User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
Pneumonia decision tool reduces death in ED patients
a 3-year, pragmatic, cluster-controlled study shows.
“We designed the ePNa specifically to require minimal input from the clinician so everything it does is already in the electronic medical record,” Nathan Dean, MD, University of Utah, Salt Lake City, told this news organization.
“So it’s actually putting the guideline recommendations into effect for physicians so that they can make better decisions by having all this information – it’s a comprehensive best practice kind of tool where best practices are likely to make the biggest difference for patients with a high severity of illness,” he added.
The study was published online in the American Journal of Respiratory and Critical Care Medicine.
Guideline-based tool
The ePNa makes use of pneumonia guidelines of 2007 and 2019 from the American Thoracic Society/Infectious Disease Society of America. The system was deployed into six geographic clusters of 16 Intermountain hospital EDs at 2-month intervals between December 2017 and November 2018. Simultaneous deployment was impractical, as implementation of the tool takes education, monitoring, and feedback that can be facilitated by focusing on only a few hospitals at a time.
The decision support tool gathers key patient indicators including age, fever, oxygen saturation, vital signs, and laboratory and chest imaging results to offer recommendations on care, including appropriate antibiotic therapy, microbiology studies, and whether a given patient should be sent to the intensive care unit, admitted to hospital, or may safely be discharged home.
Investigators analyzed a total of 6,848 patients, of whom 4,536 were managed for pneumonia before the ePNa was deployed and 2,312 after deployment.
The median age of patients was 67 years (interquartile range, 50-79 years). Roughly half were female and almost all were White. “Observed 30-day all-cause mortality including both outpatients and inpatients was 8.6% before deployment versus 4.8% after deployment of ePNa,” Dr. Dean and colleagues reported.
Adjusted for severity of illness, the odds ratio for lower mortality post-ePNa launch was 0.62 (95% confidence interval, 0.49-0.79; P < .0010) “and lower morality was consistent across hospital clusters.”
Compared with patients who were discharged home, reductions in mortality were greatest in patients who were directly admitted to ICUs from the ED (OR, 0.32; 95% CI, 0.14-0.77; P = .01). The OR for patients admitted to the medical floor was 0.53 (95% CI, 0.25-1.1; P = .09), which did not reach statistical significance.
Dr. Dean explained that the reductions in mortality were seen among those with the most severe illness, in whom best practices would benefit the most. In contrast, patients who are sent home on an antibiotic are at low risk for mortality while patients admitted to the medical floor may well have another, more lethal illness from which they end up dying, rather than simple pneumonia.
“For me, this was a clear demonstration that these best practices made the biggest difference in patients who were sick and who did not have any underlying disease that was going to kill them anyway,” he emphasized. On the other hand, both 30-day mortality and 7-day secondary hospital admission were higher among patients the tool recommended for hospital ward admission but who were discharged home from the ED.
“This was an unexpected finding,” Dr. Dean observed. However, as he explained, the authors reviewed 25% of randomly selected patients who fell into this subgroup and discovered that the ePNa tool was used in only about 20% of patients – “so doctors did not use the tool in the majority of this group.”
In addition, some of these patients declined hospital admission, so the doctors may have recommended that they be admitted but the patients said no. “The hypothesis here is that if they had been admitted to the hospital, they may have had a lower mortality risk,” Dr. Dean said.
Noticeable changes
Another noticeable change following the introduction of the ePNa tool was that guideline-concordant antibiotic prescribing increased in the 8 hours after patients presented to the ED, from 79.5% prior to the tool’s launch to 87.9%, again after adjusting for pneumonia severity (P < .001). Use of broad-spectrum antibiotics was not significantly different between the two treatment intervals, but administration of antibiotics active against methicillin-resistant Staphylococcus aureus dropped significantly between the two treatment intervals (P < .001). And the mean time from admission to the ED to the first antibiotic taken was slightly faster, improving from 159.4 minutes (95% CI, 156.9-161.9 minutes) prior to the ePNa launch to 150.9 minutes (95% CI, 144.1-157.8) post deployment (P < .001).
“Overall outpatient disposition for treatment of pneumonia from the emergency department increased from 29.2% before ePNa to 46.9% [post ePNA],” the authors noted, while a similar increase was observed in patients for whom ePNA recommended outpatient care – from 49.2% pre-ePNA to 66.6% after ePNA.
Both hospital ward admission and admission to the ICU decreased after ePNa had been introduced. Despite a significant increase in the percentage of patients being discharged home, neither 7-day secondary hospital admission nor severity-adjusted, 30-day mortality were significantly different before versus after the introduction of ePNa, the authors stressed.
A limitation of the study was that the trial was confined to a single health care system in one region of the United States with a patient population that may differ from that in other regions.
Reason for its success
Asked to comment on the findings, Adam Balls, MD, emergency department chair, Intermountain Medical Center, Murray, Utah, suggested that the reason the ePNa tool has been so successful at improving care for pneumonia patients is that it puts the guidelines directly into the hands of individual providers and tells them what’s going on. (Dr. Balls was not involved in the study.) “The tool allows us to take into consideration various clinical features – a patient’s oxygen requirements and whether or not they had prior complicated pneumonias that required additional antibiotics, for example – and then it makes the best determination for not only the disposition for that patient but antibiotic treatment as well,” he said in an interview.
This then allows physicians to either appropriately discharge less severely ill patients and admit those who are more ill – “and in general, just do a better job of treating pneumonia with this tool,” Dr. Balls said. He himself uses the decision support tool when attending to his own patients with pneumonia, as he feels that the tool really does make his care of these patients better. “There is a disparity around how we treat pneumonia in the U.S.
“Clinicians sometimes have a bias or a preference for certain antibiotics and we may not be appropriately treating these patients with broad-spectrum antibiotics or are perhaps using antibiotics that are not as effective based on an individual patient scenario so this is definitely a user-friendly tool that hopefully can be deployed throughout other health care systems to improve the treatment of pneumonia overall,” Dr. Balls emphasized.
A version of this article first appeared on Medscape.com.
a 3-year, pragmatic, cluster-controlled study shows.
“We designed the ePNa specifically to require minimal input from the clinician so everything it does is already in the electronic medical record,” Nathan Dean, MD, University of Utah, Salt Lake City, told this news organization.
“So it’s actually putting the guideline recommendations into effect for physicians so that they can make better decisions by having all this information – it’s a comprehensive best practice kind of tool where best practices are likely to make the biggest difference for patients with a high severity of illness,” he added.
The study was published online in the American Journal of Respiratory and Critical Care Medicine.
Guideline-based tool
The ePNa makes use of pneumonia guidelines of 2007 and 2019 from the American Thoracic Society/Infectious Disease Society of America. The system was deployed into six geographic clusters of 16 Intermountain hospital EDs at 2-month intervals between December 2017 and November 2018. Simultaneous deployment was impractical, as implementation of the tool takes education, monitoring, and feedback that can be facilitated by focusing on only a few hospitals at a time.
The decision support tool gathers key patient indicators including age, fever, oxygen saturation, vital signs, and laboratory and chest imaging results to offer recommendations on care, including appropriate antibiotic therapy, microbiology studies, and whether a given patient should be sent to the intensive care unit, admitted to hospital, or may safely be discharged home.
Investigators analyzed a total of 6,848 patients, of whom 4,536 were managed for pneumonia before the ePNa was deployed and 2,312 after deployment.
The median age of patients was 67 years (interquartile range, 50-79 years). Roughly half were female and almost all were White. “Observed 30-day all-cause mortality including both outpatients and inpatients was 8.6% before deployment versus 4.8% after deployment of ePNa,” Dr. Dean and colleagues reported.
Adjusted for severity of illness, the odds ratio for lower mortality post-ePNa launch was 0.62 (95% confidence interval, 0.49-0.79; P < .0010) “and lower morality was consistent across hospital clusters.”
Compared with patients who were discharged home, reductions in mortality were greatest in patients who were directly admitted to ICUs from the ED (OR, 0.32; 95% CI, 0.14-0.77; P = .01). The OR for patients admitted to the medical floor was 0.53 (95% CI, 0.25-1.1; P = .09), which did not reach statistical significance.
Dr. Dean explained that the reductions in mortality were seen among those with the most severe illness, in whom best practices would benefit the most. In contrast, patients who are sent home on an antibiotic are at low risk for mortality while patients admitted to the medical floor may well have another, more lethal illness from which they end up dying, rather than simple pneumonia.
“For me, this was a clear demonstration that these best practices made the biggest difference in patients who were sick and who did not have any underlying disease that was going to kill them anyway,” he emphasized. On the other hand, both 30-day mortality and 7-day secondary hospital admission were higher among patients the tool recommended for hospital ward admission but who were discharged home from the ED.
“This was an unexpected finding,” Dr. Dean observed. However, as he explained, the authors reviewed 25% of randomly selected patients who fell into this subgroup and discovered that the ePNa tool was used in only about 20% of patients – “so doctors did not use the tool in the majority of this group.”
In addition, some of these patients declined hospital admission, so the doctors may have recommended that they be admitted but the patients said no. “The hypothesis here is that if they had been admitted to the hospital, they may have had a lower mortality risk,” Dr. Dean said.
Noticeable changes
Another noticeable change following the introduction of the ePNa tool was that guideline-concordant antibiotic prescribing increased in the 8 hours after patients presented to the ED, from 79.5% prior to the tool’s launch to 87.9%, again after adjusting for pneumonia severity (P < .001). Use of broad-spectrum antibiotics was not significantly different between the two treatment intervals, but administration of antibiotics active against methicillin-resistant Staphylococcus aureus dropped significantly between the two treatment intervals (P < .001). And the mean time from admission to the ED to the first antibiotic taken was slightly faster, improving from 159.4 minutes (95% CI, 156.9-161.9 minutes) prior to the ePNa launch to 150.9 minutes (95% CI, 144.1-157.8) post deployment (P < .001).
“Overall outpatient disposition for treatment of pneumonia from the emergency department increased from 29.2% before ePNa to 46.9% [post ePNA],” the authors noted, while a similar increase was observed in patients for whom ePNA recommended outpatient care – from 49.2% pre-ePNA to 66.6% after ePNA.
Both hospital ward admission and admission to the ICU decreased after ePNa had been introduced. Despite a significant increase in the percentage of patients being discharged home, neither 7-day secondary hospital admission nor severity-adjusted, 30-day mortality were significantly different before versus after the introduction of ePNa, the authors stressed.
A limitation of the study was that the trial was confined to a single health care system in one region of the United States with a patient population that may differ from that in other regions.
Reason for its success
Asked to comment on the findings, Adam Balls, MD, emergency department chair, Intermountain Medical Center, Murray, Utah, suggested that the reason the ePNa tool has been so successful at improving care for pneumonia patients is that it puts the guidelines directly into the hands of individual providers and tells them what’s going on. (Dr. Balls was not involved in the study.) “The tool allows us to take into consideration various clinical features – a patient’s oxygen requirements and whether or not they had prior complicated pneumonias that required additional antibiotics, for example – and then it makes the best determination for not only the disposition for that patient but antibiotic treatment as well,” he said in an interview.
This then allows physicians to either appropriately discharge less severely ill patients and admit those who are more ill – “and in general, just do a better job of treating pneumonia with this tool,” Dr. Balls said. He himself uses the decision support tool when attending to his own patients with pneumonia, as he feels that the tool really does make his care of these patients better. “There is a disparity around how we treat pneumonia in the U.S.
“Clinicians sometimes have a bias or a preference for certain antibiotics and we may not be appropriately treating these patients with broad-spectrum antibiotics or are perhaps using antibiotics that are not as effective based on an individual patient scenario so this is definitely a user-friendly tool that hopefully can be deployed throughout other health care systems to improve the treatment of pneumonia overall,” Dr. Balls emphasized.
A version of this article first appeared on Medscape.com.
a 3-year, pragmatic, cluster-controlled study shows.
“We designed the ePNa specifically to require minimal input from the clinician so everything it does is already in the electronic medical record,” Nathan Dean, MD, University of Utah, Salt Lake City, told this news organization.
“So it’s actually putting the guideline recommendations into effect for physicians so that they can make better decisions by having all this information – it’s a comprehensive best practice kind of tool where best practices are likely to make the biggest difference for patients with a high severity of illness,” he added.
The study was published online in the American Journal of Respiratory and Critical Care Medicine.
Guideline-based tool
The ePNa makes use of pneumonia guidelines of 2007 and 2019 from the American Thoracic Society/Infectious Disease Society of America. The system was deployed into six geographic clusters of 16 Intermountain hospital EDs at 2-month intervals between December 2017 and November 2018. Simultaneous deployment was impractical, as implementation of the tool takes education, monitoring, and feedback that can be facilitated by focusing on only a few hospitals at a time.
The decision support tool gathers key patient indicators including age, fever, oxygen saturation, vital signs, and laboratory and chest imaging results to offer recommendations on care, including appropriate antibiotic therapy, microbiology studies, and whether a given patient should be sent to the intensive care unit, admitted to hospital, or may safely be discharged home.
Investigators analyzed a total of 6,848 patients, of whom 4,536 were managed for pneumonia before the ePNa was deployed and 2,312 after deployment.
The median age of patients was 67 years (interquartile range, 50-79 years). Roughly half were female and almost all were White. “Observed 30-day all-cause mortality including both outpatients and inpatients was 8.6% before deployment versus 4.8% after deployment of ePNa,” Dr. Dean and colleagues reported.
Adjusted for severity of illness, the odds ratio for lower mortality post-ePNa launch was 0.62 (95% confidence interval, 0.49-0.79; P < .0010) “and lower morality was consistent across hospital clusters.”
Compared with patients who were discharged home, reductions in mortality were greatest in patients who were directly admitted to ICUs from the ED (OR, 0.32; 95% CI, 0.14-0.77; P = .01). The OR for patients admitted to the medical floor was 0.53 (95% CI, 0.25-1.1; P = .09), which did not reach statistical significance.
Dr. Dean explained that the reductions in mortality were seen among those with the most severe illness, in whom best practices would benefit the most. In contrast, patients who are sent home on an antibiotic are at low risk for mortality while patients admitted to the medical floor may well have another, more lethal illness from which they end up dying, rather than simple pneumonia.
“For me, this was a clear demonstration that these best practices made the biggest difference in patients who were sick and who did not have any underlying disease that was going to kill them anyway,” he emphasized. On the other hand, both 30-day mortality and 7-day secondary hospital admission were higher among patients the tool recommended for hospital ward admission but who were discharged home from the ED.
“This was an unexpected finding,” Dr. Dean observed. However, as he explained, the authors reviewed 25% of randomly selected patients who fell into this subgroup and discovered that the ePNa tool was used in only about 20% of patients – “so doctors did not use the tool in the majority of this group.”
In addition, some of these patients declined hospital admission, so the doctors may have recommended that they be admitted but the patients said no. “The hypothesis here is that if they had been admitted to the hospital, they may have had a lower mortality risk,” Dr. Dean said.
Noticeable changes
Another noticeable change following the introduction of the ePNa tool was that guideline-concordant antibiotic prescribing increased in the 8 hours after patients presented to the ED, from 79.5% prior to the tool’s launch to 87.9%, again after adjusting for pneumonia severity (P < .001). Use of broad-spectrum antibiotics was not significantly different between the two treatment intervals, but administration of antibiotics active against methicillin-resistant Staphylococcus aureus dropped significantly between the two treatment intervals (P < .001). And the mean time from admission to the ED to the first antibiotic taken was slightly faster, improving from 159.4 minutes (95% CI, 156.9-161.9 minutes) prior to the ePNa launch to 150.9 minutes (95% CI, 144.1-157.8) post deployment (P < .001).
“Overall outpatient disposition for treatment of pneumonia from the emergency department increased from 29.2% before ePNa to 46.9% [post ePNA],” the authors noted, while a similar increase was observed in patients for whom ePNA recommended outpatient care – from 49.2% pre-ePNA to 66.6% after ePNA.
Both hospital ward admission and admission to the ICU decreased after ePNa had been introduced. Despite a significant increase in the percentage of patients being discharged home, neither 7-day secondary hospital admission nor severity-adjusted, 30-day mortality were significantly different before versus after the introduction of ePNa, the authors stressed.
A limitation of the study was that the trial was confined to a single health care system in one region of the United States with a patient population that may differ from that in other regions.
Reason for its success
Asked to comment on the findings, Adam Balls, MD, emergency department chair, Intermountain Medical Center, Murray, Utah, suggested that the reason the ePNa tool has been so successful at improving care for pneumonia patients is that it puts the guidelines directly into the hands of individual providers and tells them what’s going on. (Dr. Balls was not involved in the study.) “The tool allows us to take into consideration various clinical features – a patient’s oxygen requirements and whether or not they had prior complicated pneumonias that required additional antibiotics, for example – and then it makes the best determination for not only the disposition for that patient but antibiotic treatment as well,” he said in an interview.
This then allows physicians to either appropriately discharge less severely ill patients and admit those who are more ill – “and in general, just do a better job of treating pneumonia with this tool,” Dr. Balls said. He himself uses the decision support tool when attending to his own patients with pneumonia, as he feels that the tool really does make his care of these patients better. “There is a disparity around how we treat pneumonia in the U.S.
“Clinicians sometimes have a bias or a preference for certain antibiotics and we may not be appropriately treating these patients with broad-spectrum antibiotics or are perhaps using antibiotics that are not as effective based on an individual patient scenario so this is definitely a user-friendly tool that hopefully can be deployed throughout other health care systems to improve the treatment of pneumonia overall,” Dr. Balls emphasized.
A version of this article first appeared on Medscape.com.
FROM THE AMERICAN JOURNAL OF RESPIRATORY AND CRITICAL CARE MEDICINE
‘Profound implications’: COVID ups diabetes risk 40% a year later
COVID-19 infection appears to significantly raise the risk for diabetes by about 40% at 1 year, indicate new data from a very large Veterans Administration population.
“If patients have a prior history of COVID-19, that’s a risk factor for diabetes and they should certainly be screened for diabetes,” study coauthor Ziyad Al-Aly, MD, a nephrologist and chief of research and development at VA St. Louis Health Care, told this news organization.
“It’s still premature to make guidelines. I think we have to process the data landscape to understand what this all really means, but it’s really, really clear that all these roads are pointing in one direction, that COVID-19 increases the risk of diabetes up to a year later. The risk is small but not negligible,” he said.
The database includes over 8 million people and 180,000 with a prior COVID-19 diagnosis. Significantly increased diabetes risks compared to those not infected ranging from 31% to more than double were found in an analysis of subgroups based on diabetes risk score, body mass index, age, race, prediabetes status, and deprivation level, even after adjustment for confounding factors.
There was a gradient of diabetes risk by COVID-19 severity – i.e., whether patients had not been hospitalized, had been hospitalized, or stayed in intensive care – but a significant excess diabetes burden was seen even among those with “mild” COVID-19. The diabetes risk was also elevated compared to both contemporary and historical controls.
The study was published March 21 in The Lancet Diabetes & Endocrinology, by Yan Xie, MPH, also of VA St Louis Health Care, along with Dr. Al-Aly.
The data align with those from another study just published from a nationwide German primary care database. That study was smaller and of shorter duration than the new VA study but consistent, said Dr. Al-Aly, a clinical epidemiologist at Washington University, St. Louis.
Millions more with new diabetes as late manifestation of COVID-19
“Millions of people in the U.S. have had COVID-19, so this is going to translate to literally millions more people with new-onset diabetes. Better to identify them early so they can be adequately treated,” Dr. Al-Aly said in an interview.
“The long-term implications of SARS-CoV-2 infection increasing diabetes risk are profound,” Venkat Narayan, MD, and Lisa R. Staimez, PhD, both of the Rollins School of Public Health and Emory Global Diabetes Research Center at Emory University, Atlanta, said in an accompanying editorial.
“With large and growing numbers of people worldwide infected with SARS-CoV-2 (434,154,739 cumulative cases by Feb. 28, 2022), any COVID-19-related increases in diabetes incidence could lead to unprecedented cases of diabetes worldwide – wreaking havoc on already over-stretched and under-resourced clinical and public health systems globally, with devastating tolls in terms of deaths and suffering,” they added.
Medscape Medical News contributor Eric Topol MD, of Scripps Research Institute, La Jolla, Calif., agrees. He said these new data “are most profound. The researchers found a 40% increase in diabetes that wasn’t present at 1 month after COVID-19 but at 1 year, it was. Some kind of late manifestation is happening here.”
Dr. Al-Aly told this news organization that the mechanisms for the association are unknown and likely to be heterogeneous. Among the people who already had risk factors for type 2 diabetes, such as obesity or metabolic syndrome, SARS-CoV-2 could simply accelerate that process and “put them over the edge” to overt diabetes.
However, for those without diabetes risk factors, “COVID-19 with all the inflammation it provokes in the body could be leading to de novo disease.” (Diabetes status was ascertained by ICD-10 codes and only about 0.70% of the total were recorded as type 1 diabetes. But, since autoantibody testing wasn’t routinely conducted, it’s unknown how many of the cases may have been type 1 misclassified as type 2, Dr. Al-Aly acknowledged.)
Diabetes risk significantly increased after COVID-19 in all analyses
The analysis included 181,280 patients in the U.S. Department of Veterans Affairs health care database with a COVID-19 diagnosis who survived for at least 30 days afterward during March 2020 through Sept. 30, 2021, with 4,118,441 contemporary controls without COVID-19 seen during 2019, and a historical control group of 4,286,911 people seen at the VA in 2017. Average follow-up was about a year.
Compared with the contemporary controls, the COVID-19 group had an excess diabetes burden of 13.46 per 1,000 person-years with a hazard ratio of 1.40. They had an increased 12.35 per 1,000 person-year risk for incident use of glucose-lowering medications, with a hazard ratio of 1.85. Similar results were seen with the historical controls.
Subgroup analyses showed an increased risk for diabetes following COVID-19 infection by age (≤ 65 years and > 65 years), race (White and Black), sex (male and female), BMI categories (> 18.5 to ≤ 25 kg/m², > 25 to ≤ 30 kg/m², and > 30 kg/m²), and area deprivation index quartiles. The increased risk was also seen across diabetes risk score quartiles.
Notably, COVID-19 significantly elevated the diabetes risk by 59% even for the subgroup with BMI between 18 and 25 kg/m², and by 38% among those with the lowest diabetes risk score quartile.
The COVID-19 population included 162,096 who were not hospitalized, 15,078 hospitalized, and 4,106 admitted to intensive care. Here, the hazard ratios for diabetes compared to the contemporary controls were 1.25, 2.73, and 3.76, respectively, all significant.
Dr. Al-Aly said that his group is now further analyzing the VA data for other outcomes including cardiovascular disease and kidney disease, as well as the now well-documented long COVID symptoms including fatigue, pain, and neurocognitive dysfunction.
They’re also investigating the impact of the COVID-19 vaccine to see whether the risks are mitigated in the case of breakthrough infections: “We’re doing a broad systematic assessment. The next paper will be more comprehensive.”
Dr. Narayan and Dr. Staimez wrote: “The potential connection between COVID-19 and diabetes highlights that infectious diseases (eg, SARS-CoV-2) and chronic diseases (eg, diabetes) cannot be viewed in siloes. When we emerge out of the pandemic, the much-neglected non-communicable diseases, such as type 2 diabetes, will continue their relentless trajectory, possibly in an accelerated manner, as the leading burdens of global health.”
Dr. Al-Aly declared support from the U.S. Department of Veterans Affairs for the submitted work. He has received consultation fees from Gilead Sciences and funding (unrelated to this work) from Tonix Pharmaceuticals. He is a member of the board of directors for Veterans Research and Education Foundation of Saint Louis, associate editor for the Journal of the American Society of Nephrology, and a member of multiple editorial boards. Dr. Narayan and Dr. Staimez have received support from the National Institutes of Health.
A version of this article first appeared on Medscape.com.
COVID-19 infection appears to significantly raise the risk for diabetes by about 40% at 1 year, indicate new data from a very large Veterans Administration population.
“If patients have a prior history of COVID-19, that’s a risk factor for diabetes and they should certainly be screened for diabetes,” study coauthor Ziyad Al-Aly, MD, a nephrologist and chief of research and development at VA St. Louis Health Care, told this news organization.
“It’s still premature to make guidelines. I think we have to process the data landscape to understand what this all really means, but it’s really, really clear that all these roads are pointing in one direction, that COVID-19 increases the risk of diabetes up to a year later. The risk is small but not negligible,” he said.
The database includes over 8 million people and 180,000 with a prior COVID-19 diagnosis. Significantly increased diabetes risks compared to those not infected ranging from 31% to more than double were found in an analysis of subgroups based on diabetes risk score, body mass index, age, race, prediabetes status, and deprivation level, even after adjustment for confounding factors.
There was a gradient of diabetes risk by COVID-19 severity – i.e., whether patients had not been hospitalized, had been hospitalized, or stayed in intensive care – but a significant excess diabetes burden was seen even among those with “mild” COVID-19. The diabetes risk was also elevated compared to both contemporary and historical controls.
The study was published March 21 in The Lancet Diabetes & Endocrinology, by Yan Xie, MPH, also of VA St Louis Health Care, along with Dr. Al-Aly.
The data align with those from another study just published from a nationwide German primary care database. That study was smaller and of shorter duration than the new VA study but consistent, said Dr. Al-Aly, a clinical epidemiologist at Washington University, St. Louis.
Millions more with new diabetes as late manifestation of COVID-19
“Millions of people in the U.S. have had COVID-19, so this is going to translate to literally millions more people with new-onset diabetes. Better to identify them early so they can be adequately treated,” Dr. Al-Aly said in an interview.
“The long-term implications of SARS-CoV-2 infection increasing diabetes risk are profound,” Venkat Narayan, MD, and Lisa R. Staimez, PhD, both of the Rollins School of Public Health and Emory Global Diabetes Research Center at Emory University, Atlanta, said in an accompanying editorial.
“With large and growing numbers of people worldwide infected with SARS-CoV-2 (434,154,739 cumulative cases by Feb. 28, 2022), any COVID-19-related increases in diabetes incidence could lead to unprecedented cases of diabetes worldwide – wreaking havoc on already over-stretched and under-resourced clinical and public health systems globally, with devastating tolls in terms of deaths and suffering,” they added.
Medscape Medical News contributor Eric Topol MD, of Scripps Research Institute, La Jolla, Calif., agrees. He said these new data “are most profound. The researchers found a 40% increase in diabetes that wasn’t present at 1 month after COVID-19 but at 1 year, it was. Some kind of late manifestation is happening here.”
Dr. Al-Aly told this news organization that the mechanisms for the association are unknown and likely to be heterogeneous. Among the people who already had risk factors for type 2 diabetes, such as obesity or metabolic syndrome, SARS-CoV-2 could simply accelerate that process and “put them over the edge” to overt diabetes.
However, for those without diabetes risk factors, “COVID-19 with all the inflammation it provokes in the body could be leading to de novo disease.” (Diabetes status was ascertained by ICD-10 codes and only about 0.70% of the total were recorded as type 1 diabetes. But, since autoantibody testing wasn’t routinely conducted, it’s unknown how many of the cases may have been type 1 misclassified as type 2, Dr. Al-Aly acknowledged.)
Diabetes risk significantly increased after COVID-19 in all analyses
The analysis included 181,280 patients in the U.S. Department of Veterans Affairs health care database with a COVID-19 diagnosis who survived for at least 30 days afterward during March 2020 through Sept. 30, 2021, with 4,118,441 contemporary controls without COVID-19 seen during 2019, and a historical control group of 4,286,911 people seen at the VA in 2017. Average follow-up was about a year.
Compared with the contemporary controls, the COVID-19 group had an excess diabetes burden of 13.46 per 1,000 person-years with a hazard ratio of 1.40. They had an increased 12.35 per 1,000 person-year risk for incident use of glucose-lowering medications, with a hazard ratio of 1.85. Similar results were seen with the historical controls.
Subgroup analyses showed an increased risk for diabetes following COVID-19 infection by age (≤ 65 years and > 65 years), race (White and Black), sex (male and female), BMI categories (> 18.5 to ≤ 25 kg/m², > 25 to ≤ 30 kg/m², and > 30 kg/m²), and area deprivation index quartiles. The increased risk was also seen across diabetes risk score quartiles.
Notably, COVID-19 significantly elevated the diabetes risk by 59% even for the subgroup with BMI between 18 and 25 kg/m², and by 38% among those with the lowest diabetes risk score quartile.
The COVID-19 population included 162,096 who were not hospitalized, 15,078 hospitalized, and 4,106 admitted to intensive care. Here, the hazard ratios for diabetes compared to the contemporary controls were 1.25, 2.73, and 3.76, respectively, all significant.
Dr. Al-Aly said that his group is now further analyzing the VA data for other outcomes including cardiovascular disease and kidney disease, as well as the now well-documented long COVID symptoms including fatigue, pain, and neurocognitive dysfunction.
They’re also investigating the impact of the COVID-19 vaccine to see whether the risks are mitigated in the case of breakthrough infections: “We’re doing a broad systematic assessment. The next paper will be more comprehensive.”
Dr. Narayan and Dr. Staimez wrote: “The potential connection between COVID-19 and diabetes highlights that infectious diseases (eg, SARS-CoV-2) and chronic diseases (eg, diabetes) cannot be viewed in siloes. When we emerge out of the pandemic, the much-neglected non-communicable diseases, such as type 2 diabetes, will continue their relentless trajectory, possibly in an accelerated manner, as the leading burdens of global health.”
Dr. Al-Aly declared support from the U.S. Department of Veterans Affairs for the submitted work. He has received consultation fees from Gilead Sciences and funding (unrelated to this work) from Tonix Pharmaceuticals. He is a member of the board of directors for Veterans Research and Education Foundation of Saint Louis, associate editor for the Journal of the American Society of Nephrology, and a member of multiple editorial boards. Dr. Narayan and Dr. Staimez have received support from the National Institutes of Health.
A version of this article first appeared on Medscape.com.
COVID-19 infection appears to significantly raise the risk for diabetes by about 40% at 1 year, indicate new data from a very large Veterans Administration population.
“If patients have a prior history of COVID-19, that’s a risk factor for diabetes and they should certainly be screened for diabetes,” study coauthor Ziyad Al-Aly, MD, a nephrologist and chief of research and development at VA St. Louis Health Care, told this news organization.
“It’s still premature to make guidelines. I think we have to process the data landscape to understand what this all really means, but it’s really, really clear that all these roads are pointing in one direction, that COVID-19 increases the risk of diabetes up to a year later. The risk is small but not negligible,” he said.
The database includes over 8 million people and 180,000 with a prior COVID-19 diagnosis. Significantly increased diabetes risks compared to those not infected ranging from 31% to more than double were found in an analysis of subgroups based on diabetes risk score, body mass index, age, race, prediabetes status, and deprivation level, even after adjustment for confounding factors.
There was a gradient of diabetes risk by COVID-19 severity – i.e., whether patients had not been hospitalized, had been hospitalized, or stayed in intensive care – but a significant excess diabetes burden was seen even among those with “mild” COVID-19. The diabetes risk was also elevated compared to both contemporary and historical controls.
The study was published March 21 in The Lancet Diabetes & Endocrinology, by Yan Xie, MPH, also of VA St Louis Health Care, along with Dr. Al-Aly.
The data align with those from another study just published from a nationwide German primary care database. That study was smaller and of shorter duration than the new VA study but consistent, said Dr. Al-Aly, a clinical epidemiologist at Washington University, St. Louis.
Millions more with new diabetes as late manifestation of COVID-19
“Millions of people in the U.S. have had COVID-19, so this is going to translate to literally millions more people with new-onset diabetes. Better to identify them early so they can be adequately treated,” Dr. Al-Aly said in an interview.
“The long-term implications of SARS-CoV-2 infection increasing diabetes risk are profound,” Venkat Narayan, MD, and Lisa R. Staimez, PhD, both of the Rollins School of Public Health and Emory Global Diabetes Research Center at Emory University, Atlanta, said in an accompanying editorial.
“With large and growing numbers of people worldwide infected with SARS-CoV-2 (434,154,739 cumulative cases by Feb. 28, 2022), any COVID-19-related increases in diabetes incidence could lead to unprecedented cases of diabetes worldwide – wreaking havoc on already over-stretched and under-resourced clinical and public health systems globally, with devastating tolls in terms of deaths and suffering,” they added.
Medscape Medical News contributor Eric Topol MD, of Scripps Research Institute, La Jolla, Calif., agrees. He said these new data “are most profound. The researchers found a 40% increase in diabetes that wasn’t present at 1 month after COVID-19 but at 1 year, it was. Some kind of late manifestation is happening here.”
Dr. Al-Aly told this news organization that the mechanisms for the association are unknown and likely to be heterogeneous. Among the people who already had risk factors for type 2 diabetes, such as obesity or metabolic syndrome, SARS-CoV-2 could simply accelerate that process and “put them over the edge” to overt diabetes.
However, for those without diabetes risk factors, “COVID-19 with all the inflammation it provokes in the body could be leading to de novo disease.” (Diabetes status was ascertained by ICD-10 codes and only about 0.70% of the total were recorded as type 1 diabetes. But, since autoantibody testing wasn’t routinely conducted, it’s unknown how many of the cases may have been type 1 misclassified as type 2, Dr. Al-Aly acknowledged.)
Diabetes risk significantly increased after COVID-19 in all analyses
The analysis included 181,280 patients in the U.S. Department of Veterans Affairs health care database with a COVID-19 diagnosis who survived for at least 30 days afterward during March 2020 through Sept. 30, 2021, with 4,118,441 contemporary controls without COVID-19 seen during 2019, and a historical control group of 4,286,911 people seen at the VA in 2017. Average follow-up was about a year.
Compared with the contemporary controls, the COVID-19 group had an excess diabetes burden of 13.46 per 1,000 person-years with a hazard ratio of 1.40. They had an increased 12.35 per 1,000 person-year risk for incident use of glucose-lowering medications, with a hazard ratio of 1.85. Similar results were seen with the historical controls.
Subgroup analyses showed an increased risk for diabetes following COVID-19 infection by age (≤ 65 years and > 65 years), race (White and Black), sex (male and female), BMI categories (> 18.5 to ≤ 25 kg/m², > 25 to ≤ 30 kg/m², and > 30 kg/m²), and area deprivation index quartiles. The increased risk was also seen across diabetes risk score quartiles.
Notably, COVID-19 significantly elevated the diabetes risk by 59% even for the subgroup with BMI between 18 and 25 kg/m², and by 38% among those with the lowest diabetes risk score quartile.
The COVID-19 population included 162,096 who were not hospitalized, 15,078 hospitalized, and 4,106 admitted to intensive care. Here, the hazard ratios for diabetes compared to the contemporary controls were 1.25, 2.73, and 3.76, respectively, all significant.
Dr. Al-Aly said that his group is now further analyzing the VA data for other outcomes including cardiovascular disease and kidney disease, as well as the now well-documented long COVID symptoms including fatigue, pain, and neurocognitive dysfunction.
They’re also investigating the impact of the COVID-19 vaccine to see whether the risks are mitigated in the case of breakthrough infections: “We’re doing a broad systematic assessment. The next paper will be more comprehensive.”
Dr. Narayan and Dr. Staimez wrote: “The potential connection between COVID-19 and diabetes highlights that infectious diseases (eg, SARS-CoV-2) and chronic diseases (eg, diabetes) cannot be viewed in siloes. When we emerge out of the pandemic, the much-neglected non-communicable diseases, such as type 2 diabetes, will continue their relentless trajectory, possibly in an accelerated manner, as the leading burdens of global health.”
Dr. Al-Aly declared support from the U.S. Department of Veterans Affairs for the submitted work. He has received consultation fees from Gilead Sciences and funding (unrelated to this work) from Tonix Pharmaceuticals. He is a member of the board of directors for Veterans Research and Education Foundation of Saint Louis, associate editor for the Journal of the American Society of Nephrology, and a member of multiple editorial boards. Dr. Narayan and Dr. Staimez have received support from the National Institutes of Health.
A version of this article first appeared on Medscape.com.
FROM THE LANCET DIABETES & ENDOCRINOLOGY
Are ‘antibiotic diets’ good practice?
Antibiotics are among the most commonly prescribed medications in both outpatient and hospital settings. Global efforts at curbing antibiotic-resistant strains have prompted clinicians to pursue better stewardship, whereby they limit their prescribing of such medications to those who truly need them.
Yet there’s another possible means of addressing antibiotic resistance – using dietary interventions to reduce the gastrointestinal complaints that so often accompany the use of antibiotics: vomiting, nausea, diarrhea, bloating/indigestion, abdominal pain, and loss of appetite. Far from being a mere nuisance, these complications can have major ramifications.
“Often [these side effects] will result in people stopping an antibiotic they need or taking a second- or third-generation one, which potentially causes even more problems with resistance,” explained Daniel J. Merenstein, MD, a professor of family medicine at Georgetown University, Washington, who has conducted multiple trials on antibiotic stewardship and probiotics.
And it’s not just clinicians who would like to find a way around these common complaints. On lifestyle blogs such as Livestrong and Goop, medical news websites, and via academic institutions, patients can learn how the right probiotic supplement or certain fermented or high-fiber foods, for example, might spare them the upset stomach that often accompanies a course of antibiotics.
Yet according to experts in the field, there are notable questions to be answered about whether there’s evidence to support this approach.
The cost of GI discomfort
Pinpointing the exact manner by which antibiotics upend GI tracts is a complex task, according to Gail Cresci, PhD, RD, a microbiome researcher in the department of pediatric gastroenterology, hepatology, and nutrition and director of nutrition research at the Cleveland Clinic.
“A lot of different mechanisms can go awry,” Dr. Cresci said. “But normally, the good microbes are there to help keep the balance, so when that balance is gone, the pathogens can take over and lead to this disruption.”
According to Lynne V. McFarland, PhD, a recently retired infectious disease epidemiologist who now serves as a private consultant, such complications can have substantial ramifications for patients and health care systems.
“If you’re an inpatient and develop antibiotic-associated diarrhea, it usually increases the length of stay from 8 to 20 days. It also increases the cost of care. And small children who have this can get severely dehydrated, which can become life threatening.”
Proponents of probiotics
Several of the researchers who were interviewed believe there’s convincing evidence supporting probiotics for the treatment of common antibiotic-related GI complaints. In many instances, they were involved in the studies themselves.
During Dr. McFarland’s 4-decade career in probiotics research, she has participated in early animal studies with strains such as Saccharomyces boulardii and was involved in meta-analyses of their role in Clostridioides difficile infection and associated diarrhea and even as a potential GI intervention for COVID-19 patients (Antibiotics (Basel). 2021 Apr 9;10(4):408).
In mouse model studies from 2013 and 2018, Dr. Cresci and colleagues showed that the probiotic strains Lactobacillus GG and Faecalibacterium prausnitzii reduced the structural gut changes that lead to antibiotic-associated diarrhea and minimized the risk of C. difficile infection.
In a 2021 randomized controlled trial led by Merenstein, healthy participants were given a trial of amoxicillin/clavulanate (days 1-7) in conjunction with either yogurt containing the probiotic Bifidobacterium animalis subsp lactis BB-12 or control yogurt (days 1-14). After assessing feces samples over a 30-day period, they found that those who received the probiotic yogurt had a significantly smaller decrease in short-chain fatty acid levels and a more stable taxonomic microbiota profile than control persons.
Dr. Merenstein said that, on the basis of results such as these and others, he’s comfortable being relatively definitive about the value of probiotics.
“I believe it’s close to standard of care that if you’re prescribing antibiotics, especially for more than 7 days, you really need to put people on probiotics that have been studied, simply because the evidence is robust enough now,” he said.
Even for proponents, there are caveats
However, all the researchers recommending the use of probiotics did so with caveats. First and foremost, they advise clinicians that the term “probiotics” is an imprecise catchall and is essentially meaningless.
“A lot of products label themselves as probiotic. It’s a great marketing scheme, but many of the products out there aren’t really probiotics; they’re not proven with randomized control trials and don’t have the scientific background,” said Dr. McFarland. “We’ve found that the efficacy is extremely strain specific and disease specific. A strain may work for one disease and not work for another.”
In 2018, Dr. McFarland coauthored an evidence-based practical guide to help clinicians and patients identify the specific strain that works in certain indications. Dr. Cresci recommends that clinicians consult websites such as Probiotics.org or the National Institutes of Health’s database to find the strains that have been proven to work in well-designed clinical trials.
There was also agreement that, to date, the most robust data support probiotics for the treatment of antibiotic-associated diarrhea.
Although the optimal timing of probiotics is a subject of debate, most proponents agreed that the general rule is “the sooner the better.”
Dr. McFarland recommended incorporating probiotics within 24 hours of starting an antibiotic “because the damage done to your GI tract microbiome is pretty quick, and the probiotics work best if they’re established before major disruption occurs.” She added that patients should continue taking probiotics for 2-8 weeks after stopping antibiotics.
“It takes a long time for your normal flora to get restored,” she said. “It’s best to cover your bases.”
For others, the evidence is not definitive
Opinions on the value of probiotics to combat antibiotic-related GI side effects are divergent, though.
“I would not recommend the routine use of probiotics, and certainly not in the prevention of C. difficile or antibiotic-related diarrhea,” said David A. Johnson, MD, professor of medicine and chief of gastroenterology at Eastern Virginia Medical School, Norfolk. “I think the evidence does not support that, and I stand strongly on that recommendation.”
Dr. Johnson cited the 2020 guidelines from the American Gastroenterological Association, which offer only a conditional recommendation for the use of specific probiotics and only in preventing antibiotic-associated C. difficile infection.
Geoffrey A. Preidis, MD, PhD, an assistant professor of pediatrics in the section of gastroenterology, hepatology, and nutrition at Baylor College of Medicine, Houston, served as a coauthor of the AGA’s guidelines. He noted that after reviewing 39 published trials of approximately 10,000 patients given probiotics while receiving antibiotics, the authors “did find some evidence that specific probiotics might decrease the risk of C. difficile diarrhea, but the quality of that evidence was low.”
Dr. Preidis attributed this to the lack of well-designed multicenter trials that can isolate the effects of certain strains and determine their benefit in this application.
“The majority of published trials have not reported safety data as rigorously as these data are reported in pharmaceutical trials, so the risk of side effects could be higher than we think,” said Dr. Preidis. “As living microbes, probiotics can move from the intestines into the bloodstream, causing sepsis. Contamination in the manufacturing process has been reported. There might be other long-term effects that we are not yet aware of.”
When asked to characterize the available data on probiotics, Dr. Johnson replied, “I’d generally label it, ‘caveat emptor.’ ”
Dr. McFarland agreed that the field would benefit from better-designed studies and called out meta-analyses that pool outcomes with various strains for particular criticism.
“When researchers do that, it’s no longer valid and shouldn’t have been published, in my opinion,” she told this news organization.
‘Antibiotic diets’ as a possible approach
A compromise between the two sides may be the so-called antibiotic diet. The theory behind such diets is that foods and beverages with biome-boosting properties may be a risk-free intervention that patients can adopt to alleviate antibiotic-related side effects.
“You want your diet to include more soluble fibers to help support the good bacteria, particularly when you’re taking antibiotics,” said Dr. Cresci. “You can get this through eating fresh fruits and vegetables, whole grains, and foods that have more prebiotic, like potatoes. You can also eat fermented food, such as kefir, kombucha, kimchi, and yogurt, so you’re adding more beneficial bacteria into your intestinal tract.”
There is ample published evidence that such foods can boost microbiome diversity and decrease inflammation, including a July 2021 study in Cell. However, the protection this may confer while taking antibiotics isn’t known. Establishing a clear role for the efficacy of such interventions is made additionally difficult by the well-established limitations of conducting dietary clinical trials.
Dr. Merenstein said that there is no compelling evidence that antibiotic-related complications can be offset by changing what goes onto our dinner plates. He joked: “We can’t say, ‘Here’s amoxicillin for your ear infection, now make sure you increase your fermented food, fiber, and water.’ ”
Dr. Johnson said he’s intrigued by studies of prebiotics – fibers that boost beneficial bacteria in the GI system.
“I would love to have more findings about prebiotic identification; that is, things we could do in a healthy way to keep the gut balanced while it’s subject to a change with antibiotics,” he said. “We’re just not there yet.”
Dr. Johnson added that he generally recommends that patients taking antibiotics eat “a bland diet, avoiding things that may have been provocative in the past.”
If patients are already enjoying foods with microbiome-boosting reputations, Dr. Johnson sees “very little downside to continuing that [while on antibiotics].” However, he noted that the period in which you’re taking antibiotics isn’t ideal for trying new foods, given the lack of experience with how the gut bacteria will react.
There are data about foods to avoid while taking antibiotics, which generally fall in line with common dietary knowledge. Many patients may know not to drink grapefruit juice with certain medications, but it’s worth a reminder. Certain antibiotics may also require delaying or avoiding dairy products, although this does not apply to yogurt.
A fiber-deficient diet can aggravate microbiota collapse following antibiotics. In a 2020 study, researchers showed that people on a high-fat diet who were using antibiotics were 8.6 times more likely to have preinflammatory bowel disease than those eating low-fat foods and who had no recent history of antibiotic use. Mouse model data from the same study indicated that poor diet and antibiotics may have worked in conjunction to reduce oxygen in the gut.
Dr. McFarland noted that building a healthy microbiome is a lifelong pursuit and that several factors (for example, environmental or genetic) are out of individuals’ hands. The general public might want a quick fix – ironically, one of the main drivers behind their requesting and receiving antibiotics when they’re not indicated – but it’s likely not available to them.
“You can’t eat one salad and suddenly have a healthy gut, unfortunately.”
A version of this article first appeared on Medscape.com.
Antibiotics are among the most commonly prescribed medications in both outpatient and hospital settings. Global efforts at curbing antibiotic-resistant strains have prompted clinicians to pursue better stewardship, whereby they limit their prescribing of such medications to those who truly need them.
Yet there’s another possible means of addressing antibiotic resistance – using dietary interventions to reduce the gastrointestinal complaints that so often accompany the use of antibiotics: vomiting, nausea, diarrhea, bloating/indigestion, abdominal pain, and loss of appetite. Far from being a mere nuisance, these complications can have major ramifications.
“Often [these side effects] will result in people stopping an antibiotic they need or taking a second- or third-generation one, which potentially causes even more problems with resistance,” explained Daniel J. Merenstein, MD, a professor of family medicine at Georgetown University, Washington, who has conducted multiple trials on antibiotic stewardship and probiotics.
And it’s not just clinicians who would like to find a way around these common complaints. On lifestyle blogs such as Livestrong and Goop, medical news websites, and via academic institutions, patients can learn how the right probiotic supplement or certain fermented or high-fiber foods, for example, might spare them the upset stomach that often accompanies a course of antibiotics.
Yet according to experts in the field, there are notable questions to be answered about whether there’s evidence to support this approach.
The cost of GI discomfort
Pinpointing the exact manner by which antibiotics upend GI tracts is a complex task, according to Gail Cresci, PhD, RD, a microbiome researcher in the department of pediatric gastroenterology, hepatology, and nutrition and director of nutrition research at the Cleveland Clinic.
“A lot of different mechanisms can go awry,” Dr. Cresci said. “But normally, the good microbes are there to help keep the balance, so when that balance is gone, the pathogens can take over and lead to this disruption.”
According to Lynne V. McFarland, PhD, a recently retired infectious disease epidemiologist who now serves as a private consultant, such complications can have substantial ramifications for patients and health care systems.
“If you’re an inpatient and develop antibiotic-associated diarrhea, it usually increases the length of stay from 8 to 20 days. It also increases the cost of care. And small children who have this can get severely dehydrated, which can become life threatening.”
Proponents of probiotics
Several of the researchers who were interviewed believe there’s convincing evidence supporting probiotics for the treatment of common antibiotic-related GI complaints. In many instances, they were involved in the studies themselves.
During Dr. McFarland’s 4-decade career in probiotics research, she has participated in early animal studies with strains such as Saccharomyces boulardii and was involved in meta-analyses of their role in Clostridioides difficile infection and associated diarrhea and even as a potential GI intervention for COVID-19 patients (Antibiotics (Basel). 2021 Apr 9;10(4):408).
In mouse model studies from 2013 and 2018, Dr. Cresci and colleagues showed that the probiotic strains Lactobacillus GG and Faecalibacterium prausnitzii reduced the structural gut changes that lead to antibiotic-associated diarrhea and minimized the risk of C. difficile infection.
In a 2021 randomized controlled trial led by Merenstein, healthy participants were given a trial of amoxicillin/clavulanate (days 1-7) in conjunction with either yogurt containing the probiotic Bifidobacterium animalis subsp lactis BB-12 or control yogurt (days 1-14). After assessing feces samples over a 30-day period, they found that those who received the probiotic yogurt had a significantly smaller decrease in short-chain fatty acid levels and a more stable taxonomic microbiota profile than control persons.
Dr. Merenstein said that, on the basis of results such as these and others, he’s comfortable being relatively definitive about the value of probiotics.
“I believe it’s close to standard of care that if you’re prescribing antibiotics, especially for more than 7 days, you really need to put people on probiotics that have been studied, simply because the evidence is robust enough now,” he said.
Even for proponents, there are caveats
However, all the researchers recommending the use of probiotics did so with caveats. First and foremost, they advise clinicians that the term “probiotics” is an imprecise catchall and is essentially meaningless.
“A lot of products label themselves as probiotic. It’s a great marketing scheme, but many of the products out there aren’t really probiotics; they’re not proven with randomized control trials and don’t have the scientific background,” said Dr. McFarland. “We’ve found that the efficacy is extremely strain specific and disease specific. A strain may work for one disease and not work for another.”
In 2018, Dr. McFarland coauthored an evidence-based practical guide to help clinicians and patients identify the specific strain that works in certain indications. Dr. Cresci recommends that clinicians consult websites such as Probiotics.org or the National Institutes of Health’s database to find the strains that have been proven to work in well-designed clinical trials.
There was also agreement that, to date, the most robust data support probiotics for the treatment of antibiotic-associated diarrhea.
Although the optimal timing of probiotics is a subject of debate, most proponents agreed that the general rule is “the sooner the better.”
Dr. McFarland recommended incorporating probiotics within 24 hours of starting an antibiotic “because the damage done to your GI tract microbiome is pretty quick, and the probiotics work best if they’re established before major disruption occurs.” She added that patients should continue taking probiotics for 2-8 weeks after stopping antibiotics.
“It takes a long time for your normal flora to get restored,” she said. “It’s best to cover your bases.”
For others, the evidence is not definitive
Opinions on the value of probiotics to combat antibiotic-related GI side effects are divergent, though.
“I would not recommend the routine use of probiotics, and certainly not in the prevention of C. difficile or antibiotic-related diarrhea,” said David A. Johnson, MD, professor of medicine and chief of gastroenterology at Eastern Virginia Medical School, Norfolk. “I think the evidence does not support that, and I stand strongly on that recommendation.”
Dr. Johnson cited the 2020 guidelines from the American Gastroenterological Association, which offer only a conditional recommendation for the use of specific probiotics and only in preventing antibiotic-associated C. difficile infection.
Geoffrey A. Preidis, MD, PhD, an assistant professor of pediatrics in the section of gastroenterology, hepatology, and nutrition at Baylor College of Medicine, Houston, served as a coauthor of the AGA’s guidelines. He noted that after reviewing 39 published trials of approximately 10,000 patients given probiotics while receiving antibiotics, the authors “did find some evidence that specific probiotics might decrease the risk of C. difficile diarrhea, but the quality of that evidence was low.”
Dr. Preidis attributed this to the lack of well-designed multicenter trials that can isolate the effects of certain strains and determine their benefit in this application.
“The majority of published trials have not reported safety data as rigorously as these data are reported in pharmaceutical trials, so the risk of side effects could be higher than we think,” said Dr. Preidis. “As living microbes, probiotics can move from the intestines into the bloodstream, causing sepsis. Contamination in the manufacturing process has been reported. There might be other long-term effects that we are not yet aware of.”
When asked to characterize the available data on probiotics, Dr. Johnson replied, “I’d generally label it, ‘caveat emptor.’ ”
Dr. McFarland agreed that the field would benefit from better-designed studies and called out meta-analyses that pool outcomes with various strains for particular criticism.
“When researchers do that, it’s no longer valid and shouldn’t have been published, in my opinion,” she told this news organization.
‘Antibiotic diets’ as a possible approach
A compromise between the two sides may be the so-called antibiotic diet. The theory behind such diets is that foods and beverages with biome-boosting properties may be a risk-free intervention that patients can adopt to alleviate antibiotic-related side effects.
“You want your diet to include more soluble fibers to help support the good bacteria, particularly when you’re taking antibiotics,” said Dr. Cresci. “You can get this through eating fresh fruits and vegetables, whole grains, and foods that have more prebiotic, like potatoes. You can also eat fermented food, such as kefir, kombucha, kimchi, and yogurt, so you’re adding more beneficial bacteria into your intestinal tract.”
There is ample published evidence that such foods can boost microbiome diversity and decrease inflammation, including a July 2021 study in Cell. However, the protection this may confer while taking antibiotics isn’t known. Establishing a clear role for the efficacy of such interventions is made additionally difficult by the well-established limitations of conducting dietary clinical trials.
Dr. Merenstein said that there is no compelling evidence that antibiotic-related complications can be offset by changing what goes onto our dinner plates. He joked: “We can’t say, ‘Here’s amoxicillin for your ear infection, now make sure you increase your fermented food, fiber, and water.’ ”
Dr. Johnson said he’s intrigued by studies of prebiotics – fibers that boost beneficial bacteria in the GI system.
“I would love to have more findings about prebiotic identification; that is, things we could do in a healthy way to keep the gut balanced while it’s subject to a change with antibiotics,” he said. “We’re just not there yet.”
Dr. Johnson added that he generally recommends that patients taking antibiotics eat “a bland diet, avoiding things that may have been provocative in the past.”
If patients are already enjoying foods with microbiome-boosting reputations, Dr. Johnson sees “very little downside to continuing that [while on antibiotics].” However, he noted that the period in which you’re taking antibiotics isn’t ideal for trying new foods, given the lack of experience with how the gut bacteria will react.
There are data about foods to avoid while taking antibiotics, which generally fall in line with common dietary knowledge. Many patients may know not to drink grapefruit juice with certain medications, but it’s worth a reminder. Certain antibiotics may also require delaying or avoiding dairy products, although this does not apply to yogurt.
A fiber-deficient diet can aggravate microbiota collapse following antibiotics. In a 2020 study, researchers showed that people on a high-fat diet who were using antibiotics were 8.6 times more likely to have preinflammatory bowel disease than those eating low-fat foods and who had no recent history of antibiotic use. Mouse model data from the same study indicated that poor diet and antibiotics may have worked in conjunction to reduce oxygen in the gut.
Dr. McFarland noted that building a healthy microbiome is a lifelong pursuit and that several factors (for example, environmental or genetic) are out of individuals’ hands. The general public might want a quick fix – ironically, one of the main drivers behind their requesting and receiving antibiotics when they’re not indicated – but it’s likely not available to them.
“You can’t eat one salad and suddenly have a healthy gut, unfortunately.”
A version of this article first appeared on Medscape.com.
Antibiotics are among the most commonly prescribed medications in both outpatient and hospital settings. Global efforts at curbing antibiotic-resistant strains have prompted clinicians to pursue better stewardship, whereby they limit their prescribing of such medications to those who truly need them.
Yet there’s another possible means of addressing antibiotic resistance – using dietary interventions to reduce the gastrointestinal complaints that so often accompany the use of antibiotics: vomiting, nausea, diarrhea, bloating/indigestion, abdominal pain, and loss of appetite. Far from being a mere nuisance, these complications can have major ramifications.
“Often [these side effects] will result in people stopping an antibiotic they need or taking a second- or third-generation one, which potentially causes even more problems with resistance,” explained Daniel J. Merenstein, MD, a professor of family medicine at Georgetown University, Washington, who has conducted multiple trials on antibiotic stewardship and probiotics.
And it’s not just clinicians who would like to find a way around these common complaints. On lifestyle blogs such as Livestrong and Goop, medical news websites, and via academic institutions, patients can learn how the right probiotic supplement or certain fermented or high-fiber foods, for example, might spare them the upset stomach that often accompanies a course of antibiotics.
Yet according to experts in the field, there are notable questions to be answered about whether there’s evidence to support this approach.
The cost of GI discomfort
Pinpointing the exact manner by which antibiotics upend GI tracts is a complex task, according to Gail Cresci, PhD, RD, a microbiome researcher in the department of pediatric gastroenterology, hepatology, and nutrition and director of nutrition research at the Cleveland Clinic.
“A lot of different mechanisms can go awry,” Dr. Cresci said. “But normally, the good microbes are there to help keep the balance, so when that balance is gone, the pathogens can take over and lead to this disruption.”
According to Lynne V. McFarland, PhD, a recently retired infectious disease epidemiologist who now serves as a private consultant, such complications can have substantial ramifications for patients and health care systems.
“If you’re an inpatient and develop antibiotic-associated diarrhea, it usually increases the length of stay from 8 to 20 days. It also increases the cost of care. And small children who have this can get severely dehydrated, which can become life threatening.”
Proponents of probiotics
Several of the researchers who were interviewed believe there’s convincing evidence supporting probiotics for the treatment of common antibiotic-related GI complaints. In many instances, they were involved in the studies themselves.
During Dr. McFarland’s 4-decade career in probiotics research, she has participated in early animal studies with strains such as Saccharomyces boulardii and was involved in meta-analyses of their role in Clostridioides difficile infection and associated diarrhea and even as a potential GI intervention for COVID-19 patients (Antibiotics (Basel). 2021 Apr 9;10(4):408).
In mouse model studies from 2013 and 2018, Dr. Cresci and colleagues showed that the probiotic strains Lactobacillus GG and Faecalibacterium prausnitzii reduced the structural gut changes that lead to antibiotic-associated diarrhea and minimized the risk of C. difficile infection.
In a 2021 randomized controlled trial led by Merenstein, healthy participants were given a trial of amoxicillin/clavulanate (days 1-7) in conjunction with either yogurt containing the probiotic Bifidobacterium animalis subsp lactis BB-12 or control yogurt (days 1-14). After assessing feces samples over a 30-day period, they found that those who received the probiotic yogurt had a significantly smaller decrease in short-chain fatty acid levels and a more stable taxonomic microbiota profile than control persons.
Dr. Merenstein said that, on the basis of results such as these and others, he’s comfortable being relatively definitive about the value of probiotics.
“I believe it’s close to standard of care that if you’re prescribing antibiotics, especially for more than 7 days, you really need to put people on probiotics that have been studied, simply because the evidence is robust enough now,” he said.
Even for proponents, there are caveats
However, all the researchers recommending the use of probiotics did so with caveats. First and foremost, they advise clinicians that the term “probiotics” is an imprecise catchall and is essentially meaningless.
“A lot of products label themselves as probiotic. It’s a great marketing scheme, but many of the products out there aren’t really probiotics; they’re not proven with randomized control trials and don’t have the scientific background,” said Dr. McFarland. “We’ve found that the efficacy is extremely strain specific and disease specific. A strain may work for one disease and not work for another.”
In 2018, Dr. McFarland coauthored an evidence-based practical guide to help clinicians and patients identify the specific strain that works in certain indications. Dr. Cresci recommends that clinicians consult websites such as Probiotics.org or the National Institutes of Health’s database to find the strains that have been proven to work in well-designed clinical trials.
There was also agreement that, to date, the most robust data support probiotics for the treatment of antibiotic-associated diarrhea.
Although the optimal timing of probiotics is a subject of debate, most proponents agreed that the general rule is “the sooner the better.”
Dr. McFarland recommended incorporating probiotics within 24 hours of starting an antibiotic “because the damage done to your GI tract microbiome is pretty quick, and the probiotics work best if they’re established before major disruption occurs.” She added that patients should continue taking probiotics for 2-8 weeks after stopping antibiotics.
“It takes a long time for your normal flora to get restored,” she said. “It’s best to cover your bases.”
For others, the evidence is not definitive
Opinions on the value of probiotics to combat antibiotic-related GI side effects are divergent, though.
“I would not recommend the routine use of probiotics, and certainly not in the prevention of C. difficile or antibiotic-related diarrhea,” said David A. Johnson, MD, professor of medicine and chief of gastroenterology at Eastern Virginia Medical School, Norfolk. “I think the evidence does not support that, and I stand strongly on that recommendation.”
Dr. Johnson cited the 2020 guidelines from the American Gastroenterological Association, which offer only a conditional recommendation for the use of specific probiotics and only in preventing antibiotic-associated C. difficile infection.
Geoffrey A. Preidis, MD, PhD, an assistant professor of pediatrics in the section of gastroenterology, hepatology, and nutrition at Baylor College of Medicine, Houston, served as a coauthor of the AGA’s guidelines. He noted that after reviewing 39 published trials of approximately 10,000 patients given probiotics while receiving antibiotics, the authors “did find some evidence that specific probiotics might decrease the risk of C. difficile diarrhea, but the quality of that evidence was low.”
Dr. Preidis attributed this to the lack of well-designed multicenter trials that can isolate the effects of certain strains and determine their benefit in this application.
“The majority of published trials have not reported safety data as rigorously as these data are reported in pharmaceutical trials, so the risk of side effects could be higher than we think,” said Dr. Preidis. “As living microbes, probiotics can move from the intestines into the bloodstream, causing sepsis. Contamination in the manufacturing process has been reported. There might be other long-term effects that we are not yet aware of.”
When asked to characterize the available data on probiotics, Dr. Johnson replied, “I’d generally label it, ‘caveat emptor.’ ”
Dr. McFarland agreed that the field would benefit from better-designed studies and called out meta-analyses that pool outcomes with various strains for particular criticism.
“When researchers do that, it’s no longer valid and shouldn’t have been published, in my opinion,” she told this news organization.
‘Antibiotic diets’ as a possible approach
A compromise between the two sides may be the so-called antibiotic diet. The theory behind such diets is that foods and beverages with biome-boosting properties may be a risk-free intervention that patients can adopt to alleviate antibiotic-related side effects.
“You want your diet to include more soluble fibers to help support the good bacteria, particularly when you’re taking antibiotics,” said Dr. Cresci. “You can get this through eating fresh fruits and vegetables, whole grains, and foods that have more prebiotic, like potatoes. You can also eat fermented food, such as kefir, kombucha, kimchi, and yogurt, so you’re adding more beneficial bacteria into your intestinal tract.”
There is ample published evidence that such foods can boost microbiome diversity and decrease inflammation, including a July 2021 study in Cell. However, the protection this may confer while taking antibiotics isn’t known. Establishing a clear role for the efficacy of such interventions is made additionally difficult by the well-established limitations of conducting dietary clinical trials.
Dr. Merenstein said that there is no compelling evidence that antibiotic-related complications can be offset by changing what goes onto our dinner plates. He joked: “We can’t say, ‘Here’s amoxicillin for your ear infection, now make sure you increase your fermented food, fiber, and water.’ ”
Dr. Johnson said he’s intrigued by studies of prebiotics – fibers that boost beneficial bacteria in the GI system.
“I would love to have more findings about prebiotic identification; that is, things we could do in a healthy way to keep the gut balanced while it’s subject to a change with antibiotics,” he said. “We’re just not there yet.”
Dr. Johnson added that he generally recommends that patients taking antibiotics eat “a bland diet, avoiding things that may have been provocative in the past.”
If patients are already enjoying foods with microbiome-boosting reputations, Dr. Johnson sees “very little downside to continuing that [while on antibiotics].” However, he noted that the period in which you’re taking antibiotics isn’t ideal for trying new foods, given the lack of experience with how the gut bacteria will react.
There are data about foods to avoid while taking antibiotics, which generally fall in line with common dietary knowledge. Many patients may know not to drink grapefruit juice with certain medications, but it’s worth a reminder. Certain antibiotics may also require delaying or avoiding dairy products, although this does not apply to yogurt.
A fiber-deficient diet can aggravate microbiota collapse following antibiotics. In a 2020 study, researchers showed that people on a high-fat diet who were using antibiotics were 8.6 times more likely to have preinflammatory bowel disease than those eating low-fat foods and who had no recent history of antibiotic use. Mouse model data from the same study indicated that poor diet and antibiotics may have worked in conjunction to reduce oxygen in the gut.
Dr. McFarland noted that building a healthy microbiome is a lifelong pursuit and that several factors (for example, environmental or genetic) are out of individuals’ hands. The general public might want a quick fix – ironically, one of the main drivers behind their requesting and receiving antibiotics when they’re not indicated – but it’s likely not available to them.
“You can’t eat one salad and suddenly have a healthy gut, unfortunately.”
A version of this article first appeared on Medscape.com.
Children and COVID: CDC gives perspective on hospitalizations
New COVID-19 cases in children fell by 23% as the latest weekly count dropped to its lowest level since July of 2021, based on data from the American Academy of Pediatrics and the Children’s Hospital Association.
, when the early stages of the Delta surge led to 23,551 cases, the AAP and CHA said in their weekly COVID report.
The two organizations put the total number of cases at nearly 12.8 million from the start of the pandemic to March 17, with children representing 19.0% of cases among all ages. The Centers for Disease Control and Prevention puts the cumulative number of COVID-19 cases at almost 12.0 million as of March 21, or 17.5% of the nationwide total.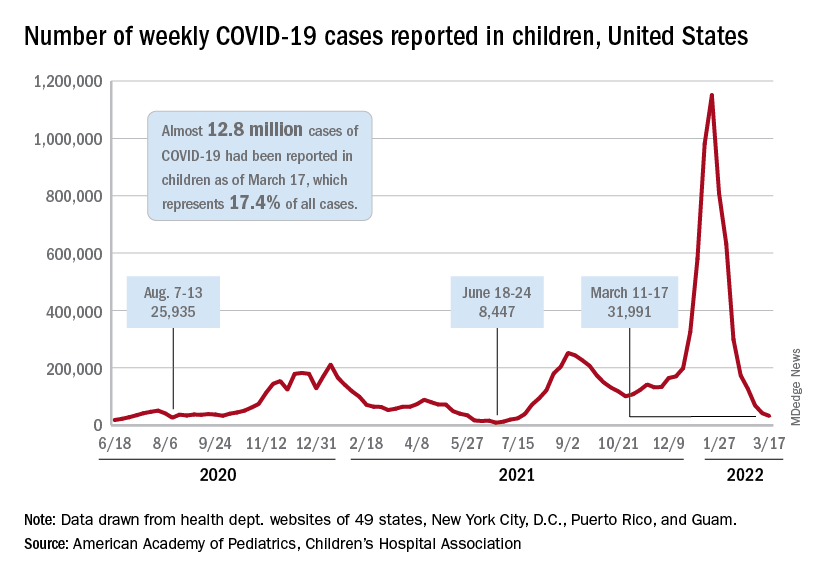
COVID-related hospitalizations also continue to fall, and two new studies from the CDC put children’s experiences during the Omicron surge and the larger pandemic into perspective.
One study showed that hospitalization rates for children aged 4 years and younger during the Omicron surge were five times higher than at the peak of the Delta surge, with the highest rates occurring in infants under 6 months of age. That report was based on the CDC’s COVID-19–Associated Hospitalization Surveillance Network (COVID-NET), which covers 99 counties across 14 states (MMWR. 2022 March 18;71[11]:429-36).
The second study compared child hospitalizations during 1 year of the COVID pandemic (Oct. 1, 2020, to Sept. 30, 2021) with three influenza seasons (2017-2018 through 2019-2020). The pre-Omicron hospitalization rate for those under age 18 years, 48.2 per 100,000 children, was higher than any of the three flu seasons: 33.5 per 100,000 in 2017-2018, 33.8 in 2018-2019, and 41.7 for 2019-2020, the investigators said in a medRxiv preprint.
Most of the increased COVID burden fell on adolescents aged 12-17, they said. The COVID hospitalization rate for that age group was 59.9 per 100,000, versus 12.2-14.1 for influenza, while children aged 5-11 had a COVID-related rate of 25.0 and flu-related rates of 24.3-31.7, and those aged 0-4 had rates of 66.8 for COVID and 70.9-91.5 for the flu, Miranda J. Delahoy of the CDC’s COVID-19 Response Team and associates reported.
New COVID-19 cases in children fell by 23% as the latest weekly count dropped to its lowest level since July of 2021, based on data from the American Academy of Pediatrics and the Children’s Hospital Association.
, when the early stages of the Delta surge led to 23,551 cases, the AAP and CHA said in their weekly COVID report.
The two organizations put the total number of cases at nearly 12.8 million from the start of the pandemic to March 17, with children representing 19.0% of cases among all ages. The Centers for Disease Control and Prevention puts the cumulative number of COVID-19 cases at almost 12.0 million as of March 21, or 17.5% of the nationwide total.
COVID-related hospitalizations also continue to fall, and two new studies from the CDC put children’s experiences during the Omicron surge and the larger pandemic into perspective.
One study showed that hospitalization rates for children aged 4 years and younger during the Omicron surge were five times higher than at the peak of the Delta surge, with the highest rates occurring in infants under 6 months of age. That report was based on the CDC’s COVID-19–Associated Hospitalization Surveillance Network (COVID-NET), which covers 99 counties across 14 states (MMWR. 2022 March 18;71[11]:429-36).
The second study compared child hospitalizations during 1 year of the COVID pandemic (Oct. 1, 2020, to Sept. 30, 2021) with three influenza seasons (2017-2018 through 2019-2020). The pre-Omicron hospitalization rate for those under age 18 years, 48.2 per 100,000 children, was higher than any of the three flu seasons: 33.5 per 100,000 in 2017-2018, 33.8 in 2018-2019, and 41.7 for 2019-2020, the investigators said in a medRxiv preprint.
Most of the increased COVID burden fell on adolescents aged 12-17, they said. The COVID hospitalization rate for that age group was 59.9 per 100,000, versus 12.2-14.1 for influenza, while children aged 5-11 had a COVID-related rate of 25.0 and flu-related rates of 24.3-31.7, and those aged 0-4 had rates of 66.8 for COVID and 70.9-91.5 for the flu, Miranda J. Delahoy of the CDC’s COVID-19 Response Team and associates reported.
New COVID-19 cases in children fell by 23% as the latest weekly count dropped to its lowest level since July of 2021, based on data from the American Academy of Pediatrics and the Children’s Hospital Association.
, when the early stages of the Delta surge led to 23,551 cases, the AAP and CHA said in their weekly COVID report.
The two organizations put the total number of cases at nearly 12.8 million from the start of the pandemic to March 17, with children representing 19.0% of cases among all ages. The Centers for Disease Control and Prevention puts the cumulative number of COVID-19 cases at almost 12.0 million as of March 21, or 17.5% of the nationwide total.
COVID-related hospitalizations also continue to fall, and two new studies from the CDC put children’s experiences during the Omicron surge and the larger pandemic into perspective.
One study showed that hospitalization rates for children aged 4 years and younger during the Omicron surge were five times higher than at the peak of the Delta surge, with the highest rates occurring in infants under 6 months of age. That report was based on the CDC’s COVID-19–Associated Hospitalization Surveillance Network (COVID-NET), which covers 99 counties across 14 states (MMWR. 2022 March 18;71[11]:429-36).
The second study compared child hospitalizations during 1 year of the COVID pandemic (Oct. 1, 2020, to Sept. 30, 2021) with three influenza seasons (2017-2018 through 2019-2020). The pre-Omicron hospitalization rate for those under age 18 years, 48.2 per 100,000 children, was higher than any of the three flu seasons: 33.5 per 100,000 in 2017-2018, 33.8 in 2018-2019, and 41.7 for 2019-2020, the investigators said in a medRxiv preprint.
Most of the increased COVID burden fell on adolescents aged 12-17, they said. The COVID hospitalization rate for that age group was 59.9 per 100,000, versus 12.2-14.1 for influenza, while children aged 5-11 had a COVID-related rate of 25.0 and flu-related rates of 24.3-31.7, and those aged 0-4 had rates of 66.8 for COVID and 70.9-91.5 for the flu, Miranda J. Delahoy of the CDC’s COVID-19 Response Team and associates reported.
Mild COVID-19 infection linked to later type 2 diabetes
People who recover from a mild case of COVID-19 appear to have an increased risk for subsequent new-onset type 2 diabetes but not other types of diabetes, new data suggest.
“If confirmed, the results of the present study indicate that diabetes screening in individuals who have recovered from even mild COVID-19 should be recommended,” say Wolfgang Rathmann, MD, of the Leibniz Center for Diabetes Research at Heinrich Heine University, Düsseldorf, Germany, and colleagues.
The findings, from a nationwide primary care database in Germany, were recently published in Diabetologia.
These primary care data align with those from other studies of more seriously ill patients with COVID-19 that found increased rates of type 2 diabetes diagnoses in the subsequent months following illness, they point out.
“COVID-19 infection may lead to diabetes by upregulation of the immune system after remission, which may induce pancreatic beta-cell dysfunction and insulin resistance, or patients may have been at risk for developing diabetes due to having obesity or prediabetes, and the stress COVID-19 put on their bodies sped it up,” said Dr. Rathmann in a press release.
However, because the patients with COVID-19 in the study were only followed for about 3 months, “further follow-up is needed to understand whether type 2 diabetes after mild COVID-19 is just temporary and can be reversed after they have fully recovered or whether it leads to a chronic condition,” he noted.
Increase in type 2 diabetes 3 months after mild COVID-19
The retrospective cohort analysis was performed using data from the Disease Analyzer, a representative panel of 1,171 physician practices in Germany, from March 2020 to January 2021, with follow-up through July 2021.
Individuals with a history of COVID-19 or diabetes and those taking corticosteroids within 30 days after the index dates were excluded.
A total of 35,865 patients with confirmed SARS-CoV-2 infection were propensity score-matched on a one-to-one basis for sex, age, health insurance, and comorbidities with those who had acute respiratory tract infections (controls) but were COVID-19 negative. Median follow-up was 119 days for the COVID-19 group and 161 days for controls.
There was a 28% increased risk of type 2 diabetes for those who had COVID-19 versus controls (15.8 per 1,000 person-years vs. 12.3 per 1,000 person-years, respectively, which was significantly different, and an incidence rate ratio of 1.28).
The incidence of other types of diabetes or unspecified diabetes for the COVID-19 and control groups did not differ significantly (4.3 per 1,000 person-years vs. 3.7 per 1,000 person-years; IRR, 1.17).
Similar findings were seen in sensitivity analyses by glucose-lowering medication prescriptions and by ICD-10 codes.
Although type 2 diabetes is not likely to be a problem for the vast majority of people who have mild COVID-19, the authors recommend that anyone who has recovered from COVID-19 be aware of the warning signs and symptoms such as fatigue, frequent urination, and increased thirst, and seek treatment right away.
CoviDiab registry tracking type 1 and type 2 diabetes
Over the course of the pandemic, there have been conflicting data on whether COVID-19 induces or reveals a propensity for type 1 and type 2 diabetes.
The CoviDiab global registry is tracking this and will include diabetes type for adults and children.
The aim is to have “as many as possible cases of new-onset diabetes for which we can have also a minimum set of clinical data including type of diabetes and A1c,” coprincipal investigator Francesco Rubino, MD, of King’s College London, previously told this news organization.
“By looking at this information we can infer whether a role of COVID-19 in triggering diabetes is clinically plausible – or not – and what type of diabetes is most frequently associated with COVID-19.”
Rubino said that the CoviDiab team is approaching the data with the assumption that, at least in adults diagnosed with type 2 diabetes, the explanation might be that the person already had undiagnosed diabetes or the hyperglycemia may be stress-induced and temporary.
The German Diabetes Center is funded by the German Federal Ministry of Health and the Ministry of Culture and Science of the State of North Rhine-Westphalia. Dr. Rathmann has reported receiving consulting fees for attending educational sessions or advisory boards for AstraZeneca, Boehringer Ingelheim, and Novo Nordisk and institutional research grants from Novo Nordisk outside of the topic of the current work.
A version of this article first appeared on Medscape.com.
People who recover from a mild case of COVID-19 appear to have an increased risk for subsequent new-onset type 2 diabetes but not other types of diabetes, new data suggest.
“If confirmed, the results of the present study indicate that diabetes screening in individuals who have recovered from even mild COVID-19 should be recommended,” say Wolfgang Rathmann, MD, of the Leibniz Center for Diabetes Research at Heinrich Heine University, Düsseldorf, Germany, and colleagues.
The findings, from a nationwide primary care database in Germany, were recently published in Diabetologia.
These primary care data align with those from other studies of more seriously ill patients with COVID-19 that found increased rates of type 2 diabetes diagnoses in the subsequent months following illness, they point out.
“COVID-19 infection may lead to diabetes by upregulation of the immune system after remission, which may induce pancreatic beta-cell dysfunction and insulin resistance, or patients may have been at risk for developing diabetes due to having obesity or prediabetes, and the stress COVID-19 put on their bodies sped it up,” said Dr. Rathmann in a press release.
However, because the patients with COVID-19 in the study were only followed for about 3 months, “further follow-up is needed to understand whether type 2 diabetes after mild COVID-19 is just temporary and can be reversed after they have fully recovered or whether it leads to a chronic condition,” he noted.
Increase in type 2 diabetes 3 months after mild COVID-19
The retrospective cohort analysis was performed using data from the Disease Analyzer, a representative panel of 1,171 physician practices in Germany, from March 2020 to January 2021, with follow-up through July 2021.
Individuals with a history of COVID-19 or diabetes and those taking corticosteroids within 30 days after the index dates were excluded.
A total of 35,865 patients with confirmed SARS-CoV-2 infection were propensity score-matched on a one-to-one basis for sex, age, health insurance, and comorbidities with those who had acute respiratory tract infections (controls) but were COVID-19 negative. Median follow-up was 119 days for the COVID-19 group and 161 days for controls.
There was a 28% increased risk of type 2 diabetes for those who had COVID-19 versus controls (15.8 per 1,000 person-years vs. 12.3 per 1,000 person-years, respectively, which was significantly different, and an incidence rate ratio of 1.28).
The incidence of other types of diabetes or unspecified diabetes for the COVID-19 and control groups did not differ significantly (4.3 per 1,000 person-years vs. 3.7 per 1,000 person-years; IRR, 1.17).
Similar findings were seen in sensitivity analyses by glucose-lowering medication prescriptions and by ICD-10 codes.
Although type 2 diabetes is not likely to be a problem for the vast majority of people who have mild COVID-19, the authors recommend that anyone who has recovered from COVID-19 be aware of the warning signs and symptoms such as fatigue, frequent urination, and increased thirst, and seek treatment right away.
CoviDiab registry tracking type 1 and type 2 diabetes
Over the course of the pandemic, there have been conflicting data on whether COVID-19 induces or reveals a propensity for type 1 and type 2 diabetes.
The CoviDiab global registry is tracking this and will include diabetes type for adults and children.
The aim is to have “as many as possible cases of new-onset diabetes for which we can have also a minimum set of clinical data including type of diabetes and A1c,” coprincipal investigator Francesco Rubino, MD, of King’s College London, previously told this news organization.
“By looking at this information we can infer whether a role of COVID-19 in triggering diabetes is clinically plausible – or not – and what type of diabetes is most frequently associated with COVID-19.”
Rubino said that the CoviDiab team is approaching the data with the assumption that, at least in adults diagnosed with type 2 diabetes, the explanation might be that the person already had undiagnosed diabetes or the hyperglycemia may be stress-induced and temporary.
The German Diabetes Center is funded by the German Federal Ministry of Health and the Ministry of Culture and Science of the State of North Rhine-Westphalia. Dr. Rathmann has reported receiving consulting fees for attending educational sessions or advisory boards for AstraZeneca, Boehringer Ingelheim, and Novo Nordisk and institutional research grants from Novo Nordisk outside of the topic of the current work.
A version of this article first appeared on Medscape.com.
People who recover from a mild case of COVID-19 appear to have an increased risk for subsequent new-onset type 2 diabetes but not other types of diabetes, new data suggest.
“If confirmed, the results of the present study indicate that diabetes screening in individuals who have recovered from even mild COVID-19 should be recommended,” say Wolfgang Rathmann, MD, of the Leibniz Center for Diabetes Research at Heinrich Heine University, Düsseldorf, Germany, and colleagues.
The findings, from a nationwide primary care database in Germany, were recently published in Diabetologia.
These primary care data align with those from other studies of more seriously ill patients with COVID-19 that found increased rates of type 2 diabetes diagnoses in the subsequent months following illness, they point out.
“COVID-19 infection may lead to diabetes by upregulation of the immune system after remission, which may induce pancreatic beta-cell dysfunction and insulin resistance, or patients may have been at risk for developing diabetes due to having obesity or prediabetes, and the stress COVID-19 put on their bodies sped it up,” said Dr. Rathmann in a press release.
However, because the patients with COVID-19 in the study were only followed for about 3 months, “further follow-up is needed to understand whether type 2 diabetes after mild COVID-19 is just temporary and can be reversed after they have fully recovered or whether it leads to a chronic condition,” he noted.
Increase in type 2 diabetes 3 months after mild COVID-19
The retrospective cohort analysis was performed using data from the Disease Analyzer, a representative panel of 1,171 physician practices in Germany, from March 2020 to January 2021, with follow-up through July 2021.
Individuals with a history of COVID-19 or diabetes and those taking corticosteroids within 30 days after the index dates were excluded.
A total of 35,865 patients with confirmed SARS-CoV-2 infection were propensity score-matched on a one-to-one basis for sex, age, health insurance, and comorbidities with those who had acute respiratory tract infections (controls) but were COVID-19 negative. Median follow-up was 119 days for the COVID-19 group and 161 days for controls.
There was a 28% increased risk of type 2 diabetes for those who had COVID-19 versus controls (15.8 per 1,000 person-years vs. 12.3 per 1,000 person-years, respectively, which was significantly different, and an incidence rate ratio of 1.28).
The incidence of other types of diabetes or unspecified diabetes for the COVID-19 and control groups did not differ significantly (4.3 per 1,000 person-years vs. 3.7 per 1,000 person-years; IRR, 1.17).
Similar findings were seen in sensitivity analyses by glucose-lowering medication prescriptions and by ICD-10 codes.
Although type 2 diabetes is not likely to be a problem for the vast majority of people who have mild COVID-19, the authors recommend that anyone who has recovered from COVID-19 be aware of the warning signs and symptoms such as fatigue, frequent urination, and increased thirst, and seek treatment right away.
CoviDiab registry tracking type 1 and type 2 diabetes
Over the course of the pandemic, there have been conflicting data on whether COVID-19 induces or reveals a propensity for type 1 and type 2 diabetes.
The CoviDiab global registry is tracking this and will include diabetes type for adults and children.
The aim is to have “as many as possible cases of new-onset diabetes for which we can have also a minimum set of clinical data including type of diabetes and A1c,” coprincipal investigator Francesco Rubino, MD, of King’s College London, previously told this news organization.
“By looking at this information we can infer whether a role of COVID-19 in triggering diabetes is clinically plausible – or not – and what type of diabetes is most frequently associated with COVID-19.”
Rubino said that the CoviDiab team is approaching the data with the assumption that, at least in adults diagnosed with type 2 diabetes, the explanation might be that the person already had undiagnosed diabetes or the hyperglycemia may be stress-induced and temporary.
The German Diabetes Center is funded by the German Federal Ministry of Health and the Ministry of Culture and Science of the State of North Rhine-Westphalia. Dr. Rathmann has reported receiving consulting fees for attending educational sessions or advisory boards for AstraZeneca, Boehringer Ingelheim, and Novo Nordisk and institutional research grants from Novo Nordisk outside of the topic of the current work.
A version of this article first appeared on Medscape.com.
FROM DIABETOLOGIA
Third-party vendor physicians more likely to prescribe antibiotics during acute care telehealth visits
Third-party vendor physicians appear to be more much more likely than their system-employed counterparts to prescribe antibiotics during acute care telehealth visits for acute respiratory infection (ARI), according to a study in the Journal of Telemedicine and Telecare.
As health systems expand their direct-to-consumer (DTC) virtual care, the quality of that care seems to vary, write the authors. Patients with ARI symptoms make up about one-third of virtual visits. Prescribing practice is a commonly cited measure of care quality for ARI, which is usually viral and rarely benefits from antibiotics.
“When providing care through telehealth, hospital-affiliated emergency physicians practiced better antibiotic stewardship than vendor-supplied, non–hospital-affiliated physicians,” lead study author Kathleen Li, MD, MS, a clinical lecturer in the department of emergency medicine at the University of Michigan, Ann Arbor, told this news organization.
“We had a sense that a difference existed, but the magnitude of the difference was larger than expected,” she said.
Dr. Li and her colleagues retrospectively analyzed on-demand telehealth visits available to health system employees and dependents of a large urban academic health system from March 2018, when the service began, through July 2019.
All 16 affiliated physicians providing ARI care were board-certified in emergency medicine, compared with 2 (8%) of the 25 unaffiliated (vendor-employed) physicians. Most unaffiliated physicians were known to be board-certified in family medicine, internal medicine, or pediatrics.
Unaffiliated physicians were not given access to the health system’s electronic medical record. Instead, all their patient histories, exams, assessments, plans, impressions, and discharge instructions were scanned into the electronic medical record system by other staff the next day.
Unaffiliated doctors were more than twice as likely to prescribe antibiotics
The researchers extracted data on all 257 virtual ARI visits from the electronic health record system, including prescriptions and medication therapeutic class. They performed multivariable logistic regression, adjusting for patient age and time of visit (weekday vs. weekend; day vs. overnight).
Antibiotic prescription rates were similar between weekday and weekend visits, and between day and night visits. Regardless of provider status, older patients were more likely to be prescribed antibiotics (P = .01).
Overall, affiliated physicians prescribed antibiotics during 18% of visits, whereas vendor physicians prescribed antibiotics during 37% of visits. After adjustments, the odds were 2.3 times higher that a patient in a telehealth visit with a vendor provider would be prescribed antibiotics (95% confidence interval, 1.1-4.5).
The predicted antibiotic prescribing rate for ARI was 19% (95% confidence interval, 13%-25%) for affiliated providers vs. 35% (95% CI, 22%-47%) for unaffiliated providers, an average marginal effect of 15% (95% CI, 2%-29%). The difference was even greater (average marginal effect 20%, 95% CI, 4%-35%) when children and patients over 65 were excluded.
Consistent, high-quality care and antibiotic stewardship are needed in all settings
Three experts who were not involved in the study commented on the study.
Joshua W. Elder, MD, MPH, MHS, medical director of Telehealth Express Care (direct-to-consumer telemedicine) at UC Davis Health in Sacramento, Calif., said, “An important unanswered question is how factors such as communication (policy and procedures, practice guidelines), connection (electronic health records), and reimbursement and incentives that health system and vendor-based providers received impacted this outcome.
“As the volume of virtual practices grows, most health systems will need to create a hybrid between health-system-employed and vendor-and/or-payer-supplied physicians,” he added. “Finding ways to create similar quality and outcomes will be essential in the evolving digital health infrastructure being developed.”
Charles Teixeira, DO, an infectious disease specialist at the Medical University of South Carolina in Charleston, said that this study highlighted the need to consistently provide high-quality, evidence-based care regardless of the encounter setting.
“It was important to compare the prescribing practices for commonly used medications, especially those as important as antibiotics,” he added. “Overprescribing antibiotics can have a progressive, long-term effect on a community and increase the risk for patients to develop multidrug-resistant bacteria.”
Jeffrey A. Linder, MD, MPH, the chief of general internal medicine and geriatrics in the department of medicine at Northwestern University in Chicago, commended the authors for investigating the quality of telehealth.
“The major limitation,” he found, “is that the investigators lumped all ARI visits – including those that are potentially antibiotic appropriate (e.g., otitis media, pharyngitis, sinusitis), those that are non–antibiotic appropriate (e.g., bronchitis, influenza, laryngitis, URI, viral syndrome), and those that are nonspecific symptoms (e.g., cough, congestion, fever, sore throat) – into the same category.”
No clinical information was collected or presented that would enable the reader to tell if these two groups of physicians were evaluating different patient populations or even if they just diagnosed patients differently,” he added.
“Our study did not delve into why we saw the difference,” Dr. Li explained. “Exploring potential reasons further will have important implications for how to optimally deliver care via telehealth.”
All authors and independent experts have disclosed no relevant financial relationships. The study received no financial support.
A version of this article first appeared on Medscape.com.
Third-party vendor physicians appear to be more much more likely than their system-employed counterparts to prescribe antibiotics during acute care telehealth visits for acute respiratory infection (ARI), according to a study in the Journal of Telemedicine and Telecare.
As health systems expand their direct-to-consumer (DTC) virtual care, the quality of that care seems to vary, write the authors. Patients with ARI symptoms make up about one-third of virtual visits. Prescribing practice is a commonly cited measure of care quality for ARI, which is usually viral and rarely benefits from antibiotics.
“When providing care through telehealth, hospital-affiliated emergency physicians practiced better antibiotic stewardship than vendor-supplied, non–hospital-affiliated physicians,” lead study author Kathleen Li, MD, MS, a clinical lecturer in the department of emergency medicine at the University of Michigan, Ann Arbor, told this news organization.
“We had a sense that a difference existed, but the magnitude of the difference was larger than expected,” she said.
Dr. Li and her colleagues retrospectively analyzed on-demand telehealth visits available to health system employees and dependents of a large urban academic health system from March 2018, when the service began, through July 2019.
All 16 affiliated physicians providing ARI care were board-certified in emergency medicine, compared with 2 (8%) of the 25 unaffiliated (vendor-employed) physicians. Most unaffiliated physicians were known to be board-certified in family medicine, internal medicine, or pediatrics.
Unaffiliated physicians were not given access to the health system’s electronic medical record. Instead, all their patient histories, exams, assessments, plans, impressions, and discharge instructions were scanned into the electronic medical record system by other staff the next day.
Unaffiliated doctors were more than twice as likely to prescribe antibiotics
The researchers extracted data on all 257 virtual ARI visits from the electronic health record system, including prescriptions and medication therapeutic class. They performed multivariable logistic regression, adjusting for patient age and time of visit (weekday vs. weekend; day vs. overnight).
Antibiotic prescription rates were similar between weekday and weekend visits, and between day and night visits. Regardless of provider status, older patients were more likely to be prescribed antibiotics (P = .01).
Overall, affiliated physicians prescribed antibiotics during 18% of visits, whereas vendor physicians prescribed antibiotics during 37% of visits. After adjustments, the odds were 2.3 times higher that a patient in a telehealth visit with a vendor provider would be prescribed antibiotics (95% confidence interval, 1.1-4.5).
The predicted antibiotic prescribing rate for ARI was 19% (95% confidence interval, 13%-25%) for affiliated providers vs. 35% (95% CI, 22%-47%) for unaffiliated providers, an average marginal effect of 15% (95% CI, 2%-29%). The difference was even greater (average marginal effect 20%, 95% CI, 4%-35%) when children and patients over 65 were excluded.
Consistent, high-quality care and antibiotic stewardship are needed in all settings
Three experts who were not involved in the study commented on the study.
Joshua W. Elder, MD, MPH, MHS, medical director of Telehealth Express Care (direct-to-consumer telemedicine) at UC Davis Health in Sacramento, Calif., said, “An important unanswered question is how factors such as communication (policy and procedures, practice guidelines), connection (electronic health records), and reimbursement and incentives that health system and vendor-based providers received impacted this outcome.
“As the volume of virtual practices grows, most health systems will need to create a hybrid between health-system-employed and vendor-and/or-payer-supplied physicians,” he added. “Finding ways to create similar quality and outcomes will be essential in the evolving digital health infrastructure being developed.”
Charles Teixeira, DO, an infectious disease specialist at the Medical University of South Carolina in Charleston, said that this study highlighted the need to consistently provide high-quality, evidence-based care regardless of the encounter setting.
“It was important to compare the prescribing practices for commonly used medications, especially those as important as antibiotics,” he added. “Overprescribing antibiotics can have a progressive, long-term effect on a community and increase the risk for patients to develop multidrug-resistant bacteria.”
Jeffrey A. Linder, MD, MPH, the chief of general internal medicine and geriatrics in the department of medicine at Northwestern University in Chicago, commended the authors for investigating the quality of telehealth.
“The major limitation,” he found, “is that the investigators lumped all ARI visits – including those that are potentially antibiotic appropriate (e.g., otitis media, pharyngitis, sinusitis), those that are non–antibiotic appropriate (e.g., bronchitis, influenza, laryngitis, URI, viral syndrome), and those that are nonspecific symptoms (e.g., cough, congestion, fever, sore throat) – into the same category.”
No clinical information was collected or presented that would enable the reader to tell if these two groups of physicians were evaluating different patient populations or even if they just diagnosed patients differently,” he added.
“Our study did not delve into why we saw the difference,” Dr. Li explained. “Exploring potential reasons further will have important implications for how to optimally deliver care via telehealth.”
All authors and independent experts have disclosed no relevant financial relationships. The study received no financial support.
A version of this article first appeared on Medscape.com.
Third-party vendor physicians appear to be more much more likely than their system-employed counterparts to prescribe antibiotics during acute care telehealth visits for acute respiratory infection (ARI), according to a study in the Journal of Telemedicine and Telecare.
As health systems expand their direct-to-consumer (DTC) virtual care, the quality of that care seems to vary, write the authors. Patients with ARI symptoms make up about one-third of virtual visits. Prescribing practice is a commonly cited measure of care quality for ARI, which is usually viral and rarely benefits from antibiotics.
“When providing care through telehealth, hospital-affiliated emergency physicians practiced better antibiotic stewardship than vendor-supplied, non–hospital-affiliated physicians,” lead study author Kathleen Li, MD, MS, a clinical lecturer in the department of emergency medicine at the University of Michigan, Ann Arbor, told this news organization.
“We had a sense that a difference existed, but the magnitude of the difference was larger than expected,” she said.
Dr. Li and her colleagues retrospectively analyzed on-demand telehealth visits available to health system employees and dependents of a large urban academic health system from March 2018, when the service began, through July 2019.
All 16 affiliated physicians providing ARI care were board-certified in emergency medicine, compared with 2 (8%) of the 25 unaffiliated (vendor-employed) physicians. Most unaffiliated physicians were known to be board-certified in family medicine, internal medicine, or pediatrics.
Unaffiliated physicians were not given access to the health system’s electronic medical record. Instead, all their patient histories, exams, assessments, plans, impressions, and discharge instructions were scanned into the electronic medical record system by other staff the next day.
Unaffiliated doctors were more than twice as likely to prescribe antibiotics
The researchers extracted data on all 257 virtual ARI visits from the electronic health record system, including prescriptions and medication therapeutic class. They performed multivariable logistic regression, adjusting for patient age and time of visit (weekday vs. weekend; day vs. overnight).
Antibiotic prescription rates were similar between weekday and weekend visits, and between day and night visits. Regardless of provider status, older patients were more likely to be prescribed antibiotics (P = .01).
Overall, affiliated physicians prescribed antibiotics during 18% of visits, whereas vendor physicians prescribed antibiotics during 37% of visits. After adjustments, the odds were 2.3 times higher that a patient in a telehealth visit with a vendor provider would be prescribed antibiotics (95% confidence interval, 1.1-4.5).
The predicted antibiotic prescribing rate for ARI was 19% (95% confidence interval, 13%-25%) for affiliated providers vs. 35% (95% CI, 22%-47%) for unaffiliated providers, an average marginal effect of 15% (95% CI, 2%-29%). The difference was even greater (average marginal effect 20%, 95% CI, 4%-35%) when children and patients over 65 were excluded.
Consistent, high-quality care and antibiotic stewardship are needed in all settings
Three experts who were not involved in the study commented on the study.
Joshua W. Elder, MD, MPH, MHS, medical director of Telehealth Express Care (direct-to-consumer telemedicine) at UC Davis Health in Sacramento, Calif., said, “An important unanswered question is how factors such as communication (policy and procedures, practice guidelines), connection (electronic health records), and reimbursement and incentives that health system and vendor-based providers received impacted this outcome.
“As the volume of virtual practices grows, most health systems will need to create a hybrid between health-system-employed and vendor-and/or-payer-supplied physicians,” he added. “Finding ways to create similar quality and outcomes will be essential in the evolving digital health infrastructure being developed.”
Charles Teixeira, DO, an infectious disease specialist at the Medical University of South Carolina in Charleston, said that this study highlighted the need to consistently provide high-quality, evidence-based care regardless of the encounter setting.
“It was important to compare the prescribing practices for commonly used medications, especially those as important as antibiotics,” he added. “Overprescribing antibiotics can have a progressive, long-term effect on a community and increase the risk for patients to develop multidrug-resistant bacteria.”
Jeffrey A. Linder, MD, MPH, the chief of general internal medicine and geriatrics in the department of medicine at Northwestern University in Chicago, commended the authors for investigating the quality of telehealth.
“The major limitation,” he found, “is that the investigators lumped all ARI visits – including those that are potentially antibiotic appropriate (e.g., otitis media, pharyngitis, sinusitis), those that are non–antibiotic appropriate (e.g., bronchitis, influenza, laryngitis, URI, viral syndrome), and those that are nonspecific symptoms (e.g., cough, congestion, fever, sore throat) – into the same category.”
No clinical information was collected or presented that would enable the reader to tell if these two groups of physicians were evaluating different patient populations or even if they just diagnosed patients differently,” he added.
“Our study did not delve into why we saw the difference,” Dr. Li explained. “Exploring potential reasons further will have important implications for how to optimally deliver care via telehealth.”
All authors and independent experts have disclosed no relevant financial relationships. The study received no financial support.
A version of this article first appeared on Medscape.com.
FROM JOURNAL OF TELEMEDICINE AND TELECARE
WHO issues new TB guidelines for children and adolescents
The World Health Organization now recommends shortened treatment for children with mild tuberculosis, as well as two oral TB treatments (bedaquiline and delamanid) for use in children of all ages. The updated guidelines for TB management in children and adolescents were announced March 21 ahead of World Tuberculosis Day on March 24.
The agency also called for increased investment in global TB programs, noting that in 2020, TB deaths increased for the first time in over a decade. “We cannot falter in our commitment to reach and save every man, woman, child, family, and community impacted by this deadly disease,” said Tereza Kasaeva, MD, PhD, director of the WHO Global Tuberculosis Programme during a press conference.
TB is the 13th-leading cause of death and the second top infectious killer after COVID-19, with more than 4,100 people dying from TB every day. WHO estimates that 1.1 million children fall ill with TB each year.
Calls for investment
The increase in TB deaths from 1.4 million in 2019 to 1.5 million in 2020 was coupled with a decrease in funding. From 2019-2020, global spending for TB diagnostic, treatment, and prevention services fell from $5.8 billion to $5.3 billion. This is less than half of the $13 billion target funding amount for 2022, Dr. Kasaeva said.
Efforts to expand access to TB care have fallen short mainly because of this lack of funding, especially for children. In 2020, about 63% of children under 15 years of age with TB either did not receive or were not reported to have access to TB diagnosis and treatment services, which rose to 72% in children under age 5. Almost two-thirds of children under age 5 also did not receive TB preventive treatment in 2022, according to WHO statistics.
The socioeconomic ramifications of the COVID-19 pandemic as well as ongoing conflict in Eastern Europe, Africa, and the Middle East have “further exacerbated the situation,” Dr. Kasaeva said. “This conveys the urgent need to dramatically increase investments to ramp up the fight against TB and achieve commitments to end TB made by global leaders.”
Dr. Kasaeva laid out WHO’s main points for global investment in TB care:
- Increase domestic and international funding to close gaps in TB research and program implementation. For countries with smaller economies, increased international investment will be necessary in the short or medium term to help regain progress.
- Double funding for TB research, including vaccines.
- Invest in sustaining TB programs and services during the COVID-19 pandemic and ongoing crises so care is not disrupted.
New guidelines
Dr. Kasaeva also noted that adoption of WHO’s new guidelines for children and adolescents should be fast-tracked to improve access to and quality of care. The updates include:
- Rapid molecular tests called Xpert Ultra should be used as the initial test for TB in children and adolescents.
- Diagnostic testing can now include noninvasive specimens, like stool samples.
- Children with mild TB can be treated with a , rather than 6 months. This shortened regimen will allow children to return to school faster and save money for families and the health care system, said Kerri Viney, MD, PhD, a team lead for the WHO Tuberculosis Programme, with a focus on vulnerable populations, including children. She presented the new guidelines during the WHO press conference.
- The recommended treatment regimen for TB meningitis has also been shortened from 12 to 6 months.
Two oral medications for drug-resistant TB (bedaquiline and delamanid) are now recommended for use in children of all ages. “There is no longer a need for painful injections that can have serious side effects, including deafness,” Dr. Viney said.
Health systems should develop new models of decentralized and integrated TB care to bring TB care closer to where children live.
The guidelines are available on the WHO website.
“The WHO guidelines issued today are a game changer for children and adolescents with TB,” Dr. Kasaeva said. The next step is assisting countries in implementing these updates so that children and adolescents globally have access to high quality TB care,” Dr. Viney added. “We have the policy recommendations. We have the implementation guidance, we have child-friendly formulations of TB medicines,” she said. “Let us not wait any longer. Let us invest to end TB in children and adolescents.”
A version of this article first appeared on Medscape.com.
The World Health Organization now recommends shortened treatment for children with mild tuberculosis, as well as two oral TB treatments (bedaquiline and delamanid) for use in children of all ages. The updated guidelines for TB management in children and adolescents were announced March 21 ahead of World Tuberculosis Day on March 24.
The agency also called for increased investment in global TB programs, noting that in 2020, TB deaths increased for the first time in over a decade. “We cannot falter in our commitment to reach and save every man, woman, child, family, and community impacted by this deadly disease,” said Tereza Kasaeva, MD, PhD, director of the WHO Global Tuberculosis Programme during a press conference.
TB is the 13th-leading cause of death and the second top infectious killer after COVID-19, with more than 4,100 people dying from TB every day. WHO estimates that 1.1 million children fall ill with TB each year.
Calls for investment
The increase in TB deaths from 1.4 million in 2019 to 1.5 million in 2020 was coupled with a decrease in funding. From 2019-2020, global spending for TB diagnostic, treatment, and prevention services fell from $5.8 billion to $5.3 billion. This is less than half of the $13 billion target funding amount for 2022, Dr. Kasaeva said.
Efforts to expand access to TB care have fallen short mainly because of this lack of funding, especially for children. In 2020, about 63% of children under 15 years of age with TB either did not receive or were not reported to have access to TB diagnosis and treatment services, which rose to 72% in children under age 5. Almost two-thirds of children under age 5 also did not receive TB preventive treatment in 2022, according to WHO statistics.
The socioeconomic ramifications of the COVID-19 pandemic as well as ongoing conflict in Eastern Europe, Africa, and the Middle East have “further exacerbated the situation,” Dr. Kasaeva said. “This conveys the urgent need to dramatically increase investments to ramp up the fight against TB and achieve commitments to end TB made by global leaders.”
Dr. Kasaeva laid out WHO’s main points for global investment in TB care:
- Increase domestic and international funding to close gaps in TB research and program implementation. For countries with smaller economies, increased international investment will be necessary in the short or medium term to help regain progress.
- Double funding for TB research, including vaccines.
- Invest in sustaining TB programs and services during the COVID-19 pandemic and ongoing crises so care is not disrupted.
New guidelines
Dr. Kasaeva also noted that adoption of WHO’s new guidelines for children and adolescents should be fast-tracked to improve access to and quality of care. The updates include:
- Rapid molecular tests called Xpert Ultra should be used as the initial test for TB in children and adolescents.
- Diagnostic testing can now include noninvasive specimens, like stool samples.
- Children with mild TB can be treated with a , rather than 6 months. This shortened regimen will allow children to return to school faster and save money for families and the health care system, said Kerri Viney, MD, PhD, a team lead for the WHO Tuberculosis Programme, with a focus on vulnerable populations, including children. She presented the new guidelines during the WHO press conference.
- The recommended treatment regimen for TB meningitis has also been shortened from 12 to 6 months.
Two oral medications for drug-resistant TB (bedaquiline and delamanid) are now recommended for use in children of all ages. “There is no longer a need for painful injections that can have serious side effects, including deafness,” Dr. Viney said.
Health systems should develop new models of decentralized and integrated TB care to bring TB care closer to where children live.
The guidelines are available on the WHO website.
“The WHO guidelines issued today are a game changer for children and adolescents with TB,” Dr. Kasaeva said. The next step is assisting countries in implementing these updates so that children and adolescents globally have access to high quality TB care,” Dr. Viney added. “We have the policy recommendations. We have the implementation guidance, we have child-friendly formulations of TB medicines,” she said. “Let us not wait any longer. Let us invest to end TB in children and adolescents.”
A version of this article first appeared on Medscape.com.
The World Health Organization now recommends shortened treatment for children with mild tuberculosis, as well as two oral TB treatments (bedaquiline and delamanid) for use in children of all ages. The updated guidelines for TB management in children and adolescents were announced March 21 ahead of World Tuberculosis Day on March 24.
The agency also called for increased investment in global TB programs, noting that in 2020, TB deaths increased for the first time in over a decade. “We cannot falter in our commitment to reach and save every man, woman, child, family, and community impacted by this deadly disease,” said Tereza Kasaeva, MD, PhD, director of the WHO Global Tuberculosis Programme during a press conference.
TB is the 13th-leading cause of death and the second top infectious killer after COVID-19, with more than 4,100 people dying from TB every day. WHO estimates that 1.1 million children fall ill with TB each year.
Calls for investment
The increase in TB deaths from 1.4 million in 2019 to 1.5 million in 2020 was coupled with a decrease in funding. From 2019-2020, global spending for TB diagnostic, treatment, and prevention services fell from $5.8 billion to $5.3 billion. This is less than half of the $13 billion target funding amount for 2022, Dr. Kasaeva said.
Efforts to expand access to TB care have fallen short mainly because of this lack of funding, especially for children. In 2020, about 63% of children under 15 years of age with TB either did not receive or were not reported to have access to TB diagnosis and treatment services, which rose to 72% in children under age 5. Almost two-thirds of children under age 5 also did not receive TB preventive treatment in 2022, according to WHO statistics.
The socioeconomic ramifications of the COVID-19 pandemic as well as ongoing conflict in Eastern Europe, Africa, and the Middle East have “further exacerbated the situation,” Dr. Kasaeva said. “This conveys the urgent need to dramatically increase investments to ramp up the fight against TB and achieve commitments to end TB made by global leaders.”
Dr. Kasaeva laid out WHO’s main points for global investment in TB care:
- Increase domestic and international funding to close gaps in TB research and program implementation. For countries with smaller economies, increased international investment will be necessary in the short or medium term to help regain progress.
- Double funding for TB research, including vaccines.
- Invest in sustaining TB programs and services during the COVID-19 pandemic and ongoing crises so care is not disrupted.
New guidelines
Dr. Kasaeva also noted that adoption of WHO’s new guidelines for children and adolescents should be fast-tracked to improve access to and quality of care. The updates include:
- Rapid molecular tests called Xpert Ultra should be used as the initial test for TB in children and adolescents.
- Diagnostic testing can now include noninvasive specimens, like stool samples.
- Children with mild TB can be treated with a , rather than 6 months. This shortened regimen will allow children to return to school faster and save money for families and the health care system, said Kerri Viney, MD, PhD, a team lead for the WHO Tuberculosis Programme, with a focus on vulnerable populations, including children. She presented the new guidelines during the WHO press conference.
- The recommended treatment regimen for TB meningitis has also been shortened from 12 to 6 months.
Two oral medications for drug-resistant TB (bedaquiline and delamanid) are now recommended for use in children of all ages. “There is no longer a need for painful injections that can have serious side effects, including deafness,” Dr. Viney said.
Health systems should develop new models of decentralized and integrated TB care to bring TB care closer to where children live.
The guidelines are available on the WHO website.
“The WHO guidelines issued today are a game changer for children and adolescents with TB,” Dr. Kasaeva said. The next step is assisting countries in implementing these updates so that children and adolescents globally have access to high quality TB care,” Dr. Viney added. “We have the policy recommendations. We have the implementation guidance, we have child-friendly formulations of TB medicines,” she said. “Let us not wait any longer. Let us invest to end TB in children and adolescents.”
A version of this article first appeared on Medscape.com.
COVID-19 doesn’t spike A1c levels
Key takeaways
Results from a retrospective, observational, case-control study of more than 20,000 people from a single U.S. medical center showed a statistically significant but clinically insignificant increase in A1c in people following COVID-19 infection, in both those with and without diabetes.
After people received a diagnosis of COVID-19 infection, they were 40% more likely to also receive a diagnosis of type 2 diabetes, compared with people who tested negative for COVID-19, a difference that was significant and could be explained by the increased medical care received by people who test positive for COVID-19.
The risk of incident diabetic ketoacidosis (DKA) among people who tested positive for COVID-19 was significantly higher among those with pre-existing type 2 diabetes, those using insulin, and among Black individuals.
Why this matters
The authors said that their study is the first report of evidence that infection with COVID-19 affects A1c levels in a large, real-world clinical cohort.
Until now, the impact of COVID-19 infection on A1c remained unclear. Results from previous studies indicated that COVID-19 infection may increase A1c levels, but the studied cohorts were small and lacked uninfected controls.
The current study included 8,755 people infected with COVID-19, had data from both before and after the infection on diabetes status and A1c levels, and also included many matched, uninfected people who served as controls.
Study design
Data came from a Cleveland Clinic registry that included 81,093 people who tested positive for COVID-19 between March 2020 and May 2021 and 153,034 matched individuals who tested negative for COVID-19 during the same period.
The researchers retrospectively selected patients with an A1c recorded within 12 months before their COVID-19 test, as well as a second A1c value recorded within 12 months after COVID-19 testing. This produced a study cohort of 8,755 COVID-positive people and 11,998 matched people who tested negative for COVID-19.
To evaluate the risk of DKA onset after COVID-19 infection, the authors identified two sub-cohorts that excluded those with a history of DKA. The sub-cohorts were 701 people with type 1 diabetes and 21,830 with type 2 diabetes.
Key results
The investigators found a statistically significant but clinically insignificant A1c increase following a positive COVID-19 test, an average A1c increase of 0.06 percentage points. Those who tested negative for COVID-19 had a clinically insignificant change in their average A1c level that was of borderline statistical significance, an average increase of 0.02 percentage points (P = .05).
The statistically significant but clinically insignificant increase in A1c following infection with COVID-19 was similar in people with and without type 2 diabetes prior to infection.
In patients with type 2 diabetes who became infected with COVID-19, the researchers saw significant positive associations between higher A1c levels before infection and time to hospitalization (hazard ratio, 1.07), need for assisted breathing (HR, 1.06), and ICU admission (HR, 1.07).
Following a COVID-19 infection, people were 40% more likely to receive a diagnosis of incident type 2 diabetes, compared with matched uninfected people. The authors said a possible explanation is that after diagnosis of COVID-19, infected people in general received more intensified care that led to better identification of those with underlying type 2 diabetes.
The 701 people included with pre-existing type 1 diabetes showed no significant difference in their rate of developing DKA between those infected and not infected with COVID-19.
Among the 21,830 people with pre-existing type 2 diabetes, the DKA risk was a significant 35% greater for those who were infected with COVID-19, compared with those who were uninfected. The magnitude of this increased relative risk was even higher among the patients with type 2 diabetes who used insulin as part of their treatment.
The difference in DKA risk didn’t differ between Black and White patients who were not infected with COVID-19, but among those infected by COVID-19, Black patients were more than twice as likely to be diagnosed with DKA, compared with White patients, a significant difference.
Black patients with type 2 diabetes who became infected with COVID-19 had a significant (63%) increased rate of DKA compared with Black patients with type 2 diabetes who remained uninfected.
Limitations
The study included patients with A1c measurements made up to 12 months prior to their COVID-19 test, and hence comorbid conditions, medication changes during this period, or other factors may have affected subsequent A1c levels. To address this, the authors also assessed outcomes at 3- and 6-month intervals, which produced results consistent with the 12-month findings.
The researchers did not have A1c values for many of the more than 234,000 people in the entire registry who underwent COVID-19 testing from March 2020-May 2021 at the Cleveland Clinic, omissions that may have biased the study cohort.
This was a single-center study. Some patients may have received care outside of the center, hence records of those episodes could not be included.
Disclosures
The study received no commercial funding. Four authors received consulting and speaker honoraria and research funding from AstraZeneca, Bayer, Boehringer Ingelheim, Corcept Therapeutics, Diasome, Eli Lilly, Merck, Novo Nordisk, and Sanofi. Three authors have intellectual property related to treatment decisionmaking in the context of type 2 diabetes.
This is a summary of a preprint research study “Impacts of COVID-19 on glycemia and risk of diabetic ketoacidosis,” written by researchers at the Cleveland Clinic on medRxiv. The study has not yet been peer reviewed. The full text of the study can be found on medRxiv.org.
A version of this article first appeared on Medscape.com.
Key takeaways
Results from a retrospective, observational, case-control study of more than 20,000 people from a single U.S. medical center showed a statistically significant but clinically insignificant increase in A1c in people following COVID-19 infection, in both those with and without diabetes.
After people received a diagnosis of COVID-19 infection, they were 40% more likely to also receive a diagnosis of type 2 diabetes, compared with people who tested negative for COVID-19, a difference that was significant and could be explained by the increased medical care received by people who test positive for COVID-19.
The risk of incident diabetic ketoacidosis (DKA) among people who tested positive for COVID-19 was significantly higher among those with pre-existing type 2 diabetes, those using insulin, and among Black individuals.
Why this matters
The authors said that their study is the first report of evidence that infection with COVID-19 affects A1c levels in a large, real-world clinical cohort.
Until now, the impact of COVID-19 infection on A1c remained unclear. Results from previous studies indicated that COVID-19 infection may increase A1c levels, but the studied cohorts were small and lacked uninfected controls.
The current study included 8,755 people infected with COVID-19, had data from both before and after the infection on diabetes status and A1c levels, and also included many matched, uninfected people who served as controls.
Study design
Data came from a Cleveland Clinic registry that included 81,093 people who tested positive for COVID-19 between March 2020 and May 2021 and 153,034 matched individuals who tested negative for COVID-19 during the same period.
The researchers retrospectively selected patients with an A1c recorded within 12 months before their COVID-19 test, as well as a second A1c value recorded within 12 months after COVID-19 testing. This produced a study cohort of 8,755 COVID-positive people and 11,998 matched people who tested negative for COVID-19.
To evaluate the risk of DKA onset after COVID-19 infection, the authors identified two sub-cohorts that excluded those with a history of DKA. The sub-cohorts were 701 people with type 1 diabetes and 21,830 with type 2 diabetes.
Key results
The investigators found a statistically significant but clinically insignificant A1c increase following a positive COVID-19 test, an average A1c increase of 0.06 percentage points. Those who tested negative for COVID-19 had a clinically insignificant change in their average A1c level that was of borderline statistical significance, an average increase of 0.02 percentage points (P = .05).
The statistically significant but clinically insignificant increase in A1c following infection with COVID-19 was similar in people with and without type 2 diabetes prior to infection.
In patients with type 2 diabetes who became infected with COVID-19, the researchers saw significant positive associations between higher A1c levels before infection and time to hospitalization (hazard ratio, 1.07), need for assisted breathing (HR, 1.06), and ICU admission (HR, 1.07).
Following a COVID-19 infection, people were 40% more likely to receive a diagnosis of incident type 2 diabetes, compared with matched uninfected people. The authors said a possible explanation is that after diagnosis of COVID-19, infected people in general received more intensified care that led to better identification of those with underlying type 2 diabetes.
The 701 people included with pre-existing type 1 diabetes showed no significant difference in their rate of developing DKA between those infected and not infected with COVID-19.
Among the 21,830 people with pre-existing type 2 diabetes, the DKA risk was a significant 35% greater for those who were infected with COVID-19, compared with those who were uninfected. The magnitude of this increased relative risk was even higher among the patients with type 2 diabetes who used insulin as part of their treatment.
The difference in DKA risk didn’t differ between Black and White patients who were not infected with COVID-19, but among those infected by COVID-19, Black patients were more than twice as likely to be diagnosed with DKA, compared with White patients, a significant difference.
Black patients with type 2 diabetes who became infected with COVID-19 had a significant (63%) increased rate of DKA compared with Black patients with type 2 diabetes who remained uninfected.
Limitations
The study included patients with A1c measurements made up to 12 months prior to their COVID-19 test, and hence comorbid conditions, medication changes during this period, or other factors may have affected subsequent A1c levels. To address this, the authors also assessed outcomes at 3- and 6-month intervals, which produced results consistent with the 12-month findings.
The researchers did not have A1c values for many of the more than 234,000 people in the entire registry who underwent COVID-19 testing from March 2020-May 2021 at the Cleveland Clinic, omissions that may have biased the study cohort.
This was a single-center study. Some patients may have received care outside of the center, hence records of those episodes could not be included.
Disclosures
The study received no commercial funding. Four authors received consulting and speaker honoraria and research funding from AstraZeneca, Bayer, Boehringer Ingelheim, Corcept Therapeutics, Diasome, Eli Lilly, Merck, Novo Nordisk, and Sanofi. Three authors have intellectual property related to treatment decisionmaking in the context of type 2 diabetes.
This is a summary of a preprint research study “Impacts of COVID-19 on glycemia and risk of diabetic ketoacidosis,” written by researchers at the Cleveland Clinic on medRxiv. The study has not yet been peer reviewed. The full text of the study can be found on medRxiv.org.
A version of this article first appeared on Medscape.com.
Key takeaways
Results from a retrospective, observational, case-control study of more than 20,000 people from a single U.S. medical center showed a statistically significant but clinically insignificant increase in A1c in people following COVID-19 infection, in both those with and without diabetes.
After people received a diagnosis of COVID-19 infection, they were 40% more likely to also receive a diagnosis of type 2 diabetes, compared with people who tested negative for COVID-19, a difference that was significant and could be explained by the increased medical care received by people who test positive for COVID-19.
The risk of incident diabetic ketoacidosis (DKA) among people who tested positive for COVID-19 was significantly higher among those with pre-existing type 2 diabetes, those using insulin, and among Black individuals.
Why this matters
The authors said that their study is the first report of evidence that infection with COVID-19 affects A1c levels in a large, real-world clinical cohort.
Until now, the impact of COVID-19 infection on A1c remained unclear. Results from previous studies indicated that COVID-19 infection may increase A1c levels, but the studied cohorts were small and lacked uninfected controls.
The current study included 8,755 people infected with COVID-19, had data from both before and after the infection on diabetes status and A1c levels, and also included many matched, uninfected people who served as controls.
Study design
Data came from a Cleveland Clinic registry that included 81,093 people who tested positive for COVID-19 between March 2020 and May 2021 and 153,034 matched individuals who tested negative for COVID-19 during the same period.
The researchers retrospectively selected patients with an A1c recorded within 12 months before their COVID-19 test, as well as a second A1c value recorded within 12 months after COVID-19 testing. This produced a study cohort of 8,755 COVID-positive people and 11,998 matched people who tested negative for COVID-19.
To evaluate the risk of DKA onset after COVID-19 infection, the authors identified two sub-cohorts that excluded those with a history of DKA. The sub-cohorts were 701 people with type 1 diabetes and 21,830 with type 2 diabetes.
Key results
The investigators found a statistically significant but clinically insignificant A1c increase following a positive COVID-19 test, an average A1c increase of 0.06 percentage points. Those who tested negative for COVID-19 had a clinically insignificant change in their average A1c level that was of borderline statistical significance, an average increase of 0.02 percentage points (P = .05).
The statistically significant but clinically insignificant increase in A1c following infection with COVID-19 was similar in people with and without type 2 diabetes prior to infection.
In patients with type 2 diabetes who became infected with COVID-19, the researchers saw significant positive associations between higher A1c levels before infection and time to hospitalization (hazard ratio, 1.07), need for assisted breathing (HR, 1.06), and ICU admission (HR, 1.07).
Following a COVID-19 infection, people were 40% more likely to receive a diagnosis of incident type 2 diabetes, compared with matched uninfected people. The authors said a possible explanation is that after diagnosis of COVID-19, infected people in general received more intensified care that led to better identification of those with underlying type 2 diabetes.
The 701 people included with pre-existing type 1 diabetes showed no significant difference in their rate of developing DKA between those infected and not infected with COVID-19.
Among the 21,830 people with pre-existing type 2 diabetes, the DKA risk was a significant 35% greater for those who were infected with COVID-19, compared with those who were uninfected. The magnitude of this increased relative risk was even higher among the patients with type 2 diabetes who used insulin as part of their treatment.
The difference in DKA risk didn’t differ between Black and White patients who were not infected with COVID-19, but among those infected by COVID-19, Black patients were more than twice as likely to be diagnosed with DKA, compared with White patients, a significant difference.
Black patients with type 2 diabetes who became infected with COVID-19 had a significant (63%) increased rate of DKA compared with Black patients with type 2 diabetes who remained uninfected.
Limitations
The study included patients with A1c measurements made up to 12 months prior to their COVID-19 test, and hence comorbid conditions, medication changes during this period, or other factors may have affected subsequent A1c levels. To address this, the authors also assessed outcomes at 3- and 6-month intervals, which produced results consistent with the 12-month findings.
The researchers did not have A1c values for many of the more than 234,000 people in the entire registry who underwent COVID-19 testing from March 2020-May 2021 at the Cleveland Clinic, omissions that may have biased the study cohort.
This was a single-center study. Some patients may have received care outside of the center, hence records of those episodes could not be included.
Disclosures
The study received no commercial funding. Four authors received consulting and speaker honoraria and research funding from AstraZeneca, Bayer, Boehringer Ingelheim, Corcept Therapeutics, Diasome, Eli Lilly, Merck, Novo Nordisk, and Sanofi. Three authors have intellectual property related to treatment decisionmaking in the context of type 2 diabetes.
This is a summary of a preprint research study “Impacts of COVID-19 on glycemia and risk of diabetic ketoacidosis,” written by researchers at the Cleveland Clinic on medRxiv. The study has not yet been peer reviewed. The full text of the study can be found on medRxiv.org.
A version of this article first appeared on Medscape.com.
Yes, Russian docs should be shut out of medical associations, says ethicist
This transcript has been edited for clarity.
Hi. I’m Art Caplan. I’m at the division of medical ethics at the NYU Grossman School of Medicine.
There are many difficult moral issues that are being fueled by the terrible war that Russia is waging against Ukraine. I think there is no way to justify anything that the Russians are doing. Ukraine did not do anything to violate Russian integrity, Russian territorial integrity, or anything by way of being aggressive toward Russia.
Russia decided at some point it wanted the Ukraine back. Putin has a dream, as the long-standing leader of Russia, to restore the Soviet empire, and Ukraine is top of the list of the places that he wants back for a variety of reasons.
We’re not here to debate the merits and demerits of this terrible act of war.
The European Society of Cardiology made a decision very recently to drop, as members, both Russia and Belarus, Russia’s ally in this aggressive war against Ukraine. They basically found it intolerable to have business as usual with these subsidiary cardiology societies as part of the ongoing activities of the European group.
The sole goal of this overarching European group is to reduce the health burden of cardiovascular disease. It doesn’t have political goals. It doesn’t have much to say about anything other than, “Let’s get evidence-based medicine used to try and prevent heart disease or treat heart disease.” So there’s noble intent.
Many of its members asked, “What are we doing in politics? Why are we punishing Russian and Belarussian cardiologists, acting as if somehow they are responsible for what the Russian army is doing or for what Putin has decided to do? Why are we acting against them? They are just trying to fight heart disease. That’s a legitimate goal for any doctor, public health official, or scientist.” They didn’t see, as members, why this exclusion had taken place.
I believe the exclusion is appropriate and some of the membership, obviously, in the European Society of Cardiology, agrees. It’s not because they’re holding doctors or scientists directly accountable for Putin’s war crimes, ethnic cleansing assault, or bombing and shelling of hospitals, maternity hospitals, and civilians.
They understand that these scientists and doctors have little to do with such things, but we are in a new form of warfare, and that warfare is basically economic and sociologic: turning Russia, as an inexcusably aggressive state, into a pariah.
The reason to break the ties is that that is the way to bring pressure upon Putin and his kleptocratic, oligarchic advisers to stop the attack, to try and bring down their economy, to say, “Business is not going to go on as usual. You will be excluded from normal scientific and medical commerce. We’re not going to be holding conferences or exchanging ideas,” and in my view, extending it to say, “We’re not taking your papers, we’re not publishing anything you do. We’re not even having you speak at our meetings until this war, this aggressive invasion, and these war crimes come to a halt.”
There is actually a basis for this action. It isn’t in the organization’s own bylaws, which as I said, are very simple — reduce cardiovascular disease burden — but they are a member of a broader group, the Biomedical Alliance in Europe, which does have a very explicit code of ethics.
I’m going to read you a little bit from that code. It says healthcare organizations should uphold and promote equality, diversity and inclusion, accountability, transparency, and equality. They also say that all members, including the European Society of Cardiology, should be committed both to the Declaration of Helsinki, a fundamental medical ethics document, and the Declaration of Geneva. These rules refer to the highest respect of human beings, responsible resource allocation, and preservation of the environment, among other things.
What the organization is doing is consistent with the code of ethics that the broader organization of all the medical societies of Europe say that these individual groups should be doing. You can’t collaborate with war criminals. You can’t act as if business as usual is going on. That’s not inclusive. That’s not respect for diversity.
I think the Ukrainian medical societies of cardiology and other specialties would find it grimly ironic to say that keeping Russian and Belarus members makes sense, given what’s going on in their country and what is happening to them. They’re under attack. They’re being killed. Their healthcare institutions are being indiscriminately shelled and bombed.
It’s very hard — and I understand that — to say we’re going to punish scientists. We’re going to, perhaps, even cause public health problems in Russia because we’re not going to collaborate right now with doctors and scientists in cardiology or any other medical specialty. I think it’s what has to be done.
We’re in a new era of trying to combat what is basically organized, international ethnic terrorism, complete with war crimes. We fight financially. We fight by isolating. We fight by excluding. It’s painful. It’s difficult. It’s somewhat unfair to individuals.
Only through that kind of pain are we going to get the kind of pressure that will achieve justice. I think that is a goal that we have to commend the European Society of Cardiology for honoring.
Dr. Caplan is director of the division of medical ethics at New York University. He is the author or editor of 35 books and 750 peer-reviewed articles as well as a frequent commentator in the media on bioethical issues. He has served as a director, officer, partner, employee, adviser, consultant, or trustee for Johnson & Johnson’s Panel for Compassionate Drug Use (an unpaid position), and is a contributing author and adviser for Medscape. A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
Hi. I’m Art Caplan. I’m at the division of medical ethics at the NYU Grossman School of Medicine.
There are many difficult moral issues that are being fueled by the terrible war that Russia is waging against Ukraine. I think there is no way to justify anything that the Russians are doing. Ukraine did not do anything to violate Russian integrity, Russian territorial integrity, or anything by way of being aggressive toward Russia.
Russia decided at some point it wanted the Ukraine back. Putin has a dream, as the long-standing leader of Russia, to restore the Soviet empire, and Ukraine is top of the list of the places that he wants back for a variety of reasons.
We’re not here to debate the merits and demerits of this terrible act of war.
The European Society of Cardiology made a decision very recently to drop, as members, both Russia and Belarus, Russia’s ally in this aggressive war against Ukraine. They basically found it intolerable to have business as usual with these subsidiary cardiology societies as part of the ongoing activities of the European group.
The sole goal of this overarching European group is to reduce the health burden of cardiovascular disease. It doesn’t have political goals. It doesn’t have much to say about anything other than, “Let’s get evidence-based medicine used to try and prevent heart disease or treat heart disease.” So there’s noble intent.
Many of its members asked, “What are we doing in politics? Why are we punishing Russian and Belarussian cardiologists, acting as if somehow they are responsible for what the Russian army is doing or for what Putin has decided to do? Why are we acting against them? They are just trying to fight heart disease. That’s a legitimate goal for any doctor, public health official, or scientist.” They didn’t see, as members, why this exclusion had taken place.
I believe the exclusion is appropriate and some of the membership, obviously, in the European Society of Cardiology, agrees. It’s not because they’re holding doctors or scientists directly accountable for Putin’s war crimes, ethnic cleansing assault, or bombing and shelling of hospitals, maternity hospitals, and civilians.
They understand that these scientists and doctors have little to do with such things, but we are in a new form of warfare, and that warfare is basically economic and sociologic: turning Russia, as an inexcusably aggressive state, into a pariah.
The reason to break the ties is that that is the way to bring pressure upon Putin and his kleptocratic, oligarchic advisers to stop the attack, to try and bring down their economy, to say, “Business is not going to go on as usual. You will be excluded from normal scientific and medical commerce. We’re not going to be holding conferences or exchanging ideas,” and in my view, extending it to say, “We’re not taking your papers, we’re not publishing anything you do. We’re not even having you speak at our meetings until this war, this aggressive invasion, and these war crimes come to a halt.”
There is actually a basis for this action. It isn’t in the organization’s own bylaws, which as I said, are very simple — reduce cardiovascular disease burden — but they are a member of a broader group, the Biomedical Alliance in Europe, which does have a very explicit code of ethics.
I’m going to read you a little bit from that code. It says healthcare organizations should uphold and promote equality, diversity and inclusion, accountability, transparency, and equality. They also say that all members, including the European Society of Cardiology, should be committed both to the Declaration of Helsinki, a fundamental medical ethics document, and the Declaration of Geneva. These rules refer to the highest respect of human beings, responsible resource allocation, and preservation of the environment, among other things.
What the organization is doing is consistent with the code of ethics that the broader organization of all the medical societies of Europe say that these individual groups should be doing. You can’t collaborate with war criminals. You can’t act as if business as usual is going on. That’s not inclusive. That’s not respect for diversity.
I think the Ukrainian medical societies of cardiology and other specialties would find it grimly ironic to say that keeping Russian and Belarus members makes sense, given what’s going on in their country and what is happening to them. They’re under attack. They’re being killed. Their healthcare institutions are being indiscriminately shelled and bombed.
It’s very hard — and I understand that — to say we’re going to punish scientists. We’re going to, perhaps, even cause public health problems in Russia because we’re not going to collaborate right now with doctors and scientists in cardiology or any other medical specialty. I think it’s what has to be done.
We’re in a new era of trying to combat what is basically organized, international ethnic terrorism, complete with war crimes. We fight financially. We fight by isolating. We fight by excluding. It’s painful. It’s difficult. It’s somewhat unfair to individuals.
Only through that kind of pain are we going to get the kind of pressure that will achieve justice. I think that is a goal that we have to commend the European Society of Cardiology for honoring.
Dr. Caplan is director of the division of medical ethics at New York University. He is the author or editor of 35 books and 750 peer-reviewed articles as well as a frequent commentator in the media on bioethical issues. He has served as a director, officer, partner, employee, adviser, consultant, or trustee for Johnson & Johnson’s Panel for Compassionate Drug Use (an unpaid position), and is a contributing author and adviser for Medscape. A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
Hi. I’m Art Caplan. I’m at the division of medical ethics at the NYU Grossman School of Medicine.
There are many difficult moral issues that are being fueled by the terrible war that Russia is waging against Ukraine. I think there is no way to justify anything that the Russians are doing. Ukraine did not do anything to violate Russian integrity, Russian territorial integrity, or anything by way of being aggressive toward Russia.
Russia decided at some point it wanted the Ukraine back. Putin has a dream, as the long-standing leader of Russia, to restore the Soviet empire, and Ukraine is top of the list of the places that he wants back for a variety of reasons.
We’re not here to debate the merits and demerits of this terrible act of war.
The European Society of Cardiology made a decision very recently to drop, as members, both Russia and Belarus, Russia’s ally in this aggressive war against Ukraine. They basically found it intolerable to have business as usual with these subsidiary cardiology societies as part of the ongoing activities of the European group.
The sole goal of this overarching European group is to reduce the health burden of cardiovascular disease. It doesn’t have political goals. It doesn’t have much to say about anything other than, “Let’s get evidence-based medicine used to try and prevent heart disease or treat heart disease.” So there’s noble intent.
Many of its members asked, “What are we doing in politics? Why are we punishing Russian and Belarussian cardiologists, acting as if somehow they are responsible for what the Russian army is doing or for what Putin has decided to do? Why are we acting against them? They are just trying to fight heart disease. That’s a legitimate goal for any doctor, public health official, or scientist.” They didn’t see, as members, why this exclusion had taken place.
I believe the exclusion is appropriate and some of the membership, obviously, in the European Society of Cardiology, agrees. It’s not because they’re holding doctors or scientists directly accountable for Putin’s war crimes, ethnic cleansing assault, or bombing and shelling of hospitals, maternity hospitals, and civilians.
They understand that these scientists and doctors have little to do with such things, but we are in a new form of warfare, and that warfare is basically economic and sociologic: turning Russia, as an inexcusably aggressive state, into a pariah.
The reason to break the ties is that that is the way to bring pressure upon Putin and his kleptocratic, oligarchic advisers to stop the attack, to try and bring down their economy, to say, “Business is not going to go on as usual. You will be excluded from normal scientific and medical commerce. We’re not going to be holding conferences or exchanging ideas,” and in my view, extending it to say, “We’re not taking your papers, we’re not publishing anything you do. We’re not even having you speak at our meetings until this war, this aggressive invasion, and these war crimes come to a halt.”
There is actually a basis for this action. It isn’t in the organization’s own bylaws, which as I said, are very simple — reduce cardiovascular disease burden — but they are a member of a broader group, the Biomedical Alliance in Europe, which does have a very explicit code of ethics.
I’m going to read you a little bit from that code. It says healthcare organizations should uphold and promote equality, diversity and inclusion, accountability, transparency, and equality. They also say that all members, including the European Society of Cardiology, should be committed both to the Declaration of Helsinki, a fundamental medical ethics document, and the Declaration of Geneva. These rules refer to the highest respect of human beings, responsible resource allocation, and preservation of the environment, among other things.
What the organization is doing is consistent with the code of ethics that the broader organization of all the medical societies of Europe say that these individual groups should be doing. You can’t collaborate with war criminals. You can’t act as if business as usual is going on. That’s not inclusive. That’s not respect for diversity.
I think the Ukrainian medical societies of cardiology and other specialties would find it grimly ironic to say that keeping Russian and Belarus members makes sense, given what’s going on in their country and what is happening to them. They’re under attack. They’re being killed. Their healthcare institutions are being indiscriminately shelled and bombed.
It’s very hard — and I understand that — to say we’re going to punish scientists. We’re going to, perhaps, even cause public health problems in Russia because we’re not going to collaborate right now with doctors and scientists in cardiology or any other medical specialty. I think it’s what has to be done.
We’re in a new era of trying to combat what is basically organized, international ethnic terrorism, complete with war crimes. We fight financially. We fight by isolating. We fight by excluding. It’s painful. It’s difficult. It’s somewhat unfair to individuals.
Only through that kind of pain are we going to get the kind of pressure that will achieve justice. I think that is a goal that we have to commend the European Society of Cardiology for honoring.
Dr. Caplan is director of the division of medical ethics at New York University. He is the author or editor of 35 books and 750 peer-reviewed articles as well as a frequent commentator in the media on bioethical issues. He has served as a director, officer, partner, employee, adviser, consultant, or trustee for Johnson & Johnson’s Panel for Compassionate Drug Use (an unpaid position), and is a contributing author and adviser for Medscape. A version of this article first appeared on Medscape.com.
U.S. health officials tracking COVID-19 increase in U.K.
according to NPR.
Daily cases counts have increased 38% in the past week, according to the latest data from the U.K. Health Security Agency. Hospitalizations are up about 25% as well.
“Over the last year or so, what happens in the U.K. usually happens here a few weeks later,” Anthony S. Fauci, MD, director of the National Institute of Allergy and Infectious Diseases, told NPR.
“And right now, the U.K. is seeing somewhat of a rebound in cases,” he said.
Health officials in the United Kingdom have noted the latest increase is likely due to the contagious BA.2 Omicron subvariant, the recent loosening of coronavirus restrictions, and waning immunity from vaccinations and infections.
“All three of those factors we have here in the United States,” Dr. Fauci said. “So I would not be surprised if, in the next few weeks, we see either a plateauing … of cases or even [the curve] rebounds and slightly goes up.”
Right now, COVID-19 cases in the United Stastes have dropped to their lowest levels since July 2021, according to the latest Centers for Disease Control and Prevention data, with fewer than 30,000 daily cases. At the same time, the rate of decline in cases has slowed significantly and is beginning to plateau.
Public health experts are also pointing to wastewater surveillance data that shows an uptick in viral activity across the country. The CDC’s wastewater dashboard indicates that about 35% of sites that monitor wastewater are seeing an increase, with consistent growth in Florida, Rhode Island, and West Virginia.
“The power of wastewater surveillance is that it’s an early warning system,” Amy Kirby, the program lead for the CDC’s National Wastewater Surveillance System, told NPR.
“We are seeing evidence of increases in some communities across the country,” she said. “What looked like noise at the beginning of the week is starting to look like a true signal here at the end of the week.”
The wastewater system doesn’t distinguish between Omicron and subvariants such as BA.2. However, other CDC data has found an increase in BA.2 cases in the United States, making up about a quarter of new COVID-19 cases.
The BA.2 variant has roughly doubled each week for the last month, which means it could become the dominant coronavirus strain in the United States in coming weeks, according to USA Today. Cases appear to be spreading more quickly in the Northeast and West, making up about 39% of cases in New York and New Jersey last week.
BA.2 also accounts for nearly 39% of cases across the Northeast, including Connecticut, Maine, Massachusetts, New Hampshire, Rhode Island and Vermont, USA Today reported. In the West, which includes Arizona, California and Nevada, the subvariant makes up about 28% of new cases. In the upper West, which includes Alaska, Oregon and Washington, about 26% of cases are BA.2.
The good news is that BA.2 “doesn’t seem to evade our vaccines or immunity any more than the prior Omicron [variant]. And it doesn’t seem to lead to any more increased severity of disease,” Rochelle Walensky, MD, the CDC director, told NPR’s Morning Edition on March 18.
The effects of BA.2 will likely depend on the immunity profile in the United States, including how long it’s been since someone was vaccinated, boosted, or recovered from an infection, she said.
Health officials are watching other countries with BA.2 increases, such as Germany, Italy, and the Netherlands. Many European countries have been reporting an uptick but not implementing major restrictions or shutdowns, USA Today reported.
The BA.2 variant likely won’t lead to a major surge in severe disease or strict COVID-19 measures, Dr. Fauci told NPR, but some coronavirus protocols may need to be implemented again if cases grow dramatically.
“We must be ready to pivot and, if necessary, to go back to stricter mitigation with regard to masks,” he said.
A version of this article first appeared on WebMD.com.
according to NPR.
Daily cases counts have increased 38% in the past week, according to the latest data from the U.K. Health Security Agency. Hospitalizations are up about 25% as well.
“Over the last year or so, what happens in the U.K. usually happens here a few weeks later,” Anthony S. Fauci, MD, director of the National Institute of Allergy and Infectious Diseases, told NPR.
“And right now, the U.K. is seeing somewhat of a rebound in cases,” he said.
Health officials in the United Kingdom have noted the latest increase is likely due to the contagious BA.2 Omicron subvariant, the recent loosening of coronavirus restrictions, and waning immunity from vaccinations and infections.
“All three of those factors we have here in the United States,” Dr. Fauci said. “So I would not be surprised if, in the next few weeks, we see either a plateauing … of cases or even [the curve] rebounds and slightly goes up.”
Right now, COVID-19 cases in the United Stastes have dropped to their lowest levels since July 2021, according to the latest Centers for Disease Control and Prevention data, with fewer than 30,000 daily cases. At the same time, the rate of decline in cases has slowed significantly and is beginning to plateau.
Public health experts are also pointing to wastewater surveillance data that shows an uptick in viral activity across the country. The CDC’s wastewater dashboard indicates that about 35% of sites that monitor wastewater are seeing an increase, with consistent growth in Florida, Rhode Island, and West Virginia.
“The power of wastewater surveillance is that it’s an early warning system,” Amy Kirby, the program lead for the CDC’s National Wastewater Surveillance System, told NPR.
“We are seeing evidence of increases in some communities across the country,” she said. “What looked like noise at the beginning of the week is starting to look like a true signal here at the end of the week.”
The wastewater system doesn’t distinguish between Omicron and subvariants such as BA.2. However, other CDC data has found an increase in BA.2 cases in the United States, making up about a quarter of new COVID-19 cases.
The BA.2 variant has roughly doubled each week for the last month, which means it could become the dominant coronavirus strain in the United States in coming weeks, according to USA Today. Cases appear to be spreading more quickly in the Northeast and West, making up about 39% of cases in New York and New Jersey last week.
BA.2 also accounts for nearly 39% of cases across the Northeast, including Connecticut, Maine, Massachusetts, New Hampshire, Rhode Island and Vermont, USA Today reported. In the West, which includes Arizona, California and Nevada, the subvariant makes up about 28% of new cases. In the upper West, which includes Alaska, Oregon and Washington, about 26% of cases are BA.2.
The good news is that BA.2 “doesn’t seem to evade our vaccines or immunity any more than the prior Omicron [variant]. And it doesn’t seem to lead to any more increased severity of disease,” Rochelle Walensky, MD, the CDC director, told NPR’s Morning Edition on March 18.
The effects of BA.2 will likely depend on the immunity profile in the United States, including how long it’s been since someone was vaccinated, boosted, or recovered from an infection, she said.
Health officials are watching other countries with BA.2 increases, such as Germany, Italy, and the Netherlands. Many European countries have been reporting an uptick but not implementing major restrictions or shutdowns, USA Today reported.
The BA.2 variant likely won’t lead to a major surge in severe disease or strict COVID-19 measures, Dr. Fauci told NPR, but some coronavirus protocols may need to be implemented again if cases grow dramatically.
“We must be ready to pivot and, if necessary, to go back to stricter mitigation with regard to masks,” he said.
A version of this article first appeared on WebMD.com.
according to NPR.
Daily cases counts have increased 38% in the past week, according to the latest data from the U.K. Health Security Agency. Hospitalizations are up about 25% as well.
“Over the last year or so, what happens in the U.K. usually happens here a few weeks later,” Anthony S. Fauci, MD, director of the National Institute of Allergy and Infectious Diseases, told NPR.
“And right now, the U.K. is seeing somewhat of a rebound in cases,” he said.
Health officials in the United Kingdom have noted the latest increase is likely due to the contagious BA.2 Omicron subvariant, the recent loosening of coronavirus restrictions, and waning immunity from vaccinations and infections.
“All three of those factors we have here in the United States,” Dr. Fauci said. “So I would not be surprised if, in the next few weeks, we see either a plateauing … of cases or even [the curve] rebounds and slightly goes up.”
Right now, COVID-19 cases in the United Stastes have dropped to their lowest levels since July 2021, according to the latest Centers for Disease Control and Prevention data, with fewer than 30,000 daily cases. At the same time, the rate of decline in cases has slowed significantly and is beginning to plateau.
Public health experts are also pointing to wastewater surveillance data that shows an uptick in viral activity across the country. The CDC’s wastewater dashboard indicates that about 35% of sites that monitor wastewater are seeing an increase, with consistent growth in Florida, Rhode Island, and West Virginia.
“The power of wastewater surveillance is that it’s an early warning system,” Amy Kirby, the program lead for the CDC’s National Wastewater Surveillance System, told NPR.
“We are seeing evidence of increases in some communities across the country,” she said. “What looked like noise at the beginning of the week is starting to look like a true signal here at the end of the week.”
The wastewater system doesn’t distinguish between Omicron and subvariants such as BA.2. However, other CDC data has found an increase in BA.2 cases in the United States, making up about a quarter of new COVID-19 cases.
The BA.2 variant has roughly doubled each week for the last month, which means it could become the dominant coronavirus strain in the United States in coming weeks, according to USA Today. Cases appear to be spreading more quickly in the Northeast and West, making up about 39% of cases in New York and New Jersey last week.
BA.2 also accounts for nearly 39% of cases across the Northeast, including Connecticut, Maine, Massachusetts, New Hampshire, Rhode Island and Vermont, USA Today reported. In the West, which includes Arizona, California and Nevada, the subvariant makes up about 28% of new cases. In the upper West, which includes Alaska, Oregon and Washington, about 26% of cases are BA.2.
The good news is that BA.2 “doesn’t seem to evade our vaccines or immunity any more than the prior Omicron [variant]. And it doesn’t seem to lead to any more increased severity of disease,” Rochelle Walensky, MD, the CDC director, told NPR’s Morning Edition on March 18.
The effects of BA.2 will likely depend on the immunity profile in the United States, including how long it’s been since someone was vaccinated, boosted, or recovered from an infection, she said.
Health officials are watching other countries with BA.2 increases, such as Germany, Italy, and the Netherlands. Many European countries have been reporting an uptick but not implementing major restrictions or shutdowns, USA Today reported.
The BA.2 variant likely won’t lead to a major surge in severe disease or strict COVID-19 measures, Dr. Fauci told NPR, but some coronavirus protocols may need to be implemented again if cases grow dramatically.
“We must be ready to pivot and, if necessary, to go back to stricter mitigation with regard to masks,” he said.
A version of this article first appeared on WebMD.com.