User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
Severe outcomes increased in youth hospitalized after positive COVID-19 test
Approximately 3% of youth who tested positive for COVID-19 in an emergency department setting had severe outcomes after 2 weeks, but this risk was 0.5% among those not admitted to the hospital, based on data from more than 3,000 individuals aged 18 and younger.
In the early stages of the COVID-19 pandemic, youth younger than 18 years accounted for fewer than 5% of reported cases, but now account for approximately 25% of positive cases, wrote Anna L. Funk, PhD, of the University of Calgary, Alberta, Canada, and colleagues.
However, the risk of severe outcomes of youth with COVID-19 remains poorly understood and data from large studies are lacking, they noted.
In a prospective cohort study published in JAMA Network Open, the researchers reviewed data from 3,221 children and adolescents who were tested for COVID-19 at one of 41 emergency departments in 10 countries including Argentina, Australia, Canada, Costa Rica, Italy, New Zealand, Paraguay, Singapore, Spain, and the United States between March 2020 and June 2021. Positive infections were confirmed by polymerase chain reaction (PCR) testing. At 14 days’ follow-up after a positive test, 735 patients (22.8%), were hospitalized, 107 (3.3%) had severe outcomes, and 4 (0.12%) had died. Severe outcomes were significantly more likely in children aged 5-10 years and 10-18 years vs. less than 1 year (odds ratios, 1.60 and 2.39, respectively), and in children with a self-reported chronic illness (OR, 2.34) or a prior episode of pneumonia (OR, 3.15).
Severe outcomes were more likely in patients who presented with symptoms that started 4-7 days before seeking care, compared with those whose symptoms started 0-3 days before seeking care (OR, 2.22).
The researchers also reviewed data from a subgroup of 2,510 individuals who were discharged home from the ED after initial testing. At 14 days’ follow-up, 50 of these patients (2.0%) were hospitalized and 12 (0.5%) had severe outcomes. In addition, the researchers found that the risk of severe outcomes among hospitalized COVID-19–positive youth was nearly four times higher, compared with hospitalized youth who tested negative for COVID-19 (risk difference, 3.9%).
Previous retrospective studies of severe outcomes in children and adolescents with COVID-19 have yielded varying results, in part because of the variation in study populations, the researchers noted in their discussion of the findings. “Our study population provides a risk estimate for youths brought for ED care.” Therefore, “Our lower estimate of severe disease likely reflects our stringent definition, which required the occurrence of complications or specific invasive interventions,” they said.
The study limitations included the potential overestimation of the risk of severe outcomes because patients were recruited in the ED, the researchers noted. Other limitations included variation in regional case definitions, screening criteria, and testing capacity among different sites and time periods. “Thus, 5% of our SARS-CoV-2–positive participants were asymptomatic – most of whom were tested as they were positive contacts of known cases or as part of routine screening procedures,” they said. The findings also are not generalizable to all community EDs and did not account for variants, they added.
However, the results were strengthened by the ability to compare outcomes for children with positive tests to similar children with negative tests, and add to the literature showing an increased risk of severe outcomes for those hospitalized with positive tests, the researchers concluded.
Data may inform clinical decisions
“The data [in the current study] are concerning for severe outcomes for children even prior to the Omicron strain,” said Margaret Thew, DNP, FP-BC, of Children’s Wisconsin-Milwaukee Hospital, in an interview. “Presently, the number of children infected with the Omicron strain is much higher and hospitalizations among children are at their highest since COVID-19 began,” she said. “For medical providers caring for this population, the study sheds light on pediatric patients who may be at higher risk of severe illness when they become infected with COVID-19,” she added.
“I was surprised by how high the number of pediatric patients hospitalized (22%) and the percentage (3%) with severe disease were during this time,” given that the timeline for these data preceded the spread of the Omicron strain, said Ms. Thew. “The risk of prior pneumonia was quite surprising. I do not recall seeing prior pneumonia as a risk factor for more severe COVID-19 with children or adults,” she added.
The take-home messaging for clinicians caring for children and adolescents is the added knowledge of the risk factors for severe outcomes from COVID-19, including the 10-18 age range, chronic illness, prior pneumonia, and longer symptom duration before seeking care in the ED, Ms. Thew emphasized.
However, additional research is needed on the impact of the new strains of COVID-19 on pediatric and adolescent hospitalizations, Ms. Thew said. Research also is needed on the other illnesses that have resulted from COVID-19, including illness requiring antibiotic use or medical interventions or treatments, and on the risk of combined COVID-19 and influenza viruses, she noted.
The study was supported by the Canadian Institutes of Health Research, Alberta Innovates, the Alberta Health Services University of Calgary Clinical Research Fund, the Alberta Children’s Hospital Research Institute, the COVID-19 Research Accelerator Funding Track (CRAFT) Program at the University of California, Davis, and the Cincinnati Children’s Hospital Medical Center Division of Emergency Medicine Small Grants Program. Lead author Dr. Funk was supported by the University of Calgary Eyes-High Post-Doctoral Research Fund, but had no financial conflicts to disclose. Ms. Thew had no financial conflicts to disclose and serves on the Editorial Advisory Board of Pediatric News.
Approximately 3% of youth who tested positive for COVID-19 in an emergency department setting had severe outcomes after 2 weeks, but this risk was 0.5% among those not admitted to the hospital, based on data from more than 3,000 individuals aged 18 and younger.
In the early stages of the COVID-19 pandemic, youth younger than 18 years accounted for fewer than 5% of reported cases, but now account for approximately 25% of positive cases, wrote Anna L. Funk, PhD, of the University of Calgary, Alberta, Canada, and colleagues.
However, the risk of severe outcomes of youth with COVID-19 remains poorly understood and data from large studies are lacking, they noted.
In a prospective cohort study published in JAMA Network Open, the researchers reviewed data from 3,221 children and adolescents who were tested for COVID-19 at one of 41 emergency departments in 10 countries including Argentina, Australia, Canada, Costa Rica, Italy, New Zealand, Paraguay, Singapore, Spain, and the United States between March 2020 and June 2021. Positive infections were confirmed by polymerase chain reaction (PCR) testing. At 14 days’ follow-up after a positive test, 735 patients (22.8%), were hospitalized, 107 (3.3%) had severe outcomes, and 4 (0.12%) had died. Severe outcomes were significantly more likely in children aged 5-10 years and 10-18 years vs. less than 1 year (odds ratios, 1.60 and 2.39, respectively), and in children with a self-reported chronic illness (OR, 2.34) or a prior episode of pneumonia (OR, 3.15).
Severe outcomes were more likely in patients who presented with symptoms that started 4-7 days before seeking care, compared with those whose symptoms started 0-3 days before seeking care (OR, 2.22).
The researchers also reviewed data from a subgroup of 2,510 individuals who were discharged home from the ED after initial testing. At 14 days’ follow-up, 50 of these patients (2.0%) were hospitalized and 12 (0.5%) had severe outcomes. In addition, the researchers found that the risk of severe outcomes among hospitalized COVID-19–positive youth was nearly four times higher, compared with hospitalized youth who tested negative for COVID-19 (risk difference, 3.9%).
Previous retrospective studies of severe outcomes in children and adolescents with COVID-19 have yielded varying results, in part because of the variation in study populations, the researchers noted in their discussion of the findings. “Our study population provides a risk estimate for youths brought for ED care.” Therefore, “Our lower estimate of severe disease likely reflects our stringent definition, which required the occurrence of complications or specific invasive interventions,” they said.
The study limitations included the potential overestimation of the risk of severe outcomes because patients were recruited in the ED, the researchers noted. Other limitations included variation in regional case definitions, screening criteria, and testing capacity among different sites and time periods. “Thus, 5% of our SARS-CoV-2–positive participants were asymptomatic – most of whom were tested as they were positive contacts of known cases or as part of routine screening procedures,” they said. The findings also are not generalizable to all community EDs and did not account for variants, they added.
However, the results were strengthened by the ability to compare outcomes for children with positive tests to similar children with negative tests, and add to the literature showing an increased risk of severe outcomes for those hospitalized with positive tests, the researchers concluded.
Data may inform clinical decisions
“The data [in the current study] are concerning for severe outcomes for children even prior to the Omicron strain,” said Margaret Thew, DNP, FP-BC, of Children’s Wisconsin-Milwaukee Hospital, in an interview. “Presently, the number of children infected with the Omicron strain is much higher and hospitalizations among children are at their highest since COVID-19 began,” she said. “For medical providers caring for this population, the study sheds light on pediatric patients who may be at higher risk of severe illness when they become infected with COVID-19,” she added.
“I was surprised by how high the number of pediatric patients hospitalized (22%) and the percentage (3%) with severe disease were during this time,” given that the timeline for these data preceded the spread of the Omicron strain, said Ms. Thew. “The risk of prior pneumonia was quite surprising. I do not recall seeing prior pneumonia as a risk factor for more severe COVID-19 with children or adults,” she added.
The take-home messaging for clinicians caring for children and adolescents is the added knowledge of the risk factors for severe outcomes from COVID-19, including the 10-18 age range, chronic illness, prior pneumonia, and longer symptom duration before seeking care in the ED, Ms. Thew emphasized.
However, additional research is needed on the impact of the new strains of COVID-19 on pediatric and adolescent hospitalizations, Ms. Thew said. Research also is needed on the other illnesses that have resulted from COVID-19, including illness requiring antibiotic use or medical interventions or treatments, and on the risk of combined COVID-19 and influenza viruses, she noted.
The study was supported by the Canadian Institutes of Health Research, Alberta Innovates, the Alberta Health Services University of Calgary Clinical Research Fund, the Alberta Children’s Hospital Research Institute, the COVID-19 Research Accelerator Funding Track (CRAFT) Program at the University of California, Davis, and the Cincinnati Children’s Hospital Medical Center Division of Emergency Medicine Small Grants Program. Lead author Dr. Funk was supported by the University of Calgary Eyes-High Post-Doctoral Research Fund, but had no financial conflicts to disclose. Ms. Thew had no financial conflicts to disclose and serves on the Editorial Advisory Board of Pediatric News.
Approximately 3% of youth who tested positive for COVID-19 in an emergency department setting had severe outcomes after 2 weeks, but this risk was 0.5% among those not admitted to the hospital, based on data from more than 3,000 individuals aged 18 and younger.
In the early stages of the COVID-19 pandemic, youth younger than 18 years accounted for fewer than 5% of reported cases, but now account for approximately 25% of positive cases, wrote Anna L. Funk, PhD, of the University of Calgary, Alberta, Canada, and colleagues.
However, the risk of severe outcomes of youth with COVID-19 remains poorly understood and data from large studies are lacking, they noted.
In a prospective cohort study published in JAMA Network Open, the researchers reviewed data from 3,221 children and adolescents who were tested for COVID-19 at one of 41 emergency departments in 10 countries including Argentina, Australia, Canada, Costa Rica, Italy, New Zealand, Paraguay, Singapore, Spain, and the United States between March 2020 and June 2021. Positive infections were confirmed by polymerase chain reaction (PCR) testing. At 14 days’ follow-up after a positive test, 735 patients (22.8%), were hospitalized, 107 (3.3%) had severe outcomes, and 4 (0.12%) had died. Severe outcomes were significantly more likely in children aged 5-10 years and 10-18 years vs. less than 1 year (odds ratios, 1.60 and 2.39, respectively), and in children with a self-reported chronic illness (OR, 2.34) or a prior episode of pneumonia (OR, 3.15).
Severe outcomes were more likely in patients who presented with symptoms that started 4-7 days before seeking care, compared with those whose symptoms started 0-3 days before seeking care (OR, 2.22).
The researchers also reviewed data from a subgroup of 2,510 individuals who were discharged home from the ED after initial testing. At 14 days’ follow-up, 50 of these patients (2.0%) were hospitalized and 12 (0.5%) had severe outcomes. In addition, the researchers found that the risk of severe outcomes among hospitalized COVID-19–positive youth was nearly four times higher, compared with hospitalized youth who tested negative for COVID-19 (risk difference, 3.9%).
Previous retrospective studies of severe outcomes in children and adolescents with COVID-19 have yielded varying results, in part because of the variation in study populations, the researchers noted in their discussion of the findings. “Our study population provides a risk estimate for youths brought for ED care.” Therefore, “Our lower estimate of severe disease likely reflects our stringent definition, which required the occurrence of complications or specific invasive interventions,” they said.
The study limitations included the potential overestimation of the risk of severe outcomes because patients were recruited in the ED, the researchers noted. Other limitations included variation in regional case definitions, screening criteria, and testing capacity among different sites and time periods. “Thus, 5% of our SARS-CoV-2–positive participants were asymptomatic – most of whom were tested as they were positive contacts of known cases or as part of routine screening procedures,” they said. The findings also are not generalizable to all community EDs and did not account for variants, they added.
However, the results were strengthened by the ability to compare outcomes for children with positive tests to similar children with negative tests, and add to the literature showing an increased risk of severe outcomes for those hospitalized with positive tests, the researchers concluded.
Data may inform clinical decisions
“The data [in the current study] are concerning for severe outcomes for children even prior to the Omicron strain,” said Margaret Thew, DNP, FP-BC, of Children’s Wisconsin-Milwaukee Hospital, in an interview. “Presently, the number of children infected with the Omicron strain is much higher and hospitalizations among children are at their highest since COVID-19 began,” she said. “For medical providers caring for this population, the study sheds light on pediatric patients who may be at higher risk of severe illness when they become infected with COVID-19,” she added.
“I was surprised by how high the number of pediatric patients hospitalized (22%) and the percentage (3%) with severe disease were during this time,” given that the timeline for these data preceded the spread of the Omicron strain, said Ms. Thew. “The risk of prior pneumonia was quite surprising. I do not recall seeing prior pneumonia as a risk factor for more severe COVID-19 with children or adults,” she added.
The take-home messaging for clinicians caring for children and adolescents is the added knowledge of the risk factors for severe outcomes from COVID-19, including the 10-18 age range, chronic illness, prior pneumonia, and longer symptom duration before seeking care in the ED, Ms. Thew emphasized.
However, additional research is needed on the impact of the new strains of COVID-19 on pediatric and adolescent hospitalizations, Ms. Thew said. Research also is needed on the other illnesses that have resulted from COVID-19, including illness requiring antibiotic use or medical interventions or treatments, and on the risk of combined COVID-19 and influenza viruses, she noted.
The study was supported by the Canadian Institutes of Health Research, Alberta Innovates, the Alberta Health Services University of Calgary Clinical Research Fund, the Alberta Children’s Hospital Research Institute, the COVID-19 Research Accelerator Funding Track (CRAFT) Program at the University of California, Davis, and the Cincinnati Children’s Hospital Medical Center Division of Emergency Medicine Small Grants Program. Lead author Dr. Funk was supported by the University of Calgary Eyes-High Post-Doctoral Research Fund, but had no financial conflicts to disclose. Ms. Thew had no financial conflicts to disclose and serves on the Editorial Advisory Board of Pediatric News.
FROM JAMA NETWORK OPEN
Pediatric community-acquired pneumonia: 5 days of antibiotics better than 10 days
The evidence is in: and had the added benefit of a lower risk of inducing antibiotic resistance, according to the randomized, controlled SCOUT-CAP trial.
“Several studies have shown shorter antibiotic courses to be non-inferior to the standard treatment strategy, but in our study, we show that a shortened 5-day course of therapy was superior to standard therapy because the short course achieved similar outcomes with fewer days of antibiotics,” Derek Williams, MD, MPH, Vanderbilt University Medical Center, Nashville, Tenn., said in an email.
“These data are immediately applicable to frontline clinicians, and we hope this study will shift the paradigm towards more judicious treatment approaches for childhood pneumonia, resulting in care that is safer and more effective,” he added.
The study was published online Jan. 18 in JAMA Pediatrics.
Uncomplicated CAP
The study enrolled children aged 6 months to 71 months diagnosed with uncomplicated CAP who demonstrated early clinical improvement in response to 5 days of antibiotic treatment. Participants were prescribed either amoxicillin, amoxicillin and clavulanate, or cefdinir according to standard of care and were randomized on day 6 to another 5 days of their initially prescribed antibiotic course or to placebo.
“Those assessed on day 6 were eligible only if they had not yet received a dose of antibiotic therapy on that day,” the authors write. The primary endpoint was end-of-treatment response, adjusted for the duration of antibiotic risk as assessed by RADAR. As the authors explain, RADAR is a composite endpoint that ranks each child’s clinical response, resolution of symptoms, and antibiotic-associated adverse effects (AEs) in an ordinal desirability of outcome ranking, or DOOR.
“There were no differences between strategies in the DOOR or in its individual components,” Dr. Williams and colleagues point out. A total of 380 children took part in the study. The mean age of participants was 35.7 months, and half were male.
Over 90% of children randomized to active therapy were prescribed amoxicillin. “Fewer than 10% of children in either strategy had an inadequate clinical response,” the authors report.
However, the 5-day antibiotic strategy had a 69% (95% CI, 63%-75%) probability of children achieving a more desirable RADAR outcome compared with the standard, 10-day course, as assessed either on days 6 to 10 at outcome assessment visit one (OAV1) or at OAV2 on days 19 to 25.
There were also no significant differences between the two groups in the percentage of participants with persistent symptoms at either assessment point, they note. At assessment visit one, 40% of children assigned to the short-course strategy and 37% of children assigned to the 10-day strategy reported an antibiotic-related AE, most of which were mild.
Resistome analysis
Some 171 children were included in a resistome analysis in which throat swabs were collected between study days 19 and 25 to quantify antibiotic resistance genes in oropharyngeal flora. The total number of resistance genes per prokaryotic cell (RGPC) was significantly lower in children treated with antibiotics for 5 days compared with children who were treated for 10 days.
Specifically, the median number of total RGPC was 1.17 (95% CI, 0.35-2.43) for the short-course strategy and 1.33 (95% CI, 0.46-11.08) for the standard-course strategy (P = .01). Similarly, the median number of β-lactamase RGPC was 0.55 (0.18-1.24) for the short-course strategy and 0.60 (0.21-2.45) for the standard-course strategy (P = .03).
“Providing the shortest duration of antibiotics necessary to effectively treat an infection is a central tenet of antimicrobial stewardship and a convenient and cost-effective strategy for caregivers,” the authors observe. For example, reducing treatment from 10 to 5 days for outpatient CAP could reduce the number of days spent on antibiotics by up to 7.5 million days in the U.S. each year.
“If we can safely reduce antibiotic exposure, we can minimize antibiotic side effects while also helping to slow antibiotic resistance,” Dr. Williams pointed out.
Fewer days of having to give their child repeated doses of antibiotics is also more convenient for families, he added.
Asked to comment on the study, David Greenberg, MD, professor of pediatrics and infectious diseases, Ben Gurion University of the Negev, Israel, explained that the length of antibiotic therapy as recommended by various guidelines is more or less arbitrary, some infections being excepted.
“There have been no studies evaluating the recommendation for a 100-day treatment course, and it’s kind of a joke because if you look at the treatment of just about any infection, it’s either for 7 days or 14 days or even 20 days because it’s easy to calculate – it’s not that anybody proved that treatment of whatever infection it is should last this long,” he told this news organization.
Moreover, adherence to a shorter antibiotic course is much better than it is to a longer course. If, for example, physicians tell a mother to take two bottles of antibiotics for a treatment course of 10 days, she’ll finish the first bottle which is good for 5 days and, because the child is fine, “she forgets about the second bottle,” Dr. Greenberg said.
In one of the first studies to compare a short versus long course of antibiotic therapy in uncomplicated CAP in young children, Dr. Greenberg and colleagues initially compared a 3-day course of high-dose amoxicillin to a 10-day course of the same treatment, but the 3-day course was associated with an unacceptable failure rate. (At the time, the World Health Organization was recommending a 3-day course of antibiotics for the treatment of uncomplicated CAP in children.)
They stopped the study and then initiated a second study in which they compared a 5-day course of the same antibiotic to a 10-day course and found the 5-day course was comparable to the 10-day course in terms of clinical cure rates. As a result of his study, Dr. Greenberg has long since prescribed a 5-day course of antibiotics for his own patients.
“Five days is good,” he affirmed. “And if patients start a 10-day course of an antibiotic for, say, a urinary tract infection and a subsequent culture comes back negative, they don’t have to finish the antibiotics either.” Dr. Greenberg said.
Dr. Williams said he has no financial ties to industry. Dr. Greenberg said he has served as a consultant for Pfizer, Merck, Johnson & Johnson, and AstraZeneca. He is also a founder of the company Beyond Air.
A version of this article first appeared on Medscape.com.
The evidence is in: and had the added benefit of a lower risk of inducing antibiotic resistance, according to the randomized, controlled SCOUT-CAP trial.
“Several studies have shown shorter antibiotic courses to be non-inferior to the standard treatment strategy, but in our study, we show that a shortened 5-day course of therapy was superior to standard therapy because the short course achieved similar outcomes with fewer days of antibiotics,” Derek Williams, MD, MPH, Vanderbilt University Medical Center, Nashville, Tenn., said in an email.
“These data are immediately applicable to frontline clinicians, and we hope this study will shift the paradigm towards more judicious treatment approaches for childhood pneumonia, resulting in care that is safer and more effective,” he added.
The study was published online Jan. 18 in JAMA Pediatrics.
Uncomplicated CAP
The study enrolled children aged 6 months to 71 months diagnosed with uncomplicated CAP who demonstrated early clinical improvement in response to 5 days of antibiotic treatment. Participants were prescribed either amoxicillin, amoxicillin and clavulanate, or cefdinir according to standard of care and were randomized on day 6 to another 5 days of their initially prescribed antibiotic course or to placebo.
“Those assessed on day 6 were eligible only if they had not yet received a dose of antibiotic therapy on that day,” the authors write. The primary endpoint was end-of-treatment response, adjusted for the duration of antibiotic risk as assessed by RADAR. As the authors explain, RADAR is a composite endpoint that ranks each child’s clinical response, resolution of symptoms, and antibiotic-associated adverse effects (AEs) in an ordinal desirability of outcome ranking, or DOOR.
“There were no differences between strategies in the DOOR or in its individual components,” Dr. Williams and colleagues point out. A total of 380 children took part in the study. The mean age of participants was 35.7 months, and half were male.
Over 90% of children randomized to active therapy were prescribed amoxicillin. “Fewer than 10% of children in either strategy had an inadequate clinical response,” the authors report.
However, the 5-day antibiotic strategy had a 69% (95% CI, 63%-75%) probability of children achieving a more desirable RADAR outcome compared with the standard, 10-day course, as assessed either on days 6 to 10 at outcome assessment visit one (OAV1) or at OAV2 on days 19 to 25.
There were also no significant differences between the two groups in the percentage of participants with persistent symptoms at either assessment point, they note. At assessment visit one, 40% of children assigned to the short-course strategy and 37% of children assigned to the 10-day strategy reported an antibiotic-related AE, most of which were mild.
Resistome analysis
Some 171 children were included in a resistome analysis in which throat swabs were collected between study days 19 and 25 to quantify antibiotic resistance genes in oropharyngeal flora. The total number of resistance genes per prokaryotic cell (RGPC) was significantly lower in children treated with antibiotics for 5 days compared with children who were treated for 10 days.
Specifically, the median number of total RGPC was 1.17 (95% CI, 0.35-2.43) for the short-course strategy and 1.33 (95% CI, 0.46-11.08) for the standard-course strategy (P = .01). Similarly, the median number of β-lactamase RGPC was 0.55 (0.18-1.24) for the short-course strategy and 0.60 (0.21-2.45) for the standard-course strategy (P = .03).
“Providing the shortest duration of antibiotics necessary to effectively treat an infection is a central tenet of antimicrobial stewardship and a convenient and cost-effective strategy for caregivers,” the authors observe. For example, reducing treatment from 10 to 5 days for outpatient CAP could reduce the number of days spent on antibiotics by up to 7.5 million days in the U.S. each year.
“If we can safely reduce antibiotic exposure, we can minimize antibiotic side effects while also helping to slow antibiotic resistance,” Dr. Williams pointed out.
Fewer days of having to give their child repeated doses of antibiotics is also more convenient for families, he added.
Asked to comment on the study, David Greenberg, MD, professor of pediatrics and infectious diseases, Ben Gurion University of the Negev, Israel, explained that the length of antibiotic therapy as recommended by various guidelines is more or less arbitrary, some infections being excepted.
“There have been no studies evaluating the recommendation for a 100-day treatment course, and it’s kind of a joke because if you look at the treatment of just about any infection, it’s either for 7 days or 14 days or even 20 days because it’s easy to calculate – it’s not that anybody proved that treatment of whatever infection it is should last this long,” he told this news organization.
Moreover, adherence to a shorter antibiotic course is much better than it is to a longer course. If, for example, physicians tell a mother to take two bottles of antibiotics for a treatment course of 10 days, she’ll finish the first bottle which is good for 5 days and, because the child is fine, “she forgets about the second bottle,” Dr. Greenberg said.
In one of the first studies to compare a short versus long course of antibiotic therapy in uncomplicated CAP in young children, Dr. Greenberg and colleagues initially compared a 3-day course of high-dose amoxicillin to a 10-day course of the same treatment, but the 3-day course was associated with an unacceptable failure rate. (At the time, the World Health Organization was recommending a 3-day course of antibiotics for the treatment of uncomplicated CAP in children.)
They stopped the study and then initiated a second study in which they compared a 5-day course of the same antibiotic to a 10-day course and found the 5-day course was comparable to the 10-day course in terms of clinical cure rates. As a result of his study, Dr. Greenberg has long since prescribed a 5-day course of antibiotics for his own patients.
“Five days is good,” he affirmed. “And if patients start a 10-day course of an antibiotic for, say, a urinary tract infection and a subsequent culture comes back negative, they don’t have to finish the antibiotics either.” Dr. Greenberg said.
Dr. Williams said he has no financial ties to industry. Dr. Greenberg said he has served as a consultant for Pfizer, Merck, Johnson & Johnson, and AstraZeneca. He is also a founder of the company Beyond Air.
A version of this article first appeared on Medscape.com.
The evidence is in: and had the added benefit of a lower risk of inducing antibiotic resistance, according to the randomized, controlled SCOUT-CAP trial.
“Several studies have shown shorter antibiotic courses to be non-inferior to the standard treatment strategy, but in our study, we show that a shortened 5-day course of therapy was superior to standard therapy because the short course achieved similar outcomes with fewer days of antibiotics,” Derek Williams, MD, MPH, Vanderbilt University Medical Center, Nashville, Tenn., said in an email.
“These data are immediately applicable to frontline clinicians, and we hope this study will shift the paradigm towards more judicious treatment approaches for childhood pneumonia, resulting in care that is safer and more effective,” he added.
The study was published online Jan. 18 in JAMA Pediatrics.
Uncomplicated CAP
The study enrolled children aged 6 months to 71 months diagnosed with uncomplicated CAP who demonstrated early clinical improvement in response to 5 days of antibiotic treatment. Participants were prescribed either amoxicillin, amoxicillin and clavulanate, or cefdinir according to standard of care and were randomized on day 6 to another 5 days of their initially prescribed antibiotic course or to placebo.
“Those assessed on day 6 were eligible only if they had not yet received a dose of antibiotic therapy on that day,” the authors write. The primary endpoint was end-of-treatment response, adjusted for the duration of antibiotic risk as assessed by RADAR. As the authors explain, RADAR is a composite endpoint that ranks each child’s clinical response, resolution of symptoms, and antibiotic-associated adverse effects (AEs) in an ordinal desirability of outcome ranking, or DOOR.
“There were no differences between strategies in the DOOR or in its individual components,” Dr. Williams and colleagues point out. A total of 380 children took part in the study. The mean age of participants was 35.7 months, and half were male.
Over 90% of children randomized to active therapy were prescribed amoxicillin. “Fewer than 10% of children in either strategy had an inadequate clinical response,” the authors report.
However, the 5-day antibiotic strategy had a 69% (95% CI, 63%-75%) probability of children achieving a more desirable RADAR outcome compared with the standard, 10-day course, as assessed either on days 6 to 10 at outcome assessment visit one (OAV1) or at OAV2 on days 19 to 25.
There were also no significant differences between the two groups in the percentage of participants with persistent symptoms at either assessment point, they note. At assessment visit one, 40% of children assigned to the short-course strategy and 37% of children assigned to the 10-day strategy reported an antibiotic-related AE, most of which were mild.
Resistome analysis
Some 171 children were included in a resistome analysis in which throat swabs were collected between study days 19 and 25 to quantify antibiotic resistance genes in oropharyngeal flora. The total number of resistance genes per prokaryotic cell (RGPC) was significantly lower in children treated with antibiotics for 5 days compared with children who were treated for 10 days.
Specifically, the median number of total RGPC was 1.17 (95% CI, 0.35-2.43) for the short-course strategy and 1.33 (95% CI, 0.46-11.08) for the standard-course strategy (P = .01). Similarly, the median number of β-lactamase RGPC was 0.55 (0.18-1.24) for the short-course strategy and 0.60 (0.21-2.45) for the standard-course strategy (P = .03).
“Providing the shortest duration of antibiotics necessary to effectively treat an infection is a central tenet of antimicrobial stewardship and a convenient and cost-effective strategy for caregivers,” the authors observe. For example, reducing treatment from 10 to 5 days for outpatient CAP could reduce the number of days spent on antibiotics by up to 7.5 million days in the U.S. each year.
“If we can safely reduce antibiotic exposure, we can minimize antibiotic side effects while also helping to slow antibiotic resistance,” Dr. Williams pointed out.
Fewer days of having to give their child repeated doses of antibiotics is also more convenient for families, he added.
Asked to comment on the study, David Greenberg, MD, professor of pediatrics and infectious diseases, Ben Gurion University of the Negev, Israel, explained that the length of antibiotic therapy as recommended by various guidelines is more or less arbitrary, some infections being excepted.
“There have been no studies evaluating the recommendation for a 100-day treatment course, and it’s kind of a joke because if you look at the treatment of just about any infection, it’s either for 7 days or 14 days or even 20 days because it’s easy to calculate – it’s not that anybody proved that treatment of whatever infection it is should last this long,” he told this news organization.
Moreover, adherence to a shorter antibiotic course is much better than it is to a longer course. If, for example, physicians tell a mother to take two bottles of antibiotics for a treatment course of 10 days, she’ll finish the first bottle which is good for 5 days and, because the child is fine, “she forgets about the second bottle,” Dr. Greenberg said.
In one of the first studies to compare a short versus long course of antibiotic therapy in uncomplicated CAP in young children, Dr. Greenberg and colleagues initially compared a 3-day course of high-dose amoxicillin to a 10-day course of the same treatment, but the 3-day course was associated with an unacceptable failure rate. (At the time, the World Health Organization was recommending a 3-day course of antibiotics for the treatment of uncomplicated CAP in children.)
They stopped the study and then initiated a second study in which they compared a 5-day course of the same antibiotic to a 10-day course and found the 5-day course was comparable to the 10-day course in terms of clinical cure rates. As a result of his study, Dr. Greenberg has long since prescribed a 5-day course of antibiotics for his own patients.
“Five days is good,” he affirmed. “And if patients start a 10-day course of an antibiotic for, say, a urinary tract infection and a subsequent culture comes back negative, they don’t have to finish the antibiotics either.” Dr. Greenberg said.
Dr. Williams said he has no financial ties to industry. Dr. Greenberg said he has served as a consultant for Pfizer, Merck, Johnson & Johnson, and AstraZeneca. He is also a founder of the company Beyond Air.
A version of this article first appeared on Medscape.com.
Appendectomy or antibiotics? Large trial helps decision-making
The presence of mineralized stool, known as appendicolith, was associated with a nearly twofold increased risk of undergoing appendectomy within 30 days of initiating antibiotics, write David Flum, MD, of the University of Washington, Seattle, and coauthors in a paper published in JAMA Surgery on Jan. 12, 2021.
But the surprise was the lack of an association between appendectomy and factors often presumed to be consistent with more severe appendicitis.
Physicians have had their own ideas about what factors make a patient more likely to need an appendectomy after an initial round of treatment with antibiotics, such as a high white blood cell count or a perforation seen on CT scan, Dr. Flum said in an interview. But the research didn’t support some of these theories.
“This is why we do the studies,” Dr. Flum said. “Sometimes we find out that our hunches were wrong.”
Dr. Flum and coauthors measured the association between different patient factors and disease severity and the need for appendectomy following a course of antibiotics. They used adjusted odds ratios to describe these relationships while accounting for other differences.
An OR of 1.0 – or when the confidence interval around an OR crosses 1 – signals that there is no association between that factor and appendectomy. Positive ORs with confidence intervals that exclude 1.0 suggest the factor was associated with appendectomy.
The OR was 1.99 for the presence of appendicolith, a finding with a 95% confidence interval of 1.28-3.10. The OR was 1.53 (95% CI, 1.01-2.31) for female sex.
But the OR was 1.14 (95% CI, 0.66-1.98) for perforation, abscess, or fat stranding.
The OR was 1.09 (95% CI, 1.00-1.18) for radiographic finding of a larger appendix, as measured by diameter.
And the OR was 1.03 (95% CI, 0.98-1.09) for having a higher white blood cell count, as measured by a 1,000-cells/mcL increase.
Appy or not?
This paper draws from the Comparison of Outcomes of Antibiotic Drugs and Appendectomy (CODA) trial (NCT02800785), for which top-line results were published in 2020 in the New England Journal of Medicine. In that paper, Dr. Flum and colleagues reported on results for 1,552 adults (414 with an appendicolith) who were evenly randomized to either antibiotics treatment or appendectomy. After 30 days, antibiotics were found to be noninferior to appendectomy, as reported by this news organization.
The federal Patient-Centered Outcomes Research Institute funded the CODA research. Dr. Flum said the National Institutes of Health had not appeared interested in funding a look at the different options available to patients experiencing appendicitis. Congress created PCORI as part of the Affordable Care Act of 2010, seeking to encourage researchers to study which treatments best serve patients through direct comparisons. Its support was critical for Dr. Flum and colleagues in seeking to help people weigh their options for treating appendicitis.
The CODA study “models what the patient’s experience is like, and this has not been the focus of NIH as much,” Dr. Flum said.
The CODA team has sought to make it easy for patients to consider what its findings and other research on appendicitis mean for them. They created an online decision-making tool, available at the aptly named http://www.appyornot.org/ website, which has videos in English and Spanish explaining patients’ options in simple terms. The website also asks questions about personal preferences, priorities, and resources to help them choose a treatment based on their individual situation.
Shift away from ‘paternalistic framing’
In the past, surgeons focused on the risk for patients from procedures, making the decisions for them about whether or not to proceed. There’s now a drive to shift away from this “paternalistic framing” toward shared decision-making, Dr. Flum said.
Surgeons need to have conversations with their patients about what’s happening in their lives as well as to assess their fears and concerns about treatment options, he said. These are aspects of patient care that were not covered in medical school or surgical training, but they lead to “less paternalistic” treatment. A patient’s decision about whether to choose surgery or antibiotics for appendicitis may hinge on factors such as insurance coverage, access to childcare, and the ability to miss days of work.
Dr. Flum said his fellow surgeons by and large have reacted well to the CODA team’s work.
“To their credit, the surgical community has embraced a healthy skepticism about the role of surgery,” Dr. Flum said.
The guidelines of the American College of Surgeons state that there is “high-quality evidence” that most patients with appendicitis can be managed with antibiotics instead of appendectomy (69% overall avoid appendectomy by 90 days, 75% of those without appendicolith, and 59% of those with appendicolith).
“Based on the surgeon’s judgment, patient preferences, and local resources (e.g., hospital staff, bed, and PPE supply availability) antibiotics are an acceptable first-line treatment, with appendectomy offered for those with worsening or recurrent symptoms,” the ACS guidelines say.
In an interview, Samir M. Fakhry, MD, vice president of HCA Center for Trauma and Acute Care Surgery Research in Nashville, Tenn., agreed with Dr. Flum about the shift taking place in medicine.
The CODA research, including the new paper in JAMA Surgery, makes it easier for physicians to work with patients and their families to reach decisions about how to treat appendicitis, Dr. Fakhry said.
These important discussions take time, he said, and patients must be allowed that time. Patients might feel misled, for example, if a surgeon pressed for appendectomy without explaining that a course of antibiotics may have served them well. Other patients may opt for surgery right away, especially in cases with appendicoliths, to avoid the potential for repeat episodes of medical care.
“You’ve got people who just want to get it done and over with. You’ve got people who want to avoid surgery no matter what,” Dr. Fakhry said. “It’s not just about the science and the data.”
This study was supported by a grant from PCORI. The authors reported having served as consultants or reviewers or have received fees for work outside of this paper from Stryker, Kerecis, Acera, Medline, Shriner’s Research Fund, UpToDate, and Tetraphase Pharmaceuticals Stryker.
A version of this article first appeared on Medscape.com.
The presence of mineralized stool, known as appendicolith, was associated with a nearly twofold increased risk of undergoing appendectomy within 30 days of initiating antibiotics, write David Flum, MD, of the University of Washington, Seattle, and coauthors in a paper published in JAMA Surgery on Jan. 12, 2021.
But the surprise was the lack of an association between appendectomy and factors often presumed to be consistent with more severe appendicitis.
Physicians have had their own ideas about what factors make a patient more likely to need an appendectomy after an initial round of treatment with antibiotics, such as a high white blood cell count or a perforation seen on CT scan, Dr. Flum said in an interview. But the research didn’t support some of these theories.
“This is why we do the studies,” Dr. Flum said. “Sometimes we find out that our hunches were wrong.”
Dr. Flum and coauthors measured the association between different patient factors and disease severity and the need for appendectomy following a course of antibiotics. They used adjusted odds ratios to describe these relationships while accounting for other differences.
An OR of 1.0 – or when the confidence interval around an OR crosses 1 – signals that there is no association between that factor and appendectomy. Positive ORs with confidence intervals that exclude 1.0 suggest the factor was associated with appendectomy.
The OR was 1.99 for the presence of appendicolith, a finding with a 95% confidence interval of 1.28-3.10. The OR was 1.53 (95% CI, 1.01-2.31) for female sex.
But the OR was 1.14 (95% CI, 0.66-1.98) for perforation, abscess, or fat stranding.
The OR was 1.09 (95% CI, 1.00-1.18) for radiographic finding of a larger appendix, as measured by diameter.
And the OR was 1.03 (95% CI, 0.98-1.09) for having a higher white blood cell count, as measured by a 1,000-cells/mcL increase.
Appy or not?
This paper draws from the Comparison of Outcomes of Antibiotic Drugs and Appendectomy (CODA) trial (NCT02800785), for which top-line results were published in 2020 in the New England Journal of Medicine. In that paper, Dr. Flum and colleagues reported on results for 1,552 adults (414 with an appendicolith) who were evenly randomized to either antibiotics treatment or appendectomy. After 30 days, antibiotics were found to be noninferior to appendectomy, as reported by this news organization.
The federal Patient-Centered Outcomes Research Institute funded the CODA research. Dr. Flum said the National Institutes of Health had not appeared interested in funding a look at the different options available to patients experiencing appendicitis. Congress created PCORI as part of the Affordable Care Act of 2010, seeking to encourage researchers to study which treatments best serve patients through direct comparisons. Its support was critical for Dr. Flum and colleagues in seeking to help people weigh their options for treating appendicitis.
The CODA study “models what the patient’s experience is like, and this has not been the focus of NIH as much,” Dr. Flum said.
The CODA team has sought to make it easy for patients to consider what its findings and other research on appendicitis mean for them. They created an online decision-making tool, available at the aptly named http://www.appyornot.org/ website, which has videos in English and Spanish explaining patients’ options in simple terms. The website also asks questions about personal preferences, priorities, and resources to help them choose a treatment based on their individual situation.
Shift away from ‘paternalistic framing’
In the past, surgeons focused on the risk for patients from procedures, making the decisions for them about whether or not to proceed. There’s now a drive to shift away from this “paternalistic framing” toward shared decision-making, Dr. Flum said.
Surgeons need to have conversations with their patients about what’s happening in their lives as well as to assess their fears and concerns about treatment options, he said. These are aspects of patient care that were not covered in medical school or surgical training, but they lead to “less paternalistic” treatment. A patient’s decision about whether to choose surgery or antibiotics for appendicitis may hinge on factors such as insurance coverage, access to childcare, and the ability to miss days of work.
Dr. Flum said his fellow surgeons by and large have reacted well to the CODA team’s work.
“To their credit, the surgical community has embraced a healthy skepticism about the role of surgery,” Dr. Flum said.
The guidelines of the American College of Surgeons state that there is “high-quality evidence” that most patients with appendicitis can be managed with antibiotics instead of appendectomy (69% overall avoid appendectomy by 90 days, 75% of those without appendicolith, and 59% of those with appendicolith).
“Based on the surgeon’s judgment, patient preferences, and local resources (e.g., hospital staff, bed, and PPE supply availability) antibiotics are an acceptable first-line treatment, with appendectomy offered for those with worsening or recurrent symptoms,” the ACS guidelines say.
In an interview, Samir M. Fakhry, MD, vice president of HCA Center for Trauma and Acute Care Surgery Research in Nashville, Tenn., agreed with Dr. Flum about the shift taking place in medicine.
The CODA research, including the new paper in JAMA Surgery, makes it easier for physicians to work with patients and their families to reach decisions about how to treat appendicitis, Dr. Fakhry said.
These important discussions take time, he said, and patients must be allowed that time. Patients might feel misled, for example, if a surgeon pressed for appendectomy without explaining that a course of antibiotics may have served them well. Other patients may opt for surgery right away, especially in cases with appendicoliths, to avoid the potential for repeat episodes of medical care.
“You’ve got people who just want to get it done and over with. You’ve got people who want to avoid surgery no matter what,” Dr. Fakhry said. “It’s not just about the science and the data.”
This study was supported by a grant from PCORI. The authors reported having served as consultants or reviewers or have received fees for work outside of this paper from Stryker, Kerecis, Acera, Medline, Shriner’s Research Fund, UpToDate, and Tetraphase Pharmaceuticals Stryker.
A version of this article first appeared on Medscape.com.
The presence of mineralized stool, known as appendicolith, was associated with a nearly twofold increased risk of undergoing appendectomy within 30 days of initiating antibiotics, write David Flum, MD, of the University of Washington, Seattle, and coauthors in a paper published in JAMA Surgery on Jan. 12, 2021.
But the surprise was the lack of an association between appendectomy and factors often presumed to be consistent with more severe appendicitis.
Physicians have had their own ideas about what factors make a patient more likely to need an appendectomy after an initial round of treatment with antibiotics, such as a high white blood cell count or a perforation seen on CT scan, Dr. Flum said in an interview. But the research didn’t support some of these theories.
“This is why we do the studies,” Dr. Flum said. “Sometimes we find out that our hunches were wrong.”
Dr. Flum and coauthors measured the association between different patient factors and disease severity and the need for appendectomy following a course of antibiotics. They used adjusted odds ratios to describe these relationships while accounting for other differences.
An OR of 1.0 – or when the confidence interval around an OR crosses 1 – signals that there is no association between that factor and appendectomy. Positive ORs with confidence intervals that exclude 1.0 suggest the factor was associated with appendectomy.
The OR was 1.99 for the presence of appendicolith, a finding with a 95% confidence interval of 1.28-3.10. The OR was 1.53 (95% CI, 1.01-2.31) for female sex.
But the OR was 1.14 (95% CI, 0.66-1.98) for perforation, abscess, or fat stranding.
The OR was 1.09 (95% CI, 1.00-1.18) for radiographic finding of a larger appendix, as measured by diameter.
And the OR was 1.03 (95% CI, 0.98-1.09) for having a higher white blood cell count, as measured by a 1,000-cells/mcL increase.
Appy or not?
This paper draws from the Comparison of Outcomes of Antibiotic Drugs and Appendectomy (CODA) trial (NCT02800785), for which top-line results were published in 2020 in the New England Journal of Medicine. In that paper, Dr. Flum and colleagues reported on results for 1,552 adults (414 with an appendicolith) who were evenly randomized to either antibiotics treatment or appendectomy. After 30 days, antibiotics were found to be noninferior to appendectomy, as reported by this news organization.
The federal Patient-Centered Outcomes Research Institute funded the CODA research. Dr. Flum said the National Institutes of Health had not appeared interested in funding a look at the different options available to patients experiencing appendicitis. Congress created PCORI as part of the Affordable Care Act of 2010, seeking to encourage researchers to study which treatments best serve patients through direct comparisons. Its support was critical for Dr. Flum and colleagues in seeking to help people weigh their options for treating appendicitis.
The CODA study “models what the patient’s experience is like, and this has not been the focus of NIH as much,” Dr. Flum said.
The CODA team has sought to make it easy for patients to consider what its findings and other research on appendicitis mean for them. They created an online decision-making tool, available at the aptly named http://www.appyornot.org/ website, which has videos in English and Spanish explaining patients’ options in simple terms. The website also asks questions about personal preferences, priorities, and resources to help them choose a treatment based on their individual situation.
Shift away from ‘paternalistic framing’
In the past, surgeons focused on the risk for patients from procedures, making the decisions for them about whether or not to proceed. There’s now a drive to shift away from this “paternalistic framing” toward shared decision-making, Dr. Flum said.
Surgeons need to have conversations with their patients about what’s happening in their lives as well as to assess their fears and concerns about treatment options, he said. These are aspects of patient care that were not covered in medical school or surgical training, but they lead to “less paternalistic” treatment. A patient’s decision about whether to choose surgery or antibiotics for appendicitis may hinge on factors such as insurance coverage, access to childcare, and the ability to miss days of work.
Dr. Flum said his fellow surgeons by and large have reacted well to the CODA team’s work.
“To their credit, the surgical community has embraced a healthy skepticism about the role of surgery,” Dr. Flum said.
The guidelines of the American College of Surgeons state that there is “high-quality evidence” that most patients with appendicitis can be managed with antibiotics instead of appendectomy (69% overall avoid appendectomy by 90 days, 75% of those without appendicolith, and 59% of those with appendicolith).
“Based on the surgeon’s judgment, patient preferences, and local resources (e.g., hospital staff, bed, and PPE supply availability) antibiotics are an acceptable first-line treatment, with appendectomy offered for those with worsening or recurrent symptoms,” the ACS guidelines say.
In an interview, Samir M. Fakhry, MD, vice president of HCA Center for Trauma and Acute Care Surgery Research in Nashville, Tenn., agreed with Dr. Flum about the shift taking place in medicine.
The CODA research, including the new paper in JAMA Surgery, makes it easier for physicians to work with patients and their families to reach decisions about how to treat appendicitis, Dr. Fakhry said.
These important discussions take time, he said, and patients must be allowed that time. Patients might feel misled, for example, if a surgeon pressed for appendectomy without explaining that a course of antibiotics may have served them well. Other patients may opt for surgery right away, especially in cases with appendicoliths, to avoid the potential for repeat episodes of medical care.
“You’ve got people who just want to get it done and over with. You’ve got people who want to avoid surgery no matter what,” Dr. Fakhry said. “It’s not just about the science and the data.”
This study was supported by a grant from PCORI. The authors reported having served as consultants or reviewers or have received fees for work outside of this paper from Stryker, Kerecis, Acera, Medline, Shriner’s Research Fund, UpToDate, and Tetraphase Pharmaceuticals Stryker.
A version of this article first appeared on Medscape.com.
FROM JAMA SURGERY
When the patient wants to speak to a manager
A patient swore at me the other day. Not as in “she used a curse word.” As in she spewed fury, spitting out a vulgar, adverbial word before “... terrible doctor” while jabbing her finger toward me. In my 15 years of practice, I’d never had that happen before. Equally surprising, I was not surprised by her outburst. The level of incivility from patients is at an all-time high.
Her anger was misdirected. She wanted me to write a letter to her employer excusing her from getting a vaccine. It was neither indicated nor ethical for me to do so. I did my best to redirect her, but without success. As our chief of service, I often help with service concerns and am happy to see patients who want another opinion or want to speak with the department head (aka, “the manager”). Usually I can help. Lately, it’s become harder.
Not only are such rude incidents more frequent, but they are also more dramatic and inappropriate. For example, I cannot imagine writing a complaint against a doctor stating that she must be a foreign medical grad (as it happens, she’s Ivy League-trained) or demanding money back when a biopsy result turned out to be benign, or threatening to report a doctor to the medical board because he failed to schedule a follow-up appointment (that doctor had been retired for months). Patients have hung up on our staff mid-sentence and slammed a clinic door when they left in a huff. Why are so many previously sensible people throwing childlike tantrums?
It’s the same phenomenon happening to our fellow service agents across all industries. The Federal Aviation Administration’s graph of unruly passenger incidents is a flat line from 1995 to 2019, then it goes straight vertical. A recent survey showed that Americans’ sense of civility is low and worse, that people’s expectations that civility will improve is going down. It’s palpable. Last month, I witnessed a man and woman screaming at each other over Christmas lights in a busy store. An army of aproned walkie-talkie staff surrounded them and escorted them out – their coordination and efficiency clearly indicated they’d done this before. Customers everywhere are mad, frustrated, disenfranchised. Lately, a lot of things just are not working out for them. Supplies are out. Kids are sent home from school. No elective surgery appointments are available. The insta-gratification they’ve grown accustomed to from Amazon and DoorDash is colliding with the reality that not everything works that way.
The word “patient’’ you’ll recall comes from the Latin “patior,” meaning to suffer or bear. With virus variants raging, inflation growing, and call center wait times approaching infinity, many of our patients, it seems, cannot bear any more. I’m confident this situation will improve and our patients will be more reasonable in their expectations, but I am afraid that, in the end, we’ll have lost some decorum and dignity that we may never find again in medicine.
For my potty-mouthed patient, I made an excuse to leave the room to get my dermatoscope and walked out. It gave her time to calm down. I returned in a few minutes to do a skin exam. As I was wrapping up, I advised her that she cannot raise her voice or use offensive language and that she should know that I and everyone in our office cares about her and wants to help. She did apologize for her behavior, but then had to add that, if I really cared, I’d write the letter for her.
I guess the customer is not always right.
Dr. Benabio is director of Healthcare Transformation and chief of dermatology at Kaiser Permanente San Diego. The opinions expressed in this column are his own and do not represent those of Kaiser Permanente. Dr. Benabio is @Dermdoc on Twitter. Write to him at [email protected]
A patient swore at me the other day. Not as in “she used a curse word.” As in she spewed fury, spitting out a vulgar, adverbial word before “... terrible doctor” while jabbing her finger toward me. In my 15 years of practice, I’d never had that happen before. Equally surprising, I was not surprised by her outburst. The level of incivility from patients is at an all-time high.
Her anger was misdirected. She wanted me to write a letter to her employer excusing her from getting a vaccine. It was neither indicated nor ethical for me to do so. I did my best to redirect her, but without success. As our chief of service, I often help with service concerns and am happy to see patients who want another opinion or want to speak with the department head (aka, “the manager”). Usually I can help. Lately, it’s become harder.
Not only are such rude incidents more frequent, but they are also more dramatic and inappropriate. For example, I cannot imagine writing a complaint against a doctor stating that she must be a foreign medical grad (as it happens, she’s Ivy League-trained) or demanding money back when a biopsy result turned out to be benign, or threatening to report a doctor to the medical board because he failed to schedule a follow-up appointment (that doctor had been retired for months). Patients have hung up on our staff mid-sentence and slammed a clinic door when they left in a huff. Why are so many previously sensible people throwing childlike tantrums?
It’s the same phenomenon happening to our fellow service agents across all industries. The Federal Aviation Administration’s graph of unruly passenger incidents is a flat line from 1995 to 2019, then it goes straight vertical. A recent survey showed that Americans’ sense of civility is low and worse, that people’s expectations that civility will improve is going down. It’s palpable. Last month, I witnessed a man and woman screaming at each other over Christmas lights in a busy store. An army of aproned walkie-talkie staff surrounded them and escorted them out – their coordination and efficiency clearly indicated they’d done this before. Customers everywhere are mad, frustrated, disenfranchised. Lately, a lot of things just are not working out for them. Supplies are out. Kids are sent home from school. No elective surgery appointments are available. The insta-gratification they’ve grown accustomed to from Amazon and DoorDash is colliding with the reality that not everything works that way.
The word “patient’’ you’ll recall comes from the Latin “patior,” meaning to suffer or bear. With virus variants raging, inflation growing, and call center wait times approaching infinity, many of our patients, it seems, cannot bear any more. I’m confident this situation will improve and our patients will be more reasonable in their expectations, but I am afraid that, in the end, we’ll have lost some decorum and dignity that we may never find again in medicine.
For my potty-mouthed patient, I made an excuse to leave the room to get my dermatoscope and walked out. It gave her time to calm down. I returned in a few minutes to do a skin exam. As I was wrapping up, I advised her that she cannot raise her voice or use offensive language and that she should know that I and everyone in our office cares about her and wants to help. She did apologize for her behavior, but then had to add that, if I really cared, I’d write the letter for her.
I guess the customer is not always right.
Dr. Benabio is director of Healthcare Transformation and chief of dermatology at Kaiser Permanente San Diego. The opinions expressed in this column are his own and do not represent those of Kaiser Permanente. Dr. Benabio is @Dermdoc on Twitter. Write to him at [email protected]
A patient swore at me the other day. Not as in “she used a curse word.” As in she spewed fury, spitting out a vulgar, adverbial word before “... terrible doctor” while jabbing her finger toward me. In my 15 years of practice, I’d never had that happen before. Equally surprising, I was not surprised by her outburst. The level of incivility from patients is at an all-time high.
Her anger was misdirected. She wanted me to write a letter to her employer excusing her from getting a vaccine. It was neither indicated nor ethical for me to do so. I did my best to redirect her, but without success. As our chief of service, I often help with service concerns and am happy to see patients who want another opinion or want to speak with the department head (aka, “the manager”). Usually I can help. Lately, it’s become harder.
Not only are such rude incidents more frequent, but they are also more dramatic and inappropriate. For example, I cannot imagine writing a complaint against a doctor stating that she must be a foreign medical grad (as it happens, she’s Ivy League-trained) or demanding money back when a biopsy result turned out to be benign, or threatening to report a doctor to the medical board because he failed to schedule a follow-up appointment (that doctor had been retired for months). Patients have hung up on our staff mid-sentence and slammed a clinic door when they left in a huff. Why are so many previously sensible people throwing childlike tantrums?
It’s the same phenomenon happening to our fellow service agents across all industries. The Federal Aviation Administration’s graph of unruly passenger incidents is a flat line from 1995 to 2019, then it goes straight vertical. A recent survey showed that Americans’ sense of civility is low and worse, that people’s expectations that civility will improve is going down. It’s palpable. Last month, I witnessed a man and woman screaming at each other over Christmas lights in a busy store. An army of aproned walkie-talkie staff surrounded them and escorted them out – their coordination and efficiency clearly indicated they’d done this before. Customers everywhere are mad, frustrated, disenfranchised. Lately, a lot of things just are not working out for them. Supplies are out. Kids are sent home from school. No elective surgery appointments are available. The insta-gratification they’ve grown accustomed to from Amazon and DoorDash is colliding with the reality that not everything works that way.
The word “patient’’ you’ll recall comes from the Latin “patior,” meaning to suffer or bear. With virus variants raging, inflation growing, and call center wait times approaching infinity, many of our patients, it seems, cannot bear any more. I’m confident this situation will improve and our patients will be more reasonable in their expectations, but I am afraid that, in the end, we’ll have lost some decorum and dignity that we may never find again in medicine.
For my potty-mouthed patient, I made an excuse to leave the room to get my dermatoscope and walked out. It gave her time to calm down. I returned in a few minutes to do a skin exam. As I was wrapping up, I advised her that she cannot raise her voice or use offensive language and that she should know that I and everyone in our office cares about her and wants to help. She did apologize for her behavior, but then had to add that, if I really cared, I’d write the letter for her.
I guess the customer is not always right.
Dr. Benabio is director of Healthcare Transformation and chief of dermatology at Kaiser Permanente San Diego. The opinions expressed in this column are his own and do not represent those of Kaiser Permanente. Dr. Benabio is @Dermdoc on Twitter. Write to him at [email protected]
Study finds genetic factor for COVID smell and taste loss
, according to a new study published in the journal Nature Genetics
The finding could eventually help the 1.6 million people in the United States who still can’t smell or have had a change in their ability to smell more than 6 months after getting the coronavirus. The exact cause related to COVID-19 is still unknown, but researchers believe it could be because of damage in a part of the nose called the olfactory epithelium.
“How we get from infection to smell loss remains unclear,” Justin Turner, MD, an associate professor of otolaryngology at Vanderbilt University, Nashville, Tenn., told NBC News. Dr. Turner was not part of the research team.
“Early data suggest that supporting cells of the olfactory epithelium are the ones mostly being infected by the virus, and presumably this leads to the death of the neurons themselves,” he said. “But we don’t really, really know why and when that happens, and why it seems to preferentially happen in certain individuals.”
Researchers at 23andMe, a genomics and biotechnology company, did the study as part of a larger COVID-19 project, which includes people in the United States and the United Kingdom. They analyzed data from nearly 70,000 people who took online surveys after receiving a positive coronavirus test. Among those, 68% reported a loss of smell or taste as a symptom.
The study team compared the genetic differences between those who lost their sense of smell and taste and those who didn’t. They found that a location near two olfactory genes – UGT2A1 and UGT2A2 – is associated with COVID-19 loss of smell and taste. The genetic risk factor makes it 11% more likely for a person with COVID-19 to lose their sense of smell or taste.
The research team also found that women were 11% more likely than men to report a loss of smell and taste. About 73% of those who reported a loss of smell and taste were ages 26-35.
The researchers aren’t sure how the genes are involved, though they suspect that infected cells could lead to smell loss. Typically, the genes are expressed in tissue inside the nose involved with smell and play a role in processing things that have an odor. To use the findings, researchers need to learn more about the genes, how they are expressed, and what their functions are, NBC News reported.
The findings could help lead to treatments. Other research has shown that the loss of taste and smell is related to a “failure to protect the sensory cells of the nose and tongue from viral infection,” Danielle Reed, PhD, associate director of the Monell Chemical Senses Center in Philadelphia, told NBC News. She was not part of the research team but studies person-to-person differences in the loss of these senses because of COVID-19.
“This study suggests a different direction,” she said. “The pathways that break down the chemicals that cause taste and smell in the first place might be over or underactive, reducing or distorting the ability to taste and smell.”
A version of this article first appeared on WebMD.com.
, according to a new study published in the journal Nature Genetics
The finding could eventually help the 1.6 million people in the United States who still can’t smell or have had a change in their ability to smell more than 6 months after getting the coronavirus. The exact cause related to COVID-19 is still unknown, but researchers believe it could be because of damage in a part of the nose called the olfactory epithelium.
“How we get from infection to smell loss remains unclear,” Justin Turner, MD, an associate professor of otolaryngology at Vanderbilt University, Nashville, Tenn., told NBC News. Dr. Turner was not part of the research team.
“Early data suggest that supporting cells of the olfactory epithelium are the ones mostly being infected by the virus, and presumably this leads to the death of the neurons themselves,” he said. “But we don’t really, really know why and when that happens, and why it seems to preferentially happen in certain individuals.”
Researchers at 23andMe, a genomics and biotechnology company, did the study as part of a larger COVID-19 project, which includes people in the United States and the United Kingdom. They analyzed data from nearly 70,000 people who took online surveys after receiving a positive coronavirus test. Among those, 68% reported a loss of smell or taste as a symptom.
The study team compared the genetic differences between those who lost their sense of smell and taste and those who didn’t. They found that a location near two olfactory genes – UGT2A1 and UGT2A2 – is associated with COVID-19 loss of smell and taste. The genetic risk factor makes it 11% more likely for a person with COVID-19 to lose their sense of smell or taste.
The research team also found that women were 11% more likely than men to report a loss of smell and taste. About 73% of those who reported a loss of smell and taste were ages 26-35.
The researchers aren’t sure how the genes are involved, though they suspect that infected cells could lead to smell loss. Typically, the genes are expressed in tissue inside the nose involved with smell and play a role in processing things that have an odor. To use the findings, researchers need to learn more about the genes, how they are expressed, and what their functions are, NBC News reported.
The findings could help lead to treatments. Other research has shown that the loss of taste and smell is related to a “failure to protect the sensory cells of the nose and tongue from viral infection,” Danielle Reed, PhD, associate director of the Monell Chemical Senses Center in Philadelphia, told NBC News. She was not part of the research team but studies person-to-person differences in the loss of these senses because of COVID-19.
“This study suggests a different direction,” she said. “The pathways that break down the chemicals that cause taste and smell in the first place might be over or underactive, reducing or distorting the ability to taste and smell.”
A version of this article first appeared on WebMD.com.
, according to a new study published in the journal Nature Genetics
The finding could eventually help the 1.6 million people in the United States who still can’t smell or have had a change in their ability to smell more than 6 months after getting the coronavirus. The exact cause related to COVID-19 is still unknown, but researchers believe it could be because of damage in a part of the nose called the olfactory epithelium.
“How we get from infection to smell loss remains unclear,” Justin Turner, MD, an associate professor of otolaryngology at Vanderbilt University, Nashville, Tenn., told NBC News. Dr. Turner was not part of the research team.
“Early data suggest that supporting cells of the olfactory epithelium are the ones mostly being infected by the virus, and presumably this leads to the death of the neurons themselves,” he said. “But we don’t really, really know why and when that happens, and why it seems to preferentially happen in certain individuals.”
Researchers at 23andMe, a genomics and biotechnology company, did the study as part of a larger COVID-19 project, which includes people in the United States and the United Kingdom. They analyzed data from nearly 70,000 people who took online surveys after receiving a positive coronavirus test. Among those, 68% reported a loss of smell or taste as a symptom.
The study team compared the genetic differences between those who lost their sense of smell and taste and those who didn’t. They found that a location near two olfactory genes – UGT2A1 and UGT2A2 – is associated with COVID-19 loss of smell and taste. The genetic risk factor makes it 11% more likely for a person with COVID-19 to lose their sense of smell or taste.
The research team also found that women were 11% more likely than men to report a loss of smell and taste. About 73% of those who reported a loss of smell and taste were ages 26-35.
The researchers aren’t sure how the genes are involved, though they suspect that infected cells could lead to smell loss. Typically, the genes are expressed in tissue inside the nose involved with smell and play a role in processing things that have an odor. To use the findings, researchers need to learn more about the genes, how they are expressed, and what their functions are, NBC News reported.
The findings could help lead to treatments. Other research has shown that the loss of taste and smell is related to a “failure to protect the sensory cells of the nose and tongue from viral infection,” Danielle Reed, PhD, associate director of the Monell Chemical Senses Center in Philadelphia, told NBC News. She was not part of the research team but studies person-to-person differences in the loss of these senses because of COVID-19.
“This study suggests a different direction,” she said. “The pathways that break down the chemicals that cause taste and smell in the first place might be over or underactive, reducing or distorting the ability to taste and smell.”
A version of this article first appeared on WebMD.com.
FROM NATURE GENETICS
Fourth vaccine shot less effective against Omicron, Israeli study says
, according to new research at an Israeli hospital.
The preliminary results, released on Jan. 17, challenge the idea of giving a second booster dose to slow the spread of the coronavirus, according to USA Today.
“Despite increased antibody levels, the fourth vaccine only offers a partial defense against the virus,” Gili Regev-Yochay, MD, director of the hospital’s infection prevention and control units, told reporters.
“The vaccines, which were more effective against previous variants, offer less protection versus Omicron,” she said.
In a clinical trial, 274 medical workers at Sheba Medical Center near Tel Aviv received a fourth vaccine dose in December – 154 got the Pfizer vaccine and 120 got the Moderna vaccine – after previously getting three Pfizer shots.
Both groups received a boost in antibodies that was “slightly higher” than after the third shot, Dr. Regev-Yochay said. But when compared with a control group that didn’t receive the fourth dose, the extra boost didn’t prevent the spread of Omicron.
“We see many infected with Omicron who received the fourth dose,” Dr. Regev-Yochay said. “Granted, a bit less than in the control group, but still a lot of infections.”
Some public health officials in Israel say the campaign for fourth doses is still worthwhile, according to The Times of Israel. The vaccine still works well against the Alpha and Delta variants, Dr. Regev-Yochay said, and a fourth shot should go to older adults and those who face higher risks for severe COVID-19.
Hours after releasing the preliminary results, Sheba Medical Center published a statement calling for “continuing the vaccination drive for risk groups at this time, even though the vaccine doesn’t provide optimal protection against getting infected with the variant.” News outlets reported that the hospital was pressured into issuing the statement after Israel’s Health Ministry didn’t like the release of the early study results, The Times of Israel reported.
The second booster “returns the level of antibodies to what it was at the beginning of the third booster,” Nachman Ash, MD, director of Israel’s Health Ministry, told Channel 13 TV in Israel, according to The Associated Press.
“That has great importance, especially among the older population,” he said.
As of Sunday, more than 500,000 people in Israel had received fourth doses since the country began offering them last month to medical workers, immunocompromised patients, and people ages 60 years and older, the AP reported. At the same time, the country has faced a recent coronavirus surge that has led to record-breaking numbers of cases and rising hospitalizations.
On Tuesday, the Israeli government said it would shorten the mandatory quarantine period from 7 days to 5 days, the AP reported.
“This decision will enable us to continue safeguarding public health on the one hand and to keep the economy going at this time on the other, even though it is difficult, so that we can get through this wave safely,” Prime Minister Naftali Bennett said.
A version of this article first appeared on WebMD.com.
, according to new research at an Israeli hospital.
The preliminary results, released on Jan. 17, challenge the idea of giving a second booster dose to slow the spread of the coronavirus, according to USA Today.
“Despite increased antibody levels, the fourth vaccine only offers a partial defense against the virus,” Gili Regev-Yochay, MD, director of the hospital’s infection prevention and control units, told reporters.
“The vaccines, which were more effective against previous variants, offer less protection versus Omicron,” she said.
In a clinical trial, 274 medical workers at Sheba Medical Center near Tel Aviv received a fourth vaccine dose in December – 154 got the Pfizer vaccine and 120 got the Moderna vaccine – after previously getting three Pfizer shots.
Both groups received a boost in antibodies that was “slightly higher” than after the third shot, Dr. Regev-Yochay said. But when compared with a control group that didn’t receive the fourth dose, the extra boost didn’t prevent the spread of Omicron.
“We see many infected with Omicron who received the fourth dose,” Dr. Regev-Yochay said. “Granted, a bit less than in the control group, but still a lot of infections.”
Some public health officials in Israel say the campaign for fourth doses is still worthwhile, according to The Times of Israel. The vaccine still works well against the Alpha and Delta variants, Dr. Regev-Yochay said, and a fourth shot should go to older adults and those who face higher risks for severe COVID-19.
Hours after releasing the preliminary results, Sheba Medical Center published a statement calling for “continuing the vaccination drive for risk groups at this time, even though the vaccine doesn’t provide optimal protection against getting infected with the variant.” News outlets reported that the hospital was pressured into issuing the statement after Israel’s Health Ministry didn’t like the release of the early study results, The Times of Israel reported.
The second booster “returns the level of antibodies to what it was at the beginning of the third booster,” Nachman Ash, MD, director of Israel’s Health Ministry, told Channel 13 TV in Israel, according to The Associated Press.
“That has great importance, especially among the older population,” he said.
As of Sunday, more than 500,000 people in Israel had received fourth doses since the country began offering them last month to medical workers, immunocompromised patients, and people ages 60 years and older, the AP reported. At the same time, the country has faced a recent coronavirus surge that has led to record-breaking numbers of cases and rising hospitalizations.
On Tuesday, the Israeli government said it would shorten the mandatory quarantine period from 7 days to 5 days, the AP reported.
“This decision will enable us to continue safeguarding public health on the one hand and to keep the economy going at this time on the other, even though it is difficult, so that we can get through this wave safely,” Prime Minister Naftali Bennett said.
A version of this article first appeared on WebMD.com.
, according to new research at an Israeli hospital.
The preliminary results, released on Jan. 17, challenge the idea of giving a second booster dose to slow the spread of the coronavirus, according to USA Today.
“Despite increased antibody levels, the fourth vaccine only offers a partial defense against the virus,” Gili Regev-Yochay, MD, director of the hospital’s infection prevention and control units, told reporters.
“The vaccines, which were more effective against previous variants, offer less protection versus Omicron,” she said.
In a clinical trial, 274 medical workers at Sheba Medical Center near Tel Aviv received a fourth vaccine dose in December – 154 got the Pfizer vaccine and 120 got the Moderna vaccine – after previously getting three Pfizer shots.
Both groups received a boost in antibodies that was “slightly higher” than after the third shot, Dr. Regev-Yochay said. But when compared with a control group that didn’t receive the fourth dose, the extra boost didn’t prevent the spread of Omicron.
“We see many infected with Omicron who received the fourth dose,” Dr. Regev-Yochay said. “Granted, a bit less than in the control group, but still a lot of infections.”
Some public health officials in Israel say the campaign for fourth doses is still worthwhile, according to The Times of Israel. The vaccine still works well against the Alpha and Delta variants, Dr. Regev-Yochay said, and a fourth shot should go to older adults and those who face higher risks for severe COVID-19.
Hours after releasing the preliminary results, Sheba Medical Center published a statement calling for “continuing the vaccination drive for risk groups at this time, even though the vaccine doesn’t provide optimal protection against getting infected with the variant.” News outlets reported that the hospital was pressured into issuing the statement after Israel’s Health Ministry didn’t like the release of the early study results, The Times of Israel reported.
The second booster “returns the level of antibodies to what it was at the beginning of the third booster,” Nachman Ash, MD, director of Israel’s Health Ministry, told Channel 13 TV in Israel, according to The Associated Press.
“That has great importance, especially among the older population,” he said.
As of Sunday, more than 500,000 people in Israel had received fourth doses since the country began offering them last month to medical workers, immunocompromised patients, and people ages 60 years and older, the AP reported. At the same time, the country has faced a recent coronavirus surge that has led to record-breaking numbers of cases and rising hospitalizations.
On Tuesday, the Israeli government said it would shorten the mandatory quarantine period from 7 days to 5 days, the AP reported.
“This decision will enable us to continue safeguarding public health on the one hand and to keep the economy going at this time on the other, even though it is difficult, so that we can get through this wave safely,” Prime Minister Naftali Bennett said.
A version of this article first appeared on WebMD.com.
Children and COVID: U.S. sees almost 1 million new cases
Another record week for COVID-19 brought almost 1 million new cases to the children of the United States, according to new data from the American Academy of Pediatrics and the Children’s Hospital Association.
The pre-Omicron high for new cases in a week – 252,000 during the Delta surge of the late summer and early fall – has been surpassed each of the last 3 weeks and now stands at 981,000 (Jan. 7-13), according to the AAP/CHA weekly COVID-19 report. Over the 3-week stretch from Dec. 17 to Jan. 13, weekly cases increased by 394%.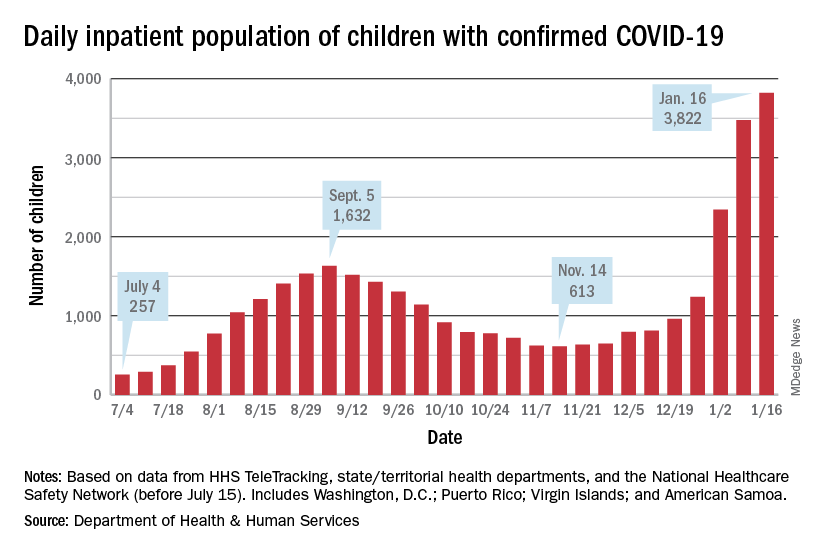
Hospitalizations also climbed to new heights, as daily admissions reached 1.23 per 100,000 children on Jan. 14, an increase of 547% since Nov. 30, when the rate was 0.19 per 100,000. Before Omicron, the highest rate for children was 0.47 per 100,000, based on data from the Centers for Disease Control and Prevention.
The inpatient population count, meanwhile, has followed suit. On Jan. 16, there were 3,822 children hospitalized in pediatric inpatient beds with laboratory-confirmed COVID-19, which is 523% higher than the 613 children who were hospitalized on Nov. 14, according to the Department of Health & Human Services. In the last week, though, the population was up by just 10%.
The one thing that has not surged in the last few weeks is vaccination. Among children aged 5-11 years, the weekly count of those who have received at least one dose dropped by 34% over the last 5 weeks, falling from 527,000 for Dec.11-17 to 347,000 during Jan. 8-14, the CDC said on the COVID Data Tracker, which also noted that just 18.4% of this age group is fully vaccinated.
The situation was reversed in children aged 12-15, who were up by 36% over that same time, but their numbers were much smaller: 78,000 for the week of Dec. 11-17 and 106,000 for Jan. 8-14. Those aged 16-17 were up by just 4% over that 5-week span, the CDC data show.
Over the course of the entire pandemic, almost 9.5 million cases of COVID-19 in children have been reported, and children represent 17.8% of all cases reported in 49 states (excluding New York but including New York City), the District of Columbia, Puerto Rico, and Guam, the AAP and CHA said in their report.
Three states (Alabama, Nebraska, and Texas) stopped public reporting over the summer, but many states count individuals up to age 19 as children, and others (South Carolina, Tennessee, and West Virginia) go up to age 20, the AAP and CHA noted. The CDC, by comparison, puts the number of cases for those aged 0-17 at 8.3 million, but that estimate is based on only 51 million of the nearly 67 million U.S. cases as of Jan. 18.
Another record week for COVID-19 brought almost 1 million new cases to the children of the United States, according to new data from the American Academy of Pediatrics and the Children’s Hospital Association.
The pre-Omicron high for new cases in a week – 252,000 during the Delta surge of the late summer and early fall – has been surpassed each of the last 3 weeks and now stands at 981,000 (Jan. 7-13), according to the AAP/CHA weekly COVID-19 report. Over the 3-week stretch from Dec. 17 to Jan. 13, weekly cases increased by 394%.
Hospitalizations also climbed to new heights, as daily admissions reached 1.23 per 100,000 children on Jan. 14, an increase of 547% since Nov. 30, when the rate was 0.19 per 100,000. Before Omicron, the highest rate for children was 0.47 per 100,000, based on data from the Centers for Disease Control and Prevention.
The inpatient population count, meanwhile, has followed suit. On Jan. 16, there were 3,822 children hospitalized in pediatric inpatient beds with laboratory-confirmed COVID-19, which is 523% higher than the 613 children who were hospitalized on Nov. 14, according to the Department of Health & Human Services. In the last week, though, the population was up by just 10%.
The one thing that has not surged in the last few weeks is vaccination. Among children aged 5-11 years, the weekly count of those who have received at least one dose dropped by 34% over the last 5 weeks, falling from 527,000 for Dec.11-17 to 347,000 during Jan. 8-14, the CDC said on the COVID Data Tracker, which also noted that just 18.4% of this age group is fully vaccinated.
The situation was reversed in children aged 12-15, who were up by 36% over that same time, but their numbers were much smaller: 78,000 for the week of Dec. 11-17 and 106,000 for Jan. 8-14. Those aged 16-17 were up by just 4% over that 5-week span, the CDC data show.
Over the course of the entire pandemic, almost 9.5 million cases of COVID-19 in children have been reported, and children represent 17.8% of all cases reported in 49 states (excluding New York but including New York City), the District of Columbia, Puerto Rico, and Guam, the AAP and CHA said in their report.
Three states (Alabama, Nebraska, and Texas) stopped public reporting over the summer, but many states count individuals up to age 19 as children, and others (South Carolina, Tennessee, and West Virginia) go up to age 20, the AAP and CHA noted. The CDC, by comparison, puts the number of cases for those aged 0-17 at 8.3 million, but that estimate is based on only 51 million of the nearly 67 million U.S. cases as of Jan. 18.
Another record week for COVID-19 brought almost 1 million new cases to the children of the United States, according to new data from the American Academy of Pediatrics and the Children’s Hospital Association.
The pre-Omicron high for new cases in a week – 252,000 during the Delta surge of the late summer and early fall – has been surpassed each of the last 3 weeks and now stands at 981,000 (Jan. 7-13), according to the AAP/CHA weekly COVID-19 report. Over the 3-week stretch from Dec. 17 to Jan. 13, weekly cases increased by 394%.
Hospitalizations also climbed to new heights, as daily admissions reached 1.23 per 100,000 children on Jan. 14, an increase of 547% since Nov. 30, when the rate was 0.19 per 100,000. Before Omicron, the highest rate for children was 0.47 per 100,000, based on data from the Centers for Disease Control and Prevention.
The inpatient population count, meanwhile, has followed suit. On Jan. 16, there were 3,822 children hospitalized in pediatric inpatient beds with laboratory-confirmed COVID-19, which is 523% higher than the 613 children who were hospitalized on Nov. 14, according to the Department of Health & Human Services. In the last week, though, the population was up by just 10%.
The one thing that has not surged in the last few weeks is vaccination. Among children aged 5-11 years, the weekly count of those who have received at least one dose dropped by 34% over the last 5 weeks, falling from 527,000 for Dec.11-17 to 347,000 during Jan. 8-14, the CDC said on the COVID Data Tracker, which also noted that just 18.4% of this age group is fully vaccinated.
The situation was reversed in children aged 12-15, who were up by 36% over that same time, but their numbers were much smaller: 78,000 for the week of Dec. 11-17 and 106,000 for Jan. 8-14. Those aged 16-17 were up by just 4% over that 5-week span, the CDC data show.
Over the course of the entire pandemic, almost 9.5 million cases of COVID-19 in children have been reported, and children represent 17.8% of all cases reported in 49 states (excluding New York but including New York City), the District of Columbia, Puerto Rico, and Guam, the AAP and CHA said in their report.
Three states (Alabama, Nebraska, and Texas) stopped public reporting over the summer, but many states count individuals up to age 19 as children, and others (South Carolina, Tennessee, and West Virginia) go up to age 20, the AAP and CHA noted. The CDC, by comparison, puts the number of cases for those aged 0-17 at 8.3 million, but that estimate is based on only 51 million of the nearly 67 million U.S. cases as of Jan. 18.
Negative home COVID test no ‘free pass’ for kids, study finds
With the country looking increasingly to rapid testing as an off-ramp from the COVID-19 pandemic, a new study shows that the performance of the tests in children falls below standards set by regulatory agencies in the United States and elsewhere for diagnostic accuracy.
Experts said the findings, from a meta-analysis by researchers in the United Kingdom and Germany, underscore that, while a positive result on a rapid test is almost certainly an indicator of infection, negative results often are unreliable and can lead to a false sense of security.
“Real-life performance of current antigen tests for professional use in pediatric populations is below the minimum performance criteria set by WHO, the United States Food and Drug Administration, or the Medicines and Healthcare products Regulatory Agency (U.K.),” according to Naomi Fujita-Rohwerder, PhD, a research associate at the Cologne-based German Institute for Quality and Efficiency in Health Care (IQWiG), and her colleagues, whose study appears in BMJ Evidence-Based Medicine.
The researchers said that the study suggests that performance of rapid testing in a pediatric population is comparable to that in adults. However, they said they could not identify any studies investigating self-testing in children, which also could affect test performance.
Egon Ozer, MD, PhD, director of the center for pathogen genomics and microbial evolution at Northwestern University in Chicago, said the finding that specificity was high but sensitivity was middling “suggests that we should be very careful about interpreting negative antigen test results in children and recognize that there is a fair amount of uncertainty in the tests in this situation.”
Researchers from IQWiG, which examines the advantages and disadvantages of medical interventions, and the University of Manchester (England), conducted the systematic review and meta-analysis, which they described as the first of its kind to evaluate the diagnostic accuracy of rapid point-of-care tests for current SARS-CoV-2 infections in children.
They compiled information from 17 studies with a total 6,355 participants. They compared all antigen tests to reverse-transcription polymerase chain reaction (PCR). The studies compared eight antigen tests from six different brands. The rapid antigen tests, available from pharmacies and online stores, are widely used for self-testing in schools and testing toddlers before kindergarten.
The pooled diagnostic sensitivity of antigen tests was 64.2% and specificity was 99.1%.
Dr. Ozer noted that the analysis “was not able to address important outstanding questions such as the likelihood of transmitting infection with a false-negative antigen test versus a true-negative antigen test or how much repeated testing can increase the sensitivity.”
“In Europe, we don’t know how most tests perform in real life,” Dr. Fujita-Rohwerder said. “And even in countries like the United States, where market access is more stringent, we don’t know whether self-testing performed by children or sample collection in toddlers by laypersons has a significant impact on the diagnostic accuracy. Also, diagnostic accuracy estimates reported in our study may not apply to the current omicron or future variants of SARS-CoV-2 or vaccinated children. Hopefully, these essential gaps in the evidence will get addressed soon.”
Dr. Ozer said one takeaway from this study is negative antigen tests should not be considered a “free pass” in children, especially if the child is symptomatic, has been recently exposed to COVID-19, or is planning to spend time with individuals with conditions that place them at high risk for complications of COVID-19 infection. “In such cases, consider getting PCR testing or at least performing a repeat antigen test 36-48 hours after the first negative,” he said.
Dr. Fujita-Rohwerder said the low diagnostic sensitivity may affect the use of the tests. The gaps in evidence her group found in their study point to research needed to support evidence-based decision-making. “In particular, evidence is needed on real-life performance of tests in schools, self-testing performed by children, and kindergarten, [particularly] sample collection in toddlers by laypersons,” she said.
However, she stressed, testing is only a single measure. “Effectively reducing the spread of SARS-CoV-2 during the current pandemic requires multilayered mitigation measures,” she said. “Rapid testing represents one single layer. It can have its use at the population level, even though the sensitivity of antigen tests is lower than expected. However, antigen-based rapid testing is not a magic bullet: If your kid tests negative, do not disregard other mitigation measures.”
Edward Campbell, PhD, a virologist at Loyola University of Chicago, who serves on the board of LaGrange Elementary School District 102 outside Chicago, said the findings were unsurprising.
“This study generally looks consistent with what is known for adults. These rapid antigen tests are less sensitive than other tests,” said Dr. Campbell, who also runs a testing company for private schools in the Chicago area using reverse transcription-loop-mediated isothermal amplification technology. Even so, he said, “These tests are still effective at identifying people who are infectious to some degree. Never miss an opportunity to test.”
Dr. Fujita-Rohwerder disclosed no relevant financial conflicts of interest. Dr. Campbell owns Safeguard Surveillance.
With the country looking increasingly to rapid testing as an off-ramp from the COVID-19 pandemic, a new study shows that the performance of the tests in children falls below standards set by regulatory agencies in the United States and elsewhere for diagnostic accuracy.
Experts said the findings, from a meta-analysis by researchers in the United Kingdom and Germany, underscore that, while a positive result on a rapid test is almost certainly an indicator of infection, negative results often are unreliable and can lead to a false sense of security.
“Real-life performance of current antigen tests for professional use in pediatric populations is below the minimum performance criteria set by WHO, the United States Food and Drug Administration, or the Medicines and Healthcare products Regulatory Agency (U.K.),” according to Naomi Fujita-Rohwerder, PhD, a research associate at the Cologne-based German Institute for Quality and Efficiency in Health Care (IQWiG), and her colleagues, whose study appears in BMJ Evidence-Based Medicine.
The researchers said that the study suggests that performance of rapid testing in a pediatric population is comparable to that in adults. However, they said they could not identify any studies investigating self-testing in children, which also could affect test performance.
Egon Ozer, MD, PhD, director of the center for pathogen genomics and microbial evolution at Northwestern University in Chicago, said the finding that specificity was high but sensitivity was middling “suggests that we should be very careful about interpreting negative antigen test results in children and recognize that there is a fair amount of uncertainty in the tests in this situation.”
Researchers from IQWiG, which examines the advantages and disadvantages of medical interventions, and the University of Manchester (England), conducted the systematic review and meta-analysis, which they described as the first of its kind to evaluate the diagnostic accuracy of rapid point-of-care tests for current SARS-CoV-2 infections in children.
They compiled information from 17 studies with a total 6,355 participants. They compared all antigen tests to reverse-transcription polymerase chain reaction (PCR). The studies compared eight antigen tests from six different brands. The rapid antigen tests, available from pharmacies and online stores, are widely used for self-testing in schools and testing toddlers before kindergarten.
The pooled diagnostic sensitivity of antigen tests was 64.2% and specificity was 99.1%.
Dr. Ozer noted that the analysis “was not able to address important outstanding questions such as the likelihood of transmitting infection with a false-negative antigen test versus a true-negative antigen test or how much repeated testing can increase the sensitivity.”
“In Europe, we don’t know how most tests perform in real life,” Dr. Fujita-Rohwerder said. “And even in countries like the United States, where market access is more stringent, we don’t know whether self-testing performed by children or sample collection in toddlers by laypersons has a significant impact on the diagnostic accuracy. Also, diagnostic accuracy estimates reported in our study may not apply to the current omicron or future variants of SARS-CoV-2 or vaccinated children. Hopefully, these essential gaps in the evidence will get addressed soon.”
Dr. Ozer said one takeaway from this study is negative antigen tests should not be considered a “free pass” in children, especially if the child is symptomatic, has been recently exposed to COVID-19, or is planning to spend time with individuals with conditions that place them at high risk for complications of COVID-19 infection. “In such cases, consider getting PCR testing or at least performing a repeat antigen test 36-48 hours after the first negative,” he said.
Dr. Fujita-Rohwerder said the low diagnostic sensitivity may affect the use of the tests. The gaps in evidence her group found in their study point to research needed to support evidence-based decision-making. “In particular, evidence is needed on real-life performance of tests in schools, self-testing performed by children, and kindergarten, [particularly] sample collection in toddlers by laypersons,” she said.
However, she stressed, testing is only a single measure. “Effectively reducing the spread of SARS-CoV-2 during the current pandemic requires multilayered mitigation measures,” she said. “Rapid testing represents one single layer. It can have its use at the population level, even though the sensitivity of antigen tests is lower than expected. However, antigen-based rapid testing is not a magic bullet: If your kid tests negative, do not disregard other mitigation measures.”
Edward Campbell, PhD, a virologist at Loyola University of Chicago, who serves on the board of LaGrange Elementary School District 102 outside Chicago, said the findings were unsurprising.
“This study generally looks consistent with what is known for adults. These rapid antigen tests are less sensitive than other tests,” said Dr. Campbell, who also runs a testing company for private schools in the Chicago area using reverse transcription-loop-mediated isothermal amplification technology. Even so, he said, “These tests are still effective at identifying people who are infectious to some degree. Never miss an opportunity to test.”
Dr. Fujita-Rohwerder disclosed no relevant financial conflicts of interest. Dr. Campbell owns Safeguard Surveillance.
With the country looking increasingly to rapid testing as an off-ramp from the COVID-19 pandemic, a new study shows that the performance of the tests in children falls below standards set by regulatory agencies in the United States and elsewhere for diagnostic accuracy.
Experts said the findings, from a meta-analysis by researchers in the United Kingdom and Germany, underscore that, while a positive result on a rapid test is almost certainly an indicator of infection, negative results often are unreliable and can lead to a false sense of security.
“Real-life performance of current antigen tests for professional use in pediatric populations is below the minimum performance criteria set by WHO, the United States Food and Drug Administration, or the Medicines and Healthcare products Regulatory Agency (U.K.),” according to Naomi Fujita-Rohwerder, PhD, a research associate at the Cologne-based German Institute for Quality and Efficiency in Health Care (IQWiG), and her colleagues, whose study appears in BMJ Evidence-Based Medicine.
The researchers said that the study suggests that performance of rapid testing in a pediatric population is comparable to that in adults. However, they said they could not identify any studies investigating self-testing in children, which also could affect test performance.
Egon Ozer, MD, PhD, director of the center for pathogen genomics and microbial evolution at Northwestern University in Chicago, said the finding that specificity was high but sensitivity was middling “suggests that we should be very careful about interpreting negative antigen test results in children and recognize that there is a fair amount of uncertainty in the tests in this situation.”
Researchers from IQWiG, which examines the advantages and disadvantages of medical interventions, and the University of Manchester (England), conducted the systematic review and meta-analysis, which they described as the first of its kind to evaluate the diagnostic accuracy of rapid point-of-care tests for current SARS-CoV-2 infections in children.
They compiled information from 17 studies with a total 6,355 participants. They compared all antigen tests to reverse-transcription polymerase chain reaction (PCR). The studies compared eight antigen tests from six different brands. The rapid antigen tests, available from pharmacies and online stores, are widely used for self-testing in schools and testing toddlers before kindergarten.
The pooled diagnostic sensitivity of antigen tests was 64.2% and specificity was 99.1%.
Dr. Ozer noted that the analysis “was not able to address important outstanding questions such as the likelihood of transmitting infection with a false-negative antigen test versus a true-negative antigen test or how much repeated testing can increase the sensitivity.”
“In Europe, we don’t know how most tests perform in real life,” Dr. Fujita-Rohwerder said. “And even in countries like the United States, where market access is more stringent, we don’t know whether self-testing performed by children or sample collection in toddlers by laypersons has a significant impact on the diagnostic accuracy. Also, diagnostic accuracy estimates reported in our study may not apply to the current omicron or future variants of SARS-CoV-2 or vaccinated children. Hopefully, these essential gaps in the evidence will get addressed soon.”
Dr. Ozer said one takeaway from this study is negative antigen tests should not be considered a “free pass” in children, especially if the child is symptomatic, has been recently exposed to COVID-19, or is planning to spend time with individuals with conditions that place them at high risk for complications of COVID-19 infection. “In such cases, consider getting PCR testing or at least performing a repeat antigen test 36-48 hours after the first negative,” he said.
Dr. Fujita-Rohwerder said the low diagnostic sensitivity may affect the use of the tests. The gaps in evidence her group found in their study point to research needed to support evidence-based decision-making. “In particular, evidence is needed on real-life performance of tests in schools, self-testing performed by children, and kindergarten, [particularly] sample collection in toddlers by laypersons,” she said.
However, she stressed, testing is only a single measure. “Effectively reducing the spread of SARS-CoV-2 during the current pandemic requires multilayered mitigation measures,” she said. “Rapid testing represents one single layer. It can have its use at the population level, even though the sensitivity of antigen tests is lower than expected. However, antigen-based rapid testing is not a magic bullet: If your kid tests negative, do not disregard other mitigation measures.”
Edward Campbell, PhD, a virologist at Loyola University of Chicago, who serves on the board of LaGrange Elementary School District 102 outside Chicago, said the findings were unsurprising.
“This study generally looks consistent with what is known for adults. These rapid antigen tests are less sensitive than other tests,” said Dr. Campbell, who also runs a testing company for private schools in the Chicago area using reverse transcription-loop-mediated isothermal amplification technology. Even so, he said, “These tests are still effective at identifying people who are infectious to some degree. Never miss an opportunity to test.”
Dr. Fujita-Rohwerder disclosed no relevant financial conflicts of interest. Dr. Campbell owns Safeguard Surveillance.
BMJ EVIDENCE-BASED MEDICINE
Feds’ website for free at-home COVID tests launches day early
The Biden administration’s new no-cost, at-home testing program launched Jan. 18, a day ahead of schedule.
The administration said 500 million tests are available to be delivered to homes across the country. This accounts for half of the president’s recent pledge to purchase 1 billion free at-home COVID-19 tests to distribute to the American public.
On a Jan. 14 call with reporters, senior White House officials offered some details about the new program.
Here’s what we know so far.
How do I order my free tests?
Americans can visit COVIDtests.gov to order their rapid at-home tests. You can also order directly from the U.S. Postal Service website. After you order, you’ll receive a confirmation email that promises to send tracking information once your order ships.
What information do I need to order the tests?
You only need your name and home mailing address.
There is also an option to provide your email address to get updates on the status of your order.
What if someone needs help ordering the tests?
There will be a free call-in line for people needing more help, including those having trouble accessing the internet, according to White House officials.
What tests will be available?
There are nine at-home tests available through FDA emergency use authorization. According to the Frequently Asked Questions section of COVIDtests.gov, "You will not be able to choose the brand you order as part of this program.”
How long will it take to get the tests once I order them?
Tests are expected to ship 7 to 12 days after you order them.
But White House officials say that the time frame will likely shorten as the program gains steam.
How many can I order?
There’s a limit of four tests per residential mailing address.
For larger families, White House officials suggest trying other free testing options, like visiting COVID-19 testing sites or your local health center.
Is this a one-time opportunity?
The White House doesn’t say, but officials did mention that if you run out of your four free tests, there are many other ways to access free at-home tests, such as COVID-19 testing sites, pharmacies, and community health centers.
The free tests available through COVIDtests.gov are in addition to an estimated 375 million at-home rapid tests on the market in the U.S. this month.
When should people use a rapid at-home test?
The CDC and experts with other public health groups agree that Americans should consider using at-home rapid tests in the following situations:
- If they begin to have symptoms consistent with COVID-19;
- At least 5 days after close contact with someone who has COVID;
- If someone is indoors with a group of people who are at risk of severe disease or are unvaccinated.
Are at-home rapid tests accurate?
The U.S. Department of Health and Human Services and other federal officials confirmed through studies that all tests distributed through this program can detect the Omicron variant. These agencies also confirmed that their performance is consistent with the FDA’s emergency use authorization.
Is the website designed to handle high demand?
After the original website to sign up for health insurance under the Affordable Care Act crashed repeatedly at launch, the government says it has prepared for high demand for ordering at-home rapid tests.
The U.S. Digital Service (USDS), an organization founded after Healthcare.gov, has partnered with the Postal Service to plan for the launch.
The Postal Service has expanded its staffing, similar to what’s done during the holidays.
All orders in the continental United States will be shipped through first-class mail, with shipments to Alaska, Hawaii, U.S. territories, and military and overseas addresses sent through priority mail.
A version of this article first appeared on WebMD.com.
The Biden administration’s new no-cost, at-home testing program launched Jan. 18, a day ahead of schedule.
The administration said 500 million tests are available to be delivered to homes across the country. This accounts for half of the president’s recent pledge to purchase 1 billion free at-home COVID-19 tests to distribute to the American public.
On a Jan. 14 call with reporters, senior White House officials offered some details about the new program.
Here’s what we know so far.
How do I order my free tests?
Americans can visit COVIDtests.gov to order their rapid at-home tests. You can also order directly from the U.S. Postal Service website. After you order, you’ll receive a confirmation email that promises to send tracking information once your order ships.
What information do I need to order the tests?
You only need your name and home mailing address.
There is also an option to provide your email address to get updates on the status of your order.
What if someone needs help ordering the tests?
There will be a free call-in line for people needing more help, including those having trouble accessing the internet, according to White House officials.
What tests will be available?
There are nine at-home tests available through FDA emergency use authorization. According to the Frequently Asked Questions section of COVIDtests.gov, "You will not be able to choose the brand you order as part of this program.”
How long will it take to get the tests once I order them?
Tests are expected to ship 7 to 12 days after you order them.
But White House officials say that the time frame will likely shorten as the program gains steam.
How many can I order?
There’s a limit of four tests per residential mailing address.
For larger families, White House officials suggest trying other free testing options, like visiting COVID-19 testing sites or your local health center.
Is this a one-time opportunity?
The White House doesn’t say, but officials did mention that if you run out of your four free tests, there are many other ways to access free at-home tests, such as COVID-19 testing sites, pharmacies, and community health centers.
The free tests available through COVIDtests.gov are in addition to an estimated 375 million at-home rapid tests on the market in the U.S. this month.
When should people use a rapid at-home test?
The CDC and experts with other public health groups agree that Americans should consider using at-home rapid tests in the following situations:
- If they begin to have symptoms consistent with COVID-19;
- At least 5 days after close contact with someone who has COVID;
- If someone is indoors with a group of people who are at risk of severe disease or are unvaccinated.
Are at-home rapid tests accurate?
The U.S. Department of Health and Human Services and other federal officials confirmed through studies that all tests distributed through this program can detect the Omicron variant. These agencies also confirmed that their performance is consistent with the FDA’s emergency use authorization.
Is the website designed to handle high demand?
After the original website to sign up for health insurance under the Affordable Care Act crashed repeatedly at launch, the government says it has prepared for high demand for ordering at-home rapid tests.
The U.S. Digital Service (USDS), an organization founded after Healthcare.gov, has partnered with the Postal Service to plan for the launch.
The Postal Service has expanded its staffing, similar to what’s done during the holidays.
All orders in the continental United States will be shipped through first-class mail, with shipments to Alaska, Hawaii, U.S. territories, and military and overseas addresses sent through priority mail.
A version of this article first appeared on WebMD.com.
The Biden administration’s new no-cost, at-home testing program launched Jan. 18, a day ahead of schedule.
The administration said 500 million tests are available to be delivered to homes across the country. This accounts for half of the president’s recent pledge to purchase 1 billion free at-home COVID-19 tests to distribute to the American public.
On a Jan. 14 call with reporters, senior White House officials offered some details about the new program.
Here’s what we know so far.
How do I order my free tests?
Americans can visit COVIDtests.gov to order their rapid at-home tests. You can also order directly from the U.S. Postal Service website. After you order, you’ll receive a confirmation email that promises to send tracking information once your order ships.
What information do I need to order the tests?
You only need your name and home mailing address.
There is also an option to provide your email address to get updates on the status of your order.
What if someone needs help ordering the tests?
There will be a free call-in line for people needing more help, including those having trouble accessing the internet, according to White House officials.
What tests will be available?
There are nine at-home tests available through FDA emergency use authorization. According to the Frequently Asked Questions section of COVIDtests.gov, "You will not be able to choose the brand you order as part of this program.”
How long will it take to get the tests once I order them?
Tests are expected to ship 7 to 12 days after you order them.
But White House officials say that the time frame will likely shorten as the program gains steam.
How many can I order?
There’s a limit of four tests per residential mailing address.
For larger families, White House officials suggest trying other free testing options, like visiting COVID-19 testing sites or your local health center.
Is this a one-time opportunity?
The White House doesn’t say, but officials did mention that if you run out of your four free tests, there are many other ways to access free at-home tests, such as COVID-19 testing sites, pharmacies, and community health centers.
The free tests available through COVIDtests.gov are in addition to an estimated 375 million at-home rapid tests on the market in the U.S. this month.
When should people use a rapid at-home test?
The CDC and experts with other public health groups agree that Americans should consider using at-home rapid tests in the following situations:
- If they begin to have symptoms consistent with COVID-19;
- At least 5 days after close contact with someone who has COVID;
- If someone is indoors with a group of people who are at risk of severe disease or are unvaccinated.
Are at-home rapid tests accurate?
The U.S. Department of Health and Human Services and other federal officials confirmed through studies that all tests distributed through this program can detect the Omicron variant. These agencies also confirmed that their performance is consistent with the FDA’s emergency use authorization.
Is the website designed to handle high demand?
After the original website to sign up for health insurance under the Affordable Care Act crashed repeatedly at launch, the government says it has prepared for high demand for ordering at-home rapid tests.
The U.S. Digital Service (USDS), an organization founded after Healthcare.gov, has partnered with the Postal Service to plan for the launch.
The Postal Service has expanded its staffing, similar to what’s done during the holidays.
All orders in the continental United States will be shipped through first-class mail, with shipments to Alaska, Hawaii, U.S. territories, and military and overseas addresses sent through priority mail.
A version of this article first appeared on WebMD.com.
Antibiotics used in newborns despite low risk for sepsis
Antibiotics were administered to newborns at low risk for early-onset sepsis as frequently as to newborns with EOS risk factors, based on data from approximately 7,500 infants.
EOS remains a significant cause of morbidity and mortality, and predicting which newborns are at risk remains a challenge for neonatal care that often drives high rates of antibiotic use, Dustin D. Flannery, DO, of Children’s Hospital of Philadelphia and colleagues wrote.
Antibiotic exposures are associated with short- and long-term adverse effects in both preterm and term infants, which highlights the need for improved risk assessment in this population, the researchers said.
“A robust estimate of EOS risk in relation to delivery characteristics among infants of all gestational ages at birth could significantly contribute to newborn clinical management by identifying newborns unlikely to benefit from empirical antibiotic therapy,” they emphasized.
In a study published in Pediatrics, the researchers identified 7,540 infants born between Jan. 1, 2009, and Dec. 31, 2014, at two high-risk perinatal units in Philadelphia. Gestational age ranged from 22 to 43 weeks. Criteria for low risk of EOS were determined via an algorithm that included cesarean delivery (with or without labor or membrane rupture), and no antepartum concerns for intra-amniotic infection or nonreassuring fetal status.
A total of 6,428 infants did not meet the low-risk criteria; another 1,121 infants met the low-risk criteria. The primary outcome of EOS was defined as growth of a pathogen in at least 1 blood and/or cerebrospinal fluid culture obtained at 72 hours or less after birth. Overall, 41 infants who did not meet the low-risk criteria developed EOS; none of the infants who met the low-risk criteria developed EOS. Secondary outcomes included initiation of empirical antibiotics at 72 hours or less after birth and the duration of antibiotic use.
Although fewer low-risk infants received antibiotics, compared with infants with EOS (80.4% vs. 91.0%, P < .001), the duration of antibiotic use was not significantly different between the groups, with an adjusted difference of 0.6 hours.
Among infants who did not meet low-risk criteria, 157 were started on antibiotics for each case of EOS, the researchers noted in their discussion of the findings. “Because no cases of EOS were identified in the low-risk group, this proportion could not be calculated but suggests that antibiotic exposure in this group was disproportionately higher for incidence of EOS.”
The study findings were limited by several factors including the possible lack of generalizability to other centers and the use of data from a period before more refined EOS strategies, the researchers noted. Other limitations include the inability to assess the effect of lab results on antibiotic use, a lack of data on the exact indication for delivery, and potential misclassification bias.
Risk assessment tools should not be used alone, but should be used to inform clinical decision-making, the researchers emphasized. However, the results were strengthened by the inclusion of moderately preterm infants, who are rarely studied, and the clinical utility of the risk algorithm used in the study. “The implications of our study include potential adjustments to sepsis risk assessment in term infants, and confirmation and enhancement of previous studies that identify a subset of lower-risk preterm infants,” who may be spared empirical or prolonged antibiotic exposure, they concluded.
Data inform intelligent antibiotic use
“Early-onset sepsis is predominantly caused by exposure of the fetus or neonate to ascending maternal colonization or infection by gastrointestinal or genitourinary bacteria,” Iris Krishna, MD, of Emory University, Atlanta, said in an interview. “Scenarios where there is limited neonatal exposure to these organisms would decrease the risk of development of EOS, therefore it is not surprising that delivery characteristics of low-risk deliveries as defined by investigators – the absence of labor, absence of intra-amniotic infection, rupture of membranes at time of delivery, and cesarean delivery – would have resulted in decreased likelihood of EOS.”
Inappropriate antibiotic use contributes to the development of resistant and more virulent strains of bacteria. A growing body of literature also suggests that early antibiotic usage in newborns may affect the neonatal gut microbiome, which is important for development of the neonatal immune system. Early alterations of the microbiome may have long-term implications,” Dr. Krishna said.
“Understanding the delivery characteristics that increase the risk of EOS are crucial to optimizing the use of antibiotics and thereby minimize potential harm to newborns,” she said. “Studies such as the current study are needed develop EOS prediction tools to improve antibiotic utilization.” More research is needed not only to adequately predict EOS, but to explore how antibiotics affect the neonatal microbiome, and how clinicians can circumvent potential adverse implications with antibiotic use to improve long-term health, Dr. Krishna concluded.
The study received no outside funding. The researchers had no financial conflicts to disclose. Dr. Krishna had no financial conflicts to disclose and serves on the editorial advisory board of Ob.Gyn. News.
Antibiotics were administered to newborns at low risk for early-onset sepsis as frequently as to newborns with EOS risk factors, based on data from approximately 7,500 infants.
EOS remains a significant cause of morbidity and mortality, and predicting which newborns are at risk remains a challenge for neonatal care that often drives high rates of antibiotic use, Dustin D. Flannery, DO, of Children’s Hospital of Philadelphia and colleagues wrote.
Antibiotic exposures are associated with short- and long-term adverse effects in both preterm and term infants, which highlights the need for improved risk assessment in this population, the researchers said.
“A robust estimate of EOS risk in relation to delivery characteristics among infants of all gestational ages at birth could significantly contribute to newborn clinical management by identifying newborns unlikely to benefit from empirical antibiotic therapy,” they emphasized.
In a study published in Pediatrics, the researchers identified 7,540 infants born between Jan. 1, 2009, and Dec. 31, 2014, at two high-risk perinatal units in Philadelphia. Gestational age ranged from 22 to 43 weeks. Criteria for low risk of EOS were determined via an algorithm that included cesarean delivery (with or without labor or membrane rupture), and no antepartum concerns for intra-amniotic infection or nonreassuring fetal status.
A total of 6,428 infants did not meet the low-risk criteria; another 1,121 infants met the low-risk criteria. The primary outcome of EOS was defined as growth of a pathogen in at least 1 blood and/or cerebrospinal fluid culture obtained at 72 hours or less after birth. Overall, 41 infants who did not meet the low-risk criteria developed EOS; none of the infants who met the low-risk criteria developed EOS. Secondary outcomes included initiation of empirical antibiotics at 72 hours or less after birth and the duration of antibiotic use.
Although fewer low-risk infants received antibiotics, compared with infants with EOS (80.4% vs. 91.0%, P < .001), the duration of antibiotic use was not significantly different between the groups, with an adjusted difference of 0.6 hours.
Among infants who did not meet low-risk criteria, 157 were started on antibiotics for each case of EOS, the researchers noted in their discussion of the findings. “Because no cases of EOS were identified in the low-risk group, this proportion could not be calculated but suggests that antibiotic exposure in this group was disproportionately higher for incidence of EOS.”
The study findings were limited by several factors including the possible lack of generalizability to other centers and the use of data from a period before more refined EOS strategies, the researchers noted. Other limitations include the inability to assess the effect of lab results on antibiotic use, a lack of data on the exact indication for delivery, and potential misclassification bias.
Risk assessment tools should not be used alone, but should be used to inform clinical decision-making, the researchers emphasized. However, the results were strengthened by the inclusion of moderately preterm infants, who are rarely studied, and the clinical utility of the risk algorithm used in the study. “The implications of our study include potential adjustments to sepsis risk assessment in term infants, and confirmation and enhancement of previous studies that identify a subset of lower-risk preterm infants,” who may be spared empirical or prolonged antibiotic exposure, they concluded.
Data inform intelligent antibiotic use
“Early-onset sepsis is predominantly caused by exposure of the fetus or neonate to ascending maternal colonization or infection by gastrointestinal or genitourinary bacteria,” Iris Krishna, MD, of Emory University, Atlanta, said in an interview. “Scenarios where there is limited neonatal exposure to these organisms would decrease the risk of development of EOS, therefore it is not surprising that delivery characteristics of low-risk deliveries as defined by investigators – the absence of labor, absence of intra-amniotic infection, rupture of membranes at time of delivery, and cesarean delivery – would have resulted in decreased likelihood of EOS.”
Inappropriate antibiotic use contributes to the development of resistant and more virulent strains of bacteria. A growing body of literature also suggests that early antibiotic usage in newborns may affect the neonatal gut microbiome, which is important for development of the neonatal immune system. Early alterations of the microbiome may have long-term implications,” Dr. Krishna said.
“Understanding the delivery characteristics that increase the risk of EOS are crucial to optimizing the use of antibiotics and thereby minimize potential harm to newborns,” she said. “Studies such as the current study are needed develop EOS prediction tools to improve antibiotic utilization.” More research is needed not only to adequately predict EOS, but to explore how antibiotics affect the neonatal microbiome, and how clinicians can circumvent potential adverse implications with antibiotic use to improve long-term health, Dr. Krishna concluded.
The study received no outside funding. The researchers had no financial conflicts to disclose. Dr. Krishna had no financial conflicts to disclose and serves on the editorial advisory board of Ob.Gyn. News.
Antibiotics were administered to newborns at low risk for early-onset sepsis as frequently as to newborns with EOS risk factors, based on data from approximately 7,500 infants.
EOS remains a significant cause of morbidity and mortality, and predicting which newborns are at risk remains a challenge for neonatal care that often drives high rates of antibiotic use, Dustin D. Flannery, DO, of Children’s Hospital of Philadelphia and colleagues wrote.
Antibiotic exposures are associated with short- and long-term adverse effects in both preterm and term infants, which highlights the need for improved risk assessment in this population, the researchers said.
“A robust estimate of EOS risk in relation to delivery characteristics among infants of all gestational ages at birth could significantly contribute to newborn clinical management by identifying newborns unlikely to benefit from empirical antibiotic therapy,” they emphasized.
In a study published in Pediatrics, the researchers identified 7,540 infants born between Jan. 1, 2009, and Dec. 31, 2014, at two high-risk perinatal units in Philadelphia. Gestational age ranged from 22 to 43 weeks. Criteria for low risk of EOS were determined via an algorithm that included cesarean delivery (with or without labor or membrane rupture), and no antepartum concerns for intra-amniotic infection or nonreassuring fetal status.
A total of 6,428 infants did not meet the low-risk criteria; another 1,121 infants met the low-risk criteria. The primary outcome of EOS was defined as growth of a pathogen in at least 1 blood and/or cerebrospinal fluid culture obtained at 72 hours or less after birth. Overall, 41 infants who did not meet the low-risk criteria developed EOS; none of the infants who met the low-risk criteria developed EOS. Secondary outcomes included initiation of empirical antibiotics at 72 hours or less after birth and the duration of antibiotic use.
Although fewer low-risk infants received antibiotics, compared with infants with EOS (80.4% vs. 91.0%, P < .001), the duration of antibiotic use was not significantly different between the groups, with an adjusted difference of 0.6 hours.
Among infants who did not meet low-risk criteria, 157 were started on antibiotics for each case of EOS, the researchers noted in their discussion of the findings. “Because no cases of EOS were identified in the low-risk group, this proportion could not be calculated but suggests that antibiotic exposure in this group was disproportionately higher for incidence of EOS.”
The study findings were limited by several factors including the possible lack of generalizability to other centers and the use of data from a period before more refined EOS strategies, the researchers noted. Other limitations include the inability to assess the effect of lab results on antibiotic use, a lack of data on the exact indication for delivery, and potential misclassification bias.
Risk assessment tools should not be used alone, but should be used to inform clinical decision-making, the researchers emphasized. However, the results were strengthened by the inclusion of moderately preterm infants, who are rarely studied, and the clinical utility of the risk algorithm used in the study. “The implications of our study include potential adjustments to sepsis risk assessment in term infants, and confirmation and enhancement of previous studies that identify a subset of lower-risk preterm infants,” who may be spared empirical or prolonged antibiotic exposure, they concluded.
Data inform intelligent antibiotic use
“Early-onset sepsis is predominantly caused by exposure of the fetus or neonate to ascending maternal colonization or infection by gastrointestinal or genitourinary bacteria,” Iris Krishna, MD, of Emory University, Atlanta, said in an interview. “Scenarios where there is limited neonatal exposure to these organisms would decrease the risk of development of EOS, therefore it is not surprising that delivery characteristics of low-risk deliveries as defined by investigators – the absence of labor, absence of intra-amniotic infection, rupture of membranes at time of delivery, and cesarean delivery – would have resulted in decreased likelihood of EOS.”
Inappropriate antibiotic use contributes to the development of resistant and more virulent strains of bacteria. A growing body of literature also suggests that early antibiotic usage in newborns may affect the neonatal gut microbiome, which is important for development of the neonatal immune system. Early alterations of the microbiome may have long-term implications,” Dr. Krishna said.
“Understanding the delivery characteristics that increase the risk of EOS are crucial to optimizing the use of antibiotics and thereby minimize potential harm to newborns,” she said. “Studies such as the current study are needed develop EOS prediction tools to improve antibiotic utilization.” More research is needed not only to adequately predict EOS, but to explore how antibiotics affect the neonatal microbiome, and how clinicians can circumvent potential adverse implications with antibiotic use to improve long-term health, Dr. Krishna concluded.
The study received no outside funding. The researchers had no financial conflicts to disclose. Dr. Krishna had no financial conflicts to disclose and serves on the editorial advisory board of Ob.Gyn. News.
FROM PEDIATRICS

