User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
nav[contains(@class, 'nav-ce-stack nav-ce-stack__large-screen')]
header[@id='header']
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'main-prefix')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
div[contains(@class, 'view-medstat-quiz-listing-panes')]
div[contains(@class, 'pane-article-sidebar-latest-news')]
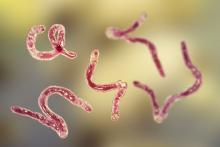
Helpful giant rodents and our old friend, the hookworm
Rat-ting out coronavirus
Did you know there is a possibility that giant rodents could rat out coronavirus? Not many people are keen on the presence of a 3-foot-long African giant pouched rat, but they have already been trained to sniff out diseases that are dangerous to humans, such as brucellosis and tuberculosis, according to researchers at the University of Glasgow.
Professor Dan Haydon and his associates believe there is a good possibility that the rats can be trained to sniff out COVID-19. Dogs have been helpful in sniffing for COVID-19 at airports and are being trained to detect it through armpit sweat, making detection of the virus easier for travelers and staff. Even robots have gotten into the COVID-19 detecting act.
Since African giant pouched rats can grow to be the size of a small dog and “are easily tamed as companion animals,” it seems likely that they have the potential to do the same. That is, sniffing for COVID-19, not appearing at your local airport. That’s still gross.
Stay healthy, get a parasite bestie
The key to health could actually be swimming around in your gut. Researchers from University College London have found that a parasitic worm could be the answer to longevity and avoidance of chronic diseases.
The seeming immunity from inflammatory diseases such as arthritis, diabetes, and multiple sclerosis may come from helminth parasites, or hookworms – parasites that have been coexisting harmlessly with the human body for thousands of years. The investigators went so far as to call them “old friends,” but the kind that you rarely see at reunions or call up for a favor.
As a result of modern sanitation and improved hygiene, humans and hookworms are seeing much less of each other, which may be a factor in the rise of “aging-associated inflammation” such as COVID-19 symptoms, they suggested. So is there a way to get these old friends back?
“Restorative hookworm treatments” could help with heart disease or dementia, according to the investigators, but maybe you’re not totally on board with getting an actual parasite in your system. We get it. There are helminth-derived proteins that have already been tested to get the job done.
Maybe old friends really do make the best friends.
I love the smell of microbe-infected aerosols in the morning
Have you gone into a public restroom and just stood around for a while appreciating the fine aromas? No? You haven’t? You do your business and get out? Well, it’s a good thing you act like a normal person, because the aerosols released when toilets flush can contain all sorts of nasty bacteria and viruses.
The authors of a new study published in Physics of Fluids came to this groundbreaking conclusion by going to a public bathroom, sticking particle counters above a urinal and a toilet, and letting them sit for a while. After 3 hours and 100 flushes, the ambient level of particles 0.3-3 mcm in diameter had increased dramatically, with particles sized 0.5-1 mcm particularly prone to lingering. For those particles, the level from baseline increased by over 200%.
This is a major concern, the researchers said, because the sort of microbes that are expelled through feces, urine, and vomit can include some pretty nasty things. Ebola, noroviruses that can cause food poisoning, and even good old SARS-CoV-2 can be expelled from the body but remain viable for a time in these aerosols. The researchers recommended improving the ventilation systems in restrooms so that aerosols don’t hang around for hours at a time. Plus, it might make the place not smell like a, uh, public restroom.
Not to question the research and the people behind it, but we’re not sure how necessary it was to give people another reason not to hang out in a place where hundreds, if not thousands, of people come to relieve themselves. There’s a reason we’re supposed to “stop and smell the roses” and not “stop and smell the public bathroom.”
World ends not with a bang but with a cheeseburger
Speaking of old sayings, one of our favorites, “You are what you eat,” may offer a culinary explanation for those who do enjoy the ambiance of a fine, aerosol-infested public restroom.
That explanation involves the high-calorie, high-fat smorgasbord known as the Western diet and some mice who were forced to consume it. Those mice, it turns out, were more anxious and less cognitively advanced than their counterparts who were not eating “highly palatable, energy dense foods (e.g., high saturated fat, high sugar) that are commonly consumed by humans,” according to the authors of a recent literature review.
“Consumption of a Western diet is related to poorer cognitive performance across the lifespan,” the investigators said, adding that consumption of a Western diet “during critical early life stages of development has negative consequences on various cognitive abilities later in adulthood.”
To show their appreciation for the sacrifices these brave test subjects had made in the name of science, the scientists released the Western diet–addled rodents from captivity. Not only did they survive and thrive in the wilds of darkest suburbia, but within 6 months almost half of them were running for Congress.
Rat-ting out coronavirus
Did you know there is a possibility that giant rodents could rat out coronavirus? Not many people are keen on the presence of a 3-foot-long African giant pouched rat, but they have already been trained to sniff out diseases that are dangerous to humans, such as brucellosis and tuberculosis, according to researchers at the University of Glasgow.
Professor Dan Haydon and his associates believe there is a good possibility that the rats can be trained to sniff out COVID-19. Dogs have been helpful in sniffing for COVID-19 at airports and are being trained to detect it through armpit sweat, making detection of the virus easier for travelers and staff. Even robots have gotten into the COVID-19 detecting act.
Since African giant pouched rats can grow to be the size of a small dog and “are easily tamed as companion animals,” it seems likely that they have the potential to do the same. That is, sniffing for COVID-19, not appearing at your local airport. That’s still gross.
Stay healthy, get a parasite bestie
The key to health could actually be swimming around in your gut. Researchers from University College London have found that a parasitic worm could be the answer to longevity and avoidance of chronic diseases.
The seeming immunity from inflammatory diseases such as arthritis, diabetes, and multiple sclerosis may come from helminth parasites, or hookworms – parasites that have been coexisting harmlessly with the human body for thousands of years. The investigators went so far as to call them “old friends,” but the kind that you rarely see at reunions or call up for a favor.
As a result of modern sanitation and improved hygiene, humans and hookworms are seeing much less of each other, which may be a factor in the rise of “aging-associated inflammation” such as COVID-19 symptoms, they suggested. So is there a way to get these old friends back?
“Restorative hookworm treatments” could help with heart disease or dementia, according to the investigators, but maybe you’re not totally on board with getting an actual parasite in your system. We get it. There are helminth-derived proteins that have already been tested to get the job done.
Maybe old friends really do make the best friends.
I love the smell of microbe-infected aerosols in the morning
Have you gone into a public restroom and just stood around for a while appreciating the fine aromas? No? You haven’t? You do your business and get out? Well, it’s a good thing you act like a normal person, because the aerosols released when toilets flush can contain all sorts of nasty bacteria and viruses.
The authors of a new study published in Physics of Fluids came to this groundbreaking conclusion by going to a public bathroom, sticking particle counters above a urinal and a toilet, and letting them sit for a while. After 3 hours and 100 flushes, the ambient level of particles 0.3-3 mcm in diameter had increased dramatically, with particles sized 0.5-1 mcm particularly prone to lingering. For those particles, the level from baseline increased by over 200%.
This is a major concern, the researchers said, because the sort of microbes that are expelled through feces, urine, and vomit can include some pretty nasty things. Ebola, noroviruses that can cause food poisoning, and even good old SARS-CoV-2 can be expelled from the body but remain viable for a time in these aerosols. The researchers recommended improving the ventilation systems in restrooms so that aerosols don’t hang around for hours at a time. Plus, it might make the place not smell like a, uh, public restroom.
Not to question the research and the people behind it, but we’re not sure how necessary it was to give people another reason not to hang out in a place where hundreds, if not thousands, of people come to relieve themselves. There’s a reason we’re supposed to “stop and smell the roses” and not “stop and smell the public bathroom.”
World ends not with a bang but with a cheeseburger
Speaking of old sayings, one of our favorites, “You are what you eat,” may offer a culinary explanation for those who do enjoy the ambiance of a fine, aerosol-infested public restroom.
That explanation involves the high-calorie, high-fat smorgasbord known as the Western diet and some mice who were forced to consume it. Those mice, it turns out, were more anxious and less cognitively advanced than their counterparts who were not eating “highly palatable, energy dense foods (e.g., high saturated fat, high sugar) that are commonly consumed by humans,” according to the authors of a recent literature review.
“Consumption of a Western diet is related to poorer cognitive performance across the lifespan,” the investigators said, adding that consumption of a Western diet “during critical early life stages of development has negative consequences on various cognitive abilities later in adulthood.”
To show their appreciation for the sacrifices these brave test subjects had made in the name of science, the scientists released the Western diet–addled rodents from captivity. Not only did they survive and thrive in the wilds of darkest suburbia, but within 6 months almost half of them were running for Congress.
Rat-ting out coronavirus
Did you know there is a possibility that giant rodents could rat out coronavirus? Not many people are keen on the presence of a 3-foot-long African giant pouched rat, but they have already been trained to sniff out diseases that are dangerous to humans, such as brucellosis and tuberculosis, according to researchers at the University of Glasgow.
Professor Dan Haydon and his associates believe there is a good possibility that the rats can be trained to sniff out COVID-19. Dogs have been helpful in sniffing for COVID-19 at airports and are being trained to detect it through armpit sweat, making detection of the virus easier for travelers and staff. Even robots have gotten into the COVID-19 detecting act.
Since African giant pouched rats can grow to be the size of a small dog and “are easily tamed as companion animals,” it seems likely that they have the potential to do the same. That is, sniffing for COVID-19, not appearing at your local airport. That’s still gross.
Stay healthy, get a parasite bestie
The key to health could actually be swimming around in your gut. Researchers from University College London have found that a parasitic worm could be the answer to longevity and avoidance of chronic diseases.
The seeming immunity from inflammatory diseases such as arthritis, diabetes, and multiple sclerosis may come from helminth parasites, or hookworms – parasites that have been coexisting harmlessly with the human body for thousands of years. The investigators went so far as to call them “old friends,” but the kind that you rarely see at reunions or call up for a favor.
As a result of modern sanitation and improved hygiene, humans and hookworms are seeing much less of each other, which may be a factor in the rise of “aging-associated inflammation” such as COVID-19 symptoms, they suggested. So is there a way to get these old friends back?
“Restorative hookworm treatments” could help with heart disease or dementia, according to the investigators, but maybe you’re not totally on board with getting an actual parasite in your system. We get it. There are helminth-derived proteins that have already been tested to get the job done.
Maybe old friends really do make the best friends.
I love the smell of microbe-infected aerosols in the morning
Have you gone into a public restroom and just stood around for a while appreciating the fine aromas? No? You haven’t? You do your business and get out? Well, it’s a good thing you act like a normal person, because the aerosols released when toilets flush can contain all sorts of nasty bacteria and viruses.
The authors of a new study published in Physics of Fluids came to this groundbreaking conclusion by going to a public bathroom, sticking particle counters above a urinal and a toilet, and letting them sit for a while. After 3 hours and 100 flushes, the ambient level of particles 0.3-3 mcm in diameter had increased dramatically, with particles sized 0.5-1 mcm particularly prone to lingering. For those particles, the level from baseline increased by over 200%.
This is a major concern, the researchers said, because the sort of microbes that are expelled through feces, urine, and vomit can include some pretty nasty things. Ebola, noroviruses that can cause food poisoning, and even good old SARS-CoV-2 can be expelled from the body but remain viable for a time in these aerosols. The researchers recommended improving the ventilation systems in restrooms so that aerosols don’t hang around for hours at a time. Plus, it might make the place not smell like a, uh, public restroom.
Not to question the research and the people behind it, but we’re not sure how necessary it was to give people another reason not to hang out in a place where hundreds, if not thousands, of people come to relieve themselves. There’s a reason we’re supposed to “stop and smell the roses” and not “stop and smell the public bathroom.”
World ends not with a bang but with a cheeseburger
Speaking of old sayings, one of our favorites, “You are what you eat,” may offer a culinary explanation for those who do enjoy the ambiance of a fine, aerosol-infested public restroom.
That explanation involves the high-calorie, high-fat smorgasbord known as the Western diet and some mice who were forced to consume it. Those mice, it turns out, were more anxious and less cognitively advanced than their counterparts who were not eating “highly palatable, energy dense foods (e.g., high saturated fat, high sugar) that are commonly consumed by humans,” according to the authors of a recent literature review.
“Consumption of a Western diet is related to poorer cognitive performance across the lifespan,” the investigators said, adding that consumption of a Western diet “during critical early life stages of development has negative consequences on various cognitive abilities later in adulthood.”
To show their appreciation for the sacrifices these brave test subjects had made in the name of science, the scientists released the Western diet–addled rodents from captivity. Not only did they survive and thrive in the wilds of darkest suburbia, but within 6 months almost half of them were running for Congress.
Gene therapy shows promise for Sanfilippo syndrome
. Most of the benefit from the treatment came in patients who began treatment at younger age, but comparisons to natural history controls showed profound improvement among many recipients, some of whom attained normal developmental trajectories.
The study was presented at the American Academy of Neurology’s 2021 annual meeting by Kevin Flanigan, MD, an attending neurologist at Nationwide Children’s Hospital in Columbus, Ohio. He highlighted the improved developmental outcomes. “There’s been nothing shown to change the cognitive pathway of the disease. This is the first time it’s been seen as a treatment effect,” Dr. Flanigan said during a follow-up Q&A session.
The therapy was delivered using an adeno-associated virus-9 (AAV-9) vector, which led one questioner to ask about potential safety concerns, since AAV-associated risks date back to the death of Jesse Gelsinger in 1999. “There is concern about AAV therapies related to immune responses to potentially complement-mediated activation and thrombocytopenic syndrome, which has led to clinical holds on some other AAV-9 products related to muscular dystrophies. We’ve not seen signals of anything reminiscent of that, and we’re at AAV-9 dosages that are quite similar to what’s been used elsewhere in the field,” said Dr. Flanigan.
The results have him optimistic about the therapy. “I do think if it continues to be increasing divergent from the natural history, it will be questionable as to whether a subsequent trial will be necessary for this. That’s a decision for the [Food and Drug Administration] and the company to decide. Each observation point that goes by, each patient treated, and each time we get more data, I get more and more confident. It’s really gratifying to watch,” said Dr. Flanigan.
The study confirms the potential of gene replacement therapy autosomal recessive conditions, according to Nicholas Johnson, MD, associate professor of neurology at Virginia Commonwealth University, Richmond, as well as a fellow of the American Academy of Neurology. “Where the genetic problem is loss of gene function, the ability to replace that gene using a viral approach is going to be transformative across the board for many of these different conditions, including Sanfilippo syndrome,” said Dr. Johnson, who attended the session but was not involved in the research.
Toxicity could remain an issue, even in the absence of AAV-based safety concerns. “The rate limiting step in terms of gene replacement therapy development likely relates to the ability to provide those therapies to larger adults, because many approaches are weight based and it’s unclear what the upper limit of toxicity would be for adults,” said Dr. Johnson.
Transpher A study results
Dr. Flanigan presented results from Transpher A, a phase 1/2 clinical trial that has enrolled 20 patients to date in three cohorts: Cohort 1, with 3 patients, received 5 x 1,012 vg/kg, and had a mean follow-up of 58 months; cohort 2, with 3 patients, received 1 x 1,013 vg/kg, and had a mean follow-up of 49 months; and cohort 3, with 14 patients, received 3 x 1,013 vg/kg, with a mean follow-up of 24 months. Included patients ranged from birth to age 2, or older than age 2 with a development quotient of 60 or higher on the Bayley Scale.
Dr. Flanigan showed a plot of developmental progress compared with natural history controls, which showed that patients treated before age 2 or with a developmental quotient of 60 or higher had improved outcomes compared to other patients in the high dose cohort. They continued to show normal developmental progression at 30-36 months post treatment, at a time when the natural history data suggested they would suffer cognitive decline. Two years after administration, this group had cerebral spinal fluid levels of heparan sulfate that fell below the lower limit of detection. Patients in the high-dose cohort had normalized CSF levels of GM2 and GM3 gangliosides, and there were reductions in plasma heparan sulfate and urinary glycosaminoglycans. There was also a sustained decrease in liver volume.
The highest dose group was originally given to older patients, and most were similar to the natural history cohort, though some did stabilize. “More compellingly, patients (in the high-dose group) who were treated younger actually showed continued increase in development. One individual follows the normal development quotient line, and we would say that these are really quite distinct from what we typically see in patients,” said Dr. Flanigan.
The treatment was well tolerated. There were no deaths or treatment-related serious adverse events, and no clinically-significant adverse events within the first 5 years of follow-up.
The study was funded by Abeona Therapeutics. Dr. Flanigan has been on advisory boards for Apic Bio and 4D Molecular Therapeutics, consulted for Encoded Therapeutics, and has received royalties from Audentes Therapeutics. Dr. Flanigan has received funding from and been a consultant for Avidity.
. Most of the benefit from the treatment came in patients who began treatment at younger age, but comparisons to natural history controls showed profound improvement among many recipients, some of whom attained normal developmental trajectories.
The study was presented at the American Academy of Neurology’s 2021 annual meeting by Kevin Flanigan, MD, an attending neurologist at Nationwide Children’s Hospital in Columbus, Ohio. He highlighted the improved developmental outcomes. “There’s been nothing shown to change the cognitive pathway of the disease. This is the first time it’s been seen as a treatment effect,” Dr. Flanigan said during a follow-up Q&A session.
The therapy was delivered using an adeno-associated virus-9 (AAV-9) vector, which led one questioner to ask about potential safety concerns, since AAV-associated risks date back to the death of Jesse Gelsinger in 1999. “There is concern about AAV therapies related to immune responses to potentially complement-mediated activation and thrombocytopenic syndrome, which has led to clinical holds on some other AAV-9 products related to muscular dystrophies. We’ve not seen signals of anything reminiscent of that, and we’re at AAV-9 dosages that are quite similar to what’s been used elsewhere in the field,” said Dr. Flanigan.
The results have him optimistic about the therapy. “I do think if it continues to be increasing divergent from the natural history, it will be questionable as to whether a subsequent trial will be necessary for this. That’s a decision for the [Food and Drug Administration] and the company to decide. Each observation point that goes by, each patient treated, and each time we get more data, I get more and more confident. It’s really gratifying to watch,” said Dr. Flanigan.
The study confirms the potential of gene replacement therapy autosomal recessive conditions, according to Nicholas Johnson, MD, associate professor of neurology at Virginia Commonwealth University, Richmond, as well as a fellow of the American Academy of Neurology. “Where the genetic problem is loss of gene function, the ability to replace that gene using a viral approach is going to be transformative across the board for many of these different conditions, including Sanfilippo syndrome,” said Dr. Johnson, who attended the session but was not involved in the research.
Toxicity could remain an issue, even in the absence of AAV-based safety concerns. “The rate limiting step in terms of gene replacement therapy development likely relates to the ability to provide those therapies to larger adults, because many approaches are weight based and it’s unclear what the upper limit of toxicity would be for adults,” said Dr. Johnson.
Transpher A study results
Dr. Flanigan presented results from Transpher A, a phase 1/2 clinical trial that has enrolled 20 patients to date in three cohorts: Cohort 1, with 3 patients, received 5 x 1,012 vg/kg, and had a mean follow-up of 58 months; cohort 2, with 3 patients, received 1 x 1,013 vg/kg, and had a mean follow-up of 49 months; and cohort 3, with 14 patients, received 3 x 1,013 vg/kg, with a mean follow-up of 24 months. Included patients ranged from birth to age 2, or older than age 2 with a development quotient of 60 or higher on the Bayley Scale.
Dr. Flanigan showed a plot of developmental progress compared with natural history controls, which showed that patients treated before age 2 or with a developmental quotient of 60 or higher had improved outcomes compared to other patients in the high dose cohort. They continued to show normal developmental progression at 30-36 months post treatment, at a time when the natural history data suggested they would suffer cognitive decline. Two years after administration, this group had cerebral spinal fluid levels of heparan sulfate that fell below the lower limit of detection. Patients in the high-dose cohort had normalized CSF levels of GM2 and GM3 gangliosides, and there were reductions in plasma heparan sulfate and urinary glycosaminoglycans. There was also a sustained decrease in liver volume.
The highest dose group was originally given to older patients, and most were similar to the natural history cohort, though some did stabilize. “More compellingly, patients (in the high-dose group) who were treated younger actually showed continued increase in development. One individual follows the normal development quotient line, and we would say that these are really quite distinct from what we typically see in patients,” said Dr. Flanigan.
The treatment was well tolerated. There were no deaths or treatment-related serious adverse events, and no clinically-significant adverse events within the first 5 years of follow-up.
The study was funded by Abeona Therapeutics. Dr. Flanigan has been on advisory boards for Apic Bio and 4D Molecular Therapeutics, consulted for Encoded Therapeutics, and has received royalties from Audentes Therapeutics. Dr. Flanigan has received funding from and been a consultant for Avidity.
. Most of the benefit from the treatment came in patients who began treatment at younger age, but comparisons to natural history controls showed profound improvement among many recipients, some of whom attained normal developmental trajectories.
The study was presented at the American Academy of Neurology’s 2021 annual meeting by Kevin Flanigan, MD, an attending neurologist at Nationwide Children’s Hospital in Columbus, Ohio. He highlighted the improved developmental outcomes. “There’s been nothing shown to change the cognitive pathway of the disease. This is the first time it’s been seen as a treatment effect,” Dr. Flanigan said during a follow-up Q&A session.
The therapy was delivered using an adeno-associated virus-9 (AAV-9) vector, which led one questioner to ask about potential safety concerns, since AAV-associated risks date back to the death of Jesse Gelsinger in 1999. “There is concern about AAV therapies related to immune responses to potentially complement-mediated activation and thrombocytopenic syndrome, which has led to clinical holds on some other AAV-9 products related to muscular dystrophies. We’ve not seen signals of anything reminiscent of that, and we’re at AAV-9 dosages that are quite similar to what’s been used elsewhere in the field,” said Dr. Flanigan.
The results have him optimistic about the therapy. “I do think if it continues to be increasing divergent from the natural history, it will be questionable as to whether a subsequent trial will be necessary for this. That’s a decision for the [Food and Drug Administration] and the company to decide. Each observation point that goes by, each patient treated, and each time we get more data, I get more and more confident. It’s really gratifying to watch,” said Dr. Flanigan.
The study confirms the potential of gene replacement therapy autosomal recessive conditions, according to Nicholas Johnson, MD, associate professor of neurology at Virginia Commonwealth University, Richmond, as well as a fellow of the American Academy of Neurology. “Where the genetic problem is loss of gene function, the ability to replace that gene using a viral approach is going to be transformative across the board for many of these different conditions, including Sanfilippo syndrome,” said Dr. Johnson, who attended the session but was not involved in the research.
Toxicity could remain an issue, even in the absence of AAV-based safety concerns. “The rate limiting step in terms of gene replacement therapy development likely relates to the ability to provide those therapies to larger adults, because many approaches are weight based and it’s unclear what the upper limit of toxicity would be for adults,” said Dr. Johnson.
Transpher A study results
Dr. Flanigan presented results from Transpher A, a phase 1/2 clinical trial that has enrolled 20 patients to date in three cohorts: Cohort 1, with 3 patients, received 5 x 1,012 vg/kg, and had a mean follow-up of 58 months; cohort 2, with 3 patients, received 1 x 1,013 vg/kg, and had a mean follow-up of 49 months; and cohort 3, with 14 patients, received 3 x 1,013 vg/kg, with a mean follow-up of 24 months. Included patients ranged from birth to age 2, or older than age 2 with a development quotient of 60 or higher on the Bayley Scale.
Dr. Flanigan showed a plot of developmental progress compared with natural history controls, which showed that patients treated before age 2 or with a developmental quotient of 60 or higher had improved outcomes compared to other patients in the high dose cohort. They continued to show normal developmental progression at 30-36 months post treatment, at a time when the natural history data suggested they would suffer cognitive decline. Two years after administration, this group had cerebral spinal fluid levels of heparan sulfate that fell below the lower limit of detection. Patients in the high-dose cohort had normalized CSF levels of GM2 and GM3 gangliosides, and there were reductions in plasma heparan sulfate and urinary glycosaminoglycans. There was also a sustained decrease in liver volume.
The highest dose group was originally given to older patients, and most were similar to the natural history cohort, though some did stabilize. “More compellingly, patients (in the high-dose group) who were treated younger actually showed continued increase in development. One individual follows the normal development quotient line, and we would say that these are really quite distinct from what we typically see in patients,” said Dr. Flanigan.
The treatment was well tolerated. There were no deaths or treatment-related serious adverse events, and no clinically-significant adverse events within the first 5 years of follow-up.
The study was funded by Abeona Therapeutics. Dr. Flanigan has been on advisory boards for Apic Bio and 4D Molecular Therapeutics, consulted for Encoded Therapeutics, and has received royalties from Audentes Therapeutics. Dr. Flanigan has received funding from and been a consultant for Avidity.
FROM AAN 2021
Investigative gepant liver profile comparable with standard of care
according to results of a multicenter, open-label trial presented at the 2021 annual meeting of the American Academy of Neurology.
The trial included 739 patients, 543 of whom were randomized to daily oral atogepant with the remainder assigned to the existing standard of care oral migraine prevention medication, said Messoud Ashina, MD, PhD, of the Danish Headache Center at the University of Copenhagen. Initially, 67% (n = 364) of the atogepant patients reported treatment-emergent adverse events (TEAEs). However, he noted, the rate of TEAEs related to treatment was actually 18% (n = 98), and the rate of serious AEs was 4.4% (n = 24); 31 patients (5.7%) discontinued therapy because of TEAEs.
Those rates compared favorably with the standard of care group, Dr. Ashina said. In that group, the rate of TEAEs was 78.6% (154/196), and the rate of treatment-related TEAEs was 36.2% (n = 71).
In the atogepant group, the most common TEAEs were upper respiratory tract infection (10.3%, n = 56), constipation (7.2%, n = 39) and nausea (6.3%, n = 34). “With constipation in particular most cases were mild to moderate,” Dr. Ashina said. “Only one case was considered severe and it resolved before the end of the trial.” One patient discontinued treatment because of constipation.
Most significantly, said Dr. Ashina, “No hepatic safety issues were identified.” In the atogepant group, 2.4% of patients (n = 13) had ALT/AST levels at three times the upper limit of normal versus 3.2% (n = 6) in the standard of care group.
During question-and-answer, Dr. Ashina was pressed on the rate or urinary tract infections in the atogepant patients – 5.2% (n = 28), a measure not reported in the standard of care group – but he said there was no indication the UTIs resulted from atogepant itself. “I assume if there was some problems with urinary tract infections because of the kidneys then you would expect to see the lab data showing that,” he added in an interview. “There were no differences in lab abnormalities between the two groups.”
While Dr. Ashina said “I don’t think it’s something of concern” with regard to the UTI risk, he added: “It doesn’t mean that we don’t have to be careful. As physicians, we have to exhibit pharmacovigilance all the time, especially with the new drugs coming out over the next 5 years. But don’t panic.”
Stephen D. Silberstein, MD, director of the headache center at Thomas Jefferson University, Philadelphia, agreed that the hepatic values reported in the open-label trial are important. “What’s really nice about this study is the fact that we now know that this particular gepant when given on a regular basis for 1 year has no problem with liver or kidney function,” he said in an interview.
Dr. Ashina said once-daily oral atogepant could potentially be a desirable alternative migraine preventive treatment to monoclonal antibodies and their quarterly injections and a second-line therapy when other treatments don’t work. However, Dr. Silverstein noted that patients typically aren’t as compliant with self-administered oral medications as they are with periodic injections.
Allergan/AbbVie sponsored the trial. Dr. Ashina reported financial relationships with Allergan/AbbVie, Amgen, Eli Lilly, Lundbeck, Novartis, and Teva. Dr. Silberstein is a trial investigator.
according to results of a multicenter, open-label trial presented at the 2021 annual meeting of the American Academy of Neurology.
The trial included 739 patients, 543 of whom were randomized to daily oral atogepant with the remainder assigned to the existing standard of care oral migraine prevention medication, said Messoud Ashina, MD, PhD, of the Danish Headache Center at the University of Copenhagen. Initially, 67% (n = 364) of the atogepant patients reported treatment-emergent adverse events (TEAEs). However, he noted, the rate of TEAEs related to treatment was actually 18% (n = 98), and the rate of serious AEs was 4.4% (n = 24); 31 patients (5.7%) discontinued therapy because of TEAEs.
Those rates compared favorably with the standard of care group, Dr. Ashina said. In that group, the rate of TEAEs was 78.6% (154/196), and the rate of treatment-related TEAEs was 36.2% (n = 71).
In the atogepant group, the most common TEAEs were upper respiratory tract infection (10.3%, n = 56), constipation (7.2%, n = 39) and nausea (6.3%, n = 34). “With constipation in particular most cases were mild to moderate,” Dr. Ashina said. “Only one case was considered severe and it resolved before the end of the trial.” One patient discontinued treatment because of constipation.
Most significantly, said Dr. Ashina, “No hepatic safety issues were identified.” In the atogepant group, 2.4% of patients (n = 13) had ALT/AST levels at three times the upper limit of normal versus 3.2% (n = 6) in the standard of care group.
During question-and-answer, Dr. Ashina was pressed on the rate or urinary tract infections in the atogepant patients – 5.2% (n = 28), a measure not reported in the standard of care group – but he said there was no indication the UTIs resulted from atogepant itself. “I assume if there was some problems with urinary tract infections because of the kidneys then you would expect to see the lab data showing that,” he added in an interview. “There were no differences in lab abnormalities between the two groups.”
While Dr. Ashina said “I don’t think it’s something of concern” with regard to the UTI risk, he added: “It doesn’t mean that we don’t have to be careful. As physicians, we have to exhibit pharmacovigilance all the time, especially with the new drugs coming out over the next 5 years. But don’t panic.”
Stephen D. Silberstein, MD, director of the headache center at Thomas Jefferson University, Philadelphia, agreed that the hepatic values reported in the open-label trial are important. “What’s really nice about this study is the fact that we now know that this particular gepant when given on a regular basis for 1 year has no problem with liver or kidney function,” he said in an interview.
Dr. Ashina said once-daily oral atogepant could potentially be a desirable alternative migraine preventive treatment to monoclonal antibodies and their quarterly injections and a second-line therapy when other treatments don’t work. However, Dr. Silverstein noted that patients typically aren’t as compliant with self-administered oral medications as they are with periodic injections.
Allergan/AbbVie sponsored the trial. Dr. Ashina reported financial relationships with Allergan/AbbVie, Amgen, Eli Lilly, Lundbeck, Novartis, and Teva. Dr. Silberstein is a trial investigator.
according to results of a multicenter, open-label trial presented at the 2021 annual meeting of the American Academy of Neurology.
The trial included 739 patients, 543 of whom were randomized to daily oral atogepant with the remainder assigned to the existing standard of care oral migraine prevention medication, said Messoud Ashina, MD, PhD, of the Danish Headache Center at the University of Copenhagen. Initially, 67% (n = 364) of the atogepant patients reported treatment-emergent adverse events (TEAEs). However, he noted, the rate of TEAEs related to treatment was actually 18% (n = 98), and the rate of serious AEs was 4.4% (n = 24); 31 patients (5.7%) discontinued therapy because of TEAEs.
Those rates compared favorably with the standard of care group, Dr. Ashina said. In that group, the rate of TEAEs was 78.6% (154/196), and the rate of treatment-related TEAEs was 36.2% (n = 71).
In the atogepant group, the most common TEAEs were upper respiratory tract infection (10.3%, n = 56), constipation (7.2%, n = 39) and nausea (6.3%, n = 34). “With constipation in particular most cases were mild to moderate,” Dr. Ashina said. “Only one case was considered severe and it resolved before the end of the trial.” One patient discontinued treatment because of constipation.
Most significantly, said Dr. Ashina, “No hepatic safety issues were identified.” In the atogepant group, 2.4% of patients (n = 13) had ALT/AST levels at three times the upper limit of normal versus 3.2% (n = 6) in the standard of care group.
During question-and-answer, Dr. Ashina was pressed on the rate or urinary tract infections in the atogepant patients – 5.2% (n = 28), a measure not reported in the standard of care group – but he said there was no indication the UTIs resulted from atogepant itself. “I assume if there was some problems with urinary tract infections because of the kidneys then you would expect to see the lab data showing that,” he added in an interview. “There were no differences in lab abnormalities between the two groups.”
While Dr. Ashina said “I don’t think it’s something of concern” with regard to the UTI risk, he added: “It doesn’t mean that we don’t have to be careful. As physicians, we have to exhibit pharmacovigilance all the time, especially with the new drugs coming out over the next 5 years. But don’t panic.”
Stephen D. Silberstein, MD, director of the headache center at Thomas Jefferson University, Philadelphia, agreed that the hepatic values reported in the open-label trial are important. “What’s really nice about this study is the fact that we now know that this particular gepant when given on a regular basis for 1 year has no problem with liver or kidney function,” he said in an interview.
Dr. Ashina said once-daily oral atogepant could potentially be a desirable alternative migraine preventive treatment to monoclonal antibodies and their quarterly injections and a second-line therapy when other treatments don’t work. However, Dr. Silverstein noted that patients typically aren’t as compliant with self-administered oral medications as they are with periodic injections.
Allergan/AbbVie sponsored the trial. Dr. Ashina reported financial relationships with Allergan/AbbVie, Amgen, Eli Lilly, Lundbeck, Novartis, and Teva. Dr. Silberstein is a trial investigator.
FROM AAN 2021
The neurology of long-haul COVID-19
Long-haul neurologic symptoms of COVID-19 seem to be distinct from neurologic conditions found in acute disease. Much work remains to be done to understand the biological mechanisms behind these problems, but inflammation and autoimmune responses may play a role in some cases.
Those were some of the takeaways from a talk by Serena Spudich, MD, who presented her research at the 2021 annual meeting of the American Academy of Neurology. Dr. Spudich is the division chief of neurologic infections and global neurology and codirector of the Center for Neuroepidemiology and Clinical Neurological Research at Yale University, New Haven, Conn.
Examining the nervous system’s involvement in COVID-19
Even early on in the pandemic, it became clear that there were lingering complaints of neuromuscular problems, cognitive dysfunction, and mood and psychiatric issues. Breathing and heart rate problems also can arise. “There seems to be a preponderance of syndromes that reflect involvement of the nervous system,” said Dr. Spudich.
To try to understand the etiology of these persistent problems, Dr. Spudich said it’s important to examine the nervous system’s involvement in acute COVID-19. She has been involved in these efforts since early in the pandemic, when she ran an inpatient consult service at Yale dedicated to neurologic effects of acute COVID-19. She witnessed complications including stroke, encephalopathy, and seizures, among others.
Stroke during acute COVID-19 seemed to be associated with inflammation and endothelial activation or endotheliopathy. SARS-CoV-2 has been undetectable in the cerebrospinal fluid (CSF) of patients with acute COVID-19 and neurologic symptoms, but inflammatory cytokines can be present along with increased frequency of B cells. Anti–SARS-CoV-2 antibodies have also been found in CSF, some of which were auto reactive to brain tissue. The immune response was altered, compared with healthy controls, and in the CNS, compared with in the blood, “raising the question of whether inflammation and autoimmunity may be underlying causes of these syndromes,” said Dr. Spudich.
She also pointed to an MRI study of autopsied brain tissue of patients with COVID-19 and neurologic complications, which showed indications of both hemorrhagic and ischemic microvascular injury. “It’s just a reminder that, during acute COVID-19, there may be inflammation in the brain, there may be autoimmune reactions, and there may be vascular changes that underlie some of the neurologic syndromes that are seen,” said Dr. Spudich.
A panoply of different syndromes
In October, Yale set up a post-COVID neurologic clinic that brought together pulmonary, cardiology, and psychiatric specialists, many of whom saw the same patients, about 60% of whom had cognitive impairment, more than 40% had neuromuscular problems, and over 30% headache. “There’s not a single entity of a post-COVID neurologic syndrome. There’s a panoply of different syndromes that may have similar or distinct etiologies,” said Dr. Spudich.
Most patients were in their 30s, 40s, or 50s. That doesn’t necessarily mean this is the most common age range for these issues, though. There could be some bias if these individuals are seeking specialty care because they expected to recover from COVID-19 quickly. But it could be that there is something biologically unique among this age group that predisposes them to complications. Regardless, two out of three patients were never hospitalized, “suggesting that even mild COVID-19 can lead to some long-term sequelae,” said Dr. Spudich.
One potential explanation for long-term neurologic syndromes is that they are an extension of the inflammation, autoimmunity, and immune perturbation occurring during acute disease. One study looked at 18 cancer patients who had neurologic complications with COVID-19. Two months after onset, they had elevated markers of neuroinflammation and neuronal injury in the cerebral spinal fluid compared to cancer patients with no history of COVID-19.
Looking for biologic markers
An Italian study looked at patients who were evaluated during acute hospitalization and again 3 months later, and found that some markers of inflation in the blood were associated with later cognitive impairment. The patients were more severely ill, so it’s not clear what the findings mean for patients who present with neurologic symptoms after milder illness.
A PET scan study of 35 patients with persistent neurologic symptoms found patterns of reduced fluorodeoxyglucose uptake in some regions of the brain that are believed to be associated with some symptoms. Lower values were associated with greater severity for symptoms like memory dysfunction, and anosmia. “Why there might be hypometabolism in these regions I think needs to be assessed and used as a biomarker to associate hypometabolism with other kinds of processes in blood and spinal fluid,” said Dr. Spudich.
Along with colleagues at Yale, Dr. Spudich is conducting the MIND study, which is using PET and MRI imaging along with blood and CSF biomarkers to track the progress of patients after COVID-19. There are few results to discuss since only 20 patients have been recruited so far, except that brain imaging and blood values are generally normal despite neurologic complaints. Most were not hospitalized for COVID-19. Dr. Spudich highlighted one man in his 30s who developed new-onset psychosis, despite no previous history. Although clinical tests were all negative, a novel autoantibody detection method revealed a previously unknown autoreactive antibody in his spinal fluid. “This may suggest that there is autoantibody production in some individuals with post–COVID-19 psychosis, and potentially other syndromes,” said Dr. Spudich.
The research task ahead
The case illustrates the task ahead for neurology. “There’s a real research mandate to understand the biological substrates of these diverse disorders, not only to address the emergent public health concern and reduce the stigma in our patients, but to develop targeted therapeutic interventions,” said Dr. Spudich.
Anna Cervantes-Arslanian, MD, an associate professor of neurology at Boston University who also treats and studies patients with post-COVID neurologic symptoms, agreed with that assessment. “It’s not like every patient that has muscle aches and fatigue also has brain fog. It’s really hard to parse them out into specific phenotypes that are pretty classic. Some people will have all of those things, some will have very few of them,” said Dr. Cervantes-Arslanian. “We need to be able to identify them sand see if there is clustering of symptoms so we can better look into what the biological underpinnings are. That’s the first step to thinking about a therapeutic target.”
Dr. Spudich and Dr. Cervantes-Arslanian had no relevant financial disclosures.
Long-haul neurologic symptoms of COVID-19 seem to be distinct from neurologic conditions found in acute disease. Much work remains to be done to understand the biological mechanisms behind these problems, but inflammation and autoimmune responses may play a role in some cases.
Those were some of the takeaways from a talk by Serena Spudich, MD, who presented her research at the 2021 annual meeting of the American Academy of Neurology. Dr. Spudich is the division chief of neurologic infections and global neurology and codirector of the Center for Neuroepidemiology and Clinical Neurological Research at Yale University, New Haven, Conn.
Examining the nervous system’s involvement in COVID-19
Even early on in the pandemic, it became clear that there were lingering complaints of neuromuscular problems, cognitive dysfunction, and mood and psychiatric issues. Breathing and heart rate problems also can arise. “There seems to be a preponderance of syndromes that reflect involvement of the nervous system,” said Dr. Spudich.
To try to understand the etiology of these persistent problems, Dr. Spudich said it’s important to examine the nervous system’s involvement in acute COVID-19. She has been involved in these efforts since early in the pandemic, when she ran an inpatient consult service at Yale dedicated to neurologic effects of acute COVID-19. She witnessed complications including stroke, encephalopathy, and seizures, among others.
Stroke during acute COVID-19 seemed to be associated with inflammation and endothelial activation or endotheliopathy. SARS-CoV-2 has been undetectable in the cerebrospinal fluid (CSF) of patients with acute COVID-19 and neurologic symptoms, but inflammatory cytokines can be present along with increased frequency of B cells. Anti–SARS-CoV-2 antibodies have also been found in CSF, some of which were auto reactive to brain tissue. The immune response was altered, compared with healthy controls, and in the CNS, compared with in the blood, “raising the question of whether inflammation and autoimmunity may be underlying causes of these syndromes,” said Dr. Spudich.
She also pointed to an MRI study of autopsied brain tissue of patients with COVID-19 and neurologic complications, which showed indications of both hemorrhagic and ischemic microvascular injury. “It’s just a reminder that, during acute COVID-19, there may be inflammation in the brain, there may be autoimmune reactions, and there may be vascular changes that underlie some of the neurologic syndromes that are seen,” said Dr. Spudich.
A panoply of different syndromes
In October, Yale set up a post-COVID neurologic clinic that brought together pulmonary, cardiology, and psychiatric specialists, many of whom saw the same patients, about 60% of whom had cognitive impairment, more than 40% had neuromuscular problems, and over 30% headache. “There’s not a single entity of a post-COVID neurologic syndrome. There’s a panoply of different syndromes that may have similar or distinct etiologies,” said Dr. Spudich.
Most patients were in their 30s, 40s, or 50s. That doesn’t necessarily mean this is the most common age range for these issues, though. There could be some bias if these individuals are seeking specialty care because they expected to recover from COVID-19 quickly. But it could be that there is something biologically unique among this age group that predisposes them to complications. Regardless, two out of three patients were never hospitalized, “suggesting that even mild COVID-19 can lead to some long-term sequelae,” said Dr. Spudich.
One potential explanation for long-term neurologic syndromes is that they are an extension of the inflammation, autoimmunity, and immune perturbation occurring during acute disease. One study looked at 18 cancer patients who had neurologic complications with COVID-19. Two months after onset, they had elevated markers of neuroinflammation and neuronal injury in the cerebral spinal fluid compared to cancer patients with no history of COVID-19.
Looking for biologic markers
An Italian study looked at patients who were evaluated during acute hospitalization and again 3 months later, and found that some markers of inflation in the blood were associated with later cognitive impairment. The patients were more severely ill, so it’s not clear what the findings mean for patients who present with neurologic symptoms after milder illness.
A PET scan study of 35 patients with persistent neurologic symptoms found patterns of reduced fluorodeoxyglucose uptake in some regions of the brain that are believed to be associated with some symptoms. Lower values were associated with greater severity for symptoms like memory dysfunction, and anosmia. “Why there might be hypometabolism in these regions I think needs to be assessed and used as a biomarker to associate hypometabolism with other kinds of processes in blood and spinal fluid,” said Dr. Spudich.
Along with colleagues at Yale, Dr. Spudich is conducting the MIND study, which is using PET and MRI imaging along with blood and CSF biomarkers to track the progress of patients after COVID-19. There are few results to discuss since only 20 patients have been recruited so far, except that brain imaging and blood values are generally normal despite neurologic complaints. Most were not hospitalized for COVID-19. Dr. Spudich highlighted one man in his 30s who developed new-onset psychosis, despite no previous history. Although clinical tests were all negative, a novel autoantibody detection method revealed a previously unknown autoreactive antibody in his spinal fluid. “This may suggest that there is autoantibody production in some individuals with post–COVID-19 psychosis, and potentially other syndromes,” said Dr. Spudich.
The research task ahead
The case illustrates the task ahead for neurology. “There’s a real research mandate to understand the biological substrates of these diverse disorders, not only to address the emergent public health concern and reduce the stigma in our patients, but to develop targeted therapeutic interventions,” said Dr. Spudich.
Anna Cervantes-Arslanian, MD, an associate professor of neurology at Boston University who also treats and studies patients with post-COVID neurologic symptoms, agreed with that assessment. “It’s not like every patient that has muscle aches and fatigue also has brain fog. It’s really hard to parse them out into specific phenotypes that are pretty classic. Some people will have all of those things, some will have very few of them,” said Dr. Cervantes-Arslanian. “We need to be able to identify them sand see if there is clustering of symptoms so we can better look into what the biological underpinnings are. That’s the first step to thinking about a therapeutic target.”
Dr. Spudich and Dr. Cervantes-Arslanian had no relevant financial disclosures.
Long-haul neurologic symptoms of COVID-19 seem to be distinct from neurologic conditions found in acute disease. Much work remains to be done to understand the biological mechanisms behind these problems, but inflammation and autoimmune responses may play a role in some cases.
Those were some of the takeaways from a talk by Serena Spudich, MD, who presented her research at the 2021 annual meeting of the American Academy of Neurology. Dr. Spudich is the division chief of neurologic infections and global neurology and codirector of the Center for Neuroepidemiology and Clinical Neurological Research at Yale University, New Haven, Conn.
Examining the nervous system’s involvement in COVID-19
Even early on in the pandemic, it became clear that there were lingering complaints of neuromuscular problems, cognitive dysfunction, and mood and psychiatric issues. Breathing and heart rate problems also can arise. “There seems to be a preponderance of syndromes that reflect involvement of the nervous system,” said Dr. Spudich.
To try to understand the etiology of these persistent problems, Dr. Spudich said it’s important to examine the nervous system’s involvement in acute COVID-19. She has been involved in these efforts since early in the pandemic, when she ran an inpatient consult service at Yale dedicated to neurologic effects of acute COVID-19. She witnessed complications including stroke, encephalopathy, and seizures, among others.
Stroke during acute COVID-19 seemed to be associated with inflammation and endothelial activation or endotheliopathy. SARS-CoV-2 has been undetectable in the cerebrospinal fluid (CSF) of patients with acute COVID-19 and neurologic symptoms, but inflammatory cytokines can be present along with increased frequency of B cells. Anti–SARS-CoV-2 antibodies have also been found in CSF, some of which were auto reactive to brain tissue. The immune response was altered, compared with healthy controls, and in the CNS, compared with in the blood, “raising the question of whether inflammation and autoimmunity may be underlying causes of these syndromes,” said Dr. Spudich.
She also pointed to an MRI study of autopsied brain tissue of patients with COVID-19 and neurologic complications, which showed indications of both hemorrhagic and ischemic microvascular injury. “It’s just a reminder that, during acute COVID-19, there may be inflammation in the brain, there may be autoimmune reactions, and there may be vascular changes that underlie some of the neurologic syndromes that are seen,” said Dr. Spudich.
A panoply of different syndromes
In October, Yale set up a post-COVID neurologic clinic that brought together pulmonary, cardiology, and psychiatric specialists, many of whom saw the same patients, about 60% of whom had cognitive impairment, more than 40% had neuromuscular problems, and over 30% headache. “There’s not a single entity of a post-COVID neurologic syndrome. There’s a panoply of different syndromes that may have similar or distinct etiologies,” said Dr. Spudich.
Most patients were in their 30s, 40s, or 50s. That doesn’t necessarily mean this is the most common age range for these issues, though. There could be some bias if these individuals are seeking specialty care because they expected to recover from COVID-19 quickly. But it could be that there is something biologically unique among this age group that predisposes them to complications. Regardless, two out of three patients were never hospitalized, “suggesting that even mild COVID-19 can lead to some long-term sequelae,” said Dr. Spudich.
One potential explanation for long-term neurologic syndromes is that they are an extension of the inflammation, autoimmunity, and immune perturbation occurring during acute disease. One study looked at 18 cancer patients who had neurologic complications with COVID-19. Two months after onset, they had elevated markers of neuroinflammation and neuronal injury in the cerebral spinal fluid compared to cancer patients with no history of COVID-19.
Looking for biologic markers
An Italian study looked at patients who were evaluated during acute hospitalization and again 3 months later, and found that some markers of inflation in the blood were associated with later cognitive impairment. The patients were more severely ill, so it’s not clear what the findings mean for patients who present with neurologic symptoms after milder illness.
A PET scan study of 35 patients with persistent neurologic symptoms found patterns of reduced fluorodeoxyglucose uptake in some regions of the brain that are believed to be associated with some symptoms. Lower values were associated with greater severity for symptoms like memory dysfunction, and anosmia. “Why there might be hypometabolism in these regions I think needs to be assessed and used as a biomarker to associate hypometabolism with other kinds of processes in blood and spinal fluid,” said Dr. Spudich.
Along with colleagues at Yale, Dr. Spudich is conducting the MIND study, which is using PET and MRI imaging along with blood and CSF biomarkers to track the progress of patients after COVID-19. There are few results to discuss since only 20 patients have been recruited so far, except that brain imaging and blood values are generally normal despite neurologic complaints. Most were not hospitalized for COVID-19. Dr. Spudich highlighted one man in his 30s who developed new-onset psychosis, despite no previous history. Although clinical tests were all negative, a novel autoantibody detection method revealed a previously unknown autoreactive antibody in his spinal fluid. “This may suggest that there is autoantibody production in some individuals with post–COVID-19 psychosis, and potentially other syndromes,” said Dr. Spudich.
The research task ahead
The case illustrates the task ahead for neurology. “There’s a real research mandate to understand the biological substrates of these diverse disorders, not only to address the emergent public health concern and reduce the stigma in our patients, but to develop targeted therapeutic interventions,” said Dr. Spudich.
Anna Cervantes-Arslanian, MD, an associate professor of neurology at Boston University who also treats and studies patients with post-COVID neurologic symptoms, agreed with that assessment. “It’s not like every patient that has muscle aches and fatigue also has brain fog. It’s really hard to parse them out into specific phenotypes that are pretty classic. Some people will have all of those things, some will have very few of them,” said Dr. Cervantes-Arslanian. “We need to be able to identify them sand see if there is clustering of symptoms so we can better look into what the biological underpinnings are. That’s the first step to thinking about a therapeutic target.”
Dr. Spudich and Dr. Cervantes-Arslanian had no relevant financial disclosures.
FROM AAN 2021
Most patients with chronic inflammatory diseases have sufficient response to COVID-19 vaccination
Glucocorticoids and B-cell–depleting therapies are trouble spots
Although most patients with chronic inflammatory diseases mounted immune responses after two doses of mRNA-based COVID-19 vaccines, glucocorticoids and B-cell–depleting therapies markedly reduced the response, according to a recently published preprint of a new study.
The study, published on MedRxiv and not yet peer reviewed, involved a prospective look at 133 patients with chronic inflammatory disease (CID) and 53 patients with healthy immune systems at Washington University, St. Louis, and the University of California, San Francisco. It is regarded as the largest and most detailed study yet in how vaccines perform in people with immune-mediated inflammatory disease. The patients were enrolled between December 2020 and March 2021, and the most common diseases were inflammatory bowel disease (32%), rheumatoid arthritis (29%), spondyloarthritis (15%), and systemic lupus erythematosus (11%).
A ‘modest’ reduction in antibody response
Senior author Alfred Kim, MD, PhD, of the department of medicine at Washington University, said the overall results so far are encouraging.
“Most patients with an autoimmune disease that are on immunosuppression can mount antibody responses,” he said. “We’re seeing the majority of our subjects respond.”
The immune-healthy controls and most of the patients with CID had a robust immune response against the spike protein, although the CID group had a mean reduction in antibody titers that was three times lower than the controls (P = .0092). The CID group similarly had a 2.7-fold reduction in preventing neutralization, or halting the virus’ ability to infect (P < .0001), researchers reported.
This reduction in response is “modest,” he said.
“Is the level of reduction going to be detrimental for protection? Time will tell,” he said, adding that researchers anticipate that it won’t have a critical effect on protection because responses tended to be within the range of the immunocompetent controls, who themselves had wildly varied antibody titers across a 20-fold range. “ ‘Optimal’ isn’t necessarily the same as ‘sufficient.’ ”
Type of medication has big impact on antibody titers
But there was a wide variety of effects on the immune response depending on the medication. Glucorticoids resulted in a response that was 10 times lower than the immune-healthy controls, as well as fewer circulating plasmablasts after vaccination. Researchers found that 98% of controls were seropositive for antibody, compared with 92% of those with CID who were not taking prednisone, and 65% of CID patients on prednisone (P = .0006 and .0115, respectively). Prevention of neutralization of the virus was similarly reduced in those groups, compared with the controls. Dr. Kim noted this was a small sample size, with about 15 patients. These effects were seen regardless of the dose.
“We would’ve anticipated this would have been dose dependent, so this was a little bit surprising,” Dr. Kim said.
B-cell–depleting therapies, such as rituximab (Rituxan) and ocrelizumab (Ocrevus), reduced antibody titers by 36 times, compared with controls (P < .0001), with a similar reduction in preventing infection (P = .0066), the researchers found. The reduction in antibody titers was the most pronounced among those who had received B-cell–depleting therapies within the previous 6 months. Dr. Kim noted this was a small sample size, with about 10 patients.
CID study subjects taking an antimetabolite, including methotrexate, had an average of a two- to threefold reduction in antibody titers and in neutralization (P = .0006). This reduction was greatest with methotrexate, researchers found (P = .0027).
JAK inhibitors also significantly reduced antibody titers (P = .0066), but the reduction in neutralization of the virus was not significant. In addition, researchers found a reduction in antibody titers, the prevention of viral infection, and circulating plasmablasts among those on tumor necrosis factor (TNF) inhibitors, compared with controls, but these were insignificant statistically except for virus neutralization.
Dr. Kim said he hopes the glucocorticoid data spur physicians to try harder to wean patients off the drugs, when possible, in keeping with recommendations already in place.
“The general culture in rheumatology has been very lax about the need to reduce glucocorticoids,” he said. “This reinvigorates that call.” Questions about possible drug holidays from glucocorticoids remain, regarding how long a holiday would be needed, he said. He noted that many patients on glucocorticoids nonetheless mounted responses.
Those on B-cell–depleting therapies present a “much more difficult” question, he said. Some patients possibly could wait a bit longer than their normal, every-6-month schedule, but it’s an individual decision, he said. Since a booster of influenza vaccine has been found to enhance the response even within the 6-month window among ocrelizumab patients, a booster of COVID-19 vaccine might also help, although this remains to be studied.
The study group has already increased its sample size and is looking at adverse reactions and long-term immune responses, Dr. Kim said.
Encouraging, rather than discouraging, results
Leonard Calabrese, DO, professor of medicine at the Cleveland Clinic in Ohio, said the findings shouldn’t discourage clinicians from encouraging vaccination.
“There’s still a preponderance of people who will develop a robust antibody vaccine response,” he said.
He cautioned that the findings look only at antibodies to the spike protein and at plasmablasts. The reduction in these titers is “of concern,” he said, but “we don’t really know with certainty what are the effects of these drugs, and these data are on the overall biologic protective effect of the vaccine. There’s much more to a vaccine response than anti–spike protein and plasmablasts,” including cell-mediated immune response.
For an individual patient, the findings “mean a lot,” he said.
“I think that people who are on significant prednisone and B-cell–depleting agents, I think you have to share with them that there’s a reasonable chance that you’re not going to be making a response similar to healthy people,” he said. “Thus, even with your vaccine, we’re not going to cut you loose to do things that are violating social distancing and group settings. … Should you be hugging your grandchildren if you’re a rituximab vaccine recipient? I think I would wait until we have a little bit more data.”
Kevin Winthrop, MD, MPH, professor of ophthalmology at Oregon Health & Science University, Portland, where he studies vaccinations in the immunocompromised, said that glucocorticoids tend to have little effect on vaccinations generally at low doses.
When effects are seen they can be difficult to interpret, he said.
“It’s hard to extricate that from the effect of the underlying disease,” he said. The drug can be a proxy for worse disease control.
Although it’s a small study, it’s reassuring that overall the responses were similar to healthy controls.
For B-cell–depleting therapies, his usual guidance is to not give vaccine until a patient is at least 3 months out from their last dose, and not to restart until at least 2 weeks after vaccination.
“It’s not surprising that some of these DMARDs [disease-modifying antirheumatic drugs] do negatively affect vaccine response, particularly B-cell–depletion therapy. We need to do some studies to find a way to overcome that, or optimize delivery of the vaccine.”
Dr. Kim reported participating in consulting, advisory board, or speaker’s bureau for Alexion, Aurinia, Annexon Biosciences, Exagen Diagnostics, and GlaxoSmithKline, and receiving funding under a sponsored research agreement unrelated to the data in the paper from GlaxoSmithKline. Dr. Winthrop reported receiving consulting fees from Pfizer, AbbVie, UCB, Eli Lilly, Galapagos, GlaxoSmithKline, Roche, Gilead, Bristol-Myers Squibb, Regeneron, Sanofi, AstraZeneca, Novartis, and research grants from Bristol-Myers Squibb and Pfizer. Dr. Calabrese reported no relevant disclosures.
Glucocorticoids and B-cell–depleting therapies are trouble spots
Glucocorticoids and B-cell–depleting therapies are trouble spots
Although most patients with chronic inflammatory diseases mounted immune responses after two doses of mRNA-based COVID-19 vaccines, glucocorticoids and B-cell–depleting therapies markedly reduced the response, according to a recently published preprint of a new study.
The study, published on MedRxiv and not yet peer reviewed, involved a prospective look at 133 patients with chronic inflammatory disease (CID) and 53 patients with healthy immune systems at Washington University, St. Louis, and the University of California, San Francisco. It is regarded as the largest and most detailed study yet in how vaccines perform in people with immune-mediated inflammatory disease. The patients were enrolled between December 2020 and March 2021, and the most common diseases were inflammatory bowel disease (32%), rheumatoid arthritis (29%), spondyloarthritis (15%), and systemic lupus erythematosus (11%).
A ‘modest’ reduction in antibody response
Senior author Alfred Kim, MD, PhD, of the department of medicine at Washington University, said the overall results so far are encouraging.
“Most patients with an autoimmune disease that are on immunosuppression can mount antibody responses,” he said. “We’re seeing the majority of our subjects respond.”
The immune-healthy controls and most of the patients with CID had a robust immune response against the spike protein, although the CID group had a mean reduction in antibody titers that was three times lower than the controls (P = .0092). The CID group similarly had a 2.7-fold reduction in preventing neutralization, or halting the virus’ ability to infect (P < .0001), researchers reported.
This reduction in response is “modest,” he said.
“Is the level of reduction going to be detrimental for protection? Time will tell,” he said, adding that researchers anticipate that it won’t have a critical effect on protection because responses tended to be within the range of the immunocompetent controls, who themselves had wildly varied antibody titers across a 20-fold range. “ ‘Optimal’ isn’t necessarily the same as ‘sufficient.’ ”
Type of medication has big impact on antibody titers
But there was a wide variety of effects on the immune response depending on the medication. Glucorticoids resulted in a response that was 10 times lower than the immune-healthy controls, as well as fewer circulating plasmablasts after vaccination. Researchers found that 98% of controls were seropositive for antibody, compared with 92% of those with CID who were not taking prednisone, and 65% of CID patients on prednisone (P = .0006 and .0115, respectively). Prevention of neutralization of the virus was similarly reduced in those groups, compared with the controls. Dr. Kim noted this was a small sample size, with about 15 patients. These effects were seen regardless of the dose.
“We would’ve anticipated this would have been dose dependent, so this was a little bit surprising,” Dr. Kim said.
B-cell–depleting therapies, such as rituximab (Rituxan) and ocrelizumab (Ocrevus), reduced antibody titers by 36 times, compared with controls (P < .0001), with a similar reduction in preventing infection (P = .0066), the researchers found. The reduction in antibody titers was the most pronounced among those who had received B-cell–depleting therapies within the previous 6 months. Dr. Kim noted this was a small sample size, with about 10 patients.
CID study subjects taking an antimetabolite, including methotrexate, had an average of a two- to threefold reduction in antibody titers and in neutralization (P = .0006). This reduction was greatest with methotrexate, researchers found (P = .0027).
JAK inhibitors also significantly reduced antibody titers (P = .0066), but the reduction in neutralization of the virus was not significant. In addition, researchers found a reduction in antibody titers, the prevention of viral infection, and circulating plasmablasts among those on tumor necrosis factor (TNF) inhibitors, compared with controls, but these were insignificant statistically except for virus neutralization.
Dr. Kim said he hopes the glucocorticoid data spur physicians to try harder to wean patients off the drugs, when possible, in keeping with recommendations already in place.
“The general culture in rheumatology has been very lax about the need to reduce glucocorticoids,” he said. “This reinvigorates that call.” Questions about possible drug holidays from glucocorticoids remain, regarding how long a holiday would be needed, he said. He noted that many patients on glucocorticoids nonetheless mounted responses.
Those on B-cell–depleting therapies present a “much more difficult” question, he said. Some patients possibly could wait a bit longer than their normal, every-6-month schedule, but it’s an individual decision, he said. Since a booster of influenza vaccine has been found to enhance the response even within the 6-month window among ocrelizumab patients, a booster of COVID-19 vaccine might also help, although this remains to be studied.
The study group has already increased its sample size and is looking at adverse reactions and long-term immune responses, Dr. Kim said.
Encouraging, rather than discouraging, results
Leonard Calabrese, DO, professor of medicine at the Cleveland Clinic in Ohio, said the findings shouldn’t discourage clinicians from encouraging vaccination.
“There’s still a preponderance of people who will develop a robust antibody vaccine response,” he said.
He cautioned that the findings look only at antibodies to the spike protein and at plasmablasts. The reduction in these titers is “of concern,” he said, but “we don’t really know with certainty what are the effects of these drugs, and these data are on the overall biologic protective effect of the vaccine. There’s much more to a vaccine response than anti–spike protein and plasmablasts,” including cell-mediated immune response.
For an individual patient, the findings “mean a lot,” he said.
“I think that people who are on significant prednisone and B-cell–depleting agents, I think you have to share with them that there’s a reasonable chance that you’re not going to be making a response similar to healthy people,” he said. “Thus, even with your vaccine, we’re not going to cut you loose to do things that are violating social distancing and group settings. … Should you be hugging your grandchildren if you’re a rituximab vaccine recipient? I think I would wait until we have a little bit more data.”
Kevin Winthrop, MD, MPH, professor of ophthalmology at Oregon Health & Science University, Portland, where he studies vaccinations in the immunocompromised, said that glucocorticoids tend to have little effect on vaccinations generally at low doses.
When effects are seen they can be difficult to interpret, he said.
“It’s hard to extricate that from the effect of the underlying disease,” he said. The drug can be a proxy for worse disease control.
Although it’s a small study, it’s reassuring that overall the responses were similar to healthy controls.
For B-cell–depleting therapies, his usual guidance is to not give vaccine until a patient is at least 3 months out from their last dose, and not to restart until at least 2 weeks after vaccination.
“It’s not surprising that some of these DMARDs [disease-modifying antirheumatic drugs] do negatively affect vaccine response, particularly B-cell–depletion therapy. We need to do some studies to find a way to overcome that, or optimize delivery of the vaccine.”
Dr. Kim reported participating in consulting, advisory board, or speaker’s bureau for Alexion, Aurinia, Annexon Biosciences, Exagen Diagnostics, and GlaxoSmithKline, and receiving funding under a sponsored research agreement unrelated to the data in the paper from GlaxoSmithKline. Dr. Winthrop reported receiving consulting fees from Pfizer, AbbVie, UCB, Eli Lilly, Galapagos, GlaxoSmithKline, Roche, Gilead, Bristol-Myers Squibb, Regeneron, Sanofi, AstraZeneca, Novartis, and research grants from Bristol-Myers Squibb and Pfizer. Dr. Calabrese reported no relevant disclosures.
Although most patients with chronic inflammatory diseases mounted immune responses after two doses of mRNA-based COVID-19 vaccines, glucocorticoids and B-cell–depleting therapies markedly reduced the response, according to a recently published preprint of a new study.
The study, published on MedRxiv and not yet peer reviewed, involved a prospective look at 133 patients with chronic inflammatory disease (CID) and 53 patients with healthy immune systems at Washington University, St. Louis, and the University of California, San Francisco. It is regarded as the largest and most detailed study yet in how vaccines perform in people with immune-mediated inflammatory disease. The patients were enrolled between December 2020 and March 2021, and the most common diseases were inflammatory bowel disease (32%), rheumatoid arthritis (29%), spondyloarthritis (15%), and systemic lupus erythematosus (11%).
A ‘modest’ reduction in antibody response
Senior author Alfred Kim, MD, PhD, of the department of medicine at Washington University, said the overall results so far are encouraging.
“Most patients with an autoimmune disease that are on immunosuppression can mount antibody responses,” he said. “We’re seeing the majority of our subjects respond.”
The immune-healthy controls and most of the patients with CID had a robust immune response against the spike protein, although the CID group had a mean reduction in antibody titers that was three times lower than the controls (P = .0092). The CID group similarly had a 2.7-fold reduction in preventing neutralization, or halting the virus’ ability to infect (P < .0001), researchers reported.
This reduction in response is “modest,” he said.
“Is the level of reduction going to be detrimental for protection? Time will tell,” he said, adding that researchers anticipate that it won’t have a critical effect on protection because responses tended to be within the range of the immunocompetent controls, who themselves had wildly varied antibody titers across a 20-fold range. “ ‘Optimal’ isn’t necessarily the same as ‘sufficient.’ ”
Type of medication has big impact on antibody titers
But there was a wide variety of effects on the immune response depending on the medication. Glucorticoids resulted in a response that was 10 times lower than the immune-healthy controls, as well as fewer circulating plasmablasts after vaccination. Researchers found that 98% of controls were seropositive for antibody, compared with 92% of those with CID who were not taking prednisone, and 65% of CID patients on prednisone (P = .0006 and .0115, respectively). Prevention of neutralization of the virus was similarly reduced in those groups, compared with the controls. Dr. Kim noted this was a small sample size, with about 15 patients. These effects were seen regardless of the dose.
“We would’ve anticipated this would have been dose dependent, so this was a little bit surprising,” Dr. Kim said.
B-cell–depleting therapies, such as rituximab (Rituxan) and ocrelizumab (Ocrevus), reduced antibody titers by 36 times, compared with controls (P < .0001), with a similar reduction in preventing infection (P = .0066), the researchers found. The reduction in antibody titers was the most pronounced among those who had received B-cell–depleting therapies within the previous 6 months. Dr. Kim noted this was a small sample size, with about 10 patients.
CID study subjects taking an antimetabolite, including methotrexate, had an average of a two- to threefold reduction in antibody titers and in neutralization (P = .0006). This reduction was greatest with methotrexate, researchers found (P = .0027).
JAK inhibitors also significantly reduced antibody titers (P = .0066), but the reduction in neutralization of the virus was not significant. In addition, researchers found a reduction in antibody titers, the prevention of viral infection, and circulating plasmablasts among those on tumor necrosis factor (TNF) inhibitors, compared with controls, but these were insignificant statistically except for virus neutralization.
Dr. Kim said he hopes the glucocorticoid data spur physicians to try harder to wean patients off the drugs, when possible, in keeping with recommendations already in place.
“The general culture in rheumatology has been very lax about the need to reduce glucocorticoids,” he said. “This reinvigorates that call.” Questions about possible drug holidays from glucocorticoids remain, regarding how long a holiday would be needed, he said. He noted that many patients on glucocorticoids nonetheless mounted responses.
Those on B-cell–depleting therapies present a “much more difficult” question, he said. Some patients possibly could wait a bit longer than their normal, every-6-month schedule, but it’s an individual decision, he said. Since a booster of influenza vaccine has been found to enhance the response even within the 6-month window among ocrelizumab patients, a booster of COVID-19 vaccine might also help, although this remains to be studied.
The study group has already increased its sample size and is looking at adverse reactions and long-term immune responses, Dr. Kim said.
Encouraging, rather than discouraging, results
Leonard Calabrese, DO, professor of medicine at the Cleveland Clinic in Ohio, said the findings shouldn’t discourage clinicians from encouraging vaccination.
“There’s still a preponderance of people who will develop a robust antibody vaccine response,” he said.
He cautioned that the findings look only at antibodies to the spike protein and at plasmablasts. The reduction in these titers is “of concern,” he said, but “we don’t really know with certainty what are the effects of these drugs, and these data are on the overall biologic protective effect of the vaccine. There’s much more to a vaccine response than anti–spike protein and plasmablasts,” including cell-mediated immune response.
For an individual patient, the findings “mean a lot,” he said.
“I think that people who are on significant prednisone and B-cell–depleting agents, I think you have to share with them that there’s a reasonable chance that you’re not going to be making a response similar to healthy people,” he said. “Thus, even with your vaccine, we’re not going to cut you loose to do things that are violating social distancing and group settings. … Should you be hugging your grandchildren if you’re a rituximab vaccine recipient? I think I would wait until we have a little bit more data.”
Kevin Winthrop, MD, MPH, professor of ophthalmology at Oregon Health & Science University, Portland, where he studies vaccinations in the immunocompromised, said that glucocorticoids tend to have little effect on vaccinations generally at low doses.
When effects are seen they can be difficult to interpret, he said.
“It’s hard to extricate that from the effect of the underlying disease,” he said. The drug can be a proxy for worse disease control.
Although it’s a small study, it’s reassuring that overall the responses were similar to healthy controls.
For B-cell–depleting therapies, his usual guidance is to not give vaccine until a patient is at least 3 months out from their last dose, and not to restart until at least 2 weeks after vaccination.
“It’s not surprising that some of these DMARDs [disease-modifying antirheumatic drugs] do negatively affect vaccine response, particularly B-cell–depletion therapy. We need to do some studies to find a way to overcome that, or optimize delivery of the vaccine.”
Dr. Kim reported participating in consulting, advisory board, or speaker’s bureau for Alexion, Aurinia, Annexon Biosciences, Exagen Diagnostics, and GlaxoSmithKline, and receiving funding under a sponsored research agreement unrelated to the data in the paper from GlaxoSmithKline. Dr. Winthrop reported receiving consulting fees from Pfizer, AbbVie, UCB, Eli Lilly, Galapagos, GlaxoSmithKline, Roche, Gilead, Bristol-Myers Squibb, Regeneron, Sanofi, AstraZeneca, Novartis, and research grants from Bristol-Myers Squibb and Pfizer. Dr. Calabrese reported no relevant disclosures.
FROM MEDRXIV
Stroke is ‘not a common complication’ in COVID-19
One study showed a stroke rate of 2.2% among patients with COVID-19 admitted to intensive care in 52 different countries. Another found a stroke rate of 1.48% in patients hospitalized with COVID-19 from 70 different countries. These researchers also found a reduction in stroke presentations and stroke care during the pandemic.
Both studies will be presented at the American Academy of Neurology’s 2021 annual meeting.
“Stroke has been a known serious complication of COVID-19, with some studies reporting a higher-than-expected occurrence, especially in young people,” said coauthor of the intensive care study, Jonathon Fanning, MBBS, PhD, University of Queensland, Brisbane, Australia.
“However, among the sickest of COVID patients – those admitted to an ICU – our research found that stroke was not a common complication and that ischemic stroke did not increase the risk of death,” he added.
Hemorrhagic stroke more common?
In this study, researchers analyzed a database of 2,699 patients who were admitted to the intensive care unit with COVID-19 in 52 countries and found that 59 of these patients (2.2%) subsequently sustained a stroke.
Most of the strokes identified in this cohort were hemorrhagic (46%), with 32% being ischemic and 22% unspecified. Hemorrhagic stroke was associated with a fivefold increased risk for death compared with patients who did not have a stroke. Of those with a hemorrhagic stroke, 72% died, but only 15% died of the stroke. Rather, multiorgan failure was the leading cause of death.
There was no association between ischemic stroke and mortality.
“There is scarce research on new-onset stroke complicating ICU admissions, and many of the limitations of assessing stroke in ICU populations confound the true values and result in variability in reported incidence anywhere from a 1%-4% incidence,” Dr. Fanning said.
He noted that a large Korean study had shown a 1.2% rate of stroke in patients without COVID admitted to non-neurologic ICUs. “In light of this, I think this 2% is higher than we would expect in a general ICU population, but in the context of earlier reports of COVID-19–associated risk for stroke, this figure is actually somewhat reassuring,” Dr. Fanning said.
Asked how this study compared with the large American Heart Association study recently reported that showed an overall rate of ischemic stroke of 0.75%, Dr. Fanning said the two studies reported on different populations, which makes them difficult to compare.
“Our study specifically reports on new-onset stroke complicating ICU admission,” he noted. “The AHA study is a large study of all patients admitted to hospital, but both studies identified less than previous estimates of COVID-related stroke.”
Largest sample to date
The other study, which includes 119,967 COVID-19 hospitalizations and represents the largest sample reporting the concomitant diagnoses of stroke and SARS-CoV-2 infection to date, was presented at the AAN meeting by Thanh N. Nguyen, MD, a professor at Boston University.
This study has also been published online in Neurology, with first author Raul G. Nogueira, MD, Emory University, Atlanta.
In this international observational, retrospective study across 6 continents, 70 countries, and 457 stroke centers, there was a 1.48% stroke rate across 119,967 COVID-19 hospitalizations. SARS-CoV-2 infection was noted in 3.3% (1,722) of all stroke admissions, which numbered 52,026.
The researchers identified stroke diagnoses by the International Classification of Diseases, 10th revision, codes and/or classifications in stroke center databases, and rates of stroke hospitalizations and numbers of patients receiving thrombolysis were compared between the first 4 months of the pandemic (March to June 2020) compared with two control 4-month periods.
Global decline in stroke care during pandemic
Results showed a global decline in the number of stroke patients admitted to the hospital as well as acute stroke treatments, such as thrombolysis, during the first wave of the COVID-19 pandemic. The researchers found that there were 91,373 stroke admissions in the 4 months immediately before the pandemic, compared with 80,894 admissions during the first 4 pandemic months, representing an 11.5% decline.
They also report that 13,334 stroke patients received intravenous thrombolysis in the 4 months preceding the pandemic, compared with 11,570 during the first 4 pandemic months, representing a 13.2% drop.
Interhospital transfers after thrombolysis for a higher level of stroke care decreased from 1,337 before the pandemic to 1,178 during the pandemic, a reduction of 11.9%.
There were greater declines in primary compared with comprehensive stroke centers for stroke hospitalizations (change, –17.3% vs. –10.3%) and for the number of patients receiving thrombolysis (change, –15.5% vs. –12.6%).
The volume of stroke hospitalizations increased by 9.5% in the two later pandemic months (May, June) versus the two earlier months (March, April), with greater recovery in hospitals with lower COVID-19 hospitalization volume, high-volume stroke centers, and comprehensive stroke centers.
Dr. Nguyen suggested that reasons for the reductions in these stroke numbers at the beginning of the pandemic could include a reduction in stroke risk due to a reduction of exposure to other viral infections or patients not presenting to the hospital for fear of contracting the coronavirus.
The higher recovery of stroke volume in high-volume stroke centers and comprehensive stroke centers may represent patients with higher needs – those having more severe strokes – seeking care more frequently than those with milder symptoms, she noted.
“Preserving access to stroke care and emergency stroke care amidst a pandemic is as important as educating patients on the importance of presenting to the hospital in the event of stroke-like symptoms,” Dr. Nguyen concluded.
“We continue to advocate that if a patient has stroke-like symptoms, such as loss of speech, strength, vision, or balance, it is important for the patient to seek medical care as an emergency, as there are treatments that can improve a patient’s ability to recover from disabling stroke in earlier rather than later time windows,” she added.
In the publication, the authors wrote, “Our results concur with other recent reports on the collateral effects of the COVID-19 pandemic on stroke systems of care,” but added that “this is among the first descriptions of the change at a global level, including primary and comprehensive stroke centers.”
They said that hospital access related to high COVID-19 burden was unlikely a factor because the decline was seen in centers with a few or no patients with COVID-19. They suggested that patient fear of contracting coronavirus may have played a role, along with a decrease in presentation of transient ischemic attacks, mild strokes, or moderate strokes, and physical distancing measures may have prevented the timely witnessing of a stroke.
A version of this article first appeared on Medscape.com.
One study showed a stroke rate of 2.2% among patients with COVID-19 admitted to intensive care in 52 different countries. Another found a stroke rate of 1.48% in patients hospitalized with COVID-19 from 70 different countries. These researchers also found a reduction in stroke presentations and stroke care during the pandemic.
Both studies will be presented at the American Academy of Neurology’s 2021 annual meeting.
“Stroke has been a known serious complication of COVID-19, with some studies reporting a higher-than-expected occurrence, especially in young people,” said coauthor of the intensive care study, Jonathon Fanning, MBBS, PhD, University of Queensland, Brisbane, Australia.
“However, among the sickest of COVID patients – those admitted to an ICU – our research found that stroke was not a common complication and that ischemic stroke did not increase the risk of death,” he added.
Hemorrhagic stroke more common?
In this study, researchers analyzed a database of 2,699 patients who were admitted to the intensive care unit with COVID-19 in 52 countries and found that 59 of these patients (2.2%) subsequently sustained a stroke.
Most of the strokes identified in this cohort were hemorrhagic (46%), with 32% being ischemic and 22% unspecified. Hemorrhagic stroke was associated with a fivefold increased risk for death compared with patients who did not have a stroke. Of those with a hemorrhagic stroke, 72% died, but only 15% died of the stroke. Rather, multiorgan failure was the leading cause of death.
There was no association between ischemic stroke and mortality.
“There is scarce research on new-onset stroke complicating ICU admissions, and many of the limitations of assessing stroke in ICU populations confound the true values and result in variability in reported incidence anywhere from a 1%-4% incidence,” Dr. Fanning said.
He noted that a large Korean study had shown a 1.2% rate of stroke in patients without COVID admitted to non-neurologic ICUs. “In light of this, I think this 2% is higher than we would expect in a general ICU population, but in the context of earlier reports of COVID-19–associated risk for stroke, this figure is actually somewhat reassuring,” Dr. Fanning said.
Asked how this study compared with the large American Heart Association study recently reported that showed an overall rate of ischemic stroke of 0.75%, Dr. Fanning said the two studies reported on different populations, which makes them difficult to compare.
“Our study specifically reports on new-onset stroke complicating ICU admission,” he noted. “The AHA study is a large study of all patients admitted to hospital, but both studies identified less than previous estimates of COVID-related stroke.”
Largest sample to date
The other study, which includes 119,967 COVID-19 hospitalizations and represents the largest sample reporting the concomitant diagnoses of stroke and SARS-CoV-2 infection to date, was presented at the AAN meeting by Thanh N. Nguyen, MD, a professor at Boston University.
This study has also been published online in Neurology, with first author Raul G. Nogueira, MD, Emory University, Atlanta.
In this international observational, retrospective study across 6 continents, 70 countries, and 457 stroke centers, there was a 1.48% stroke rate across 119,967 COVID-19 hospitalizations. SARS-CoV-2 infection was noted in 3.3% (1,722) of all stroke admissions, which numbered 52,026.
The researchers identified stroke diagnoses by the International Classification of Diseases, 10th revision, codes and/or classifications in stroke center databases, and rates of stroke hospitalizations and numbers of patients receiving thrombolysis were compared between the first 4 months of the pandemic (March to June 2020) compared with two control 4-month periods.
Global decline in stroke care during pandemic
Results showed a global decline in the number of stroke patients admitted to the hospital as well as acute stroke treatments, such as thrombolysis, during the first wave of the COVID-19 pandemic. The researchers found that there were 91,373 stroke admissions in the 4 months immediately before the pandemic, compared with 80,894 admissions during the first 4 pandemic months, representing an 11.5% decline.
They also report that 13,334 stroke patients received intravenous thrombolysis in the 4 months preceding the pandemic, compared with 11,570 during the first 4 pandemic months, representing a 13.2% drop.
Interhospital transfers after thrombolysis for a higher level of stroke care decreased from 1,337 before the pandemic to 1,178 during the pandemic, a reduction of 11.9%.
There were greater declines in primary compared with comprehensive stroke centers for stroke hospitalizations (change, –17.3% vs. –10.3%) and for the number of patients receiving thrombolysis (change, –15.5% vs. –12.6%).
The volume of stroke hospitalizations increased by 9.5% in the two later pandemic months (May, June) versus the two earlier months (March, April), with greater recovery in hospitals with lower COVID-19 hospitalization volume, high-volume stroke centers, and comprehensive stroke centers.
Dr. Nguyen suggested that reasons for the reductions in these stroke numbers at the beginning of the pandemic could include a reduction in stroke risk due to a reduction of exposure to other viral infections or patients not presenting to the hospital for fear of contracting the coronavirus.
The higher recovery of stroke volume in high-volume stroke centers and comprehensive stroke centers may represent patients with higher needs – those having more severe strokes – seeking care more frequently than those with milder symptoms, she noted.
“Preserving access to stroke care and emergency stroke care amidst a pandemic is as important as educating patients on the importance of presenting to the hospital in the event of stroke-like symptoms,” Dr. Nguyen concluded.
“We continue to advocate that if a patient has stroke-like symptoms, such as loss of speech, strength, vision, or balance, it is important for the patient to seek medical care as an emergency, as there are treatments that can improve a patient’s ability to recover from disabling stroke in earlier rather than later time windows,” she added.
In the publication, the authors wrote, “Our results concur with other recent reports on the collateral effects of the COVID-19 pandemic on stroke systems of care,” but added that “this is among the first descriptions of the change at a global level, including primary and comprehensive stroke centers.”
They said that hospital access related to high COVID-19 burden was unlikely a factor because the decline was seen in centers with a few or no patients with COVID-19. They suggested that patient fear of contracting coronavirus may have played a role, along with a decrease in presentation of transient ischemic attacks, mild strokes, or moderate strokes, and physical distancing measures may have prevented the timely witnessing of a stroke.
A version of this article first appeared on Medscape.com.
One study showed a stroke rate of 2.2% among patients with COVID-19 admitted to intensive care in 52 different countries. Another found a stroke rate of 1.48% in patients hospitalized with COVID-19 from 70 different countries. These researchers also found a reduction in stroke presentations and stroke care during the pandemic.
Both studies will be presented at the American Academy of Neurology’s 2021 annual meeting.
“Stroke has been a known serious complication of COVID-19, with some studies reporting a higher-than-expected occurrence, especially in young people,” said coauthor of the intensive care study, Jonathon Fanning, MBBS, PhD, University of Queensland, Brisbane, Australia.
“However, among the sickest of COVID patients – those admitted to an ICU – our research found that stroke was not a common complication and that ischemic stroke did not increase the risk of death,” he added.
Hemorrhagic stroke more common?
In this study, researchers analyzed a database of 2,699 patients who were admitted to the intensive care unit with COVID-19 in 52 countries and found that 59 of these patients (2.2%) subsequently sustained a stroke.
Most of the strokes identified in this cohort were hemorrhagic (46%), with 32% being ischemic and 22% unspecified. Hemorrhagic stroke was associated with a fivefold increased risk for death compared with patients who did not have a stroke. Of those with a hemorrhagic stroke, 72% died, but only 15% died of the stroke. Rather, multiorgan failure was the leading cause of death.
There was no association between ischemic stroke and mortality.
“There is scarce research on new-onset stroke complicating ICU admissions, and many of the limitations of assessing stroke in ICU populations confound the true values and result in variability in reported incidence anywhere from a 1%-4% incidence,” Dr. Fanning said.
He noted that a large Korean study had shown a 1.2% rate of stroke in patients without COVID admitted to non-neurologic ICUs. “In light of this, I think this 2% is higher than we would expect in a general ICU population, but in the context of earlier reports of COVID-19–associated risk for stroke, this figure is actually somewhat reassuring,” Dr. Fanning said.
Asked how this study compared with the large American Heart Association study recently reported that showed an overall rate of ischemic stroke of 0.75%, Dr. Fanning said the two studies reported on different populations, which makes them difficult to compare.
“Our study specifically reports on new-onset stroke complicating ICU admission,” he noted. “The AHA study is a large study of all patients admitted to hospital, but both studies identified less than previous estimates of COVID-related stroke.”
Largest sample to date
The other study, which includes 119,967 COVID-19 hospitalizations and represents the largest sample reporting the concomitant diagnoses of stroke and SARS-CoV-2 infection to date, was presented at the AAN meeting by Thanh N. Nguyen, MD, a professor at Boston University.
This study has also been published online in Neurology, with first author Raul G. Nogueira, MD, Emory University, Atlanta.
In this international observational, retrospective study across 6 continents, 70 countries, and 457 stroke centers, there was a 1.48% stroke rate across 119,967 COVID-19 hospitalizations. SARS-CoV-2 infection was noted in 3.3% (1,722) of all stroke admissions, which numbered 52,026.
The researchers identified stroke diagnoses by the International Classification of Diseases, 10th revision, codes and/or classifications in stroke center databases, and rates of stroke hospitalizations and numbers of patients receiving thrombolysis were compared between the first 4 months of the pandemic (March to June 2020) compared with two control 4-month periods.
Global decline in stroke care during pandemic
Results showed a global decline in the number of stroke patients admitted to the hospital as well as acute stroke treatments, such as thrombolysis, during the first wave of the COVID-19 pandemic. The researchers found that there were 91,373 stroke admissions in the 4 months immediately before the pandemic, compared with 80,894 admissions during the first 4 pandemic months, representing an 11.5% decline.
They also report that 13,334 stroke patients received intravenous thrombolysis in the 4 months preceding the pandemic, compared with 11,570 during the first 4 pandemic months, representing a 13.2% drop.
Interhospital transfers after thrombolysis for a higher level of stroke care decreased from 1,337 before the pandemic to 1,178 during the pandemic, a reduction of 11.9%.
There were greater declines in primary compared with comprehensive stroke centers for stroke hospitalizations (change, –17.3% vs. –10.3%) and for the number of patients receiving thrombolysis (change, –15.5% vs. –12.6%).
The volume of stroke hospitalizations increased by 9.5% in the two later pandemic months (May, June) versus the two earlier months (March, April), with greater recovery in hospitals with lower COVID-19 hospitalization volume, high-volume stroke centers, and comprehensive stroke centers.
Dr. Nguyen suggested that reasons for the reductions in these stroke numbers at the beginning of the pandemic could include a reduction in stroke risk due to a reduction of exposure to other viral infections or patients not presenting to the hospital for fear of contracting the coronavirus.
The higher recovery of stroke volume in high-volume stroke centers and comprehensive stroke centers may represent patients with higher needs – those having more severe strokes – seeking care more frequently than those with milder symptoms, she noted.
“Preserving access to stroke care and emergency stroke care amidst a pandemic is as important as educating patients on the importance of presenting to the hospital in the event of stroke-like symptoms,” Dr. Nguyen concluded.
“We continue to advocate that if a patient has stroke-like symptoms, such as loss of speech, strength, vision, or balance, it is important for the patient to seek medical care as an emergency, as there are treatments that can improve a patient’s ability to recover from disabling stroke in earlier rather than later time windows,” she added.
In the publication, the authors wrote, “Our results concur with other recent reports on the collateral effects of the COVID-19 pandemic on stroke systems of care,” but added that “this is among the first descriptions of the change at a global level, including primary and comprehensive stroke centers.”
They said that hospital access related to high COVID-19 burden was unlikely a factor because the decline was seen in centers with a few or no patients with COVID-19. They suggested that patient fear of contracting coronavirus may have played a role, along with a decrease in presentation of transient ischemic attacks, mild strokes, or moderate strokes, and physical distancing measures may have prevented the timely witnessing of a stroke.
A version of this article first appeared on Medscape.com.
From AAN 2021
Ten reasons airborne transmission of SARS-CoV-2 appears airtight
The scientific evidence for airborne transmission of the SARS-CoV-2 virus from different researchers all point in the same direction – that infectious aerosols are the principal means of person-to-person transmission, according to experts.
Not that it’s without controversy.
The science backing aerosol transmission “is clear-cut, but it is not accepted in many circles,” Trisha Greenhalgh, PhD, said in an interview.
“In particular, some in the evidence-based medicine movement and some infectious diseases clinicians are remarkably resistant to the evidence,” added Dr. Greenhalgh, professor of primary care health sciences at the University of Oxford (England).
“It’s very hard to see why, since the evidence all stacks up,” Dr. Greenhalgh said.
“The scientific evidence on spread from both near-field and far-field aerosols has been clear since early on in the pandemic, but there was resistance to acknowledging this in some circles, including the medical journals,” Joseph G. Allen, DSc, MPH, told this news organization when asked to comment.
“This is the week the dam broke. Three new commentaries came out … in top medical journals – BMJ, The Lancet, JAMA – all making the same point that aerosols are the dominant mode of transmission,” added Dr. Allen, associate professor of exposure assessment science at the Harvard T.H. Chan School of Public Health in Boston.
Dr. Greenhalgh and colleagues point to an increase in COVID-19 cases in the aftermath of so-called “super-spreader” events, spread of SARS-CoV-2 to people across different hotel rooms, and the relatively lower transmission detected after outdoor events.
Top 10 reasons
They outlined 10 scientific reasons backing airborne transmission in a commentary published online April 15 in The Lancet:
- The dominance of airborne transmission is supported by long-range transmission observed at super-spreader events.
- Long-range transmission has been reported among rooms at COVID-19 quarantine hotels, settings where infected people never spent time in the same room.
- Asymptomatic individuals account for an estimated 33%-59% of SARS-CoV-2 transmission, and could be spreading the virus through speaking, which produces thousands of aerosol particles and few large droplets.
- Transmission outdoors and in well-ventilated indoor spaces is lower than in enclosed spaces.
- Nosocomial infections are reported in health care settings where protective measures address large droplets but not aerosols.
- Viable SARS-CoV-2 has been detected in the air of hospital rooms and in the car of an infected person.
- Investigators found SARS-CoV-2 in hospital air filters and building ducts.
- It’s not just humans – infected animals can infect animals in other cages connected only through an air duct.
- No strong evidence refutes airborne transmission, and contact tracing supports secondary transmission in crowded, poorly ventilated indoor spaces.
- Only limited evidence supports other means of SARS-CoV-2 transmission, including through fomites or large droplets.
“We thought we’d summarize [the evidence] to clarify the arguments for and against. We looked hard for evidence against but found none,” Dr. Greenhalgh said.
“Although other routes can contribute, we believe that the airborne route is likely to be dominant,” the authors note.
The evidence on airborne transmission was there very early on but the Centers for Disease Control and Prevention, World Health Organization, and others repeated the message that the primary concern was droplets and fomites.
Response to a review
The top 10 list is also part rebuttal of a systematic review funded by the WHO and published last month that points to inconclusive evidence for airborne transmission. The researchers involved with that review state that “the lack of recoverable viral culture samples of SARS-CoV-2 prevents firm conclusions to be drawn about airborne transmission.”
However, Dr. Greenhalgh and colleagues note that “this conclusion, and the wide circulation of the review’s findings, is concerning because of the public health implications.”
The current authors also argue that enough evidence already exists on airborne transmission. “Policy should change. We don’t need more research on this topic; we need different policy,” Dr. Greenhalgh said. “We need ventilation front and center, air filtration when necessary, and better-fitting masks worn whenever indoors.”
Dr. Allen agreed that guidance hasn’t always kept pace with the science. “With all of the new evidence accumulated on airborne transmission since last winter, there is still widespread confusion in the public about modes of transmission,” he said. Dr. Allen also serves as commissioner of The Lancet COVID-19 Commission and is chair of the commission’s Task Force on Safe Work, Safe Schools, and Safe Travel.
“It was only just last week that CDC pulled back on guidance on ‘deep cleaning’ and in its place correctly said that the risk from touching surfaces is low,” he added. “The science has been clear on this for over a year, but official guidance was only recently updated.”
As a result, many companies and organizations continued to focus on “hygiene theatre,” Dr. Allen said, “wasting resources on overcleaning surfaces. Unbelievably, many schools still close for an entire day each week for deep cleaning and some still quarantine library books. The message that shared air is the problem, not shared surfaces, is a message that still needs to be reinforced.”
The National Institute for Health Research, Economic and Social Research Council, and Wellcome support Dr. Greenhalgh’s research. Dr. Greenhalgh and Dr. Allen had no relevant financial relationships to disclose.
A version of this article first appeared on Medscape.com.
The scientific evidence for airborne transmission of the SARS-CoV-2 virus from different researchers all point in the same direction – that infectious aerosols are the principal means of person-to-person transmission, according to experts.
Not that it’s without controversy.
The science backing aerosol transmission “is clear-cut, but it is not accepted in many circles,” Trisha Greenhalgh, PhD, said in an interview.
“In particular, some in the evidence-based medicine movement and some infectious diseases clinicians are remarkably resistant to the evidence,” added Dr. Greenhalgh, professor of primary care health sciences at the University of Oxford (England).
“It’s very hard to see why, since the evidence all stacks up,” Dr. Greenhalgh said.
“The scientific evidence on spread from both near-field and far-field aerosols has been clear since early on in the pandemic, but there was resistance to acknowledging this in some circles, including the medical journals,” Joseph G. Allen, DSc, MPH, told this news organization when asked to comment.
“This is the week the dam broke. Three new commentaries came out … in top medical journals – BMJ, The Lancet, JAMA – all making the same point that aerosols are the dominant mode of transmission,” added Dr. Allen, associate professor of exposure assessment science at the Harvard T.H. Chan School of Public Health in Boston.
Dr. Greenhalgh and colleagues point to an increase in COVID-19 cases in the aftermath of so-called “super-spreader” events, spread of SARS-CoV-2 to people across different hotel rooms, and the relatively lower transmission detected after outdoor events.
Top 10 reasons
They outlined 10 scientific reasons backing airborne transmission in a commentary published online April 15 in The Lancet:
- The dominance of airborne transmission is supported by long-range transmission observed at super-spreader events.
- Long-range transmission has been reported among rooms at COVID-19 quarantine hotels, settings where infected people never spent time in the same room.
- Asymptomatic individuals account for an estimated 33%-59% of SARS-CoV-2 transmission, and could be spreading the virus through speaking, which produces thousands of aerosol particles and few large droplets.
- Transmission outdoors and in well-ventilated indoor spaces is lower than in enclosed spaces.
- Nosocomial infections are reported in health care settings where protective measures address large droplets but not aerosols.
- Viable SARS-CoV-2 has been detected in the air of hospital rooms and in the car of an infected person.
- Investigators found SARS-CoV-2 in hospital air filters and building ducts.
- It’s not just humans – infected animals can infect animals in other cages connected only through an air duct.
- No strong evidence refutes airborne transmission, and contact tracing supports secondary transmission in crowded, poorly ventilated indoor spaces.
- Only limited evidence supports other means of SARS-CoV-2 transmission, including through fomites or large droplets.
“We thought we’d summarize [the evidence] to clarify the arguments for and against. We looked hard for evidence against but found none,” Dr. Greenhalgh said.
“Although other routes can contribute, we believe that the airborne route is likely to be dominant,” the authors note.
The evidence on airborne transmission was there very early on but the Centers for Disease Control and Prevention, World Health Organization, and others repeated the message that the primary concern was droplets and fomites.
Response to a review
The top 10 list is also part rebuttal of a systematic review funded by the WHO and published last month that points to inconclusive evidence for airborne transmission. The researchers involved with that review state that “the lack of recoverable viral culture samples of SARS-CoV-2 prevents firm conclusions to be drawn about airborne transmission.”
However, Dr. Greenhalgh and colleagues note that “this conclusion, and the wide circulation of the review’s findings, is concerning because of the public health implications.”
The current authors also argue that enough evidence already exists on airborne transmission. “Policy should change. We don’t need more research on this topic; we need different policy,” Dr. Greenhalgh said. “We need ventilation front and center, air filtration when necessary, and better-fitting masks worn whenever indoors.”
Dr. Allen agreed that guidance hasn’t always kept pace with the science. “With all of the new evidence accumulated on airborne transmission since last winter, there is still widespread confusion in the public about modes of transmission,” he said. Dr. Allen also serves as commissioner of The Lancet COVID-19 Commission and is chair of the commission’s Task Force on Safe Work, Safe Schools, and Safe Travel.
“It was only just last week that CDC pulled back on guidance on ‘deep cleaning’ and in its place correctly said that the risk from touching surfaces is low,” he added. “The science has been clear on this for over a year, but official guidance was only recently updated.”
As a result, many companies and organizations continued to focus on “hygiene theatre,” Dr. Allen said, “wasting resources on overcleaning surfaces. Unbelievably, many schools still close for an entire day each week for deep cleaning and some still quarantine library books. The message that shared air is the problem, not shared surfaces, is a message that still needs to be reinforced.”
The National Institute for Health Research, Economic and Social Research Council, and Wellcome support Dr. Greenhalgh’s research. Dr. Greenhalgh and Dr. Allen had no relevant financial relationships to disclose.
A version of this article first appeared on Medscape.com.
The scientific evidence for airborne transmission of the SARS-CoV-2 virus from different researchers all point in the same direction – that infectious aerosols are the principal means of person-to-person transmission, according to experts.
Not that it’s without controversy.
The science backing aerosol transmission “is clear-cut, but it is not accepted in many circles,” Trisha Greenhalgh, PhD, said in an interview.
“In particular, some in the evidence-based medicine movement and some infectious diseases clinicians are remarkably resistant to the evidence,” added Dr. Greenhalgh, professor of primary care health sciences at the University of Oxford (England).
“It’s very hard to see why, since the evidence all stacks up,” Dr. Greenhalgh said.
“The scientific evidence on spread from both near-field and far-field aerosols has been clear since early on in the pandemic, but there was resistance to acknowledging this in some circles, including the medical journals,” Joseph G. Allen, DSc, MPH, told this news organization when asked to comment.
“This is the week the dam broke. Three new commentaries came out … in top medical journals – BMJ, The Lancet, JAMA – all making the same point that aerosols are the dominant mode of transmission,” added Dr. Allen, associate professor of exposure assessment science at the Harvard T.H. Chan School of Public Health in Boston.
Dr. Greenhalgh and colleagues point to an increase in COVID-19 cases in the aftermath of so-called “super-spreader” events, spread of SARS-CoV-2 to people across different hotel rooms, and the relatively lower transmission detected after outdoor events.
Top 10 reasons
They outlined 10 scientific reasons backing airborne transmission in a commentary published online April 15 in The Lancet:
- The dominance of airborne transmission is supported by long-range transmission observed at super-spreader events.
- Long-range transmission has been reported among rooms at COVID-19 quarantine hotels, settings where infected people never spent time in the same room.
- Asymptomatic individuals account for an estimated 33%-59% of SARS-CoV-2 transmission, and could be spreading the virus through speaking, which produces thousands of aerosol particles and few large droplets.
- Transmission outdoors and in well-ventilated indoor spaces is lower than in enclosed spaces.
- Nosocomial infections are reported in health care settings where protective measures address large droplets but not aerosols.
- Viable SARS-CoV-2 has been detected in the air of hospital rooms and in the car of an infected person.
- Investigators found SARS-CoV-2 in hospital air filters and building ducts.
- It’s not just humans – infected animals can infect animals in other cages connected only through an air duct.
- No strong evidence refutes airborne transmission, and contact tracing supports secondary transmission in crowded, poorly ventilated indoor spaces.
- Only limited evidence supports other means of SARS-CoV-2 transmission, including through fomites or large droplets.
“We thought we’d summarize [the evidence] to clarify the arguments for and against. We looked hard for evidence against but found none,” Dr. Greenhalgh said.
“Although other routes can contribute, we believe that the airborne route is likely to be dominant,” the authors note.
The evidence on airborne transmission was there very early on but the Centers for Disease Control and Prevention, World Health Organization, and others repeated the message that the primary concern was droplets and fomites.
Response to a review
The top 10 list is also part rebuttal of a systematic review funded by the WHO and published last month that points to inconclusive evidence for airborne transmission. The researchers involved with that review state that “the lack of recoverable viral culture samples of SARS-CoV-2 prevents firm conclusions to be drawn about airborne transmission.”
However, Dr. Greenhalgh and colleagues note that “this conclusion, and the wide circulation of the review’s findings, is concerning because of the public health implications.”
The current authors also argue that enough evidence already exists on airborne transmission. “Policy should change. We don’t need more research on this topic; we need different policy,” Dr. Greenhalgh said. “We need ventilation front and center, air filtration when necessary, and better-fitting masks worn whenever indoors.”
Dr. Allen agreed that guidance hasn’t always kept pace with the science. “With all of the new evidence accumulated on airborne transmission since last winter, there is still widespread confusion in the public about modes of transmission,” he said. Dr. Allen also serves as commissioner of The Lancet COVID-19 Commission and is chair of the commission’s Task Force on Safe Work, Safe Schools, and Safe Travel.
“It was only just last week that CDC pulled back on guidance on ‘deep cleaning’ and in its place correctly said that the risk from touching surfaces is low,” he added. “The science has been clear on this for over a year, but official guidance was only recently updated.”
As a result, many companies and organizations continued to focus on “hygiene theatre,” Dr. Allen said, “wasting resources on overcleaning surfaces. Unbelievably, many schools still close for an entire day each week for deep cleaning and some still quarantine library books. The message that shared air is the problem, not shared surfaces, is a message that still needs to be reinforced.”
The National Institute for Health Research, Economic and Social Research Council, and Wellcome support Dr. Greenhalgh’s research. Dr. Greenhalgh and Dr. Allen had no relevant financial relationships to disclose.
A version of this article first appeared on Medscape.com.
Study shows potential of remote Parkinson’s disease genotyping
according to a pilot study sponsored by the Parkinson’s Foundation.
“Overall we found high levels of participant satisfaction with Parkinson’s testing and genetic counseling and no significant difference in outcomes concerning satisfaction, knowledge, and impact of genetic testing between disclosure of results and genetic counseling in-person by either a neurologist or a genetic counselor or via telephone by a remove genetic counselor at a centralized center,” said Jennifer L. Verbrugge, MS, a genetic counselor at Indiana University, Indianapolis, in reporting results of the PD GENEration pilot study, presented at the 2021 annual meeting of the American Academy of Neurology.
COVID complication
The study launched in the summer of 2019 with the goal of enrolling 600 participants. However, because of the COVID-19 pandemic, enrollment was truncated. The pilot study eventually enrolled 289 patients, 205 of whom returned their postgenetic counseling surveys, Ms. Verbrugge said. The pilot study goal was to evaluate the feasibility and impact of in-person versus remote genetic testing and counseling for people with Parkinson’s disease.
“The study hopes to reach its ultimate goal, which is to deliver Parkinson’s disease–related genetic testing and counseling to upward of 15,000 people with Parkinson’s,” Ms. Verbrugge said. The program is also planning to expand to include Spanish speakers.
In the pilot study, genetic results were positive in 17% of patients, with 15% (n = 42) having positive heterozygous variants and 8% having variants of uncertain significance. “We did not see significant differences in these outcomes when we compared the mode and genetic counselors involved,” Ms. Verbrugge said.
The study did find that in-person testing and counseling “was associated with increased participant feelings that they were partners in care,” Ms. Verbrugge added. “This is something we are going to continue to evaluate as time goes on.”
However, as the COVID-19 pandemic pushed clinicians to develop virtual platforms, it resulted in a function through which participants can complete all genetic study activities remotely, she said. The study organizers anticipate that as pandemic restrictions ease, they will be able to reach their original goal of 600 participants along with those recruited in an expansion phase.
“As restrictions related to the pandemic ease, we anticipate that more Parkinson’s disease gene-targeted clinical trials will emerge, with aims to recruit people who carry certain gene variants,” Ms. Verbrugge said in an interview. “Many people with Parkinson’s disease may therefore benefit from genetic testing and learning if they carry a Parkinson’s disease related gene variant from participation in the PD GENEration study.”
Increasing patient access
To scale up to the 15,000-population goal for the program, PD GENEration has launched a new protocol designed to increase patient access at new study sites, Ms. Verbrugge said. “This protocol includes an abbreviate version of the clinical data collected, while it maintains the critical component of genetic counseling in the testing process.”
Going forward, the PD GENEration study will focus on improving access to genetic testing and counseling in underrepresented and rural populations, Ms. Verbrugge said. “These efforts will also generate valuable genomic data, allowing researchers to learn more about the causes of Parkinson’s disease in diverse and underrepresented populations. The study will be expanding research efforts concerning the genomic data to gain insights about the seven key genes studied as well as new genes linked to Parkinson’s disease.”
The work of the PD GENEration study is timely, said David K. Simon, MD, PhD, of Harvard Medical School and director of the Parkinson’s Disease & Movement Disorders Center at Beth Israel Deaconess Medical Center, both in Boston. “This is very important to identify such patients now, as clinical trials targeting people with specific genetic mutations or variants are coming soon, and in some cases already are underway. The feasibility and speed of enrollment for those trials will be greatly facilitate if we know in advance who are the people with Parkinson’s disease who have mutations that would make them eligible for the particular trials.”
The fact that the study provides free genetic testing to people with Parkinson’s disease isn’t to be overlooked. “This was an important study to address the question of whether or not remote genetic counseling was feasible and effective, and the results are meaningful given the randomized prospective design,” Dr. Simon said.
Ms. Verbrugge has no relevant relationships to disclose. Dr. Simon reports receiving research funding from the Parkinson’s Foundation.
according to a pilot study sponsored by the Parkinson’s Foundation.
“Overall we found high levels of participant satisfaction with Parkinson’s testing and genetic counseling and no significant difference in outcomes concerning satisfaction, knowledge, and impact of genetic testing between disclosure of results and genetic counseling in-person by either a neurologist or a genetic counselor or via telephone by a remove genetic counselor at a centralized center,” said Jennifer L. Verbrugge, MS, a genetic counselor at Indiana University, Indianapolis, in reporting results of the PD GENEration pilot study, presented at the 2021 annual meeting of the American Academy of Neurology.
COVID complication
The study launched in the summer of 2019 with the goal of enrolling 600 participants. However, because of the COVID-19 pandemic, enrollment was truncated. The pilot study eventually enrolled 289 patients, 205 of whom returned their postgenetic counseling surveys, Ms. Verbrugge said. The pilot study goal was to evaluate the feasibility and impact of in-person versus remote genetic testing and counseling for people with Parkinson’s disease.
“The study hopes to reach its ultimate goal, which is to deliver Parkinson’s disease–related genetic testing and counseling to upward of 15,000 people with Parkinson’s,” Ms. Verbrugge said. The program is also planning to expand to include Spanish speakers.
In the pilot study, genetic results were positive in 17% of patients, with 15% (n = 42) having positive heterozygous variants and 8% having variants of uncertain significance. “We did not see significant differences in these outcomes when we compared the mode and genetic counselors involved,” Ms. Verbrugge said.
The study did find that in-person testing and counseling “was associated with increased participant feelings that they were partners in care,” Ms. Verbrugge added. “This is something we are going to continue to evaluate as time goes on.”
However, as the COVID-19 pandemic pushed clinicians to develop virtual platforms, it resulted in a function through which participants can complete all genetic study activities remotely, she said. The study organizers anticipate that as pandemic restrictions ease, they will be able to reach their original goal of 600 participants along with those recruited in an expansion phase.
“As restrictions related to the pandemic ease, we anticipate that more Parkinson’s disease gene-targeted clinical trials will emerge, with aims to recruit people who carry certain gene variants,” Ms. Verbrugge said in an interview. “Many people with Parkinson’s disease may therefore benefit from genetic testing and learning if they carry a Parkinson’s disease related gene variant from participation in the PD GENEration study.”
Increasing patient access
To scale up to the 15,000-population goal for the program, PD GENEration has launched a new protocol designed to increase patient access at new study sites, Ms. Verbrugge said. “This protocol includes an abbreviate version of the clinical data collected, while it maintains the critical component of genetic counseling in the testing process.”
Going forward, the PD GENEration study will focus on improving access to genetic testing and counseling in underrepresented and rural populations, Ms. Verbrugge said. “These efforts will also generate valuable genomic data, allowing researchers to learn more about the causes of Parkinson’s disease in diverse and underrepresented populations. The study will be expanding research efforts concerning the genomic data to gain insights about the seven key genes studied as well as new genes linked to Parkinson’s disease.”
The work of the PD GENEration study is timely, said David K. Simon, MD, PhD, of Harvard Medical School and director of the Parkinson’s Disease & Movement Disorders Center at Beth Israel Deaconess Medical Center, both in Boston. “This is very important to identify such patients now, as clinical trials targeting people with specific genetic mutations or variants are coming soon, and in some cases already are underway. The feasibility and speed of enrollment for those trials will be greatly facilitate if we know in advance who are the people with Parkinson’s disease who have mutations that would make them eligible for the particular trials.”
The fact that the study provides free genetic testing to people with Parkinson’s disease isn’t to be overlooked. “This was an important study to address the question of whether or not remote genetic counseling was feasible and effective, and the results are meaningful given the randomized prospective design,” Dr. Simon said.
Ms. Verbrugge has no relevant relationships to disclose. Dr. Simon reports receiving research funding from the Parkinson’s Foundation.
according to a pilot study sponsored by the Parkinson’s Foundation.
“Overall we found high levels of participant satisfaction with Parkinson’s testing and genetic counseling and no significant difference in outcomes concerning satisfaction, knowledge, and impact of genetic testing between disclosure of results and genetic counseling in-person by either a neurologist or a genetic counselor or via telephone by a remove genetic counselor at a centralized center,” said Jennifer L. Verbrugge, MS, a genetic counselor at Indiana University, Indianapolis, in reporting results of the PD GENEration pilot study, presented at the 2021 annual meeting of the American Academy of Neurology.
COVID complication
The study launched in the summer of 2019 with the goal of enrolling 600 participants. However, because of the COVID-19 pandemic, enrollment was truncated. The pilot study eventually enrolled 289 patients, 205 of whom returned their postgenetic counseling surveys, Ms. Verbrugge said. The pilot study goal was to evaluate the feasibility and impact of in-person versus remote genetic testing and counseling for people with Parkinson’s disease.
“The study hopes to reach its ultimate goal, which is to deliver Parkinson’s disease–related genetic testing and counseling to upward of 15,000 people with Parkinson’s,” Ms. Verbrugge said. The program is also planning to expand to include Spanish speakers.
In the pilot study, genetic results were positive in 17% of patients, with 15% (n = 42) having positive heterozygous variants and 8% having variants of uncertain significance. “We did not see significant differences in these outcomes when we compared the mode and genetic counselors involved,” Ms. Verbrugge said.
The study did find that in-person testing and counseling “was associated with increased participant feelings that they were partners in care,” Ms. Verbrugge added. “This is something we are going to continue to evaluate as time goes on.”
However, as the COVID-19 pandemic pushed clinicians to develop virtual platforms, it resulted in a function through which participants can complete all genetic study activities remotely, she said. The study organizers anticipate that as pandemic restrictions ease, they will be able to reach their original goal of 600 participants along with those recruited in an expansion phase.
“As restrictions related to the pandemic ease, we anticipate that more Parkinson’s disease gene-targeted clinical trials will emerge, with aims to recruit people who carry certain gene variants,” Ms. Verbrugge said in an interview. “Many people with Parkinson’s disease may therefore benefit from genetic testing and learning if they carry a Parkinson’s disease related gene variant from participation in the PD GENEration study.”
Increasing patient access
To scale up to the 15,000-population goal for the program, PD GENEration has launched a new protocol designed to increase patient access at new study sites, Ms. Verbrugge said. “This protocol includes an abbreviate version of the clinical data collected, while it maintains the critical component of genetic counseling in the testing process.”
Going forward, the PD GENEration study will focus on improving access to genetic testing and counseling in underrepresented and rural populations, Ms. Verbrugge said. “These efforts will also generate valuable genomic data, allowing researchers to learn more about the causes of Parkinson’s disease in diverse and underrepresented populations. The study will be expanding research efforts concerning the genomic data to gain insights about the seven key genes studied as well as new genes linked to Parkinson’s disease.”
The work of the PD GENEration study is timely, said David K. Simon, MD, PhD, of Harvard Medical School and director of the Parkinson’s Disease & Movement Disorders Center at Beth Israel Deaconess Medical Center, both in Boston. “This is very important to identify such patients now, as clinical trials targeting people with specific genetic mutations or variants are coming soon, and in some cases already are underway. The feasibility and speed of enrollment for those trials will be greatly facilitate if we know in advance who are the people with Parkinson’s disease who have mutations that would make them eligible for the particular trials.”
The fact that the study provides free genetic testing to people with Parkinson’s disease isn’t to be overlooked. “This was an important study to address the question of whether or not remote genetic counseling was feasible and effective, and the results are meaningful given the randomized prospective design,” Dr. Simon said.
Ms. Verbrugge has no relevant relationships to disclose. Dr. Simon reports receiving research funding from the Parkinson’s Foundation.
FROM AAN 2021
The ripple effect
“I want my life to mean something.”
She was young, energetic, and idealistic. She was seeing me for her migraines, but our conversation had turned to her applying for medical school.
“I want my life to mean something.”
I reflected on that later. I once said similar things, but now found myself wondering, two-thirds of the way through my career, did it?
The world certainly isn’t safer, geopolitically or environmentally, now than it was when I left medical school. Hell, the idea that there’d actually be another worldwide pandemic was pretty much beyond me then. That seemed so 1918.
I can’t even say I’ve made a huge difference in medicine. I’m not on the front line of research, inventing cures and tests that will change what we do.
I’m certainly far removed from the front lines of the pandemic.
But realistically, none of those things were ever really my goal, either.
“I want my life to mean something.”
Sometimes it’s hard to think I’ve made a difference. Day in and day out I’m at my office, quietly sitting behind a desk and trying to look smart. For all good intentions ... at some point it’s just a job.
Then I realized that the job isn’t about me. It’s about her, and the many other people who come to me for help. The real meaning is the impact on their lives.
Anytime we see a patient and make their lives better, either through treatment or compassion, it creates a ripple effect. It helps their family, friends, and coworkers. Whether we’re actually giving help or just understanding.
It might even inspire one of them to go into medicine, because my generation will be drifting toward retirement in the next 10 years.
Our care may set off a chain reaction we can’t see. Perhaps the patient you help will return to work and initiate some action that will bring a marked benefit to us all. Or someone in their circle, freed or inspired by their improvement, will bring about such a change.
And that’s meaning enough for me.
Dr. Block has a solo neurology practice in Scottsdale, Ariz.
“I want my life to mean something.”
She was young, energetic, and idealistic. She was seeing me for her migraines, but our conversation had turned to her applying for medical school.
“I want my life to mean something.”
I reflected on that later. I once said similar things, but now found myself wondering, two-thirds of the way through my career, did it?
The world certainly isn’t safer, geopolitically or environmentally, now than it was when I left medical school. Hell, the idea that there’d actually be another worldwide pandemic was pretty much beyond me then. That seemed so 1918.
I can’t even say I’ve made a huge difference in medicine. I’m not on the front line of research, inventing cures and tests that will change what we do.
I’m certainly far removed from the front lines of the pandemic.
But realistically, none of those things were ever really my goal, either.
“I want my life to mean something.”
Sometimes it’s hard to think I’ve made a difference. Day in and day out I’m at my office, quietly sitting behind a desk and trying to look smart. For all good intentions ... at some point it’s just a job.
Then I realized that the job isn’t about me. It’s about her, and the many other people who come to me for help. The real meaning is the impact on their lives.
Anytime we see a patient and make their lives better, either through treatment or compassion, it creates a ripple effect. It helps their family, friends, and coworkers. Whether we’re actually giving help or just understanding.
It might even inspire one of them to go into medicine, because my generation will be drifting toward retirement in the next 10 years.
Our care may set off a chain reaction we can’t see. Perhaps the patient you help will return to work and initiate some action that will bring a marked benefit to us all. Or someone in their circle, freed or inspired by their improvement, will bring about such a change.
And that’s meaning enough for me.
Dr. Block has a solo neurology practice in Scottsdale, Ariz.
“I want my life to mean something.”
She was young, energetic, and idealistic. She was seeing me for her migraines, but our conversation had turned to her applying for medical school.
“I want my life to mean something.”
I reflected on that later. I once said similar things, but now found myself wondering, two-thirds of the way through my career, did it?
The world certainly isn’t safer, geopolitically or environmentally, now than it was when I left medical school. Hell, the idea that there’d actually be another worldwide pandemic was pretty much beyond me then. That seemed so 1918.
I can’t even say I’ve made a huge difference in medicine. I’m not on the front line of research, inventing cures and tests that will change what we do.
I’m certainly far removed from the front lines of the pandemic.
But realistically, none of those things were ever really my goal, either.
“I want my life to mean something.”
Sometimes it’s hard to think I’ve made a difference. Day in and day out I’m at my office, quietly sitting behind a desk and trying to look smart. For all good intentions ... at some point it’s just a job.
Then I realized that the job isn’t about me. It’s about her, and the many other people who come to me for help. The real meaning is the impact on their lives.
Anytime we see a patient and make their lives better, either through treatment or compassion, it creates a ripple effect. It helps their family, friends, and coworkers. Whether we’re actually giving help or just understanding.
It might even inspire one of them to go into medicine, because my generation will be drifting toward retirement in the next 10 years.
Our care may set off a chain reaction we can’t see. Perhaps the patient you help will return to work and initiate some action that will bring a marked benefit to us all. Or someone in their circle, freed or inspired by their improvement, will bring about such a change.
And that’s meaning enough for me.
Dr. Block has a solo neurology practice in Scottsdale, Ariz.
Rimegepant looks safe in migraine patients with cardiovascular risk
Results from a 1-year, open-label safety study suggest that Patients who fall into this category may be ineligible for treatment with triptans.
There are mechanistic concerns with rimegepant and related CGRP receptor antagonists. They block CGRP’s effect in the central nervous system, but CGRP is also active in blood vessels and the heart, leading to the possibility that countering its vasodilating effect could expose organs to risk of ischemia.
The Food and Drug Administration approved rimegepant in 2020 for treatment of acute migraine attacks. Sponsor Biohaven is also seeking approval for migraine prevention after a successful phase 3 study published January 2021 in The Lancet.
Susan Hutchinson, MD, who is a headache specialist at Orange County Migraine & Headache Center in Irvine, Calif., presented the results at the American Academy of Neurology’s 2021 annual meeting. The open-label study suggested that rimegepant is generally safe. “The proportion of subjects reporting at least one adverse event was similar among subjects whether they had zero, one, or two or more cardiovascular disease risk factors, and also among those with low and moderate to high 10-year cardiovascular risk, as determined by the Framingham Risk Score,” said Dr. Hutchinson during her presentation.
Still, there was one concerning case: A 53-year-old man experienced an attack of angina. But he already had angina prior to the study, was being treated for hypercholesterolemia, and had current or former exposure to statins. “This adverse event was deemed by the investigator to not be related to rimegepant,” said Dr. Hutchinson.
During the following question-and-answer session, an attendee pressed Dr. Hutchinson about the case, and she admitted to some initial doubts. “That was my concern when I saw those slides. I’m like, ‘oh, my goodness.’ ” She clarified that the man’s angina history dated to 2016, which was several years before the trial, and the episode of angina occurred 7 months after the first dose of rimegepant. “He was treated with nitroglycerin and taken out of the trial,” said Dr. Hutchinson.
Proper patient selection is key
The research adds to the literature on rimegepant by providing data on multiple uses, as opposed to the phase 3 study, which only looked at single use, according to Olivia Begasse de Dhaem, MD, who is a neurology attending physician at Stamford (Conn.) Health and was the session moderator. Rimegepant and other oral CGRP receptor antagonists, including the FDA-approved ubrogepant and the investigative drug atogepant, will help fill the gap of patients who don’t tolerate or are ineligible for triptans, she said.
Dr. Begasse de Dhaem pointed out that patient selection remains important. “I think the main thing for patient care is to look at whether the patient we are treating would fit within the inclusion criteria, or would have been excluded from this study,” said Dr. Begasse de Dhaem. Specifically, according to its clinicaltrials.gov page, the trial excluded patients with hemiplegic and basilar migraine, as well as patients with uncontrolled, unstable, or recently diagnosed cardiovascular disease, those with a body mass index of 30 kg/m2 or higher, and hemoglobin A1c levels of 6.5% or higher. “This also looked at people with less than 15 migraine days per month, so it’s limited in how much we can extrapolate to people with chronic migraine who may take more than 7.7 rimegepant [doses, the mean value taken by trial participants] per month,” Dr. Begasse de Dhaem added.
She also applauded the inclusion of older patients in the study, noting that most migraine studies have an upper age limit.
The study included subjects who experienced 2-14 moderate or severe migraine attacks per month, and they were allowed to take other migraine medications. Cardiovascular risk factors did not prevent entry to the trial and, like the previous pivotal trial, the long-term safety study admitted subjects older than 65. Among the study cohort, 1,514 participants were told to treat migraine pain of any intensity with 75 mg rimegepant up to once per day on an as-needed basis (PRN), and a second group of 286 were told to take 75 mg rimegepant every other day for 12 weeks, along with PRN dosing on nonscheduled treatment days.
Nearly 90% of subjects were female, the mean age was 43.1 years, and 3.7% were age 65 or older. Among the study participants, 40.8% had cardiovascular risk factors, including 28.8% with one risk factor, and 12.1% with two or more. About 7% had a moderate to high (≥10%) 10-year cardiovascular risk by Framingham Risk Score, 23.6% had a family history of coronary artery disease, 11.7% were being treated for hypertension, 10.4% smoked, 8.3% were being treated with a statin, and 3.0% had a history of diabetes.
In total, subjects were exposed to 112,014 doses of rimegepant, a mean of 7.7 doses per 4-week period. The exposure was similar across all risk groups, which included zero risk factors, one risk factor, and two or more risk factors; FRS of less than 10%; and FRS of 10% or greater. The most common adverse events were upper respiratory tract infection (8.8%), nasopharyngitis (6.8%), and sinusitis (5.1%). The frequency of one or more adverse events was similar among those with zero cardiovascular risk factors (59.6%), one risk factor (61.4%), two or more risk factors (62.2%), FRS less than 10% (59.9%), and FRS of 10% or greater (59.9%).
The study was funded by Biohaven Pharmaceuticals. Dr. Hutchinson has been a consultant or advisory board member for Biohaven, Alder, Allergan, Amgen, Avanir, electroCore, Lilly, Novartis, Promius, Supernus, and Teva. She has been on the speaker’s bureau for Allergan, Amgen, Avanir, electroCore, Lilly, Novartis, Promius, Supernus, and Teva. Dr. Begasse de Dhaem has no relevant financial disclosures.
Results from a 1-year, open-label safety study suggest that Patients who fall into this category may be ineligible for treatment with triptans.
There are mechanistic concerns with rimegepant and related CGRP receptor antagonists. They block CGRP’s effect in the central nervous system, but CGRP is also active in blood vessels and the heart, leading to the possibility that countering its vasodilating effect could expose organs to risk of ischemia.
The Food and Drug Administration approved rimegepant in 2020 for treatment of acute migraine attacks. Sponsor Biohaven is also seeking approval for migraine prevention after a successful phase 3 study published January 2021 in The Lancet.
Susan Hutchinson, MD, who is a headache specialist at Orange County Migraine & Headache Center in Irvine, Calif., presented the results at the American Academy of Neurology’s 2021 annual meeting. The open-label study suggested that rimegepant is generally safe. “The proportion of subjects reporting at least one adverse event was similar among subjects whether they had zero, one, or two or more cardiovascular disease risk factors, and also among those with low and moderate to high 10-year cardiovascular risk, as determined by the Framingham Risk Score,” said Dr. Hutchinson during her presentation.
Still, there was one concerning case: A 53-year-old man experienced an attack of angina. But he already had angina prior to the study, was being treated for hypercholesterolemia, and had current or former exposure to statins. “This adverse event was deemed by the investigator to not be related to rimegepant,” said Dr. Hutchinson.
During the following question-and-answer session, an attendee pressed Dr. Hutchinson about the case, and she admitted to some initial doubts. “That was my concern when I saw those slides. I’m like, ‘oh, my goodness.’ ” She clarified that the man’s angina history dated to 2016, which was several years before the trial, and the episode of angina occurred 7 months after the first dose of rimegepant. “He was treated with nitroglycerin and taken out of the trial,” said Dr. Hutchinson.
Proper patient selection is key
The research adds to the literature on rimegepant by providing data on multiple uses, as opposed to the phase 3 study, which only looked at single use, according to Olivia Begasse de Dhaem, MD, who is a neurology attending physician at Stamford (Conn.) Health and was the session moderator. Rimegepant and other oral CGRP receptor antagonists, including the FDA-approved ubrogepant and the investigative drug atogepant, will help fill the gap of patients who don’t tolerate or are ineligible for triptans, she said.
Dr. Begasse de Dhaem pointed out that patient selection remains important. “I think the main thing for patient care is to look at whether the patient we are treating would fit within the inclusion criteria, or would have been excluded from this study,” said Dr. Begasse de Dhaem. Specifically, according to its clinicaltrials.gov page, the trial excluded patients with hemiplegic and basilar migraine, as well as patients with uncontrolled, unstable, or recently diagnosed cardiovascular disease, those with a body mass index of 30 kg/m2 or higher, and hemoglobin A1c levels of 6.5% or higher. “This also looked at people with less than 15 migraine days per month, so it’s limited in how much we can extrapolate to people with chronic migraine who may take more than 7.7 rimegepant [doses, the mean value taken by trial participants] per month,” Dr. Begasse de Dhaem added.
She also applauded the inclusion of older patients in the study, noting that most migraine studies have an upper age limit.
The study included subjects who experienced 2-14 moderate or severe migraine attacks per month, and they were allowed to take other migraine medications. Cardiovascular risk factors did not prevent entry to the trial and, like the previous pivotal trial, the long-term safety study admitted subjects older than 65. Among the study cohort, 1,514 participants were told to treat migraine pain of any intensity with 75 mg rimegepant up to once per day on an as-needed basis (PRN), and a second group of 286 were told to take 75 mg rimegepant every other day for 12 weeks, along with PRN dosing on nonscheduled treatment days.
Nearly 90% of subjects were female, the mean age was 43.1 years, and 3.7% were age 65 or older. Among the study participants, 40.8% had cardiovascular risk factors, including 28.8% with one risk factor, and 12.1% with two or more. About 7% had a moderate to high (≥10%) 10-year cardiovascular risk by Framingham Risk Score, 23.6% had a family history of coronary artery disease, 11.7% were being treated for hypertension, 10.4% smoked, 8.3% were being treated with a statin, and 3.0% had a history of diabetes.
In total, subjects were exposed to 112,014 doses of rimegepant, a mean of 7.7 doses per 4-week period. The exposure was similar across all risk groups, which included zero risk factors, one risk factor, and two or more risk factors; FRS of less than 10%; and FRS of 10% or greater. The most common adverse events were upper respiratory tract infection (8.8%), nasopharyngitis (6.8%), and sinusitis (5.1%). The frequency of one or more adverse events was similar among those with zero cardiovascular risk factors (59.6%), one risk factor (61.4%), two or more risk factors (62.2%), FRS less than 10% (59.9%), and FRS of 10% or greater (59.9%).
The study was funded by Biohaven Pharmaceuticals. Dr. Hutchinson has been a consultant or advisory board member for Biohaven, Alder, Allergan, Amgen, Avanir, electroCore, Lilly, Novartis, Promius, Supernus, and Teva. She has been on the speaker’s bureau for Allergan, Amgen, Avanir, electroCore, Lilly, Novartis, Promius, Supernus, and Teva. Dr. Begasse de Dhaem has no relevant financial disclosures.
Results from a 1-year, open-label safety study suggest that Patients who fall into this category may be ineligible for treatment with triptans.
There are mechanistic concerns with rimegepant and related CGRP receptor antagonists. They block CGRP’s effect in the central nervous system, but CGRP is also active in blood vessels and the heart, leading to the possibility that countering its vasodilating effect could expose organs to risk of ischemia.
The Food and Drug Administration approved rimegepant in 2020 for treatment of acute migraine attacks. Sponsor Biohaven is also seeking approval for migraine prevention after a successful phase 3 study published January 2021 in The Lancet.
Susan Hutchinson, MD, who is a headache specialist at Orange County Migraine & Headache Center in Irvine, Calif., presented the results at the American Academy of Neurology’s 2021 annual meeting. The open-label study suggested that rimegepant is generally safe. “The proportion of subjects reporting at least one adverse event was similar among subjects whether they had zero, one, or two or more cardiovascular disease risk factors, and also among those with low and moderate to high 10-year cardiovascular risk, as determined by the Framingham Risk Score,” said Dr. Hutchinson during her presentation.
Still, there was one concerning case: A 53-year-old man experienced an attack of angina. But he already had angina prior to the study, was being treated for hypercholesterolemia, and had current or former exposure to statins. “This adverse event was deemed by the investigator to not be related to rimegepant,” said Dr. Hutchinson.
During the following question-and-answer session, an attendee pressed Dr. Hutchinson about the case, and she admitted to some initial doubts. “That was my concern when I saw those slides. I’m like, ‘oh, my goodness.’ ” She clarified that the man’s angina history dated to 2016, which was several years before the trial, and the episode of angina occurred 7 months after the first dose of rimegepant. “He was treated with nitroglycerin and taken out of the trial,” said Dr. Hutchinson.
Proper patient selection is key
The research adds to the literature on rimegepant by providing data on multiple uses, as opposed to the phase 3 study, which only looked at single use, according to Olivia Begasse de Dhaem, MD, who is a neurology attending physician at Stamford (Conn.) Health and was the session moderator. Rimegepant and other oral CGRP receptor antagonists, including the FDA-approved ubrogepant and the investigative drug atogepant, will help fill the gap of patients who don’t tolerate or are ineligible for triptans, she said.
Dr. Begasse de Dhaem pointed out that patient selection remains important. “I think the main thing for patient care is to look at whether the patient we are treating would fit within the inclusion criteria, or would have been excluded from this study,” said Dr. Begasse de Dhaem. Specifically, according to its clinicaltrials.gov page, the trial excluded patients with hemiplegic and basilar migraine, as well as patients with uncontrolled, unstable, or recently diagnosed cardiovascular disease, those with a body mass index of 30 kg/m2 or higher, and hemoglobin A1c levels of 6.5% or higher. “This also looked at people with less than 15 migraine days per month, so it’s limited in how much we can extrapolate to people with chronic migraine who may take more than 7.7 rimegepant [doses, the mean value taken by trial participants] per month,” Dr. Begasse de Dhaem added.
She also applauded the inclusion of older patients in the study, noting that most migraine studies have an upper age limit.
The study included subjects who experienced 2-14 moderate or severe migraine attacks per month, and they were allowed to take other migraine medications. Cardiovascular risk factors did not prevent entry to the trial and, like the previous pivotal trial, the long-term safety study admitted subjects older than 65. Among the study cohort, 1,514 participants were told to treat migraine pain of any intensity with 75 mg rimegepant up to once per day on an as-needed basis (PRN), and a second group of 286 were told to take 75 mg rimegepant every other day for 12 weeks, along with PRN dosing on nonscheduled treatment days.
Nearly 90% of subjects were female, the mean age was 43.1 years, and 3.7% were age 65 or older. Among the study participants, 40.8% had cardiovascular risk factors, including 28.8% with one risk factor, and 12.1% with two or more. About 7% had a moderate to high (≥10%) 10-year cardiovascular risk by Framingham Risk Score, 23.6% had a family history of coronary artery disease, 11.7% were being treated for hypertension, 10.4% smoked, 8.3% were being treated with a statin, and 3.0% had a history of diabetes.
In total, subjects were exposed to 112,014 doses of rimegepant, a mean of 7.7 doses per 4-week period. The exposure was similar across all risk groups, which included zero risk factors, one risk factor, and two or more risk factors; FRS of less than 10%; and FRS of 10% or greater. The most common adverse events were upper respiratory tract infection (8.8%), nasopharyngitis (6.8%), and sinusitis (5.1%). The frequency of one or more adverse events was similar among those with zero cardiovascular risk factors (59.6%), one risk factor (61.4%), two or more risk factors (62.2%), FRS less than 10% (59.9%), and FRS of 10% or greater (59.9%).
The study was funded by Biohaven Pharmaceuticals. Dr. Hutchinson has been a consultant or advisory board member for Biohaven, Alder, Allergan, Amgen, Avanir, electroCore, Lilly, Novartis, Promius, Supernus, and Teva. She has been on the speaker’s bureau for Allergan, Amgen, Avanir, electroCore, Lilly, Novartis, Promius, Supernus, and Teva. Dr. Begasse de Dhaem has no relevant financial disclosures.
FROM AAN 2021