User login
Make the Diagnosis - February 2016
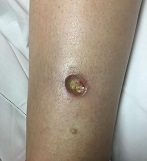
Diagnosis: Pyoderma gangrenosum
Pyoderma gangrenosum (PG) is an uncommon, noninfectious neutrophilic dermatosis that results in chronic ulcerative lesions. This disease process favors adult women and can be associated with systemic diseases in the majority of cases. The most common underlying systemic ailments include inflammatory bowel disease, arthritis, infection, and hematologic malignancy; it can also be drug induced.
Typically, the lesions begin as an erythematous pustule or nodule on an extremity. As was the case with our patient, a history of a "spider bite" or other arthropod assault may be elicited in the history as patients try to attribute a cause to the development of the initial ulceration. The pustule then develops into an ulcer with a characteristic necrotic, violaceous undermined border with a purulent base. Also, this disease process is associated with pathergy, in which minor trauma can induce additional lesions at remote sites.
There are four well-known types of pyoderma gangrenosum including the classic ulcerative lesions, pustular, bullous, and superficial granulomatous type, also known as vegetative PG. The pustular type may be seen more frequently in patients with inflammatory bowel disease, the bullous type may predominate in hematologic disorders, and the superficial granulomatous type is known to occur following surgery or other trauma.
The pathology of lesions can be nonspecific. However, in untreated lesions, widespread infiltration of neutrophils can be demonstrated at the base of the ulcers with accompanying necrosis at the periphery of lesions.
Dr. Bilu Martin is in private practice at Premier Dermatology, MD, in Aventura, Fla. More diagnostic cases are available at edermatologynews.com. To submit your case for possible publication, send an email to [email protected].
Diagnosis: Pyoderma gangrenosum
Pyoderma gangrenosum (PG) is an uncommon, noninfectious neutrophilic dermatosis that results in chronic ulcerative lesions. This disease process favors adult women and can be associated with systemic diseases in the majority of cases. The most common underlying systemic ailments include inflammatory bowel disease, arthritis, infection, and hematologic malignancy; it can also be drug induced.
Typically, the lesions begin as an erythematous pustule or nodule on an extremity. As was the case with our patient, a history of a "spider bite" or other arthropod assault may be elicited in the history as patients try to attribute a cause to the development of the initial ulceration. The pustule then develops into an ulcer with a characteristic necrotic, violaceous undermined border with a purulent base. Also, this disease process is associated with pathergy, in which minor trauma can induce additional lesions at remote sites.
There are four well-known types of pyoderma gangrenosum including the classic ulcerative lesions, pustular, bullous, and superficial granulomatous type, also known as vegetative PG. The pustular type may be seen more frequently in patients with inflammatory bowel disease, the bullous type may predominate in hematologic disorders, and the superficial granulomatous type is known to occur following surgery or other trauma.
The pathology of lesions can be nonspecific. However, in untreated lesions, widespread infiltration of neutrophils can be demonstrated at the base of the ulcers with accompanying necrosis at the periphery of lesions.
Dr. Bilu Martin is in private practice at Premier Dermatology, MD, in Aventura, Fla. More diagnostic cases are available at edermatologynews.com. To submit your case for possible publication, send an email to [email protected].
Diagnosis: Pyoderma gangrenosum
Pyoderma gangrenosum (PG) is an uncommon, noninfectious neutrophilic dermatosis that results in chronic ulcerative lesions. This disease process favors adult women and can be associated with systemic diseases in the majority of cases. The most common underlying systemic ailments include inflammatory bowel disease, arthritis, infection, and hematologic malignancy; it can also be drug induced.
Typically, the lesions begin as an erythematous pustule or nodule on an extremity. As was the case with our patient, a history of a "spider bite" or other arthropod assault may be elicited in the history as patients try to attribute a cause to the development of the initial ulceration. The pustule then develops into an ulcer with a characteristic necrotic, violaceous undermined border with a purulent base. Also, this disease process is associated with pathergy, in which minor trauma can induce additional lesions at remote sites.
There are four well-known types of pyoderma gangrenosum including the classic ulcerative lesions, pustular, bullous, and superficial granulomatous type, also known as vegetative PG. The pustular type may be seen more frequently in patients with inflammatory bowel disease, the bullous type may predominate in hematologic disorders, and the superficial granulomatous type is known to occur following surgery or other trauma.
The pathology of lesions can be nonspecific. However, in untreated lesions, widespread infiltration of neutrophils can be demonstrated at the base of the ulcers with accompanying necrosis at the periphery of lesions.
Dr. Bilu Martin is in private practice at Premier Dermatology, MD, in Aventura, Fla. More diagnostic cases are available at edermatologynews.com. To submit your case for possible publication, send an email to [email protected].
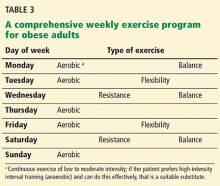
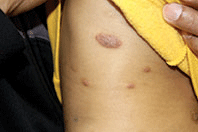
A 42-year-old woman with a 10-year history of Crohn's disease, treated with weekly subcutaneous injections of adalimumab, and hypertension presented with ulcerations on the lower extremities. She stated that the ulcerations began after she had been camping and reported being bitten by several ants during the trip, approximately 3 months earlier.
Pediatric Dermatology Consult - February 2016
By Catalina Matiz, M.D., and David Ginsberg
Nummular eczema
Nummular eczema is not an uncommon dermatosis that presents in pediatric and adult patients; its name, which derives from the Latin word nummulus (coin-like), refers to the coined-shape plaques that characterize this condition. It also has been referred to as discoid eczema and nummular dermatitis.1
The lesions begin as erythematous papules and vesicles that extend into larger oval or circular plaques that often become crusted, and can later progress to dry and scaly plaques.1,2 Patients often complain of intense pruritus.1 The lesions can be single or multiple, and more commonly occur on the extensor extremities as well as the trunk, and rarely affect the neck and the head.1-3 The pathophysiology of nummular eczema is not fully understood. It can occur in patients that exhibit atopic manifestations such as atopic dermatitis and other allergies, but there has been no clear link found between nummular eczema and atopy.3,4
Many theories exist implicating causative factors including Staphylococcus aureus colonization and xerosis.1 Similarly, some physicians believe that patch testing can be useful in these patients because of the potential for exacerbation caused by environmental allergens, but there is still no agreement on the ultimate cause.5 There is a higher incidence in males than females, and in the pediatric population, it is more common among “school aged” children between the ages of 2-12.6 Overall, nummular eczema is more commonly seen in adults, but it can occur at any age.2,3,6
Differential diagnosis
Nummular eczema is commonly mistaken as tinea corporis.1 The coined shape lesions, from which nummular eczema gets its name, can resemble the characteristic annular shape plaques of “ring worm,” but a potassium hydroxide (KOH) test or a fungal culture are simple ways to differentiate between the two conditions.
Nummular eczema occasionally can be confused for psoriasis as both entities can present with oval plaques. Psoriasis lesions tend to be pinker and less erythematous than nummular eczema lesions and most psoriasis plaques present with a characteristic silver scale.7 Clinically, nummular eczema is frequently associated with extreme pruritus, while in psoriasis the pruritus is less prominent.7
A biopsy would yield a more definitive diagnosis in difficult cases. Histologically, nummular eczema resembles other forms of spongiotic dermatitis, while psoriasis has very distinct histological features.7 Differentiating between contact dermatitis and nummular eczema relies on a thorough history of known allergies and potential exposure to environmental allergens. If history alone does not yield a definitive diagnosis and a suspicion for contact allergy is high, patch testing could help support one diagnosis over the other.5
Treatment
The generally accepted first line therapy includes mid to high potency topical corticosteroids in an ointment preparation or else under occlusion.1,4 Other topical agents used include tar preparations and calcineurin inhibitors.4 Intralesional corticosteroid injection can be used to treat isolated lesions that fail to respond to topical treatments.4
As with almost all manifestations of dermatitis, general gentle skin care measures and daily moisturizing are recommended.1 For more severe cases in older children, narrow-band UVB light therapy can be helpful.1 Due to their efficacy in treatment of other forms of refractory dermatitis, systemic therapy with cyclosporine, azathioprine, mycophenolate mofetil, and methotrexate can be used in cases in which phototherapy fails or is not accessible.4
In cases recalcitrant to topical therapies, secondary staphylococcal infection always should be ruled out and treated with systemic antimicrobials such as first generation cephalosporins.1
References
- Eczematous eruptions in childhood in “Hurwitz Clinical Pediatric Dermatology,” 4th ed. (New York, N.Y.: Elsevier, pp. 59-60
- Acta Derm Venereol. 1961;41:453-60.
- Acta Derm Venereol. 1969;49(2):189-96.
- Australas J Dermatol. 2010 May;51(2):128-30.
- Contact Dermatitis. 1997 May;36(5):261-4.
- Ped Dermatol. 2012 Oct;29(5):580-3.
- Dermatol Ther. 2006 Mar-Apr;19(2):73-82.
Dr. Matiz is assistant professor of dermatology at Rady Children’s Hospital San Diego–University of California, San Diego and Mr. Ginsberg is a research associate at the hospital. Dr. Matiz and Mr. Ginsberg said they have no relevant financial disclosures.
By Catalina Matiz, M.D., and David Ginsberg
Nummular eczema
Nummular eczema is not an uncommon dermatosis that presents in pediatric and adult patients; its name, which derives from the Latin word nummulus (coin-like), refers to the coined-shape plaques that characterize this condition. It also has been referred to as discoid eczema and nummular dermatitis.1
The lesions begin as erythematous papules and vesicles that extend into larger oval or circular plaques that often become crusted, and can later progress to dry and scaly plaques.1,2 Patients often complain of intense pruritus.1 The lesions can be single or multiple, and more commonly occur on the extensor extremities as well as the trunk, and rarely affect the neck and the head.1-3 The pathophysiology of nummular eczema is not fully understood. It can occur in patients that exhibit atopic manifestations such as atopic dermatitis and other allergies, but there has been no clear link found between nummular eczema and atopy.3,4
Many theories exist implicating causative factors including Staphylococcus aureus colonization and xerosis.1 Similarly, some physicians believe that patch testing can be useful in these patients because of the potential for exacerbation caused by environmental allergens, but there is still no agreement on the ultimate cause.5 There is a higher incidence in males than females, and in the pediatric population, it is more common among “school aged” children between the ages of 2-12.6 Overall, nummular eczema is more commonly seen in adults, but it can occur at any age.2,3,6
Differential diagnosis
Nummular eczema is commonly mistaken as tinea corporis.1 The coined shape lesions, from which nummular eczema gets its name, can resemble the characteristic annular shape plaques of “ring worm,” but a potassium hydroxide (KOH) test or a fungal culture are simple ways to differentiate between the two conditions.
Nummular eczema occasionally can be confused for psoriasis as both entities can present with oval plaques. Psoriasis lesions tend to be pinker and less erythematous than nummular eczema lesions and most psoriasis plaques present with a characteristic silver scale.7 Clinically, nummular eczema is frequently associated with extreme pruritus, while in psoriasis the pruritus is less prominent.7
A biopsy would yield a more definitive diagnosis in difficult cases. Histologically, nummular eczema resembles other forms of spongiotic dermatitis, while psoriasis has very distinct histological features.7 Differentiating between contact dermatitis and nummular eczema relies on a thorough history of known allergies and potential exposure to environmental allergens. If history alone does not yield a definitive diagnosis and a suspicion for contact allergy is high, patch testing could help support one diagnosis over the other.5
Treatment
The generally accepted first line therapy includes mid to high potency topical corticosteroids in an ointment preparation or else under occlusion.1,4 Other topical agents used include tar preparations and calcineurin inhibitors.4 Intralesional corticosteroid injection can be used to treat isolated lesions that fail to respond to topical treatments.4
As with almost all manifestations of dermatitis, general gentle skin care measures and daily moisturizing are recommended.1 For more severe cases in older children, narrow-band UVB light therapy can be helpful.1 Due to their efficacy in treatment of other forms of refractory dermatitis, systemic therapy with cyclosporine, azathioprine, mycophenolate mofetil, and methotrexate can be used in cases in which phototherapy fails or is not accessible.4
In cases recalcitrant to topical therapies, secondary staphylococcal infection always should be ruled out and treated with systemic antimicrobials such as first generation cephalosporins.1
References
- Eczematous eruptions in childhood in “Hurwitz Clinical Pediatric Dermatology,” 4th ed. (New York, N.Y.: Elsevier, pp. 59-60
- Acta Derm Venereol. 1961;41:453-60.
- Acta Derm Venereol. 1969;49(2):189-96.
- Australas J Dermatol. 2010 May;51(2):128-30.
- Contact Dermatitis. 1997 May;36(5):261-4.
- Ped Dermatol. 2012 Oct;29(5):580-3.
- Dermatol Ther. 2006 Mar-Apr;19(2):73-82.
Dr. Matiz is assistant professor of dermatology at Rady Children’s Hospital San Diego–University of California, San Diego and Mr. Ginsberg is a research associate at the hospital. Dr. Matiz and Mr. Ginsberg said they have no relevant financial disclosures.
By Catalina Matiz, M.D., and David Ginsberg
Nummular eczema
Nummular eczema is not an uncommon dermatosis that presents in pediatric and adult patients; its name, which derives from the Latin word nummulus (coin-like), refers to the coined-shape plaques that characterize this condition. It also has been referred to as discoid eczema and nummular dermatitis.1
The lesions begin as erythematous papules and vesicles that extend into larger oval or circular plaques that often become crusted, and can later progress to dry and scaly plaques.1,2 Patients often complain of intense pruritus.1 The lesions can be single or multiple, and more commonly occur on the extensor extremities as well as the trunk, and rarely affect the neck and the head.1-3 The pathophysiology of nummular eczema is not fully understood. It can occur in patients that exhibit atopic manifestations such as atopic dermatitis and other allergies, but there has been no clear link found between nummular eczema and atopy.3,4
Many theories exist implicating causative factors including Staphylococcus aureus colonization and xerosis.1 Similarly, some physicians believe that patch testing can be useful in these patients because of the potential for exacerbation caused by environmental allergens, but there is still no agreement on the ultimate cause.5 There is a higher incidence in males than females, and in the pediatric population, it is more common among “school aged” children between the ages of 2-12.6 Overall, nummular eczema is more commonly seen in adults, but it can occur at any age.2,3,6
Differential diagnosis
Nummular eczema is commonly mistaken as tinea corporis.1 The coined shape lesions, from which nummular eczema gets its name, can resemble the characteristic annular shape plaques of “ring worm,” but a potassium hydroxide (KOH) test or a fungal culture are simple ways to differentiate between the two conditions.
Nummular eczema occasionally can be confused for psoriasis as both entities can present with oval plaques. Psoriasis lesions tend to be pinker and less erythematous than nummular eczema lesions and most psoriasis plaques present with a characteristic silver scale.7 Clinically, nummular eczema is frequently associated with extreme pruritus, while in psoriasis the pruritus is less prominent.7
A biopsy would yield a more definitive diagnosis in difficult cases. Histologically, nummular eczema resembles other forms of spongiotic dermatitis, while psoriasis has very distinct histological features.7 Differentiating between contact dermatitis and nummular eczema relies on a thorough history of known allergies and potential exposure to environmental allergens. If history alone does not yield a definitive diagnosis and a suspicion for contact allergy is high, patch testing could help support one diagnosis over the other.5
Treatment
The generally accepted first line therapy includes mid to high potency topical corticosteroids in an ointment preparation or else under occlusion.1,4 Other topical agents used include tar preparations and calcineurin inhibitors.4 Intralesional corticosteroid injection can be used to treat isolated lesions that fail to respond to topical treatments.4
As with almost all manifestations of dermatitis, general gentle skin care measures and daily moisturizing are recommended.1 For more severe cases in older children, narrow-band UVB light therapy can be helpful.1 Due to their efficacy in treatment of other forms of refractory dermatitis, systemic therapy with cyclosporine, azathioprine, mycophenolate mofetil, and methotrexate can be used in cases in which phototherapy fails or is not accessible.4
In cases recalcitrant to topical therapies, secondary staphylococcal infection always should be ruled out and treated with systemic antimicrobials such as first generation cephalosporins.1
References
- Eczematous eruptions in childhood in “Hurwitz Clinical Pediatric Dermatology,” 4th ed. (New York, N.Y.: Elsevier, pp. 59-60
- Acta Derm Venereol. 1961;41:453-60.
- Acta Derm Venereol. 1969;49(2):189-96.
- Australas J Dermatol. 2010 May;51(2):128-30.
- Contact Dermatitis. 1997 May;36(5):261-4.
- Ped Dermatol. 2012 Oct;29(5):580-3.
- Dermatol Ther. 2006 Mar-Apr;19(2):73-82.
Dr. Matiz is assistant professor of dermatology at Rady Children’s Hospital San Diego–University of California, San Diego and Mr. Ginsberg is a research associate at the hospital. Dr. Matiz and Mr. Ginsberg said they have no relevant financial disclosures.
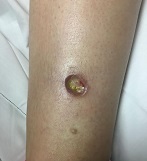
A 9-month-old male with no significant previous medical history presents with a very itchy rash that has been present for 6 weeks. His mother reports that the lesions began as small red bumps on the extremities and his torso, that developed over the course of a few weeks into large round, red, very pruritic plaques. He has been treated with an antifungal cream for several weeks without resolution, and most recently his mother has been applying hydrocortisone 2.5% cream with no improvement either. On exam, the patient is a well appearing infant, who is visibly irritated due to the pruritus accompanying his rash. There are several 1-cm to 4-cm round and oval dry, scaly, erythematous plaques on the trunk (see photo) and a few on the extremities. There is no generalized xerosis.
High Headache Frequency Is More Likely During Perimenopause
Women in perimenopause are at increased risk of high-frequency headache, compared with premenopausal women, according to data published online ahead of print January 21 in Headache. Women in menopause also are at increased risk of high-frequency headache, but the effect of menopause on headache frequency may be mediated or confounded by medication overuse or depression.
“Our results confirm the commonly held belief that the perimenopause worsens headache, but challenge the idea that migraine ‘always’ improves during the menopause,” said Vincent T. Martin, MD, Professor of Internal Medicine in the University of Cincinnati’s (UC) Division of General Internal Medicine and codirector of the Headache and Facial Pain Program at the UC Neuroscience Institute. “Recognition of the increased risk of high-frequency headache during the menopausal transition suggests a need for optimized preventive treatment of migraine during this time of women’s life.”
Research has suggested a lower prevalence of headache or migraine during menopause, compared with premenopause. No previous studies have analyzed whether frequency of headache attacks changes during the menopausal transition among women with migraine, however. Dr. Martin and colleagues sought to determine whether the percentage of female migraineurs with high-frequency headache, defined as 10 or more days/month, is greater during the perimenopausal and menopausal time periods, compared with the premenopausal period. The researchers also set out to examine whether any increase in high-frequency headache during a particular reproductive phase was restricted to the early or late stages of the phase.
An Analysis of AMPP Data
To answer their questions, the investigators conducted a cross-sectional study using data from the American Migraine Prevalence and Prevention (AMPP) study. The AMPP researchers elicited data about headache from 162,756 respondents age 12 or older in 2004 and invited a random subset of 24,000 people age 18 or older with self-reported severe headache to participate in annual follow-up surveys for the subsequent five years. Follow-up surveys included questions about sociodemographics (eg, BMI, smoking, and household income) and headache types and characteristics, in addition to the Migraine Disability Assessment Score. Dr. Martin and colleagues examined data from the 2006 follow-up survey because it contained questions on the menstrual cycle.
Eligible participants in the cross-sectional study were women with a diagnosis of migraine between ages 35 and 65. Women who were pregnant, breastfeeding, had a history of hysterectomy or oophorectomy, or used hormonal therapies were excluded from the analysis. The investigators classified respondents as premenopause, perimenopause, and menopause according to Stages of Reproductive Aging Workshop criteria.
Late Perimenopause and Headache Frequency
The analysis included 3,664 women, of whom 3,454 had episodic migraine and 210 had chronic migraine. In all, 1,263 women were classified as premenopausal, 1,283 as perimenopausal, and 1,118 as menopausal. Compared with women in premenopause, women in perimenopause and menopause used more migraine preventives and were more likely to overuse medication.
Approximately 8% of premenopausal women had high-frequency headache, compared with 12.2% of perimenopausal women and 12.0% of postmenopausal women. After adjustments for sociodemographics alone, the odds ratios (ORs) of high-frequency headache were 1.62 for perimenopausal women and 1.76 for menopausal women, compared with premenopausal women. After adjustment for BMI, current migraine preventive use, medication overuse, and depression, the OR decreased, but remained significant in the perimenopausal group (OR, 1.42) and lost significance for the menopausal group (OR, 1.27). Depression and medication overuse significantly increased the likelihood of high-frequency headache.
When the researchers examined participants in the early and late stages of perimenopause and adjusted data for all covariates, women in late perimenopause had an increased likelihood of high-frequency headache (OR, 1.72), but women in early perimenopause had a statistically insignificant increased risk of this outcome (OR, 1.22), compared with premenopausal women. When the researchers examined the early and late stages of menopause, compared with premenopause, they found no significant difference in risk of high-frequency headache after controlling for all covariates.
Results Contradict Common Belief
“These results suggest that the hormonal milieu of the late perimenopause is particularly provocative for high-frequency headache among migraineurs,” said Dr. Martin. Because the researchers did not collect data on premenstrual syndrome (PMS) disorder, they could not determine whether the increased risk for high-frequency headache during perimenopause only occurred in female migraineurs with PMS or in the entire population.
Epidemiologic studies have contributed to an impression that migraine prevalence declines in menopausal women, but the current study’s results contradict this impression. “Our study used high-frequency headache as its primary outcome measure, rather than migraine prevalence. It is plausible that during menopause, migraine prevalence decreases and migraine attacks occur more frequently in subgroups of women,” said Dr. Martin. “Women, as they get older, develop lots of aches and pains, joint [pain], and back pain, and it is possible [that] their overuse of pain medications for headache and other conditions might actually drive an increase in headaches for the menopause group,” he added.
Estrogen withdrawal in the late luteal phase, low serum levels of estrogen or progesterone, and increased uterine prostaglandin release could precipitate headache during the menopausal transition, said the researchers. These hormonal changes also may change the characteristics of the menstrual cycle, which could in turn affect headache frequency.
An advantage of the cross-sectional analysis is that it was a large population-based study of persons with migraine “that should have wide generalizability to the general population,” said Dr. Martin. The outcome measure of high-frequency headache, however, was not limited to migraine, but included all headaches. In addition, headache frequency was self-reported, and investigators did not confirm it with daily headache diaries. Finally, the researchers did not account or control for aura. “Our results should be considered preliminary until confirmed in future studies,” Dr. Martin concluded.
—Erik Greb
Suggested Reading
Martin VT, Pavlovic J, Fanning KM, et al. Perimenopause and menopause are associated with high frequency headache in women with migraine: results of the American Migraine Prevalence and Prevention Study. Headache. 2016 Jan 21 [Epub ahead of print].
Women in perimenopause are at increased risk of high-frequency headache, compared with premenopausal women, according to data published online ahead of print January 21 in Headache. Women in menopause also are at increased risk of high-frequency headache, but the effect of menopause on headache frequency may be mediated or confounded by medication overuse or depression.
“Our results confirm the commonly held belief that the perimenopause worsens headache, but challenge the idea that migraine ‘always’ improves during the menopause,” said Vincent T. Martin, MD, Professor of Internal Medicine in the University of Cincinnati’s (UC) Division of General Internal Medicine and codirector of the Headache and Facial Pain Program at the UC Neuroscience Institute. “Recognition of the increased risk of high-frequency headache during the menopausal transition suggests a need for optimized preventive treatment of migraine during this time of women’s life.”
Research has suggested a lower prevalence of headache or migraine during menopause, compared with premenopause. No previous studies have analyzed whether frequency of headache attacks changes during the menopausal transition among women with migraine, however. Dr. Martin and colleagues sought to determine whether the percentage of female migraineurs with high-frequency headache, defined as 10 or more days/month, is greater during the perimenopausal and menopausal time periods, compared with the premenopausal period. The researchers also set out to examine whether any increase in high-frequency headache during a particular reproductive phase was restricted to the early or late stages of the phase.
An Analysis of AMPP Data
To answer their questions, the investigators conducted a cross-sectional study using data from the American Migraine Prevalence and Prevention (AMPP) study. The AMPP researchers elicited data about headache from 162,756 respondents age 12 or older in 2004 and invited a random subset of 24,000 people age 18 or older with self-reported severe headache to participate in annual follow-up surveys for the subsequent five years. Follow-up surveys included questions about sociodemographics (eg, BMI, smoking, and household income) and headache types and characteristics, in addition to the Migraine Disability Assessment Score. Dr. Martin and colleagues examined data from the 2006 follow-up survey because it contained questions on the menstrual cycle.
Eligible participants in the cross-sectional study were women with a diagnosis of migraine between ages 35 and 65. Women who were pregnant, breastfeeding, had a history of hysterectomy or oophorectomy, or used hormonal therapies were excluded from the analysis. The investigators classified respondents as premenopause, perimenopause, and menopause according to Stages of Reproductive Aging Workshop criteria.
Late Perimenopause and Headache Frequency
The analysis included 3,664 women, of whom 3,454 had episodic migraine and 210 had chronic migraine. In all, 1,263 women were classified as premenopausal, 1,283 as perimenopausal, and 1,118 as menopausal. Compared with women in premenopause, women in perimenopause and menopause used more migraine preventives and were more likely to overuse medication.
Approximately 8% of premenopausal women had high-frequency headache, compared with 12.2% of perimenopausal women and 12.0% of postmenopausal women. After adjustments for sociodemographics alone, the odds ratios (ORs) of high-frequency headache were 1.62 for perimenopausal women and 1.76 for menopausal women, compared with premenopausal women. After adjustment for BMI, current migraine preventive use, medication overuse, and depression, the OR decreased, but remained significant in the perimenopausal group (OR, 1.42) and lost significance for the menopausal group (OR, 1.27). Depression and medication overuse significantly increased the likelihood of high-frequency headache.
When the researchers examined participants in the early and late stages of perimenopause and adjusted data for all covariates, women in late perimenopause had an increased likelihood of high-frequency headache (OR, 1.72), but women in early perimenopause had a statistically insignificant increased risk of this outcome (OR, 1.22), compared with premenopausal women. When the researchers examined the early and late stages of menopause, compared with premenopause, they found no significant difference in risk of high-frequency headache after controlling for all covariates.
Results Contradict Common Belief
“These results suggest that the hormonal milieu of the late perimenopause is particularly provocative for high-frequency headache among migraineurs,” said Dr. Martin. Because the researchers did not collect data on premenstrual syndrome (PMS) disorder, they could not determine whether the increased risk for high-frequency headache during perimenopause only occurred in female migraineurs with PMS or in the entire population.
Epidemiologic studies have contributed to an impression that migraine prevalence declines in menopausal women, but the current study’s results contradict this impression. “Our study used high-frequency headache as its primary outcome measure, rather than migraine prevalence. It is plausible that during menopause, migraine prevalence decreases and migraine attacks occur more frequently in subgroups of women,” said Dr. Martin. “Women, as they get older, develop lots of aches and pains, joint [pain], and back pain, and it is possible [that] their overuse of pain medications for headache and other conditions might actually drive an increase in headaches for the menopause group,” he added.
Estrogen withdrawal in the late luteal phase, low serum levels of estrogen or progesterone, and increased uterine prostaglandin release could precipitate headache during the menopausal transition, said the researchers. These hormonal changes also may change the characteristics of the menstrual cycle, which could in turn affect headache frequency.
An advantage of the cross-sectional analysis is that it was a large population-based study of persons with migraine “that should have wide generalizability to the general population,” said Dr. Martin. The outcome measure of high-frequency headache, however, was not limited to migraine, but included all headaches. In addition, headache frequency was self-reported, and investigators did not confirm it with daily headache diaries. Finally, the researchers did not account or control for aura. “Our results should be considered preliminary until confirmed in future studies,” Dr. Martin concluded.
—Erik Greb
Women in perimenopause are at increased risk of high-frequency headache, compared with premenopausal women, according to data published online ahead of print January 21 in Headache. Women in menopause also are at increased risk of high-frequency headache, but the effect of menopause on headache frequency may be mediated or confounded by medication overuse or depression.
“Our results confirm the commonly held belief that the perimenopause worsens headache, but challenge the idea that migraine ‘always’ improves during the menopause,” said Vincent T. Martin, MD, Professor of Internal Medicine in the University of Cincinnati’s (UC) Division of General Internal Medicine and codirector of the Headache and Facial Pain Program at the UC Neuroscience Institute. “Recognition of the increased risk of high-frequency headache during the menopausal transition suggests a need for optimized preventive treatment of migraine during this time of women’s life.”
Research has suggested a lower prevalence of headache or migraine during menopause, compared with premenopause. No previous studies have analyzed whether frequency of headache attacks changes during the menopausal transition among women with migraine, however. Dr. Martin and colleagues sought to determine whether the percentage of female migraineurs with high-frequency headache, defined as 10 or more days/month, is greater during the perimenopausal and menopausal time periods, compared with the premenopausal period. The researchers also set out to examine whether any increase in high-frequency headache during a particular reproductive phase was restricted to the early or late stages of the phase.
An Analysis of AMPP Data
To answer their questions, the investigators conducted a cross-sectional study using data from the American Migraine Prevalence and Prevention (AMPP) study. The AMPP researchers elicited data about headache from 162,756 respondents age 12 or older in 2004 and invited a random subset of 24,000 people age 18 or older with self-reported severe headache to participate in annual follow-up surveys for the subsequent five years. Follow-up surveys included questions about sociodemographics (eg, BMI, smoking, and household income) and headache types and characteristics, in addition to the Migraine Disability Assessment Score. Dr. Martin and colleagues examined data from the 2006 follow-up survey because it contained questions on the menstrual cycle.
Eligible participants in the cross-sectional study were women with a diagnosis of migraine between ages 35 and 65. Women who were pregnant, breastfeeding, had a history of hysterectomy or oophorectomy, or used hormonal therapies were excluded from the analysis. The investigators classified respondents as premenopause, perimenopause, and menopause according to Stages of Reproductive Aging Workshop criteria.
Late Perimenopause and Headache Frequency
The analysis included 3,664 women, of whom 3,454 had episodic migraine and 210 had chronic migraine. In all, 1,263 women were classified as premenopausal, 1,283 as perimenopausal, and 1,118 as menopausal. Compared with women in premenopause, women in perimenopause and menopause used more migraine preventives and were more likely to overuse medication.
Approximately 8% of premenopausal women had high-frequency headache, compared with 12.2% of perimenopausal women and 12.0% of postmenopausal women. After adjustments for sociodemographics alone, the odds ratios (ORs) of high-frequency headache were 1.62 for perimenopausal women and 1.76 for menopausal women, compared with premenopausal women. After adjustment for BMI, current migraine preventive use, medication overuse, and depression, the OR decreased, but remained significant in the perimenopausal group (OR, 1.42) and lost significance for the menopausal group (OR, 1.27). Depression and medication overuse significantly increased the likelihood of high-frequency headache.
When the researchers examined participants in the early and late stages of perimenopause and adjusted data for all covariates, women in late perimenopause had an increased likelihood of high-frequency headache (OR, 1.72), but women in early perimenopause had a statistically insignificant increased risk of this outcome (OR, 1.22), compared with premenopausal women. When the researchers examined the early and late stages of menopause, compared with premenopause, they found no significant difference in risk of high-frequency headache after controlling for all covariates.
Results Contradict Common Belief
“These results suggest that the hormonal milieu of the late perimenopause is particularly provocative for high-frequency headache among migraineurs,” said Dr. Martin. Because the researchers did not collect data on premenstrual syndrome (PMS) disorder, they could not determine whether the increased risk for high-frequency headache during perimenopause only occurred in female migraineurs with PMS or in the entire population.
Epidemiologic studies have contributed to an impression that migraine prevalence declines in menopausal women, but the current study’s results contradict this impression. “Our study used high-frequency headache as its primary outcome measure, rather than migraine prevalence. It is plausible that during menopause, migraine prevalence decreases and migraine attacks occur more frequently in subgroups of women,” said Dr. Martin. “Women, as they get older, develop lots of aches and pains, joint [pain], and back pain, and it is possible [that] their overuse of pain medications for headache and other conditions might actually drive an increase in headaches for the menopause group,” he added.
Estrogen withdrawal in the late luteal phase, low serum levels of estrogen or progesterone, and increased uterine prostaglandin release could precipitate headache during the menopausal transition, said the researchers. These hormonal changes also may change the characteristics of the menstrual cycle, which could in turn affect headache frequency.
An advantage of the cross-sectional analysis is that it was a large population-based study of persons with migraine “that should have wide generalizability to the general population,” said Dr. Martin. The outcome measure of high-frequency headache, however, was not limited to migraine, but included all headaches. In addition, headache frequency was self-reported, and investigators did not confirm it with daily headache diaries. Finally, the researchers did not account or control for aura. “Our results should be considered preliminary until confirmed in future studies,” Dr. Martin concluded.
—Erik Greb
Suggested Reading
Martin VT, Pavlovic J, Fanning KM, et al. Perimenopause and menopause are associated with high frequency headache in women with migraine: results of the American Migraine Prevalence and Prevention Study. Headache. 2016 Jan 21 [Epub ahead of print].
Suggested Reading
Martin VT, Pavlovic J, Fanning KM, et al. Perimenopause and menopause are associated with high frequency headache in women with migraine: results of the American Migraine Prevalence and Prevention Study. Headache. 2016 Jan 21 [Epub ahead of print].
Hope may not be the best component of an exercise regimen
Judging by the crowd and the difficulty in finding a locker at my gym on January 1, a lot of people are serious about their 2016 New Year’s resolution to exercise and lose weight. But as most of us have experienced personally and professionally, embarking on a well-intended effort to exercise in the hope of losing weight more often results in frustration than a trip to the store to buy smaller-sized clothes.
The frequent answer to the question “What did the doctor say at your visit?” provides a partial explanation of this phenomenon: “Nothing, just that I should exercise and lose weight” is the usual hackneyed response. Nothing—as in nothing unexpected, nothing significant, and nothing specific was said. It is with this lack of specific advice that I feel many of us let our patients down.
We admonish patients to eat fewer calories, avoid the evil carbs, walk 10,000 steps, ride a bike, use the elliptical, or swim three times a week. There is a concrete but broad nature to these suggestions, but there is also a familiarity and a lack of specificity that leaves patients feeling that there is no science behind them. And the truth is that many of us are not comfortable enough with current data from our exercise physiology colleagues to have a detailed discussion with our patients that pairs their specific goals with an exercise regimen and diet most likely to be beneficial. We may fear sounding like the morning talk show doctors, offering sound bites instead of engaging in an evidence-based dialogue with our patients.
Many of our patients cannot afford a personal trainer to guide and cajole them through a successful regimen—assuming that they, or we, can separate myth and fact and choose an appropriate trainer. We should try to be their guide and sounding board as well as coach and cheerleader.
In this issue of the Journal, John and Christopher Higgins present a primer on the background information to use when talking with our patients about starting an exercise program focused on weight loss. They provide useful references that support specific approaches to achieve realistic expectations, and they review and compare various strategies.
I’m sure by March it will again be easier to find a locker at my gym. And I hope by then that my new workout plan will be more scientifically based, as well as a bit more effective. Even data-based hope springs eternal.
Judging by the crowd and the difficulty in finding a locker at my gym on January 1, a lot of people are serious about their 2016 New Year’s resolution to exercise and lose weight. But as most of us have experienced personally and professionally, embarking on a well-intended effort to exercise in the hope of losing weight more often results in frustration than a trip to the store to buy smaller-sized clothes.
The frequent answer to the question “What did the doctor say at your visit?” provides a partial explanation of this phenomenon: “Nothing, just that I should exercise and lose weight” is the usual hackneyed response. Nothing—as in nothing unexpected, nothing significant, and nothing specific was said. It is with this lack of specific advice that I feel many of us let our patients down.
We admonish patients to eat fewer calories, avoid the evil carbs, walk 10,000 steps, ride a bike, use the elliptical, or swim three times a week. There is a concrete but broad nature to these suggestions, but there is also a familiarity and a lack of specificity that leaves patients feeling that there is no science behind them. And the truth is that many of us are not comfortable enough with current data from our exercise physiology colleagues to have a detailed discussion with our patients that pairs their specific goals with an exercise regimen and diet most likely to be beneficial. We may fear sounding like the morning talk show doctors, offering sound bites instead of engaging in an evidence-based dialogue with our patients.
Many of our patients cannot afford a personal trainer to guide and cajole them through a successful regimen—assuming that they, or we, can separate myth and fact and choose an appropriate trainer. We should try to be their guide and sounding board as well as coach and cheerleader.
In this issue of the Journal, John and Christopher Higgins present a primer on the background information to use when talking with our patients about starting an exercise program focused on weight loss. They provide useful references that support specific approaches to achieve realistic expectations, and they review and compare various strategies.
I’m sure by March it will again be easier to find a locker at my gym. And I hope by then that my new workout plan will be more scientifically based, as well as a bit more effective. Even data-based hope springs eternal.
Judging by the crowd and the difficulty in finding a locker at my gym on January 1, a lot of people are serious about their 2016 New Year’s resolution to exercise and lose weight. But as most of us have experienced personally and professionally, embarking on a well-intended effort to exercise in the hope of losing weight more often results in frustration than a trip to the store to buy smaller-sized clothes.
The frequent answer to the question “What did the doctor say at your visit?” provides a partial explanation of this phenomenon: “Nothing, just that I should exercise and lose weight” is the usual hackneyed response. Nothing—as in nothing unexpected, nothing significant, and nothing specific was said. It is with this lack of specific advice that I feel many of us let our patients down.
We admonish patients to eat fewer calories, avoid the evil carbs, walk 10,000 steps, ride a bike, use the elliptical, or swim three times a week. There is a concrete but broad nature to these suggestions, but there is also a familiarity and a lack of specificity that leaves patients feeling that there is no science behind them. And the truth is that many of us are not comfortable enough with current data from our exercise physiology colleagues to have a detailed discussion with our patients that pairs their specific goals with an exercise regimen and diet most likely to be beneficial. We may fear sounding like the morning talk show doctors, offering sound bites instead of engaging in an evidence-based dialogue with our patients.
Many of our patients cannot afford a personal trainer to guide and cajole them through a successful regimen—assuming that they, or we, can separate myth and fact and choose an appropriate trainer. We should try to be their guide and sounding board as well as coach and cheerleader.
In this issue of the Journal, John and Christopher Higgins present a primer on the background information to use when talking with our patients about starting an exercise program focused on weight loss. They provide useful references that support specific approaches to achieve realistic expectations, and they review and compare various strategies.
I’m sure by March it will again be easier to find a locker at my gym. And I hope by then that my new workout plan will be more scientifically based, as well as a bit more effective. Even data-based hope springs eternal.
The ethics of ICDs: History and future directions
In 1975, Julia and Joseph Quinlan approached the administrator of St. Clare’s Hospital in Denville, New Jersey, and requested that the mechanical ventilator on which their adopted daughter, Karen, was dependent be turned off. Karen Ann Quinlan, 21 years old, was in a permanent vegetative state after a severe anoxic event, and her parents had been informed by the hospital’s medical staff that she would never regain consciousness.
To the Quinlans’ request to withdraw the ventilator, the hospital administrator replied, “You have to understand our position, Mrs. Quinlan. In this hospital we don’t kill people.”1
The administrator’s response was consistent with prevailing ethical and legal perspectives, analyses, and directives at that time related to discontinuation of life-sustaining treatment. In the mid-1970s, the American Medical Association’s position was that it was permissible to not put a patient on a ventilator (ie, a physician could withhold a life-sustaining treatment), but once a patient was on a ventilator, it was not permissible to take the patient off if the intention was to allow death to occur.1 However, the New Jersey Supreme Court ultimately found this distinction between withholding and withdrawing unconvincing, and ruled unanimously that Karen Quinlan’s ventilator could be turned off.2
THE HASTINGS CENTER REPORT: STOPPING IS THE SAME AS NOT STARTING
During the subsequent decade, further ethical analysis and additional legal cases resulted in new insights and more nuanced thinking about forgoing life-sustaining treatment.
These developments were summarized in a 1987 report by the Hastings Center,3 a leading bioethics research and policy institute. The report provided normative guidance for the termination of life-sustaining treatment and for the care of dying patients. It acknowledged that deciding not to start a life-sustaining treatment can emotionally and psychologically affect healthcare professionals differently than deciding to stop such a treatment. However, the report also asserted that there is no morally important difference between withholding and withdrawing such treatments.
Reflecting a partnership model between patients and professionals for healthcare decision-making, and affirming the ethical significance of both a burden-benefit analysis and patient autonomy, the report stated that when a patient or surrogate in collaboration with a responsible healthcare professional decides that a treatment under way and the life it supports have become more burdensome than beneficial to the patient, that is sufficient reason to stop. There is no ethical requirement that treatment, once initiated, must continue against the patient’s wishes or when the surrogate determines that it is more burdensome than beneficial from the patient’s perspective. In fact, imposing treatment in such circumstances violates the patient’s right to self-determination.3
The report noted further that, because of frequent uncertainty about the efficacy of proposed treatments, it is preferable to initiate time-limited trials of treatments and then later stop them if they prove ineffective or become overly burdensome from a patient’s perspective.
ICDs ARE LIKE OTHER LIFE-SUSTAINING THERAPIES
In this issue of Cleveland Clinic Journal of Medicine, Baibars et al4 address the question of how implantable cardioverter-defibrillators (ICDs) should be managed at the end of life. The historical events and developments recounted above regarding withdrawing life-sustaining technologies are an appropriate context for ethically assessing the management of ICDs for dying patients.
Obviously, ICDs are not ventilators, but like ventilators, they are life-sustaining therapy, as are dialysis machines, blood transfusions, medically supplied nutrition and hydration, ventricular assist devices, and other implantable electronic cardiac devices such as pacemakers. Each of these life-sustaining therapies, depending on a patient’s clinical condition, underlying illness, and comorbidities, can become a death-prolonging technology.
An ethical framework and analysis about whether to continue any life-sustaining therapy, including an ICD, must include an assessment of the benefit-to-burden ratio from the patient’s perspective. Does the therapy enhance or maintain a quality of life acceptable to the patient? Or has it become overly burdensome and does it maintain a quality of life the patient finds (or would find) unacceptable? If the latter is true, and especially in the context of an underlying terminal condition, then shifting the goals of care to focus on comfort is always appropriate and ethically justified. Treatments—including ICDs—that do not contribute to patient comfort should be withdrawn.
TOWARD COMPETENCY IN ETHICAL MANAGEMENT
Baibars et al note that much more needs to be done to enhance competencies, increase proficiencies, and mitigate the moral distress of healthcare professionals caring for dying patients with ICDs and other devices. To help clinicians achieve a personal and professional “comfort zone” for ethically managing patients with ICDs, we recommend that healthcare institutions, medical schools, and nursing schools take the following steps:
Develop comprehensive end-of-life policies, procedures, and protocols that incorporate specific guidance for managing cardiac devices and that have been endorsed by a hospital ethics committee. Such guidance can be informative and educational and can ensure that decisions and resulting actions (including stopping cardiac devices) are ethically supportable.
Provide more palliative care training in medical and nursing schools, residency programs, and continuing education activities so that front-line clinicians can deliver “basic,” “primary” palliative care not requiring specialty palliative medicine. This training, called for in the Institute of Medicine’s 2014 report, Dying in America,5 should include explicit ethics discussions about managing cardiac devices at the end of life.
Provide ongoing training in communication skills needed for all patient-professional encounters. Effectively engaging patients in goals-of-care discussions, especially patients with life-limiting illnesses such as heart failure, cannot be achieved without these skills.
- Pence G. Comas: Karen Quinlan and Nancy Cruzan. In: Classic Cases in Medical Ethics: Accounts of Cases That Have Shaped Medical Ethics, With Philosophical, Legal, and Historical Backgrounds, 3rd edition. Boston: McGraw-Hill; 2000:29–55.
- In the matter of Karen Quinlan, an alleged incompetent. In re Quinlan. 70 N.J. 10, 355 A.2d 647 (1976), cert. denied, 429 U.S. 922 (1976).
- Wolf SM. Hastings Center. Guidelines on the Termination of Life-Sustaining Treatment and Care of the Dying: A Report by the Hastings Center. The Hastings Center: Briarcliff Manor, NY; 1987.
- Baibars MM, Alraies MC, Kabach A, Pritzker M. Can patients opt to turn off implantable cardioverter-defibrillators near the end of life? Cleve Clin J Med 2016; 83:97–98.
- National Academy of Sciences. Dying in America: improving quality and honoring individual p near the end of life. www.iom.edu/Reports/2014/Dying-In-America-Improving-Quality-and-Honoring-Individual-P-Near-the-End-of-Life.aspx. Accessed January 4, 2016.
In 1975, Julia and Joseph Quinlan approached the administrator of St. Clare’s Hospital in Denville, New Jersey, and requested that the mechanical ventilator on which their adopted daughter, Karen, was dependent be turned off. Karen Ann Quinlan, 21 years old, was in a permanent vegetative state after a severe anoxic event, and her parents had been informed by the hospital’s medical staff that she would never regain consciousness.
To the Quinlans’ request to withdraw the ventilator, the hospital administrator replied, “You have to understand our position, Mrs. Quinlan. In this hospital we don’t kill people.”1
The administrator’s response was consistent with prevailing ethical and legal perspectives, analyses, and directives at that time related to discontinuation of life-sustaining treatment. In the mid-1970s, the American Medical Association’s position was that it was permissible to not put a patient on a ventilator (ie, a physician could withhold a life-sustaining treatment), but once a patient was on a ventilator, it was not permissible to take the patient off if the intention was to allow death to occur.1 However, the New Jersey Supreme Court ultimately found this distinction between withholding and withdrawing unconvincing, and ruled unanimously that Karen Quinlan’s ventilator could be turned off.2
THE HASTINGS CENTER REPORT: STOPPING IS THE SAME AS NOT STARTING
During the subsequent decade, further ethical analysis and additional legal cases resulted in new insights and more nuanced thinking about forgoing life-sustaining treatment.
These developments were summarized in a 1987 report by the Hastings Center,3 a leading bioethics research and policy institute. The report provided normative guidance for the termination of life-sustaining treatment and for the care of dying patients. It acknowledged that deciding not to start a life-sustaining treatment can emotionally and psychologically affect healthcare professionals differently than deciding to stop such a treatment. However, the report also asserted that there is no morally important difference between withholding and withdrawing such treatments.
Reflecting a partnership model between patients and professionals for healthcare decision-making, and affirming the ethical significance of both a burden-benefit analysis and patient autonomy, the report stated that when a patient or surrogate in collaboration with a responsible healthcare professional decides that a treatment under way and the life it supports have become more burdensome than beneficial to the patient, that is sufficient reason to stop. There is no ethical requirement that treatment, once initiated, must continue against the patient’s wishes or when the surrogate determines that it is more burdensome than beneficial from the patient’s perspective. In fact, imposing treatment in such circumstances violates the patient’s right to self-determination.3
The report noted further that, because of frequent uncertainty about the efficacy of proposed treatments, it is preferable to initiate time-limited trials of treatments and then later stop them if they prove ineffective or become overly burdensome from a patient’s perspective.
ICDs ARE LIKE OTHER LIFE-SUSTAINING THERAPIES
In this issue of Cleveland Clinic Journal of Medicine, Baibars et al4 address the question of how implantable cardioverter-defibrillators (ICDs) should be managed at the end of life. The historical events and developments recounted above regarding withdrawing life-sustaining technologies are an appropriate context for ethically assessing the management of ICDs for dying patients.
Obviously, ICDs are not ventilators, but like ventilators, they are life-sustaining therapy, as are dialysis machines, blood transfusions, medically supplied nutrition and hydration, ventricular assist devices, and other implantable electronic cardiac devices such as pacemakers. Each of these life-sustaining therapies, depending on a patient’s clinical condition, underlying illness, and comorbidities, can become a death-prolonging technology.
An ethical framework and analysis about whether to continue any life-sustaining therapy, including an ICD, must include an assessment of the benefit-to-burden ratio from the patient’s perspective. Does the therapy enhance or maintain a quality of life acceptable to the patient? Or has it become overly burdensome and does it maintain a quality of life the patient finds (or would find) unacceptable? If the latter is true, and especially in the context of an underlying terminal condition, then shifting the goals of care to focus on comfort is always appropriate and ethically justified. Treatments—including ICDs—that do not contribute to patient comfort should be withdrawn.
TOWARD COMPETENCY IN ETHICAL MANAGEMENT
Baibars et al note that much more needs to be done to enhance competencies, increase proficiencies, and mitigate the moral distress of healthcare professionals caring for dying patients with ICDs and other devices. To help clinicians achieve a personal and professional “comfort zone” for ethically managing patients with ICDs, we recommend that healthcare institutions, medical schools, and nursing schools take the following steps:
Develop comprehensive end-of-life policies, procedures, and protocols that incorporate specific guidance for managing cardiac devices and that have been endorsed by a hospital ethics committee. Such guidance can be informative and educational and can ensure that decisions and resulting actions (including stopping cardiac devices) are ethically supportable.
Provide more palliative care training in medical and nursing schools, residency programs, and continuing education activities so that front-line clinicians can deliver “basic,” “primary” palliative care not requiring specialty palliative medicine. This training, called for in the Institute of Medicine’s 2014 report, Dying in America,5 should include explicit ethics discussions about managing cardiac devices at the end of life.
Provide ongoing training in communication skills needed for all patient-professional encounters. Effectively engaging patients in goals-of-care discussions, especially patients with life-limiting illnesses such as heart failure, cannot be achieved without these skills.
In 1975, Julia and Joseph Quinlan approached the administrator of St. Clare’s Hospital in Denville, New Jersey, and requested that the mechanical ventilator on which their adopted daughter, Karen, was dependent be turned off. Karen Ann Quinlan, 21 years old, was in a permanent vegetative state after a severe anoxic event, and her parents had been informed by the hospital’s medical staff that she would never regain consciousness.
To the Quinlans’ request to withdraw the ventilator, the hospital administrator replied, “You have to understand our position, Mrs. Quinlan. In this hospital we don’t kill people.”1
The administrator’s response was consistent with prevailing ethical and legal perspectives, analyses, and directives at that time related to discontinuation of life-sustaining treatment. In the mid-1970s, the American Medical Association’s position was that it was permissible to not put a patient on a ventilator (ie, a physician could withhold a life-sustaining treatment), but once a patient was on a ventilator, it was not permissible to take the patient off if the intention was to allow death to occur.1 However, the New Jersey Supreme Court ultimately found this distinction between withholding and withdrawing unconvincing, and ruled unanimously that Karen Quinlan’s ventilator could be turned off.2
THE HASTINGS CENTER REPORT: STOPPING IS THE SAME AS NOT STARTING
During the subsequent decade, further ethical analysis and additional legal cases resulted in new insights and more nuanced thinking about forgoing life-sustaining treatment.
These developments were summarized in a 1987 report by the Hastings Center,3 a leading bioethics research and policy institute. The report provided normative guidance for the termination of life-sustaining treatment and for the care of dying patients. It acknowledged that deciding not to start a life-sustaining treatment can emotionally and psychologically affect healthcare professionals differently than deciding to stop such a treatment. However, the report also asserted that there is no morally important difference between withholding and withdrawing such treatments.
Reflecting a partnership model between patients and professionals for healthcare decision-making, and affirming the ethical significance of both a burden-benefit analysis and patient autonomy, the report stated that when a patient or surrogate in collaboration with a responsible healthcare professional decides that a treatment under way and the life it supports have become more burdensome than beneficial to the patient, that is sufficient reason to stop. There is no ethical requirement that treatment, once initiated, must continue against the patient’s wishes or when the surrogate determines that it is more burdensome than beneficial from the patient’s perspective. In fact, imposing treatment in such circumstances violates the patient’s right to self-determination.3
The report noted further that, because of frequent uncertainty about the efficacy of proposed treatments, it is preferable to initiate time-limited trials of treatments and then later stop them if they prove ineffective or become overly burdensome from a patient’s perspective.
ICDs ARE LIKE OTHER LIFE-SUSTAINING THERAPIES
In this issue of Cleveland Clinic Journal of Medicine, Baibars et al4 address the question of how implantable cardioverter-defibrillators (ICDs) should be managed at the end of life. The historical events and developments recounted above regarding withdrawing life-sustaining technologies are an appropriate context for ethically assessing the management of ICDs for dying patients.
Obviously, ICDs are not ventilators, but like ventilators, they are life-sustaining therapy, as are dialysis machines, blood transfusions, medically supplied nutrition and hydration, ventricular assist devices, and other implantable electronic cardiac devices such as pacemakers. Each of these life-sustaining therapies, depending on a patient’s clinical condition, underlying illness, and comorbidities, can become a death-prolonging technology.
An ethical framework and analysis about whether to continue any life-sustaining therapy, including an ICD, must include an assessment of the benefit-to-burden ratio from the patient’s perspective. Does the therapy enhance or maintain a quality of life acceptable to the patient? Or has it become overly burdensome and does it maintain a quality of life the patient finds (or would find) unacceptable? If the latter is true, and especially in the context of an underlying terminal condition, then shifting the goals of care to focus on comfort is always appropriate and ethically justified. Treatments—including ICDs—that do not contribute to patient comfort should be withdrawn.
TOWARD COMPETENCY IN ETHICAL MANAGEMENT
Baibars et al note that much more needs to be done to enhance competencies, increase proficiencies, and mitigate the moral distress of healthcare professionals caring for dying patients with ICDs and other devices. To help clinicians achieve a personal and professional “comfort zone” for ethically managing patients with ICDs, we recommend that healthcare institutions, medical schools, and nursing schools take the following steps:
Develop comprehensive end-of-life policies, procedures, and protocols that incorporate specific guidance for managing cardiac devices and that have been endorsed by a hospital ethics committee. Such guidance can be informative and educational and can ensure that decisions and resulting actions (including stopping cardiac devices) are ethically supportable.
Provide more palliative care training in medical and nursing schools, residency programs, and continuing education activities so that front-line clinicians can deliver “basic,” “primary” palliative care not requiring specialty palliative medicine. This training, called for in the Institute of Medicine’s 2014 report, Dying in America,5 should include explicit ethics discussions about managing cardiac devices at the end of life.
Provide ongoing training in communication skills needed for all patient-professional encounters. Effectively engaging patients in goals-of-care discussions, especially patients with life-limiting illnesses such as heart failure, cannot be achieved without these skills.
- Pence G. Comas: Karen Quinlan and Nancy Cruzan. In: Classic Cases in Medical Ethics: Accounts of Cases That Have Shaped Medical Ethics, With Philosophical, Legal, and Historical Backgrounds, 3rd edition. Boston: McGraw-Hill; 2000:29–55.
- In the matter of Karen Quinlan, an alleged incompetent. In re Quinlan. 70 N.J. 10, 355 A.2d 647 (1976), cert. denied, 429 U.S. 922 (1976).
- Wolf SM. Hastings Center. Guidelines on the Termination of Life-Sustaining Treatment and Care of the Dying: A Report by the Hastings Center. The Hastings Center: Briarcliff Manor, NY; 1987.
- Baibars MM, Alraies MC, Kabach A, Pritzker M. Can patients opt to turn off implantable cardioverter-defibrillators near the end of life? Cleve Clin J Med 2016; 83:97–98.
- National Academy of Sciences. Dying in America: improving quality and honoring individual p near the end of life. www.iom.edu/Reports/2014/Dying-In-America-Improving-Quality-and-Honoring-Individual-P-Near-the-End-of-Life.aspx. Accessed January 4, 2016.
- Pence G. Comas: Karen Quinlan and Nancy Cruzan. In: Classic Cases in Medical Ethics: Accounts of Cases That Have Shaped Medical Ethics, With Philosophical, Legal, and Historical Backgrounds, 3rd edition. Boston: McGraw-Hill; 2000:29–55.
- In the matter of Karen Quinlan, an alleged incompetent. In re Quinlan. 70 N.J. 10, 355 A.2d 647 (1976), cert. denied, 429 U.S. 922 (1976).
- Wolf SM. Hastings Center. Guidelines on the Termination of Life-Sustaining Treatment and Care of the Dying: A Report by the Hastings Center. The Hastings Center: Briarcliff Manor, NY; 1987.
- Baibars MM, Alraies MC, Kabach A, Pritzker M. Can patients opt to turn off implantable cardioverter-defibrillators near the end of life? Cleve Clin J Med 2016; 83:97–98.
- National Academy of Sciences. Dying in America: improving quality and honoring individual p near the end of life. www.iom.edu/Reports/2014/Dying-In-America-Improving-Quality-and-Honoring-Individual-P-Near-the-End-of-Life.aspx. Accessed January 4, 2016.
Veterans, guilt, and suicide risk: An opportunity to collaborate with chaplains?
Suicidal behavior is a major cause of morbidity and mortality in the United States,1 and active-duty and reserve military personnel and veterans account for nearly 18% of suicide deaths.2 By one estimate, as many as 22 veterans die by suicide each day.3 These rates are thought to be due to a higher incidence of mental illness in certain veteran populations relative to the general population.4–8 Consequently, a number of mental health services are available to veterans in a variety of clinical and community settings.
Chaplains and clinicians bring complementary skills and services to the problem of suicide risk among veterans. In particular, helping at-risk veterans deal with experiences of guilt is an opportunity for interdisciplinary collaboration. Available literature supports the potential utility of chaplaincy services in supporting at-risk veteran populations.9–15
But while most healthcare facilities have chaplains on staff, there is little information to guide any such collaboration. Further, healthcare providers appear to have a limited understanding of chaplaincy services, the “language” within which chaplains operate, or the roles chaplains play in healthcare settings.16
In the following discussion, using the example of experiences of guilt, we offer our insights and suggestions on how chaplaincy services may prove useful in alleviating this complex emotion in veterans at risk of suicide.
BENEFITS OF TALKING TO A CHAPLAIN
Collaboration between healthcare providers and pastoral care professionals has been suggested as a means of enhancing the treatment of patients with mental illness.17,18 Chaplains draw from a variety of faith traditions and are usually trained to respond to the needs of people from a variety of religious and spiritual backgrounds. They provide some non-faithbased services (eg, crisis intervention, life review, bereavement counseling) resembling those also provided in formal mental healthcare settings.19 By facilitating religious and spiritual coping and religious practice and responding to religious and spiritual needs, chaplains also offer a level of support not typically offered by formal mental healthcare providers.20
Veterans at risk of suicide sometimes look to pastoral care providers, particularly chaplains, for mental health support.9,10 Research on the effects of chaplaincy services on suicidal behavior is just beginning to emerge.15 Still, the US Department of Health and Human Services has recognized pastoral care services as having a “beneficial and therapeutic effect on the medical condition of a patient.”11
For example, in one study, hospital inpatients reported higher satisfaction if they had been visited by a chaplain.12 Chaplains help align treatment plans with patient values and wishes.13 In another study,14 patients undergoing coronary artery bypass grafting who were randomized to receive five visits from a chaplain were found to have a higher rate of positive religious coping (eg, forgiveness, letting go of anger). Positive religious coping has been correlated with lower levels of psychological stress and better mental health outcomes.
EXPERIENCING GUILT IS LINKED TO RISK OF SUICIDE
Suicidal behavior is complex, multifaceted, and linked to genetic, neurologic, psychological, social, and cultural factors.21
Assessing for and addressing certain complex emotions, such as guilt and shame, is an important part of suicide prevention efforts. Guilt is defined as a “controllable psychological state that is typically linked to a specific action or behavior, and which entails regret or remorse.”22
Guilt has been linked to risk of suicide in veterans.23–25 In one study, close to 75% of veterans who had thought about suicide said they frequently experienced guilt about having violated the precepts of their faith group, family, God, life, or the military.26
Such findings suggest that the sense of guilt experienced by some at-risk veterans may be grounded in a variety of contexts. For example, faith communities that place a strong emphasis on obedience to moral, ethical, and religious precepts may contribute to the experience of guilt unless balanced by a message of grace or favor from a benevolent God or deity. Without this balance, engaging in activities that are not fully sanctioned by one’s faith community may lead to guilt.
Families might also contribute to veterans’ experiences of guilt by placing unrealistic expectations on them. And the family environment may not be conducive to resolving feelings of guilt in veterans, harboring resentment and antipathies and making it very difficult to alleviate any ensuing sense of distress.
CLINICIAN’S ROLE IN ASSESSING GUILT
In addressing and assessing guilt in veterans at risk of suicide, clinicians should try to recognize the source and clinical implications of this emotion.
Recognize the source of guilt
Guilt may indicate a clinical disorder such as a mood disorder (eg, major depression).27 Mood disorders significantly increase the risk of suicidal behavior.28,29
Beyond diagnosing a clinical disorder, prescribing pharmacotherapy, and referring for mental healthcare services, recognizing the source of this emotion remains an important part of addressing a patient’s experience of guilt. Especially when associated with a clinical disorder, guilt is often irrational and excessive and does not appropriately reflect the experience or situation in question.
Case conceptualization, defined as “synthesizing the patient’s experience with relevant clinical theory and research,”30 can be used to understand the context in which the guilt-inducing action or behavior occurred and the veteran’s own interpretation of his or her actions. Understanding the source of the patient’s guilt could be used to plan treatment and resolve any underlying sense of distress.
As with other negative emotions, the affective component of guilt is often the result of cognitive distortions made as the person tries to make sense of what has occurred or to reconcile beliefs of right and wrong with the guilt-inducing behavior.31 The common cognitive errors associated with guilt include:
- Hindsight bias (a belief that one should have known what was going to happen as a result of one’s actions)
- Responsibility distortion (a belief that one’s actions directly caused an adverse event)
- Justification distortion (a belief that one’s actions were not justified by the situation)
- Wrongdoing distortion (a belief that one violated one’s own standards of right and wrong).31
Cognitive therapy to counter cognitive distortions
A variety of clinical options exist to help veterans manage and resolve guilt.
Cognitive therapy can counter the cognitive distortions that drive feelings of guilt. The goal is to guide patients to examine the evidence, process the event, and realize that their behavior was appropriate for the given situation. Cognitive processing therapy and prolonged exposure therapy have both been shown to decrease trauma-related guilt, though cognitive processing therapy was found to be better at decreasing guilt that arose from cognitive distortions.32
Guilt and suicide ideation have also been associated with a belief that one’s actions constituted an unforgivable sin.33 Responding to these inherently religious-spiritual cognitive distortions may be beyond the scope of expertise for many healthcare professionals. In such cases, it may be prudent to consider complementing clinical services with pastoral care. It follows that pastoral care services should only be provided if the veteran voices a desire and readiness for them. The clinician and chaplain can then work together to provide coordinated care to best meet the patient’s needs, to address the experience of guilt, and to alleviate the sense of distress.
A CHAPLAIN’S PERSPECTIVE ON GUILT
A prominent feature of pastoral practice is helping people, including at-risk veterans, resolve feelings of guilt regardless of the context on which the emotion is founded (eg, religion, shame).10 For many people, guilt is an impenetrable barrier, preventing resolution of whatever experience led to a sense of inner turmoil.
Forgiveness
In the context of pastoral care, resolution of guilt is ordinarily tied to a need for forgiveness. There are multiple ways in which forgiveness can be grounded in religious and spiritual contexts.34 Examples include forgiving others (ie, forswearing resentment, anger, or hatred directed toward another person), being forgiven by God or another benevolent deity, and forgiving oneself for violating perceived personal transgressions.35 In some cases, divine forgiveness may be conditional on interpersonal forgiveness.36 Forgiveness is also sometimes seen as a remedy for sin and a way to restore moral order.37
Some people may initially think they can never be forgiven. With time and the weight of one’s experiences, the impossibility of forgiveness can become so ingrained that it becomes a core belief. These core beliefs set up a vicious circle of thoughts and feelings, in which people and places and events from the past are continuously brought forward into the present. Anger and resentment become the steady diet for the tormented self that feels forever powerless over experienced injustices. These relived experiences drive the person into a deep isolation where the self becomes less human—a thing, an object. This experience of losing oneself proves excruciating and often leads to contemplation of suicide as a way to resolve anguish.
Hope emerges
Pastoral care services provide a means to reframe one’s core beliefs, manage and resolve the burden of guilt, and uncover new motivation for living.
The practice of spiritual direction within the discipline of pastoral care listens for these inner movements and encourages the person to give voice to them in his or her own words. No longer limited by a diminished, tormented self, the real self begins to relate to another reality that changes his or her identity, relieves the burden of guilt, and gives reason, purpose, and meaning to life.
Even with this opportunity for a new life, however, cognitive distortions based on a disproportional “faith-based prism” may persist. In this case, clinicians and chaplains must work closely together to reframe old understandings of self and incorrect understandings of religion and spirituality into one that continues to reinforce this newfound sense of hope.38
A VETERAN OF IRAQ WITH SUICIDE IDEATION
The following case illustrates how clinicians and chaplains may be able to work together to help facilitate the resolution of guilt.
A veteran who had served in Iraq had entered the Domiciliary Care Program at a US Department of Veterans Affairs medical center. He reported experiencing problems with guilt, forgiveness, and suicide ideation. A clinical therapeutic program was prescribed after a psychological evaluation uncovered that he was also struggling with depression and posttraumatic stress disorder.
His mental healthcare providers recognized the importance of incorporating a religious-spiritual component into the therapeutic plan, and so consulted with a chaplain to plan a suitable course of action. Specifically, this veteran reported feeling that he could not be forgiven for his military experiences, a feeling that was giving way to alienation and isolation from the God of his faith tradition.
The chaplain helped this veteran reflect on his military experiences, giving him the perspective he needed to view his God as one who truly loves him. He recognized instances in which he could have lost his life had it not been for others who intervened on his behalf at just the right time. This awareness caused him to think about his life differently, challenging him to reframe his relationship with God. Instead of simple coincidences, the veteran began to consider the mystery behind these times and places.
Over time and in keeping with the tenets of his faith tradition, the veteran stated that he was ultimately able to accept and receive God’s love and forgiveness. He now reports that these inner spiritual movements serve as a source of support during occasional relapses into emotional distress. These movements allow him to consider the mystery of his present life and its value based on his experience of his God’s love and forgiveness.
CARE FOR SUICIDE SURVIVORS
The experience of guilt is not limited to veterans. Those bereaved by suicide are also left to manage their own experiences of the loss and ensuing complex emotions. Friends and loved ones who survive a suicide decedent may experience guilt, feeling that they somehow contributed to or failed to prevent the suicide. Such feelings of guilt are hypothesized to lower the threshold for suicidal behavior in those bereaved.39
Guilt and shame are also frequently encountered in survivors of nonfatal suicide attempts.40 Chaplaincy services might also prove useful for these individuals.
TIME IS EVERYTHING
Patients who may have an active psychopathology should have their clinical therapeutic needs attended to first. If the clinician deems pastoral care services to be an appropriate complementary support option, care should be taken to select a pastoral care provider who is adequately prepared for this role. Different professional organizations (eg, Association of Professional Chaplains) have established board-certification procedures, minimum education requirements, and supervised practical experience required for chaplaincy certification.
Also, spiritual growth and development remain a core focus of pastoral practice. Clinicians should discontinue any collaboration with pastoral care providers who question an individual’s faith or commitment to his or her faith, or who promote thinking or actions that could be deleterious to the patient’s therapeutic trajectory.
SUMMING UP
We have here presented our perspectives on how chaplaincy services can be used to complement clinical services in support of at-risk veterans struggling with experiences of guilt. Unfortunately, the current level of collaboration between chaplains and clinicians in support of at-risk veteran populations is limited.20 Our hope is that clinicians managing these at-risk patients will develop a greater awareness of how chaplaincy services might be able to help in alleviating experiences of guilt in at-risk veteran populations. A further hope is that such cases will serve as an opportunity for greater interdisciplinary collaboration, benefiting at-risk veterans most in need of support.
Acknowledgment: Dr. Rasmussen was supported by the Office of Academic Affiliations, Advanced Fellowship Program in Mental Illness Research and Treatment, US Department of Veterans Affairs, VISN 2 Center of Excellence for Suicide Prevention.
- Centers for Disease Control and Prevention (CDC). Suicide and self-inflicted injury. www.cdc.gov/nchs/fastats/suicide.htm. Accessed November 12, 2015.
- Centers for Disease Control and Prevention (CDC). National violent death reporting system (NVDRS). https://wisqars.cdc.gov:8443/nvdrs/nvdrsDisplay.jsp. Accessed November 12, 2015.
- Kemp JE, Bossarte R. Suicide data report, 2012. www.sprc.org/library_resources/items/suicide-data-report-2012. Accessed November 12, 2015.
- Bullman TA, Kang HK. The risk of suicide among wounded Vietnam veterans. Am J Public Health 1996; 86:662–667.
- Kang HK, Bullman TA. Is there an epidemic of suicides among current and former US military personnel? Ann Epidemiol 2009; 19:757–760.
- LeardMann CA, Powell TM, Smith TC, et al. Risk factors associated with suicide in current and former US military personnel. JAMA 2013; 310:496–506.
- Mrnak-Meyer J, Tate SR, Tripp JC, Worley MJ, Jajodia A, McQuaid JR. Predictors of suicide-related hospitalization among US veterans receiving treatment for comorbid depression and substance dependence: who is the riskiest of the risky? Suicide Life Threat Behav 2011; 41:532–542.
- Pietrzak RH, Russo AR, Ling Q, Southwick SM. Suicidal ideation in treatment-seeking veterans of Operations Enduring Freedom and Iraqi Freedom: the role of coping strategies, resilience, and social support. J Psychiatr Res 2011; 45:720–726.
- Kopacz MS, McCarten JM, Pollitt MJ. VHA chaplaincy contact with veterans at increased risk of suicide. South Med J 2014; 107: 661–664.
- Kopacz MS. Providing pastoral care services in a clinical setting to veterans at-risk of suicide. J Relig Health 2013; 52:759–767.
- Medicare program; payment for nursing and allied health education. Health Care Financing Administration (HCFA), HHS. Final rule. Fed Regist 2001; 66:3358–3376.
- Marin DB, Sharma V, Sosunov E, Egorova N, Goldstein R, Handzo GF. Relationship between chaplain visits and patient satisfaction. J Health Care Chaplain 2015; 21:14–24.
- Flannelly KJ, Emanuel LL, Handzo GF, Galek K, Silton NR, Carlson M. A national study of chaplaincy services and end-of-life outcomes. BMC Palliat Care 2012; 11:10.
- Bay PS, Beckman D, Trippi J, Gunderman R, Terry C. The effect of pastoral care services on anxiety, depression, hope, religious coping, and religious problem solving styles: a randomized controlled study. J Relig Health 2008; 47:57–69.
- Kopacz MS, Nieuwsma JA, Jackson GL, et al. Chaplains’ engagement with suicidality among their service users: findings from the VA/DoD Integrated Mental Health Strategy. Suicide Life Threat Behav 2015. [Epub ahead of print.]
- Flannelly KJ, Galek K, Bucchino J, Handzo GF, Tannenbaum HP. Department directors’ perceptions of the roles and functions of hospital chaplains: a national survey. Hosp Top 2005; 83:19–27.
- Farrell JL, Goebert DA. Collaboration between psychiatrists and clergy in recognizing and treating serious mental illness. Psychiatr Serv 2008; 59:437–440.
- Weaver AJ, Flannelly KJ, Flannelly LT, Oppenheimer JE. Collaboration between clergy and mental health professionals: a review of professional health care journals from 1980 through 1999. Counsel Val 2003; 47:162–171.
- Handzo GF, Flannelly KJ, Kudler T, et al. What do chaplains really do? II. Interventions in the New York chaplaincy study. J Health Care Chaplain 2008; 14:39–56.
- Kopacz MS, Pollitt MJ. Delivering chaplaincy services to veterans at increased risk of suicide. J Health Care Chaplain 2015; 21:1–13.
- Knox KL, Bossarte RM. Suicide prevention for veterans and active duty personnel. Am J Public Health 2012;102(suppl 1):S8–S9.
- Bryan CJ, Morrow CE, Etienne N, Ray-Sannerud B. Guilt, shame, and suicidal ideation in a military outpatient clinical sample. Depress Anxiety 2013; 30:55–60.
- Ganz D, Sher L. Educating medical professionals about suicide prevention among military veterans. Int J Adolesc Med Health 2013; 25:187–191.
- Hendin H, Haas AP. Suicide and guilt as manifestations of PTSD in Vietnam combat veterans. Am J Psychiatry 1991; 148:586–591.
- Maguen S, Metzler TJ, Bosch J, Marmar CR, Knight SJ, Neylan TC. Killing in combat may be independently associated with suicidal ideation. Depress Anxiety 2012; 29:918–923.
- Kopacz MS, McCarten JM, Vance CG, Connery AL. A preliminary study for exploring different sources of guilt in a sample of veterans who sought chaplaincy services. Mil Psychol 2015; 27:1–8.
- Buck CJ. 2013 ICD-9-CM for physicians. St. Louis, MO: Saunders; 2013.
- Angst F, Stassen HH, Clayton PJ, Angst J. Mortality of patients with mood disorders: follow-up over 34-38 years. J Affect Disord 2002; 68:167–181.
- Nierenberg AA, Gray SM, Grandin LD. Mood disorders and suicide. J Clin Psychiatry 2001; 62(suppl 25):27–30.
- Macneil CA, Hasty MK, Conus P, Berk M. Is diagnosis enough to guide interventions in mental health? Using case formulation in clinical practice. BMC Med 2012; 10:111.
- Kubany ES, Manke FP. Cognitive therapy for trauma-related guilt: conceptual bases and treatment outlines. Cogn Behav Pract 1995; 2:27–61.
- Resick PA, Nishith P, Weaver TL, Astin MC, Feuer CA. Comparison of cognitive-processing therapy with prolonged exposure and a waiting condition for the treatment of chronic posttraumatic stress disorder in female rape victims. J Consult Clin Psychol 2002; 70:867–879.
- Exline JJ, Yali AM, Sanderson WC. Guilt, discord, and alienation: the role of religious strain in depression and suicidality. J Clin Psychol 2000; 56:1481–1496.
- Musick MA. Multiple forms of forgiveness and their relationship with aging and religion, In: Schaie KW, Krause N, Booth A, editors. Religious Influences on Health and Well-being in the Elderly. New York, NY: Springer Publishing Company; 2004:202–214.
- Kaplan BH, Munroe-Blum H, Blazer DG. Religion, health and forgiveness: tradition and challenges. In: Levin JS, editor. Religion in Aging and Health. Theoretical Foundations and Methodological Frontiers. Thousand Oaks, CA: SAGE Focus Edition; 1994:52–77.
- Worthington EL Jr, Berry JW, Parrott L III. Unforgiveness, forgiveness, religion and health. In: Plante TG, Sherman AC, editors. Faith and Health. Psychological Perspectives. New York, NY: Guilford Press; 2001:107–138.
- Enright RD, Gassin EA, Wu GR. Forgiveness: a developmental view. J Moral Educ 1992; 21:99–114.
- Kopacz MS, O’Reilly LM, Van Inwagen CC, et al. Understanding the role of chaplains in veteran suicide prevention efforts: a discussion paper. SAGE Open 2014; 4:1–10.
- Young IT, Iglewicz A, Glorioso D, et al. Suicide bereavement and complicated grief. Dialogues Clin Neurosci 2012; 14:177–186.
- Wiklander M, Samuelsson M, Asberg M. Shame reactions after suicide attempt. Scand J Caring Sci 2003; 17:293–300.
Suicidal behavior is a major cause of morbidity and mortality in the United States,1 and active-duty and reserve military personnel and veterans account for nearly 18% of suicide deaths.2 By one estimate, as many as 22 veterans die by suicide each day.3 These rates are thought to be due to a higher incidence of mental illness in certain veteran populations relative to the general population.4–8 Consequently, a number of mental health services are available to veterans in a variety of clinical and community settings.
Chaplains and clinicians bring complementary skills and services to the problem of suicide risk among veterans. In particular, helping at-risk veterans deal with experiences of guilt is an opportunity for interdisciplinary collaboration. Available literature supports the potential utility of chaplaincy services in supporting at-risk veteran populations.9–15
But while most healthcare facilities have chaplains on staff, there is little information to guide any such collaboration. Further, healthcare providers appear to have a limited understanding of chaplaincy services, the “language” within which chaplains operate, or the roles chaplains play in healthcare settings.16
In the following discussion, using the example of experiences of guilt, we offer our insights and suggestions on how chaplaincy services may prove useful in alleviating this complex emotion in veterans at risk of suicide.
BENEFITS OF TALKING TO A CHAPLAIN
Collaboration between healthcare providers and pastoral care professionals has been suggested as a means of enhancing the treatment of patients with mental illness.17,18 Chaplains draw from a variety of faith traditions and are usually trained to respond to the needs of people from a variety of religious and spiritual backgrounds. They provide some non-faithbased services (eg, crisis intervention, life review, bereavement counseling) resembling those also provided in formal mental healthcare settings.19 By facilitating religious and spiritual coping and religious practice and responding to religious and spiritual needs, chaplains also offer a level of support not typically offered by formal mental healthcare providers.20
Veterans at risk of suicide sometimes look to pastoral care providers, particularly chaplains, for mental health support.9,10 Research on the effects of chaplaincy services on suicidal behavior is just beginning to emerge.15 Still, the US Department of Health and Human Services has recognized pastoral care services as having a “beneficial and therapeutic effect on the medical condition of a patient.”11
For example, in one study, hospital inpatients reported higher satisfaction if they had been visited by a chaplain.12 Chaplains help align treatment plans with patient values and wishes.13 In another study,14 patients undergoing coronary artery bypass grafting who were randomized to receive five visits from a chaplain were found to have a higher rate of positive religious coping (eg, forgiveness, letting go of anger). Positive religious coping has been correlated with lower levels of psychological stress and better mental health outcomes.
EXPERIENCING GUILT IS LINKED TO RISK OF SUICIDE
Suicidal behavior is complex, multifaceted, and linked to genetic, neurologic, psychological, social, and cultural factors.21
Assessing for and addressing certain complex emotions, such as guilt and shame, is an important part of suicide prevention efforts. Guilt is defined as a “controllable psychological state that is typically linked to a specific action or behavior, and which entails regret or remorse.”22
Guilt has been linked to risk of suicide in veterans.23–25 In one study, close to 75% of veterans who had thought about suicide said they frequently experienced guilt about having violated the precepts of their faith group, family, God, life, or the military.26
Such findings suggest that the sense of guilt experienced by some at-risk veterans may be grounded in a variety of contexts. For example, faith communities that place a strong emphasis on obedience to moral, ethical, and religious precepts may contribute to the experience of guilt unless balanced by a message of grace or favor from a benevolent God or deity. Without this balance, engaging in activities that are not fully sanctioned by one’s faith community may lead to guilt.
Families might also contribute to veterans’ experiences of guilt by placing unrealistic expectations on them. And the family environment may not be conducive to resolving feelings of guilt in veterans, harboring resentment and antipathies and making it very difficult to alleviate any ensuing sense of distress.
CLINICIAN’S ROLE IN ASSESSING GUILT
In addressing and assessing guilt in veterans at risk of suicide, clinicians should try to recognize the source and clinical implications of this emotion.
Recognize the source of guilt
Guilt may indicate a clinical disorder such as a mood disorder (eg, major depression).27 Mood disorders significantly increase the risk of suicidal behavior.28,29
Beyond diagnosing a clinical disorder, prescribing pharmacotherapy, and referring for mental healthcare services, recognizing the source of this emotion remains an important part of addressing a patient’s experience of guilt. Especially when associated with a clinical disorder, guilt is often irrational and excessive and does not appropriately reflect the experience or situation in question.
Case conceptualization, defined as “synthesizing the patient’s experience with relevant clinical theory and research,”30 can be used to understand the context in which the guilt-inducing action or behavior occurred and the veteran’s own interpretation of his or her actions. Understanding the source of the patient’s guilt could be used to plan treatment and resolve any underlying sense of distress.
As with other negative emotions, the affective component of guilt is often the result of cognitive distortions made as the person tries to make sense of what has occurred or to reconcile beliefs of right and wrong with the guilt-inducing behavior.31 The common cognitive errors associated with guilt include:
- Hindsight bias (a belief that one should have known what was going to happen as a result of one’s actions)
- Responsibility distortion (a belief that one’s actions directly caused an adverse event)
- Justification distortion (a belief that one’s actions were not justified by the situation)
- Wrongdoing distortion (a belief that one violated one’s own standards of right and wrong).31
Cognitive therapy to counter cognitive distortions
A variety of clinical options exist to help veterans manage and resolve guilt.
Cognitive therapy can counter the cognitive distortions that drive feelings of guilt. The goal is to guide patients to examine the evidence, process the event, and realize that their behavior was appropriate for the given situation. Cognitive processing therapy and prolonged exposure therapy have both been shown to decrease trauma-related guilt, though cognitive processing therapy was found to be better at decreasing guilt that arose from cognitive distortions.32
Guilt and suicide ideation have also been associated with a belief that one’s actions constituted an unforgivable sin.33 Responding to these inherently religious-spiritual cognitive distortions may be beyond the scope of expertise for many healthcare professionals. In such cases, it may be prudent to consider complementing clinical services with pastoral care. It follows that pastoral care services should only be provided if the veteran voices a desire and readiness for them. The clinician and chaplain can then work together to provide coordinated care to best meet the patient’s needs, to address the experience of guilt, and to alleviate the sense of distress.
A CHAPLAIN’S PERSPECTIVE ON GUILT
A prominent feature of pastoral practice is helping people, including at-risk veterans, resolve feelings of guilt regardless of the context on which the emotion is founded (eg, religion, shame).10 For many people, guilt is an impenetrable barrier, preventing resolution of whatever experience led to a sense of inner turmoil.
Forgiveness
In the context of pastoral care, resolution of guilt is ordinarily tied to a need for forgiveness. There are multiple ways in which forgiveness can be grounded in religious and spiritual contexts.34 Examples include forgiving others (ie, forswearing resentment, anger, or hatred directed toward another person), being forgiven by God or another benevolent deity, and forgiving oneself for violating perceived personal transgressions.35 In some cases, divine forgiveness may be conditional on interpersonal forgiveness.36 Forgiveness is also sometimes seen as a remedy for sin and a way to restore moral order.37
Some people may initially think they can never be forgiven. With time and the weight of one’s experiences, the impossibility of forgiveness can become so ingrained that it becomes a core belief. These core beliefs set up a vicious circle of thoughts and feelings, in which people and places and events from the past are continuously brought forward into the present. Anger and resentment become the steady diet for the tormented self that feels forever powerless over experienced injustices. These relived experiences drive the person into a deep isolation where the self becomes less human—a thing, an object. This experience of losing oneself proves excruciating and often leads to contemplation of suicide as a way to resolve anguish.
Hope emerges
Pastoral care services provide a means to reframe one’s core beliefs, manage and resolve the burden of guilt, and uncover new motivation for living.
The practice of spiritual direction within the discipline of pastoral care listens for these inner movements and encourages the person to give voice to them in his or her own words. No longer limited by a diminished, tormented self, the real self begins to relate to another reality that changes his or her identity, relieves the burden of guilt, and gives reason, purpose, and meaning to life.
Even with this opportunity for a new life, however, cognitive distortions based on a disproportional “faith-based prism” may persist. In this case, clinicians and chaplains must work closely together to reframe old understandings of self and incorrect understandings of religion and spirituality into one that continues to reinforce this newfound sense of hope.38
A VETERAN OF IRAQ WITH SUICIDE IDEATION
The following case illustrates how clinicians and chaplains may be able to work together to help facilitate the resolution of guilt.
A veteran who had served in Iraq had entered the Domiciliary Care Program at a US Department of Veterans Affairs medical center. He reported experiencing problems with guilt, forgiveness, and suicide ideation. A clinical therapeutic program was prescribed after a psychological evaluation uncovered that he was also struggling with depression and posttraumatic stress disorder.
His mental healthcare providers recognized the importance of incorporating a religious-spiritual component into the therapeutic plan, and so consulted with a chaplain to plan a suitable course of action. Specifically, this veteran reported feeling that he could not be forgiven for his military experiences, a feeling that was giving way to alienation and isolation from the God of his faith tradition.
The chaplain helped this veteran reflect on his military experiences, giving him the perspective he needed to view his God as one who truly loves him. He recognized instances in which he could have lost his life had it not been for others who intervened on his behalf at just the right time. This awareness caused him to think about his life differently, challenging him to reframe his relationship with God. Instead of simple coincidences, the veteran began to consider the mystery behind these times and places.
Over time and in keeping with the tenets of his faith tradition, the veteran stated that he was ultimately able to accept and receive God’s love and forgiveness. He now reports that these inner spiritual movements serve as a source of support during occasional relapses into emotional distress. These movements allow him to consider the mystery of his present life and its value based on his experience of his God’s love and forgiveness.
CARE FOR SUICIDE SURVIVORS
The experience of guilt is not limited to veterans. Those bereaved by suicide are also left to manage their own experiences of the loss and ensuing complex emotions. Friends and loved ones who survive a suicide decedent may experience guilt, feeling that they somehow contributed to or failed to prevent the suicide. Such feelings of guilt are hypothesized to lower the threshold for suicidal behavior in those bereaved.39
Guilt and shame are also frequently encountered in survivors of nonfatal suicide attempts.40 Chaplaincy services might also prove useful for these individuals.
TIME IS EVERYTHING
Patients who may have an active psychopathology should have their clinical therapeutic needs attended to first. If the clinician deems pastoral care services to be an appropriate complementary support option, care should be taken to select a pastoral care provider who is adequately prepared for this role. Different professional organizations (eg, Association of Professional Chaplains) have established board-certification procedures, minimum education requirements, and supervised practical experience required for chaplaincy certification.
Also, spiritual growth and development remain a core focus of pastoral practice. Clinicians should discontinue any collaboration with pastoral care providers who question an individual’s faith or commitment to his or her faith, or who promote thinking or actions that could be deleterious to the patient’s therapeutic trajectory.
SUMMING UP
We have here presented our perspectives on how chaplaincy services can be used to complement clinical services in support of at-risk veterans struggling with experiences of guilt. Unfortunately, the current level of collaboration between chaplains and clinicians in support of at-risk veteran populations is limited.20 Our hope is that clinicians managing these at-risk patients will develop a greater awareness of how chaplaincy services might be able to help in alleviating experiences of guilt in at-risk veteran populations. A further hope is that such cases will serve as an opportunity for greater interdisciplinary collaboration, benefiting at-risk veterans most in need of support.
Acknowledgment: Dr. Rasmussen was supported by the Office of Academic Affiliations, Advanced Fellowship Program in Mental Illness Research and Treatment, US Department of Veterans Affairs, VISN 2 Center of Excellence for Suicide Prevention.
Suicidal behavior is a major cause of morbidity and mortality in the United States,1 and active-duty and reserve military personnel and veterans account for nearly 18% of suicide deaths.2 By one estimate, as many as 22 veterans die by suicide each day.3 These rates are thought to be due to a higher incidence of mental illness in certain veteran populations relative to the general population.4–8 Consequently, a number of mental health services are available to veterans in a variety of clinical and community settings.
Chaplains and clinicians bring complementary skills and services to the problem of suicide risk among veterans. In particular, helping at-risk veterans deal with experiences of guilt is an opportunity for interdisciplinary collaboration. Available literature supports the potential utility of chaplaincy services in supporting at-risk veteran populations.9–15
But while most healthcare facilities have chaplains on staff, there is little information to guide any such collaboration. Further, healthcare providers appear to have a limited understanding of chaplaincy services, the “language” within which chaplains operate, or the roles chaplains play in healthcare settings.16
In the following discussion, using the example of experiences of guilt, we offer our insights and suggestions on how chaplaincy services may prove useful in alleviating this complex emotion in veterans at risk of suicide.
BENEFITS OF TALKING TO A CHAPLAIN
Collaboration between healthcare providers and pastoral care professionals has been suggested as a means of enhancing the treatment of patients with mental illness.17,18 Chaplains draw from a variety of faith traditions and are usually trained to respond to the needs of people from a variety of religious and spiritual backgrounds. They provide some non-faithbased services (eg, crisis intervention, life review, bereavement counseling) resembling those also provided in formal mental healthcare settings.19 By facilitating religious and spiritual coping and religious practice and responding to religious and spiritual needs, chaplains also offer a level of support not typically offered by formal mental healthcare providers.20
Veterans at risk of suicide sometimes look to pastoral care providers, particularly chaplains, for mental health support.9,10 Research on the effects of chaplaincy services on suicidal behavior is just beginning to emerge.15 Still, the US Department of Health and Human Services has recognized pastoral care services as having a “beneficial and therapeutic effect on the medical condition of a patient.”11
For example, in one study, hospital inpatients reported higher satisfaction if they had been visited by a chaplain.12 Chaplains help align treatment plans with patient values and wishes.13 In another study,14 patients undergoing coronary artery bypass grafting who were randomized to receive five visits from a chaplain were found to have a higher rate of positive religious coping (eg, forgiveness, letting go of anger). Positive religious coping has been correlated with lower levels of psychological stress and better mental health outcomes.
EXPERIENCING GUILT IS LINKED TO RISK OF SUICIDE
Suicidal behavior is complex, multifaceted, and linked to genetic, neurologic, psychological, social, and cultural factors.21
Assessing for and addressing certain complex emotions, such as guilt and shame, is an important part of suicide prevention efforts. Guilt is defined as a “controllable psychological state that is typically linked to a specific action or behavior, and which entails regret or remorse.”22
Guilt has been linked to risk of suicide in veterans.23–25 In one study, close to 75% of veterans who had thought about suicide said they frequently experienced guilt about having violated the precepts of their faith group, family, God, life, or the military.26
Such findings suggest that the sense of guilt experienced by some at-risk veterans may be grounded in a variety of contexts. For example, faith communities that place a strong emphasis on obedience to moral, ethical, and religious precepts may contribute to the experience of guilt unless balanced by a message of grace or favor from a benevolent God or deity. Without this balance, engaging in activities that are not fully sanctioned by one’s faith community may lead to guilt.
Families might also contribute to veterans’ experiences of guilt by placing unrealistic expectations on them. And the family environment may not be conducive to resolving feelings of guilt in veterans, harboring resentment and antipathies and making it very difficult to alleviate any ensuing sense of distress.
CLINICIAN’S ROLE IN ASSESSING GUILT
In addressing and assessing guilt in veterans at risk of suicide, clinicians should try to recognize the source and clinical implications of this emotion.
Recognize the source of guilt
Guilt may indicate a clinical disorder such as a mood disorder (eg, major depression).27 Mood disorders significantly increase the risk of suicidal behavior.28,29
Beyond diagnosing a clinical disorder, prescribing pharmacotherapy, and referring for mental healthcare services, recognizing the source of this emotion remains an important part of addressing a patient’s experience of guilt. Especially when associated with a clinical disorder, guilt is often irrational and excessive and does not appropriately reflect the experience or situation in question.
Case conceptualization, defined as “synthesizing the patient’s experience with relevant clinical theory and research,”30 can be used to understand the context in which the guilt-inducing action or behavior occurred and the veteran’s own interpretation of his or her actions. Understanding the source of the patient’s guilt could be used to plan treatment and resolve any underlying sense of distress.
As with other negative emotions, the affective component of guilt is often the result of cognitive distortions made as the person tries to make sense of what has occurred or to reconcile beliefs of right and wrong with the guilt-inducing behavior.31 The common cognitive errors associated with guilt include:
- Hindsight bias (a belief that one should have known what was going to happen as a result of one’s actions)
- Responsibility distortion (a belief that one’s actions directly caused an adverse event)
- Justification distortion (a belief that one’s actions were not justified by the situation)
- Wrongdoing distortion (a belief that one violated one’s own standards of right and wrong).31
Cognitive therapy to counter cognitive distortions
A variety of clinical options exist to help veterans manage and resolve guilt.
Cognitive therapy can counter the cognitive distortions that drive feelings of guilt. The goal is to guide patients to examine the evidence, process the event, and realize that their behavior was appropriate for the given situation. Cognitive processing therapy and prolonged exposure therapy have both been shown to decrease trauma-related guilt, though cognitive processing therapy was found to be better at decreasing guilt that arose from cognitive distortions.32
Guilt and suicide ideation have also been associated with a belief that one’s actions constituted an unforgivable sin.33 Responding to these inherently religious-spiritual cognitive distortions may be beyond the scope of expertise for many healthcare professionals. In such cases, it may be prudent to consider complementing clinical services with pastoral care. It follows that pastoral care services should only be provided if the veteran voices a desire and readiness for them. The clinician and chaplain can then work together to provide coordinated care to best meet the patient’s needs, to address the experience of guilt, and to alleviate the sense of distress.
A CHAPLAIN’S PERSPECTIVE ON GUILT
A prominent feature of pastoral practice is helping people, including at-risk veterans, resolve feelings of guilt regardless of the context on which the emotion is founded (eg, religion, shame).10 For many people, guilt is an impenetrable barrier, preventing resolution of whatever experience led to a sense of inner turmoil.
Forgiveness
In the context of pastoral care, resolution of guilt is ordinarily tied to a need for forgiveness. There are multiple ways in which forgiveness can be grounded in religious and spiritual contexts.34 Examples include forgiving others (ie, forswearing resentment, anger, or hatred directed toward another person), being forgiven by God or another benevolent deity, and forgiving oneself for violating perceived personal transgressions.35 In some cases, divine forgiveness may be conditional on interpersonal forgiveness.36 Forgiveness is also sometimes seen as a remedy for sin and a way to restore moral order.37
Some people may initially think they can never be forgiven. With time and the weight of one’s experiences, the impossibility of forgiveness can become so ingrained that it becomes a core belief. These core beliefs set up a vicious circle of thoughts and feelings, in which people and places and events from the past are continuously brought forward into the present. Anger and resentment become the steady diet for the tormented self that feels forever powerless over experienced injustices. These relived experiences drive the person into a deep isolation where the self becomes less human—a thing, an object. This experience of losing oneself proves excruciating and often leads to contemplation of suicide as a way to resolve anguish.
Hope emerges
Pastoral care services provide a means to reframe one’s core beliefs, manage and resolve the burden of guilt, and uncover new motivation for living.
The practice of spiritual direction within the discipline of pastoral care listens for these inner movements and encourages the person to give voice to them in his or her own words. No longer limited by a diminished, tormented self, the real self begins to relate to another reality that changes his or her identity, relieves the burden of guilt, and gives reason, purpose, and meaning to life.
Even with this opportunity for a new life, however, cognitive distortions based on a disproportional “faith-based prism” may persist. In this case, clinicians and chaplains must work closely together to reframe old understandings of self and incorrect understandings of religion and spirituality into one that continues to reinforce this newfound sense of hope.38
A VETERAN OF IRAQ WITH SUICIDE IDEATION
The following case illustrates how clinicians and chaplains may be able to work together to help facilitate the resolution of guilt.
A veteran who had served in Iraq had entered the Domiciliary Care Program at a US Department of Veterans Affairs medical center. He reported experiencing problems with guilt, forgiveness, and suicide ideation. A clinical therapeutic program was prescribed after a psychological evaluation uncovered that he was also struggling with depression and posttraumatic stress disorder.
His mental healthcare providers recognized the importance of incorporating a religious-spiritual component into the therapeutic plan, and so consulted with a chaplain to plan a suitable course of action. Specifically, this veteran reported feeling that he could not be forgiven for his military experiences, a feeling that was giving way to alienation and isolation from the God of his faith tradition.
The chaplain helped this veteran reflect on his military experiences, giving him the perspective he needed to view his God as one who truly loves him. He recognized instances in which he could have lost his life had it not been for others who intervened on his behalf at just the right time. This awareness caused him to think about his life differently, challenging him to reframe his relationship with God. Instead of simple coincidences, the veteran began to consider the mystery behind these times and places.
Over time and in keeping with the tenets of his faith tradition, the veteran stated that he was ultimately able to accept and receive God’s love and forgiveness. He now reports that these inner spiritual movements serve as a source of support during occasional relapses into emotional distress. These movements allow him to consider the mystery of his present life and its value based on his experience of his God’s love and forgiveness.
CARE FOR SUICIDE SURVIVORS
The experience of guilt is not limited to veterans. Those bereaved by suicide are also left to manage their own experiences of the loss and ensuing complex emotions. Friends and loved ones who survive a suicide decedent may experience guilt, feeling that they somehow contributed to or failed to prevent the suicide. Such feelings of guilt are hypothesized to lower the threshold for suicidal behavior in those bereaved.39
Guilt and shame are also frequently encountered in survivors of nonfatal suicide attempts.40 Chaplaincy services might also prove useful for these individuals.
TIME IS EVERYTHING
Patients who may have an active psychopathology should have their clinical therapeutic needs attended to first. If the clinician deems pastoral care services to be an appropriate complementary support option, care should be taken to select a pastoral care provider who is adequately prepared for this role. Different professional organizations (eg, Association of Professional Chaplains) have established board-certification procedures, minimum education requirements, and supervised practical experience required for chaplaincy certification.
Also, spiritual growth and development remain a core focus of pastoral practice. Clinicians should discontinue any collaboration with pastoral care providers who question an individual’s faith or commitment to his or her faith, or who promote thinking or actions that could be deleterious to the patient’s therapeutic trajectory.
SUMMING UP
We have here presented our perspectives on how chaplaincy services can be used to complement clinical services in support of at-risk veterans struggling with experiences of guilt. Unfortunately, the current level of collaboration between chaplains and clinicians in support of at-risk veteran populations is limited.20 Our hope is that clinicians managing these at-risk patients will develop a greater awareness of how chaplaincy services might be able to help in alleviating experiences of guilt in at-risk veteran populations. A further hope is that such cases will serve as an opportunity for greater interdisciplinary collaboration, benefiting at-risk veterans most in need of support.
Acknowledgment: Dr. Rasmussen was supported by the Office of Academic Affiliations, Advanced Fellowship Program in Mental Illness Research and Treatment, US Department of Veterans Affairs, VISN 2 Center of Excellence for Suicide Prevention.
- Centers for Disease Control and Prevention (CDC). Suicide and self-inflicted injury. www.cdc.gov/nchs/fastats/suicide.htm. Accessed November 12, 2015.
- Centers for Disease Control and Prevention (CDC). National violent death reporting system (NVDRS). https://wisqars.cdc.gov:8443/nvdrs/nvdrsDisplay.jsp. Accessed November 12, 2015.
- Kemp JE, Bossarte R. Suicide data report, 2012. www.sprc.org/library_resources/items/suicide-data-report-2012. Accessed November 12, 2015.
- Bullman TA, Kang HK. The risk of suicide among wounded Vietnam veterans. Am J Public Health 1996; 86:662–667.
- Kang HK, Bullman TA. Is there an epidemic of suicides among current and former US military personnel? Ann Epidemiol 2009; 19:757–760.
- LeardMann CA, Powell TM, Smith TC, et al. Risk factors associated with suicide in current and former US military personnel. JAMA 2013; 310:496–506.
- Mrnak-Meyer J, Tate SR, Tripp JC, Worley MJ, Jajodia A, McQuaid JR. Predictors of suicide-related hospitalization among US veterans receiving treatment for comorbid depression and substance dependence: who is the riskiest of the risky? Suicide Life Threat Behav 2011; 41:532–542.
- Pietrzak RH, Russo AR, Ling Q, Southwick SM. Suicidal ideation in treatment-seeking veterans of Operations Enduring Freedom and Iraqi Freedom: the role of coping strategies, resilience, and social support. J Psychiatr Res 2011; 45:720–726.
- Kopacz MS, McCarten JM, Pollitt MJ. VHA chaplaincy contact with veterans at increased risk of suicide. South Med J 2014; 107: 661–664.
- Kopacz MS. Providing pastoral care services in a clinical setting to veterans at-risk of suicide. J Relig Health 2013; 52:759–767.
- Medicare program; payment for nursing and allied health education. Health Care Financing Administration (HCFA), HHS. Final rule. Fed Regist 2001; 66:3358–3376.
- Marin DB, Sharma V, Sosunov E, Egorova N, Goldstein R, Handzo GF. Relationship between chaplain visits and patient satisfaction. J Health Care Chaplain 2015; 21:14–24.
- Flannelly KJ, Emanuel LL, Handzo GF, Galek K, Silton NR, Carlson M. A national study of chaplaincy services and end-of-life outcomes. BMC Palliat Care 2012; 11:10.
- Bay PS, Beckman D, Trippi J, Gunderman R, Terry C. The effect of pastoral care services on anxiety, depression, hope, religious coping, and religious problem solving styles: a randomized controlled study. J Relig Health 2008; 47:57–69.
- Kopacz MS, Nieuwsma JA, Jackson GL, et al. Chaplains’ engagement with suicidality among their service users: findings from the VA/DoD Integrated Mental Health Strategy. Suicide Life Threat Behav 2015. [Epub ahead of print.]
- Flannelly KJ, Galek K, Bucchino J, Handzo GF, Tannenbaum HP. Department directors’ perceptions of the roles and functions of hospital chaplains: a national survey. Hosp Top 2005; 83:19–27.
- Farrell JL, Goebert DA. Collaboration between psychiatrists and clergy in recognizing and treating serious mental illness. Psychiatr Serv 2008; 59:437–440.
- Weaver AJ, Flannelly KJ, Flannelly LT, Oppenheimer JE. Collaboration between clergy and mental health professionals: a review of professional health care journals from 1980 through 1999. Counsel Val 2003; 47:162–171.
- Handzo GF, Flannelly KJ, Kudler T, et al. What do chaplains really do? II. Interventions in the New York chaplaincy study. J Health Care Chaplain 2008; 14:39–56.
- Kopacz MS, Pollitt MJ. Delivering chaplaincy services to veterans at increased risk of suicide. J Health Care Chaplain 2015; 21:1–13.
- Knox KL, Bossarte RM. Suicide prevention for veterans and active duty personnel. Am J Public Health 2012;102(suppl 1):S8–S9.
- Bryan CJ, Morrow CE, Etienne N, Ray-Sannerud B. Guilt, shame, and suicidal ideation in a military outpatient clinical sample. Depress Anxiety 2013; 30:55–60.
- Ganz D, Sher L. Educating medical professionals about suicide prevention among military veterans. Int J Adolesc Med Health 2013; 25:187–191.
- Hendin H, Haas AP. Suicide and guilt as manifestations of PTSD in Vietnam combat veterans. Am J Psychiatry 1991; 148:586–591.
- Maguen S, Metzler TJ, Bosch J, Marmar CR, Knight SJ, Neylan TC. Killing in combat may be independently associated with suicidal ideation. Depress Anxiety 2012; 29:918–923.
- Kopacz MS, McCarten JM, Vance CG, Connery AL. A preliminary study for exploring different sources of guilt in a sample of veterans who sought chaplaincy services. Mil Psychol 2015; 27:1–8.
- Buck CJ. 2013 ICD-9-CM for physicians. St. Louis, MO: Saunders; 2013.
- Angst F, Stassen HH, Clayton PJ, Angst J. Mortality of patients with mood disorders: follow-up over 34-38 years. J Affect Disord 2002; 68:167–181.
- Nierenberg AA, Gray SM, Grandin LD. Mood disorders and suicide. J Clin Psychiatry 2001; 62(suppl 25):27–30.
- Macneil CA, Hasty MK, Conus P, Berk M. Is diagnosis enough to guide interventions in mental health? Using case formulation in clinical practice. BMC Med 2012; 10:111.
- Kubany ES, Manke FP. Cognitive therapy for trauma-related guilt: conceptual bases and treatment outlines. Cogn Behav Pract 1995; 2:27–61.
- Resick PA, Nishith P, Weaver TL, Astin MC, Feuer CA. Comparison of cognitive-processing therapy with prolonged exposure and a waiting condition for the treatment of chronic posttraumatic stress disorder in female rape victims. J Consult Clin Psychol 2002; 70:867–879.
- Exline JJ, Yali AM, Sanderson WC. Guilt, discord, and alienation: the role of religious strain in depression and suicidality. J Clin Psychol 2000; 56:1481–1496.
- Musick MA. Multiple forms of forgiveness and their relationship with aging and religion, In: Schaie KW, Krause N, Booth A, editors. Religious Influences on Health and Well-being in the Elderly. New York, NY: Springer Publishing Company; 2004:202–214.
- Kaplan BH, Munroe-Blum H, Blazer DG. Religion, health and forgiveness: tradition and challenges. In: Levin JS, editor. Religion in Aging and Health. Theoretical Foundations and Methodological Frontiers. Thousand Oaks, CA: SAGE Focus Edition; 1994:52–77.
- Worthington EL Jr, Berry JW, Parrott L III. Unforgiveness, forgiveness, religion and health. In: Plante TG, Sherman AC, editors. Faith and Health. Psychological Perspectives. New York, NY: Guilford Press; 2001:107–138.
- Enright RD, Gassin EA, Wu GR. Forgiveness: a developmental view. J Moral Educ 1992; 21:99–114.
- Kopacz MS, O’Reilly LM, Van Inwagen CC, et al. Understanding the role of chaplains in veteran suicide prevention efforts: a discussion paper. SAGE Open 2014; 4:1–10.
- Young IT, Iglewicz A, Glorioso D, et al. Suicide bereavement and complicated grief. Dialogues Clin Neurosci 2012; 14:177–186.
- Wiklander M, Samuelsson M, Asberg M. Shame reactions after suicide attempt. Scand J Caring Sci 2003; 17:293–300.
- Centers for Disease Control and Prevention (CDC). Suicide and self-inflicted injury. www.cdc.gov/nchs/fastats/suicide.htm. Accessed November 12, 2015.
- Centers for Disease Control and Prevention (CDC). National violent death reporting system (NVDRS). https://wisqars.cdc.gov:8443/nvdrs/nvdrsDisplay.jsp. Accessed November 12, 2015.
- Kemp JE, Bossarte R. Suicide data report, 2012. www.sprc.org/library_resources/items/suicide-data-report-2012. Accessed November 12, 2015.
- Bullman TA, Kang HK. The risk of suicide among wounded Vietnam veterans. Am J Public Health 1996; 86:662–667.
- Kang HK, Bullman TA. Is there an epidemic of suicides among current and former US military personnel? Ann Epidemiol 2009; 19:757–760.
- LeardMann CA, Powell TM, Smith TC, et al. Risk factors associated with suicide in current and former US military personnel. JAMA 2013; 310:496–506.
- Mrnak-Meyer J, Tate SR, Tripp JC, Worley MJ, Jajodia A, McQuaid JR. Predictors of suicide-related hospitalization among US veterans receiving treatment for comorbid depression and substance dependence: who is the riskiest of the risky? Suicide Life Threat Behav 2011; 41:532–542.
- Pietrzak RH, Russo AR, Ling Q, Southwick SM. Suicidal ideation in treatment-seeking veterans of Operations Enduring Freedom and Iraqi Freedom: the role of coping strategies, resilience, and social support. J Psychiatr Res 2011; 45:720–726.
- Kopacz MS, McCarten JM, Pollitt MJ. VHA chaplaincy contact with veterans at increased risk of suicide. South Med J 2014; 107: 661–664.
- Kopacz MS. Providing pastoral care services in a clinical setting to veterans at-risk of suicide. J Relig Health 2013; 52:759–767.
- Medicare program; payment for nursing and allied health education. Health Care Financing Administration (HCFA), HHS. Final rule. Fed Regist 2001; 66:3358–3376.
- Marin DB, Sharma V, Sosunov E, Egorova N, Goldstein R, Handzo GF. Relationship between chaplain visits and patient satisfaction. J Health Care Chaplain 2015; 21:14–24.
- Flannelly KJ, Emanuel LL, Handzo GF, Galek K, Silton NR, Carlson M. A national study of chaplaincy services and end-of-life outcomes. BMC Palliat Care 2012; 11:10.
- Bay PS, Beckman D, Trippi J, Gunderman R, Terry C. The effect of pastoral care services on anxiety, depression, hope, religious coping, and religious problem solving styles: a randomized controlled study. J Relig Health 2008; 47:57–69.
- Kopacz MS, Nieuwsma JA, Jackson GL, et al. Chaplains’ engagement with suicidality among their service users: findings from the VA/DoD Integrated Mental Health Strategy. Suicide Life Threat Behav 2015. [Epub ahead of print.]
- Flannelly KJ, Galek K, Bucchino J, Handzo GF, Tannenbaum HP. Department directors’ perceptions of the roles and functions of hospital chaplains: a national survey. Hosp Top 2005; 83:19–27.
- Farrell JL, Goebert DA. Collaboration between psychiatrists and clergy in recognizing and treating serious mental illness. Psychiatr Serv 2008; 59:437–440.
- Weaver AJ, Flannelly KJ, Flannelly LT, Oppenheimer JE. Collaboration between clergy and mental health professionals: a review of professional health care journals from 1980 through 1999. Counsel Val 2003; 47:162–171.
- Handzo GF, Flannelly KJ, Kudler T, et al. What do chaplains really do? II. Interventions in the New York chaplaincy study. J Health Care Chaplain 2008; 14:39–56.
- Kopacz MS, Pollitt MJ. Delivering chaplaincy services to veterans at increased risk of suicide. J Health Care Chaplain 2015; 21:1–13.
- Knox KL, Bossarte RM. Suicide prevention for veterans and active duty personnel. Am J Public Health 2012;102(suppl 1):S8–S9.
- Bryan CJ, Morrow CE, Etienne N, Ray-Sannerud B. Guilt, shame, and suicidal ideation in a military outpatient clinical sample. Depress Anxiety 2013; 30:55–60.
- Ganz D, Sher L. Educating medical professionals about suicide prevention among military veterans. Int J Adolesc Med Health 2013; 25:187–191.
- Hendin H, Haas AP. Suicide and guilt as manifestations of PTSD in Vietnam combat veterans. Am J Psychiatry 1991; 148:586–591.
- Maguen S, Metzler TJ, Bosch J, Marmar CR, Knight SJ, Neylan TC. Killing in combat may be independently associated with suicidal ideation. Depress Anxiety 2012; 29:918–923.
- Kopacz MS, McCarten JM, Vance CG, Connery AL. A preliminary study for exploring different sources of guilt in a sample of veterans who sought chaplaincy services. Mil Psychol 2015; 27:1–8.
- Buck CJ. 2013 ICD-9-CM for physicians. St. Louis, MO: Saunders; 2013.
- Angst F, Stassen HH, Clayton PJ, Angst J. Mortality of patients with mood disorders: follow-up over 34-38 years. J Affect Disord 2002; 68:167–181.
- Nierenberg AA, Gray SM, Grandin LD. Mood disorders and suicide. J Clin Psychiatry 2001; 62(suppl 25):27–30.
- Macneil CA, Hasty MK, Conus P, Berk M. Is diagnosis enough to guide interventions in mental health? Using case formulation in clinical practice. BMC Med 2012; 10:111.
- Kubany ES, Manke FP. Cognitive therapy for trauma-related guilt: conceptual bases and treatment outlines. Cogn Behav Pract 1995; 2:27–61.
- Resick PA, Nishith P, Weaver TL, Astin MC, Feuer CA. Comparison of cognitive-processing therapy with prolonged exposure and a waiting condition for the treatment of chronic posttraumatic stress disorder in female rape victims. J Consult Clin Psychol 2002; 70:867–879.
- Exline JJ, Yali AM, Sanderson WC. Guilt, discord, and alienation: the role of religious strain in depression and suicidality. J Clin Psychol 2000; 56:1481–1496.
- Musick MA. Multiple forms of forgiveness and their relationship with aging and religion, In: Schaie KW, Krause N, Booth A, editors. Religious Influences on Health and Well-being in the Elderly. New York, NY: Springer Publishing Company; 2004:202–214.
- Kaplan BH, Munroe-Blum H, Blazer DG. Religion, health and forgiveness: tradition and challenges. In: Levin JS, editor. Religion in Aging and Health. Theoretical Foundations and Methodological Frontiers. Thousand Oaks, CA: SAGE Focus Edition; 1994:52–77.
- Worthington EL Jr, Berry JW, Parrott L III. Unforgiveness, forgiveness, religion and health. In: Plante TG, Sherman AC, editors. Faith and Health. Psychological Perspectives. New York, NY: Guilford Press; 2001:107–138.
- Enright RD, Gassin EA, Wu GR. Forgiveness: a developmental view. J Moral Educ 1992; 21:99–114.
- Kopacz MS, O’Reilly LM, Van Inwagen CC, et al. Understanding the role of chaplains in veteran suicide prevention efforts: a discussion paper. SAGE Open 2014; 4:1–10.
- Young IT, Iglewicz A, Glorioso D, et al. Suicide bereavement and complicated grief. Dialogues Clin Neurosci 2012; 14:177–186.
- Wiklander M, Samuelsson M, Asberg M. Shame reactions after suicide attempt. Scand J Caring Sci 2003; 17:293–300.
Many shades of guilt
In their commentary, Kopacz et al1 propose that collaboration with professionally trained and certified clinical chaplains provides an opportunity for interdisciplinary care with increased benefit to veterans at risk of suicide. They rightly identify the pivotal issue of guilt as one that falls squarely in the domain of spiritual (or pastoral) care.
As professionals involved in the training of board-certifiable chaplains (and one of us is a veteran), we find that guilt in patients with suicidal tendencies is a profoundly spiritual issue that can be addressed effectively through collaboration among chaplains, physicians, and mental health providers.
Guilt is a serious spiritual condition that can easily be undertreated, or treated under the rubric of depression, which is related but not identical. Undertreatment occurs when caregivers, eager to see the guilt-sufferer experience relief, inadvertently short-circuit the necessary process of working through, rather than around, the guilt. Allowing patients to talk about their feelings of guilt without minimizing those feelings can be helpful even if, as Kopacz et al point out, the feelings are often irrational. We believe that people have an innate need to be truly heard and understood before they can become open to a reinterpretation of their feelings. Only then can the seeds of self-forgiveness begin to take root.
Hearing the words “There is hope for you to feel forgiven” can be more helpful than hearing “You didn’t really do anything bad,” particularly if the patient is religious. Hearing these words from a chaplain is often more effective than hearing them from a lay person, just as many of us take basic health information more seriously when we hear it from a physician. Even if the veteran is not overtly religious, there may be a unique exchange between that person and a religious authority when it concerns the violation of a millenniaold, widely known teaching from the Bible, such as “Thou shalt not kill.”
Kopacz et al also rightly suggest that unless religious prohibitions have been balanced with teachings on forgiveness and grace, the teachings can actually exacerbate feelings of guilt and elevate them to harmful proportions, especially in the potentially vulnerable psyche of a veteran who may have been traumatized. If there is no religious or spiritual guidance for balancing prohibitions with graces, the patient may be left to spiral in an unending loop of guilt with no way out.
We therefore propose the following categories for different types (or “shades”) of guilt that can be effectively addressed by chaplains in concert with other members of the healthcare team. For simplicity, we call these types real guilt, survivor guilt, mistaken guilt, and complex-compound guilt.
REAL GUILT
An important role professional chaplains can play is to allow patients (in this case, veterans) to express their remorse and regret for violations of their own moral codes. In many cases, they have in fact hurt or killed another person, and they need the chance to unburden their hearts and spirits, especially if they were taught that killing people is a sin. Veterans who have harmed or killed others, even if under orders, are often left with bona fide feelings of guilt that need to be aired and released in a safe and confidential environment. This is often most effective when done by someone who not only is trained in nonjudgmental and nondirective listening, but also is a religious authority who can assure the patient of his or her innate worthiness and of the ability to be forgiven.
As Kopacz et al note, guilt is linked to a specific action or behavior and usually entails regret or remorse. Many veterans belong to or have had exposure to faith groups with strong moral codes and prohibitions, and so may see the chaplain as having authority to act as confessor and granter of absolution.
SURVIVOR GUILT
Survivor guilt is commonly understood as the feeling of surviving a terrible event or situation while someone else did not. Those who suffer from survivor guilt judge themselves unworthy of survival and believe the deceased to have been more courageous, virtuous, or somehow a better person than they. They torture themselves with ideas of the deceased person’s virtues—imagined or real—and sometimes go on to believe that “It should have been me who was killed.”
The burden of feeling that the wrong person died can be overwhelming. If these people are not helped to see their own worth and helped to find outlets for their sense of having been spared (by God, by their own wits, or by sheer luck), they are likely to struggle more. This is related but not identical to what we call mistaken guilt. The two types are similar because they share a sense of randomness and helplessness, but they are different for reasons we will explain below.
Chaplains can be particularly effective partners in the care of veterans with survivor guilt, helping them to make meaning out of a life-changing event, rather than find meaning inherently in that event. Meaning, purpose, and “God’s plan for my life” are common themes in the pastoral conversation that can provide a compass for the disoriented survivor.
MISTAKEN GUILT
Mistaken guilt describes when a person who is involved in the death of another but is absolutely blameless—and could not possibly have prevented that death—literally “mis-takes” the guilt upon himself or herself in spite of the facts. Because of the helplessness induced by this feeling, mistaken guilt can be more difficult to treat than other forms. These patients continue to suffer despite assurances that the death occurred through absolutely no fault of their own.2
Hickling3 has written extensively about this phenomenon in innocent motor vehicle drivers who cause pedestrian deaths, and he considers this type of guilt one of the most difficult to recover from precisely because of the helplessness factor. He has explained that if patients can find a real reason by which they were culpable for what happened, they can change their ways. But if they were absolutely innocent (as in many incidents in training or combat), they often cannot make sense of what happened in a way that allows them to move on because there is nothing they could have done differently and therefore nothing they can change.4
These patients almost certainly need long-term intervention such as cognitive behavioral therapy in order to train their mind away from such destructive thoughts. However, they are also very likely to be helped by a chaplain if they find that the event triggers memories of other past infractions of which they may need to unburden themselves (ie, confess).
COMPOUND-COMPLEX GUILT
As the name implies, compound-complex guilt is a combination of the other types and may have additional layers.
Compound-complex guilt leaves sufferers literally feeling guilty for feeling guilty. Though this may border on a genuine clinical disorder, it is also to some degree normal (eg, due to cultural taboos and norms) for people to feel culpable for not being able to “move on” or “forgive themselves” as quickly as others may want them to. Buddhists call this tendency the “second arrow effect.” The first arrow is the feeling of guilt (or other painful feeling) that strikes the individual, but the second arrow is the one he or she drives in afterward by thinking it is wrong or weak to even have the feeling.
Patients who suffer from this type of guilt blame themselves for the conundrum they are in and feel even worse. This is not unlike the vortex of unresolved and complicated grief.
Those who suffer from compound-complex guilt may layer the primary guilt with additional guilt for feeling weak, for needing help, or for asking for help. Especially in the culture of the military, the fear of stigma when asking for help (especially with mental health) is still quite strong. Therefore, chaplains can serve as a less threatening entry point for the veteran needing multiple professionals involved in his or her care.
NONJUDGMENTAL LISTENING
Nonjudgmental listening is essential to get at the source or sources of guilt, regardless of the type, in order to allow the wounds to air out and begin healing. Many veterans suffering from guilt may need intensive pharmacologic and cognitive therapy to fully recover, and care from a chaplain is not a substitute for psychiatric evaluation and treatment, especially if there is a risk of suicide.
However, chaplains may be able to help with the “deep work” of spiritual healing that is part of veterans’ overall recovery. This is true not only because chaplains are especially trained to do this, but also because they are the team members most likely to have uniquely spiritual language to speak to the condition. The language of confession, absolution, repentance, redemption, atonement, and forgiveness is language of the spiritual realm.
In addition, chaplains’ freedom from hourly billing concerns and their often less formalized interactions with patients may help to build trust. Well-trained chaplains, who are often quite gifted at creating an atmosphere of reverence and safety (sanctuary) in the most unlikely situations, are well suited to help the interdisciplinary team treat this vulnerable patient population.
SUGGESTED READING
For more insights into the role of chaplains on the interdisciplinary healthcare team, we recommend the following book: Cadge W. Paging God: Religion in the Halls of Medicine. Chicago, IL: University of Chicago Press; 2012.
- Kopacz MS, Rasmussen KA, Searle RF, Wozniak BM, Titus CE. Veterans, guilt, and suicide risk: chaplains can help. Cleve Clin J Med 2016; 83:101–105.
- Life after death: Act one—guilty as not charged. Darin Strauss. This American Life. www.thisamericanlife.org. Episode 359. Aired July 18, 2008. www.thisamericanlife.org/radio-archives/episode/359/life-after-death?act=1#play. Accessed December 10, 2015.
- Hickling EJ, Blanchard EB. Overcoming the Trauma of Your Motor Vehicle Accident: A Cognitive-behavioral Treatment Program. New York, NY: Oxford University Press; 2006.
- Hickling EJ. Transforming Tragedy: Finding Growth Following Life’s Traumas. North Charleston, SC: CreateSpace; 2012.
In their commentary, Kopacz et al1 propose that collaboration with professionally trained and certified clinical chaplains provides an opportunity for interdisciplinary care with increased benefit to veterans at risk of suicide. They rightly identify the pivotal issue of guilt as one that falls squarely in the domain of spiritual (or pastoral) care.
As professionals involved in the training of board-certifiable chaplains (and one of us is a veteran), we find that guilt in patients with suicidal tendencies is a profoundly spiritual issue that can be addressed effectively through collaboration among chaplains, physicians, and mental health providers.
Guilt is a serious spiritual condition that can easily be undertreated, or treated under the rubric of depression, which is related but not identical. Undertreatment occurs when caregivers, eager to see the guilt-sufferer experience relief, inadvertently short-circuit the necessary process of working through, rather than around, the guilt. Allowing patients to talk about their feelings of guilt without minimizing those feelings can be helpful even if, as Kopacz et al point out, the feelings are often irrational. We believe that people have an innate need to be truly heard and understood before they can become open to a reinterpretation of their feelings. Only then can the seeds of self-forgiveness begin to take root.
Hearing the words “There is hope for you to feel forgiven” can be more helpful than hearing “You didn’t really do anything bad,” particularly if the patient is religious. Hearing these words from a chaplain is often more effective than hearing them from a lay person, just as many of us take basic health information more seriously when we hear it from a physician. Even if the veteran is not overtly religious, there may be a unique exchange between that person and a religious authority when it concerns the violation of a millenniaold, widely known teaching from the Bible, such as “Thou shalt not kill.”
Kopacz et al also rightly suggest that unless religious prohibitions have been balanced with teachings on forgiveness and grace, the teachings can actually exacerbate feelings of guilt and elevate them to harmful proportions, especially in the potentially vulnerable psyche of a veteran who may have been traumatized. If there is no religious or spiritual guidance for balancing prohibitions with graces, the patient may be left to spiral in an unending loop of guilt with no way out.
We therefore propose the following categories for different types (or “shades”) of guilt that can be effectively addressed by chaplains in concert with other members of the healthcare team. For simplicity, we call these types real guilt, survivor guilt, mistaken guilt, and complex-compound guilt.
REAL GUILT
An important role professional chaplains can play is to allow patients (in this case, veterans) to express their remorse and regret for violations of their own moral codes. In many cases, they have in fact hurt or killed another person, and they need the chance to unburden their hearts and spirits, especially if they were taught that killing people is a sin. Veterans who have harmed or killed others, even if under orders, are often left with bona fide feelings of guilt that need to be aired and released in a safe and confidential environment. This is often most effective when done by someone who not only is trained in nonjudgmental and nondirective listening, but also is a religious authority who can assure the patient of his or her innate worthiness and of the ability to be forgiven.
As Kopacz et al note, guilt is linked to a specific action or behavior and usually entails regret or remorse. Many veterans belong to or have had exposure to faith groups with strong moral codes and prohibitions, and so may see the chaplain as having authority to act as confessor and granter of absolution.
SURVIVOR GUILT
Survivor guilt is commonly understood as the feeling of surviving a terrible event or situation while someone else did not. Those who suffer from survivor guilt judge themselves unworthy of survival and believe the deceased to have been more courageous, virtuous, or somehow a better person than they. They torture themselves with ideas of the deceased person’s virtues—imagined or real—and sometimes go on to believe that “It should have been me who was killed.”
The burden of feeling that the wrong person died can be overwhelming. If these people are not helped to see their own worth and helped to find outlets for their sense of having been spared (by God, by their own wits, or by sheer luck), they are likely to struggle more. This is related but not identical to what we call mistaken guilt. The two types are similar because they share a sense of randomness and helplessness, but they are different for reasons we will explain below.
Chaplains can be particularly effective partners in the care of veterans with survivor guilt, helping them to make meaning out of a life-changing event, rather than find meaning inherently in that event. Meaning, purpose, and “God’s plan for my life” are common themes in the pastoral conversation that can provide a compass for the disoriented survivor.
MISTAKEN GUILT
Mistaken guilt describes when a person who is involved in the death of another but is absolutely blameless—and could not possibly have prevented that death—literally “mis-takes” the guilt upon himself or herself in spite of the facts. Because of the helplessness induced by this feeling, mistaken guilt can be more difficult to treat than other forms. These patients continue to suffer despite assurances that the death occurred through absolutely no fault of their own.2
Hickling3 has written extensively about this phenomenon in innocent motor vehicle drivers who cause pedestrian deaths, and he considers this type of guilt one of the most difficult to recover from precisely because of the helplessness factor. He has explained that if patients can find a real reason by which they were culpable for what happened, they can change their ways. But if they were absolutely innocent (as in many incidents in training or combat), they often cannot make sense of what happened in a way that allows them to move on because there is nothing they could have done differently and therefore nothing they can change.4
These patients almost certainly need long-term intervention such as cognitive behavioral therapy in order to train their mind away from such destructive thoughts. However, they are also very likely to be helped by a chaplain if they find that the event triggers memories of other past infractions of which they may need to unburden themselves (ie, confess).
COMPOUND-COMPLEX GUILT
As the name implies, compound-complex guilt is a combination of the other types and may have additional layers.
Compound-complex guilt leaves sufferers literally feeling guilty for feeling guilty. Though this may border on a genuine clinical disorder, it is also to some degree normal (eg, due to cultural taboos and norms) for people to feel culpable for not being able to “move on” or “forgive themselves” as quickly as others may want them to. Buddhists call this tendency the “second arrow effect.” The first arrow is the feeling of guilt (or other painful feeling) that strikes the individual, but the second arrow is the one he or she drives in afterward by thinking it is wrong or weak to even have the feeling.
Patients who suffer from this type of guilt blame themselves for the conundrum they are in and feel even worse. This is not unlike the vortex of unresolved and complicated grief.
Those who suffer from compound-complex guilt may layer the primary guilt with additional guilt for feeling weak, for needing help, or for asking for help. Especially in the culture of the military, the fear of stigma when asking for help (especially with mental health) is still quite strong. Therefore, chaplains can serve as a less threatening entry point for the veteran needing multiple professionals involved in his or her care.
NONJUDGMENTAL LISTENING
Nonjudgmental listening is essential to get at the source or sources of guilt, regardless of the type, in order to allow the wounds to air out and begin healing. Many veterans suffering from guilt may need intensive pharmacologic and cognitive therapy to fully recover, and care from a chaplain is not a substitute for psychiatric evaluation and treatment, especially if there is a risk of suicide.
However, chaplains may be able to help with the “deep work” of spiritual healing that is part of veterans’ overall recovery. This is true not only because chaplains are especially trained to do this, but also because they are the team members most likely to have uniquely spiritual language to speak to the condition. The language of confession, absolution, repentance, redemption, atonement, and forgiveness is language of the spiritual realm.
In addition, chaplains’ freedom from hourly billing concerns and their often less formalized interactions with patients may help to build trust. Well-trained chaplains, who are often quite gifted at creating an atmosphere of reverence and safety (sanctuary) in the most unlikely situations, are well suited to help the interdisciplinary team treat this vulnerable patient population.
SUGGESTED READING
For more insights into the role of chaplains on the interdisciplinary healthcare team, we recommend the following book: Cadge W. Paging God: Religion in the Halls of Medicine. Chicago, IL: University of Chicago Press; 2012.
In their commentary, Kopacz et al1 propose that collaboration with professionally trained and certified clinical chaplains provides an opportunity for interdisciplinary care with increased benefit to veterans at risk of suicide. They rightly identify the pivotal issue of guilt as one that falls squarely in the domain of spiritual (or pastoral) care.
As professionals involved in the training of board-certifiable chaplains (and one of us is a veteran), we find that guilt in patients with suicidal tendencies is a profoundly spiritual issue that can be addressed effectively through collaboration among chaplains, physicians, and mental health providers.
Guilt is a serious spiritual condition that can easily be undertreated, or treated under the rubric of depression, which is related but not identical. Undertreatment occurs when caregivers, eager to see the guilt-sufferer experience relief, inadvertently short-circuit the necessary process of working through, rather than around, the guilt. Allowing patients to talk about their feelings of guilt without minimizing those feelings can be helpful even if, as Kopacz et al point out, the feelings are often irrational. We believe that people have an innate need to be truly heard and understood before they can become open to a reinterpretation of their feelings. Only then can the seeds of self-forgiveness begin to take root.
Hearing the words “There is hope for you to feel forgiven” can be more helpful than hearing “You didn’t really do anything bad,” particularly if the patient is religious. Hearing these words from a chaplain is often more effective than hearing them from a lay person, just as many of us take basic health information more seriously when we hear it from a physician. Even if the veteran is not overtly religious, there may be a unique exchange between that person and a religious authority when it concerns the violation of a millenniaold, widely known teaching from the Bible, such as “Thou shalt not kill.”
Kopacz et al also rightly suggest that unless religious prohibitions have been balanced with teachings on forgiveness and grace, the teachings can actually exacerbate feelings of guilt and elevate them to harmful proportions, especially in the potentially vulnerable psyche of a veteran who may have been traumatized. If there is no religious or spiritual guidance for balancing prohibitions with graces, the patient may be left to spiral in an unending loop of guilt with no way out.
We therefore propose the following categories for different types (or “shades”) of guilt that can be effectively addressed by chaplains in concert with other members of the healthcare team. For simplicity, we call these types real guilt, survivor guilt, mistaken guilt, and complex-compound guilt.
REAL GUILT
An important role professional chaplains can play is to allow patients (in this case, veterans) to express their remorse and regret for violations of their own moral codes. In many cases, they have in fact hurt or killed another person, and they need the chance to unburden their hearts and spirits, especially if they were taught that killing people is a sin. Veterans who have harmed or killed others, even if under orders, are often left with bona fide feelings of guilt that need to be aired and released in a safe and confidential environment. This is often most effective when done by someone who not only is trained in nonjudgmental and nondirective listening, but also is a religious authority who can assure the patient of his or her innate worthiness and of the ability to be forgiven.
As Kopacz et al note, guilt is linked to a specific action or behavior and usually entails regret or remorse. Many veterans belong to or have had exposure to faith groups with strong moral codes and prohibitions, and so may see the chaplain as having authority to act as confessor and granter of absolution.
SURVIVOR GUILT
Survivor guilt is commonly understood as the feeling of surviving a terrible event or situation while someone else did not. Those who suffer from survivor guilt judge themselves unworthy of survival and believe the deceased to have been more courageous, virtuous, or somehow a better person than they. They torture themselves with ideas of the deceased person’s virtues—imagined or real—and sometimes go on to believe that “It should have been me who was killed.”
The burden of feeling that the wrong person died can be overwhelming. If these people are not helped to see their own worth and helped to find outlets for their sense of having been spared (by God, by their own wits, or by sheer luck), they are likely to struggle more. This is related but not identical to what we call mistaken guilt. The two types are similar because they share a sense of randomness and helplessness, but they are different for reasons we will explain below.
Chaplains can be particularly effective partners in the care of veterans with survivor guilt, helping them to make meaning out of a life-changing event, rather than find meaning inherently in that event. Meaning, purpose, and “God’s plan for my life” are common themes in the pastoral conversation that can provide a compass for the disoriented survivor.
MISTAKEN GUILT
Mistaken guilt describes when a person who is involved in the death of another but is absolutely blameless—and could not possibly have prevented that death—literally “mis-takes” the guilt upon himself or herself in spite of the facts. Because of the helplessness induced by this feeling, mistaken guilt can be more difficult to treat than other forms. These patients continue to suffer despite assurances that the death occurred through absolutely no fault of their own.2
Hickling3 has written extensively about this phenomenon in innocent motor vehicle drivers who cause pedestrian deaths, and he considers this type of guilt one of the most difficult to recover from precisely because of the helplessness factor. He has explained that if patients can find a real reason by which they were culpable for what happened, they can change their ways. But if they were absolutely innocent (as in many incidents in training or combat), they often cannot make sense of what happened in a way that allows them to move on because there is nothing they could have done differently and therefore nothing they can change.4
These patients almost certainly need long-term intervention such as cognitive behavioral therapy in order to train their mind away from such destructive thoughts. However, they are also very likely to be helped by a chaplain if they find that the event triggers memories of other past infractions of which they may need to unburden themselves (ie, confess).
COMPOUND-COMPLEX GUILT
As the name implies, compound-complex guilt is a combination of the other types and may have additional layers.
Compound-complex guilt leaves sufferers literally feeling guilty for feeling guilty. Though this may border on a genuine clinical disorder, it is also to some degree normal (eg, due to cultural taboos and norms) for people to feel culpable for not being able to “move on” or “forgive themselves” as quickly as others may want them to. Buddhists call this tendency the “second arrow effect.” The first arrow is the feeling of guilt (or other painful feeling) that strikes the individual, but the second arrow is the one he or she drives in afterward by thinking it is wrong or weak to even have the feeling.
Patients who suffer from this type of guilt blame themselves for the conundrum they are in and feel even worse. This is not unlike the vortex of unresolved and complicated grief.
Those who suffer from compound-complex guilt may layer the primary guilt with additional guilt for feeling weak, for needing help, or for asking for help. Especially in the culture of the military, the fear of stigma when asking for help (especially with mental health) is still quite strong. Therefore, chaplains can serve as a less threatening entry point for the veteran needing multiple professionals involved in his or her care.
NONJUDGMENTAL LISTENING
Nonjudgmental listening is essential to get at the source or sources of guilt, regardless of the type, in order to allow the wounds to air out and begin healing. Many veterans suffering from guilt may need intensive pharmacologic and cognitive therapy to fully recover, and care from a chaplain is not a substitute for psychiatric evaluation and treatment, especially if there is a risk of suicide.
However, chaplains may be able to help with the “deep work” of spiritual healing that is part of veterans’ overall recovery. This is true not only because chaplains are especially trained to do this, but also because they are the team members most likely to have uniquely spiritual language to speak to the condition. The language of confession, absolution, repentance, redemption, atonement, and forgiveness is language of the spiritual realm.
In addition, chaplains’ freedom from hourly billing concerns and their often less formalized interactions with patients may help to build trust. Well-trained chaplains, who are often quite gifted at creating an atmosphere of reverence and safety (sanctuary) in the most unlikely situations, are well suited to help the interdisciplinary team treat this vulnerable patient population.
SUGGESTED READING
For more insights into the role of chaplains on the interdisciplinary healthcare team, we recommend the following book: Cadge W. Paging God: Religion in the Halls of Medicine. Chicago, IL: University of Chicago Press; 2012.
- Kopacz MS, Rasmussen KA, Searle RF, Wozniak BM, Titus CE. Veterans, guilt, and suicide risk: chaplains can help. Cleve Clin J Med 2016; 83:101–105.
- Life after death: Act one—guilty as not charged. Darin Strauss. This American Life. www.thisamericanlife.org. Episode 359. Aired July 18, 2008. www.thisamericanlife.org/radio-archives/episode/359/life-after-death?act=1#play. Accessed December 10, 2015.
- Hickling EJ, Blanchard EB. Overcoming the Trauma of Your Motor Vehicle Accident: A Cognitive-behavioral Treatment Program. New York, NY: Oxford University Press; 2006.
- Hickling EJ. Transforming Tragedy: Finding Growth Following Life’s Traumas. North Charleston, SC: CreateSpace; 2012.
- Kopacz MS, Rasmussen KA, Searle RF, Wozniak BM, Titus CE. Veterans, guilt, and suicide risk: chaplains can help. Cleve Clin J Med 2016; 83:101–105.
- Life after death: Act one—guilty as not charged. Darin Strauss. This American Life. www.thisamericanlife.org. Episode 359. Aired July 18, 2008. www.thisamericanlife.org/radio-archives/episode/359/life-after-death?act=1#play. Accessed December 10, 2015.
- Hickling EJ, Blanchard EB. Overcoming the Trauma of Your Motor Vehicle Accident: A Cognitive-behavioral Treatment Program. New York, NY: Oxford University Press; 2006.
- Hickling EJ. Transforming Tragedy: Finding Growth Following Life’s Traumas. North Charleston, SC: CreateSpace; 2012.
Can patients opt to turn off implantable cardioverter-defibrillators near the end of life?
Yes. Although implantable cardioverter-defibrillators (ICDs) prevent sudden cardiac death in patients with advanced heart failure, their benefit in terminally ill patients is small.1 Furthermore, the shocks they deliver at the end of life can cause distress. Therefore, it is reasonable to consider ICD deactivation if the patient or family wishes.
A DIFFICULT DECISION
End-of-life decisions place significant emotional burdens on patients, their families, and their healthcare providers and can have social and legal consequences.
Turning off an ICD is an especially difficult decision, considering that these devices protect against sudden cardiac death and fatal arrhythmias. Also, patients and their representatives may find it more difficult to withdraw from active care than to forgo further interventions (more on this below), and they may misunderstand discussions about ICD deactivation, perceiving them as the beginning of abandonment.
ICD DEACTIVATION IS OFTEN DONE HAPHAZARDLY OR NOT AT ALL
Many healthcare providers are not trained in or comfortable with discussing end-of-life issues, and many hospitals and hospice programs lack policies and protocols for managing implanted devices at the end of life. Consequently, ICD management at the end of life varies among providers and tends to be suboptimal.2
In a report of a survey in 414 hospice facilities, 97% of facilities reported that they admitted patients with ICDs, but only 10% had a policy on device deactivation.3
In a survey of 47 European medical centers, only 4% said they addressed ICD deactivation with their patients.4
A study of 125 patients with ICDs who had died found that 52% had do-not-resuscitate orders. Nevertheless, in 100 patients the ICD had remained active in the last 24 hours of their life, and 31 of these patients had received shocks during their last 24 hours.5
In a survey of next of kin of patients with ICDs who had died of any cause,6 in only 27 of 100 cases had the clinician discussed ICD deactivation, and about three-fourths of these discussions had occurred during the last few days of life. Twenty-seven patients had received ICD discharges in the last month of life, and 8% had received a discharge during the final minutes.
TRAINING AND PROTOCOLS ARE NEEDED
Healthcare professionals need education about device deactivation at the end of life so that they are comfortable communicating with patients and families about this critical issue. To this end, several cardiac and palliative care societies have jointly released an expert statement on managing ICDs and other implantable devices in end-of-life situations.7
Many providers harbor a misunderstanding of the difference between withholding a device and withdrawing (or turning off) a device that is already implanted.2 Some mistakenly believe they would be committing a crime by deactivating an implanted life-sustaining device. Legally and ethically, there is no difference between withholding a device and withdrawing a device. Legally, carrying out a request to withdraw life-sustaining treatment is neither physician-assisted suicide nor euthanasia.
DISCUSSION SHOULD BEGIN EARLY AND SHOULD BE ONGOING
The discussion of ICD deactivation should begin before the device is implanted and should continue as the patient’s health status changes. In a survey, 40% of patients said they felt that ICD deactivation should be discussed before the device is implanted, and only 5% felt that this discussion should be undertaken in the last days of life.8
At the least, it is important to identify patients with ICDs on admission to hospice and to have policies in place that ensure adequate patient education to make an informed decision about ICD deactivation at the end of life.
The topic should be discussed when goals of care change and when do-not-resuscitate status is addressed, and also when advanced directives are being acknowledged. If the patient or his or her legal representative wishes to keep the ICD turned on, that wish should be respected. The essence of a discussion is not to impose the providers’ choice on the patient, but to help the patient make the right decision for himself or herself. Of note, patients entering hospice do not have to have do-not-resuscitate status.
We believe that device management in end-of-life circumstances should be part of the discussion of the goals of care. Accordingly, healthcare providers need to be familiar with device management and to have a higher comfort level in addressing such sensitive topics with patients facing the end of life, as well as with their families.
It is also advisable to apply protocols within hospice services to address ICD management options for the patient and the legal representative. An early decision regarding end-of-life deactivation will help patients avoid distressing ICD discharges and the related emotional distress in their last moments.
- Barsheshet A, Moss AJ, Huang DT, McNitt S, Zareba W, Goldenberg I. Applicability of a risk score for prediction of the long-term (8-year) benefit of the implantable cardioverter-defibrillator. J Am Coll Cardiol 2012; 59:2075–2079.
- Kapa S, Mueller PS, Hayes DL, Asirvatham SJ. Perspectives on withdrawing pacemaker and implantable cardioverter-defibrillator therapies at end of life: results of a survey of medical and legal professionals and patients. Mayo Clin Proc 2010; 85:981–990.
- Goldstein N, Carlson M, Livote E, Kutner JS. Brief communication: management of implantable cardioverter-defibrillators in hospice: a nationwide survey. Ann Intern Med 2010; 152:296–299.
- Marinskis G, van Erven L; EHRA Scientific Initiatives Committtee. Deactivation of implanted cardioverter-defibrillators at the end of life: results of the EHRA survey. Europace 2010; 12:1176–1177.
- Kinch Westerdahl A, Sjoblom J, Mattiasson AC, Rosenqvist M, Frykman V. Implantable cardioverter-defibrillator therapy before death: high risk for painful shocks at end of life. Circulation 2014; 129:422–429.
- Goldstein NE, Lampert R, Bradley E, Lynn J, Krumholz HM. Management of implantable cardioverter defibrillators in end-of-life care. Ann Intern Med 2004; 141:835–838.
- Lampert R, Hayes DL, Annas GJ, et al; American College of Cardiology; American Geriatrics Society; American Academy of Hospice and Palliative Medicine; American Heart Association; European Heart Rhythm Association; Hospice and Palliative Nurses Association. HRS expert consensus statement on the management of cardiovascular implantable electronic devices (CIEDs) in patients nearing end of life or requesting withdrawal of therapy. Heart Rhythm 2010; 7:1008–1026.
- Raphael CE, Koa-Wing M, Stain N, Wright I, Francis DP, Kanagaratnam P. Implantable cardioverter-defibrillator recipient attitudes towards device activation: how much do patients want to know? Pacing Clin Electrophysiol 2011; 34:1628–1633.
Yes. Although implantable cardioverter-defibrillators (ICDs) prevent sudden cardiac death in patients with advanced heart failure, their benefit in terminally ill patients is small.1 Furthermore, the shocks they deliver at the end of life can cause distress. Therefore, it is reasonable to consider ICD deactivation if the patient or family wishes.
A DIFFICULT DECISION
End-of-life decisions place significant emotional burdens on patients, their families, and their healthcare providers and can have social and legal consequences.
Turning off an ICD is an especially difficult decision, considering that these devices protect against sudden cardiac death and fatal arrhythmias. Also, patients and their representatives may find it more difficult to withdraw from active care than to forgo further interventions (more on this below), and they may misunderstand discussions about ICD deactivation, perceiving them as the beginning of abandonment.
ICD DEACTIVATION IS OFTEN DONE HAPHAZARDLY OR NOT AT ALL
Many healthcare providers are not trained in or comfortable with discussing end-of-life issues, and many hospitals and hospice programs lack policies and protocols for managing implanted devices at the end of life. Consequently, ICD management at the end of life varies among providers and tends to be suboptimal.2
In a report of a survey in 414 hospice facilities, 97% of facilities reported that they admitted patients with ICDs, but only 10% had a policy on device deactivation.3
In a survey of 47 European medical centers, only 4% said they addressed ICD deactivation with their patients.4
A study of 125 patients with ICDs who had died found that 52% had do-not-resuscitate orders. Nevertheless, in 100 patients the ICD had remained active in the last 24 hours of their life, and 31 of these patients had received shocks during their last 24 hours.5
In a survey of next of kin of patients with ICDs who had died of any cause,6 in only 27 of 100 cases had the clinician discussed ICD deactivation, and about three-fourths of these discussions had occurred during the last few days of life. Twenty-seven patients had received ICD discharges in the last month of life, and 8% had received a discharge during the final minutes.
TRAINING AND PROTOCOLS ARE NEEDED
Healthcare professionals need education about device deactivation at the end of life so that they are comfortable communicating with patients and families about this critical issue. To this end, several cardiac and palliative care societies have jointly released an expert statement on managing ICDs and other implantable devices in end-of-life situations.7
Many providers harbor a misunderstanding of the difference between withholding a device and withdrawing (or turning off) a device that is already implanted.2 Some mistakenly believe they would be committing a crime by deactivating an implanted life-sustaining device. Legally and ethically, there is no difference between withholding a device and withdrawing a device. Legally, carrying out a request to withdraw life-sustaining treatment is neither physician-assisted suicide nor euthanasia.
DISCUSSION SHOULD BEGIN EARLY AND SHOULD BE ONGOING
The discussion of ICD deactivation should begin before the device is implanted and should continue as the patient’s health status changes. In a survey, 40% of patients said they felt that ICD deactivation should be discussed before the device is implanted, and only 5% felt that this discussion should be undertaken in the last days of life.8
At the least, it is important to identify patients with ICDs on admission to hospice and to have policies in place that ensure adequate patient education to make an informed decision about ICD deactivation at the end of life.
The topic should be discussed when goals of care change and when do-not-resuscitate status is addressed, and also when advanced directives are being acknowledged. If the patient or his or her legal representative wishes to keep the ICD turned on, that wish should be respected. The essence of a discussion is not to impose the providers’ choice on the patient, but to help the patient make the right decision for himself or herself. Of note, patients entering hospice do not have to have do-not-resuscitate status.
We believe that device management in end-of-life circumstances should be part of the discussion of the goals of care. Accordingly, healthcare providers need to be familiar with device management and to have a higher comfort level in addressing such sensitive topics with patients facing the end of life, as well as with their families.
It is also advisable to apply protocols within hospice services to address ICD management options for the patient and the legal representative. An early decision regarding end-of-life deactivation will help patients avoid distressing ICD discharges and the related emotional distress in their last moments.
Yes. Although implantable cardioverter-defibrillators (ICDs) prevent sudden cardiac death in patients with advanced heart failure, their benefit in terminally ill patients is small.1 Furthermore, the shocks they deliver at the end of life can cause distress. Therefore, it is reasonable to consider ICD deactivation if the patient or family wishes.
A DIFFICULT DECISION
End-of-life decisions place significant emotional burdens on patients, their families, and their healthcare providers and can have social and legal consequences.
Turning off an ICD is an especially difficult decision, considering that these devices protect against sudden cardiac death and fatal arrhythmias. Also, patients and their representatives may find it more difficult to withdraw from active care than to forgo further interventions (more on this below), and they may misunderstand discussions about ICD deactivation, perceiving them as the beginning of abandonment.
ICD DEACTIVATION IS OFTEN DONE HAPHAZARDLY OR NOT AT ALL
Many healthcare providers are not trained in or comfortable with discussing end-of-life issues, and many hospitals and hospice programs lack policies and protocols for managing implanted devices at the end of life. Consequently, ICD management at the end of life varies among providers and tends to be suboptimal.2
In a report of a survey in 414 hospice facilities, 97% of facilities reported that they admitted patients with ICDs, but only 10% had a policy on device deactivation.3
In a survey of 47 European medical centers, only 4% said they addressed ICD deactivation with their patients.4
A study of 125 patients with ICDs who had died found that 52% had do-not-resuscitate orders. Nevertheless, in 100 patients the ICD had remained active in the last 24 hours of their life, and 31 of these patients had received shocks during their last 24 hours.5
In a survey of next of kin of patients with ICDs who had died of any cause,6 in only 27 of 100 cases had the clinician discussed ICD deactivation, and about three-fourths of these discussions had occurred during the last few days of life. Twenty-seven patients had received ICD discharges in the last month of life, and 8% had received a discharge during the final minutes.
TRAINING AND PROTOCOLS ARE NEEDED
Healthcare professionals need education about device deactivation at the end of life so that they are comfortable communicating with patients and families about this critical issue. To this end, several cardiac and palliative care societies have jointly released an expert statement on managing ICDs and other implantable devices in end-of-life situations.7
Many providers harbor a misunderstanding of the difference between withholding a device and withdrawing (or turning off) a device that is already implanted.2 Some mistakenly believe they would be committing a crime by deactivating an implanted life-sustaining device. Legally and ethically, there is no difference between withholding a device and withdrawing a device. Legally, carrying out a request to withdraw life-sustaining treatment is neither physician-assisted suicide nor euthanasia.
DISCUSSION SHOULD BEGIN EARLY AND SHOULD BE ONGOING
The discussion of ICD deactivation should begin before the device is implanted and should continue as the patient’s health status changes. In a survey, 40% of patients said they felt that ICD deactivation should be discussed before the device is implanted, and only 5% felt that this discussion should be undertaken in the last days of life.8
At the least, it is important to identify patients with ICDs on admission to hospice and to have policies in place that ensure adequate patient education to make an informed decision about ICD deactivation at the end of life.
The topic should be discussed when goals of care change and when do-not-resuscitate status is addressed, and also when advanced directives are being acknowledged. If the patient or his or her legal representative wishes to keep the ICD turned on, that wish should be respected. The essence of a discussion is not to impose the providers’ choice on the patient, but to help the patient make the right decision for himself or herself. Of note, patients entering hospice do not have to have do-not-resuscitate status.
We believe that device management in end-of-life circumstances should be part of the discussion of the goals of care. Accordingly, healthcare providers need to be familiar with device management and to have a higher comfort level in addressing such sensitive topics with patients facing the end of life, as well as with their families.
It is also advisable to apply protocols within hospice services to address ICD management options for the patient and the legal representative. An early decision regarding end-of-life deactivation will help patients avoid distressing ICD discharges and the related emotional distress in their last moments.
- Barsheshet A, Moss AJ, Huang DT, McNitt S, Zareba W, Goldenberg I. Applicability of a risk score for prediction of the long-term (8-year) benefit of the implantable cardioverter-defibrillator. J Am Coll Cardiol 2012; 59:2075–2079.
- Kapa S, Mueller PS, Hayes DL, Asirvatham SJ. Perspectives on withdrawing pacemaker and implantable cardioverter-defibrillator therapies at end of life: results of a survey of medical and legal professionals and patients. Mayo Clin Proc 2010; 85:981–990.
- Goldstein N, Carlson M, Livote E, Kutner JS. Brief communication: management of implantable cardioverter-defibrillators in hospice: a nationwide survey. Ann Intern Med 2010; 152:296–299.
- Marinskis G, van Erven L; EHRA Scientific Initiatives Committtee. Deactivation of implanted cardioverter-defibrillators at the end of life: results of the EHRA survey. Europace 2010; 12:1176–1177.
- Kinch Westerdahl A, Sjoblom J, Mattiasson AC, Rosenqvist M, Frykman V. Implantable cardioverter-defibrillator therapy before death: high risk for painful shocks at end of life. Circulation 2014; 129:422–429.
- Goldstein NE, Lampert R, Bradley E, Lynn J, Krumholz HM. Management of implantable cardioverter defibrillators in end-of-life care. Ann Intern Med 2004; 141:835–838.
- Lampert R, Hayes DL, Annas GJ, et al; American College of Cardiology; American Geriatrics Society; American Academy of Hospice and Palliative Medicine; American Heart Association; European Heart Rhythm Association; Hospice and Palliative Nurses Association. HRS expert consensus statement on the management of cardiovascular implantable electronic devices (CIEDs) in patients nearing end of life or requesting withdrawal of therapy. Heart Rhythm 2010; 7:1008–1026.
- Raphael CE, Koa-Wing M, Stain N, Wright I, Francis DP, Kanagaratnam P. Implantable cardioverter-defibrillator recipient attitudes towards device activation: how much do patients want to know? Pacing Clin Electrophysiol 2011; 34:1628–1633.
- Barsheshet A, Moss AJ, Huang DT, McNitt S, Zareba W, Goldenberg I. Applicability of a risk score for prediction of the long-term (8-year) benefit of the implantable cardioverter-defibrillator. J Am Coll Cardiol 2012; 59:2075–2079.
- Kapa S, Mueller PS, Hayes DL, Asirvatham SJ. Perspectives on withdrawing pacemaker and implantable cardioverter-defibrillator therapies at end of life: results of a survey of medical and legal professionals and patients. Mayo Clin Proc 2010; 85:981–990.
- Goldstein N, Carlson M, Livote E, Kutner JS. Brief communication: management of implantable cardioverter-defibrillators in hospice: a nationwide survey. Ann Intern Med 2010; 152:296–299.
- Marinskis G, van Erven L; EHRA Scientific Initiatives Committtee. Deactivation of implanted cardioverter-defibrillators at the end of life: results of the EHRA survey. Europace 2010; 12:1176–1177.
- Kinch Westerdahl A, Sjoblom J, Mattiasson AC, Rosenqvist M, Frykman V. Implantable cardioverter-defibrillator therapy before death: high risk for painful shocks at end of life. Circulation 2014; 129:422–429.
- Goldstein NE, Lampert R, Bradley E, Lynn J, Krumholz HM. Management of implantable cardioverter defibrillators in end-of-life care. Ann Intern Med 2004; 141:835–838.
- Lampert R, Hayes DL, Annas GJ, et al; American College of Cardiology; American Geriatrics Society; American Academy of Hospice and Palliative Medicine; American Heart Association; European Heart Rhythm Association; Hospice and Palliative Nurses Association. HRS expert consensus statement on the management of cardiovascular implantable electronic devices (CIEDs) in patients nearing end of life or requesting withdrawal of therapy. Heart Rhythm 2010; 7:1008–1026.
- Raphael CE, Koa-Wing M, Stain N, Wright I, Francis DP, Kanagaratnam P. Implantable cardioverter-defibrillator recipient attitudes towards device activation: how much do patients want to know? Pacing Clin Electrophysiol 2011; 34:1628–1633.
Common neurologic emergencies for nonneurologists: When minutes count
Neurologic emergencies such as acute stroke, status epilepticus, subarachnoid hemorrhage, neuromuscular weakness, and spinal cord injury affect millions of Americans yearly.1,2 These conditions can be difficult to diagnose, and delays in recognition and treatment can have devastating results. Consequently, it is important for nonneurologists to be able to quickly recognize these conditions and initiate timely management, often while awaiting neurologic consultation.
Here, we review how to recognize and treat these common, serious conditions.
ACUTE ISCHEMIC STROKE: TIME IS OF THE ESSENCE
Stroke is the fourth leading cause of death in the United States and is one of the most common causes of disability worldwide.3–5 About 85% of strokes are ischemic, resulting from diminished vascular supply to the brain. Symptoms such as facial droop, unilateral weakness or numbness, aphasia, gaze deviation, and unsteadiness of gait may be seen. Time is of the essence, as all currently available interventions are safe and effective only within defined time windows.
Diagnosis and assessment
When acute ischemic stroke is suspected, the clinical history, time of onset, and basic neurologic examination should be obtained quickly.
The National Institutes of Health (NIH) stroke scale is an objective marker for assessing stroke severity as well as evolution of disease and should be obtained in all stroke patients. Scores range from 0 (best) to 42 (worst) (www.ninds.nih.gov/doctors/NIH_Stroke_Scale.pdf).
Time of onset of symptoms is essential to determine, since it guides eligibility for acute therapies. Clinicians should ascertain the last time the patient was seen to be neurologically well in order to estimate this time window as closely as possible.
Laboratory tests should include a fingerstick blood glucose measurement, coagulation studies, complete blood cell count, and basic metabolic profile.
Computed tomography (CT) of the head without contrast should be obtained immediately to exclude acute hemorrhage and any alternative diagnoses that could explain the patient’s symptoms. Acute brain ischemia is often not apparent on CT during the first few hours of injury. Therefore, a patient presenting with new focal neurologic deficits and an unremarkable result on CT of the head should be treated as having had an acute ischemic stroke, and interventional therapies should be considered.
Stroke mimics should be considered and treated, as appropriate (Table 1).
Acute management of ischemic stroke
Acute treatment should not be delayed by obtaining chest radiography, inserting a Foley catheter, or obtaining an electrocardiogram. The longer the time that elapses before treatment, the worse the functional outcome, underscoring the need for rapid decision-making.6–8
Lowering the head of the bed may provide benefit by promoting blood flow to ischemic brain tissue.9 However, this should not be done in patients with significantly elevated intracerebral pressure and concern for herniation.
Permissive hypertension (antihypertensive treatment only for blood pressure greater than 220/110 mm Hg) should be allowed per national guidelines to provide adequate perfusion to brain areas at risk of injury.10
Tissue plasminogen activator. Patients with ischemic stroke who present within 3 hours of symptom onset should be considered for intravenous administration of tissue plasminogen activator (tPA), a safe and effective therapy with nearly 2 decades of evidence to support its use.10 The treating physician should carefully review the risks and benefits of this therapy.
To receive tPA, the patient must have all of the following:
- Clinical diagnosis of ischemic stroke with measurable neurologic deficit
- Onset of symptoms within the past 3 hours
- Age 18 or older.
The patient must not have any of the following:
- Significant stroke within the past 3 months
- Severe traumatic head injury within the past 3 months
- History of significant intracerebral hemorrhage
- Previously ruptured arteriovenous malformation or intracranial aneurysm
- Central nervous system neoplasm
- Arterial puncture at a noncompressible site within the past 7 days
- Evidence of hemorrhage on CT of the head
- Evidence of ischemia in greater than 33% of the cerebral hemisphere on head CT
- History and symptoms strongly suggesting subarachnoid hemorrhage
- Persistent hypertension (systolic pressure ≥ 185 mm Hg or diastolic pressure ≥ 110 mm Hg)
- Evidence of acute significant bleeding (external or internal)
- Hypoglycemia—ie, serum glucose less than 50 mg/dL (< 2.8 mmol/L)
- Thrombocytopenia (platelet count < 100 × 109/L)
- Significant coagulopathy (international normalized ratio > 1.7, prothrombin time > 15 seconds, or abnormally elevated activated partial thromboplastin time)
- Current use of a factor Xa inhibitor or direct thrombin inhibitor.
Relative contraindications:
- Minor or rapidly resolving symptoms
- Major surgery or trauma within the past 14 days
- Gastrointestinal or urinary tract bleeding within the past 21 days
- Myocardial infarction in the past 3 months
- Unruptured intracranial aneurysm
- Seizure occurring at stroke onset
- Pregnancy.
If these criteria are satisfied, tPA should be given at a dose of 0.9 mg/kg intravenously over 60 minutes. Ten percent of the dose should be given as an initial bolus, followed by a constant infusion of the remaining 90% over 1 hour.
If tPA is given, the blood pressure must be kept lower than 185/110 mm Hg to minimize the risk of symptomatic intracerebral hemorrhage.
A subset of patients may benefit from receiving intravenous tPA between 3 and 4.5 hours after the onset of stroke symptoms. These include patients who are no more than 80 years old, who have not recently used oral anticoagulants, who do not have severe neurologic injury (ie, do not have NIH Stroke Scale scores > 25), and who do not have diabetes mellitus or a history of ischemic stroke.11 Although many hospitals have such a protocol for tPA up to 4.5 hours after the onset of stroke symptoms, this time window is not currently approved by the US Food and Drug Administration.
Intra-arterial therapy. Based on recent trials, some patients may benefit further from intra-arterial thrombolysis or mechanical thrombectomy, both delivered during catheter-based cerebral angiography, independent of intravenous tPA administration.12,13 These patients should be evaluated on a case-by-case basis by a neurologist and neurointerventional team. Time windows for these treatments generally extend to 6 hours from stroke onset and perhaps even longer in some situations (eg, basilar artery occlusion).
An antiplatelet agent should be started quickly in all stroke patients who do not receive tPA. Patients who receive tPA can begin receiving an antiplatelet agent 24 hours afterward.
Unfractionated heparin. There is no evidence to support the use of unfractionated heparin in most cases of acute ischemic stroke.10
Glucose control (in the range of 140–180 mg/dL) and fever control remain essential elements of post-acute stroke care to provide additional protection to the damaged brain.
For ischemic stroke due to atrial fibrillation
In ischemic stroke due to atrial fibrillation, early anticoagulation should be considered, based on the CHA2DS2-VASC risk of ischemic stroke vs the HAS-BLED risk of hemorrhage (calculators available at www.mdcalc.com).
In general, anticoagulation may be withheld during the first 72 hours while further stroke workup and evaluation of extent of injury are carried out, as there is an increased risk of hemorrhagic transformation of the ischemic stroke. Often, anticoagulation is resumed at a full dose between 72 hours and 2 weeks of the ischemic stroke.
ACUTE HEMORRHAGIC STROKE: BLOOD PRESSURE, COAGULATION
Approximately 15% of strokes are caused by intracerebral hemorrhage, which can be detected with noncontrast head CT with a sensitivity of 98.6% within 6 hours of the onset of bleeding.14 A common underlying cause of intracerebral hemorrhage is chronic poorly controlled hypertension, causing rupture of damaged (or “lipohyalinized”) vessels with resultant blood extravasation into the brain parenchyma. Other causes are less common (Table 2).
Treatment of acute hemorrhagic stroke
Acute treatment of intracerebral hemorrhage includes blood pressure control, reversal of underlying coagulopathy or anticoagulation, and sometimes intracranial pressure control. There is little role for surgery in most cases, based on findings of randomized trials.15
Blood pressure control. Many studies have investigated optimal blood pressure goals in acute intracerebral hemorrhage. Recent data suggest that early aggressive therapy, targeting a systolic blood pressure goal less than 140 mm Hg within the first hour, is safe and can lead to better functional outcomes than a more conservative blood-pressure-lowering target.16 Rapid-onset, short-acting antihypertensive agents in intravenous form, such as nicardipine and labetalol, are frequently used. Of note, this treatment strategy for hemorrhagic stroke is in direct contrast to the treatment of ischemic stroke, in which permissive hypertension (blood pressure goal < 220/110 mm Hg) is often pursued.
Reversal of any coagulation abnormalities should be done quickly in intracranial hemorrhage. Warfarin use has been shown to be a strong independent predictor of intracranial hemorrhage expansion, which increases the risk of death.17,18
Increasingly, agents other than vitamin K or fresh-frozen plasma are being used to rapidly reverse anticoagulation, including prothrombin complex concentrate (available in three- and four-factor preparations) and recombinant factor VIIa. While four-factor prothrombin complex concentrate and recombinant factor VIIa have been shown to be more efficacious than fresh-frozen plasma, there are limited data directly comparing these newer reversal agents against each other.19 The use of these medications is limited by availability and practitioner familiarity.20–22
Reversing anticoagulation due to target-specific oral anticoagulants. The acute management of intracranial hemorrhage in patients taking the new target-specific oral anticoagulants (eg, dabigatran, apixaban, rivaroxaban, edoxaban) remains challenging. Laboratory tests such as factor Xa levels are not readily available in many institutions and do not provide results in a timely fashion, and in the interim, acute hemorrhage and clinical deterioration may occur. Management strategies involve giving fresh-frozen plasma, prothrombin complex concentrate, and consideration of hemodialysis.23 Dabigatran reversal with idarucizumab has recently been shown to have efficacy.24
Vigilance for elevated intracranial pressure. Intracranial hemorrhage can occasionally cause elevated intracranial pressure, which should be treated rapidly. Any acute decline in mental status in a patient with intracranial hemorrhage requires emergency imaging to evaluate for expansion of hemorrhage.
SUBARACHNOID HEMORRHAGE
The sudden onset of a “thunderclap” headache (often described by patients as “the worst headache of my life”) suggests subarachnoid hemorrhage.
In contrast to intracranial hemorrhage, in subarachnoid hemorrhage blood collects mainly in the cerebral spinal fluid-containing spaces surrounding the brain, leading to a higher incidence of hydrocephalus from impaired drainage of cerebrospinal fluid. Nontraumatic subarachnoid hemorrhage is most often caused by rupture of an intracranial aneurysm, which can be a devastating event, with death rates approaching 50%.25
Diagnosis of subarachnoid hemorrhage
Noncontrast CT of the head is the main modality for diagnosing subarachnoid hemorrhage. Blood within the subarachnoid space is demonstrable in 92% of cases if CT is performed within the first 24 hours of hemorrhage, with an initial sensitivity of about 95% within the first 6 hours of onset.14,26,27 The longer CT is delayed, the lower the sensitivity.
Some studies suggest that a protocol of CT followed by CT angiography can safely exclude aneurysmal subarachnoid hemorrhage and obviate the need for lumbar puncture. However, further research is required to validate this approach.28
Lumbar puncture. If clinical suspicion of subarachnoid hemorrhage remains strong even though initial CT is negative, lumbar puncture must be performed for cerebrospinal fluid analysis.29 Xanthochromia (a yellowish pigmentation of the cerebrospinal fluid due to the degeneration of blood products that occurs within 8 to 12 hours of bleeding) should raise the alarm for subarachnoid hemorrhage; this sign may be present up to 4 weeks after the bleeding event.30
If lumbar puncture is contraindicated, then aneurysmal subarachnoid hemorrhage has not been ruled out, and further neurologic consultation should be pursued.
Management of subarachnoid hemorrhage
Early management of blood pressure for a ruptured intracranial aneurysm follows strategies similar to those for intracranial hemorrhage. Further investigation is rapidly directed toward an underlying vascular malformation, with intracranial vessel imaging such as CT angiography, magnetic resonance angiography, or the gold standard test—catheter-based cerebral angiography.
Aneurysms are treated (or “secured”) either by surgical clipping or by endovascular coiling. Endovascular coiling is preferable in cases in which both can be safely attempted.31 If the facility lacks the resources to do these procedures, the patient should be referred to a nearby tertiary care center.
INTRACRANIAL HYPERTENSION: DANGER OF BRAIN HERNIATION
A number of conditions can cause an acute intracranial pressure elevation. The danger of brain herniation requires that therapies be implemented rapidly to prevent catastrophic neurologic injury. In many situations, nonneurologists are the first responders and therefore should be familiar with basic intracranial pressure management.
Initial symptoms of acute rise in intracranial pressure
As intracranial pressure rises, pressure is typically equally distributed throughout the cranial vault, leading to dysfunction of the ascending reticular activating system, which clinically manifests as the inability to stay alert despite varying degrees of noxious stimulation. Progressive cranial neuropathies (often starting with pupillary abnormalities) and coma are often seen in this setting as the upper brainstem begins to be compressed.
Initial assessment and treatment of elevated intracranial pressure
Noncontrast CT of the head is often obtained immediately when acutely elevated intracranial pressure is suspected. If clinical examination and radiographic findings are consistent with intracranial hypertension, prompt measures can be started at the bedside.
Elevate the head of the bed to 30 degrees to promote venous drainage and reduce intracranial pressure. (In contrast, most other hemodynamically unstable patients are placed flat or in the Trendelenburg position.)
Intubation should be done quickly in cases of airway compromise, and hyperventilation should be started with a goal Paco2 of 30 to 35 mm Hg. This hypocarbic strategy promotes cerebral vasoconstriction and a transient decrease in intracranial pressure.
Hyperosmolar therapy allows for transient intracranial volume decompression and is the mainstay of emergency medical treatment of intracranial hypertension. Mannitol is a hyperosmolar polysaccharide that promotes osmotic diuresis and removes excessive cerebral water. In the acute setting, it can be given as an intravenous bolus of 1 to 2 g/kg through a peripheral intravenous line, followed by a bolus every 4 to 6 hours. Hypotension can occur after diuresis, and renal function should be closely monitored since frequent mannitol use can promote acute tubular necrosis. In patients who are anuric, the medication is typically not used.
Hypertonic saline (typically 3% sodium chloride, though different concentrations are available) is an alternative that helps draw interstitial fluid into the intravascular space, decreasing cerebral edema and maintaining hemodynamic stability. Relative contraindications include congestive heart failure or renal failure leading to pulmonary edema from volume overload. Hypertonic saline can be given as a bolus or a constant infusion. Some institutions have rapid access to 23.4% saline, which can be given as a 30-mL bolus but typically requires a central venous catheter for rapid infusion.
Comatose patients with radiographic findings of hydrocephalus, epidural or subdural hematoma, or mass effect with midline shift warrant prompt neurosurgical consultation for further surgical measures of intracranial pressure control and monitoring.
The ‘blown’ pupil
The physician should be concerned about elevated intracranial pressure if a patient has mydriasis, ie, an abnormally dilated (“blown”) pupil, which is a worrisome sign in the setting of true intracranial hypertension. However, many different processes can cause mydriasis and should be kept in mind when evaluating this finding (Table 3).32 If radiographic findings do not suggest elevated intracranial pressure, further workup into these other processes should be pursued.
STATUS EPILEPTICUS: SEIZURE CONTROL IS IMPORTANT
A continuous unremitting seizure lasting longer than 5 minutes or recurrent seizure activity in a patient who does not regain consciousness between seizures should be treated as status epilepticus. All seizure types carry the risk of progressing to status epilepticus, and responsiveness to antiepileptic drug therapy is inversely related to the duration of seizures. It is imperative that seizure activity be treated early and aggressively to prevent recalcitrant seizure activity, neuronal damage, and progression to status epilepticus.33
Once the ABCs of emergency stabilization have been performed (ie, airway, breathing, circulation), antiepileptic drug therapy should start immediately using established algorithms (Figure 1).34–36 During the course of treatment, the reliability of the neurologic examination may be limited due to medication effects or continued status epilepticus, making continuous video electroencephalographic monitoring often necessary to guide further therapy in patients who are not rapidly recovering.34–38
Once status epilepticus has resolved, further investigation into the underlying cause should be pursued quickly, especially in patients without a previous diagnosis of epilepsy. Head CT with contrast or magnetic resonance imaging can be used to look for any structural abnormality that may explain seizures. Basic laboratory tests including toxicology screening can identify a common trigger such as hypoglycemia or stimulant use. Fever or other possible signs of meningitis should be investigated further with cerebrospinal fluid analysis.
SPINAL CORD INJURY
Acute spinal cord injury can lead to substantial long-term neurologic impairment and should be suspected in any patient presenting with focal motor loss, sensory loss, or both with sparing of the cranial nerves and mental status. Causes of injury include compression (traumatic or nontraumatic) and inflammatory and noninflammatory myelopathies.
The location of the injury can be inferred by analyzing the symptoms, which can point to the cord level and indicate whether the anterior or posterior of the cord is involved. Anterior cord injury tends to affect the descending corticospinal and pyramidal tracts, resulting in motor deficits and weakness. Posterior cord injury involves the dorsal columns, leading to deficits of vibration sensation and proprioception. High cervical cord injuries tend to involve varying degrees of quadriparesis, sensory loss, and sometimes respiratory compromise. A clinical history of bilateral lower-extremity weakness, a “band-like” sensory complaint around the lower chest or abdomen, or both, can suggest thoracic cord involvement. Symptoms isolated to one or both lower extremities along with lower back pain and bowel or bladder involvement may point to injury of the lumbosacral cord.
Basic management of spinal cord injury includes decompression of the bladder and initial protection against further injury with a stabilizing collar or brace.
Magnetic resonance imaging with and without contrast is the ideal study to evaluate injuries to the spinal cord itself. While CT is helpful in identifying bony disease of the spinal column (eg, evaluating traumatic fractures), it is not helpful in viewing intrinsic cord pathology.
Traumatic myelopathy
Traumatic spinal cord injury is usually suggested by the clinical history and confirmed with CT. In this setting, early consultation with a neurosurgeon is required to prevent permanent cord injury.
Guidelines suggest maintaining a mean arterial pressure greater than 85 to 90 mm Hg for the first 7 days after traumatic spinal cord injury, a particular problem in the setting of hemodynamic instability, which can accompany lesions above the midthoracic level.39,40
Patients with vertebral body misalignment should be placed in an appropriate stabilizing collar or brace until a medically trained professional deems it appropriate to discontinue the device, or until surgical stabilization is performed.
Methylprednisone is a controversial intervention for acute spinal cord trauma, lacking clear benefit in meta-analyses.41
Nontraumatic compressive myelopathy
Patients with nontraumatic compressive myelopathy tend to present with varying degrees of back pain and worsening sensorimotor function. The differential diagnosis includes epidural abscesses, hematoma, metastatic neoplasm, and osteophyte compression (Table 4). The clinical history helps to guide therapy and should involve assessment for previous spinal column injury, immunocompromised state, travel history (which provides information on risks of exposure to a variety of diseases, including infections), and constitutional symptoms such as fever and weight loss.
Epidural abscess can have devastating results if missed. Red flags such as recent illness, intravenous drug use, focal back pain, fever, worsening numbness or weakness, and bowel or bladder incontinence should raise suspicion of this disorder. Emergency magnetic resonance imaging is required to diagnose this condition, and treatment involves urgent administration of antibiotics and consideration of surgical drainage.
Noncompressive myelopathies
There are numerous causes of noncompressive spinal cord injury (Table 4), and the etiology may be inflammatory (eg, “myelitis”) or noninflammatory. The diagnostic workup may require both magnetic resonance imaging and cerebrospinal fluid analysis. Acute disease-targeted therapy is rarely indicated and can be deferred until a full diagnostic workup has been completed.
NEUROMUSCULAR DISEASE: IS VENTILATION NEEDED?
Diseases involving the motor components of the peripheral nervous system (Table 5) share the common risk of causing ventilatory failure due to weakness of the diaphragm, intercostal muscles, and upper-airway muscles. Clinicians need to be aware of this risk and view these disorders as neurologic emergencies.
Determining when these patients require mechanical intubation is a challenge. Serial measurements of maximum inspiratory force and vital capacity are important and can be accomplished quickly at the bedside by a respiratory therapist. A maximum inspiratory force less than –30 cm H2O or a vital capacity less than 20 mL/kg, or both, are worrisome markers that raise concern for impending ventilatory failure. Serial measurements can detect changes in these values that might indicate the need for elective intubation. In any patient presenting with weakness of the limbs, these measurements are an important step in the initial evaluation.
Myasthenic crisis
Myasthenia gravis is caused by autoantibodies directed against postsynaptic acetylcholine receptors. Patients demonstrate muscle weakness, usually in a proximal pattern, with fatigue, respiratory distress, nasal speech, ophthalmoparesis, and dysphagia. Exacerbations can occur as a response to recent infection, surgery, or medications such as neuromuscular blocking agents or aminoglycosides.
Myasthenic crisis, while uncommon, is a life-threatening emergency characterized by bulbar or respiratory failure secondary to muscle weakness. It can occur in patients already diagnosed with myasthenia gravis or may be the initial manifestation of the disease.42–49 Intubation and mechanical ventilation are frequently required. Postoperative myasthenic patients in whom extubation has been delayed more than 24 hours should be considered in crisis.45
The diagnosis of myasthenia gravis can be made by serum autoantibody testing, electromyography, and nerve conduction studies (with repetitive stimulation) or administration of edrophonium in patients with obvious ptosis.
The mainstay of therapy for myasthenic crisis is either intravenous immunoglobulin at a dose of 2 g/kg over 2 to 5 days or plasmapheresis (5–7 exchanges over 7–14 days). Corticosteroids are not recommended in myasthenic crisis in patients who are not intubated, as they can potentiate an initial worsening of crisis. Once the patient begins to show clinical improvement, outpatient pyridostigmine and immunosuppressive medications can be resumed at a low dose and titrated as tolerated.
Acute inflammatory demyelinating polyneuropathy (Guillain-Barré syndrome)
Acute inflammatory demyelinating polyneuropathy is an autoimmune disorder involving autoantibodies against axons or myelin in the peripheral nervous system.
This disease should be suspected in a patient who is developing worsening muscle weakness (usually with areflexia) over the course of days to weeks. Occasionally, a recent diarrheal or other systemic infectious trigger can be identified. Blood pressure instability and cardiac arrhythmia can also be seen due to autonomic nerve involvement. Although classically described as an “ascending paralysis,” other variants of this disease have distinct clinical presentations (eg, the descending paralysis, ataxia, areflexia, ophthalmoparesis of the Miller Fisher syndrome).
Acute inflammatory demyelinating polyneuropathy is diagnosed by electromyography and nerve conduction studies. A cerebrospinal fluid profile demonstrating elevated protein and few white blood cells is typical.
Treatment, as in myasthenic crisis, involves intravenous immunoglobulin or plasmapheresis. Corticosteroids are ineffective. Anticipation of ventilatory failure and expectant intubation is essential, given the progressive nature of the disorder.50
- Pitts SR, Niska RW, Xu J, Burt CW. National hospital ambulatory medical care survey: 2006 emergency department summary. Natl Health Stat Report 2008; 7:1–38.
- McMullan JT, Knight WA, Clark JF, Beyette FR, Pancioli A. Time-critical neurological emergencies: the unfulfilled role for point-of-care testing. Int J Emerg Med 2010; 3:127–131.
- Centers for Disease Control and Prevention (CDC). Prevalence of stroke: United States, 2006–2010. MMWR Morb Mortal Wkly Rep 2012; 61:379–382.
- Lozano R, Naghavi M, Foreman K, et al. Global and regional mortality from 235 causes of death for 20 age groups in 1990 and 2010: a systematic analysis for the global burden of disease study 2010. Lancet 2012; 380:2095–2128.
- Vos T, Flaxman AD, Naghavi M, et al. Years lived with disability (YLDs) for 1,160 sequelae of 289 diseases and injuries 1990–2010: a systematic analysis for the global burden of disease study 2010. Lancet 2012; 380:2163–2196.
- Tissue plasminogen activator for acute ischemic stroke. The National Institute of Neurological Disorders and Stroke rt-PA stroke study group. N Engl J Med 1995; 333:1581–1587.
- Hacke W, Donnan G, Fieschi C, et al; ATLANTIS Trials Investigators; ECASS Trials Investigators; NINDS rt-PA Study Group Investigators. Association of outcome with early stroke treatment: pooled analysis of ATLANTIS, ECASS, and NINDS rt-PA stroke trials. Lancet 2004; 363:768–774.
- Saver JL, Fonarrow GC, Smith EE, et al. Time to treatment with intravenous tissue plasminogen activator and outcome from acute ischemic stroke. JAMA 2013; 309:2480–2488.
- Wojner-Alexander AW, Garami Z, Chernyshev OY, Alexandrov AV. Heads down: flat positioning improves blood flow velocity in acute ischemic stroke. Neurology 2005; 64:1354–1357.
- Jauch EC, Saver JL, Adams HP Jr, et al; American Heart Association Stroke Council; Council on Cardiovascular Nursing; Council on Peripheral Vascular Disease; Council on Clinical Cardiology. Guidelines for the early management of patients with acute ischemic stroke: a guideline for healthcare professionals from the American Heart Association/American Stroke Association. Stroke 2013; 44:870–947.
- Hacke W, Kaste M, Bluhmki E, et al; ECASS Investigators. Thrombolysis with alteplase 3 to 4.5 hours after acute ischemic stroke. N Engl J Med 2008; 359:1317–1329.
- Berkhemer OA, Fransen PSS, Beumer D, et al; MR CLEAN Investigators. A randomized trial of intraarterial treatment for acute ischemic stroke. N Eng J Med 2015; 372:11–20.
- Campbell BC, Mitchell PJ, Kleinig TJ, et al; EXTEND-IA Investigators. Endovascular therapy for ischemic stroke with perfusion-imaging selection. N Engl J Med 2015; 372:1009–1018.
- Backes D, Rinkel GJ, Kemperman H, Linn FH, Vergouwen MD. Time-dependent test characteristics of head computed tomography in patients suspected of nontraumatic subarachnoid hemorrhage. Stroke 2012; 43:2115–2119.
- Mendelow AD, Gregson BA, Fernandes HM, et al; STICH investigators. Early surgery versus initial conservative treatment in patients with spontaneous supratentorial intracerebral haematomas in the International Surgical Trial in Intracerebral Haemorrhage (STICH): a randomised trial. Lancet 2005; 365: 387–397.
- Anderson CS, Helley E, Huang Y, et al; INTERACT2 Investigators. Rapid blood-pressure lowering in patients with acute intracerebral hemorrhage. N Engl J Med 2013; 368:2355–2365.
- Flibotte JJ, Hagan N, O'Donnell J, Greenberg SM, Rosand J. Warfarin, hematoma expansion, and outcome of intracerebral hemorrhage. Neurology 2004; 63:1059–1064.
- Davis SM, Broderick J, Hennerici M, et al; Recombinant Activated Factor VII Intracerebral Hemorrhage Trial Investigators. Hematoma growth is a determinant of mortality and poor outcome after intracerebral hemorrhage. Neurology 2006; 66:1175–1181.
- Woo CH, Patel N, Conell C, et al. Rapid warfarin reversal in the setting of intracranial hemorrhage: a comparison of plasma, recombinant activated factor VII, and prothrombin complex concentrate. World Neurosurg 2014; 81:110–115.
- Broderick J, Connolly S, Feldmann E, et al; American Heart Association; American Stroke Association Stroke Council; High Blood Pressure Research Council; Quality of Care and Outcomes in Research Interdisciplinary Working Group. Guidelines for the management of spontaneous intracerebral hemorrhage in adults: 2007 update: a guideline from the American Heart Association/American Stroke Association Stroke Council, High Blood Pressure Research Council, and the Quality of Care and Outcomes in Research Interdisciplinary Working Group. Stroke 2007; 38:2001–2023.
- Goldstein JN, Thomas SH, Frontiero V, et al. Timing of fresh frozen plasma administration and rapid correction of coagulopathy in warfarin-related intracerebral hemorrhage. Stroke 2006, 37:151–155.
- Chapman SA, Irwin ED, Beal AL, Kulinski NM, Hutson KE, Thorson MA. Prothrombin complex concentrate versus standard therapies for INR reversal in trauma patients receiving warfarin. Ann Pharmacother 2011; 45:869–875.
- Fawole A, Daw HA, Crowther MA. Practical management of bleeding due to the anticoagulants dabigatran, rivaroxaban, and apixaban. Cleve Clin J Med 2013; 80:443–451.
- Pollack CV Jr, Reilly PA, Eikelboom J, et al. Idarucizumab for dabigatran reversal. N Engl J Med 2015; 373:511-520.
- Broderick JP, Brott TG, Duldner JE, Tomsick T, Leach A. Initial and recurrent bleeding are the major causes of death following subarachnoid hemorrhage. Stroke 1994; 25:1342–1347.
- Kassell NF, Torner JC, Haley EC Jr, Jane JA, Adams HP, Kongable GL. The international cooperative study on the timing of aneurysm surgery. Part 1: overall management results. J Neurosurg 1990; 73:18–36.
- Perry JJ, Stiell IG, Sivilotti ML, et al. Sensitivity of computed tomography performed within six hours of onset of headache for diagnosis of subarachnoid haemorrhage: prospective cohort study. BMJ 2011; 343:d4277.
- McCormack RF, Hutson A. Can computed tomography angiography of the brain replace lumbar puncture in the evaluation of acute-onset headache after a negative noncontrast cranial computed tomography scan? Acad Emerg Med 2010; 17:444–451.
- Connolly ES Jr, Rabinstein AA, Carhuapoma JR, et al; American Heart Association Stroke Council; Council on Cardiovascular Radiology and Intervention; Council on Cardiovascular Nursing; Council on Cardiovascular Surgery and Anesthesia; Council on Clinical Cardiology. Guidelines for the management of aneurysmal subarachnoid hemorrhage: a guideline for healthcare professionals from the American Heart Association/American Stroke Association. Stroke 2012; 43:1711–1737.
- Vermuelen M, Hasan D, Blijenberg BG, Hijdra A, van Gijn J. Xanthochromia after subarachnoid haemorrhage needs no revisitation. J Neurol Neurosurg Psychiatry 1989; 52:826–828.
- Molyneaux AJ, Kerr RS, Yu LM, et al; International Subarachnoid Aneurysm Trial (ISAT) Collaborative Group. International subarachnoid hemorrhage trial (ISAT) of neurosurgical clipping versus endovascular coiling in 2,143 patients with ruptured intracranial aneurysms: a randomised comparison of effects on survival, dependency, seizures, rebleeding, subgroups, and aneurysm occlusion. Lancet 2005; 366:809–817.
- Caglayan HZ, Colpak IA, Kansu T. A diagnostic challenge: dilated pupil. Curr Opin Ophthalmol 2013; 24:550–557.
- Brophy GM, Bell R, Claassen J, et al; Neurocritical Care Society Status Epilepticus Guideline Writing Committee. Guidelines for the evaluation and management of status epilepticus. Neurocrit Care 2012; 17:3–23.
- Chang CW, Bleck TP. Status epilepticus. Neurol Clin 1995; 13:529–548.
- Treiman DM. Generalized convulsive status epilepticus in the adult. Epilepsia 1993; 34(suppl 1):S2–S11.
- Leppick IE. Status epilepticus: the next decade. Neurology 1990; 40(suppl 2):4–9.
- Aranda A, Foucart G, Ducassé JL, Grolleau S, McGonigal A, Valton L. Generalized convulsive status epilepticus management in adults: a cohort study with evaluation of professional practice. Epilepsia 2010; 51:2159–2167.
- DeLorenzo RJ, Waterhouse EJ, Towne AR, et al. Persistent nonconvulsive status epilepticus after the control of convulsive status epilepticus. Epilepsia 1998; 39:833–840.
- Casha S, Christie S. A systematic review of intensive cardiopulmonary management after spinal cord injury. J Neurotrauma 2011; 28:1479–1495.
- Walters BC, Hadley MN, Hurlbert RJ, et al; American Association of Neurological Surgeons; Congress of Neurological Surgeons. Guidelines for the management of acute cervical spine and spinal cord injuries: 2013 update. Neurosurgery 2013; 60(suppl 1):82–91.
- Hurlbert RJ, Hadley MN, Walters BC, et al. Pharmacological therapy for acute spinal cord injury. Neurosurgery 2013; 72(suppl 2):93–105.
- Cohen MS, Younger D. Aspects of the natural history of myasthenia gravis: crisis and death. Ann NY Acad Sci 1981; 377:670–677.
- Belack RS, Sanders DB. On the concept of myasthenic crisis. J Clin Neuromuscul Dis 2002; 4:40–42.
- Chaudhuri A, Behan PO. Myasthenic crisis. QJM 2009; 102:97–107.
- Mayer SA. Intensive care of the myasthenic patient. Neurology 1997; 48(suppl 5):70S–75S.
- Jani-Acsadi A, Lisak RP. Myasthenic crisis: guidelines for prevention and treatment. J Neurol Sci 2007; 261:127–133.
- Bershad EM, Feen ES, Suarez JI. Myasthenia gravis crisis. South Med J 2008; 101:63–69.
- Ahmed S, Kirmani JF, Janjua N, et al. An update on myasthenic crisis. Curr Treat Options Neurol 2005; 7:129–141.
- Godoy DA, Vaz de Mello LJ, Masotti L, Napoli MD. The myasthenic patient in crisis: an update of the management in neurointensive care unit. Arq Neuropsiquiatr 2013; 71:627–639.
- Hughes RA, Wijdicks EF, Benson E, et al; Multidisciplinary Consensus Group. Supportive care for patients with Guillain-Barré syndrome: Arch Neurol 2005; 62:1194–1198.
Neurologic emergencies such as acute stroke, status epilepticus, subarachnoid hemorrhage, neuromuscular weakness, and spinal cord injury affect millions of Americans yearly.1,2 These conditions can be difficult to diagnose, and delays in recognition and treatment can have devastating results. Consequently, it is important for nonneurologists to be able to quickly recognize these conditions and initiate timely management, often while awaiting neurologic consultation.
Here, we review how to recognize and treat these common, serious conditions.
ACUTE ISCHEMIC STROKE: TIME IS OF THE ESSENCE
Stroke is the fourth leading cause of death in the United States and is one of the most common causes of disability worldwide.3–5 About 85% of strokes are ischemic, resulting from diminished vascular supply to the brain. Symptoms such as facial droop, unilateral weakness or numbness, aphasia, gaze deviation, and unsteadiness of gait may be seen. Time is of the essence, as all currently available interventions are safe and effective only within defined time windows.
Diagnosis and assessment
When acute ischemic stroke is suspected, the clinical history, time of onset, and basic neurologic examination should be obtained quickly.
The National Institutes of Health (NIH) stroke scale is an objective marker for assessing stroke severity as well as evolution of disease and should be obtained in all stroke patients. Scores range from 0 (best) to 42 (worst) (www.ninds.nih.gov/doctors/NIH_Stroke_Scale.pdf).
Time of onset of symptoms is essential to determine, since it guides eligibility for acute therapies. Clinicians should ascertain the last time the patient was seen to be neurologically well in order to estimate this time window as closely as possible.
Laboratory tests should include a fingerstick blood glucose measurement, coagulation studies, complete blood cell count, and basic metabolic profile.
Computed tomography (CT) of the head without contrast should be obtained immediately to exclude acute hemorrhage and any alternative diagnoses that could explain the patient’s symptoms. Acute brain ischemia is often not apparent on CT during the first few hours of injury. Therefore, a patient presenting with new focal neurologic deficits and an unremarkable result on CT of the head should be treated as having had an acute ischemic stroke, and interventional therapies should be considered.
Stroke mimics should be considered and treated, as appropriate (Table 1).
Acute management of ischemic stroke
Acute treatment should not be delayed by obtaining chest radiography, inserting a Foley catheter, or obtaining an electrocardiogram. The longer the time that elapses before treatment, the worse the functional outcome, underscoring the need for rapid decision-making.6–8
Lowering the head of the bed may provide benefit by promoting blood flow to ischemic brain tissue.9 However, this should not be done in patients with significantly elevated intracerebral pressure and concern for herniation.
Permissive hypertension (antihypertensive treatment only for blood pressure greater than 220/110 mm Hg) should be allowed per national guidelines to provide adequate perfusion to brain areas at risk of injury.10
Tissue plasminogen activator. Patients with ischemic stroke who present within 3 hours of symptom onset should be considered for intravenous administration of tissue plasminogen activator (tPA), a safe and effective therapy with nearly 2 decades of evidence to support its use.10 The treating physician should carefully review the risks and benefits of this therapy.
To receive tPA, the patient must have all of the following:
- Clinical diagnosis of ischemic stroke with measurable neurologic deficit
- Onset of symptoms within the past 3 hours
- Age 18 or older.
The patient must not have any of the following:
- Significant stroke within the past 3 months
- Severe traumatic head injury within the past 3 months
- History of significant intracerebral hemorrhage
- Previously ruptured arteriovenous malformation or intracranial aneurysm
- Central nervous system neoplasm
- Arterial puncture at a noncompressible site within the past 7 days
- Evidence of hemorrhage on CT of the head
- Evidence of ischemia in greater than 33% of the cerebral hemisphere on head CT
- History and symptoms strongly suggesting subarachnoid hemorrhage
- Persistent hypertension (systolic pressure ≥ 185 mm Hg or diastolic pressure ≥ 110 mm Hg)
- Evidence of acute significant bleeding (external or internal)
- Hypoglycemia—ie, serum glucose less than 50 mg/dL (< 2.8 mmol/L)
- Thrombocytopenia (platelet count < 100 × 109/L)
- Significant coagulopathy (international normalized ratio > 1.7, prothrombin time > 15 seconds, or abnormally elevated activated partial thromboplastin time)
- Current use of a factor Xa inhibitor or direct thrombin inhibitor.
Relative contraindications:
- Minor or rapidly resolving symptoms
- Major surgery or trauma within the past 14 days
- Gastrointestinal or urinary tract bleeding within the past 21 days
- Myocardial infarction in the past 3 months
- Unruptured intracranial aneurysm
- Seizure occurring at stroke onset
- Pregnancy.
If these criteria are satisfied, tPA should be given at a dose of 0.9 mg/kg intravenously over 60 minutes. Ten percent of the dose should be given as an initial bolus, followed by a constant infusion of the remaining 90% over 1 hour.
If tPA is given, the blood pressure must be kept lower than 185/110 mm Hg to minimize the risk of symptomatic intracerebral hemorrhage.
A subset of patients may benefit from receiving intravenous tPA between 3 and 4.5 hours after the onset of stroke symptoms. These include patients who are no more than 80 years old, who have not recently used oral anticoagulants, who do not have severe neurologic injury (ie, do not have NIH Stroke Scale scores > 25), and who do not have diabetes mellitus or a history of ischemic stroke.11 Although many hospitals have such a protocol for tPA up to 4.5 hours after the onset of stroke symptoms, this time window is not currently approved by the US Food and Drug Administration.
Intra-arterial therapy. Based on recent trials, some patients may benefit further from intra-arterial thrombolysis or mechanical thrombectomy, both delivered during catheter-based cerebral angiography, independent of intravenous tPA administration.12,13 These patients should be evaluated on a case-by-case basis by a neurologist and neurointerventional team. Time windows for these treatments generally extend to 6 hours from stroke onset and perhaps even longer in some situations (eg, basilar artery occlusion).
An antiplatelet agent should be started quickly in all stroke patients who do not receive tPA. Patients who receive tPA can begin receiving an antiplatelet agent 24 hours afterward.
Unfractionated heparin. There is no evidence to support the use of unfractionated heparin in most cases of acute ischemic stroke.10
Glucose control (in the range of 140–180 mg/dL) and fever control remain essential elements of post-acute stroke care to provide additional protection to the damaged brain.
For ischemic stroke due to atrial fibrillation
In ischemic stroke due to atrial fibrillation, early anticoagulation should be considered, based on the CHA2DS2-VASC risk of ischemic stroke vs the HAS-BLED risk of hemorrhage (calculators available at www.mdcalc.com).
In general, anticoagulation may be withheld during the first 72 hours while further stroke workup and evaluation of extent of injury are carried out, as there is an increased risk of hemorrhagic transformation of the ischemic stroke. Often, anticoagulation is resumed at a full dose between 72 hours and 2 weeks of the ischemic stroke.
ACUTE HEMORRHAGIC STROKE: BLOOD PRESSURE, COAGULATION
Approximately 15% of strokes are caused by intracerebral hemorrhage, which can be detected with noncontrast head CT with a sensitivity of 98.6% within 6 hours of the onset of bleeding.14 A common underlying cause of intracerebral hemorrhage is chronic poorly controlled hypertension, causing rupture of damaged (or “lipohyalinized”) vessels with resultant blood extravasation into the brain parenchyma. Other causes are less common (Table 2).
Treatment of acute hemorrhagic stroke
Acute treatment of intracerebral hemorrhage includes blood pressure control, reversal of underlying coagulopathy or anticoagulation, and sometimes intracranial pressure control. There is little role for surgery in most cases, based on findings of randomized trials.15
Blood pressure control. Many studies have investigated optimal blood pressure goals in acute intracerebral hemorrhage. Recent data suggest that early aggressive therapy, targeting a systolic blood pressure goal less than 140 mm Hg within the first hour, is safe and can lead to better functional outcomes than a more conservative blood-pressure-lowering target.16 Rapid-onset, short-acting antihypertensive agents in intravenous form, such as nicardipine and labetalol, are frequently used. Of note, this treatment strategy for hemorrhagic stroke is in direct contrast to the treatment of ischemic stroke, in which permissive hypertension (blood pressure goal < 220/110 mm Hg) is often pursued.
Reversal of any coagulation abnormalities should be done quickly in intracranial hemorrhage. Warfarin use has been shown to be a strong independent predictor of intracranial hemorrhage expansion, which increases the risk of death.17,18
Increasingly, agents other than vitamin K or fresh-frozen plasma are being used to rapidly reverse anticoagulation, including prothrombin complex concentrate (available in three- and four-factor preparations) and recombinant factor VIIa. While four-factor prothrombin complex concentrate and recombinant factor VIIa have been shown to be more efficacious than fresh-frozen plasma, there are limited data directly comparing these newer reversal agents against each other.19 The use of these medications is limited by availability and practitioner familiarity.20–22
Reversing anticoagulation due to target-specific oral anticoagulants. The acute management of intracranial hemorrhage in patients taking the new target-specific oral anticoagulants (eg, dabigatran, apixaban, rivaroxaban, edoxaban) remains challenging. Laboratory tests such as factor Xa levels are not readily available in many institutions and do not provide results in a timely fashion, and in the interim, acute hemorrhage and clinical deterioration may occur. Management strategies involve giving fresh-frozen plasma, prothrombin complex concentrate, and consideration of hemodialysis.23 Dabigatran reversal with idarucizumab has recently been shown to have efficacy.24
Vigilance for elevated intracranial pressure. Intracranial hemorrhage can occasionally cause elevated intracranial pressure, which should be treated rapidly. Any acute decline in mental status in a patient with intracranial hemorrhage requires emergency imaging to evaluate for expansion of hemorrhage.
SUBARACHNOID HEMORRHAGE
The sudden onset of a “thunderclap” headache (often described by patients as “the worst headache of my life”) suggests subarachnoid hemorrhage.
In contrast to intracranial hemorrhage, in subarachnoid hemorrhage blood collects mainly in the cerebral spinal fluid-containing spaces surrounding the brain, leading to a higher incidence of hydrocephalus from impaired drainage of cerebrospinal fluid. Nontraumatic subarachnoid hemorrhage is most often caused by rupture of an intracranial aneurysm, which can be a devastating event, with death rates approaching 50%.25
Diagnosis of subarachnoid hemorrhage
Noncontrast CT of the head is the main modality for diagnosing subarachnoid hemorrhage. Blood within the subarachnoid space is demonstrable in 92% of cases if CT is performed within the first 24 hours of hemorrhage, with an initial sensitivity of about 95% within the first 6 hours of onset.14,26,27 The longer CT is delayed, the lower the sensitivity.
Some studies suggest that a protocol of CT followed by CT angiography can safely exclude aneurysmal subarachnoid hemorrhage and obviate the need for lumbar puncture. However, further research is required to validate this approach.28
Lumbar puncture. If clinical suspicion of subarachnoid hemorrhage remains strong even though initial CT is negative, lumbar puncture must be performed for cerebrospinal fluid analysis.29 Xanthochromia (a yellowish pigmentation of the cerebrospinal fluid due to the degeneration of blood products that occurs within 8 to 12 hours of bleeding) should raise the alarm for subarachnoid hemorrhage; this sign may be present up to 4 weeks after the bleeding event.30
If lumbar puncture is contraindicated, then aneurysmal subarachnoid hemorrhage has not been ruled out, and further neurologic consultation should be pursued.
Management of subarachnoid hemorrhage
Early management of blood pressure for a ruptured intracranial aneurysm follows strategies similar to those for intracranial hemorrhage. Further investigation is rapidly directed toward an underlying vascular malformation, with intracranial vessel imaging such as CT angiography, magnetic resonance angiography, or the gold standard test—catheter-based cerebral angiography.
Aneurysms are treated (or “secured”) either by surgical clipping or by endovascular coiling. Endovascular coiling is preferable in cases in which both can be safely attempted.31 If the facility lacks the resources to do these procedures, the patient should be referred to a nearby tertiary care center.
INTRACRANIAL HYPERTENSION: DANGER OF BRAIN HERNIATION
A number of conditions can cause an acute intracranial pressure elevation. The danger of brain herniation requires that therapies be implemented rapidly to prevent catastrophic neurologic injury. In many situations, nonneurologists are the first responders and therefore should be familiar with basic intracranial pressure management.
Initial symptoms of acute rise in intracranial pressure
As intracranial pressure rises, pressure is typically equally distributed throughout the cranial vault, leading to dysfunction of the ascending reticular activating system, which clinically manifests as the inability to stay alert despite varying degrees of noxious stimulation. Progressive cranial neuropathies (often starting with pupillary abnormalities) and coma are often seen in this setting as the upper brainstem begins to be compressed.
Initial assessment and treatment of elevated intracranial pressure
Noncontrast CT of the head is often obtained immediately when acutely elevated intracranial pressure is suspected. If clinical examination and radiographic findings are consistent with intracranial hypertension, prompt measures can be started at the bedside.
Elevate the head of the bed to 30 degrees to promote venous drainage and reduce intracranial pressure. (In contrast, most other hemodynamically unstable patients are placed flat or in the Trendelenburg position.)
Intubation should be done quickly in cases of airway compromise, and hyperventilation should be started with a goal Paco2 of 30 to 35 mm Hg. This hypocarbic strategy promotes cerebral vasoconstriction and a transient decrease in intracranial pressure.
Hyperosmolar therapy allows for transient intracranial volume decompression and is the mainstay of emergency medical treatment of intracranial hypertension. Mannitol is a hyperosmolar polysaccharide that promotes osmotic diuresis and removes excessive cerebral water. In the acute setting, it can be given as an intravenous bolus of 1 to 2 g/kg through a peripheral intravenous line, followed by a bolus every 4 to 6 hours. Hypotension can occur after diuresis, and renal function should be closely monitored since frequent mannitol use can promote acute tubular necrosis. In patients who are anuric, the medication is typically not used.
Hypertonic saline (typically 3% sodium chloride, though different concentrations are available) is an alternative that helps draw interstitial fluid into the intravascular space, decreasing cerebral edema and maintaining hemodynamic stability. Relative contraindications include congestive heart failure or renal failure leading to pulmonary edema from volume overload. Hypertonic saline can be given as a bolus or a constant infusion. Some institutions have rapid access to 23.4% saline, which can be given as a 30-mL bolus but typically requires a central venous catheter for rapid infusion.
Comatose patients with radiographic findings of hydrocephalus, epidural or subdural hematoma, or mass effect with midline shift warrant prompt neurosurgical consultation for further surgical measures of intracranial pressure control and monitoring.
The ‘blown’ pupil
The physician should be concerned about elevated intracranial pressure if a patient has mydriasis, ie, an abnormally dilated (“blown”) pupil, which is a worrisome sign in the setting of true intracranial hypertension. However, many different processes can cause mydriasis and should be kept in mind when evaluating this finding (Table 3).32 If radiographic findings do not suggest elevated intracranial pressure, further workup into these other processes should be pursued.
STATUS EPILEPTICUS: SEIZURE CONTROL IS IMPORTANT
A continuous unremitting seizure lasting longer than 5 minutes or recurrent seizure activity in a patient who does not regain consciousness between seizures should be treated as status epilepticus. All seizure types carry the risk of progressing to status epilepticus, and responsiveness to antiepileptic drug therapy is inversely related to the duration of seizures. It is imperative that seizure activity be treated early and aggressively to prevent recalcitrant seizure activity, neuronal damage, and progression to status epilepticus.33
Once the ABCs of emergency stabilization have been performed (ie, airway, breathing, circulation), antiepileptic drug therapy should start immediately using established algorithms (Figure 1).34–36 During the course of treatment, the reliability of the neurologic examination may be limited due to medication effects or continued status epilepticus, making continuous video electroencephalographic monitoring often necessary to guide further therapy in patients who are not rapidly recovering.34–38
Once status epilepticus has resolved, further investigation into the underlying cause should be pursued quickly, especially in patients without a previous diagnosis of epilepsy. Head CT with contrast or magnetic resonance imaging can be used to look for any structural abnormality that may explain seizures. Basic laboratory tests including toxicology screening can identify a common trigger such as hypoglycemia or stimulant use. Fever or other possible signs of meningitis should be investigated further with cerebrospinal fluid analysis.
SPINAL CORD INJURY
Acute spinal cord injury can lead to substantial long-term neurologic impairment and should be suspected in any patient presenting with focal motor loss, sensory loss, or both with sparing of the cranial nerves and mental status. Causes of injury include compression (traumatic or nontraumatic) and inflammatory and noninflammatory myelopathies.
The location of the injury can be inferred by analyzing the symptoms, which can point to the cord level and indicate whether the anterior or posterior of the cord is involved. Anterior cord injury tends to affect the descending corticospinal and pyramidal tracts, resulting in motor deficits and weakness. Posterior cord injury involves the dorsal columns, leading to deficits of vibration sensation and proprioception. High cervical cord injuries tend to involve varying degrees of quadriparesis, sensory loss, and sometimes respiratory compromise. A clinical history of bilateral lower-extremity weakness, a “band-like” sensory complaint around the lower chest or abdomen, or both, can suggest thoracic cord involvement. Symptoms isolated to one or both lower extremities along with lower back pain and bowel or bladder involvement may point to injury of the lumbosacral cord.
Basic management of spinal cord injury includes decompression of the bladder and initial protection against further injury with a stabilizing collar or brace.
Magnetic resonance imaging with and without contrast is the ideal study to evaluate injuries to the spinal cord itself. While CT is helpful in identifying bony disease of the spinal column (eg, evaluating traumatic fractures), it is not helpful in viewing intrinsic cord pathology.
Traumatic myelopathy
Traumatic spinal cord injury is usually suggested by the clinical history and confirmed with CT. In this setting, early consultation with a neurosurgeon is required to prevent permanent cord injury.
Guidelines suggest maintaining a mean arterial pressure greater than 85 to 90 mm Hg for the first 7 days after traumatic spinal cord injury, a particular problem in the setting of hemodynamic instability, which can accompany lesions above the midthoracic level.39,40
Patients with vertebral body misalignment should be placed in an appropriate stabilizing collar or brace until a medically trained professional deems it appropriate to discontinue the device, or until surgical stabilization is performed.
Methylprednisone is a controversial intervention for acute spinal cord trauma, lacking clear benefit in meta-analyses.41
Nontraumatic compressive myelopathy
Patients with nontraumatic compressive myelopathy tend to present with varying degrees of back pain and worsening sensorimotor function. The differential diagnosis includes epidural abscesses, hematoma, metastatic neoplasm, and osteophyte compression (Table 4). The clinical history helps to guide therapy and should involve assessment for previous spinal column injury, immunocompromised state, travel history (which provides information on risks of exposure to a variety of diseases, including infections), and constitutional symptoms such as fever and weight loss.
Epidural abscess can have devastating results if missed. Red flags such as recent illness, intravenous drug use, focal back pain, fever, worsening numbness or weakness, and bowel or bladder incontinence should raise suspicion of this disorder. Emergency magnetic resonance imaging is required to diagnose this condition, and treatment involves urgent administration of antibiotics and consideration of surgical drainage.
Noncompressive myelopathies
There are numerous causes of noncompressive spinal cord injury (Table 4), and the etiology may be inflammatory (eg, “myelitis”) or noninflammatory. The diagnostic workup may require both magnetic resonance imaging and cerebrospinal fluid analysis. Acute disease-targeted therapy is rarely indicated and can be deferred until a full diagnostic workup has been completed.
NEUROMUSCULAR DISEASE: IS VENTILATION NEEDED?
Diseases involving the motor components of the peripheral nervous system (Table 5) share the common risk of causing ventilatory failure due to weakness of the diaphragm, intercostal muscles, and upper-airway muscles. Clinicians need to be aware of this risk and view these disorders as neurologic emergencies.
Determining when these patients require mechanical intubation is a challenge. Serial measurements of maximum inspiratory force and vital capacity are important and can be accomplished quickly at the bedside by a respiratory therapist. A maximum inspiratory force less than –30 cm H2O or a vital capacity less than 20 mL/kg, or both, are worrisome markers that raise concern for impending ventilatory failure. Serial measurements can detect changes in these values that might indicate the need for elective intubation. In any patient presenting with weakness of the limbs, these measurements are an important step in the initial evaluation.
Myasthenic crisis
Myasthenia gravis is caused by autoantibodies directed against postsynaptic acetylcholine receptors. Patients demonstrate muscle weakness, usually in a proximal pattern, with fatigue, respiratory distress, nasal speech, ophthalmoparesis, and dysphagia. Exacerbations can occur as a response to recent infection, surgery, or medications such as neuromuscular blocking agents or aminoglycosides.
Myasthenic crisis, while uncommon, is a life-threatening emergency characterized by bulbar or respiratory failure secondary to muscle weakness. It can occur in patients already diagnosed with myasthenia gravis or may be the initial manifestation of the disease.42–49 Intubation and mechanical ventilation are frequently required. Postoperative myasthenic patients in whom extubation has been delayed more than 24 hours should be considered in crisis.45
The diagnosis of myasthenia gravis can be made by serum autoantibody testing, electromyography, and nerve conduction studies (with repetitive stimulation) or administration of edrophonium in patients with obvious ptosis.
The mainstay of therapy for myasthenic crisis is either intravenous immunoglobulin at a dose of 2 g/kg over 2 to 5 days or plasmapheresis (5–7 exchanges over 7–14 days). Corticosteroids are not recommended in myasthenic crisis in patients who are not intubated, as they can potentiate an initial worsening of crisis. Once the patient begins to show clinical improvement, outpatient pyridostigmine and immunosuppressive medications can be resumed at a low dose and titrated as tolerated.
Acute inflammatory demyelinating polyneuropathy (Guillain-Barré syndrome)
Acute inflammatory demyelinating polyneuropathy is an autoimmune disorder involving autoantibodies against axons or myelin in the peripheral nervous system.
This disease should be suspected in a patient who is developing worsening muscle weakness (usually with areflexia) over the course of days to weeks. Occasionally, a recent diarrheal or other systemic infectious trigger can be identified. Blood pressure instability and cardiac arrhythmia can also be seen due to autonomic nerve involvement. Although classically described as an “ascending paralysis,” other variants of this disease have distinct clinical presentations (eg, the descending paralysis, ataxia, areflexia, ophthalmoparesis of the Miller Fisher syndrome).
Acute inflammatory demyelinating polyneuropathy is diagnosed by electromyography and nerve conduction studies. A cerebrospinal fluid profile demonstrating elevated protein and few white blood cells is typical.
Treatment, as in myasthenic crisis, involves intravenous immunoglobulin or plasmapheresis. Corticosteroids are ineffective. Anticipation of ventilatory failure and expectant intubation is essential, given the progressive nature of the disorder.50
Neurologic emergencies such as acute stroke, status epilepticus, subarachnoid hemorrhage, neuromuscular weakness, and spinal cord injury affect millions of Americans yearly.1,2 These conditions can be difficult to diagnose, and delays in recognition and treatment can have devastating results. Consequently, it is important for nonneurologists to be able to quickly recognize these conditions and initiate timely management, often while awaiting neurologic consultation.
Here, we review how to recognize and treat these common, serious conditions.
ACUTE ISCHEMIC STROKE: TIME IS OF THE ESSENCE
Stroke is the fourth leading cause of death in the United States and is one of the most common causes of disability worldwide.3–5 About 85% of strokes are ischemic, resulting from diminished vascular supply to the brain. Symptoms such as facial droop, unilateral weakness or numbness, aphasia, gaze deviation, and unsteadiness of gait may be seen. Time is of the essence, as all currently available interventions are safe and effective only within defined time windows.
Diagnosis and assessment
When acute ischemic stroke is suspected, the clinical history, time of onset, and basic neurologic examination should be obtained quickly.
The National Institutes of Health (NIH) stroke scale is an objective marker for assessing stroke severity as well as evolution of disease and should be obtained in all stroke patients. Scores range from 0 (best) to 42 (worst) (www.ninds.nih.gov/doctors/NIH_Stroke_Scale.pdf).
Time of onset of symptoms is essential to determine, since it guides eligibility for acute therapies. Clinicians should ascertain the last time the patient was seen to be neurologically well in order to estimate this time window as closely as possible.
Laboratory tests should include a fingerstick blood glucose measurement, coagulation studies, complete blood cell count, and basic metabolic profile.
Computed tomography (CT) of the head without contrast should be obtained immediately to exclude acute hemorrhage and any alternative diagnoses that could explain the patient’s symptoms. Acute brain ischemia is often not apparent on CT during the first few hours of injury. Therefore, a patient presenting with new focal neurologic deficits and an unremarkable result on CT of the head should be treated as having had an acute ischemic stroke, and interventional therapies should be considered.
Stroke mimics should be considered and treated, as appropriate (Table 1).
Acute management of ischemic stroke
Acute treatment should not be delayed by obtaining chest radiography, inserting a Foley catheter, or obtaining an electrocardiogram. The longer the time that elapses before treatment, the worse the functional outcome, underscoring the need for rapid decision-making.6–8
Lowering the head of the bed may provide benefit by promoting blood flow to ischemic brain tissue.9 However, this should not be done in patients with significantly elevated intracerebral pressure and concern for herniation.
Permissive hypertension (antihypertensive treatment only for blood pressure greater than 220/110 mm Hg) should be allowed per national guidelines to provide adequate perfusion to brain areas at risk of injury.10
Tissue plasminogen activator. Patients with ischemic stroke who present within 3 hours of symptom onset should be considered for intravenous administration of tissue plasminogen activator (tPA), a safe and effective therapy with nearly 2 decades of evidence to support its use.10 The treating physician should carefully review the risks and benefits of this therapy.
To receive tPA, the patient must have all of the following:
- Clinical diagnosis of ischemic stroke with measurable neurologic deficit
- Onset of symptoms within the past 3 hours
- Age 18 or older.
The patient must not have any of the following:
- Significant stroke within the past 3 months
- Severe traumatic head injury within the past 3 months
- History of significant intracerebral hemorrhage
- Previously ruptured arteriovenous malformation or intracranial aneurysm
- Central nervous system neoplasm
- Arterial puncture at a noncompressible site within the past 7 days
- Evidence of hemorrhage on CT of the head
- Evidence of ischemia in greater than 33% of the cerebral hemisphere on head CT
- History and symptoms strongly suggesting subarachnoid hemorrhage
- Persistent hypertension (systolic pressure ≥ 185 mm Hg or diastolic pressure ≥ 110 mm Hg)
- Evidence of acute significant bleeding (external or internal)
- Hypoglycemia—ie, serum glucose less than 50 mg/dL (< 2.8 mmol/L)
- Thrombocytopenia (platelet count < 100 × 109/L)
- Significant coagulopathy (international normalized ratio > 1.7, prothrombin time > 15 seconds, or abnormally elevated activated partial thromboplastin time)
- Current use of a factor Xa inhibitor or direct thrombin inhibitor.
Relative contraindications:
- Minor or rapidly resolving symptoms
- Major surgery or trauma within the past 14 days
- Gastrointestinal or urinary tract bleeding within the past 21 days
- Myocardial infarction in the past 3 months
- Unruptured intracranial aneurysm
- Seizure occurring at stroke onset
- Pregnancy.
If these criteria are satisfied, tPA should be given at a dose of 0.9 mg/kg intravenously over 60 minutes. Ten percent of the dose should be given as an initial bolus, followed by a constant infusion of the remaining 90% over 1 hour.
If tPA is given, the blood pressure must be kept lower than 185/110 mm Hg to minimize the risk of symptomatic intracerebral hemorrhage.
A subset of patients may benefit from receiving intravenous tPA between 3 and 4.5 hours after the onset of stroke symptoms. These include patients who are no more than 80 years old, who have not recently used oral anticoagulants, who do not have severe neurologic injury (ie, do not have NIH Stroke Scale scores > 25), and who do not have diabetes mellitus or a history of ischemic stroke.11 Although many hospitals have such a protocol for tPA up to 4.5 hours after the onset of stroke symptoms, this time window is not currently approved by the US Food and Drug Administration.
Intra-arterial therapy. Based on recent trials, some patients may benefit further from intra-arterial thrombolysis or mechanical thrombectomy, both delivered during catheter-based cerebral angiography, independent of intravenous tPA administration.12,13 These patients should be evaluated on a case-by-case basis by a neurologist and neurointerventional team. Time windows for these treatments generally extend to 6 hours from stroke onset and perhaps even longer in some situations (eg, basilar artery occlusion).
An antiplatelet agent should be started quickly in all stroke patients who do not receive tPA. Patients who receive tPA can begin receiving an antiplatelet agent 24 hours afterward.
Unfractionated heparin. There is no evidence to support the use of unfractionated heparin in most cases of acute ischemic stroke.10
Glucose control (in the range of 140–180 mg/dL) and fever control remain essential elements of post-acute stroke care to provide additional protection to the damaged brain.
For ischemic stroke due to atrial fibrillation
In ischemic stroke due to atrial fibrillation, early anticoagulation should be considered, based on the CHA2DS2-VASC risk of ischemic stroke vs the HAS-BLED risk of hemorrhage (calculators available at www.mdcalc.com).
In general, anticoagulation may be withheld during the first 72 hours while further stroke workup and evaluation of extent of injury are carried out, as there is an increased risk of hemorrhagic transformation of the ischemic stroke. Often, anticoagulation is resumed at a full dose between 72 hours and 2 weeks of the ischemic stroke.
ACUTE HEMORRHAGIC STROKE: BLOOD PRESSURE, COAGULATION
Approximately 15% of strokes are caused by intracerebral hemorrhage, which can be detected with noncontrast head CT with a sensitivity of 98.6% within 6 hours of the onset of bleeding.14 A common underlying cause of intracerebral hemorrhage is chronic poorly controlled hypertension, causing rupture of damaged (or “lipohyalinized”) vessels with resultant blood extravasation into the brain parenchyma. Other causes are less common (Table 2).
Treatment of acute hemorrhagic stroke
Acute treatment of intracerebral hemorrhage includes blood pressure control, reversal of underlying coagulopathy or anticoagulation, and sometimes intracranial pressure control. There is little role for surgery in most cases, based on findings of randomized trials.15
Blood pressure control. Many studies have investigated optimal blood pressure goals in acute intracerebral hemorrhage. Recent data suggest that early aggressive therapy, targeting a systolic blood pressure goal less than 140 mm Hg within the first hour, is safe and can lead to better functional outcomes than a more conservative blood-pressure-lowering target.16 Rapid-onset, short-acting antihypertensive agents in intravenous form, such as nicardipine and labetalol, are frequently used. Of note, this treatment strategy for hemorrhagic stroke is in direct contrast to the treatment of ischemic stroke, in which permissive hypertension (blood pressure goal < 220/110 mm Hg) is often pursued.
Reversal of any coagulation abnormalities should be done quickly in intracranial hemorrhage. Warfarin use has been shown to be a strong independent predictor of intracranial hemorrhage expansion, which increases the risk of death.17,18
Increasingly, agents other than vitamin K or fresh-frozen plasma are being used to rapidly reverse anticoagulation, including prothrombin complex concentrate (available in three- and four-factor preparations) and recombinant factor VIIa. While four-factor prothrombin complex concentrate and recombinant factor VIIa have been shown to be more efficacious than fresh-frozen plasma, there are limited data directly comparing these newer reversal agents against each other.19 The use of these medications is limited by availability and practitioner familiarity.20–22
Reversing anticoagulation due to target-specific oral anticoagulants. The acute management of intracranial hemorrhage in patients taking the new target-specific oral anticoagulants (eg, dabigatran, apixaban, rivaroxaban, edoxaban) remains challenging. Laboratory tests such as factor Xa levels are not readily available in many institutions and do not provide results in a timely fashion, and in the interim, acute hemorrhage and clinical deterioration may occur. Management strategies involve giving fresh-frozen plasma, prothrombin complex concentrate, and consideration of hemodialysis.23 Dabigatran reversal with idarucizumab has recently been shown to have efficacy.24
Vigilance for elevated intracranial pressure. Intracranial hemorrhage can occasionally cause elevated intracranial pressure, which should be treated rapidly. Any acute decline in mental status in a patient with intracranial hemorrhage requires emergency imaging to evaluate for expansion of hemorrhage.
SUBARACHNOID HEMORRHAGE
The sudden onset of a “thunderclap” headache (often described by patients as “the worst headache of my life”) suggests subarachnoid hemorrhage.
In contrast to intracranial hemorrhage, in subarachnoid hemorrhage blood collects mainly in the cerebral spinal fluid-containing spaces surrounding the brain, leading to a higher incidence of hydrocephalus from impaired drainage of cerebrospinal fluid. Nontraumatic subarachnoid hemorrhage is most often caused by rupture of an intracranial aneurysm, which can be a devastating event, with death rates approaching 50%.25
Diagnosis of subarachnoid hemorrhage
Noncontrast CT of the head is the main modality for diagnosing subarachnoid hemorrhage. Blood within the subarachnoid space is demonstrable in 92% of cases if CT is performed within the first 24 hours of hemorrhage, with an initial sensitivity of about 95% within the first 6 hours of onset.14,26,27 The longer CT is delayed, the lower the sensitivity.
Some studies suggest that a protocol of CT followed by CT angiography can safely exclude aneurysmal subarachnoid hemorrhage and obviate the need for lumbar puncture. However, further research is required to validate this approach.28
Lumbar puncture. If clinical suspicion of subarachnoid hemorrhage remains strong even though initial CT is negative, lumbar puncture must be performed for cerebrospinal fluid analysis.29 Xanthochromia (a yellowish pigmentation of the cerebrospinal fluid due to the degeneration of blood products that occurs within 8 to 12 hours of bleeding) should raise the alarm for subarachnoid hemorrhage; this sign may be present up to 4 weeks after the bleeding event.30
If lumbar puncture is contraindicated, then aneurysmal subarachnoid hemorrhage has not been ruled out, and further neurologic consultation should be pursued.
Management of subarachnoid hemorrhage
Early management of blood pressure for a ruptured intracranial aneurysm follows strategies similar to those for intracranial hemorrhage. Further investigation is rapidly directed toward an underlying vascular malformation, with intracranial vessel imaging such as CT angiography, magnetic resonance angiography, or the gold standard test—catheter-based cerebral angiography.
Aneurysms are treated (or “secured”) either by surgical clipping or by endovascular coiling. Endovascular coiling is preferable in cases in which both can be safely attempted.31 If the facility lacks the resources to do these procedures, the patient should be referred to a nearby tertiary care center.
INTRACRANIAL HYPERTENSION: DANGER OF BRAIN HERNIATION
A number of conditions can cause an acute intracranial pressure elevation. The danger of brain herniation requires that therapies be implemented rapidly to prevent catastrophic neurologic injury. In many situations, nonneurologists are the first responders and therefore should be familiar with basic intracranial pressure management.
Initial symptoms of acute rise in intracranial pressure
As intracranial pressure rises, pressure is typically equally distributed throughout the cranial vault, leading to dysfunction of the ascending reticular activating system, which clinically manifests as the inability to stay alert despite varying degrees of noxious stimulation. Progressive cranial neuropathies (often starting with pupillary abnormalities) and coma are often seen in this setting as the upper brainstem begins to be compressed.
Initial assessment and treatment of elevated intracranial pressure
Noncontrast CT of the head is often obtained immediately when acutely elevated intracranial pressure is suspected. If clinical examination and radiographic findings are consistent with intracranial hypertension, prompt measures can be started at the bedside.
Elevate the head of the bed to 30 degrees to promote venous drainage and reduce intracranial pressure. (In contrast, most other hemodynamically unstable patients are placed flat or in the Trendelenburg position.)
Intubation should be done quickly in cases of airway compromise, and hyperventilation should be started with a goal Paco2 of 30 to 35 mm Hg. This hypocarbic strategy promotes cerebral vasoconstriction and a transient decrease in intracranial pressure.
Hyperosmolar therapy allows for transient intracranial volume decompression and is the mainstay of emergency medical treatment of intracranial hypertension. Mannitol is a hyperosmolar polysaccharide that promotes osmotic diuresis and removes excessive cerebral water. In the acute setting, it can be given as an intravenous bolus of 1 to 2 g/kg through a peripheral intravenous line, followed by a bolus every 4 to 6 hours. Hypotension can occur after diuresis, and renal function should be closely monitored since frequent mannitol use can promote acute tubular necrosis. In patients who are anuric, the medication is typically not used.
Hypertonic saline (typically 3% sodium chloride, though different concentrations are available) is an alternative that helps draw interstitial fluid into the intravascular space, decreasing cerebral edema and maintaining hemodynamic stability. Relative contraindications include congestive heart failure or renal failure leading to pulmonary edema from volume overload. Hypertonic saline can be given as a bolus or a constant infusion. Some institutions have rapid access to 23.4% saline, which can be given as a 30-mL bolus but typically requires a central venous catheter for rapid infusion.
Comatose patients with radiographic findings of hydrocephalus, epidural or subdural hematoma, or mass effect with midline shift warrant prompt neurosurgical consultation for further surgical measures of intracranial pressure control and monitoring.
The ‘blown’ pupil
The physician should be concerned about elevated intracranial pressure if a patient has mydriasis, ie, an abnormally dilated (“blown”) pupil, which is a worrisome sign in the setting of true intracranial hypertension. However, many different processes can cause mydriasis and should be kept in mind when evaluating this finding (Table 3).32 If radiographic findings do not suggest elevated intracranial pressure, further workup into these other processes should be pursued.
STATUS EPILEPTICUS: SEIZURE CONTROL IS IMPORTANT
A continuous unremitting seizure lasting longer than 5 minutes or recurrent seizure activity in a patient who does not regain consciousness between seizures should be treated as status epilepticus. All seizure types carry the risk of progressing to status epilepticus, and responsiveness to antiepileptic drug therapy is inversely related to the duration of seizures. It is imperative that seizure activity be treated early and aggressively to prevent recalcitrant seizure activity, neuronal damage, and progression to status epilepticus.33
Once the ABCs of emergency stabilization have been performed (ie, airway, breathing, circulation), antiepileptic drug therapy should start immediately using established algorithms (Figure 1).34–36 During the course of treatment, the reliability of the neurologic examination may be limited due to medication effects or continued status epilepticus, making continuous video electroencephalographic monitoring often necessary to guide further therapy in patients who are not rapidly recovering.34–38
Once status epilepticus has resolved, further investigation into the underlying cause should be pursued quickly, especially in patients without a previous diagnosis of epilepsy. Head CT with contrast or magnetic resonance imaging can be used to look for any structural abnormality that may explain seizures. Basic laboratory tests including toxicology screening can identify a common trigger such as hypoglycemia or stimulant use. Fever or other possible signs of meningitis should be investigated further with cerebrospinal fluid analysis.
SPINAL CORD INJURY
Acute spinal cord injury can lead to substantial long-term neurologic impairment and should be suspected in any patient presenting with focal motor loss, sensory loss, or both with sparing of the cranial nerves and mental status. Causes of injury include compression (traumatic or nontraumatic) and inflammatory and noninflammatory myelopathies.
The location of the injury can be inferred by analyzing the symptoms, which can point to the cord level and indicate whether the anterior or posterior of the cord is involved. Anterior cord injury tends to affect the descending corticospinal and pyramidal tracts, resulting in motor deficits and weakness. Posterior cord injury involves the dorsal columns, leading to deficits of vibration sensation and proprioception. High cervical cord injuries tend to involve varying degrees of quadriparesis, sensory loss, and sometimes respiratory compromise. A clinical history of bilateral lower-extremity weakness, a “band-like” sensory complaint around the lower chest or abdomen, or both, can suggest thoracic cord involvement. Symptoms isolated to one or both lower extremities along with lower back pain and bowel or bladder involvement may point to injury of the lumbosacral cord.
Basic management of spinal cord injury includes decompression of the bladder and initial protection against further injury with a stabilizing collar or brace.
Magnetic resonance imaging with and without contrast is the ideal study to evaluate injuries to the spinal cord itself. While CT is helpful in identifying bony disease of the spinal column (eg, evaluating traumatic fractures), it is not helpful in viewing intrinsic cord pathology.
Traumatic myelopathy
Traumatic spinal cord injury is usually suggested by the clinical history and confirmed with CT. In this setting, early consultation with a neurosurgeon is required to prevent permanent cord injury.
Guidelines suggest maintaining a mean arterial pressure greater than 85 to 90 mm Hg for the first 7 days after traumatic spinal cord injury, a particular problem in the setting of hemodynamic instability, which can accompany lesions above the midthoracic level.39,40
Patients with vertebral body misalignment should be placed in an appropriate stabilizing collar or brace until a medically trained professional deems it appropriate to discontinue the device, or until surgical stabilization is performed.
Methylprednisone is a controversial intervention for acute spinal cord trauma, lacking clear benefit in meta-analyses.41
Nontraumatic compressive myelopathy
Patients with nontraumatic compressive myelopathy tend to present with varying degrees of back pain and worsening sensorimotor function. The differential diagnosis includes epidural abscesses, hematoma, metastatic neoplasm, and osteophyte compression (Table 4). The clinical history helps to guide therapy and should involve assessment for previous spinal column injury, immunocompromised state, travel history (which provides information on risks of exposure to a variety of diseases, including infections), and constitutional symptoms such as fever and weight loss.
Epidural abscess can have devastating results if missed. Red flags such as recent illness, intravenous drug use, focal back pain, fever, worsening numbness or weakness, and bowel or bladder incontinence should raise suspicion of this disorder. Emergency magnetic resonance imaging is required to diagnose this condition, and treatment involves urgent administration of antibiotics and consideration of surgical drainage.
Noncompressive myelopathies
There are numerous causes of noncompressive spinal cord injury (Table 4), and the etiology may be inflammatory (eg, “myelitis”) or noninflammatory. The diagnostic workup may require both magnetic resonance imaging and cerebrospinal fluid analysis. Acute disease-targeted therapy is rarely indicated and can be deferred until a full diagnostic workup has been completed.
NEUROMUSCULAR DISEASE: IS VENTILATION NEEDED?
Diseases involving the motor components of the peripheral nervous system (Table 5) share the common risk of causing ventilatory failure due to weakness of the diaphragm, intercostal muscles, and upper-airway muscles. Clinicians need to be aware of this risk and view these disorders as neurologic emergencies.
Determining when these patients require mechanical intubation is a challenge. Serial measurements of maximum inspiratory force and vital capacity are important and can be accomplished quickly at the bedside by a respiratory therapist. A maximum inspiratory force less than –30 cm H2O or a vital capacity less than 20 mL/kg, or both, are worrisome markers that raise concern for impending ventilatory failure. Serial measurements can detect changes in these values that might indicate the need for elective intubation. In any patient presenting with weakness of the limbs, these measurements are an important step in the initial evaluation.
Myasthenic crisis
Myasthenia gravis is caused by autoantibodies directed against postsynaptic acetylcholine receptors. Patients demonstrate muscle weakness, usually in a proximal pattern, with fatigue, respiratory distress, nasal speech, ophthalmoparesis, and dysphagia. Exacerbations can occur as a response to recent infection, surgery, or medications such as neuromuscular blocking agents or aminoglycosides.
Myasthenic crisis, while uncommon, is a life-threatening emergency characterized by bulbar or respiratory failure secondary to muscle weakness. It can occur in patients already diagnosed with myasthenia gravis or may be the initial manifestation of the disease.42–49 Intubation and mechanical ventilation are frequently required. Postoperative myasthenic patients in whom extubation has been delayed more than 24 hours should be considered in crisis.45
The diagnosis of myasthenia gravis can be made by serum autoantibody testing, electromyography, and nerve conduction studies (with repetitive stimulation) or administration of edrophonium in patients with obvious ptosis.
The mainstay of therapy for myasthenic crisis is either intravenous immunoglobulin at a dose of 2 g/kg over 2 to 5 days or plasmapheresis (5–7 exchanges over 7–14 days). Corticosteroids are not recommended in myasthenic crisis in patients who are not intubated, as they can potentiate an initial worsening of crisis. Once the patient begins to show clinical improvement, outpatient pyridostigmine and immunosuppressive medications can be resumed at a low dose and titrated as tolerated.
Acute inflammatory demyelinating polyneuropathy (Guillain-Barré syndrome)
Acute inflammatory demyelinating polyneuropathy is an autoimmune disorder involving autoantibodies against axons or myelin in the peripheral nervous system.
This disease should be suspected in a patient who is developing worsening muscle weakness (usually with areflexia) over the course of days to weeks. Occasionally, a recent diarrheal or other systemic infectious trigger can be identified. Blood pressure instability and cardiac arrhythmia can also be seen due to autonomic nerve involvement. Although classically described as an “ascending paralysis,” other variants of this disease have distinct clinical presentations (eg, the descending paralysis, ataxia, areflexia, ophthalmoparesis of the Miller Fisher syndrome).
Acute inflammatory demyelinating polyneuropathy is diagnosed by electromyography and nerve conduction studies. A cerebrospinal fluid profile demonstrating elevated protein and few white blood cells is typical.
Treatment, as in myasthenic crisis, involves intravenous immunoglobulin or plasmapheresis. Corticosteroids are ineffective. Anticipation of ventilatory failure and expectant intubation is essential, given the progressive nature of the disorder.50
- Pitts SR, Niska RW, Xu J, Burt CW. National hospital ambulatory medical care survey: 2006 emergency department summary. Natl Health Stat Report 2008; 7:1–38.
- McMullan JT, Knight WA, Clark JF, Beyette FR, Pancioli A. Time-critical neurological emergencies: the unfulfilled role for point-of-care testing. Int J Emerg Med 2010; 3:127–131.
- Centers for Disease Control and Prevention (CDC). Prevalence of stroke: United States, 2006–2010. MMWR Morb Mortal Wkly Rep 2012; 61:379–382.
- Lozano R, Naghavi M, Foreman K, et al. Global and regional mortality from 235 causes of death for 20 age groups in 1990 and 2010: a systematic analysis for the global burden of disease study 2010. Lancet 2012; 380:2095–2128.
- Vos T, Flaxman AD, Naghavi M, et al. Years lived with disability (YLDs) for 1,160 sequelae of 289 diseases and injuries 1990–2010: a systematic analysis for the global burden of disease study 2010. Lancet 2012; 380:2163–2196.
- Tissue plasminogen activator for acute ischemic stroke. The National Institute of Neurological Disorders and Stroke rt-PA stroke study group. N Engl J Med 1995; 333:1581–1587.
- Hacke W, Donnan G, Fieschi C, et al; ATLANTIS Trials Investigators; ECASS Trials Investigators; NINDS rt-PA Study Group Investigators. Association of outcome with early stroke treatment: pooled analysis of ATLANTIS, ECASS, and NINDS rt-PA stroke trials. Lancet 2004; 363:768–774.
- Saver JL, Fonarrow GC, Smith EE, et al. Time to treatment with intravenous tissue plasminogen activator and outcome from acute ischemic stroke. JAMA 2013; 309:2480–2488.
- Wojner-Alexander AW, Garami Z, Chernyshev OY, Alexandrov AV. Heads down: flat positioning improves blood flow velocity in acute ischemic stroke. Neurology 2005; 64:1354–1357.
- Jauch EC, Saver JL, Adams HP Jr, et al; American Heart Association Stroke Council; Council on Cardiovascular Nursing; Council on Peripheral Vascular Disease; Council on Clinical Cardiology. Guidelines for the early management of patients with acute ischemic stroke: a guideline for healthcare professionals from the American Heart Association/American Stroke Association. Stroke 2013; 44:870–947.
- Hacke W, Kaste M, Bluhmki E, et al; ECASS Investigators. Thrombolysis with alteplase 3 to 4.5 hours after acute ischemic stroke. N Engl J Med 2008; 359:1317–1329.
- Berkhemer OA, Fransen PSS, Beumer D, et al; MR CLEAN Investigators. A randomized trial of intraarterial treatment for acute ischemic stroke. N Eng J Med 2015; 372:11–20.
- Campbell BC, Mitchell PJ, Kleinig TJ, et al; EXTEND-IA Investigators. Endovascular therapy for ischemic stroke with perfusion-imaging selection. N Engl J Med 2015; 372:1009–1018.
- Backes D, Rinkel GJ, Kemperman H, Linn FH, Vergouwen MD. Time-dependent test characteristics of head computed tomography in patients suspected of nontraumatic subarachnoid hemorrhage. Stroke 2012; 43:2115–2119.
- Mendelow AD, Gregson BA, Fernandes HM, et al; STICH investigators. Early surgery versus initial conservative treatment in patients with spontaneous supratentorial intracerebral haematomas in the International Surgical Trial in Intracerebral Haemorrhage (STICH): a randomised trial. Lancet 2005; 365: 387–397.
- Anderson CS, Helley E, Huang Y, et al; INTERACT2 Investigators. Rapid blood-pressure lowering in patients with acute intracerebral hemorrhage. N Engl J Med 2013; 368:2355–2365.
- Flibotte JJ, Hagan N, O'Donnell J, Greenberg SM, Rosand J. Warfarin, hematoma expansion, and outcome of intracerebral hemorrhage. Neurology 2004; 63:1059–1064.
- Davis SM, Broderick J, Hennerici M, et al; Recombinant Activated Factor VII Intracerebral Hemorrhage Trial Investigators. Hematoma growth is a determinant of mortality and poor outcome after intracerebral hemorrhage. Neurology 2006; 66:1175–1181.
- Woo CH, Patel N, Conell C, et al. Rapid warfarin reversal in the setting of intracranial hemorrhage: a comparison of plasma, recombinant activated factor VII, and prothrombin complex concentrate. World Neurosurg 2014; 81:110–115.
- Broderick J, Connolly S, Feldmann E, et al; American Heart Association; American Stroke Association Stroke Council; High Blood Pressure Research Council; Quality of Care and Outcomes in Research Interdisciplinary Working Group. Guidelines for the management of spontaneous intracerebral hemorrhage in adults: 2007 update: a guideline from the American Heart Association/American Stroke Association Stroke Council, High Blood Pressure Research Council, and the Quality of Care and Outcomes in Research Interdisciplinary Working Group. Stroke 2007; 38:2001–2023.
- Goldstein JN, Thomas SH, Frontiero V, et al. Timing of fresh frozen plasma administration and rapid correction of coagulopathy in warfarin-related intracerebral hemorrhage. Stroke 2006, 37:151–155.
- Chapman SA, Irwin ED, Beal AL, Kulinski NM, Hutson KE, Thorson MA. Prothrombin complex concentrate versus standard therapies for INR reversal in trauma patients receiving warfarin. Ann Pharmacother 2011; 45:869–875.
- Fawole A, Daw HA, Crowther MA. Practical management of bleeding due to the anticoagulants dabigatran, rivaroxaban, and apixaban. Cleve Clin J Med 2013; 80:443–451.
- Pollack CV Jr, Reilly PA, Eikelboom J, et al. Idarucizumab for dabigatran reversal. N Engl J Med 2015; 373:511-520.
- Broderick JP, Brott TG, Duldner JE, Tomsick T, Leach A. Initial and recurrent bleeding are the major causes of death following subarachnoid hemorrhage. Stroke 1994; 25:1342–1347.
- Kassell NF, Torner JC, Haley EC Jr, Jane JA, Adams HP, Kongable GL. The international cooperative study on the timing of aneurysm surgery. Part 1: overall management results. J Neurosurg 1990; 73:18–36.
- Perry JJ, Stiell IG, Sivilotti ML, et al. Sensitivity of computed tomography performed within six hours of onset of headache for diagnosis of subarachnoid haemorrhage: prospective cohort study. BMJ 2011; 343:d4277.
- McCormack RF, Hutson A. Can computed tomography angiography of the brain replace lumbar puncture in the evaluation of acute-onset headache after a negative noncontrast cranial computed tomography scan? Acad Emerg Med 2010; 17:444–451.
- Connolly ES Jr, Rabinstein AA, Carhuapoma JR, et al; American Heart Association Stroke Council; Council on Cardiovascular Radiology and Intervention; Council on Cardiovascular Nursing; Council on Cardiovascular Surgery and Anesthesia; Council on Clinical Cardiology. Guidelines for the management of aneurysmal subarachnoid hemorrhage: a guideline for healthcare professionals from the American Heart Association/American Stroke Association. Stroke 2012; 43:1711–1737.
- Vermuelen M, Hasan D, Blijenberg BG, Hijdra A, van Gijn J. Xanthochromia after subarachnoid haemorrhage needs no revisitation. J Neurol Neurosurg Psychiatry 1989; 52:826–828.
- Molyneaux AJ, Kerr RS, Yu LM, et al; International Subarachnoid Aneurysm Trial (ISAT) Collaborative Group. International subarachnoid hemorrhage trial (ISAT) of neurosurgical clipping versus endovascular coiling in 2,143 patients with ruptured intracranial aneurysms: a randomised comparison of effects on survival, dependency, seizures, rebleeding, subgroups, and aneurysm occlusion. Lancet 2005; 366:809–817.
- Caglayan HZ, Colpak IA, Kansu T. A diagnostic challenge: dilated pupil. Curr Opin Ophthalmol 2013; 24:550–557.
- Brophy GM, Bell R, Claassen J, et al; Neurocritical Care Society Status Epilepticus Guideline Writing Committee. Guidelines for the evaluation and management of status epilepticus. Neurocrit Care 2012; 17:3–23.
- Chang CW, Bleck TP. Status epilepticus. Neurol Clin 1995; 13:529–548.
- Treiman DM. Generalized convulsive status epilepticus in the adult. Epilepsia 1993; 34(suppl 1):S2–S11.
- Leppick IE. Status epilepticus: the next decade. Neurology 1990; 40(suppl 2):4–9.
- Aranda A, Foucart G, Ducassé JL, Grolleau S, McGonigal A, Valton L. Generalized convulsive status epilepticus management in adults: a cohort study with evaluation of professional practice. Epilepsia 2010; 51:2159–2167.
- DeLorenzo RJ, Waterhouse EJ, Towne AR, et al. Persistent nonconvulsive status epilepticus after the control of convulsive status epilepticus. Epilepsia 1998; 39:833–840.
- Casha S, Christie S. A systematic review of intensive cardiopulmonary management after spinal cord injury. J Neurotrauma 2011; 28:1479–1495.
- Walters BC, Hadley MN, Hurlbert RJ, et al; American Association of Neurological Surgeons; Congress of Neurological Surgeons. Guidelines for the management of acute cervical spine and spinal cord injuries: 2013 update. Neurosurgery 2013; 60(suppl 1):82–91.
- Hurlbert RJ, Hadley MN, Walters BC, et al. Pharmacological therapy for acute spinal cord injury. Neurosurgery 2013; 72(suppl 2):93–105.
- Cohen MS, Younger D. Aspects of the natural history of myasthenia gravis: crisis and death. Ann NY Acad Sci 1981; 377:670–677.
- Belack RS, Sanders DB. On the concept of myasthenic crisis. J Clin Neuromuscul Dis 2002; 4:40–42.
- Chaudhuri A, Behan PO. Myasthenic crisis. QJM 2009; 102:97–107.
- Mayer SA. Intensive care of the myasthenic patient. Neurology 1997; 48(suppl 5):70S–75S.
- Jani-Acsadi A, Lisak RP. Myasthenic crisis: guidelines for prevention and treatment. J Neurol Sci 2007; 261:127–133.
- Bershad EM, Feen ES, Suarez JI. Myasthenia gravis crisis. South Med J 2008; 101:63–69.
- Ahmed S, Kirmani JF, Janjua N, et al. An update on myasthenic crisis. Curr Treat Options Neurol 2005; 7:129–141.
- Godoy DA, Vaz de Mello LJ, Masotti L, Napoli MD. The myasthenic patient in crisis: an update of the management in neurointensive care unit. Arq Neuropsiquiatr 2013; 71:627–639.
- Hughes RA, Wijdicks EF, Benson E, et al; Multidisciplinary Consensus Group. Supportive care for patients with Guillain-Barré syndrome: Arch Neurol 2005; 62:1194–1198.
- Pitts SR, Niska RW, Xu J, Burt CW. National hospital ambulatory medical care survey: 2006 emergency department summary. Natl Health Stat Report 2008; 7:1–38.
- McMullan JT, Knight WA, Clark JF, Beyette FR, Pancioli A. Time-critical neurological emergencies: the unfulfilled role for point-of-care testing. Int J Emerg Med 2010; 3:127–131.
- Centers for Disease Control and Prevention (CDC). Prevalence of stroke: United States, 2006–2010. MMWR Morb Mortal Wkly Rep 2012; 61:379–382.
- Lozano R, Naghavi M, Foreman K, et al. Global and regional mortality from 235 causes of death for 20 age groups in 1990 and 2010: a systematic analysis for the global burden of disease study 2010. Lancet 2012; 380:2095–2128.
- Vos T, Flaxman AD, Naghavi M, et al. Years lived with disability (YLDs) for 1,160 sequelae of 289 diseases and injuries 1990–2010: a systematic analysis for the global burden of disease study 2010. Lancet 2012; 380:2163–2196.
- Tissue plasminogen activator for acute ischemic stroke. The National Institute of Neurological Disorders and Stroke rt-PA stroke study group. N Engl J Med 1995; 333:1581–1587.
- Hacke W, Donnan G, Fieschi C, et al; ATLANTIS Trials Investigators; ECASS Trials Investigators; NINDS rt-PA Study Group Investigators. Association of outcome with early stroke treatment: pooled analysis of ATLANTIS, ECASS, and NINDS rt-PA stroke trials. Lancet 2004; 363:768–774.
- Saver JL, Fonarrow GC, Smith EE, et al. Time to treatment with intravenous tissue plasminogen activator and outcome from acute ischemic stroke. JAMA 2013; 309:2480–2488.
- Wojner-Alexander AW, Garami Z, Chernyshev OY, Alexandrov AV. Heads down: flat positioning improves blood flow velocity in acute ischemic stroke. Neurology 2005; 64:1354–1357.
- Jauch EC, Saver JL, Adams HP Jr, et al; American Heart Association Stroke Council; Council on Cardiovascular Nursing; Council on Peripheral Vascular Disease; Council on Clinical Cardiology. Guidelines for the early management of patients with acute ischemic stroke: a guideline for healthcare professionals from the American Heart Association/American Stroke Association. Stroke 2013; 44:870–947.
- Hacke W, Kaste M, Bluhmki E, et al; ECASS Investigators. Thrombolysis with alteplase 3 to 4.5 hours after acute ischemic stroke. N Engl J Med 2008; 359:1317–1329.
- Berkhemer OA, Fransen PSS, Beumer D, et al; MR CLEAN Investigators. A randomized trial of intraarterial treatment for acute ischemic stroke. N Eng J Med 2015; 372:11–20.
- Campbell BC, Mitchell PJ, Kleinig TJ, et al; EXTEND-IA Investigators. Endovascular therapy for ischemic stroke with perfusion-imaging selection. N Engl J Med 2015; 372:1009–1018.
- Backes D, Rinkel GJ, Kemperman H, Linn FH, Vergouwen MD. Time-dependent test characteristics of head computed tomography in patients suspected of nontraumatic subarachnoid hemorrhage. Stroke 2012; 43:2115–2119.
- Mendelow AD, Gregson BA, Fernandes HM, et al; STICH investigators. Early surgery versus initial conservative treatment in patients with spontaneous supratentorial intracerebral haematomas in the International Surgical Trial in Intracerebral Haemorrhage (STICH): a randomised trial. Lancet 2005; 365: 387–397.
- Anderson CS, Helley E, Huang Y, et al; INTERACT2 Investigators. Rapid blood-pressure lowering in patients with acute intracerebral hemorrhage. N Engl J Med 2013; 368:2355–2365.
- Flibotte JJ, Hagan N, O'Donnell J, Greenberg SM, Rosand J. Warfarin, hematoma expansion, and outcome of intracerebral hemorrhage. Neurology 2004; 63:1059–1064.
- Davis SM, Broderick J, Hennerici M, et al; Recombinant Activated Factor VII Intracerebral Hemorrhage Trial Investigators. Hematoma growth is a determinant of mortality and poor outcome after intracerebral hemorrhage. Neurology 2006; 66:1175–1181.
- Woo CH, Patel N, Conell C, et al. Rapid warfarin reversal in the setting of intracranial hemorrhage: a comparison of plasma, recombinant activated factor VII, and prothrombin complex concentrate. World Neurosurg 2014; 81:110–115.
- Broderick J, Connolly S, Feldmann E, et al; American Heart Association; American Stroke Association Stroke Council; High Blood Pressure Research Council; Quality of Care and Outcomes in Research Interdisciplinary Working Group. Guidelines for the management of spontaneous intracerebral hemorrhage in adults: 2007 update: a guideline from the American Heart Association/American Stroke Association Stroke Council, High Blood Pressure Research Council, and the Quality of Care and Outcomes in Research Interdisciplinary Working Group. Stroke 2007; 38:2001–2023.
- Goldstein JN, Thomas SH, Frontiero V, et al. Timing of fresh frozen plasma administration and rapid correction of coagulopathy in warfarin-related intracerebral hemorrhage. Stroke 2006, 37:151–155.
- Chapman SA, Irwin ED, Beal AL, Kulinski NM, Hutson KE, Thorson MA. Prothrombin complex concentrate versus standard therapies for INR reversal in trauma patients receiving warfarin. Ann Pharmacother 2011; 45:869–875.
- Fawole A, Daw HA, Crowther MA. Practical management of bleeding due to the anticoagulants dabigatran, rivaroxaban, and apixaban. Cleve Clin J Med 2013; 80:443–451.
- Pollack CV Jr, Reilly PA, Eikelboom J, et al. Idarucizumab for dabigatran reversal. N Engl J Med 2015; 373:511-520.
- Broderick JP, Brott TG, Duldner JE, Tomsick T, Leach A. Initial and recurrent bleeding are the major causes of death following subarachnoid hemorrhage. Stroke 1994; 25:1342–1347.
- Kassell NF, Torner JC, Haley EC Jr, Jane JA, Adams HP, Kongable GL. The international cooperative study on the timing of aneurysm surgery. Part 1: overall management results. J Neurosurg 1990; 73:18–36.
- Perry JJ, Stiell IG, Sivilotti ML, et al. Sensitivity of computed tomography performed within six hours of onset of headache for diagnosis of subarachnoid haemorrhage: prospective cohort study. BMJ 2011; 343:d4277.
- McCormack RF, Hutson A. Can computed tomography angiography of the brain replace lumbar puncture in the evaluation of acute-onset headache after a negative noncontrast cranial computed tomography scan? Acad Emerg Med 2010; 17:444–451.
- Connolly ES Jr, Rabinstein AA, Carhuapoma JR, et al; American Heart Association Stroke Council; Council on Cardiovascular Radiology and Intervention; Council on Cardiovascular Nursing; Council on Cardiovascular Surgery and Anesthesia; Council on Clinical Cardiology. Guidelines for the management of aneurysmal subarachnoid hemorrhage: a guideline for healthcare professionals from the American Heart Association/American Stroke Association. Stroke 2012; 43:1711–1737.
- Vermuelen M, Hasan D, Blijenberg BG, Hijdra A, van Gijn J. Xanthochromia after subarachnoid haemorrhage needs no revisitation. J Neurol Neurosurg Psychiatry 1989; 52:826–828.
- Molyneaux AJ, Kerr RS, Yu LM, et al; International Subarachnoid Aneurysm Trial (ISAT) Collaborative Group. International subarachnoid hemorrhage trial (ISAT) of neurosurgical clipping versus endovascular coiling in 2,143 patients with ruptured intracranial aneurysms: a randomised comparison of effects on survival, dependency, seizures, rebleeding, subgroups, and aneurysm occlusion. Lancet 2005; 366:809–817.
- Caglayan HZ, Colpak IA, Kansu T. A diagnostic challenge: dilated pupil. Curr Opin Ophthalmol 2013; 24:550–557.
- Brophy GM, Bell R, Claassen J, et al; Neurocritical Care Society Status Epilepticus Guideline Writing Committee. Guidelines for the evaluation and management of status epilepticus. Neurocrit Care 2012; 17:3–23.
- Chang CW, Bleck TP. Status epilepticus. Neurol Clin 1995; 13:529–548.
- Treiman DM. Generalized convulsive status epilepticus in the adult. Epilepsia 1993; 34(suppl 1):S2–S11.
- Leppick IE. Status epilepticus: the next decade. Neurology 1990; 40(suppl 2):4–9.
- Aranda A, Foucart G, Ducassé JL, Grolleau S, McGonigal A, Valton L. Generalized convulsive status epilepticus management in adults: a cohort study with evaluation of professional practice. Epilepsia 2010; 51:2159–2167.
- DeLorenzo RJ, Waterhouse EJ, Towne AR, et al. Persistent nonconvulsive status epilepticus after the control of convulsive status epilepticus. Epilepsia 1998; 39:833–840.
- Casha S, Christie S. A systematic review of intensive cardiopulmonary management after spinal cord injury. J Neurotrauma 2011; 28:1479–1495.
- Walters BC, Hadley MN, Hurlbert RJ, et al; American Association of Neurological Surgeons; Congress of Neurological Surgeons. Guidelines for the management of acute cervical spine and spinal cord injuries: 2013 update. Neurosurgery 2013; 60(suppl 1):82–91.
- Hurlbert RJ, Hadley MN, Walters BC, et al. Pharmacological therapy for acute spinal cord injury. Neurosurgery 2013; 72(suppl 2):93–105.
- Cohen MS, Younger D. Aspects of the natural history of myasthenia gravis: crisis and death. Ann NY Acad Sci 1981; 377:670–677.
- Belack RS, Sanders DB. On the concept of myasthenic crisis. J Clin Neuromuscul Dis 2002; 4:40–42.
- Chaudhuri A, Behan PO. Myasthenic crisis. QJM 2009; 102:97–107.
- Mayer SA. Intensive care of the myasthenic patient. Neurology 1997; 48(suppl 5):70S–75S.
- Jani-Acsadi A, Lisak RP. Myasthenic crisis: guidelines for prevention and treatment. J Neurol Sci 2007; 261:127–133.
- Bershad EM, Feen ES, Suarez JI. Myasthenia gravis crisis. South Med J 2008; 101:63–69.
- Ahmed S, Kirmani JF, Janjua N, et al. An update on myasthenic crisis. Curr Treat Options Neurol 2005; 7:129–141.
- Godoy DA, Vaz de Mello LJ, Masotti L, Napoli MD. The myasthenic patient in crisis: an update of the management in neurointensive care unit. Arq Neuropsiquiatr 2013; 71:627–639.
- Hughes RA, Wijdicks EF, Benson E, et al; Multidisciplinary Consensus Group. Supportive care for patients with Guillain-Barré syndrome: Arch Neurol 2005; 62:1194–1198.
KEY POINTS
- Patients with possible acute ischemic stroke should be assessed quickly to see if they should receive tissue plasminogen activator, which should be started within 3 hours of stroke onset. Computed tomography (CT) of the head without contrast should be done immediately to rule out acute hemorrhagic stroke.
- Acute treatment of intracerebral hemorrhage includes blood pressure control, reversal of underlying coagulopathy, and sometimes intracranial pressure control.
- If the clinical suspicion of subarachnoid hemorrhage remains strong even though initial CT was negative, lumbar puncture is mandatory.
- Hyperosmolar therapy is the mainstay of emergency medical treatment of intracranial hypertension.
- Seizure activity must be treated aggressively to prevent recalcitrant seizure activity, neuronal damage, and progression to status epilepticus.
Prescribing exercise to help your patients lose weight
Although exercise is probably less effective than diet in reducing weight, most studies show that adding it to a diet regimen will increase the weight loss.1,2 Guidelines from the American Heart Association, American College of Cardiology, and Obesity Society recommend a comprehensive lifestyle program that includes a low-calorie diet as well as an increase in physical activity.3
Here, we review the many benefits of exercise for obese patients, not only in terms of weight loss, but also its positive cardiovascular and metabolic effects. Then we discuss how to motivate and prescribe exercise for this challenging group.
EXERCISE IMPROVES WEIGHT LOSS
Increasing energy expenditure by exercising can mobilize and burn stored fat and thus lead to weight loss.4
Typically, with no changes in caloric intake, exercising 60 minutes at low intensity most days of the week will remove up to 0.5 lb per week.5 Exercising harder for longer will take off more weight, up to 3 lb per week.1,6 Some practitioners believe that the total volume of exercise (frequency multiplied by time) is more important than the intensity in determining the amount of weight loss.2,7,8
Ross et al9 randomized 101 obese men to try to lose weight by exercising at a low to moderate intensity, to try to lose weight by dieting, to exercise without the goal of losing weight, or to do nothing (the control group). About half the participants declined or dropped out, but 52 completed the trial. The weight-loss-through-exercise group had lost approximately 15 lb by 12 weeks; the diet group lost a similar amount. Total body fat, visceral fat, and abdominal obesity were all reduced with both diet- and exercise-induced weight loss.
In a study in 130 severely obese adults, after 6 months of high-intensity physical activity for a mean duration of 71 minutes per week, those on an exercise-and-diet regimen lost an average of 24 lb, compared with 18 lb with diet alone.10
Another trial involved obese patients who were instructed to jog the equivalent of 20 miles (32.2 km) a week, with no restriction on caloric intake.11 They lost only 2.9 kg (6.5 lb) over 8 months. Increased food intake explained this minimal weight loss.
In an analysis of 20 studies, exercise-only interventions of 4 months or less resulted in a mean weekly weight loss of 0.4 lb (0.2 kg), with a total loss of about 5 lb (2.3 kg).12
A systematic review of 15 studies noted that aerobic exercise for 3 months or more resulted in a significant reduction in visceral adipose tissue in overweight men and women as measured by computed tomography.13
Effects that different types of exercise have on weight loss
In a study of 119 sedentary adults who were overweight or obese and who were randomized to aerobic, resistance, or combined aerobic-resistance training over 8 months, those involved in aerobic or combined aerobic and resistance training had the greatest reduction in total body and fat mass.14 Given that the combined aerobic-resistance training program required twice the time commitment of the aerobic-alone program, the authors suggested that the most efficient manner of reducing body and fat mass is aerobic training alone.14 In contrast, if the goal is to increase lean muscle mass rather than lose weight and fat, then resistance training would be preferred.14
A meta-analysis confirmed the benefit of aerobic exercise, which resulted in significantly more loss in weight (1.2 kg, 2.6 lb), waist circumference (1.57 cm), and fat mass (1.2 kg, 2.6 lb) than resistance training.15 However, combined aerobic and resistance training was even better, with significantly more weight loss (2.0 kg, 4.4 lb) and fat mass reduction (1.9 kg, 4.2 lb).15
In summary, aerobic and combined aerobic-resistance training appear to be more effective for weight management in obese people than resistance training alone.
ADDITIONAL BENEFITS OF EXERCISE
Increasing regular physical activity through structured exercise has the additional benefits of improving physical fitness, flexibility, mobility, and cardiovascular health.16,17
Even before patients lose a significant amount of weight (eg, 10%), low-intensity exercise such as walking 30 to 60 minutes most days of the week will rapidly improve cardiorespiratory fitness and have positive effects on cardiovascular risk factors such as hypertension, elevated blood glucose, and dyslipidemia.18,19 Aerobic exercise and resistance training also reduce chronic inflammation, which is a strong indicator of future disease, especially in obese patients who have high levels of inflammatory biomarkers.20,21
Even if he or she does not lose much weight, an obese exercising person with good cardiorespiratory fitness has lower cardiovascular risk than a person who is not obese but is poorly conditioned.22
Exercise lowers blood pressure
Overactivity of the sympathetic nervous system is thought to account for over 50% of all cases of hypertension.23 Obesity in concert with diabetes is characterized by sympathetic overactivity and progressive loss of cardiac parasympathetic activity.24 Cardiac autonomic neuropathy is an underestimated risk factor for the increased cardiovascular morbidity and mortality associated with obesity and diabetes, and physical exercise may promote restoration of cardioprotective autonomic modulation in the heart.24
Several studies have shown that aerobic endurance exercise lowers blood pressure in patients with hypertension, and reduction in sympathetic neural activity has been reported as one of the main mechanisms explaining this effect.23 Another mechanism is endothelium-mediated vasodilation: even a single exercise session may increase the bioavailability of nitric oxide and decrease postexercise blood pressure.25
Different types of exercise have been shown to have different effects on blood pressure.
Aerobic training has been shown to reduce systolic blood pressure by 5.2 to 11.0 mm Hg and diastolic blood pressure by 3.0 to 7.7 mm Hg.26
The hypotensive effect of endurance aerobic training is probably mediated at least in part by a reduction in systemic vascular resistance through decreased activity of the sympathetic and renin-angiotensin systems and through improved insulin sensitivity.26 Other factors that may be involved include improved endothelium-dependent vasodilation, enhanced baroreceptor sensitivity, and arterial compliance.26
Dynamic resistance exercise has less of an effect than aerobic exercise, but it has been shown to reduce systolic blood pressure by 0.5 to 4.8 mm Hg and diastolic blood pressure by 0.5 to 4.1 mm Hg.26
In a meta-analysis of studies of resistance training lasting more than 1 month in healthy adults age 18 and older, the authors noted that resistance training induced a significant blood pressure reduction in 28 normotensive or prehypertensive study groups (–3.9/–3.9 mm Hg), whereas the reduction was not significant for the five hypertensive study groups.27
Isometric resistance exercise has been associated with small cardiovascular benefits, but has been shown to reduce systolic blood pressure by 10.5 to 16.5 mm Hg and diastolic blood pressure by 0.62 to 16.4 mm Hg.26
Exercise improves type 2 diabetes
Regular physical activity improves glycemic control and can prevent or delay the onset of type 2 diabetes mellitus.28 Furthermore, physical activity positively affects lipid levels, lowers blood pressure, reduces the rate of cardiovascular events, and restores quality of life in patients with type 2 diabetes.24,29
A meta-analysis of the effect of supervised exercise in adults with type 2 diabetes found that structured exercise achieved the following:
- Lowered systolic blood pressure by 2.42 mm Hg (95% confidence interval 0.45–4.39)
- Lowered diastolic blood pressure by 2.23 mm Hg (1.25–3.21)
- Raised the level of high-density lipoprotein cholesterol by 0.04 mmol/L (0.02–0.07)
- Lowered the level of low-density lipoprotein cholesterol by 0.16 mmol/L (0.01–0.30).30
The metabolic stress from physical exercise can increase oxidation of carbohydrates during exercise, increase postexercise consumption of oxygen (which can increase the rate of fat oxidation during recovery periods after exercise), improve glucose tolerance and insulin sensitivity, and reduce glycemia for 2 to 72 hours depending on the intensity and duration of the exercise.25
Exercise lowers the Framingham risk score
Exercise improves several of the risk factors for coronary artery disease used in calculating the Framingham risk score—ie, systolic blood pressure, total cholesterol, and high-density lipoprotein cholesterol—and thus can significantly lower this number. (It is important to remember that the Framingham score is a surrogate end point of cardiovascular risk that may correlate with a real clinical end point but does not necessarily have a guaranteed relationship.)
In a study of a 12-week exercise program in middle-aged women (ages 40–55), treadmill running for 30 minutes a day 3 days a week significantly reduced 10-year cardiovascular risk scores: 10-year risk 2.2% vs 4.3% in the nonexercising group.31 Others have also shown that enhanced levels of fitness are associated with lower 10-year Framingham risk estimates.32
A study of 31 healthy sedentary adults ages 50 to 65 who were randomized to an unsupervised but pedometer-monitored home-based walking program of 30 minutes of brisk walking 5 days a week noted significant reductions in systolic and diastolic blood pressure and stroke risk, and increased functional capacity in the walking group at 12 weeks.33 Thus, the Framingham risk scores were significantly lower in the exercising group than in with the control group.33
Given that overweight and obese patients who are starting to exercise may find jogging or running daunting, it should also be noted that three brisk 10-minute walks a day are at least as effective as one continuous 30-minute walk in reducing cardiovascular risk in previously sedentary people.34
SETTING ‘SMART’ GOALS
Because obese adults typically do not comply well with prescriptions for exercise, it is important to educate them about its benefits and to provide tools such as perceived exertion scales so they can monitor their exercise, document their performance, and chart their progress; smartphone apps can also be helpful.35 Supervised exercise may improve compliance and results.36 Initially, personal trainers are excellent for starting a habit change, but they are expensive. Virtual trainers are now available and cost far less.37
People do not become obese overnight.They gain weight over a long time. Likewise, weight reduction takes time if done in a sustainable and healthy manner. Thus, SMART goals—specific, measurable, attainable, realistic, timely—should be set to sustain the self-discipline required.
EXERCISE RECOMMENDATIONS
Any exercise program should target 30 to 60 minutes of effort per day, most days of the week, ie, 150 to 300 minutes per week or more.38 But beginners should start low and go slow to avoid dropout, musculoskeletal strain, and joint injury.
The American College of Sports Medicine (ACSM)38,39 recommends combining aerobic and progressive resistance exercise as the core components of an exercise program. The aerobic component can include anaerobic high-intensity interval training (see discussion below). In addition, we recommend flexibility and balance exercises for obese patients.40
Combining aerobic and resistance exercises likely results in greater decreases in abdominal adiposity in the obese.41 In addition, the aerobic portion of a combined exercise regimen can improve functional capacity, and the resistance portion may prevent injury by strengthening the muscles, bones, and joint support systems.42 Adding exercises that promote flexibility and balance helps with range of motion and prevents injuries while exercising.43 These exercises not only expend calories during the exercise itself, but also increase resting energy expenditure for the remainder of the day, as the effects of the raised metabolism persist for hours.44
Aerobic exercise is the foundation
Aerobic exercises that involve large muscle groups, especially walking, should be the foundation of cardiopulmonary exercise for obese persons.45 Many patients can tolerate weight-bearing exercises such as walking or bike riding, but for some, exercises with limited or no weight-bearing such as swimming or aqua-aerobics are better.46
Tips for prescribing. Patients should exercise:
- On 5 or 6 days each week
- At low to moderate intensity (30%–60% of maximum oxygen consumption [Vo2 max])
- For at least 150 minutes per week, with a long-term goal of 300 minutes per week
- By walking, riding a stationary bicycle, or swimming.38,47
To mobilize and use free fatty acids as an energy source, lower-intensity longer-duration aerobic exercise is preferred.5 Thus, frequent, low-intensity or moderate-intensity training (30%–60% of Vo2 max) of longer duration (at least 60 minutes) may be the best approach to losing body fat in obese persons.5,48 Early on in the exercise program, keep the intensity low, as high-intensity training will preferentially use stored glycogen or carbohydrate as an energy substrate rather than free fatty acids or fat.5
With light-moderate exercise, the heart rate will increase and patients will perspire, but they still should be able to carry on a conversation.
Measure (or have patients measure) the heart rate using the radial artery in their wrist after 6 minutes of walking. A pulse of 100 beats per minute or more is associated with an exercise intensity of approximately 50% (or more) of Vo2 max.5
A study of 136 obese men and women who exercised for 6 months found that those doing aerobic exercise only and those doing a combination of aerobic and resistance exercise had greater cardiopulmonary fitness, greater reductions in abdominal and visceral fat, and more improved insulin sensitivity than those doing resistance exercise only.41 Although the aerobiconly group lost more weight (6 lb) than the aerobic-plus-resistance group (5.1 lb) and the resistance-only group (1.4 lb), combining aerobic and resistance exercise is considered optimal.
All physical activity is beneficial, but activities that have less impact on the joints are less likely to cause injuries and joint pain. Aerobic activities that are especially useful in obese adults include walking at a speed of at least 2.5 miles per hour, bicycling, jogging, treadmill walking, swimming, aqua-aerobics, rowing, and low-impact aerobics classes.
Walking is the easiest way for most people to start their program, as it is safe, accessible, and relatively cheap with respect to equipment.35 Adding a simple pedometer or smartphone app to measure the amount of exercise, together with physician counseling, may improve compliance and thus weight loss.49,50
Obese patients may have been inactive for quite a while. Therefore, the sessions should be short and low-intensity at first, then steadily progress.51 To minimize dropout, avoid hard exercise too soon for people with a low exercise capacity or high body mass index at baseline, and give positive feedback and encouragement at each visit.52
It is reasonable to introduce other aerobic exercises to vary the routine, use other muscle groups, and reduce the chance of injury from overuse of one muscle or joint group. Then, as cardiorespiratory fitness improves, the patient will be more confident about trying activities that are more challenging, such as jogging and aerobics classes. An aerobic exercise program consisting only of swimming is less efficacious for weight loss in this population.53
High-intensity interval training
High-intensity interval training involves relatively brief bursts of vigorous exercise separated by periods of recovery and is a time-efficient, novel alternative to continuous exercise.54 The exercise component is anaerobic, meaning muscle movement that does not require oxygen. Anaerobic exercise uses fast-twitch muscle fibers, and thus helps that musculature to become stronger, larger, and more toned. Evidence suggests that high-intensity interval training induces health-enhancing adaptations similar to those of continuous exercise, despite a substantially lower time commitment.41
The ACSM recommends that most adults engage in moderate-intensity cardiorespiratory exercise training for at least 30 minutes a day on at least 5 days a week for a total of at least 150 minutes per week, or high-intensity cardiorespiratory exercise training for at least 20 minutes a day on at least 3 days a week for a goal of 75 minutes a week.38 Thus, high-intensity interval training may be attractive for obese patients because it entails a shorter time commitment to achieve similar weight loss and improved insulin sensitivity than low-intensity or moderate-intensity continuous exercise.
High-intensity exercise has been shown to be effective for obese patients if they can do it.54–56 In one study,57 134 obese patients, mean age 53, underwent supervised high-intensity interval training with resistance training two or three times a week, were encouraged to perform one or two additional exercise sessions a week (unsupervised), and were counseled to follow a Mediterranean diet. At 9 months, investigators noted a significant reduction in body mass, waist circumference, and fat mass.
A study of 12 weeks of high-intensity interval training, moderate-intensity interval training, or no exercise in 34 obese adolescent girls noted that body mass and percentage body fat were significantly decreased with both interval training regimens. However, the high-intensity group had greater reductions in waist circumference and more significant improvements in blood lipid levels, adiponectin levels, and insulin sensitivity.58
Of 62 overweight and obese patients (mean age 53.3, mean body mass index 35.8 kg/m2), 97% adhered to a program of high-intensity interval training over 9 months, which resulted in an average weekly energy expenditure of 1,582 kcal.55 Clinically and statistically significant improvements occurred in body mass (–5.3 kg), body mass index (–1.9 kg/m2), and waist circumference (–5.8 cm) (P < .0001 for all variables). Total fat mass, trunk fat mass, and lipid levels also significantly improved (P < .0001), and the prevalence of metabolic syndrome was reduced by 32.5% (P < .05).
In a meta-analysis of the effect of exercise on overweight adults, training of moderate or high intensity was noted to have the highest potential to reduce visceral adipose tissue in overweight men and women.13 Another meta-analysis noted that high-intensity interval training appeared to promote more improvement in fitness and similar improvements in some cardiometabolic risk factors than moderate exercise performed for at least 8 to 12 weeks in overweight patients.56
A typical progressive exercise program for obese adults is shown in Table 1.
Progressive resistance exercise
Progressive resistance exercises are generally easier for obese patients, as they are not aerobically challenging, allow patients to exercise around physically active people who thus motivate them, and encourage positive feelings about completing their exercise sets.59 The result is improved muscular fitness, socialization, and increased confidence in their abilities (self-efficacy).59
Progressive resistance exercises also promote favorable energy balance and reduced visceral fat deposition through enhanced basal metabolism and activity levels while counteracting age- and disease-related muscle wasting.59 They have been shown to improve cognitive ability, self-esteem, movement control, muscle mass, strength, glucose control, insulin sensitivity, resting blood pressure, lipid profile, and bone mineral density and to reduce fat weight, low back pain, arthritic discomfort, insomnia, anxiety, and depression.60
Gym neophytes should spend a few sessions with a personal trainer to learn how to use the equipment.
While the primary goal of resistance training is more muscle strength, it can reduce fat and weight, burning up to 170 kcal in a 20-minute intense exercise session.61 It reduces both total body fat and visceral adipose tissue, thus benefiting obese persons by reducing insulin resistance.62 All exercise, and especially resistance exercise, can help to strengthen the musculoskeletal system, reduce muscle atrophy, and improve bone mineral density.63
The ACSM guidelines38 recommend progressive resistance exercise on 2 or 3 nonconsecutive days a week. It should involve:
- Exercises that work 8 to 10 muscle groups per session
- Two to four sets of 8 to 12 repetitions for each muscle group.
Exercising on nonconsecutive days allows time for the complete cycle of muscle tissue remodeling.64 Such self-regulated intensity reduces the likelihood of excessive delayed-onset muscle soreness, which can discourage new participants.65
To prevent muscle injury, obese people should begin with low-intensity workouts using lower resistance, one set of 8 to 12 repetitions 2 days a week. Then, they should gradually but progressively increase the intensity, volume, and frequency of the training.47 This will obviate a plateau in training and will maximize musculoskeletal adaptation. The prescription should include exercises for the upper body (eg, biceps curls), lower body (eg, leg presses), and the midsection (eg, abdominal curl-ups, which give better abdominal muscle engagement and less risk to the back than crunches) and focus on the correct exercise form and function rather than the amount of resistance or weight lifted.
A typical progressive resistance exercise program for obese adults is shown in Table 2.
Flexibility exercise
Flexibility exercise involves stretching to improve the movement of muscles, joints, and ligaments.45 While not specifically used in an energy-expenditure strategy, flexibility (or mobility) exercises help to increase or maintain joint range of motion and can reduce muscle and joint pain associated with obesity and exercise.66
The ACSM recommends that stretching exercises be done when the muscles are warm after a brief warm-up or exercise session.38 Typically, muscles should be stretched for at least 15 seconds, and stretching is recommended at a frequency of 2 to 4 days per week.38
A good way to incorporate flexibility exercise is to join a yoga class, as yoga has been shown to improve strength and flexibility and may help control physiologic variables such as blood pressure, lipids, respiration, heart rate, and metabolic rate to improve overall exercise capacity in obese patients.67
Balance exercise
Balance exercises help obese patients improve their stability. Poor balance is associated with injuries, accidents, and falls during activities of daily living.68
Balance, the ability to maintain the body’s center of gravity within its base of support, can be categorized as static (sustaining the body in static equilibrium or within its base of support) or dynamic (maintaining equilibrium during a transition from a dynamic to a static state), which is more challenging.69 Doing both static and dynamic balance training maximizes balance and stability.69 While most activities that involve moving the body or body parts (such as walking) will improve balance, some additional balance exercises can be beneficial.
Balance exercises can be done without any equipment. Examples are balancing on one foot for 15 seconds and standing up and sitting down without using the hands. However, specific equipment can help, including physioballs, stability balls, cut-in-half stability balls, balance discs, balance wedges, wobble boards, rocker boards, and Indo boards.70 In fact, balance boards and stability balls engage more muscle fibers in other areas of the body (lower back, lower abs, quads, hamstrings, and calves) than exercises done without those balancing devices.71
Balance training for at least 10 minutes a day, 3 days a week, for 4 weeks that incorporates various methods of balance training appears to improve balance.56 Obese patients commencing a program should start with static balance exercises and then progress to dynamic ones. In addition, as balance training progresses, obese patients can integrate balance and stability training exercises with other pieces of equipment, such as performing squats on a balance board, and then gradually add weights (eg, dumbbells) to the exercise.
An example of a weekly comprehensive exercise program for an obese patient that incorporates all major exercise types is provided in Table 3. In addition, some smartphone apps that are especially helpful in overweight newcomers to exercise include Couch-to-5K, GymGoal 2, Moves, Fitbit, Workout Trainer, Endomondo, MapMyFitness, Fitocracy, and Fitness Buddy.
BARIATRIC SURGERY AND LIFESTYLE MANAGEMENT FOR OBESITY
Bariatric surgery is a safe and effective treatment for severe obesity and comorbidities including type 2 diabetes mellitus, but weight loss and health outcomes vary considerably among individuals.72,73 Of importance, postoperative weight loss after bariatric surgery and long-term weight loss largely depend on the extent to which patients can make and sustain changes to their lifestyle, including diet, exercise, and behavior modification.72,74
Exercise, especially supervised, is associated with more weight loss after bariatric surgery.61 In a meta-analysis of bariatric patients, exercise participants involved in moderate or greater levels of exercise lost a mean of 3.6 kg more than the minimal exercise groups.75 Another meta-analysis noted the beneficial effects of exercise incorporating more than 30 minutes a day of moderate physical activity following bariatric surgery and was associated with a greater weight loss of over 4% of body mass index.76 These findings were consistent with those of yet another meta-analysis.77
In summary, exercise appears to significantly increase weight loss after bariatric surgery.
TREATMENT CONSIDERATIONS IN MORBID OBESITY
Challenges faced by severely obese or morbidly obese patients affect their exercise options. The types of exercise they are able to perform are limited in most cases to very-low-impact, low-intensity exercises, which may not be as efficient in weight loss or weight maintenance.48 Therefore, it may be prudent to set more conservative weight-loss goals for them, especially early in the program. Compliance and success rates may be better with low-impact activities such as walking, water aerobics, stationary cycling, and resistance training in the severely obese population.
The more severe the obesity, the more comorbidities such as diabetes, hypertension, hyperlipidemia, arthritis, sleep apnea, gastroesophageal reflux disease, and the greater the risk of metabolic syndrome—and conversely, the greater the potential benefit from bariatric surgery followed by exercise.74
A LONG-TERM ENDEAVOR
For obese patients, a comprehensive exercise program will improve functional status, favorably influence cardiovascular risk factors, and help with weight loss or weight maintenance.
Managing obesity is a long-term endeavor.78 For it to succeed, both the patient and the physician need to keep up their efforts. To keep the patient from becoming discouraged, the clinician should focus not just on weight, but also on improvements in metabolic profile and cardiorespiratory fitness. In addition, a careful evaluation, a clear exercise prescription, defined goals, ongoing monitoring (by the patient and the provider), frequent feedback, and charting of progress will improve daily performance and the chance of long-term success.
- Thorogood A, Mottillo S, Shimony A, et al. Isolated aerobic exercise and weight loss: a systematic review and meta-analysis of randomized controlled trials. Am J Med 2011; 124:747–755.
- Church T. Exercise in obesity, metabolic syndrome, and diabetes. Prog Cardiovasc Dis 2011; 53:412–418.
- Jensen MD, Ryan DH, Apovian CM, et al; American College of Cardiology/American Heart Association Task Force on Practice Guidelines; Obesity Society. 2013 AHA/ACC/TOS guideline for the management of overweight and obesity in adults: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines and The Obesity Society. Circulation 2014; 129(suppl 2):S102–S138.
- Strasser B. Physical activity in obesity and metabolic syndrome. Ann N Y Acad Sci 2013; 1281:141–159.
- Poirier P, Despres JP. Exercise in weight management of obesity. Cardiol Clin 2001; 19:459–470.
- Shaw K, Gennat H, O’Rourke P, Del Mar C. Exercise for overweight or obesity. Cochrane Database Syst Rev 2006; 4:CD003817.
- Slentz CA, Houmard JA, Kraus WE. Exercise, abdominal obesity, skeletal muscle, and metabolic risk: evidence for a dose response. Obesity (Silver Spring) 2009; 17(suppl 3):S27–S33.
- Ross R, Hudson R, Stotz PJ, Lam M. Effects of exercise amount and intensity on abdominal obesity and glucose tolerance in obese adults: a randomized trial. Ann Intern Med 2015; 162:325–334.
- Ross R, Dagnone D, Jones PJ, et al. Reduction in obesity and related comorbid conditions after diet-induced weight loss or exercise-induced weight loss in men. A randomized, controlled trial. Ann Intern Med 2000; 133:92–103.
- Goodpaster BH, Delany JP, Otto AD, et al. Effects of diet and physical activity interventions on weight loss and cardiometabolic risk factors in severely obese adults: a randomized trial. JAMA 2010; 304:1795–1802.
- Slentz CA, Duscha BD, Johnson JL, et al. Effects of the amount of exercise on body weight, body composition, and measures of central obesity: STRRIDE—a randomized controlled study. Arch Intern Med 2004; 164:31–39.
- Ross R, Janssen I. Physical activity, total and regional obesity: dose-response considerations. Med Sci Sports Exerc 2001; 33(suppl 6):S521–S529.
- Vissers D, Hens W, Taeymans J, Baeyens JP, Poortmans J, Van Gaal L. The effect of exercise on visceral adipose tissue in overweight adults: a systematic review and meta-analysis. PLoS One 2013; 8:e56415.
- Willis LH, Slentz CA, Bateman LA, et al. Effects of aerobic and/or resistance training on body mass and fat mass in overweight or obese adults. J Appl Physiol (1985) 2012; 113:1831–1837.
- Schwingshackl L, Dias S, Strasser B, Hoffmann G. Impact of different training modalities on anthropometric and metabolic characteristics in overweight/obese subjects: a systematic review and network meta-analysis. PLoS One 2013; 8:e82853.
- Cook CM, Schoeller DA. Physical activity and weight control: conflicting findings. Curr Opin Clin Nutr Metab Care 2011; 14:419–424.
- Choo J, Lee J, Cho JH, Burke LE, Sekikawa A, Jae SY. Effects of weight management by exercise modes on markers of subclinical atherosclerosis and cardiometabolic profile among women with abdominal obesity: a randomized controlled trial. BMC Cardiovasc Disord 2014; 14:82.
- Carroll S, Dudfield M. What is the relationship between exercise and metabolic abnormalities? A review of the metabolic syndrome. Sports Med 2004; 34:371–418.
- Shaibi GQ, Ryder JR, Kim JY, Barraza E. Exercise for obese youth: refocusing attention from weight loss to health gains. Exerc Sport Sci Rev 2015; 43:41–47.
- You T, Arsenis NC, Disanzo BL, Lamonte MJ. Effects of exercise training on chronic inflammation in obesity: current evidence and potential mechanisms. Sports Med 2013; 43:243–256.
- Bluher S, Petroff D, Wagner A, et al. The one year exercise and lifestyle intervention program KLAKS: effects on anthropometric parameters, cardiometabolic risk factors and glycemic control in childhood obesity. Metabolism 2014; 63:422–430.
- Lee CD, Blair SN, Jackson AS. Cardiorespiratory fitness, body composition, and all-cause and cardiovascular disease mortality in men. Am J Clin Nutr 1999; 69:373–380.
- Leosco D, Parisi V, Femminella GD, et al. Effects of exercise training on cardiovascular adrenergic system. Front Physiol 2013; 4:348.
- Voulgari C, Pagoni S, Vinik A, Poirier P. Exercise improves cardiac autonomic function in obesity and diabetes. Metabolism 2013; 62:609–621.
- Asano RY, Sales MM, Browne RA, et al. Acute effects of physical exercise in type 2 diabetes: a review. World J Diabetes 2014; 5:659–665.
- Brook RD, Appel LJ, Rubenfire M, et al; American Heart Association Professional Education Committee of the Council for High Blood Pressure Research, Council on Cardiovascular and Stroke Nursing, Council on Epidemiology and Prevention, and Council on Nutrition, Physical Activity. Beyond medications and diet: alternative approaches to lowering blood pressure: a scientific statement from the American Heart Association. Hypertension 2013; 61:1360–1383.
- Cornelissen VA, Fagard RH, Coeckelberghs E, Vanhees L. Impact of resistance training on blood pressure and other cardiovascular risk factors: a meta-analysis of randomized, controlled trials. Hypertension 2011; 58:950–958.
- Ades PA. A lifestyle program of exercise and weight loss is effective in preventing and treating type 2 diabetes mellitus: why are programs not more available? Prev Med 2015: S0091–7435(15)00085–7.
- Ades PA, Savage PD, Marney AM, Harvey J, Evans KA. Remission of recently diagnosed type 2 diabetes mellitus with weight loss and exercise. J Cardiopulm Rehabil Prev 2015; 35:193–197.
- Hayashino Y, Jackson JL, Fukumori N, Nakamura F, Fukuhara S. Effects of supervised exercise on lipid profiles and blood pressure control in people with type 2 diabetes mellitus: a meta-analysis of randomized controlled trials. Diabetes Res Clin Pract 2012; 98:349–360.
- Saffi MA, Polanczyk CA, Rabelo-Silva ER. Lifestyle interventions reduce cardiovascular risk in patients with coronary artery disease: a randomized clinical trial. Eur J Cardiovasc Nurs 2014; 13:436–443.
- LaMonte MJ, Durstine JL, Addy CL, Irwin ML, Ainsworth BE. Physical activity, physical fitness, and Framingham 10-year risk score: the cross-cultural activity participation study. J Cardiopulm Rehabil 2001; 21:63–70.
- Tully MA, Cupples ME, Chan WS, McGlade K, Young IS. Brisk walking, fitness, and cardiovascular risk: a randomized controlled trial in primary care. Prev Med 2005; 41:622–628.
- Murphy M, Nevill A, Neville C, Biddle S, Hardman A. Accumulating brisk walking for fitness, cardiovascular risk, and psychological health. Med Sci Sports Exerc 2002; 34:1468–1474.
- Colley RC, Hills AP, King NA, Byrne NM. Exercise-induced energy expenditure: implications for exercise prescription and obesity. Patient Educ Couns 2010; 79:327–332.
- Baillot A, Mampuya WM, Comeau E, Méziat-Burdin A, Langlois MF. Feasibility and impacts of supervised exercise training in subjects with obesity awaiting bariatric surgery: a pilot study. Obes Surg 2013; 23:882–891.
- Lowe S, ÓLaighin G. The age of the virtual trainer. Procedia Engineering 2012; 34:242–247.
- Garber CE, Blissmer B, Deschenes MR, et al; American College of Sports Medicine. American College of Sports Medicine position stand. Quantity and quality of exercise for developing and maintaining cardiorespiratory, musculoskeletal, and neuromotor fitness in apparently healthy adults: guidance for prescribing exercise. Med Sci Sports Exerc 2011; 43:1334–1359.
- Donnelly JE, Blair SN, Jakicic JM, Manore MM, Rankin JW, Smith BK; American College of Sports Medicine. American College of Sports Medicine position stand. Appropriate physical activity intervention strategies for weight loss and prevention of weight regain for adults. Med Sci Sports Exerc 2009; 41:459–471.
- Montero-Fernandez N, Serra-Rexach JA. Role of exercise on sarcopenia in the elderly. Eur J Phys Rehabil Med 2013; 49:131–143.
- Davidson LE, Hudson R, Kilpatrick K, et al. Effects of exercise modality on insulin resistance and functional limitation in older adults: a randomized controlled trial. Arch Intern Med 2009; 169:122–131.
- Liu CJ, Latham NK. Progressive resistance strength training for improving physical function in older adults. Cochrane Database Syst Rev 2009; 3:CD002759.
- Manini TM, Newman AB, Fielding R, et al; LIFE Research Group. Effects of exercise on mobility in obese and nonobese older adults. Obesity (Silver Spring) 2010; 18:1168–1175.
- Hackney KJ, Engels HJ, Gretebeck RJ. Resting energy expenditure and delayed-onset muscle soreness after full-body resistance training with an eccentric concentration. J Strength Cond Res 2008; 22:1602–1609.
- Siddiqui NI, Nessa A, Hossain MA. Regular physical exercise: way to healthy life. Mymensingh Med J 2010; 19:154–158.
- Chicco AJ. Exercise training in prevention and rehabilitation: which training mode is best? Minerva Cardioangiol 2008; 56:557–570.
- Westcott WL, Winett RA, Annesi JJ, Wojcik JR, Anderson ES, Madden PJ. Prescribing physical activity: applying the ACSM protocols for exercise type, intensity, and duration across 3 training frequencies. Phys Sportsmed 2009; 37:51–58.
- Mougios V1, Kazaki M, Christoulas K, Ziogas G, Petridou A. Does the intensity of an exercise programme modulate body composition changes? Int J Sports Med 2006; 27:178–181.
- Richardson CR, Newton TL, Abraham JJ, Sen A, Jimbo M, Swartz AM. A meta-analysis of pedometer-based walking interventions and weight loss. Ann Fam Med 2008; 6:69–77.
- Stovitz SD, VanWormer JJ, Center BA, Bremer KL. Pedometers as a means to increase ambulatory activity for patients seen at a family medicine clinic. J Am Board Fam Pract 2005; 18:335–343.
- Lepor NE, Fouchia DD, McCullough PA. New vistas for the treatment of obesity: turning the tide against the leading cause of morbidity and cardiovascular mortality in the developed world. Rev Cardiovasc Med 2013; 14:20–40.
- Wittmer M, Volpatti M, Piazzalonga S, Hoffmann A. Expectation, satisfaction, and predictors of dropout in cardiac rehabilitation. Eur J Prev Cardiol 2012; 19:1082–1088.
- Gwinup G. Weight loss without dietary restriction: efficacy of different forms of aerobic exercise. Am J Sports Med 1987; 15:275–279.
- Jung ME, Bourne JE, Little JP. Where does HIT fit? An examination of the affective response to high-intensity intervals in comparison to continuous moderate- and continuous vigorous-intensity exercise in the exercise intensity-affect continuum. PLoS One 2014; 9:e114541.
- Gremeaux V, Drigny J, Nigam A, et al. Long-term lifestyle intervention with optimized high-intensity interval training improves body composition, cardiometabolic risk, and exercise parameters in patients with abdominal obesity. Am J Phys Med Rehabil 2012; 91:941–950.
- Kessler HS, Sisson SB, Short KR. The potential for high-intensity interval training to reduce cardiometabolic disease risk. Sports Med 2012; 42:489–509.
- Dalzill C, Nigam A, Juneau M, et al. Intensive lifestyle intervention improves cardiometabolic and exercise parameters in metabolically healthy obese and metabolically unhealthy obese individuals. Can J Cardiol 2014; 30:434–440.
- Racil G, Ben Ounis O, Hammouda O, et al. Effects of high vs moderate exercise intensity during interval training on lipids and adiponectin levels in obese young females. Eur J Appl Physiol 2013; 113:2531–2540.
- Willey KA, Singh MA. Battling insulin resistance in elderly obese people with type 2 diabetes: bring on the heavy weights. Diabetes Care 2003; 26:1580–1588.
- Westcott WL. Resistance training is medicine: effects of strength training on health. Curr Sports Med Rep 2012; 11:209–216.
- Haltom RW, Kraemer RR, Sloan RA, Hebert EP, Frank K, Tryniecki JL. Circuit weight training and its effects on excess postexercise oxygen consumption. Med Sci Sports Exerc 1999; 31:1613–1618.
- Strasser B, Schobersberger W. Evidence for resistance training as a treatment therapy in obesity. J Obes 2011; pii:482564.
- Fonseca H, Moreira-Gonçalves D, Coriolano HJ, Duarte JA. Bone quality: the determinants of bone strength and fragility. Sports Med 2014; 44:37–53.
- Candow DG, Burke DG. Effect of short-term equal-volume resistance training with different workout frequency on muscle mass and strength in untrained men and women. J Strength Cond Res 2007; 21:204–207.
- Trost Z, France CR, Thomas JS. Pain-related fear and avoidance of physical exertion following delayed-onset muscle soreness. Pain 2011; 152:1540–1547.
- Mathus-Vliegen EM. Obesity and the elderly. J Clin Gastroenterol 2012; 46:533–544.
- Dhananjai S, Sadashiv, Tiwari S, Dutt K, Kumar R. Reducing psychological distress and obesity through yoga practice. Int J Yoga 2013; 6:66–70.
- Mathus-Vliegen EM; Obesity Management Task Force of the European Association for the Study of Obesity. Prevalence, pathophysiology, health consequences and treatment options of obesity in the elderly: a guideline. Obes Facts 2012; 5:460–483.
- DiStefano LJ, Clark MA, Padua DA. Evidence supporting balance training in healthy individuals: a systemic review. J Strength Cond Res 2009; 23:2718–2731.
- Ogaya S, Ikezoe T, Soda N, Ichihashi N. Effects of balance training using wobble boards in the elderly. J Strength Cond Res 2011; 25:2616–2622.
- Sukalinggam CL, Sukalinggam GL, Kasim F, Yusof A. Stability ball training on lower back strength has greater effect in untrained female compared to male. J Hum Kinet 2012; 33:133–141.
- Kalarchian M, Turk M, Elliott J, Gourash W. Lifestyle management for enhancing outcomes after bariatric surgery. Curr Diab Rep 2014; 14:540.
- Rothwell L, Kow L, Toouli J. Effect of a post-operative structured exercise programme on short-term weight loss after obesity surgery using adjustable gastric bands. Obes Surg 2015; 25:126–128.
- Mechanick JI, Youdim A, Jones DB, et al. Clinical practice guidelines for the perioperative nutritional, metabolic, and nonsurgical support of the bariatric surgery patient—2013 update: cosponsored by American Association of Clinical Endocrinologists, the Obesity Society, and American Society for Metabolic & Bariatric Surgery. Surg Obes Relat Dis 2013; 9:159–191.
- Egberts K, Brown WA, Brennan L, O’Brien PE. Does exercise improve weight loss after bariatric surgery? A systematic review. Obes Surg 2012; 22:335–341.
- Livhits M, Mercado C, Yermilov I, et al. Exercise following bariatric surgery: systematic review. Obes Surg 2010; 20:657–665.
- Jacobi D, Ciangura C, Couet C, Oppert JM. Physical activity and weight loss following bariatric surgery. Obes Rev 2011; 12:366–377.
- Wadden TA, Foster GD, Letizia KA. One-year behavioral treatment of obesity: comparison of moderate and severe caloric restriction and the effects of weight maintenance therapy. J Consult Clin Psychol 1994; 62:165–171.
Although exercise is probably less effective than diet in reducing weight, most studies show that adding it to a diet regimen will increase the weight loss.1,2 Guidelines from the American Heart Association, American College of Cardiology, and Obesity Society recommend a comprehensive lifestyle program that includes a low-calorie diet as well as an increase in physical activity.3
Here, we review the many benefits of exercise for obese patients, not only in terms of weight loss, but also its positive cardiovascular and metabolic effects. Then we discuss how to motivate and prescribe exercise for this challenging group.
EXERCISE IMPROVES WEIGHT LOSS
Increasing energy expenditure by exercising can mobilize and burn stored fat and thus lead to weight loss.4
Typically, with no changes in caloric intake, exercising 60 minutes at low intensity most days of the week will remove up to 0.5 lb per week.5 Exercising harder for longer will take off more weight, up to 3 lb per week.1,6 Some practitioners believe that the total volume of exercise (frequency multiplied by time) is more important than the intensity in determining the amount of weight loss.2,7,8
Ross et al9 randomized 101 obese men to try to lose weight by exercising at a low to moderate intensity, to try to lose weight by dieting, to exercise without the goal of losing weight, or to do nothing (the control group). About half the participants declined or dropped out, but 52 completed the trial. The weight-loss-through-exercise group had lost approximately 15 lb by 12 weeks; the diet group lost a similar amount. Total body fat, visceral fat, and abdominal obesity were all reduced with both diet- and exercise-induced weight loss.
In a study in 130 severely obese adults, after 6 months of high-intensity physical activity for a mean duration of 71 minutes per week, those on an exercise-and-diet regimen lost an average of 24 lb, compared with 18 lb with diet alone.10
Another trial involved obese patients who were instructed to jog the equivalent of 20 miles (32.2 km) a week, with no restriction on caloric intake.11 They lost only 2.9 kg (6.5 lb) over 8 months. Increased food intake explained this minimal weight loss.
In an analysis of 20 studies, exercise-only interventions of 4 months or less resulted in a mean weekly weight loss of 0.4 lb (0.2 kg), with a total loss of about 5 lb (2.3 kg).12
A systematic review of 15 studies noted that aerobic exercise for 3 months or more resulted in a significant reduction in visceral adipose tissue in overweight men and women as measured by computed tomography.13
Effects that different types of exercise have on weight loss
In a study of 119 sedentary adults who were overweight or obese and who were randomized to aerobic, resistance, or combined aerobic-resistance training over 8 months, those involved in aerobic or combined aerobic and resistance training had the greatest reduction in total body and fat mass.14 Given that the combined aerobic-resistance training program required twice the time commitment of the aerobic-alone program, the authors suggested that the most efficient manner of reducing body and fat mass is aerobic training alone.14 In contrast, if the goal is to increase lean muscle mass rather than lose weight and fat, then resistance training would be preferred.14
A meta-analysis confirmed the benefit of aerobic exercise, which resulted in significantly more loss in weight (1.2 kg, 2.6 lb), waist circumference (1.57 cm), and fat mass (1.2 kg, 2.6 lb) than resistance training.15 However, combined aerobic and resistance training was even better, with significantly more weight loss (2.0 kg, 4.4 lb) and fat mass reduction (1.9 kg, 4.2 lb).15
In summary, aerobic and combined aerobic-resistance training appear to be more effective for weight management in obese people than resistance training alone.
ADDITIONAL BENEFITS OF EXERCISE
Increasing regular physical activity through structured exercise has the additional benefits of improving physical fitness, flexibility, mobility, and cardiovascular health.16,17
Even before patients lose a significant amount of weight (eg, 10%), low-intensity exercise such as walking 30 to 60 minutes most days of the week will rapidly improve cardiorespiratory fitness and have positive effects on cardiovascular risk factors such as hypertension, elevated blood glucose, and dyslipidemia.18,19 Aerobic exercise and resistance training also reduce chronic inflammation, which is a strong indicator of future disease, especially in obese patients who have high levels of inflammatory biomarkers.20,21
Even if he or she does not lose much weight, an obese exercising person with good cardiorespiratory fitness has lower cardiovascular risk than a person who is not obese but is poorly conditioned.22
Exercise lowers blood pressure
Overactivity of the sympathetic nervous system is thought to account for over 50% of all cases of hypertension.23 Obesity in concert with diabetes is characterized by sympathetic overactivity and progressive loss of cardiac parasympathetic activity.24 Cardiac autonomic neuropathy is an underestimated risk factor for the increased cardiovascular morbidity and mortality associated with obesity and diabetes, and physical exercise may promote restoration of cardioprotective autonomic modulation in the heart.24
Several studies have shown that aerobic endurance exercise lowers blood pressure in patients with hypertension, and reduction in sympathetic neural activity has been reported as one of the main mechanisms explaining this effect.23 Another mechanism is endothelium-mediated vasodilation: even a single exercise session may increase the bioavailability of nitric oxide and decrease postexercise blood pressure.25
Different types of exercise have been shown to have different effects on blood pressure.
Aerobic training has been shown to reduce systolic blood pressure by 5.2 to 11.0 mm Hg and diastolic blood pressure by 3.0 to 7.7 mm Hg.26
The hypotensive effect of endurance aerobic training is probably mediated at least in part by a reduction in systemic vascular resistance through decreased activity of the sympathetic and renin-angiotensin systems and through improved insulin sensitivity.26 Other factors that may be involved include improved endothelium-dependent vasodilation, enhanced baroreceptor sensitivity, and arterial compliance.26
Dynamic resistance exercise has less of an effect than aerobic exercise, but it has been shown to reduce systolic blood pressure by 0.5 to 4.8 mm Hg and diastolic blood pressure by 0.5 to 4.1 mm Hg.26
In a meta-analysis of studies of resistance training lasting more than 1 month in healthy adults age 18 and older, the authors noted that resistance training induced a significant blood pressure reduction in 28 normotensive or prehypertensive study groups (–3.9/–3.9 mm Hg), whereas the reduction was not significant for the five hypertensive study groups.27
Isometric resistance exercise has been associated with small cardiovascular benefits, but has been shown to reduce systolic blood pressure by 10.5 to 16.5 mm Hg and diastolic blood pressure by 0.62 to 16.4 mm Hg.26
Exercise improves type 2 diabetes
Regular physical activity improves glycemic control and can prevent or delay the onset of type 2 diabetes mellitus.28 Furthermore, physical activity positively affects lipid levels, lowers blood pressure, reduces the rate of cardiovascular events, and restores quality of life in patients with type 2 diabetes.24,29
A meta-analysis of the effect of supervised exercise in adults with type 2 diabetes found that structured exercise achieved the following:
- Lowered systolic blood pressure by 2.42 mm Hg (95% confidence interval 0.45–4.39)
- Lowered diastolic blood pressure by 2.23 mm Hg (1.25–3.21)
- Raised the level of high-density lipoprotein cholesterol by 0.04 mmol/L (0.02–0.07)
- Lowered the level of low-density lipoprotein cholesterol by 0.16 mmol/L (0.01–0.30).30
The metabolic stress from physical exercise can increase oxidation of carbohydrates during exercise, increase postexercise consumption of oxygen (which can increase the rate of fat oxidation during recovery periods after exercise), improve glucose tolerance and insulin sensitivity, and reduce glycemia for 2 to 72 hours depending on the intensity and duration of the exercise.25
Exercise lowers the Framingham risk score
Exercise improves several of the risk factors for coronary artery disease used in calculating the Framingham risk score—ie, systolic blood pressure, total cholesterol, and high-density lipoprotein cholesterol—and thus can significantly lower this number. (It is important to remember that the Framingham score is a surrogate end point of cardiovascular risk that may correlate with a real clinical end point but does not necessarily have a guaranteed relationship.)
In a study of a 12-week exercise program in middle-aged women (ages 40–55), treadmill running for 30 minutes a day 3 days a week significantly reduced 10-year cardiovascular risk scores: 10-year risk 2.2% vs 4.3% in the nonexercising group.31 Others have also shown that enhanced levels of fitness are associated with lower 10-year Framingham risk estimates.32
A study of 31 healthy sedentary adults ages 50 to 65 who were randomized to an unsupervised but pedometer-monitored home-based walking program of 30 minutes of brisk walking 5 days a week noted significant reductions in systolic and diastolic blood pressure and stroke risk, and increased functional capacity in the walking group at 12 weeks.33 Thus, the Framingham risk scores were significantly lower in the exercising group than in with the control group.33
Given that overweight and obese patients who are starting to exercise may find jogging or running daunting, it should also be noted that three brisk 10-minute walks a day are at least as effective as one continuous 30-minute walk in reducing cardiovascular risk in previously sedentary people.34
SETTING ‘SMART’ GOALS
Because obese adults typically do not comply well with prescriptions for exercise, it is important to educate them about its benefits and to provide tools such as perceived exertion scales so they can monitor their exercise, document their performance, and chart their progress; smartphone apps can also be helpful.35 Supervised exercise may improve compliance and results.36 Initially, personal trainers are excellent for starting a habit change, but they are expensive. Virtual trainers are now available and cost far less.37
People do not become obese overnight.They gain weight over a long time. Likewise, weight reduction takes time if done in a sustainable and healthy manner. Thus, SMART goals—specific, measurable, attainable, realistic, timely—should be set to sustain the self-discipline required.
EXERCISE RECOMMENDATIONS
Any exercise program should target 30 to 60 minutes of effort per day, most days of the week, ie, 150 to 300 minutes per week or more.38 But beginners should start low and go slow to avoid dropout, musculoskeletal strain, and joint injury.
The American College of Sports Medicine (ACSM)38,39 recommends combining aerobic and progressive resistance exercise as the core components of an exercise program. The aerobic component can include anaerobic high-intensity interval training (see discussion below). In addition, we recommend flexibility and balance exercises for obese patients.40
Combining aerobic and resistance exercises likely results in greater decreases in abdominal adiposity in the obese.41 In addition, the aerobic portion of a combined exercise regimen can improve functional capacity, and the resistance portion may prevent injury by strengthening the muscles, bones, and joint support systems.42 Adding exercises that promote flexibility and balance helps with range of motion and prevents injuries while exercising.43 These exercises not only expend calories during the exercise itself, but also increase resting energy expenditure for the remainder of the day, as the effects of the raised metabolism persist for hours.44
Aerobic exercise is the foundation
Aerobic exercises that involve large muscle groups, especially walking, should be the foundation of cardiopulmonary exercise for obese persons.45 Many patients can tolerate weight-bearing exercises such as walking or bike riding, but for some, exercises with limited or no weight-bearing such as swimming or aqua-aerobics are better.46
Tips for prescribing. Patients should exercise:
- On 5 or 6 days each week
- At low to moderate intensity (30%–60% of maximum oxygen consumption [Vo2 max])
- For at least 150 minutes per week, with a long-term goal of 300 minutes per week
- By walking, riding a stationary bicycle, or swimming.38,47
To mobilize and use free fatty acids as an energy source, lower-intensity longer-duration aerobic exercise is preferred.5 Thus, frequent, low-intensity or moderate-intensity training (30%–60% of Vo2 max) of longer duration (at least 60 minutes) may be the best approach to losing body fat in obese persons.5,48 Early on in the exercise program, keep the intensity low, as high-intensity training will preferentially use stored glycogen or carbohydrate as an energy substrate rather than free fatty acids or fat.5
With light-moderate exercise, the heart rate will increase and patients will perspire, but they still should be able to carry on a conversation.
Measure (or have patients measure) the heart rate using the radial artery in their wrist after 6 minutes of walking. A pulse of 100 beats per minute or more is associated with an exercise intensity of approximately 50% (or more) of Vo2 max.5
A study of 136 obese men and women who exercised for 6 months found that those doing aerobic exercise only and those doing a combination of aerobic and resistance exercise had greater cardiopulmonary fitness, greater reductions in abdominal and visceral fat, and more improved insulin sensitivity than those doing resistance exercise only.41 Although the aerobiconly group lost more weight (6 lb) than the aerobic-plus-resistance group (5.1 lb) and the resistance-only group (1.4 lb), combining aerobic and resistance exercise is considered optimal.
All physical activity is beneficial, but activities that have less impact on the joints are less likely to cause injuries and joint pain. Aerobic activities that are especially useful in obese adults include walking at a speed of at least 2.5 miles per hour, bicycling, jogging, treadmill walking, swimming, aqua-aerobics, rowing, and low-impact aerobics classes.
Walking is the easiest way for most people to start their program, as it is safe, accessible, and relatively cheap with respect to equipment.35 Adding a simple pedometer or smartphone app to measure the amount of exercise, together with physician counseling, may improve compliance and thus weight loss.49,50
Obese patients may have been inactive for quite a while. Therefore, the sessions should be short and low-intensity at first, then steadily progress.51 To minimize dropout, avoid hard exercise too soon for people with a low exercise capacity or high body mass index at baseline, and give positive feedback and encouragement at each visit.52
It is reasonable to introduce other aerobic exercises to vary the routine, use other muscle groups, and reduce the chance of injury from overuse of one muscle or joint group. Then, as cardiorespiratory fitness improves, the patient will be more confident about trying activities that are more challenging, such as jogging and aerobics classes. An aerobic exercise program consisting only of swimming is less efficacious for weight loss in this population.53
High-intensity interval training
High-intensity interval training involves relatively brief bursts of vigorous exercise separated by periods of recovery and is a time-efficient, novel alternative to continuous exercise.54 The exercise component is anaerobic, meaning muscle movement that does not require oxygen. Anaerobic exercise uses fast-twitch muscle fibers, and thus helps that musculature to become stronger, larger, and more toned. Evidence suggests that high-intensity interval training induces health-enhancing adaptations similar to those of continuous exercise, despite a substantially lower time commitment.41
The ACSM recommends that most adults engage in moderate-intensity cardiorespiratory exercise training for at least 30 minutes a day on at least 5 days a week for a total of at least 150 minutes per week, or high-intensity cardiorespiratory exercise training for at least 20 minutes a day on at least 3 days a week for a goal of 75 minutes a week.38 Thus, high-intensity interval training may be attractive for obese patients because it entails a shorter time commitment to achieve similar weight loss and improved insulin sensitivity than low-intensity or moderate-intensity continuous exercise.
High-intensity exercise has been shown to be effective for obese patients if they can do it.54–56 In one study,57 134 obese patients, mean age 53, underwent supervised high-intensity interval training with resistance training two or three times a week, were encouraged to perform one or two additional exercise sessions a week (unsupervised), and were counseled to follow a Mediterranean diet. At 9 months, investigators noted a significant reduction in body mass, waist circumference, and fat mass.
A study of 12 weeks of high-intensity interval training, moderate-intensity interval training, or no exercise in 34 obese adolescent girls noted that body mass and percentage body fat were significantly decreased with both interval training regimens. However, the high-intensity group had greater reductions in waist circumference and more significant improvements in blood lipid levels, adiponectin levels, and insulin sensitivity.58
Of 62 overweight and obese patients (mean age 53.3, mean body mass index 35.8 kg/m2), 97% adhered to a program of high-intensity interval training over 9 months, which resulted in an average weekly energy expenditure of 1,582 kcal.55 Clinically and statistically significant improvements occurred in body mass (–5.3 kg), body mass index (–1.9 kg/m2), and waist circumference (–5.8 cm) (P < .0001 for all variables). Total fat mass, trunk fat mass, and lipid levels also significantly improved (P < .0001), and the prevalence of metabolic syndrome was reduced by 32.5% (P < .05).
In a meta-analysis of the effect of exercise on overweight adults, training of moderate or high intensity was noted to have the highest potential to reduce visceral adipose tissue in overweight men and women.13 Another meta-analysis noted that high-intensity interval training appeared to promote more improvement in fitness and similar improvements in some cardiometabolic risk factors than moderate exercise performed for at least 8 to 12 weeks in overweight patients.56
A typical progressive exercise program for obese adults is shown in Table 1.
Progressive resistance exercise
Progressive resistance exercises are generally easier for obese patients, as they are not aerobically challenging, allow patients to exercise around physically active people who thus motivate them, and encourage positive feelings about completing their exercise sets.59 The result is improved muscular fitness, socialization, and increased confidence in their abilities (self-efficacy).59
Progressive resistance exercises also promote favorable energy balance and reduced visceral fat deposition through enhanced basal metabolism and activity levels while counteracting age- and disease-related muscle wasting.59 They have been shown to improve cognitive ability, self-esteem, movement control, muscle mass, strength, glucose control, insulin sensitivity, resting blood pressure, lipid profile, and bone mineral density and to reduce fat weight, low back pain, arthritic discomfort, insomnia, anxiety, and depression.60
Gym neophytes should spend a few sessions with a personal trainer to learn how to use the equipment.
While the primary goal of resistance training is more muscle strength, it can reduce fat and weight, burning up to 170 kcal in a 20-minute intense exercise session.61 It reduces both total body fat and visceral adipose tissue, thus benefiting obese persons by reducing insulin resistance.62 All exercise, and especially resistance exercise, can help to strengthen the musculoskeletal system, reduce muscle atrophy, and improve bone mineral density.63
The ACSM guidelines38 recommend progressive resistance exercise on 2 or 3 nonconsecutive days a week. It should involve:
- Exercises that work 8 to 10 muscle groups per session
- Two to four sets of 8 to 12 repetitions for each muscle group.
Exercising on nonconsecutive days allows time for the complete cycle of muscle tissue remodeling.64 Such self-regulated intensity reduces the likelihood of excessive delayed-onset muscle soreness, which can discourage new participants.65
To prevent muscle injury, obese people should begin with low-intensity workouts using lower resistance, one set of 8 to 12 repetitions 2 days a week. Then, they should gradually but progressively increase the intensity, volume, and frequency of the training.47 This will obviate a plateau in training and will maximize musculoskeletal adaptation. The prescription should include exercises for the upper body (eg, biceps curls), lower body (eg, leg presses), and the midsection (eg, abdominal curl-ups, which give better abdominal muscle engagement and less risk to the back than crunches) and focus on the correct exercise form and function rather than the amount of resistance or weight lifted.
A typical progressive resistance exercise program for obese adults is shown in Table 2.
Flexibility exercise
Flexibility exercise involves stretching to improve the movement of muscles, joints, and ligaments.45 While not specifically used in an energy-expenditure strategy, flexibility (or mobility) exercises help to increase or maintain joint range of motion and can reduce muscle and joint pain associated with obesity and exercise.66
The ACSM recommends that stretching exercises be done when the muscles are warm after a brief warm-up or exercise session.38 Typically, muscles should be stretched for at least 15 seconds, and stretching is recommended at a frequency of 2 to 4 days per week.38
A good way to incorporate flexibility exercise is to join a yoga class, as yoga has been shown to improve strength and flexibility and may help control physiologic variables such as blood pressure, lipids, respiration, heart rate, and metabolic rate to improve overall exercise capacity in obese patients.67
Balance exercise
Balance exercises help obese patients improve their stability. Poor balance is associated with injuries, accidents, and falls during activities of daily living.68
Balance, the ability to maintain the body’s center of gravity within its base of support, can be categorized as static (sustaining the body in static equilibrium or within its base of support) or dynamic (maintaining equilibrium during a transition from a dynamic to a static state), which is more challenging.69 Doing both static and dynamic balance training maximizes balance and stability.69 While most activities that involve moving the body or body parts (such as walking) will improve balance, some additional balance exercises can be beneficial.
Balance exercises can be done without any equipment. Examples are balancing on one foot for 15 seconds and standing up and sitting down without using the hands. However, specific equipment can help, including physioballs, stability balls, cut-in-half stability balls, balance discs, balance wedges, wobble boards, rocker boards, and Indo boards.70 In fact, balance boards and stability balls engage more muscle fibers in other areas of the body (lower back, lower abs, quads, hamstrings, and calves) than exercises done without those balancing devices.71
Balance training for at least 10 minutes a day, 3 days a week, for 4 weeks that incorporates various methods of balance training appears to improve balance.56 Obese patients commencing a program should start with static balance exercises and then progress to dynamic ones. In addition, as balance training progresses, obese patients can integrate balance and stability training exercises with other pieces of equipment, such as performing squats on a balance board, and then gradually add weights (eg, dumbbells) to the exercise.
An example of a weekly comprehensive exercise program for an obese patient that incorporates all major exercise types is provided in Table 3. In addition, some smartphone apps that are especially helpful in overweight newcomers to exercise include Couch-to-5K, GymGoal 2, Moves, Fitbit, Workout Trainer, Endomondo, MapMyFitness, Fitocracy, and Fitness Buddy.
BARIATRIC SURGERY AND LIFESTYLE MANAGEMENT FOR OBESITY
Bariatric surgery is a safe and effective treatment for severe obesity and comorbidities including type 2 diabetes mellitus, but weight loss and health outcomes vary considerably among individuals.72,73 Of importance, postoperative weight loss after bariatric surgery and long-term weight loss largely depend on the extent to which patients can make and sustain changes to their lifestyle, including diet, exercise, and behavior modification.72,74
Exercise, especially supervised, is associated with more weight loss after bariatric surgery.61 In a meta-analysis of bariatric patients, exercise participants involved in moderate or greater levels of exercise lost a mean of 3.6 kg more than the minimal exercise groups.75 Another meta-analysis noted the beneficial effects of exercise incorporating more than 30 minutes a day of moderate physical activity following bariatric surgery and was associated with a greater weight loss of over 4% of body mass index.76 These findings were consistent with those of yet another meta-analysis.77
In summary, exercise appears to significantly increase weight loss after bariatric surgery.
TREATMENT CONSIDERATIONS IN MORBID OBESITY
Challenges faced by severely obese or morbidly obese patients affect their exercise options. The types of exercise they are able to perform are limited in most cases to very-low-impact, low-intensity exercises, which may not be as efficient in weight loss or weight maintenance.48 Therefore, it may be prudent to set more conservative weight-loss goals for them, especially early in the program. Compliance and success rates may be better with low-impact activities such as walking, water aerobics, stationary cycling, and resistance training in the severely obese population.
The more severe the obesity, the more comorbidities such as diabetes, hypertension, hyperlipidemia, arthritis, sleep apnea, gastroesophageal reflux disease, and the greater the risk of metabolic syndrome—and conversely, the greater the potential benefit from bariatric surgery followed by exercise.74
A LONG-TERM ENDEAVOR
For obese patients, a comprehensive exercise program will improve functional status, favorably influence cardiovascular risk factors, and help with weight loss or weight maintenance.
Managing obesity is a long-term endeavor.78 For it to succeed, both the patient and the physician need to keep up their efforts. To keep the patient from becoming discouraged, the clinician should focus not just on weight, but also on improvements in metabolic profile and cardiorespiratory fitness. In addition, a careful evaluation, a clear exercise prescription, defined goals, ongoing monitoring (by the patient and the provider), frequent feedback, and charting of progress will improve daily performance and the chance of long-term success.
Although exercise is probably less effective than diet in reducing weight, most studies show that adding it to a diet regimen will increase the weight loss.1,2 Guidelines from the American Heart Association, American College of Cardiology, and Obesity Society recommend a comprehensive lifestyle program that includes a low-calorie diet as well as an increase in physical activity.3
Here, we review the many benefits of exercise for obese patients, not only in terms of weight loss, but also its positive cardiovascular and metabolic effects. Then we discuss how to motivate and prescribe exercise for this challenging group.
EXERCISE IMPROVES WEIGHT LOSS
Increasing energy expenditure by exercising can mobilize and burn stored fat and thus lead to weight loss.4
Typically, with no changes in caloric intake, exercising 60 minutes at low intensity most days of the week will remove up to 0.5 lb per week.5 Exercising harder for longer will take off more weight, up to 3 lb per week.1,6 Some practitioners believe that the total volume of exercise (frequency multiplied by time) is more important than the intensity in determining the amount of weight loss.2,7,8
Ross et al9 randomized 101 obese men to try to lose weight by exercising at a low to moderate intensity, to try to lose weight by dieting, to exercise without the goal of losing weight, or to do nothing (the control group). About half the participants declined or dropped out, but 52 completed the trial. The weight-loss-through-exercise group had lost approximately 15 lb by 12 weeks; the diet group lost a similar amount. Total body fat, visceral fat, and abdominal obesity were all reduced with both diet- and exercise-induced weight loss.
In a study in 130 severely obese adults, after 6 months of high-intensity physical activity for a mean duration of 71 minutes per week, those on an exercise-and-diet regimen lost an average of 24 lb, compared with 18 lb with diet alone.10
Another trial involved obese patients who were instructed to jog the equivalent of 20 miles (32.2 km) a week, with no restriction on caloric intake.11 They lost only 2.9 kg (6.5 lb) over 8 months. Increased food intake explained this minimal weight loss.
In an analysis of 20 studies, exercise-only interventions of 4 months or less resulted in a mean weekly weight loss of 0.4 lb (0.2 kg), with a total loss of about 5 lb (2.3 kg).12
A systematic review of 15 studies noted that aerobic exercise for 3 months or more resulted in a significant reduction in visceral adipose tissue in overweight men and women as measured by computed tomography.13
Effects that different types of exercise have on weight loss
In a study of 119 sedentary adults who were overweight or obese and who were randomized to aerobic, resistance, or combined aerobic-resistance training over 8 months, those involved in aerobic or combined aerobic and resistance training had the greatest reduction in total body and fat mass.14 Given that the combined aerobic-resistance training program required twice the time commitment of the aerobic-alone program, the authors suggested that the most efficient manner of reducing body and fat mass is aerobic training alone.14 In contrast, if the goal is to increase lean muscle mass rather than lose weight and fat, then resistance training would be preferred.14
A meta-analysis confirmed the benefit of aerobic exercise, which resulted in significantly more loss in weight (1.2 kg, 2.6 lb), waist circumference (1.57 cm), and fat mass (1.2 kg, 2.6 lb) than resistance training.15 However, combined aerobic and resistance training was even better, with significantly more weight loss (2.0 kg, 4.4 lb) and fat mass reduction (1.9 kg, 4.2 lb).15
In summary, aerobic and combined aerobic-resistance training appear to be more effective for weight management in obese people than resistance training alone.
ADDITIONAL BENEFITS OF EXERCISE
Increasing regular physical activity through structured exercise has the additional benefits of improving physical fitness, flexibility, mobility, and cardiovascular health.16,17
Even before patients lose a significant amount of weight (eg, 10%), low-intensity exercise such as walking 30 to 60 minutes most days of the week will rapidly improve cardiorespiratory fitness and have positive effects on cardiovascular risk factors such as hypertension, elevated blood glucose, and dyslipidemia.18,19 Aerobic exercise and resistance training also reduce chronic inflammation, which is a strong indicator of future disease, especially in obese patients who have high levels of inflammatory biomarkers.20,21
Even if he or she does not lose much weight, an obese exercising person with good cardiorespiratory fitness has lower cardiovascular risk than a person who is not obese but is poorly conditioned.22
Exercise lowers blood pressure
Overactivity of the sympathetic nervous system is thought to account for over 50% of all cases of hypertension.23 Obesity in concert with diabetes is characterized by sympathetic overactivity and progressive loss of cardiac parasympathetic activity.24 Cardiac autonomic neuropathy is an underestimated risk factor for the increased cardiovascular morbidity and mortality associated with obesity and diabetes, and physical exercise may promote restoration of cardioprotective autonomic modulation in the heart.24
Several studies have shown that aerobic endurance exercise lowers blood pressure in patients with hypertension, and reduction in sympathetic neural activity has been reported as one of the main mechanisms explaining this effect.23 Another mechanism is endothelium-mediated vasodilation: even a single exercise session may increase the bioavailability of nitric oxide and decrease postexercise blood pressure.25
Different types of exercise have been shown to have different effects on blood pressure.
Aerobic training has been shown to reduce systolic blood pressure by 5.2 to 11.0 mm Hg and diastolic blood pressure by 3.0 to 7.7 mm Hg.26
The hypotensive effect of endurance aerobic training is probably mediated at least in part by a reduction in systemic vascular resistance through decreased activity of the sympathetic and renin-angiotensin systems and through improved insulin sensitivity.26 Other factors that may be involved include improved endothelium-dependent vasodilation, enhanced baroreceptor sensitivity, and arterial compliance.26
Dynamic resistance exercise has less of an effect than aerobic exercise, but it has been shown to reduce systolic blood pressure by 0.5 to 4.8 mm Hg and diastolic blood pressure by 0.5 to 4.1 mm Hg.26
In a meta-analysis of studies of resistance training lasting more than 1 month in healthy adults age 18 and older, the authors noted that resistance training induced a significant blood pressure reduction in 28 normotensive or prehypertensive study groups (–3.9/–3.9 mm Hg), whereas the reduction was not significant for the five hypertensive study groups.27
Isometric resistance exercise has been associated with small cardiovascular benefits, but has been shown to reduce systolic blood pressure by 10.5 to 16.5 mm Hg and diastolic blood pressure by 0.62 to 16.4 mm Hg.26
Exercise improves type 2 diabetes
Regular physical activity improves glycemic control and can prevent or delay the onset of type 2 diabetes mellitus.28 Furthermore, physical activity positively affects lipid levels, lowers blood pressure, reduces the rate of cardiovascular events, and restores quality of life in patients with type 2 diabetes.24,29
A meta-analysis of the effect of supervised exercise in adults with type 2 diabetes found that structured exercise achieved the following:
- Lowered systolic blood pressure by 2.42 mm Hg (95% confidence interval 0.45–4.39)
- Lowered diastolic blood pressure by 2.23 mm Hg (1.25–3.21)
- Raised the level of high-density lipoprotein cholesterol by 0.04 mmol/L (0.02–0.07)
- Lowered the level of low-density lipoprotein cholesterol by 0.16 mmol/L (0.01–0.30).30
The metabolic stress from physical exercise can increase oxidation of carbohydrates during exercise, increase postexercise consumption of oxygen (which can increase the rate of fat oxidation during recovery periods after exercise), improve glucose tolerance and insulin sensitivity, and reduce glycemia for 2 to 72 hours depending on the intensity and duration of the exercise.25
Exercise lowers the Framingham risk score
Exercise improves several of the risk factors for coronary artery disease used in calculating the Framingham risk score—ie, systolic blood pressure, total cholesterol, and high-density lipoprotein cholesterol—and thus can significantly lower this number. (It is important to remember that the Framingham score is a surrogate end point of cardiovascular risk that may correlate with a real clinical end point but does not necessarily have a guaranteed relationship.)
In a study of a 12-week exercise program in middle-aged women (ages 40–55), treadmill running for 30 minutes a day 3 days a week significantly reduced 10-year cardiovascular risk scores: 10-year risk 2.2% vs 4.3% in the nonexercising group.31 Others have also shown that enhanced levels of fitness are associated with lower 10-year Framingham risk estimates.32
A study of 31 healthy sedentary adults ages 50 to 65 who were randomized to an unsupervised but pedometer-monitored home-based walking program of 30 minutes of brisk walking 5 days a week noted significant reductions in systolic and diastolic blood pressure and stroke risk, and increased functional capacity in the walking group at 12 weeks.33 Thus, the Framingham risk scores were significantly lower in the exercising group than in with the control group.33
Given that overweight and obese patients who are starting to exercise may find jogging or running daunting, it should also be noted that three brisk 10-minute walks a day are at least as effective as one continuous 30-minute walk in reducing cardiovascular risk in previously sedentary people.34
SETTING ‘SMART’ GOALS
Because obese adults typically do not comply well with prescriptions for exercise, it is important to educate them about its benefits and to provide tools such as perceived exertion scales so they can monitor their exercise, document their performance, and chart their progress; smartphone apps can also be helpful.35 Supervised exercise may improve compliance and results.36 Initially, personal trainers are excellent for starting a habit change, but they are expensive. Virtual trainers are now available and cost far less.37
People do not become obese overnight.They gain weight over a long time. Likewise, weight reduction takes time if done in a sustainable and healthy manner. Thus, SMART goals—specific, measurable, attainable, realistic, timely—should be set to sustain the self-discipline required.
EXERCISE RECOMMENDATIONS
Any exercise program should target 30 to 60 minutes of effort per day, most days of the week, ie, 150 to 300 minutes per week or more.38 But beginners should start low and go slow to avoid dropout, musculoskeletal strain, and joint injury.
The American College of Sports Medicine (ACSM)38,39 recommends combining aerobic and progressive resistance exercise as the core components of an exercise program. The aerobic component can include anaerobic high-intensity interval training (see discussion below). In addition, we recommend flexibility and balance exercises for obese patients.40
Combining aerobic and resistance exercises likely results in greater decreases in abdominal adiposity in the obese.41 In addition, the aerobic portion of a combined exercise regimen can improve functional capacity, and the resistance portion may prevent injury by strengthening the muscles, bones, and joint support systems.42 Adding exercises that promote flexibility and balance helps with range of motion and prevents injuries while exercising.43 These exercises not only expend calories during the exercise itself, but also increase resting energy expenditure for the remainder of the day, as the effects of the raised metabolism persist for hours.44
Aerobic exercise is the foundation
Aerobic exercises that involve large muscle groups, especially walking, should be the foundation of cardiopulmonary exercise for obese persons.45 Many patients can tolerate weight-bearing exercises such as walking or bike riding, but for some, exercises with limited or no weight-bearing such as swimming or aqua-aerobics are better.46
Tips for prescribing. Patients should exercise:
- On 5 or 6 days each week
- At low to moderate intensity (30%–60% of maximum oxygen consumption [Vo2 max])
- For at least 150 minutes per week, with a long-term goal of 300 minutes per week
- By walking, riding a stationary bicycle, or swimming.38,47
To mobilize and use free fatty acids as an energy source, lower-intensity longer-duration aerobic exercise is preferred.5 Thus, frequent, low-intensity or moderate-intensity training (30%–60% of Vo2 max) of longer duration (at least 60 minutes) may be the best approach to losing body fat in obese persons.5,48 Early on in the exercise program, keep the intensity low, as high-intensity training will preferentially use stored glycogen or carbohydrate as an energy substrate rather than free fatty acids or fat.5
With light-moderate exercise, the heart rate will increase and patients will perspire, but they still should be able to carry on a conversation.
Measure (or have patients measure) the heart rate using the radial artery in their wrist after 6 minutes of walking. A pulse of 100 beats per minute or more is associated with an exercise intensity of approximately 50% (or more) of Vo2 max.5
A study of 136 obese men and women who exercised for 6 months found that those doing aerobic exercise only and those doing a combination of aerobic and resistance exercise had greater cardiopulmonary fitness, greater reductions in abdominal and visceral fat, and more improved insulin sensitivity than those doing resistance exercise only.41 Although the aerobiconly group lost more weight (6 lb) than the aerobic-plus-resistance group (5.1 lb) and the resistance-only group (1.4 lb), combining aerobic and resistance exercise is considered optimal.
All physical activity is beneficial, but activities that have less impact on the joints are less likely to cause injuries and joint pain. Aerobic activities that are especially useful in obese adults include walking at a speed of at least 2.5 miles per hour, bicycling, jogging, treadmill walking, swimming, aqua-aerobics, rowing, and low-impact aerobics classes.
Walking is the easiest way for most people to start their program, as it is safe, accessible, and relatively cheap with respect to equipment.35 Adding a simple pedometer or smartphone app to measure the amount of exercise, together with physician counseling, may improve compliance and thus weight loss.49,50
Obese patients may have been inactive for quite a while. Therefore, the sessions should be short and low-intensity at first, then steadily progress.51 To minimize dropout, avoid hard exercise too soon for people with a low exercise capacity or high body mass index at baseline, and give positive feedback and encouragement at each visit.52
It is reasonable to introduce other aerobic exercises to vary the routine, use other muscle groups, and reduce the chance of injury from overuse of one muscle or joint group. Then, as cardiorespiratory fitness improves, the patient will be more confident about trying activities that are more challenging, such as jogging and aerobics classes. An aerobic exercise program consisting only of swimming is less efficacious for weight loss in this population.53
High-intensity interval training
High-intensity interval training involves relatively brief bursts of vigorous exercise separated by periods of recovery and is a time-efficient, novel alternative to continuous exercise.54 The exercise component is anaerobic, meaning muscle movement that does not require oxygen. Anaerobic exercise uses fast-twitch muscle fibers, and thus helps that musculature to become stronger, larger, and more toned. Evidence suggests that high-intensity interval training induces health-enhancing adaptations similar to those of continuous exercise, despite a substantially lower time commitment.41
The ACSM recommends that most adults engage in moderate-intensity cardiorespiratory exercise training for at least 30 minutes a day on at least 5 days a week for a total of at least 150 minutes per week, or high-intensity cardiorespiratory exercise training for at least 20 minutes a day on at least 3 days a week for a goal of 75 minutes a week.38 Thus, high-intensity interval training may be attractive for obese patients because it entails a shorter time commitment to achieve similar weight loss and improved insulin sensitivity than low-intensity or moderate-intensity continuous exercise.
High-intensity exercise has been shown to be effective for obese patients if they can do it.54–56 In one study,57 134 obese patients, mean age 53, underwent supervised high-intensity interval training with resistance training two or three times a week, were encouraged to perform one or two additional exercise sessions a week (unsupervised), and were counseled to follow a Mediterranean diet. At 9 months, investigators noted a significant reduction in body mass, waist circumference, and fat mass.
A study of 12 weeks of high-intensity interval training, moderate-intensity interval training, or no exercise in 34 obese adolescent girls noted that body mass and percentage body fat were significantly decreased with both interval training regimens. However, the high-intensity group had greater reductions in waist circumference and more significant improvements in blood lipid levels, adiponectin levels, and insulin sensitivity.58
Of 62 overweight and obese patients (mean age 53.3, mean body mass index 35.8 kg/m2), 97% adhered to a program of high-intensity interval training over 9 months, which resulted in an average weekly energy expenditure of 1,582 kcal.55 Clinically and statistically significant improvements occurred in body mass (–5.3 kg), body mass index (–1.9 kg/m2), and waist circumference (–5.8 cm) (P < .0001 for all variables). Total fat mass, trunk fat mass, and lipid levels also significantly improved (P < .0001), and the prevalence of metabolic syndrome was reduced by 32.5% (P < .05).
In a meta-analysis of the effect of exercise on overweight adults, training of moderate or high intensity was noted to have the highest potential to reduce visceral adipose tissue in overweight men and women.13 Another meta-analysis noted that high-intensity interval training appeared to promote more improvement in fitness and similar improvements in some cardiometabolic risk factors than moderate exercise performed for at least 8 to 12 weeks in overweight patients.56
A typical progressive exercise program for obese adults is shown in Table 1.
Progressive resistance exercise
Progressive resistance exercises are generally easier for obese patients, as they are not aerobically challenging, allow patients to exercise around physically active people who thus motivate them, and encourage positive feelings about completing their exercise sets.59 The result is improved muscular fitness, socialization, and increased confidence in their abilities (self-efficacy).59
Progressive resistance exercises also promote favorable energy balance and reduced visceral fat deposition through enhanced basal metabolism and activity levels while counteracting age- and disease-related muscle wasting.59 They have been shown to improve cognitive ability, self-esteem, movement control, muscle mass, strength, glucose control, insulin sensitivity, resting blood pressure, lipid profile, and bone mineral density and to reduce fat weight, low back pain, arthritic discomfort, insomnia, anxiety, and depression.60
Gym neophytes should spend a few sessions with a personal trainer to learn how to use the equipment.
While the primary goal of resistance training is more muscle strength, it can reduce fat and weight, burning up to 170 kcal in a 20-minute intense exercise session.61 It reduces both total body fat and visceral adipose tissue, thus benefiting obese persons by reducing insulin resistance.62 All exercise, and especially resistance exercise, can help to strengthen the musculoskeletal system, reduce muscle atrophy, and improve bone mineral density.63
The ACSM guidelines38 recommend progressive resistance exercise on 2 or 3 nonconsecutive days a week. It should involve:
- Exercises that work 8 to 10 muscle groups per session
- Two to four sets of 8 to 12 repetitions for each muscle group.
Exercising on nonconsecutive days allows time for the complete cycle of muscle tissue remodeling.64 Such self-regulated intensity reduces the likelihood of excessive delayed-onset muscle soreness, which can discourage new participants.65
To prevent muscle injury, obese people should begin with low-intensity workouts using lower resistance, one set of 8 to 12 repetitions 2 days a week. Then, they should gradually but progressively increase the intensity, volume, and frequency of the training.47 This will obviate a plateau in training and will maximize musculoskeletal adaptation. The prescription should include exercises for the upper body (eg, biceps curls), lower body (eg, leg presses), and the midsection (eg, abdominal curl-ups, which give better abdominal muscle engagement and less risk to the back than crunches) and focus on the correct exercise form and function rather than the amount of resistance or weight lifted.
A typical progressive resistance exercise program for obese adults is shown in Table 2.
Flexibility exercise
Flexibility exercise involves stretching to improve the movement of muscles, joints, and ligaments.45 While not specifically used in an energy-expenditure strategy, flexibility (or mobility) exercises help to increase or maintain joint range of motion and can reduce muscle and joint pain associated with obesity and exercise.66
The ACSM recommends that stretching exercises be done when the muscles are warm after a brief warm-up or exercise session.38 Typically, muscles should be stretched for at least 15 seconds, and stretching is recommended at a frequency of 2 to 4 days per week.38
A good way to incorporate flexibility exercise is to join a yoga class, as yoga has been shown to improve strength and flexibility and may help control physiologic variables such as blood pressure, lipids, respiration, heart rate, and metabolic rate to improve overall exercise capacity in obese patients.67
Balance exercise
Balance exercises help obese patients improve their stability. Poor balance is associated with injuries, accidents, and falls during activities of daily living.68
Balance, the ability to maintain the body’s center of gravity within its base of support, can be categorized as static (sustaining the body in static equilibrium or within its base of support) or dynamic (maintaining equilibrium during a transition from a dynamic to a static state), which is more challenging.69 Doing both static and dynamic balance training maximizes balance and stability.69 While most activities that involve moving the body or body parts (such as walking) will improve balance, some additional balance exercises can be beneficial.
Balance exercises can be done without any equipment. Examples are balancing on one foot for 15 seconds and standing up and sitting down without using the hands. However, specific equipment can help, including physioballs, stability balls, cut-in-half stability balls, balance discs, balance wedges, wobble boards, rocker boards, and Indo boards.70 In fact, balance boards and stability balls engage more muscle fibers in other areas of the body (lower back, lower abs, quads, hamstrings, and calves) than exercises done without those balancing devices.71
Balance training for at least 10 minutes a day, 3 days a week, for 4 weeks that incorporates various methods of balance training appears to improve balance.56 Obese patients commencing a program should start with static balance exercises and then progress to dynamic ones. In addition, as balance training progresses, obese patients can integrate balance and stability training exercises with other pieces of equipment, such as performing squats on a balance board, and then gradually add weights (eg, dumbbells) to the exercise.
An example of a weekly comprehensive exercise program for an obese patient that incorporates all major exercise types is provided in Table 3. In addition, some smartphone apps that are especially helpful in overweight newcomers to exercise include Couch-to-5K, GymGoal 2, Moves, Fitbit, Workout Trainer, Endomondo, MapMyFitness, Fitocracy, and Fitness Buddy.
BARIATRIC SURGERY AND LIFESTYLE MANAGEMENT FOR OBESITY
Bariatric surgery is a safe and effective treatment for severe obesity and comorbidities including type 2 diabetes mellitus, but weight loss and health outcomes vary considerably among individuals.72,73 Of importance, postoperative weight loss after bariatric surgery and long-term weight loss largely depend on the extent to which patients can make and sustain changes to their lifestyle, including diet, exercise, and behavior modification.72,74
Exercise, especially supervised, is associated with more weight loss after bariatric surgery.61 In a meta-analysis of bariatric patients, exercise participants involved in moderate or greater levels of exercise lost a mean of 3.6 kg more than the minimal exercise groups.75 Another meta-analysis noted the beneficial effects of exercise incorporating more than 30 minutes a day of moderate physical activity following bariatric surgery and was associated with a greater weight loss of over 4% of body mass index.76 These findings were consistent with those of yet another meta-analysis.77
In summary, exercise appears to significantly increase weight loss after bariatric surgery.
TREATMENT CONSIDERATIONS IN MORBID OBESITY
Challenges faced by severely obese or morbidly obese patients affect their exercise options. The types of exercise they are able to perform are limited in most cases to very-low-impact, low-intensity exercises, which may not be as efficient in weight loss or weight maintenance.48 Therefore, it may be prudent to set more conservative weight-loss goals for them, especially early in the program. Compliance and success rates may be better with low-impact activities such as walking, water aerobics, stationary cycling, and resistance training in the severely obese population.
The more severe the obesity, the more comorbidities such as diabetes, hypertension, hyperlipidemia, arthritis, sleep apnea, gastroesophageal reflux disease, and the greater the risk of metabolic syndrome—and conversely, the greater the potential benefit from bariatric surgery followed by exercise.74
A LONG-TERM ENDEAVOR
For obese patients, a comprehensive exercise program will improve functional status, favorably influence cardiovascular risk factors, and help with weight loss or weight maintenance.
Managing obesity is a long-term endeavor.78 For it to succeed, both the patient and the physician need to keep up their efforts. To keep the patient from becoming discouraged, the clinician should focus not just on weight, but also on improvements in metabolic profile and cardiorespiratory fitness. In addition, a careful evaluation, a clear exercise prescription, defined goals, ongoing monitoring (by the patient and the provider), frequent feedback, and charting of progress will improve daily performance and the chance of long-term success.
- Thorogood A, Mottillo S, Shimony A, et al. Isolated aerobic exercise and weight loss: a systematic review and meta-analysis of randomized controlled trials. Am J Med 2011; 124:747–755.
- Church T. Exercise in obesity, metabolic syndrome, and diabetes. Prog Cardiovasc Dis 2011; 53:412–418.
- Jensen MD, Ryan DH, Apovian CM, et al; American College of Cardiology/American Heart Association Task Force on Practice Guidelines; Obesity Society. 2013 AHA/ACC/TOS guideline for the management of overweight and obesity in adults: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines and The Obesity Society. Circulation 2014; 129(suppl 2):S102–S138.
- Strasser B. Physical activity in obesity and metabolic syndrome. Ann N Y Acad Sci 2013; 1281:141–159.
- Poirier P, Despres JP. Exercise in weight management of obesity. Cardiol Clin 2001; 19:459–470.
- Shaw K, Gennat H, O’Rourke P, Del Mar C. Exercise for overweight or obesity. Cochrane Database Syst Rev 2006; 4:CD003817.
- Slentz CA, Houmard JA, Kraus WE. Exercise, abdominal obesity, skeletal muscle, and metabolic risk: evidence for a dose response. Obesity (Silver Spring) 2009; 17(suppl 3):S27–S33.
- Ross R, Hudson R, Stotz PJ, Lam M. Effects of exercise amount and intensity on abdominal obesity and glucose tolerance in obese adults: a randomized trial. Ann Intern Med 2015; 162:325–334.
- Ross R, Dagnone D, Jones PJ, et al. Reduction in obesity and related comorbid conditions after diet-induced weight loss or exercise-induced weight loss in men. A randomized, controlled trial. Ann Intern Med 2000; 133:92–103.
- Goodpaster BH, Delany JP, Otto AD, et al. Effects of diet and physical activity interventions on weight loss and cardiometabolic risk factors in severely obese adults: a randomized trial. JAMA 2010; 304:1795–1802.
- Slentz CA, Duscha BD, Johnson JL, et al. Effects of the amount of exercise on body weight, body composition, and measures of central obesity: STRRIDE—a randomized controlled study. Arch Intern Med 2004; 164:31–39.
- Ross R, Janssen I. Physical activity, total and regional obesity: dose-response considerations. Med Sci Sports Exerc 2001; 33(suppl 6):S521–S529.
- Vissers D, Hens W, Taeymans J, Baeyens JP, Poortmans J, Van Gaal L. The effect of exercise on visceral adipose tissue in overweight adults: a systematic review and meta-analysis. PLoS One 2013; 8:e56415.
- Willis LH, Slentz CA, Bateman LA, et al. Effects of aerobic and/or resistance training on body mass and fat mass in overweight or obese adults. J Appl Physiol (1985) 2012; 113:1831–1837.
- Schwingshackl L, Dias S, Strasser B, Hoffmann G. Impact of different training modalities on anthropometric and metabolic characteristics in overweight/obese subjects: a systematic review and network meta-analysis. PLoS One 2013; 8:e82853.
- Cook CM, Schoeller DA. Physical activity and weight control: conflicting findings. Curr Opin Clin Nutr Metab Care 2011; 14:419–424.
- Choo J, Lee J, Cho JH, Burke LE, Sekikawa A, Jae SY. Effects of weight management by exercise modes on markers of subclinical atherosclerosis and cardiometabolic profile among women with abdominal obesity: a randomized controlled trial. BMC Cardiovasc Disord 2014; 14:82.
- Carroll S, Dudfield M. What is the relationship between exercise and metabolic abnormalities? A review of the metabolic syndrome. Sports Med 2004; 34:371–418.
- Shaibi GQ, Ryder JR, Kim JY, Barraza E. Exercise for obese youth: refocusing attention from weight loss to health gains. Exerc Sport Sci Rev 2015; 43:41–47.
- You T, Arsenis NC, Disanzo BL, Lamonte MJ. Effects of exercise training on chronic inflammation in obesity: current evidence and potential mechanisms. Sports Med 2013; 43:243–256.
- Bluher S, Petroff D, Wagner A, et al. The one year exercise and lifestyle intervention program KLAKS: effects on anthropometric parameters, cardiometabolic risk factors and glycemic control in childhood obesity. Metabolism 2014; 63:422–430.
- Lee CD, Blair SN, Jackson AS. Cardiorespiratory fitness, body composition, and all-cause and cardiovascular disease mortality in men. Am J Clin Nutr 1999; 69:373–380.
- Leosco D, Parisi V, Femminella GD, et al. Effects of exercise training on cardiovascular adrenergic system. Front Physiol 2013; 4:348.
- Voulgari C, Pagoni S, Vinik A, Poirier P. Exercise improves cardiac autonomic function in obesity and diabetes. Metabolism 2013; 62:609–621.
- Asano RY, Sales MM, Browne RA, et al. Acute effects of physical exercise in type 2 diabetes: a review. World J Diabetes 2014; 5:659–665.
- Brook RD, Appel LJ, Rubenfire M, et al; American Heart Association Professional Education Committee of the Council for High Blood Pressure Research, Council on Cardiovascular and Stroke Nursing, Council on Epidemiology and Prevention, and Council on Nutrition, Physical Activity. Beyond medications and diet: alternative approaches to lowering blood pressure: a scientific statement from the American Heart Association. Hypertension 2013; 61:1360–1383.
- Cornelissen VA, Fagard RH, Coeckelberghs E, Vanhees L. Impact of resistance training on blood pressure and other cardiovascular risk factors: a meta-analysis of randomized, controlled trials. Hypertension 2011; 58:950–958.
- Ades PA. A lifestyle program of exercise and weight loss is effective in preventing and treating type 2 diabetes mellitus: why are programs not more available? Prev Med 2015: S0091–7435(15)00085–7.
- Ades PA, Savage PD, Marney AM, Harvey J, Evans KA. Remission of recently diagnosed type 2 diabetes mellitus with weight loss and exercise. J Cardiopulm Rehabil Prev 2015; 35:193–197.
- Hayashino Y, Jackson JL, Fukumori N, Nakamura F, Fukuhara S. Effects of supervised exercise on lipid profiles and blood pressure control in people with type 2 diabetes mellitus: a meta-analysis of randomized controlled trials. Diabetes Res Clin Pract 2012; 98:349–360.
- Saffi MA, Polanczyk CA, Rabelo-Silva ER. Lifestyle interventions reduce cardiovascular risk in patients with coronary artery disease: a randomized clinical trial. Eur J Cardiovasc Nurs 2014; 13:436–443.
- LaMonte MJ, Durstine JL, Addy CL, Irwin ML, Ainsworth BE. Physical activity, physical fitness, and Framingham 10-year risk score: the cross-cultural activity participation study. J Cardiopulm Rehabil 2001; 21:63–70.
- Tully MA, Cupples ME, Chan WS, McGlade K, Young IS. Brisk walking, fitness, and cardiovascular risk: a randomized controlled trial in primary care. Prev Med 2005; 41:622–628.
- Murphy M, Nevill A, Neville C, Biddle S, Hardman A. Accumulating brisk walking for fitness, cardiovascular risk, and psychological health. Med Sci Sports Exerc 2002; 34:1468–1474.
- Colley RC, Hills AP, King NA, Byrne NM. Exercise-induced energy expenditure: implications for exercise prescription and obesity. Patient Educ Couns 2010; 79:327–332.
- Baillot A, Mampuya WM, Comeau E, Méziat-Burdin A, Langlois MF. Feasibility and impacts of supervised exercise training in subjects with obesity awaiting bariatric surgery: a pilot study. Obes Surg 2013; 23:882–891.
- Lowe S, ÓLaighin G. The age of the virtual trainer. Procedia Engineering 2012; 34:242–247.
- Garber CE, Blissmer B, Deschenes MR, et al; American College of Sports Medicine. American College of Sports Medicine position stand. Quantity and quality of exercise for developing and maintaining cardiorespiratory, musculoskeletal, and neuromotor fitness in apparently healthy adults: guidance for prescribing exercise. Med Sci Sports Exerc 2011; 43:1334–1359.
- Donnelly JE, Blair SN, Jakicic JM, Manore MM, Rankin JW, Smith BK; American College of Sports Medicine. American College of Sports Medicine position stand. Appropriate physical activity intervention strategies for weight loss and prevention of weight regain for adults. Med Sci Sports Exerc 2009; 41:459–471.
- Montero-Fernandez N, Serra-Rexach JA. Role of exercise on sarcopenia in the elderly. Eur J Phys Rehabil Med 2013; 49:131–143.
- Davidson LE, Hudson R, Kilpatrick K, et al. Effects of exercise modality on insulin resistance and functional limitation in older adults: a randomized controlled trial. Arch Intern Med 2009; 169:122–131.
- Liu CJ, Latham NK. Progressive resistance strength training for improving physical function in older adults. Cochrane Database Syst Rev 2009; 3:CD002759.
- Manini TM, Newman AB, Fielding R, et al; LIFE Research Group. Effects of exercise on mobility in obese and nonobese older adults. Obesity (Silver Spring) 2010; 18:1168–1175.
- Hackney KJ, Engels HJ, Gretebeck RJ. Resting energy expenditure and delayed-onset muscle soreness after full-body resistance training with an eccentric concentration. J Strength Cond Res 2008; 22:1602–1609.
- Siddiqui NI, Nessa A, Hossain MA. Regular physical exercise: way to healthy life. Mymensingh Med J 2010; 19:154–158.
- Chicco AJ. Exercise training in prevention and rehabilitation: which training mode is best? Minerva Cardioangiol 2008; 56:557–570.
- Westcott WL, Winett RA, Annesi JJ, Wojcik JR, Anderson ES, Madden PJ. Prescribing physical activity: applying the ACSM protocols for exercise type, intensity, and duration across 3 training frequencies. Phys Sportsmed 2009; 37:51–58.
- Mougios V1, Kazaki M, Christoulas K, Ziogas G, Petridou A. Does the intensity of an exercise programme modulate body composition changes? Int J Sports Med 2006; 27:178–181.
- Richardson CR, Newton TL, Abraham JJ, Sen A, Jimbo M, Swartz AM. A meta-analysis of pedometer-based walking interventions and weight loss. Ann Fam Med 2008; 6:69–77.
- Stovitz SD, VanWormer JJ, Center BA, Bremer KL. Pedometers as a means to increase ambulatory activity for patients seen at a family medicine clinic. J Am Board Fam Pract 2005; 18:335–343.
- Lepor NE, Fouchia DD, McCullough PA. New vistas for the treatment of obesity: turning the tide against the leading cause of morbidity and cardiovascular mortality in the developed world. Rev Cardiovasc Med 2013; 14:20–40.
- Wittmer M, Volpatti M, Piazzalonga S, Hoffmann A. Expectation, satisfaction, and predictors of dropout in cardiac rehabilitation. Eur J Prev Cardiol 2012; 19:1082–1088.
- Gwinup G. Weight loss without dietary restriction: efficacy of different forms of aerobic exercise. Am J Sports Med 1987; 15:275–279.
- Jung ME, Bourne JE, Little JP. Where does HIT fit? An examination of the affective response to high-intensity intervals in comparison to continuous moderate- and continuous vigorous-intensity exercise in the exercise intensity-affect continuum. PLoS One 2014; 9:e114541.
- Gremeaux V, Drigny J, Nigam A, et al. Long-term lifestyle intervention with optimized high-intensity interval training improves body composition, cardiometabolic risk, and exercise parameters in patients with abdominal obesity. Am J Phys Med Rehabil 2012; 91:941–950.
- Kessler HS, Sisson SB, Short KR. The potential for high-intensity interval training to reduce cardiometabolic disease risk. Sports Med 2012; 42:489–509.
- Dalzill C, Nigam A, Juneau M, et al. Intensive lifestyle intervention improves cardiometabolic and exercise parameters in metabolically healthy obese and metabolically unhealthy obese individuals. Can J Cardiol 2014; 30:434–440.
- Racil G, Ben Ounis O, Hammouda O, et al. Effects of high vs moderate exercise intensity during interval training on lipids and adiponectin levels in obese young females. Eur J Appl Physiol 2013; 113:2531–2540.
- Willey KA, Singh MA. Battling insulin resistance in elderly obese people with type 2 diabetes: bring on the heavy weights. Diabetes Care 2003; 26:1580–1588.
- Westcott WL. Resistance training is medicine: effects of strength training on health. Curr Sports Med Rep 2012; 11:209–216.
- Haltom RW, Kraemer RR, Sloan RA, Hebert EP, Frank K, Tryniecki JL. Circuit weight training and its effects on excess postexercise oxygen consumption. Med Sci Sports Exerc 1999; 31:1613–1618.
- Strasser B, Schobersberger W. Evidence for resistance training as a treatment therapy in obesity. J Obes 2011; pii:482564.
- Fonseca H, Moreira-Gonçalves D, Coriolano HJ, Duarte JA. Bone quality: the determinants of bone strength and fragility. Sports Med 2014; 44:37–53.
- Candow DG, Burke DG. Effect of short-term equal-volume resistance training with different workout frequency on muscle mass and strength in untrained men and women. J Strength Cond Res 2007; 21:204–207.
- Trost Z, France CR, Thomas JS. Pain-related fear and avoidance of physical exertion following delayed-onset muscle soreness. Pain 2011; 152:1540–1547.
- Mathus-Vliegen EM. Obesity and the elderly. J Clin Gastroenterol 2012; 46:533–544.
- Dhananjai S, Sadashiv, Tiwari S, Dutt K, Kumar R. Reducing psychological distress and obesity through yoga practice. Int J Yoga 2013; 6:66–70.
- Mathus-Vliegen EM; Obesity Management Task Force of the European Association for the Study of Obesity. Prevalence, pathophysiology, health consequences and treatment options of obesity in the elderly: a guideline. Obes Facts 2012; 5:460–483.
- DiStefano LJ, Clark MA, Padua DA. Evidence supporting balance training in healthy individuals: a systemic review. J Strength Cond Res 2009; 23:2718–2731.
- Ogaya S, Ikezoe T, Soda N, Ichihashi N. Effects of balance training using wobble boards in the elderly. J Strength Cond Res 2011; 25:2616–2622.
- Sukalinggam CL, Sukalinggam GL, Kasim F, Yusof A. Stability ball training on lower back strength has greater effect in untrained female compared to male. J Hum Kinet 2012; 33:133–141.
- Kalarchian M, Turk M, Elliott J, Gourash W. Lifestyle management for enhancing outcomes after bariatric surgery. Curr Diab Rep 2014; 14:540.
- Rothwell L, Kow L, Toouli J. Effect of a post-operative structured exercise programme on short-term weight loss after obesity surgery using adjustable gastric bands. Obes Surg 2015; 25:126–128.
- Mechanick JI, Youdim A, Jones DB, et al. Clinical practice guidelines for the perioperative nutritional, metabolic, and nonsurgical support of the bariatric surgery patient—2013 update: cosponsored by American Association of Clinical Endocrinologists, the Obesity Society, and American Society for Metabolic & Bariatric Surgery. Surg Obes Relat Dis 2013; 9:159–191.
- Egberts K, Brown WA, Brennan L, O’Brien PE. Does exercise improve weight loss after bariatric surgery? A systematic review. Obes Surg 2012; 22:335–341.
- Livhits M, Mercado C, Yermilov I, et al. Exercise following bariatric surgery: systematic review. Obes Surg 2010; 20:657–665.
- Jacobi D, Ciangura C, Couet C, Oppert JM. Physical activity and weight loss following bariatric surgery. Obes Rev 2011; 12:366–377.
- Wadden TA, Foster GD, Letizia KA. One-year behavioral treatment of obesity: comparison of moderate and severe caloric restriction and the effects of weight maintenance therapy. J Consult Clin Psychol 1994; 62:165–171.
- Thorogood A, Mottillo S, Shimony A, et al. Isolated aerobic exercise and weight loss: a systematic review and meta-analysis of randomized controlled trials. Am J Med 2011; 124:747–755.
- Church T. Exercise in obesity, metabolic syndrome, and diabetes. Prog Cardiovasc Dis 2011; 53:412–418.
- Jensen MD, Ryan DH, Apovian CM, et al; American College of Cardiology/American Heart Association Task Force on Practice Guidelines; Obesity Society. 2013 AHA/ACC/TOS guideline for the management of overweight and obesity in adults: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines and The Obesity Society. Circulation 2014; 129(suppl 2):S102–S138.
- Strasser B. Physical activity in obesity and metabolic syndrome. Ann N Y Acad Sci 2013; 1281:141–159.
- Poirier P, Despres JP. Exercise in weight management of obesity. Cardiol Clin 2001; 19:459–470.
- Shaw K, Gennat H, O’Rourke P, Del Mar C. Exercise for overweight or obesity. Cochrane Database Syst Rev 2006; 4:CD003817.
- Slentz CA, Houmard JA, Kraus WE. Exercise, abdominal obesity, skeletal muscle, and metabolic risk: evidence for a dose response. Obesity (Silver Spring) 2009; 17(suppl 3):S27–S33.
- Ross R, Hudson R, Stotz PJ, Lam M. Effects of exercise amount and intensity on abdominal obesity and glucose tolerance in obese adults: a randomized trial. Ann Intern Med 2015; 162:325–334.
- Ross R, Dagnone D, Jones PJ, et al. Reduction in obesity and related comorbid conditions after diet-induced weight loss or exercise-induced weight loss in men. A randomized, controlled trial. Ann Intern Med 2000; 133:92–103.
- Goodpaster BH, Delany JP, Otto AD, et al. Effects of diet and physical activity interventions on weight loss and cardiometabolic risk factors in severely obese adults: a randomized trial. JAMA 2010; 304:1795–1802.
- Slentz CA, Duscha BD, Johnson JL, et al. Effects of the amount of exercise on body weight, body composition, and measures of central obesity: STRRIDE—a randomized controlled study. Arch Intern Med 2004; 164:31–39.
- Ross R, Janssen I. Physical activity, total and regional obesity: dose-response considerations. Med Sci Sports Exerc 2001; 33(suppl 6):S521–S529.
- Vissers D, Hens W, Taeymans J, Baeyens JP, Poortmans J, Van Gaal L. The effect of exercise on visceral adipose tissue in overweight adults: a systematic review and meta-analysis. PLoS One 2013; 8:e56415.
- Willis LH, Slentz CA, Bateman LA, et al. Effects of aerobic and/or resistance training on body mass and fat mass in overweight or obese adults. J Appl Physiol (1985) 2012; 113:1831–1837.
- Schwingshackl L, Dias S, Strasser B, Hoffmann G. Impact of different training modalities on anthropometric and metabolic characteristics in overweight/obese subjects: a systematic review and network meta-analysis. PLoS One 2013; 8:e82853.
- Cook CM, Schoeller DA. Physical activity and weight control: conflicting findings. Curr Opin Clin Nutr Metab Care 2011; 14:419–424.
- Choo J, Lee J, Cho JH, Burke LE, Sekikawa A, Jae SY. Effects of weight management by exercise modes on markers of subclinical atherosclerosis and cardiometabolic profile among women with abdominal obesity: a randomized controlled trial. BMC Cardiovasc Disord 2014; 14:82.
- Carroll S, Dudfield M. What is the relationship between exercise and metabolic abnormalities? A review of the metabolic syndrome. Sports Med 2004; 34:371–418.
- Shaibi GQ, Ryder JR, Kim JY, Barraza E. Exercise for obese youth: refocusing attention from weight loss to health gains. Exerc Sport Sci Rev 2015; 43:41–47.
- You T, Arsenis NC, Disanzo BL, Lamonte MJ. Effects of exercise training on chronic inflammation in obesity: current evidence and potential mechanisms. Sports Med 2013; 43:243–256.
- Bluher S, Petroff D, Wagner A, et al. The one year exercise and lifestyle intervention program KLAKS: effects on anthropometric parameters, cardiometabolic risk factors and glycemic control in childhood obesity. Metabolism 2014; 63:422–430.
- Lee CD, Blair SN, Jackson AS. Cardiorespiratory fitness, body composition, and all-cause and cardiovascular disease mortality in men. Am J Clin Nutr 1999; 69:373–380.
- Leosco D, Parisi V, Femminella GD, et al. Effects of exercise training on cardiovascular adrenergic system. Front Physiol 2013; 4:348.
- Voulgari C, Pagoni S, Vinik A, Poirier P. Exercise improves cardiac autonomic function in obesity and diabetes. Metabolism 2013; 62:609–621.
- Asano RY, Sales MM, Browne RA, et al. Acute effects of physical exercise in type 2 diabetes: a review. World J Diabetes 2014; 5:659–665.
- Brook RD, Appel LJ, Rubenfire M, et al; American Heart Association Professional Education Committee of the Council for High Blood Pressure Research, Council on Cardiovascular and Stroke Nursing, Council on Epidemiology and Prevention, and Council on Nutrition, Physical Activity. Beyond medications and diet: alternative approaches to lowering blood pressure: a scientific statement from the American Heart Association. Hypertension 2013; 61:1360–1383.
- Cornelissen VA, Fagard RH, Coeckelberghs E, Vanhees L. Impact of resistance training on blood pressure and other cardiovascular risk factors: a meta-analysis of randomized, controlled trials. Hypertension 2011; 58:950–958.
- Ades PA. A lifestyle program of exercise and weight loss is effective in preventing and treating type 2 diabetes mellitus: why are programs not more available? Prev Med 2015: S0091–7435(15)00085–7.
- Ades PA, Savage PD, Marney AM, Harvey J, Evans KA. Remission of recently diagnosed type 2 diabetes mellitus with weight loss and exercise. J Cardiopulm Rehabil Prev 2015; 35:193–197.
- Hayashino Y, Jackson JL, Fukumori N, Nakamura F, Fukuhara S. Effects of supervised exercise on lipid profiles and blood pressure control in people with type 2 diabetes mellitus: a meta-analysis of randomized controlled trials. Diabetes Res Clin Pract 2012; 98:349–360.
- Saffi MA, Polanczyk CA, Rabelo-Silva ER. Lifestyle interventions reduce cardiovascular risk in patients with coronary artery disease: a randomized clinical trial. Eur J Cardiovasc Nurs 2014; 13:436–443.
- LaMonte MJ, Durstine JL, Addy CL, Irwin ML, Ainsworth BE. Physical activity, physical fitness, and Framingham 10-year risk score: the cross-cultural activity participation study. J Cardiopulm Rehabil 2001; 21:63–70.
- Tully MA, Cupples ME, Chan WS, McGlade K, Young IS. Brisk walking, fitness, and cardiovascular risk: a randomized controlled trial in primary care. Prev Med 2005; 41:622–628.
- Murphy M, Nevill A, Neville C, Biddle S, Hardman A. Accumulating brisk walking for fitness, cardiovascular risk, and psychological health. Med Sci Sports Exerc 2002; 34:1468–1474.
- Colley RC, Hills AP, King NA, Byrne NM. Exercise-induced energy expenditure: implications for exercise prescription and obesity. Patient Educ Couns 2010; 79:327–332.
- Baillot A, Mampuya WM, Comeau E, Méziat-Burdin A, Langlois MF. Feasibility and impacts of supervised exercise training in subjects with obesity awaiting bariatric surgery: a pilot study. Obes Surg 2013; 23:882–891.
- Lowe S, ÓLaighin G. The age of the virtual trainer. Procedia Engineering 2012; 34:242–247.
- Garber CE, Blissmer B, Deschenes MR, et al; American College of Sports Medicine. American College of Sports Medicine position stand. Quantity and quality of exercise for developing and maintaining cardiorespiratory, musculoskeletal, and neuromotor fitness in apparently healthy adults: guidance for prescribing exercise. Med Sci Sports Exerc 2011; 43:1334–1359.
- Donnelly JE, Blair SN, Jakicic JM, Manore MM, Rankin JW, Smith BK; American College of Sports Medicine. American College of Sports Medicine position stand. Appropriate physical activity intervention strategies for weight loss and prevention of weight regain for adults. Med Sci Sports Exerc 2009; 41:459–471.
- Montero-Fernandez N, Serra-Rexach JA. Role of exercise on sarcopenia in the elderly. Eur J Phys Rehabil Med 2013; 49:131–143.
- Davidson LE, Hudson R, Kilpatrick K, et al. Effects of exercise modality on insulin resistance and functional limitation in older adults: a randomized controlled trial. Arch Intern Med 2009; 169:122–131.
- Liu CJ, Latham NK. Progressive resistance strength training for improving physical function in older adults. Cochrane Database Syst Rev 2009; 3:CD002759.
- Manini TM, Newman AB, Fielding R, et al; LIFE Research Group. Effects of exercise on mobility in obese and nonobese older adults. Obesity (Silver Spring) 2010; 18:1168–1175.
- Hackney KJ, Engels HJ, Gretebeck RJ. Resting energy expenditure and delayed-onset muscle soreness after full-body resistance training with an eccentric concentration. J Strength Cond Res 2008; 22:1602–1609.
- Siddiqui NI, Nessa A, Hossain MA. Regular physical exercise: way to healthy life. Mymensingh Med J 2010; 19:154–158.
- Chicco AJ. Exercise training in prevention and rehabilitation: which training mode is best? Minerva Cardioangiol 2008; 56:557–570.
- Westcott WL, Winett RA, Annesi JJ, Wojcik JR, Anderson ES, Madden PJ. Prescribing physical activity: applying the ACSM protocols for exercise type, intensity, and duration across 3 training frequencies. Phys Sportsmed 2009; 37:51–58.
- Mougios V1, Kazaki M, Christoulas K, Ziogas G, Petridou A. Does the intensity of an exercise programme modulate body composition changes? Int J Sports Med 2006; 27:178–181.
- Richardson CR, Newton TL, Abraham JJ, Sen A, Jimbo M, Swartz AM. A meta-analysis of pedometer-based walking interventions and weight loss. Ann Fam Med 2008; 6:69–77.
- Stovitz SD, VanWormer JJ, Center BA, Bremer KL. Pedometers as a means to increase ambulatory activity for patients seen at a family medicine clinic. J Am Board Fam Pract 2005; 18:335–343.
- Lepor NE, Fouchia DD, McCullough PA. New vistas for the treatment of obesity: turning the tide against the leading cause of morbidity and cardiovascular mortality in the developed world. Rev Cardiovasc Med 2013; 14:20–40.
- Wittmer M, Volpatti M, Piazzalonga S, Hoffmann A. Expectation, satisfaction, and predictors of dropout in cardiac rehabilitation. Eur J Prev Cardiol 2012; 19:1082–1088.
- Gwinup G. Weight loss without dietary restriction: efficacy of different forms of aerobic exercise. Am J Sports Med 1987; 15:275–279.
- Jung ME, Bourne JE, Little JP. Where does HIT fit? An examination of the affective response to high-intensity intervals in comparison to continuous moderate- and continuous vigorous-intensity exercise in the exercise intensity-affect continuum. PLoS One 2014; 9:e114541.
- Gremeaux V, Drigny J, Nigam A, et al. Long-term lifestyle intervention with optimized high-intensity interval training improves body composition, cardiometabolic risk, and exercise parameters in patients with abdominal obesity. Am J Phys Med Rehabil 2012; 91:941–950.
- Kessler HS, Sisson SB, Short KR. The potential for high-intensity interval training to reduce cardiometabolic disease risk. Sports Med 2012; 42:489–509.
- Dalzill C, Nigam A, Juneau M, et al. Intensive lifestyle intervention improves cardiometabolic and exercise parameters in metabolically healthy obese and metabolically unhealthy obese individuals. Can J Cardiol 2014; 30:434–440.
- Racil G, Ben Ounis O, Hammouda O, et al. Effects of high vs moderate exercise intensity during interval training on lipids and adiponectin levels in obese young females. Eur J Appl Physiol 2013; 113:2531–2540.
- Willey KA, Singh MA. Battling insulin resistance in elderly obese people with type 2 diabetes: bring on the heavy weights. Diabetes Care 2003; 26:1580–1588.
- Westcott WL. Resistance training is medicine: effects of strength training on health. Curr Sports Med Rep 2012; 11:209–216.
- Haltom RW, Kraemer RR, Sloan RA, Hebert EP, Frank K, Tryniecki JL. Circuit weight training and its effects on excess postexercise oxygen consumption. Med Sci Sports Exerc 1999; 31:1613–1618.
- Strasser B, Schobersberger W. Evidence for resistance training as a treatment therapy in obesity. J Obes 2011; pii:482564.
- Fonseca H, Moreira-Gonçalves D, Coriolano HJ, Duarte JA. Bone quality: the determinants of bone strength and fragility. Sports Med 2014; 44:37–53.
- Candow DG, Burke DG. Effect of short-term equal-volume resistance training with different workout frequency on muscle mass and strength in untrained men and women. J Strength Cond Res 2007; 21:204–207.
- Trost Z, France CR, Thomas JS. Pain-related fear and avoidance of physical exertion following delayed-onset muscle soreness. Pain 2011; 152:1540–1547.
- Mathus-Vliegen EM. Obesity and the elderly. J Clin Gastroenterol 2012; 46:533–544.
- Dhananjai S, Sadashiv, Tiwari S, Dutt K, Kumar R. Reducing psychological distress and obesity through yoga practice. Int J Yoga 2013; 6:66–70.
- Mathus-Vliegen EM; Obesity Management Task Force of the European Association for the Study of Obesity. Prevalence, pathophysiology, health consequences and treatment options of obesity in the elderly: a guideline. Obes Facts 2012; 5:460–483.
- DiStefano LJ, Clark MA, Padua DA. Evidence supporting balance training in healthy individuals: a systemic review. J Strength Cond Res 2009; 23:2718–2731.
- Ogaya S, Ikezoe T, Soda N, Ichihashi N. Effects of balance training using wobble boards in the elderly. J Strength Cond Res 2011; 25:2616–2622.
- Sukalinggam CL, Sukalinggam GL, Kasim F, Yusof A. Stability ball training on lower back strength has greater effect in untrained female compared to male. J Hum Kinet 2012; 33:133–141.
- Kalarchian M, Turk M, Elliott J, Gourash W. Lifestyle management for enhancing outcomes after bariatric surgery. Curr Diab Rep 2014; 14:540.
- Rothwell L, Kow L, Toouli J. Effect of a post-operative structured exercise programme on short-term weight loss after obesity surgery using adjustable gastric bands. Obes Surg 2015; 25:126–128.
- Mechanick JI, Youdim A, Jones DB, et al. Clinical practice guidelines for the perioperative nutritional, metabolic, and nonsurgical support of the bariatric surgery patient—2013 update: cosponsored by American Association of Clinical Endocrinologists, the Obesity Society, and American Society for Metabolic & Bariatric Surgery. Surg Obes Relat Dis 2013; 9:159–191.
- Egberts K, Brown WA, Brennan L, O’Brien PE. Does exercise improve weight loss after bariatric surgery? A systematic review. Obes Surg 2012; 22:335–341.
- Livhits M, Mercado C, Yermilov I, et al. Exercise following bariatric surgery: systematic review. Obes Surg 2010; 20:657–665.
- Jacobi D, Ciangura C, Couet C, Oppert JM. Physical activity and weight loss following bariatric surgery. Obes Rev 2011; 12:366–377.
- Wadden TA, Foster GD, Letizia KA. One-year behavioral treatment of obesity: comparison of moderate and severe caloric restriction and the effects of weight maintenance therapy. J Consult Clin Psychol 1994; 62:165–171.
KEY POINTS
- Exercise not only helps people lose weight and keep it off, it lowers blood pressure, improves lipid levels, improves insulin sensitivity, and lowers blood glucose levels.
- Of the various types of exercise, aerobic exercise provides the most benefits, but resistance, flexibility, and balance exercises have additional value. Specifically, continuous moderate-intensity aerobic or high-intensity interval training in combination with some resistance exercises appears to be most effective for weight management.
- For people who are extremely obese, low-impact exercises performed for a longer duration may be more manageable and are still effective.
- The clinician should monitor the patient’s compliance and progress and give appropriate encouragement and feedback for sustained success.