User login
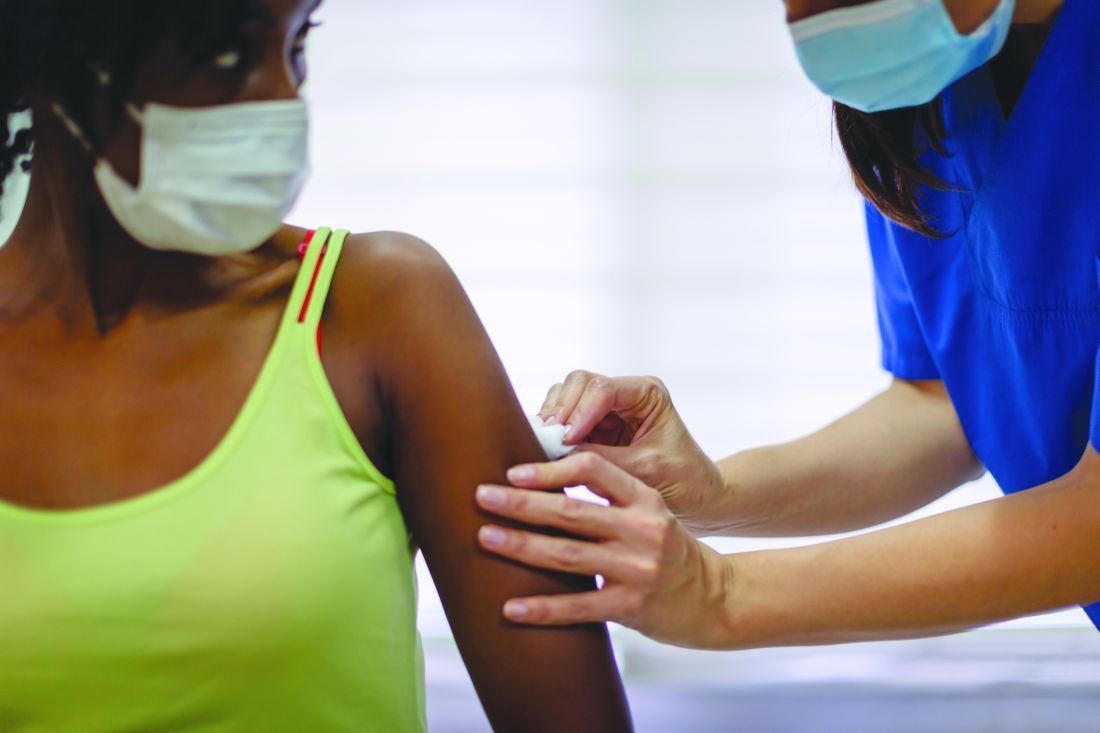
Researchers stress importance of second COVID-19 vaccine dose for infliximab users
Patients being treated with infliximab had weakened immune responses to the first dose of the ChAdOx1 nCoV-19 (Oxford/AstraZeneca) and BNT162b2 (Pfizer/BioNTech) vaccines, compared with patients on vedolizumab (Entyvio), although a very significant number of patients from both groups seroconverted after their second dose, according to a new U.K. study of patients with inflammatory bowel disease (IBD).
“Antibody testing and adapted vaccine schedules should be considered to protect these at-risk patients,” Nicholas A. Kennedy, PhD, MBBS, of the University of Exeter (England) and colleagues wrote in a preprint published March 29 on MedRxiv.
Infliximab is an anti–tumor necrosis factor (anti-TNF) monoclonal antibody that’s approved to treat adult and pediatric Crohn’s disease and ulcerative colitis, as well as rheumatoid arthritis, ankylosing spondylitis, psoriatic arthritis, and plaque psoriasis, whereas vedolizumab, a gut selective anti-integrin alpha4beta7 monoclonal antibody that is not associated with impaired systemic immune responses, is approved to treat Crohn’s disease and ulcerative colitis in adults.
A previous study from Kennedy and colleagues revealed that IBD patients on infliximab showed a weakened COVID-19 antibody response compared with patients on vedolizumab. To determine if treatment with anti-TNF drugs impacted the efficacy of the first shot of these two-dose COVID-19 vaccines, the researchers used data from the CLARITY IBD study to assess 865 infliximab- and 428 vedolizumab-treated participants without evidence of prior SARS-CoV-2 infection who had received uninterrupted biologic therapy since being recruited between Sept. 22 and Dec. 23, 2020.
In the 3-10 weeks after initial vaccination, geometric mean concentrations for SARS-CoV-2 anti-spike protein receptor-binding protein antibodies were lower in patients on infliximab, compared with patients on vedolizumab for both the Pfizer (6.0 U/mL [5.9] versus 28.8 U/mL [5.4], P < .0001) and AstraZeneca (4.7 U/mL [4.9] versus 13.8 U/mL [5.9]; P < .0001) vaccines. The researchers’ multivariable models reinforced those findings, with antibody concentrations lower in infliximab-treated patients for both the Pfizer (fold change, 0.29; 95% confidence interval, 0.21-0.40; P < .0001) and AstraZeneca (FC, 0.39; 95% CI, 0.30-0.51; P < .0001) vaccines.
After second doses of the two-dose Pfizer vaccine, 85% of patients on infliximab and 86% of patients on vedolizumab seroconverted (P = .68); similarly high seroconversion rates were seen in patients who had been infected with SARS-CoV-2 prior to receiving either vaccine. Several patient characteristics were associated with lower antibody concentrations regardless of vaccine type: being 60 years or older, use of immunomodulators, having Crohn’s disease, and being a smoker. Alternatively, non-White ethnicity was associated with higher antibody concentrations.
Evidence has ‘unclear clinical significance’
“These data, which require peer review, do not change my opinion on the safety and efficacy of COVID-19 vaccines in patients taking TNF inhibitors such as infliximab as monotherapy for the treatment of psoriatic disease,” Joel M. Gelfand MD, director of the psoriasis and phototherapy treatment center at the University of Pennsylvania, Philadelphia, said in an interview.
“First, two peer-reviewed studies found good antibody response in patients on TNF inhibitors receiving COVID-19 vaccines (doi: 10.1136/annrheumdis-2021-220289; 10.1136/annrheumdis-2021-220272). Second, antibody responses were robust in the small cohort that received the second dose of a COVID-19 vaccine. We already know that, for the two messenger RNA-based vaccines available under emergency use authorization in the U.S., a second dose is required for optimal efficacy. Thus, evidence of a reduced antibody response after just one dose is of unclear clinical significance. Third, antibody responses are only a surrogate marker, and a low antibody response doesn’t necessarily mean the patient will not be protected by the vaccine.”
Focus on the second dose of a two-dose regimen
“Tell me about the response in people who got both doses of a vaccine that you’re supposed to get both doses of,” Jeffrey Curtis, MD, professor of medicine in the division of clinical immunology and rheumatology at the University of Alabama at Birmingham, said in an interview. “The number of patients in that subset was small [n = 27] but in my opinion that’s the most clinically relevant analysis and the one that patients and clinicians want answered.”
He also emphasized the uncertainty around what ‘protection’ means in these early days of studying COVID-19 vaccine responses. “You can define seroprotection or seroconversion as some absolute level of an antibody response, but if you want to say ‘Mrs. Smith, your antibody level was X,’ on whatever arbitrary scale with whoever’s arbitrary lab test, nobody actually knows that Mrs. Smith is now protected from SARS-CoV-2, or how protected,” he said.
“What is not terribly controversial is: If you can’t detect antibodies, the vaccine didn’t ‘take,’ if you will. But if I tell you that the mean antibody level was X with one drug and then 2X with another drug, does that mean that you’re twice as protected? We don’t know that. I’m fearful that people are looking at these studies and thinking that more is better. It might be, but we don’t know that to be true.”
Debating the cause of weakened immune responses
“The biological plausibility of being on an anti-TNF affecting your immune reaction to a messenger RNA or even a replication-deficient viral vector vaccine doesn’t make sense,” David T. Rubin, MD, professor of medicine at the University of Chicago and chair of the National Scientific Advisory Committee of the Crohn’s and Colitis Foundation, said in an interview.
“I’m sure immunologists may differ with me on this, but given what we have come to appreciate about these vaccine mechanisms, this finding doesn’t make intuitive sense. So we need to make sure that, when this happens, we look to the next studies and try to understand, was there any other confounder that may have resulted in these findings that was not adequately adjusted for or addressed in some other way?
“When you have a study of this size, you argue, ‘Because it’s so large, any effect that was seen must be real,’ ” he added. “Alternatively, to have a study of this size, by its very nature you are limited in being able to control for certain other factors or differences between the groups.”
That said, he commended the authors for their study and acknowledged the potential questions it raises about the single-shot Johnson & Johnson vaccine. “If you only get one and you’re on infliximab, this study implies that maybe that’s not enough,” he said. “Despite the fact that Johnson & Johnson was approved as a single dose, it may be necessary to think about it as the first of two, or maybe it’s not the preferred vaccine in this group of patients.”
The study was supported by the Royal Devon and Exeter and Hull University Hospital Foundation NHS Trusts and unrestricted educational grants from Biogen (Switzerland), Celltrion Healthcare (South Korea), Galapagos NV (Belgium), and F. Hoffmann-La Roche (Switzerland). The authors acknowledged numerous potential conflicts of interest, including receiving grants, personal fees, and nonfinancial support from various pharmaceutical companies.
Patients being treated with infliximab had weakened immune responses to the first dose of the ChAdOx1 nCoV-19 (Oxford/AstraZeneca) and BNT162b2 (Pfizer/BioNTech) vaccines, compared with patients on vedolizumab (Entyvio), although a very significant number of patients from both groups seroconverted after their second dose, according to a new U.K. study of patients with inflammatory bowel disease (IBD).
“Antibody testing and adapted vaccine schedules should be considered to protect these at-risk patients,” Nicholas A. Kennedy, PhD, MBBS, of the University of Exeter (England) and colleagues wrote in a preprint published March 29 on MedRxiv.
Infliximab is an anti–tumor necrosis factor (anti-TNF) monoclonal antibody that’s approved to treat adult and pediatric Crohn’s disease and ulcerative colitis, as well as rheumatoid arthritis, ankylosing spondylitis, psoriatic arthritis, and plaque psoriasis, whereas vedolizumab, a gut selective anti-integrin alpha4beta7 monoclonal antibody that is not associated with impaired systemic immune responses, is approved to treat Crohn’s disease and ulcerative colitis in adults.
A previous study from Kennedy and colleagues revealed that IBD patients on infliximab showed a weakened COVID-19 antibody response compared with patients on vedolizumab. To determine if treatment with anti-TNF drugs impacted the efficacy of the first shot of these two-dose COVID-19 vaccines, the researchers used data from the CLARITY IBD study to assess 865 infliximab- and 428 vedolizumab-treated participants without evidence of prior SARS-CoV-2 infection who had received uninterrupted biologic therapy since being recruited between Sept. 22 and Dec. 23, 2020.
In the 3-10 weeks after initial vaccination, geometric mean concentrations for SARS-CoV-2 anti-spike protein receptor-binding protein antibodies were lower in patients on infliximab, compared with patients on vedolizumab for both the Pfizer (6.0 U/mL [5.9] versus 28.8 U/mL [5.4], P < .0001) and AstraZeneca (4.7 U/mL [4.9] versus 13.8 U/mL [5.9]; P < .0001) vaccines. The researchers’ multivariable models reinforced those findings, with antibody concentrations lower in infliximab-treated patients for both the Pfizer (fold change, 0.29; 95% confidence interval, 0.21-0.40; P < .0001) and AstraZeneca (FC, 0.39; 95% CI, 0.30-0.51; P < .0001) vaccines.
After second doses of the two-dose Pfizer vaccine, 85% of patients on infliximab and 86% of patients on vedolizumab seroconverted (P = .68); similarly high seroconversion rates were seen in patients who had been infected with SARS-CoV-2 prior to receiving either vaccine. Several patient characteristics were associated with lower antibody concentrations regardless of vaccine type: being 60 years or older, use of immunomodulators, having Crohn’s disease, and being a smoker. Alternatively, non-White ethnicity was associated with higher antibody concentrations.
Evidence has ‘unclear clinical significance’
“These data, which require peer review, do not change my opinion on the safety and efficacy of COVID-19 vaccines in patients taking TNF inhibitors such as infliximab as monotherapy for the treatment of psoriatic disease,” Joel M. Gelfand MD, director of the psoriasis and phototherapy treatment center at the University of Pennsylvania, Philadelphia, said in an interview.
“First, two peer-reviewed studies found good antibody response in patients on TNF inhibitors receiving COVID-19 vaccines (doi: 10.1136/annrheumdis-2021-220289; 10.1136/annrheumdis-2021-220272). Second, antibody responses were robust in the small cohort that received the second dose of a COVID-19 vaccine. We already know that, for the two messenger RNA-based vaccines available under emergency use authorization in the U.S., a second dose is required for optimal efficacy. Thus, evidence of a reduced antibody response after just one dose is of unclear clinical significance. Third, antibody responses are only a surrogate marker, and a low antibody response doesn’t necessarily mean the patient will not be protected by the vaccine.”
Focus on the second dose of a two-dose regimen
“Tell me about the response in people who got both doses of a vaccine that you’re supposed to get both doses of,” Jeffrey Curtis, MD, professor of medicine in the division of clinical immunology and rheumatology at the University of Alabama at Birmingham, said in an interview. “The number of patients in that subset was small [n = 27] but in my opinion that’s the most clinically relevant analysis and the one that patients and clinicians want answered.”
He also emphasized the uncertainty around what ‘protection’ means in these early days of studying COVID-19 vaccine responses. “You can define seroprotection or seroconversion as some absolute level of an antibody response, but if you want to say ‘Mrs. Smith, your antibody level was X,’ on whatever arbitrary scale with whoever’s arbitrary lab test, nobody actually knows that Mrs. Smith is now protected from SARS-CoV-2, or how protected,” he said.
“What is not terribly controversial is: If you can’t detect antibodies, the vaccine didn’t ‘take,’ if you will. But if I tell you that the mean antibody level was X with one drug and then 2X with another drug, does that mean that you’re twice as protected? We don’t know that. I’m fearful that people are looking at these studies and thinking that more is better. It might be, but we don’t know that to be true.”
Debating the cause of weakened immune responses
“The biological plausibility of being on an anti-TNF affecting your immune reaction to a messenger RNA or even a replication-deficient viral vector vaccine doesn’t make sense,” David T. Rubin, MD, professor of medicine at the University of Chicago and chair of the National Scientific Advisory Committee of the Crohn’s and Colitis Foundation, said in an interview.
“I’m sure immunologists may differ with me on this, but given what we have come to appreciate about these vaccine mechanisms, this finding doesn’t make intuitive sense. So we need to make sure that, when this happens, we look to the next studies and try to understand, was there any other confounder that may have resulted in these findings that was not adequately adjusted for or addressed in some other way?
“When you have a study of this size, you argue, ‘Because it’s so large, any effect that was seen must be real,’ ” he added. “Alternatively, to have a study of this size, by its very nature you are limited in being able to control for certain other factors or differences between the groups.”
That said, he commended the authors for their study and acknowledged the potential questions it raises about the single-shot Johnson & Johnson vaccine. “If you only get one and you’re on infliximab, this study implies that maybe that’s not enough,” he said. “Despite the fact that Johnson & Johnson was approved as a single dose, it may be necessary to think about it as the first of two, or maybe it’s not the preferred vaccine in this group of patients.”
The study was supported by the Royal Devon and Exeter and Hull University Hospital Foundation NHS Trusts and unrestricted educational grants from Biogen (Switzerland), Celltrion Healthcare (South Korea), Galapagos NV (Belgium), and F. Hoffmann-La Roche (Switzerland). The authors acknowledged numerous potential conflicts of interest, including receiving grants, personal fees, and nonfinancial support from various pharmaceutical companies.
Patients being treated with infliximab had weakened immune responses to the first dose of the ChAdOx1 nCoV-19 (Oxford/AstraZeneca) and BNT162b2 (Pfizer/BioNTech) vaccines, compared with patients on vedolizumab (Entyvio), although a very significant number of patients from both groups seroconverted after their second dose, according to a new U.K. study of patients with inflammatory bowel disease (IBD).
“Antibody testing and adapted vaccine schedules should be considered to protect these at-risk patients,” Nicholas A. Kennedy, PhD, MBBS, of the University of Exeter (England) and colleagues wrote in a preprint published March 29 on MedRxiv.
Infliximab is an anti–tumor necrosis factor (anti-TNF) monoclonal antibody that’s approved to treat adult and pediatric Crohn’s disease and ulcerative colitis, as well as rheumatoid arthritis, ankylosing spondylitis, psoriatic arthritis, and plaque psoriasis, whereas vedolizumab, a gut selective anti-integrin alpha4beta7 monoclonal antibody that is not associated with impaired systemic immune responses, is approved to treat Crohn’s disease and ulcerative colitis in adults.
A previous study from Kennedy and colleagues revealed that IBD patients on infliximab showed a weakened COVID-19 antibody response compared with patients on vedolizumab. To determine if treatment with anti-TNF drugs impacted the efficacy of the first shot of these two-dose COVID-19 vaccines, the researchers used data from the CLARITY IBD study to assess 865 infliximab- and 428 vedolizumab-treated participants without evidence of prior SARS-CoV-2 infection who had received uninterrupted biologic therapy since being recruited between Sept. 22 and Dec. 23, 2020.
In the 3-10 weeks after initial vaccination, geometric mean concentrations for SARS-CoV-2 anti-spike protein receptor-binding protein antibodies were lower in patients on infliximab, compared with patients on vedolizumab for both the Pfizer (6.0 U/mL [5.9] versus 28.8 U/mL [5.4], P < .0001) and AstraZeneca (4.7 U/mL [4.9] versus 13.8 U/mL [5.9]; P < .0001) vaccines. The researchers’ multivariable models reinforced those findings, with antibody concentrations lower in infliximab-treated patients for both the Pfizer (fold change, 0.29; 95% confidence interval, 0.21-0.40; P < .0001) and AstraZeneca (FC, 0.39; 95% CI, 0.30-0.51; P < .0001) vaccines.
After second doses of the two-dose Pfizer vaccine, 85% of patients on infliximab and 86% of patients on vedolizumab seroconverted (P = .68); similarly high seroconversion rates were seen in patients who had been infected with SARS-CoV-2 prior to receiving either vaccine. Several patient characteristics were associated with lower antibody concentrations regardless of vaccine type: being 60 years or older, use of immunomodulators, having Crohn’s disease, and being a smoker. Alternatively, non-White ethnicity was associated with higher antibody concentrations.
Evidence has ‘unclear clinical significance’
“These data, which require peer review, do not change my opinion on the safety and efficacy of COVID-19 vaccines in patients taking TNF inhibitors such as infliximab as monotherapy for the treatment of psoriatic disease,” Joel M. Gelfand MD, director of the psoriasis and phototherapy treatment center at the University of Pennsylvania, Philadelphia, said in an interview.
“First, two peer-reviewed studies found good antibody response in patients on TNF inhibitors receiving COVID-19 vaccines (doi: 10.1136/annrheumdis-2021-220289; 10.1136/annrheumdis-2021-220272). Second, antibody responses were robust in the small cohort that received the second dose of a COVID-19 vaccine. We already know that, for the two messenger RNA-based vaccines available under emergency use authorization in the U.S., a second dose is required for optimal efficacy. Thus, evidence of a reduced antibody response after just one dose is of unclear clinical significance. Third, antibody responses are only a surrogate marker, and a low antibody response doesn’t necessarily mean the patient will not be protected by the vaccine.”
Focus on the second dose of a two-dose regimen
“Tell me about the response in people who got both doses of a vaccine that you’re supposed to get both doses of,” Jeffrey Curtis, MD, professor of medicine in the division of clinical immunology and rheumatology at the University of Alabama at Birmingham, said in an interview. “The number of patients in that subset was small [n = 27] but in my opinion that’s the most clinically relevant analysis and the one that patients and clinicians want answered.”
He also emphasized the uncertainty around what ‘protection’ means in these early days of studying COVID-19 vaccine responses. “You can define seroprotection or seroconversion as some absolute level of an antibody response, but if you want to say ‘Mrs. Smith, your antibody level was X,’ on whatever arbitrary scale with whoever’s arbitrary lab test, nobody actually knows that Mrs. Smith is now protected from SARS-CoV-2, or how protected,” he said.
“What is not terribly controversial is: If you can’t detect antibodies, the vaccine didn’t ‘take,’ if you will. But if I tell you that the mean antibody level was X with one drug and then 2X with another drug, does that mean that you’re twice as protected? We don’t know that. I’m fearful that people are looking at these studies and thinking that more is better. It might be, but we don’t know that to be true.”
Debating the cause of weakened immune responses
“The biological plausibility of being on an anti-TNF affecting your immune reaction to a messenger RNA or even a replication-deficient viral vector vaccine doesn’t make sense,” David T. Rubin, MD, professor of medicine at the University of Chicago and chair of the National Scientific Advisory Committee of the Crohn’s and Colitis Foundation, said in an interview.
“I’m sure immunologists may differ with me on this, but given what we have come to appreciate about these vaccine mechanisms, this finding doesn’t make intuitive sense. So we need to make sure that, when this happens, we look to the next studies and try to understand, was there any other confounder that may have resulted in these findings that was not adequately adjusted for or addressed in some other way?
“When you have a study of this size, you argue, ‘Because it’s so large, any effect that was seen must be real,’ ” he added. “Alternatively, to have a study of this size, by its very nature you are limited in being able to control for certain other factors or differences between the groups.”
That said, he commended the authors for their study and acknowledged the potential questions it raises about the single-shot Johnson & Johnson vaccine. “If you only get one and you’re on infliximab, this study implies that maybe that’s not enough,” he said. “Despite the fact that Johnson & Johnson was approved as a single dose, it may be necessary to think about it as the first of two, or maybe it’s not the preferred vaccine in this group of patients.”
The study was supported by the Royal Devon and Exeter and Hull University Hospital Foundation NHS Trusts and unrestricted educational grants from Biogen (Switzerland), Celltrion Healthcare (South Korea), Galapagos NV (Belgium), and F. Hoffmann-La Roche (Switzerland). The authors acknowledged numerous potential conflicts of interest, including receiving grants, personal fees, and nonfinancial support from various pharmaceutical companies.
FROM MEDRXIV
Bimekizumab superior to adalimumab in head-to-head psoriasis study
for treatment of moderate to severe plaque psoriasis in the head-to-head, phase 3 BE SURE trial, Jerry Bagel, MD, said at Innovations in Dermatology: Virtual Spring Conference 2021.
“Results demonstrated that bimekizumab was superior to adalimumab over 16 weeks of treatment in terms of the speed, depth, and durability of skin clearance,” reported Dr. Bagel, a dermatologist at the Psoriasis Center of Central New Jersey, East Windsor.
The Food and Drug Administration is now reviewing UCB’s application for marketing approval of bimekizumab for treatment of moderate to severe psoriasis in adults.
BE SURE was a 478-patient, double-blind, phase 3 trial in which patients were randomized to one of three regimens: 320 mg of bimekizumab every 4 weeks; the tumor necrosis factor blocker adalimumab (Humira) at 40 mg every 2 weeks for 24 weeks, followed by a switch to bimekizumab at 320 mg every 4 weeks; or 320 mg of bimekizumab every 4 weeks for 16 weeks, then ratcheting back to dosing every 8 weeks. The trial concluded at week 56, Dr. Bagel explained at the conference sponsored by MedscapeLIVE! and the producers of the Hawaii Dermatology Seminar and Caribbean Dermatology Symposium.
The two coprimary endpoints were the 16-week rates of a 90% improvement from baseline in Psoriasis Area and Severity Index score, or PASI 90 response, and an Investigator’s Global Assessment (IGA) score of 0 or 1, meaning clear or almost clear. Bimekizumab every 4 weeks bested adalimumab on both endpoints, with a PASI 90 rate of 86.2%, compared with 47.2%, and a IGA 0/1 rate of 85.3% versus 57.2%. The 16-week PASI 100 response rate was 60.8% with bimekizumab and 23.9% with adalimumab.
The response to bimekizumab was notably fast: already by week 4, the PASI 75 rate was 76.4%, compared with 31.4% with adalimumab. And once patients switched from adalimumab to bimekizumab at week 24, their response rates shot up rapidly. Bimekizumab was equally effective whether dosed at 320 mg every 4 weeks or at maintenance dosing every 8 weeks, such that at week 56 patients in all three study arms had PASI 90 rates of 82%-84%.
The most frequent treatment-emergent adverse events associated with bimekizumab were oral candidiasis, nasopharyngitis, and upper respiratory tract infection. The oral candidiasis, which occurred in 13.2% of patients on bimekizumab every 4 weeks, was mainly mild to moderate, localized, and in no instance led to discontinuation of therapy, according to Dr. Bagel.
“Very impressive data,” commented session comoderator Linda Stein Gold, MD. “This study shows some data that’s potentially unprecedented. Bimekizumab was superior to one of the drugs that we know, we’ve used, and know is very, very effective.”
“Note the speed of this drug,” added comoderator Bruce E. Strober, MD, PhD, of Yale University, New Haven, Conn., and Central Connecticut Dermatology, Cromwell, Conn. “It achieved at week 4 the efficacy that it took adalimumab until week 16 to reach. So it is a very fast drug. Bimekizumab will be the fastest drug you’ve ever, ever worked with.”
“You’ll see in the bimekizumab studies about a fivefold increased frequency of oral candidiasis relative to our more legacy IL-17 inhibitors, such as ixekizumab, secukinumab, and brodalumab. I think that means approximately one in five or one in six patients will have some form of candidiasis when you treat them with bimekizumab,” he said. Therefore, he added, “in some patients you’ll have to manage oral candidiasis. Most affected patients don’t leave the studies, so it’s manageable, but you’ll have to become something of an authority on how to treat with, for example, oral antifungal swish-and-swallow, swish-and-spit, or oral fluconazole. And some of these patients will have recurrent infections.”
It’s a prospect that doesn’t concern Dr. Stein Gold. “This is a side effect that we can treat. We can see it, we’re comfortable with it, and it’s certainly something we can get a handle on,” said Dr. Stein Gold, director of dermatology clinical research at the Henry Ford Health System in Detroit.
BE SURE was funded by UCB. Dr. Bagel reported serving as a speaker for, consultant to, and paid investigator for AbbVie, Celgene, Eli Lilly, Leo Pharma, Novartis, and Ortho Pharmaceuticals. Dr. Stein Gold and Dr. Strober reported having financial relationships with numerous pharmaceutical companies.
MedscapeLIVE! and this news organization are owned by the same parent company.
for treatment of moderate to severe plaque psoriasis in the head-to-head, phase 3 BE SURE trial, Jerry Bagel, MD, said at Innovations in Dermatology: Virtual Spring Conference 2021.
“Results demonstrated that bimekizumab was superior to adalimumab over 16 weeks of treatment in terms of the speed, depth, and durability of skin clearance,” reported Dr. Bagel, a dermatologist at the Psoriasis Center of Central New Jersey, East Windsor.
The Food and Drug Administration is now reviewing UCB’s application for marketing approval of bimekizumab for treatment of moderate to severe psoriasis in adults.
BE SURE was a 478-patient, double-blind, phase 3 trial in which patients were randomized to one of three regimens: 320 mg of bimekizumab every 4 weeks; the tumor necrosis factor blocker adalimumab (Humira) at 40 mg every 2 weeks for 24 weeks, followed by a switch to bimekizumab at 320 mg every 4 weeks; or 320 mg of bimekizumab every 4 weeks for 16 weeks, then ratcheting back to dosing every 8 weeks. The trial concluded at week 56, Dr. Bagel explained at the conference sponsored by MedscapeLIVE! and the producers of the Hawaii Dermatology Seminar and Caribbean Dermatology Symposium.
The two coprimary endpoints were the 16-week rates of a 90% improvement from baseline in Psoriasis Area and Severity Index score, or PASI 90 response, and an Investigator’s Global Assessment (IGA) score of 0 or 1, meaning clear or almost clear. Bimekizumab every 4 weeks bested adalimumab on both endpoints, with a PASI 90 rate of 86.2%, compared with 47.2%, and a IGA 0/1 rate of 85.3% versus 57.2%. The 16-week PASI 100 response rate was 60.8% with bimekizumab and 23.9% with adalimumab.
The response to bimekizumab was notably fast: already by week 4, the PASI 75 rate was 76.4%, compared with 31.4% with adalimumab. And once patients switched from adalimumab to bimekizumab at week 24, their response rates shot up rapidly. Bimekizumab was equally effective whether dosed at 320 mg every 4 weeks or at maintenance dosing every 8 weeks, such that at week 56 patients in all three study arms had PASI 90 rates of 82%-84%.
The most frequent treatment-emergent adverse events associated with bimekizumab were oral candidiasis, nasopharyngitis, and upper respiratory tract infection. The oral candidiasis, which occurred in 13.2% of patients on bimekizumab every 4 weeks, was mainly mild to moderate, localized, and in no instance led to discontinuation of therapy, according to Dr. Bagel.
“Very impressive data,” commented session comoderator Linda Stein Gold, MD. “This study shows some data that’s potentially unprecedented. Bimekizumab was superior to one of the drugs that we know, we’ve used, and know is very, very effective.”
“Note the speed of this drug,” added comoderator Bruce E. Strober, MD, PhD, of Yale University, New Haven, Conn., and Central Connecticut Dermatology, Cromwell, Conn. “It achieved at week 4 the efficacy that it took adalimumab until week 16 to reach. So it is a very fast drug. Bimekizumab will be the fastest drug you’ve ever, ever worked with.”
“You’ll see in the bimekizumab studies about a fivefold increased frequency of oral candidiasis relative to our more legacy IL-17 inhibitors, such as ixekizumab, secukinumab, and brodalumab. I think that means approximately one in five or one in six patients will have some form of candidiasis when you treat them with bimekizumab,” he said. Therefore, he added, “in some patients you’ll have to manage oral candidiasis. Most affected patients don’t leave the studies, so it’s manageable, but you’ll have to become something of an authority on how to treat with, for example, oral antifungal swish-and-swallow, swish-and-spit, or oral fluconazole. And some of these patients will have recurrent infections.”
It’s a prospect that doesn’t concern Dr. Stein Gold. “This is a side effect that we can treat. We can see it, we’re comfortable with it, and it’s certainly something we can get a handle on,” said Dr. Stein Gold, director of dermatology clinical research at the Henry Ford Health System in Detroit.
BE SURE was funded by UCB. Dr. Bagel reported serving as a speaker for, consultant to, and paid investigator for AbbVie, Celgene, Eli Lilly, Leo Pharma, Novartis, and Ortho Pharmaceuticals. Dr. Stein Gold and Dr. Strober reported having financial relationships with numerous pharmaceutical companies.
MedscapeLIVE! and this news organization are owned by the same parent company.
for treatment of moderate to severe plaque psoriasis in the head-to-head, phase 3 BE SURE trial, Jerry Bagel, MD, said at Innovations in Dermatology: Virtual Spring Conference 2021.
“Results demonstrated that bimekizumab was superior to adalimumab over 16 weeks of treatment in terms of the speed, depth, and durability of skin clearance,” reported Dr. Bagel, a dermatologist at the Psoriasis Center of Central New Jersey, East Windsor.
The Food and Drug Administration is now reviewing UCB’s application for marketing approval of bimekizumab for treatment of moderate to severe psoriasis in adults.
BE SURE was a 478-patient, double-blind, phase 3 trial in which patients were randomized to one of three regimens: 320 mg of bimekizumab every 4 weeks; the tumor necrosis factor blocker adalimumab (Humira) at 40 mg every 2 weeks for 24 weeks, followed by a switch to bimekizumab at 320 mg every 4 weeks; or 320 mg of bimekizumab every 4 weeks for 16 weeks, then ratcheting back to dosing every 8 weeks. The trial concluded at week 56, Dr. Bagel explained at the conference sponsored by MedscapeLIVE! and the producers of the Hawaii Dermatology Seminar and Caribbean Dermatology Symposium.
The two coprimary endpoints were the 16-week rates of a 90% improvement from baseline in Psoriasis Area and Severity Index score, or PASI 90 response, and an Investigator’s Global Assessment (IGA) score of 0 or 1, meaning clear or almost clear. Bimekizumab every 4 weeks bested adalimumab on both endpoints, with a PASI 90 rate of 86.2%, compared with 47.2%, and a IGA 0/1 rate of 85.3% versus 57.2%. The 16-week PASI 100 response rate was 60.8% with bimekizumab and 23.9% with adalimumab.
The response to bimekizumab was notably fast: already by week 4, the PASI 75 rate was 76.4%, compared with 31.4% with adalimumab. And once patients switched from adalimumab to bimekizumab at week 24, their response rates shot up rapidly. Bimekizumab was equally effective whether dosed at 320 mg every 4 weeks or at maintenance dosing every 8 weeks, such that at week 56 patients in all three study arms had PASI 90 rates of 82%-84%.
The most frequent treatment-emergent adverse events associated with bimekizumab were oral candidiasis, nasopharyngitis, and upper respiratory tract infection. The oral candidiasis, which occurred in 13.2% of patients on bimekizumab every 4 weeks, was mainly mild to moderate, localized, and in no instance led to discontinuation of therapy, according to Dr. Bagel.
“Very impressive data,” commented session comoderator Linda Stein Gold, MD. “This study shows some data that’s potentially unprecedented. Bimekizumab was superior to one of the drugs that we know, we’ve used, and know is very, very effective.”
“Note the speed of this drug,” added comoderator Bruce E. Strober, MD, PhD, of Yale University, New Haven, Conn., and Central Connecticut Dermatology, Cromwell, Conn. “It achieved at week 4 the efficacy that it took adalimumab until week 16 to reach. So it is a very fast drug. Bimekizumab will be the fastest drug you’ve ever, ever worked with.”
“You’ll see in the bimekizumab studies about a fivefold increased frequency of oral candidiasis relative to our more legacy IL-17 inhibitors, such as ixekizumab, secukinumab, and brodalumab. I think that means approximately one in five or one in six patients will have some form of candidiasis when you treat them with bimekizumab,” he said. Therefore, he added, “in some patients you’ll have to manage oral candidiasis. Most affected patients don’t leave the studies, so it’s manageable, but you’ll have to become something of an authority on how to treat with, for example, oral antifungal swish-and-swallow, swish-and-spit, or oral fluconazole. And some of these patients will have recurrent infections.”
It’s a prospect that doesn’t concern Dr. Stein Gold. “This is a side effect that we can treat. We can see it, we’re comfortable with it, and it’s certainly something we can get a handle on,” said Dr. Stein Gold, director of dermatology clinical research at the Henry Ford Health System in Detroit.
BE SURE was funded by UCB. Dr. Bagel reported serving as a speaker for, consultant to, and paid investigator for AbbVie, Celgene, Eli Lilly, Leo Pharma, Novartis, and Ortho Pharmaceuticals. Dr. Stein Gold and Dr. Strober reported having financial relationships with numerous pharmaceutical companies.
MedscapeLIVE! and this news organization are owned by the same parent company.
FROM INNOVATIONS IN DERMATOLOGY
Clinical Edge Journal Scan Commentary: PsA April 2021
Psoriatic arthritis may be associated with excess mortality risk but the current evidence to support excess mortality in psoriatic arthritis is inconclusive. Nevertheless, identifying risk factors for mortality in patients with psoriatic arthritis is important so that modifiable factors could be addressed. Vela et al investigated whether the cumulative pain experienced by psoriatic arthritis patients was associated with increased mortality. Using data from the Danish nationwide registry of biological therapies (DANBIO), the authors showed that although there was a significant association between pain intensity and mortality (odds ratio 1.06 (95%CI 1.02 to 1.10) per 5 VAS unit increase), no association was found when the analyses was adjusted for confounders including age, CRP, joint counts, HAQ score, treatment, and comorbidities. As expected, recent glucocorticoid use, chronic obstructive pulmonary disease, diabetes mellitus, cancer and cardiovascular disease were associated with excess mortality. The results indicate that mortality in psoriatic arthritis is primarily driven by associated comorbidities; holistic management of psoriatic disease should include management of associated comorbidities.
Comorbidities have a major impact on patients with psoriatic arthritis and influence a patient’s quality of life and function as well as treatment response. Neuropsychiatric comorbidities have been less studied in psoriatic arthritis. An intriguing study by Garcia et al indicate that cognitive impairment may be associated with psoriatic arthritis. In a small cross-sectional study, they demonstrated that patients with psoriatic arthritis score worse on the Montreal Cognitive Assessment (MoCA) tool compared to controls, with executive skills, naming, language, and abstraction being most affected. Further research is required to explore whether other comorbidities such as cerebrovascular disease, depression or sleep disturbances explain the cognitive impairment. Depression and anxiety are also associated with reduced likelihood of achieving remission or low disease activity state in psoriatic arthritis. In a study of 743 patients, Wong et al demonstrated that patients with depression or anxiety are less likely to achieve a state of sustained minimal disease activity. These studies once again highlight the management of comorbidities to achieve improved outcomes in patients with psoriatic arthritis.
New data also indicate efficacy of targeted therapy in psoriatic arthritis. In two phase 3 trials funded by LEO pharma, Mease et al report that brodalumab, an interleukin 17 receptor inhibitor that is already available for the treatment for psoriasis, is efficacious in the treatment of psoriatic arthritis. Although the trial was terminated early, pooled data from the two trials showed that higher proportions of patients on 140 mg and 210 mg of brodalumab achieved American College of Rheumatology (ACR)20 response at week 16 compared to placebo group (45.8% and 47.9%, respectively vs. 20.9%; P less than .0001).
There are limited data on treatment withdrawal in patients with psoriatic arthritis. Coates et al investigated whether continued treatment with ixekizumab, an interleukin 17A inhibitor was superior to withdrawing ixekizumab in maintaining minimal disease activity state in patents with psoriatic arthritis. They showed that more patients relapsed rapidly after ixekizumab was withdrawn compared to those continuing ixekizumab treatment. Importantly, >95% of patients who relapsed with treatment withdrawal re-achieved MDA on retreatment with ixekizumab within a median duration of 4.1 weeks. Thus, ixekizumab treatment is best maintained after a patient achieves a state of minimal disease activity. If the treatment needs to be interrupted (e.g., infection, surgery), most patients will re-achieve the state of minimal disease activity on retreatment.
Finally, research continues to demonstrate delayed diagnosis of psoriatic arthritis. Karmacharya et al showed that only 45% of patients receive a diagnosis of psoriatic arthritis by 2 years after symptom onset. This study from the population-based Rochester Epidemiology Project indicates that earlier age of onset of symptoms, the presence of higher body mass index and enthesitis are associated with diagnostic delay. Further education of health care providers and patients with psoriasis about psoriatic arthritis may help reduce diagnostic delay; delayed diagnosis leads to poorer long-term outcomes.
Psoriatic arthritis may be associated with excess mortality risk but the current evidence to support excess mortality in psoriatic arthritis is inconclusive. Nevertheless, identifying risk factors for mortality in patients with psoriatic arthritis is important so that modifiable factors could be addressed. Vela et al investigated whether the cumulative pain experienced by psoriatic arthritis patients was associated with increased mortality. Using data from the Danish nationwide registry of biological therapies (DANBIO), the authors showed that although there was a significant association between pain intensity and mortality (odds ratio 1.06 (95%CI 1.02 to 1.10) per 5 VAS unit increase), no association was found when the analyses was adjusted for confounders including age, CRP, joint counts, HAQ score, treatment, and comorbidities. As expected, recent glucocorticoid use, chronic obstructive pulmonary disease, diabetes mellitus, cancer and cardiovascular disease were associated with excess mortality. The results indicate that mortality in psoriatic arthritis is primarily driven by associated comorbidities; holistic management of psoriatic disease should include management of associated comorbidities.
Comorbidities have a major impact on patients with psoriatic arthritis and influence a patient’s quality of life and function as well as treatment response. Neuropsychiatric comorbidities have been less studied in psoriatic arthritis. An intriguing study by Garcia et al indicate that cognitive impairment may be associated with psoriatic arthritis. In a small cross-sectional study, they demonstrated that patients with psoriatic arthritis score worse on the Montreal Cognitive Assessment (MoCA) tool compared to controls, with executive skills, naming, language, and abstraction being most affected. Further research is required to explore whether other comorbidities such as cerebrovascular disease, depression or sleep disturbances explain the cognitive impairment. Depression and anxiety are also associated with reduced likelihood of achieving remission or low disease activity state in psoriatic arthritis. In a study of 743 patients, Wong et al demonstrated that patients with depression or anxiety are less likely to achieve a state of sustained minimal disease activity. These studies once again highlight the management of comorbidities to achieve improved outcomes in patients with psoriatic arthritis.
New data also indicate efficacy of targeted therapy in psoriatic arthritis. In two phase 3 trials funded by LEO pharma, Mease et al report that brodalumab, an interleukin 17 receptor inhibitor that is already available for the treatment for psoriasis, is efficacious in the treatment of psoriatic arthritis. Although the trial was terminated early, pooled data from the two trials showed that higher proportions of patients on 140 mg and 210 mg of brodalumab achieved American College of Rheumatology (ACR)20 response at week 16 compared to placebo group (45.8% and 47.9%, respectively vs. 20.9%; P less than .0001).
There are limited data on treatment withdrawal in patients with psoriatic arthritis. Coates et al investigated whether continued treatment with ixekizumab, an interleukin 17A inhibitor was superior to withdrawing ixekizumab in maintaining minimal disease activity state in patents with psoriatic arthritis. They showed that more patients relapsed rapidly after ixekizumab was withdrawn compared to those continuing ixekizumab treatment. Importantly, >95% of patients who relapsed with treatment withdrawal re-achieved MDA on retreatment with ixekizumab within a median duration of 4.1 weeks. Thus, ixekizumab treatment is best maintained after a patient achieves a state of minimal disease activity. If the treatment needs to be interrupted (e.g., infection, surgery), most patients will re-achieve the state of minimal disease activity on retreatment.
Finally, research continues to demonstrate delayed diagnosis of psoriatic arthritis. Karmacharya et al showed that only 45% of patients receive a diagnosis of psoriatic arthritis by 2 years after symptom onset. This study from the population-based Rochester Epidemiology Project indicates that earlier age of onset of symptoms, the presence of higher body mass index and enthesitis are associated with diagnostic delay. Further education of health care providers and patients with psoriasis about psoriatic arthritis may help reduce diagnostic delay; delayed diagnosis leads to poorer long-term outcomes.
Psoriatic arthritis may be associated with excess mortality risk but the current evidence to support excess mortality in psoriatic arthritis is inconclusive. Nevertheless, identifying risk factors for mortality in patients with psoriatic arthritis is important so that modifiable factors could be addressed. Vela et al investigated whether the cumulative pain experienced by psoriatic arthritis patients was associated with increased mortality. Using data from the Danish nationwide registry of biological therapies (DANBIO), the authors showed that although there was a significant association between pain intensity and mortality (odds ratio 1.06 (95%CI 1.02 to 1.10) per 5 VAS unit increase), no association was found when the analyses was adjusted for confounders including age, CRP, joint counts, HAQ score, treatment, and comorbidities. As expected, recent glucocorticoid use, chronic obstructive pulmonary disease, diabetes mellitus, cancer and cardiovascular disease were associated with excess mortality. The results indicate that mortality in psoriatic arthritis is primarily driven by associated comorbidities; holistic management of psoriatic disease should include management of associated comorbidities.
Comorbidities have a major impact on patients with psoriatic arthritis and influence a patient’s quality of life and function as well as treatment response. Neuropsychiatric comorbidities have been less studied in psoriatic arthritis. An intriguing study by Garcia et al indicate that cognitive impairment may be associated with psoriatic arthritis. In a small cross-sectional study, they demonstrated that patients with psoriatic arthritis score worse on the Montreal Cognitive Assessment (MoCA) tool compared to controls, with executive skills, naming, language, and abstraction being most affected. Further research is required to explore whether other comorbidities such as cerebrovascular disease, depression or sleep disturbances explain the cognitive impairment. Depression and anxiety are also associated with reduced likelihood of achieving remission or low disease activity state in psoriatic arthritis. In a study of 743 patients, Wong et al demonstrated that patients with depression or anxiety are less likely to achieve a state of sustained minimal disease activity. These studies once again highlight the management of comorbidities to achieve improved outcomes in patients with psoriatic arthritis.
New data also indicate efficacy of targeted therapy in psoriatic arthritis. In two phase 3 trials funded by LEO pharma, Mease et al report that brodalumab, an interleukin 17 receptor inhibitor that is already available for the treatment for psoriasis, is efficacious in the treatment of psoriatic arthritis. Although the trial was terminated early, pooled data from the two trials showed that higher proportions of patients on 140 mg and 210 mg of brodalumab achieved American College of Rheumatology (ACR)20 response at week 16 compared to placebo group (45.8% and 47.9%, respectively vs. 20.9%; P less than .0001).
There are limited data on treatment withdrawal in patients with psoriatic arthritis. Coates et al investigated whether continued treatment with ixekizumab, an interleukin 17A inhibitor was superior to withdrawing ixekizumab in maintaining minimal disease activity state in patents with psoriatic arthritis. They showed that more patients relapsed rapidly after ixekizumab was withdrawn compared to those continuing ixekizumab treatment. Importantly, >95% of patients who relapsed with treatment withdrawal re-achieved MDA on retreatment with ixekizumab within a median duration of 4.1 weeks. Thus, ixekizumab treatment is best maintained after a patient achieves a state of minimal disease activity. If the treatment needs to be interrupted (e.g., infection, surgery), most patients will re-achieve the state of minimal disease activity on retreatment.
Finally, research continues to demonstrate delayed diagnosis of psoriatic arthritis. Karmacharya et al showed that only 45% of patients receive a diagnosis of psoriatic arthritis by 2 years after symptom onset. This study from the population-based Rochester Epidemiology Project indicates that earlier age of onset of symptoms, the presence of higher body mass index and enthesitis are associated with diagnostic delay. Further education of health care providers and patients with psoriasis about psoriatic arthritis may help reduce diagnostic delay; delayed diagnosis leads to poorer long-term outcomes.
Over half of PsA patients not diagnosed within 2 years of symptom onset
Key clinical point: More than half of patients with PsA had a diagnostic delay of >2 years.
Major finding: The percentage of patients receiving a diagnosis of PsA by 2 years after symptom onset was 45%. Factors associated with a diagnostic delay of >2 years included earlier age at onset of symptoms, higher body mass index, and enthesitis.
Study details: The data come from a retrospective, population-based cohort of 164 incident adult PsA patients.
Disclosures: The study used resources of the Rochester Epidemiology Project, which is supported by various National Institutes of Health grants. P Karmacharya and A Duarte-García reported receiving individual research grants. A Ogdie and JM Davis reported ties with various pharmaceutical companies. The remaining authors declared no conflicts of interest.
Source: Karmacharya P et al. J Rheumatol. 2021 Feb 15. doi: 10.3899/jrheum.201199.
Key clinical point: More than half of patients with PsA had a diagnostic delay of >2 years.
Major finding: The percentage of patients receiving a diagnosis of PsA by 2 years after symptom onset was 45%. Factors associated with a diagnostic delay of >2 years included earlier age at onset of symptoms, higher body mass index, and enthesitis.
Study details: The data come from a retrospective, population-based cohort of 164 incident adult PsA patients.
Disclosures: The study used resources of the Rochester Epidemiology Project, which is supported by various National Institutes of Health grants. P Karmacharya and A Duarte-García reported receiving individual research grants. A Ogdie and JM Davis reported ties with various pharmaceutical companies. The remaining authors declared no conflicts of interest.
Source: Karmacharya P et al. J Rheumatol. 2021 Feb 15. doi: 10.3899/jrheum.201199.
Key clinical point: More than half of patients with PsA had a diagnostic delay of >2 years.
Major finding: The percentage of patients receiving a diagnosis of PsA by 2 years after symptom onset was 45%. Factors associated with a diagnostic delay of >2 years included earlier age at onset of symptoms, higher body mass index, and enthesitis.
Study details: The data come from a retrospective, population-based cohort of 164 incident adult PsA patients.
Disclosures: The study used resources of the Rochester Epidemiology Project, which is supported by various National Institutes of Health grants. P Karmacharya and A Duarte-García reported receiving individual research grants. A Ogdie and JM Davis reported ties with various pharmaceutical companies. The remaining authors declared no conflicts of interest.
Source: Karmacharya P et al. J Rheumatol. 2021 Feb 15. doi: 10.3899/jrheum.201199.
Agreement between modalities for imaging of finger joints in PsA patients
Key clinical point: There is favorable agreement between ultrasound (US) and magnetic resonance imaging (MRI) for detecting inflammatory changes in finger joints of patients with psoriatic arthritis (PsA). Additionally, there is a favorable agreement between US, radiography and MRI for detecting destructive changes.
Major finding: The absolute agreement between US and MRI was in the range of good-to-very good for detecting synovitis (85%-96%), flexor tenosynovitis (93%-98%), and extensor paratenonitis (95%-98%). Agreement between US, MRI and radiography was also good-to-very good for detecting erosions (96%-98%) and bone proliferations (71%-93%).
Study details: The data come from a study of 100 consecutive PsA patients who underwent clinical assessment and concomitant radiographic, US and MRI evaluations of the metacarpophalangeal, proximal interphalangeal and distal interphalangeal joints of one hand.
Disclosures: The study was supported by an investigator-initiated research grant sponsored by Johnson and Johnson. The authors declared no conflicts of interest.
Source: Polachek A et al. Rheumatology (Oxford). 2021 Mar 17. doi: 10.1093/rheumatology/keab272.
Key clinical point: There is favorable agreement between ultrasound (US) and magnetic resonance imaging (MRI) for detecting inflammatory changes in finger joints of patients with psoriatic arthritis (PsA). Additionally, there is a favorable agreement between US, radiography and MRI for detecting destructive changes.
Major finding: The absolute agreement between US and MRI was in the range of good-to-very good for detecting synovitis (85%-96%), flexor tenosynovitis (93%-98%), and extensor paratenonitis (95%-98%). Agreement between US, MRI and radiography was also good-to-very good for detecting erosions (96%-98%) and bone proliferations (71%-93%).
Study details: The data come from a study of 100 consecutive PsA patients who underwent clinical assessment and concomitant radiographic, US and MRI evaluations of the metacarpophalangeal, proximal interphalangeal and distal interphalangeal joints of one hand.
Disclosures: The study was supported by an investigator-initiated research grant sponsored by Johnson and Johnson. The authors declared no conflicts of interest.
Source: Polachek A et al. Rheumatology (Oxford). 2021 Mar 17. doi: 10.1093/rheumatology/keab272.
Key clinical point: There is favorable agreement between ultrasound (US) and magnetic resonance imaging (MRI) for detecting inflammatory changes in finger joints of patients with psoriatic arthritis (PsA). Additionally, there is a favorable agreement between US, radiography and MRI for detecting destructive changes.
Major finding: The absolute agreement between US and MRI was in the range of good-to-very good for detecting synovitis (85%-96%), flexor tenosynovitis (93%-98%), and extensor paratenonitis (95%-98%). Agreement between US, MRI and radiography was also good-to-very good for detecting erosions (96%-98%) and bone proliferations (71%-93%).
Study details: The data come from a study of 100 consecutive PsA patients who underwent clinical assessment and concomitant radiographic, US and MRI evaluations of the metacarpophalangeal, proximal interphalangeal and distal interphalangeal joints of one hand.
Disclosures: The study was supported by an investigator-initiated research grant sponsored by Johnson and Johnson. The authors declared no conflicts of interest.
Source: Polachek A et al. Rheumatology (Oxford). 2021 Mar 17. doi: 10.1093/rheumatology/keab272.
Cognitive impairment is a manifestation of PsA
Key clinical point: Cognitive impairment may be one of the neurological manifestations of psoriatic arthritis (PsA).
Major finding: Patients with PsA scored worse on the Montreal Cognitive Assessment (MoCA) vs. controls (P = .01). Additionally, the proportion of patients with cognitive impairment according to MoCA was significantly higher among cases vs. controls (91.9% vs. 58.3%, P = .002).
Study details: The data come from a cross-sectional case-control study involving 37 patients with PsA and 36 healthy controls.
Disclosures: The study did not receive any funding. The authors declared no conflicts of interest.
Source: Garcia LOKL et al. Acta Neurol Belg. 2021 Mar 13. doi: 10.1007/s13760-021-01644-y.
Key clinical point: Cognitive impairment may be one of the neurological manifestations of psoriatic arthritis (PsA).
Major finding: Patients with PsA scored worse on the Montreal Cognitive Assessment (MoCA) vs. controls (P = .01). Additionally, the proportion of patients with cognitive impairment according to MoCA was significantly higher among cases vs. controls (91.9% vs. 58.3%, P = .002).
Study details: The data come from a cross-sectional case-control study involving 37 patients with PsA and 36 healthy controls.
Disclosures: The study did not receive any funding. The authors declared no conflicts of interest.
Source: Garcia LOKL et al. Acta Neurol Belg. 2021 Mar 13. doi: 10.1007/s13760-021-01644-y.
Key clinical point: Cognitive impairment may be one of the neurological manifestations of psoriatic arthritis (PsA).
Major finding: Patients with PsA scored worse on the Montreal Cognitive Assessment (MoCA) vs. controls (P = .01). Additionally, the proportion of patients with cognitive impairment according to MoCA was significantly higher among cases vs. controls (91.9% vs. 58.3%, P = .002).
Study details: The data come from a cross-sectional case-control study involving 37 patients with PsA and 36 healthy controls.
Disclosures: The study did not receive any funding. The authors declared no conflicts of interest.
Source: Garcia LOKL et al. Acta Neurol Belg. 2021 Mar 13. doi: 10.1007/s13760-021-01644-y.
Intravenous golimumab improves HRQoL and productivity in patients with active PsA
Key clinical point: In patients with active psoriatic arthritis (PsA), intravenous (IV) golimumab improved health-related quality of life (HRQoL) and productivity as early as 8 weeks and maintained improvement through 1 year; and these improvements were associated with improvements in disease activity and patient functional capability outcomes.
Major finding: At week 8, patients receiving IV golimumab vs. placebo had greater improvements in EuroQol-5 dimension-5 level index (0.14 vs. 0.04) and visual analog scale (VAS; 17.16 vs. 3.69), daily productivity VAS (−2.91 vs. −0.71), and Work Limitations Questionnaire productivity loss score (−2.92 vs. −0.78). At week 52, improvements were similar in the golimumab and placebo-crossover groups. HRQoL and productivity correlated with disease activity and functional capability, with continued association from week 8 through week 52.
Study details: In this phase 3 GO-VIBRANT trial, 480 patients with PsA were randomly assigned to receive IV golimumab 2 mg/kg (n=241) at weeks 0, 4, and then every 8 weeks (q8w) through week 52 or placebo (n=239) at weeks 0, 4, and then q8w, with crossover to IV golimumab 2 mg/kg at weeks 24, 28, and then q8w through week 52.
Disclosures: This study was supported by Janssen Research & Development, LLC, Spring House, PA. Some study investigators are employees of Janssen Global Services, LLC and own stock in Johnson & Johnson, of which Janssen Global Services, LLC is a wholly owned subsidiary. Some study authors reported receiving support from and consulting for Janssen.
Source: Ogdie A et al. Clin Rheumatol. 2021 Mar 2. doi: 10.1007/s10067-021-05639-1.
Key clinical point: In patients with active psoriatic arthritis (PsA), intravenous (IV) golimumab improved health-related quality of life (HRQoL) and productivity as early as 8 weeks and maintained improvement through 1 year; and these improvements were associated with improvements in disease activity and patient functional capability outcomes.
Major finding: At week 8, patients receiving IV golimumab vs. placebo had greater improvements in EuroQol-5 dimension-5 level index (0.14 vs. 0.04) and visual analog scale (VAS; 17.16 vs. 3.69), daily productivity VAS (−2.91 vs. −0.71), and Work Limitations Questionnaire productivity loss score (−2.92 vs. −0.78). At week 52, improvements were similar in the golimumab and placebo-crossover groups. HRQoL and productivity correlated with disease activity and functional capability, with continued association from week 8 through week 52.
Study details: In this phase 3 GO-VIBRANT trial, 480 patients with PsA were randomly assigned to receive IV golimumab 2 mg/kg (n=241) at weeks 0, 4, and then every 8 weeks (q8w) through week 52 or placebo (n=239) at weeks 0, 4, and then q8w, with crossover to IV golimumab 2 mg/kg at weeks 24, 28, and then q8w through week 52.
Disclosures: This study was supported by Janssen Research & Development, LLC, Spring House, PA. Some study investigators are employees of Janssen Global Services, LLC and own stock in Johnson & Johnson, of which Janssen Global Services, LLC is a wholly owned subsidiary. Some study authors reported receiving support from and consulting for Janssen.
Source: Ogdie A et al. Clin Rheumatol. 2021 Mar 2. doi: 10.1007/s10067-021-05639-1.
Key clinical point: In patients with active psoriatic arthritis (PsA), intravenous (IV) golimumab improved health-related quality of life (HRQoL) and productivity as early as 8 weeks and maintained improvement through 1 year; and these improvements were associated with improvements in disease activity and patient functional capability outcomes.
Major finding: At week 8, patients receiving IV golimumab vs. placebo had greater improvements in EuroQol-5 dimension-5 level index (0.14 vs. 0.04) and visual analog scale (VAS; 17.16 vs. 3.69), daily productivity VAS (−2.91 vs. −0.71), and Work Limitations Questionnaire productivity loss score (−2.92 vs. −0.78). At week 52, improvements were similar in the golimumab and placebo-crossover groups. HRQoL and productivity correlated with disease activity and functional capability, with continued association from week 8 through week 52.
Study details: In this phase 3 GO-VIBRANT trial, 480 patients with PsA were randomly assigned to receive IV golimumab 2 mg/kg (n=241) at weeks 0, 4, and then every 8 weeks (q8w) through week 52 or placebo (n=239) at weeks 0, 4, and then q8w, with crossover to IV golimumab 2 mg/kg at weeks 24, 28, and then q8w through week 52.
Disclosures: This study was supported by Janssen Research & Development, LLC, Spring House, PA. Some study investigators are employees of Janssen Global Services, LLC and own stock in Johnson & Johnson, of which Janssen Global Services, LLC is a wholly owned subsidiary. Some study authors reported receiving support from and consulting for Janssen.
Source: Ogdie A et al. Clin Rheumatol. 2021 Mar 2. doi: 10.1007/s10067-021-05639-1.
Depression and anxiety linked to reduced probability of achieving sustained MDA in PsA
Key clinical point: The presence of depression/anxiety symptoms reduces the probability of achieving sustained minimal disease activity (MDA) in patients with psoriatic arthritis (PsA), regardless of the method used to define depression/anxiety.
Major finding: When depression/anxiety was defined as a score of 38 or lower on the Mental Component Summary of the Short Form-36 questionnaire (definition 1), the odds ratio (OR) for reaching sustained MDA was 0.30 (P less than .0001). The OR values were 0.34 (P less than .0001) and 0.47 (P less than .0001) for a score of 56 or lower on the Mental Health sub-scale (definition 2) and for rheumatologist’s report of a diagnosis or treatment for depression/anxiety (definition 3), respectively.
Study details: The data come from a study of 743 patients with PsA.
Disclosures: No study sponsor was identified. Several of the authors are affiliated with the Psoriatic Disease Program of the Krembil Research Institute of University Health Network in Toronto, which is supported by the Krembil Foundation. Dr. A Wong was supported by a Krembil Psoriatic Arthritis Fellowship and Dr. V Chandran was supported by a Pfizer Chair Rheumatology Research Award from the Department of Medicine, University of Toronto.
Source: Wong A et al. Arthritis Care Res (Hoboken). 2021 Mar 4. doi: 10.1002/acr.24593.
Key clinical point: The presence of depression/anxiety symptoms reduces the probability of achieving sustained minimal disease activity (MDA) in patients with psoriatic arthritis (PsA), regardless of the method used to define depression/anxiety.
Major finding: When depression/anxiety was defined as a score of 38 or lower on the Mental Component Summary of the Short Form-36 questionnaire (definition 1), the odds ratio (OR) for reaching sustained MDA was 0.30 (P less than .0001). The OR values were 0.34 (P less than .0001) and 0.47 (P less than .0001) for a score of 56 or lower on the Mental Health sub-scale (definition 2) and for rheumatologist’s report of a diagnosis or treatment for depression/anxiety (definition 3), respectively.
Study details: The data come from a study of 743 patients with PsA.
Disclosures: No study sponsor was identified. Several of the authors are affiliated with the Psoriatic Disease Program of the Krembil Research Institute of University Health Network in Toronto, which is supported by the Krembil Foundation. Dr. A Wong was supported by a Krembil Psoriatic Arthritis Fellowship and Dr. V Chandran was supported by a Pfizer Chair Rheumatology Research Award from the Department of Medicine, University of Toronto.
Source: Wong A et al. Arthritis Care Res (Hoboken). 2021 Mar 4. doi: 10.1002/acr.24593.
Key clinical point: The presence of depression/anxiety symptoms reduces the probability of achieving sustained minimal disease activity (MDA) in patients with psoriatic arthritis (PsA), regardless of the method used to define depression/anxiety.
Major finding: When depression/anxiety was defined as a score of 38 or lower on the Mental Component Summary of the Short Form-36 questionnaire (definition 1), the odds ratio (OR) for reaching sustained MDA was 0.30 (P less than .0001). The OR values were 0.34 (P less than .0001) and 0.47 (P less than .0001) for a score of 56 or lower on the Mental Health sub-scale (definition 2) and for rheumatologist’s report of a diagnosis or treatment for depression/anxiety (definition 3), respectively.
Study details: The data come from a study of 743 patients with PsA.
Disclosures: No study sponsor was identified. Several of the authors are affiliated with the Psoriatic Disease Program of the Krembil Research Institute of University Health Network in Toronto, which is supported by the Krembil Foundation. Dr. A Wong was supported by a Krembil Psoriatic Arthritis Fellowship and Dr. V Chandran was supported by a Pfizer Chair Rheumatology Research Award from the Department of Medicine, University of Toronto.
Source: Wong A et al. Arthritis Care Res (Hoboken). 2021 Mar 4. doi: 10.1002/acr.24593.
Risk factors for progression from subclinical to clinical psoriatic arthritis
Key clinical point: Alcohol consumption, older age, suffering from fatty liver when less than 45 years old, and elevated high-sensitivity C-reactive protein (hs-CRP) level appear to increase the risk of transition from subclinical to clinical psoriatic arthritis (PsA).
Major finding: Older age (greater than 45 years; odds ratio [OR], 10.15; P = 0.00), alcohol consumption (OR, 3.43; P = .03), and elevated hs-CRP (OR, 1.05; P = 0.03) were associated with an increased risk of progression from subclinical PsA to clinical PsA. For patients aged less than 45 years old, the association between fatty liver and clinical PsA was statistically significant.
Study details: The data come from a retrospective case-control study of 25 patients with clinically confirmed PsA (cases) and 137 controls without confirmed PsA.
Disclosures: This study was funded by the 1.3.5 Project for Disciplines of Excellence, West China Hospital, Sichuan University, Chengdu, Sichuan, China and West China Precision Medicine Industrial Technology Institutes. The authors declared no conflicts of interest.
Source: Wang Y et al. Rheumatol Ther. 2021 Mar 5. doi: 10.1007/s40744-021-00295-y.
Key clinical point: Alcohol consumption, older age, suffering from fatty liver when less than 45 years old, and elevated high-sensitivity C-reactive protein (hs-CRP) level appear to increase the risk of transition from subclinical to clinical psoriatic arthritis (PsA).
Major finding: Older age (greater than 45 years; odds ratio [OR], 10.15; P = 0.00), alcohol consumption (OR, 3.43; P = .03), and elevated hs-CRP (OR, 1.05; P = 0.03) were associated with an increased risk of progression from subclinical PsA to clinical PsA. For patients aged less than 45 years old, the association between fatty liver and clinical PsA was statistically significant.
Study details: The data come from a retrospective case-control study of 25 patients with clinically confirmed PsA (cases) and 137 controls without confirmed PsA.
Disclosures: This study was funded by the 1.3.5 Project for Disciplines of Excellence, West China Hospital, Sichuan University, Chengdu, Sichuan, China and West China Precision Medicine Industrial Technology Institutes. The authors declared no conflicts of interest.
Source: Wang Y et al. Rheumatol Ther. 2021 Mar 5. doi: 10.1007/s40744-021-00295-y.
Key clinical point: Alcohol consumption, older age, suffering from fatty liver when less than 45 years old, and elevated high-sensitivity C-reactive protein (hs-CRP) level appear to increase the risk of transition from subclinical to clinical psoriatic arthritis (PsA).
Major finding: Older age (greater than 45 years; odds ratio [OR], 10.15; P = 0.00), alcohol consumption (OR, 3.43; P = .03), and elevated hs-CRP (OR, 1.05; P = 0.03) were associated with an increased risk of progression from subclinical PsA to clinical PsA. For patients aged less than 45 years old, the association between fatty liver and clinical PsA was statistically significant.
Study details: The data come from a retrospective case-control study of 25 patients with clinically confirmed PsA (cases) and 137 controls without confirmed PsA.
Disclosures: This study was funded by the 1.3.5 Project for Disciplines of Excellence, West China Hospital, Sichuan University, Chengdu, Sichuan, China and West China Precision Medicine Industrial Technology Institutes. The authors declared no conflicts of interest.
Source: Wang Y et al. Rheumatol Ther. 2021 Mar 5. doi: 10.1007/s40744-021-00295-y.
Continuing ixekizumab superior to withdrawal in stable psoriatic arthritis
Key clinical point: Continued ixekizumab (IXE) therapy was superior to IXE withdrawal in maintaining minimal disease activity (MDA) in biologic-naive patients with psoriatic arthritis (PsA). In case of treatment interruption, re-treatment with IXE following relapse may restore disease control.
Major finding: Patients relapsed more rapidly with IXE withdrawal (median, 22.3 weeks) vs. continued IXE treatment (median was not estimable because less than 50% of patients had relapsed by the end of study period; P less than .0001). The cumulative relapse rate from week 24 to week 104 was higher for the withdrawal vs. continued treatment group (85% vs. 38%; P less than .0001). Median time to re-achieving MDA on retreatment was 4.1 weeks; 64 (95.5%) of 67 patients who relapsed with treatment withdrawal re-achieved MDA on retreatment.
Study details: SPIRIT-P3 was a multicenter, randomized, double-blind withdrawal study of 394 biologic-naive patients with PsA who received open-label IXE (160 mg at week 0, 80 mg every 2 weeks) for 36 weeks. Between weeks 36 and 64, 158 patients who achieved sustained MDA (greater than 3 months) were randomized (1:1) to continue on 80 mg IXE every 2 weeks or placebo until week 104.
Disclosures: This study was funded by Eli Lilly and Company. Some study investigators reported owning stock in, being an employee of, receiving support from, and/or consulting for Eli Lilly and Company.
Source: Coates LC et al. Arthritis Rheumatol. 2021 Mar 7. doi: 10.1002/art.41716.
Key clinical point: Continued ixekizumab (IXE) therapy was superior to IXE withdrawal in maintaining minimal disease activity (MDA) in biologic-naive patients with psoriatic arthritis (PsA). In case of treatment interruption, re-treatment with IXE following relapse may restore disease control.
Major finding: Patients relapsed more rapidly with IXE withdrawal (median, 22.3 weeks) vs. continued IXE treatment (median was not estimable because less than 50% of patients had relapsed by the end of study period; P less than .0001). The cumulative relapse rate from week 24 to week 104 was higher for the withdrawal vs. continued treatment group (85% vs. 38%; P less than .0001). Median time to re-achieving MDA on retreatment was 4.1 weeks; 64 (95.5%) of 67 patients who relapsed with treatment withdrawal re-achieved MDA on retreatment.
Study details: SPIRIT-P3 was a multicenter, randomized, double-blind withdrawal study of 394 biologic-naive patients with PsA who received open-label IXE (160 mg at week 0, 80 mg every 2 weeks) for 36 weeks. Between weeks 36 and 64, 158 patients who achieved sustained MDA (greater than 3 months) were randomized (1:1) to continue on 80 mg IXE every 2 weeks or placebo until week 104.
Disclosures: This study was funded by Eli Lilly and Company. Some study investigators reported owning stock in, being an employee of, receiving support from, and/or consulting for Eli Lilly and Company.
Source: Coates LC et al. Arthritis Rheumatol. 2021 Mar 7. doi: 10.1002/art.41716.
Key clinical point: Continued ixekizumab (IXE) therapy was superior to IXE withdrawal in maintaining minimal disease activity (MDA) in biologic-naive patients with psoriatic arthritis (PsA). In case of treatment interruption, re-treatment with IXE following relapse may restore disease control.
Major finding: Patients relapsed more rapidly with IXE withdrawal (median, 22.3 weeks) vs. continued IXE treatment (median was not estimable because less than 50% of patients had relapsed by the end of study period; P less than .0001). The cumulative relapse rate from week 24 to week 104 was higher for the withdrawal vs. continued treatment group (85% vs. 38%; P less than .0001). Median time to re-achieving MDA on retreatment was 4.1 weeks; 64 (95.5%) of 67 patients who relapsed with treatment withdrawal re-achieved MDA on retreatment.
Study details: SPIRIT-P3 was a multicenter, randomized, double-blind withdrawal study of 394 biologic-naive patients with PsA who received open-label IXE (160 mg at week 0, 80 mg every 2 weeks) for 36 weeks. Between weeks 36 and 64, 158 patients who achieved sustained MDA (greater than 3 months) were randomized (1:1) to continue on 80 mg IXE every 2 weeks or placebo until week 104.
Disclosures: This study was funded by Eli Lilly and Company. Some study investigators reported owning stock in, being an employee of, receiving support from, and/or consulting for Eli Lilly and Company.
Source: Coates LC et al. Arthritis Rheumatol. 2021 Mar 7. doi: 10.1002/art.41716.