User login
Overprescription of Opioids in Women of Childbearing Age
Despite the risks of birth defects and other complications, opioids are prescribed for > 33% of reproductive-aged women enrolled in Medicaid and 28% of those with private insurance, according to a study by the CDC.
Related: Acute Upper Abdominal Pain in Early Pregnancy
“Many women taking these medicines may not know they are pregnant,” said CDC Director Tom Frieden, MD, MPH. “That’s why it’s critical for health care professionals to take a thorough health assessment before prescribing these medicines to women of reproductive age."
Related: Reducing Opioid Use for Chronic Pain
Studies have suggested that giving opioids to women who are pregnant raises the risks of neural tube defects, congenital heart defects, and gastroschisis, as well as neonatal abstinence syndrome.
Related: Maternal Morbidity: Higher Risks for Minorities
In the CDC study, researchers looked at data from health insurance claims from 2008 to 2012 and from Medicaid. The most frequently prescribed opioids were hydrocodone, codeine, and oxycodone. Prescription rates for women of reproductive age were highest in the South and lowest in the northeastern U.S. Prescriptions were nearly 1.5 times higher for white women compared with that of black or Hispanic women. The consistently higher rate of prescribing to Medicaid-enrolled women is of concern, the researchers say, because about 50% of U.S. births are in that group.
Despite the risks of birth defects and other complications, opioids are prescribed for > 33% of reproductive-aged women enrolled in Medicaid and 28% of those with private insurance, according to a study by the CDC.
Related: Acute Upper Abdominal Pain in Early Pregnancy
“Many women taking these medicines may not know they are pregnant,” said CDC Director Tom Frieden, MD, MPH. “That’s why it’s critical for health care professionals to take a thorough health assessment before prescribing these medicines to women of reproductive age."
Related: Reducing Opioid Use for Chronic Pain
Studies have suggested that giving opioids to women who are pregnant raises the risks of neural tube defects, congenital heart defects, and gastroschisis, as well as neonatal abstinence syndrome.
Related: Maternal Morbidity: Higher Risks for Minorities
In the CDC study, researchers looked at data from health insurance claims from 2008 to 2012 and from Medicaid. The most frequently prescribed opioids were hydrocodone, codeine, and oxycodone. Prescription rates for women of reproductive age were highest in the South and lowest in the northeastern U.S. Prescriptions were nearly 1.5 times higher for white women compared with that of black or Hispanic women. The consistently higher rate of prescribing to Medicaid-enrolled women is of concern, the researchers say, because about 50% of U.S. births are in that group.
Despite the risks of birth defects and other complications, opioids are prescribed for > 33% of reproductive-aged women enrolled in Medicaid and 28% of those with private insurance, according to a study by the CDC.
Related: Acute Upper Abdominal Pain in Early Pregnancy
“Many women taking these medicines may not know they are pregnant,” said CDC Director Tom Frieden, MD, MPH. “That’s why it’s critical for health care professionals to take a thorough health assessment before prescribing these medicines to women of reproductive age."
Related: Reducing Opioid Use for Chronic Pain
Studies have suggested that giving opioids to women who are pregnant raises the risks of neural tube defects, congenital heart defects, and gastroschisis, as well as neonatal abstinence syndrome.
Related: Maternal Morbidity: Higher Risks for Minorities
In the CDC study, researchers looked at data from health insurance claims from 2008 to 2012 and from Medicaid. The most frequently prescribed opioids were hydrocodone, codeine, and oxycodone. Prescription rates for women of reproductive age were highest in the South and lowest in the northeastern U.S. Prescriptions were nearly 1.5 times higher for white women compared with that of black or Hispanic women. The consistently higher rate of prescribing to Medicaid-enrolled women is of concern, the researchers say, because about 50% of U.S. births are in that group.
VHA Looks to Reduce Overdoses With Opioid Therapy Risk Report
To promote the use of the Opioid Therapy Risk Report, a computer tool designed to reduce opiate overprescribing, VA Interim Under Secretary for Health Dr. Carolyn Clancy visited more than 2,000 VHA primary care providers during the first week of March.
The risk report allows VHA staff to review information about pain treatment and the dosages of narcotics and other sedative medications in one place, helping to monitor patients already using high doses of opioids or those who have medical risk factors that put them at an increased risk of complications from opioid medications.
“It is critical that we ensure systemwide implementation of the Opioid Therapy Risk Report in the weeks ahead,” Dr. Clancy said.
According to Jennifer Kryskalla, PharmD, pharmacoeconomist and pharmacy data analyst for VISN 22 and coauthor of the September 2014 Federal Practitioner article “Using Dashboard Technology to Monitor Overdose Risk,” the Opioid Therapy Tool grew out of local dashboards like the one developed in VISN 22.
“VISNs 21 and 22 both had opioid dashboards, and all were invited to provide feedback on the development. These tools help our prescribers provide better, safer care for our veterans,” she explained.
“The Opioid Therapy Risk Report promotes a dialogue among the various disciplines responsible for providing care to veterans who have, in many cases, complex comorbidities that involve pain, combat injuries, and psychiatric conditions, such as PTSD and depression, related to high risk for suicide,” Peter Hauser, MD, the Mental Health Care Services lead in VISN 22 told Federal Practitioner. “However, the IT tool alone is just a beginning. A major part of the solution is to provide veterans with chronic pain and the clinicians who serve them other alternatives for pain relief, ones that also address psychiatric comorbidities.”
For more on the challenges in lowering veterans’ opioid use and reducing overdose risk, listen to this exclusive interview with Dr. Kryskalla and Dr. Hauser: "Lowering Veterans' Opioid Use and Reducing Overdose Risk."
To promote the use of the Opioid Therapy Risk Report, a computer tool designed to reduce opiate overprescribing, VA Interim Under Secretary for Health Dr. Carolyn Clancy visited more than 2,000 VHA primary care providers during the first week of March.
The risk report allows VHA staff to review information about pain treatment and the dosages of narcotics and other sedative medications in one place, helping to monitor patients already using high doses of opioids or those who have medical risk factors that put them at an increased risk of complications from opioid medications.
“It is critical that we ensure systemwide implementation of the Opioid Therapy Risk Report in the weeks ahead,” Dr. Clancy said.
According to Jennifer Kryskalla, PharmD, pharmacoeconomist and pharmacy data analyst for VISN 22 and coauthor of the September 2014 Federal Practitioner article “Using Dashboard Technology to Monitor Overdose Risk,” the Opioid Therapy Tool grew out of local dashboards like the one developed in VISN 22.
“VISNs 21 and 22 both had opioid dashboards, and all were invited to provide feedback on the development. These tools help our prescribers provide better, safer care for our veterans,” she explained.
“The Opioid Therapy Risk Report promotes a dialogue among the various disciplines responsible for providing care to veterans who have, in many cases, complex comorbidities that involve pain, combat injuries, and psychiatric conditions, such as PTSD and depression, related to high risk for suicide,” Peter Hauser, MD, the Mental Health Care Services lead in VISN 22 told Federal Practitioner. “However, the IT tool alone is just a beginning. A major part of the solution is to provide veterans with chronic pain and the clinicians who serve them other alternatives for pain relief, ones that also address psychiatric comorbidities.”
For more on the challenges in lowering veterans’ opioid use and reducing overdose risk, listen to this exclusive interview with Dr. Kryskalla and Dr. Hauser: "Lowering Veterans' Opioid Use and Reducing Overdose Risk."
To promote the use of the Opioid Therapy Risk Report, a computer tool designed to reduce opiate overprescribing, VA Interim Under Secretary for Health Dr. Carolyn Clancy visited more than 2,000 VHA primary care providers during the first week of March.
The risk report allows VHA staff to review information about pain treatment and the dosages of narcotics and other sedative medications in one place, helping to monitor patients already using high doses of opioids or those who have medical risk factors that put them at an increased risk of complications from opioid medications.
“It is critical that we ensure systemwide implementation of the Opioid Therapy Risk Report in the weeks ahead,” Dr. Clancy said.
According to Jennifer Kryskalla, PharmD, pharmacoeconomist and pharmacy data analyst for VISN 22 and coauthor of the September 2014 Federal Practitioner article “Using Dashboard Technology to Monitor Overdose Risk,” the Opioid Therapy Tool grew out of local dashboards like the one developed in VISN 22.
“VISNs 21 and 22 both had opioid dashboards, and all were invited to provide feedback on the development. These tools help our prescribers provide better, safer care for our veterans,” she explained.
“The Opioid Therapy Risk Report promotes a dialogue among the various disciplines responsible for providing care to veterans who have, in many cases, complex comorbidities that involve pain, combat injuries, and psychiatric conditions, such as PTSD and depression, related to high risk for suicide,” Peter Hauser, MD, the Mental Health Care Services lead in VISN 22 told Federal Practitioner. “However, the IT tool alone is just a beginning. A major part of the solution is to provide veterans with chronic pain and the clinicians who serve them other alternatives for pain relief, ones that also address psychiatric comorbidities.”
For more on the challenges in lowering veterans’ opioid use and reducing overdose risk, listen to this exclusive interview with Dr. Kryskalla and Dr. Hauser: "Lowering Veterans' Opioid Use and Reducing Overdose Risk."
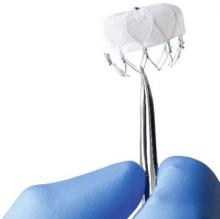
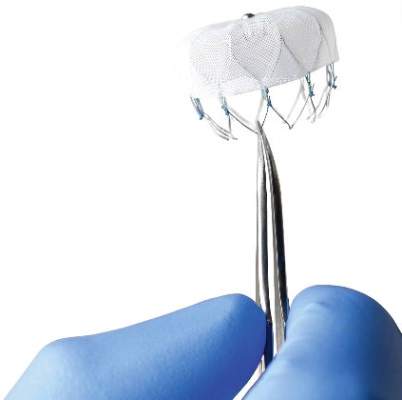
Novel Watchman device approved as warfarin alternative in atrial fib
The Watchman left atrial appendage (LAA) closure device has been approved in the United States as an alternative to warfarin for patients with nonvalvular atrial fibrillation, for a narrower indication than the one submitted for approval to the Food and Drug Administration.
The device is a percutaneously delivered permanent cardiac implant placed in the LAA to prevent the embolization of thrombi formed in the LAA, and is manufactured by Boston Scientific. The FDA approved the Watchman for reducing the risk of thromboembolism from the LAA in patients with nonvalvular atrial fibrillation “who are at increased risk for stroke and systemic embolism based on CHADS2 or CHA2DS2-VASc scores, are deemed by their physicians to be suitable for warfarin; and have an appropriate rationale to seek a nonpharmacologic alternative to warfarin, taking into account the safety and effectiveness of the device, compared to warfarin,” according to a statement issued by the company on March 13.
The approved indication is worded differently than the proposed indication that was submitted to the FDA for approval and discussed at an FDA panel meeting in October, to “prevent thromboembolism from the left atrial appendage.” The changes in the indication include the replacement of “prevent” with “reduce the risk” of thromboembolism, and the addition of the following qualifiers: In patients who “are deemed by their physicians to be suitable for warfarin,” and who have “an appropriate rationale to seek a nonpharmacologic alternative to warfarin, taking into account the safety and effectiveness of the device compared to warfarin.”
“These changes were made to more accurately reflect the appropriate patient population for this device,” according to an FDA spokesperson.
At a meeting in October 2014, the FDA’s Circulatory System Devices Panel voted 6-5 with one abstention that the benefits of the device outweighed its risks for the proposed indication, but several panelists who voted no said they would support approval of a second-line indication. In addition, panelists voting on both sides of this question said that the indication was too broad and should be revised to describe the device as a second-line alternative to warfarin, making clear it is not appropriate for all warfarin-eligible patients. (At the meeting, the panel unanimously agreed that there was “reasonable assurance” that the device was safe for use in this population.)
At the first advisory panel meeting on the device, in December 2013, the panel voted 13-1 to recommend approval, based on data from the PREVAIL and PROTECT-AF studies, which compared the device to chronic warfarin, and information from the Continued Access to PREVAIL (CAP2) registry.PREVAIL compared implantation of the device – with 45 days of warfarin plus 81 mg of aspirin for 45 days, followed by 325 mg of aspirin and 75 mg of clopidogrel through 6 months, followed by 325 mg of aspirin a day indefinitely – to chronic warfarin.
The October meeting was convened by the FDA to review longer follow-up data from PREVAIL, which found additional cases of ischemic strokes in the Watchman group and none in the warfarin-treated group.
The Watchman device has been available outside of the United States since 2009, is registered in 75 countries, and has been used to treat more than 10,000 patients, according to Boston Scientific.
The Watchman left atrial appendage (LAA) closure device has been approved in the United States as an alternative to warfarin for patients with nonvalvular atrial fibrillation, for a narrower indication than the one submitted for approval to the Food and Drug Administration.
The device is a percutaneously delivered permanent cardiac implant placed in the LAA to prevent the embolization of thrombi formed in the LAA, and is manufactured by Boston Scientific. The FDA approved the Watchman for reducing the risk of thromboembolism from the LAA in patients with nonvalvular atrial fibrillation “who are at increased risk for stroke and systemic embolism based on CHADS2 or CHA2DS2-VASc scores, are deemed by their physicians to be suitable for warfarin; and have an appropriate rationale to seek a nonpharmacologic alternative to warfarin, taking into account the safety and effectiveness of the device, compared to warfarin,” according to a statement issued by the company on March 13.
The approved indication is worded differently than the proposed indication that was submitted to the FDA for approval and discussed at an FDA panel meeting in October, to “prevent thromboembolism from the left atrial appendage.” The changes in the indication include the replacement of “prevent” with “reduce the risk” of thromboembolism, and the addition of the following qualifiers: In patients who “are deemed by their physicians to be suitable for warfarin,” and who have “an appropriate rationale to seek a nonpharmacologic alternative to warfarin, taking into account the safety and effectiveness of the device compared to warfarin.”
“These changes were made to more accurately reflect the appropriate patient population for this device,” according to an FDA spokesperson.
At a meeting in October 2014, the FDA’s Circulatory System Devices Panel voted 6-5 with one abstention that the benefits of the device outweighed its risks for the proposed indication, but several panelists who voted no said they would support approval of a second-line indication. In addition, panelists voting on both sides of this question said that the indication was too broad and should be revised to describe the device as a second-line alternative to warfarin, making clear it is not appropriate for all warfarin-eligible patients. (At the meeting, the panel unanimously agreed that there was “reasonable assurance” that the device was safe for use in this population.)
At the first advisory panel meeting on the device, in December 2013, the panel voted 13-1 to recommend approval, based on data from the PREVAIL and PROTECT-AF studies, which compared the device to chronic warfarin, and information from the Continued Access to PREVAIL (CAP2) registry.PREVAIL compared implantation of the device – with 45 days of warfarin plus 81 mg of aspirin for 45 days, followed by 325 mg of aspirin and 75 mg of clopidogrel through 6 months, followed by 325 mg of aspirin a day indefinitely – to chronic warfarin.
The October meeting was convened by the FDA to review longer follow-up data from PREVAIL, which found additional cases of ischemic strokes in the Watchman group and none in the warfarin-treated group.
The Watchman device has been available outside of the United States since 2009, is registered in 75 countries, and has been used to treat more than 10,000 patients, according to Boston Scientific.
The Watchman left atrial appendage (LAA) closure device has been approved in the United States as an alternative to warfarin for patients with nonvalvular atrial fibrillation, for a narrower indication than the one submitted for approval to the Food and Drug Administration.
The device is a percutaneously delivered permanent cardiac implant placed in the LAA to prevent the embolization of thrombi formed in the LAA, and is manufactured by Boston Scientific. The FDA approved the Watchman for reducing the risk of thromboembolism from the LAA in patients with nonvalvular atrial fibrillation “who are at increased risk for stroke and systemic embolism based on CHADS2 or CHA2DS2-VASc scores, are deemed by their physicians to be suitable for warfarin; and have an appropriate rationale to seek a nonpharmacologic alternative to warfarin, taking into account the safety and effectiveness of the device, compared to warfarin,” according to a statement issued by the company on March 13.
The approved indication is worded differently than the proposed indication that was submitted to the FDA for approval and discussed at an FDA panel meeting in October, to “prevent thromboembolism from the left atrial appendage.” The changes in the indication include the replacement of “prevent” with “reduce the risk” of thromboembolism, and the addition of the following qualifiers: In patients who “are deemed by their physicians to be suitable for warfarin,” and who have “an appropriate rationale to seek a nonpharmacologic alternative to warfarin, taking into account the safety and effectiveness of the device compared to warfarin.”
“These changes were made to more accurately reflect the appropriate patient population for this device,” according to an FDA spokesperson.
At a meeting in October 2014, the FDA’s Circulatory System Devices Panel voted 6-5 with one abstention that the benefits of the device outweighed its risks for the proposed indication, but several panelists who voted no said they would support approval of a second-line indication. In addition, panelists voting on both sides of this question said that the indication was too broad and should be revised to describe the device as a second-line alternative to warfarin, making clear it is not appropriate for all warfarin-eligible patients. (At the meeting, the panel unanimously agreed that there was “reasonable assurance” that the device was safe for use in this population.)
At the first advisory panel meeting on the device, in December 2013, the panel voted 13-1 to recommend approval, based on data from the PREVAIL and PROTECT-AF studies, which compared the device to chronic warfarin, and information from the Continued Access to PREVAIL (CAP2) registry.PREVAIL compared implantation of the device – with 45 days of warfarin plus 81 mg of aspirin for 45 days, followed by 325 mg of aspirin and 75 mg of clopidogrel through 6 months, followed by 325 mg of aspirin a day indefinitely – to chronic warfarin.
The October meeting was convened by the FDA to review longer follow-up data from PREVAIL, which found additional cases of ischemic strokes in the Watchman group and none in the warfarin-treated group.
The Watchman device has been available outside of the United States since 2009, is registered in 75 countries, and has been used to treat more than 10,000 patients, according to Boston Scientific.
Registration Is Open for 35th Annual Wheelchair Games
The largest annual wheelchair sports event in the world is open for registration through April 15, 2015. The National Veterans Wheelchair Games is a sports and rehabilitation program cosponsored since 1985 by VA and Paralyzed Veterans of America for veterans with spinal cord injuries, amputations, or neurologic problems. Each year, the games attract > 600 athletes who compete in archery, basketball, bowling, hand cycling, power soccer, quad rugby, swimming, weight lifting, and other sports.
Related: Advances in Prosthetics Restore High Levels of Physical Activity
This year’s games, with the theme BIG City, BIG Wheels, BIG Heart, will take place in Dallas, Texas, June 21 through June 26. Jeff Milligan, director of VA North Texas Health Care System, and Steven Ray, chapter president of Lone Star Paralyzed Veterans, are hosting the event.
For more information, visit http://www.wheelchairgames.org.
The largest annual wheelchair sports event in the world is open for registration through April 15, 2015. The National Veterans Wheelchair Games is a sports and rehabilitation program cosponsored since 1985 by VA and Paralyzed Veterans of America for veterans with spinal cord injuries, amputations, or neurologic problems. Each year, the games attract > 600 athletes who compete in archery, basketball, bowling, hand cycling, power soccer, quad rugby, swimming, weight lifting, and other sports.
Related: Advances in Prosthetics Restore High Levels of Physical Activity
This year’s games, with the theme BIG City, BIG Wheels, BIG Heart, will take place in Dallas, Texas, June 21 through June 26. Jeff Milligan, director of VA North Texas Health Care System, and Steven Ray, chapter president of Lone Star Paralyzed Veterans, are hosting the event.
For more information, visit http://www.wheelchairgames.org.
The largest annual wheelchair sports event in the world is open for registration through April 15, 2015. The National Veterans Wheelchair Games is a sports and rehabilitation program cosponsored since 1985 by VA and Paralyzed Veterans of America for veterans with spinal cord injuries, amputations, or neurologic problems. Each year, the games attract > 600 athletes who compete in archery, basketball, bowling, hand cycling, power soccer, quad rugby, swimming, weight lifting, and other sports.
Related: Advances in Prosthetics Restore High Levels of Physical Activity
This year’s games, with the theme BIG City, BIG Wheels, BIG Heart, will take place in Dallas, Texas, June 21 through June 26. Jeff Milligan, director of VA North Texas Health Care System, and Steven Ray, chapter president of Lone Star Paralyzed Veterans, are hosting the event.
For more information, visit http://www.wheelchairgames.org.
CDC monitoring Americans after potential Ebola exposure in West Africa
The Centers for Disease Control and Prevention continues to monitor potential Ebola exposure among several American citizens following the identification of an American volunteer health care worker who tested positive for the virus. That person was admitted to the NIH Clinical Center on March 13 for care and treatment.
CDC and the State Department are working to return to the United States an unspecified number of American citizens who were potential exposed to the index patient, None have tested positive for Ebola virus disease so far. These individuals will be monitored near the University of Nebraska Medical Center, the National Institutes of Health, or Emory University Hospital.
For more information, visit the CDC website.
The Centers for Disease Control and Prevention continues to monitor potential Ebola exposure among several American citizens following the identification of an American volunteer health care worker who tested positive for the virus. That person was admitted to the NIH Clinical Center on March 13 for care and treatment.
CDC and the State Department are working to return to the United States an unspecified number of American citizens who were potential exposed to the index patient, None have tested positive for Ebola virus disease so far. These individuals will be monitored near the University of Nebraska Medical Center, the National Institutes of Health, or Emory University Hospital.
For more information, visit the CDC website.
The Centers for Disease Control and Prevention continues to monitor potential Ebola exposure among several American citizens following the identification of an American volunteer health care worker who tested positive for the virus. That person was admitted to the NIH Clinical Center on March 13 for care and treatment.
CDC and the State Department are working to return to the United States an unspecified number of American citizens who were potential exposed to the index patient, None have tested positive for Ebola virus disease so far. These individuals will be monitored near the University of Nebraska Medical Center, the National Institutes of Health, or Emory University Hospital.
For more information, visit the CDC website.
U.S. volunteer infected with Ebola in Sierra Leone
The Centers for Disease Control and Prevention will conduct contact tracing of individuals in Sierra Leone, including several American citizens, who may have come into contact with an American health worker who has become infected with the Ebola virus.
The patient returned to the United States by medevac and has been admitted to the National Institutes of Health Clinical Center for treatment, the agency said in a written statement.
Although none of the individuals currently being monitored in Sierra Leone have tested positive for Ebola, they will voluntarily self-isolate and remain under direct active monitoring for the 21-day incubation period before returning to the United States.
In the event that another Ebola case is confirmed, the CDC and the State Department are developing contingency plans for returning the other American health care workers to the United States by noncommercial air transport.
The Centers for Disease Control and Prevention will conduct contact tracing of individuals in Sierra Leone, including several American citizens, who may have come into contact with an American health worker who has become infected with the Ebola virus.
The patient returned to the United States by medevac and has been admitted to the National Institutes of Health Clinical Center for treatment, the agency said in a written statement.
Although none of the individuals currently being monitored in Sierra Leone have tested positive for Ebola, they will voluntarily self-isolate and remain under direct active monitoring for the 21-day incubation period before returning to the United States.
In the event that another Ebola case is confirmed, the CDC and the State Department are developing contingency plans for returning the other American health care workers to the United States by noncommercial air transport.
The Centers for Disease Control and Prevention will conduct contact tracing of individuals in Sierra Leone, including several American citizens, who may have come into contact with an American health worker who has become infected with the Ebola virus.
The patient returned to the United States by medevac and has been admitted to the National Institutes of Health Clinical Center for treatment, the agency said in a written statement.
Although none of the individuals currently being monitored in Sierra Leone have tested positive for Ebola, they will voluntarily self-isolate and remain under direct active monitoring for the 21-day incubation period before returning to the United States.
In the event that another Ebola case is confirmed, the CDC and the State Department are developing contingency plans for returning the other American health care workers to the United States by noncommercial air transport.
House negotiating SGR fix, repeal could come soon
Negotiations are apparently underway in the House of Representatives over the Medicare Sustainable Growth Rate (SGR) formula, and agreement on a possible repeal could be on the horizon.
The new efforts will hopefully lead to an improved, permanent payment system that ensures Medicare is on a sustainable path going forward, said Sen. Ron Wyden (D-Ore.), ranking member of the Senate Finance Committee.
“I’ve been in Congress long enough to be skeptical of rumors, but what we are hearing from the House suggests there is real movement to fully repeal and replace the flawed formula for paying Medicare providers known as SGR,” Sen. Wyden said in a statement. “If what we’re hearing is true, it’s good news and moves us closer to something I’ve been working tirelessly to achieve – a payment formula that stands on its own, doesn’t require annual and expensive ‘patches,’ and which opens the door to improving the way care is delivered.”
The current SGR patch expires March 31, after which physicians can expect a 21% pay cut.
A new analysis of last year’s repeal legislation notes the bill has strengths and weaknesses for physicians (Health Aff. 2015 doi: 10.1377/hlthaff.2014.1429). Current congressional deliberations are focused on how to pay for the SGR fix, with wide consensus that the 2014 legislation will remain the basic model for reform.
The SGR Repeal and Medicare Provider Payment Modernization Act of 2014 includes opportunities for specialists to participate in pay-for-performance programs and other alternative payment models, according to author James Reschovsky, a senior fellow at policy research firm Mathematica Policy Research. The legislation would create two new payment pathways for doctors, one for those who want to continue to receive fee-for-service payments, and another for those already participating in value-based payment models, including accountable care arrangements and bundled payment initiatives.
But the bill fails to address distortions in Medicare’s fee-for-service fee schedule, which some argue overvalues specialty care services, while undervaluing preventive care, Mr. Reschovsky and his colleagues said. Without fixing that defect, specialists may find it more lucrative to stay in Medicare’s fee-for-service program and continue to concentrate on providing high-cost services. Attempts to bring down costs through ACOs and other alternative payment methods also will be difficult as long as the fee-for-service pay rates underlying these programs are skewed.
“Correcting fee schedule valuations will be a substantial and controversial undertaking,” they wrote. “But it is one that is vitally important to the SGR fix’s prospects for success.”
The SGR Repeal and Medicare Provider Payment Modernization Act of 2014 passed the House last year, but was not taken up by the Senate
On Twitter @legal_med
Negotiations are apparently underway in the House of Representatives over the Medicare Sustainable Growth Rate (SGR) formula, and agreement on a possible repeal could be on the horizon.
The new efforts will hopefully lead to an improved, permanent payment system that ensures Medicare is on a sustainable path going forward, said Sen. Ron Wyden (D-Ore.), ranking member of the Senate Finance Committee.
“I’ve been in Congress long enough to be skeptical of rumors, but what we are hearing from the House suggests there is real movement to fully repeal and replace the flawed formula for paying Medicare providers known as SGR,” Sen. Wyden said in a statement. “If what we’re hearing is true, it’s good news and moves us closer to something I’ve been working tirelessly to achieve – a payment formula that stands on its own, doesn’t require annual and expensive ‘patches,’ and which opens the door to improving the way care is delivered.”
The current SGR patch expires March 31, after which physicians can expect a 21% pay cut.
A new analysis of last year’s repeal legislation notes the bill has strengths and weaknesses for physicians (Health Aff. 2015 doi: 10.1377/hlthaff.2014.1429). Current congressional deliberations are focused on how to pay for the SGR fix, with wide consensus that the 2014 legislation will remain the basic model for reform.
The SGR Repeal and Medicare Provider Payment Modernization Act of 2014 includes opportunities for specialists to participate in pay-for-performance programs and other alternative payment models, according to author James Reschovsky, a senior fellow at policy research firm Mathematica Policy Research. The legislation would create two new payment pathways for doctors, one for those who want to continue to receive fee-for-service payments, and another for those already participating in value-based payment models, including accountable care arrangements and bundled payment initiatives.
But the bill fails to address distortions in Medicare’s fee-for-service fee schedule, which some argue overvalues specialty care services, while undervaluing preventive care, Mr. Reschovsky and his colleagues said. Without fixing that defect, specialists may find it more lucrative to stay in Medicare’s fee-for-service program and continue to concentrate on providing high-cost services. Attempts to bring down costs through ACOs and other alternative payment methods also will be difficult as long as the fee-for-service pay rates underlying these programs are skewed.
“Correcting fee schedule valuations will be a substantial and controversial undertaking,” they wrote. “But it is one that is vitally important to the SGR fix’s prospects for success.”
The SGR Repeal and Medicare Provider Payment Modernization Act of 2014 passed the House last year, but was not taken up by the Senate
On Twitter @legal_med
Negotiations are apparently underway in the House of Representatives over the Medicare Sustainable Growth Rate (SGR) formula, and agreement on a possible repeal could be on the horizon.
The new efforts will hopefully lead to an improved, permanent payment system that ensures Medicare is on a sustainable path going forward, said Sen. Ron Wyden (D-Ore.), ranking member of the Senate Finance Committee.
“I’ve been in Congress long enough to be skeptical of rumors, but what we are hearing from the House suggests there is real movement to fully repeal and replace the flawed formula for paying Medicare providers known as SGR,” Sen. Wyden said in a statement. “If what we’re hearing is true, it’s good news and moves us closer to something I’ve been working tirelessly to achieve – a payment formula that stands on its own, doesn’t require annual and expensive ‘patches,’ and which opens the door to improving the way care is delivered.”
The current SGR patch expires March 31, after which physicians can expect a 21% pay cut.
A new analysis of last year’s repeal legislation notes the bill has strengths and weaknesses for physicians (Health Aff. 2015 doi: 10.1377/hlthaff.2014.1429). Current congressional deliberations are focused on how to pay for the SGR fix, with wide consensus that the 2014 legislation will remain the basic model for reform.
The SGR Repeal and Medicare Provider Payment Modernization Act of 2014 includes opportunities for specialists to participate in pay-for-performance programs and other alternative payment models, according to author James Reschovsky, a senior fellow at policy research firm Mathematica Policy Research. The legislation would create two new payment pathways for doctors, one for those who want to continue to receive fee-for-service payments, and another for those already participating in value-based payment models, including accountable care arrangements and bundled payment initiatives.
But the bill fails to address distortions in Medicare’s fee-for-service fee schedule, which some argue overvalues specialty care services, while undervaluing preventive care, Mr. Reschovsky and his colleagues said. Without fixing that defect, specialists may find it more lucrative to stay in Medicare’s fee-for-service program and continue to concentrate on providing high-cost services. Attempts to bring down costs through ACOs and other alternative payment methods also will be difficult as long as the fee-for-service pay rates underlying these programs are skewed.
“Correcting fee schedule valuations will be a substantial and controversial undertaking,” they wrote. “But it is one that is vitally important to the SGR fix’s prospects for success.”
The SGR Repeal and Medicare Provider Payment Modernization Act of 2014 passed the House last year, but was not taken up by the Senate
On Twitter @legal_med
Faster Response to Overdoses
Lowering the rate of deaths from opioid overdoses in Indian Country is the idea behind the Law Enforcement Naloxone Toolkit, developed by IHS, U.S. Department of Justice, and various HHS agencies.
Related: Lowering Veterans' Opioid Use and Reducing Overdose Risk
Opioid overdoses can quickly turn fatal—within 45 to 90 minutes—but naloxone, provided in time, can restore breathing within 2 to 5 minutes, potentially preventing brain damage and death. Law enforcement officers are often first on the scene of an overdose, so their actions can mean the difference between life and death, according to the IHS.
Related: Reducing Opioid Use for Chronic Pain
The downloadable toolkit offers answers to frequent questions about naloxone and offers sample documents and templates, such as data collection forms. The toolkit is provided free to all interested tribal and IHS facilities.
Related: SAMHSA Awards Funds for Tribal Youth Programs
As of October 2014, law enforcement agencies in at least 19 states have implemented naloxone programs.
Lowering the rate of deaths from opioid overdoses in Indian Country is the idea behind the Law Enforcement Naloxone Toolkit, developed by IHS, U.S. Department of Justice, and various HHS agencies.
Related: Lowering Veterans' Opioid Use and Reducing Overdose Risk
Opioid overdoses can quickly turn fatal—within 45 to 90 minutes—but naloxone, provided in time, can restore breathing within 2 to 5 minutes, potentially preventing brain damage and death. Law enforcement officers are often first on the scene of an overdose, so their actions can mean the difference between life and death, according to the IHS.
Related: Reducing Opioid Use for Chronic Pain
The downloadable toolkit offers answers to frequent questions about naloxone and offers sample documents and templates, such as data collection forms. The toolkit is provided free to all interested tribal and IHS facilities.
Related: SAMHSA Awards Funds for Tribal Youth Programs
As of October 2014, law enforcement agencies in at least 19 states have implemented naloxone programs.
Lowering the rate of deaths from opioid overdoses in Indian Country is the idea behind the Law Enforcement Naloxone Toolkit, developed by IHS, U.S. Department of Justice, and various HHS agencies.
Related: Lowering Veterans' Opioid Use and Reducing Overdose Risk
Opioid overdoses can quickly turn fatal—within 45 to 90 minutes—but naloxone, provided in time, can restore breathing within 2 to 5 minutes, potentially preventing brain damage and death. Law enforcement officers are often first on the scene of an overdose, so their actions can mean the difference between life and death, according to the IHS.
Related: Reducing Opioid Use for Chronic Pain
The downloadable toolkit offers answers to frequent questions about naloxone and offers sample documents and templates, such as data collection forms. The toolkit is provided free to all interested tribal and IHS facilities.
Related: SAMHSA Awards Funds for Tribal Youth Programs
As of October 2014, law enforcement agencies in at least 19 states have implemented naloxone programs.
Despite requirement, only 13% of clinical trials report results
Only 13% of the clinical trials legally required since 2007 to report their findings to ClinicalTrials.gov actually do so, according to a report published online in the New England Journal of Medicine.
The Food and Drug Administration Amendments Act requires sponsors of most clinical trials to register and report their basic summary results within 1 year of either completing data collection for the primary outcome or of terminating. Failure to report study findings is punishable by sanctions including civil penalties of up to $10,000 per day and loss of funding.
The reporting regulation “reflect[s] the ethical obligation of researchers and sponsors to respect human trial participants through fidelity to commitments made explicit in informed consent: namely, to make results of trials available to contribute to generalizable knowledge,” said Dr. Monique L. Anderson of the Duke Clinical Research Institute, Durham, N.C., and her associates.
The law was enacted because of public concern that sponsors and investigators were “selectively publishing trials that favored interests of the sponsors, and that journals were selectively reporting positive findings.” Among other detriments, the failure to report results could harm participants in similar studies by failing to warn them of possible risks, the investigators noted.
They assessed compliance with the law more than 5 years after it was enacted, using a National Library of Medicine algorithm to identify 13,327 eligible clinical trials that were either completed or terminated during 2008-2012.
Only 13.4% of them reported their results within 1 year, and only 38.3% reported their results at any time during the study period (N. Engl. J. Med. 2015;372:1031-9). Moreover, “despite ethical mandates, statutory obligations, and considerable societal pressure, most trials that were funded by the NIH or other government or academic institutions ... have yet to report results at ClinicalTrials.gov, whereas the medical-products industry has been more responsive to the legal mandate,” the researchers explained.
At 1 year, the rate of reporting was 17.0% for industry-sponsored trials, 8.1% for NIH-funded trials, and 5.7% for other government- or academically funded trials. The corresponding rates of reporting at 5 years were only slightly better, at 41.5%, 38.9%, and 27.7%, respectively.
Phase I, II, and III clinical trials were the less likely than phase IV trials were to report their results in a timely fashion. These earlier-phase studies “are primarily focused on proof-of-concept demonstrations and as such represent valuable, closely guarded intellectual property,” Dr. Anderson and her associates said.
Despite the regulation’s threat of penalties, no enforcement has yet occurred, the researchers noted, in part because this portion of the FDA Administration and Amendments Act is still under public discussion and hasn’t been finalized.
Only 13% of the clinical trials legally required since 2007 to report their findings to ClinicalTrials.gov actually do so, according to a report published online in the New England Journal of Medicine.
The Food and Drug Administration Amendments Act requires sponsors of most clinical trials to register and report their basic summary results within 1 year of either completing data collection for the primary outcome or of terminating. Failure to report study findings is punishable by sanctions including civil penalties of up to $10,000 per day and loss of funding.
The reporting regulation “reflect[s] the ethical obligation of researchers and sponsors to respect human trial participants through fidelity to commitments made explicit in informed consent: namely, to make results of trials available to contribute to generalizable knowledge,” said Dr. Monique L. Anderson of the Duke Clinical Research Institute, Durham, N.C., and her associates.
The law was enacted because of public concern that sponsors and investigators were “selectively publishing trials that favored interests of the sponsors, and that journals were selectively reporting positive findings.” Among other detriments, the failure to report results could harm participants in similar studies by failing to warn them of possible risks, the investigators noted.
They assessed compliance with the law more than 5 years after it was enacted, using a National Library of Medicine algorithm to identify 13,327 eligible clinical trials that were either completed or terminated during 2008-2012.
Only 13.4% of them reported their results within 1 year, and only 38.3% reported their results at any time during the study period (N. Engl. J. Med. 2015;372:1031-9). Moreover, “despite ethical mandates, statutory obligations, and considerable societal pressure, most trials that were funded by the NIH or other government or academic institutions ... have yet to report results at ClinicalTrials.gov, whereas the medical-products industry has been more responsive to the legal mandate,” the researchers explained.
At 1 year, the rate of reporting was 17.0% for industry-sponsored trials, 8.1% for NIH-funded trials, and 5.7% for other government- or academically funded trials. The corresponding rates of reporting at 5 years were only slightly better, at 41.5%, 38.9%, and 27.7%, respectively.
Phase I, II, and III clinical trials were the less likely than phase IV trials were to report their results in a timely fashion. These earlier-phase studies “are primarily focused on proof-of-concept demonstrations and as such represent valuable, closely guarded intellectual property,” Dr. Anderson and her associates said.
Despite the regulation’s threat of penalties, no enforcement has yet occurred, the researchers noted, in part because this portion of the FDA Administration and Amendments Act is still under public discussion and hasn’t been finalized.
Only 13% of the clinical trials legally required since 2007 to report their findings to ClinicalTrials.gov actually do so, according to a report published online in the New England Journal of Medicine.
The Food and Drug Administration Amendments Act requires sponsors of most clinical trials to register and report their basic summary results within 1 year of either completing data collection for the primary outcome or of terminating. Failure to report study findings is punishable by sanctions including civil penalties of up to $10,000 per day and loss of funding.
The reporting regulation “reflect[s] the ethical obligation of researchers and sponsors to respect human trial participants through fidelity to commitments made explicit in informed consent: namely, to make results of trials available to contribute to generalizable knowledge,” said Dr. Monique L. Anderson of the Duke Clinical Research Institute, Durham, N.C., and her associates.
The law was enacted because of public concern that sponsors and investigators were “selectively publishing trials that favored interests of the sponsors, and that journals were selectively reporting positive findings.” Among other detriments, the failure to report results could harm participants in similar studies by failing to warn them of possible risks, the investigators noted.
They assessed compliance with the law more than 5 years after it was enacted, using a National Library of Medicine algorithm to identify 13,327 eligible clinical trials that were either completed or terminated during 2008-2012.
Only 13.4% of them reported their results within 1 year, and only 38.3% reported their results at any time during the study period (N. Engl. J. Med. 2015;372:1031-9). Moreover, “despite ethical mandates, statutory obligations, and considerable societal pressure, most trials that were funded by the NIH or other government or academic institutions ... have yet to report results at ClinicalTrials.gov, whereas the medical-products industry has been more responsive to the legal mandate,” the researchers explained.
At 1 year, the rate of reporting was 17.0% for industry-sponsored trials, 8.1% for NIH-funded trials, and 5.7% for other government- or academically funded trials. The corresponding rates of reporting at 5 years were only slightly better, at 41.5%, 38.9%, and 27.7%, respectively.
Phase I, II, and III clinical trials were the less likely than phase IV trials were to report their results in a timely fashion. These earlier-phase studies “are primarily focused on proof-of-concept demonstrations and as such represent valuable, closely guarded intellectual property,” Dr. Anderson and her associates said.
Despite the regulation’s threat of penalties, no enforcement has yet occurred, the researchers noted, in part because this portion of the FDA Administration and Amendments Act is still under public discussion and hasn’t been finalized.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Key clinical point: Only 13% of clinical trials legally required to report their findings to ClinicalTrials.gov actually do so.
Major finding: Only 13.4% of 13,327 clinical trials reported their results within 1 year, and only 38.3% of those trials reported results at any time during the 5-year study period.
Data source: An analysis of 13,327 clinical trials completed between 2008 and 2012 that were required to report their results to ClinicalTrials.gov within 1 year.
Disclosures: The study was supported by the Clinical Trials Transformation Initiative and the NIH Common Fund. Dr. Anderson reported having no financial disclosures; two of her associates reported ties to numerous industry sources.
Heroin overdoses up dramatically since 2010
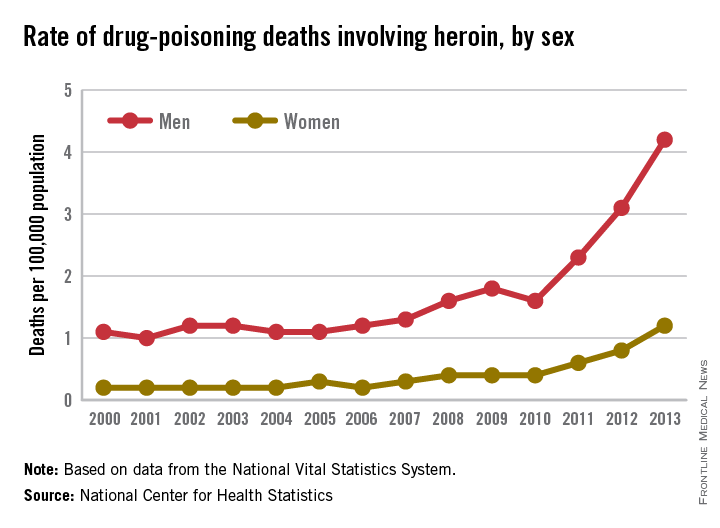
Drug-poisoning deaths involving heroin have soared since 2000, and most of the increase occurred since 2010, according to a report from the National Center for Health Statistics.
From 2010 to 2013, the rate of heroin overdose deaths increased 163% for men, from a rate of 1.6/100,000 population in 2010 to 4.2 in 2013. For women, the death rate increased by 200%, from 0.4/100,000 in 2010 to 1.2/100,000 in 2013. From 2000 to 2010, however, the rate of increase was much slower, with the death rate increasing from 1.1 to 1.6 for men and from 0.2 to 0.4 for women.

The overall rate for heroin overdose from 2000 to 2013 increased from 0.7 to 2.7/100,000. Most of this increase occurred from 2010 to 2013: From 2000 to 2010, the death rate increased to only 1/100,000, a growth rate of 6%, but after 2010, the rate grew by 37% per year, the NCHS reported.
In 2013, non-Hispanic whites aged 18-44 years had the highest heroin poisoning death rate among measured racial/ethnic groups at 7/100,000. In 2000, older, non-Hispanic blacks aged 45-64 years had the highest death rate among the reported racial/ethnic groups at 2/100,000. The death rate for whites aged 18-44 in 2000 was 1.2/100,000, meaning that the death rate increased by 483% from 2000 to 2013. For non-Hispanic blacks aged 45-64, the death rate in 2013 was 4.9, an increase of 145%.
The number of heroin-related overdose deaths climbed in every region of the country from 2000 through 2013. The largest change in heroin overdose by region occurred in the Midwest, where the death rate rose from 0.4/100,000 in 2000 to 4.3 in 2013, an increase of 975%, said the NCHS report, which used data collected by the National Vital Statistics System.
Drug-poisoning deaths involving heroin have soared since 2000, and most of the increase occurred since 2010, according to a report from the National Center for Health Statistics.
From 2010 to 2013, the rate of heroin overdose deaths increased 163% for men, from a rate of 1.6/100,000 population in 2010 to 4.2 in 2013. For women, the death rate increased by 200%, from 0.4/100,000 in 2010 to 1.2/100,000 in 2013. From 2000 to 2010, however, the rate of increase was much slower, with the death rate increasing from 1.1 to 1.6 for men and from 0.2 to 0.4 for women.

The overall rate for heroin overdose from 2000 to 2013 increased from 0.7 to 2.7/100,000. Most of this increase occurred from 2010 to 2013: From 2000 to 2010, the death rate increased to only 1/100,000, a growth rate of 6%, but after 2010, the rate grew by 37% per year, the NCHS reported.
In 2013, non-Hispanic whites aged 18-44 years had the highest heroin poisoning death rate among measured racial/ethnic groups at 7/100,000. In 2000, older, non-Hispanic blacks aged 45-64 years had the highest death rate among the reported racial/ethnic groups at 2/100,000. The death rate for whites aged 18-44 in 2000 was 1.2/100,000, meaning that the death rate increased by 483% from 2000 to 2013. For non-Hispanic blacks aged 45-64, the death rate in 2013 was 4.9, an increase of 145%.
The number of heroin-related overdose deaths climbed in every region of the country from 2000 through 2013. The largest change in heroin overdose by region occurred in the Midwest, where the death rate rose from 0.4/100,000 in 2000 to 4.3 in 2013, an increase of 975%, said the NCHS report, which used data collected by the National Vital Statistics System.
Drug-poisoning deaths involving heroin have soared since 2000, and most of the increase occurred since 2010, according to a report from the National Center for Health Statistics.
From 2010 to 2013, the rate of heroin overdose deaths increased 163% for men, from a rate of 1.6/100,000 population in 2010 to 4.2 in 2013. For women, the death rate increased by 200%, from 0.4/100,000 in 2010 to 1.2/100,000 in 2013. From 2000 to 2010, however, the rate of increase was much slower, with the death rate increasing from 1.1 to 1.6 for men and from 0.2 to 0.4 for women.

The overall rate for heroin overdose from 2000 to 2013 increased from 0.7 to 2.7/100,000. Most of this increase occurred from 2010 to 2013: From 2000 to 2010, the death rate increased to only 1/100,000, a growth rate of 6%, but after 2010, the rate grew by 37% per year, the NCHS reported.
In 2013, non-Hispanic whites aged 18-44 years had the highest heroin poisoning death rate among measured racial/ethnic groups at 7/100,000. In 2000, older, non-Hispanic blacks aged 45-64 years had the highest death rate among the reported racial/ethnic groups at 2/100,000. The death rate for whites aged 18-44 in 2000 was 1.2/100,000, meaning that the death rate increased by 483% from 2000 to 2013. For non-Hispanic blacks aged 45-64, the death rate in 2013 was 4.9, an increase of 145%.
The number of heroin-related overdose deaths climbed in every region of the country from 2000 through 2013. The largest change in heroin overdose by region occurred in the Midwest, where the death rate rose from 0.4/100,000 in 2000 to 4.3 in 2013, an increase of 975%, said the NCHS report, which used data collected by the National Vital Statistics System.