User login
‘Phenomenal’ results with CAR T cells in R/R multiple myeloma
Patients with multiple myeloma that has continued to progress despite many lines of therapy have shown deep and durable responses to a new chimeric antigen receptor (CAR) T-cell therapy, idecabtagene vicleucel (ide-cel, under development by Bristol-Myers Squibb and Bluebird Bio).
An expert not involved in the trial described the results as “phenomenal.”
Krina Patel, MD, an associate professor in the department of lymphoma/myeloma at the University of Texas MD Anderson Cancer Center, Houston, said that “the response rate of 73% in a patient population with a median of six lines of therapy, and with one-third of those patients achieving a deep response of complete response or better, is phenomenal.”
“We are very excited as a myeloma community for this study of idecabtagene vicleucel for relapsed/refractory patients,” Dr. Patel said.
The new data on ide-cell, from a trial in 128 patients, were published Feb. 25 in the New England Journal of Medicine.
Lead investigator of the study Nikhil Munshi, MD, of Dana-Farber Cancer Institute, Boston, said: “The results of this trial represent a true turning point in the treatment of this disease. In my 30 years of treating myeloma, I have not seen any other therapy as effective in this group of patients.”
Both experts highlighted the poor prognosis for this population of relapsed/refractory patients. Recent decades have seen a flurry of new agents for myeloma, and there are now three main classes of agents: immunomodulatory agents, proteasome inhibitors, and anti-CD38 antibodies. Nevertheless, in some patients, the disease continues to progress. For patients who have failed all three classes of drugs, the median progression-free survival is about 3-4 months, with a median overall survival of 8-9 months.
Product is awaiting approval
Ide-cel is currently awaiting FDA approval, with a decision date slated for March 27.
Several CAR T-cell products are already marketed for use in certain leukemias and lymphomas, and there is another for use in multiple myeloma, ciltacabtagene autoleucel (cilta-cel, under development by Janssen), that is awaiting approval in Europe.
Strong and sustained responses
The trial involved 128 patients treated with ide-cel infusions. At the time of data cutoff for this report (Jan. 14, 2020), 62 patients remained in the primary study. Of the 128 treated patients, the median age was 61 years and the median time since diagnosis was 6 years. About half (51%) had a high tumor burden (≥50% bone marrow plasma cells), 39% had extramedullary disease, 16% had stage III disease, and 35% had a high-risk cytogenetic abnormality, defined as del(17p), t(4;14), or t(14;16).
Patients in the cohort had received a median of six previous antimyeloma regimens (range, 3-16), and most of the patients (120, 94%) had undergone autologous hematopoietic stem cell transplants. In addition, the majority of patients (84%) had disease that was triple refractory (to an immunomodulatory agent, a proteasome inhibitor, and an anti-CD38 antibody), 60% had disease that was penta exposed (to bortezomib, carfilzomib, lenalidomide, pomalidomide, and daratumumab), and 26% had disease that was penta refractory.
At a median follow-up of 13.3 months, 94 of 128 patients (73%) showed a response to therapy (P < .001), with 42 (33%) showing a complete or stringent complete response, and 67 patients (52%) showing a “very good partial response or better.”
Overall median progression-free survival was 8.8 months at the 450×106 dose but more than double that (20.2 months) for patients who achieved a complete or stringent complete response. Estimated median overall survival was 19.4 months, with an overall survival of 78% at 12 months. The authors noted that overall survival data are not yet mature.
After experiencing disease progression, 28 patients were retreated with ide-cel, with 6 patients showing a second response. The durations of response ranged from 1.9 to 6.8 months.
All patients in the cohort experienced adverse events, primarily grade 3 or 4 events that occurred in 127 patients (99%). The most common events reported were hematologic toxicities, including neutropenia in 114 patients (89%), anemia in 77 (60%), and thrombocytopenia in 67 (52%), and were at least partially related to the lymphodepleting chemotherapy administered before ide-cel infusion, the authors note. Cytokine-release syndrome occurred in 107 patients (84%), primarily grade 1 or 2.
“Results of the KarMMa study support substantial antitumor activity for ide-cel across a target dose range of 150×106 to 450×106 CAR+ T cells,” the authors conclude. “The 450×106 dose appeared to be somewhat more effective than the other doses.”
New option?
“What this study further highlights is that higher cell dose tends to increase cell expansion, which correlates to improved response and duration of response,” said Dr. Patel.
Importantly, multiple vulnerable subgroups experienced impressive outcomes, such as those who are older or with high risk or extramedullary disease, she noted.
“My patients who have undergone this therapy, albeit on other clinical trials, all say that their quality of life during this time of remission is priceless,” Dr. Patel added. “The is the first therapy in the relapsed/refractory setting that allows patients to have a significant chemo-free period. We need to find more ways to do this for our patients.”
The study was supported by Bluebird Bio and Bristol-Myers Squibb. Dr. Patel has served on the advisory board for Janssen and Bristol-Myers Squibb. She also reports a speaking engagement with Oncopeptides. Dr. Munshi acts as a consultant for several pharmaceutical companies, and many coauthors also have relationships with industry, as listed in the original article.
A version of this article first appeared on Medscape.com.
Patients with multiple myeloma that has continued to progress despite many lines of therapy have shown deep and durable responses to a new chimeric antigen receptor (CAR) T-cell therapy, idecabtagene vicleucel (ide-cel, under development by Bristol-Myers Squibb and Bluebird Bio).
An expert not involved in the trial described the results as “phenomenal.”
Krina Patel, MD, an associate professor in the department of lymphoma/myeloma at the University of Texas MD Anderson Cancer Center, Houston, said that “the response rate of 73% in a patient population with a median of six lines of therapy, and with one-third of those patients achieving a deep response of complete response or better, is phenomenal.”
“We are very excited as a myeloma community for this study of idecabtagene vicleucel for relapsed/refractory patients,” Dr. Patel said.
The new data on ide-cell, from a trial in 128 patients, were published Feb. 25 in the New England Journal of Medicine.
Lead investigator of the study Nikhil Munshi, MD, of Dana-Farber Cancer Institute, Boston, said: “The results of this trial represent a true turning point in the treatment of this disease. In my 30 years of treating myeloma, I have not seen any other therapy as effective in this group of patients.”
Both experts highlighted the poor prognosis for this population of relapsed/refractory patients. Recent decades have seen a flurry of new agents for myeloma, and there are now three main classes of agents: immunomodulatory agents, proteasome inhibitors, and anti-CD38 antibodies. Nevertheless, in some patients, the disease continues to progress. For patients who have failed all three classes of drugs, the median progression-free survival is about 3-4 months, with a median overall survival of 8-9 months.
Product is awaiting approval
Ide-cel is currently awaiting FDA approval, with a decision date slated for March 27.
Several CAR T-cell products are already marketed for use in certain leukemias and lymphomas, and there is another for use in multiple myeloma, ciltacabtagene autoleucel (cilta-cel, under development by Janssen), that is awaiting approval in Europe.
Strong and sustained responses
The trial involved 128 patients treated with ide-cel infusions. At the time of data cutoff for this report (Jan. 14, 2020), 62 patients remained in the primary study. Of the 128 treated patients, the median age was 61 years and the median time since diagnosis was 6 years. About half (51%) had a high tumor burden (≥50% bone marrow plasma cells), 39% had extramedullary disease, 16% had stage III disease, and 35% had a high-risk cytogenetic abnormality, defined as del(17p), t(4;14), or t(14;16).
Patients in the cohort had received a median of six previous antimyeloma regimens (range, 3-16), and most of the patients (120, 94%) had undergone autologous hematopoietic stem cell transplants. In addition, the majority of patients (84%) had disease that was triple refractory (to an immunomodulatory agent, a proteasome inhibitor, and an anti-CD38 antibody), 60% had disease that was penta exposed (to bortezomib, carfilzomib, lenalidomide, pomalidomide, and daratumumab), and 26% had disease that was penta refractory.
At a median follow-up of 13.3 months, 94 of 128 patients (73%) showed a response to therapy (P < .001), with 42 (33%) showing a complete or stringent complete response, and 67 patients (52%) showing a “very good partial response or better.”
Overall median progression-free survival was 8.8 months at the 450×106 dose but more than double that (20.2 months) for patients who achieved a complete or stringent complete response. Estimated median overall survival was 19.4 months, with an overall survival of 78% at 12 months. The authors noted that overall survival data are not yet mature.
After experiencing disease progression, 28 patients were retreated with ide-cel, with 6 patients showing a second response. The durations of response ranged from 1.9 to 6.8 months.
All patients in the cohort experienced adverse events, primarily grade 3 or 4 events that occurred in 127 patients (99%). The most common events reported were hematologic toxicities, including neutropenia in 114 patients (89%), anemia in 77 (60%), and thrombocytopenia in 67 (52%), and were at least partially related to the lymphodepleting chemotherapy administered before ide-cel infusion, the authors note. Cytokine-release syndrome occurred in 107 patients (84%), primarily grade 1 or 2.
“Results of the KarMMa study support substantial antitumor activity for ide-cel across a target dose range of 150×106 to 450×106 CAR+ T cells,” the authors conclude. “The 450×106 dose appeared to be somewhat more effective than the other doses.”
New option?
“What this study further highlights is that higher cell dose tends to increase cell expansion, which correlates to improved response and duration of response,” said Dr. Patel.
Importantly, multiple vulnerable subgroups experienced impressive outcomes, such as those who are older or with high risk or extramedullary disease, she noted.
“My patients who have undergone this therapy, albeit on other clinical trials, all say that their quality of life during this time of remission is priceless,” Dr. Patel added. “The is the first therapy in the relapsed/refractory setting that allows patients to have a significant chemo-free period. We need to find more ways to do this for our patients.”
The study was supported by Bluebird Bio and Bristol-Myers Squibb. Dr. Patel has served on the advisory board for Janssen and Bristol-Myers Squibb. She also reports a speaking engagement with Oncopeptides. Dr. Munshi acts as a consultant for several pharmaceutical companies, and many coauthors also have relationships with industry, as listed in the original article.
A version of this article first appeared on Medscape.com.
Patients with multiple myeloma that has continued to progress despite many lines of therapy have shown deep and durable responses to a new chimeric antigen receptor (CAR) T-cell therapy, idecabtagene vicleucel (ide-cel, under development by Bristol-Myers Squibb and Bluebird Bio).
An expert not involved in the trial described the results as “phenomenal.”
Krina Patel, MD, an associate professor in the department of lymphoma/myeloma at the University of Texas MD Anderson Cancer Center, Houston, said that “the response rate of 73% in a patient population with a median of six lines of therapy, and with one-third of those patients achieving a deep response of complete response or better, is phenomenal.”
“We are very excited as a myeloma community for this study of idecabtagene vicleucel for relapsed/refractory patients,” Dr. Patel said.
The new data on ide-cell, from a trial in 128 patients, were published Feb. 25 in the New England Journal of Medicine.
Lead investigator of the study Nikhil Munshi, MD, of Dana-Farber Cancer Institute, Boston, said: “The results of this trial represent a true turning point in the treatment of this disease. In my 30 years of treating myeloma, I have not seen any other therapy as effective in this group of patients.”
Both experts highlighted the poor prognosis for this population of relapsed/refractory patients. Recent decades have seen a flurry of new agents for myeloma, and there are now three main classes of agents: immunomodulatory agents, proteasome inhibitors, and anti-CD38 antibodies. Nevertheless, in some patients, the disease continues to progress. For patients who have failed all three classes of drugs, the median progression-free survival is about 3-4 months, with a median overall survival of 8-9 months.
Product is awaiting approval
Ide-cel is currently awaiting FDA approval, with a decision date slated for March 27.
Several CAR T-cell products are already marketed for use in certain leukemias and lymphomas, and there is another for use in multiple myeloma, ciltacabtagene autoleucel (cilta-cel, under development by Janssen), that is awaiting approval in Europe.
Strong and sustained responses
The trial involved 128 patients treated with ide-cel infusions. At the time of data cutoff for this report (Jan. 14, 2020), 62 patients remained in the primary study. Of the 128 treated patients, the median age was 61 years and the median time since diagnosis was 6 years. About half (51%) had a high tumor burden (≥50% bone marrow plasma cells), 39% had extramedullary disease, 16% had stage III disease, and 35% had a high-risk cytogenetic abnormality, defined as del(17p), t(4;14), or t(14;16).
Patients in the cohort had received a median of six previous antimyeloma regimens (range, 3-16), and most of the patients (120, 94%) had undergone autologous hematopoietic stem cell transplants. In addition, the majority of patients (84%) had disease that was triple refractory (to an immunomodulatory agent, a proteasome inhibitor, and an anti-CD38 antibody), 60% had disease that was penta exposed (to bortezomib, carfilzomib, lenalidomide, pomalidomide, and daratumumab), and 26% had disease that was penta refractory.
At a median follow-up of 13.3 months, 94 of 128 patients (73%) showed a response to therapy (P < .001), with 42 (33%) showing a complete or stringent complete response, and 67 patients (52%) showing a “very good partial response or better.”
Overall median progression-free survival was 8.8 months at the 450×106 dose but more than double that (20.2 months) for patients who achieved a complete or stringent complete response. Estimated median overall survival was 19.4 months, with an overall survival of 78% at 12 months. The authors noted that overall survival data are not yet mature.
After experiencing disease progression, 28 patients were retreated with ide-cel, with 6 patients showing a second response. The durations of response ranged from 1.9 to 6.8 months.
All patients in the cohort experienced adverse events, primarily grade 3 or 4 events that occurred in 127 patients (99%). The most common events reported were hematologic toxicities, including neutropenia in 114 patients (89%), anemia in 77 (60%), and thrombocytopenia in 67 (52%), and were at least partially related to the lymphodepleting chemotherapy administered before ide-cel infusion, the authors note. Cytokine-release syndrome occurred in 107 patients (84%), primarily grade 1 or 2.
“Results of the KarMMa study support substantial antitumor activity for ide-cel across a target dose range of 150×106 to 450×106 CAR+ T cells,” the authors conclude. “The 450×106 dose appeared to be somewhat more effective than the other doses.”
New option?
“What this study further highlights is that higher cell dose tends to increase cell expansion, which correlates to improved response and duration of response,” said Dr. Patel.
Importantly, multiple vulnerable subgroups experienced impressive outcomes, such as those who are older or with high risk or extramedullary disease, she noted.
“My patients who have undergone this therapy, albeit on other clinical trials, all say that their quality of life during this time of remission is priceless,” Dr. Patel added. “The is the first therapy in the relapsed/refractory setting that allows patients to have a significant chemo-free period. We need to find more ways to do this for our patients.”
The study was supported by Bluebird Bio and Bristol-Myers Squibb. Dr. Patel has served on the advisory board for Janssen and Bristol-Myers Squibb. She also reports a speaking engagement with Oncopeptides. Dr. Munshi acts as a consultant for several pharmaceutical companies, and many coauthors also have relationships with industry, as listed in the original article.
A version of this article first appeared on Medscape.com.
Rural women receive antibiotics for longer than necessary for UTIs
Women living in rural areas were significantly more likely than were those in urban areas to receive inappropriate antibiotic prescriptions for urinary tract infections, based on data from an observational cohort study of more than 600,000 women.
Uncomplicated urinary tract infections (UTIs) are common among otherwise healthy women in the United States, and certain antibiotics are recommended as first-line therapy, wrote Abbye W. Clark, MD, of Washington University, St. Louis, and colleagues.
“However, the majority of antibiotic prescriptions for uncomplicated UTI are suboptimal because they are written for nonrecommended agents and durations,” they said.
Addressing rural health disparities has become a focus in the United States, and previous studies of respiratory tract infections have shown differences in antibiotic prescribing based on geographic region; “however, no large-scale studies have evaluated rural-urban differences in inappropriate outpatient prescribing for UTI,” they added.
In a study published in Infection Control & Hospital Epidemiology, the researchers identified 670,450 women aged 18-44 years who received oral antibiotics for uncomplicated UTIs between 2010 to 2015, using a commercial insurance database to determine diagnosis and antibiotic prescription information. Women were defined as urban if they lived in a metropolitan statistical area of at least 50,000 inhabitants (86.2%); all other women were defined as rural (13.8%). The median age was 30 years for both groups.
Overall, 46.7% of the women received prescriptions for inappropriate antibiotics, and 76.1% received antibiotics for inappropriate durations.
Antibiotics and durations were defined as appropriate or inappropriate based on current clinical guidelines. “We classified first-line agents (nitrofurantoin, TMP-SMX, fosfomycin) as appropriate and non–first-line agents (fluoroquinolones, beta-lactams) as inappropriate,” the researchers said.
The regimens classified as appropriate duration were “nitrofurantoin 5-day regimen, TMP-SMX (including TMP monotherapy) 3-day regimen, fosfomycin 1-day regimen, fluoroquinolones 3-day regimen, and beta-lactams 3- to 7-day regimen. All other regimens were classified as inappropriate duration,” they noted.
More rural women receive long-duration antibiotics
In a multivariate analysis, similar percentages of antibiotics for rural and urban women consisted of inappropriate agents (45.9% vs. 46.9%) including use of fluoroquinolones (41.0% vs. 41.7%) and beta-lactams (4.8% vs. 5.0%).
However, across all antibiotics, women in rural areas were more likely than were women in urban areas to receive prescriptions for inappropriately long durations (83.9% vs. 75.9%, adjusted risk ratio 1.10).
The percentage of women who received inappropriate antibiotic agents was not significantly different based on geographic region of the country.
From 2011 to 2015, the quarterly proportion of women overall who received inappropriate agents and antibiotics for inappropriate durations decreased slightly (48.5% to 43.7% and 78.3% to 73.4%, respectively), the researchers noted.
The study findings were limited by several factors including the potentially lenient definition of antibiotic duration, a study population that disproportionately oversampled from the South and undersampled from the West, use of ZIP codes to determine rural vs. urban status, lack of data on race and income, and lack of access to urine culture results, the researchers noted.
However, “our study identified rural-urban differences in antibiotic prescribing, including an actionable disparity in the duration of antibiotics that disproportionately affects women who live in rural locations,” they said.
“Given the large quantity of inappropriate prescriptions annually in the U.S., as well as the negative patient- and society-level consequences of unnecessary exposure to antibiotics, antimicrobial stewardship interventions are needed to improve outpatient UTI antibiotic prescribing, particularly in rural settings,” they concluded.
Data support need for education and stewardship
“This manuscript provides valuable information to all women’s health providers regarding the importance of antibiotic stewardship,” David M. Jaspan, DO, and Natasha Abdullah, MD, Einstein Medical Center, Philadelphia, said in an interview. Whether urban or rural, over 45% of the patients received inappropriate non–first-line treatment and 76% of the prescriptions were for an inappropriate duration (98.8% for longer than recommended), they emphasized.
“The potential negative impact of antibiotic resistance, coupled with the potential for increased side effects, should prompt providers to ensure that when treating uncomplicated UTIs in women, that the choice of treatment and the duration of treatment is tailored to the patient’s needs,” the Dr. Jaspan and Dr. Abdullah said.
To improve antibiotic prescribing, especially at the local and regional level, “We encourage providers to familiarize themselves with local information as it pertains to known resistance when prescribing empiric treatment regimens for uncomplicated UTIs,” they said.
The study was supported by the National Center for Advancing Translational Sciences at the National Institutes of Health. Lead author Dr. Clark, as well as Dr. Jaspan and Dr. Abdullah, had no financial conflicts to disclose.
Women living in rural areas were significantly more likely than were those in urban areas to receive inappropriate antibiotic prescriptions for urinary tract infections, based on data from an observational cohort study of more than 600,000 women.
Uncomplicated urinary tract infections (UTIs) are common among otherwise healthy women in the United States, and certain antibiotics are recommended as first-line therapy, wrote Abbye W. Clark, MD, of Washington University, St. Louis, and colleagues.
“However, the majority of antibiotic prescriptions for uncomplicated UTI are suboptimal because they are written for nonrecommended agents and durations,” they said.
Addressing rural health disparities has become a focus in the United States, and previous studies of respiratory tract infections have shown differences in antibiotic prescribing based on geographic region; “however, no large-scale studies have evaluated rural-urban differences in inappropriate outpatient prescribing for UTI,” they added.
In a study published in Infection Control & Hospital Epidemiology, the researchers identified 670,450 women aged 18-44 years who received oral antibiotics for uncomplicated UTIs between 2010 to 2015, using a commercial insurance database to determine diagnosis and antibiotic prescription information. Women were defined as urban if they lived in a metropolitan statistical area of at least 50,000 inhabitants (86.2%); all other women were defined as rural (13.8%). The median age was 30 years for both groups.
Overall, 46.7% of the women received prescriptions for inappropriate antibiotics, and 76.1% received antibiotics for inappropriate durations.
Antibiotics and durations were defined as appropriate or inappropriate based on current clinical guidelines. “We classified first-line agents (nitrofurantoin, TMP-SMX, fosfomycin) as appropriate and non–first-line agents (fluoroquinolones, beta-lactams) as inappropriate,” the researchers said.
The regimens classified as appropriate duration were “nitrofurantoin 5-day regimen, TMP-SMX (including TMP monotherapy) 3-day regimen, fosfomycin 1-day regimen, fluoroquinolones 3-day regimen, and beta-lactams 3- to 7-day regimen. All other regimens were classified as inappropriate duration,” they noted.
More rural women receive long-duration antibiotics
In a multivariate analysis, similar percentages of antibiotics for rural and urban women consisted of inappropriate agents (45.9% vs. 46.9%) including use of fluoroquinolones (41.0% vs. 41.7%) and beta-lactams (4.8% vs. 5.0%).
However, across all antibiotics, women in rural areas were more likely than were women in urban areas to receive prescriptions for inappropriately long durations (83.9% vs. 75.9%, adjusted risk ratio 1.10).
The percentage of women who received inappropriate antibiotic agents was not significantly different based on geographic region of the country.
From 2011 to 2015, the quarterly proportion of women overall who received inappropriate agents and antibiotics for inappropriate durations decreased slightly (48.5% to 43.7% and 78.3% to 73.4%, respectively), the researchers noted.
The study findings were limited by several factors including the potentially lenient definition of antibiotic duration, a study population that disproportionately oversampled from the South and undersampled from the West, use of ZIP codes to determine rural vs. urban status, lack of data on race and income, and lack of access to urine culture results, the researchers noted.
However, “our study identified rural-urban differences in antibiotic prescribing, including an actionable disparity in the duration of antibiotics that disproportionately affects women who live in rural locations,” they said.
“Given the large quantity of inappropriate prescriptions annually in the U.S., as well as the negative patient- and society-level consequences of unnecessary exposure to antibiotics, antimicrobial stewardship interventions are needed to improve outpatient UTI antibiotic prescribing, particularly in rural settings,” they concluded.
Data support need for education and stewardship
“This manuscript provides valuable information to all women’s health providers regarding the importance of antibiotic stewardship,” David M. Jaspan, DO, and Natasha Abdullah, MD, Einstein Medical Center, Philadelphia, said in an interview. Whether urban or rural, over 45% of the patients received inappropriate non–first-line treatment and 76% of the prescriptions were for an inappropriate duration (98.8% for longer than recommended), they emphasized.
“The potential negative impact of antibiotic resistance, coupled with the potential for increased side effects, should prompt providers to ensure that when treating uncomplicated UTIs in women, that the choice of treatment and the duration of treatment is tailored to the patient’s needs,” the Dr. Jaspan and Dr. Abdullah said.
To improve antibiotic prescribing, especially at the local and regional level, “We encourage providers to familiarize themselves with local information as it pertains to known resistance when prescribing empiric treatment regimens for uncomplicated UTIs,” they said.
The study was supported by the National Center for Advancing Translational Sciences at the National Institutes of Health. Lead author Dr. Clark, as well as Dr. Jaspan and Dr. Abdullah, had no financial conflicts to disclose.
Women living in rural areas were significantly more likely than were those in urban areas to receive inappropriate antibiotic prescriptions for urinary tract infections, based on data from an observational cohort study of more than 600,000 women.
Uncomplicated urinary tract infections (UTIs) are common among otherwise healthy women in the United States, and certain antibiotics are recommended as first-line therapy, wrote Abbye W. Clark, MD, of Washington University, St. Louis, and colleagues.
“However, the majority of antibiotic prescriptions for uncomplicated UTI are suboptimal because they are written for nonrecommended agents and durations,” they said.
Addressing rural health disparities has become a focus in the United States, and previous studies of respiratory tract infections have shown differences in antibiotic prescribing based on geographic region; “however, no large-scale studies have evaluated rural-urban differences in inappropriate outpatient prescribing for UTI,” they added.
In a study published in Infection Control & Hospital Epidemiology, the researchers identified 670,450 women aged 18-44 years who received oral antibiotics for uncomplicated UTIs between 2010 to 2015, using a commercial insurance database to determine diagnosis and antibiotic prescription information. Women were defined as urban if they lived in a metropolitan statistical area of at least 50,000 inhabitants (86.2%); all other women were defined as rural (13.8%). The median age was 30 years for both groups.
Overall, 46.7% of the women received prescriptions for inappropriate antibiotics, and 76.1% received antibiotics for inappropriate durations.
Antibiotics and durations were defined as appropriate or inappropriate based on current clinical guidelines. “We classified first-line agents (nitrofurantoin, TMP-SMX, fosfomycin) as appropriate and non–first-line agents (fluoroquinolones, beta-lactams) as inappropriate,” the researchers said.
The regimens classified as appropriate duration were “nitrofurantoin 5-day regimen, TMP-SMX (including TMP monotherapy) 3-day regimen, fosfomycin 1-day regimen, fluoroquinolones 3-day regimen, and beta-lactams 3- to 7-day regimen. All other regimens were classified as inappropriate duration,” they noted.
More rural women receive long-duration antibiotics
In a multivariate analysis, similar percentages of antibiotics for rural and urban women consisted of inappropriate agents (45.9% vs. 46.9%) including use of fluoroquinolones (41.0% vs. 41.7%) and beta-lactams (4.8% vs. 5.0%).
However, across all antibiotics, women in rural areas were more likely than were women in urban areas to receive prescriptions for inappropriately long durations (83.9% vs. 75.9%, adjusted risk ratio 1.10).
The percentage of women who received inappropriate antibiotic agents was not significantly different based on geographic region of the country.
From 2011 to 2015, the quarterly proportion of women overall who received inappropriate agents and antibiotics for inappropriate durations decreased slightly (48.5% to 43.7% and 78.3% to 73.4%, respectively), the researchers noted.
The study findings were limited by several factors including the potentially lenient definition of antibiotic duration, a study population that disproportionately oversampled from the South and undersampled from the West, use of ZIP codes to determine rural vs. urban status, lack of data on race and income, and lack of access to urine culture results, the researchers noted.
However, “our study identified rural-urban differences in antibiotic prescribing, including an actionable disparity in the duration of antibiotics that disproportionately affects women who live in rural locations,” they said.
“Given the large quantity of inappropriate prescriptions annually in the U.S., as well as the negative patient- and society-level consequences of unnecessary exposure to antibiotics, antimicrobial stewardship interventions are needed to improve outpatient UTI antibiotic prescribing, particularly in rural settings,” they concluded.
Data support need for education and stewardship
“This manuscript provides valuable information to all women’s health providers regarding the importance of antibiotic stewardship,” David M. Jaspan, DO, and Natasha Abdullah, MD, Einstein Medical Center, Philadelphia, said in an interview. Whether urban or rural, over 45% of the patients received inappropriate non–first-line treatment and 76% of the prescriptions were for an inappropriate duration (98.8% for longer than recommended), they emphasized.
“The potential negative impact of antibiotic resistance, coupled with the potential for increased side effects, should prompt providers to ensure that when treating uncomplicated UTIs in women, that the choice of treatment and the duration of treatment is tailored to the patient’s needs,” the Dr. Jaspan and Dr. Abdullah said.
To improve antibiotic prescribing, especially at the local and regional level, “We encourage providers to familiarize themselves with local information as it pertains to known resistance when prescribing empiric treatment regimens for uncomplicated UTIs,” they said.
The study was supported by the National Center for Advancing Translational Sciences at the National Institutes of Health. Lead author Dr. Clark, as well as Dr. Jaspan and Dr. Abdullah, had no financial conflicts to disclose.
FROM INFECTION CONTROL & HOSPITAL EPIDEMIOLOGY
No vascular benefit of testosterone over exercise in aging men
Exercise training – but not testosterone therapy – improved vascular health in aging men with widening midsections and low to normal testosterone, new research suggests.
“Previous studies have suggested that men with higher levels of testosterone, who were more physically active, might have better health outcomes,” Bu Beng Yeap, MBBS, PhD, University of Western Australia, Perth, said in an interview. “We formulated the hypothesis that the combination of testosterone treatment and exercise training would improve the health of arteries more than either alone.”
To test this hypothesis, the investigators randomly assigned 80 men, aged 50-70 years, to 12 weeks of 5% testosterone cream 2 mL applied daily or placebo plus a supervised exercise program that included machine-based resistance and aerobic (cycling) exercises two to three times a week or no additional exercise.
The men (mean age, 59 years) had low-normal testosterone (6-14 nmol/L), a waist circumference of at least 95 cm (37.4 inches), and no known cardiovascular disease (CVD), type 1 diabetes, or other clinically significant illnesses. Current smokers and men on testosterone or medications that would alter testosterone levels were also excluded.
High-resolution ultrasound of the brachial artery was used to assess flow-mediated dilation (FMD) and sublingual glyceryl trinitrate (GTN) responses. FMD has been shown to be predictive of CVD risk, with a 1% increase in FMD associated with a 9%-13% decrease in future CVD events.
Based on participants’ daily dairies, testosterone adherence was 97.6%. Exercise adherence was 96.5% for twice-weekly attendance and 80.0% for thrice-weekly attendance, with no between-group differences.
As reported Feb. 22, 2021, in Hypertension, testosterone levels increased, on average, 3.0 nmol/L in both testosterone groups by week 12 (P = .003). In all, 62% of these men had levels of the hormone exceeding 14 nmol/L, compared with 29% of those receiving placebo.
Testosterone levels improved with exercise training plus placebo by 0.9 nmol/L, but fell with no exercise and placebo by 0.9 nmol/L.
In terms of vascular function, exercise training increased FMD when expressed as both the delta change (mm; P = .004) and relative rise from baseline diameter (%; P = .033).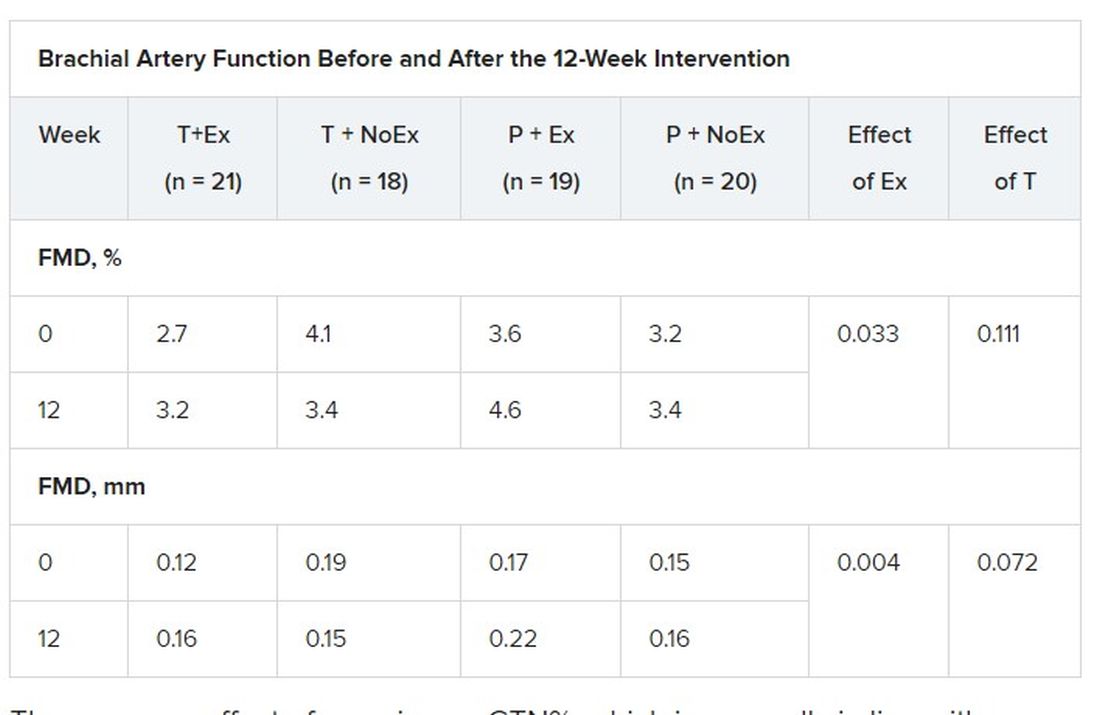
There was no effect of exercise on GTN%, which is generally in line with exercise literature indicating that shear-mediated adaptations in response to episodic exercise occur largely in endothelial cells, the authors noted.
Testosterone did not affect any measures of FMD nor was there an effect on GTN response, despite previous evidence that lower testosterone doses might enhance smooth muscle function.
“Our main finding was that testosterone – at this dose over this duration of treatment – did not have a beneficial effect on artery health, nor did it enhance the effect of exercise,” said Dr. Yeap, who is also president of the Endocrine Society of Australia. “For middle-aged and older men wanting to improve the health of their arteries, exercise is better than testosterone!”
Shalender Bhasin, MBBS, director of research programs in men’s health, aging, and metabolism at Brigham and Women’s Hospital and professor of medicine at Harvard Medical School, both in Boston, said the study is interesting from a mechanistic perspective and adds to the overall body of evidence on how testosterone affects performance, but was narrowly focused.
“They looked at very specific markers and what they’re showing is that this is not the mechanism by which testosterone improves performance,” he said. “That may be so, but it doesn’t negate the finding that testosterone improves endurance and has other vascular effects: it increases capillarity, increases blood flow to the tissues, and improves myocardial function.”
Although well done, the study doesn’t get at the larger question of whether testosterone increases cardiovascular risk, observed Dr. Bhasin. “None of the randomized studies have been large enough or long enough to determine the effect on cardiovascular events rates. There’s a lot of argument on both sides but we need some data to address that.”
The 6,000-patient TRAVERSE trial is specifically looking at long-term major cardiovascular events with topical testosterone, compared with placebo, in hypogonadal men aged 45-80 years age who have evidence of or are at increased risk for CVD. The study, which is set to be completed in April 2022, should also provide information on fracture risk in these men, said Dr. Bhasin, one of the trial’s principal investigators and lead author of the Endocrine Society’s 2018 clinical practice guideline on testosterone therapy for hypogonadism in men.
William Evans, MD, adjunct professor of human nutrition, University of California, Berkley, said in an interview that the positive effects of testosterone occur at much lower doses in men and women who are hypogonadal but, in this particular population, exercise is the key and the major recommendation.
“Testosterone has been overprescribed and overadvertised for essentially a lifetime of sedentary living, and it’s advertised as a way to get all that back without having to work for it,” he said. “Exercise has a profound and positive effect on control of blood pressure, function, and strength, and testosterone may only affect in people who are sick, people who have really low levels.”
The study was funded by the Heart Foundation of Australia. Lawley Pharmaceuticals provided the study medication and placebo. Dr. Yeap has received speaker honoraria and conference support from Bayer, Eli Lilly, and Besins Healthcare; research support from Bayer, Lily, and Lawley; and served as an adviser for Lily, Besins Healthcare, Ferring, and Lawley. Dr. Shalender reports consultation or advisement for GTx, Pfizer, and TAP; grant or other research support from Solvay and GlaxoSmithKline; and honoraria from Solvay and Auxilium. Dr. Evans reported having no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
Exercise training – but not testosterone therapy – improved vascular health in aging men with widening midsections and low to normal testosterone, new research suggests.
“Previous studies have suggested that men with higher levels of testosterone, who were more physically active, might have better health outcomes,” Bu Beng Yeap, MBBS, PhD, University of Western Australia, Perth, said in an interview. “We formulated the hypothesis that the combination of testosterone treatment and exercise training would improve the health of arteries more than either alone.”
To test this hypothesis, the investigators randomly assigned 80 men, aged 50-70 years, to 12 weeks of 5% testosterone cream 2 mL applied daily or placebo plus a supervised exercise program that included machine-based resistance and aerobic (cycling) exercises two to three times a week or no additional exercise.
The men (mean age, 59 years) had low-normal testosterone (6-14 nmol/L), a waist circumference of at least 95 cm (37.4 inches), and no known cardiovascular disease (CVD), type 1 diabetes, or other clinically significant illnesses. Current smokers and men on testosterone or medications that would alter testosterone levels were also excluded.
High-resolution ultrasound of the brachial artery was used to assess flow-mediated dilation (FMD) and sublingual glyceryl trinitrate (GTN) responses. FMD has been shown to be predictive of CVD risk, with a 1% increase in FMD associated with a 9%-13% decrease in future CVD events.
Based on participants’ daily dairies, testosterone adherence was 97.6%. Exercise adherence was 96.5% for twice-weekly attendance and 80.0% for thrice-weekly attendance, with no between-group differences.
As reported Feb. 22, 2021, in Hypertension, testosterone levels increased, on average, 3.0 nmol/L in both testosterone groups by week 12 (P = .003). In all, 62% of these men had levels of the hormone exceeding 14 nmol/L, compared with 29% of those receiving placebo.
Testosterone levels improved with exercise training plus placebo by 0.9 nmol/L, but fell with no exercise and placebo by 0.9 nmol/L.
In terms of vascular function, exercise training increased FMD when expressed as both the delta change (mm; P = .004) and relative rise from baseline diameter (%; P = .033).
There was no effect of exercise on GTN%, which is generally in line with exercise literature indicating that shear-mediated adaptations in response to episodic exercise occur largely in endothelial cells, the authors noted.
Testosterone did not affect any measures of FMD nor was there an effect on GTN response, despite previous evidence that lower testosterone doses might enhance smooth muscle function.
“Our main finding was that testosterone – at this dose over this duration of treatment – did not have a beneficial effect on artery health, nor did it enhance the effect of exercise,” said Dr. Yeap, who is also president of the Endocrine Society of Australia. “For middle-aged and older men wanting to improve the health of their arteries, exercise is better than testosterone!”
Shalender Bhasin, MBBS, director of research programs in men’s health, aging, and metabolism at Brigham and Women’s Hospital and professor of medicine at Harvard Medical School, both in Boston, said the study is interesting from a mechanistic perspective and adds to the overall body of evidence on how testosterone affects performance, but was narrowly focused.
“They looked at very specific markers and what they’re showing is that this is not the mechanism by which testosterone improves performance,” he said. “That may be so, but it doesn’t negate the finding that testosterone improves endurance and has other vascular effects: it increases capillarity, increases blood flow to the tissues, and improves myocardial function.”
Although well done, the study doesn’t get at the larger question of whether testosterone increases cardiovascular risk, observed Dr. Bhasin. “None of the randomized studies have been large enough or long enough to determine the effect on cardiovascular events rates. There’s a lot of argument on both sides but we need some data to address that.”
The 6,000-patient TRAVERSE trial is specifically looking at long-term major cardiovascular events with topical testosterone, compared with placebo, in hypogonadal men aged 45-80 years age who have evidence of or are at increased risk for CVD. The study, which is set to be completed in April 2022, should also provide information on fracture risk in these men, said Dr. Bhasin, one of the trial’s principal investigators and lead author of the Endocrine Society’s 2018 clinical practice guideline on testosterone therapy for hypogonadism in men.
William Evans, MD, adjunct professor of human nutrition, University of California, Berkley, said in an interview that the positive effects of testosterone occur at much lower doses in men and women who are hypogonadal but, in this particular population, exercise is the key and the major recommendation.
“Testosterone has been overprescribed and overadvertised for essentially a lifetime of sedentary living, and it’s advertised as a way to get all that back without having to work for it,” he said. “Exercise has a profound and positive effect on control of blood pressure, function, and strength, and testosterone may only affect in people who are sick, people who have really low levels.”
The study was funded by the Heart Foundation of Australia. Lawley Pharmaceuticals provided the study medication and placebo. Dr. Yeap has received speaker honoraria and conference support from Bayer, Eli Lilly, and Besins Healthcare; research support from Bayer, Lily, and Lawley; and served as an adviser for Lily, Besins Healthcare, Ferring, and Lawley. Dr. Shalender reports consultation or advisement for GTx, Pfizer, and TAP; grant or other research support from Solvay and GlaxoSmithKline; and honoraria from Solvay and Auxilium. Dr. Evans reported having no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
Exercise training – but not testosterone therapy – improved vascular health in aging men with widening midsections and low to normal testosterone, new research suggests.
“Previous studies have suggested that men with higher levels of testosterone, who were more physically active, might have better health outcomes,” Bu Beng Yeap, MBBS, PhD, University of Western Australia, Perth, said in an interview. “We formulated the hypothesis that the combination of testosterone treatment and exercise training would improve the health of arteries more than either alone.”
To test this hypothesis, the investigators randomly assigned 80 men, aged 50-70 years, to 12 weeks of 5% testosterone cream 2 mL applied daily or placebo plus a supervised exercise program that included machine-based resistance and aerobic (cycling) exercises two to three times a week or no additional exercise.
The men (mean age, 59 years) had low-normal testosterone (6-14 nmol/L), a waist circumference of at least 95 cm (37.4 inches), and no known cardiovascular disease (CVD), type 1 diabetes, or other clinically significant illnesses. Current smokers and men on testosterone or medications that would alter testosterone levels were also excluded.
High-resolution ultrasound of the brachial artery was used to assess flow-mediated dilation (FMD) and sublingual glyceryl trinitrate (GTN) responses. FMD has been shown to be predictive of CVD risk, with a 1% increase in FMD associated with a 9%-13% decrease in future CVD events.
Based on participants’ daily dairies, testosterone adherence was 97.6%. Exercise adherence was 96.5% for twice-weekly attendance and 80.0% for thrice-weekly attendance, with no between-group differences.
As reported Feb. 22, 2021, in Hypertension, testosterone levels increased, on average, 3.0 nmol/L in both testosterone groups by week 12 (P = .003). In all, 62% of these men had levels of the hormone exceeding 14 nmol/L, compared with 29% of those receiving placebo.
Testosterone levels improved with exercise training plus placebo by 0.9 nmol/L, but fell with no exercise and placebo by 0.9 nmol/L.
In terms of vascular function, exercise training increased FMD when expressed as both the delta change (mm; P = .004) and relative rise from baseline diameter (%; P = .033).
There was no effect of exercise on GTN%, which is generally in line with exercise literature indicating that shear-mediated adaptations in response to episodic exercise occur largely in endothelial cells, the authors noted.
Testosterone did not affect any measures of FMD nor was there an effect on GTN response, despite previous evidence that lower testosterone doses might enhance smooth muscle function.
“Our main finding was that testosterone – at this dose over this duration of treatment – did not have a beneficial effect on artery health, nor did it enhance the effect of exercise,” said Dr. Yeap, who is also president of the Endocrine Society of Australia. “For middle-aged and older men wanting to improve the health of their arteries, exercise is better than testosterone!”
Shalender Bhasin, MBBS, director of research programs in men’s health, aging, and metabolism at Brigham and Women’s Hospital and professor of medicine at Harvard Medical School, both in Boston, said the study is interesting from a mechanistic perspective and adds to the overall body of evidence on how testosterone affects performance, but was narrowly focused.
“They looked at very specific markers and what they’re showing is that this is not the mechanism by which testosterone improves performance,” he said. “That may be so, but it doesn’t negate the finding that testosterone improves endurance and has other vascular effects: it increases capillarity, increases blood flow to the tissues, and improves myocardial function.”
Although well done, the study doesn’t get at the larger question of whether testosterone increases cardiovascular risk, observed Dr. Bhasin. “None of the randomized studies have been large enough or long enough to determine the effect on cardiovascular events rates. There’s a lot of argument on both sides but we need some data to address that.”
The 6,000-patient TRAVERSE trial is specifically looking at long-term major cardiovascular events with topical testosterone, compared with placebo, in hypogonadal men aged 45-80 years age who have evidence of or are at increased risk for CVD. The study, which is set to be completed in April 2022, should also provide information on fracture risk in these men, said Dr. Bhasin, one of the trial’s principal investigators and lead author of the Endocrine Society’s 2018 clinical practice guideline on testosterone therapy for hypogonadism in men.
William Evans, MD, adjunct professor of human nutrition, University of California, Berkley, said in an interview that the positive effects of testosterone occur at much lower doses in men and women who are hypogonadal but, in this particular population, exercise is the key and the major recommendation.
“Testosterone has been overprescribed and overadvertised for essentially a lifetime of sedentary living, and it’s advertised as a way to get all that back without having to work for it,” he said. “Exercise has a profound and positive effect on control of blood pressure, function, and strength, and testosterone may only affect in people who are sick, people who have really low levels.”
The study was funded by the Heart Foundation of Australia. Lawley Pharmaceuticals provided the study medication and placebo. Dr. Yeap has received speaker honoraria and conference support from Bayer, Eli Lilly, and Besins Healthcare; research support from Bayer, Lily, and Lawley; and served as an adviser for Lily, Besins Healthcare, Ferring, and Lawley. Dr. Shalender reports consultation or advisement for GTx, Pfizer, and TAP; grant or other research support from Solvay and GlaxoSmithKline; and honoraria from Solvay and Auxilium. Dr. Evans reported having no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
Earlier antibiotic initiation for sepsis did not lead to overuse
There has been a marked increase in the time to antibiotic administration for ICU patients with sepsis across Veterans Affairs (VA) hospitals, but there is no evidence that they are being given inappropriately, according to new findings.
Accelerating time-to-antibiotics in sepsis means that patients will be treated earlier, but it could also result in more patients receiving antibiotics, including those without infection. This in turn may contribute to antimicrobial resistance.
“The time to antibiotics for sepsis accelerated across VA hospitals, and declined from 5.8 to 4.8 hours between 2013 and 2018,” said lead study author Sarah Seelye, PhD, data scientist at the U.S. Department of Veterans Affairs, Ann Arbor, Mich. “Despite this, there was no evidence between hospital level antibiotic acceleration in sepsis and antibiotic use among all patients with potential sepsis.”
The results were presented at the Critical Care Congress sponsored by the Society of Critical Care Medicine, which was held virtually this year.
“Many hospitals have initiated programs like this to accelerate the use of antibiotics in patients with severe sepsis, but at the same time, there is growing concern that earlier antibiotic initiation may result in increased antibiotic treatment overall, including those without infection,” said Dr. Seelye. “However, to date, there is little evidence to support this claim.”
The goal of their study was to investigate whether hospital-level acceleration in antibiotic timing for sepsis was associated with increasing antibiotic use among patients hospitalized with potential infection.
They identified 1,101,239 hospitalizations for potential infection in 132 VA hospitals during the period from 2013 to 2018. Of these patients, 608,128 (55.2%) received antibiotics within 48 hours of presentation to the emergency department. A total of 117,435 (10.7%) met the criteria for sepsis.
Hospitals were classified into tertiles of antibiotic acceleration for sepsis: rapid, slow, and flat.
In the VA system, patients with severe sepsis began receiving faster antibiotic treatment in 2017, compared with earlier years. In 2017-2018 more than 20% of sepsis patients had received their first treatment within 2 hours, compared with 14% in 2013-1014.
In 2017-2018, more than 20% of sepsis patients had received their first treatment within 2 hours, compared with 14% in 2013-1014.
Hospitals categorized as rapid accelerators decreased their time to antibiotic initiation from 6.4 hours to 4.5 hours, while slow accelerators went from 5.6 to 4.6 hours from 2013 to 2018, and flat accelerators remained stable during the time period (5.3 hours down to 5.2 hours).
However, statistical analysis showed no real difference between the three groups in antibiotic prescribing.
“Despite this, there was no evidence between hospital-level antibiotic acceleration in sepsis and antibiotic use among all patients with potential sepsis,” said Dr. Seelye.
Weighing in on the study results, Craig M. Coopersmith, MD, professor of surgery at Emory University, Atlanta, noted that these results are very convincing, considering the size of the study and that it encompassed 132 different facilities.
“It’s difficult to say how generalizable these results are but they are definitely generalizable to all hospitals in the VA system,” he said. “In general, there are similarities between large health care systems, and it would be surprising if we found the opposite to be true in non-VA health systems.”
However, he emphasized that there is some possibility that the results would not be identical because different health care systems have different methods of providing care.
“This paper does show that you can get antibiotics into patients faster, which can be life saving, without inappropriately using them on everybody,” Dr. Coopersmith said.
He explained that there is more attention being paid now to antibiotic stewardship, compared with 10 or 15 years ago. “Given the choice of giving someone a single dose of antibiotics who may not need it, as opposed to withholding them from someone who is septic which is life threatening, the risk benefit ratio weighs heavily towards starting them early,” he said. “And then escalate rapidly.”
There has been a marked increase in the time to antibiotic administration for ICU patients with sepsis across Veterans Affairs (VA) hospitals, but there is no evidence that they are being given inappropriately, according to new findings.
Accelerating time-to-antibiotics in sepsis means that patients will be treated earlier, but it could also result in more patients receiving antibiotics, including those without infection. This in turn may contribute to antimicrobial resistance.
“The time to antibiotics for sepsis accelerated across VA hospitals, and declined from 5.8 to 4.8 hours between 2013 and 2018,” said lead study author Sarah Seelye, PhD, data scientist at the U.S. Department of Veterans Affairs, Ann Arbor, Mich. “Despite this, there was no evidence between hospital level antibiotic acceleration in sepsis and antibiotic use among all patients with potential sepsis.”
The results were presented at the Critical Care Congress sponsored by the Society of Critical Care Medicine, which was held virtually this year.
“Many hospitals have initiated programs like this to accelerate the use of antibiotics in patients with severe sepsis, but at the same time, there is growing concern that earlier antibiotic initiation may result in increased antibiotic treatment overall, including those without infection,” said Dr. Seelye. “However, to date, there is little evidence to support this claim.”
The goal of their study was to investigate whether hospital-level acceleration in antibiotic timing for sepsis was associated with increasing antibiotic use among patients hospitalized with potential infection.
They identified 1,101,239 hospitalizations for potential infection in 132 VA hospitals during the period from 2013 to 2018. Of these patients, 608,128 (55.2%) received antibiotics within 48 hours of presentation to the emergency department. A total of 117,435 (10.7%) met the criteria for sepsis.
Hospitals were classified into tertiles of antibiotic acceleration for sepsis: rapid, slow, and flat.
In the VA system, patients with severe sepsis began receiving faster antibiotic treatment in 2017, compared with earlier years. In 2017-2018 more than 20% of sepsis patients had received their first treatment within 2 hours, compared with 14% in 2013-1014.
In 2017-2018, more than 20% of sepsis patients had received their first treatment within 2 hours, compared with 14% in 2013-1014.
Hospitals categorized as rapid accelerators decreased their time to antibiotic initiation from 6.4 hours to 4.5 hours, while slow accelerators went from 5.6 to 4.6 hours from 2013 to 2018, and flat accelerators remained stable during the time period (5.3 hours down to 5.2 hours).
However, statistical analysis showed no real difference between the three groups in antibiotic prescribing.
“Despite this, there was no evidence between hospital-level antibiotic acceleration in sepsis and antibiotic use among all patients with potential sepsis,” said Dr. Seelye.
Weighing in on the study results, Craig M. Coopersmith, MD, professor of surgery at Emory University, Atlanta, noted that these results are very convincing, considering the size of the study and that it encompassed 132 different facilities.
“It’s difficult to say how generalizable these results are but they are definitely generalizable to all hospitals in the VA system,” he said. “In general, there are similarities between large health care systems, and it would be surprising if we found the opposite to be true in non-VA health systems.”
However, he emphasized that there is some possibility that the results would not be identical because different health care systems have different methods of providing care.
“This paper does show that you can get antibiotics into patients faster, which can be life saving, without inappropriately using them on everybody,” Dr. Coopersmith said.
He explained that there is more attention being paid now to antibiotic stewardship, compared with 10 or 15 years ago. “Given the choice of giving someone a single dose of antibiotics who may not need it, as opposed to withholding them from someone who is septic which is life threatening, the risk benefit ratio weighs heavily towards starting them early,” he said. “And then escalate rapidly.”
There has been a marked increase in the time to antibiotic administration for ICU patients with sepsis across Veterans Affairs (VA) hospitals, but there is no evidence that they are being given inappropriately, according to new findings.
Accelerating time-to-antibiotics in sepsis means that patients will be treated earlier, but it could also result in more patients receiving antibiotics, including those without infection. This in turn may contribute to antimicrobial resistance.
“The time to antibiotics for sepsis accelerated across VA hospitals, and declined from 5.8 to 4.8 hours between 2013 and 2018,” said lead study author Sarah Seelye, PhD, data scientist at the U.S. Department of Veterans Affairs, Ann Arbor, Mich. “Despite this, there was no evidence between hospital level antibiotic acceleration in sepsis and antibiotic use among all patients with potential sepsis.”
The results were presented at the Critical Care Congress sponsored by the Society of Critical Care Medicine, which was held virtually this year.
“Many hospitals have initiated programs like this to accelerate the use of antibiotics in patients with severe sepsis, but at the same time, there is growing concern that earlier antibiotic initiation may result in increased antibiotic treatment overall, including those without infection,” said Dr. Seelye. “However, to date, there is little evidence to support this claim.”
The goal of their study was to investigate whether hospital-level acceleration in antibiotic timing for sepsis was associated with increasing antibiotic use among patients hospitalized with potential infection.
They identified 1,101,239 hospitalizations for potential infection in 132 VA hospitals during the period from 2013 to 2018. Of these patients, 608,128 (55.2%) received antibiotics within 48 hours of presentation to the emergency department. A total of 117,435 (10.7%) met the criteria for sepsis.
Hospitals were classified into tertiles of antibiotic acceleration for sepsis: rapid, slow, and flat.
In the VA system, patients with severe sepsis began receiving faster antibiotic treatment in 2017, compared with earlier years. In 2017-2018 more than 20% of sepsis patients had received their first treatment within 2 hours, compared with 14% in 2013-1014.
In 2017-2018, more than 20% of sepsis patients had received their first treatment within 2 hours, compared with 14% in 2013-1014.
Hospitals categorized as rapid accelerators decreased their time to antibiotic initiation from 6.4 hours to 4.5 hours, while slow accelerators went from 5.6 to 4.6 hours from 2013 to 2018, and flat accelerators remained stable during the time period (5.3 hours down to 5.2 hours).
However, statistical analysis showed no real difference between the three groups in antibiotic prescribing.
“Despite this, there was no evidence between hospital-level antibiotic acceleration in sepsis and antibiotic use among all patients with potential sepsis,” said Dr. Seelye.
Weighing in on the study results, Craig M. Coopersmith, MD, professor of surgery at Emory University, Atlanta, noted that these results are very convincing, considering the size of the study and that it encompassed 132 different facilities.
“It’s difficult to say how generalizable these results are but they are definitely generalizable to all hospitals in the VA system,” he said. “In general, there are similarities between large health care systems, and it would be surprising if we found the opposite to be true in non-VA health systems.”
However, he emphasized that there is some possibility that the results would not be identical because different health care systems have different methods of providing care.
“This paper does show that you can get antibiotics into patients faster, which can be life saving, without inappropriately using them on everybody,” Dr. Coopersmith said.
He explained that there is more attention being paid now to antibiotic stewardship, compared with 10 or 15 years ago. “Given the choice of giving someone a single dose of antibiotics who may not need it, as opposed to withholding them from someone who is septic which is life threatening, the risk benefit ratio weighs heavily towards starting them early,” he said. “And then escalate rapidly.”
FROM CCC50
New ASH guidelines: VTE prevention and treatment in cancer patients
New guidelines from the American Society of Hematology “strongly recommend” using no thromboprophylaxis over using parenteral thromboprophylaxis in ambulatory patients receiving cancer chemotherapy who have low venous thromboembolism (VTE) risk, and using no thromboprophylaxis over oral thromboprophylaxis with vitamin K antagonists in those at any VTE risk level.
The evidence-based guidelines for the prevention and treatment of VTE in patient with cancer, published online in Blood Advances, also include a “conditional recommendation” for using either thromboprophylaxis with the direct oral anticoagulants (DOACs) apixaban or rivaroxaban or using no thromboprophylaxis in ambulatory patients with intermediate risk and using the DOACs over no thromboprophylaxis in those with high VTE risk.
The purpose of the guidelines, which also address VTE prophylaxis in hospitalized patients with cancer and the use of anticoagulation for VTE treatment in patients with cancer, is to provide clinical decision support for shared decision-making by patients and clinicians, Gary H. Lyman, MD, of Fred Hutchinson Cancer Research Center, Seattle and Marc Carrier, MD, of the University of Ottawa, and their colleagues from the multidisciplinary guidelines panel explained.
“The recommendations take into consideration the strength of the evidence, risks of mortality, VTE, and bleeding, as well as quality of life, acceptability, and cost considerations,” they wrote, noting that VTE is a common complication in patients with cancer, who are at markedly increased risk for morbidity and mortality from VTE.
Levels of evidence
The panel members relied on updated and original systematic evidence reviews. Conditional recommendations, as opposed to strong recommendations, are defined by the panel as “suggestions,” and all 33 recommendations that make up the guidelines include a statement on the strength of the relevant evidence.
For example, the thromboprophylaxis recommendations for low, intermediate, and high VTE risk are made based on “moderate certainty in the evidence of effects,” and the recommendation for no thromboprophylaxis over oral thromboprophylaxis with vitamin K antagonists is a strong recommendation based on “very low certainty in the evidence of benefits, but high certainty about the harms.”
The guidelines panel also strongly recommends, based on moderate certainty in the evidence of effects, using low-molecular-weight heparin over unfractionated heparin for the initial treatment of VTE in patients with cancer, and suggests, based on “very low certainty in the evidence of effects,” using LMWH over fondaparinux in this setting.
In addition to primary prophylaxis in ambulatory and hospitalized patients and initial VTE treatment, they also address primary prophylaxis for patients with cancer who have a central venous catheter, VTE treatment in surgical patients with cancer, short-term VTE treatment, and long-term VTE treatment.
For example, the guidelines panel conditionally recommends:
- Not using parenteral or oral thromboprophylaxis in patients with cancer and a central venous catheter
- Using LMWH or fondaparinux for surgical patients with cancer
- Using DOACS for the short-term treatment of VTE, and LMWH or DOACs for the long-term treatment of VTE in patients with cancer.
The perils of VTE
VTE in patients with cancer can interfere with treatment, increase mortality risk, and increase costs, the authors noted, adding that VTE can also adversely affect cancer patients’ quality of life.
“Some have even reported the experience of VTE to be more upsetting than that of the cancer,” they wrote. “More than 50% of thrombotic events occur within 3 months of the cancer diagnosis, a time when most cancer treatments will be underway. Patients, who are still coming to terms with a recent cancer diagnosis, often view the occurrence of VTE as a further threat to life, confirmation of the severity of their condition, and a poor prognostic sign.”
Therefore, the new guidelines aim to reduce VTE frequency, risk of bleeding complications, morbidity, and costs, thereby improving quality of life and the patient experience, the authors said, noting that three other recent guidelines on VTEs in patients with cancer have been published: the 2019 American Society of Clinical Oncology guidelines, the 2019 International Initiative on Thrombosis and Cancer guidelines, and the 2020 National Comprehensive Cancer Network guidelines.
The ASH guidelines are similar in many ways to the other guidelines, but differ in some ways, as well. An example is the timing of initiation of pharmacological thromboprophylaxis in patients undergoing cancer-related major abdominal surgery. The ASCO and ITAC guidelines advise starting thromboprophylaxis preoperatively, whereas the ASH guidelines recommend initiating thromboprophylaxis postoperatively, citing “the limited advantages to initiating thromboprophylaxis preoperatively, in addition to the potential bleeding and logistical considerations associated with neuraxial anesthesia.”
These differences highlight a lack of data in that setting and the need for additional studies, the authors said.
ASH vs. ASCO
James Douketis, MD, a practicing clinician and professor of medicine at McMaster University, Hamilton, Ont., highlighted another difference between the ASH and ASCO guidelines.
“For the treatment of [cancer-associated thrombosis], ASCO gives a strong recommendation to use LMWH or DOACs (with some caveats), which is easy to follow. ASH, on the other hand, suggests LMWH or a DOAC for the first 7-10 days, DOACs for the first 3-6 months, and back to LMWH or DOACs after 6 months,” he said in an interview.
The recommendation is “very evidence based but ambiguous and not helpful for the practicing clinician,” added Dr. Douketis, who helped develop the ITAC guidelines, but was not part of the ASH or ASCO guideline panels.
ASCO also provides a clear recommendation for giving VTE prophylaxis for 4 weeks after cancer surgery in patients with high VTE risk, whereas ASH gives “a somewhat vague recommendation” for thromboprophylaxis after hospital discharge.
The guidelines are “pretty well aligned” with respect to recommendations on VTE prophylaxis in medical cancer patients receiving chemotherapy, and although the “extremely academic” ASH guidelines were developed by “a superb team using the same evidence and excellent methodology,” they are interpreted in slightly different ways and fall short when it comes to being clinician friendly, Dr. Douketis said.
“At the end of day, for practicing clinicians, the ASH guidelines don’t provide a message that’s easy to digest,” he added.
ASH has, however, provided a resource page that includes tools and information for implementing the guidelines in clinical practice, and will maintain the guidelines “through surveillance for new evidence, ongoing review by experts, and regular revisions,” the authors said.
New guidelines from the American Society of Hematology “strongly recommend” using no thromboprophylaxis over using parenteral thromboprophylaxis in ambulatory patients receiving cancer chemotherapy who have low venous thromboembolism (VTE) risk, and using no thromboprophylaxis over oral thromboprophylaxis with vitamin K antagonists in those at any VTE risk level.
The evidence-based guidelines for the prevention and treatment of VTE in patient with cancer, published online in Blood Advances, also include a “conditional recommendation” for using either thromboprophylaxis with the direct oral anticoagulants (DOACs) apixaban or rivaroxaban or using no thromboprophylaxis in ambulatory patients with intermediate risk and using the DOACs over no thromboprophylaxis in those with high VTE risk.
The purpose of the guidelines, which also address VTE prophylaxis in hospitalized patients with cancer and the use of anticoagulation for VTE treatment in patients with cancer, is to provide clinical decision support for shared decision-making by patients and clinicians, Gary H. Lyman, MD, of Fred Hutchinson Cancer Research Center, Seattle and Marc Carrier, MD, of the University of Ottawa, and their colleagues from the multidisciplinary guidelines panel explained.
“The recommendations take into consideration the strength of the evidence, risks of mortality, VTE, and bleeding, as well as quality of life, acceptability, and cost considerations,” they wrote, noting that VTE is a common complication in patients with cancer, who are at markedly increased risk for morbidity and mortality from VTE.
Levels of evidence
The panel members relied on updated and original systematic evidence reviews. Conditional recommendations, as opposed to strong recommendations, are defined by the panel as “suggestions,” and all 33 recommendations that make up the guidelines include a statement on the strength of the relevant evidence.
For example, the thromboprophylaxis recommendations for low, intermediate, and high VTE risk are made based on “moderate certainty in the evidence of effects,” and the recommendation for no thromboprophylaxis over oral thromboprophylaxis with vitamin K antagonists is a strong recommendation based on “very low certainty in the evidence of benefits, but high certainty about the harms.”
The guidelines panel also strongly recommends, based on moderate certainty in the evidence of effects, using low-molecular-weight heparin over unfractionated heparin for the initial treatment of VTE in patients with cancer, and suggests, based on “very low certainty in the evidence of effects,” using LMWH over fondaparinux in this setting.
In addition to primary prophylaxis in ambulatory and hospitalized patients and initial VTE treatment, they also address primary prophylaxis for patients with cancer who have a central venous catheter, VTE treatment in surgical patients with cancer, short-term VTE treatment, and long-term VTE treatment.
For example, the guidelines panel conditionally recommends:
- Not using parenteral or oral thromboprophylaxis in patients with cancer and a central venous catheter
- Using LMWH or fondaparinux for surgical patients with cancer
- Using DOACS for the short-term treatment of VTE, and LMWH or DOACs for the long-term treatment of VTE in patients with cancer.
The perils of VTE
VTE in patients with cancer can interfere with treatment, increase mortality risk, and increase costs, the authors noted, adding that VTE can also adversely affect cancer patients’ quality of life.
“Some have even reported the experience of VTE to be more upsetting than that of the cancer,” they wrote. “More than 50% of thrombotic events occur within 3 months of the cancer diagnosis, a time when most cancer treatments will be underway. Patients, who are still coming to terms with a recent cancer diagnosis, often view the occurrence of VTE as a further threat to life, confirmation of the severity of their condition, and a poor prognostic sign.”
Therefore, the new guidelines aim to reduce VTE frequency, risk of bleeding complications, morbidity, and costs, thereby improving quality of life and the patient experience, the authors said, noting that three other recent guidelines on VTEs in patients with cancer have been published: the 2019 American Society of Clinical Oncology guidelines, the 2019 International Initiative on Thrombosis and Cancer guidelines, and the 2020 National Comprehensive Cancer Network guidelines.
The ASH guidelines are similar in many ways to the other guidelines, but differ in some ways, as well. An example is the timing of initiation of pharmacological thromboprophylaxis in patients undergoing cancer-related major abdominal surgery. The ASCO and ITAC guidelines advise starting thromboprophylaxis preoperatively, whereas the ASH guidelines recommend initiating thromboprophylaxis postoperatively, citing “the limited advantages to initiating thromboprophylaxis preoperatively, in addition to the potential bleeding and logistical considerations associated with neuraxial anesthesia.”
These differences highlight a lack of data in that setting and the need for additional studies, the authors said.
ASH vs. ASCO
James Douketis, MD, a practicing clinician and professor of medicine at McMaster University, Hamilton, Ont., highlighted another difference between the ASH and ASCO guidelines.
“For the treatment of [cancer-associated thrombosis], ASCO gives a strong recommendation to use LMWH or DOACs (with some caveats), which is easy to follow. ASH, on the other hand, suggests LMWH or a DOAC for the first 7-10 days, DOACs for the first 3-6 months, and back to LMWH or DOACs after 6 months,” he said in an interview.
The recommendation is “very evidence based but ambiguous and not helpful for the practicing clinician,” added Dr. Douketis, who helped develop the ITAC guidelines, but was not part of the ASH or ASCO guideline panels.
ASCO also provides a clear recommendation for giving VTE prophylaxis for 4 weeks after cancer surgery in patients with high VTE risk, whereas ASH gives “a somewhat vague recommendation” for thromboprophylaxis after hospital discharge.
The guidelines are “pretty well aligned” with respect to recommendations on VTE prophylaxis in medical cancer patients receiving chemotherapy, and although the “extremely academic” ASH guidelines were developed by “a superb team using the same evidence and excellent methodology,” they are interpreted in slightly different ways and fall short when it comes to being clinician friendly, Dr. Douketis said.
“At the end of day, for practicing clinicians, the ASH guidelines don’t provide a message that’s easy to digest,” he added.
ASH has, however, provided a resource page that includes tools and information for implementing the guidelines in clinical practice, and will maintain the guidelines “through surveillance for new evidence, ongoing review by experts, and regular revisions,” the authors said.
New guidelines from the American Society of Hematology “strongly recommend” using no thromboprophylaxis over using parenteral thromboprophylaxis in ambulatory patients receiving cancer chemotherapy who have low venous thromboembolism (VTE) risk, and using no thromboprophylaxis over oral thromboprophylaxis with vitamin K antagonists in those at any VTE risk level.
The evidence-based guidelines for the prevention and treatment of VTE in patient with cancer, published online in Blood Advances, also include a “conditional recommendation” for using either thromboprophylaxis with the direct oral anticoagulants (DOACs) apixaban or rivaroxaban or using no thromboprophylaxis in ambulatory patients with intermediate risk and using the DOACs over no thromboprophylaxis in those with high VTE risk.
The purpose of the guidelines, which also address VTE prophylaxis in hospitalized patients with cancer and the use of anticoagulation for VTE treatment in patients with cancer, is to provide clinical decision support for shared decision-making by patients and clinicians, Gary H. Lyman, MD, of Fred Hutchinson Cancer Research Center, Seattle and Marc Carrier, MD, of the University of Ottawa, and their colleagues from the multidisciplinary guidelines panel explained.
“The recommendations take into consideration the strength of the evidence, risks of mortality, VTE, and bleeding, as well as quality of life, acceptability, and cost considerations,” they wrote, noting that VTE is a common complication in patients with cancer, who are at markedly increased risk for morbidity and mortality from VTE.
Levels of evidence
The panel members relied on updated and original systematic evidence reviews. Conditional recommendations, as opposed to strong recommendations, are defined by the panel as “suggestions,” and all 33 recommendations that make up the guidelines include a statement on the strength of the relevant evidence.
For example, the thromboprophylaxis recommendations for low, intermediate, and high VTE risk are made based on “moderate certainty in the evidence of effects,” and the recommendation for no thromboprophylaxis over oral thromboprophylaxis with vitamin K antagonists is a strong recommendation based on “very low certainty in the evidence of benefits, but high certainty about the harms.”
The guidelines panel also strongly recommends, based on moderate certainty in the evidence of effects, using low-molecular-weight heparin over unfractionated heparin for the initial treatment of VTE in patients with cancer, and suggests, based on “very low certainty in the evidence of effects,” using LMWH over fondaparinux in this setting.
In addition to primary prophylaxis in ambulatory and hospitalized patients and initial VTE treatment, they also address primary prophylaxis for patients with cancer who have a central venous catheter, VTE treatment in surgical patients with cancer, short-term VTE treatment, and long-term VTE treatment.
For example, the guidelines panel conditionally recommends:
- Not using parenteral or oral thromboprophylaxis in patients with cancer and a central venous catheter
- Using LMWH or fondaparinux for surgical patients with cancer
- Using DOACS for the short-term treatment of VTE, and LMWH or DOACs for the long-term treatment of VTE in patients with cancer.
The perils of VTE
VTE in patients with cancer can interfere with treatment, increase mortality risk, and increase costs, the authors noted, adding that VTE can also adversely affect cancer patients’ quality of life.
“Some have even reported the experience of VTE to be more upsetting than that of the cancer,” they wrote. “More than 50% of thrombotic events occur within 3 months of the cancer diagnosis, a time when most cancer treatments will be underway. Patients, who are still coming to terms with a recent cancer diagnosis, often view the occurrence of VTE as a further threat to life, confirmation of the severity of their condition, and a poor prognostic sign.”
Therefore, the new guidelines aim to reduce VTE frequency, risk of bleeding complications, morbidity, and costs, thereby improving quality of life and the patient experience, the authors said, noting that three other recent guidelines on VTEs in patients with cancer have been published: the 2019 American Society of Clinical Oncology guidelines, the 2019 International Initiative on Thrombosis and Cancer guidelines, and the 2020 National Comprehensive Cancer Network guidelines.
The ASH guidelines are similar in many ways to the other guidelines, but differ in some ways, as well. An example is the timing of initiation of pharmacological thromboprophylaxis in patients undergoing cancer-related major abdominal surgery. The ASCO and ITAC guidelines advise starting thromboprophylaxis preoperatively, whereas the ASH guidelines recommend initiating thromboprophylaxis postoperatively, citing “the limited advantages to initiating thromboprophylaxis preoperatively, in addition to the potential bleeding and logistical considerations associated with neuraxial anesthesia.”
These differences highlight a lack of data in that setting and the need for additional studies, the authors said.
ASH vs. ASCO
James Douketis, MD, a practicing clinician and professor of medicine at McMaster University, Hamilton, Ont., highlighted another difference between the ASH and ASCO guidelines.
“For the treatment of [cancer-associated thrombosis], ASCO gives a strong recommendation to use LMWH or DOACs (with some caveats), which is easy to follow. ASH, on the other hand, suggests LMWH or a DOAC for the first 7-10 days, DOACs for the first 3-6 months, and back to LMWH or DOACs after 6 months,” he said in an interview.
The recommendation is “very evidence based but ambiguous and not helpful for the practicing clinician,” added Dr. Douketis, who helped develop the ITAC guidelines, but was not part of the ASH or ASCO guideline panels.
ASCO also provides a clear recommendation for giving VTE prophylaxis for 4 weeks after cancer surgery in patients with high VTE risk, whereas ASH gives “a somewhat vague recommendation” for thromboprophylaxis after hospital discharge.
The guidelines are “pretty well aligned” with respect to recommendations on VTE prophylaxis in medical cancer patients receiving chemotherapy, and although the “extremely academic” ASH guidelines were developed by “a superb team using the same evidence and excellent methodology,” they are interpreted in slightly different ways and fall short when it comes to being clinician friendly, Dr. Douketis said.
“At the end of day, for practicing clinicians, the ASH guidelines don’t provide a message that’s easy to digest,” he added.
ASH has, however, provided a resource page that includes tools and information for implementing the guidelines in clinical practice, and will maintain the guidelines “through surveillance for new evidence, ongoing review by experts, and regular revisions,” the authors said.
FROM BLOOD ADVANCES
Mepolizumab reduced exacerbations in patients with asthma and atopy, depression comorbidities
, according to research from the annual meeting of the American Academy of Allergy, Asthma, and Immunology.
“Mepolizumab has clearly been shown to improve severe asthma control in many clinical trials, but atopy, obesity, and depression/anxiety affect patients with asthma at an increased rate,” Thomas B. Casale, MD, former AAAAI president and professor of medicine and pediatrics at the University of South Florida in Tampa, said in a presentation at the meeting. “Yet, few studies have examined whether asthma therapy with these comorbidities works.”
Dr. Casale and colleagues performed a retrospective analysis of patients in the United States from the MarketScan Commercial and Medicare Supplemental Database between November 2014 and December 2018 who had atopy, obesity, or depression/anxiety in addition to asthma and were receiving mepolizumab. Atopy in the study was defined as allergic rhinitis, anaphylaxis, atopic dermatitis, conjunctivitis, eosinophilic esophagitis, and food allergies. Patients were at least age 12 years, had at least one diagnosis for asthma, at least one diagnosis code for atopic disease, obesity, or depression/anxiety at baseline, and at least two administrations of mepolizumab within 180 days.
The researchers examined the number of exacerbations, oral corticosteroid (OCS) claims, and OCS bursts per year at 12-month follow-up, compared with baseline. They identified exacerbations by examining patients who had an emergency department or outpatient claim related to their asthma, and a claim for systemic corticosteroids made in the 4 days prior to or 5 days after a visit, or if their inpatient hospital admission contained a primary asthma diagnosis. Dr. Casale and colleagues measured OCS bursts as a pharmacy claim of at least 20 mg of prednisone per day for between 3 and 28 days plus a claim for an emergency department visit related to asthma in the 7 days prior or 6 days after the claim.
At baseline, patients across all groups were mean age 50.5-52.4 years with a Charleson Comorbidity Index score between 1.1 and 1.4, a majority were women (59.0%-72.0%) and nearly all were commercially insured (88.0%-90.0%). Patients who used biologics at baseline and/or used a biologic that wasn’t mepolizumab during the follow-up period were excluded.
Medication claims in the groups included inhaled corticosteroids (ICS) (36.8%-48.6%), ICS/long-acting beta-agonist (LABA) (60.2%-63.0%), LABA/ long-acting muscarinic antagonist (LAMA) (1.2%-3.5%), ICS/LABA/LAMA (21.2%-25.1%), short-acting beta-agonist (SABA) (83.2%-87.7%), LAMA alone (33.5%-42.1%), or leukotriene receptor antagonist (LTRA).
In the non–mutually exclusive group of patients with atopy (468 patients), 28.0% had comorbid obesity and 26.0% had comorbid depression/anxiety. For patients with obesity categorized in a non–mutually exclusive subgroup (171 patients), 79.0% had comorbid atopy and 32.0% had comorbid depression/anxiety. Among patients with non–mutually exclusive depression/anxiety (173 patients), 70.0% had comorbid atopy, while 32.0% had comorbid obesity.
The results showed the mean number of overall exacerbations decreased by 48% at 12 months in the atopic group (2.3 vs. 1.2; P < .001), 52% in the group with obesity (2.5 vs. 1.2; P < .001), and 38% in the depression/anxiety group (2.4 vs. 1.5; P < .001). The mean number of exacerbations leading to hospitalizations decreased by 64% in the atopic group (0.11 vs. 0.04; P < .001), 65% in the group with obesity (0.20 vs. 0.07; P < .001), and 68% in the group with depression/anxiety (0.22 vs. 0.07; P < .001).
The researchers also found the mean number of OCS claims and OCS bursts also significantly decreased over the 12-month follow-up period. Mean OCS claims decreased by 33% for patients in the atopic group (5.5 vs. 3.7; P < .001), by 38% in the group with obesity (6.1 vs. 3.8; P < .001), and by 31% in the group with depression/anxiety (6.2 vs. 4.3; P < .001).
The mean number of OCS bursts also significantly decreased by 40% in the atopic group (2.0 vs. 2.1; P < .001), 48% in the group with obesity (2.3 vs. 1.2; P < .001), and by 37% in the group with depression/anxiety (1.9 vs. 1.2; P < .001). In total, 69% of patients with comorbid atopy, 70.8% of patients with comorbid obesity, and 68.2% of patients with comorbid depression/anxiety experienced a mean decrease in their OCS dose over 12 months.
“These data demonstrate that patients with asthma and atopy, obesity, or depression and anxiety have significantly fewer exacerbations and reduced OCS use in a real-world setting with treatment of mepolizumab,” Dr. Casale said. “Thus, holistic patient care for severe asthma is critical, and mepolizumab provides tangible clinical benefit despite the complexities of medical comorbidities.”
This study was funded by GlaxoSmithKline, and the company also funded graphic design support of the poster. Dr. Casale reports he has received research funds from GlaxoSmithKline. Four authors report being current or former GlaxoSmithKline employees; three authors report holding stock and/or shares of GlaxoSmithKline. Three authors are IBM Watson Health employees, a company GlaxoSmithKline has provided research funding.
, according to research from the annual meeting of the American Academy of Allergy, Asthma, and Immunology.
“Mepolizumab has clearly been shown to improve severe asthma control in many clinical trials, but atopy, obesity, and depression/anxiety affect patients with asthma at an increased rate,” Thomas B. Casale, MD, former AAAAI president and professor of medicine and pediatrics at the University of South Florida in Tampa, said in a presentation at the meeting. “Yet, few studies have examined whether asthma therapy with these comorbidities works.”
Dr. Casale and colleagues performed a retrospective analysis of patients in the United States from the MarketScan Commercial and Medicare Supplemental Database between November 2014 and December 2018 who had atopy, obesity, or depression/anxiety in addition to asthma and were receiving mepolizumab. Atopy in the study was defined as allergic rhinitis, anaphylaxis, atopic dermatitis, conjunctivitis, eosinophilic esophagitis, and food allergies. Patients were at least age 12 years, had at least one diagnosis for asthma, at least one diagnosis code for atopic disease, obesity, or depression/anxiety at baseline, and at least two administrations of mepolizumab within 180 days.
The researchers examined the number of exacerbations, oral corticosteroid (OCS) claims, and OCS bursts per year at 12-month follow-up, compared with baseline. They identified exacerbations by examining patients who had an emergency department or outpatient claim related to their asthma, and a claim for systemic corticosteroids made in the 4 days prior to or 5 days after a visit, or if their inpatient hospital admission contained a primary asthma diagnosis. Dr. Casale and colleagues measured OCS bursts as a pharmacy claim of at least 20 mg of prednisone per day for between 3 and 28 days plus a claim for an emergency department visit related to asthma in the 7 days prior or 6 days after the claim.
At baseline, patients across all groups were mean age 50.5-52.4 years with a Charleson Comorbidity Index score between 1.1 and 1.4, a majority were women (59.0%-72.0%) and nearly all were commercially insured (88.0%-90.0%). Patients who used biologics at baseline and/or used a biologic that wasn’t mepolizumab during the follow-up period were excluded.
Medication claims in the groups included inhaled corticosteroids (ICS) (36.8%-48.6%), ICS/long-acting beta-agonist (LABA) (60.2%-63.0%), LABA/ long-acting muscarinic antagonist (LAMA) (1.2%-3.5%), ICS/LABA/LAMA (21.2%-25.1%), short-acting beta-agonist (SABA) (83.2%-87.7%), LAMA alone (33.5%-42.1%), or leukotriene receptor antagonist (LTRA).
In the non–mutually exclusive group of patients with atopy (468 patients), 28.0% had comorbid obesity and 26.0% had comorbid depression/anxiety. For patients with obesity categorized in a non–mutually exclusive subgroup (171 patients), 79.0% had comorbid atopy and 32.0% had comorbid depression/anxiety. Among patients with non–mutually exclusive depression/anxiety (173 patients), 70.0% had comorbid atopy, while 32.0% had comorbid obesity.
The results showed the mean number of overall exacerbations decreased by 48% at 12 months in the atopic group (2.3 vs. 1.2; P < .001), 52% in the group with obesity (2.5 vs. 1.2; P < .001), and 38% in the depression/anxiety group (2.4 vs. 1.5; P < .001). The mean number of exacerbations leading to hospitalizations decreased by 64% in the atopic group (0.11 vs. 0.04; P < .001), 65% in the group with obesity (0.20 vs. 0.07; P < .001), and 68% in the group with depression/anxiety (0.22 vs. 0.07; P < .001).
The researchers also found the mean number of OCS claims and OCS bursts also significantly decreased over the 12-month follow-up period. Mean OCS claims decreased by 33% for patients in the atopic group (5.5 vs. 3.7; P < .001), by 38% in the group with obesity (6.1 vs. 3.8; P < .001), and by 31% in the group with depression/anxiety (6.2 vs. 4.3; P < .001).
The mean number of OCS bursts also significantly decreased by 40% in the atopic group (2.0 vs. 2.1; P < .001), 48% in the group with obesity (2.3 vs. 1.2; P < .001), and by 37% in the group with depression/anxiety (1.9 vs. 1.2; P < .001). In total, 69% of patients with comorbid atopy, 70.8% of patients with comorbid obesity, and 68.2% of patients with comorbid depression/anxiety experienced a mean decrease in their OCS dose over 12 months.
“These data demonstrate that patients with asthma and atopy, obesity, or depression and anxiety have significantly fewer exacerbations and reduced OCS use in a real-world setting with treatment of mepolizumab,” Dr. Casale said. “Thus, holistic patient care for severe asthma is critical, and mepolizumab provides tangible clinical benefit despite the complexities of medical comorbidities.”
This study was funded by GlaxoSmithKline, and the company also funded graphic design support of the poster. Dr. Casale reports he has received research funds from GlaxoSmithKline. Four authors report being current or former GlaxoSmithKline employees; three authors report holding stock and/or shares of GlaxoSmithKline. Three authors are IBM Watson Health employees, a company GlaxoSmithKline has provided research funding.
, according to research from the annual meeting of the American Academy of Allergy, Asthma, and Immunology.
“Mepolizumab has clearly been shown to improve severe asthma control in many clinical trials, but atopy, obesity, and depression/anxiety affect patients with asthma at an increased rate,” Thomas B. Casale, MD, former AAAAI president and professor of medicine and pediatrics at the University of South Florida in Tampa, said in a presentation at the meeting. “Yet, few studies have examined whether asthma therapy with these comorbidities works.”
Dr. Casale and colleagues performed a retrospective analysis of patients in the United States from the MarketScan Commercial and Medicare Supplemental Database between November 2014 and December 2018 who had atopy, obesity, or depression/anxiety in addition to asthma and were receiving mepolizumab. Atopy in the study was defined as allergic rhinitis, anaphylaxis, atopic dermatitis, conjunctivitis, eosinophilic esophagitis, and food allergies. Patients were at least age 12 years, had at least one diagnosis for asthma, at least one diagnosis code for atopic disease, obesity, or depression/anxiety at baseline, and at least two administrations of mepolizumab within 180 days.
The researchers examined the number of exacerbations, oral corticosteroid (OCS) claims, and OCS bursts per year at 12-month follow-up, compared with baseline. They identified exacerbations by examining patients who had an emergency department or outpatient claim related to their asthma, and a claim for systemic corticosteroids made in the 4 days prior to or 5 days after a visit, or if their inpatient hospital admission contained a primary asthma diagnosis. Dr. Casale and colleagues measured OCS bursts as a pharmacy claim of at least 20 mg of prednisone per day for between 3 and 28 days plus a claim for an emergency department visit related to asthma in the 7 days prior or 6 days after the claim.
At baseline, patients across all groups were mean age 50.5-52.4 years with a Charleson Comorbidity Index score between 1.1 and 1.4, a majority were women (59.0%-72.0%) and nearly all were commercially insured (88.0%-90.0%). Patients who used biologics at baseline and/or used a biologic that wasn’t mepolizumab during the follow-up period were excluded.
Medication claims in the groups included inhaled corticosteroids (ICS) (36.8%-48.6%), ICS/long-acting beta-agonist (LABA) (60.2%-63.0%), LABA/ long-acting muscarinic antagonist (LAMA) (1.2%-3.5%), ICS/LABA/LAMA (21.2%-25.1%), short-acting beta-agonist (SABA) (83.2%-87.7%), LAMA alone (33.5%-42.1%), or leukotriene receptor antagonist (LTRA).
In the non–mutually exclusive group of patients with atopy (468 patients), 28.0% had comorbid obesity and 26.0% had comorbid depression/anxiety. For patients with obesity categorized in a non–mutually exclusive subgroup (171 patients), 79.0% had comorbid atopy and 32.0% had comorbid depression/anxiety. Among patients with non–mutually exclusive depression/anxiety (173 patients), 70.0% had comorbid atopy, while 32.0% had comorbid obesity.
The results showed the mean number of overall exacerbations decreased by 48% at 12 months in the atopic group (2.3 vs. 1.2; P < .001), 52% in the group with obesity (2.5 vs. 1.2; P < .001), and 38% in the depression/anxiety group (2.4 vs. 1.5; P < .001). The mean number of exacerbations leading to hospitalizations decreased by 64% in the atopic group (0.11 vs. 0.04; P < .001), 65% in the group with obesity (0.20 vs. 0.07; P < .001), and 68% in the group with depression/anxiety (0.22 vs. 0.07; P < .001).
The researchers also found the mean number of OCS claims and OCS bursts also significantly decreased over the 12-month follow-up period. Mean OCS claims decreased by 33% for patients in the atopic group (5.5 vs. 3.7; P < .001), by 38% in the group with obesity (6.1 vs. 3.8; P < .001), and by 31% in the group with depression/anxiety (6.2 vs. 4.3; P < .001).
The mean number of OCS bursts also significantly decreased by 40% in the atopic group (2.0 vs. 2.1; P < .001), 48% in the group with obesity (2.3 vs. 1.2; P < .001), and by 37% in the group with depression/anxiety (1.9 vs. 1.2; P < .001). In total, 69% of patients with comorbid atopy, 70.8% of patients with comorbid obesity, and 68.2% of patients with comorbid depression/anxiety experienced a mean decrease in their OCS dose over 12 months.
“These data demonstrate that patients with asthma and atopy, obesity, or depression and anxiety have significantly fewer exacerbations and reduced OCS use in a real-world setting with treatment of mepolizumab,” Dr. Casale said. “Thus, holistic patient care for severe asthma is critical, and mepolizumab provides tangible clinical benefit despite the complexities of medical comorbidities.”
This study was funded by GlaxoSmithKline, and the company also funded graphic design support of the poster. Dr. Casale reports he has received research funds from GlaxoSmithKline. Four authors report being current or former GlaxoSmithKline employees; three authors report holding stock and/or shares of GlaxoSmithKline. Three authors are IBM Watson Health employees, a company GlaxoSmithKline has provided research funding.
FROM AAAAI 2021
Bladder cancer indication withdrawn for durvalumab
The change does not affect this indication outside the United States, nor does it affect other approved durvalumab indications within the United States.
For example, durvalumab remains approved by the Food and Drug Administration in the curative-intent setting of unresectable, stage III non–small cell lung cancer after chemoradiotherapy and for the treatment of extensive-stage small cell lung cancer.
AstraZeneca is continuing with clinical trials of durvalumab in various combinations for the treatment of bladder cancer.
Granted accelerated approval
Durvalumab was granted accelerated approval in May 2017 by the FDA specifically for the treatment of patients with locally advanced or metastatic urothelial carcinoma who experience disease progression during or following platinum-containing chemotherapy or who experience disease progression within 12 months of neoadjuvant or adjuvant treatment with that chemotherapy.
That accelerated approval was based on the surrogate markers of tumor response rate and duration of response from Study 1108, a phase 1/2 trial. In this trial, the overall response rate was 17.8% in a cohort of 191 patients with locally advanced or metastatic urothelial cancer that had progressed during or after a platinum-based regimen.
However, in the confirmatory phase 3 DANUBE trial in patients with unresectable metastatic bladder cancer, neither durvalumab nor durvalumab plus tremelimumab met the primary endpoint of improving overall survival in comparison with standard-of-care chemotherapy.
“While the withdrawal in previously treated metastatic bladder cancer is disappointing, we respect the principles FDA set out when the accelerated approval pathway was founded,” Dave Fredrickson, executive vice president, Oncology Business Unit, AstraZeneca, said in a company press statement.
A version of this article first appeared on Medscape.com.
The change does not affect this indication outside the United States, nor does it affect other approved durvalumab indications within the United States.
For example, durvalumab remains approved by the Food and Drug Administration in the curative-intent setting of unresectable, stage III non–small cell lung cancer after chemoradiotherapy and for the treatment of extensive-stage small cell lung cancer.
AstraZeneca is continuing with clinical trials of durvalumab in various combinations for the treatment of bladder cancer.
Granted accelerated approval
Durvalumab was granted accelerated approval in May 2017 by the FDA specifically for the treatment of patients with locally advanced or metastatic urothelial carcinoma who experience disease progression during or following platinum-containing chemotherapy or who experience disease progression within 12 months of neoadjuvant or adjuvant treatment with that chemotherapy.
That accelerated approval was based on the surrogate markers of tumor response rate and duration of response from Study 1108, a phase 1/2 trial. In this trial, the overall response rate was 17.8% in a cohort of 191 patients with locally advanced or metastatic urothelial cancer that had progressed during or after a platinum-based regimen.
However, in the confirmatory phase 3 DANUBE trial in patients with unresectable metastatic bladder cancer, neither durvalumab nor durvalumab plus tremelimumab met the primary endpoint of improving overall survival in comparison with standard-of-care chemotherapy.
“While the withdrawal in previously treated metastatic bladder cancer is disappointing, we respect the principles FDA set out when the accelerated approval pathway was founded,” Dave Fredrickson, executive vice president, Oncology Business Unit, AstraZeneca, said in a company press statement.
A version of this article first appeared on Medscape.com.
The change does not affect this indication outside the United States, nor does it affect other approved durvalumab indications within the United States.
For example, durvalumab remains approved by the Food and Drug Administration in the curative-intent setting of unresectable, stage III non–small cell lung cancer after chemoradiotherapy and for the treatment of extensive-stage small cell lung cancer.
AstraZeneca is continuing with clinical trials of durvalumab in various combinations for the treatment of bladder cancer.
Granted accelerated approval
Durvalumab was granted accelerated approval in May 2017 by the FDA specifically for the treatment of patients with locally advanced or metastatic urothelial carcinoma who experience disease progression during or following platinum-containing chemotherapy or who experience disease progression within 12 months of neoadjuvant or adjuvant treatment with that chemotherapy.
That accelerated approval was based on the surrogate markers of tumor response rate and duration of response from Study 1108, a phase 1/2 trial. In this trial, the overall response rate was 17.8% in a cohort of 191 patients with locally advanced or metastatic urothelial cancer that had progressed during or after a platinum-based regimen.
However, in the confirmatory phase 3 DANUBE trial in patients with unresectable metastatic bladder cancer, neither durvalumab nor durvalumab plus tremelimumab met the primary endpoint of improving overall survival in comparison with standard-of-care chemotherapy.
“While the withdrawal in previously treated metastatic bladder cancer is disappointing, we respect the principles FDA set out when the accelerated approval pathway was founded,” Dave Fredrickson, executive vice president, Oncology Business Unit, AstraZeneca, said in a company press statement.
A version of this article first appeared on Medscape.com.
FDA approves cemiplimab-rwlc for NSCLC with PD-L1 expression
Specifically, the indication is for first-line treatment as monotherapy for patients with locally advanced or metastatic disease who are not candidates for surgical resection or definitive chemoradiotherapy and whose tumors have a high expression of programmed death–ligand 1 (PD-L1) (Tumor Proportion Score >50%), as determined by an FDA-approved test, with no EGFR, ALK, or ROS1 aberrations.
This is the third indication for cemiplimab-rlwc, a monoclonal antibody and PD-1 inhibitor.
In February, it was approved as the first immunotherapy to treat patients with locally advanced or metastatic basal cell carcinoma that was previously treated with a hedgehog pathway inhibitor or for whom a hedgehog inhibitor is inappropriate.
Cemiplimab-rlwc previously received FDA approval in 2018 for locally advanced or metastatic cutaneous squamous cell carcinoma for patients who were not eligible for curative surgery or radiotherapy. At the time, Karl Lewis, MD, a professor at the University of Colorado at Denver, Aurora, and a trial investigator, predicted that the drug “will change the treatment paradigm for patients with advanced basal cell carcinoma.”
Outperforms chemotherapy
The approval for use in NSCLC is based on results from the phase 3, open-label EMPOWER-Lung 1 trial, which randomly assigned 710 patients in a 1:1 ratio to receive either cemiplimab-rwlc or platinum-doublet chemotherapy. Patients had either locally advanced NSCLC and were not candidates for surgical resection or definitive chemoradiotherapy, or they had metastatic NSCLC.
Patients in the experimental arm received cemiplimab-rwlc 350 mg intravenously every 3 weeks. The primary efficacy outcome measures were overall survival (OS) and progression-free survival (PFS), determined on the basis of blinded independent central review.
Results showed statistically significant improvements in both outcomes. Median OS was 22.1 months with cemiplimab-rwlc versus 14.3 months with chemotherapy (hazard ratio, 0.68; P = .0022). Median PFS was 6.2 months versus 5.6 months (HR, 0.59; P < .0001).
The confirmed overall response rate was 37% for the cemiplimab arm versus 21% for the chemotherapy arm.
The most common adverse reactions (>10%) with cemiplimab-rlwc were musculoskeletal pain, rash, anemia, fatigue, decreased appetite, pneumonia, and cough.
This approval “means physicians and patients have a potent new treatment option against this deadly disease,” said Naiyer Rizvi, MD, Price Family Professor of Medicine, director of thoracic oncology, and codirector of cancer immunotherapy at Columbia University Irving Medical Center, New York, in a statement. He was a steering committee member on the EMPOWER-Lung-1 Trial.
“Notably, Libtayo was approved based on a pivotal trial where most chemotherapy patients crossed over to Libtayo following disease progression, and that allowed for frequently underrepresented patients who had pretreated and clinically stable brain metastases or who had locally advanced disease and were not candidates for definitive chemoradiation,” said Dr. Rizvi. “This gives doctors important new data when considering Libtayo for the varied patients and situations they treat in daily clinical practice.”
A version of this article first appeared on Medscape.com.
Specifically, the indication is for first-line treatment as monotherapy for patients with locally advanced or metastatic disease who are not candidates for surgical resection or definitive chemoradiotherapy and whose tumors have a high expression of programmed death–ligand 1 (PD-L1) (Tumor Proportion Score >50%), as determined by an FDA-approved test, with no EGFR, ALK, or ROS1 aberrations.
This is the third indication for cemiplimab-rlwc, a monoclonal antibody and PD-1 inhibitor.
In February, it was approved as the first immunotherapy to treat patients with locally advanced or metastatic basal cell carcinoma that was previously treated with a hedgehog pathway inhibitor or for whom a hedgehog inhibitor is inappropriate.
Cemiplimab-rlwc previously received FDA approval in 2018 for locally advanced or metastatic cutaneous squamous cell carcinoma for patients who were not eligible for curative surgery or radiotherapy. At the time, Karl Lewis, MD, a professor at the University of Colorado at Denver, Aurora, and a trial investigator, predicted that the drug “will change the treatment paradigm for patients with advanced basal cell carcinoma.”
Outperforms chemotherapy
The approval for use in NSCLC is based on results from the phase 3, open-label EMPOWER-Lung 1 trial, which randomly assigned 710 patients in a 1:1 ratio to receive either cemiplimab-rwlc or platinum-doublet chemotherapy. Patients had either locally advanced NSCLC and were not candidates for surgical resection or definitive chemoradiotherapy, or they had metastatic NSCLC.
Patients in the experimental arm received cemiplimab-rwlc 350 mg intravenously every 3 weeks. The primary efficacy outcome measures were overall survival (OS) and progression-free survival (PFS), determined on the basis of blinded independent central review.
Results showed statistically significant improvements in both outcomes. Median OS was 22.1 months with cemiplimab-rwlc versus 14.3 months with chemotherapy (hazard ratio, 0.68; P = .0022). Median PFS was 6.2 months versus 5.6 months (HR, 0.59; P < .0001).
The confirmed overall response rate was 37% for the cemiplimab arm versus 21% for the chemotherapy arm.
The most common adverse reactions (>10%) with cemiplimab-rlwc were musculoskeletal pain, rash, anemia, fatigue, decreased appetite, pneumonia, and cough.
This approval “means physicians and patients have a potent new treatment option against this deadly disease,” said Naiyer Rizvi, MD, Price Family Professor of Medicine, director of thoracic oncology, and codirector of cancer immunotherapy at Columbia University Irving Medical Center, New York, in a statement. He was a steering committee member on the EMPOWER-Lung-1 Trial.
“Notably, Libtayo was approved based on a pivotal trial where most chemotherapy patients crossed over to Libtayo following disease progression, and that allowed for frequently underrepresented patients who had pretreated and clinically stable brain metastases or who had locally advanced disease and were not candidates for definitive chemoradiation,” said Dr. Rizvi. “This gives doctors important new data when considering Libtayo for the varied patients and situations they treat in daily clinical practice.”
A version of this article first appeared on Medscape.com.
Specifically, the indication is for first-line treatment as monotherapy for patients with locally advanced or metastatic disease who are not candidates for surgical resection or definitive chemoradiotherapy and whose tumors have a high expression of programmed death–ligand 1 (PD-L1) (Tumor Proportion Score >50%), as determined by an FDA-approved test, with no EGFR, ALK, or ROS1 aberrations.
This is the third indication for cemiplimab-rlwc, a monoclonal antibody and PD-1 inhibitor.
In February, it was approved as the first immunotherapy to treat patients with locally advanced or metastatic basal cell carcinoma that was previously treated with a hedgehog pathway inhibitor or for whom a hedgehog inhibitor is inappropriate.
Cemiplimab-rlwc previously received FDA approval in 2018 for locally advanced or metastatic cutaneous squamous cell carcinoma for patients who were not eligible for curative surgery or radiotherapy. At the time, Karl Lewis, MD, a professor at the University of Colorado at Denver, Aurora, and a trial investigator, predicted that the drug “will change the treatment paradigm for patients with advanced basal cell carcinoma.”
Outperforms chemotherapy
The approval for use in NSCLC is based on results from the phase 3, open-label EMPOWER-Lung 1 trial, which randomly assigned 710 patients in a 1:1 ratio to receive either cemiplimab-rwlc or platinum-doublet chemotherapy. Patients had either locally advanced NSCLC and were not candidates for surgical resection or definitive chemoradiotherapy, or they had metastatic NSCLC.
Patients in the experimental arm received cemiplimab-rwlc 350 mg intravenously every 3 weeks. The primary efficacy outcome measures were overall survival (OS) and progression-free survival (PFS), determined on the basis of blinded independent central review.
Results showed statistically significant improvements in both outcomes. Median OS was 22.1 months with cemiplimab-rwlc versus 14.3 months with chemotherapy (hazard ratio, 0.68; P = .0022). Median PFS was 6.2 months versus 5.6 months (HR, 0.59; P < .0001).
The confirmed overall response rate was 37% for the cemiplimab arm versus 21% for the chemotherapy arm.
The most common adverse reactions (>10%) with cemiplimab-rlwc were musculoskeletal pain, rash, anemia, fatigue, decreased appetite, pneumonia, and cough.
This approval “means physicians and patients have a potent new treatment option against this deadly disease,” said Naiyer Rizvi, MD, Price Family Professor of Medicine, director of thoracic oncology, and codirector of cancer immunotherapy at Columbia University Irving Medical Center, New York, in a statement. He was a steering committee member on the EMPOWER-Lung-1 Trial.
“Notably, Libtayo was approved based on a pivotal trial where most chemotherapy patients crossed over to Libtayo following disease progression, and that allowed for frequently underrepresented patients who had pretreated and clinically stable brain metastases or who had locally advanced disease and were not candidates for definitive chemoradiation,” said Dr. Rizvi. “This gives doctors important new data when considering Libtayo for the varied patients and situations they treat in daily clinical practice.”
A version of this article first appeared on Medscape.com.
Tips offered for treating co-occurring ADHD and SUDs
When Frances R. Levin, MD, began her clinical psychiatry career in the mid-1990s, she spent a lot of time educating colleagues about the validity of an ADHD diagnosis in adults.
“That’s no longer an issue,” Dr. Levin, the Kennedy-Leavy Professor of Psychiatry at Columbia University, New York, said during an annual psychopharmacology update held by the Nevada Psychiatric Association. “But at the time, we often thought, ‘ADHD is something that’s specific to people who are stimulant users.’ In fact, what we found over the years was that these rates are elevated in a range of substance use populations.”
According to National Comorbidity Survey, a nontreatment sample of more than 3,000 adults, individuals who have SUD have two to three times the risk of having ADHD, while individuals who have ADHD have about three times the rate of having an SUD, compared with those who don’t (Am J Psychiatry. 2006;163[4]:716-23). “When you move to treatment samples, the rates also remain quite high,” said Dr. Levin, who is also chief of the division of substance use disorders at the medical center.
“In the general population, the rates of ADHD are 2%-4%. When we look at people who are coming in specifically for treatment of their SUD, the rates are substantially higher, ranging from 10% to 24%.”
According to a 2014 review of medical literature, potential reasons for the association between ADHD and SUD vary and include underlying biologic deficits, such as parental SUDs and genetics; conduct disorder symptoms, such as defiance, rule breaking, and delinquency; poor performance in school, such as low grades, grade retention, or drop-out; and social difficulties, such as rejection from conventional groups or few quality friendships (Annu Rev Clin Psychol. 2014;10:607-39). Other potential pathways include neurocognitive deficits, stress-negative affect models, impulsive anger, and other underlying traits.
One key reason to treat ADHD in patients with SUDs is that they tend to develop the SUD earlier when the ADHD is present, Dr. Levin said. They’re also less likely to be retained in treatment and have a reduced likelihood of going into remission if dependence develops. “Even when they do achieve remission, it seems to take longer for people to reach remission,” she said. “They have more treatment exposure yet do less well in treatment. This can make it more challenging to treat this population.”
One common assumption from clinicians regarding patients with ADHD and a concomitant SUD is that standard treatments for ADHD do not work in active substance users. Another is that, even if treatments work for ADHD, they do not affect the substance use disorder. “Understandably, there is also concern that active substance abusers will misuse and divert their medications,” she said. “Finally, there are often additional psychiatric comorbidities that may make it harder to effectively treat individuals with ADHD and SUD.”
Since 2002, 15 double-blind outpatient studies using stimulants/atomoxetine to treat substance abusers with ADHD have appeared in the medical literature, Dr. Levin said. Only three have included adolescents. “That’s surprising, because up to 40% of kids who come in for treatment, often for cannabis use disorder, will have ADHD, yet there is very little guidance from empirical studies as to how to best treat them,” she said. “There have been several studies looking at atomoxetine to treat substance abusers with ADHD, but results have been mixed. In the cannabis use populations, atomoxetine has not been shown to be effective in treating the substance use disorder, and results are mixed regarding superiority in reducing ADHD symptoms. There is one study showing that ADHD is more likely to be improved in adults with alcohol use disorders with mixed results regarding the alcohol use.”
Overall, most of the outpatient and inpatient studies conducted in this population have demonstrated some signal in terms of reducing ADHD, she said, while a minority of the outpatient studies suggest some benefit in terms of substance use. “What’s interesting is that when you see a response in terms of the ADHD, you often see an improvement in the substance use as well,” Dr. Levin said. “This potentially suggests that patients may be self-medicating their ADHD symptoms or that if the ADHD responds to treatment, then the patient may benefit from the psychosocial interventions that targets the SUD.”
A separate meta-analysis involving more than 1,000 patients found mixed results from pharmacologic interventions and concluded that, while they modestly improved ADHD symptoms, no beneficial effect was seen on drug abstinence or on treatment discontinuation (J Psychopharmacol. 2015 Jan;29[1]:15-23). “I would argue that you don’t need to be as nihilistic about this as the meta-analysis might suggest, because the devil’s in the details,” said Dr. Levin, whose own research was included in the work.
“First of all, many of the studies had high drop-out rates. The outcome measures were variable, and some of the studies used formulations with poor bioavailability. Also, trials that evaluated atomoxetine or stimulants were combined, which may be problematic given the different mechanisms of action. Further, the meta-analysis did not include two recent placebo-controlled trials in adults with stimulant-use disorders that both found that higher dosing of a long-acting stimulant resulted in greater improvements in ADHD symptoms and stimulant use” (Addict. 2014;109[3]:440-9 and JAMA Psychiatry. 2015;72[6]:593-602).
Dr. Levin went on to note that there are few empirical data to guide treatment for those who have multiple psychiatric disorders, let alone treatment for ADHD and SUDs without additional psychiatric disorders. The challenge is what to treat first and/or how to treat the concomitant conditions safely.
“Generally, if possible, treat what is most clinically impairing first,” she said. “Overall, both stimulants and atomoxetine may work for ADHD even in the presence of additional depression, anxiety disorders, and substance use disorders.”
She cautioned against treating a patient with ADHD medication if there is a preexisting psychosis or bipolar illness. “If you start a stimulant or atomoxetine and psychosis or mania occurs, you clearly want to stop the medication and reassess,” she said. Researchers found that the risk of precipitating mania with a stimulant is uncommon if you alleviate symptoms first with a mood stabilizer. “This is a situation where you probably want to treat the bipolar illness first, but it does not preclude the treatment of ADHD once the mood stabilization has occurred,” she said.
In patients with ADHD and anxiety, she often treats the ADHD first, “because oftentimes the anxiety is driven by the procrastination and the inability to get things done,” she explained. “It’s important to determine whether the anxiety is an independent disorder rather than symptoms of ADHD. Inner restlessness can be described as anxiety.”
When there are concerns that preclude the use of a controlled medication, there are medications, in addition to atomoxetine, that might be considered. While bupropion is not Food and Drug Administration approved for ADHD, it might be useful in comorbid mood disorders for nicotine dependence. Other off-label medications that may help include guanfacine, modafinil, and tricyclic antidepressants.
“To date, robust dosing of long-acting amphetamine or methylphenidate formulations have been shown to be effective for patients with stimulant-use disorder, but as mentioned earlier, the data only come from two studies,” she said.
In order to determine whether stimulant treatment is yielding a benefit in a patient with co-occurring ADHD and SUD, she recommends carrying out a structured assessment of ADHD symptoms. Monitoring for functional improvement is also key.
“If there is no improvement in social, occupational, or academic settings and the patient is still actively using drugs, then there is no reason to keep prescribing,” she said. Close monitoring for cardiovascular or other psychiatric symptoms are key as well. Further, for those individuals with both ADHD and a substance-use disorder, it is critical that both are targeted for treatment.
Dr. Levin reported that she has received research, training, or salary support from the National Institute on Drug Abuse, New York state, and the Substance Abuse and Mental Health Services Administration. She has also received or currently receives industry support from Indivior and U.S. World Meds and for medication and from Major League Baseball. In addition, Dr. Levin has been an unpaid scientific advisory board member for Alkermes, Indivior, and Novartis.
When Frances R. Levin, MD, began her clinical psychiatry career in the mid-1990s, she spent a lot of time educating colleagues about the validity of an ADHD diagnosis in adults.
“That’s no longer an issue,” Dr. Levin, the Kennedy-Leavy Professor of Psychiatry at Columbia University, New York, said during an annual psychopharmacology update held by the Nevada Psychiatric Association. “But at the time, we often thought, ‘ADHD is something that’s specific to people who are stimulant users.’ In fact, what we found over the years was that these rates are elevated in a range of substance use populations.”
According to National Comorbidity Survey, a nontreatment sample of more than 3,000 adults, individuals who have SUD have two to three times the risk of having ADHD, while individuals who have ADHD have about three times the rate of having an SUD, compared with those who don’t (Am J Psychiatry. 2006;163[4]:716-23). “When you move to treatment samples, the rates also remain quite high,” said Dr. Levin, who is also chief of the division of substance use disorders at the medical center.
“In the general population, the rates of ADHD are 2%-4%. When we look at people who are coming in specifically for treatment of their SUD, the rates are substantially higher, ranging from 10% to 24%.”
According to a 2014 review of medical literature, potential reasons for the association between ADHD and SUD vary and include underlying biologic deficits, such as parental SUDs and genetics; conduct disorder symptoms, such as defiance, rule breaking, and delinquency; poor performance in school, such as low grades, grade retention, or drop-out; and social difficulties, such as rejection from conventional groups or few quality friendships (Annu Rev Clin Psychol. 2014;10:607-39). Other potential pathways include neurocognitive deficits, stress-negative affect models, impulsive anger, and other underlying traits.
One key reason to treat ADHD in patients with SUDs is that they tend to develop the SUD earlier when the ADHD is present, Dr. Levin said. They’re also less likely to be retained in treatment and have a reduced likelihood of going into remission if dependence develops. “Even when they do achieve remission, it seems to take longer for people to reach remission,” she said. “They have more treatment exposure yet do less well in treatment. This can make it more challenging to treat this population.”
One common assumption from clinicians regarding patients with ADHD and a concomitant SUD is that standard treatments for ADHD do not work in active substance users. Another is that, even if treatments work for ADHD, they do not affect the substance use disorder. “Understandably, there is also concern that active substance abusers will misuse and divert their medications,” she said. “Finally, there are often additional psychiatric comorbidities that may make it harder to effectively treat individuals with ADHD and SUD.”
Since 2002, 15 double-blind outpatient studies using stimulants/atomoxetine to treat substance abusers with ADHD have appeared in the medical literature, Dr. Levin said. Only three have included adolescents. “That’s surprising, because up to 40% of kids who come in for treatment, often for cannabis use disorder, will have ADHD, yet there is very little guidance from empirical studies as to how to best treat them,” she said. “There have been several studies looking at atomoxetine to treat substance abusers with ADHD, but results have been mixed. In the cannabis use populations, atomoxetine has not been shown to be effective in treating the substance use disorder, and results are mixed regarding superiority in reducing ADHD symptoms. There is one study showing that ADHD is more likely to be improved in adults with alcohol use disorders with mixed results regarding the alcohol use.”
Overall, most of the outpatient and inpatient studies conducted in this population have demonstrated some signal in terms of reducing ADHD, she said, while a minority of the outpatient studies suggest some benefit in terms of substance use. “What’s interesting is that when you see a response in terms of the ADHD, you often see an improvement in the substance use as well,” Dr. Levin said. “This potentially suggests that patients may be self-medicating their ADHD symptoms or that if the ADHD responds to treatment, then the patient may benefit from the psychosocial interventions that targets the SUD.”
A separate meta-analysis involving more than 1,000 patients found mixed results from pharmacologic interventions and concluded that, while they modestly improved ADHD symptoms, no beneficial effect was seen on drug abstinence or on treatment discontinuation (J Psychopharmacol. 2015 Jan;29[1]:15-23). “I would argue that you don’t need to be as nihilistic about this as the meta-analysis might suggest, because the devil’s in the details,” said Dr. Levin, whose own research was included in the work.
“First of all, many of the studies had high drop-out rates. The outcome measures were variable, and some of the studies used formulations with poor bioavailability. Also, trials that evaluated atomoxetine or stimulants were combined, which may be problematic given the different mechanisms of action. Further, the meta-analysis did not include two recent placebo-controlled trials in adults with stimulant-use disorders that both found that higher dosing of a long-acting stimulant resulted in greater improvements in ADHD symptoms and stimulant use” (Addict. 2014;109[3]:440-9 and JAMA Psychiatry. 2015;72[6]:593-602).
Dr. Levin went on to note that there are few empirical data to guide treatment for those who have multiple psychiatric disorders, let alone treatment for ADHD and SUDs without additional psychiatric disorders. The challenge is what to treat first and/or how to treat the concomitant conditions safely.
“Generally, if possible, treat what is most clinically impairing first,” she said. “Overall, both stimulants and atomoxetine may work for ADHD even in the presence of additional depression, anxiety disorders, and substance use disorders.”
She cautioned against treating a patient with ADHD medication if there is a preexisting psychosis or bipolar illness. “If you start a stimulant or atomoxetine and psychosis or mania occurs, you clearly want to stop the medication and reassess,” she said. Researchers found that the risk of precipitating mania with a stimulant is uncommon if you alleviate symptoms first with a mood stabilizer. “This is a situation where you probably want to treat the bipolar illness first, but it does not preclude the treatment of ADHD once the mood stabilization has occurred,” she said.
In patients with ADHD and anxiety, she often treats the ADHD first, “because oftentimes the anxiety is driven by the procrastination and the inability to get things done,” she explained. “It’s important to determine whether the anxiety is an independent disorder rather than symptoms of ADHD. Inner restlessness can be described as anxiety.”
When there are concerns that preclude the use of a controlled medication, there are medications, in addition to atomoxetine, that might be considered. While bupropion is not Food and Drug Administration approved for ADHD, it might be useful in comorbid mood disorders for nicotine dependence. Other off-label medications that may help include guanfacine, modafinil, and tricyclic antidepressants.
“To date, robust dosing of long-acting amphetamine or methylphenidate formulations have been shown to be effective for patients with stimulant-use disorder, but as mentioned earlier, the data only come from two studies,” she said.
In order to determine whether stimulant treatment is yielding a benefit in a patient with co-occurring ADHD and SUD, she recommends carrying out a structured assessment of ADHD symptoms. Monitoring for functional improvement is also key.
“If there is no improvement in social, occupational, or academic settings and the patient is still actively using drugs, then there is no reason to keep prescribing,” she said. Close monitoring for cardiovascular or other psychiatric symptoms are key as well. Further, for those individuals with both ADHD and a substance-use disorder, it is critical that both are targeted for treatment.
Dr. Levin reported that she has received research, training, or salary support from the National Institute on Drug Abuse, New York state, and the Substance Abuse and Mental Health Services Administration. She has also received or currently receives industry support from Indivior and U.S. World Meds and for medication and from Major League Baseball. In addition, Dr. Levin has been an unpaid scientific advisory board member for Alkermes, Indivior, and Novartis.
When Frances R. Levin, MD, began her clinical psychiatry career in the mid-1990s, she spent a lot of time educating colleagues about the validity of an ADHD diagnosis in adults.
“That’s no longer an issue,” Dr. Levin, the Kennedy-Leavy Professor of Psychiatry at Columbia University, New York, said during an annual psychopharmacology update held by the Nevada Psychiatric Association. “But at the time, we often thought, ‘ADHD is something that’s specific to people who are stimulant users.’ In fact, what we found over the years was that these rates are elevated in a range of substance use populations.”
According to National Comorbidity Survey, a nontreatment sample of more than 3,000 adults, individuals who have SUD have two to three times the risk of having ADHD, while individuals who have ADHD have about three times the rate of having an SUD, compared with those who don’t (Am J Psychiatry. 2006;163[4]:716-23). “When you move to treatment samples, the rates also remain quite high,” said Dr. Levin, who is also chief of the division of substance use disorders at the medical center.
“In the general population, the rates of ADHD are 2%-4%. When we look at people who are coming in specifically for treatment of their SUD, the rates are substantially higher, ranging from 10% to 24%.”
According to a 2014 review of medical literature, potential reasons for the association between ADHD and SUD vary and include underlying biologic deficits, such as parental SUDs and genetics; conduct disorder symptoms, such as defiance, rule breaking, and delinquency; poor performance in school, such as low grades, grade retention, or drop-out; and social difficulties, such as rejection from conventional groups or few quality friendships (Annu Rev Clin Psychol. 2014;10:607-39). Other potential pathways include neurocognitive deficits, stress-negative affect models, impulsive anger, and other underlying traits.
One key reason to treat ADHD in patients with SUDs is that they tend to develop the SUD earlier when the ADHD is present, Dr. Levin said. They’re also less likely to be retained in treatment and have a reduced likelihood of going into remission if dependence develops. “Even when they do achieve remission, it seems to take longer for people to reach remission,” she said. “They have more treatment exposure yet do less well in treatment. This can make it more challenging to treat this population.”
One common assumption from clinicians regarding patients with ADHD and a concomitant SUD is that standard treatments for ADHD do not work in active substance users. Another is that, even if treatments work for ADHD, they do not affect the substance use disorder. “Understandably, there is also concern that active substance abusers will misuse and divert their medications,” she said. “Finally, there are often additional psychiatric comorbidities that may make it harder to effectively treat individuals with ADHD and SUD.”
Since 2002, 15 double-blind outpatient studies using stimulants/atomoxetine to treat substance abusers with ADHD have appeared in the medical literature, Dr. Levin said. Only three have included adolescents. “That’s surprising, because up to 40% of kids who come in for treatment, often for cannabis use disorder, will have ADHD, yet there is very little guidance from empirical studies as to how to best treat them,” she said. “There have been several studies looking at atomoxetine to treat substance abusers with ADHD, but results have been mixed. In the cannabis use populations, atomoxetine has not been shown to be effective in treating the substance use disorder, and results are mixed regarding superiority in reducing ADHD symptoms. There is one study showing that ADHD is more likely to be improved in adults with alcohol use disorders with mixed results regarding the alcohol use.”
Overall, most of the outpatient and inpatient studies conducted in this population have demonstrated some signal in terms of reducing ADHD, she said, while a minority of the outpatient studies suggest some benefit in terms of substance use. “What’s interesting is that when you see a response in terms of the ADHD, you often see an improvement in the substance use as well,” Dr. Levin said. “This potentially suggests that patients may be self-medicating their ADHD symptoms or that if the ADHD responds to treatment, then the patient may benefit from the psychosocial interventions that targets the SUD.”
A separate meta-analysis involving more than 1,000 patients found mixed results from pharmacologic interventions and concluded that, while they modestly improved ADHD symptoms, no beneficial effect was seen on drug abstinence or on treatment discontinuation (J Psychopharmacol. 2015 Jan;29[1]:15-23). “I would argue that you don’t need to be as nihilistic about this as the meta-analysis might suggest, because the devil’s in the details,” said Dr. Levin, whose own research was included in the work.
“First of all, many of the studies had high drop-out rates. The outcome measures were variable, and some of the studies used formulations with poor bioavailability. Also, trials that evaluated atomoxetine or stimulants were combined, which may be problematic given the different mechanisms of action. Further, the meta-analysis did not include two recent placebo-controlled trials in adults with stimulant-use disorders that both found that higher dosing of a long-acting stimulant resulted in greater improvements in ADHD symptoms and stimulant use” (Addict. 2014;109[3]:440-9 and JAMA Psychiatry. 2015;72[6]:593-602).
Dr. Levin went on to note that there are few empirical data to guide treatment for those who have multiple psychiatric disorders, let alone treatment for ADHD and SUDs without additional psychiatric disorders. The challenge is what to treat first and/or how to treat the concomitant conditions safely.
“Generally, if possible, treat what is most clinically impairing first,” she said. “Overall, both stimulants and atomoxetine may work for ADHD even in the presence of additional depression, anxiety disorders, and substance use disorders.”
She cautioned against treating a patient with ADHD medication if there is a preexisting psychosis or bipolar illness. “If you start a stimulant or atomoxetine and psychosis or mania occurs, you clearly want to stop the medication and reassess,” she said. Researchers found that the risk of precipitating mania with a stimulant is uncommon if you alleviate symptoms first with a mood stabilizer. “This is a situation where you probably want to treat the bipolar illness first, but it does not preclude the treatment of ADHD once the mood stabilization has occurred,” she said.
In patients with ADHD and anxiety, she often treats the ADHD first, “because oftentimes the anxiety is driven by the procrastination and the inability to get things done,” she explained. “It’s important to determine whether the anxiety is an independent disorder rather than symptoms of ADHD. Inner restlessness can be described as anxiety.”
When there are concerns that preclude the use of a controlled medication, there are medications, in addition to atomoxetine, that might be considered. While bupropion is not Food and Drug Administration approved for ADHD, it might be useful in comorbid mood disorders for nicotine dependence. Other off-label medications that may help include guanfacine, modafinil, and tricyclic antidepressants.
“To date, robust dosing of long-acting amphetamine or methylphenidate formulations have been shown to be effective for patients with stimulant-use disorder, but as mentioned earlier, the data only come from two studies,” she said.
In order to determine whether stimulant treatment is yielding a benefit in a patient with co-occurring ADHD and SUD, she recommends carrying out a structured assessment of ADHD symptoms. Monitoring for functional improvement is also key.
“If there is no improvement in social, occupational, or academic settings and the patient is still actively using drugs, then there is no reason to keep prescribing,” she said. Close monitoring for cardiovascular or other psychiatric symptoms are key as well. Further, for those individuals with both ADHD and a substance-use disorder, it is critical that both are targeted for treatment.
Dr. Levin reported that she has received research, training, or salary support from the National Institute on Drug Abuse, New York state, and the Substance Abuse and Mental Health Services Administration. She has also received or currently receives industry support from Indivior and U.S. World Meds and for medication and from Major League Baseball. In addition, Dr. Levin has been an unpaid scientific advisory board member for Alkermes, Indivior, and Novartis.
FROM NPA 2021
Large study finds trans men on testosterone at risk for blood clots
Over 10% of transgender men (females transitioning to male) who take testosterone develop high hematocrit levels that could put them at greater risk for a thrombotic event, and the largest increase in levels occurs in the first year after starting therapy, a new Dutch study indicates.
Erythrocytosis, defined as a hematocrit greater than 0.50 L/L, is a potentially serious side effect of testosterone therapy, say Milou Cecilia Madsen, MD, and colleagues in their article published online Feb. 18, 2021, in the Journal of Clinical Endocrinology & Metabolism.
When hematocrit was measured twice, 11.1% of the cohort of 1073 trans men had levels in excess of 0.50 L/L over a 20-year follow-up.
“Erythrocytosis is common in transgender men treated with testosterone, especially in those who smoke, have [a] high BMI [body mass index], and [who] use testosterone injections,” Dr. Madsen, of the VU University Medical Center Amsterdam, said in a statement from the Endocrine Society.
“A reasonable first step in the care of transgender men with high red blood cells while on testosterone is to advise them to quit smoking, switch injectable testosterone to gel, and, if BMI is high, to lose weight,” she added.
First large study of testosterone in trans men with 20-year follow-up
Transgender men often undergo testosterone therapy as part of gender-affirming treatment.
Secondary erythrocytosis, a condition where the body makes too many red blood cells, is a common side effect of testosterone therapy that can increase the risk of thrombolic events, heart attack, and stroke, Dr. Madsen and colleagues explained.
This is the first study of a large cohort of trans men taking testosterone therapy followed for up to 20 years. Because of the large sample size, statistical analysis with many determinants could be performed. And because of the long follow-up, a clear time relation between initiation of testosterone therapy and hematocrit could be studied, they noted.
Participants were part of the Amsterdam Cohort of Gender Dysphoria study, a large cohort of individuals seen at the Center of Expertise on Gender Dysphoria at Amsterdam University Medical Center between 1972 and 2015.
Laboratory measurements taken between 2004 and 2018 were available for analysis. Trans men visited the center every 3-6 months during their first year of testosterone therapy and were then monitored every year or every other year.
Long-acting undecanoate injection was associated with the highest risk of a hematocrit level greater than 0.50 L/L, and the risk of erythrocytosis in those who took long-acting intramuscular injections was about threefold higher, compared with testosterone gel (adjusted odds ratio, 3.1).
In contrast, short-acting ester injections and oral administration of testosterone had a similar risk for erythrocytosis, as did testosterone gel.
Other determinants of elevated hematocrit included smoking, medical history of a number of comorbid conditions, and older age on initiation of testosterone.
In contrast, “higher testosterone levels per se were not associated with an increased odds of hematocrit greater than 0.50 L/L”, the authors noted.
Current advice for trans men based on old guidance for hypogonadism
The authors said that current advice for trans men is based on recommendations for testosterone-treated hypogonadal cis men (those assigned male at birth) from 2008, which advises a hematocrit greater than 0.50 L/L has a moderate to high risk of adverse outcome. For levels greater than 0.54 L/L, cessation of testosterone therapy, a dose reduction, or therapeutic phlebotomy to reduce the risk of adverse events is advised. For levels 0.50-0.54 L/L, no clear advice is given.
But questions remain as to whether these guidelines are applicable to trans men because the duration of testosterone therapy is much longer in trans men and hormone treatment often cannot be discontinued without causing distress.
Meanwhile, hematology guidelines indicate an upper limit for hematocrit for cis females of 0.48 L/L.
“It could be argued that the upper limit for cis females should be applied, as trans men are born with female genetics,” the authors said. “This is a subject for further research.”
Duration of testosterone therapy impacts risk of erythrocytosis
In the study, the researchers found that longer duration of testosterone therapy increased the risk of developing hematocrit levels greater than 0.50 L/L. For example, after 1 year, the cumulative incidence of erythrocytosis was 8%; after 10 years, it was 38%; and after 14 years, it was 50%.
Until more specific guidance is developed for trans men, if hematocrit levels rise to 0.50-0.54 L/L, the researchers suggested taking “reasonable” steps to prevent a further increase:
- Consider switching patients who use injectable testosterone to transdermal products.
- Advise patients with a BMI greater than 25 kg/m2 to lose weight to attain a BMI of 18.5-25.
- Advise patients to stop smoking.
- Pursue treatment optimization for chronic lung disease or sleep apnea.
The study had no external funding. The authors reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Over 10% of transgender men (females transitioning to male) who take testosterone develop high hematocrit levels that could put them at greater risk for a thrombotic event, and the largest increase in levels occurs in the first year after starting therapy, a new Dutch study indicates.
Erythrocytosis, defined as a hematocrit greater than 0.50 L/L, is a potentially serious side effect of testosterone therapy, say Milou Cecilia Madsen, MD, and colleagues in their article published online Feb. 18, 2021, in the Journal of Clinical Endocrinology & Metabolism.
When hematocrit was measured twice, 11.1% of the cohort of 1073 trans men had levels in excess of 0.50 L/L over a 20-year follow-up.
“Erythrocytosis is common in transgender men treated with testosterone, especially in those who smoke, have [a] high BMI [body mass index], and [who] use testosterone injections,” Dr. Madsen, of the VU University Medical Center Amsterdam, said in a statement from the Endocrine Society.
“A reasonable first step in the care of transgender men with high red blood cells while on testosterone is to advise them to quit smoking, switch injectable testosterone to gel, and, if BMI is high, to lose weight,” she added.
First large study of testosterone in trans men with 20-year follow-up
Transgender men often undergo testosterone therapy as part of gender-affirming treatment.
Secondary erythrocytosis, a condition where the body makes too many red blood cells, is a common side effect of testosterone therapy that can increase the risk of thrombolic events, heart attack, and stroke, Dr. Madsen and colleagues explained.
This is the first study of a large cohort of trans men taking testosterone therapy followed for up to 20 years. Because of the large sample size, statistical analysis with many determinants could be performed. And because of the long follow-up, a clear time relation between initiation of testosterone therapy and hematocrit could be studied, they noted.
Participants were part of the Amsterdam Cohort of Gender Dysphoria study, a large cohort of individuals seen at the Center of Expertise on Gender Dysphoria at Amsterdam University Medical Center between 1972 and 2015.
Laboratory measurements taken between 2004 and 2018 were available for analysis. Trans men visited the center every 3-6 months during their first year of testosterone therapy and were then monitored every year or every other year.
Long-acting undecanoate injection was associated with the highest risk of a hematocrit level greater than 0.50 L/L, and the risk of erythrocytosis in those who took long-acting intramuscular injections was about threefold higher, compared with testosterone gel (adjusted odds ratio, 3.1).
In contrast, short-acting ester injections and oral administration of testosterone had a similar risk for erythrocytosis, as did testosterone gel.
Other determinants of elevated hematocrit included smoking, medical history of a number of comorbid conditions, and older age on initiation of testosterone.
In contrast, “higher testosterone levels per se were not associated with an increased odds of hematocrit greater than 0.50 L/L”, the authors noted.
Current advice for trans men based on old guidance for hypogonadism
The authors said that current advice for trans men is based on recommendations for testosterone-treated hypogonadal cis men (those assigned male at birth) from 2008, which advises a hematocrit greater than 0.50 L/L has a moderate to high risk of adverse outcome. For levels greater than 0.54 L/L, cessation of testosterone therapy, a dose reduction, or therapeutic phlebotomy to reduce the risk of adverse events is advised. For levels 0.50-0.54 L/L, no clear advice is given.
But questions remain as to whether these guidelines are applicable to trans men because the duration of testosterone therapy is much longer in trans men and hormone treatment often cannot be discontinued without causing distress.
Meanwhile, hematology guidelines indicate an upper limit for hematocrit for cis females of 0.48 L/L.
“It could be argued that the upper limit for cis females should be applied, as trans men are born with female genetics,” the authors said. “This is a subject for further research.”
Duration of testosterone therapy impacts risk of erythrocytosis
In the study, the researchers found that longer duration of testosterone therapy increased the risk of developing hematocrit levels greater than 0.50 L/L. For example, after 1 year, the cumulative incidence of erythrocytosis was 8%; after 10 years, it was 38%; and after 14 years, it was 50%.
Until more specific guidance is developed for trans men, if hematocrit levels rise to 0.50-0.54 L/L, the researchers suggested taking “reasonable” steps to prevent a further increase:
- Consider switching patients who use injectable testosterone to transdermal products.
- Advise patients with a BMI greater than 25 kg/m2 to lose weight to attain a BMI of 18.5-25.
- Advise patients to stop smoking.
- Pursue treatment optimization for chronic lung disease or sleep apnea.
The study had no external funding. The authors reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Over 10% of transgender men (females transitioning to male) who take testosterone develop high hematocrit levels that could put them at greater risk for a thrombotic event, and the largest increase in levels occurs in the first year after starting therapy, a new Dutch study indicates.
Erythrocytosis, defined as a hematocrit greater than 0.50 L/L, is a potentially serious side effect of testosterone therapy, say Milou Cecilia Madsen, MD, and colleagues in their article published online Feb. 18, 2021, in the Journal of Clinical Endocrinology & Metabolism.
When hematocrit was measured twice, 11.1% of the cohort of 1073 trans men had levels in excess of 0.50 L/L over a 20-year follow-up.
“Erythrocytosis is common in transgender men treated with testosterone, especially in those who smoke, have [a] high BMI [body mass index], and [who] use testosterone injections,” Dr. Madsen, of the VU University Medical Center Amsterdam, said in a statement from the Endocrine Society.
“A reasonable first step in the care of transgender men with high red blood cells while on testosterone is to advise them to quit smoking, switch injectable testosterone to gel, and, if BMI is high, to lose weight,” she added.
First large study of testosterone in trans men with 20-year follow-up
Transgender men often undergo testosterone therapy as part of gender-affirming treatment.
Secondary erythrocytosis, a condition where the body makes too many red blood cells, is a common side effect of testosterone therapy that can increase the risk of thrombolic events, heart attack, and stroke, Dr. Madsen and colleagues explained.
This is the first study of a large cohort of trans men taking testosterone therapy followed for up to 20 years. Because of the large sample size, statistical analysis with many determinants could be performed. And because of the long follow-up, a clear time relation between initiation of testosterone therapy and hematocrit could be studied, they noted.
Participants were part of the Amsterdam Cohort of Gender Dysphoria study, a large cohort of individuals seen at the Center of Expertise on Gender Dysphoria at Amsterdam University Medical Center between 1972 and 2015.
Laboratory measurements taken between 2004 and 2018 were available for analysis. Trans men visited the center every 3-6 months during their first year of testosterone therapy and were then monitored every year or every other year.
Long-acting undecanoate injection was associated with the highest risk of a hematocrit level greater than 0.50 L/L, and the risk of erythrocytosis in those who took long-acting intramuscular injections was about threefold higher, compared with testosterone gel (adjusted odds ratio, 3.1).
In contrast, short-acting ester injections and oral administration of testosterone had a similar risk for erythrocytosis, as did testosterone gel.
Other determinants of elevated hematocrit included smoking, medical history of a number of comorbid conditions, and older age on initiation of testosterone.
In contrast, “higher testosterone levels per se were not associated with an increased odds of hematocrit greater than 0.50 L/L”, the authors noted.
Current advice for trans men based on old guidance for hypogonadism
The authors said that current advice for trans men is based on recommendations for testosterone-treated hypogonadal cis men (those assigned male at birth) from 2008, which advises a hematocrit greater than 0.50 L/L has a moderate to high risk of adverse outcome. For levels greater than 0.54 L/L, cessation of testosterone therapy, a dose reduction, or therapeutic phlebotomy to reduce the risk of adverse events is advised. For levels 0.50-0.54 L/L, no clear advice is given.
But questions remain as to whether these guidelines are applicable to trans men because the duration of testosterone therapy is much longer in trans men and hormone treatment often cannot be discontinued without causing distress.
Meanwhile, hematology guidelines indicate an upper limit for hematocrit for cis females of 0.48 L/L.
“It could be argued that the upper limit for cis females should be applied, as trans men are born with female genetics,” the authors said. “This is a subject for further research.”
Duration of testosterone therapy impacts risk of erythrocytosis
In the study, the researchers found that longer duration of testosterone therapy increased the risk of developing hematocrit levels greater than 0.50 L/L. For example, after 1 year, the cumulative incidence of erythrocytosis was 8%; after 10 years, it was 38%; and after 14 years, it was 50%.
Until more specific guidance is developed for trans men, if hematocrit levels rise to 0.50-0.54 L/L, the researchers suggested taking “reasonable” steps to prevent a further increase:
- Consider switching patients who use injectable testosterone to transdermal products.
- Advise patients with a BMI greater than 25 kg/m2 to lose weight to attain a BMI of 18.5-25.
- Advise patients to stop smoking.
- Pursue treatment optimization for chronic lung disease or sleep apnea.
The study had no external funding. The authors reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.





