User login
Lifestyle Medicine: Not Just for the Wealthy
Primary care clinicians understand that addressing lifestyle-related chronic disease health disparities in minority and lower-income communities is a significant opportunity to alleviate unnecessary suffering. Disparate health outcomes associated with underlying comorbidities during the COVID pandemic exposed the urgency of this problem.
When it comes to delivering evidence-based therapeutic lifestyle behavior interventions to these populations, however, there is a misconception that lifestyle medicine is only for the wealthy. Such a misconception needlessly widens the gap in health disparities because the truth is that everyone deserves access to lifestyle medicine. Fortunately, there are numerous successful examples of delivering these services to underresourced patients. We can all contribute to narrowing health inequities by sourcing increasingly abundant lifestyle medicine resources.
All patients’ lived experiences are unique, and there is a wide range of potential challenges to achieving lifestyle behavior change. Ignoring these obstacles is a disservice to patients and almost certainly results in treatment failure. Requirements to document SDOH have been a tremendous initial step.
The next step is to have conversations with every patient about the powerful outcomes of even small lifestyle changes. All too often, clinicians forgo conversations on lifestyle change with patients affected by adverse SDOH and assume that social obstacles automatically mean that patients are neither willing nor able to attempt behavior modification. Instead, it is an opportunity for clinicians, particularly those certified in lifestyle medicine, to meet patients where they are, work with them to identify solutions, and provide referrals to community-based organizations with resources to help.
Small Steps to Big Changes
Not all lifestyle behavior interventions need to be programmatic or time intensive. Clinicians can guide patients toward simple but specific actions that can make a difference in health outcomes over time. Small steps, like eating one can of beans or two bags of frozen leafy greens each week, are a good start toward adjusted eating patterns. The American College of Lifestyle Medicine offers a whole-food, plant-predominant meal guide to share with patients.
Individuals can increase their physical activity in their living rooms by doing sit-to-stands or balancing on one leg. Deep breathing and establishing a sleep routine are other lifestyle behavior changes without a price tag.
It is true that early adopters of lifestyle medicine often had difficulty practicing in underresourced communities. Those practitioners were forced to operate on a cash-pay basis, making access to care cost-prohibitive for many patients. However, board certification has been available since 2017, and lifestyle medicine is being integrated into medical schools and residency programs. Many such board-certified clinicians now work in large health systems and bill under the usual methods. There are also frameworks, such as the community-engaged lifestyle medicine model, showing how to treat patients affected by adverse SDOH effectively.
For example, patients at risk for malnutrition because of illnesses like chronic kidney disease, cancer, and heart failure receive medically tailored meals and access to a registered dietitian through a partnership between UC San Diego Health and Mama’s Kitchen. In Pennsylvania’s Lehigh Valley, where 1 in 10 of the approximately 700,000 residents face food insecurity, the Kellyn Foundation delivers fresh food through the Eat Real Food Mobile Market and offers whole-food, plant-predominant cooking classes, interactive elementary school programs focused on healthy lifestyle choices, and therapeutic lifestyle-change programs in community locations. Three months after launching new mobile market sites in Allentown, 1200 households were utilizing $15 weekly food vouchers through the program. Lifestyle medicine clinicians serve inner-city and rural areas in independent practices, large health systems, and community-based practice activities.
To improve access to lifestyle medicine in underresourced communities, more clinicians trained and certified in lifestyle medicine are needed. The Health Equity Achieved through Lifestyle Medicine Initiative supports a diverse lifestyle medicine workforce by offering scholarships to clinicians underrepresented in medicine and is working to train and certify at least one physician within each of the 1400 federally qualified health centers where clinicians are on the front lines of delivering care to the most underserved populations.
A meaningful first step for clinicians to address health disparities is to screen patients for and document SDOH. The American Academy of Family Physicians offers useful tools to screen patients, identify community-based resources, and help patients create action plans to overcome health risks and improve outcomes. In a promising trend to better support addressing SDOH in clinical care, the 2024 Medicare Physician Fee Schedule final rule included new codes to support this effort.
Not every patient will be ready or willing to begin a lifestyle medicine treatment plan. Still, all of them will be grateful for the opportunity to decide for themselves. If we are invested in narrowing health inequities, lifestyle medicine and behavior change must be a topic in clinical encounters with all our patients.
Dr. Collings, director of lifestyle medicine, Silicon Valley Medical Development, and past president, American College of Lifestyle Medicine, Mountain View, California, has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
Primary care clinicians understand that addressing lifestyle-related chronic disease health disparities in minority and lower-income communities is a significant opportunity to alleviate unnecessary suffering. Disparate health outcomes associated with underlying comorbidities during the COVID pandemic exposed the urgency of this problem.
When it comes to delivering evidence-based therapeutic lifestyle behavior interventions to these populations, however, there is a misconception that lifestyle medicine is only for the wealthy. Such a misconception needlessly widens the gap in health disparities because the truth is that everyone deserves access to lifestyle medicine. Fortunately, there are numerous successful examples of delivering these services to underresourced patients. We can all contribute to narrowing health inequities by sourcing increasingly abundant lifestyle medicine resources.
All patients’ lived experiences are unique, and there is a wide range of potential challenges to achieving lifestyle behavior change. Ignoring these obstacles is a disservice to patients and almost certainly results in treatment failure. Requirements to document SDOH have been a tremendous initial step.
The next step is to have conversations with every patient about the powerful outcomes of even small lifestyle changes. All too often, clinicians forgo conversations on lifestyle change with patients affected by adverse SDOH and assume that social obstacles automatically mean that patients are neither willing nor able to attempt behavior modification. Instead, it is an opportunity for clinicians, particularly those certified in lifestyle medicine, to meet patients where they are, work with them to identify solutions, and provide referrals to community-based organizations with resources to help.
Small Steps to Big Changes
Not all lifestyle behavior interventions need to be programmatic or time intensive. Clinicians can guide patients toward simple but specific actions that can make a difference in health outcomes over time. Small steps, like eating one can of beans or two bags of frozen leafy greens each week, are a good start toward adjusted eating patterns. The American College of Lifestyle Medicine offers a whole-food, plant-predominant meal guide to share with patients.
Individuals can increase their physical activity in their living rooms by doing sit-to-stands or balancing on one leg. Deep breathing and establishing a sleep routine are other lifestyle behavior changes without a price tag.
It is true that early adopters of lifestyle medicine often had difficulty practicing in underresourced communities. Those practitioners were forced to operate on a cash-pay basis, making access to care cost-prohibitive for many patients. However, board certification has been available since 2017, and lifestyle medicine is being integrated into medical schools and residency programs. Many such board-certified clinicians now work in large health systems and bill under the usual methods. There are also frameworks, such as the community-engaged lifestyle medicine model, showing how to treat patients affected by adverse SDOH effectively.
For example, patients at risk for malnutrition because of illnesses like chronic kidney disease, cancer, and heart failure receive medically tailored meals and access to a registered dietitian through a partnership between UC San Diego Health and Mama’s Kitchen. In Pennsylvania’s Lehigh Valley, where 1 in 10 of the approximately 700,000 residents face food insecurity, the Kellyn Foundation delivers fresh food through the Eat Real Food Mobile Market and offers whole-food, plant-predominant cooking classes, interactive elementary school programs focused on healthy lifestyle choices, and therapeutic lifestyle-change programs in community locations. Three months after launching new mobile market sites in Allentown, 1200 households were utilizing $15 weekly food vouchers through the program. Lifestyle medicine clinicians serve inner-city and rural areas in independent practices, large health systems, and community-based practice activities.
To improve access to lifestyle medicine in underresourced communities, more clinicians trained and certified in lifestyle medicine are needed. The Health Equity Achieved through Lifestyle Medicine Initiative supports a diverse lifestyle medicine workforce by offering scholarships to clinicians underrepresented in medicine and is working to train and certify at least one physician within each of the 1400 federally qualified health centers where clinicians are on the front lines of delivering care to the most underserved populations.
A meaningful first step for clinicians to address health disparities is to screen patients for and document SDOH. The American Academy of Family Physicians offers useful tools to screen patients, identify community-based resources, and help patients create action plans to overcome health risks and improve outcomes. In a promising trend to better support addressing SDOH in clinical care, the 2024 Medicare Physician Fee Schedule final rule included new codes to support this effort.
Not every patient will be ready or willing to begin a lifestyle medicine treatment plan. Still, all of them will be grateful for the opportunity to decide for themselves. If we are invested in narrowing health inequities, lifestyle medicine and behavior change must be a topic in clinical encounters with all our patients.
Dr. Collings, director of lifestyle medicine, Silicon Valley Medical Development, and past president, American College of Lifestyle Medicine, Mountain View, California, has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
Primary care clinicians understand that addressing lifestyle-related chronic disease health disparities in minority and lower-income communities is a significant opportunity to alleviate unnecessary suffering. Disparate health outcomes associated with underlying comorbidities during the COVID pandemic exposed the urgency of this problem.
When it comes to delivering evidence-based therapeutic lifestyle behavior interventions to these populations, however, there is a misconception that lifestyle medicine is only for the wealthy. Such a misconception needlessly widens the gap in health disparities because the truth is that everyone deserves access to lifestyle medicine. Fortunately, there are numerous successful examples of delivering these services to underresourced patients. We can all contribute to narrowing health inequities by sourcing increasingly abundant lifestyle medicine resources.
All patients’ lived experiences are unique, and there is a wide range of potential challenges to achieving lifestyle behavior change. Ignoring these obstacles is a disservice to patients and almost certainly results in treatment failure. Requirements to document SDOH have been a tremendous initial step.
The next step is to have conversations with every patient about the powerful outcomes of even small lifestyle changes. All too often, clinicians forgo conversations on lifestyle change with patients affected by adverse SDOH and assume that social obstacles automatically mean that patients are neither willing nor able to attempt behavior modification. Instead, it is an opportunity for clinicians, particularly those certified in lifestyle medicine, to meet patients where they are, work with them to identify solutions, and provide referrals to community-based organizations with resources to help.
Small Steps to Big Changes
Not all lifestyle behavior interventions need to be programmatic or time intensive. Clinicians can guide patients toward simple but specific actions that can make a difference in health outcomes over time. Small steps, like eating one can of beans or two bags of frozen leafy greens each week, are a good start toward adjusted eating patterns. The American College of Lifestyle Medicine offers a whole-food, plant-predominant meal guide to share with patients.
Individuals can increase their physical activity in their living rooms by doing sit-to-stands or balancing on one leg. Deep breathing and establishing a sleep routine are other lifestyle behavior changes without a price tag.
It is true that early adopters of lifestyle medicine often had difficulty practicing in underresourced communities. Those practitioners were forced to operate on a cash-pay basis, making access to care cost-prohibitive for many patients. However, board certification has been available since 2017, and lifestyle medicine is being integrated into medical schools and residency programs. Many such board-certified clinicians now work in large health systems and bill under the usual methods. There are also frameworks, such as the community-engaged lifestyle medicine model, showing how to treat patients affected by adverse SDOH effectively.
For example, patients at risk for malnutrition because of illnesses like chronic kidney disease, cancer, and heart failure receive medically tailored meals and access to a registered dietitian through a partnership between UC San Diego Health and Mama’s Kitchen. In Pennsylvania’s Lehigh Valley, where 1 in 10 of the approximately 700,000 residents face food insecurity, the Kellyn Foundation delivers fresh food through the Eat Real Food Mobile Market and offers whole-food, plant-predominant cooking classes, interactive elementary school programs focused on healthy lifestyle choices, and therapeutic lifestyle-change programs in community locations. Three months after launching new mobile market sites in Allentown, 1200 households were utilizing $15 weekly food vouchers through the program. Lifestyle medicine clinicians serve inner-city and rural areas in independent practices, large health systems, and community-based practice activities.
To improve access to lifestyle medicine in underresourced communities, more clinicians trained and certified in lifestyle medicine are needed. The Health Equity Achieved through Lifestyle Medicine Initiative supports a diverse lifestyle medicine workforce by offering scholarships to clinicians underrepresented in medicine and is working to train and certify at least one physician within each of the 1400 federally qualified health centers where clinicians are on the front lines of delivering care to the most underserved populations.
A meaningful first step for clinicians to address health disparities is to screen patients for and document SDOH. The American Academy of Family Physicians offers useful tools to screen patients, identify community-based resources, and help patients create action plans to overcome health risks and improve outcomes. In a promising trend to better support addressing SDOH in clinical care, the 2024 Medicare Physician Fee Schedule final rule included new codes to support this effort.
Not every patient will be ready or willing to begin a lifestyle medicine treatment plan. Still, all of them will be grateful for the opportunity to decide for themselves. If we are invested in narrowing health inequities, lifestyle medicine and behavior change must be a topic in clinical encounters with all our patients.
Dr. Collings, director of lifestyle medicine, Silicon Valley Medical Development, and past president, American College of Lifestyle Medicine, Mountain View, California, has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
Which Medication Is Best? VA Genetic Tests May Have the Answer
The US Department of Veterans Affairs (VA) now has a permanent pharmacogenomics service that provides genetic tests to give clinicians insight into the best medication options for their patients.
The tests, which have no extra cost, are available to all veterans, said pharmacist Jill S. Bates, PharmD, MS, executive director of the VA National Pharmacogenomics Program, who spoke in an interview and a presentation at the annual meeting of the Association of VA Hematology/Oncology.
Genetic testing is “a tool that can help optimize care that we provide for veterans,” she said. “Pharmacogenomics is additional information to help the clinician make a decision. We know that most veterans—greater than 90%—carry a variant in a pharmacogenomics gene that is actionable.”
The genetic tests can provide insight into the optimal medication for multiple conditions such as mental illness, gastrointestinal disorders, cancer, pain, and heart disease. According to a 2019 analysis of over 6 years of data, more than half of the VA patient population used medications whose efficacy may have been affected by detectable genetic variants.
For instance, Bates said tests can let clinicians know whether patients are susceptible to statin-associated muscle adverse effects if they take simvastatin, the cholesterol medication. An estimated 25.6% of the VA population has this variant.
Elsewhere on the cardiac front, an estimated 58.3% of the VA population has a genetic variant that increases sensitivity to the blood thinner warfarin.
Testing could help psychiatrists determine whether certain medications should not be prescribed—or should be prescribed at lower doses—in patients who’ve had adverse reactions to antidepressants, Bates said.
In cancer, Bates said, genetic testing can identify patients who have a genetic variant that boosts toxicity from fluoropyrimidine chemotherapy treatments, which include capecitabine, floxuridine, and fluorouracil. Meanwhile, an estimated 0.9% will have no reaction or limited reaction to capecitabine and fluorouracil, and 4.8% will have hypersensitivity to carbamazepine and oxcarbazepine.
Tests can also identify a genetic variant that can lead to poor metabolism of the chemotherapy drug irinotecan, which is used to treat colon cancer. “In those patients, you’d want to reduce the dose by 20%,” Bates said. In other cases, alternate drugs may be the best strategy to address genetic variations.
Prior to 2019, clinicians had to order pharmacogenomic tests outside of the VA system, according to Bates. That year, a donation from Sanford Health brought VA pharmacogenomics to 40 pilot sites. Since then, more than 88,000 tests have been performed.
The VA has now made its pharmacogenomic program permanent, Bates said. As of early September, testing was available at 139 VA sites and is coming soon to 4 more. It’s not available at another 23 sites that are scattered across the country.
A tool in the VA electronic health record now reminds clinicians about the availability of genetic testing and allows them to order tests. However, testing isn’t available for patients who have had liver transplants or certain bone marrow transplants.
The VA is working on developing decision-making tools to help clinicians determine when the tests are appropriate, Bates said. It typically takes 2 to 3 weeks to get results, she said, adding that external laboratories provide results. “We eventually would like to bring in all pharmacogenomics testing to be conducted within the VA enterprise.”
Bates reported that she had no disclosures.
The US Department of Veterans Affairs (VA) now has a permanent pharmacogenomics service that provides genetic tests to give clinicians insight into the best medication options for their patients.
The tests, which have no extra cost, are available to all veterans, said pharmacist Jill S. Bates, PharmD, MS, executive director of the VA National Pharmacogenomics Program, who spoke in an interview and a presentation at the annual meeting of the Association of VA Hematology/Oncology.
Genetic testing is “a tool that can help optimize care that we provide for veterans,” she said. “Pharmacogenomics is additional information to help the clinician make a decision. We know that most veterans—greater than 90%—carry a variant in a pharmacogenomics gene that is actionable.”
The genetic tests can provide insight into the optimal medication for multiple conditions such as mental illness, gastrointestinal disorders, cancer, pain, and heart disease. According to a 2019 analysis of over 6 years of data, more than half of the VA patient population used medications whose efficacy may have been affected by detectable genetic variants.
For instance, Bates said tests can let clinicians know whether patients are susceptible to statin-associated muscle adverse effects if they take simvastatin, the cholesterol medication. An estimated 25.6% of the VA population has this variant.
Elsewhere on the cardiac front, an estimated 58.3% of the VA population has a genetic variant that increases sensitivity to the blood thinner warfarin.
Testing could help psychiatrists determine whether certain medications should not be prescribed—or should be prescribed at lower doses—in patients who’ve had adverse reactions to antidepressants, Bates said.
In cancer, Bates said, genetic testing can identify patients who have a genetic variant that boosts toxicity from fluoropyrimidine chemotherapy treatments, which include capecitabine, floxuridine, and fluorouracil. Meanwhile, an estimated 0.9% will have no reaction or limited reaction to capecitabine and fluorouracil, and 4.8% will have hypersensitivity to carbamazepine and oxcarbazepine.
Tests can also identify a genetic variant that can lead to poor metabolism of the chemotherapy drug irinotecan, which is used to treat colon cancer. “In those patients, you’d want to reduce the dose by 20%,” Bates said. In other cases, alternate drugs may be the best strategy to address genetic variations.
Prior to 2019, clinicians had to order pharmacogenomic tests outside of the VA system, according to Bates. That year, a donation from Sanford Health brought VA pharmacogenomics to 40 pilot sites. Since then, more than 88,000 tests have been performed.
The VA has now made its pharmacogenomic program permanent, Bates said. As of early September, testing was available at 139 VA sites and is coming soon to 4 more. It’s not available at another 23 sites that are scattered across the country.
A tool in the VA electronic health record now reminds clinicians about the availability of genetic testing and allows them to order tests. However, testing isn’t available for patients who have had liver transplants or certain bone marrow transplants.
The VA is working on developing decision-making tools to help clinicians determine when the tests are appropriate, Bates said. It typically takes 2 to 3 weeks to get results, she said, adding that external laboratories provide results. “We eventually would like to bring in all pharmacogenomics testing to be conducted within the VA enterprise.”
Bates reported that she had no disclosures.
The US Department of Veterans Affairs (VA) now has a permanent pharmacogenomics service that provides genetic tests to give clinicians insight into the best medication options for their patients.
The tests, which have no extra cost, are available to all veterans, said pharmacist Jill S. Bates, PharmD, MS, executive director of the VA National Pharmacogenomics Program, who spoke in an interview and a presentation at the annual meeting of the Association of VA Hematology/Oncology.
Genetic testing is “a tool that can help optimize care that we provide for veterans,” she said. “Pharmacogenomics is additional information to help the clinician make a decision. We know that most veterans—greater than 90%—carry a variant in a pharmacogenomics gene that is actionable.”
The genetic tests can provide insight into the optimal medication for multiple conditions such as mental illness, gastrointestinal disorders, cancer, pain, and heart disease. According to a 2019 analysis of over 6 years of data, more than half of the VA patient population used medications whose efficacy may have been affected by detectable genetic variants.
For instance, Bates said tests can let clinicians know whether patients are susceptible to statin-associated muscle adverse effects if they take simvastatin, the cholesterol medication. An estimated 25.6% of the VA population has this variant.
Elsewhere on the cardiac front, an estimated 58.3% of the VA population has a genetic variant that increases sensitivity to the blood thinner warfarin.
Testing could help psychiatrists determine whether certain medications should not be prescribed—or should be prescribed at lower doses—in patients who’ve had adverse reactions to antidepressants, Bates said.
In cancer, Bates said, genetic testing can identify patients who have a genetic variant that boosts toxicity from fluoropyrimidine chemotherapy treatments, which include capecitabine, floxuridine, and fluorouracil. Meanwhile, an estimated 0.9% will have no reaction or limited reaction to capecitabine and fluorouracil, and 4.8% will have hypersensitivity to carbamazepine and oxcarbazepine.
Tests can also identify a genetic variant that can lead to poor metabolism of the chemotherapy drug irinotecan, which is used to treat colon cancer. “In those patients, you’d want to reduce the dose by 20%,” Bates said. In other cases, alternate drugs may be the best strategy to address genetic variations.
Prior to 2019, clinicians had to order pharmacogenomic tests outside of the VA system, according to Bates. That year, a donation from Sanford Health brought VA pharmacogenomics to 40 pilot sites. Since then, more than 88,000 tests have been performed.
The VA has now made its pharmacogenomic program permanent, Bates said. As of early September, testing was available at 139 VA sites and is coming soon to 4 more. It’s not available at another 23 sites that are scattered across the country.
A tool in the VA electronic health record now reminds clinicians about the availability of genetic testing and allows them to order tests. However, testing isn’t available for patients who have had liver transplants or certain bone marrow transplants.
The VA is working on developing decision-making tools to help clinicians determine when the tests are appropriate, Bates said. It typically takes 2 to 3 weeks to get results, she said, adding that external laboratories provide results. “We eventually would like to bring in all pharmacogenomics testing to be conducted within the VA enterprise.”
Bates reported that she had no disclosures.
New Hypertension Approach Hits Multiple Targets at Low Dose
LONDON — , according to experts evaluating the new approach.
This multidrug strategy — in which ultralow-dose triple combinations can be used as a starting treatment and four full-dose combinations can be used to treat resistant hypertension — has shown an impressive ability to lower blood pressure in several new studies.
But will it catch on as a routine treatment recommendation in current practice?
Studies of treatment strategies that involve an ultralow quarter dose of three drugs that lower blood pressure and then escalation to a half-dose triple combination and then to a full-dose triple combination, all given as a single pill, were presented at the European Society of Cardiology (ESC) Congress 2024. Another strategy presented involves a four-drug full-dose combination in patients with resistant hypertension.
Start With Low Doses of Three Drugs
The triple-combination pill contains telmisartan (an angiotensin blocker), amlodipine (a calcium channel blocker), and indapamide (a diuretic). The three medications are used at three doses: Quarter, half, and standard.
“The idea is to start treatment with a little bit of the three main drug classes instead of the full dose of one drug and then to increase the triple-combination doses as required to get to blood pressure goal,” said Anthony Rodgers, PhD, from the team at The George Institute for Global Health, Sydney, Australia, that is developing this triple-combination product.
“Using three different mechanisms right from the beginning covers all the bases and leads to improved blood pressure reduction while just using very small doses of each agent. This represents a completely new approach that could transform the management of hypertension,” he reported.
Single-pill triple-combination antihypertensive formulations exist already, but the component drugs are all at standard doses. Such combinations were designed to improve adherence in patients with hard-to-control blood pressure who need more than two full-dose medications, he explained.
“We are suggesting a completely different concept using much lower doses of the triple combination right from the beginning of treatment,” Dr. Rodgers explained. “Convenience and adherence will be an added advantage, but there’s more to it than that. It’s about combining the different mechanisms of three separate drug classes to get a better antihypertensive effect and being able to do this right from the start of treatment in patients with mildly elevated blood pressure, as well as those with higher levels.”
Proof-of-concept trials of this approach have been conducted, but no commercial low-dose triple-combination product has been available.
The George Institute is now developing such a product — through George Medicines, its commercial arm — with the aim of bringing the triple-combination pill to market in both high- and low-income countries. An approval submission has been filed in the United States.
Dr. Rodgers presented two studies that assessed the triple combination. One showed that the quarter dose reduced blood pressure significantly better than placebo in patients with mildly elevated blood pressure. The second showed that half and standard doses of the three medications were more effective at lowering blood pressure than three dual combinations at the same doses.
The VERONICA Trial
The triple combination was also assessed in the VERONICA study, which showed that among Black adults in Nigeria with uncontrolled hypertension, blood pressure was lower and control was better with the low-dose triple-combination pill than with standard care, and tolerability was good.
In VERONICA, recently published in JAMA, 300 patients with a mean baseline blood pressure of 151/97 mm Hg at home and 156/97 mm Hg in the clinic were randomly assigned to receive the triple-combination pill or standard care.
In the triple-combination group, patients started with the quarter-dose pill, then accelerated, as necessary, to the half-dose and standard-dose pills.
In the standard care group, patients started with amlodipine (5 mg), which was stepped up at monthly intervals so patients could achieve a target blood pressure < 140/90 mm Hg as follows: Amlodipine (5 mg) plus losartan (50 mg); then amlodipine (10 mg) plus losartan (100 mg); then amlodipine (10 mg), losartan (100 mg), plus hydrochlorothiazide (25 mg); and finally referral to a specialist if the target blood pressure was still not achieved.
At month 6, mean home systolic blood pressure was, on average, 31 mm Hg lower in the triple-combination group and 26 mm Hg lower in the standard care group (adjusted difference, −5.8 mm Hg; P < .001).
More patients in the triple-combination group than in the standard care group achieved clinic blood pressure control, defined as blood pressure < 140/90 mm Hg (82% vs 72%), and more patients achieved home blood pressure control, defined as blood pressure below 130/80 mm Hg (62% vs 28%).
No participants discontinued treatment due to adverse events, and adverse events of special interest were reported by just 2% and 3% patients in the triple-combination and standard care groups, respectively.
At month 6, however, more participants in the triple-combination group than in the standard care group had serum potassium levels < 3.5 mmol/L (34% vs 18%), although fewer participants in both the groups had potassium levels < 3.0 mmol/L (10% vs 5%).
Hypokalemia may be the consequence of low dietary potassium intake in Africa, and co-administration with potassium-enriched salt substitution should be evaluated, said Dike Ojji, MBBS, PhD, University of Abuja, Nigeria, who was the lead investigator of VERONICA.
“These findings have broad clinical and public health implications, given that improved hypertension control is a priority in Africa and globally. The results underscore the need for combination therapy to be the cornerstone of effective treatment regimens,” Dr. Ojji said.
Missed Targets
“It has taken a long time for the penny to drop as to why the existing antihypertensive treatment paradigm does not work so well,” Dr. Rodgers pointed out. “What tends to happen in clinical practice is that people start on one drug and blood pressure falls a bit, then no further action is taken. But this is not usually enough to get to target. With our approach of using three drugs at low doses straight away, we can often get the blood pressure controlled to target much more quickly with one tablet.”
Low doses of the triple-combination pill should also have a favorable adverse-effect profile and fewer drug interactions, as these issues are generally seen much more frequently with higher doses of drugs, he explained.
This low-dose triple-combination approach could help manage the current epidemic of hypertension and cardiometabolic disease, said Pam Taub, MD, director of preventive cardiology at UC San Diego Health System.
“We are in a new era of cardiometabolic disease, and one of the fundamental drivers of atherosclerotic cardiovascular disease is hypertension, which is prevalent in patients with diabetes, in those with obesity, and is a contributor to chronic kidney disease,” she said.
“We really need to be addressing hypertension very early to prevent this end-organ damage, but because hypertension tends to occur alongside multiple other comorbidities, patients are often on many different medications and are overwhelmed by the burden of polypharmacy.”
Dr. Taub described this triple-combination approach as “looking at hypertension treatment through a new lens.”
“We’ve always been taught to maximize the dose of one agent before we go to a new agent,” she said. “These studies are fundamentally challenging that paradigm. From a pathophysiological and mechanistic perspective, we are seeing that lower doses of different medications can really harness some unique synergistic mechanisms, which can be beneficial for patients.”
But not all experts are convinced that this approach will be a popular option in all countries.
Although this approach makes sense, in that the different agents work synergistically to give a better antihypertensive effect, many physicians could be uncomfortable with the idea of giving multiple medications straight off as the first step of treatment, said Eugene Yang, MD, from the University of Washington in Seattle.
If the patient develops a side effect, it will not be clear which medication is causing it, making it difficult to know which one to stop, he pointed out.
“These studies confirm that a low-dose multidrug-combination pill is effective at lowering blood pressure, but we already have previous studies showing this,” he added. “The issue is how we translate this into patient care. It would be great if we could get people to use it, but I think concerns from both clinicians and patients about identifying the source of any side effects may be a stumbling block.”
The approach is more likely to be adopted in low- to middle-income countries, where there is limited access to healthcare and where the population-wide control of blood pressure makes sense, said Dr. Yang.
Most current guidelines now recommend initiating therapy with two agents, ideally, as a single-pill combination product. “We have finally acknowledged that the vast majority of patients need two drugs. That’s a good starting point. This low-dose triple combination could be an interesting new approach,” said Neil Poulter, MD, professor of preventive cardiovascular medicine at Imperial College London, England.
This approach is in line with the idea that single-pill combinations are the way forward for hypertension therapy, he added.
“The triple combination is attractive, in that you are never quite sure which particular mechanism is driving an individual’s elevated blood pressure, so if you can target three different mechanisms at the same time, you’ve got more chance of a good hit,” Dr. Poulter said.
“The VERONICA trial showed a very good result on lowering BP using this low-dose triple combination as a starting point and increasing quickly to single-pill combinations of triple half doses, then triple full doses, as required. But I think we need more evidence on how this compares to current practice than just this one study in Africa to make this an acceptable routine approach on a global level,” he said.
QUADRO: Four-Drug Combo in Resistant Hypertension
Another scenario in which single-pill antihypertensive combinations could be particularly useful is at the other end of the spectrum: The treatment of patients with resistant hypertension.
The QUADRO study showed that a single pill containing perindopril, indapamide, amlodipine, and bisoprolol is better at lowering blood pressure than the triple combination of perindopril, indapamide, and amlodipine.
The primary endpoint — office sitting systolic blood pressure at 16 weeks — was 8 mm Hg lower with the quadruple combination than with the triple combination. And mean ambulatory 24-hour systolic blood pressure was 7.5 mm Hg lower with the four-drug combination.
This was the first study of a single-pill quadruple combination in patients with resistant hypertension, which is a “difficult-to-treat condition demanding a high number of pills with not enough safe and practical options,” said Stefano Taddei, MD, from the University of Pisa, Italy, when he presented the study at the ESC meeting.
Using “four well-established drugs in a single-combination pill may improve adherence and should be an innovative solution for resistant and difficult-to-treat hypertensive patients,” he said.
Nonadherence is a big problem in patients with resistant hypertension. “It is really difficult to get patients to take three or four antihypertensive agents along with all the other medications they have for other comorbidities,” Dr. Taub pointed out. “We really need to think about combination formulations that reduce the pill burden for our patients.”
Around 10% patients with hypertension may require a fourth drug, so a four-drug single-pill combination therefore makes good sense, said Dr. Poulter.
But the choice of the fourth drug is the subject of debate. The PATHWAY trial showed spironolactone to be the most effective fourth agent, but it can cause side effects, such as gynecomastia and hyperkalemia.
“The beta-blocker in the four-drug combination product used in the QUADRO study may not be as effective as spironolactone at lowering blood pressure,” Dr. Poulter explained, noting that beta-blockers have known side effects. However, “they are often already recommended for patients with very common comorbidities, such as arrhythmias, history of MI, heart failure, angina. In that regard, it makes sense to have a beta-blocker in there.”
The four-drug combination used in the QUADRO study led to a bigger reduction — by 8 mm Hg — than the three-drug combination. “That’s pretty good. I thought this was a very useful and interesting study,” he said.
There could be a role for a four-drug combination product in resistant hypertension. “Whatever we can do to improve adherence and reduce blood pressure is good thing,” said Dr. Yang.
However, a mineralocorticoid receptor antagonist (such as spironolactone) might be better as the fourth drug; that is what is recommended in the resistant hypertension algorithm.
Lower Blood Pressure, Better Outcomes
“What we are seeing in these trials is that across a wide spectrum of patients with hypertension or resistant hypertension, combination pills are superior to standard practice for BP lowering, and that will lead to improved outcomes,” said Dr. Taub.
“For years, such single-pill combinations have been viewed as ‘bad medicine’ in hypertension,” Dr. Poulter added. “That is clearly not the case, as these studies are showing. And single-pill combination therapies are used extensively in practically every other area of medicine. We are starting to accept them now in the blood pressure community, and I think the use of triple and quadruple combinations, as in these studies, has a real logic to it. But for this approach to be useful, these single-pill combinations must be made available, cheaply, across the world, especially in low- and middle-income countries where hypertension rates are a particular problem.”
A version of this article appeared on Medscape.com.
LONDON — , according to experts evaluating the new approach.
This multidrug strategy — in which ultralow-dose triple combinations can be used as a starting treatment and four full-dose combinations can be used to treat resistant hypertension — has shown an impressive ability to lower blood pressure in several new studies.
But will it catch on as a routine treatment recommendation in current practice?
Studies of treatment strategies that involve an ultralow quarter dose of three drugs that lower blood pressure and then escalation to a half-dose triple combination and then to a full-dose triple combination, all given as a single pill, were presented at the European Society of Cardiology (ESC) Congress 2024. Another strategy presented involves a four-drug full-dose combination in patients with resistant hypertension.
Start With Low Doses of Three Drugs
The triple-combination pill contains telmisartan (an angiotensin blocker), amlodipine (a calcium channel blocker), and indapamide (a diuretic). The three medications are used at three doses: Quarter, half, and standard.
“The idea is to start treatment with a little bit of the three main drug classes instead of the full dose of one drug and then to increase the triple-combination doses as required to get to blood pressure goal,” said Anthony Rodgers, PhD, from the team at The George Institute for Global Health, Sydney, Australia, that is developing this triple-combination product.
“Using three different mechanisms right from the beginning covers all the bases and leads to improved blood pressure reduction while just using very small doses of each agent. This represents a completely new approach that could transform the management of hypertension,” he reported.
Single-pill triple-combination antihypertensive formulations exist already, but the component drugs are all at standard doses. Such combinations were designed to improve adherence in patients with hard-to-control blood pressure who need more than two full-dose medications, he explained.
“We are suggesting a completely different concept using much lower doses of the triple combination right from the beginning of treatment,” Dr. Rodgers explained. “Convenience and adherence will be an added advantage, but there’s more to it than that. It’s about combining the different mechanisms of three separate drug classes to get a better antihypertensive effect and being able to do this right from the start of treatment in patients with mildly elevated blood pressure, as well as those with higher levels.”
Proof-of-concept trials of this approach have been conducted, but no commercial low-dose triple-combination product has been available.
The George Institute is now developing such a product — through George Medicines, its commercial arm — with the aim of bringing the triple-combination pill to market in both high- and low-income countries. An approval submission has been filed in the United States.
Dr. Rodgers presented two studies that assessed the triple combination. One showed that the quarter dose reduced blood pressure significantly better than placebo in patients with mildly elevated blood pressure. The second showed that half and standard doses of the three medications were more effective at lowering blood pressure than three dual combinations at the same doses.
The VERONICA Trial
The triple combination was also assessed in the VERONICA study, which showed that among Black adults in Nigeria with uncontrolled hypertension, blood pressure was lower and control was better with the low-dose triple-combination pill than with standard care, and tolerability was good.
In VERONICA, recently published in JAMA, 300 patients with a mean baseline blood pressure of 151/97 mm Hg at home and 156/97 mm Hg in the clinic were randomly assigned to receive the triple-combination pill or standard care.
In the triple-combination group, patients started with the quarter-dose pill, then accelerated, as necessary, to the half-dose and standard-dose pills.
In the standard care group, patients started with amlodipine (5 mg), which was stepped up at monthly intervals so patients could achieve a target blood pressure < 140/90 mm Hg as follows: Amlodipine (5 mg) plus losartan (50 mg); then amlodipine (10 mg) plus losartan (100 mg); then amlodipine (10 mg), losartan (100 mg), plus hydrochlorothiazide (25 mg); and finally referral to a specialist if the target blood pressure was still not achieved.
At month 6, mean home systolic blood pressure was, on average, 31 mm Hg lower in the triple-combination group and 26 mm Hg lower in the standard care group (adjusted difference, −5.8 mm Hg; P < .001).
More patients in the triple-combination group than in the standard care group achieved clinic blood pressure control, defined as blood pressure < 140/90 mm Hg (82% vs 72%), and more patients achieved home blood pressure control, defined as blood pressure below 130/80 mm Hg (62% vs 28%).
No participants discontinued treatment due to adverse events, and adverse events of special interest were reported by just 2% and 3% patients in the triple-combination and standard care groups, respectively.
At month 6, however, more participants in the triple-combination group than in the standard care group had serum potassium levels < 3.5 mmol/L (34% vs 18%), although fewer participants in both the groups had potassium levels < 3.0 mmol/L (10% vs 5%).
Hypokalemia may be the consequence of low dietary potassium intake in Africa, and co-administration with potassium-enriched salt substitution should be evaluated, said Dike Ojji, MBBS, PhD, University of Abuja, Nigeria, who was the lead investigator of VERONICA.
“These findings have broad clinical and public health implications, given that improved hypertension control is a priority in Africa and globally. The results underscore the need for combination therapy to be the cornerstone of effective treatment regimens,” Dr. Ojji said.
Missed Targets
“It has taken a long time for the penny to drop as to why the existing antihypertensive treatment paradigm does not work so well,” Dr. Rodgers pointed out. “What tends to happen in clinical practice is that people start on one drug and blood pressure falls a bit, then no further action is taken. But this is not usually enough to get to target. With our approach of using three drugs at low doses straight away, we can often get the blood pressure controlled to target much more quickly with one tablet.”
Low doses of the triple-combination pill should also have a favorable adverse-effect profile and fewer drug interactions, as these issues are generally seen much more frequently with higher doses of drugs, he explained.
This low-dose triple-combination approach could help manage the current epidemic of hypertension and cardiometabolic disease, said Pam Taub, MD, director of preventive cardiology at UC San Diego Health System.
“We are in a new era of cardiometabolic disease, and one of the fundamental drivers of atherosclerotic cardiovascular disease is hypertension, which is prevalent in patients with diabetes, in those with obesity, and is a contributor to chronic kidney disease,” she said.
“We really need to be addressing hypertension very early to prevent this end-organ damage, but because hypertension tends to occur alongside multiple other comorbidities, patients are often on many different medications and are overwhelmed by the burden of polypharmacy.”
Dr. Taub described this triple-combination approach as “looking at hypertension treatment through a new lens.”
“We’ve always been taught to maximize the dose of one agent before we go to a new agent,” she said. “These studies are fundamentally challenging that paradigm. From a pathophysiological and mechanistic perspective, we are seeing that lower doses of different medications can really harness some unique synergistic mechanisms, which can be beneficial for patients.”
But not all experts are convinced that this approach will be a popular option in all countries.
Although this approach makes sense, in that the different agents work synergistically to give a better antihypertensive effect, many physicians could be uncomfortable with the idea of giving multiple medications straight off as the first step of treatment, said Eugene Yang, MD, from the University of Washington in Seattle.
If the patient develops a side effect, it will not be clear which medication is causing it, making it difficult to know which one to stop, he pointed out.
“These studies confirm that a low-dose multidrug-combination pill is effective at lowering blood pressure, but we already have previous studies showing this,” he added. “The issue is how we translate this into patient care. It would be great if we could get people to use it, but I think concerns from both clinicians and patients about identifying the source of any side effects may be a stumbling block.”
The approach is more likely to be adopted in low- to middle-income countries, where there is limited access to healthcare and where the population-wide control of blood pressure makes sense, said Dr. Yang.
Most current guidelines now recommend initiating therapy with two agents, ideally, as a single-pill combination product. “We have finally acknowledged that the vast majority of patients need two drugs. That’s a good starting point. This low-dose triple combination could be an interesting new approach,” said Neil Poulter, MD, professor of preventive cardiovascular medicine at Imperial College London, England.
This approach is in line with the idea that single-pill combinations are the way forward for hypertension therapy, he added.
“The triple combination is attractive, in that you are never quite sure which particular mechanism is driving an individual’s elevated blood pressure, so if you can target three different mechanisms at the same time, you’ve got more chance of a good hit,” Dr. Poulter said.
“The VERONICA trial showed a very good result on lowering BP using this low-dose triple combination as a starting point and increasing quickly to single-pill combinations of triple half doses, then triple full doses, as required. But I think we need more evidence on how this compares to current practice than just this one study in Africa to make this an acceptable routine approach on a global level,” he said.
QUADRO: Four-Drug Combo in Resistant Hypertension
Another scenario in which single-pill antihypertensive combinations could be particularly useful is at the other end of the spectrum: The treatment of patients with resistant hypertension.
The QUADRO study showed that a single pill containing perindopril, indapamide, amlodipine, and bisoprolol is better at lowering blood pressure than the triple combination of perindopril, indapamide, and amlodipine.
The primary endpoint — office sitting systolic blood pressure at 16 weeks — was 8 mm Hg lower with the quadruple combination than with the triple combination. And mean ambulatory 24-hour systolic blood pressure was 7.5 mm Hg lower with the four-drug combination.
This was the first study of a single-pill quadruple combination in patients with resistant hypertension, which is a “difficult-to-treat condition demanding a high number of pills with not enough safe and practical options,” said Stefano Taddei, MD, from the University of Pisa, Italy, when he presented the study at the ESC meeting.
Using “four well-established drugs in a single-combination pill may improve adherence and should be an innovative solution for resistant and difficult-to-treat hypertensive patients,” he said.
Nonadherence is a big problem in patients with resistant hypertension. “It is really difficult to get patients to take three or four antihypertensive agents along with all the other medications they have for other comorbidities,” Dr. Taub pointed out. “We really need to think about combination formulations that reduce the pill burden for our patients.”
Around 10% patients with hypertension may require a fourth drug, so a four-drug single-pill combination therefore makes good sense, said Dr. Poulter.
But the choice of the fourth drug is the subject of debate. The PATHWAY trial showed spironolactone to be the most effective fourth agent, but it can cause side effects, such as gynecomastia and hyperkalemia.
“The beta-blocker in the four-drug combination product used in the QUADRO study may not be as effective as spironolactone at lowering blood pressure,” Dr. Poulter explained, noting that beta-blockers have known side effects. However, “they are often already recommended for patients with very common comorbidities, such as arrhythmias, history of MI, heart failure, angina. In that regard, it makes sense to have a beta-blocker in there.”
The four-drug combination used in the QUADRO study led to a bigger reduction — by 8 mm Hg — than the three-drug combination. “That’s pretty good. I thought this was a very useful and interesting study,” he said.
There could be a role for a four-drug combination product in resistant hypertension. “Whatever we can do to improve adherence and reduce blood pressure is good thing,” said Dr. Yang.
However, a mineralocorticoid receptor antagonist (such as spironolactone) might be better as the fourth drug; that is what is recommended in the resistant hypertension algorithm.
Lower Blood Pressure, Better Outcomes
“What we are seeing in these trials is that across a wide spectrum of patients with hypertension or resistant hypertension, combination pills are superior to standard practice for BP lowering, and that will lead to improved outcomes,” said Dr. Taub.
“For years, such single-pill combinations have been viewed as ‘bad medicine’ in hypertension,” Dr. Poulter added. “That is clearly not the case, as these studies are showing. And single-pill combination therapies are used extensively in practically every other area of medicine. We are starting to accept them now in the blood pressure community, and I think the use of triple and quadruple combinations, as in these studies, has a real logic to it. But for this approach to be useful, these single-pill combinations must be made available, cheaply, across the world, especially in low- and middle-income countries where hypertension rates are a particular problem.”
A version of this article appeared on Medscape.com.
LONDON — , according to experts evaluating the new approach.
This multidrug strategy — in which ultralow-dose triple combinations can be used as a starting treatment and four full-dose combinations can be used to treat resistant hypertension — has shown an impressive ability to lower blood pressure in several new studies.
But will it catch on as a routine treatment recommendation in current practice?
Studies of treatment strategies that involve an ultralow quarter dose of three drugs that lower blood pressure and then escalation to a half-dose triple combination and then to a full-dose triple combination, all given as a single pill, were presented at the European Society of Cardiology (ESC) Congress 2024. Another strategy presented involves a four-drug full-dose combination in patients with resistant hypertension.
Start With Low Doses of Three Drugs
The triple-combination pill contains telmisartan (an angiotensin blocker), amlodipine (a calcium channel blocker), and indapamide (a diuretic). The three medications are used at three doses: Quarter, half, and standard.
“The idea is to start treatment with a little bit of the three main drug classes instead of the full dose of one drug and then to increase the triple-combination doses as required to get to blood pressure goal,” said Anthony Rodgers, PhD, from the team at The George Institute for Global Health, Sydney, Australia, that is developing this triple-combination product.
“Using three different mechanisms right from the beginning covers all the bases and leads to improved blood pressure reduction while just using very small doses of each agent. This represents a completely new approach that could transform the management of hypertension,” he reported.
Single-pill triple-combination antihypertensive formulations exist already, but the component drugs are all at standard doses. Such combinations were designed to improve adherence in patients with hard-to-control blood pressure who need more than two full-dose medications, he explained.
“We are suggesting a completely different concept using much lower doses of the triple combination right from the beginning of treatment,” Dr. Rodgers explained. “Convenience and adherence will be an added advantage, but there’s more to it than that. It’s about combining the different mechanisms of three separate drug classes to get a better antihypertensive effect and being able to do this right from the start of treatment in patients with mildly elevated blood pressure, as well as those with higher levels.”
Proof-of-concept trials of this approach have been conducted, but no commercial low-dose triple-combination product has been available.
The George Institute is now developing such a product — through George Medicines, its commercial arm — with the aim of bringing the triple-combination pill to market in both high- and low-income countries. An approval submission has been filed in the United States.
Dr. Rodgers presented two studies that assessed the triple combination. One showed that the quarter dose reduced blood pressure significantly better than placebo in patients with mildly elevated blood pressure. The second showed that half and standard doses of the three medications were more effective at lowering blood pressure than three dual combinations at the same doses.
The VERONICA Trial
The triple combination was also assessed in the VERONICA study, which showed that among Black adults in Nigeria with uncontrolled hypertension, blood pressure was lower and control was better with the low-dose triple-combination pill than with standard care, and tolerability was good.
In VERONICA, recently published in JAMA, 300 patients with a mean baseline blood pressure of 151/97 mm Hg at home and 156/97 mm Hg in the clinic were randomly assigned to receive the triple-combination pill or standard care.
In the triple-combination group, patients started with the quarter-dose pill, then accelerated, as necessary, to the half-dose and standard-dose pills.
In the standard care group, patients started with amlodipine (5 mg), which was stepped up at monthly intervals so patients could achieve a target blood pressure < 140/90 mm Hg as follows: Amlodipine (5 mg) plus losartan (50 mg); then amlodipine (10 mg) plus losartan (100 mg); then amlodipine (10 mg), losartan (100 mg), plus hydrochlorothiazide (25 mg); and finally referral to a specialist if the target blood pressure was still not achieved.
At month 6, mean home systolic blood pressure was, on average, 31 mm Hg lower in the triple-combination group and 26 mm Hg lower in the standard care group (adjusted difference, −5.8 mm Hg; P < .001).
More patients in the triple-combination group than in the standard care group achieved clinic blood pressure control, defined as blood pressure < 140/90 mm Hg (82% vs 72%), and more patients achieved home blood pressure control, defined as blood pressure below 130/80 mm Hg (62% vs 28%).
No participants discontinued treatment due to adverse events, and adverse events of special interest were reported by just 2% and 3% patients in the triple-combination and standard care groups, respectively.
At month 6, however, more participants in the triple-combination group than in the standard care group had serum potassium levels < 3.5 mmol/L (34% vs 18%), although fewer participants in both the groups had potassium levels < 3.0 mmol/L (10% vs 5%).
Hypokalemia may be the consequence of low dietary potassium intake in Africa, and co-administration with potassium-enriched salt substitution should be evaluated, said Dike Ojji, MBBS, PhD, University of Abuja, Nigeria, who was the lead investigator of VERONICA.
“These findings have broad clinical and public health implications, given that improved hypertension control is a priority in Africa and globally. The results underscore the need for combination therapy to be the cornerstone of effective treatment regimens,” Dr. Ojji said.
Missed Targets
“It has taken a long time for the penny to drop as to why the existing antihypertensive treatment paradigm does not work so well,” Dr. Rodgers pointed out. “What tends to happen in clinical practice is that people start on one drug and blood pressure falls a bit, then no further action is taken. But this is not usually enough to get to target. With our approach of using three drugs at low doses straight away, we can often get the blood pressure controlled to target much more quickly with one tablet.”
Low doses of the triple-combination pill should also have a favorable adverse-effect profile and fewer drug interactions, as these issues are generally seen much more frequently with higher doses of drugs, he explained.
This low-dose triple-combination approach could help manage the current epidemic of hypertension and cardiometabolic disease, said Pam Taub, MD, director of preventive cardiology at UC San Diego Health System.
“We are in a new era of cardiometabolic disease, and one of the fundamental drivers of atherosclerotic cardiovascular disease is hypertension, which is prevalent in patients with diabetes, in those with obesity, and is a contributor to chronic kidney disease,” she said.
“We really need to be addressing hypertension very early to prevent this end-organ damage, but because hypertension tends to occur alongside multiple other comorbidities, patients are often on many different medications and are overwhelmed by the burden of polypharmacy.”
Dr. Taub described this triple-combination approach as “looking at hypertension treatment through a new lens.”
“We’ve always been taught to maximize the dose of one agent before we go to a new agent,” she said. “These studies are fundamentally challenging that paradigm. From a pathophysiological and mechanistic perspective, we are seeing that lower doses of different medications can really harness some unique synergistic mechanisms, which can be beneficial for patients.”
But not all experts are convinced that this approach will be a popular option in all countries.
Although this approach makes sense, in that the different agents work synergistically to give a better antihypertensive effect, many physicians could be uncomfortable with the idea of giving multiple medications straight off as the first step of treatment, said Eugene Yang, MD, from the University of Washington in Seattle.
If the patient develops a side effect, it will not be clear which medication is causing it, making it difficult to know which one to stop, he pointed out.
“These studies confirm that a low-dose multidrug-combination pill is effective at lowering blood pressure, but we already have previous studies showing this,” he added. “The issue is how we translate this into patient care. It would be great if we could get people to use it, but I think concerns from both clinicians and patients about identifying the source of any side effects may be a stumbling block.”
The approach is more likely to be adopted in low- to middle-income countries, where there is limited access to healthcare and where the population-wide control of blood pressure makes sense, said Dr. Yang.
Most current guidelines now recommend initiating therapy with two agents, ideally, as a single-pill combination product. “We have finally acknowledged that the vast majority of patients need two drugs. That’s a good starting point. This low-dose triple combination could be an interesting new approach,” said Neil Poulter, MD, professor of preventive cardiovascular medicine at Imperial College London, England.
This approach is in line with the idea that single-pill combinations are the way forward for hypertension therapy, he added.
“The triple combination is attractive, in that you are never quite sure which particular mechanism is driving an individual’s elevated blood pressure, so if you can target three different mechanisms at the same time, you’ve got more chance of a good hit,” Dr. Poulter said.
“The VERONICA trial showed a very good result on lowering BP using this low-dose triple combination as a starting point and increasing quickly to single-pill combinations of triple half doses, then triple full doses, as required. But I think we need more evidence on how this compares to current practice than just this one study in Africa to make this an acceptable routine approach on a global level,” he said.
QUADRO: Four-Drug Combo in Resistant Hypertension
Another scenario in which single-pill antihypertensive combinations could be particularly useful is at the other end of the spectrum: The treatment of patients with resistant hypertension.
The QUADRO study showed that a single pill containing perindopril, indapamide, amlodipine, and bisoprolol is better at lowering blood pressure than the triple combination of perindopril, indapamide, and amlodipine.
The primary endpoint — office sitting systolic blood pressure at 16 weeks — was 8 mm Hg lower with the quadruple combination than with the triple combination. And mean ambulatory 24-hour systolic blood pressure was 7.5 mm Hg lower with the four-drug combination.
This was the first study of a single-pill quadruple combination in patients with resistant hypertension, which is a “difficult-to-treat condition demanding a high number of pills with not enough safe and practical options,” said Stefano Taddei, MD, from the University of Pisa, Italy, when he presented the study at the ESC meeting.
Using “four well-established drugs in a single-combination pill may improve adherence and should be an innovative solution for resistant and difficult-to-treat hypertensive patients,” he said.
Nonadherence is a big problem in patients with resistant hypertension. “It is really difficult to get patients to take three or four antihypertensive agents along with all the other medications they have for other comorbidities,” Dr. Taub pointed out. “We really need to think about combination formulations that reduce the pill burden for our patients.”
Around 10% patients with hypertension may require a fourth drug, so a four-drug single-pill combination therefore makes good sense, said Dr. Poulter.
But the choice of the fourth drug is the subject of debate. The PATHWAY trial showed spironolactone to be the most effective fourth agent, but it can cause side effects, such as gynecomastia and hyperkalemia.
“The beta-blocker in the four-drug combination product used in the QUADRO study may not be as effective as spironolactone at lowering blood pressure,” Dr. Poulter explained, noting that beta-blockers have known side effects. However, “they are often already recommended for patients with very common comorbidities, such as arrhythmias, history of MI, heart failure, angina. In that regard, it makes sense to have a beta-blocker in there.”
The four-drug combination used in the QUADRO study led to a bigger reduction — by 8 mm Hg — than the three-drug combination. “That’s pretty good. I thought this was a very useful and interesting study,” he said.
There could be a role for a four-drug combination product in resistant hypertension. “Whatever we can do to improve adherence and reduce blood pressure is good thing,” said Dr. Yang.
However, a mineralocorticoid receptor antagonist (such as spironolactone) might be better as the fourth drug; that is what is recommended in the resistant hypertension algorithm.
Lower Blood Pressure, Better Outcomes
“What we are seeing in these trials is that across a wide spectrum of patients with hypertension or resistant hypertension, combination pills are superior to standard practice for BP lowering, and that will lead to improved outcomes,” said Dr. Taub.
“For years, such single-pill combinations have been viewed as ‘bad medicine’ in hypertension,” Dr. Poulter added. “That is clearly not the case, as these studies are showing. And single-pill combination therapies are used extensively in practically every other area of medicine. We are starting to accept them now in the blood pressure community, and I think the use of triple and quadruple combinations, as in these studies, has a real logic to it. But for this approach to be useful, these single-pill combinations must be made available, cheaply, across the world, especially in low- and middle-income countries where hypertension rates are a particular problem.”
A version of this article appeared on Medscape.com.
FROM ESC CONGRESS 2024
Modified Sleep Apnea Index Score Associated With CV Risk
Use of a modified sleep apnea index can identify cardiovascular risk factors in adults with moderate to severe obstructive sleep apnea (OSA), according to results from a new study presented at the American Academy of Otolaryngology–Head and Neck Surgery 2024 Annual Meeting.
, said Jennifer A. Goldfarb, MHS, a medical student at Thomas Jefferson University, Philadelphia, Pennsylvania, who presented the findings.
OSA has an association with many negative cardiovascular comorbidities; however, “the AHI provides only a single metric and does not provide a holistic assessment of the individual patient’s disease severity,” said senior author Colin T. Huntley, MD, also of Thomas Jefferson University.
“OSA is very complex, and having a robust system to assess the disease may be a better predictor of overall severity,” he told this news organization.
Previous research has shown a correlation between mSASI and mean arterial pressure and serum C-reactive protein in OSA patients, but the connection with cardiovascular risk factors has not been well studied, Ms. Goldfarb noted.
In the retrospective cohort study, Ms. Goldfarb and colleagues looked at mSASI scores from 260 CPAP-intolerant patients with OSA who underwent upper airway stimulation, maxillomandibular advancement, or expansion sphincter pharyngoplasty at a single sleep surgery clinic between 2014 and 2021. The mSASI uses a score of 1-3, with 3 as the highest level of OSA severity.
Cardiovascular risk factors were assessed at the patient’s initial evaluation by the sleep surgery team. They included coronary artery disease, type 2 diabetes, atrial fibrillation, congestive heart failure, hypertension, and cerebrovascular accident.
A total of 142 patients (55%) had an mSASI of 1; 91 (35%) had an mSASI of 2; and 27 (10%) had an mSASI of 3. At least one cardiovascular risk factor was present in 58%, 68%, and 63% of these groups, respectively (P = .3).
Stratifying participants by mSASI scores, the researchers found that patients with an mSASI of 2 or 3 were significantly more likely than those with an mSASI of 1 to have more cardiovascular risk factors on initial presentation, and were significantly more likely to be diagnosed with hypertension (P = .02 for both).
Using the AHI, however, patients with moderate to severe OSA (AHI > 15) had a similar number of cardiovascular risk factors as those with mild OSA (P > .05).
“A higher mSASI score, which represents worse disease, was associated with a higher Framingham risk score, which supported our hypothesis; however, the AHI was not found to be associated with an increased Framingham score,” Dr. Huntley told this news organization.
Takeaways and Next Steps
These results suggest that the AHI, while a good metric, might not be the best tool for assessment of overall disease severity, given the complexity of OSA, the impact of the disease on patient quality of life, and the risk for downstream cardiovascular disease, said Dr. Huntley.
The findings were limited by the retrospective design and use of data from a single center.
Population-level data are needed to identify variables that may be meaningful to create a future tool that provides the best picture of the individual patient’s disease, he added. Additional prospective data are also needed to assess the impact of the scoring system on long-term treatment outcomes.
“The current study is especially interesting as we are just beginning to understand the factors that predict cardiovascular risk for patients with obstructive sleep apnea,” Megan Durr, MD, of the University of California, San Francisco, said in an interview.
“For a long time, we primarily looked at the AHI and/or oxygen levels during sleep as risk factors, and we haven’t looked as much at other factors.” said Dr. Durr, who served as a moderator for the session in which the study was presented.
The current findings provide a more comprehensive look at cardiovascular risk; the inclusion of patient anatomy and symptoms add to the knowledge of this topic, and will lead to further work in this area, she added.
The study received no outside funding. The researchers had no financial conflicts to disclose. Dr. Huntley disclosed receiving research support from Nyxoah and Inspire, and serving as a consultant for Nyxoah, Inspire, and Avivomed.
Dr. Durr had no financial conflicts to disclose.
A version of this article appeared on Medscape.com.
Use of a modified sleep apnea index can identify cardiovascular risk factors in adults with moderate to severe obstructive sleep apnea (OSA), according to results from a new study presented at the American Academy of Otolaryngology–Head and Neck Surgery 2024 Annual Meeting.
, said Jennifer A. Goldfarb, MHS, a medical student at Thomas Jefferson University, Philadelphia, Pennsylvania, who presented the findings.
OSA has an association with many negative cardiovascular comorbidities; however, “the AHI provides only a single metric and does not provide a holistic assessment of the individual patient’s disease severity,” said senior author Colin T. Huntley, MD, also of Thomas Jefferson University.
“OSA is very complex, and having a robust system to assess the disease may be a better predictor of overall severity,” he told this news organization.
Previous research has shown a correlation between mSASI and mean arterial pressure and serum C-reactive protein in OSA patients, but the connection with cardiovascular risk factors has not been well studied, Ms. Goldfarb noted.
In the retrospective cohort study, Ms. Goldfarb and colleagues looked at mSASI scores from 260 CPAP-intolerant patients with OSA who underwent upper airway stimulation, maxillomandibular advancement, or expansion sphincter pharyngoplasty at a single sleep surgery clinic between 2014 and 2021. The mSASI uses a score of 1-3, with 3 as the highest level of OSA severity.
Cardiovascular risk factors were assessed at the patient’s initial evaluation by the sleep surgery team. They included coronary artery disease, type 2 diabetes, atrial fibrillation, congestive heart failure, hypertension, and cerebrovascular accident.
A total of 142 patients (55%) had an mSASI of 1; 91 (35%) had an mSASI of 2; and 27 (10%) had an mSASI of 3. At least one cardiovascular risk factor was present in 58%, 68%, and 63% of these groups, respectively (P = .3).
Stratifying participants by mSASI scores, the researchers found that patients with an mSASI of 2 or 3 were significantly more likely than those with an mSASI of 1 to have more cardiovascular risk factors on initial presentation, and were significantly more likely to be diagnosed with hypertension (P = .02 for both).
Using the AHI, however, patients with moderate to severe OSA (AHI > 15) had a similar number of cardiovascular risk factors as those with mild OSA (P > .05).
“A higher mSASI score, which represents worse disease, was associated with a higher Framingham risk score, which supported our hypothesis; however, the AHI was not found to be associated with an increased Framingham score,” Dr. Huntley told this news organization.
Takeaways and Next Steps
These results suggest that the AHI, while a good metric, might not be the best tool for assessment of overall disease severity, given the complexity of OSA, the impact of the disease on patient quality of life, and the risk for downstream cardiovascular disease, said Dr. Huntley.
The findings were limited by the retrospective design and use of data from a single center.
Population-level data are needed to identify variables that may be meaningful to create a future tool that provides the best picture of the individual patient’s disease, he added. Additional prospective data are also needed to assess the impact of the scoring system on long-term treatment outcomes.
“The current study is especially interesting as we are just beginning to understand the factors that predict cardiovascular risk for patients with obstructive sleep apnea,” Megan Durr, MD, of the University of California, San Francisco, said in an interview.
“For a long time, we primarily looked at the AHI and/or oxygen levels during sleep as risk factors, and we haven’t looked as much at other factors.” said Dr. Durr, who served as a moderator for the session in which the study was presented.
The current findings provide a more comprehensive look at cardiovascular risk; the inclusion of patient anatomy and symptoms add to the knowledge of this topic, and will lead to further work in this area, she added.
The study received no outside funding. The researchers had no financial conflicts to disclose. Dr. Huntley disclosed receiving research support from Nyxoah and Inspire, and serving as a consultant for Nyxoah, Inspire, and Avivomed.
Dr. Durr had no financial conflicts to disclose.
A version of this article appeared on Medscape.com.
Use of a modified sleep apnea index can identify cardiovascular risk factors in adults with moderate to severe obstructive sleep apnea (OSA), according to results from a new study presented at the American Academy of Otolaryngology–Head and Neck Surgery 2024 Annual Meeting.
, said Jennifer A. Goldfarb, MHS, a medical student at Thomas Jefferson University, Philadelphia, Pennsylvania, who presented the findings.
OSA has an association with many negative cardiovascular comorbidities; however, “the AHI provides only a single metric and does not provide a holistic assessment of the individual patient’s disease severity,” said senior author Colin T. Huntley, MD, also of Thomas Jefferson University.
“OSA is very complex, and having a robust system to assess the disease may be a better predictor of overall severity,” he told this news organization.
Previous research has shown a correlation between mSASI and mean arterial pressure and serum C-reactive protein in OSA patients, but the connection with cardiovascular risk factors has not been well studied, Ms. Goldfarb noted.
In the retrospective cohort study, Ms. Goldfarb and colleagues looked at mSASI scores from 260 CPAP-intolerant patients with OSA who underwent upper airway stimulation, maxillomandibular advancement, or expansion sphincter pharyngoplasty at a single sleep surgery clinic between 2014 and 2021. The mSASI uses a score of 1-3, with 3 as the highest level of OSA severity.
Cardiovascular risk factors were assessed at the patient’s initial evaluation by the sleep surgery team. They included coronary artery disease, type 2 diabetes, atrial fibrillation, congestive heart failure, hypertension, and cerebrovascular accident.
A total of 142 patients (55%) had an mSASI of 1; 91 (35%) had an mSASI of 2; and 27 (10%) had an mSASI of 3. At least one cardiovascular risk factor was present in 58%, 68%, and 63% of these groups, respectively (P = .3).
Stratifying participants by mSASI scores, the researchers found that patients with an mSASI of 2 or 3 were significantly more likely than those with an mSASI of 1 to have more cardiovascular risk factors on initial presentation, and were significantly more likely to be diagnosed with hypertension (P = .02 for both).
Using the AHI, however, patients with moderate to severe OSA (AHI > 15) had a similar number of cardiovascular risk factors as those with mild OSA (P > .05).
“A higher mSASI score, which represents worse disease, was associated with a higher Framingham risk score, which supported our hypothesis; however, the AHI was not found to be associated with an increased Framingham score,” Dr. Huntley told this news organization.
Takeaways and Next Steps
These results suggest that the AHI, while a good metric, might not be the best tool for assessment of overall disease severity, given the complexity of OSA, the impact of the disease on patient quality of life, and the risk for downstream cardiovascular disease, said Dr. Huntley.
The findings were limited by the retrospective design and use of data from a single center.
Population-level data are needed to identify variables that may be meaningful to create a future tool that provides the best picture of the individual patient’s disease, he added. Additional prospective data are also needed to assess the impact of the scoring system on long-term treatment outcomes.
“The current study is especially interesting as we are just beginning to understand the factors that predict cardiovascular risk for patients with obstructive sleep apnea,” Megan Durr, MD, of the University of California, San Francisco, said in an interview.
“For a long time, we primarily looked at the AHI and/or oxygen levels during sleep as risk factors, and we haven’t looked as much at other factors.” said Dr. Durr, who served as a moderator for the session in which the study was presented.
The current findings provide a more comprehensive look at cardiovascular risk; the inclusion of patient anatomy and symptoms add to the knowledge of this topic, and will lead to further work in this area, she added.
The study received no outside funding. The researchers had no financial conflicts to disclose. Dr. Huntley disclosed receiving research support from Nyxoah and Inspire, and serving as a consultant for Nyxoah, Inspire, and Avivomed.
Dr. Durr had no financial conflicts to disclose.
A version of this article appeared on Medscape.com.
FROM AAO-HNS 2024
Walking App Works Only if Users Think It Does
TOPLINE:
Apps designed to increase physical activity may be useful in increasing daily step counts for users who believe the intervention beneficial, but not for those who do not. The app’s effectiveness is notably influenced by how users perceive its utility.
METHODOLOGY:
- Researchers conducted a randomized controlled trial from February 2021 to May 2022 to evaluate the effectiveness of SNapp, an adaptive app designed to promote walking through tailored coaching content.
- Overall, 176 adults (76% women; mean age, 56 years) were randomly assigned to use the app plus tailored coaching content (SNapp group; n = 87) or only the step counter app (control group; n = 89).
- SNapp’s coaching content provided personalized feedback on step counts and recommendations for increasing walking, while also considering individual preferences for behavior change techniques.
- The primary outcome was the daily step count recorded by the app, which was updated on an hourly basis in a database over an intervention period of 12 months.
- Perceptions of ease of use and usefulness were assessed to determine their effect on the effectiveness of the app.
TAKEAWAY:
- Intervention group participants used the app nearly 30% of days, while those using the app alone showed almost identical use.
- The SNapp intervention did not significantly affect the step counts on average over time (B, −202.30; 95% CI, −889.7 to 485.1).
- Perceived usefulness significantly moderated the intervention effect of SNapp (B, 344.38; 90% CI, 40.4-648.3), but perceived ease of use did not (B, 38.60; 90% CI, −276.5 to 353.7).
- Among participants with a high perceived usefulness, the SNapp group had a higher median step count than the control group (median difference, 1260 steps; 90% CI, −3243.7 to 1298.2); however, this difference was not statistically significant.
IN PRACTICE:
“This study shows that perceived usefulness is also an important factor influencing behavioral effects. Hence, it is essential for apps to be perceived as useful to effectively improve users’ activity levels,” the authors wrote.
SOURCE:
The study was led by Anne L. Vos, PhD, of the Amsterdam School of Communication Research at the University of Amsterdam, in the Netherlands. It was published online on September 16, 2024, in the American Journal of Preventive Medicine.
LIMITATIONS:
The study’s recruitment strategy primarily attracted highly educated individuals, limiting generalizability. The app’s accuracy in measuring steps could be improved, as it sometimes underestimated step counts. Researchers also were unable to check if participants read messages from coaches.
DISCLOSURES:
The study was supported by grants from the Dutch Heart Foundation and the Netherlands Organisation for Health Research and Development. No relevant conflicts of interest were disclosed by the authors.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
TOPLINE:
Apps designed to increase physical activity may be useful in increasing daily step counts for users who believe the intervention beneficial, but not for those who do not. The app’s effectiveness is notably influenced by how users perceive its utility.
METHODOLOGY:
- Researchers conducted a randomized controlled trial from February 2021 to May 2022 to evaluate the effectiveness of SNapp, an adaptive app designed to promote walking through tailored coaching content.
- Overall, 176 adults (76% women; mean age, 56 years) were randomly assigned to use the app plus tailored coaching content (SNapp group; n = 87) or only the step counter app (control group; n = 89).
- SNapp’s coaching content provided personalized feedback on step counts and recommendations for increasing walking, while also considering individual preferences for behavior change techniques.
- The primary outcome was the daily step count recorded by the app, which was updated on an hourly basis in a database over an intervention period of 12 months.
- Perceptions of ease of use and usefulness were assessed to determine their effect on the effectiveness of the app.
TAKEAWAY:
- Intervention group participants used the app nearly 30% of days, while those using the app alone showed almost identical use.
- The SNapp intervention did not significantly affect the step counts on average over time (B, −202.30; 95% CI, −889.7 to 485.1).
- Perceived usefulness significantly moderated the intervention effect of SNapp (B, 344.38; 90% CI, 40.4-648.3), but perceived ease of use did not (B, 38.60; 90% CI, −276.5 to 353.7).
- Among participants with a high perceived usefulness, the SNapp group had a higher median step count than the control group (median difference, 1260 steps; 90% CI, −3243.7 to 1298.2); however, this difference was not statistically significant.
IN PRACTICE:
“This study shows that perceived usefulness is also an important factor influencing behavioral effects. Hence, it is essential for apps to be perceived as useful to effectively improve users’ activity levels,” the authors wrote.
SOURCE:
The study was led by Anne L. Vos, PhD, of the Amsterdam School of Communication Research at the University of Amsterdam, in the Netherlands. It was published online on September 16, 2024, in the American Journal of Preventive Medicine.
LIMITATIONS:
The study’s recruitment strategy primarily attracted highly educated individuals, limiting generalizability. The app’s accuracy in measuring steps could be improved, as it sometimes underestimated step counts. Researchers also were unable to check if participants read messages from coaches.
DISCLOSURES:
The study was supported by grants from the Dutch Heart Foundation and the Netherlands Organisation for Health Research and Development. No relevant conflicts of interest were disclosed by the authors.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
TOPLINE:
Apps designed to increase physical activity may be useful in increasing daily step counts for users who believe the intervention beneficial, but not for those who do not. The app’s effectiveness is notably influenced by how users perceive its utility.
METHODOLOGY:
- Researchers conducted a randomized controlled trial from February 2021 to May 2022 to evaluate the effectiveness of SNapp, an adaptive app designed to promote walking through tailored coaching content.
- Overall, 176 adults (76% women; mean age, 56 years) were randomly assigned to use the app plus tailored coaching content (SNapp group; n = 87) or only the step counter app (control group; n = 89).
- SNapp’s coaching content provided personalized feedback on step counts and recommendations for increasing walking, while also considering individual preferences for behavior change techniques.
- The primary outcome was the daily step count recorded by the app, which was updated on an hourly basis in a database over an intervention period of 12 months.
- Perceptions of ease of use and usefulness were assessed to determine their effect on the effectiveness of the app.
TAKEAWAY:
- Intervention group participants used the app nearly 30% of days, while those using the app alone showed almost identical use.
- The SNapp intervention did not significantly affect the step counts on average over time (B, −202.30; 95% CI, −889.7 to 485.1).
- Perceived usefulness significantly moderated the intervention effect of SNapp (B, 344.38; 90% CI, 40.4-648.3), but perceived ease of use did not (B, 38.60; 90% CI, −276.5 to 353.7).
- Among participants with a high perceived usefulness, the SNapp group had a higher median step count than the control group (median difference, 1260 steps; 90% CI, −3243.7 to 1298.2); however, this difference was not statistically significant.
IN PRACTICE:
“This study shows that perceived usefulness is also an important factor influencing behavioral effects. Hence, it is essential for apps to be perceived as useful to effectively improve users’ activity levels,” the authors wrote.
SOURCE:
The study was led by Anne L. Vos, PhD, of the Amsterdam School of Communication Research at the University of Amsterdam, in the Netherlands. It was published online on September 16, 2024, in the American Journal of Preventive Medicine.
LIMITATIONS:
The study’s recruitment strategy primarily attracted highly educated individuals, limiting generalizability. The app’s accuracy in measuring steps could be improved, as it sometimes underestimated step counts. Researchers also were unable to check if participants read messages from coaches.
DISCLOSURES:
The study was supported by grants from the Dutch Heart Foundation and the Netherlands Organisation for Health Research and Development. No relevant conflicts of interest were disclosed by the authors.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
Environmental, Metabolic Factors Driving Global Rise in Stroke
Air pollution, high temperatures, and metabolic risk factors are driving global increases in stroke, contributing to 12 million cases and more than 7 million deaths from stroke each year, new data from the Global Burden of Disease (GBD) study showed.
Between 1990 and 2021, the number of people who experienced a stroke increased to 11.9 million (up by 70% since 1990), while the number of stroke survivors rose to 93.8 million (up by 86%), and stroke-related deaths rose to 7.3 million (up by 44%), making stroke the third leading cause of death worldwide after ischemic heart disease and COVID-19, investigators found.
Stroke is highly preventable, the investigators noted, with 84% of the stroke burden in 2021 attributable to 23 modifiable risk factors, including air pollution, excess body weight, high blood pressure, smoking, and physical inactivity.
This means there are “tremendous opportunities to alter the trajectory of stroke risk for the next generation,” Catherine O. Johnson, MPH, PhD, co-author and lead research scientist at the Institute for Health Metrics and Evaluation (IHME), University of Washington, Seattle, said in a news release.
The study was published online in The Lancet Neurology.
Top Risk Factor for Subarachnoid Hemorrhage
Since 1990, the contribution of high temperatures to poor health and early death due to stroke has risen 72%, a trend likely to increase in the future — underscoring the impact of environmental factors on the growing stroke burden, the authors said.
“Given that ambient air pollution is reciprocally linked with ambient temperature and climate change, the importance of urgent climate actions and measures to reduce air pollution cannot be overestimated,” Dr. Johnson said.
Mitchell S.V. Elkind, MD, MS, chief clinical science officer for the American Heart Association, who wasn’t involved in the study, told this news organization that environmental factors such as air pollution, particulate matter from wildfires and other sources, and excessive heat are now recognized as major contributors to the risk for stroke. “This should not be surprising as we have long recognized the risks of stroke associated with toxins in cigarette smoke, which likely share mechanisms for vascular damage with pollutants,” Dr. Elkind said.
The data also reveal for the first time that ambient particulate matter air pollution is a top risk factor for subarachnoid hemorrhage, contributing to 14% of the death and disability caused by this serious stroke subtype, on a par with smoking.
Dr. Elkind noted that smoking is “a major risk factor for subarachnoid hemorrhage. It makes sense that particulate air pollution would therefore similarly be a risk factor for subarachnoid hemorrhage, which similarly damages blood vessels. Prior studies were likely too small or did not assess the role of air pollution in subarachnoid hemorrhage.”
The analysis also showed substantial increases between 1990 and 2021 in the global stroke burden linked to high body mass index (up by 88%), high blood sugar (up 32%), a diet high in sugar-sweetened drinks (up 23%), low physical activity (up 11%), high systolic blood pressure (up 7%), and a diet low in omega-6 polyunsaturated fatty acids (up 5%).
“And with increasing exposure to risk factors such as high blood sugar and diet high in sugar-sweetened drinks, there is a critical need for interventions focused on obesity and metabolic syndromes,” Dr. Johnson said.
“Identifying sustainable ways to work with communities to take action to prevent and control modifiable risk factors for stroke is essential to address this growing crisis,” she added.
Prevention Strategies Fall Short
The data also showed that stroke-related disability-adjusted life-years rose from around 121.4 million years of healthy life lost in 1990 to 160.5 million years in 2021, making stroke the fourth leading cause of health loss worldwide after COVID-19, ischemic heart disease, and neonatal disorders.
“The global growth of the number of people who develop stroke and died from or remain disabled by stroke is growing fast, strongly suggesting that currently used stroke prevention strategies are not sufficiently effective,” lead author Valery L. Feigin, MD, PhD, from Auckland University of Technology, Auckland, New Zealand, and affiliate professor at IHME, said in the release.
“New, proven effective population-wide and motivational individual prevention strategies that could be applied to all people at risk of having a stroke, regardless of the level of risk, as recommended in the recent Lancet Neurology Commission on Stroke should be implemented across the globe urgently,” said Dr. Feigin.
Dr. Elkind said the AHA supports research on the effects of air quality on risk for vascular injury and stroke and has “long advocated for policies to mitigate the adverse health impacts of air pollutants, including reduction of vehicle emissions and renewable portfolio standards, taking into account racial, ethnic, and economic disparities.”
“AHA, and the healthcare sector more broadly, must take a leadership role in recommending policies to improve environmental air quality and in working with the private sector and industry to improve air quality,” Dr. Elkind said.
In an accompanying commentary, Ming Liu, MD, and Simiao Wu, MD, PhD, West China Hospital, Sichuan University, Chengdu, China, wrote that “pragmatic solutions to the enormous and increasing stroke burden include surveillance, prevention, acute care, and rehabilitation.”
“Surveillance strategies include establishing a national-level framework for regular monitoring of stroke burden, risk factors, and healthcare services via community-based surveys and health records,” they noted.
“Artificial intelligence and mobile technologies might not only facilitate the dissemination of evidence-based health services but also increase the number of data sources and encourage participation of multidisciplinary collaborators, potentially improving the validity and accuracy of future GBD estimates,” they added.
This study was funded by the Bill & Melinda Gates Foundation. Author disclosures are listed with the original article.
A version of this article first appeared on Medscape.com.
Air pollution, high temperatures, and metabolic risk factors are driving global increases in stroke, contributing to 12 million cases and more than 7 million deaths from stroke each year, new data from the Global Burden of Disease (GBD) study showed.
Between 1990 and 2021, the number of people who experienced a stroke increased to 11.9 million (up by 70% since 1990), while the number of stroke survivors rose to 93.8 million (up by 86%), and stroke-related deaths rose to 7.3 million (up by 44%), making stroke the third leading cause of death worldwide after ischemic heart disease and COVID-19, investigators found.
Stroke is highly preventable, the investigators noted, with 84% of the stroke burden in 2021 attributable to 23 modifiable risk factors, including air pollution, excess body weight, high blood pressure, smoking, and physical inactivity.
This means there are “tremendous opportunities to alter the trajectory of stroke risk for the next generation,” Catherine O. Johnson, MPH, PhD, co-author and lead research scientist at the Institute for Health Metrics and Evaluation (IHME), University of Washington, Seattle, said in a news release.
The study was published online in The Lancet Neurology.
Top Risk Factor for Subarachnoid Hemorrhage
Since 1990, the contribution of high temperatures to poor health and early death due to stroke has risen 72%, a trend likely to increase in the future — underscoring the impact of environmental factors on the growing stroke burden, the authors said.
“Given that ambient air pollution is reciprocally linked with ambient temperature and climate change, the importance of urgent climate actions and measures to reduce air pollution cannot be overestimated,” Dr. Johnson said.
Mitchell S.V. Elkind, MD, MS, chief clinical science officer for the American Heart Association, who wasn’t involved in the study, told this news organization that environmental factors such as air pollution, particulate matter from wildfires and other sources, and excessive heat are now recognized as major contributors to the risk for stroke. “This should not be surprising as we have long recognized the risks of stroke associated with toxins in cigarette smoke, which likely share mechanisms for vascular damage with pollutants,” Dr. Elkind said.
The data also reveal for the first time that ambient particulate matter air pollution is a top risk factor for subarachnoid hemorrhage, contributing to 14% of the death and disability caused by this serious stroke subtype, on a par with smoking.
Dr. Elkind noted that smoking is “a major risk factor for subarachnoid hemorrhage. It makes sense that particulate air pollution would therefore similarly be a risk factor for subarachnoid hemorrhage, which similarly damages blood vessels. Prior studies were likely too small or did not assess the role of air pollution in subarachnoid hemorrhage.”
The analysis also showed substantial increases between 1990 and 2021 in the global stroke burden linked to high body mass index (up by 88%), high blood sugar (up 32%), a diet high in sugar-sweetened drinks (up 23%), low physical activity (up 11%), high systolic blood pressure (up 7%), and a diet low in omega-6 polyunsaturated fatty acids (up 5%).
“And with increasing exposure to risk factors such as high blood sugar and diet high in sugar-sweetened drinks, there is a critical need for interventions focused on obesity and metabolic syndromes,” Dr. Johnson said.
“Identifying sustainable ways to work with communities to take action to prevent and control modifiable risk factors for stroke is essential to address this growing crisis,” she added.
Prevention Strategies Fall Short
The data also showed that stroke-related disability-adjusted life-years rose from around 121.4 million years of healthy life lost in 1990 to 160.5 million years in 2021, making stroke the fourth leading cause of health loss worldwide after COVID-19, ischemic heart disease, and neonatal disorders.
“The global growth of the number of people who develop stroke and died from or remain disabled by stroke is growing fast, strongly suggesting that currently used stroke prevention strategies are not sufficiently effective,” lead author Valery L. Feigin, MD, PhD, from Auckland University of Technology, Auckland, New Zealand, and affiliate professor at IHME, said in the release.
“New, proven effective population-wide and motivational individual prevention strategies that could be applied to all people at risk of having a stroke, regardless of the level of risk, as recommended in the recent Lancet Neurology Commission on Stroke should be implemented across the globe urgently,” said Dr. Feigin.
Dr. Elkind said the AHA supports research on the effects of air quality on risk for vascular injury and stroke and has “long advocated for policies to mitigate the adverse health impacts of air pollutants, including reduction of vehicle emissions and renewable portfolio standards, taking into account racial, ethnic, and economic disparities.”
“AHA, and the healthcare sector more broadly, must take a leadership role in recommending policies to improve environmental air quality and in working with the private sector and industry to improve air quality,” Dr. Elkind said.
In an accompanying commentary, Ming Liu, MD, and Simiao Wu, MD, PhD, West China Hospital, Sichuan University, Chengdu, China, wrote that “pragmatic solutions to the enormous and increasing stroke burden include surveillance, prevention, acute care, and rehabilitation.”
“Surveillance strategies include establishing a national-level framework for regular monitoring of stroke burden, risk factors, and healthcare services via community-based surveys and health records,” they noted.
“Artificial intelligence and mobile technologies might not only facilitate the dissemination of evidence-based health services but also increase the number of data sources and encourage participation of multidisciplinary collaborators, potentially improving the validity and accuracy of future GBD estimates,” they added.
This study was funded by the Bill & Melinda Gates Foundation. Author disclosures are listed with the original article.
A version of this article first appeared on Medscape.com.
Air pollution, high temperatures, and metabolic risk factors are driving global increases in stroke, contributing to 12 million cases and more than 7 million deaths from stroke each year, new data from the Global Burden of Disease (GBD) study showed.
Between 1990 and 2021, the number of people who experienced a stroke increased to 11.9 million (up by 70% since 1990), while the number of stroke survivors rose to 93.8 million (up by 86%), and stroke-related deaths rose to 7.3 million (up by 44%), making stroke the third leading cause of death worldwide after ischemic heart disease and COVID-19, investigators found.
Stroke is highly preventable, the investigators noted, with 84% of the stroke burden in 2021 attributable to 23 modifiable risk factors, including air pollution, excess body weight, high blood pressure, smoking, and physical inactivity.
This means there are “tremendous opportunities to alter the trajectory of stroke risk for the next generation,” Catherine O. Johnson, MPH, PhD, co-author and lead research scientist at the Institute for Health Metrics and Evaluation (IHME), University of Washington, Seattle, said in a news release.
The study was published online in The Lancet Neurology.
Top Risk Factor for Subarachnoid Hemorrhage
Since 1990, the contribution of high temperatures to poor health and early death due to stroke has risen 72%, a trend likely to increase in the future — underscoring the impact of environmental factors on the growing stroke burden, the authors said.
“Given that ambient air pollution is reciprocally linked with ambient temperature and climate change, the importance of urgent climate actions and measures to reduce air pollution cannot be overestimated,” Dr. Johnson said.
Mitchell S.V. Elkind, MD, MS, chief clinical science officer for the American Heart Association, who wasn’t involved in the study, told this news organization that environmental factors such as air pollution, particulate matter from wildfires and other sources, and excessive heat are now recognized as major contributors to the risk for stroke. “This should not be surprising as we have long recognized the risks of stroke associated with toxins in cigarette smoke, which likely share mechanisms for vascular damage with pollutants,” Dr. Elkind said.
The data also reveal for the first time that ambient particulate matter air pollution is a top risk factor for subarachnoid hemorrhage, contributing to 14% of the death and disability caused by this serious stroke subtype, on a par with smoking.
Dr. Elkind noted that smoking is “a major risk factor for subarachnoid hemorrhage. It makes sense that particulate air pollution would therefore similarly be a risk factor for subarachnoid hemorrhage, which similarly damages blood vessels. Prior studies were likely too small or did not assess the role of air pollution in subarachnoid hemorrhage.”
The analysis also showed substantial increases between 1990 and 2021 in the global stroke burden linked to high body mass index (up by 88%), high blood sugar (up 32%), a diet high in sugar-sweetened drinks (up 23%), low physical activity (up 11%), high systolic blood pressure (up 7%), and a diet low in omega-6 polyunsaturated fatty acids (up 5%).
“And with increasing exposure to risk factors such as high blood sugar and diet high in sugar-sweetened drinks, there is a critical need for interventions focused on obesity and metabolic syndromes,” Dr. Johnson said.
“Identifying sustainable ways to work with communities to take action to prevent and control modifiable risk factors for stroke is essential to address this growing crisis,” she added.
Prevention Strategies Fall Short
The data also showed that stroke-related disability-adjusted life-years rose from around 121.4 million years of healthy life lost in 1990 to 160.5 million years in 2021, making stroke the fourth leading cause of health loss worldwide after COVID-19, ischemic heart disease, and neonatal disorders.
“The global growth of the number of people who develop stroke and died from or remain disabled by stroke is growing fast, strongly suggesting that currently used stroke prevention strategies are not sufficiently effective,” lead author Valery L. Feigin, MD, PhD, from Auckland University of Technology, Auckland, New Zealand, and affiliate professor at IHME, said in the release.
“New, proven effective population-wide and motivational individual prevention strategies that could be applied to all people at risk of having a stroke, regardless of the level of risk, as recommended in the recent Lancet Neurology Commission on Stroke should be implemented across the globe urgently,” said Dr. Feigin.
Dr. Elkind said the AHA supports research on the effects of air quality on risk for vascular injury and stroke and has “long advocated for policies to mitigate the adverse health impacts of air pollutants, including reduction of vehicle emissions and renewable portfolio standards, taking into account racial, ethnic, and economic disparities.”
“AHA, and the healthcare sector more broadly, must take a leadership role in recommending policies to improve environmental air quality and in working with the private sector and industry to improve air quality,” Dr. Elkind said.
In an accompanying commentary, Ming Liu, MD, and Simiao Wu, MD, PhD, West China Hospital, Sichuan University, Chengdu, China, wrote that “pragmatic solutions to the enormous and increasing stroke burden include surveillance, prevention, acute care, and rehabilitation.”
“Surveillance strategies include establishing a national-level framework for regular monitoring of stroke burden, risk factors, and healthcare services via community-based surveys and health records,” they noted.
“Artificial intelligence and mobile technologies might not only facilitate the dissemination of evidence-based health services but also increase the number of data sources and encourage participation of multidisciplinary collaborators, potentially improving the validity and accuracy of future GBD estimates,” they added.
This study was funded by the Bill & Melinda Gates Foundation. Author disclosures are listed with the original article.
A version of this article first appeared on Medscape.com.
Controlling Six Risk Factors Can Combat CKD in Obesity
TOPLINE:
Optimal management of blood pressure, A1c levels, low-density lipoprotein cholesterol (LDL-C), albuminuria, smoking, and physical activity may reduce the excess risk for chronic kidney disease (CKD) typically linked to obesity. The protective effect is more pronounced in men, in those with lower healthy food scores, and in users of diabetes medication.
METHODOLOGY:
- Obesity is a significant risk factor for CKD, but it is unknown if managing multiple other obesity-related CKD risk factors can mitigate the excess CKD risk.
- Researchers assessed CKD risk factor control in 97,538 participants with obesity from the UK Biobank and compared them with an equal number of age- and sex-matched control participants with normal body weight and no CKD at baseline.
- Participants with obesity were assessed for six modifiable risk factors: Blood pressure, A1c levels, LDL-C, albuminuria, smoking, and physical activity.
- Overall, 2487, 12,720, 32,388, 36,988, and 15,381 participants with obesity had at most two, three, four, five, and six risk factors under combined control, respectively, with the two or fewer group serving as the reference.
- The primary outcome was incident CKD and the degree of combined risk factor control in persons. The CKD risk and risk factor control in participants with obesity were also compared with CKD incidence in matched normal weight participants.
TAKEAWAY:
- During a median follow-up period of 10.8 years, 3954 cases of incident CKD were reported in participants with obesity and 1498 cases in matched persons of normal body mass index (BMI).
- In a stepwise pattern, optimal control of each additional risk factor was associated with 11% (adjusted hazard ratio [aHR], 0.89; 95% CI, 0.86-0.91) reduction in the incidence of CKD events, down to a 49% reduction in CKD incidence (aHR, 0.51; 95% CI, 0.43-0.61) for combined control of all six risk factors in participants with obesity.
- The protective effect of combined control of risk factors was more pronounced in men vs women, in those with lower vs higher healthy diet scores, and in users vs nonusers of diabetes medication.
- A similar stepwise pattern emerged between the number of risk factors controlled and CKD risk in participants with obesity compared with matched individuals of normal BMI, with the excess CKD risk eliminated in participants with obesity with six risk factors under control.
IN PRACTICE:
“Comprehensive control of risk factors might effectively neutralize the excessive CKD risk associated with obesity, emphasizing the potential of a joint management approach in the prevention of CKD in this population,” the authors wrote.
SOURCE:
The study was led by Rui Tang, MS, Department of Epidemiology, School of Public Health and Tropical Medicine, Tulane University, New Orleans, Louisiana. It was published online in Diabetes, Obesity and Metabolism.
LIMITATIONS:
The evaluated risk factors for CKD were arbitrarily selected, which may not represent the ideal group. The study did not consider the time-varying effect of joint risk factor control owing to the lack of some variables such as A1c. The generalizability of the findings was limited because over 90% of the UK Biobank cohort is composed of White people and individuals with healthier behaviors compared with the overall UK population.
DISCLOSURES:
The study was supported by grants from the US National Heart, Lung, and Blood Institute and the National Institute of Diabetes and Digestive and Kidney Diseases. The authors declared no conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
TOPLINE:
Optimal management of blood pressure, A1c levels, low-density lipoprotein cholesterol (LDL-C), albuminuria, smoking, and physical activity may reduce the excess risk for chronic kidney disease (CKD) typically linked to obesity. The protective effect is more pronounced in men, in those with lower healthy food scores, and in users of diabetes medication.
METHODOLOGY:
- Obesity is a significant risk factor for CKD, but it is unknown if managing multiple other obesity-related CKD risk factors can mitigate the excess CKD risk.
- Researchers assessed CKD risk factor control in 97,538 participants with obesity from the UK Biobank and compared them with an equal number of age- and sex-matched control participants with normal body weight and no CKD at baseline.
- Participants with obesity were assessed for six modifiable risk factors: Blood pressure, A1c levels, LDL-C, albuminuria, smoking, and physical activity.
- Overall, 2487, 12,720, 32,388, 36,988, and 15,381 participants with obesity had at most two, three, four, five, and six risk factors under combined control, respectively, with the two or fewer group serving as the reference.
- The primary outcome was incident CKD and the degree of combined risk factor control in persons. The CKD risk and risk factor control in participants with obesity were also compared with CKD incidence in matched normal weight participants.
TAKEAWAY:
- During a median follow-up period of 10.8 years, 3954 cases of incident CKD were reported in participants with obesity and 1498 cases in matched persons of normal body mass index (BMI).
- In a stepwise pattern, optimal control of each additional risk factor was associated with 11% (adjusted hazard ratio [aHR], 0.89; 95% CI, 0.86-0.91) reduction in the incidence of CKD events, down to a 49% reduction in CKD incidence (aHR, 0.51; 95% CI, 0.43-0.61) for combined control of all six risk factors in participants with obesity.
- The protective effect of combined control of risk factors was more pronounced in men vs women, in those with lower vs higher healthy diet scores, and in users vs nonusers of diabetes medication.
- A similar stepwise pattern emerged between the number of risk factors controlled and CKD risk in participants with obesity compared with matched individuals of normal BMI, with the excess CKD risk eliminated in participants with obesity with six risk factors under control.
IN PRACTICE:
“Comprehensive control of risk factors might effectively neutralize the excessive CKD risk associated with obesity, emphasizing the potential of a joint management approach in the prevention of CKD in this population,” the authors wrote.
SOURCE:
The study was led by Rui Tang, MS, Department of Epidemiology, School of Public Health and Tropical Medicine, Tulane University, New Orleans, Louisiana. It was published online in Diabetes, Obesity and Metabolism.
LIMITATIONS:
The evaluated risk factors for CKD were arbitrarily selected, which may not represent the ideal group. The study did not consider the time-varying effect of joint risk factor control owing to the lack of some variables such as A1c. The generalizability of the findings was limited because over 90% of the UK Biobank cohort is composed of White people and individuals with healthier behaviors compared with the overall UK population.
DISCLOSURES:
The study was supported by grants from the US National Heart, Lung, and Blood Institute and the National Institute of Diabetes and Digestive and Kidney Diseases. The authors declared no conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
TOPLINE:
Optimal management of blood pressure, A1c levels, low-density lipoprotein cholesterol (LDL-C), albuminuria, smoking, and physical activity may reduce the excess risk for chronic kidney disease (CKD) typically linked to obesity. The protective effect is more pronounced in men, in those with lower healthy food scores, and in users of diabetes medication.
METHODOLOGY:
- Obesity is a significant risk factor for CKD, but it is unknown if managing multiple other obesity-related CKD risk factors can mitigate the excess CKD risk.
- Researchers assessed CKD risk factor control in 97,538 participants with obesity from the UK Biobank and compared them with an equal number of age- and sex-matched control participants with normal body weight and no CKD at baseline.
- Participants with obesity were assessed for six modifiable risk factors: Blood pressure, A1c levels, LDL-C, albuminuria, smoking, and physical activity.
- Overall, 2487, 12,720, 32,388, 36,988, and 15,381 participants with obesity had at most two, three, four, five, and six risk factors under combined control, respectively, with the two or fewer group serving as the reference.
- The primary outcome was incident CKD and the degree of combined risk factor control in persons. The CKD risk and risk factor control in participants with obesity were also compared with CKD incidence in matched normal weight participants.
TAKEAWAY:
- During a median follow-up period of 10.8 years, 3954 cases of incident CKD were reported in participants with obesity and 1498 cases in matched persons of normal body mass index (BMI).
- In a stepwise pattern, optimal control of each additional risk factor was associated with 11% (adjusted hazard ratio [aHR], 0.89; 95% CI, 0.86-0.91) reduction in the incidence of CKD events, down to a 49% reduction in CKD incidence (aHR, 0.51; 95% CI, 0.43-0.61) for combined control of all six risk factors in participants with obesity.
- The protective effect of combined control of risk factors was more pronounced in men vs women, in those with lower vs higher healthy diet scores, and in users vs nonusers of diabetes medication.
- A similar stepwise pattern emerged between the number of risk factors controlled and CKD risk in participants with obesity compared with matched individuals of normal BMI, with the excess CKD risk eliminated in participants with obesity with six risk factors under control.
IN PRACTICE:
“Comprehensive control of risk factors might effectively neutralize the excessive CKD risk associated with obesity, emphasizing the potential of a joint management approach in the prevention of CKD in this population,” the authors wrote.
SOURCE:
The study was led by Rui Tang, MS, Department of Epidemiology, School of Public Health and Tropical Medicine, Tulane University, New Orleans, Louisiana. It was published online in Diabetes, Obesity and Metabolism.
LIMITATIONS:
The evaluated risk factors for CKD were arbitrarily selected, which may not represent the ideal group. The study did not consider the time-varying effect of joint risk factor control owing to the lack of some variables such as A1c. The generalizability of the findings was limited because over 90% of the UK Biobank cohort is composed of White people and individuals with healthier behaviors compared with the overall UK population.
DISCLOSURES:
The study was supported by grants from the US National Heart, Lung, and Blood Institute and the National Institute of Diabetes and Digestive and Kidney Diseases. The authors declared no conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
Rheumatology Clinic Interventions for Smoking, Blood Pressure ‘Make a Big Difference’
Two relatively simple interventions — addressing high blood pressure (BP) and smoking cessation — could make a huge difference for patients with rheumatic disease. Patients with autoimmune disease are up to three times more likely to develop cardiovascular disease (CVD) than the general population. In addition to compounding CVD, smoking is tied to the development of certain autoimmune conditions, as well as worse outcomes. Christie Bartels, MD, chief of the Division of Rheumatology at the University of Wisconsin School of Medicine and Public Health, Madison, has focused her research on improving cardiac health in inflammatory diseases. This news organization spoke with Bartels about two short interventions she developed that tackle hypertension and smoking cessation during regular visits, each taking less than 3 minutes.
How Do These Programs Address Cardiac Disease Prevention?
The BP and Quit Connect programs help clinics systematically address the two most modifiable risk factors for CVD: high BP and smoking. There’s also evidence that addressing these two risk factors improves outcomes in rheumatic diseases. Hypertension predicts an increase in lupus damage. Particularly in lupus nephritis, hypertension will increase the risk for CVD and kidney failure. People who use tobacco have worse outcomes in diseases like rheumatoid arthritis, psoriatic arthritis, and lupus, as well as more CVD, and antirheumatic drugs may not work as well.
In 90 seconds to 3 minutes, staff can do protocol-based care, which we’ve done across 20,000-plus visits. We showed we can improve population level rates of high BP and BP control, as well as increase smoking quitting rates across different patient settings.
What Is the Quit Connect Program?
The Quit Connect program is a 10- to 90-second point of care intervention. During rooming, staff (medical assistants and nurses) ask patients: “A) Do you smoke? and B) Have you thought about cutting back or quitting in the next 30 days?”
It turns out, when you ask the question that way, between a third and a half of people say that they’ve thought about cutting back or quitting. Then, we can get patients connected directly to Quitline, a free public service across all 50 states that smokers can use to get cessation support.
If patients are ready, we ask if we can arrange for them to receive a call from a Quitline coach about setting a quit date or receiving free nicotine replacement therapy. The beautiful thing is when that all happens, A) it’s free to the patient, and B) the results from the Quitline can be recorded right back to the electronic health record.
In our most recent publication in Arthritis Care & Research, we documented bringing Quit Connect to Grady Hospital in downtown Atlanta. It’s a safety net hospital, where 80% patients are Black and 70%-80% patients are on public insurance or uninsured. Using this protocol, we improved Quitline referrals 20-fold.
What Is the BP Connect Program?
At least half of the encounters in United States happen in specialty clinics. Unfortunately, when patients get their BP measured in a specialty clinic that’s not a cardiology or a vascular clinic, often, even if the pressure is high, the clinic doesn’t give patients feedback on that. The problem is because we haven’t said anything, that gives people the false reassurance that their BP is okay.
We’ve developed a 3-minute protocol to ask, advise, and connect. The idea is that if we measure a high BP, then we remeasure and confirm that it’s high. Then, we advise why it matters in rheumatic disease: Patients with rheumatic diseases are already at an increased risk for heart disease, and controlling BP can make a big difference. Then, we connect patients with high BP back to primary care.
Specifically, a SmartSet — an electronic medical record feature — prompts different actions based on confirmed high BP readings:
- If systolic BP ≥ 140-159, the SmartSet directs scheduling a visit to a nurse or primary care provider.
- If systolic BP ≥ 160-179, the next primary care visit anticipates the need to see a prescriber.
- If systolic BP ≥ 180, then the medical assistant or nurse at the visit is instructed to notify the provider who can arrange a provider-to-provider handoff for safety to exclude a hypertensive emergency.
That order goes to the scheduler to call primary care to coordinate follow-up. BP Connect doubled the likelihood of a guideline-recommended follow-up in primary care within 30 days. All patients benefited, and disparities decreased. BP Connect has had 1100 downloads, and both BP and Quit Connect programs are endorsed by the Centers for Disease Control and Prevention and Million Hearts.
How Do These Programs Affect Clinical Practice?
We developed these interventions with a health system engineer, and we time stamped everything. Part of the sustainability of this model is that it fits within a regular workflow. As a practicing rheumatologist, I understand that time is a precious commodity.
The interventions are in partnership with frontline staff. We’ve received feedback that they feel pride participating in these initiatives. They can say, because of me, 30 patients followed up last month for high BP, or 10 patients took a referral to the Quitline last year. We celebrate these accomplishments with the staff.
What Are the Next Steps for These Programs?
Public-facing toolkits for both BP and Quit Connect programs are available online. We have implemented [these programs] in a rural setting, in an urban setting, in Milwaukee and in Atlanta, and we are looking in the future to do a larger, multistate implementation study. If folks are interested, we’d love to partner with them to look at disseminating this further.
A version of this article appeared on Medscape.com.
Two relatively simple interventions — addressing high blood pressure (BP) and smoking cessation — could make a huge difference for patients with rheumatic disease. Patients with autoimmune disease are up to three times more likely to develop cardiovascular disease (CVD) than the general population. In addition to compounding CVD, smoking is tied to the development of certain autoimmune conditions, as well as worse outcomes. Christie Bartels, MD, chief of the Division of Rheumatology at the University of Wisconsin School of Medicine and Public Health, Madison, has focused her research on improving cardiac health in inflammatory diseases. This news organization spoke with Bartels about two short interventions she developed that tackle hypertension and smoking cessation during regular visits, each taking less than 3 minutes.
How Do These Programs Address Cardiac Disease Prevention?
The BP and Quit Connect programs help clinics systematically address the two most modifiable risk factors for CVD: high BP and smoking. There’s also evidence that addressing these two risk factors improves outcomes in rheumatic diseases. Hypertension predicts an increase in lupus damage. Particularly in lupus nephritis, hypertension will increase the risk for CVD and kidney failure. People who use tobacco have worse outcomes in diseases like rheumatoid arthritis, psoriatic arthritis, and lupus, as well as more CVD, and antirheumatic drugs may not work as well.
In 90 seconds to 3 minutes, staff can do protocol-based care, which we’ve done across 20,000-plus visits. We showed we can improve population level rates of high BP and BP control, as well as increase smoking quitting rates across different patient settings.
What Is the Quit Connect Program?
The Quit Connect program is a 10- to 90-second point of care intervention. During rooming, staff (medical assistants and nurses) ask patients: “A) Do you smoke? and B) Have you thought about cutting back or quitting in the next 30 days?”
It turns out, when you ask the question that way, between a third and a half of people say that they’ve thought about cutting back or quitting. Then, we can get patients connected directly to Quitline, a free public service across all 50 states that smokers can use to get cessation support.
If patients are ready, we ask if we can arrange for them to receive a call from a Quitline coach about setting a quit date or receiving free nicotine replacement therapy. The beautiful thing is when that all happens, A) it’s free to the patient, and B) the results from the Quitline can be recorded right back to the electronic health record.
In our most recent publication in Arthritis Care & Research, we documented bringing Quit Connect to Grady Hospital in downtown Atlanta. It’s a safety net hospital, where 80% patients are Black and 70%-80% patients are on public insurance or uninsured. Using this protocol, we improved Quitline referrals 20-fold.
What Is the BP Connect Program?
At least half of the encounters in United States happen in specialty clinics. Unfortunately, when patients get their BP measured in a specialty clinic that’s not a cardiology or a vascular clinic, often, even if the pressure is high, the clinic doesn’t give patients feedback on that. The problem is because we haven’t said anything, that gives people the false reassurance that their BP is okay.
We’ve developed a 3-minute protocol to ask, advise, and connect. The idea is that if we measure a high BP, then we remeasure and confirm that it’s high. Then, we advise why it matters in rheumatic disease: Patients with rheumatic diseases are already at an increased risk for heart disease, and controlling BP can make a big difference. Then, we connect patients with high BP back to primary care.
Specifically, a SmartSet — an electronic medical record feature — prompts different actions based on confirmed high BP readings:
- If systolic BP ≥ 140-159, the SmartSet directs scheduling a visit to a nurse or primary care provider.
- If systolic BP ≥ 160-179, the next primary care visit anticipates the need to see a prescriber.
- If systolic BP ≥ 180, then the medical assistant or nurse at the visit is instructed to notify the provider who can arrange a provider-to-provider handoff for safety to exclude a hypertensive emergency.
That order goes to the scheduler to call primary care to coordinate follow-up. BP Connect doubled the likelihood of a guideline-recommended follow-up in primary care within 30 days. All patients benefited, and disparities decreased. BP Connect has had 1100 downloads, and both BP and Quit Connect programs are endorsed by the Centers for Disease Control and Prevention and Million Hearts.
How Do These Programs Affect Clinical Practice?
We developed these interventions with a health system engineer, and we time stamped everything. Part of the sustainability of this model is that it fits within a regular workflow. As a practicing rheumatologist, I understand that time is a precious commodity.
The interventions are in partnership with frontline staff. We’ve received feedback that they feel pride participating in these initiatives. They can say, because of me, 30 patients followed up last month for high BP, or 10 patients took a referral to the Quitline last year. We celebrate these accomplishments with the staff.
What Are the Next Steps for These Programs?
Public-facing toolkits for both BP and Quit Connect programs are available online. We have implemented [these programs] in a rural setting, in an urban setting, in Milwaukee and in Atlanta, and we are looking in the future to do a larger, multistate implementation study. If folks are interested, we’d love to partner with them to look at disseminating this further.
A version of this article appeared on Medscape.com.
Two relatively simple interventions — addressing high blood pressure (BP) and smoking cessation — could make a huge difference for patients with rheumatic disease. Patients with autoimmune disease are up to three times more likely to develop cardiovascular disease (CVD) than the general population. In addition to compounding CVD, smoking is tied to the development of certain autoimmune conditions, as well as worse outcomes. Christie Bartels, MD, chief of the Division of Rheumatology at the University of Wisconsin School of Medicine and Public Health, Madison, has focused her research on improving cardiac health in inflammatory diseases. This news organization spoke with Bartels about two short interventions she developed that tackle hypertension and smoking cessation during regular visits, each taking less than 3 minutes.
How Do These Programs Address Cardiac Disease Prevention?
The BP and Quit Connect programs help clinics systematically address the two most modifiable risk factors for CVD: high BP and smoking. There’s also evidence that addressing these two risk factors improves outcomes in rheumatic diseases. Hypertension predicts an increase in lupus damage. Particularly in lupus nephritis, hypertension will increase the risk for CVD and kidney failure. People who use tobacco have worse outcomes in diseases like rheumatoid arthritis, psoriatic arthritis, and lupus, as well as more CVD, and antirheumatic drugs may not work as well.
In 90 seconds to 3 minutes, staff can do protocol-based care, which we’ve done across 20,000-plus visits. We showed we can improve population level rates of high BP and BP control, as well as increase smoking quitting rates across different patient settings.
What Is the Quit Connect Program?
The Quit Connect program is a 10- to 90-second point of care intervention. During rooming, staff (medical assistants and nurses) ask patients: “A) Do you smoke? and B) Have you thought about cutting back or quitting in the next 30 days?”
It turns out, when you ask the question that way, between a third and a half of people say that they’ve thought about cutting back or quitting. Then, we can get patients connected directly to Quitline, a free public service across all 50 states that smokers can use to get cessation support.
If patients are ready, we ask if we can arrange for them to receive a call from a Quitline coach about setting a quit date or receiving free nicotine replacement therapy. The beautiful thing is when that all happens, A) it’s free to the patient, and B) the results from the Quitline can be recorded right back to the electronic health record.
In our most recent publication in Arthritis Care & Research, we documented bringing Quit Connect to Grady Hospital in downtown Atlanta. It’s a safety net hospital, where 80% patients are Black and 70%-80% patients are on public insurance or uninsured. Using this protocol, we improved Quitline referrals 20-fold.
What Is the BP Connect Program?
At least half of the encounters in United States happen in specialty clinics. Unfortunately, when patients get their BP measured in a specialty clinic that’s not a cardiology or a vascular clinic, often, even if the pressure is high, the clinic doesn’t give patients feedback on that. The problem is because we haven’t said anything, that gives people the false reassurance that their BP is okay.
We’ve developed a 3-minute protocol to ask, advise, and connect. The idea is that if we measure a high BP, then we remeasure and confirm that it’s high. Then, we advise why it matters in rheumatic disease: Patients with rheumatic diseases are already at an increased risk for heart disease, and controlling BP can make a big difference. Then, we connect patients with high BP back to primary care.
Specifically, a SmartSet — an electronic medical record feature — prompts different actions based on confirmed high BP readings:
- If systolic BP ≥ 140-159, the SmartSet directs scheduling a visit to a nurse or primary care provider.
- If systolic BP ≥ 160-179, the next primary care visit anticipates the need to see a prescriber.
- If systolic BP ≥ 180, then the medical assistant or nurse at the visit is instructed to notify the provider who can arrange a provider-to-provider handoff for safety to exclude a hypertensive emergency.
That order goes to the scheduler to call primary care to coordinate follow-up. BP Connect doubled the likelihood of a guideline-recommended follow-up in primary care within 30 days. All patients benefited, and disparities decreased. BP Connect has had 1100 downloads, and both BP and Quit Connect programs are endorsed by the Centers for Disease Control and Prevention and Million Hearts.
How Do These Programs Affect Clinical Practice?
We developed these interventions with a health system engineer, and we time stamped everything. Part of the sustainability of this model is that it fits within a regular workflow. As a practicing rheumatologist, I understand that time is a precious commodity.
The interventions are in partnership with frontline staff. We’ve received feedback that they feel pride participating in these initiatives. They can say, because of me, 30 patients followed up last month for high BP, or 10 patients took a referral to the Quitline last year. We celebrate these accomplishments with the staff.
What Are the Next Steps for These Programs?
Public-facing toolkits for both BP and Quit Connect programs are available online. We have implemented [these programs] in a rural setting, in an urban setting, in Milwaukee and in Atlanta, and we are looking in the future to do a larger, multistate implementation study. If folks are interested, we’d love to partner with them to look at disseminating this further.
A version of this article appeared on Medscape.com.
Coffee’s ‘Sweet Spot’: Daily Consumption and Cardiometabolic Risk
Each and every day, 1 billion people on this planet ingest a particular psychoactive substance. This chemical has fairly profound physiologic effects. It increases levels of nitric oxide in the blood, leads to vasodilation, and, of course, makes you feel more awake. The substance comes in many forms but almost always in a liquid medium. Do you have it yet? That’s right. The substance is caffeine, quite possibly the healthiest recreational drug that has ever been discovered.
This might be my New England upbringing speaking, but when it comes to lifestyle and health, one of the rules I’ve internalized is that things that are pleasurable are generally bad for you. I know, I know — some of you love to exercise. Some of you love doing crosswords. But you know what I mean. I’m talking French fries, smoked meats, drugs, smoking, alcohol, binge-watching Firefly. You’d be suspicious if a study came out suggesting that eating ice cream in bed reduces your risk for heart attack, and so would I. So I’m always on the lookout for those unicorns of lifestyle factors, those rare things that you want to do and are also good for you.
So far, the data are strong for three things: sleeping, (safe) sexual activity, and coffee. You’ll have to stay tuned for articles about the first two. Today, we’re brewing up some deeper insights about the power of java.
I was inspired to write this article because of a paper, “Habitual Coffee, Tea, and Caffeine Consumption, Circulating Metabolites, and the Risk of Cardiometabolic Multimorbidity,” appearing September 17 in The Journal of Clinical Endocrinology and Metabolism (JCEM).
This is not the first study to suggest that coffee intake may be beneficial. A 2013 meta-analysis summarized the results of 36 studies with more than a million participants and found a U-shaped relationship between coffee intake and cardiovascular risk. The sweet spot was at three to five cups a day; people drinking that much coffee had about a 15% reduced risk for cardiovascular disease compared with nondrinkers.
But here’s the thing. Coffee contains caffeine, but it is much more than that. It is a heady brew of various chemicals and compounds, phenols, and chlorogenic acids. And, of course, you can get caffeine from stuff that isn’t coffee — natural things like tea — and decidedly unnatural things like energy drinks. How do you figure out where the benefit really lies?
The JCEM study leveraged the impressive UK Biobank dataset to figure this out. The Biobank recruited more than half a million people from the UK between 2006 and 2010 and collected a wealth of data from each of them: surveys, blood samples, biometrics, medical imaging — the works. And then they followed what would happen to those people medically over time. It’s a pretty amazing resource.
But for the purposes of this study, what you need to know is that just under 200,000 of those participants met the key criteria for this study: being free from cardiovascular disease at baseline; having completed a detailed survey about their coffee, tea, and other caffeinated beverage intake; and having adequate follow-up. A subset of that number, just under 100,000, had metabolomic data — which is where this study really gets interesting.
We’ll dive into the metabolome in a moment, but first let’s just talk about the main finding, the relationship between coffee, tea, or caffeine and cardiovascular disease. But to do that, we need to acknowledge that people who drink a lot of coffee are different from people who don’t, and it might be those differences, not the coffee itself, that are beneficial.
What were those differences? People who drank more coffee tended to be a bit older, were less likely to be female, and were slightly more likely to engage in physical activity. They ate less processed meat but also fewer vegetables. Some of those factors, like being female, are generally protective against cardiovascular disease; but some, like age, are definitely not. The authors adjusted for these and multiple other factors, including alcohol intake, BMI, kidney function, and many others to try to disentangle the effect of being the type of person who drinks a lot of coffee from the drinking a lot of coffee itself.
These are the results of the fully adjusted model. Compared with nonconsumers, you can see that people in the higher range of coffee, tea, or just caffeine intake have almost a 40% reduction in cardiovascular disease in follow-up.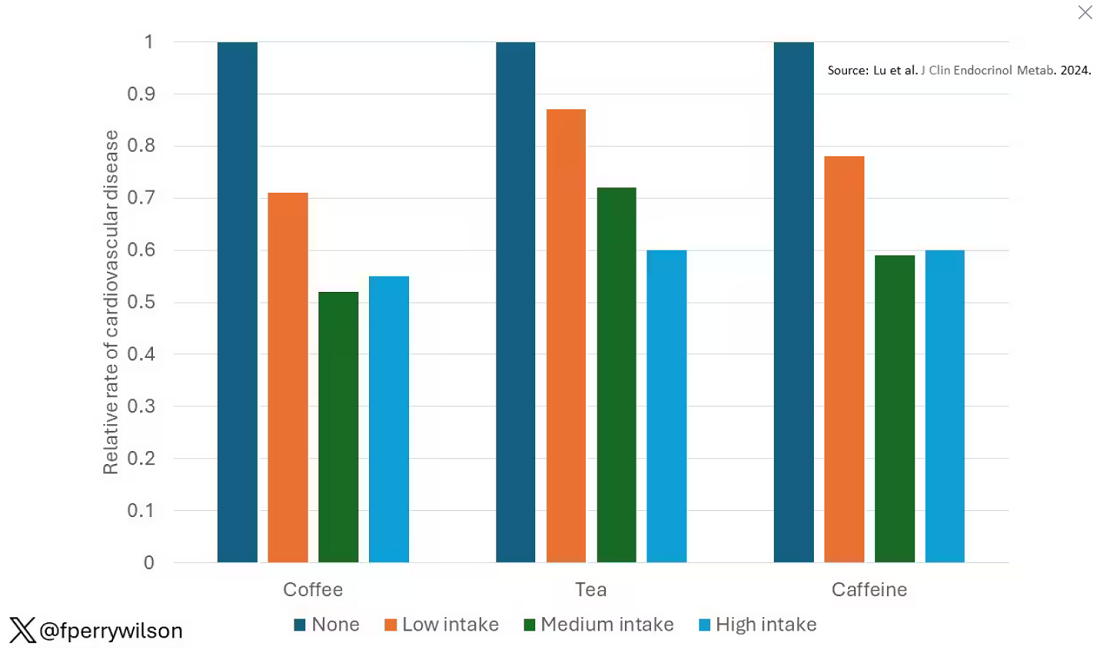
Looking at the benefit across the spectrum of intake, you again see that U-shaped curve, suggesting that a sweet spot for daily consumption can be found around 3 cups of coffee or tea (or 250 mg of caffeine). A standard energy drink contains about 120 mg of caffeine. 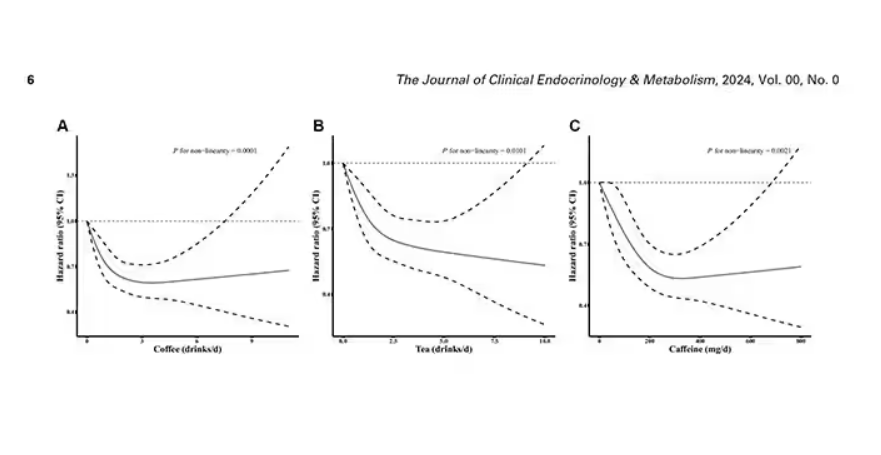
But if this is true, it would be good to know why. To figure that out, the authors turned to the metabolome. The idea here is that your body is constantly breaking stuff down, taking all these proteins and chemicals and compounds that we ingest and turning them into metabolites. Using advanced measurement techniques, researchers can measure hundreds or even thousands of metabolites from a single blood sample. They provide information, obviously, about the food you eat and the drinks you drink, but what is really intriguing is that some metabolites are associated with better health and some with worse
In this study, researchers measured 168 individual metabolites. Eighty of them, nearly half, were significantly altered in people who drank more coffee.
This figure summarizes the findings, and yes, this is way too complicated. 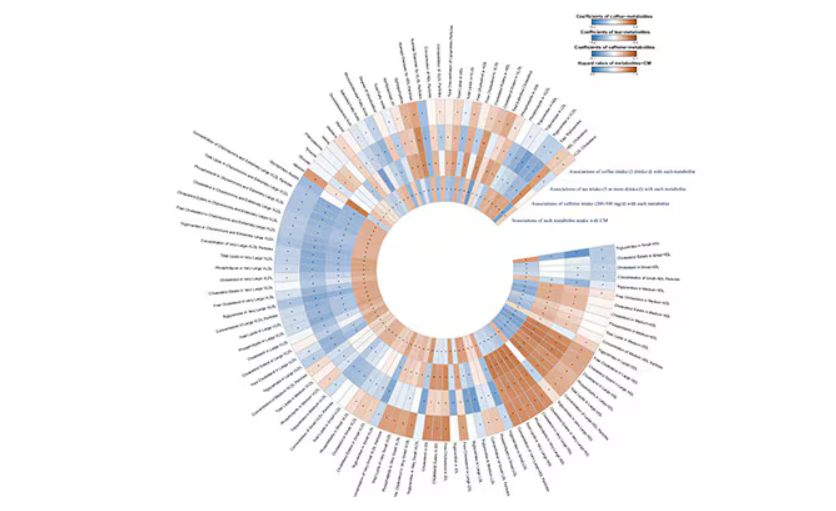
But here’s how to interpret it. The inner ring shows you how certain metabolites are associated with cardiovascular disease. The outer rings show you how those metabolites are associated with coffee, tea, or caffeine. The interesting part is that the sections of the ring (outer rings and inner rings) are very different colors.
Like here.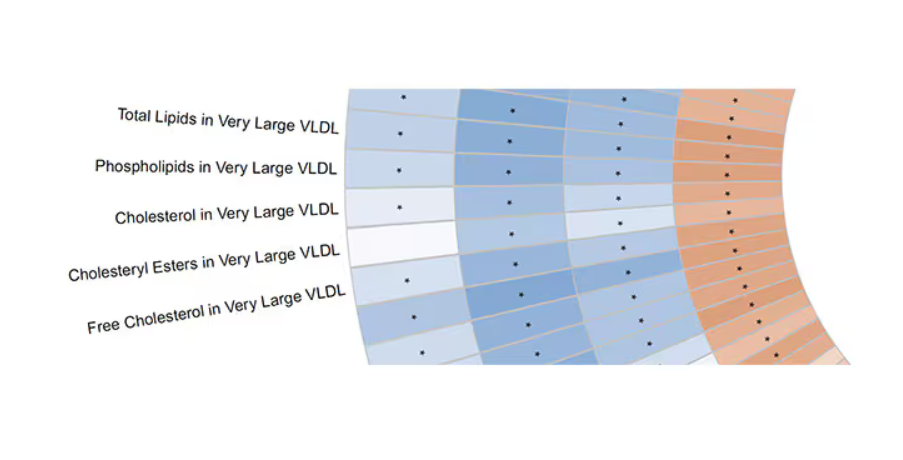
What you see here is a fairly profound effect that coffee, tea, or caffeine intake has on metabolites of VLDL — bad cholesterol. The beverages lower it, and, of course, higher levels lead to cardiovascular disease. This means that this is a potential causal pathway from coffee intake to heart protection.
And that’s not the only one.
You see a similar relationship for saturated fatty acids. Higher levels lead to cardiovascular disease, and coffee intake lowers levels. The reverse works too: Lower levels of histidine (an amino acid) increase cardiovascular risk, and coffee seems to raise those levels.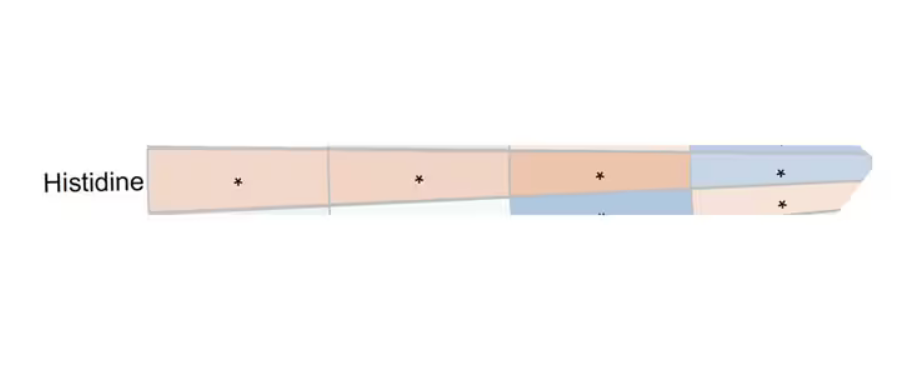
Is this all too good to be true? It’s hard to say. The data on coffee’s benefits have been remarkably consistent. Still, I wouldn’t be a good doctor if I didn’t mention that clearly there is a difference between a cup of black coffee and a venti caramel Frappuccino.
Nevertheless, coffee remains firmly in my holy trinity of enjoyable things that are, for whatever reason, still good for you. So, when you’re having that second, or third, or maybe fourth cup of the day, you can take that to heart.
Dr. Wilson, associate professor of medicine and public health and director of Yale’s Clinical and Translational Research Accelerator, reported no conflicts of interest.
A version of this article first appeared on Medscape.com.
Each and every day, 1 billion people on this planet ingest a particular psychoactive substance. This chemical has fairly profound physiologic effects. It increases levels of nitric oxide in the blood, leads to vasodilation, and, of course, makes you feel more awake. The substance comes in many forms but almost always in a liquid medium. Do you have it yet? That’s right. The substance is caffeine, quite possibly the healthiest recreational drug that has ever been discovered.
This might be my New England upbringing speaking, but when it comes to lifestyle and health, one of the rules I’ve internalized is that things that are pleasurable are generally bad for you. I know, I know — some of you love to exercise. Some of you love doing crosswords. But you know what I mean. I’m talking French fries, smoked meats, drugs, smoking, alcohol, binge-watching Firefly. You’d be suspicious if a study came out suggesting that eating ice cream in bed reduces your risk for heart attack, and so would I. So I’m always on the lookout for those unicorns of lifestyle factors, those rare things that you want to do and are also good for you.
So far, the data are strong for three things: sleeping, (safe) sexual activity, and coffee. You’ll have to stay tuned for articles about the first two. Today, we’re brewing up some deeper insights about the power of java.
I was inspired to write this article because of a paper, “Habitual Coffee, Tea, and Caffeine Consumption, Circulating Metabolites, and the Risk of Cardiometabolic Multimorbidity,” appearing September 17 in The Journal of Clinical Endocrinology and Metabolism (JCEM).
This is not the first study to suggest that coffee intake may be beneficial. A 2013 meta-analysis summarized the results of 36 studies with more than a million participants and found a U-shaped relationship between coffee intake and cardiovascular risk. The sweet spot was at three to five cups a day; people drinking that much coffee had about a 15% reduced risk for cardiovascular disease compared with nondrinkers.
But here’s the thing. Coffee contains caffeine, but it is much more than that. It is a heady brew of various chemicals and compounds, phenols, and chlorogenic acids. And, of course, you can get caffeine from stuff that isn’t coffee — natural things like tea — and decidedly unnatural things like energy drinks. How do you figure out where the benefit really lies?
The JCEM study leveraged the impressive UK Biobank dataset to figure this out. The Biobank recruited more than half a million people from the UK between 2006 and 2010 and collected a wealth of data from each of them: surveys, blood samples, biometrics, medical imaging — the works. And then they followed what would happen to those people medically over time. It’s a pretty amazing resource.
But for the purposes of this study, what you need to know is that just under 200,000 of those participants met the key criteria for this study: being free from cardiovascular disease at baseline; having completed a detailed survey about their coffee, tea, and other caffeinated beverage intake; and having adequate follow-up. A subset of that number, just under 100,000, had metabolomic data — which is where this study really gets interesting.
We’ll dive into the metabolome in a moment, but first let’s just talk about the main finding, the relationship between coffee, tea, or caffeine and cardiovascular disease. But to do that, we need to acknowledge that people who drink a lot of coffee are different from people who don’t, and it might be those differences, not the coffee itself, that are beneficial.
What were those differences? People who drank more coffee tended to be a bit older, were less likely to be female, and were slightly more likely to engage in physical activity. They ate less processed meat but also fewer vegetables. Some of those factors, like being female, are generally protective against cardiovascular disease; but some, like age, are definitely not. The authors adjusted for these and multiple other factors, including alcohol intake, BMI, kidney function, and many others to try to disentangle the effect of being the type of person who drinks a lot of coffee from the drinking a lot of coffee itself.
These are the results of the fully adjusted model. Compared with nonconsumers, you can see that people in the higher range of coffee, tea, or just caffeine intake have almost a 40% reduction in cardiovascular disease in follow-up.
Looking at the benefit across the spectrum of intake, you again see that U-shaped curve, suggesting that a sweet spot for daily consumption can be found around 3 cups of coffee or tea (or 250 mg of caffeine). A standard energy drink contains about 120 mg of caffeine. 
But if this is true, it would be good to know why. To figure that out, the authors turned to the metabolome. The idea here is that your body is constantly breaking stuff down, taking all these proteins and chemicals and compounds that we ingest and turning them into metabolites. Using advanced measurement techniques, researchers can measure hundreds or even thousands of metabolites from a single blood sample. They provide information, obviously, about the food you eat and the drinks you drink, but what is really intriguing is that some metabolites are associated with better health and some with worse
In this study, researchers measured 168 individual metabolites. Eighty of them, nearly half, were significantly altered in people who drank more coffee.
This figure summarizes the findings, and yes, this is way too complicated. 
But here’s how to interpret it. The inner ring shows you how certain metabolites are associated with cardiovascular disease. The outer rings show you how those metabolites are associated with coffee, tea, or caffeine. The interesting part is that the sections of the ring (outer rings and inner rings) are very different colors.
Like here.
What you see here is a fairly profound effect that coffee, tea, or caffeine intake has on metabolites of VLDL — bad cholesterol. The beverages lower it, and, of course, higher levels lead to cardiovascular disease. This means that this is a potential causal pathway from coffee intake to heart protection.
And that’s not the only one.
You see a similar relationship for saturated fatty acids. Higher levels lead to cardiovascular disease, and coffee intake lowers levels. The reverse works too: Lower levels of histidine (an amino acid) increase cardiovascular risk, and coffee seems to raise those levels.
Is this all too good to be true? It’s hard to say. The data on coffee’s benefits have been remarkably consistent. Still, I wouldn’t be a good doctor if I didn’t mention that clearly there is a difference between a cup of black coffee and a venti caramel Frappuccino.
Nevertheless, coffee remains firmly in my holy trinity of enjoyable things that are, for whatever reason, still good for you. So, when you’re having that second, or third, or maybe fourth cup of the day, you can take that to heart.
Dr. Wilson, associate professor of medicine and public health and director of Yale’s Clinical and Translational Research Accelerator, reported no conflicts of interest.
A version of this article first appeared on Medscape.com.
Each and every day, 1 billion people on this planet ingest a particular psychoactive substance. This chemical has fairly profound physiologic effects. It increases levels of nitric oxide in the blood, leads to vasodilation, and, of course, makes you feel more awake. The substance comes in many forms but almost always in a liquid medium. Do you have it yet? That’s right. The substance is caffeine, quite possibly the healthiest recreational drug that has ever been discovered.
This might be my New England upbringing speaking, but when it comes to lifestyle and health, one of the rules I’ve internalized is that things that are pleasurable are generally bad for you. I know, I know — some of you love to exercise. Some of you love doing crosswords. But you know what I mean. I’m talking French fries, smoked meats, drugs, smoking, alcohol, binge-watching Firefly. You’d be suspicious if a study came out suggesting that eating ice cream in bed reduces your risk for heart attack, and so would I. So I’m always on the lookout for those unicorns of lifestyle factors, those rare things that you want to do and are also good for you.
So far, the data are strong for three things: sleeping, (safe) sexual activity, and coffee. You’ll have to stay tuned for articles about the first two. Today, we’re brewing up some deeper insights about the power of java.
I was inspired to write this article because of a paper, “Habitual Coffee, Tea, and Caffeine Consumption, Circulating Metabolites, and the Risk of Cardiometabolic Multimorbidity,” appearing September 17 in The Journal of Clinical Endocrinology and Metabolism (JCEM).
This is not the first study to suggest that coffee intake may be beneficial. A 2013 meta-analysis summarized the results of 36 studies with more than a million participants and found a U-shaped relationship between coffee intake and cardiovascular risk. The sweet spot was at three to five cups a day; people drinking that much coffee had about a 15% reduced risk for cardiovascular disease compared with nondrinkers.
But here’s the thing. Coffee contains caffeine, but it is much more than that. It is a heady brew of various chemicals and compounds, phenols, and chlorogenic acids. And, of course, you can get caffeine from stuff that isn’t coffee — natural things like tea — and decidedly unnatural things like energy drinks. How do you figure out where the benefit really lies?
The JCEM study leveraged the impressive UK Biobank dataset to figure this out. The Biobank recruited more than half a million people from the UK between 2006 and 2010 and collected a wealth of data from each of them: surveys, blood samples, biometrics, medical imaging — the works. And then they followed what would happen to those people medically over time. It’s a pretty amazing resource.
But for the purposes of this study, what you need to know is that just under 200,000 of those participants met the key criteria for this study: being free from cardiovascular disease at baseline; having completed a detailed survey about their coffee, tea, and other caffeinated beverage intake; and having adequate follow-up. A subset of that number, just under 100,000, had metabolomic data — which is where this study really gets interesting.
We’ll dive into the metabolome in a moment, but first let’s just talk about the main finding, the relationship between coffee, tea, or caffeine and cardiovascular disease. But to do that, we need to acknowledge that people who drink a lot of coffee are different from people who don’t, and it might be those differences, not the coffee itself, that are beneficial.
What were those differences? People who drank more coffee tended to be a bit older, were less likely to be female, and were slightly more likely to engage in physical activity. They ate less processed meat but also fewer vegetables. Some of those factors, like being female, are generally protective against cardiovascular disease; but some, like age, are definitely not. The authors adjusted for these and multiple other factors, including alcohol intake, BMI, kidney function, and many others to try to disentangle the effect of being the type of person who drinks a lot of coffee from the drinking a lot of coffee itself.
These are the results of the fully adjusted model. Compared with nonconsumers, you can see that people in the higher range of coffee, tea, or just caffeine intake have almost a 40% reduction in cardiovascular disease in follow-up.
Looking at the benefit across the spectrum of intake, you again see that U-shaped curve, suggesting that a sweet spot for daily consumption can be found around 3 cups of coffee or tea (or 250 mg of caffeine). A standard energy drink contains about 120 mg of caffeine. 
But if this is true, it would be good to know why. To figure that out, the authors turned to the metabolome. The idea here is that your body is constantly breaking stuff down, taking all these proteins and chemicals and compounds that we ingest and turning them into metabolites. Using advanced measurement techniques, researchers can measure hundreds or even thousands of metabolites from a single blood sample. They provide information, obviously, about the food you eat and the drinks you drink, but what is really intriguing is that some metabolites are associated with better health and some with worse
In this study, researchers measured 168 individual metabolites. Eighty of them, nearly half, were significantly altered in people who drank more coffee.
This figure summarizes the findings, and yes, this is way too complicated. 
But here’s how to interpret it. The inner ring shows you how certain metabolites are associated with cardiovascular disease. The outer rings show you how those metabolites are associated with coffee, tea, or caffeine. The interesting part is that the sections of the ring (outer rings and inner rings) are very different colors.
Like here.
What you see here is a fairly profound effect that coffee, tea, or caffeine intake has on metabolites of VLDL — bad cholesterol. The beverages lower it, and, of course, higher levels lead to cardiovascular disease. This means that this is a potential causal pathway from coffee intake to heart protection.
And that’s not the only one.
You see a similar relationship for saturated fatty acids. Higher levels lead to cardiovascular disease, and coffee intake lowers levels. The reverse works too: Lower levels of histidine (an amino acid) increase cardiovascular risk, and coffee seems to raise those levels.
Is this all too good to be true? It’s hard to say. The data on coffee’s benefits have been remarkably consistent. Still, I wouldn’t be a good doctor if I didn’t mention that clearly there is a difference between a cup of black coffee and a venti caramel Frappuccino.
Nevertheless, coffee remains firmly in my holy trinity of enjoyable things that are, for whatever reason, still good for you. So, when you’re having that second, or third, or maybe fourth cup of the day, you can take that to heart.
Dr. Wilson, associate professor of medicine and public health and director of Yale’s Clinical and Translational Research Accelerator, reported no conflicts of interest.
A version of this article first appeared on Medscape.com.
Can AI Improve Cardiomyopathy Detection in Pregnant Women?
TOPLINE:
Artificial intelligence (AI)–guided screening using digital stethoscopes doubled the detection of left ventricular systolic dysfunction (LVSD) in pregnant and postpartum women in Nigeria. Cardiomyopathy during pregnancy and post partum is challenging to diagnose because of symptom overlap with normal pregnancy changes. AI-guided screening showed a significant improvement in diagnosis rates, compared with usual care.
METHODOLOGY:
- Researchers conducted an open-label, randomized clinical trial involving 1232 pregnant and postpartum women in Nigeria.
- Participants were randomized to either AI-guided screening using digital stethoscopes and 12-lead ECGs or usual care.
- The primary outcome was the identification of LVSD confirmed by echocardiography.
- Secondary outcomes were AI model performance across subgroups and the effectiveness of AI in identifying various levels of LVSD.
TAKEAWAY:
- AI-guided screening using digital stethoscopes detected LVSD in 4.1% of participants, compared with 2.0% of controls (P = .032).
- The 12-lead AI-ECG model detected LVSD in 3.4% of participants in the intervention arm, compared with 2.0% of those in the control arm (P = .125).
- No serious adverse events related to study participation were reported.
- The study highlighted the potential of AI-guided screening to improve the diagnosis of pregnancy-related cardiomyopathy.
IN PRACTICE:
“Delays in the diagnosis of cardiomyopathy during the peripartum period is associated with poorer outcomes as such, it is imperative that we are able to identify cardiac dysfunction early so that appropriate care can be initiated to reduce associated adverse maternal and infant outcomes,” wrote the authors of the study.
SOURCE:
This study was led by Demilade A. Adedinsewo, MBchB, Mayo Clinic in Jacksonville, Florida. It was published online in Nature Medicine.
LIMITATIONS:
The study’s pragmatic design and enrollment at teaching hospitals with echocardiography capabilities limited generalizability. Two thirds of participants were in the third trimester or postpartum at study entry, which limited follow-up visits. The study did not require completion of all seven visits, which led to potential attrition bias. The selected cutoff for LVSD (left ventricular ejection fraction < 50%) did not match the original model specifications, which potentially affected results.
DISCLOSURES:
Dr. Adedinsewo disclosed receiving grants from the Mayo Clinic BIRCWH program funded by the National Institutes of Health. Two coauthors reported holding patents for AI algorithms licensed to Anumana, AliveCor, and Eko Health. Additional disclosures are noted in the original article.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
TOPLINE:
Artificial intelligence (AI)–guided screening using digital stethoscopes doubled the detection of left ventricular systolic dysfunction (LVSD) in pregnant and postpartum women in Nigeria. Cardiomyopathy during pregnancy and post partum is challenging to diagnose because of symptom overlap with normal pregnancy changes. AI-guided screening showed a significant improvement in diagnosis rates, compared with usual care.
METHODOLOGY:
- Researchers conducted an open-label, randomized clinical trial involving 1232 pregnant and postpartum women in Nigeria.
- Participants were randomized to either AI-guided screening using digital stethoscopes and 12-lead ECGs or usual care.
- The primary outcome was the identification of LVSD confirmed by echocardiography.
- Secondary outcomes were AI model performance across subgroups and the effectiveness of AI in identifying various levels of LVSD.
TAKEAWAY:
- AI-guided screening using digital stethoscopes detected LVSD in 4.1% of participants, compared with 2.0% of controls (P = .032).
- The 12-lead AI-ECG model detected LVSD in 3.4% of participants in the intervention arm, compared with 2.0% of those in the control arm (P = .125).
- No serious adverse events related to study participation were reported.
- The study highlighted the potential of AI-guided screening to improve the diagnosis of pregnancy-related cardiomyopathy.
IN PRACTICE:
“Delays in the diagnosis of cardiomyopathy during the peripartum period is associated with poorer outcomes as such, it is imperative that we are able to identify cardiac dysfunction early so that appropriate care can be initiated to reduce associated adverse maternal and infant outcomes,” wrote the authors of the study.
SOURCE:
This study was led by Demilade A. Adedinsewo, MBchB, Mayo Clinic in Jacksonville, Florida. It was published online in Nature Medicine.
LIMITATIONS:
The study’s pragmatic design and enrollment at teaching hospitals with echocardiography capabilities limited generalizability. Two thirds of participants were in the third trimester or postpartum at study entry, which limited follow-up visits. The study did not require completion of all seven visits, which led to potential attrition bias. The selected cutoff for LVSD (left ventricular ejection fraction < 50%) did not match the original model specifications, which potentially affected results.
DISCLOSURES:
Dr. Adedinsewo disclosed receiving grants from the Mayo Clinic BIRCWH program funded by the National Institutes of Health. Two coauthors reported holding patents for AI algorithms licensed to Anumana, AliveCor, and Eko Health. Additional disclosures are noted in the original article.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
TOPLINE:
Artificial intelligence (AI)–guided screening using digital stethoscopes doubled the detection of left ventricular systolic dysfunction (LVSD) in pregnant and postpartum women in Nigeria. Cardiomyopathy during pregnancy and post partum is challenging to diagnose because of symptom overlap with normal pregnancy changes. AI-guided screening showed a significant improvement in diagnosis rates, compared with usual care.
METHODOLOGY:
- Researchers conducted an open-label, randomized clinical trial involving 1232 pregnant and postpartum women in Nigeria.
- Participants were randomized to either AI-guided screening using digital stethoscopes and 12-lead ECGs or usual care.
- The primary outcome was the identification of LVSD confirmed by echocardiography.
- Secondary outcomes were AI model performance across subgroups and the effectiveness of AI in identifying various levels of LVSD.
TAKEAWAY:
- AI-guided screening using digital stethoscopes detected LVSD in 4.1% of participants, compared with 2.0% of controls (P = .032).
- The 12-lead AI-ECG model detected LVSD in 3.4% of participants in the intervention arm, compared with 2.0% of those in the control arm (P = .125).
- No serious adverse events related to study participation were reported.
- The study highlighted the potential of AI-guided screening to improve the diagnosis of pregnancy-related cardiomyopathy.
IN PRACTICE:
“Delays in the diagnosis of cardiomyopathy during the peripartum period is associated with poorer outcomes as such, it is imperative that we are able to identify cardiac dysfunction early so that appropriate care can be initiated to reduce associated adverse maternal and infant outcomes,” wrote the authors of the study.
SOURCE:
This study was led by Demilade A. Adedinsewo, MBchB, Mayo Clinic in Jacksonville, Florida. It was published online in Nature Medicine.
LIMITATIONS:
The study’s pragmatic design and enrollment at teaching hospitals with echocardiography capabilities limited generalizability. Two thirds of participants were in the third trimester or postpartum at study entry, which limited follow-up visits. The study did not require completion of all seven visits, which led to potential attrition bias. The selected cutoff for LVSD (left ventricular ejection fraction < 50%) did not match the original model specifications, which potentially affected results.
DISCLOSURES:
Dr. Adedinsewo disclosed receiving grants from the Mayo Clinic BIRCWH program funded by the National Institutes of Health. Two coauthors reported holding patents for AI algorithms licensed to Anumana, AliveCor, and Eko Health. Additional disclosures are noted in the original article.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.