User login
VA Home Telehealth Program for Initiating and Optimizing Heart Failure Guideline-Directed Medical Therapy
Heart failure (HF) is a chronic, progressive condition that is characterized by the heart’s inability to effectively pump blood throughout the body. In 2018, approximately 6.2 million US adults had HF, and 13.4% of all death certificates noted HF as a precipitating factor.1 Patients not receiving appropriate guideline-directed medical therapy (GDMT) face a 29% excess mortality risk over a 2-year period.2 Each additional GDMT included in a patient’s regimen significantly reduces all-cause mortality.3
The Change the Management of Patients with Heart Failure (CHAMP) registry reports that only about 1% of patients with HF are prescribed 3 agents from contemporary GDMT at target doses, highlighting the need for optimizing clinicians’ approaches to GDMT.4 Similarly, The Get With The Guidelines Heart Failure Registry has noted that only 20.2% of patients with HF with reduced ejection fraction (HFrEF) are prescribed a sodium-glucose cotransporter 2 inhibitor (SGLT2i) following hospital discharge for HFrEF exacerbation.5 Overall, treatment rates with GDMT saw limited improvement between 2013 and 2019, with no significant difference between groups in mortality, indicating the need for optimized methods to encourage the initiation of GDMT.6
Remote monitoring and telecare are novel ways to improve GDMT rates in those with HFrEF. However, data are inconsistent regarding the impact of remote HF monitoring and improvements in GDMT or HF-related outcomes.6-10 The modalities of remote monitoring for GDMT vary among studies, but the potential for telehealth monitoring to improve GDMT, thereby potentially reducing HF-related hospitalizations, is clear.
Telemonitoring has demonstrated improved participant adherence with weight monitoring, although the withdrawal rate was high, and has the potential to reduce all-cause mortality and HF-related hospitalizations.11,12 Telemonitoring for GDMT optimization led to an increase in the proportion of patients who achieved optimal GDMT doses, a decrease in the time to dose optimization, and a reduction in the number of clinic visits.13 Remote GDMT titration was accomplished in the general patient population with HFrEF; however, in populations already followed by cardiologists or HF specialists, remote optimization strategies did not yield different proportions of GDMT use.14 The aim of this study was to assess the impact of the home telehealth (HT) monitoring program on the initiation and optimization of HF GDMT among veterans with HFrEF at the Veterans Affairs Ann Arbor Healthcare System (VAAAHS) in Michigan.
Methods
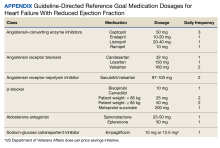
This was a single-center retrospective study of Computerized Patient Record System (CPRS)data. Patients at the VAAAHS were evaluated if they were diagnosed with HFrEF and were eligible for enrollment in the HT monitoring program. Eligibility criteria included a diagnosis of stage C HF, irrespective of EF, and a history of any HF-related hospitalization. We focused on patients with HFrEF due to stronger guideline-based recommendations for certain pharmacotherapies as compared with HF with mildly reduced ejection fraction (HFmrEF) and preserved ejection fraction (HFpEF). Initial patient data for HT enrolling were accessed using the Heart Failure Dashboard via the US Department of Veterans Affairs (VA) Academic Detailing Service. The target daily doses of typical agents used in HFrEF GDMT are listed in the Appendix.
The HT program is an embedded model in which HT nurses receive remote data from the patient and triage that with the VAAAHS cardiology team. Patients’ questions, concerns, and/or vital signs are recovered remotely. In this model, nurses are embedded in the cardiology team, working with the cardiologists, cardiology clinical pharmacist, and/or cardiology nurse practitioners to make medication interventions. Data are recorded with an HT device, including weight, blood pressure (BP), heart rate, and pulse oximetry. HT nurses are also available to the patient via phone or video. The program uses a 180-day disease management protocol for HF via remote device, enabling the patient to answer questions and receive education on their disease daily. Responses to questions and data are then reviewed by an HT nurse remotely during business hours and triaged as appropriate with the cardiology team. Data can be communicated to the cardiology team via the patient record, eliminating the need for the cardiology team to use the proprietary portal affiliated with the HT device.
Study Sample
Patient information was obtained from a list of 417 patients eligible for enrollment in the HT program; the list was sent to the HT program for review and enrollment. Patient data were extracted from the VAAAHS HF Dashboard and included all patients with HFrEF and available data on the platform. The sample for the retrospective chart review included 40 adults who had HFrEF, defined as a left ventricular EF (LVEF) of ≤ 40% as evidenced by a transthoracic echocardiogram or cardiac magnetic resonance imaging. These patients were contacted and agreed to enroll in the HT monitoring program. The HT program population was compared against a control group of 33 patients who were ineligible for the HT program. Patients were deemed ineligible for HT if they resided in a nursing home, lacked a VAAAHS primary care clinician, or declined participation in the HT program.
Procedures
Patients who declined participation in the HT program followed the standard of care, which was limited to visits with primary care clinicians and/or cardiologists as per the follow-up plan. Patient data were collected over 12 months. The study was approved by the VAAAHS Institutional Review Board (reference number, 1703034), Research and Development Committee, and Research Administration.
Primary and Secondary Goals
The primary goal of the study was to assess the impact of the HT program on drug interventions, specifically initiating and titrating HFrEF pharmacotherapies. Interventions were based on GDMT with known mortality- and morbidity-reducing properties when used at their maximum tolerated doses, including angiotensin-converting enzyme inhibitors (ACEi), angiotensin receptor-neprilysin inhibitor (ARNi), or angiotensin receptor blockers (ARB), with a preference for ARNi, β-blockers for HFrEF (metoprolol succinate, bisoprolol, or carvedilol), aldosterone antagonists, and SGLT2is.
Secondary goals included HF-related hospitalizations, medication adherence, time to enrollment in HT, time to laboratory analysis after the initiation or titration of an ACEi/ARB/ARNi or aldosterone antagonist, and time enrolled in the HT program. Patients were considered adherent if their drug refill history showed consistent fills of their medications. The χ2 test was used for total interventions made during the study period and Fisher exact test for all others.
Results
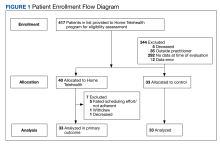
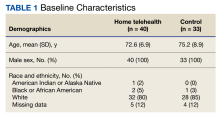
Patient data were collected between July 2022 and June 2023. All 73 patients were male, and the mean age in the HT group (n = 40) was 72.6 years and 75.2 years for the control group (n = 33). Overall, the baseline demographics were similar between the groups (Table 1). Of 40 patients screened for enrollment in the HT program, 33 were included in the analysis (Figure 1).
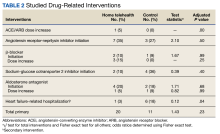
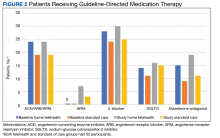
At baseline, the HT group included more individuals than the control group on ACEi/ARB/ARNi (24 vs 19, respectively), β-blocker (28 vs 24, respectively), SGLT2i (14 vs 11, respectively), and aldosterone antagonist (15 vs 9, respectively) (Figure 2). There were 20 interventions made in the HT group compared with 11 therapy changes in the control arm during the study (odds ratio, 1.43; P = .23) (Table 2). In the HT group, 1 patient achieved an ACEi target dose, 3 patients achieved a β-blocker target dose, and 7 achieved a target dose of spironolactone (titration is not required for SGLT2i therapy and is counted as target dose). In the HT group, 17 patients were on ≥ 3 recommended agents, while 9 patients were taking 4 agents. Seven of 20 HT group interventions resulted in titration to the target dose. In the control group, no patients achieved an ARNi target dose, 3 patients achieved a β-blocker target dose, and 2 patients achieved a spironolactone antagonist target dose. In the control arm, 7 patients were on ≥ 3 GDMTs, and 2 were taking 4 agents. No patient in either group achieved a target dose of 4 agents. Five of 11 control group interventions resulted in initiation or titration of GDMT to the target dose.
Of the 40 HT group patients, 7 were excluded from analysis (3 failed to schedule HT, 1 was at a long-term care facility, 1 was nonadherent, 1 declined participation, and 1 died) and 33 remained in the program for a mean (SD) 5.3 (3.5) months. Death rates were tracked during the study: 1 patient died in the HT group and 3 in the control group.
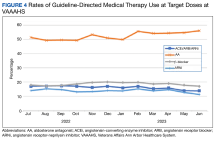
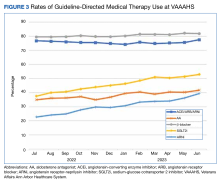
We analyzed the overall percentage of VAAAHS patients with HFrEF who were on appropriate GDMT. Given the ongoing drive to improve HF-related outcomes, HT interventions could not be compared to a static population, so the HT and control patients were compared with the rates of GDMT at VAAAHS, which was available in the Academic Detailing Service Heart Failure Dashboard (Figure 3). Titration and optimization rates were also compared (Figure 4). From July 2022 to June 2023, ARNi use increased by 16.6%, aldosterone antagonist by 6.8%, and β-blockers by 2.4%. Target doses of GDMTs were more difficult to achieve in the hospital system. There was an increase in aldosterone antagonist target dose achievement by 4.7%, but overall there were decreases in target doses in other GDMTs: ACEi/ARB/ARNi target dose use decreased by 3.2%, ARNi target dose use decreased by 2.7% and target β-blocker use decreased by 0.9%.
Discussion
Telehealth yielded clinically important interventions, with some titrations bringing patients to their target doses of medications for HFrEF. The 20 interventions made in the HT group can be largely attributed to the nurses’ efforts to alert clinicians to drug titrations or ACEi/ARB to ARNi transitions. Although the findings were not statistically significant, the difference in the number of drug therapy changes supports the use of the HT program for a GDMT optimization strategy. Patients may be difficult to titrate secondary to adverse effects that make medication initiation or titration inappropriate, such as hypotension and hyperkalemia, although this was not observed in this small sample size. Considering a mean HT enrollment of 5.3 months, many patients had adequate disease assessment and medication titration. Given that patients are discharged from the service once deemed appropriate, this decreases the burden on the patient and increases the utility and implementation of the HT program for other patients.
A surprising finding of this study was the lower rate of HF-related hospitalizations in the HT group. Although not the primary subject of interest in the study, it reinforced the importance of close health care professional follow-up for patients living with HF. Telehealth may improve communication and shared decision making over medication use. Because the finding was unanticipated, the rate of diuretic adjustments was not tracked.
Patients were reevaluated every 6 months for willingness to continue the program, adherence, and clinical needs. These results are similar to those of other trials that demonstrated improved rates of GDMT in the setting of pharmacist- or nurse-led HF treatment optimization.15,16 These positive results differ from other trials incorporating remote monitoring regarding patient continuation in HT programs. For example, in a study by Ding and colleagues, the withdrawal rate from their monitoring service was about 22%, while in our study only 1 patient withdrew from the HT program.11
The HT program resulted in fewer hospitalizations than the control arm. There were 6 HF-related hospitalizations in the control group, although 5 involved a single patient. Typically, such a patient would be encouraged to follow HT monitoring after just 1 HF-related hospitalization; however, the patient declined to participate.
Early optimization of GDMT in patients who were recently discharged from the hospital for an HF-related hospitalization yields a reduction in hospital rehospitalization.17 GDMT optimization has unequivocal benefits in HF outcomes. Unfortunately, the issues surrounding methodologies on how to best optimize GDMT are lacking. While HT has been found to be feasible in the aid of optimizing medical therapy, the TIM-HF trial concluded that remote monitoring services had no significant benefit in reducing mortality.7,8 On the other hand, the OptiLink HF Study showed that when clinicians respond to remote monitoring prompts from fluid index threshold crossing alerts, these interventions are associated with significantly improved clinical outcomes in patients with implantable cardioverter-defibrillators and advanced HF.9 In contrast to previous trials, the AMULET trial showed that remote monitoring compared with standard care significantly reduced the risk of HF hospitalization or cardiovascular death during the 12-month follow-up among patients with HF and LVEF ≤ 49% after an episode of acute exacerbation.10 Additionally, patients who received skilled home health services and participated in remote monitoring for their chronic HF experienced a reduction in all-cause 30-day readmission.18
Given the contrasting evidence regarding remote monitoring and variable modalities of implementing interventions, we investigated whether HT monitoring yields improvements in GDMT optimization. We found that HT nurses were able to nearly double the rate of interventions for patients with HFrEF. The HT program in providing expanded services will require adequate staffing responsibilities and support. The HT program is geared toward following a large, diverse patient population, such as those with chronic obstructive pulmonary disease, hypertension, and HF. We only evaluated services for patients with HFrEF, but the program also follows patients with HfmrEF and HfpEF. These patients were not included as GDMT optimization differs for patients with an LVEF > 40%.19,20
The lower rates of achieving target doses of GDMTs were likely obstructed by continuous use of initial drug doses and further limited by lack of follow-up. When compared with the rest of the VAAAHS, there was a greater effort to increase ARNi use in the HT group as 7 of 33 patients (21%) were started on ARNi compared with a background increase of ARNi use of 17%. There was a lower mortality rate observed in the HT group compared with the control group. One patient in each group died of unrelated causes, while 2 deaths in the control group were due to worsening HF. The difference in mortality is likely multifactorial, possibly related to the control group’s greater disease burden or higher mean age (75.2 years vs 72.6 years).
Limitations
This was an observational cohort design, which is subject to bias. Thus, the findings of this study are entirely hypothesis-generating and a randomized controlled trial would be necessary for clearer results. Second, low numbers of participants may have skewed the data set. Given the observational nature of the study, this nonetheless is a positive finding to support the HT program for assisting with HF monitoring and prompting drug interventions. Due to the low number of participants, a single patient may have skewed the results with 5 hospitalizations.
Conclusions
This pilot study demonstrates the applicability of HT monitoring to optimize veteran HFrEF GDMT. The HT program yielded numerically relevant interventions and fewer HF-related hospitalizations compared with the control arm. The study shows the feasibility of the program to safely optimize GDMT toward their target doses and may serve as an additional catalyst to further develop HT programs specifically targeted toward HF monitoring and management. Cost-savings analyses would likely need to demonstrate the cost utility of such a service.
We thank the home telehealth nursing staff for their assistance in data collection and enrollment of patients into the monitoring program.
1. Tsao CW, Aday AW, Almarzooq ZI, et al. Heart disease and stroke statistics-2022 update: a report from the American Heart Association. Circulation. 2022;145(8):e153-e639. doi:10.1161/CIR.0000000000001052
2. McCullough PA, Mehta HS, Barker CM, et al. Mortality and guideline-directed medical therapy in real-world heart failure patients with reduced ejection fraction. Clin Cardiol. 2021;44(9):1192-1198. doi:10.1002/clc.23664
3. Tromp J, Ouwerkerk W, van Veldhuisen DJ, et al. A systematic review and network meta-analysis of pharmacological treatment of heart failure with reduced ejection fraction. JACC Heart Fail. 2022;10(2):73-84. doi:10.1016/j.jchf.2021.09.004
4. Greene SJ, Butler J, Albert NM, et al. Medical therapy for heart failure with reduced ejection fraction: the CHAMP-HF Registry. J Am Coll Cardiol. 2018;72(4):351-366. doi:10.1016/j.jacc.2018.04.070
5. Pierce JB, Vaduganathan M, Fonarow GC, et al. Contemporary use of sodium-glucose cotransporter-2 inhibitor therapy among patients hospitalized for heart failure with reduced ejection fraction in the US: The Get With The Guidelines-Heart Failure Registry. JAMA Cardiol. 2023;8(7):652-661. doi:10.1001/jamacardio.2023.1266
6. Sandhu AT, Kohsaka S, Turakhia MP, Lewis EF, Heidenreich PA. Evaluation of quality of care for US veterans with recent-onset heart failure with reduced ejection fraction. JAMA Cardiol. 2022;7(2):130-139. doi:10.1001/jamacardio.2021.4585 7. Rahimi K, Nazarzadeh M, Pinho-Gomes AC, et al. Home monitoring with technology-supported management in chronic heart failure: a randomised trial. Heart. 2020;106(20):1573-1578. doi:10.1136/heartjnl-2020-316773 8. Koehler F, Winkler S, Schieber M, et al. Impact of remote telemedical management on mortality and hospitalizations in ambulatory patients with chronic heart failure: the telemedical interventional monitoring in heart failure study. Circulation. 2011;123(17):1873-1880. doi:10.1161/CIRCULATIONAHA.111.018473
9. Wintrich J, Pavlicek V, Brachmann J, et al. Remote monitoring with appropriate reaction to alerts was associated with improved outcomes in chronic heart failure: results from the OptiLink HF study. Circ Arrhythm Electrophysiol. 2021;14(1):e008693. doi:10.1161/CIRCEP.120.008693
10. Krzesinski P, Jankowska EA, Siebert J, et al. Effects of an outpatient intervention comprising nurse-led non-invasive assessments, telemedicine support and remote cardiologists’ decisions in patients with heart failure (AMULET study): a randomised controlled trial. Eur J Heart Fail. 2022;24(3):565-577. doi:10.1002/ejhf.2358
11. Ding H, Jayasena R, Chen SH, et al. The effects of telemonitoring on patient compliance with self-management recommendations and outcomes of the innovative telemonitoring enhanced care program for chronic heart failure: randomized controlled trial. J Med Internet Res. 2020;22(7):e17559. doi:10.2196/17559
12. Kitsiou S, Pare G, Jaana M. Effects of home telemonitoring interventions on patients with chronic heart failure: an overview of systematic reviews. J Med Internet Res. 2015;17(3):e63. doi:10.2196/jmir.4174
13. Artanian V, Ross HJ, Rac VE, O’Sullivan M, Brahmbhatt DH, Seto E. Impact of remote titration combined with telemonitoring on the optimization of guideline-directed medical therapy for patients with heart failure: internal pilot of a randomized controlled trial. JMIR Cardio. 2020;4(1):e21962. doi:10.2196/21962
14. Desai AS, Maclean T, Blood AJ, et al. Remote optimization of guideline-directed medical therapy in patients with heart failure with reduced ejection fraction. JAMA Cardiol. 2020;5(12):1430-1434. doi:10.1001/jamacardio.2020.3757
15. Patil T, Ali S, Kaur A, et al. Impact of pharmacist-led heart failure clinic on optimization of guideline-directed medical therapy (PHARM-HF). J Cardiovasc Transl Res. 2022;15(6):1424-1435. doi:10.1007/s12265-022-10262-9
16. Zheng J, Mednick T, Heidenreich PA, Sandhu AT. Pharmacist- and nurse-led medical optimization in heart failure: a systematic review and meta-analysis. J Card Fail. 2023;29(7):1000-1013. doi:10.1016/j.cardfail.2023.03.012
17. Mebazaa A, Davison B, Chioncel O, et al. Safety, tolerability and efficacy of up-titration of guideline-directed medical therapies for acute heart failure (STRONG-HF): a multinational, open-label, randomised, trial. Lancet. 2022;400(10367):1938-1952. doi:10.1016/S0140-6736(22)02076-1
18. O’Connor M, Asdornwised U, Dempsey ML, et al. Using telehealth to reduce all-cause 30-day hospital readmissions among heart failure patients receiving skilled home health services. Appl Clin Inform. 2016;7(2):238-47. doi:10.4338/ACI-2015-11-SOA-0157
19. Heidenreich PA, Bozkurt B, Aguilar D, et al. 2022 AHA/ACC/HFSA Guideline for the Management of Heart Failure: Executive Summary: A Report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. Circulation. 2022;145(18):e876-e894. doi:10.1161/CIR.0000000000001062
20. Kittleson MM, Panjrath GS, Amancherla K, et al. 2023 ACC Expert Consensus Decision Pathway on Management of Heart Failure With Preserved Ejection Fraction: A Report of the American College of Cardiology Solution Set Oversight Committee. J Am Coll Cardiol. 2023;81(18):1835-1878. doi:10.1016/j.jacc.2023.03.393
Heart failure (HF) is a chronic, progressive condition that is characterized by the heart’s inability to effectively pump blood throughout the body. In 2018, approximately 6.2 million US adults had HF, and 13.4% of all death certificates noted HF as a precipitating factor.1 Patients not receiving appropriate guideline-directed medical therapy (GDMT) face a 29% excess mortality risk over a 2-year period.2 Each additional GDMT included in a patient’s regimen significantly reduces all-cause mortality.3
The Change the Management of Patients with Heart Failure (CHAMP) registry reports that only about 1% of patients with HF are prescribed 3 agents from contemporary GDMT at target doses, highlighting the need for optimizing clinicians’ approaches to GDMT.4 Similarly, The Get With The Guidelines Heart Failure Registry has noted that only 20.2% of patients with HF with reduced ejection fraction (HFrEF) are prescribed a sodium-glucose cotransporter 2 inhibitor (SGLT2i) following hospital discharge for HFrEF exacerbation.5 Overall, treatment rates with GDMT saw limited improvement between 2013 and 2019, with no significant difference between groups in mortality, indicating the need for optimized methods to encourage the initiation of GDMT.6
Remote monitoring and telecare are novel ways to improve GDMT rates in those with HFrEF. However, data are inconsistent regarding the impact of remote HF monitoring and improvements in GDMT or HF-related outcomes.6-10 The modalities of remote monitoring for GDMT vary among studies, but the potential for telehealth monitoring to improve GDMT, thereby potentially reducing HF-related hospitalizations, is clear.
Telemonitoring has demonstrated improved participant adherence with weight monitoring, although the withdrawal rate was high, and has the potential to reduce all-cause mortality and HF-related hospitalizations.11,12 Telemonitoring for GDMT optimization led to an increase in the proportion of patients who achieved optimal GDMT doses, a decrease in the time to dose optimization, and a reduction in the number of clinic visits.13 Remote GDMT titration was accomplished in the general patient population with HFrEF; however, in populations already followed by cardiologists or HF specialists, remote optimization strategies did not yield different proportions of GDMT use.14 The aim of this study was to assess the impact of the home telehealth (HT) monitoring program on the initiation and optimization of HF GDMT among veterans with HFrEF at the Veterans Affairs Ann Arbor Healthcare System (VAAAHS) in Michigan.
Methods
This was a single-center retrospective study of Computerized Patient Record System (CPRS)data. Patients at the VAAAHS were evaluated if they were diagnosed with HFrEF and were eligible for enrollment in the HT monitoring program. Eligibility criteria included a diagnosis of stage C HF, irrespective of EF, and a history of any HF-related hospitalization. We focused on patients with HFrEF due to stronger guideline-based recommendations for certain pharmacotherapies as compared with HF with mildly reduced ejection fraction (HFmrEF) and preserved ejection fraction (HFpEF). Initial patient data for HT enrolling were accessed using the Heart Failure Dashboard via the US Department of Veterans Affairs (VA) Academic Detailing Service. The target daily doses of typical agents used in HFrEF GDMT are listed in the Appendix.
The HT program is an embedded model in which HT nurses receive remote data from the patient and triage that with the VAAAHS cardiology team. Patients’ questions, concerns, and/or vital signs are recovered remotely. In this model, nurses are embedded in the cardiology team, working with the cardiologists, cardiology clinical pharmacist, and/or cardiology nurse practitioners to make medication interventions. Data are recorded with an HT device, including weight, blood pressure (BP), heart rate, and pulse oximetry. HT nurses are also available to the patient via phone or video. The program uses a 180-day disease management protocol for HF via remote device, enabling the patient to answer questions and receive education on their disease daily. Responses to questions and data are then reviewed by an HT nurse remotely during business hours and triaged as appropriate with the cardiology team. Data can be communicated to the cardiology team via the patient record, eliminating the need for the cardiology team to use the proprietary portal affiliated with the HT device.
Study Sample
Patient information was obtained from a list of 417 patients eligible for enrollment in the HT program; the list was sent to the HT program for review and enrollment. Patient data were extracted from the VAAAHS HF Dashboard and included all patients with HFrEF and available data on the platform. The sample for the retrospective chart review included 40 adults who had HFrEF, defined as a left ventricular EF (LVEF) of ≤ 40% as evidenced by a transthoracic echocardiogram or cardiac magnetic resonance imaging. These patients were contacted and agreed to enroll in the HT monitoring program. The HT program population was compared against a control group of 33 patients who were ineligible for the HT program. Patients were deemed ineligible for HT if they resided in a nursing home, lacked a VAAAHS primary care clinician, or declined participation in the HT program.
Procedures
Patients who declined participation in the HT program followed the standard of care, which was limited to visits with primary care clinicians and/or cardiologists as per the follow-up plan. Patient data were collected over 12 months. The study was approved by the VAAAHS Institutional Review Board (reference number, 1703034), Research and Development Committee, and Research Administration.
Primary and Secondary Goals
The primary goal of the study was to assess the impact of the HT program on drug interventions, specifically initiating and titrating HFrEF pharmacotherapies. Interventions were based on GDMT with known mortality- and morbidity-reducing properties when used at their maximum tolerated doses, including angiotensin-converting enzyme inhibitors (ACEi), angiotensin receptor-neprilysin inhibitor (ARNi), or angiotensin receptor blockers (ARB), with a preference for ARNi, β-blockers for HFrEF (metoprolol succinate, bisoprolol, or carvedilol), aldosterone antagonists, and SGLT2is.
Secondary goals included HF-related hospitalizations, medication adherence, time to enrollment in HT, time to laboratory analysis after the initiation or titration of an ACEi/ARB/ARNi or aldosterone antagonist, and time enrolled in the HT program. Patients were considered adherent if their drug refill history showed consistent fills of their medications. The χ2 test was used for total interventions made during the study period and Fisher exact test for all others.
Results
Patient data were collected between July 2022 and June 2023. All 73 patients were male, and the mean age in the HT group (n = 40) was 72.6 years and 75.2 years for the control group (n = 33). Overall, the baseline demographics were similar between the groups (Table 1). Of 40 patients screened for enrollment in the HT program, 33 were included in the analysis (Figure 1).
At baseline, the HT group included more individuals than the control group on ACEi/ARB/ARNi (24 vs 19, respectively), β-blocker (28 vs 24, respectively), SGLT2i (14 vs 11, respectively), and aldosterone antagonist (15 vs 9, respectively) (Figure 2). There were 20 interventions made in the HT group compared with 11 therapy changes in the control arm during the study (odds ratio, 1.43; P = .23) (Table 2). In the HT group, 1 patient achieved an ACEi target dose, 3 patients achieved a β-blocker target dose, and 7 achieved a target dose of spironolactone (titration is not required for SGLT2i therapy and is counted as target dose). In the HT group, 17 patients were on ≥ 3 recommended agents, while 9 patients were taking 4 agents. Seven of 20 HT group interventions resulted in titration to the target dose. In the control group, no patients achieved an ARNi target dose, 3 patients achieved a β-blocker target dose, and 2 patients achieved a spironolactone antagonist target dose. In the control arm, 7 patients were on ≥ 3 GDMTs, and 2 were taking 4 agents. No patient in either group achieved a target dose of 4 agents. Five of 11 control group interventions resulted in initiation or titration of GDMT to the target dose.
Of the 40 HT group patients, 7 were excluded from analysis (3 failed to schedule HT, 1 was at a long-term care facility, 1 was nonadherent, 1 declined participation, and 1 died) and 33 remained in the program for a mean (SD) 5.3 (3.5) months. Death rates were tracked during the study: 1 patient died in the HT group and 3 in the control group.
We analyzed the overall percentage of VAAAHS patients with HFrEF who were on appropriate GDMT. Given the ongoing drive to improve HF-related outcomes, HT interventions could not be compared to a static population, so the HT and control patients were compared with the rates of GDMT at VAAAHS, which was available in the Academic Detailing Service Heart Failure Dashboard (Figure 3). Titration and optimization rates were also compared (Figure 4). From July 2022 to June 2023, ARNi use increased by 16.6%, aldosterone antagonist by 6.8%, and β-blockers by 2.4%. Target doses of GDMTs were more difficult to achieve in the hospital system. There was an increase in aldosterone antagonist target dose achievement by 4.7%, but overall there were decreases in target doses in other GDMTs: ACEi/ARB/ARNi target dose use decreased by 3.2%, ARNi target dose use decreased by 2.7% and target β-blocker use decreased by 0.9%.
Discussion
Telehealth yielded clinically important interventions, with some titrations bringing patients to their target doses of medications for HFrEF. The 20 interventions made in the HT group can be largely attributed to the nurses’ efforts to alert clinicians to drug titrations or ACEi/ARB to ARNi transitions. Although the findings were not statistically significant, the difference in the number of drug therapy changes supports the use of the HT program for a GDMT optimization strategy. Patients may be difficult to titrate secondary to adverse effects that make medication initiation or titration inappropriate, such as hypotension and hyperkalemia, although this was not observed in this small sample size. Considering a mean HT enrollment of 5.3 months, many patients had adequate disease assessment and medication titration. Given that patients are discharged from the service once deemed appropriate, this decreases the burden on the patient and increases the utility and implementation of the HT program for other patients.
A surprising finding of this study was the lower rate of HF-related hospitalizations in the HT group. Although not the primary subject of interest in the study, it reinforced the importance of close health care professional follow-up for patients living with HF. Telehealth may improve communication and shared decision making over medication use. Because the finding was unanticipated, the rate of diuretic adjustments was not tracked.
Patients were reevaluated every 6 months for willingness to continue the program, adherence, and clinical needs. These results are similar to those of other trials that demonstrated improved rates of GDMT in the setting of pharmacist- or nurse-led HF treatment optimization.15,16 These positive results differ from other trials incorporating remote monitoring regarding patient continuation in HT programs. For example, in a study by Ding and colleagues, the withdrawal rate from their monitoring service was about 22%, while in our study only 1 patient withdrew from the HT program.11
The HT program resulted in fewer hospitalizations than the control arm. There were 6 HF-related hospitalizations in the control group, although 5 involved a single patient. Typically, such a patient would be encouraged to follow HT monitoring after just 1 HF-related hospitalization; however, the patient declined to participate.
Early optimization of GDMT in patients who were recently discharged from the hospital for an HF-related hospitalization yields a reduction in hospital rehospitalization.17 GDMT optimization has unequivocal benefits in HF outcomes. Unfortunately, the issues surrounding methodologies on how to best optimize GDMT are lacking. While HT has been found to be feasible in the aid of optimizing medical therapy, the TIM-HF trial concluded that remote monitoring services had no significant benefit in reducing mortality.7,8 On the other hand, the OptiLink HF Study showed that when clinicians respond to remote monitoring prompts from fluid index threshold crossing alerts, these interventions are associated with significantly improved clinical outcomes in patients with implantable cardioverter-defibrillators and advanced HF.9 In contrast to previous trials, the AMULET trial showed that remote monitoring compared with standard care significantly reduced the risk of HF hospitalization or cardiovascular death during the 12-month follow-up among patients with HF and LVEF ≤ 49% after an episode of acute exacerbation.10 Additionally, patients who received skilled home health services and participated in remote monitoring for their chronic HF experienced a reduction in all-cause 30-day readmission.18
Given the contrasting evidence regarding remote monitoring and variable modalities of implementing interventions, we investigated whether HT monitoring yields improvements in GDMT optimization. We found that HT nurses were able to nearly double the rate of interventions for patients with HFrEF. The HT program in providing expanded services will require adequate staffing responsibilities and support. The HT program is geared toward following a large, diverse patient population, such as those with chronic obstructive pulmonary disease, hypertension, and HF. We only evaluated services for patients with HFrEF, but the program also follows patients with HfmrEF and HfpEF. These patients were not included as GDMT optimization differs for patients with an LVEF > 40%.19,20
The lower rates of achieving target doses of GDMTs were likely obstructed by continuous use of initial drug doses and further limited by lack of follow-up. When compared with the rest of the VAAAHS, there was a greater effort to increase ARNi use in the HT group as 7 of 33 patients (21%) were started on ARNi compared with a background increase of ARNi use of 17%. There was a lower mortality rate observed in the HT group compared with the control group. One patient in each group died of unrelated causes, while 2 deaths in the control group were due to worsening HF. The difference in mortality is likely multifactorial, possibly related to the control group’s greater disease burden or higher mean age (75.2 years vs 72.6 years).
Limitations
This was an observational cohort design, which is subject to bias. Thus, the findings of this study are entirely hypothesis-generating and a randomized controlled trial would be necessary for clearer results. Second, low numbers of participants may have skewed the data set. Given the observational nature of the study, this nonetheless is a positive finding to support the HT program for assisting with HF monitoring and prompting drug interventions. Due to the low number of participants, a single patient may have skewed the results with 5 hospitalizations.
Conclusions
This pilot study demonstrates the applicability of HT monitoring to optimize veteran HFrEF GDMT. The HT program yielded numerically relevant interventions and fewer HF-related hospitalizations compared with the control arm. The study shows the feasibility of the program to safely optimize GDMT toward their target doses and may serve as an additional catalyst to further develop HT programs specifically targeted toward HF monitoring and management. Cost-savings analyses would likely need to demonstrate the cost utility of such a service.
We thank the home telehealth nursing staff for their assistance in data collection and enrollment of patients into the monitoring program.
Heart failure (HF) is a chronic, progressive condition that is characterized by the heart’s inability to effectively pump blood throughout the body. In 2018, approximately 6.2 million US adults had HF, and 13.4% of all death certificates noted HF as a precipitating factor.1 Patients not receiving appropriate guideline-directed medical therapy (GDMT) face a 29% excess mortality risk over a 2-year period.2 Each additional GDMT included in a patient’s regimen significantly reduces all-cause mortality.3
The Change the Management of Patients with Heart Failure (CHAMP) registry reports that only about 1% of patients with HF are prescribed 3 agents from contemporary GDMT at target doses, highlighting the need for optimizing clinicians’ approaches to GDMT.4 Similarly, The Get With The Guidelines Heart Failure Registry has noted that only 20.2% of patients with HF with reduced ejection fraction (HFrEF) are prescribed a sodium-glucose cotransporter 2 inhibitor (SGLT2i) following hospital discharge for HFrEF exacerbation.5 Overall, treatment rates with GDMT saw limited improvement between 2013 and 2019, with no significant difference between groups in mortality, indicating the need for optimized methods to encourage the initiation of GDMT.6
Remote monitoring and telecare are novel ways to improve GDMT rates in those with HFrEF. However, data are inconsistent regarding the impact of remote HF monitoring and improvements in GDMT or HF-related outcomes.6-10 The modalities of remote monitoring for GDMT vary among studies, but the potential for telehealth monitoring to improve GDMT, thereby potentially reducing HF-related hospitalizations, is clear.
Telemonitoring has demonstrated improved participant adherence with weight monitoring, although the withdrawal rate was high, and has the potential to reduce all-cause mortality and HF-related hospitalizations.11,12 Telemonitoring for GDMT optimization led to an increase in the proportion of patients who achieved optimal GDMT doses, a decrease in the time to dose optimization, and a reduction in the number of clinic visits.13 Remote GDMT titration was accomplished in the general patient population with HFrEF; however, in populations already followed by cardiologists or HF specialists, remote optimization strategies did not yield different proportions of GDMT use.14 The aim of this study was to assess the impact of the home telehealth (HT) monitoring program on the initiation and optimization of HF GDMT among veterans with HFrEF at the Veterans Affairs Ann Arbor Healthcare System (VAAAHS) in Michigan.
Methods
This was a single-center retrospective study of Computerized Patient Record System (CPRS)data. Patients at the VAAAHS were evaluated if they were diagnosed with HFrEF and were eligible for enrollment in the HT monitoring program. Eligibility criteria included a diagnosis of stage C HF, irrespective of EF, and a history of any HF-related hospitalization. We focused on patients with HFrEF due to stronger guideline-based recommendations for certain pharmacotherapies as compared with HF with mildly reduced ejection fraction (HFmrEF) and preserved ejection fraction (HFpEF). Initial patient data for HT enrolling were accessed using the Heart Failure Dashboard via the US Department of Veterans Affairs (VA) Academic Detailing Service. The target daily doses of typical agents used in HFrEF GDMT are listed in the Appendix.
The HT program is an embedded model in which HT nurses receive remote data from the patient and triage that with the VAAAHS cardiology team. Patients’ questions, concerns, and/or vital signs are recovered remotely. In this model, nurses are embedded in the cardiology team, working with the cardiologists, cardiology clinical pharmacist, and/or cardiology nurse practitioners to make medication interventions. Data are recorded with an HT device, including weight, blood pressure (BP), heart rate, and pulse oximetry. HT nurses are also available to the patient via phone or video. The program uses a 180-day disease management protocol for HF via remote device, enabling the patient to answer questions and receive education on their disease daily. Responses to questions and data are then reviewed by an HT nurse remotely during business hours and triaged as appropriate with the cardiology team. Data can be communicated to the cardiology team via the patient record, eliminating the need for the cardiology team to use the proprietary portal affiliated with the HT device.
Study Sample
Patient information was obtained from a list of 417 patients eligible for enrollment in the HT program; the list was sent to the HT program for review and enrollment. Patient data were extracted from the VAAAHS HF Dashboard and included all patients with HFrEF and available data on the platform. The sample for the retrospective chart review included 40 adults who had HFrEF, defined as a left ventricular EF (LVEF) of ≤ 40% as evidenced by a transthoracic echocardiogram or cardiac magnetic resonance imaging. These patients were contacted and agreed to enroll in the HT monitoring program. The HT program population was compared against a control group of 33 patients who were ineligible for the HT program. Patients were deemed ineligible for HT if they resided in a nursing home, lacked a VAAAHS primary care clinician, or declined participation in the HT program.
Procedures
Patients who declined participation in the HT program followed the standard of care, which was limited to visits with primary care clinicians and/or cardiologists as per the follow-up plan. Patient data were collected over 12 months. The study was approved by the VAAAHS Institutional Review Board (reference number, 1703034), Research and Development Committee, and Research Administration.
Primary and Secondary Goals
The primary goal of the study was to assess the impact of the HT program on drug interventions, specifically initiating and titrating HFrEF pharmacotherapies. Interventions were based on GDMT with known mortality- and morbidity-reducing properties when used at their maximum tolerated doses, including angiotensin-converting enzyme inhibitors (ACEi), angiotensin receptor-neprilysin inhibitor (ARNi), or angiotensin receptor blockers (ARB), with a preference for ARNi, β-blockers for HFrEF (metoprolol succinate, bisoprolol, or carvedilol), aldosterone antagonists, and SGLT2is.
Secondary goals included HF-related hospitalizations, medication adherence, time to enrollment in HT, time to laboratory analysis after the initiation or titration of an ACEi/ARB/ARNi or aldosterone antagonist, and time enrolled in the HT program. Patients were considered adherent if their drug refill history showed consistent fills of their medications. The χ2 test was used for total interventions made during the study period and Fisher exact test for all others.
Results
Patient data were collected between July 2022 and June 2023. All 73 patients were male, and the mean age in the HT group (n = 40) was 72.6 years and 75.2 years for the control group (n = 33). Overall, the baseline demographics were similar between the groups (Table 1). Of 40 patients screened for enrollment in the HT program, 33 were included in the analysis (Figure 1).
At baseline, the HT group included more individuals than the control group on ACEi/ARB/ARNi (24 vs 19, respectively), β-blocker (28 vs 24, respectively), SGLT2i (14 vs 11, respectively), and aldosterone antagonist (15 vs 9, respectively) (Figure 2). There were 20 interventions made in the HT group compared with 11 therapy changes in the control arm during the study (odds ratio, 1.43; P = .23) (Table 2). In the HT group, 1 patient achieved an ACEi target dose, 3 patients achieved a β-blocker target dose, and 7 achieved a target dose of spironolactone (titration is not required for SGLT2i therapy and is counted as target dose). In the HT group, 17 patients were on ≥ 3 recommended agents, while 9 patients were taking 4 agents. Seven of 20 HT group interventions resulted in titration to the target dose. In the control group, no patients achieved an ARNi target dose, 3 patients achieved a β-blocker target dose, and 2 patients achieved a spironolactone antagonist target dose. In the control arm, 7 patients were on ≥ 3 GDMTs, and 2 were taking 4 agents. No patient in either group achieved a target dose of 4 agents. Five of 11 control group interventions resulted in initiation or titration of GDMT to the target dose.
Of the 40 HT group patients, 7 were excluded from analysis (3 failed to schedule HT, 1 was at a long-term care facility, 1 was nonadherent, 1 declined participation, and 1 died) and 33 remained in the program for a mean (SD) 5.3 (3.5) months. Death rates were tracked during the study: 1 patient died in the HT group and 3 in the control group.
We analyzed the overall percentage of VAAAHS patients with HFrEF who were on appropriate GDMT. Given the ongoing drive to improve HF-related outcomes, HT interventions could not be compared to a static population, so the HT and control patients were compared with the rates of GDMT at VAAAHS, which was available in the Academic Detailing Service Heart Failure Dashboard (Figure 3). Titration and optimization rates were also compared (Figure 4). From July 2022 to June 2023, ARNi use increased by 16.6%, aldosterone antagonist by 6.8%, and β-blockers by 2.4%. Target doses of GDMTs were more difficult to achieve in the hospital system. There was an increase in aldosterone antagonist target dose achievement by 4.7%, but overall there were decreases in target doses in other GDMTs: ACEi/ARB/ARNi target dose use decreased by 3.2%, ARNi target dose use decreased by 2.7% and target β-blocker use decreased by 0.9%.
Discussion
Telehealth yielded clinically important interventions, with some titrations bringing patients to their target doses of medications for HFrEF. The 20 interventions made in the HT group can be largely attributed to the nurses’ efforts to alert clinicians to drug titrations or ACEi/ARB to ARNi transitions. Although the findings were not statistically significant, the difference in the number of drug therapy changes supports the use of the HT program for a GDMT optimization strategy. Patients may be difficult to titrate secondary to adverse effects that make medication initiation or titration inappropriate, such as hypotension and hyperkalemia, although this was not observed in this small sample size. Considering a mean HT enrollment of 5.3 months, many patients had adequate disease assessment and medication titration. Given that patients are discharged from the service once deemed appropriate, this decreases the burden on the patient and increases the utility and implementation of the HT program for other patients.
A surprising finding of this study was the lower rate of HF-related hospitalizations in the HT group. Although not the primary subject of interest in the study, it reinforced the importance of close health care professional follow-up for patients living with HF. Telehealth may improve communication and shared decision making over medication use. Because the finding was unanticipated, the rate of diuretic adjustments was not tracked.
Patients were reevaluated every 6 months for willingness to continue the program, adherence, and clinical needs. These results are similar to those of other trials that demonstrated improved rates of GDMT in the setting of pharmacist- or nurse-led HF treatment optimization.15,16 These positive results differ from other trials incorporating remote monitoring regarding patient continuation in HT programs. For example, in a study by Ding and colleagues, the withdrawal rate from their monitoring service was about 22%, while in our study only 1 patient withdrew from the HT program.11
The HT program resulted in fewer hospitalizations than the control arm. There were 6 HF-related hospitalizations in the control group, although 5 involved a single patient. Typically, such a patient would be encouraged to follow HT monitoring after just 1 HF-related hospitalization; however, the patient declined to participate.
Early optimization of GDMT in patients who were recently discharged from the hospital for an HF-related hospitalization yields a reduction in hospital rehospitalization.17 GDMT optimization has unequivocal benefits in HF outcomes. Unfortunately, the issues surrounding methodologies on how to best optimize GDMT are lacking. While HT has been found to be feasible in the aid of optimizing medical therapy, the TIM-HF trial concluded that remote monitoring services had no significant benefit in reducing mortality.7,8 On the other hand, the OptiLink HF Study showed that when clinicians respond to remote monitoring prompts from fluid index threshold crossing alerts, these interventions are associated with significantly improved clinical outcomes in patients with implantable cardioverter-defibrillators and advanced HF.9 In contrast to previous trials, the AMULET trial showed that remote monitoring compared with standard care significantly reduced the risk of HF hospitalization or cardiovascular death during the 12-month follow-up among patients with HF and LVEF ≤ 49% after an episode of acute exacerbation.10 Additionally, patients who received skilled home health services and participated in remote monitoring for their chronic HF experienced a reduction in all-cause 30-day readmission.18
Given the contrasting evidence regarding remote monitoring and variable modalities of implementing interventions, we investigated whether HT monitoring yields improvements in GDMT optimization. We found that HT nurses were able to nearly double the rate of interventions for patients with HFrEF. The HT program in providing expanded services will require adequate staffing responsibilities and support. The HT program is geared toward following a large, diverse patient population, such as those with chronic obstructive pulmonary disease, hypertension, and HF. We only evaluated services for patients with HFrEF, but the program also follows patients with HfmrEF and HfpEF. These patients were not included as GDMT optimization differs for patients with an LVEF > 40%.19,20
The lower rates of achieving target doses of GDMTs were likely obstructed by continuous use of initial drug doses and further limited by lack of follow-up. When compared with the rest of the VAAAHS, there was a greater effort to increase ARNi use in the HT group as 7 of 33 patients (21%) were started on ARNi compared with a background increase of ARNi use of 17%. There was a lower mortality rate observed in the HT group compared with the control group. One patient in each group died of unrelated causes, while 2 deaths in the control group were due to worsening HF. The difference in mortality is likely multifactorial, possibly related to the control group’s greater disease burden or higher mean age (75.2 years vs 72.6 years).
Limitations
This was an observational cohort design, which is subject to bias. Thus, the findings of this study are entirely hypothesis-generating and a randomized controlled trial would be necessary for clearer results. Second, low numbers of participants may have skewed the data set. Given the observational nature of the study, this nonetheless is a positive finding to support the HT program for assisting with HF monitoring and prompting drug interventions. Due to the low number of participants, a single patient may have skewed the results with 5 hospitalizations.
Conclusions
This pilot study demonstrates the applicability of HT monitoring to optimize veteran HFrEF GDMT. The HT program yielded numerically relevant interventions and fewer HF-related hospitalizations compared with the control arm. The study shows the feasibility of the program to safely optimize GDMT toward their target doses and may serve as an additional catalyst to further develop HT programs specifically targeted toward HF monitoring and management. Cost-savings analyses would likely need to demonstrate the cost utility of such a service.
We thank the home telehealth nursing staff for their assistance in data collection and enrollment of patients into the monitoring program.
1. Tsao CW, Aday AW, Almarzooq ZI, et al. Heart disease and stroke statistics-2022 update: a report from the American Heart Association. Circulation. 2022;145(8):e153-e639. doi:10.1161/CIR.0000000000001052
2. McCullough PA, Mehta HS, Barker CM, et al. Mortality and guideline-directed medical therapy in real-world heart failure patients with reduced ejection fraction. Clin Cardiol. 2021;44(9):1192-1198. doi:10.1002/clc.23664
3. Tromp J, Ouwerkerk W, van Veldhuisen DJ, et al. A systematic review and network meta-analysis of pharmacological treatment of heart failure with reduced ejection fraction. JACC Heart Fail. 2022;10(2):73-84. doi:10.1016/j.jchf.2021.09.004
4. Greene SJ, Butler J, Albert NM, et al. Medical therapy for heart failure with reduced ejection fraction: the CHAMP-HF Registry. J Am Coll Cardiol. 2018;72(4):351-366. doi:10.1016/j.jacc.2018.04.070
5. Pierce JB, Vaduganathan M, Fonarow GC, et al. Contemporary use of sodium-glucose cotransporter-2 inhibitor therapy among patients hospitalized for heart failure with reduced ejection fraction in the US: The Get With The Guidelines-Heart Failure Registry. JAMA Cardiol. 2023;8(7):652-661. doi:10.1001/jamacardio.2023.1266
6. Sandhu AT, Kohsaka S, Turakhia MP, Lewis EF, Heidenreich PA. Evaluation of quality of care for US veterans with recent-onset heart failure with reduced ejection fraction. JAMA Cardiol. 2022;7(2):130-139. doi:10.1001/jamacardio.2021.4585 7. Rahimi K, Nazarzadeh M, Pinho-Gomes AC, et al. Home monitoring with technology-supported management in chronic heart failure: a randomised trial. Heart. 2020;106(20):1573-1578. doi:10.1136/heartjnl-2020-316773 8. Koehler F, Winkler S, Schieber M, et al. Impact of remote telemedical management on mortality and hospitalizations in ambulatory patients with chronic heart failure: the telemedical interventional monitoring in heart failure study. Circulation. 2011;123(17):1873-1880. doi:10.1161/CIRCULATIONAHA.111.018473
9. Wintrich J, Pavlicek V, Brachmann J, et al. Remote monitoring with appropriate reaction to alerts was associated with improved outcomes in chronic heart failure: results from the OptiLink HF study. Circ Arrhythm Electrophysiol. 2021;14(1):e008693. doi:10.1161/CIRCEP.120.008693
10. Krzesinski P, Jankowska EA, Siebert J, et al. Effects of an outpatient intervention comprising nurse-led non-invasive assessments, telemedicine support and remote cardiologists’ decisions in patients with heart failure (AMULET study): a randomised controlled trial. Eur J Heart Fail. 2022;24(3):565-577. doi:10.1002/ejhf.2358
11. Ding H, Jayasena R, Chen SH, et al. The effects of telemonitoring on patient compliance with self-management recommendations and outcomes of the innovative telemonitoring enhanced care program for chronic heart failure: randomized controlled trial. J Med Internet Res. 2020;22(7):e17559. doi:10.2196/17559
12. Kitsiou S, Pare G, Jaana M. Effects of home telemonitoring interventions on patients with chronic heart failure: an overview of systematic reviews. J Med Internet Res. 2015;17(3):e63. doi:10.2196/jmir.4174
13. Artanian V, Ross HJ, Rac VE, O’Sullivan M, Brahmbhatt DH, Seto E. Impact of remote titration combined with telemonitoring on the optimization of guideline-directed medical therapy for patients with heart failure: internal pilot of a randomized controlled trial. JMIR Cardio. 2020;4(1):e21962. doi:10.2196/21962
14. Desai AS, Maclean T, Blood AJ, et al. Remote optimization of guideline-directed medical therapy in patients with heart failure with reduced ejection fraction. JAMA Cardiol. 2020;5(12):1430-1434. doi:10.1001/jamacardio.2020.3757
15. Patil T, Ali S, Kaur A, et al. Impact of pharmacist-led heart failure clinic on optimization of guideline-directed medical therapy (PHARM-HF). J Cardiovasc Transl Res. 2022;15(6):1424-1435. doi:10.1007/s12265-022-10262-9
16. Zheng J, Mednick T, Heidenreich PA, Sandhu AT. Pharmacist- and nurse-led medical optimization in heart failure: a systematic review and meta-analysis. J Card Fail. 2023;29(7):1000-1013. doi:10.1016/j.cardfail.2023.03.012
17. Mebazaa A, Davison B, Chioncel O, et al. Safety, tolerability and efficacy of up-titration of guideline-directed medical therapies for acute heart failure (STRONG-HF): a multinational, open-label, randomised, trial. Lancet. 2022;400(10367):1938-1952. doi:10.1016/S0140-6736(22)02076-1
18. O’Connor M, Asdornwised U, Dempsey ML, et al. Using telehealth to reduce all-cause 30-day hospital readmissions among heart failure patients receiving skilled home health services. Appl Clin Inform. 2016;7(2):238-47. doi:10.4338/ACI-2015-11-SOA-0157
19. Heidenreich PA, Bozkurt B, Aguilar D, et al. 2022 AHA/ACC/HFSA Guideline for the Management of Heart Failure: Executive Summary: A Report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. Circulation. 2022;145(18):e876-e894. doi:10.1161/CIR.0000000000001062
20. Kittleson MM, Panjrath GS, Amancherla K, et al. 2023 ACC Expert Consensus Decision Pathway on Management of Heart Failure With Preserved Ejection Fraction: A Report of the American College of Cardiology Solution Set Oversight Committee. J Am Coll Cardiol. 2023;81(18):1835-1878. doi:10.1016/j.jacc.2023.03.393
1. Tsao CW, Aday AW, Almarzooq ZI, et al. Heart disease and stroke statistics-2022 update: a report from the American Heart Association. Circulation. 2022;145(8):e153-e639. doi:10.1161/CIR.0000000000001052
2. McCullough PA, Mehta HS, Barker CM, et al. Mortality and guideline-directed medical therapy in real-world heart failure patients with reduced ejection fraction. Clin Cardiol. 2021;44(9):1192-1198. doi:10.1002/clc.23664
3. Tromp J, Ouwerkerk W, van Veldhuisen DJ, et al. A systematic review and network meta-analysis of pharmacological treatment of heart failure with reduced ejection fraction. JACC Heart Fail. 2022;10(2):73-84. doi:10.1016/j.jchf.2021.09.004
4. Greene SJ, Butler J, Albert NM, et al. Medical therapy for heart failure with reduced ejection fraction: the CHAMP-HF Registry. J Am Coll Cardiol. 2018;72(4):351-366. doi:10.1016/j.jacc.2018.04.070
5. Pierce JB, Vaduganathan M, Fonarow GC, et al. Contemporary use of sodium-glucose cotransporter-2 inhibitor therapy among patients hospitalized for heart failure with reduced ejection fraction in the US: The Get With The Guidelines-Heart Failure Registry. JAMA Cardiol. 2023;8(7):652-661. doi:10.1001/jamacardio.2023.1266
6. Sandhu AT, Kohsaka S, Turakhia MP, Lewis EF, Heidenreich PA. Evaluation of quality of care for US veterans with recent-onset heart failure with reduced ejection fraction. JAMA Cardiol. 2022;7(2):130-139. doi:10.1001/jamacardio.2021.4585 7. Rahimi K, Nazarzadeh M, Pinho-Gomes AC, et al. Home monitoring with technology-supported management in chronic heart failure: a randomised trial. Heart. 2020;106(20):1573-1578. doi:10.1136/heartjnl-2020-316773 8. Koehler F, Winkler S, Schieber M, et al. Impact of remote telemedical management on mortality and hospitalizations in ambulatory patients with chronic heart failure: the telemedical interventional monitoring in heart failure study. Circulation. 2011;123(17):1873-1880. doi:10.1161/CIRCULATIONAHA.111.018473
9. Wintrich J, Pavlicek V, Brachmann J, et al. Remote monitoring with appropriate reaction to alerts was associated with improved outcomes in chronic heart failure: results from the OptiLink HF study. Circ Arrhythm Electrophysiol. 2021;14(1):e008693. doi:10.1161/CIRCEP.120.008693
10. Krzesinski P, Jankowska EA, Siebert J, et al. Effects of an outpatient intervention comprising nurse-led non-invasive assessments, telemedicine support and remote cardiologists’ decisions in patients with heart failure (AMULET study): a randomised controlled trial. Eur J Heart Fail. 2022;24(3):565-577. doi:10.1002/ejhf.2358
11. Ding H, Jayasena R, Chen SH, et al. The effects of telemonitoring on patient compliance with self-management recommendations and outcomes of the innovative telemonitoring enhanced care program for chronic heart failure: randomized controlled trial. J Med Internet Res. 2020;22(7):e17559. doi:10.2196/17559
12. Kitsiou S, Pare G, Jaana M. Effects of home telemonitoring interventions on patients with chronic heart failure: an overview of systematic reviews. J Med Internet Res. 2015;17(3):e63. doi:10.2196/jmir.4174
13. Artanian V, Ross HJ, Rac VE, O’Sullivan M, Brahmbhatt DH, Seto E. Impact of remote titration combined with telemonitoring on the optimization of guideline-directed medical therapy for patients with heart failure: internal pilot of a randomized controlled trial. JMIR Cardio. 2020;4(1):e21962. doi:10.2196/21962
14. Desai AS, Maclean T, Blood AJ, et al. Remote optimization of guideline-directed medical therapy in patients with heart failure with reduced ejection fraction. JAMA Cardiol. 2020;5(12):1430-1434. doi:10.1001/jamacardio.2020.3757
15. Patil T, Ali S, Kaur A, et al. Impact of pharmacist-led heart failure clinic on optimization of guideline-directed medical therapy (PHARM-HF). J Cardiovasc Transl Res. 2022;15(6):1424-1435. doi:10.1007/s12265-022-10262-9
16. Zheng J, Mednick T, Heidenreich PA, Sandhu AT. Pharmacist- and nurse-led medical optimization in heart failure: a systematic review and meta-analysis. J Card Fail. 2023;29(7):1000-1013. doi:10.1016/j.cardfail.2023.03.012
17. Mebazaa A, Davison B, Chioncel O, et al. Safety, tolerability and efficacy of up-titration of guideline-directed medical therapies for acute heart failure (STRONG-HF): a multinational, open-label, randomised, trial. Lancet. 2022;400(10367):1938-1952. doi:10.1016/S0140-6736(22)02076-1
18. O’Connor M, Asdornwised U, Dempsey ML, et al. Using telehealth to reduce all-cause 30-day hospital readmissions among heart failure patients receiving skilled home health services. Appl Clin Inform. 2016;7(2):238-47. doi:10.4338/ACI-2015-11-SOA-0157
19. Heidenreich PA, Bozkurt B, Aguilar D, et al. 2022 AHA/ACC/HFSA Guideline for the Management of Heart Failure: Executive Summary: A Report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. Circulation. 2022;145(18):e876-e894. doi:10.1161/CIR.0000000000001062
20. Kittleson MM, Panjrath GS, Amancherla K, et al. 2023 ACC Expert Consensus Decision Pathway on Management of Heart Failure With Preserved Ejection Fraction: A Report of the American College of Cardiology Solution Set Oversight Committee. J Am Coll Cardiol. 2023;81(18):1835-1878. doi:10.1016/j.jacc.2023.03.393
AHA, AAP update neonatal resuscitation guidelines
The 2023 focused update was prompted by four systematic literature reviews by the International Liaison Committee on Resuscitation (ILCOR) Neonatal Life Support Task Force.
“Evidence evaluations by the ILCOR play a large role in the group’s process and timing of updates,” Henry Lee, MD, co-chair of the writing group, said in an interview.
He noted that updated recommendations do not change prior recommendations from the 2020 guidelines.
“However, they provide additional details to consider in neonatal resuscitation that could lead to changes in some practice in various settings,” said Dr. Lee, medical director of the University of California San Diego neonatal intensive care unit.
The focused update was simultaneously published online November 16 in Circulation and in Pediatrics.
Dr. Lee noted that effective positive-pressure ventilation (PPV) is the priority in newborn infants who need support after birth.
And while the 2020 update provided some details on devices to be used for PPV, the 2023 focused update gives guidance on use of T-piece resuscitators for providing PPV, which may be particularly helpful for preterm infants, and the use of supraglottic airways as a primary interface to deliver PPV, he explained.
Specifically, the updated guidelines state that use of a T-piece resuscitator to deliver PPV is preferred to the use of a self-inflating bag.
Because both T-piece resuscitators and flow-inflating bags require a compressed gas source to function, a self-inflating bag should be available as a backup in the event of compressed gas failure when using either of these devices.
Use of a supraglottic airway may be considered as the primary interface to administer PPV instead of a face mask for newborn infants delivered at 34 0/7 weeks’ gestation or later.
Continued Emphasis on Delayed Cord Clamping
The updated guidelines “continue to emphasize delayed cord clamping for both term and preterm newborn infants when clinically possible. There is also a new recommendation for nonvigorous infants born 35-42 weeks’ gestational age to consider umbilical cord milking,” Dr. Lee said in an interview.
Specifically, the guidelines state:
- For term and late preterm newborn infants ≥34 weeks’ gestation, and preterm newborn infants <34 weeks’ gestation, who do not require resuscitation, delayed cord clamping (≥30 seconds) can be beneficial compared with early cord clamping (<30 seconds).
- For term and late preterm newborn infants ≥34 weeks’ gestation who do not require resuscitation, intact cord milking is not known to be beneficial compared with delayed cord clamping (≥30 seconds).
- For preterm newborn infants between 28- and 34-weeks’ gestation who do not require resuscitation and in whom delayed cord clamping cannot be performed, intact cord milking may be reasonable.
- For preterm newborn infants <28 weeks’ gestation, intact cord milking is not recommended.
- For nonvigorous term and late preterm infants (35-42 weeks’ gestation), intact cord milking may be reasonable compared with early cord clamping (<30 seconds).
The guidelines also highlight the following knowledge gaps that require further research:
- Optimal management of the umbilical cord in term, late preterm, and preterm infants who require resuscitation at delivery
- Longer-term outcome data, such as anemia during infancy and neurodevelopmental outcomes, for all umbilical cord management strategies
- Cost-effectiveness of a T-piece resuscitator compared with a self-inflating bag
- The effect of a self-inflating bag with a positive end-expiratory pressure valve on outcomes in preterm newborn infants
- Comparison of either a T-piece resuscitator or a self-inflating bag with a flow-inflating bag for administering PPV
- Comparison of clinical outcomes by gestational age for any PPV device
- Comparison of supraglottic airway devices and face masks as the primary interface for PPV in high-resourced settings
- The amount and type of training required for successful supraglottic airway insertion and the potential for skill decay
- The utility of supraglottic airway devices for suctioning secretions from the airway
- The efficacy of a supraglottic airway during advanced neonatal resuscitation requiring chest compressions or the delivery of intratracheal medications
This research had no commercial funding. The authors report no relevant financial relationships.
A version of this article appeared on Medscape.com.
The 2023 focused update was prompted by four systematic literature reviews by the International Liaison Committee on Resuscitation (ILCOR) Neonatal Life Support Task Force.
“Evidence evaluations by the ILCOR play a large role in the group’s process and timing of updates,” Henry Lee, MD, co-chair of the writing group, said in an interview.
He noted that updated recommendations do not change prior recommendations from the 2020 guidelines.
“However, they provide additional details to consider in neonatal resuscitation that could lead to changes in some practice in various settings,” said Dr. Lee, medical director of the University of California San Diego neonatal intensive care unit.
The focused update was simultaneously published online November 16 in Circulation and in Pediatrics.
Dr. Lee noted that effective positive-pressure ventilation (PPV) is the priority in newborn infants who need support after birth.
And while the 2020 update provided some details on devices to be used for PPV, the 2023 focused update gives guidance on use of T-piece resuscitators for providing PPV, which may be particularly helpful for preterm infants, and the use of supraglottic airways as a primary interface to deliver PPV, he explained.
Specifically, the updated guidelines state that use of a T-piece resuscitator to deliver PPV is preferred to the use of a self-inflating bag.
Because both T-piece resuscitators and flow-inflating bags require a compressed gas source to function, a self-inflating bag should be available as a backup in the event of compressed gas failure when using either of these devices.
Use of a supraglottic airway may be considered as the primary interface to administer PPV instead of a face mask for newborn infants delivered at 34 0/7 weeks’ gestation or later.
Continued Emphasis on Delayed Cord Clamping
The updated guidelines “continue to emphasize delayed cord clamping for both term and preterm newborn infants when clinically possible. There is also a new recommendation for nonvigorous infants born 35-42 weeks’ gestational age to consider umbilical cord milking,” Dr. Lee said in an interview.
Specifically, the guidelines state:
- For term and late preterm newborn infants ≥34 weeks’ gestation, and preterm newborn infants <34 weeks’ gestation, who do not require resuscitation, delayed cord clamping (≥30 seconds) can be beneficial compared with early cord clamping (<30 seconds).
- For term and late preterm newborn infants ≥34 weeks’ gestation who do not require resuscitation, intact cord milking is not known to be beneficial compared with delayed cord clamping (≥30 seconds).
- For preterm newborn infants between 28- and 34-weeks’ gestation who do not require resuscitation and in whom delayed cord clamping cannot be performed, intact cord milking may be reasonable.
- For preterm newborn infants <28 weeks’ gestation, intact cord milking is not recommended.
- For nonvigorous term and late preterm infants (35-42 weeks’ gestation), intact cord milking may be reasonable compared with early cord clamping (<30 seconds).
The guidelines also highlight the following knowledge gaps that require further research:
- Optimal management of the umbilical cord in term, late preterm, and preterm infants who require resuscitation at delivery
- Longer-term outcome data, such as anemia during infancy and neurodevelopmental outcomes, for all umbilical cord management strategies
- Cost-effectiveness of a T-piece resuscitator compared with a self-inflating bag
- The effect of a self-inflating bag with a positive end-expiratory pressure valve on outcomes in preterm newborn infants
- Comparison of either a T-piece resuscitator or a self-inflating bag with a flow-inflating bag for administering PPV
- Comparison of clinical outcomes by gestational age for any PPV device
- Comparison of supraglottic airway devices and face masks as the primary interface for PPV in high-resourced settings
- The amount and type of training required for successful supraglottic airway insertion and the potential for skill decay
- The utility of supraglottic airway devices for suctioning secretions from the airway
- The efficacy of a supraglottic airway during advanced neonatal resuscitation requiring chest compressions or the delivery of intratracheal medications
This research had no commercial funding. The authors report no relevant financial relationships.
A version of this article appeared on Medscape.com.
The 2023 focused update was prompted by four systematic literature reviews by the International Liaison Committee on Resuscitation (ILCOR) Neonatal Life Support Task Force.
“Evidence evaluations by the ILCOR play a large role in the group’s process and timing of updates,” Henry Lee, MD, co-chair of the writing group, said in an interview.
He noted that updated recommendations do not change prior recommendations from the 2020 guidelines.
“However, they provide additional details to consider in neonatal resuscitation that could lead to changes in some practice in various settings,” said Dr. Lee, medical director of the University of California San Diego neonatal intensive care unit.
The focused update was simultaneously published online November 16 in Circulation and in Pediatrics.
Dr. Lee noted that effective positive-pressure ventilation (PPV) is the priority in newborn infants who need support after birth.
And while the 2020 update provided some details on devices to be used for PPV, the 2023 focused update gives guidance on use of T-piece resuscitators for providing PPV, which may be particularly helpful for preterm infants, and the use of supraglottic airways as a primary interface to deliver PPV, he explained.
Specifically, the updated guidelines state that use of a T-piece resuscitator to deliver PPV is preferred to the use of a self-inflating bag.
Because both T-piece resuscitators and flow-inflating bags require a compressed gas source to function, a self-inflating bag should be available as a backup in the event of compressed gas failure when using either of these devices.
Use of a supraglottic airway may be considered as the primary interface to administer PPV instead of a face mask for newborn infants delivered at 34 0/7 weeks’ gestation or later.
Continued Emphasis on Delayed Cord Clamping
The updated guidelines “continue to emphasize delayed cord clamping for both term and preterm newborn infants when clinically possible. There is also a new recommendation for nonvigorous infants born 35-42 weeks’ gestational age to consider umbilical cord milking,” Dr. Lee said in an interview.
Specifically, the guidelines state:
- For term and late preterm newborn infants ≥34 weeks’ gestation, and preterm newborn infants <34 weeks’ gestation, who do not require resuscitation, delayed cord clamping (≥30 seconds) can be beneficial compared with early cord clamping (<30 seconds).
- For term and late preterm newborn infants ≥34 weeks’ gestation who do not require resuscitation, intact cord milking is not known to be beneficial compared with delayed cord clamping (≥30 seconds).
- For preterm newborn infants between 28- and 34-weeks’ gestation who do not require resuscitation and in whom delayed cord clamping cannot be performed, intact cord milking may be reasonable.
- For preterm newborn infants <28 weeks’ gestation, intact cord milking is not recommended.
- For nonvigorous term and late preterm infants (35-42 weeks’ gestation), intact cord milking may be reasonable compared with early cord clamping (<30 seconds).
The guidelines also highlight the following knowledge gaps that require further research:
- Optimal management of the umbilical cord in term, late preterm, and preterm infants who require resuscitation at delivery
- Longer-term outcome data, such as anemia during infancy and neurodevelopmental outcomes, for all umbilical cord management strategies
- Cost-effectiveness of a T-piece resuscitator compared with a self-inflating bag
- The effect of a self-inflating bag with a positive end-expiratory pressure valve on outcomes in preterm newborn infants
- Comparison of either a T-piece resuscitator or a self-inflating bag with a flow-inflating bag for administering PPV
- Comparison of clinical outcomes by gestational age for any PPV device
- Comparison of supraglottic airway devices and face masks as the primary interface for PPV in high-resourced settings
- The amount and type of training required for successful supraglottic airway insertion and the potential for skill decay
- The utility of supraglottic airway devices for suctioning secretions from the airway
- The efficacy of a supraglottic airway during advanced neonatal resuscitation requiring chest compressions or the delivery of intratracheal medications
This research had no commercial funding. The authors report no relevant financial relationships.
A version of this article appeared on Medscape.com.
ACC/AHA issue updated atrial fibrillation guideline
The American College of Cardiology (ACC), the American Heart Association (AHA), the American College of Chest Physicians (ACCP), and the Heart Rhythm Society (HRS) have issued an updated guideline for preventing and optimally managing atrial fibrillation (AF).
The 2023 ACC/AHA/ACCP/HRS Guideline for Diagnosis and Management of Atrial Fibrillation was published online in the Journal of the American College of Cardiology and Circulation.
“The new guideline has important changes,” including a new way to classify AF, Jose Joglar, MD, professor of cardiac electrophysiology at UT Southwestern Medical Center in Dallas, Texas, and chair of the writing committee, said in an interview.
The previous classification was largely based only on arrhythmia duration and tended to emphasize specific therapeutic interventions rather than a more holistic and multidisciplinary management approach, Dr. Joglar explained.
, from prevention, lifestyle and risk factor modification, screening, and therapy.
Stage 1: At risk for AF due to the presence of risk factors
Stage 2: Pre-AF, with evidence of structural or electrical findings predisposing to AF
Stage 3: AF, including paroxysmal (3A), persistent (3B), long-standing persistent (3C), successful AF ablation (3D)
Stage 4: Permanent AF
The updated guideline recognizes lifestyle and risk factor modification as a “pillar” of AF management and offers “more prescriptive” recommendations, including management of obesity, weight loss, physical activity, smoking cessation, alcohol moderation, hypertension, and other comorbidities.
“We should not only be telling patients they need to be healthy, which doesn’t mean much to a patient, we need to tell them precisely what they need to do. For example, how much exercise to do or how much weight to lose to have a benefit,” Dr. Joglar said in an interview.
The good news for many people, he noted, is that coffee, which has had a “bad reputation,” is okay, as the latest data show it doesn’t seem to exacerbate AF.
The new guideline continues to endorse use of the CHA2DS2-VASc score as the predictor of choice to determine the risk of stroke, but it also allows for flexibility to use other calculators when uncertainty exists or when other risk factors, such as kidney disease, need to be included.
With the emergence of “new and consistent” evidence, the guideline also emphasizes the importance of early and continued management of patients with AF with a focus on maintaining sinus rhythm and minimizing AF burden.
Catheter ablation of AF is given a class 1 indication as first-line therapy in selected patients, including those with heart failure with reduced ejection fraction.
That’s based on recent randomized studies that have shown catheter ablation to be “superior to pharmacological therapy” for rhythm control in appropriately selected patients, Dr. Joglar told this news organization.
“There’s no need to try pharmacological therapies after a discussion between the patient and doctor and they decide that they want to proceed with the most effective intervention,” he added.
The new guideline also upgrades the class of recommendation for left atrial appendage occlusion devices to 2a, compared with the 2019 AF Focused Update, for use of these devices in patients with long-term contraindications to anticoagulation.
It also provides updated recommendations for AF detected via implantable devices and wearables as well as recommendations for patients with AF identified during medical illness or surgery.
Development of the guideline had no commercial funding. Disclosures for the writing group are available with the original articles.
A version of this article appeared on Medscape.com.
The American College of Cardiology (ACC), the American Heart Association (AHA), the American College of Chest Physicians (ACCP), and the Heart Rhythm Society (HRS) have issued an updated guideline for preventing and optimally managing atrial fibrillation (AF).
The 2023 ACC/AHA/ACCP/HRS Guideline for Diagnosis and Management of Atrial Fibrillation was published online in the Journal of the American College of Cardiology and Circulation.
“The new guideline has important changes,” including a new way to classify AF, Jose Joglar, MD, professor of cardiac electrophysiology at UT Southwestern Medical Center in Dallas, Texas, and chair of the writing committee, said in an interview.
The previous classification was largely based only on arrhythmia duration and tended to emphasize specific therapeutic interventions rather than a more holistic and multidisciplinary management approach, Dr. Joglar explained.
, from prevention, lifestyle and risk factor modification, screening, and therapy.
Stage 1: At risk for AF due to the presence of risk factors
Stage 2: Pre-AF, with evidence of structural or electrical findings predisposing to AF
Stage 3: AF, including paroxysmal (3A), persistent (3B), long-standing persistent (3C), successful AF ablation (3D)
Stage 4: Permanent AF
The updated guideline recognizes lifestyle and risk factor modification as a “pillar” of AF management and offers “more prescriptive” recommendations, including management of obesity, weight loss, physical activity, smoking cessation, alcohol moderation, hypertension, and other comorbidities.
“We should not only be telling patients they need to be healthy, which doesn’t mean much to a patient, we need to tell them precisely what they need to do. For example, how much exercise to do or how much weight to lose to have a benefit,” Dr. Joglar said in an interview.
The good news for many people, he noted, is that coffee, which has had a “bad reputation,” is okay, as the latest data show it doesn’t seem to exacerbate AF.
The new guideline continues to endorse use of the CHA2DS2-VASc score as the predictor of choice to determine the risk of stroke, but it also allows for flexibility to use other calculators when uncertainty exists or when other risk factors, such as kidney disease, need to be included.
With the emergence of “new and consistent” evidence, the guideline also emphasizes the importance of early and continued management of patients with AF with a focus on maintaining sinus rhythm and minimizing AF burden.
Catheter ablation of AF is given a class 1 indication as first-line therapy in selected patients, including those with heart failure with reduced ejection fraction.
That’s based on recent randomized studies that have shown catheter ablation to be “superior to pharmacological therapy” for rhythm control in appropriately selected patients, Dr. Joglar told this news organization.
“There’s no need to try pharmacological therapies after a discussion between the patient and doctor and they decide that they want to proceed with the most effective intervention,” he added.
The new guideline also upgrades the class of recommendation for left atrial appendage occlusion devices to 2a, compared with the 2019 AF Focused Update, for use of these devices in patients with long-term contraindications to anticoagulation.
It also provides updated recommendations for AF detected via implantable devices and wearables as well as recommendations for patients with AF identified during medical illness or surgery.
Development of the guideline had no commercial funding. Disclosures for the writing group are available with the original articles.
A version of this article appeared on Medscape.com.
The American College of Cardiology (ACC), the American Heart Association (AHA), the American College of Chest Physicians (ACCP), and the Heart Rhythm Society (HRS) have issued an updated guideline for preventing and optimally managing atrial fibrillation (AF).
The 2023 ACC/AHA/ACCP/HRS Guideline for Diagnosis and Management of Atrial Fibrillation was published online in the Journal of the American College of Cardiology and Circulation.
“The new guideline has important changes,” including a new way to classify AF, Jose Joglar, MD, professor of cardiac electrophysiology at UT Southwestern Medical Center in Dallas, Texas, and chair of the writing committee, said in an interview.
The previous classification was largely based only on arrhythmia duration and tended to emphasize specific therapeutic interventions rather than a more holistic and multidisciplinary management approach, Dr. Joglar explained.
, from prevention, lifestyle and risk factor modification, screening, and therapy.
Stage 1: At risk for AF due to the presence of risk factors
Stage 2: Pre-AF, with evidence of structural or electrical findings predisposing to AF
Stage 3: AF, including paroxysmal (3A), persistent (3B), long-standing persistent (3C), successful AF ablation (3D)
Stage 4: Permanent AF
The updated guideline recognizes lifestyle and risk factor modification as a “pillar” of AF management and offers “more prescriptive” recommendations, including management of obesity, weight loss, physical activity, smoking cessation, alcohol moderation, hypertension, and other comorbidities.
“We should not only be telling patients they need to be healthy, which doesn’t mean much to a patient, we need to tell them precisely what they need to do. For example, how much exercise to do or how much weight to lose to have a benefit,” Dr. Joglar said in an interview.
The good news for many people, he noted, is that coffee, which has had a “bad reputation,” is okay, as the latest data show it doesn’t seem to exacerbate AF.
The new guideline continues to endorse use of the CHA2DS2-VASc score as the predictor of choice to determine the risk of stroke, but it also allows for flexibility to use other calculators when uncertainty exists or when other risk factors, such as kidney disease, need to be included.
With the emergence of “new and consistent” evidence, the guideline also emphasizes the importance of early and continued management of patients with AF with a focus on maintaining sinus rhythm and minimizing AF burden.
Catheter ablation of AF is given a class 1 indication as first-line therapy in selected patients, including those with heart failure with reduced ejection fraction.
That’s based on recent randomized studies that have shown catheter ablation to be “superior to pharmacological therapy” for rhythm control in appropriately selected patients, Dr. Joglar told this news organization.
“There’s no need to try pharmacological therapies after a discussion between the patient and doctor and they decide that they want to proceed with the most effective intervention,” he added.
The new guideline also upgrades the class of recommendation for left atrial appendage occlusion devices to 2a, compared with the 2019 AF Focused Update, for use of these devices in patients with long-term contraindications to anticoagulation.
It also provides updated recommendations for AF detected via implantable devices and wearables as well as recommendations for patients with AF identified during medical illness or surgery.
Development of the guideline had no commercial funding. Disclosures for the writing group are available with the original articles.
A version of this article appeared on Medscape.com.
Vagus nerve stimulation promising in POTS
TOPLINE:
possibly through decreased antiadrenergic autoantibodies and inflammatory cytokines, and improved cardiac autonomic function, in a small proof-of-concept study.
METHODOLOGY:
The double-blind study included 25 female patients with POTS, a syndrome of orthostatic intolerance (mean age 31 years and 81% Caucasian), who were randomly assigned to transcutaneous vagus nerve stimulation (tVNS) to the right tragus or sham stimulation to the earlobe, a site devoid of vagal innervation.
After training, patients delivered the tVNS themselves at a frequency of 20 Hz and pulse width of 200 ms during 1-hour daily sessions over 2 months.
At baseline and 2 months, patients underwent a tilt test to determine postural tachycardia; they remained supine for 25 minutes, followed by 10 minutes of standing, as tolerated.
Researchers used electrocardiogram data to examine heart rate and blood samples to assess serum cytokines and antiautonomic autoantibodies.
The primary outcome was a comparison of orthostatic tachycardia (standing – supine) between the two arms at 2 months.
TAKEAWAY:
At 2 months, postural tachycardia was significantly less in the active vs sham arm (mean postural increase in heart rate 17.6 beats/min vs 31.7 beats/min; P = .01).
There was a significant decrease in beta 1-adrenergic receptor (beta 1-AR; P = .01) and alpha-1-AR (P = .04) autoantibody activity in the active vs sham group, which may account at least in part for the reduced orthostatic tachycardia, although the exact mechanisms for this effect have not been clearly defined, the authors said.
Serum tumor necrosis factor-alpha (TNF-alpha) levels were significantly decreased in the active group relative to the sham group (8.3 pg/mL vs 13.9 pg/mL; P = .01).
As for heart rate variability, change in low frequency (LF) and high frequency (HF) from supine to standing was significantly decreased, and postural change in LF/HF ratio, a surrogate for sympathovagal balance, was significantly lower in the active group compared with the sham group.
IN PRACTICE:
“Collectively, these data suggest that tVNS, a low-cost, low-risk intervention, applied for a short period of time in selected patients with POTS, may result in a significant amelioration of their disease,” the authors conclude.
SOURCE:
The study was led by Stavros Stavrakis, MD, PhD, University of Oklahoma Health Sciences Center, Oklahoma City. It was published online in JACC: Clinical Electrophysiology..
LIMITATIONS:
The study had a small sample size, included only females, and extended only up to 2 months. As there was no improvement on the overall score from the Composite Autonomic Symptom Score 31 (COMPASS-31) questionnaire, researchers can’t conclude tVNS improved patient reported outcomes. The study used 1 hour of daily stimulation but the optimal duration and ideal timing of tVNS is yet to be determined.
DISCLOSURES:
The study was supported by the National Institutes of Health (NIH)/National Heart, Lung, and Blood Institute, NIH/National Institute of General Medical Sciences, and individual donations from Francie Fitzgerald and family through the OU Foundation Fund. The authors have no relevant conflicts of interest.
A version of this article appeared on Medscape.com.
TOPLINE:
possibly through decreased antiadrenergic autoantibodies and inflammatory cytokines, and improved cardiac autonomic function, in a small proof-of-concept study.
METHODOLOGY:
The double-blind study included 25 female patients with POTS, a syndrome of orthostatic intolerance (mean age 31 years and 81% Caucasian), who were randomly assigned to transcutaneous vagus nerve stimulation (tVNS) to the right tragus or sham stimulation to the earlobe, a site devoid of vagal innervation.
After training, patients delivered the tVNS themselves at a frequency of 20 Hz and pulse width of 200 ms during 1-hour daily sessions over 2 months.
At baseline and 2 months, patients underwent a tilt test to determine postural tachycardia; they remained supine for 25 minutes, followed by 10 minutes of standing, as tolerated.
Researchers used electrocardiogram data to examine heart rate and blood samples to assess serum cytokines and antiautonomic autoantibodies.
The primary outcome was a comparison of orthostatic tachycardia (standing – supine) between the two arms at 2 months.
TAKEAWAY:
At 2 months, postural tachycardia was significantly less in the active vs sham arm (mean postural increase in heart rate 17.6 beats/min vs 31.7 beats/min; P = .01).
There was a significant decrease in beta 1-adrenergic receptor (beta 1-AR; P = .01) and alpha-1-AR (P = .04) autoantibody activity in the active vs sham group, which may account at least in part for the reduced orthostatic tachycardia, although the exact mechanisms for this effect have not been clearly defined, the authors said.
Serum tumor necrosis factor-alpha (TNF-alpha) levels were significantly decreased in the active group relative to the sham group (8.3 pg/mL vs 13.9 pg/mL; P = .01).
As for heart rate variability, change in low frequency (LF) and high frequency (HF) from supine to standing was significantly decreased, and postural change in LF/HF ratio, a surrogate for sympathovagal balance, was significantly lower in the active group compared with the sham group.
IN PRACTICE:
“Collectively, these data suggest that tVNS, a low-cost, low-risk intervention, applied for a short period of time in selected patients with POTS, may result in a significant amelioration of their disease,” the authors conclude.
SOURCE:
The study was led by Stavros Stavrakis, MD, PhD, University of Oklahoma Health Sciences Center, Oklahoma City. It was published online in JACC: Clinical Electrophysiology..
LIMITATIONS:
The study had a small sample size, included only females, and extended only up to 2 months. As there was no improvement on the overall score from the Composite Autonomic Symptom Score 31 (COMPASS-31) questionnaire, researchers can’t conclude tVNS improved patient reported outcomes. The study used 1 hour of daily stimulation but the optimal duration and ideal timing of tVNS is yet to be determined.
DISCLOSURES:
The study was supported by the National Institutes of Health (NIH)/National Heart, Lung, and Blood Institute, NIH/National Institute of General Medical Sciences, and individual donations from Francie Fitzgerald and family through the OU Foundation Fund. The authors have no relevant conflicts of interest.
A version of this article appeared on Medscape.com.
TOPLINE:
possibly through decreased antiadrenergic autoantibodies and inflammatory cytokines, and improved cardiac autonomic function, in a small proof-of-concept study.
METHODOLOGY:
The double-blind study included 25 female patients with POTS, a syndrome of orthostatic intolerance (mean age 31 years and 81% Caucasian), who were randomly assigned to transcutaneous vagus nerve stimulation (tVNS) to the right tragus or sham stimulation to the earlobe, a site devoid of vagal innervation.
After training, patients delivered the tVNS themselves at a frequency of 20 Hz and pulse width of 200 ms during 1-hour daily sessions over 2 months.
At baseline and 2 months, patients underwent a tilt test to determine postural tachycardia; they remained supine for 25 minutes, followed by 10 minutes of standing, as tolerated.
Researchers used electrocardiogram data to examine heart rate and blood samples to assess serum cytokines and antiautonomic autoantibodies.
The primary outcome was a comparison of orthostatic tachycardia (standing – supine) between the two arms at 2 months.
TAKEAWAY:
At 2 months, postural tachycardia was significantly less in the active vs sham arm (mean postural increase in heart rate 17.6 beats/min vs 31.7 beats/min; P = .01).
There was a significant decrease in beta 1-adrenergic receptor (beta 1-AR; P = .01) and alpha-1-AR (P = .04) autoantibody activity in the active vs sham group, which may account at least in part for the reduced orthostatic tachycardia, although the exact mechanisms for this effect have not been clearly defined, the authors said.
Serum tumor necrosis factor-alpha (TNF-alpha) levels were significantly decreased in the active group relative to the sham group (8.3 pg/mL vs 13.9 pg/mL; P = .01).
As for heart rate variability, change in low frequency (LF) and high frequency (HF) from supine to standing was significantly decreased, and postural change in LF/HF ratio, a surrogate for sympathovagal balance, was significantly lower in the active group compared with the sham group.
IN PRACTICE:
“Collectively, these data suggest that tVNS, a low-cost, low-risk intervention, applied for a short period of time in selected patients with POTS, may result in a significant amelioration of their disease,” the authors conclude.
SOURCE:
The study was led by Stavros Stavrakis, MD, PhD, University of Oklahoma Health Sciences Center, Oklahoma City. It was published online in JACC: Clinical Electrophysiology..
LIMITATIONS:
The study had a small sample size, included only females, and extended only up to 2 months. As there was no improvement on the overall score from the Composite Autonomic Symptom Score 31 (COMPASS-31) questionnaire, researchers can’t conclude tVNS improved patient reported outcomes. The study used 1 hour of daily stimulation but the optimal duration and ideal timing of tVNS is yet to be determined.
DISCLOSURES:
The study was supported by the National Institutes of Health (NIH)/National Heart, Lung, and Blood Institute, NIH/National Institute of General Medical Sciences, and individual donations from Francie Fitzgerald and family through the OU Foundation Fund. The authors have no relevant conflicts of interest.
A version of this article appeared on Medscape.com.
PTSD symptoms in women tied to worse heart, brain health
TOPLINE:
METHODOLOGY:
- Researchers conducted a cross-sectional study of 274 women (mean age, 59 years) participating in the MsBrain study of menopause and brain health.
- As part of the study, the women completed the PTSD Checklist–Civilian Version and underwent physical and neuropsychological testing, as well as carotid artery ultrasonography and brain MRI.
- Outcomes of interest were associations of PTSD symptoms with carotid intima media thickness (IMT), brain white matter hyperintensity volume (WMHV), and cognition, assessed in linear regression models.
- Interactions by APOEε4 were assessed; covariates included age, race/ethnicity, education, and CVD risk factors.
TAKEAWAY:
- Higher PTSD symptoms were associated with greater carotid IMT (P = .03); associations of PTSD symptoms with neurocognitive outcomes varied significantly by APOEε4 status.
- Among APOEε4 carriers, PTSD symptoms were associated with greater whole-brain WMHV (P = .009), periventricular WMHV (P = .02), deep WMHV (P = .01), and frontal WMHV (P = .04) in multivariable models.
- APOEε4 carriers with PTSD symptoms also had poorer cognition, specifically attention and working memory (P = .02), semantic fluency (P = .01), perceptual speed (P = .002) and processing speed (P = .002), in multivariable models.
IN PRACTICE:
“This study sheds important insight on the implications of PTSD symptoms to women’s cardiovascular and neurocognitive health. Our findings indicate that the APOEε4 genotype may identify a group of women with PTSD symptoms at particular risk for poor neurocognitive health,” the authors wrote.
SOURCE:
The study, with first author Rebecca Thurston, PhD, of the department of psychiatry, University of Pittsburgh, was published online in JAMA Network Open.
LIMITATIONS:
No diagnostic clinical interviews were conducted, and PTSD treatment was not assessed. All participants identified as cisgender, and most were non-Hispanic Black or White. The study was observational and cross-sectional, precluding assertions about directionality or causality.
DISCLOSURES:
The study was funded by the National Institutes of Health, the University of Pittsburgh Clinical and Translational Science Institute, and the University of Pittsburgh Small Molecule Biomarker Core. Dr. Thurston reported receiving personal fees from Astellas Pharma, Bayer, Hello Therapeutics, Vira Health, and Happify Health outside the submitted work.
A version of this article first appeared on Medscape.com.
TOPLINE:
METHODOLOGY:
- Researchers conducted a cross-sectional study of 274 women (mean age, 59 years) participating in the MsBrain study of menopause and brain health.
- As part of the study, the women completed the PTSD Checklist–Civilian Version and underwent physical and neuropsychological testing, as well as carotid artery ultrasonography and brain MRI.
- Outcomes of interest were associations of PTSD symptoms with carotid intima media thickness (IMT), brain white matter hyperintensity volume (WMHV), and cognition, assessed in linear regression models.
- Interactions by APOEε4 were assessed; covariates included age, race/ethnicity, education, and CVD risk factors.
TAKEAWAY:
- Higher PTSD symptoms were associated with greater carotid IMT (P = .03); associations of PTSD symptoms with neurocognitive outcomes varied significantly by APOEε4 status.
- Among APOEε4 carriers, PTSD symptoms were associated with greater whole-brain WMHV (P = .009), periventricular WMHV (P = .02), deep WMHV (P = .01), and frontal WMHV (P = .04) in multivariable models.
- APOEε4 carriers with PTSD symptoms also had poorer cognition, specifically attention and working memory (P = .02), semantic fluency (P = .01), perceptual speed (P = .002) and processing speed (P = .002), in multivariable models.
IN PRACTICE:
“This study sheds important insight on the implications of PTSD symptoms to women’s cardiovascular and neurocognitive health. Our findings indicate that the APOEε4 genotype may identify a group of women with PTSD symptoms at particular risk for poor neurocognitive health,” the authors wrote.
SOURCE:
The study, with first author Rebecca Thurston, PhD, of the department of psychiatry, University of Pittsburgh, was published online in JAMA Network Open.
LIMITATIONS:
No diagnostic clinical interviews were conducted, and PTSD treatment was not assessed. All participants identified as cisgender, and most were non-Hispanic Black or White. The study was observational and cross-sectional, precluding assertions about directionality or causality.
DISCLOSURES:
The study was funded by the National Institutes of Health, the University of Pittsburgh Clinical and Translational Science Institute, and the University of Pittsburgh Small Molecule Biomarker Core. Dr. Thurston reported receiving personal fees from Astellas Pharma, Bayer, Hello Therapeutics, Vira Health, and Happify Health outside the submitted work.
A version of this article first appeared on Medscape.com.
TOPLINE:
METHODOLOGY:
- Researchers conducted a cross-sectional study of 274 women (mean age, 59 years) participating in the MsBrain study of menopause and brain health.
- As part of the study, the women completed the PTSD Checklist–Civilian Version and underwent physical and neuropsychological testing, as well as carotid artery ultrasonography and brain MRI.
- Outcomes of interest were associations of PTSD symptoms with carotid intima media thickness (IMT), brain white matter hyperintensity volume (WMHV), and cognition, assessed in linear regression models.
- Interactions by APOEε4 were assessed; covariates included age, race/ethnicity, education, and CVD risk factors.
TAKEAWAY:
- Higher PTSD symptoms were associated with greater carotid IMT (P = .03); associations of PTSD symptoms with neurocognitive outcomes varied significantly by APOEε4 status.
- Among APOEε4 carriers, PTSD symptoms were associated with greater whole-brain WMHV (P = .009), periventricular WMHV (P = .02), deep WMHV (P = .01), and frontal WMHV (P = .04) in multivariable models.
- APOEε4 carriers with PTSD symptoms also had poorer cognition, specifically attention and working memory (P = .02), semantic fluency (P = .01), perceptual speed (P = .002) and processing speed (P = .002), in multivariable models.
IN PRACTICE:
“This study sheds important insight on the implications of PTSD symptoms to women’s cardiovascular and neurocognitive health. Our findings indicate that the APOEε4 genotype may identify a group of women with PTSD symptoms at particular risk for poor neurocognitive health,” the authors wrote.
SOURCE:
The study, with first author Rebecca Thurston, PhD, of the department of psychiatry, University of Pittsburgh, was published online in JAMA Network Open.
LIMITATIONS:
No diagnostic clinical interviews were conducted, and PTSD treatment was not assessed. All participants identified as cisgender, and most were non-Hispanic Black or White. The study was observational and cross-sectional, precluding assertions about directionality or causality.
DISCLOSURES:
The study was funded by the National Institutes of Health, the University of Pittsburgh Clinical and Translational Science Institute, and the University of Pittsburgh Small Molecule Biomarker Core. Dr. Thurston reported receiving personal fees from Astellas Pharma, Bayer, Hello Therapeutics, Vira Health, and Happify Health outside the submitted work.
A version of this article first appeared on Medscape.com.
Bariatric surgery still best option for some with obesity
Bariatric surgery continues to play a major role in obesity management despite the emergence of potent new weight-loss medications, according to two experts who spoke at an Endocrine Society science writers briefing.
“Bariatric surgery is safe, effective, and unfortunately underutilized for treating obesity and its complications,” said Jaime Almandoz, MD, medical director of the Weight Wellness Program at the University of Texas Southwestern Medical Center, Dallas.
Added Dr. Almandoz, who is triple board-certified in internal medicine, endocrinology, and obesity medicine, “Sometimes this gets presented in a linear fashion. ‘We’ll try lifestyle first, and if that doesn’t work, we’ll try medications, and if that doesn’t work, we’ll try surgery.’ But sometimes we might need to go straight to surgery instead of going through medications first, because it may be the most effective and evidence-based treatment for the person in the office in front of you.”
Moreover, he pointed out that currently, Medicare and many private insurers don’t cover antiobesity medications but do cover bariatric surgery.
Indeed, Srividya Kidambi, MD, professor and chief of endocrinology and molecular medicine at the Medical College of Wisconsin/Froedtert Hospital, Milwaukee, said there are certain types of patients for whom she might consider bariatric surgery first. One would be a person with a body mass index (BMI) greater than 40 kg/m2 or with a BMI greater than 35 kg/m2 and severe comorbidities.
Another, she said, would be young, relatively healthy people with obesity who have no comorbid conditions. “We know that if we stop the medication, the weight comes back. So, if I see a 20- to 25-year-old, am I really to commit them to lifelong therapy, or is bariatric surgery a better option in these cases? These drugs have not been around that long ... so I tend to recommend bariatric surgery in some patients.”
During the recent briefing, Dr. Almandoz summarized the evidence base for the benefits of bariatric surgery beyond weight loss, which include remission of type 2 diabetes and fatty liver disease, reduction of the risks of cardiovascular disease and cancer, and increased life expectancy.
“Everyone seems to be talking about GLP-1s for facilitating weight loss and treating obesity. ... What I want to do is provide a counterpoint to accessible therapies that are covered by more insurance plans and that may, in fact, have a better evidence base for treating obesity and its related complications,” he said in his introduction.
Bariatric surgery has been used for decades, and many centers of excellence perform it, with greatly reduced complication rates seen today than in the past. “It’s comparable to having a gallbladder surgery in terms of perioperative risk,” he noted.
Medicare and private insurers generally cover bariatric surgery for people with BMI greater than 40 kg/m2 or 35-39 kg/m2 and at least one weight-related comorbidity, including type 2 diabetes, obstructive sleep apnea, hypertension, atherosclerotic disease, hyperlipidemia, and fatty liver disease.
Data suggest that weight reduction of about 3% can lead to meaningful reductions in blood glucose and triglyceride levels, but weight loss of 15% or greater is associated with reductions in cardiovascular events and type 2 diabetes remission. Lifestyle modification typically produces about 5% weight loss, compared with 20%-35% with bariatric surgery with sleeve gastrectomy or gastric bypass.
Older weight loss medications produced weight loss of 5%-10%; only the newer medications, semaglutide 2.4 mg and tirzepatide, come close to that. Weight loss with semaglutide is about 15%, while tirzepatide can produce weight loss of up to 22%. But, there are still issues with affordability, access, and lack of coverage, Dr. Almandoz noted.
One recent randomized trial of more than 400 individuals showed that bariatric surgery was more effective than lifestyle and medical therapies for treating metabolic-associated steatohepatitis without worsening of fibrosis.
Another showed that the surgery was associated with fewer major adverse liver outcomes among people who already had MASH. That same study showed a 70% reduction in cardiovascular events with bariatric surgery.
For patients with type 2 diabetes, numerous trials have demonstrated long-term remission and reduced A1c at 5 years and 10 years post surgery, along with reductions in microvascular and macrovascular complications.
Other data suggest that a shorter history of type 2 diabetes is among the factors predicting remission with bariatric surgery. “Oftentimes, both patients and providers will wait until the diabetes is quite advanced before they even have the conversation about weight loss or even bariatric surgery. This suggests that if we intervene earlier in the course of disease, when it is less severe and less advanced, we have a higher rate of causing remission in the diabetes,” Dr. Almandoz said.
The American Diabetes Association’s Standards of Care incorporate bariatric surgery as either “recommended” or “may be considered” to treat type 2 diabetes, depending on BMI level, for those who don’t achieve durable weight loss with nonsurgical methods, he noted.
A retrospective cohort study showed significant reductions in cardiovascular outcomes with bariatric surgery among people with baseline cardiovascular disease. “This is not just about bariatric surgery to cause weight loss. This is about the multitude of effects that happen when we treat obesity as a disease with highly effective therapies such as surgery,” he said.
Even cancer risk and cancer-related mortality were significantly reduced with bariatric surgery, another study found.
And in the long-term Swedish Obese Subjects Study, among people with obesity, bariatric surgery was associated with a 3-year increase in life expectancy, compared with not undergoing surgery.
However, Dr. Almandoz also pointed out that some patients may benefit from both weight-loss medication and bariatric surgery. “Once someone has undergone pharmacotherapy, there may still be a role for bariatric procedures in helping to optimize body weight and control body weight long term. And likewise for those who have undergone bariatric surgery, there’s also a role for pharmacotherapy in terms of treating insufficient weight loss or weight recurrence after bariatric surgery. ... So I think there’s clearly a role for integration of therapies.”
Dr. Almandoz serves as consultant/advisory board member for Novo Nordisk, Boehringer Ingelheim, and Eli Lilly. Dr. Kidambi is director of TOPS Center for Metabolic Research and is medical editor of TOPS Magazine, for which her institution receives an honorarium.
A version of this article first appeared on Medscape.com.
Bariatric surgery continues to play a major role in obesity management despite the emergence of potent new weight-loss medications, according to two experts who spoke at an Endocrine Society science writers briefing.
“Bariatric surgery is safe, effective, and unfortunately underutilized for treating obesity and its complications,” said Jaime Almandoz, MD, medical director of the Weight Wellness Program at the University of Texas Southwestern Medical Center, Dallas.
Added Dr. Almandoz, who is triple board-certified in internal medicine, endocrinology, and obesity medicine, “Sometimes this gets presented in a linear fashion. ‘We’ll try lifestyle first, and if that doesn’t work, we’ll try medications, and if that doesn’t work, we’ll try surgery.’ But sometimes we might need to go straight to surgery instead of going through medications first, because it may be the most effective and evidence-based treatment for the person in the office in front of you.”
Moreover, he pointed out that currently, Medicare and many private insurers don’t cover antiobesity medications but do cover bariatric surgery.
Indeed, Srividya Kidambi, MD, professor and chief of endocrinology and molecular medicine at the Medical College of Wisconsin/Froedtert Hospital, Milwaukee, said there are certain types of patients for whom she might consider bariatric surgery first. One would be a person with a body mass index (BMI) greater than 40 kg/m2 or with a BMI greater than 35 kg/m2 and severe comorbidities.
Another, she said, would be young, relatively healthy people with obesity who have no comorbid conditions. “We know that if we stop the medication, the weight comes back. So, if I see a 20- to 25-year-old, am I really to commit them to lifelong therapy, or is bariatric surgery a better option in these cases? These drugs have not been around that long ... so I tend to recommend bariatric surgery in some patients.”
During the recent briefing, Dr. Almandoz summarized the evidence base for the benefits of bariatric surgery beyond weight loss, which include remission of type 2 diabetes and fatty liver disease, reduction of the risks of cardiovascular disease and cancer, and increased life expectancy.
“Everyone seems to be talking about GLP-1s for facilitating weight loss and treating obesity. ... What I want to do is provide a counterpoint to accessible therapies that are covered by more insurance plans and that may, in fact, have a better evidence base for treating obesity and its related complications,” he said in his introduction.
Bariatric surgery has been used for decades, and many centers of excellence perform it, with greatly reduced complication rates seen today than in the past. “It’s comparable to having a gallbladder surgery in terms of perioperative risk,” he noted.
Medicare and private insurers generally cover bariatric surgery for people with BMI greater than 40 kg/m2 or 35-39 kg/m2 and at least one weight-related comorbidity, including type 2 diabetes, obstructive sleep apnea, hypertension, atherosclerotic disease, hyperlipidemia, and fatty liver disease.
Data suggest that weight reduction of about 3% can lead to meaningful reductions in blood glucose and triglyceride levels, but weight loss of 15% or greater is associated with reductions in cardiovascular events and type 2 diabetes remission. Lifestyle modification typically produces about 5% weight loss, compared with 20%-35% with bariatric surgery with sleeve gastrectomy or gastric bypass.
Older weight loss medications produced weight loss of 5%-10%; only the newer medications, semaglutide 2.4 mg and tirzepatide, come close to that. Weight loss with semaglutide is about 15%, while tirzepatide can produce weight loss of up to 22%. But, there are still issues with affordability, access, and lack of coverage, Dr. Almandoz noted.
One recent randomized trial of more than 400 individuals showed that bariatric surgery was more effective than lifestyle and medical therapies for treating metabolic-associated steatohepatitis without worsening of fibrosis.
Another showed that the surgery was associated with fewer major adverse liver outcomes among people who already had MASH. That same study showed a 70% reduction in cardiovascular events with bariatric surgery.
For patients with type 2 diabetes, numerous trials have demonstrated long-term remission and reduced A1c at 5 years and 10 years post surgery, along with reductions in microvascular and macrovascular complications.
Other data suggest that a shorter history of type 2 diabetes is among the factors predicting remission with bariatric surgery. “Oftentimes, both patients and providers will wait until the diabetes is quite advanced before they even have the conversation about weight loss or even bariatric surgery. This suggests that if we intervene earlier in the course of disease, when it is less severe and less advanced, we have a higher rate of causing remission in the diabetes,” Dr. Almandoz said.
The American Diabetes Association’s Standards of Care incorporate bariatric surgery as either “recommended” or “may be considered” to treat type 2 diabetes, depending on BMI level, for those who don’t achieve durable weight loss with nonsurgical methods, he noted.
A retrospective cohort study showed significant reductions in cardiovascular outcomes with bariatric surgery among people with baseline cardiovascular disease. “This is not just about bariatric surgery to cause weight loss. This is about the multitude of effects that happen when we treat obesity as a disease with highly effective therapies such as surgery,” he said.
Even cancer risk and cancer-related mortality were significantly reduced with bariatric surgery, another study found.
And in the long-term Swedish Obese Subjects Study, among people with obesity, bariatric surgery was associated with a 3-year increase in life expectancy, compared with not undergoing surgery.
However, Dr. Almandoz also pointed out that some patients may benefit from both weight-loss medication and bariatric surgery. “Once someone has undergone pharmacotherapy, there may still be a role for bariatric procedures in helping to optimize body weight and control body weight long term. And likewise for those who have undergone bariatric surgery, there’s also a role for pharmacotherapy in terms of treating insufficient weight loss or weight recurrence after bariatric surgery. ... So I think there’s clearly a role for integration of therapies.”
Dr. Almandoz serves as consultant/advisory board member for Novo Nordisk, Boehringer Ingelheim, and Eli Lilly. Dr. Kidambi is director of TOPS Center for Metabolic Research and is medical editor of TOPS Magazine, for which her institution receives an honorarium.
A version of this article first appeared on Medscape.com.
Bariatric surgery continues to play a major role in obesity management despite the emergence of potent new weight-loss medications, according to two experts who spoke at an Endocrine Society science writers briefing.
“Bariatric surgery is safe, effective, and unfortunately underutilized for treating obesity and its complications,” said Jaime Almandoz, MD, medical director of the Weight Wellness Program at the University of Texas Southwestern Medical Center, Dallas.
Added Dr. Almandoz, who is triple board-certified in internal medicine, endocrinology, and obesity medicine, “Sometimes this gets presented in a linear fashion. ‘We’ll try lifestyle first, and if that doesn’t work, we’ll try medications, and if that doesn’t work, we’ll try surgery.’ But sometimes we might need to go straight to surgery instead of going through medications first, because it may be the most effective and evidence-based treatment for the person in the office in front of you.”
Moreover, he pointed out that currently, Medicare and many private insurers don’t cover antiobesity medications but do cover bariatric surgery.
Indeed, Srividya Kidambi, MD, professor and chief of endocrinology and molecular medicine at the Medical College of Wisconsin/Froedtert Hospital, Milwaukee, said there are certain types of patients for whom she might consider bariatric surgery first. One would be a person with a body mass index (BMI) greater than 40 kg/m2 or with a BMI greater than 35 kg/m2 and severe comorbidities.
Another, she said, would be young, relatively healthy people with obesity who have no comorbid conditions. “We know that if we stop the medication, the weight comes back. So, if I see a 20- to 25-year-old, am I really to commit them to lifelong therapy, or is bariatric surgery a better option in these cases? These drugs have not been around that long ... so I tend to recommend bariatric surgery in some patients.”
During the recent briefing, Dr. Almandoz summarized the evidence base for the benefits of bariatric surgery beyond weight loss, which include remission of type 2 diabetes and fatty liver disease, reduction of the risks of cardiovascular disease and cancer, and increased life expectancy.
“Everyone seems to be talking about GLP-1s for facilitating weight loss and treating obesity. ... What I want to do is provide a counterpoint to accessible therapies that are covered by more insurance plans and that may, in fact, have a better evidence base for treating obesity and its related complications,” he said in his introduction.
Bariatric surgery has been used for decades, and many centers of excellence perform it, with greatly reduced complication rates seen today than in the past. “It’s comparable to having a gallbladder surgery in terms of perioperative risk,” he noted.
Medicare and private insurers generally cover bariatric surgery for people with BMI greater than 40 kg/m2 or 35-39 kg/m2 and at least one weight-related comorbidity, including type 2 diabetes, obstructive sleep apnea, hypertension, atherosclerotic disease, hyperlipidemia, and fatty liver disease.
Data suggest that weight reduction of about 3% can lead to meaningful reductions in blood glucose and triglyceride levels, but weight loss of 15% or greater is associated with reductions in cardiovascular events and type 2 diabetes remission. Lifestyle modification typically produces about 5% weight loss, compared with 20%-35% with bariatric surgery with sleeve gastrectomy or gastric bypass.
Older weight loss medications produced weight loss of 5%-10%; only the newer medications, semaglutide 2.4 mg and tirzepatide, come close to that. Weight loss with semaglutide is about 15%, while tirzepatide can produce weight loss of up to 22%. But, there are still issues with affordability, access, and lack of coverage, Dr. Almandoz noted.
One recent randomized trial of more than 400 individuals showed that bariatric surgery was more effective than lifestyle and medical therapies for treating metabolic-associated steatohepatitis without worsening of fibrosis.
Another showed that the surgery was associated with fewer major adverse liver outcomes among people who already had MASH. That same study showed a 70% reduction in cardiovascular events with bariatric surgery.
For patients with type 2 diabetes, numerous trials have demonstrated long-term remission and reduced A1c at 5 years and 10 years post surgery, along with reductions in microvascular and macrovascular complications.
Other data suggest that a shorter history of type 2 diabetes is among the factors predicting remission with bariatric surgery. “Oftentimes, both patients and providers will wait until the diabetes is quite advanced before they even have the conversation about weight loss or even bariatric surgery. This suggests that if we intervene earlier in the course of disease, when it is less severe and less advanced, we have a higher rate of causing remission in the diabetes,” Dr. Almandoz said.
The American Diabetes Association’s Standards of Care incorporate bariatric surgery as either “recommended” or “may be considered” to treat type 2 diabetes, depending on BMI level, for those who don’t achieve durable weight loss with nonsurgical methods, he noted.
A retrospective cohort study showed significant reductions in cardiovascular outcomes with bariatric surgery among people with baseline cardiovascular disease. “This is not just about bariatric surgery to cause weight loss. This is about the multitude of effects that happen when we treat obesity as a disease with highly effective therapies such as surgery,” he said.
Even cancer risk and cancer-related mortality were significantly reduced with bariatric surgery, another study found.
And in the long-term Swedish Obese Subjects Study, among people with obesity, bariatric surgery was associated with a 3-year increase in life expectancy, compared with not undergoing surgery.
However, Dr. Almandoz also pointed out that some patients may benefit from both weight-loss medication and bariatric surgery. “Once someone has undergone pharmacotherapy, there may still be a role for bariatric procedures in helping to optimize body weight and control body weight long term. And likewise for those who have undergone bariatric surgery, there’s also a role for pharmacotherapy in terms of treating insufficient weight loss or weight recurrence after bariatric surgery. ... So I think there’s clearly a role for integration of therapies.”
Dr. Almandoz serves as consultant/advisory board member for Novo Nordisk, Boehringer Ingelheim, and Eli Lilly. Dr. Kidambi is director of TOPS Center for Metabolic Research and is medical editor of TOPS Magazine, for which her institution receives an honorarium.
A version of this article first appeared on Medscape.com.
Long-term use of ADHD meds and CVD risk: New data
results of a large Swedish nested case-control study suggest.
The increased risk was evident only for hypertension and arterial disease, was dose dependent, and was higher for stimulant than nonstimulant ADHD medications.
“Clinicians should be vigilant in monitoring signs and symptoms of cardiovascular diseases, particularly among those receiving higher doses,” Zheng Chang, PhD, principal researcher, department of medical epidemiology and biostatistics, Karolinska Institutet, Stockholm, said in an interview.
“Treatment decisions, as always, should be based on careful weighing of potential benefits and risks at individual patient level, rather than simple one-size-fits-all recommendations,” Dr. Chang added.
The study was published online in JAMA Psychiatry
Filling in the research gaps
The use of medications to treat ADHD has increased markedly over the past decades in both children and adults. The potential risk for CVD associated with long-term ADHD medication use remains unclear. Most “longitudinal” studies that have looked at the association have an average follow-up time of no more than 2 years, the authors note.
In contrast, the Swedish study assessed the association between cumulative use of ADHD medication in children and adults followed for up to 14 years and also looked at whether associations differ across types of medication and dosages, types of CVD, gender, and age.
Among 278,027 individuals aged 6-64 years diagnosed with ADHD or dispensed ADHD medication, 10,388 with CVD were identified and matched to 51,672 controls without CVD.
Longer cumulative duration of ADHD medication use was associated with a statistically significant increased risk for CVD, compared with no use.
When the risk for specific CVDs was examined, long-term use of ADHD medication (compared with no use) was associated with an increased risk for hypertension and arterial disease but not arrhythmias, heart failure, ischemic heart disease, thromboembolic disease, or cerebrovascular disease.
For hypertension, the adjusted odds ratio was 1.72 (95% confidence interval, 1.51-1.97) for 3 to ≤ 5 years and 1.80 (95% CI, 1.55-2.08) for > 5 years of medication use. For arterial disease, the AOR was 1.65 (95% CI, 1.11-2.45) for 3 to ≤ 5 years and 1.49 (95% CI, 0.96-2.32) for > 5 years of use.
Stimulants confer greatest risk
Across the 14-year follow-up period, each additional year of ADHD medication use was associated with an average 4% increased CVD risk, with a larger 8% increased risk in the first 3 years of cumulative use, followed by stable risk over the remaining follow-up.
Similar risks were observed in children and adults, as well as in females and males.
When focusing on specific ADHD medications, compared with no use, long-term use of the stimulant methylphenidate was associated with an increased risk for CVD (AOR, 1.20 [95% CI, 1.10-1.31] for 3 to ≤ 5 years and 1.19 [95% CI, 1.08-1.31] for > 5 years).
The same was true for long-term use of the stimulant lisdexamfetamine (AOR, 1.23 [95% CI, 1.05-1.44] for 2 to ≤ 3 years and 1.17 [95% CI, 0.98-1.40] for > 3 years).
In contrast, use of the nonstimulant atomoxetine was associated with elevated CVD risk only for the first year of use (AOR, 1.07; 95% CI, 1.01-1.13).
The increased risk for CVD occurred only above certain average daily doses: 45 mg for methylphenidate and lisdexamfetamine, 22.5 mg for amphetamines, and 120 mg for atomoxetine.
The authors note that, although they accounted for a wide range of potential confounding variables, considering the observational nature of the study and the possibility of residual confounding, they could not prove causality.
‘Tricky trade-offs’
The coauthors of an editorial in JAMA Psychiatry (2023 Nov 22. doi: 10.1001/jamapsychiatry.2023.4126) note that the study “should remind us that clinical decision-making is often based on tricky trade-offs that should be considered at the individual patient level.”
Given that hypertension is the leading cause of CV morbidity and mortality worldwide, the increased likelihood of hypertension with long-term use of ADHD medications “cannot be disregarded,” write Samuele Cortese, MD, PhD, and Cristiano Fava, MD, PhD, with University of Southampton (England).
“These findings are especially relevant given the reported association between ADHD and physical conditions, such as obesity, which further contribute to increased cardiovascular risk,” they add.
Dr. Cortese and Dr. Fava say that the increased CV risk – averaging 4% per year and stabilizing after 3 years of treatment – “should be carefully weighed against the established benefits, on a case-by-case basis.”
“Importantly,” they write, “large real-world self-controlled studies have shown that individuals with ADHD experience significantly fewer unintentional physical injuries, motor vehicle crashes, substance use disorders, and criminal acts, as well as improved academic functioning, during periods when they are taking, compared with periods when they are not taking, methylphenidate.”
The risk-benefit ratio, however, may be lower in people with preexisting heart conditions. However, more evidence and precise recommendations are needed in relation to the treatment of individuals with ADHD and preexisting CV conditions, the editorial writers say.
This study was supported by grants from the Swedish Research Council for Health, Working Life, and Welfare and the European Union’s Horizon 2020 research and innovation program. The authors and editorial writers have no relevant conflicts of interest.
A version of this article appeared on Medscape.com.
results of a large Swedish nested case-control study suggest.
The increased risk was evident only for hypertension and arterial disease, was dose dependent, and was higher for stimulant than nonstimulant ADHD medications.
“Clinicians should be vigilant in monitoring signs and symptoms of cardiovascular diseases, particularly among those receiving higher doses,” Zheng Chang, PhD, principal researcher, department of medical epidemiology and biostatistics, Karolinska Institutet, Stockholm, said in an interview.
“Treatment decisions, as always, should be based on careful weighing of potential benefits and risks at individual patient level, rather than simple one-size-fits-all recommendations,” Dr. Chang added.
The study was published online in JAMA Psychiatry
Filling in the research gaps
The use of medications to treat ADHD has increased markedly over the past decades in both children and adults. The potential risk for CVD associated with long-term ADHD medication use remains unclear. Most “longitudinal” studies that have looked at the association have an average follow-up time of no more than 2 years, the authors note.
In contrast, the Swedish study assessed the association between cumulative use of ADHD medication in children and adults followed for up to 14 years and also looked at whether associations differ across types of medication and dosages, types of CVD, gender, and age.
Among 278,027 individuals aged 6-64 years diagnosed with ADHD or dispensed ADHD medication, 10,388 with CVD were identified and matched to 51,672 controls without CVD.
Longer cumulative duration of ADHD medication use was associated with a statistically significant increased risk for CVD, compared with no use.
When the risk for specific CVDs was examined, long-term use of ADHD medication (compared with no use) was associated with an increased risk for hypertension and arterial disease but not arrhythmias, heart failure, ischemic heart disease, thromboembolic disease, or cerebrovascular disease.
For hypertension, the adjusted odds ratio was 1.72 (95% confidence interval, 1.51-1.97) for 3 to ≤ 5 years and 1.80 (95% CI, 1.55-2.08) for > 5 years of medication use. For arterial disease, the AOR was 1.65 (95% CI, 1.11-2.45) for 3 to ≤ 5 years and 1.49 (95% CI, 0.96-2.32) for > 5 years of use.
Stimulants confer greatest risk
Across the 14-year follow-up period, each additional year of ADHD medication use was associated with an average 4% increased CVD risk, with a larger 8% increased risk in the first 3 years of cumulative use, followed by stable risk over the remaining follow-up.
Similar risks were observed in children and adults, as well as in females and males.
When focusing on specific ADHD medications, compared with no use, long-term use of the stimulant methylphenidate was associated with an increased risk for CVD (AOR, 1.20 [95% CI, 1.10-1.31] for 3 to ≤ 5 years and 1.19 [95% CI, 1.08-1.31] for > 5 years).
The same was true for long-term use of the stimulant lisdexamfetamine (AOR, 1.23 [95% CI, 1.05-1.44] for 2 to ≤ 3 years and 1.17 [95% CI, 0.98-1.40] for > 3 years).
In contrast, use of the nonstimulant atomoxetine was associated with elevated CVD risk only for the first year of use (AOR, 1.07; 95% CI, 1.01-1.13).
The increased risk for CVD occurred only above certain average daily doses: 45 mg for methylphenidate and lisdexamfetamine, 22.5 mg for amphetamines, and 120 mg for atomoxetine.
The authors note that, although they accounted for a wide range of potential confounding variables, considering the observational nature of the study and the possibility of residual confounding, they could not prove causality.
‘Tricky trade-offs’
The coauthors of an editorial in JAMA Psychiatry (2023 Nov 22. doi: 10.1001/jamapsychiatry.2023.4126) note that the study “should remind us that clinical decision-making is often based on tricky trade-offs that should be considered at the individual patient level.”
Given that hypertension is the leading cause of CV morbidity and mortality worldwide, the increased likelihood of hypertension with long-term use of ADHD medications “cannot be disregarded,” write Samuele Cortese, MD, PhD, and Cristiano Fava, MD, PhD, with University of Southampton (England).
“These findings are especially relevant given the reported association between ADHD and physical conditions, such as obesity, which further contribute to increased cardiovascular risk,” they add.
Dr. Cortese and Dr. Fava say that the increased CV risk – averaging 4% per year and stabilizing after 3 years of treatment – “should be carefully weighed against the established benefits, on a case-by-case basis.”
“Importantly,” they write, “large real-world self-controlled studies have shown that individuals with ADHD experience significantly fewer unintentional physical injuries, motor vehicle crashes, substance use disorders, and criminal acts, as well as improved academic functioning, during periods when they are taking, compared with periods when they are not taking, methylphenidate.”
The risk-benefit ratio, however, may be lower in people with preexisting heart conditions. However, more evidence and precise recommendations are needed in relation to the treatment of individuals with ADHD and preexisting CV conditions, the editorial writers say.
This study was supported by grants from the Swedish Research Council for Health, Working Life, and Welfare and the European Union’s Horizon 2020 research and innovation program. The authors and editorial writers have no relevant conflicts of interest.
A version of this article appeared on Medscape.com.
results of a large Swedish nested case-control study suggest.
The increased risk was evident only for hypertension and arterial disease, was dose dependent, and was higher for stimulant than nonstimulant ADHD medications.
“Clinicians should be vigilant in monitoring signs and symptoms of cardiovascular diseases, particularly among those receiving higher doses,” Zheng Chang, PhD, principal researcher, department of medical epidemiology and biostatistics, Karolinska Institutet, Stockholm, said in an interview.
“Treatment decisions, as always, should be based on careful weighing of potential benefits and risks at individual patient level, rather than simple one-size-fits-all recommendations,” Dr. Chang added.
The study was published online in JAMA Psychiatry
Filling in the research gaps
The use of medications to treat ADHD has increased markedly over the past decades in both children and adults. The potential risk for CVD associated with long-term ADHD medication use remains unclear. Most “longitudinal” studies that have looked at the association have an average follow-up time of no more than 2 years, the authors note.
In contrast, the Swedish study assessed the association between cumulative use of ADHD medication in children and adults followed for up to 14 years and also looked at whether associations differ across types of medication and dosages, types of CVD, gender, and age.
Among 278,027 individuals aged 6-64 years diagnosed with ADHD or dispensed ADHD medication, 10,388 with CVD were identified and matched to 51,672 controls without CVD.
Longer cumulative duration of ADHD medication use was associated with a statistically significant increased risk for CVD, compared with no use.
When the risk for specific CVDs was examined, long-term use of ADHD medication (compared with no use) was associated with an increased risk for hypertension and arterial disease but not arrhythmias, heart failure, ischemic heart disease, thromboembolic disease, or cerebrovascular disease.
For hypertension, the adjusted odds ratio was 1.72 (95% confidence interval, 1.51-1.97) for 3 to ≤ 5 years and 1.80 (95% CI, 1.55-2.08) for > 5 years of medication use. For arterial disease, the AOR was 1.65 (95% CI, 1.11-2.45) for 3 to ≤ 5 years and 1.49 (95% CI, 0.96-2.32) for > 5 years of use.
Stimulants confer greatest risk
Across the 14-year follow-up period, each additional year of ADHD medication use was associated with an average 4% increased CVD risk, with a larger 8% increased risk in the first 3 years of cumulative use, followed by stable risk over the remaining follow-up.
Similar risks were observed in children and adults, as well as in females and males.
When focusing on specific ADHD medications, compared with no use, long-term use of the stimulant methylphenidate was associated with an increased risk for CVD (AOR, 1.20 [95% CI, 1.10-1.31] for 3 to ≤ 5 years and 1.19 [95% CI, 1.08-1.31] for > 5 years).
The same was true for long-term use of the stimulant lisdexamfetamine (AOR, 1.23 [95% CI, 1.05-1.44] for 2 to ≤ 3 years and 1.17 [95% CI, 0.98-1.40] for > 3 years).
In contrast, use of the nonstimulant atomoxetine was associated with elevated CVD risk only for the first year of use (AOR, 1.07; 95% CI, 1.01-1.13).
The increased risk for CVD occurred only above certain average daily doses: 45 mg for methylphenidate and lisdexamfetamine, 22.5 mg for amphetamines, and 120 mg for atomoxetine.
The authors note that, although they accounted for a wide range of potential confounding variables, considering the observational nature of the study and the possibility of residual confounding, they could not prove causality.
‘Tricky trade-offs’
The coauthors of an editorial in JAMA Psychiatry (2023 Nov 22. doi: 10.1001/jamapsychiatry.2023.4126) note that the study “should remind us that clinical decision-making is often based on tricky trade-offs that should be considered at the individual patient level.”
Given that hypertension is the leading cause of CV morbidity and mortality worldwide, the increased likelihood of hypertension with long-term use of ADHD medications “cannot be disregarded,” write Samuele Cortese, MD, PhD, and Cristiano Fava, MD, PhD, with University of Southampton (England).
“These findings are especially relevant given the reported association between ADHD and physical conditions, such as obesity, which further contribute to increased cardiovascular risk,” they add.
Dr. Cortese and Dr. Fava say that the increased CV risk – averaging 4% per year and stabilizing after 3 years of treatment – “should be carefully weighed against the established benefits, on a case-by-case basis.”
“Importantly,” they write, “large real-world self-controlled studies have shown that individuals with ADHD experience significantly fewer unintentional physical injuries, motor vehicle crashes, substance use disorders, and criminal acts, as well as improved academic functioning, during periods when they are taking, compared with periods when they are not taking, methylphenidate.”
The risk-benefit ratio, however, may be lower in people with preexisting heart conditions. However, more evidence and precise recommendations are needed in relation to the treatment of individuals with ADHD and preexisting CV conditions, the editorial writers say.
This study was supported by grants from the Swedish Research Council for Health, Working Life, and Welfare and the European Union’s Horizon 2020 research and innovation program. The authors and editorial writers have no relevant conflicts of interest.
A version of this article appeared on Medscape.com.
FROM JAMA PSYCHIATRY
‘Smart’ stethoscope spots peripartum cardiomyopathy
in a large study of obstetric patients in Nigeria.
Demilade A. Adedinsewo, MD, MPH, from Mayo Clinic, Jacksonville, Fla., reported these findings from the Screening for Pregnancy Related Heart Failure in Nigeria (SPEC-AI Nigeria) trial in a press briefing and in a late-breaking trial session at the annual scientific sessions of the American Heart Association.
“The key takeaway,” Dr. Adedinsewo said in an interview, “is recognizing that a simple, low-impact tool like a digital stethoscope can dramatically improve the diagnosis of a life-threatening condition, and we can treat it. A large proportion of the women will recover; if we identify them early and treat them appropriately, we can reduce the risk of dying.”
If the device predicted low ejection fraction, the patient went on to have an echocardiogram to confirm cardiomyopathy, defined as a left ventricular ejection fraction (LVEF) <50%.
Peripartum cardiomyopathy was detected in 4% of the women who were screened with this tool, compared with 1.8% of those who received usual care, which included a traditional ECG.
“I believe that the control arm also has about 4% of cardiomyopathy cases, but because they didn’t have the same screening and echo, we’re missing them,” Dr. Adedinsewo said.
Diagnosis of peripartum cardiomyopathy is challenging, she noted, owing to overlap of common symptoms in pregnancy, such as lower-extremity swelling, fatigue, and shortness of breath with mild activity, which are also cardinal symptoms of heart failure.
“We were really impressed by the effectiveness of the tool, looking at how accurate it was when it comes to the sensitivity,” she added. She noted that the digital stethoscope correctly identified 92% of women with LVEF < 50% and 100% of those with LVEF < 40%.
This was the first large, clinical trial to evaluate an AI intervention in pregnancy. The investigators used a portable, battery-operated device that yielded AI results in real time.
Nigeria has the highest rate of pericardium cardiomyopathy of any country. However, one study showed a 16-fold higher rate of cardiomyopathy among African American women, compared with White women in the United States, Dr. Adedinsewo noted. “It will be important to identify who we should be screening to identify more cases,” she said.
A digital stethoscope that provides an ECG is currently available, but the algorithm that powers detection of cardiomyopathy is not yet commercially available.
Findings ‘absolutely startling’
The study discussant in the press briefing, Alexander Tarlochan Singh Sandhu, MD, from Stanford (Calif.) University congratulated the authors on this “valuable study that uses AI tools to solve a real health problem.”
Finding that 4% of the women in the intervention arm had reduced ejection fraction is “absolutely startling,” he said, “and speaks to how important improving our diagnosis in this space is.
“Where the burden of disease is high, a tool like this can be so incredibly valuable,” he said. He noted that the investigators identified 2% more patients with peripartum cardiomyopathy.
“This is an example of the potential of AI tools that can actually improve access to care and improve quality of care in resource-limited settings,” he said. “We need to move to understanding how to implement this into subsequent care [and] figure out what the next steps are to improve their outcomes.”
“The main takeaway is that, in areas where there is a very high prevalence of a morbid condition, a prescreening tool like this may be helpful” for diagnosis, the assigned discussant in the session, Marco Perez, MD, also from Stanford University, told this news organization.
The number of women needed to screen to detect peripartum cardiomyopathy by echocardiography alone is 1 in 23 in Nigeria and 1 in 970 in the United States, he said.
With an AI tool such as this one (sensitivity, 92%; specificity, 80%), the number needed to screen would be 1 in 5.7 in Nigeria and 1 in 194 in the United States, he estimates on the basis of incidence data.
“Because it is so common in Nigeria, a screening method makes a lot of sense,” Dr. Perez said. “The big question that remains is, what is the best screening modality?
“Certainly, this tool helped in bringing down the number of echoes needed to find a case, from the mid 20s down to about 5 or 6, so it certainly does seem to be helpful.”
However, the investigators did not say whether this tool is better than a clinical review of ECG or an AI analysis of ECG alone. It’s not clear whether the phonocardiogram component is significant in conjunction with the ECG component.
Nevertheless, “In a place where there’s a very high prevalence of peripartum cardiomyopathy, like Haiti, like Nigeria, doing something like this makes a lot of sense.
“For the U.S. and the rest of the world, where the prevalence is much lower, even with a tool like this you still would need to do a lot of echoes to find one case, and that may end up not being cost-effective. You would need to screen 200 women with echo to find one case.”
AI-guided screening study
Nigeria has the highest reported incidence of peripartum cardiomyopathy mortality (1 in 100 live births) and the highest number of maternal deaths.
In the United States, where rates of peripartum cardiomyopathy are much lower, maternal deaths are nevertheless higher than in other developed countries and have trended up over the past 3 decades; cardiomyopathy is a key contributor.
The investigators enrolled 1,195 women who were pregnant or had given birth in the past 12 months. The patients were from six teaching hospitals in Nigeria (two in the north and four in the south). They were randomly assigned in a 1:1 ratio to the intervention group (587) or the control group (608).
In the intervention group, clinicians used a smart stethoscope to record a phonocardiogram and a single-lead ECG reading in the V2 position and in an angled position on the patient’s chest wall and to record an ECG from the patient’s fingers. The recordings were sent to a Bluetooth-enabled mobile device (tablet or smartphone), which displayed the phonocardiogram and ECG images and that indicated whether the ejection fraction was normal or low. All patients in the intervention group received an echocardiogram.
In the control group, patients received usual care plus a traditional ECG. They were not required to have an echocardiogram because undergoing an echocardiogram is not part of usual care; however, they could receive an echocardiogram if the ECG suggested that they might need further testing.
The mean age of all the patients was 31 years, and all were Black. At study entry, 73% were pregnant, and 26% were post partum. They had similar comorbidities.
The primary outcome, cardiomyopathy (LVEF <50%) was detected in 24 of 587 patients (4.1%) in the intervention group and in 11 of 608 patients (1.8%) in the control group (odds ratio, 2.3; 95% confidence interval, 1.1-4.8; P = .02).
For the detection of LVEF <50%, the sensitivity was 92% and the specificity was 80%. For the detection of LVEF <40% (a secondary outcome), the sensitivity was 100% and the specificity was 79%.
Dr. Adedinsewo is supported by the Mayo Building Interdisciplinary Research Careers in Women’s Health (BIRCWH) Program, which is funded by the National Institutes of Health. The trial was funded by Mayo Clinic (Centers for Digital Health and Community Health and Engagement Research) and in part by the Mayo Clinic BIRCWH Program. Portable ECG, phonocardiogram recordings, and AI predictions using the digital stethoscope were extracted by the Eko Health team and were sent to the coordinating center for analysis. Eko Health had no role in study design, data collection, data analysis, or data interpretation.
A version of this article appeared on Medscape.com.
in a large study of obstetric patients in Nigeria.
Demilade A. Adedinsewo, MD, MPH, from Mayo Clinic, Jacksonville, Fla., reported these findings from the Screening for Pregnancy Related Heart Failure in Nigeria (SPEC-AI Nigeria) trial in a press briefing and in a late-breaking trial session at the annual scientific sessions of the American Heart Association.
“The key takeaway,” Dr. Adedinsewo said in an interview, “is recognizing that a simple, low-impact tool like a digital stethoscope can dramatically improve the diagnosis of a life-threatening condition, and we can treat it. A large proportion of the women will recover; if we identify them early and treat them appropriately, we can reduce the risk of dying.”
If the device predicted low ejection fraction, the patient went on to have an echocardiogram to confirm cardiomyopathy, defined as a left ventricular ejection fraction (LVEF) <50%.
Peripartum cardiomyopathy was detected in 4% of the women who were screened with this tool, compared with 1.8% of those who received usual care, which included a traditional ECG.
“I believe that the control arm also has about 4% of cardiomyopathy cases, but because they didn’t have the same screening and echo, we’re missing them,” Dr. Adedinsewo said.
Diagnosis of peripartum cardiomyopathy is challenging, she noted, owing to overlap of common symptoms in pregnancy, such as lower-extremity swelling, fatigue, and shortness of breath with mild activity, which are also cardinal symptoms of heart failure.
“We were really impressed by the effectiveness of the tool, looking at how accurate it was when it comes to the sensitivity,” she added. She noted that the digital stethoscope correctly identified 92% of women with LVEF < 50% and 100% of those with LVEF < 40%.
This was the first large, clinical trial to evaluate an AI intervention in pregnancy. The investigators used a portable, battery-operated device that yielded AI results in real time.
Nigeria has the highest rate of pericardium cardiomyopathy of any country. However, one study showed a 16-fold higher rate of cardiomyopathy among African American women, compared with White women in the United States, Dr. Adedinsewo noted. “It will be important to identify who we should be screening to identify more cases,” she said.
A digital stethoscope that provides an ECG is currently available, but the algorithm that powers detection of cardiomyopathy is not yet commercially available.
Findings ‘absolutely startling’
The study discussant in the press briefing, Alexander Tarlochan Singh Sandhu, MD, from Stanford (Calif.) University congratulated the authors on this “valuable study that uses AI tools to solve a real health problem.”
Finding that 4% of the women in the intervention arm had reduced ejection fraction is “absolutely startling,” he said, “and speaks to how important improving our diagnosis in this space is.
“Where the burden of disease is high, a tool like this can be so incredibly valuable,” he said. He noted that the investigators identified 2% more patients with peripartum cardiomyopathy.
“This is an example of the potential of AI tools that can actually improve access to care and improve quality of care in resource-limited settings,” he said. “We need to move to understanding how to implement this into subsequent care [and] figure out what the next steps are to improve their outcomes.”
“The main takeaway is that, in areas where there is a very high prevalence of a morbid condition, a prescreening tool like this may be helpful” for diagnosis, the assigned discussant in the session, Marco Perez, MD, also from Stanford University, told this news organization.
The number of women needed to screen to detect peripartum cardiomyopathy by echocardiography alone is 1 in 23 in Nigeria and 1 in 970 in the United States, he said.
With an AI tool such as this one (sensitivity, 92%; specificity, 80%), the number needed to screen would be 1 in 5.7 in Nigeria and 1 in 194 in the United States, he estimates on the basis of incidence data.
“Because it is so common in Nigeria, a screening method makes a lot of sense,” Dr. Perez said. “The big question that remains is, what is the best screening modality?
“Certainly, this tool helped in bringing down the number of echoes needed to find a case, from the mid 20s down to about 5 or 6, so it certainly does seem to be helpful.”
However, the investigators did not say whether this tool is better than a clinical review of ECG or an AI analysis of ECG alone. It’s not clear whether the phonocardiogram component is significant in conjunction with the ECG component.
Nevertheless, “In a place where there’s a very high prevalence of peripartum cardiomyopathy, like Haiti, like Nigeria, doing something like this makes a lot of sense.
“For the U.S. and the rest of the world, where the prevalence is much lower, even with a tool like this you still would need to do a lot of echoes to find one case, and that may end up not being cost-effective. You would need to screen 200 women with echo to find one case.”
AI-guided screening study
Nigeria has the highest reported incidence of peripartum cardiomyopathy mortality (1 in 100 live births) and the highest number of maternal deaths.
In the United States, where rates of peripartum cardiomyopathy are much lower, maternal deaths are nevertheless higher than in other developed countries and have trended up over the past 3 decades; cardiomyopathy is a key contributor.
The investigators enrolled 1,195 women who were pregnant or had given birth in the past 12 months. The patients were from six teaching hospitals in Nigeria (two in the north and four in the south). They were randomly assigned in a 1:1 ratio to the intervention group (587) or the control group (608).
In the intervention group, clinicians used a smart stethoscope to record a phonocardiogram and a single-lead ECG reading in the V2 position and in an angled position on the patient’s chest wall and to record an ECG from the patient’s fingers. The recordings were sent to a Bluetooth-enabled mobile device (tablet or smartphone), which displayed the phonocardiogram and ECG images and that indicated whether the ejection fraction was normal or low. All patients in the intervention group received an echocardiogram.
In the control group, patients received usual care plus a traditional ECG. They were not required to have an echocardiogram because undergoing an echocardiogram is not part of usual care; however, they could receive an echocardiogram if the ECG suggested that they might need further testing.
The mean age of all the patients was 31 years, and all were Black. At study entry, 73% were pregnant, and 26% were post partum. They had similar comorbidities.
The primary outcome, cardiomyopathy (LVEF <50%) was detected in 24 of 587 patients (4.1%) in the intervention group and in 11 of 608 patients (1.8%) in the control group (odds ratio, 2.3; 95% confidence interval, 1.1-4.8; P = .02).
For the detection of LVEF <50%, the sensitivity was 92% and the specificity was 80%. For the detection of LVEF <40% (a secondary outcome), the sensitivity was 100% and the specificity was 79%.
Dr. Adedinsewo is supported by the Mayo Building Interdisciplinary Research Careers in Women’s Health (BIRCWH) Program, which is funded by the National Institutes of Health. The trial was funded by Mayo Clinic (Centers for Digital Health and Community Health and Engagement Research) and in part by the Mayo Clinic BIRCWH Program. Portable ECG, phonocardiogram recordings, and AI predictions using the digital stethoscope were extracted by the Eko Health team and were sent to the coordinating center for analysis. Eko Health had no role in study design, data collection, data analysis, or data interpretation.
A version of this article appeared on Medscape.com.
in a large study of obstetric patients in Nigeria.
Demilade A. Adedinsewo, MD, MPH, from Mayo Clinic, Jacksonville, Fla., reported these findings from the Screening for Pregnancy Related Heart Failure in Nigeria (SPEC-AI Nigeria) trial in a press briefing and in a late-breaking trial session at the annual scientific sessions of the American Heart Association.
“The key takeaway,” Dr. Adedinsewo said in an interview, “is recognizing that a simple, low-impact tool like a digital stethoscope can dramatically improve the diagnosis of a life-threatening condition, and we can treat it. A large proportion of the women will recover; if we identify them early and treat them appropriately, we can reduce the risk of dying.”
If the device predicted low ejection fraction, the patient went on to have an echocardiogram to confirm cardiomyopathy, defined as a left ventricular ejection fraction (LVEF) <50%.
Peripartum cardiomyopathy was detected in 4% of the women who were screened with this tool, compared with 1.8% of those who received usual care, which included a traditional ECG.
“I believe that the control arm also has about 4% of cardiomyopathy cases, but because they didn’t have the same screening and echo, we’re missing them,” Dr. Adedinsewo said.
Diagnosis of peripartum cardiomyopathy is challenging, she noted, owing to overlap of common symptoms in pregnancy, such as lower-extremity swelling, fatigue, and shortness of breath with mild activity, which are also cardinal symptoms of heart failure.
“We were really impressed by the effectiveness of the tool, looking at how accurate it was when it comes to the sensitivity,” she added. She noted that the digital stethoscope correctly identified 92% of women with LVEF < 50% and 100% of those with LVEF < 40%.
This was the first large, clinical trial to evaluate an AI intervention in pregnancy. The investigators used a portable, battery-operated device that yielded AI results in real time.
Nigeria has the highest rate of pericardium cardiomyopathy of any country. However, one study showed a 16-fold higher rate of cardiomyopathy among African American women, compared with White women in the United States, Dr. Adedinsewo noted. “It will be important to identify who we should be screening to identify more cases,” she said.
A digital stethoscope that provides an ECG is currently available, but the algorithm that powers detection of cardiomyopathy is not yet commercially available.
Findings ‘absolutely startling’
The study discussant in the press briefing, Alexander Tarlochan Singh Sandhu, MD, from Stanford (Calif.) University congratulated the authors on this “valuable study that uses AI tools to solve a real health problem.”
Finding that 4% of the women in the intervention arm had reduced ejection fraction is “absolutely startling,” he said, “and speaks to how important improving our diagnosis in this space is.
“Where the burden of disease is high, a tool like this can be so incredibly valuable,” he said. He noted that the investigators identified 2% more patients with peripartum cardiomyopathy.
“This is an example of the potential of AI tools that can actually improve access to care and improve quality of care in resource-limited settings,” he said. “We need to move to understanding how to implement this into subsequent care [and] figure out what the next steps are to improve their outcomes.”
“The main takeaway is that, in areas where there is a very high prevalence of a morbid condition, a prescreening tool like this may be helpful” for diagnosis, the assigned discussant in the session, Marco Perez, MD, also from Stanford University, told this news organization.
The number of women needed to screen to detect peripartum cardiomyopathy by echocardiography alone is 1 in 23 in Nigeria and 1 in 970 in the United States, he said.
With an AI tool such as this one (sensitivity, 92%; specificity, 80%), the number needed to screen would be 1 in 5.7 in Nigeria and 1 in 194 in the United States, he estimates on the basis of incidence data.
“Because it is so common in Nigeria, a screening method makes a lot of sense,” Dr. Perez said. “The big question that remains is, what is the best screening modality?
“Certainly, this tool helped in bringing down the number of echoes needed to find a case, from the mid 20s down to about 5 or 6, so it certainly does seem to be helpful.”
However, the investigators did not say whether this tool is better than a clinical review of ECG or an AI analysis of ECG alone. It’s not clear whether the phonocardiogram component is significant in conjunction with the ECG component.
Nevertheless, “In a place where there’s a very high prevalence of peripartum cardiomyopathy, like Haiti, like Nigeria, doing something like this makes a lot of sense.
“For the U.S. and the rest of the world, where the prevalence is much lower, even with a tool like this you still would need to do a lot of echoes to find one case, and that may end up not being cost-effective. You would need to screen 200 women with echo to find one case.”
AI-guided screening study
Nigeria has the highest reported incidence of peripartum cardiomyopathy mortality (1 in 100 live births) and the highest number of maternal deaths.
In the United States, where rates of peripartum cardiomyopathy are much lower, maternal deaths are nevertheless higher than in other developed countries and have trended up over the past 3 decades; cardiomyopathy is a key contributor.
The investigators enrolled 1,195 women who were pregnant or had given birth in the past 12 months. The patients were from six teaching hospitals in Nigeria (two in the north and four in the south). They were randomly assigned in a 1:1 ratio to the intervention group (587) or the control group (608).
In the intervention group, clinicians used a smart stethoscope to record a phonocardiogram and a single-lead ECG reading in the V2 position and in an angled position on the patient’s chest wall and to record an ECG from the patient’s fingers. The recordings were sent to a Bluetooth-enabled mobile device (tablet or smartphone), which displayed the phonocardiogram and ECG images and that indicated whether the ejection fraction was normal or low. All patients in the intervention group received an echocardiogram.
In the control group, patients received usual care plus a traditional ECG. They were not required to have an echocardiogram because undergoing an echocardiogram is not part of usual care; however, they could receive an echocardiogram if the ECG suggested that they might need further testing.
The mean age of all the patients was 31 years, and all were Black. At study entry, 73% were pregnant, and 26% were post partum. They had similar comorbidities.
The primary outcome, cardiomyopathy (LVEF <50%) was detected in 24 of 587 patients (4.1%) in the intervention group and in 11 of 608 patients (1.8%) in the control group (odds ratio, 2.3; 95% confidence interval, 1.1-4.8; P = .02).
For the detection of LVEF <50%, the sensitivity was 92% and the specificity was 80%. For the detection of LVEF <40% (a secondary outcome), the sensitivity was 100% and the specificity was 79%.
Dr. Adedinsewo is supported by the Mayo Building Interdisciplinary Research Careers in Women’s Health (BIRCWH) Program, which is funded by the National Institutes of Health. The trial was funded by Mayo Clinic (Centers for Digital Health and Community Health and Engagement Research) and in part by the Mayo Clinic BIRCWH Program. Portable ECG, phonocardiogram recordings, and AI predictions using the digital stethoscope were extracted by the Eko Health team and were sent to the coordinating center for analysis. Eko Health had no role in study design, data collection, data analysis, or data interpretation.
A version of this article appeared on Medscape.com.
FROM AHA 2023
At-home blood pressure monitoring is cost effective over long term
Despite higher upfront costs, enhanced home blood pressure monitoring by hypertension patients could be cost-effective compared with standard clinical care over the longer term, a systematic review in JAMA Network Open found.
In an analysis of 16 studies, at-home blood pressure (HBPM) monitoring, particularly using automatic 24-hour continuous measurements alone or combined with additional support or team-based care, appeared to be economical over a minimum 10-year period compared with usual care – higher expenditures for equipment and training notwithstanding.
“Our findings suggest that clinicians, hospitals, health care systems, third-party payers, and other stakeholders should consider the long-term incremental benefits and improvements in patients’ blood pressure, quality of life, and reductions in adverse outcomes,” wrote Michelle A. Hayek, of the Population Informatics Lab, department of industrial and systems engineering, at Texas A&M University, College Station, Tex., and colleagues.
HBPM increased considerably during the COVID pandemic and is expected to increase further in the next decade, according to lead author Theodoros Giannouchos, PhD, MS, MPharm, assistant professor in the department of health policy and organization in the School of Public Health at the University of Alabama at Birmingham. “Because home blood pressure monitoring might add costs to insurers, patients, and the health care system – at least short term – we noticed a gap in the updated literature on whether this method is cost-effective relative to in-office monitoring. Hence, we conducted this review.”
Six of the 16 studies were conducted in the United States and six in the United Kingdom; 14 used a health care insurance system perspective to determine costs. In nearly half, quality-adjusted life-years gained and cost per 1–mm Hg reduction in blood pressure were used as outcomes.
Self-monitoring included self measurements transmitted to health care professionals and involved either periodic readings, such as twice each morning and evening during the first week of every month, 3 times per week, or 24-hour ambulatory readings with a portable device every 20 or 30 minutes. Among studies comparing HBPM alone versus 24-hour ambulatory BP monitoring (ABPM) or HBPM combined with additional support or team-based care, the latter two approaches were more cost effective. The benefits would appear to offset the costs of more resource-intensive at-home self-monitoring methods over office care and traditional at-home monitoring only.
In addition, the authors noted, ABPM in particular might detect elevated in-office, or white-coat hypertension, and masked hypertension, the latter referring to normal BP readings measured in the office but actual elevated pressures in the everyday home setting. An estimated 17.1 million adults in the United States have masked hypertension, and the authors say the new approach would allow early tailored interventions to mitigate the risk of masked hypertension or prevent unnecessary treatment because of white-coat hypertension. “Because of the growing market in blood pressure monitors, the technology and accuracy of monitors is expected to improve even more,” Dr. Giannouchos said. “If these technologies are properly used, they can improve patients’ quality of life and health outcomes at a justified level of cost.”
The findings align with previous research that synthesized costs and benefits of self-monitoring methods across various diseases and settings.
“Future work is needed to compare these alternatives directly from a cost-effectiveness standpoint and to provide clinicians, stakeholders, and patients with more evidence to prioritize specific home-based BP programs,” the authors wrote.
This research was supported by the Texas A&M President’s Office X-grant initiative, National Science Foundation PATHS-UP, and Population Informatics Lab. A study coauthor reported grants from National Science Foundation during the conduct of the study.
Despite higher upfront costs, enhanced home blood pressure monitoring by hypertension patients could be cost-effective compared with standard clinical care over the longer term, a systematic review in JAMA Network Open found.
In an analysis of 16 studies, at-home blood pressure (HBPM) monitoring, particularly using automatic 24-hour continuous measurements alone or combined with additional support or team-based care, appeared to be economical over a minimum 10-year period compared with usual care – higher expenditures for equipment and training notwithstanding.
“Our findings suggest that clinicians, hospitals, health care systems, third-party payers, and other stakeholders should consider the long-term incremental benefits and improvements in patients’ blood pressure, quality of life, and reductions in adverse outcomes,” wrote Michelle A. Hayek, of the Population Informatics Lab, department of industrial and systems engineering, at Texas A&M University, College Station, Tex., and colleagues.
HBPM increased considerably during the COVID pandemic and is expected to increase further in the next decade, according to lead author Theodoros Giannouchos, PhD, MS, MPharm, assistant professor in the department of health policy and organization in the School of Public Health at the University of Alabama at Birmingham. “Because home blood pressure monitoring might add costs to insurers, patients, and the health care system – at least short term – we noticed a gap in the updated literature on whether this method is cost-effective relative to in-office monitoring. Hence, we conducted this review.”
Six of the 16 studies were conducted in the United States and six in the United Kingdom; 14 used a health care insurance system perspective to determine costs. In nearly half, quality-adjusted life-years gained and cost per 1–mm Hg reduction in blood pressure were used as outcomes.
Self-monitoring included self measurements transmitted to health care professionals and involved either periodic readings, such as twice each morning and evening during the first week of every month, 3 times per week, or 24-hour ambulatory readings with a portable device every 20 or 30 minutes. Among studies comparing HBPM alone versus 24-hour ambulatory BP monitoring (ABPM) or HBPM combined with additional support or team-based care, the latter two approaches were more cost effective. The benefits would appear to offset the costs of more resource-intensive at-home self-monitoring methods over office care and traditional at-home monitoring only.
In addition, the authors noted, ABPM in particular might detect elevated in-office, or white-coat hypertension, and masked hypertension, the latter referring to normal BP readings measured in the office but actual elevated pressures in the everyday home setting. An estimated 17.1 million adults in the United States have masked hypertension, and the authors say the new approach would allow early tailored interventions to mitigate the risk of masked hypertension or prevent unnecessary treatment because of white-coat hypertension. “Because of the growing market in blood pressure monitors, the technology and accuracy of monitors is expected to improve even more,” Dr. Giannouchos said. “If these technologies are properly used, they can improve patients’ quality of life and health outcomes at a justified level of cost.”
The findings align with previous research that synthesized costs and benefits of self-monitoring methods across various diseases and settings.
“Future work is needed to compare these alternatives directly from a cost-effectiveness standpoint and to provide clinicians, stakeholders, and patients with more evidence to prioritize specific home-based BP programs,” the authors wrote.
This research was supported by the Texas A&M President’s Office X-grant initiative, National Science Foundation PATHS-UP, and Population Informatics Lab. A study coauthor reported grants from National Science Foundation during the conduct of the study.
Despite higher upfront costs, enhanced home blood pressure monitoring by hypertension patients could be cost-effective compared with standard clinical care over the longer term, a systematic review in JAMA Network Open found.
In an analysis of 16 studies, at-home blood pressure (HBPM) monitoring, particularly using automatic 24-hour continuous measurements alone or combined with additional support or team-based care, appeared to be economical over a minimum 10-year period compared with usual care – higher expenditures for equipment and training notwithstanding.
“Our findings suggest that clinicians, hospitals, health care systems, third-party payers, and other stakeholders should consider the long-term incremental benefits and improvements in patients’ blood pressure, quality of life, and reductions in adverse outcomes,” wrote Michelle A. Hayek, of the Population Informatics Lab, department of industrial and systems engineering, at Texas A&M University, College Station, Tex., and colleagues.
HBPM increased considerably during the COVID pandemic and is expected to increase further in the next decade, according to lead author Theodoros Giannouchos, PhD, MS, MPharm, assistant professor in the department of health policy and organization in the School of Public Health at the University of Alabama at Birmingham. “Because home blood pressure monitoring might add costs to insurers, patients, and the health care system – at least short term – we noticed a gap in the updated literature on whether this method is cost-effective relative to in-office monitoring. Hence, we conducted this review.”
Six of the 16 studies were conducted in the United States and six in the United Kingdom; 14 used a health care insurance system perspective to determine costs. In nearly half, quality-adjusted life-years gained and cost per 1–mm Hg reduction in blood pressure were used as outcomes.
Self-monitoring included self measurements transmitted to health care professionals and involved either periodic readings, such as twice each morning and evening during the first week of every month, 3 times per week, or 24-hour ambulatory readings with a portable device every 20 or 30 minutes. Among studies comparing HBPM alone versus 24-hour ambulatory BP monitoring (ABPM) or HBPM combined with additional support or team-based care, the latter two approaches were more cost effective. The benefits would appear to offset the costs of more resource-intensive at-home self-monitoring methods over office care and traditional at-home monitoring only.
In addition, the authors noted, ABPM in particular might detect elevated in-office, or white-coat hypertension, and masked hypertension, the latter referring to normal BP readings measured in the office but actual elevated pressures in the everyday home setting. An estimated 17.1 million adults in the United States have masked hypertension, and the authors say the new approach would allow early tailored interventions to mitigate the risk of masked hypertension or prevent unnecessary treatment because of white-coat hypertension. “Because of the growing market in blood pressure monitors, the technology and accuracy of monitors is expected to improve even more,” Dr. Giannouchos said. “If these technologies are properly used, they can improve patients’ quality of life and health outcomes at a justified level of cost.”
The findings align with previous research that synthesized costs and benefits of self-monitoring methods across various diseases and settings.
“Future work is needed to compare these alternatives directly from a cost-effectiveness standpoint and to provide clinicians, stakeholders, and patients with more evidence to prioritize specific home-based BP programs,” the authors wrote.
This research was supported by the Texas A&M President’s Office X-grant initiative, National Science Foundation PATHS-UP, and Population Informatics Lab. A study coauthor reported grants from National Science Foundation during the conduct of the study.
FROM JAMA NETWORK OPEN
FDA OKs symplicity renal denervation system for hypertension
the company has announced.
The Symplicity Spyral system, also known as the Symplicity blood pressure procedure, provides a catheter-based approach to denervate the renal arteries using radiofrequency energy.
Back in August, the FDA’s Circulatory System Devices Panel unanimously agreed that the Symplicity Spyral system is safe, but the panel was split on its efficacy and whether the benefits outweighed the risks associated with its use.
As part of the Medtronic SPYRAL HTN Global Clinical Program, more than 25,000 patients have been treated worldwide, both in the presence and absence of medication, and in patients with high baseline cardiovascular risk.
The SPYRAL HTN-OFF study enrolled patients with hypertension whose medications could be stopped at the start of the trial.
The primary effectiveness endpoint was the mean difference in the baseline adjusted 24-hour ambulatory systolic blood pressure (ASBP) from baseline to 3 months after renal denervation or sham procedure. The study showed a statistically significant reduction of 3.9 mm Hg ASBP in patients who received the device, compared with sham-control patients.
The SPYRAL HTN-ON study evaluated patients with uncontrolled hypertension who continued taking their BP medications during treatment with either the Spyral renal denervation device or a sham device.
The primary endpoint was the mean difference in the baseline adjusted 24-hour ambulatory systolic blood pressure at 6 months. The study showed a nonsignificant 24-hour 0.03 mm Hg reduction in ASBP in active-treatment patients, compared with sham-control patients.
“The Symplicity blood pressure procedure is safe and effective, providing significant ‘always-on’ blood pressure reductions for patients,” David Kandzari, MD, chief, Piedmont Heart Institute, Atlanta, and co–principal investigator of the SPYRAL clinical program, said in the news release.
“This approval paves the way for a transformation in hypertension treatment, offering a solution that complements medication and lifestyle changes,” added co–principal investigator Raymond Townsend, MD, with University of Pennsylvania, Philadelphia.
Medtronic notes that patient preference and shared decision-making are key components when considering the Symplicity blood pressure procedure.
In a patient preference study led by Medtronic, when presented with an interventional treatment with BP reduction and potential risks in line with those of the Symplicity blood pressure procedure, approximately one-third of patients were likely to choose the interventional treatment.
Earlier in November, the FDA also approved the Paradise Ultrasound Renal Denervation system (Recor Medical, Otsuka Medical Devices) for the treatment of hypertension.
A version of this article first appeared on Medscape.com.
the company has announced.
The Symplicity Spyral system, also known as the Symplicity blood pressure procedure, provides a catheter-based approach to denervate the renal arteries using radiofrequency energy.
Back in August, the FDA’s Circulatory System Devices Panel unanimously agreed that the Symplicity Spyral system is safe, but the panel was split on its efficacy and whether the benefits outweighed the risks associated with its use.
As part of the Medtronic SPYRAL HTN Global Clinical Program, more than 25,000 patients have been treated worldwide, both in the presence and absence of medication, and in patients with high baseline cardiovascular risk.
The SPYRAL HTN-OFF study enrolled patients with hypertension whose medications could be stopped at the start of the trial.
The primary effectiveness endpoint was the mean difference in the baseline adjusted 24-hour ambulatory systolic blood pressure (ASBP) from baseline to 3 months after renal denervation or sham procedure. The study showed a statistically significant reduction of 3.9 mm Hg ASBP in patients who received the device, compared with sham-control patients.
The SPYRAL HTN-ON study evaluated patients with uncontrolled hypertension who continued taking their BP medications during treatment with either the Spyral renal denervation device or a sham device.
The primary endpoint was the mean difference in the baseline adjusted 24-hour ambulatory systolic blood pressure at 6 months. The study showed a nonsignificant 24-hour 0.03 mm Hg reduction in ASBP in active-treatment patients, compared with sham-control patients.
“The Symplicity blood pressure procedure is safe and effective, providing significant ‘always-on’ blood pressure reductions for patients,” David Kandzari, MD, chief, Piedmont Heart Institute, Atlanta, and co–principal investigator of the SPYRAL clinical program, said in the news release.
“This approval paves the way for a transformation in hypertension treatment, offering a solution that complements medication and lifestyle changes,” added co–principal investigator Raymond Townsend, MD, with University of Pennsylvania, Philadelphia.
Medtronic notes that patient preference and shared decision-making are key components when considering the Symplicity blood pressure procedure.
In a patient preference study led by Medtronic, when presented with an interventional treatment with BP reduction and potential risks in line with those of the Symplicity blood pressure procedure, approximately one-third of patients were likely to choose the interventional treatment.
Earlier in November, the FDA also approved the Paradise Ultrasound Renal Denervation system (Recor Medical, Otsuka Medical Devices) for the treatment of hypertension.
A version of this article first appeared on Medscape.com.
the company has announced.
The Symplicity Spyral system, also known as the Symplicity blood pressure procedure, provides a catheter-based approach to denervate the renal arteries using radiofrequency energy.
Back in August, the FDA’s Circulatory System Devices Panel unanimously agreed that the Symplicity Spyral system is safe, but the panel was split on its efficacy and whether the benefits outweighed the risks associated with its use.
As part of the Medtronic SPYRAL HTN Global Clinical Program, more than 25,000 patients have been treated worldwide, both in the presence and absence of medication, and in patients with high baseline cardiovascular risk.
The SPYRAL HTN-OFF study enrolled patients with hypertension whose medications could be stopped at the start of the trial.
The primary effectiveness endpoint was the mean difference in the baseline adjusted 24-hour ambulatory systolic blood pressure (ASBP) from baseline to 3 months after renal denervation or sham procedure. The study showed a statistically significant reduction of 3.9 mm Hg ASBP in patients who received the device, compared with sham-control patients.
The SPYRAL HTN-ON study evaluated patients with uncontrolled hypertension who continued taking their BP medications during treatment with either the Spyral renal denervation device or a sham device.
The primary endpoint was the mean difference in the baseline adjusted 24-hour ambulatory systolic blood pressure at 6 months. The study showed a nonsignificant 24-hour 0.03 mm Hg reduction in ASBP in active-treatment patients, compared with sham-control patients.
“The Symplicity blood pressure procedure is safe and effective, providing significant ‘always-on’ blood pressure reductions for patients,” David Kandzari, MD, chief, Piedmont Heart Institute, Atlanta, and co–principal investigator of the SPYRAL clinical program, said in the news release.
“This approval paves the way for a transformation in hypertension treatment, offering a solution that complements medication and lifestyle changes,” added co–principal investigator Raymond Townsend, MD, with University of Pennsylvania, Philadelphia.
Medtronic notes that patient preference and shared decision-making are key components when considering the Symplicity blood pressure procedure.
In a patient preference study led by Medtronic, when presented with an interventional treatment with BP reduction and potential risks in line with those of the Symplicity blood pressure procedure, approximately one-third of patients were likely to choose the interventional treatment.
Earlier in November, the FDA also approved the Paradise Ultrasound Renal Denervation system (Recor Medical, Otsuka Medical Devices) for the treatment of hypertension.
A version of this article first appeared on Medscape.com.