User login
Consider potty seats when you see contact dermatitis on toddler bottoms
In such cases, be on the alert for contact dermatitis, reported Claire O. Dorfman, DO, of Lehigh Valley Health Network, Allentown, Pa., and her associates at Hershey (Pa.) Medical Center.
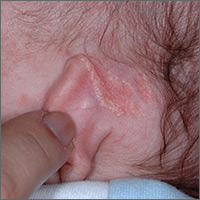
A 3-year-old white boy with a 6-month history of a pruritic rash on his buttocks and bilateral posterior thighs was treated without improvement at the pediatric dermatology clinic with low-potency topical corticosteroids, as well as topical antibiotic and antifungal agents.
Only mild improvement was seen once disposable paper toilet seat covers were added to treatment regimen. Following the purchase of a new potty seat through an online retailer, the child’s mother discovered a number of consumer product reviews also detailing similar complaints about the manufacturer, Prince Lionheart WeePOD Basix, by more than 30 other consumers. Photos highlighting identical rash presentation in other toddlers confirmed that the toilet seat was responsible for the allergic reaction. A warning had been posted by the manufacturer but this warning was not provided by the online retailer.
Use of the seat was immediately discontinued, and complete resolution of lesions was achieved within 1 month; subsequently, a report to the Consumer Product Safety Commission was made.
Allergic contact dermatitis to toilet seats is becoming increasingly common, the authors noted. Although the source of allergies is varied, wood historically has been identified as the most common material associated with the condition. Polypropylene and polyurethane foam also have been found to cause irritation. However, in the case reported by Dr. Dorfman and her associates, the precise irritant could not be identified because of the atypical pattern of the lesions and their irregular presentation on the buttocks and thighs. They speculated that this irregularity could be attributed to “the small, round shape of the seat and the squirmy behavior of a toddler,” because the typical arciform distribution was not present. Relief was not achieved with the paper liners because they did not completely cover the seat.
Because the rash resolved when the seat was replaced, parents declined patch testing. As a result, it was not possible to identify the specific allergenic component of the polyurethane. The polyurethanes used to make the seats are synthetic polymers that contain isocyanates, and frequently diaminodiphenylmethane, a curing agent. Possible allergy to the dyes used during manufacture also was considered but the presenting rash was reported in all four of the available colors made.
Although it was speculated that exposure to cleansers could be to blame for possible irritant dermatitis given reports of cracking of the potty seat, the mother and several online reviews indicated only soap and water were used, not harsh cleaning agents.
The clinicians had no relevant financial disclosures.
SOURCE: Dorfman CO et al. Pediatr Dermatol. 2018 May 29. doi: 10.1111/pde.13534.
In such cases, be on the alert for contact dermatitis, reported Claire O. Dorfman, DO, of Lehigh Valley Health Network, Allentown, Pa., and her associates at Hershey (Pa.) Medical Center.
A 3-year-old white boy with a 6-month history of a pruritic rash on his buttocks and bilateral posterior thighs was treated without improvement at the pediatric dermatology clinic with low-potency topical corticosteroids, as well as topical antibiotic and antifungal agents.
Only mild improvement was seen once disposable paper toilet seat covers were added to treatment regimen. Following the purchase of a new potty seat through an online retailer, the child’s mother discovered a number of consumer product reviews also detailing similar complaints about the manufacturer, Prince Lionheart WeePOD Basix, by more than 30 other consumers. Photos highlighting identical rash presentation in other toddlers confirmed that the toilet seat was responsible for the allergic reaction. A warning had been posted by the manufacturer but this warning was not provided by the online retailer.
Use of the seat was immediately discontinued, and complete resolution of lesions was achieved within 1 month; subsequently, a report to the Consumer Product Safety Commission was made.
Allergic contact dermatitis to toilet seats is becoming increasingly common, the authors noted. Although the source of allergies is varied, wood historically has been identified as the most common material associated with the condition. Polypropylene and polyurethane foam also have been found to cause irritation. However, in the case reported by Dr. Dorfman and her associates, the precise irritant could not be identified because of the atypical pattern of the lesions and their irregular presentation on the buttocks and thighs. They speculated that this irregularity could be attributed to “the small, round shape of the seat and the squirmy behavior of a toddler,” because the typical arciform distribution was not present. Relief was not achieved with the paper liners because they did not completely cover the seat.
Because the rash resolved when the seat was replaced, parents declined patch testing. As a result, it was not possible to identify the specific allergenic component of the polyurethane. The polyurethanes used to make the seats are synthetic polymers that contain isocyanates, and frequently diaminodiphenylmethane, a curing agent. Possible allergy to the dyes used during manufacture also was considered but the presenting rash was reported in all four of the available colors made.
Although it was speculated that exposure to cleansers could be to blame for possible irritant dermatitis given reports of cracking of the potty seat, the mother and several online reviews indicated only soap and water were used, not harsh cleaning agents.
The clinicians had no relevant financial disclosures.
SOURCE: Dorfman CO et al. Pediatr Dermatol. 2018 May 29. doi: 10.1111/pde.13534.
In such cases, be on the alert for contact dermatitis, reported Claire O. Dorfman, DO, of Lehigh Valley Health Network, Allentown, Pa., and her associates at Hershey (Pa.) Medical Center.
A 3-year-old white boy with a 6-month history of a pruritic rash on his buttocks and bilateral posterior thighs was treated without improvement at the pediatric dermatology clinic with low-potency topical corticosteroids, as well as topical antibiotic and antifungal agents.
Only mild improvement was seen once disposable paper toilet seat covers were added to treatment regimen. Following the purchase of a new potty seat through an online retailer, the child’s mother discovered a number of consumer product reviews also detailing similar complaints about the manufacturer, Prince Lionheart WeePOD Basix, by more than 30 other consumers. Photos highlighting identical rash presentation in other toddlers confirmed that the toilet seat was responsible for the allergic reaction. A warning had been posted by the manufacturer but this warning was not provided by the online retailer.
Use of the seat was immediately discontinued, and complete resolution of lesions was achieved within 1 month; subsequently, a report to the Consumer Product Safety Commission was made.
Allergic contact dermatitis to toilet seats is becoming increasingly common, the authors noted. Although the source of allergies is varied, wood historically has been identified as the most common material associated with the condition. Polypropylene and polyurethane foam also have been found to cause irritation. However, in the case reported by Dr. Dorfman and her associates, the precise irritant could not be identified because of the atypical pattern of the lesions and their irregular presentation on the buttocks and thighs. They speculated that this irregularity could be attributed to “the small, round shape of the seat and the squirmy behavior of a toddler,” because the typical arciform distribution was not present. Relief was not achieved with the paper liners because they did not completely cover the seat.
Because the rash resolved when the seat was replaced, parents declined patch testing. As a result, it was not possible to identify the specific allergenic component of the polyurethane. The polyurethanes used to make the seats are synthetic polymers that contain isocyanates, and frequently diaminodiphenylmethane, a curing agent. Possible allergy to the dyes used during manufacture also was considered but the presenting rash was reported in all four of the available colors made.
Although it was speculated that exposure to cleansers could be to blame for possible irritant dermatitis given reports of cracking of the potty seat, the mother and several online reviews indicated only soap and water were used, not harsh cleaning agents.
The clinicians had no relevant financial disclosures.
SOURCE: Dorfman CO et al. Pediatr Dermatol. 2018 May 29. doi: 10.1111/pde.13534.
FROM THE JOURNAL OF PEDIATRIC DERMATOLOGY
Key clinical point: Suspect contact dermatitis in cases of unexplained pruritic rash.
Major finding: Allergic contact dermatitis to toilet seats is becoming increasingly common.
Study details: A case study.
Disclosures: The authors had no relevant financial disclosures.
Source: Dorfman CO et al. Pediatr Dermatol. 2018 May 29. doi: 10.1111/pde.13534.
Ozenoxacin is a novel treatment option for impetigo
Ozenoxacin (Xepi cream), a potent novel topical antibacterial agent, is effective and well tolerated for treatment of impetigo in patients aged 2 months and older, according to results of a phase 3 randomized, double-blind, vehicle-controlled clinical trial.
A 1% cream formulation of ozenoxacin had a clinical and microbiologic response that was rapid and superior to placebo, investigators reported in this second phase 3 pivotal study, which demonstrated the efficacy of ozenoxacin cream in impetigo patients.
“With concerns over widespread antibiotic resistance, ozenoxacin is an important potential treatment option with an expanded spectrum against bacterial pathogens, including those resistant to mupirocin, ciprofloxacin, and methicillin, including MRSA [methicillin-resistant Staphylococcus aureus],” Dr. Rosen and his colleagues wrote in a report on their study in JAMA Dermatology.
The clinical trial included 412 patients aged 2 months or older with impetigo. Clinical success, defined as complete absence of the treated lesions, was seen after 5 days of treatment in 112 of the 206 (54.4%) patients randomized to ozenoxacin versus 78 of 206 patients (37.9%) who received placebo (P = .001), which confirmed the superiority of ozenoxacin, Dr. Rosen and his coauthors wrote.
In addition, microbiologic response was seen in 87.2% of patients in the ozenoxacin group during therapy (day 3) versus 63.9% in the placebo group (P = .002); by the end of therapy (day 6), response rates were 92.0% and 73.1%, respectively (P = .005).
All patients with drug-resistant infections had clinical cure or improvement at the end of treatment, including 10 patients with mupirocin-resistant S. aureus and 8 patients with MRSA, investigators wrote.
Adverse event rates were low for both ozenoxacin and placebo groups, at 3.9% and 3.4%, respectively. One patient in the ozenoxacin group experienced a serious adverse event “at least potentially related” to treatment (rosacea and seborrheic dermatitis).
“The lack of systemic adverse effects is consistent with previous studies that demonstrated that topically applied ozenoxacin has negligible systemic absorption,” Dr. Rosen and his coauthors wrote.
The study was partly supported by Ferrer Internacional; two coauthors of the study reported employment with that company. Dr. Rosen reported consulting work with Medimetriks Pharmaceuticals, which developed the agent.
SOURCE: Rosen T et al. JAMA Dermatol. 2018 Jun 13. doi: 10.1001/jamadermatol.2018.1103.
Ozenoxacin (Xepi cream), a potent novel topical antibacterial agent, is effective and well tolerated for treatment of impetigo in patients aged 2 months and older, according to results of a phase 3 randomized, double-blind, vehicle-controlled clinical trial.
A 1% cream formulation of ozenoxacin had a clinical and microbiologic response that was rapid and superior to placebo, investigators reported in this second phase 3 pivotal study, which demonstrated the efficacy of ozenoxacin cream in impetigo patients.
“With concerns over widespread antibiotic resistance, ozenoxacin is an important potential treatment option with an expanded spectrum against bacterial pathogens, including those resistant to mupirocin, ciprofloxacin, and methicillin, including MRSA [methicillin-resistant Staphylococcus aureus],” Dr. Rosen and his colleagues wrote in a report on their study in JAMA Dermatology.
The clinical trial included 412 patients aged 2 months or older with impetigo. Clinical success, defined as complete absence of the treated lesions, was seen after 5 days of treatment in 112 of the 206 (54.4%) patients randomized to ozenoxacin versus 78 of 206 patients (37.9%) who received placebo (P = .001), which confirmed the superiority of ozenoxacin, Dr. Rosen and his coauthors wrote.
In addition, microbiologic response was seen in 87.2% of patients in the ozenoxacin group during therapy (day 3) versus 63.9% in the placebo group (P = .002); by the end of therapy (day 6), response rates were 92.0% and 73.1%, respectively (P = .005).
All patients with drug-resistant infections had clinical cure or improvement at the end of treatment, including 10 patients with mupirocin-resistant S. aureus and 8 patients with MRSA, investigators wrote.
Adverse event rates were low for both ozenoxacin and placebo groups, at 3.9% and 3.4%, respectively. One patient in the ozenoxacin group experienced a serious adverse event “at least potentially related” to treatment (rosacea and seborrheic dermatitis).
“The lack of systemic adverse effects is consistent with previous studies that demonstrated that topically applied ozenoxacin has negligible systemic absorption,” Dr. Rosen and his coauthors wrote.
The study was partly supported by Ferrer Internacional; two coauthors of the study reported employment with that company. Dr. Rosen reported consulting work with Medimetriks Pharmaceuticals, which developed the agent.
SOURCE: Rosen T et al. JAMA Dermatol. 2018 Jun 13. doi: 10.1001/jamadermatol.2018.1103.
Ozenoxacin (Xepi cream), a potent novel topical antibacterial agent, is effective and well tolerated for treatment of impetigo in patients aged 2 months and older, according to results of a phase 3 randomized, double-blind, vehicle-controlled clinical trial.
A 1% cream formulation of ozenoxacin had a clinical and microbiologic response that was rapid and superior to placebo, investigators reported in this second phase 3 pivotal study, which demonstrated the efficacy of ozenoxacin cream in impetigo patients.
“With concerns over widespread antibiotic resistance, ozenoxacin is an important potential treatment option with an expanded spectrum against bacterial pathogens, including those resistant to mupirocin, ciprofloxacin, and methicillin, including MRSA [methicillin-resistant Staphylococcus aureus],” Dr. Rosen and his colleagues wrote in a report on their study in JAMA Dermatology.
The clinical trial included 412 patients aged 2 months or older with impetigo. Clinical success, defined as complete absence of the treated lesions, was seen after 5 days of treatment in 112 of the 206 (54.4%) patients randomized to ozenoxacin versus 78 of 206 patients (37.9%) who received placebo (P = .001), which confirmed the superiority of ozenoxacin, Dr. Rosen and his coauthors wrote.
In addition, microbiologic response was seen in 87.2% of patients in the ozenoxacin group during therapy (day 3) versus 63.9% in the placebo group (P = .002); by the end of therapy (day 6), response rates were 92.0% and 73.1%, respectively (P = .005).
All patients with drug-resistant infections had clinical cure or improvement at the end of treatment, including 10 patients with mupirocin-resistant S. aureus and 8 patients with MRSA, investigators wrote.
Adverse event rates were low for both ozenoxacin and placebo groups, at 3.9% and 3.4%, respectively. One patient in the ozenoxacin group experienced a serious adverse event “at least potentially related” to treatment (rosacea and seborrheic dermatitis).
“The lack of systemic adverse effects is consistent with previous studies that demonstrated that topically applied ozenoxacin has negligible systemic absorption,” Dr. Rosen and his coauthors wrote.
The study was partly supported by Ferrer Internacional; two coauthors of the study reported employment with that company. Dr. Rosen reported consulting work with Medimetriks Pharmaceuticals, which developed the agent.
SOURCE: Rosen T et al. JAMA Dermatol. 2018 Jun 13. doi: 10.1001/jamadermatol.2018.1103.
FROM JAMA DERMATOLOGY
Key clinical point: Ozenoxacin, a novel topical antibacterial agent, is effective and well tolerated for treatment of impetigo
Major finding: Clinical response was seen in 54.4% of ozenoxacin-treated patients and 37.9% of placebo-treated patients (P = .001).
Study details: A phase 3 randomized, double-blind, vehicle-controlled clinical trial that included 411 patients aged 2 months or older with impetigo.
Disclosures: Partial support came from Ferrer Internacional; two coauthors worked for that company. The study’s first author reported consulting work with Medimetriks, the developer of the agent.
Source: Rosen T et al. JAMA Dermatol. 2018 Jun 13. doi: 10.1001/jamadermatol.2018.1103.
JAK inhibitor therapy promising for refractory dermatomyositis
ORLANDO – The according to Ruth Ann Vleugels, MD, director of the autoimmune skin diseases program at Brigham and Women’s Hospital, Boston.
“We will have patients who essentially fail all of our typical therapies and are still coming to us for help. This is a huge challenge,” said Dr. Vleugels, who, several years ago, started to use tofacitinib to treat these patients. “Similar to my colleagues who use tofacitinib to treat alopecia areata, we often have to push” beyond the dose used to treat rheumatoid arthritis, to 10 mg twice a day, she said at the International Conference on Cutaneous Lupus Erythematosus. Tofacitinib helps counter the overexpression of interferon in DM.
Getting insurance coverage for this off-label indication can be tough, however, but Dr. Vleugels said she’s had success when she tells insurers that tofacitinib will likely reduce the need for IVIg.
It’s safe to keep patients on methotrexate if there are concerns about muscle involvement while their skin is brought under control with tofacitinib. In terms of side effects, “we see increased shingles,” so recommending the shingles vaccine for these patients is a good idea, she added.
It’s also important to counsel DM patients that they are at particular risk for skin reactions with antimalarials, which can be serious, so that, “if there is a drug reaction that develops, it’s noticed right away” and the drug can be stopped, she said. “If you have a patient who has very severe disease, I might skip over an antimalarial altogether,” she commented.
Methotrexate is the next option, especially if there are work ups for cancer or the patients have cancer, but Dr. Femia, director of inpatient dermatology at NYU, said she leans towards mycophenolate if there’s concern about lung involvement.
The next step, if necessary, is IVIg, which she said is “particularly helpful” for recalcitrant skin disease and can help some patients discontinue other immunosuppressives. To counter headache, a common side effect, she will space dosing out over 3 days, instead of the usual 2, and have a bag of saline administered before and after the infusion to keep patients hydrated; this counters the headache-inducing viscosity of IVIg.
Patients often see a result after the first infusion, but if there’s no benefit by the third cycle, “it’s probably time to move on,” she said. “If you have a refractory muscle disease patient and skin isn’t the main issue, rituximab is reasonable to try,” she added, noting that the benefit of tumor necrosis factor blockers, “at best, is very mixed in the DM population. They are very low down in the treatment algorithm.”
Dr. Vleugels and Dr. Femia are both Pfizer investigators.
ORLANDO – The according to Ruth Ann Vleugels, MD, director of the autoimmune skin diseases program at Brigham and Women’s Hospital, Boston.
“We will have patients who essentially fail all of our typical therapies and are still coming to us for help. This is a huge challenge,” said Dr. Vleugels, who, several years ago, started to use tofacitinib to treat these patients. “Similar to my colleagues who use tofacitinib to treat alopecia areata, we often have to push” beyond the dose used to treat rheumatoid arthritis, to 10 mg twice a day, she said at the International Conference on Cutaneous Lupus Erythematosus. Tofacitinib helps counter the overexpression of interferon in DM.
Getting insurance coverage for this off-label indication can be tough, however, but Dr. Vleugels said she’s had success when she tells insurers that tofacitinib will likely reduce the need for IVIg.
It’s safe to keep patients on methotrexate if there are concerns about muscle involvement while their skin is brought under control with tofacitinib. In terms of side effects, “we see increased shingles,” so recommending the shingles vaccine for these patients is a good idea, she added.
It’s also important to counsel DM patients that they are at particular risk for skin reactions with antimalarials, which can be serious, so that, “if there is a drug reaction that develops, it’s noticed right away” and the drug can be stopped, she said. “If you have a patient who has very severe disease, I might skip over an antimalarial altogether,” she commented.
Methotrexate is the next option, especially if there are work ups for cancer or the patients have cancer, but Dr. Femia, director of inpatient dermatology at NYU, said she leans towards mycophenolate if there’s concern about lung involvement.
The next step, if necessary, is IVIg, which she said is “particularly helpful” for recalcitrant skin disease and can help some patients discontinue other immunosuppressives. To counter headache, a common side effect, she will space dosing out over 3 days, instead of the usual 2, and have a bag of saline administered before and after the infusion to keep patients hydrated; this counters the headache-inducing viscosity of IVIg.
Patients often see a result after the first infusion, but if there’s no benefit by the third cycle, “it’s probably time to move on,” she said. “If you have a refractory muscle disease patient and skin isn’t the main issue, rituximab is reasonable to try,” she added, noting that the benefit of tumor necrosis factor blockers, “at best, is very mixed in the DM population. They are very low down in the treatment algorithm.”
Dr. Vleugels and Dr. Femia are both Pfizer investigators.
ORLANDO – The according to Ruth Ann Vleugels, MD, director of the autoimmune skin diseases program at Brigham and Women’s Hospital, Boston.
“We will have patients who essentially fail all of our typical therapies and are still coming to us for help. This is a huge challenge,” said Dr. Vleugels, who, several years ago, started to use tofacitinib to treat these patients. “Similar to my colleagues who use tofacitinib to treat alopecia areata, we often have to push” beyond the dose used to treat rheumatoid arthritis, to 10 mg twice a day, she said at the International Conference on Cutaneous Lupus Erythematosus. Tofacitinib helps counter the overexpression of interferon in DM.
Getting insurance coverage for this off-label indication can be tough, however, but Dr. Vleugels said she’s had success when she tells insurers that tofacitinib will likely reduce the need for IVIg.
It’s safe to keep patients on methotrexate if there are concerns about muscle involvement while their skin is brought under control with tofacitinib. In terms of side effects, “we see increased shingles,” so recommending the shingles vaccine for these patients is a good idea, she added.
It’s also important to counsel DM patients that they are at particular risk for skin reactions with antimalarials, which can be serious, so that, “if there is a drug reaction that develops, it’s noticed right away” and the drug can be stopped, she said. “If you have a patient who has very severe disease, I might skip over an antimalarial altogether,” she commented.
Methotrexate is the next option, especially if there are work ups for cancer or the patients have cancer, but Dr. Femia, director of inpatient dermatology at NYU, said she leans towards mycophenolate if there’s concern about lung involvement.
The next step, if necessary, is IVIg, which she said is “particularly helpful” for recalcitrant skin disease and can help some patients discontinue other immunosuppressives. To counter headache, a common side effect, she will space dosing out over 3 days, instead of the usual 2, and have a bag of saline administered before and after the infusion to keep patients hydrated; this counters the headache-inducing viscosity of IVIg.
Patients often see a result after the first infusion, but if there’s no benefit by the third cycle, “it’s probably time to move on,” she said. “If you have a refractory muscle disease patient and skin isn’t the main issue, rituximab is reasonable to try,” she added, noting that the benefit of tumor necrosis factor blockers, “at best, is very mixed in the DM population. They are very low down in the treatment algorithm.”
Dr. Vleugels and Dr. Femia are both Pfizer investigators.
EXPERT ANALYSIS FROM ICCLE 2018
Pediatric Dermatology Consult - July 2018
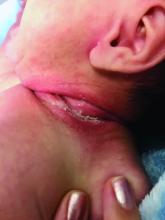
Frequently misdiagnosed, streptococcal intertrigo more commonly affects infants and toddlers but is rarely reported, especially compared with other Streptococcus pyogenes infections, including impetigo, erysipelas, and cellulitis.1
Intertrigo, meaning “between” (inter) and “to rub” (terere) in Latin, describes any skin disorder involving two opposing skin surfaces that touch or rub to cause friction.2 The continuous chaffing, coupled with moisture trapped within the skin folds, leads to irritation and maceration, which provides an ideal environment for pathogens to thrive. Thus, frictional dermatitides that arise may become secondarily infected with one or more microorganisms, such as Candida albicans, Staphylococcus aureus, Streptococcus pyogenes, and even organisms less commonly associated with cutaneous infection, such as Proteus mirabilis.3
Streptococcal intertrigo may affect any intertriginous area, but most commonly it affects the folds of the neck; this is likely because of the combination of the deep folds that develop in shorter, infantile necks and the moisture from drool and saliva that pools in the area.5,6 In addition to these cervical folds, other intertriginous areas commonly are affected, including the inguinal, axillary, popliteal, posterior auricular, perianal, and genital folds.
Perianal streptococcal disease may present in a similar manner as streptococcal intertrigo, manifesting as well-demarcated, beefy red plaques in the skin folds around the anus and, in females, frequently perivaginally.7 Unlike streptococcal intertrigo, perianal streptococcal disease is often characterized by pain, pruritus, and fissuring of the involved area.8 It is associated with pharyngeal colonization of group A beta-hemolytic streptococci.7
Diagnosis is straight forward and may be confirmed by a positive streptococcal rapid antigen test of swab specimens of one or more surfaces of affected skin or by culture from a skin swab yielding growth of the organism.1,5 Skin biopsy is not necessary. If the index of suspicion for candida is high, a potassium hydroxide preparation and culture may be performed. Checking serum anti-DNase B antibodies, antistreptolysin O, and pharyngeal cultures is often unrevealing.9 A urinalysis may be performed to assess for poststreptococcal glomerulonephritis if the patient later develops facial or orbital edema, hypertension, hematuria, or lethargy.9
Treatment consists of systemic antistreptococcal therapy; oral amoxicillin and penicillin frequently have been used.9 Moisture in the area should be reduced with application of absorptive powders and physical barriers, such as zinc oxide, after gentle cleansing of the area.5
Other diagnoses to consider when evaluating dermatitides affecting skin folds include: other infectious causes, which may be ruled out by fungal or bacterial culture; inverse psoriasis, which will frequently demonstrate scale; atopic dermatitis, which will be pruritic with history of atopy; irritant or contact dermatitis, which will often have correlating clinical history; seborrheic dermatitis, which will often involve greasiness and scale; and less commonly, acrodermatitis enteropathica, which will be accompanied by diarrhea and hair loss.2,9 Scabies also may be on the differential if the patient endorses severe pruritus with close contacts with similar symptoms.
Ms. Han is a medical student at the University of California, San Diego. Dr. Eichenfield is chief of pediatric and adolescent dermatology at Rady Children’s Hospital–San Diego. He is vice chair of the department of dermatology and a professor of dermatology and pediatrics at the university. They had no conflicts of interest or disclosures to report.
References
1. Pediatr Dermatol. 2014 Mar-Apr;31(2):e71-2.
2. Clin Dermatol. 2011 Mar-Apr;29(2):173-9.
3. Pediatrics. 2003 Dec;112(6 pt 1):1427-9.
4. BMJ Case Rep. 2018 Mar 20. doi: 10.1136/bcr-2018-224179.
5. Pediatr Infect Dis J. 2012 Aug;31(8):872-3.
6. J Pediatr. 2015 May;166(5):1318.
7. J Pediatr. 2015 Sep;167(3):687-93.e1-2.
8. Pediatrics in Review. 1991;12(8):248-55.
9. J Pediatr. 2017 May;184:230-1.e1.
Frequently misdiagnosed, streptococcal intertrigo more commonly affects infants and toddlers but is rarely reported, especially compared with other Streptococcus pyogenes infections, including impetigo, erysipelas, and cellulitis.1
Intertrigo, meaning “between” (inter) and “to rub” (terere) in Latin, describes any skin disorder involving two opposing skin surfaces that touch or rub to cause friction.2 The continuous chaffing, coupled with moisture trapped within the skin folds, leads to irritation and maceration, which provides an ideal environment for pathogens to thrive. Thus, frictional dermatitides that arise may become secondarily infected with one or more microorganisms, such as Candida albicans, Staphylococcus aureus, Streptococcus pyogenes, and even organisms less commonly associated with cutaneous infection, such as Proteus mirabilis.3
Streptococcal intertrigo may affect any intertriginous area, but most commonly it affects the folds of the neck; this is likely because of the combination of the deep folds that develop in shorter, infantile necks and the moisture from drool and saliva that pools in the area.5,6 In addition to these cervical folds, other intertriginous areas commonly are affected, including the inguinal, axillary, popliteal, posterior auricular, perianal, and genital folds.
Perianal streptococcal disease may present in a similar manner as streptococcal intertrigo, manifesting as well-demarcated, beefy red plaques in the skin folds around the anus and, in females, frequently perivaginally.7 Unlike streptococcal intertrigo, perianal streptococcal disease is often characterized by pain, pruritus, and fissuring of the involved area.8 It is associated with pharyngeal colonization of group A beta-hemolytic streptococci.7
Diagnosis is straight forward and may be confirmed by a positive streptococcal rapid antigen test of swab specimens of one or more surfaces of affected skin or by culture from a skin swab yielding growth of the organism.1,5 Skin biopsy is not necessary. If the index of suspicion for candida is high, a potassium hydroxide preparation and culture may be performed. Checking serum anti-DNase B antibodies, antistreptolysin O, and pharyngeal cultures is often unrevealing.9 A urinalysis may be performed to assess for poststreptococcal glomerulonephritis if the patient later develops facial or orbital edema, hypertension, hematuria, or lethargy.9
Treatment consists of systemic antistreptococcal therapy; oral amoxicillin and penicillin frequently have been used.9 Moisture in the area should be reduced with application of absorptive powders and physical barriers, such as zinc oxide, after gentle cleansing of the area.5
Other diagnoses to consider when evaluating dermatitides affecting skin folds include: other infectious causes, which may be ruled out by fungal or bacterial culture; inverse psoriasis, which will frequently demonstrate scale; atopic dermatitis, which will be pruritic with history of atopy; irritant or contact dermatitis, which will often have correlating clinical history; seborrheic dermatitis, which will often involve greasiness and scale; and less commonly, acrodermatitis enteropathica, which will be accompanied by diarrhea and hair loss.2,9 Scabies also may be on the differential if the patient endorses severe pruritus with close contacts with similar symptoms.
Ms. Han is a medical student at the University of California, San Diego. Dr. Eichenfield is chief of pediatric and adolescent dermatology at Rady Children’s Hospital–San Diego. He is vice chair of the department of dermatology and a professor of dermatology and pediatrics at the university. They had no conflicts of interest or disclosures to report.
References
1. Pediatr Dermatol. 2014 Mar-Apr;31(2):e71-2.
2. Clin Dermatol. 2011 Mar-Apr;29(2):173-9.
3. Pediatrics. 2003 Dec;112(6 pt 1):1427-9.
4. BMJ Case Rep. 2018 Mar 20. doi: 10.1136/bcr-2018-224179.
5. Pediatr Infect Dis J. 2012 Aug;31(8):872-3.
6. J Pediatr. 2015 May;166(5):1318.
7. J Pediatr. 2015 Sep;167(3):687-93.e1-2.
8. Pediatrics in Review. 1991;12(8):248-55.
9. J Pediatr. 2017 May;184:230-1.e1.
Frequently misdiagnosed, streptococcal intertrigo more commonly affects infants and toddlers but is rarely reported, especially compared with other Streptococcus pyogenes infections, including impetigo, erysipelas, and cellulitis.1
Intertrigo, meaning “between” (inter) and “to rub” (terere) in Latin, describes any skin disorder involving two opposing skin surfaces that touch or rub to cause friction.2 The continuous chaffing, coupled with moisture trapped within the skin folds, leads to irritation and maceration, which provides an ideal environment for pathogens to thrive. Thus, frictional dermatitides that arise may become secondarily infected with one or more microorganisms, such as Candida albicans, Staphylococcus aureus, Streptococcus pyogenes, and even organisms less commonly associated with cutaneous infection, such as Proteus mirabilis.3
Streptococcal intertrigo may affect any intertriginous area, but most commonly it affects the folds of the neck; this is likely because of the combination of the deep folds that develop in shorter, infantile necks and the moisture from drool and saliva that pools in the area.5,6 In addition to these cervical folds, other intertriginous areas commonly are affected, including the inguinal, axillary, popliteal, posterior auricular, perianal, and genital folds.
Perianal streptococcal disease may present in a similar manner as streptococcal intertrigo, manifesting as well-demarcated, beefy red plaques in the skin folds around the anus and, in females, frequently perivaginally.7 Unlike streptococcal intertrigo, perianal streptococcal disease is often characterized by pain, pruritus, and fissuring of the involved area.8 It is associated with pharyngeal colonization of group A beta-hemolytic streptococci.7
Diagnosis is straight forward and may be confirmed by a positive streptococcal rapid antigen test of swab specimens of one or more surfaces of affected skin or by culture from a skin swab yielding growth of the organism.1,5 Skin biopsy is not necessary. If the index of suspicion for candida is high, a potassium hydroxide preparation and culture may be performed. Checking serum anti-DNase B antibodies, antistreptolysin O, and pharyngeal cultures is often unrevealing.9 A urinalysis may be performed to assess for poststreptococcal glomerulonephritis if the patient later develops facial or orbital edema, hypertension, hematuria, or lethargy.9
Treatment consists of systemic antistreptococcal therapy; oral amoxicillin and penicillin frequently have been used.9 Moisture in the area should be reduced with application of absorptive powders and physical barriers, such as zinc oxide, after gentle cleansing of the area.5
Other diagnoses to consider when evaluating dermatitides affecting skin folds include: other infectious causes, which may be ruled out by fungal or bacterial culture; inverse psoriasis, which will frequently demonstrate scale; atopic dermatitis, which will be pruritic with history of atopy; irritant or contact dermatitis, which will often have correlating clinical history; seborrheic dermatitis, which will often involve greasiness and scale; and less commonly, acrodermatitis enteropathica, which will be accompanied by diarrhea and hair loss.2,9 Scabies also may be on the differential if the patient endorses severe pruritus with close contacts with similar symptoms.
Ms. Han is a medical student at the University of California, San Diego. Dr. Eichenfield is chief of pediatric and adolescent dermatology at Rady Children’s Hospital–San Diego. He is vice chair of the department of dermatology and a professor of dermatology and pediatrics at the university. They had no conflicts of interest or disclosures to report.
References
1. Pediatr Dermatol. 2014 Mar-Apr;31(2):e71-2.
2. Clin Dermatol. 2011 Mar-Apr;29(2):173-9.
3. Pediatrics. 2003 Dec;112(6 pt 1):1427-9.
4. BMJ Case Rep. 2018 Mar 20. doi: 10.1136/bcr-2018-224179.
5. Pediatr Infect Dis J. 2012 Aug;31(8):872-3.
6. J Pediatr. 2015 May;166(5):1318.
7. J Pediatr. 2015 Sep;167(3):687-93.e1-2.
8. Pediatrics in Review. 1991;12(8):248-55.
9. J Pediatr. 2017 May;184:230-1.e1.
An 8-week-old male with a history of cradle cap presented for a second evaluation of an erythematous rash on the neck that started 1.5 weeks before, and it had since worsened. The parents note that their infant has been more irritable, but they otherwise deny any fever, diarrhea, constipation, or decrease in oral intake.
The patient’s first evaluation had been 3 days prior; nystatin cream was prescribed, and the parents applied it twice a day but without improvement to the rash. The patient also had a rash behind the ears bilaterally, which was treated with hydrocortisone 2.5% ointment with some improvement
On physical exam, the central neck is covered by a bright, beefy red, erythematous plaque with distinct borders and strong odor. There is faint scale and superficial desquamation between the skin folds. There are no surrounding papules or pustules. The patient’s chin is moist with drool. In the postauricular skin folds bilaterally, there are fainter but still erythematous plaques with mild scale.
Mother Knows Best
About six months ago, an 8-year-old girl developed an asymptomatic rash near her ear. Her mother suspects it is psoriasis, which runs heavily in the family—but their primary care provider favors a fungal diagnosis. He prescribes a succession of topical and oral antifungal medications (including nystatin and terbinafine), which yield no discernable improvement. At this point, referral to dermatology is made.
The child’s mother denies any history of recent infections (eg, strep throat) on her daughter’s behalf. Furthermore, there are no reports of pain associated with the rash or elsewhere.
EXAMINATION
The rash, which is confined to the external right ear, is composed of uniformly smooth white scale on a faintly salmon base. The entire lesion measures about 3 cm at its widest point, and the margins are arciform and well-defined.

No such lesions are seen elsewhere, but tiny pits can be seen on one fingernail.
What is the diagnosis?
DISCUSSION
A punch biopsy could have confirmed the diagnosis, but with the family history, classic appearance, and lack of response to antifungal medication, there was little doubt that this was a case of psoriasis. This autoimmune disease affects nearly 3% of the white population in this country and has a genetic component about 30% of the time.
In psoriasis, keratinocytes matriculate upward from the basal layer to the skin surface at four times the normal rate—so quickly that they have no chance to lose their nuclei (as they normally would). They then pile up, creating plaques of micaceous white scale on a salmon-pink base. Histologically, the smoothly undulating dermoepidermal junction is jammed together, producing fused ridges with clumps of neutrophils on their tips.
While it favors extensor surfaces of extremities, psoriasis can show up anywhere on the body—on the genitals, mouth, and in the nails, where it can cause pits, dystrophy, discoloration, onycholysis, and onychorrhexis.
Unfortunately, this is probably just the beginning of this child’s psoriasis. The good news is that we’re in a golden age of psoriasis treatment, with more drugs than ever to choose from and even more in development. For this patient, we used a keratolytic agent (urea lotion) to thin out the surface scale, in order to allow a class 4 steroid cream to reach the pink inflammatory portion. Within a month, most of this patch had cleared, though we can be fairly sure it and others like it will be back. Education and ongoing follow-up will be needed, in case she is among the 20% to 25% of patients who will develop psoriatic arthropathy, a crippling form of arthritis.
It is certainly possible to develop a fungal infection on or in an ear, but for that to happen, there has to be a source (eg, animal, person, soil). Moreover, the scale would look entirely different, with clearing centers and advancing margins. The likely truth is that this was called “fungal” for lack of any other suspects.
TAKE-HOME LEARNING POINTS
- White scale on a salmon-pink base typifies psoriasis vulgaris, a very common diagnosis that is often mistaken for fungal infection; biopsy can be extremely helpful in establishing or ruling out this diagnosis.
- Psoriasis has a genetic basis, with many gene loci identified to date, but only about 30% of affected patients can attest to a family history.
- In addition to having unsightly, often itchy lesions, psoriasis patients are also at risk for psoriatic arthropathy, a potentially crippling condition.
- The best news is that we have many drugs with which to treat this disease, including a whole family of drugs termed “the biologics,” which directly (and successfully!) address the disease.
About six months ago, an 8-year-old girl developed an asymptomatic rash near her ear. Her mother suspects it is psoriasis, which runs heavily in the family—but their primary care provider favors a fungal diagnosis. He prescribes a succession of topical and oral antifungal medications (including nystatin and terbinafine), which yield no discernable improvement. At this point, referral to dermatology is made.
The child’s mother denies any history of recent infections (eg, strep throat) on her daughter’s behalf. Furthermore, there are no reports of pain associated with the rash or elsewhere.
EXAMINATION
The rash, which is confined to the external right ear, is composed of uniformly smooth white scale on a faintly salmon base. The entire lesion measures about 3 cm at its widest point, and the margins are arciform and well-defined.

No such lesions are seen elsewhere, but tiny pits can be seen on one fingernail.
What is the diagnosis?
DISCUSSION
A punch biopsy could have confirmed the diagnosis, but with the family history, classic appearance, and lack of response to antifungal medication, there was little doubt that this was a case of psoriasis. This autoimmune disease affects nearly 3% of the white population in this country and has a genetic component about 30% of the time.
In psoriasis, keratinocytes matriculate upward from the basal layer to the skin surface at four times the normal rate—so quickly that they have no chance to lose their nuclei (as they normally would). They then pile up, creating plaques of micaceous white scale on a salmon-pink base. Histologically, the smoothly undulating dermoepidermal junction is jammed together, producing fused ridges with clumps of neutrophils on their tips.
While it favors extensor surfaces of extremities, psoriasis can show up anywhere on the body—on the genitals, mouth, and in the nails, where it can cause pits, dystrophy, discoloration, onycholysis, and onychorrhexis.
Unfortunately, this is probably just the beginning of this child’s psoriasis. The good news is that we’re in a golden age of psoriasis treatment, with more drugs than ever to choose from and even more in development. For this patient, we used a keratolytic agent (urea lotion) to thin out the surface scale, in order to allow a class 4 steroid cream to reach the pink inflammatory portion. Within a month, most of this patch had cleared, though we can be fairly sure it and others like it will be back. Education and ongoing follow-up will be needed, in case she is among the 20% to 25% of patients who will develop psoriatic arthropathy, a crippling form of arthritis.
It is certainly possible to develop a fungal infection on or in an ear, but for that to happen, there has to be a source (eg, animal, person, soil). Moreover, the scale would look entirely different, with clearing centers and advancing margins. The likely truth is that this was called “fungal” for lack of any other suspects.
TAKE-HOME LEARNING POINTS
- White scale on a salmon-pink base typifies psoriasis vulgaris, a very common diagnosis that is often mistaken for fungal infection; biopsy can be extremely helpful in establishing or ruling out this diagnosis.
- Psoriasis has a genetic basis, with many gene loci identified to date, but only about 30% of affected patients can attest to a family history.
- In addition to having unsightly, often itchy lesions, psoriasis patients are also at risk for psoriatic arthropathy, a potentially crippling condition.
- The best news is that we have many drugs with which to treat this disease, including a whole family of drugs termed “the biologics,” which directly (and successfully!) address the disease.
About six months ago, an 8-year-old girl developed an asymptomatic rash near her ear. Her mother suspects it is psoriasis, which runs heavily in the family—but their primary care provider favors a fungal diagnosis. He prescribes a succession of topical and oral antifungal medications (including nystatin and terbinafine), which yield no discernable improvement. At this point, referral to dermatology is made.
The child’s mother denies any history of recent infections (eg, strep throat) on her daughter’s behalf. Furthermore, there are no reports of pain associated with the rash or elsewhere.
EXAMINATION
The rash, which is confined to the external right ear, is composed of uniformly smooth white scale on a faintly salmon base. The entire lesion measures about 3 cm at its widest point, and the margins are arciform and well-defined.

No such lesions are seen elsewhere, but tiny pits can be seen on one fingernail.
What is the diagnosis?
DISCUSSION
A punch biopsy could have confirmed the diagnosis, but with the family history, classic appearance, and lack of response to antifungal medication, there was little doubt that this was a case of psoriasis. This autoimmune disease affects nearly 3% of the white population in this country and has a genetic component about 30% of the time.
In psoriasis, keratinocytes matriculate upward from the basal layer to the skin surface at four times the normal rate—so quickly that they have no chance to lose their nuclei (as they normally would). They then pile up, creating plaques of micaceous white scale on a salmon-pink base. Histologically, the smoothly undulating dermoepidermal junction is jammed together, producing fused ridges with clumps of neutrophils on their tips.
While it favors extensor surfaces of extremities, psoriasis can show up anywhere on the body—on the genitals, mouth, and in the nails, where it can cause pits, dystrophy, discoloration, onycholysis, and onychorrhexis.
Unfortunately, this is probably just the beginning of this child’s psoriasis. The good news is that we’re in a golden age of psoriasis treatment, with more drugs than ever to choose from and even more in development. For this patient, we used a keratolytic agent (urea lotion) to thin out the surface scale, in order to allow a class 4 steroid cream to reach the pink inflammatory portion. Within a month, most of this patch had cleared, though we can be fairly sure it and others like it will be back. Education and ongoing follow-up will be needed, in case she is among the 20% to 25% of patients who will develop psoriatic arthropathy, a crippling form of arthritis.
It is certainly possible to develop a fungal infection on or in an ear, but for that to happen, there has to be a source (eg, animal, person, soil). Moreover, the scale would look entirely different, with clearing centers and advancing margins. The likely truth is that this was called “fungal” for lack of any other suspects.
TAKE-HOME LEARNING POINTS
- White scale on a salmon-pink base typifies psoriasis vulgaris, a very common diagnosis that is often mistaken for fungal infection; biopsy can be extremely helpful in establishing or ruling out this diagnosis.
- Psoriasis has a genetic basis, with many gene loci identified to date, but only about 30% of affected patients can attest to a family history.
- In addition to having unsightly, often itchy lesions, psoriasis patients are also at risk for psoriatic arthropathy, a potentially crippling condition.
- The best news is that we have many drugs with which to treat this disease, including a whole family of drugs termed “the biologics,” which directly (and successfully!) address the disease.
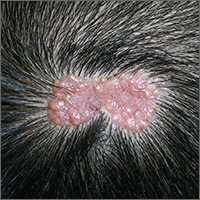
Growth on scalp
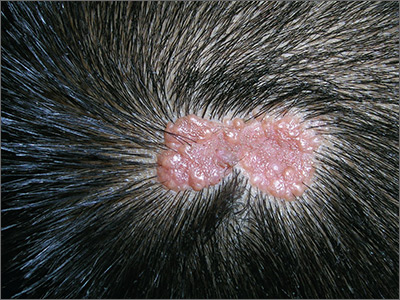
The family physician diagnosed a nevus sebaceous (NS) in this patient.
There are 3 stages of evolution paralleling the histologic differentiation of normal sebaceous glands:
- Infancy and young children. The lesion is smooth to slightly papillated, waxy, and hairless. (See Photo Rounds Friday, 6/15/18.)
- Puberty. Epidermal hyperplasia results in verrucous irregularity of the surface and coverage with numerous closely aggregated yellow-to-brown papules (this case).
- Development of secondary appendageal tumors. This occurs in 20% to 30% of patients. Most lesions are benign, but single (most commonly basal cell carcinoma) or multiple malignant tumors of both epidermal and adnexal origins may be seen. These malignancies are rarely seen in childhood.
In this case, a biopsy was not needed because the clinical picture was clear and no operative intervention was planned. When needed, a shave biopsy should provide adequate tissue for diagnosis because the pathology is epidermal and in the upper dermis. The NS need not be removed to prevent malignant transformation.
The FP explained that hair usually doesn’t grow where an NS is, and it was okay to proceed with observation only. He advised the patient’s father that if any changes were to occur, he would be happy to refer the child for surgical removal. The boy was not worried about the appearance of the NS and did not want to have surgery.
Photos and text for Photo Rounds Friday courtesy of Richard P. Usatine, MD. This case was adapted from: Smith M. Epidermal nevus and nevus sebaceous. In: Usatine R, Smith M, Mayeaux EJ, et al. Color Atlas of Family Medicine, 2nd ed. New York, NY: McGraw-Hill; 2013:958-962.
To learn more about the Color Atlas of Family Medicine, see: www.amazon.com/Color-Family-Medicine-Richard-Usatine/dp/0071769641/.
You can now get the second edition of the Color Atlas of Family Medicine as an app by clicking on this link: usatinemedia.com.

The family physician diagnosed a nevus sebaceous (NS) in this patient.
There are 3 stages of evolution paralleling the histologic differentiation of normal sebaceous glands:
- Infancy and young children. The lesion is smooth to slightly papillated, waxy, and hairless. (See Photo Rounds Friday, 6/15/18.)
- Puberty. Epidermal hyperplasia results in verrucous irregularity of the surface and coverage with numerous closely aggregated yellow-to-brown papules (this case).
- Development of secondary appendageal tumors. This occurs in 20% to 30% of patients. Most lesions are benign, but single (most commonly basal cell carcinoma) or multiple malignant tumors of both epidermal and adnexal origins may be seen. These malignancies are rarely seen in childhood.
In this case, a biopsy was not needed because the clinical picture was clear and no operative intervention was planned. When needed, a shave biopsy should provide adequate tissue for diagnosis because the pathology is epidermal and in the upper dermis. The NS need not be removed to prevent malignant transformation.
The FP explained that hair usually doesn’t grow where an NS is, and it was okay to proceed with observation only. He advised the patient’s father that if any changes were to occur, he would be happy to refer the child for surgical removal. The boy was not worried about the appearance of the NS and did not want to have surgery.
Photos and text for Photo Rounds Friday courtesy of Richard P. Usatine, MD. This case was adapted from: Smith M. Epidermal nevus and nevus sebaceous. In: Usatine R, Smith M, Mayeaux EJ, et al. Color Atlas of Family Medicine, 2nd ed. New York, NY: McGraw-Hill; 2013:958-962.
To learn more about the Color Atlas of Family Medicine, see: www.amazon.com/Color-Family-Medicine-Richard-Usatine/dp/0071769641/.
You can now get the second edition of the Color Atlas of Family Medicine as an app by clicking on this link: usatinemedia.com.

The family physician diagnosed a nevus sebaceous (NS) in this patient.
There are 3 stages of evolution paralleling the histologic differentiation of normal sebaceous glands:
- Infancy and young children. The lesion is smooth to slightly papillated, waxy, and hairless. (See Photo Rounds Friday, 6/15/18.)
- Puberty. Epidermal hyperplasia results in verrucous irregularity of the surface and coverage with numerous closely aggregated yellow-to-brown papules (this case).
- Development of secondary appendageal tumors. This occurs in 20% to 30% of patients. Most lesions are benign, but single (most commonly basal cell carcinoma) or multiple malignant tumors of both epidermal and adnexal origins may be seen. These malignancies are rarely seen in childhood.
In this case, a biopsy was not needed because the clinical picture was clear and no operative intervention was planned. When needed, a shave biopsy should provide adequate tissue for diagnosis because the pathology is epidermal and in the upper dermis. The NS need not be removed to prevent malignant transformation.
The FP explained that hair usually doesn’t grow where an NS is, and it was okay to proceed with observation only. He advised the patient’s father that if any changes were to occur, he would be happy to refer the child for surgical removal. The boy was not worried about the appearance of the NS and did not want to have surgery.
Photos and text for Photo Rounds Friday courtesy of Richard P. Usatine, MD. This case was adapted from: Smith M. Epidermal nevus and nevus sebaceous. In: Usatine R, Smith M, Mayeaux EJ, et al. Color Atlas of Family Medicine, 2nd ed. New York, NY: McGraw-Hill; 2013:958-962.
To learn more about the Color Atlas of Family Medicine, see: www.amazon.com/Color-Family-Medicine-Richard-Usatine/dp/0071769641/.
You can now get the second edition of the Color Atlas of Family Medicine as an app by clicking on this link: usatinemedia.com.
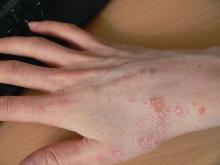
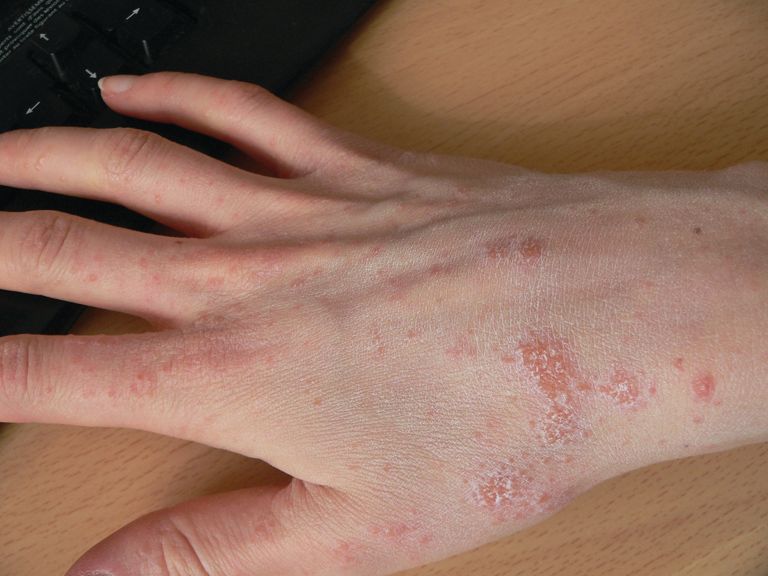
Relapsing scabies? Nails may hold a clue
cautioned Marie Chinazzo, MD, of Centre Hospitalier Régional et Universitaire Tours, France, and her associates.
Nails can harbor mites, representing a potential source for relapse, not only in children, but also in adults.
Few studies have addressed scabies on the nails, which is typically observed in immunocompromised adults with crusted scabies, but also rarely in healthy adults and children.
In an observational, multicenter, prospective study conducted between June 2015 and January 2017, 47 pediatric patients with common scabies, including 3 children under 2 years of age, presented with mites on the first toenail/thumbnail; two of them had already completed treatment and were experiencing relapse. All children with dermatologic diagnosis that was confirmed by visual inspection of “the delta sign” (presence of the mite seen as a triangle representing the head) using dermoscopy or by microscopic identification of Sarcoptes scabiei were included in the study. Dermatologists were required to complete a standardized questionnaire for each participant. Full body inspections and nail samplings also were done.
Clinical nail damage, consisting of hyperkeratosis, onycholysis, onychoschizia, and pachyonychia, appeared in 5 of the 47 patients (11%). No other cause of nail damage was determined in four of the cases, for which mites were not directly visualized, the researchers noted. The report was published in the Journal of Pediatrics.
Of the 47 confirmed cases, 26 were female; 23 were under 2 years of age; 20 were 2-12 years; and 4 were older than 12. Ten cases presented with significant medical history; none were classified as immunocompromised.
Fully 42 of the 47 children (89%) reported pruritus, and of these, 64% also had pruritus present in the family home; 60% of siblings and 45% of parents were affected.
None were diagnosed with crusted scabies. The mean delay from disease onset to diagnosis was 55 days. In 38% of cases, previous treatment for scabies had been rendered.
Treatments varied based on presentation. Ivermectin, esdepallethrin, and 40% urea were repeated after 10 days in at least one case. In another case, an entire family was treated once with topical 5% permethrin; once the child experienced relapse, oral ivermectin was employed. In the case of an 18-month-old girl with pruritus and skin lesions, topical corticosteroid was used for 10 days until such time that dermatoscopy revealed the “delta sign” and 5% topical permethrin was added.
The authors observed that nail scabies in the medical literature is more commonly seen in immunocompromised patients with crusted scabies and higher concentrations of parasites. They were able to locate only three other reports, all in adults, of nail scabies occurring with common scabies.
“Treatment of nail scabies is difficult and is not highly evidence based,” cautioned Dr. Chinazzo and her associates. The primary study limitations were the small patient population and that nail sampling was taken only from the first fingers and toes, which could mean that the number of mites present is actually underestimated, they added.
The authors had no relevant financial disclosures.
SOURCE: Chinazzo M et al. J Pediatr. 2018. doi: 10.1016/j.jpeds.2018.01.038.
cautioned Marie Chinazzo, MD, of Centre Hospitalier Régional et Universitaire Tours, France, and her associates.
Nails can harbor mites, representing a potential source for relapse, not only in children, but also in adults.
Few studies have addressed scabies on the nails, which is typically observed in immunocompromised adults with crusted scabies, but also rarely in healthy adults and children.
In an observational, multicenter, prospective study conducted between June 2015 and January 2017, 47 pediatric patients with common scabies, including 3 children under 2 years of age, presented with mites on the first toenail/thumbnail; two of them had already completed treatment and were experiencing relapse. All children with dermatologic diagnosis that was confirmed by visual inspection of “the delta sign” (presence of the mite seen as a triangle representing the head) using dermoscopy or by microscopic identification of Sarcoptes scabiei were included in the study. Dermatologists were required to complete a standardized questionnaire for each participant. Full body inspections and nail samplings also were done.
Clinical nail damage, consisting of hyperkeratosis, onycholysis, onychoschizia, and pachyonychia, appeared in 5 of the 47 patients (11%). No other cause of nail damage was determined in four of the cases, for which mites were not directly visualized, the researchers noted. The report was published in the Journal of Pediatrics.
Of the 47 confirmed cases, 26 were female; 23 were under 2 years of age; 20 were 2-12 years; and 4 were older than 12. Ten cases presented with significant medical history; none were classified as immunocompromised.
Fully 42 of the 47 children (89%) reported pruritus, and of these, 64% also had pruritus present in the family home; 60% of siblings and 45% of parents were affected.
None were diagnosed with crusted scabies. The mean delay from disease onset to diagnosis was 55 days. In 38% of cases, previous treatment for scabies had been rendered.
Treatments varied based on presentation. Ivermectin, esdepallethrin, and 40% urea were repeated after 10 days in at least one case. In another case, an entire family was treated once with topical 5% permethrin; once the child experienced relapse, oral ivermectin was employed. In the case of an 18-month-old girl with pruritus and skin lesions, topical corticosteroid was used for 10 days until such time that dermatoscopy revealed the “delta sign” and 5% topical permethrin was added.
The authors observed that nail scabies in the medical literature is more commonly seen in immunocompromised patients with crusted scabies and higher concentrations of parasites. They were able to locate only three other reports, all in adults, of nail scabies occurring with common scabies.
“Treatment of nail scabies is difficult and is not highly evidence based,” cautioned Dr. Chinazzo and her associates. The primary study limitations were the small patient population and that nail sampling was taken only from the first fingers and toes, which could mean that the number of mites present is actually underestimated, they added.
The authors had no relevant financial disclosures.
SOURCE: Chinazzo M et al. J Pediatr. 2018. doi: 10.1016/j.jpeds.2018.01.038.
cautioned Marie Chinazzo, MD, of Centre Hospitalier Régional et Universitaire Tours, France, and her associates.
Nails can harbor mites, representing a potential source for relapse, not only in children, but also in adults.
Few studies have addressed scabies on the nails, which is typically observed in immunocompromised adults with crusted scabies, but also rarely in healthy adults and children.
In an observational, multicenter, prospective study conducted between June 2015 and January 2017, 47 pediatric patients with common scabies, including 3 children under 2 years of age, presented with mites on the first toenail/thumbnail; two of them had already completed treatment and were experiencing relapse. All children with dermatologic diagnosis that was confirmed by visual inspection of “the delta sign” (presence of the mite seen as a triangle representing the head) using dermoscopy or by microscopic identification of Sarcoptes scabiei were included in the study. Dermatologists were required to complete a standardized questionnaire for each participant. Full body inspections and nail samplings also were done.
Clinical nail damage, consisting of hyperkeratosis, onycholysis, onychoschizia, and pachyonychia, appeared in 5 of the 47 patients (11%). No other cause of nail damage was determined in four of the cases, for which mites were not directly visualized, the researchers noted. The report was published in the Journal of Pediatrics.
Of the 47 confirmed cases, 26 were female; 23 were under 2 years of age; 20 were 2-12 years; and 4 were older than 12. Ten cases presented with significant medical history; none were classified as immunocompromised.
Fully 42 of the 47 children (89%) reported pruritus, and of these, 64% also had pruritus present in the family home; 60% of siblings and 45% of parents were affected.
None were diagnosed with crusted scabies. The mean delay from disease onset to diagnosis was 55 days. In 38% of cases, previous treatment for scabies had been rendered.
Treatments varied based on presentation. Ivermectin, esdepallethrin, and 40% urea were repeated after 10 days in at least one case. In another case, an entire family was treated once with topical 5% permethrin; once the child experienced relapse, oral ivermectin was employed. In the case of an 18-month-old girl with pruritus and skin lesions, topical corticosteroid was used for 10 days until such time that dermatoscopy revealed the “delta sign” and 5% topical permethrin was added.
The authors observed that nail scabies in the medical literature is more commonly seen in immunocompromised patients with crusted scabies and higher concentrations of parasites. They were able to locate only three other reports, all in adults, of nail scabies occurring with common scabies.
“Treatment of nail scabies is difficult and is not highly evidence based,” cautioned Dr. Chinazzo and her associates. The primary study limitations were the small patient population and that nail sampling was taken only from the first fingers and toes, which could mean that the number of mites present is actually underestimated, they added.
The authors had no relevant financial disclosures.
SOURCE: Chinazzo M et al. J Pediatr. 2018. doi: 10.1016/j.jpeds.2018.01.038.
FROM THE JOURNAL OF PEDIATRICS
Key clinical point: Pediatric relapse estimated as high as 66%.
Major finding: Nail scabies found in great toenail, not fingernails.
Study details: Observational multicenter prospective study of 47 pediatric patients with common scabies.
Disclosures: The authors had no relevant financial disclosures.
Source: Chinazzo M et al. J Pediatr. 2018. doi: 10.1016/j.jpeds.2018.01.038.
Inflammatory myopathy following tattoo reported in an immunocompromised patient
A case of inflammatory .
Writing in the June 18 online edition of BMJ Case Reports, clinicians described a 31-year-old woman who was on long-term immunosuppressive therapy after bilateral lung transplants for cystic fibrosis.
The woman received a large, colored tattoo on her upper leg, with no immediate complications beyond the usual mild skin irritation. However, 9 days later, she developed pain in her left thigh and knee that was severe enough to require analgesic treatment that included tramadol and paracetamol.
The pain settled over the following few months, but the woman continued to experience a sense of fullness from her hip to knee along the medial side of her thigh. She presented to a rheumatology clinic 10 months after she was tattooed, with pain that was still constant and disturbing her sleep, but with no apparent aggravating factors and, otherwise, she was in good health.
Work-up included an MRI that showed focal inflammation of the vastus medialis muscle, particularly in the distal third, but a biopsy found no bacterial growth, nor was there any bacterial or fungal infection found in fluid drawn from the knee. However, histopathology revealed scattered internal nuclei, atrophic fibers, a mild perivascular inflammatory infiltrate, and upregulation of human leukocyte antigen.
In the report, William T. Wilson, MD, and his colleagues from the department of trauma and orthopedics, NHS Greater Glasgow and Clyde, Glasgow, said that these findings gave the impression of an inflammatory myopathy in which the pathologic response may have been influenced by the immunosuppression.
“To our knowledge, there have been no previously reported cases of tattoo-associated reactions causing an inflammatory myopathy,” they wrote. “This could be a rare occurrence or represent an underdiagnosis for patients presenting with similar symptoms having had tattoos.”
The authors suggested there was a chance that the myopathy may have been stimulated by a toxin or pathogen introduced during tattoo procedure. However, they pointed out that they could not identify a causative pathogen, although the timing of onset and location of symptoms correlated with the tattoo application.
“This case serves as a reminder to consider tattoo-related complications as part of the differential diagnosis when patients, especially the immune-suppressed, present with unusual atraumatic musculoskeletal symptoms,” they wrote.
After the biopsy, the woman received physiotherapy in the form of basic quadriceps-strengthening exercises. Her condition did not start to improve until about 1 year after the onset of symptoms, and by 3 years, she had no more pain and had resumed normal activities.
No funding or conflicts of interest were declared.
SOURCE: Wilson W et al. BMJ Case Rep. 2018. Jun 18. doi: 10.1136/bcr-2018-224968.
A case of inflammatory .
Writing in the June 18 online edition of BMJ Case Reports, clinicians described a 31-year-old woman who was on long-term immunosuppressive therapy after bilateral lung transplants for cystic fibrosis.
The woman received a large, colored tattoo on her upper leg, with no immediate complications beyond the usual mild skin irritation. However, 9 days later, she developed pain in her left thigh and knee that was severe enough to require analgesic treatment that included tramadol and paracetamol.
The pain settled over the following few months, but the woman continued to experience a sense of fullness from her hip to knee along the medial side of her thigh. She presented to a rheumatology clinic 10 months after she was tattooed, with pain that was still constant and disturbing her sleep, but with no apparent aggravating factors and, otherwise, she was in good health.
Work-up included an MRI that showed focal inflammation of the vastus medialis muscle, particularly in the distal third, but a biopsy found no bacterial growth, nor was there any bacterial or fungal infection found in fluid drawn from the knee. However, histopathology revealed scattered internal nuclei, atrophic fibers, a mild perivascular inflammatory infiltrate, and upregulation of human leukocyte antigen.
In the report, William T. Wilson, MD, and his colleagues from the department of trauma and orthopedics, NHS Greater Glasgow and Clyde, Glasgow, said that these findings gave the impression of an inflammatory myopathy in which the pathologic response may have been influenced by the immunosuppression.
“To our knowledge, there have been no previously reported cases of tattoo-associated reactions causing an inflammatory myopathy,” they wrote. “This could be a rare occurrence or represent an underdiagnosis for patients presenting with similar symptoms having had tattoos.”
The authors suggested there was a chance that the myopathy may have been stimulated by a toxin or pathogen introduced during tattoo procedure. However, they pointed out that they could not identify a causative pathogen, although the timing of onset and location of symptoms correlated with the tattoo application.
“This case serves as a reminder to consider tattoo-related complications as part of the differential diagnosis when patients, especially the immune-suppressed, present with unusual atraumatic musculoskeletal symptoms,” they wrote.
After the biopsy, the woman received physiotherapy in the form of basic quadriceps-strengthening exercises. Her condition did not start to improve until about 1 year after the onset of symptoms, and by 3 years, she had no more pain and had resumed normal activities.
No funding or conflicts of interest were declared.
SOURCE: Wilson W et al. BMJ Case Rep. 2018. Jun 18. doi: 10.1136/bcr-2018-224968.
A case of inflammatory .
Writing in the June 18 online edition of BMJ Case Reports, clinicians described a 31-year-old woman who was on long-term immunosuppressive therapy after bilateral lung transplants for cystic fibrosis.
The woman received a large, colored tattoo on her upper leg, with no immediate complications beyond the usual mild skin irritation. However, 9 days later, she developed pain in her left thigh and knee that was severe enough to require analgesic treatment that included tramadol and paracetamol.
The pain settled over the following few months, but the woman continued to experience a sense of fullness from her hip to knee along the medial side of her thigh. She presented to a rheumatology clinic 10 months after she was tattooed, with pain that was still constant and disturbing her sleep, but with no apparent aggravating factors and, otherwise, she was in good health.
Work-up included an MRI that showed focal inflammation of the vastus medialis muscle, particularly in the distal third, but a biopsy found no bacterial growth, nor was there any bacterial or fungal infection found in fluid drawn from the knee. However, histopathology revealed scattered internal nuclei, atrophic fibers, a mild perivascular inflammatory infiltrate, and upregulation of human leukocyte antigen.
In the report, William T. Wilson, MD, and his colleagues from the department of trauma and orthopedics, NHS Greater Glasgow and Clyde, Glasgow, said that these findings gave the impression of an inflammatory myopathy in which the pathologic response may have been influenced by the immunosuppression.
“To our knowledge, there have been no previously reported cases of tattoo-associated reactions causing an inflammatory myopathy,” they wrote. “This could be a rare occurrence or represent an underdiagnosis for patients presenting with similar symptoms having had tattoos.”
The authors suggested there was a chance that the myopathy may have been stimulated by a toxin or pathogen introduced during tattoo procedure. However, they pointed out that they could not identify a causative pathogen, although the timing of onset and location of symptoms correlated with the tattoo application.
“This case serves as a reminder to consider tattoo-related complications as part of the differential diagnosis when patients, especially the immune-suppressed, present with unusual atraumatic musculoskeletal symptoms,” they wrote.
After the biopsy, the woman received physiotherapy in the form of basic quadriceps-strengthening exercises. Her condition did not start to improve until about 1 year after the onset of symptoms, and by 3 years, she had no more pain and had resumed normal activities.
No funding or conflicts of interest were declared.
SOURCE: Wilson W et al. BMJ Case Rep. 2018. Jun 18. doi: 10.1136/bcr-2018-224968.
FROM BMJ CASE REPORTS
Key clinical point: Consider tattoo-induced myopathy in immunosuppressed individuals.
Major finding: Tattooing has been linked to inflammatory myopathy in an immunosuppressed woman.
Study details: A report of a 31-year-old double lung transplant recipient who developed inflammatory myopathy after receiving a tattoo.
Disclosures: No funding or conflicts of interest were declared.
Source: Wilson W et al. BMJ Case Rep. 2018. Jun 18. doi:10.1136/bcr-2018-224968.
Impact of varicella vaccination on herpes zoster is not what was expected
MALMO, SWEDEN – The unique 20-year U.S. experience with pediatric universal varicella vaccination hasn’t resulted in the anticipated increase in herpes zoster predicted by the exogenous boosting hypothesis, Lara J. Wolfson, PhD, reported at the annual meeting of the European Society for Paediatric Infectious Diseases.
In fact, the opposite has occurred. And this finding – based upon hard data – should be of considerable interest to European health officials who have been considering introducing universal varicella vaccination into their national health care systems but have refrained because of theoretical concerns raised by the venerable exogenous boosting hypothesis, noted Dr. Wolfson, director of outcomes research at the Merck Center for Observational and Real-World Evidence, Kenilworth, N.J.

The exogenous boosting hypothesis, which dates back to the mid-1960s, holds that reexposure to wild circulating varicella virus prevents development of herpes zoster later in life. Conversely, by vaccinating children against varicella, opportunities are diminished for reexposure to wild type virus among adults who weren’t vaccinated against varicella, so the hypothesis would predict an increase in the incidence of herpes zoster that should peak 15-35 years after introduction of universal varicella vaccination.
“The same virus that causes varicella in children later reactivates after going dormant in the dorsal root ganglia, and it reactivates as herpes zoster, which is 10 times more severe than chicken pox and leads to 10 times the health care costs. So if in fact implementing a universal varicella vaccine program would lead to an increased incidence of herpes zoster, this would be a bad thing,” the researcher explained.
However, the predictive models based upon the exogenous boosting hypothesis are built upon scanty data. And the models have great difficulty in adjusting for the changes in population dynamics that have occurred in the United States and Western Europe during the past quarter century: namely, declining birth rates coupled with survival to an older age.
Dr. Wolfson presented a retrospective study of deidentified administrative claims data from the MarketScan database covering roughly one-fifth of the U.S. population during 1991-2016. Her analysis broke down the annual incidence of varicella and herpes zoster in three eras: 1991-1995, which was the pre–varicella vaccination period; 1996-2006, when single-dose universal varicella vaccination of children was recommended; and 2007-2016, when two-dose vaccination became standard.
The first key study finding was that herpes zoster rates in the United States already were climbing across all age groups back in 1991-1995; that is, before introduction of universal varicella vaccination. Why? Probably because of those changes in population dynamics, although that’s speculative. The second key finding was that contrary to the exogenous boosting hypothesis prediction that the annual incidence of herpes zoster would accelerate after introduction of universal varicella vaccination, the rate of increase slowed, then plateaued during 2013-2016, most prominently in individuals aged 65 or older.
“In comparing the pre–universal varicella vaccination period to the one- or two-dose period or the total 20 years of vaccination, what we saw consistently across every age group is that herpes zoster is decelerating. There is actually less increase in the rate of herpes zoster than before varicella vaccination,” Dr. Wolfson said.
Uptake of the herpes zoster vaccine, introduced in the United States in 2008, was too low during the study years to account for this trend, she added.
Most dramatically, the incidence of herpes zoster among youths under age 18 years plummeted by 61.4%, from 88 per 100,000 person-years in 1991-1995 to 34 per 100,000 in 2016.
And of course, varicella disease has sharply declined in all age groups following the introduction of universal pediatric varicella vaccination, Dr. Wolfson observed.
Her study was supported by her employer, Merck.
MALMO, SWEDEN – The unique 20-year U.S. experience with pediatric universal varicella vaccination hasn’t resulted in the anticipated increase in herpes zoster predicted by the exogenous boosting hypothesis, Lara J. Wolfson, PhD, reported at the annual meeting of the European Society for Paediatric Infectious Diseases.
In fact, the opposite has occurred. And this finding – based upon hard data – should be of considerable interest to European health officials who have been considering introducing universal varicella vaccination into their national health care systems but have refrained because of theoretical concerns raised by the venerable exogenous boosting hypothesis, noted Dr. Wolfson, director of outcomes research at the Merck Center for Observational and Real-World Evidence, Kenilworth, N.J.

The exogenous boosting hypothesis, which dates back to the mid-1960s, holds that reexposure to wild circulating varicella virus prevents development of herpes zoster later in life. Conversely, by vaccinating children against varicella, opportunities are diminished for reexposure to wild type virus among adults who weren’t vaccinated against varicella, so the hypothesis would predict an increase in the incidence of herpes zoster that should peak 15-35 years after introduction of universal varicella vaccination.
“The same virus that causes varicella in children later reactivates after going dormant in the dorsal root ganglia, and it reactivates as herpes zoster, which is 10 times more severe than chicken pox and leads to 10 times the health care costs. So if in fact implementing a universal varicella vaccine program would lead to an increased incidence of herpes zoster, this would be a bad thing,” the researcher explained.
However, the predictive models based upon the exogenous boosting hypothesis are built upon scanty data. And the models have great difficulty in adjusting for the changes in population dynamics that have occurred in the United States and Western Europe during the past quarter century: namely, declining birth rates coupled with survival to an older age.
Dr. Wolfson presented a retrospective study of deidentified administrative claims data from the MarketScan database covering roughly one-fifth of the U.S. population during 1991-2016. Her analysis broke down the annual incidence of varicella and herpes zoster in three eras: 1991-1995, which was the pre–varicella vaccination period; 1996-2006, when single-dose universal varicella vaccination of children was recommended; and 2007-2016, when two-dose vaccination became standard.
The first key study finding was that herpes zoster rates in the United States already were climbing across all age groups back in 1991-1995; that is, before introduction of universal varicella vaccination. Why? Probably because of those changes in population dynamics, although that’s speculative. The second key finding was that contrary to the exogenous boosting hypothesis prediction that the annual incidence of herpes zoster would accelerate after introduction of universal varicella vaccination, the rate of increase slowed, then plateaued during 2013-2016, most prominently in individuals aged 65 or older.
“In comparing the pre–universal varicella vaccination period to the one- or two-dose period or the total 20 years of vaccination, what we saw consistently across every age group is that herpes zoster is decelerating. There is actually less increase in the rate of herpes zoster than before varicella vaccination,” Dr. Wolfson said.
Uptake of the herpes zoster vaccine, introduced in the United States in 2008, was too low during the study years to account for this trend, she added.
Most dramatically, the incidence of herpes zoster among youths under age 18 years plummeted by 61.4%, from 88 per 100,000 person-years in 1991-1995 to 34 per 100,000 in 2016.
And of course, varicella disease has sharply declined in all age groups following the introduction of universal pediatric varicella vaccination, Dr. Wolfson observed.
Her study was supported by her employer, Merck.
MALMO, SWEDEN – The unique 20-year U.S. experience with pediatric universal varicella vaccination hasn’t resulted in the anticipated increase in herpes zoster predicted by the exogenous boosting hypothesis, Lara J. Wolfson, PhD, reported at the annual meeting of the European Society for Paediatric Infectious Diseases.
In fact, the opposite has occurred. And this finding – based upon hard data – should be of considerable interest to European health officials who have been considering introducing universal varicella vaccination into their national health care systems but have refrained because of theoretical concerns raised by the venerable exogenous boosting hypothesis, noted Dr. Wolfson, director of outcomes research at the Merck Center for Observational and Real-World Evidence, Kenilworth, N.J.

The exogenous boosting hypothesis, which dates back to the mid-1960s, holds that reexposure to wild circulating varicella virus prevents development of herpes zoster later in life. Conversely, by vaccinating children against varicella, opportunities are diminished for reexposure to wild type virus among adults who weren’t vaccinated against varicella, so the hypothesis would predict an increase in the incidence of herpes zoster that should peak 15-35 years after introduction of universal varicella vaccination.
“The same virus that causes varicella in children later reactivates after going dormant in the dorsal root ganglia, and it reactivates as herpes zoster, which is 10 times more severe than chicken pox and leads to 10 times the health care costs. So if in fact implementing a universal varicella vaccine program would lead to an increased incidence of herpes zoster, this would be a bad thing,” the researcher explained.
However, the predictive models based upon the exogenous boosting hypothesis are built upon scanty data. And the models have great difficulty in adjusting for the changes in population dynamics that have occurred in the United States and Western Europe during the past quarter century: namely, declining birth rates coupled with survival to an older age.
Dr. Wolfson presented a retrospective study of deidentified administrative claims data from the MarketScan database covering roughly one-fifth of the U.S. population during 1991-2016. Her analysis broke down the annual incidence of varicella and herpes zoster in three eras: 1991-1995, which was the pre–varicella vaccination period; 1996-2006, when single-dose universal varicella vaccination of children was recommended; and 2007-2016, when two-dose vaccination became standard.
The first key study finding was that herpes zoster rates in the United States already were climbing across all age groups back in 1991-1995; that is, before introduction of universal varicella vaccination. Why? Probably because of those changes in population dynamics, although that’s speculative. The second key finding was that contrary to the exogenous boosting hypothesis prediction that the annual incidence of herpes zoster would accelerate after introduction of universal varicella vaccination, the rate of increase slowed, then plateaued during 2013-2016, most prominently in individuals aged 65 or older.
“In comparing the pre–universal varicella vaccination period to the one- or two-dose period or the total 20 years of vaccination, what we saw consistently across every age group is that herpes zoster is decelerating. There is actually less increase in the rate of herpes zoster than before varicella vaccination,” Dr. Wolfson said.
Uptake of the herpes zoster vaccine, introduced in the United States in 2008, was too low during the study years to account for this trend, she added.
Most dramatically, the incidence of herpes zoster among youths under age 18 years plummeted by 61.4%, from 88 per 100,000 person-years in 1991-1995 to 34 per 100,000 in 2016.
And of course, varicella disease has sharply declined in all age groups following the introduction of universal pediatric varicella vaccination, Dr. Wolfson observed.
Her study was supported by her employer, Merck.
REPORTING FROM ESPID 2018
Key clinical point: The exogenous boosting hypothesis that universal pediatric varicella vaccination would result in an increase in herpes zoster hasn’t been borne out by the U.S. experience.
Major finding: rather than accelerating as some had forecast.
Study details: This was a retrospective study of the annual incidence of varicella and herpes zoster during 1991-2016 in roughly one-fifth of the U.S. population.
Disclosures: The study was sponsored by Merck and presented by a company employee.
Growth behind infant’s ear
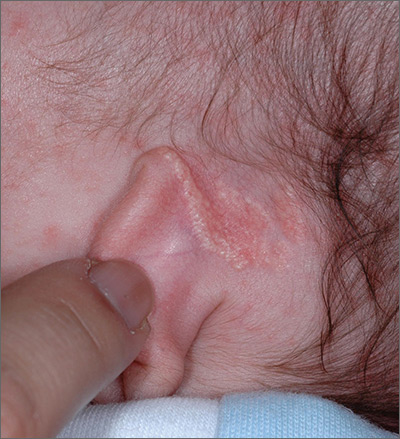
The FP recognized the growth as an early nevus sebaceous (NS).
NS may be present at birth or noted in early childhood and occurs in males and females equally. In early stages of development, it appears skin-colored and waxy. Because of the potential for malignant transformation, particularly following puberty, many authors have recommended early complete plastic surgical excision. However, in a retrospective analysis of 757 cases of NS from 1996 to 2002 in children <16 years, investigators found no malignancies and questioned the need for prophylactic surgical removal.
The FP emphasized that this condition was benign and would likely get darker and more raised over time. He said that he would keep an eye on it during future visits and that the boy could choose to have it removed when he became an adult. The FP explained that reasons for removal include cosmetic issues and the prevention of malignant changes.
Photos and text for Photo Rounds Friday courtesy of Richard P. Usatine, MD. This case was adapted from: Smith M. Epidermal nevus and nevus sebaceous. In: Usatine R, Smith M, Mayeaux EJ, et al. Color Atlas of Family Medicine, 2nd ed. New York, NY: McGraw-Hill; 2013:958-962.
To learn more about the Color Atlas of Family Medicine, see: www.amazon.com/Color-Family-Medicine-Richard-Usatine/dp/0071769641/.
You can now get the second edition of the Color Atlas of Family Medicine as an app by clicking on this link: usatinemedia.com.

The FP recognized the growth as an early nevus sebaceous (NS).
NS may be present at birth or noted in early childhood and occurs in males and females equally. In early stages of development, it appears skin-colored and waxy. Because of the potential for malignant transformation, particularly following puberty, many authors have recommended early complete plastic surgical excision. However, in a retrospective analysis of 757 cases of NS from 1996 to 2002 in children <16 years, investigators found no malignancies and questioned the need for prophylactic surgical removal.
The FP emphasized that this condition was benign and would likely get darker and more raised over time. He said that he would keep an eye on it during future visits and that the boy could choose to have it removed when he became an adult. The FP explained that reasons for removal include cosmetic issues and the prevention of malignant changes.
Photos and text for Photo Rounds Friday courtesy of Richard P. Usatine, MD. This case was adapted from: Smith M. Epidermal nevus and nevus sebaceous. In: Usatine R, Smith M, Mayeaux EJ, et al. Color Atlas of Family Medicine, 2nd ed. New York, NY: McGraw-Hill; 2013:958-962.
To learn more about the Color Atlas of Family Medicine, see: www.amazon.com/Color-Family-Medicine-Richard-Usatine/dp/0071769641/.
You can now get the second edition of the Color Atlas of Family Medicine as an app by clicking on this link: usatinemedia.com.

The FP recognized the growth as an early nevus sebaceous (NS).
NS may be present at birth or noted in early childhood and occurs in males and females equally. In early stages of development, it appears skin-colored and waxy. Because of the potential for malignant transformation, particularly following puberty, many authors have recommended early complete plastic surgical excision. However, in a retrospective analysis of 757 cases of NS from 1996 to 2002 in children <16 years, investigators found no malignancies and questioned the need for prophylactic surgical removal.
The FP emphasized that this condition was benign and would likely get darker and more raised over time. He said that he would keep an eye on it during future visits and that the boy could choose to have it removed when he became an adult. The FP explained that reasons for removal include cosmetic issues and the prevention of malignant changes.
Photos and text for Photo Rounds Friday courtesy of Richard P. Usatine, MD. This case was adapted from: Smith M. Epidermal nevus and nevus sebaceous. In: Usatine R, Smith M, Mayeaux EJ, et al. Color Atlas of Family Medicine, 2nd ed. New York, NY: McGraw-Hill; 2013:958-962.
To learn more about the Color Atlas of Family Medicine, see: www.amazon.com/Color-Family-Medicine-Richard-Usatine/dp/0071769641/.
You can now get the second edition of the Color Atlas of Family Medicine as an app by clicking on this link: usatinemedia.com.