User login
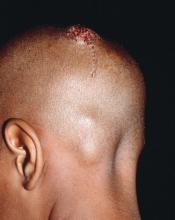
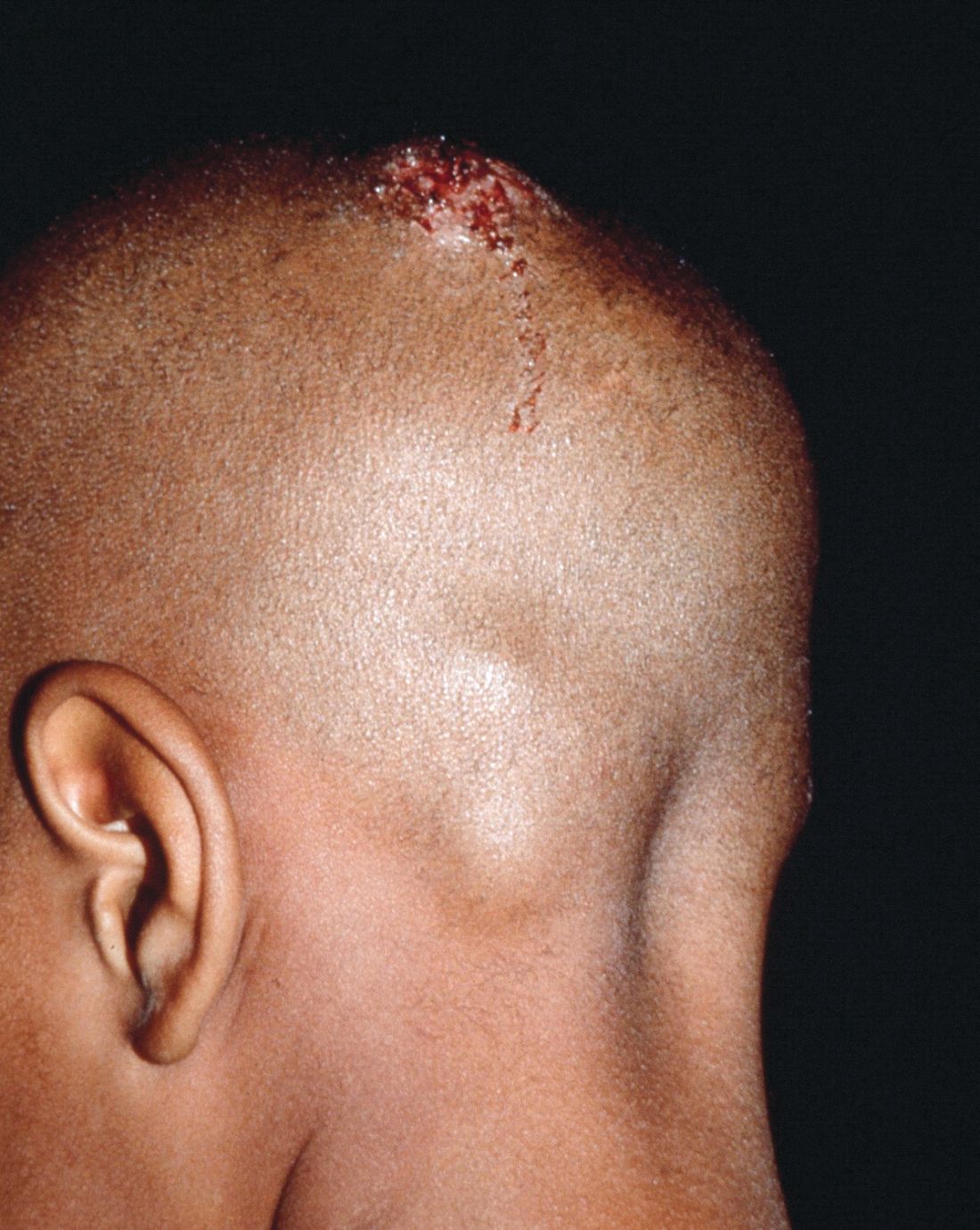
Early diagnosis, treatment key to prevent permanent baldness in tinea capitis
and the substantially reduced overall quality of health, reported Aditya K. Gupta, MD, PhD, of Mediprobe Research and the University of Toronto, and his associates.
In a systematic review of both randomized, controlled trials and clinical trials published before June 1, 2017, the authors sought to identify differences between treatment medications and significant adverse side effects, and to evaluate the most effective methods for diagnosis. The study criteria included trials with clinical and mycologic diagnosis of tinea capitis, evaluation of efficacy rates and/or safety measures in participants aged 18 years or younger, yielded a total of 4,190 studies in this article published in Pediatric Dermatology.
Dr. Gupta and his colleagues evaluated efficacy rates that reported on mycologic cure (negative mycologic testing), clinical cure (complete absence of signs and symptoms), and complete cure (both mycologic and clinical cure). Trichophyton tonsurans was the most common organism reported in North America, and Wood’s light examination/light microscopic examination was the most common hair sample collection method identified.
In a population of 3,998 children who received treatment across all studies, five oral antifungals were used (terbinafine, griseofulvin, itraconazole, ketoconazole, and fluconazole). In addition, several studies examined the safety and effectiveness of combined oral and topical treatment in 833 children, while 25 children received topical-only therapy.
Although topical treatment may be useful adjunctively, some studies noted that oral treatment is necessary for effective resolution of tinea capitis. While some experts recommend continuing topical treatment until clinical and mycologic cure are achieved, the authors cautioned that “the presence of a topical antifungal in a culture media would likely lead to a false negative result, so a clinical confirmation is necessary.”
Adverse events
Altogether, 295 drug-related adverse effects were reported: 51.2% from terbinafine, 26.8% from griseofulvin, 12.2% from fluconazole, 8.5% from itraconazole, and 1.4% from ketoconazole; all were transient and mild to moderate in severity.
Of the total population observed, just 50 children (1.3% of 3,998) ceased treatment because of adverse effects of the medication.
Therapy choices
Of the 75 antifungal treatment combinations identified, cure rates were highest with continuous itraconazole and terbinafine (mycologic), griseofulvin and terbinafine (clinical), and griseofulvin and terbinafine (complete). Griseofulvin was more effective at treating Microsporum than Trichophyton infections, fluconazole was comparably effective in treating both Microsporum and Trichophyton infections, and continuous itraconazole and terbinafine were more effective at curing Trichophyton infections than Microsporum, noted Dr. Gupta and his associates.
Terbinafine treatment for Trichophyton infections was found to be effective at just 4 weeks, however, oral terbinafine was singularly responsible for more than half of adverse events reported. The authors suggested that this might be from its extensive biodistribution. In such cases, the authors recommended baseline monitoring of transaminase.
Although griseofulvin is the most widely prescribed medication for pediatric tinea capitis, primarily because of its cost effectiveness and accessibility, a 2016 Cochrane review found that newer treatments – terbinafine, itraconazole and fluconazole – offer comparative effectiveness in cases of Trichophyton infection. The relatively higher cost of these treatments and the prevalence of tinea capitis in lower socioeconomic populations, however, may render them impractical, the authors noted. As recent clinical trials have suggested significantly larger, weight-normalized doses are required in children to approximate the exposure estimates of adults, this should be of key consideration when choosing appropriate, cost-effective treatments.
Diagnostic issues
T. tonsurans cases of tinea capitis are most prevalent in North America, and recent data suggest they are on the rise. The organism typically infects human skin and hair, and can to survive for lengthy periods on inanimate objects, including combs, brushes, sheets, and blankets. Researchers credit the growing number of cases in North America to several factors. Infections from the fungus have become increasingly common in the United States and Canada as a consequence of changing travel and immigration patterns. In addition, many physicians still turn to fluorescence (Wood’s light examination) in diagnosing tinea capitis, but T. tonsurans does not show up with fluorescence and typically does not present with the classic black dots characteristic of other fungal species. As a result, many cases in North America are misdiagnosed as seborrhea, dandruff, and impetigo, and subsequently undertreated, leading to spread of the infection. It was noted that more than half of the included studies used some form of Wood’s light examination.
Of all the techniques addressed, microscopy was found to be the fastest, but not always the most accurate, means of diagnosing tinea capitis. Dr. Gupta and his associates advised that diagnosis confirmation and precise species identification is best obtained with cultured scrapings, but this process can take 3 weeks or longer.
While fomites and hair care practices play a key role in tinea capitis infection, large family size, crowded living conditions, and low socioeconomic status are predisposing factors. Those who come in contact with infected patients should be considered possible asymptomatic carriers and be evaluated accordingly for treatment and to prevent spread of infection, the authors advised. Furthermore, recent studies recognized the impracticality of isolating children recently treated with oral therapy from classrooms since shedding of spores can continue for months.
The researchers had no relevant financial disclosures to report.
SOURCE: Gupta AK et al. Ped Dermatol. 2018 May 24. doi: 10.1111/jdv.15088.
and the substantially reduced overall quality of health, reported Aditya K. Gupta, MD, PhD, of Mediprobe Research and the University of Toronto, and his associates.
In a systematic review of both randomized, controlled trials and clinical trials published before June 1, 2017, the authors sought to identify differences between treatment medications and significant adverse side effects, and to evaluate the most effective methods for diagnosis. The study criteria included trials with clinical and mycologic diagnosis of tinea capitis, evaluation of efficacy rates and/or safety measures in participants aged 18 years or younger, yielded a total of 4,190 studies in this article published in Pediatric Dermatology.
Dr. Gupta and his colleagues evaluated efficacy rates that reported on mycologic cure (negative mycologic testing), clinical cure (complete absence of signs and symptoms), and complete cure (both mycologic and clinical cure). Trichophyton tonsurans was the most common organism reported in North America, and Wood’s light examination/light microscopic examination was the most common hair sample collection method identified.
In a population of 3,998 children who received treatment across all studies, five oral antifungals were used (terbinafine, griseofulvin, itraconazole, ketoconazole, and fluconazole). In addition, several studies examined the safety and effectiveness of combined oral and topical treatment in 833 children, while 25 children received topical-only therapy.
Although topical treatment may be useful adjunctively, some studies noted that oral treatment is necessary for effective resolution of tinea capitis. While some experts recommend continuing topical treatment until clinical and mycologic cure are achieved, the authors cautioned that “the presence of a topical antifungal in a culture media would likely lead to a false negative result, so a clinical confirmation is necessary.”
Adverse events
Altogether, 295 drug-related adverse effects were reported: 51.2% from terbinafine, 26.8% from griseofulvin, 12.2% from fluconazole, 8.5% from itraconazole, and 1.4% from ketoconazole; all were transient and mild to moderate in severity.
Of the total population observed, just 50 children (1.3% of 3,998) ceased treatment because of adverse effects of the medication.
Therapy choices
Of the 75 antifungal treatment combinations identified, cure rates were highest with continuous itraconazole and terbinafine (mycologic), griseofulvin and terbinafine (clinical), and griseofulvin and terbinafine (complete). Griseofulvin was more effective at treating Microsporum than Trichophyton infections, fluconazole was comparably effective in treating both Microsporum and Trichophyton infections, and continuous itraconazole and terbinafine were more effective at curing Trichophyton infections than Microsporum, noted Dr. Gupta and his associates.
Terbinafine treatment for Trichophyton infections was found to be effective at just 4 weeks, however, oral terbinafine was singularly responsible for more than half of adverse events reported. The authors suggested that this might be from its extensive biodistribution. In such cases, the authors recommended baseline monitoring of transaminase.
Although griseofulvin is the most widely prescribed medication for pediatric tinea capitis, primarily because of its cost effectiveness and accessibility, a 2016 Cochrane review found that newer treatments – terbinafine, itraconazole and fluconazole – offer comparative effectiveness in cases of Trichophyton infection. The relatively higher cost of these treatments and the prevalence of tinea capitis in lower socioeconomic populations, however, may render them impractical, the authors noted. As recent clinical trials have suggested significantly larger, weight-normalized doses are required in children to approximate the exposure estimates of adults, this should be of key consideration when choosing appropriate, cost-effective treatments.
Diagnostic issues
T. tonsurans cases of tinea capitis are most prevalent in North America, and recent data suggest they are on the rise. The organism typically infects human skin and hair, and can to survive for lengthy periods on inanimate objects, including combs, brushes, sheets, and blankets. Researchers credit the growing number of cases in North America to several factors. Infections from the fungus have become increasingly common in the United States and Canada as a consequence of changing travel and immigration patterns. In addition, many physicians still turn to fluorescence (Wood’s light examination) in diagnosing tinea capitis, but T. tonsurans does not show up with fluorescence and typically does not present with the classic black dots characteristic of other fungal species. As a result, many cases in North America are misdiagnosed as seborrhea, dandruff, and impetigo, and subsequently undertreated, leading to spread of the infection. It was noted that more than half of the included studies used some form of Wood’s light examination.
Of all the techniques addressed, microscopy was found to be the fastest, but not always the most accurate, means of diagnosing tinea capitis. Dr. Gupta and his associates advised that diagnosis confirmation and precise species identification is best obtained with cultured scrapings, but this process can take 3 weeks or longer.
While fomites and hair care practices play a key role in tinea capitis infection, large family size, crowded living conditions, and low socioeconomic status are predisposing factors. Those who come in contact with infected patients should be considered possible asymptomatic carriers and be evaluated accordingly for treatment and to prevent spread of infection, the authors advised. Furthermore, recent studies recognized the impracticality of isolating children recently treated with oral therapy from classrooms since shedding of spores can continue for months.
The researchers had no relevant financial disclosures to report.
SOURCE: Gupta AK et al. Ped Dermatol. 2018 May 24. doi: 10.1111/jdv.15088.
and the substantially reduced overall quality of health, reported Aditya K. Gupta, MD, PhD, of Mediprobe Research and the University of Toronto, and his associates.
In a systematic review of both randomized, controlled trials and clinical trials published before June 1, 2017, the authors sought to identify differences between treatment medications and significant adverse side effects, and to evaluate the most effective methods for diagnosis. The study criteria included trials with clinical and mycologic diagnosis of tinea capitis, evaluation of efficacy rates and/or safety measures in participants aged 18 years or younger, yielded a total of 4,190 studies in this article published in Pediatric Dermatology.
Dr. Gupta and his colleagues evaluated efficacy rates that reported on mycologic cure (negative mycologic testing), clinical cure (complete absence of signs and symptoms), and complete cure (both mycologic and clinical cure). Trichophyton tonsurans was the most common organism reported in North America, and Wood’s light examination/light microscopic examination was the most common hair sample collection method identified.
In a population of 3,998 children who received treatment across all studies, five oral antifungals were used (terbinafine, griseofulvin, itraconazole, ketoconazole, and fluconazole). In addition, several studies examined the safety and effectiveness of combined oral and topical treatment in 833 children, while 25 children received topical-only therapy.
Although topical treatment may be useful adjunctively, some studies noted that oral treatment is necessary for effective resolution of tinea capitis. While some experts recommend continuing topical treatment until clinical and mycologic cure are achieved, the authors cautioned that “the presence of a topical antifungal in a culture media would likely lead to a false negative result, so a clinical confirmation is necessary.”
Adverse events
Altogether, 295 drug-related adverse effects were reported: 51.2% from terbinafine, 26.8% from griseofulvin, 12.2% from fluconazole, 8.5% from itraconazole, and 1.4% from ketoconazole; all were transient and mild to moderate in severity.
Of the total population observed, just 50 children (1.3% of 3,998) ceased treatment because of adverse effects of the medication.
Therapy choices
Of the 75 antifungal treatment combinations identified, cure rates were highest with continuous itraconazole and terbinafine (mycologic), griseofulvin and terbinafine (clinical), and griseofulvin and terbinafine (complete). Griseofulvin was more effective at treating Microsporum than Trichophyton infections, fluconazole was comparably effective in treating both Microsporum and Trichophyton infections, and continuous itraconazole and terbinafine were more effective at curing Trichophyton infections than Microsporum, noted Dr. Gupta and his associates.
Terbinafine treatment for Trichophyton infections was found to be effective at just 4 weeks, however, oral terbinafine was singularly responsible for more than half of adverse events reported. The authors suggested that this might be from its extensive biodistribution. In such cases, the authors recommended baseline monitoring of transaminase.
Although griseofulvin is the most widely prescribed medication for pediatric tinea capitis, primarily because of its cost effectiveness and accessibility, a 2016 Cochrane review found that newer treatments – terbinafine, itraconazole and fluconazole – offer comparative effectiveness in cases of Trichophyton infection. The relatively higher cost of these treatments and the prevalence of tinea capitis in lower socioeconomic populations, however, may render them impractical, the authors noted. As recent clinical trials have suggested significantly larger, weight-normalized doses are required in children to approximate the exposure estimates of adults, this should be of key consideration when choosing appropriate, cost-effective treatments.
Diagnostic issues
T. tonsurans cases of tinea capitis are most prevalent in North America, and recent data suggest they are on the rise. The organism typically infects human skin and hair, and can to survive for lengthy periods on inanimate objects, including combs, brushes, sheets, and blankets. Researchers credit the growing number of cases in North America to several factors. Infections from the fungus have become increasingly common in the United States and Canada as a consequence of changing travel and immigration patterns. In addition, many physicians still turn to fluorescence (Wood’s light examination) in diagnosing tinea capitis, but T. tonsurans does not show up with fluorescence and typically does not present with the classic black dots characteristic of other fungal species. As a result, many cases in North America are misdiagnosed as seborrhea, dandruff, and impetigo, and subsequently undertreated, leading to spread of the infection. It was noted that more than half of the included studies used some form of Wood’s light examination.
Of all the techniques addressed, microscopy was found to be the fastest, but not always the most accurate, means of diagnosing tinea capitis. Dr. Gupta and his associates advised that diagnosis confirmation and precise species identification is best obtained with cultured scrapings, but this process can take 3 weeks or longer.
While fomites and hair care practices play a key role in tinea capitis infection, large family size, crowded living conditions, and low socioeconomic status are predisposing factors. Those who come in contact with infected patients should be considered possible asymptomatic carriers and be evaluated accordingly for treatment and to prevent spread of infection, the authors advised. Furthermore, recent studies recognized the impracticality of isolating children recently treated with oral therapy from classrooms since shedding of spores can continue for months.
The researchers had no relevant financial disclosures to report.
SOURCE: Gupta AK et al. Ped Dermatol. 2018 May 24. doi: 10.1111/jdv.15088.
FROM PEDIATRIC DERMATOLOGY
Key clinical point: The psychosocial impact and overall lower quality of health associated with tinea capitis is significant.
Major finding: Wood’s light should not be the only method of organism identification.
Study details: A systematic literature review of 4,190 studies.
Disclosures: The researchers had no relevant financial disclosures.
Source: Gupta AK et al. Ped Dermatol. 2018 May 24. doi: 10.1111/jdv.15088.
Nevus count tied to BCC risk
ORLANDO – The more , according to a review of over 200,000 subjects in decades-long health professional cohorts.
It’s well known that nevi increase the risk of melanoma, and the study confirmed that fact. The basal cell carcinoma finding, however, is novel. “The relationship between nevi and non-melanoma skin cancer has not [previously] been clearly demonstrated in large population cohorts,” said lead investigator Erin X. Wei, MD, a dermatologist at Brigham and Women’s Hospital, Boston.
“Nevus count serves as a convenient maker to identify patients at risk for both melanoma and basal cell carcinoma. Providers should be aware of these increased risks in patients with any nevi on the extremity, particularly 15 or more,” she said at the International Investigative Dermatology meeting.
There was no association, meanwhile, between nevus counts and squamous cell carcinoma (SCC).
The team reviewed 176,317 women in the Nurses’ Health Study 1 and 2, as well as 32,383 men in the Health Professionals Follow-up Study. Subjects were enrolled in the 1980s and followed through 2012. They reported nevus counts on their arms or legs at baseline, and filled out questionnaires on a regular basis that, among many other things, asked about new skin cancer diagnoses.
Overall, there were 30,457 incident basal cell carcinomas (BCCs), 1,704 incident melanomas, and 2,296 incident SCCs. Melanomas and SCCs – as well as a portion of BCCs – were confirmed by histology.
The team correlated the skin cancer incidence with how many moles subjects reported at baseline: zero, 1-5, 6-14, or 15 or more.
“Surprisingly, having any nevi on an extremity was associated with a significant increase in the risk of basal cell carcinoma,” in a dose-dependent manner, with 15 or more conferring a 40% increased risk of BCC, compared to subjects with no extremity nevi, Dr. Wei said (P less than .0001).
Even one mole also increased the risk of melanoma; having six or more nearly tripled it, again in a dose-dependent fashion (P less than .0001). Extremity nevi increased the risk of melanoma across all anatomic sites, including head, neck, and trunk.
The findings were statistically significant, and adjusted for age, body mass index, smoking, sun exposure, sunburn history, and other confounders.
There was no industry funding for the work, and the investigators had no relevant disclosures.
SOURCE: Wei EX et al. 2018 International Investigative Dermatology meeting abstract 233
ORLANDO – The more , according to a review of over 200,000 subjects in decades-long health professional cohorts.
It’s well known that nevi increase the risk of melanoma, and the study confirmed that fact. The basal cell carcinoma finding, however, is novel. “The relationship between nevi and non-melanoma skin cancer has not [previously] been clearly demonstrated in large population cohorts,” said lead investigator Erin X. Wei, MD, a dermatologist at Brigham and Women’s Hospital, Boston.
“Nevus count serves as a convenient maker to identify patients at risk for both melanoma and basal cell carcinoma. Providers should be aware of these increased risks in patients with any nevi on the extremity, particularly 15 or more,” she said at the International Investigative Dermatology meeting.
There was no association, meanwhile, between nevus counts and squamous cell carcinoma (SCC).
The team reviewed 176,317 women in the Nurses’ Health Study 1 and 2, as well as 32,383 men in the Health Professionals Follow-up Study. Subjects were enrolled in the 1980s and followed through 2012. They reported nevus counts on their arms or legs at baseline, and filled out questionnaires on a regular basis that, among many other things, asked about new skin cancer diagnoses.
Overall, there were 30,457 incident basal cell carcinomas (BCCs), 1,704 incident melanomas, and 2,296 incident SCCs. Melanomas and SCCs – as well as a portion of BCCs – were confirmed by histology.
The team correlated the skin cancer incidence with how many moles subjects reported at baseline: zero, 1-5, 6-14, or 15 or more.
“Surprisingly, having any nevi on an extremity was associated with a significant increase in the risk of basal cell carcinoma,” in a dose-dependent manner, with 15 or more conferring a 40% increased risk of BCC, compared to subjects with no extremity nevi, Dr. Wei said (P less than .0001).
Even one mole also increased the risk of melanoma; having six or more nearly tripled it, again in a dose-dependent fashion (P less than .0001). Extremity nevi increased the risk of melanoma across all anatomic sites, including head, neck, and trunk.
The findings were statistically significant, and adjusted for age, body mass index, smoking, sun exposure, sunburn history, and other confounders.
There was no industry funding for the work, and the investigators had no relevant disclosures.
SOURCE: Wei EX et al. 2018 International Investigative Dermatology meeting abstract 233
ORLANDO – The more , according to a review of over 200,000 subjects in decades-long health professional cohorts.
It’s well known that nevi increase the risk of melanoma, and the study confirmed that fact. The basal cell carcinoma finding, however, is novel. “The relationship between nevi and non-melanoma skin cancer has not [previously] been clearly demonstrated in large population cohorts,” said lead investigator Erin X. Wei, MD, a dermatologist at Brigham and Women’s Hospital, Boston.
“Nevus count serves as a convenient maker to identify patients at risk for both melanoma and basal cell carcinoma. Providers should be aware of these increased risks in patients with any nevi on the extremity, particularly 15 or more,” she said at the International Investigative Dermatology meeting.
There was no association, meanwhile, between nevus counts and squamous cell carcinoma (SCC).
The team reviewed 176,317 women in the Nurses’ Health Study 1 and 2, as well as 32,383 men in the Health Professionals Follow-up Study. Subjects were enrolled in the 1980s and followed through 2012. They reported nevus counts on their arms or legs at baseline, and filled out questionnaires on a regular basis that, among many other things, asked about new skin cancer diagnoses.
Overall, there were 30,457 incident basal cell carcinomas (BCCs), 1,704 incident melanomas, and 2,296 incident SCCs. Melanomas and SCCs – as well as a portion of BCCs – were confirmed by histology.
The team correlated the skin cancer incidence with how many moles subjects reported at baseline: zero, 1-5, 6-14, or 15 or more.
“Surprisingly, having any nevi on an extremity was associated with a significant increase in the risk of basal cell carcinoma,” in a dose-dependent manner, with 15 or more conferring a 40% increased risk of BCC, compared to subjects with no extremity nevi, Dr. Wei said (P less than .0001).
Even one mole also increased the risk of melanoma; having six or more nearly tripled it, again in a dose-dependent fashion (P less than .0001). Extremity nevi increased the risk of melanoma across all anatomic sites, including head, neck, and trunk.
The findings were statistically significant, and adjusted for age, body mass index, smoking, sun exposure, sunburn history, and other confounders.
There was no industry funding for the work, and the investigators had no relevant disclosures.
SOURCE: Wei EX et al. 2018 International Investigative Dermatology meeting abstract 233
REPORTING FROM IID 2018
Key clinical point: The more nevi a person has, the greater the risk of basal cell carcinoma.
Major finding: Having 15 or more moles on the arms and legs increased the risk 40% (P less than .0001).
Study details: Review of over 200,000 subjects in decades-long health professional cohorts
Disclosures: There was no industry funding for the work, and the investigators had no relevant disclosures.
Source: Wei EX et al. 2018 International Investigative Dermatology meeting abstract 233
Allergies linked to autism spectrum disorder in children
The prevalence of food, respiratory, and skin allergy is greater in U.S. children with autism spectrum disorder (ASD) than in U.S. children without the disorder, according to findings published June 8 in JAMA Network Open.
An analysis of data from the National Health Interview Survey found that the weighted prevalence of food, respiratory, and skin allergies was 11.25%, 18.73%, and 16.81%, respectively, in children with ASD, compared with 4.25%, 12.08%, and 9.84%, respectively, in children without ASD (P less than .001).
Survey data were collected between 1997 and 2016, and included patients aged 3-17 years. Allergic conditions were defined by the respondent, usually a parent, answering in the affirmative that the child had any kind of food, digestive, respiratory, or skin allergy in the past 12 months. ASD was defined based on an affirmative response to a question asking whether the child received an ASD diagnosis from a health professional. The question was asked as part of a 10-condition checklist from 1997 to 2013, and as a standalone item from 2014 onward, with revised wording to distinguish autism, Asperger’s disorder, pervasive developmental disorder, and ASD, wrote Guifeng Xu, MD, of the department of epidemiology at the University of Iowa, Iowa City, and her coauthors.
Of the 199,520 children included in the study, 8,734 had food allergy, 24,555 had respiratory allergy, and 19,399 had skin allergy. An ASD diagnosis was reported in 1,868 children. The weighted prevalence was 4.31% for food allergy (95% confidence interval, 4.20%-4.43%), 12.15% for respiratory allergy (95% CI, 11.92%-12.38%), and 9.91% for skin allergy (95% CI, 9.72%-10.10%), the authors said.
Children with ASD were more likely than were children without ASD to have food allergy, respiratory allergy, and skin allergy (P less than .001). After adjustment for factors including age, sex, ethnicity, and family education level, the odds ratio of ASD was more than double among children with food allergy, compared with those without food allergy (odds ratio, 2.72; 95% CI, 2.26-3.28; P less than .001).
Respiratory allergy and skin allergy also were significantly associated with ASD, but to a lesser degree, with an OR of 1.53 (95% CI, 1.32-1.78; P less than .001) for respiratory allergy and 1.80 (95% CI, 1.55-2.09; P less than .001) for skin allergy, Dr. Xu and her colleagues reported.
,” though the underlying mechanisms still need to be identified, the authors wrote.
Particularly striking were the connections found between ASD and food allergy. “Although the underlying mechanisms for the observed association between food allergy and ASD remain to be elucidated, the gut-brain-behavior axis could be one of the potential mechanisms,” Dr. Xu and her coauthors wrote. “Previous studies found higher prevalence of gastrointestinal symptoms among children with ASD.”
Limitations to the study include possible recall bias and misreporting, as well as an absence of information about onset of allergy and ASD diagnosis.
“Large prospective cohort studies starting from birth or early life are needed to confirm our findings,” the authors concluded.
No conflicts of interest were reported.
SOURCE: Guifeng X et al. JAMA Network Open. 2018 Jun 8. doi: 10.1001/jamanetworkopen.2018.0279.
The results of this study add to a “growing body of literature supporting an immune-mediated subtype of autism spectrum disorder (ASD),” Christopher J. McDougle, MD, wrote in an editorial published with the study.
Although prior studies have identified an association between ASD and respiratory and skin allergy, this study is “the first to document the association of food allergy with ASD with confidence, in part based on the large sample size they accessed,” he said (JAMA Network Open. 2018;1[2]:e180280. doi: 10.1001/jamanetworkopen.2018.0280).
Dr. McDougle is affiliated with the Lurie Center for Autism at Massachusetts General Hospital.
The results of this study add to a “growing body of literature supporting an immune-mediated subtype of autism spectrum disorder (ASD),” Christopher J. McDougle, MD, wrote in an editorial published with the study.
Although prior studies have identified an association between ASD and respiratory and skin allergy, this study is “the first to document the association of food allergy with ASD with confidence, in part based on the large sample size they accessed,” he said (JAMA Network Open. 2018;1[2]:e180280. doi: 10.1001/jamanetworkopen.2018.0280).
Dr. McDougle is affiliated with the Lurie Center for Autism at Massachusetts General Hospital.
The results of this study add to a “growing body of literature supporting an immune-mediated subtype of autism spectrum disorder (ASD),” Christopher J. McDougle, MD, wrote in an editorial published with the study.
Although prior studies have identified an association between ASD and respiratory and skin allergy, this study is “the first to document the association of food allergy with ASD with confidence, in part based on the large sample size they accessed,” he said (JAMA Network Open. 2018;1[2]:e180280. doi: 10.1001/jamanetworkopen.2018.0280).
Dr. McDougle is affiliated with the Lurie Center for Autism at Massachusetts General Hospital.
The prevalence of food, respiratory, and skin allergy is greater in U.S. children with autism spectrum disorder (ASD) than in U.S. children without the disorder, according to findings published June 8 in JAMA Network Open.
An analysis of data from the National Health Interview Survey found that the weighted prevalence of food, respiratory, and skin allergies was 11.25%, 18.73%, and 16.81%, respectively, in children with ASD, compared with 4.25%, 12.08%, and 9.84%, respectively, in children without ASD (P less than .001).
Survey data were collected between 1997 and 2016, and included patients aged 3-17 years. Allergic conditions were defined by the respondent, usually a parent, answering in the affirmative that the child had any kind of food, digestive, respiratory, or skin allergy in the past 12 months. ASD was defined based on an affirmative response to a question asking whether the child received an ASD diagnosis from a health professional. The question was asked as part of a 10-condition checklist from 1997 to 2013, and as a standalone item from 2014 onward, with revised wording to distinguish autism, Asperger’s disorder, pervasive developmental disorder, and ASD, wrote Guifeng Xu, MD, of the department of epidemiology at the University of Iowa, Iowa City, and her coauthors.
Of the 199,520 children included in the study, 8,734 had food allergy, 24,555 had respiratory allergy, and 19,399 had skin allergy. An ASD diagnosis was reported in 1,868 children. The weighted prevalence was 4.31% for food allergy (95% confidence interval, 4.20%-4.43%), 12.15% for respiratory allergy (95% CI, 11.92%-12.38%), and 9.91% for skin allergy (95% CI, 9.72%-10.10%), the authors said.
Children with ASD were more likely than were children without ASD to have food allergy, respiratory allergy, and skin allergy (P less than .001). After adjustment for factors including age, sex, ethnicity, and family education level, the odds ratio of ASD was more than double among children with food allergy, compared with those without food allergy (odds ratio, 2.72; 95% CI, 2.26-3.28; P less than .001).
Respiratory allergy and skin allergy also were significantly associated with ASD, but to a lesser degree, with an OR of 1.53 (95% CI, 1.32-1.78; P less than .001) for respiratory allergy and 1.80 (95% CI, 1.55-2.09; P less than .001) for skin allergy, Dr. Xu and her colleagues reported.
,” though the underlying mechanisms still need to be identified, the authors wrote.
Particularly striking were the connections found between ASD and food allergy. “Although the underlying mechanisms for the observed association between food allergy and ASD remain to be elucidated, the gut-brain-behavior axis could be one of the potential mechanisms,” Dr. Xu and her coauthors wrote. “Previous studies found higher prevalence of gastrointestinal symptoms among children with ASD.”
Limitations to the study include possible recall bias and misreporting, as well as an absence of information about onset of allergy and ASD diagnosis.
“Large prospective cohort studies starting from birth or early life are needed to confirm our findings,” the authors concluded.
No conflicts of interest were reported.
SOURCE: Guifeng X et al. JAMA Network Open. 2018 Jun 8. doi: 10.1001/jamanetworkopen.2018.0279.
The prevalence of food, respiratory, and skin allergy is greater in U.S. children with autism spectrum disorder (ASD) than in U.S. children without the disorder, according to findings published June 8 in JAMA Network Open.
An analysis of data from the National Health Interview Survey found that the weighted prevalence of food, respiratory, and skin allergies was 11.25%, 18.73%, and 16.81%, respectively, in children with ASD, compared with 4.25%, 12.08%, and 9.84%, respectively, in children without ASD (P less than .001).
Survey data were collected between 1997 and 2016, and included patients aged 3-17 years. Allergic conditions were defined by the respondent, usually a parent, answering in the affirmative that the child had any kind of food, digestive, respiratory, or skin allergy in the past 12 months. ASD was defined based on an affirmative response to a question asking whether the child received an ASD diagnosis from a health professional. The question was asked as part of a 10-condition checklist from 1997 to 2013, and as a standalone item from 2014 onward, with revised wording to distinguish autism, Asperger’s disorder, pervasive developmental disorder, and ASD, wrote Guifeng Xu, MD, of the department of epidemiology at the University of Iowa, Iowa City, and her coauthors.
Of the 199,520 children included in the study, 8,734 had food allergy, 24,555 had respiratory allergy, and 19,399 had skin allergy. An ASD diagnosis was reported in 1,868 children. The weighted prevalence was 4.31% for food allergy (95% confidence interval, 4.20%-4.43%), 12.15% for respiratory allergy (95% CI, 11.92%-12.38%), and 9.91% for skin allergy (95% CI, 9.72%-10.10%), the authors said.
Children with ASD were more likely than were children without ASD to have food allergy, respiratory allergy, and skin allergy (P less than .001). After adjustment for factors including age, sex, ethnicity, and family education level, the odds ratio of ASD was more than double among children with food allergy, compared with those without food allergy (odds ratio, 2.72; 95% CI, 2.26-3.28; P less than .001).
Respiratory allergy and skin allergy also were significantly associated with ASD, but to a lesser degree, with an OR of 1.53 (95% CI, 1.32-1.78; P less than .001) for respiratory allergy and 1.80 (95% CI, 1.55-2.09; P less than .001) for skin allergy, Dr. Xu and her colleagues reported.
,” though the underlying mechanisms still need to be identified, the authors wrote.
Particularly striking were the connections found between ASD and food allergy. “Although the underlying mechanisms for the observed association between food allergy and ASD remain to be elucidated, the gut-brain-behavior axis could be one of the potential mechanisms,” Dr. Xu and her coauthors wrote. “Previous studies found higher prevalence of gastrointestinal symptoms among children with ASD.”
Limitations to the study include possible recall bias and misreporting, as well as an absence of information about onset of allergy and ASD diagnosis.
“Large prospective cohort studies starting from birth or early life are needed to confirm our findings,” the authors concluded.
No conflicts of interest were reported.
SOURCE: Guifeng X et al. JAMA Network Open. 2018 Jun 8. doi: 10.1001/jamanetworkopen.2018.0279.
FROM JAMA NETWORK OPEN
Key clinical point: The prevalence of food, respiratory, and skin allergy was greater in children with autism spectrum disorder (ASD).
Major finding: The weighted prevalence of food, respiratory, and skin allergies was 11.25%, 18.73%, and 16.81%, respectively, in children with ASD, compared with 4.25%, 12.08%, and 9.84%, respectively, in children without ASD (P less than .001).
Study details: A population-based study of 199,520 children aged 3-17 years in the National Health Interview Survey.
Disclosures: No conflicts of interest were reported.
Source: Guifeng X et al. JAMA Network Open. 2018 Jun 8. doi: 10.1001/jamanetworkopen.2018.0279.
Pemphigus remission rate tops 80% with rituximab
ORLANDO – , and could be approved for the indication soon.
With approval pending, “rituximab is quickly emerging as frontline therapy” for pemphigus, so “we should begin to prepare to answer our patients’ questions. It’s likely they will be interested in its use,” said Carolyn Kushner, a medical student and dermatology research fellow at the University of Pennsylvania, Philadelphia. Rituximab manufacturer Genentech announced the priority review for this indication in a Feb. 2018 press release.
“We get a lot of questions in the clinic,” she said at the International Investigative Dermatology meeting. Patients with pemphigus want to know how well rituximab will work, and if they’ll be able to go off other medications. They wonder if it’s safe, and when they’ll need to be retreated. The goal of the study was to provide information for both clinicians and patients regarding what to expect from the treatment.
Overall, 54 patients (48%) achieved a complete response off therapy (CROT) after their first treatment cycle, meaning they had no new lesions for at least 2 months off of all systemic and topical treatments. The median time to a complete response was 7.4 months, and the median time to relapse was 20.9 months after the first infusion. An additional 15 patients (13%) had a complete remission with minimal therapy after one cycle.
In short, “61% of patients achieved complete healing of their skin after one cycle,” Ms. Kushner said. The number rose to 82% (93 patients) when those who had more than one cycle were included. The maximum in the study was seven. Among all patients, the median time from the first to second rituximab dose was 25.1 months.
When age, sex, and disease duration were controlled for, patients who received lymphoma dosing – 375 mg/m2 weekly for 4 weeks – were 2.7 times more likely to achieve CROT than those on the rheumatoid arthritis dosing, two 1,000 mg IV infusions 2 weeks apart (P = .037). “We almost never use RA dosing now,” she said.
The odds of success also increased with age, with patients 45 years and older 3.5 to almost 7 times more likely to achieve CROT than younger patients, also a statistically significant finding.
There were four serious adverse events across 155 cycles of the lymphoma regimen, and one with 90 cycles of arthritis dosing, all infectious and none fatal. Ms. Kushner cautioned the true rate was probably higher, since their review data might have missed some cases.
Race, sex, and disease duration had no significant effect on response rates. About 60% of the patients were women.
Rituximab, approved in 1997, is a CD20-directed cytolytic antibody.
There was no industry funding for the work, and Ms. Kushner didn’t have any disclosures.
SOURCE: Kushner CJ et al. IID 2018, Abstract 552.
ORLANDO – , and could be approved for the indication soon.
With approval pending, “rituximab is quickly emerging as frontline therapy” for pemphigus, so “we should begin to prepare to answer our patients’ questions. It’s likely they will be interested in its use,” said Carolyn Kushner, a medical student and dermatology research fellow at the University of Pennsylvania, Philadelphia. Rituximab manufacturer Genentech announced the priority review for this indication in a Feb. 2018 press release.
“We get a lot of questions in the clinic,” she said at the International Investigative Dermatology meeting. Patients with pemphigus want to know how well rituximab will work, and if they’ll be able to go off other medications. They wonder if it’s safe, and when they’ll need to be retreated. The goal of the study was to provide information for both clinicians and patients regarding what to expect from the treatment.
Overall, 54 patients (48%) achieved a complete response off therapy (CROT) after their first treatment cycle, meaning they had no new lesions for at least 2 months off of all systemic and topical treatments. The median time to a complete response was 7.4 months, and the median time to relapse was 20.9 months after the first infusion. An additional 15 patients (13%) had a complete remission with minimal therapy after one cycle.
In short, “61% of patients achieved complete healing of their skin after one cycle,” Ms. Kushner said. The number rose to 82% (93 patients) when those who had more than one cycle were included. The maximum in the study was seven. Among all patients, the median time from the first to second rituximab dose was 25.1 months.
When age, sex, and disease duration were controlled for, patients who received lymphoma dosing – 375 mg/m2 weekly for 4 weeks – were 2.7 times more likely to achieve CROT than those on the rheumatoid arthritis dosing, two 1,000 mg IV infusions 2 weeks apart (P = .037). “We almost never use RA dosing now,” she said.
The odds of success also increased with age, with patients 45 years and older 3.5 to almost 7 times more likely to achieve CROT than younger patients, also a statistically significant finding.
There were four serious adverse events across 155 cycles of the lymphoma regimen, and one with 90 cycles of arthritis dosing, all infectious and none fatal. Ms. Kushner cautioned the true rate was probably higher, since their review data might have missed some cases.
Race, sex, and disease duration had no significant effect on response rates. About 60% of the patients were women.
Rituximab, approved in 1997, is a CD20-directed cytolytic antibody.
There was no industry funding for the work, and Ms. Kushner didn’t have any disclosures.
SOURCE: Kushner CJ et al. IID 2018, Abstract 552.
ORLANDO – , and could be approved for the indication soon.
With approval pending, “rituximab is quickly emerging as frontline therapy” for pemphigus, so “we should begin to prepare to answer our patients’ questions. It’s likely they will be interested in its use,” said Carolyn Kushner, a medical student and dermatology research fellow at the University of Pennsylvania, Philadelphia. Rituximab manufacturer Genentech announced the priority review for this indication in a Feb. 2018 press release.
“We get a lot of questions in the clinic,” she said at the International Investigative Dermatology meeting. Patients with pemphigus want to know how well rituximab will work, and if they’ll be able to go off other medications. They wonder if it’s safe, and when they’ll need to be retreated. The goal of the study was to provide information for both clinicians and patients regarding what to expect from the treatment.
Overall, 54 patients (48%) achieved a complete response off therapy (CROT) after their first treatment cycle, meaning they had no new lesions for at least 2 months off of all systemic and topical treatments. The median time to a complete response was 7.4 months, and the median time to relapse was 20.9 months after the first infusion. An additional 15 patients (13%) had a complete remission with minimal therapy after one cycle.
In short, “61% of patients achieved complete healing of their skin after one cycle,” Ms. Kushner said. The number rose to 82% (93 patients) when those who had more than one cycle were included. The maximum in the study was seven. Among all patients, the median time from the first to second rituximab dose was 25.1 months.
When age, sex, and disease duration were controlled for, patients who received lymphoma dosing – 375 mg/m2 weekly for 4 weeks – were 2.7 times more likely to achieve CROT than those on the rheumatoid arthritis dosing, two 1,000 mg IV infusions 2 weeks apart (P = .037). “We almost never use RA dosing now,” she said.
The odds of success also increased with age, with patients 45 years and older 3.5 to almost 7 times more likely to achieve CROT than younger patients, also a statistically significant finding.
There were four serious adverse events across 155 cycles of the lymphoma regimen, and one with 90 cycles of arthritis dosing, all infectious and none fatal. Ms. Kushner cautioned the true rate was probably higher, since their review data might have missed some cases.
Race, sex, and disease duration had no significant effect on response rates. About 60% of the patients were women.
Rituximab, approved in 1997, is a CD20-directed cytolytic antibody.
There was no industry funding for the work, and Ms. Kushner didn’t have any disclosures.
SOURCE: Kushner CJ et al. IID 2018, Abstract 552.
REPORTING FROM IID 2018
Key clinical point: Rituximab puts the majority of pemphigus patients in remission, with a median time between doses of about 2 years.
Major finding: Sixty-one percent of patients achieved complete healing of their skin after one cycle, increasing to 82% when those who had more than one cycle were included.
Study details: A single-center review of 113 patients
Disclosures: There was no industry funding, and the lead investigator had no disclosures.
Source: Kushner CJ et al. IID 2018, Abstract 552.
Tiny Tot, Big Lesion
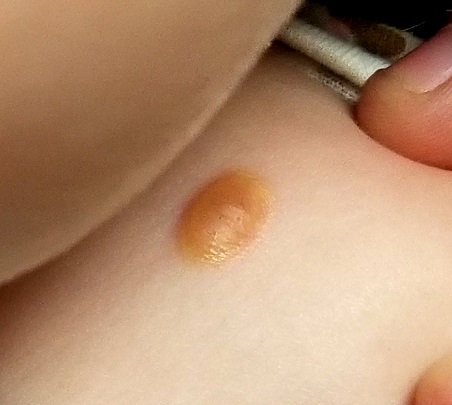
About six months ago, the parents of this 1-year-old boy first noticed the lesion on his shoulder. It started as a pinpoint papule but has grown to its current size—at which point, it caught their full attention. Although there are no associated symptoms, the parents request referral to dermatology to clear up the matter.
The child is reportedly healthy in all other respects, maintaining weight as expected, and normally active and reactive to verbal and visual stimuli.
EXAMINATION
A distinctive orangish brown, ovoid, 8 x 4–mm nodule is located on the child’s right superior shoulder. The lesion has a smooth, soft surface, and there is no tenderness on palpation. No additional lesions are seen elsewhere.
Eye examination reveals normal and symmetrical red reflexes.
What is the diagnosis?
DISCUSSION
Juvenile xanthogranuloma (JXG) is a rare, benign variant of non-Langerhans cell histiocytosis. This patient’s lesion is typical, but JXG can vary in appearance; some patients present with darker or larger lesions—or multiple lesions.
JXGs are essentially granulomatous tumors that, on histologic examination, display multinucleated giant cells called Touton giant cells. These macrophage-derived foam cells are seen in lesions with high lipid content.
JXG tends to favor the neck, face, and trunk but can appear around or (rarely) inside the eye, typically unilaterally in the iris. Benign in all other respects, ocular JXG lesions can cause spontaneous hyphema, glaucoma, or blindness; they must therefore be dealt with by a specialist. Fortunately, only about 10% of patients display ocular involvement.
JXGs can be confused with compound nevi, warts, or Spitz tumors. Therefore, biopsy is often necessary to establish the diagnosis.
TAKE-HOME LEARNING POINTS
- Juvenile xanthogranuloma (JXG) is a rare non-Langerhans cell tumor usually seen on the neck, face, or trunk of children younger than 2.
- The orangish brown, soft appearance of this patient’s papule was typical.
- Although atypical JXG lesions may require shave biopsy to confirm the diagnosis, they typically resolve on their own without treatment.
- When JXG lesions appear in the eye (most commonly in the iris), there is potential for serious complications, including heterochromia, glaucoma, spontaneous hyphema, or even blindness.
About six months ago, the parents of this 1-year-old boy first noticed the lesion on his shoulder. It started as a pinpoint papule but has grown to its current size—at which point, it caught their full attention. Although there are no associated symptoms, the parents request referral to dermatology to clear up the matter.
The child is reportedly healthy in all other respects, maintaining weight as expected, and normally active and reactive to verbal and visual stimuli.
EXAMINATION
A distinctive orangish brown, ovoid, 8 x 4–mm nodule is located on the child’s right superior shoulder. The lesion has a smooth, soft surface, and there is no tenderness on palpation. No additional lesions are seen elsewhere.

Eye examination reveals normal and symmetrical red reflexes.
What is the diagnosis?
DISCUSSION
Juvenile xanthogranuloma (JXG) is a rare, benign variant of non-Langerhans cell histiocytosis. This patient’s lesion is typical, but JXG can vary in appearance; some patients present with darker or larger lesions—or multiple lesions.
JXGs are essentially granulomatous tumors that, on histologic examination, display multinucleated giant cells called Touton giant cells. These macrophage-derived foam cells are seen in lesions with high lipid content.
JXG tends to favor the neck, face, and trunk but can appear around or (rarely) inside the eye, typically unilaterally in the iris. Benign in all other respects, ocular JXG lesions can cause spontaneous hyphema, glaucoma, or blindness; they must therefore be dealt with by a specialist. Fortunately, only about 10% of patients display ocular involvement.
JXGs can be confused with compound nevi, warts, or Spitz tumors. Therefore, biopsy is often necessary to establish the diagnosis.
TAKE-HOME LEARNING POINTS
- Juvenile xanthogranuloma (JXG) is a rare non-Langerhans cell tumor usually seen on the neck, face, or trunk of children younger than 2.
- The orangish brown, soft appearance of this patient’s papule was typical.
- Although atypical JXG lesions may require shave biopsy to confirm the diagnosis, they typically resolve on their own without treatment.
- When JXG lesions appear in the eye (most commonly in the iris), there is potential for serious complications, including heterochromia, glaucoma, spontaneous hyphema, or even blindness.
About six months ago, the parents of this 1-year-old boy first noticed the lesion on his shoulder. It started as a pinpoint papule but has grown to its current size—at which point, it caught their full attention. Although there are no associated symptoms, the parents request referral to dermatology to clear up the matter.
The child is reportedly healthy in all other respects, maintaining weight as expected, and normally active and reactive to verbal and visual stimuli.
EXAMINATION
A distinctive orangish brown, ovoid, 8 x 4–mm nodule is located on the child’s right superior shoulder. The lesion has a smooth, soft surface, and there is no tenderness on palpation. No additional lesions are seen elsewhere.

Eye examination reveals normal and symmetrical red reflexes.
What is the diagnosis?
DISCUSSION
Juvenile xanthogranuloma (JXG) is a rare, benign variant of non-Langerhans cell histiocytosis. This patient’s lesion is typical, but JXG can vary in appearance; some patients present with darker or larger lesions—or multiple lesions.
JXGs are essentially granulomatous tumors that, on histologic examination, display multinucleated giant cells called Touton giant cells. These macrophage-derived foam cells are seen in lesions with high lipid content.
JXG tends to favor the neck, face, and trunk but can appear around or (rarely) inside the eye, typically unilaterally in the iris. Benign in all other respects, ocular JXG lesions can cause spontaneous hyphema, glaucoma, or blindness; they must therefore be dealt with by a specialist. Fortunately, only about 10% of patients display ocular involvement.
JXGs can be confused with compound nevi, warts, or Spitz tumors. Therefore, biopsy is often necessary to establish the diagnosis.
TAKE-HOME LEARNING POINTS
- Juvenile xanthogranuloma (JXG) is a rare non-Langerhans cell tumor usually seen on the neck, face, or trunk of children younger than 2.
- The orangish brown, soft appearance of this patient’s papule was typical.
- Although atypical JXG lesions may require shave biopsy to confirm the diagnosis, they typically resolve on their own without treatment.
- When JXG lesions appear in the eye (most commonly in the iris), there is potential for serious complications, including heterochromia, glaucoma, spontaneous hyphema, or even blindness.
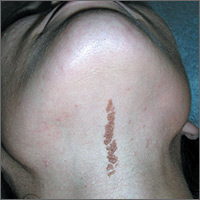
Growth on neck
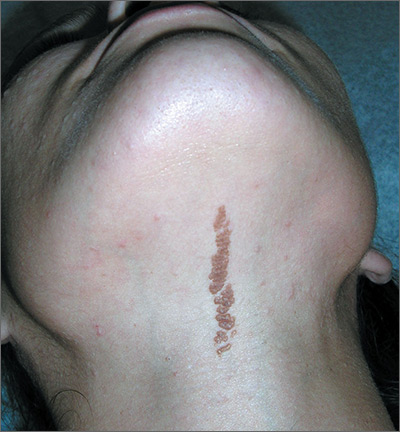
The FP recognized the lesion as a linear epidermal nevus.
Epidermal nevi (EN) are congenital hamartomas of ectodermal origin that are uncommon (occurring in < 1% of newborns and children), sporadic, and usually present at birth, although they can appear in early childhood. EN are associated with disorders of the eye, nervous system, and musculoskeletal system in 10% to 30% of patients.
EN are linear, round or oblong, well circumscribed, elevated, and flat topped. EN are often yellow-tan to dark brown in color, with a surface that is uniformly velvety or warty. They most commonly occur on the head and neck, although they can occur on the trunk and proximal extremities.
The FP determined that the patient had no neurological, musculoskeletal, or vision problems that could be associated with a linear epidermal nevus syndrome and reassured the patient and his mother that the nevus was not dangerous and did not need to be removed.
Photos and text for Photo Rounds Friday courtesy of Richard P. Usatine, MD. This case was adapted from: Smith M. Epidermal nevus and nevus sebaceous. In: Usatine R, Smith M, Mayeaux EJ, et al. Color Atlas of Family Medicine, 2nd ed. New York, NY: McGraw-Hill; 2013:958-962.
To learn more about the Color Atlas of Family Medicine, see: www.amazon.com/Color-Family-Medicine-Richard-Usatine/dp/0071769641/.
You can now get the second edition of the Color Atlas of Family Medicine as an app by clicking on this link: usatinemedia.com.

The FP recognized the lesion as a linear epidermal nevus.
Epidermal nevi (EN) are congenital hamartomas of ectodermal origin that are uncommon (occurring in < 1% of newborns and children), sporadic, and usually present at birth, although they can appear in early childhood. EN are associated with disorders of the eye, nervous system, and musculoskeletal system in 10% to 30% of patients.
EN are linear, round or oblong, well circumscribed, elevated, and flat topped. EN are often yellow-tan to dark brown in color, with a surface that is uniformly velvety or warty. They most commonly occur on the head and neck, although they can occur on the trunk and proximal extremities.
The FP determined that the patient had no neurological, musculoskeletal, or vision problems that could be associated with a linear epidermal nevus syndrome and reassured the patient and his mother that the nevus was not dangerous and did not need to be removed.
Photos and text for Photo Rounds Friday courtesy of Richard P. Usatine, MD. This case was adapted from: Smith M. Epidermal nevus and nevus sebaceous. In: Usatine R, Smith M, Mayeaux EJ, et al. Color Atlas of Family Medicine, 2nd ed. New York, NY: McGraw-Hill; 2013:958-962.
To learn more about the Color Atlas of Family Medicine, see: www.amazon.com/Color-Family-Medicine-Richard-Usatine/dp/0071769641/.
You can now get the second edition of the Color Atlas of Family Medicine as an app by clicking on this link: usatinemedia.com.

The FP recognized the lesion as a linear epidermal nevus.
Epidermal nevi (EN) are congenital hamartomas of ectodermal origin that are uncommon (occurring in < 1% of newborns and children), sporadic, and usually present at birth, although they can appear in early childhood. EN are associated with disorders of the eye, nervous system, and musculoskeletal system in 10% to 30% of patients.
EN are linear, round or oblong, well circumscribed, elevated, and flat topped. EN are often yellow-tan to dark brown in color, with a surface that is uniformly velvety or warty. They most commonly occur on the head and neck, although they can occur on the trunk and proximal extremities.
The FP determined that the patient had no neurological, musculoskeletal, or vision problems that could be associated with a linear epidermal nevus syndrome and reassured the patient and his mother that the nevus was not dangerous and did not need to be removed.
Photos and text for Photo Rounds Friday courtesy of Richard P. Usatine, MD. This case was adapted from: Smith M. Epidermal nevus and nevus sebaceous. In: Usatine R, Smith M, Mayeaux EJ, et al. Color Atlas of Family Medicine, 2nd ed. New York, NY: McGraw-Hill; 2013:958-962.
To learn more about the Color Atlas of Family Medicine, see: www.amazon.com/Color-Family-Medicine-Richard-Usatine/dp/0071769641/.
You can now get the second edition of the Color Atlas of Family Medicine as an app by clicking on this link: usatinemedia.com.
Cutaneous lupus: Switching antimalarials can delay immunosuppressive therapy
ORLANDO – , according to Anthony Fernandez, MD, PhD, director of medical and inpatient dermatology at the Cleveland Clinic.
A switch to chloroquine, or adding quinacrine, might do the trick, saving at least some patients from having to move on to immunosuppressive therapy, Dr. Fernandez said at the International Conference on Cutaneous Lupus Erythematosus.
“What we are learning from the literature is that we can switch from one antimalarial to another. We need to think about this in our algorithms before reaching for potentially more toxic immunosuppressives,” Dr. Fernandez said.
As for quinacrine, about two-thirds of patients who fail hydroxychloroquine or chloroquine will have a positive response to quinacrine if added (Br J Dermatol. 2017 Jul;177[1]:188-96). “It’s important to remember that we are not adding any ocular toxicity” with quinacrine, he said.
Quinacrine does come with a major concern of its own: the risk of aplastic anemia. However, this seems to occur with doses higher than 100 mg/day, which are no longer recommended; there have been no reports of aplastic anemia in patients on 100 mg/day or less.
Quinacrine “is an underutilized antimalarial. I think a lot of people don’t know about it or know how to get it. It can be compounded into capsules for patients,” and for a reasonable price, at about $20 for a month supply at some pharmacies, Dr. Fernandez said.
Meanwhile, because of the risk of retinal toxicity with hydroxychloroquine, there’s been a shift in recent years from dosing up to 6.5 mg/kg per day of ideal body weight to a ceiling of 5 mg/kg per day of actual body weight (JAMA Ophthalmol. 2014 Dec;132[12]:1453-60), and a ceiling of 2.3 mg/kg per day actual body weight for chloroquine.
The idea was to prevent overdosing in people who are under their ideal body weight, but there have been concerns about the efficacy of the new dosing regimen in other patients. Dr. Fernandez has not seen evidence of this. “We do adhere to the new dosing recommendations” at the Cleveland Clinic, and “personally, I think we are seeing similar efficacy,” he said.
The most important risk factor for retinal toxicity is cumulative dose. The risk seems to be extremely low in the first 5 years, but increases afterwards. Most patients who develop retinal toxicity have taken a cumulative hydroxychloroquine dose of 1,000 g, equal to about 400 mg/d for 7 years. “The longer you are on the medicine, the higher your risk of developing retinal toxicity,” he noted.
Regardless of weight, it’s recommended to limit hydroxychloroquine to 400 mg daily and chloroquine to 250 mg daily, with a baseline ocular exam, and – barring any intervening problems – annual screening after 5 years.
In patients with highly active skin disease at baseline, Dr. Fernandez said he will sometimes start hydroxychloroquine higher than 400 mg initially but will bring patients back down to 400 mg after a few months.
Dr. Fernandez had no relevant disclosures.
ORLANDO – , according to Anthony Fernandez, MD, PhD, director of medical and inpatient dermatology at the Cleveland Clinic.
A switch to chloroquine, or adding quinacrine, might do the trick, saving at least some patients from having to move on to immunosuppressive therapy, Dr. Fernandez said at the International Conference on Cutaneous Lupus Erythematosus.
“What we are learning from the literature is that we can switch from one antimalarial to another. We need to think about this in our algorithms before reaching for potentially more toxic immunosuppressives,” Dr. Fernandez said.
As for quinacrine, about two-thirds of patients who fail hydroxychloroquine or chloroquine will have a positive response to quinacrine if added (Br J Dermatol. 2017 Jul;177[1]:188-96). “It’s important to remember that we are not adding any ocular toxicity” with quinacrine, he said.
Quinacrine does come with a major concern of its own: the risk of aplastic anemia. However, this seems to occur with doses higher than 100 mg/day, which are no longer recommended; there have been no reports of aplastic anemia in patients on 100 mg/day or less.
Quinacrine “is an underutilized antimalarial. I think a lot of people don’t know about it or know how to get it. It can be compounded into capsules for patients,” and for a reasonable price, at about $20 for a month supply at some pharmacies, Dr. Fernandez said.
Meanwhile, because of the risk of retinal toxicity with hydroxychloroquine, there’s been a shift in recent years from dosing up to 6.5 mg/kg per day of ideal body weight to a ceiling of 5 mg/kg per day of actual body weight (JAMA Ophthalmol. 2014 Dec;132[12]:1453-60), and a ceiling of 2.3 mg/kg per day actual body weight for chloroquine.
The idea was to prevent overdosing in people who are under their ideal body weight, but there have been concerns about the efficacy of the new dosing regimen in other patients. Dr. Fernandez has not seen evidence of this. “We do adhere to the new dosing recommendations” at the Cleveland Clinic, and “personally, I think we are seeing similar efficacy,” he said.
The most important risk factor for retinal toxicity is cumulative dose. The risk seems to be extremely low in the first 5 years, but increases afterwards. Most patients who develop retinal toxicity have taken a cumulative hydroxychloroquine dose of 1,000 g, equal to about 400 mg/d for 7 years. “The longer you are on the medicine, the higher your risk of developing retinal toxicity,” he noted.
Regardless of weight, it’s recommended to limit hydroxychloroquine to 400 mg daily and chloroquine to 250 mg daily, with a baseline ocular exam, and – barring any intervening problems – annual screening after 5 years.
In patients with highly active skin disease at baseline, Dr. Fernandez said he will sometimes start hydroxychloroquine higher than 400 mg initially but will bring patients back down to 400 mg after a few months.
Dr. Fernandez had no relevant disclosures.
ORLANDO – , according to Anthony Fernandez, MD, PhD, director of medical and inpatient dermatology at the Cleveland Clinic.
A switch to chloroquine, or adding quinacrine, might do the trick, saving at least some patients from having to move on to immunosuppressive therapy, Dr. Fernandez said at the International Conference on Cutaneous Lupus Erythematosus.
“What we are learning from the literature is that we can switch from one antimalarial to another. We need to think about this in our algorithms before reaching for potentially more toxic immunosuppressives,” Dr. Fernandez said.
As for quinacrine, about two-thirds of patients who fail hydroxychloroquine or chloroquine will have a positive response to quinacrine if added (Br J Dermatol. 2017 Jul;177[1]:188-96). “It’s important to remember that we are not adding any ocular toxicity” with quinacrine, he said.
Quinacrine does come with a major concern of its own: the risk of aplastic anemia. However, this seems to occur with doses higher than 100 mg/day, which are no longer recommended; there have been no reports of aplastic anemia in patients on 100 mg/day or less.
Quinacrine “is an underutilized antimalarial. I think a lot of people don’t know about it or know how to get it. It can be compounded into capsules for patients,” and for a reasonable price, at about $20 for a month supply at some pharmacies, Dr. Fernandez said.
Meanwhile, because of the risk of retinal toxicity with hydroxychloroquine, there’s been a shift in recent years from dosing up to 6.5 mg/kg per day of ideal body weight to a ceiling of 5 mg/kg per day of actual body weight (JAMA Ophthalmol. 2014 Dec;132[12]:1453-60), and a ceiling of 2.3 mg/kg per day actual body weight for chloroquine.
The idea was to prevent overdosing in people who are under their ideal body weight, but there have been concerns about the efficacy of the new dosing regimen in other patients. Dr. Fernandez has not seen evidence of this. “We do adhere to the new dosing recommendations” at the Cleveland Clinic, and “personally, I think we are seeing similar efficacy,” he said.
The most important risk factor for retinal toxicity is cumulative dose. The risk seems to be extremely low in the first 5 years, but increases afterwards. Most patients who develop retinal toxicity have taken a cumulative hydroxychloroquine dose of 1,000 g, equal to about 400 mg/d for 7 years. “The longer you are on the medicine, the higher your risk of developing retinal toxicity,” he noted.
Regardless of weight, it’s recommended to limit hydroxychloroquine to 400 mg daily and chloroquine to 250 mg daily, with a baseline ocular exam, and – barring any intervening problems – annual screening after 5 years.
In patients with highly active skin disease at baseline, Dr. Fernandez said he will sometimes start hydroxychloroquine higher than 400 mg initially but will bring patients back down to 400 mg after a few months.
Dr. Fernandez had no relevant disclosures.
EXPERT ANALYSIS FROM ICCLE 2018
Hydroxychloroquine throws off Quantiferon-TB Gold results, study finds
ORLANDO – according to investigators from the University of Pennsylvania, Philadelphia.
Among 46 patients with lupus, dermatomyositis, or blistering diseases who had been on hydroxychloroquine within a year of testing, QuantiFERON-TB Gold (QFT-G) – the go-to TB test in many places – yielded indeterminate results in 37%. Meanwhile, just 9.6% of tests were indeterminate among 73 patients with those diseases who had not been on hydroxychloroquine (P less than .001). The findings could not be explained by concomitant use of prednisone and other immunosuppressives; there were no statistically significant differences between the groups. “This was shocking to us. We need to come up with a better screening test in this patient population,” said lead investigator Rebecca Gaffney, a research fellow at the University of Pennsylvania, and a medical student at Robert Wood Johnson Medical School, New Brunswick, NJ.*
It’s widely known that immunosuppressives interfere with QFT-G results, but antimalarials are considered immunomodulators, not immunosuppressives. The new study is probably the first to investigate the issue. The team is now pitting QFT-G against another TB blood test, the T-SPOT, in 100 patients to see if it’s a better option, in a trial that they expect to complete in 2018.
The investigators have a hunch that the T-SPOT might be better because, while QFT-G measures interferon-gamma concentrations in response to TB antigens, the T-SPOT “counts cells first to make sure you have a standard amount of cells, then looks at how many cells are releasing interferon-gamma,” Ms. Gaffney said, adding that “it seems like a more sensitive test,” especially for lymphocytopenic autoimmune patients. “We are really excited to see if there’s a better test for our patients, given all the clinical trials we do. We want to see what’s best, so there’s no barrier to receiving therapy.”
Subjects were around 50 years old on average, and the majority were women. Most were white, and about 20% were black.
There was no industry funding for the work, and Ms. Gaffney reported no disclosures.
*This article was updated on June 13. 2018.
ORLANDO – according to investigators from the University of Pennsylvania, Philadelphia.
Among 46 patients with lupus, dermatomyositis, or blistering diseases who had been on hydroxychloroquine within a year of testing, QuantiFERON-TB Gold (QFT-G) – the go-to TB test in many places – yielded indeterminate results in 37%. Meanwhile, just 9.6% of tests were indeterminate among 73 patients with those diseases who had not been on hydroxychloroquine (P less than .001). The findings could not be explained by concomitant use of prednisone and other immunosuppressives; there were no statistically significant differences between the groups. “This was shocking to us. We need to come up with a better screening test in this patient population,” said lead investigator Rebecca Gaffney, a research fellow at the University of Pennsylvania, and a medical student at Robert Wood Johnson Medical School, New Brunswick, NJ.*
It’s widely known that immunosuppressives interfere with QFT-G results, but antimalarials are considered immunomodulators, not immunosuppressives. The new study is probably the first to investigate the issue. The team is now pitting QFT-G against another TB blood test, the T-SPOT, in 100 patients to see if it’s a better option, in a trial that they expect to complete in 2018.
The investigators have a hunch that the T-SPOT might be better because, while QFT-G measures interferon-gamma concentrations in response to TB antigens, the T-SPOT “counts cells first to make sure you have a standard amount of cells, then looks at how many cells are releasing interferon-gamma,” Ms. Gaffney said, adding that “it seems like a more sensitive test,” especially for lymphocytopenic autoimmune patients. “We are really excited to see if there’s a better test for our patients, given all the clinical trials we do. We want to see what’s best, so there’s no barrier to receiving therapy.”
Subjects were around 50 years old on average, and the majority were women. Most were white, and about 20% were black.
There was no industry funding for the work, and Ms. Gaffney reported no disclosures.
*This article was updated on June 13. 2018.
ORLANDO – according to investigators from the University of Pennsylvania, Philadelphia.
Among 46 patients with lupus, dermatomyositis, or blistering diseases who had been on hydroxychloroquine within a year of testing, QuantiFERON-TB Gold (QFT-G) – the go-to TB test in many places – yielded indeterminate results in 37%. Meanwhile, just 9.6% of tests were indeterminate among 73 patients with those diseases who had not been on hydroxychloroquine (P less than .001). The findings could not be explained by concomitant use of prednisone and other immunosuppressives; there were no statistically significant differences between the groups. “This was shocking to us. We need to come up with a better screening test in this patient population,” said lead investigator Rebecca Gaffney, a research fellow at the University of Pennsylvania, and a medical student at Robert Wood Johnson Medical School, New Brunswick, NJ.*
It’s widely known that immunosuppressives interfere with QFT-G results, but antimalarials are considered immunomodulators, not immunosuppressives. The new study is probably the first to investigate the issue. The team is now pitting QFT-G against another TB blood test, the T-SPOT, in 100 patients to see if it’s a better option, in a trial that they expect to complete in 2018.
The investigators have a hunch that the T-SPOT might be better because, while QFT-G measures interferon-gamma concentrations in response to TB antigens, the T-SPOT “counts cells first to make sure you have a standard amount of cells, then looks at how many cells are releasing interferon-gamma,” Ms. Gaffney said, adding that “it seems like a more sensitive test,” especially for lymphocytopenic autoimmune patients. “We are really excited to see if there’s a better test for our patients, given all the clinical trials we do. We want to see what’s best, so there’s no barrier to receiving therapy.”
Subjects were around 50 years old on average, and the majority were women. Most were white, and about 20% were black.
There was no industry funding for the work, and Ms. Gaffney reported no disclosures.
*This article was updated on June 13. 2018.
REPORTING FROM ICCLE 2018
Timely dermatomyositis diagnosis, treatment remain elusive
ORLANDO – There was a median 1-year delay between the onset of symptoms and diagnosis of classic dermatomyositis, and a 17-month delay before diagnosis of amyopathic dermatomyositis, based on a review of 232 dermatomyositis patients seen at the University of Pennsylvania, Philadelphia.
Just 103 (44.4%) patients were diagnosed with dermatomyositis (DM) right out of the gate. Among the other 129, 48 (37.2%) were diagnosed with lupus, 38 (29.5%) with undifferentiated connective tissue disease, 10 (7.8%) went undiagnosed, and 33 (25.5%) were diagnosed with rosacea, psoriasis, rheumatoid arthritis, fibromyalgia, lichen planus, and a number of other conditions. By the time the DM diagnosis was finally confirmed, almost every patient had Gottron’s papules or sign.
Misdiagnosis of dermatomyositis (DM) is nothing new, but the study brings home just how common the problem is, even at a major academic medical institution.
One of the take homes is that , and remain vigilant for erythema on the lateral thighs or nasolabial fold, Gottron’s papules, and other diagnostic giveaways, the researchers said.
Interface dermatitis, in particular, can’t be relied on to differentiate the conditions. A better option is checking for lupus bands and membrane attack complexes on direct immunofluorescence.
There’s also just not enough awareness that dermatomyositis can present without the classic muscle symptoms and findings, i.e. clinically amyopathic DM. While 49 of 120 patients with classic dermatomyositis (40.8%) were misdiagnosed or undiagnosed in the study, the number rose to 80 of 112 (71.4%) among amyopathic patients.
“We saw that there was a much higher rate of misdiagnosis in patients who didn’t have any muscle disease. We have to raise awareness that amyopathic dermatomyositis is a very prevalent condition,” Dr. Patel said at the International Conference on Cutaneous Lupus Erythematosus.
“There might be some level of subclinical muscle activity where, if you did an MRI, you might see inflammation, but the patient doesn’t report any symptoms. There are also patients that don’t have any muscle findings on MRI, or elevated muscle enzymes, but still have the skin findings,” he said.
Perhaps the markedly increased risk of cancer in DM, especially within a year or 2 of symptom onset, is the strongest argument for earlier diagnosis. “There’s also a risk of interstitial lung disease, so making sure that you’re getting pulmonary function tests and age-appropriate malignancy screening in a timely fashion is very important,” Dr. Patel said.
Also, although many of the initial treatments for DM – sun protection and topical steroids and calcineurin inhibitors, for instance – are the same as for cutaneous lupus, medications like mycophenolate mofetil and methotrexate are used more readily. The sooner DM is recognized for what it is, the sooner patients can get relief, he said.
Almost all the patients were white women. The majority were 40-80 years old.
There was no industry funding for the work, and Dr. Patel didn’t have any disclosures.
SOURCE: da Silva DM et al. Presented at the 2018 International Conference on Cutaneous Lupus Erythematosus
ORLANDO – There was a median 1-year delay between the onset of symptoms and diagnosis of classic dermatomyositis, and a 17-month delay before diagnosis of amyopathic dermatomyositis, based on a review of 232 dermatomyositis patients seen at the University of Pennsylvania, Philadelphia.
Just 103 (44.4%) patients were diagnosed with dermatomyositis (DM) right out of the gate. Among the other 129, 48 (37.2%) were diagnosed with lupus, 38 (29.5%) with undifferentiated connective tissue disease, 10 (7.8%) went undiagnosed, and 33 (25.5%) were diagnosed with rosacea, psoriasis, rheumatoid arthritis, fibromyalgia, lichen planus, and a number of other conditions. By the time the DM diagnosis was finally confirmed, almost every patient had Gottron’s papules or sign.
Misdiagnosis of dermatomyositis (DM) is nothing new, but the study brings home just how common the problem is, even at a major academic medical institution.
One of the take homes is that , and remain vigilant for erythema on the lateral thighs or nasolabial fold, Gottron’s papules, and other diagnostic giveaways, the researchers said.
Interface dermatitis, in particular, can’t be relied on to differentiate the conditions. A better option is checking for lupus bands and membrane attack complexes on direct immunofluorescence.
There’s also just not enough awareness that dermatomyositis can present without the classic muscle symptoms and findings, i.e. clinically amyopathic DM. While 49 of 120 patients with classic dermatomyositis (40.8%) were misdiagnosed or undiagnosed in the study, the number rose to 80 of 112 (71.4%) among amyopathic patients.
“We saw that there was a much higher rate of misdiagnosis in patients who didn’t have any muscle disease. We have to raise awareness that amyopathic dermatomyositis is a very prevalent condition,” Dr. Patel said at the International Conference on Cutaneous Lupus Erythematosus.
“There might be some level of subclinical muscle activity where, if you did an MRI, you might see inflammation, but the patient doesn’t report any symptoms. There are also patients that don’t have any muscle findings on MRI, or elevated muscle enzymes, but still have the skin findings,” he said.
Perhaps the markedly increased risk of cancer in DM, especially within a year or 2 of symptom onset, is the strongest argument for earlier diagnosis. “There’s also a risk of interstitial lung disease, so making sure that you’re getting pulmonary function tests and age-appropriate malignancy screening in a timely fashion is very important,” Dr. Patel said.
Also, although many of the initial treatments for DM – sun protection and topical steroids and calcineurin inhibitors, for instance – are the same as for cutaneous lupus, medications like mycophenolate mofetil and methotrexate are used more readily. The sooner DM is recognized for what it is, the sooner patients can get relief, he said.
Almost all the patients were white women. The majority were 40-80 years old.
There was no industry funding for the work, and Dr. Patel didn’t have any disclosures.
SOURCE: da Silva DM et al. Presented at the 2018 International Conference on Cutaneous Lupus Erythematosus
ORLANDO – There was a median 1-year delay between the onset of symptoms and diagnosis of classic dermatomyositis, and a 17-month delay before diagnosis of amyopathic dermatomyositis, based on a review of 232 dermatomyositis patients seen at the University of Pennsylvania, Philadelphia.
Just 103 (44.4%) patients were diagnosed with dermatomyositis (DM) right out of the gate. Among the other 129, 48 (37.2%) were diagnosed with lupus, 38 (29.5%) with undifferentiated connective tissue disease, 10 (7.8%) went undiagnosed, and 33 (25.5%) were diagnosed with rosacea, psoriasis, rheumatoid arthritis, fibromyalgia, lichen planus, and a number of other conditions. By the time the DM diagnosis was finally confirmed, almost every patient had Gottron’s papules or sign.
Misdiagnosis of dermatomyositis (DM) is nothing new, but the study brings home just how common the problem is, even at a major academic medical institution.
One of the take homes is that , and remain vigilant for erythema on the lateral thighs or nasolabial fold, Gottron’s papules, and other diagnostic giveaways, the researchers said.
Interface dermatitis, in particular, can’t be relied on to differentiate the conditions. A better option is checking for lupus bands and membrane attack complexes on direct immunofluorescence.
There’s also just not enough awareness that dermatomyositis can present without the classic muscle symptoms and findings, i.e. clinically amyopathic DM. While 49 of 120 patients with classic dermatomyositis (40.8%) were misdiagnosed or undiagnosed in the study, the number rose to 80 of 112 (71.4%) among amyopathic patients.
“We saw that there was a much higher rate of misdiagnosis in patients who didn’t have any muscle disease. We have to raise awareness that amyopathic dermatomyositis is a very prevalent condition,” Dr. Patel said at the International Conference on Cutaneous Lupus Erythematosus.
“There might be some level of subclinical muscle activity where, if you did an MRI, you might see inflammation, but the patient doesn’t report any symptoms. There are also patients that don’t have any muscle findings on MRI, or elevated muscle enzymes, but still have the skin findings,” he said.
Perhaps the markedly increased risk of cancer in DM, especially within a year or 2 of symptom onset, is the strongest argument for earlier diagnosis. “There’s also a risk of interstitial lung disease, so making sure that you’re getting pulmonary function tests and age-appropriate malignancy screening in a timely fashion is very important,” Dr. Patel said.
Also, although many of the initial treatments for DM – sun protection and topical steroids and calcineurin inhibitors, for instance – are the same as for cutaneous lupus, medications like mycophenolate mofetil and methotrexate are used more readily. The sooner DM is recognized for what it is, the sooner patients can get relief, he said.
Almost all the patients were white women. The majority were 40-80 years old.
There was no industry funding for the work, and Dr. Patel didn’t have any disclosures.
SOURCE: da Silva DM et al. Presented at the 2018 International Conference on Cutaneous Lupus Erythematosus
REPORTING FROM ICCLE 2018
Key clinical point: Clinicians need to do a better job of catching DM early.
Major finding: It took a median of 12.2 months after the start of symptoms to diagnose classic dermatomyositis, and 17.1 months to diagnose amyopathic dermatomyositis.
Study details: Review of 232 patients
Disclosures: There was no industry funding, and the presenter didn’t have any disclosures.
Source: da Silva DM et al. Presented at the 2018 International Conference on Cutaneous Lupus Erythematosus
The Evidence for Herbal and Botanical Remedies, Part 2
More than a third of American adults use complementary and alternative medicine.1 Unfortunately, the public’s enthusiasm for herbal products is not always consistent with the scientific evidence supporting their use. In part one of this series, we discussed the studies that have been done on capsaicin, butterbur, green tea, and peppermint. In this installment, we outline the research on 5 additional remedies: turmeric/curcumin, which may be of benefit in ulcerative colitis; chamomile, which appears to offer relief to patients with anxiety; rosemary, which may help treat alopecia; as well as coffee and cocoa, which may have some cardiovascular benefits (TABLE).
Turmeric/curcumin
Overview
Turmeric (Curcuma longa), a relative of ginger, has been used for 4000 years to treat a variety of conditions.2,3 Curcumin is the yellow pigment isolated from the rhizomes of Curcuma longa, commonly known as turmeric.3 Turmeric powder contains 5% curcumin, which is the main biologically active compound. Although it grows in many tropical locations, most turmeric is grown in India, where it is used as a main ingredient in curry. The roots and bulbs of turmeric that are used in medicine are generally boiled and dried, which results in a yellow powder.
Turmeric has been used in both Ayurvedic and Chinese medicine for its anti-inflammatory properties, in the treatment of digestive and liver problems, to fight infections, and to help heal skin diseases and wounds.3-7
Functional GI disorders. A recent review noted that curcumin has been shown in several preclinical studies and uncontrolled clinical trials to have effects on gut inflammation, gut permeability, and the brain-gut axis, especially in functional GI disorders.7 A double-blind, placebo-controlled study from 1989 found that turmeric reduced symptoms of bloating and gas in subjects suffering from undifferentiated dyspepsia.8
Ulcerative colitis (UC). A 2012 Cochrane review noted that curcumin appears to be a safe and effective therapy for maintenance of remission in quiescent UC when given as adjunctive therapy along with mesalamine or sulfasalazine.9 In a 2015 randomized controlled trial (RCT), the addition of curcumin to mesalamine therapy was superior to the combination of placebo and mesalamine in inducing clinical and endoscopic remission in patients with mild-to-moderate active UC, producing no apparent adverse effects.10
Osteoarthritis (OA). Because of turmeric’s ability to reduce inflammation, it may help relieve OA pain.3 Clinical evidence is scant for the anti-arthritic efficacy of turmeric dietary supplements, although animal studies indicate that turmeric prevents inflammation through regulation of NF-kappaB-regulated genes that regulate the immune and inflammatory response.6 Inflammatory cell influx, joint levels of prostaglandin E2, and periarticular osteoclast formation were also inhibited by turmeric extract treatment.6
A 2013 review of turmeric for OA concluded that observational studies and in vitro results are promising for the use of curcumin for OA, but well-designed clinical studies were lacking and are needed to support the efficacy of curcumin in OA patients.11 However, in a 2014 randomized trial of 367 patients, turmeric appeared to be similar in efficacy to ibuprofen for the treatment of pain and disability in adults with knee OA.12 The curcumin (turmeric) group also had fewer adverse effects.12
Cancer. There has been a great deal of research on turmeric’s anti-cancer properties, but clinical evidence is lacking. In vitro evidence, animal studies, and small clinical trials suggest that curcumin may help prevent or treat several types of cancers, but the overall evidence is poor. Nonetheless, curcumin and turmeric have been or are currently being evaluated for the treatment or prevention of prostate, liver, breast, skin, gynecologic, hematologic, pulmonary, thymic, bone, brain, and colon cancer.13-18
Oral submucous fibrosis. A small randomized trial found improvement in oral function with curcumin lozenges, when compared to placebo, indicating that turmeric may hold promise as a treatment of oral submucous fibrosis.19
Uveitis. A small pilot study of 32 patients suggested that oral curcumin may be as effective as corticosteroids for uveitis.20
Heart disease. Curcumin may have a cardiovascular protective role, as it has been shown to reduce atherosclerosis, but a reduction in myocardial infarction or stroke has not been documented.21
Alzheimer’s dementia. Animal studies have shown a reduction in amyloid plaque formation with curcumin.22
Adverse effects (and precautions)
Turmeric in food is considered safe. A variety of animal and human studies have also indicated that curcumin is safe and well tolerated, even at very high doses.13 However, taking large amounts of turmeric for long periods of time could cause stomach upset and gastric ulcers. In addition, patients with gallstones or bile obstruction should use it with caution due to increased bile production.7
Because turmeric may lower blood sugar levels, patients with diabetes should monitor for hypoglycemia when using turmeric in combination with diabetic medications. Similarly, those with bleeding disorders taking blood thinners should use turmeric and curcumin with caution, because it can inhibit platelet aggregation.23
Although it is safe to eat foods with turmeric during pregnancy, pregnant and breastfeeding women should not take turmeric supplements, as the safety of large doses in pregnancy is unknown.
The bottom line
Turmeric/curcumin has anti-inflammatory properties and may be useful as an adjunct for ulcerative colitis and to improve the symptoms of OA. It may also have anti-carcinogenic properties, although definitive data are lacking. Those with a history of gastrointestinal conditions such as gastric ulcer, patients taking blood thinners, and patients with diabetes who are prone to low blood sugar levels should use turmeric/curcumin with caution.
Chamomile
Overview
Chamomile, a member of the Asteraceae/Compositae family, is one of the oldest herbal medicines. It has been used for hay fever, inflammation, muscle spasms, menstrual disorders, insomnia, ulcers, wounds, gastrointestinal disorders, rheumatic pain, and hemorrhoids. Essential oils of chamomile are used extensively in cosmetics and aromatherapy. Many different preparations have been developed, the most popular being herbal tea.24
Individuals with a hypersensitivity to plants of the Asteraceae (Compositae) family such as ragweed (Ambrosia spp.), marigold flower (Calendula officinalis), and chrysanthemum (Chrysanthemum spp.) may show a similar reaction to chamomile.25
Anxiety. A controlled clinical trial of chamomile extract for generalized anxiety disorder (GAD) suggested that it may have modest anxiolytic activity in patients with mild to moderate GAD.26 Another randomized, double-blind, placebo-controlled trial found oral chamomile extract was efficacious and well-tolerated in patients experiencing mild to moderate GAD and may provide an alternative therapeutic anxiolytic for patients with mild GAD.25 In addition to its anxiolytic activity, chamomile may also provide clinically meaningful antidepressant activity.26
Insomnia. Chamomile may have some impact on sleep diary measures (total sleep time, sleep efficiency, sleep latency, wake after sleep onset, sleep quality, and number of awakenings) relative to placebo in adults with chronic primary insomnia, according to a small randomized, double-blind, placebo-controlled pilot trial involving 34 patients.27 However, a systematic review found no statistically significant difference between any herbal medicine (including chamomile) and placebo, for clinical efficacy in patients with insomnia. A similar, or smaller, number of adverse events per person were reported with chamomile compared with placebo, suggesting safe use.28
Infantile colic. A small prospective double-blind study on the use of chamomile-containing tea on infantile colic showed statistically significant symptom improvement in tea-treated infants. The study did note, however, that prolonged ingestion of herbal teas may lead to decreased milk intake.29,30
Adverse effects
As noted earlier, a systematic review found that the number of adverse events per person reported with chamomile was comparable to the number associated with placebo, suggesting that it is safe.28
The bottom line
Chamomile appears to be safe with minimal adverse effects and may be effective for the treatment of anxiety, insomnia, and infantile colic.
Rosemary
Overview
Rosemary, officially known as Rosmarinus officinalis, is a medicinal evergreen plant native to the Mediterranean area that appears to increase microcapillary perfusion.31
Alopecia. A randomized double-blind controlled trial found that essential oils including rosemary oil (as well as thyme, lavender, and cedarwood) massaged into the scalp improved hair growth in almost half of patients with alopecia areata after 7 months.32 Another randomized trial comparing rosemary oil to minoxidil 2% for androgenetic alopecia showed a significant increase in hair count at the 6-month endpoint compared with the baseline, but no significant difference was found between the study groups regarding hair count either at Month 3 or Month 6 (P >.05). 31
Adverse effects
In the randomized trial described above comparing rosemary oil to minoxidil 2%, adverse effects appeared to be rare for topical rosemary oil. Scalp itching was more frequent in the minoxidil group.31
The bottom line
Topical rosemary oil may be useful in the treatment of alopecia with minimal adverse effects.
Coffee/caffeine
Overview
Coffee is one of the most widely used botanicals with approximately 3.5 billion cups of coffee consumed per day worldwide. It is a popular beverage because of its unique aromatic taste and its use as a central nervous system stimulant. The coffee tree (genus coffea) is found throughout Latin America, Africa, and eastern Asia. Two of the most common commercially grown species are Coffea arabica (Arabicas) and Coffea canephora (Robusta). Processing and roasting methods may differ and produce variations in flavor and aroma. The degree of roasting also affects the caffeine content.
Coffee consumption leads to increased alertness and can boost mental performance. Based on the literature and US Food and Drug Administration recommendations, four 8-oz cups of coffee (about 400 mg of caffeine) daily is an acceptable average amount of caffeine. More than 500 mg/d is considered excessive use of coffee.33,34
Overall mortality. A 2008 study showed that regular coffee was not associated with increased or decreased mortality in both men and women.35 However, more recent studies show an inverse relationship between mortality and coffee consumption.
Specifically, a 2014 meta-analysis found an inverse relationship between coffee and mortality.36 A large prospective cohort study from 2015 that included 79,234 women and 76,704 men found that drinking coffee was inversely associated with overall mortality.37 In this cohort study, an inverse association were observed for deaths from heart disease, respiratory disease, diabetes, and self-harm.37 While mechanisms were not analyzed, coffee may reduce mortality risk by affecting inflammation, lung function, insulin sensitivity, and depression.
Cardiovascular disease. Coffee consumption may modestly reduce the risk of stroke, according to a prospective cohort study of 83,076 women from the Nurses’ Health Study who were followed for 24 years.38 Reduced cardiovascular mortality was also found in a large prospective cohort study, as noted in the mortality discussion above.37 A 2014 meta-analysis concluded that coffee consumption is inversely associated with cardiovascular mortality. Drinking 3 or 4 cups a day appears to be the amount that may decrease one’s risk of death when compared to those who do not drink coffee at all.36
Liver disease. Friedrich et al performed a study involving 379 patients with end stage liver disease, and found that coffee consumption delayed the progression of disease in patients with both alcoholic liver disease and primary sclerosing cholangitis.39 Coffee consumption also increased long-term survival after liver transplantation.39 However, the study found that coffee did not have any effect on patients with chronic viral hepatitis.
In a 2016 meta-analysis, caffeinated coffee consumption reduced hepatic fibrosis of nonalcoholic fatty liver disease, although caffeine consumption did not reduce the prevalence of nonalcoholic fatty liver disease.40 Another meta-analysis, including 16 studies, also found caffeine reduced the risk for hepatic fibrosis and cirrhosis.41
Depression. Based on 2 different systematic reviews and meta-analyses from 2016, coffee consumption appears to have a significant protective effect, decreasing the risk of developing depression.40,42
Alzheimer’s disease/dementia. Coffee, tea, and caffeine consumption show promise in reducing the risk of cognitive decline and dementia. Individuals who consume one to 2 cups of coffee per day had a decreased incidence of mild cognitive impairment compared to non-drinkers.43 A 2015 Japanese study also found an inverse association between coffee consumption and dementia among women, nonsmokers, and those who do not drink alcohol.44 Most recently, a 2016 study, the Women’s Health Initiative Memory Study, looked at incident dementia rates in women >65 years of age with high vs low caffeine intake. Women with higher caffeine intake were less likely to develop dementia or any cognitive impairment compared with those consuming <64 mg/day.45
Type 2 diabetes. A 2009 prospective cohort study, which included 40,011 participants followed for more than 10 years, found that drinking at least 3 cups of coffee or tea was associated with a lowered risk of type 2 diabetes.46 A 2009 systematic review of 20 cohort studies showed that high intakes of coffee, decaffeinated coffee, and tea are associated with a reduced risk of diabetes.47
Melanoma.
Adverse effects
Despite the many potential benefits of coffee, caffeine is a potent drug that should be used with caution.49 People with underlying heart problems should avoid caffeine due to concern that it may cause palpitations from tachycardia. It may worsen anxiety problems or depression. Coffee may increase the production of stomach acids, which can worsen acid reflux or stomach ulcers.
Caffeine is a potent diuretic and may decrease absorption of calcium and cause OA. Caffeine may cause dependence and withdrawal symptoms. Some of the symptoms of withdrawal include drowsiness, headaches, irritability, nausea, and vomiting. It may disrupt sleeping patterns by causing jitters and sleeplessness.49 Additionally, large amounts of caffeine may cause overdose and death.
The bottom line
Regular coffee intake is associated with a lower risk of mortality, reduced cardiovascular events, and a reduction in liver disease progression. Coffee may also have some utility for improving cognitive function and reducing the risk of type 2 diabetes. Caffeinated coffee should be limited to no more than 32 oz per day, due to the risk of insomnia, palpitations, anxiety, and gastritis.
Chocolate/cocoa
Overview
Few natural products have been claimed to successfully treat as many disorders as chocolate. The modern concept of chocolate as food has overshadowed its traditional medicinal use, although recent trials have looked at evidence for some of its traditional uses. Chocolate is processed from the pod of the cacao plant. The earliest evidence for its medical use is in Mayan civilizations, and for most of its approximately 4000-year history, chocolate was consumed as a bitter drink referred to as the “drink of the Gods.” The traditional drink was mixed with water, vanilla, honey, chili peppers, and other spices. Important components in chocolate include flavonoids (antioxidants), cocoa butter, caffeine, theobromine, and phenylethylamine.
Chocolate has stimulating, anti-inflammatory, neuroprotective, and cardioprotective effects, and improves the bioavailability of nitric oxide, which can improve blood pressure and platelet function.50 Epicatechin (an antioxidant) in cocoa is primarily responsible for its favorable impact on vascular endothelium via its effect on both acute and chronic upregulation of nitric oxide production. Other cardiovascular effects are mediated by the anti-inflammatory effects of cocoa polyphenols, and modulated through the activity of NF-kappaB.51
Dark chocolate appears to have the greatest benefit, as milk binds to antioxidants in chocolate, making them unavailable. Therefore, milk chocolate is not a good antioxidant source. There is no specific amount of chocolate that is known to be ideal, but an average of one to 2 ounces per day is often used in studies.
Cardiovascular effects. Chocolate does contain saturated fat, but a comparative, double-blind study found that short-term use of cocoa powder lowered plasma low-density lipoprotein (LDL) cholesterol, oxidized LDL, and apo B concentrations, and the plasma high-density lipoprotein (HDL) cholesterol concentration increased, relative to baseline in the low-, middle-, and high-cocoa groups.52 A small randomized crossover trial without clinical outcomes indicated that chocolate may increase HDL cholesterol without increasing weight.53
A meta-analysis of short-term (2-12 weeks) treatment with dark chocolate/cocoa products showed reductions in LDL and total cholesterol, but no changes in HDL or triglycerides.54 Another meta-analysis of RCTs, however, showed no short-term effect of cocoa/chocolate on lipid concentrations.55 A randomized, placebo-controlled double-blind study of 62 patients with diabetes and hypertension showed that high polyphenol chocolate improved triglyceride levels.56
Multiple studies have shown that chocolate is associated with a reduction in cardiovascular risk.57-59 A best case scenario analysis using a Markov model to predict the long-term effectiveness and cost effectiveness of daily dark chocolate consumption in a population with metabolic syndrome at high risk of cardiovascular disease concluded that daily consumption of dark chocolate can reduce cardiovascular events by 85 per 10,000 population treated over 10 years. The study concluded that $42 could be cost effectively spent per person per year on prevention strategies using dark chocolate.59
In addition, a meta-analysis of 7 observational studies showed that high levels of chocolate consumption (any type) were associated with a 29% reduction in stroke compared with the lowest levels of chocolate intake.57 Results of a similar meta-analysis from Neurology in 2012 also suggested that moderate chocolate consumption (any type) may lower the risk of stroke.60
That said, 2 systematic reviews specifically relating to the risk of coronary heart disease and chocolate intake were inconclusive.61-62
Blood pressure (BP). An RCT published in JAMA indicates that inclusion of small amounts of polyphenol-rich dark chocolate as part of a usual diet efficiently reduced BP and improved the formation of vasodilative nitric oxide.63 A meta-analysis of 10 RCTs also showed mean BP change in the active cocoa treatment arms across all trials was -4.5 mm Hg (95% confidence interval (CI), -5.9 to -3.2; P<.001) for systolic BP and -2.5 mm Hg (95% CI, -3.9 to -1.2; P<.001) for diastolic BP.64
A Cochrane Review meta-analysis of 20 studies revealed a statistically significant BP-reducing effect of flavanol-rich cocoa products compared with control in short-term trials of 2 to 18 weeks' duration.65 Because studies have shown improvement in BP with chocolate intake, investigations into a role of chocolate in the prevention of preeclampsia have been undertaken. In some studies, chocolate intake was associated with reduced odds of preeclampsia and gestational hypertension.66,67
Diabetes. Chocolate may exert significant vascular protection because of its antioxidant properties and possible increase of nitric oxide bioavailability, which can influence glucose uptake. A small trial comparing the effects of either dark or white chocolate bars (which do not contain the polyphenols) showed improved BP and glucose and insulin responses to an oral glucose tolerance test in healthy subjects on dark chocolate, but not white chocolate.68 A comparison of chocolate consumption and risk of diabetes in the Physicians’ Health Study showed an inverse relationship between chocolate intake with incident disease, but this association appeared only to apply in younger and normal-body weight men after controlling for comprehensive lifestyles, including total energy consumption.69
Fatigue. The effect of chocolate on a person’s energy level has been noted for centuries.70 A small randomized trial showed improved energy levels in those treated with higher chocolate intakes. In a double-blind, randomized, clinical pilot crossover study, high cocoa liquor/polyphenol rich chocolate, reduced fatigue in subjects with chronic fatigue syndrome.71
Anxiety. A small randomized trial showed chocolate decreased anxiety in high-anxiety trait subjects and improved the anxiety level and the energy levels of low-anxiety trait participants.72
Eye effects. The literature presents conflicting evidence regarding the effect of flavonoids on patients with glaucoma and ocular hypertension. However, a meta-analysis showed that flavonoids have a promising role in improving visual function in patients with glaucoma and ocular hypertension, and appear to play a part in both improving and slowing the progression of visual field loss.73
Cognitive decline. Chocolate intake (any type) was associated with a lower risk of cognitive decline (RR = 0.59; 95% CI, 0.38-0.92) with the greatest benefit noted in those who averaged more than one chocolate bar or one tablespoon of cocoa powder per week. This protective effect was observed only among subjects with an average daily consumption of caffeine <75 mg (69% of the participants; RR = 0.50; 95% CI, 0.31-0.82).74
The bottom line
Chocolate with high cocoa content (dark chocolate) appears to be safe and beneficial as part of a healthy diet and lifestyle that includes exercise and stress reduction to decrease cardiovascular risk and may improve energy levels.
CORRESPONDENCE
Michael Malone, MD, Family and Community Medicine, Penn State College of Medicine, 500 University Drive, Hershey, PA 17033; [email protected].
1. National Center for Complementary and Integrative Health. The use of complementary and alternative medicine in the United States. Available at: https://nccih.nih.gov/research/statistics/2007/camsurvey_fs1.htm. Accessed Nov 28, 2017.
2. Aggarwal BB. Curcumin-free turmeric exhibits anti-inflammatory and anticancer activities: identification of novel components of turmeric. Mol Nutr Food Res. 2013;57:1529-1542.
3. Henrotin Y, Clutterbuck AL, Allaway D, et al. Biological actions of curcumin on articular chondrocytes. Osteoarthritis Cartilage. 2010;18:141-149.
4. Asher GN, Spelman K. Clinical utility of curcumin extract. Altern Ther Health Med. 2013;19:20-22.
5. Phan TT, See P, Lee ST, et al. Protective effects of curcumin against oxidative damage on skin cells in vitro: its implication for wound healing. J Trauma. 2001;51:927-931.
6. Funk JL, Frye JB, Oyarzo JN, et al. Efficacy and mechanism of action of turmeric supplements in the treatment of experimental arthritis. Arthritis Rheum. 2006;54:3452-3464.
7. Patcharatrakul T, Gonlachanvit S. Chili peppers, curcumins, and prebiotics in gastrointestinal health and disease. Curr Gastroenterol Rep. 2016;18:19.
8. Thamlikitkul V, Bunyapraphatsara N, Dechatiwongse T, et al. Randomized double blind study of Curcuma domestica Val. for dyspepsia. J Med Assoc Thai. 1989;72:613-620.
9. Kumar S, Ahuja V, Sankar MJ, et al. Curcumin for maintenance of remission in ulcerative colitis. Cochrane Database Syst Rev. 2012;10:CD008424.
10. Lang A, Salomon N, Wu JC, et al. Curcumin in combination with mesalamine induces remission in patients with mild-to-moderate ulcerative colitis in a randomized controlled trial. Clin Gastroenterol Hepatol. 2015;13:1444-1449.e1.
11. Henrotin Y, Priem F, Mobasheri A. Curcumin: a new paradigm and therapeutic opportunity for the treatment of osteoarthritis: curcumin for osteoarthritis management. Springerplus. 2013;2:56.
12. Kuptniratsaikul V, Dajpratham P, Taechaarpornkul W, et al. Efficacy and safety of Curcuma domestica extracts compared with ibuprofen in patients with knee osteoarthritis: a multicenter study. Clin Interv Aging. 2014;9:451-458.
13. Shehzad A, Lee J, Lee YS. Curcumin in various cancers. Biofactors. 2013;39:56-68.
14. Sordillo LA, Sordillo PP, Helson L. Curcumin for the treatment of glioblastoma. Anticancer Res. 2015;35:6373-6378.
15. Darvesh AS, Aggarwal BB, Bishayee A. Curcumin and liver cancer: a review. Curr Pharm Biotechnol. 2012;13:218-228.
16. Nagaraju GP, Aliya S, Zafar SF, et al. The impact of curcumin on breast cancer. Integr Biol (Camb). 2012;4:996-1007.
17. Johnson JJ, Mukhtar H. Curcumin for chemoprevention of colon cancer. Cancer Lett. 2007;255:170-181.
18. Dorai T, Cao YC, Dorai B, et al. Therapeutic potential of curcumin in human prostate cancer. III. Curcumin inhibits proliferation, induces apoptosis, and inhibits angiogenesis of LNCaP prostate cancer cells in vivo. Prostate. 2001;47:293-303.
19. Hazarey VK, Sakrikar AR, Ganvir SM. Efficacy of curcumin in the treatment for oral submucous fibrosis - a randomized clinical trial. J Oral Maxillofac Pathol. 2015;19:145-152.
20. Lal B, Kapoor AK, Asthana OP, et al. Efficacy of curcumin in the management of chronic anterior uveitis. Phytother Res. 1999;13:318-322.
21. Kapakos G, Youreva V, Srivastava AK. Cardiovascular protection by curcumin: molecular aspects. Indian J Biochem Biophys. 2012;49:306-315.
22. Yang F, Lim GP, Begum AN, et al. Curcumin inhibits formation of amyloid beta oligomers and fibrils, binds plaques, and reduces amyloid in vivo. J Biol Chem. 2005;280:5892-5901.
23. Heck AM, DeWitt BA, Lukes AL. Potential interactions between alternative therapies and warfarin. Am J Health Syst Pharm. 2000;57:1221-1227.
24. Srivastava JK, Shankar E, Gupta S. Chamomile: a herbal medicine of the past with bright future. Mol Med Rep. 2010;3:895-901.
25. Ross SM. Generalized anxiety disorder (GAD): efficacy of standardized matricaria recutita (german chamomile) extract in the treatment of generalized anxiety disorder. Holistic Nursing Practice. 2013;27:366- 368.
26. Amsterdam JD, Li Y, Soeller I, et al. A randomized, double-blind, placebo-controlled trial of oral Matricaria recutita (chamomile) extract therapy for generalized anxiety disorder. J Clin Psychopharmacol. 2009;29:378-382.
27. Zick SM, Wright BD, Sen A, et al. Preliminary examination of the efficacy and safety of a standardized chamomile extract for chronic primary insomnia: a randomized placebo-controlled pilot study. BMC Complement Altern Med. 2011;11:78.
28. Leach MJ, Page AT. Herbal medicine for insomnia: a systematic review and meta-analysis. Sleep Med Rev. 2015;24:1-12.
29. Weizman Z, Alkrinawi S, Goldfarb D, et al. Efficacy of herbal tea preparation in infantile colic. J Pediatr. 1993;122:650.
30. Crotteau CA, Wright ST, Eglash A. Clinical inquiries. What is the best treatment for infants with colic? J Fam Pract. 2006;55:634-636.
31. Panahi Y, Taghizadeh M, Marzony ET, et al. Rosemary oil vs minoxidil 2% for the treatment of androgenetic alopecia: a randomized comparative trial. Skinmed. 2015;13:15-21.
32. Hay IC, Jamieson M, Ormerod AD. Randomized trial of aromatherapy. Successful treatment for alopecia areata. Arch Dermatol. 1998;134:1349-1352.
33. Caffeine and kids: FDA takes a closer look. Available at: https://www.fda.gov/ForConsumers/ConsumerUpdates/ucm350570.htm. Accessed: November 1, 2017.
34. Torpy JM, Livingston EH. Energy Drinks. JAMA. 2013;309:297.
35. Lopez-Garcia E, van Dam RM, Li TY, et al. The relationship of coffee consumption with mortality. Ann Intern Med. 2008;148:904-914.
36. Crippa A, Discacciati A, Larsson SC, et al. Coffee consumption and mortality from all causes, cardiovascular disease, and cancer: a dose-response meta-analysis. Am J Epidemiol. 2014;180:763-775.
37. Loftfield E, Freedman ND, Graubard BI, et al. Association of coffee consumption with overall and cause-specific mortality in a large US prospective cohort study. Am J Epidemiol. 2015;182:1010-1022.
38. Lopez-Garcia E, Rodriguez-Artalejo F, Rexrode KM, et al. Coffee consumption and risk of stroke in women. Circulation. 2009;119:1116-1123.
39. Friedrich K, Smit M, Wannhoff A, et al. Coffee consumption protects against progression in liver cirrhosis and increases long-term survival after liver transplantation. J Gastroenterol Hepatol. 2016;31:1470-1475.
40. Wang L, Shen X, Wu Y, et al. Coffee and caffeine consumption and depression: a meta-analysis of observational studies. Aust N Z J Psychiatry. 2016;50:228-242.
41. Liu F, Wang X, Wu G, et al. Coffee consumption decreases risks for hepatic fibrosis and cirrhosis: a meta-analysis. PLoS One. 2015;10:e0142457.
42. Grosso G, Micek A, Castellano S, et al. Coffee, tea, caffeine and risk of depression: a systematic review and dose-response meta-analysis of observational studies. Mol Nutr Food Res. 2016;60:223-234.
43. Solfrizzi V, Panza F, Imbimbo BP, et al. Italian longitudinal study on aging working group. Coffee consumption habits and the risk of mild cognitive impairment: The Italian Longitudinal Study on Aging. J Alzheimers Dis. 2015;47:889-899.
44. Sugiyama K, Tomata Y, Kaiho Y, et al. Association between coffee consumption and incident risk of disabling dementia in elderly japanese: The Ohsaki Cohort 2006 Study. J Alzheimers Dis. 2015;50:491-500.
45. Driscoll I, Shumaker SA, Snively BM, et al. Relationships between caffeine intake and risk for probable dementia or global cognitive impairment: The Women’s Health Initiative Memory Study. J Gerontol A Biol Sci Med Sci. 2016;71:1596-1602.
46. van Dieren S, Uiterwaal CS, van der Schouw YT, et al. Coffee and tea consumption and risk of type 2 diabetes. Diabetologia. 2009;52:2561-2569.
47. Wang J, Li X, Zhang D. Coffee consumption and the risk of cutaneous melanoma: a meta-analysis. Eur J Nutr. 2016;55:1317-1329.
48. Liu J, Shen B, Shi M, et al. Higher caffeinated coffee intake is associated with reduced malignant melanoma risk: a meta-analysis study. PLoS One. 2016;11:e0147056.
49. Wikoff D, Welsh BT, Henderson R, et al. Systematic review of the potential adverse effects of caffeine consumption in healthy adults, pregnant women, adolescents, and children. Food Chem Toxical. 2017;109(Pt 1):585-648.
50. Verna R. The history and science of chocolate. Malays J Pathol. 2013;35:111-121.
51. Katz DL, Doughty K, Ali A. Cocoa and chocolate in human health and disease. Antioxid Redox Signal. 2011;15:2779-2811.
52. Baba S, Natsume M, Yasuda A, et al. Plasma LDL and HDL cholesterol and oxidized LDL concentrations are altered in normo- and hypercholesterolemic humans after intake of different levels of cocoa powder. J Nutr. 2007;137:1436-1441.
53. Mellor DD, Sathyapalan T, Kilpatrick ES, et al. High-cocoa polyphenol-rich chocolate improves HDL cholesterol in type 2 diabetes patients. Diabet Med. 2010;27:1318-1321.
54. Tokede OA, Gaziano JM, Djoussé L. Effects of cocoa products/dark chocolate on serum lipids: a meta-analysis. Eur J Clin Nutr. 2011;65:879-886.
55. Jia L, Liu X, Bai YY, et al. Short-term effect of cocoa product consumption on lipid profile: a meta-analysis of randomized controlled trials. Am J Clin Nutr. 2010;92:218-225.
56. Rostami A, Khalili M, Haghighat N, et al. High-cocoa polyphenol-rich chocolate improves blood pressure in patients with diabetes and hypertension. ARYA Atheroscler. 2015;11:21-29.
57. Buitrago-Lopez A, Sanderson J, Johnson L, et al. Chocolate consumption and cardiometabolic disorders: systematic review and meta-analysis. BMJ. 2011;26;343:d4488.
58. Wang X, Ouyang YY, Liu J, et al. Flavonoid intake and risk of CVD: a systematic review and meta-analysis of prospective cohort studies. Br J Nutr. 2014;111:1-11.
59. Zomer E, Owen A, Magliano DJ, et al. The effectiveness and cost effectiveness of dark chocolate consumption as prevention therapy in people at high risk of cardiovascular disease: best case scenario analysis using a Markov model. BMJ. 2012;344:e3657.
60. Larsson SC, Virtamo J, Wolk A. Chocolate consumption and risk of stroke: a prospective cohort of men and meta-analysis. Neurology. 2012;79:1223-1229.
61. Khawaja O, Gaziano JM, Djoussé L. Chocolate and coronary heart disease: a systematic review. Curr Atheroscler Rep. 2011;13:447-452.
62. Jacques PF, Cassidy A, Rogers G, et al. Dietary flavonoid intakes and CVD incidence in the Framingham Offspring Cohort. Br J Nutr. 2015;114:1496-1503.
63. Taubert D, Roesen R, Lehmann C, et al. Effects of low habitual cocoa intake on blood pressure and bioactive nitric oxide: a randomized controlled trial. JAMA. 2007;298:49-60.
64. Desch S, Schmidt J, Kobler D, et al. Effect of cocoa products on blood pressure: systematic review and meta-analysis. Am J Hypertens. 2010;23:97-103.
65. Ried K, Sullivan TR, Fakler P, et al. Effect of cocoa on blood pressure. Cochrane Database Syst Rev. 2012;8:CD008893.
66. Saftlas AF, Triche EW, Beydoun H, et al. Does chocolate intake during pregnancy reduce the risks of preeclampsia and gestational hypertension? Ann Epidemiol. 2010;20:584-591.
67. Triche EW, Grosso LM, Belanger K, et al. Chocolate consumption in pregnancy and reduced likelihood of preeclampsia. Epidemiology. 2008;19:459-464.
68. Grassi D, Lippi C, Necozione S, et al. Short-term administration of dark chocolate is followed by a significant increase in insulin sensitivity and a decrease in blood pressure in healthy persons. Am J Clin Nutr. 2005;81:611-614.
69. Matsumoto C, Petrone AB, Sesso HD, et al. Chocolate consumption and risk of diabetes mellitus in the Physicians’ Health Study. Am J Clin Nutr. 2015;101:362-367.
70. Lippi D. Chocolate in history: food, medicine, medi-food. Nutrients. 2013;5:1573-1584.
71. Sathyapalan T, Beckett S, Rigby AS, et al. High cocoa polyphenol rich chocolate may reduce the burden of the symptoms in chronic fatigue syndrome. Nutr J. 2010;9:55.
72. Martin FP, Antille N, Rezzi S, et al. Everyday eating experiences of chocolate and non-chocolate snacks impact postprandial anxiety, energy and emotional states. Nutrients. 2012;4:554-567.
73. Patel S, Mathan JJ, Vaghefi E, et al. The effect of flavonoids on visual function in patients with glaucoma or ocular hypertension: a systematic review and meta-analysis. Graefes Arch Clin Exp Ophthalmol. 2015;253:1841-1850.
74. Moreira A, Diógenes MJ, de Mendonça A, et al. Chocolate consumption is associated with a lower risk of cognitive decline. J Alzheimers Dis. 2016;53:85-93.
More than a third of American adults use complementary and alternative medicine.1 Unfortunately, the public’s enthusiasm for herbal products is not always consistent with the scientific evidence supporting their use. In part one of this series, we discussed the studies that have been done on capsaicin, butterbur, green tea, and peppermint. In this installment, we outline the research on 5 additional remedies: turmeric/curcumin, which may be of benefit in ulcerative colitis; chamomile, which appears to offer relief to patients with anxiety; rosemary, which may help treat alopecia; as well as coffee and cocoa, which may have some cardiovascular benefits (TABLE).

Turmeric/curcumin
Overview
Turmeric (Curcuma longa), a relative of ginger, has been used for 4000 years to treat a variety of conditions.2,3 Curcumin is the yellow pigment isolated from the rhizomes of Curcuma longa, commonly known as turmeric.3 Turmeric powder contains 5% curcumin, which is the main biologically active compound. Although it grows in many tropical locations, most turmeric is grown in India, where it is used as a main ingredient in curry. The roots and bulbs of turmeric that are used in medicine are generally boiled and dried, which results in a yellow powder.
Turmeric has been used in both Ayurvedic and Chinese medicine for its anti-inflammatory properties, in the treatment of digestive and liver problems, to fight infections, and to help heal skin diseases and wounds.3-7
Functional GI disorders. A recent review noted that curcumin has been shown in several preclinical studies and uncontrolled clinical trials to have effects on gut inflammation, gut permeability, and the brain-gut axis, especially in functional GI disorders.7 A double-blind, placebo-controlled study from 1989 found that turmeric reduced symptoms of bloating and gas in subjects suffering from undifferentiated dyspepsia.8
Ulcerative colitis (UC). A 2012 Cochrane review noted that curcumin appears to be a safe and effective therapy for maintenance of remission in quiescent UC when given as adjunctive therapy along with mesalamine or sulfasalazine.9 In a 2015 randomized controlled trial (RCT), the addition of curcumin to mesalamine therapy was superior to the combination of placebo and mesalamine in inducing clinical and endoscopic remission in patients with mild-to-moderate active UC, producing no apparent adverse effects.10
Osteoarthritis (OA). Because of turmeric’s ability to reduce inflammation, it may help relieve OA pain.3 Clinical evidence is scant for the anti-arthritic efficacy of turmeric dietary supplements, although animal studies indicate that turmeric prevents inflammation through regulation of NF-kappaB-regulated genes that regulate the immune and inflammatory response.6 Inflammatory cell influx, joint levels of prostaglandin E2, and periarticular osteoclast formation were also inhibited by turmeric extract treatment.6
A 2013 review of turmeric for OA concluded that observational studies and in vitro results are promising for the use of curcumin for OA, but well-designed clinical studies were lacking and are needed to support the efficacy of curcumin in OA patients.11 However, in a 2014 randomized trial of 367 patients, turmeric appeared to be similar in efficacy to ibuprofen for the treatment of pain and disability in adults with knee OA.12 The curcumin (turmeric) group also had fewer adverse effects.12
Cancer. There has been a great deal of research on turmeric’s anti-cancer properties, but clinical evidence is lacking. In vitro evidence, animal studies, and small clinical trials suggest that curcumin may help prevent or treat several types of cancers, but the overall evidence is poor. Nonetheless, curcumin and turmeric have been or are currently being evaluated for the treatment or prevention of prostate, liver, breast, skin, gynecologic, hematologic, pulmonary, thymic, bone, brain, and colon cancer.13-18
Oral submucous fibrosis. A small randomized trial found improvement in oral function with curcumin lozenges, when compared to placebo, indicating that turmeric may hold promise as a treatment of oral submucous fibrosis.19
Uveitis. A small pilot study of 32 patients suggested that oral curcumin may be as effective as corticosteroids for uveitis.20
Heart disease. Curcumin may have a cardiovascular protective role, as it has been shown to reduce atherosclerosis, but a reduction in myocardial infarction or stroke has not been documented.21
Alzheimer’s dementia. Animal studies have shown a reduction in amyloid plaque formation with curcumin.22
Adverse effects (and precautions)
Turmeric in food is considered safe. A variety of animal and human studies have also indicated that curcumin is safe and well tolerated, even at very high doses.13 However, taking large amounts of turmeric for long periods of time could cause stomach upset and gastric ulcers. In addition, patients with gallstones or bile obstruction should use it with caution due to increased bile production.7
Because turmeric may lower blood sugar levels, patients with diabetes should monitor for hypoglycemia when using turmeric in combination with diabetic medications. Similarly, those with bleeding disorders taking blood thinners should use turmeric and curcumin with caution, because it can inhibit platelet aggregation.23
Although it is safe to eat foods with turmeric during pregnancy, pregnant and breastfeeding women should not take turmeric supplements, as the safety of large doses in pregnancy is unknown.
The bottom line
Turmeric/curcumin has anti-inflammatory properties and may be useful as an adjunct for ulcerative colitis and to improve the symptoms of OA. It may also have anti-carcinogenic properties, although definitive data are lacking. Those with a history of gastrointestinal conditions such as gastric ulcer, patients taking blood thinners, and patients with diabetes who are prone to low blood sugar levels should use turmeric/curcumin with caution.
Chamomile
Overview
Chamomile, a member of the Asteraceae/Compositae family, is one of the oldest herbal medicines. It has been used for hay fever, inflammation, muscle spasms, menstrual disorders, insomnia, ulcers, wounds, gastrointestinal disorders, rheumatic pain, and hemorrhoids. Essential oils of chamomile are used extensively in cosmetics and aromatherapy. Many different preparations have been developed, the most popular being herbal tea.24
Individuals with a hypersensitivity to plants of the Asteraceae (Compositae) family such as ragweed (Ambrosia spp.), marigold flower (Calendula officinalis), and chrysanthemum (Chrysanthemum spp.) may show a similar reaction to chamomile.25
Anxiety. A controlled clinical trial of chamomile extract for generalized anxiety disorder (GAD) suggested that it may have modest anxiolytic activity in patients with mild to moderate GAD.26 Another randomized, double-blind, placebo-controlled trial found oral chamomile extract was efficacious and well-tolerated in patients experiencing mild to moderate GAD and may provide an alternative therapeutic anxiolytic for patients with mild GAD.25 In addition to its anxiolytic activity, chamomile may also provide clinically meaningful antidepressant activity.26
Insomnia. Chamomile may have some impact on sleep diary measures (total sleep time, sleep efficiency, sleep latency, wake after sleep onset, sleep quality, and number of awakenings) relative to placebo in adults with chronic primary insomnia, according to a small randomized, double-blind, placebo-controlled pilot trial involving 34 patients.27 However, a systematic review found no statistically significant difference between any herbal medicine (including chamomile) and placebo, for clinical efficacy in patients with insomnia. A similar, or smaller, number of adverse events per person were reported with chamomile compared with placebo, suggesting safe use.28
Infantile colic. A small prospective double-blind study on the use of chamomile-containing tea on infantile colic showed statistically significant symptom improvement in tea-treated infants. The study did note, however, that prolonged ingestion of herbal teas may lead to decreased milk intake.29,30
Adverse effects
As noted earlier, a systematic review found that the number of adverse events per person reported with chamomile was comparable to the number associated with placebo, suggesting that it is safe.28
The bottom line
Chamomile appears to be safe with minimal adverse effects and may be effective for the treatment of anxiety, insomnia, and infantile colic.
Rosemary
Overview
Rosemary, officially known as Rosmarinus officinalis, is a medicinal evergreen plant native to the Mediterranean area that appears to increase microcapillary perfusion.31
Alopecia. A randomized double-blind controlled trial found that essential oils including rosemary oil (as well as thyme, lavender, and cedarwood) massaged into the scalp improved hair growth in almost half of patients with alopecia areata after 7 months.32 Another randomized trial comparing rosemary oil to minoxidil 2% for androgenetic alopecia showed a significant increase in hair count at the 6-month endpoint compared with the baseline, but no significant difference was found between the study groups regarding hair count either at Month 3 or Month 6 (P >.05). 31
Adverse effects
In the randomized trial described above comparing rosemary oil to minoxidil 2%, adverse effects appeared to be rare for topical rosemary oil. Scalp itching was more frequent in the minoxidil group.31
The bottom line
Topical rosemary oil may be useful in the treatment of alopecia with minimal adverse effects.
Coffee/caffeine
Overview
Coffee is one of the most widely used botanicals with approximately 3.5 billion cups of coffee consumed per day worldwide. It is a popular beverage because of its unique aromatic taste and its use as a central nervous system stimulant. The coffee tree (genus coffea) is found throughout Latin America, Africa, and eastern Asia. Two of the most common commercially grown species are Coffea arabica (Arabicas) and Coffea canephora (Robusta). Processing and roasting methods may differ and produce variations in flavor and aroma. The degree of roasting also affects the caffeine content.
Coffee consumption leads to increased alertness and can boost mental performance. Based on the literature and US Food and Drug Administration recommendations, four 8-oz cups of coffee (about 400 mg of caffeine) daily is an acceptable average amount of caffeine. More than 500 mg/d is considered excessive use of coffee.33,34
Overall mortality. A 2008 study showed that regular coffee was not associated with increased or decreased mortality in both men and women.35 However, more recent studies show an inverse relationship between mortality and coffee consumption.
Specifically, a 2014 meta-analysis found an inverse relationship between coffee and mortality.36 A large prospective cohort study from 2015 that included 79,234 women and 76,704 men found that drinking coffee was inversely associated with overall mortality.37 In this cohort study, an inverse association were observed for deaths from heart disease, respiratory disease, diabetes, and self-harm.37 While mechanisms were not analyzed, coffee may reduce mortality risk by affecting inflammation, lung function, insulin sensitivity, and depression.
Cardiovascular disease. Coffee consumption may modestly reduce the risk of stroke, according to a prospective cohort study of 83,076 women from the Nurses’ Health Study who were followed for 24 years.38 Reduced cardiovascular mortality was also found in a large prospective cohort study, as noted in the mortality discussion above.37 A 2014 meta-analysis concluded that coffee consumption is inversely associated with cardiovascular mortality. Drinking 3 or 4 cups a day appears to be the amount that may decrease one’s risk of death when compared to those who do not drink coffee at all.36
Liver disease. Friedrich et al performed a study involving 379 patients with end stage liver disease, and found that coffee consumption delayed the progression of disease in patients with both alcoholic liver disease and primary sclerosing cholangitis.39 Coffee consumption also increased long-term survival after liver transplantation.39 However, the study found that coffee did not have any effect on patients with chronic viral hepatitis.
In a 2016 meta-analysis, caffeinated coffee consumption reduced hepatic fibrosis of nonalcoholic fatty liver disease, although caffeine consumption did not reduce the prevalence of nonalcoholic fatty liver disease.40 Another meta-analysis, including 16 studies, also found caffeine reduced the risk for hepatic fibrosis and cirrhosis.41
Depression. Based on 2 different systematic reviews and meta-analyses from 2016, coffee consumption appears to have a significant protective effect, decreasing the risk of developing depression.40,42
Alzheimer’s disease/dementia. Coffee, tea, and caffeine consumption show promise in reducing the risk of cognitive decline and dementia. Individuals who consume one to 2 cups of coffee per day had a decreased incidence of mild cognitive impairment compared to non-drinkers.43 A 2015 Japanese study also found an inverse association between coffee consumption and dementia among women, nonsmokers, and those who do not drink alcohol.44 Most recently, a 2016 study, the Women’s Health Initiative Memory Study, looked at incident dementia rates in women >65 years of age with high vs low caffeine intake. Women with higher caffeine intake were less likely to develop dementia or any cognitive impairment compared with those consuming <64 mg/day.45
Type 2 diabetes. A 2009 prospective cohort study, which included 40,011 participants followed for more than 10 years, found that drinking at least 3 cups of coffee or tea was associated with a lowered risk of type 2 diabetes.46 A 2009 systematic review of 20 cohort studies showed that high intakes of coffee, decaffeinated coffee, and tea are associated with a reduced risk of diabetes.47
Melanoma.
Adverse effects
Despite the many potential benefits of coffee, caffeine is a potent drug that should be used with caution.49 People with underlying heart problems should avoid caffeine due to concern that it may cause palpitations from tachycardia. It may worsen anxiety problems or depression. Coffee may increase the production of stomach acids, which can worsen acid reflux or stomach ulcers.
Caffeine is a potent diuretic and may decrease absorption of calcium and cause OA. Caffeine may cause dependence and withdrawal symptoms. Some of the symptoms of withdrawal include drowsiness, headaches, irritability, nausea, and vomiting. It may disrupt sleeping patterns by causing jitters and sleeplessness.49 Additionally, large amounts of caffeine may cause overdose and death.
The bottom line
Regular coffee intake is associated with a lower risk of mortality, reduced cardiovascular events, and a reduction in liver disease progression. Coffee may also have some utility for improving cognitive function and reducing the risk of type 2 diabetes. Caffeinated coffee should be limited to no more than 32 oz per day, due to the risk of insomnia, palpitations, anxiety, and gastritis.
Chocolate/cocoa
Overview
Few natural products have been claimed to successfully treat as many disorders as chocolate. The modern concept of chocolate as food has overshadowed its traditional medicinal use, although recent trials have looked at evidence for some of its traditional uses. Chocolate is processed from the pod of the cacao plant. The earliest evidence for its medical use is in Mayan civilizations, and for most of its approximately 4000-year history, chocolate was consumed as a bitter drink referred to as the “drink of the Gods.” The traditional drink was mixed with water, vanilla, honey, chili peppers, and other spices. Important components in chocolate include flavonoids (antioxidants), cocoa butter, caffeine, theobromine, and phenylethylamine.
Chocolate has stimulating, anti-inflammatory, neuroprotective, and cardioprotective effects, and improves the bioavailability of nitric oxide, which can improve blood pressure and platelet function.50 Epicatechin (an antioxidant) in cocoa is primarily responsible for its favorable impact on vascular endothelium via its effect on both acute and chronic upregulation of nitric oxide production. Other cardiovascular effects are mediated by the anti-inflammatory effects of cocoa polyphenols, and modulated through the activity of NF-kappaB.51
Dark chocolate appears to have the greatest benefit, as milk binds to antioxidants in chocolate, making them unavailable. Therefore, milk chocolate is not a good antioxidant source. There is no specific amount of chocolate that is known to be ideal, but an average of one to 2 ounces per day is often used in studies.
Cardiovascular effects. Chocolate does contain saturated fat, but a comparative, double-blind study found that short-term use of cocoa powder lowered plasma low-density lipoprotein (LDL) cholesterol, oxidized LDL, and apo B concentrations, and the plasma high-density lipoprotein (HDL) cholesterol concentration increased, relative to baseline in the low-, middle-, and high-cocoa groups.52 A small randomized crossover trial without clinical outcomes indicated that chocolate may increase HDL cholesterol without increasing weight.53
A meta-analysis of short-term (2-12 weeks) treatment with dark chocolate/cocoa products showed reductions in LDL and total cholesterol, but no changes in HDL or triglycerides.54 Another meta-analysis of RCTs, however, showed no short-term effect of cocoa/chocolate on lipid concentrations.55 A randomized, placebo-controlled double-blind study of 62 patients with diabetes and hypertension showed that high polyphenol chocolate improved triglyceride levels.56
Multiple studies have shown that chocolate is associated with a reduction in cardiovascular risk.57-59 A best case scenario analysis using a Markov model to predict the long-term effectiveness and cost effectiveness of daily dark chocolate consumption in a population with metabolic syndrome at high risk of cardiovascular disease concluded that daily consumption of dark chocolate can reduce cardiovascular events by 85 per 10,000 population treated over 10 years. The study concluded that $42 could be cost effectively spent per person per year on prevention strategies using dark chocolate.59
In addition, a meta-analysis of 7 observational studies showed that high levels of chocolate consumption (any type) were associated with a 29% reduction in stroke compared with the lowest levels of chocolate intake.57 Results of a similar meta-analysis from Neurology in 2012 also suggested that moderate chocolate consumption (any type) may lower the risk of stroke.60
That said, 2 systematic reviews specifically relating to the risk of coronary heart disease and chocolate intake were inconclusive.61-62
Blood pressure (BP). An RCT published in JAMA indicates that inclusion of small amounts of polyphenol-rich dark chocolate as part of a usual diet efficiently reduced BP and improved the formation of vasodilative nitric oxide.63 A meta-analysis of 10 RCTs also showed mean BP change in the active cocoa treatment arms across all trials was -4.5 mm Hg (95% confidence interval (CI), -5.9 to -3.2; P<.001) for systolic BP and -2.5 mm Hg (95% CI, -3.9 to -1.2; P<.001) for diastolic BP.64
A Cochrane Review meta-analysis of 20 studies revealed a statistically significant BP-reducing effect of flavanol-rich cocoa products compared with control in short-term trials of 2 to 18 weeks' duration.65 Because studies have shown improvement in BP with chocolate intake, investigations into a role of chocolate in the prevention of preeclampsia have been undertaken. In some studies, chocolate intake was associated with reduced odds of preeclampsia and gestational hypertension.66,67
Diabetes. Chocolate may exert significant vascular protection because of its antioxidant properties and possible increase of nitric oxide bioavailability, which can influence glucose uptake. A small trial comparing the effects of either dark or white chocolate bars (which do not contain the polyphenols) showed improved BP and glucose and insulin responses to an oral glucose tolerance test in healthy subjects on dark chocolate, but not white chocolate.68 A comparison of chocolate consumption and risk of diabetes in the Physicians’ Health Study showed an inverse relationship between chocolate intake with incident disease, but this association appeared only to apply in younger and normal-body weight men after controlling for comprehensive lifestyles, including total energy consumption.69
Fatigue. The effect of chocolate on a person’s energy level has been noted for centuries.70 A small randomized trial showed improved energy levels in those treated with higher chocolate intakes. In a double-blind, randomized, clinical pilot crossover study, high cocoa liquor/polyphenol rich chocolate, reduced fatigue in subjects with chronic fatigue syndrome.71
Anxiety. A small randomized trial showed chocolate decreased anxiety in high-anxiety trait subjects and improved the anxiety level and the energy levels of low-anxiety trait participants.72
Eye effects. The literature presents conflicting evidence regarding the effect of flavonoids on patients with glaucoma and ocular hypertension. However, a meta-analysis showed that flavonoids have a promising role in improving visual function in patients with glaucoma and ocular hypertension, and appear to play a part in both improving and slowing the progression of visual field loss.73
Cognitive decline. Chocolate intake (any type) was associated with a lower risk of cognitive decline (RR = 0.59; 95% CI, 0.38-0.92) with the greatest benefit noted in those who averaged more than one chocolate bar or one tablespoon of cocoa powder per week. This protective effect was observed only among subjects with an average daily consumption of caffeine <75 mg (69% of the participants; RR = 0.50; 95% CI, 0.31-0.82).74
The bottom line
Chocolate with high cocoa content (dark chocolate) appears to be safe and beneficial as part of a healthy diet and lifestyle that includes exercise and stress reduction to decrease cardiovascular risk and may improve energy levels.
CORRESPONDENCE
Michael Malone, MD, Family and Community Medicine, Penn State College of Medicine, 500 University Drive, Hershey, PA 17033; [email protected].
More than a third of American adults use complementary and alternative medicine.1 Unfortunately, the public’s enthusiasm for herbal products is not always consistent with the scientific evidence supporting their use. In part one of this series, we discussed the studies that have been done on capsaicin, butterbur, green tea, and peppermint. In this installment, we outline the research on 5 additional remedies: turmeric/curcumin, which may be of benefit in ulcerative colitis; chamomile, which appears to offer relief to patients with anxiety; rosemary, which may help treat alopecia; as well as coffee and cocoa, which may have some cardiovascular benefits (TABLE).

Turmeric/curcumin
Overview
Turmeric (Curcuma longa), a relative of ginger, has been used for 4000 years to treat a variety of conditions.2,3 Curcumin is the yellow pigment isolated from the rhizomes of Curcuma longa, commonly known as turmeric.3 Turmeric powder contains 5% curcumin, which is the main biologically active compound. Although it grows in many tropical locations, most turmeric is grown in India, where it is used as a main ingredient in curry. The roots and bulbs of turmeric that are used in medicine are generally boiled and dried, which results in a yellow powder.
Turmeric has been used in both Ayurvedic and Chinese medicine for its anti-inflammatory properties, in the treatment of digestive and liver problems, to fight infections, and to help heal skin diseases and wounds.3-7
Functional GI disorders. A recent review noted that curcumin has been shown in several preclinical studies and uncontrolled clinical trials to have effects on gut inflammation, gut permeability, and the brain-gut axis, especially in functional GI disorders.7 A double-blind, placebo-controlled study from 1989 found that turmeric reduced symptoms of bloating and gas in subjects suffering from undifferentiated dyspepsia.8
Ulcerative colitis (UC). A 2012 Cochrane review noted that curcumin appears to be a safe and effective therapy for maintenance of remission in quiescent UC when given as adjunctive therapy along with mesalamine or sulfasalazine.9 In a 2015 randomized controlled trial (RCT), the addition of curcumin to mesalamine therapy was superior to the combination of placebo and mesalamine in inducing clinical and endoscopic remission in patients with mild-to-moderate active UC, producing no apparent adverse effects.10
Osteoarthritis (OA). Because of turmeric’s ability to reduce inflammation, it may help relieve OA pain.3 Clinical evidence is scant for the anti-arthritic efficacy of turmeric dietary supplements, although animal studies indicate that turmeric prevents inflammation through regulation of NF-kappaB-regulated genes that regulate the immune and inflammatory response.6 Inflammatory cell influx, joint levels of prostaglandin E2, and periarticular osteoclast formation were also inhibited by turmeric extract treatment.6
A 2013 review of turmeric for OA concluded that observational studies and in vitro results are promising for the use of curcumin for OA, but well-designed clinical studies were lacking and are needed to support the efficacy of curcumin in OA patients.11 However, in a 2014 randomized trial of 367 patients, turmeric appeared to be similar in efficacy to ibuprofen for the treatment of pain and disability in adults with knee OA.12 The curcumin (turmeric) group also had fewer adverse effects.12
Cancer. There has been a great deal of research on turmeric’s anti-cancer properties, but clinical evidence is lacking. In vitro evidence, animal studies, and small clinical trials suggest that curcumin may help prevent or treat several types of cancers, but the overall evidence is poor. Nonetheless, curcumin and turmeric have been or are currently being evaluated for the treatment or prevention of prostate, liver, breast, skin, gynecologic, hematologic, pulmonary, thymic, bone, brain, and colon cancer.13-18
Oral submucous fibrosis. A small randomized trial found improvement in oral function with curcumin lozenges, when compared to placebo, indicating that turmeric may hold promise as a treatment of oral submucous fibrosis.19
Uveitis. A small pilot study of 32 patients suggested that oral curcumin may be as effective as corticosteroids for uveitis.20
Heart disease. Curcumin may have a cardiovascular protective role, as it has been shown to reduce atherosclerosis, but a reduction in myocardial infarction or stroke has not been documented.21
Alzheimer’s dementia. Animal studies have shown a reduction in amyloid plaque formation with curcumin.22
Adverse effects (and precautions)
Turmeric in food is considered safe. A variety of animal and human studies have also indicated that curcumin is safe and well tolerated, even at very high doses.13 However, taking large amounts of turmeric for long periods of time could cause stomach upset and gastric ulcers. In addition, patients with gallstones or bile obstruction should use it with caution due to increased bile production.7
Because turmeric may lower blood sugar levels, patients with diabetes should monitor for hypoglycemia when using turmeric in combination with diabetic medications. Similarly, those with bleeding disorders taking blood thinners should use turmeric and curcumin with caution, because it can inhibit platelet aggregation.23
Although it is safe to eat foods with turmeric during pregnancy, pregnant and breastfeeding women should not take turmeric supplements, as the safety of large doses in pregnancy is unknown.
The bottom line
Turmeric/curcumin has anti-inflammatory properties and may be useful as an adjunct for ulcerative colitis and to improve the symptoms of OA. It may also have anti-carcinogenic properties, although definitive data are lacking. Those with a history of gastrointestinal conditions such as gastric ulcer, patients taking blood thinners, and patients with diabetes who are prone to low blood sugar levels should use turmeric/curcumin with caution.
Chamomile
Overview
Chamomile, a member of the Asteraceae/Compositae family, is one of the oldest herbal medicines. It has been used for hay fever, inflammation, muscle spasms, menstrual disorders, insomnia, ulcers, wounds, gastrointestinal disorders, rheumatic pain, and hemorrhoids. Essential oils of chamomile are used extensively in cosmetics and aromatherapy. Many different preparations have been developed, the most popular being herbal tea.24
Individuals with a hypersensitivity to plants of the Asteraceae (Compositae) family such as ragweed (Ambrosia spp.), marigold flower (Calendula officinalis), and chrysanthemum (Chrysanthemum spp.) may show a similar reaction to chamomile.25
Anxiety. A controlled clinical trial of chamomile extract for generalized anxiety disorder (GAD) suggested that it may have modest anxiolytic activity in patients with mild to moderate GAD.26 Another randomized, double-blind, placebo-controlled trial found oral chamomile extract was efficacious and well-tolerated in patients experiencing mild to moderate GAD and may provide an alternative therapeutic anxiolytic for patients with mild GAD.25 In addition to its anxiolytic activity, chamomile may also provide clinically meaningful antidepressant activity.26
Insomnia. Chamomile may have some impact on sleep diary measures (total sleep time, sleep efficiency, sleep latency, wake after sleep onset, sleep quality, and number of awakenings) relative to placebo in adults with chronic primary insomnia, according to a small randomized, double-blind, placebo-controlled pilot trial involving 34 patients.27 However, a systematic review found no statistically significant difference between any herbal medicine (including chamomile) and placebo, for clinical efficacy in patients with insomnia. A similar, or smaller, number of adverse events per person were reported with chamomile compared with placebo, suggesting safe use.28
Infantile colic. A small prospective double-blind study on the use of chamomile-containing tea on infantile colic showed statistically significant symptom improvement in tea-treated infants. The study did note, however, that prolonged ingestion of herbal teas may lead to decreased milk intake.29,30
Adverse effects
As noted earlier, a systematic review found that the number of adverse events per person reported with chamomile was comparable to the number associated with placebo, suggesting that it is safe.28
The bottom line
Chamomile appears to be safe with minimal adverse effects and may be effective for the treatment of anxiety, insomnia, and infantile colic.
Rosemary
Overview
Rosemary, officially known as Rosmarinus officinalis, is a medicinal evergreen plant native to the Mediterranean area that appears to increase microcapillary perfusion.31
Alopecia. A randomized double-blind controlled trial found that essential oils including rosemary oil (as well as thyme, lavender, and cedarwood) massaged into the scalp improved hair growth in almost half of patients with alopecia areata after 7 months.32 Another randomized trial comparing rosemary oil to minoxidil 2% for androgenetic alopecia showed a significant increase in hair count at the 6-month endpoint compared with the baseline, but no significant difference was found between the study groups regarding hair count either at Month 3 or Month 6 (P >.05). 31
Adverse effects
In the randomized trial described above comparing rosemary oil to minoxidil 2%, adverse effects appeared to be rare for topical rosemary oil. Scalp itching was more frequent in the minoxidil group.31
The bottom line
Topical rosemary oil may be useful in the treatment of alopecia with minimal adverse effects.
Coffee/caffeine
Overview
Coffee is one of the most widely used botanicals with approximately 3.5 billion cups of coffee consumed per day worldwide. It is a popular beverage because of its unique aromatic taste and its use as a central nervous system stimulant. The coffee tree (genus coffea) is found throughout Latin America, Africa, and eastern Asia. Two of the most common commercially grown species are Coffea arabica (Arabicas) and Coffea canephora (Robusta). Processing and roasting methods may differ and produce variations in flavor and aroma. The degree of roasting also affects the caffeine content.
Coffee consumption leads to increased alertness and can boost mental performance. Based on the literature and US Food and Drug Administration recommendations, four 8-oz cups of coffee (about 400 mg of caffeine) daily is an acceptable average amount of caffeine. More than 500 mg/d is considered excessive use of coffee.33,34
Overall mortality. A 2008 study showed that regular coffee was not associated with increased or decreased mortality in both men and women.35 However, more recent studies show an inverse relationship between mortality and coffee consumption.
Specifically, a 2014 meta-analysis found an inverse relationship between coffee and mortality.36 A large prospective cohort study from 2015 that included 79,234 women and 76,704 men found that drinking coffee was inversely associated with overall mortality.37 In this cohort study, an inverse association were observed for deaths from heart disease, respiratory disease, diabetes, and self-harm.37 While mechanisms were not analyzed, coffee may reduce mortality risk by affecting inflammation, lung function, insulin sensitivity, and depression.
Cardiovascular disease. Coffee consumption may modestly reduce the risk of stroke, according to a prospective cohort study of 83,076 women from the Nurses’ Health Study who were followed for 24 years.38 Reduced cardiovascular mortality was also found in a large prospective cohort study, as noted in the mortality discussion above.37 A 2014 meta-analysis concluded that coffee consumption is inversely associated with cardiovascular mortality. Drinking 3 or 4 cups a day appears to be the amount that may decrease one’s risk of death when compared to those who do not drink coffee at all.36
Liver disease. Friedrich et al performed a study involving 379 patients with end stage liver disease, and found that coffee consumption delayed the progression of disease in patients with both alcoholic liver disease and primary sclerosing cholangitis.39 Coffee consumption also increased long-term survival after liver transplantation.39 However, the study found that coffee did not have any effect on patients with chronic viral hepatitis.
In a 2016 meta-analysis, caffeinated coffee consumption reduced hepatic fibrosis of nonalcoholic fatty liver disease, although caffeine consumption did not reduce the prevalence of nonalcoholic fatty liver disease.40 Another meta-analysis, including 16 studies, also found caffeine reduced the risk for hepatic fibrosis and cirrhosis.41
Depression. Based on 2 different systematic reviews and meta-analyses from 2016, coffee consumption appears to have a significant protective effect, decreasing the risk of developing depression.40,42
Alzheimer’s disease/dementia. Coffee, tea, and caffeine consumption show promise in reducing the risk of cognitive decline and dementia. Individuals who consume one to 2 cups of coffee per day had a decreased incidence of mild cognitive impairment compared to non-drinkers.43 A 2015 Japanese study also found an inverse association between coffee consumption and dementia among women, nonsmokers, and those who do not drink alcohol.44 Most recently, a 2016 study, the Women’s Health Initiative Memory Study, looked at incident dementia rates in women >65 years of age with high vs low caffeine intake. Women with higher caffeine intake were less likely to develop dementia or any cognitive impairment compared with those consuming <64 mg/day.45
Type 2 diabetes. A 2009 prospective cohort study, which included 40,011 participants followed for more than 10 years, found that drinking at least 3 cups of coffee or tea was associated with a lowered risk of type 2 diabetes.46 A 2009 systematic review of 20 cohort studies showed that high intakes of coffee, decaffeinated coffee, and tea are associated with a reduced risk of diabetes.47
Melanoma.
Adverse effects
Despite the many potential benefits of coffee, caffeine is a potent drug that should be used with caution.49 People with underlying heart problems should avoid caffeine due to concern that it may cause palpitations from tachycardia. It may worsen anxiety problems or depression. Coffee may increase the production of stomach acids, which can worsen acid reflux or stomach ulcers.
Caffeine is a potent diuretic and may decrease absorption of calcium and cause OA. Caffeine may cause dependence and withdrawal symptoms. Some of the symptoms of withdrawal include drowsiness, headaches, irritability, nausea, and vomiting. It may disrupt sleeping patterns by causing jitters and sleeplessness.49 Additionally, large amounts of caffeine may cause overdose and death.
The bottom line
Regular coffee intake is associated with a lower risk of mortality, reduced cardiovascular events, and a reduction in liver disease progression. Coffee may also have some utility for improving cognitive function and reducing the risk of type 2 diabetes. Caffeinated coffee should be limited to no more than 32 oz per day, due to the risk of insomnia, palpitations, anxiety, and gastritis.
Chocolate/cocoa
Overview
Few natural products have been claimed to successfully treat as many disorders as chocolate. The modern concept of chocolate as food has overshadowed its traditional medicinal use, although recent trials have looked at evidence for some of its traditional uses. Chocolate is processed from the pod of the cacao plant. The earliest evidence for its medical use is in Mayan civilizations, and for most of its approximately 4000-year history, chocolate was consumed as a bitter drink referred to as the “drink of the Gods.” The traditional drink was mixed with water, vanilla, honey, chili peppers, and other spices. Important components in chocolate include flavonoids (antioxidants), cocoa butter, caffeine, theobromine, and phenylethylamine.
Chocolate has stimulating, anti-inflammatory, neuroprotective, and cardioprotective effects, and improves the bioavailability of nitric oxide, which can improve blood pressure and platelet function.50 Epicatechin (an antioxidant) in cocoa is primarily responsible for its favorable impact on vascular endothelium via its effect on both acute and chronic upregulation of nitric oxide production. Other cardiovascular effects are mediated by the anti-inflammatory effects of cocoa polyphenols, and modulated through the activity of NF-kappaB.51
Dark chocolate appears to have the greatest benefit, as milk binds to antioxidants in chocolate, making them unavailable. Therefore, milk chocolate is not a good antioxidant source. There is no specific amount of chocolate that is known to be ideal, but an average of one to 2 ounces per day is often used in studies.
Cardiovascular effects. Chocolate does contain saturated fat, but a comparative, double-blind study found that short-term use of cocoa powder lowered plasma low-density lipoprotein (LDL) cholesterol, oxidized LDL, and apo B concentrations, and the plasma high-density lipoprotein (HDL) cholesterol concentration increased, relative to baseline in the low-, middle-, and high-cocoa groups.52 A small randomized crossover trial without clinical outcomes indicated that chocolate may increase HDL cholesterol without increasing weight.53
A meta-analysis of short-term (2-12 weeks) treatment with dark chocolate/cocoa products showed reductions in LDL and total cholesterol, but no changes in HDL or triglycerides.54 Another meta-analysis of RCTs, however, showed no short-term effect of cocoa/chocolate on lipid concentrations.55 A randomized, placebo-controlled double-blind study of 62 patients with diabetes and hypertension showed that high polyphenol chocolate improved triglyceride levels.56
Multiple studies have shown that chocolate is associated with a reduction in cardiovascular risk.57-59 A best case scenario analysis using a Markov model to predict the long-term effectiveness and cost effectiveness of daily dark chocolate consumption in a population with metabolic syndrome at high risk of cardiovascular disease concluded that daily consumption of dark chocolate can reduce cardiovascular events by 85 per 10,000 population treated over 10 years. The study concluded that $42 could be cost effectively spent per person per year on prevention strategies using dark chocolate.59
In addition, a meta-analysis of 7 observational studies showed that high levels of chocolate consumption (any type) were associated with a 29% reduction in stroke compared with the lowest levels of chocolate intake.57 Results of a similar meta-analysis from Neurology in 2012 also suggested that moderate chocolate consumption (any type) may lower the risk of stroke.60
That said, 2 systematic reviews specifically relating to the risk of coronary heart disease and chocolate intake were inconclusive.61-62
Blood pressure (BP). An RCT published in JAMA indicates that inclusion of small amounts of polyphenol-rich dark chocolate as part of a usual diet efficiently reduced BP and improved the formation of vasodilative nitric oxide.63 A meta-analysis of 10 RCTs also showed mean BP change in the active cocoa treatment arms across all trials was -4.5 mm Hg (95% confidence interval (CI), -5.9 to -3.2; P<.001) for systolic BP and -2.5 mm Hg (95% CI, -3.9 to -1.2; P<.001) for diastolic BP.64
A Cochrane Review meta-analysis of 20 studies revealed a statistically significant BP-reducing effect of flavanol-rich cocoa products compared with control in short-term trials of 2 to 18 weeks' duration.65 Because studies have shown improvement in BP with chocolate intake, investigations into a role of chocolate in the prevention of preeclampsia have been undertaken. In some studies, chocolate intake was associated with reduced odds of preeclampsia and gestational hypertension.66,67
Diabetes. Chocolate may exert significant vascular protection because of its antioxidant properties and possible increase of nitric oxide bioavailability, which can influence glucose uptake. A small trial comparing the effects of either dark or white chocolate bars (which do not contain the polyphenols) showed improved BP and glucose and insulin responses to an oral glucose tolerance test in healthy subjects on dark chocolate, but not white chocolate.68 A comparison of chocolate consumption and risk of diabetes in the Physicians’ Health Study showed an inverse relationship between chocolate intake with incident disease, but this association appeared only to apply in younger and normal-body weight men after controlling for comprehensive lifestyles, including total energy consumption.69
Fatigue. The effect of chocolate on a person’s energy level has been noted for centuries.70 A small randomized trial showed improved energy levels in those treated with higher chocolate intakes. In a double-blind, randomized, clinical pilot crossover study, high cocoa liquor/polyphenol rich chocolate, reduced fatigue in subjects with chronic fatigue syndrome.71
Anxiety. A small randomized trial showed chocolate decreased anxiety in high-anxiety trait subjects and improved the anxiety level and the energy levels of low-anxiety trait participants.72
Eye effects. The literature presents conflicting evidence regarding the effect of flavonoids on patients with glaucoma and ocular hypertension. However, a meta-analysis showed that flavonoids have a promising role in improving visual function in patients with glaucoma and ocular hypertension, and appear to play a part in both improving and slowing the progression of visual field loss.73
Cognitive decline. Chocolate intake (any type) was associated with a lower risk of cognitive decline (RR = 0.59; 95% CI, 0.38-0.92) with the greatest benefit noted in those who averaged more than one chocolate bar or one tablespoon of cocoa powder per week. This protective effect was observed only among subjects with an average daily consumption of caffeine <75 mg (69% of the participants; RR = 0.50; 95% CI, 0.31-0.82).74
The bottom line
Chocolate with high cocoa content (dark chocolate) appears to be safe and beneficial as part of a healthy diet and lifestyle that includes exercise and stress reduction to decrease cardiovascular risk and may improve energy levels.
CORRESPONDENCE
Michael Malone, MD, Family and Community Medicine, Penn State College of Medicine, 500 University Drive, Hershey, PA 17033; [email protected].
1. National Center for Complementary and Integrative Health. The use of complementary and alternative medicine in the United States. Available at: https://nccih.nih.gov/research/statistics/2007/camsurvey_fs1.htm. Accessed Nov 28, 2017.
2. Aggarwal BB. Curcumin-free turmeric exhibits anti-inflammatory and anticancer activities: identification of novel components of turmeric. Mol Nutr Food Res. 2013;57:1529-1542.
3. Henrotin Y, Clutterbuck AL, Allaway D, et al. Biological actions of curcumin on articular chondrocytes. Osteoarthritis Cartilage. 2010;18:141-149.
4. Asher GN, Spelman K. Clinical utility of curcumin extract. Altern Ther Health Med. 2013;19:20-22.
5. Phan TT, See P, Lee ST, et al. Protective effects of curcumin against oxidative damage on skin cells in vitro: its implication for wound healing. J Trauma. 2001;51:927-931.
6. Funk JL, Frye JB, Oyarzo JN, et al. Efficacy and mechanism of action of turmeric supplements in the treatment of experimental arthritis. Arthritis Rheum. 2006;54:3452-3464.
7. Patcharatrakul T, Gonlachanvit S. Chili peppers, curcumins, and prebiotics in gastrointestinal health and disease. Curr Gastroenterol Rep. 2016;18:19.
8. Thamlikitkul V, Bunyapraphatsara N, Dechatiwongse T, et al. Randomized double blind study of Curcuma domestica Val. for dyspepsia. J Med Assoc Thai. 1989;72:613-620.
9. Kumar S, Ahuja V, Sankar MJ, et al. Curcumin for maintenance of remission in ulcerative colitis. Cochrane Database Syst Rev. 2012;10:CD008424.
10. Lang A, Salomon N, Wu JC, et al. Curcumin in combination with mesalamine induces remission in patients with mild-to-moderate ulcerative colitis in a randomized controlled trial. Clin Gastroenterol Hepatol. 2015;13:1444-1449.e1.
11. Henrotin Y, Priem F, Mobasheri A. Curcumin: a new paradigm and therapeutic opportunity for the treatment of osteoarthritis: curcumin for osteoarthritis management. Springerplus. 2013;2:56.
12. Kuptniratsaikul V, Dajpratham P, Taechaarpornkul W, et al. Efficacy and safety of Curcuma domestica extracts compared with ibuprofen in patients with knee osteoarthritis: a multicenter study. Clin Interv Aging. 2014;9:451-458.
13. Shehzad A, Lee J, Lee YS. Curcumin in various cancers. Biofactors. 2013;39:56-68.
14. Sordillo LA, Sordillo PP, Helson L. Curcumin for the treatment of glioblastoma. Anticancer Res. 2015;35:6373-6378.
15. Darvesh AS, Aggarwal BB, Bishayee A. Curcumin and liver cancer: a review. Curr Pharm Biotechnol. 2012;13:218-228.
16. Nagaraju GP, Aliya S, Zafar SF, et al. The impact of curcumin on breast cancer. Integr Biol (Camb). 2012;4:996-1007.
17. Johnson JJ, Mukhtar H. Curcumin for chemoprevention of colon cancer. Cancer Lett. 2007;255:170-181.
18. Dorai T, Cao YC, Dorai B, et al. Therapeutic potential of curcumin in human prostate cancer. III. Curcumin inhibits proliferation, induces apoptosis, and inhibits angiogenesis of LNCaP prostate cancer cells in vivo. Prostate. 2001;47:293-303.
19. Hazarey VK, Sakrikar AR, Ganvir SM. Efficacy of curcumin in the treatment for oral submucous fibrosis - a randomized clinical trial. J Oral Maxillofac Pathol. 2015;19:145-152.
20. Lal B, Kapoor AK, Asthana OP, et al. Efficacy of curcumin in the management of chronic anterior uveitis. Phytother Res. 1999;13:318-322.
21. Kapakos G, Youreva V, Srivastava AK. Cardiovascular protection by curcumin: molecular aspects. Indian J Biochem Biophys. 2012;49:306-315.
22. Yang F, Lim GP, Begum AN, et al. Curcumin inhibits formation of amyloid beta oligomers and fibrils, binds plaques, and reduces amyloid in vivo. J Biol Chem. 2005;280:5892-5901.
23. Heck AM, DeWitt BA, Lukes AL. Potential interactions between alternative therapies and warfarin. Am J Health Syst Pharm. 2000;57:1221-1227.
24. Srivastava JK, Shankar E, Gupta S. Chamomile: a herbal medicine of the past with bright future. Mol Med Rep. 2010;3:895-901.
25. Ross SM. Generalized anxiety disorder (GAD): efficacy of standardized matricaria recutita (german chamomile) extract in the treatment of generalized anxiety disorder. Holistic Nursing Practice. 2013;27:366- 368.
26. Amsterdam JD, Li Y, Soeller I, et al. A randomized, double-blind, placebo-controlled trial of oral Matricaria recutita (chamomile) extract therapy for generalized anxiety disorder. J Clin Psychopharmacol. 2009;29:378-382.
27. Zick SM, Wright BD, Sen A, et al. Preliminary examination of the efficacy and safety of a standardized chamomile extract for chronic primary insomnia: a randomized placebo-controlled pilot study. BMC Complement Altern Med. 2011;11:78.
28. Leach MJ, Page AT. Herbal medicine for insomnia: a systematic review and meta-analysis. Sleep Med Rev. 2015;24:1-12.
29. Weizman Z, Alkrinawi S, Goldfarb D, et al. Efficacy of herbal tea preparation in infantile colic. J Pediatr. 1993;122:650.
30. Crotteau CA, Wright ST, Eglash A. Clinical inquiries. What is the best treatment for infants with colic? J Fam Pract. 2006;55:634-636.
31. Panahi Y, Taghizadeh M, Marzony ET, et al. Rosemary oil vs minoxidil 2% for the treatment of androgenetic alopecia: a randomized comparative trial. Skinmed. 2015;13:15-21.
32. Hay IC, Jamieson M, Ormerod AD. Randomized trial of aromatherapy. Successful treatment for alopecia areata. Arch Dermatol. 1998;134:1349-1352.
33. Caffeine and kids: FDA takes a closer look. Available at: https://www.fda.gov/ForConsumers/ConsumerUpdates/ucm350570.htm. Accessed: November 1, 2017.
34. Torpy JM, Livingston EH. Energy Drinks. JAMA. 2013;309:297.
35. Lopez-Garcia E, van Dam RM, Li TY, et al. The relationship of coffee consumption with mortality. Ann Intern Med. 2008;148:904-914.
36. Crippa A, Discacciati A, Larsson SC, et al. Coffee consumption and mortality from all causes, cardiovascular disease, and cancer: a dose-response meta-analysis. Am J Epidemiol. 2014;180:763-775.
37. Loftfield E, Freedman ND, Graubard BI, et al. Association of coffee consumption with overall and cause-specific mortality in a large US prospective cohort study. Am J Epidemiol. 2015;182:1010-1022.
38. Lopez-Garcia E, Rodriguez-Artalejo F, Rexrode KM, et al. Coffee consumption and risk of stroke in women. Circulation. 2009;119:1116-1123.
39. Friedrich K, Smit M, Wannhoff A, et al. Coffee consumption protects against progression in liver cirrhosis and increases long-term survival after liver transplantation. J Gastroenterol Hepatol. 2016;31:1470-1475.
40. Wang L, Shen X, Wu Y, et al. Coffee and caffeine consumption and depression: a meta-analysis of observational studies. Aust N Z J Psychiatry. 2016;50:228-242.
41. Liu F, Wang X, Wu G, et al. Coffee consumption decreases risks for hepatic fibrosis and cirrhosis: a meta-analysis. PLoS One. 2015;10:e0142457.
42. Grosso G, Micek A, Castellano S, et al. Coffee, tea, caffeine and risk of depression: a systematic review and dose-response meta-analysis of observational studies. Mol Nutr Food Res. 2016;60:223-234.
43. Solfrizzi V, Panza F, Imbimbo BP, et al. Italian longitudinal study on aging working group. Coffee consumption habits and the risk of mild cognitive impairment: The Italian Longitudinal Study on Aging. J Alzheimers Dis. 2015;47:889-899.
44. Sugiyama K, Tomata Y, Kaiho Y, et al. Association between coffee consumption and incident risk of disabling dementia in elderly japanese: The Ohsaki Cohort 2006 Study. J Alzheimers Dis. 2015;50:491-500.
45. Driscoll I, Shumaker SA, Snively BM, et al. Relationships between caffeine intake and risk for probable dementia or global cognitive impairment: The Women’s Health Initiative Memory Study. J Gerontol A Biol Sci Med Sci. 2016;71:1596-1602.
46. van Dieren S, Uiterwaal CS, van der Schouw YT, et al. Coffee and tea consumption and risk of type 2 diabetes. Diabetologia. 2009;52:2561-2569.
47. Wang J, Li X, Zhang D. Coffee consumption and the risk of cutaneous melanoma: a meta-analysis. Eur J Nutr. 2016;55:1317-1329.
48. Liu J, Shen B, Shi M, et al. Higher caffeinated coffee intake is associated with reduced malignant melanoma risk: a meta-analysis study. PLoS One. 2016;11:e0147056.
49. Wikoff D, Welsh BT, Henderson R, et al. Systematic review of the potential adverse effects of caffeine consumption in healthy adults, pregnant women, adolescents, and children. Food Chem Toxical. 2017;109(Pt 1):585-648.
50. Verna R. The history and science of chocolate. Malays J Pathol. 2013;35:111-121.
51. Katz DL, Doughty K, Ali A. Cocoa and chocolate in human health and disease. Antioxid Redox Signal. 2011;15:2779-2811.
52. Baba S, Natsume M, Yasuda A, et al. Plasma LDL and HDL cholesterol and oxidized LDL concentrations are altered in normo- and hypercholesterolemic humans after intake of different levels of cocoa powder. J Nutr. 2007;137:1436-1441.
53. Mellor DD, Sathyapalan T, Kilpatrick ES, et al. High-cocoa polyphenol-rich chocolate improves HDL cholesterol in type 2 diabetes patients. Diabet Med. 2010;27:1318-1321.
54. Tokede OA, Gaziano JM, Djoussé L. Effects of cocoa products/dark chocolate on serum lipids: a meta-analysis. Eur J Clin Nutr. 2011;65:879-886.
55. Jia L, Liu X, Bai YY, et al. Short-term effect of cocoa product consumption on lipid profile: a meta-analysis of randomized controlled trials. Am J Clin Nutr. 2010;92:218-225.
56. Rostami A, Khalili M, Haghighat N, et al. High-cocoa polyphenol-rich chocolate improves blood pressure in patients with diabetes and hypertension. ARYA Atheroscler. 2015;11:21-29.
57. Buitrago-Lopez A, Sanderson J, Johnson L, et al. Chocolate consumption and cardiometabolic disorders: systematic review and meta-analysis. BMJ. 2011;26;343:d4488.
58. Wang X, Ouyang YY, Liu J, et al. Flavonoid intake and risk of CVD: a systematic review and meta-analysis of prospective cohort studies. Br J Nutr. 2014;111:1-11.
59. Zomer E, Owen A, Magliano DJ, et al. The effectiveness and cost effectiveness of dark chocolate consumption as prevention therapy in people at high risk of cardiovascular disease: best case scenario analysis using a Markov model. BMJ. 2012;344:e3657.
60. Larsson SC, Virtamo J, Wolk A. Chocolate consumption and risk of stroke: a prospective cohort of men and meta-analysis. Neurology. 2012;79:1223-1229.
61. Khawaja O, Gaziano JM, Djoussé L. Chocolate and coronary heart disease: a systematic review. Curr Atheroscler Rep. 2011;13:447-452.
62. Jacques PF, Cassidy A, Rogers G, et al. Dietary flavonoid intakes and CVD incidence in the Framingham Offspring Cohort. Br J Nutr. 2015;114:1496-1503.
63. Taubert D, Roesen R, Lehmann C, et al. Effects of low habitual cocoa intake on blood pressure and bioactive nitric oxide: a randomized controlled trial. JAMA. 2007;298:49-60.
64. Desch S, Schmidt J, Kobler D, et al. Effect of cocoa products on blood pressure: systematic review and meta-analysis. Am J Hypertens. 2010;23:97-103.
65. Ried K, Sullivan TR, Fakler P, et al. Effect of cocoa on blood pressure. Cochrane Database Syst Rev. 2012;8:CD008893.
66. Saftlas AF, Triche EW, Beydoun H, et al. Does chocolate intake during pregnancy reduce the risks of preeclampsia and gestational hypertension? Ann Epidemiol. 2010;20:584-591.
67. Triche EW, Grosso LM, Belanger K, et al. Chocolate consumption in pregnancy and reduced likelihood of preeclampsia. Epidemiology. 2008;19:459-464.
68. Grassi D, Lippi C, Necozione S, et al. Short-term administration of dark chocolate is followed by a significant increase in insulin sensitivity and a decrease in blood pressure in healthy persons. Am J Clin Nutr. 2005;81:611-614.
69. Matsumoto C, Petrone AB, Sesso HD, et al. Chocolate consumption and risk of diabetes mellitus in the Physicians’ Health Study. Am J Clin Nutr. 2015;101:362-367.
70. Lippi D. Chocolate in history: food, medicine, medi-food. Nutrients. 2013;5:1573-1584.
71. Sathyapalan T, Beckett S, Rigby AS, et al. High cocoa polyphenol rich chocolate may reduce the burden of the symptoms in chronic fatigue syndrome. Nutr J. 2010;9:55.
72. Martin FP, Antille N, Rezzi S, et al. Everyday eating experiences of chocolate and non-chocolate snacks impact postprandial anxiety, energy and emotional states. Nutrients. 2012;4:554-567.
73. Patel S, Mathan JJ, Vaghefi E, et al. The effect of flavonoids on visual function in patients with glaucoma or ocular hypertension: a systematic review and meta-analysis. Graefes Arch Clin Exp Ophthalmol. 2015;253:1841-1850.
74. Moreira A, Diógenes MJ, de Mendonça A, et al. Chocolate consumption is associated with a lower risk of cognitive decline. J Alzheimers Dis. 2016;53:85-93.
1. National Center for Complementary and Integrative Health. The use of complementary and alternative medicine in the United States. Available at: https://nccih.nih.gov/research/statistics/2007/camsurvey_fs1.htm. Accessed Nov 28, 2017.
2. Aggarwal BB. Curcumin-free turmeric exhibits anti-inflammatory and anticancer activities: identification of novel components of turmeric. Mol Nutr Food Res. 2013;57:1529-1542.
3. Henrotin Y, Clutterbuck AL, Allaway D, et al. Biological actions of curcumin on articular chondrocytes. Osteoarthritis Cartilage. 2010;18:141-149.
4. Asher GN, Spelman K. Clinical utility of curcumin extract. Altern Ther Health Med. 2013;19:20-22.
5. Phan TT, See P, Lee ST, et al. Protective effects of curcumin against oxidative damage on skin cells in vitro: its implication for wound healing. J Trauma. 2001;51:927-931.
6. Funk JL, Frye JB, Oyarzo JN, et al. Efficacy and mechanism of action of turmeric supplements in the treatment of experimental arthritis. Arthritis Rheum. 2006;54:3452-3464.
7. Patcharatrakul T, Gonlachanvit S. Chili peppers, curcumins, and prebiotics in gastrointestinal health and disease. Curr Gastroenterol Rep. 2016;18:19.
8. Thamlikitkul V, Bunyapraphatsara N, Dechatiwongse T, et al. Randomized double blind study of Curcuma domestica Val. for dyspepsia. J Med Assoc Thai. 1989;72:613-620.
9. Kumar S, Ahuja V, Sankar MJ, et al. Curcumin for maintenance of remission in ulcerative colitis. Cochrane Database Syst Rev. 2012;10:CD008424.
10. Lang A, Salomon N, Wu JC, et al. Curcumin in combination with mesalamine induces remission in patients with mild-to-moderate ulcerative colitis in a randomized controlled trial. Clin Gastroenterol Hepatol. 2015;13:1444-1449.e1.
11. Henrotin Y, Priem F, Mobasheri A. Curcumin: a new paradigm and therapeutic opportunity for the treatment of osteoarthritis: curcumin for osteoarthritis management. Springerplus. 2013;2:56.
12. Kuptniratsaikul V, Dajpratham P, Taechaarpornkul W, et al. Efficacy and safety of Curcuma domestica extracts compared with ibuprofen in patients with knee osteoarthritis: a multicenter study. Clin Interv Aging. 2014;9:451-458.
13. Shehzad A, Lee J, Lee YS. Curcumin in various cancers. Biofactors. 2013;39:56-68.
14. Sordillo LA, Sordillo PP, Helson L. Curcumin for the treatment of glioblastoma. Anticancer Res. 2015;35:6373-6378.
15. Darvesh AS, Aggarwal BB, Bishayee A. Curcumin and liver cancer: a review. Curr Pharm Biotechnol. 2012;13:218-228.
16. Nagaraju GP, Aliya S, Zafar SF, et al. The impact of curcumin on breast cancer. Integr Biol (Camb). 2012;4:996-1007.
17. Johnson JJ, Mukhtar H. Curcumin for chemoprevention of colon cancer. Cancer Lett. 2007;255:170-181.
18. Dorai T, Cao YC, Dorai B, et al. Therapeutic potential of curcumin in human prostate cancer. III. Curcumin inhibits proliferation, induces apoptosis, and inhibits angiogenesis of LNCaP prostate cancer cells in vivo. Prostate. 2001;47:293-303.
19. Hazarey VK, Sakrikar AR, Ganvir SM. Efficacy of curcumin in the treatment for oral submucous fibrosis - a randomized clinical trial. J Oral Maxillofac Pathol. 2015;19:145-152.
20. Lal B, Kapoor AK, Asthana OP, et al. Efficacy of curcumin in the management of chronic anterior uveitis. Phytother Res. 1999;13:318-322.
21. Kapakos G, Youreva V, Srivastava AK. Cardiovascular protection by curcumin: molecular aspects. Indian J Biochem Biophys. 2012;49:306-315.
22. Yang F, Lim GP, Begum AN, et al. Curcumin inhibits formation of amyloid beta oligomers and fibrils, binds plaques, and reduces amyloid in vivo. J Biol Chem. 2005;280:5892-5901.
23. Heck AM, DeWitt BA, Lukes AL. Potential interactions between alternative therapies and warfarin. Am J Health Syst Pharm. 2000;57:1221-1227.
24. Srivastava JK, Shankar E, Gupta S. Chamomile: a herbal medicine of the past with bright future. Mol Med Rep. 2010;3:895-901.
25. Ross SM. Generalized anxiety disorder (GAD): efficacy of standardized matricaria recutita (german chamomile) extract in the treatment of generalized anxiety disorder. Holistic Nursing Practice. 2013;27:366- 368.
26. Amsterdam JD, Li Y, Soeller I, et al. A randomized, double-blind, placebo-controlled trial of oral Matricaria recutita (chamomile) extract therapy for generalized anxiety disorder. J Clin Psychopharmacol. 2009;29:378-382.
27. Zick SM, Wright BD, Sen A, et al. Preliminary examination of the efficacy and safety of a standardized chamomile extract for chronic primary insomnia: a randomized placebo-controlled pilot study. BMC Complement Altern Med. 2011;11:78.
28. Leach MJ, Page AT. Herbal medicine for insomnia: a systematic review and meta-analysis. Sleep Med Rev. 2015;24:1-12.
29. Weizman Z, Alkrinawi S, Goldfarb D, et al. Efficacy of herbal tea preparation in infantile colic. J Pediatr. 1993;122:650.
30. Crotteau CA, Wright ST, Eglash A. Clinical inquiries. What is the best treatment for infants with colic? J Fam Pract. 2006;55:634-636.
31. Panahi Y, Taghizadeh M, Marzony ET, et al. Rosemary oil vs minoxidil 2% for the treatment of androgenetic alopecia: a randomized comparative trial. Skinmed. 2015;13:15-21.
32. Hay IC, Jamieson M, Ormerod AD. Randomized trial of aromatherapy. Successful treatment for alopecia areata. Arch Dermatol. 1998;134:1349-1352.
33. Caffeine and kids: FDA takes a closer look. Available at: https://www.fda.gov/ForConsumers/ConsumerUpdates/ucm350570.htm. Accessed: November 1, 2017.
34. Torpy JM, Livingston EH. Energy Drinks. JAMA. 2013;309:297.
35. Lopez-Garcia E, van Dam RM, Li TY, et al. The relationship of coffee consumption with mortality. Ann Intern Med. 2008;148:904-914.
36. Crippa A, Discacciati A, Larsson SC, et al. Coffee consumption and mortality from all causes, cardiovascular disease, and cancer: a dose-response meta-analysis. Am J Epidemiol. 2014;180:763-775.
37. Loftfield E, Freedman ND, Graubard BI, et al. Association of coffee consumption with overall and cause-specific mortality in a large US prospective cohort study. Am J Epidemiol. 2015;182:1010-1022.
38. Lopez-Garcia E, Rodriguez-Artalejo F, Rexrode KM, et al. Coffee consumption and risk of stroke in women. Circulation. 2009;119:1116-1123.
39. Friedrich K, Smit M, Wannhoff A, et al. Coffee consumption protects against progression in liver cirrhosis and increases long-term survival after liver transplantation. J Gastroenterol Hepatol. 2016;31:1470-1475.
40. Wang L, Shen X, Wu Y, et al. Coffee and caffeine consumption and depression: a meta-analysis of observational studies. Aust N Z J Psychiatry. 2016;50:228-242.
41. Liu F, Wang X, Wu G, et al. Coffee consumption decreases risks for hepatic fibrosis and cirrhosis: a meta-analysis. PLoS One. 2015;10:e0142457.
42. Grosso G, Micek A, Castellano S, et al. Coffee, tea, caffeine and risk of depression: a systematic review and dose-response meta-analysis of observational studies. Mol Nutr Food Res. 2016;60:223-234.
43. Solfrizzi V, Panza F, Imbimbo BP, et al. Italian longitudinal study on aging working group. Coffee consumption habits and the risk of mild cognitive impairment: The Italian Longitudinal Study on Aging. J Alzheimers Dis. 2015;47:889-899.
44. Sugiyama K, Tomata Y, Kaiho Y, et al. Association between coffee consumption and incident risk of disabling dementia in elderly japanese: The Ohsaki Cohort 2006 Study. J Alzheimers Dis. 2015;50:491-500.
45. Driscoll I, Shumaker SA, Snively BM, et al. Relationships between caffeine intake and risk for probable dementia or global cognitive impairment: The Women’s Health Initiative Memory Study. J Gerontol A Biol Sci Med Sci. 2016;71:1596-1602.
46. van Dieren S, Uiterwaal CS, van der Schouw YT, et al. Coffee and tea consumption and risk of type 2 diabetes. Diabetologia. 2009;52:2561-2569.
47. Wang J, Li X, Zhang D. Coffee consumption and the risk of cutaneous melanoma: a meta-analysis. Eur J Nutr. 2016;55:1317-1329.
48. Liu J, Shen B, Shi M, et al. Higher caffeinated coffee intake is associated with reduced malignant melanoma risk: a meta-analysis study. PLoS One. 2016;11:e0147056.
49. Wikoff D, Welsh BT, Henderson R, et al. Systematic review of the potential adverse effects of caffeine consumption in healthy adults, pregnant women, adolescents, and children. Food Chem Toxical. 2017;109(Pt 1):585-648.
50. Verna R. The history and science of chocolate. Malays J Pathol. 2013;35:111-121.
51. Katz DL, Doughty K, Ali A. Cocoa and chocolate in human health and disease. Antioxid Redox Signal. 2011;15:2779-2811.
52. Baba S, Natsume M, Yasuda A, et al. Plasma LDL and HDL cholesterol and oxidized LDL concentrations are altered in normo- and hypercholesterolemic humans after intake of different levels of cocoa powder. J Nutr. 2007;137:1436-1441.
53. Mellor DD, Sathyapalan T, Kilpatrick ES, et al. High-cocoa polyphenol-rich chocolate improves HDL cholesterol in type 2 diabetes patients. Diabet Med. 2010;27:1318-1321.
54. Tokede OA, Gaziano JM, Djoussé L. Effects of cocoa products/dark chocolate on serum lipids: a meta-analysis. Eur J Clin Nutr. 2011;65:879-886.
55. Jia L, Liu X, Bai YY, et al. Short-term effect of cocoa product consumption on lipid profile: a meta-analysis of randomized controlled trials. Am J Clin Nutr. 2010;92:218-225.
56. Rostami A, Khalili M, Haghighat N, et al. High-cocoa polyphenol-rich chocolate improves blood pressure in patients with diabetes and hypertension. ARYA Atheroscler. 2015;11:21-29.
57. Buitrago-Lopez A, Sanderson J, Johnson L, et al. Chocolate consumption and cardiometabolic disorders: systematic review and meta-analysis. BMJ. 2011;26;343:d4488.
58. Wang X, Ouyang YY, Liu J, et al. Flavonoid intake and risk of CVD: a systematic review and meta-analysis of prospective cohort studies. Br J Nutr. 2014;111:1-11.
59. Zomer E, Owen A, Magliano DJ, et al. The effectiveness and cost effectiveness of dark chocolate consumption as prevention therapy in people at high risk of cardiovascular disease: best case scenario analysis using a Markov model. BMJ. 2012;344:e3657.
60. Larsson SC, Virtamo J, Wolk A. Chocolate consumption and risk of stroke: a prospective cohort of men and meta-analysis. Neurology. 2012;79:1223-1229.
61. Khawaja O, Gaziano JM, Djoussé L. Chocolate and coronary heart disease: a systematic review. Curr Atheroscler Rep. 2011;13:447-452.
62. Jacques PF, Cassidy A, Rogers G, et al. Dietary flavonoid intakes and CVD incidence in the Framingham Offspring Cohort. Br J Nutr. 2015;114:1496-1503.
63. Taubert D, Roesen R, Lehmann C, et al. Effects of low habitual cocoa intake on blood pressure and bioactive nitric oxide: a randomized controlled trial. JAMA. 2007;298:49-60.
64. Desch S, Schmidt J, Kobler D, et al. Effect of cocoa products on blood pressure: systematic review and meta-analysis. Am J Hypertens. 2010;23:97-103.
65. Ried K, Sullivan TR, Fakler P, et al. Effect of cocoa on blood pressure. Cochrane Database Syst Rev. 2012;8:CD008893.
66. Saftlas AF, Triche EW, Beydoun H, et al. Does chocolate intake during pregnancy reduce the risks of preeclampsia and gestational hypertension? Ann Epidemiol. 2010;20:584-591.
67. Triche EW, Grosso LM, Belanger K, et al. Chocolate consumption in pregnancy and reduced likelihood of preeclampsia. Epidemiology. 2008;19:459-464.
68. Grassi D, Lippi C, Necozione S, et al. Short-term administration of dark chocolate is followed by a significant increase in insulin sensitivity and a decrease in blood pressure in healthy persons. Am J Clin Nutr. 2005;81:611-614.
69. Matsumoto C, Petrone AB, Sesso HD, et al. Chocolate consumption and risk of diabetes mellitus in the Physicians’ Health Study. Am J Clin Nutr. 2015;101:362-367.
70. Lippi D. Chocolate in history: food, medicine, medi-food. Nutrients. 2013;5:1573-1584.
71. Sathyapalan T, Beckett S, Rigby AS, et al. High cocoa polyphenol rich chocolate may reduce the burden of the symptoms in chronic fatigue syndrome. Nutr J. 2010;9:55.
72. Martin FP, Antille N, Rezzi S, et al. Everyday eating experiences of chocolate and non-chocolate snacks impact postprandial anxiety, energy and emotional states. Nutrients. 2012;4:554-567.
73. Patel S, Mathan JJ, Vaghefi E, et al. The effect of flavonoids on visual function in patients with glaucoma or ocular hypertension: a systematic review and meta-analysis. Graefes Arch Clin Exp Ophthalmol. 2015;253:1841-1850.
74. Moreira A, Diógenes MJ, de Mendonça A, et al. Chocolate consumption is associated with a lower risk of cognitive decline. J Alzheimers Dis. 2016;53:85-93.
PRACTICE RECOMMENDATIONS
› Inform patients that curcumin appears to be a safe and effective adjunctive therapy for ulcerative colitis when used along with mesalamine or sulfasalazine. B
› Recommend chamomile extract to patients experiencing mild to moderate generalized anxiety disorder. B
› Tell patients that coffee is associated with a lower risk of mortality, reduced cardiovascular events, and a reduction in liver disease progression (in patients with end-stage liver disease). B
Strength of recommendation (SOR)
A Good-quality patient-oriented evidence
B Inconsistent or limited-quality patient-oriented evidence
C Consensus, usual practice, opinion, disease-oriented evidence, case series