User login
Cirrhosis, Child-Pugh score predict ERCP complications
Cirrhosis may increase the risk of complications from endoscopic retrograde cholangiopancreatography (ERCP), according to a retrospective study involving almost 700 patients.
The study also showed that Child-Pugh class was a better predictor of risk than Model for End-Stage Liver Disease (MELD) score, reported lead author Michelle Bernshteyn, MD, a third-year internal medicine resident at State University of New York, Syracuse , and colleagues.
“There remains a scarcity in the literature regarding complications and adverse effects after ERCP in cirrhotic patients, particularly those incorporating Child-Pugh class and MELD score or type of intervention as predictors,” Dr. Bernshteyn said during a virtual presentation at the American College of Gastroenterology annual meeting. “Furthermore, literature review demonstrates inconsistency among results.”
To gain clarity, Dr. Bernshteyn and colleagues reviewed electronic medical records from 692 patients who underwent ERCP, of whom 174 had cirrhosis and 518 did not. For all patients, the investigators analyzed demographics, comorbidities, indications for ERCP, type of sedation, type of intervention, and complications within a 30-day period. Complications included bleeding, pancreatitis, cholangitis, perforation, mortality caused by ERCP, and mortality from other causes. Patients with cirrhosis were further analyzed based on etiology of cirrhosis, Child-Pugh class, and MELD score.
The analysis revealed that complications were significantly more common in patients with cirrhosis than in those without cirrhosis (21.30% vs. 13.51%; P = .015). No specific complications were significantly more common in patients with cirrhosis than in those without cirrhosis.
In patients with cirrhosis, 41.18% of Child-Pugh class C patients had complications, compared with 15.15% of class B patients and 19.30% of class A patients (P = .010). In contrast, MELD scores were not significantly associated with adverse events.
Further analysis showed that, in patients without cirrhosis, diagnostic-only ERCP and underlying chronic obstructive pulmonary disease were associated with high rates of complications (P = .039 and P = .003, respectively). In patients with cirrhosis, underlying chronic obstructive pulmonary disease and hypertension predicted adverse events (P = .009 and P = .003, respectively).
“The results of our study reaffirm that liver cirrhosis has an impact on the occurrence of complications during ERCP,” Dr. Bernshteyn said. “Child-Pugh class seems to be more reliable as compared to MELD score in predicting complications of ERCP in cirrhosis patients,” she added. “However, we are also aware that Child-Pugh and MELD scores are complementary to each other while evaluating outcomes of any surgery in patients with cirrhosis.”
In 2017, Udayakumar Navaneethan, MD, a gastroenterologist at AdventHealth Orlando’s Center for Interventional Endoscopy, and an assistant professor at the University of Central Florida, Orlando, and colleagues published a national database study concerning the safety of ERCP in patients with liver cirrhosis.
“[The present] study is important as it highlights the fact that ERCP is associated with significant complications in cirrhotic patients compared to those without cirrhosis,” Dr. Navaneethan said when asked to comment. “Also, Child-Pugh score appeared to be more reliable than MELD score in predicting complications of ERCP in cirrhotic patients.”
He went on to explain relevance for practicing clinicians. “The clinical implications of the study are that a detailed risk-benefit discussion needs to be done with patients with liver cirrhosis, particularly with advanced liver disease Child-Pugh class C, irrespective of the etiology,” Dr. Navaneethan said. “ERCP should be performed when there is clear evidence that the benefits outweigh the risks.” The investigators and Dr. Navaneethan reported no conflicts of interest.
SOURCE: Bernshteyn M et al. ACG 2020, Abstract S0982.
Cirrhosis may increase the risk of complications from endoscopic retrograde cholangiopancreatography (ERCP), according to a retrospective study involving almost 700 patients.
The study also showed that Child-Pugh class was a better predictor of risk than Model for End-Stage Liver Disease (MELD) score, reported lead author Michelle Bernshteyn, MD, a third-year internal medicine resident at State University of New York, Syracuse , and colleagues.
“There remains a scarcity in the literature regarding complications and adverse effects after ERCP in cirrhotic patients, particularly those incorporating Child-Pugh class and MELD score or type of intervention as predictors,” Dr. Bernshteyn said during a virtual presentation at the American College of Gastroenterology annual meeting. “Furthermore, literature review demonstrates inconsistency among results.”
To gain clarity, Dr. Bernshteyn and colleagues reviewed electronic medical records from 692 patients who underwent ERCP, of whom 174 had cirrhosis and 518 did not. For all patients, the investigators analyzed demographics, comorbidities, indications for ERCP, type of sedation, type of intervention, and complications within a 30-day period. Complications included bleeding, pancreatitis, cholangitis, perforation, mortality caused by ERCP, and mortality from other causes. Patients with cirrhosis were further analyzed based on etiology of cirrhosis, Child-Pugh class, and MELD score.
The analysis revealed that complications were significantly more common in patients with cirrhosis than in those without cirrhosis (21.30% vs. 13.51%; P = .015). No specific complications were significantly more common in patients with cirrhosis than in those without cirrhosis.
In patients with cirrhosis, 41.18% of Child-Pugh class C patients had complications, compared with 15.15% of class B patients and 19.30% of class A patients (P = .010). In contrast, MELD scores were not significantly associated with adverse events.
Further analysis showed that, in patients without cirrhosis, diagnostic-only ERCP and underlying chronic obstructive pulmonary disease were associated with high rates of complications (P = .039 and P = .003, respectively). In patients with cirrhosis, underlying chronic obstructive pulmonary disease and hypertension predicted adverse events (P = .009 and P = .003, respectively).
“The results of our study reaffirm that liver cirrhosis has an impact on the occurrence of complications during ERCP,” Dr. Bernshteyn said. “Child-Pugh class seems to be more reliable as compared to MELD score in predicting complications of ERCP in cirrhosis patients,” she added. “However, we are also aware that Child-Pugh and MELD scores are complementary to each other while evaluating outcomes of any surgery in patients with cirrhosis.”
In 2017, Udayakumar Navaneethan, MD, a gastroenterologist at AdventHealth Orlando’s Center for Interventional Endoscopy, and an assistant professor at the University of Central Florida, Orlando, and colleagues published a national database study concerning the safety of ERCP in patients with liver cirrhosis.
“[The present] study is important as it highlights the fact that ERCP is associated with significant complications in cirrhotic patients compared to those without cirrhosis,” Dr. Navaneethan said when asked to comment. “Also, Child-Pugh score appeared to be more reliable than MELD score in predicting complications of ERCP in cirrhotic patients.”
He went on to explain relevance for practicing clinicians. “The clinical implications of the study are that a detailed risk-benefit discussion needs to be done with patients with liver cirrhosis, particularly with advanced liver disease Child-Pugh class C, irrespective of the etiology,” Dr. Navaneethan said. “ERCP should be performed when there is clear evidence that the benefits outweigh the risks.” The investigators and Dr. Navaneethan reported no conflicts of interest.
SOURCE: Bernshteyn M et al. ACG 2020, Abstract S0982.
Cirrhosis may increase the risk of complications from endoscopic retrograde cholangiopancreatography (ERCP), according to a retrospective study involving almost 700 patients.
The study also showed that Child-Pugh class was a better predictor of risk than Model for End-Stage Liver Disease (MELD) score, reported lead author Michelle Bernshteyn, MD, a third-year internal medicine resident at State University of New York, Syracuse , and colleagues.
“There remains a scarcity in the literature regarding complications and adverse effects after ERCP in cirrhotic patients, particularly those incorporating Child-Pugh class and MELD score or type of intervention as predictors,” Dr. Bernshteyn said during a virtual presentation at the American College of Gastroenterology annual meeting. “Furthermore, literature review demonstrates inconsistency among results.”
To gain clarity, Dr. Bernshteyn and colleagues reviewed electronic medical records from 692 patients who underwent ERCP, of whom 174 had cirrhosis and 518 did not. For all patients, the investigators analyzed demographics, comorbidities, indications for ERCP, type of sedation, type of intervention, and complications within a 30-day period. Complications included bleeding, pancreatitis, cholangitis, perforation, mortality caused by ERCP, and mortality from other causes. Patients with cirrhosis were further analyzed based on etiology of cirrhosis, Child-Pugh class, and MELD score.
The analysis revealed that complications were significantly more common in patients with cirrhosis than in those without cirrhosis (21.30% vs. 13.51%; P = .015). No specific complications were significantly more common in patients with cirrhosis than in those without cirrhosis.
In patients with cirrhosis, 41.18% of Child-Pugh class C patients had complications, compared with 15.15% of class B patients and 19.30% of class A patients (P = .010). In contrast, MELD scores were not significantly associated with adverse events.
Further analysis showed that, in patients without cirrhosis, diagnostic-only ERCP and underlying chronic obstructive pulmonary disease were associated with high rates of complications (P = .039 and P = .003, respectively). In patients with cirrhosis, underlying chronic obstructive pulmonary disease and hypertension predicted adverse events (P = .009 and P = .003, respectively).
“The results of our study reaffirm that liver cirrhosis has an impact on the occurrence of complications during ERCP,” Dr. Bernshteyn said. “Child-Pugh class seems to be more reliable as compared to MELD score in predicting complications of ERCP in cirrhosis patients,” she added. “However, we are also aware that Child-Pugh and MELD scores are complementary to each other while evaluating outcomes of any surgery in patients with cirrhosis.”
In 2017, Udayakumar Navaneethan, MD, a gastroenterologist at AdventHealth Orlando’s Center for Interventional Endoscopy, and an assistant professor at the University of Central Florida, Orlando, and colleagues published a national database study concerning the safety of ERCP in patients with liver cirrhosis.
“[The present] study is important as it highlights the fact that ERCP is associated with significant complications in cirrhotic patients compared to those without cirrhosis,” Dr. Navaneethan said when asked to comment. “Also, Child-Pugh score appeared to be more reliable than MELD score in predicting complications of ERCP in cirrhotic patients.”
He went on to explain relevance for practicing clinicians. “The clinical implications of the study are that a detailed risk-benefit discussion needs to be done with patients with liver cirrhosis, particularly with advanced liver disease Child-Pugh class C, irrespective of the etiology,” Dr. Navaneethan said. “ERCP should be performed when there is clear evidence that the benefits outweigh the risks.” The investigators and Dr. Navaneethan reported no conflicts of interest.
SOURCE: Bernshteyn M et al. ACG 2020, Abstract S0982.
FROM ACG 2020
Preemptive CMV monitoring beats prophylaxis post liver transplant
Preemptive monitoring and treatment of cytomegalovirus infections in CMV-seronegative liver transplant recipients who receive organs from CMV-positive donors appears to be better at preventing infections than a viral prophylaxis strategy, according to infectious disease and organ transplant specialists.
In a study published in JAMA that may have gotten scant notice because of its publication during the early days of the COVID-19 pandemic, investigators at the University of Pittsburgh and other transplant centers reported results of a randomized clinical trial comparing the two CMV management strategies, and found that the incidence of CMV disease was significantly lower for patients who were started on valganciclovir when asymptomatic CMV viremia was detected, compared with patients on antiviral prophylaxis with valganciclovir.
The study “is a significant game changer for the field of transplantation,” commented Michael G. Ison, MD, professor of infectious diseases and organ transplantation at Northwestern University, Chicago.
Dr. Ison discussed the study and its implications during a session on potentially practice-changing clinical trials presented virtually during IDWeek 2020, an annual scientific meeting on infectious diseases.
In the trial, Nina Singh, MD, and colleagues randomly assigned 100 CMV-seronegative liver transplant recipients to receive preemptive therapy, in which patients underwent weekly testing for 100 days with a highly sensitive real-time plasma polymerase chain reaction assay for CMV. If viremia at any level was detected, the patients received oral valganciclovir 900 mg twice daily until two consecutive tests performed 1 week apart came back negative.
The remaining 105 patients were randomly assigned to 100 days of oral prophylaxis with 900 mg valganciclovir twice daily, started within 10 days of transplant.
CMV disease incidence lower
The incidence of CMV disease within 12 months of transplants, the primary outcome, was 9% in the preemptive therapy group, compared with 19% in the prophylaxis group (P = .04)
The difference between the groups was largely accounted for by a reduction in disease onset beyond 100 days in the preemptive therapy group (6% vs. 17%, respectively, P = .01)
There were no significant differences in secondary endpoints of rejection, opportunistic infections, graft loss because of retransplantation, neutropenia, or receipt of one or more doses of granulocyte colony–stimulating factor for the management of neutropenia.
At 1-year follow-up, the incidence of all-cause mortality was 15% in the preemptive therapy group, and 19% in the prophylaxis group; the difference was not statistically significant.
“While most transplant centers utilize universal prophylaxis, I think that this study really suggests that preemptive monitoring, if it can be safely accomplished at your center, may be of the greatest benefit to your patients,” Dr. Ison said.
He noted that Singh et al. also looked in an exploratory analysis at CMV-specific immunity and observed that patients assigned to preemptive therapy “clearly had better CMV-specific immunity, whether CD4 or CD8 cells, and had higher lymphocyte numbers than those patients that had received universal prophylaxis.”
In a comment, Sarah Doernberg, MD, from the division of infectious diseases at the University of California, San Francisco, agreed that “exploratory analysis of CMV-specific immune responses suggested increased CMV-specific immunity in those in the preemptive group, a finding that warrants further study. The feasibility of adopting reliable preemptive monitoring must be considered as individual centers ponder adopting this approach.”
Dr. Doernberg moderated the session where Dr. Ison discussed the data, but was not involved in the research.
The study by Singh et al. was supported by the National Institutes of Health. Dr. Singh reported research grants from NIH. Dr. Ison disclosed research support and paid consultation for several companies. Dr. Doernberg disclosed consulting for Basilea and Genentech.
Preemptive monitoring and treatment of cytomegalovirus infections in CMV-seronegative liver transplant recipients who receive organs from CMV-positive donors appears to be better at preventing infections than a viral prophylaxis strategy, according to infectious disease and organ transplant specialists.
In a study published in JAMA that may have gotten scant notice because of its publication during the early days of the COVID-19 pandemic, investigators at the University of Pittsburgh and other transplant centers reported results of a randomized clinical trial comparing the two CMV management strategies, and found that the incidence of CMV disease was significantly lower for patients who were started on valganciclovir when asymptomatic CMV viremia was detected, compared with patients on antiviral prophylaxis with valganciclovir.
The study “is a significant game changer for the field of transplantation,” commented Michael G. Ison, MD, professor of infectious diseases and organ transplantation at Northwestern University, Chicago.
Dr. Ison discussed the study and its implications during a session on potentially practice-changing clinical trials presented virtually during IDWeek 2020, an annual scientific meeting on infectious diseases.
In the trial, Nina Singh, MD, and colleagues randomly assigned 100 CMV-seronegative liver transplant recipients to receive preemptive therapy, in which patients underwent weekly testing for 100 days with a highly sensitive real-time plasma polymerase chain reaction assay for CMV. If viremia at any level was detected, the patients received oral valganciclovir 900 mg twice daily until two consecutive tests performed 1 week apart came back negative.
The remaining 105 patients were randomly assigned to 100 days of oral prophylaxis with 900 mg valganciclovir twice daily, started within 10 days of transplant.
CMV disease incidence lower
The incidence of CMV disease within 12 months of transplants, the primary outcome, was 9% in the preemptive therapy group, compared with 19% in the prophylaxis group (P = .04)
The difference between the groups was largely accounted for by a reduction in disease onset beyond 100 days in the preemptive therapy group (6% vs. 17%, respectively, P = .01)
There were no significant differences in secondary endpoints of rejection, opportunistic infections, graft loss because of retransplantation, neutropenia, or receipt of one or more doses of granulocyte colony–stimulating factor for the management of neutropenia.
At 1-year follow-up, the incidence of all-cause mortality was 15% in the preemptive therapy group, and 19% in the prophylaxis group; the difference was not statistically significant.
“While most transplant centers utilize universal prophylaxis, I think that this study really suggests that preemptive monitoring, if it can be safely accomplished at your center, may be of the greatest benefit to your patients,” Dr. Ison said.
He noted that Singh et al. also looked in an exploratory analysis at CMV-specific immunity and observed that patients assigned to preemptive therapy “clearly had better CMV-specific immunity, whether CD4 or CD8 cells, and had higher lymphocyte numbers than those patients that had received universal prophylaxis.”
In a comment, Sarah Doernberg, MD, from the division of infectious diseases at the University of California, San Francisco, agreed that “exploratory analysis of CMV-specific immune responses suggested increased CMV-specific immunity in those in the preemptive group, a finding that warrants further study. The feasibility of adopting reliable preemptive monitoring must be considered as individual centers ponder adopting this approach.”
Dr. Doernberg moderated the session where Dr. Ison discussed the data, but was not involved in the research.
The study by Singh et al. was supported by the National Institutes of Health. Dr. Singh reported research grants from NIH. Dr. Ison disclosed research support and paid consultation for several companies. Dr. Doernberg disclosed consulting for Basilea and Genentech.
Preemptive monitoring and treatment of cytomegalovirus infections in CMV-seronegative liver transplant recipients who receive organs from CMV-positive donors appears to be better at preventing infections than a viral prophylaxis strategy, according to infectious disease and organ transplant specialists.
In a study published in JAMA that may have gotten scant notice because of its publication during the early days of the COVID-19 pandemic, investigators at the University of Pittsburgh and other transplant centers reported results of a randomized clinical trial comparing the two CMV management strategies, and found that the incidence of CMV disease was significantly lower for patients who were started on valganciclovir when asymptomatic CMV viremia was detected, compared with patients on antiviral prophylaxis with valganciclovir.
The study “is a significant game changer for the field of transplantation,” commented Michael G. Ison, MD, professor of infectious diseases and organ transplantation at Northwestern University, Chicago.
Dr. Ison discussed the study and its implications during a session on potentially practice-changing clinical trials presented virtually during IDWeek 2020, an annual scientific meeting on infectious diseases.
In the trial, Nina Singh, MD, and colleagues randomly assigned 100 CMV-seronegative liver transplant recipients to receive preemptive therapy, in which patients underwent weekly testing for 100 days with a highly sensitive real-time plasma polymerase chain reaction assay for CMV. If viremia at any level was detected, the patients received oral valganciclovir 900 mg twice daily until two consecutive tests performed 1 week apart came back negative.
The remaining 105 patients were randomly assigned to 100 days of oral prophylaxis with 900 mg valganciclovir twice daily, started within 10 days of transplant.
CMV disease incidence lower
The incidence of CMV disease within 12 months of transplants, the primary outcome, was 9% in the preemptive therapy group, compared with 19% in the prophylaxis group (P = .04)
The difference between the groups was largely accounted for by a reduction in disease onset beyond 100 days in the preemptive therapy group (6% vs. 17%, respectively, P = .01)
There were no significant differences in secondary endpoints of rejection, opportunistic infections, graft loss because of retransplantation, neutropenia, or receipt of one or more doses of granulocyte colony–stimulating factor for the management of neutropenia.
At 1-year follow-up, the incidence of all-cause mortality was 15% in the preemptive therapy group, and 19% in the prophylaxis group; the difference was not statistically significant.
“While most transplant centers utilize universal prophylaxis, I think that this study really suggests that preemptive monitoring, if it can be safely accomplished at your center, may be of the greatest benefit to your patients,” Dr. Ison said.
He noted that Singh et al. also looked in an exploratory analysis at CMV-specific immunity and observed that patients assigned to preemptive therapy “clearly had better CMV-specific immunity, whether CD4 or CD8 cells, and had higher lymphocyte numbers than those patients that had received universal prophylaxis.”
In a comment, Sarah Doernberg, MD, from the division of infectious diseases at the University of California, San Francisco, agreed that “exploratory analysis of CMV-specific immune responses suggested increased CMV-specific immunity in those in the preemptive group, a finding that warrants further study. The feasibility of adopting reliable preemptive monitoring must be considered as individual centers ponder adopting this approach.”
Dr. Doernberg moderated the session where Dr. Ison discussed the data, but was not involved in the research.
The study by Singh et al. was supported by the National Institutes of Health. Dr. Singh reported research grants from NIH. Dr. Ison disclosed research support and paid consultation for several companies. Dr. Doernberg disclosed consulting for Basilea and Genentech.
FROM IDWEEK 2020
INR fails to predict bleeding in patients with cirrhosis
International normalized ratio (INR) does not predict periprocedural bleeding in patients with cirrhosis, according to a meta-analysis of 29 studies.
This finding should deter the common practice of delivering blood products to cirrhotic patients with an elevated INR, reported lead author Alexander J. Kovalic, MD, of Novant Forsyth Medical Center in Winston Salem, N.C., and colleagues.
“INR measurement among cirrhotic patients is important in MELD [Model for End-Stage Liver Disease] prognostication and assessment of underlying hepatic synthetic function, however the INR alone does not capture the complicated interplay of anticoagulant and procoagulant deficiencies present in cirrhotic coagulopathy,” Dr. Kovalic and colleagues wrote in Alimentary Pharmacology & Therapeutics. “Yet, the ‘correction’ of these aberrancies among peripheral coagulation tests remains common ... even in modern practice, and not uncommonly occurs in the periprocedural setting.”
According to investigators, addressing INR with blood transfusion can have a litany of negative effects. Beyond the risks faced by all patient populations, increasing blood volume in those with cirrhosis can increase portal venous pressure, thereby raising risks of portal gastropathy or variceal hemorrhage. In addition, giving plasma products to patients with cirrhotic coagulopathy may further disrupt the balance between anticoagulants and procoagulants, potentially triggering disseminated intravascular coagulation.
Dr. Kovalic and colleagues noted that the lack of correlation between peripheral coagulation tests and bleeding risk has been a longstanding subject of investigation, citing studies from as early as 1981.
To add further weight to this body of evidence, the investigators conducted a systematic review and meta-analysis involving 13,276 patients with cirrhosis who underwent various procedures between 1999 and 2019. Primary outcomes included periprocedural bleeding events and the association between preprocedural INR and periprocedural bleeding events. Secondary outcomes included mortality, quantity of blood and/or plasma products used, and relationship between preprocedural platelet count and periprocedural bleeding events.
The analysis showed that preprocedural INR was not significantly associated with periprocedural bleeding events (pooled odds ratio, 1.52; 95% confidence interval, 0.99-2.33; P = .06), a finding that held across INR threshold subgroups. Similarly, no significant difference was found between mean INR of patients who had bleeding events versus that of those who did not (pooled mean difference, 0.05; 95% CI, 0.03-0.13; P = .23).
Preprocedural platelet count was also a poor predictor of periprocedural bleeding, with a pooled odds ratio of 1.24 (95% CI, 0.55-2.77; P = .60), although the investigators noted that platelet count thresholds varied widely across studies, from 30 to 150 × 109/L. When studies were stratified by procedural bleeding risk or procedure type, subgroup effects were no longer significant. Other secondary endpoints were incalculable because of insufficient data.
“Hopefully, these findings will spark initiation of more large-scale, higher-quality studies ... to reinforce minimizing administration of fresh frozen plasma for inappropriate correction of INR, which carries a multitude of adverse effects among cirrhotic [patients],” the investigators concluded.
According to Stephen H. Caldwell, MD, of the University of Virginia in Charlottesville, “The present paper augments accumulating literature over the past 15 years that INR should be discarded as a measure of procedure-related bleeding risk.”
Dr. Caldwell pointed out that “bleeding in cirrhosis is usually related to portal hypertension not with impaired hemostasis, with the occasional exception of hyperfibrinolysis, which is very different from a prolonged INR.”
He went on to suggest that the present findings should dissuade clinicians from a practice that, for some, is reflexive rather than evidence based.
“It’s remarkable how many medical practices become entrenched based on hand-me-down teaching during our early training years, and remain so for many years beyond as we disperse into various medical and surgical fields,” Dr. Caldwell said. “These learned approaches to common problems can clearly persist for generations despite overwhelming evidence to the contrary that usually evolve slowly and well-insulated within subspecialties or sub-subspecialties, and hence take several generations of training to diffuse into the wider practice of medical care for common problems. These may become matters of expedience in decision-making, much like the old antibiotic conundrum of ‘no-think-a-cillin,’ as critics referred to over-use of broad spectrum antibiotics. And so it has been with the INR.”The investigators disclosed relationships with AbbVie, Eisai, Gilead, and others. Dr. Caldwell disclosed research support from Daiichi concerning the potential role of anticoagulation therapy in preventing cirrhosis progression.
SOURCE: Kovalic AJ et al. Aliment Pharmacol Ther. 2020 Sep 10. doi: 10.1111/apt.16078.
International normalized ratio (INR) does not predict periprocedural bleeding in patients with cirrhosis, according to a meta-analysis of 29 studies.
This finding should deter the common practice of delivering blood products to cirrhotic patients with an elevated INR, reported lead author Alexander J. Kovalic, MD, of Novant Forsyth Medical Center in Winston Salem, N.C., and colleagues.
“INR measurement among cirrhotic patients is important in MELD [Model for End-Stage Liver Disease] prognostication and assessment of underlying hepatic synthetic function, however the INR alone does not capture the complicated interplay of anticoagulant and procoagulant deficiencies present in cirrhotic coagulopathy,” Dr. Kovalic and colleagues wrote in Alimentary Pharmacology & Therapeutics. “Yet, the ‘correction’ of these aberrancies among peripheral coagulation tests remains common ... even in modern practice, and not uncommonly occurs in the periprocedural setting.”
According to investigators, addressing INR with blood transfusion can have a litany of negative effects. Beyond the risks faced by all patient populations, increasing blood volume in those with cirrhosis can increase portal venous pressure, thereby raising risks of portal gastropathy or variceal hemorrhage. In addition, giving plasma products to patients with cirrhotic coagulopathy may further disrupt the balance between anticoagulants and procoagulants, potentially triggering disseminated intravascular coagulation.
Dr. Kovalic and colleagues noted that the lack of correlation between peripheral coagulation tests and bleeding risk has been a longstanding subject of investigation, citing studies from as early as 1981.
To add further weight to this body of evidence, the investigators conducted a systematic review and meta-analysis involving 13,276 patients with cirrhosis who underwent various procedures between 1999 and 2019. Primary outcomes included periprocedural bleeding events and the association between preprocedural INR and periprocedural bleeding events. Secondary outcomes included mortality, quantity of blood and/or plasma products used, and relationship between preprocedural platelet count and periprocedural bleeding events.
The analysis showed that preprocedural INR was not significantly associated with periprocedural bleeding events (pooled odds ratio, 1.52; 95% confidence interval, 0.99-2.33; P = .06), a finding that held across INR threshold subgroups. Similarly, no significant difference was found between mean INR of patients who had bleeding events versus that of those who did not (pooled mean difference, 0.05; 95% CI, 0.03-0.13; P = .23).
Preprocedural platelet count was also a poor predictor of periprocedural bleeding, with a pooled odds ratio of 1.24 (95% CI, 0.55-2.77; P = .60), although the investigators noted that platelet count thresholds varied widely across studies, from 30 to 150 × 109/L. When studies were stratified by procedural bleeding risk or procedure type, subgroup effects were no longer significant. Other secondary endpoints were incalculable because of insufficient data.
“Hopefully, these findings will spark initiation of more large-scale, higher-quality studies ... to reinforce minimizing administration of fresh frozen plasma for inappropriate correction of INR, which carries a multitude of adverse effects among cirrhotic [patients],” the investigators concluded.
According to Stephen H. Caldwell, MD, of the University of Virginia in Charlottesville, “The present paper augments accumulating literature over the past 15 years that INR should be discarded as a measure of procedure-related bleeding risk.”
Dr. Caldwell pointed out that “bleeding in cirrhosis is usually related to portal hypertension not with impaired hemostasis, with the occasional exception of hyperfibrinolysis, which is very different from a prolonged INR.”
He went on to suggest that the present findings should dissuade clinicians from a practice that, for some, is reflexive rather than evidence based.
“It’s remarkable how many medical practices become entrenched based on hand-me-down teaching during our early training years, and remain so for many years beyond as we disperse into various medical and surgical fields,” Dr. Caldwell said. “These learned approaches to common problems can clearly persist for generations despite overwhelming evidence to the contrary that usually evolve slowly and well-insulated within subspecialties or sub-subspecialties, and hence take several generations of training to diffuse into the wider practice of medical care for common problems. These may become matters of expedience in decision-making, much like the old antibiotic conundrum of ‘no-think-a-cillin,’ as critics referred to over-use of broad spectrum antibiotics. And so it has been with the INR.”The investigators disclosed relationships with AbbVie, Eisai, Gilead, and others. Dr. Caldwell disclosed research support from Daiichi concerning the potential role of anticoagulation therapy in preventing cirrhosis progression.
SOURCE: Kovalic AJ et al. Aliment Pharmacol Ther. 2020 Sep 10. doi: 10.1111/apt.16078.
International normalized ratio (INR) does not predict periprocedural bleeding in patients with cirrhosis, according to a meta-analysis of 29 studies.
This finding should deter the common practice of delivering blood products to cirrhotic patients with an elevated INR, reported lead author Alexander J. Kovalic, MD, of Novant Forsyth Medical Center in Winston Salem, N.C., and colleagues.
“INR measurement among cirrhotic patients is important in MELD [Model for End-Stage Liver Disease] prognostication and assessment of underlying hepatic synthetic function, however the INR alone does not capture the complicated interplay of anticoagulant and procoagulant deficiencies present in cirrhotic coagulopathy,” Dr. Kovalic and colleagues wrote in Alimentary Pharmacology & Therapeutics. “Yet, the ‘correction’ of these aberrancies among peripheral coagulation tests remains common ... even in modern practice, and not uncommonly occurs in the periprocedural setting.”
According to investigators, addressing INR with blood transfusion can have a litany of negative effects. Beyond the risks faced by all patient populations, increasing blood volume in those with cirrhosis can increase portal venous pressure, thereby raising risks of portal gastropathy or variceal hemorrhage. In addition, giving plasma products to patients with cirrhotic coagulopathy may further disrupt the balance between anticoagulants and procoagulants, potentially triggering disseminated intravascular coagulation.
Dr. Kovalic and colleagues noted that the lack of correlation between peripheral coagulation tests and bleeding risk has been a longstanding subject of investigation, citing studies from as early as 1981.
To add further weight to this body of evidence, the investigators conducted a systematic review and meta-analysis involving 13,276 patients with cirrhosis who underwent various procedures between 1999 and 2019. Primary outcomes included periprocedural bleeding events and the association between preprocedural INR and periprocedural bleeding events. Secondary outcomes included mortality, quantity of blood and/or plasma products used, and relationship between preprocedural platelet count and periprocedural bleeding events.
The analysis showed that preprocedural INR was not significantly associated with periprocedural bleeding events (pooled odds ratio, 1.52; 95% confidence interval, 0.99-2.33; P = .06), a finding that held across INR threshold subgroups. Similarly, no significant difference was found between mean INR of patients who had bleeding events versus that of those who did not (pooled mean difference, 0.05; 95% CI, 0.03-0.13; P = .23).
Preprocedural platelet count was also a poor predictor of periprocedural bleeding, with a pooled odds ratio of 1.24 (95% CI, 0.55-2.77; P = .60), although the investigators noted that platelet count thresholds varied widely across studies, from 30 to 150 × 109/L. When studies were stratified by procedural bleeding risk or procedure type, subgroup effects were no longer significant. Other secondary endpoints were incalculable because of insufficient data.
“Hopefully, these findings will spark initiation of more large-scale, higher-quality studies ... to reinforce minimizing administration of fresh frozen plasma for inappropriate correction of INR, which carries a multitude of adverse effects among cirrhotic [patients],” the investigators concluded.
According to Stephen H. Caldwell, MD, of the University of Virginia in Charlottesville, “The present paper augments accumulating literature over the past 15 years that INR should be discarded as a measure of procedure-related bleeding risk.”
Dr. Caldwell pointed out that “bleeding in cirrhosis is usually related to portal hypertension not with impaired hemostasis, with the occasional exception of hyperfibrinolysis, which is very different from a prolonged INR.”
He went on to suggest that the present findings should dissuade clinicians from a practice that, for some, is reflexive rather than evidence based.
“It’s remarkable how many medical practices become entrenched based on hand-me-down teaching during our early training years, and remain so for many years beyond as we disperse into various medical and surgical fields,” Dr. Caldwell said. “These learned approaches to common problems can clearly persist for generations despite overwhelming evidence to the contrary that usually evolve slowly and well-insulated within subspecialties or sub-subspecialties, and hence take several generations of training to diffuse into the wider practice of medical care for common problems. These may become matters of expedience in decision-making, much like the old antibiotic conundrum of ‘no-think-a-cillin,’ as critics referred to over-use of broad spectrum antibiotics. And so it has been with the INR.”The investigators disclosed relationships with AbbVie, Eisai, Gilead, and others. Dr. Caldwell disclosed research support from Daiichi concerning the potential role of anticoagulation therapy in preventing cirrhosis progression.
SOURCE: Kovalic AJ et al. Aliment Pharmacol Ther. 2020 Sep 10. doi: 10.1111/apt.16078.
FROM ALIMENTARY PHARMACOLOGY & THERAPEUTICS
Time to screen for liver disease in type 2 diabetes?
With high rates of fatty liver disease known to occur among people with type 2 diabetes, is it time to introduce routine liver screening into daily diabetes practice? The answer depends on whom you ask, and then there are still some important caveats.
From the hepatologist’s perspective, there is no excuse not to consider liver surveillance now that noninvasive screening methods are available, suggested Michael Trauner, MD, of the Medical University of Vienna.
“From a practical standpoint, I think every type 2 diabetic over 50 years of age is at high risk,” and consequently should be screened at diagnosis, Dr. Trauner said during a debate at the virtual annual meeting of the European Association for the Study of Diabetes. “I would screen at diagnosis and then decide on recall depending on noninvasive fibrosis markers.”
“It’s a rising problem that we are facing these days,” observed Michael Roden, MD, chair and professor of internal medicine, endocrinology and metabolic diseases at Heinrich-Heine University in Düsseldorf, Germany, and who cochaired the session. Not only do people with type 2 diabetes have an increased risk for developing liver diseases, but also there’s a higher risk for those with fatty liver diseases developing type 2 diabetes.
A meta-analysis published in Gut in just last week illustrates just how big a problem this is – nonalcoholic fatty liver disease (NAFLD) “doubled the risk of type 2 diabetes,” said Dr Rosen, who is also the director of the division of endocrinology and diabetology at University Clinics Düsseldorf. That analysis was based on more than 500,000 people, almost 28,000 of whom had incident diabetes over a 5-year period.
Screening tools scarce
This makes liver screening in type 2 diabetes patients “a formidable challenge,” cautioned Gianluca Perseghin, MD, professor of endocrinology at the Monza (Italy) Polyclinic and the University of Milano-Bicocca in Milan.
“Hepatologists generally see only the most severe cases,” Dr. Perseghin said. Diabetologists and endocrinologists would be likely to see huge numbers of patients that could potentially be at risk for liver disease and following the recommendations set out in the joint European Association for the Study of the Liver/EASD/European Association for the Study of Obesity guidelines would result in a huge number of patients being identified and potentially needing referral, he argued.
“At this stage, we need to build friendly, reliable and cost-effective screening process to be applied in the health systems,” Dr. Perseghin suggested. He proposed that liver surveillance would need to be not only personalized on a patient level, but also at the infrastructure level. Measuring liver enzymes, for example, was going to be less accurate in picking up liver disease but blood tests were widely available, whereas imaging methods were not going to be something all diabetes clinics would have immediate access to.
“There are clearly a lot of provocative decisions still to be made,” acknowledged Philip Newsome, PhD, FRCPE, an honorary consultant hepatologist at the University of Birmingham (England) and who cochaired the debate.
“We need to demonstrate that looking for the presence of liver disease in this cohort changes their outcomes in a way that is cost effective,” Dr. Newsome, who is also the secretary general of EASL.
“Tests are evolving, but more importantly, treatments are evolving. So, the decision around cost effectiveness will clearly change,” he added.
NAFLD therapies unclar
“There are still a lot of questions,” Dr. Newsome said during a Novo-Nordisk–sponsored “Meet the Expert” session discussing EASL-EASD-EASO guidelines. “We don’t have any licensed therapies at the moment. But there’s been a huge amount of investment, looking at all sorts of different approaches.”
Dr. Newsome added: “We also don’t know how to monitor these patients. Most of the noninvasive are very useful for staging patients, but we don’t really understand how useful they are for monitoring changes in fibrosis.”
Diabetologist Hannele Yki-Järvinen, MD, PhD, of the University of Helsinki, gave her thoughts on the topic during the same session.
“We should add FIB-4 [Fibrosis-4 index] to the annual exam and ask the lab to calculate FIB-4 automatically,” Dr. Yki-Järvinen said. FIB-4is calculated using the patients age and the results of readily available blood tests that measure the AST/ALT ratio and the platelet count.
Dr. Trauner has received advisory fees and grant support from various companies with an interest in developing liver-directed therapies, and is also a coinventor of 24-norursodeoxycholic acid under development for cholestatic liver disease and potentially NAFLD. Dr. Perseghin has received honoraria and grant support from various pharmaceutical companies with an interest in diabetes care. Dr. Roden did not provide any disclosures. Dr. Newsome has received research grants from Boehringer Ingelheim and Novo Nordisk and acted as a consultant to many pharmaceutical companies. Dr. Yki-Järvinen disclosed receiving consultancy fees from Eli Lilly, MSD, and Novo Nordisk.
SOURCE: Trauner M; Persghin G. EASD 2020, Session S27.
With high rates of fatty liver disease known to occur among people with type 2 diabetes, is it time to introduce routine liver screening into daily diabetes practice? The answer depends on whom you ask, and then there are still some important caveats.
From the hepatologist’s perspective, there is no excuse not to consider liver surveillance now that noninvasive screening methods are available, suggested Michael Trauner, MD, of the Medical University of Vienna.
“From a practical standpoint, I think every type 2 diabetic over 50 years of age is at high risk,” and consequently should be screened at diagnosis, Dr. Trauner said during a debate at the virtual annual meeting of the European Association for the Study of Diabetes. “I would screen at diagnosis and then decide on recall depending on noninvasive fibrosis markers.”
“It’s a rising problem that we are facing these days,” observed Michael Roden, MD, chair and professor of internal medicine, endocrinology and metabolic diseases at Heinrich-Heine University in Düsseldorf, Germany, and who cochaired the session. Not only do people with type 2 diabetes have an increased risk for developing liver diseases, but also there’s a higher risk for those with fatty liver diseases developing type 2 diabetes.
A meta-analysis published in Gut in just last week illustrates just how big a problem this is – nonalcoholic fatty liver disease (NAFLD) “doubled the risk of type 2 diabetes,” said Dr Rosen, who is also the director of the division of endocrinology and diabetology at University Clinics Düsseldorf. That analysis was based on more than 500,000 people, almost 28,000 of whom had incident diabetes over a 5-year period.
Screening tools scarce
This makes liver screening in type 2 diabetes patients “a formidable challenge,” cautioned Gianluca Perseghin, MD, professor of endocrinology at the Monza (Italy) Polyclinic and the University of Milano-Bicocca in Milan.
“Hepatologists generally see only the most severe cases,” Dr. Perseghin said. Diabetologists and endocrinologists would be likely to see huge numbers of patients that could potentially be at risk for liver disease and following the recommendations set out in the joint European Association for the Study of the Liver/EASD/European Association for the Study of Obesity guidelines would result in a huge number of patients being identified and potentially needing referral, he argued.
“At this stage, we need to build friendly, reliable and cost-effective screening process to be applied in the health systems,” Dr. Perseghin suggested. He proposed that liver surveillance would need to be not only personalized on a patient level, but also at the infrastructure level. Measuring liver enzymes, for example, was going to be less accurate in picking up liver disease but blood tests were widely available, whereas imaging methods were not going to be something all diabetes clinics would have immediate access to.
“There are clearly a lot of provocative decisions still to be made,” acknowledged Philip Newsome, PhD, FRCPE, an honorary consultant hepatologist at the University of Birmingham (England) and who cochaired the debate.
“We need to demonstrate that looking for the presence of liver disease in this cohort changes their outcomes in a way that is cost effective,” Dr. Newsome, who is also the secretary general of EASL.
“Tests are evolving, but more importantly, treatments are evolving. So, the decision around cost effectiveness will clearly change,” he added.
NAFLD therapies unclar
“There are still a lot of questions,” Dr. Newsome said during a Novo-Nordisk–sponsored “Meet the Expert” session discussing EASL-EASD-EASO guidelines. “We don’t have any licensed therapies at the moment. But there’s been a huge amount of investment, looking at all sorts of different approaches.”
Dr. Newsome added: “We also don’t know how to monitor these patients. Most of the noninvasive are very useful for staging patients, but we don’t really understand how useful they are for monitoring changes in fibrosis.”
Diabetologist Hannele Yki-Järvinen, MD, PhD, of the University of Helsinki, gave her thoughts on the topic during the same session.
“We should add FIB-4 [Fibrosis-4 index] to the annual exam and ask the lab to calculate FIB-4 automatically,” Dr. Yki-Järvinen said. FIB-4is calculated using the patients age and the results of readily available blood tests that measure the AST/ALT ratio and the platelet count.
Dr. Trauner has received advisory fees and grant support from various companies with an interest in developing liver-directed therapies, and is also a coinventor of 24-norursodeoxycholic acid under development for cholestatic liver disease and potentially NAFLD. Dr. Perseghin has received honoraria and grant support from various pharmaceutical companies with an interest in diabetes care. Dr. Roden did not provide any disclosures. Dr. Newsome has received research grants from Boehringer Ingelheim and Novo Nordisk and acted as a consultant to many pharmaceutical companies. Dr. Yki-Järvinen disclosed receiving consultancy fees from Eli Lilly, MSD, and Novo Nordisk.
SOURCE: Trauner M; Persghin G. EASD 2020, Session S27.
With high rates of fatty liver disease known to occur among people with type 2 diabetes, is it time to introduce routine liver screening into daily diabetes practice? The answer depends on whom you ask, and then there are still some important caveats.
From the hepatologist’s perspective, there is no excuse not to consider liver surveillance now that noninvasive screening methods are available, suggested Michael Trauner, MD, of the Medical University of Vienna.
“From a practical standpoint, I think every type 2 diabetic over 50 years of age is at high risk,” and consequently should be screened at diagnosis, Dr. Trauner said during a debate at the virtual annual meeting of the European Association for the Study of Diabetes. “I would screen at diagnosis and then decide on recall depending on noninvasive fibrosis markers.”
“It’s a rising problem that we are facing these days,” observed Michael Roden, MD, chair and professor of internal medicine, endocrinology and metabolic diseases at Heinrich-Heine University in Düsseldorf, Germany, and who cochaired the session. Not only do people with type 2 diabetes have an increased risk for developing liver diseases, but also there’s a higher risk for those with fatty liver diseases developing type 2 diabetes.
A meta-analysis published in Gut in just last week illustrates just how big a problem this is – nonalcoholic fatty liver disease (NAFLD) “doubled the risk of type 2 diabetes,” said Dr Rosen, who is also the director of the division of endocrinology and diabetology at University Clinics Düsseldorf. That analysis was based on more than 500,000 people, almost 28,000 of whom had incident diabetes over a 5-year period.
Screening tools scarce
This makes liver screening in type 2 diabetes patients “a formidable challenge,” cautioned Gianluca Perseghin, MD, professor of endocrinology at the Monza (Italy) Polyclinic and the University of Milano-Bicocca in Milan.
“Hepatologists generally see only the most severe cases,” Dr. Perseghin said. Diabetologists and endocrinologists would be likely to see huge numbers of patients that could potentially be at risk for liver disease and following the recommendations set out in the joint European Association for the Study of the Liver/EASD/European Association for the Study of Obesity guidelines would result in a huge number of patients being identified and potentially needing referral, he argued.
“At this stage, we need to build friendly, reliable and cost-effective screening process to be applied in the health systems,” Dr. Perseghin suggested. He proposed that liver surveillance would need to be not only personalized on a patient level, but also at the infrastructure level. Measuring liver enzymes, for example, was going to be less accurate in picking up liver disease but blood tests were widely available, whereas imaging methods were not going to be something all diabetes clinics would have immediate access to.
“There are clearly a lot of provocative decisions still to be made,” acknowledged Philip Newsome, PhD, FRCPE, an honorary consultant hepatologist at the University of Birmingham (England) and who cochaired the debate.
“We need to demonstrate that looking for the presence of liver disease in this cohort changes their outcomes in a way that is cost effective,” Dr. Newsome, who is also the secretary general of EASL.
“Tests are evolving, but more importantly, treatments are evolving. So, the decision around cost effectiveness will clearly change,” he added.
NAFLD therapies unclar
“There are still a lot of questions,” Dr. Newsome said during a Novo-Nordisk–sponsored “Meet the Expert” session discussing EASL-EASD-EASO guidelines. “We don’t have any licensed therapies at the moment. But there’s been a huge amount of investment, looking at all sorts of different approaches.”
Dr. Newsome added: “We also don’t know how to monitor these patients. Most of the noninvasive are very useful for staging patients, but we don’t really understand how useful they are for monitoring changes in fibrosis.”
Diabetologist Hannele Yki-Järvinen, MD, PhD, of the University of Helsinki, gave her thoughts on the topic during the same session.
“We should add FIB-4 [Fibrosis-4 index] to the annual exam and ask the lab to calculate FIB-4 automatically,” Dr. Yki-Järvinen said. FIB-4is calculated using the patients age and the results of readily available blood tests that measure the AST/ALT ratio and the platelet count.
Dr. Trauner has received advisory fees and grant support from various companies with an interest in developing liver-directed therapies, and is also a coinventor of 24-norursodeoxycholic acid under development for cholestatic liver disease and potentially NAFLD. Dr. Perseghin has received honoraria and grant support from various pharmaceutical companies with an interest in diabetes care. Dr. Roden did not provide any disclosures. Dr. Newsome has received research grants from Boehringer Ingelheim and Novo Nordisk and acted as a consultant to many pharmaceutical companies. Dr. Yki-Järvinen disclosed receiving consultancy fees from Eli Lilly, MSD, and Novo Nordisk.
SOURCE: Trauner M; Persghin G. EASD 2020, Session S27.
REPORTING FROM EASD 2020
Address root causes to manage NASH
Not only the prevalence, but the impact of nonalcoholic fatty liver disease (NAFLD) is increasing in much of the world, Arun J. Sanyal, MD, said in a virtual presentation at the meeting jointly provided by Rutgers and Global Academy for Medical Education. “It is currently estimated that the number of people living with cirrhosis or with decompensated cirrhosis will increase two- to threefold from 2015 to 2030,” which underlines the public health impact and the need for improved treatment paradigms, he emphasized.
“The thing to remember about NAFLD is that it does not exist in a vacuum,” Dr. Sanyal said. NAFLD is a multisystem disorder. Most patients have concomitant cardiovascular disease, but others may have type 2 diabetes, hypertension, and dyslipidemia, all of which are now accepted as risk factors for nonalcoholic steatohepatitis (NASH), he said.
“What ties these conditions together is metabolic stress leading to systemic inflammation and fibrosis. This is primarily due to diet-induced obesity. If you think about treating all of these competing risks to the patient’s life, the optimal way is to treat the root cause,” he said.
Various options exist to manage the conditions that can lead to NASH, but several of these also appear promising as a treatment of NASH, Dr. Sanyal said. Glucagonlike peptide–1 agonists and sodium-glucose transporter 2 inhibitors have been shown to improve multiple outcomes of interest in type 2 diabetes. However, insulin can cause weight gain at the expense of controlling HbA1C levels, he said.
Bariatric surgery can improve histology, but many patients with advanced fibrosis do not demonstrate improvement in fibrosis. Also, bariatric surgery has its own associated morbidity, including an increased suicide rate across multiple studies, Dr. Sanyal noted.
A new and interesting option is duodenal mucosal resurfacing (DMR) “a novel, minimally invasive outpatient upper-endoscopic procedure,” said Dr. Sanyal. DMR involves use of a catheter to perform a submucosal lift and hydrothermal mucosal ablation, prompting healthy epithelial regrowth, he explained. “The mucosa sloughs off, fresh epithelium grows in, and the hormonal signal from the gut to the rest of the body is restored to a more normal pattern,” he noted.
In the REVITA-2 study of patients with diabetes and NAFLD, the average fat loss was 5.4% in those randomized to DMR vs. 2.4% in sham-procedure patients and represented “quite significant defatting of the liver,” Dr. Sanyal said.
Dr. Sanyal then focused on fatty liver disease. “The first step when you see a patient with fatty liver disease is to see how scarred is the liver, and whether the patient has silent cirrhosis. The more scarred the liver, the greater risk of liver-related outcomes,” he said. The goal of therapy for these patients is to reduce the risk of progression to cirrhosis, he added. Dr. Sanyal recommended evaluating fibrosis using the Fibrosis 4 score (Fib4). “If the Fib4 is less than 1.3, the likelihood of significant scarring in the liver is less than 10%,” he said. On the other hand, a Fib4 greater than 2.67 suggests advanced fibrosis, he noted.
Overall, the goals of treatment for NASH patients are to prevent cirrhosis, reduce decompensation, and prevent hepatocellular carcinoma, said Dr. Sanyal.
“The ideal drug for NASH should also help other end organs, or at least be neutral,” said Dr. Sanyal.
Current frontline therapies for precirrhotic NASH include thiazolidinediones (TZD), farnesoid X receptor (FXR)/fibroblast growth factor 19 (FGF-19), FGF21, thyroxine B-R, and glucagonlike peptide-1. Clinical evidence varies based on different populations, endpoints, assessment methods, and treatment duration, he said.
Looking ahead to the next decade, a NASH management paradigm will likely play out that can be applied in the clinic today, Dr. Sanyal said. First, make an initial assessment of the status of the end organs. Start with a weight-loss regimen; use statins and GLP-1 and SGLT2 inhibitors as needed. Follow and reassess, and if the patient still has disease, progress to targeted therapy for active NASH while continuing to encourage weight loss and healthy living, he said.
“The ultimate proof that what we are doing is working is that we are improving mortality, reducing health care costs, and improving patients’ function and quality of life,” he concluded.
Dr. Sanyal is president of Sanyal Biotechnologies. He also disclosed stock options for Durect, Exhalenz, Galmed, Genfit, Immuton, Indalo, and Tiziana, as well as various relationships with Allergan, AMRA, Astra Zeneca-Medimmune, Birdrock, Boehringer Ingelheim, Bristol Myers, Echosense, GE, Genentech, Gilead, Hemoshear, IFMO, Innovate, Intercept, Lilly, Lipocine, Merck, Novartis, Novo Nordisk, OWL, Pfizer, RedX, Sundise, Tern, and Zydus.
Global Academy for Medical Education and this news organization are owned by the same parent company.
Help your patients understand their risks for NASH by sharing AGA patient education at http://ow.ly/5AAk30rbK5y.
Not only the prevalence, but the impact of nonalcoholic fatty liver disease (NAFLD) is increasing in much of the world, Arun J. Sanyal, MD, said in a virtual presentation at the meeting jointly provided by Rutgers and Global Academy for Medical Education. “It is currently estimated that the number of people living with cirrhosis or with decompensated cirrhosis will increase two- to threefold from 2015 to 2030,” which underlines the public health impact and the need for improved treatment paradigms, he emphasized.
“The thing to remember about NAFLD is that it does not exist in a vacuum,” Dr. Sanyal said. NAFLD is a multisystem disorder. Most patients have concomitant cardiovascular disease, but others may have type 2 diabetes, hypertension, and dyslipidemia, all of which are now accepted as risk factors for nonalcoholic steatohepatitis (NASH), he said.
“What ties these conditions together is metabolic stress leading to systemic inflammation and fibrosis. This is primarily due to diet-induced obesity. If you think about treating all of these competing risks to the patient’s life, the optimal way is to treat the root cause,” he said.
Various options exist to manage the conditions that can lead to NASH, but several of these also appear promising as a treatment of NASH, Dr. Sanyal said. Glucagonlike peptide–1 agonists and sodium-glucose transporter 2 inhibitors have been shown to improve multiple outcomes of interest in type 2 diabetes. However, insulin can cause weight gain at the expense of controlling HbA1C levels, he said.
Bariatric surgery can improve histology, but many patients with advanced fibrosis do not demonstrate improvement in fibrosis. Also, bariatric surgery has its own associated morbidity, including an increased suicide rate across multiple studies, Dr. Sanyal noted.
A new and interesting option is duodenal mucosal resurfacing (DMR) “a novel, minimally invasive outpatient upper-endoscopic procedure,” said Dr. Sanyal. DMR involves use of a catheter to perform a submucosal lift and hydrothermal mucosal ablation, prompting healthy epithelial regrowth, he explained. “The mucosa sloughs off, fresh epithelium grows in, and the hormonal signal from the gut to the rest of the body is restored to a more normal pattern,” he noted.
In the REVITA-2 study of patients with diabetes and NAFLD, the average fat loss was 5.4% in those randomized to DMR vs. 2.4% in sham-procedure patients and represented “quite significant defatting of the liver,” Dr. Sanyal said.
Dr. Sanyal then focused on fatty liver disease. “The first step when you see a patient with fatty liver disease is to see how scarred is the liver, and whether the patient has silent cirrhosis. The more scarred the liver, the greater risk of liver-related outcomes,” he said. The goal of therapy for these patients is to reduce the risk of progression to cirrhosis, he added. Dr. Sanyal recommended evaluating fibrosis using the Fibrosis 4 score (Fib4). “If the Fib4 is less than 1.3, the likelihood of significant scarring in the liver is less than 10%,” he said. On the other hand, a Fib4 greater than 2.67 suggests advanced fibrosis, he noted.
Overall, the goals of treatment for NASH patients are to prevent cirrhosis, reduce decompensation, and prevent hepatocellular carcinoma, said Dr. Sanyal.
“The ideal drug for NASH should also help other end organs, or at least be neutral,” said Dr. Sanyal.
Current frontline therapies for precirrhotic NASH include thiazolidinediones (TZD), farnesoid X receptor (FXR)/fibroblast growth factor 19 (FGF-19), FGF21, thyroxine B-R, and glucagonlike peptide-1. Clinical evidence varies based on different populations, endpoints, assessment methods, and treatment duration, he said.
Looking ahead to the next decade, a NASH management paradigm will likely play out that can be applied in the clinic today, Dr. Sanyal said. First, make an initial assessment of the status of the end organs. Start with a weight-loss regimen; use statins and GLP-1 and SGLT2 inhibitors as needed. Follow and reassess, and if the patient still has disease, progress to targeted therapy for active NASH while continuing to encourage weight loss and healthy living, he said.
“The ultimate proof that what we are doing is working is that we are improving mortality, reducing health care costs, and improving patients’ function and quality of life,” he concluded.
Dr. Sanyal is president of Sanyal Biotechnologies. He also disclosed stock options for Durect, Exhalenz, Galmed, Genfit, Immuton, Indalo, and Tiziana, as well as various relationships with Allergan, AMRA, Astra Zeneca-Medimmune, Birdrock, Boehringer Ingelheim, Bristol Myers, Echosense, GE, Genentech, Gilead, Hemoshear, IFMO, Innovate, Intercept, Lilly, Lipocine, Merck, Novartis, Novo Nordisk, OWL, Pfizer, RedX, Sundise, Tern, and Zydus.
Global Academy for Medical Education and this news organization are owned by the same parent company.
Help your patients understand their risks for NASH by sharing AGA patient education at http://ow.ly/5AAk30rbK5y.
Not only the prevalence, but the impact of nonalcoholic fatty liver disease (NAFLD) is increasing in much of the world, Arun J. Sanyal, MD, said in a virtual presentation at the meeting jointly provided by Rutgers and Global Academy for Medical Education. “It is currently estimated that the number of people living with cirrhosis or with decompensated cirrhosis will increase two- to threefold from 2015 to 2030,” which underlines the public health impact and the need for improved treatment paradigms, he emphasized.
“The thing to remember about NAFLD is that it does not exist in a vacuum,” Dr. Sanyal said. NAFLD is a multisystem disorder. Most patients have concomitant cardiovascular disease, but others may have type 2 diabetes, hypertension, and dyslipidemia, all of which are now accepted as risk factors for nonalcoholic steatohepatitis (NASH), he said.
“What ties these conditions together is metabolic stress leading to systemic inflammation and fibrosis. This is primarily due to diet-induced obesity. If you think about treating all of these competing risks to the patient’s life, the optimal way is to treat the root cause,” he said.
Various options exist to manage the conditions that can lead to NASH, but several of these also appear promising as a treatment of NASH, Dr. Sanyal said. Glucagonlike peptide–1 agonists and sodium-glucose transporter 2 inhibitors have been shown to improve multiple outcomes of interest in type 2 diabetes. However, insulin can cause weight gain at the expense of controlling HbA1C levels, he said.
Bariatric surgery can improve histology, but many patients with advanced fibrosis do not demonstrate improvement in fibrosis. Also, bariatric surgery has its own associated morbidity, including an increased suicide rate across multiple studies, Dr. Sanyal noted.
A new and interesting option is duodenal mucosal resurfacing (DMR) “a novel, minimally invasive outpatient upper-endoscopic procedure,” said Dr. Sanyal. DMR involves use of a catheter to perform a submucosal lift and hydrothermal mucosal ablation, prompting healthy epithelial regrowth, he explained. “The mucosa sloughs off, fresh epithelium grows in, and the hormonal signal from the gut to the rest of the body is restored to a more normal pattern,” he noted.
In the REVITA-2 study of patients with diabetes and NAFLD, the average fat loss was 5.4% in those randomized to DMR vs. 2.4% in sham-procedure patients and represented “quite significant defatting of the liver,” Dr. Sanyal said.
Dr. Sanyal then focused on fatty liver disease. “The first step when you see a patient with fatty liver disease is to see how scarred is the liver, and whether the patient has silent cirrhosis. The more scarred the liver, the greater risk of liver-related outcomes,” he said. The goal of therapy for these patients is to reduce the risk of progression to cirrhosis, he added. Dr. Sanyal recommended evaluating fibrosis using the Fibrosis 4 score (Fib4). “If the Fib4 is less than 1.3, the likelihood of significant scarring in the liver is less than 10%,” he said. On the other hand, a Fib4 greater than 2.67 suggests advanced fibrosis, he noted.
Overall, the goals of treatment for NASH patients are to prevent cirrhosis, reduce decompensation, and prevent hepatocellular carcinoma, said Dr. Sanyal.
“The ideal drug for NASH should also help other end organs, or at least be neutral,” said Dr. Sanyal.
Current frontline therapies for precirrhotic NASH include thiazolidinediones (TZD), farnesoid X receptor (FXR)/fibroblast growth factor 19 (FGF-19), FGF21, thyroxine B-R, and glucagonlike peptide-1. Clinical evidence varies based on different populations, endpoints, assessment methods, and treatment duration, he said.
Looking ahead to the next decade, a NASH management paradigm will likely play out that can be applied in the clinic today, Dr. Sanyal said. First, make an initial assessment of the status of the end organs. Start with a weight-loss regimen; use statins and GLP-1 and SGLT2 inhibitors as needed. Follow and reassess, and if the patient still has disease, progress to targeted therapy for active NASH while continuing to encourage weight loss and healthy living, he said.
“The ultimate proof that what we are doing is working is that we are improving mortality, reducing health care costs, and improving patients’ function and quality of life,” he concluded.
Dr. Sanyal is president of Sanyal Biotechnologies. He also disclosed stock options for Durect, Exhalenz, Galmed, Genfit, Immuton, Indalo, and Tiziana, as well as various relationships with Allergan, AMRA, Astra Zeneca-Medimmune, Birdrock, Boehringer Ingelheim, Bristol Myers, Echosense, GE, Genentech, Gilead, Hemoshear, IFMO, Innovate, Intercept, Lilly, Lipocine, Merck, Novartis, Novo Nordisk, OWL, Pfizer, RedX, Sundise, Tern, and Zydus.
Global Academy for Medical Education and this news organization are owned by the same parent company.
Help your patients understand their risks for NASH by sharing AGA patient education at http://ow.ly/5AAk30rbK5y.
FROM DIGESTIVE DISEASES: NEW ADVANCES
Hepatocellular carcinoma shows risk factor shift
Rates of hepatocellular carcinoma (HCC) continue to rise in the United States, but unevenly so given how the incidence has become highest in the Hispanic population, which is reflected in increased rates in the southern and western states, Hashem B. El-Serag, MD, of Baylor College of Medicine, Houston, said in a virtual presentation at the annual Digestive Diseases: New Advances, which is jointly provided by Rutgers and Global Academy for Medical Education.
In addition to this demographic shift, the risk factors for HCC are shifting, he said. Hepatitis C virus (HCV) has been the dominant risk factor for HCC; for patients with active HCV, the factors historically associated with increased HCC risk have included alcohol consumption, obesity, diabetes, coinfection, and genetics, he said.
This pattern is starting to change. In fact, for patients with active HCV, antiviral treatment with a sustained virologic response has surfaced as the most significant risk factor in the development of HCC, said Dr. El-Serag: Among these patients, sustained virologic response from direct-acting antivirals is associated with a significant reduction in HCC risk. However, it is important to recognize that a residual risk of HCC remains that doesn’t go away for several years, he noted.
“Who are those people who got treated, got cured, and still developed HCC? Those with cirrhosis at the time of treatment,” he said. Those with cirrhosis have cumulative incidence of 1.8% per year, but those without cirrhosis had very low risk, he said.
Some good news in HCC is that rates appear to be declining among young men, and this is thought to be one of the groups who are achieving a cure of HCV, he said.
“One would hope, if goals for HCV elimination are met, that will translate into massive reduction of HCC,” he said.
“The issue now for hepatitis is finding infected patients and curing them,” he noted.
Dr. El-Serag touched on hepatitis B (HBV), which continues to be the driving force of hepatitis infections globally. However, in patients who receive and respond to antiviral treatment “there is a significant and considerable reduction in HCC in the context of hepatitis B” similar to that seen with hepatitis C. Vaccination programs for HBV have started to make the desired impact of reducing HCC in HBV-endemic areas, he noted.
However, current risk factors for HCC are related less to HCV and HBV and more to metabolic syndrome because more people are treated for HCV and HBV, Dr. El-Serag said. He went on to address the new dominant global risk factor for HCC: obesity. Based on data from multiple studies, those who are obese, defined as a body mass index greater than 30 kg/m2, carry a twofold increased risk of developing HCC, he said.
To reduce this risk, treatment targets might address intermediate factors such as abdominal obesity, said Dr. El-Serag. He cited a study published in Hepatology in which individuals in the highest tertile for waist-hip ratio had a threefold higher risk of HCC, compared with those in the lowest tertile.
In addition, consideration of obesity must include type 2 diabetes, which is often linked to obesity and occurs in approximately one-third of adults in the United States, Dr. El-Serag said.
Treatment of type 2 diabetes may make a difference in HCC risk reduction, Dr. El-Serag noted. “The impact of treatment of diabetes on HCC risk is an area of intense interest,” he said. Based on the latest research, “the bottom line is that those treated with metformin experience a 50% reduction in the risk of HCC,” he said
Dr. El-Serag also acknowledged the impact of other risk factors for HCC: the use of statins and the presence of nonalcoholic fatty liver disease (NAFLD).
Dr. El-Serag noted that, among NAFLD patients, subgroups at even greater risk for HCC include those with diabetes, those older than 65 years, Hispanic race, and those with cirrhosis. These patients should be candidates for surveillance. Metabolic dysfunction traits such as obesity and diabetes are very common conditions, so it’s important to look at other, more specific factors, he added. “I hope that there will be tools to help clinicians classify or risk-stratify patients into different buckets,” he said.
Areas for further research on HCC continue to include risk stratification, mechanisms of action, and HCC prevention related to treatment of metabolic syndrome, he emphasized.
Dr. El-Serag had no financial conflicts to disclose. Global Academy for Medical Education and this news organization are owned by the same parent company.
*This story was updated on Oct. 28, 2020.
SOURCE: El-Serag HB. Digestive Diseases: New Advances 2020.
Rates of hepatocellular carcinoma (HCC) continue to rise in the United States, but unevenly so given how the incidence has become highest in the Hispanic population, which is reflected in increased rates in the southern and western states, Hashem B. El-Serag, MD, of Baylor College of Medicine, Houston, said in a virtual presentation at the annual Digestive Diseases: New Advances, which is jointly provided by Rutgers and Global Academy for Medical Education.
In addition to this demographic shift, the risk factors for HCC are shifting, he said. Hepatitis C virus (HCV) has been the dominant risk factor for HCC; for patients with active HCV, the factors historically associated with increased HCC risk have included alcohol consumption, obesity, diabetes, coinfection, and genetics, he said.
This pattern is starting to change. In fact, for patients with active HCV, antiviral treatment with a sustained virologic response has surfaced as the most significant risk factor in the development of HCC, said Dr. El-Serag: Among these patients, sustained virologic response from direct-acting antivirals is associated with a significant reduction in HCC risk. However, it is important to recognize that a residual risk of HCC remains that doesn’t go away for several years, he noted.
“Who are those people who got treated, got cured, and still developed HCC? Those with cirrhosis at the time of treatment,” he said. Those with cirrhosis have cumulative incidence of 1.8% per year, but those without cirrhosis had very low risk, he said.
Some good news in HCC is that rates appear to be declining among young men, and this is thought to be one of the groups who are achieving a cure of HCV, he said.
“One would hope, if goals for HCV elimination are met, that will translate into massive reduction of HCC,” he said.
“The issue now for hepatitis is finding infected patients and curing them,” he noted.
Dr. El-Serag touched on hepatitis B (HBV), which continues to be the driving force of hepatitis infections globally. However, in patients who receive and respond to antiviral treatment “there is a significant and considerable reduction in HCC in the context of hepatitis B” similar to that seen with hepatitis C. Vaccination programs for HBV have started to make the desired impact of reducing HCC in HBV-endemic areas, he noted.
However, current risk factors for HCC are related less to HCV and HBV and more to metabolic syndrome because more people are treated for HCV and HBV, Dr. El-Serag said. He went on to address the new dominant global risk factor for HCC: obesity. Based on data from multiple studies, those who are obese, defined as a body mass index greater than 30 kg/m2, carry a twofold increased risk of developing HCC, he said.
To reduce this risk, treatment targets might address intermediate factors such as abdominal obesity, said Dr. El-Serag. He cited a study published in Hepatology in which individuals in the highest tertile for waist-hip ratio had a threefold higher risk of HCC, compared with those in the lowest tertile.
In addition, consideration of obesity must include type 2 diabetes, which is often linked to obesity and occurs in approximately one-third of adults in the United States, Dr. El-Serag said.
Treatment of type 2 diabetes may make a difference in HCC risk reduction, Dr. El-Serag noted. “The impact of treatment of diabetes on HCC risk is an area of intense interest,” he said. Based on the latest research, “the bottom line is that those treated with metformin experience a 50% reduction in the risk of HCC,” he said
Dr. El-Serag also acknowledged the impact of other risk factors for HCC: the use of statins and the presence of nonalcoholic fatty liver disease (NAFLD).
Dr. El-Serag noted that, among NAFLD patients, subgroups at even greater risk for HCC include those with diabetes, those older than 65 years, Hispanic race, and those with cirrhosis. These patients should be candidates for surveillance. Metabolic dysfunction traits such as obesity and diabetes are very common conditions, so it’s important to look at other, more specific factors, he added. “I hope that there will be tools to help clinicians classify or risk-stratify patients into different buckets,” he said.
Areas for further research on HCC continue to include risk stratification, mechanisms of action, and HCC prevention related to treatment of metabolic syndrome, he emphasized.
Dr. El-Serag had no financial conflicts to disclose. Global Academy for Medical Education and this news organization are owned by the same parent company.
*This story was updated on Oct. 28, 2020.
SOURCE: El-Serag HB. Digestive Diseases: New Advances 2020.
Rates of hepatocellular carcinoma (HCC) continue to rise in the United States, but unevenly so given how the incidence has become highest in the Hispanic population, which is reflected in increased rates in the southern and western states, Hashem B. El-Serag, MD, of Baylor College of Medicine, Houston, said in a virtual presentation at the annual Digestive Diseases: New Advances, which is jointly provided by Rutgers and Global Academy for Medical Education.
In addition to this demographic shift, the risk factors for HCC are shifting, he said. Hepatitis C virus (HCV) has been the dominant risk factor for HCC; for patients with active HCV, the factors historically associated with increased HCC risk have included alcohol consumption, obesity, diabetes, coinfection, and genetics, he said.
This pattern is starting to change. In fact, for patients with active HCV, antiviral treatment with a sustained virologic response has surfaced as the most significant risk factor in the development of HCC, said Dr. El-Serag: Among these patients, sustained virologic response from direct-acting antivirals is associated with a significant reduction in HCC risk. However, it is important to recognize that a residual risk of HCC remains that doesn’t go away for several years, he noted.
“Who are those people who got treated, got cured, and still developed HCC? Those with cirrhosis at the time of treatment,” he said. Those with cirrhosis have cumulative incidence of 1.8% per year, but those without cirrhosis had very low risk, he said.
Some good news in HCC is that rates appear to be declining among young men, and this is thought to be one of the groups who are achieving a cure of HCV, he said.
“One would hope, if goals for HCV elimination are met, that will translate into massive reduction of HCC,” he said.
“The issue now for hepatitis is finding infected patients and curing them,” he noted.
Dr. El-Serag touched on hepatitis B (HBV), which continues to be the driving force of hepatitis infections globally. However, in patients who receive and respond to antiviral treatment “there is a significant and considerable reduction in HCC in the context of hepatitis B” similar to that seen with hepatitis C. Vaccination programs for HBV have started to make the desired impact of reducing HCC in HBV-endemic areas, he noted.
However, current risk factors for HCC are related less to HCV and HBV and more to metabolic syndrome because more people are treated for HCV and HBV, Dr. El-Serag said. He went on to address the new dominant global risk factor for HCC: obesity. Based on data from multiple studies, those who are obese, defined as a body mass index greater than 30 kg/m2, carry a twofold increased risk of developing HCC, he said.
To reduce this risk, treatment targets might address intermediate factors such as abdominal obesity, said Dr. El-Serag. He cited a study published in Hepatology in which individuals in the highest tertile for waist-hip ratio had a threefold higher risk of HCC, compared with those in the lowest tertile.
In addition, consideration of obesity must include type 2 diabetes, which is often linked to obesity and occurs in approximately one-third of adults in the United States, Dr. El-Serag said.
Treatment of type 2 diabetes may make a difference in HCC risk reduction, Dr. El-Serag noted. “The impact of treatment of diabetes on HCC risk is an area of intense interest,” he said. Based on the latest research, “the bottom line is that those treated with metformin experience a 50% reduction in the risk of HCC,” he said
Dr. El-Serag also acknowledged the impact of other risk factors for HCC: the use of statins and the presence of nonalcoholic fatty liver disease (NAFLD).
Dr. El-Serag noted that, among NAFLD patients, subgroups at even greater risk for HCC include those with diabetes, those older than 65 years, Hispanic race, and those with cirrhosis. These patients should be candidates for surveillance. Metabolic dysfunction traits such as obesity and diabetes are very common conditions, so it’s important to look at other, more specific factors, he added. “I hope that there will be tools to help clinicians classify or risk-stratify patients into different buckets,” he said.
Areas for further research on HCC continue to include risk stratification, mechanisms of action, and HCC prevention related to treatment of metabolic syndrome, he emphasized.
Dr. El-Serag had no financial conflicts to disclose. Global Academy for Medical Education and this news organization are owned by the same parent company.
*This story was updated on Oct. 28, 2020.
SOURCE: El-Serag HB. Digestive Diseases: New Advances 2020.
FROM DIGESTIVE DISEASES: NEW ADVANCES
Lusutrombopag found safe, effective for severe thrombocytopenia in patients with hepatocellular carcinoma
For patients with severe thrombocytopenia and chronic liver diseases, including hepatocellular carcinoma, treatment with lusutrombopag prior to invasive procedures significantly decreased the need for platelet transfusions without increasing the need for rescue treatment for bleeding or the rate of thromboembolic events.
In a post hoc analysis of data from 270 patients in two manufacturer-sponsored, multicenter, randomized, double-blind, placebo-controlled, phase 3 trials, significantly more lusutrombopag recipients met the primary efficacy endpoint, including patients with hepatocellular carcinoma (68.0% vs. 8.9% in the placebo group; P < .0001) and those without it (77.0% vs. 21.6%; P < .0001). Rates of treatment-emergent adverse events were similar between the lusutrombopag and placebo groups, and patients with hepatocellular carcinoma were not at increased risk for thrombosis, Naim Alkhouri, MD, of Texas Liver Institute in San Antonio, and associates wrote in Clinical Gastroenterology and Hepatology.
Platelet transfusion is the treatment mainstay for patients with thrombocytopenia related to cirrhosis who are undergoing invasive procedures, but its effects are short-lived, and at least one in five transfusions fails. Thrombopoietin agonists such as lusutrombopag are efficacious and approved in this setting, but they can be prothrombotic, particularly in patients with hepatocellular carcinoma, who already are at heightened risk for portal vein thrombosis.
Dr. Alkhouri and associates performed an integrated analysis of the PLUS 1 trial (Japan, October 2013–May 2014) and the L-PLUS 2 (global, June 2015–April 2017). Participants were adults with Child-Pugh Class A or B chronic liver disease and baseline platelet counts under 50 x 109 per L who were scheduled for invasive procedures. Of the 270 patients, 95 had hepatocellular carcinoma. Patients were randomly assigned on a one-to-one basis to receive either lusutrombopag (3 mg) or placebo daily for up to 7 days before procedures. The primary endpoint was the percentage of patients in the per-protocol population who did not need a platelet transfusion before the invasive procedure or rescue therapy within 7 days afterward.
The treatment and placebo arms were similar except that patients with hepatocellular carcinoma were about 10 years older on average. In patients with hepatocellular carcinoma, 60.5% more lusutrombopag recipients than placebo recipients met the primary endpoint, and rates of bleeding-related adverse events were 9.1% and 15.7%, respectively. In patients with other chronic liver diseases, 52.6% more lusutrombopag recipients met the primary endpoint. Rates of bleeding-related adverse events were 5% and 10.6%.
“Approximately 88% of patients with hepatocellular carcinoma underwent a liver-related procedure, compared with approximately 10% of patients without hepatocellular carcinoma,” the investigators wrote. “This is significant because ablations or transcatheter arterial chemoembolizations can be associated with serious bleeding complications. It is clinically important that, given the greater number of liver-related procedures, the incidence of bleeding-related adverse events was lower in patients treated with lusutrombopag than placebo.”
Imaging after the procedures confirmed low rates of thromboses in both groups and subgroups. Four patients developed portal vein thromboses, including two lusutrombopag recipients (one of whom had hepatocellular carcinoma) and two placebo recipients without hepatocellular carcinoma.
These trials excluded patients undergoing major surgical procedures and those with decompensated cirrhosis; portal vein thrombosis; hematopoietic tumors; aplastic anemia; myelodysplastic syndrome; myelofibrosis; liver transplantation; splenectomy; and thrombocytopenia that was congenital, autoimmune, or drug induced. “A limitation of this study was the high rate of protocol violations related to platelet transfusions,” the researchers noted. “A number of patients [42 in all] were excluded from the per-protocol population owing to receipt of unnecessary platelet transfusions, or because they did not receive a needed platelet transfusion.”Shionogi makes lusutrombopag and sponsored the study. Dr. Alkhouri reported an advisory relationship with Shionogi and Dova Pharma. Two coinvestigators reported being employed by Shionogi. Three coinvestigators also disclosed ties to Shionogi and to several other pharmaceutical companies.
SOURCE: Alkhouri N et al. Clin Gastroenterol Hepatol. 2020 Mar 20. doi: 10.1016/j.cgh.2020.03.032.
Thrombocytopenia is of clinical concern in patients with cirrhosis, as it complicates routine patient care and results in delayed or canceled procedures due to concern for risk of bleeding. In the last few years, availability of thrombopoietin (TPO) receptor agonists have facilitated the performance of elective invasive procedures in cirrhotic patients with severe thrombocytopenia.
In this integrated analysis of data from two phase 3 studies, Alkhouri et al. demonstrated the efficacy of a novel TPO receptor agonist, lusutrombopag, in reducing bleeding events and need for platelet transfusion in cirrhotic patients undergoing invasive procedures. The risk for thrombosis-related adverse events was not increased in lusutrombopag recipients with or without HCC. Previous studies with another TPO, eltrombopag, resulted in high rate of symptomatic portal vein thrombosis. Avatrombopag, a recently approved TPO receptor agonist reported few thrombotic symptomatic events but no prospective imaging for evaluation of thrombotic events was included in the protocol. A unique strength of this study was inclusion of prospective imaging for evaluation of portal vein thrombosis. Strategic scheduling is required with use of TPO agonists. Lusutrombopag can be given orally in convenient daily doses and provides a 7-10-day procedural window for scheduling and performing elective invasive procedures. However, because of several days of lag period for platelet production, these agents cannot be used for emergent cases.
Gagan K. Sood, MD, AGAF, FAASLD, is an associate professor of medicine and surgery, division of gastroenterology and hepatology and division of abdominal transplantation, Baylor College of Medicine, Houston. He has no conflicts of interest.
Thrombocytopenia is of clinical concern in patients with cirrhosis, as it complicates routine patient care and results in delayed or canceled procedures due to concern for risk of bleeding. In the last few years, availability of thrombopoietin (TPO) receptor agonists have facilitated the performance of elective invasive procedures in cirrhotic patients with severe thrombocytopenia.
In this integrated analysis of data from two phase 3 studies, Alkhouri et al. demonstrated the efficacy of a novel TPO receptor agonist, lusutrombopag, in reducing bleeding events and need for platelet transfusion in cirrhotic patients undergoing invasive procedures. The risk for thrombosis-related adverse events was not increased in lusutrombopag recipients with or without HCC. Previous studies with another TPO, eltrombopag, resulted in high rate of symptomatic portal vein thrombosis. Avatrombopag, a recently approved TPO receptor agonist reported few thrombotic symptomatic events but no prospective imaging for evaluation of thrombotic events was included in the protocol. A unique strength of this study was inclusion of prospective imaging for evaluation of portal vein thrombosis. Strategic scheduling is required with use of TPO agonists. Lusutrombopag can be given orally in convenient daily doses and provides a 7-10-day procedural window for scheduling and performing elective invasive procedures. However, because of several days of lag period for platelet production, these agents cannot be used for emergent cases.
Gagan K. Sood, MD, AGAF, FAASLD, is an associate professor of medicine and surgery, division of gastroenterology and hepatology and division of abdominal transplantation, Baylor College of Medicine, Houston. He has no conflicts of interest.
Thrombocytopenia is of clinical concern in patients with cirrhosis, as it complicates routine patient care and results in delayed or canceled procedures due to concern for risk of bleeding. In the last few years, availability of thrombopoietin (TPO) receptor agonists have facilitated the performance of elective invasive procedures in cirrhotic patients with severe thrombocytopenia.
In this integrated analysis of data from two phase 3 studies, Alkhouri et al. demonstrated the efficacy of a novel TPO receptor agonist, lusutrombopag, in reducing bleeding events and need for platelet transfusion in cirrhotic patients undergoing invasive procedures. The risk for thrombosis-related adverse events was not increased in lusutrombopag recipients with or without HCC. Previous studies with another TPO, eltrombopag, resulted in high rate of symptomatic portal vein thrombosis. Avatrombopag, a recently approved TPO receptor agonist reported few thrombotic symptomatic events but no prospective imaging for evaluation of thrombotic events was included in the protocol. A unique strength of this study was inclusion of prospective imaging for evaluation of portal vein thrombosis. Strategic scheduling is required with use of TPO agonists. Lusutrombopag can be given orally in convenient daily doses and provides a 7-10-day procedural window for scheduling and performing elective invasive procedures. However, because of several days of lag period for platelet production, these agents cannot be used for emergent cases.
Gagan K. Sood, MD, AGAF, FAASLD, is an associate professor of medicine and surgery, division of gastroenterology and hepatology and division of abdominal transplantation, Baylor College of Medicine, Houston. He has no conflicts of interest.
For patients with severe thrombocytopenia and chronic liver diseases, including hepatocellular carcinoma, treatment with lusutrombopag prior to invasive procedures significantly decreased the need for platelet transfusions without increasing the need for rescue treatment for bleeding or the rate of thromboembolic events.
In a post hoc analysis of data from 270 patients in two manufacturer-sponsored, multicenter, randomized, double-blind, placebo-controlled, phase 3 trials, significantly more lusutrombopag recipients met the primary efficacy endpoint, including patients with hepatocellular carcinoma (68.0% vs. 8.9% in the placebo group; P < .0001) and those without it (77.0% vs. 21.6%; P < .0001). Rates of treatment-emergent adverse events were similar between the lusutrombopag and placebo groups, and patients with hepatocellular carcinoma were not at increased risk for thrombosis, Naim Alkhouri, MD, of Texas Liver Institute in San Antonio, and associates wrote in Clinical Gastroenterology and Hepatology.
Platelet transfusion is the treatment mainstay for patients with thrombocytopenia related to cirrhosis who are undergoing invasive procedures, but its effects are short-lived, and at least one in five transfusions fails. Thrombopoietin agonists such as lusutrombopag are efficacious and approved in this setting, but they can be prothrombotic, particularly in patients with hepatocellular carcinoma, who already are at heightened risk for portal vein thrombosis.
Dr. Alkhouri and associates performed an integrated analysis of the PLUS 1 trial (Japan, October 2013–May 2014) and the L-PLUS 2 (global, June 2015–April 2017). Participants were adults with Child-Pugh Class A or B chronic liver disease and baseline platelet counts under 50 x 109 per L who were scheduled for invasive procedures. Of the 270 patients, 95 had hepatocellular carcinoma. Patients were randomly assigned on a one-to-one basis to receive either lusutrombopag (3 mg) or placebo daily for up to 7 days before procedures. The primary endpoint was the percentage of patients in the per-protocol population who did not need a platelet transfusion before the invasive procedure or rescue therapy within 7 days afterward.
The treatment and placebo arms were similar except that patients with hepatocellular carcinoma were about 10 years older on average. In patients with hepatocellular carcinoma, 60.5% more lusutrombopag recipients than placebo recipients met the primary endpoint, and rates of bleeding-related adverse events were 9.1% and 15.7%, respectively. In patients with other chronic liver diseases, 52.6% more lusutrombopag recipients met the primary endpoint. Rates of bleeding-related adverse events were 5% and 10.6%.
“Approximately 88% of patients with hepatocellular carcinoma underwent a liver-related procedure, compared with approximately 10% of patients without hepatocellular carcinoma,” the investigators wrote. “This is significant because ablations or transcatheter arterial chemoembolizations can be associated with serious bleeding complications. It is clinically important that, given the greater number of liver-related procedures, the incidence of bleeding-related adverse events was lower in patients treated with lusutrombopag than placebo.”
Imaging after the procedures confirmed low rates of thromboses in both groups and subgroups. Four patients developed portal vein thromboses, including two lusutrombopag recipients (one of whom had hepatocellular carcinoma) and two placebo recipients without hepatocellular carcinoma.
These trials excluded patients undergoing major surgical procedures and those with decompensated cirrhosis; portal vein thrombosis; hematopoietic tumors; aplastic anemia; myelodysplastic syndrome; myelofibrosis; liver transplantation; splenectomy; and thrombocytopenia that was congenital, autoimmune, or drug induced. “A limitation of this study was the high rate of protocol violations related to platelet transfusions,” the researchers noted. “A number of patients [42 in all] were excluded from the per-protocol population owing to receipt of unnecessary platelet transfusions, or because they did not receive a needed platelet transfusion.”Shionogi makes lusutrombopag and sponsored the study. Dr. Alkhouri reported an advisory relationship with Shionogi and Dova Pharma. Two coinvestigators reported being employed by Shionogi. Three coinvestigators also disclosed ties to Shionogi and to several other pharmaceutical companies.
SOURCE: Alkhouri N et al. Clin Gastroenterol Hepatol. 2020 Mar 20. doi: 10.1016/j.cgh.2020.03.032.
For patients with severe thrombocytopenia and chronic liver diseases, including hepatocellular carcinoma, treatment with lusutrombopag prior to invasive procedures significantly decreased the need for platelet transfusions without increasing the need for rescue treatment for bleeding or the rate of thromboembolic events.
In a post hoc analysis of data from 270 patients in two manufacturer-sponsored, multicenter, randomized, double-blind, placebo-controlled, phase 3 trials, significantly more lusutrombopag recipients met the primary efficacy endpoint, including patients with hepatocellular carcinoma (68.0% vs. 8.9% in the placebo group; P < .0001) and those without it (77.0% vs. 21.6%; P < .0001). Rates of treatment-emergent adverse events were similar between the lusutrombopag and placebo groups, and patients with hepatocellular carcinoma were not at increased risk for thrombosis, Naim Alkhouri, MD, of Texas Liver Institute in San Antonio, and associates wrote in Clinical Gastroenterology and Hepatology.
Platelet transfusion is the treatment mainstay for patients with thrombocytopenia related to cirrhosis who are undergoing invasive procedures, but its effects are short-lived, and at least one in five transfusions fails. Thrombopoietin agonists such as lusutrombopag are efficacious and approved in this setting, but they can be prothrombotic, particularly in patients with hepatocellular carcinoma, who already are at heightened risk for portal vein thrombosis.
Dr. Alkhouri and associates performed an integrated analysis of the PLUS 1 trial (Japan, October 2013–May 2014) and the L-PLUS 2 (global, June 2015–April 2017). Participants were adults with Child-Pugh Class A or B chronic liver disease and baseline platelet counts under 50 x 109 per L who were scheduled for invasive procedures. Of the 270 patients, 95 had hepatocellular carcinoma. Patients were randomly assigned on a one-to-one basis to receive either lusutrombopag (3 mg) or placebo daily for up to 7 days before procedures. The primary endpoint was the percentage of patients in the per-protocol population who did not need a platelet transfusion before the invasive procedure or rescue therapy within 7 days afterward.
The treatment and placebo arms were similar except that patients with hepatocellular carcinoma were about 10 years older on average. In patients with hepatocellular carcinoma, 60.5% more lusutrombopag recipients than placebo recipients met the primary endpoint, and rates of bleeding-related adverse events were 9.1% and 15.7%, respectively. In patients with other chronic liver diseases, 52.6% more lusutrombopag recipients met the primary endpoint. Rates of bleeding-related adverse events were 5% and 10.6%.
“Approximately 88% of patients with hepatocellular carcinoma underwent a liver-related procedure, compared with approximately 10% of patients without hepatocellular carcinoma,” the investigators wrote. “This is significant because ablations or transcatheter arterial chemoembolizations can be associated with serious bleeding complications. It is clinically important that, given the greater number of liver-related procedures, the incidence of bleeding-related adverse events was lower in patients treated with lusutrombopag than placebo.”
Imaging after the procedures confirmed low rates of thromboses in both groups and subgroups. Four patients developed portal vein thromboses, including two lusutrombopag recipients (one of whom had hepatocellular carcinoma) and two placebo recipients without hepatocellular carcinoma.
These trials excluded patients undergoing major surgical procedures and those with decompensated cirrhosis; portal vein thrombosis; hematopoietic tumors; aplastic anemia; myelodysplastic syndrome; myelofibrosis; liver transplantation; splenectomy; and thrombocytopenia that was congenital, autoimmune, or drug induced. “A limitation of this study was the high rate of protocol violations related to platelet transfusions,” the researchers noted. “A number of patients [42 in all] were excluded from the per-protocol population owing to receipt of unnecessary platelet transfusions, or because they did not receive a needed platelet transfusion.”Shionogi makes lusutrombopag and sponsored the study. Dr. Alkhouri reported an advisory relationship with Shionogi and Dova Pharma. Two coinvestigators reported being employed by Shionogi. Three coinvestigators also disclosed ties to Shionogi and to several other pharmaceutical companies.
SOURCE: Alkhouri N et al. Clin Gastroenterol Hepatol. 2020 Mar 20. doi: 10.1016/j.cgh.2020.03.032.
FROM CLINICAL GASTROENTEROLOGY AND HEPATOLOGY
Bariatric surgery achieved long-term resolution of NASH without worsening fibrosis
Bariatric surgery resolved nonalcoholic steatohepatitis (NASH) without worsening fibrosis in 84% of patients with evaluable biopsies, according to the findings of a prospective study.
The study included 180 severely or morbidly obese adults (body mass index >35 kg/m2) with NASH who underwent bariatric surgery at a center in France. Among 94 patients evaluated 5 years later, 68% had follow-up liver biopsies, of whom 84% (95% confidence interval, 73.1%-92.2%) met the primary endpoint of resolution of NASH without worsening of fibrosis. All histologic aspects of NASH had improved, median nonalcoholic fatty liver disease scores (NAS) fell from 5 (interquartile range, 4 to 5) to 1 (IQR, 0-2; P < .001), and 90% of patients achieved at least a 2-point NAS improvement. Hepatocellular ballooning also improved in 87.5% of patients. Baseline severity of NASH did not affect the chances of it resolving at 5 years. “The reduction of fibrosis [was] progressive, beginning during the first year and continuing through 5 years,” Guillaume Lassailly, MD, and associates wrote in Gastroenterology.
NASH is a priority for clinical research because of the substantial risk for subsequent cirrhosis, added Dr. Lassailly of CHU Lille (France). For NASH to resolve, most patients need to lose at least 7%-10% of their body weight, but “only 10% of patients reach this objective with lifestyle therapy at 1 year, and less than half maintain the weight loss 5 years later.” Despite ongoing drug development efforts, no medications have been approved for treating NASH. Although weight loss after bariatric surgery has been reported to resolve NASH in approximately 80% of patients at 1 year, longer-term data have been unavailable, and it has remained unclear whether bariatric surgery can slow or halt fibrosis progression.
All patients in this study had biopsy-confirmed NASH and at least a 5-year history of severe or morbid obesity as well as at least one comorbidity, such as diabetes mellitus or arterial hypertension. Patients were not heavy drinkers, and none had detectable markers of chronic liver disease.
Bariatric surgery produced a median 12-kg/m2 drop in body mass index. At 5-year follow-up, 93% of patients meeting or exceeding this threshold who had biopsies performed showed resolution of NASH without worsening of fibrosis. Furthermore, 56% of patients (95% CI, 42.4%-69.3%) had no histologic evidence of fibrosis, including 45.5% of patients who had bridging fibrosis at baseline.
Participants in this study received intensive preoperative support, including evaluations by numerous specialists, a nutrition plan, and a 6- to 12-month therapeutic education program. Bariatric surgery techniques included Roux-en-Y gastric bypass, gastric banding, and sleeve gastrectomy. A subgroup analysis linked gastric bypass to a significantly higher probability of meeting the primary endpoint, compared with gastric banding. Refusal was the most common reason for not having a follow-up biopsy, the researchers said. “Patients without liver biopsy after bariatric surgery were not significantly different from those with a histological follow-up except for a lower BMI at 1 year. Baseline fibrosis did not influence the probability of undergoing histological reevaluation at 5 years.”
Two study participants died from surgical complications within 1 month after surgery, and one patient died from cardiac dysfunction 4 years later. No fatality was deemed liver related.
The study was funded by the French Ministry of Health, Conseil Régional Nord-Pas de Calais, National de la Recherche, and the European commission (FEDER). The researchers reported having no conflicts of interest.
SOURCE: Lassailly G et al. Gastroenterology. 2020 Jun 15. doi: 10.1053/j.gastro.2020.06.006.
As obesity prevalence increases at an alarming pace, nonalcoholic steatohepatitis (NASH) has become the most common indication for liver transplantation in women and the second most common in men in the United States. Impeding the inflammation and reversing the resultant fibrosis prior to the development of end-stage liver disease and needing liver transplantation are essential goals in NASH management. The lack of Food and Drug Administration–approved pharmacotherapy triggered interest in the effect of weight loss on NASH and short-term benefits were noted.
In this article, Lassailly et al. demonstrated long-term benefits of bariatric surgery in patients with NASH. They prospectively enrolled 180 patients and histologically followed 64 patients at 1 year and 5 years postoperatively. NASH resolved in 84% of patients and fibrosis regressed in >70%. Importantly, advanced fibrosis (F3) regressed in 15/19 patients. Cirrhosis regressed to F3 in two-thirds of patients. No liver-related mortality or decompensation was observed.
These favorable outcomes embolden the practice of referring NASH patients with morbid obesity to bariatric surgery before liver disease severity becomes prohibitive of this approach. NASH pharmacotherapy may become available in the future. However, we must not forget that cardiovascular disease remains a common cause of morbidity and mortality in NASH patients.
With these study findings and previously established benefits of bariatric surgery on mitigating cardiovascular risk and treating relevant metabolic derangements (e.g., diabetes mellitus), pursuing bariatric surgery in NASH patients may be the seed that, if planted early on, can later flourish with resolution of NASH, prevention of cardiovascular disease, metabolic optimization, and potentially longer and healthier life.
Manhal J. Izzy, MD, is assistant professor of medicine, Vanderbilt Digestive Disease Center, Vanderbilt University, Nashville, Tenn.
As obesity prevalence increases at an alarming pace, nonalcoholic steatohepatitis (NASH) has become the most common indication for liver transplantation in women and the second most common in men in the United States. Impeding the inflammation and reversing the resultant fibrosis prior to the development of end-stage liver disease and needing liver transplantation are essential goals in NASH management. The lack of Food and Drug Administration–approved pharmacotherapy triggered interest in the effect of weight loss on NASH and short-term benefits were noted.
In this article, Lassailly et al. demonstrated long-term benefits of bariatric surgery in patients with NASH. They prospectively enrolled 180 patients and histologically followed 64 patients at 1 year and 5 years postoperatively. NASH resolved in 84% of patients and fibrosis regressed in >70%. Importantly, advanced fibrosis (F3) regressed in 15/19 patients. Cirrhosis regressed to F3 in two-thirds of patients. No liver-related mortality or decompensation was observed.
These favorable outcomes embolden the practice of referring NASH patients with morbid obesity to bariatric surgery before liver disease severity becomes prohibitive of this approach. NASH pharmacotherapy may become available in the future. However, we must not forget that cardiovascular disease remains a common cause of morbidity and mortality in NASH patients.
With these study findings and previously established benefits of bariatric surgery on mitigating cardiovascular risk and treating relevant metabolic derangements (e.g., diabetes mellitus), pursuing bariatric surgery in NASH patients may be the seed that, if planted early on, can later flourish with resolution of NASH, prevention of cardiovascular disease, metabolic optimization, and potentially longer and healthier life.
Manhal J. Izzy, MD, is assistant professor of medicine, Vanderbilt Digestive Disease Center, Vanderbilt University, Nashville, Tenn.
As obesity prevalence increases at an alarming pace, nonalcoholic steatohepatitis (NASH) has become the most common indication for liver transplantation in women and the second most common in men in the United States. Impeding the inflammation and reversing the resultant fibrosis prior to the development of end-stage liver disease and needing liver transplantation are essential goals in NASH management. The lack of Food and Drug Administration–approved pharmacotherapy triggered interest in the effect of weight loss on NASH and short-term benefits were noted.
In this article, Lassailly et al. demonstrated long-term benefits of bariatric surgery in patients with NASH. They prospectively enrolled 180 patients and histologically followed 64 patients at 1 year and 5 years postoperatively. NASH resolved in 84% of patients and fibrosis regressed in >70%. Importantly, advanced fibrosis (F3) regressed in 15/19 patients. Cirrhosis regressed to F3 in two-thirds of patients. No liver-related mortality or decompensation was observed.
These favorable outcomes embolden the practice of referring NASH patients with morbid obesity to bariatric surgery before liver disease severity becomes prohibitive of this approach. NASH pharmacotherapy may become available in the future. However, we must not forget that cardiovascular disease remains a common cause of morbidity and mortality in NASH patients.
With these study findings and previously established benefits of bariatric surgery on mitigating cardiovascular risk and treating relevant metabolic derangements (e.g., diabetes mellitus), pursuing bariatric surgery in NASH patients may be the seed that, if planted early on, can later flourish with resolution of NASH, prevention of cardiovascular disease, metabolic optimization, and potentially longer and healthier life.
Manhal J. Izzy, MD, is assistant professor of medicine, Vanderbilt Digestive Disease Center, Vanderbilt University, Nashville, Tenn.
Bariatric surgery resolved nonalcoholic steatohepatitis (NASH) without worsening fibrosis in 84% of patients with evaluable biopsies, according to the findings of a prospective study.
The study included 180 severely or morbidly obese adults (body mass index >35 kg/m2) with NASH who underwent bariatric surgery at a center in France. Among 94 patients evaluated 5 years later, 68% had follow-up liver biopsies, of whom 84% (95% confidence interval, 73.1%-92.2%) met the primary endpoint of resolution of NASH without worsening of fibrosis. All histologic aspects of NASH had improved, median nonalcoholic fatty liver disease scores (NAS) fell from 5 (interquartile range, 4 to 5) to 1 (IQR, 0-2; P < .001), and 90% of patients achieved at least a 2-point NAS improvement. Hepatocellular ballooning also improved in 87.5% of patients. Baseline severity of NASH did not affect the chances of it resolving at 5 years. “The reduction of fibrosis [was] progressive, beginning during the first year and continuing through 5 years,” Guillaume Lassailly, MD, and associates wrote in Gastroenterology.
NASH is a priority for clinical research because of the substantial risk for subsequent cirrhosis, added Dr. Lassailly of CHU Lille (France). For NASH to resolve, most patients need to lose at least 7%-10% of their body weight, but “only 10% of patients reach this objective with lifestyle therapy at 1 year, and less than half maintain the weight loss 5 years later.” Despite ongoing drug development efforts, no medications have been approved for treating NASH. Although weight loss after bariatric surgery has been reported to resolve NASH in approximately 80% of patients at 1 year, longer-term data have been unavailable, and it has remained unclear whether bariatric surgery can slow or halt fibrosis progression.
All patients in this study had biopsy-confirmed NASH and at least a 5-year history of severe or morbid obesity as well as at least one comorbidity, such as diabetes mellitus or arterial hypertension. Patients were not heavy drinkers, and none had detectable markers of chronic liver disease.
Bariatric surgery produced a median 12-kg/m2 drop in body mass index. At 5-year follow-up, 93% of patients meeting or exceeding this threshold who had biopsies performed showed resolution of NASH without worsening of fibrosis. Furthermore, 56% of patients (95% CI, 42.4%-69.3%) had no histologic evidence of fibrosis, including 45.5% of patients who had bridging fibrosis at baseline.
Participants in this study received intensive preoperative support, including evaluations by numerous specialists, a nutrition plan, and a 6- to 12-month therapeutic education program. Bariatric surgery techniques included Roux-en-Y gastric bypass, gastric banding, and sleeve gastrectomy. A subgroup analysis linked gastric bypass to a significantly higher probability of meeting the primary endpoint, compared with gastric banding. Refusal was the most common reason for not having a follow-up biopsy, the researchers said. “Patients without liver biopsy after bariatric surgery were not significantly different from those with a histological follow-up except for a lower BMI at 1 year. Baseline fibrosis did not influence the probability of undergoing histological reevaluation at 5 years.”
Two study participants died from surgical complications within 1 month after surgery, and one patient died from cardiac dysfunction 4 years later. No fatality was deemed liver related.
The study was funded by the French Ministry of Health, Conseil Régional Nord-Pas de Calais, National de la Recherche, and the European commission (FEDER). The researchers reported having no conflicts of interest.
SOURCE: Lassailly G et al. Gastroenterology. 2020 Jun 15. doi: 10.1053/j.gastro.2020.06.006.
Bariatric surgery resolved nonalcoholic steatohepatitis (NASH) without worsening fibrosis in 84% of patients with evaluable biopsies, according to the findings of a prospective study.
The study included 180 severely or morbidly obese adults (body mass index >35 kg/m2) with NASH who underwent bariatric surgery at a center in France. Among 94 patients evaluated 5 years later, 68% had follow-up liver biopsies, of whom 84% (95% confidence interval, 73.1%-92.2%) met the primary endpoint of resolution of NASH without worsening of fibrosis. All histologic aspects of NASH had improved, median nonalcoholic fatty liver disease scores (NAS) fell from 5 (interquartile range, 4 to 5) to 1 (IQR, 0-2; P < .001), and 90% of patients achieved at least a 2-point NAS improvement. Hepatocellular ballooning also improved in 87.5% of patients. Baseline severity of NASH did not affect the chances of it resolving at 5 years. “The reduction of fibrosis [was] progressive, beginning during the first year and continuing through 5 years,” Guillaume Lassailly, MD, and associates wrote in Gastroenterology.
NASH is a priority for clinical research because of the substantial risk for subsequent cirrhosis, added Dr. Lassailly of CHU Lille (France). For NASH to resolve, most patients need to lose at least 7%-10% of their body weight, but “only 10% of patients reach this objective with lifestyle therapy at 1 year, and less than half maintain the weight loss 5 years later.” Despite ongoing drug development efforts, no medications have been approved for treating NASH. Although weight loss after bariatric surgery has been reported to resolve NASH in approximately 80% of patients at 1 year, longer-term data have been unavailable, and it has remained unclear whether bariatric surgery can slow or halt fibrosis progression.
All patients in this study had biopsy-confirmed NASH and at least a 5-year history of severe or morbid obesity as well as at least one comorbidity, such as diabetes mellitus or arterial hypertension. Patients were not heavy drinkers, and none had detectable markers of chronic liver disease.
Bariatric surgery produced a median 12-kg/m2 drop in body mass index. At 5-year follow-up, 93% of patients meeting or exceeding this threshold who had biopsies performed showed resolution of NASH without worsening of fibrosis. Furthermore, 56% of patients (95% CI, 42.4%-69.3%) had no histologic evidence of fibrosis, including 45.5% of patients who had bridging fibrosis at baseline.
Participants in this study received intensive preoperative support, including evaluations by numerous specialists, a nutrition plan, and a 6- to 12-month therapeutic education program. Bariatric surgery techniques included Roux-en-Y gastric bypass, gastric banding, and sleeve gastrectomy. A subgroup analysis linked gastric bypass to a significantly higher probability of meeting the primary endpoint, compared with gastric banding. Refusal was the most common reason for not having a follow-up biopsy, the researchers said. “Patients without liver biopsy after bariatric surgery were not significantly different from those with a histological follow-up except for a lower BMI at 1 year. Baseline fibrosis did not influence the probability of undergoing histological reevaluation at 5 years.”
Two study participants died from surgical complications within 1 month after surgery, and one patient died from cardiac dysfunction 4 years later. No fatality was deemed liver related.
The study was funded by the French Ministry of Health, Conseil Régional Nord-Pas de Calais, National de la Recherche, and the European commission (FEDER). The researchers reported having no conflicts of interest.
SOURCE: Lassailly G et al. Gastroenterology. 2020 Jun 15. doi: 10.1053/j.gastro.2020.06.006.
FROM GASTROENTEROLOGY
Should beta-blockers be used in portal hypertension?
Dear colleagues and friends,
Thank you for your continued support of the Perspectives debates. In this edition, Dr. Guadalupe Garcia-Tsao and Dr. Marwan Ghabril discuss the rationale for and against beta-blocker therapy in portal hypertension, and ultimately highlight the nuances required for appropriate decision-making. This topic invariably generates controversy and debate, and is broadly relevant to general GI and hepatology practices. I hope you will find it as informative as I did, and I welcome your comments and suggestions for future topics at [email protected].
Charles J. Kahi, MD, MS, AGAF, professor of medicine, Indiana University, Indianapolis. He is also an associate editor for GI & Hepatology News.
Beta-blockers in portal hypertension – Yes!
BY GUADALUPE GARCIA-TSAO, MD
Portal hypertension is the main consequence of cirrhosis and is responsible for most of its complications. In compensated cirrhosis, a threshold portal pressure gradient, as determined by the hepatic venous pressure gradient (HVPG), of at least 10 mm Hg is the strongest predictor of clinical decompensation (ascites, variceal hemorrhage, or encephalopathy) which is, in turn, the main determinant of death in cirrhosis.
Portal hypertension is initially caused by an increase in intrahepatic resistance that leads to mild portal hypertension (HVPG, 5-10 mm Hg) but is then enhanced and maintained by an increase in portal venous inflow that leads to clinically significant portal hypertension (HVPG, at least 10 mm Hg).
Portal pressure can be reduced by either ameliorating intrahepatic resistance (which is mostly caused by structural changes that are difficult to reverse) and/or by decreasing portal vein blood inflow (the most modifiable pathogenic mechanism). For over 30 years, treatment of portal hypertension has been based on the use of nonselective beta-blockers (NSBB), drugs that decrease portal pressure through a reduction in splanchnic blood flow. Reduction in portal pressure is greater with NSBB (propranolol, nadolol) than with selective beta-blockers because, as demonstrated experimentally, the main portal pressure–reducing effect stems from splanchnic vasoconstriction because of beta2-adrenergic blockade. This has been confirmed in patients with cirrhosis, in whom the reduction in HVPG is greater with NSBB than with selective BB. On the other hand, carvedilol, an NSBB that also has a vasodilatory alpha1 adrenergic blocking effect, has a greater effect in reducing HVPG, compared with traditional NSBB.
A significant decrease in portal pressure has been associated with better outcomes in cirrhosis. A favorable portal pressure reduction (“response”) has been traditionally defined as a decrease in HVPG below 12 mm Hg or greater than 20% from baseline, although even decreases of 10% are associated with better outcomes. Initial studies had been focused on variceal hemorrhage, a complication that is clearly related to portal hypertension. In this setting, reducing portal pressure clearly leads to a decreased in the incidence of variceal hemorrhage and a decrease in mortality.1 More recently, the focus has been on preventing decompensation (in compensated cirrhosis) and preventing further decompensation/death (in decompensated cirrhosis).
In compensated cirrhosis, a recent meta-analysis of clinical trials of prevention of variceal hemorrhage showed that patients with varices (therefore with clinically significant portal hypertension) without ascites who were NSBB hemodynamic responders, had a reduced risk of developing not only variceal hemorrhage but also ascites and/or encephalopathy, and had lower mortality.2 More importantly, a recent seminal randomized, double-blind, placebo-controlled trial performed in patients with compensated cirrhosis and clinically significant portal hypertension with no or small varices, showed that NSBB (propranolol or carvedilol) led to a significantly lower incidence of decompensation, with ascites being the single event that was significantly lower in the NSBB group.3 This study thereby demonstrates that NSBBs not only reduce the risk of variceal hemorrhage, as previously demonstrated, but also significantly reduces the probability of developing ascites, the most common complication of cirrhosis.
In decompensated cirrhosis, a recent meta-analysis of clinical trials of prevention of variceal hemorrhage showed that patients with varices and ascites (decompensated) who were NSBB responders, had a reduced risk of developing not only variceal hemorrhage but also refractory ascites, spontaneous bacterial peritonitis and/or hepatorenal syndrome, and also had a lower mortality.2 In patients with variceal hemorrhage, the recommended therapy to prevent recurrent variceal hemorrhage is the combination of NSBB plus variceal ligation but this is based on trials that combined compensated and decompensated patients. An individual patient data meta-analysis of these trials showed that, in the group of patients with decompensated Child-Pugh class B/C cirrhosis, rebleeding and mortality were higher with ligation alone, compared with combined therapy with NSBBs and ligation, underlining that NSBB is the key element of combination therapy in these patients.4
There is a fading controversy regarding the potential for increased mortality with the use of NSBBs in patients with refractory ascites and SBP, reported in two retrospective studies.5 These studies lacked information regarding the number of patients in whom NSBBs were withdrawn before the last observation and number of patients in whom NSBBs were started in the course of follow-up. Notably, a recent meta-analysis that included these and subsequent retrospective studies, encompassing a collective of over 1,300 patients, demonstrated that NSBB use in patients with ascites is not related to increased mortality.1,4
Nevertheless, NSBBs should be used cautiously in patients with cirrhosis and ascites. Hemodynamic alterations typical of decompensated cirrhosis are maximal in patients with refractory ascites and spontaneous bacterial peritonitis and the use of NSBB in this setting could lead to worsening hemodynamics, with decreased mean arterial pressure and renal perfusion that could in turn lead to acute kidney injury and death. In studies showing a deleterious effect of NSBB, the mean arterial pressure was significantly lower in patients in the NSBB group.5 In a recent retrospective study, the beneficial effect of NSBBs in patients with refractory ascites, spontaneous bacterial peritonitis, and acute-on-chronic liver failure appeared to apply only to those with a mean arterial pressure of at least 65 mm Hg.6 This evidence has led to guideline recommendations that limit the dose of NSBB to a maximum of 160 mg/day for propranolol or 80 mg/day for nadolol in patients with ascites with close follow-up of arterial blood pressure. Carvedilol should preferably not be used. In the presence of a systolic blood pressure <90 mm Hg or acute kidney injury, NSBBs should be dose-reduced or discontinued. If a precipitant for hypotension is identified (e.g., spontaneous bacterial peritonitis), NSBB can be reinitiated once the precipitating event is resolved and hypotension/acute kidney injury has resolved.
In conclusion, NSBBs are a definite “yes” in the management of cirrhosis and portal hypertension as they prevent poor outcomes (including death) in patients with both compensated and decompensated cirrhosis. In patients with ascites and spontaneous bacterial peritonitis, NSBBs could have deleterious effects but these can be prevented by careful monitoring of blood pressure.
References
1. D’Amico G et al. Gastroenterology. 2006;131:1611-24.
2. Turco L et al. Clin Gastroenterol Hepatol. 2020;18:313-27.
3. Villanueva C et al. Lancet. 2019;393:1597-608.
4. Albillos A et al. Hepatology. 2017;66:1219-31.
5. Garcia-Tsao G. J Hepatol. 2016 Mar;64(3):532-4.
6. Tergast TL et al. Aliment Pharmacol. Ther 2019;50:696-706.
Dr. Garcia-Tsao is professor of medicine, digestive diseases; chief, digestive diseases, Veterans Affairs Connecticut Healthcare System; director, clinical and translational core, Yale Liver Center; program director, VA Connecticut Hepatitis C Resource Center, New Haven. She has no conflicts.
Can be a double-edged blade too dangerous to wield
BY MARWAN S. GHABRIL, MD, AGAF
Nonselective beta-blockers (NSBB) are a cornerstone in the primary and secondary prophylaxis of variceal bleeding in patients with cirrhosis and clinically significant portal hypertension (CSPH), defined as a hepatic venous pressure gradient (HVPG) of at least 10 mm Hg. In the absence of routine HVPG measurement in most clinical practices, NSBB therapy is targeted to a 25% heart rate reduction or target heart rate of 55-60 beats per minute. There is ample evidence supporting this indication for NSBB as summarized in the Baveno VI consensus recommendations in a wide range of liver disease severity, encompassing patients with low-risk small esophageal varices to those with large varices, stigmata, and advanced Child-Pugh class. Tasked with the contrarian perspective on NSBB use, the argument for caution hinges on observations that disease progression can shift the balance of risk and reward to NSBB minimization or avoidance. Understanding the hyperdynamic circulation in worsening portal hypertension of cirrhosis is paramount to these considerations.
The pathophysiology of portal hypertension
Portal hypertension arises as a result of both increased portal venous inflow and increased hepatic sinusoidal resistance and is characterized by splanchnic and systemic vasodilation and reduced effective systemic arterial volume. Compensatory mechanisms include systemic neurohormonal activation, increased heart rate and cardiac output, sodium and water retention (increased plasma volume), and vasoconstrictor system activation. These mechanisms suffice in restoring effective arterial volume initially but also contribute to increased splanchnic/portal inflow and portal hypertension. In advanced decompensation the cardiovascular reserve is overwhelmed with progressive systemic vasodilation, worsening sodium and water retention, vasoconstriction of vital organ vascular beds and an ineffective hyperdynamic state (tachycardia, inadequate cardiac output, and systemic hypotension). This pathophysiological state is heralded clinically by the development of worsening or refractory ascites, and belies the development of other complications of advanced cirrhosis including hyponatremia and hepatorenal syndrome.
The beneficial effects of NSBB in decreasing portal hypertension are mediated by inhibition of splanchnic vasodilation and cardiac effects (reduced heart rate and cardiac output) leading to reduced portal inflow. However, these cardiac effects can be deleterious to systemic hemodynamics in more advanced disease, particularly with acute insults that exacerbate arterial hypovolemia such as bleeding or infection. As such, blunting of sympathetic drive by NSBB carries different degrees of tolerance and risk depending on the hemodynamic reserve in the hyperdynamic state.
Reported clinical experiences
The controversy over NSBB use in advanced cirrhosis arises out of heterogeneous, commonly retrospective datasets and nonrandomized cohorts, with conflicting reports of positive, negative, or neutral effects on mortality and acute kidney injury. Not surprisingly, studies describing detrimental effects of NSBB are based on patients with strictly defined refractory ascites or those with spontaneous bacterial peritonitis.1,2 Importantly, these studies also describe significantly lower blood pressure in at-risk NSBB treated patients, This baseline hemodynamic difference is either not observed,or not explicitly compared in studies/subcohorts with decreased or unaffected mortality with NSBB use in advanced cirrhosis.2
In the largest prospective study of NSBB in cirrhosis with ascites (which used data from three randomized satavaptan trials), NSBB users and nonusers were more closely matched for baseline mean arterial pressure.3 There was no effect of NSBB on mortality but there was a 29% rate of NSBB discontinuation (i.e., intolerance) during the year of follow-up. Predictors of NSBB discontinuation were hospitalization, variceal bleeding, infection, hepatorenal syndrome, Child-Pugh class C, and refractory ascites. Furthermore, NSBB discontinuation was associated with a notable increase in mortality. Similarly, clinically driven discontinuation of NSBB was observed in half of hospitalized patients with acute-on-chronic liver failure in the prospective CANONIC study, and was also associated with significantly higher short-term mortality.
It is possible that NSBB tolerance may select patients with adequate hemodynamic reserve despite the severity of other liver decompensations. Conversely, intolerance of therapeutic NSBB may signify evolving inadequacy of hemodynamic reserve, giving rise to two distinctly different risk/benefit profiles. This double-edged blade perspective is supported by findings of impaired cardiac output in patients with refractory ascites with impaired renal perfusion, and increased wait-list mortality with NSBB use in patients with compromised global cardiac function.4,5
When is caution due?
Rather than a “therapeutic window” that is either wide open or suddenly shut, in nonhospitalized patients risk is on a continuum and there are no agreed upon liver-specific parameters that define strict barriers to NSBB treatment. Refractory ascites may not absolutely define the closure of this window but should put clinicians on notice for a patient’s vulnerability. The Baveno VI recommendations echo the need for caution, with NSBB in refractory ascites with close monitoring of blood pressure, serum sodium, and creatinine. Treatment cessation, reduction or temporary withholding, and careful reintroduction (with reversible insults) are advised in patients with systolic blood pressure <90 mm Hg, serum Na <130 mEq/L, or those with acute kidney injury.
In the absence of randomized trials that account for cirrhotic cardiomyopathy and cardiac reserve, the risks and putative nonvariceal benefits of NSBB (e.g., reducing gut bacterial translocation) are not truly defined in this patient population. We lack HVPG-based or surrogate assessments in routine practice to determine which patients are hemodynamically benefiting from NSBB therapy, or reliable indicators of imminent NSBB intolerance or risk. While the indications for NSBB may expand to prevention of decompensation, serious questions about their safety are being asked in advanced decompensation. Poor tolerance of therapeutic NSBB dosing and unquantified, but likely negative, impact on quality of life raise additional questions. In a shared decision-making partnership, the patient’s perspective on the utility, tolerance, and monitoring of NSBB therapy in preventing variceal bleeding is vital, particularly when there are endoscopic or earlier shunting alternatives. “Primum non nocere” is not a gladiatorial cry, and in the wrong patients NSBB can be a double-edged blade too dangerous to wield.
References
1. Serste T et al. Hepatology. 2010;52:1017-22.
2. Mandorfer M et al. Gastroenterology. 2014;146:1680-90 e1.
3. Bossen L et al. Hepatology. 2016;63:1968-76.
4. Giannelli V et al. J Hepatol. 2020;72:463-71.
5. Tellez L et al. J Hepatol. 2020 May 20. doi: 10.1016/j.jhep.2020.05.011.
Dr. Ghabril is a gastroenterologist with the Indiana University, Indianapolis. He has no conflicts.
Dear colleagues and friends,
Thank you for your continued support of the Perspectives debates. In this edition, Dr. Guadalupe Garcia-Tsao and Dr. Marwan Ghabril discuss the rationale for and against beta-blocker therapy in portal hypertension, and ultimately highlight the nuances required for appropriate decision-making. This topic invariably generates controversy and debate, and is broadly relevant to general GI and hepatology practices. I hope you will find it as informative as I did, and I welcome your comments and suggestions for future topics at [email protected].
Charles J. Kahi, MD, MS, AGAF, professor of medicine, Indiana University, Indianapolis. He is also an associate editor for GI & Hepatology News.
Beta-blockers in portal hypertension – Yes!
BY GUADALUPE GARCIA-TSAO, MD
Portal hypertension is the main consequence of cirrhosis and is responsible for most of its complications. In compensated cirrhosis, a threshold portal pressure gradient, as determined by the hepatic venous pressure gradient (HVPG), of at least 10 mm Hg is the strongest predictor of clinical decompensation (ascites, variceal hemorrhage, or encephalopathy) which is, in turn, the main determinant of death in cirrhosis.
Portal hypertension is initially caused by an increase in intrahepatic resistance that leads to mild portal hypertension (HVPG, 5-10 mm Hg) but is then enhanced and maintained by an increase in portal venous inflow that leads to clinically significant portal hypertension (HVPG, at least 10 mm Hg).
Portal pressure can be reduced by either ameliorating intrahepatic resistance (which is mostly caused by structural changes that are difficult to reverse) and/or by decreasing portal vein blood inflow (the most modifiable pathogenic mechanism). For over 30 years, treatment of portal hypertension has been based on the use of nonselective beta-blockers (NSBB), drugs that decrease portal pressure through a reduction in splanchnic blood flow. Reduction in portal pressure is greater with NSBB (propranolol, nadolol) than with selective beta-blockers because, as demonstrated experimentally, the main portal pressure–reducing effect stems from splanchnic vasoconstriction because of beta2-adrenergic blockade. This has been confirmed in patients with cirrhosis, in whom the reduction in HVPG is greater with NSBB than with selective BB. On the other hand, carvedilol, an NSBB that also has a vasodilatory alpha1 adrenergic blocking effect, has a greater effect in reducing HVPG, compared with traditional NSBB.
A significant decrease in portal pressure has been associated with better outcomes in cirrhosis. A favorable portal pressure reduction (“response”) has been traditionally defined as a decrease in HVPG below 12 mm Hg or greater than 20% from baseline, although even decreases of 10% are associated with better outcomes. Initial studies had been focused on variceal hemorrhage, a complication that is clearly related to portal hypertension. In this setting, reducing portal pressure clearly leads to a decreased in the incidence of variceal hemorrhage and a decrease in mortality.1 More recently, the focus has been on preventing decompensation (in compensated cirrhosis) and preventing further decompensation/death (in decompensated cirrhosis).
In compensated cirrhosis, a recent meta-analysis of clinical trials of prevention of variceal hemorrhage showed that patients with varices (therefore with clinically significant portal hypertension) without ascites who were NSBB hemodynamic responders, had a reduced risk of developing not only variceal hemorrhage but also ascites and/or encephalopathy, and had lower mortality.2 More importantly, a recent seminal randomized, double-blind, placebo-controlled trial performed in patients with compensated cirrhosis and clinically significant portal hypertension with no or small varices, showed that NSBB (propranolol or carvedilol) led to a significantly lower incidence of decompensation, with ascites being the single event that was significantly lower in the NSBB group.3 This study thereby demonstrates that NSBBs not only reduce the risk of variceal hemorrhage, as previously demonstrated, but also significantly reduces the probability of developing ascites, the most common complication of cirrhosis.
In decompensated cirrhosis, a recent meta-analysis of clinical trials of prevention of variceal hemorrhage showed that patients with varices and ascites (decompensated) who were NSBB responders, had a reduced risk of developing not only variceal hemorrhage but also refractory ascites, spontaneous bacterial peritonitis and/or hepatorenal syndrome, and also had a lower mortality.2 In patients with variceal hemorrhage, the recommended therapy to prevent recurrent variceal hemorrhage is the combination of NSBB plus variceal ligation but this is based on trials that combined compensated and decompensated patients. An individual patient data meta-analysis of these trials showed that, in the group of patients with decompensated Child-Pugh class B/C cirrhosis, rebleeding and mortality were higher with ligation alone, compared with combined therapy with NSBBs and ligation, underlining that NSBB is the key element of combination therapy in these patients.4
There is a fading controversy regarding the potential for increased mortality with the use of NSBBs in patients with refractory ascites and SBP, reported in two retrospective studies.5 These studies lacked information regarding the number of patients in whom NSBBs were withdrawn before the last observation and number of patients in whom NSBBs were started in the course of follow-up. Notably, a recent meta-analysis that included these and subsequent retrospective studies, encompassing a collective of over 1,300 patients, demonstrated that NSBB use in patients with ascites is not related to increased mortality.1,4
Nevertheless, NSBBs should be used cautiously in patients with cirrhosis and ascites. Hemodynamic alterations typical of decompensated cirrhosis are maximal in patients with refractory ascites and spontaneous bacterial peritonitis and the use of NSBB in this setting could lead to worsening hemodynamics, with decreased mean arterial pressure and renal perfusion that could in turn lead to acute kidney injury and death. In studies showing a deleterious effect of NSBB, the mean arterial pressure was significantly lower in patients in the NSBB group.5 In a recent retrospective study, the beneficial effect of NSBBs in patients with refractory ascites, spontaneous bacterial peritonitis, and acute-on-chronic liver failure appeared to apply only to those with a mean arterial pressure of at least 65 mm Hg.6 This evidence has led to guideline recommendations that limit the dose of NSBB to a maximum of 160 mg/day for propranolol or 80 mg/day for nadolol in patients with ascites with close follow-up of arterial blood pressure. Carvedilol should preferably not be used. In the presence of a systolic blood pressure <90 mm Hg or acute kidney injury, NSBBs should be dose-reduced or discontinued. If a precipitant for hypotension is identified (e.g., spontaneous bacterial peritonitis), NSBB can be reinitiated once the precipitating event is resolved and hypotension/acute kidney injury has resolved.
In conclusion, NSBBs are a definite “yes” in the management of cirrhosis and portal hypertension as they prevent poor outcomes (including death) in patients with both compensated and decompensated cirrhosis. In patients with ascites and spontaneous bacterial peritonitis, NSBBs could have deleterious effects but these can be prevented by careful monitoring of blood pressure.
References
1. D’Amico G et al. Gastroenterology. 2006;131:1611-24.
2. Turco L et al. Clin Gastroenterol Hepatol. 2020;18:313-27.
3. Villanueva C et al. Lancet. 2019;393:1597-608.
4. Albillos A et al. Hepatology. 2017;66:1219-31.
5. Garcia-Tsao G. J Hepatol. 2016 Mar;64(3):532-4.
6. Tergast TL et al. Aliment Pharmacol. Ther 2019;50:696-706.
Dr. Garcia-Tsao is professor of medicine, digestive diseases; chief, digestive diseases, Veterans Affairs Connecticut Healthcare System; director, clinical and translational core, Yale Liver Center; program director, VA Connecticut Hepatitis C Resource Center, New Haven. She has no conflicts.
Can be a double-edged blade too dangerous to wield
BY MARWAN S. GHABRIL, MD, AGAF
Nonselective beta-blockers (NSBB) are a cornerstone in the primary and secondary prophylaxis of variceal bleeding in patients with cirrhosis and clinically significant portal hypertension (CSPH), defined as a hepatic venous pressure gradient (HVPG) of at least 10 mm Hg. In the absence of routine HVPG measurement in most clinical practices, NSBB therapy is targeted to a 25% heart rate reduction or target heart rate of 55-60 beats per minute. There is ample evidence supporting this indication for NSBB as summarized in the Baveno VI consensus recommendations in a wide range of liver disease severity, encompassing patients with low-risk small esophageal varices to those with large varices, stigmata, and advanced Child-Pugh class. Tasked with the contrarian perspective on NSBB use, the argument for caution hinges on observations that disease progression can shift the balance of risk and reward to NSBB minimization or avoidance. Understanding the hyperdynamic circulation in worsening portal hypertension of cirrhosis is paramount to these considerations.
The pathophysiology of portal hypertension
Portal hypertension arises as a result of both increased portal venous inflow and increased hepatic sinusoidal resistance and is characterized by splanchnic and systemic vasodilation and reduced effective systemic arterial volume. Compensatory mechanisms include systemic neurohormonal activation, increased heart rate and cardiac output, sodium and water retention (increased plasma volume), and vasoconstrictor system activation. These mechanisms suffice in restoring effective arterial volume initially but also contribute to increased splanchnic/portal inflow and portal hypertension. In advanced decompensation the cardiovascular reserve is overwhelmed with progressive systemic vasodilation, worsening sodium and water retention, vasoconstriction of vital organ vascular beds and an ineffective hyperdynamic state (tachycardia, inadequate cardiac output, and systemic hypotension). This pathophysiological state is heralded clinically by the development of worsening or refractory ascites, and belies the development of other complications of advanced cirrhosis including hyponatremia and hepatorenal syndrome.
The beneficial effects of NSBB in decreasing portal hypertension are mediated by inhibition of splanchnic vasodilation and cardiac effects (reduced heart rate and cardiac output) leading to reduced portal inflow. However, these cardiac effects can be deleterious to systemic hemodynamics in more advanced disease, particularly with acute insults that exacerbate arterial hypovolemia such as bleeding or infection. As such, blunting of sympathetic drive by NSBB carries different degrees of tolerance and risk depending on the hemodynamic reserve in the hyperdynamic state.
Reported clinical experiences
The controversy over NSBB use in advanced cirrhosis arises out of heterogeneous, commonly retrospective datasets and nonrandomized cohorts, with conflicting reports of positive, negative, or neutral effects on mortality and acute kidney injury. Not surprisingly, studies describing detrimental effects of NSBB are based on patients with strictly defined refractory ascites or those with spontaneous bacterial peritonitis.1,2 Importantly, these studies also describe significantly lower blood pressure in at-risk NSBB treated patients, This baseline hemodynamic difference is either not observed,or not explicitly compared in studies/subcohorts with decreased or unaffected mortality with NSBB use in advanced cirrhosis.2
In the largest prospective study of NSBB in cirrhosis with ascites (which used data from three randomized satavaptan trials), NSBB users and nonusers were more closely matched for baseline mean arterial pressure.3 There was no effect of NSBB on mortality but there was a 29% rate of NSBB discontinuation (i.e., intolerance) during the year of follow-up. Predictors of NSBB discontinuation were hospitalization, variceal bleeding, infection, hepatorenal syndrome, Child-Pugh class C, and refractory ascites. Furthermore, NSBB discontinuation was associated with a notable increase in mortality. Similarly, clinically driven discontinuation of NSBB was observed in half of hospitalized patients with acute-on-chronic liver failure in the prospective CANONIC study, and was also associated with significantly higher short-term mortality.
It is possible that NSBB tolerance may select patients with adequate hemodynamic reserve despite the severity of other liver decompensations. Conversely, intolerance of therapeutic NSBB may signify evolving inadequacy of hemodynamic reserve, giving rise to two distinctly different risk/benefit profiles. This double-edged blade perspective is supported by findings of impaired cardiac output in patients with refractory ascites with impaired renal perfusion, and increased wait-list mortality with NSBB use in patients with compromised global cardiac function.4,5
When is caution due?
Rather than a “therapeutic window” that is either wide open or suddenly shut, in nonhospitalized patients risk is on a continuum and there are no agreed upon liver-specific parameters that define strict barriers to NSBB treatment. Refractory ascites may not absolutely define the closure of this window but should put clinicians on notice for a patient’s vulnerability. The Baveno VI recommendations echo the need for caution, with NSBB in refractory ascites with close monitoring of blood pressure, serum sodium, and creatinine. Treatment cessation, reduction or temporary withholding, and careful reintroduction (with reversible insults) are advised in patients with systolic blood pressure <90 mm Hg, serum Na <130 mEq/L, or those with acute kidney injury.
In the absence of randomized trials that account for cirrhotic cardiomyopathy and cardiac reserve, the risks and putative nonvariceal benefits of NSBB (e.g., reducing gut bacterial translocation) are not truly defined in this patient population. We lack HVPG-based or surrogate assessments in routine practice to determine which patients are hemodynamically benefiting from NSBB therapy, or reliable indicators of imminent NSBB intolerance or risk. While the indications for NSBB may expand to prevention of decompensation, serious questions about their safety are being asked in advanced decompensation. Poor tolerance of therapeutic NSBB dosing and unquantified, but likely negative, impact on quality of life raise additional questions. In a shared decision-making partnership, the patient’s perspective on the utility, tolerance, and monitoring of NSBB therapy in preventing variceal bleeding is vital, particularly when there are endoscopic or earlier shunting alternatives. “Primum non nocere” is not a gladiatorial cry, and in the wrong patients NSBB can be a double-edged blade too dangerous to wield.
References
1. Serste T et al. Hepatology. 2010;52:1017-22.
2. Mandorfer M et al. Gastroenterology. 2014;146:1680-90 e1.
3. Bossen L et al. Hepatology. 2016;63:1968-76.
4. Giannelli V et al. J Hepatol. 2020;72:463-71.
5. Tellez L et al. J Hepatol. 2020 May 20. doi: 10.1016/j.jhep.2020.05.011.
Dr. Ghabril is a gastroenterologist with the Indiana University, Indianapolis. He has no conflicts.
Dear colleagues and friends,
Thank you for your continued support of the Perspectives debates. In this edition, Dr. Guadalupe Garcia-Tsao and Dr. Marwan Ghabril discuss the rationale for and against beta-blocker therapy in portal hypertension, and ultimately highlight the nuances required for appropriate decision-making. This topic invariably generates controversy and debate, and is broadly relevant to general GI and hepatology practices. I hope you will find it as informative as I did, and I welcome your comments and suggestions for future topics at [email protected].
Charles J. Kahi, MD, MS, AGAF, professor of medicine, Indiana University, Indianapolis. He is also an associate editor for GI & Hepatology News.
Beta-blockers in portal hypertension – Yes!
BY GUADALUPE GARCIA-TSAO, MD
Portal hypertension is the main consequence of cirrhosis and is responsible for most of its complications. In compensated cirrhosis, a threshold portal pressure gradient, as determined by the hepatic venous pressure gradient (HVPG), of at least 10 mm Hg is the strongest predictor of clinical decompensation (ascites, variceal hemorrhage, or encephalopathy) which is, in turn, the main determinant of death in cirrhosis.
Portal hypertension is initially caused by an increase in intrahepatic resistance that leads to mild portal hypertension (HVPG, 5-10 mm Hg) but is then enhanced and maintained by an increase in portal venous inflow that leads to clinically significant portal hypertension (HVPG, at least 10 mm Hg).
Portal pressure can be reduced by either ameliorating intrahepatic resistance (which is mostly caused by structural changes that are difficult to reverse) and/or by decreasing portal vein blood inflow (the most modifiable pathogenic mechanism). For over 30 years, treatment of portal hypertension has been based on the use of nonselective beta-blockers (NSBB), drugs that decrease portal pressure through a reduction in splanchnic blood flow. Reduction in portal pressure is greater with NSBB (propranolol, nadolol) than with selective beta-blockers because, as demonstrated experimentally, the main portal pressure–reducing effect stems from splanchnic vasoconstriction because of beta2-adrenergic blockade. This has been confirmed in patients with cirrhosis, in whom the reduction in HVPG is greater with NSBB than with selective BB. On the other hand, carvedilol, an NSBB that also has a vasodilatory alpha1 adrenergic blocking effect, has a greater effect in reducing HVPG, compared with traditional NSBB.
A significant decrease in portal pressure has been associated with better outcomes in cirrhosis. A favorable portal pressure reduction (“response”) has been traditionally defined as a decrease in HVPG below 12 mm Hg or greater than 20% from baseline, although even decreases of 10% are associated with better outcomes. Initial studies had been focused on variceal hemorrhage, a complication that is clearly related to portal hypertension. In this setting, reducing portal pressure clearly leads to a decreased in the incidence of variceal hemorrhage and a decrease in mortality.1 More recently, the focus has been on preventing decompensation (in compensated cirrhosis) and preventing further decompensation/death (in decompensated cirrhosis).
In compensated cirrhosis, a recent meta-analysis of clinical trials of prevention of variceal hemorrhage showed that patients with varices (therefore with clinically significant portal hypertension) without ascites who were NSBB hemodynamic responders, had a reduced risk of developing not only variceal hemorrhage but also ascites and/or encephalopathy, and had lower mortality.2 More importantly, a recent seminal randomized, double-blind, placebo-controlled trial performed in patients with compensated cirrhosis and clinically significant portal hypertension with no or small varices, showed that NSBB (propranolol or carvedilol) led to a significantly lower incidence of decompensation, with ascites being the single event that was significantly lower in the NSBB group.3 This study thereby demonstrates that NSBBs not only reduce the risk of variceal hemorrhage, as previously demonstrated, but also significantly reduces the probability of developing ascites, the most common complication of cirrhosis.
In decompensated cirrhosis, a recent meta-analysis of clinical trials of prevention of variceal hemorrhage showed that patients with varices and ascites (decompensated) who were NSBB responders, had a reduced risk of developing not only variceal hemorrhage but also refractory ascites, spontaneous bacterial peritonitis and/or hepatorenal syndrome, and also had a lower mortality.2 In patients with variceal hemorrhage, the recommended therapy to prevent recurrent variceal hemorrhage is the combination of NSBB plus variceal ligation but this is based on trials that combined compensated and decompensated patients. An individual patient data meta-analysis of these trials showed that, in the group of patients with decompensated Child-Pugh class B/C cirrhosis, rebleeding and mortality were higher with ligation alone, compared with combined therapy with NSBBs and ligation, underlining that NSBB is the key element of combination therapy in these patients.4
There is a fading controversy regarding the potential for increased mortality with the use of NSBBs in patients with refractory ascites and SBP, reported in two retrospective studies.5 These studies lacked information regarding the number of patients in whom NSBBs were withdrawn before the last observation and number of patients in whom NSBBs were started in the course of follow-up. Notably, a recent meta-analysis that included these and subsequent retrospective studies, encompassing a collective of over 1,300 patients, demonstrated that NSBB use in patients with ascites is not related to increased mortality.1,4
Nevertheless, NSBBs should be used cautiously in patients with cirrhosis and ascites. Hemodynamic alterations typical of decompensated cirrhosis are maximal in patients with refractory ascites and spontaneous bacterial peritonitis and the use of NSBB in this setting could lead to worsening hemodynamics, with decreased mean arterial pressure and renal perfusion that could in turn lead to acute kidney injury and death. In studies showing a deleterious effect of NSBB, the mean arterial pressure was significantly lower in patients in the NSBB group.5 In a recent retrospective study, the beneficial effect of NSBBs in patients with refractory ascites, spontaneous bacterial peritonitis, and acute-on-chronic liver failure appeared to apply only to those with a mean arterial pressure of at least 65 mm Hg.6 This evidence has led to guideline recommendations that limit the dose of NSBB to a maximum of 160 mg/day for propranolol or 80 mg/day for nadolol in patients with ascites with close follow-up of arterial blood pressure. Carvedilol should preferably not be used. In the presence of a systolic blood pressure <90 mm Hg or acute kidney injury, NSBBs should be dose-reduced or discontinued. If a precipitant for hypotension is identified (e.g., spontaneous bacterial peritonitis), NSBB can be reinitiated once the precipitating event is resolved and hypotension/acute kidney injury has resolved.
In conclusion, NSBBs are a definite “yes” in the management of cirrhosis and portal hypertension as they prevent poor outcomes (including death) in patients with both compensated and decompensated cirrhosis. In patients with ascites and spontaneous bacterial peritonitis, NSBBs could have deleterious effects but these can be prevented by careful monitoring of blood pressure.
References
1. D’Amico G et al. Gastroenterology. 2006;131:1611-24.
2. Turco L et al. Clin Gastroenterol Hepatol. 2020;18:313-27.
3. Villanueva C et al. Lancet. 2019;393:1597-608.
4. Albillos A et al. Hepatology. 2017;66:1219-31.
5. Garcia-Tsao G. J Hepatol. 2016 Mar;64(3):532-4.
6. Tergast TL et al. Aliment Pharmacol. Ther 2019;50:696-706.
Dr. Garcia-Tsao is professor of medicine, digestive diseases; chief, digestive diseases, Veterans Affairs Connecticut Healthcare System; director, clinical and translational core, Yale Liver Center; program director, VA Connecticut Hepatitis C Resource Center, New Haven. She has no conflicts.
Can be a double-edged blade too dangerous to wield
BY MARWAN S. GHABRIL, MD, AGAF
Nonselective beta-blockers (NSBB) are a cornerstone in the primary and secondary prophylaxis of variceal bleeding in patients with cirrhosis and clinically significant portal hypertension (CSPH), defined as a hepatic venous pressure gradient (HVPG) of at least 10 mm Hg. In the absence of routine HVPG measurement in most clinical practices, NSBB therapy is targeted to a 25% heart rate reduction or target heart rate of 55-60 beats per minute. There is ample evidence supporting this indication for NSBB as summarized in the Baveno VI consensus recommendations in a wide range of liver disease severity, encompassing patients with low-risk small esophageal varices to those with large varices, stigmata, and advanced Child-Pugh class. Tasked with the contrarian perspective on NSBB use, the argument for caution hinges on observations that disease progression can shift the balance of risk and reward to NSBB minimization or avoidance. Understanding the hyperdynamic circulation in worsening portal hypertension of cirrhosis is paramount to these considerations.
The pathophysiology of portal hypertension
Portal hypertension arises as a result of both increased portal venous inflow and increased hepatic sinusoidal resistance and is characterized by splanchnic and systemic vasodilation and reduced effective systemic arterial volume. Compensatory mechanisms include systemic neurohormonal activation, increased heart rate and cardiac output, sodium and water retention (increased plasma volume), and vasoconstrictor system activation. These mechanisms suffice in restoring effective arterial volume initially but also contribute to increased splanchnic/portal inflow and portal hypertension. In advanced decompensation the cardiovascular reserve is overwhelmed with progressive systemic vasodilation, worsening sodium and water retention, vasoconstriction of vital organ vascular beds and an ineffective hyperdynamic state (tachycardia, inadequate cardiac output, and systemic hypotension). This pathophysiological state is heralded clinically by the development of worsening or refractory ascites, and belies the development of other complications of advanced cirrhosis including hyponatremia and hepatorenal syndrome.
The beneficial effects of NSBB in decreasing portal hypertension are mediated by inhibition of splanchnic vasodilation and cardiac effects (reduced heart rate and cardiac output) leading to reduced portal inflow. However, these cardiac effects can be deleterious to systemic hemodynamics in more advanced disease, particularly with acute insults that exacerbate arterial hypovolemia such as bleeding or infection. As such, blunting of sympathetic drive by NSBB carries different degrees of tolerance and risk depending on the hemodynamic reserve in the hyperdynamic state.
Reported clinical experiences
The controversy over NSBB use in advanced cirrhosis arises out of heterogeneous, commonly retrospective datasets and nonrandomized cohorts, with conflicting reports of positive, negative, or neutral effects on mortality and acute kidney injury. Not surprisingly, studies describing detrimental effects of NSBB are based on patients with strictly defined refractory ascites or those with spontaneous bacterial peritonitis.1,2 Importantly, these studies also describe significantly lower blood pressure in at-risk NSBB treated patients, This baseline hemodynamic difference is either not observed,or not explicitly compared in studies/subcohorts with decreased or unaffected mortality with NSBB use in advanced cirrhosis.2
In the largest prospective study of NSBB in cirrhosis with ascites (which used data from three randomized satavaptan trials), NSBB users and nonusers were more closely matched for baseline mean arterial pressure.3 There was no effect of NSBB on mortality but there was a 29% rate of NSBB discontinuation (i.e., intolerance) during the year of follow-up. Predictors of NSBB discontinuation were hospitalization, variceal bleeding, infection, hepatorenal syndrome, Child-Pugh class C, and refractory ascites. Furthermore, NSBB discontinuation was associated with a notable increase in mortality. Similarly, clinically driven discontinuation of NSBB was observed in half of hospitalized patients with acute-on-chronic liver failure in the prospective CANONIC study, and was also associated with significantly higher short-term mortality.
It is possible that NSBB tolerance may select patients with adequate hemodynamic reserve despite the severity of other liver decompensations. Conversely, intolerance of therapeutic NSBB may signify evolving inadequacy of hemodynamic reserve, giving rise to two distinctly different risk/benefit profiles. This double-edged blade perspective is supported by findings of impaired cardiac output in patients with refractory ascites with impaired renal perfusion, and increased wait-list mortality with NSBB use in patients with compromised global cardiac function.4,5
When is caution due?
Rather than a “therapeutic window” that is either wide open or suddenly shut, in nonhospitalized patients risk is on a continuum and there are no agreed upon liver-specific parameters that define strict barriers to NSBB treatment. Refractory ascites may not absolutely define the closure of this window but should put clinicians on notice for a patient’s vulnerability. The Baveno VI recommendations echo the need for caution, with NSBB in refractory ascites with close monitoring of blood pressure, serum sodium, and creatinine. Treatment cessation, reduction or temporary withholding, and careful reintroduction (with reversible insults) are advised in patients with systolic blood pressure <90 mm Hg, serum Na <130 mEq/L, or those with acute kidney injury.
In the absence of randomized trials that account for cirrhotic cardiomyopathy and cardiac reserve, the risks and putative nonvariceal benefits of NSBB (e.g., reducing gut bacterial translocation) are not truly defined in this patient population. We lack HVPG-based or surrogate assessments in routine practice to determine which patients are hemodynamically benefiting from NSBB therapy, or reliable indicators of imminent NSBB intolerance or risk. While the indications for NSBB may expand to prevention of decompensation, serious questions about their safety are being asked in advanced decompensation. Poor tolerance of therapeutic NSBB dosing and unquantified, but likely negative, impact on quality of life raise additional questions. In a shared decision-making partnership, the patient’s perspective on the utility, tolerance, and monitoring of NSBB therapy in preventing variceal bleeding is vital, particularly when there are endoscopic or earlier shunting alternatives. “Primum non nocere” is not a gladiatorial cry, and in the wrong patients NSBB can be a double-edged blade too dangerous to wield.
References
1. Serste T et al. Hepatology. 2010;52:1017-22.
2. Mandorfer M et al. Gastroenterology. 2014;146:1680-90 e1.
3. Bossen L et al. Hepatology. 2016;63:1968-76.
4. Giannelli V et al. J Hepatol. 2020;72:463-71.
5. Tellez L et al. J Hepatol. 2020 May 20. doi: 10.1016/j.jhep.2020.05.011.
Dr. Ghabril is a gastroenterologist with the Indiana University, Indianapolis. He has no conflicts.
Tools emerging to predict liver failure in cirrhosis
Systemic inflammation and portal hypertension are key predictors of acute-on-chronic liver failure (ACLF) in the 3 months after a hospital stay for acute decompensated cirrhosis and also of death after 12 months, a preliminary analysis of data from the PREDICT study shows.
“Before this, we never had any patient signatures to identify ACLF,” said Jonel Trebicka, MD, PhD, from the JW Goethe University Hospital in Frankfurt, Germany.
Now, Dr. Trebicka’s team has “characterized the phenotypes in pre-ACLF that will progress within 3 months,” he said in an interview. “Those with high levels of inflammatory proteins, white blood cell count, are more likely to develop ACLF.”
ACLF is a highly complex disorder that can lead liver, cardiovascular, renal, cerebral, pulmonary, intestinal, adrenal, and immune systems to fail, Dr. Trebicka explained when he discussed the analysis – published online in the Journal of Hepatology – during the virtual International Liver Congress (ILC) 2020.
The chance of survival after the onset of ACLF is low – the 28-day survival rate is 30% – and “the only treatment we have is liver transplant,” he said.
For their prospective observational study, Dr. Trebicka and his colleagues assessed 1071 participants from 48 European hospitals in 14 countries who were admitted for an episode of acute decompensation, defined as the development of ascites, hepatic encephalopathy, gastrointestinal hemorrhage, infection, or a combination thereof.
The researchers identified three distinct clinical courses for a patient hospitalized with acute decompensated cirrhosis that will help clinicians predict the development of ACLF.
At study enrollment, more than half of the patients at highest risk for ACLF had pre-ACLF and high-grade systemic inflammation. The patients at intermediate risk had unstable decompensated cirrhosis with low-grade systemic inflammation and complications related to severe portal hypertension. And those at lowest risk for ACLF had stable decompensated cirrhosis and no severe systemic inflammation or portal hypertension complications, and did not develop ACLF or another episode of acute decompensation in the subsequent 3 months.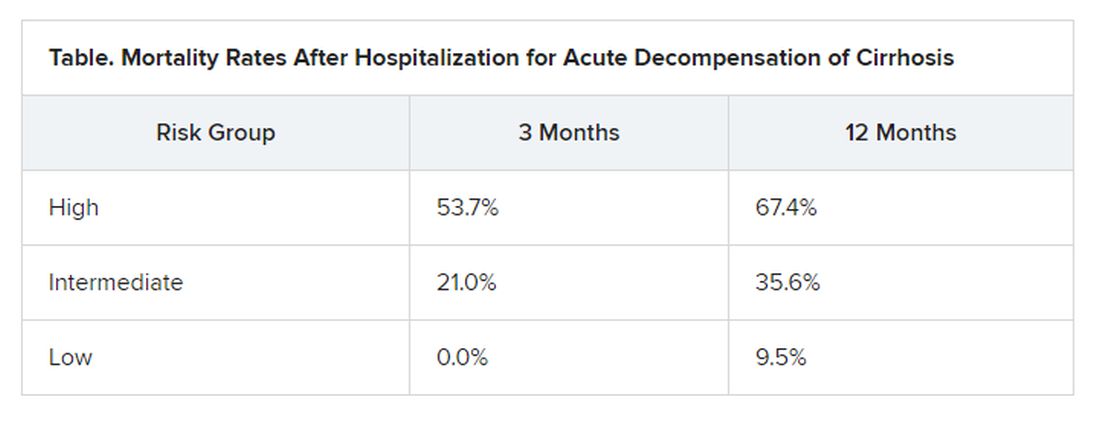
“There have been hints of possible phenotypes before – for stable and unstable ACLF – but we never had anything specific to diagnose,” Trebicka reported.
“We found that there are two main mechanisms in the development of ACLF that are most important,” he said. The first is systemic inflammation with high levels of proteins, which “leads to organ failure. This is the most striking acute mechanism.”
The second is the development of portal hypertension. “This is slower, but also very important, causing increased pressure in the portal vein, and leading to bleeding if the pressure is too great,” he said.
More tools emerging to help predict ACLF
The Albumin-functionality-test (AFT), which uses serum albumin levels to evaluate liver and kidney function, might also be useful in the prediction of ACLF and 12-month survival, according to a separate study an Italian group presented at the virtual ILC.
“Our main results are that parameters from albumin predict the development of ACLF in acute decompensated patients with the same diagnostic performance as the CLIF-AD score,” said Katja Waterstradt, PhD, from the University of Bologna in Italy.
And when the two tests are combined, diagnostic performance is increased, she added.
Dr. Trebicka has disclosed no relevant financial relationships. Dr. Waterstrand is a researcher for MedInnovation GmbH.
This article first appeared on Medscape.com.
Systemic inflammation and portal hypertension are key predictors of acute-on-chronic liver failure (ACLF) in the 3 months after a hospital stay for acute decompensated cirrhosis and also of death after 12 months, a preliminary analysis of data from the PREDICT study shows.
“Before this, we never had any patient signatures to identify ACLF,” said Jonel Trebicka, MD, PhD, from the JW Goethe University Hospital in Frankfurt, Germany.
Now, Dr. Trebicka’s team has “characterized the phenotypes in pre-ACLF that will progress within 3 months,” he said in an interview. “Those with high levels of inflammatory proteins, white blood cell count, are more likely to develop ACLF.”
ACLF is a highly complex disorder that can lead liver, cardiovascular, renal, cerebral, pulmonary, intestinal, adrenal, and immune systems to fail, Dr. Trebicka explained when he discussed the analysis – published online in the Journal of Hepatology – during the virtual International Liver Congress (ILC) 2020.
The chance of survival after the onset of ACLF is low – the 28-day survival rate is 30% – and “the only treatment we have is liver transplant,” he said.
For their prospective observational study, Dr. Trebicka and his colleagues assessed 1071 participants from 48 European hospitals in 14 countries who were admitted for an episode of acute decompensation, defined as the development of ascites, hepatic encephalopathy, gastrointestinal hemorrhage, infection, or a combination thereof.
The researchers identified three distinct clinical courses for a patient hospitalized with acute decompensated cirrhosis that will help clinicians predict the development of ACLF.
At study enrollment, more than half of the patients at highest risk for ACLF had pre-ACLF and high-grade systemic inflammation. The patients at intermediate risk had unstable decompensated cirrhosis with low-grade systemic inflammation and complications related to severe portal hypertension. And those at lowest risk for ACLF had stable decompensated cirrhosis and no severe systemic inflammation or portal hypertension complications, and did not develop ACLF or another episode of acute decompensation in the subsequent 3 months.
“There have been hints of possible phenotypes before – for stable and unstable ACLF – but we never had anything specific to diagnose,” Trebicka reported.
“We found that there are two main mechanisms in the development of ACLF that are most important,” he said. The first is systemic inflammation with high levels of proteins, which “leads to organ failure. This is the most striking acute mechanism.”
The second is the development of portal hypertension. “This is slower, but also very important, causing increased pressure in the portal vein, and leading to bleeding if the pressure is too great,” he said.
More tools emerging to help predict ACLF
The Albumin-functionality-test (AFT), which uses serum albumin levels to evaluate liver and kidney function, might also be useful in the prediction of ACLF and 12-month survival, according to a separate study an Italian group presented at the virtual ILC.
“Our main results are that parameters from albumin predict the development of ACLF in acute decompensated patients with the same diagnostic performance as the CLIF-AD score,” said Katja Waterstradt, PhD, from the University of Bologna in Italy.
And when the two tests are combined, diagnostic performance is increased, she added.
Dr. Trebicka has disclosed no relevant financial relationships. Dr. Waterstrand is a researcher for MedInnovation GmbH.
This article first appeared on Medscape.com.
Systemic inflammation and portal hypertension are key predictors of acute-on-chronic liver failure (ACLF) in the 3 months after a hospital stay for acute decompensated cirrhosis and also of death after 12 months, a preliminary analysis of data from the PREDICT study shows.
“Before this, we never had any patient signatures to identify ACLF,” said Jonel Trebicka, MD, PhD, from the JW Goethe University Hospital in Frankfurt, Germany.
Now, Dr. Trebicka’s team has “characterized the phenotypes in pre-ACLF that will progress within 3 months,” he said in an interview. “Those with high levels of inflammatory proteins, white blood cell count, are more likely to develop ACLF.”
ACLF is a highly complex disorder that can lead liver, cardiovascular, renal, cerebral, pulmonary, intestinal, adrenal, and immune systems to fail, Dr. Trebicka explained when he discussed the analysis – published online in the Journal of Hepatology – during the virtual International Liver Congress (ILC) 2020.
The chance of survival after the onset of ACLF is low – the 28-day survival rate is 30% – and “the only treatment we have is liver transplant,” he said.
For their prospective observational study, Dr. Trebicka and his colleagues assessed 1071 participants from 48 European hospitals in 14 countries who were admitted for an episode of acute decompensation, defined as the development of ascites, hepatic encephalopathy, gastrointestinal hemorrhage, infection, or a combination thereof.
The researchers identified three distinct clinical courses for a patient hospitalized with acute decompensated cirrhosis that will help clinicians predict the development of ACLF.
At study enrollment, more than half of the patients at highest risk for ACLF had pre-ACLF and high-grade systemic inflammation. The patients at intermediate risk had unstable decompensated cirrhosis with low-grade systemic inflammation and complications related to severe portal hypertension. And those at lowest risk for ACLF had stable decompensated cirrhosis and no severe systemic inflammation or portal hypertension complications, and did not develop ACLF or another episode of acute decompensation in the subsequent 3 months.
“There have been hints of possible phenotypes before – for stable and unstable ACLF – but we never had anything specific to diagnose,” Trebicka reported.
“We found that there are two main mechanisms in the development of ACLF that are most important,” he said. The first is systemic inflammation with high levels of proteins, which “leads to organ failure. This is the most striking acute mechanism.”
The second is the development of portal hypertension. “This is slower, but also very important, causing increased pressure in the portal vein, and leading to bleeding if the pressure is too great,” he said.
More tools emerging to help predict ACLF
The Albumin-functionality-test (AFT), which uses serum albumin levels to evaluate liver and kidney function, might also be useful in the prediction of ACLF and 12-month survival, according to a separate study an Italian group presented at the virtual ILC.
“Our main results are that parameters from albumin predict the development of ACLF in acute decompensated patients with the same diagnostic performance as the CLIF-AD score,” said Katja Waterstradt, PhD, from the University of Bologna in Italy.
And when the two tests are combined, diagnostic performance is increased, she added.
Dr. Trebicka has disclosed no relevant financial relationships. Dr. Waterstrand is a researcher for MedInnovation GmbH.
This article first appeared on Medscape.com.