User login
Logistical hassles hinder lifesaving lung cancer screenings
Screening high-risk populations for lung cancer saves lives. The National Lung Screening Trial (NLST) demonstrated a 20% relative reduction in lung cancer mortality with annual screening over 3 years with low-dose CT as compared with x-rays. The NELSON trial found a higher benefit: Men at high risk for lung cancer had a 26% reduced risk of dying from lung cancer and women had a 61% reduced risk over 10 years. However,
There are many reasons, but I submit that at least one hurdle is related to the difficulties associated with ordering the low-dose CT in the electronic medical record (EMR) and following the results. The rules and regulations around lung cancer screening are complex. First, the ordering provider must be able to determine if the patient is eligible for screening and has insurance coverage – a complicated procedure, which is constantly in flux, and is based on age, smoking history, smoke-free interval, and type of insurance coverage. Most EMRs do not have a way of flagging high-risk individuals, and clinic coordinators (for those practices that have one) are often put in charge of determining eligibility.
Secondly, the health care provider must order the scan. Unlike mammography, people must have a prescreening visit with a physician or other health care provider – a visit which is poorly compensated, and often must be supported by the institution. Many EMRs also do not have a smooth mechanism to make sure all the “boxes have been checked” before the scan can be ordered. Is there a complete smoking history? Has the patient had their prescreening visit? Has the patient been counseled regarding tobacco use? Has eligibility for insurance payment been confirmed?
Coordinating follow-up is cumbersome. Abnormal findings are common and usually nonmalignant, but must be followed up, and the follow-up recommendations are complicated and are based upon the appearance of the finding. This may be difficult for a general practitioner, so referrals to pulmonologists are often scheduled. Best practices state the patient be followed in a multidisciplinary pulmonary module clinic, but again, most multidisciplinary pulmonary module clinics are found in the academic setting.
All this involves a lot of back-and-forth for the patient: First to see their primary care physician, then to see a pulmonologist or other health care provider for the counseling regarding risks and benefits of screening and the importance of smoking cessation, and then a visit to a radiologist as well as a visit to a smoking cessation clinic, then a return follow-up visit. Academic medical centers and NCI-approved cancer centers often have these procedures worked out, but many private or smaller practices do not. Yes, the local IT folks can modify an EMR, but in a small practice, there are other “more important” problems that take precedence.
Would coupling lung cancer screening with breast cancer scanning help? One study followed 874 women who attended mammographic screening and found that over 11% were at high risk for lung cancer. This would appear to be an ideal “teaching moment” to educate the importance of lung cancer screening to women. It could also cut down on some of the logistical issues associated with lung cancer screening, particularly if a health care provider and coordinator were immediately available for counseling and eligibility determination at the time of the mammography visit. The radiology clinic staff could schedule a scan and return visit while the patient was still in the mammography suite.
Of course, the logistical hassle is just one of many associated with lung cancer screening. The internal stigma the patient may experience about their smoking history, the unconscious bias on the part of many health professionals, the many other screening and prevention regulations providers are now required to follow, the institution’s reluctance to support a screening program, the rushed pace in many clinics: These all certainly contribute to the problem. However, we have overcome all of these issues when it comes to mammography, such as work flow, stigma, logistical issues, seamless incorporation of ordering scans and referrals to specialists in the EMR, etc. We can and must do the same for our patients at high risk for lung cancer, and routine scheduling of mammograms and low-dose CTs may help.
Dr. Schiller is a medical oncologist and founding member of Oncologists United for Climate and Health. She is a former board member of the International Association for the Study of Lung Cancer and a current board member of the Lung Cancer Research Foundation. Ivy Elkins, cofounder of EGFR Resisters, a patient, survivor, and caregiver advocacy group, contributed to this article.
Screening high-risk populations for lung cancer saves lives. The National Lung Screening Trial (NLST) demonstrated a 20% relative reduction in lung cancer mortality with annual screening over 3 years with low-dose CT as compared with x-rays. The NELSON trial found a higher benefit: Men at high risk for lung cancer had a 26% reduced risk of dying from lung cancer and women had a 61% reduced risk over 10 years. However,
There are many reasons, but I submit that at least one hurdle is related to the difficulties associated with ordering the low-dose CT in the electronic medical record (EMR) and following the results. The rules and regulations around lung cancer screening are complex. First, the ordering provider must be able to determine if the patient is eligible for screening and has insurance coverage – a complicated procedure, which is constantly in flux, and is based on age, smoking history, smoke-free interval, and type of insurance coverage. Most EMRs do not have a way of flagging high-risk individuals, and clinic coordinators (for those practices that have one) are often put in charge of determining eligibility.
Secondly, the health care provider must order the scan. Unlike mammography, people must have a prescreening visit with a physician or other health care provider – a visit which is poorly compensated, and often must be supported by the institution. Many EMRs also do not have a smooth mechanism to make sure all the “boxes have been checked” before the scan can be ordered. Is there a complete smoking history? Has the patient had their prescreening visit? Has the patient been counseled regarding tobacco use? Has eligibility for insurance payment been confirmed?
Coordinating follow-up is cumbersome. Abnormal findings are common and usually nonmalignant, but must be followed up, and the follow-up recommendations are complicated and are based upon the appearance of the finding. This may be difficult for a general practitioner, so referrals to pulmonologists are often scheduled. Best practices state the patient be followed in a multidisciplinary pulmonary module clinic, but again, most multidisciplinary pulmonary module clinics are found in the academic setting.
All this involves a lot of back-and-forth for the patient: First to see their primary care physician, then to see a pulmonologist or other health care provider for the counseling regarding risks and benefits of screening and the importance of smoking cessation, and then a visit to a radiologist as well as a visit to a smoking cessation clinic, then a return follow-up visit. Academic medical centers and NCI-approved cancer centers often have these procedures worked out, but many private or smaller practices do not. Yes, the local IT folks can modify an EMR, but in a small practice, there are other “more important” problems that take precedence.
Would coupling lung cancer screening with breast cancer scanning help? One study followed 874 women who attended mammographic screening and found that over 11% were at high risk for lung cancer. This would appear to be an ideal “teaching moment” to educate the importance of lung cancer screening to women. It could also cut down on some of the logistical issues associated with lung cancer screening, particularly if a health care provider and coordinator were immediately available for counseling and eligibility determination at the time of the mammography visit. The radiology clinic staff could schedule a scan and return visit while the patient was still in the mammography suite.
Of course, the logistical hassle is just one of many associated with lung cancer screening. The internal stigma the patient may experience about their smoking history, the unconscious bias on the part of many health professionals, the many other screening and prevention regulations providers are now required to follow, the institution’s reluctance to support a screening program, the rushed pace in many clinics: These all certainly contribute to the problem. However, we have overcome all of these issues when it comes to mammography, such as work flow, stigma, logistical issues, seamless incorporation of ordering scans and referrals to specialists in the EMR, etc. We can and must do the same for our patients at high risk for lung cancer, and routine scheduling of mammograms and low-dose CTs may help.
Dr. Schiller is a medical oncologist and founding member of Oncologists United for Climate and Health. She is a former board member of the International Association for the Study of Lung Cancer and a current board member of the Lung Cancer Research Foundation. Ivy Elkins, cofounder of EGFR Resisters, a patient, survivor, and caregiver advocacy group, contributed to this article.
Screening high-risk populations for lung cancer saves lives. The National Lung Screening Trial (NLST) demonstrated a 20% relative reduction in lung cancer mortality with annual screening over 3 years with low-dose CT as compared with x-rays. The NELSON trial found a higher benefit: Men at high risk for lung cancer had a 26% reduced risk of dying from lung cancer and women had a 61% reduced risk over 10 years. However,
There are many reasons, but I submit that at least one hurdle is related to the difficulties associated with ordering the low-dose CT in the electronic medical record (EMR) and following the results. The rules and regulations around lung cancer screening are complex. First, the ordering provider must be able to determine if the patient is eligible for screening and has insurance coverage – a complicated procedure, which is constantly in flux, and is based on age, smoking history, smoke-free interval, and type of insurance coverage. Most EMRs do not have a way of flagging high-risk individuals, and clinic coordinators (for those practices that have one) are often put in charge of determining eligibility.
Secondly, the health care provider must order the scan. Unlike mammography, people must have a prescreening visit with a physician or other health care provider – a visit which is poorly compensated, and often must be supported by the institution. Many EMRs also do not have a smooth mechanism to make sure all the “boxes have been checked” before the scan can be ordered. Is there a complete smoking history? Has the patient had their prescreening visit? Has the patient been counseled regarding tobacco use? Has eligibility for insurance payment been confirmed?
Coordinating follow-up is cumbersome. Abnormal findings are common and usually nonmalignant, but must be followed up, and the follow-up recommendations are complicated and are based upon the appearance of the finding. This may be difficult for a general practitioner, so referrals to pulmonologists are often scheduled. Best practices state the patient be followed in a multidisciplinary pulmonary module clinic, but again, most multidisciplinary pulmonary module clinics are found in the academic setting.
All this involves a lot of back-and-forth for the patient: First to see their primary care physician, then to see a pulmonologist or other health care provider for the counseling regarding risks and benefits of screening and the importance of smoking cessation, and then a visit to a radiologist as well as a visit to a smoking cessation clinic, then a return follow-up visit. Academic medical centers and NCI-approved cancer centers often have these procedures worked out, but many private or smaller practices do not. Yes, the local IT folks can modify an EMR, but in a small practice, there are other “more important” problems that take precedence.
Would coupling lung cancer screening with breast cancer scanning help? One study followed 874 women who attended mammographic screening and found that over 11% were at high risk for lung cancer. This would appear to be an ideal “teaching moment” to educate the importance of lung cancer screening to women. It could also cut down on some of the logistical issues associated with lung cancer screening, particularly if a health care provider and coordinator were immediately available for counseling and eligibility determination at the time of the mammography visit. The radiology clinic staff could schedule a scan and return visit while the patient was still in the mammography suite.
Of course, the logistical hassle is just one of many associated with lung cancer screening. The internal stigma the patient may experience about their smoking history, the unconscious bias on the part of many health professionals, the many other screening and prevention regulations providers are now required to follow, the institution’s reluctance to support a screening program, the rushed pace in many clinics: These all certainly contribute to the problem. However, we have overcome all of these issues when it comes to mammography, such as work flow, stigma, logistical issues, seamless incorporation of ordering scans and referrals to specialists in the EMR, etc. We can and must do the same for our patients at high risk for lung cancer, and routine scheduling of mammograms and low-dose CTs may help.
Dr. Schiller is a medical oncologist and founding member of Oncologists United for Climate and Health. She is a former board member of the International Association for the Study of Lung Cancer and a current board member of the Lung Cancer Research Foundation. Ivy Elkins, cofounder of EGFR Resisters, a patient, survivor, and caregiver advocacy group, contributed to this article.
NSCLC- Clinical Presentation
Complaints of cough and fatigue
The history and findings in this case are suggestive of non–small cell lung cancer (NSCLC) large cell carcinoma.
Lung cancer is the most common cancer worldwide and has the highest mortality rate of all cancers. It comprises two major subtypes: NSCLC and small cell lung cancer (SCLC). Histologically, NSCLC is further classified as adenocarcinoma, squamous cell carcinoma, and large cell carcinoma with or without neuroendocrine features. Large cell carcinoma accounts for 9% of all cases and is frequently associated with poor prognosis. Most patients with NSCLC large cell carcinoma are older than 60 years and are diagnosed with stage III or IV disease. NSCLC large cell carcinoma appears to occur more commonly in men than in women and in patients with a history of smoking. It often presents as a large mass with central necrosis.
NSCLC is often asymptomatic in its early stages. The most frequently reported signs and symptoms of lung cancer include:
• Cough
• Chest pain
• Shortness of breath
• Coughing up blood
• Wheezing
• Hoarseness
• Recurring infections, such as bronchitis and pneumonia
• Weight loss and loss of appetite
• Fatigue
Signs and symptoms of metastatic disease may include bone pain, spinal cord impingement, or neurologic problems, such as headache, weakness or numbness of limbs, dizziness, and seizures.
All patients with NSCLC require a complete staging workup to evaluate the extent of disease because stage plays a central role in treatment selection. After physical examination and a complete blood count, a chest radiograph is often the first test performed. Chest radiographs may show a pulmonary nodule, mass, or infiltrate; mediastinal widening; atelectasis; hilar enlargement; and/or pleural effusion.
Various methods are available to confirm the diagnosis, and the method chosen may be determined at least in part by lesion location. These include:
• Bronchoscopy
• Sputum cytology
• Mediastinoscopy
• Thoracentesis
• Thoracoscopy
• Transthoracic needle biopsy (CT- or fluoroscopy-guided)
According to 2023 guidelines from the National Comprehensive Cancer Network (NCCN), the diagnosis of NSCLC large cell carcinoma requires a thoroughly sampled resected tumor with immunohistochemical stains that exclude adenocarcinoma (TTF-1, napsin A) and squamous cell (p40, p63) carcinoma. Nonresected specimens or cytology specimens are insufficient for its diagnosis. NSCLC large cell carcinoma lacks the cytologic, architectural, and histochemical features of small cell carcinoma, adenocarcinoma, or squamous cell carcinoma and is undifferentiated.
When the NSCLC histologic subtype is determined, molecular testing should be performed as part of broad molecular profiling with the goal of identifying rare driver mutations for which effective drugs may already be available or to appropriately counsel patients regarding the availability of clinical trials. NSCLC diagnostic standards include the detection of EGFR, BRAF, and MET mutations, ERBB2 (HER2) expression, and the analysis of ALK, ROS1, RET, and NTRK translocations. In addition, analysis of programmed death-ligand 1 expression is necessary to identify patients who may benefit from the use of immune checkpoint inhibitors.
Surgery combined with chemotherapy has been shown to improve the prognosis of patients with NSCLC large cell carcinoma. Preferred regimens in various lines of treatment and according to molecular characteristics can be found in the NCCN guidelines.
Karl J. D'Silva, MD, Clinical Assistant Professor, Department of Medicine, Tufts University School of Medicine, Boston; Medical Director, Department of Oncology and Hematology, Lahey Hospital and Medical Center, Peabody, Massachusetts.
Karl J. D'Silva, MD, has disclosed no relevant financial relationships.
Image Quizzes are fictional or fictionalized clinical scenarios intended to provide evidence-based educational takeaways.
The history and findings in this case are suggestive of non–small cell lung cancer (NSCLC) large cell carcinoma.
Lung cancer is the most common cancer worldwide and has the highest mortality rate of all cancers. It comprises two major subtypes: NSCLC and small cell lung cancer (SCLC). Histologically, NSCLC is further classified as adenocarcinoma, squamous cell carcinoma, and large cell carcinoma with or without neuroendocrine features. Large cell carcinoma accounts for 9% of all cases and is frequently associated with poor prognosis. Most patients with NSCLC large cell carcinoma are older than 60 years and are diagnosed with stage III or IV disease. NSCLC large cell carcinoma appears to occur more commonly in men than in women and in patients with a history of smoking. It often presents as a large mass with central necrosis.
NSCLC is often asymptomatic in its early stages. The most frequently reported signs and symptoms of lung cancer include:
• Cough
• Chest pain
• Shortness of breath
• Coughing up blood
• Wheezing
• Hoarseness
• Recurring infections, such as bronchitis and pneumonia
• Weight loss and loss of appetite
• Fatigue
Signs and symptoms of metastatic disease may include bone pain, spinal cord impingement, or neurologic problems, such as headache, weakness or numbness of limbs, dizziness, and seizures.
All patients with NSCLC require a complete staging workup to evaluate the extent of disease because stage plays a central role in treatment selection. After physical examination and a complete blood count, a chest radiograph is often the first test performed. Chest radiographs may show a pulmonary nodule, mass, or infiltrate; mediastinal widening; atelectasis; hilar enlargement; and/or pleural effusion.
Various methods are available to confirm the diagnosis, and the method chosen may be determined at least in part by lesion location. These include:
• Bronchoscopy
• Sputum cytology
• Mediastinoscopy
• Thoracentesis
• Thoracoscopy
• Transthoracic needle biopsy (CT- or fluoroscopy-guided)
According to 2023 guidelines from the National Comprehensive Cancer Network (NCCN), the diagnosis of NSCLC large cell carcinoma requires a thoroughly sampled resected tumor with immunohistochemical stains that exclude adenocarcinoma (TTF-1, napsin A) and squamous cell (p40, p63) carcinoma. Nonresected specimens or cytology specimens are insufficient for its diagnosis. NSCLC large cell carcinoma lacks the cytologic, architectural, and histochemical features of small cell carcinoma, adenocarcinoma, or squamous cell carcinoma and is undifferentiated.
When the NSCLC histologic subtype is determined, molecular testing should be performed as part of broad molecular profiling with the goal of identifying rare driver mutations for which effective drugs may already be available or to appropriately counsel patients regarding the availability of clinical trials. NSCLC diagnostic standards include the detection of EGFR, BRAF, and MET mutations, ERBB2 (HER2) expression, and the analysis of ALK, ROS1, RET, and NTRK translocations. In addition, analysis of programmed death-ligand 1 expression is necessary to identify patients who may benefit from the use of immune checkpoint inhibitors.
Surgery combined with chemotherapy has been shown to improve the prognosis of patients with NSCLC large cell carcinoma. Preferred regimens in various lines of treatment and according to molecular characteristics can be found in the NCCN guidelines.
Karl J. D'Silva, MD, Clinical Assistant Professor, Department of Medicine, Tufts University School of Medicine, Boston; Medical Director, Department of Oncology and Hematology, Lahey Hospital and Medical Center, Peabody, Massachusetts.
Karl J. D'Silva, MD, has disclosed no relevant financial relationships.
Image Quizzes are fictional or fictionalized clinical scenarios intended to provide evidence-based educational takeaways.
The history and findings in this case are suggestive of non–small cell lung cancer (NSCLC) large cell carcinoma.
Lung cancer is the most common cancer worldwide and has the highest mortality rate of all cancers. It comprises two major subtypes: NSCLC and small cell lung cancer (SCLC). Histologically, NSCLC is further classified as adenocarcinoma, squamous cell carcinoma, and large cell carcinoma with or without neuroendocrine features. Large cell carcinoma accounts for 9% of all cases and is frequently associated with poor prognosis. Most patients with NSCLC large cell carcinoma are older than 60 years and are diagnosed with stage III or IV disease. NSCLC large cell carcinoma appears to occur more commonly in men than in women and in patients with a history of smoking. It often presents as a large mass with central necrosis.
NSCLC is often asymptomatic in its early stages. The most frequently reported signs and symptoms of lung cancer include:
• Cough
• Chest pain
• Shortness of breath
• Coughing up blood
• Wheezing
• Hoarseness
• Recurring infections, such as bronchitis and pneumonia
• Weight loss and loss of appetite
• Fatigue
Signs and symptoms of metastatic disease may include bone pain, spinal cord impingement, or neurologic problems, such as headache, weakness or numbness of limbs, dizziness, and seizures.
All patients with NSCLC require a complete staging workup to evaluate the extent of disease because stage plays a central role in treatment selection. After physical examination and a complete blood count, a chest radiograph is often the first test performed. Chest radiographs may show a pulmonary nodule, mass, or infiltrate; mediastinal widening; atelectasis; hilar enlargement; and/or pleural effusion.
Various methods are available to confirm the diagnosis, and the method chosen may be determined at least in part by lesion location. These include:
• Bronchoscopy
• Sputum cytology
• Mediastinoscopy
• Thoracentesis
• Thoracoscopy
• Transthoracic needle biopsy (CT- or fluoroscopy-guided)
According to 2023 guidelines from the National Comprehensive Cancer Network (NCCN), the diagnosis of NSCLC large cell carcinoma requires a thoroughly sampled resected tumor with immunohistochemical stains that exclude adenocarcinoma (TTF-1, napsin A) and squamous cell (p40, p63) carcinoma. Nonresected specimens or cytology specimens are insufficient for its diagnosis. NSCLC large cell carcinoma lacks the cytologic, architectural, and histochemical features of small cell carcinoma, adenocarcinoma, or squamous cell carcinoma and is undifferentiated.
When the NSCLC histologic subtype is determined, molecular testing should be performed as part of broad molecular profiling with the goal of identifying rare driver mutations for which effective drugs may already be available or to appropriately counsel patients regarding the availability of clinical trials. NSCLC diagnostic standards include the detection of EGFR, BRAF, and MET mutations, ERBB2 (HER2) expression, and the analysis of ALK, ROS1, RET, and NTRK translocations. In addition, analysis of programmed death-ligand 1 expression is necessary to identify patients who may benefit from the use of immune checkpoint inhibitors.
Surgery combined with chemotherapy has been shown to improve the prognosis of patients with NSCLC large cell carcinoma. Preferred regimens in various lines of treatment and according to molecular characteristics can be found in the NCCN guidelines.
Karl J. D'Silva, MD, Clinical Assistant Professor, Department of Medicine, Tufts University School of Medicine, Boston; Medical Director, Department of Oncology and Hematology, Lahey Hospital and Medical Center, Peabody, Massachusetts.
Karl J. D'Silva, MD, has disclosed no relevant financial relationships.
Image Quizzes are fictional or fictionalized clinical scenarios intended to provide evidence-based educational takeaways.
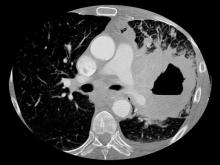
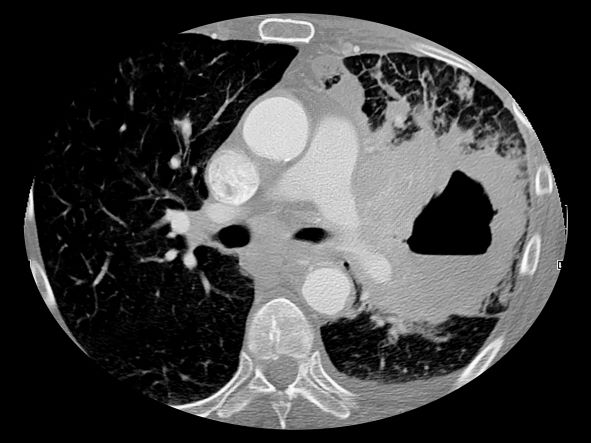
A 67-year-old White man presents to the emergency department with reports of cough, dyspnea, fatigue, hoarseness, and unintentional weight loss. The patient states that his symptoms began approximately 3 weeks earlier and have progressively worsened. In the past year, he has been treated twice for respiratory infections (bronchitis and pneumonia approximately 6 and 9 months before the current presentation, respectively). He has a 45-year history of smoking (45 pack-years). The patient's vital signs include temperature of 100.4 °F, blood pressure of 142/80 mm Hg, and pulse ox of 95%. Physical examination reveals rales in the left side of the chest and decreased breath sounds in bilateral bases of the lungs. The patient appears cachexic. He is 6 ft 2 in and weighs 163 lb.
A chest radiograph reveals a large mass in the left lung field. A subsequent CT of the chest reveals encasement of the left upper and lower lobe bronchus with extensive mediastinal lymphadenopathy and areas of necrosis. Immunohistochemical analysis of the resected tumor reveals a malignant, poorly differentiated epithelial neoplasm composed of large, atypical cells. There is no morphologic or immunohistochemical evidence of glandular, squamous, or neuroendocrine differentiation.
NSCLC- The Basics
Woman presents with cough and bronchorrhea
Bronchioalveolar cell carcinoma (BAC) is a variant of non–small cell lung cancer (NSCLC) that, in recent years, has received a new identity in some of the literature. Adenocarcinoma in situ (AIS) and minimally invasive adenocarcinoma (MIA) are relatively new entities that in some published literature have replaced the term BAC. The National Comprehensive Cancer Network recognizes these terms. AIS is defined as a localized adenocarcinoma of < 3 cm that exhibits a lepidic growth pattern, with neoplastic cells along the alveolar structures but without stromal, vascular, or pleural invasion. MIA refers to small, solitary adenocarcinomas < 3 cm with either pure lepidic growth or predominant lepidic growth with ≤ 5 mm of stromal invasion. BAC has unique epidemiologic, pathologic, and clinical features compared with other NSCLC subtypes. For example, although it is smoking-related, the relationship of BAC to smoking is less strong than with other types of NSCLC. About a third of patients with BAC are never-smokers.
There are also some unique radiographic features — its presentation may be confused with pneumonia or other inflammatory conditions in the lung, and only after a patient does not improve after a course of antibiotics should a diagnosis of BAC be considered. Unlike other types of lung cancer where chemotherapy may be the first plan of attack, surgery is often the first choice for treating BAC, particularly when there is no mediastinal node involvement (10%-25% of cases) or distal metastases (5% of cases). BAC usually harbors EGFR mutation. It is responsive to new targeted therapies for lung cancer, particularly osimertinib, afatinib, erlotinib, and gefitinib. Thus, people with BAC are good candidates for genetic testing.
Karl J. D'Silva, MD, Clinical Assistant Professor, Department of Medicine, Tufts University School of Medicine, Boston; Medical Director, Department of Oncology and Hematology, Lahey Hospital and Medical Center, Peabody, Massachusetts
Karl J. D'Silva, MD, has disclosed no relevant financial relationships.
Bronchioalveolar cell carcinoma (BAC) is a variant of non–small cell lung cancer (NSCLC) that, in recent years, has received a new identity in some of the literature. Adenocarcinoma in situ (AIS) and minimally invasive adenocarcinoma (MIA) are relatively new entities that in some published literature have replaced the term BAC. The National Comprehensive Cancer Network recognizes these terms. AIS is defined as a localized adenocarcinoma of < 3 cm that exhibits a lepidic growth pattern, with neoplastic cells along the alveolar structures but without stromal, vascular, or pleural invasion. MIA refers to small, solitary adenocarcinomas < 3 cm with either pure lepidic growth or predominant lepidic growth with ≤ 5 mm of stromal invasion. BAC has unique epidemiologic, pathologic, and clinical features compared with other NSCLC subtypes. For example, although it is smoking-related, the relationship of BAC to smoking is less strong than with other types of NSCLC. About a third of patients with BAC are never-smokers.
There are also some unique radiographic features — its presentation may be confused with pneumonia or other inflammatory conditions in the lung, and only after a patient does not improve after a course of antibiotics should a diagnosis of BAC be considered. Unlike other types of lung cancer where chemotherapy may be the first plan of attack, surgery is often the first choice for treating BAC, particularly when there is no mediastinal node involvement (10%-25% of cases) or distal metastases (5% of cases). BAC usually harbors EGFR mutation. It is responsive to new targeted therapies for lung cancer, particularly osimertinib, afatinib, erlotinib, and gefitinib. Thus, people with BAC are good candidates for genetic testing.
Karl J. D'Silva, MD, Clinical Assistant Professor, Department of Medicine, Tufts University School of Medicine, Boston; Medical Director, Department of Oncology and Hematology, Lahey Hospital and Medical Center, Peabody, Massachusetts
Karl J. D'Silva, MD, has disclosed no relevant financial relationships.
Bronchioalveolar cell carcinoma (BAC) is a variant of non–small cell lung cancer (NSCLC) that, in recent years, has received a new identity in some of the literature. Adenocarcinoma in situ (AIS) and minimally invasive adenocarcinoma (MIA) are relatively new entities that in some published literature have replaced the term BAC. The National Comprehensive Cancer Network recognizes these terms. AIS is defined as a localized adenocarcinoma of < 3 cm that exhibits a lepidic growth pattern, with neoplastic cells along the alveolar structures but without stromal, vascular, or pleural invasion. MIA refers to small, solitary adenocarcinomas < 3 cm with either pure lepidic growth or predominant lepidic growth with ≤ 5 mm of stromal invasion. BAC has unique epidemiologic, pathologic, and clinical features compared with other NSCLC subtypes. For example, although it is smoking-related, the relationship of BAC to smoking is less strong than with other types of NSCLC. About a third of patients with BAC are never-smokers.
There are also some unique radiographic features — its presentation may be confused with pneumonia or other inflammatory conditions in the lung, and only after a patient does not improve after a course of antibiotics should a diagnosis of BAC be considered. Unlike other types of lung cancer where chemotherapy may be the first plan of attack, surgery is often the first choice for treating BAC, particularly when there is no mediastinal node involvement (10%-25% of cases) or distal metastases (5% of cases). BAC usually harbors EGFR mutation. It is responsive to new targeted therapies for lung cancer, particularly osimertinib, afatinib, erlotinib, and gefitinib. Thus, people with BAC are good candidates for genetic testing.
Karl J. D'Silva, MD, Clinical Assistant Professor, Department of Medicine, Tufts University School of Medicine, Boston; Medical Director, Department of Oncology and Hematology, Lahey Hospital and Medical Center, Peabody, Massachusetts
Karl J. D'Silva, MD, has disclosed no relevant financial relationships.
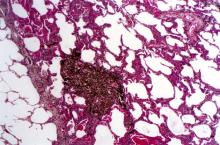
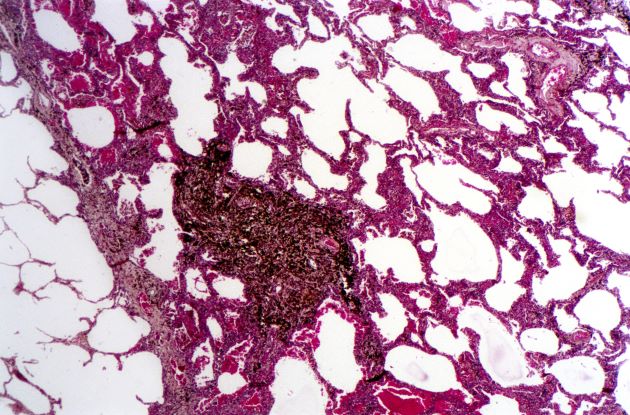
A 50-year-old woman, a never-smoker, presented with complaints of intermittent cough and shortness of breath for 3 months, associated with bronchorrhea (copious watery sputum production). She had lost 15 pounds in the past 2 months and had dyspnea on exertion for 1 month. Her pulse rate was 88/min, respiratory rate 18/min, and oxygen saturation 96% on room air. A chest x-ray (posteroanterior view) showed dense opacity in the right lower zone. Contrast-enhanced CT of the thorax showed diffuse ground-glass opacities around nodules and consolidation involving the apical and basal segments of the right lower lobe. Despite several courses of antimicrobials, bronchodilators, and IV corticosteroid therapy, the patient's condition worsened.
Breathing easier: The growing adoption of indwelling pleural catheters
Thoracic Oncology Network
Interventional Procedures Section
The management of recurrent pleural effusions is challenging. These devices have become an important treatment option in patients with malignant pleural effusions (MPE), particularly those with a nonexpandable lung (Feller-Kopman DJ, et al. Am J Respir Crit Care Med. 2018;198[7]:839) and when talc pleurodesis is unsuccessful in patients with an expandable lung (Dresler CM, et al. Chest. 2005;127[3]:909).
Over the last 5 years, studies evaluating the use of IPCs in treating nonmalignant pleural disease have proliferated. These studies have included and shown the successful treatment of pleural effusions due to end-stage renal disease, advanced heart failure (Walker SP, et al. Eur Respir J. 2022;59[2]:2101362), and cirrhosis, especially when a transjugular intrahepatic portosystemic shunt or liver transplant is not an option (Shojaee S, et al., Chest. 2019;155[3]:546). Compared with MPE, the rate of pleurodesis is generally lower and takes longer when an IPC is used to manage a nonmalignant pleural disease. Infection is the most common complication; most cases can be managed without catheter removal.
With many cited advantages, the IPC is an essential tool in the armamentarium of the chest physician and interventional radiologist. Indwelling pleural catheters have proven applications beyond MPE. When applied in a multidisciplinary fashion involving subspecialists and considering the patient’s goals, using an IPC can help achieve a crucial patient-centric goal in managing a recurrent nonmalignant pleural effusion.
Samiksha Gupta, MD
2nd Year Fellow
Sameer Kaushik Avasarala, MD
Section Member-at-Large
Thoracic Oncology Network
Interventional Procedures Section
The management of recurrent pleural effusions is challenging. These devices have become an important treatment option in patients with malignant pleural effusions (MPE), particularly those with a nonexpandable lung (Feller-Kopman DJ, et al. Am J Respir Crit Care Med. 2018;198[7]:839) and when talc pleurodesis is unsuccessful in patients with an expandable lung (Dresler CM, et al. Chest. 2005;127[3]:909).
Over the last 5 years, studies evaluating the use of IPCs in treating nonmalignant pleural disease have proliferated. These studies have included and shown the successful treatment of pleural effusions due to end-stage renal disease, advanced heart failure (Walker SP, et al. Eur Respir J. 2022;59[2]:2101362), and cirrhosis, especially when a transjugular intrahepatic portosystemic shunt or liver transplant is not an option (Shojaee S, et al., Chest. 2019;155[3]:546). Compared with MPE, the rate of pleurodesis is generally lower and takes longer when an IPC is used to manage a nonmalignant pleural disease. Infection is the most common complication; most cases can be managed without catheter removal.
With many cited advantages, the IPC is an essential tool in the armamentarium of the chest physician and interventional radiologist. Indwelling pleural catheters have proven applications beyond MPE. When applied in a multidisciplinary fashion involving subspecialists and considering the patient’s goals, using an IPC can help achieve a crucial patient-centric goal in managing a recurrent nonmalignant pleural effusion.
Samiksha Gupta, MD
2nd Year Fellow
Sameer Kaushik Avasarala, MD
Section Member-at-Large
Thoracic Oncology Network
Interventional Procedures Section
The management of recurrent pleural effusions is challenging. These devices have become an important treatment option in patients with malignant pleural effusions (MPE), particularly those with a nonexpandable lung (Feller-Kopman DJ, et al. Am J Respir Crit Care Med. 2018;198[7]:839) and when talc pleurodesis is unsuccessful in patients with an expandable lung (Dresler CM, et al. Chest. 2005;127[3]:909).
Over the last 5 years, studies evaluating the use of IPCs in treating nonmalignant pleural disease have proliferated. These studies have included and shown the successful treatment of pleural effusions due to end-stage renal disease, advanced heart failure (Walker SP, et al. Eur Respir J. 2022;59[2]:2101362), and cirrhosis, especially when a transjugular intrahepatic portosystemic shunt or liver transplant is not an option (Shojaee S, et al., Chest. 2019;155[3]:546). Compared with MPE, the rate of pleurodesis is generally lower and takes longer when an IPC is used to manage a nonmalignant pleural disease. Infection is the most common complication; most cases can be managed without catheter removal.
With many cited advantages, the IPC is an essential tool in the armamentarium of the chest physician and interventional radiologist. Indwelling pleural catheters have proven applications beyond MPE. When applied in a multidisciplinary fashion involving subspecialists and considering the patient’s goals, using an IPC can help achieve a crucial patient-centric goal in managing a recurrent nonmalignant pleural effusion.
Samiksha Gupta, MD
2nd Year Fellow
Sameer Kaushik Avasarala, MD
Section Member-at-Large
Standard-of-care therapy in lung cancer: Be open to new ideas
This transcript has been edited for clarity.
I’ll focus on some important topics related to decision-making and daily practice, and the practitioners’ thoughts from the meeting.
There’s no doubt that our outcomes are better for patients, but it’s much harder to make the best choice and I think there’s more pressure on us to make the best choice.
Topic one was the need for next-generation sequencing (NGS) testing. I’ll put it before you that every patient needs NGS testing at the time of diagnosis. It really shouldn’t be put off. How to do that is a topic for another day, but you need NGS testing.
Moving along with this, even when you’re thinking you’re going to go down the road of a checkpoint inhibitor with chemotherapy, the recent Food and Drug Administration approval for cemiplimab and chemotherapy says that you have to make sure that patients don’t have EGFR or ALK aberrations. Now, for cemiplimab, you have to make sure they don’t have ROS1 aberrations.
You need NGS testing to find those targets and give patients a targeted therapy. Even if you want to give a checkpoint inhibitor with or without chemotherapy, you need to have NGS testing.
Second, the way to get the most comprehensive analysis of targets for which there are therapeutic avenues is to do more comprehensive NGS testing, including both DNA and RNA. Not all the panels do this right now, and you really need that RNA-based testing to find all the fusions that are druggable by the current medications that we have.
Bottom line: NGS testing should be done for everybody, and you need to do the most comprehensive panel available both for DNA and RNA.
The next topic that there was great agreement on was the emergence of antibody-drug conjugates. I think everybody’s excited. All of them have shown evidence of benefit. There are varying degrees of side effects, and we’ll learn how to deal with those. They’re new drugs, they’re here, and they’re safe.
There are a couple of things to consider, though. Number one, these drugs do have chemotherapy and they have side effects from chemotherapy. I think the consensus is that when you treat patients with an antibody-drug conjugate, you need to give antiemetic regimens, at least for trastuzumab and the other deruxtecan drugs. You need to give a regimen for highly emetogenic chemotherapy as prophylactic antiemetics. I think that was a consensus thought.
Second, these drugs are making us rethink what it means to have the expression of the protein. I’m totally struck that for trastuzumab deruxtecan, patritumab deruxtecan, and datopotamab deruxtecan, the degree of protein expression is not particularly relevant, and these drugs can work in all patients. There have been cases clearly shown that datopotamab deruxtecan and patritumab deruxtecan both have benefit in patients with EGFR mutations after progression on osimertinib.
This idea of a need for overexpression, and maybe even the idea of testing, is being challenged now. These drugs seem to work as long as some protein is present. They don’t work in every patient, but they work in the vast majority. This thinking about overexpression with the antibody-drug conjugates is probably going to need to be reevaluated.
Last are some thoughts about our targeted therapies. Again, we have more targets. We have EGFR exon 20, for example, and more drugs for MET. I’d like to share a couple of thoughts on what the experts presented at the meeting.
First, although we have a bunch of new targeted agents for patients with EGFR-mutant cancers, probably the thing that’s going to change therapy now is adding chemotherapy to these agents. We may also use circulating tumor (ctDNA) to help guide us to identify which patients would be more likely to benefit from a chemotherapy with osimertinib. I see that as a trend and as a strategy that we’re likely to see move forward.
Another is in the ALK space. I know we’ve gotten very comfortable giving alectinib and brigatinib, but when you look at all the data, it points to lorlatinib perhaps being a better first-line therapy.
I think the experts thought lorlatinib would be a good drug. Yes, it has a different spectrum of side effects. The central nervous system (CNS) side effects are something we have to learn how to take care of; however, we can do that. Generally, with dose reduction, those side effects are manageable.
If you can get better outcomes in general and in patients with brain metastases, it may make some sense to displace our go-to first-line drugs, brigatinib and alectinib, with lorlatinib.
Changes in practice are happening now. There are drugs available. I urge oncologists to be open to rethinking what your standard of care is and also open to rethinking how these drugs work and to go with the data that we have.
We’re doing much better now, but the best is yet to come.
Mark G. Kris, MD, is chief of the thoracic oncology service and the William and Joy Ruane Chair in Thoracic Oncology at Memorial Sloan Kettering Cancer Center in New York City. His research interests include targeted therapies for lung cancer, multimodality therapy, the development of new anticancer drugs, and symptom management with a focus on preventing emesis. A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
I’ll focus on some important topics related to decision-making and daily practice, and the practitioners’ thoughts from the meeting.
There’s no doubt that our outcomes are better for patients, but it’s much harder to make the best choice and I think there’s more pressure on us to make the best choice.
Topic one was the need for next-generation sequencing (NGS) testing. I’ll put it before you that every patient needs NGS testing at the time of diagnosis. It really shouldn’t be put off. How to do that is a topic for another day, but you need NGS testing.
Moving along with this, even when you’re thinking you’re going to go down the road of a checkpoint inhibitor with chemotherapy, the recent Food and Drug Administration approval for cemiplimab and chemotherapy says that you have to make sure that patients don’t have EGFR or ALK aberrations. Now, for cemiplimab, you have to make sure they don’t have ROS1 aberrations.
You need NGS testing to find those targets and give patients a targeted therapy. Even if you want to give a checkpoint inhibitor with or without chemotherapy, you need to have NGS testing.
Second, the way to get the most comprehensive analysis of targets for which there are therapeutic avenues is to do more comprehensive NGS testing, including both DNA and RNA. Not all the panels do this right now, and you really need that RNA-based testing to find all the fusions that are druggable by the current medications that we have.
Bottom line: NGS testing should be done for everybody, and you need to do the most comprehensive panel available both for DNA and RNA.
The next topic that there was great agreement on was the emergence of antibody-drug conjugates. I think everybody’s excited. All of them have shown evidence of benefit. There are varying degrees of side effects, and we’ll learn how to deal with those. They’re new drugs, they’re here, and they’re safe.
There are a couple of things to consider, though. Number one, these drugs do have chemotherapy and they have side effects from chemotherapy. I think the consensus is that when you treat patients with an antibody-drug conjugate, you need to give antiemetic regimens, at least for trastuzumab and the other deruxtecan drugs. You need to give a regimen for highly emetogenic chemotherapy as prophylactic antiemetics. I think that was a consensus thought.
Second, these drugs are making us rethink what it means to have the expression of the protein. I’m totally struck that for trastuzumab deruxtecan, patritumab deruxtecan, and datopotamab deruxtecan, the degree of protein expression is not particularly relevant, and these drugs can work in all patients. There have been cases clearly shown that datopotamab deruxtecan and patritumab deruxtecan both have benefit in patients with EGFR mutations after progression on osimertinib.
This idea of a need for overexpression, and maybe even the idea of testing, is being challenged now. These drugs seem to work as long as some protein is present. They don’t work in every patient, but they work in the vast majority. This thinking about overexpression with the antibody-drug conjugates is probably going to need to be reevaluated.
Last are some thoughts about our targeted therapies. Again, we have more targets. We have EGFR exon 20, for example, and more drugs for MET. I’d like to share a couple of thoughts on what the experts presented at the meeting.
First, although we have a bunch of new targeted agents for patients with EGFR-mutant cancers, probably the thing that’s going to change therapy now is adding chemotherapy to these agents. We may also use circulating tumor (ctDNA) to help guide us to identify which patients would be more likely to benefit from a chemotherapy with osimertinib. I see that as a trend and as a strategy that we’re likely to see move forward.
Another is in the ALK space. I know we’ve gotten very comfortable giving alectinib and brigatinib, but when you look at all the data, it points to lorlatinib perhaps being a better first-line therapy.
I think the experts thought lorlatinib would be a good drug. Yes, it has a different spectrum of side effects. The central nervous system (CNS) side effects are something we have to learn how to take care of; however, we can do that. Generally, with dose reduction, those side effects are manageable.
If you can get better outcomes in general and in patients with brain metastases, it may make some sense to displace our go-to first-line drugs, brigatinib and alectinib, with lorlatinib.
Changes in practice are happening now. There are drugs available. I urge oncologists to be open to rethinking what your standard of care is and also open to rethinking how these drugs work and to go with the data that we have.
We’re doing much better now, but the best is yet to come.
Mark G. Kris, MD, is chief of the thoracic oncology service and the William and Joy Ruane Chair in Thoracic Oncology at Memorial Sloan Kettering Cancer Center in New York City. His research interests include targeted therapies for lung cancer, multimodality therapy, the development of new anticancer drugs, and symptom management with a focus on preventing emesis. A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
I’ll focus on some important topics related to decision-making and daily practice, and the practitioners’ thoughts from the meeting.
There’s no doubt that our outcomes are better for patients, but it’s much harder to make the best choice and I think there’s more pressure on us to make the best choice.
Topic one was the need for next-generation sequencing (NGS) testing. I’ll put it before you that every patient needs NGS testing at the time of diagnosis. It really shouldn’t be put off. How to do that is a topic for another day, but you need NGS testing.
Moving along with this, even when you’re thinking you’re going to go down the road of a checkpoint inhibitor with chemotherapy, the recent Food and Drug Administration approval for cemiplimab and chemotherapy says that you have to make sure that patients don’t have EGFR or ALK aberrations. Now, for cemiplimab, you have to make sure they don’t have ROS1 aberrations.
You need NGS testing to find those targets and give patients a targeted therapy. Even if you want to give a checkpoint inhibitor with or without chemotherapy, you need to have NGS testing.
Second, the way to get the most comprehensive analysis of targets for which there are therapeutic avenues is to do more comprehensive NGS testing, including both DNA and RNA. Not all the panels do this right now, and you really need that RNA-based testing to find all the fusions that are druggable by the current medications that we have.
Bottom line: NGS testing should be done for everybody, and you need to do the most comprehensive panel available both for DNA and RNA.
The next topic that there was great agreement on was the emergence of antibody-drug conjugates. I think everybody’s excited. All of them have shown evidence of benefit. There are varying degrees of side effects, and we’ll learn how to deal with those. They’re new drugs, they’re here, and they’re safe.
There are a couple of things to consider, though. Number one, these drugs do have chemotherapy and they have side effects from chemotherapy. I think the consensus is that when you treat patients with an antibody-drug conjugate, you need to give antiemetic regimens, at least for trastuzumab and the other deruxtecan drugs. You need to give a regimen for highly emetogenic chemotherapy as prophylactic antiemetics. I think that was a consensus thought.
Second, these drugs are making us rethink what it means to have the expression of the protein. I’m totally struck that for trastuzumab deruxtecan, patritumab deruxtecan, and datopotamab deruxtecan, the degree of protein expression is not particularly relevant, and these drugs can work in all patients. There have been cases clearly shown that datopotamab deruxtecan and patritumab deruxtecan both have benefit in patients with EGFR mutations after progression on osimertinib.
This idea of a need for overexpression, and maybe even the idea of testing, is being challenged now. These drugs seem to work as long as some protein is present. They don’t work in every patient, but they work in the vast majority. This thinking about overexpression with the antibody-drug conjugates is probably going to need to be reevaluated.
Last are some thoughts about our targeted therapies. Again, we have more targets. We have EGFR exon 20, for example, and more drugs for MET. I’d like to share a couple of thoughts on what the experts presented at the meeting.
First, although we have a bunch of new targeted agents for patients with EGFR-mutant cancers, probably the thing that’s going to change therapy now is adding chemotherapy to these agents. We may also use circulating tumor (ctDNA) to help guide us to identify which patients would be more likely to benefit from a chemotherapy with osimertinib. I see that as a trend and as a strategy that we’re likely to see move forward.
Another is in the ALK space. I know we’ve gotten very comfortable giving alectinib and brigatinib, but when you look at all the data, it points to lorlatinib perhaps being a better first-line therapy.
I think the experts thought lorlatinib would be a good drug. Yes, it has a different spectrum of side effects. The central nervous system (CNS) side effects are something we have to learn how to take care of; however, we can do that. Generally, with dose reduction, those side effects are manageable.
If you can get better outcomes in general and in patients with brain metastases, it may make some sense to displace our go-to first-line drugs, brigatinib and alectinib, with lorlatinib.
Changes in practice are happening now. There are drugs available. I urge oncologists to be open to rethinking what your standard of care is and also open to rethinking how these drugs work and to go with the data that we have.
We’re doing much better now, but the best is yet to come.
Mark G. Kris, MD, is chief of the thoracic oncology service and the William and Joy Ruane Chair in Thoracic Oncology at Memorial Sloan Kettering Cancer Center in New York City. His research interests include targeted therapies for lung cancer, multimodality therapy, the development of new anticancer drugs, and symptom management with a focus on preventing emesis. A version of this article first appeared on Medscape.com.
Myths about smoking, diet, alcohol, and cancer persist
FRANCE – Conducted every 5 years since 2005, the Cancer Survey documents the knowledge, perceptions, and way of life of the French people in relation to cancer. The researchers analyzed responses to telephone interviews of a representative sample of almost 5,000 individuals aged 15-85 years.
This study shows how thinking has changed over time and how difficult it is to alter preconceived notions.
Is cancer hereditary?
The report shows that 67.7% of respondents believe that cancer is a hereditary disease. Respondents were asked to explain their answer. “Data show that medical practices for cancer treatment substantiate this belief [that cancer is hereditary],” wrote the authors of the report.
“Indeed, health care professionals almost systematically ask questions about family history of breast cancer and, when a family member has been diagnosed with cancer, medical monitoring of other family members is often sought out, thus reinforcing the belief that cancer is hereditary,” they said.
Furthermore, there seems to be confusion regarding the role of genes in the development of cancer. A person can inherit cancer-predisposing genes, not cancer itself. The authors highlighted their concern that this confusion may “lead people to think that prevention measures are unnecessary because cancer is inherited.”
Misconceptions about smoking
About 41% of smokers think that the length of time one has been smoking is the biggest determining factor for developing cancer; 58.1% think the number of cigarettes smoked per day has a bigger impact.
Experts at InCA and SPF put the debate to rest, stating that prolonged exposure to carcinogenic substances is far more toxic. As for the danger threshold concerning the number of cigarettes smoked per day, respondents believed this to be 9.2 cigarettes per day, on average. They believed that the danger threshold for the number of years as an active smoker is 13.4, on average.
“The [survey] respondents clearly understand that smoking carries a risk, but many smokers think that light smoking or smoking for a short period of time doesn’t carry any risks.” Yet it is understood that even occasional tobacco consumption increases mortality.
This was not the only misconception regarding smoking and its relationship with cancer. About 34% of survey respondents agreed with the following statement: “Smoking doesn’t cause cancer unless you’re a heavy smoker and have smoked for a long time.” Furthermore, 43.3% agreed with the statement, “Pollution is more likely to cause cancer than smoking,” 54.6% think that “exercising cleans your lungs of tobacco,” and 61.6% think that “a smoker can prevent developing cancer caused by smoking if they know to quit on time.”
Overweight and obesity
Although diet and excess weight represent the third and fourth biggest avoidable cancer risk factors, after smoking and alcohol, only 30% of survey respondents knew of this link.
“Among the causes of cancer known and cited by respondents without prompting, excessive weight and obesity were mentioned only 100 times out of 12,558 responses,” highlighted the authors of the report. The explanation put forward by the authors is that discourse about diet has been more focused on diet as a protective health factor, especially in preventing cardiovascular diseases. “The link between cancer and diet is less prominent in the public space,” they noted.
Breastfeeding and cancer
About 63% of survey respondents, which for the first time included both women and men, believe that breastfeeding does not affect mothers’ risk of breast cancer, but this is a misconception. And almost 1 in 3 respondents said that breastfeeding provides health benefits for the mother.
Artificial UV rays
Exposure to UV rays, whether of natural or artificial origin, is a major risk factor for skin cancer. However, 1 in 5 people (20.9%) think that a session in a tanning bed is less harmful than sun exposure.
Daily stress
Regarding psychological factors linked to cancer, the authors noted that risk factors not supported by scientific evidence were, ironically, cited more often by respondents than proven risk factors. There is a real knowledge gap between scientific data and the beliefs of the French people. For example, “working at night” is largely not seen as a risk factor, but data show that it presents a clear risk. However, “not being able to express one’s feelings,” “having been weakened by traumatic experiences,” and “being exposed to the stress of modern life” are seen as risk factors of cancer, without any scientific evidence.
Cigarettes and e-cigarettes
About 53% of respondents agreed that “e-cigarettes are just as harmful or more harmful than traditional cigarettes.” Nicotine and the flavors in e-cigarettes are largely perceived as “very” or “extremely” harmful to the health of a person. However, the authors note that “no published study on nicotine substitutes has shown harmful effects on the health of a person, let alone determined it a risk factor for cancer. The nicotine doses in e-cigarettes are similar to traditional nicotine substitutes, and no cytotoxic effect of nicotine in its inhaled form has been found.” There seems to be confusion between dependence and risk of cancer.
Alcohol consumption
Eight of 10 respondents believe that “some people can drink a lot of alcohol all their life without ever getting cancer,” which goes against the scientific literature. The authors of the report state that the negative effects of alcohol on health seem poorly understood. Although alcohol is the second biggest cause of cancer, only a third of survey respondents cited it without having been prompted as one of the main causes of cancer. And 23.5% even think that “in terms of decreasing your risk of cancer, it’s better to drink a little wine than to drink no wine at all.”
This article was translated from the Medscape French edition. A version of this article appeared on Medscape.com.
FRANCE – Conducted every 5 years since 2005, the Cancer Survey documents the knowledge, perceptions, and way of life of the French people in relation to cancer. The researchers analyzed responses to telephone interviews of a representative sample of almost 5,000 individuals aged 15-85 years.
This study shows how thinking has changed over time and how difficult it is to alter preconceived notions.
Is cancer hereditary?
The report shows that 67.7% of respondents believe that cancer is a hereditary disease. Respondents were asked to explain their answer. “Data show that medical practices for cancer treatment substantiate this belief [that cancer is hereditary],” wrote the authors of the report.
“Indeed, health care professionals almost systematically ask questions about family history of breast cancer and, when a family member has been diagnosed with cancer, medical monitoring of other family members is often sought out, thus reinforcing the belief that cancer is hereditary,” they said.
Furthermore, there seems to be confusion regarding the role of genes in the development of cancer. A person can inherit cancer-predisposing genes, not cancer itself. The authors highlighted their concern that this confusion may “lead people to think that prevention measures are unnecessary because cancer is inherited.”
Misconceptions about smoking
About 41% of smokers think that the length of time one has been smoking is the biggest determining factor for developing cancer; 58.1% think the number of cigarettes smoked per day has a bigger impact.
Experts at InCA and SPF put the debate to rest, stating that prolonged exposure to carcinogenic substances is far more toxic. As for the danger threshold concerning the number of cigarettes smoked per day, respondents believed this to be 9.2 cigarettes per day, on average. They believed that the danger threshold for the number of years as an active smoker is 13.4, on average.
“The [survey] respondents clearly understand that smoking carries a risk, but many smokers think that light smoking or smoking for a short period of time doesn’t carry any risks.” Yet it is understood that even occasional tobacco consumption increases mortality.
This was not the only misconception regarding smoking and its relationship with cancer. About 34% of survey respondents agreed with the following statement: “Smoking doesn’t cause cancer unless you’re a heavy smoker and have smoked for a long time.” Furthermore, 43.3% agreed with the statement, “Pollution is more likely to cause cancer than smoking,” 54.6% think that “exercising cleans your lungs of tobacco,” and 61.6% think that “a smoker can prevent developing cancer caused by smoking if they know to quit on time.”
Overweight and obesity
Although diet and excess weight represent the third and fourth biggest avoidable cancer risk factors, after smoking and alcohol, only 30% of survey respondents knew of this link.
“Among the causes of cancer known and cited by respondents without prompting, excessive weight and obesity were mentioned only 100 times out of 12,558 responses,” highlighted the authors of the report. The explanation put forward by the authors is that discourse about diet has been more focused on diet as a protective health factor, especially in preventing cardiovascular diseases. “The link between cancer and diet is less prominent in the public space,” they noted.
Breastfeeding and cancer
About 63% of survey respondents, which for the first time included both women and men, believe that breastfeeding does not affect mothers’ risk of breast cancer, but this is a misconception. And almost 1 in 3 respondents said that breastfeeding provides health benefits for the mother.
Artificial UV rays
Exposure to UV rays, whether of natural or artificial origin, is a major risk factor for skin cancer. However, 1 in 5 people (20.9%) think that a session in a tanning bed is less harmful than sun exposure.
Daily stress
Regarding psychological factors linked to cancer, the authors noted that risk factors not supported by scientific evidence were, ironically, cited more often by respondents than proven risk factors. There is a real knowledge gap between scientific data and the beliefs of the French people. For example, “working at night” is largely not seen as a risk factor, but data show that it presents a clear risk. However, “not being able to express one’s feelings,” “having been weakened by traumatic experiences,” and “being exposed to the stress of modern life” are seen as risk factors of cancer, without any scientific evidence.
Cigarettes and e-cigarettes
About 53% of respondents agreed that “e-cigarettes are just as harmful or more harmful than traditional cigarettes.” Nicotine and the flavors in e-cigarettes are largely perceived as “very” or “extremely” harmful to the health of a person. However, the authors note that “no published study on nicotine substitutes has shown harmful effects on the health of a person, let alone determined it a risk factor for cancer. The nicotine doses in e-cigarettes are similar to traditional nicotine substitutes, and no cytotoxic effect of nicotine in its inhaled form has been found.” There seems to be confusion between dependence and risk of cancer.
Alcohol consumption
Eight of 10 respondents believe that “some people can drink a lot of alcohol all their life without ever getting cancer,” which goes against the scientific literature. The authors of the report state that the negative effects of alcohol on health seem poorly understood. Although alcohol is the second biggest cause of cancer, only a third of survey respondents cited it without having been prompted as one of the main causes of cancer. And 23.5% even think that “in terms of decreasing your risk of cancer, it’s better to drink a little wine than to drink no wine at all.”
This article was translated from the Medscape French edition. A version of this article appeared on Medscape.com.
FRANCE – Conducted every 5 years since 2005, the Cancer Survey documents the knowledge, perceptions, and way of life of the French people in relation to cancer. The researchers analyzed responses to telephone interviews of a representative sample of almost 5,000 individuals aged 15-85 years.
This study shows how thinking has changed over time and how difficult it is to alter preconceived notions.
Is cancer hereditary?
The report shows that 67.7% of respondents believe that cancer is a hereditary disease. Respondents were asked to explain their answer. “Data show that medical practices for cancer treatment substantiate this belief [that cancer is hereditary],” wrote the authors of the report.
“Indeed, health care professionals almost systematically ask questions about family history of breast cancer and, when a family member has been diagnosed with cancer, medical monitoring of other family members is often sought out, thus reinforcing the belief that cancer is hereditary,” they said.
Furthermore, there seems to be confusion regarding the role of genes in the development of cancer. A person can inherit cancer-predisposing genes, not cancer itself. The authors highlighted their concern that this confusion may “lead people to think that prevention measures are unnecessary because cancer is inherited.”
Misconceptions about smoking
About 41% of smokers think that the length of time one has been smoking is the biggest determining factor for developing cancer; 58.1% think the number of cigarettes smoked per day has a bigger impact.
Experts at InCA and SPF put the debate to rest, stating that prolonged exposure to carcinogenic substances is far more toxic. As for the danger threshold concerning the number of cigarettes smoked per day, respondents believed this to be 9.2 cigarettes per day, on average. They believed that the danger threshold for the number of years as an active smoker is 13.4, on average.
“The [survey] respondents clearly understand that smoking carries a risk, but many smokers think that light smoking or smoking for a short period of time doesn’t carry any risks.” Yet it is understood that even occasional tobacco consumption increases mortality.
This was not the only misconception regarding smoking and its relationship with cancer. About 34% of survey respondents agreed with the following statement: “Smoking doesn’t cause cancer unless you’re a heavy smoker and have smoked for a long time.” Furthermore, 43.3% agreed with the statement, “Pollution is more likely to cause cancer than smoking,” 54.6% think that “exercising cleans your lungs of tobacco,” and 61.6% think that “a smoker can prevent developing cancer caused by smoking if they know to quit on time.”
Overweight and obesity
Although diet and excess weight represent the third and fourth biggest avoidable cancer risk factors, after smoking and alcohol, only 30% of survey respondents knew of this link.
“Among the causes of cancer known and cited by respondents without prompting, excessive weight and obesity were mentioned only 100 times out of 12,558 responses,” highlighted the authors of the report. The explanation put forward by the authors is that discourse about diet has been more focused on diet as a protective health factor, especially in preventing cardiovascular diseases. “The link between cancer and diet is less prominent in the public space,” they noted.
Breastfeeding and cancer
About 63% of survey respondents, which for the first time included both women and men, believe that breastfeeding does not affect mothers’ risk of breast cancer, but this is a misconception. And almost 1 in 3 respondents said that breastfeeding provides health benefits for the mother.
Artificial UV rays
Exposure to UV rays, whether of natural or artificial origin, is a major risk factor for skin cancer. However, 1 in 5 people (20.9%) think that a session in a tanning bed is less harmful than sun exposure.
Daily stress
Regarding psychological factors linked to cancer, the authors noted that risk factors not supported by scientific evidence were, ironically, cited more often by respondents than proven risk factors. There is a real knowledge gap between scientific data and the beliefs of the French people. For example, “working at night” is largely not seen as a risk factor, but data show that it presents a clear risk. However, “not being able to express one’s feelings,” “having been weakened by traumatic experiences,” and “being exposed to the stress of modern life” are seen as risk factors of cancer, without any scientific evidence.
Cigarettes and e-cigarettes
About 53% of respondents agreed that “e-cigarettes are just as harmful or more harmful than traditional cigarettes.” Nicotine and the flavors in e-cigarettes are largely perceived as “very” or “extremely” harmful to the health of a person. However, the authors note that “no published study on nicotine substitutes has shown harmful effects on the health of a person, let alone determined it a risk factor for cancer. The nicotine doses in e-cigarettes are similar to traditional nicotine substitutes, and no cytotoxic effect of nicotine in its inhaled form has been found.” There seems to be confusion between dependence and risk of cancer.
Alcohol consumption
Eight of 10 respondents believe that “some people can drink a lot of alcohol all their life without ever getting cancer,” which goes against the scientific literature. The authors of the report state that the negative effects of alcohol on health seem poorly understood. Although alcohol is the second biggest cause of cancer, only a third of survey respondents cited it without having been prompted as one of the main causes of cancer. And 23.5% even think that “in terms of decreasing your risk of cancer, it’s better to drink a little wine than to drink no wine at all.”
This article was translated from the Medscape French edition. A version of this article appeared on Medscape.com.
Advanced imaging technology could help predict lung cancer progression after surgery
, according to new data.
The technology, known as highly multiplexed imaging mass cytometry (IMC), can provide cellular-level detail of the tumor immune microenvironment, which may allow clinicians to identify patients who need additional treatment, as well as those who don’t.
“It is well known that the frequency of certain cell populations within the tumor microenvironment correlates with clinical outcomes. These observations help us understand the biology underlying cancer progression,” senior author Logan Walsh, PhD, assistant professor of human genetics and the Rosalind Goodman Chair in Lung Cancer Research at McGill University’s Rosalind and Morris Goodman Cancer Institute, Montreal, said in an interview.
“We wanted to test whether using completely unbiased AI could find and use the spatial topography of the tumor microenvironment from IMC data to predict clinical outcomes,” he said. “It turns out the answer is yes! AI can predict clinical outcomes when combined with IMC with extremely high accuracy from a single 1-mm2 tumor core.”
The study was published on in Nature.
The immune landscape
Lung cancer is the leading cause of cancer-related death in Canada, surpassing breast, colon, and prostate cancer deaths combined, the study authors write.
Lung adenocarcinoma, a non–small cell lung cancer, is the most common subtype and is characterized by distinct cellular and molecular features. The tumor immune microenvironment influences disease progression and therapy response, the authors write. Understanding the spatial landscape of the microenvironment could provide insight into disease progression, therapeutic vulnerabilities, and biomarkers of response to existing treatments.
In a collaborative study, Dr. Walsh and colleagues from McGill University and Université Laval profiled the cellular composition and spatial organization of the tumor immune microenvironment in tumors from 416 patients with lung adenocarcinoma across five histologic patterns. They used IMC to assess at samples from the universities’ biobanks that patients had provided for research purposes.
The research team detected more than 1.6 million cells, which allowed spatial analysis of immune lineages and activation states with distinct clinical correlates, including survival. They used a supervised lineage assignment approach to classify 14 distinct immune cell populations, along with tumor cells and endothelial cells.
High-grade solid tumors had the greatest immune infiltrate (44.6%), compared with micropapillary (37%), acinar (39.7%), papillary (32.8%), and lepidic architectures (32.7%). Macrophages were the most frequent cell population in the tumor immune microenvironment, representing 12.3% of total cells and 34.1% of immune cells.
The prevalence of CD163+ macrophages was strongly correlated with FOXP3+ immunoregulatory T cells in the solid pattern. This relationship was less pronounced in low-grade lepidic and papillary architectures. This finding could suggest an interplay between macrophage and T-cell populations in the tumor immune microenvironment across lung adenocarcinoma patterns.
Using a deep neural network model, the researchers also analyzed the relationship between immune populations and clinical or pathologic variables by examining the frequency of individual cell types as a percentage of total cells in each image. Each image was cross-referenced with clinical data from patients, including sex, age, body mass index, smoking status, stage, progression, survival, and histologic subtype.
Overall, the researchers found that various clinical outcomes, including cancer progression, could be predicted with high accuracy using a single 1-mm2 tumor core. For instance, they could predict progression in stage IA and IB resected lung cancer with 95.9% accuracy.
Additional applications
“We were not surprised that AI was able to predict clinical outcomes, but we were surprised that it was able to do so with such high accuracy and precision,” said Dr. Walsh. “We were also surprised to learn that our predictions were equally accurate using only six-plex data, compared with 35-plex. This hinted to us that we could potentially scale down the number of markers to a practical number that would be amenable to technologies available in routine pathology labs.”
Dr. Walsh and colleagues are now validating the predictive tool using a lower-plex technology. In addition, they are investigating the immune landscapes of primary and metastatic brain tumors.
“This study is important, as it helps us to understand and appreciate the biological and mechanistic factors that may influence treatment outcomes. Our standard clinical predictors for predicting risk of recurrence and probability of response to therapy are not optimal,” Yee Ung, MD, an associate professor of radiation oncology at Sunnybrook Health Sciences Centre, Toronto, said in an interview.
Dr. Ung, who wasn’t involved with this study, has researched noninvasive hypoxia imaging and targeting in lung cancer. Ideally, he said, future studies should incorporate the use of noninvasive imaging predictive factors, in addition to the tumor immune microenvironment and clinical factors, to predict outcomes and provide personalized treatment.
“As we begin to investigate and understand more about cancer biology down to the cellular and molecular level, we need to strategically use AI methodologies in the processing and analysis of data,” he said.
The study was supported by the McGill Interdisciplinary Initiative in Infection and Immunity, the Brain Tumour Funders’ Collaborative, the Canadian Institutes of Health Research, and the Canadian Foundation for Innovation. Dr. Walsh and Dr. Ung have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
, according to new data.
The technology, known as highly multiplexed imaging mass cytometry (IMC), can provide cellular-level detail of the tumor immune microenvironment, which may allow clinicians to identify patients who need additional treatment, as well as those who don’t.
“It is well known that the frequency of certain cell populations within the tumor microenvironment correlates with clinical outcomes. These observations help us understand the biology underlying cancer progression,” senior author Logan Walsh, PhD, assistant professor of human genetics and the Rosalind Goodman Chair in Lung Cancer Research at McGill University’s Rosalind and Morris Goodman Cancer Institute, Montreal, said in an interview.
“We wanted to test whether using completely unbiased AI could find and use the spatial topography of the tumor microenvironment from IMC data to predict clinical outcomes,” he said. “It turns out the answer is yes! AI can predict clinical outcomes when combined with IMC with extremely high accuracy from a single 1-mm2 tumor core.”
The study was published on in Nature.
The immune landscape
Lung cancer is the leading cause of cancer-related death in Canada, surpassing breast, colon, and prostate cancer deaths combined, the study authors write.
Lung adenocarcinoma, a non–small cell lung cancer, is the most common subtype and is characterized by distinct cellular and molecular features. The tumor immune microenvironment influences disease progression and therapy response, the authors write. Understanding the spatial landscape of the microenvironment could provide insight into disease progression, therapeutic vulnerabilities, and biomarkers of response to existing treatments.
In a collaborative study, Dr. Walsh and colleagues from McGill University and Université Laval profiled the cellular composition and spatial organization of the tumor immune microenvironment in tumors from 416 patients with lung adenocarcinoma across five histologic patterns. They used IMC to assess at samples from the universities’ biobanks that patients had provided for research purposes.
The research team detected more than 1.6 million cells, which allowed spatial analysis of immune lineages and activation states with distinct clinical correlates, including survival. They used a supervised lineage assignment approach to classify 14 distinct immune cell populations, along with tumor cells and endothelial cells.
High-grade solid tumors had the greatest immune infiltrate (44.6%), compared with micropapillary (37%), acinar (39.7%), papillary (32.8%), and lepidic architectures (32.7%). Macrophages were the most frequent cell population in the tumor immune microenvironment, representing 12.3% of total cells and 34.1% of immune cells.
The prevalence of CD163+ macrophages was strongly correlated with FOXP3+ immunoregulatory T cells in the solid pattern. This relationship was less pronounced in low-grade lepidic and papillary architectures. This finding could suggest an interplay between macrophage and T-cell populations in the tumor immune microenvironment across lung adenocarcinoma patterns.
Using a deep neural network model, the researchers also analyzed the relationship between immune populations and clinical or pathologic variables by examining the frequency of individual cell types as a percentage of total cells in each image. Each image was cross-referenced with clinical data from patients, including sex, age, body mass index, smoking status, stage, progression, survival, and histologic subtype.
Overall, the researchers found that various clinical outcomes, including cancer progression, could be predicted with high accuracy using a single 1-mm2 tumor core. For instance, they could predict progression in stage IA and IB resected lung cancer with 95.9% accuracy.
Additional applications
“We were not surprised that AI was able to predict clinical outcomes, but we were surprised that it was able to do so with such high accuracy and precision,” said Dr. Walsh. “We were also surprised to learn that our predictions were equally accurate using only six-plex data, compared with 35-plex. This hinted to us that we could potentially scale down the number of markers to a practical number that would be amenable to technologies available in routine pathology labs.”
Dr. Walsh and colleagues are now validating the predictive tool using a lower-plex technology. In addition, they are investigating the immune landscapes of primary and metastatic brain tumors.
“This study is important, as it helps us to understand and appreciate the biological and mechanistic factors that may influence treatment outcomes. Our standard clinical predictors for predicting risk of recurrence and probability of response to therapy are not optimal,” Yee Ung, MD, an associate professor of radiation oncology at Sunnybrook Health Sciences Centre, Toronto, said in an interview.
Dr. Ung, who wasn’t involved with this study, has researched noninvasive hypoxia imaging and targeting in lung cancer. Ideally, he said, future studies should incorporate the use of noninvasive imaging predictive factors, in addition to the tumor immune microenvironment and clinical factors, to predict outcomes and provide personalized treatment.
“As we begin to investigate and understand more about cancer biology down to the cellular and molecular level, we need to strategically use AI methodologies in the processing and analysis of data,” he said.
The study was supported by the McGill Interdisciplinary Initiative in Infection and Immunity, the Brain Tumour Funders’ Collaborative, the Canadian Institutes of Health Research, and the Canadian Foundation for Innovation. Dr. Walsh and Dr. Ung have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
, according to new data.
The technology, known as highly multiplexed imaging mass cytometry (IMC), can provide cellular-level detail of the tumor immune microenvironment, which may allow clinicians to identify patients who need additional treatment, as well as those who don’t.
“It is well known that the frequency of certain cell populations within the tumor microenvironment correlates with clinical outcomes. These observations help us understand the biology underlying cancer progression,” senior author Logan Walsh, PhD, assistant professor of human genetics and the Rosalind Goodman Chair in Lung Cancer Research at McGill University’s Rosalind and Morris Goodman Cancer Institute, Montreal, said in an interview.
“We wanted to test whether using completely unbiased AI could find and use the spatial topography of the tumor microenvironment from IMC data to predict clinical outcomes,” he said. “It turns out the answer is yes! AI can predict clinical outcomes when combined with IMC with extremely high accuracy from a single 1-mm2 tumor core.”
The study was published on in Nature.
The immune landscape
Lung cancer is the leading cause of cancer-related death in Canada, surpassing breast, colon, and prostate cancer deaths combined, the study authors write.
Lung adenocarcinoma, a non–small cell lung cancer, is the most common subtype and is characterized by distinct cellular and molecular features. The tumor immune microenvironment influences disease progression and therapy response, the authors write. Understanding the spatial landscape of the microenvironment could provide insight into disease progression, therapeutic vulnerabilities, and biomarkers of response to existing treatments.
In a collaborative study, Dr. Walsh and colleagues from McGill University and Université Laval profiled the cellular composition and spatial organization of the tumor immune microenvironment in tumors from 416 patients with lung adenocarcinoma across five histologic patterns. They used IMC to assess at samples from the universities’ biobanks that patients had provided for research purposes.
The research team detected more than 1.6 million cells, which allowed spatial analysis of immune lineages and activation states with distinct clinical correlates, including survival. They used a supervised lineage assignment approach to classify 14 distinct immune cell populations, along with tumor cells and endothelial cells.
High-grade solid tumors had the greatest immune infiltrate (44.6%), compared with micropapillary (37%), acinar (39.7%), papillary (32.8%), and lepidic architectures (32.7%). Macrophages were the most frequent cell population in the tumor immune microenvironment, representing 12.3% of total cells and 34.1% of immune cells.
The prevalence of CD163+ macrophages was strongly correlated with FOXP3+ immunoregulatory T cells in the solid pattern. This relationship was less pronounced in low-grade lepidic and papillary architectures. This finding could suggest an interplay between macrophage and T-cell populations in the tumor immune microenvironment across lung adenocarcinoma patterns.
Using a deep neural network model, the researchers also analyzed the relationship between immune populations and clinical or pathologic variables by examining the frequency of individual cell types as a percentage of total cells in each image. Each image was cross-referenced with clinical data from patients, including sex, age, body mass index, smoking status, stage, progression, survival, and histologic subtype.
Overall, the researchers found that various clinical outcomes, including cancer progression, could be predicted with high accuracy using a single 1-mm2 tumor core. For instance, they could predict progression in stage IA and IB resected lung cancer with 95.9% accuracy.
Additional applications
“We were not surprised that AI was able to predict clinical outcomes, but we were surprised that it was able to do so with such high accuracy and precision,” said Dr. Walsh. “We were also surprised to learn that our predictions were equally accurate using only six-plex data, compared with 35-plex. This hinted to us that we could potentially scale down the number of markers to a practical number that would be amenable to technologies available in routine pathology labs.”
Dr. Walsh and colleagues are now validating the predictive tool using a lower-plex technology. In addition, they are investigating the immune landscapes of primary and metastatic brain tumors.
“This study is important, as it helps us to understand and appreciate the biological and mechanistic factors that may influence treatment outcomes. Our standard clinical predictors for predicting risk of recurrence and probability of response to therapy are not optimal,” Yee Ung, MD, an associate professor of radiation oncology at Sunnybrook Health Sciences Centre, Toronto, said in an interview.
Dr. Ung, who wasn’t involved with this study, has researched noninvasive hypoxia imaging and targeting in lung cancer. Ideally, he said, future studies should incorporate the use of noninvasive imaging predictive factors, in addition to the tumor immune microenvironment and clinical factors, to predict outcomes and provide personalized treatment.
“As we begin to investigate and understand more about cancer biology down to the cellular and molecular level, we need to strategically use AI methodologies in the processing and analysis of data,” he said.
The study was supported by the McGill Interdisciplinary Initiative in Infection and Immunity, the Brain Tumour Funders’ Collaborative, the Canadian Institutes of Health Research, and the Canadian Foundation for Innovation. Dr. Walsh and Dr. Ung have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM NATURE
Postop RT: Meaningful survival improvement in N2 lung cancer
This transcript has been edited for clarity.
I’m Mark Kris from Memorial Sloan Kettering, speaking today about a topic that’s become quite controversial, which is
Data from clinical trials and data from a SEER study showed approximately 7% improvement in overall survival in patients with N2 disease who received PORT. There has been a very clear demonstration of an improved local control rate in every trial that’s ever looked at PORT.
However, there was a randomized trial, the Lung ART trial, where patients were randomized to get PORT or not. PORT was delivered in a way that is not routinely used now. In that trial, the benefit of PORT was found in terms of local control, almost doubling control within the mediastinum.
The difference in overall survival was less than 12%. Again, I’m not surprised to see that because the improvement in overall survival is probably somewhere between 5% and 10%. They also found an excess of deaths, probably due to cardiac causes from the radiation in the radiation arm.
However, the trial used a type of radiation not used at this point – it used conformal, but now we would use 3D. And its ability at the time of the trial to estimate and lower cardiac risk was not what it is today. Owing to the design of the trial, it was not a significant difference and has largely been interpreted as saying that the PORT doesn’t work.
First, let’s please go to the guidelines. I’m going to the ASCO guidelines, which say that patients with mediastinal disease should not routinely get PORT, but they should be routinely referred to a radiation oncologist for consideration of PORT. I don’t think anything that’s been published so far changes that.
I think each case needs to be individualized and requires the specialty care of a radiation oncologist to weigh the pros and cons of PORT. It also depends upon the treatment plan. Can the heart be spared? Are there radiation techniques available that would eliminate or lessen heart exposure, such as using protons? The point is that PORT is still needed.
When we look at the trials of patients receiving adjuvant therapy – and I’m looking particularly at the ADAURA trial where patients received adjuvant osimertinib – the greatest number of failures now is in the chest. We have to look for good ways to cut down on failure in the chest. Unfortunately, failure in the chest means ultimately failure and lack of cure, and we have to do a better job at that. I think PORT can play a role there.
Please, when you have patients with N2 disease, after the completion of systemic therapies, think about the use of PORT and get the advice of a radiation oncologist to meet with the patient, review their clinical situation, and assess whether or not PORT could be useful for that patient.
That is following the NCCN guidelines, which were not changed on the basis of the Lung ART paper. I think we owe it to our patients to make sure that those who could benefit from this additional therapy receive it.
I’ll put it to you that radiation delivered in the most innovative way – taking very careful account of the effects on the heart – can improve local control. There’s no question about that. I think PORT has the ability to improve survival by a small amount – probably less than 12%, which I will agree the Lung ART trial showed – but still an important amount for patients with this condition.
Mark G. Kris, MD, is chief of the thoracic oncology service and the William and Joy Ruane Chair in Thoracic Oncology at Memorial Sloan Kettering Cancer Center in New York City. He reported conflicts of interest with Arial Pharmaceuticals, Pfizer, PUMA, and Roche/Genentech. A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
I’m Mark Kris from Memorial Sloan Kettering, speaking today about a topic that’s become quite controversial, which is
Data from clinical trials and data from a SEER study showed approximately 7% improvement in overall survival in patients with N2 disease who received PORT. There has been a very clear demonstration of an improved local control rate in every trial that’s ever looked at PORT.
However, there was a randomized trial, the Lung ART trial, where patients were randomized to get PORT or not. PORT was delivered in a way that is not routinely used now. In that trial, the benefit of PORT was found in terms of local control, almost doubling control within the mediastinum.
The difference in overall survival was less than 12%. Again, I’m not surprised to see that because the improvement in overall survival is probably somewhere between 5% and 10%. They also found an excess of deaths, probably due to cardiac causes from the radiation in the radiation arm.
However, the trial used a type of radiation not used at this point – it used conformal, but now we would use 3D. And its ability at the time of the trial to estimate and lower cardiac risk was not what it is today. Owing to the design of the trial, it was not a significant difference and has largely been interpreted as saying that the PORT doesn’t work.
First, let’s please go to the guidelines. I’m going to the ASCO guidelines, which say that patients with mediastinal disease should not routinely get PORT, but they should be routinely referred to a radiation oncologist for consideration of PORT. I don’t think anything that’s been published so far changes that.
I think each case needs to be individualized and requires the specialty care of a radiation oncologist to weigh the pros and cons of PORT. It also depends upon the treatment plan. Can the heart be spared? Are there radiation techniques available that would eliminate or lessen heart exposure, such as using protons? The point is that PORT is still needed.
When we look at the trials of patients receiving adjuvant therapy – and I’m looking particularly at the ADAURA trial where patients received adjuvant osimertinib – the greatest number of failures now is in the chest. We have to look for good ways to cut down on failure in the chest. Unfortunately, failure in the chest means ultimately failure and lack of cure, and we have to do a better job at that. I think PORT can play a role there.
Please, when you have patients with N2 disease, after the completion of systemic therapies, think about the use of PORT and get the advice of a radiation oncologist to meet with the patient, review their clinical situation, and assess whether or not PORT could be useful for that patient.
That is following the NCCN guidelines, which were not changed on the basis of the Lung ART paper. I think we owe it to our patients to make sure that those who could benefit from this additional therapy receive it.
I’ll put it to you that radiation delivered in the most innovative way – taking very careful account of the effects on the heart – can improve local control. There’s no question about that. I think PORT has the ability to improve survival by a small amount – probably less than 12%, which I will agree the Lung ART trial showed – but still an important amount for patients with this condition.
Mark G. Kris, MD, is chief of the thoracic oncology service and the William and Joy Ruane Chair in Thoracic Oncology at Memorial Sloan Kettering Cancer Center in New York City. He reported conflicts of interest with Arial Pharmaceuticals, Pfizer, PUMA, and Roche/Genentech. A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
I’m Mark Kris from Memorial Sloan Kettering, speaking today about a topic that’s become quite controversial, which is
Data from clinical trials and data from a SEER study showed approximately 7% improvement in overall survival in patients with N2 disease who received PORT. There has been a very clear demonstration of an improved local control rate in every trial that’s ever looked at PORT.
However, there was a randomized trial, the Lung ART trial, where patients were randomized to get PORT or not. PORT was delivered in a way that is not routinely used now. In that trial, the benefit of PORT was found in terms of local control, almost doubling control within the mediastinum.
The difference in overall survival was less than 12%. Again, I’m not surprised to see that because the improvement in overall survival is probably somewhere between 5% and 10%. They also found an excess of deaths, probably due to cardiac causes from the radiation in the radiation arm.
However, the trial used a type of radiation not used at this point – it used conformal, but now we would use 3D. And its ability at the time of the trial to estimate and lower cardiac risk was not what it is today. Owing to the design of the trial, it was not a significant difference and has largely been interpreted as saying that the PORT doesn’t work.
First, let’s please go to the guidelines. I’m going to the ASCO guidelines, which say that patients with mediastinal disease should not routinely get PORT, but they should be routinely referred to a radiation oncologist for consideration of PORT. I don’t think anything that’s been published so far changes that.
I think each case needs to be individualized and requires the specialty care of a radiation oncologist to weigh the pros and cons of PORT. It also depends upon the treatment plan. Can the heart be spared? Are there radiation techniques available that would eliminate or lessen heart exposure, such as using protons? The point is that PORT is still needed.
When we look at the trials of patients receiving adjuvant therapy – and I’m looking particularly at the ADAURA trial where patients received adjuvant osimertinib – the greatest number of failures now is in the chest. We have to look for good ways to cut down on failure in the chest. Unfortunately, failure in the chest means ultimately failure and lack of cure, and we have to do a better job at that. I think PORT can play a role there.
Please, when you have patients with N2 disease, after the completion of systemic therapies, think about the use of PORT and get the advice of a radiation oncologist to meet with the patient, review their clinical situation, and assess whether or not PORT could be useful for that patient.
That is following the NCCN guidelines, which were not changed on the basis of the Lung ART paper. I think we owe it to our patients to make sure that those who could benefit from this additional therapy receive it.
I’ll put it to you that radiation delivered in the most innovative way – taking very careful account of the effects on the heart – can improve local control. There’s no question about that. I think PORT has the ability to improve survival by a small amount – probably less than 12%, which I will agree the Lung ART trial showed – but still an important amount for patients with this condition.
Mark G. Kris, MD, is chief of the thoracic oncology service and the William and Joy Ruane Chair in Thoracic Oncology at Memorial Sloan Kettering Cancer Center in New York City. He reported conflicts of interest with Arial Pharmaceuticals, Pfizer, PUMA, and Roche/Genentech. A version of this article first appeared on Medscape.com.