User login
DMTs in Aging MS Patients: When and How to Stop
NASHVILLE, TENNESSEE — Individuals with multiple sclerosis are living longer, healthier lives. More than half of patients with MS are 55 years or older, and the incidence of late-onset MS is rising.
This can lead to complex treatment decisions, according to Amy Perrin Ross, APN, MSN, CNRN, MSCN, who is the neuroscience program coordinator at Loyola Medical Center in Maywood, Illinois.
said Ms. Ross, during a presentation at the annual meeting of the Consortium of Multiple Sclerosis Centers. But there is little evidence to support treatment decisions, since there are few older patients enrolled in clinical trials. The average age is around 30-34 years.
MS in Older Patients
Aging is associated with immune system changes. There is a decline in inflammatory activity and an accompanying 17% reduction in the relapse rate with every 5 years of advancing age, and the majority of relapses occur within 30 years of onset. The bad news is that patients have reduced capacity to recover from relapses as they age.
“When I’m talking to patients about pros and cons [of treatment], I do mention that, yes, your relapse rate might be less, but as we age, we have less of an ability to completely recover,” said Ms. Ross.
The efficacy of disease-modifying therapies (DMTs) goes down with advancing age. One meta-analyis of 38 randomized trials and 13 therapies found that benefit with respect to disease progression generally disappeared by the age of 53. “Age is an essential modifier of drug efficacy,” said Ms. Ross.
On the other hand, another meta-analysis found that success in treating relapses was similar across age groups. “So it seems that we can successfully treat our patients’ relapses: There was no significant association between age and reductions in annualized relapse rate,” she said, though she noted that clinical trial populations are likely to be dissimilar to aging patients, many of whom have gone years without experiencing a relapse.
Aging can also lead to differences in potential adverse effects of DMTs. Patients with MS experience faster immunosenescence, in which normal changes to the innate and adaptive immune system are accelerated. This can lead to greater risk of infection, and other adverse events can include post-administration reactions and changes to serum IgG levels.
Other conditions that should be monitored for include progressive multifocal leukoencephalopathy, and malignancies are more prevalent among people with MS than the general population, although it is unclear if this is due to the use of DMTs or other factors, or even just coincidence, said Ms. Ross. “Those are all things to keep in mind as we’re pushing forward with therapy for patients,” she said.
Comorbidities that occur with aging can also affect treatment outcomes, and could tip the balance against use of DMTs in some situations.
What Does the Literature Say?
There has been a range of retrospective studies looking at the results of discontinuation of DMTs with advancing age, and the results have been mixed. Some factors are associated with greater likelihood of disease reactivation, including younger age, female sex, shorter duration without a relapse, MRI activity, and degree of disability.
A study of a French registry including patients aged 50 years and older who went off DMTs found that 100% of patients who discontinued therapy were on older injectable DMTs, and 34.9% of that group restarted therapy over a mean follow-up of 7 years. The risk of relapse or disability progression was similar between the groups, but discontinuers who started with Expanded Disability Status Scale (EDSS) scores lower than 6.0 were more likely to reach an EDSS score of 6.0.
The DISCOMS study compared 259 patients randomized to continue DMTs versus discontinuation of DMTs. “What they found was that noninferiority was not shown. Disease activity, such as relapses and new lesions, [occurred in] 12% of the discontinuers and 5% of the continuers,” said Ms. Ross.
One option to balance risk and benefit is DMT de-escalation, with the aim to match disease therapy with disease activity over time. A 2023 survey of 224 neurologists to identify characteristics in older patients that would prompt de-escalation. The most common reasons were overall safety or comorbidity concerns (62% endorsed), high risk of infection (59%), low disease activity or stable disease (50%), concerns about efficacy (41%), high disability (37%), and patient choice (36%). About 7% reported that they generally do not de-escalate.
The preferred de-escalation therapies included glatiramer acetate (29%), fumarates (27%), teriflunomide (23%), and interferon betas (21%).
Ms. Ross noted that the study was likely conducted around the height of the COVID-19 pandemic. “So I wonder if some of these results might be a little bit different [than if it was conducted at a different time],” she said.
Other Concerns and Options
During the Q&A session, one audience member asked if physicians should consider low-efficacy medications in older patients with the idea that they at least get a little bit of protection.
Patricia Coyle, MD, who also presented during the session, framed her response around whether the patient had relapsing or progressive MS. “If somebody has had relapsing MS and has never transitioned to progressive MS, and they’re 70, maybe they don’t need to be on any DMT. If there’s no longer a focal inflammatory relapsing phase, if we could feel confident on that possibility, then maybe they don’t need to be on a relapsing DMT,” said Dr. Coyle, who is director of the MS Comprehensive Care Center at Stony Brook University Medical Center in Stony Brook, New York.
Alternatively, if a patient has progressive MS, she said she would recommend discontinuing treatment if she believes the patient is being harmed by it, to focus instead on health and wellness.
Another questioner wondered what to do with a 70-year-old patient who has had no infections, has normal IgG, but insists on continuing high-efficacy B-cell therapy. Dr. Coyle responded that she would tell the patient that she believes it isn’t offering any benefit, but if the patient insisted, she would continue: “I’m not living with MS the way they are. If they tell me, ‘I believe it’s helping me and I want to stay on it,’ then so long as I don’t think I’m overtly harming them, I’m going to treat them.”
Ms. Ross agreed, and suggested that ceding to the patient’s will is an important consideration. “I think sometimes what we’re doing, if we’re not causing harm, what we’re doing is bolstering these people’s ability to continue to have hope, and that in my mind is a big part of managing their disease,” she said.
Ms. Ross has financial relationships with Alexion Pharmaceuticals, Amgen/Horizon, ArgenX, Banner, Bristol Myers Squibb, EMD Serono, Roche, Sandoz, TG Therapeutics, UCB, and Viatris. Dr. Coyle has consulted for Accordant, Amgen, Bristol Myers Squibb, EMD Serono, Genentech, GlaxoSmithKline, Horizon Therapeutics, LabCorp, Eli Lilly, Mylan, Novartis, and Sanofi Genzyme. She has received research funding from Celgene, CorEvitas, Genentech/Roche, NINDS, and Sanofi Genzyme.
NASHVILLE, TENNESSEE — Individuals with multiple sclerosis are living longer, healthier lives. More than half of patients with MS are 55 years or older, and the incidence of late-onset MS is rising.
This can lead to complex treatment decisions, according to Amy Perrin Ross, APN, MSN, CNRN, MSCN, who is the neuroscience program coordinator at Loyola Medical Center in Maywood, Illinois.
said Ms. Ross, during a presentation at the annual meeting of the Consortium of Multiple Sclerosis Centers. But there is little evidence to support treatment decisions, since there are few older patients enrolled in clinical trials. The average age is around 30-34 years.
MS in Older Patients
Aging is associated with immune system changes. There is a decline in inflammatory activity and an accompanying 17% reduction in the relapse rate with every 5 years of advancing age, and the majority of relapses occur within 30 years of onset. The bad news is that patients have reduced capacity to recover from relapses as they age.
“When I’m talking to patients about pros and cons [of treatment], I do mention that, yes, your relapse rate might be less, but as we age, we have less of an ability to completely recover,” said Ms. Ross.
The efficacy of disease-modifying therapies (DMTs) goes down with advancing age. One meta-analyis of 38 randomized trials and 13 therapies found that benefit with respect to disease progression generally disappeared by the age of 53. “Age is an essential modifier of drug efficacy,” said Ms. Ross.
On the other hand, another meta-analysis found that success in treating relapses was similar across age groups. “So it seems that we can successfully treat our patients’ relapses: There was no significant association between age and reductions in annualized relapse rate,” she said, though she noted that clinical trial populations are likely to be dissimilar to aging patients, many of whom have gone years without experiencing a relapse.
Aging can also lead to differences in potential adverse effects of DMTs. Patients with MS experience faster immunosenescence, in which normal changes to the innate and adaptive immune system are accelerated. This can lead to greater risk of infection, and other adverse events can include post-administration reactions and changes to serum IgG levels.
Other conditions that should be monitored for include progressive multifocal leukoencephalopathy, and malignancies are more prevalent among people with MS than the general population, although it is unclear if this is due to the use of DMTs or other factors, or even just coincidence, said Ms. Ross. “Those are all things to keep in mind as we’re pushing forward with therapy for patients,” she said.
Comorbidities that occur with aging can also affect treatment outcomes, and could tip the balance against use of DMTs in some situations.
What Does the Literature Say?
There has been a range of retrospective studies looking at the results of discontinuation of DMTs with advancing age, and the results have been mixed. Some factors are associated with greater likelihood of disease reactivation, including younger age, female sex, shorter duration without a relapse, MRI activity, and degree of disability.
A study of a French registry including patients aged 50 years and older who went off DMTs found that 100% of patients who discontinued therapy were on older injectable DMTs, and 34.9% of that group restarted therapy over a mean follow-up of 7 years. The risk of relapse or disability progression was similar between the groups, but discontinuers who started with Expanded Disability Status Scale (EDSS) scores lower than 6.0 were more likely to reach an EDSS score of 6.0.
The DISCOMS study compared 259 patients randomized to continue DMTs versus discontinuation of DMTs. “What they found was that noninferiority was not shown. Disease activity, such as relapses and new lesions, [occurred in] 12% of the discontinuers and 5% of the continuers,” said Ms. Ross.
One option to balance risk and benefit is DMT de-escalation, with the aim to match disease therapy with disease activity over time. A 2023 survey of 224 neurologists to identify characteristics in older patients that would prompt de-escalation. The most common reasons were overall safety or comorbidity concerns (62% endorsed), high risk of infection (59%), low disease activity or stable disease (50%), concerns about efficacy (41%), high disability (37%), and patient choice (36%). About 7% reported that they generally do not de-escalate.
The preferred de-escalation therapies included glatiramer acetate (29%), fumarates (27%), teriflunomide (23%), and interferon betas (21%).
Ms. Ross noted that the study was likely conducted around the height of the COVID-19 pandemic. “So I wonder if some of these results might be a little bit different [than if it was conducted at a different time],” she said.
Other Concerns and Options
During the Q&A session, one audience member asked if physicians should consider low-efficacy medications in older patients with the idea that they at least get a little bit of protection.
Patricia Coyle, MD, who also presented during the session, framed her response around whether the patient had relapsing or progressive MS. “If somebody has had relapsing MS and has never transitioned to progressive MS, and they’re 70, maybe they don’t need to be on any DMT. If there’s no longer a focal inflammatory relapsing phase, if we could feel confident on that possibility, then maybe they don’t need to be on a relapsing DMT,” said Dr. Coyle, who is director of the MS Comprehensive Care Center at Stony Brook University Medical Center in Stony Brook, New York.
Alternatively, if a patient has progressive MS, she said she would recommend discontinuing treatment if she believes the patient is being harmed by it, to focus instead on health and wellness.
Another questioner wondered what to do with a 70-year-old patient who has had no infections, has normal IgG, but insists on continuing high-efficacy B-cell therapy. Dr. Coyle responded that she would tell the patient that she believes it isn’t offering any benefit, but if the patient insisted, she would continue: “I’m not living with MS the way they are. If they tell me, ‘I believe it’s helping me and I want to stay on it,’ then so long as I don’t think I’m overtly harming them, I’m going to treat them.”
Ms. Ross agreed, and suggested that ceding to the patient’s will is an important consideration. “I think sometimes what we’re doing, if we’re not causing harm, what we’re doing is bolstering these people’s ability to continue to have hope, and that in my mind is a big part of managing their disease,” she said.
Ms. Ross has financial relationships with Alexion Pharmaceuticals, Amgen/Horizon, ArgenX, Banner, Bristol Myers Squibb, EMD Serono, Roche, Sandoz, TG Therapeutics, UCB, and Viatris. Dr. Coyle has consulted for Accordant, Amgen, Bristol Myers Squibb, EMD Serono, Genentech, GlaxoSmithKline, Horizon Therapeutics, LabCorp, Eli Lilly, Mylan, Novartis, and Sanofi Genzyme. She has received research funding from Celgene, CorEvitas, Genentech/Roche, NINDS, and Sanofi Genzyme.
NASHVILLE, TENNESSEE — Individuals with multiple sclerosis are living longer, healthier lives. More than half of patients with MS are 55 years or older, and the incidence of late-onset MS is rising.
This can lead to complex treatment decisions, according to Amy Perrin Ross, APN, MSN, CNRN, MSCN, who is the neuroscience program coordinator at Loyola Medical Center in Maywood, Illinois.
said Ms. Ross, during a presentation at the annual meeting of the Consortium of Multiple Sclerosis Centers. But there is little evidence to support treatment decisions, since there are few older patients enrolled in clinical trials. The average age is around 30-34 years.
MS in Older Patients
Aging is associated with immune system changes. There is a decline in inflammatory activity and an accompanying 17% reduction in the relapse rate with every 5 years of advancing age, and the majority of relapses occur within 30 years of onset. The bad news is that patients have reduced capacity to recover from relapses as they age.
“When I’m talking to patients about pros and cons [of treatment], I do mention that, yes, your relapse rate might be less, but as we age, we have less of an ability to completely recover,” said Ms. Ross.
The efficacy of disease-modifying therapies (DMTs) goes down with advancing age. One meta-analyis of 38 randomized trials and 13 therapies found that benefit with respect to disease progression generally disappeared by the age of 53. “Age is an essential modifier of drug efficacy,” said Ms. Ross.
On the other hand, another meta-analysis found that success in treating relapses was similar across age groups. “So it seems that we can successfully treat our patients’ relapses: There was no significant association between age and reductions in annualized relapse rate,” she said, though she noted that clinical trial populations are likely to be dissimilar to aging patients, many of whom have gone years without experiencing a relapse.
Aging can also lead to differences in potential adverse effects of DMTs. Patients with MS experience faster immunosenescence, in which normal changes to the innate and adaptive immune system are accelerated. This can lead to greater risk of infection, and other adverse events can include post-administration reactions and changes to serum IgG levels.
Other conditions that should be monitored for include progressive multifocal leukoencephalopathy, and malignancies are more prevalent among people with MS than the general population, although it is unclear if this is due to the use of DMTs or other factors, or even just coincidence, said Ms. Ross. “Those are all things to keep in mind as we’re pushing forward with therapy for patients,” she said.
Comorbidities that occur with aging can also affect treatment outcomes, and could tip the balance against use of DMTs in some situations.
What Does the Literature Say?
There has been a range of retrospective studies looking at the results of discontinuation of DMTs with advancing age, and the results have been mixed. Some factors are associated with greater likelihood of disease reactivation, including younger age, female sex, shorter duration without a relapse, MRI activity, and degree of disability.
A study of a French registry including patients aged 50 years and older who went off DMTs found that 100% of patients who discontinued therapy were on older injectable DMTs, and 34.9% of that group restarted therapy over a mean follow-up of 7 years. The risk of relapse or disability progression was similar between the groups, but discontinuers who started with Expanded Disability Status Scale (EDSS) scores lower than 6.0 were more likely to reach an EDSS score of 6.0.
The DISCOMS study compared 259 patients randomized to continue DMTs versus discontinuation of DMTs. “What they found was that noninferiority was not shown. Disease activity, such as relapses and new lesions, [occurred in] 12% of the discontinuers and 5% of the continuers,” said Ms. Ross.
One option to balance risk and benefit is DMT de-escalation, with the aim to match disease therapy with disease activity over time. A 2023 survey of 224 neurologists to identify characteristics in older patients that would prompt de-escalation. The most common reasons were overall safety or comorbidity concerns (62% endorsed), high risk of infection (59%), low disease activity or stable disease (50%), concerns about efficacy (41%), high disability (37%), and patient choice (36%). About 7% reported that they generally do not de-escalate.
The preferred de-escalation therapies included glatiramer acetate (29%), fumarates (27%), teriflunomide (23%), and interferon betas (21%).
Ms. Ross noted that the study was likely conducted around the height of the COVID-19 pandemic. “So I wonder if some of these results might be a little bit different [than if it was conducted at a different time],” she said.
Other Concerns and Options
During the Q&A session, one audience member asked if physicians should consider low-efficacy medications in older patients with the idea that they at least get a little bit of protection.
Patricia Coyle, MD, who also presented during the session, framed her response around whether the patient had relapsing or progressive MS. “If somebody has had relapsing MS and has never transitioned to progressive MS, and they’re 70, maybe they don’t need to be on any DMT. If there’s no longer a focal inflammatory relapsing phase, if we could feel confident on that possibility, then maybe they don’t need to be on a relapsing DMT,” said Dr. Coyle, who is director of the MS Comprehensive Care Center at Stony Brook University Medical Center in Stony Brook, New York.
Alternatively, if a patient has progressive MS, she said she would recommend discontinuing treatment if she believes the patient is being harmed by it, to focus instead on health and wellness.
Another questioner wondered what to do with a 70-year-old patient who has had no infections, has normal IgG, but insists on continuing high-efficacy B-cell therapy. Dr. Coyle responded that she would tell the patient that she believes it isn’t offering any benefit, but if the patient insisted, she would continue: “I’m not living with MS the way they are. If they tell me, ‘I believe it’s helping me and I want to stay on it,’ then so long as I don’t think I’m overtly harming them, I’m going to treat them.”
Ms. Ross agreed, and suggested that ceding to the patient’s will is an important consideration. “I think sometimes what we’re doing, if we’re not causing harm, what we’re doing is bolstering these people’s ability to continue to have hope, and that in my mind is a big part of managing their disease,” she said.
Ms. Ross has financial relationships with Alexion Pharmaceuticals, Amgen/Horizon, ArgenX, Banner, Bristol Myers Squibb, EMD Serono, Roche, Sandoz, TG Therapeutics, UCB, and Viatris. Dr. Coyle has consulted for Accordant, Amgen, Bristol Myers Squibb, EMD Serono, Genentech, GlaxoSmithKline, Horizon Therapeutics, LabCorp, Eli Lilly, Mylan, Novartis, and Sanofi Genzyme. She has received research funding from Celgene, CorEvitas, Genentech/Roche, NINDS, and Sanofi Genzyme.
FROM CMSC 2024
Migraine Disability Nearly Doubled in US Between 2005 and 2018
, a new systematic review showed.
“The disability trend could reflect changes in reporting, study methodology, social, and societal changes, or changes in exacerbating or remediating factors that make migraine more disabling,” wrote lead investigator Fred Cohen, MD, of Center for Headache and Facial Pain, Department of Neurology, Icahn School of Medicine, Mount Sinai, New York City, and colleagues.
The study was published online in Headache.
Researchers conducted a systematic review of population-based US epidemiologic studies focusing on the prevalence and/or burden of migraine, all published before February 2022. Studies on migraine, episodic migraine, and/or chronic migraine were included.
The primary measure of disease burden was the Migraine Disability Assessment Scale (MIDAS), which measures days lost to migraine over a 3-month period in three domains and defines groups with moderate or severe disability (grades III and IV, respectively), using cut-scores.
Of 1609 studies initially reviewed, the researchers included 26 publications from 11 US population-based studies.
For the past 30 years, the prevalence of migraine in the population has remained largely stable, ranging from 12% to 15% in the overall population, from 17% to 19% in women, and from 6% to 7% in men.
In adults overall, chronic migraine prevalence is 0.91% (1.3% in women and 0.5% in men), while in adolescents, the prevalence is 0.8%.
Although prevalence remained roughly the same during the 30 years, the proportion of people with migraine and moderate to severe MIDAS disability (grades III-IV) has trended upward across studies during part of the study period, increasing from 22% in 2005 to 42% in 2018.
Throughout the years studied, a consistently higher proportion of women versus men were assigned MIDAS grades III-IV.
Although researchers said the exact reason for the increase is unknown, possible explanations include changes in study methodology from mailed questionnaires to web surveys or the decline in participation rate in web surveys. It is also possible that people with migraine may be more willing to report disability than they used to be, authors wrote.
Increased MIDAS scores may be attributable to some environmental risk factor that exacerbates migraine without modifying its prevalence, such as worsening air quality, an increase in natural disasters, or increased opioid use for migraine, they added.
The reason for increased moderate to severe disability in women may be attributable to the fact that migraine is “most common in mid-life, a period characterized by familial and work responsibilities, which may engender a higher risk of burden for working women,” authors wrote. The link between migraine attacks and menstrual cycles may also explain observed gender differences in disability.
In general, the most frequently reported burdens associated with migraine included missed work and school and family and social functioning.
It is “surprising that improvements in treatment have not been associated with reductions in disability,” researchers noted.
No financial support was provided for this study. Dr. Cohen serves as an assistant editor for Headache. He has received honoraria from Springer Nature and MedLink Neurology. Other authors’ disclosures are listed on the original paper.
A version of this article appeared on Medscape.com.
, a new systematic review showed.
“The disability trend could reflect changes in reporting, study methodology, social, and societal changes, or changes in exacerbating or remediating factors that make migraine more disabling,” wrote lead investigator Fred Cohen, MD, of Center for Headache and Facial Pain, Department of Neurology, Icahn School of Medicine, Mount Sinai, New York City, and colleagues.
The study was published online in Headache.
Researchers conducted a systematic review of population-based US epidemiologic studies focusing on the prevalence and/or burden of migraine, all published before February 2022. Studies on migraine, episodic migraine, and/or chronic migraine were included.
The primary measure of disease burden was the Migraine Disability Assessment Scale (MIDAS), which measures days lost to migraine over a 3-month period in three domains and defines groups with moderate or severe disability (grades III and IV, respectively), using cut-scores.
Of 1609 studies initially reviewed, the researchers included 26 publications from 11 US population-based studies.
For the past 30 years, the prevalence of migraine in the population has remained largely stable, ranging from 12% to 15% in the overall population, from 17% to 19% in women, and from 6% to 7% in men.
In adults overall, chronic migraine prevalence is 0.91% (1.3% in women and 0.5% in men), while in adolescents, the prevalence is 0.8%.
Although prevalence remained roughly the same during the 30 years, the proportion of people with migraine and moderate to severe MIDAS disability (grades III-IV) has trended upward across studies during part of the study period, increasing from 22% in 2005 to 42% in 2018.
Throughout the years studied, a consistently higher proportion of women versus men were assigned MIDAS grades III-IV.
Although researchers said the exact reason for the increase is unknown, possible explanations include changes in study methodology from mailed questionnaires to web surveys or the decline in participation rate in web surveys. It is also possible that people with migraine may be more willing to report disability than they used to be, authors wrote.
Increased MIDAS scores may be attributable to some environmental risk factor that exacerbates migraine without modifying its prevalence, such as worsening air quality, an increase in natural disasters, or increased opioid use for migraine, they added.
The reason for increased moderate to severe disability in women may be attributable to the fact that migraine is “most common in mid-life, a period characterized by familial and work responsibilities, which may engender a higher risk of burden for working women,” authors wrote. The link between migraine attacks and menstrual cycles may also explain observed gender differences in disability.
In general, the most frequently reported burdens associated with migraine included missed work and school and family and social functioning.
It is “surprising that improvements in treatment have not been associated with reductions in disability,” researchers noted.
No financial support was provided for this study. Dr. Cohen serves as an assistant editor for Headache. He has received honoraria from Springer Nature and MedLink Neurology. Other authors’ disclosures are listed on the original paper.
A version of this article appeared on Medscape.com.
, a new systematic review showed.
“The disability trend could reflect changes in reporting, study methodology, social, and societal changes, or changes in exacerbating or remediating factors that make migraine more disabling,” wrote lead investigator Fred Cohen, MD, of Center for Headache and Facial Pain, Department of Neurology, Icahn School of Medicine, Mount Sinai, New York City, and colleagues.
The study was published online in Headache.
Researchers conducted a systematic review of population-based US epidemiologic studies focusing on the prevalence and/or burden of migraine, all published before February 2022. Studies on migraine, episodic migraine, and/or chronic migraine were included.
The primary measure of disease burden was the Migraine Disability Assessment Scale (MIDAS), which measures days lost to migraine over a 3-month period in three domains and defines groups with moderate or severe disability (grades III and IV, respectively), using cut-scores.
Of 1609 studies initially reviewed, the researchers included 26 publications from 11 US population-based studies.
For the past 30 years, the prevalence of migraine in the population has remained largely stable, ranging from 12% to 15% in the overall population, from 17% to 19% in women, and from 6% to 7% in men.
In adults overall, chronic migraine prevalence is 0.91% (1.3% in women and 0.5% in men), while in adolescents, the prevalence is 0.8%.
Although prevalence remained roughly the same during the 30 years, the proportion of people with migraine and moderate to severe MIDAS disability (grades III-IV) has trended upward across studies during part of the study period, increasing from 22% in 2005 to 42% in 2018.
Throughout the years studied, a consistently higher proportion of women versus men were assigned MIDAS grades III-IV.
Although researchers said the exact reason for the increase is unknown, possible explanations include changes in study methodology from mailed questionnaires to web surveys or the decline in participation rate in web surveys. It is also possible that people with migraine may be more willing to report disability than they used to be, authors wrote.
Increased MIDAS scores may be attributable to some environmental risk factor that exacerbates migraine without modifying its prevalence, such as worsening air quality, an increase in natural disasters, or increased opioid use for migraine, they added.
The reason for increased moderate to severe disability in women may be attributable to the fact that migraine is “most common in mid-life, a period characterized by familial and work responsibilities, which may engender a higher risk of burden for working women,” authors wrote. The link between migraine attacks and menstrual cycles may also explain observed gender differences in disability.
In general, the most frequently reported burdens associated with migraine included missed work and school and family and social functioning.
It is “surprising that improvements in treatment have not been associated with reductions in disability,” researchers noted.
No financial support was provided for this study. Dr. Cohen serves as an assistant editor for Headache. He has received honoraria from Springer Nature and MedLink Neurology. Other authors’ disclosures are listed on the original paper.
A version of this article appeared on Medscape.com.
FROM HEADACHE
Guidelines on Rapid Blood Pressure Reduction in Acute Ischemic Stroke Challenged
BASEL, SWITZERLAND — New findings challenge the practice of rapidly lowering blood pressure (BP) in acute ischemic stroke to allow for speedy thrombolysis.
The observational cluster study showed that , even though this meant fewer patient received thrombolysis.
“We found insufficient evidence to recommend active blood pressure lowering in patients with ischemic stroke who have blood pressure levels exceeding the guidelines but are otherwise eligible for thrombolytic therapy,” said senior study author Nyika D. Kruyt, MD, PhD, Leiden University Medical Center, the Netherlands.
“Our results suggest that if the blood pressure is too high for thrombolysis, then it is best to wait and only treat with thrombolysis if the blood pressure drops spontaneously,” Dr. Kruyt said.
The findings were presented at the European Stroke Organisation Conference (ESOC) annual meeting and published online in The Lancet Neurology.
Guidelines Without Evidence?
Owing to concerns about high BP increasing the risk for intracerebral hemorrhage after thrombolysis, the original trials evaluating thrombolysis in stroke set an arbitrary threshold of 185/110 mm Hg, which has been incorporated into stroke guidelines. These trials cautioned against lowering BP rapidly, which is not included in guidelines.
Most stroke centers therefore tend to rapidly lower BP in patients who have values greater than 185/110 mm Hg and who are otherwise eligible for thrombolysis, investigators noted. Because thrombolysis is more effective the earlier it is administered, there is some urgency to reduce the BP quickly when patients first arrive at the hospital.
“But there has never been any evidence for the lowering of blood pressure with IV [intravenous] antihypertensives before thrombolysis, and some centers have never adopted this approach because of concerns that a rapid decline in blood pressure may reduce perfusion of the brain at a time when there is already ischemia present,” Dr. Kruyt noted.
However, if BP is lowered quickly, there is a greater chance that patients will not be able to receive thrombolysis because the 4.5-hour time limit could be exceeded.
For the prospective, observational TRUTH study, researchers compared outcomes in 853 patients treated at 27 stroke centers in the Netherlands with an active BP-lowering strategy vs 199 patients treated at 10 hospitals with no such strategy.
Baseline characteristics of participants in the two groups were similar.
Results showed a strong trend toward worse outcomes in participants whose BP was lowered, with an adjusted odds ratio (aOR) for a shift toward a worse 90-day functional outcome on the modified Rankin Scale of 1.27 (95% CI, 0.96-1.68).
This was despite the fact that many more patients whose BP was reduced received thrombolysis (94% vs 52% of those with no BP lowering) and had shorter times to treatment, with average door-to-needle times of 35 minutes (vs 47 minutes among those with no BP lowering).
Symptomatic intracranial hemorrhage occurred in 5% of the active BP-lowering group versus 3% of those who did not have their BP lowered (aOR, 1.28; 95% CI, 0.62-2.62).
Reconsider Guidelines?
These results are consistent with those from the INTERACT4 trial, which was also presented at the ESOC 2024 meeting. That trial showed a harmful effect of lowering BP in the ambulance in patients with acute ischemic stroke, but a beneficial effect in patients with hemorrhagic stroke.
“I think the guidelines need to be reconsidered after these studies and we should refrain from active blood pressure–lowering in patients with acute ischemic stroke,” Dr. Kruyt said.
But he acknowledged that not rapidly lowering BP will mean fewer patients will be able to receive thrombolysis within the 4.5-hour treatment window.
Dr. Kruyt estimated that the combination of being eligible for thrombolysis, with the only exclusion criterion being BP greater than 185/110 mm Hg, applies to about 10%-15% of patients.
“If we have a watch-and-wait policy, then about half of those patients will still get treated with thrombolysis within in the 4.5-hour limit but later than if blood pressure was reduced with IV antihypertensives,” he added.
Dr. Kruyt noted that there has never been a randomized trial on the practice of BP lowering in order to be able to administer thrombolysis.
“The 185/110 mm Hg blood pressure level is an arbitrary threshold that was chosen for the original thrombolysis stroke trials,” he said. “I believe we need trials to investigate whether we can give thrombolysis safely to patients with higher blood pressure levels than this, without needing to rapidly reduce the pressure.”
Caution Advised
Discussing the TRUTH study at the ESOC meeting, Guillaume Turc, MD, professor of neurology at Sainte-Anne Hospital, Paris, said he thought the findings were “very thought provoking.”
Simona Sacco, MD, professor of neurology at the University of L’Aquila, Italy, said the result was surprising, but she advised caution in acting on this finding.
“I don’t think this study can change practice or guidelines as it is not a randomized trial. Yes, it can generate a hypothesis, but we need more research before changing clinical practice,” she said.
In an accompanying editorial, Verónica Olavarría, MD, Clínica Alemana Universidad del Desarrollo, Santiago, Chile, also suggested the trial should be interpreted cautiously because there was “insufficient evidence for a definitive conclusion.”
But Dr. Kruyt noted that even though the TRUTH study was not a randomized trial, the results are in line with those of recent randomized trials such as INTERACT4.
He added that the ENCHANTED trial also showed no benefit of intensive BP management immediately after thrombolysis in mild to moderate stroke and even suggested harm in severe stroke. And other trials (OPTIMAL-BP and ENCHANTED2/MT) have shown worse outcomes with BP lowering in patients with acute ischemic stroke undergoing thrombectomy.
“All these studies are showing similar signals throughout the whole timeline in acute ischemic stroke. The results are very much in line with each other. I think this strengthens our findings,” Dr. Kruyt said.
“With this data, I think the guidelines should be revised, and until randomized data become available showing that reducing blood pressure in acute ischemic stroke patients in order for them to receive early thrombolysis is beneficial, then we should refrain from doing so,” he added.
INTERACT4 investigator Craig Anderson, MD, George Institute for Global Health, Sydney, New South Wales, Australia, agreed.
“The TRUTH study gives the same message as INTERACT4. They are completely in line with each other, both suggesting harm with blood pressure lowering in acute ischemic stroke. These two together are going to rattle the cage around blood pressure control in acute ischemic stroke patients,” Dr. Anderson said.
The TRUTH study was funded by a grant from Fonds NutsOhra. Dr. Kruyt reported no relevant financial disclosures. Dr. Olavarría reported receiving a grant from Boehringer Ingelheim for the RECCA registry and honoraria from Novo Nordisk.
A version of this article appeared on Medscape.com.
BASEL, SWITZERLAND — New findings challenge the practice of rapidly lowering blood pressure (BP) in acute ischemic stroke to allow for speedy thrombolysis.
The observational cluster study showed that , even though this meant fewer patient received thrombolysis.
“We found insufficient evidence to recommend active blood pressure lowering in patients with ischemic stroke who have blood pressure levels exceeding the guidelines but are otherwise eligible for thrombolytic therapy,” said senior study author Nyika D. Kruyt, MD, PhD, Leiden University Medical Center, the Netherlands.
“Our results suggest that if the blood pressure is too high for thrombolysis, then it is best to wait and only treat with thrombolysis if the blood pressure drops spontaneously,” Dr. Kruyt said.
The findings were presented at the European Stroke Organisation Conference (ESOC) annual meeting and published online in The Lancet Neurology.
Guidelines Without Evidence?
Owing to concerns about high BP increasing the risk for intracerebral hemorrhage after thrombolysis, the original trials evaluating thrombolysis in stroke set an arbitrary threshold of 185/110 mm Hg, which has been incorporated into stroke guidelines. These trials cautioned against lowering BP rapidly, which is not included in guidelines.
Most stroke centers therefore tend to rapidly lower BP in patients who have values greater than 185/110 mm Hg and who are otherwise eligible for thrombolysis, investigators noted. Because thrombolysis is more effective the earlier it is administered, there is some urgency to reduce the BP quickly when patients first arrive at the hospital.
“But there has never been any evidence for the lowering of blood pressure with IV [intravenous] antihypertensives before thrombolysis, and some centers have never adopted this approach because of concerns that a rapid decline in blood pressure may reduce perfusion of the brain at a time when there is already ischemia present,” Dr. Kruyt noted.
However, if BP is lowered quickly, there is a greater chance that patients will not be able to receive thrombolysis because the 4.5-hour time limit could be exceeded.
For the prospective, observational TRUTH study, researchers compared outcomes in 853 patients treated at 27 stroke centers in the Netherlands with an active BP-lowering strategy vs 199 patients treated at 10 hospitals with no such strategy.
Baseline characteristics of participants in the two groups were similar.
Results showed a strong trend toward worse outcomes in participants whose BP was lowered, with an adjusted odds ratio (aOR) for a shift toward a worse 90-day functional outcome on the modified Rankin Scale of 1.27 (95% CI, 0.96-1.68).
This was despite the fact that many more patients whose BP was reduced received thrombolysis (94% vs 52% of those with no BP lowering) and had shorter times to treatment, with average door-to-needle times of 35 minutes (vs 47 minutes among those with no BP lowering).
Symptomatic intracranial hemorrhage occurred in 5% of the active BP-lowering group versus 3% of those who did not have their BP lowered (aOR, 1.28; 95% CI, 0.62-2.62).
Reconsider Guidelines?
These results are consistent with those from the INTERACT4 trial, which was also presented at the ESOC 2024 meeting. That trial showed a harmful effect of lowering BP in the ambulance in patients with acute ischemic stroke, but a beneficial effect in patients with hemorrhagic stroke.
“I think the guidelines need to be reconsidered after these studies and we should refrain from active blood pressure–lowering in patients with acute ischemic stroke,” Dr. Kruyt said.
But he acknowledged that not rapidly lowering BP will mean fewer patients will be able to receive thrombolysis within the 4.5-hour treatment window.
Dr. Kruyt estimated that the combination of being eligible for thrombolysis, with the only exclusion criterion being BP greater than 185/110 mm Hg, applies to about 10%-15% of patients.
“If we have a watch-and-wait policy, then about half of those patients will still get treated with thrombolysis within in the 4.5-hour limit but later than if blood pressure was reduced with IV antihypertensives,” he added.
Dr. Kruyt noted that there has never been a randomized trial on the practice of BP lowering in order to be able to administer thrombolysis.
“The 185/110 mm Hg blood pressure level is an arbitrary threshold that was chosen for the original thrombolysis stroke trials,” he said. “I believe we need trials to investigate whether we can give thrombolysis safely to patients with higher blood pressure levels than this, without needing to rapidly reduce the pressure.”
Caution Advised
Discussing the TRUTH study at the ESOC meeting, Guillaume Turc, MD, professor of neurology at Sainte-Anne Hospital, Paris, said he thought the findings were “very thought provoking.”
Simona Sacco, MD, professor of neurology at the University of L’Aquila, Italy, said the result was surprising, but she advised caution in acting on this finding.
“I don’t think this study can change practice or guidelines as it is not a randomized trial. Yes, it can generate a hypothesis, but we need more research before changing clinical practice,” she said.
In an accompanying editorial, Verónica Olavarría, MD, Clínica Alemana Universidad del Desarrollo, Santiago, Chile, also suggested the trial should be interpreted cautiously because there was “insufficient evidence for a definitive conclusion.”
But Dr. Kruyt noted that even though the TRUTH study was not a randomized trial, the results are in line with those of recent randomized trials such as INTERACT4.
He added that the ENCHANTED trial also showed no benefit of intensive BP management immediately after thrombolysis in mild to moderate stroke and even suggested harm in severe stroke. And other trials (OPTIMAL-BP and ENCHANTED2/MT) have shown worse outcomes with BP lowering in patients with acute ischemic stroke undergoing thrombectomy.
“All these studies are showing similar signals throughout the whole timeline in acute ischemic stroke. The results are very much in line with each other. I think this strengthens our findings,” Dr. Kruyt said.
“With this data, I think the guidelines should be revised, and until randomized data become available showing that reducing blood pressure in acute ischemic stroke patients in order for them to receive early thrombolysis is beneficial, then we should refrain from doing so,” he added.
INTERACT4 investigator Craig Anderson, MD, George Institute for Global Health, Sydney, New South Wales, Australia, agreed.
“The TRUTH study gives the same message as INTERACT4. They are completely in line with each other, both suggesting harm with blood pressure lowering in acute ischemic stroke. These two together are going to rattle the cage around blood pressure control in acute ischemic stroke patients,” Dr. Anderson said.
The TRUTH study was funded by a grant from Fonds NutsOhra. Dr. Kruyt reported no relevant financial disclosures. Dr. Olavarría reported receiving a grant from Boehringer Ingelheim for the RECCA registry and honoraria from Novo Nordisk.
A version of this article appeared on Medscape.com.
BASEL, SWITZERLAND — New findings challenge the practice of rapidly lowering blood pressure (BP) in acute ischemic stroke to allow for speedy thrombolysis.
The observational cluster study showed that , even though this meant fewer patient received thrombolysis.
“We found insufficient evidence to recommend active blood pressure lowering in patients with ischemic stroke who have blood pressure levels exceeding the guidelines but are otherwise eligible for thrombolytic therapy,” said senior study author Nyika D. Kruyt, MD, PhD, Leiden University Medical Center, the Netherlands.
“Our results suggest that if the blood pressure is too high for thrombolysis, then it is best to wait and only treat with thrombolysis if the blood pressure drops spontaneously,” Dr. Kruyt said.
The findings were presented at the European Stroke Organisation Conference (ESOC) annual meeting and published online in The Lancet Neurology.
Guidelines Without Evidence?
Owing to concerns about high BP increasing the risk for intracerebral hemorrhage after thrombolysis, the original trials evaluating thrombolysis in stroke set an arbitrary threshold of 185/110 mm Hg, which has been incorporated into stroke guidelines. These trials cautioned against lowering BP rapidly, which is not included in guidelines.
Most stroke centers therefore tend to rapidly lower BP in patients who have values greater than 185/110 mm Hg and who are otherwise eligible for thrombolysis, investigators noted. Because thrombolysis is more effective the earlier it is administered, there is some urgency to reduce the BP quickly when patients first arrive at the hospital.
“But there has never been any evidence for the lowering of blood pressure with IV [intravenous] antihypertensives before thrombolysis, and some centers have never adopted this approach because of concerns that a rapid decline in blood pressure may reduce perfusion of the brain at a time when there is already ischemia present,” Dr. Kruyt noted.
However, if BP is lowered quickly, there is a greater chance that patients will not be able to receive thrombolysis because the 4.5-hour time limit could be exceeded.
For the prospective, observational TRUTH study, researchers compared outcomes in 853 patients treated at 27 stroke centers in the Netherlands with an active BP-lowering strategy vs 199 patients treated at 10 hospitals with no such strategy.
Baseline characteristics of participants in the two groups were similar.
Results showed a strong trend toward worse outcomes in participants whose BP was lowered, with an adjusted odds ratio (aOR) for a shift toward a worse 90-day functional outcome on the modified Rankin Scale of 1.27 (95% CI, 0.96-1.68).
This was despite the fact that many more patients whose BP was reduced received thrombolysis (94% vs 52% of those with no BP lowering) and had shorter times to treatment, with average door-to-needle times of 35 minutes (vs 47 minutes among those with no BP lowering).
Symptomatic intracranial hemorrhage occurred in 5% of the active BP-lowering group versus 3% of those who did not have their BP lowered (aOR, 1.28; 95% CI, 0.62-2.62).
Reconsider Guidelines?
These results are consistent with those from the INTERACT4 trial, which was also presented at the ESOC 2024 meeting. That trial showed a harmful effect of lowering BP in the ambulance in patients with acute ischemic stroke, but a beneficial effect in patients with hemorrhagic stroke.
“I think the guidelines need to be reconsidered after these studies and we should refrain from active blood pressure–lowering in patients with acute ischemic stroke,” Dr. Kruyt said.
But he acknowledged that not rapidly lowering BP will mean fewer patients will be able to receive thrombolysis within the 4.5-hour treatment window.
Dr. Kruyt estimated that the combination of being eligible for thrombolysis, with the only exclusion criterion being BP greater than 185/110 mm Hg, applies to about 10%-15% of patients.
“If we have a watch-and-wait policy, then about half of those patients will still get treated with thrombolysis within in the 4.5-hour limit but later than if blood pressure was reduced with IV antihypertensives,” he added.
Dr. Kruyt noted that there has never been a randomized trial on the practice of BP lowering in order to be able to administer thrombolysis.
“The 185/110 mm Hg blood pressure level is an arbitrary threshold that was chosen for the original thrombolysis stroke trials,” he said. “I believe we need trials to investigate whether we can give thrombolysis safely to patients with higher blood pressure levels than this, without needing to rapidly reduce the pressure.”
Caution Advised
Discussing the TRUTH study at the ESOC meeting, Guillaume Turc, MD, professor of neurology at Sainte-Anne Hospital, Paris, said he thought the findings were “very thought provoking.”
Simona Sacco, MD, professor of neurology at the University of L’Aquila, Italy, said the result was surprising, but she advised caution in acting on this finding.
“I don’t think this study can change practice or guidelines as it is not a randomized trial. Yes, it can generate a hypothesis, but we need more research before changing clinical practice,” she said.
In an accompanying editorial, Verónica Olavarría, MD, Clínica Alemana Universidad del Desarrollo, Santiago, Chile, also suggested the trial should be interpreted cautiously because there was “insufficient evidence for a definitive conclusion.”
But Dr. Kruyt noted that even though the TRUTH study was not a randomized trial, the results are in line with those of recent randomized trials such as INTERACT4.
He added that the ENCHANTED trial also showed no benefit of intensive BP management immediately after thrombolysis in mild to moderate stroke and even suggested harm in severe stroke. And other trials (OPTIMAL-BP and ENCHANTED2/MT) have shown worse outcomes with BP lowering in patients with acute ischemic stroke undergoing thrombectomy.
“All these studies are showing similar signals throughout the whole timeline in acute ischemic stroke. The results are very much in line with each other. I think this strengthens our findings,” Dr. Kruyt said.
“With this data, I think the guidelines should be revised, and until randomized data become available showing that reducing blood pressure in acute ischemic stroke patients in order for them to receive early thrombolysis is beneficial, then we should refrain from doing so,” he added.
INTERACT4 investigator Craig Anderson, MD, George Institute for Global Health, Sydney, New South Wales, Australia, agreed.
“The TRUTH study gives the same message as INTERACT4. They are completely in line with each other, both suggesting harm with blood pressure lowering in acute ischemic stroke. These two together are going to rattle the cage around blood pressure control in acute ischemic stroke patients,” Dr. Anderson said.
The TRUTH study was funded by a grant from Fonds NutsOhra. Dr. Kruyt reported no relevant financial disclosures. Dr. Olavarría reported receiving a grant from Boehringer Ingelheim for the RECCA registry and honoraria from Novo Nordisk.
A version of this article appeared on Medscape.com.
FROM ESOC 2024
IV Thrombolysis Offers No Benefit for Mild Stroke
BASEL, SWITZERLAND — , a new trial has concluded.
Results from the randomized controlled trial TEMPO-2 showed no benefit from treatment with tenecteplase following ischemic stroke. In addition, investigators found a small increased risk for symptomatic intracranial hemorrhage (ICH) and more deaths in the tenecteplase group compared with the control group.
The research suggests that although it makes sense to open up vessels in patients with minor stroke, they didn’t do better with thrombolysis.
“This is not the result we were hoping for, but I think the question of whether to treat these minor stroke patients who are not disabled has now been answered,” said lead investigator Shelagh B. Coutts, MD, University of Calgary, Alberta, Canada.
“After these results, I think we should scan these patients, admit them, give them dual antiplatelet therapy and IV fluids, and watch them like a hawk. If they deteriorate, we can intervene at that point.”
The findings were presented at the European Stroke Organization Conference (ESOC) 2024 annual meeting and published online simultaneously in The Lancet.
Very Little Data
Up to half of patients with ischemic stroke initially present with minimal symptoms, which are not disabling, investigators noted. Despite having low scores on the National Institutes of Health Stroke Scale (NIHSS) that typically range from 0 to 5, a third of these patients are dead or disabled at 90-day follow-up if thrombolysis is withheld.
Patients with minor deficits and evidence of an intracranial occlusion are a subpopulation at a high risk for early neurological deterioration, which most often occurs within the first 24 hours after presentation.
However, many physicians have concerns about giving thrombolysis to these patients because of the potential harm from bleeding in the absence of major deficits, and most trials of thrombolysis have excluded patients with minor stroke. That leaves very little high-quality data to guide practice for these patients.
Two previous studies have compared alteplase with antiplatelet agents in minor stroke, but no trial has specifically looked at the subset of patients with minor stroke who have intracranial occlusion. The TEMPO-2 trial was conducted to evaluate the use of tenecteplase in this patient population.
The multicenter, parallel group, open-label study was conducted at 48 hospitals in Australia, Austria, Brazil, Canada, Finland, Ireland, New Zealand, Singapore, Spain, and the United Kingdom.
The trial included patients with minor acute ischemic stroke (NIHSS score of 0-5) and intracranial occlusion or focal perfusion abnormality who were within 12 hours from stroke onset.
Patients received IV tenecteplase (0.25 mg/kg) or non-thrombolytic standard of care (control). Most patients in the control group were treated with dual antiplatelet therapy with aspirin and clopidogrel (57%) or aspirin monotherapy (23%).
The trial was stopped early for futility after 886 patients had been enrolled. The median NIHSS score was 2.
The primary outcome — a return to baseline functioning on the modified Rankin Scale score at 90 days — occurred in 75% of the control group and in 72% of the tenecteplase group (risk ratio [RR], 0.96; P = .29).
Although there were significantly more patients with early recanalization and an NIHSS score of 0 at day 5 or discharge after tenecteplase treatment, this did not translate into improved functional outcomes at 90 days.
More patients died in the tenecteplase group compared with the control group (5% vs 1%; adjusted hazard ratio, 3.8; P = .0085).
There were eight (2%) symptomatic ICHs in the tenecteplase group versus two (< 1%) in the control group (RR, 4.2; P = .059).
The ICH rate was not different in patients treated after 4.5 hours versus before 4.5 hours. The subgroup of patients treated at 4.5-12.0 hours showed weaker evidence of better outcomes with thrombolysis than those treated before 4.5 hours, suggesting that the 12-hour window for TEMPO-2 did not explain the absence of benefit seen with tenecteplase.
Patients in the control group did better than expected, which may have been the result of chance, patient selection, or greater use of dual antiplatelet therapy, researchers noted.
Despite higher recanalization rates in the tenecteplase group (48% vs 22%), there was no change in the rate of stroke progression between groups, with an 8% rate of progression seen overall in the study.
Noting that previous studies have shown that patients with minor stroke and intracranial occlusion are at a risk for both progression and disability, the authors suggested that good supportive care may have improved outcomes in both groups.
More Trials Needed
Commenting on the study at the ESOC meeting, Urs Fischer, MD, Basel University Hospital, Switzerland, said “What should we do for patients with mild stroke with vessel occlusion has been a huge unanswered question. The TEMPO-2 study did not show a benefit with thrombolysis, and there was a tendency toward an increased risk of ICH. This is an important finding.”
In an accompanying editorial, Simona Sacco, MD, University of L’Aquila, Italy, and Guillaume Turc, MD, Université Paris Cité, France, noted that different minor ischemic stroke populations pose different therapeutic challenges.
Observational data suggest a benefit of endovascular treatment for minor stroke with large vessel occlusion, and dedicated randomized controlled trials in this group are ongoing, they added.
Early dual antiplatelet treatment is now the recommended treatment of minor stroke and should therefore be the active comparator for non-cardioembolic strokes in future trials.
While TEMPO-2 did not prove that tenecteplase is better than the standard of care for the acute treatment of minor stroke, Dr. Sacco and Dr. Turc said the study confirms that tenecteplase is associated with a high rate of recanalization.
“Fast recanalization with intravenous thrombolysis, endovascular treatment, proper patient selection, and combination with dual antiplatelet treatment or early initiation of anticoagulants may translate into tangible clinical benefits for patients with minor ischemic stroke, which should be tested in future studies,” they wrote.
This trial was funded by grants from the Canadian Institutes of Health Research, Heart and Stroke Foundation of Canada, and the British Heart Foundation. Boehringer Ingelheim provided tenecteplase for the study. Dr. Coutts reported no conflicts of interest. Dr. Sacco reported receiving grants for research from Novartis and Uriach; consulting fees from Novartis, Allergan-AbbVie, Teva, Lilly, Lundbeck, Pfizer, Novo Nordisk, Abbott, and AstraZeneca; payment for lectures from Novartis, Allergan-AbbVie, Teva, Lilly, Lundbeck, Pfizer, Novo Nordisk, Abbott, and AstraZeneca; and support for attending conferences from Lilly, Novartis, Teva, Lundbeck, and Pfizer. She is president elect of the European Stroke Organization and editor-in-chief of Cephalalgia. Dr. Turc reported payment for lectures from Guerbet France, is a member of the scientific advisory board of AI-Stroke, and is the Secretary General of the European Stroke Organisation.
A version of this article appeared on Medscape.com.
BASEL, SWITZERLAND — , a new trial has concluded.
Results from the randomized controlled trial TEMPO-2 showed no benefit from treatment with tenecteplase following ischemic stroke. In addition, investigators found a small increased risk for symptomatic intracranial hemorrhage (ICH) and more deaths in the tenecteplase group compared with the control group.
The research suggests that although it makes sense to open up vessels in patients with minor stroke, they didn’t do better with thrombolysis.
“This is not the result we were hoping for, but I think the question of whether to treat these minor stroke patients who are not disabled has now been answered,” said lead investigator Shelagh B. Coutts, MD, University of Calgary, Alberta, Canada.
“After these results, I think we should scan these patients, admit them, give them dual antiplatelet therapy and IV fluids, and watch them like a hawk. If they deteriorate, we can intervene at that point.”
The findings were presented at the European Stroke Organization Conference (ESOC) 2024 annual meeting and published online simultaneously in The Lancet.
Very Little Data
Up to half of patients with ischemic stroke initially present with minimal symptoms, which are not disabling, investigators noted. Despite having low scores on the National Institutes of Health Stroke Scale (NIHSS) that typically range from 0 to 5, a third of these patients are dead or disabled at 90-day follow-up if thrombolysis is withheld.
Patients with minor deficits and evidence of an intracranial occlusion are a subpopulation at a high risk for early neurological deterioration, which most often occurs within the first 24 hours after presentation.
However, many physicians have concerns about giving thrombolysis to these patients because of the potential harm from bleeding in the absence of major deficits, and most trials of thrombolysis have excluded patients with minor stroke. That leaves very little high-quality data to guide practice for these patients.
Two previous studies have compared alteplase with antiplatelet agents in minor stroke, but no trial has specifically looked at the subset of patients with minor stroke who have intracranial occlusion. The TEMPO-2 trial was conducted to evaluate the use of tenecteplase in this patient population.
The multicenter, parallel group, open-label study was conducted at 48 hospitals in Australia, Austria, Brazil, Canada, Finland, Ireland, New Zealand, Singapore, Spain, and the United Kingdom.
The trial included patients with minor acute ischemic stroke (NIHSS score of 0-5) and intracranial occlusion or focal perfusion abnormality who were within 12 hours from stroke onset.
Patients received IV tenecteplase (0.25 mg/kg) or non-thrombolytic standard of care (control). Most patients in the control group were treated with dual antiplatelet therapy with aspirin and clopidogrel (57%) or aspirin monotherapy (23%).
The trial was stopped early for futility after 886 patients had been enrolled. The median NIHSS score was 2.
The primary outcome — a return to baseline functioning on the modified Rankin Scale score at 90 days — occurred in 75% of the control group and in 72% of the tenecteplase group (risk ratio [RR], 0.96; P = .29).
Although there were significantly more patients with early recanalization and an NIHSS score of 0 at day 5 or discharge after tenecteplase treatment, this did not translate into improved functional outcomes at 90 days.
More patients died in the tenecteplase group compared with the control group (5% vs 1%; adjusted hazard ratio, 3.8; P = .0085).
There were eight (2%) symptomatic ICHs in the tenecteplase group versus two (< 1%) in the control group (RR, 4.2; P = .059).
The ICH rate was not different in patients treated after 4.5 hours versus before 4.5 hours. The subgroup of patients treated at 4.5-12.0 hours showed weaker evidence of better outcomes with thrombolysis than those treated before 4.5 hours, suggesting that the 12-hour window for TEMPO-2 did not explain the absence of benefit seen with tenecteplase.
Patients in the control group did better than expected, which may have been the result of chance, patient selection, or greater use of dual antiplatelet therapy, researchers noted.
Despite higher recanalization rates in the tenecteplase group (48% vs 22%), there was no change in the rate of stroke progression between groups, with an 8% rate of progression seen overall in the study.
Noting that previous studies have shown that patients with minor stroke and intracranial occlusion are at a risk for both progression and disability, the authors suggested that good supportive care may have improved outcomes in both groups.
More Trials Needed
Commenting on the study at the ESOC meeting, Urs Fischer, MD, Basel University Hospital, Switzerland, said “What should we do for patients with mild stroke with vessel occlusion has been a huge unanswered question. The TEMPO-2 study did not show a benefit with thrombolysis, and there was a tendency toward an increased risk of ICH. This is an important finding.”
In an accompanying editorial, Simona Sacco, MD, University of L’Aquila, Italy, and Guillaume Turc, MD, Université Paris Cité, France, noted that different minor ischemic stroke populations pose different therapeutic challenges.
Observational data suggest a benefit of endovascular treatment for minor stroke with large vessel occlusion, and dedicated randomized controlled trials in this group are ongoing, they added.
Early dual antiplatelet treatment is now the recommended treatment of minor stroke and should therefore be the active comparator for non-cardioembolic strokes in future trials.
While TEMPO-2 did not prove that tenecteplase is better than the standard of care for the acute treatment of minor stroke, Dr. Sacco and Dr. Turc said the study confirms that tenecteplase is associated with a high rate of recanalization.
“Fast recanalization with intravenous thrombolysis, endovascular treatment, proper patient selection, and combination with dual antiplatelet treatment or early initiation of anticoagulants may translate into tangible clinical benefits for patients with minor ischemic stroke, which should be tested in future studies,” they wrote.
This trial was funded by grants from the Canadian Institutes of Health Research, Heart and Stroke Foundation of Canada, and the British Heart Foundation. Boehringer Ingelheim provided tenecteplase for the study. Dr. Coutts reported no conflicts of interest. Dr. Sacco reported receiving grants for research from Novartis and Uriach; consulting fees from Novartis, Allergan-AbbVie, Teva, Lilly, Lundbeck, Pfizer, Novo Nordisk, Abbott, and AstraZeneca; payment for lectures from Novartis, Allergan-AbbVie, Teva, Lilly, Lundbeck, Pfizer, Novo Nordisk, Abbott, and AstraZeneca; and support for attending conferences from Lilly, Novartis, Teva, Lundbeck, and Pfizer. She is president elect of the European Stroke Organization and editor-in-chief of Cephalalgia. Dr. Turc reported payment for lectures from Guerbet France, is a member of the scientific advisory board of AI-Stroke, and is the Secretary General of the European Stroke Organisation.
A version of this article appeared on Medscape.com.
BASEL, SWITZERLAND — , a new trial has concluded.
Results from the randomized controlled trial TEMPO-2 showed no benefit from treatment with tenecteplase following ischemic stroke. In addition, investigators found a small increased risk for symptomatic intracranial hemorrhage (ICH) and more deaths in the tenecteplase group compared with the control group.
The research suggests that although it makes sense to open up vessels in patients with minor stroke, they didn’t do better with thrombolysis.
“This is not the result we were hoping for, but I think the question of whether to treat these minor stroke patients who are not disabled has now been answered,” said lead investigator Shelagh B. Coutts, MD, University of Calgary, Alberta, Canada.
“After these results, I think we should scan these patients, admit them, give them dual antiplatelet therapy and IV fluids, and watch them like a hawk. If they deteriorate, we can intervene at that point.”
The findings were presented at the European Stroke Organization Conference (ESOC) 2024 annual meeting and published online simultaneously in The Lancet.
Very Little Data
Up to half of patients with ischemic stroke initially present with minimal symptoms, which are not disabling, investigators noted. Despite having low scores on the National Institutes of Health Stroke Scale (NIHSS) that typically range from 0 to 5, a third of these patients are dead or disabled at 90-day follow-up if thrombolysis is withheld.
Patients with minor deficits and evidence of an intracranial occlusion are a subpopulation at a high risk for early neurological deterioration, which most often occurs within the first 24 hours after presentation.
However, many physicians have concerns about giving thrombolysis to these patients because of the potential harm from bleeding in the absence of major deficits, and most trials of thrombolysis have excluded patients with minor stroke. That leaves very little high-quality data to guide practice for these patients.
Two previous studies have compared alteplase with antiplatelet agents in minor stroke, but no trial has specifically looked at the subset of patients with minor stroke who have intracranial occlusion. The TEMPO-2 trial was conducted to evaluate the use of tenecteplase in this patient population.
The multicenter, parallel group, open-label study was conducted at 48 hospitals in Australia, Austria, Brazil, Canada, Finland, Ireland, New Zealand, Singapore, Spain, and the United Kingdom.
The trial included patients with minor acute ischemic stroke (NIHSS score of 0-5) and intracranial occlusion or focal perfusion abnormality who were within 12 hours from stroke onset.
Patients received IV tenecteplase (0.25 mg/kg) or non-thrombolytic standard of care (control). Most patients in the control group were treated with dual antiplatelet therapy with aspirin and clopidogrel (57%) or aspirin monotherapy (23%).
The trial was stopped early for futility after 886 patients had been enrolled. The median NIHSS score was 2.
The primary outcome — a return to baseline functioning on the modified Rankin Scale score at 90 days — occurred in 75% of the control group and in 72% of the tenecteplase group (risk ratio [RR], 0.96; P = .29).
Although there were significantly more patients with early recanalization and an NIHSS score of 0 at day 5 or discharge after tenecteplase treatment, this did not translate into improved functional outcomes at 90 days.
More patients died in the tenecteplase group compared with the control group (5% vs 1%; adjusted hazard ratio, 3.8; P = .0085).
There were eight (2%) symptomatic ICHs in the tenecteplase group versus two (< 1%) in the control group (RR, 4.2; P = .059).
The ICH rate was not different in patients treated after 4.5 hours versus before 4.5 hours. The subgroup of patients treated at 4.5-12.0 hours showed weaker evidence of better outcomes with thrombolysis than those treated before 4.5 hours, suggesting that the 12-hour window for TEMPO-2 did not explain the absence of benefit seen with tenecteplase.
Patients in the control group did better than expected, which may have been the result of chance, patient selection, or greater use of dual antiplatelet therapy, researchers noted.
Despite higher recanalization rates in the tenecteplase group (48% vs 22%), there was no change in the rate of stroke progression between groups, with an 8% rate of progression seen overall in the study.
Noting that previous studies have shown that patients with minor stroke and intracranial occlusion are at a risk for both progression and disability, the authors suggested that good supportive care may have improved outcomes in both groups.
More Trials Needed
Commenting on the study at the ESOC meeting, Urs Fischer, MD, Basel University Hospital, Switzerland, said “What should we do for patients with mild stroke with vessel occlusion has been a huge unanswered question. The TEMPO-2 study did not show a benefit with thrombolysis, and there was a tendency toward an increased risk of ICH. This is an important finding.”
In an accompanying editorial, Simona Sacco, MD, University of L’Aquila, Italy, and Guillaume Turc, MD, Université Paris Cité, France, noted that different minor ischemic stroke populations pose different therapeutic challenges.
Observational data suggest a benefit of endovascular treatment for minor stroke with large vessel occlusion, and dedicated randomized controlled trials in this group are ongoing, they added.
Early dual antiplatelet treatment is now the recommended treatment of minor stroke and should therefore be the active comparator for non-cardioembolic strokes in future trials.
While TEMPO-2 did not prove that tenecteplase is better than the standard of care for the acute treatment of minor stroke, Dr. Sacco and Dr. Turc said the study confirms that tenecteplase is associated with a high rate of recanalization.
“Fast recanalization with intravenous thrombolysis, endovascular treatment, proper patient selection, and combination with dual antiplatelet treatment or early initiation of anticoagulants may translate into tangible clinical benefits for patients with minor ischemic stroke, which should be tested in future studies,” they wrote.
This trial was funded by grants from the Canadian Institutes of Health Research, Heart and Stroke Foundation of Canada, and the British Heart Foundation. Boehringer Ingelheim provided tenecteplase for the study. Dr. Coutts reported no conflicts of interest. Dr. Sacco reported receiving grants for research from Novartis and Uriach; consulting fees from Novartis, Allergan-AbbVie, Teva, Lilly, Lundbeck, Pfizer, Novo Nordisk, Abbott, and AstraZeneca; payment for lectures from Novartis, Allergan-AbbVie, Teva, Lilly, Lundbeck, Pfizer, Novo Nordisk, Abbott, and AstraZeneca; and support for attending conferences from Lilly, Novartis, Teva, Lundbeck, and Pfizer. She is president elect of the European Stroke Organization and editor-in-chief of Cephalalgia. Dr. Turc reported payment for lectures from Guerbet France, is a member of the scientific advisory board of AI-Stroke, and is the Secretary General of the European Stroke Organisation.
A version of this article appeared on Medscape.com.
FROM ESOC 2024
PCPs May Have a New Tool to Help Identify Autism in Young Children
Incorporating eye-tracking biomarkers into pediatric autism assessments may make identifying the condition easier, according to new findings published in JAMA Network Open.
Researchers created an artificial intelligence–based tool to help primary care clinicians and pediatricians spot potential cases of the neurological condition, according to Brandon Keehn, PhD, associate professor in the Department of Speech, Language, and Hearing Sciences at Purdue University in West Lafayette, Indiana, and an author of the study.
Most primary care clinicians do not receive specialized training in identifying autism, and around a third diagnose the condition with uncertainty, according to Dr. Keehn. The tool helps clinicians by incorporating their diagnosis and self-reported level of certainty with eye-tracking biomarkers. A clinical psychologist also assessed children, either verifying or confuting the earlier results.
The tool produced the same diagnosis as that from a psychologist in 90% of cases. When children were assessed using eye biomarkers alone, the diagnosis was aligned with that of a psychologist 77% of the time.
“This is the first step in demonstrating both that eye-tracking biomarkers are sensitive to autism and whether or not these biomarkers provide extra clinical information for primary care physicians to more accurately diagnose autism,” Dr. Keehn told this news organization.
The study took place between 2019 and 2022 and included 146 children between 14 and 48 months old who were treated at seven primary care practices in Indiana. Dr. Keehn and colleagues asked primary care clinicians to rate their level of certainty in their diagnosis.
During the biomarker test, toddlers watched cartoons while researchers tracked their eye movements. Six biomarkers included in the test were based on previous research linking eye movements to autism, according to Dr. Keehn.
These included whether toddlers looked more at images of people or geometric patterns and the speed and size of pupil dilation when exposed to bright light.
Most toddlers produced a positive result for autism in only one biomarker test. Dr. Keehn said this confirms that children should be tested for a variety of biomarkers because each patient’s condition manifests differently.
Dr. Keehn said his team is still a few steps away from determining how the model would work in a real clinical setting and that they are planning more research with a larger study population.
Alice Kuo, MD, a pediatrician specializing in autism at the University of California, Los Angeles (UCLA), said primary care clinicians should feel comfortable making an autism diagnosis.
“Any tool that helps them to do that can be useful, since wait times for a specialist can take years,” Dr. Kuo, also the director of the Autism Intervention Research Network on Physical Health at UCLA, said.
However, Dr. Kuo said she is concerned about the cases that were falsely identified as positive or negative.
“To be told your kid is autistic when he’s not, or to be told your kid is not when he clinically is, has huge ramifications,” she said.
The study was funded by the National Institute of Mental Health, the Riley Children’s Foundation, and the Indiana Clinical and Translational Sciences Institute. Dr. Keehn reported payments for workshops on the use of the Autism Diagnostic Observation Schedule.
A version of this article appeared on Medscape.com .
Incorporating eye-tracking biomarkers into pediatric autism assessments may make identifying the condition easier, according to new findings published in JAMA Network Open.
Researchers created an artificial intelligence–based tool to help primary care clinicians and pediatricians spot potential cases of the neurological condition, according to Brandon Keehn, PhD, associate professor in the Department of Speech, Language, and Hearing Sciences at Purdue University in West Lafayette, Indiana, and an author of the study.
Most primary care clinicians do not receive specialized training in identifying autism, and around a third diagnose the condition with uncertainty, according to Dr. Keehn. The tool helps clinicians by incorporating their diagnosis and self-reported level of certainty with eye-tracking biomarkers. A clinical psychologist also assessed children, either verifying or confuting the earlier results.
The tool produced the same diagnosis as that from a psychologist in 90% of cases. When children were assessed using eye biomarkers alone, the diagnosis was aligned with that of a psychologist 77% of the time.
“This is the first step in demonstrating both that eye-tracking biomarkers are sensitive to autism and whether or not these biomarkers provide extra clinical information for primary care physicians to more accurately diagnose autism,” Dr. Keehn told this news organization.
The study took place between 2019 and 2022 and included 146 children between 14 and 48 months old who were treated at seven primary care practices in Indiana. Dr. Keehn and colleagues asked primary care clinicians to rate their level of certainty in their diagnosis.
During the biomarker test, toddlers watched cartoons while researchers tracked their eye movements. Six biomarkers included in the test were based on previous research linking eye movements to autism, according to Dr. Keehn.
These included whether toddlers looked more at images of people or geometric patterns and the speed and size of pupil dilation when exposed to bright light.
Most toddlers produced a positive result for autism in only one biomarker test. Dr. Keehn said this confirms that children should be tested for a variety of biomarkers because each patient’s condition manifests differently.
Dr. Keehn said his team is still a few steps away from determining how the model would work in a real clinical setting and that they are planning more research with a larger study population.
Alice Kuo, MD, a pediatrician specializing in autism at the University of California, Los Angeles (UCLA), said primary care clinicians should feel comfortable making an autism diagnosis.
“Any tool that helps them to do that can be useful, since wait times for a specialist can take years,” Dr. Kuo, also the director of the Autism Intervention Research Network on Physical Health at UCLA, said.
However, Dr. Kuo said she is concerned about the cases that were falsely identified as positive or negative.
“To be told your kid is autistic when he’s not, or to be told your kid is not when he clinically is, has huge ramifications,” she said.
The study was funded by the National Institute of Mental Health, the Riley Children’s Foundation, and the Indiana Clinical and Translational Sciences Institute. Dr. Keehn reported payments for workshops on the use of the Autism Diagnostic Observation Schedule.
A version of this article appeared on Medscape.com .
Incorporating eye-tracking biomarkers into pediatric autism assessments may make identifying the condition easier, according to new findings published in JAMA Network Open.
Researchers created an artificial intelligence–based tool to help primary care clinicians and pediatricians spot potential cases of the neurological condition, according to Brandon Keehn, PhD, associate professor in the Department of Speech, Language, and Hearing Sciences at Purdue University in West Lafayette, Indiana, and an author of the study.
Most primary care clinicians do not receive specialized training in identifying autism, and around a third diagnose the condition with uncertainty, according to Dr. Keehn. The tool helps clinicians by incorporating their diagnosis and self-reported level of certainty with eye-tracking biomarkers. A clinical psychologist also assessed children, either verifying or confuting the earlier results.
The tool produced the same diagnosis as that from a psychologist in 90% of cases. When children were assessed using eye biomarkers alone, the diagnosis was aligned with that of a psychologist 77% of the time.
“This is the first step in demonstrating both that eye-tracking biomarkers are sensitive to autism and whether or not these biomarkers provide extra clinical information for primary care physicians to more accurately diagnose autism,” Dr. Keehn told this news organization.
The study took place between 2019 and 2022 and included 146 children between 14 and 48 months old who were treated at seven primary care practices in Indiana. Dr. Keehn and colleagues asked primary care clinicians to rate their level of certainty in their diagnosis.
During the biomarker test, toddlers watched cartoons while researchers tracked their eye movements. Six biomarkers included in the test were based on previous research linking eye movements to autism, according to Dr. Keehn.
These included whether toddlers looked more at images of people or geometric patterns and the speed and size of pupil dilation when exposed to bright light.
Most toddlers produced a positive result for autism in only one biomarker test. Dr. Keehn said this confirms that children should be tested for a variety of biomarkers because each patient’s condition manifests differently.
Dr. Keehn said his team is still a few steps away from determining how the model would work in a real clinical setting and that they are planning more research with a larger study population.
Alice Kuo, MD, a pediatrician specializing in autism at the University of California, Los Angeles (UCLA), said primary care clinicians should feel comfortable making an autism diagnosis.
“Any tool that helps them to do that can be useful, since wait times for a specialist can take years,” Dr. Kuo, also the director of the Autism Intervention Research Network on Physical Health at UCLA, said.
However, Dr. Kuo said she is concerned about the cases that were falsely identified as positive or negative.
“To be told your kid is autistic when he’s not, or to be told your kid is not when he clinically is, has huge ramifications,” she said.
The study was funded by the National Institute of Mental Health, the Riley Children’s Foundation, and the Indiana Clinical and Translational Sciences Institute. Dr. Keehn reported payments for workshops on the use of the Autism Diagnostic Observation Schedule.
A version of this article appeared on Medscape.com .
FROM JAMA NETWORK OPEN
Ultraprocessed Foods May Be an Independent Risk Factor for Poor Brain Health
, new research suggests.
Observations from a large cohort of adults followed for more than 10 years suggested that eating more ultraprocessed foods (UPFs) may increase the risk for cognitive decline and stroke, while eating more unprocessed or minimally processed foods may lower the risk.
“The first key takeaway is that the type of food that we eat matters for brain health, but it’s equally important to think about how it’s made and handled when thinking about brain health,” said study investigator W. Taylor Kimberly, MD, PhD, with Massachusetts General Hospital in Boston.
“The second is that it’s not just all a bad news story because while increased consumption of ultra-processed foods is associated with a higher risk of cognitive impairment and stroke, unprocessed foods appear to be protective,” Dr. Kimberly added.
The study was published online on May 22 in Neurology.
Food Processing Matters
UPFs are highly manipulated, low in protein and fiber, and packed with added ingredients, including sugar, fat, and salt. Examples of UPFs are soft drinks, chips, chocolate, candy, ice cream, sweetened breakfast cereals, packaged soups, chicken nuggets, hot dogs, and fries.
Unprocessed or minimally processed foods include meats such as simple cuts of beef, pork, and chicken, and vegetables and fruits.
Research has shown associations between high UPF consumption and increased risk for metabolic and neurologic disorders.
As reported previously, in the ELSA-Brasil study, higher intake of UPFs was significantly associated with a faster rate of decline in executive and global cognitive function.
Yet, it’s unclear whether the extent of food processing contributes to the risk of adverse neurologic outcomes independent of dietary patterns.
Dr. Kimberly and colleagues examined the association of food processing levels with the risk for cognitive impairment and stroke in the long-running REGARDS study, a large prospective US cohort of Black and White adults aged 45 years and older.
Food processing levels were defined by the NOVA food classification system, which ranges from unprocessed or minimally processed foods (NOVA1) to UPFs (NOVA4). Dietary patterns were characterized based on food frequency questionnaires.
In the cognitive impairment cohort, 768 of 14,175 adults without evidence of impairment at baseline who underwent follow-up testing developed cognitive impairment.
Diet an Opportunity to Protect Brain Health
In multivariable Cox proportional hazards models adjusting for age, sex, high blood pressure, and other factors, a 10% increase in relative intake of UPFs was associated with a 16% higher risk for cognitive impairment (hazard ratio [HR], 1.16). Conversely, a higher intake of unprocessed or minimally processed foods correlated with a 12% lower risk for cognitive impairment (HR, 0.88).
In the stroke cohort, 1108 of 20,243 adults without a history of stroke had a stroke during the follow-up.
In multivariable Cox models, greater intake of UPFs was associated with an 8% increased risk for stroke (HR, 1.08), while greater intake of unprocessed or minimally processed foods correlated with a 9% lower risk for stroke (HR, 0.91).
The effect of UPFs on stroke risk was greater among Black than among White adults (UPF-by-race interaction HR, 1.15).
The associations between UPFs and both cognitive impairment and stroke were independent of adherence to the Mediterranean diet, the Dietary Approaches to Stop Hypertension (DASH) diet, and the Mediterranean-DASH Intervention for Neurodegenerative Delay diet.
These results “highlight the possibility that we have the capacity to maintain our brain health and prevent poor brain health outcomes by focusing on unprocessed foods in the long term,” Dr. Kimberly said.
He cautioned that this was “an observational study and not an interventional study, so we can’t say with certainty that substituting ultra-processed foods with unprocessed foods will definitively improve brain health,” Dr. Kimberly said. “That’s a clinical trial question that has not been done but our results certainly are provocative.”
Consider UPFs in National Guidelines?
The coauthors of an accompanying editorial said the “robust” results from Kimberly and colleagues highlight the “significant role of food processing levels and their relationship with adverse neurologic outcomes, independent of conventional dietary patterns.”
Peipei Gao, MS, with Harvard T.H. Chan School of Public Health, and Zhendong Mei, PhD, with Harvard Medical School, both in Boston, noted that the mechanisms underlying the impact of UPFs on adverse neurologic outcomes “can be attributed not only to their nutritional profiles,” including poor nutrient composition and high glycemic load, “but also to the presence of additives including emulsifiers, colorants, sweeteners, and nitrates/nitrites, which have been associated with disruptions in the gut microbial ecosystem and inflammation.
“Understanding how food processing levels are associated with human health offers a fresh take on the saying ‘you are what you eat,’ ” the editorialists wrote.
This new study, they noted, adds to the evidence by highlighting the link between UPFs and brain health, independent of traditional dietary patterns and “raises questions about whether considerations of UPFs should be included in dietary guidelines, as well as national and global public health policies for improving brain health.”
The editorialists called for large prospective population studies and randomized controlled trials to better understand the link between UPF consumption and brain health. “In addition, mechanistic studies are warranted to identify specific foods, detrimental processes, and additives that play a role in UPFs and their association with neurologic disorders,” they concluded.
Funding for the study was provided by the National Institute of Neurological Disorders and Stroke, the National Institute on Aging, National Institutes of Health, and Department of Health and Human Services. The authors and editorial writers had no relevant disclosures.
A version of this article appeared on Medscape.com.
, new research suggests.
Observations from a large cohort of adults followed for more than 10 years suggested that eating more ultraprocessed foods (UPFs) may increase the risk for cognitive decline and stroke, while eating more unprocessed or minimally processed foods may lower the risk.
“The first key takeaway is that the type of food that we eat matters for brain health, but it’s equally important to think about how it’s made and handled when thinking about brain health,” said study investigator W. Taylor Kimberly, MD, PhD, with Massachusetts General Hospital in Boston.
“The second is that it’s not just all a bad news story because while increased consumption of ultra-processed foods is associated with a higher risk of cognitive impairment and stroke, unprocessed foods appear to be protective,” Dr. Kimberly added.
The study was published online on May 22 in Neurology.
Food Processing Matters
UPFs are highly manipulated, low in protein and fiber, and packed with added ingredients, including sugar, fat, and salt. Examples of UPFs are soft drinks, chips, chocolate, candy, ice cream, sweetened breakfast cereals, packaged soups, chicken nuggets, hot dogs, and fries.
Unprocessed or minimally processed foods include meats such as simple cuts of beef, pork, and chicken, and vegetables and fruits.
Research has shown associations between high UPF consumption and increased risk for metabolic and neurologic disorders.
As reported previously, in the ELSA-Brasil study, higher intake of UPFs was significantly associated with a faster rate of decline in executive and global cognitive function.
Yet, it’s unclear whether the extent of food processing contributes to the risk of adverse neurologic outcomes independent of dietary patterns.
Dr. Kimberly and colleagues examined the association of food processing levels with the risk for cognitive impairment and stroke in the long-running REGARDS study, a large prospective US cohort of Black and White adults aged 45 years and older.
Food processing levels were defined by the NOVA food classification system, which ranges from unprocessed or minimally processed foods (NOVA1) to UPFs (NOVA4). Dietary patterns were characterized based on food frequency questionnaires.
In the cognitive impairment cohort, 768 of 14,175 adults without evidence of impairment at baseline who underwent follow-up testing developed cognitive impairment.
Diet an Opportunity to Protect Brain Health
In multivariable Cox proportional hazards models adjusting for age, sex, high blood pressure, and other factors, a 10% increase in relative intake of UPFs was associated with a 16% higher risk for cognitive impairment (hazard ratio [HR], 1.16). Conversely, a higher intake of unprocessed or minimally processed foods correlated with a 12% lower risk for cognitive impairment (HR, 0.88).
In the stroke cohort, 1108 of 20,243 adults without a history of stroke had a stroke during the follow-up.
In multivariable Cox models, greater intake of UPFs was associated with an 8% increased risk for stroke (HR, 1.08), while greater intake of unprocessed or minimally processed foods correlated with a 9% lower risk for stroke (HR, 0.91).
The effect of UPFs on stroke risk was greater among Black than among White adults (UPF-by-race interaction HR, 1.15).
The associations between UPFs and both cognitive impairment and stroke were independent of adherence to the Mediterranean diet, the Dietary Approaches to Stop Hypertension (DASH) diet, and the Mediterranean-DASH Intervention for Neurodegenerative Delay diet.
These results “highlight the possibility that we have the capacity to maintain our brain health and prevent poor brain health outcomes by focusing on unprocessed foods in the long term,” Dr. Kimberly said.
He cautioned that this was “an observational study and not an interventional study, so we can’t say with certainty that substituting ultra-processed foods with unprocessed foods will definitively improve brain health,” Dr. Kimberly said. “That’s a clinical trial question that has not been done but our results certainly are provocative.”
Consider UPFs in National Guidelines?
The coauthors of an accompanying editorial said the “robust” results from Kimberly and colleagues highlight the “significant role of food processing levels and their relationship with adverse neurologic outcomes, independent of conventional dietary patterns.”
Peipei Gao, MS, with Harvard T.H. Chan School of Public Health, and Zhendong Mei, PhD, with Harvard Medical School, both in Boston, noted that the mechanisms underlying the impact of UPFs on adverse neurologic outcomes “can be attributed not only to their nutritional profiles,” including poor nutrient composition and high glycemic load, “but also to the presence of additives including emulsifiers, colorants, sweeteners, and nitrates/nitrites, which have been associated with disruptions in the gut microbial ecosystem and inflammation.
“Understanding how food processing levels are associated with human health offers a fresh take on the saying ‘you are what you eat,’ ” the editorialists wrote.
This new study, they noted, adds to the evidence by highlighting the link between UPFs and brain health, independent of traditional dietary patterns and “raises questions about whether considerations of UPFs should be included in dietary guidelines, as well as national and global public health policies for improving brain health.”
The editorialists called for large prospective population studies and randomized controlled trials to better understand the link between UPF consumption and brain health. “In addition, mechanistic studies are warranted to identify specific foods, detrimental processes, and additives that play a role in UPFs and their association with neurologic disorders,” they concluded.
Funding for the study was provided by the National Institute of Neurological Disorders and Stroke, the National Institute on Aging, National Institutes of Health, and Department of Health and Human Services. The authors and editorial writers had no relevant disclosures.
A version of this article appeared on Medscape.com.
, new research suggests.
Observations from a large cohort of adults followed for more than 10 years suggested that eating more ultraprocessed foods (UPFs) may increase the risk for cognitive decline and stroke, while eating more unprocessed or minimally processed foods may lower the risk.
“The first key takeaway is that the type of food that we eat matters for brain health, but it’s equally important to think about how it’s made and handled when thinking about brain health,” said study investigator W. Taylor Kimberly, MD, PhD, with Massachusetts General Hospital in Boston.
“The second is that it’s not just all a bad news story because while increased consumption of ultra-processed foods is associated with a higher risk of cognitive impairment and stroke, unprocessed foods appear to be protective,” Dr. Kimberly added.
The study was published online on May 22 in Neurology.
Food Processing Matters
UPFs are highly manipulated, low in protein and fiber, and packed with added ingredients, including sugar, fat, and salt. Examples of UPFs are soft drinks, chips, chocolate, candy, ice cream, sweetened breakfast cereals, packaged soups, chicken nuggets, hot dogs, and fries.
Unprocessed or minimally processed foods include meats such as simple cuts of beef, pork, and chicken, and vegetables and fruits.
Research has shown associations between high UPF consumption and increased risk for metabolic and neurologic disorders.
As reported previously, in the ELSA-Brasil study, higher intake of UPFs was significantly associated with a faster rate of decline in executive and global cognitive function.
Yet, it’s unclear whether the extent of food processing contributes to the risk of adverse neurologic outcomes independent of dietary patterns.
Dr. Kimberly and colleagues examined the association of food processing levels with the risk for cognitive impairment and stroke in the long-running REGARDS study, a large prospective US cohort of Black and White adults aged 45 years and older.
Food processing levels were defined by the NOVA food classification system, which ranges from unprocessed or minimally processed foods (NOVA1) to UPFs (NOVA4). Dietary patterns were characterized based on food frequency questionnaires.
In the cognitive impairment cohort, 768 of 14,175 adults without evidence of impairment at baseline who underwent follow-up testing developed cognitive impairment.
Diet an Opportunity to Protect Brain Health
In multivariable Cox proportional hazards models adjusting for age, sex, high blood pressure, and other factors, a 10% increase in relative intake of UPFs was associated with a 16% higher risk for cognitive impairment (hazard ratio [HR], 1.16). Conversely, a higher intake of unprocessed or minimally processed foods correlated with a 12% lower risk for cognitive impairment (HR, 0.88).
In the stroke cohort, 1108 of 20,243 adults without a history of stroke had a stroke during the follow-up.
In multivariable Cox models, greater intake of UPFs was associated with an 8% increased risk for stroke (HR, 1.08), while greater intake of unprocessed or minimally processed foods correlated with a 9% lower risk for stroke (HR, 0.91).
The effect of UPFs on stroke risk was greater among Black than among White adults (UPF-by-race interaction HR, 1.15).
The associations between UPFs and both cognitive impairment and stroke were independent of adherence to the Mediterranean diet, the Dietary Approaches to Stop Hypertension (DASH) diet, and the Mediterranean-DASH Intervention for Neurodegenerative Delay diet.
These results “highlight the possibility that we have the capacity to maintain our brain health and prevent poor brain health outcomes by focusing on unprocessed foods in the long term,” Dr. Kimberly said.
He cautioned that this was “an observational study and not an interventional study, so we can’t say with certainty that substituting ultra-processed foods with unprocessed foods will definitively improve brain health,” Dr. Kimberly said. “That’s a clinical trial question that has not been done but our results certainly are provocative.”
Consider UPFs in National Guidelines?
The coauthors of an accompanying editorial said the “robust” results from Kimberly and colleagues highlight the “significant role of food processing levels and their relationship with adverse neurologic outcomes, independent of conventional dietary patterns.”
Peipei Gao, MS, with Harvard T.H. Chan School of Public Health, and Zhendong Mei, PhD, with Harvard Medical School, both in Boston, noted that the mechanisms underlying the impact of UPFs on adverse neurologic outcomes “can be attributed not only to their nutritional profiles,” including poor nutrient composition and high glycemic load, “but also to the presence of additives including emulsifiers, colorants, sweeteners, and nitrates/nitrites, which have been associated with disruptions in the gut microbial ecosystem and inflammation.
“Understanding how food processing levels are associated with human health offers a fresh take on the saying ‘you are what you eat,’ ” the editorialists wrote.
This new study, they noted, adds to the evidence by highlighting the link between UPFs and brain health, independent of traditional dietary patterns and “raises questions about whether considerations of UPFs should be included in dietary guidelines, as well as national and global public health policies for improving brain health.”
The editorialists called for large prospective population studies and randomized controlled trials to better understand the link between UPF consumption and brain health. “In addition, mechanistic studies are warranted to identify specific foods, detrimental processes, and additives that play a role in UPFs and their association with neurologic disorders,” they concluded.
Funding for the study was provided by the National Institute of Neurological Disorders and Stroke, the National Institute on Aging, National Institutes of Health, and Department of Health and Human Services. The authors and editorial writers had no relevant disclosures.
A version of this article appeared on Medscape.com.
FROM NEUROLOGY
Fluoride, Water, and Kids’ Brains: It’s Complicated
This transcript has been edited for clarity.
I recently looked back at my folder full of these medical study commentaries, this weekly video series we call Impact Factor, and realized that I’ve been doing this for a long time. More than 400 articles, believe it or not.
I’ve learned a lot in that time — about medicine, of course — but also about how people react to certain topics. If you’ve been with me this whole time, or even for just a chunk of it, you’ll know that I tend to take a measured approach to most topics. No one study is ever truly definitive, after all. But regardless of how even-keeled I may be, there are some topics that I just know in advance are going to be a bit divisive: studies about gun control; studies about vitamin D; and, of course, studies about fluoride.
Shall We Shake This Hornet’s Nest?
The fluoridation of the US water system began in 1945 with the goal of reducing cavities in the population. The CDC named water fluoridation one of the 10 great public health achievements of the 20th century, along with such inarguable achievements as the recognition of tobacco as a health hazard.
But fluoridation has never been without its detractors. One problem is that the spectrum of beliefs about the potential harm of fluoridation is huge. On one end, you have science-based concerns such as the recognition that excessive fluoride intake can cause fluorosis and stain tooth enamel. I’ll note that the EPA regulates fluoride levels — there is a fair amount of naturally occurring fluoride in water tables around the world — to prevent this. And, of course, on the other end of the spectrum, you have beliefs that are essentially conspiracy theories: “They” add fluoride to the water supply to control us.
The challenge for me is that when one “side” of a scientific debate includes the crazy theories, it can be hard to discuss that whole spectrum, since there are those who will see evidence of any adverse fluoride effect as confirmation that the conspiracy theory is true.
I can’t help this. So I’ll just say this up front: I am about to tell you about a study that shows some potential risk from fluoride exposure. I will tell you up front that there are some significant caveats to the study that call the results into question. And I will tell you up front that no one is controlling your mind, or my mind, with fluoride; they do it with social media.
Let’s Dive Into These Shark-Infested, Fluoridated Waters
We’re talking about the study, “Maternal Urinary Fluoride and Child Neurobehavior at Age 36 Months,” which appears in JAMA Network Open.
It’s a study of 229 mother-child pairs from the Los Angeles area. The moms had their urinary fluoride level measured once before 30 weeks of gestation. A neurobehavioral battery called the Preschool Child Behavior Checklist was administered to the children at age 36 months.
The main thing you’ll hear about this study — in headlines, Facebook posts, and manifestos locked in drawers somewhere — is the primary result: A 0.68-mg/L increase in urinary fluoride in the mothers, about 25 percentile points, was associated with a doubling of the risk for neurobehavioral problems in their kids when they were 3 years old.
Yikes.
But this is not a randomized trial. Researchers didn’t randomly assign some women to have high fluoride intake and some women to have low fluoride intake. They knew that other factors that might lead to neurobehavioral problems could also lead to higher fluoride intake. They represent these factors in what’s known as a directed acyclic graph, as seen here, and account for them statistically using a regression equation.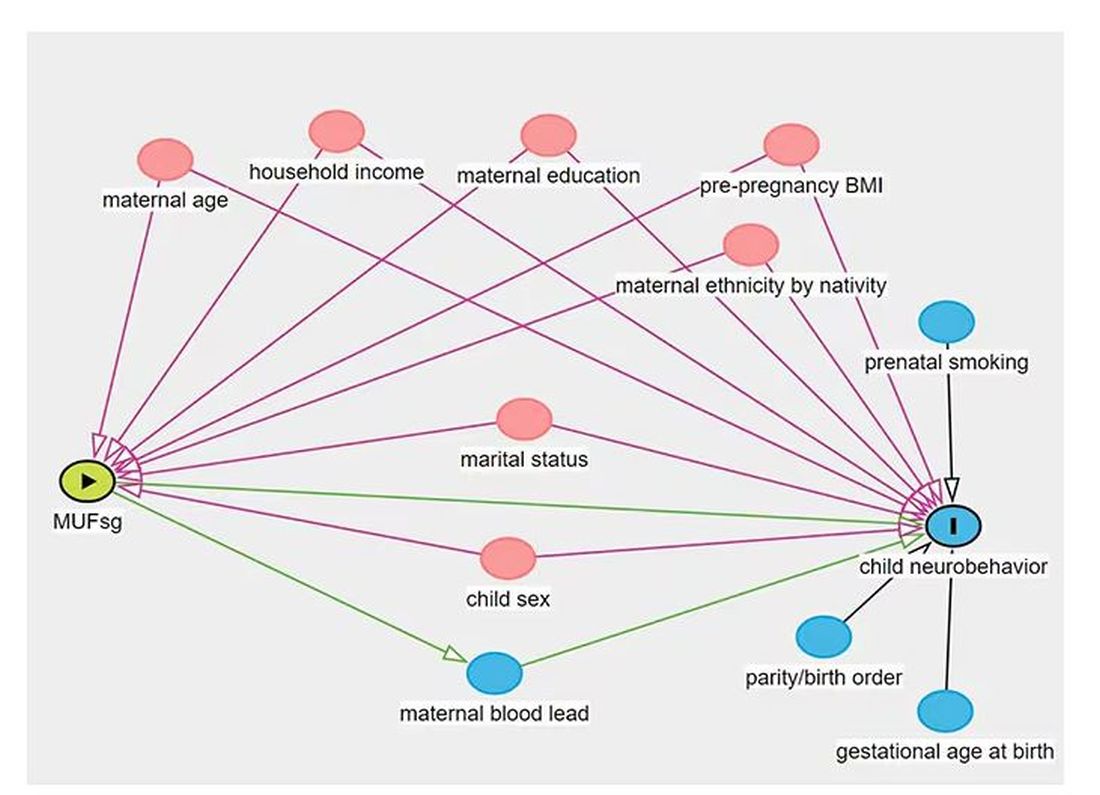
Not represented here are neighborhood characteristics. Los Angeles does not have uniformly fluoridated water, and neurobehavioral problems in kids are strongly linked to stressors in their environments. Fluoride level could be an innocent bystander.
I’m really just describing the classic issue of correlation versus causation here, the bane of all observational research and — let’s be honest — a bit of a crutch that allows us to disregard the results of studies we don’t like, provided the study wasn’t a randomized trial.
But I have a deeper issue with this study than the old “failure to adjust for relevant confounders” thing, as important as that is.
The exposure of interest in this study is maternal urinary fluoride, as measured in a spot sample. It’s not often that I get to go deep on nephrology in this space, but let’s think about that for a second. Let’s assume for a moment that fluoride is toxic to the developing fetal brain, the main concern raised by the results of the study. How would that work? Presumably, mom would be ingesting fluoride from various sources (like the water supply), and that fluoride would get into her blood, and from her blood across the placenta to the baby’s blood, and into the baby’s brain.
Is Urinary Fluoride a Good Measure of Blood Fluoride?
It’s not great. Empirically, we have data that tell us that levels of urine fluoride are not all that similar to levels of serum fluoride. In 2014, a study investigated the correlation between urine and serum fluoride in a cohort of 60 schoolchildren and found a correlation coefficient of around 0.5.
Why isn’t urine fluoride a great proxy for serum fluoride? The most obvious reason is the urine concentration. Human urine concentration can range from about 50 mmol to 1200 mmol (a 24-fold difference) depending on hydration status. Over the course of 24 hours, for example, the amount of fluoride you put out in your urine may be fairly stable in relation to intake, but for a spot urine sample it would be wildly variable. The authors know this, of course, and so they divide the measured urine fluoride by the specific gravity of the urine to give a sort of “dilution adjusted” value. That’s what is actually used in this study. But specific gravity is, itself, an imperfect measure of how dilute the urine is.
This is something that comes up a lot in urinary biomarker research and it’s not that hard to get around. The best thing would be to just measure blood levels of fluoride. The second best option is 24-hour fluoride excretion. After that, the next best thing would be to adjust the spot concentration by other markers of urinary dilution — creatinine or osmolality — as sensitivity analyses. Any of these approaches would lend credence to the results of the study.
Urinary fluoride excretion is pH dependent. The more acidic the urine, the less fluoride is excreted. Many things — including, importantly, diet — affect urine pH. And it is not a stretch to think that diet may also affect the developing fetus. Neither urine pH nor dietary habits were accounted for in this study.
So, here we are. We have an observational study suggesting a harm that may be associated with fluoride. There may be a causal link here, in which case we need further studies to weigh the harm against the more well-established public health benefit. Or, this is all correlation — an illusion created by the limitations of observational data, and the unique challenges of estimating intake from a single urine sample. In other words, this study has something for everyone, fluoride boosters and skeptics alike. Let the arguments begin. But, if possible, leave me out of it.
Dr. Wilson is associate professor of medicine and public health and director of the Clinical and Translational Research Accelerator at Yale University, New Haven, Conn. He has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
This transcript has been edited for clarity.
I recently looked back at my folder full of these medical study commentaries, this weekly video series we call Impact Factor, and realized that I’ve been doing this for a long time. More than 400 articles, believe it or not.
I’ve learned a lot in that time — about medicine, of course — but also about how people react to certain topics. If you’ve been with me this whole time, or even for just a chunk of it, you’ll know that I tend to take a measured approach to most topics. No one study is ever truly definitive, after all. But regardless of how even-keeled I may be, there are some topics that I just know in advance are going to be a bit divisive: studies about gun control; studies about vitamin D; and, of course, studies about fluoride.
Shall We Shake This Hornet’s Nest?
The fluoridation of the US water system began in 1945 with the goal of reducing cavities in the population. The CDC named water fluoridation one of the 10 great public health achievements of the 20th century, along with such inarguable achievements as the recognition of tobacco as a health hazard.
But fluoridation has never been without its detractors. One problem is that the spectrum of beliefs about the potential harm of fluoridation is huge. On one end, you have science-based concerns such as the recognition that excessive fluoride intake can cause fluorosis and stain tooth enamel. I’ll note that the EPA regulates fluoride levels — there is a fair amount of naturally occurring fluoride in water tables around the world — to prevent this. And, of course, on the other end of the spectrum, you have beliefs that are essentially conspiracy theories: “They” add fluoride to the water supply to control us.
The challenge for me is that when one “side” of a scientific debate includes the crazy theories, it can be hard to discuss that whole spectrum, since there are those who will see evidence of any adverse fluoride effect as confirmation that the conspiracy theory is true.
I can’t help this. So I’ll just say this up front: I am about to tell you about a study that shows some potential risk from fluoride exposure. I will tell you up front that there are some significant caveats to the study that call the results into question. And I will tell you up front that no one is controlling your mind, or my mind, with fluoride; they do it with social media.
Let’s Dive Into These Shark-Infested, Fluoridated Waters
We’re talking about the study, “Maternal Urinary Fluoride and Child Neurobehavior at Age 36 Months,” which appears in JAMA Network Open.
It’s a study of 229 mother-child pairs from the Los Angeles area. The moms had their urinary fluoride level measured once before 30 weeks of gestation. A neurobehavioral battery called the Preschool Child Behavior Checklist was administered to the children at age 36 months.
The main thing you’ll hear about this study — in headlines, Facebook posts, and manifestos locked in drawers somewhere — is the primary result: A 0.68-mg/L increase in urinary fluoride in the mothers, about 25 percentile points, was associated with a doubling of the risk for neurobehavioral problems in their kids when they were 3 years old.
Yikes.
But this is not a randomized trial. Researchers didn’t randomly assign some women to have high fluoride intake and some women to have low fluoride intake. They knew that other factors that might lead to neurobehavioral problems could also lead to higher fluoride intake. They represent these factors in what’s known as a directed acyclic graph, as seen here, and account for them statistically using a regression equation.
Not represented here are neighborhood characteristics. Los Angeles does not have uniformly fluoridated water, and neurobehavioral problems in kids are strongly linked to stressors in their environments. Fluoride level could be an innocent bystander.
I’m really just describing the classic issue of correlation versus causation here, the bane of all observational research and — let’s be honest — a bit of a crutch that allows us to disregard the results of studies we don’t like, provided the study wasn’t a randomized trial.
But I have a deeper issue with this study than the old “failure to adjust for relevant confounders” thing, as important as that is.
The exposure of interest in this study is maternal urinary fluoride, as measured in a spot sample. It’s not often that I get to go deep on nephrology in this space, but let’s think about that for a second. Let’s assume for a moment that fluoride is toxic to the developing fetal brain, the main concern raised by the results of the study. How would that work? Presumably, mom would be ingesting fluoride from various sources (like the water supply), and that fluoride would get into her blood, and from her blood across the placenta to the baby’s blood, and into the baby’s brain.
Is Urinary Fluoride a Good Measure of Blood Fluoride?
It’s not great. Empirically, we have data that tell us that levels of urine fluoride are not all that similar to levels of serum fluoride. In 2014, a study investigated the correlation between urine and serum fluoride in a cohort of 60 schoolchildren and found a correlation coefficient of around 0.5.
Why isn’t urine fluoride a great proxy for serum fluoride? The most obvious reason is the urine concentration. Human urine concentration can range from about 50 mmol to 1200 mmol (a 24-fold difference) depending on hydration status. Over the course of 24 hours, for example, the amount of fluoride you put out in your urine may be fairly stable in relation to intake, but for a spot urine sample it would be wildly variable. The authors know this, of course, and so they divide the measured urine fluoride by the specific gravity of the urine to give a sort of “dilution adjusted” value. That’s what is actually used in this study. But specific gravity is, itself, an imperfect measure of how dilute the urine is.
This is something that comes up a lot in urinary biomarker research and it’s not that hard to get around. The best thing would be to just measure blood levels of fluoride. The second best option is 24-hour fluoride excretion. After that, the next best thing would be to adjust the spot concentration by other markers of urinary dilution — creatinine or osmolality — as sensitivity analyses. Any of these approaches would lend credence to the results of the study.
Urinary fluoride excretion is pH dependent. The more acidic the urine, the less fluoride is excreted. Many things — including, importantly, diet — affect urine pH. And it is not a stretch to think that diet may also affect the developing fetus. Neither urine pH nor dietary habits were accounted for in this study.
So, here we are. We have an observational study suggesting a harm that may be associated with fluoride. There may be a causal link here, in which case we need further studies to weigh the harm against the more well-established public health benefit. Or, this is all correlation — an illusion created by the limitations of observational data, and the unique challenges of estimating intake from a single urine sample. In other words, this study has something for everyone, fluoride boosters and skeptics alike. Let the arguments begin. But, if possible, leave me out of it.
Dr. Wilson is associate professor of medicine and public health and director of the Clinical and Translational Research Accelerator at Yale University, New Haven, Conn. He has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
This transcript has been edited for clarity.
I recently looked back at my folder full of these medical study commentaries, this weekly video series we call Impact Factor, and realized that I’ve been doing this for a long time. More than 400 articles, believe it or not.
I’ve learned a lot in that time — about medicine, of course — but also about how people react to certain topics. If you’ve been with me this whole time, or even for just a chunk of it, you’ll know that I tend to take a measured approach to most topics. No one study is ever truly definitive, after all. But regardless of how even-keeled I may be, there are some topics that I just know in advance are going to be a bit divisive: studies about gun control; studies about vitamin D; and, of course, studies about fluoride.
Shall We Shake This Hornet’s Nest?
The fluoridation of the US water system began in 1945 with the goal of reducing cavities in the population. The CDC named water fluoridation one of the 10 great public health achievements of the 20th century, along with such inarguable achievements as the recognition of tobacco as a health hazard.
But fluoridation has never been without its detractors. One problem is that the spectrum of beliefs about the potential harm of fluoridation is huge. On one end, you have science-based concerns such as the recognition that excessive fluoride intake can cause fluorosis and stain tooth enamel. I’ll note that the EPA regulates fluoride levels — there is a fair amount of naturally occurring fluoride in water tables around the world — to prevent this. And, of course, on the other end of the spectrum, you have beliefs that are essentially conspiracy theories: “They” add fluoride to the water supply to control us.
The challenge for me is that when one “side” of a scientific debate includes the crazy theories, it can be hard to discuss that whole spectrum, since there are those who will see evidence of any adverse fluoride effect as confirmation that the conspiracy theory is true.
I can’t help this. So I’ll just say this up front: I am about to tell you about a study that shows some potential risk from fluoride exposure. I will tell you up front that there are some significant caveats to the study that call the results into question. And I will tell you up front that no one is controlling your mind, or my mind, with fluoride; they do it with social media.
Let’s Dive Into These Shark-Infested, Fluoridated Waters
We’re talking about the study, “Maternal Urinary Fluoride and Child Neurobehavior at Age 36 Months,” which appears in JAMA Network Open.
It’s a study of 229 mother-child pairs from the Los Angeles area. The moms had their urinary fluoride level measured once before 30 weeks of gestation. A neurobehavioral battery called the Preschool Child Behavior Checklist was administered to the children at age 36 months.
The main thing you’ll hear about this study — in headlines, Facebook posts, and manifestos locked in drawers somewhere — is the primary result: A 0.68-mg/L increase in urinary fluoride in the mothers, about 25 percentile points, was associated with a doubling of the risk for neurobehavioral problems in their kids when they were 3 years old.
Yikes.
But this is not a randomized trial. Researchers didn’t randomly assign some women to have high fluoride intake and some women to have low fluoride intake. They knew that other factors that might lead to neurobehavioral problems could also lead to higher fluoride intake. They represent these factors in what’s known as a directed acyclic graph, as seen here, and account for them statistically using a regression equation.
Not represented here are neighborhood characteristics. Los Angeles does not have uniformly fluoridated water, and neurobehavioral problems in kids are strongly linked to stressors in their environments. Fluoride level could be an innocent bystander.
I’m really just describing the classic issue of correlation versus causation here, the bane of all observational research and — let’s be honest — a bit of a crutch that allows us to disregard the results of studies we don’t like, provided the study wasn’t a randomized trial.
But I have a deeper issue with this study than the old “failure to adjust for relevant confounders” thing, as important as that is.
The exposure of interest in this study is maternal urinary fluoride, as measured in a spot sample. It’s not often that I get to go deep on nephrology in this space, but let’s think about that for a second. Let’s assume for a moment that fluoride is toxic to the developing fetal brain, the main concern raised by the results of the study. How would that work? Presumably, mom would be ingesting fluoride from various sources (like the water supply), and that fluoride would get into her blood, and from her blood across the placenta to the baby’s blood, and into the baby’s brain.
Is Urinary Fluoride a Good Measure of Blood Fluoride?
It’s not great. Empirically, we have data that tell us that levels of urine fluoride are not all that similar to levels of serum fluoride. In 2014, a study investigated the correlation between urine and serum fluoride in a cohort of 60 schoolchildren and found a correlation coefficient of around 0.5.
Why isn’t urine fluoride a great proxy for serum fluoride? The most obvious reason is the urine concentration. Human urine concentration can range from about 50 mmol to 1200 mmol (a 24-fold difference) depending on hydration status. Over the course of 24 hours, for example, the amount of fluoride you put out in your urine may be fairly stable in relation to intake, but for a spot urine sample it would be wildly variable. The authors know this, of course, and so they divide the measured urine fluoride by the specific gravity of the urine to give a sort of “dilution adjusted” value. That’s what is actually used in this study. But specific gravity is, itself, an imperfect measure of how dilute the urine is.
This is something that comes up a lot in urinary biomarker research and it’s not that hard to get around. The best thing would be to just measure blood levels of fluoride. The second best option is 24-hour fluoride excretion. After that, the next best thing would be to adjust the spot concentration by other markers of urinary dilution — creatinine or osmolality — as sensitivity analyses. Any of these approaches would lend credence to the results of the study.
Urinary fluoride excretion is pH dependent. The more acidic the urine, the less fluoride is excreted. Many things — including, importantly, diet — affect urine pH. And it is not a stretch to think that diet may also affect the developing fetus. Neither urine pH nor dietary habits were accounted for in this study.
So, here we are. We have an observational study suggesting a harm that may be associated with fluoride. There may be a causal link here, in which case we need further studies to weigh the harm against the more well-established public health benefit. Or, this is all correlation — an illusion created by the limitations of observational data, and the unique challenges of estimating intake from a single urine sample. In other words, this study has something for everyone, fluoride boosters and skeptics alike. Let the arguments begin. But, if possible, leave me out of it.
Dr. Wilson is associate professor of medicine and public health and director of the Clinical and Translational Research Accelerator at Yale University, New Haven, Conn. He has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
New Expert Guidance on Antiseizure Medication Use During Pregnancy
New expert guidance to help clinicians manage the treatment of patients with epilepsy during pregnancy has been released.
Issued by the American Academy of Neurology, the American Epilepsy Society, and the Society for Maternal-Fetal Medicine, the new practice guideline covers the use of antiseizure medications (ASMs) and folic acid supplementation before conception and during pregnancy.
“Most children born to people with epilepsy are healthy, but there is a small risk of pregnancy-related problems, partly due to seizures and partly due to the effects of antiseizure medications,” the guidelines’ lead author Alison M. Pack, MD, MPH, professor of neurology and chief of the Epilepsy and Sleep Division, Columbia University, New York City, said in a news release.
“This guideline provides recommendations regarding the effects of antiseizure medications and folic acid supplementation on malformations at birth and the development of children during pregnancy, so that doctors and people with epilepsy can determine which treatments may be best for them,” she added.
The guideline was published online in Neurology.
Why Now?
The new guideline updates the 2009 guidance on epilepsy management during pregnancy. Since then, Dr. Pack told this news organization, there has been a wealth of new data on differential effects of different ASMs — notably, lamotrigine and levetiracetam — the most commonly prescribed medications in this population.
“In this guideline, we were able to assess differential effects of different ASMs on outcomes of interest, including major congenital malformations [MCMs], perinatal outcomes, and neurodevelopmental outcomes. In addition, we looked at the effect of folic acid supplementation on each of these outcomes,” she said.
The overarching goals of care for patients are to “optimize health outcomes both for individuals and their future offspring,” the authors wrote. Shared decision-making, they add, leads to better decision-making by providing a better understanding of the available treatment options and their potential risks, resulting in enhanced decision-making that aligns with personal values.
Clinicians should recommend ASMs that optimize seizure control and fetal outcomes, in the event of a pregnancy, at the earliest possible preconception time, the guideline authors note.
“Overall, treating clinicians need to balance treating the person with epilepsy to control convulsive seizures (generalized tonic-clonic seizures and focal-to-bilateral tonic-clonic seizures) to minimize potential risks to the birth parent and the possible risks of certain ASMs on the fetus if pregnancy occurs,” they wrote.
If a patient is already pregnant, the experts recommend that clinicians “exercise caution” in removing or replacing an ASM that controls convulsive seizures, even if it’s “not an optimal choice” for the fetus.
In addition, they advise that ASM levels should be monitored throughout the pregnancy, guided by individual ASM pharmacokinetics and an individual patient’s clinical presentation. ASM dose, they note, should be adjusted during pregnancy in response to decreasing serum ASM levels or worsening seizure control.
The authors point out that there are limited data on “pregnancy-related outcomes with respect to acetazolamide, eslicarbazepine, ethosuximide, lacosamide, nitrazepam, perampanel, piracetam, pregabalin, rufinamide, stiripentol, tiagabine, and vigabatrin.”
Patients should be informed that the birth prevalence of any major congenital malformation in the general population ranges between 2.4% and 2.9%.
If Feasible, Avoid Valproic Acid
“One of the most important take-home messages is that valproic acid has the highest unadjusted birth prevalence of all major congenital malformations — 9.7% — and the highest unadjusted birth prevalence of neural tube defects at 1.4%,” Dr. Pack said. As a result, the guideline authors advise against using valproic acid, if clinically feasible.
Valproic acid also has the highest prevalence of negative neurodevelopmental outcomes, including a reduction in global IQ and an increased prevalence of autism spectrum disorder (ASD). Patients should be counseled accordingly and advised of the increased risk for ASD and decreased IQ resulting from valproic acid.
Clinicians should consider using lamotrigine, levetiracetam, or oxcarbazepine when appropriate. Serum concentrations of most ASMs have a “defined therapeutic window” for effective seizure control and that concentration may decrease during pregnancy, particularly with lamotrigine and levetiracetam, the authors note.
Phenobarbital, topiramate, and valproic acid should because of the increased risk for cardiac malformations, oral clefts, and urogenital and renal malformations.
Fetal screening for major congenital malformations is recommended to enable early detection and timely intervention in patients treated with any ASM during pregnancy Patients receiving phenobarbital during pregnancy should also undergo fetal cardiac screenings.
Valproic acid and topiramate are also associated with children who are small for their gestational age. To enable early identification of fetal growth restriction, patients taking valproic acid or topiramate should be monitored. In addition, children exposed to these medications in utero should be monitored during childhood to ensure they are meeting age-appropriate developmental milestones.
Folic acid taken during pregnancy can reduce the prevalence of negative neurodevelopment outcomes, but not major congenital malformations, Dr. Pack noted.
“Due to limited available data, we were unable to define an optimal dose of folic acid supplementation beyond at least 0.4 mg/d,” Dr. Pack said. “Future studies, preferably randomized clinical trials, are needed to better define the optimal dose.”
She emphasized that epilepsy is one of the most common neurologic disorders, and 1 in 5 of those affected are people of childbearing potential. Understanding the effects of ASMs on pregnancy outcomes is critical for physicians who manage these patients.
Uncertainty Remains
Commenting for this news organization, Kimford Meador, MD, a professor in the Department of Neurology and Neurological Sciences at Stanford University School of Medicine , Stanford Neuroscience Health Center, Palo Alto, California, noted that the new guidelines reflect the gains in knowledge since 2009 and that the recommendations are “reasonable, based on available data.”
However, “one very important point is how much remains unknown,” said Dr. Meador, who was not involved in writing the current guideline. “Many ASMs have no data, and several have estimates based on small samples or a single observational study.” Thus, “the risks for the majority of ASMs are uncertain.”
Given that randomized trials “are not possible in this population, and that all observational studies are subject to residual confounding, a reliable signal across multiple studies in humans is required to be certain of findings,” he stated.
This practice guideline was developed with financial support from the American Academy of Neurology. Dr. Pack serves on the editorial board for the journal Epilepsy Currents, receives royalties from UpToDate, receives funding from the National Institutes of Health for serving as coinvestigator and site principal investigator for the Maternal Outcomes and Neurodevelopmental Effects of Antiepileptic Drugs (MONEAD) study, and receives funding from Bayer for serving as a co-investigator on a study on women with epilepsy initiating a progestin intrauterine device. One of Dr. Pack’s immediate family members has received personal compensation for serving as an employee of REGENEXBIO. The other authors’ disclosures are listed on the original paper. Dr. Meador has received research support from the National Institutes of Health, Veterans Administration, Eisai, Inc, and Suno Medtronic Navigation, Inc, and the Epilepsy Study Consortium pays Dr. Meador’s university for his research on the Human Epilepsy Project and consultant time related to Eisai, UCB Pharma, and Xenon.
A version of this article first appeared on Medscape.com.
New expert guidance to help clinicians manage the treatment of patients with epilepsy during pregnancy has been released.
Issued by the American Academy of Neurology, the American Epilepsy Society, and the Society for Maternal-Fetal Medicine, the new practice guideline covers the use of antiseizure medications (ASMs) and folic acid supplementation before conception and during pregnancy.
“Most children born to people with epilepsy are healthy, but there is a small risk of pregnancy-related problems, partly due to seizures and partly due to the effects of antiseizure medications,” the guidelines’ lead author Alison M. Pack, MD, MPH, professor of neurology and chief of the Epilepsy and Sleep Division, Columbia University, New York City, said in a news release.
“This guideline provides recommendations regarding the effects of antiseizure medications and folic acid supplementation on malformations at birth and the development of children during pregnancy, so that doctors and people with epilepsy can determine which treatments may be best for them,” she added.
The guideline was published online in Neurology.
Why Now?
The new guideline updates the 2009 guidance on epilepsy management during pregnancy. Since then, Dr. Pack told this news organization, there has been a wealth of new data on differential effects of different ASMs — notably, lamotrigine and levetiracetam — the most commonly prescribed medications in this population.
“In this guideline, we were able to assess differential effects of different ASMs on outcomes of interest, including major congenital malformations [MCMs], perinatal outcomes, and neurodevelopmental outcomes. In addition, we looked at the effect of folic acid supplementation on each of these outcomes,” she said.
The overarching goals of care for patients are to “optimize health outcomes both for individuals and their future offspring,” the authors wrote. Shared decision-making, they add, leads to better decision-making by providing a better understanding of the available treatment options and their potential risks, resulting in enhanced decision-making that aligns with personal values.
Clinicians should recommend ASMs that optimize seizure control and fetal outcomes, in the event of a pregnancy, at the earliest possible preconception time, the guideline authors note.
“Overall, treating clinicians need to balance treating the person with epilepsy to control convulsive seizures (generalized tonic-clonic seizures and focal-to-bilateral tonic-clonic seizures) to minimize potential risks to the birth parent and the possible risks of certain ASMs on the fetus if pregnancy occurs,” they wrote.
If a patient is already pregnant, the experts recommend that clinicians “exercise caution” in removing or replacing an ASM that controls convulsive seizures, even if it’s “not an optimal choice” for the fetus.
In addition, they advise that ASM levels should be monitored throughout the pregnancy, guided by individual ASM pharmacokinetics and an individual patient’s clinical presentation. ASM dose, they note, should be adjusted during pregnancy in response to decreasing serum ASM levels or worsening seizure control.
The authors point out that there are limited data on “pregnancy-related outcomes with respect to acetazolamide, eslicarbazepine, ethosuximide, lacosamide, nitrazepam, perampanel, piracetam, pregabalin, rufinamide, stiripentol, tiagabine, and vigabatrin.”
Patients should be informed that the birth prevalence of any major congenital malformation in the general population ranges between 2.4% and 2.9%.
If Feasible, Avoid Valproic Acid
“One of the most important take-home messages is that valproic acid has the highest unadjusted birth prevalence of all major congenital malformations — 9.7% — and the highest unadjusted birth prevalence of neural tube defects at 1.4%,” Dr. Pack said. As a result, the guideline authors advise against using valproic acid, if clinically feasible.
Valproic acid also has the highest prevalence of negative neurodevelopmental outcomes, including a reduction in global IQ and an increased prevalence of autism spectrum disorder (ASD). Patients should be counseled accordingly and advised of the increased risk for ASD and decreased IQ resulting from valproic acid.
Clinicians should consider using lamotrigine, levetiracetam, or oxcarbazepine when appropriate. Serum concentrations of most ASMs have a “defined therapeutic window” for effective seizure control and that concentration may decrease during pregnancy, particularly with lamotrigine and levetiracetam, the authors note.
Phenobarbital, topiramate, and valproic acid should because of the increased risk for cardiac malformations, oral clefts, and urogenital and renal malformations.
Fetal screening for major congenital malformations is recommended to enable early detection and timely intervention in patients treated with any ASM during pregnancy Patients receiving phenobarbital during pregnancy should also undergo fetal cardiac screenings.
Valproic acid and topiramate are also associated with children who are small for their gestational age. To enable early identification of fetal growth restriction, patients taking valproic acid or topiramate should be monitored. In addition, children exposed to these medications in utero should be monitored during childhood to ensure they are meeting age-appropriate developmental milestones.
Folic acid taken during pregnancy can reduce the prevalence of negative neurodevelopment outcomes, but not major congenital malformations, Dr. Pack noted.
“Due to limited available data, we were unable to define an optimal dose of folic acid supplementation beyond at least 0.4 mg/d,” Dr. Pack said. “Future studies, preferably randomized clinical trials, are needed to better define the optimal dose.”
She emphasized that epilepsy is one of the most common neurologic disorders, and 1 in 5 of those affected are people of childbearing potential. Understanding the effects of ASMs on pregnancy outcomes is critical for physicians who manage these patients.
Uncertainty Remains
Commenting for this news organization, Kimford Meador, MD, a professor in the Department of Neurology and Neurological Sciences at Stanford University School of Medicine , Stanford Neuroscience Health Center, Palo Alto, California, noted that the new guidelines reflect the gains in knowledge since 2009 and that the recommendations are “reasonable, based on available data.”
However, “one very important point is how much remains unknown,” said Dr. Meador, who was not involved in writing the current guideline. “Many ASMs have no data, and several have estimates based on small samples or a single observational study.” Thus, “the risks for the majority of ASMs are uncertain.”
Given that randomized trials “are not possible in this population, and that all observational studies are subject to residual confounding, a reliable signal across multiple studies in humans is required to be certain of findings,” he stated.
This practice guideline was developed with financial support from the American Academy of Neurology. Dr. Pack serves on the editorial board for the journal Epilepsy Currents, receives royalties from UpToDate, receives funding from the National Institutes of Health for serving as coinvestigator and site principal investigator for the Maternal Outcomes and Neurodevelopmental Effects of Antiepileptic Drugs (MONEAD) study, and receives funding from Bayer for serving as a co-investigator on a study on women with epilepsy initiating a progestin intrauterine device. One of Dr. Pack’s immediate family members has received personal compensation for serving as an employee of REGENEXBIO. The other authors’ disclosures are listed on the original paper. Dr. Meador has received research support from the National Institutes of Health, Veterans Administration, Eisai, Inc, and Suno Medtronic Navigation, Inc, and the Epilepsy Study Consortium pays Dr. Meador’s university for his research on the Human Epilepsy Project and consultant time related to Eisai, UCB Pharma, and Xenon.
A version of this article first appeared on Medscape.com.
New expert guidance to help clinicians manage the treatment of patients with epilepsy during pregnancy has been released.
Issued by the American Academy of Neurology, the American Epilepsy Society, and the Society for Maternal-Fetal Medicine, the new practice guideline covers the use of antiseizure medications (ASMs) and folic acid supplementation before conception and during pregnancy.
“Most children born to people with epilepsy are healthy, but there is a small risk of pregnancy-related problems, partly due to seizures and partly due to the effects of antiseizure medications,” the guidelines’ lead author Alison M. Pack, MD, MPH, professor of neurology and chief of the Epilepsy and Sleep Division, Columbia University, New York City, said in a news release.
“This guideline provides recommendations regarding the effects of antiseizure medications and folic acid supplementation on malformations at birth and the development of children during pregnancy, so that doctors and people with epilepsy can determine which treatments may be best for them,” she added.
The guideline was published online in Neurology.
Why Now?
The new guideline updates the 2009 guidance on epilepsy management during pregnancy. Since then, Dr. Pack told this news organization, there has been a wealth of new data on differential effects of different ASMs — notably, lamotrigine and levetiracetam — the most commonly prescribed medications in this population.
“In this guideline, we were able to assess differential effects of different ASMs on outcomes of interest, including major congenital malformations [MCMs], perinatal outcomes, and neurodevelopmental outcomes. In addition, we looked at the effect of folic acid supplementation on each of these outcomes,” she said.
The overarching goals of care for patients are to “optimize health outcomes both for individuals and their future offspring,” the authors wrote. Shared decision-making, they add, leads to better decision-making by providing a better understanding of the available treatment options and their potential risks, resulting in enhanced decision-making that aligns with personal values.
Clinicians should recommend ASMs that optimize seizure control and fetal outcomes, in the event of a pregnancy, at the earliest possible preconception time, the guideline authors note.
“Overall, treating clinicians need to balance treating the person with epilepsy to control convulsive seizures (generalized tonic-clonic seizures and focal-to-bilateral tonic-clonic seizures) to minimize potential risks to the birth parent and the possible risks of certain ASMs on the fetus if pregnancy occurs,” they wrote.
If a patient is already pregnant, the experts recommend that clinicians “exercise caution” in removing or replacing an ASM that controls convulsive seizures, even if it’s “not an optimal choice” for the fetus.
In addition, they advise that ASM levels should be monitored throughout the pregnancy, guided by individual ASM pharmacokinetics and an individual patient’s clinical presentation. ASM dose, they note, should be adjusted during pregnancy in response to decreasing serum ASM levels or worsening seizure control.
The authors point out that there are limited data on “pregnancy-related outcomes with respect to acetazolamide, eslicarbazepine, ethosuximide, lacosamide, nitrazepam, perampanel, piracetam, pregabalin, rufinamide, stiripentol, tiagabine, and vigabatrin.”
Patients should be informed that the birth prevalence of any major congenital malformation in the general population ranges between 2.4% and 2.9%.
If Feasible, Avoid Valproic Acid
“One of the most important take-home messages is that valproic acid has the highest unadjusted birth prevalence of all major congenital malformations — 9.7% — and the highest unadjusted birth prevalence of neural tube defects at 1.4%,” Dr. Pack said. As a result, the guideline authors advise against using valproic acid, if clinically feasible.
Valproic acid also has the highest prevalence of negative neurodevelopmental outcomes, including a reduction in global IQ and an increased prevalence of autism spectrum disorder (ASD). Patients should be counseled accordingly and advised of the increased risk for ASD and decreased IQ resulting from valproic acid.
Clinicians should consider using lamotrigine, levetiracetam, or oxcarbazepine when appropriate. Serum concentrations of most ASMs have a “defined therapeutic window” for effective seizure control and that concentration may decrease during pregnancy, particularly with lamotrigine and levetiracetam, the authors note.
Phenobarbital, topiramate, and valproic acid should because of the increased risk for cardiac malformations, oral clefts, and urogenital and renal malformations.
Fetal screening for major congenital malformations is recommended to enable early detection and timely intervention in patients treated with any ASM during pregnancy Patients receiving phenobarbital during pregnancy should also undergo fetal cardiac screenings.
Valproic acid and topiramate are also associated with children who are small for their gestational age. To enable early identification of fetal growth restriction, patients taking valproic acid or topiramate should be monitored. In addition, children exposed to these medications in utero should be monitored during childhood to ensure they are meeting age-appropriate developmental milestones.
Folic acid taken during pregnancy can reduce the prevalence of negative neurodevelopment outcomes, but not major congenital malformations, Dr. Pack noted.
“Due to limited available data, we were unable to define an optimal dose of folic acid supplementation beyond at least 0.4 mg/d,” Dr. Pack said. “Future studies, preferably randomized clinical trials, are needed to better define the optimal dose.”
She emphasized that epilepsy is one of the most common neurologic disorders, and 1 in 5 of those affected are people of childbearing potential. Understanding the effects of ASMs on pregnancy outcomes is critical for physicians who manage these patients.
Uncertainty Remains
Commenting for this news organization, Kimford Meador, MD, a professor in the Department of Neurology and Neurological Sciences at Stanford University School of Medicine , Stanford Neuroscience Health Center, Palo Alto, California, noted that the new guidelines reflect the gains in knowledge since 2009 and that the recommendations are “reasonable, based on available data.”
However, “one very important point is how much remains unknown,” said Dr. Meador, who was not involved in writing the current guideline. “Many ASMs have no data, and several have estimates based on small samples or a single observational study.” Thus, “the risks for the majority of ASMs are uncertain.”
Given that randomized trials “are not possible in this population, and that all observational studies are subject to residual confounding, a reliable signal across multiple studies in humans is required to be certain of findings,” he stated.
This practice guideline was developed with financial support from the American Academy of Neurology. Dr. Pack serves on the editorial board for the journal Epilepsy Currents, receives royalties from UpToDate, receives funding from the National Institutes of Health for serving as coinvestigator and site principal investigator for the Maternal Outcomes and Neurodevelopmental Effects of Antiepileptic Drugs (MONEAD) study, and receives funding from Bayer for serving as a co-investigator on a study on women with epilepsy initiating a progestin intrauterine device. One of Dr. Pack’s immediate family members has received personal compensation for serving as an employee of REGENEXBIO. The other authors’ disclosures are listed on the original paper. Dr. Meador has received research support from the National Institutes of Health, Veterans Administration, Eisai, Inc, and Suno Medtronic Navigation, Inc, and the Epilepsy Study Consortium pays Dr. Meador’s university for his research on the Human Epilepsy Project and consultant time related to Eisai, UCB Pharma, and Xenon.
A version of this article first appeared on Medscape.com.
FROM NEUROLOGY
Global Analysis Identifies Drugs Associated With SJS-TEN in Children
TOPLINE:
METHODOLOGY:
- SJS and TEN are rare, life-threatening mucocutaneous reactions mainly associated with medications, but large pharmacovigilance studies of drugs associated with SJS-TEN in the pediatric population are still lacking.
- Using the WHO’s pharmacovigilance database (VigiBase) containing individual case safety reports from January 1967 to July 2022, researchers identified 7342 adverse drug reaction reports of SJS-TEN in children (younger than 18 years; median age, 9 years) in all six continents. Median onset was 5 days, and 3.2% were fatal.
- They analyzed drugs reported as suspected treatments, and for each molecule, they performed a case–non-case study to assess a potential pharmacovigilance signal by computing the information component (IC).
- A positive IC value suggested more frequent reporting of a specific drug-adverse reaction pair. A positive IC025, a traditional threshold for statistical signal detection, is suggestive of a potential pharmacovigilance signal.
TAKEAWAY:
- Overall, 165 drugs were associated with a diagnosis of SJS-TEN; antiepileptic and anti-infectious drugs were the most common drug classes represented.
- The five most frequently reported drugs were carbamazepine (11.7%), lamotrigine (10.6%), sulfamethoxazole-trimethoprim (9%), acetaminophen (8.4%), and phenytoin (6.6%). The five drugs with the highest IC025 were lamotrigine, carbamazepine, phenobarbital, phenytoin, and nimesulide.
- All antiepileptics, many antibiotic families, dapsone, antiretroviral drugs, some antifungal drugs, and nonsteroidal anti-inflammatory drugs were identified in reports, with penicillins the most frequently reported antibiotic family and sulfonamides having the strongest pharmacovigilance signal.
- Vaccines were not associated with significant signals.
IN PRACTICE:
The study provides an update on “the spectrum of drugs potentially associated with SJS-TEN in the pediatric population,” the authors concluded, and “underlines the importance of reporting to pharmacovigilance the suspicion of this severe side effect of drugs with the most precise and detailed clinical description possible.”
SOURCE:
The study, led by Pauline Bataille, MD, of the Department of Pediatric Dermatology, Hôpital Necker-Enfants Malades, Paris City University, France, was published online in the Journal of the European Academy of Dermatology and Venereology.
LIMITATIONS:
Limitations include the possibility that some cases could have had an infectious or idiopathic cause not related to a drug and the lack of detailed clinical data in the database.
DISCLOSURES:
This study did not receive any funding. The authors declared no conflict of interest.
A version of this article first appeared on Medscape.com.
TOPLINE:
METHODOLOGY:
- SJS and TEN are rare, life-threatening mucocutaneous reactions mainly associated with medications, but large pharmacovigilance studies of drugs associated with SJS-TEN in the pediatric population are still lacking.
- Using the WHO’s pharmacovigilance database (VigiBase) containing individual case safety reports from January 1967 to July 2022, researchers identified 7342 adverse drug reaction reports of SJS-TEN in children (younger than 18 years; median age, 9 years) in all six continents. Median onset was 5 days, and 3.2% were fatal.
- They analyzed drugs reported as suspected treatments, and for each molecule, they performed a case–non-case study to assess a potential pharmacovigilance signal by computing the information component (IC).
- A positive IC value suggested more frequent reporting of a specific drug-adverse reaction pair. A positive IC025, a traditional threshold for statistical signal detection, is suggestive of a potential pharmacovigilance signal.
TAKEAWAY:
- Overall, 165 drugs were associated with a diagnosis of SJS-TEN; antiepileptic and anti-infectious drugs were the most common drug classes represented.
- The five most frequently reported drugs were carbamazepine (11.7%), lamotrigine (10.6%), sulfamethoxazole-trimethoprim (9%), acetaminophen (8.4%), and phenytoin (6.6%). The five drugs with the highest IC025 were lamotrigine, carbamazepine, phenobarbital, phenytoin, and nimesulide.
- All antiepileptics, many antibiotic families, dapsone, antiretroviral drugs, some antifungal drugs, and nonsteroidal anti-inflammatory drugs were identified in reports, with penicillins the most frequently reported antibiotic family and sulfonamides having the strongest pharmacovigilance signal.
- Vaccines were not associated with significant signals.
IN PRACTICE:
The study provides an update on “the spectrum of drugs potentially associated with SJS-TEN in the pediatric population,” the authors concluded, and “underlines the importance of reporting to pharmacovigilance the suspicion of this severe side effect of drugs with the most precise and detailed clinical description possible.”
SOURCE:
The study, led by Pauline Bataille, MD, of the Department of Pediatric Dermatology, Hôpital Necker-Enfants Malades, Paris City University, France, was published online in the Journal of the European Academy of Dermatology and Venereology.
LIMITATIONS:
Limitations include the possibility that some cases could have had an infectious or idiopathic cause not related to a drug and the lack of detailed clinical data in the database.
DISCLOSURES:
This study did not receive any funding. The authors declared no conflict of interest.
A version of this article first appeared on Medscape.com.
TOPLINE:
METHODOLOGY:
- SJS and TEN are rare, life-threatening mucocutaneous reactions mainly associated with medications, but large pharmacovigilance studies of drugs associated with SJS-TEN in the pediatric population are still lacking.
- Using the WHO’s pharmacovigilance database (VigiBase) containing individual case safety reports from January 1967 to July 2022, researchers identified 7342 adverse drug reaction reports of SJS-TEN in children (younger than 18 years; median age, 9 years) in all six continents. Median onset was 5 days, and 3.2% were fatal.
- They analyzed drugs reported as suspected treatments, and for each molecule, they performed a case–non-case study to assess a potential pharmacovigilance signal by computing the information component (IC).
- A positive IC value suggested more frequent reporting of a specific drug-adverse reaction pair. A positive IC025, a traditional threshold for statistical signal detection, is suggestive of a potential pharmacovigilance signal.
TAKEAWAY:
- Overall, 165 drugs were associated with a diagnosis of SJS-TEN; antiepileptic and anti-infectious drugs were the most common drug classes represented.
- The five most frequently reported drugs were carbamazepine (11.7%), lamotrigine (10.6%), sulfamethoxazole-trimethoprim (9%), acetaminophen (8.4%), and phenytoin (6.6%). The five drugs with the highest IC025 were lamotrigine, carbamazepine, phenobarbital, phenytoin, and nimesulide.
- All antiepileptics, many antibiotic families, dapsone, antiretroviral drugs, some antifungal drugs, and nonsteroidal anti-inflammatory drugs were identified in reports, with penicillins the most frequently reported antibiotic family and sulfonamides having the strongest pharmacovigilance signal.
- Vaccines were not associated with significant signals.
IN PRACTICE:
The study provides an update on “the spectrum of drugs potentially associated with SJS-TEN in the pediatric population,” the authors concluded, and “underlines the importance of reporting to pharmacovigilance the suspicion of this severe side effect of drugs with the most precise and detailed clinical description possible.”
SOURCE:
The study, led by Pauline Bataille, MD, of the Department of Pediatric Dermatology, Hôpital Necker-Enfants Malades, Paris City University, France, was published online in the Journal of the European Academy of Dermatology and Venereology.
LIMITATIONS:
Limitations include the possibility that some cases could have had an infectious or idiopathic cause not related to a drug and the lack of detailed clinical data in the database.
DISCLOSURES:
This study did not receive any funding. The authors declared no conflict of interest.
A version of this article first appeared on Medscape.com.
Lecanemab’s Promise and Peril: Alzheimer’s Treatment Dilemma
Clinicians interested in treating patients with symptoms of mild cognitive impairment or mild dementia should carefully analyze the potential benefits and harms of monoclonal amyloid beta therapy, including likelihood of side effects and overall burden on the patient, according to researchers at the annual meeting of the American Geriatrics Society (AGS).
Lecanemab (Leqembi) may help some patients by lowering the level of beta-amyloid protein in the brain. Results from a phase 3 trial presented at the conference showed participants with Alzheimer’s disease had a 27% slower progression of the disease compared with placebo.
But clinicians must weigh that advantage against risks and contraindications, according to Esther Oh, MD, PhD, an associate professor in the Division of Geriatric Medicine and Gerontology and co-director of the Johns Hopkins Memory and Alzheimer’s Treatment Center, Johns Hopkins University, Baltimore, Maryland, who spoke during a plenary session. Lecanemab gained accelerated approval by the US Food and Drug Administration in January 2023 and full approval in July 2023.
The results from CLARITY, an 18-month, multicenter, double-blind trial involving 1795 participants aged 50-90 years, showed that the variation between treatment and placebo did not meet the criteria for a minimum clinically important difference for mild cognitive impairment or mild Alzheimer’s disease.
Even more concerning to Dr. Oh was the rate of amyloid-related abnormalities on brain imaging, which can cause brain edema and hemorrhage (12.6% and 17.3%, respectively). Almost 85% of cases were asymptomatic.
The risk for abnormalities indicates that thrombolytics are contraindicated for patients taking the drug, according to Dr. Oh.
“Appropriate use recommendations exclude vitamin K antagonists such as warfarin, direct oral anticoagulants and heparin, although aspirin and other antiplatelet agents are allowed,” Dr. Oh said during the presentation.
Blood biomarkers, PET imaging, and levels of amyloid-beta proteins in cerebrospinal fluid are used to determine eligibility for lecanemab. However, tau biomarkers may indicate signs of cognitive impairment decades prior to symptoms. Some evidence indicates that the drug may be more effective in individuals with low tau levels that are evident in earlier stages of disease. Tau can also be determined from cerebrospinal fluid, however, “we do not factor in tau protein as a biomarker for treatment eligibility, but this may become an important biomarker in the future,” Dr. Oh said.
Lecanemab is cost-prohibitive for many patients, with an annual price tag of $26,000. Treatment also requires monthly infusions, a PET, intravenous administration, lab work, multiple MRIs, and potentially an APOE4 serum test.
Medicare covers the majority of services, but patients are responsible for deductibles and copays, an estimated $7000 annually, according to Shari Ling, MD, deputy chief medical officer with the US Centers for Medicare & Medicaid Services, who also spoke during the session. Supplemental or other insurance such as Medicaid are also not included in this estimate.
The Medicare population is growing more complex over time, Dr. Ling said. In 2021, 54% of beneficiaries had five or more comorbidities, which can affect eligibility for lecanemab.
“Across the healthcare system, we are learning what is necessary for coordination of delivery, for evaluation of people who receive these treatments, and for the care that is not anticipated,” Dr. Ling noted.
Neither speaker reported any financial conflicts of interest.
A version of this article first appeared on Medscape.com.
Clinicians interested in treating patients with symptoms of mild cognitive impairment or mild dementia should carefully analyze the potential benefits and harms of monoclonal amyloid beta therapy, including likelihood of side effects and overall burden on the patient, according to researchers at the annual meeting of the American Geriatrics Society (AGS).
Lecanemab (Leqembi) may help some patients by lowering the level of beta-amyloid protein in the brain. Results from a phase 3 trial presented at the conference showed participants with Alzheimer’s disease had a 27% slower progression of the disease compared with placebo.
But clinicians must weigh that advantage against risks and contraindications, according to Esther Oh, MD, PhD, an associate professor in the Division of Geriatric Medicine and Gerontology and co-director of the Johns Hopkins Memory and Alzheimer’s Treatment Center, Johns Hopkins University, Baltimore, Maryland, who spoke during a plenary session. Lecanemab gained accelerated approval by the US Food and Drug Administration in January 2023 and full approval in July 2023.
The results from CLARITY, an 18-month, multicenter, double-blind trial involving 1795 participants aged 50-90 years, showed that the variation between treatment and placebo did not meet the criteria for a minimum clinically important difference for mild cognitive impairment or mild Alzheimer’s disease.
Even more concerning to Dr. Oh was the rate of amyloid-related abnormalities on brain imaging, which can cause brain edema and hemorrhage (12.6% and 17.3%, respectively). Almost 85% of cases were asymptomatic.
The risk for abnormalities indicates that thrombolytics are contraindicated for patients taking the drug, according to Dr. Oh.
“Appropriate use recommendations exclude vitamin K antagonists such as warfarin, direct oral anticoagulants and heparin, although aspirin and other antiplatelet agents are allowed,” Dr. Oh said during the presentation.
Blood biomarkers, PET imaging, and levels of amyloid-beta proteins in cerebrospinal fluid are used to determine eligibility for lecanemab. However, tau biomarkers may indicate signs of cognitive impairment decades prior to symptoms. Some evidence indicates that the drug may be more effective in individuals with low tau levels that are evident in earlier stages of disease. Tau can also be determined from cerebrospinal fluid, however, “we do not factor in tau protein as a biomarker for treatment eligibility, but this may become an important biomarker in the future,” Dr. Oh said.
Lecanemab is cost-prohibitive for many patients, with an annual price tag of $26,000. Treatment also requires monthly infusions, a PET, intravenous administration, lab work, multiple MRIs, and potentially an APOE4 serum test.
Medicare covers the majority of services, but patients are responsible for deductibles and copays, an estimated $7000 annually, according to Shari Ling, MD, deputy chief medical officer with the US Centers for Medicare & Medicaid Services, who also spoke during the session. Supplemental or other insurance such as Medicaid are also not included in this estimate.
The Medicare population is growing more complex over time, Dr. Ling said. In 2021, 54% of beneficiaries had five or more comorbidities, which can affect eligibility for lecanemab.
“Across the healthcare system, we are learning what is necessary for coordination of delivery, for evaluation of people who receive these treatments, and for the care that is not anticipated,” Dr. Ling noted.
Neither speaker reported any financial conflicts of interest.
A version of this article first appeared on Medscape.com.
Clinicians interested in treating patients with symptoms of mild cognitive impairment or mild dementia should carefully analyze the potential benefits and harms of monoclonal amyloid beta therapy, including likelihood of side effects and overall burden on the patient, according to researchers at the annual meeting of the American Geriatrics Society (AGS).
Lecanemab (Leqembi) may help some patients by lowering the level of beta-amyloid protein in the brain. Results from a phase 3 trial presented at the conference showed participants with Alzheimer’s disease had a 27% slower progression of the disease compared with placebo.
But clinicians must weigh that advantage against risks and contraindications, according to Esther Oh, MD, PhD, an associate professor in the Division of Geriatric Medicine and Gerontology and co-director of the Johns Hopkins Memory and Alzheimer’s Treatment Center, Johns Hopkins University, Baltimore, Maryland, who spoke during a plenary session. Lecanemab gained accelerated approval by the US Food and Drug Administration in January 2023 and full approval in July 2023.
The results from CLARITY, an 18-month, multicenter, double-blind trial involving 1795 participants aged 50-90 years, showed that the variation between treatment and placebo did not meet the criteria for a minimum clinically important difference for mild cognitive impairment or mild Alzheimer’s disease.
Even more concerning to Dr. Oh was the rate of amyloid-related abnormalities on brain imaging, which can cause brain edema and hemorrhage (12.6% and 17.3%, respectively). Almost 85% of cases were asymptomatic.
The risk for abnormalities indicates that thrombolytics are contraindicated for patients taking the drug, according to Dr. Oh.
“Appropriate use recommendations exclude vitamin K antagonists such as warfarin, direct oral anticoagulants and heparin, although aspirin and other antiplatelet agents are allowed,” Dr. Oh said during the presentation.
Blood biomarkers, PET imaging, and levels of amyloid-beta proteins in cerebrospinal fluid are used to determine eligibility for lecanemab. However, tau biomarkers may indicate signs of cognitive impairment decades prior to symptoms. Some evidence indicates that the drug may be more effective in individuals with low tau levels that are evident in earlier stages of disease. Tau can also be determined from cerebrospinal fluid, however, “we do not factor in tau protein as a biomarker for treatment eligibility, but this may become an important biomarker in the future,” Dr. Oh said.
Lecanemab is cost-prohibitive for many patients, with an annual price tag of $26,000. Treatment also requires monthly infusions, a PET, intravenous administration, lab work, multiple MRIs, and potentially an APOE4 serum test.
Medicare covers the majority of services, but patients are responsible for deductibles and copays, an estimated $7000 annually, according to Shari Ling, MD, deputy chief medical officer with the US Centers for Medicare & Medicaid Services, who also spoke during the session. Supplemental or other insurance such as Medicaid are also not included in this estimate.
The Medicare population is growing more complex over time, Dr. Ling said. In 2021, 54% of beneficiaries had five or more comorbidities, which can affect eligibility for lecanemab.
“Across the healthcare system, we are learning what is necessary for coordination of delivery, for evaluation of people who receive these treatments, and for the care that is not anticipated,” Dr. Ling noted.
Neither speaker reported any financial conflicts of interest.
A version of this article first appeared on Medscape.com.
FROM AGS 2024