User login
Which perioperative strategies for transvaginal cervical cerclage are backed by data?
In contemporary medical practice, we expect our clinical actions to reflect the best and most current evidence. In many cases, however, the evidence available to us is weak or irrelevant. In their investigation, Berghella and colleagues set out to assess the quality of evidence in the setting of transvaginal cervical cerclage by reviewing the published data on selected perioperative strategies. They elected to perform a systematic review, as opposed to a narrative review (a simple opinion piece), because this approach follows an explicit process designed to limit bias and random error in the interpretation of scientific research.
The studies they analyzed vary from observational investigations to randomized trials, generating considerable heterogeneity in the data. Therefore, it would not have been feasible or appropriate for them to combine the results in a quantitative review (ie, meta-analysis). Their solution: to limit the analysis to a qualitative systematic review.
The term “systematic review” implies that investigators have an accurate and comprehensive understanding of existent data, with each study representing one contribution to a much larger body of knowledge. Over the years, Berghella and colleagues have contributed extensively to the literature on cervical cerclage and are well qualified to provide an analytic framework for the flood of published information on this practice. Although they focused primarily on how to perform cerclage, a discussion of when to perform cerclage cannot be separated from any consideration of efficacy.
When, exactly, is cerclage indicated?
The original indication for cerclage, established more than 50 years ago, required both a history of second-trimester loss and asymptomatic cervical changes in the current pregnancy. Since then, many cerclages have been performed on the basis of history alone or on current cervical changes regardless of history. However, the most recent professional guidelines reconfirm that any cerclage procedure should be supported by both historical and contemporaneous findings.1,2
Investigators have demonstrated that the measurement of cervical length by transvaginal ultrasound should generally be an integral part of clinical evaluation for asymptomatic cervical changes. Indeed, sonographic assessment has emerged as a tool capable of reducing “overcall” and unnecessary intervention.3 On the other hand, a meta-analysis of four randomized trials of ultrasound-indicated cerclage found it to be beneficial in women with a short cervix only if they also had a history of preterm delivery.4
In addition, randomized trials have documented a benefit for cerclage in two other clinical contexts:
- Results from a secondary analysis of data from a large randomized trial published in 1993 suggest that elective cerclage can be based on history alone in women with three or more second-trimester losses or preterm births.5
- Authors of a randomized trial published in 2003 suggested that women with advanced cervical changes, such as dilatation of the external os with exposure of the fetal membranes, may benefit from “emergency” cerclage even in the absence of a prior preterm delivery.6
How the data were analyzed
The data included in the review were analyzed separately, according to three widely accepted indications for cerclage:
- history-indicated: a history of three or more second-trimester losses and/or preterm births
- ultrasound-indicated: ultrasonographic detection of a cervical length of less than 25 mm, as measured by transvaginal ultrasound, in a woman with a history of second-trimester loss or preterm birth
- physical-examination–indicated: physical examination (manual or with a speculum) that confirms a dilated cervix.
Granted, this terminology can be confusing, as in the case of ultrasound-indicated cerclage, which includes aspects of the patient’s history. Moreover, I doubt that the studies included in this analysis always adhered to these definitions. The heterogeneity of the study population and the ambiguity of these definitions may limit the applicability of findings. In fact, they constitute the major (albeit practically unavoidable) limitation of this review.
The optimal approach to subclinical infection is unclear
Although there is a consensus that overt intra-amniotic infection is an absolute contraindication to cerclage, the implications of subclinical intra-amniotic infection in asymptomatic women are unclear. About 50% of women considered for emergency cerclage are likely to have intra-amniotic infection.7 An ongoing randomized trial is expected to elucidate the benefit of precerclage amniocentesis in such cases.
The sonographic detection of sludge in the amniotic fluid also has been associated with intra-amniotic infection. However, after analyzing the data, Berghella and colleagues did not find adequate justification for amniocentesis in this setting. A more practical question might be whether cerclage is advisable at all when sludge is present. Data from a recently reported abstract suggest that the presence of sludge increases the likelihood of early preterm birth independent of cervical length.8
Other gray areas
Another absolute contraindication to cerclage is the presence of painful uterine contractions in a woman exhibiting cervical change. The study findings seemed to imply that when uterine contractions are detected via tocodynamometric monitoring but are not experienced by the patient, cerclage may be appropriate. In my opinion, this issue represents another open clinical question.
On at least one occasion, I have discovered that the patient scheduled for cerclage has not undergone preoperative screening for genital tract infections. In such a scenario, it is unclear whether it is best to cancel the procedure or to proceed. Berghella and colleagues concluded that evidence is insufficient to support routine, universal screening for genital tract infection prior to cerclage. In the absence of risk factors for infection, it may be appropriate to proceed with cerclage.
The issue of combined use of cerclage and 17α hydroxyprogesterone caproate also was addressed in this review. Berghella and colleagues recommended that progestin supplementation be continued if cerclage is subsequently performed. I, too, follow this approach, although the two interventions (progestin followed by cerclage) have not been studied in this sequence.
Related article: A stepwise approach to cervical cerclage (June 2012)
Data are limited, and conflicting, on the use of 17α hydroxyprogesterone caproate after cerclage. A small randomized trial suggested benefit,9 while a retrospective cohort study10 and a secondary analysis of data from a randomized trial of cerclage11 found no benefit for the addition of 17α hydroxyprogesterone caproate.
Berghella and colleagues advocated against reinforcing (second or repeat) cerclage, an intervention that may be associated with a higher incidence of preterm birth.12 Accordingly, continuing sonographic assessment of cervical length after cerclage may be futile. What this evidence means for practice
Increasing evidence suggests that cervical shortening is not limited to an innate or acquired cervical weakness but represents an early, asymptomatic phase on the pathway to preterm birth. Nevertheless, cerclage continues to be practiced widely and has been shown to be beneficial in selected populations.
We clinicians often face complex cases of presumed cervical insufficiency for which there are no simple or unequivocal recommendations. This appraisal by Berghella and colleagues will help us separate evidence-validated approaches from misconceptions in the technical aspects of cerclage.
--Alex C. Vidaeff, MD, MPH
We want to hear from you! Tell us what you think.
- Royal College of Obstetricians and Gynaecologists. Cervical cerclage [guidelines]. http://www.rcog.org.uk/womens-health/clinical-guidance/cervical-cerclage-green-top-60. Accessed September 18, 2013.
- ACOG Practice Bulletin #130: Prediction and prevention of preterm birth. Obstet Gynecol. 2012;120:964–973.
- Grimes-Dennis J, Berghella V. Cervical length and prediction of preterm delivery. Curr Opin Obstet Gynecol. 2007;19(2):191–195.
- Berghella V, Odibo AO, To MS, et al. Cerclage for short cervix on ultrasound: meta-analysis of trials using individual patient-level data. Obstet Gynecol. 2005;106(1):181–189.
- Working Party on Cervical Cerclage. Final report of the Medical Research Council/Royal College of Obstetricians and Gynaecologists multicenter randomised trial of cervical cerclage. Br J Obstet Gynaecol. 1993;100(6):516–523.
- Althuisius SM, Dekker GA, Hummel P, van Geijin HP. Cervical incompetence prevention randomized cerclage trial: emergency cerclage with bed rest versus bed rest alone. Am J Obstet Gynecol. 2003;189(4):907–910.
- Romero R, Gonzalez R, Sepulveda W, et al. Infection and labor. VIII. Microbial invasion of the amniotic cavity in patients with suspected cervical incompetence: prevalence and clinical significance. Am J Obstet Gynecol. 1992(4 Pt 1);167:1086–1091.
- Saade G. Cervical funneling or intraamniotic debris and preterm birth in nulliparous women with short cervix. Am J Obstet Gynecol. 2013;208(1):S4.
- Yemini M, Borenstein R, Dreazen E, et al. Prevention of premature labor by 17 alpha-hydroxyprogesterone caproate. Am J Obstet Gynecol. 1985;151(5):574–577.
- Rebarber A, Cleary-Goldman J, Istwan NB, et al. The use of 17 alpha-hydroxyprogesterone caproate (17P) in women with cervical cerclage. Am J Perinatol. 2008;25(5):271–275.
- Berghella V, Figueroa D, Szychowski JM, et al. 17 alpha-hydroxyprogesterone caproate for the prevention of preterm birth in women with prior preterm birth and a short cervical length. Am J Obstet Gynecol. 2010;202(4):351.e1–e6.
- Baxter JK, Airoldi J, Berghella V. Short cervical length after history-indicated cerclage: is a reinforcing cerclage beneficial? Am J Obstet Gynecol. 2005;193(3 Pt 2):1204–1207.
In contemporary medical practice, we expect our clinical actions to reflect the best and most current evidence. In many cases, however, the evidence available to us is weak or irrelevant. In their investigation, Berghella and colleagues set out to assess the quality of evidence in the setting of transvaginal cervical cerclage by reviewing the published data on selected perioperative strategies. They elected to perform a systematic review, as opposed to a narrative review (a simple opinion piece), because this approach follows an explicit process designed to limit bias and random error in the interpretation of scientific research.
The studies they analyzed vary from observational investigations to randomized trials, generating considerable heterogeneity in the data. Therefore, it would not have been feasible or appropriate for them to combine the results in a quantitative review (ie, meta-analysis). Their solution: to limit the analysis to a qualitative systematic review.
The term “systematic review” implies that investigators have an accurate and comprehensive understanding of existent data, with each study representing one contribution to a much larger body of knowledge. Over the years, Berghella and colleagues have contributed extensively to the literature on cervical cerclage and are well qualified to provide an analytic framework for the flood of published information on this practice. Although they focused primarily on how to perform cerclage, a discussion of when to perform cerclage cannot be separated from any consideration of efficacy.
When, exactly, is cerclage indicated?
The original indication for cerclage, established more than 50 years ago, required both a history of second-trimester loss and asymptomatic cervical changes in the current pregnancy. Since then, many cerclages have been performed on the basis of history alone or on current cervical changes regardless of history. However, the most recent professional guidelines reconfirm that any cerclage procedure should be supported by both historical and contemporaneous findings.1,2
Investigators have demonstrated that the measurement of cervical length by transvaginal ultrasound should generally be an integral part of clinical evaluation for asymptomatic cervical changes. Indeed, sonographic assessment has emerged as a tool capable of reducing “overcall” and unnecessary intervention.3 On the other hand, a meta-analysis of four randomized trials of ultrasound-indicated cerclage found it to be beneficial in women with a short cervix only if they also had a history of preterm delivery.4
In addition, randomized trials have documented a benefit for cerclage in two other clinical contexts:
- Results from a secondary analysis of data from a large randomized trial published in 1993 suggest that elective cerclage can be based on history alone in women with three or more second-trimester losses or preterm births.5
- Authors of a randomized trial published in 2003 suggested that women with advanced cervical changes, such as dilatation of the external os with exposure of the fetal membranes, may benefit from “emergency” cerclage even in the absence of a prior preterm delivery.6
How the data were analyzed
The data included in the review were analyzed separately, according to three widely accepted indications for cerclage:
- history-indicated: a history of three or more second-trimester losses and/or preterm births
- ultrasound-indicated: ultrasonographic detection of a cervical length of less than 25 mm, as measured by transvaginal ultrasound, in a woman with a history of second-trimester loss or preterm birth
- physical-examination–indicated: physical examination (manual or with a speculum) that confirms a dilated cervix.
Granted, this terminology can be confusing, as in the case of ultrasound-indicated cerclage, which includes aspects of the patient’s history. Moreover, I doubt that the studies included in this analysis always adhered to these definitions. The heterogeneity of the study population and the ambiguity of these definitions may limit the applicability of findings. In fact, they constitute the major (albeit practically unavoidable) limitation of this review.
The optimal approach to subclinical infection is unclear
Although there is a consensus that overt intra-amniotic infection is an absolute contraindication to cerclage, the implications of subclinical intra-amniotic infection in asymptomatic women are unclear. About 50% of women considered for emergency cerclage are likely to have intra-amniotic infection.7 An ongoing randomized trial is expected to elucidate the benefit of precerclage amniocentesis in such cases.
The sonographic detection of sludge in the amniotic fluid also has been associated with intra-amniotic infection. However, after analyzing the data, Berghella and colleagues did not find adequate justification for amniocentesis in this setting. A more practical question might be whether cerclage is advisable at all when sludge is present. Data from a recently reported abstract suggest that the presence of sludge increases the likelihood of early preterm birth independent of cervical length.8
Other gray areas
Another absolute contraindication to cerclage is the presence of painful uterine contractions in a woman exhibiting cervical change. The study findings seemed to imply that when uterine contractions are detected via tocodynamometric monitoring but are not experienced by the patient, cerclage may be appropriate. In my opinion, this issue represents another open clinical question.
On at least one occasion, I have discovered that the patient scheduled for cerclage has not undergone preoperative screening for genital tract infections. In such a scenario, it is unclear whether it is best to cancel the procedure or to proceed. Berghella and colleagues concluded that evidence is insufficient to support routine, universal screening for genital tract infection prior to cerclage. In the absence of risk factors for infection, it may be appropriate to proceed with cerclage.
The issue of combined use of cerclage and 17α hydroxyprogesterone caproate also was addressed in this review. Berghella and colleagues recommended that progestin supplementation be continued if cerclage is subsequently performed. I, too, follow this approach, although the two interventions (progestin followed by cerclage) have not been studied in this sequence.
Related article: A stepwise approach to cervical cerclage (June 2012)
Data are limited, and conflicting, on the use of 17α hydroxyprogesterone caproate after cerclage. A small randomized trial suggested benefit,9 while a retrospective cohort study10 and a secondary analysis of data from a randomized trial of cerclage11 found no benefit for the addition of 17α hydroxyprogesterone caproate.
Berghella and colleagues advocated against reinforcing (second or repeat) cerclage, an intervention that may be associated with a higher incidence of preterm birth.12 Accordingly, continuing sonographic assessment of cervical length after cerclage may be futile. What this evidence means for practice
Increasing evidence suggests that cervical shortening is not limited to an innate or acquired cervical weakness but represents an early, asymptomatic phase on the pathway to preterm birth. Nevertheless, cerclage continues to be practiced widely and has been shown to be beneficial in selected populations.
We clinicians often face complex cases of presumed cervical insufficiency for which there are no simple or unequivocal recommendations. This appraisal by Berghella and colleagues will help us separate evidence-validated approaches from misconceptions in the technical aspects of cerclage.
--Alex C. Vidaeff, MD, MPH
We want to hear from you! Tell us what you think.
In contemporary medical practice, we expect our clinical actions to reflect the best and most current evidence. In many cases, however, the evidence available to us is weak or irrelevant. In their investigation, Berghella and colleagues set out to assess the quality of evidence in the setting of transvaginal cervical cerclage by reviewing the published data on selected perioperative strategies. They elected to perform a systematic review, as opposed to a narrative review (a simple opinion piece), because this approach follows an explicit process designed to limit bias and random error in the interpretation of scientific research.
The studies they analyzed vary from observational investigations to randomized trials, generating considerable heterogeneity in the data. Therefore, it would not have been feasible or appropriate for them to combine the results in a quantitative review (ie, meta-analysis). Their solution: to limit the analysis to a qualitative systematic review.
The term “systematic review” implies that investigators have an accurate and comprehensive understanding of existent data, with each study representing one contribution to a much larger body of knowledge. Over the years, Berghella and colleagues have contributed extensively to the literature on cervical cerclage and are well qualified to provide an analytic framework for the flood of published information on this practice. Although they focused primarily on how to perform cerclage, a discussion of when to perform cerclage cannot be separated from any consideration of efficacy.
When, exactly, is cerclage indicated?
The original indication for cerclage, established more than 50 years ago, required both a history of second-trimester loss and asymptomatic cervical changes in the current pregnancy. Since then, many cerclages have been performed on the basis of history alone or on current cervical changes regardless of history. However, the most recent professional guidelines reconfirm that any cerclage procedure should be supported by both historical and contemporaneous findings.1,2
Investigators have demonstrated that the measurement of cervical length by transvaginal ultrasound should generally be an integral part of clinical evaluation for asymptomatic cervical changes. Indeed, sonographic assessment has emerged as a tool capable of reducing “overcall” and unnecessary intervention.3 On the other hand, a meta-analysis of four randomized trials of ultrasound-indicated cerclage found it to be beneficial in women with a short cervix only if they also had a history of preterm delivery.4
In addition, randomized trials have documented a benefit for cerclage in two other clinical contexts:
- Results from a secondary analysis of data from a large randomized trial published in 1993 suggest that elective cerclage can be based on history alone in women with three or more second-trimester losses or preterm births.5
- Authors of a randomized trial published in 2003 suggested that women with advanced cervical changes, such as dilatation of the external os with exposure of the fetal membranes, may benefit from “emergency” cerclage even in the absence of a prior preterm delivery.6
How the data were analyzed
The data included in the review were analyzed separately, according to three widely accepted indications for cerclage:
- history-indicated: a history of three or more second-trimester losses and/or preterm births
- ultrasound-indicated: ultrasonographic detection of a cervical length of less than 25 mm, as measured by transvaginal ultrasound, in a woman with a history of second-trimester loss or preterm birth
- physical-examination–indicated: physical examination (manual or with a speculum) that confirms a dilated cervix.
Granted, this terminology can be confusing, as in the case of ultrasound-indicated cerclage, which includes aspects of the patient’s history. Moreover, I doubt that the studies included in this analysis always adhered to these definitions. The heterogeneity of the study population and the ambiguity of these definitions may limit the applicability of findings. In fact, they constitute the major (albeit practically unavoidable) limitation of this review.
The optimal approach to subclinical infection is unclear
Although there is a consensus that overt intra-amniotic infection is an absolute contraindication to cerclage, the implications of subclinical intra-amniotic infection in asymptomatic women are unclear. About 50% of women considered for emergency cerclage are likely to have intra-amniotic infection.7 An ongoing randomized trial is expected to elucidate the benefit of precerclage amniocentesis in such cases.
The sonographic detection of sludge in the amniotic fluid also has been associated with intra-amniotic infection. However, after analyzing the data, Berghella and colleagues did not find adequate justification for amniocentesis in this setting. A more practical question might be whether cerclage is advisable at all when sludge is present. Data from a recently reported abstract suggest that the presence of sludge increases the likelihood of early preterm birth independent of cervical length.8
Other gray areas
Another absolute contraindication to cerclage is the presence of painful uterine contractions in a woman exhibiting cervical change. The study findings seemed to imply that when uterine contractions are detected via tocodynamometric monitoring but are not experienced by the patient, cerclage may be appropriate. In my opinion, this issue represents another open clinical question.
On at least one occasion, I have discovered that the patient scheduled for cerclage has not undergone preoperative screening for genital tract infections. In such a scenario, it is unclear whether it is best to cancel the procedure or to proceed. Berghella and colleagues concluded that evidence is insufficient to support routine, universal screening for genital tract infection prior to cerclage. In the absence of risk factors for infection, it may be appropriate to proceed with cerclage.
The issue of combined use of cerclage and 17α hydroxyprogesterone caproate also was addressed in this review. Berghella and colleagues recommended that progestin supplementation be continued if cerclage is subsequently performed. I, too, follow this approach, although the two interventions (progestin followed by cerclage) have not been studied in this sequence.
Related article: A stepwise approach to cervical cerclage (June 2012)
Data are limited, and conflicting, on the use of 17α hydroxyprogesterone caproate after cerclage. A small randomized trial suggested benefit,9 while a retrospective cohort study10 and a secondary analysis of data from a randomized trial of cerclage11 found no benefit for the addition of 17α hydroxyprogesterone caproate.
Berghella and colleagues advocated against reinforcing (second or repeat) cerclage, an intervention that may be associated with a higher incidence of preterm birth.12 Accordingly, continuing sonographic assessment of cervical length after cerclage may be futile. What this evidence means for practice
Increasing evidence suggests that cervical shortening is not limited to an innate or acquired cervical weakness but represents an early, asymptomatic phase on the pathway to preterm birth. Nevertheless, cerclage continues to be practiced widely and has been shown to be beneficial in selected populations.
We clinicians often face complex cases of presumed cervical insufficiency for which there are no simple or unequivocal recommendations. This appraisal by Berghella and colleagues will help us separate evidence-validated approaches from misconceptions in the technical aspects of cerclage.
--Alex C. Vidaeff, MD, MPH
We want to hear from you! Tell us what you think.
- Royal College of Obstetricians and Gynaecologists. Cervical cerclage [guidelines]. http://www.rcog.org.uk/womens-health/clinical-guidance/cervical-cerclage-green-top-60. Accessed September 18, 2013.
- ACOG Practice Bulletin #130: Prediction and prevention of preterm birth. Obstet Gynecol. 2012;120:964–973.
- Grimes-Dennis J, Berghella V. Cervical length and prediction of preterm delivery. Curr Opin Obstet Gynecol. 2007;19(2):191–195.
- Berghella V, Odibo AO, To MS, et al. Cerclage for short cervix on ultrasound: meta-analysis of trials using individual patient-level data. Obstet Gynecol. 2005;106(1):181–189.
- Working Party on Cervical Cerclage. Final report of the Medical Research Council/Royal College of Obstetricians and Gynaecologists multicenter randomised trial of cervical cerclage. Br J Obstet Gynaecol. 1993;100(6):516–523.
- Althuisius SM, Dekker GA, Hummel P, van Geijin HP. Cervical incompetence prevention randomized cerclage trial: emergency cerclage with bed rest versus bed rest alone. Am J Obstet Gynecol. 2003;189(4):907–910.
- Romero R, Gonzalez R, Sepulveda W, et al. Infection and labor. VIII. Microbial invasion of the amniotic cavity in patients with suspected cervical incompetence: prevalence and clinical significance. Am J Obstet Gynecol. 1992(4 Pt 1);167:1086–1091.
- Saade G. Cervical funneling or intraamniotic debris and preterm birth in nulliparous women with short cervix. Am J Obstet Gynecol. 2013;208(1):S4.
- Yemini M, Borenstein R, Dreazen E, et al. Prevention of premature labor by 17 alpha-hydroxyprogesterone caproate. Am J Obstet Gynecol. 1985;151(5):574–577.
- Rebarber A, Cleary-Goldman J, Istwan NB, et al. The use of 17 alpha-hydroxyprogesterone caproate (17P) in women with cervical cerclage. Am J Perinatol. 2008;25(5):271–275.
- Berghella V, Figueroa D, Szychowski JM, et al. 17 alpha-hydroxyprogesterone caproate for the prevention of preterm birth in women with prior preterm birth and a short cervical length. Am J Obstet Gynecol. 2010;202(4):351.e1–e6.
- Baxter JK, Airoldi J, Berghella V. Short cervical length after history-indicated cerclage: is a reinforcing cerclage beneficial? Am J Obstet Gynecol. 2005;193(3 Pt 2):1204–1207.
- Royal College of Obstetricians and Gynaecologists. Cervical cerclage [guidelines]. http://www.rcog.org.uk/womens-health/clinical-guidance/cervical-cerclage-green-top-60. Accessed September 18, 2013.
- ACOG Practice Bulletin #130: Prediction and prevention of preterm birth. Obstet Gynecol. 2012;120:964–973.
- Grimes-Dennis J, Berghella V. Cervical length and prediction of preterm delivery. Curr Opin Obstet Gynecol. 2007;19(2):191–195.
- Berghella V, Odibo AO, To MS, et al. Cerclage for short cervix on ultrasound: meta-analysis of trials using individual patient-level data. Obstet Gynecol. 2005;106(1):181–189.
- Working Party on Cervical Cerclage. Final report of the Medical Research Council/Royal College of Obstetricians and Gynaecologists multicenter randomised trial of cervical cerclage. Br J Obstet Gynaecol. 1993;100(6):516–523.
- Althuisius SM, Dekker GA, Hummel P, van Geijin HP. Cervical incompetence prevention randomized cerclage trial: emergency cerclage with bed rest versus bed rest alone. Am J Obstet Gynecol. 2003;189(4):907–910.
- Romero R, Gonzalez R, Sepulveda W, et al. Infection and labor. VIII. Microbial invasion of the amniotic cavity in patients with suspected cervical incompetence: prevalence and clinical significance. Am J Obstet Gynecol. 1992(4 Pt 1);167:1086–1091.
- Saade G. Cervical funneling or intraamniotic debris and preterm birth in nulliparous women with short cervix. Am J Obstet Gynecol. 2013;208(1):S4.
- Yemini M, Borenstein R, Dreazen E, et al. Prevention of premature labor by 17 alpha-hydroxyprogesterone caproate. Am J Obstet Gynecol. 1985;151(5):574–577.
- Rebarber A, Cleary-Goldman J, Istwan NB, et al. The use of 17 alpha-hydroxyprogesterone caproate (17P) in women with cervical cerclage. Am J Perinatol. 2008;25(5):271–275.
- Berghella V, Figueroa D, Szychowski JM, et al. 17 alpha-hydroxyprogesterone caproate for the prevention of preterm birth in women with prior preterm birth and a short cervical length. Am J Obstet Gynecol. 2010;202(4):351.e1–e6.
- Baxter JK, Airoldi J, Berghella V. Short cervical length after history-indicated cerclage: is a reinforcing cerclage beneficial? Am J Obstet Gynecol. 2005;193(3 Pt 2):1204–1207.
A stepwise approach to managing eclampsia and other hypertensive emergencies
CASE: MISSED PREECLAMPSIA
At her first prenatal visit at 14 weeks’ gestation, a 41-year-old woman (G2P1) presents with a dichorionic twin gestation, blood pressure (BP) of 105/68 mm Hg, and a body mass index (BMI) of 40 kg/m2. The pregnancy was achieved through in vitro fertilization. Ten years earlier, the patient’s first pregnancy was complicated by preeclampsia, requiring preterm delivery at 33 weeks’ gestation.
By 28 weeks’ gestation, the patient has gained 26 lb. Her BP is 120/70 mm Hg, with no proteinuria detected by urine dipstick. By 30 weeks, she has gained an additional 8 lb, her BP is 142/84 mm Hg, and no proteinuria is detected. At 32 weeks, her BP is 140/92 mm Hg, she has gained another 8 lb, and no proteinuria is present. She also reports new-onset headaches that do not respond to over-the-counter analgesics. She is sent to the obstetric triage area for BP monitoring, blood testing for preeclampsia and nonstress test fetal monitoring.
During the 2-hour observation period, the patient continues to report headaches, and swelling of her face and hands is present. Her systolic BP values range from 132 to 152 mm Hg, and diastolic values range from 80 to 96 mm Hg. No proteinuria is detected, blood testing results for preeclampsia (complete blood count, liver enzymes, serum creatinine, and uric acid) are normal, and the nonstress tests are reactive in both fetuses.
The patient is given a diagnosis of gestational hypertension, along with a prescription for oral labetalol 200 mg daily and two tablets of acetaminophen with codeine for the headaches (to be taken every 6 hours as needed). She is sent home with instructions to return to her physician’s office in 1 week.
Two days later, she wakes in the middle of the night with a severe headache, blurred vision, and vomiting. Her husband calls the obstetrician’s answering service and is instructed to call 911 immediately. While waiting for an ambulance, the patient experiences a grand mal eclamptic convulsion. A second convulsion occurs during her transfer to the ED.
This scenario could have been avoided.
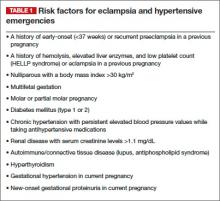
The obstetrician in this case was negligent for failing to recognize preeclampsia in a patient who had two clear risk factors for it: multifetal gestation and a history of early-onset (<37 weeks) preeclampsia in an earlier pregnancy (other risk factors are listed in TABLE 1).
As a result, the patient developed eclampsia, a serious condition that can lead to grave maternal complications (TABLE 2), including death. It also can cause fetal complications, including growth restriction, hypoxia, acidosis, preterm birth, long-term developmental deficits, and death.1,2
The obstetrician in this case also overlooked published evidence indicating that, in the setting of hypertension and headaches, as many as 20% to 30% of pregnant women whose tests for proteinuria show a negative or trace result via dipstick will develop eclampsia.3 Instead of initiating outpatient administration of oral antihypertensive agents, the obstetrician should have hospitalized this patient for at least 48 hours, with steroid administration, to determine whether outpatient management was feasible.
Related article: 10 practical, evidence-based recommendations to improve outcomes in women who have eclampsia Baha Sibai, MD (November 2011)
Defining eclampsia
Eclampsia is marked by the onset of convulsions (during pregnancy or postpartum) in association with gestational hypertension alone, proteinuria, preeclampsia, or superimposed preeclampsia. Although it is rare, eclampsia is potentially life-threatening. For that reason, obstetricians, anesthesiologists, ED physicians, neurologists, and critical-care physicians should be well versed in its diagnosis and management. In this article, I focus on management.
A few preliminary points
Eclampsia can develop any time during the antenatal period (>16 weeks’ gestation), during labor and delivery, and as long as 6 weeks after delivery. Therefore, we should be vigilant for preeclampsia whenever a pregnant patient visits our office, as well as when she makes unscheduled visits to the ED or obstetric triage area or is hospitalized.
Early recognition of women at high risk for preeclampsia and eclampsia may allow for prompt intervention, including early hospitalization for close observation prior to delivery and postpartum.1,2,4–10
Hospitalization of high-risk women allows for use of antihypertensive agents to treat severe BP, administration of magnesium sulfate to prevent convulsions, and timely delivery of the infant. It also allows for intensive maternal support during and after an eclamptic seizure.
Hospitalization is essential for women who exhibit features that suggest severe disease. More specifically, the presence of gestational hypertension with any of the following features is an indication for immediate hospitalization for evaluation and management:
- persistent severe hypertension (systolic
BP ≥160 mm Hg or diastolic BP ≥110 mm Hg) for at least 1 hour - gestational hypertension requiring oral antihypertensive therapy
- progressive and excessive weight gain (≥20 lb prior to 28 weeks’ gestation)
- generalized swelling (edema of hands or face)
- new-onset or persistent headaches despite analgesics
- persistent visual changes (blurred vision, scotomata, photophobia, double vision)
- shortness of breath, dyspnea, orthopnea, or tightness in the chest
- persistent retrosternal chest pain, severe epigastric or right upper quadrant pain
- persistent nausea, vomiting, malaise
- altered mental state, confusion, numbness, tingling, or motor weakness
- platelet count below 100 3 103 µL
- aspartate aminotransferase (AST), alanine aminotransferase (ALT), or lactic acid dehydrogenase (LDH) levels more than twice the upper limit of normal
- serum creatinine level >1.1 mg/dL
- suspected abruptio placentae.
A stepwise approach to eclampsia
Eclampsia is an obstetric emergency. Inadequate preparation for it or an inappropriate response to maternal and fetal conditions during and after an eclamptic convulsion can be detrimental to the mother and fetus. All obstetric units should have up-to-date protocols in place and should conduct mandatory drills to prepare nursing staff, obstetric providers, and anesthesia staff working in these units to manage eclampsia.
Step 1: Let the seizure run its course
During a seizure, resist the impulse to administer anticonvulsive drugs, including intravenous (IV) magnesium sulfate, because most eclamptic convulsions are self-limiting. Also abstain from administering medications such as IV phenytoin, diazepam, or midazolam, as these drugs are less effective than magnesium sulfate, and some can suppress the laryngeal reflex, increasing the risk of aspiration.
If the patient develops status epilepticus, initiate muscle paralysis and intubate her.
Step 2: Support the maternal condition
It is vital to support maternal respiratory and cardiovascular functions to prevent hypoxia, acidosis, and cardiorespiratory arrest.
Begin by establishing airway patency and maternal oxygenation during and after the convulsion. Administer oxygen via a face mask, with or without a reservoir, at a rate of 8 to 10 L/min.
During the apneic period (see “Profile of an eclamptic seizure” on page 46), the patient will develop hypoxia. Use pulse oximetry to monitor oxygen saturation, with the goal of keeping it above 94%. Arterial blood gas analysis is required if oxygen saturation remains below 92% or if pulmonary edema or aspiration is suspected.
If the patient develops recurrent seizures, status epilepticus, florid alveolar pulmonary edema, or respiratory arrest, intubate her immediately.
Step 3: Prevent maternal injury and aspiration
Secure the side rails of the patient’s bed by elevating them to prevent a fall, and make sure they are padded to prevent trauma during convulsions and afterward, when some women become combative and agitated. Position the patient in a lateral decubitus position to minimize aspiration of oral secretions. If any secretions or vomitus are present, remove them via suction.
Step 4: After the convulsion, give magnesium sulfate
Magnesium is the drug of choice for seizure prophylaxis in women with preeclampsia and severe symptoms, and to prevent recurrent seizures in women with eclampsia.
In the latter group, once the eclamptic convulsion has ended, give a loading dose of IV magnesium (6 g/100 mL over 20 minutes), followed by a continuous infusion of 2 g/h for at least 24 hours. If the patient develops a second seizure during the maintenance infusion, administer another bolus of magnesium (2 g/100 mL over 3–5 minutes).
Step 5: Treat severe hypertension
If severe hypertension persists for 60 minutes or longer, it can lead to injury of the brain, heart, and kidneys. To avoid these complications, it is essential to reduce BP to a safe range and maintain that level without compromising cerebral perfusion pressure and uteroplacental blood flow (which already may be reduced in some patients).
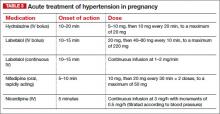
The goal of antihypertensive therapy is to keep systolic BP between 140 and 155 mm Hg and diastolic values between 90 and 105 mm Hg.9 Several agents are available for the treatment of severe hypertension during pregnancy and postpartum. The most commonly used IV medications for this purpose are labetalol and hydralazine. Another option is oral, rapidly acting
nifedipine.
Several randomized trials have compared efficacy and side effects between IV bolus injections of hydralazine; IV labetalol; and oral, rapidly acting nifedipine. In general, the findings of these studies suggest that either IV hydralazine or labetalol or oral nifedipine can be used to treat severe hypertension in pregnancy, as long as the provider is familiar with the dose to be used, the expected onset of action, and potential side effects (TABLE 3).
Women who develop generalized swelling or hemoconcentration (hematocrit ≥40%), or both, usually experience markedly reduced plasma volume. For this reason, these women will benefit from treatment with labetalol. If this is ineffective, then add IV hydralazine. However, delay administration of a rapidly acting vasodilator such as hydralazine to prevent an excessive hypotensive response and a secondary reduction in tissue perfusion and uteroplacental blood flow. Rather, administer a bolus infusion of 250 to 500 mL of isotonic saline before giving a vasodilator.
Additional details about the use of antihypertensive drugs are given in the section on other hypertensive emergencies below.
Step 6: Evaluate the patient for complications
Pulmonary edema can develop in patients with eclampsia or another hypertensive emergency. Suspect it if the patient has respiratory symptoms in association with tachypnea, tachycardia, or sustained oxygen saturation values below 93%, as well as when the patient exhibits basal rales during auscultation of the lungs. Treatment involves the administration of oxygen and IV furosemide (20–40 mg push), repeated as needed.
Some women with eclampsia may develop severe cerebral edema, hemorrhage, or both. The edema can be vasogenic or cytotoxic, leading to increased intracerebral pressure. Suspect edema or hemorrhage if the patient remains unresponsive, continues to experience convulsions despite therapy, or exhibits sensory or motor neurologic deficits. In such cases, neuroimaging is indicated, and the patient should be managed in consultation with neurology or neurosurgery.
Step 7: Begin the process of induction and delivery
Once the patient has been stabilized—and not before—initiate the induction process. Be aware that during and after the convulsion, changes in fetal heart rate (FHR) and uterine monitoring will usually be evident:
- prolonged deceleration or bradycardia (3–10 minutes)
- compensatory tachycardia, decreased beat-to-beat variability
- transient recurrent decelerations
- increased uterine tone and greater frequency of uterine activity.
These changes in FHR and uterine activity usually last 3 to 15 minutes. For this reason, it is important to avoid rushing the patient for cesarean delivery, as FHR and uterine activity are likely to return to normal after maternal resuscitation and stabilization. If not, consider other causes, such as abruptio placentae.
Eclampsia itself is not an indication for cesarean delivery. The selection of mode of delivery should be based on the presence or absence of labor, the cervical Bishop score, fetal gestational age, fetal presentation, and overall fetal condition.
Choosing an anesthetic
Regional analgesia/anesthesia is the method of choice for most women with eclampsia. However, regional anesthesia is to be avoided in the presence of disseminated intravascular coagulation or thrombocytopenia (the threshold platelet count is usually less than 75 x 103 µL. In such a case, IV analgesia can be used during labor, and general anesthesia may be appropriate for cesarean delivery. Both spinal and epidural analgesia and anesthesia are appropriate for women with eclampsia.
How to manage other hypertensive emergencies
A hypertensive emergency during pregnancy or postpartum involves acute-onset, persistent (>15 minutes), severe systolic BP (≥160 mm Hg) or severe diastolic BP (≥110 mm Hg), or both. The first step in such an emergency is to ensure the accurate measurement of BP using standard techniques.
Patients with acute-onset, persistent, severe BP should be hospitalized promptly for evaluation and treatment to prevent organ damage. Once such a patient is hospitalized, BP should be recorded every 15 minutes, with continuous FHR monitoring to ensure fetal viability.
Related article: Failure to diagnose preeclampsia and more (Medical Verdicts, February 2013)
The timing of initiation of antihypertensive medications, as well as determination of the type of medication best suited for the patient, should be based on:
- systolic and diastolic BP levels
- maternal clinical and laboratory findings
- presence of associated symptoms
- preexisting medical comorbidities
- whether the patient is antepartum or postpartum.
For example, a sustained BP level of 200/120 mm Hg requires therapy after 15 minutes, whereas observation may be suitable for as long as 60 minutes for a sustained BP of 160/72 mm Hg during labor.
Rapid reduction of systolic BP can lead to marked reductions in uteroplacental blood flow and a nonreassuring FHR tracing. Moreover, a rapid reduction of severe systolic BP in patients who have constricted plasma volume can reduce perfusion to the kidney, brain, and placenta. However, sustained BP of 165/100 mm Hg in association with central nervous system signs or symptoms, congestive heart failure, thrombocytopenia, or postpartum status requires therapy within 1 hour.
In general, it is difficult to obtain accurate BP recordings using noninvasive electronic instruments during labor because of the effects of labor on systolic BP and the lack of standardized methods for positioning of the arm cuff and the patient.
For these reasons, the decision about when to start acute antihypertensive therapy, based on systolic or diastolic BP, or both, should be individualized. And the choice of antihypertensive agent should be based on maternal clinical findings.
Choosing an antihypertensive agent
Because both hydralazine and nifedipine are associated with tachycardia, avoid them in patients with a heart rate above 110 bpm, using labetalol instead.10
In patients with bradycardia (heart rate <60 bpm), asthma, or congestive heart failure, however, labetalol should be avoided. In these populations, hydralazine or nifedipine is the drug of choice. Nifedipine is associated with improved renal blood flow and a resultant increase in urine output, making it preferable for patients with decreased urine output or severe postpartum hypertension.10
One theoretical concern is that the combined use of nifedipine and magnesium sulfate can cause excessive hypotension and neuromuscular blockage. As a result, some experts recommend that nifedipine be avoided in patients receiving magnesium sulfate. However, a recent review of this subject concluded that combined use of these drugs does not increase the risks of excessive hypotension and neuromuscular blockage in patients with severe hypertension or preeclampsia.
The initial dose of labetalol, when it is your chosen agent, is 20 mg IV, with BP measured 10 minutes later. If the target BP threshold is not achieved, administer 40 mg, 80 mg, and 80 mg at 10-minute intervals, as needed, again measuring BP 10 minutes after every dose. If, after a maximum dose of 240 mg, the desired BP threshold still has not been reached, give 5 to 10 mg IV hydralazine and measure BP 20 minutes later. If the target BP threshold still has not been achieved, it is essential to obtain consultation on the need for continuous infusion of labetalol, nicardipine, or sodium nitroprusside.
The initial dose of hydralazine, when it is your chosen agent, is 5 to 10 mg IV, with BP measured 20 minutes later. If needed, give another 10 mg and measure BP after another 20-minute interval. After a maximum dose of hydralazine 20 mg, switch to IV labetalol, using the regimen described above for labetalol, if the BP threshold still has not been achieved.
Nitroglycerin may be helpful in carefully selected patients
This drug is an arterial—but mostly venous—dilator. It is administered via IV infusion at an initial rate of 5 µg/min, with the rate gradually increased every 3 to 5 minutes (titrated to BP) to a maximum dose of 100 µg/min. It is the drug of choice in any hypertensive emergency associated with pulmonary edema and for control of hypertension associated with tracheal manipulation during intubation and extubation with general anesthesia.
Nitroglycerin is contraindicated in hypertensive encephalopathy because it increases cerebral blood flow and intracranial pressure. This drug should be administered only under the supervision of an experienced obstetric intensivist.
Sodium nitroprusside: Only in an ICU
This agent causes arterial and venous relaxation by interfering with the influx and intracellular activation of calcium. It is the drug of choice in hypertensive encephalopathy because it controls both afterload (vascular resistance) and preload (fluid status). It should be used only in the setting of intensive care.
The recommended dose is IV infusion at a rate of 0.25 to 5.00 µg/kg/min. Sodium nitroprusside has an immediate onset of action and may continue to exert an effect 3 to 5 minutes after discontinuation. Any hypotension caused by the drug should subside within minutes after discontinuation of the drip, due to the drug’s short half-life.
Nitroprusside is metabolized into thiocyanate and excreted in the urine. Cyanide can accumulate with large doses (>10 µg/kg/min) or prolonged administration (>48 hours), or if the patient has renal insufficiency or decreased hepatic metabolism. Signs of toxicity include anorexia, disorientation, headache, fatigue, restlessness, tinnitus, delirium, hallucinations, nausea, vomiting, and metabolic acidosis. When infused at a rate of less than 2 µg/kg/min, however, cyanide toxicity is unlikely.
As is the case with nitroglycerin, this drug should be administered only under the supervision of an experienced obstetric intensivist.
Case: Resolved
Upon arrival at the ED, the patient exhibits shallow, rapid breathing and foaming from the mouth. She is placed in a lateral decubitus position, an oral airway is established, and all secretions are suctioned. Oxygen is administered via face mask at a rate of 8 L/min. Her initial oxygen saturation level is 92%. IV access is secured, and a loading dose of magnesium sulfate 6 g is given over 20 minutes. Oxygen saturation increases to 94% to 96%. Auscultation of both lungs is normal.
The patient remains in a postictal state for about 15 minutes, but then orients to name, place, and time. FHR monitoring of both fetuses reveals a normal baseline with moderate variability, as well as variable decelerations in the presenting twin.
A maintenance dose of magnesium sulfate is initiated at a rate of 2 g/h, with the BP level recorded every 15 minutes. Systolic values remain between 170 and 180 mm Hg, and diastolic values between 108 and 112 mm Hg for 60 minutes. The obstetrician administers IV labetalol (20 mg) over 2 minutes. About 15 minutes later, the BP level is 154/100 mm Hg, with values remaining in the range of 150 to 156 mm Hg systolic and 92 to 104 mm Hg diastolic.
Ultrasonography reveals that the presenting twin is in a breech position, with estimated fetal weight below the 10th percentile and oligohydramnios. As a result, the obstetrician elects to proceed to cesarean delivery. The twins are delivered by cesarean section using spinal anesthesia. Although the infants are premature, there are no complications.Profile of an eclamptic seizure
Witnessing an eclamptic convulsion can be a frightening experience for nurses and medical providers. The convulsion usually lasts 60 to 90 seconds and occurs in two phases:
- Phase 1 (15–25 seconds) involves facial twitching, rolling of the eyes, and stiffening of the body, with generalized muscular contractions.
- Phase 2 (20–50 seconds) involves alternate contraction and relaxation of the muscles of the body in rapid succession, starting in the face and spreading throughout the body. Foaming at the mouth also occurs, and the patient may bite her tongue if it isn’t protected.
Apnea develops during and immediately after the convulsion, lasting about 120 seconds. A period of hyperventilation follows to compensate for the respiratory acidosis during the apneic period.
A postictal state follows the convulsion, and the patient usually remembers nothing of the episode. Some patients also become restless, combative, and agitated, requiring sedation. Aspiration is possible during or after the convulsion.
We want to hear from you! Tell us what you think.
- Sibai BM. Diagnosis, prevention, and management of eclampsia. Obstet Gynecol. 2005;105(2):402–410.
- Sibai BM. Diagnosis and management of gestational hypertension and preeclampsia. Obstet Gynecol. 2003;102(1):182–192.
- Meyer NL, Mercer BM, Friedman SA, Sibai BM. Urinary dipstick protein: a poor predictor of absent or severe proteinuria. Am J Obstet Gynecol. 1994;170(1 Pt 1):137–141.
- Knight M; UK Obstetric Surveillance System (UKOSS). Eclampsia in the United Kingdom 2005. BJOG. 2007;114(9):1072–1078.
- ACOG Practice Bulletin #33: Diagnosis and management of preeclampsia and eclampsia. Obstet Gynecol. 2002;99:159–167.
- Sibai BM, Stella CL. Diagnosis and management of atypical preeclampsia–eclampsia. Am J Obstet Gynecol. 2009;200(5):481.e1–e7.
- Sibai BM. Etiology and management of postpartum hypertension-preeclampsia. Am J Obstet Gynecol. 2012;206(6):470–475.
- ACOG Committee Opinion #514: Emergent therapy for acute-onset, severe hypertension with preeclampsia or eclampsia. Obstet Gynecol. 2011;118:1465–1468.
- Liu S, Joseph KS, Liston RM, et al. Incidence, risk factors, and associated complications of eclampsia. Obstet Gynecol. 2011;118(5):987–994.
- Raheem IA, Saaid R, Omar Sz, Tan PC. Oral nifedipine versus intravenous labetalol for acute blood pressure control in hypertensive emergencies of pregnancy: a randomized trial. BJOG. 2012;119(1):78–85.
CASE: MISSED PREECLAMPSIA
At her first prenatal visit at 14 weeks’ gestation, a 41-year-old woman (G2P1) presents with a dichorionic twin gestation, blood pressure (BP) of 105/68 mm Hg, and a body mass index (BMI) of 40 kg/m2. The pregnancy was achieved through in vitro fertilization. Ten years earlier, the patient’s first pregnancy was complicated by preeclampsia, requiring preterm delivery at 33 weeks’ gestation.
By 28 weeks’ gestation, the patient has gained 26 lb. Her BP is 120/70 mm Hg, with no proteinuria detected by urine dipstick. By 30 weeks, she has gained an additional 8 lb, her BP is 142/84 mm Hg, and no proteinuria is detected. At 32 weeks, her BP is 140/92 mm Hg, she has gained another 8 lb, and no proteinuria is present. She also reports new-onset headaches that do not respond to over-the-counter analgesics. She is sent to the obstetric triage area for BP monitoring, blood testing for preeclampsia and nonstress test fetal monitoring.
During the 2-hour observation period, the patient continues to report headaches, and swelling of her face and hands is present. Her systolic BP values range from 132 to 152 mm Hg, and diastolic values range from 80 to 96 mm Hg. No proteinuria is detected, blood testing results for preeclampsia (complete blood count, liver enzymes, serum creatinine, and uric acid) are normal, and the nonstress tests are reactive in both fetuses.
The patient is given a diagnosis of gestational hypertension, along with a prescription for oral labetalol 200 mg daily and two tablets of acetaminophen with codeine for the headaches (to be taken every 6 hours as needed). She is sent home with instructions to return to her physician’s office in 1 week.
Two days later, she wakes in the middle of the night with a severe headache, blurred vision, and vomiting. Her husband calls the obstetrician’s answering service and is instructed to call 911 immediately. While waiting for an ambulance, the patient experiences a grand mal eclamptic convulsion. A second convulsion occurs during her transfer to the ED.
This scenario could have been avoided.
The obstetrician in this case was negligent for failing to recognize preeclampsia in a patient who had two clear risk factors for it: multifetal gestation and a history of early-onset (<37 weeks) preeclampsia in an earlier pregnancy (other risk factors are listed in TABLE 1).
As a result, the patient developed eclampsia, a serious condition that can lead to grave maternal complications (TABLE 2), including death. It also can cause fetal complications, including growth restriction, hypoxia, acidosis, preterm birth, long-term developmental deficits, and death.1,2
The obstetrician in this case also overlooked published evidence indicating that, in the setting of hypertension and headaches, as many as 20% to 30% of pregnant women whose tests for proteinuria show a negative or trace result via dipstick will develop eclampsia.3 Instead of initiating outpatient administration of oral antihypertensive agents, the obstetrician should have hospitalized this patient for at least 48 hours, with steroid administration, to determine whether outpatient management was feasible.
Related article: 10 practical, evidence-based recommendations to improve outcomes in women who have eclampsia Baha Sibai, MD (November 2011)
Defining eclampsia
Eclampsia is marked by the onset of convulsions (during pregnancy or postpartum) in association with gestational hypertension alone, proteinuria, preeclampsia, or superimposed preeclampsia. Although it is rare, eclampsia is potentially life-threatening. For that reason, obstetricians, anesthesiologists, ED physicians, neurologists, and critical-care physicians should be well versed in its diagnosis and management. In this article, I focus on management.
A few preliminary points
Eclampsia can develop any time during the antenatal period (>16 weeks’ gestation), during labor and delivery, and as long as 6 weeks after delivery. Therefore, we should be vigilant for preeclampsia whenever a pregnant patient visits our office, as well as when she makes unscheduled visits to the ED or obstetric triage area or is hospitalized.
Early recognition of women at high risk for preeclampsia and eclampsia may allow for prompt intervention, including early hospitalization for close observation prior to delivery and postpartum.1,2,4–10
Hospitalization of high-risk women allows for use of antihypertensive agents to treat severe BP, administration of magnesium sulfate to prevent convulsions, and timely delivery of the infant. It also allows for intensive maternal support during and after an eclamptic seizure.
Hospitalization is essential for women who exhibit features that suggest severe disease. More specifically, the presence of gestational hypertension with any of the following features is an indication for immediate hospitalization for evaluation and management:
- persistent severe hypertension (systolic
BP ≥160 mm Hg or diastolic BP ≥110 mm Hg) for at least 1 hour - gestational hypertension requiring oral antihypertensive therapy
- progressive and excessive weight gain (≥20 lb prior to 28 weeks’ gestation)
- generalized swelling (edema of hands or face)
- new-onset or persistent headaches despite analgesics
- persistent visual changes (blurred vision, scotomata, photophobia, double vision)
- shortness of breath, dyspnea, orthopnea, or tightness in the chest
- persistent retrosternal chest pain, severe epigastric or right upper quadrant pain
- persistent nausea, vomiting, malaise
- altered mental state, confusion, numbness, tingling, or motor weakness
- platelet count below 100 3 103 µL
- aspartate aminotransferase (AST), alanine aminotransferase (ALT), or lactic acid dehydrogenase (LDH) levels more than twice the upper limit of normal
- serum creatinine level >1.1 mg/dL
- suspected abruptio placentae.
A stepwise approach to eclampsia
Eclampsia is an obstetric emergency. Inadequate preparation for it or an inappropriate response to maternal and fetal conditions during and after an eclamptic convulsion can be detrimental to the mother and fetus. All obstetric units should have up-to-date protocols in place and should conduct mandatory drills to prepare nursing staff, obstetric providers, and anesthesia staff working in these units to manage eclampsia.
Step 1: Let the seizure run its course
During a seizure, resist the impulse to administer anticonvulsive drugs, including intravenous (IV) magnesium sulfate, because most eclamptic convulsions are self-limiting. Also abstain from administering medications such as IV phenytoin, diazepam, or midazolam, as these drugs are less effective than magnesium sulfate, and some can suppress the laryngeal reflex, increasing the risk of aspiration.
If the patient develops status epilepticus, initiate muscle paralysis and intubate her.
Step 2: Support the maternal condition
It is vital to support maternal respiratory and cardiovascular functions to prevent hypoxia, acidosis, and cardiorespiratory arrest.
Begin by establishing airway patency and maternal oxygenation during and after the convulsion. Administer oxygen via a face mask, with or without a reservoir, at a rate of 8 to 10 L/min.
During the apneic period (see “Profile of an eclamptic seizure” on page 46), the patient will develop hypoxia. Use pulse oximetry to monitor oxygen saturation, with the goal of keeping it above 94%. Arterial blood gas analysis is required if oxygen saturation remains below 92% or if pulmonary edema or aspiration is suspected.
If the patient develops recurrent seizures, status epilepticus, florid alveolar pulmonary edema, or respiratory arrest, intubate her immediately.
Step 3: Prevent maternal injury and aspiration
Secure the side rails of the patient’s bed by elevating them to prevent a fall, and make sure they are padded to prevent trauma during convulsions and afterward, when some women become combative and agitated. Position the patient in a lateral decubitus position to minimize aspiration of oral secretions. If any secretions or vomitus are present, remove them via suction.
Step 4: After the convulsion, give magnesium sulfate
Magnesium is the drug of choice for seizure prophylaxis in women with preeclampsia and severe symptoms, and to prevent recurrent seizures in women with eclampsia.
In the latter group, once the eclamptic convulsion has ended, give a loading dose of IV magnesium (6 g/100 mL over 20 minutes), followed by a continuous infusion of 2 g/h for at least 24 hours. If the patient develops a second seizure during the maintenance infusion, administer another bolus of magnesium (2 g/100 mL over 3–5 minutes).
Step 5: Treat severe hypertension
If severe hypertension persists for 60 minutes or longer, it can lead to injury of the brain, heart, and kidneys. To avoid these complications, it is essential to reduce BP to a safe range and maintain that level without compromising cerebral perfusion pressure and uteroplacental blood flow (which already may be reduced in some patients).
The goal of antihypertensive therapy is to keep systolic BP between 140 and 155 mm Hg and diastolic values between 90 and 105 mm Hg.9 Several agents are available for the treatment of severe hypertension during pregnancy and postpartum. The most commonly used IV medications for this purpose are labetalol and hydralazine. Another option is oral, rapidly acting
nifedipine.
Several randomized trials have compared efficacy and side effects between IV bolus injections of hydralazine; IV labetalol; and oral, rapidly acting nifedipine. In general, the findings of these studies suggest that either IV hydralazine or labetalol or oral nifedipine can be used to treat severe hypertension in pregnancy, as long as the provider is familiar with the dose to be used, the expected onset of action, and potential side effects (TABLE 3).
Women who develop generalized swelling or hemoconcentration (hematocrit ≥40%), or both, usually experience markedly reduced plasma volume. For this reason, these women will benefit from treatment with labetalol. If this is ineffective, then add IV hydralazine. However, delay administration of a rapidly acting vasodilator such as hydralazine to prevent an excessive hypotensive response and a secondary reduction in tissue perfusion and uteroplacental blood flow. Rather, administer a bolus infusion of 250 to 500 mL of isotonic saline before giving a vasodilator.
Additional details about the use of antihypertensive drugs are given in the section on other hypertensive emergencies below.
Step 6: Evaluate the patient for complications
Pulmonary edema can develop in patients with eclampsia or another hypertensive emergency. Suspect it if the patient has respiratory symptoms in association with tachypnea, tachycardia, or sustained oxygen saturation values below 93%, as well as when the patient exhibits basal rales during auscultation of the lungs. Treatment involves the administration of oxygen and IV furosemide (20–40 mg push), repeated as needed.
Some women with eclampsia may develop severe cerebral edema, hemorrhage, or both. The edema can be vasogenic or cytotoxic, leading to increased intracerebral pressure. Suspect edema or hemorrhage if the patient remains unresponsive, continues to experience convulsions despite therapy, or exhibits sensory or motor neurologic deficits. In such cases, neuroimaging is indicated, and the patient should be managed in consultation with neurology or neurosurgery.
Step 7: Begin the process of induction and delivery
Once the patient has been stabilized—and not before—initiate the induction process. Be aware that during and after the convulsion, changes in fetal heart rate (FHR) and uterine monitoring will usually be evident:
- prolonged deceleration or bradycardia (3–10 minutes)
- compensatory tachycardia, decreased beat-to-beat variability
- transient recurrent decelerations
- increased uterine tone and greater frequency of uterine activity.
These changes in FHR and uterine activity usually last 3 to 15 minutes. For this reason, it is important to avoid rushing the patient for cesarean delivery, as FHR and uterine activity are likely to return to normal after maternal resuscitation and stabilization. If not, consider other causes, such as abruptio placentae.
Eclampsia itself is not an indication for cesarean delivery. The selection of mode of delivery should be based on the presence or absence of labor, the cervical Bishop score, fetal gestational age, fetal presentation, and overall fetal condition.
Choosing an anesthetic
Regional analgesia/anesthesia is the method of choice for most women with eclampsia. However, regional anesthesia is to be avoided in the presence of disseminated intravascular coagulation or thrombocytopenia (the threshold platelet count is usually less than 75 x 103 µL. In such a case, IV analgesia can be used during labor, and general anesthesia may be appropriate for cesarean delivery. Both spinal and epidural analgesia and anesthesia are appropriate for women with eclampsia.
How to manage other hypertensive emergencies
A hypertensive emergency during pregnancy or postpartum involves acute-onset, persistent (>15 minutes), severe systolic BP (≥160 mm Hg) or severe diastolic BP (≥110 mm Hg), or both. The first step in such an emergency is to ensure the accurate measurement of BP using standard techniques.
Patients with acute-onset, persistent, severe BP should be hospitalized promptly for evaluation and treatment to prevent organ damage. Once such a patient is hospitalized, BP should be recorded every 15 minutes, with continuous FHR monitoring to ensure fetal viability.
Related article: Failure to diagnose preeclampsia and more (Medical Verdicts, February 2013)
The timing of initiation of antihypertensive medications, as well as determination of the type of medication best suited for the patient, should be based on:
- systolic and diastolic BP levels
- maternal clinical and laboratory findings
- presence of associated symptoms
- preexisting medical comorbidities
- whether the patient is antepartum or postpartum.
For example, a sustained BP level of 200/120 mm Hg requires therapy after 15 minutes, whereas observation may be suitable for as long as 60 minutes for a sustained BP of 160/72 mm Hg during labor.
Rapid reduction of systolic BP can lead to marked reductions in uteroplacental blood flow and a nonreassuring FHR tracing. Moreover, a rapid reduction of severe systolic BP in patients who have constricted plasma volume can reduce perfusion to the kidney, brain, and placenta. However, sustained BP of 165/100 mm Hg in association with central nervous system signs or symptoms, congestive heart failure, thrombocytopenia, or postpartum status requires therapy within 1 hour.
In general, it is difficult to obtain accurate BP recordings using noninvasive electronic instruments during labor because of the effects of labor on systolic BP and the lack of standardized methods for positioning of the arm cuff and the patient.
For these reasons, the decision about when to start acute antihypertensive therapy, based on systolic or diastolic BP, or both, should be individualized. And the choice of antihypertensive agent should be based on maternal clinical findings.
Choosing an antihypertensive agent
Because both hydralazine and nifedipine are associated with tachycardia, avoid them in patients with a heart rate above 110 bpm, using labetalol instead.10
In patients with bradycardia (heart rate <60 bpm), asthma, or congestive heart failure, however, labetalol should be avoided. In these populations, hydralazine or nifedipine is the drug of choice. Nifedipine is associated with improved renal blood flow and a resultant increase in urine output, making it preferable for patients with decreased urine output or severe postpartum hypertension.10
One theoretical concern is that the combined use of nifedipine and magnesium sulfate can cause excessive hypotension and neuromuscular blockage. As a result, some experts recommend that nifedipine be avoided in patients receiving magnesium sulfate. However, a recent review of this subject concluded that combined use of these drugs does not increase the risks of excessive hypotension and neuromuscular blockage in patients with severe hypertension or preeclampsia.
The initial dose of labetalol, when it is your chosen agent, is 20 mg IV, with BP measured 10 minutes later. If the target BP threshold is not achieved, administer 40 mg, 80 mg, and 80 mg at 10-minute intervals, as needed, again measuring BP 10 minutes after every dose. If, after a maximum dose of 240 mg, the desired BP threshold still has not been reached, give 5 to 10 mg IV hydralazine and measure BP 20 minutes later. If the target BP threshold still has not been achieved, it is essential to obtain consultation on the need for continuous infusion of labetalol, nicardipine, or sodium nitroprusside.
The initial dose of hydralazine, when it is your chosen agent, is 5 to 10 mg IV, with BP measured 20 minutes later. If needed, give another 10 mg and measure BP after another 20-minute interval. After a maximum dose of hydralazine 20 mg, switch to IV labetalol, using the regimen described above for labetalol, if the BP threshold still has not been achieved.
Nitroglycerin may be helpful in carefully selected patients
This drug is an arterial—but mostly venous—dilator. It is administered via IV infusion at an initial rate of 5 µg/min, with the rate gradually increased every 3 to 5 minutes (titrated to BP) to a maximum dose of 100 µg/min. It is the drug of choice in any hypertensive emergency associated with pulmonary edema and for control of hypertension associated with tracheal manipulation during intubation and extubation with general anesthesia.
Nitroglycerin is contraindicated in hypertensive encephalopathy because it increases cerebral blood flow and intracranial pressure. This drug should be administered only under the supervision of an experienced obstetric intensivist.
Sodium nitroprusside: Only in an ICU
This agent causes arterial and venous relaxation by interfering with the influx and intracellular activation of calcium. It is the drug of choice in hypertensive encephalopathy because it controls both afterload (vascular resistance) and preload (fluid status). It should be used only in the setting of intensive care.
The recommended dose is IV infusion at a rate of 0.25 to 5.00 µg/kg/min. Sodium nitroprusside has an immediate onset of action and may continue to exert an effect 3 to 5 minutes after discontinuation. Any hypotension caused by the drug should subside within minutes after discontinuation of the drip, due to the drug’s short half-life.
Nitroprusside is metabolized into thiocyanate and excreted in the urine. Cyanide can accumulate with large doses (>10 µg/kg/min) or prolonged administration (>48 hours), or if the patient has renal insufficiency or decreased hepatic metabolism. Signs of toxicity include anorexia, disorientation, headache, fatigue, restlessness, tinnitus, delirium, hallucinations, nausea, vomiting, and metabolic acidosis. When infused at a rate of less than 2 µg/kg/min, however, cyanide toxicity is unlikely.
As is the case with nitroglycerin, this drug should be administered only under the supervision of an experienced obstetric intensivist.
Case: Resolved
Upon arrival at the ED, the patient exhibits shallow, rapid breathing and foaming from the mouth. She is placed in a lateral decubitus position, an oral airway is established, and all secretions are suctioned. Oxygen is administered via face mask at a rate of 8 L/min. Her initial oxygen saturation level is 92%. IV access is secured, and a loading dose of magnesium sulfate 6 g is given over 20 minutes. Oxygen saturation increases to 94% to 96%. Auscultation of both lungs is normal.
The patient remains in a postictal state for about 15 minutes, but then orients to name, place, and time. FHR monitoring of both fetuses reveals a normal baseline with moderate variability, as well as variable decelerations in the presenting twin.
A maintenance dose of magnesium sulfate is initiated at a rate of 2 g/h, with the BP level recorded every 15 minutes. Systolic values remain between 170 and 180 mm Hg, and diastolic values between 108 and 112 mm Hg for 60 minutes. The obstetrician administers IV labetalol (20 mg) over 2 minutes. About 15 minutes later, the BP level is 154/100 mm Hg, with values remaining in the range of 150 to 156 mm Hg systolic and 92 to 104 mm Hg diastolic.
Ultrasonography reveals that the presenting twin is in a breech position, with estimated fetal weight below the 10th percentile and oligohydramnios. As a result, the obstetrician elects to proceed to cesarean delivery. The twins are delivered by cesarean section using spinal anesthesia. Although the infants are premature, there are no complications.Profile of an eclamptic seizure
Witnessing an eclamptic convulsion can be a frightening experience for nurses and medical providers. The convulsion usually lasts 60 to 90 seconds and occurs in two phases:
- Phase 1 (15–25 seconds) involves facial twitching, rolling of the eyes, and stiffening of the body, with generalized muscular contractions.
- Phase 2 (20–50 seconds) involves alternate contraction and relaxation of the muscles of the body in rapid succession, starting in the face and spreading throughout the body. Foaming at the mouth also occurs, and the patient may bite her tongue if it isn’t protected.
Apnea develops during and immediately after the convulsion, lasting about 120 seconds. A period of hyperventilation follows to compensate for the respiratory acidosis during the apneic period.
A postictal state follows the convulsion, and the patient usually remembers nothing of the episode. Some patients also become restless, combative, and agitated, requiring sedation. Aspiration is possible during or after the convulsion.
We want to hear from you! Tell us what you think.
CASE: MISSED PREECLAMPSIA
At her first prenatal visit at 14 weeks’ gestation, a 41-year-old woman (G2P1) presents with a dichorionic twin gestation, blood pressure (BP) of 105/68 mm Hg, and a body mass index (BMI) of 40 kg/m2. The pregnancy was achieved through in vitro fertilization. Ten years earlier, the patient’s first pregnancy was complicated by preeclampsia, requiring preterm delivery at 33 weeks’ gestation.
By 28 weeks’ gestation, the patient has gained 26 lb. Her BP is 120/70 mm Hg, with no proteinuria detected by urine dipstick. By 30 weeks, she has gained an additional 8 lb, her BP is 142/84 mm Hg, and no proteinuria is detected. At 32 weeks, her BP is 140/92 mm Hg, she has gained another 8 lb, and no proteinuria is present. She also reports new-onset headaches that do not respond to over-the-counter analgesics. She is sent to the obstetric triage area for BP monitoring, blood testing for preeclampsia and nonstress test fetal monitoring.
During the 2-hour observation period, the patient continues to report headaches, and swelling of her face and hands is present. Her systolic BP values range from 132 to 152 mm Hg, and diastolic values range from 80 to 96 mm Hg. No proteinuria is detected, blood testing results for preeclampsia (complete blood count, liver enzymes, serum creatinine, and uric acid) are normal, and the nonstress tests are reactive in both fetuses.
The patient is given a diagnosis of gestational hypertension, along with a prescription for oral labetalol 200 mg daily and two tablets of acetaminophen with codeine for the headaches (to be taken every 6 hours as needed). She is sent home with instructions to return to her physician’s office in 1 week.
Two days later, she wakes in the middle of the night with a severe headache, blurred vision, and vomiting. Her husband calls the obstetrician’s answering service and is instructed to call 911 immediately. While waiting for an ambulance, the patient experiences a grand mal eclamptic convulsion. A second convulsion occurs during her transfer to the ED.
This scenario could have been avoided.
The obstetrician in this case was negligent for failing to recognize preeclampsia in a patient who had two clear risk factors for it: multifetal gestation and a history of early-onset (<37 weeks) preeclampsia in an earlier pregnancy (other risk factors are listed in TABLE 1).
As a result, the patient developed eclampsia, a serious condition that can lead to grave maternal complications (TABLE 2), including death. It also can cause fetal complications, including growth restriction, hypoxia, acidosis, preterm birth, long-term developmental deficits, and death.1,2
The obstetrician in this case also overlooked published evidence indicating that, in the setting of hypertension and headaches, as many as 20% to 30% of pregnant women whose tests for proteinuria show a negative or trace result via dipstick will develop eclampsia.3 Instead of initiating outpatient administration of oral antihypertensive agents, the obstetrician should have hospitalized this patient for at least 48 hours, with steroid administration, to determine whether outpatient management was feasible.
Related article: 10 practical, evidence-based recommendations to improve outcomes in women who have eclampsia Baha Sibai, MD (November 2011)
Defining eclampsia
Eclampsia is marked by the onset of convulsions (during pregnancy or postpartum) in association with gestational hypertension alone, proteinuria, preeclampsia, or superimposed preeclampsia. Although it is rare, eclampsia is potentially life-threatening. For that reason, obstetricians, anesthesiologists, ED physicians, neurologists, and critical-care physicians should be well versed in its diagnosis and management. In this article, I focus on management.
A few preliminary points
Eclampsia can develop any time during the antenatal period (>16 weeks’ gestation), during labor and delivery, and as long as 6 weeks after delivery. Therefore, we should be vigilant for preeclampsia whenever a pregnant patient visits our office, as well as when she makes unscheduled visits to the ED or obstetric triage area or is hospitalized.
Early recognition of women at high risk for preeclampsia and eclampsia may allow for prompt intervention, including early hospitalization for close observation prior to delivery and postpartum.1,2,4–10
Hospitalization of high-risk women allows for use of antihypertensive agents to treat severe BP, administration of magnesium sulfate to prevent convulsions, and timely delivery of the infant. It also allows for intensive maternal support during and after an eclamptic seizure.
Hospitalization is essential for women who exhibit features that suggest severe disease. More specifically, the presence of gestational hypertension with any of the following features is an indication for immediate hospitalization for evaluation and management:
- persistent severe hypertension (systolic
BP ≥160 mm Hg or diastolic BP ≥110 mm Hg) for at least 1 hour - gestational hypertension requiring oral antihypertensive therapy
- progressive and excessive weight gain (≥20 lb prior to 28 weeks’ gestation)
- generalized swelling (edema of hands or face)
- new-onset or persistent headaches despite analgesics
- persistent visual changes (blurred vision, scotomata, photophobia, double vision)
- shortness of breath, dyspnea, orthopnea, or tightness in the chest
- persistent retrosternal chest pain, severe epigastric or right upper quadrant pain
- persistent nausea, vomiting, malaise
- altered mental state, confusion, numbness, tingling, or motor weakness
- platelet count below 100 3 103 µL
- aspartate aminotransferase (AST), alanine aminotransferase (ALT), or lactic acid dehydrogenase (LDH) levels more than twice the upper limit of normal
- serum creatinine level >1.1 mg/dL
- suspected abruptio placentae.
A stepwise approach to eclampsia
Eclampsia is an obstetric emergency. Inadequate preparation for it or an inappropriate response to maternal and fetal conditions during and after an eclamptic convulsion can be detrimental to the mother and fetus. All obstetric units should have up-to-date protocols in place and should conduct mandatory drills to prepare nursing staff, obstetric providers, and anesthesia staff working in these units to manage eclampsia.
Step 1: Let the seizure run its course
During a seizure, resist the impulse to administer anticonvulsive drugs, including intravenous (IV) magnesium sulfate, because most eclamptic convulsions are self-limiting. Also abstain from administering medications such as IV phenytoin, diazepam, or midazolam, as these drugs are less effective than magnesium sulfate, and some can suppress the laryngeal reflex, increasing the risk of aspiration.
If the patient develops status epilepticus, initiate muscle paralysis and intubate her.
Step 2: Support the maternal condition
It is vital to support maternal respiratory and cardiovascular functions to prevent hypoxia, acidosis, and cardiorespiratory arrest.
Begin by establishing airway patency and maternal oxygenation during and after the convulsion. Administer oxygen via a face mask, with or without a reservoir, at a rate of 8 to 10 L/min.
During the apneic period (see “Profile of an eclamptic seizure” on page 46), the patient will develop hypoxia. Use pulse oximetry to monitor oxygen saturation, with the goal of keeping it above 94%. Arterial blood gas analysis is required if oxygen saturation remains below 92% or if pulmonary edema or aspiration is suspected.
If the patient develops recurrent seizures, status epilepticus, florid alveolar pulmonary edema, or respiratory arrest, intubate her immediately.
Step 3: Prevent maternal injury and aspiration
Secure the side rails of the patient’s bed by elevating them to prevent a fall, and make sure they are padded to prevent trauma during convulsions and afterward, when some women become combative and agitated. Position the patient in a lateral decubitus position to minimize aspiration of oral secretions. If any secretions or vomitus are present, remove them via suction.
Step 4: After the convulsion, give magnesium sulfate
Magnesium is the drug of choice for seizure prophylaxis in women with preeclampsia and severe symptoms, and to prevent recurrent seizures in women with eclampsia.
In the latter group, once the eclamptic convulsion has ended, give a loading dose of IV magnesium (6 g/100 mL over 20 minutes), followed by a continuous infusion of 2 g/h for at least 24 hours. If the patient develops a second seizure during the maintenance infusion, administer another bolus of magnesium (2 g/100 mL over 3–5 minutes).
Step 5: Treat severe hypertension
If severe hypertension persists for 60 minutes or longer, it can lead to injury of the brain, heart, and kidneys. To avoid these complications, it is essential to reduce BP to a safe range and maintain that level without compromising cerebral perfusion pressure and uteroplacental blood flow (which already may be reduced in some patients).
The goal of antihypertensive therapy is to keep systolic BP between 140 and 155 mm Hg and diastolic values between 90 and 105 mm Hg.9 Several agents are available for the treatment of severe hypertension during pregnancy and postpartum. The most commonly used IV medications for this purpose are labetalol and hydralazine. Another option is oral, rapidly acting
nifedipine.
Several randomized trials have compared efficacy and side effects between IV bolus injections of hydralazine; IV labetalol; and oral, rapidly acting nifedipine. In general, the findings of these studies suggest that either IV hydralazine or labetalol or oral nifedipine can be used to treat severe hypertension in pregnancy, as long as the provider is familiar with the dose to be used, the expected onset of action, and potential side effects (TABLE 3).
Women who develop generalized swelling or hemoconcentration (hematocrit ≥40%), or both, usually experience markedly reduced plasma volume. For this reason, these women will benefit from treatment with labetalol. If this is ineffective, then add IV hydralazine. However, delay administration of a rapidly acting vasodilator such as hydralazine to prevent an excessive hypotensive response and a secondary reduction in tissue perfusion and uteroplacental blood flow. Rather, administer a bolus infusion of 250 to 500 mL of isotonic saline before giving a vasodilator.
Additional details about the use of antihypertensive drugs are given in the section on other hypertensive emergencies below.
Step 6: Evaluate the patient for complications
Pulmonary edema can develop in patients with eclampsia or another hypertensive emergency. Suspect it if the patient has respiratory symptoms in association with tachypnea, tachycardia, or sustained oxygen saturation values below 93%, as well as when the patient exhibits basal rales during auscultation of the lungs. Treatment involves the administration of oxygen and IV furosemide (20–40 mg push), repeated as needed.
Some women with eclampsia may develop severe cerebral edema, hemorrhage, or both. The edema can be vasogenic or cytotoxic, leading to increased intracerebral pressure. Suspect edema or hemorrhage if the patient remains unresponsive, continues to experience convulsions despite therapy, or exhibits sensory or motor neurologic deficits. In such cases, neuroimaging is indicated, and the patient should be managed in consultation with neurology or neurosurgery.
Step 7: Begin the process of induction and delivery
Once the patient has been stabilized—and not before—initiate the induction process. Be aware that during and after the convulsion, changes in fetal heart rate (FHR) and uterine monitoring will usually be evident:
- prolonged deceleration or bradycardia (3–10 minutes)
- compensatory tachycardia, decreased beat-to-beat variability
- transient recurrent decelerations
- increased uterine tone and greater frequency of uterine activity.
These changes in FHR and uterine activity usually last 3 to 15 minutes. For this reason, it is important to avoid rushing the patient for cesarean delivery, as FHR and uterine activity are likely to return to normal after maternal resuscitation and stabilization. If not, consider other causes, such as abruptio placentae.
Eclampsia itself is not an indication for cesarean delivery. The selection of mode of delivery should be based on the presence or absence of labor, the cervical Bishop score, fetal gestational age, fetal presentation, and overall fetal condition.
Choosing an anesthetic
Regional analgesia/anesthesia is the method of choice for most women with eclampsia. However, regional anesthesia is to be avoided in the presence of disseminated intravascular coagulation or thrombocytopenia (the threshold platelet count is usually less than 75 x 103 µL. In such a case, IV analgesia can be used during labor, and general anesthesia may be appropriate for cesarean delivery. Both spinal and epidural analgesia and anesthesia are appropriate for women with eclampsia.
How to manage other hypertensive emergencies
A hypertensive emergency during pregnancy or postpartum involves acute-onset, persistent (>15 minutes), severe systolic BP (≥160 mm Hg) or severe diastolic BP (≥110 mm Hg), or both. The first step in such an emergency is to ensure the accurate measurement of BP using standard techniques.
Patients with acute-onset, persistent, severe BP should be hospitalized promptly for evaluation and treatment to prevent organ damage. Once such a patient is hospitalized, BP should be recorded every 15 minutes, with continuous FHR monitoring to ensure fetal viability.
Related article: Failure to diagnose preeclampsia and more (Medical Verdicts, February 2013)
The timing of initiation of antihypertensive medications, as well as determination of the type of medication best suited for the patient, should be based on:
- systolic and diastolic BP levels
- maternal clinical and laboratory findings
- presence of associated symptoms
- preexisting medical comorbidities
- whether the patient is antepartum or postpartum.
For example, a sustained BP level of 200/120 mm Hg requires therapy after 15 minutes, whereas observation may be suitable for as long as 60 minutes for a sustained BP of 160/72 mm Hg during labor.
Rapid reduction of systolic BP can lead to marked reductions in uteroplacental blood flow and a nonreassuring FHR tracing. Moreover, a rapid reduction of severe systolic BP in patients who have constricted plasma volume can reduce perfusion to the kidney, brain, and placenta. However, sustained BP of 165/100 mm Hg in association with central nervous system signs or symptoms, congestive heart failure, thrombocytopenia, or postpartum status requires therapy within 1 hour.
In general, it is difficult to obtain accurate BP recordings using noninvasive electronic instruments during labor because of the effects of labor on systolic BP and the lack of standardized methods for positioning of the arm cuff and the patient.
For these reasons, the decision about when to start acute antihypertensive therapy, based on systolic or diastolic BP, or both, should be individualized. And the choice of antihypertensive agent should be based on maternal clinical findings.
Choosing an antihypertensive agent
Because both hydralazine and nifedipine are associated with tachycardia, avoid them in patients with a heart rate above 110 bpm, using labetalol instead.10
In patients with bradycardia (heart rate <60 bpm), asthma, or congestive heart failure, however, labetalol should be avoided. In these populations, hydralazine or nifedipine is the drug of choice. Nifedipine is associated with improved renal blood flow and a resultant increase in urine output, making it preferable for patients with decreased urine output or severe postpartum hypertension.10
One theoretical concern is that the combined use of nifedipine and magnesium sulfate can cause excessive hypotension and neuromuscular blockage. As a result, some experts recommend that nifedipine be avoided in patients receiving magnesium sulfate. However, a recent review of this subject concluded that combined use of these drugs does not increase the risks of excessive hypotension and neuromuscular blockage in patients with severe hypertension or preeclampsia.
The initial dose of labetalol, when it is your chosen agent, is 20 mg IV, with BP measured 10 minutes later. If the target BP threshold is not achieved, administer 40 mg, 80 mg, and 80 mg at 10-minute intervals, as needed, again measuring BP 10 minutes after every dose. If, after a maximum dose of 240 mg, the desired BP threshold still has not been reached, give 5 to 10 mg IV hydralazine and measure BP 20 minutes later. If the target BP threshold still has not been achieved, it is essential to obtain consultation on the need for continuous infusion of labetalol, nicardipine, or sodium nitroprusside.
The initial dose of hydralazine, when it is your chosen agent, is 5 to 10 mg IV, with BP measured 20 minutes later. If needed, give another 10 mg and measure BP after another 20-minute interval. After a maximum dose of hydralazine 20 mg, switch to IV labetalol, using the regimen described above for labetalol, if the BP threshold still has not been achieved.
Nitroglycerin may be helpful in carefully selected patients
This drug is an arterial—but mostly venous—dilator. It is administered via IV infusion at an initial rate of 5 µg/min, with the rate gradually increased every 3 to 5 minutes (titrated to BP) to a maximum dose of 100 µg/min. It is the drug of choice in any hypertensive emergency associated with pulmonary edema and for control of hypertension associated with tracheal manipulation during intubation and extubation with general anesthesia.
Nitroglycerin is contraindicated in hypertensive encephalopathy because it increases cerebral blood flow and intracranial pressure. This drug should be administered only under the supervision of an experienced obstetric intensivist.
Sodium nitroprusside: Only in an ICU
This agent causes arterial and venous relaxation by interfering with the influx and intracellular activation of calcium. It is the drug of choice in hypertensive encephalopathy because it controls both afterload (vascular resistance) and preload (fluid status). It should be used only in the setting of intensive care.
The recommended dose is IV infusion at a rate of 0.25 to 5.00 µg/kg/min. Sodium nitroprusside has an immediate onset of action and may continue to exert an effect 3 to 5 minutes after discontinuation. Any hypotension caused by the drug should subside within minutes after discontinuation of the drip, due to the drug’s short half-life.
Nitroprusside is metabolized into thiocyanate and excreted in the urine. Cyanide can accumulate with large doses (>10 µg/kg/min) or prolonged administration (>48 hours), or if the patient has renal insufficiency or decreased hepatic metabolism. Signs of toxicity include anorexia, disorientation, headache, fatigue, restlessness, tinnitus, delirium, hallucinations, nausea, vomiting, and metabolic acidosis. When infused at a rate of less than 2 µg/kg/min, however, cyanide toxicity is unlikely.
As is the case with nitroglycerin, this drug should be administered only under the supervision of an experienced obstetric intensivist.
Case: Resolved
Upon arrival at the ED, the patient exhibits shallow, rapid breathing and foaming from the mouth. She is placed in a lateral decubitus position, an oral airway is established, and all secretions are suctioned. Oxygen is administered via face mask at a rate of 8 L/min. Her initial oxygen saturation level is 92%. IV access is secured, and a loading dose of magnesium sulfate 6 g is given over 20 minutes. Oxygen saturation increases to 94% to 96%. Auscultation of both lungs is normal.
The patient remains in a postictal state for about 15 minutes, but then orients to name, place, and time. FHR monitoring of both fetuses reveals a normal baseline with moderate variability, as well as variable decelerations in the presenting twin.
A maintenance dose of magnesium sulfate is initiated at a rate of 2 g/h, with the BP level recorded every 15 minutes. Systolic values remain between 170 and 180 mm Hg, and diastolic values between 108 and 112 mm Hg for 60 minutes. The obstetrician administers IV labetalol (20 mg) over 2 minutes. About 15 minutes later, the BP level is 154/100 mm Hg, with values remaining in the range of 150 to 156 mm Hg systolic and 92 to 104 mm Hg diastolic.
Ultrasonography reveals that the presenting twin is in a breech position, with estimated fetal weight below the 10th percentile and oligohydramnios. As a result, the obstetrician elects to proceed to cesarean delivery. The twins are delivered by cesarean section using spinal anesthesia. Although the infants are premature, there are no complications.Profile of an eclamptic seizure
Witnessing an eclamptic convulsion can be a frightening experience for nurses and medical providers. The convulsion usually lasts 60 to 90 seconds and occurs in two phases:
- Phase 1 (15–25 seconds) involves facial twitching, rolling of the eyes, and stiffening of the body, with generalized muscular contractions.
- Phase 2 (20–50 seconds) involves alternate contraction and relaxation of the muscles of the body in rapid succession, starting in the face and spreading throughout the body. Foaming at the mouth also occurs, and the patient may bite her tongue if it isn’t protected.
Apnea develops during and immediately after the convulsion, lasting about 120 seconds. A period of hyperventilation follows to compensate for the respiratory acidosis during the apneic period.
A postictal state follows the convulsion, and the patient usually remembers nothing of the episode. Some patients also become restless, combative, and agitated, requiring sedation. Aspiration is possible during or after the convulsion.
We want to hear from you! Tell us what you think.
- Sibai BM. Diagnosis, prevention, and management of eclampsia. Obstet Gynecol. 2005;105(2):402–410.
- Sibai BM. Diagnosis and management of gestational hypertension and preeclampsia. Obstet Gynecol. 2003;102(1):182–192.
- Meyer NL, Mercer BM, Friedman SA, Sibai BM. Urinary dipstick protein: a poor predictor of absent or severe proteinuria. Am J Obstet Gynecol. 1994;170(1 Pt 1):137–141.
- Knight M; UK Obstetric Surveillance System (UKOSS). Eclampsia in the United Kingdom 2005. BJOG. 2007;114(9):1072–1078.
- ACOG Practice Bulletin #33: Diagnosis and management of preeclampsia and eclampsia. Obstet Gynecol. 2002;99:159–167.
- Sibai BM, Stella CL. Diagnosis and management of atypical preeclampsia–eclampsia. Am J Obstet Gynecol. 2009;200(5):481.e1–e7.
- Sibai BM. Etiology and management of postpartum hypertension-preeclampsia. Am J Obstet Gynecol. 2012;206(6):470–475.
- ACOG Committee Opinion #514: Emergent therapy for acute-onset, severe hypertension with preeclampsia or eclampsia. Obstet Gynecol. 2011;118:1465–1468.
- Liu S, Joseph KS, Liston RM, et al. Incidence, risk factors, and associated complications of eclampsia. Obstet Gynecol. 2011;118(5):987–994.
- Raheem IA, Saaid R, Omar Sz, Tan PC. Oral nifedipine versus intravenous labetalol for acute blood pressure control in hypertensive emergencies of pregnancy: a randomized trial. BJOG. 2012;119(1):78–85.
- Sibai BM. Diagnosis, prevention, and management of eclampsia. Obstet Gynecol. 2005;105(2):402–410.
- Sibai BM. Diagnosis and management of gestational hypertension and preeclampsia. Obstet Gynecol. 2003;102(1):182–192.
- Meyer NL, Mercer BM, Friedman SA, Sibai BM. Urinary dipstick protein: a poor predictor of absent or severe proteinuria. Am J Obstet Gynecol. 1994;170(1 Pt 1):137–141.
- Knight M; UK Obstetric Surveillance System (UKOSS). Eclampsia in the United Kingdom 2005. BJOG. 2007;114(9):1072–1078.
- ACOG Practice Bulletin #33: Diagnosis and management of preeclampsia and eclampsia. Obstet Gynecol. 2002;99:159–167.
- Sibai BM, Stella CL. Diagnosis and management of atypical preeclampsia–eclampsia. Am J Obstet Gynecol. 2009;200(5):481.e1–e7.
- Sibai BM. Etiology and management of postpartum hypertension-preeclampsia. Am J Obstet Gynecol. 2012;206(6):470–475.
- ACOG Committee Opinion #514: Emergent therapy for acute-onset, severe hypertension with preeclampsia or eclampsia. Obstet Gynecol. 2011;118:1465–1468.
- Liu S, Joseph KS, Liston RM, et al. Incidence, risk factors, and associated complications of eclampsia. Obstet Gynecol. 2011;118(5):987–994.
- Raheem IA, Saaid R, Omar Sz, Tan PC. Oral nifedipine versus intravenous labetalol for acute blood pressure control in hypertensive emergencies of pregnancy: a randomized trial. BJOG. 2012;119(1):78–85.
Why obesity spells higher cesarean section rates
DENVER – The obesity epidemic has been a major driver of the steep rise in cesarean section rates documented nationally since the year 2000.
The contemporary trend is nicely captured by data on nearly 207,000 pregnancies in the mid-to-late years of the past decade collected by the Consortium on Safe Labor, a group of 19 university and community hospitals in all nine American College of Obstetricians and Gynecologists districts. As prepregnancy body mass index increased, so did the cesarean section rate, Dr. Mark Alanis observed at the annual meeting of the Society of Ob/Gyn Hospitalists.
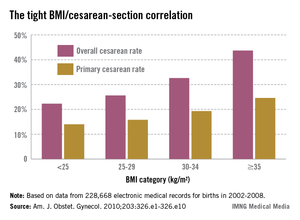
The Consortium used as their highest body mass index (BMI) category those women with a prepregnancy BMI of 35 kg/m2 or more, who made up 21% of their study population. Nationally, 7.6% of the obstetric population is extremely obese as defined by a BMI of 40 kg/m2 or more. The superobese – women with a prepregnancy BMI of 50 kg/m2 or more – make up a smaller subset, but they pose a special challenge for obstetricians. In the three studies that have examined cesarean section rates in superobese patients, one of which was conducted by Dr. Alanis, the rates were 55%, 49%, and 56%.
"It makes you wonder if it’s worth doing a randomized, controlled trial of scheduled primary cesarean section for women with a BMI of 50 kg/m2 or more. Is it more cost effective to try to do labor or just do the cesarean section? We don’t know," he said.
Why do obese women have a higher rate of cesarean section? The answer is beyond dispute, yet not widely appreciated; it’s because obese women tend to have ineffective uterine contractions in early labor, according to Dr. Alanis of the University of Colorado, Denver.
Persuasive evidence exists that greater body weight correlates with slowed cervical dilation. The first stage of labor is prolonged by about 2 hours in obese women. Dystocia occurs before 7 cm.
"You do these cesarean sections between 4 and 7 cm. You don’t do them after 7 cm very often," he noted. "The bottom line is if you can get your obese patient past 7 cm and she’s clearly in active labor, she’ll deliver just as easily as a lean patient."
Compelling data provided by the National Institutes of Health Maternal-Fetal Medicine Units Network show no differences in uterine forces or the length of the second stage of labor depending upon prepregnancy BMI in 5,341 nulliparous women.
"Maternal BMI in nulliparous women reaching the second stage is not associated with a higher incidence of cesarean delivery," according to the investigators (Obstet. Gynecol. 2011;118:1309-13).
This finding confirms earlier work by Australian/New Zealand clinical trialists in a study involving 2,629 nulliparous women who went into labor after 37 weeks’ gestation. Being overweight or obese, respectively, was associated with adjusted 39% and 2.9-fold increased likelihood of cesarean delivery during the first stage of labor, compared with normal-weight women, but no increase in second-stage cesarean delivery (Obstet. Gynecol. 2011;117:1315-22).
These findings of an overall longer duration and slower progression of the early part of the first stage of labor have led to a proposal for adoption of a separate obese labor curve (Obstet. Gynecol. 2012;120:130-5).
"We probably should," according to Dr. Alanis.
The indication for cesarean section in obese patients is disproportionately failure to progress. Fetal distress as a trigger for cesarean delivery is not more common than in normal-weight women.
The explanation for the slower course of first-stage labor in obese women remains unclear, according to Dr. Alanis. Neither smooth muscle content nor contraction strength differs between obese and leaner women.
Dr. Alanis offers vaginal birth after cesarean section to obese patients he considers to have a decent chance of success. But he explains to them that they have a higher risk of uterine rupture, are more likely to develop endometritis, and are less likely to have a successful vaginal delivery than are normal-weight women, based upon data from the Maternal-Fetal Medicine Units Network.
"So approach TOLAC [trial of labor after cesarean] cautiously," he advised.
Dr. Alanis reported having no financial interests germane to his presentation.
DENVER – The obesity epidemic has been a major driver of the steep rise in cesarean section rates documented nationally since the year 2000.
The contemporary trend is nicely captured by data on nearly 207,000 pregnancies in the mid-to-late years of the past decade collected by the Consortium on Safe Labor, a group of 19 university and community hospitals in all nine American College of Obstetricians and Gynecologists districts. As prepregnancy body mass index increased, so did the cesarean section rate, Dr. Mark Alanis observed at the annual meeting of the Society of Ob/Gyn Hospitalists.

The Consortium used as their highest body mass index (BMI) category those women with a prepregnancy BMI of 35 kg/m2 or more, who made up 21% of their study population. Nationally, 7.6% of the obstetric population is extremely obese as defined by a BMI of 40 kg/m2 or more. The superobese – women with a prepregnancy BMI of 50 kg/m2 or more – make up a smaller subset, but they pose a special challenge for obstetricians. In the three studies that have examined cesarean section rates in superobese patients, one of which was conducted by Dr. Alanis, the rates were 55%, 49%, and 56%.
"It makes you wonder if it’s worth doing a randomized, controlled trial of scheduled primary cesarean section for women with a BMI of 50 kg/m2 or more. Is it more cost effective to try to do labor or just do the cesarean section? We don’t know," he said.
Why do obese women have a higher rate of cesarean section? The answer is beyond dispute, yet not widely appreciated; it’s because obese women tend to have ineffective uterine contractions in early labor, according to Dr. Alanis of the University of Colorado, Denver.
Persuasive evidence exists that greater body weight correlates with slowed cervical dilation. The first stage of labor is prolonged by about 2 hours in obese women. Dystocia occurs before 7 cm.
"You do these cesarean sections between 4 and 7 cm. You don’t do them after 7 cm very often," he noted. "The bottom line is if you can get your obese patient past 7 cm and she’s clearly in active labor, she’ll deliver just as easily as a lean patient."
Compelling data provided by the National Institutes of Health Maternal-Fetal Medicine Units Network show no differences in uterine forces or the length of the second stage of labor depending upon prepregnancy BMI in 5,341 nulliparous women.
"Maternal BMI in nulliparous women reaching the second stage is not associated with a higher incidence of cesarean delivery," according to the investigators (Obstet. Gynecol. 2011;118:1309-13).
This finding confirms earlier work by Australian/New Zealand clinical trialists in a study involving 2,629 nulliparous women who went into labor after 37 weeks’ gestation. Being overweight or obese, respectively, was associated with adjusted 39% and 2.9-fold increased likelihood of cesarean delivery during the first stage of labor, compared with normal-weight women, but no increase in second-stage cesarean delivery (Obstet. Gynecol. 2011;117:1315-22).
These findings of an overall longer duration and slower progression of the early part of the first stage of labor have led to a proposal for adoption of a separate obese labor curve (Obstet. Gynecol. 2012;120:130-5).
"We probably should," according to Dr. Alanis.
The indication for cesarean section in obese patients is disproportionately failure to progress. Fetal distress as a trigger for cesarean delivery is not more common than in normal-weight women.
The explanation for the slower course of first-stage labor in obese women remains unclear, according to Dr. Alanis. Neither smooth muscle content nor contraction strength differs between obese and leaner women.
Dr. Alanis offers vaginal birth after cesarean section to obese patients he considers to have a decent chance of success. But he explains to them that they have a higher risk of uterine rupture, are more likely to develop endometritis, and are less likely to have a successful vaginal delivery than are normal-weight women, based upon data from the Maternal-Fetal Medicine Units Network.
"So approach TOLAC [trial of labor after cesarean] cautiously," he advised.
Dr. Alanis reported having no financial interests germane to his presentation.
DENVER – The obesity epidemic has been a major driver of the steep rise in cesarean section rates documented nationally since the year 2000.
The contemporary trend is nicely captured by data on nearly 207,000 pregnancies in the mid-to-late years of the past decade collected by the Consortium on Safe Labor, a group of 19 university and community hospitals in all nine American College of Obstetricians and Gynecologists districts. As prepregnancy body mass index increased, so did the cesarean section rate, Dr. Mark Alanis observed at the annual meeting of the Society of Ob/Gyn Hospitalists.

The Consortium used as their highest body mass index (BMI) category those women with a prepregnancy BMI of 35 kg/m2 or more, who made up 21% of their study population. Nationally, 7.6% of the obstetric population is extremely obese as defined by a BMI of 40 kg/m2 or more. The superobese – women with a prepregnancy BMI of 50 kg/m2 or more – make up a smaller subset, but they pose a special challenge for obstetricians. In the three studies that have examined cesarean section rates in superobese patients, one of which was conducted by Dr. Alanis, the rates were 55%, 49%, and 56%.
"It makes you wonder if it’s worth doing a randomized, controlled trial of scheduled primary cesarean section for women with a BMI of 50 kg/m2 or more. Is it more cost effective to try to do labor or just do the cesarean section? We don’t know," he said.
Why do obese women have a higher rate of cesarean section? The answer is beyond dispute, yet not widely appreciated; it’s because obese women tend to have ineffective uterine contractions in early labor, according to Dr. Alanis of the University of Colorado, Denver.
Persuasive evidence exists that greater body weight correlates with slowed cervical dilation. The first stage of labor is prolonged by about 2 hours in obese women. Dystocia occurs before 7 cm.
"You do these cesarean sections between 4 and 7 cm. You don’t do them after 7 cm very often," he noted. "The bottom line is if you can get your obese patient past 7 cm and she’s clearly in active labor, she’ll deliver just as easily as a lean patient."
Compelling data provided by the National Institutes of Health Maternal-Fetal Medicine Units Network show no differences in uterine forces or the length of the second stage of labor depending upon prepregnancy BMI in 5,341 nulliparous women.
"Maternal BMI in nulliparous women reaching the second stage is not associated with a higher incidence of cesarean delivery," according to the investigators (Obstet. Gynecol. 2011;118:1309-13).
This finding confirms earlier work by Australian/New Zealand clinical trialists in a study involving 2,629 nulliparous women who went into labor after 37 weeks’ gestation. Being overweight or obese, respectively, was associated with adjusted 39% and 2.9-fold increased likelihood of cesarean delivery during the first stage of labor, compared with normal-weight women, but no increase in second-stage cesarean delivery (Obstet. Gynecol. 2011;117:1315-22).
These findings of an overall longer duration and slower progression of the early part of the first stage of labor have led to a proposal for adoption of a separate obese labor curve (Obstet. Gynecol. 2012;120:130-5).
"We probably should," according to Dr. Alanis.
The indication for cesarean section in obese patients is disproportionately failure to progress. Fetal distress as a trigger for cesarean delivery is not more common than in normal-weight women.
The explanation for the slower course of first-stage labor in obese women remains unclear, according to Dr. Alanis. Neither smooth muscle content nor contraction strength differs between obese and leaner women.
Dr. Alanis offers vaginal birth after cesarean section to obese patients he considers to have a decent chance of success. But he explains to them that they have a higher risk of uterine rupture, are more likely to develop endometritis, and are less likely to have a successful vaginal delivery than are normal-weight women, based upon data from the Maternal-Fetal Medicine Units Network.
"So approach TOLAC [trial of labor after cesarean] cautiously," he advised.
Dr. Alanis reported having no financial interests germane to his presentation.
EXPERT ANALYSIS FROM THE SOGH ANNUAL CLINICAL MEETING
Brain shadowing sign indicates fetal craniosynostosis
SYDNEY, AUSTRALIA – The brain shadowing sign – a sharply demarcated area of acoustic shadowing on ultrasound – is a novel and easily identifiable marker for fetal craniosynostosis, a small multicenter retrospective study showed.
An analysis of ultrasound images from 16 patients with a postnatal diagnosis of fetal craniosynostosis, presented at the International Society of Ultrasound in Obstetrics and Gynecology world congress, showed the brain shadowing sign was clearly depicted in all cases, even when the suture was only partly closed.
Coauthor Gustavo Malinger, director of the ob.gyn. ultrasound unit at the Tel Aviv Sourasky Medical Centre, said prenatal diagnosis of fetal craniosynostosis is often difficult, particularly if it involves the sagittal suture and occurs in the absence of a family history. As a result, most cases are delivered without a diagnosis.
"With the metopic suture, you have a chance to make a diagnosis because the shape of the head is quite characteristic. So with the third-trimester examination, when you perform a weight estimation or something like that, you see that something is wrong," Dr. Malinger said in an interview.
"But when the sagittal suture is closed, you don’t see it because you come from the side; you obtain axial planes and you never reach the sagittal suture."
The brain shadowing sign occurs because of a failure of the acoustic wave to cross the cortical bone, and appears as a line separating a zone of clearly defined brain anatomy and a zone of relative shadow.
The sign is easily identifiable, is not dependent on fetal position, and does not require visualization with high-definition 3D transducers.
All patients underwent a multiplanar neurosonographic exam with transvaginal and transabdominal ultrasound.
The mean gestational age at diagnosis was 29 weeks, and the diagnosis was made in the second trimester in only five cases. Dr. Malinger said that while it was possible in some cases to diagnose very severe cases at 22 weeks, fetal craniosynostosis tends to develop later in the pregnancy.
Prenatal diagnosis did not necessarily enable any kind of intervention, he added, but it was important in enabling counselling to be provided to families to prepare them for what to expect.
"They are ready to know, and we send them to a neurosurgical consultation in utero, so the neurosurgeon can explain that in most of the cases, it’s going to be OK, and that we’ll follow up," Dr. Malinger said.
Of the 16 patients included in the study, 14 presented with single-suture craniosynostosis: 6 involved metopic suture; 5, sagittal suture; and 3, coronal suture.
Nine cases were isolated fetal craniosynostosis, three were cases of Apert syndrome, and four also presented with associated CNS and non-CNS anomalies.
Dr. Malinger said the earlier diagnosis of fetal craniosynostosis could enable discovery of other related malformations that might otherwise have been missed.
No financial conflicts were reported.
SYDNEY, AUSTRALIA – The brain shadowing sign – a sharply demarcated area of acoustic shadowing on ultrasound – is a novel and easily identifiable marker for fetal craniosynostosis, a small multicenter retrospective study showed.
An analysis of ultrasound images from 16 patients with a postnatal diagnosis of fetal craniosynostosis, presented at the International Society of Ultrasound in Obstetrics and Gynecology world congress, showed the brain shadowing sign was clearly depicted in all cases, even when the suture was only partly closed.
Coauthor Gustavo Malinger, director of the ob.gyn. ultrasound unit at the Tel Aviv Sourasky Medical Centre, said prenatal diagnosis of fetal craniosynostosis is often difficult, particularly if it involves the sagittal suture and occurs in the absence of a family history. As a result, most cases are delivered without a diagnosis.
"With the metopic suture, you have a chance to make a diagnosis because the shape of the head is quite characteristic. So with the third-trimester examination, when you perform a weight estimation or something like that, you see that something is wrong," Dr. Malinger said in an interview.
"But when the sagittal suture is closed, you don’t see it because you come from the side; you obtain axial planes and you never reach the sagittal suture."
The brain shadowing sign occurs because of a failure of the acoustic wave to cross the cortical bone, and appears as a line separating a zone of clearly defined brain anatomy and a zone of relative shadow.
The sign is easily identifiable, is not dependent on fetal position, and does not require visualization with high-definition 3D transducers.
All patients underwent a multiplanar neurosonographic exam with transvaginal and transabdominal ultrasound.
The mean gestational age at diagnosis was 29 weeks, and the diagnosis was made in the second trimester in only five cases. Dr. Malinger said that while it was possible in some cases to diagnose very severe cases at 22 weeks, fetal craniosynostosis tends to develop later in the pregnancy.
Prenatal diagnosis did not necessarily enable any kind of intervention, he added, but it was important in enabling counselling to be provided to families to prepare them for what to expect.
"They are ready to know, and we send them to a neurosurgical consultation in utero, so the neurosurgeon can explain that in most of the cases, it’s going to be OK, and that we’ll follow up," Dr. Malinger said.
Of the 16 patients included in the study, 14 presented with single-suture craniosynostosis: 6 involved metopic suture; 5, sagittal suture; and 3, coronal suture.
Nine cases were isolated fetal craniosynostosis, three were cases of Apert syndrome, and four also presented with associated CNS and non-CNS anomalies.
Dr. Malinger said the earlier diagnosis of fetal craniosynostosis could enable discovery of other related malformations that might otherwise have been missed.
No financial conflicts were reported.
SYDNEY, AUSTRALIA – The brain shadowing sign – a sharply demarcated area of acoustic shadowing on ultrasound – is a novel and easily identifiable marker for fetal craniosynostosis, a small multicenter retrospective study showed.
An analysis of ultrasound images from 16 patients with a postnatal diagnosis of fetal craniosynostosis, presented at the International Society of Ultrasound in Obstetrics and Gynecology world congress, showed the brain shadowing sign was clearly depicted in all cases, even when the suture was only partly closed.
Coauthor Gustavo Malinger, director of the ob.gyn. ultrasound unit at the Tel Aviv Sourasky Medical Centre, said prenatal diagnosis of fetal craniosynostosis is often difficult, particularly if it involves the sagittal suture and occurs in the absence of a family history. As a result, most cases are delivered without a diagnosis.
"With the metopic suture, you have a chance to make a diagnosis because the shape of the head is quite characteristic. So with the third-trimester examination, when you perform a weight estimation or something like that, you see that something is wrong," Dr. Malinger said in an interview.
"But when the sagittal suture is closed, you don’t see it because you come from the side; you obtain axial planes and you never reach the sagittal suture."
The brain shadowing sign occurs because of a failure of the acoustic wave to cross the cortical bone, and appears as a line separating a zone of clearly defined brain anatomy and a zone of relative shadow.
The sign is easily identifiable, is not dependent on fetal position, and does not require visualization with high-definition 3D transducers.
All patients underwent a multiplanar neurosonographic exam with transvaginal and transabdominal ultrasound.
The mean gestational age at diagnosis was 29 weeks, and the diagnosis was made in the second trimester in only five cases. Dr. Malinger said that while it was possible in some cases to diagnose very severe cases at 22 weeks, fetal craniosynostosis tends to develop later in the pregnancy.
Prenatal diagnosis did not necessarily enable any kind of intervention, he added, but it was important in enabling counselling to be provided to families to prepare them for what to expect.
"They are ready to know, and we send them to a neurosurgical consultation in utero, so the neurosurgeon can explain that in most of the cases, it’s going to be OK, and that we’ll follow up," Dr. Malinger said.
Of the 16 patients included in the study, 14 presented with single-suture craniosynostosis: 6 involved metopic suture; 5, sagittal suture; and 3, coronal suture.
Nine cases were isolated fetal craniosynostosis, three were cases of Apert syndrome, and four also presented with associated CNS and non-CNS anomalies.
Dr. Malinger said the earlier diagnosis of fetal craniosynostosis could enable discovery of other related malformations that might otherwise have been missed.
No financial conflicts were reported.
AT THE ISUOG WORLD CONGRESS
Miscarriage risk is not increased with subchorionic hematoma
SYDNEY – The presence of subchorionic bleeding around the gestational sac does not appear to increase the risk of miscarriage, according to data from a prospective observational study.
In a study of 1,115 women in early pregnancy, 142 (13%) had documented subchorionic hematoma. The women were diagnosed either after presenting with bleeding or during a routine dating scan. Researchers looked at the outcomes of pregnancy in both groups at the end of the first trimester and the final pregnancy outcome. They found no statistically significant association between the presence of subchorionic bleeding and miscarriage, according to findings presented at the International Society on Ultrasound in Obstetrics and Gynecology world congress.
Lead author Dr. Nicole Stamatopoulos said the study came about because she observed a significant number of women attending the unit with subchorionic hematoma.
Looking at viable pregnancies that later miscarry, "we wondered if the bleeding around the gestational sac was one of the reasons why these women were miscarrying," said Dr. Stamatopoulos, a researcher at Nepean Hospital in Penrith, Sydney.
Dr. Stamatopoulos said that a diagnosis of subchorionic hematoma – which is one of the most common sonographic abnormalities with live embryos – often caused women a lot of concern, particularly when it was accompanied by vaginal bleeding.
She said the findings should offer some reassurance to these women.
"If you’ve got an embryo that’s got a heart rate, and someone’s come in with bleeding, and there is a subchorionic hematoma, I think you can reassure those women," Dr. Stamatopoulos said in an interview.
However, she said that reassurance could not necessarily be extended to women in whom a viable pregnancy was yet to be established.
The causes of subchorionic hematoma are not well understood, but the condition may result from trauma or may be related to the process of the trophoblast embedding into the uterus, Dr. Stamatopoulos said.
The next step in the study is to explore a possible relationship between the size of the hematoma and pregnancy outcomes.
"I’ve got data relating to the actual size of the subchorionic hematoma, to see whether or not, if it’s a really large one, does that increase the risk of miscarriage? Or if it’s just small, is it not a problem?" she said.
There were no relevant financial conflicts of interest declared.
SYDNEY – The presence of subchorionic bleeding around the gestational sac does not appear to increase the risk of miscarriage, according to data from a prospective observational study.
In a study of 1,115 women in early pregnancy, 142 (13%) had documented subchorionic hematoma. The women were diagnosed either after presenting with bleeding or during a routine dating scan. Researchers looked at the outcomes of pregnancy in both groups at the end of the first trimester and the final pregnancy outcome. They found no statistically significant association between the presence of subchorionic bleeding and miscarriage, according to findings presented at the International Society on Ultrasound in Obstetrics and Gynecology world congress.
Lead author Dr. Nicole Stamatopoulos said the study came about because she observed a significant number of women attending the unit with subchorionic hematoma.
Looking at viable pregnancies that later miscarry, "we wondered if the bleeding around the gestational sac was one of the reasons why these women were miscarrying," said Dr. Stamatopoulos, a researcher at Nepean Hospital in Penrith, Sydney.
Dr. Stamatopoulos said that a diagnosis of subchorionic hematoma – which is one of the most common sonographic abnormalities with live embryos – often caused women a lot of concern, particularly when it was accompanied by vaginal bleeding.
She said the findings should offer some reassurance to these women.
"If you’ve got an embryo that’s got a heart rate, and someone’s come in with bleeding, and there is a subchorionic hematoma, I think you can reassure those women," Dr. Stamatopoulos said in an interview.
However, she said that reassurance could not necessarily be extended to women in whom a viable pregnancy was yet to be established.
The causes of subchorionic hematoma are not well understood, but the condition may result from trauma or may be related to the process of the trophoblast embedding into the uterus, Dr. Stamatopoulos said.
The next step in the study is to explore a possible relationship between the size of the hematoma and pregnancy outcomes.
"I’ve got data relating to the actual size of the subchorionic hematoma, to see whether or not, if it’s a really large one, does that increase the risk of miscarriage? Or if it’s just small, is it not a problem?" she said.
There were no relevant financial conflicts of interest declared.
SYDNEY – The presence of subchorionic bleeding around the gestational sac does not appear to increase the risk of miscarriage, according to data from a prospective observational study.
In a study of 1,115 women in early pregnancy, 142 (13%) had documented subchorionic hematoma. The women were diagnosed either after presenting with bleeding or during a routine dating scan. Researchers looked at the outcomes of pregnancy in both groups at the end of the first trimester and the final pregnancy outcome. They found no statistically significant association between the presence of subchorionic bleeding and miscarriage, according to findings presented at the International Society on Ultrasound in Obstetrics and Gynecology world congress.
Lead author Dr. Nicole Stamatopoulos said the study came about because she observed a significant number of women attending the unit with subchorionic hematoma.
Looking at viable pregnancies that later miscarry, "we wondered if the bleeding around the gestational sac was one of the reasons why these women were miscarrying," said Dr. Stamatopoulos, a researcher at Nepean Hospital in Penrith, Sydney.
Dr. Stamatopoulos said that a diagnosis of subchorionic hematoma – which is one of the most common sonographic abnormalities with live embryos – often caused women a lot of concern, particularly when it was accompanied by vaginal bleeding.
She said the findings should offer some reassurance to these women.
"If you’ve got an embryo that’s got a heart rate, and someone’s come in with bleeding, and there is a subchorionic hematoma, I think you can reassure those women," Dr. Stamatopoulos said in an interview.
However, she said that reassurance could not necessarily be extended to women in whom a viable pregnancy was yet to be established.
The causes of subchorionic hematoma are not well understood, but the condition may result from trauma or may be related to the process of the trophoblast embedding into the uterus, Dr. Stamatopoulos said.
The next step in the study is to explore a possible relationship between the size of the hematoma and pregnancy outcomes.
"I’ve got data relating to the actual size of the subchorionic hematoma, to see whether or not, if it’s a really large one, does that increase the risk of miscarriage? Or if it’s just small, is it not a problem?" she said.
There were no relevant financial conflicts of interest declared.
AT THE ISUOG WORLD CONGRESS
Evaluation for possible early pregnancy failure
DENVER – There is a slew of ultrasound findings that are equivocal or downright worrisome for the diagnosis of early pregnancy failure, but the list of definitive findings is short – and even those are fraught with controversy, according to Dr. Roxanne Vrees.
"In a highly desired pregnancy, even with ultrasound findings that are perhaps suggestive of early pregnancy failure, I think watchful waiting has a really important role," said Dr. Vrees, an ob.gyn. at Brown University in Providence, R.I.
She is also on the staff at the Women and Infants Hospital in Providence, where she works in a unique women’s emergency department staffed 24/7 exclusively by ob.gyn. attending physicians. Traditional emergency medicine physicians are not in the picture. This busy women’s ED averages nearly 30,000 visits annually, so Dr. Vrees and her colleagues have acquired considerable experience in emergency ob.gyn. However, because the facility is licensed by the state as an ED, by law any patient who comes in must be treated, so medical backup is available around the clock for patients who present with chest pain or otherwise fall outside the generalist ob.gyn.’s scope.
Early pregnancy failure occurs in 15%-20% of clinically recognized pregnancies. The most common symptom is vaginal bleeding, which occurs in one-quarter of all known first-trimester pregnancies, half of which end in pregnancy failure.
Dr. Vrees emphasized that no single aspect of the work-up for early pregnancy failure should drive patient management. That ought to be based upon a combination of the patient’s symptoms, laboratory findings, physical exam, and pelvic ultrasound.
In the setting of worrisome ultrasound findings, the most important step in management is to get a repeat ultrasound under real-time observation. The use of cine loops in order to visualize the entire gestational sac is valuable.
"Most obstetricians underutilize this. You’re unlikely to miss a yolk sac or embryo, and you’re getting a true report of sac diameter, not a random measurement from a snapshot," she explained at the annual meeting of the Society of Ob/Gyn Hospitalists.
Worrisome but nondefinitive findings suggestive of early pregnancy loss include a slow fetal heart rate, an unusual appearance of the uterine lining, and a sac that is small, grossly distorted, enlarged, irregularly contoured, or low in position.
Maternal gestational age is a key consideration in defining a slow fetal heart rate by M-mode ultrasound. At a menstrual age of 6.2 weeks or less, a normal fetal heart rate is 100 bpm or more; less than 90 bpm is considered slow. In contrast, at 6.3-7.0 weeks, normal is defined as 120 bpm or more, and a fetal heart rate of less than 110 bpm is considered slow. When a slow fetal heart rate is detected at 6.0-7.0 weeks, the risk of subsequent first-trimester fetal demise remains elevated at about 25% even if the heart rate is normal at follow-up at 8.0 weeks (Radiology;2005;236:643-6).
The absolute ultrasound criteria for early pregnancy failure used at the Women and Infants Hospital as well as in many other settings are no fetal heart beat in an embryo more than 5 mm in crown-rump length, or menstrual age known to be greater than 6.5 weeks with no heart beat.
However, a group of investigators led by Dr. Yazan Abdallah of Imperial College London has argued that current definitions used to diagnose early pregnancy failure are potentially unsafe and could result in inadvertent termination of wanted pregnancies. Given the inherent inter- and intraobserver variation in ultrasound measurements, they have urged more conservative criteria for the definitive diagnosis of early pregnancy failure: specifically, a crown-rump length cutoff of greater than 7 mm instead of the widely used 5 mm, and a mean gestational sac diameter cutoff of more than 25 mm.
Dr. Abdallah and his coworkers support their argument on the basis of their observational, prospective cross-sectional study involving 1,060 consecutive women diagnosed with intrauterine pregnancy of uncertain viability. This diagnosis was based upon a symptom-generated ultrasound that showed either an empty gestational sac; a gestational sac with a yolk sac but no embryo when the mean gestational sac diameter was either less than 20 mm or less than 30 mm; or an embryo with an absent heart beat and a crown-rump length of either less than 6 mm or less than 8 mm.
The primary endpoint was a viable pregnancy upon routine first-trimester screening ultrasound at 11-14 weeks. At that point, 473 of the women had viable pregnancies and 587 did not.
When neither the yolk sac nor the embryo was visualized on the initial ultrasound, the false-positive rate for diagnosis of early pregnancy failure was 4.4% when a mean gestational sac diameter of 16 mm was used as a cutoff and 0.5% when 20 mm was the cutoff. Only when a cutoff of 21 mm was utilized did the false-positive rate fall to 0.
If a yolk sac was present but an embryo wasn’t, the false-positive rate was 2.6% with a mean gestational sac diameter cutoff of 16 mm and 0.4% with a cutoff of 20 mm. Again, there were no false positives when a cutoff of 21 mm was employed.
When a yolk sac and embryo were visible but a fetal heartbeat was not apparent, the false-positive rate for miscarriage was 8.3% with a crown-rump length cutoff of 5 mm. At a cutoff of 5.3 mm, there were no false-positive results (Ultrasound Obstet. Gynecol. 2011;38:497-502).
When Dr. Vrees asked the audience for a show of hands as to who utilizes a mean gestational sac diameter cutoff of 21 mm to define early pregnancy loss in the absence of both a yolk sac and embryo, only a couple of ob.gyns. responded affirmatively. Some audience members indicated they use a cutoff as low as 16 mm.
"We see great variability at our institution, too, in the definition of early pregnancy failure based upon mean gestational sac diameter," according to Dr. Vrees.
The lack of unanimity on this point, coupled with the remote likelihood of physical harm in waiting 7-10 days to repeat an ultrasound scan, figure prominently in her advocacy of expectant management.
She reported having no relevant financial disclosures.
DENVER – There is a slew of ultrasound findings that are equivocal or downright worrisome for the diagnosis of early pregnancy failure, but the list of definitive findings is short – and even those are fraught with controversy, according to Dr. Roxanne Vrees.
"In a highly desired pregnancy, even with ultrasound findings that are perhaps suggestive of early pregnancy failure, I think watchful waiting has a really important role," said Dr. Vrees, an ob.gyn. at Brown University in Providence, R.I.
She is also on the staff at the Women and Infants Hospital in Providence, where she works in a unique women’s emergency department staffed 24/7 exclusively by ob.gyn. attending physicians. Traditional emergency medicine physicians are not in the picture. This busy women’s ED averages nearly 30,000 visits annually, so Dr. Vrees and her colleagues have acquired considerable experience in emergency ob.gyn. However, because the facility is licensed by the state as an ED, by law any patient who comes in must be treated, so medical backup is available around the clock for patients who present with chest pain or otherwise fall outside the generalist ob.gyn.’s scope.
Early pregnancy failure occurs in 15%-20% of clinically recognized pregnancies. The most common symptom is vaginal bleeding, which occurs in one-quarter of all known first-trimester pregnancies, half of which end in pregnancy failure.
Dr. Vrees emphasized that no single aspect of the work-up for early pregnancy failure should drive patient management. That ought to be based upon a combination of the patient’s symptoms, laboratory findings, physical exam, and pelvic ultrasound.
In the setting of worrisome ultrasound findings, the most important step in management is to get a repeat ultrasound under real-time observation. The use of cine loops in order to visualize the entire gestational sac is valuable.
"Most obstetricians underutilize this. You’re unlikely to miss a yolk sac or embryo, and you’re getting a true report of sac diameter, not a random measurement from a snapshot," she explained at the annual meeting of the Society of Ob/Gyn Hospitalists.
Worrisome but nondefinitive findings suggestive of early pregnancy loss include a slow fetal heart rate, an unusual appearance of the uterine lining, and a sac that is small, grossly distorted, enlarged, irregularly contoured, or low in position.
Maternal gestational age is a key consideration in defining a slow fetal heart rate by M-mode ultrasound. At a menstrual age of 6.2 weeks or less, a normal fetal heart rate is 100 bpm or more; less than 90 bpm is considered slow. In contrast, at 6.3-7.0 weeks, normal is defined as 120 bpm or more, and a fetal heart rate of less than 110 bpm is considered slow. When a slow fetal heart rate is detected at 6.0-7.0 weeks, the risk of subsequent first-trimester fetal demise remains elevated at about 25% even if the heart rate is normal at follow-up at 8.0 weeks (Radiology;2005;236:643-6).
The absolute ultrasound criteria for early pregnancy failure used at the Women and Infants Hospital as well as in many other settings are no fetal heart beat in an embryo more than 5 mm in crown-rump length, or menstrual age known to be greater than 6.5 weeks with no heart beat.
However, a group of investigators led by Dr. Yazan Abdallah of Imperial College London has argued that current definitions used to diagnose early pregnancy failure are potentially unsafe and could result in inadvertent termination of wanted pregnancies. Given the inherent inter- and intraobserver variation in ultrasound measurements, they have urged more conservative criteria for the definitive diagnosis of early pregnancy failure: specifically, a crown-rump length cutoff of greater than 7 mm instead of the widely used 5 mm, and a mean gestational sac diameter cutoff of more than 25 mm.
Dr. Abdallah and his coworkers support their argument on the basis of their observational, prospective cross-sectional study involving 1,060 consecutive women diagnosed with intrauterine pregnancy of uncertain viability. This diagnosis was based upon a symptom-generated ultrasound that showed either an empty gestational sac; a gestational sac with a yolk sac but no embryo when the mean gestational sac diameter was either less than 20 mm or less than 30 mm; or an embryo with an absent heart beat and a crown-rump length of either less than 6 mm or less than 8 mm.
The primary endpoint was a viable pregnancy upon routine first-trimester screening ultrasound at 11-14 weeks. At that point, 473 of the women had viable pregnancies and 587 did not.
When neither the yolk sac nor the embryo was visualized on the initial ultrasound, the false-positive rate for diagnosis of early pregnancy failure was 4.4% when a mean gestational sac diameter of 16 mm was used as a cutoff and 0.5% when 20 mm was the cutoff. Only when a cutoff of 21 mm was utilized did the false-positive rate fall to 0.
If a yolk sac was present but an embryo wasn’t, the false-positive rate was 2.6% with a mean gestational sac diameter cutoff of 16 mm and 0.4% with a cutoff of 20 mm. Again, there were no false positives when a cutoff of 21 mm was employed.
When a yolk sac and embryo were visible but a fetal heartbeat was not apparent, the false-positive rate for miscarriage was 8.3% with a crown-rump length cutoff of 5 mm. At a cutoff of 5.3 mm, there were no false-positive results (Ultrasound Obstet. Gynecol. 2011;38:497-502).
When Dr. Vrees asked the audience for a show of hands as to who utilizes a mean gestational sac diameter cutoff of 21 mm to define early pregnancy loss in the absence of both a yolk sac and embryo, only a couple of ob.gyns. responded affirmatively. Some audience members indicated they use a cutoff as low as 16 mm.
"We see great variability at our institution, too, in the definition of early pregnancy failure based upon mean gestational sac diameter," according to Dr. Vrees.
The lack of unanimity on this point, coupled with the remote likelihood of physical harm in waiting 7-10 days to repeat an ultrasound scan, figure prominently in her advocacy of expectant management.
She reported having no relevant financial disclosures.
DENVER – There is a slew of ultrasound findings that are equivocal or downright worrisome for the diagnosis of early pregnancy failure, but the list of definitive findings is short – and even those are fraught with controversy, according to Dr. Roxanne Vrees.
"In a highly desired pregnancy, even with ultrasound findings that are perhaps suggestive of early pregnancy failure, I think watchful waiting has a really important role," said Dr. Vrees, an ob.gyn. at Brown University in Providence, R.I.
She is also on the staff at the Women and Infants Hospital in Providence, where she works in a unique women’s emergency department staffed 24/7 exclusively by ob.gyn. attending physicians. Traditional emergency medicine physicians are not in the picture. This busy women’s ED averages nearly 30,000 visits annually, so Dr. Vrees and her colleagues have acquired considerable experience in emergency ob.gyn. However, because the facility is licensed by the state as an ED, by law any patient who comes in must be treated, so medical backup is available around the clock for patients who present with chest pain or otherwise fall outside the generalist ob.gyn.’s scope.
Early pregnancy failure occurs in 15%-20% of clinically recognized pregnancies. The most common symptom is vaginal bleeding, which occurs in one-quarter of all known first-trimester pregnancies, half of which end in pregnancy failure.
Dr. Vrees emphasized that no single aspect of the work-up for early pregnancy failure should drive patient management. That ought to be based upon a combination of the patient’s symptoms, laboratory findings, physical exam, and pelvic ultrasound.
In the setting of worrisome ultrasound findings, the most important step in management is to get a repeat ultrasound under real-time observation. The use of cine loops in order to visualize the entire gestational sac is valuable.
"Most obstetricians underutilize this. You’re unlikely to miss a yolk sac or embryo, and you’re getting a true report of sac diameter, not a random measurement from a snapshot," she explained at the annual meeting of the Society of Ob/Gyn Hospitalists.
Worrisome but nondefinitive findings suggestive of early pregnancy loss include a slow fetal heart rate, an unusual appearance of the uterine lining, and a sac that is small, grossly distorted, enlarged, irregularly contoured, or low in position.
Maternal gestational age is a key consideration in defining a slow fetal heart rate by M-mode ultrasound. At a menstrual age of 6.2 weeks or less, a normal fetal heart rate is 100 bpm or more; less than 90 bpm is considered slow. In contrast, at 6.3-7.0 weeks, normal is defined as 120 bpm or more, and a fetal heart rate of less than 110 bpm is considered slow. When a slow fetal heart rate is detected at 6.0-7.0 weeks, the risk of subsequent first-trimester fetal demise remains elevated at about 25% even if the heart rate is normal at follow-up at 8.0 weeks (Radiology;2005;236:643-6).
The absolute ultrasound criteria for early pregnancy failure used at the Women and Infants Hospital as well as in many other settings are no fetal heart beat in an embryo more than 5 mm in crown-rump length, or menstrual age known to be greater than 6.5 weeks with no heart beat.
However, a group of investigators led by Dr. Yazan Abdallah of Imperial College London has argued that current definitions used to diagnose early pregnancy failure are potentially unsafe and could result in inadvertent termination of wanted pregnancies. Given the inherent inter- and intraobserver variation in ultrasound measurements, they have urged more conservative criteria for the definitive diagnosis of early pregnancy failure: specifically, a crown-rump length cutoff of greater than 7 mm instead of the widely used 5 mm, and a mean gestational sac diameter cutoff of more than 25 mm.
Dr. Abdallah and his coworkers support their argument on the basis of their observational, prospective cross-sectional study involving 1,060 consecutive women diagnosed with intrauterine pregnancy of uncertain viability. This diagnosis was based upon a symptom-generated ultrasound that showed either an empty gestational sac; a gestational sac with a yolk sac but no embryo when the mean gestational sac diameter was either less than 20 mm or less than 30 mm; or an embryo with an absent heart beat and a crown-rump length of either less than 6 mm or less than 8 mm.
The primary endpoint was a viable pregnancy upon routine first-trimester screening ultrasound at 11-14 weeks. At that point, 473 of the women had viable pregnancies and 587 did not.
When neither the yolk sac nor the embryo was visualized on the initial ultrasound, the false-positive rate for diagnosis of early pregnancy failure was 4.4% when a mean gestational sac diameter of 16 mm was used as a cutoff and 0.5% when 20 mm was the cutoff. Only when a cutoff of 21 mm was utilized did the false-positive rate fall to 0.
If a yolk sac was present but an embryo wasn’t, the false-positive rate was 2.6% with a mean gestational sac diameter cutoff of 16 mm and 0.4% with a cutoff of 20 mm. Again, there were no false positives when a cutoff of 21 mm was employed.
When a yolk sac and embryo were visible but a fetal heartbeat was not apparent, the false-positive rate for miscarriage was 8.3% with a crown-rump length cutoff of 5 mm. At a cutoff of 5.3 mm, there were no false-positive results (Ultrasound Obstet. Gynecol. 2011;38:497-502).
When Dr. Vrees asked the audience for a show of hands as to who utilizes a mean gestational sac diameter cutoff of 21 mm to define early pregnancy loss in the absence of both a yolk sac and embryo, only a couple of ob.gyns. responded affirmatively. Some audience members indicated they use a cutoff as low as 16 mm.
"We see great variability at our institution, too, in the definition of early pregnancy failure based upon mean gestational sac diameter," according to Dr. Vrees.
The lack of unanimity on this point, coupled with the remote likelihood of physical harm in waiting 7-10 days to repeat an ultrasound scan, figure prominently in her advocacy of expectant management.
She reported having no relevant financial disclosures.
EXPERT ANALYSIS FROM THE SOGH ANNUAL CLINICAL MEETING
Study shows no benefit with cesarean section for twins
No significant differences were seen in the risk of adverse neonatal outcomes between women with twin pregnancy who underwent planned cesarean delivery and those who underwent planned vaginal delivery as part of the randomized Twin Birth Study.
The rate of a composite outcome of fetal or neonatal death or serious neonatal morbidity among 1,393 women in 25 countries assigned to undergo planned cesarean delivery was 2.2%, compared with 1.9% among 1,393 women assigned to planned vaginal delivery (odds ratio with planned cesarean delivery, 1.16), Dr. Jon F.R. Barrett of Sunnybrook Health Sciences Centre, Toronto, and his colleagues reported on behalf of the Twin Birth Study Collaborative Group.
Another outcome – a composite of maternal death or serious maternal morbidity – also did not differ between the groups, occurring in 7.3% of the cesarean group and 8.5% of the vaginal delivery group. This finding, however, may be explained in part by the high rate of cesarean section in the planned vaginal delivery group, they said (N. Engl. J. Med. 2013;369:1295-305).
Secondary outcomes, including death or a poor neurodevelopmental outcome among the children at 2 years and problematic incontinence among the mothers at 2 years, will be reported subsequently.
Participants in the Twin Birth Study were women between 32 weeks 0 days gestation and 38 weeks 6 days gestation between Dec. 13, 2003, and April 4, 2011, who were pregnant with twins with an estimated weight of between 1,500 g and 4,000 g each and whose first twin was in the cephalic presentation. The women were randomized to planned cesarean delivery or vaginal delivery with cesarean only if indicated and were followed until 28 days after delivery.
Most (90.7%) in the cesarean delivery group delivered by cesarean as planned, and 43.8% in the planned vaginal delivery group required cesarean delivery, the investigators said
The findings are of note because the twin pregnancy rate has increased over time, largely due to assisted reproductive technologies, and the rate of cesarean section for twins also has increased in the wake of studies suggesting a possible benefit of cesarean delivery with respect to perinatal outcomes.
"There are several possible reasons why our results differ from previous observational data: we avoided selection bias, we ensured the presence of an experienced obstetrician at delivery, and many of the twins in our study were born preterm," the investigators said, adding that no significant interaction between treatment group and baseline variables was seen, which suggests that no significant benefit occurred with planned cesarean delivery for any subgroup tested.
"However, our study was not powered for these subgroup analyses. Further study may be warranted for the gestational-age subgroup of 37 to 38 weeks, particularly given the limited number of infants in this subgroup," they said.
Overall, the results of this study are consistent with no more than a 23% reduction and no more than a 74% increase in the odds of fetal or neonatal death or serious neonatal morbidity with planned cesarean vs. planned vaginal delivery, they said.
"In conclusion, we found no benefits of planned cesarean section, as compared with planned vaginal delivery, for the delivery of twins between 32 and 38 weeks of gestation, if the first twin was in the cephalic presentation," they wrote.
This study was supported by a grant from the Canadian Institutes of Health Research. The authors reported having no relevant conflicts of interest.
Although the Twin Birth Study was a "massive logistic effort," the findings are unlikely to lead to a major change in the use of cesarean delivery for twin pregnancies, according to Dr. Michael F. Greene.
For one thing, the results do not indicate that all sets of twin should be delivered vaginally, he said.
"Obstetricians exercising their best clinical judgment delivered both twins by cesarean section in nearly 40% of the women assigned to planned vaginal delivery, which undoubtedly contributed to the salutary outcomes," he said, noting that the results nonetheless suggest that "a plan to deliver appropriately selected sets of twins vaginally is a reasonably safe choice in skilled hands."
Still, are the results likely to "bend the cesarean delivery curve for twin pregnancies?" he asked.
"Given the trends in patient demographic characteristics and preferences, the virtual disappearance of vaginal delivery in cases of breech presentation, and the dramatic reduction in instrumented vaginal delivery (and the associated gradual disappearance of the skills necessary to perform these procedures among obstetricians), it seems unlikely that we will see a major change in use of cesarean delivery for twins nationwide," he said.
Dr. Greene is an ob.gyn. at Massachusetts General Hospital, Boston, and an associate editor for the New England Journal of Medicine. He reported having no other disclosures. Dr. Greene’s remarks were in an editorial in response to Dr. Barrett and his colleagues’ study (N. Engl. J. Med. 2013;369:1365-6).
Although the Twin Birth Study was a "massive logistic effort," the findings are unlikely to lead to a major change in the use of cesarean delivery for twin pregnancies, according to Dr. Michael F. Greene.
For one thing, the results do not indicate that all sets of twin should be delivered vaginally, he said.
"Obstetricians exercising their best clinical judgment delivered both twins by cesarean section in nearly 40% of the women assigned to planned vaginal delivery, which undoubtedly contributed to the salutary outcomes," he said, noting that the results nonetheless suggest that "a plan to deliver appropriately selected sets of twins vaginally is a reasonably safe choice in skilled hands."
Still, are the results likely to "bend the cesarean delivery curve for twin pregnancies?" he asked.
"Given the trends in patient demographic characteristics and preferences, the virtual disappearance of vaginal delivery in cases of breech presentation, and the dramatic reduction in instrumented vaginal delivery (and the associated gradual disappearance of the skills necessary to perform these procedures among obstetricians), it seems unlikely that we will see a major change in use of cesarean delivery for twins nationwide," he said.
Dr. Greene is an ob.gyn. at Massachusetts General Hospital, Boston, and an associate editor for the New England Journal of Medicine. He reported having no other disclosures. Dr. Greene’s remarks were in an editorial in response to Dr. Barrett and his colleagues’ study (N. Engl. J. Med. 2013;369:1365-6).
Although the Twin Birth Study was a "massive logistic effort," the findings are unlikely to lead to a major change in the use of cesarean delivery for twin pregnancies, according to Dr. Michael F. Greene.
For one thing, the results do not indicate that all sets of twin should be delivered vaginally, he said.
"Obstetricians exercising their best clinical judgment delivered both twins by cesarean section in nearly 40% of the women assigned to planned vaginal delivery, which undoubtedly contributed to the salutary outcomes," he said, noting that the results nonetheless suggest that "a plan to deliver appropriately selected sets of twins vaginally is a reasonably safe choice in skilled hands."
Still, are the results likely to "bend the cesarean delivery curve for twin pregnancies?" he asked.
"Given the trends in patient demographic characteristics and preferences, the virtual disappearance of vaginal delivery in cases of breech presentation, and the dramatic reduction in instrumented vaginal delivery (and the associated gradual disappearance of the skills necessary to perform these procedures among obstetricians), it seems unlikely that we will see a major change in use of cesarean delivery for twins nationwide," he said.
Dr. Greene is an ob.gyn. at Massachusetts General Hospital, Boston, and an associate editor for the New England Journal of Medicine. He reported having no other disclosures. Dr. Greene’s remarks were in an editorial in response to Dr. Barrett and his colleagues’ study (N. Engl. J. Med. 2013;369:1365-6).
No significant differences were seen in the risk of adverse neonatal outcomes between women with twin pregnancy who underwent planned cesarean delivery and those who underwent planned vaginal delivery as part of the randomized Twin Birth Study.
The rate of a composite outcome of fetal or neonatal death or serious neonatal morbidity among 1,393 women in 25 countries assigned to undergo planned cesarean delivery was 2.2%, compared with 1.9% among 1,393 women assigned to planned vaginal delivery (odds ratio with planned cesarean delivery, 1.16), Dr. Jon F.R. Barrett of Sunnybrook Health Sciences Centre, Toronto, and his colleagues reported on behalf of the Twin Birth Study Collaborative Group.
Another outcome – a composite of maternal death or serious maternal morbidity – also did not differ between the groups, occurring in 7.3% of the cesarean group and 8.5% of the vaginal delivery group. This finding, however, may be explained in part by the high rate of cesarean section in the planned vaginal delivery group, they said (N. Engl. J. Med. 2013;369:1295-305).
Secondary outcomes, including death or a poor neurodevelopmental outcome among the children at 2 years and problematic incontinence among the mothers at 2 years, will be reported subsequently.
Participants in the Twin Birth Study were women between 32 weeks 0 days gestation and 38 weeks 6 days gestation between Dec. 13, 2003, and April 4, 2011, who were pregnant with twins with an estimated weight of between 1,500 g and 4,000 g each and whose first twin was in the cephalic presentation. The women were randomized to planned cesarean delivery or vaginal delivery with cesarean only if indicated and were followed until 28 days after delivery.
Most (90.7%) in the cesarean delivery group delivered by cesarean as planned, and 43.8% in the planned vaginal delivery group required cesarean delivery, the investigators said
The findings are of note because the twin pregnancy rate has increased over time, largely due to assisted reproductive technologies, and the rate of cesarean section for twins also has increased in the wake of studies suggesting a possible benefit of cesarean delivery with respect to perinatal outcomes.
"There are several possible reasons why our results differ from previous observational data: we avoided selection bias, we ensured the presence of an experienced obstetrician at delivery, and many of the twins in our study were born preterm," the investigators said, adding that no significant interaction between treatment group and baseline variables was seen, which suggests that no significant benefit occurred with planned cesarean delivery for any subgroup tested.
"However, our study was not powered for these subgroup analyses. Further study may be warranted for the gestational-age subgroup of 37 to 38 weeks, particularly given the limited number of infants in this subgroup," they said.
Overall, the results of this study are consistent with no more than a 23% reduction and no more than a 74% increase in the odds of fetal or neonatal death or serious neonatal morbidity with planned cesarean vs. planned vaginal delivery, they said.
"In conclusion, we found no benefits of planned cesarean section, as compared with planned vaginal delivery, for the delivery of twins between 32 and 38 weeks of gestation, if the first twin was in the cephalic presentation," they wrote.
This study was supported by a grant from the Canadian Institutes of Health Research. The authors reported having no relevant conflicts of interest.
No significant differences were seen in the risk of adverse neonatal outcomes between women with twin pregnancy who underwent planned cesarean delivery and those who underwent planned vaginal delivery as part of the randomized Twin Birth Study.
The rate of a composite outcome of fetal or neonatal death or serious neonatal morbidity among 1,393 women in 25 countries assigned to undergo planned cesarean delivery was 2.2%, compared with 1.9% among 1,393 women assigned to planned vaginal delivery (odds ratio with planned cesarean delivery, 1.16), Dr. Jon F.R. Barrett of Sunnybrook Health Sciences Centre, Toronto, and his colleagues reported on behalf of the Twin Birth Study Collaborative Group.
Another outcome – a composite of maternal death or serious maternal morbidity – also did not differ between the groups, occurring in 7.3% of the cesarean group and 8.5% of the vaginal delivery group. This finding, however, may be explained in part by the high rate of cesarean section in the planned vaginal delivery group, they said (N. Engl. J. Med. 2013;369:1295-305).
Secondary outcomes, including death or a poor neurodevelopmental outcome among the children at 2 years and problematic incontinence among the mothers at 2 years, will be reported subsequently.
Participants in the Twin Birth Study were women between 32 weeks 0 days gestation and 38 weeks 6 days gestation between Dec. 13, 2003, and April 4, 2011, who were pregnant with twins with an estimated weight of between 1,500 g and 4,000 g each and whose first twin was in the cephalic presentation. The women were randomized to planned cesarean delivery or vaginal delivery with cesarean only if indicated and were followed until 28 days after delivery.
Most (90.7%) in the cesarean delivery group delivered by cesarean as planned, and 43.8% in the planned vaginal delivery group required cesarean delivery, the investigators said
The findings are of note because the twin pregnancy rate has increased over time, largely due to assisted reproductive technologies, and the rate of cesarean section for twins also has increased in the wake of studies suggesting a possible benefit of cesarean delivery with respect to perinatal outcomes.
"There are several possible reasons why our results differ from previous observational data: we avoided selection bias, we ensured the presence of an experienced obstetrician at delivery, and many of the twins in our study were born preterm," the investigators said, adding that no significant interaction between treatment group and baseline variables was seen, which suggests that no significant benefit occurred with planned cesarean delivery for any subgroup tested.
"However, our study was not powered for these subgroup analyses. Further study may be warranted for the gestational-age subgroup of 37 to 38 weeks, particularly given the limited number of infants in this subgroup," they said.
Overall, the results of this study are consistent with no more than a 23% reduction and no more than a 74% increase in the odds of fetal or neonatal death or serious neonatal morbidity with planned cesarean vs. planned vaginal delivery, they said.
"In conclusion, we found no benefits of planned cesarean section, as compared with planned vaginal delivery, for the delivery of twins between 32 and 38 weeks of gestation, if the first twin was in the cephalic presentation," they wrote.
This study was supported by a grant from the Canadian Institutes of Health Research. The authors reported having no relevant conflicts of interest.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Major finding: The rates of adverse neonatal outcome with cesarean vs. vaginal delivery were similar (2.2% and 1.9%, respectively).
Data source: The randomized Twin Birth Study, involving 2,804 women.
Disclosures: This study was supported by a grant from the Canadian Institutes of Health Research. The authors reported having no relevant conflicts of interest.
CDC urges flu vaccination for all, especially pregnant women
In an appeal for increased vaccination against influenza, the Centers for Disease Control and Prevention (CDC) announced that, although vaccination coverage has increased for some groups of the population, the nation needs to do better. Recommendations from the Advisory Committee on Immunization Practices reported seasonal influenza vaccination data in the CDC’s Morbidity and Mortality Weekly Report (MMWR) on September 20, 2013.1
More children aged 6 months through 17 years received the influenza vaccine during the 2012–2013 season (56.6%), up 5.1% from the 2011–2012 season. Smaller increases were reported for adults aged 18 years and older at 41.5%, up 2.7% from 2011–2012. Overall, 45% of the entire US population aged 6 months and older was vaccinated in 2012–2013. However, this falls short of public health goals.2
Howard K. Koh, MD, Assistant Secretary for Health at the US Department of Health and Human Services, pointed out that, although the increases in number of those who received the influenza vaccine last year is significant, there is more work to do. “Despite substantial progress, we can do even more to make our country healthier through prevention. Flu vaccination should represent a simple investment we make year in and year out to maximize the gift of health.”2
What vaccines will be available for the 2013-2014 season?
The 2013-2014 influenza vaccine is detailed in MMWR1:
Risk of complications from influenza
Mothers. Vaccination to prevent influenza is particularly important for those who are at increased risk for severe complications from influenza. Pregnant and postpartum women are at higher risk for severe illness and complications from influenza than women who are not pregnant because of changes in the immune system, heart, and lungs during pregnancy.1
Increased severity of influenza among pregnant women was reported during pandemics in 1918–1919, 1957–1958, and 2009–2010. Severe infections were also observed among postpartum (within 2 weeks after delivery) women during the 2009–2010 pandemic.1
One study by Cox et al indicated an increase in delivery complications, including fetal distress, preterm labor, and cesarean delivery, for pregnant women with respiratory hospitalizations during the flu season.1
Vaccination coverage has increased in pregnant women in the last few years, but, unlike other groups where rates continue to increase, coverage in pregnant women appears to have stalled at around 50%.2
Pregnant women who were treated with antivirals more than 4 days after symptoms arose were more likely to be admitted to an ICU (57% vs 9%; 95% confidence interval [CI] 3.5–10.6] than those treated within 2 days on symptom onset.1
Infants. Pregnant women have protective levels of anti-influenza antibodies after vaccination that are passively transferred to their children. In a randomized controlled trial of vaccinated pregnant women who later breastfed their children, infants of vaccinated women had a 63% reduction in laboratory-confirmed influenza during the first 6 months of life. In addition, authors Zaman et al reported a 29% reduction in respiratory illness with fever among the infants and a 36% reduction in respiratory illness with fever among their mothers during the first 6 months after birth.1
Another study by Eick et al found that infants aged less than 6 months who were born to women who were given influenza vaccination during pregnancy had significantly reduced risk for influenza virus infection (relative risk [RR]: 0.59; 95% CI = 0.37–0.93) and hospitalization for influenza-like illness (ILI) (RR: 0.61; 95% CI = 0.45–0.84).1
Infants born to women with laboratory-confirmed flu during pregnancy did not have higher rates of low birthweight, congenital abnormalities, or lower Apgar scores compared with infants born to uninfected women.1
ACOG recommendations for pregnant and postpartum women
Because of the increased risk for serious illness and complications from influenza, the American College of Obstetricians and Gynecologists (ACOG) recommends that all women who are pregnant or who might be pregnant in the upcoming influenza season receive the IIV vaccine. This vaccination can be administered at any time during pregnancy, before and during the influenza season.3
LAIV is not recommended for pregnant women. Postpartum women can receive either LAIV or IIV. Pregnant and postpartum women do not need to avoid contact with persons recently vaccinated with LAIV.1
Recommendations for nonpregnant women
The CDC continues to recommend annual influenza vaccination for all persons ages 6 months or older. Among healthy younger adults, seasonal influenza is typically less acute and results less often in hospitalization, as compared with children aged <5 years, adults aged ≥65 years, pregnant women, or persons with chronic medical conditions. The flu is a significant reason for outpatient medical visits and worker absenteeism among healthy adults aged 19 through 49 years.1
For specific guidelines, visit the CDC’s “Influenza Vaccination: A Summary for Clinicians” at http://www.cdc.gov/flu/professionals/vaccination/vax-summary.htm.
What can you, as a physician, do?
Health-care professionals are critical to the vaccination program and need to lead by example. “If you are around people at high risk for flu complications, you need to get vaccinated,” said Anne Schuchat, MD, Assistant Surgeon General, US Publish Health Service, and Director of the National Center for Immunization and Respiratory Diseases at the CDC.2
The CDC reported that 72% of health-care personal received a vaccination in 2012–2013. Physicians had the highest coverage at 92.3%.2
“Patients also look to you,” said Dr. Schuchat. “Data show that a personal recommendation makes a big difference to patients.”2
For example, pregnant women whose physicians recommend flu vaccination are five times more likely to get vaccinated. Dr. Schuchat also reminded healthcare professionals that they should begin vaccinating patients as soon as vaccine is available in their area.
- Grahskopf LA, Shay DK, Shimabukuro TT, et al; Advisory Committee on Immunization Practices. Prevention and control of seasonal influenza with Vaccines. Centers for Disease Control and Prevention. MMWR Morb Mortal Wkly Rep. 2013;62(RR07):1–43. http://www.cdc.gov/mmwr/preview/mmwrhtml/rr6207a1.htm?s_cid=rr6207a1_w. Accessed September 26, 2013.
- Flu vaccination coverage on the rise; health officials urge continued vigilance [press release] (Susan Heins, e-mail communication, September 26, 2013).
- Committee of Obstetric Practice; American College of Obstetricians and Gynecologists. Committee Opinion No. 468: Influenza vaccination during pregnancy. Obstet Gynecol. 2010;116(4):1006–1007.
In an appeal for increased vaccination against influenza, the Centers for Disease Control and Prevention (CDC) announced that, although vaccination coverage has increased for some groups of the population, the nation needs to do better. Recommendations from the Advisory Committee on Immunization Practices reported seasonal influenza vaccination data in the CDC’s Morbidity and Mortality Weekly Report (MMWR) on September 20, 2013.1
More children aged 6 months through 17 years received the influenza vaccine during the 2012–2013 season (56.6%), up 5.1% from the 2011–2012 season. Smaller increases were reported for adults aged 18 years and older at 41.5%, up 2.7% from 2011–2012. Overall, 45% of the entire US population aged 6 months and older was vaccinated in 2012–2013. However, this falls short of public health goals.2
Howard K. Koh, MD, Assistant Secretary for Health at the US Department of Health and Human Services, pointed out that, although the increases in number of those who received the influenza vaccine last year is significant, there is more work to do. “Despite substantial progress, we can do even more to make our country healthier through prevention. Flu vaccination should represent a simple investment we make year in and year out to maximize the gift of health.”2
What vaccines will be available for the 2013-2014 season?
The 2013-2014 influenza vaccine is detailed in MMWR1:
Risk of complications from influenza
Mothers. Vaccination to prevent influenza is particularly important for those who are at increased risk for severe complications from influenza. Pregnant and postpartum women are at higher risk for severe illness and complications from influenza than women who are not pregnant because of changes in the immune system, heart, and lungs during pregnancy.1
Increased severity of influenza among pregnant women was reported during pandemics in 1918–1919, 1957–1958, and 2009–2010. Severe infections were also observed among postpartum (within 2 weeks after delivery) women during the 2009–2010 pandemic.1
One study by Cox et al indicated an increase in delivery complications, including fetal distress, preterm labor, and cesarean delivery, for pregnant women with respiratory hospitalizations during the flu season.1
Vaccination coverage has increased in pregnant women in the last few years, but, unlike other groups where rates continue to increase, coverage in pregnant women appears to have stalled at around 50%.2
Pregnant women who were treated with antivirals more than 4 days after symptoms arose were more likely to be admitted to an ICU (57% vs 9%; 95% confidence interval [CI] 3.5–10.6] than those treated within 2 days on symptom onset.1
Infants. Pregnant women have protective levels of anti-influenza antibodies after vaccination that are passively transferred to their children. In a randomized controlled trial of vaccinated pregnant women who later breastfed their children, infants of vaccinated women had a 63% reduction in laboratory-confirmed influenza during the first 6 months of life. In addition, authors Zaman et al reported a 29% reduction in respiratory illness with fever among the infants and a 36% reduction in respiratory illness with fever among their mothers during the first 6 months after birth.1
Another study by Eick et al found that infants aged less than 6 months who were born to women who were given influenza vaccination during pregnancy had significantly reduced risk for influenza virus infection (relative risk [RR]: 0.59; 95% CI = 0.37–0.93) and hospitalization for influenza-like illness (ILI) (RR: 0.61; 95% CI = 0.45–0.84).1
Infants born to women with laboratory-confirmed flu during pregnancy did not have higher rates of low birthweight, congenital abnormalities, or lower Apgar scores compared with infants born to uninfected women.1
ACOG recommendations for pregnant and postpartum women
Because of the increased risk for serious illness and complications from influenza, the American College of Obstetricians and Gynecologists (ACOG) recommends that all women who are pregnant or who might be pregnant in the upcoming influenza season receive the IIV vaccine. This vaccination can be administered at any time during pregnancy, before and during the influenza season.3
LAIV is not recommended for pregnant women. Postpartum women can receive either LAIV or IIV. Pregnant and postpartum women do not need to avoid contact with persons recently vaccinated with LAIV.1
Recommendations for nonpregnant women
The CDC continues to recommend annual influenza vaccination for all persons ages 6 months or older. Among healthy younger adults, seasonal influenza is typically less acute and results less often in hospitalization, as compared with children aged <5 years, adults aged ≥65 years, pregnant women, or persons with chronic medical conditions. The flu is a significant reason for outpatient medical visits and worker absenteeism among healthy adults aged 19 through 49 years.1
For specific guidelines, visit the CDC’s “Influenza Vaccination: A Summary for Clinicians” at http://www.cdc.gov/flu/professionals/vaccination/vax-summary.htm.
What can you, as a physician, do?
Health-care professionals are critical to the vaccination program and need to lead by example. “If you are around people at high risk for flu complications, you need to get vaccinated,” said Anne Schuchat, MD, Assistant Surgeon General, US Publish Health Service, and Director of the National Center for Immunization and Respiratory Diseases at the CDC.2
The CDC reported that 72% of health-care personal received a vaccination in 2012–2013. Physicians had the highest coverage at 92.3%.2
“Patients also look to you,” said Dr. Schuchat. “Data show that a personal recommendation makes a big difference to patients.”2
For example, pregnant women whose physicians recommend flu vaccination are five times more likely to get vaccinated. Dr. Schuchat also reminded healthcare professionals that they should begin vaccinating patients as soon as vaccine is available in their area.
In an appeal for increased vaccination against influenza, the Centers for Disease Control and Prevention (CDC) announced that, although vaccination coverage has increased for some groups of the population, the nation needs to do better. Recommendations from the Advisory Committee on Immunization Practices reported seasonal influenza vaccination data in the CDC’s Morbidity and Mortality Weekly Report (MMWR) on September 20, 2013.1
More children aged 6 months through 17 years received the influenza vaccine during the 2012–2013 season (56.6%), up 5.1% from the 2011–2012 season. Smaller increases were reported for adults aged 18 years and older at 41.5%, up 2.7% from 2011–2012. Overall, 45% of the entire US population aged 6 months and older was vaccinated in 2012–2013. However, this falls short of public health goals.2
Howard K. Koh, MD, Assistant Secretary for Health at the US Department of Health and Human Services, pointed out that, although the increases in number of those who received the influenza vaccine last year is significant, there is more work to do. “Despite substantial progress, we can do even more to make our country healthier through prevention. Flu vaccination should represent a simple investment we make year in and year out to maximize the gift of health.”2
What vaccines will be available for the 2013-2014 season?
The 2013-2014 influenza vaccine is detailed in MMWR1:
Risk of complications from influenza
Mothers. Vaccination to prevent influenza is particularly important for those who are at increased risk for severe complications from influenza. Pregnant and postpartum women are at higher risk for severe illness and complications from influenza than women who are not pregnant because of changes in the immune system, heart, and lungs during pregnancy.1
Increased severity of influenza among pregnant women was reported during pandemics in 1918–1919, 1957–1958, and 2009–2010. Severe infections were also observed among postpartum (within 2 weeks after delivery) women during the 2009–2010 pandemic.1
One study by Cox et al indicated an increase in delivery complications, including fetal distress, preterm labor, and cesarean delivery, for pregnant women with respiratory hospitalizations during the flu season.1
Vaccination coverage has increased in pregnant women in the last few years, but, unlike other groups where rates continue to increase, coverage in pregnant women appears to have stalled at around 50%.2
Pregnant women who were treated with antivirals more than 4 days after symptoms arose were more likely to be admitted to an ICU (57% vs 9%; 95% confidence interval [CI] 3.5–10.6] than those treated within 2 days on symptom onset.1
Infants. Pregnant women have protective levels of anti-influenza antibodies after vaccination that are passively transferred to their children. In a randomized controlled trial of vaccinated pregnant women who later breastfed their children, infants of vaccinated women had a 63% reduction in laboratory-confirmed influenza during the first 6 months of life. In addition, authors Zaman et al reported a 29% reduction in respiratory illness with fever among the infants and a 36% reduction in respiratory illness with fever among their mothers during the first 6 months after birth.1
Another study by Eick et al found that infants aged less than 6 months who were born to women who were given influenza vaccination during pregnancy had significantly reduced risk for influenza virus infection (relative risk [RR]: 0.59; 95% CI = 0.37–0.93) and hospitalization for influenza-like illness (ILI) (RR: 0.61; 95% CI = 0.45–0.84).1
Infants born to women with laboratory-confirmed flu during pregnancy did not have higher rates of low birthweight, congenital abnormalities, or lower Apgar scores compared with infants born to uninfected women.1
ACOG recommendations for pregnant and postpartum women
Because of the increased risk for serious illness and complications from influenza, the American College of Obstetricians and Gynecologists (ACOG) recommends that all women who are pregnant or who might be pregnant in the upcoming influenza season receive the IIV vaccine. This vaccination can be administered at any time during pregnancy, before and during the influenza season.3
LAIV is not recommended for pregnant women. Postpartum women can receive either LAIV or IIV. Pregnant and postpartum women do not need to avoid contact with persons recently vaccinated with LAIV.1
Recommendations for nonpregnant women
The CDC continues to recommend annual influenza vaccination for all persons ages 6 months or older. Among healthy younger adults, seasonal influenza is typically less acute and results less often in hospitalization, as compared with children aged <5 years, adults aged ≥65 years, pregnant women, or persons with chronic medical conditions. The flu is a significant reason for outpatient medical visits and worker absenteeism among healthy adults aged 19 through 49 years.1
For specific guidelines, visit the CDC’s “Influenza Vaccination: A Summary for Clinicians” at http://www.cdc.gov/flu/professionals/vaccination/vax-summary.htm.
What can you, as a physician, do?
Health-care professionals are critical to the vaccination program and need to lead by example. “If you are around people at high risk for flu complications, you need to get vaccinated,” said Anne Schuchat, MD, Assistant Surgeon General, US Publish Health Service, and Director of the National Center for Immunization and Respiratory Diseases at the CDC.2
The CDC reported that 72% of health-care personal received a vaccination in 2012–2013. Physicians had the highest coverage at 92.3%.2
“Patients also look to you,” said Dr. Schuchat. “Data show that a personal recommendation makes a big difference to patients.”2
For example, pregnant women whose physicians recommend flu vaccination are five times more likely to get vaccinated. Dr. Schuchat also reminded healthcare professionals that they should begin vaccinating patients as soon as vaccine is available in their area.
- Grahskopf LA, Shay DK, Shimabukuro TT, et al; Advisory Committee on Immunization Practices. Prevention and control of seasonal influenza with Vaccines. Centers for Disease Control and Prevention. MMWR Morb Mortal Wkly Rep. 2013;62(RR07):1–43. http://www.cdc.gov/mmwr/preview/mmwrhtml/rr6207a1.htm?s_cid=rr6207a1_w. Accessed September 26, 2013.
- Flu vaccination coverage on the rise; health officials urge continued vigilance [press release] (Susan Heins, e-mail communication, September 26, 2013).
- Committee of Obstetric Practice; American College of Obstetricians and Gynecologists. Committee Opinion No. 468: Influenza vaccination during pregnancy. Obstet Gynecol. 2010;116(4):1006–1007.
- Grahskopf LA, Shay DK, Shimabukuro TT, et al; Advisory Committee on Immunization Practices. Prevention and control of seasonal influenza with Vaccines. Centers for Disease Control and Prevention. MMWR Morb Mortal Wkly Rep. 2013;62(RR07):1–43. http://www.cdc.gov/mmwr/preview/mmwrhtml/rr6207a1.htm?s_cid=rr6207a1_w. Accessed September 26, 2013.
- Flu vaccination coverage on the rise; health officials urge continued vigilance [press release] (Susan Heins, e-mail communication, September 26, 2013).
- Committee of Obstetric Practice; American College of Obstetricians and Gynecologists. Committee Opinion No. 468: Influenza vaccination during pregnancy. Obstet Gynecol. 2010;116(4):1006–1007.
Gestational diabetes linked to sleep apnea
Women with gestational diabetes are nearly seven times more likely to have sleep apnea and to sleep an average of one hour less nightly than do expectant mothers without the condition, a small observational study has shown.
"It is common for pregnant women to experience sleep disruptions, but the risk of developing obstructive sleep apnea increases substantially in women who have gestational diabetes," Dr. Sirimon Reutrakul of Rush University Medical Center in Chicago, said in a statement. "Nearly 75% of the participants in our study who had gestational diabetes also suffered from obstructive sleep apnea."
Reports in the literature link sleep apnea in pregnancy to complications such as preeclampsia, hypertension, low birth weights, preterm delivery, and other pregnancy-related adverse outcomes.
Use of continuous positive airway pressure (CPAP) early in pregnancy for women with hypertension and chronic snoring was noted by the investigators to be associated with better blood pressure control and pregnancy outcomes. CPAP treatment in nonpregnant type 2 diabetes patients with sleep apnea has been shown occasionally effective in improving glucose control. Because there are currently no data on the effects of sleep apnea treatment in gestational diabetes, whether CPAP treatment might affect glucose metabolism and pregnancy outcomes is unknown.
Dr. Reutrakul and his associates cited previous studies that increased sleep apnea leads to poor glycemic control, as well as reports that sleep apnea is a major risk factor for insulin resistance regardless of body mass index (BMI). With these data in mind, the investigators compared metabolic and sleep apnea measures in 15 pregnant women who did not have gestational diabetes; 15 pregnant women with gestational diabetes; and 15 obese controls (BMI = 31.0 +/– 4.3 kg/m2) who were neither pregnant nor diabetic. The groups were matched for age, race, and in the pregnant groups, and prepregnancy BMI.
All gestating women were expecting singletons and were either in the latter part of their second term, or the early part of their third. The average gestational age was 28.2 +/– 3.7 weeks in the women with gestational diabetes, and 30.9 +/– 2.0 weeks in the pregnant group without.
The women with gestational diabetes had notably higher measures of prepregnancy BMI than did the pregnant women without gestational diabetes. The BMI of the gestational diabetes cohort at the time of the sleep apnea monitoring also tended to be higher. Most of the pregnant women were overweight (BMI 25.0-29.9 kg/m2) or obese (BMI = 30 kg/m2) based on the prepregnancy BMI (93% of those with gestational diabetes; 67% of those without).
The number of apneas and hypopneas per hour of sleep were measured using polysomnography. Sleep apnea was diagnosed in the women if their apnea-hypopnea index (AHI) score was greater than or equal to 5. Dr. Reutrakul and his colleagues wrote that theirs was the first study to use polysomnography to evaluate overall sleep quality, including apnea, in women with gestational diabetes, compared with pregnant women without gestational diabetes, controlling for confounding factors. Wake time after sleep onset equaled the number of minutes participants were awake between sleep onset and end of the session.
There were a number of statistically significant findings:
After they adjusted for prepregnancy BMI, Dr. Reutrakul and his associates found that a diagnosis of gestational diabetes was strongly associated with a diagnosis of sleep apnea (odds ratio, 6.60; 95% confidence interval, 1.15-37.96). The researchers also found that pregnant women with gestational diabetes slept a median average of 1 hour less than the other pregnant women did (397 minutes vs. 464 minutes). The gestational diabetes cohort also had a median AHI approximately four times higher than the pregnant women without gestational diabetes had (8.2 vs. 2.0), and a higher overall rate of sleep apnea than did the nongestational diabetes group (73% vs. 27%). Compared with controls, a higher AHI was found in pregnant women without gestational diabetes (2.0 vs. 0.5), as was more disrupted sleep as reflected by a higher wake time after sleep onset (66 vs. 21 minutes). Their median microarousal index was also higher (16.4 vs. 10.6).
The researchers noted that women with gestational diabetes gained less weight during pregnancy than the pregnant women without gestational diabetes (BMI increased respectively by 2.2 +/– 2.0 vs. 4.6 +/– 1.9 kg/m2), ruling out any "strong association" between gestational weight gain, gestational diabetes, and sleep apnea.
According to the authors, the study also was limited by its cross-sectional design, which does not indicate whether sleep apnea causes gestational diabetes, or vice versa. Regardless, Dr. Reutrakul stated, "Based on these findings, women who have gestational diabetes should be considered for evaluation for obstructive sleep apnea, especially if other risk factors such as hypertension or obesity are present, and women already diagnosed with sleep apnea should be monitored for signs of gestational diabetes during pregnancy."
A member of the research team reported financial ties with Pfizer and other industry-related research grant support. See study for list of disclosures. This study was supported by the ResMed Foundation; the diabetes research training center at the University of Chicago, a specialized center of research on Women’s Health; and the National Institutes of Health.
Women with gestational diabetes are nearly seven times more likely to have sleep apnea and to sleep an average of one hour less nightly than do expectant mothers without the condition, a small observational study has shown.
"It is common for pregnant women to experience sleep disruptions, but the risk of developing obstructive sleep apnea increases substantially in women who have gestational diabetes," Dr. Sirimon Reutrakul of Rush University Medical Center in Chicago, said in a statement. "Nearly 75% of the participants in our study who had gestational diabetes also suffered from obstructive sleep apnea."
Reports in the literature link sleep apnea in pregnancy to complications such as preeclampsia, hypertension, low birth weights, preterm delivery, and other pregnancy-related adverse outcomes.
Use of continuous positive airway pressure (CPAP) early in pregnancy for women with hypertension and chronic snoring was noted by the investigators to be associated with better blood pressure control and pregnancy outcomes. CPAP treatment in nonpregnant type 2 diabetes patients with sleep apnea has been shown occasionally effective in improving glucose control. Because there are currently no data on the effects of sleep apnea treatment in gestational diabetes, whether CPAP treatment might affect glucose metabolism and pregnancy outcomes is unknown.
Dr. Reutrakul and his associates cited previous studies that increased sleep apnea leads to poor glycemic control, as well as reports that sleep apnea is a major risk factor for insulin resistance regardless of body mass index (BMI). With these data in mind, the investigators compared metabolic and sleep apnea measures in 15 pregnant women who did not have gestational diabetes; 15 pregnant women with gestational diabetes; and 15 obese controls (BMI = 31.0 +/– 4.3 kg/m2) who were neither pregnant nor diabetic. The groups were matched for age, race, and in the pregnant groups, and prepregnancy BMI.
All gestating women were expecting singletons and were either in the latter part of their second term, or the early part of their third. The average gestational age was 28.2 +/– 3.7 weeks in the women with gestational diabetes, and 30.9 +/– 2.0 weeks in the pregnant group without.
The women with gestational diabetes had notably higher measures of prepregnancy BMI than did the pregnant women without gestational diabetes. The BMI of the gestational diabetes cohort at the time of the sleep apnea monitoring also tended to be higher. Most of the pregnant women were overweight (BMI 25.0-29.9 kg/m2) or obese (BMI = 30 kg/m2) based on the prepregnancy BMI (93% of those with gestational diabetes; 67% of those without).
The number of apneas and hypopneas per hour of sleep were measured using polysomnography. Sleep apnea was diagnosed in the women if their apnea-hypopnea index (AHI) score was greater than or equal to 5. Dr. Reutrakul and his colleagues wrote that theirs was the first study to use polysomnography to evaluate overall sleep quality, including apnea, in women with gestational diabetes, compared with pregnant women without gestational diabetes, controlling for confounding factors. Wake time after sleep onset equaled the number of minutes participants were awake between sleep onset and end of the session.
There were a number of statistically significant findings:
After they adjusted for prepregnancy BMI, Dr. Reutrakul and his associates found that a diagnosis of gestational diabetes was strongly associated with a diagnosis of sleep apnea (odds ratio, 6.60; 95% confidence interval, 1.15-37.96). The researchers also found that pregnant women with gestational diabetes slept a median average of 1 hour less than the other pregnant women did (397 minutes vs. 464 minutes). The gestational diabetes cohort also had a median AHI approximately four times higher than the pregnant women without gestational diabetes had (8.2 vs. 2.0), and a higher overall rate of sleep apnea than did the nongestational diabetes group (73% vs. 27%). Compared with controls, a higher AHI was found in pregnant women without gestational diabetes (2.0 vs. 0.5), as was more disrupted sleep as reflected by a higher wake time after sleep onset (66 vs. 21 minutes). Their median microarousal index was also higher (16.4 vs. 10.6).
The researchers noted that women with gestational diabetes gained less weight during pregnancy than the pregnant women without gestational diabetes (BMI increased respectively by 2.2 +/– 2.0 vs. 4.6 +/– 1.9 kg/m2), ruling out any "strong association" between gestational weight gain, gestational diabetes, and sleep apnea.
According to the authors, the study also was limited by its cross-sectional design, which does not indicate whether sleep apnea causes gestational diabetes, or vice versa. Regardless, Dr. Reutrakul stated, "Based on these findings, women who have gestational diabetes should be considered for evaluation for obstructive sleep apnea, especially if other risk factors such as hypertension or obesity are present, and women already diagnosed with sleep apnea should be monitored for signs of gestational diabetes during pregnancy."
A member of the research team reported financial ties with Pfizer and other industry-related research grant support. See study for list of disclosures. This study was supported by the ResMed Foundation; the diabetes research training center at the University of Chicago, a specialized center of research on Women’s Health; and the National Institutes of Health.
Women with gestational diabetes are nearly seven times more likely to have sleep apnea and to sleep an average of one hour less nightly than do expectant mothers without the condition, a small observational study has shown.
"It is common for pregnant women to experience sleep disruptions, but the risk of developing obstructive sleep apnea increases substantially in women who have gestational diabetes," Dr. Sirimon Reutrakul of Rush University Medical Center in Chicago, said in a statement. "Nearly 75% of the participants in our study who had gestational diabetes also suffered from obstructive sleep apnea."
Reports in the literature link sleep apnea in pregnancy to complications such as preeclampsia, hypertension, low birth weights, preterm delivery, and other pregnancy-related adverse outcomes.
Use of continuous positive airway pressure (CPAP) early in pregnancy for women with hypertension and chronic snoring was noted by the investigators to be associated with better blood pressure control and pregnancy outcomes. CPAP treatment in nonpregnant type 2 diabetes patients with sleep apnea has been shown occasionally effective in improving glucose control. Because there are currently no data on the effects of sleep apnea treatment in gestational diabetes, whether CPAP treatment might affect glucose metabolism and pregnancy outcomes is unknown.
Dr. Reutrakul and his associates cited previous studies that increased sleep apnea leads to poor glycemic control, as well as reports that sleep apnea is a major risk factor for insulin resistance regardless of body mass index (BMI). With these data in mind, the investigators compared metabolic and sleep apnea measures in 15 pregnant women who did not have gestational diabetes; 15 pregnant women with gestational diabetes; and 15 obese controls (BMI = 31.0 +/– 4.3 kg/m2) who were neither pregnant nor diabetic. The groups were matched for age, race, and in the pregnant groups, and prepregnancy BMI.
All gestating women were expecting singletons and were either in the latter part of their second term, or the early part of their third. The average gestational age was 28.2 +/– 3.7 weeks in the women with gestational diabetes, and 30.9 +/– 2.0 weeks in the pregnant group without.
The women with gestational diabetes had notably higher measures of prepregnancy BMI than did the pregnant women without gestational diabetes. The BMI of the gestational diabetes cohort at the time of the sleep apnea monitoring also tended to be higher. Most of the pregnant women were overweight (BMI 25.0-29.9 kg/m2) or obese (BMI = 30 kg/m2) based on the prepregnancy BMI (93% of those with gestational diabetes; 67% of those without).
The number of apneas and hypopneas per hour of sleep were measured using polysomnography. Sleep apnea was diagnosed in the women if their apnea-hypopnea index (AHI) score was greater than or equal to 5. Dr. Reutrakul and his colleagues wrote that theirs was the first study to use polysomnography to evaluate overall sleep quality, including apnea, in women with gestational diabetes, compared with pregnant women without gestational diabetes, controlling for confounding factors. Wake time after sleep onset equaled the number of minutes participants were awake between sleep onset and end of the session.
There were a number of statistically significant findings:
After they adjusted for prepregnancy BMI, Dr. Reutrakul and his associates found that a diagnosis of gestational diabetes was strongly associated with a diagnosis of sleep apnea (odds ratio, 6.60; 95% confidence interval, 1.15-37.96). The researchers also found that pregnant women with gestational diabetes slept a median average of 1 hour less than the other pregnant women did (397 minutes vs. 464 minutes). The gestational diabetes cohort also had a median AHI approximately four times higher than the pregnant women without gestational diabetes had (8.2 vs. 2.0), and a higher overall rate of sleep apnea than did the nongestational diabetes group (73% vs. 27%). Compared with controls, a higher AHI was found in pregnant women without gestational diabetes (2.0 vs. 0.5), as was more disrupted sleep as reflected by a higher wake time after sleep onset (66 vs. 21 minutes). Their median microarousal index was also higher (16.4 vs. 10.6).
The researchers noted that women with gestational diabetes gained less weight during pregnancy than the pregnant women without gestational diabetes (BMI increased respectively by 2.2 +/– 2.0 vs. 4.6 +/– 1.9 kg/m2), ruling out any "strong association" between gestational weight gain, gestational diabetes, and sleep apnea.
According to the authors, the study also was limited by its cross-sectional design, which does not indicate whether sleep apnea causes gestational diabetes, or vice versa. Regardless, Dr. Reutrakul stated, "Based on these findings, women who have gestational diabetes should be considered for evaluation for obstructive sleep apnea, especially if other risk factors such as hypertension or obesity are present, and women already diagnosed with sleep apnea should be monitored for signs of gestational diabetes during pregnancy."
A member of the research team reported financial ties with Pfizer and other industry-related research grant support. See study for list of disclosures. This study was supported by the ResMed Foundation; the diabetes research training center at the University of Chicago, a specialized center of research on Women’s Health; and the National Institutes of Health.
JOURNAL OF CLINICAL ENDOCRINOLOGY & METABOLISM
Major finding: Women with gestational diabetes are nearly seven times more likely to have sleep apnea and sleep an hour less than other pregnant women do.
Data source: Observational case-control study of 45 women.
Disclosures: A member of the research team reported financial ties with Pfizer and other industry-related research grant support. See study for list of disclosures. This study was supported by the ResMed Foundation; the diabetes research training center at the University of Chicago, a specialized center of research on Women’s Health; and the National Institutes of Health.
Implementing hospital laborist program cut cesarean rates
DENVER – The newly published first data showing improved clinical outcomes after adoption of a full-time hospital laborist program was roundly celebrated at the annual meeting of the Society of Ob/Gyn Hospitalists.
Dr. Thomas J. Garite presented highlights of this freshly published retrospective observational study (Am. J. Obstet. Gynecol. 2013;209:251.e1-6) conducted at a large-delivery-volume tertiary hospital in Las Vegas. Dr. Garite and his coinvestigators, led by Dr. Brian K. Iriye, compared hospital-wide cesarean delivery rates for 6,206 nulliparous, term, singleton live births during 2006-2011.
This was a period of change in how labor and delivery was organized at the hospital. During the first 16 months of the study period, the traditional private-practice model of patient care was in place, with ob.gyns. on call and no laborists in the house. This was followed by a 14-month interlude in which local private-practice ob.gyns. got together and made sure that a community physician was continuously in-hospital to provide laborist coverage.
"I call that the doc-in-a-box model," said Dr. Garite, professor emeritus and former chair of obstetrics and gynecology at the University of California, Irvine.
Finally came a 24-month period with full-time laborists – that is, ob.gyns. without a private practice – providing in-hospital coverage 24/7.
In a multivariate logistic regression analysis adjusted for potential confounders, the hospital’s cesarean section rate was roughly 25% lower after implementation of the full-time laborist program than in either of the other two periods.
"I haven’t seen other studies to date that demonstrate these kinds of outcome advantages for this kind of practice. I think we’re going to see a lot more. But until we do, a lot of people who don’t like change are going to be saying, ‘Wait, where’s the proof?’ Well, this is the beginning of the proof of something I believe in strongly," declared Dr. Garite, who is also editor-in-chief of the American Journal of Obstetrics and Gynecology and chief clinical officer at PeriGen, a provider of fetal surveillance systems.
Society of Ob/Gyn Hospitalists (SOGH) board member Dr. Jennifer Tessmer-Tuck hailed the new study as "the best and almost the only" clinical outcome data to date showing the advantages of the ob.gyn. hospitalist model of care. And it was a long time coming, she noted: a full 10½ years since Dr. Louis Weinstein of the Medical College of Ohio, Toledo, heralded the birth of a radically different form of ob.gyn. practice in his seminal essay "The laborist: A new focus of practice for the obstetrician" (Am. J. Obstet. Gynecol. 2003;188:310-2).
"We have a lot to do. SOGH would really like to have more of a research platform and be able to put ourselves out there. There’s really a gap in care, and we’re hoping to jump in and fill it," said Dr. Tessmer-Tuck, director of North Memorial Medical Center Laborist Associates in Robbinsdale, Minn.
But while the SOGH leadership is eager to see the field assume a bigger research presence, it’s a challenge. Most society members, when they talk about why they became hospitalists, say they had burned out in traditional private practice, with its demanding on-call schedule. They sought well-defined hours, perhaps more family time. Given those priorities, taking on a research project can sound daunting, even though the fruits of such a project might enhance the standing of the young subspecialty.
Dr. Garite reported that the cesarean section rate at the tertiary center was 33.2% during the 24 months when full-time laborists were on hand, compared with 39.2% under the traditional private practice model with no laborists, and 38.7% with laborist coverage by community staff. In a multivariate logistic regression analysis adjusted for maternal age, physician age, race, gestational age, induction of labor, birth weight, and maternal weight, the hospital’s cesarean section rate after the introduction of full-time laborists was 27% lower than in the earlier period of no laborists and 23% less than with community laborist care.
There were no differences between the three groups in rates of low Apgar scores, metabolic acidosis, or any other parameters of adverse neonatal or maternal outcome.
During the study years of 2006-2011, cesarean section rates at the other hospitals in the city were either stable or rising.
Asked why hospital-wide cesarean section rates dropped significantly once full-time obstetric hospitalists were in place, Dr. Garite replied, "It’s not, for example, the patient with abruption who comes in the door; she’s going to get a cesarean section whether a hospitalist is there or some other doctor is covering. Instead, it’s the patient who has what I call ‘failure to wait,’ a.k.a. failure to progress, or the 4 o’clock induction that hasn’t made any progress ... There are lots of examples of why cesarean section rates change with a hospitalist in place, especially if you look at the correlation between cesarean sections and time of day."
Dr. Tessmer-Tuck said she found the Las Vegas study highly relevant because lots of hospitals throughout the country are now going through a similar transition from traditional on-call practice to around-the-clock coverage provided by rotating private practice community laborists, while pondering a possible move to full-time laborists.
"This is where many of our hospitals are at: They’re in the middle phase, with private-practice docs being paid to stay in-house 24 hours in case there’s an emergency," according to Dr. Tessmer-Tuck.
She said she found particularly impressive the investigators’ calculation that a full-time laborist resulted in an average of one fewer cesarean section every 2 days in a population of primiparous, term, singleton patients, with a resultant estimated savings in patient care costs of $2,823-$3,305 per day. Because a laborist might be paid $2,500 per 24-hour shift, the reduced cesarean section rate alone covers the laborist’s salary. Those are the sort of numbers hospital administrators find persuasive.
"This is a message you guys should take home with you when you go back to your own program," she said.
While the Las Vegas study provides the first evidence to be published in a major peer-reviewed journal demonstrating superior clinical outcomes with the full-time laborist model, Dr. Tessmer-Tuck noted that in addition there are several published studies suggesting that hospitals experience fewer adverse events and markedly lower payouts for bad outcomes after they implement multipronged, comprehensive obstetric patient safety programs that include bringing a laborist on board.
"Liability has become a huge issue for us. Many hospitals implement hospitalist programs mainly in order to reduce liability," according to Dr. Tessmer-Tuck.
She cited a study by ob.gyns. at New York Presbyterian/Weill Cornell Medical Center in which they analyzed the impact of a comprehensive patient safety program initiated in stages beginning in 2003. The interventions included mandatory labor and delivery team training aimed at enhancing physician/nurse communication, development of standardized management protocols, training in fetal heart rate monitoring interpretation, creation of a patient safety nurse position, and, in 2006, introduction of a laborist.
It’s not possible to parse out just how much of the improvement in response to the multipronged safety program was the result of adopting the laborist model, Dr. Tessmer-Tuck said, but she noted the average yearly compensation payments for patient claims or lawsuits were $27.6 million during 2003-2006, plummeting to $2.5 million per year in 2007-2009, after the laborist was in place. Moreover, sentinel adverse events such as maternal death or severe neurodevelopmental impairment in a child decreased from five in the year 2000 to none in 2008 and 2009 (Am. J. Obstet. Gynecol. 2011;204:97-105).
Ob.gyns. at Yale–New Haven (Conn.) Hospital introduced a similar comprehensive patient safety program, also including implementation of a 24-hour obstetrics hospitalist, during 2004-2006. During 3 years of prospective follow-up involving nearly 14,000 deliveries, they documented a significant linear decline in obstetric adverse outcomes (Am. J. Obstet. Gynecol. 2009;200:492e1-8). They also administered a validated workplace safety attitude questionnaire four times during 2004-2009 and documented marked improvement over time in favorable scores in the domains of job satisfaction, teamwork, and safety culture (Am J. Obstet. Gynecol. 2011;204:216.e1-6).
Dr. Garite and Dr. Tessmer-Tuck reported having no germane financial relationships.
DENVER – The newly published first data showing improved clinical outcomes after adoption of a full-time hospital laborist program was roundly celebrated at the annual meeting of the Society of Ob/Gyn Hospitalists.
Dr. Thomas J. Garite presented highlights of this freshly published retrospective observational study (Am. J. Obstet. Gynecol. 2013;209:251.e1-6) conducted at a large-delivery-volume tertiary hospital in Las Vegas. Dr. Garite and his coinvestigators, led by Dr. Brian K. Iriye, compared hospital-wide cesarean delivery rates for 6,206 nulliparous, term, singleton live births during 2006-2011.
This was a period of change in how labor and delivery was organized at the hospital. During the first 16 months of the study period, the traditional private-practice model of patient care was in place, with ob.gyns. on call and no laborists in the house. This was followed by a 14-month interlude in which local private-practice ob.gyns. got together and made sure that a community physician was continuously in-hospital to provide laborist coverage.
"I call that the doc-in-a-box model," said Dr. Garite, professor emeritus and former chair of obstetrics and gynecology at the University of California, Irvine.
Finally came a 24-month period with full-time laborists – that is, ob.gyns. without a private practice – providing in-hospital coverage 24/7.
In a multivariate logistic regression analysis adjusted for potential confounders, the hospital’s cesarean section rate was roughly 25% lower after implementation of the full-time laborist program than in either of the other two periods.
"I haven’t seen other studies to date that demonstrate these kinds of outcome advantages for this kind of practice. I think we’re going to see a lot more. But until we do, a lot of people who don’t like change are going to be saying, ‘Wait, where’s the proof?’ Well, this is the beginning of the proof of something I believe in strongly," declared Dr. Garite, who is also editor-in-chief of the American Journal of Obstetrics and Gynecology and chief clinical officer at PeriGen, a provider of fetal surveillance systems.
Society of Ob/Gyn Hospitalists (SOGH) board member Dr. Jennifer Tessmer-Tuck hailed the new study as "the best and almost the only" clinical outcome data to date showing the advantages of the ob.gyn. hospitalist model of care. And it was a long time coming, she noted: a full 10½ years since Dr. Louis Weinstein of the Medical College of Ohio, Toledo, heralded the birth of a radically different form of ob.gyn. practice in his seminal essay "The laborist: A new focus of practice for the obstetrician" (Am. J. Obstet. Gynecol. 2003;188:310-2).
"We have a lot to do. SOGH would really like to have more of a research platform and be able to put ourselves out there. There’s really a gap in care, and we’re hoping to jump in and fill it," said Dr. Tessmer-Tuck, director of North Memorial Medical Center Laborist Associates in Robbinsdale, Minn.
But while the SOGH leadership is eager to see the field assume a bigger research presence, it’s a challenge. Most society members, when they talk about why they became hospitalists, say they had burned out in traditional private practice, with its demanding on-call schedule. They sought well-defined hours, perhaps more family time. Given those priorities, taking on a research project can sound daunting, even though the fruits of such a project might enhance the standing of the young subspecialty.
Dr. Garite reported that the cesarean section rate at the tertiary center was 33.2% during the 24 months when full-time laborists were on hand, compared with 39.2% under the traditional private practice model with no laborists, and 38.7% with laborist coverage by community staff. In a multivariate logistic regression analysis adjusted for maternal age, physician age, race, gestational age, induction of labor, birth weight, and maternal weight, the hospital’s cesarean section rate after the introduction of full-time laborists was 27% lower than in the earlier period of no laborists and 23% less than with community laborist care.
There were no differences between the three groups in rates of low Apgar scores, metabolic acidosis, or any other parameters of adverse neonatal or maternal outcome.
During the study years of 2006-2011, cesarean section rates at the other hospitals in the city were either stable or rising.
Asked why hospital-wide cesarean section rates dropped significantly once full-time obstetric hospitalists were in place, Dr. Garite replied, "It’s not, for example, the patient with abruption who comes in the door; she’s going to get a cesarean section whether a hospitalist is there or some other doctor is covering. Instead, it’s the patient who has what I call ‘failure to wait,’ a.k.a. failure to progress, or the 4 o’clock induction that hasn’t made any progress ... There are lots of examples of why cesarean section rates change with a hospitalist in place, especially if you look at the correlation between cesarean sections and time of day."
Dr. Tessmer-Tuck said she found the Las Vegas study highly relevant because lots of hospitals throughout the country are now going through a similar transition from traditional on-call practice to around-the-clock coverage provided by rotating private practice community laborists, while pondering a possible move to full-time laborists.
"This is where many of our hospitals are at: They’re in the middle phase, with private-practice docs being paid to stay in-house 24 hours in case there’s an emergency," according to Dr. Tessmer-Tuck.
She said she found particularly impressive the investigators’ calculation that a full-time laborist resulted in an average of one fewer cesarean section every 2 days in a population of primiparous, term, singleton patients, with a resultant estimated savings in patient care costs of $2,823-$3,305 per day. Because a laborist might be paid $2,500 per 24-hour shift, the reduced cesarean section rate alone covers the laborist’s salary. Those are the sort of numbers hospital administrators find persuasive.
"This is a message you guys should take home with you when you go back to your own program," she said.
While the Las Vegas study provides the first evidence to be published in a major peer-reviewed journal demonstrating superior clinical outcomes with the full-time laborist model, Dr. Tessmer-Tuck noted that in addition there are several published studies suggesting that hospitals experience fewer adverse events and markedly lower payouts for bad outcomes after they implement multipronged, comprehensive obstetric patient safety programs that include bringing a laborist on board.
"Liability has become a huge issue for us. Many hospitals implement hospitalist programs mainly in order to reduce liability," according to Dr. Tessmer-Tuck.
She cited a study by ob.gyns. at New York Presbyterian/Weill Cornell Medical Center in which they analyzed the impact of a comprehensive patient safety program initiated in stages beginning in 2003. The interventions included mandatory labor and delivery team training aimed at enhancing physician/nurse communication, development of standardized management protocols, training in fetal heart rate monitoring interpretation, creation of a patient safety nurse position, and, in 2006, introduction of a laborist.
It’s not possible to parse out just how much of the improvement in response to the multipronged safety program was the result of adopting the laborist model, Dr. Tessmer-Tuck said, but she noted the average yearly compensation payments for patient claims or lawsuits were $27.6 million during 2003-2006, plummeting to $2.5 million per year in 2007-2009, after the laborist was in place. Moreover, sentinel adverse events such as maternal death or severe neurodevelopmental impairment in a child decreased from five in the year 2000 to none in 2008 and 2009 (Am. J. Obstet. Gynecol. 2011;204:97-105).
Ob.gyns. at Yale–New Haven (Conn.) Hospital introduced a similar comprehensive patient safety program, also including implementation of a 24-hour obstetrics hospitalist, during 2004-2006. During 3 years of prospective follow-up involving nearly 14,000 deliveries, they documented a significant linear decline in obstetric adverse outcomes (Am. J. Obstet. Gynecol. 2009;200:492e1-8). They also administered a validated workplace safety attitude questionnaire four times during 2004-2009 and documented marked improvement over time in favorable scores in the domains of job satisfaction, teamwork, and safety culture (Am J. Obstet. Gynecol. 2011;204:216.e1-6).
Dr. Garite and Dr. Tessmer-Tuck reported having no germane financial relationships.
DENVER – The newly published first data showing improved clinical outcomes after adoption of a full-time hospital laborist program was roundly celebrated at the annual meeting of the Society of Ob/Gyn Hospitalists.
Dr. Thomas J. Garite presented highlights of this freshly published retrospective observational study (Am. J. Obstet. Gynecol. 2013;209:251.e1-6) conducted at a large-delivery-volume tertiary hospital in Las Vegas. Dr. Garite and his coinvestigators, led by Dr. Brian K. Iriye, compared hospital-wide cesarean delivery rates for 6,206 nulliparous, term, singleton live births during 2006-2011.
This was a period of change in how labor and delivery was organized at the hospital. During the first 16 months of the study period, the traditional private-practice model of patient care was in place, with ob.gyns. on call and no laborists in the house. This was followed by a 14-month interlude in which local private-practice ob.gyns. got together and made sure that a community physician was continuously in-hospital to provide laborist coverage.
"I call that the doc-in-a-box model," said Dr. Garite, professor emeritus and former chair of obstetrics and gynecology at the University of California, Irvine.
Finally came a 24-month period with full-time laborists – that is, ob.gyns. without a private practice – providing in-hospital coverage 24/7.
In a multivariate logistic regression analysis adjusted for potential confounders, the hospital’s cesarean section rate was roughly 25% lower after implementation of the full-time laborist program than in either of the other two periods.
"I haven’t seen other studies to date that demonstrate these kinds of outcome advantages for this kind of practice. I think we’re going to see a lot more. But until we do, a lot of people who don’t like change are going to be saying, ‘Wait, where’s the proof?’ Well, this is the beginning of the proof of something I believe in strongly," declared Dr. Garite, who is also editor-in-chief of the American Journal of Obstetrics and Gynecology and chief clinical officer at PeriGen, a provider of fetal surveillance systems.
Society of Ob/Gyn Hospitalists (SOGH) board member Dr. Jennifer Tessmer-Tuck hailed the new study as "the best and almost the only" clinical outcome data to date showing the advantages of the ob.gyn. hospitalist model of care. And it was a long time coming, she noted: a full 10½ years since Dr. Louis Weinstein of the Medical College of Ohio, Toledo, heralded the birth of a radically different form of ob.gyn. practice in his seminal essay "The laborist: A new focus of practice for the obstetrician" (Am. J. Obstet. Gynecol. 2003;188:310-2).
"We have a lot to do. SOGH would really like to have more of a research platform and be able to put ourselves out there. There’s really a gap in care, and we’re hoping to jump in and fill it," said Dr. Tessmer-Tuck, director of North Memorial Medical Center Laborist Associates in Robbinsdale, Minn.
But while the SOGH leadership is eager to see the field assume a bigger research presence, it’s a challenge. Most society members, when they talk about why they became hospitalists, say they had burned out in traditional private practice, with its demanding on-call schedule. They sought well-defined hours, perhaps more family time. Given those priorities, taking on a research project can sound daunting, even though the fruits of such a project might enhance the standing of the young subspecialty.
Dr. Garite reported that the cesarean section rate at the tertiary center was 33.2% during the 24 months when full-time laborists were on hand, compared with 39.2% under the traditional private practice model with no laborists, and 38.7% with laborist coverage by community staff. In a multivariate logistic regression analysis adjusted for maternal age, physician age, race, gestational age, induction of labor, birth weight, and maternal weight, the hospital’s cesarean section rate after the introduction of full-time laborists was 27% lower than in the earlier period of no laborists and 23% less than with community laborist care.
There were no differences between the three groups in rates of low Apgar scores, metabolic acidosis, or any other parameters of adverse neonatal or maternal outcome.
During the study years of 2006-2011, cesarean section rates at the other hospitals in the city were either stable or rising.
Asked why hospital-wide cesarean section rates dropped significantly once full-time obstetric hospitalists were in place, Dr. Garite replied, "It’s not, for example, the patient with abruption who comes in the door; she’s going to get a cesarean section whether a hospitalist is there or some other doctor is covering. Instead, it’s the patient who has what I call ‘failure to wait,’ a.k.a. failure to progress, or the 4 o’clock induction that hasn’t made any progress ... There are lots of examples of why cesarean section rates change with a hospitalist in place, especially if you look at the correlation between cesarean sections and time of day."
Dr. Tessmer-Tuck said she found the Las Vegas study highly relevant because lots of hospitals throughout the country are now going through a similar transition from traditional on-call practice to around-the-clock coverage provided by rotating private practice community laborists, while pondering a possible move to full-time laborists.
"This is where many of our hospitals are at: They’re in the middle phase, with private-practice docs being paid to stay in-house 24 hours in case there’s an emergency," according to Dr. Tessmer-Tuck.
She said she found particularly impressive the investigators’ calculation that a full-time laborist resulted in an average of one fewer cesarean section every 2 days in a population of primiparous, term, singleton patients, with a resultant estimated savings in patient care costs of $2,823-$3,305 per day. Because a laborist might be paid $2,500 per 24-hour shift, the reduced cesarean section rate alone covers the laborist’s salary. Those are the sort of numbers hospital administrators find persuasive.
"This is a message you guys should take home with you when you go back to your own program," she said.
While the Las Vegas study provides the first evidence to be published in a major peer-reviewed journal demonstrating superior clinical outcomes with the full-time laborist model, Dr. Tessmer-Tuck noted that in addition there are several published studies suggesting that hospitals experience fewer adverse events and markedly lower payouts for bad outcomes after they implement multipronged, comprehensive obstetric patient safety programs that include bringing a laborist on board.
"Liability has become a huge issue for us. Many hospitals implement hospitalist programs mainly in order to reduce liability," according to Dr. Tessmer-Tuck.
She cited a study by ob.gyns. at New York Presbyterian/Weill Cornell Medical Center in which they analyzed the impact of a comprehensive patient safety program initiated in stages beginning in 2003. The interventions included mandatory labor and delivery team training aimed at enhancing physician/nurse communication, development of standardized management protocols, training in fetal heart rate monitoring interpretation, creation of a patient safety nurse position, and, in 2006, introduction of a laborist.
It’s not possible to parse out just how much of the improvement in response to the multipronged safety program was the result of adopting the laborist model, Dr. Tessmer-Tuck said, but she noted the average yearly compensation payments for patient claims or lawsuits were $27.6 million during 2003-2006, plummeting to $2.5 million per year in 2007-2009, after the laborist was in place. Moreover, sentinel adverse events such as maternal death or severe neurodevelopmental impairment in a child decreased from five in the year 2000 to none in 2008 and 2009 (Am. J. Obstet. Gynecol. 2011;204:97-105).
Ob.gyns. at Yale–New Haven (Conn.) Hospital introduced a similar comprehensive patient safety program, also including implementation of a 24-hour obstetrics hospitalist, during 2004-2006. During 3 years of prospective follow-up involving nearly 14,000 deliveries, they documented a significant linear decline in obstetric adverse outcomes (Am. J. Obstet. Gynecol. 2009;200:492e1-8). They also administered a validated workplace safety attitude questionnaire four times during 2004-2009 and documented marked improvement over time in favorable scores in the domains of job satisfaction, teamwork, and safety culture (Am J. Obstet. Gynecol. 2011;204:216.e1-6).
Dr. Garite and Dr. Tessmer-Tuck reported having no germane financial relationships.
AT THE SOGH ANNUAL CLINICAL MEETING