User login
Most in-hospital pneumonia deaths may not be preventable
Most in-hospital deaths from community-acquired pneumonia are not preventable with current medical therapy, according to an analysis of deaths at five U.S. hospitals with expertise in pneumonia care.
Adults who are hospitalized with community-acquired pneumonia (CAP) are at high risk for short-term mortality but it is unclear whether an improvement in care could lower this risk, noted the study authors led by Grant W. Waterer, MBBS, PhD, of Northwestern University, Chicago.
“Understanding the circumstances in which CAP patients die could facilitate improvements in the management of CAP by enabling future improvement efforts to focus on common preventable causes of death,” they wrote. Their report was published in CHEST®.
They therefore performed a secondary analysis of the Etiology of Pneumonia in the Community (EPIC) study involving adults hospitalized with CAP between January 2010 and June 2012 across five tertiary-care hospitals in the United States.
The clinical characteristics of patients who died in the hospital were compared with those of patients who survived to hospital discharge. Chronic heart failure, chronic obstructive pulmonary disease, coronary artery disease, chronic liver disease, cerebrovascular disease, cancer (excluding skin cancer), and diabetes were considered as severe chronic comorbidities based on their association with increased mortality and ICU admission in CAP severity scores.
Deaths caused by septic shock, respiratory failure, multisystem organ failure, cardiopulmonary arrest prior to stabilization of CAP, and endocarditis, were considered to be directly related to CAP.
Conversely, causes of death indirectly related to CAP included acute cardiovascular disease, stroke, acute renal failure, and secondary infections developed after hospitalization. Deaths caused by cancer, cirrhosis, and chronic neurologic conditions were considered unrelated to CAP.
Medical notes were assessed to determine whether the patient received management consistent with current recommendations; for example, antibiotics consistent with guidelines from the Infectious Diseases Society of America.
End-of-life limitations in care, such as patient/family decision not to proceed with full medical treatment, also were considered by the research team.
Results showed that among the 2,320 patients with radiographically confirmed CAP, 52 died during initial hospitalization, 33 of whom were aged 65 years or older, and 32 of whom had two or more chronic comorbidities.
Most of the in-hospital deaths occurred early in the hospitalization: 35 within the first 10 days of admission, and 5 after 30 days in hospital.
CAP was judged by an expert physician review panel to be the direct cause of death in 27 of the patients, 10 with CAP having an indirect role with major contribution, 9 with CAP having an indirect role with minor contribution, and 6 with CAP having no role in death.
Do-not-resuscitate orders were present at the time of death for 21 of the patients.
Forty-five of the patients were admitted to an ICU, with 37 dying in the ICU. The eight patients who died on the ward after transfer out of the ICU had end-of-life limitations of care in place.
The researchers noted that the number of patients dying in the ICU was greater in the United States, possibly because in Europe fewer patients are admitted to an ICU.
“This discrepancy likely reflects cultural differences between the U.S. and Europe in the role of intensive care for patients with advanced age and/or advanced comorbid conditions,” they noted.
Overall, the physician review panel identified nine patients who had a lapse in quality of in-hospital CAP care, with four of the deaths potentially linked to this lapse in care.
However, two of the patients had end-of-life limitations of care in place, which according to the authors meant that “only two patients undergoing full medical treatment without end-of-life limitations of care had an identified lapse in quality of in-hospital pneumonia care potentially contributing to in-hospital death, including one with a delay in antibiotics for over an hour in the presence of shock and one with initial antibiotics not consistent with IDSA/ATS guidelines.”
The research team concluded that most in-hospital deaths among adult patients admitted with CAP in their study would not have been preventable with higher quality in-hospital pneumonia care.
“Many of the in-hospital deaths among patients admitted with CAP occurred in older patients with severe comorbidities and end-of-life limitations in care,” they noted.
They said the influence of end-of-life limitations on care short of full palliation was an important finding, with all patients who died outside the ICU having end-of-life limitations in care.
“Current diagnostic related group (DRG) and international classification of diseases (ICD) coding systems do not have the necessary nuances to capture these limitations of care, yet they are clearly important factors in determining whether patients experience in-hospital death,” they added.
Dr. Waterer reported no conflicts. Two coauthors reported potential conflicts of interest in relation to consulting fees from several pharmaceutical companies.
SOURCE: Waterer G. et al. CHEST 2018;154(3):628-35. doi: 10.1016/j.chest.2018.05.021.
Most in-hospital deaths from community-acquired pneumonia are not preventable with current medical therapy, according to an analysis of deaths at five U.S. hospitals with expertise in pneumonia care.
Adults who are hospitalized with community-acquired pneumonia (CAP) are at high risk for short-term mortality but it is unclear whether an improvement in care could lower this risk, noted the study authors led by Grant W. Waterer, MBBS, PhD, of Northwestern University, Chicago.
“Understanding the circumstances in which CAP patients die could facilitate improvements in the management of CAP by enabling future improvement efforts to focus on common preventable causes of death,” they wrote. Their report was published in CHEST®.
They therefore performed a secondary analysis of the Etiology of Pneumonia in the Community (EPIC) study involving adults hospitalized with CAP between January 2010 and June 2012 across five tertiary-care hospitals in the United States.
The clinical characteristics of patients who died in the hospital were compared with those of patients who survived to hospital discharge. Chronic heart failure, chronic obstructive pulmonary disease, coronary artery disease, chronic liver disease, cerebrovascular disease, cancer (excluding skin cancer), and diabetes were considered as severe chronic comorbidities based on their association with increased mortality and ICU admission in CAP severity scores.
Deaths caused by septic shock, respiratory failure, multisystem organ failure, cardiopulmonary arrest prior to stabilization of CAP, and endocarditis, were considered to be directly related to CAP.
Conversely, causes of death indirectly related to CAP included acute cardiovascular disease, stroke, acute renal failure, and secondary infections developed after hospitalization. Deaths caused by cancer, cirrhosis, and chronic neurologic conditions were considered unrelated to CAP.
Medical notes were assessed to determine whether the patient received management consistent with current recommendations; for example, antibiotics consistent with guidelines from the Infectious Diseases Society of America.
End-of-life limitations in care, such as patient/family decision not to proceed with full medical treatment, also were considered by the research team.
Results showed that among the 2,320 patients with radiographically confirmed CAP, 52 died during initial hospitalization, 33 of whom were aged 65 years or older, and 32 of whom had two or more chronic comorbidities.
Most of the in-hospital deaths occurred early in the hospitalization: 35 within the first 10 days of admission, and 5 after 30 days in hospital.
CAP was judged by an expert physician review panel to be the direct cause of death in 27 of the patients, 10 with CAP having an indirect role with major contribution, 9 with CAP having an indirect role with minor contribution, and 6 with CAP having no role in death.
Do-not-resuscitate orders were present at the time of death for 21 of the patients.
Forty-five of the patients were admitted to an ICU, with 37 dying in the ICU. The eight patients who died on the ward after transfer out of the ICU had end-of-life limitations of care in place.
The researchers noted that the number of patients dying in the ICU was greater in the United States, possibly because in Europe fewer patients are admitted to an ICU.
“This discrepancy likely reflects cultural differences between the U.S. and Europe in the role of intensive care for patients with advanced age and/or advanced comorbid conditions,” they noted.
Overall, the physician review panel identified nine patients who had a lapse in quality of in-hospital CAP care, with four of the deaths potentially linked to this lapse in care.
However, two of the patients had end-of-life limitations of care in place, which according to the authors meant that “only two patients undergoing full medical treatment without end-of-life limitations of care had an identified lapse in quality of in-hospital pneumonia care potentially contributing to in-hospital death, including one with a delay in antibiotics for over an hour in the presence of shock and one with initial antibiotics not consistent with IDSA/ATS guidelines.”
The research team concluded that most in-hospital deaths among adult patients admitted with CAP in their study would not have been preventable with higher quality in-hospital pneumonia care.
“Many of the in-hospital deaths among patients admitted with CAP occurred in older patients with severe comorbidities and end-of-life limitations in care,” they noted.
They said the influence of end-of-life limitations on care short of full palliation was an important finding, with all patients who died outside the ICU having end-of-life limitations in care.
“Current diagnostic related group (DRG) and international classification of diseases (ICD) coding systems do not have the necessary nuances to capture these limitations of care, yet they are clearly important factors in determining whether patients experience in-hospital death,” they added.
Dr. Waterer reported no conflicts. Two coauthors reported potential conflicts of interest in relation to consulting fees from several pharmaceutical companies.
SOURCE: Waterer G. et al. CHEST 2018;154(3):628-35. doi: 10.1016/j.chest.2018.05.021.
Most in-hospital deaths from community-acquired pneumonia are not preventable with current medical therapy, according to an analysis of deaths at five U.S. hospitals with expertise in pneumonia care.
Adults who are hospitalized with community-acquired pneumonia (CAP) are at high risk for short-term mortality but it is unclear whether an improvement in care could lower this risk, noted the study authors led by Grant W. Waterer, MBBS, PhD, of Northwestern University, Chicago.
“Understanding the circumstances in which CAP patients die could facilitate improvements in the management of CAP by enabling future improvement efforts to focus on common preventable causes of death,” they wrote. Their report was published in CHEST®.
They therefore performed a secondary analysis of the Etiology of Pneumonia in the Community (EPIC) study involving adults hospitalized with CAP between January 2010 and June 2012 across five tertiary-care hospitals in the United States.
The clinical characteristics of patients who died in the hospital were compared with those of patients who survived to hospital discharge. Chronic heart failure, chronic obstructive pulmonary disease, coronary artery disease, chronic liver disease, cerebrovascular disease, cancer (excluding skin cancer), and diabetes were considered as severe chronic comorbidities based on their association with increased mortality and ICU admission in CAP severity scores.
Deaths caused by septic shock, respiratory failure, multisystem organ failure, cardiopulmonary arrest prior to stabilization of CAP, and endocarditis, were considered to be directly related to CAP.
Conversely, causes of death indirectly related to CAP included acute cardiovascular disease, stroke, acute renal failure, and secondary infections developed after hospitalization. Deaths caused by cancer, cirrhosis, and chronic neurologic conditions were considered unrelated to CAP.
Medical notes were assessed to determine whether the patient received management consistent with current recommendations; for example, antibiotics consistent with guidelines from the Infectious Diseases Society of America.
End-of-life limitations in care, such as patient/family decision not to proceed with full medical treatment, also were considered by the research team.
Results showed that among the 2,320 patients with radiographically confirmed CAP, 52 died during initial hospitalization, 33 of whom were aged 65 years or older, and 32 of whom had two or more chronic comorbidities.
Most of the in-hospital deaths occurred early in the hospitalization: 35 within the first 10 days of admission, and 5 after 30 days in hospital.
CAP was judged by an expert physician review panel to be the direct cause of death in 27 of the patients, 10 with CAP having an indirect role with major contribution, 9 with CAP having an indirect role with minor contribution, and 6 with CAP having no role in death.
Do-not-resuscitate orders were present at the time of death for 21 of the patients.
Forty-five of the patients were admitted to an ICU, with 37 dying in the ICU. The eight patients who died on the ward after transfer out of the ICU had end-of-life limitations of care in place.
The researchers noted that the number of patients dying in the ICU was greater in the United States, possibly because in Europe fewer patients are admitted to an ICU.
“This discrepancy likely reflects cultural differences between the U.S. and Europe in the role of intensive care for patients with advanced age and/or advanced comorbid conditions,” they noted.
Overall, the physician review panel identified nine patients who had a lapse in quality of in-hospital CAP care, with four of the deaths potentially linked to this lapse in care.
However, two of the patients had end-of-life limitations of care in place, which according to the authors meant that “only two patients undergoing full medical treatment without end-of-life limitations of care had an identified lapse in quality of in-hospital pneumonia care potentially contributing to in-hospital death, including one with a delay in antibiotics for over an hour in the presence of shock and one with initial antibiotics not consistent with IDSA/ATS guidelines.”
The research team concluded that most in-hospital deaths among adult patients admitted with CAP in their study would not have been preventable with higher quality in-hospital pneumonia care.
“Many of the in-hospital deaths among patients admitted with CAP occurred in older patients with severe comorbidities and end-of-life limitations in care,” they noted.
They said the influence of end-of-life limitations on care short of full palliation was an important finding, with all patients who died outside the ICU having end-of-life limitations in care.
“Current diagnostic related group (DRG) and international classification of diseases (ICD) coding systems do not have the necessary nuances to capture these limitations of care, yet they are clearly important factors in determining whether patients experience in-hospital death,” they added.
Dr. Waterer reported no conflicts. Two coauthors reported potential conflicts of interest in relation to consulting fees from several pharmaceutical companies.
SOURCE: Waterer G. et al. CHEST 2018;154(3):628-35. doi: 10.1016/j.chest.2018.05.021.
FROM CHEST
Key clinical point: Most in-hospital deaths from community-acquired pneumonia are not preventable with current medical therapy.
Major finding: Two out of 52 patients who died in-hospital from community-acquired pneumonia (CAP) who were undergoing full medical treatment without end-of-life limitations of care had an identified lapse in quality of in-hospital pneumonia care that potentially contributed to their death.
Study details: A secondary analysis of the prospective multicenter Etiology of Pneumonia in the Community (EPIC) study involving 2,320 adults with radiographically confirmed CAP.
Disclosures: Dr. Waterer reported no conflicts. Two coauthors reported potential conflicts of interest in relation to consulting fees from several pharmaceutical companies.
Source: Waterer G. et al. CHEST 2018;154(3):628-35.
UN aims to eradicate TB by 2030
A concerted a lethal disease affecting one-quarter of the world’s population by the year 2030.
On September 26 the United Nations General Assembly will convene a high-level meeting of global stakeholders to solidify the eradication plan, addressing the global crisis of tuberculosis (TB), the world’s most deadly infectious disease.
“We must seize the moment,” said Tereza Kasaeva, MD, director of the World Health Organization’s global TB program, speaking at a telebriefing and press conference accompanying the release of the World Health Organization’s annual global tuberculosis report. “It’s unacceptable in the 21st century that millions lose their lives to this preventable and curable disease.”
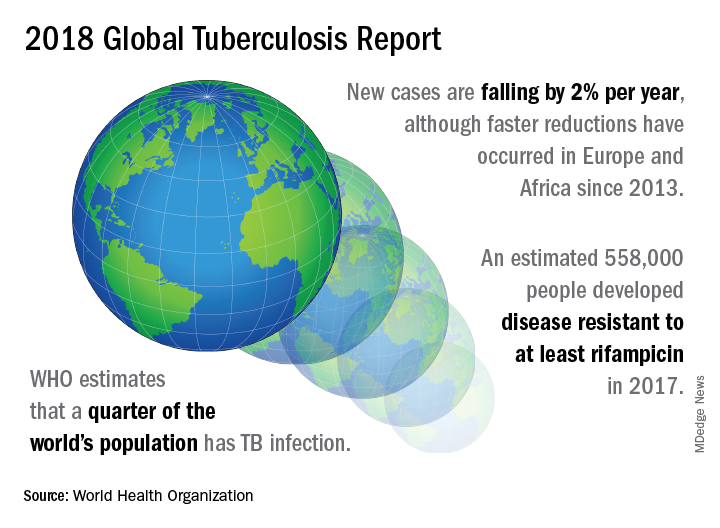
TB caused 1.6 million deaths globally in 2017, and the World Health Organization (WHO) estimates that of the 10 million new cases of TB last year, 558,000 are multi-drug resistant (MDR) infections.
Though death rates and new cases are falling globally each year, significantly more resources are needed to boost access to preventive treatment for latent TB infection; “Most people needing it are not yet accessing care,” according to the press briefing accompanying the report.
A review and commentary on TB incubation and latency published in BMJ (2018;362:k2738 doi: 10.1136/bmj.k2738; e-pub 23 Aug 2018) has called into question the focus preventive treatment of latent cases at the expense of reaching those most likely to die from TB (e.g., HIV patients, children of individuals living with active TB). The authors state that “latent” TB is identified by indirect evidence of present or past infection with Mycobacterium tuberculosis as inferred by a detectable adaptive immune response to M tuberculosis antigens. Active TB infection is overwhelmingly the result of a primary infection and almost always occurs within two years.
In order to meet the ambitious goal of TB eradication by the year 2030, treatment coverage must rise to 90% globally from the current 64%, according to the report.
Progress in southern Africa and in the Russian Federation, where efforts have led to a 30% reduction in TB mortality and a decrease in incidence of 5% per year, show that steep reductions in TB are possible when resources are brought to bear on the problem, said Dr. Kasaeva. “We should acknowledge that actions in some countries and regions show that progress can accelerate,” she said. Still, she noted, “Four thousand lives per day are lost to TB. Tuberculosis is the leading killer of people living with HIV, and the major cause of deaths related to antimicrobial resistance” at a global level.
Two thirds of all TB cases occur in eight countries, with India, China, and Indonesia leading this group. About half of the cases of MDR TB occur in India, China, and Russia, said Dr. Kasaeva, and globally only one in four individuals with MDR TB who need access to treatment have received it. “We need to urgently tackle the multidrug resistant TB public health crisis,” she said.
Major impediments to successful public health efforts against TB are underdiagnosis and underreporting: It is estimated that 3.6 million of 2017’s 10 million new cases were not officially recorded or reported. Countries where these problems are most serious include India, Indonesia, and Nigeria. Fewer than half of the children with TB are reported globally, according to the report.
People living with HIV/AIDS who are also infected with TB number nearly 1,000,000, but only about half of these were officially reported in 2017.
In terms of prevention priorities, WHO has recommended targeting treatment of latent TB in two groups: People living with HIV/AIDS, and children under the age of 5 years who live in households with TB-infected individuals.
“To enable these actions,” said Dr. Kasaeva, “we need strengthened commitments not just for TB care, but for overall health services. So the aim for universal coverage is real.” Underreporting is particularly prevalent in lower income countries with large unregulated private sectors, she said, though India and Indonesia have taken corrective steps to increase reporting.
A meaningful global initiative will not come cheap: The current annual shortfall in funding for TB prevention, diagnosis, and treatment is about $3.5 billion. By the year 2022, the gap between funding and what’s needed to stay on track for the 2030 target will be over $6 billion, said Dr. Kasaeva.
The best use of increased resources for TB eradication will be in locally focused efforts, said Irene Koek, MD, the United States Agency for International Development’s deputy administrator for global health. “It is likely that each region requires a tailored response.” Further, “to improve quality of care we need to ensure that services are patient centered,” she said at the press conference.
To that end, Dr. Koek expects that at the upcoming high-level meeting, the United Nations member states will be called on to develop an open framework, with clear accountability for monitoring and reviewing progress. The road forward should “celebrate accomplishments and acknowledge shortcomings,” she said. Some recent studies have shown that treatment success rates above 80% for patients with MDR TB can be achieved.
“Lessons learned from these experiences should be documented and shared in order to replicate success globally,” said Dr. Koek.
The United States, said Dr. Koek, is the leading global investor in TB research and treatment. “We welcome increased partnerships, especially with countries with the highest burden, to end global suffering from this disease.”
Eric Goosby, MD, the United Nations special envoy on TB, used his speaking time to lend some perspective to the social framework around TB’s longtime lethality.
There are aspects of TB infection that differentiate it from HIV/AIDS, said Dr. Goosby, who has spent most of his clinical and public health career on HIV/AIDS treatment and prevention. In contrast to an infection that at present requires a lifetime of treatment, TB can ordinarily be treated in 6 months, making it an unpleasant episode that an individual may be eager to move past. Additionally, the fact that TB has had a “hold on the world since the time of the ancient Egyptians” may paradoxically have served to lessen urgency in research and treatment efforts, he noted.
Dr. Goosby also spoke of the stigma surrounding TB, whose sufferers are likely to be facing dire poverty, malnutrition, and other infectious disease burdens. Civil society concerned with TB, he said, has spoken up “for those without a voice, for those who have difficulty advocating for themselves.”
Dr. Kasaeva agreed, noting that TB “affects the poorest of the poor, which makes it extraordinarily difficult for activism to come from that population.”
However, others have spoken for those affected, said Dr. Goosby. “The TB civil society has put its heart and soul this last year into gathering political will from leaders around the world…. It’s not a passive effort; it involves a lot of work.” During the past year of concerted effort, he said, “All of us have known the difficulty of pushing a political leader up that learning curve.”
As the upcoming high-level meeting approaches, those who have been working on the effort can feel the momentum, said Dr. Goosby. Still, he noted, “While there’s a significant step forward, this is not the time for a victory dance. This is really the time for a reflection...Do we understand the burden in our respective countries, and has the response been adequate?”
The goal for the meeting is to have leaders “step up to commit, not for one day, or for one meeting, but for the duration of the effort,” said Dr. Goosby. “We must make sure that the words that we hear next week from our leaders translate into action...Next week the world will say, ‘No more. No longer. No one is immune to TB. Tuberculosis is preventable; tuberculosis is treatable; tuberculosis is curable.’”
The BMJ commentary, by Marcel A. Behr, MD, of McGill International TB Centre, Infectious Diseases and Immunity in Global Health Program, McGill University Health Centre Research Institute, and his colleagues, recommend caution when building a prevention strategy around treating many millions of individuals with “latent” TB. They wrote, “Immunoreactivity to TB does not necessarily indicate the presence of live bacteria, as reactivity can persist after infection has been cleared. Classifying two billion people with evidence of immunoreactivity as having latent TB infection may divert fundamental research and public health interventions away from transmissible active TB disease and newly infected people at highest risk of progression to disease.”
This story was updated on 09/24/2018
A concerted a lethal disease affecting one-quarter of the world’s population by the year 2030.

On September 26 the United Nations General Assembly will convene a high-level meeting of global stakeholders to solidify the eradication plan, addressing the global crisis of tuberculosis (TB), the world’s most deadly infectious disease.
“We must seize the moment,” said Tereza Kasaeva, MD, director of the World Health Organization’s global TB program, speaking at a telebriefing and press conference accompanying the release of the World Health Organization’s annual global tuberculosis report. “It’s unacceptable in the 21st century that millions lose their lives to this preventable and curable disease.”
TB caused 1.6 million deaths globally in 2017, and the World Health Organization (WHO) estimates that of the 10 million new cases of TB last year, 558,000 are multi-drug resistant (MDR) infections.
Though death rates and new cases are falling globally each year, significantly more resources are needed to boost access to preventive treatment for latent TB infection; “Most people needing it are not yet accessing care,” according to the press briefing accompanying the report.
A review and commentary on TB incubation and latency published in BMJ (2018;362:k2738 doi: 10.1136/bmj.k2738; e-pub 23 Aug 2018) has called into question the focus preventive treatment of latent cases at the expense of reaching those most likely to die from TB (e.g., HIV patients, children of individuals living with active TB). The authors state that “latent” TB is identified by indirect evidence of present or past infection with Mycobacterium tuberculosis as inferred by a detectable adaptive immune response to M tuberculosis antigens. Active TB infection is overwhelmingly the result of a primary infection and almost always occurs within two years.
In order to meet the ambitious goal of TB eradication by the year 2030, treatment coverage must rise to 90% globally from the current 64%, according to the report.
Progress in southern Africa and in the Russian Federation, where efforts have led to a 30% reduction in TB mortality and a decrease in incidence of 5% per year, show that steep reductions in TB are possible when resources are brought to bear on the problem, said Dr. Kasaeva. “We should acknowledge that actions in some countries and regions show that progress can accelerate,” she said. Still, she noted, “Four thousand lives per day are lost to TB. Tuberculosis is the leading killer of people living with HIV, and the major cause of deaths related to antimicrobial resistance” at a global level.
Two thirds of all TB cases occur in eight countries, with India, China, and Indonesia leading this group. About half of the cases of MDR TB occur in India, China, and Russia, said Dr. Kasaeva, and globally only one in four individuals with MDR TB who need access to treatment have received it. “We need to urgently tackle the multidrug resistant TB public health crisis,” she said.
Major impediments to successful public health efforts against TB are underdiagnosis and underreporting: It is estimated that 3.6 million of 2017’s 10 million new cases were not officially recorded or reported. Countries where these problems are most serious include India, Indonesia, and Nigeria. Fewer than half of the children with TB are reported globally, according to the report.
People living with HIV/AIDS who are also infected with TB number nearly 1,000,000, but only about half of these were officially reported in 2017.
In terms of prevention priorities, WHO has recommended targeting treatment of latent TB in two groups: People living with HIV/AIDS, and children under the age of 5 years who live in households with TB-infected individuals.
“To enable these actions,” said Dr. Kasaeva, “we need strengthened commitments not just for TB care, but for overall health services. So the aim for universal coverage is real.” Underreporting is particularly prevalent in lower income countries with large unregulated private sectors, she said, though India and Indonesia have taken corrective steps to increase reporting.
A meaningful global initiative will not come cheap: The current annual shortfall in funding for TB prevention, diagnosis, and treatment is about $3.5 billion. By the year 2022, the gap between funding and what’s needed to stay on track for the 2030 target will be over $6 billion, said Dr. Kasaeva.
The best use of increased resources for TB eradication will be in locally focused efforts, said Irene Koek, MD, the United States Agency for International Development’s deputy administrator for global health. “It is likely that each region requires a tailored response.” Further, “to improve quality of care we need to ensure that services are patient centered,” she said at the press conference.
To that end, Dr. Koek expects that at the upcoming high-level meeting, the United Nations member states will be called on to develop an open framework, with clear accountability for monitoring and reviewing progress. The road forward should “celebrate accomplishments and acknowledge shortcomings,” she said. Some recent studies have shown that treatment success rates above 80% for patients with MDR TB can be achieved.
“Lessons learned from these experiences should be documented and shared in order to replicate success globally,” said Dr. Koek.
The United States, said Dr. Koek, is the leading global investor in TB research and treatment. “We welcome increased partnerships, especially with countries with the highest burden, to end global suffering from this disease.”
Eric Goosby, MD, the United Nations special envoy on TB, used his speaking time to lend some perspective to the social framework around TB’s longtime lethality.
There are aspects of TB infection that differentiate it from HIV/AIDS, said Dr. Goosby, who has spent most of his clinical and public health career on HIV/AIDS treatment and prevention. In contrast to an infection that at present requires a lifetime of treatment, TB can ordinarily be treated in 6 months, making it an unpleasant episode that an individual may be eager to move past. Additionally, the fact that TB has had a “hold on the world since the time of the ancient Egyptians” may paradoxically have served to lessen urgency in research and treatment efforts, he noted.
Dr. Goosby also spoke of the stigma surrounding TB, whose sufferers are likely to be facing dire poverty, malnutrition, and other infectious disease burdens. Civil society concerned with TB, he said, has spoken up “for those without a voice, for those who have difficulty advocating for themselves.”
Dr. Kasaeva agreed, noting that TB “affects the poorest of the poor, which makes it extraordinarily difficult for activism to come from that population.”
However, others have spoken for those affected, said Dr. Goosby. “The TB civil society has put its heart and soul this last year into gathering political will from leaders around the world…. It’s not a passive effort; it involves a lot of work.” During the past year of concerted effort, he said, “All of us have known the difficulty of pushing a political leader up that learning curve.”
As the upcoming high-level meeting approaches, those who have been working on the effort can feel the momentum, said Dr. Goosby. Still, he noted, “While there’s a significant step forward, this is not the time for a victory dance. This is really the time for a reflection...Do we understand the burden in our respective countries, and has the response been adequate?”
The goal for the meeting is to have leaders “step up to commit, not for one day, or for one meeting, but for the duration of the effort,” said Dr. Goosby. “We must make sure that the words that we hear next week from our leaders translate into action...Next week the world will say, ‘No more. No longer. No one is immune to TB. Tuberculosis is preventable; tuberculosis is treatable; tuberculosis is curable.’”
The BMJ commentary, by Marcel A. Behr, MD, of McGill International TB Centre, Infectious Diseases and Immunity in Global Health Program, McGill University Health Centre Research Institute, and his colleagues, recommend caution when building a prevention strategy around treating many millions of individuals with “latent” TB. They wrote, “Immunoreactivity to TB does not necessarily indicate the presence of live bacteria, as reactivity can persist after infection has been cleared. Classifying two billion people with evidence of immunoreactivity as having latent TB infection may divert fundamental research and public health interventions away from transmissible active TB disease and newly infected people at highest risk of progression to disease.”
This story was updated on 09/24/2018
A concerted a lethal disease affecting one-quarter of the world’s population by the year 2030.

On September 26 the United Nations General Assembly will convene a high-level meeting of global stakeholders to solidify the eradication plan, addressing the global crisis of tuberculosis (TB), the world’s most deadly infectious disease.
“We must seize the moment,” said Tereza Kasaeva, MD, director of the World Health Organization’s global TB program, speaking at a telebriefing and press conference accompanying the release of the World Health Organization’s annual global tuberculosis report. “It’s unacceptable in the 21st century that millions lose their lives to this preventable and curable disease.”
TB caused 1.6 million deaths globally in 2017, and the World Health Organization (WHO) estimates that of the 10 million new cases of TB last year, 558,000 are multi-drug resistant (MDR) infections.
Though death rates and new cases are falling globally each year, significantly more resources are needed to boost access to preventive treatment for latent TB infection; “Most people needing it are not yet accessing care,” according to the press briefing accompanying the report.
A review and commentary on TB incubation and latency published in BMJ (2018;362:k2738 doi: 10.1136/bmj.k2738; e-pub 23 Aug 2018) has called into question the focus preventive treatment of latent cases at the expense of reaching those most likely to die from TB (e.g., HIV patients, children of individuals living with active TB). The authors state that “latent” TB is identified by indirect evidence of present or past infection with Mycobacterium tuberculosis as inferred by a detectable adaptive immune response to M tuberculosis antigens. Active TB infection is overwhelmingly the result of a primary infection and almost always occurs within two years.
In order to meet the ambitious goal of TB eradication by the year 2030, treatment coverage must rise to 90% globally from the current 64%, according to the report.
Progress in southern Africa and in the Russian Federation, where efforts have led to a 30% reduction in TB mortality and a decrease in incidence of 5% per year, show that steep reductions in TB are possible when resources are brought to bear on the problem, said Dr. Kasaeva. “We should acknowledge that actions in some countries and regions show that progress can accelerate,” she said. Still, she noted, “Four thousand lives per day are lost to TB. Tuberculosis is the leading killer of people living with HIV, and the major cause of deaths related to antimicrobial resistance” at a global level.
Two thirds of all TB cases occur in eight countries, with India, China, and Indonesia leading this group. About half of the cases of MDR TB occur in India, China, and Russia, said Dr. Kasaeva, and globally only one in four individuals with MDR TB who need access to treatment have received it. “We need to urgently tackle the multidrug resistant TB public health crisis,” she said.
Major impediments to successful public health efforts against TB are underdiagnosis and underreporting: It is estimated that 3.6 million of 2017’s 10 million new cases were not officially recorded or reported. Countries where these problems are most serious include India, Indonesia, and Nigeria. Fewer than half of the children with TB are reported globally, according to the report.
People living with HIV/AIDS who are also infected with TB number nearly 1,000,000, but only about half of these were officially reported in 2017.
In terms of prevention priorities, WHO has recommended targeting treatment of latent TB in two groups: People living with HIV/AIDS, and children under the age of 5 years who live in households with TB-infected individuals.
“To enable these actions,” said Dr. Kasaeva, “we need strengthened commitments not just for TB care, but for overall health services. So the aim for universal coverage is real.” Underreporting is particularly prevalent in lower income countries with large unregulated private sectors, she said, though India and Indonesia have taken corrective steps to increase reporting.
A meaningful global initiative will not come cheap: The current annual shortfall in funding for TB prevention, diagnosis, and treatment is about $3.5 billion. By the year 2022, the gap between funding and what’s needed to stay on track for the 2030 target will be over $6 billion, said Dr. Kasaeva.
The best use of increased resources for TB eradication will be in locally focused efforts, said Irene Koek, MD, the United States Agency for International Development’s deputy administrator for global health. “It is likely that each region requires a tailored response.” Further, “to improve quality of care we need to ensure that services are patient centered,” she said at the press conference.
To that end, Dr. Koek expects that at the upcoming high-level meeting, the United Nations member states will be called on to develop an open framework, with clear accountability for monitoring and reviewing progress. The road forward should “celebrate accomplishments and acknowledge shortcomings,” she said. Some recent studies have shown that treatment success rates above 80% for patients with MDR TB can be achieved.
“Lessons learned from these experiences should be documented and shared in order to replicate success globally,” said Dr. Koek.
The United States, said Dr. Koek, is the leading global investor in TB research and treatment. “We welcome increased partnerships, especially with countries with the highest burden, to end global suffering from this disease.”
Eric Goosby, MD, the United Nations special envoy on TB, used his speaking time to lend some perspective to the social framework around TB’s longtime lethality.
There are aspects of TB infection that differentiate it from HIV/AIDS, said Dr. Goosby, who has spent most of his clinical and public health career on HIV/AIDS treatment and prevention. In contrast to an infection that at present requires a lifetime of treatment, TB can ordinarily be treated in 6 months, making it an unpleasant episode that an individual may be eager to move past. Additionally, the fact that TB has had a “hold on the world since the time of the ancient Egyptians” may paradoxically have served to lessen urgency in research and treatment efforts, he noted.
Dr. Goosby also spoke of the stigma surrounding TB, whose sufferers are likely to be facing dire poverty, malnutrition, and other infectious disease burdens. Civil society concerned with TB, he said, has spoken up “for those without a voice, for those who have difficulty advocating for themselves.”
Dr. Kasaeva agreed, noting that TB “affects the poorest of the poor, which makes it extraordinarily difficult for activism to come from that population.”
However, others have spoken for those affected, said Dr. Goosby. “The TB civil society has put its heart and soul this last year into gathering political will from leaders around the world…. It’s not a passive effort; it involves a lot of work.” During the past year of concerted effort, he said, “All of us have known the difficulty of pushing a political leader up that learning curve.”
As the upcoming high-level meeting approaches, those who have been working on the effort can feel the momentum, said Dr. Goosby. Still, he noted, “While there’s a significant step forward, this is not the time for a victory dance. This is really the time for a reflection...Do we understand the burden in our respective countries, and has the response been adequate?”
The goal for the meeting is to have leaders “step up to commit, not for one day, or for one meeting, but for the duration of the effort,” said Dr. Goosby. “We must make sure that the words that we hear next week from our leaders translate into action...Next week the world will say, ‘No more. No longer. No one is immune to TB. Tuberculosis is preventable; tuberculosis is treatable; tuberculosis is curable.’”
The BMJ commentary, by Marcel A. Behr, MD, of McGill International TB Centre, Infectious Diseases and Immunity in Global Health Program, McGill University Health Centre Research Institute, and his colleagues, recommend caution when building a prevention strategy around treating many millions of individuals with “latent” TB. They wrote, “Immunoreactivity to TB does not necessarily indicate the presence of live bacteria, as reactivity can persist after infection has been cleared. Classifying two billion people with evidence of immunoreactivity as having latent TB infection may divert fundamental research and public health interventions away from transmissible active TB disease and newly infected people at highest risk of progression to disease.”
This story was updated on 09/24/2018
FROM A WORLD HEALTH ORGANIZATION PRESS CONFERENCE
Variation in bacterial drug susceptibility tied to TB relapse risk
Higher pretreatment drug concentrations close to a resistance breakpoint for susceptibility were associated with greater relapse risk in TB, based on data from 54 patients who relapsed and 63 who were treated and cured.
“We postulated that drug-susceptible Mycobacterium tuberculosis might have a graded spectrum of susceptibilities that could be used to determine the risk of relapse,” wrote Roberto Colangeli, PhD, of Rutgers University, Newark, N.J., and his colleagues.
In a study published in the New England Journal of Medicine, the researchers examined pretreatment bacterial isolates from adults with TB who had experienced relapse and those who were cured. Using these isolates, they identified the minimum inhibitory concentration (MIC) – the lowest concentration of the drug that prevents visible bacterial growth in culture – for isoniazid and rifampin.
Overall, after controlling for other potential relapse risk factors, higher pretreatment MIC values for both isoniazid and rifampin were associated with an increased relapse risk. For isoniazid, the average MIC below the breakpoint was 0.0334 mcg/mL for relapsed patients and 0.0286 mcg/mL for cured patients. For rifampin, the average MIC below the breakpoint was 0.0695 mcg/mL for relapsed patients and 0.0453 mcg/mL for cured patients. The higher values for the relapsed versus cured patients were represented by factors of 1.17 and 1.53 for isoniazid and rifampin, respectively.
The average age of the patients was 41 years; 83% were men, and 35% were non-Hispanic white.
The study findings were limited by several factors, including the small sample size, retrospective design, and inability to test MIC values from primary cultures versus subcultures, the researchers wrote. However, the results suggest an impact of MIC values on treatment outcomes, and “additional studies that are performed in larger, well-defined prospective cohorts and that include MIC testing of pretreatment culture isolates will be useful to better validate these findings,”
The study was funded by the National Institute of Allergy and Infectious Diseases. Dr. Colangeli reported no financial conflicts. Dr. Alland disclosed funding from Cepheid and several current and pending patents in the United States and Europe, with some royalties paid to Cepheid.
SOURCE: Colangeli R et al. N Engl J Med. 2018;379:823-33.
Although standard four-drug therapy has been shown to cure 90% of patients in several clinical trials, patients do relapse for reasons such as poor treatment adherence and “variations in the characteristics of the infected patients or the infecting pathogens,” wrote Eric J. Rubin, MD, in an accompanying editorial (N Engl J Med. 2018;379:882-3).
Current antibiotic susceptibility thresholds are often set by committees using models, said Dr. Rubin. “Given the uncertainties in modeling, as has been seen in clinical studies, these breakpoints can be imperfect predictors of treatment response.”
Dr. Rubin proposed that minimum inhibitory concentration (MIC) concentrations could be an alternative to in vitro testing as a predictor of treatment response.
“The clinical laboratory provides us not only with a breakpoint interpretation but also with raw data, a quantitative assessment of MIC values,” he noted. “These values can be thought of more as probabilities of successful therapy than as absolute thresholds, a change in attitude that may dispel a false sense of security about the choice of regimen in the treatment of patients with tuberculosis.”
Dr. Rubin is affiliated with the department of immunology and infectious diseases at the Harvard School of Public Health, Boston. He had no relevant financial conflicts to disclose.
Although standard four-drug therapy has been shown to cure 90% of patients in several clinical trials, patients do relapse for reasons such as poor treatment adherence and “variations in the characteristics of the infected patients or the infecting pathogens,” wrote Eric J. Rubin, MD, in an accompanying editorial (N Engl J Med. 2018;379:882-3).
Current antibiotic susceptibility thresholds are often set by committees using models, said Dr. Rubin. “Given the uncertainties in modeling, as has been seen in clinical studies, these breakpoints can be imperfect predictors of treatment response.”
Dr. Rubin proposed that minimum inhibitory concentration (MIC) concentrations could be an alternative to in vitro testing as a predictor of treatment response.
“The clinical laboratory provides us not only with a breakpoint interpretation but also with raw data, a quantitative assessment of MIC values,” he noted. “These values can be thought of more as probabilities of successful therapy than as absolute thresholds, a change in attitude that may dispel a false sense of security about the choice of regimen in the treatment of patients with tuberculosis.”
Dr. Rubin is affiliated with the department of immunology and infectious diseases at the Harvard School of Public Health, Boston. He had no relevant financial conflicts to disclose.
Although standard four-drug therapy has been shown to cure 90% of patients in several clinical trials, patients do relapse for reasons such as poor treatment adherence and “variations in the characteristics of the infected patients or the infecting pathogens,” wrote Eric J. Rubin, MD, in an accompanying editorial (N Engl J Med. 2018;379:882-3).
Current antibiotic susceptibility thresholds are often set by committees using models, said Dr. Rubin. “Given the uncertainties in modeling, as has been seen in clinical studies, these breakpoints can be imperfect predictors of treatment response.”
Dr. Rubin proposed that minimum inhibitory concentration (MIC) concentrations could be an alternative to in vitro testing as a predictor of treatment response.
“The clinical laboratory provides us not only with a breakpoint interpretation but also with raw data, a quantitative assessment of MIC values,” he noted. “These values can be thought of more as probabilities of successful therapy than as absolute thresholds, a change in attitude that may dispel a false sense of security about the choice of regimen in the treatment of patients with tuberculosis.”
Dr. Rubin is affiliated with the department of immunology and infectious diseases at the Harvard School of Public Health, Boston. He had no relevant financial conflicts to disclose.
Higher pretreatment drug concentrations close to a resistance breakpoint for susceptibility were associated with greater relapse risk in TB, based on data from 54 patients who relapsed and 63 who were treated and cured.
“We postulated that drug-susceptible Mycobacterium tuberculosis might have a graded spectrum of susceptibilities that could be used to determine the risk of relapse,” wrote Roberto Colangeli, PhD, of Rutgers University, Newark, N.J., and his colleagues.
In a study published in the New England Journal of Medicine, the researchers examined pretreatment bacterial isolates from adults with TB who had experienced relapse and those who were cured. Using these isolates, they identified the minimum inhibitory concentration (MIC) – the lowest concentration of the drug that prevents visible bacterial growth in culture – for isoniazid and rifampin.
Overall, after controlling for other potential relapse risk factors, higher pretreatment MIC values for both isoniazid and rifampin were associated with an increased relapse risk. For isoniazid, the average MIC below the breakpoint was 0.0334 mcg/mL for relapsed patients and 0.0286 mcg/mL for cured patients. For rifampin, the average MIC below the breakpoint was 0.0695 mcg/mL for relapsed patients and 0.0453 mcg/mL for cured patients. The higher values for the relapsed versus cured patients were represented by factors of 1.17 and 1.53 for isoniazid and rifampin, respectively.
The average age of the patients was 41 years; 83% were men, and 35% were non-Hispanic white.
The study findings were limited by several factors, including the small sample size, retrospective design, and inability to test MIC values from primary cultures versus subcultures, the researchers wrote. However, the results suggest an impact of MIC values on treatment outcomes, and “additional studies that are performed in larger, well-defined prospective cohorts and that include MIC testing of pretreatment culture isolates will be useful to better validate these findings,”
The study was funded by the National Institute of Allergy and Infectious Diseases. Dr. Colangeli reported no financial conflicts. Dr. Alland disclosed funding from Cepheid and several current and pending patents in the United States and Europe, with some royalties paid to Cepheid.
SOURCE: Colangeli R et al. N Engl J Med. 2018;379:823-33.
Higher pretreatment drug concentrations close to a resistance breakpoint for susceptibility were associated with greater relapse risk in TB, based on data from 54 patients who relapsed and 63 who were treated and cured.
“We postulated that drug-susceptible Mycobacterium tuberculosis might have a graded spectrum of susceptibilities that could be used to determine the risk of relapse,” wrote Roberto Colangeli, PhD, of Rutgers University, Newark, N.J., and his colleagues.
In a study published in the New England Journal of Medicine, the researchers examined pretreatment bacterial isolates from adults with TB who had experienced relapse and those who were cured. Using these isolates, they identified the minimum inhibitory concentration (MIC) – the lowest concentration of the drug that prevents visible bacterial growth in culture – for isoniazid and rifampin.
Overall, after controlling for other potential relapse risk factors, higher pretreatment MIC values for both isoniazid and rifampin were associated with an increased relapse risk. For isoniazid, the average MIC below the breakpoint was 0.0334 mcg/mL for relapsed patients and 0.0286 mcg/mL for cured patients. For rifampin, the average MIC below the breakpoint was 0.0695 mcg/mL for relapsed patients and 0.0453 mcg/mL for cured patients. The higher values for the relapsed versus cured patients were represented by factors of 1.17 and 1.53 for isoniazid and rifampin, respectively.
The average age of the patients was 41 years; 83% were men, and 35% were non-Hispanic white.
The study findings were limited by several factors, including the small sample size, retrospective design, and inability to test MIC values from primary cultures versus subcultures, the researchers wrote. However, the results suggest an impact of MIC values on treatment outcomes, and “additional studies that are performed in larger, well-defined prospective cohorts and that include MIC testing of pretreatment culture isolates will be useful to better validate these findings,”
The study was funded by the National Institute of Allergy and Infectious Diseases. Dr. Colangeli reported no financial conflicts. Dr. Alland disclosed funding from Cepheid and several current and pending patents in the United States and Europe, with some royalties paid to Cepheid.
SOURCE: Colangeli R et al. N Engl J Med. 2018;379:823-33.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Key clinical point: Risk of TB relapse increased with higher pretreatment minimum inhibitory concentration values for either isoniazid or rifampin.
Major finding: The higher values for the relapsed versus cured patients were represented by factors of 1.17 and 1.53 for isoniazid and rifampin, respectively.
Study details: The data come from a retrospective study of isolates from 54 patients with TB who relapsed and 63 who were treated and cured.
Disclosures: The study was funded by the National Institute of Allergy and Infectious Diseases. Dr. Colangeli reported no financial conflicts. Dr. Alland disclosed funding from Cepheid and several current and pending patents in the United States and Europe, with some royalties paid to Cepheid.
Source: Colangeli R et al. N Engl J Med. 2018;379:823-33.
RSV-related risk of hospitalization higher in Down syndrome patients
reported Andrea Beckhaus, MD, and Jose Castro-Rodriguez, MD, PhD, at the Pontificia Universidad Católica de Chile in Santiago.
Because DS is the most common chromosomal disorder, affecting 1 in every 800 children born worldwide, and respiratory infections are the leading cause of hospitalization in children with DS, especially during the first year of life, the results of this study are important and have considerable consequences for public health, the authors observed.
The economic burden on families of children with DS and the health care systems that treat them is, not surprisingly, significantly higher given their sevenfold higher need for supplemental oxygen therapy, threefold likely increased risk of ICU admission, fivefold higher likely need for ventilator support, and average increased length of hospital stay by nearly 5 days.
In the 15 years from 1997 to 2012, RSV-related hospital charges for infants with DS increased by nearly 80% from $10,141 (US dollars) to $18,217, compared with charges for infants not at high risk, which increased by more than 60% from $6,983 to $11,273 during the same period, according to a study (PLoS One. 2016;11[4]:e0152208).
Dr. Beckhaus and Dr. Castro-Rodriguez conducted their systematic review and meta-analysis to evaluate RSV-associated morbidity in children with DS. Following a search of four electronic data bases, a total of 12 studies published between 2004 and 2017 across 10 different countries were identified, including six in Europe, three in Asia, two in the United States, and one in Latin America. Altogether, 3,662 children with DS and 1,145,509 without DS were included in the review.
“Any potential strategy to reduce RSV infection (e.g., prophylaxis with monoclonal antibodies or new vaccines) in children with DS could decrease their morbidity and mortality,” the authors noted. Specifically, they cited the humanized monoclonal antibody palivizumab, which is used for prophylaxis against RSV. Palivizumab is known to reduce hospitalizations in children at high risk from comorbid conditions, including chronic lung disease, hemodynamically significant congenital heart disease (CHD), neuromuscular disease, immunodeficiency, and prematurity. At present, palivizumab is not recommended by the American Academy of Pediatrics for routine use to prevent RSV infection in patients with DS who are not already qualified for other reasons because there are insufficient data to recommend routine prophylactic use of the drug in children with Down syndrome.
The authors cited recent studies in Canada and Japan that may warrant further reconsideration of the AAP’s recommendations, however. A fourfold lowering of RSV-related hospitalizations was observed during the first 2 years of life among 532 children with DS receiving palivizumab who were included in a recent prospective Canadian study. The drug also was found to be safe and effective for preventing lower respiratory tract infections caused by RSV in a recent Japanese multicenter postmarketing surveillance study that evaluated palivizumab prophylaxis for RSV infection in 138 children with DS who did not have hemodynamically significant CHD; only 2 of the children treated required hospitalization.
“More cost-utility studies used to determine the efficacy of RSV immunoprophylaxis in this specific high-risk patient population need to be done,” Dr. Beckhaus and Dr. Castro-Rodriguez recommended.
The review was not without limitations. Not all of the studies included subgroups of participants with DS with CHD and without CHD or other additional risk factors. However, when only those studies were considered that had data for participants with DS without other risk factors, almost all results were similar.
One key strength of the study concerned the study methodology. Using the Newcastle–Ottawa scale, the risk of bias among the 12 studies was generally low, the total number of participants was considerably high, and the outcomes selected had importance for the patients and also public health implications. “It is important to remark that the vast majority of the outcomes that were analyzed had no or unimportant bias,” they said.
Dr. Beckhaus and Dr. Castro-Rodriguez had no relevant disclosures to disclose.
SOURCE: Beckhaus A et al. J Pediatrics. 2018;142(3):e20180225.
reported Andrea Beckhaus, MD, and Jose Castro-Rodriguez, MD, PhD, at the Pontificia Universidad Católica de Chile in Santiago.
Because DS is the most common chromosomal disorder, affecting 1 in every 800 children born worldwide, and respiratory infections are the leading cause of hospitalization in children with DS, especially during the first year of life, the results of this study are important and have considerable consequences for public health, the authors observed.
The economic burden on families of children with DS and the health care systems that treat them is, not surprisingly, significantly higher given their sevenfold higher need for supplemental oxygen therapy, threefold likely increased risk of ICU admission, fivefold higher likely need for ventilator support, and average increased length of hospital stay by nearly 5 days.
In the 15 years from 1997 to 2012, RSV-related hospital charges for infants with DS increased by nearly 80% from $10,141 (US dollars) to $18,217, compared with charges for infants not at high risk, which increased by more than 60% from $6,983 to $11,273 during the same period, according to a study (PLoS One. 2016;11[4]:e0152208).
Dr. Beckhaus and Dr. Castro-Rodriguez conducted their systematic review and meta-analysis to evaluate RSV-associated morbidity in children with DS. Following a search of four electronic data bases, a total of 12 studies published between 2004 and 2017 across 10 different countries were identified, including six in Europe, three in Asia, two in the United States, and one in Latin America. Altogether, 3,662 children with DS and 1,145,509 without DS were included in the review.
“Any potential strategy to reduce RSV infection (e.g., prophylaxis with monoclonal antibodies or new vaccines) in children with DS could decrease their morbidity and mortality,” the authors noted. Specifically, they cited the humanized monoclonal antibody palivizumab, which is used for prophylaxis against RSV. Palivizumab is known to reduce hospitalizations in children at high risk from comorbid conditions, including chronic lung disease, hemodynamically significant congenital heart disease (CHD), neuromuscular disease, immunodeficiency, and prematurity. At present, palivizumab is not recommended by the American Academy of Pediatrics for routine use to prevent RSV infection in patients with DS who are not already qualified for other reasons because there are insufficient data to recommend routine prophylactic use of the drug in children with Down syndrome.
The authors cited recent studies in Canada and Japan that may warrant further reconsideration of the AAP’s recommendations, however. A fourfold lowering of RSV-related hospitalizations was observed during the first 2 years of life among 532 children with DS receiving palivizumab who were included in a recent prospective Canadian study. The drug also was found to be safe and effective for preventing lower respiratory tract infections caused by RSV in a recent Japanese multicenter postmarketing surveillance study that evaluated palivizumab prophylaxis for RSV infection in 138 children with DS who did not have hemodynamically significant CHD; only 2 of the children treated required hospitalization.
“More cost-utility studies used to determine the efficacy of RSV immunoprophylaxis in this specific high-risk patient population need to be done,” Dr. Beckhaus and Dr. Castro-Rodriguez recommended.
The review was not without limitations. Not all of the studies included subgroups of participants with DS with CHD and without CHD or other additional risk factors. However, when only those studies were considered that had data for participants with DS without other risk factors, almost all results were similar.
One key strength of the study concerned the study methodology. Using the Newcastle–Ottawa scale, the risk of bias among the 12 studies was generally low, the total number of participants was considerably high, and the outcomes selected had importance for the patients and also public health implications. “It is important to remark that the vast majority of the outcomes that were analyzed had no or unimportant bias,” they said.
Dr. Beckhaus and Dr. Castro-Rodriguez had no relevant disclosures to disclose.
SOURCE: Beckhaus A et al. J Pediatrics. 2018;142(3):e20180225.
reported Andrea Beckhaus, MD, and Jose Castro-Rodriguez, MD, PhD, at the Pontificia Universidad Católica de Chile in Santiago.
Because DS is the most common chromosomal disorder, affecting 1 in every 800 children born worldwide, and respiratory infections are the leading cause of hospitalization in children with DS, especially during the first year of life, the results of this study are important and have considerable consequences for public health, the authors observed.
The economic burden on families of children with DS and the health care systems that treat them is, not surprisingly, significantly higher given their sevenfold higher need for supplemental oxygen therapy, threefold likely increased risk of ICU admission, fivefold higher likely need for ventilator support, and average increased length of hospital stay by nearly 5 days.
In the 15 years from 1997 to 2012, RSV-related hospital charges for infants with DS increased by nearly 80% from $10,141 (US dollars) to $18,217, compared with charges for infants not at high risk, which increased by more than 60% from $6,983 to $11,273 during the same period, according to a study (PLoS One. 2016;11[4]:e0152208).
Dr. Beckhaus and Dr. Castro-Rodriguez conducted their systematic review and meta-analysis to evaluate RSV-associated morbidity in children with DS. Following a search of four electronic data bases, a total of 12 studies published between 2004 and 2017 across 10 different countries were identified, including six in Europe, three in Asia, two in the United States, and one in Latin America. Altogether, 3,662 children with DS and 1,145,509 without DS were included in the review.
“Any potential strategy to reduce RSV infection (e.g., prophylaxis with monoclonal antibodies or new vaccines) in children with DS could decrease their morbidity and mortality,” the authors noted. Specifically, they cited the humanized monoclonal antibody palivizumab, which is used for prophylaxis against RSV. Palivizumab is known to reduce hospitalizations in children at high risk from comorbid conditions, including chronic lung disease, hemodynamically significant congenital heart disease (CHD), neuromuscular disease, immunodeficiency, and prematurity. At present, palivizumab is not recommended by the American Academy of Pediatrics for routine use to prevent RSV infection in patients with DS who are not already qualified for other reasons because there are insufficient data to recommend routine prophylactic use of the drug in children with Down syndrome.
The authors cited recent studies in Canada and Japan that may warrant further reconsideration of the AAP’s recommendations, however. A fourfold lowering of RSV-related hospitalizations was observed during the first 2 years of life among 532 children with DS receiving palivizumab who were included in a recent prospective Canadian study. The drug also was found to be safe and effective for preventing lower respiratory tract infections caused by RSV in a recent Japanese multicenter postmarketing surveillance study that evaluated palivizumab prophylaxis for RSV infection in 138 children with DS who did not have hemodynamically significant CHD; only 2 of the children treated required hospitalization.
“More cost-utility studies used to determine the efficacy of RSV immunoprophylaxis in this specific high-risk patient population need to be done,” Dr. Beckhaus and Dr. Castro-Rodriguez recommended.
The review was not without limitations. Not all of the studies included subgroups of participants with DS with CHD and without CHD or other additional risk factors. However, when only those studies were considered that had data for participants with DS without other risk factors, almost all results were similar.
One key strength of the study concerned the study methodology. Using the Newcastle–Ottawa scale, the risk of bias among the 12 studies was generally low, the total number of participants was considerably high, and the outcomes selected had importance for the patients and also public health implications. “It is important to remark that the vast majority of the outcomes that were analyzed had no or unimportant bias,” they said.
Dr. Beckhaus and Dr. Castro-Rodriguez had no relevant disclosures to disclose.
SOURCE: Beckhaus A et al. J Pediatrics. 2018;142(3):e20180225.
FROM PEDIATRICS
Key clinical point: Strategies to reduce RSV are needed to decrease morbidity and mortality in children with Down syndrome.
Major finding: Down syndrome children with RSV-related hospitalization had sevenfold higher need for supplemental oxygen therapy, threefold likely increased risk of ICU admission, fivefold higher likely need for ventilator support, and average increased length of hospital stay by nearly 5 days.
Study details: Systematic review and 12-study meta-analysis of 3,662 children with DS and 1,145,509 without DS.
Disclosures: The authors had no relevant financial disclosures.
Source: Beckhaus A et al. J Pediatrics. 2018;142(3):e20180225.
FDA warns against azithromycin in blood or lymph node cancers
The Food and Drug Administration has issued a in patients with blood or lymph node cancers who have received donor stem cell transplants.
This use of azithromycin can lead to increased risk of cancer relapse and death in this population. The FDA is continuing to review data and is expected to issue further recommendations.
Patients with blood or lymph node cancers are at an increased risk of bronchiolitis obliterans syndrome after donor stem cell transplant; although azithromycin is not approved for prevention of this condition, the antibiotic is sometimes prescribed for that purpose.
A French study of 480 patients was undertaken to assess the effectiveness of this prophylaxis but revealed the increased risk of relapse and death and was halted 13 months after completing enrollment. The rate of cancer relapse was 32.9% in the azithromycin group and just 20.8% in the placebo group; the 2-year survival rate was 56.6% in the azithromycin group and 70.1% in the placebo group (JAMA 2017;318[6]:557-66).
Bronchiolitis obliterans syndrome is marked by inflammation and scarring of the airways that leads to severe shortness of breath and dry cough. There are no known effective antibiotic treatments for prophylaxis of the condition, according to the FDA.
FDA officials are advising physicians not to prescribe long-term azithromycin in this population. Patients who have had a stem cell transplant and are already taking the antibiotic, should consult a doctor before discontinuing.
The manufacturer of brand name azithromycin (Zithromax) has issued a Dear Healthcare Provider letter about the safety issue, and more information can be found in the FDA’s safety announcement.
The Food and Drug Administration has issued a in patients with blood or lymph node cancers who have received donor stem cell transplants.
This use of azithromycin can lead to increased risk of cancer relapse and death in this population. The FDA is continuing to review data and is expected to issue further recommendations.
Patients with blood or lymph node cancers are at an increased risk of bronchiolitis obliterans syndrome after donor stem cell transplant; although azithromycin is not approved for prevention of this condition, the antibiotic is sometimes prescribed for that purpose.
A French study of 480 patients was undertaken to assess the effectiveness of this prophylaxis but revealed the increased risk of relapse and death and was halted 13 months after completing enrollment. The rate of cancer relapse was 32.9% in the azithromycin group and just 20.8% in the placebo group; the 2-year survival rate was 56.6% in the azithromycin group and 70.1% in the placebo group (JAMA 2017;318[6]:557-66).
Bronchiolitis obliterans syndrome is marked by inflammation and scarring of the airways that leads to severe shortness of breath and dry cough. There are no known effective antibiotic treatments for prophylaxis of the condition, according to the FDA.
FDA officials are advising physicians not to prescribe long-term azithromycin in this population. Patients who have had a stem cell transplant and are already taking the antibiotic, should consult a doctor before discontinuing.
The manufacturer of brand name azithromycin (Zithromax) has issued a Dear Healthcare Provider letter about the safety issue, and more information can be found in the FDA’s safety announcement.
The Food and Drug Administration has issued a in patients with blood or lymph node cancers who have received donor stem cell transplants.
This use of azithromycin can lead to increased risk of cancer relapse and death in this population. The FDA is continuing to review data and is expected to issue further recommendations.
Patients with blood or lymph node cancers are at an increased risk of bronchiolitis obliterans syndrome after donor stem cell transplant; although azithromycin is not approved for prevention of this condition, the antibiotic is sometimes prescribed for that purpose.
A French study of 480 patients was undertaken to assess the effectiveness of this prophylaxis but revealed the increased risk of relapse and death and was halted 13 months after completing enrollment. The rate of cancer relapse was 32.9% in the azithromycin group and just 20.8% in the placebo group; the 2-year survival rate was 56.6% in the azithromycin group and 70.1% in the placebo group (JAMA 2017;318[6]:557-66).
Bronchiolitis obliterans syndrome is marked by inflammation and scarring of the airways that leads to severe shortness of breath and dry cough. There are no known effective antibiotic treatments for prophylaxis of the condition, according to the FDA.
FDA officials are advising physicians not to prescribe long-term azithromycin in this population. Patients who have had a stem cell transplant and are already taking the antibiotic, should consult a doctor before discontinuing.
The manufacturer of brand name azithromycin (Zithromax) has issued a Dear Healthcare Provider letter about the safety issue, and more information can be found in the FDA’s safety announcement.