User login
Antibiotic resistance rises among pneumococcus strains in kids
Antibiotic resistance in strains of Streptococcus pneumoniae has been rising since 2013 because of changing susceptibility profiles, based on data from 1,201 isolates collected from 448 children in primary care settings.
“New strains expressing capsular serotypes not included in the 13-valent pneumococcal conjugate vaccine are emerging to cause disease, and strains that acquire antibiotic resistance are increasing in frequency due to their survival of the fittest advantage,” wrote Ravinder Kaur, PhD, of Rochester (N.Y.) General Hospital Research Institute, and colleagues.
Similar Darwinian principles occurred after the introduction of PCV-7, the study authors added.
In a prospective cohort study published in Clinical Infectious Diseases, the researchers reviewed 1,201 isolates collected from the nasopharynx during healthy periods, and from the nasopharynx and middle ear fluid (MEF) during episodes of acute otitis media, in children aged 6-36 months who were seen in primary care settings.
The isolates were collected during 2006-2016 to reflect the pre- and post-PCV13 era. Children received PCV-7 from 2006 until April 2010, and received PCV-13 after April 2010.
Overall, the number of acute otitis media (AOM) cases caused by S. pneumoniae was not significantly different between the PCV-7 and PCV-13 eras, nor was the frequency of pneumococci identified in the nasopharynx during healthy visits and visits at the start of an AOM infection.
The researchers examined susceptibility using minimum inhibitory concentrations (MIC). During healthy visits, the MIC50 of isolated pneumococci was low (no greater than 0.06 mcg/mL) for all four beta-lactam drugs tested. And it didn’t change significantly over the study years.
In contrast, among the nasopharyngeal and MEF isolates during AOM, the MIC50 to penicillin, amoxicillin, ceftriaxone, and meropenem during 2013-2016 rose significantly, the investigators said.
A change in antibiotic susceptibility within a subtype also contributed to the development of PCV-13 resistance.
The study authors identified three serotypes that affected the changes in susceptibility in their study population. Serotypes 35B and 35F increased their beta-lactam resistance during 2013-2016, and serotype 11A had a higher MIC to quinolones and became more prevalent during 2013-2016. Those three serotypes accounted for most of the change in antibiotic susceptibility, the researchers said.
In addition, “the frequency of strains resistant to penicillin and amoxicillin decreased with the introduction of PCV-13, but rebounded to levels similar to those before PCV-13 introduction by 2015-2016,” the investigators noted.
The study findings were limited by several factors, including the homogeneous study population and potential lack of generalizability to other settings. In addition, the researchers did not study antibiotic consumption or antibiotic treatment failure, and they could not account for potential AOM cases that may have been treated in settings other than primary care.
However, the investigators said the results support the need for additional studies and attention to the development of the next generation of PCVs, the PCV-15 and PCV-20. Both include serotypes 22F and 33F, but neither includes 35B or 35F. The PCV-20 also includes 11A and 15B.
The study was supported in part by the National Institutes of Health and Sanofi Pasteur. Some isolates collected during the 2010-2013 time period were part of a study supported by Pfizer. The researchers had no relevant financial conflicts to disclose.
SOURCE: Kaur R et al. Clin Inf Dis. 2020 Feb 18. doi: 10.1093/cid/ciaa157.
Dr. Kaur and colleagues report their analysis of pneumococcal resistance among nasopharyngeal and middle ear isolates (90% nasopharyngeal and 10% middle ear) collected between 2008 and 2016. They demonstrate the dominant role that nonvaccine serotypes play in carriage and acute otitis media (AOM) in children, and by extension potentially the entire spectrum of pneumococcal disease in the 13-valent pneumococcal conjugate vaccine (PCV13) era. Nonsusceptibility to beta-lactams was reported for one-third of isolates with the increase in the most recent reported years (2013-2016).
What are the implications for treatment of pneumococcal infections? For AOM, amoxicillin minimum inhibitory concentrations (MIC) were all less than 4 mcg/mL, which is the pharmacodynamic breakpoint for high-dose (90 mg/kg per day) AOM regimens; these data support continued use of high-dose amoxicillin for children with AOM that requires antimicrobial treatment. Resistance to macrolides (erythromycin and likely azithromycin) occurred in approximately one-third of isolates; however, in contrast to beta-lactams (amoxicillin), higher macrolide doses do not overcome resistance. Thus macrolide use for AOM appears limited to those with beta-lactam allergy and no better alternative drug, i.e., expect failure in one-third of AOM patients if macrolides are used. For ceftriaxone, no 2013-2016 isolate had a MIC over 0.5 mcg/mL, implying that ceftriaxone remains appropriate first-line therapy for serious pneumococcal disease and effective for pneumococcal AOM when oral drugs have failed or are not an option because of repeated emesis. Interestingly, trimethoprim/sulfamethoxazole (T/S) had lower resistance rates against the nonvaccine “bad boy” serogroup 35 (8%-15%), compared with cephalosporins (32%-57%). Perhaps we are back to the future and T/S will again have a role against pneumococcal AOM. Of note, no isolate was resistant to levofloxacin or linezolid. Linezolid or macrolide use alone must be considered with the caveat that nontypeable Haemophilus influenzae now likely surpasses pneumococcus as an AOM pathogen, and neither drug class is active against nontypeable H. influenzae.
What are the implications for prevention? This is one of many studies in the post-PCV era reporting serotype replacement with nonvaccine serotypes. But most prior studies reported reduced overall disease burden; in other words, the absolute number of pneumococcal infections was reduced, but residual AOM nonvaccine types dominated as the etiology. The current study, however, suggests that the overall number of AOM episodes may not be less because increases in AOM caused by nonvaccine serotypes may be offsetting declines in AOM caused by vaccine serotypes. This concept contrasts to multiple large epidemiologic studies demonstrating a decline in overall incidence of AOM office visits/episodes and several Israeli studies reporting a decline in pneumococcal AOM in children who warrant tympanocentesis. These new data are food for thought, but antibiotic resistance can vary regionally, so confirmation based on data from other regions seems warranted.
Next-generation vaccines will need to consider which serotypes are prevalent in pneumococcal disease, including AOM, as we continue into the PCV13 era. However, serotypes causing invasive pneumococcal disease and pneumonia would be higher priorities than AOM. Indeed, several candidate PCV vaccines are currently in clinical trials adding up to seven serotypes, including most of the newly emerging invasive disease serotypes. One downside to the newer PCVs is lack of serogroup 35, a prominent culprit in AOM resistance in the current report.
Stephen I. Pelton, MD, is professor of pediatrics and epidemiology at Boston University and senior attending physician at Boston Medical Center. Christopher J. Harrison, MD, is professor of pediatrics and pediatric infectious diseases at Children’s Mercy Hospital–Kansas City, Mo. Dr. Pelton has received honorarium from Merck Vaccines, Pfizer, and Sanofi for participation in advisory board meeting on pneumococcal vaccine and/or membership on the Data and Safety Monitoring Board. Boston Medical Center has received investigator-initiated research grants from Merck Vaccines and Pfizer.
Children’s Mercy Hospital – Kansas City Boston Medical Center has received funding from GlaxoSmithKline, Merck, and Pfizer for research vaccine studies, and from Pfizer and Merck for investigator-initiated research grants for in vitro pneumococcal investigations on which Dr. Harrison is an investigator.
Dr. Kaur and colleagues report their analysis of pneumococcal resistance among nasopharyngeal and middle ear isolates (90% nasopharyngeal and 10% middle ear) collected between 2008 and 2016. They demonstrate the dominant role that nonvaccine serotypes play in carriage and acute otitis media (AOM) in children, and by extension potentially the entire spectrum of pneumococcal disease in the 13-valent pneumococcal conjugate vaccine (PCV13) era. Nonsusceptibility to beta-lactams was reported for one-third of isolates with the increase in the most recent reported years (2013-2016).
What are the implications for treatment of pneumococcal infections? For AOM, amoxicillin minimum inhibitory concentrations (MIC) were all less than 4 mcg/mL, which is the pharmacodynamic breakpoint for high-dose (90 mg/kg per day) AOM regimens; these data support continued use of high-dose amoxicillin for children with AOM that requires antimicrobial treatment. Resistance to macrolides (erythromycin and likely azithromycin) occurred in approximately one-third of isolates; however, in contrast to beta-lactams (amoxicillin), higher macrolide doses do not overcome resistance. Thus macrolide use for AOM appears limited to those with beta-lactam allergy and no better alternative drug, i.e., expect failure in one-third of AOM patients if macrolides are used. For ceftriaxone, no 2013-2016 isolate had a MIC over 0.5 mcg/mL, implying that ceftriaxone remains appropriate first-line therapy for serious pneumococcal disease and effective for pneumococcal AOM when oral drugs have failed or are not an option because of repeated emesis. Interestingly, trimethoprim/sulfamethoxazole (T/S) had lower resistance rates against the nonvaccine “bad boy” serogroup 35 (8%-15%), compared with cephalosporins (32%-57%). Perhaps we are back to the future and T/S will again have a role against pneumococcal AOM. Of note, no isolate was resistant to levofloxacin or linezolid. Linezolid or macrolide use alone must be considered with the caveat that nontypeable Haemophilus influenzae now likely surpasses pneumococcus as an AOM pathogen, and neither drug class is active against nontypeable H. influenzae.
What are the implications for prevention? This is one of many studies in the post-PCV era reporting serotype replacement with nonvaccine serotypes. But most prior studies reported reduced overall disease burden; in other words, the absolute number of pneumococcal infections was reduced, but residual AOM nonvaccine types dominated as the etiology. The current study, however, suggests that the overall number of AOM episodes may not be less because increases in AOM caused by nonvaccine serotypes may be offsetting declines in AOM caused by vaccine serotypes. This concept contrasts to multiple large epidemiologic studies demonstrating a decline in overall incidence of AOM office visits/episodes and several Israeli studies reporting a decline in pneumococcal AOM in children who warrant tympanocentesis. These new data are food for thought, but antibiotic resistance can vary regionally, so confirmation based on data from other regions seems warranted.
Next-generation vaccines will need to consider which serotypes are prevalent in pneumococcal disease, including AOM, as we continue into the PCV13 era. However, serotypes causing invasive pneumococcal disease and pneumonia would be higher priorities than AOM. Indeed, several candidate PCV vaccines are currently in clinical trials adding up to seven serotypes, including most of the newly emerging invasive disease serotypes. One downside to the newer PCVs is lack of serogroup 35, a prominent culprit in AOM resistance in the current report.
Stephen I. Pelton, MD, is professor of pediatrics and epidemiology at Boston University and senior attending physician at Boston Medical Center. Christopher J. Harrison, MD, is professor of pediatrics and pediatric infectious diseases at Children’s Mercy Hospital–Kansas City, Mo. Dr. Pelton has received honorarium from Merck Vaccines, Pfizer, and Sanofi for participation in advisory board meeting on pneumococcal vaccine and/or membership on the Data and Safety Monitoring Board. Boston Medical Center has received investigator-initiated research grants from Merck Vaccines and Pfizer.
Children’s Mercy Hospital – Kansas City Boston Medical Center has received funding from GlaxoSmithKline, Merck, and Pfizer for research vaccine studies, and from Pfizer and Merck for investigator-initiated research grants for in vitro pneumococcal investigations on which Dr. Harrison is an investigator.
Dr. Kaur and colleagues report their analysis of pneumococcal resistance among nasopharyngeal and middle ear isolates (90% nasopharyngeal and 10% middle ear) collected between 2008 and 2016. They demonstrate the dominant role that nonvaccine serotypes play in carriage and acute otitis media (AOM) in children, and by extension potentially the entire spectrum of pneumococcal disease in the 13-valent pneumococcal conjugate vaccine (PCV13) era. Nonsusceptibility to beta-lactams was reported for one-third of isolates with the increase in the most recent reported years (2013-2016).
What are the implications for treatment of pneumococcal infections? For AOM, amoxicillin minimum inhibitory concentrations (MIC) were all less than 4 mcg/mL, which is the pharmacodynamic breakpoint for high-dose (90 mg/kg per day) AOM regimens; these data support continued use of high-dose amoxicillin for children with AOM that requires antimicrobial treatment. Resistance to macrolides (erythromycin and likely azithromycin) occurred in approximately one-third of isolates; however, in contrast to beta-lactams (amoxicillin), higher macrolide doses do not overcome resistance. Thus macrolide use for AOM appears limited to those with beta-lactam allergy and no better alternative drug, i.e., expect failure in one-third of AOM patients if macrolides are used. For ceftriaxone, no 2013-2016 isolate had a MIC over 0.5 mcg/mL, implying that ceftriaxone remains appropriate first-line therapy for serious pneumococcal disease and effective for pneumococcal AOM when oral drugs have failed or are not an option because of repeated emesis. Interestingly, trimethoprim/sulfamethoxazole (T/S) had lower resistance rates against the nonvaccine “bad boy” serogroup 35 (8%-15%), compared with cephalosporins (32%-57%). Perhaps we are back to the future and T/S will again have a role against pneumococcal AOM. Of note, no isolate was resistant to levofloxacin or linezolid. Linezolid or macrolide use alone must be considered with the caveat that nontypeable Haemophilus influenzae now likely surpasses pneumococcus as an AOM pathogen, and neither drug class is active against nontypeable H. influenzae.
What are the implications for prevention? This is one of many studies in the post-PCV era reporting serotype replacement with nonvaccine serotypes. But most prior studies reported reduced overall disease burden; in other words, the absolute number of pneumococcal infections was reduced, but residual AOM nonvaccine types dominated as the etiology. The current study, however, suggests that the overall number of AOM episodes may not be less because increases in AOM caused by nonvaccine serotypes may be offsetting declines in AOM caused by vaccine serotypes. This concept contrasts to multiple large epidemiologic studies demonstrating a decline in overall incidence of AOM office visits/episodes and several Israeli studies reporting a decline in pneumococcal AOM in children who warrant tympanocentesis. These new data are food for thought, but antibiotic resistance can vary regionally, so confirmation based on data from other regions seems warranted.
Next-generation vaccines will need to consider which serotypes are prevalent in pneumococcal disease, including AOM, as we continue into the PCV13 era. However, serotypes causing invasive pneumococcal disease and pneumonia would be higher priorities than AOM. Indeed, several candidate PCV vaccines are currently in clinical trials adding up to seven serotypes, including most of the newly emerging invasive disease serotypes. One downside to the newer PCVs is lack of serogroup 35, a prominent culprit in AOM resistance in the current report.
Stephen I. Pelton, MD, is professor of pediatrics and epidemiology at Boston University and senior attending physician at Boston Medical Center. Christopher J. Harrison, MD, is professor of pediatrics and pediatric infectious diseases at Children’s Mercy Hospital–Kansas City, Mo. Dr. Pelton has received honorarium from Merck Vaccines, Pfizer, and Sanofi for participation in advisory board meeting on pneumococcal vaccine and/or membership on the Data and Safety Monitoring Board. Boston Medical Center has received investigator-initiated research grants from Merck Vaccines and Pfizer.
Children’s Mercy Hospital – Kansas City Boston Medical Center has received funding from GlaxoSmithKline, Merck, and Pfizer for research vaccine studies, and from Pfizer and Merck for investigator-initiated research grants for in vitro pneumococcal investigations on which Dr. Harrison is an investigator.
Antibiotic resistance in strains of Streptococcus pneumoniae has been rising since 2013 because of changing susceptibility profiles, based on data from 1,201 isolates collected from 448 children in primary care settings.
“New strains expressing capsular serotypes not included in the 13-valent pneumococcal conjugate vaccine are emerging to cause disease, and strains that acquire antibiotic resistance are increasing in frequency due to their survival of the fittest advantage,” wrote Ravinder Kaur, PhD, of Rochester (N.Y.) General Hospital Research Institute, and colleagues.
Similar Darwinian principles occurred after the introduction of PCV-7, the study authors added.
In a prospective cohort study published in Clinical Infectious Diseases, the researchers reviewed 1,201 isolates collected from the nasopharynx during healthy periods, and from the nasopharynx and middle ear fluid (MEF) during episodes of acute otitis media, in children aged 6-36 months who were seen in primary care settings.
The isolates were collected during 2006-2016 to reflect the pre- and post-PCV13 era. Children received PCV-7 from 2006 until April 2010, and received PCV-13 after April 2010.
Overall, the number of acute otitis media (AOM) cases caused by S. pneumoniae was not significantly different between the PCV-7 and PCV-13 eras, nor was the frequency of pneumococci identified in the nasopharynx during healthy visits and visits at the start of an AOM infection.
The researchers examined susceptibility using minimum inhibitory concentrations (MIC). During healthy visits, the MIC50 of isolated pneumococci was low (no greater than 0.06 mcg/mL) for all four beta-lactam drugs tested. And it didn’t change significantly over the study years.
In contrast, among the nasopharyngeal and MEF isolates during AOM, the MIC50 to penicillin, amoxicillin, ceftriaxone, and meropenem during 2013-2016 rose significantly, the investigators said.
A change in antibiotic susceptibility within a subtype also contributed to the development of PCV-13 resistance.
The study authors identified three serotypes that affected the changes in susceptibility in their study population. Serotypes 35B and 35F increased their beta-lactam resistance during 2013-2016, and serotype 11A had a higher MIC to quinolones and became more prevalent during 2013-2016. Those three serotypes accounted for most of the change in antibiotic susceptibility, the researchers said.
In addition, “the frequency of strains resistant to penicillin and amoxicillin decreased with the introduction of PCV-13, but rebounded to levels similar to those before PCV-13 introduction by 2015-2016,” the investigators noted.
The study findings were limited by several factors, including the homogeneous study population and potential lack of generalizability to other settings. In addition, the researchers did not study antibiotic consumption or antibiotic treatment failure, and they could not account for potential AOM cases that may have been treated in settings other than primary care.
However, the investigators said the results support the need for additional studies and attention to the development of the next generation of PCVs, the PCV-15 and PCV-20. Both include serotypes 22F and 33F, but neither includes 35B or 35F. The PCV-20 also includes 11A and 15B.
The study was supported in part by the National Institutes of Health and Sanofi Pasteur. Some isolates collected during the 2010-2013 time period were part of a study supported by Pfizer. The researchers had no relevant financial conflicts to disclose.
SOURCE: Kaur R et al. Clin Inf Dis. 2020 Feb 18. doi: 10.1093/cid/ciaa157.
Antibiotic resistance in strains of Streptococcus pneumoniae has been rising since 2013 because of changing susceptibility profiles, based on data from 1,201 isolates collected from 448 children in primary care settings.
“New strains expressing capsular serotypes not included in the 13-valent pneumococcal conjugate vaccine are emerging to cause disease, and strains that acquire antibiotic resistance are increasing in frequency due to their survival of the fittest advantage,” wrote Ravinder Kaur, PhD, of Rochester (N.Y.) General Hospital Research Institute, and colleagues.
Similar Darwinian principles occurred after the introduction of PCV-7, the study authors added.
In a prospective cohort study published in Clinical Infectious Diseases, the researchers reviewed 1,201 isolates collected from the nasopharynx during healthy periods, and from the nasopharynx and middle ear fluid (MEF) during episodes of acute otitis media, in children aged 6-36 months who were seen in primary care settings.
The isolates were collected during 2006-2016 to reflect the pre- and post-PCV13 era. Children received PCV-7 from 2006 until April 2010, and received PCV-13 after April 2010.
Overall, the number of acute otitis media (AOM) cases caused by S. pneumoniae was not significantly different between the PCV-7 and PCV-13 eras, nor was the frequency of pneumococci identified in the nasopharynx during healthy visits and visits at the start of an AOM infection.
The researchers examined susceptibility using minimum inhibitory concentrations (MIC). During healthy visits, the MIC50 of isolated pneumococci was low (no greater than 0.06 mcg/mL) for all four beta-lactam drugs tested. And it didn’t change significantly over the study years.
In contrast, among the nasopharyngeal and MEF isolates during AOM, the MIC50 to penicillin, amoxicillin, ceftriaxone, and meropenem during 2013-2016 rose significantly, the investigators said.
A change in antibiotic susceptibility within a subtype also contributed to the development of PCV-13 resistance.
The study authors identified three serotypes that affected the changes in susceptibility in their study population. Serotypes 35B and 35F increased their beta-lactam resistance during 2013-2016, and serotype 11A had a higher MIC to quinolones and became more prevalent during 2013-2016. Those three serotypes accounted for most of the change in antibiotic susceptibility, the researchers said.
In addition, “the frequency of strains resistant to penicillin and amoxicillin decreased with the introduction of PCV-13, but rebounded to levels similar to those before PCV-13 introduction by 2015-2016,” the investigators noted.
The study findings were limited by several factors, including the homogeneous study population and potential lack of generalizability to other settings. In addition, the researchers did not study antibiotic consumption or antibiotic treatment failure, and they could not account for potential AOM cases that may have been treated in settings other than primary care.
However, the investigators said the results support the need for additional studies and attention to the development of the next generation of PCVs, the PCV-15 and PCV-20. Both include serotypes 22F and 33F, but neither includes 35B or 35F. The PCV-20 also includes 11A and 15B.
The study was supported in part by the National Institutes of Health and Sanofi Pasteur. Some isolates collected during the 2010-2013 time period were part of a study supported by Pfizer. The researchers had no relevant financial conflicts to disclose.
SOURCE: Kaur R et al. Clin Inf Dis. 2020 Feb 18. doi: 10.1093/cid/ciaa157.
FROM CLINICAL INFECTIOUS DISEASES
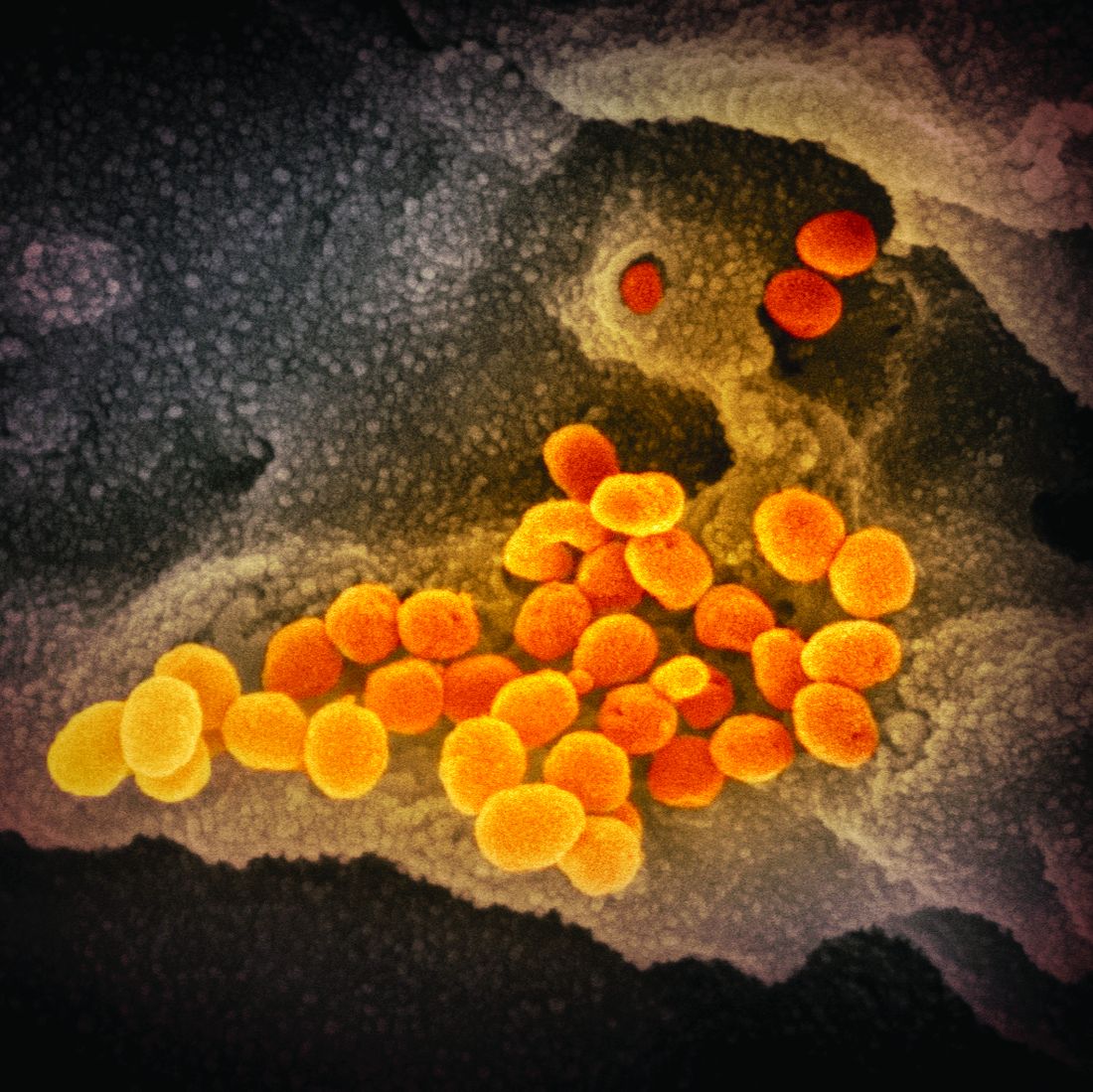
U.S. reports first death from COVID-19, possible outbreak at long-term care facility
The first death in the United States from the novel coronavirus (COVID-19) was a Washington state man in his 50s who had underlying health conditions, state health officials announced on Feb 29. At the same time, officials there are investigating a possible COVID-19 outbreak at a long-term care facility.
Washington state officials reported two other presumptive positive cases of COVID-19, both of whom are associated with LifeCare of Kirkland, Washington. One is a woman in her 70s who is a resident at the facility and the other is a woman in her 40s who is a health care worker at the facility.
Additionally, many residents and staff members at the facility have reported respiratory symptoms, according to Jeff Duchin, MD, health officer for public health in Seattle and King County. Among the more than 100 residents at the facility, 27 have respiratory symptoms; while among the 180 staff members, 25 have reported symptoms.
Overall, these reports bring the total number of U.S. COVID-19 cases detected by the public health system to 22, though that number is expected to climb as these investigations continue.
The general risk to the American public is still low, including residents in long-term care facilities, Nancy Messonnier, MD, director of the National Center for Immunization and Respiratory Diseases at the Centers for Disease Control and Prevention, said during the Feb. 29 press briefing. Older people are are higher risk, however, and long-term care facilities should emphasize handwashing and the early identification of individuals with symptoms.
Dr. Duchin added that health care workers who are sick should stay home and that visitors should be screened for symptoms, the same advice offered to limit the spread of influenza at long-term care facilities.

The CDC briefing comes after President Trump held his own press conference at the White House where he identified the person who had died as being a woman in her 50s who was medically at risk.
During that press conference, Anthony S. Fauci, MD, director of the National Institute of Allergy and Infectious Diseases, said that the current pattern of disease with COVID-19 suggests that 75%-80% of patients will have mild illness and recover, while 15%-20% will require advanced medical care.
For the most part, the more serious cases will occur in those who are elderly or have underlying medical conditions. There is “no indication” that individuals who recover from the virus are becoming re-infected, Dr. Fauci said.
The administration also announced a series of actions aimed at slowing the spread of the virus and responding to it. On March 2, President Trump will meet with leaders in the pharmaceutical industry at the White House to discuss vaccine development. The administration is also working to ensure an adequate supply of face masks. Vice President Mike Pence said there are currently more than 40 million masks available, but that the administration has received promises of 35 million more masks per month from manufacturers. Access to masks will be prioritized for high-risk health care workers, Vice President Pence said. “The average American does not need to go out and buy a mask,” he added.
Additionally, Vice President Pence announced new travel restrictions with Iran that would bar entry to the United States for any foreign national who visited Iran in the last 14 days. The federal government is also advising Americans not to travel to the regions in Italy and South Korea that have been most affected by COVID-19. The government is also working with officials in Italy and South Korea to conduct medical screening of anyone coming into the United States from those countries.
The first death in the United States from the novel coronavirus (COVID-19) was a Washington state man in his 50s who had underlying health conditions, state health officials announced on Feb 29. At the same time, officials there are investigating a possible COVID-19 outbreak at a long-term care facility.
Washington state officials reported two other presumptive positive cases of COVID-19, both of whom are associated with LifeCare of Kirkland, Washington. One is a woman in her 70s who is a resident at the facility and the other is a woman in her 40s who is a health care worker at the facility.
Additionally, many residents and staff members at the facility have reported respiratory symptoms, according to Jeff Duchin, MD, health officer for public health in Seattle and King County. Among the more than 100 residents at the facility, 27 have respiratory symptoms; while among the 180 staff members, 25 have reported symptoms.
Overall, these reports bring the total number of U.S. COVID-19 cases detected by the public health system to 22, though that number is expected to climb as these investigations continue.
The general risk to the American public is still low, including residents in long-term care facilities, Nancy Messonnier, MD, director of the National Center for Immunization and Respiratory Diseases at the Centers for Disease Control and Prevention, said during the Feb. 29 press briefing. Older people are are higher risk, however, and long-term care facilities should emphasize handwashing and the early identification of individuals with symptoms.
Dr. Duchin added that health care workers who are sick should stay home and that visitors should be screened for symptoms, the same advice offered to limit the spread of influenza at long-term care facilities.

The CDC briefing comes after President Trump held his own press conference at the White House where he identified the person who had died as being a woman in her 50s who was medically at risk.
During that press conference, Anthony S. Fauci, MD, director of the National Institute of Allergy and Infectious Diseases, said that the current pattern of disease with COVID-19 suggests that 75%-80% of patients will have mild illness and recover, while 15%-20% will require advanced medical care.
For the most part, the more serious cases will occur in those who are elderly or have underlying medical conditions. There is “no indication” that individuals who recover from the virus are becoming re-infected, Dr. Fauci said.
The administration also announced a series of actions aimed at slowing the spread of the virus and responding to it. On March 2, President Trump will meet with leaders in the pharmaceutical industry at the White House to discuss vaccine development. The administration is also working to ensure an adequate supply of face masks. Vice President Mike Pence said there are currently more than 40 million masks available, but that the administration has received promises of 35 million more masks per month from manufacturers. Access to masks will be prioritized for high-risk health care workers, Vice President Pence said. “The average American does not need to go out and buy a mask,” he added.
Additionally, Vice President Pence announced new travel restrictions with Iran that would bar entry to the United States for any foreign national who visited Iran in the last 14 days. The federal government is also advising Americans not to travel to the regions in Italy and South Korea that have been most affected by COVID-19. The government is also working with officials in Italy and South Korea to conduct medical screening of anyone coming into the United States from those countries.
The first death in the United States from the novel coronavirus (COVID-19) was a Washington state man in his 50s who had underlying health conditions, state health officials announced on Feb 29. At the same time, officials there are investigating a possible COVID-19 outbreak at a long-term care facility.
Washington state officials reported two other presumptive positive cases of COVID-19, both of whom are associated with LifeCare of Kirkland, Washington. One is a woman in her 70s who is a resident at the facility and the other is a woman in her 40s who is a health care worker at the facility.
Additionally, many residents and staff members at the facility have reported respiratory symptoms, according to Jeff Duchin, MD, health officer for public health in Seattle and King County. Among the more than 100 residents at the facility, 27 have respiratory symptoms; while among the 180 staff members, 25 have reported symptoms.
Overall, these reports bring the total number of U.S. COVID-19 cases detected by the public health system to 22, though that number is expected to climb as these investigations continue.
The general risk to the American public is still low, including residents in long-term care facilities, Nancy Messonnier, MD, director of the National Center for Immunization and Respiratory Diseases at the Centers for Disease Control and Prevention, said during the Feb. 29 press briefing. Older people are are higher risk, however, and long-term care facilities should emphasize handwashing and the early identification of individuals with symptoms.
Dr. Duchin added that health care workers who are sick should stay home and that visitors should be screened for symptoms, the same advice offered to limit the spread of influenza at long-term care facilities.

The CDC briefing comes after President Trump held his own press conference at the White House where he identified the person who had died as being a woman in her 50s who was medically at risk.
During that press conference, Anthony S. Fauci, MD, director of the National Institute of Allergy and Infectious Diseases, said that the current pattern of disease with COVID-19 suggests that 75%-80% of patients will have mild illness and recover, while 15%-20% will require advanced medical care.
For the most part, the more serious cases will occur in those who are elderly or have underlying medical conditions. There is “no indication” that individuals who recover from the virus are becoming re-infected, Dr. Fauci said.
The administration also announced a series of actions aimed at slowing the spread of the virus and responding to it. On March 2, President Trump will meet with leaders in the pharmaceutical industry at the White House to discuss vaccine development. The administration is also working to ensure an adequate supply of face masks. Vice President Mike Pence said there are currently more than 40 million masks available, but that the administration has received promises of 35 million more masks per month from manufacturers. Access to masks will be prioritized for high-risk health care workers, Vice President Pence said. “The average American does not need to go out and buy a mask,” he added.
Additionally, Vice President Pence announced new travel restrictions with Iran that would bar entry to the United States for any foreign national who visited Iran in the last 14 days. The federal government is also advising Americans not to travel to the regions in Italy and South Korea that have been most affected by COVID-19. The government is also working with officials in Italy and South Korea to conduct medical screening of anyone coming into the United States from those countries.
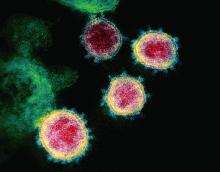
CDC revises COVID-19 test kits, broadens ‘person under investigation’ definition
In a telebriefing on the COVID-19 outbreak, Nancy Messonnier, MD, director of the National Center for Immunization and Respiratory Diseases at .

The definition has been revised “to meet the needs of this rapidly evolving situation,” she said. The new PUI definition includes travel to more geographic areas to reflect this past week’s marked uptick in coronavirus activity in Italy and Iran. In addition to these countries and China, recent travel to Japan or South Korea also constitutes an epidemiologic risk factor which, in conjunction with clinical features, warrant an individual being classified as a PUI. These five countries each now have widespread person-to-person transmission of the virus.
Dr. Messonnier left open the possibility that the PUI definition would continue to evolve if such transmission within communities becomes more common. Asked whether the small number of U.S. cases thus might be an artifact of low test volumes, she said, “We aggressively controlled our borders to slow the spread. This was an intentional U.S. strategy. The CDC has always had the capacity to test rapidly from the time the sequence was available. ...We have been testing aggressively.”
The original PUI definition, she explained, emphasized individuals with fever, cough, or trouble breathing who had traveled recently from areas with COVID-19 activity, in particular China’s Hubei province. “We have been most focused on symptomatic people who are closely linked to, or who had, travel history, but our criteria also allow for clinical discretion,” she said. “There is no substitute for an astute clinician on the front lines of patient care.”
The first COVID-19 case from person-to-person spread was reported on Feb. 27. “At this time, we don’t know how or where this person became infected,” said Dr. Messonnier, although investigations are still underway. She responded to a question about whether the CDC delayed allowing COVID-19 testing for the patient for several days, as was reported in some media accounts. “According to CDC records, the first call we got was Feb. 23,” when public health officials in California reported a severely ill person with no travel abroad and no known contacts with individuals that would trigger suspicions for coronavirus. The CDC recommended COVID-19 testing on that day, she said.
Dr. Messonnier declined to answer questions about a whistleblower report alleging improper training and inadequate protective measures for Department of Health & Human Services workers at the quarantine center at Travis Air Force Base, Calif.
Dr. Messonnier said that the CDC has been working closely with the Food and Drug Administration to address problems with the COVID-19 test kits that were unusable because of a large number of indeterminate results. The two agencies together have determined that of the three reactions that were initially deemed necessary for a definitive COVID-19 diagnosis, just two are sufficient, so new kits that omit the problematic chemical are being manufactured and distributed.
These new kits are rapidly being made available; the goal, said Dr. Messonnier, is to have to state and local public health departments equipped with test kits by about March 7.
As local tests become available, the most updated information will be coming from state and local public health departments, she stressed, adding that the CDC would continue to update case counts on Monday, Wednesday, and Friday of each week. Procedures are being developed for the management of patients presumed to have COVID-19, where local health departments see positive tests but the mandatory CDC confirmatory test hasn’t been completed.
While new cases emerge across Europe and Asia, China’s earlier COVID-19 explosion seems to be slowing. “It’s really good news that the case counts in China are decreasing,” both for the well-being of that country’s citizens, and as a sign of the disease’s potential global effects, said Dr. Messonnier. She added that epidemiologists and mathematical modelers are parsing case fatality rates as well.
She advised health care providers and public health officials to keep abreast of changes in CDC guidance by checking frequently at https://www.cdc.gov/coronavirus/2019-ncov/index.html.
In a telebriefing on the COVID-19 outbreak, Nancy Messonnier, MD, director of the National Center for Immunization and Respiratory Diseases at .

The definition has been revised “to meet the needs of this rapidly evolving situation,” she said. The new PUI definition includes travel to more geographic areas to reflect this past week’s marked uptick in coronavirus activity in Italy and Iran. In addition to these countries and China, recent travel to Japan or South Korea also constitutes an epidemiologic risk factor which, in conjunction with clinical features, warrant an individual being classified as a PUI. These five countries each now have widespread person-to-person transmission of the virus.
Dr. Messonnier left open the possibility that the PUI definition would continue to evolve if such transmission within communities becomes more common. Asked whether the small number of U.S. cases thus might be an artifact of low test volumes, she said, “We aggressively controlled our borders to slow the spread. This was an intentional U.S. strategy. The CDC has always had the capacity to test rapidly from the time the sequence was available. ...We have been testing aggressively.”
The original PUI definition, she explained, emphasized individuals with fever, cough, or trouble breathing who had traveled recently from areas with COVID-19 activity, in particular China’s Hubei province. “We have been most focused on symptomatic people who are closely linked to, or who had, travel history, but our criteria also allow for clinical discretion,” she said. “There is no substitute for an astute clinician on the front lines of patient care.”
The first COVID-19 case from person-to-person spread was reported on Feb. 27. “At this time, we don’t know how or where this person became infected,” said Dr. Messonnier, although investigations are still underway. She responded to a question about whether the CDC delayed allowing COVID-19 testing for the patient for several days, as was reported in some media accounts. “According to CDC records, the first call we got was Feb. 23,” when public health officials in California reported a severely ill person with no travel abroad and no known contacts with individuals that would trigger suspicions for coronavirus. The CDC recommended COVID-19 testing on that day, she said.
Dr. Messonnier declined to answer questions about a whistleblower report alleging improper training and inadequate protective measures for Department of Health & Human Services workers at the quarantine center at Travis Air Force Base, Calif.
Dr. Messonnier said that the CDC has been working closely with the Food and Drug Administration to address problems with the COVID-19 test kits that were unusable because of a large number of indeterminate results. The two agencies together have determined that of the three reactions that were initially deemed necessary for a definitive COVID-19 diagnosis, just two are sufficient, so new kits that omit the problematic chemical are being manufactured and distributed.
These new kits are rapidly being made available; the goal, said Dr. Messonnier, is to have to state and local public health departments equipped with test kits by about March 7.
As local tests become available, the most updated information will be coming from state and local public health departments, she stressed, adding that the CDC would continue to update case counts on Monday, Wednesday, and Friday of each week. Procedures are being developed for the management of patients presumed to have COVID-19, where local health departments see positive tests but the mandatory CDC confirmatory test hasn’t been completed.
While new cases emerge across Europe and Asia, China’s earlier COVID-19 explosion seems to be slowing. “It’s really good news that the case counts in China are decreasing,” both for the well-being of that country’s citizens, and as a sign of the disease’s potential global effects, said Dr. Messonnier. She added that epidemiologists and mathematical modelers are parsing case fatality rates as well.
She advised health care providers and public health officials to keep abreast of changes in CDC guidance by checking frequently at https://www.cdc.gov/coronavirus/2019-ncov/index.html.
In a telebriefing on the COVID-19 outbreak, Nancy Messonnier, MD, director of the National Center for Immunization and Respiratory Diseases at .

The definition has been revised “to meet the needs of this rapidly evolving situation,” she said. The new PUI definition includes travel to more geographic areas to reflect this past week’s marked uptick in coronavirus activity in Italy and Iran. In addition to these countries and China, recent travel to Japan or South Korea also constitutes an epidemiologic risk factor which, in conjunction with clinical features, warrant an individual being classified as a PUI. These five countries each now have widespread person-to-person transmission of the virus.
Dr. Messonnier left open the possibility that the PUI definition would continue to evolve if such transmission within communities becomes more common. Asked whether the small number of U.S. cases thus might be an artifact of low test volumes, she said, “We aggressively controlled our borders to slow the spread. This was an intentional U.S. strategy. The CDC has always had the capacity to test rapidly from the time the sequence was available. ...We have been testing aggressively.”
The original PUI definition, she explained, emphasized individuals with fever, cough, or trouble breathing who had traveled recently from areas with COVID-19 activity, in particular China’s Hubei province. “We have been most focused on symptomatic people who are closely linked to, or who had, travel history, but our criteria also allow for clinical discretion,” she said. “There is no substitute for an astute clinician on the front lines of patient care.”
The first COVID-19 case from person-to-person spread was reported on Feb. 27. “At this time, we don’t know how or where this person became infected,” said Dr. Messonnier, although investigations are still underway. She responded to a question about whether the CDC delayed allowing COVID-19 testing for the patient for several days, as was reported in some media accounts. “According to CDC records, the first call we got was Feb. 23,” when public health officials in California reported a severely ill person with no travel abroad and no known contacts with individuals that would trigger suspicions for coronavirus. The CDC recommended COVID-19 testing on that day, she said.
Dr. Messonnier declined to answer questions about a whistleblower report alleging improper training and inadequate protective measures for Department of Health & Human Services workers at the quarantine center at Travis Air Force Base, Calif.
Dr. Messonnier said that the CDC has been working closely with the Food and Drug Administration to address problems with the COVID-19 test kits that were unusable because of a large number of indeterminate results. The two agencies together have determined that of the three reactions that were initially deemed necessary for a definitive COVID-19 diagnosis, just two are sufficient, so new kits that omit the problematic chemical are being manufactured and distributed.
These new kits are rapidly being made available; the goal, said Dr. Messonnier, is to have to state and local public health departments equipped with test kits by about March 7.
As local tests become available, the most updated information will be coming from state and local public health departments, she stressed, adding that the CDC would continue to update case counts on Monday, Wednesday, and Friday of each week. Procedures are being developed for the management of patients presumed to have COVID-19, where local health departments see positive tests but the mandatory CDC confirmatory test hasn’t been completed.
While new cases emerge across Europe and Asia, China’s earlier COVID-19 explosion seems to be slowing. “It’s really good news that the case counts in China are decreasing,” both for the well-being of that country’s citizens, and as a sign of the disease’s potential global effects, said Dr. Messonnier. She added that epidemiologists and mathematical modelers are parsing case fatality rates as well.
She advised health care providers and public health officials to keep abreast of changes in CDC guidance by checking frequently at https://www.cdc.gov/coronavirus/2019-ncov/index.html.
REPORTING FROM A CDC BRIEFING
Children bearing the brunt of declining flu activity
National flu activity decreased for the second consecutive week, but pediatric mortality is heading in the opposite direction, according to the Centers for Disease Control and Prevention.
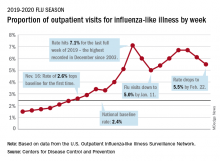
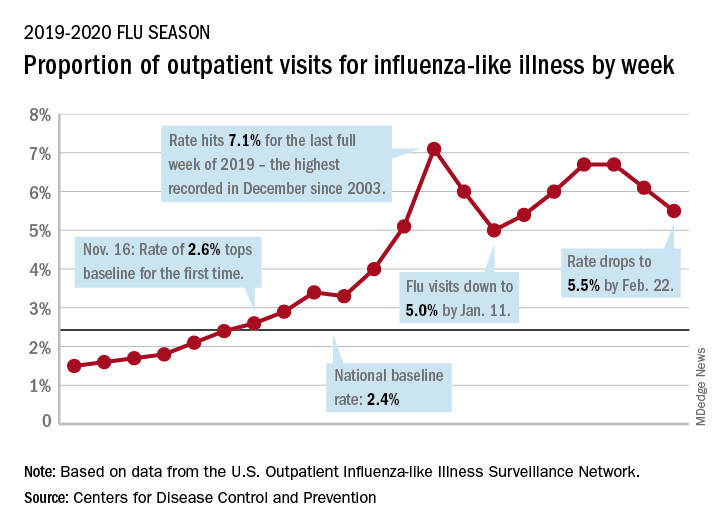
Influenza-like illness (ILI) represented 5.5% of all visits to outpatient health care providers during the week ending Feb. 22, compared with 6.1% the previous week, the CDC’s influenza division reported Feb. 28. The ILI visit rate had reached 6.6% in early February after dropping to 5.0% in mid-January, following a rise to a season-high 7.1% in the last week of December.
Another measure of ILI activity, the percentage of laboratory specimens testing positive, also declined for the second week in a row. The rate was 26.4% for the week ending Feb. 22, which is down from the season high of 30.3% reached 2 weeks before, the influenza division said.
ILI-related deaths among children, however, are not dropping. The total for 2019-2020 is now up to 125, and that “number is higher for the same time period than in every season since reporting began in 2004-05, except for the 2009 pandemic,” the CDC noted.
Hospitalization rates, which have been fairly typical in the general population, also are elevated for young adults and school-aged children, the agency said, and “rates among children 0-4 years old are now the highest CDC has on record at this point in the season, surpassing rates reported during the second wave of the 2009 H1N1 pandemic.”
National flu activity decreased for the second consecutive week, but pediatric mortality is heading in the opposite direction, according to the Centers for Disease Control and Prevention.
Influenza-like illness (ILI) represented 5.5% of all visits to outpatient health care providers during the week ending Feb. 22, compared with 6.1% the previous week, the CDC’s influenza division reported Feb. 28. The ILI visit rate had reached 6.6% in early February after dropping to 5.0% in mid-January, following a rise to a season-high 7.1% in the last week of December.
Another measure of ILI activity, the percentage of laboratory specimens testing positive, also declined for the second week in a row. The rate was 26.4% for the week ending Feb. 22, which is down from the season high of 30.3% reached 2 weeks before, the influenza division said.
ILI-related deaths among children, however, are not dropping. The total for 2019-2020 is now up to 125, and that “number is higher for the same time period than in every season since reporting began in 2004-05, except for the 2009 pandemic,” the CDC noted.
Hospitalization rates, which have been fairly typical in the general population, also are elevated for young adults and school-aged children, the agency said, and “rates among children 0-4 years old are now the highest CDC has on record at this point in the season, surpassing rates reported during the second wave of the 2009 H1N1 pandemic.”
National flu activity decreased for the second consecutive week, but pediatric mortality is heading in the opposite direction, according to the Centers for Disease Control and Prevention.
Influenza-like illness (ILI) represented 5.5% of all visits to outpatient health care providers during the week ending Feb. 22, compared with 6.1% the previous week, the CDC’s influenza division reported Feb. 28. The ILI visit rate had reached 6.6% in early February after dropping to 5.0% in mid-January, following a rise to a season-high 7.1% in the last week of December.
Another measure of ILI activity, the percentage of laboratory specimens testing positive, also declined for the second week in a row. The rate was 26.4% for the week ending Feb. 22, which is down from the season high of 30.3% reached 2 weeks before, the influenza division said.
ILI-related deaths among children, however, are not dropping. The total for 2019-2020 is now up to 125, and that “number is higher for the same time period than in every season since reporting began in 2004-05, except for the 2009 pandemic,” the CDC noted.
Hospitalization rates, which have been fairly typical in the general population, also are elevated for young adults and school-aged children, the agency said, and “rates among children 0-4 years old are now the highest CDC has on record at this point in the season, surpassing rates reported during the second wave of the 2009 H1N1 pandemic.”
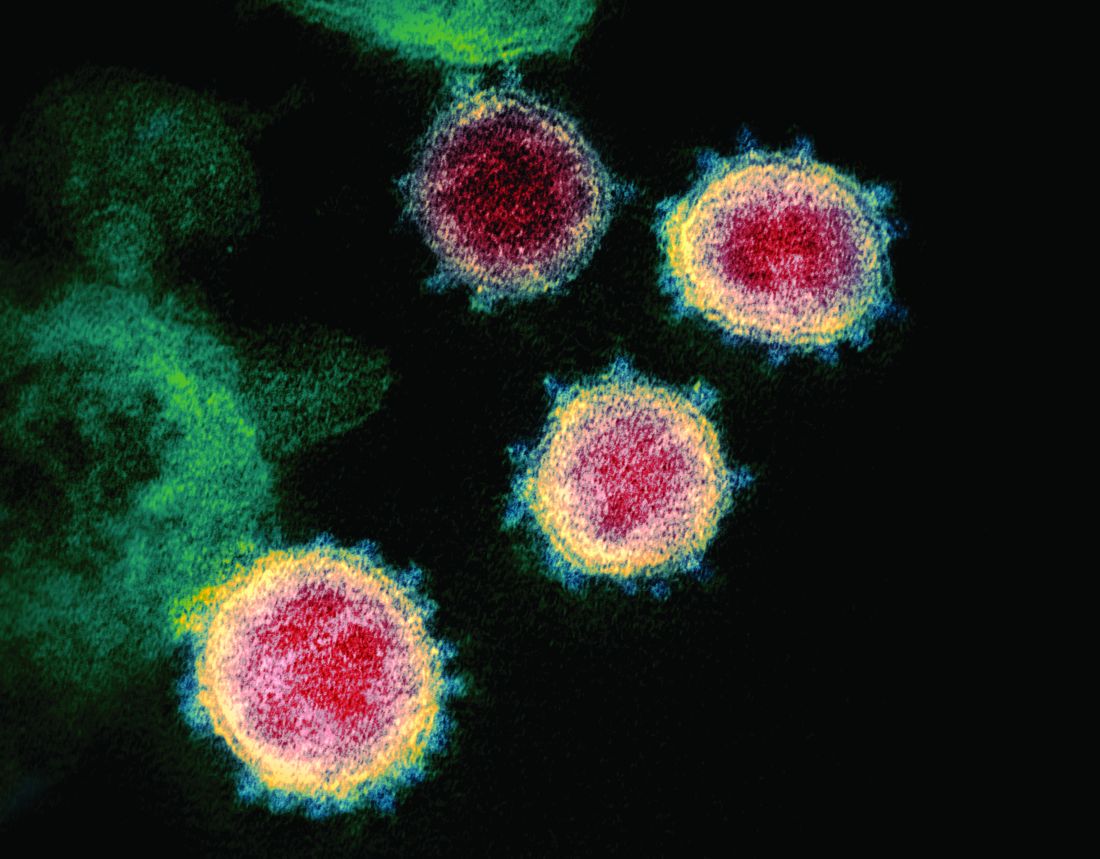
What hospitalists need to know about COVID-19
This article last updated 4/8/20. (Disclaimer: The information in this article may not be updated regularly. For more COVID-19 coverage, bookmark our COVID-19 updates page. The editors of The Hospitalist encourage clinicians to also review information on the CDC website and on the AHA website.)
An infectious disease outbreak that began in December 2019 in Wuhan (Hubei Province), China, was found to be caused by the seventh strain of coronavirus, initially called the novel (new) coronavirus. The virus was later labeled as severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). The disease caused by SARS-CoV-2 is named COVID-19. Until 2019, only six strains of human coronaviruses had previously been identified.
As of April 8, 2020, according to the U.S. Centers for Disease Control and Prevention, COVID-19 has been detected in at least 209 countries and has spread to every contintent except Antarctica. More than 1,469,245 people have become infected globally, and at least 86,278 have died. Based on the cases detected and tested in the United States through the U.S. public health surveillance systems, we have had 406,693 confirmed cases and 13,089 deaths.
On March 11, 2020, the World Health Organization formally declared the COVID-19 outbreak to be a pandemic.
As the number of cases increases in the United States, we hope to provide answers about some common questions regarding COVID-19. The information summarized in this article is obtained and modified from the CDC.
What are the clinical features of COVID-19?
Ranges from asymptomatic infection, a mild disease with nonspecific signs and symptoms of acute respiratory illness, to severe pneumonia with respiratory failure and septic shock.
Who is at risk for COVID-19?
Persons who have had prolonged, unprotected close contact with a patient with symptomatic, confirmed COVID-19, and those with recent travel to China, especially Hubei Province.
Who is at risk for severe disease from COVID-19?
Older adults and persons who have underlying chronic medical conditions, such as immunocompromising conditions.
How is COVID-19 spread?
Person-to-person, mainly through respiratory droplets. SARS-CoV-2 has been isolated from upper respiratory tract specimens and bronchoalveolar lavage fluid.
When is someone infectious?
Incubation period may range from 2 to 14 days. Detection of viral RNA does not necessarily mean that infectious virus is present, as it may be detectable in the upper or lower respiratory tract for weeks after illness onset.
Can someone who has been quarantined for COVID-19 spread the illness to others?
For COVID-19, the period of quarantine is 14 days from the last date of exposure, because 14 days is the longest incubation period seen for similar coronaviruses. Someone who has been released from COVID-19 quarantine is not considered a risk for spreading the virus to others because they have not developed illness during the incubation period.
Can a person test negative and later test positive for COVID-19?
Yes. In the early stages of infection, it is possible the virus will not be detected.
Do patients with confirmed or suspected COVID-19 need to be admitted to the hospital?
Not all patients with COVID-19 require hospital admission. Patients whose clinical presentation warrants inpatient clinical management for supportive medical care should be admitted to the hospital under appropriate isolation precautions. The decision to monitor these patients in the inpatient or outpatient setting should be made on a case-by-case basis.
What should you do if you suspect a patient for COVID-19?
Immediately notify both infection control personnel at your health care facility and your local or state health department. State health departments that have identified a person under investigation (PUI) should immediately contact CDC’s Emergency Operations Center (EOC) at 770-488-7100 and complete a COVID-19 PUI case investigation form.
CDC’s EOC will assist local/state health departments to collect, store, and ship specimens appropriately to CDC, including during after-hours or on weekends/holidays.
What type of isolation is needed for COVID-19?
Airborne Infection Isolation Room (AIIR) using Standard, Contact, and Airborne Precautions with eye protection.
How should health care personnel protect themselves when evaluating a patient who may have COVID-19?
Standard Precautions, Contact Precautions, Airborne Precautions, and use eye protection (e.g., goggles or a face shield).
What face mask do health care workers wear for respiratory protection?
A fit-tested NIOSH-certified disposable N95 filtering facepiece respirator should be worn before entry into the patient room or care area. Disposable respirators should be removed and discarded after exiting the patient’s room or care area and closing the door. Perform hand hygiene after discarding the respirator.
If reusable respirators (e.g., powered air purifying respirator/PAPR) are used, they must be cleaned and disinfected according to manufacturer’s reprocessing instructions prior to re-use.
What should you tell the patient if COVID-19 is suspected or confirmed?
Patients with suspected or confirmed COVID-19 should be asked to wear a surgical mask as soon as they are identified, to prevent spread to others.
Should any diagnostic or therapeutic interventions be withheld because of concerns about the transmission of COVID-19?
No.
How do you test a patient for SARS-CoV-2, the virus that causes COVID-19?
At this time, diagnostic testing for COVID-19 can be conducted only at CDC.
The CDC recommends collecting and testing multiple clinical specimens from different sites, including two specimen types – lower respiratory and upper respiratory (nasopharyngeal and oropharyngeal aspirates or washes, nasopharyngeal and oropharyngeal swabs, bronchioalveolar lavage, tracheal aspirates, sputum, and serum) using a real-time reverse transcription PCR (rRT-PCR) assay for SARS-CoV-2. Specimens should be collected as soon as possible once a PUI is identified regardless of the time of symptom onset. Turnaround time for the PCR assay testing is about 24-48 hours.
Testing for other respiratory pathogens should not delay specimen shipping to CDC. If a PUI tests positive for another respiratory pathogen, after clinical evaluation and consultation with public health authorities, they may no longer be considered a PUI.
Will existing respiratory virus panels detect SARS-CoV-2, the virus that causes COVID-19?
No.
How is COVID-19 treated?
Symptomatic management. Corticosteroids are not routinely recommended for viral pneumonia or acute respiratory distress syndrome and should be avoided unless they are indicated for another reason (e.g., COPD exacerbation, refractory septic shock following Surviving Sepsis Campaign Guidelines). There are currently no antiviral drugs licensed by the U.S. Food and Drug Administration to treat COVID-19.
What is considered ‘close contact’ for health care exposures?
Being within approximately 6 feet (2 meters), of a person with COVID-19 for a prolonged period of time (such as caring for or visiting the patient, or sitting within 6 feet of the patient in a health care waiting area or room); or having unprotected direct contact with infectious secretions or excretions of the patient (e.g., being coughed on, touching used tissues with a bare hand). However, until more is known about transmission risks, it would be reasonable to consider anything longer than a brief (e.g., less than 1-2 minutes) exposure as prolonged.
What happens if the health care personnel (HCP) are exposed to confirmed COVID-19 patients? What’s the protocol for HCP exposed to persons under investigation (PUI) if test results are delayed beyond 48-72 hours?
Management is similar in both these scenarios. CDC categorized exposures as high, medium, low, and no identifiable risk. High- and medium-risk exposures are managed similarly with active monitoring for COVID-19 until 14 days after last potential exposure and exclude from work for 14 days after last exposure. Active monitoring means that the state or local public health authority assumes responsibility for establishing regular communication with potentially exposed people to assess for the presence of fever or respiratory symptoms (e.g., cough, shortness of breath, sore throat). For HCP with high- or medium-risk exposures, CDC recommends this communication occurs at least once each day. For full details, please see www.cdc.gov/coronavirus/2019-ncov/hcp/guidance-risk-assesment-hcp.html.
Should postexposure prophylaxis be used for people who may have been exposed to COVID-19?
None available.
COVID-19 test results are negative in a symptomatic patient you suspected of COVID-19? What does it mean?
A negative test result for a sample collected while a person has symptoms likely means that the COVID-19 virus is not causing their current illness.
What if your hospital does not have an Airborne Infection Isolation Room (AIIR) for COVID-19 patients?
Transfer the patient to a facility that has an available AIIR. If a transfer is impractical or not medically appropriate, the patient should be cared for in a single-person room and the door should be kept closed. The room should ideally not have an exhaust that is recirculated within the building without high-efficiency particulate air (HEPA) filtration. Health care personnel should still use gloves, gown, respiratory and eye protection and follow all other recommended infection prevention and control practices when caring for these patients.
What if your hospital does not have enough Airborne Infection Isolation Rooms (AIIR) for COVID-19 patients?
Prioritize patients for AIIR who are symptomatic with severe illness (e.g., those requiring ventilator support).
When can patients with confirmed COVID-19 be discharged from the hospital?
Patients can be discharged from the health care facility whenever clinically indicated. Isolation should be maintained at home if the patient returns home before the time period recommended for discontinuation of hospital transmission-based precautions.
Considerations to discontinue transmission-based precautions include all of the following:
- Resolution of fever, without the use of antipyretic medication.
- Improvement in illness signs and symptoms.
- Negative rRT-PCR results from at least two consecutive sets of paired nasopharyngeal and throat swabs specimens collected at least 24 hours apart (total of four negative specimens – two nasopharyngeal and two throat) from a patient with COVID-19 are needed before discontinuing transmission-based precautions.
Should people be concerned about pets or other animals and COVID-19?
To date, CDC has not received any reports of pets or other animals becoming sick with COVID-19.
Should patients avoid contact with pets or other animals if they are sick with COVID-19?
Patients should restrict contact with pets and other animals while they are sick with COVID-19, just like they would around other people.
Does CDC recommend the use of face masks in the community to prevent COVID-19?
CDC does not recommend that people who are well wear a face mask to protect themselves from respiratory illnesses, including COVID-19. A face mask should be used by people who have COVID-19 and are showing symptoms to protect others from the risk of getting infected.
Should medical waste or general waste from health care facilities treating PUIs and patients with confirmed COVID-19 be handled any differently or need any additional disinfection?
No. CDC’s guidance states that management of laundry, food service utensils, and medical waste should be performed in accordance with routine procedures.
Can people who recover from COVID-19 be infected again?
Unknown. The immune response to COVID-19 is not yet understood.
What is the mortality rate of COVID-19, and how does it compare to the mortality rate of influenza (flu)?
The average 10-year mortality rate for flu, using CDC data, is found to be around 0.1%. Even though this percentage appears to be small, influenza is estimated to be responsible for 30,000 to 40,000 deaths annually in the U.S.
According to statistics released by the Chinese Center for Disease Control and Prevention on Feb. 17, the mortality rate of COVID-19 is estimated to be around 2.3%. This calculation was based on cases reported through Feb. 11, and calcuated by dividing the number of coronavirus-related deaths at the time (1,023) by the number of confirmed cases (44,672) of COVID-19 infection. However, this report has its limitations, since Chinese officials have a vague way of defining who has COVID-19 infection.
The World Health Organization (WHO) currently estimates the mortality rate for COVID-19 to be between 2% and 4%.
Dr. Sitammagari is a co-medical director for quality and assistant professor of internal medicine at Atrium Health, Charlotte, N.C. He is also a physician advisor. He currently serves as treasurer for the NC-Triangle Chapter of the Society of Hospital Medicine and as an editorial board member of The Hospitalist.
Dr. Skandhan is a hospitalist and member of the Core Faculty for the Internal Medicine Residency Program at Southeast Health (SEH), Dothan Ala., and an assistant professor at the Alabama College of Osteopathic Medicine. He serves as the medical director/physician liaison for the Clinical Documentation Program at SEH and also as the director for physician integration for Southeast Health Statera Network, an Accountable Care Organization. Dr. Skandhan was a cofounder of the Wiregrass chapter of SHM and currently serves on the Advisory board. He is also a member of the editorial board of The Hospitalist.
Dr. Dahlin is a second-year internal medicine resident at Southeast Health, Dothan, Ala. She serves as her class representative and is the cochair/resident liaison for the research committee at SEH. Dr. Dahlin also serves as a resident liaison for the Wiregrass chapter of SHM.
This article last updated 4/8/20. (Disclaimer: The information in this article may not be updated regularly. For more COVID-19 coverage, bookmark our COVID-19 updates page. The editors of The Hospitalist encourage clinicians to also review information on the CDC website and on the AHA website.)
An infectious disease outbreak that began in December 2019 in Wuhan (Hubei Province), China, was found to be caused by the seventh strain of coronavirus, initially called the novel (new) coronavirus. The virus was later labeled as severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). The disease caused by SARS-CoV-2 is named COVID-19. Until 2019, only six strains of human coronaviruses had previously been identified.
As of April 8, 2020, according to the U.S. Centers for Disease Control and Prevention, COVID-19 has been detected in at least 209 countries and has spread to every contintent except Antarctica. More than 1,469,245 people have become infected globally, and at least 86,278 have died. Based on the cases detected and tested in the United States through the U.S. public health surveillance systems, we have had 406,693 confirmed cases and 13,089 deaths.
On March 11, 2020, the World Health Organization formally declared the COVID-19 outbreak to be a pandemic.
As the number of cases increases in the United States, we hope to provide answers about some common questions regarding COVID-19. The information summarized in this article is obtained and modified from the CDC.
What are the clinical features of COVID-19?
Ranges from asymptomatic infection, a mild disease with nonspecific signs and symptoms of acute respiratory illness, to severe pneumonia with respiratory failure and septic shock.
Who is at risk for COVID-19?
Persons who have had prolonged, unprotected close contact with a patient with symptomatic, confirmed COVID-19, and those with recent travel to China, especially Hubei Province.
Who is at risk for severe disease from COVID-19?
Older adults and persons who have underlying chronic medical conditions, such as immunocompromising conditions.
How is COVID-19 spread?
Person-to-person, mainly through respiratory droplets. SARS-CoV-2 has been isolated from upper respiratory tract specimens and bronchoalveolar lavage fluid.
When is someone infectious?
Incubation period may range from 2 to 14 days. Detection of viral RNA does not necessarily mean that infectious virus is present, as it may be detectable in the upper or lower respiratory tract for weeks after illness onset.
Can someone who has been quarantined for COVID-19 spread the illness to others?
For COVID-19, the period of quarantine is 14 days from the last date of exposure, because 14 days is the longest incubation period seen for similar coronaviruses. Someone who has been released from COVID-19 quarantine is not considered a risk for spreading the virus to others because they have not developed illness during the incubation period.
Can a person test negative and later test positive for COVID-19?
Yes. In the early stages of infection, it is possible the virus will not be detected.
Do patients with confirmed or suspected COVID-19 need to be admitted to the hospital?
Not all patients with COVID-19 require hospital admission. Patients whose clinical presentation warrants inpatient clinical management for supportive medical care should be admitted to the hospital under appropriate isolation precautions. The decision to monitor these patients in the inpatient or outpatient setting should be made on a case-by-case basis.
What should you do if you suspect a patient for COVID-19?
Immediately notify both infection control personnel at your health care facility and your local or state health department. State health departments that have identified a person under investigation (PUI) should immediately contact CDC’s Emergency Operations Center (EOC) at 770-488-7100 and complete a COVID-19 PUI case investigation form.
CDC’s EOC will assist local/state health departments to collect, store, and ship specimens appropriately to CDC, including during after-hours or on weekends/holidays.
What type of isolation is needed for COVID-19?
Airborne Infection Isolation Room (AIIR) using Standard, Contact, and Airborne Precautions with eye protection.
How should health care personnel protect themselves when evaluating a patient who may have COVID-19?
Standard Precautions, Contact Precautions, Airborne Precautions, and use eye protection (e.g., goggles or a face shield).
What face mask do health care workers wear for respiratory protection?
A fit-tested NIOSH-certified disposable N95 filtering facepiece respirator should be worn before entry into the patient room or care area. Disposable respirators should be removed and discarded after exiting the patient’s room or care area and closing the door. Perform hand hygiene after discarding the respirator.
If reusable respirators (e.g., powered air purifying respirator/PAPR) are used, they must be cleaned and disinfected according to manufacturer’s reprocessing instructions prior to re-use.
What should you tell the patient if COVID-19 is suspected or confirmed?
Patients with suspected or confirmed COVID-19 should be asked to wear a surgical mask as soon as they are identified, to prevent spread to others.
Should any diagnostic or therapeutic interventions be withheld because of concerns about the transmission of COVID-19?
No.
How do you test a patient for SARS-CoV-2, the virus that causes COVID-19?
At this time, diagnostic testing for COVID-19 can be conducted only at CDC.
The CDC recommends collecting and testing multiple clinical specimens from different sites, including two specimen types – lower respiratory and upper respiratory (nasopharyngeal and oropharyngeal aspirates or washes, nasopharyngeal and oropharyngeal swabs, bronchioalveolar lavage, tracheal aspirates, sputum, and serum) using a real-time reverse transcription PCR (rRT-PCR) assay for SARS-CoV-2. Specimens should be collected as soon as possible once a PUI is identified regardless of the time of symptom onset. Turnaround time for the PCR assay testing is about 24-48 hours.
Testing for other respiratory pathogens should not delay specimen shipping to CDC. If a PUI tests positive for another respiratory pathogen, after clinical evaluation and consultation with public health authorities, they may no longer be considered a PUI.
Will existing respiratory virus panels detect SARS-CoV-2, the virus that causes COVID-19?
No.
How is COVID-19 treated?
Symptomatic management. Corticosteroids are not routinely recommended for viral pneumonia or acute respiratory distress syndrome and should be avoided unless they are indicated for another reason (e.g., COPD exacerbation, refractory septic shock following Surviving Sepsis Campaign Guidelines). There are currently no antiviral drugs licensed by the U.S. Food and Drug Administration to treat COVID-19.
What is considered ‘close contact’ for health care exposures?
Being within approximately 6 feet (2 meters), of a person with COVID-19 for a prolonged period of time (such as caring for or visiting the patient, or sitting within 6 feet of the patient in a health care waiting area or room); or having unprotected direct contact with infectious secretions or excretions of the patient (e.g., being coughed on, touching used tissues with a bare hand). However, until more is known about transmission risks, it would be reasonable to consider anything longer than a brief (e.g., less than 1-2 minutes) exposure as prolonged.
What happens if the health care personnel (HCP) are exposed to confirmed COVID-19 patients? What’s the protocol for HCP exposed to persons under investigation (PUI) if test results are delayed beyond 48-72 hours?
Management is similar in both these scenarios. CDC categorized exposures as high, medium, low, and no identifiable risk. High- and medium-risk exposures are managed similarly with active monitoring for COVID-19 until 14 days after last potential exposure and exclude from work for 14 days after last exposure. Active monitoring means that the state or local public health authority assumes responsibility for establishing regular communication with potentially exposed people to assess for the presence of fever or respiratory symptoms (e.g., cough, shortness of breath, sore throat). For HCP with high- or medium-risk exposures, CDC recommends this communication occurs at least once each day. For full details, please see www.cdc.gov/coronavirus/2019-ncov/hcp/guidance-risk-assesment-hcp.html.
Should postexposure prophylaxis be used for people who may have been exposed to COVID-19?
None available.
COVID-19 test results are negative in a symptomatic patient you suspected of COVID-19? What does it mean?
A negative test result for a sample collected while a person has symptoms likely means that the COVID-19 virus is not causing their current illness.
What if your hospital does not have an Airborne Infection Isolation Room (AIIR) for COVID-19 patients?
Transfer the patient to a facility that has an available AIIR. If a transfer is impractical or not medically appropriate, the patient should be cared for in a single-person room and the door should be kept closed. The room should ideally not have an exhaust that is recirculated within the building without high-efficiency particulate air (HEPA) filtration. Health care personnel should still use gloves, gown, respiratory and eye protection and follow all other recommended infection prevention and control practices when caring for these patients.
What if your hospital does not have enough Airborne Infection Isolation Rooms (AIIR) for COVID-19 patients?
Prioritize patients for AIIR who are symptomatic with severe illness (e.g., those requiring ventilator support).
When can patients with confirmed COVID-19 be discharged from the hospital?
Patients can be discharged from the health care facility whenever clinically indicated. Isolation should be maintained at home if the patient returns home before the time period recommended for discontinuation of hospital transmission-based precautions.
Considerations to discontinue transmission-based precautions include all of the following:
- Resolution of fever, without the use of antipyretic medication.
- Improvement in illness signs and symptoms.
- Negative rRT-PCR results from at least two consecutive sets of paired nasopharyngeal and throat swabs specimens collected at least 24 hours apart (total of four negative specimens – two nasopharyngeal and two throat) from a patient with COVID-19 are needed before discontinuing transmission-based precautions.
Should people be concerned about pets or other animals and COVID-19?
To date, CDC has not received any reports of pets or other animals becoming sick with COVID-19.
Should patients avoid contact with pets or other animals if they are sick with COVID-19?
Patients should restrict contact with pets and other animals while they are sick with COVID-19, just like they would around other people.
Does CDC recommend the use of face masks in the community to prevent COVID-19?
CDC does not recommend that people who are well wear a face mask to protect themselves from respiratory illnesses, including COVID-19. A face mask should be used by people who have COVID-19 and are showing symptoms to protect others from the risk of getting infected.
Should medical waste or general waste from health care facilities treating PUIs and patients with confirmed COVID-19 be handled any differently or need any additional disinfection?
No. CDC’s guidance states that management of laundry, food service utensils, and medical waste should be performed in accordance with routine procedures.
Can people who recover from COVID-19 be infected again?
Unknown. The immune response to COVID-19 is not yet understood.
What is the mortality rate of COVID-19, and how does it compare to the mortality rate of influenza (flu)?
The average 10-year mortality rate for flu, using CDC data, is found to be around 0.1%. Even though this percentage appears to be small, influenza is estimated to be responsible for 30,000 to 40,000 deaths annually in the U.S.
According to statistics released by the Chinese Center for Disease Control and Prevention on Feb. 17, the mortality rate of COVID-19 is estimated to be around 2.3%. This calculation was based on cases reported through Feb. 11, and calcuated by dividing the number of coronavirus-related deaths at the time (1,023) by the number of confirmed cases (44,672) of COVID-19 infection. However, this report has its limitations, since Chinese officials have a vague way of defining who has COVID-19 infection.
The World Health Organization (WHO) currently estimates the mortality rate for COVID-19 to be between 2% and 4%.
Dr. Sitammagari is a co-medical director for quality and assistant professor of internal medicine at Atrium Health, Charlotte, N.C. He is also a physician advisor. He currently serves as treasurer for the NC-Triangle Chapter of the Society of Hospital Medicine and as an editorial board member of The Hospitalist.
Dr. Skandhan is a hospitalist and member of the Core Faculty for the Internal Medicine Residency Program at Southeast Health (SEH), Dothan Ala., and an assistant professor at the Alabama College of Osteopathic Medicine. He serves as the medical director/physician liaison for the Clinical Documentation Program at SEH and also as the director for physician integration for Southeast Health Statera Network, an Accountable Care Organization. Dr. Skandhan was a cofounder of the Wiregrass chapter of SHM and currently serves on the Advisory board. He is also a member of the editorial board of The Hospitalist.
Dr. Dahlin is a second-year internal medicine resident at Southeast Health, Dothan, Ala. She serves as her class representative and is the cochair/resident liaison for the research committee at SEH. Dr. Dahlin also serves as a resident liaison for the Wiregrass chapter of SHM.
This article last updated 4/8/20. (Disclaimer: The information in this article may not be updated regularly. For more COVID-19 coverage, bookmark our COVID-19 updates page. The editors of The Hospitalist encourage clinicians to also review information on the CDC website and on the AHA website.)
An infectious disease outbreak that began in December 2019 in Wuhan (Hubei Province), China, was found to be caused by the seventh strain of coronavirus, initially called the novel (new) coronavirus. The virus was later labeled as severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). The disease caused by SARS-CoV-2 is named COVID-19. Until 2019, only six strains of human coronaviruses had previously been identified.
As of April 8, 2020, according to the U.S. Centers for Disease Control and Prevention, COVID-19 has been detected in at least 209 countries and has spread to every contintent except Antarctica. More than 1,469,245 people have become infected globally, and at least 86,278 have died. Based on the cases detected and tested in the United States through the U.S. public health surveillance systems, we have had 406,693 confirmed cases and 13,089 deaths.
On March 11, 2020, the World Health Organization formally declared the COVID-19 outbreak to be a pandemic.
As the number of cases increases in the United States, we hope to provide answers about some common questions regarding COVID-19. The information summarized in this article is obtained and modified from the CDC.
What are the clinical features of COVID-19?
Ranges from asymptomatic infection, a mild disease with nonspecific signs and symptoms of acute respiratory illness, to severe pneumonia with respiratory failure and septic shock.
Who is at risk for COVID-19?
Persons who have had prolonged, unprotected close contact with a patient with symptomatic, confirmed COVID-19, and those with recent travel to China, especially Hubei Province.
Who is at risk for severe disease from COVID-19?
Older adults and persons who have underlying chronic medical conditions, such as immunocompromising conditions.
How is COVID-19 spread?
Person-to-person, mainly through respiratory droplets. SARS-CoV-2 has been isolated from upper respiratory tract specimens and bronchoalveolar lavage fluid.
When is someone infectious?
Incubation period may range from 2 to 14 days. Detection of viral RNA does not necessarily mean that infectious virus is present, as it may be detectable in the upper or lower respiratory tract for weeks after illness onset.
Can someone who has been quarantined for COVID-19 spread the illness to others?
For COVID-19, the period of quarantine is 14 days from the last date of exposure, because 14 days is the longest incubation period seen for similar coronaviruses. Someone who has been released from COVID-19 quarantine is not considered a risk for spreading the virus to others because they have not developed illness during the incubation period.
Can a person test negative and later test positive for COVID-19?
Yes. In the early stages of infection, it is possible the virus will not be detected.
Do patients with confirmed or suspected COVID-19 need to be admitted to the hospital?
Not all patients with COVID-19 require hospital admission. Patients whose clinical presentation warrants inpatient clinical management for supportive medical care should be admitted to the hospital under appropriate isolation precautions. The decision to monitor these patients in the inpatient or outpatient setting should be made on a case-by-case basis.
What should you do if you suspect a patient for COVID-19?
Immediately notify both infection control personnel at your health care facility and your local or state health department. State health departments that have identified a person under investigation (PUI) should immediately contact CDC’s Emergency Operations Center (EOC) at 770-488-7100 and complete a COVID-19 PUI case investigation form.
CDC’s EOC will assist local/state health departments to collect, store, and ship specimens appropriately to CDC, including during after-hours or on weekends/holidays.
What type of isolation is needed for COVID-19?
Airborne Infection Isolation Room (AIIR) using Standard, Contact, and Airborne Precautions with eye protection.
How should health care personnel protect themselves when evaluating a patient who may have COVID-19?
Standard Precautions, Contact Precautions, Airborne Precautions, and use eye protection (e.g., goggles or a face shield).
What face mask do health care workers wear for respiratory protection?
A fit-tested NIOSH-certified disposable N95 filtering facepiece respirator should be worn before entry into the patient room or care area. Disposable respirators should be removed and discarded after exiting the patient’s room or care area and closing the door. Perform hand hygiene after discarding the respirator.
If reusable respirators (e.g., powered air purifying respirator/PAPR) are used, they must be cleaned and disinfected according to manufacturer’s reprocessing instructions prior to re-use.
What should you tell the patient if COVID-19 is suspected or confirmed?
Patients with suspected or confirmed COVID-19 should be asked to wear a surgical mask as soon as they are identified, to prevent spread to others.
Should any diagnostic or therapeutic interventions be withheld because of concerns about the transmission of COVID-19?
No.
How do you test a patient for SARS-CoV-2, the virus that causes COVID-19?
At this time, diagnostic testing for COVID-19 can be conducted only at CDC.
The CDC recommends collecting and testing multiple clinical specimens from different sites, including two specimen types – lower respiratory and upper respiratory (nasopharyngeal and oropharyngeal aspirates or washes, nasopharyngeal and oropharyngeal swabs, bronchioalveolar lavage, tracheal aspirates, sputum, and serum) using a real-time reverse transcription PCR (rRT-PCR) assay for SARS-CoV-2. Specimens should be collected as soon as possible once a PUI is identified regardless of the time of symptom onset. Turnaround time for the PCR assay testing is about 24-48 hours.
Testing for other respiratory pathogens should not delay specimen shipping to CDC. If a PUI tests positive for another respiratory pathogen, after clinical evaluation and consultation with public health authorities, they may no longer be considered a PUI.
Will existing respiratory virus panels detect SARS-CoV-2, the virus that causes COVID-19?
No.
How is COVID-19 treated?
Symptomatic management. Corticosteroids are not routinely recommended for viral pneumonia or acute respiratory distress syndrome and should be avoided unless they are indicated for another reason (e.g., COPD exacerbation, refractory septic shock following Surviving Sepsis Campaign Guidelines). There are currently no antiviral drugs licensed by the U.S. Food and Drug Administration to treat COVID-19.
What is considered ‘close contact’ for health care exposures?
Being within approximately 6 feet (2 meters), of a person with COVID-19 for a prolonged period of time (such as caring for or visiting the patient, or sitting within 6 feet of the patient in a health care waiting area or room); or having unprotected direct contact with infectious secretions or excretions of the patient (e.g., being coughed on, touching used tissues with a bare hand). However, until more is known about transmission risks, it would be reasonable to consider anything longer than a brief (e.g., less than 1-2 minutes) exposure as prolonged.
What happens if the health care personnel (HCP) are exposed to confirmed COVID-19 patients? What’s the protocol for HCP exposed to persons under investigation (PUI) if test results are delayed beyond 48-72 hours?
Management is similar in both these scenarios. CDC categorized exposures as high, medium, low, and no identifiable risk. High- and medium-risk exposures are managed similarly with active monitoring for COVID-19 until 14 days after last potential exposure and exclude from work for 14 days after last exposure. Active monitoring means that the state or local public health authority assumes responsibility for establishing regular communication with potentially exposed people to assess for the presence of fever or respiratory symptoms (e.g., cough, shortness of breath, sore throat). For HCP with high- or medium-risk exposures, CDC recommends this communication occurs at least once each day. For full details, please see www.cdc.gov/coronavirus/2019-ncov/hcp/guidance-risk-assesment-hcp.html.
Should postexposure prophylaxis be used for people who may have been exposed to COVID-19?
None available.
COVID-19 test results are negative in a symptomatic patient you suspected of COVID-19? What does it mean?
A negative test result for a sample collected while a person has symptoms likely means that the COVID-19 virus is not causing their current illness.
What if your hospital does not have an Airborne Infection Isolation Room (AIIR) for COVID-19 patients?
Transfer the patient to a facility that has an available AIIR. If a transfer is impractical or not medically appropriate, the patient should be cared for in a single-person room and the door should be kept closed. The room should ideally not have an exhaust that is recirculated within the building without high-efficiency particulate air (HEPA) filtration. Health care personnel should still use gloves, gown, respiratory and eye protection and follow all other recommended infection prevention and control practices when caring for these patients.
What if your hospital does not have enough Airborne Infection Isolation Rooms (AIIR) for COVID-19 patients?
Prioritize patients for AIIR who are symptomatic with severe illness (e.g., those requiring ventilator support).
When can patients with confirmed COVID-19 be discharged from the hospital?
Patients can be discharged from the health care facility whenever clinically indicated. Isolation should be maintained at home if the patient returns home before the time period recommended for discontinuation of hospital transmission-based precautions.
Considerations to discontinue transmission-based precautions include all of the following:
- Resolution of fever, without the use of antipyretic medication.
- Improvement in illness signs and symptoms.
- Negative rRT-PCR results from at least two consecutive sets of paired nasopharyngeal and throat swabs specimens collected at least 24 hours apart (total of four negative specimens – two nasopharyngeal and two throat) from a patient with COVID-19 are needed before discontinuing transmission-based precautions.
Should people be concerned about pets or other animals and COVID-19?
To date, CDC has not received any reports of pets or other animals becoming sick with COVID-19.
Should patients avoid contact with pets or other animals if they are sick with COVID-19?
Patients should restrict contact with pets and other animals while they are sick with COVID-19, just like they would around other people.
Does CDC recommend the use of face masks in the community to prevent COVID-19?
CDC does not recommend that people who are well wear a face mask to protect themselves from respiratory illnesses, including COVID-19. A face mask should be used by people who have COVID-19 and are showing symptoms to protect others from the risk of getting infected.
Should medical waste or general waste from health care facilities treating PUIs and patients with confirmed COVID-19 be handled any differently or need any additional disinfection?
No. CDC’s guidance states that management of laundry, food service utensils, and medical waste should be performed in accordance with routine procedures.
Can people who recover from COVID-19 be infected again?
Unknown. The immune response to COVID-19 is not yet understood.
What is the mortality rate of COVID-19, and how does it compare to the mortality rate of influenza (flu)?
The average 10-year mortality rate for flu, using CDC data, is found to be around 0.1%. Even though this percentage appears to be small, influenza is estimated to be responsible for 30,000 to 40,000 deaths annually in the U.S.
According to statistics released by the Chinese Center for Disease Control and Prevention on Feb. 17, the mortality rate of COVID-19 is estimated to be around 2.3%. This calculation was based on cases reported through Feb. 11, and calcuated by dividing the number of coronavirus-related deaths at the time (1,023) by the number of confirmed cases (44,672) of COVID-19 infection. However, this report has its limitations, since Chinese officials have a vague way of defining who has COVID-19 infection.
The World Health Organization (WHO) currently estimates the mortality rate for COVID-19 to be between 2% and 4%.
Dr. Sitammagari is a co-medical director for quality and assistant professor of internal medicine at Atrium Health, Charlotte, N.C. He is also a physician advisor. He currently serves as treasurer for the NC-Triangle Chapter of the Society of Hospital Medicine and as an editorial board member of The Hospitalist.
Dr. Skandhan is a hospitalist and member of the Core Faculty for the Internal Medicine Residency Program at Southeast Health (SEH), Dothan Ala., and an assistant professor at the Alabama College of Osteopathic Medicine. He serves as the medical director/physician liaison for the Clinical Documentation Program at SEH and also as the director for physician integration for Southeast Health Statera Network, an Accountable Care Organization. Dr. Skandhan was a cofounder of the Wiregrass chapter of SHM and currently serves on the Advisory board. He is also a member of the editorial board of The Hospitalist.
Dr. Dahlin is a second-year internal medicine resident at Southeast Health, Dothan, Ala. She serves as her class representative and is the cochair/resident liaison for the research committee at SEH. Dr. Dahlin also serves as a resident liaison for the Wiregrass chapter of SHM.
ACIP: Flu vaccines for older adults show similar safety profiles
The Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices (ACIP) recommends that age-appropriate vaccines be used when possible, said Kenneth E. Schmader, MD, professor of medicine at Duke University, Durham, N.C. However, no study to date had directly compared the safety of the trivalent high dose (HD-IIV3) and adjuvanted (aIIV3) vaccines or their impact on health-related quality of life. Dr. Schmader presented findings from a randomized trial at the February ACIP meeting.
To compare the safety of the vaccines, the researchers recruited community-dwelling volunteers aged 65 years and older who were cognitively intact, not immunosuppressed, and had no contraindications for influenza vaccination. A total of 378 individuals were randomized to aIIV3 and 379 to HD-IIV3. The average age was 72 years; 80 individuals in the aIIV3 group and 83 in the HDIIV3 group were 80 years and older. The primary outcome was moderate or severe injection site pain.
Overall, the proportion of participants with moderate or severe injection site pain was not significantly different after aIIV3 vs. HD-IIV3 (3.2% vs. 5.8%).
Nine participants in the aIIV3 group and three participants in the HD-IIV3 group experienced at least one serious adverse event, but no serious adverse events were deemed vaccine related, and the occurrence of serious adverse events was not significantly different between groups.
In addition, measures of short-term, postvaccination health-related quality of life were not significantly different between the groups. Changes in scores from day 1 prevaccination to day 3 postvaccination on the EuroQOL-5 dimensions-5 levels (EQ-5D-5L) were –0.05 for both groups.
The findings were limited in part by the lack of inclusion of older adults in nursing homes or similar settings, Dr. Schmader noted. However, the results suggest that “from the standpoint of safety, either vaccine is an acceptable option for the prevention of influenza in older adults.”
Studies comparing the immunogenicity of the vaccines are ongoing, and the data should be available within the next few months, he noted.
Dr. Schmader had no financial conflicts to disclose.
The Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices (ACIP) recommends that age-appropriate vaccines be used when possible, said Kenneth E. Schmader, MD, professor of medicine at Duke University, Durham, N.C. However, no study to date had directly compared the safety of the trivalent high dose (HD-IIV3) and adjuvanted (aIIV3) vaccines or their impact on health-related quality of life. Dr. Schmader presented findings from a randomized trial at the February ACIP meeting.
To compare the safety of the vaccines, the researchers recruited community-dwelling volunteers aged 65 years and older who were cognitively intact, not immunosuppressed, and had no contraindications for influenza vaccination. A total of 378 individuals were randomized to aIIV3 and 379 to HD-IIV3. The average age was 72 years; 80 individuals in the aIIV3 group and 83 in the HDIIV3 group were 80 years and older. The primary outcome was moderate or severe injection site pain.
Overall, the proportion of participants with moderate or severe injection site pain was not significantly different after aIIV3 vs. HD-IIV3 (3.2% vs. 5.8%).
Nine participants in the aIIV3 group and three participants in the HD-IIV3 group experienced at least one serious adverse event, but no serious adverse events were deemed vaccine related, and the occurrence of serious adverse events was not significantly different between groups.
In addition, measures of short-term, postvaccination health-related quality of life were not significantly different between the groups. Changes in scores from day 1 prevaccination to day 3 postvaccination on the EuroQOL-5 dimensions-5 levels (EQ-5D-5L) were –0.05 for both groups.
The findings were limited in part by the lack of inclusion of older adults in nursing homes or similar settings, Dr. Schmader noted. However, the results suggest that “from the standpoint of safety, either vaccine is an acceptable option for the prevention of influenza in older adults.”
Studies comparing the immunogenicity of the vaccines are ongoing, and the data should be available within the next few months, he noted.
Dr. Schmader had no financial conflicts to disclose.
The Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices (ACIP) recommends that age-appropriate vaccines be used when possible, said Kenneth E. Schmader, MD, professor of medicine at Duke University, Durham, N.C. However, no study to date had directly compared the safety of the trivalent high dose (HD-IIV3) and adjuvanted (aIIV3) vaccines or their impact on health-related quality of life. Dr. Schmader presented findings from a randomized trial at the February ACIP meeting.
To compare the safety of the vaccines, the researchers recruited community-dwelling volunteers aged 65 years and older who were cognitively intact, not immunosuppressed, and had no contraindications for influenza vaccination. A total of 378 individuals were randomized to aIIV3 and 379 to HD-IIV3. The average age was 72 years; 80 individuals in the aIIV3 group and 83 in the HDIIV3 group were 80 years and older. The primary outcome was moderate or severe injection site pain.
Overall, the proportion of participants with moderate or severe injection site pain was not significantly different after aIIV3 vs. HD-IIV3 (3.2% vs. 5.8%).
Nine participants in the aIIV3 group and three participants in the HD-IIV3 group experienced at least one serious adverse event, but no serious adverse events were deemed vaccine related, and the occurrence of serious adverse events was not significantly different between groups.
In addition, measures of short-term, postvaccination health-related quality of life were not significantly different between the groups. Changes in scores from day 1 prevaccination to day 3 postvaccination on the EuroQOL-5 dimensions-5 levels (EQ-5D-5L) were –0.05 for both groups.
The findings were limited in part by the lack of inclusion of older adults in nursing homes or similar settings, Dr. Schmader noted. However, the results suggest that “from the standpoint of safety, either vaccine is an acceptable option for the prevention of influenza in older adults.”
Studies comparing the immunogenicity of the vaccines are ongoing, and the data should be available within the next few months, he noted.
Dr. Schmader had no financial conflicts to disclose.
FROM AN ACIP MEETING
Pence named COVID-19 point person as CDC reports possible community spread
Vice President Mike Pence will be the White House point person quarterbacking the administration’s response to COVID-19, although President Donald Trump was quick to dismiss the notion that he is a so-called coronavirus “czar.”
President Trump introduced Vice President Pence in this role during a Feb. 26 press conference. The same night, officials at the Centers for Disease Control and Prevention announced the first case of possible community spread of the novel coronavirus in the United States.
“I am going to be putting our vice president, Mike Pence, in charge, and Mike will be working with the professionals, the doctors, and everybody else that is working” on this, President Trump said.
“Mike is going to be in charge and Mike will report back to me, but he’s got a certain talent for this,” President Trump continued, noting that while Vice President Pence was governor of Indiana, his was the first state to have a patient affected by the 2014 Middle East Respiratory Syndrome coronavirus (MERS-CoV) outbreak, so he has experience in a similar situation.
“I know full well the importance of presidential leadership, the importance of administration leadership, and the vital role of partnerships of state and local governments and health authorities in responding to the potential threat of dangerous infectious diseases,” Vice President Pence said.
He said that his role will be to continue to meet with the Coronavirus Task Force and bring to the president “the best options for action and to see to the safety and well being and health of the American people. I will also be continuing to reach out to governors [and] state and local officials.”
Vice President Pence said he will also be working with Congress to ensure that resources are available.
It was noted during the press conference that some members of Congress consider the $2.5 billion in emergency appropriations requested by the White House to be inadequate and that the legislative branch is working to provide more funding.
Vice President Pence’s new role does not change the command structure of the Coronavirus Task Force, which is currently led by Department of Health & Human Services Secretary Alex Azar.
Speaking at the press conference, Secretary Azar noted that he is still chairman of the task force. “Having the vice president gives me the biggest stick one can have in the government on this whole-of-government approach.”
He emphatically stated, “not in the least,” in response to a question about whether he felt he was being replaced. “When this was mentioned to me, I said I was delighted that I get to have the vice president helping in this way. Delighted.”
The announcement came as President Trump continued to downplay the threat of the coronavirus to U.S. citizens, going so far as to contradict CDC officials who have stated that it is a matter of when, not if, there will be community spread in the United States.
“I don’t think it’s inevitable,” President Trump said. “I think that there’s a chance that it could get worse. There’s a chance it could get fairly substantially worse, but nothing’s inevitable.”
Immediately after President Trump wrapped up his statement, however, the CDC formally announced the first case of possible community spread of the coronavirus. In a statement issued to the press, the agency announced the 15th confirmed case in the United States, a person in California “who reportedly did not have relevant travel history or exposure to another known patient” with the coronavirus.
“This case was detected through the U.S. public health system – picked up by astute clinicians,” CDC added, noting it will continue to provide updates on the evolving situation.
Vice President Mike Pence will be the White House point person quarterbacking the administration’s response to COVID-19, although President Donald Trump was quick to dismiss the notion that he is a so-called coronavirus “czar.”
President Trump introduced Vice President Pence in this role during a Feb. 26 press conference. The same night, officials at the Centers for Disease Control and Prevention announced the first case of possible community spread of the novel coronavirus in the United States.
“I am going to be putting our vice president, Mike Pence, in charge, and Mike will be working with the professionals, the doctors, and everybody else that is working” on this, President Trump said.
“Mike is going to be in charge and Mike will report back to me, but he’s got a certain talent for this,” President Trump continued, noting that while Vice President Pence was governor of Indiana, his was the first state to have a patient affected by the 2014 Middle East Respiratory Syndrome coronavirus (MERS-CoV) outbreak, so he has experience in a similar situation.
“I know full well the importance of presidential leadership, the importance of administration leadership, and the vital role of partnerships of state and local governments and health authorities in responding to the potential threat of dangerous infectious diseases,” Vice President Pence said.
He said that his role will be to continue to meet with the Coronavirus Task Force and bring to the president “the best options for action and to see to the safety and well being and health of the American people. I will also be continuing to reach out to governors [and] state and local officials.”
Vice President Pence said he will also be working with Congress to ensure that resources are available.
It was noted during the press conference that some members of Congress consider the $2.5 billion in emergency appropriations requested by the White House to be inadequate and that the legislative branch is working to provide more funding.
Vice President Pence’s new role does not change the command structure of the Coronavirus Task Force, which is currently led by Department of Health & Human Services Secretary Alex Azar.
Speaking at the press conference, Secretary Azar noted that he is still chairman of the task force. “Having the vice president gives me the biggest stick one can have in the government on this whole-of-government approach.”
He emphatically stated, “not in the least,” in response to a question about whether he felt he was being replaced. “When this was mentioned to me, I said I was delighted that I get to have the vice president helping in this way. Delighted.”
The announcement came as President Trump continued to downplay the threat of the coronavirus to U.S. citizens, going so far as to contradict CDC officials who have stated that it is a matter of when, not if, there will be community spread in the United States.
“I don’t think it’s inevitable,” President Trump said. “I think that there’s a chance that it could get worse. There’s a chance it could get fairly substantially worse, but nothing’s inevitable.”
Immediately after President Trump wrapped up his statement, however, the CDC formally announced the first case of possible community spread of the coronavirus. In a statement issued to the press, the agency announced the 15th confirmed case in the United States, a person in California “who reportedly did not have relevant travel history or exposure to another known patient” with the coronavirus.
“This case was detected through the U.S. public health system – picked up by astute clinicians,” CDC added, noting it will continue to provide updates on the evolving situation.
Vice President Mike Pence will be the White House point person quarterbacking the administration’s response to COVID-19, although President Donald Trump was quick to dismiss the notion that he is a so-called coronavirus “czar.”
President Trump introduced Vice President Pence in this role during a Feb. 26 press conference. The same night, officials at the Centers for Disease Control and Prevention announced the first case of possible community spread of the novel coronavirus in the United States.
“I am going to be putting our vice president, Mike Pence, in charge, and Mike will be working with the professionals, the doctors, and everybody else that is working” on this, President Trump said.
“Mike is going to be in charge and Mike will report back to me, but he’s got a certain talent for this,” President Trump continued, noting that while Vice President Pence was governor of Indiana, his was the first state to have a patient affected by the 2014 Middle East Respiratory Syndrome coronavirus (MERS-CoV) outbreak, so he has experience in a similar situation.
“I know full well the importance of presidential leadership, the importance of administration leadership, and the vital role of partnerships of state and local governments and health authorities in responding to the potential threat of dangerous infectious diseases,” Vice President Pence said.
He said that his role will be to continue to meet with the Coronavirus Task Force and bring to the president “the best options for action and to see to the safety and well being and health of the American people. I will also be continuing to reach out to governors [and] state and local officials.”
Vice President Pence said he will also be working with Congress to ensure that resources are available.
It was noted during the press conference that some members of Congress consider the $2.5 billion in emergency appropriations requested by the White House to be inadequate and that the legislative branch is working to provide more funding.
Vice President Pence’s new role does not change the command structure of the Coronavirus Task Force, which is currently led by Department of Health & Human Services Secretary Alex Azar.
Speaking at the press conference, Secretary Azar noted that he is still chairman of the task force. “Having the vice president gives me the biggest stick one can have in the government on this whole-of-government approach.”
He emphatically stated, “not in the least,” in response to a question about whether he felt he was being replaced. “When this was mentioned to me, I said I was delighted that I get to have the vice president helping in this way. Delighted.”
The announcement came as President Trump continued to downplay the threat of the coronavirus to U.S. citizens, going so far as to contradict CDC officials who have stated that it is a matter of when, not if, there will be community spread in the United States.
“I don’t think it’s inevitable,” President Trump said. “I think that there’s a chance that it could get worse. There’s a chance it could get fairly substantially worse, but nothing’s inevitable.”
Immediately after President Trump wrapped up his statement, however, the CDC formally announced the first case of possible community spread of the coronavirus. In a statement issued to the press, the agency announced the 15th confirmed case in the United States, a person in California “who reportedly did not have relevant travel history or exposure to another known patient” with the coronavirus.
“This case was detected through the U.S. public health system – picked up by astute clinicians,” CDC added, noting it will continue to provide updates on the evolving situation.
COVID-19: Time to ‘take the risk of scaring people’
It’s past time to call the novel coronavirus, COVID-19, a pandemic and “time to push people to prepare, and guide their prep,” according to risk communication experts.
Medical messaging about containing or stopping the spread of the virus is doing more harm than good, write Peter Sandman, PhD, and Jody Lanard, MD, both based in New York City, in a recent blog post.
“We are near-certain that the desperate-sounding last-ditch containment messaging of recent days is contributing to a massive global misperception,” they warn.
“The most crucial (and overdue) risk communication task … is to help people visualize their communities when ‘keeping it out’ – containment – is no longer relevant.”
That message is embraced by several experts who spoke to Medscape Medical News.
“I’m jealous of what [they] have written: It is so clear, so correct, and so practical,” said David Fisman, MD, MPH, professor of epidemiology at the University of Toronto, Canada. “I think WHO [World Health Organization] is shying away from the P word,” he continued, referring to the organization’s continuing decision not to call the outbreak a pandemic.
“I fully support exactly what [Sandman and Lanard] are saying,” said Michael Osterholm, PhD, MPH, professor of environmental health sciences and director of the Center for Infectious Disease Research and Policy (CIDRAP) at the University of Minnesota in Minneapolis.
Sandman and Lanard write. “Hardly any officials are telling civil society and the general public how to get ready for this pandemic.”
Effective communication should inform people of what to expect now, they continue: “[T]he end of most quarantines, travel restrictions, contact tracing, and other measures designed to keep ‘them’ from infecting ‘us,’ and the switch to measures like canceling mass events designed to keep us from infecting each other.”
Among the new messages that should be delivered are things like:
- Stockpiling nonperishable food and prescription meds.
- Considering care of sick family members.
- Cross-training work personnel so one person’s absence won’t derail an organization’s ability to function.
“We hope that governments and healthcare institutions are using this time wisely,” Sandman and Lanard continue. “We know that ordinary citizens are not being asked to do so. In most countries … ordinary citizens have not been asked to prepare. Instead, they have been led to expect that their governments will keep the virus from their doors.”
This article first appeared on Medscape.com.
It’s past time to call the novel coronavirus, COVID-19, a pandemic and “time to push people to prepare, and guide their prep,” according to risk communication experts.
Medical messaging about containing or stopping the spread of the virus is doing more harm than good, write Peter Sandman, PhD, and Jody Lanard, MD, both based in New York City, in a recent blog post.
“We are near-certain that the desperate-sounding last-ditch containment messaging of recent days is contributing to a massive global misperception,” they warn.
“The most crucial (and overdue) risk communication task … is to help people visualize their communities when ‘keeping it out’ – containment – is no longer relevant.”
That message is embraced by several experts who spoke to Medscape Medical News.
“I’m jealous of what [they] have written: It is so clear, so correct, and so practical,” said David Fisman, MD, MPH, professor of epidemiology at the University of Toronto, Canada. “I think WHO [World Health Organization] is shying away from the P word,” he continued, referring to the organization’s continuing decision not to call the outbreak a pandemic.
“I fully support exactly what [Sandman and Lanard] are saying,” said Michael Osterholm, PhD, MPH, professor of environmental health sciences and director of the Center for Infectious Disease Research and Policy (CIDRAP) at the University of Minnesota in Minneapolis.
Sandman and Lanard write. “Hardly any officials are telling civil society and the general public how to get ready for this pandemic.”
Effective communication should inform people of what to expect now, they continue: “[T]he end of most quarantines, travel restrictions, contact tracing, and other measures designed to keep ‘them’ from infecting ‘us,’ and the switch to measures like canceling mass events designed to keep us from infecting each other.”
Among the new messages that should be delivered are things like:
- Stockpiling nonperishable food and prescription meds.
- Considering care of sick family members.
- Cross-training work personnel so one person’s absence won’t derail an organization’s ability to function.
“We hope that governments and healthcare institutions are using this time wisely,” Sandman and Lanard continue. “We know that ordinary citizens are not being asked to do so. In most countries … ordinary citizens have not been asked to prepare. Instead, they have been led to expect that their governments will keep the virus from their doors.”
This article first appeared on Medscape.com.
It’s past time to call the novel coronavirus, COVID-19, a pandemic and “time to push people to prepare, and guide their prep,” according to risk communication experts.
Medical messaging about containing or stopping the spread of the virus is doing more harm than good, write Peter Sandman, PhD, and Jody Lanard, MD, both based in New York City, in a recent blog post.
“We are near-certain that the desperate-sounding last-ditch containment messaging of recent days is contributing to a massive global misperception,” they warn.
“The most crucial (and overdue) risk communication task … is to help people visualize their communities when ‘keeping it out’ – containment – is no longer relevant.”
That message is embraced by several experts who spoke to Medscape Medical News.
“I’m jealous of what [they] have written: It is so clear, so correct, and so practical,” said David Fisman, MD, MPH, professor of epidemiology at the University of Toronto, Canada. “I think WHO [World Health Organization] is shying away from the P word,” he continued, referring to the organization’s continuing decision not to call the outbreak a pandemic.
“I fully support exactly what [Sandman and Lanard] are saying,” said Michael Osterholm, PhD, MPH, professor of environmental health sciences and director of the Center for Infectious Disease Research and Policy (CIDRAP) at the University of Minnesota in Minneapolis.
Sandman and Lanard write. “Hardly any officials are telling civil society and the general public how to get ready for this pandemic.”
Effective communication should inform people of what to expect now, they continue: “[T]he end of most quarantines, travel restrictions, contact tracing, and other measures designed to keep ‘them’ from infecting ‘us,’ and the switch to measures like canceling mass events designed to keep us from infecting each other.”
Among the new messages that should be delivered are things like:
- Stockpiling nonperishable food and prescription meds.
- Considering care of sick family members.
- Cross-training work personnel so one person’s absence won’t derail an organization’s ability to function.
“We hope that governments and healthcare institutions are using this time wisely,” Sandman and Lanard continue. “We know that ordinary citizens are not being asked to do so. In most countries … ordinary citizens have not been asked to prepare. Instead, they have been led to expect that their governments will keep the virus from their doors.”
This article first appeared on Medscape.com.
CDC expects eventual community spread of coronavirus in U.S.
“We have for many weeks been saying that, while we hope this is not going to be severe, we are planning as if it is,” Nancy Messonnier, MD, director of the National Center for Immunization and Respiratory Diseases at the CDC, said during a Feb. 25, 2020, telebriefing with reporters. “The data over the last week and the spread in other countries has certainly raised our level of concern and raised our level expectation that we are going to have community spread here.”
Dr. Messonnier noted that the coronavirus is now showing signs of community spread without a known source of exposure in a number of countries, including in Hong Kong, Iran, Italy, Japan, Singapore, South Korea, Taiwan, and Thailand. This has now raised the belief that there will be more widespread outbreaks in the United States.
“What we still don’t know is what that will look like,” she said. “As many of you know, we can have community spread in the United States and have it be reasonably mild. We can have community spread in the U.S. and have it be very severe. That is what we don’t completely know yet and we certainly also don’t exactly know when it is going to happen.”
She reiterated the number of actions being taken to slow the potential spread in the United States, including detecting, tracking, and isolating all cases, as well as restricting travel into the United States and issuing travel advisories for countries where coronavirus outbreaks are known.
“We are doing this with the goal of slowing the introduction of this new virus into the U.S. and buying us more time to prepare,” Dr. Messonnier said, noting the containment strategies have been largely successful, though it will be more difficult as more countries experience community spread of the virus.
Dr. Messonnier also reiterated that at this time there are no vaccines and no medicines to treat the coronavirus. She stressed the need to adhere to nonpharmaceutical interventions (NPIs), as they will be “the most important tools in our response to this virus.”
She said the NPIs will vary based on the severity of the outbreak in any given local community and include personal protective measures that individuals can take every day (many of which mirror the recommendations for preventing the spread of the seasonal flu virus), community NPIs that involve social distancing measures designed to keep people away from others, and environmental NPIs such as surface cleaning measures.
CDC’s latest warning comes as parent agency the Department of Health & Human Services is seeking $2.5 billion in funds from Congress to address the coronavirus outbreak.
During a separate press conference on the same day, HHS Secretary Alex Azar noted that there are five major priorities related to those funds, which would be used in the current year, including expansion of surveillance work within the influenza surveillance network; supporting public health preparedness and response for state and local governments; support the development of therapeutics and the development of vaccines; and the purchase of personal protective equipment for national stockpiles.
Anthony S. Fauci, MD, director of the National Institute of Allergy and Infectious Disease at the National Institutes of Health, added during the press conference that vaccine work is in progress and could be ready for phase 1 testing within a month and a half. If all goes well, it would still be at least 12 - 18 months following the completion of a phase 2 trial before it could be produced for mass consumption.
“It is certainly conceivable that this issue with this coronavirus will go well beyond this season into next season,” Dr. Fauci said. “So a vaccine may not solve the problems of the next couple of months, but it certainly would be an important tool that we would have and we will keep you posted on that.”
He also mentioned that NIAID is looking at a number of candidates for therapeutic treatment of coronavirus. He highlighted Gilead’s remdesivir, a nucleotide analog, as one which undergoing two trials – a randomized controlled trial in China and a copy of that trial in Nebraska among patients with the coronavirus who were taken from the Diamond Princess cruise line in Japan.
“I am optimistic that we will at least get an answer if we do have do have a therapy that really is a gamechanger because then we could do something from the standpoint of intervention for those who are sick,” Dr. Fauci said.
UPDATE: This story was updated 2/25 at 4:51 p.m. ET
“We have for many weeks been saying that, while we hope this is not going to be severe, we are planning as if it is,” Nancy Messonnier, MD, director of the National Center for Immunization and Respiratory Diseases at the CDC, said during a Feb. 25, 2020, telebriefing with reporters. “The data over the last week and the spread in other countries has certainly raised our level of concern and raised our level expectation that we are going to have community spread here.”
Dr. Messonnier noted that the coronavirus is now showing signs of community spread without a known source of exposure in a number of countries, including in Hong Kong, Iran, Italy, Japan, Singapore, South Korea, Taiwan, and Thailand. This has now raised the belief that there will be more widespread outbreaks in the United States.
“What we still don’t know is what that will look like,” she said. “As many of you know, we can have community spread in the United States and have it be reasonably mild. We can have community spread in the U.S. and have it be very severe. That is what we don’t completely know yet and we certainly also don’t exactly know when it is going to happen.”
She reiterated the number of actions being taken to slow the potential spread in the United States, including detecting, tracking, and isolating all cases, as well as restricting travel into the United States and issuing travel advisories for countries where coronavirus outbreaks are known.
“We are doing this with the goal of slowing the introduction of this new virus into the U.S. and buying us more time to prepare,” Dr. Messonnier said, noting the containment strategies have been largely successful, though it will be more difficult as more countries experience community spread of the virus.
Dr. Messonnier also reiterated that at this time there are no vaccines and no medicines to treat the coronavirus. She stressed the need to adhere to nonpharmaceutical interventions (NPIs), as they will be “the most important tools in our response to this virus.”
She said the NPIs will vary based on the severity of the outbreak in any given local community and include personal protective measures that individuals can take every day (many of which mirror the recommendations for preventing the spread of the seasonal flu virus), community NPIs that involve social distancing measures designed to keep people away from others, and environmental NPIs such as surface cleaning measures.
CDC’s latest warning comes as parent agency the Department of Health & Human Services is seeking $2.5 billion in funds from Congress to address the coronavirus outbreak.
During a separate press conference on the same day, HHS Secretary Alex Azar noted that there are five major priorities related to those funds, which would be used in the current year, including expansion of surveillance work within the influenza surveillance network; supporting public health preparedness and response for state and local governments; support the development of therapeutics and the development of vaccines; and the purchase of personal protective equipment for national stockpiles.
Anthony S. Fauci, MD, director of the National Institute of Allergy and Infectious Disease at the National Institutes of Health, added during the press conference that vaccine work is in progress and could be ready for phase 1 testing within a month and a half. If all goes well, it would still be at least 12 - 18 months following the completion of a phase 2 trial before it could be produced for mass consumption.
“It is certainly conceivable that this issue with this coronavirus will go well beyond this season into next season,” Dr. Fauci said. “So a vaccine may not solve the problems of the next couple of months, but it certainly would be an important tool that we would have and we will keep you posted on that.”
He also mentioned that NIAID is looking at a number of candidates for therapeutic treatment of coronavirus. He highlighted Gilead’s remdesivir, a nucleotide analog, as one which undergoing two trials – a randomized controlled trial in China and a copy of that trial in Nebraska among patients with the coronavirus who were taken from the Diamond Princess cruise line in Japan.
“I am optimistic that we will at least get an answer if we do have do have a therapy that really is a gamechanger because then we could do something from the standpoint of intervention for those who are sick,” Dr. Fauci said.
UPDATE: This story was updated 2/25 at 4:51 p.m. ET
“We have for many weeks been saying that, while we hope this is not going to be severe, we are planning as if it is,” Nancy Messonnier, MD, director of the National Center for Immunization and Respiratory Diseases at the CDC, said during a Feb. 25, 2020, telebriefing with reporters. “The data over the last week and the spread in other countries has certainly raised our level of concern and raised our level expectation that we are going to have community spread here.”
Dr. Messonnier noted that the coronavirus is now showing signs of community spread without a known source of exposure in a number of countries, including in Hong Kong, Iran, Italy, Japan, Singapore, South Korea, Taiwan, and Thailand. This has now raised the belief that there will be more widespread outbreaks in the United States.
“What we still don’t know is what that will look like,” she said. “As many of you know, we can have community spread in the United States and have it be reasonably mild. We can have community spread in the U.S. and have it be very severe. That is what we don’t completely know yet and we certainly also don’t exactly know when it is going to happen.”
She reiterated the number of actions being taken to slow the potential spread in the United States, including detecting, tracking, and isolating all cases, as well as restricting travel into the United States and issuing travel advisories for countries where coronavirus outbreaks are known.
“We are doing this with the goal of slowing the introduction of this new virus into the U.S. and buying us more time to prepare,” Dr. Messonnier said, noting the containment strategies have been largely successful, though it will be more difficult as more countries experience community spread of the virus.
Dr. Messonnier also reiterated that at this time there are no vaccines and no medicines to treat the coronavirus. She stressed the need to adhere to nonpharmaceutical interventions (NPIs), as they will be “the most important tools in our response to this virus.”
She said the NPIs will vary based on the severity of the outbreak in any given local community and include personal protective measures that individuals can take every day (many of which mirror the recommendations for preventing the spread of the seasonal flu virus), community NPIs that involve social distancing measures designed to keep people away from others, and environmental NPIs such as surface cleaning measures.
CDC’s latest warning comes as parent agency the Department of Health & Human Services is seeking $2.5 billion in funds from Congress to address the coronavirus outbreak.
During a separate press conference on the same day, HHS Secretary Alex Azar noted that there are five major priorities related to those funds, which would be used in the current year, including expansion of surveillance work within the influenza surveillance network; supporting public health preparedness and response for state and local governments; support the development of therapeutics and the development of vaccines; and the purchase of personal protective equipment for national stockpiles.
Anthony S. Fauci, MD, director of the National Institute of Allergy and Infectious Disease at the National Institutes of Health, added during the press conference that vaccine work is in progress and could be ready for phase 1 testing within a month and a half. If all goes well, it would still be at least 12 - 18 months following the completion of a phase 2 trial before it could be produced for mass consumption.
“It is certainly conceivable that this issue with this coronavirus will go well beyond this season into next season,” Dr. Fauci said. “So a vaccine may not solve the problems of the next couple of months, but it certainly would be an important tool that we would have and we will keep you posted on that.”
He also mentioned that NIAID is looking at a number of candidates for therapeutic treatment of coronavirus. He highlighted Gilead’s remdesivir, a nucleotide analog, as one which undergoing two trials – a randomized controlled trial in China and a copy of that trial in Nebraska among patients with the coronavirus who were taken from the Diamond Princess cruise line in Japan.
“I am optimistic that we will at least get an answer if we do have do have a therapy that really is a gamechanger because then we could do something from the standpoint of intervention for those who are sick,” Dr. Fauci said.
UPDATE: This story was updated 2/25 at 4:51 p.m. ET
ID Blog: SARS-CoV-2 – What’s in a name?
Coming up with a moniker for the new coronavirus shows the perils of naming names.
There is no Baby Book of Names or hurricane alphabet to readily name diseases and their causal entities. Throughout history and even in the modern era, a host of considerations have intruded on the decision as to what to call these blights upon humanity. Names have varied from inflammatory to misleading, from colloquial to scientific. And when it concerns a new epidemiological entity such as the latest coronavirus outbreak originating in China, health organizations, media, politicians, scientific taxonomy commissions, and the public at large all have a stake in the naming.
From “Wuhan virus” to “novel coronavirus-2019” to “COVID-19 virus,” the name of the new coronavirus that first appeared in China has been evolving to its now official designation: SARS-CoV-2 (severe acute respiratory syndrome coronavirus 2). But where did the final name come from, how does such a name become official, and who makes it so?
Virus taxonomy
The Coronavirus Study Group (CSG) of the International Committee on Taxonomy of Viruses (ICTV) named the new coronavirus SARS-CoV-2 based upon its genetic relationship to the original SARS-CoV that caused an outbreak of disease in 2002–2003.
According to the ICTV website, the first internationally organized attempts to introduce order into the bewildering variety of viruses took place at the International Congress of Microbiology held in Moscow in 1966 where a committee was created that later became the ICTV and was given the task of developing a single, universal taxonomic scheme for all the viruses infecting animals, plants, fungi, bacteria, and archaea. The ICTV was created as a committee of the virology division of the International Union of Microbiological Societies and is governed by statutes approved by the virology division. Virus classification and nomenclature are subject to rules set out in an International Code.
These designate that: “The universal virus classification system shall employ the hierarchical levels of realm, subrealm, kingdom, subkingdom, phylum, subphylum, class, subclass, order, suborder, family, subfamily, genus, subgenus and species.”
Many of the topmost areas of classification are based on whether the viruses are DNA or RNA, single or double stranded, and have a simple protein shell or a complex lipoprotein envelope. Other levels of classification include host species, type of replication, and type of diseases they cause, the later exemplified in the SARS designation for this virus.
There are 98 international study groups (SGs) covering all major virus orders, families, and genera that are part of the ICTV, and it was the one dedicated to the single-stranded RNA coronaviruses, the CSG, that came up with the SARS-CoV-2 name and first referenced it in their Feb 11 publication in the Cold Springs Harbor preprint journal bioRxiv.
“Based on phylogeny, taxonomy and established practice, the CSG formally recognizes this virus as a sister to severe acute respiratory syndrome coronaviruses (SARS-CoVs) of the species severe acute respiratory syndrome–related coronavirus and designates it as severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2),” they wrote.
According to the National Center for Biotechnology Information Taxonomy Browser, with respect to the original SARS CoV virus, of which this is a relative, the full taxonomic designation is: Viruses, Riboviria, Nidovirales, Cornidovirineae, Coronaviridae, Orthocoronavirinae, Betacoronavirus, Sarbecovirus.
The problem with naming names
The World Health Organization currently is not using the official scientific name of the virus, but rather is merely labeling it with regard to the disease: COVID-19, which simply refers to coronavirus disease 2019.
They are following a modern standard by which disease names avoid inflammatory connotations with people and places. Too often in the past from syphilis as the “French pox,” the 1918 influenza as the “Spanish flu,” AIDS as the “gay plague,” Middle East Respiratory Syndrome (MERS), and the currently named “WuFlu,” which made an appearance early in the new outbreak and which is symbolic of a sudden wave of anti-Asian, and specifically Chinese, prejudice.
Chinatown districts even in the United States are being affected economically through unwarranted fear associated with the virus. And there have been equivalently virulent outbreaks of hate speech against Asian individuals in places untouched by the new virus.
However, although SARS-CoV-2 as a name avoids such problems, different considerations led the WHO to reject it in its discussions, determining that its use ties it to tightly to the much more deadly SARS-CoV-1 virus in the public mind, risking greater fear and panic, especially in Asia, where SARS-CoV-1 had the biggest impact.
Back in 1896, William Sykes, MD, writing in the first flush of the triumph of germ theory in modern medicine, attempted to give some guidance to how medical science should best come up with new names of diseases by merging the demands of common parlance with those of taxonomic legitimacy. His “On the Origin and History of Disease-Names,” published in the Lancet, had clearcut advice: “It is vain to attempt to replace a folk name or one widely adopted by the people by a new one deliberately coined by scholars, and this for the following reasons: first, whatever names may be accepted by medical men must be translated by them into the vernacular of their patients, and by a resulting reaction the vernacular name comes to be the commoner one with themselves; and, secondly, there is no continuity or unchangeableness in the terms invented by savants, which are amended, improved upon, and displaced by the next writer on the subject, or, even more absurdly still, by the very inventors themselves in a subsequent publication.”
This is the reason that virus taxonomy provides names based upon unchangeable scientific descriptors of the actual disease causing entity, as illustrated by the decisions of the ICTV. In addition, the genomic sequences being provided by the scientific community are all being organized under the SARS-CoV-2 name and thus are cementing that moniker as the only acceptable scientific one.
Whether the rest of the world universally adopts SARS-CoV-2 as a name is still in question. If the outbreak spreads significantly beyond its current limits, fear and confusion – and simply the need for a more familiar-sounding label – may lead the general public to adopt more colloquial designations than those that science attempts to impose, as Dr. Sykes suggested back in 1896. That remains to be seen.
[email protected]
Mark Lesney is the managing editor of MDedge.com/IDPractioner. He has a PhD in plant virology and a PhD in the history of science, with a focus on the history of biotechnology and medicine. He has served as an adjunct assistant professor of the department of biochemistry and molecular & cellular biology at Georgetown University, Washington.
Coming up with a moniker for the new coronavirus shows the perils of naming names.
Coming up with a moniker for the new coronavirus shows the perils of naming names.
There is no Baby Book of Names or hurricane alphabet to readily name diseases and their causal entities. Throughout history and even in the modern era, a host of considerations have intruded on the decision as to what to call these blights upon humanity. Names have varied from inflammatory to misleading, from colloquial to scientific. And when it concerns a new epidemiological entity such as the latest coronavirus outbreak originating in China, health organizations, media, politicians, scientific taxonomy commissions, and the public at large all have a stake in the naming.
From “Wuhan virus” to “novel coronavirus-2019” to “COVID-19 virus,” the name of the new coronavirus that first appeared in China has been evolving to its now official designation: SARS-CoV-2 (severe acute respiratory syndrome coronavirus 2). But where did the final name come from, how does such a name become official, and who makes it so?
Virus taxonomy
The Coronavirus Study Group (CSG) of the International Committee on Taxonomy of Viruses (ICTV) named the new coronavirus SARS-CoV-2 based upon its genetic relationship to the original SARS-CoV that caused an outbreak of disease in 2002–2003.
According to the ICTV website, the first internationally organized attempts to introduce order into the bewildering variety of viruses took place at the International Congress of Microbiology held in Moscow in 1966 where a committee was created that later became the ICTV and was given the task of developing a single, universal taxonomic scheme for all the viruses infecting animals, plants, fungi, bacteria, and archaea. The ICTV was created as a committee of the virology division of the International Union of Microbiological Societies and is governed by statutes approved by the virology division. Virus classification and nomenclature are subject to rules set out in an International Code.
These designate that: “The universal virus classification system shall employ the hierarchical levels of realm, subrealm, kingdom, subkingdom, phylum, subphylum, class, subclass, order, suborder, family, subfamily, genus, subgenus and species.”
Many of the topmost areas of classification are based on whether the viruses are DNA or RNA, single or double stranded, and have a simple protein shell or a complex lipoprotein envelope. Other levels of classification include host species, type of replication, and type of diseases they cause, the later exemplified in the SARS designation for this virus.
There are 98 international study groups (SGs) covering all major virus orders, families, and genera that are part of the ICTV, and it was the one dedicated to the single-stranded RNA coronaviruses, the CSG, that came up with the SARS-CoV-2 name and first referenced it in their Feb 11 publication in the Cold Springs Harbor preprint journal bioRxiv.
“Based on phylogeny, taxonomy and established practice, the CSG formally recognizes this virus as a sister to severe acute respiratory syndrome coronaviruses (SARS-CoVs) of the species severe acute respiratory syndrome–related coronavirus and designates it as severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2),” they wrote.
According to the National Center for Biotechnology Information Taxonomy Browser, with respect to the original SARS CoV virus, of which this is a relative, the full taxonomic designation is: Viruses, Riboviria, Nidovirales, Cornidovirineae, Coronaviridae, Orthocoronavirinae, Betacoronavirus, Sarbecovirus.
The problem with naming names
The World Health Organization currently is not using the official scientific name of the virus, but rather is merely labeling it with regard to the disease: COVID-19, which simply refers to coronavirus disease 2019.
They are following a modern standard by which disease names avoid inflammatory connotations with people and places. Too often in the past from syphilis as the “French pox,” the 1918 influenza as the “Spanish flu,” AIDS as the “gay plague,” Middle East Respiratory Syndrome (MERS), and the currently named “WuFlu,” which made an appearance early in the new outbreak and which is symbolic of a sudden wave of anti-Asian, and specifically Chinese, prejudice.
Chinatown districts even in the United States are being affected economically through unwarranted fear associated with the virus. And there have been equivalently virulent outbreaks of hate speech against Asian individuals in places untouched by the new virus.
However, although SARS-CoV-2 as a name avoids such problems, different considerations led the WHO to reject it in its discussions, determining that its use ties it to tightly to the much more deadly SARS-CoV-1 virus in the public mind, risking greater fear and panic, especially in Asia, where SARS-CoV-1 had the biggest impact.
Back in 1896, William Sykes, MD, writing in the first flush of the triumph of germ theory in modern medicine, attempted to give some guidance to how medical science should best come up with new names of diseases by merging the demands of common parlance with those of taxonomic legitimacy. His “On the Origin and History of Disease-Names,” published in the Lancet, had clearcut advice: “It is vain to attempt to replace a folk name or one widely adopted by the people by a new one deliberately coined by scholars, and this for the following reasons: first, whatever names may be accepted by medical men must be translated by them into the vernacular of their patients, and by a resulting reaction the vernacular name comes to be the commoner one with themselves; and, secondly, there is no continuity or unchangeableness in the terms invented by savants, which are amended, improved upon, and displaced by the next writer on the subject, or, even more absurdly still, by the very inventors themselves in a subsequent publication.”
This is the reason that virus taxonomy provides names based upon unchangeable scientific descriptors of the actual disease causing entity, as illustrated by the decisions of the ICTV. In addition, the genomic sequences being provided by the scientific community are all being organized under the SARS-CoV-2 name and thus are cementing that moniker as the only acceptable scientific one.
Whether the rest of the world universally adopts SARS-CoV-2 as a name is still in question. If the outbreak spreads significantly beyond its current limits, fear and confusion – and simply the need for a more familiar-sounding label – may lead the general public to adopt more colloquial designations than those that science attempts to impose, as Dr. Sykes suggested back in 1896. That remains to be seen.
[email protected]
Mark Lesney is the managing editor of MDedge.com/IDPractioner. He has a PhD in plant virology and a PhD in the history of science, with a focus on the history of biotechnology and medicine. He has served as an adjunct assistant professor of the department of biochemistry and molecular & cellular biology at Georgetown University, Washington.
There is no Baby Book of Names or hurricane alphabet to readily name diseases and their causal entities. Throughout history and even in the modern era, a host of considerations have intruded on the decision as to what to call these blights upon humanity. Names have varied from inflammatory to misleading, from colloquial to scientific. And when it concerns a new epidemiological entity such as the latest coronavirus outbreak originating in China, health organizations, media, politicians, scientific taxonomy commissions, and the public at large all have a stake in the naming.
From “Wuhan virus” to “novel coronavirus-2019” to “COVID-19 virus,” the name of the new coronavirus that first appeared in China has been evolving to its now official designation: SARS-CoV-2 (severe acute respiratory syndrome coronavirus 2). But where did the final name come from, how does such a name become official, and who makes it so?
Virus taxonomy
The Coronavirus Study Group (CSG) of the International Committee on Taxonomy of Viruses (ICTV) named the new coronavirus SARS-CoV-2 based upon its genetic relationship to the original SARS-CoV that caused an outbreak of disease in 2002–2003.
According to the ICTV website, the first internationally organized attempts to introduce order into the bewildering variety of viruses took place at the International Congress of Microbiology held in Moscow in 1966 where a committee was created that later became the ICTV and was given the task of developing a single, universal taxonomic scheme for all the viruses infecting animals, plants, fungi, bacteria, and archaea. The ICTV was created as a committee of the virology division of the International Union of Microbiological Societies and is governed by statutes approved by the virology division. Virus classification and nomenclature are subject to rules set out in an International Code.
These designate that: “The universal virus classification system shall employ the hierarchical levels of realm, subrealm, kingdom, subkingdom, phylum, subphylum, class, subclass, order, suborder, family, subfamily, genus, subgenus and species.”
Many of the topmost areas of classification are based on whether the viruses are DNA or RNA, single or double stranded, and have a simple protein shell or a complex lipoprotein envelope. Other levels of classification include host species, type of replication, and type of diseases they cause, the later exemplified in the SARS designation for this virus.
There are 98 international study groups (SGs) covering all major virus orders, families, and genera that are part of the ICTV, and it was the one dedicated to the single-stranded RNA coronaviruses, the CSG, that came up with the SARS-CoV-2 name and first referenced it in their Feb 11 publication in the Cold Springs Harbor preprint journal bioRxiv.
“Based on phylogeny, taxonomy and established practice, the CSG formally recognizes this virus as a sister to severe acute respiratory syndrome coronaviruses (SARS-CoVs) of the species severe acute respiratory syndrome–related coronavirus and designates it as severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2),” they wrote.
According to the National Center for Biotechnology Information Taxonomy Browser, with respect to the original SARS CoV virus, of which this is a relative, the full taxonomic designation is: Viruses, Riboviria, Nidovirales, Cornidovirineae, Coronaviridae, Orthocoronavirinae, Betacoronavirus, Sarbecovirus.
The problem with naming names
The World Health Organization currently is not using the official scientific name of the virus, but rather is merely labeling it with regard to the disease: COVID-19, which simply refers to coronavirus disease 2019.
They are following a modern standard by which disease names avoid inflammatory connotations with people and places. Too often in the past from syphilis as the “French pox,” the 1918 influenza as the “Spanish flu,” AIDS as the “gay plague,” Middle East Respiratory Syndrome (MERS), and the currently named “WuFlu,” which made an appearance early in the new outbreak and which is symbolic of a sudden wave of anti-Asian, and specifically Chinese, prejudice.
Chinatown districts even in the United States are being affected economically through unwarranted fear associated with the virus. And there have been equivalently virulent outbreaks of hate speech against Asian individuals in places untouched by the new virus.
However, although SARS-CoV-2 as a name avoids such problems, different considerations led the WHO to reject it in its discussions, determining that its use ties it to tightly to the much more deadly SARS-CoV-1 virus in the public mind, risking greater fear and panic, especially in Asia, where SARS-CoV-1 had the biggest impact.
Back in 1896, William Sykes, MD, writing in the first flush of the triumph of germ theory in modern medicine, attempted to give some guidance to how medical science should best come up with new names of diseases by merging the demands of common parlance with those of taxonomic legitimacy. His “On the Origin and History of Disease-Names,” published in the Lancet, had clearcut advice: “It is vain to attempt to replace a folk name or one widely adopted by the people by a new one deliberately coined by scholars, and this for the following reasons: first, whatever names may be accepted by medical men must be translated by them into the vernacular of their patients, and by a resulting reaction the vernacular name comes to be the commoner one with themselves; and, secondly, there is no continuity or unchangeableness in the terms invented by savants, which are amended, improved upon, and displaced by the next writer on the subject, or, even more absurdly still, by the very inventors themselves in a subsequent publication.”
This is the reason that virus taxonomy provides names based upon unchangeable scientific descriptors of the actual disease causing entity, as illustrated by the decisions of the ICTV. In addition, the genomic sequences being provided by the scientific community are all being organized under the SARS-CoV-2 name and thus are cementing that moniker as the only acceptable scientific one.
Whether the rest of the world universally adopts SARS-CoV-2 as a name is still in question. If the outbreak spreads significantly beyond its current limits, fear and confusion – and simply the need for a more familiar-sounding label – may lead the general public to adopt more colloquial designations than those that science attempts to impose, as Dr. Sykes suggested back in 1896. That remains to be seen.
[email protected]
Mark Lesney is the managing editor of MDedge.com/IDPractioner. He has a PhD in plant virology and a PhD in the history of science, with a focus on the history of biotechnology and medicine. He has served as an adjunct assistant professor of the department of biochemistry and molecular & cellular biology at Georgetown University, Washington.