User login
Nocturnal hypoxemia predicts incident atrial fibrillation in sleep apnea patients
Researchers have found a significant independent association between nocturnal hypoxemia and risk of incident atrial fibrillation (AF) in patients with obstructive sleep apnea (OSA), which they believe may help prevent the development of AF in this population, according to recent research published in the journal CHEST®.
“These findings are consistent with those of previous studies suggesting that these groups, who are typically at somewhat lower risk of [atrial fibrillation], might be especially vulnerable to the effects of [obstructive sleep apnea] and hypoxemia,” Tetyana Kendzerska, MD, PhD, of the University of Ottawa in Ottawa, and her colleagues wrote in their study.
They performed an analysis of 8,256 patients with data linked to a provincial health administrative database who had suspected OSA who underwent a sleep study at a large academic hospital between 1994 and 2010. The patients were median 47 years old; 62% of the cohort were men, 28% had an apnea-hypopnea index (AHI) of greater than 30 events per hour, and 6% spent more than 30% of the time during sleep with less than 90% oxygen saturation.
Overall, 173 of 8,256 patients (2.1%) developed AF during the study period. In patients with suspected OSA and who were arrhythmia free, nocturnal hypoxemia significantly increased the risk of incident hospitalized AF (hazard ratio, 2.47; 95% confidence interval, 1.64-3.71) over median 10 years of follow-up (interquartile range, 7-13 years) after the researchers controlled for age, sex, chronic obstructive pulmonary disease, history of heart failure, smoking status, nocturnal hypoxemia, and pulmonary embolism, and this association remained significant after adjustment for body mass index and hypertension (HR, 1.77; 95% CI, 1.15-2.74).
“These findings support a relationship between OSA, chronic nocturnal hypoxemia, and the development of [atrial fibrillation], and may be used to identify those patients with OSA who are at greatest risk of developing AF.”
Researchers cited the observational design, retrospective data collection, and examining hospitalized AF only as limitations in the study. However, they noted that clinical data was collected prospectively and the inability to adjust to positive airway pressure would push results to the null with regard to unstudied confounders.
The authors reported no relevant conflicts of interest.
SOURCE: Kendzerska T et al. CHEST. 2018;doi:10.1016/j.chest.2018.08.1075.
Researchers have found a significant independent association between nocturnal hypoxemia and risk of incident atrial fibrillation (AF) in patients with obstructive sleep apnea (OSA), which they believe may help prevent the development of AF in this population, according to recent research published in the journal CHEST®.
“These findings are consistent with those of previous studies suggesting that these groups, who are typically at somewhat lower risk of [atrial fibrillation], might be especially vulnerable to the effects of [obstructive sleep apnea] and hypoxemia,” Tetyana Kendzerska, MD, PhD, of the University of Ottawa in Ottawa, and her colleagues wrote in their study.
They performed an analysis of 8,256 patients with data linked to a provincial health administrative database who had suspected OSA who underwent a sleep study at a large academic hospital between 1994 and 2010. The patients were median 47 years old; 62% of the cohort were men, 28% had an apnea-hypopnea index (AHI) of greater than 30 events per hour, and 6% spent more than 30% of the time during sleep with less than 90% oxygen saturation.
Overall, 173 of 8,256 patients (2.1%) developed AF during the study period. In patients with suspected OSA and who were arrhythmia free, nocturnal hypoxemia significantly increased the risk of incident hospitalized AF (hazard ratio, 2.47; 95% confidence interval, 1.64-3.71) over median 10 years of follow-up (interquartile range, 7-13 years) after the researchers controlled for age, sex, chronic obstructive pulmonary disease, history of heart failure, smoking status, nocturnal hypoxemia, and pulmonary embolism, and this association remained significant after adjustment for body mass index and hypertension (HR, 1.77; 95% CI, 1.15-2.74).
“These findings support a relationship between OSA, chronic nocturnal hypoxemia, and the development of [atrial fibrillation], and may be used to identify those patients with OSA who are at greatest risk of developing AF.”
Researchers cited the observational design, retrospective data collection, and examining hospitalized AF only as limitations in the study. However, they noted that clinical data was collected prospectively and the inability to adjust to positive airway pressure would push results to the null with regard to unstudied confounders.
The authors reported no relevant conflicts of interest.
SOURCE: Kendzerska T et al. CHEST. 2018;doi:10.1016/j.chest.2018.08.1075.
Researchers have found a significant independent association between nocturnal hypoxemia and risk of incident atrial fibrillation (AF) in patients with obstructive sleep apnea (OSA), which they believe may help prevent the development of AF in this population, according to recent research published in the journal CHEST®.
“These findings are consistent with those of previous studies suggesting that these groups, who are typically at somewhat lower risk of [atrial fibrillation], might be especially vulnerable to the effects of [obstructive sleep apnea] and hypoxemia,” Tetyana Kendzerska, MD, PhD, of the University of Ottawa in Ottawa, and her colleagues wrote in their study.
They performed an analysis of 8,256 patients with data linked to a provincial health administrative database who had suspected OSA who underwent a sleep study at a large academic hospital between 1994 and 2010. The patients were median 47 years old; 62% of the cohort were men, 28% had an apnea-hypopnea index (AHI) of greater than 30 events per hour, and 6% spent more than 30% of the time during sleep with less than 90% oxygen saturation.
Overall, 173 of 8,256 patients (2.1%) developed AF during the study period. In patients with suspected OSA and who were arrhythmia free, nocturnal hypoxemia significantly increased the risk of incident hospitalized AF (hazard ratio, 2.47; 95% confidence interval, 1.64-3.71) over median 10 years of follow-up (interquartile range, 7-13 years) after the researchers controlled for age, sex, chronic obstructive pulmonary disease, history of heart failure, smoking status, nocturnal hypoxemia, and pulmonary embolism, and this association remained significant after adjustment for body mass index and hypertension (HR, 1.77; 95% CI, 1.15-2.74).
“These findings support a relationship between OSA, chronic nocturnal hypoxemia, and the development of [atrial fibrillation], and may be used to identify those patients with OSA who are at greatest risk of developing AF.”
Researchers cited the observational design, retrospective data collection, and examining hospitalized AF only as limitations in the study. However, they noted that clinical data was collected prospectively and the inability to adjust to positive airway pressure would push results to the null with regard to unstudied confounders.
The authors reported no relevant conflicts of interest.
SOURCE: Kendzerska T et al. CHEST. 2018;doi:10.1016/j.chest.2018.08.1075.
FROM CHEST
Key clinical point: In patients with suspected obstructive sleep apnea and who were arrhythmia free, nocturnal hypoxemia increased the risk of incident hospitalized atrial fibrillation by 77%.
Major finding: Nocturnal hypoxemia significantly predicted atrial fibrillation (hazard ratio, 2.47; 95% confidence interval, 1.64-3.71) and remained significant after adjustment for body mass index and hypertension (HR, 1.77; 95% CI, 1.15-2.74).
Study details: An observational study of 8,256 arrhythmia-free patients with suspected obstructive sleep apnea who underwent a sleep study at a large academic hospital between 1994 and 2010.
Disclosures: The authors report no relevant conflicts of interest.
Source: Kendzerska T et al. CHEST. 2018;doi:10.1016/j.chest.2018.08.1075.
Single-item scale effective for assessing sleep quality
The single-item sleep quality scale (SQS) produced favorable results comparable to other complex, time-intensive assessment tools, according to findings published in the Journal of Clinical Sleep Medicine.
In a study of 70 insomnia patients and 651 depression patients, concurrent criterion validity analysis yielded strong correlations between the SQS and the morning-questionnaire insomnia (MQI) and Pittsburgh Sleep Quality Index (PSQI) in patients with insomnia and depression, respectively. The investigators wrote, “The single-item format enables a patient-reported rating of sleep quality over a 7-day recall period without greatly increasing the patient’s burden. The use of a discretizing visual analog scale (VAS) increases the potential for a more sensitive measurement.” The SQS is a quick but accurate self-reported assessment of sleep quality.
The SQS was validated based on two studies. Eligible patients in the 4-week, randomized, multicenter insomnia study were aged 30-75 years and were receiving a Food and Drug Administration–approved hypnotic agent as usual treatment for insomnia based on criteria from the Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition (DSM-IV). The MQI was used daily for the duration of the study, wrote Ellen Snyder, PhD, of Merck & Co., in Kenilworth, N.J., and her coauthors.
The depression study was a randomized, double-blind, parallel-group, 12-month international trial evaluating the safety of the substance P antagonist aprepitant, compared with paroxetine hydrochloride. Patients were aged 18 years or older, with a DSM-IV diagnosis of major depressive disorder. Patients completed the SQS and PSQI at baseline, week 1, and week 8.
In insomnia patients, a Pearson correlation of –.76 was found at week 1 for the SQS in relation to the MQI.
In patients with depression, Goodman-Kruskal correlation coefficients for the SQS in relation to the Pittsburgh Sleep Quality Index (PSQI) were –.87, –.88, and –.92 at baseline, week 1, and week 8, respectively.
Correlations were negative because “better sleep quality is associated with a lower score on the MQI and PSQI, but a higher score on the SQS,” the authors noted.
The results support the use of the SQS as a “practical sleep measure that can effectively gauge sleep quality without significantly increasing the burden of clinical trial participants,” compared with lengthier assessments such as the MQI and PSQI, they added.
Funding for the study was provided by Merck Sharp & Dohme.
SOURCE: Snyder E et al. J Clin Sleep Med. 2018;14(11):1849-57.
The single-item sleep quality scale (SQS) produced favorable results comparable to other complex, time-intensive assessment tools, according to findings published in the Journal of Clinical Sleep Medicine.
In a study of 70 insomnia patients and 651 depression patients, concurrent criterion validity analysis yielded strong correlations between the SQS and the morning-questionnaire insomnia (MQI) and Pittsburgh Sleep Quality Index (PSQI) in patients with insomnia and depression, respectively. The investigators wrote, “The single-item format enables a patient-reported rating of sleep quality over a 7-day recall period without greatly increasing the patient’s burden. The use of a discretizing visual analog scale (VAS) increases the potential for a more sensitive measurement.” The SQS is a quick but accurate self-reported assessment of sleep quality.
The SQS was validated based on two studies. Eligible patients in the 4-week, randomized, multicenter insomnia study were aged 30-75 years and were receiving a Food and Drug Administration–approved hypnotic agent as usual treatment for insomnia based on criteria from the Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition (DSM-IV). The MQI was used daily for the duration of the study, wrote Ellen Snyder, PhD, of Merck & Co., in Kenilworth, N.J., and her coauthors.
The depression study was a randomized, double-blind, parallel-group, 12-month international trial evaluating the safety of the substance P antagonist aprepitant, compared with paroxetine hydrochloride. Patients were aged 18 years or older, with a DSM-IV diagnosis of major depressive disorder. Patients completed the SQS and PSQI at baseline, week 1, and week 8.
In insomnia patients, a Pearson correlation of –.76 was found at week 1 for the SQS in relation to the MQI.
In patients with depression, Goodman-Kruskal correlation coefficients for the SQS in relation to the Pittsburgh Sleep Quality Index (PSQI) were –.87, –.88, and –.92 at baseline, week 1, and week 8, respectively.
Correlations were negative because “better sleep quality is associated with a lower score on the MQI and PSQI, but a higher score on the SQS,” the authors noted.
The results support the use of the SQS as a “practical sleep measure that can effectively gauge sleep quality without significantly increasing the burden of clinical trial participants,” compared with lengthier assessments such as the MQI and PSQI, they added.
Funding for the study was provided by Merck Sharp & Dohme.
SOURCE: Snyder E et al. J Clin Sleep Med. 2018;14(11):1849-57.
The single-item sleep quality scale (SQS) produced favorable results comparable to other complex, time-intensive assessment tools, according to findings published in the Journal of Clinical Sleep Medicine.
In a study of 70 insomnia patients and 651 depression patients, concurrent criterion validity analysis yielded strong correlations between the SQS and the morning-questionnaire insomnia (MQI) and Pittsburgh Sleep Quality Index (PSQI) in patients with insomnia and depression, respectively. The investigators wrote, “The single-item format enables a patient-reported rating of sleep quality over a 7-day recall period without greatly increasing the patient’s burden. The use of a discretizing visual analog scale (VAS) increases the potential for a more sensitive measurement.” The SQS is a quick but accurate self-reported assessment of sleep quality.
The SQS was validated based on two studies. Eligible patients in the 4-week, randomized, multicenter insomnia study were aged 30-75 years and were receiving a Food and Drug Administration–approved hypnotic agent as usual treatment for insomnia based on criteria from the Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition (DSM-IV). The MQI was used daily for the duration of the study, wrote Ellen Snyder, PhD, of Merck & Co., in Kenilworth, N.J., and her coauthors.
The depression study was a randomized, double-blind, parallel-group, 12-month international trial evaluating the safety of the substance P antagonist aprepitant, compared with paroxetine hydrochloride. Patients were aged 18 years or older, with a DSM-IV diagnosis of major depressive disorder. Patients completed the SQS and PSQI at baseline, week 1, and week 8.
In insomnia patients, a Pearson correlation of –.76 was found at week 1 for the SQS in relation to the MQI.
In patients with depression, Goodman-Kruskal correlation coefficients for the SQS in relation to the Pittsburgh Sleep Quality Index (PSQI) were –.87, –.88, and –.92 at baseline, week 1, and week 8, respectively.
Correlations were negative because “better sleep quality is associated with a lower score on the MQI and PSQI, but a higher score on the SQS,” the authors noted.
The results support the use of the SQS as a “practical sleep measure that can effectively gauge sleep quality without significantly increasing the burden of clinical trial participants,” compared with lengthier assessments such as the MQI and PSQI, they added.
Funding for the study was provided by Merck Sharp & Dohme.
SOURCE: Snyder E et al. J Clin Sleep Med. 2018;14(11):1849-57.
FROM THE JOURNAL OF CLINICAL SLEEP MEDICINE
Key clinical point: The
Major finding: Week 1 Pearson correlation was –0.76 between the SQS and the morning-questionnaire insomnia (MQI); week 8 Goodman-Kruskal correlation between SQS and the Pittsburgh Sleep Quality Index (PSQI) was –0.92.
Study details: An analysis of SQS versus other measures in 70 insomnia patients and 651 depression patients.
Disclosures: Funding for the study was provided by Merck Sharp & Dohme.
Source: Snyder E et al. J Clin Sleep Med. 2018;14(11):1849-57
Clinical trial: Treating Sleep Apnea in Female Veterans
The Treating Sleep Apnea in Women Veterans study is an interventional, randomized, controlled trial for woman veterans recently diagnosed with sleep apnea and prescribed positive airway pressure (PAP) treatment with at least one risk factor for sleep-disordered breathing (SDB).
The trial will compare the efficacy of two different programs combining patient education with behavioral techniques to improve adherence to PAP therapy in women veterans. The recommended first-line treatment for SDB is PAP therapy, but women have been shown to have lower adherence to men, especially in woman veterans, who experience significant sleep disturbance and other consequences of sleep disorders. No study has tested possible interventions to increase PAP adherence in this population.
Among inclusion criteria are being a woman veteran aged at least 18 years, having received care from a Veterans Affairs facility, and having been diagnosed with sleep apnea with an apnea-hypopnea index of 5 or greater. Patients will be excluded if they are currently using a sleep apnea treatment, are pregnant, are an active substance user or started recovery in the past 90 days, are too ill to participate, do not have transportation, cannot self-consent, have unstable housing, have another sleep disorder that accounts for sleep disturbance, or have no sleep complaints or symptoms.
The primary outcome measures are PAP adherence 3 months post PAP initiation and sleep quality by patient-reported sleep questionnaire 3 months after randomizations. In addition, remote PAP usage data will be collected for 12 months post PAP initiation.
The estimated primary completion date is Dec. 21, 2021, and the estimated study completion date is Jan. 31, 2022. About 300 patients are estimated to be recruited.
Find more information on the study page at Clinicaltrials.gov.
The Treating Sleep Apnea in Women Veterans study is an interventional, randomized, controlled trial for woman veterans recently diagnosed with sleep apnea and prescribed positive airway pressure (PAP) treatment with at least one risk factor for sleep-disordered breathing (SDB).
The trial will compare the efficacy of two different programs combining patient education with behavioral techniques to improve adherence to PAP therapy in women veterans. The recommended first-line treatment for SDB is PAP therapy, but women have been shown to have lower adherence to men, especially in woman veterans, who experience significant sleep disturbance and other consequences of sleep disorders. No study has tested possible interventions to increase PAP adherence in this population.
Among inclusion criteria are being a woman veteran aged at least 18 years, having received care from a Veterans Affairs facility, and having been diagnosed with sleep apnea with an apnea-hypopnea index of 5 or greater. Patients will be excluded if they are currently using a sleep apnea treatment, are pregnant, are an active substance user or started recovery in the past 90 days, are too ill to participate, do not have transportation, cannot self-consent, have unstable housing, have another sleep disorder that accounts for sleep disturbance, or have no sleep complaints or symptoms.
The primary outcome measures are PAP adherence 3 months post PAP initiation and sleep quality by patient-reported sleep questionnaire 3 months after randomizations. In addition, remote PAP usage data will be collected for 12 months post PAP initiation.
The estimated primary completion date is Dec. 21, 2021, and the estimated study completion date is Jan. 31, 2022. About 300 patients are estimated to be recruited.
Find more information on the study page at Clinicaltrials.gov.
The Treating Sleep Apnea in Women Veterans study is an interventional, randomized, controlled trial for woman veterans recently diagnosed with sleep apnea and prescribed positive airway pressure (PAP) treatment with at least one risk factor for sleep-disordered breathing (SDB).
The trial will compare the efficacy of two different programs combining patient education with behavioral techniques to improve adherence to PAP therapy in women veterans. The recommended first-line treatment for SDB is PAP therapy, but women have been shown to have lower adherence to men, especially in woman veterans, who experience significant sleep disturbance and other consequences of sleep disorders. No study has tested possible interventions to increase PAP adherence in this population.
Among inclusion criteria are being a woman veteran aged at least 18 years, having received care from a Veterans Affairs facility, and having been diagnosed with sleep apnea with an apnea-hypopnea index of 5 or greater. Patients will be excluded if they are currently using a sleep apnea treatment, are pregnant, are an active substance user or started recovery in the past 90 days, are too ill to participate, do not have transportation, cannot self-consent, have unstable housing, have another sleep disorder that accounts for sleep disturbance, or have no sleep complaints or symptoms.
The primary outcome measures are PAP adherence 3 months post PAP initiation and sleep quality by patient-reported sleep questionnaire 3 months after randomizations. In addition, remote PAP usage data will be collected for 12 months post PAP initiation.
The estimated primary completion date is Dec. 21, 2021, and the estimated study completion date is Jan. 31, 2022. About 300 patients are estimated to be recruited.
Find more information on the study page at Clinicaltrials.gov.
Clinical trial: Magnetic Resonance Imaging in Obstructive Sleep Apnea
The Magnetic Resonance Imaging in Obstructive Sleep Apnea trial is an observational cohort study recruiting adults with obstructive sleep apnea undergoing surgery.
The trial will compare drug-induced sleep endoscopy and upper airway MRI in order to determine which is the better predictor of success in patients who cannot tolerate nonsurgical solutions. Upper airway MRI is a more complete evaluation during wakefulness and is cheaper than drug-induced sleep endoscopy, but no studies have thus far utilized MRI as a surgical evaluation tool.
Patients will be included if they are at least 21 years old, have moderate to severe obstructive sleep apnea, and have a body mass index less than 40 kg/m2. Exclusion criteria include prior surgery for obstructive sleep apnea; known neurologic, cardiac, pulmonary, renal, or hepatic disorders; psychiatric problems except for treated depression or mild anxiety; a coexisting sleep disorder; or another contraindication to drug-induced sleep endoscopy or MRI, such as propofol allergy.
The primary outcome measure is surgical results after 6 months, which will be measured using sleep studies. Secondary outcomes include sleep-related quality of life after 6 months and daytime sleepiness after 6 months.
The estimated primary completion date is June 2020, and the estimated study completion date is July 2020. About 40 patients are expected to be recruited.
Find more information on the study page at Clinicaltrials.gov.
The Magnetic Resonance Imaging in Obstructive Sleep Apnea trial is an observational cohort study recruiting adults with obstructive sleep apnea undergoing surgery.
The trial will compare drug-induced sleep endoscopy and upper airway MRI in order to determine which is the better predictor of success in patients who cannot tolerate nonsurgical solutions. Upper airway MRI is a more complete evaluation during wakefulness and is cheaper than drug-induced sleep endoscopy, but no studies have thus far utilized MRI as a surgical evaluation tool.
Patients will be included if they are at least 21 years old, have moderate to severe obstructive sleep apnea, and have a body mass index less than 40 kg/m2. Exclusion criteria include prior surgery for obstructive sleep apnea; known neurologic, cardiac, pulmonary, renal, or hepatic disorders; psychiatric problems except for treated depression or mild anxiety; a coexisting sleep disorder; or another contraindication to drug-induced sleep endoscopy or MRI, such as propofol allergy.
The primary outcome measure is surgical results after 6 months, which will be measured using sleep studies. Secondary outcomes include sleep-related quality of life after 6 months and daytime sleepiness after 6 months.
The estimated primary completion date is June 2020, and the estimated study completion date is July 2020. About 40 patients are expected to be recruited.
Find more information on the study page at Clinicaltrials.gov.
The Magnetic Resonance Imaging in Obstructive Sleep Apnea trial is an observational cohort study recruiting adults with obstructive sleep apnea undergoing surgery.
The trial will compare drug-induced sleep endoscopy and upper airway MRI in order to determine which is the better predictor of success in patients who cannot tolerate nonsurgical solutions. Upper airway MRI is a more complete evaluation during wakefulness and is cheaper than drug-induced sleep endoscopy, but no studies have thus far utilized MRI as a surgical evaluation tool.
Patients will be included if they are at least 21 years old, have moderate to severe obstructive sleep apnea, and have a body mass index less than 40 kg/m2. Exclusion criteria include prior surgery for obstructive sleep apnea; known neurologic, cardiac, pulmonary, renal, or hepatic disorders; psychiatric problems except for treated depression or mild anxiety; a coexisting sleep disorder; or another contraindication to drug-induced sleep endoscopy or MRI, such as propofol allergy.
The primary outcome measure is surgical results after 6 months, which will be measured using sleep studies. Secondary outcomes include sleep-related quality of life after 6 months and daytime sleepiness after 6 months.
The estimated primary completion date is June 2020, and the estimated study completion date is July 2020. About 40 patients are expected to be recruited.
Find more information on the study page at Clinicaltrials.gov.
Narcolepsy: Diagnosis and management
Narcolepsy was originally described in the late 1800s by the French physician Jean-Baptiste-Edouard Gélineau, who reported the case of a wine merchant suffering from somnolence. In this first description, he coined the term narcolepsie by joining the Greek words narke (numbness or stupor) and lepsis (attack).1
Since then, the disorder has been further characterized, and some insight into its biological underpinnings has been established. Importantly, treatments have improved and expanded, facilitating its management and thereby improving quality of life for those with the disorder.
This review focuses on clinically relevant features of the disorder and proposes management strategies.
CLINICAL FEATURES
Narcolepsy is characterized by instability of sleep-wake transitions.
Daytime sleepiness
Clinically, narcolepsy manifests with excessive daytime sleepiness that can be personally and socially disabling. Cataplexy, sleep paralysis, and hypnagogic or hypnopompic hallucinations can also be present,2,3 but they are not necessary for diagnosis. In fact, a minority of patients with narcolepsy have all these symptoms.4 Narcolepsy is divided into type 1 (with cataplexy) and type 2 (without cataplexy).2
Sleepiness tends to be worse with inactivity, and sleep can often be irresistible. Sleep attacks can come on suddenly and may be brief enough to manifest as a lapse in consciousness.
Short naps tend to be refreshing. Rapid eye movement (REM) latency—the interval between falling asleep and the onset of the REM sleep—is short in narcolepsy, and since the REM stage is when dreaming occurs, naps often include dreaming. Therefore, when taking a history, it is worthwhile to ask patients whether they dream during naps; a yes answer supports the diagnosis of narcolepsy.5
In children, sleepiness can manifest in reduced concentration and behavioral issues.6 Napping after age 5 or 6 is considered abnormal and may reflect pathologic sleepiness.7
Cataplexy
Cataplexy—transient muscle weakness triggered by emotion—is a specific feature of narcolepsy type 1. It often begins in the facial muscles and can manifest with slackening of the jaw or brief dropping of the head. However, episodes can be more dramatic and, if the trunk and limb muscles are affected, can result in collapsing to the ground.
Cataplexy usually has its onset at about the same time as the sleepiness associated with narcolepsy, but it can arise even years later.8 Episodes can last from a few seconds to 2 minutes. Consciousness is always preserved. A range of emotions can trigger cataplexy, but typically the emotion is a positive one such as laughter or excitement.9 Deep tendon reflexes disappear in cataplexy, so checking reflexes during a witnessed episode can be clinically valuable.2
Cataplexy can worsen with stress and insufficient sleep, occasionally with “status cataplecticus,” in which repeated, persistent episodes of cataplexy occur over several hours.8 Status cataplecticus can be spontaneous or an effect of withdrawal from anticataplectic medications.2
Cataplexy is thought to represent intrusion of REM sleep and its associated muscle atonia during wakefulness.
Sleep paralysis, hallucinations
Sleep paralysis and hallucinations are other features of narcolepsy that reflect this REM dissociation from sleep.
Sleep paralysis occurs most commonly upon awakening, but sometimes just before sleep onset. In most cases, it is manifested by inability to move the limbs or speak, lasting several seconds or, in rare cases, minutes at a time. Sleep paralysis can be associated with a sensation of fear or suffocation, especially when initially experienced.8
Hypnopompic hallucinations, occurring upon awakening, are more common than hypnagogic hallucinations, which are experienced before falling asleep. The hallucinations are often vivid and usually visual, although other types of hallucinations are possible. Unlike those that occur in psychotic disorders, the hallucinations tend to be associated with preserved insight that they are not real.10
Notably, both sleep paralysis and hallucinations are nonspecific symptoms that are common in the general population.8,11,12
Fragmented sleep
Although they are very sleepy, people with narcolepsy generally cannot stay asleep for very long. Their sleep tends to be extremely fragmented, and they often wake up several times a night.2
This sleep pattern reflects the inherent instability of sleep-wake transitions in narcolepsy. In fact, over a 24-hour period, adults with narcolepsy have a normal amount of sleep.13 In children, however, when narcolepsy first arises, the 24-hour sleep time can increase abruptly and can sometimes be associated with persistent cataplexy that can manifest as a clumsy gait.14
Weight gain, obstructive sleep apnea
Weight gain is common, particularly after symptom onset, and especially in children. As a result, obesity is a frequent comorbidity.15 Because obstructive sleep apnea can consequently develop, all patients with narcolepsy require screening for sleep-disordered breathing.
Other sleep disorders often accompany narcolepsy and are more common than in the general population.16 In a study incorporating both clinical and polysomnographic data of 100 patients with narcolepsy, insomnia was the most common comorbid sleep disorder, with a prevalence of 28%; others were REM sleep behavior disorder (24%), restless legs syndrome (24%), obstructive sleep apnea (21%), and non-REM parasomnias.17
PSYCHOSOCIAL CONSEQUENCES
Narcolepsy has significant psychosocial consequences. As a result of their symptoms, people with narcolepsy may not be able to meet academic or work-related demands.
Additionally, their risk of a motor vehicle accident is 3 to 4 times higher than in the general population, and more than one-third of patients have been in an accident due to sleepiness.18 There is some evidence to show that treatment eliminates this risk.19
Few systematic studies have examined mood disorders in narcolepsy. However, studies tend to show a higher prevalence of psychiatric disorders than in the general population, with depression and anxiety the most com-mon.20,21
DIAGNOSIS IS OFTEN DELAYED
The prevalence of narcolepsy type 1 is between 25 and 100 per 100,000 people.22 In a Mayo Clinic study,23 the incidence of narcolepsy type 1 was estimated to be 0.74 per 100,000 person-years. Epidemiologic data on narcolepsy type 2 are sparse, but patients with narcolepsy without cataplexy are thought to represent only 36% of all narcolepsy patients.23
Diagnosis is often delayed, with the average time between the onset of symptoms and the diagnosis ranging from 8 to 22 years. With increasing awareness, the efficiency of the diagnostic process is improving, and this delay is expected to lessen accordingly.24
Symptoms most commonly arise in the second decade; but the age at onset ranges significantly, between the first and fifth decades. Narcolepsy has a bimodal distribution in incidence, with the biggest peak at approximately age 15 and second smaller peak in the mid-30s. Some studies have suggested a slight male predominance.23,25
DIAGNOSIS
History is key
The history should include specific questions about the hallmark features of narcolepsy, including cataplexy, sleep paralysis, and sleep-related hallucinations. For individual assessment of subjective sleepiness, the Epworth Sleepiness Scale or Pediatric Daytime Sleepiness Scale can be administered quickly in the office setting.26,27
The Epworth score is calculated from the self-rated likelihood of falling asleep in 8 different situations, with possible scores of 0 (would never doze) to 3 (high chance of dozing) on each question, for a total possible score of 0 to 24. Normal total scores are between 0 and 10, while scores greater than 10 reflect pathologic sleepiness. Scores on the Epworth Sleepiness Scale in those with narcolepsy tend to reflect moderate to severe sleepiness, or at least 13, as opposed to patients with obstructive sleep apnea, whose scores commonly reflect milder sleepiness.28
Testing with actigraphy and polysomnography
It is imperative to rule out insufficient sleep and other sleep disorders as a cause of daytime sleepiness. This can be done with a careful clinical history, actigraphy with sleep logs, and polysomnography.
In the 2 to 4 weeks before actigraphy and subsequent testing, all medications with alerting or sedating properties (including antidepressants) should be tapered off to prevent influence on the results of the study.
Delayed sleep-phase disorder presents at a similar age as narcolepsy and can be associated with similar degrees of sleepiness. However, individuals with delayed sleep phase disorder have an inappropriately timed sleep-wake cycle so that there is a shift in their desired sleep onset and awakening times. It is common—prevalence estimates vary but average about 1% in the general population.29
Insufficient sleep syndrome is even more common, especially in teenagers and young adults, with increasing family, social, and academic demands. Sleep needs vary across the life span. A teenager needs 8 to 10 hours of sleep per night, and a young adult needs 7 to 9 hours. A study of 1,285 high school students found that 10.4% were not getting enough sleep.30
If actigraphy data suggest a circadian rhythm disorder or insufficient sleep that could explain the symptoms of sleepiness, then further testing should be halted and these specific issues should be addressed. In these cases, working with the patient toward maintaining a regular sleep-wake schedule with 7 to 8 hours of nightly sleep will often resolve symptoms.
If actigraphy demonstrates the patient is maintaining a regular sleep schedule and allowing adequate time for nightly sleep, the next step is polysomnography.
Polysomnography is performed to detect other disorders that can disrupt sleep, such as sleep-disordered breathing or periodic limb movement disorder.2,5 In addition, polysomnography can provide assurance that adequate sleep was obtained prior to the next step in testing.
Multiple sleep latency test
If sufficient sleep is obtained on polysomnograpy (at least 6 hours for an adult) and no other sleep disorder is identified, a multiple sleep latency test is performed. A urine toxicology screen is typically performed on the day of the test to ensure that drugs are not affecting the results.
The multiple sleep latency test consists of 4 to 5 nap opportunities at 2-hour intervals in a quiet dark room conducive to sleep, during which both sleep and REM latency are recorded. The sleep latency of those with narcolepsy is significantly shortened, and the diagnosis of narcolepsy requires an average sleep latency of less than 8 minutes.
Given the propensity for REM sleep in narcolepsy, another essential feature for diagnosis is the sleep-onset REM period (SOREMP). A SOREMP is defined as a REM latency of less than 15 minutes. A diagnosis of narcolepsy re-quires a SOREMP in at least 2 of the naps in a multiple sleep latency test (or 1 nap if the shortened REM latency is seen during polysomnography).31
The multiple sleep latency test has an imperfect sensitivity, though, and should be repeated when there is a high suspicion of narcolepsy.32–34 It is not completely specific either, and false-positive results occur. In fact, SOREMPs can be seen in the general population, particularly in those with a circadian rhythm disorder, insufficient sleep, or sleep-disordered breathing. Two or more SOREMPs in an multiple sleep latency test can be seen in a small proportion of the general population.35 The results of a multiple sleep latency test should be interpreted in the clinical context.
Differential diagnosis
Narcolepsy type 1 is distinguished from type 2 by the presence of cataplexy. A cerebrospinal fluid hypocretin 1 level of 110 pg/mL or less, or less than one-third of the mean value obtained in normal individuals, can substitute for the multiple sleep latency test in diagnosing narcolepsy type 1.31 Currently, hypocretin testing is generally not performed in clinical practice, although it may become a routine part of the narcolepsy evaluation in the future.
Thus, according to the International Classification of Sleep Disorders, 3rd edition,31 the diagnosis of narcolepsy type 1 requires excessive daytime sleepiness for at least 3 months that cannot be explained by another sleep disorder, medical or neurologic disorder, mental disorder, medication use, or substance use disorder, and at least 1 of the following:
- Cataplexy and mean sleep latency of 8 minutes or less with at least 2 SOREMPs on multiple sleep latency testing (1 of which can be on the preceding night’s polysomography)
- Cerebrospinal fluid hypocretin 1 levels less than 110 pg/mL or one-third the baseline normal levels and mean sleep latency ≤ 8 minutes with ≥ 2 SOREMPs on multiple sleep latency testing.
Similarly, the diagnosis of narcolepsy type 2 requires excessive daytime sleepiness for at least 3 months that cannot be explained by another sleep disorder, medical or neurological disorder, mental disorder, medication use, or substance use disorder, plus:
- Mean sleep latency of 8 minutes or less with at least 2 SOREMPs on multiple sleep latency testing.
Idiopathic hypersomnia, another disorder of central hypersomnolence, is also characterized by disabling sleepiness. It is diagnostically differentiated from narcolepsy, as there are fewer than 2 SOREMPs. As opposed to narcolepsy, in which naps tend to be refreshing, even prolonged naps in idiopathic hypersomnia are often not helpful in restoring wakefulness. In idiopathic hypersomnia, sleep is usually not fragmented, and there are few nocturnal arousals. Sleep times can often be prolonged as well, whereas in narcolepsy total sleep time through the day may not be increased but is not consolidated.
Kleine-Levin syndrome is a rarer disorder of hypersomnia. It is episodic compared with the relatively persistent sleepiness in narcolepsy and idiopathic hypersomnia. Periods of hypersomnia occur intermittently for days to weeks and are accompanied by cognitive and behavioral changes including hyperphagia and hypersexuality.4
LINKED TO HYPOCRETIN DEFICIENCY
Over the past 2 decades, the underlying pathophysiology of narcolepsy type 1 has been better characterized.
Narcolepsy type 1 has been linked to a deficiency in hypocretin in the central nervous system.36 Hypocretin (also known as orexin) is a hormone produced in the hypothalamus that acts on multiple brain regions and maintains alertness. For unclear reasons, hypothalamic neurons producing hypocretin are selectively reduced in narcolepsy type 1. Hypocretin also stabilizes wakefulness and inhibits REM sleep; therefore, hypocretin deficiency can lead to inappropriate intrusions of REM sleep onto wakefulness, leading to the hallmark features of narcolepsy—cataplexy, sleep-related hallucinations, and sleep paralysis.37 According to one theory, cataplexy is triggered by emotional stimuli because of a pathway between the medial prefrontal cortex and the amygdala to the pons.38
Cerebrospinal fluid levels of hypocretin in patients with narcolepsy type 2 tend to be normal, and the biologic underpinnings of narcolepsy type 2 remain mysterious. However, in the subgroup of those with narcolepsy type 2 in which hypocretin is low, many individuals go on to develop cataplexy, thereby evolving to narcolepsy type 1.36
POSSIBLE AUTOIMMUNE BASIS
Narcolepsy is typically a sporadic disorder, although familial cases have been described. The risk of a parent with narcolepsy having a child who is affected is approximately 1%.5
Narcolepsy type 1 is strongly associated with HLA-DQB1*0602, with up to 95% of those affected having at least one allele.39 Having 2 copies of the allele further increases the risk of developing narcolepsy.40 However, this allele is far from specific for narcolepsy with cataplexy, as it occurs in 12% to 38% of the general population.41 Therefore, HLA typing currently has limited clinical utility. The exact cause is as yet unknown, but substantial literature proposes an autoimmune basis of the disorder, given the strong association with the HLA subtype.42–44
After the 2009 H1N1 influenza pandemic, there was a significant increase in the incidence of narcolepsy with cataplexy, which again sparked interest in an autoimmune etiology underlying the disorder. Pandemrix, an H1N1 vaccine produced as a result of the 2009 pandemic, appeared to also be associated with an increase in the incidence of narcolepsy. An association with other upper respiratory infections has also been noted, further supporting a possible autoimmune basis.
A few studies have looked for serum autoantibodies involved in the pathogenesis of narcolepsy. Thus far, only one has been identified, an antibody to Tribbles homolog 2, found in 20% to 40% of those with new onset of nar-colepsy.42–44
TREATMENTS FOR DAYTIME SLEEPINESS
As with many chronic disorders, the treatment of narcolepsy consists of symptomatic rather than curative management, which can be done through both pharmacologic and nonpharmacologic means.
Nondrug measures
Scheduled naps lasting 15 to 20 minutes can help improve alertness.45 A consistent sleep schedule with good sleep hygiene, ensuring sufficient nightly sleep, is also important. In one study, the combination of scheduled naps and regular nocturnal sleep times reduced the level of daytime sleepiness and unintentional daytime sleep. Daytime naps were most helpful for those with the highest degree of daytime sleepiness.45
Strategic use of caffeine can be helpful and can reduce dependence on pharmacologic treatment.
Screening should be performed routinely for other sleep disorders, such as sleep-disordered breathing, which should be treated if identified.5,18 When being treated for other medical conditions, individuals with narcolepsy should avoid medications that can cause sedation, such as opiates or barbiturates; alcohol should be minimized or avoided.
Networking with other individuals with narcolepsy through support groups such as Narcolepsy Network can be valuable for learning coping skills and connecting with community resources. Psychological counseling for the patient, and sometimes the family, can also be useful. School-age children may need special accommodations such as schedule adjustments to allow for scheduled naps or frequent breaks to maintain alertness.
People with narcolepsy tend to function better in careers that do not require long periods of sitting, as sleepiness tends to be worse, but instead offer flexibility and require higher levels of activity that tend to combat sleepiness. They should not work as commercial drivers.18
Medications
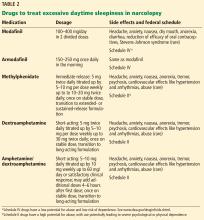
While behavioral interventions in narcolepsy are vital, they are rarely sufficient, and drugs that promote daytime wakefulness are used as an adjunct (Table 2).46
Realistic expectations should be established when starting, as some degree of residual sleepiness usually remains even with optimal medical therapy. Medications should be strategically scheduled to maximize alertness during necessary times such as at work or school or during driving. Patients should specifically be counseled to avoid driving if sleepy.18,47
Modafinil is often used as a first-line therapy, given its favorable side-effect profile and low potential for abuse. Its pharmacologic action has been debated but it probably acts as a selective dopamine reuptake inhibitor. It is typically taken twice daily (upon waking and early afternoon) and is usually well tolerated.
Potential side effects include headache, nausea, dry mouth, anorexia, diarrhea, and, rarely, Stevens-Johnson syndrome. Cardiovascular side effects are minimal, making it a favorable choice in older patients.18,48
A trial in 283 patients showed significantly lower levels of sleepiness in patients taking modafinil 200 mg or 400 mg than in a control group. Other trials have supported these findings and showed improved driving performance on modafinil.18
Notably, modafinil can increase the metabolism of oral contraceptives, thereby reducing their efficacy. Women of childbearing age should be warned about this interaction and should be transitioned to nonhormonal forms of contraception.2,47
Armodafinil, a purified R-isomer of modafinil, has a longer half-life and requires only once-daily dosing.5
If modafinil or armodafinil fails to optimally manage daytime sleepiness, a traditional stimulant such as methylphenidate or an amphetamine is often used.
Methylphenidate and amphetamines primarily inhibit the reuptake and increase the release of the monoamines, mainly dopamine, and to a lesser degree serotonin and norepinephrine.
These drugs have more significant adverse effects that can involve the cardiovascular system, causing hypertension and arrhythmias. Anorexia, weight loss, and, particularly with high doses, psychosis can occur.49
These drugs should be avoided in patients with a history of significant cardiovascular disease. Before starting stimulant therapy, a thorough cardiovascular examination should be done, often including electrocardiography to ensure there is no baseline arrhythmia.
Patients on these medications should be followed closely to ensure that blood pressure, pulse, and weight are not negatively affected.18,50 Addiction and tolerance can develop with these drugs, and follow-up should include assessment for dependence. Some states may require prescription drug monitoring to ensure the drugs are not being abused or diverted.
Short- and long-acting formulations of both methylphenidate and amphetamines are available, and a long-acting form is often used in conjunction with a short-acting form as needed.18
Addiction and drug-seeking behavior can develop but are unusual in those taking stimulants to treat narcolepsy.49
Follow-up
Residual daytime sleepiness can be measured subjectively through the Epworth Sleepiness Scale on follow-up. If necessary, a maintenance-of-wakefulness test can provide an objective assessment of treatment efficacy.18
As narcolepsy is a chronic disorder, treatment should evolve with time. Most medications that treat narcolepsy are categorized by the US Food and Drug Administration as pregnancy category C, as we do not have adequate studies in human pregnancies to evaluate their effects. When a patient with narcolepsy becomes pregnant, she should be counseled about the risks and benefits of remaining on therapy. Treatment should balance the risks of sleepiness with the potential risks of remaining on medications.50 In the elderly, as cardiovascular comorbidities tend to increase, the risks and benefits of therapy should be routinely reevaluated.
For cataplexy
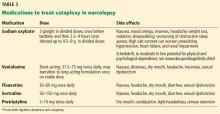
Sodium oxybate,51–53 the most potent anticataplectic drug, is the sodium salt of gamma hydroxybutyrate, a metabolite of gamma-aminobutyric acid. Sodium oxybate can be prescribed in the United States, Canada, and Europe. The American Academy of Sleep Medicine recommends sodium oxybate for cataplexy, daytime sleepiness, and disrupted sleep based on 3 level-1 studies and 2 level-4 studies.46
Sodium oxybate increases slow-wave sleep, improves sleep continuity, and often helps to mitigate daytime sleepiness. Due to its short half-life, its administration is unusual: the first dose is taken before bedtime and the second dose 2.5 to 4 hours later. Some patients set an alarm clock to take the second dose, while others awaken spontaneously to take the second dose. Most patients find that with adherence to dosing and safety instructions, sodium oxybate can serve as a highly effective form of treatment of both excessive sleepiness and cataplexy and may reduce the need for stimulant-based therapies.
The most common adverse effects are nausea, mood swings, and enuresis. Occasionally, psychosis can result and limit use of the drug. Obstructive sleep apnea can also develop or worsen.52 Because of its high salt content, sodium oxybate should be used with caution in those with heart failure, hypertension, or renal impairment. Its relative, gamma hydroxybutyrate, causes rapid sedation and has been notorious for illegal use as a date rape drug.
In the United States, sodium oxybate is distributed only through a central pharmacy to mitigate potential abuse. Due to this system, the rates of diversion are extremely low, estimated in a postmarketing analysis to be 1 instance per 5,200 patients treated. In the same study, abuse and dependence were both rare as well, about 1 case for every 2,600 and 6,500 patients treated.6,18,52,53
Antidepressants promote the action of norepinephrine and, to a lesser degree, serotonin, thereby suppressing REM sleep.
Venlafaxine, a serotonin-norepinephrine reuptake inhibitor, is often used as a first-line treatment for cataplexy. Selective serotonin reuptake inhibitors such as fluoxetine are also used with success. Tricyclic antidepressants such as protriptyline or clomipramine are extremely effective for cataplexy, but are rarely used due to their adverse effects.2,47
FUTURE WORK
While our understanding of narcolepsy has advanced, there are still gaps in our knowledge of the disorder—namely, the specific trigger for the loss of hypocretin neurons in type 1 narcolepsy and the underlying pathophysiology of type 2.
A number of emerging therapies target the hypocretin system, including peptide replacement, neuronal transplant, and immunotherapy preventing hypocretin neuronal cell death.50,54,55 Additional drugs designed to improve alertness that do not involve the hypocretin system are also being developed, including a histamine inverse agonist.50,56 Sodium oxybate and modafinil, although currently approved for use in adults, are still off-label in pediatric practice. Studies of the safety and efficacy of these medications in children are needed.7,57
- Gélineau J. De la narcolepsie. Gazette des Hôpitaux Civils et Militaires 1880; part a, 53:626–628, part b, 54:635–637.
- Dauvilliers Y, Arnulf I, Mignot E. Narcolepsy with cataplexy. Lancet 2007; 369(9560):499–511. doi:10.1016/S0140-6736(07)60237-2
- Scammell TE. Clinical features and diagnosis of narcolepsy in adults. In: Eichler AF, ed. UpToDate. Waltham, MA: UpToDate; 2018. www.uptodate.com. Accessed October 31, 2018.
- Morrish E, King MA, Smith IE, Shneerson JM. Factors associated with a delay in the diagnosis of narcolepsy. Sleep Med 2004; 5(1):37–41. pmid:14725825
- Scammell TE. Narcolepsy. N Engl J Med 2015; 373(27):2654–2662. doi:10.1056/NEJMra1500587
- Babiker MO, Prasad M. Narcolepsy in children: a diagnostic and management approach. Pediatr Neurol 2015; 52(6):557–565. doi:10.1016/j.pediatrneurol.2015.02.020
- Kotagal S. Narcolepsy in children. In: UpToDate, Eichler AF, ed. UpToDate, Waltham, MA. www.uptodate.com. Accessed October 31, 2018.
- Scammell TE. The neurobiology, diagnosis, and treatment of narcolepsy. Ann Neurol 2003; 53(2):154–166. doi:10.1002/ana.10444
- Overeem S, van Nues SJ, van der Zande WL, Donjacour CE, van Mierlo P, Lammers GJ. The clinical features of cataplexy: a questionnaire study in narcolepsy patients with and without hypocretin-1 deficiency. Sleep Med 2011; 12(1):12–18. doi:10.1016/j.sleep.2010.05.010
- Plazzi G, Fabbri C, Pizza F, Serretti A. Schizophrenia-like symptoms in narcolepsy type 1: shared and distinctive clinical characteristics. Neuropsychobiology 2015; 71(4):218–224. doi:10.1159/000432400
- Ohayon MM. Prevalence of hallucinations and their pathological associations in the general population. Psychiatry Res 2000; 97(2-3):153–164. pmid:11166087
- Sharpless BA, Barber JP. Lifetime prevalence rates of sleep paralysis: a systematic review. Sleep Med Rev 2011;5(5):311–315. doi:10.1016/j.smrv.2011.01.007
- Broughton R, Dunham W, Newman J, Lutley K, Duschesne P, Rivers M. Ambulatory 24 hour sleep-wake monitoring in narcolepsy-cataplexy compared to matched controls. Electroencephalogr Clin Neurophysiol 1988; 70(6):473–481. pmid:2461281
- Pizza F, Franceschini C, Peltola H, et al. Clinical and polysomnographic course of childhood narcolepsy with cataplexy. Brain 2013; 136(pt 12):3787–3795. doi:10.1093/brain/awt277
- Kotagal S, Krahn LE, Slocumb N. A putative link between childhood narcolepsy and obesity. Sleep Med 2004; 5(2):147–150. doi:10.1016/j.sleep.2003.10.006
- Pizza F, Tartarotti S, Poryazova R, Baumann CR, Bassetti CL. Sleep-disordered breathing and periodic limb movements in narcolepsy with cataplexy: a systematic analysis of 35 consecutive patients. Eur Neurol 2013; 70(1-2):22–26. doi:10.1159/000348719
- Frauscher B, Ehrmann L, Mitterling T, et al. Delayed diagnosis, range of severity, and multiple sleep comorbidities: a clinical and polysomnographic analysis of 100 patients of the Innsbruck narcolepsy cohort. J Clin Sleep Med 2013; 9(8):805–812. doi:10.5664/jcsm.2926
- Scammell TE. Treatment of narcolepsy in adults. In: Eichler AF, ed. UpToDate, Waltham, MA. www.uptodate.com. Accessed October 31, 2018.
- Pizza F, Jaussent I, Lopez R, et al. Car crashes and central disorders of hypersomnolence: a French study. PLoS One 2015; 10(6):e0129386. doi:10.1371/journal.pone.0129386
- Fortuyn HD, Lappenschaar MA, Furer JW, et al. Anxiety and mood disorders in narcolepsy: a case-control study. Gen Hosp Psychiatry 2010; 32(1):49–56. doi:10.1016/j.genhosppsych.2009.08.007
- Ruoff CM, Reaven NL, Funk SE, et al. High rates of psychiatric comorbidity in narcolepsy: findings from the Burden of Narcolepsy Disease (BOND) study of 9,312 patients in the United States. J Clin Psychiatry 2017; 78(2):171–176. doi:10.4088/JCP.15m10262
- Longstreth WT Jr, Koepsell TD, Ton TG, Hendrickson AF, van Belle G. The epidemiology of narcolepsy. Sleep. 2007; 30(1):13–26. pmid:17310860
- Silber MH, Krahn LE, Olson EJ, Pankratz VS. The epidemiology of narcolepsy in Olmsted County, Minnesota: a population-based study. Sleep 2002; 25(2):197–202. pmid:11902429
- Thorpy MJ, Krieger AC. Delayed diagnosis of narcolepsy: characterization and impact. Sleep Med 2014; 15(5):502–507. doi:10.1016/j.sleep.2014.01.015
- Dauvilliers Y, Montplaisir J, Molinari N, et al. Age at onset of narcolepsy in two large populations of patients in France and Quebec. Neurology 2001; 57(11):2029–2033. pmid:11739821
- Johns MW. A new method for measuring daytime sleepiness: the Epworth sleepiness scale. Sleep 1991; 14(6):540–545. pmid:1798888
- Drake C, Nickel C, Burduvali E, Roth T, Jefferson C, Badia P. The pediatric daytime sleepiness scale (PDSS): sleep habits and school outcomes in middle-school children. Sleep 2003; 26(4):455–458. pmid:12841372
- van der Heide A, van Schie MK, Lammers GJ, et al. Comparing treatment effect measurements in narcolepsy: the sustained attention to response task, Epworth sleepiness scale and maintenance of wakefulness test. Sleep 2015; 38(7):1051–1058. doi:10.5665/sleep.4810
- Nesbitt AD. Delayed sleep-wake phase disorder. J Thorac Dis 2018; 10(suppl 1):S103–S111. doi:10.21037/jtd.2018.01.11
- Pallesen S, Saxvig IW, Molde H, Sørensen E, Wilhelmsen-Langeland A, Bjorvatn B. Brief report: behaviorally induced insufficient sleep syndrome in older adolescents: prevalence and correlates. J Adolesc 2011; 34(2):391–395. doi:10.1016/j.adolescence.2010.02.005
- American Academy of Sleep Medicine. International Classification of Sleep Disorders. 3rd ed. Darien, IL: American Academy of Sleep Disorders; 2014.
- Trotti LM, Staab BA, Rye DB. Test-retest reliability of the multiple sleep latency test in narcolepsy without cataplexy and idiopathic hypersomnia. J Clin Sleep Med 2013; 9(8):789–795. doi:10.5664/jcsm.2922
- Andlauer O, Moore H, Jouhier L, et al. Nocturnal rapid eye movement sleep latency for identifying patients with narcolepsy/hypocretin deficiency. JAMA Neurol 2013; 70(7):891–902. doi:10.1001/jamaneurol.2013.1589
- Cairns A, Bogan R. Prevalence and clinical correlates of a short onset REM period (SOREMP) during routine PSG. Sleep 2015; 38(10):1575–1581. doi:10.5665/sleep.5050
- Mignot E, Lin L, Finn L, et al. Correlates of sleep-onset REM periods during the multiple sleep latency test in community adults. Brain 2006; 129(6):1609–1623. doi:10.1093/brain/awl079
- Nishino S, Ripley B, Overeem S, Lammers GJ, Mignot E. Hypocretin (orexin) deficiency in human narcolepsy. Lancet 2000; 355(9197):39–40. doi:10.1016/S0140-6736(99)05582-8
- Peyron C, Faraco J, Rogers W, et al. A mutation in a case of early onset narcolepsy and a generalized absence of hypocretin peptides in human narcoleptic brains. Nat Med 2000; 6(9):991–997. doi:10.1038/79690
- Oishi Y, Williams RH, Agostinelli L, et al. Role of the medial prefrontal cortex in cataplexy. J Neurosci 2013; 33(23):9743–9751. doi:10.1523/JNEUROSCI.0499-13.2013
- Mignot E, Hayduk R, Black J, Grumet FC, Guilleminault C. HLA DQB1*0602 is associated with cataplexy in 509 narcoleptic patients.. Sleep 1997; 20(11):1012–1020. pmid:9456467
- Pelin Z, Guilleminault C, Risch N, Grumet FC, Mignot E. HLA-DQB1*0602 homozygosity increases relative risk for narcolepsy but not disease severity in two ethnic groups. US Modafinil in Narcolepsy Multicenter Study Group. Tissue Antigens 1998; 51(1):96–100. pmid:9459509
- Akintomide GS, Rickards H. Narcolepsy: a review. Neuropsychiatr Dis Treat 2011; 7(1):507–518. doi:10.2147/NDT.S23624
- Mahlios J, De la Herrán-Arita AK, Mignot E. The autoimmune basis of narcolepsy. Curr Opin Neurobiol 2013; 23(5):767–773. doi:10.1016/j.conb.2013.04.013
- Degn M, Kornum BR. Type 1 narcolepsy: a CD8(+) T cell-mediated disease? Ann N Y Acad Sci 2015;1 351:80–88. doi:10.1111/nyas.12793
- Liblau RS, Vassalli A, Seifinejad A, Tafti M. Hypocretin (orexin) biology and the pathophysiology of narcolepsy with cataplexy. Lancet Neurol 2015; 14(3):318–328. doi:10.1016/S1474-4422(14)70218-2
- Rogers AE, Aldrich MS, Lin X. A comparison of three different sleep schedules for reducing daytime sleepiness in narcolepsy. Sleep 2001; 24(4):385–391. pmid:11403522
- Morgenthaler TI, Kapur VK, Brown TM, et al; Standards of Practice Committee of the American Academy of Sleep Medicine. Practice parameters for the treatment of narcolepsy and other hypersomnias of central origin. Sleep 2007; 30(12):1705–1711. pmid:18246980
- Mignot EJ. A practical guide to the therapy of narcolepsy and hypersomnia syndromes. Neurotherapeutics 2012; 9(4):739–752. doi:10.1007/s13311-012-0150-9
- Roth T, Schwartz JR, Hirshkowitz M, Erman MK, Dayno JM, Arora S. Evaluation of the safety of modafinil for treatment of excessive sleepiness. J Clin Sleep Med 2007; 3(6):595–602. pmid:17993041
- Auger RR, Goodman SH, Silber MH, Krahn LE, Pankratz VS, Slocumb NL. Risks of high-dose stimulants in the treatment of disorders of excessive somnolence: a case-control study. Sleep 2005; 28(6):667–672. pmid:16477952
- Abad VC, Guilleminault C. New developments in the management of narcolepsy. Nat Sci Sleep 2017; 9:39–57. doi:10.2147/NSS.S103467
- Drakatos P, Lykouras D, D’Ancona G, et al. Safety and efficacy of long-term use of sodium oxybate for narcolepsy with cataplexy in routine clinical practice. Sleep Med 2017; 35:80–84. doi:10.1016/j.sleep.2017.03.028
- Mansukhani MP, Kotagal S. Sodium oxybate in the treatment of childhood narcolepsy–cataplexy: a retrospective study. Sleep Med 2012; 13(6):606–610. doi:10.1016/j.sleep.2011.10.032
- Wang YG, Swick TJ, Carter LP, Thorpy MJ, Benowitz NL. Safety overview of postmarketing and clinical experience of sodium oxybate (Xyrem): abuse, misuse, dependence, and diversion. J Clin Sleep Med 2009; 5(4):365–371. pmid:19968016
- Weinhold SL, Seeck-Hirschner M, Nowak A, Hallschmid M, Göder R, Baier PC. The effect of intranasal orexin-A (hypocretin-1) on sleep, wakefulness and attention in narcolepsy with cataplexy. Behav Brain Res 2014; 262:8–13. doi:10.1016/j.bbr.2013.12.045
- Arias-Carrión O, Murillo-Rodriguez E. Effects of hypocretin/orexin cell transplantation on narcoleptic-like sleep behavior in rats. PLoS One 2014; 9(4):e95342. doi:10.1371/journal.pone.0095342
- Leu-Semenescu S, Nittur N, Golmard JL, Arnulf I. Effects of pitolisant, a histamine H3 inverse agonist, in drug-resistant idiopathic and symptomatic hypersomnia: a chart review. Sleep Med 2014; 15(6):681–687. doi:10.1016/j.sleep.2014.01.021
- Lecendreux M, Bruni O, Franco P, et al. Clinical experience suggests that modafinil is an effective and safe treatment for paediatric narcolepsy. J Sleep Res 2012; 21(4):481–483. doi:10.1111/j.1365-2869.2011.00991.x
Narcolepsy was originally described in the late 1800s by the French physician Jean-Baptiste-Edouard Gélineau, who reported the case of a wine merchant suffering from somnolence. In this first description, he coined the term narcolepsie by joining the Greek words narke (numbness or stupor) and lepsis (attack).1
Since then, the disorder has been further characterized, and some insight into its biological underpinnings has been established. Importantly, treatments have improved and expanded, facilitating its management and thereby improving quality of life for those with the disorder.
This review focuses on clinically relevant features of the disorder and proposes management strategies.
CLINICAL FEATURES
Narcolepsy is characterized by instability of sleep-wake transitions.
Daytime sleepiness
Clinically, narcolepsy manifests with excessive daytime sleepiness that can be personally and socially disabling. Cataplexy, sleep paralysis, and hypnagogic or hypnopompic hallucinations can also be present,2,3 but they are not necessary for diagnosis. In fact, a minority of patients with narcolepsy have all these symptoms.4 Narcolepsy is divided into type 1 (with cataplexy) and type 2 (without cataplexy).2
Sleepiness tends to be worse with inactivity, and sleep can often be irresistible. Sleep attacks can come on suddenly and may be brief enough to manifest as a lapse in consciousness.
Short naps tend to be refreshing. Rapid eye movement (REM) latency—the interval between falling asleep and the onset of the REM sleep—is short in narcolepsy, and since the REM stage is when dreaming occurs, naps often include dreaming. Therefore, when taking a history, it is worthwhile to ask patients whether they dream during naps; a yes answer supports the diagnosis of narcolepsy.5
In children, sleepiness can manifest in reduced concentration and behavioral issues.6 Napping after age 5 or 6 is considered abnormal and may reflect pathologic sleepiness.7
Cataplexy
Cataplexy—transient muscle weakness triggered by emotion—is a specific feature of narcolepsy type 1. It often begins in the facial muscles and can manifest with slackening of the jaw or brief dropping of the head. However, episodes can be more dramatic and, if the trunk and limb muscles are affected, can result in collapsing to the ground.
Cataplexy usually has its onset at about the same time as the sleepiness associated with narcolepsy, but it can arise even years later.8 Episodes can last from a few seconds to 2 minutes. Consciousness is always preserved. A range of emotions can trigger cataplexy, but typically the emotion is a positive one such as laughter or excitement.9 Deep tendon reflexes disappear in cataplexy, so checking reflexes during a witnessed episode can be clinically valuable.2
Cataplexy can worsen with stress and insufficient sleep, occasionally with “status cataplecticus,” in which repeated, persistent episodes of cataplexy occur over several hours.8 Status cataplecticus can be spontaneous or an effect of withdrawal from anticataplectic medications.2
Cataplexy is thought to represent intrusion of REM sleep and its associated muscle atonia during wakefulness.
Sleep paralysis, hallucinations
Sleep paralysis and hallucinations are other features of narcolepsy that reflect this REM dissociation from sleep.
Sleep paralysis occurs most commonly upon awakening, but sometimes just before sleep onset. In most cases, it is manifested by inability to move the limbs or speak, lasting several seconds or, in rare cases, minutes at a time. Sleep paralysis can be associated with a sensation of fear or suffocation, especially when initially experienced.8
Hypnopompic hallucinations, occurring upon awakening, are more common than hypnagogic hallucinations, which are experienced before falling asleep. The hallucinations are often vivid and usually visual, although other types of hallucinations are possible. Unlike those that occur in psychotic disorders, the hallucinations tend to be associated with preserved insight that they are not real.10
Notably, both sleep paralysis and hallucinations are nonspecific symptoms that are common in the general population.8,11,12
Fragmented sleep
Although they are very sleepy, people with narcolepsy generally cannot stay asleep for very long. Their sleep tends to be extremely fragmented, and they often wake up several times a night.2
This sleep pattern reflects the inherent instability of sleep-wake transitions in narcolepsy. In fact, over a 24-hour period, adults with narcolepsy have a normal amount of sleep.13 In children, however, when narcolepsy first arises, the 24-hour sleep time can increase abruptly and can sometimes be associated with persistent cataplexy that can manifest as a clumsy gait.14
Weight gain, obstructive sleep apnea
Weight gain is common, particularly after symptom onset, and especially in children. As a result, obesity is a frequent comorbidity.15 Because obstructive sleep apnea can consequently develop, all patients with narcolepsy require screening for sleep-disordered breathing.
Other sleep disorders often accompany narcolepsy and are more common than in the general population.16 In a study incorporating both clinical and polysomnographic data of 100 patients with narcolepsy, insomnia was the most common comorbid sleep disorder, with a prevalence of 28%; others were REM sleep behavior disorder (24%), restless legs syndrome (24%), obstructive sleep apnea (21%), and non-REM parasomnias.17
PSYCHOSOCIAL CONSEQUENCES
Narcolepsy has significant psychosocial consequences. As a result of their symptoms, people with narcolepsy may not be able to meet academic or work-related demands.
Additionally, their risk of a motor vehicle accident is 3 to 4 times higher than in the general population, and more than one-third of patients have been in an accident due to sleepiness.18 There is some evidence to show that treatment eliminates this risk.19
Few systematic studies have examined mood disorders in narcolepsy. However, studies tend to show a higher prevalence of psychiatric disorders than in the general population, with depression and anxiety the most com-mon.20,21
DIAGNOSIS IS OFTEN DELAYED
The prevalence of narcolepsy type 1 is between 25 and 100 per 100,000 people.22 In a Mayo Clinic study,23 the incidence of narcolepsy type 1 was estimated to be 0.74 per 100,000 person-years. Epidemiologic data on narcolepsy type 2 are sparse, but patients with narcolepsy without cataplexy are thought to represent only 36% of all narcolepsy patients.23
Diagnosis is often delayed, with the average time between the onset of symptoms and the diagnosis ranging from 8 to 22 years. With increasing awareness, the efficiency of the diagnostic process is improving, and this delay is expected to lessen accordingly.24
Symptoms most commonly arise in the second decade; but the age at onset ranges significantly, between the first and fifth decades. Narcolepsy has a bimodal distribution in incidence, with the biggest peak at approximately age 15 and second smaller peak in the mid-30s. Some studies have suggested a slight male predominance.23,25
DIAGNOSIS
History is key
The history should include specific questions about the hallmark features of narcolepsy, including cataplexy, sleep paralysis, and sleep-related hallucinations. For individual assessment of subjective sleepiness, the Epworth Sleepiness Scale or Pediatric Daytime Sleepiness Scale can be administered quickly in the office setting.26,27
The Epworth score is calculated from the self-rated likelihood of falling asleep in 8 different situations, with possible scores of 0 (would never doze) to 3 (high chance of dozing) on each question, for a total possible score of 0 to 24. Normal total scores are between 0 and 10, while scores greater than 10 reflect pathologic sleepiness. Scores on the Epworth Sleepiness Scale in those with narcolepsy tend to reflect moderate to severe sleepiness, or at least 13, as opposed to patients with obstructive sleep apnea, whose scores commonly reflect milder sleepiness.28
Testing with actigraphy and polysomnography
It is imperative to rule out insufficient sleep and other sleep disorders as a cause of daytime sleepiness. This can be done with a careful clinical history, actigraphy with sleep logs, and polysomnography.
In the 2 to 4 weeks before actigraphy and subsequent testing, all medications with alerting or sedating properties (including antidepressants) should be tapered off to prevent influence on the results of the study.
Delayed sleep-phase disorder presents at a similar age as narcolepsy and can be associated with similar degrees of sleepiness. However, individuals with delayed sleep phase disorder have an inappropriately timed sleep-wake cycle so that there is a shift in their desired sleep onset and awakening times. It is common—prevalence estimates vary but average about 1% in the general population.29
Insufficient sleep syndrome is even more common, especially in teenagers and young adults, with increasing family, social, and academic demands. Sleep needs vary across the life span. A teenager needs 8 to 10 hours of sleep per night, and a young adult needs 7 to 9 hours. A study of 1,285 high school students found that 10.4% were not getting enough sleep.30
If actigraphy data suggest a circadian rhythm disorder or insufficient sleep that could explain the symptoms of sleepiness, then further testing should be halted and these specific issues should be addressed. In these cases, working with the patient toward maintaining a regular sleep-wake schedule with 7 to 8 hours of nightly sleep will often resolve symptoms.
If actigraphy demonstrates the patient is maintaining a regular sleep schedule and allowing adequate time for nightly sleep, the next step is polysomnography.
Polysomnography is performed to detect other disorders that can disrupt sleep, such as sleep-disordered breathing or periodic limb movement disorder.2,5 In addition, polysomnography can provide assurance that adequate sleep was obtained prior to the next step in testing.
Multiple sleep latency test
If sufficient sleep is obtained on polysomnograpy (at least 6 hours for an adult) and no other sleep disorder is identified, a multiple sleep latency test is performed. A urine toxicology screen is typically performed on the day of the test to ensure that drugs are not affecting the results.
The multiple sleep latency test consists of 4 to 5 nap opportunities at 2-hour intervals in a quiet dark room conducive to sleep, during which both sleep and REM latency are recorded. The sleep latency of those with narcolepsy is significantly shortened, and the diagnosis of narcolepsy requires an average sleep latency of less than 8 minutes.
Given the propensity for REM sleep in narcolepsy, another essential feature for diagnosis is the sleep-onset REM period (SOREMP). A SOREMP is defined as a REM latency of less than 15 minutes. A diagnosis of narcolepsy re-quires a SOREMP in at least 2 of the naps in a multiple sleep latency test (or 1 nap if the shortened REM latency is seen during polysomnography).31
The multiple sleep latency test has an imperfect sensitivity, though, and should be repeated when there is a high suspicion of narcolepsy.32–34 It is not completely specific either, and false-positive results occur. In fact, SOREMPs can be seen in the general population, particularly in those with a circadian rhythm disorder, insufficient sleep, or sleep-disordered breathing. Two or more SOREMPs in an multiple sleep latency test can be seen in a small proportion of the general population.35 The results of a multiple sleep latency test should be interpreted in the clinical context.
Differential diagnosis
Narcolepsy type 1 is distinguished from type 2 by the presence of cataplexy. A cerebrospinal fluid hypocretin 1 level of 110 pg/mL or less, or less than one-third of the mean value obtained in normal individuals, can substitute for the multiple sleep latency test in diagnosing narcolepsy type 1.31 Currently, hypocretin testing is generally not performed in clinical practice, although it may become a routine part of the narcolepsy evaluation in the future.
Thus, according to the International Classification of Sleep Disorders, 3rd edition,31 the diagnosis of narcolepsy type 1 requires excessive daytime sleepiness for at least 3 months that cannot be explained by another sleep disorder, medical or neurologic disorder, mental disorder, medication use, or substance use disorder, and at least 1 of the following:
- Cataplexy and mean sleep latency of 8 minutes or less with at least 2 SOREMPs on multiple sleep latency testing (1 of which can be on the preceding night’s polysomography)
- Cerebrospinal fluid hypocretin 1 levels less than 110 pg/mL or one-third the baseline normal levels and mean sleep latency ≤ 8 minutes with ≥ 2 SOREMPs on multiple sleep latency testing.
Similarly, the diagnosis of narcolepsy type 2 requires excessive daytime sleepiness for at least 3 months that cannot be explained by another sleep disorder, medical or neurological disorder, mental disorder, medication use, or substance use disorder, plus:
- Mean sleep latency of 8 minutes or less with at least 2 SOREMPs on multiple sleep latency testing.
Idiopathic hypersomnia, another disorder of central hypersomnolence, is also characterized by disabling sleepiness. It is diagnostically differentiated from narcolepsy, as there are fewer than 2 SOREMPs. As opposed to narcolepsy, in which naps tend to be refreshing, even prolonged naps in idiopathic hypersomnia are often not helpful in restoring wakefulness. In idiopathic hypersomnia, sleep is usually not fragmented, and there are few nocturnal arousals. Sleep times can often be prolonged as well, whereas in narcolepsy total sleep time through the day may not be increased but is not consolidated.
Kleine-Levin syndrome is a rarer disorder of hypersomnia. It is episodic compared with the relatively persistent sleepiness in narcolepsy and idiopathic hypersomnia. Periods of hypersomnia occur intermittently for days to weeks and are accompanied by cognitive and behavioral changes including hyperphagia and hypersexuality.4
LINKED TO HYPOCRETIN DEFICIENCY
Over the past 2 decades, the underlying pathophysiology of narcolepsy type 1 has been better characterized.
Narcolepsy type 1 has been linked to a deficiency in hypocretin in the central nervous system.36 Hypocretin (also known as orexin) is a hormone produced in the hypothalamus that acts on multiple brain regions and maintains alertness. For unclear reasons, hypothalamic neurons producing hypocretin are selectively reduced in narcolepsy type 1. Hypocretin also stabilizes wakefulness and inhibits REM sleep; therefore, hypocretin deficiency can lead to inappropriate intrusions of REM sleep onto wakefulness, leading to the hallmark features of narcolepsy—cataplexy, sleep-related hallucinations, and sleep paralysis.37 According to one theory, cataplexy is triggered by emotional stimuli because of a pathway between the medial prefrontal cortex and the amygdala to the pons.38
Cerebrospinal fluid levels of hypocretin in patients with narcolepsy type 2 tend to be normal, and the biologic underpinnings of narcolepsy type 2 remain mysterious. However, in the subgroup of those with narcolepsy type 2 in which hypocretin is low, many individuals go on to develop cataplexy, thereby evolving to narcolepsy type 1.36
POSSIBLE AUTOIMMUNE BASIS
Narcolepsy is typically a sporadic disorder, although familial cases have been described. The risk of a parent with narcolepsy having a child who is affected is approximately 1%.5
Narcolepsy type 1 is strongly associated with HLA-DQB1*0602, with up to 95% of those affected having at least one allele.39 Having 2 copies of the allele further increases the risk of developing narcolepsy.40 However, this allele is far from specific for narcolepsy with cataplexy, as it occurs in 12% to 38% of the general population.41 Therefore, HLA typing currently has limited clinical utility. The exact cause is as yet unknown, but substantial literature proposes an autoimmune basis of the disorder, given the strong association with the HLA subtype.42–44
After the 2009 H1N1 influenza pandemic, there was a significant increase in the incidence of narcolepsy with cataplexy, which again sparked interest in an autoimmune etiology underlying the disorder. Pandemrix, an H1N1 vaccine produced as a result of the 2009 pandemic, appeared to also be associated with an increase in the incidence of narcolepsy. An association with other upper respiratory infections has also been noted, further supporting a possible autoimmune basis.
A few studies have looked for serum autoantibodies involved in the pathogenesis of narcolepsy. Thus far, only one has been identified, an antibody to Tribbles homolog 2, found in 20% to 40% of those with new onset of nar-colepsy.42–44
TREATMENTS FOR DAYTIME SLEEPINESS
As with many chronic disorders, the treatment of narcolepsy consists of symptomatic rather than curative management, which can be done through both pharmacologic and nonpharmacologic means.
Nondrug measures
Scheduled naps lasting 15 to 20 minutes can help improve alertness.45 A consistent sleep schedule with good sleep hygiene, ensuring sufficient nightly sleep, is also important. In one study, the combination of scheduled naps and regular nocturnal sleep times reduced the level of daytime sleepiness and unintentional daytime sleep. Daytime naps were most helpful for those with the highest degree of daytime sleepiness.45
Strategic use of caffeine can be helpful and can reduce dependence on pharmacologic treatment.
Screening should be performed routinely for other sleep disorders, such as sleep-disordered breathing, which should be treated if identified.5,18 When being treated for other medical conditions, individuals with narcolepsy should avoid medications that can cause sedation, such as opiates or barbiturates; alcohol should be minimized or avoided.
Networking with other individuals with narcolepsy through support groups such as Narcolepsy Network can be valuable for learning coping skills and connecting with community resources. Psychological counseling for the patient, and sometimes the family, can also be useful. School-age children may need special accommodations such as schedule adjustments to allow for scheduled naps or frequent breaks to maintain alertness.
People with narcolepsy tend to function better in careers that do not require long periods of sitting, as sleepiness tends to be worse, but instead offer flexibility and require higher levels of activity that tend to combat sleepiness. They should not work as commercial drivers.18
Medications
While behavioral interventions in narcolepsy are vital, they are rarely sufficient, and drugs that promote daytime wakefulness are used as an adjunct (Table 2).46
Realistic expectations should be established when starting, as some degree of residual sleepiness usually remains even with optimal medical therapy. Medications should be strategically scheduled to maximize alertness during necessary times such as at work or school or during driving. Patients should specifically be counseled to avoid driving if sleepy.18,47
Modafinil is often used as a first-line therapy, given its favorable side-effect profile and low potential for abuse. Its pharmacologic action has been debated but it probably acts as a selective dopamine reuptake inhibitor. It is typically taken twice daily (upon waking and early afternoon) and is usually well tolerated.
Potential side effects include headache, nausea, dry mouth, anorexia, diarrhea, and, rarely, Stevens-Johnson syndrome. Cardiovascular side effects are minimal, making it a favorable choice in older patients.18,48
A trial in 283 patients showed significantly lower levels of sleepiness in patients taking modafinil 200 mg or 400 mg than in a control group. Other trials have supported these findings and showed improved driving performance on modafinil.18
Notably, modafinil can increase the metabolism of oral contraceptives, thereby reducing their efficacy. Women of childbearing age should be warned about this interaction and should be transitioned to nonhormonal forms of contraception.2,47
Armodafinil, a purified R-isomer of modafinil, has a longer half-life and requires only once-daily dosing.5
If modafinil or armodafinil fails to optimally manage daytime sleepiness, a traditional stimulant such as methylphenidate or an amphetamine is often used.
Methylphenidate and amphetamines primarily inhibit the reuptake and increase the release of the monoamines, mainly dopamine, and to a lesser degree serotonin and norepinephrine.
These drugs have more significant adverse effects that can involve the cardiovascular system, causing hypertension and arrhythmias. Anorexia, weight loss, and, particularly with high doses, psychosis can occur.49
These drugs should be avoided in patients with a history of significant cardiovascular disease. Before starting stimulant therapy, a thorough cardiovascular examination should be done, often including electrocardiography to ensure there is no baseline arrhythmia.
Patients on these medications should be followed closely to ensure that blood pressure, pulse, and weight are not negatively affected.18,50 Addiction and tolerance can develop with these drugs, and follow-up should include assessment for dependence. Some states may require prescription drug monitoring to ensure the drugs are not being abused or diverted.
Short- and long-acting formulations of both methylphenidate and amphetamines are available, and a long-acting form is often used in conjunction with a short-acting form as needed.18
Addiction and drug-seeking behavior can develop but are unusual in those taking stimulants to treat narcolepsy.49
Follow-up
Residual daytime sleepiness can be measured subjectively through the Epworth Sleepiness Scale on follow-up. If necessary, a maintenance-of-wakefulness test can provide an objective assessment of treatment efficacy.18
As narcolepsy is a chronic disorder, treatment should evolve with time. Most medications that treat narcolepsy are categorized by the US Food and Drug Administration as pregnancy category C, as we do not have adequate studies in human pregnancies to evaluate their effects. When a patient with narcolepsy becomes pregnant, she should be counseled about the risks and benefits of remaining on therapy. Treatment should balance the risks of sleepiness with the potential risks of remaining on medications.50 In the elderly, as cardiovascular comorbidities tend to increase, the risks and benefits of therapy should be routinely reevaluated.
For cataplexy
Sodium oxybate,51–53 the most potent anticataplectic drug, is the sodium salt of gamma hydroxybutyrate, a metabolite of gamma-aminobutyric acid. Sodium oxybate can be prescribed in the United States, Canada, and Europe. The American Academy of Sleep Medicine recommends sodium oxybate for cataplexy, daytime sleepiness, and disrupted sleep based on 3 level-1 studies and 2 level-4 studies.46
Sodium oxybate increases slow-wave sleep, improves sleep continuity, and often helps to mitigate daytime sleepiness. Due to its short half-life, its administration is unusual: the first dose is taken before bedtime and the second dose 2.5 to 4 hours later. Some patients set an alarm clock to take the second dose, while others awaken spontaneously to take the second dose. Most patients find that with adherence to dosing and safety instructions, sodium oxybate can serve as a highly effective form of treatment of both excessive sleepiness and cataplexy and may reduce the need for stimulant-based therapies.
The most common adverse effects are nausea, mood swings, and enuresis. Occasionally, psychosis can result and limit use of the drug. Obstructive sleep apnea can also develop or worsen.52 Because of its high salt content, sodium oxybate should be used with caution in those with heart failure, hypertension, or renal impairment. Its relative, gamma hydroxybutyrate, causes rapid sedation and has been notorious for illegal use as a date rape drug.
In the United States, sodium oxybate is distributed only through a central pharmacy to mitigate potential abuse. Due to this system, the rates of diversion are extremely low, estimated in a postmarketing analysis to be 1 instance per 5,200 patients treated. In the same study, abuse and dependence were both rare as well, about 1 case for every 2,600 and 6,500 patients treated.6,18,52,53
Antidepressants promote the action of norepinephrine and, to a lesser degree, serotonin, thereby suppressing REM sleep.
Venlafaxine, a serotonin-norepinephrine reuptake inhibitor, is often used as a first-line treatment for cataplexy. Selective serotonin reuptake inhibitors such as fluoxetine are also used with success. Tricyclic antidepressants such as protriptyline or clomipramine are extremely effective for cataplexy, but are rarely used due to their adverse effects.2,47
FUTURE WORK
While our understanding of narcolepsy has advanced, there are still gaps in our knowledge of the disorder—namely, the specific trigger for the loss of hypocretin neurons in type 1 narcolepsy and the underlying pathophysiology of type 2.
A number of emerging therapies target the hypocretin system, including peptide replacement, neuronal transplant, and immunotherapy preventing hypocretin neuronal cell death.50,54,55 Additional drugs designed to improve alertness that do not involve the hypocretin system are also being developed, including a histamine inverse agonist.50,56 Sodium oxybate and modafinil, although currently approved for use in adults, are still off-label in pediatric practice. Studies of the safety and efficacy of these medications in children are needed.7,57
Narcolepsy was originally described in the late 1800s by the French physician Jean-Baptiste-Edouard Gélineau, who reported the case of a wine merchant suffering from somnolence. In this first description, he coined the term narcolepsie by joining the Greek words narke (numbness or stupor) and lepsis (attack).1
Since then, the disorder has been further characterized, and some insight into its biological underpinnings has been established. Importantly, treatments have improved and expanded, facilitating its management and thereby improving quality of life for those with the disorder.
This review focuses on clinically relevant features of the disorder and proposes management strategies.
CLINICAL FEATURES
Narcolepsy is characterized by instability of sleep-wake transitions.
Daytime sleepiness
Clinically, narcolepsy manifests with excessive daytime sleepiness that can be personally and socially disabling. Cataplexy, sleep paralysis, and hypnagogic or hypnopompic hallucinations can also be present,2,3 but they are not necessary for diagnosis. In fact, a minority of patients with narcolepsy have all these symptoms.4 Narcolepsy is divided into type 1 (with cataplexy) and type 2 (without cataplexy).2
Sleepiness tends to be worse with inactivity, and sleep can often be irresistible. Sleep attacks can come on suddenly and may be brief enough to manifest as a lapse in consciousness.
Short naps tend to be refreshing. Rapid eye movement (REM) latency—the interval between falling asleep and the onset of the REM sleep—is short in narcolepsy, and since the REM stage is when dreaming occurs, naps often include dreaming. Therefore, when taking a history, it is worthwhile to ask patients whether they dream during naps; a yes answer supports the diagnosis of narcolepsy.5
In children, sleepiness can manifest in reduced concentration and behavioral issues.6 Napping after age 5 or 6 is considered abnormal and may reflect pathologic sleepiness.7
Cataplexy
Cataplexy—transient muscle weakness triggered by emotion—is a specific feature of narcolepsy type 1. It often begins in the facial muscles and can manifest with slackening of the jaw or brief dropping of the head. However, episodes can be more dramatic and, if the trunk and limb muscles are affected, can result in collapsing to the ground.
Cataplexy usually has its onset at about the same time as the sleepiness associated with narcolepsy, but it can arise even years later.8 Episodes can last from a few seconds to 2 minutes. Consciousness is always preserved. A range of emotions can trigger cataplexy, but typically the emotion is a positive one such as laughter or excitement.9 Deep tendon reflexes disappear in cataplexy, so checking reflexes during a witnessed episode can be clinically valuable.2
Cataplexy can worsen with stress and insufficient sleep, occasionally with “status cataplecticus,” in which repeated, persistent episodes of cataplexy occur over several hours.8 Status cataplecticus can be spontaneous or an effect of withdrawal from anticataplectic medications.2
Cataplexy is thought to represent intrusion of REM sleep and its associated muscle atonia during wakefulness.
Sleep paralysis, hallucinations
Sleep paralysis and hallucinations are other features of narcolepsy that reflect this REM dissociation from sleep.
Sleep paralysis occurs most commonly upon awakening, but sometimes just before sleep onset. In most cases, it is manifested by inability to move the limbs or speak, lasting several seconds or, in rare cases, minutes at a time. Sleep paralysis can be associated with a sensation of fear or suffocation, especially when initially experienced.8
Hypnopompic hallucinations, occurring upon awakening, are more common than hypnagogic hallucinations, which are experienced before falling asleep. The hallucinations are often vivid and usually visual, although other types of hallucinations are possible. Unlike those that occur in psychotic disorders, the hallucinations tend to be associated with preserved insight that they are not real.10
Notably, both sleep paralysis and hallucinations are nonspecific symptoms that are common in the general population.8,11,12
Fragmented sleep
Although they are very sleepy, people with narcolepsy generally cannot stay asleep for very long. Their sleep tends to be extremely fragmented, and they often wake up several times a night.2
This sleep pattern reflects the inherent instability of sleep-wake transitions in narcolepsy. In fact, over a 24-hour period, adults with narcolepsy have a normal amount of sleep.13 In children, however, when narcolepsy first arises, the 24-hour sleep time can increase abruptly and can sometimes be associated with persistent cataplexy that can manifest as a clumsy gait.14
Weight gain, obstructive sleep apnea
Weight gain is common, particularly after symptom onset, and especially in children. As a result, obesity is a frequent comorbidity.15 Because obstructive sleep apnea can consequently develop, all patients with narcolepsy require screening for sleep-disordered breathing.
Other sleep disorders often accompany narcolepsy and are more common than in the general population.16 In a study incorporating both clinical and polysomnographic data of 100 patients with narcolepsy, insomnia was the most common comorbid sleep disorder, with a prevalence of 28%; others were REM sleep behavior disorder (24%), restless legs syndrome (24%), obstructive sleep apnea (21%), and non-REM parasomnias.17
PSYCHOSOCIAL CONSEQUENCES
Narcolepsy has significant psychosocial consequences. As a result of their symptoms, people with narcolepsy may not be able to meet academic or work-related demands.
Additionally, their risk of a motor vehicle accident is 3 to 4 times higher than in the general population, and more than one-third of patients have been in an accident due to sleepiness.18 There is some evidence to show that treatment eliminates this risk.19
Few systematic studies have examined mood disorders in narcolepsy. However, studies tend to show a higher prevalence of psychiatric disorders than in the general population, with depression and anxiety the most com-mon.20,21
DIAGNOSIS IS OFTEN DELAYED
The prevalence of narcolepsy type 1 is between 25 and 100 per 100,000 people.22 In a Mayo Clinic study,23 the incidence of narcolepsy type 1 was estimated to be 0.74 per 100,000 person-years. Epidemiologic data on narcolepsy type 2 are sparse, but patients with narcolepsy without cataplexy are thought to represent only 36% of all narcolepsy patients.23
Diagnosis is often delayed, with the average time between the onset of symptoms and the diagnosis ranging from 8 to 22 years. With increasing awareness, the efficiency of the diagnostic process is improving, and this delay is expected to lessen accordingly.24
Symptoms most commonly arise in the second decade; but the age at onset ranges significantly, between the first and fifth decades. Narcolepsy has a bimodal distribution in incidence, with the biggest peak at approximately age 15 and second smaller peak in the mid-30s. Some studies have suggested a slight male predominance.23,25
DIAGNOSIS
History is key
The history should include specific questions about the hallmark features of narcolepsy, including cataplexy, sleep paralysis, and sleep-related hallucinations. For individual assessment of subjective sleepiness, the Epworth Sleepiness Scale or Pediatric Daytime Sleepiness Scale can be administered quickly in the office setting.26,27
The Epworth score is calculated from the self-rated likelihood of falling asleep in 8 different situations, with possible scores of 0 (would never doze) to 3 (high chance of dozing) on each question, for a total possible score of 0 to 24. Normal total scores are between 0 and 10, while scores greater than 10 reflect pathologic sleepiness. Scores on the Epworth Sleepiness Scale in those with narcolepsy tend to reflect moderate to severe sleepiness, or at least 13, as opposed to patients with obstructive sleep apnea, whose scores commonly reflect milder sleepiness.28
Testing with actigraphy and polysomnography
It is imperative to rule out insufficient sleep and other sleep disorders as a cause of daytime sleepiness. This can be done with a careful clinical history, actigraphy with sleep logs, and polysomnography.
In the 2 to 4 weeks before actigraphy and subsequent testing, all medications with alerting or sedating properties (including antidepressants) should be tapered off to prevent influence on the results of the study.
Delayed sleep-phase disorder presents at a similar age as narcolepsy and can be associated with similar degrees of sleepiness. However, individuals with delayed sleep phase disorder have an inappropriately timed sleep-wake cycle so that there is a shift in their desired sleep onset and awakening times. It is common—prevalence estimates vary but average about 1% in the general population.29
Insufficient sleep syndrome is even more common, especially in teenagers and young adults, with increasing family, social, and academic demands. Sleep needs vary across the life span. A teenager needs 8 to 10 hours of sleep per night, and a young adult needs 7 to 9 hours. A study of 1,285 high school students found that 10.4% were not getting enough sleep.30
If actigraphy data suggest a circadian rhythm disorder or insufficient sleep that could explain the symptoms of sleepiness, then further testing should be halted and these specific issues should be addressed. In these cases, working with the patient toward maintaining a regular sleep-wake schedule with 7 to 8 hours of nightly sleep will often resolve symptoms.
If actigraphy demonstrates the patient is maintaining a regular sleep schedule and allowing adequate time for nightly sleep, the next step is polysomnography.
Polysomnography is performed to detect other disorders that can disrupt sleep, such as sleep-disordered breathing or periodic limb movement disorder.2,5 In addition, polysomnography can provide assurance that adequate sleep was obtained prior to the next step in testing.
Multiple sleep latency test
If sufficient sleep is obtained on polysomnograpy (at least 6 hours for an adult) and no other sleep disorder is identified, a multiple sleep latency test is performed. A urine toxicology screen is typically performed on the day of the test to ensure that drugs are not affecting the results.
The multiple sleep latency test consists of 4 to 5 nap opportunities at 2-hour intervals in a quiet dark room conducive to sleep, during which both sleep and REM latency are recorded. The sleep latency of those with narcolepsy is significantly shortened, and the diagnosis of narcolepsy requires an average sleep latency of less than 8 minutes.
Given the propensity for REM sleep in narcolepsy, another essential feature for diagnosis is the sleep-onset REM period (SOREMP). A SOREMP is defined as a REM latency of less than 15 minutes. A diagnosis of narcolepsy re-quires a SOREMP in at least 2 of the naps in a multiple sleep latency test (or 1 nap if the shortened REM latency is seen during polysomnography).31
The multiple sleep latency test has an imperfect sensitivity, though, and should be repeated when there is a high suspicion of narcolepsy.32–34 It is not completely specific either, and false-positive results occur. In fact, SOREMPs can be seen in the general population, particularly in those with a circadian rhythm disorder, insufficient sleep, or sleep-disordered breathing. Two or more SOREMPs in an multiple sleep latency test can be seen in a small proportion of the general population.35 The results of a multiple sleep latency test should be interpreted in the clinical context.
Differential diagnosis
Narcolepsy type 1 is distinguished from type 2 by the presence of cataplexy. A cerebrospinal fluid hypocretin 1 level of 110 pg/mL or less, or less than one-third of the mean value obtained in normal individuals, can substitute for the multiple sleep latency test in diagnosing narcolepsy type 1.31 Currently, hypocretin testing is generally not performed in clinical practice, although it may become a routine part of the narcolepsy evaluation in the future.
Thus, according to the International Classification of Sleep Disorders, 3rd edition,31 the diagnosis of narcolepsy type 1 requires excessive daytime sleepiness for at least 3 months that cannot be explained by another sleep disorder, medical or neurologic disorder, mental disorder, medication use, or substance use disorder, and at least 1 of the following:
- Cataplexy and mean sleep latency of 8 minutes or less with at least 2 SOREMPs on multiple sleep latency testing (1 of which can be on the preceding night’s polysomography)
- Cerebrospinal fluid hypocretin 1 levels less than 110 pg/mL or one-third the baseline normal levels and mean sleep latency ≤ 8 minutes with ≥ 2 SOREMPs on multiple sleep latency testing.
Similarly, the diagnosis of narcolepsy type 2 requires excessive daytime sleepiness for at least 3 months that cannot be explained by another sleep disorder, medical or neurological disorder, mental disorder, medication use, or substance use disorder, plus:
- Mean sleep latency of 8 minutes or less with at least 2 SOREMPs on multiple sleep latency testing.
Idiopathic hypersomnia, another disorder of central hypersomnolence, is also characterized by disabling sleepiness. It is diagnostically differentiated from narcolepsy, as there are fewer than 2 SOREMPs. As opposed to narcolepsy, in which naps tend to be refreshing, even prolonged naps in idiopathic hypersomnia are often not helpful in restoring wakefulness. In idiopathic hypersomnia, sleep is usually not fragmented, and there are few nocturnal arousals. Sleep times can often be prolonged as well, whereas in narcolepsy total sleep time through the day may not be increased but is not consolidated.
Kleine-Levin syndrome is a rarer disorder of hypersomnia. It is episodic compared with the relatively persistent sleepiness in narcolepsy and idiopathic hypersomnia. Periods of hypersomnia occur intermittently for days to weeks and are accompanied by cognitive and behavioral changes including hyperphagia and hypersexuality.4
LINKED TO HYPOCRETIN DEFICIENCY
Over the past 2 decades, the underlying pathophysiology of narcolepsy type 1 has been better characterized.
Narcolepsy type 1 has been linked to a deficiency in hypocretin in the central nervous system.36 Hypocretin (also known as orexin) is a hormone produced in the hypothalamus that acts on multiple brain regions and maintains alertness. For unclear reasons, hypothalamic neurons producing hypocretin are selectively reduced in narcolepsy type 1. Hypocretin also stabilizes wakefulness and inhibits REM sleep; therefore, hypocretin deficiency can lead to inappropriate intrusions of REM sleep onto wakefulness, leading to the hallmark features of narcolepsy—cataplexy, sleep-related hallucinations, and sleep paralysis.37 According to one theory, cataplexy is triggered by emotional stimuli because of a pathway between the medial prefrontal cortex and the amygdala to the pons.38
Cerebrospinal fluid levels of hypocretin in patients with narcolepsy type 2 tend to be normal, and the biologic underpinnings of narcolepsy type 2 remain mysterious. However, in the subgroup of those with narcolepsy type 2 in which hypocretin is low, many individuals go on to develop cataplexy, thereby evolving to narcolepsy type 1.36
POSSIBLE AUTOIMMUNE BASIS
Narcolepsy is typically a sporadic disorder, although familial cases have been described. The risk of a parent with narcolepsy having a child who is affected is approximately 1%.5
Narcolepsy type 1 is strongly associated with HLA-DQB1*0602, with up to 95% of those affected having at least one allele.39 Having 2 copies of the allele further increases the risk of developing narcolepsy.40 However, this allele is far from specific for narcolepsy with cataplexy, as it occurs in 12% to 38% of the general population.41 Therefore, HLA typing currently has limited clinical utility. The exact cause is as yet unknown, but substantial literature proposes an autoimmune basis of the disorder, given the strong association with the HLA subtype.42–44
After the 2009 H1N1 influenza pandemic, there was a significant increase in the incidence of narcolepsy with cataplexy, which again sparked interest in an autoimmune etiology underlying the disorder. Pandemrix, an H1N1 vaccine produced as a result of the 2009 pandemic, appeared to also be associated with an increase in the incidence of narcolepsy. An association with other upper respiratory infections has also been noted, further supporting a possible autoimmune basis.
A few studies have looked for serum autoantibodies involved in the pathogenesis of narcolepsy. Thus far, only one has been identified, an antibody to Tribbles homolog 2, found in 20% to 40% of those with new onset of nar-colepsy.42–44
TREATMENTS FOR DAYTIME SLEEPINESS
As with many chronic disorders, the treatment of narcolepsy consists of symptomatic rather than curative management, which can be done through both pharmacologic and nonpharmacologic means.
Nondrug measures
Scheduled naps lasting 15 to 20 minutes can help improve alertness.45 A consistent sleep schedule with good sleep hygiene, ensuring sufficient nightly sleep, is also important. In one study, the combination of scheduled naps and regular nocturnal sleep times reduced the level of daytime sleepiness and unintentional daytime sleep. Daytime naps were most helpful for those with the highest degree of daytime sleepiness.45
Strategic use of caffeine can be helpful and can reduce dependence on pharmacologic treatment.
Screening should be performed routinely for other sleep disorders, such as sleep-disordered breathing, which should be treated if identified.5,18 When being treated for other medical conditions, individuals with narcolepsy should avoid medications that can cause sedation, such as opiates or barbiturates; alcohol should be minimized or avoided.
Networking with other individuals with narcolepsy through support groups such as Narcolepsy Network can be valuable for learning coping skills and connecting with community resources. Psychological counseling for the patient, and sometimes the family, can also be useful. School-age children may need special accommodations such as schedule adjustments to allow for scheduled naps or frequent breaks to maintain alertness.
People with narcolepsy tend to function better in careers that do not require long periods of sitting, as sleepiness tends to be worse, but instead offer flexibility and require higher levels of activity that tend to combat sleepiness. They should not work as commercial drivers.18
Medications
While behavioral interventions in narcolepsy are vital, they are rarely sufficient, and drugs that promote daytime wakefulness are used as an adjunct (Table 2).46
Realistic expectations should be established when starting, as some degree of residual sleepiness usually remains even with optimal medical therapy. Medications should be strategically scheduled to maximize alertness during necessary times such as at work or school or during driving. Patients should specifically be counseled to avoid driving if sleepy.18,47
Modafinil is often used as a first-line therapy, given its favorable side-effect profile and low potential for abuse. Its pharmacologic action has been debated but it probably acts as a selective dopamine reuptake inhibitor. It is typically taken twice daily (upon waking and early afternoon) and is usually well tolerated.
Potential side effects include headache, nausea, dry mouth, anorexia, diarrhea, and, rarely, Stevens-Johnson syndrome. Cardiovascular side effects are minimal, making it a favorable choice in older patients.18,48
A trial in 283 patients showed significantly lower levels of sleepiness in patients taking modafinil 200 mg or 400 mg than in a control group. Other trials have supported these findings and showed improved driving performance on modafinil.18
Notably, modafinil can increase the metabolism of oral contraceptives, thereby reducing their efficacy. Women of childbearing age should be warned about this interaction and should be transitioned to nonhormonal forms of contraception.2,47
Armodafinil, a purified R-isomer of modafinil, has a longer half-life and requires only once-daily dosing.5
If modafinil or armodafinil fails to optimally manage daytime sleepiness, a traditional stimulant such as methylphenidate or an amphetamine is often used.
Methylphenidate and amphetamines primarily inhibit the reuptake and increase the release of the monoamines, mainly dopamine, and to a lesser degree serotonin and norepinephrine.
These drugs have more significant adverse effects that can involve the cardiovascular system, causing hypertension and arrhythmias. Anorexia, weight loss, and, particularly with high doses, psychosis can occur.49
These drugs should be avoided in patients with a history of significant cardiovascular disease. Before starting stimulant therapy, a thorough cardiovascular examination should be done, often including electrocardiography to ensure there is no baseline arrhythmia.
Patients on these medications should be followed closely to ensure that blood pressure, pulse, and weight are not negatively affected.18,50 Addiction and tolerance can develop with these drugs, and follow-up should include assessment for dependence. Some states may require prescription drug monitoring to ensure the drugs are not being abused or diverted.
Short- and long-acting formulations of both methylphenidate and amphetamines are available, and a long-acting form is often used in conjunction with a short-acting form as needed.18
Addiction and drug-seeking behavior can develop but are unusual in those taking stimulants to treat narcolepsy.49
Follow-up
Residual daytime sleepiness can be measured subjectively through the Epworth Sleepiness Scale on follow-up. If necessary, a maintenance-of-wakefulness test can provide an objective assessment of treatment efficacy.18
As narcolepsy is a chronic disorder, treatment should evolve with time. Most medications that treat narcolepsy are categorized by the US Food and Drug Administration as pregnancy category C, as we do not have adequate studies in human pregnancies to evaluate their effects. When a patient with narcolepsy becomes pregnant, she should be counseled about the risks and benefits of remaining on therapy. Treatment should balance the risks of sleepiness with the potential risks of remaining on medications.50 In the elderly, as cardiovascular comorbidities tend to increase, the risks and benefits of therapy should be routinely reevaluated.
For cataplexy
Sodium oxybate,51–53 the most potent anticataplectic drug, is the sodium salt of gamma hydroxybutyrate, a metabolite of gamma-aminobutyric acid. Sodium oxybate can be prescribed in the United States, Canada, and Europe. The American Academy of Sleep Medicine recommends sodium oxybate for cataplexy, daytime sleepiness, and disrupted sleep based on 3 level-1 studies and 2 level-4 studies.46
Sodium oxybate increases slow-wave sleep, improves sleep continuity, and often helps to mitigate daytime sleepiness. Due to its short half-life, its administration is unusual: the first dose is taken before bedtime and the second dose 2.5 to 4 hours later. Some patients set an alarm clock to take the second dose, while others awaken spontaneously to take the second dose. Most patients find that with adherence to dosing and safety instructions, sodium oxybate can serve as a highly effective form of treatment of both excessive sleepiness and cataplexy and may reduce the need for stimulant-based therapies.
The most common adverse effects are nausea, mood swings, and enuresis. Occasionally, psychosis can result and limit use of the drug. Obstructive sleep apnea can also develop or worsen.52 Because of its high salt content, sodium oxybate should be used with caution in those with heart failure, hypertension, or renal impairment. Its relative, gamma hydroxybutyrate, causes rapid sedation and has been notorious for illegal use as a date rape drug.
In the United States, sodium oxybate is distributed only through a central pharmacy to mitigate potential abuse. Due to this system, the rates of diversion are extremely low, estimated in a postmarketing analysis to be 1 instance per 5,200 patients treated. In the same study, abuse and dependence were both rare as well, about 1 case for every 2,600 and 6,500 patients treated.6,18,52,53
Antidepressants promote the action of norepinephrine and, to a lesser degree, serotonin, thereby suppressing REM sleep.
Venlafaxine, a serotonin-norepinephrine reuptake inhibitor, is often used as a first-line treatment for cataplexy. Selective serotonin reuptake inhibitors such as fluoxetine are also used with success. Tricyclic antidepressants such as protriptyline or clomipramine are extremely effective for cataplexy, but are rarely used due to their adverse effects.2,47
FUTURE WORK
While our understanding of narcolepsy has advanced, there are still gaps in our knowledge of the disorder—namely, the specific trigger for the loss of hypocretin neurons in type 1 narcolepsy and the underlying pathophysiology of type 2.
A number of emerging therapies target the hypocretin system, including peptide replacement, neuronal transplant, and immunotherapy preventing hypocretin neuronal cell death.50,54,55 Additional drugs designed to improve alertness that do not involve the hypocretin system are also being developed, including a histamine inverse agonist.50,56 Sodium oxybate and modafinil, although currently approved for use in adults, are still off-label in pediatric practice. Studies of the safety and efficacy of these medications in children are needed.7,57
- Gélineau J. De la narcolepsie. Gazette des Hôpitaux Civils et Militaires 1880; part a, 53:626–628, part b, 54:635–637.
- Dauvilliers Y, Arnulf I, Mignot E. Narcolepsy with cataplexy. Lancet 2007; 369(9560):499–511. doi:10.1016/S0140-6736(07)60237-2
- Scammell TE. Clinical features and diagnosis of narcolepsy in adults. In: Eichler AF, ed. UpToDate. Waltham, MA: UpToDate; 2018. www.uptodate.com. Accessed October 31, 2018.
- Morrish E, King MA, Smith IE, Shneerson JM. Factors associated with a delay in the diagnosis of narcolepsy. Sleep Med 2004; 5(1):37–41. pmid:14725825
- Scammell TE. Narcolepsy. N Engl J Med 2015; 373(27):2654–2662. doi:10.1056/NEJMra1500587
- Babiker MO, Prasad M. Narcolepsy in children: a diagnostic and management approach. Pediatr Neurol 2015; 52(6):557–565. doi:10.1016/j.pediatrneurol.2015.02.020
- Kotagal S. Narcolepsy in children. In: UpToDate, Eichler AF, ed. UpToDate, Waltham, MA. www.uptodate.com. Accessed October 31, 2018.
- Scammell TE. The neurobiology, diagnosis, and treatment of narcolepsy. Ann Neurol 2003; 53(2):154–166. doi:10.1002/ana.10444
- Overeem S, van Nues SJ, van der Zande WL, Donjacour CE, van Mierlo P, Lammers GJ. The clinical features of cataplexy: a questionnaire study in narcolepsy patients with and without hypocretin-1 deficiency. Sleep Med 2011; 12(1):12–18. doi:10.1016/j.sleep.2010.05.010
- Plazzi G, Fabbri C, Pizza F, Serretti A. Schizophrenia-like symptoms in narcolepsy type 1: shared and distinctive clinical characteristics. Neuropsychobiology 2015; 71(4):218–224. doi:10.1159/000432400
- Ohayon MM. Prevalence of hallucinations and their pathological associations in the general population. Psychiatry Res 2000; 97(2-3):153–164. pmid:11166087
- Sharpless BA, Barber JP. Lifetime prevalence rates of sleep paralysis: a systematic review. Sleep Med Rev 2011;5(5):311–315. doi:10.1016/j.smrv.2011.01.007
- Broughton R, Dunham W, Newman J, Lutley K, Duschesne P, Rivers M. Ambulatory 24 hour sleep-wake monitoring in narcolepsy-cataplexy compared to matched controls. Electroencephalogr Clin Neurophysiol 1988; 70(6):473–481. pmid:2461281
- Pizza F, Franceschini C, Peltola H, et al. Clinical and polysomnographic course of childhood narcolepsy with cataplexy. Brain 2013; 136(pt 12):3787–3795. doi:10.1093/brain/awt277
- Kotagal S, Krahn LE, Slocumb N. A putative link between childhood narcolepsy and obesity. Sleep Med 2004; 5(2):147–150. doi:10.1016/j.sleep.2003.10.006
- Pizza F, Tartarotti S, Poryazova R, Baumann CR, Bassetti CL. Sleep-disordered breathing and periodic limb movements in narcolepsy with cataplexy: a systematic analysis of 35 consecutive patients. Eur Neurol 2013; 70(1-2):22–26. doi:10.1159/000348719
- Frauscher B, Ehrmann L, Mitterling T, et al. Delayed diagnosis, range of severity, and multiple sleep comorbidities: a clinical and polysomnographic analysis of 100 patients of the Innsbruck narcolepsy cohort. J Clin Sleep Med 2013; 9(8):805–812. doi:10.5664/jcsm.2926
- Scammell TE. Treatment of narcolepsy in adults. In: Eichler AF, ed. UpToDate, Waltham, MA. www.uptodate.com. Accessed October 31, 2018.
- Pizza F, Jaussent I, Lopez R, et al. Car crashes and central disorders of hypersomnolence: a French study. PLoS One 2015; 10(6):e0129386. doi:10.1371/journal.pone.0129386
- Fortuyn HD, Lappenschaar MA, Furer JW, et al. Anxiety and mood disorders in narcolepsy: a case-control study. Gen Hosp Psychiatry 2010; 32(1):49–56. doi:10.1016/j.genhosppsych.2009.08.007
- Ruoff CM, Reaven NL, Funk SE, et al. High rates of psychiatric comorbidity in narcolepsy: findings from the Burden of Narcolepsy Disease (BOND) study of 9,312 patients in the United States. J Clin Psychiatry 2017; 78(2):171–176. doi:10.4088/JCP.15m10262
- Longstreth WT Jr, Koepsell TD, Ton TG, Hendrickson AF, van Belle G. The epidemiology of narcolepsy. Sleep. 2007; 30(1):13–26. pmid:17310860
- Silber MH, Krahn LE, Olson EJ, Pankratz VS. The epidemiology of narcolepsy in Olmsted County, Minnesota: a population-based study. Sleep 2002; 25(2):197–202. pmid:11902429
- Thorpy MJ, Krieger AC. Delayed diagnosis of narcolepsy: characterization and impact. Sleep Med 2014; 15(5):502–507. doi:10.1016/j.sleep.2014.01.015
- Dauvilliers Y, Montplaisir J, Molinari N, et al. Age at onset of narcolepsy in two large populations of patients in France and Quebec. Neurology 2001; 57(11):2029–2033. pmid:11739821
- Johns MW. A new method for measuring daytime sleepiness: the Epworth sleepiness scale. Sleep 1991; 14(6):540–545. pmid:1798888
- Drake C, Nickel C, Burduvali E, Roth T, Jefferson C, Badia P. The pediatric daytime sleepiness scale (PDSS): sleep habits and school outcomes in middle-school children. Sleep 2003; 26(4):455–458. pmid:12841372
- van der Heide A, van Schie MK, Lammers GJ, et al. Comparing treatment effect measurements in narcolepsy: the sustained attention to response task, Epworth sleepiness scale and maintenance of wakefulness test. Sleep 2015; 38(7):1051–1058. doi:10.5665/sleep.4810
- Nesbitt AD. Delayed sleep-wake phase disorder. J Thorac Dis 2018; 10(suppl 1):S103–S111. doi:10.21037/jtd.2018.01.11
- Pallesen S, Saxvig IW, Molde H, Sørensen E, Wilhelmsen-Langeland A, Bjorvatn B. Brief report: behaviorally induced insufficient sleep syndrome in older adolescents: prevalence and correlates. J Adolesc 2011; 34(2):391–395. doi:10.1016/j.adolescence.2010.02.005
- American Academy of Sleep Medicine. International Classification of Sleep Disorders. 3rd ed. Darien, IL: American Academy of Sleep Disorders; 2014.
- Trotti LM, Staab BA, Rye DB. Test-retest reliability of the multiple sleep latency test in narcolepsy without cataplexy and idiopathic hypersomnia. J Clin Sleep Med 2013; 9(8):789–795. doi:10.5664/jcsm.2922
- Andlauer O, Moore H, Jouhier L, et al. Nocturnal rapid eye movement sleep latency for identifying patients with narcolepsy/hypocretin deficiency. JAMA Neurol 2013; 70(7):891–902. doi:10.1001/jamaneurol.2013.1589
- Cairns A, Bogan R. Prevalence and clinical correlates of a short onset REM period (SOREMP) during routine PSG. Sleep 2015; 38(10):1575–1581. doi:10.5665/sleep.5050
- Mignot E, Lin L, Finn L, et al. Correlates of sleep-onset REM periods during the multiple sleep latency test in community adults. Brain 2006; 129(6):1609–1623. doi:10.1093/brain/awl079
- Nishino S, Ripley B, Overeem S, Lammers GJ, Mignot E. Hypocretin (orexin) deficiency in human narcolepsy. Lancet 2000; 355(9197):39–40. doi:10.1016/S0140-6736(99)05582-8
- Peyron C, Faraco J, Rogers W, et al. A mutation in a case of early onset narcolepsy and a generalized absence of hypocretin peptides in human narcoleptic brains. Nat Med 2000; 6(9):991–997. doi:10.1038/79690
- Oishi Y, Williams RH, Agostinelli L, et al. Role of the medial prefrontal cortex in cataplexy. J Neurosci 2013; 33(23):9743–9751. doi:10.1523/JNEUROSCI.0499-13.2013
- Mignot E, Hayduk R, Black J, Grumet FC, Guilleminault C. HLA DQB1*0602 is associated with cataplexy in 509 narcoleptic patients.. Sleep 1997; 20(11):1012–1020. pmid:9456467
- Pelin Z, Guilleminault C, Risch N, Grumet FC, Mignot E. HLA-DQB1*0602 homozygosity increases relative risk for narcolepsy but not disease severity in two ethnic groups. US Modafinil in Narcolepsy Multicenter Study Group. Tissue Antigens 1998; 51(1):96–100. pmid:9459509
- Akintomide GS, Rickards H. Narcolepsy: a review. Neuropsychiatr Dis Treat 2011; 7(1):507–518. doi:10.2147/NDT.S23624
- Mahlios J, De la Herrán-Arita AK, Mignot E. The autoimmune basis of narcolepsy. Curr Opin Neurobiol 2013; 23(5):767–773. doi:10.1016/j.conb.2013.04.013
- Degn M, Kornum BR. Type 1 narcolepsy: a CD8(+) T cell-mediated disease? Ann N Y Acad Sci 2015;1 351:80–88. doi:10.1111/nyas.12793
- Liblau RS, Vassalli A, Seifinejad A, Tafti M. Hypocretin (orexin) biology and the pathophysiology of narcolepsy with cataplexy. Lancet Neurol 2015; 14(3):318–328. doi:10.1016/S1474-4422(14)70218-2
- Rogers AE, Aldrich MS, Lin X. A comparison of three different sleep schedules for reducing daytime sleepiness in narcolepsy. Sleep 2001; 24(4):385–391. pmid:11403522
- Morgenthaler TI, Kapur VK, Brown TM, et al; Standards of Practice Committee of the American Academy of Sleep Medicine. Practice parameters for the treatment of narcolepsy and other hypersomnias of central origin. Sleep 2007; 30(12):1705–1711. pmid:18246980
- Mignot EJ. A practical guide to the therapy of narcolepsy and hypersomnia syndromes. Neurotherapeutics 2012; 9(4):739–752. doi:10.1007/s13311-012-0150-9
- Roth T, Schwartz JR, Hirshkowitz M, Erman MK, Dayno JM, Arora S. Evaluation of the safety of modafinil for treatment of excessive sleepiness. J Clin Sleep Med 2007; 3(6):595–602. pmid:17993041
- Auger RR, Goodman SH, Silber MH, Krahn LE, Pankratz VS, Slocumb NL. Risks of high-dose stimulants in the treatment of disorders of excessive somnolence: a case-control study. Sleep 2005; 28(6):667–672. pmid:16477952
- Abad VC, Guilleminault C. New developments in the management of narcolepsy. Nat Sci Sleep 2017; 9:39–57. doi:10.2147/NSS.S103467
- Drakatos P, Lykouras D, D’Ancona G, et al. Safety and efficacy of long-term use of sodium oxybate for narcolepsy with cataplexy in routine clinical practice. Sleep Med 2017; 35:80–84. doi:10.1016/j.sleep.2017.03.028
- Mansukhani MP, Kotagal S. Sodium oxybate in the treatment of childhood narcolepsy–cataplexy: a retrospective study. Sleep Med 2012; 13(6):606–610. doi:10.1016/j.sleep.2011.10.032
- Wang YG, Swick TJ, Carter LP, Thorpy MJ, Benowitz NL. Safety overview of postmarketing and clinical experience of sodium oxybate (Xyrem): abuse, misuse, dependence, and diversion. J Clin Sleep Med 2009; 5(4):365–371. pmid:19968016
- Weinhold SL, Seeck-Hirschner M, Nowak A, Hallschmid M, Göder R, Baier PC. The effect of intranasal orexin-A (hypocretin-1) on sleep, wakefulness and attention in narcolepsy with cataplexy. Behav Brain Res 2014; 262:8–13. doi:10.1016/j.bbr.2013.12.045
- Arias-Carrión O, Murillo-Rodriguez E. Effects of hypocretin/orexin cell transplantation on narcoleptic-like sleep behavior in rats. PLoS One 2014; 9(4):e95342. doi:10.1371/journal.pone.0095342
- Leu-Semenescu S, Nittur N, Golmard JL, Arnulf I. Effects of pitolisant, a histamine H3 inverse agonist, in drug-resistant idiopathic and symptomatic hypersomnia: a chart review. Sleep Med 2014; 15(6):681–687. doi:10.1016/j.sleep.2014.01.021
- Lecendreux M, Bruni O, Franco P, et al. Clinical experience suggests that modafinil is an effective and safe treatment for paediatric narcolepsy. J Sleep Res 2012; 21(4):481–483. doi:10.1111/j.1365-2869.2011.00991.x
- Gélineau J. De la narcolepsie. Gazette des Hôpitaux Civils et Militaires 1880; part a, 53:626–628, part b, 54:635–637.
- Dauvilliers Y, Arnulf I, Mignot E. Narcolepsy with cataplexy. Lancet 2007; 369(9560):499–511. doi:10.1016/S0140-6736(07)60237-2
- Scammell TE. Clinical features and diagnosis of narcolepsy in adults. In: Eichler AF, ed. UpToDate. Waltham, MA: UpToDate; 2018. www.uptodate.com. Accessed October 31, 2018.
- Morrish E, King MA, Smith IE, Shneerson JM. Factors associated with a delay in the diagnosis of narcolepsy. Sleep Med 2004; 5(1):37–41. pmid:14725825
- Scammell TE. Narcolepsy. N Engl J Med 2015; 373(27):2654–2662. doi:10.1056/NEJMra1500587
- Babiker MO, Prasad M. Narcolepsy in children: a diagnostic and management approach. Pediatr Neurol 2015; 52(6):557–565. doi:10.1016/j.pediatrneurol.2015.02.020
- Kotagal S. Narcolepsy in children. In: UpToDate, Eichler AF, ed. UpToDate, Waltham, MA. www.uptodate.com. Accessed October 31, 2018.
- Scammell TE. The neurobiology, diagnosis, and treatment of narcolepsy. Ann Neurol 2003; 53(2):154–166. doi:10.1002/ana.10444
- Overeem S, van Nues SJ, van der Zande WL, Donjacour CE, van Mierlo P, Lammers GJ. The clinical features of cataplexy: a questionnaire study in narcolepsy patients with and without hypocretin-1 deficiency. Sleep Med 2011; 12(1):12–18. doi:10.1016/j.sleep.2010.05.010
- Plazzi G, Fabbri C, Pizza F, Serretti A. Schizophrenia-like symptoms in narcolepsy type 1: shared and distinctive clinical characteristics. Neuropsychobiology 2015; 71(4):218–224. doi:10.1159/000432400
- Ohayon MM. Prevalence of hallucinations and their pathological associations in the general population. Psychiatry Res 2000; 97(2-3):153–164. pmid:11166087
- Sharpless BA, Barber JP. Lifetime prevalence rates of sleep paralysis: a systematic review. Sleep Med Rev 2011;5(5):311–315. doi:10.1016/j.smrv.2011.01.007
- Broughton R, Dunham W, Newman J, Lutley K, Duschesne P, Rivers M. Ambulatory 24 hour sleep-wake monitoring in narcolepsy-cataplexy compared to matched controls. Electroencephalogr Clin Neurophysiol 1988; 70(6):473–481. pmid:2461281
- Pizza F, Franceschini C, Peltola H, et al. Clinical and polysomnographic course of childhood narcolepsy with cataplexy. Brain 2013; 136(pt 12):3787–3795. doi:10.1093/brain/awt277
- Kotagal S, Krahn LE, Slocumb N. A putative link between childhood narcolepsy and obesity. Sleep Med 2004; 5(2):147–150. doi:10.1016/j.sleep.2003.10.006
- Pizza F, Tartarotti S, Poryazova R, Baumann CR, Bassetti CL. Sleep-disordered breathing and periodic limb movements in narcolepsy with cataplexy: a systematic analysis of 35 consecutive patients. Eur Neurol 2013; 70(1-2):22–26. doi:10.1159/000348719
- Frauscher B, Ehrmann L, Mitterling T, et al. Delayed diagnosis, range of severity, and multiple sleep comorbidities: a clinical and polysomnographic analysis of 100 patients of the Innsbruck narcolepsy cohort. J Clin Sleep Med 2013; 9(8):805–812. doi:10.5664/jcsm.2926
- Scammell TE. Treatment of narcolepsy in adults. In: Eichler AF, ed. UpToDate, Waltham, MA. www.uptodate.com. Accessed October 31, 2018.
- Pizza F, Jaussent I, Lopez R, et al. Car crashes and central disorders of hypersomnolence: a French study. PLoS One 2015; 10(6):e0129386. doi:10.1371/journal.pone.0129386
- Fortuyn HD, Lappenschaar MA, Furer JW, et al. Anxiety and mood disorders in narcolepsy: a case-control study. Gen Hosp Psychiatry 2010; 32(1):49–56. doi:10.1016/j.genhosppsych.2009.08.007
- Ruoff CM, Reaven NL, Funk SE, et al. High rates of psychiatric comorbidity in narcolepsy: findings from the Burden of Narcolepsy Disease (BOND) study of 9,312 patients in the United States. J Clin Psychiatry 2017; 78(2):171–176. doi:10.4088/JCP.15m10262
- Longstreth WT Jr, Koepsell TD, Ton TG, Hendrickson AF, van Belle G. The epidemiology of narcolepsy. Sleep. 2007; 30(1):13–26. pmid:17310860
- Silber MH, Krahn LE, Olson EJ, Pankratz VS. The epidemiology of narcolepsy in Olmsted County, Minnesota: a population-based study. Sleep 2002; 25(2):197–202. pmid:11902429
- Thorpy MJ, Krieger AC. Delayed diagnosis of narcolepsy: characterization and impact. Sleep Med 2014; 15(5):502–507. doi:10.1016/j.sleep.2014.01.015
- Dauvilliers Y, Montplaisir J, Molinari N, et al. Age at onset of narcolepsy in two large populations of patients in France and Quebec. Neurology 2001; 57(11):2029–2033. pmid:11739821
- Johns MW. A new method for measuring daytime sleepiness: the Epworth sleepiness scale. Sleep 1991; 14(6):540–545. pmid:1798888
- Drake C, Nickel C, Burduvali E, Roth T, Jefferson C, Badia P. The pediatric daytime sleepiness scale (PDSS): sleep habits and school outcomes in middle-school children. Sleep 2003; 26(4):455–458. pmid:12841372
- van der Heide A, van Schie MK, Lammers GJ, et al. Comparing treatment effect measurements in narcolepsy: the sustained attention to response task, Epworth sleepiness scale and maintenance of wakefulness test. Sleep 2015; 38(7):1051–1058. doi:10.5665/sleep.4810
- Nesbitt AD. Delayed sleep-wake phase disorder. J Thorac Dis 2018; 10(suppl 1):S103–S111. doi:10.21037/jtd.2018.01.11
- Pallesen S, Saxvig IW, Molde H, Sørensen E, Wilhelmsen-Langeland A, Bjorvatn B. Brief report: behaviorally induced insufficient sleep syndrome in older adolescents: prevalence and correlates. J Adolesc 2011; 34(2):391–395. doi:10.1016/j.adolescence.2010.02.005
- American Academy of Sleep Medicine. International Classification of Sleep Disorders. 3rd ed. Darien, IL: American Academy of Sleep Disorders; 2014.
- Trotti LM, Staab BA, Rye DB. Test-retest reliability of the multiple sleep latency test in narcolepsy without cataplexy and idiopathic hypersomnia. J Clin Sleep Med 2013; 9(8):789–795. doi:10.5664/jcsm.2922
- Andlauer O, Moore H, Jouhier L, et al. Nocturnal rapid eye movement sleep latency for identifying patients with narcolepsy/hypocretin deficiency. JAMA Neurol 2013; 70(7):891–902. doi:10.1001/jamaneurol.2013.1589
- Cairns A, Bogan R. Prevalence and clinical correlates of a short onset REM period (SOREMP) during routine PSG. Sleep 2015; 38(10):1575–1581. doi:10.5665/sleep.5050
- Mignot E, Lin L, Finn L, et al. Correlates of sleep-onset REM periods during the multiple sleep latency test in community adults. Brain 2006; 129(6):1609–1623. doi:10.1093/brain/awl079
- Nishino S, Ripley B, Overeem S, Lammers GJ, Mignot E. Hypocretin (orexin) deficiency in human narcolepsy. Lancet 2000; 355(9197):39–40. doi:10.1016/S0140-6736(99)05582-8
- Peyron C, Faraco J, Rogers W, et al. A mutation in a case of early onset narcolepsy and a generalized absence of hypocretin peptides in human narcoleptic brains. Nat Med 2000; 6(9):991–997. doi:10.1038/79690
- Oishi Y, Williams RH, Agostinelli L, et al. Role of the medial prefrontal cortex in cataplexy. J Neurosci 2013; 33(23):9743–9751. doi:10.1523/JNEUROSCI.0499-13.2013
- Mignot E, Hayduk R, Black J, Grumet FC, Guilleminault C. HLA DQB1*0602 is associated with cataplexy in 509 narcoleptic patients.. Sleep 1997; 20(11):1012–1020. pmid:9456467
- Pelin Z, Guilleminault C, Risch N, Grumet FC, Mignot E. HLA-DQB1*0602 homozygosity increases relative risk for narcolepsy but not disease severity in two ethnic groups. US Modafinil in Narcolepsy Multicenter Study Group. Tissue Antigens 1998; 51(1):96–100. pmid:9459509
- Akintomide GS, Rickards H. Narcolepsy: a review. Neuropsychiatr Dis Treat 2011; 7(1):507–518. doi:10.2147/NDT.S23624
- Mahlios J, De la Herrán-Arita AK, Mignot E. The autoimmune basis of narcolepsy. Curr Opin Neurobiol 2013; 23(5):767–773. doi:10.1016/j.conb.2013.04.013
- Degn M, Kornum BR. Type 1 narcolepsy: a CD8(+) T cell-mediated disease? Ann N Y Acad Sci 2015;1 351:80–88. doi:10.1111/nyas.12793
- Liblau RS, Vassalli A, Seifinejad A, Tafti M. Hypocretin (orexin) biology and the pathophysiology of narcolepsy with cataplexy. Lancet Neurol 2015; 14(3):318–328. doi:10.1016/S1474-4422(14)70218-2
- Rogers AE, Aldrich MS, Lin X. A comparison of three different sleep schedules for reducing daytime sleepiness in narcolepsy. Sleep 2001; 24(4):385–391. pmid:11403522
- Morgenthaler TI, Kapur VK, Brown TM, et al; Standards of Practice Committee of the American Academy of Sleep Medicine. Practice parameters for the treatment of narcolepsy and other hypersomnias of central origin. Sleep 2007; 30(12):1705–1711. pmid:18246980
- Mignot EJ. A practical guide to the therapy of narcolepsy and hypersomnia syndromes. Neurotherapeutics 2012; 9(4):739–752. doi:10.1007/s13311-012-0150-9
- Roth T, Schwartz JR, Hirshkowitz M, Erman MK, Dayno JM, Arora S. Evaluation of the safety of modafinil for treatment of excessive sleepiness. J Clin Sleep Med 2007; 3(6):595–602. pmid:17993041
- Auger RR, Goodman SH, Silber MH, Krahn LE, Pankratz VS, Slocumb NL. Risks of high-dose stimulants in the treatment of disorders of excessive somnolence: a case-control study. Sleep 2005; 28(6):667–672. pmid:16477952
- Abad VC, Guilleminault C. New developments in the management of narcolepsy. Nat Sci Sleep 2017; 9:39–57. doi:10.2147/NSS.S103467
- Drakatos P, Lykouras D, D’Ancona G, et al. Safety and efficacy of long-term use of sodium oxybate for narcolepsy with cataplexy in routine clinical practice. Sleep Med 2017; 35:80–84. doi:10.1016/j.sleep.2017.03.028
- Mansukhani MP, Kotagal S. Sodium oxybate in the treatment of childhood narcolepsy–cataplexy: a retrospective study. Sleep Med 2012; 13(6):606–610. doi:10.1016/j.sleep.2011.10.032
- Wang YG, Swick TJ, Carter LP, Thorpy MJ, Benowitz NL. Safety overview of postmarketing and clinical experience of sodium oxybate (Xyrem): abuse, misuse, dependence, and diversion. J Clin Sleep Med 2009; 5(4):365–371. pmid:19968016
- Weinhold SL, Seeck-Hirschner M, Nowak A, Hallschmid M, Göder R, Baier PC. The effect of intranasal orexin-A (hypocretin-1) on sleep, wakefulness and attention in narcolepsy with cataplexy. Behav Brain Res 2014; 262:8–13. doi:10.1016/j.bbr.2013.12.045
- Arias-Carrión O, Murillo-Rodriguez E. Effects of hypocretin/orexin cell transplantation on narcoleptic-like sleep behavior in rats. PLoS One 2014; 9(4):e95342. doi:10.1371/journal.pone.0095342
- Leu-Semenescu S, Nittur N, Golmard JL, Arnulf I. Effects of pitolisant, a histamine H3 inverse agonist, in drug-resistant idiopathic and symptomatic hypersomnia: a chart review. Sleep Med 2014; 15(6):681–687. doi:10.1016/j.sleep.2014.01.021
- Lecendreux M, Bruni O, Franco P, et al. Clinical experience suggests that modafinil is an effective and safe treatment for paediatric narcolepsy. J Sleep Res 2012; 21(4):481–483. doi:10.1111/j.1365-2869.2011.00991.x
KEY POINTS
- Features of narcolepsy include daytime sleepiness, sleep attacks, cataplexy (in narcolepsy type 1), sleep paralysis, and sleep-related hallucinations.
- People with narcolepsy feel sleepy and can fall asleep quickly, but they do not stay asleep for long. They go into rapid eye movement sleep soon after falling asleep. Total sleep time is normal, but sleep is fragmented.
- Scheduled naps lasting 15 to 20 minutes can improve alertness. A consistent sleep schedule with good sleep hygiene is also important.
- Modafinil, methylphenidate, and amphetamines are used to manage daytime sleepiness, and sodium oxybate and antidepressants are used for cataplexy.
Night sweats
A 46-year-old man comes to clinic for evaluation of night sweats. He has been having drenching night sweats for the past 3 months. He has to change his night shirt at least once per night. He has had a 10-pound weight gain over the past 6 months. No chest pain, nausea, or fatigue. He has had a cough for the past 6 months.
Which is the most likely diagnosis?
A. Gastroesophageal reflux disease.
B. Tuberculosis.
C. Lymphoma.
D. Multiple myeloma.
Night sweats are a common symptom in the general population, estimated to occur in about 10% of people. They can range in frequency and severity. We become most concerned when the patient is concerned, usually when they report drenching night sweats.
What do we need to know about this symptom to help us think of more likely causes and guide us in a more appropriate workup?
Night sweats do not seem to be a bad prognostic symptom. James W. Mold, MD, and his colleagues looked at the prognostic significance of night sweats in two cohorts of elderly patients.1 The prevalence of night sweats in this study was 10%. These two cohorts were followed for a little more than 7 years. More than 1,500 patients were included in the two cohorts. Patients who reported night sweats were not more likely to die, or die sooner, than were those who didn’t have night sweats. The severity of the night sweats did not make a difference.
Lea et al. described the prevalence of night sweats among different inpatient populations, with a range from 33% in surgical and medicine patients, to 60% on obstetrics service.2
Night sweats are common, and don’t appear to be correlated with worse prognosis. So, what are the likely common causes?
There just aren’t good studies on causes of night sweats, but there are studies that suggest that they are seen in some very common diseases. It is always good to look at medication lists as a start when evaluating unexplained symptoms.
Dr. Mold, along with Barbara J. Holtzclaw, PhD, reported higher odds ratios for night sweats for patients on SSRIs (OR, 3.01), angiotensin receptor blockers (OR, 3.44) and thyroid hormone supplements (OR, 2.53).3 W.A. Reynolds, MD, looked at the prevalence of night sweats in a GI practice.4 A total of 41% of the patients reported night sweats, and 12 of 12 patients with GERD who had night sweats had resolution of the night sweats with effective treatment of the GERD.
Dr. Mold and his colleagues found that night sweats were associated with several sleep-related symptoms, including waking up with a bitter taste in the mouth (OR, 1.94), daytime tiredness (OR, 1.99), and legs jerking during sleep (OR, 1.87).5
Erna Arnardottir, PhD, and her colleagues found that obstructive sleep apnea was associated with frequent nocturnal sweating.6 They found that 31% of men and 33% of women with OSA had nocturnal sweating, compared with about 10% of the general population. When the OSA patients were treated with positive airway pressure, the prevalence of nocturnal sweating decreased to 11.5%, similar to general population numbers.
Pearl: Night sweats are associated with common conditions: medications, GERD, and sleep disorders. These are more likely than lymphoma and tuberculosis.
Dr. Paauw is professor of medicine in the division of general internal medicine at the University of Washington, Seattle, and serves as third-year medical student clerkship director at the University of Washington. Contact Dr. Paauw at [email protected].
References
1. J Am Board Fam Med. 2010 Jan-Feb;23(1):97-103.
2. South Med J. 1985 Sep;78(9):1065-7.
3. Drugs Real World Outcomes. 2015 Mar;2(1):29-33.
4. J Clin Gastroenterol. 1989 Oct;11(5):590-1.
5. Ann Fam Med. 2006 Sep-Oct;4(5):423-6.
6. BMJ Open. 2013 May 14;3(5).
A 46-year-old man comes to clinic for evaluation of night sweats. He has been having drenching night sweats for the past 3 months. He has to change his night shirt at least once per night. He has had a 10-pound weight gain over the past 6 months. No chest pain, nausea, or fatigue. He has had a cough for the past 6 months.
Which is the most likely diagnosis?
A. Gastroesophageal reflux disease.
B. Tuberculosis.
C. Lymphoma.
D. Multiple myeloma.
Night sweats are a common symptom in the general population, estimated to occur in about 10% of people. They can range in frequency and severity. We become most concerned when the patient is concerned, usually when they report drenching night sweats.
What do we need to know about this symptom to help us think of more likely causes and guide us in a more appropriate workup?
Night sweats do not seem to be a bad prognostic symptom. James W. Mold, MD, and his colleagues looked at the prognostic significance of night sweats in two cohorts of elderly patients.1 The prevalence of night sweats in this study was 10%. These two cohorts were followed for a little more than 7 years. More than 1,500 patients were included in the two cohorts. Patients who reported night sweats were not more likely to die, or die sooner, than were those who didn’t have night sweats. The severity of the night sweats did not make a difference.
Lea et al. described the prevalence of night sweats among different inpatient populations, with a range from 33% in surgical and medicine patients, to 60% on obstetrics service.2
Night sweats are common, and don’t appear to be correlated with worse prognosis. So, what are the likely common causes?
There just aren’t good studies on causes of night sweats, but there are studies that suggest that they are seen in some very common diseases. It is always good to look at medication lists as a start when evaluating unexplained symptoms.
Dr. Mold, along with Barbara J. Holtzclaw, PhD, reported higher odds ratios for night sweats for patients on SSRIs (OR, 3.01), angiotensin receptor blockers (OR, 3.44) and thyroid hormone supplements (OR, 2.53).3 W.A. Reynolds, MD, looked at the prevalence of night sweats in a GI practice.4 A total of 41% of the patients reported night sweats, and 12 of 12 patients with GERD who had night sweats had resolution of the night sweats with effective treatment of the GERD.
Dr. Mold and his colleagues found that night sweats were associated with several sleep-related symptoms, including waking up with a bitter taste in the mouth (OR, 1.94), daytime tiredness (OR, 1.99), and legs jerking during sleep (OR, 1.87).5
Erna Arnardottir, PhD, and her colleagues found that obstructive sleep apnea was associated with frequent nocturnal sweating.6 They found that 31% of men and 33% of women with OSA had nocturnal sweating, compared with about 10% of the general population. When the OSA patients were treated with positive airway pressure, the prevalence of nocturnal sweating decreased to 11.5%, similar to general population numbers.
Pearl: Night sweats are associated with common conditions: medications, GERD, and sleep disorders. These are more likely than lymphoma and tuberculosis.
Dr. Paauw is professor of medicine in the division of general internal medicine at the University of Washington, Seattle, and serves as third-year medical student clerkship director at the University of Washington. Contact Dr. Paauw at [email protected].
References
1. J Am Board Fam Med. 2010 Jan-Feb;23(1):97-103.
2. South Med J. 1985 Sep;78(9):1065-7.
3. Drugs Real World Outcomes. 2015 Mar;2(1):29-33.
4. J Clin Gastroenterol. 1989 Oct;11(5):590-1.
5. Ann Fam Med. 2006 Sep-Oct;4(5):423-6.
6. BMJ Open. 2013 May 14;3(5).
A 46-year-old man comes to clinic for evaluation of night sweats. He has been having drenching night sweats for the past 3 months. He has to change his night shirt at least once per night. He has had a 10-pound weight gain over the past 6 months. No chest pain, nausea, or fatigue. He has had a cough for the past 6 months.
Which is the most likely diagnosis?
A. Gastroesophageal reflux disease.
B. Tuberculosis.
C. Lymphoma.
D. Multiple myeloma.
Night sweats are a common symptom in the general population, estimated to occur in about 10% of people. They can range in frequency and severity. We become most concerned when the patient is concerned, usually when they report drenching night sweats.
What do we need to know about this symptom to help us think of more likely causes and guide us in a more appropriate workup?
Night sweats do not seem to be a bad prognostic symptom. James W. Mold, MD, and his colleagues looked at the prognostic significance of night sweats in two cohorts of elderly patients.1 The prevalence of night sweats in this study was 10%. These two cohorts were followed for a little more than 7 years. More than 1,500 patients were included in the two cohorts. Patients who reported night sweats were not more likely to die, or die sooner, than were those who didn’t have night sweats. The severity of the night sweats did not make a difference.
Lea et al. described the prevalence of night sweats among different inpatient populations, with a range from 33% in surgical and medicine patients, to 60% on obstetrics service.2
Night sweats are common, and don’t appear to be correlated with worse prognosis. So, what are the likely common causes?
There just aren’t good studies on causes of night sweats, but there are studies that suggest that they are seen in some very common diseases. It is always good to look at medication lists as a start when evaluating unexplained symptoms.
Dr. Mold, along with Barbara J. Holtzclaw, PhD, reported higher odds ratios for night sweats for patients on SSRIs (OR, 3.01), angiotensin receptor blockers (OR, 3.44) and thyroid hormone supplements (OR, 2.53).3 W.A. Reynolds, MD, looked at the prevalence of night sweats in a GI practice.4 A total of 41% of the patients reported night sweats, and 12 of 12 patients with GERD who had night sweats had resolution of the night sweats with effective treatment of the GERD.
Dr. Mold and his colleagues found that night sweats were associated with several sleep-related symptoms, including waking up with a bitter taste in the mouth (OR, 1.94), daytime tiredness (OR, 1.99), and legs jerking during sleep (OR, 1.87).5
Erna Arnardottir, PhD, and her colleagues found that obstructive sleep apnea was associated with frequent nocturnal sweating.6 They found that 31% of men and 33% of women with OSA had nocturnal sweating, compared with about 10% of the general population. When the OSA patients were treated with positive airway pressure, the prevalence of nocturnal sweating decreased to 11.5%, similar to general population numbers.
Pearl: Night sweats are associated with common conditions: medications, GERD, and sleep disorders. These are more likely than lymphoma and tuberculosis.
Dr. Paauw is professor of medicine in the division of general internal medicine at the University of Washington, Seattle, and serves as third-year medical student clerkship director at the University of Washington. Contact Dr. Paauw at [email protected].
References
1. J Am Board Fam Med. 2010 Jan-Feb;23(1):97-103.
2. South Med J. 1985 Sep;78(9):1065-7.
3. Drugs Real World Outcomes. 2015 Mar;2(1):29-33.
4. J Clin Gastroenterol. 1989 Oct;11(5):590-1.
5. Ann Fam Med. 2006 Sep-Oct;4(5):423-6.
6. BMJ Open. 2013 May 14;3(5).
Intimate partner violence and PTSD increase menopausal symptom risk
Intimate partner violence or sexual assault may have a significant effect on menopausal symptoms in women, according to a cohort study published in JAMA Internal Medicine.
Researchers analyzed data from 2,016 women aged 40 years or older who were enrolled in the observational Reproductive Risks of Incontinence Study; 40% were non-Latina white, 21% were black, 20% were Latina or Hispanic, and 19% were Asian. Of this cohort, 21% had experienced emotional intimate partner violence (IPV) – 64 (3.2%) in the past 12 months – 16% had experienced physical IPV, 14% had experienced both, and 19% reported sexual assault. More than one in five women (23%) met the criteria for clinically significant PTSD.
Women who had experienced emotional domestic abuse were 36% more likely to report difficulty sleeping, 50% more like to experience night sweats, and 60% more likely to experience pain with intercourse, compared with women who had not experienced any abuse.
Physical abuse was associated with 33% higher odds of night sweats, and sexual assault was associated with 41% higher odds of vaginal dryness, 42% higher odds of vaginal irritation, and 44% higher odds of pain with intercourse.
Women with clinically significant PTSD symptoms were significantly more likely to experience all the symptoms of menopause, including twofold higher odds of pain with intercourse and threefold higher odds of difficulty sleeping. When authors accounted for the effect of PTSD symptoms in the cohort, they found that only the association between emotional abuse and night sweats or pain with intercourse, and between sexual assault and vaginal dryness, remained independently significant.
Carolyn J. Gibson, PhD, MPH, of the San Francisco Veterans Affairs Health Care System, and coauthors said that the biological and hormonal changes that underpin menopausal symptoms, as well as health risk behaviors, cardiometabolic risk factors, and other chronic health conditions associated with menopause, all are impacted by trauma and its psychological effects.
“Chronic hyperarousal and hypervigilance, common in individuals who have experienced trauma and characteristic symptoms of PTSD, may affect sleep and symptom sensitivity,” they wrote.
The reverse is also true; that the symptoms of menopause can impact the symptoms of PTSD by affecting a woman’s sense of self-efficacy, interpersonal engagements, and heighten the stress associated with this period of transition.
“The clinical management of menopause symptoms may also be enhanced by trauma-informed care, including recognition of challenges that may impair efforts to address menopause-related concerns among women affected by trauma,” the authors wrote.
Clinicians also could help by providing education about the link between trauma and health, providing their patients with a safe and supportive treatment environment, and facilitating referrals for psychological or trauma-specific services when needed, they said.
The research was supported by the San Francisco Veterans Affairs Medical Center and Kaiser Permanente Northern California, and funded by the University of California San Francisco–Kaiser Permanente Grants Program for Delivery Science, the Office of Research on Women’s Health Specialized Center of Research, and grants from the National Institute of Diabetes and Digestive and Kidney Diseases.
SOURCE: Gibson C et al. JAMA Intern Med. 2018 Nov 19. doi: 10.1001/jamainternmed.2018.5233.
An estimated 33% of women in the United States have been sexually assaulted, and an estimated 25% have experienced IPV, so be aware of how common this “wicked problem” is, the way it impacts health, and what role you can play in educating and helping patients by connecting them to available resources.
But that is not enough. Consider measures such as training yourself and staff in how to assess for IPV and sexual assault and use of EHR to integrate IPV assessment into routine clinical care, as well as developing protocols to be followed when a patient discloses IPV or sexual assault. A multidisciplinary approach also can help, including victim service advocates and behavioral health clinicians to provide care and support.
State requirements for reporting partner and sexual violence differ, so be aware of your state laws.
A strength of this study is that it included emotional as well as physical IPV, which often is left out although it has serious impacts.
Rebecca C. Thurston, PhD, is from the department of psychiatry at the University of Pittsburgh, and Elizabeth Miller, MD, PhD, is from the division of adolescent and young adult medicine at the UPMC Children’s Hospital of Pittsburgh. These comments were taken from an accompanying editorial (JAMA Intern Med. 2018 Nov 19. doi: 10.1001/jamainternmed.2018.5242). Dr. Thurston declared research support from the National Institutes of Health and consultancies for Pfizer, Procter & Gamble, and MAS Innovations.
An estimated 33% of women in the United States have been sexually assaulted, and an estimated 25% have experienced IPV, so be aware of how common this “wicked problem” is, the way it impacts health, and what role you can play in educating and helping patients by connecting them to available resources.
But that is not enough. Consider measures such as training yourself and staff in how to assess for IPV and sexual assault and use of EHR to integrate IPV assessment into routine clinical care, as well as developing protocols to be followed when a patient discloses IPV or sexual assault. A multidisciplinary approach also can help, including victim service advocates and behavioral health clinicians to provide care and support.
State requirements for reporting partner and sexual violence differ, so be aware of your state laws.
A strength of this study is that it included emotional as well as physical IPV, which often is left out although it has serious impacts.
Rebecca C. Thurston, PhD, is from the department of psychiatry at the University of Pittsburgh, and Elizabeth Miller, MD, PhD, is from the division of adolescent and young adult medicine at the UPMC Children’s Hospital of Pittsburgh. These comments were taken from an accompanying editorial (JAMA Intern Med. 2018 Nov 19. doi: 10.1001/jamainternmed.2018.5242). Dr. Thurston declared research support from the National Institutes of Health and consultancies for Pfizer, Procter & Gamble, and MAS Innovations.
An estimated 33% of women in the United States have been sexually assaulted, and an estimated 25% have experienced IPV, so be aware of how common this “wicked problem” is, the way it impacts health, and what role you can play in educating and helping patients by connecting them to available resources.
But that is not enough. Consider measures such as training yourself and staff in how to assess for IPV and sexual assault and use of EHR to integrate IPV assessment into routine clinical care, as well as developing protocols to be followed when a patient discloses IPV or sexual assault. A multidisciplinary approach also can help, including victim service advocates and behavioral health clinicians to provide care and support.
State requirements for reporting partner and sexual violence differ, so be aware of your state laws.
A strength of this study is that it included emotional as well as physical IPV, which often is left out although it has serious impacts.
Rebecca C. Thurston, PhD, is from the department of psychiatry at the University of Pittsburgh, and Elizabeth Miller, MD, PhD, is from the division of adolescent and young adult medicine at the UPMC Children’s Hospital of Pittsburgh. These comments were taken from an accompanying editorial (JAMA Intern Med. 2018 Nov 19. doi: 10.1001/jamainternmed.2018.5242). Dr. Thurston declared research support from the National Institutes of Health and consultancies for Pfizer, Procter & Gamble, and MAS Innovations.
Intimate partner violence or sexual assault may have a significant effect on menopausal symptoms in women, according to a cohort study published in JAMA Internal Medicine.
Researchers analyzed data from 2,016 women aged 40 years or older who were enrolled in the observational Reproductive Risks of Incontinence Study; 40% were non-Latina white, 21% were black, 20% were Latina or Hispanic, and 19% were Asian. Of this cohort, 21% had experienced emotional intimate partner violence (IPV) – 64 (3.2%) in the past 12 months – 16% had experienced physical IPV, 14% had experienced both, and 19% reported sexual assault. More than one in five women (23%) met the criteria for clinically significant PTSD.
Women who had experienced emotional domestic abuse were 36% more likely to report difficulty sleeping, 50% more like to experience night sweats, and 60% more likely to experience pain with intercourse, compared with women who had not experienced any abuse.
Physical abuse was associated with 33% higher odds of night sweats, and sexual assault was associated with 41% higher odds of vaginal dryness, 42% higher odds of vaginal irritation, and 44% higher odds of pain with intercourse.
Women with clinically significant PTSD symptoms were significantly more likely to experience all the symptoms of menopause, including twofold higher odds of pain with intercourse and threefold higher odds of difficulty sleeping. When authors accounted for the effect of PTSD symptoms in the cohort, they found that only the association between emotional abuse and night sweats or pain with intercourse, and between sexual assault and vaginal dryness, remained independently significant.
Carolyn J. Gibson, PhD, MPH, of the San Francisco Veterans Affairs Health Care System, and coauthors said that the biological and hormonal changes that underpin menopausal symptoms, as well as health risk behaviors, cardiometabolic risk factors, and other chronic health conditions associated with menopause, all are impacted by trauma and its psychological effects.
“Chronic hyperarousal and hypervigilance, common in individuals who have experienced trauma and characteristic symptoms of PTSD, may affect sleep and symptom sensitivity,” they wrote.
The reverse is also true; that the symptoms of menopause can impact the symptoms of PTSD by affecting a woman’s sense of self-efficacy, interpersonal engagements, and heighten the stress associated with this period of transition.
“The clinical management of menopause symptoms may also be enhanced by trauma-informed care, including recognition of challenges that may impair efforts to address menopause-related concerns among women affected by trauma,” the authors wrote.
Clinicians also could help by providing education about the link between trauma and health, providing their patients with a safe and supportive treatment environment, and facilitating referrals for psychological or trauma-specific services when needed, they said.
The research was supported by the San Francisco Veterans Affairs Medical Center and Kaiser Permanente Northern California, and funded by the University of California San Francisco–Kaiser Permanente Grants Program for Delivery Science, the Office of Research on Women’s Health Specialized Center of Research, and grants from the National Institute of Diabetes and Digestive and Kidney Diseases.
SOURCE: Gibson C et al. JAMA Intern Med. 2018 Nov 19. doi: 10.1001/jamainternmed.2018.5233.
Intimate partner violence or sexual assault may have a significant effect on menopausal symptoms in women, according to a cohort study published in JAMA Internal Medicine.
Researchers analyzed data from 2,016 women aged 40 years or older who were enrolled in the observational Reproductive Risks of Incontinence Study; 40% were non-Latina white, 21% were black, 20% were Latina or Hispanic, and 19% were Asian. Of this cohort, 21% had experienced emotional intimate partner violence (IPV) – 64 (3.2%) in the past 12 months – 16% had experienced physical IPV, 14% had experienced both, and 19% reported sexual assault. More than one in five women (23%) met the criteria for clinically significant PTSD.
Women who had experienced emotional domestic abuse were 36% more likely to report difficulty sleeping, 50% more like to experience night sweats, and 60% more likely to experience pain with intercourse, compared with women who had not experienced any abuse.
Physical abuse was associated with 33% higher odds of night sweats, and sexual assault was associated with 41% higher odds of vaginal dryness, 42% higher odds of vaginal irritation, and 44% higher odds of pain with intercourse.
Women with clinically significant PTSD symptoms were significantly more likely to experience all the symptoms of menopause, including twofold higher odds of pain with intercourse and threefold higher odds of difficulty sleeping. When authors accounted for the effect of PTSD symptoms in the cohort, they found that only the association between emotional abuse and night sweats or pain with intercourse, and between sexual assault and vaginal dryness, remained independently significant.
Carolyn J. Gibson, PhD, MPH, of the San Francisco Veterans Affairs Health Care System, and coauthors said that the biological and hormonal changes that underpin menopausal symptoms, as well as health risk behaviors, cardiometabolic risk factors, and other chronic health conditions associated with menopause, all are impacted by trauma and its psychological effects.
“Chronic hyperarousal and hypervigilance, common in individuals who have experienced trauma and characteristic symptoms of PTSD, may affect sleep and symptom sensitivity,” they wrote.
The reverse is also true; that the symptoms of menopause can impact the symptoms of PTSD by affecting a woman’s sense of self-efficacy, interpersonal engagements, and heighten the stress associated with this period of transition.
“The clinical management of menopause symptoms may also be enhanced by trauma-informed care, including recognition of challenges that may impair efforts to address menopause-related concerns among women affected by trauma,” the authors wrote.
Clinicians also could help by providing education about the link between trauma and health, providing their patients with a safe and supportive treatment environment, and facilitating referrals for psychological or trauma-specific services when needed, they said.
The research was supported by the San Francisco Veterans Affairs Medical Center and Kaiser Permanente Northern California, and funded by the University of California San Francisco–Kaiser Permanente Grants Program for Delivery Science, the Office of Research on Women’s Health Specialized Center of Research, and grants from the National Institute of Diabetes and Digestive and Kidney Diseases.
SOURCE: Gibson C et al. JAMA Intern Med. 2018 Nov 19. doi: 10.1001/jamainternmed.2018.5233.
FROM JAMA INTERNAL MEDICINE
Key clinical point: Intimate partner violence increases the risk of menopausal symptoms.
Major finding:
Study details: A cohort study in 2,016 women aged 40 years and older.
Disclosures: The research was supported by the San Francisco Veterans Affairs Medical Center and Kaiser Permanente Northern California, and funded by the University of California San Francisco–Kaiser Permanente Grants Program for Delivery Science, the Office of Research on Women’s Health Specialized Center of Research, and grants from the National Institute of Diabetes and Digestive and Kidney Diseases.
Source: Gibson C et al. JAMA Intern Med. 2018 Nov 19. doi:10.1001/jamainternmed.2018.5233.
The link between suicide and sleep
According to the Centers for Disease Control and Prevention, suicide is the 10th leading cause of mortality in the United States, with rates of suicide rising over the past 2 decades. In 2016, completed suicides accounted for approximately 45,000 deaths in the United States (Ivey-Stephenson AZ, et al. MMWR Surveill Summ. 2017;66[18]:1). While progress has been made to lower mortality rates of other leading causes of death, very little progress has been made on reducing the rates of suicide. The term “suicide,” as referred to in this article, encompasses suicidal ideation, suicidal behavior, and suicide death.
Researchers have been investigating potential risk factors and prevention strategies for suicide. The relationship between suicide and sleep disturbances, specifically insomnia and nightmares, has been well documented in the literature. Given that insomnia and nightmares are potentially modifiable risk factors, it continues to be an area of active exploration for suicide rate reduction. While there are many different types of sleep disorders, including excessive daytime sleepiness, parasomnias, obstructive sleep apnea, and restless legs syndrome, this article will focus on the relationship between insomnia and nightmares with suicide.
Insomnia
Insomnia disorder, according to the American Psychiatric Association’s DSM-5, is a dissatisfaction of sleep quantity or quality that occurs at least three nights per week for a minimum of 3 months despite adequate opportunity for sleep. This may present as difficulty with falling asleep, staying asleep, or early morning awakenings. The sleep disturbance results in functional impairment or significant distress in at least one area of life (American Psychiatric Association. Arlington, Virginia: APA; 2013). While insomnia is often a symptom of many psychiatric disorders, research has shown that insomnia is an independent risk factor for suicide, even when controlling for mental illness. Studies have shown that there is up to a 2.4 relative risk of suicide death with insomnia after adjusting for depression severity (McCall W, et al. J Clin Sleep Med. 2013;32[9]:135).
Nightmares
Nightmares, as defined by the American Psychiatric Association’s DSM-5, are “typically lengthy, elaborate, story-like sequences of dream imagery that seem real and incite anxiety, fear, or other dysphoric emotions” (American Psychiatric Association. Arlington, Virginia: APA; 2013). They are common symptoms in posttraumatic stress disorder (PTSD), with up to 90% of individuals with PTSD experiencing nightmares following a traumatic event (Littlewood DL, et al. J Clin Sleep Med. 2016;12[3]:393). Nightmares have also been shown to be an independent risk factor for suicide when controlling for mental illness. Studies have shown that nightmares are associated with an elevated risk factor of 1.5 to 3 times for suicidal ideation and 3 to 4 times for suicide attempts. The data suggest that nightmares may be a stronger risk factor for suicide than insomnia (McCall W, et al. Curr Psychiatr Rep. 2013;15[9]:389).
Proposed Mechanism
The mechanism linking insomnia and nightmares with suicide has been theorized and studied by researchers. A couple of the most noteworthy proposed psychological mechanisms involve dysfunctional beliefs and attitudes about sleep, as well as deficits in problem solving capability. Dysfunctional beliefs and attitudes about sleep (DBAS) are negative cognitions pertaining to sleep, and they have been shown to be related to the intensity of suicidal ideations. Many of the DBAS are pessimistic thoughts that contain a “hopelessness flavor” to them, which lead to the perpetuation of insomnia. Hopelessness has been found to be a strong risk factor for suicide. In addition to DBAS, insomnia has also shown to lead to impairments in complex problem solving. The lack of problem solving skills in these patients may lead to fewer quantity and quality of solutions during stressful situations and leave suicide as the perceived best or only option.
The biological theories focus on serotonin and hyperarousal mediated by the hypothalamic-pituitary-adrenal (HPA) axis. Serotonin is a neurotransmitter that is involved in the induction and maintenance of sleep. Of interesting note, low levels of serotonin’s main metabolite, 5-hydroxyindoleacetic acid (5-HIAA) have been found in the cerebrospinal fluid of suicide victims. Evidence has also shown that sleep and the HPA axis are closely related. The HPA axis is activated by stress leading to a cascade of hormones that can cause susceptibility of hyperarousal, REM alterations, and suicide. Hyperarousal, shared in context with PTSD and insomnia, can lead to hyperactivation of the noradrenergic systems in the medial prefrontal cortex, which can lead to decrease in executive decision making (McCall W, et al. Curr Psychiatr Rep. 2013;15[9]:389).
Treatment Strategies
The benefit of treating insomnia and nightmares, in regards to reducing suicidality, continues to be an area of active research. Many of the previous studies have theorized that treating symptoms of insomnia and nightmares may indirectly reduce suicide. Pharmaceutical and nonpharmaceutical treatments are currently being used to help treat patients with insomnia and nightmares, but the benefit for reducing suicidality is still unknown.
One of the main treatment modalities for insomnia is hypnotic medication; however, these medications carry their own potential risk for suicide. Reports of suicide death in conjunction with hypnotic medication has led the FDA to add warnings about the increased risk of suicide with these medications. Some of these medications include zolpidem, zaleplon, eszopiclone, doxepin, ramelteon, and suvorexant. A review of research studies and case reports was completed in 2017 and showed that there was an odds ratio of 2 to 3 for hypnotic use in suicide deaths. However, most of the studies that were reviewed reported a potential confounding bias of the individual’s current mental health state. Furthermore, many of the suicide case reports that involved hypnotics also had additional substances detected, such as alcohol. Hypnotic medication has been shown to be an effective treatment for insomnia, but caution needs to be used when prescribing these medications. Strategies that may be beneficial when using hypnotic medication to reduce the risk of an adverse outcome include using the lowest effective dose and educating the patient of not combining the medication with alcohol or other sedative/hypnotics (McCall W, et al. Am J Psychiatry. 2017;174[1]:18).
For patients who have recurrent nightmares in the context of PTSD, the alpha-1 adrenergic receptor antagonist, prazosin, may provide some benefit; however, the literature is divided. There have been several randomized, placebo-controlled clinical trials with prazosin, which has shown a moderate to large effect for alleviating trauma-related nightmares and improving sleep quality. Some of the limitations of these studies were that the trials were small to moderate in size, and the length of the trials was 15 weeks or less. In 2018, Raskin and colleagues completed a follow-up randomized, placebo-controlled study for 26 weeks with 304 participants and did not find a significant difference between prazosin and placebo in regards to nightmares and sleep quality (Raskind MA, et al. N Engl J Med. 2018;378[6]:507).
Cognitive behavioral therapy for insomnia (CBT-I) and image rehearsal therapy (IRT) are two sleep-targeted therapy modalities that are evidence based. CBT-I targets dysfunctional beliefs and attitudes regarding sleep (McCall W, et al. J Clin Sleep Med. 2013;9[2]:135). IRT, on the other hand, specifically targets nightmares by having the patient write out a narrative of the nightmare, followed by re-scripting an alternative ending to something that is less distressing. The patient will rehearse the new dream narrative before going to sleep. There is still insufficient evidence to determine if these therapies have benefit in reducing suicide (Littlewood DL, et al. J Clin Sleep Med. 2016;12[3]:393).
While the jury is still out on how best to target and treat the risk factors of insomnia and nightmares in regards to suicide, there are still steps that health-care providers can take to help keep their patients safe. During the patient interview, new or worsening insomnia and nightmares should prompt further investigation of suicidal thoughts and behaviors. After a thorough interview, treatment options, with a discussion of risks and benefits, can be tailored to the individual’s needs. Managing insomnia and nightmares may be one avenue of suicide prevention.
Drs. Locrotondo and McCall are with the Department of Psychiatry and Health Behavior at the Medical College of Georgia, Augusta University, Augusta, Georgia.
According to the Centers for Disease Control and Prevention, suicide is the 10th leading cause of mortality in the United States, with rates of suicide rising over the past 2 decades. In 2016, completed suicides accounted for approximately 45,000 deaths in the United States (Ivey-Stephenson AZ, et al. MMWR Surveill Summ. 2017;66[18]:1). While progress has been made to lower mortality rates of other leading causes of death, very little progress has been made on reducing the rates of suicide. The term “suicide,” as referred to in this article, encompasses suicidal ideation, suicidal behavior, and suicide death.
Researchers have been investigating potential risk factors and prevention strategies for suicide. The relationship between suicide and sleep disturbances, specifically insomnia and nightmares, has been well documented in the literature. Given that insomnia and nightmares are potentially modifiable risk factors, it continues to be an area of active exploration for suicide rate reduction. While there are many different types of sleep disorders, including excessive daytime sleepiness, parasomnias, obstructive sleep apnea, and restless legs syndrome, this article will focus on the relationship between insomnia and nightmares with suicide.
Insomnia
Insomnia disorder, according to the American Psychiatric Association’s DSM-5, is a dissatisfaction of sleep quantity or quality that occurs at least three nights per week for a minimum of 3 months despite adequate opportunity for sleep. This may present as difficulty with falling asleep, staying asleep, or early morning awakenings. The sleep disturbance results in functional impairment or significant distress in at least one area of life (American Psychiatric Association. Arlington, Virginia: APA; 2013). While insomnia is often a symptom of many psychiatric disorders, research has shown that insomnia is an independent risk factor for suicide, even when controlling for mental illness. Studies have shown that there is up to a 2.4 relative risk of suicide death with insomnia after adjusting for depression severity (McCall W, et al. J Clin Sleep Med. 2013;32[9]:135).
Nightmares
Nightmares, as defined by the American Psychiatric Association’s DSM-5, are “typically lengthy, elaborate, story-like sequences of dream imagery that seem real and incite anxiety, fear, or other dysphoric emotions” (American Psychiatric Association. Arlington, Virginia: APA; 2013). They are common symptoms in posttraumatic stress disorder (PTSD), with up to 90% of individuals with PTSD experiencing nightmares following a traumatic event (Littlewood DL, et al. J Clin Sleep Med. 2016;12[3]:393). Nightmares have also been shown to be an independent risk factor for suicide when controlling for mental illness. Studies have shown that nightmares are associated with an elevated risk factor of 1.5 to 3 times for suicidal ideation and 3 to 4 times for suicide attempts. The data suggest that nightmares may be a stronger risk factor for suicide than insomnia (McCall W, et al. Curr Psychiatr Rep. 2013;15[9]:389).
Proposed Mechanism
The mechanism linking insomnia and nightmares with suicide has been theorized and studied by researchers. A couple of the most noteworthy proposed psychological mechanisms involve dysfunctional beliefs and attitudes about sleep, as well as deficits in problem solving capability. Dysfunctional beliefs and attitudes about sleep (DBAS) are negative cognitions pertaining to sleep, and they have been shown to be related to the intensity of suicidal ideations. Many of the DBAS are pessimistic thoughts that contain a “hopelessness flavor” to them, which lead to the perpetuation of insomnia. Hopelessness has been found to be a strong risk factor for suicide. In addition to DBAS, insomnia has also shown to lead to impairments in complex problem solving. The lack of problem solving skills in these patients may lead to fewer quantity and quality of solutions during stressful situations and leave suicide as the perceived best or only option.
The biological theories focus on serotonin and hyperarousal mediated by the hypothalamic-pituitary-adrenal (HPA) axis. Serotonin is a neurotransmitter that is involved in the induction and maintenance of sleep. Of interesting note, low levels of serotonin’s main metabolite, 5-hydroxyindoleacetic acid (5-HIAA) have been found in the cerebrospinal fluid of suicide victims. Evidence has also shown that sleep and the HPA axis are closely related. The HPA axis is activated by stress leading to a cascade of hormones that can cause susceptibility of hyperarousal, REM alterations, and suicide. Hyperarousal, shared in context with PTSD and insomnia, can lead to hyperactivation of the noradrenergic systems in the medial prefrontal cortex, which can lead to decrease in executive decision making (McCall W, et al. Curr Psychiatr Rep. 2013;15[9]:389).
Treatment Strategies
The benefit of treating insomnia and nightmares, in regards to reducing suicidality, continues to be an area of active research. Many of the previous studies have theorized that treating symptoms of insomnia and nightmares may indirectly reduce suicide. Pharmaceutical and nonpharmaceutical treatments are currently being used to help treat patients with insomnia and nightmares, but the benefit for reducing suicidality is still unknown.
One of the main treatment modalities for insomnia is hypnotic medication; however, these medications carry their own potential risk for suicide. Reports of suicide death in conjunction with hypnotic medication has led the FDA to add warnings about the increased risk of suicide with these medications. Some of these medications include zolpidem, zaleplon, eszopiclone, doxepin, ramelteon, and suvorexant. A review of research studies and case reports was completed in 2017 and showed that there was an odds ratio of 2 to 3 for hypnotic use in suicide deaths. However, most of the studies that were reviewed reported a potential confounding bias of the individual’s current mental health state. Furthermore, many of the suicide case reports that involved hypnotics also had additional substances detected, such as alcohol. Hypnotic medication has been shown to be an effective treatment for insomnia, but caution needs to be used when prescribing these medications. Strategies that may be beneficial when using hypnotic medication to reduce the risk of an adverse outcome include using the lowest effective dose and educating the patient of not combining the medication with alcohol or other sedative/hypnotics (McCall W, et al. Am J Psychiatry. 2017;174[1]:18).
For patients who have recurrent nightmares in the context of PTSD, the alpha-1 adrenergic receptor antagonist, prazosin, may provide some benefit; however, the literature is divided. There have been several randomized, placebo-controlled clinical trials with prazosin, which has shown a moderate to large effect for alleviating trauma-related nightmares and improving sleep quality. Some of the limitations of these studies were that the trials were small to moderate in size, and the length of the trials was 15 weeks or less. In 2018, Raskin and colleagues completed a follow-up randomized, placebo-controlled study for 26 weeks with 304 participants and did not find a significant difference between prazosin and placebo in regards to nightmares and sleep quality (Raskind MA, et al. N Engl J Med. 2018;378[6]:507).
Cognitive behavioral therapy for insomnia (CBT-I) and image rehearsal therapy (IRT) are two sleep-targeted therapy modalities that are evidence based. CBT-I targets dysfunctional beliefs and attitudes regarding sleep (McCall W, et al. J Clin Sleep Med. 2013;9[2]:135). IRT, on the other hand, specifically targets nightmares by having the patient write out a narrative of the nightmare, followed by re-scripting an alternative ending to something that is less distressing. The patient will rehearse the new dream narrative before going to sleep. There is still insufficient evidence to determine if these therapies have benefit in reducing suicide (Littlewood DL, et al. J Clin Sleep Med. 2016;12[3]:393).
While the jury is still out on how best to target and treat the risk factors of insomnia and nightmares in regards to suicide, there are still steps that health-care providers can take to help keep their patients safe. During the patient interview, new or worsening insomnia and nightmares should prompt further investigation of suicidal thoughts and behaviors. After a thorough interview, treatment options, with a discussion of risks and benefits, can be tailored to the individual’s needs. Managing insomnia and nightmares may be one avenue of suicide prevention.
Drs. Locrotondo and McCall are with the Department of Psychiatry and Health Behavior at the Medical College of Georgia, Augusta University, Augusta, Georgia.
According to the Centers for Disease Control and Prevention, suicide is the 10th leading cause of mortality in the United States, with rates of suicide rising over the past 2 decades. In 2016, completed suicides accounted for approximately 45,000 deaths in the United States (Ivey-Stephenson AZ, et al. MMWR Surveill Summ. 2017;66[18]:1). While progress has been made to lower mortality rates of other leading causes of death, very little progress has been made on reducing the rates of suicide. The term “suicide,” as referred to in this article, encompasses suicidal ideation, suicidal behavior, and suicide death.
Researchers have been investigating potential risk factors and prevention strategies for suicide. The relationship between suicide and sleep disturbances, specifically insomnia and nightmares, has been well documented in the literature. Given that insomnia and nightmares are potentially modifiable risk factors, it continues to be an area of active exploration for suicide rate reduction. While there are many different types of sleep disorders, including excessive daytime sleepiness, parasomnias, obstructive sleep apnea, and restless legs syndrome, this article will focus on the relationship between insomnia and nightmares with suicide.
Insomnia
Insomnia disorder, according to the American Psychiatric Association’s DSM-5, is a dissatisfaction of sleep quantity or quality that occurs at least three nights per week for a minimum of 3 months despite adequate opportunity for sleep. This may present as difficulty with falling asleep, staying asleep, or early morning awakenings. The sleep disturbance results in functional impairment or significant distress in at least one area of life (American Psychiatric Association. Arlington, Virginia: APA; 2013). While insomnia is often a symptom of many psychiatric disorders, research has shown that insomnia is an independent risk factor for suicide, even when controlling for mental illness. Studies have shown that there is up to a 2.4 relative risk of suicide death with insomnia after adjusting for depression severity (McCall W, et al. J Clin Sleep Med. 2013;32[9]:135).
Nightmares
Nightmares, as defined by the American Psychiatric Association’s DSM-5, are “typically lengthy, elaborate, story-like sequences of dream imagery that seem real and incite anxiety, fear, or other dysphoric emotions” (American Psychiatric Association. Arlington, Virginia: APA; 2013). They are common symptoms in posttraumatic stress disorder (PTSD), with up to 90% of individuals with PTSD experiencing nightmares following a traumatic event (Littlewood DL, et al. J Clin Sleep Med. 2016;12[3]:393). Nightmares have also been shown to be an independent risk factor for suicide when controlling for mental illness. Studies have shown that nightmares are associated with an elevated risk factor of 1.5 to 3 times for suicidal ideation and 3 to 4 times for suicide attempts. The data suggest that nightmares may be a stronger risk factor for suicide than insomnia (McCall W, et al. Curr Psychiatr Rep. 2013;15[9]:389).
Proposed Mechanism
The mechanism linking insomnia and nightmares with suicide has been theorized and studied by researchers. A couple of the most noteworthy proposed psychological mechanisms involve dysfunctional beliefs and attitudes about sleep, as well as deficits in problem solving capability. Dysfunctional beliefs and attitudes about sleep (DBAS) are negative cognitions pertaining to sleep, and they have been shown to be related to the intensity of suicidal ideations. Many of the DBAS are pessimistic thoughts that contain a “hopelessness flavor” to them, which lead to the perpetuation of insomnia. Hopelessness has been found to be a strong risk factor for suicide. In addition to DBAS, insomnia has also shown to lead to impairments in complex problem solving. The lack of problem solving skills in these patients may lead to fewer quantity and quality of solutions during stressful situations and leave suicide as the perceived best or only option.
The biological theories focus on serotonin and hyperarousal mediated by the hypothalamic-pituitary-adrenal (HPA) axis. Serotonin is a neurotransmitter that is involved in the induction and maintenance of sleep. Of interesting note, low levels of serotonin’s main metabolite, 5-hydroxyindoleacetic acid (5-HIAA) have been found in the cerebrospinal fluid of suicide victims. Evidence has also shown that sleep and the HPA axis are closely related. The HPA axis is activated by stress leading to a cascade of hormones that can cause susceptibility of hyperarousal, REM alterations, and suicide. Hyperarousal, shared in context with PTSD and insomnia, can lead to hyperactivation of the noradrenergic systems in the medial prefrontal cortex, which can lead to decrease in executive decision making (McCall W, et al. Curr Psychiatr Rep. 2013;15[9]:389).
Treatment Strategies
The benefit of treating insomnia and nightmares, in regards to reducing suicidality, continues to be an area of active research. Many of the previous studies have theorized that treating symptoms of insomnia and nightmares may indirectly reduce suicide. Pharmaceutical and nonpharmaceutical treatments are currently being used to help treat patients with insomnia and nightmares, but the benefit for reducing suicidality is still unknown.
One of the main treatment modalities for insomnia is hypnotic medication; however, these medications carry their own potential risk for suicide. Reports of suicide death in conjunction with hypnotic medication has led the FDA to add warnings about the increased risk of suicide with these medications. Some of these medications include zolpidem, zaleplon, eszopiclone, doxepin, ramelteon, and suvorexant. A review of research studies and case reports was completed in 2017 and showed that there was an odds ratio of 2 to 3 for hypnotic use in suicide deaths. However, most of the studies that were reviewed reported a potential confounding bias of the individual’s current mental health state. Furthermore, many of the suicide case reports that involved hypnotics also had additional substances detected, such as alcohol. Hypnotic medication has been shown to be an effective treatment for insomnia, but caution needs to be used when prescribing these medications. Strategies that may be beneficial when using hypnotic medication to reduce the risk of an adverse outcome include using the lowest effective dose and educating the patient of not combining the medication with alcohol or other sedative/hypnotics (McCall W, et al. Am J Psychiatry. 2017;174[1]:18).
For patients who have recurrent nightmares in the context of PTSD, the alpha-1 adrenergic receptor antagonist, prazosin, may provide some benefit; however, the literature is divided. There have been several randomized, placebo-controlled clinical trials with prazosin, which has shown a moderate to large effect for alleviating trauma-related nightmares and improving sleep quality. Some of the limitations of these studies were that the trials were small to moderate in size, and the length of the trials was 15 weeks or less. In 2018, Raskin and colleagues completed a follow-up randomized, placebo-controlled study for 26 weeks with 304 participants and did not find a significant difference between prazosin and placebo in regards to nightmares and sleep quality (Raskind MA, et al. N Engl J Med. 2018;378[6]:507).
Cognitive behavioral therapy for insomnia (CBT-I) and image rehearsal therapy (IRT) are two sleep-targeted therapy modalities that are evidence based. CBT-I targets dysfunctional beliefs and attitudes regarding sleep (McCall W, et al. J Clin Sleep Med. 2013;9[2]:135). IRT, on the other hand, specifically targets nightmares by having the patient write out a narrative of the nightmare, followed by re-scripting an alternative ending to something that is less distressing. The patient will rehearse the new dream narrative before going to sleep. There is still insufficient evidence to determine if these therapies have benefit in reducing suicide (Littlewood DL, et al. J Clin Sleep Med. 2016;12[3]:393).
While the jury is still out on how best to target and treat the risk factors of insomnia and nightmares in regards to suicide, there are still steps that health-care providers can take to help keep their patients safe. During the patient interview, new or worsening insomnia and nightmares should prompt further investigation of suicidal thoughts and behaviors. After a thorough interview, treatment options, with a discussion of risks and benefits, can be tailored to the individual’s needs. Managing insomnia and nightmares may be one avenue of suicide prevention.
Drs. Locrotondo and McCall are with the Department of Psychiatry and Health Behavior at the Medical College of Georgia, Augusta University, Augusta, Georgia.
Ask depressed patients about hypersomnia to screen for mixicity
BARCELONA – Hypersomnia is a novel, previously unappreciated factor useful in tipping the balance in favor of an underlying bipolar predisposition in patients with an acute major depressive episode, Andrea Murru, MD, PhD, reported at the annual congress of the European College of Neuropsychopharmacology.
“This may help us in clinical practice. It’s an effective, costless, and highly objective clinical measure. It’s one question, and it takes a second. It’s simply asking the patient: ‘Are you sleeping more hours at night than usual?’ It’s a very simple clinical question that could really change the focus of treatment for a patient,” said Dr. Murru, a clinical psychiatrist in the bipolar disorders program of the University of Barcelona.
He presented a post hoc analysis of 2,514 acutely depressed individuals who participated in the BRIDGE-II-MIX (Bipolar Disorders: Improving Diagnosis, Guidance and Education) study, an international, multicenter, cross-sectional, observational study aimed at better characterizing clinically valid mixed features of depression indicative of concurrent manic symptoms.
“This is one of a whole series of hypothesis-generating studies from BRIDGE-II-MIX that are trying to deal with the struggle of understanding whether all the elements that favor mixicity and an underpinning bipolar diathesis are fairly represented in the diagnostic criteria in DSM-5. And what we are basically finding is the DSM-5 criteria are leaving out important symptoms that really do play a role,” the psychiatrist said in an interview.
One of those missing factors, he continued, is hypersomnia. It was present in 16.8% of the study population, and he and his coinvestigators compared them in terms of clinical variables, current and past psychiatric symptoms, and sociodemographics with the 83.2% of patients with insomnia. That is, patients who got fewer hours of sleep than normal and felt fatigued during the next day were compared with those who felt a reduced need to sleep.
The two groups differed in important ways. Hypersomnia showed a powerful correlation with a physician diagnosis of major depressive episode with atypical features, being present in 32.2% of such patients, while a mere 1.8% had insomnia. Moreover, among patients diagnosed with bipolar disorder I or II, 20.6% reported hypersomnia, a significantly higher proportion than the 16% who had insomnia.
The finding that only 5% of BRIDGE-II-MIX participants with hypersomnia met DSM-5 criteria for a mixed-state specifier, a rate not significantly different from the 8.3% figure in those with insomnia, underscores the drawbacks of the DSM-5 criteria, according to Dr. Murru. He noted that, in contrast to the DSM-5 criteria, 32.9% of the hypersomniac patients with a major depressive episode met Research Diagnostic Criteria (RDC) for a mixed-state specifier, a rate significantly higher than the 27.6% figure in patients with insomnia.
Specifically, the individual RDC mixed-state specifiers that stood out as significantly more frequent among depressed patients with hypersomnia than insomnia were racing thoughts, by a margin of 15.1% to 10.6%; impulsivity, 16.8% versus 13.2%; distractibility, 29.6% versus 23.4%; hypersexuality, which was present in 4% of patients with hypersomnia but only 2.3% with insomnia; irritable mood, 33.1% versus 24.8%; and a history of insufficient response to previous antidepressant therapy, 34.3%, compared with 27.1% in insomniacs.
When Dr. Murru and his coinvestigators performed a stepwise linear regression analysis to identify significant predictors of hypersomnia in patients with a major depressive episode, they found that the sleep abnormality keeps some interesting company. Patients with current bulimia were 4.21-fold more likely to have hypersomnia than those without the eating disorder. Current social phobia was associated with a 1.77-fold increased risk of hypersomnia; mood lability on prior antidepressant therapy carried a 1.37-fold risk, as did current mood lability; prior attempted suicide was associated with a 1.31-fold increased risk; being overweight or obese was associated with a 1.42-fold risk; currently being on a mood stabilizer carried a 1.33-fold increased risk of hypersomnia; and currently being on an atypical antipsychotic agent had a 1.36-fold greater risk.
Dr. Murru concluded that the take-home message of this study – “Of course, conceding it’s highly exploratory nature intrinsic to a post hoc analysis,” he noted – is that hypersomnia should be included among the symptoms that trigger the “with mixed features” specifier in patients with a major depressive episode.
The BRIDGE-II-MIX study was sponsored by Sanofi-Aventis. Dr. Murru reported having no financial conflicts of interest regarding the study.
BARCELONA – Hypersomnia is a novel, previously unappreciated factor useful in tipping the balance in favor of an underlying bipolar predisposition in patients with an acute major depressive episode, Andrea Murru, MD, PhD, reported at the annual congress of the European College of Neuropsychopharmacology.
“This may help us in clinical practice. It’s an effective, costless, and highly objective clinical measure. It’s one question, and it takes a second. It’s simply asking the patient: ‘Are you sleeping more hours at night than usual?’ It’s a very simple clinical question that could really change the focus of treatment for a patient,” said Dr. Murru, a clinical psychiatrist in the bipolar disorders program of the University of Barcelona.
He presented a post hoc analysis of 2,514 acutely depressed individuals who participated in the BRIDGE-II-MIX (Bipolar Disorders: Improving Diagnosis, Guidance and Education) study, an international, multicenter, cross-sectional, observational study aimed at better characterizing clinically valid mixed features of depression indicative of concurrent manic symptoms.
“This is one of a whole series of hypothesis-generating studies from BRIDGE-II-MIX that are trying to deal with the struggle of understanding whether all the elements that favor mixicity and an underpinning bipolar diathesis are fairly represented in the diagnostic criteria in DSM-5. And what we are basically finding is the DSM-5 criteria are leaving out important symptoms that really do play a role,” the psychiatrist said in an interview.
One of those missing factors, he continued, is hypersomnia. It was present in 16.8% of the study population, and he and his coinvestigators compared them in terms of clinical variables, current and past psychiatric symptoms, and sociodemographics with the 83.2% of patients with insomnia. That is, patients who got fewer hours of sleep than normal and felt fatigued during the next day were compared with those who felt a reduced need to sleep.
The two groups differed in important ways. Hypersomnia showed a powerful correlation with a physician diagnosis of major depressive episode with atypical features, being present in 32.2% of such patients, while a mere 1.8% had insomnia. Moreover, among patients diagnosed with bipolar disorder I or II, 20.6% reported hypersomnia, a significantly higher proportion than the 16% who had insomnia.
The finding that only 5% of BRIDGE-II-MIX participants with hypersomnia met DSM-5 criteria for a mixed-state specifier, a rate not significantly different from the 8.3% figure in those with insomnia, underscores the drawbacks of the DSM-5 criteria, according to Dr. Murru. He noted that, in contrast to the DSM-5 criteria, 32.9% of the hypersomniac patients with a major depressive episode met Research Diagnostic Criteria (RDC) for a mixed-state specifier, a rate significantly higher than the 27.6% figure in patients with insomnia.
Specifically, the individual RDC mixed-state specifiers that stood out as significantly more frequent among depressed patients with hypersomnia than insomnia were racing thoughts, by a margin of 15.1% to 10.6%; impulsivity, 16.8% versus 13.2%; distractibility, 29.6% versus 23.4%; hypersexuality, which was present in 4% of patients with hypersomnia but only 2.3% with insomnia; irritable mood, 33.1% versus 24.8%; and a history of insufficient response to previous antidepressant therapy, 34.3%, compared with 27.1% in insomniacs.
When Dr. Murru and his coinvestigators performed a stepwise linear regression analysis to identify significant predictors of hypersomnia in patients with a major depressive episode, they found that the sleep abnormality keeps some interesting company. Patients with current bulimia were 4.21-fold more likely to have hypersomnia than those without the eating disorder. Current social phobia was associated with a 1.77-fold increased risk of hypersomnia; mood lability on prior antidepressant therapy carried a 1.37-fold risk, as did current mood lability; prior attempted suicide was associated with a 1.31-fold increased risk; being overweight or obese was associated with a 1.42-fold risk; currently being on a mood stabilizer carried a 1.33-fold increased risk of hypersomnia; and currently being on an atypical antipsychotic agent had a 1.36-fold greater risk.
Dr. Murru concluded that the take-home message of this study – “Of course, conceding it’s highly exploratory nature intrinsic to a post hoc analysis,” he noted – is that hypersomnia should be included among the symptoms that trigger the “with mixed features” specifier in patients with a major depressive episode.
The BRIDGE-II-MIX study was sponsored by Sanofi-Aventis. Dr. Murru reported having no financial conflicts of interest regarding the study.
BARCELONA – Hypersomnia is a novel, previously unappreciated factor useful in tipping the balance in favor of an underlying bipolar predisposition in patients with an acute major depressive episode, Andrea Murru, MD, PhD, reported at the annual congress of the European College of Neuropsychopharmacology.
“This may help us in clinical practice. It’s an effective, costless, and highly objective clinical measure. It’s one question, and it takes a second. It’s simply asking the patient: ‘Are you sleeping more hours at night than usual?’ It’s a very simple clinical question that could really change the focus of treatment for a patient,” said Dr. Murru, a clinical psychiatrist in the bipolar disorders program of the University of Barcelona.
He presented a post hoc analysis of 2,514 acutely depressed individuals who participated in the BRIDGE-II-MIX (Bipolar Disorders: Improving Diagnosis, Guidance and Education) study, an international, multicenter, cross-sectional, observational study aimed at better characterizing clinically valid mixed features of depression indicative of concurrent manic symptoms.
“This is one of a whole series of hypothesis-generating studies from BRIDGE-II-MIX that are trying to deal with the struggle of understanding whether all the elements that favor mixicity and an underpinning bipolar diathesis are fairly represented in the diagnostic criteria in DSM-5. And what we are basically finding is the DSM-5 criteria are leaving out important symptoms that really do play a role,” the psychiatrist said in an interview.
One of those missing factors, he continued, is hypersomnia. It was present in 16.8% of the study population, and he and his coinvestigators compared them in terms of clinical variables, current and past psychiatric symptoms, and sociodemographics with the 83.2% of patients with insomnia. That is, patients who got fewer hours of sleep than normal and felt fatigued during the next day were compared with those who felt a reduced need to sleep.
The two groups differed in important ways. Hypersomnia showed a powerful correlation with a physician diagnosis of major depressive episode with atypical features, being present in 32.2% of such patients, while a mere 1.8% had insomnia. Moreover, among patients diagnosed with bipolar disorder I or II, 20.6% reported hypersomnia, a significantly higher proportion than the 16% who had insomnia.
The finding that only 5% of BRIDGE-II-MIX participants with hypersomnia met DSM-5 criteria for a mixed-state specifier, a rate not significantly different from the 8.3% figure in those with insomnia, underscores the drawbacks of the DSM-5 criteria, according to Dr. Murru. He noted that, in contrast to the DSM-5 criteria, 32.9% of the hypersomniac patients with a major depressive episode met Research Diagnostic Criteria (RDC) for a mixed-state specifier, a rate significantly higher than the 27.6% figure in patients with insomnia.
Specifically, the individual RDC mixed-state specifiers that stood out as significantly more frequent among depressed patients with hypersomnia than insomnia were racing thoughts, by a margin of 15.1% to 10.6%; impulsivity, 16.8% versus 13.2%; distractibility, 29.6% versus 23.4%; hypersexuality, which was present in 4% of patients with hypersomnia but only 2.3% with insomnia; irritable mood, 33.1% versus 24.8%; and a history of insufficient response to previous antidepressant therapy, 34.3%, compared with 27.1% in insomniacs.
When Dr. Murru and his coinvestigators performed a stepwise linear regression analysis to identify significant predictors of hypersomnia in patients with a major depressive episode, they found that the sleep abnormality keeps some interesting company. Patients with current bulimia were 4.21-fold more likely to have hypersomnia than those without the eating disorder. Current social phobia was associated with a 1.77-fold increased risk of hypersomnia; mood lability on prior antidepressant therapy carried a 1.37-fold risk, as did current mood lability; prior attempted suicide was associated with a 1.31-fold increased risk; being overweight or obese was associated with a 1.42-fold risk; currently being on a mood stabilizer carried a 1.33-fold increased risk of hypersomnia; and currently being on an atypical antipsychotic agent had a 1.36-fold greater risk.
Dr. Murru concluded that the take-home message of this study – “Of course, conceding it’s highly exploratory nature intrinsic to a post hoc analysis,” he noted – is that hypersomnia should be included among the symptoms that trigger the “with mixed features” specifier in patients with a major depressive episode.
The BRIDGE-II-MIX study was sponsored by Sanofi-Aventis. Dr. Murru reported having no financial conflicts of interest regarding the study.
REPORTING FROM THE ECNP CONGRESS
Key clinical point: Ask patients with a major depressive episode about hypersomnia.
Major finding: Hypersomnia in patients with an acute major depressive episode clusters with numerous elements of a bipolar diathesis.
Study details: This was a post hoc analysis of 2,514 acutely depressed individuals who participated in an international, multicenter, cross-sectional, observational study.
Disclosures: The BRIDGE-II-MIX study was sponsored by Sanofi-Aventis. The presenter reported having no financial conflicts of interest regarding the study.
Study Explores Link Between GERD and Poor Sleep Quality
The proportion of adults with GERD has increased, and GERD increases the likelihood of dissatisfaction with sleep.
ATLANTA—Chronic gastroesophageal reflux disease (GERD) is associated with various sleep disorders that might complicate the response to GERD treatment, according to an ongoing longitudinal analysis presented at the 143rd Annual Meeting of the American Neurological Association.
“We have little longitudinal information on GERD in the general population; the last published article on GERD incidence was 20 years ago,” said lead study author Maurice M. Ohayon, MD, DSc, PhD, Director of the Stanford Sleep Epidemiology Research Center in California. “As a sleep specialist, I am always interested to see how a specific medical condition may affect the sleep quality of the individuals with that condition. How we live our day has an impact on our night; it works together.”
Proton-Pump Inhibitors Are Common Treatments
To examine the long-term effects of GERD on sleep disturbances, Dr. Ohayon and his colleagues used US Census data to identify a random sample of adults in Arizona, California, Colorado, Idaho, New York, Oregon, Pennsylvania, and Texas. The researchers conducted two waves of phone interviews with the subjects three years apart, beginning in 2004. They limited their analysis to 10,930 subjects with a mean age of 43 who participated in both interviews.
Between Wave 1 and Wave 2 of phone interviews, the proportion of adults who reported having GERD rose from 10.6% to 12.4%. The prevalence of new GERD cases was 8.5% per year, while the incidence was 3.2% per year. Chronic GERD, defined as GERD present during both interview periods, was observed in 3.9% of the sample.
The researchers found that 77.3% of subjects with GERD were taking a treatment, mostly proton-pump inhibitors, to alleviate their symptoms. Those with chronic GERD were more likely to report being dissatisfied with their sleep during Wave 2 of the study, compared with Wave 1 (24.2% vs 13.5%). In addition, compared with their counterparts without GERD, those with chronic GERD were more likely to wake up at night (33.9% vs 28.3%) and to have nonrestorative sleep (15.6% vs 10.5%).
“Discomfort related to GERD may happen while you are sleeping,” said Dr. Ohayon. “It may wake you up and, if not, it may make you feel unrested when you wake up. We observed both of these symptoms in our GERD participants. Insomnia disorders were also rampant in the chronic GERD group (24.5%, compared with 14.4% in non-GERD participants). An insomnia disorder is more than just having difficulty falling asleep or waking up at night, it means that your daytime functioning is affected by the poor quality of your night.”
GERD May Promote Weight Gain
Other findings from the study were “rather alarming,” said Dr. Ohayon. For example, individuals with GERD, especially those with the chronic form, weighed much more than those with no GERD did. “Over a three-year period, the chronic GERD individuals gained one point in the BMI, which for a six-foot tall man translates into a weight gain of 30 pounds,” he said. “Of course, with that follows high blood pressure, high cholesterol, diabetes, chronic pain, and heart disease.”
He concluded that GERD has its main manifestations when affected individuals are sleeping on their backs. “The impact of GERD on the quality of sleep is major,” he said. “Sleepiness and fatigue during the day are the consequences impacting work, family, and quality of life.”
Dr. Ohayon acknowledged certain limitations of the study, including the fact that the diagnosis of GERD was based on self-report. The study was supported by an unrestricted grant from Takeda.
—Doug Brunk
The proportion of adults with GERD has increased, and GERD increases the likelihood of dissatisfaction with sleep.
The proportion of adults with GERD has increased, and GERD increases the likelihood of dissatisfaction with sleep.
ATLANTA—Chronic gastroesophageal reflux disease (GERD) is associated with various sleep disorders that might complicate the response to GERD treatment, according to an ongoing longitudinal analysis presented at the 143rd Annual Meeting of the American Neurological Association.
“We have little longitudinal information on GERD in the general population; the last published article on GERD incidence was 20 years ago,” said lead study author Maurice M. Ohayon, MD, DSc, PhD, Director of the Stanford Sleep Epidemiology Research Center in California. “As a sleep specialist, I am always interested to see how a specific medical condition may affect the sleep quality of the individuals with that condition. How we live our day has an impact on our night; it works together.”
Proton-Pump Inhibitors Are Common Treatments
To examine the long-term effects of GERD on sleep disturbances, Dr. Ohayon and his colleagues used US Census data to identify a random sample of adults in Arizona, California, Colorado, Idaho, New York, Oregon, Pennsylvania, and Texas. The researchers conducted two waves of phone interviews with the subjects three years apart, beginning in 2004. They limited their analysis to 10,930 subjects with a mean age of 43 who participated in both interviews.
Between Wave 1 and Wave 2 of phone interviews, the proportion of adults who reported having GERD rose from 10.6% to 12.4%. The prevalence of new GERD cases was 8.5% per year, while the incidence was 3.2% per year. Chronic GERD, defined as GERD present during both interview periods, was observed in 3.9% of the sample.
The researchers found that 77.3% of subjects with GERD were taking a treatment, mostly proton-pump inhibitors, to alleviate their symptoms. Those with chronic GERD were more likely to report being dissatisfied with their sleep during Wave 2 of the study, compared with Wave 1 (24.2% vs 13.5%). In addition, compared with their counterparts without GERD, those with chronic GERD were more likely to wake up at night (33.9% vs 28.3%) and to have nonrestorative sleep (15.6% vs 10.5%).
“Discomfort related to GERD may happen while you are sleeping,” said Dr. Ohayon. “It may wake you up and, if not, it may make you feel unrested when you wake up. We observed both of these symptoms in our GERD participants. Insomnia disorders were also rampant in the chronic GERD group (24.5%, compared with 14.4% in non-GERD participants). An insomnia disorder is more than just having difficulty falling asleep or waking up at night, it means that your daytime functioning is affected by the poor quality of your night.”
GERD May Promote Weight Gain
Other findings from the study were “rather alarming,” said Dr. Ohayon. For example, individuals with GERD, especially those with the chronic form, weighed much more than those with no GERD did. “Over a three-year period, the chronic GERD individuals gained one point in the BMI, which for a six-foot tall man translates into a weight gain of 30 pounds,” he said. “Of course, with that follows high blood pressure, high cholesterol, diabetes, chronic pain, and heart disease.”
He concluded that GERD has its main manifestations when affected individuals are sleeping on their backs. “The impact of GERD on the quality of sleep is major,” he said. “Sleepiness and fatigue during the day are the consequences impacting work, family, and quality of life.”
Dr. Ohayon acknowledged certain limitations of the study, including the fact that the diagnosis of GERD was based on self-report. The study was supported by an unrestricted grant from Takeda.
—Doug Brunk
ATLANTA—Chronic gastroesophageal reflux disease (GERD) is associated with various sleep disorders that might complicate the response to GERD treatment, according to an ongoing longitudinal analysis presented at the 143rd Annual Meeting of the American Neurological Association.
“We have little longitudinal information on GERD in the general population; the last published article on GERD incidence was 20 years ago,” said lead study author Maurice M. Ohayon, MD, DSc, PhD, Director of the Stanford Sleep Epidemiology Research Center in California. “As a sleep specialist, I am always interested to see how a specific medical condition may affect the sleep quality of the individuals with that condition. How we live our day has an impact on our night; it works together.”
Proton-Pump Inhibitors Are Common Treatments
To examine the long-term effects of GERD on sleep disturbances, Dr. Ohayon and his colleagues used US Census data to identify a random sample of adults in Arizona, California, Colorado, Idaho, New York, Oregon, Pennsylvania, and Texas. The researchers conducted two waves of phone interviews with the subjects three years apart, beginning in 2004. They limited their analysis to 10,930 subjects with a mean age of 43 who participated in both interviews.
Between Wave 1 and Wave 2 of phone interviews, the proportion of adults who reported having GERD rose from 10.6% to 12.4%. The prevalence of new GERD cases was 8.5% per year, while the incidence was 3.2% per year. Chronic GERD, defined as GERD present during both interview periods, was observed in 3.9% of the sample.
The researchers found that 77.3% of subjects with GERD were taking a treatment, mostly proton-pump inhibitors, to alleviate their symptoms. Those with chronic GERD were more likely to report being dissatisfied with their sleep during Wave 2 of the study, compared with Wave 1 (24.2% vs 13.5%). In addition, compared with their counterparts without GERD, those with chronic GERD were more likely to wake up at night (33.9% vs 28.3%) and to have nonrestorative sleep (15.6% vs 10.5%).
“Discomfort related to GERD may happen while you are sleeping,” said Dr. Ohayon. “It may wake you up and, if not, it may make you feel unrested when you wake up. We observed both of these symptoms in our GERD participants. Insomnia disorders were also rampant in the chronic GERD group (24.5%, compared with 14.4% in non-GERD participants). An insomnia disorder is more than just having difficulty falling asleep or waking up at night, it means that your daytime functioning is affected by the poor quality of your night.”
GERD May Promote Weight Gain
Other findings from the study were “rather alarming,” said Dr. Ohayon. For example, individuals with GERD, especially those with the chronic form, weighed much more than those with no GERD did. “Over a three-year period, the chronic GERD individuals gained one point in the BMI, which for a six-foot tall man translates into a weight gain of 30 pounds,” he said. “Of course, with that follows high blood pressure, high cholesterol, diabetes, chronic pain, and heart disease.”
He concluded that GERD has its main manifestations when affected individuals are sleeping on their backs. “The impact of GERD on the quality of sleep is major,” he said. “Sleepiness and fatigue during the day are the consequences impacting work, family, and quality of life.”
Dr. Ohayon acknowledged certain limitations of the study, including the fact that the diagnosis of GERD was based on self-report. The study was supported by an unrestricted grant from Takeda.
—Doug Brunk