User login
OV-101 shows promise for Angelman syndrome
SEATTLE – A novel extrasynaptic gamma-aminobutyric acid (GABA)–receptor agonist called OV-101 was safe and well-tolerated in adult and adolescent Angelman syndrome patients in a 12-week phase 2 trial. In a secondary analysis, the treatment appeared to improve sleep.
Angelman syndrome is associated with a microdeletion on chromosome 15 encompassing the ubiquitin protein ligase E3a (UBE3A) gene. The resulting loss of expression of the UBE3A protein leads to increases in the uptake of GABA and reduces levels of extrasynaptic GABA. Patients with Angelman syndrome typically have motor dysfunction, often extreme: “These kids are very excitable, very active, and they have lots of trouble with sleep,” said Alex Kolevzon, MD, professor of psychiatry and pediatrics at the Icahn School of Medicine at Mount Sinai, in an interview.
Dr. Kolevzon presented the results at a poster session at the annual meeting of the American Academy of Child and Adolescent Psychiatry.
The study was conducted at 12 sites in the United States and 1 in Israel. Ovid Pharmaceuticals plans to apply to the Food and Drug Administration later this year for approval. There is no existing drug for Angelman syndrome, and the study provided good safety reassurance. “There were some side effects, but for the most part we considered them mild, and only four (out of 88 subjects) discontinued because of side effects,” said Dr. Kolevzon.
The researchers used actigraphy to gain a more objective measure of sleep in the study participants. They randomized 88 patients with Angelman syndrome (aged 13-49 years) to receive placebo in the morning and 15 mg of OV-101 at night, 10 mg OVID-101 in the morning and 15 mg OVID-101 at night, or placebo both in the morning and at night.
Pyrexia occurred in 24% of the group who received the active drug only at night, 3% of the group given the twice-daily dose, and 7% of the placebo group. Seizures occurred in 7% of the once-daily group and 10% of the twice-daily group; seizures were not noted in the placebo group.
The main efficacy outcome measure was the Clinical Global Impressions-9 (CGI-9) scale. The once-daily group had a significant benefit in the sleep domain at 12 weeks, compared with placebo (difference, –0.77; P = .0141), but the twice-daily group had only a trend toward improvement in sleep (difference, –0.45; P = .1407).
Both active therapy groups had significant improvement in CGI-9 measures after 12 weeks of treatment compared to placebo – the twice-daily group (P = .0206, Fisher’s Exact Test) and the once-daily group (P = .0006, mixed model repeated measures analysis).
The actigraphy analysis, conducted in the 45% of patients who could tolerate its use, found that, compared to placebo, the once-daily dosing group experienced an 25.7 minute improvement in latency to sleep onset (P = .0147), as well an approximately 50 minute reduction in sleep time during the day, and a 3.65% improvement in sleep efficiency.
OV-101 has the potential to treat other conditions as well. “Obviously there are a lot of neurodevelopmental disorders where you see dysregulation between the GABAergic and glutamergic systems. This is a drug that has a unique effect on the GABAergic system. It’s already being studied in Fragile X syndrome, where we see this same kind of dysregulation and excess excitation,” said Dr. Kolevzon.
Dr. Kolevzon is a consultant for several drug companies including Ovid Therapeutics.
SOURCE: AACAP 2018. New Research Poster 3.1.
SEATTLE – A novel extrasynaptic gamma-aminobutyric acid (GABA)–receptor agonist called OV-101 was safe and well-tolerated in adult and adolescent Angelman syndrome patients in a 12-week phase 2 trial. In a secondary analysis, the treatment appeared to improve sleep.
Angelman syndrome is associated with a microdeletion on chromosome 15 encompassing the ubiquitin protein ligase E3a (UBE3A) gene. The resulting loss of expression of the UBE3A protein leads to increases in the uptake of GABA and reduces levels of extrasynaptic GABA. Patients with Angelman syndrome typically have motor dysfunction, often extreme: “These kids are very excitable, very active, and they have lots of trouble with sleep,” said Alex Kolevzon, MD, professor of psychiatry and pediatrics at the Icahn School of Medicine at Mount Sinai, in an interview.
Dr. Kolevzon presented the results at a poster session at the annual meeting of the American Academy of Child and Adolescent Psychiatry.
The study was conducted at 12 sites in the United States and 1 in Israel. Ovid Pharmaceuticals plans to apply to the Food and Drug Administration later this year for approval. There is no existing drug for Angelman syndrome, and the study provided good safety reassurance. “There were some side effects, but for the most part we considered them mild, and only four (out of 88 subjects) discontinued because of side effects,” said Dr. Kolevzon.
The researchers used actigraphy to gain a more objective measure of sleep in the study participants. They randomized 88 patients with Angelman syndrome (aged 13-49 years) to receive placebo in the morning and 15 mg of OV-101 at night, 10 mg OVID-101 in the morning and 15 mg OVID-101 at night, or placebo both in the morning and at night.
Pyrexia occurred in 24% of the group who received the active drug only at night, 3% of the group given the twice-daily dose, and 7% of the placebo group. Seizures occurred in 7% of the once-daily group and 10% of the twice-daily group; seizures were not noted in the placebo group.
The main efficacy outcome measure was the Clinical Global Impressions-9 (CGI-9) scale. The once-daily group had a significant benefit in the sleep domain at 12 weeks, compared with placebo (difference, –0.77; P = .0141), but the twice-daily group had only a trend toward improvement in sleep (difference, –0.45; P = .1407).
Both active therapy groups had significant improvement in CGI-9 measures after 12 weeks of treatment compared to placebo – the twice-daily group (P = .0206, Fisher’s Exact Test) and the once-daily group (P = .0006, mixed model repeated measures analysis).
The actigraphy analysis, conducted in the 45% of patients who could tolerate its use, found that, compared to placebo, the once-daily dosing group experienced an 25.7 minute improvement in latency to sleep onset (P = .0147), as well an approximately 50 minute reduction in sleep time during the day, and a 3.65% improvement in sleep efficiency.
OV-101 has the potential to treat other conditions as well. “Obviously there are a lot of neurodevelopmental disorders where you see dysregulation between the GABAergic and glutamergic systems. This is a drug that has a unique effect on the GABAergic system. It’s already being studied in Fragile X syndrome, where we see this same kind of dysregulation and excess excitation,” said Dr. Kolevzon.
Dr. Kolevzon is a consultant for several drug companies including Ovid Therapeutics.
SOURCE: AACAP 2018. New Research Poster 3.1.
SEATTLE – A novel extrasynaptic gamma-aminobutyric acid (GABA)–receptor agonist called OV-101 was safe and well-tolerated in adult and adolescent Angelman syndrome patients in a 12-week phase 2 trial. In a secondary analysis, the treatment appeared to improve sleep.
Angelman syndrome is associated with a microdeletion on chromosome 15 encompassing the ubiquitin protein ligase E3a (UBE3A) gene. The resulting loss of expression of the UBE3A protein leads to increases in the uptake of GABA and reduces levels of extrasynaptic GABA. Patients with Angelman syndrome typically have motor dysfunction, often extreme: “These kids are very excitable, very active, and they have lots of trouble with sleep,” said Alex Kolevzon, MD, professor of psychiatry and pediatrics at the Icahn School of Medicine at Mount Sinai, in an interview.
Dr. Kolevzon presented the results at a poster session at the annual meeting of the American Academy of Child and Adolescent Psychiatry.
The study was conducted at 12 sites in the United States and 1 in Israel. Ovid Pharmaceuticals plans to apply to the Food and Drug Administration later this year for approval. There is no existing drug for Angelman syndrome, and the study provided good safety reassurance. “There were some side effects, but for the most part we considered them mild, and only four (out of 88 subjects) discontinued because of side effects,” said Dr. Kolevzon.
The researchers used actigraphy to gain a more objective measure of sleep in the study participants. They randomized 88 patients with Angelman syndrome (aged 13-49 years) to receive placebo in the morning and 15 mg of OV-101 at night, 10 mg OVID-101 in the morning and 15 mg OVID-101 at night, or placebo both in the morning and at night.
Pyrexia occurred in 24% of the group who received the active drug only at night, 3% of the group given the twice-daily dose, and 7% of the placebo group. Seizures occurred in 7% of the once-daily group and 10% of the twice-daily group; seizures were not noted in the placebo group.
The main efficacy outcome measure was the Clinical Global Impressions-9 (CGI-9) scale. The once-daily group had a significant benefit in the sleep domain at 12 weeks, compared with placebo (difference, –0.77; P = .0141), but the twice-daily group had only a trend toward improvement in sleep (difference, –0.45; P = .1407).
Both active therapy groups had significant improvement in CGI-9 measures after 12 weeks of treatment compared to placebo – the twice-daily group (P = .0206, Fisher’s Exact Test) and the once-daily group (P = .0006, mixed model repeated measures analysis).
The actigraphy analysis, conducted in the 45% of patients who could tolerate its use, found that, compared to placebo, the once-daily dosing group experienced an 25.7 minute improvement in latency to sleep onset (P = .0147), as well an approximately 50 minute reduction in sleep time during the day, and a 3.65% improvement in sleep efficiency.
OV-101 has the potential to treat other conditions as well. “Obviously there are a lot of neurodevelopmental disorders where you see dysregulation between the GABAergic and glutamergic systems. This is a drug that has a unique effect on the GABAergic system. It’s already being studied in Fragile X syndrome, where we see this same kind of dysregulation and excess excitation,” said Dr. Kolevzon.
Dr. Kolevzon is a consultant for several drug companies including Ovid Therapeutics.
SOURCE: AACAP 2018. New Research Poster 3.1.
REPORTING FROM AACAP 2018
Key clinical point: A new drug may improve sleep outcomes in Angelman Syndrome.
Major finding: Patients who received a single daily dose of OV-101 scored better than study participants given placebo on the Clinical Global Impressions-Improvement scale.
Study details: Randomized, controlled phase 2 trial (n = 88).
Disclosures: The study was funded by Ovid Therapeutics. Dr. Kolevzon is a consultant for Ovid Therapeutics and several other drug companies.
Source: AACAP 2018 New Research Poster 3.1. .
Sleep: The new frontier in cardiovascular prevention
MUNICH – Getting less than 6 hours of sleep nightly on a regular basis or waking up multiple times was independently associated with increased risk of subclinical atherosclerosis in the Spanish PESA study, Fernando Dominguez, MD, reported at the annual congress of the European Society of Cardiology.
Moreover, a graded response was evident in PESA (Progression of Early Subclinical Atherosclerosis): The more times an individual typically awoke per night, the greater the number of atherosclerotic carotid or femoral artery territories documented on three-dimensional vascular ultrasound, added Dr. Dominguez of the Spanish National Center for Cardiovascular Research in Madrid.
the cardiologist said.
The cross-sectional PESA study, whose principal investigator was Valentin Fuster, MD, PhD, included 3,974 middle-aged Madrid bank employees free of known heart disease or history of stroke who wore a waistband activity monitor for a week to record sleep quantity and quality. They also underwent three-dimensional vascular ultrasound and measurement of coronary artery calcium.
PESA was one of several large studies presented at the meeting that focused on deviations from normal sleep as a marker for increased risk of cardiovascular disease and/or mortality. Of note, however, PESA was the only one to use activity monitoring technology to track sleep.
“It was essential to use objectively measured sleep variables, because they showed huge disparity with patients’ self-reports on sleep questionnaires,” Dr. Dominguez explained.
Indeed, while 10.7% of PESA participants self-reported sleeping less than 6 hours per night on the Sleep Habits Questionnaire, actigraphy showed the true rate was 27.1%.
Based on actigraphic findings, subjects were divided into tertiles based upon average hours of sleep per night, ranging from less than 6 to more than 8. They were also grouped in quintiles based upon their extent of fragmented sleep.
Subjects with short sleep were significantly older and more likely to have high blood pressure, a higher body mass index, and metabolic syndrome than those who averaged 7-8 hours of sleep. Individuals in the top quintile for sleep awakening were older and had higher prevalences of smoking and hypertension than those in the lowest quintile.
In multivariate analyses adjusted for these differences as well as for physical activity, depression, obstructive sleep apnea, daily calorie consumption, alcohol intake, and other potential confounders, subjects who slept less than 6 hours per night had a 27% greater volume of noncoronary plaque than those who slept 7-8 hours. They also had 21% more vascular territories laden with subclinical atherosclerosis. The risk of subclinical noncoronary atherosclerosis was greater among women who averaged less than 6 hours of sleep per night, representing a 48% relative risk increase in plaque volume, versus 21% in men.
At the other extreme, women who slept more than 8 hours per night had an 83% increased plaque volume, while men who slept that much had no increase in risk, compared with men who slept for 7-8 hours.
Subjects in the top quintile for sleep fragmentation had 34% more vascular territories affected by atherosclerosis than those in the lowest quintile. Their noncoronary plaque burden was 23% greater as well.
An 11-study meta-analysis
Epameinondas Fountas, MD, of the Onassis Cardiac Surgery Center in Athens, presented a meta-analysis of 11 prospective studies of the relationship between daily sleep duration and cardiovascular disease morbidity and mortality published within the past 5 years, reflecting burgeoning interest in this hot-button topic. Collectively, the meta-analysis totaled 1,000,541 adults without baseline cardiovascular disease who were followed for an average of 9.3 years.
In an analysis adjusted for numerous known cardiovascular risk factors, the Greek investigators found that short sleep duration as defined by a self-reported average of less than 6 hours per night was independently associated with a statistically significant and clinically meaningful 11% increase in the risk of diagnosis of fatal or nonfatal cardiovascular disease, compared with individuals who averaged 6-8 hours nightly. Moreover, those who averaged more than 8 hours of sleep per night were also at risk: they averaged a 32% increased risk in fatal or nonfatal cardiovascular events compared to normal 6- to 8-hour sleepers. Thus, 6-8 hours of sleep per night appears to be the sweet spot in terms of cardioprotection.
“Our message to patients is simple: Sleep well, not too long, nor too short, and be active,” Dr. Fountas said.
Numerous investigators have highlighted the pathophysiologic changes related to sleep deprivation that likely boost cardiovascular risk. These include activation of the sympathetic nervous system, increased inflammation, and disrupted glucose metabolism, he noted.
Swedes weigh in
Moa Bengtsson, a combined medical/PhD student at the University of Gothenburg (Sweden), presented a prospective study of 798 men who were 50 years old in 1993, when they underwent a physical examination and completed extensive lifestyle questionnaires that included average self-reported sleep duration. Among the 759 men still available for evaluation after 21 years, or nearly 15,000 person-years of followup, those who reported sleeping an average of 5 hours or less per night back at age 50 were 93% more likely to have experienced a major cardiovascular event by age 71 -- acute MI, stroke, coronary revascularization, heart failure hospitalization, or cardiovascular death -- compared with those who averaged 7-8 hours of shut eye.
The short sleepers had a higher prevalence of obesity, diabetes, hypertension, smoking, and physical inactivity than the men who slept 7-8 hours per night. However, these and other confounders were adjusted for in the multivariate analysis.
To place sleep abnormalities in context, Ms. Bengtssen observed that short sleep in the Gothenburg men was numerically a stronger independent risk factor for future cardiovascular events than obesity, which was associated with an 82% increase in risk, or even smoking, with a 70% increase in risk.
Men who averaged either 6 hours of sleep per night or more than 8 hours were not at increased cardiovascular risk over 21 years of followup, compared with those who slept 7-8 hours.
Like the other investigators, she noted that the studies presented at the meeting, despite their extensive adjustments for potential confounders, don’t prove a direct causal relationship between short sleep and increased cardiovascular risk. An informative next step in research, albeit a challenging one, would be to show whether improved long-term sleep habits favorably alter cardiovascular risk.
All three study investigators reported having no financial conflicts regarding their research, which was conducted free of commercial support.
MUNICH – Getting less than 6 hours of sleep nightly on a regular basis or waking up multiple times was independently associated with increased risk of subclinical atherosclerosis in the Spanish PESA study, Fernando Dominguez, MD, reported at the annual congress of the European Society of Cardiology.
Moreover, a graded response was evident in PESA (Progression of Early Subclinical Atherosclerosis): The more times an individual typically awoke per night, the greater the number of atherosclerotic carotid or femoral artery territories documented on three-dimensional vascular ultrasound, added Dr. Dominguez of the Spanish National Center for Cardiovascular Research in Madrid.
the cardiologist said.
The cross-sectional PESA study, whose principal investigator was Valentin Fuster, MD, PhD, included 3,974 middle-aged Madrid bank employees free of known heart disease or history of stroke who wore a waistband activity monitor for a week to record sleep quantity and quality. They also underwent three-dimensional vascular ultrasound and measurement of coronary artery calcium.
PESA was one of several large studies presented at the meeting that focused on deviations from normal sleep as a marker for increased risk of cardiovascular disease and/or mortality. Of note, however, PESA was the only one to use activity monitoring technology to track sleep.
“It was essential to use objectively measured sleep variables, because they showed huge disparity with patients’ self-reports on sleep questionnaires,” Dr. Dominguez explained.
Indeed, while 10.7% of PESA participants self-reported sleeping less than 6 hours per night on the Sleep Habits Questionnaire, actigraphy showed the true rate was 27.1%.
Based on actigraphic findings, subjects were divided into tertiles based upon average hours of sleep per night, ranging from less than 6 to more than 8. They were also grouped in quintiles based upon their extent of fragmented sleep.
Subjects with short sleep were significantly older and more likely to have high blood pressure, a higher body mass index, and metabolic syndrome than those who averaged 7-8 hours of sleep. Individuals in the top quintile for sleep awakening were older and had higher prevalences of smoking and hypertension than those in the lowest quintile.
In multivariate analyses adjusted for these differences as well as for physical activity, depression, obstructive sleep apnea, daily calorie consumption, alcohol intake, and other potential confounders, subjects who slept less than 6 hours per night had a 27% greater volume of noncoronary plaque than those who slept 7-8 hours. They also had 21% more vascular territories laden with subclinical atherosclerosis. The risk of subclinical noncoronary atherosclerosis was greater among women who averaged less than 6 hours of sleep per night, representing a 48% relative risk increase in plaque volume, versus 21% in men.
At the other extreme, women who slept more than 8 hours per night had an 83% increased plaque volume, while men who slept that much had no increase in risk, compared with men who slept for 7-8 hours.
Subjects in the top quintile for sleep fragmentation had 34% more vascular territories affected by atherosclerosis than those in the lowest quintile. Their noncoronary plaque burden was 23% greater as well.
An 11-study meta-analysis
Epameinondas Fountas, MD, of the Onassis Cardiac Surgery Center in Athens, presented a meta-analysis of 11 prospective studies of the relationship between daily sleep duration and cardiovascular disease morbidity and mortality published within the past 5 years, reflecting burgeoning interest in this hot-button topic. Collectively, the meta-analysis totaled 1,000,541 adults without baseline cardiovascular disease who were followed for an average of 9.3 years.
In an analysis adjusted for numerous known cardiovascular risk factors, the Greek investigators found that short sleep duration as defined by a self-reported average of less than 6 hours per night was independently associated with a statistically significant and clinically meaningful 11% increase in the risk of diagnosis of fatal or nonfatal cardiovascular disease, compared with individuals who averaged 6-8 hours nightly. Moreover, those who averaged more than 8 hours of sleep per night were also at risk: they averaged a 32% increased risk in fatal or nonfatal cardiovascular events compared to normal 6- to 8-hour sleepers. Thus, 6-8 hours of sleep per night appears to be the sweet spot in terms of cardioprotection.
“Our message to patients is simple: Sleep well, not too long, nor too short, and be active,” Dr. Fountas said.
Numerous investigators have highlighted the pathophysiologic changes related to sleep deprivation that likely boost cardiovascular risk. These include activation of the sympathetic nervous system, increased inflammation, and disrupted glucose metabolism, he noted.
Swedes weigh in
Moa Bengtsson, a combined medical/PhD student at the University of Gothenburg (Sweden), presented a prospective study of 798 men who were 50 years old in 1993, when they underwent a physical examination and completed extensive lifestyle questionnaires that included average self-reported sleep duration. Among the 759 men still available for evaluation after 21 years, or nearly 15,000 person-years of followup, those who reported sleeping an average of 5 hours or less per night back at age 50 were 93% more likely to have experienced a major cardiovascular event by age 71 -- acute MI, stroke, coronary revascularization, heart failure hospitalization, or cardiovascular death -- compared with those who averaged 7-8 hours of shut eye.
The short sleepers had a higher prevalence of obesity, diabetes, hypertension, smoking, and physical inactivity than the men who slept 7-8 hours per night. However, these and other confounders were adjusted for in the multivariate analysis.
To place sleep abnormalities in context, Ms. Bengtssen observed that short sleep in the Gothenburg men was numerically a stronger independent risk factor for future cardiovascular events than obesity, which was associated with an 82% increase in risk, or even smoking, with a 70% increase in risk.
Men who averaged either 6 hours of sleep per night or more than 8 hours were not at increased cardiovascular risk over 21 years of followup, compared with those who slept 7-8 hours.
Like the other investigators, she noted that the studies presented at the meeting, despite their extensive adjustments for potential confounders, don’t prove a direct causal relationship between short sleep and increased cardiovascular risk. An informative next step in research, albeit a challenging one, would be to show whether improved long-term sleep habits favorably alter cardiovascular risk.
All three study investigators reported having no financial conflicts regarding their research, which was conducted free of commercial support.
MUNICH – Getting less than 6 hours of sleep nightly on a regular basis or waking up multiple times was independently associated with increased risk of subclinical atherosclerosis in the Spanish PESA study, Fernando Dominguez, MD, reported at the annual congress of the European Society of Cardiology.
Moreover, a graded response was evident in PESA (Progression of Early Subclinical Atherosclerosis): The more times an individual typically awoke per night, the greater the number of atherosclerotic carotid or femoral artery territories documented on three-dimensional vascular ultrasound, added Dr. Dominguez of the Spanish National Center for Cardiovascular Research in Madrid.
the cardiologist said.
The cross-sectional PESA study, whose principal investigator was Valentin Fuster, MD, PhD, included 3,974 middle-aged Madrid bank employees free of known heart disease or history of stroke who wore a waistband activity monitor for a week to record sleep quantity and quality. They also underwent three-dimensional vascular ultrasound and measurement of coronary artery calcium.
PESA was one of several large studies presented at the meeting that focused on deviations from normal sleep as a marker for increased risk of cardiovascular disease and/or mortality. Of note, however, PESA was the only one to use activity monitoring technology to track sleep.
“It was essential to use objectively measured sleep variables, because they showed huge disparity with patients’ self-reports on sleep questionnaires,” Dr. Dominguez explained.
Indeed, while 10.7% of PESA participants self-reported sleeping less than 6 hours per night on the Sleep Habits Questionnaire, actigraphy showed the true rate was 27.1%.
Based on actigraphic findings, subjects were divided into tertiles based upon average hours of sleep per night, ranging from less than 6 to more than 8. They were also grouped in quintiles based upon their extent of fragmented sleep.
Subjects with short sleep were significantly older and more likely to have high blood pressure, a higher body mass index, and metabolic syndrome than those who averaged 7-8 hours of sleep. Individuals in the top quintile for sleep awakening were older and had higher prevalences of smoking and hypertension than those in the lowest quintile.
In multivariate analyses adjusted for these differences as well as for physical activity, depression, obstructive sleep apnea, daily calorie consumption, alcohol intake, and other potential confounders, subjects who slept less than 6 hours per night had a 27% greater volume of noncoronary plaque than those who slept 7-8 hours. They also had 21% more vascular territories laden with subclinical atherosclerosis. The risk of subclinical noncoronary atherosclerosis was greater among women who averaged less than 6 hours of sleep per night, representing a 48% relative risk increase in plaque volume, versus 21% in men.
At the other extreme, women who slept more than 8 hours per night had an 83% increased plaque volume, while men who slept that much had no increase in risk, compared with men who slept for 7-8 hours.
Subjects in the top quintile for sleep fragmentation had 34% more vascular territories affected by atherosclerosis than those in the lowest quintile. Their noncoronary plaque burden was 23% greater as well.
An 11-study meta-analysis
Epameinondas Fountas, MD, of the Onassis Cardiac Surgery Center in Athens, presented a meta-analysis of 11 prospective studies of the relationship between daily sleep duration and cardiovascular disease morbidity and mortality published within the past 5 years, reflecting burgeoning interest in this hot-button topic. Collectively, the meta-analysis totaled 1,000,541 adults without baseline cardiovascular disease who were followed for an average of 9.3 years.
In an analysis adjusted for numerous known cardiovascular risk factors, the Greek investigators found that short sleep duration as defined by a self-reported average of less than 6 hours per night was independently associated with a statistically significant and clinically meaningful 11% increase in the risk of diagnosis of fatal or nonfatal cardiovascular disease, compared with individuals who averaged 6-8 hours nightly. Moreover, those who averaged more than 8 hours of sleep per night were also at risk: they averaged a 32% increased risk in fatal or nonfatal cardiovascular events compared to normal 6- to 8-hour sleepers. Thus, 6-8 hours of sleep per night appears to be the sweet spot in terms of cardioprotection.
“Our message to patients is simple: Sleep well, not too long, nor too short, and be active,” Dr. Fountas said.
Numerous investigators have highlighted the pathophysiologic changes related to sleep deprivation that likely boost cardiovascular risk. These include activation of the sympathetic nervous system, increased inflammation, and disrupted glucose metabolism, he noted.
Swedes weigh in
Moa Bengtsson, a combined medical/PhD student at the University of Gothenburg (Sweden), presented a prospective study of 798 men who were 50 years old in 1993, when they underwent a physical examination and completed extensive lifestyle questionnaires that included average self-reported sleep duration. Among the 759 men still available for evaluation after 21 years, or nearly 15,000 person-years of followup, those who reported sleeping an average of 5 hours or less per night back at age 50 were 93% more likely to have experienced a major cardiovascular event by age 71 -- acute MI, stroke, coronary revascularization, heart failure hospitalization, or cardiovascular death -- compared with those who averaged 7-8 hours of shut eye.
The short sleepers had a higher prevalence of obesity, diabetes, hypertension, smoking, and physical inactivity than the men who slept 7-8 hours per night. However, these and other confounders were adjusted for in the multivariate analysis.
To place sleep abnormalities in context, Ms. Bengtssen observed that short sleep in the Gothenburg men was numerically a stronger independent risk factor for future cardiovascular events than obesity, which was associated with an 82% increase in risk, or even smoking, with a 70% increase in risk.
Men who averaged either 6 hours of sleep per night or more than 8 hours were not at increased cardiovascular risk over 21 years of followup, compared with those who slept 7-8 hours.
Like the other investigators, she noted that the studies presented at the meeting, despite their extensive adjustments for potential confounders, don’t prove a direct causal relationship between short sleep and increased cardiovascular risk. An informative next step in research, albeit a challenging one, would be to show whether improved long-term sleep habits favorably alter cardiovascular risk.
All three study investigators reported having no financial conflicts regarding their research, which was conducted free of commercial support.
REPORTING FROM THE ESC CONGRESS 2018
Opioids negatively affect breathing during sleep
SAN ANTONIO – Opioids do not mix well with sleep, interfering with breathing and increasing the risk of central sleep apnea, explained Anita Rajagopal, MD, a pulmonologist in private practice in Indianapolis.
“The chronic respiratory suppressant effects of opioids are well described,” Dr. Rajagopal told attendees at the annual meeting of the American College of Chest Physicians. “The most characteristic signs of chronic opioid effects are irregular central apneas, ataxic breathing, Biot’s respiration and hypoxemia, mainly during NREM sleep.”
Dr. Rajagopal reviewed the research on the effects of opioid use, primarily for therapeutic use, during sleep, especially highlighting the adverse respiratory effects.
In one small study of 24 patients, ages 18-75, who were taking long-term opioids for chronic pain, 46% had severe sleep-disordered breathing, defined as an apnea-hypopnea index greater than 30/hour (J Clin Sleep Med. 2014 Aug 15;10[8]:847-52).
When compared to sleep clinic patients referred for sleep disordered breathing, the participants taking opioids had a higher frequency of central apneas and a lower arousal index. Further, the researchers found that “morphine equivalent doses correlated with the severity of sleep-disordered breathing.”
In another study, a systematic review from 2015, researchers sought to characterize the clinical features of sleep-disordered breathing associated with chronic opioid therapy (Anesth Analg. 2015 Jun;120[6]:1273-85). They identified eight studies with 560 patients, about a quarter of whom (24%) had central sleep apnea.
Once again, “The morphine equivalent daily dose was strongly associated with the severity of the sleep disordered breathing, predominantly central sleep apnea, with a morphine equivalent daily dose of more than 200 mg being a threshold of particular concern,” the researchers reported.
Patients receiving methadone therapy for heroin addiction are not spared the respiratory risks of opioids during sleep. Dr. Rajagopal shared research revealing that patients receiving methadone treatment for at least two months had a blunted hypercapnic respiratory response and increased hypoxemic ventilatory response, changes related to respiratory rate but not tidal volume.
“All mu-opioid receptor agonists can cause complex and potentially lethal effects on respiration during sleep,” Dr. Rajagopal said as she shared evidence from a 2007 study that compared breathing patterns during sleep between 60 patients taking chronic opioids and 60 matched patients not taking opioids (J Clin Sleep Med. 2007 Aug 15;3[5]:455-61).
That study found chronic opioid use to be associated with increased central apneas and reduced arterial oxygen saturation during wakefulness and NREM sleep. Again, a dose-response relationship emerged between morphine dose equivalent and the apnea-hypopnea, obstructive apnea, hypopnea and central apnea indices (P less than .001).
Patients who took opioids long-term were also more likely to have ataxic or irregular breathing during NREM sleep, compared with patients not taking opioids.
In yet another meta-analysis and systematic review she related, researchers found across 803 patients in seven studies that long-term opioids users had a modestly increased risk for central sleep apnea but no similar increased risk for obstructive sleep apnea (J Clin Sleep Med. 2016 Apr 15;12[4]:617-25).
“REM and slow-wave sleep are decreased across all categories of opioid use — intravenous morphine, oral morphine, or methadone and heroin,” she said.
Since some patients are still going to need opioids, such as methadone therapy for those recovering from opioid use disorder, it’s important to understand appropriate effective treatments for central sleep apnea.
“CPAP [continuous positive airway pressure] is generally ineffective for opioid-induced sleep apnea and may augment central events,” Dr. Rajagopal explained, but adaptive servo ventilation (ASV) is effective for opioid-induced central apneas.
In one study of 20 patients receiving opioid therapy and referred for obstructive apnea, for example, the participants were diagnosed instead with central sleep apnea (J Clin Sleep Med. 2014 Jun 15;10[6]:637-43). The 16 patients who received CPAP continued to show central sleep apnea, with an AHI of 34 events/hour and central-apnea index (CAI) of 20 events/hour. Even after a four-week break before restarting CPAP, patients’ apnea did not resolve.
After receiving ASV, however, the average AHI dropped to 11 events/hour and CAI dropped to 0 events/hour. Those changes were accompanied by improvements in oxygen saturation, with the oxyhemoglobin saturation nadir increasing from 83% to 90%.
Similarly, a prospective multi-center observational trial assessed 27 patients with central apnea after they used ASV at home for three months (Chest. 2015 Dec;148[6]:1454-1461). The participants began with an average AHI of 55 and CAI of 23 at baseline. CPAP dropped these values only to an AHI of 33 and CAI of 10, but treatment with ASV dropped them to an AHI of 4 and CAI of 0 (P less than .001).
SAN ANTONIO – Opioids do not mix well with sleep, interfering with breathing and increasing the risk of central sleep apnea, explained Anita Rajagopal, MD, a pulmonologist in private practice in Indianapolis.
“The chronic respiratory suppressant effects of opioids are well described,” Dr. Rajagopal told attendees at the annual meeting of the American College of Chest Physicians. “The most characteristic signs of chronic opioid effects are irregular central apneas, ataxic breathing, Biot’s respiration and hypoxemia, mainly during NREM sleep.”
Dr. Rajagopal reviewed the research on the effects of opioid use, primarily for therapeutic use, during sleep, especially highlighting the adverse respiratory effects.
In one small study of 24 patients, ages 18-75, who were taking long-term opioids for chronic pain, 46% had severe sleep-disordered breathing, defined as an apnea-hypopnea index greater than 30/hour (J Clin Sleep Med. 2014 Aug 15;10[8]:847-52).
When compared to sleep clinic patients referred for sleep disordered breathing, the participants taking opioids had a higher frequency of central apneas and a lower arousal index. Further, the researchers found that “morphine equivalent doses correlated with the severity of sleep-disordered breathing.”
In another study, a systematic review from 2015, researchers sought to characterize the clinical features of sleep-disordered breathing associated with chronic opioid therapy (Anesth Analg. 2015 Jun;120[6]:1273-85). They identified eight studies with 560 patients, about a quarter of whom (24%) had central sleep apnea.
Once again, “The morphine equivalent daily dose was strongly associated with the severity of the sleep disordered breathing, predominantly central sleep apnea, with a morphine equivalent daily dose of more than 200 mg being a threshold of particular concern,” the researchers reported.
Patients receiving methadone therapy for heroin addiction are not spared the respiratory risks of opioids during sleep. Dr. Rajagopal shared research revealing that patients receiving methadone treatment for at least two months had a blunted hypercapnic respiratory response and increased hypoxemic ventilatory response, changes related to respiratory rate but not tidal volume.
“All mu-opioid receptor agonists can cause complex and potentially lethal effects on respiration during sleep,” Dr. Rajagopal said as she shared evidence from a 2007 study that compared breathing patterns during sleep between 60 patients taking chronic opioids and 60 matched patients not taking opioids (J Clin Sleep Med. 2007 Aug 15;3[5]:455-61).
That study found chronic opioid use to be associated with increased central apneas and reduced arterial oxygen saturation during wakefulness and NREM sleep. Again, a dose-response relationship emerged between morphine dose equivalent and the apnea-hypopnea, obstructive apnea, hypopnea and central apnea indices (P less than .001).
Patients who took opioids long-term were also more likely to have ataxic or irregular breathing during NREM sleep, compared with patients not taking opioids.
In yet another meta-analysis and systematic review she related, researchers found across 803 patients in seven studies that long-term opioids users had a modestly increased risk for central sleep apnea but no similar increased risk for obstructive sleep apnea (J Clin Sleep Med. 2016 Apr 15;12[4]:617-25).
“REM and slow-wave sleep are decreased across all categories of opioid use — intravenous morphine, oral morphine, or methadone and heroin,” she said.
Since some patients are still going to need opioids, such as methadone therapy for those recovering from opioid use disorder, it’s important to understand appropriate effective treatments for central sleep apnea.
“CPAP [continuous positive airway pressure] is generally ineffective for opioid-induced sleep apnea and may augment central events,” Dr. Rajagopal explained, but adaptive servo ventilation (ASV) is effective for opioid-induced central apneas.
In one study of 20 patients receiving opioid therapy and referred for obstructive apnea, for example, the participants were diagnosed instead with central sleep apnea (J Clin Sleep Med. 2014 Jun 15;10[6]:637-43). The 16 patients who received CPAP continued to show central sleep apnea, with an AHI of 34 events/hour and central-apnea index (CAI) of 20 events/hour. Even after a four-week break before restarting CPAP, patients’ apnea did not resolve.
After receiving ASV, however, the average AHI dropped to 11 events/hour and CAI dropped to 0 events/hour. Those changes were accompanied by improvements in oxygen saturation, with the oxyhemoglobin saturation nadir increasing from 83% to 90%.
Similarly, a prospective multi-center observational trial assessed 27 patients with central apnea after they used ASV at home for three months (Chest. 2015 Dec;148[6]:1454-1461). The participants began with an average AHI of 55 and CAI of 23 at baseline. CPAP dropped these values only to an AHI of 33 and CAI of 10, but treatment with ASV dropped them to an AHI of 4 and CAI of 0 (P less than .001).
SAN ANTONIO – Opioids do not mix well with sleep, interfering with breathing and increasing the risk of central sleep apnea, explained Anita Rajagopal, MD, a pulmonologist in private practice in Indianapolis.
“The chronic respiratory suppressant effects of opioids are well described,” Dr. Rajagopal told attendees at the annual meeting of the American College of Chest Physicians. “The most characteristic signs of chronic opioid effects are irregular central apneas, ataxic breathing, Biot’s respiration and hypoxemia, mainly during NREM sleep.”
Dr. Rajagopal reviewed the research on the effects of opioid use, primarily for therapeutic use, during sleep, especially highlighting the adverse respiratory effects.
In one small study of 24 patients, ages 18-75, who were taking long-term opioids for chronic pain, 46% had severe sleep-disordered breathing, defined as an apnea-hypopnea index greater than 30/hour (J Clin Sleep Med. 2014 Aug 15;10[8]:847-52).
When compared to sleep clinic patients referred for sleep disordered breathing, the participants taking opioids had a higher frequency of central apneas and a lower arousal index. Further, the researchers found that “morphine equivalent doses correlated with the severity of sleep-disordered breathing.”
In another study, a systematic review from 2015, researchers sought to characterize the clinical features of sleep-disordered breathing associated with chronic opioid therapy (Anesth Analg. 2015 Jun;120[6]:1273-85). They identified eight studies with 560 patients, about a quarter of whom (24%) had central sleep apnea.
Once again, “The morphine equivalent daily dose was strongly associated with the severity of the sleep disordered breathing, predominantly central sleep apnea, with a morphine equivalent daily dose of more than 200 mg being a threshold of particular concern,” the researchers reported.
Patients receiving methadone therapy for heroin addiction are not spared the respiratory risks of opioids during sleep. Dr. Rajagopal shared research revealing that patients receiving methadone treatment for at least two months had a blunted hypercapnic respiratory response and increased hypoxemic ventilatory response, changes related to respiratory rate but not tidal volume.
“All mu-opioid receptor agonists can cause complex and potentially lethal effects on respiration during sleep,” Dr. Rajagopal said as she shared evidence from a 2007 study that compared breathing patterns during sleep between 60 patients taking chronic opioids and 60 matched patients not taking opioids (J Clin Sleep Med. 2007 Aug 15;3[5]:455-61).
That study found chronic opioid use to be associated with increased central apneas and reduced arterial oxygen saturation during wakefulness and NREM sleep. Again, a dose-response relationship emerged between morphine dose equivalent and the apnea-hypopnea, obstructive apnea, hypopnea and central apnea indices (P less than .001).
Patients who took opioids long-term were also more likely to have ataxic or irregular breathing during NREM sleep, compared with patients not taking opioids.
In yet another meta-analysis and systematic review she related, researchers found across 803 patients in seven studies that long-term opioids users had a modestly increased risk for central sleep apnea but no similar increased risk for obstructive sleep apnea (J Clin Sleep Med. 2016 Apr 15;12[4]:617-25).
“REM and slow-wave sleep are decreased across all categories of opioid use — intravenous morphine, oral morphine, or methadone and heroin,” she said.
Since some patients are still going to need opioids, such as methadone therapy for those recovering from opioid use disorder, it’s important to understand appropriate effective treatments for central sleep apnea.
“CPAP [continuous positive airway pressure] is generally ineffective for opioid-induced sleep apnea and may augment central events,” Dr. Rajagopal explained, but adaptive servo ventilation (ASV) is effective for opioid-induced central apneas.
In one study of 20 patients receiving opioid therapy and referred for obstructive apnea, for example, the participants were diagnosed instead with central sleep apnea (J Clin Sleep Med. 2014 Jun 15;10[6]:637-43). The 16 patients who received CPAP continued to show central sleep apnea, with an AHI of 34 events/hour and central-apnea index (CAI) of 20 events/hour. Even after a four-week break before restarting CPAP, patients’ apnea did not resolve.
After receiving ASV, however, the average AHI dropped to 11 events/hour and CAI dropped to 0 events/hour. Those changes were accompanied by improvements in oxygen saturation, with the oxyhemoglobin saturation nadir increasing from 83% to 90%.
Similarly, a prospective multi-center observational trial assessed 27 patients with central apnea after they used ASV at home for three months (Chest. 2015 Dec;148[6]:1454-1461). The participants began with an average AHI of 55 and CAI of 23 at baseline. CPAP dropped these values only to an AHI of 33 and CAI of 10, but treatment with ASV dropped them to an AHI of 4 and CAI of 0 (P less than .001).
REPORTING FROM CHEST 2018
Exercise improves outcomes for patients with heart failure and OSA
Exercise may be as effective as CPAP in improving obstructive sleep apnea and quality of life in patients with heart failure, according to a study published in the October issue of Chest [https://journal.chestnet.org/article/S0012-3692(18)30790-6/fulltext].
Researchers undertook a randomized, four-arm trial in 65 patients with heart failure and obstructive sleep apnea, which compared the effects of CPAP alone, exercise alone – consisting of three supervised sessions per week for three months, or CPAP plus exercise. A control group received education sessions on the importance of exercise.
The greatest reduction in mean apnea-hypopnea index was seen in the CPAP group, who experienced a mean decrease of 24 events per hour. The exercise plus CPAP group and the exercise only groups showed a mean decrease of 10 events per hour. In contrast, the control group showed no significant decrease in the number of events per hour of sleep.
The authors commented that the change in apnea-hypopnea index was due to reduction in obstructive apneas and hypopneas, and noted the “difficulty of accurately distinguishing obstructive from central hypopneas”.
All the active interventions were associated with significant decreases in arousal index and improvements in sleep-related saturation compared to the control intervention.
Exercise – both alone and with CPAP – was associated with an increase in maximum heart rate and peak VO2, and decrease in VE/VCO2 slope compared to the CPAP-alone and control groups.
“We found that peak oxygen consumption and muscle performance improved significantly only in the exercise groups, but not with CPAP alone, even though CPAP was most effective in attenuating OSA severity,” wrote Dr. Denise M. Servantes, from the Departamento de Psicobiologia at the Universidade Federal de São Paul in Brazil, and co-authors. “Because peak VO2 is an independent predictor of survival and crucial to the optimal timing of cardiac transplantation, these findings have important clinical implications, even in patients who are adherent to CPAP.”
A significant number of participants in the active intervention groups changed New York Heart Association functional class; the number of patients in the exercise group in class I went from 0%-88% by three months, in the CPAP group it increased from 0% to 47%, and in the CPAP plus exercise group, it increased from 0% to 73%.
The study also found evidence of a trend towards improved sexual function in the participants who undertook both exercise plus CPAP.
All patients in the intervention groups showed improvements in subjective daytime sleepiness and quality of life, although improvements in the Minnesota Living with Heart Failure Questionnaire and Short Form Health Survey (SF-36) were significant only in the two groups that did exercise.
“The data suggest that exercise could be a therapeutic option for patients with HF and OSA who refuse CPAP or are intolerant to it,” the authors wrote. “In this regard, a considerable number of patients with HF and OSA do not experience subjective excessive daytime sleepiness and consequently observe no immediate benefit from using CPAP, which could contribute to poor long-term adherence.”
Individuals in the exercise group showed a slight but significant weight reduction, and those who undertook the exercise program also showed significant improvements in muscle strength and endurance compared to the control group.
The authors commented that another study examining the impact of weight loss program in people with moderate to severe obstructive sleep apnea found weight loss only or combined interventions achieved benefits for C-reactive protein levels, insulin resistance, and serum triglyceride levels. But these benefits weren’t seen with CPAP alone.
“The results of that study, and the present one emphasize the importance of adjunctive therapy of OSA with weight loss and exercise when applicable.”
However they acknowledged that the short duration of the study, and small sample size were limitations, and that this was only a preliminary investigation.
No conflicts of interest were declared.
SOURCE: Servantes D et al. Chest, 2018; 154:808-817. https://doi.org/10.1016/j.chest.2018.05.011. https://journal.chestnet.org/article/S0012-3692(18)30790-6/fulltext
Exercise may be as effective as CPAP in improving obstructive sleep apnea and quality of life in patients with heart failure, according to a study published in the October issue of Chest [https://journal.chestnet.org/article/S0012-3692(18)30790-6/fulltext].
Researchers undertook a randomized, four-arm trial in 65 patients with heart failure and obstructive sleep apnea, which compared the effects of CPAP alone, exercise alone – consisting of three supervised sessions per week for three months, or CPAP plus exercise. A control group received education sessions on the importance of exercise.
The greatest reduction in mean apnea-hypopnea index was seen in the CPAP group, who experienced a mean decrease of 24 events per hour. The exercise plus CPAP group and the exercise only groups showed a mean decrease of 10 events per hour. In contrast, the control group showed no significant decrease in the number of events per hour of sleep.
The authors commented that the change in apnea-hypopnea index was due to reduction in obstructive apneas and hypopneas, and noted the “difficulty of accurately distinguishing obstructive from central hypopneas”.
All the active interventions were associated with significant decreases in arousal index and improvements in sleep-related saturation compared to the control intervention.
Exercise – both alone and with CPAP – was associated with an increase in maximum heart rate and peak VO2, and decrease in VE/VCO2 slope compared to the CPAP-alone and control groups.
“We found that peak oxygen consumption and muscle performance improved significantly only in the exercise groups, but not with CPAP alone, even though CPAP was most effective in attenuating OSA severity,” wrote Dr. Denise M. Servantes, from the Departamento de Psicobiologia at the Universidade Federal de São Paul in Brazil, and co-authors. “Because peak VO2 is an independent predictor of survival and crucial to the optimal timing of cardiac transplantation, these findings have important clinical implications, even in patients who are adherent to CPAP.”
A significant number of participants in the active intervention groups changed New York Heart Association functional class; the number of patients in the exercise group in class I went from 0%-88% by three months, in the CPAP group it increased from 0% to 47%, and in the CPAP plus exercise group, it increased from 0% to 73%.
The study also found evidence of a trend towards improved sexual function in the participants who undertook both exercise plus CPAP.
All patients in the intervention groups showed improvements in subjective daytime sleepiness and quality of life, although improvements in the Minnesota Living with Heart Failure Questionnaire and Short Form Health Survey (SF-36) were significant only in the two groups that did exercise.
“The data suggest that exercise could be a therapeutic option for patients with HF and OSA who refuse CPAP or are intolerant to it,” the authors wrote. “In this regard, a considerable number of patients with HF and OSA do not experience subjective excessive daytime sleepiness and consequently observe no immediate benefit from using CPAP, which could contribute to poor long-term adherence.”
Individuals in the exercise group showed a slight but significant weight reduction, and those who undertook the exercise program also showed significant improvements in muscle strength and endurance compared to the control group.
The authors commented that another study examining the impact of weight loss program in people with moderate to severe obstructive sleep apnea found weight loss only or combined interventions achieved benefits for C-reactive protein levels, insulin resistance, and serum triglyceride levels. But these benefits weren’t seen with CPAP alone.
“The results of that study, and the present one emphasize the importance of adjunctive therapy of OSA with weight loss and exercise when applicable.”
However they acknowledged that the short duration of the study, and small sample size were limitations, and that this was only a preliminary investigation.
No conflicts of interest were declared.
SOURCE: Servantes D et al. Chest, 2018; 154:808-817. https://doi.org/10.1016/j.chest.2018.05.011. https://journal.chestnet.org/article/S0012-3692(18)30790-6/fulltext
Exercise may be as effective as CPAP in improving obstructive sleep apnea and quality of life in patients with heart failure, according to a study published in the October issue of Chest [https://journal.chestnet.org/article/S0012-3692(18)30790-6/fulltext].
Researchers undertook a randomized, four-arm trial in 65 patients with heart failure and obstructive sleep apnea, which compared the effects of CPAP alone, exercise alone – consisting of three supervised sessions per week for three months, or CPAP plus exercise. A control group received education sessions on the importance of exercise.
The greatest reduction in mean apnea-hypopnea index was seen in the CPAP group, who experienced a mean decrease of 24 events per hour. The exercise plus CPAP group and the exercise only groups showed a mean decrease of 10 events per hour. In contrast, the control group showed no significant decrease in the number of events per hour of sleep.
The authors commented that the change in apnea-hypopnea index was due to reduction in obstructive apneas and hypopneas, and noted the “difficulty of accurately distinguishing obstructive from central hypopneas”.
All the active interventions were associated with significant decreases in arousal index and improvements in sleep-related saturation compared to the control intervention.
Exercise – both alone and with CPAP – was associated with an increase in maximum heart rate and peak VO2, and decrease in VE/VCO2 slope compared to the CPAP-alone and control groups.
“We found that peak oxygen consumption and muscle performance improved significantly only in the exercise groups, but not with CPAP alone, even though CPAP was most effective in attenuating OSA severity,” wrote Dr. Denise M. Servantes, from the Departamento de Psicobiologia at the Universidade Federal de São Paul in Brazil, and co-authors. “Because peak VO2 is an independent predictor of survival and crucial to the optimal timing of cardiac transplantation, these findings have important clinical implications, even in patients who are adherent to CPAP.”
A significant number of participants in the active intervention groups changed New York Heart Association functional class; the number of patients in the exercise group in class I went from 0%-88% by three months, in the CPAP group it increased from 0% to 47%, and in the CPAP plus exercise group, it increased from 0% to 73%.
The study also found evidence of a trend towards improved sexual function in the participants who undertook both exercise plus CPAP.
All patients in the intervention groups showed improvements in subjective daytime sleepiness and quality of life, although improvements in the Minnesota Living with Heart Failure Questionnaire and Short Form Health Survey (SF-36) were significant only in the two groups that did exercise.
“The data suggest that exercise could be a therapeutic option for patients with HF and OSA who refuse CPAP or are intolerant to it,” the authors wrote. “In this regard, a considerable number of patients with HF and OSA do not experience subjective excessive daytime sleepiness and consequently observe no immediate benefit from using CPAP, which could contribute to poor long-term adherence.”
Individuals in the exercise group showed a slight but significant weight reduction, and those who undertook the exercise program also showed significant improvements in muscle strength and endurance compared to the control group.
The authors commented that another study examining the impact of weight loss program in people with moderate to severe obstructive sleep apnea found weight loss only or combined interventions achieved benefits for C-reactive protein levels, insulin resistance, and serum triglyceride levels. But these benefits weren’t seen with CPAP alone.
“The results of that study, and the present one emphasize the importance of adjunctive therapy of OSA with weight loss and exercise when applicable.”
However they acknowledged that the short duration of the study, and small sample size were limitations, and that this was only a preliminary investigation.
No conflicts of interest were declared.
SOURCE: Servantes D et al. Chest, 2018; 154:808-817. https://doi.org/10.1016/j.chest.2018.05.011. https://journal.chestnet.org/article/S0012-3692(18)30790-6/fulltext
FROM CHEST
Key clinical point: Exercise alone or with CPAP achieves additional improvements to quality of life in patients with heart failure and obstructive sleep apnea.
Major finding: Individuals with heart failure and obstructive sleep apnea showed significant improvements to quality of life with exercise.
Study details: Randomized controlled trial in 65 patients with heart failure and obstructive sleep apnea.
Disclosures: The study was supported by the Associacao Fundo de Incentivo a Pesquisa, Sao Paulo Research Foundation. No conflicts of interest were declared.
Source: Servantes D et al.Chest 2018;154:808-817.doi:10.1016/j.chest.2018.05.011
Management of Lewy body dementia remains complex
ATLANTA – In the not-so-distant past, neurologists viewed dementia with Lewy bodies as a disorder primarily of the brain, but it turned out to be far more complex than that.
At the annual meeting of the American Neurological Association, Bradley F. Boeve, MD, described dementia with Lewy bodies (DLB) as a systemic neurologic disorder affecting the brain, including brain stem, spinal cord, and peripheral nervous system, especially the autonomic nervous system. “This leads to the complex array of clinical manifestations, which are quite different from patient to patient cross-sectionally and longitudinally,” said Dr. Boeve, the Little Family Foundation Professor of Lewy Body Dementia in the department of neurology at the Mayo Clinic, Rochester, Minn.
, he said. The four core clinical features are Parkinsonism unrelated to medications; recurrent, fully-formed visual hallucinations; fluctuations in cognition and/or arousal; and rapid eye movement (REM) sleep behavior disorder. “This is the most predictive of all four features,” Dr. Boeve said. He described REM sleep behavior disorder as a parasomnia manifested by the tendency to repeatedly “act out one’s dreams.” The dreams tend to contain a chasing/attacking theme, and behaviors mirror dream content. Injuries to the patient and bed partner can occur.
Typically, patients will present with REM sleep behavior disorder in their 50s and 60s, and sometimes in their 30s and 40s, “decades before cognitive changes begin,” he said. “This is usually followed by Parkinsonism and visual hallucinations. That’s the prototypical DLB [case], but there are many examples where this is not followed. Prominent neuropsychiatric features can also begin before any cognitive changes.”
Neuropsychological features of DLB often include impairment of executive functions and visuospatial functions. “Early in the course of Alzheimer’s disease, typically performance on memory measures – especially delayed recall – are down and the other measures are borderline or mildly impaired,” Dr. Boeve noted. “By contrast, in DLB, attention, executive function, and visuospatial measures are down, but memory is often pretty good. What’s remarkable is that in the office setting, when you take a history the person often says, ‘I’m very forgetful,’ yet in the testing environment people tend to rise to the occasion pretty well.”
Imaging isn’t always helpful in establishing a diagnosis of DLB. MRI scans, for example, “can look pretty normal, including the hippocampi,” he said. “This is really the norm in DLB and it seems to be a disconnect. The person can have significant symptoms yet their MRI scan can be pretty normal.”
In Alzheimer’s disease, 18F-fluorodeoxyglucose-PET (FDG-PET) shows temporal, parietal, and frontal hypometabolism, sparing of the sensory-motor strip and sparing of the primary occipital cortex, while in DLB, FDG-PET shows marked deficits in the occipital regions with relative sparing of the frontal and temporal lobes. Another key neuroimaging sign of DLB is the posterior cingulate island sign, which is characterized by sparing of the posterior cingulate cortex relative to the precuneus plus cuneus on FDG-PET.
In 2017, the Dementia with Lewy Bodies Consortium published updated recommendations on the diagnosis and management of the disease (Neurology. 2017;89[1]:88-100). In its consensus report, the consortium defines probable DLB as dementia plus two or more clinical features or one core clinical feature plus one or more indicative biomarker. These biomarkers include reduced dopamine transport uptake in basal ganglia by SPECT or PET; abnormal (low uptake) meta-iodobenzylguanidine (MIBG) myocardial scintigraphy, and/or polysomnographic confirmation of REM sleep without atonia.
“Neuropathologically, limbic with or without neocortical Lewy bodies and Lewy neurites are the defining characteristics of pathologically-proven DLB,” added Dr. Boeve, a member of the DLB consortium. “The classic DLB phenotype can occur in limbic-predominant DLB. Lewy bodies in the neocortex are not necessary to cause a dementia syndrome.”
He characterized management of DLB as “very complicated. Consider symptoms as they relate to cognitive impairment, neuropsychiatric features, motor features, sleep disorders, and autonomic dysfunction.” He often asks the patient/family to prioritize the three most troublesome issues they seek to change, and develops a plan based on their input.
There is no Food and Drug Administration–approved medication for DLB, but the standard of care is an acetylcholinesterase inhibitor such as donepezil. “There is evidence that memantine can provide a modest benefit,” Dr. Boeve said. “Hypersomnia is quite prominent in DLB and worthy of assessing and treating.” Clinicians must weigh the pros and cons of pharmacotherapy with each patient. “For example, in the atypical neuroleptic class [of drugs], there may be a benefit to the hallucinations and delusions in DLB but hypersomnia can worsen,” he said. “Selecting agents is challenging but worth the effort.”
Survival is lower and more rapid with DLB, compared with Alzheimer’s. Most people pass away from primary DLB-related features or failure to thrive. The second most common is pneumonia or aspiration. Median survival was 4 years after diagnosis in one study, and end-of life discussions occurred in less than half of all patients. “This is a frustrating reminder that we as clinicians are not very good at discussing important topics such as end-of-life care with patients and their families,” Dr. Boeve said. Resources that he recommends for education and support include the Lewy Body Dementia Association and The Lewy Body Society.
At the 2016 Alzheimer’s Disease-Related Dementias Summit, clinicians formed a list of DLB research priorities (Neurology 2017;89[23]:2381-91). Among them were recommendations to “initiate clinical trials in diverse populations using therapies that address symptoms that have the greatest effect on patient function and caregiver burden” and “identify novel common and rare genetic variants, epigenetic changes, and environmental influences that affect the risk for and clinical features of” the disease.
Meanwhile, several research protocols are under way, including the development of a DLB module by the U.S. Alzheimer’s Research Disease Centers and a number of DLB-focused projects from the National Institute of Neurological Disorders and Stroke (NINDS) Parkinson’s Disease Biomarkers Program. In addition, the Lewy Body Dementia Association Research Centers of Excellence program is focused on optimizing clinical care and setting up the infrastructure for clinical trials, while the North American Prodromal Synucleinopathy Consortium is conducting longitudinal studies in those with REM sleep behavior disorder.
Dr. Boeve disclosed that he has been an investigator for clinical trials sponsored by GE Healthcare, Axovant, and Biogen. He is a member of the scientific advisory board for the Tau Consortium and has received research support from the National Institute on Aging, the NINDS, the Mangurian Foundation, and the Little Family Foundation.
ATLANTA – In the not-so-distant past, neurologists viewed dementia with Lewy bodies as a disorder primarily of the brain, but it turned out to be far more complex than that.
At the annual meeting of the American Neurological Association, Bradley F. Boeve, MD, described dementia with Lewy bodies (DLB) as a systemic neurologic disorder affecting the brain, including brain stem, spinal cord, and peripheral nervous system, especially the autonomic nervous system. “This leads to the complex array of clinical manifestations, which are quite different from patient to patient cross-sectionally and longitudinally,” said Dr. Boeve, the Little Family Foundation Professor of Lewy Body Dementia in the department of neurology at the Mayo Clinic, Rochester, Minn.
, he said. The four core clinical features are Parkinsonism unrelated to medications; recurrent, fully-formed visual hallucinations; fluctuations in cognition and/or arousal; and rapid eye movement (REM) sleep behavior disorder. “This is the most predictive of all four features,” Dr. Boeve said. He described REM sleep behavior disorder as a parasomnia manifested by the tendency to repeatedly “act out one’s dreams.” The dreams tend to contain a chasing/attacking theme, and behaviors mirror dream content. Injuries to the patient and bed partner can occur.
Typically, patients will present with REM sleep behavior disorder in their 50s and 60s, and sometimes in their 30s and 40s, “decades before cognitive changes begin,” he said. “This is usually followed by Parkinsonism and visual hallucinations. That’s the prototypical DLB [case], but there are many examples where this is not followed. Prominent neuropsychiatric features can also begin before any cognitive changes.”
Neuropsychological features of DLB often include impairment of executive functions and visuospatial functions. “Early in the course of Alzheimer’s disease, typically performance on memory measures – especially delayed recall – are down and the other measures are borderline or mildly impaired,” Dr. Boeve noted. “By contrast, in DLB, attention, executive function, and visuospatial measures are down, but memory is often pretty good. What’s remarkable is that in the office setting, when you take a history the person often says, ‘I’m very forgetful,’ yet in the testing environment people tend to rise to the occasion pretty well.”
Imaging isn’t always helpful in establishing a diagnosis of DLB. MRI scans, for example, “can look pretty normal, including the hippocampi,” he said. “This is really the norm in DLB and it seems to be a disconnect. The person can have significant symptoms yet their MRI scan can be pretty normal.”
In Alzheimer’s disease, 18F-fluorodeoxyglucose-PET (FDG-PET) shows temporal, parietal, and frontal hypometabolism, sparing of the sensory-motor strip and sparing of the primary occipital cortex, while in DLB, FDG-PET shows marked deficits in the occipital regions with relative sparing of the frontal and temporal lobes. Another key neuroimaging sign of DLB is the posterior cingulate island sign, which is characterized by sparing of the posterior cingulate cortex relative to the precuneus plus cuneus on FDG-PET.
In 2017, the Dementia with Lewy Bodies Consortium published updated recommendations on the diagnosis and management of the disease (Neurology. 2017;89[1]:88-100). In its consensus report, the consortium defines probable DLB as dementia plus two or more clinical features or one core clinical feature plus one or more indicative biomarker. These biomarkers include reduced dopamine transport uptake in basal ganglia by SPECT or PET; abnormal (low uptake) meta-iodobenzylguanidine (MIBG) myocardial scintigraphy, and/or polysomnographic confirmation of REM sleep without atonia.
“Neuropathologically, limbic with or without neocortical Lewy bodies and Lewy neurites are the defining characteristics of pathologically-proven DLB,” added Dr. Boeve, a member of the DLB consortium. “The classic DLB phenotype can occur in limbic-predominant DLB. Lewy bodies in the neocortex are not necessary to cause a dementia syndrome.”
He characterized management of DLB as “very complicated. Consider symptoms as they relate to cognitive impairment, neuropsychiatric features, motor features, sleep disorders, and autonomic dysfunction.” He often asks the patient/family to prioritize the three most troublesome issues they seek to change, and develops a plan based on their input.
There is no Food and Drug Administration–approved medication for DLB, but the standard of care is an acetylcholinesterase inhibitor such as donepezil. “There is evidence that memantine can provide a modest benefit,” Dr. Boeve said. “Hypersomnia is quite prominent in DLB and worthy of assessing and treating.” Clinicians must weigh the pros and cons of pharmacotherapy with each patient. “For example, in the atypical neuroleptic class [of drugs], there may be a benefit to the hallucinations and delusions in DLB but hypersomnia can worsen,” he said. “Selecting agents is challenging but worth the effort.”
Survival is lower and more rapid with DLB, compared with Alzheimer’s. Most people pass away from primary DLB-related features or failure to thrive. The second most common is pneumonia or aspiration. Median survival was 4 years after diagnosis in one study, and end-of life discussions occurred in less than half of all patients. “This is a frustrating reminder that we as clinicians are not very good at discussing important topics such as end-of-life care with patients and their families,” Dr. Boeve said. Resources that he recommends for education and support include the Lewy Body Dementia Association and The Lewy Body Society.
At the 2016 Alzheimer’s Disease-Related Dementias Summit, clinicians formed a list of DLB research priorities (Neurology 2017;89[23]:2381-91). Among them were recommendations to “initiate clinical trials in diverse populations using therapies that address symptoms that have the greatest effect on patient function and caregiver burden” and “identify novel common and rare genetic variants, epigenetic changes, and environmental influences that affect the risk for and clinical features of” the disease.
Meanwhile, several research protocols are under way, including the development of a DLB module by the U.S. Alzheimer’s Research Disease Centers and a number of DLB-focused projects from the National Institute of Neurological Disorders and Stroke (NINDS) Parkinson’s Disease Biomarkers Program. In addition, the Lewy Body Dementia Association Research Centers of Excellence program is focused on optimizing clinical care and setting up the infrastructure for clinical trials, while the North American Prodromal Synucleinopathy Consortium is conducting longitudinal studies in those with REM sleep behavior disorder.
Dr. Boeve disclosed that he has been an investigator for clinical trials sponsored by GE Healthcare, Axovant, and Biogen. He is a member of the scientific advisory board for the Tau Consortium and has received research support from the National Institute on Aging, the NINDS, the Mangurian Foundation, and the Little Family Foundation.
ATLANTA – In the not-so-distant past, neurologists viewed dementia with Lewy bodies as a disorder primarily of the brain, but it turned out to be far more complex than that.
At the annual meeting of the American Neurological Association, Bradley F. Boeve, MD, described dementia with Lewy bodies (DLB) as a systemic neurologic disorder affecting the brain, including brain stem, spinal cord, and peripheral nervous system, especially the autonomic nervous system. “This leads to the complex array of clinical manifestations, which are quite different from patient to patient cross-sectionally and longitudinally,” said Dr. Boeve, the Little Family Foundation Professor of Lewy Body Dementia in the department of neurology at the Mayo Clinic, Rochester, Minn.
, he said. The four core clinical features are Parkinsonism unrelated to medications; recurrent, fully-formed visual hallucinations; fluctuations in cognition and/or arousal; and rapid eye movement (REM) sleep behavior disorder. “This is the most predictive of all four features,” Dr. Boeve said. He described REM sleep behavior disorder as a parasomnia manifested by the tendency to repeatedly “act out one’s dreams.” The dreams tend to contain a chasing/attacking theme, and behaviors mirror dream content. Injuries to the patient and bed partner can occur.
Typically, patients will present with REM sleep behavior disorder in their 50s and 60s, and sometimes in their 30s and 40s, “decades before cognitive changes begin,” he said. “This is usually followed by Parkinsonism and visual hallucinations. That’s the prototypical DLB [case], but there are many examples where this is not followed. Prominent neuropsychiatric features can also begin before any cognitive changes.”
Neuropsychological features of DLB often include impairment of executive functions and visuospatial functions. “Early in the course of Alzheimer’s disease, typically performance on memory measures – especially delayed recall – are down and the other measures are borderline or mildly impaired,” Dr. Boeve noted. “By contrast, in DLB, attention, executive function, and visuospatial measures are down, but memory is often pretty good. What’s remarkable is that in the office setting, when you take a history the person often says, ‘I’m very forgetful,’ yet in the testing environment people tend to rise to the occasion pretty well.”
Imaging isn’t always helpful in establishing a diagnosis of DLB. MRI scans, for example, “can look pretty normal, including the hippocampi,” he said. “This is really the norm in DLB and it seems to be a disconnect. The person can have significant symptoms yet their MRI scan can be pretty normal.”
In Alzheimer’s disease, 18F-fluorodeoxyglucose-PET (FDG-PET) shows temporal, parietal, and frontal hypometabolism, sparing of the sensory-motor strip and sparing of the primary occipital cortex, while in DLB, FDG-PET shows marked deficits in the occipital regions with relative sparing of the frontal and temporal lobes. Another key neuroimaging sign of DLB is the posterior cingulate island sign, which is characterized by sparing of the posterior cingulate cortex relative to the precuneus plus cuneus on FDG-PET.
In 2017, the Dementia with Lewy Bodies Consortium published updated recommendations on the diagnosis and management of the disease (Neurology. 2017;89[1]:88-100). In its consensus report, the consortium defines probable DLB as dementia plus two or more clinical features or one core clinical feature plus one or more indicative biomarker. These biomarkers include reduced dopamine transport uptake in basal ganglia by SPECT or PET; abnormal (low uptake) meta-iodobenzylguanidine (MIBG) myocardial scintigraphy, and/or polysomnographic confirmation of REM sleep without atonia.
“Neuropathologically, limbic with or without neocortical Lewy bodies and Lewy neurites are the defining characteristics of pathologically-proven DLB,” added Dr. Boeve, a member of the DLB consortium. “The classic DLB phenotype can occur in limbic-predominant DLB. Lewy bodies in the neocortex are not necessary to cause a dementia syndrome.”
He characterized management of DLB as “very complicated. Consider symptoms as they relate to cognitive impairment, neuropsychiatric features, motor features, sleep disorders, and autonomic dysfunction.” He often asks the patient/family to prioritize the three most troublesome issues they seek to change, and develops a plan based on their input.
There is no Food and Drug Administration–approved medication for DLB, but the standard of care is an acetylcholinesterase inhibitor such as donepezil. “There is evidence that memantine can provide a modest benefit,” Dr. Boeve said. “Hypersomnia is quite prominent in DLB and worthy of assessing and treating.” Clinicians must weigh the pros and cons of pharmacotherapy with each patient. “For example, in the atypical neuroleptic class [of drugs], there may be a benefit to the hallucinations and delusions in DLB but hypersomnia can worsen,” he said. “Selecting agents is challenging but worth the effort.”
Survival is lower and more rapid with DLB, compared with Alzheimer’s. Most people pass away from primary DLB-related features or failure to thrive. The second most common is pneumonia or aspiration. Median survival was 4 years after diagnosis in one study, and end-of life discussions occurred in less than half of all patients. “This is a frustrating reminder that we as clinicians are not very good at discussing important topics such as end-of-life care with patients and their families,” Dr. Boeve said. Resources that he recommends for education and support include the Lewy Body Dementia Association and The Lewy Body Society.
At the 2016 Alzheimer’s Disease-Related Dementias Summit, clinicians formed a list of DLB research priorities (Neurology 2017;89[23]:2381-91). Among them were recommendations to “initiate clinical trials in diverse populations using therapies that address symptoms that have the greatest effect on patient function and caregiver burden” and “identify novel common and rare genetic variants, epigenetic changes, and environmental influences that affect the risk for and clinical features of” the disease.
Meanwhile, several research protocols are under way, including the development of a DLB module by the U.S. Alzheimer’s Research Disease Centers and a number of DLB-focused projects from the National Institute of Neurological Disorders and Stroke (NINDS) Parkinson’s Disease Biomarkers Program. In addition, the Lewy Body Dementia Association Research Centers of Excellence program is focused on optimizing clinical care and setting up the infrastructure for clinical trials, while the North American Prodromal Synucleinopathy Consortium is conducting longitudinal studies in those with REM sleep behavior disorder.
Dr. Boeve disclosed that he has been an investigator for clinical trials sponsored by GE Healthcare, Axovant, and Biogen. He is a member of the scientific advisory board for the Tau Consortium and has received research support from the National Institute on Aging, the NINDS, the Mangurian Foundation, and the Little Family Foundation.
EXPERT ANALYSIS FROM ANA 2018
The teenager who couldn’t stay awake
CASE Somnolent, confused, and hungry
Mr. G, age 14, presents to the emergency department (ED) for acute-onset hypersomnia that has gradually worsened over the course of a few days. Mr. G now sleeps most of the day, has altered mental status, and is experiencing emotional dysregulation with no clear etiology. His mother, who accompanies him to the ED, says that prior to the onset of these symptoms her son had been healthy. She notes that he has been eating more than usual, which she assumes is due to a growth spurt.
Mr. G’s symptoms began 4 days ago when he became increasingly fatigued, sleeping for 11 to 12 hours per day, with intermittent episodes of staring and unresponsiveness from which he rapidly returned to baseline. During the next 3 days, he became more confused and somnolent, and began to display bizarre behavior, including eating food out of the trash and attempting to microwave a full metal pot. He exhibited unexplained crying spells, calling out for his “mommy,” and saying he was “afraid I’m dying.”
During the 2 days before he came to our ED, Mr. G was seen at 2 other hospitals. Following extensive imaging and laboratory work-up, clinicians at these facilities attributed his symptoms to intoxication from an unknown substance. Mr. G has a history of marijuana use, and his mother reports that his friends had recently been using synthetic marijuana. However, no intoxicant was identified on urine or gas chromatography drug screening.
Mr. G’s history includes oppositional behavior, and a brief psychiatric hospitalization at age 5 for aggression. He has otherwise been healthy. His family history is significant for maternal substance use and anxiety disorders. In addition to sporadic cannabis use, Mr. G’s social history includes multiple recent family losses, previous foster care placement, and recent declining academic performance.
[polldaddy:10148168]
EVALUATION No red flags
On admission, Mr. G appears somnolent and displays disorganized speech, impulsivity, frequent disorientation, and intermittent agitation/anxiety; he sleeps 16 to 18 hours per day. Mr. G is admitted with a presumptive diagnosis of substance intoxication and transferred to the general pediatric inpatient unit. Upon arrival, he is found to be bradycardic (42 beats per minute), although afebrile with otherwise age-appropriate vitals. On exam, he is somnolent but arousable and follows simple commands.
Continue to: Mr. G undergoes a Monospot test...
Mr. G undergoes a Monospot test, which is positive, with subsequent evidence of a prior, but not active, Epstein-Barr virus (EBV) infection. He also has a mildly elevated CSF protein level, but subsequent CSF labs are negative for both infectious and non-infectious processes. An EEG reveals focal neuronal slowing.
During brief periods of wakefulness, Mr. G calls out to his mother and says, “I’m not going to make it to my birthday,” and “You’re going to have to let me go.” He occasionally becomes combative, pulling at IV lines and swearing at staff. His bradycardia resolves without intervention during his admission. On Day 8 of his hospitalization, Mr. G displays hypersexuality and makes sexually suggestive comments toward female staff members. He also experiences recurrence of hyperphagia.
On Day 10 of his stay on the pediatric unit, because of the emergence of hypersexuality and hyperphagia, along with a largely negative medical evaluation, Mr. G is transferred to the pediatric psychiatric unit for ongoing evaluation and management.
[polldaddy:10148172]
The authors’ observations
Given Mr. G’s rapid onset of confusion, hypersomnia, and emotional dysregulation, our differential diagnosis included delirium of unclear etiology, substance intoxication, autoimmune encephalitis, viral meningitis, heavy metal intoxication, primary psychotic disorder, and KLS. Mr. G underwent an extensive diagnostic evaluation, which was largely unremarkable (Table). He had a mildly elevated CSF protein level, but subsequent CSF labs were negative for both infectious and non-infectious processes. When Mr. G was transferred to the pediatric inpatient psychiatric unit on Day 10, the presumptive diagnosis was KLS.
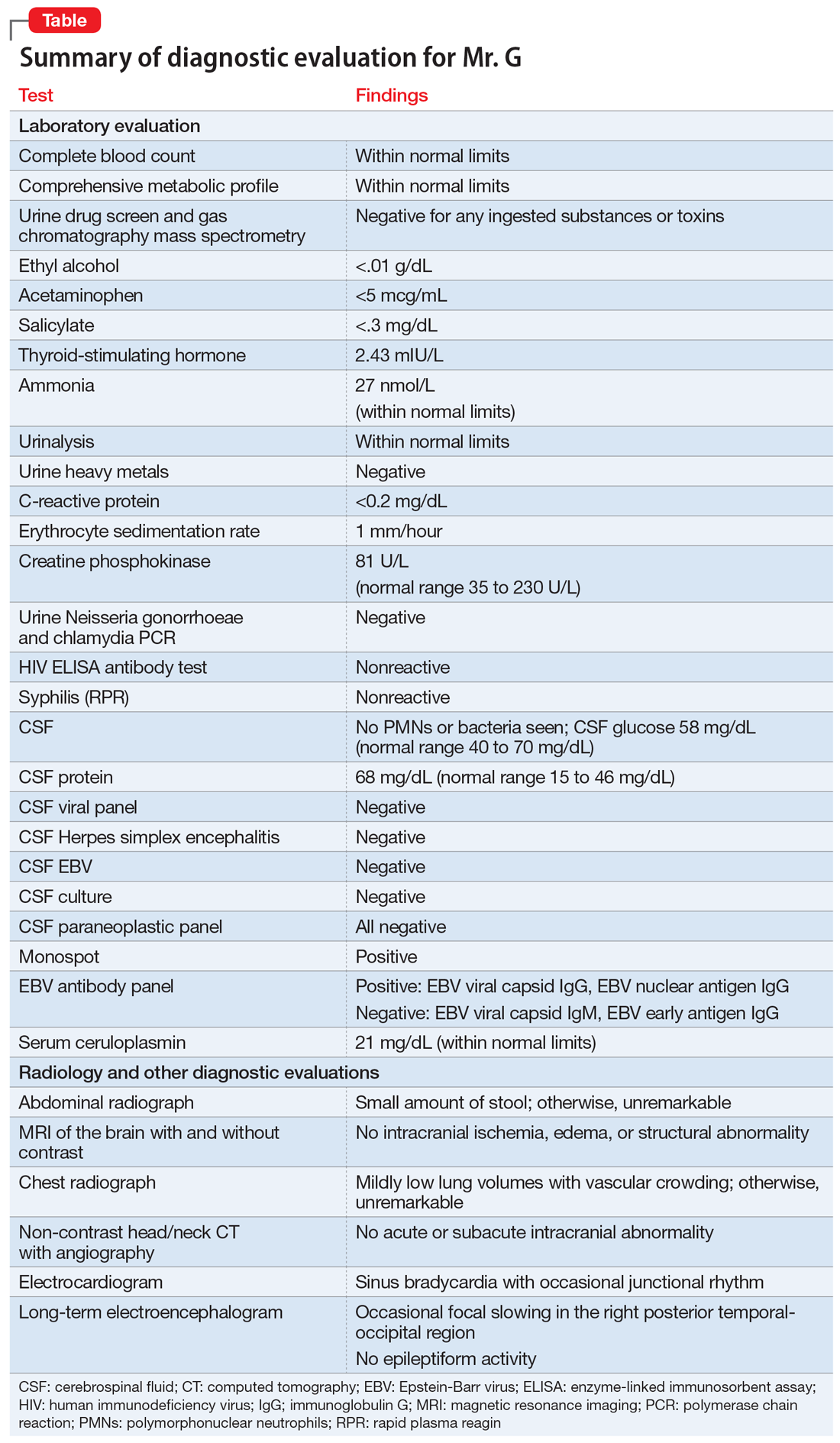
KLS is a rare neurologic disorder, with an incidence of 1 to 5 in 1 million and a 4:1 male-to-female predominance.1 It poses a diagnostic challenge due to its low prevalence and broad differential. The disorder typically presents in early adolescence and is characterized by episodes of severe hypersomnia with associated cognitive and/or behavioral disturbance2 (Box2-4). Bradycardia, as seen in Mr. G, and other forms of autonomic dysregulation have been reported in the literature, as has the focal neuronal slowing noted on Mr. G’s EEG.3

[polldaddy:10148174]
Continue to: TREATMENT Methylphenidate and a safety plan
TREATMENT Methylphenidate and a safety plan
On Day 11 of hospitalization, Mr. G is started on methylphenidate, 10 mg in the morning and 5 mg in the afternoon. After starting methylphenidate, he sustains more regular wakefulness, with improved thought organization, engagement, and fewer disruptive behaviors. He receives infrequent, as-needed doses of olanzapine, and by Day 14, he returns to his baseline behavior and cognition.
A safety plan is created for the family to address worsening symptoms or future episodes. The safety plan is developed with Mr. G and input from his family. It is to be administered in all settings and we particularly emphasized using it in the school setting, where staff may not be familiar with KLS. The safety plan involves a description of KLS, its symptoms, the risks for hypersomnolence, hypersexuality, and psychotic symptoms or behavioral dysregulation. It stresses close supervision of Mr. G, not allowing him to be unsupervised or unchaperoned on school trips or other outings, and lethal means restriction. It outlines a detailed plan if Mr. G’s behavior decompensates or escalates, including a step-wise approach to engaging psychological interventions and mental health resources, and securing crisis services as needed.
On Day 15, he is discharged to home in stable condition with outpatient mental health follow-up and continues to take the prescribed methylphenidate.
The authors’ observations
Management of KLS is primarily supportive. Stimulants may help reduce hypersomnia, impulsivity, and inattention early in the disease course.1 However, in a systematic review, 89% of patients with KLS who received methylphenidate experienced worsening or no improvement, and 11% showed only partial improvement.2 Amantadine was more promising, with 29% of patients with KLS showing partial benefit and 12% showing significant benefit.2 Multiple other pharmacologic agents have been described with varying efficacy, including lithium, valproate, risperidone, bupropion, and immunoglobulins.2 Furthermore, lithium and valproate have been suggested to be helpful in preventing recurrences in some cases.6
The circumstances surrounding Mr. G’s symptom onset are unclear and may have been multifactorial. It is possible that his prior EBV infection was a trigger for this KLS-associated episode, as EBV is a known precipitant for KLS episodes.3 Mr. G’s history of cannabis use may also have served as an early trigger for KLS.3
This case highlights the importance of multidisciplinary collaboration in a diagnostically challenging case. It emphasizes the need for a broad differential and the importance of challenging a previous diagnosis in the face of mounting evidence to the contrary. In this case, the patient’s history of irritability, aggression, and cannabis use resulted in multiple clinicians misattributing his symptoms to substance use or a primary psychiatric disorder. However, given his symptom acuity, progression, and the lack of findings on diagnostic evaluation to explain his presentation, these initial diagnoses did not explain the severity, nature, or duration of his symptoms. Keeping KLS in the differential is particularly important for patients with a prior history of psychiatric illness or substance use, because these patients are at higher risk for misattribution of symptoms to pre-existing psychiatric illness. Evolution of symptoms, a negative diagnostic evaluation, and maintaining a broad differential resulted in eventually reaching the final diagnosis of KLS and development of a longitudinal management plan.
While further work must be done to clearly define the pharmacologic approach to acute management of KLS episodes, nonpharmacologic aspects of care must not be neglected. Behavioral planning, adjustment of the environment, engagement with schools/community supports, and family education are valuable tools for facilitating the patient’s de-escalation, avoiding unneeded polypharmacy, reducing anxieties, and safeguarding the patient from unnecessary harm.7 Clinicians can support their patients’ transitions back into the community by ensuring careful outpatient follow-up for symptom monitoring and by communicating with patients’ schools and employers.
OUTCOME Asymptomatic; no recurrence of symptoms
Forty-six days after his symptoms began and 31 days after hospital discharge, Mr. G is asymptomatic with no recurrence of symptoms.
Bottom Line
Kleine-Levin syndrome (KLS) is a rare, often-overlooked condition that should be considered in the differential diagnosis for patients who present with hypersomnolence and altered mental status without a clear etiology. Rapid recognition of KLS can prevent misattribution of symptoms, unnecessary treatment, and missed opportunities for care.
1. Billiard M, Jaussent I, Dauvilliers Y, et al. Recurrent hypersomnia: a review of 339 cases. Sleep Med. 2011;15(4):247-257.
2. Arnulf I, Lin L, Gadoth N, et al. Kleine-Levin syndrome: a systematic study of 108 patients. Ann Neurol. 2008;63(4):482-493.
3. Arnulf I. Kleine-Levin syndrome: a systematic review of 186 cases in the literature. Brain. 2005;128(12):2763-2776.
4. Lisk R. Kleine-Levin syndrome. Pract Neurol. 2009;9(1);42-45.
5. de Araújo Lima TF, da Silva Behrens NS, Lopes E, et al. Kleine–Levin Syndrome: a case report. Sleep Sci. 2014;7(2):122-125.
6. Goldberg MA. The treatment of Kleine-Levin syndrome with lithium. Can J Psychiatry. 1983;28:491-493.
7. Gadoth N, Kesler A, Vainstein G, et al. Clinical and polysomnographic characteristics of 34 patients with Kleine-Levin syndrome. J Sleep Res. 2001;10(4):337-341.
CASE Somnolent, confused, and hungry
Mr. G, age 14, presents to the emergency department (ED) for acute-onset hypersomnia that has gradually worsened over the course of a few days. Mr. G now sleeps most of the day, has altered mental status, and is experiencing emotional dysregulation with no clear etiology. His mother, who accompanies him to the ED, says that prior to the onset of these symptoms her son had been healthy. She notes that he has been eating more than usual, which she assumes is due to a growth spurt.
Mr. G’s symptoms began 4 days ago when he became increasingly fatigued, sleeping for 11 to 12 hours per day, with intermittent episodes of staring and unresponsiveness from which he rapidly returned to baseline. During the next 3 days, he became more confused and somnolent, and began to display bizarre behavior, including eating food out of the trash and attempting to microwave a full metal pot. He exhibited unexplained crying spells, calling out for his “mommy,” and saying he was “afraid I’m dying.”
During the 2 days before he came to our ED, Mr. G was seen at 2 other hospitals. Following extensive imaging and laboratory work-up, clinicians at these facilities attributed his symptoms to intoxication from an unknown substance. Mr. G has a history of marijuana use, and his mother reports that his friends had recently been using synthetic marijuana. However, no intoxicant was identified on urine or gas chromatography drug screening.
Mr. G’s history includes oppositional behavior, and a brief psychiatric hospitalization at age 5 for aggression. He has otherwise been healthy. His family history is significant for maternal substance use and anxiety disorders. In addition to sporadic cannabis use, Mr. G’s social history includes multiple recent family losses, previous foster care placement, and recent declining academic performance.
[polldaddy:10148168]
EVALUATION No red flags
On admission, Mr. G appears somnolent and displays disorganized speech, impulsivity, frequent disorientation, and intermittent agitation/anxiety; he sleeps 16 to 18 hours per day. Mr. G is admitted with a presumptive diagnosis of substance intoxication and transferred to the general pediatric inpatient unit. Upon arrival, he is found to be bradycardic (42 beats per minute), although afebrile with otherwise age-appropriate vitals. On exam, he is somnolent but arousable and follows simple commands.
Continue to: Mr. G undergoes a Monospot test...
Mr. G undergoes a Monospot test, which is positive, with subsequent evidence of a prior, but not active, Epstein-Barr virus (EBV) infection. He also has a mildly elevated CSF protein level, but subsequent CSF labs are negative for both infectious and non-infectious processes. An EEG reveals focal neuronal slowing.
During brief periods of wakefulness, Mr. G calls out to his mother and says, “I’m not going to make it to my birthday,” and “You’re going to have to let me go.” He occasionally becomes combative, pulling at IV lines and swearing at staff. His bradycardia resolves without intervention during his admission. On Day 8 of his hospitalization, Mr. G displays hypersexuality and makes sexually suggestive comments toward female staff members. He also experiences recurrence of hyperphagia.
On Day 10 of his stay on the pediatric unit, because of the emergence of hypersexuality and hyperphagia, along with a largely negative medical evaluation, Mr. G is transferred to the pediatric psychiatric unit for ongoing evaluation and management.
[polldaddy:10148172]
The authors’ observations
Given Mr. G’s rapid onset of confusion, hypersomnia, and emotional dysregulation, our differential diagnosis included delirium of unclear etiology, substance intoxication, autoimmune encephalitis, viral meningitis, heavy metal intoxication, primary psychotic disorder, and KLS. Mr. G underwent an extensive diagnostic evaluation, which was largely unremarkable (Table). He had a mildly elevated CSF protein level, but subsequent CSF labs were negative for both infectious and non-infectious processes. When Mr. G was transferred to the pediatric inpatient psychiatric unit on Day 10, the presumptive diagnosis was KLS.

KLS is a rare neurologic disorder, with an incidence of 1 to 5 in 1 million and a 4:1 male-to-female predominance.1 It poses a diagnostic challenge due to its low prevalence and broad differential. The disorder typically presents in early adolescence and is characterized by episodes of severe hypersomnia with associated cognitive and/or behavioral disturbance2 (Box2-4). Bradycardia, as seen in Mr. G, and other forms of autonomic dysregulation have been reported in the literature, as has the focal neuronal slowing noted on Mr. G’s EEG.3

[polldaddy:10148174]
Continue to: TREATMENT Methylphenidate and a safety plan
TREATMENT Methylphenidate and a safety plan
On Day 11 of hospitalization, Mr. G is started on methylphenidate, 10 mg in the morning and 5 mg in the afternoon. After starting methylphenidate, he sustains more regular wakefulness, with improved thought organization, engagement, and fewer disruptive behaviors. He receives infrequent, as-needed doses of olanzapine, and by Day 14, he returns to his baseline behavior and cognition.
A safety plan is created for the family to address worsening symptoms or future episodes. The safety plan is developed with Mr. G and input from his family. It is to be administered in all settings and we particularly emphasized using it in the school setting, where staff may not be familiar with KLS. The safety plan involves a description of KLS, its symptoms, the risks for hypersomnolence, hypersexuality, and psychotic symptoms or behavioral dysregulation. It stresses close supervision of Mr. G, not allowing him to be unsupervised or unchaperoned on school trips or other outings, and lethal means restriction. It outlines a detailed plan if Mr. G’s behavior decompensates or escalates, including a step-wise approach to engaging psychological interventions and mental health resources, and securing crisis services as needed.
On Day 15, he is discharged to home in stable condition with outpatient mental health follow-up and continues to take the prescribed methylphenidate.
The authors’ observations
Management of KLS is primarily supportive. Stimulants may help reduce hypersomnia, impulsivity, and inattention early in the disease course.1 However, in a systematic review, 89% of patients with KLS who received methylphenidate experienced worsening or no improvement, and 11% showed only partial improvement.2 Amantadine was more promising, with 29% of patients with KLS showing partial benefit and 12% showing significant benefit.2 Multiple other pharmacologic agents have been described with varying efficacy, including lithium, valproate, risperidone, bupropion, and immunoglobulins.2 Furthermore, lithium and valproate have been suggested to be helpful in preventing recurrences in some cases.6
The circumstances surrounding Mr. G’s symptom onset are unclear and may have been multifactorial. It is possible that his prior EBV infection was a trigger for this KLS-associated episode, as EBV is a known precipitant for KLS episodes.3 Mr. G’s history of cannabis use may also have served as an early trigger for KLS.3
This case highlights the importance of multidisciplinary collaboration in a diagnostically challenging case. It emphasizes the need for a broad differential and the importance of challenging a previous diagnosis in the face of mounting evidence to the contrary. In this case, the patient’s history of irritability, aggression, and cannabis use resulted in multiple clinicians misattributing his symptoms to substance use or a primary psychiatric disorder. However, given his symptom acuity, progression, and the lack of findings on diagnostic evaluation to explain his presentation, these initial diagnoses did not explain the severity, nature, or duration of his symptoms. Keeping KLS in the differential is particularly important for patients with a prior history of psychiatric illness or substance use, because these patients are at higher risk for misattribution of symptoms to pre-existing psychiatric illness. Evolution of symptoms, a negative diagnostic evaluation, and maintaining a broad differential resulted in eventually reaching the final diagnosis of KLS and development of a longitudinal management plan.
While further work must be done to clearly define the pharmacologic approach to acute management of KLS episodes, nonpharmacologic aspects of care must not be neglected. Behavioral planning, adjustment of the environment, engagement with schools/community supports, and family education are valuable tools for facilitating the patient’s de-escalation, avoiding unneeded polypharmacy, reducing anxieties, and safeguarding the patient from unnecessary harm.7 Clinicians can support their patients’ transitions back into the community by ensuring careful outpatient follow-up for symptom monitoring and by communicating with patients’ schools and employers.
OUTCOME Asymptomatic; no recurrence of symptoms
Forty-six days after his symptoms began and 31 days after hospital discharge, Mr. G is asymptomatic with no recurrence of symptoms.
Bottom Line
Kleine-Levin syndrome (KLS) is a rare, often-overlooked condition that should be considered in the differential diagnosis for patients who present with hypersomnolence and altered mental status without a clear etiology. Rapid recognition of KLS can prevent misattribution of symptoms, unnecessary treatment, and missed opportunities for care.
CASE Somnolent, confused, and hungry
Mr. G, age 14, presents to the emergency department (ED) for acute-onset hypersomnia that has gradually worsened over the course of a few days. Mr. G now sleeps most of the day, has altered mental status, and is experiencing emotional dysregulation with no clear etiology. His mother, who accompanies him to the ED, says that prior to the onset of these symptoms her son had been healthy. She notes that he has been eating more than usual, which she assumes is due to a growth spurt.
Mr. G’s symptoms began 4 days ago when he became increasingly fatigued, sleeping for 11 to 12 hours per day, with intermittent episodes of staring and unresponsiveness from which he rapidly returned to baseline. During the next 3 days, he became more confused and somnolent, and began to display bizarre behavior, including eating food out of the trash and attempting to microwave a full metal pot. He exhibited unexplained crying spells, calling out for his “mommy,” and saying he was “afraid I’m dying.”
During the 2 days before he came to our ED, Mr. G was seen at 2 other hospitals. Following extensive imaging and laboratory work-up, clinicians at these facilities attributed his symptoms to intoxication from an unknown substance. Mr. G has a history of marijuana use, and his mother reports that his friends had recently been using synthetic marijuana. However, no intoxicant was identified on urine or gas chromatography drug screening.
Mr. G’s history includes oppositional behavior, and a brief psychiatric hospitalization at age 5 for aggression. He has otherwise been healthy. His family history is significant for maternal substance use and anxiety disorders. In addition to sporadic cannabis use, Mr. G’s social history includes multiple recent family losses, previous foster care placement, and recent declining academic performance.
[polldaddy:10148168]
EVALUATION No red flags
On admission, Mr. G appears somnolent and displays disorganized speech, impulsivity, frequent disorientation, and intermittent agitation/anxiety; he sleeps 16 to 18 hours per day. Mr. G is admitted with a presumptive diagnosis of substance intoxication and transferred to the general pediatric inpatient unit. Upon arrival, he is found to be bradycardic (42 beats per minute), although afebrile with otherwise age-appropriate vitals. On exam, he is somnolent but arousable and follows simple commands.
Continue to: Mr. G undergoes a Monospot test...
Mr. G undergoes a Monospot test, which is positive, with subsequent evidence of a prior, but not active, Epstein-Barr virus (EBV) infection. He also has a mildly elevated CSF protein level, but subsequent CSF labs are negative for both infectious and non-infectious processes. An EEG reveals focal neuronal slowing.
During brief periods of wakefulness, Mr. G calls out to his mother and says, “I’m not going to make it to my birthday,” and “You’re going to have to let me go.” He occasionally becomes combative, pulling at IV lines and swearing at staff. His bradycardia resolves without intervention during his admission. On Day 8 of his hospitalization, Mr. G displays hypersexuality and makes sexually suggestive comments toward female staff members. He also experiences recurrence of hyperphagia.
On Day 10 of his stay on the pediatric unit, because of the emergence of hypersexuality and hyperphagia, along with a largely negative medical evaluation, Mr. G is transferred to the pediatric psychiatric unit for ongoing evaluation and management.
[polldaddy:10148172]
The authors’ observations
Given Mr. G’s rapid onset of confusion, hypersomnia, and emotional dysregulation, our differential diagnosis included delirium of unclear etiology, substance intoxication, autoimmune encephalitis, viral meningitis, heavy metal intoxication, primary psychotic disorder, and KLS. Mr. G underwent an extensive diagnostic evaluation, which was largely unremarkable (Table). He had a mildly elevated CSF protein level, but subsequent CSF labs were negative for both infectious and non-infectious processes. When Mr. G was transferred to the pediatric inpatient psychiatric unit on Day 10, the presumptive diagnosis was KLS.

KLS is a rare neurologic disorder, with an incidence of 1 to 5 in 1 million and a 4:1 male-to-female predominance.1 It poses a diagnostic challenge due to its low prevalence and broad differential. The disorder typically presents in early adolescence and is characterized by episodes of severe hypersomnia with associated cognitive and/or behavioral disturbance2 (Box2-4). Bradycardia, as seen in Mr. G, and other forms of autonomic dysregulation have been reported in the literature, as has the focal neuronal slowing noted on Mr. G’s EEG.3

[polldaddy:10148174]
Continue to: TREATMENT Methylphenidate and a safety plan
TREATMENT Methylphenidate and a safety plan
On Day 11 of hospitalization, Mr. G is started on methylphenidate, 10 mg in the morning and 5 mg in the afternoon. After starting methylphenidate, he sustains more regular wakefulness, with improved thought organization, engagement, and fewer disruptive behaviors. He receives infrequent, as-needed doses of olanzapine, and by Day 14, he returns to his baseline behavior and cognition.
A safety plan is created for the family to address worsening symptoms or future episodes. The safety plan is developed with Mr. G and input from his family. It is to be administered in all settings and we particularly emphasized using it in the school setting, where staff may not be familiar with KLS. The safety plan involves a description of KLS, its symptoms, the risks for hypersomnolence, hypersexuality, and psychotic symptoms or behavioral dysregulation. It stresses close supervision of Mr. G, not allowing him to be unsupervised or unchaperoned on school trips or other outings, and lethal means restriction. It outlines a detailed plan if Mr. G’s behavior decompensates or escalates, including a step-wise approach to engaging psychological interventions and mental health resources, and securing crisis services as needed.
On Day 15, he is discharged to home in stable condition with outpatient mental health follow-up and continues to take the prescribed methylphenidate.
The authors’ observations
Management of KLS is primarily supportive. Stimulants may help reduce hypersomnia, impulsivity, and inattention early in the disease course.1 However, in a systematic review, 89% of patients with KLS who received methylphenidate experienced worsening or no improvement, and 11% showed only partial improvement.2 Amantadine was more promising, with 29% of patients with KLS showing partial benefit and 12% showing significant benefit.2 Multiple other pharmacologic agents have been described with varying efficacy, including lithium, valproate, risperidone, bupropion, and immunoglobulins.2 Furthermore, lithium and valproate have been suggested to be helpful in preventing recurrences in some cases.6
The circumstances surrounding Mr. G’s symptom onset are unclear and may have been multifactorial. It is possible that his prior EBV infection was a trigger for this KLS-associated episode, as EBV is a known precipitant for KLS episodes.3 Mr. G’s history of cannabis use may also have served as an early trigger for KLS.3
This case highlights the importance of multidisciplinary collaboration in a diagnostically challenging case. It emphasizes the need for a broad differential and the importance of challenging a previous diagnosis in the face of mounting evidence to the contrary. In this case, the patient’s history of irritability, aggression, and cannabis use resulted in multiple clinicians misattributing his symptoms to substance use or a primary psychiatric disorder. However, given his symptom acuity, progression, and the lack of findings on diagnostic evaluation to explain his presentation, these initial diagnoses did not explain the severity, nature, or duration of his symptoms. Keeping KLS in the differential is particularly important for patients with a prior history of psychiatric illness or substance use, because these patients are at higher risk for misattribution of symptoms to pre-existing psychiatric illness. Evolution of symptoms, a negative diagnostic evaluation, and maintaining a broad differential resulted in eventually reaching the final diagnosis of KLS and development of a longitudinal management plan.
While further work must be done to clearly define the pharmacologic approach to acute management of KLS episodes, nonpharmacologic aspects of care must not be neglected. Behavioral planning, adjustment of the environment, engagement with schools/community supports, and family education are valuable tools for facilitating the patient’s de-escalation, avoiding unneeded polypharmacy, reducing anxieties, and safeguarding the patient from unnecessary harm.7 Clinicians can support their patients’ transitions back into the community by ensuring careful outpatient follow-up for symptom monitoring and by communicating with patients’ schools and employers.
OUTCOME Asymptomatic; no recurrence of symptoms
Forty-six days after his symptoms began and 31 days after hospital discharge, Mr. G is asymptomatic with no recurrence of symptoms.
Bottom Line
Kleine-Levin syndrome (KLS) is a rare, often-overlooked condition that should be considered in the differential diagnosis for patients who present with hypersomnolence and altered mental status without a clear etiology. Rapid recognition of KLS can prevent misattribution of symptoms, unnecessary treatment, and missed opportunities for care.
1. Billiard M, Jaussent I, Dauvilliers Y, et al. Recurrent hypersomnia: a review of 339 cases. Sleep Med. 2011;15(4):247-257.
2. Arnulf I, Lin L, Gadoth N, et al. Kleine-Levin syndrome: a systematic study of 108 patients. Ann Neurol. 2008;63(4):482-493.
3. Arnulf I. Kleine-Levin syndrome: a systematic review of 186 cases in the literature. Brain. 2005;128(12):2763-2776.
4. Lisk R. Kleine-Levin syndrome. Pract Neurol. 2009;9(1);42-45.
5. de Araújo Lima TF, da Silva Behrens NS, Lopes E, et al. Kleine–Levin Syndrome: a case report. Sleep Sci. 2014;7(2):122-125.
6. Goldberg MA. The treatment of Kleine-Levin syndrome with lithium. Can J Psychiatry. 1983;28:491-493.
7. Gadoth N, Kesler A, Vainstein G, et al. Clinical and polysomnographic characteristics of 34 patients with Kleine-Levin syndrome. J Sleep Res. 2001;10(4):337-341.
1. Billiard M, Jaussent I, Dauvilliers Y, et al. Recurrent hypersomnia: a review of 339 cases. Sleep Med. 2011;15(4):247-257.
2. Arnulf I, Lin L, Gadoth N, et al. Kleine-Levin syndrome: a systematic study of 108 patients. Ann Neurol. 2008;63(4):482-493.
3. Arnulf I. Kleine-Levin syndrome: a systematic review of 186 cases in the literature. Brain. 2005;128(12):2763-2776.
4. Lisk R. Kleine-Levin syndrome. Pract Neurol. 2009;9(1);42-45.
5. de Araújo Lima TF, da Silva Behrens NS, Lopes E, et al. Kleine–Levin Syndrome: a case report. Sleep Sci. 2014;7(2):122-125.
6. Goldberg MA. The treatment of Kleine-Levin syndrome with lithium. Can J Psychiatry. 1983;28:491-493.
7. Gadoth N, Kesler A, Vainstein G, et al. Clinical and polysomnographic characteristics of 34 patients with Kleine-Levin syndrome. J Sleep Res. 2001;10(4):337-341.
Obstructive sleep apnea: A better Dx model for primary care
ABSTRACT
Purpose To derive a predictive model for obstructive sleep apnea (OSA) in primary care practice, using home-based overnight oximetry results to refine posttest probability (PTP) of disease after initial risk stratification with the Sleep Apnea Clinical Score (SACS).
Methods We performed secondary analyses on data from a SACS validation cohort, to compare the diagnostic accuracy of 3 overnight oximetry measurements (oxygen desaturation index [ODI], mean saturation, and minimum saturation) in predicting OSA. Receiver operator characteristics (ROC) were computed for each measurement independently and sequentially after risk stratifying with SACS. We examined the implications of oximetry results for OSA PTP for participants categorized as intermediate risk (SACS 6-14; 66/191 participants [35%]; OSA probability 41%). We calculated positive likelihood ratios (LR) for multiple ODI results and determined which ones allowed recalibration to high- or low-risk PTP.
Results Among the 3 oximetry findings, ODI best predicted OSA (area under the curve [AUC], 0.88; 95% confidence interval [CI], 0.83-0.93). An ODI ≥8.4 (likelihood ratio [LR], 4.19; 95% CI, 2.87-6.10) created a PTP of 77%, while an ODI of 0 to <8.4 (LR, 0.19, 95% CI, 0.12-0.33) created a 14% PTP. Sequential application of SACS and ODI results yielded an AUC result of 0.90 (95% CI, 0.85-0.95).
Conclusions SACS risk stratification provides an advantage over clinical gestalt. In those at intermediate risk, ODI results provide a simple and clinically useful way to further refine diagnostic prediction. Sequential use of SACS and selectively employed overnight oximetry may limit unnecessary polysomnography. Oximetry testing should be avoided in patients deemed low or high risk by SACS, as positive results do not substantially recalibrate risk.
Obstructive sleep apnea (OSA) is a prevalent and underdiagnosed condition. The National Sleep Foundation estimates that 18 million Americans have OSA.1 Primary care practice may be the best setting in which to identify OSA, as many of our patients have conditions frequently associated with apnea (eg, hypertension, obesity, diabetes, arrhythmia, and neurologic illness). Up to a third of patients in primary care practice may be at increased risk.2,3
Clinical guidelines of the American Academy of Sleep Medicine (AASM) recommend obtaining a sleep history to evaluate for possible OSA in 3 instances: as part of a routine health maintenance examination, during evaluation of specific complaints associated with OSA (eg, snoring, apnea, daytime sleepiness), and during comprehensive evaluations for individuals with high-risk conditions (ie, obesity, congestive heart failure, refractory hypertension, diabetes, stroke history).4
The American College of Physicians (ACP) Clinical Practice Guideline suggests assessing individuals who have unexplained daytime sleepiness.5 The ACP considers this assessment “High-Value Care,” as “evidence shows that before diagnosis, patients with OSA have higher rates of health care use, more frequent and longer hospital stays, and higher health care costs than after diagnosis.”5
Continue to: We recently validated the diagnostic accuracy...
We recently validated the diagnostic accuracy of the Sleep Apnea Clinical Score (SACS) for use in a primary care patient population suspected of having OSA.6 SACS uses historical and clinical data to derive a score that identifies a patient’s risk level.7 However, as an alternative to the 2 levels described in Flemons’ SACS,7 we propose creating 3 risk strata (FIGURE 17,8). We believe that patients at high risk (SACS ≥15) should be encouraged to undergo sleep evaluations as their posttest probability (PTP) of OSA is 75% to 80%. Individuals at low risk (SACS ≤5; PTP <20%) could receive lifestyle advice and simple clinical interventions that decrease symptoms (eg, weight loss, increased physical activity, sleeping on one’s side). For low-risk patients, clinical observation and reevaluation could take place over time with their primary care provider, without additional testing or referral to specialists.
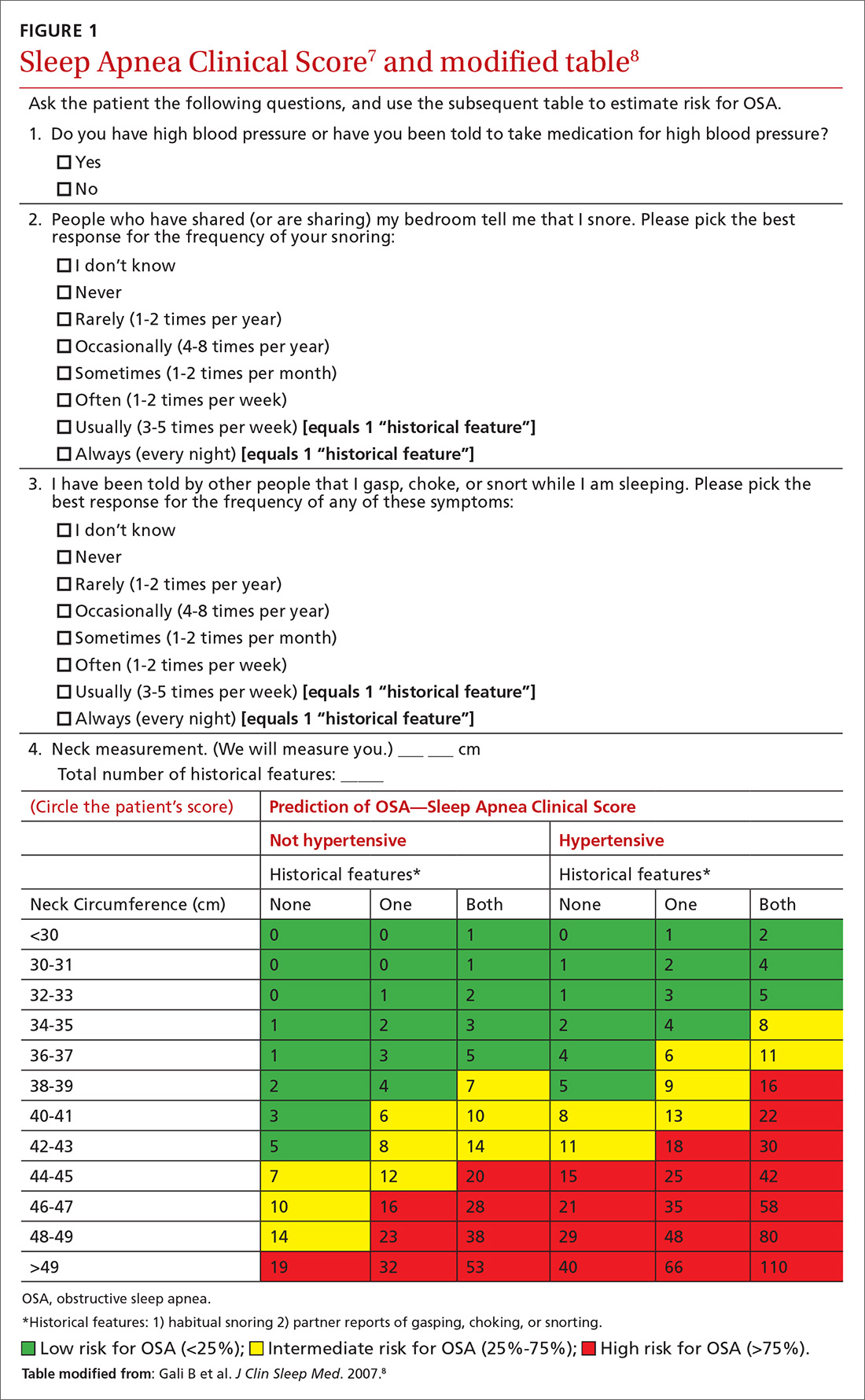
What about patients at intermediate risk? Many patients suspected of having OSA will be assigned to intermediate risk (SACS 6-14), and their PTP of OSA remains at 40% to 45%, the pre-test level most commonly encountered in suspected OSA. As polysomnography is a limited and expensive clinical resource, intermediate-risk patients would benefit from recalibration of their SACS-based risk assessment using an additional surrogate test such as home-based overnight oximetry. Our internal OSA practice guidelines recommend referral for sleep medicine consultation when oximetry results are abnormal—specifically, an oxygen desaturation index (ODI) of ≥5, a mean saturation less than 89%, and a minimum saturation of 75% or less.
Our objectives in this study were to compare the diagnostic implications of these 3 measurements from home-based overnight oximetry reports and use the most relevant result to derive a predictive model further refining PTP of OSA in a primary care patient population first stratified to intermediate risk by SACS.
METHODS
Subjects
We performed secondary analyses on data obtained from our SACS validation cohort.6 In brief, these were patients suspected of having OSA based on the presence of signs, symptoms, or associated risk factors. One hundred ninety-one patients completed all assessments. Sixty-six of 191 patients (35%) were categorized as intermediate risk (SACS 6-14; OSA probability 41% [27/66]).
Data collection and analyses
Participants completed home-based overnight oximetry using Nonin Model 2500 oximeters (Nonin Medical Inc., Plymouth, Minn). We transferred oximetry results from the sleep lab database to a statistical program for analyses of ODI, mean saturation, and minimal saturation. ODI was defined as the number of 4% drops in saturation from baseline divided by the number of hours of recording time. Although the AASM states that a diagnosis of OSA is confirmed if the number of obstructive events is more than 15 per hour or more than 5 per hour in a patient who reports related symptoms,4 we defined OSA as an apnea-hypopnea index (AHI) of >10 based on polysomnography (as this was the threshold used in the derivation cohort for SACS).7 We demonstrated the predictive ability of SACS at various AHI definitions of OSA in our validation cohort.6 The use of SACS in our validation cohort showed a statistically similar ability to predict OSA at both an AHI of 10 and 20, compared with the derivation cohort.
Continue to: We entered additional information...
We entered additional information reported directly by patients and obtained from their sleep studies into a REDCap database and transferred that to our statistical program. We used descriptive statistics to determine ranges and central tendencies of oximetry results. Receiver operator characteristic (ROC) analyses described the predictive abilities for each oximetry result individually and in serial application with prior SACS determinations. For comparison, we used the area under the ROC curve (AUC) from logistic regression to model the probability of OSA.
We calculated positive likelihood ratios (LR) and 95% confidence intervals (CI) to determine the degree of oximetry abnormality that would recalibrate risk either to a high PTP of OSA (>75%) or a low PTP (<25%). We sorted intermediate-risk SACS scores into quintiles based on ODI results to compare the resulting PTPs of OSA. We applied the PTP of OSA from our previous work (using the SACS score to compute the LR) as the new PTP, estimated the LR based on ODI, and computed an updated PTP of OSA. We also used ROC analysis to determine the optimal cutoff value of the ODI.
Finally, in accordance with our internal clinical practice recommendations, we examined the predictive ability of a “positive” ODI result of ≥5 to recalibrate risk prediction for OSA for patients in the low-risk group. We performed analyses using SAS 9.4 (SAS Institute, Cary, NC).
RESULTS
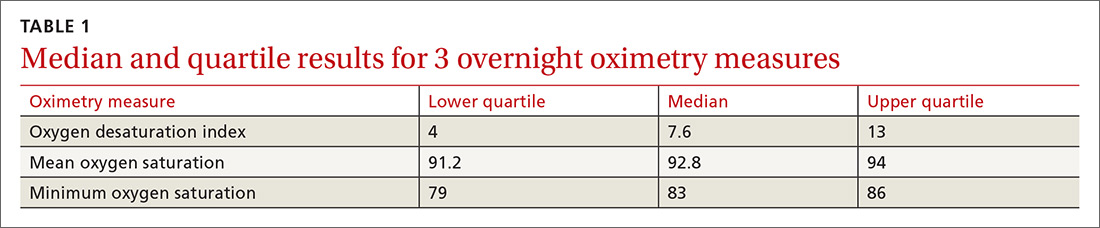
One hundred ninety-one subjects completed assessments. The median and quartile results for ODI, mean saturation, and minimum saturation are found in TABLE 1. TABLE 2 shows the distribution of patients with positive oximetry results. An ODI of 5 or greater was the most frequent abnormal result (135/191; 70.7%).
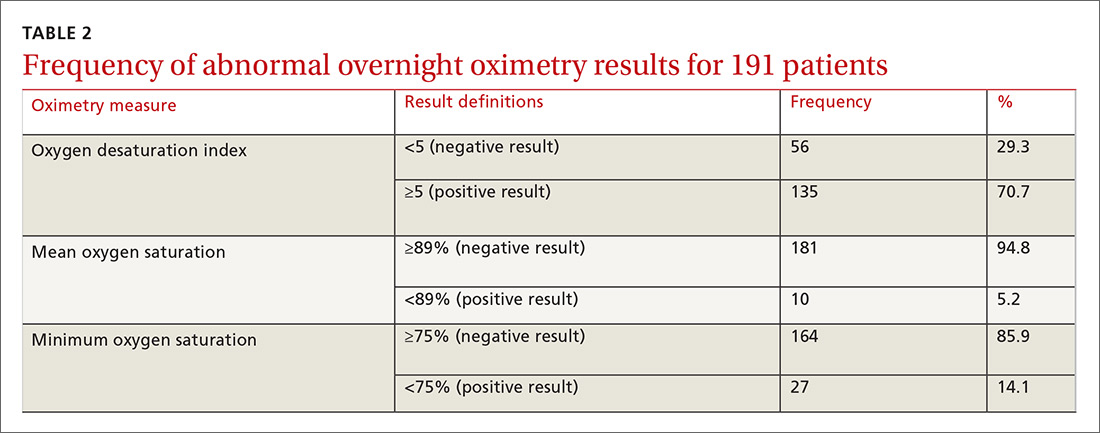
We used the AUC to measure the comparative abilities of SACS and the 3 overnight oximetry results in predicting OSA (TABLE 3). ODI results demonstrated the best ability to predict OSA, compared with polysomnography as the relative gold standard (AUC, 0.88; 95% confidence interval [CI], 0.83-0.93). Serial application of SACS and ODI yielded even better diagnostic results (AUC, 0.90; 95% CI, 0.85-0.95).
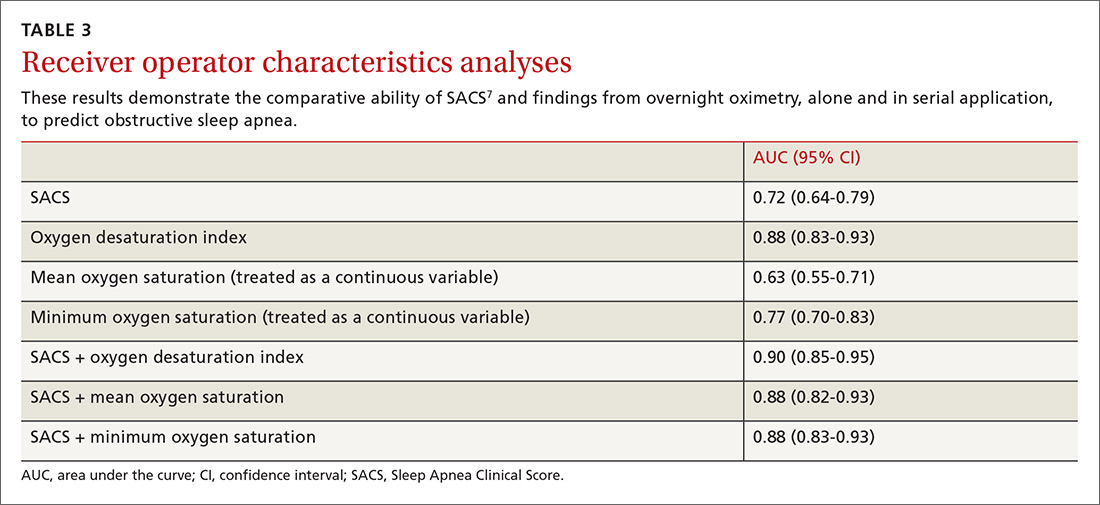
Continue to: As ODI was found to be the strongest predictor of OSA...
As ODI was found to be the strongest predictor of OSA, we grouped these results in quintiles and calculated positive LRs. TABLE 4 shows their effect on PTP of disease among patients with intermediate risk. An ODI result >10 effected an upward recalibration of disease probability (LR, 2.33; 95% CI, 1.27-4.26). The optimal cutoff of ODI to discriminate between those with and without OSA was determined by ROC analysis. An ODI greater than 8.4 created a PTP of disease of approximately 73% to 77%.
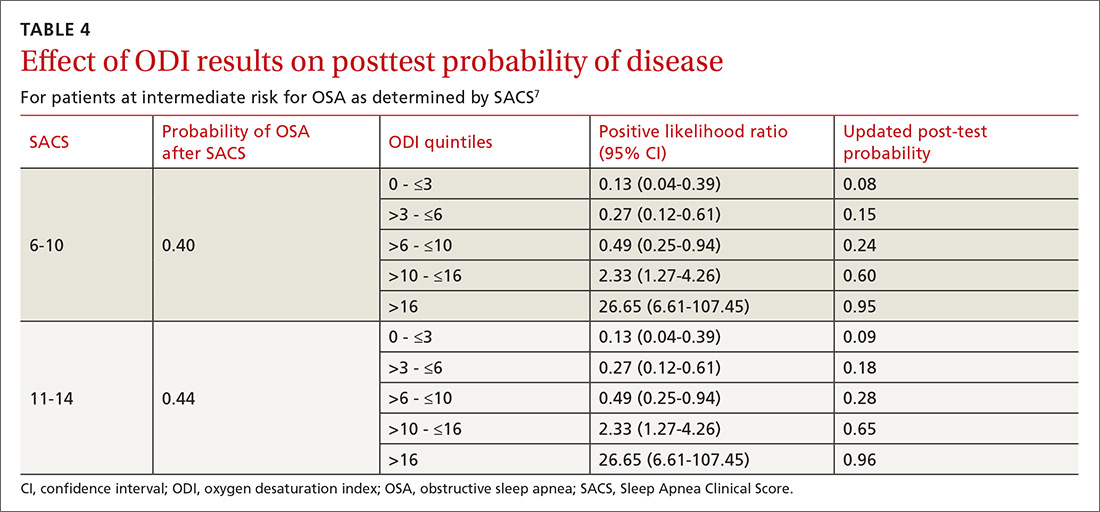
Our internal clinical guidelines recommend referring patients with an ODI of 5 or greater for sleep medicine consultation. We examined the ability of this ODI result to recalibrate disease suspicion for a patient at low risk (SACS ≤5). The LR for ODI of 5 or greater is 2.1, but this only results in a recalibration of risk from 24% pretest probability in our validation cohort to 41% PTP (95% CI, 33-49). This low cutoff for a positive test creates false-positive results more than 40% of the time due to low specificity (0.58). This is insufficient to change the suspicion of disease, resulting only in a shift to intermediate OSA risk.
DISCUSSION
Among 3 different oximetry measurements, an ODI ≥10 best predicts OSA, both independently and when used sequentially after the SACS. ODI was by far the most frequent abnormality on oximetry in our cohort, thereby increasing its utility in clinical decision making. For those subjects at intermediate risk, a cutoff of 10 for the ODI result may be a simple and clinically effective way to recalibrate risk and aid in making referral decisions. (This may also be simpler and more easily remembered by clinicians than the 8.4 ODI results from the ROC analyses.)
Assessment is inadequate without a clinical prediction rule. Unfortunately, providers cannot simply rely on clinical gestalt in diagnosing OSA. In their derivation cohort, Flemens et al examined the LRs created by SACS and by clinician prediction based on history and physical exam.7 The SACS LRs ranged from 5.17 to 0.25, a 20-fold range. This reflected superior diagnostic information compared with subjective physician impression, where LRs ranged from 3.7 to 0.52, a seven-fold range. Myers et al prepared a meta-analysis of 4 different trials that examined physicians’ ability to predict OSA.9 Despite the researchers’ use of experienced sleep medicine doctors, the overall diagnostic accuracy of clinical impression was modest (summary positive LR, 1.7; 95% CI, 1.5-2; I2 = 0%; summary negative LR, 0.67; 95% CI, 0.60-0.74; I2 = 10%; sensitivity, 58%; specificity, 67%). This is similar to reliance on a single clinical sign or symptom to predict OSA.
Wise use of oximetry augments SACS calculation. To limit unnecessary oximetry testing in low- and high-risk groups and to avoid polysomnography in cases of a low PTP of disease, we advocate limiting oximetry testing to individuals in the SACS intermediate-risk group (FIGURE 2) wherein ODI results can potentially recalibrate risk assessment up or down. (Those in the high- risk group should be referred to a sleep medicine specialist.) Our institutional recommendation of using an ODI result of ≥5 as a threshold to increase suspicion of disease requires a caveat for the low-risk group. “Positive” results at that low diagnostic threshold are frequently false.
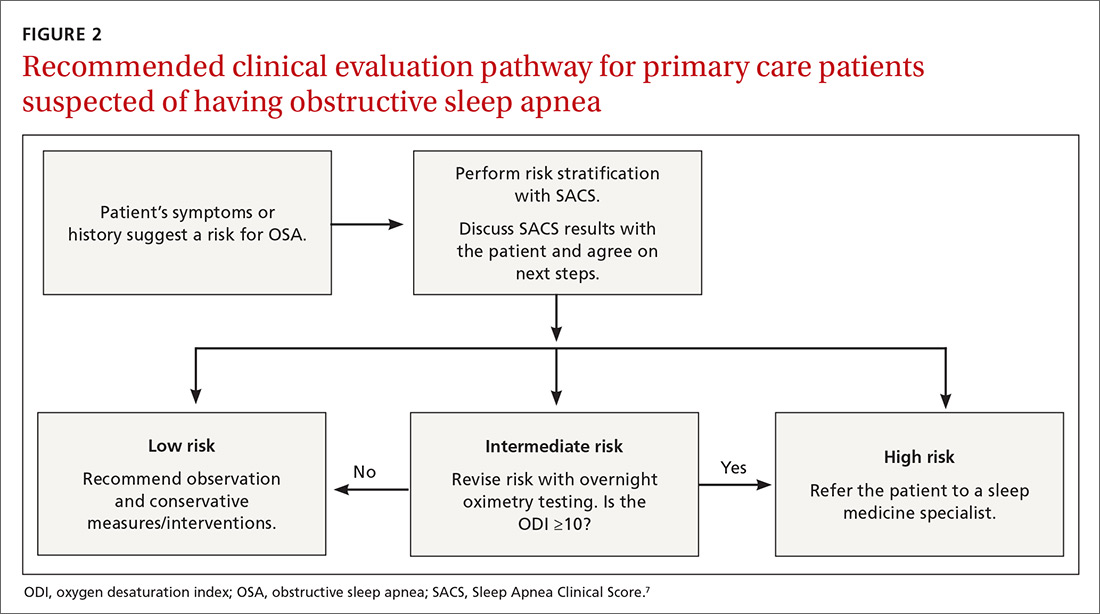
Continue to: Multiple benefits of SACS
Multiple benefits of SACS. We believe using the SACS calculation during clinical encounters with patients potentially at risk for OSA would increase diagnostic accuracy. Performing risk stratification with SACS should not be an undue burden on providers, and the increased time spent with patients has its own benefits, including helping them better understand their risk. Using this standardized process—augmented, as needed, with overnight ODI assessment—might also encourage more patients to follow through on subsequent recommendations, as their risk is further quantified objectively. Lastly, unnecessary testing with polysomnography could be avoided.
Limitations of our study. This study’s findings were derived from a patient population in a single institution. Replication of the findings from other settings would be helpful.
Looking forward. It is yet unclear if clinicians will embrace these strategies in real-world primary care practice. We have designed an implementation-and-dissemination trial to assess whether family physicians will use the SACS clinical predication rule in everyday practice and whether our evidence-based recommendations about overnight oximetry will be followed. Underlying our suggested clinical evaluation pathway (FIGURE 2) is the belief that there is value gained from sharing the decision-making process with patients. Although we provide new evidence that informs these conversations, the patient’s values and preferences are important when determining the best direction to proceed in the evaluation for suspected OSA. These recommendations are intended to aid, not replace, good clinical judgment.
Home-based sleep testing has become more widely available, is convenient for patients, and is less expensive than lab-based polysomnography. Our study did not directly address the appropriate circumstances for home studies in clinical evaluation. We rely on the expertise of our sleep medicine colleagues to determine which patients are appropriate candidates for home-based studies.
The AASM states that “portable monitors (PM) for the diagnosis of OSA should be [used] only in conjunction with a comprehensive sleep evaluation. Clinical sleep evaluations using PM must be supervised by a practitioner with board certification in sleep medicine or an individual who fulfills the eligibility criteria for the sleep medicine certification examination.”4 Additionally, the group recommends that PM “may be used in the unattended setting as an alternative to polysomnography for the diagnosis of OSA in patients with a high pretest probability of moderate to severe OSA and no comorbid sleep disorder or major comorbid medical disorders.”4
Continue to: GRANT SUPPORT
GRANT SUPPORT
The use of the REDCap database is supported by grant UL1 TR000135. This work was supported by a Mayo Foundation CR-20 grant awarded to Dr. Mookadam as Principal investigator and Dr. Grover as Coinvestigator.
Statistical analyses were supported, in part, by the Department of Family Medicine, Mayo Clinic, Scottsdale, Ariz.
CORRESPONDENCE
Michael Grover, DO, Mayo Clinic Thunderbird Primary Care Center-Family Medicine, 13737 N 92nd Street, Scottsdale, AZ 85260; [email protected]
1. National Sleep Foundation. Sleep apnea. https://sleepfoundation.org/sleep-disorders-problems/sleep-apnea. Accessed September 14, 2018.
2. Grover M, Mookadam M, Armas D, et al. Identifying patients at risk for obstructive sleep apnea in a primary care practice. J Am Board Fam Med. 2011;24:152-160.
3. Mold JW, Quattlebaum C, Schinnerer E, et al. Identification by primary care clinicians of patients with obstructive sleep apnea: a practice-based research network (PBRN) study. J Am Board Fam Med. 2011;24:138-145.
4. Epstein LJ, Kristo D, Strollo PJ, Jr., et al; Adult Obstructive Sleep Apnea Task Force of the American Academy of Sleep Medicine. Clinical guideline for the evaluation, management and long-term care of obstructive sleep apnea in adults. J Clin Sleep Med. 2009;5:263-276.
5. Qaseem A, Dallas P, Owens DK, et al. Diagnosis of obstructive sleep apnea in adults: a clinical practice guideline from the American College of Physicians. Ann Intern Med. 2014;161:210-220.
6. Grover M, Mookadam M, Chang Y-H, et al. Validating the Sleep Apnea Clinical Score for use in primary care populations. Mayo Clin Proc. 2016;91:469-476.
7. Flemons WW, Whitelaw WA, Brant R, et al. Likelihood ratios for a sleep apnea clinical prediction rule. Am J Respir Crit Care Med. 1994;150:1279-1285.
8. Gali B, Whalen FX, Gay PC, et al. Management plan to reduce risks in perioperative care of patients with presumed obstructive sleep apnea syndrome. J Clin Sleep Med. 2007;3:582-588.
9. Myers KA, Mrkobrada M, Simel DL. Does this patient have obstructive sleep apnea?: The rational clinical examination systematic review. JAMA. 2013;310(7):731-741.
ABSTRACT
Purpose To derive a predictive model for obstructive sleep apnea (OSA) in primary care practice, using home-based overnight oximetry results to refine posttest probability (PTP) of disease after initial risk stratification with the Sleep Apnea Clinical Score (SACS).
Methods We performed secondary analyses on data from a SACS validation cohort, to compare the diagnostic accuracy of 3 overnight oximetry measurements (oxygen desaturation index [ODI], mean saturation, and minimum saturation) in predicting OSA. Receiver operator characteristics (ROC) were computed for each measurement independently and sequentially after risk stratifying with SACS. We examined the implications of oximetry results for OSA PTP for participants categorized as intermediate risk (SACS 6-14; 66/191 participants [35%]; OSA probability 41%). We calculated positive likelihood ratios (LR) for multiple ODI results and determined which ones allowed recalibration to high- or low-risk PTP.
Results Among the 3 oximetry findings, ODI best predicted OSA (area under the curve [AUC], 0.88; 95% confidence interval [CI], 0.83-0.93). An ODI ≥8.4 (likelihood ratio [LR], 4.19; 95% CI, 2.87-6.10) created a PTP of 77%, while an ODI of 0 to <8.4 (LR, 0.19, 95% CI, 0.12-0.33) created a 14% PTP. Sequential application of SACS and ODI results yielded an AUC result of 0.90 (95% CI, 0.85-0.95).
Conclusions SACS risk stratification provides an advantage over clinical gestalt. In those at intermediate risk, ODI results provide a simple and clinically useful way to further refine diagnostic prediction. Sequential use of SACS and selectively employed overnight oximetry may limit unnecessary polysomnography. Oximetry testing should be avoided in patients deemed low or high risk by SACS, as positive results do not substantially recalibrate risk.
Obstructive sleep apnea (OSA) is a prevalent and underdiagnosed condition. The National Sleep Foundation estimates that 18 million Americans have OSA.1 Primary care practice may be the best setting in which to identify OSA, as many of our patients have conditions frequently associated with apnea (eg, hypertension, obesity, diabetes, arrhythmia, and neurologic illness). Up to a third of patients in primary care practice may be at increased risk.2,3
Clinical guidelines of the American Academy of Sleep Medicine (AASM) recommend obtaining a sleep history to evaluate for possible OSA in 3 instances: as part of a routine health maintenance examination, during evaluation of specific complaints associated with OSA (eg, snoring, apnea, daytime sleepiness), and during comprehensive evaluations for individuals with high-risk conditions (ie, obesity, congestive heart failure, refractory hypertension, diabetes, stroke history).4
The American College of Physicians (ACP) Clinical Practice Guideline suggests assessing individuals who have unexplained daytime sleepiness.5 The ACP considers this assessment “High-Value Care,” as “evidence shows that before diagnosis, patients with OSA have higher rates of health care use, more frequent and longer hospital stays, and higher health care costs than after diagnosis.”5
Continue to: We recently validated the diagnostic accuracy...
We recently validated the diagnostic accuracy of the Sleep Apnea Clinical Score (SACS) for use in a primary care patient population suspected of having OSA.6 SACS uses historical and clinical data to derive a score that identifies a patient’s risk level.7 However, as an alternative to the 2 levels described in Flemons’ SACS,7 we propose creating 3 risk strata (FIGURE 17,8). We believe that patients at high risk (SACS ≥15) should be encouraged to undergo sleep evaluations as their posttest probability (PTP) of OSA is 75% to 80%. Individuals at low risk (SACS ≤5; PTP <20%) could receive lifestyle advice and simple clinical interventions that decrease symptoms (eg, weight loss, increased physical activity, sleeping on one’s side). For low-risk patients, clinical observation and reevaluation could take place over time with their primary care provider, without additional testing or referral to specialists.

What about patients at intermediate risk? Many patients suspected of having OSA will be assigned to intermediate risk (SACS 6-14), and their PTP of OSA remains at 40% to 45%, the pre-test level most commonly encountered in suspected OSA. As polysomnography is a limited and expensive clinical resource, intermediate-risk patients would benefit from recalibration of their SACS-based risk assessment using an additional surrogate test such as home-based overnight oximetry. Our internal OSA practice guidelines recommend referral for sleep medicine consultation when oximetry results are abnormal—specifically, an oxygen desaturation index (ODI) of ≥5, a mean saturation less than 89%, and a minimum saturation of 75% or less.
Our objectives in this study were to compare the diagnostic implications of these 3 measurements from home-based overnight oximetry reports and use the most relevant result to derive a predictive model further refining PTP of OSA in a primary care patient population first stratified to intermediate risk by SACS.
METHODS
Subjects
We performed secondary analyses on data obtained from our SACS validation cohort.6 In brief, these were patients suspected of having OSA based on the presence of signs, symptoms, or associated risk factors. One hundred ninety-one patients completed all assessments. Sixty-six of 191 patients (35%) were categorized as intermediate risk (SACS 6-14; OSA probability 41% [27/66]).
Data collection and analyses
Participants completed home-based overnight oximetry using Nonin Model 2500 oximeters (Nonin Medical Inc., Plymouth, Minn). We transferred oximetry results from the sleep lab database to a statistical program for analyses of ODI, mean saturation, and minimal saturation. ODI was defined as the number of 4% drops in saturation from baseline divided by the number of hours of recording time. Although the AASM states that a diagnosis of OSA is confirmed if the number of obstructive events is more than 15 per hour or more than 5 per hour in a patient who reports related symptoms,4 we defined OSA as an apnea-hypopnea index (AHI) of >10 based on polysomnography (as this was the threshold used in the derivation cohort for SACS).7 We demonstrated the predictive ability of SACS at various AHI definitions of OSA in our validation cohort.6 The use of SACS in our validation cohort showed a statistically similar ability to predict OSA at both an AHI of 10 and 20, compared with the derivation cohort.
Continue to: We entered additional information...
We entered additional information reported directly by patients and obtained from their sleep studies into a REDCap database and transferred that to our statistical program. We used descriptive statistics to determine ranges and central tendencies of oximetry results. Receiver operator characteristic (ROC) analyses described the predictive abilities for each oximetry result individually and in serial application with prior SACS determinations. For comparison, we used the area under the ROC curve (AUC) from logistic regression to model the probability of OSA.
We calculated positive likelihood ratios (LR) and 95% confidence intervals (CI) to determine the degree of oximetry abnormality that would recalibrate risk either to a high PTP of OSA (>75%) or a low PTP (<25%). We sorted intermediate-risk SACS scores into quintiles based on ODI results to compare the resulting PTPs of OSA. We applied the PTP of OSA from our previous work (using the SACS score to compute the LR) as the new PTP, estimated the LR based on ODI, and computed an updated PTP of OSA. We also used ROC analysis to determine the optimal cutoff value of the ODI.
Finally, in accordance with our internal clinical practice recommendations, we examined the predictive ability of a “positive” ODI result of ≥5 to recalibrate risk prediction for OSA for patients in the low-risk group. We performed analyses using SAS 9.4 (SAS Institute, Cary, NC).
RESULTS

One hundred ninety-one subjects completed assessments. The median and quartile results for ODI, mean saturation, and minimum saturation are found in TABLE 1. TABLE 2 shows the distribution of patients with positive oximetry results. An ODI of 5 or greater was the most frequent abnormal result (135/191; 70.7%).

We used the AUC to measure the comparative abilities of SACS and the 3 overnight oximetry results in predicting OSA (TABLE 3). ODI results demonstrated the best ability to predict OSA, compared with polysomnography as the relative gold standard (AUC, 0.88; 95% confidence interval [CI], 0.83-0.93). Serial application of SACS and ODI yielded even better diagnostic results (AUC, 0.90; 95% CI, 0.85-0.95).

Continue to: As ODI was found to be the strongest predictor of OSA...
As ODI was found to be the strongest predictor of OSA, we grouped these results in quintiles and calculated positive LRs. TABLE 4 shows their effect on PTP of disease among patients with intermediate risk. An ODI result >10 effected an upward recalibration of disease probability (LR, 2.33; 95% CI, 1.27-4.26). The optimal cutoff of ODI to discriminate between those with and without OSA was determined by ROC analysis. An ODI greater than 8.4 created a PTP of disease of approximately 73% to 77%.

Our internal clinical guidelines recommend referring patients with an ODI of 5 or greater for sleep medicine consultation. We examined the ability of this ODI result to recalibrate disease suspicion for a patient at low risk (SACS ≤5). The LR for ODI of 5 or greater is 2.1, but this only results in a recalibration of risk from 24% pretest probability in our validation cohort to 41% PTP (95% CI, 33-49). This low cutoff for a positive test creates false-positive results more than 40% of the time due to low specificity (0.58). This is insufficient to change the suspicion of disease, resulting only in a shift to intermediate OSA risk.
DISCUSSION
Among 3 different oximetry measurements, an ODI ≥10 best predicts OSA, both independently and when used sequentially after the SACS. ODI was by far the most frequent abnormality on oximetry in our cohort, thereby increasing its utility in clinical decision making. For those subjects at intermediate risk, a cutoff of 10 for the ODI result may be a simple and clinically effective way to recalibrate risk and aid in making referral decisions. (This may also be simpler and more easily remembered by clinicians than the 8.4 ODI results from the ROC analyses.)
Assessment is inadequate without a clinical prediction rule. Unfortunately, providers cannot simply rely on clinical gestalt in diagnosing OSA. In their derivation cohort, Flemens et al examined the LRs created by SACS and by clinician prediction based on history and physical exam.7 The SACS LRs ranged from 5.17 to 0.25, a 20-fold range. This reflected superior diagnostic information compared with subjective physician impression, where LRs ranged from 3.7 to 0.52, a seven-fold range. Myers et al prepared a meta-analysis of 4 different trials that examined physicians’ ability to predict OSA.9 Despite the researchers’ use of experienced sleep medicine doctors, the overall diagnostic accuracy of clinical impression was modest (summary positive LR, 1.7; 95% CI, 1.5-2; I2 = 0%; summary negative LR, 0.67; 95% CI, 0.60-0.74; I2 = 10%; sensitivity, 58%; specificity, 67%). This is similar to reliance on a single clinical sign or symptom to predict OSA.
Wise use of oximetry augments SACS calculation. To limit unnecessary oximetry testing in low- and high-risk groups and to avoid polysomnography in cases of a low PTP of disease, we advocate limiting oximetry testing to individuals in the SACS intermediate-risk group (FIGURE 2) wherein ODI results can potentially recalibrate risk assessment up or down. (Those in the high- risk group should be referred to a sleep medicine specialist.) Our institutional recommendation of using an ODI result of ≥5 as a threshold to increase suspicion of disease requires a caveat for the low-risk group. “Positive” results at that low diagnostic threshold are frequently false.

Continue to: Multiple benefits of SACS
Multiple benefits of SACS. We believe using the SACS calculation during clinical encounters with patients potentially at risk for OSA would increase diagnostic accuracy. Performing risk stratification with SACS should not be an undue burden on providers, and the increased time spent with patients has its own benefits, including helping them better understand their risk. Using this standardized process—augmented, as needed, with overnight ODI assessment—might also encourage more patients to follow through on subsequent recommendations, as their risk is further quantified objectively. Lastly, unnecessary testing with polysomnography could be avoided.
Limitations of our study. This study’s findings were derived from a patient population in a single institution. Replication of the findings from other settings would be helpful.
Looking forward. It is yet unclear if clinicians will embrace these strategies in real-world primary care practice. We have designed an implementation-and-dissemination trial to assess whether family physicians will use the SACS clinical predication rule in everyday practice and whether our evidence-based recommendations about overnight oximetry will be followed. Underlying our suggested clinical evaluation pathway (FIGURE 2) is the belief that there is value gained from sharing the decision-making process with patients. Although we provide new evidence that informs these conversations, the patient’s values and preferences are important when determining the best direction to proceed in the evaluation for suspected OSA. These recommendations are intended to aid, not replace, good clinical judgment.
Home-based sleep testing has become more widely available, is convenient for patients, and is less expensive than lab-based polysomnography. Our study did not directly address the appropriate circumstances for home studies in clinical evaluation. We rely on the expertise of our sleep medicine colleagues to determine which patients are appropriate candidates for home-based studies.
The AASM states that “portable monitors (PM) for the diagnosis of OSA should be [used] only in conjunction with a comprehensive sleep evaluation. Clinical sleep evaluations using PM must be supervised by a practitioner with board certification in sleep medicine or an individual who fulfills the eligibility criteria for the sleep medicine certification examination.”4 Additionally, the group recommends that PM “may be used in the unattended setting as an alternative to polysomnography for the diagnosis of OSA in patients with a high pretest probability of moderate to severe OSA and no comorbid sleep disorder or major comorbid medical disorders.”4
Continue to: GRANT SUPPORT
GRANT SUPPORT
The use of the REDCap database is supported by grant UL1 TR000135. This work was supported by a Mayo Foundation CR-20 grant awarded to Dr. Mookadam as Principal investigator and Dr. Grover as Coinvestigator.
Statistical analyses were supported, in part, by the Department of Family Medicine, Mayo Clinic, Scottsdale, Ariz.
CORRESPONDENCE
Michael Grover, DO, Mayo Clinic Thunderbird Primary Care Center-Family Medicine, 13737 N 92nd Street, Scottsdale, AZ 85260; [email protected]
ABSTRACT
Purpose To derive a predictive model for obstructive sleep apnea (OSA) in primary care practice, using home-based overnight oximetry results to refine posttest probability (PTP) of disease after initial risk stratification with the Sleep Apnea Clinical Score (SACS).
Methods We performed secondary analyses on data from a SACS validation cohort, to compare the diagnostic accuracy of 3 overnight oximetry measurements (oxygen desaturation index [ODI], mean saturation, and minimum saturation) in predicting OSA. Receiver operator characteristics (ROC) were computed for each measurement independently and sequentially after risk stratifying with SACS. We examined the implications of oximetry results for OSA PTP for participants categorized as intermediate risk (SACS 6-14; 66/191 participants [35%]; OSA probability 41%). We calculated positive likelihood ratios (LR) for multiple ODI results and determined which ones allowed recalibration to high- or low-risk PTP.
Results Among the 3 oximetry findings, ODI best predicted OSA (area under the curve [AUC], 0.88; 95% confidence interval [CI], 0.83-0.93). An ODI ≥8.4 (likelihood ratio [LR], 4.19; 95% CI, 2.87-6.10) created a PTP of 77%, while an ODI of 0 to <8.4 (LR, 0.19, 95% CI, 0.12-0.33) created a 14% PTP. Sequential application of SACS and ODI results yielded an AUC result of 0.90 (95% CI, 0.85-0.95).
Conclusions SACS risk stratification provides an advantage over clinical gestalt. In those at intermediate risk, ODI results provide a simple and clinically useful way to further refine diagnostic prediction. Sequential use of SACS and selectively employed overnight oximetry may limit unnecessary polysomnography. Oximetry testing should be avoided in patients deemed low or high risk by SACS, as positive results do not substantially recalibrate risk.
Obstructive sleep apnea (OSA) is a prevalent and underdiagnosed condition. The National Sleep Foundation estimates that 18 million Americans have OSA.1 Primary care practice may be the best setting in which to identify OSA, as many of our patients have conditions frequently associated with apnea (eg, hypertension, obesity, diabetes, arrhythmia, and neurologic illness). Up to a third of patients in primary care practice may be at increased risk.2,3
Clinical guidelines of the American Academy of Sleep Medicine (AASM) recommend obtaining a sleep history to evaluate for possible OSA in 3 instances: as part of a routine health maintenance examination, during evaluation of specific complaints associated with OSA (eg, snoring, apnea, daytime sleepiness), and during comprehensive evaluations for individuals with high-risk conditions (ie, obesity, congestive heart failure, refractory hypertension, diabetes, stroke history).4
The American College of Physicians (ACP) Clinical Practice Guideline suggests assessing individuals who have unexplained daytime sleepiness.5 The ACP considers this assessment “High-Value Care,” as “evidence shows that before diagnosis, patients with OSA have higher rates of health care use, more frequent and longer hospital stays, and higher health care costs than after diagnosis.”5
Continue to: We recently validated the diagnostic accuracy...
We recently validated the diagnostic accuracy of the Sleep Apnea Clinical Score (SACS) for use in a primary care patient population suspected of having OSA.6 SACS uses historical and clinical data to derive a score that identifies a patient’s risk level.7 However, as an alternative to the 2 levels described in Flemons’ SACS,7 we propose creating 3 risk strata (FIGURE 17,8). We believe that patients at high risk (SACS ≥15) should be encouraged to undergo sleep evaluations as their posttest probability (PTP) of OSA is 75% to 80%. Individuals at low risk (SACS ≤5; PTP <20%) could receive lifestyle advice and simple clinical interventions that decrease symptoms (eg, weight loss, increased physical activity, sleeping on one’s side). For low-risk patients, clinical observation and reevaluation could take place over time with their primary care provider, without additional testing or referral to specialists.

What about patients at intermediate risk? Many patients suspected of having OSA will be assigned to intermediate risk (SACS 6-14), and their PTP of OSA remains at 40% to 45%, the pre-test level most commonly encountered in suspected OSA. As polysomnography is a limited and expensive clinical resource, intermediate-risk patients would benefit from recalibration of their SACS-based risk assessment using an additional surrogate test such as home-based overnight oximetry. Our internal OSA practice guidelines recommend referral for sleep medicine consultation when oximetry results are abnormal—specifically, an oxygen desaturation index (ODI) of ≥5, a mean saturation less than 89%, and a minimum saturation of 75% or less.
Our objectives in this study were to compare the diagnostic implications of these 3 measurements from home-based overnight oximetry reports and use the most relevant result to derive a predictive model further refining PTP of OSA in a primary care patient population first stratified to intermediate risk by SACS.
METHODS
Subjects
We performed secondary analyses on data obtained from our SACS validation cohort.6 In brief, these were patients suspected of having OSA based on the presence of signs, symptoms, or associated risk factors. One hundred ninety-one patients completed all assessments. Sixty-six of 191 patients (35%) were categorized as intermediate risk (SACS 6-14; OSA probability 41% [27/66]).
Data collection and analyses
Participants completed home-based overnight oximetry using Nonin Model 2500 oximeters (Nonin Medical Inc., Plymouth, Minn). We transferred oximetry results from the sleep lab database to a statistical program for analyses of ODI, mean saturation, and minimal saturation. ODI was defined as the number of 4% drops in saturation from baseline divided by the number of hours of recording time. Although the AASM states that a diagnosis of OSA is confirmed if the number of obstructive events is more than 15 per hour or more than 5 per hour in a patient who reports related symptoms,4 we defined OSA as an apnea-hypopnea index (AHI) of >10 based on polysomnography (as this was the threshold used in the derivation cohort for SACS).7 We demonstrated the predictive ability of SACS at various AHI definitions of OSA in our validation cohort.6 The use of SACS in our validation cohort showed a statistically similar ability to predict OSA at both an AHI of 10 and 20, compared with the derivation cohort.
Continue to: We entered additional information...
We entered additional information reported directly by patients and obtained from their sleep studies into a REDCap database and transferred that to our statistical program. We used descriptive statistics to determine ranges and central tendencies of oximetry results. Receiver operator characteristic (ROC) analyses described the predictive abilities for each oximetry result individually and in serial application with prior SACS determinations. For comparison, we used the area under the ROC curve (AUC) from logistic regression to model the probability of OSA.
We calculated positive likelihood ratios (LR) and 95% confidence intervals (CI) to determine the degree of oximetry abnormality that would recalibrate risk either to a high PTP of OSA (>75%) or a low PTP (<25%). We sorted intermediate-risk SACS scores into quintiles based on ODI results to compare the resulting PTPs of OSA. We applied the PTP of OSA from our previous work (using the SACS score to compute the LR) as the new PTP, estimated the LR based on ODI, and computed an updated PTP of OSA. We also used ROC analysis to determine the optimal cutoff value of the ODI.
Finally, in accordance with our internal clinical practice recommendations, we examined the predictive ability of a “positive” ODI result of ≥5 to recalibrate risk prediction for OSA for patients in the low-risk group. We performed analyses using SAS 9.4 (SAS Institute, Cary, NC).
RESULTS

One hundred ninety-one subjects completed assessments. The median and quartile results for ODI, mean saturation, and minimum saturation are found in TABLE 1. TABLE 2 shows the distribution of patients with positive oximetry results. An ODI of 5 or greater was the most frequent abnormal result (135/191; 70.7%).

We used the AUC to measure the comparative abilities of SACS and the 3 overnight oximetry results in predicting OSA (TABLE 3). ODI results demonstrated the best ability to predict OSA, compared with polysomnography as the relative gold standard (AUC, 0.88; 95% confidence interval [CI], 0.83-0.93). Serial application of SACS and ODI yielded even better diagnostic results (AUC, 0.90; 95% CI, 0.85-0.95).

Continue to: As ODI was found to be the strongest predictor of OSA...
As ODI was found to be the strongest predictor of OSA, we grouped these results in quintiles and calculated positive LRs. TABLE 4 shows their effect on PTP of disease among patients with intermediate risk. An ODI result >10 effected an upward recalibration of disease probability (LR, 2.33; 95% CI, 1.27-4.26). The optimal cutoff of ODI to discriminate between those with and without OSA was determined by ROC analysis. An ODI greater than 8.4 created a PTP of disease of approximately 73% to 77%.

Our internal clinical guidelines recommend referring patients with an ODI of 5 or greater for sleep medicine consultation. We examined the ability of this ODI result to recalibrate disease suspicion for a patient at low risk (SACS ≤5). The LR for ODI of 5 or greater is 2.1, but this only results in a recalibration of risk from 24% pretest probability in our validation cohort to 41% PTP (95% CI, 33-49). This low cutoff for a positive test creates false-positive results more than 40% of the time due to low specificity (0.58). This is insufficient to change the suspicion of disease, resulting only in a shift to intermediate OSA risk.
DISCUSSION
Among 3 different oximetry measurements, an ODI ≥10 best predicts OSA, both independently and when used sequentially after the SACS. ODI was by far the most frequent abnormality on oximetry in our cohort, thereby increasing its utility in clinical decision making. For those subjects at intermediate risk, a cutoff of 10 for the ODI result may be a simple and clinically effective way to recalibrate risk and aid in making referral decisions. (This may also be simpler and more easily remembered by clinicians than the 8.4 ODI results from the ROC analyses.)
Assessment is inadequate without a clinical prediction rule. Unfortunately, providers cannot simply rely on clinical gestalt in diagnosing OSA. In their derivation cohort, Flemens et al examined the LRs created by SACS and by clinician prediction based on history and physical exam.7 The SACS LRs ranged from 5.17 to 0.25, a 20-fold range. This reflected superior diagnostic information compared with subjective physician impression, where LRs ranged from 3.7 to 0.52, a seven-fold range. Myers et al prepared a meta-analysis of 4 different trials that examined physicians’ ability to predict OSA.9 Despite the researchers’ use of experienced sleep medicine doctors, the overall diagnostic accuracy of clinical impression was modest (summary positive LR, 1.7; 95% CI, 1.5-2; I2 = 0%; summary negative LR, 0.67; 95% CI, 0.60-0.74; I2 = 10%; sensitivity, 58%; specificity, 67%). This is similar to reliance on a single clinical sign or symptom to predict OSA.
Wise use of oximetry augments SACS calculation. To limit unnecessary oximetry testing in low- and high-risk groups and to avoid polysomnography in cases of a low PTP of disease, we advocate limiting oximetry testing to individuals in the SACS intermediate-risk group (FIGURE 2) wherein ODI results can potentially recalibrate risk assessment up or down. (Those in the high- risk group should be referred to a sleep medicine specialist.) Our institutional recommendation of using an ODI result of ≥5 as a threshold to increase suspicion of disease requires a caveat for the low-risk group. “Positive” results at that low diagnostic threshold are frequently false.

Continue to: Multiple benefits of SACS
Multiple benefits of SACS. We believe using the SACS calculation during clinical encounters with patients potentially at risk for OSA would increase diagnostic accuracy. Performing risk stratification with SACS should not be an undue burden on providers, and the increased time spent with patients has its own benefits, including helping them better understand their risk. Using this standardized process—augmented, as needed, with overnight ODI assessment—might also encourage more patients to follow through on subsequent recommendations, as their risk is further quantified objectively. Lastly, unnecessary testing with polysomnography could be avoided.
Limitations of our study. This study’s findings were derived from a patient population in a single institution. Replication of the findings from other settings would be helpful.
Looking forward. It is yet unclear if clinicians will embrace these strategies in real-world primary care practice. We have designed an implementation-and-dissemination trial to assess whether family physicians will use the SACS clinical predication rule in everyday practice and whether our evidence-based recommendations about overnight oximetry will be followed. Underlying our suggested clinical evaluation pathway (FIGURE 2) is the belief that there is value gained from sharing the decision-making process with patients. Although we provide new evidence that informs these conversations, the patient’s values and preferences are important when determining the best direction to proceed in the evaluation for suspected OSA. These recommendations are intended to aid, not replace, good clinical judgment.
Home-based sleep testing has become more widely available, is convenient for patients, and is less expensive than lab-based polysomnography. Our study did not directly address the appropriate circumstances for home studies in clinical evaluation. We rely on the expertise of our sleep medicine colleagues to determine which patients are appropriate candidates for home-based studies.
The AASM states that “portable monitors (PM) for the diagnosis of OSA should be [used] only in conjunction with a comprehensive sleep evaluation. Clinical sleep evaluations using PM must be supervised by a practitioner with board certification in sleep medicine or an individual who fulfills the eligibility criteria for the sleep medicine certification examination.”4 Additionally, the group recommends that PM “may be used in the unattended setting as an alternative to polysomnography for the diagnosis of OSA in patients with a high pretest probability of moderate to severe OSA and no comorbid sleep disorder or major comorbid medical disorders.”4
Continue to: GRANT SUPPORT
GRANT SUPPORT
The use of the REDCap database is supported by grant UL1 TR000135. This work was supported by a Mayo Foundation CR-20 grant awarded to Dr. Mookadam as Principal investigator and Dr. Grover as Coinvestigator.
Statistical analyses were supported, in part, by the Department of Family Medicine, Mayo Clinic, Scottsdale, Ariz.
CORRESPONDENCE
Michael Grover, DO, Mayo Clinic Thunderbird Primary Care Center-Family Medicine, 13737 N 92nd Street, Scottsdale, AZ 85260; [email protected]
1. National Sleep Foundation. Sleep apnea. https://sleepfoundation.org/sleep-disorders-problems/sleep-apnea. Accessed September 14, 2018.
2. Grover M, Mookadam M, Armas D, et al. Identifying patients at risk for obstructive sleep apnea in a primary care practice. J Am Board Fam Med. 2011;24:152-160.
3. Mold JW, Quattlebaum C, Schinnerer E, et al. Identification by primary care clinicians of patients with obstructive sleep apnea: a practice-based research network (PBRN) study. J Am Board Fam Med. 2011;24:138-145.
4. Epstein LJ, Kristo D, Strollo PJ, Jr., et al; Adult Obstructive Sleep Apnea Task Force of the American Academy of Sleep Medicine. Clinical guideline for the evaluation, management and long-term care of obstructive sleep apnea in adults. J Clin Sleep Med. 2009;5:263-276.
5. Qaseem A, Dallas P, Owens DK, et al. Diagnosis of obstructive sleep apnea in adults: a clinical practice guideline from the American College of Physicians. Ann Intern Med. 2014;161:210-220.
6. Grover M, Mookadam M, Chang Y-H, et al. Validating the Sleep Apnea Clinical Score for use in primary care populations. Mayo Clin Proc. 2016;91:469-476.
7. Flemons WW, Whitelaw WA, Brant R, et al. Likelihood ratios for a sleep apnea clinical prediction rule. Am J Respir Crit Care Med. 1994;150:1279-1285.
8. Gali B, Whalen FX, Gay PC, et al. Management plan to reduce risks in perioperative care of patients with presumed obstructive sleep apnea syndrome. J Clin Sleep Med. 2007;3:582-588.
9. Myers KA, Mrkobrada M, Simel DL. Does this patient have obstructive sleep apnea?: The rational clinical examination systematic review. JAMA. 2013;310(7):731-741.
1. National Sleep Foundation. Sleep apnea. https://sleepfoundation.org/sleep-disorders-problems/sleep-apnea. Accessed September 14, 2018.
2. Grover M, Mookadam M, Armas D, et al. Identifying patients at risk for obstructive sleep apnea in a primary care practice. J Am Board Fam Med. 2011;24:152-160.
3. Mold JW, Quattlebaum C, Schinnerer E, et al. Identification by primary care clinicians of patients with obstructive sleep apnea: a practice-based research network (PBRN) study. J Am Board Fam Med. 2011;24:138-145.
4. Epstein LJ, Kristo D, Strollo PJ, Jr., et al; Adult Obstructive Sleep Apnea Task Force of the American Academy of Sleep Medicine. Clinical guideline for the evaluation, management and long-term care of obstructive sleep apnea in adults. J Clin Sleep Med. 2009;5:263-276.
5. Qaseem A, Dallas P, Owens DK, et al. Diagnosis of obstructive sleep apnea in adults: a clinical practice guideline from the American College of Physicians. Ann Intern Med. 2014;161:210-220.
6. Grover M, Mookadam M, Chang Y-H, et al. Validating the Sleep Apnea Clinical Score for use in primary care populations. Mayo Clin Proc. 2016;91:469-476.
7. Flemons WW, Whitelaw WA, Brant R, et al. Likelihood ratios for a sleep apnea clinical prediction rule. Am J Respir Crit Care Med. 1994;150:1279-1285.
8. Gali B, Whalen FX, Gay PC, et al. Management plan to reduce risks in perioperative care of patients with presumed obstructive sleep apnea syndrome. J Clin Sleep Med. 2007;3:582-588.
9. Myers KA, Mrkobrada M, Simel DL. Does this patient have obstructive sleep apnea?: The rational clinical examination systematic review. JAMA. 2013;310(7):731-741.
FDA approves Xyrem to treat children with narcolepsy
The Food and Drug Administration has cleared Xyrem (sodium oxybate) oral solution to treat cataplexy and excessive daytime sleepiness in patients ages 7-17 with narcolepsy.
The central nervous system depressant previously had been approved to treat cataplexy in adults with narcolepsy.
The current approval was granted by the FDA under a Priority Review designation. Xyrem also received the FDA’s Orphan Drug designation, which is intended to encourage the development of drugs for rare diseases.
The agency noted in a press release, however, that the drug would continue to be available only through risk evaluation mitigation strategy (REMS) programs because of “the risk of serious outcomes resulting from inappropriate prescribing, misuse, abuse and diversion.” Xyrem either alone or in combination with other CNS depressants may be associated with reactions including seizure, respiratory depression, decreases in the level of consciousness, coma, and death, the FDA said.
The most common adverse reactions in pediatric patients were enuresis, nausea, headache, vomiting, weight decrease, decreased appetite, and dizziness.
For more information on prescribing Xyrem for pediatric patients, see the revised labeling information on the FDA website.
The Food and Drug Administration has cleared Xyrem (sodium oxybate) oral solution to treat cataplexy and excessive daytime sleepiness in patients ages 7-17 with narcolepsy.
The central nervous system depressant previously had been approved to treat cataplexy in adults with narcolepsy.
The current approval was granted by the FDA under a Priority Review designation. Xyrem also received the FDA’s Orphan Drug designation, which is intended to encourage the development of drugs for rare diseases.
The agency noted in a press release, however, that the drug would continue to be available only through risk evaluation mitigation strategy (REMS) programs because of “the risk of serious outcomes resulting from inappropriate prescribing, misuse, abuse and diversion.” Xyrem either alone or in combination with other CNS depressants may be associated with reactions including seizure, respiratory depression, decreases in the level of consciousness, coma, and death, the FDA said.
The most common adverse reactions in pediatric patients were enuresis, nausea, headache, vomiting, weight decrease, decreased appetite, and dizziness.
For more information on prescribing Xyrem for pediatric patients, see the revised labeling information on the FDA website.
The Food and Drug Administration has cleared Xyrem (sodium oxybate) oral solution to treat cataplexy and excessive daytime sleepiness in patients ages 7-17 with narcolepsy.
The central nervous system depressant previously had been approved to treat cataplexy in adults with narcolepsy.
The current approval was granted by the FDA under a Priority Review designation. Xyrem also received the FDA’s Orphan Drug designation, which is intended to encourage the development of drugs for rare diseases.
The agency noted in a press release, however, that the drug would continue to be available only through risk evaluation mitigation strategy (REMS) programs because of “the risk of serious outcomes resulting from inappropriate prescribing, misuse, abuse and diversion.” Xyrem either alone or in combination with other CNS depressants may be associated with reactions including seizure, respiratory depression, decreases in the level of consciousness, coma, and death, the FDA said.
The most common adverse reactions in pediatric patients were enuresis, nausea, headache, vomiting, weight decrease, decreased appetite, and dizziness.
For more information on prescribing Xyrem for pediatric patients, see the revised labeling information on the FDA website.
Pediatric OSA linked to abnormal metabolic values
SAN ANTONIO – Obstructive sleep apnea (OSA) in children is associated with an abnormal metabolic profile, but not with body mass index (BMI), according to new research.
“Screening for metabolic dysfunction in obese children with obstructive sleep apnea can help identify those at risk for cardiovascular complications,” Kanika Mathur, MD, of the Albert Einstein College of Medicine and the Children’s Hospital at Montefiore, both in New York, told attendees at the annual meeting of the American College of Chest Physicians. Dr. Mathur explained that no consensus currently exists regarding routine cardiac evaluation of children with OSA.
“The American Academy of Pediatrics does not mention any sort of cardiac evaluation in children with OSA while the most recent guidelines from the American Heart Association and the American Thoracic Society recommend echocardiographic evaluation in children with severe obstructive sleep apnea, specifically to evaluate for pulmonary hypertension and right ventricular dysfunction,” Dr. Mathur told attendees.
OSA’s association with obesity, diabetes, and hypertension is well established in adults. It is an independent risk factor for coronary artery disease, heart failure, stroke and atrial fibrillation, and research has suggested OSA treatment can reduce cardiovascular risk in adults, Dr. Mathur explained, but little data on children exist. She and her colleagues set out to understand the relationship of OSA in children with various measures of cardiovascular and metabolic health.
“Despite similar degrees of obesity and systemic blood pressure, pediatric patients with OSA had significantly higher diastolic blood pressure, heart rate, and abnormal metabolic profile, including elevated alanine transaminase, aspartate transaminase, triglycerides and hemoglobin A1c,” they found.
Their study included patients aged 3-21 years with a BMI of at least the 95th percentile who had undergone sleep study and an echocardiogram at the Children’s Hospital at Montefiore between November 2016 and November 2017.
They excluded those with comorbidities related to cardiovascular morbidity: heart disease, neuromuscular disease, sickle cell disease, rheumatologic diseases, significant cranial facial abnormalities, tracheostomy, and any lung disease. However, 7% of the patients had trisomy 21.
Among the 81 children who met their criteria, 37 were male and 44 were female, with an average age of 14 years old and a mean BMI of 39.4 kg/m2 (mean BMI z score of 2.22). Most of the patients (53.1%) had severe OSA (apnea-hypopnea index of at least 10), 21% had moderate OSA (AHI 5-9.9), 12.3% had mild OSA (AHI 2-4.9), and 13.6% did not have OSA. The median AHI of the children was 10.3.
Among all the children, “about half had elevated systolic blood pressure, which is already a risk factor for cardiovascular morbidity,” Mathur reported.
BMI, BMI z score, systolic blood pressure z score, oxygen saturation and cholesterol (overall and both HDL and LDL cholesterol levels) did not significantly differ between children who had OSA and those who did not, but diastolic blood pressure and heart rate did. Those with OSA had a diastolic blood pressure of 65 mm Hg, compared with 58 mm Hg without OSA (P = .008). Heart rate was 89 bpm in the children with OSA, compared with 78 bpm in those without (P = .004).
The children with OSA also showed higher mean levels of several other metabolic biomarkers:
- Alanine transaminase: 26 U/L with OSA vs. 18 U/L without (P = .01).
- Aspartate transaminase: 23 U/L with OSA vs. 18 U/L without (P = .03).
- Triglycerides: 138 mg/dL with OSA vs 84 mg/dL without (P = .004).
- Hemoglobin A1c: 6.2% with OSA vs. 5.4% without (P = .002).
Children with and without OSA did not have any significant differences in left atrial indexed volume, left ventricular volume, left ventricular ejection fraction, or left ventricular mass (measured by M-mode or 5/6 area length formula). Though research has shown these measures to differ in adults with and without OSA, evidence on echocardiographic changes in children has been conflicting, Dr. Mathur noted.
The researchers also conducted subanalyses according to OSA severity, but BMI, BMI Z-score, systolic or diastolic blood pressure Z-score, heart rate and oxygen saturation did not differ between those with mild OSA vs those with moderate or severe OSA. No differences in echocardiographic measurements existed between these subgroups, either.
However, children with moderate to severe OSA did have higher alanine transaminase (27 U/L with moderate to severe vs. 17 U/L with mild OSA; P = .005) and higher triglycerides (148 vs 74; P = .001).
“Certainly we need further evaluation to see the efficacy of obstructive sleep apnea therapies on metabolic dysfunction and whether weight loss needs to be an adjunct therapy for these patients,” Dr. Mathur told attendees. She also noted the need to define the role of echocardiography in managing children with OSA.
The study had several limitations, including its retrospective cross-sectional nature at a single center and its small sample size.
“Additionally, we have a wide variety of ages, which could represent different pathophysiology of the associated metabolic dysfunction in these patients,” Mathur said. “There is an inherent difficulty to performing echocardiograms in a very obese population as well.”
Both the moderators of the pediatrics section, Christopher Carroll, MD, FCCP, of Connecticut Children’s Medical Center in Hartford, and Shahid Sheikh, MD, FCCP, of Nationwide Children’s Hospital in Columbus, Ohio, were impressed with the research. Dr. Carroll called it a “very elegant” study, and Dr. Sheikh noted the need for these studies in pediatrics “so that we don’t have to rely on grown-up data,” which may or may not generalize to children.
SOURCE: CHEST 2018. https://journal.chestnet.org/article/S0012-3692(18)31935-4/fulltext
SAN ANTONIO – Obstructive sleep apnea (OSA) in children is associated with an abnormal metabolic profile, but not with body mass index (BMI), according to new research.
“Screening for metabolic dysfunction in obese children with obstructive sleep apnea can help identify those at risk for cardiovascular complications,” Kanika Mathur, MD, of the Albert Einstein College of Medicine and the Children’s Hospital at Montefiore, both in New York, told attendees at the annual meeting of the American College of Chest Physicians. Dr. Mathur explained that no consensus currently exists regarding routine cardiac evaluation of children with OSA.
“The American Academy of Pediatrics does not mention any sort of cardiac evaluation in children with OSA while the most recent guidelines from the American Heart Association and the American Thoracic Society recommend echocardiographic evaluation in children with severe obstructive sleep apnea, specifically to evaluate for pulmonary hypertension and right ventricular dysfunction,” Dr. Mathur told attendees.
OSA’s association with obesity, diabetes, and hypertension is well established in adults. It is an independent risk factor for coronary artery disease, heart failure, stroke and atrial fibrillation, and research has suggested OSA treatment can reduce cardiovascular risk in adults, Dr. Mathur explained, but little data on children exist. She and her colleagues set out to understand the relationship of OSA in children with various measures of cardiovascular and metabolic health.
“Despite similar degrees of obesity and systemic blood pressure, pediatric patients with OSA had significantly higher diastolic blood pressure, heart rate, and abnormal metabolic profile, including elevated alanine transaminase, aspartate transaminase, triglycerides and hemoglobin A1c,” they found.
Their study included patients aged 3-21 years with a BMI of at least the 95th percentile who had undergone sleep study and an echocardiogram at the Children’s Hospital at Montefiore between November 2016 and November 2017.
They excluded those with comorbidities related to cardiovascular morbidity: heart disease, neuromuscular disease, sickle cell disease, rheumatologic diseases, significant cranial facial abnormalities, tracheostomy, and any lung disease. However, 7% of the patients had trisomy 21.
Among the 81 children who met their criteria, 37 were male and 44 were female, with an average age of 14 years old and a mean BMI of 39.4 kg/m2 (mean BMI z score of 2.22). Most of the patients (53.1%) had severe OSA (apnea-hypopnea index of at least 10), 21% had moderate OSA (AHI 5-9.9), 12.3% had mild OSA (AHI 2-4.9), and 13.6% did not have OSA. The median AHI of the children was 10.3.
Among all the children, “about half had elevated systolic blood pressure, which is already a risk factor for cardiovascular morbidity,” Mathur reported.
BMI, BMI z score, systolic blood pressure z score, oxygen saturation and cholesterol (overall and both HDL and LDL cholesterol levels) did not significantly differ between children who had OSA and those who did not, but diastolic blood pressure and heart rate did. Those with OSA had a diastolic blood pressure of 65 mm Hg, compared with 58 mm Hg without OSA (P = .008). Heart rate was 89 bpm in the children with OSA, compared with 78 bpm in those without (P = .004).
The children with OSA also showed higher mean levels of several other metabolic biomarkers:
- Alanine transaminase: 26 U/L with OSA vs. 18 U/L without (P = .01).
- Aspartate transaminase: 23 U/L with OSA vs. 18 U/L without (P = .03).
- Triglycerides: 138 mg/dL with OSA vs 84 mg/dL without (P = .004).
- Hemoglobin A1c: 6.2% with OSA vs. 5.4% without (P = .002).
Children with and without OSA did not have any significant differences in left atrial indexed volume, left ventricular volume, left ventricular ejection fraction, or left ventricular mass (measured by M-mode or 5/6 area length formula). Though research has shown these measures to differ in adults with and without OSA, evidence on echocardiographic changes in children has been conflicting, Dr. Mathur noted.
The researchers also conducted subanalyses according to OSA severity, but BMI, BMI Z-score, systolic or diastolic blood pressure Z-score, heart rate and oxygen saturation did not differ between those with mild OSA vs those with moderate or severe OSA. No differences in echocardiographic measurements existed between these subgroups, either.
However, children with moderate to severe OSA did have higher alanine transaminase (27 U/L with moderate to severe vs. 17 U/L with mild OSA; P = .005) and higher triglycerides (148 vs 74; P = .001).
“Certainly we need further evaluation to see the efficacy of obstructive sleep apnea therapies on metabolic dysfunction and whether weight loss needs to be an adjunct therapy for these patients,” Dr. Mathur told attendees. She also noted the need to define the role of echocardiography in managing children with OSA.
The study had several limitations, including its retrospective cross-sectional nature at a single center and its small sample size.
“Additionally, we have a wide variety of ages, which could represent different pathophysiology of the associated metabolic dysfunction in these patients,” Mathur said. “There is an inherent difficulty to performing echocardiograms in a very obese population as well.”
Both the moderators of the pediatrics section, Christopher Carroll, MD, FCCP, of Connecticut Children’s Medical Center in Hartford, and Shahid Sheikh, MD, FCCP, of Nationwide Children’s Hospital in Columbus, Ohio, were impressed with the research. Dr. Carroll called it a “very elegant” study, and Dr. Sheikh noted the need for these studies in pediatrics “so that we don’t have to rely on grown-up data,” which may or may not generalize to children.
SOURCE: CHEST 2018. https://journal.chestnet.org/article/S0012-3692(18)31935-4/fulltext
SAN ANTONIO – Obstructive sleep apnea (OSA) in children is associated with an abnormal metabolic profile, but not with body mass index (BMI), according to new research.
“Screening for metabolic dysfunction in obese children with obstructive sleep apnea can help identify those at risk for cardiovascular complications,” Kanika Mathur, MD, of the Albert Einstein College of Medicine and the Children’s Hospital at Montefiore, both in New York, told attendees at the annual meeting of the American College of Chest Physicians. Dr. Mathur explained that no consensus currently exists regarding routine cardiac evaluation of children with OSA.
“The American Academy of Pediatrics does not mention any sort of cardiac evaluation in children with OSA while the most recent guidelines from the American Heart Association and the American Thoracic Society recommend echocardiographic evaluation in children with severe obstructive sleep apnea, specifically to evaluate for pulmonary hypertension and right ventricular dysfunction,” Dr. Mathur told attendees.
OSA’s association with obesity, diabetes, and hypertension is well established in adults. It is an independent risk factor for coronary artery disease, heart failure, stroke and atrial fibrillation, and research has suggested OSA treatment can reduce cardiovascular risk in adults, Dr. Mathur explained, but little data on children exist. She and her colleagues set out to understand the relationship of OSA in children with various measures of cardiovascular and metabolic health.
“Despite similar degrees of obesity and systemic blood pressure, pediatric patients with OSA had significantly higher diastolic blood pressure, heart rate, and abnormal metabolic profile, including elevated alanine transaminase, aspartate transaminase, triglycerides and hemoglobin A1c,” they found.
Their study included patients aged 3-21 years with a BMI of at least the 95th percentile who had undergone sleep study and an echocardiogram at the Children’s Hospital at Montefiore between November 2016 and November 2017.
They excluded those with comorbidities related to cardiovascular morbidity: heart disease, neuromuscular disease, sickle cell disease, rheumatologic diseases, significant cranial facial abnormalities, tracheostomy, and any lung disease. However, 7% of the patients had trisomy 21.
Among the 81 children who met their criteria, 37 were male and 44 were female, with an average age of 14 years old and a mean BMI of 39.4 kg/m2 (mean BMI z score of 2.22). Most of the patients (53.1%) had severe OSA (apnea-hypopnea index of at least 10), 21% had moderate OSA (AHI 5-9.9), 12.3% had mild OSA (AHI 2-4.9), and 13.6% did not have OSA. The median AHI of the children was 10.3.
Among all the children, “about half had elevated systolic blood pressure, which is already a risk factor for cardiovascular morbidity,” Mathur reported.
BMI, BMI z score, systolic blood pressure z score, oxygen saturation and cholesterol (overall and both HDL and LDL cholesterol levels) did not significantly differ between children who had OSA and those who did not, but diastolic blood pressure and heart rate did. Those with OSA had a diastolic blood pressure of 65 mm Hg, compared with 58 mm Hg without OSA (P = .008). Heart rate was 89 bpm in the children with OSA, compared with 78 bpm in those without (P = .004).
The children with OSA also showed higher mean levels of several other metabolic biomarkers:
- Alanine transaminase: 26 U/L with OSA vs. 18 U/L without (P = .01).
- Aspartate transaminase: 23 U/L with OSA vs. 18 U/L without (P = .03).
- Triglycerides: 138 mg/dL with OSA vs 84 mg/dL without (P = .004).
- Hemoglobin A1c: 6.2% with OSA vs. 5.4% without (P = .002).
Children with and without OSA did not have any significant differences in left atrial indexed volume, left ventricular volume, left ventricular ejection fraction, or left ventricular mass (measured by M-mode or 5/6 area length formula). Though research has shown these measures to differ in adults with and without OSA, evidence on echocardiographic changes in children has been conflicting, Dr. Mathur noted.
The researchers also conducted subanalyses according to OSA severity, but BMI, BMI Z-score, systolic or diastolic blood pressure Z-score, heart rate and oxygen saturation did not differ between those with mild OSA vs those with moderate or severe OSA. No differences in echocardiographic measurements existed between these subgroups, either.
However, children with moderate to severe OSA did have higher alanine transaminase (27 U/L with moderate to severe vs. 17 U/L with mild OSA; P = .005) and higher triglycerides (148 vs 74; P = .001).
“Certainly we need further evaluation to see the efficacy of obstructive sleep apnea therapies on metabolic dysfunction and whether weight loss needs to be an adjunct therapy for these patients,” Dr. Mathur told attendees. She also noted the need to define the role of echocardiography in managing children with OSA.
The study had several limitations, including its retrospective cross-sectional nature at a single center and its small sample size.
“Additionally, we have a wide variety of ages, which could represent different pathophysiology of the associated metabolic dysfunction in these patients,” Mathur said. “There is an inherent difficulty to performing echocardiograms in a very obese population as well.”
Both the moderators of the pediatrics section, Christopher Carroll, MD, FCCP, of Connecticut Children’s Medical Center in Hartford, and Shahid Sheikh, MD, FCCP, of Nationwide Children’s Hospital in Columbus, Ohio, were impressed with the research. Dr. Carroll called it a “very elegant” study, and Dr. Sheikh noted the need for these studies in pediatrics “so that we don’t have to rely on grown-up data,” which may or may not generalize to children.
SOURCE: CHEST 2018. https://journal.chestnet.org/article/S0012-3692(18)31935-4/fulltext
REPORTING FROM CHEST 2018
Key clinical point: Children with obesity and obstructive sleep apnea have an abnormal metabolic profile.
Major finding: Diastolic blood pressure (65 vs. 58 mm Hg), heart rate (89 vs. 78 bpm), triglycerides (138 vs. 84 mg/dL), alanine transaminase (26 vs. 18 U/L), aspartate transaminase (23 vs. 18 U/L) and hemoglobin A1c (6.2% vs. 5.4%) were all elevated in obese children with OSA, compared with obese children without OSA.
Study details: The findings are based on a retrospective analysis of 81 patients aged 3-21 years, from the Children’s Hospital at Montefiore between November 2016-November 2017.
Disclosures: No external funding was noted. The authors reported having no disclosures.
Study explores link between GERD and poor sleep quality
ATLANTA – results from an ongoing longitudinal analysis demonstrated.
“We have little longitudinal information on GERD in the general population; the last published article on GERD incidence was 20 years ago,” lead study author Maurice M. Ohayon, MD, DSc, PhD, said in an interview in advance of the annual meeting of the American Neurological Association. “As a sleep specialist, I am always interested to see how a specific medical condition may affect the sleep quality of the individuals with that condition. How we live our day has an impact on our night; it works together.”
In an effort to examine the long-term effects of GERD on sleep disturbances, Dr. Ohayon, director of the Stanford (Calif.) Sleep Epidemiology Research Center, and his colleagues used U.S. Census data to identify a random sample of adults in Arizona, California, Colorado, Idaho, New York, Oregon, Pennsylvania, and Texas. The researchers conducted two waves of phone interviews with the subjects 3 years apart, beginning in 2004. They limited their analysis to 10,930 subjects with a mean age of 43 years who participated in both interviews.
Between wave 1 and wave 2 of phone interviews, the proportion of adults who reported having GERD rose from 10.6% to 12.4% and the prevalence of new GERD cases was 8.5% per year, while the incidence was 3.2% per year. Chronic GERD, defined as that present during both interview periods, was observed in 3.9% of the sample.
The researchers found that 77.3% of GERD subjects were taking a treatment to alleviate their symptoms, mostly proton-pump inhibitors. Those with chronic GERD were more likely to report being dissatisfied with their sleep during wave 2 of the study, compared with wave 1 (24.2% vs. 13.5%; P less than .001). In addition, compared with their non-GERD counterparts, those with chronic GERD were more likely to wake up at night (33.9% vs. 28.3%; P less than .001) and to have nonrestorative sleep (15.6% vs. 10.5%; P less than .001).
“Discomfort related to GERD may happen while you are sleeping,” said Dr. Ohayon, who is also a professor of psychiatry and behavioral sciences at Stanford University. “It may wake you up and, if not, it may make you feel unrested when you wake up. We observed both of these symptoms in our GERD participants. Insomnia disorders were also rampant in the chronic GERD group (24.5%, compared with 14.4% in non-GERD participants). An insomnia disorder is more than just having difficulty falling asleep or waking up at night, it means that your daytime functioning is affected by the poor quality of your night.”
Dr. Ohayon said other findings from the study were “rather alarming.” For example, individuals with GERD, especially those with the chronic form, weighed much more than those with no GERD did. “Over a 3-year period, the chronic GERD individuals gained one point in the body mass index, which for a 6-foot tall man translates into a weight gain of 30 pounds,” he said. “Of course, with that follows high blood pressure, high cholesterol, diabetes, chronic pain, and heart disease.”
He concluded that GERD has its main manifestations when affected individuals are sleeping on their backs. “The impact of GERD on the quality of sleep is major,” he said. “Sleepiness and fatigue during the day are the consequences impacting work, family, and quality of life.”
Dr. Ohayon acknowledged certain limitations of the study, including the fact that GERD was based on self-report. The study was supported by an unrestricted grant from Takeda.
Source: Oyahon et al. ANA 2018, Abstract 625.
ATLANTA – results from an ongoing longitudinal analysis demonstrated.
“We have little longitudinal information on GERD in the general population; the last published article on GERD incidence was 20 years ago,” lead study author Maurice M. Ohayon, MD, DSc, PhD, said in an interview in advance of the annual meeting of the American Neurological Association. “As a sleep specialist, I am always interested to see how a specific medical condition may affect the sleep quality of the individuals with that condition. How we live our day has an impact on our night; it works together.”
In an effort to examine the long-term effects of GERD on sleep disturbances, Dr. Ohayon, director of the Stanford (Calif.) Sleep Epidemiology Research Center, and his colleagues used U.S. Census data to identify a random sample of adults in Arizona, California, Colorado, Idaho, New York, Oregon, Pennsylvania, and Texas. The researchers conducted two waves of phone interviews with the subjects 3 years apart, beginning in 2004. They limited their analysis to 10,930 subjects with a mean age of 43 years who participated in both interviews.
Between wave 1 and wave 2 of phone interviews, the proportion of adults who reported having GERD rose from 10.6% to 12.4% and the prevalence of new GERD cases was 8.5% per year, while the incidence was 3.2% per year. Chronic GERD, defined as that present during both interview periods, was observed in 3.9% of the sample.
The researchers found that 77.3% of GERD subjects were taking a treatment to alleviate their symptoms, mostly proton-pump inhibitors. Those with chronic GERD were more likely to report being dissatisfied with their sleep during wave 2 of the study, compared with wave 1 (24.2% vs. 13.5%; P less than .001). In addition, compared with their non-GERD counterparts, those with chronic GERD were more likely to wake up at night (33.9% vs. 28.3%; P less than .001) and to have nonrestorative sleep (15.6% vs. 10.5%; P less than .001).
“Discomfort related to GERD may happen while you are sleeping,” said Dr. Ohayon, who is also a professor of psychiatry and behavioral sciences at Stanford University. “It may wake you up and, if not, it may make you feel unrested when you wake up. We observed both of these symptoms in our GERD participants. Insomnia disorders were also rampant in the chronic GERD group (24.5%, compared with 14.4% in non-GERD participants). An insomnia disorder is more than just having difficulty falling asleep or waking up at night, it means that your daytime functioning is affected by the poor quality of your night.”
Dr. Ohayon said other findings from the study were “rather alarming.” For example, individuals with GERD, especially those with the chronic form, weighed much more than those with no GERD did. “Over a 3-year period, the chronic GERD individuals gained one point in the body mass index, which for a 6-foot tall man translates into a weight gain of 30 pounds,” he said. “Of course, with that follows high blood pressure, high cholesterol, diabetes, chronic pain, and heart disease.”
He concluded that GERD has its main manifestations when affected individuals are sleeping on their backs. “The impact of GERD on the quality of sleep is major,” he said. “Sleepiness and fatigue during the day are the consequences impacting work, family, and quality of life.”
Dr. Ohayon acknowledged certain limitations of the study, including the fact that GERD was based on self-report. The study was supported by an unrestricted grant from Takeda.
Source: Oyahon et al. ANA 2018, Abstract 625.
ATLANTA – results from an ongoing longitudinal analysis demonstrated.
“We have little longitudinal information on GERD in the general population; the last published article on GERD incidence was 20 years ago,” lead study author Maurice M. Ohayon, MD, DSc, PhD, said in an interview in advance of the annual meeting of the American Neurological Association. “As a sleep specialist, I am always interested to see how a specific medical condition may affect the sleep quality of the individuals with that condition. How we live our day has an impact on our night; it works together.”
In an effort to examine the long-term effects of GERD on sleep disturbances, Dr. Ohayon, director of the Stanford (Calif.) Sleep Epidemiology Research Center, and his colleagues used U.S. Census data to identify a random sample of adults in Arizona, California, Colorado, Idaho, New York, Oregon, Pennsylvania, and Texas. The researchers conducted two waves of phone interviews with the subjects 3 years apart, beginning in 2004. They limited their analysis to 10,930 subjects with a mean age of 43 years who participated in both interviews.
Between wave 1 and wave 2 of phone interviews, the proportion of adults who reported having GERD rose from 10.6% to 12.4% and the prevalence of new GERD cases was 8.5% per year, while the incidence was 3.2% per year. Chronic GERD, defined as that present during both interview periods, was observed in 3.9% of the sample.
The researchers found that 77.3% of GERD subjects were taking a treatment to alleviate their symptoms, mostly proton-pump inhibitors. Those with chronic GERD were more likely to report being dissatisfied with their sleep during wave 2 of the study, compared with wave 1 (24.2% vs. 13.5%; P less than .001). In addition, compared with their non-GERD counterparts, those with chronic GERD were more likely to wake up at night (33.9% vs. 28.3%; P less than .001) and to have nonrestorative sleep (15.6% vs. 10.5%; P less than .001).
“Discomfort related to GERD may happen while you are sleeping,” said Dr. Ohayon, who is also a professor of psychiatry and behavioral sciences at Stanford University. “It may wake you up and, if not, it may make you feel unrested when you wake up. We observed both of these symptoms in our GERD participants. Insomnia disorders were also rampant in the chronic GERD group (24.5%, compared with 14.4% in non-GERD participants). An insomnia disorder is more than just having difficulty falling asleep or waking up at night, it means that your daytime functioning is affected by the poor quality of your night.”
Dr. Ohayon said other findings from the study were “rather alarming.” For example, individuals with GERD, especially those with the chronic form, weighed much more than those with no GERD did. “Over a 3-year period, the chronic GERD individuals gained one point in the body mass index, which for a 6-foot tall man translates into a weight gain of 30 pounds,” he said. “Of course, with that follows high blood pressure, high cholesterol, diabetes, chronic pain, and heart disease.”
He concluded that GERD has its main manifestations when affected individuals are sleeping on their backs. “The impact of GERD on the quality of sleep is major,” he said. “Sleepiness and fatigue during the day are the consequences impacting work, family, and quality of life.”
Dr. Ohayon acknowledged certain limitations of the study, including the fact that GERD was based on self-report. The study was supported by an unrestricted grant from Takeda.
Source: Oyahon et al. ANA 2018, Abstract 625.
REPORTING FROM ANA 2018
Key clinical point: GERD has a major impact on quality of sleep.
Major finding: Study participants with chronic GERD were more likely to report being dissatisfied with their sleep during wave 2 of the study, compared with wave 1 (24.2% vs. 13.5%; P less than .001).
Study details: A telephone-based survey of 10,930 U.S. adults who were interviewed during two waves 3 years apart.
Disclosures: The study was supported by an unrestricted grant from Takeda.
Source: Oyahon et al. ANA 2018, Abstract 625.














