User login
Think outside lower body for pelvic pain
Also today, treating obstructive sleep apnea with positive airway pressure decreased amyloid levels, spending on medical marketing increased by more than $12 billion over that past two decades, and one expert has advice on how you can get your work published.
Amazon Alexa
Apple Podcasts
Google Podcasts
Spotify
Also today, treating obstructive sleep apnea with positive airway pressure decreased amyloid levels, spending on medical marketing increased by more than $12 billion over that past two decades, and one expert has advice on how you can get your work published.
Amazon Alexa
Apple Podcasts
Google Podcasts
Spotify
Also today, treating obstructive sleep apnea with positive airway pressure decreased amyloid levels, spending on medical marketing increased by more than $12 billion over that past two decades, and one expert has advice on how you can get your work published.
Amazon Alexa
Apple Podcasts
Google Podcasts
Spotify
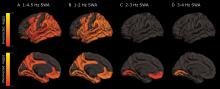
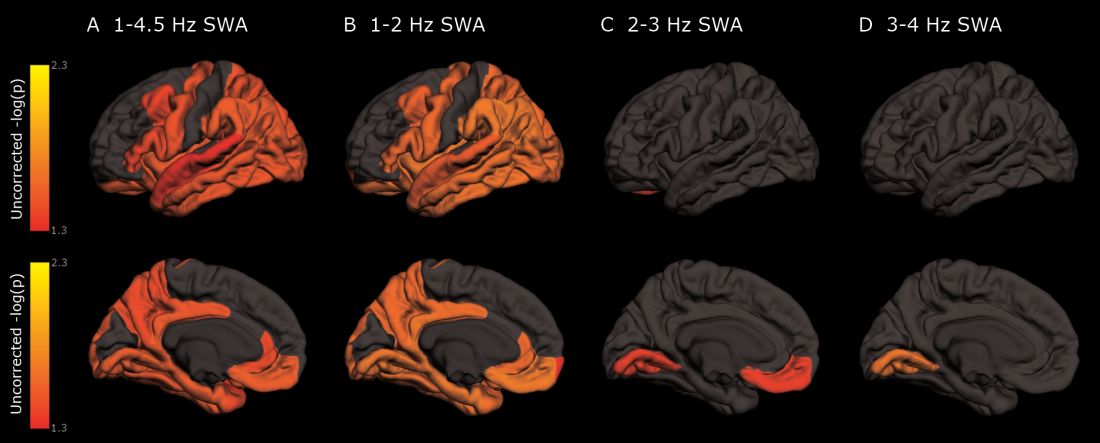
As deep sleep decreases, Alzheimer’s pathology – particularly tau – increases
The protein was evident in areas associated with memory consolidation, typically affected in Alzheimer’s disease: the entorhinal, parahippocampal, inferior parietal, insula, isthmus cingulate, lingual, supramarginal, and orbitofrontal regions.
Because the findings were observed in a population of cognitively normal and minimally impaired subjects, they suggest a role for sleep studies in assessing the risk for cognitive decline and Alzheimer’s disease, and in monitoring patients with the disease, reported Brendan P. Lucey, MD, and his colleagues. The report is in Science and Translational Medicine (Sci Transl Med. 2019 Jan 9;11:eaau6550).
“With the rising incidence of Alzheimer’s disease in an aging population, our findings have potential application in both clinical trials and patient screening for Alzheimer’s disease to noninvasively monitor for progression of Alzheimer’s disease pathology,” wrote Dr. Lucey, director of the Sleep Medicine Center and assistant professor of neurology at Washington University in St. Louis. “For instance, periodically measuring non-REM slow wave activity, in conjunction with other biomarkers, may have utility for monitoring Alzheimer’s disease risk or response to an Alzheimer’s disease treatment.”
Dr. Lucey and his colleagues examined sleep architecture and tau and amyloid deposition in 119 subjects enrolled in longitudinal aging studies. For 6 nights, subjects slept with a single-channel EEG monitor on. They also underwent cognitive testing and genotyping for Alzheimer’s disease risk factors.
Subjects were a mean of 74 years old. Almost 80% had normal cognition as measured by the Clinical Dementia Rating Scale (CDR); the remainder had very mild cognitive impairment (CDR 0.5)
Among those with positive biomarker findings, sleep architecture was altered in several ways: lower REM latency, lower wake after sleep onset, prolonged sleep-onset latency, and longer self-reported total sleep time. The differences were evident in those with normal cognition, but even more pronounced in those with mild cognitive impairment. Despite the longer sleep times, however, sleep efficiency was decreased.
Decreased non-REM slow wave activity was associated with increased tau deposition. The protein was largely concentrated in areas of typical Alzheimer’s disease pathology (entorhinal, parahippocampal, orbital frontal, precuneus, inferior parietal, and inferior temporal regions). There were no significant associations between non-REM slow wave activity and amyloid deposits.
Other sleep parameters, however, were associated with amyloid, including REM latency and sleep latency, “suggesting that as amyloid-beta deposition increased, the time to fall asleep and enter REM sleep decreased,” the investigators said.
Those with tau pathology also slept longer, reporting more daytime naps. “This suggests that participants with greater tau pathology experienced daytime sleepiness despite increased total sleep time.”
“These results, coupled with the non-REM slow wave activity findings, suggest that the quality of sleep decreases with increasing tau despite increased sleep time.” Questions about napping should probably be included in dementia screening discussions, they said.
The study was largely funded by the National Institutes of Health. Dr. Lucey had no financial conflicts.
SOURCE: Lucey BP et al. Sci Transl Med 2019 Jan 9;11:eaau6550.
The protein was evident in areas associated with memory consolidation, typically affected in Alzheimer’s disease: the entorhinal, parahippocampal, inferior parietal, insula, isthmus cingulate, lingual, supramarginal, and orbitofrontal regions.
Because the findings were observed in a population of cognitively normal and minimally impaired subjects, they suggest a role for sleep studies in assessing the risk for cognitive decline and Alzheimer’s disease, and in monitoring patients with the disease, reported Brendan P. Lucey, MD, and his colleagues. The report is in Science and Translational Medicine (Sci Transl Med. 2019 Jan 9;11:eaau6550).
“With the rising incidence of Alzheimer’s disease in an aging population, our findings have potential application in both clinical trials and patient screening for Alzheimer’s disease to noninvasively monitor for progression of Alzheimer’s disease pathology,” wrote Dr. Lucey, director of the Sleep Medicine Center and assistant professor of neurology at Washington University in St. Louis. “For instance, periodically measuring non-REM slow wave activity, in conjunction with other biomarkers, may have utility for monitoring Alzheimer’s disease risk or response to an Alzheimer’s disease treatment.”
Dr. Lucey and his colleagues examined sleep architecture and tau and amyloid deposition in 119 subjects enrolled in longitudinal aging studies. For 6 nights, subjects slept with a single-channel EEG monitor on. They also underwent cognitive testing and genotyping for Alzheimer’s disease risk factors.
Subjects were a mean of 74 years old. Almost 80% had normal cognition as measured by the Clinical Dementia Rating Scale (CDR); the remainder had very mild cognitive impairment (CDR 0.5)
Among those with positive biomarker findings, sleep architecture was altered in several ways: lower REM latency, lower wake after sleep onset, prolonged sleep-onset latency, and longer self-reported total sleep time. The differences were evident in those with normal cognition, but even more pronounced in those with mild cognitive impairment. Despite the longer sleep times, however, sleep efficiency was decreased.
Decreased non-REM slow wave activity was associated with increased tau deposition. The protein was largely concentrated in areas of typical Alzheimer’s disease pathology (entorhinal, parahippocampal, orbital frontal, precuneus, inferior parietal, and inferior temporal regions). There were no significant associations between non-REM slow wave activity and amyloid deposits.
Other sleep parameters, however, were associated with amyloid, including REM latency and sleep latency, “suggesting that as amyloid-beta deposition increased, the time to fall asleep and enter REM sleep decreased,” the investigators said.
Those with tau pathology also slept longer, reporting more daytime naps. “This suggests that participants with greater tau pathology experienced daytime sleepiness despite increased total sleep time.”
“These results, coupled with the non-REM slow wave activity findings, suggest that the quality of sleep decreases with increasing tau despite increased sleep time.” Questions about napping should probably be included in dementia screening discussions, they said.
The study was largely funded by the National Institutes of Health. Dr. Lucey had no financial conflicts.
SOURCE: Lucey BP et al. Sci Transl Med 2019 Jan 9;11:eaau6550.
The protein was evident in areas associated with memory consolidation, typically affected in Alzheimer’s disease: the entorhinal, parahippocampal, inferior parietal, insula, isthmus cingulate, lingual, supramarginal, and orbitofrontal regions.
Because the findings were observed in a population of cognitively normal and minimally impaired subjects, they suggest a role for sleep studies in assessing the risk for cognitive decline and Alzheimer’s disease, and in monitoring patients with the disease, reported Brendan P. Lucey, MD, and his colleagues. The report is in Science and Translational Medicine (Sci Transl Med. 2019 Jan 9;11:eaau6550).
“With the rising incidence of Alzheimer’s disease in an aging population, our findings have potential application in both clinical trials and patient screening for Alzheimer’s disease to noninvasively monitor for progression of Alzheimer’s disease pathology,” wrote Dr. Lucey, director of the Sleep Medicine Center and assistant professor of neurology at Washington University in St. Louis. “For instance, periodically measuring non-REM slow wave activity, in conjunction with other biomarkers, may have utility for monitoring Alzheimer’s disease risk or response to an Alzheimer’s disease treatment.”
Dr. Lucey and his colleagues examined sleep architecture and tau and amyloid deposition in 119 subjects enrolled in longitudinal aging studies. For 6 nights, subjects slept with a single-channel EEG monitor on. They also underwent cognitive testing and genotyping for Alzheimer’s disease risk factors.
Subjects were a mean of 74 years old. Almost 80% had normal cognition as measured by the Clinical Dementia Rating Scale (CDR); the remainder had very mild cognitive impairment (CDR 0.5)
Among those with positive biomarker findings, sleep architecture was altered in several ways: lower REM latency, lower wake after sleep onset, prolonged sleep-onset latency, and longer self-reported total sleep time. The differences were evident in those with normal cognition, but even more pronounced in those with mild cognitive impairment. Despite the longer sleep times, however, sleep efficiency was decreased.
Decreased non-REM slow wave activity was associated with increased tau deposition. The protein was largely concentrated in areas of typical Alzheimer’s disease pathology (entorhinal, parahippocampal, orbital frontal, precuneus, inferior parietal, and inferior temporal regions). There were no significant associations between non-REM slow wave activity and amyloid deposits.
Other sleep parameters, however, were associated with amyloid, including REM latency and sleep latency, “suggesting that as amyloid-beta deposition increased, the time to fall asleep and enter REM sleep decreased,” the investigators said.
Those with tau pathology also slept longer, reporting more daytime naps. “This suggests that participants with greater tau pathology experienced daytime sleepiness despite increased total sleep time.”
“These results, coupled with the non-REM slow wave activity findings, suggest that the quality of sleep decreases with increasing tau despite increased sleep time.” Questions about napping should probably be included in dementia screening discussions, they said.
The study was largely funded by the National Institutes of Health. Dr. Lucey had no financial conflicts.
SOURCE: Lucey BP et al. Sci Transl Med 2019 Jan 9;11:eaau6550.
FROM SCIENCE TRANSLATIONAL MEDICINE
Key clinical point: Cognitively normal subjects with tau deposition experience altered sleep patterns.
Major finding: Decreased time in non-REM deep sleep was associated with increased tau pathology in Alzheimer’s-affected brain regions and in cerebrospinal fluid.
Study details: The prospective longitudinal study comprised 119 subjects.
Disclosures: The authors reported no relevant financial disclosures.
Source: Lucey BP et al. Sci Transl Med. 2019 Jan 9;11:eaau6550.
Alcohol use, psychological distress associated with possible RBD
(RBD), according to a population-based cohort study published in Neurology. In addition, the results also replicate previous findings of an association between possible RBD and smoking, low education, and male sex.
The risk factors for RBD have been studied comparatively little. “While much is still unknown about RBD, it can be caused by medications or it may be an early sign of another neurologic condition like Parkinson’s disease, dementia with Lewy bodies, or multiple system atrophy,” according to Ronald B. Postuma, MD, an associate professor at McGill University, Montreal. “Identifying lifestyle and personal risk factors linked to this sleep disorder may lead to finding ways to reduce the chances of developing it.”
To assess sociodemographic, socioeconomic, and clinical correlates of possible RBD, Dr. Postuma and his colleagues examined baseline data collected between 2012 and 2015 in the Canadian Longitudinal Study on Aging (CLSA), which included 30,097 participants. To screen for possible RBD, the CLSA researchers asked patients, “Have you ever been told, or suspected yourself, that you seem to ‘act out your dreams’ while asleep [e.g., punching, flailing your arms in the air, making running movements, etc.]?” Participants answered additional questions to rule out RBD mimics. Patients with symptom onset before age 20 years, positive apnea screen, or a diagnosis of dementia, Alzheimer’s disease, parkinsonism, or Parkinson’s disease were excluded from analysis.
In all, 3,271 participants screened positive for possible RBD. After the investigators excluded participants with potential mimics, 958 patients (about 3.2% of the total population) remained in the analysis. Approximately 59% of patients with possible RBD were male, compared with 42% of controls. Patients with possible RBD were more likely to be married, in a common-law relationship, or widowed.
Participants with possible RBD had slightly less education (estimated mean, 13.2 years vs. 13.6 years) and lower income, compared with controls. Participants with possible RBD retired at a slightly younger age (57.5 years vs. 58.6 years) and were more likely to have retired because of health concerns (28.9% vs. 22.0%), compared with controls.
In addition, patients with possible RBD were more likely to drink more and to be moderate to heavy drinkers than controls; they were also more likely to be current or past smokers. Antidepressant use was more frequent and psychological distress was greater among participants with possible RBD.
When the investigators performed a multivariable logistic regression analysis, the associations between possible RBD and male sex and relationship status remained. Lower educational level, but not income level, also remained associated with possible RBD. Furthermore, retirement age and having reported retirement because of health concerns remained significantly associated with possible RBD, as did the amount of alcohol consumed weekly and moderate to heavy drinking. Sensitivity analyses did not change the results significantly.
One of the study’s limitations is its reliance on self-report to identify participants with possible RBD, the authors wrote. The prevalence of possible RBD in the study was 3.2%, but research using polysomnography has found a prevalence of about 1%. Thus, the majority of cases in this study may have other disorders such as restless legs syndrome or periodic limb movements. Furthermore, many participants who enact their dreams (such as unmarried people) are likely unaware of it. Finally, the researchers did not measure several variables of interest, such as consumption of caffeinated products.
“The main advantages of our current study are the large sample size; the systematic population-based sampling; the capacity to adjust for diverse potential confounding variables, including mental illness; and the ability to screen out RBD mimics,” the authors concluded.
SOURCE: Postuma RB et al. Neurology. 2018 Dec 26. doi: 10.1212/WNL.0000000000006849.
(RBD), according to a population-based cohort study published in Neurology. In addition, the results also replicate previous findings of an association between possible RBD and smoking, low education, and male sex.
The risk factors for RBD have been studied comparatively little. “While much is still unknown about RBD, it can be caused by medications or it may be an early sign of another neurologic condition like Parkinson’s disease, dementia with Lewy bodies, or multiple system atrophy,” according to Ronald B. Postuma, MD, an associate professor at McGill University, Montreal. “Identifying lifestyle and personal risk factors linked to this sleep disorder may lead to finding ways to reduce the chances of developing it.”
To assess sociodemographic, socioeconomic, and clinical correlates of possible RBD, Dr. Postuma and his colleagues examined baseline data collected between 2012 and 2015 in the Canadian Longitudinal Study on Aging (CLSA), which included 30,097 participants. To screen for possible RBD, the CLSA researchers asked patients, “Have you ever been told, or suspected yourself, that you seem to ‘act out your dreams’ while asleep [e.g., punching, flailing your arms in the air, making running movements, etc.]?” Participants answered additional questions to rule out RBD mimics. Patients with symptom onset before age 20 years, positive apnea screen, or a diagnosis of dementia, Alzheimer’s disease, parkinsonism, or Parkinson’s disease were excluded from analysis.
In all, 3,271 participants screened positive for possible RBD. After the investigators excluded participants with potential mimics, 958 patients (about 3.2% of the total population) remained in the analysis. Approximately 59% of patients with possible RBD were male, compared with 42% of controls. Patients with possible RBD were more likely to be married, in a common-law relationship, or widowed.
Participants with possible RBD had slightly less education (estimated mean, 13.2 years vs. 13.6 years) and lower income, compared with controls. Participants with possible RBD retired at a slightly younger age (57.5 years vs. 58.6 years) and were more likely to have retired because of health concerns (28.9% vs. 22.0%), compared with controls.
In addition, patients with possible RBD were more likely to drink more and to be moderate to heavy drinkers than controls; they were also more likely to be current or past smokers. Antidepressant use was more frequent and psychological distress was greater among participants with possible RBD.
When the investigators performed a multivariable logistic regression analysis, the associations between possible RBD and male sex and relationship status remained. Lower educational level, but not income level, also remained associated with possible RBD. Furthermore, retirement age and having reported retirement because of health concerns remained significantly associated with possible RBD, as did the amount of alcohol consumed weekly and moderate to heavy drinking. Sensitivity analyses did not change the results significantly.
One of the study’s limitations is its reliance on self-report to identify participants with possible RBD, the authors wrote. The prevalence of possible RBD in the study was 3.2%, but research using polysomnography has found a prevalence of about 1%. Thus, the majority of cases in this study may have other disorders such as restless legs syndrome or periodic limb movements. Furthermore, many participants who enact their dreams (such as unmarried people) are likely unaware of it. Finally, the researchers did not measure several variables of interest, such as consumption of caffeinated products.
“The main advantages of our current study are the large sample size; the systematic population-based sampling; the capacity to adjust for diverse potential confounding variables, including mental illness; and the ability to screen out RBD mimics,” the authors concluded.
SOURCE: Postuma RB et al. Neurology. 2018 Dec 26. doi: 10.1212/WNL.0000000000006849.
(RBD), according to a population-based cohort study published in Neurology. In addition, the results also replicate previous findings of an association between possible RBD and smoking, low education, and male sex.
The risk factors for RBD have been studied comparatively little. “While much is still unknown about RBD, it can be caused by medications or it may be an early sign of another neurologic condition like Parkinson’s disease, dementia with Lewy bodies, or multiple system atrophy,” according to Ronald B. Postuma, MD, an associate professor at McGill University, Montreal. “Identifying lifestyle and personal risk factors linked to this sleep disorder may lead to finding ways to reduce the chances of developing it.”
To assess sociodemographic, socioeconomic, and clinical correlates of possible RBD, Dr. Postuma and his colleagues examined baseline data collected between 2012 and 2015 in the Canadian Longitudinal Study on Aging (CLSA), which included 30,097 participants. To screen for possible RBD, the CLSA researchers asked patients, “Have you ever been told, or suspected yourself, that you seem to ‘act out your dreams’ while asleep [e.g., punching, flailing your arms in the air, making running movements, etc.]?” Participants answered additional questions to rule out RBD mimics. Patients with symptom onset before age 20 years, positive apnea screen, or a diagnosis of dementia, Alzheimer’s disease, parkinsonism, or Parkinson’s disease were excluded from analysis.
In all, 3,271 participants screened positive for possible RBD. After the investigators excluded participants with potential mimics, 958 patients (about 3.2% of the total population) remained in the analysis. Approximately 59% of patients with possible RBD were male, compared with 42% of controls. Patients with possible RBD were more likely to be married, in a common-law relationship, or widowed.
Participants with possible RBD had slightly less education (estimated mean, 13.2 years vs. 13.6 years) and lower income, compared with controls. Participants with possible RBD retired at a slightly younger age (57.5 years vs. 58.6 years) and were more likely to have retired because of health concerns (28.9% vs. 22.0%), compared with controls.
In addition, patients with possible RBD were more likely to drink more and to be moderate to heavy drinkers than controls; they were also more likely to be current or past smokers. Antidepressant use was more frequent and psychological distress was greater among participants with possible RBD.
When the investigators performed a multivariable logistic regression analysis, the associations between possible RBD and male sex and relationship status remained. Lower educational level, but not income level, also remained associated with possible RBD. Furthermore, retirement age and having reported retirement because of health concerns remained significantly associated with possible RBD, as did the amount of alcohol consumed weekly and moderate to heavy drinking. Sensitivity analyses did not change the results significantly.
One of the study’s limitations is its reliance on self-report to identify participants with possible RBD, the authors wrote. The prevalence of possible RBD in the study was 3.2%, but research using polysomnography has found a prevalence of about 1%. Thus, the majority of cases in this study may have other disorders such as restless legs syndrome or periodic limb movements. Furthermore, many participants who enact their dreams (such as unmarried people) are likely unaware of it. Finally, the researchers did not measure several variables of interest, such as consumption of caffeinated products.
“The main advantages of our current study are the large sample size; the systematic population-based sampling; the capacity to adjust for diverse potential confounding variables, including mental illness; and the ability to screen out RBD mimics,” the authors concluded.
SOURCE: Postuma RB et al. Neurology. 2018 Dec 26. doi: 10.1212/WNL.0000000000006849.
FROM NEUROLOGY
Key clinical point: Alcohol use and psychological distress are associated with possible REM sleep behavior disorder.
Major finding: A self-report questionnaire yielded a 3.2% prevalence of possible REM sleep behavior disorder.
Study details: A prospective, population-based cohort study of 30,097 participants.
Disclosures: The Canadian government provided funding for the research.
Source: Postuma RB et al. Neurology. 2018 Dec 26. doi: 10.1212/WNL.0000000000006849.
Treating OSA with positive airway pressure decreased amyloid levels in CSF
Soluble amyloid-beta in cerebrospinal fluid (CSF) decreased when subjects with obstructive sleep apnea used a positive airway pressure device with good adherence, suggesting that improving sleep could reduce the risk of Alzheimer’s disease in this population.
The small decrease in cerebrospinal amyloid-beta 40 (Ab40) and Ab42 hints at decreased neuronal release of the neurotoxic protein, wrote Yo-El S. Ju, MD, and her colleagues. The report was published online in Annals of Neurology.
Alzheimer’s disease (AD) biomarker studies typically find decreased CSF levels associated with increased Ab brain plaques. But before plaques form, increased soluble Ab in CSF is a risk factor for aggregation. Thus, higher soluble Ab levels in mid-life may suggest a risk of later Ab pathology, wrote Dr. Ju of Washington University, St. Louis.
“We tested individuals without any AD pathology as assessed by Ab42 [in CSF], a highly sensitive biomarker of amyloid plaques,” Dr. Ju and her coauthors wrote. “This means our study findings can be extrapolated to the large population of people with OSA [obstructive sleep apnea], many of whom are middle-aged or younger, and have many years to accrue benefit from AD risk reduction ... The effect of OSA on SWA [slow wave activity], Ab, and possibly tau, is a probable proximal step in a cascade whereby OSA increases the risk of AD.”
The researchers recruited 35 subjects with mild to severe OSA and without abnormal Ab levels in CSF. Subjects used auto-titrating positive airway pressure (PAP) for 1-4 months; 18 were sufficiently compliant to be included in the analysis (more than 4 hours on more than 70% of 30 preceding nights as recorded by the machine). CSF was obtained after a baseline polysomnogram and after the treatment period lasting 1-4 months.
Of the 18 analyzed patients, 7 had mild OSA and 11 had moderate to severe OSA. They were an average of nearly 57 years old with a mean body mass index of 30.4 kg/m2; 7 patients had hypertension.
PAP treatment was effective, indicated by a normalized apnea-hypopnea index and decreased time in hypoxemia. Total sleep time and sleep efficiency were unchanged, but slow-wave activity did increase. As expected, hourly arousals and time in hypoxemia decreased, and hypoxic nadir shifted from an oxygen saturation of 82.5% to 91%.
“As a group, there was no significant change in Ab with treatment,” the researchers wrote. But a correlational analysis found that “greater improvement in OSA was associated with greater decrease in Ab40 and Ab42. Additionally, we found that change in tau negatively correlated with OSA improvement.”
The team suggested a two-factor model to explain the relationship between OSA and Ab levels. “Due to decreased SWA, there would be relatively increased release of Ab into the [interstitial fluid]. However, as OSA severity worsens, pressure effects of obstructive respiratory events impede the clearance of Ab and tau out of the interstitial space, resulting in lower levels in the CSF and an inverse U-shaped curve. In this model, a small improvement in OSA may result in an increase in Ab or tau, whereas a larger improvement in OSA – that ameliorates both SWA and clearance mechanisms – will result in a decrease in Ab and tau.”
The project was funded in part by Philips-Respironics, which provided the devices, and by the National Institutes of Health. Philips-Respironics had no input or role in any other part of the study. The authors had no financial disclosures.
SOURCE: Ju YS et al. Ann Neurol. 2018 Dec 31. doi: 10.1002/ana.25408.
Soluble amyloid-beta in cerebrospinal fluid (CSF) decreased when subjects with obstructive sleep apnea used a positive airway pressure device with good adherence, suggesting that improving sleep could reduce the risk of Alzheimer’s disease in this population.
The small decrease in cerebrospinal amyloid-beta 40 (Ab40) and Ab42 hints at decreased neuronal release of the neurotoxic protein, wrote Yo-El S. Ju, MD, and her colleagues. The report was published online in Annals of Neurology.
Alzheimer’s disease (AD) biomarker studies typically find decreased CSF levels associated with increased Ab brain plaques. But before plaques form, increased soluble Ab in CSF is a risk factor for aggregation. Thus, higher soluble Ab levels in mid-life may suggest a risk of later Ab pathology, wrote Dr. Ju of Washington University, St. Louis.
“We tested individuals without any AD pathology as assessed by Ab42 [in CSF], a highly sensitive biomarker of amyloid plaques,” Dr. Ju and her coauthors wrote. “This means our study findings can be extrapolated to the large population of people with OSA [obstructive sleep apnea], many of whom are middle-aged or younger, and have many years to accrue benefit from AD risk reduction ... The effect of OSA on SWA [slow wave activity], Ab, and possibly tau, is a probable proximal step in a cascade whereby OSA increases the risk of AD.”
The researchers recruited 35 subjects with mild to severe OSA and without abnormal Ab levels in CSF. Subjects used auto-titrating positive airway pressure (PAP) for 1-4 months; 18 were sufficiently compliant to be included in the analysis (more than 4 hours on more than 70% of 30 preceding nights as recorded by the machine). CSF was obtained after a baseline polysomnogram and after the treatment period lasting 1-4 months.
Of the 18 analyzed patients, 7 had mild OSA and 11 had moderate to severe OSA. They were an average of nearly 57 years old with a mean body mass index of 30.4 kg/m2; 7 patients had hypertension.
PAP treatment was effective, indicated by a normalized apnea-hypopnea index and decreased time in hypoxemia. Total sleep time and sleep efficiency were unchanged, but slow-wave activity did increase. As expected, hourly arousals and time in hypoxemia decreased, and hypoxic nadir shifted from an oxygen saturation of 82.5% to 91%.
“As a group, there was no significant change in Ab with treatment,” the researchers wrote. But a correlational analysis found that “greater improvement in OSA was associated with greater decrease in Ab40 and Ab42. Additionally, we found that change in tau negatively correlated with OSA improvement.”
The team suggested a two-factor model to explain the relationship between OSA and Ab levels. “Due to decreased SWA, there would be relatively increased release of Ab into the [interstitial fluid]. However, as OSA severity worsens, pressure effects of obstructive respiratory events impede the clearance of Ab and tau out of the interstitial space, resulting in lower levels in the CSF and an inverse U-shaped curve. In this model, a small improvement in OSA may result in an increase in Ab or tau, whereas a larger improvement in OSA – that ameliorates both SWA and clearance mechanisms – will result in a decrease in Ab and tau.”
The project was funded in part by Philips-Respironics, which provided the devices, and by the National Institutes of Health. Philips-Respironics had no input or role in any other part of the study. The authors had no financial disclosures.
SOURCE: Ju YS et al. Ann Neurol. 2018 Dec 31. doi: 10.1002/ana.25408.
Soluble amyloid-beta in cerebrospinal fluid (CSF) decreased when subjects with obstructive sleep apnea used a positive airway pressure device with good adherence, suggesting that improving sleep could reduce the risk of Alzheimer’s disease in this population.
The small decrease in cerebrospinal amyloid-beta 40 (Ab40) and Ab42 hints at decreased neuronal release of the neurotoxic protein, wrote Yo-El S. Ju, MD, and her colleagues. The report was published online in Annals of Neurology.
Alzheimer’s disease (AD) biomarker studies typically find decreased CSF levels associated with increased Ab brain plaques. But before plaques form, increased soluble Ab in CSF is a risk factor for aggregation. Thus, higher soluble Ab levels in mid-life may suggest a risk of later Ab pathology, wrote Dr. Ju of Washington University, St. Louis.
“We tested individuals without any AD pathology as assessed by Ab42 [in CSF], a highly sensitive biomarker of amyloid plaques,” Dr. Ju and her coauthors wrote. “This means our study findings can be extrapolated to the large population of people with OSA [obstructive sleep apnea], many of whom are middle-aged or younger, and have many years to accrue benefit from AD risk reduction ... The effect of OSA on SWA [slow wave activity], Ab, and possibly tau, is a probable proximal step in a cascade whereby OSA increases the risk of AD.”
The researchers recruited 35 subjects with mild to severe OSA and without abnormal Ab levels in CSF. Subjects used auto-titrating positive airway pressure (PAP) for 1-4 months; 18 were sufficiently compliant to be included in the analysis (more than 4 hours on more than 70% of 30 preceding nights as recorded by the machine). CSF was obtained after a baseline polysomnogram and after the treatment period lasting 1-4 months.
Of the 18 analyzed patients, 7 had mild OSA and 11 had moderate to severe OSA. They were an average of nearly 57 years old with a mean body mass index of 30.4 kg/m2; 7 patients had hypertension.
PAP treatment was effective, indicated by a normalized apnea-hypopnea index and decreased time in hypoxemia. Total sleep time and sleep efficiency were unchanged, but slow-wave activity did increase. As expected, hourly arousals and time in hypoxemia decreased, and hypoxic nadir shifted from an oxygen saturation of 82.5% to 91%.
“As a group, there was no significant change in Ab with treatment,” the researchers wrote. But a correlational analysis found that “greater improvement in OSA was associated with greater decrease in Ab40 and Ab42. Additionally, we found that change in tau negatively correlated with OSA improvement.”
The team suggested a two-factor model to explain the relationship between OSA and Ab levels. “Due to decreased SWA, there would be relatively increased release of Ab into the [interstitial fluid]. However, as OSA severity worsens, pressure effects of obstructive respiratory events impede the clearance of Ab and tau out of the interstitial space, resulting in lower levels in the CSF and an inverse U-shaped curve. In this model, a small improvement in OSA may result in an increase in Ab or tau, whereas a larger improvement in OSA – that ameliorates both SWA and clearance mechanisms – will result in a decrease in Ab and tau.”
The project was funded in part by Philips-Respironics, which provided the devices, and by the National Institutes of Health. Philips-Respironics had no input or role in any other part of the study. The authors had no financial disclosures.
SOURCE: Ju YS et al. Ann Neurol. 2018 Dec 31. doi: 10.1002/ana.25408.
FROM ANNALS OF NEUROLOGY
Key clinical point:
Major finding: After treatment, a correlational analysis found decreases in amyloid-beta 40 and 42.
Study details: The prospective, interventional study comprised 18 subjects.
Disclosures: The project was funded in part by Philips-Respironics, which provided the devices, and by the National Institutes of Health. Philips-Respironics had no input or role in any other part of the study. The authors had no financial disclosures.
Source: Ju YS et al. Ann Neurol. 2018 Dec 31. doi: 10.1002/ana.25408.
Prenatal valproate and ADHD
Also today, one expert calls for better ways to preserve beta cell function in youth, synthetic opioids drive a spike in the number of fatal overdoses, and mothers may play a role in the link between depression in fathers and daughters.
Amazon Alexa
Apple Podcasts
Google Podcasts
Spotify
Also today, one expert calls for better ways to preserve beta cell function in youth, synthetic opioids drive a spike in the number of fatal overdoses, and mothers may play a role in the link between depression in fathers and daughters.
Amazon Alexa
Apple Podcasts
Google Podcasts
Spotify
Also today, one expert calls for better ways to preserve beta cell function in youth, synthetic opioids drive a spike in the number of fatal overdoses, and mothers may play a role in the link between depression in fathers and daughters.
Amazon Alexa
Apple Podcasts
Google Podcasts
Spotify
Working the night shift? Strategies for improving sleep and performance
Our 24-hour society has made night shift work essential to people in many professions, including medical specialties. Working nights disrupts homeostatic and circadian rhythms, which leads to an accumulation of sleep debt (ie, the cumulative effect of not getting enough sleep).1 This debt can affect performance by impairing processing speed, concentration, mood, and physical health.1 Night shift work takes place during the period of the sleep-wake cycle that is programmed for sleep; after the shift, workers need to sleep during the period that is least conducive to sleep.1 Research indicates that a night shift worker’s sleep can be improved by scheduling light exposure and optimizing the timing of when they start their shifts.2 However, this may not be practical because night shifts usually are scheduled at particular intervals and cannot be tailored to the individual worker’s preference. Additionally, in the short term, full circadian adaptation to night shifts is impossible.1
Because sleep and performance are complex phenomena that are difficult to control, there is no single solution to maximizing these factors when one works nights.1 The most effective approach to combating the effects of night shift work is individualized and multimodal.1 However, whether you are working a night shift or are caring for a patient who does, the following nonpharmacologic strategies can help improve sleep and performance until the body naturally adapts to working this type of schedule1,3:
Minimize sleep debt before starting aseries of night shifts by not setting an alarm on the morning before the first night shift and by napping in the afternoon for approximately 45 minutes.
Take a nap during a work break (if work demands allow you to do so). However, nap for <30 minutes to avoid slow-wave sleep and subsequent grogginess when awakening.
Expose yourself to bright light immediately upon waking and for 15 minutes 2 or 3 times during a shift to promote alertness.
Drink caffeinated beverages before and during the shift to help improve concentration and reasoning (if there is no medical contraindication to consuming caffeine). However, avoid caffeine for at least 3 hours prior to going to sleep.
Add additional checks to critical tasks, such as ordering medications, during the shift, especially during the physiological nadir in the early hours of the morning.
Continue to: Create a cool, dark, quiet environment for sleep...
Create a cool, dark, quiet environment for sleep using a comfortable mattress and pillow, blackout blinds, ear plugs, and a noise machine. Also, avoid using your smartphone or tablet while trying to go to sleep. Minimize exposure to bright light on the drive home, and stick to a routine (eg, for meals and exercise).
Avoid working too many consecutive night shifts (if possible) because this can increase sleep deprivation. Also, limiting the number of night shifts and scheduling days off can speed recovery from sleep deprivation.
1. McKenna H, Wilkes M. Optimising sleep for night shifts. BMJ. 2018;360:j5637. doi: 10.1136/bmj.5637.
2. Postnova S, Robinson PA, Postnov DD. Adaptation to shift work: physiologically based modeling of the effects of lighting and shifts’ start time. PLoS One. 2013;8(1):e53379. doi: 10.1371/journal.pone.0053379.
3. Katz PS. Back away from the donuts! Today’s Hospitalist. https://www.todayshospitalist.com/back-away-from-the-donuts/. Published January 2013. Accessed June 18, 2018.
Our 24-hour society has made night shift work essential to people in many professions, including medical specialties. Working nights disrupts homeostatic and circadian rhythms, which leads to an accumulation of sleep debt (ie, the cumulative effect of not getting enough sleep).1 This debt can affect performance by impairing processing speed, concentration, mood, and physical health.1 Night shift work takes place during the period of the sleep-wake cycle that is programmed for sleep; after the shift, workers need to sleep during the period that is least conducive to sleep.1 Research indicates that a night shift worker’s sleep can be improved by scheduling light exposure and optimizing the timing of when they start their shifts.2 However, this may not be practical because night shifts usually are scheduled at particular intervals and cannot be tailored to the individual worker’s preference. Additionally, in the short term, full circadian adaptation to night shifts is impossible.1
Because sleep and performance are complex phenomena that are difficult to control, there is no single solution to maximizing these factors when one works nights.1 The most effective approach to combating the effects of night shift work is individualized and multimodal.1 However, whether you are working a night shift or are caring for a patient who does, the following nonpharmacologic strategies can help improve sleep and performance until the body naturally adapts to working this type of schedule1,3:
Minimize sleep debt before starting aseries of night shifts by not setting an alarm on the morning before the first night shift and by napping in the afternoon for approximately 45 minutes.
Take a nap during a work break (if work demands allow you to do so). However, nap for <30 minutes to avoid slow-wave sleep and subsequent grogginess when awakening.
Expose yourself to bright light immediately upon waking and for 15 minutes 2 or 3 times during a shift to promote alertness.
Drink caffeinated beverages before and during the shift to help improve concentration and reasoning (if there is no medical contraindication to consuming caffeine). However, avoid caffeine for at least 3 hours prior to going to sleep.
Add additional checks to critical tasks, such as ordering medications, during the shift, especially during the physiological nadir in the early hours of the morning.
Continue to: Create a cool, dark, quiet environment for sleep...
Create a cool, dark, quiet environment for sleep using a comfortable mattress and pillow, blackout blinds, ear plugs, and a noise machine. Also, avoid using your smartphone or tablet while trying to go to sleep. Minimize exposure to bright light on the drive home, and stick to a routine (eg, for meals and exercise).
Avoid working too many consecutive night shifts (if possible) because this can increase sleep deprivation. Also, limiting the number of night shifts and scheduling days off can speed recovery from sleep deprivation.
Our 24-hour society has made night shift work essential to people in many professions, including medical specialties. Working nights disrupts homeostatic and circadian rhythms, which leads to an accumulation of sleep debt (ie, the cumulative effect of not getting enough sleep).1 This debt can affect performance by impairing processing speed, concentration, mood, and physical health.1 Night shift work takes place during the period of the sleep-wake cycle that is programmed for sleep; after the shift, workers need to sleep during the period that is least conducive to sleep.1 Research indicates that a night shift worker’s sleep can be improved by scheduling light exposure and optimizing the timing of when they start their shifts.2 However, this may not be practical because night shifts usually are scheduled at particular intervals and cannot be tailored to the individual worker’s preference. Additionally, in the short term, full circadian adaptation to night shifts is impossible.1
Because sleep and performance are complex phenomena that are difficult to control, there is no single solution to maximizing these factors when one works nights.1 The most effective approach to combating the effects of night shift work is individualized and multimodal.1 However, whether you are working a night shift or are caring for a patient who does, the following nonpharmacologic strategies can help improve sleep and performance until the body naturally adapts to working this type of schedule1,3:
Minimize sleep debt before starting aseries of night shifts by not setting an alarm on the morning before the first night shift and by napping in the afternoon for approximately 45 minutes.
Take a nap during a work break (if work demands allow you to do so). However, nap for <30 minutes to avoid slow-wave sleep and subsequent grogginess when awakening.
Expose yourself to bright light immediately upon waking and for 15 minutes 2 or 3 times during a shift to promote alertness.
Drink caffeinated beverages before and during the shift to help improve concentration and reasoning (if there is no medical contraindication to consuming caffeine). However, avoid caffeine for at least 3 hours prior to going to sleep.
Add additional checks to critical tasks, such as ordering medications, during the shift, especially during the physiological nadir in the early hours of the morning.
Continue to: Create a cool, dark, quiet environment for sleep...
Create a cool, dark, quiet environment for sleep using a comfortable mattress and pillow, blackout blinds, ear plugs, and a noise machine. Also, avoid using your smartphone or tablet while trying to go to sleep. Minimize exposure to bright light on the drive home, and stick to a routine (eg, for meals and exercise).
Avoid working too many consecutive night shifts (if possible) because this can increase sleep deprivation. Also, limiting the number of night shifts and scheduling days off can speed recovery from sleep deprivation.
1. McKenna H, Wilkes M. Optimising sleep for night shifts. BMJ. 2018;360:j5637. doi: 10.1136/bmj.5637.
2. Postnova S, Robinson PA, Postnov DD. Adaptation to shift work: physiologically based modeling of the effects of lighting and shifts’ start time. PLoS One. 2013;8(1):e53379. doi: 10.1371/journal.pone.0053379.
3. Katz PS. Back away from the donuts! Today’s Hospitalist. https://www.todayshospitalist.com/back-away-from-the-donuts/. Published January 2013. Accessed June 18, 2018.
1. McKenna H, Wilkes M. Optimising sleep for night shifts. BMJ. 2018;360:j5637. doi: 10.1136/bmj.5637.
2. Postnova S, Robinson PA, Postnov DD. Adaptation to shift work: physiologically based modeling of the effects of lighting and shifts’ start time. PLoS One. 2013;8(1):e53379. doi: 10.1371/journal.pone.0053379.
3. Katz PS. Back away from the donuts! Today’s Hospitalist. https://www.todayshospitalist.com/back-away-from-the-donuts/. Published January 2013. Accessed June 18, 2018.
Untreated OSA linked to resistant hypertension in black patients
according to findings published in Circulation.
In an analysis of 664 patients with hypertension, those with moderate to severe OSA had twofold higher odds of resistant hypertension, compared with those with no or mild OSA (odds ratio, 2.04; 95% confidence interval, 1.14-3.67), reported Dayna A. Johnson, PhD, of the Division of Sleep and Circadian Disorders at Brigham and Women’s Hospital, Boston, and coauthors.
Participants were enrolled in the JHSS, an ancillary trial conducted during December 2012 – May 2016 as part of the Jackson Heart Study, a longitudinal study of 5,306 black adults aged 21-95 years in Jackson, Miss. Patients included in the analysis had hypertension (defined as high blood pressure, use of antihypertensive medication, or self-reported diagnosis). Those without a valid in-home sleep apnea test and with missing data on hypertension, measured blood pressure, or use of antihypertensive medications and diuretics were excluded from analysis.
Sleep apnea was assessed using measures of nasal pressure, thoracic and abdominal inductance plethysmography, finger pulse oximetry, body position, and electrocardiography with a validated Type 3 home sleep apnea device. Obstructive apneas were identified as a flat or nearly flat amplitude of the nasal pressure signal for greater than 10 seconds, accompanied by respiratory effort on the abdominal or thoracic inductance plethysmography bands. Severity was defined by the standard Respiratory Event Index (REI) categories: fewer than 5 events (unaffected), greater than or equal to 5 events to fewer than 15 events (mild), greater than or equal to 15 events to fewer than 30 events (moderate), and greater than or equal to 30 events (severe), the authors reported.
High blood pressure (BP) was defined as systolic BP greater than or equal to 130 mm Hg or diastolic BP greater than or equal to 80 mm Hg. Controlled hypertension was defined as systolic BP less than 130 mmHg and diastolic BP less than 80 mm Hg.
Uncontrolled BP was defined as high BP with use of one or two classes of antihypertensive medications; resistant hypertension was defined as having high BP while on greater than or equal to three classes of antihypertensive medications with one being a diuretic or as using of greater than four classes of antihypertensive medications regardless of BP control, Dr. Johnson and colleagues reported.
A total of 25.7% of hypertension patients had moderate or severe OSA, though only 6% of these patients had an OSA diagnosis from a physician. In addition, 48.2% of patients had uncontrolled hypertension, and 14.5% had resistant hypertension.
Moderate or severe OSA was associated with nearly twofold higher unadjusted odds of resistant hypertension (OR, 1.92; 95% CI, 1.15-3.20). In adjusted models, moderate or severe OSA and nocturnal hypoxemia were not associated with uncontrolled hypertension but were associated with resistant hypertension (OR, 2.04; 95% CI, 1.14-3.67; OR, 1.25; 95% CI, 1.01-1.55, respectively).
Compared with no OSA, severe OSA was associated with more than three times higher odds of resistant hypertension (OR, 3.50; 95% CI, 1.54-7.91). This association was even higher after adjustment for covariates (OR, 3.58; 95% CI, 1.39-9.19).
“These data suggest that untreated OSA may contribute to the high burden of resistant hypertension in blacks,” Dr. Johnson and coauthors wrote. “Future studies should test whether diagnosis and treatment of OSA may be interventions for improving BP control” and reducing this burden, they added.
“These findings are particularly important given that most adults with OSA are undiagnosed and untreated.”
The study was funded by grants from the National Heart, Lung, and Blood Institute. One of the authors reported receiving funding from Amgen. No other disclosures were reported.
SOURCE: Johnson D et al. Circulation. 2018. doi: 10.1161/CIRCULATIONAHA.118.036675.
according to findings published in Circulation.
In an analysis of 664 patients with hypertension, those with moderate to severe OSA had twofold higher odds of resistant hypertension, compared with those with no or mild OSA (odds ratio, 2.04; 95% confidence interval, 1.14-3.67), reported Dayna A. Johnson, PhD, of the Division of Sleep and Circadian Disorders at Brigham and Women’s Hospital, Boston, and coauthors.
Participants were enrolled in the JHSS, an ancillary trial conducted during December 2012 – May 2016 as part of the Jackson Heart Study, a longitudinal study of 5,306 black adults aged 21-95 years in Jackson, Miss. Patients included in the analysis had hypertension (defined as high blood pressure, use of antihypertensive medication, or self-reported diagnosis). Those without a valid in-home sleep apnea test and with missing data on hypertension, measured blood pressure, or use of antihypertensive medications and diuretics were excluded from analysis.
Sleep apnea was assessed using measures of nasal pressure, thoracic and abdominal inductance plethysmography, finger pulse oximetry, body position, and electrocardiography with a validated Type 3 home sleep apnea device. Obstructive apneas were identified as a flat or nearly flat amplitude of the nasal pressure signal for greater than 10 seconds, accompanied by respiratory effort on the abdominal or thoracic inductance plethysmography bands. Severity was defined by the standard Respiratory Event Index (REI) categories: fewer than 5 events (unaffected), greater than or equal to 5 events to fewer than 15 events (mild), greater than or equal to 15 events to fewer than 30 events (moderate), and greater than or equal to 30 events (severe), the authors reported.
High blood pressure (BP) was defined as systolic BP greater than or equal to 130 mm Hg or diastolic BP greater than or equal to 80 mm Hg. Controlled hypertension was defined as systolic BP less than 130 mmHg and diastolic BP less than 80 mm Hg.
Uncontrolled BP was defined as high BP with use of one or two classes of antihypertensive medications; resistant hypertension was defined as having high BP while on greater than or equal to three classes of antihypertensive medications with one being a diuretic or as using of greater than four classes of antihypertensive medications regardless of BP control, Dr. Johnson and colleagues reported.
A total of 25.7% of hypertension patients had moderate or severe OSA, though only 6% of these patients had an OSA diagnosis from a physician. In addition, 48.2% of patients had uncontrolled hypertension, and 14.5% had resistant hypertension.
Moderate or severe OSA was associated with nearly twofold higher unadjusted odds of resistant hypertension (OR, 1.92; 95% CI, 1.15-3.20). In adjusted models, moderate or severe OSA and nocturnal hypoxemia were not associated with uncontrolled hypertension but were associated with resistant hypertension (OR, 2.04; 95% CI, 1.14-3.67; OR, 1.25; 95% CI, 1.01-1.55, respectively).
Compared with no OSA, severe OSA was associated with more than three times higher odds of resistant hypertension (OR, 3.50; 95% CI, 1.54-7.91). This association was even higher after adjustment for covariates (OR, 3.58; 95% CI, 1.39-9.19).
“These data suggest that untreated OSA may contribute to the high burden of resistant hypertension in blacks,” Dr. Johnson and coauthors wrote. “Future studies should test whether diagnosis and treatment of OSA may be interventions for improving BP control” and reducing this burden, they added.
“These findings are particularly important given that most adults with OSA are undiagnosed and untreated.”
The study was funded by grants from the National Heart, Lung, and Blood Institute. One of the authors reported receiving funding from Amgen. No other disclosures were reported.
SOURCE: Johnson D et al. Circulation. 2018. doi: 10.1161/CIRCULATIONAHA.118.036675.
according to findings published in Circulation.
In an analysis of 664 patients with hypertension, those with moderate to severe OSA had twofold higher odds of resistant hypertension, compared with those with no or mild OSA (odds ratio, 2.04; 95% confidence interval, 1.14-3.67), reported Dayna A. Johnson, PhD, of the Division of Sleep and Circadian Disorders at Brigham and Women’s Hospital, Boston, and coauthors.
Participants were enrolled in the JHSS, an ancillary trial conducted during December 2012 – May 2016 as part of the Jackson Heart Study, a longitudinal study of 5,306 black adults aged 21-95 years in Jackson, Miss. Patients included in the analysis had hypertension (defined as high blood pressure, use of antihypertensive medication, or self-reported diagnosis). Those without a valid in-home sleep apnea test and with missing data on hypertension, measured blood pressure, or use of antihypertensive medications and diuretics were excluded from analysis.
Sleep apnea was assessed using measures of nasal pressure, thoracic and abdominal inductance plethysmography, finger pulse oximetry, body position, and electrocardiography with a validated Type 3 home sleep apnea device. Obstructive apneas were identified as a flat or nearly flat amplitude of the nasal pressure signal for greater than 10 seconds, accompanied by respiratory effort on the abdominal or thoracic inductance plethysmography bands. Severity was defined by the standard Respiratory Event Index (REI) categories: fewer than 5 events (unaffected), greater than or equal to 5 events to fewer than 15 events (mild), greater than or equal to 15 events to fewer than 30 events (moderate), and greater than or equal to 30 events (severe), the authors reported.
High blood pressure (BP) was defined as systolic BP greater than or equal to 130 mm Hg or diastolic BP greater than or equal to 80 mm Hg. Controlled hypertension was defined as systolic BP less than 130 mmHg and diastolic BP less than 80 mm Hg.
Uncontrolled BP was defined as high BP with use of one or two classes of antihypertensive medications; resistant hypertension was defined as having high BP while on greater than or equal to three classes of antihypertensive medications with one being a diuretic or as using of greater than four classes of antihypertensive medications regardless of BP control, Dr. Johnson and colleagues reported.
A total of 25.7% of hypertension patients had moderate or severe OSA, though only 6% of these patients had an OSA diagnosis from a physician. In addition, 48.2% of patients had uncontrolled hypertension, and 14.5% had resistant hypertension.
Moderate or severe OSA was associated with nearly twofold higher unadjusted odds of resistant hypertension (OR, 1.92; 95% CI, 1.15-3.20). In adjusted models, moderate or severe OSA and nocturnal hypoxemia were not associated with uncontrolled hypertension but were associated with resistant hypertension (OR, 2.04; 95% CI, 1.14-3.67; OR, 1.25; 95% CI, 1.01-1.55, respectively).
Compared with no OSA, severe OSA was associated with more than three times higher odds of resistant hypertension (OR, 3.50; 95% CI, 1.54-7.91). This association was even higher after adjustment for covariates (OR, 3.58; 95% CI, 1.39-9.19).
“These data suggest that untreated OSA may contribute to the high burden of resistant hypertension in blacks,” Dr. Johnson and coauthors wrote. “Future studies should test whether diagnosis and treatment of OSA may be interventions for improving BP control” and reducing this burden, they added.
“These findings are particularly important given that most adults with OSA are undiagnosed and untreated.”
The study was funded by grants from the National Heart, Lung, and Blood Institute. One of the authors reported receiving funding from Amgen. No other disclosures were reported.
SOURCE: Johnson D et al. Circulation. 2018. doi: 10.1161/CIRCULATIONAHA.118.036675.
FROM CIRCULATION
Key clinical point: Untreated moderate or severe obstructive sleep apnea was associated with greater odds of resistant hypertension.
Major finding: In patients with hypertension, those with moderate to severe OSA had twofold higher odds of resistant hypertension, compared with those with no or mild OSA.
Study details: A total of 664 participants were enrolled in the JHSS, an ancillary trial as part of the Jackson Heart Study, a longitudinal study of 5,306 black adults.
Disclosures: The study was funded by grants from the National Heart, Lung, and Blood Institute. One of the authors reported receiving funding from Amgen. No other disclosures were reported.
Source: Johnson D et al. Circulation. 2018. doi: 10.1161/CIRCULATIONAHA.118.036675.
More data link severe sleep apnea and aggressive melanoma
in a study of 443 adults.
Sleep-disordered breathing (SDB) has been associated with increased cancer risk and mortality, but no large studies have examined the association in specific cancers, wrote Miguel Angel Martinez-Garcia, MD, of La Fe University and Polytechnic Hospital, Valencia, Spain, and his colleagues.
The researchers conducted a sleep study of 443 adults with melanoma within 6 months of their diagnoses. The findings were published in the journal CHEST®. Overall, patients with more severe sleep apnea were nearly twice as likely to have aggressive type melanoma, compared with those with less severe sleep apnea.
Sleep apnea was defined via the Apnea Hypopnea Index (AHI) and the desaturation indices (DI). Aggressive melanoma was defined as a Breslow index greater than 1 mm, compared with 1 mm or less.
Patients with AHI greater than 15.6 events per hour or in the DI4% tertile (more than 9.3 desaturations per hour) were approximately twice as likely (1.94 and 1.93 times, respectively) to have a more aggressive melanoma as were those with less severe sleep apnea, after adjustment for age, gender, body mass index, and melanoma location.
The average age of the patients was 60 years, 51% were male, and the average time between the melanoma diagnosis and the sleep study was 82 days. Sleep symptoms were not significantly different between the patients with aggressive or less aggressive melanoma. However, in addition to more severe sleep apnea, those with aggressive melanoma were significantly more likely to be older, male, and have a higher BMI than were those with less aggressive disease.
“Among the most salient findings were the dependence of the association between SDB and the markers of melanoma aggressiveness on both age and the actual indicators of tumor aggressiveness,” the researchers noted. The association was significant in patients younger than 55 years only if their Breslow index was greater than 2 mm, the researchers said.
The study findings were limited by the absence of full overnight polysomnography to assess SDB because not all participating centers had access to that option, the researchers noted. However, the results support an independent link between sleep apnea and common measures of aggressive melanoma, especially in younger patients, they said. “Future prospective studies are needed to confirm whether the presence and treatment of SDB and its evolution over time are also associated with poor melanoma outcomes, including death, the pathophysiological mechanisms underlying this association,” they added.
The study was supported in part by Fondo de Investigation Sanitaria, SEPAR, Red Respira, and Sociedad Valenciana de Neumología. The researchers had no financial conflicts to disclose
SOURCE: Martinez-Garcia M et al. CHEST. 2018; doi: 10.1016/j.chest.2018.07.015.
in a study of 443 adults.
Sleep-disordered breathing (SDB) has been associated with increased cancer risk and mortality, but no large studies have examined the association in specific cancers, wrote Miguel Angel Martinez-Garcia, MD, of La Fe University and Polytechnic Hospital, Valencia, Spain, and his colleagues.
The researchers conducted a sleep study of 443 adults with melanoma within 6 months of their diagnoses. The findings were published in the journal CHEST®. Overall, patients with more severe sleep apnea were nearly twice as likely to have aggressive type melanoma, compared with those with less severe sleep apnea.
Sleep apnea was defined via the Apnea Hypopnea Index (AHI) and the desaturation indices (DI). Aggressive melanoma was defined as a Breslow index greater than 1 mm, compared with 1 mm or less.
Patients with AHI greater than 15.6 events per hour or in the DI4% tertile (more than 9.3 desaturations per hour) were approximately twice as likely (1.94 and 1.93 times, respectively) to have a more aggressive melanoma as were those with less severe sleep apnea, after adjustment for age, gender, body mass index, and melanoma location.
The average age of the patients was 60 years, 51% were male, and the average time between the melanoma diagnosis and the sleep study was 82 days. Sleep symptoms were not significantly different between the patients with aggressive or less aggressive melanoma. However, in addition to more severe sleep apnea, those with aggressive melanoma were significantly more likely to be older, male, and have a higher BMI than were those with less aggressive disease.
“Among the most salient findings were the dependence of the association between SDB and the markers of melanoma aggressiveness on both age and the actual indicators of tumor aggressiveness,” the researchers noted. The association was significant in patients younger than 55 years only if their Breslow index was greater than 2 mm, the researchers said.
The study findings were limited by the absence of full overnight polysomnography to assess SDB because not all participating centers had access to that option, the researchers noted. However, the results support an independent link between sleep apnea and common measures of aggressive melanoma, especially in younger patients, they said. “Future prospective studies are needed to confirm whether the presence and treatment of SDB and its evolution over time are also associated with poor melanoma outcomes, including death, the pathophysiological mechanisms underlying this association,” they added.
The study was supported in part by Fondo de Investigation Sanitaria, SEPAR, Red Respira, and Sociedad Valenciana de Neumología. The researchers had no financial conflicts to disclose
SOURCE: Martinez-Garcia M et al. CHEST. 2018; doi: 10.1016/j.chest.2018.07.015.
in a study of 443 adults.
Sleep-disordered breathing (SDB) has been associated with increased cancer risk and mortality, but no large studies have examined the association in specific cancers, wrote Miguel Angel Martinez-Garcia, MD, of La Fe University and Polytechnic Hospital, Valencia, Spain, and his colleagues.
The researchers conducted a sleep study of 443 adults with melanoma within 6 months of their diagnoses. The findings were published in the journal CHEST®. Overall, patients with more severe sleep apnea were nearly twice as likely to have aggressive type melanoma, compared with those with less severe sleep apnea.
Sleep apnea was defined via the Apnea Hypopnea Index (AHI) and the desaturation indices (DI). Aggressive melanoma was defined as a Breslow index greater than 1 mm, compared with 1 mm or less.
Patients with AHI greater than 15.6 events per hour or in the DI4% tertile (more than 9.3 desaturations per hour) were approximately twice as likely (1.94 and 1.93 times, respectively) to have a more aggressive melanoma as were those with less severe sleep apnea, after adjustment for age, gender, body mass index, and melanoma location.
The average age of the patients was 60 years, 51% were male, and the average time between the melanoma diagnosis and the sleep study was 82 days. Sleep symptoms were not significantly different between the patients with aggressive or less aggressive melanoma. However, in addition to more severe sleep apnea, those with aggressive melanoma were significantly more likely to be older, male, and have a higher BMI than were those with less aggressive disease.
“Among the most salient findings were the dependence of the association between SDB and the markers of melanoma aggressiveness on both age and the actual indicators of tumor aggressiveness,” the researchers noted. The association was significant in patients younger than 55 years only if their Breslow index was greater than 2 mm, the researchers said.
The study findings were limited by the absence of full overnight polysomnography to assess SDB because not all participating centers had access to that option, the researchers noted. However, the results support an independent link between sleep apnea and common measures of aggressive melanoma, especially in younger patients, they said. “Future prospective studies are needed to confirm whether the presence and treatment of SDB and its evolution over time are also associated with poor melanoma outcomes, including death, the pathophysiological mechanisms underlying this association,” they added.
The study was supported in part by Fondo de Investigation Sanitaria, SEPAR, Red Respira, and Sociedad Valenciana de Neumología. The researchers had no financial conflicts to disclose
SOURCE: Martinez-Garcia M et al. CHEST. 2018; doi: 10.1016/j.chest.2018.07.015.
FROM CHEST®
Key clinical point: Severe sleep disordered breathing is independently associated with more severe melanoma.
Major finding: Patients in the upper tertiles of AHI or DI4% were 1.94 and 1.93 times more likely, respectively, to have aggressive melanoma compared with patients with less severe sleep apnea.
Study details: The data come from an observational study of 443 adults diagnosed with melanoma.
Disclosures: The study was supported in part by Fondo de Investigation Sanitaria, SEPAR, Red Respira, and Sociedad Valenciana de Neumología. The researchers had no financial conflicts to disclose.
Source: Martinez-Garcia M et al. Chest. 2018. doi: 10.1016/j.chest.2018.07.015.
Should metabolic syndrome be renamed circadian syndrome?
LOS ANGELES – In the opinion of Paul Zimmet, MD, PhD, the
This scenario created the “perfect storm” for rising rates of metabolic syndrome, which is related to low HDL cholesterol levels, central obesity, hypertension, hyperglycemia, and high triglyceride levels, Dr. Zimmet said at the World Congress on Insulin Resistance, Diabetes & Cardiovascular Disease. These cardiometabolic risk factors “all seem to cluster together in relation to the changes in our society,” he said. “It’s on that basis and research findings that I think we should understand that most of them, if not all, have been demonstrated to relate to circadian rhythm disturbance.”
In fact, the associated comorbidities sleep apnea, depression, and fatty liver disease should be included in the metabolic syndrome cluster and should be renamed the “circadian syndrome,” according to Dr. Zimmet, professor of diabetes at Monash University, Melbourne.
The term metabolic syndrome is anathema, he said. “There have been numerous different definitions, which finally led to an effort to come up with a harmonized definition” by the International Diabetes Federation Task Force on Epidemiology and Prevention, with involvement from the American Heart Association (Circulation 2009;120[16]:1640-5).
In the early 1970s, Dr. Zimmet and his colleagues at Guys Hospital in London reported on diurnal variation in glucose tolerance. “If you did a glucose tolerance test in the afternoon it could be diabetic, whereas in the morning it was normal,” he noted. “Other researchers reported similar findings. That created in my own mind interest in this area of circadian rhythm. However, I had neglected this until recently, when I was doing background research while trying to find an answer to the elusive question of a central uniting explanation for the cardiometabolic cluster constituting the metabolic syndrome.” So decades later, Dr. Zimmet extended his research to include epigenetics in the quest. Described as the study of heritable changes in gene function that occur without a change in the sequence of the DNA, epigenetic changes “are closely linked to the circadian rhythm, otherwise known as ‘the body clock,’ ” said Dr. Zimmet, who also is codirector with Naftali Stern, MD, of the Sagol Center for Epigenetics of Metabolism and Aging at Tel Aviv Medical Center. “Many aspects of human behavior and metabolism are closely linked to the circadian clock and affected by its rhythm disturbance. We decided that we wanted to further investigate this area: To what extent is circadian rhythm the central feature to explain the clustering of all of these cardiovascular and metabolic risk factors of the metabolic syndrome.”
In recent years, he has been collaborating with Noga Kronfeld-Schor, PhD, of the department of zoology at Tel Aviv (Israel) University. The research focuses on a gerbil from the Negev: Psammomys obesus (otherwise known as the Israeli fat sand rat), which develops elevated blood sugar, obesity, depression, sleep disturbance, fatty liver, and circadian dysrhythmia when removed from the desert environment to the laboratory. “These are all key features of type 2 diabetes in humans,” he said. “This is probably the best animal model of type 2 diabetes, and we felt that it was worth looking more closely to see if there was a similar relationship in humans as to whether circadian dysrhythmia would be causing all or most of these features in humans including obesity.” An epigenetic study of the gerbil in the laboratory of Prof. Sam El-Osta at Monash has shown that parental diet during early life regulated expression of genes associated with DNA methylation in the key FTO gene associated with obesity (Int J Obesity 2016;40:1079-88). It suggests that diet-induced metabolic changes can be transmitted from parent to offspring by mechanisms under epigenetic control.
Published studies from other research groups support the link between other of the cardiometabolic metabolic syndrome characteristics, epigenetic modifications, and circadian dysrhythmia including cardiovascular regulation and disease (Eur Heart J 2018;39[14]:2326-9), sleep loss and alterations in DNA methylation (Science Advances 2018;4[8]:eaar8590), and circadian dysrhythmia and fatty liver (Cell Metab 2012;15[6]:848-60). “In 2009, the FDA approved bromocriptine mesylate, a drug which has effects on circadian rhythm, for treatment of type 2 diabetes, suggesting its use in diabetes may have some role through the alteration of circadian rhythm,” continued Dr. Zimmet, who also is honorary president of the International Diabetes Federation. “Depression is also clearly linked to circadian rhythm and there is evidence from research and human studies that light therapy may be an effective treatment for type 2 diabetes and depression.”
Dr. Zimmet ended his presentation with a strong call for adding sleep apnea, fatty liver, and depression to the existing features of the metabolic syndrome “to encourage clinicians and researchers look at the picture of cardiometabolic risk much more broadly than as just a group of metabolic abnormalities,” he said. “We propose that these comorbidities be embraced within the definition of the cardiometabolic cluster and be renamed the ‘circadian syndrome.’ This cluster is now the main driver of the global chronic disease epidemic and its health burden. This is a disease of civilization – the result of the way we live.”
Dr. Zimmet reported having no disclosures.
LOS ANGELES – In the opinion of Paul Zimmet, MD, PhD, the
This scenario created the “perfect storm” for rising rates of metabolic syndrome, which is related to low HDL cholesterol levels, central obesity, hypertension, hyperglycemia, and high triglyceride levels, Dr. Zimmet said at the World Congress on Insulin Resistance, Diabetes & Cardiovascular Disease. These cardiometabolic risk factors “all seem to cluster together in relation to the changes in our society,” he said. “It’s on that basis and research findings that I think we should understand that most of them, if not all, have been demonstrated to relate to circadian rhythm disturbance.”
In fact, the associated comorbidities sleep apnea, depression, and fatty liver disease should be included in the metabolic syndrome cluster and should be renamed the “circadian syndrome,” according to Dr. Zimmet, professor of diabetes at Monash University, Melbourne.
The term metabolic syndrome is anathema, he said. “There have been numerous different definitions, which finally led to an effort to come up with a harmonized definition” by the International Diabetes Federation Task Force on Epidemiology and Prevention, with involvement from the American Heart Association (Circulation 2009;120[16]:1640-5).
In the early 1970s, Dr. Zimmet and his colleagues at Guys Hospital in London reported on diurnal variation in glucose tolerance. “If you did a glucose tolerance test in the afternoon it could be diabetic, whereas in the morning it was normal,” he noted. “Other researchers reported similar findings. That created in my own mind interest in this area of circadian rhythm. However, I had neglected this until recently, when I was doing background research while trying to find an answer to the elusive question of a central uniting explanation for the cardiometabolic cluster constituting the metabolic syndrome.” So decades later, Dr. Zimmet extended his research to include epigenetics in the quest. Described as the study of heritable changes in gene function that occur without a change in the sequence of the DNA, epigenetic changes “are closely linked to the circadian rhythm, otherwise known as ‘the body clock,’ ” said Dr. Zimmet, who also is codirector with Naftali Stern, MD, of the Sagol Center for Epigenetics of Metabolism and Aging at Tel Aviv Medical Center. “Many aspects of human behavior and metabolism are closely linked to the circadian clock and affected by its rhythm disturbance. We decided that we wanted to further investigate this area: To what extent is circadian rhythm the central feature to explain the clustering of all of these cardiovascular and metabolic risk factors of the metabolic syndrome.”
In recent years, he has been collaborating with Noga Kronfeld-Schor, PhD, of the department of zoology at Tel Aviv (Israel) University. The research focuses on a gerbil from the Negev: Psammomys obesus (otherwise known as the Israeli fat sand rat), which develops elevated blood sugar, obesity, depression, sleep disturbance, fatty liver, and circadian dysrhythmia when removed from the desert environment to the laboratory. “These are all key features of type 2 diabetes in humans,” he said. “This is probably the best animal model of type 2 diabetes, and we felt that it was worth looking more closely to see if there was a similar relationship in humans as to whether circadian dysrhythmia would be causing all or most of these features in humans including obesity.” An epigenetic study of the gerbil in the laboratory of Prof. Sam El-Osta at Monash has shown that parental diet during early life regulated expression of genes associated with DNA methylation in the key FTO gene associated with obesity (Int J Obesity 2016;40:1079-88). It suggests that diet-induced metabolic changes can be transmitted from parent to offspring by mechanisms under epigenetic control.
Published studies from other research groups support the link between other of the cardiometabolic metabolic syndrome characteristics, epigenetic modifications, and circadian dysrhythmia including cardiovascular regulation and disease (Eur Heart J 2018;39[14]:2326-9), sleep loss and alterations in DNA methylation (Science Advances 2018;4[8]:eaar8590), and circadian dysrhythmia and fatty liver (Cell Metab 2012;15[6]:848-60). “In 2009, the FDA approved bromocriptine mesylate, a drug which has effects on circadian rhythm, for treatment of type 2 diabetes, suggesting its use in diabetes may have some role through the alteration of circadian rhythm,” continued Dr. Zimmet, who also is honorary president of the International Diabetes Federation. “Depression is also clearly linked to circadian rhythm and there is evidence from research and human studies that light therapy may be an effective treatment for type 2 diabetes and depression.”
Dr. Zimmet ended his presentation with a strong call for adding sleep apnea, fatty liver, and depression to the existing features of the metabolic syndrome “to encourage clinicians and researchers look at the picture of cardiometabolic risk much more broadly than as just a group of metabolic abnormalities,” he said. “We propose that these comorbidities be embraced within the definition of the cardiometabolic cluster and be renamed the ‘circadian syndrome.’ This cluster is now the main driver of the global chronic disease epidemic and its health burden. This is a disease of civilization – the result of the way we live.”
Dr. Zimmet reported having no disclosures.
LOS ANGELES – In the opinion of Paul Zimmet, MD, PhD, the
This scenario created the “perfect storm” for rising rates of metabolic syndrome, which is related to low HDL cholesterol levels, central obesity, hypertension, hyperglycemia, and high triglyceride levels, Dr. Zimmet said at the World Congress on Insulin Resistance, Diabetes & Cardiovascular Disease. These cardiometabolic risk factors “all seem to cluster together in relation to the changes in our society,” he said. “It’s on that basis and research findings that I think we should understand that most of them, if not all, have been demonstrated to relate to circadian rhythm disturbance.”
In fact, the associated comorbidities sleep apnea, depression, and fatty liver disease should be included in the metabolic syndrome cluster and should be renamed the “circadian syndrome,” according to Dr. Zimmet, professor of diabetes at Monash University, Melbourne.
The term metabolic syndrome is anathema, he said. “There have been numerous different definitions, which finally led to an effort to come up with a harmonized definition” by the International Diabetes Federation Task Force on Epidemiology and Prevention, with involvement from the American Heart Association (Circulation 2009;120[16]:1640-5).
In the early 1970s, Dr. Zimmet and his colleagues at Guys Hospital in London reported on diurnal variation in glucose tolerance. “If you did a glucose tolerance test in the afternoon it could be diabetic, whereas in the morning it was normal,” he noted. “Other researchers reported similar findings. That created in my own mind interest in this area of circadian rhythm. However, I had neglected this until recently, when I was doing background research while trying to find an answer to the elusive question of a central uniting explanation for the cardiometabolic cluster constituting the metabolic syndrome.” So decades later, Dr. Zimmet extended his research to include epigenetics in the quest. Described as the study of heritable changes in gene function that occur without a change in the sequence of the DNA, epigenetic changes “are closely linked to the circadian rhythm, otherwise known as ‘the body clock,’ ” said Dr. Zimmet, who also is codirector with Naftali Stern, MD, of the Sagol Center for Epigenetics of Metabolism and Aging at Tel Aviv Medical Center. “Many aspects of human behavior and metabolism are closely linked to the circadian clock and affected by its rhythm disturbance. We decided that we wanted to further investigate this area: To what extent is circadian rhythm the central feature to explain the clustering of all of these cardiovascular and metabolic risk factors of the metabolic syndrome.”
In recent years, he has been collaborating with Noga Kronfeld-Schor, PhD, of the department of zoology at Tel Aviv (Israel) University. The research focuses on a gerbil from the Negev: Psammomys obesus (otherwise known as the Israeli fat sand rat), which develops elevated blood sugar, obesity, depression, sleep disturbance, fatty liver, and circadian dysrhythmia when removed from the desert environment to the laboratory. “These are all key features of type 2 diabetes in humans,” he said. “This is probably the best animal model of type 2 diabetes, and we felt that it was worth looking more closely to see if there was a similar relationship in humans as to whether circadian dysrhythmia would be causing all or most of these features in humans including obesity.” An epigenetic study of the gerbil in the laboratory of Prof. Sam El-Osta at Monash has shown that parental diet during early life regulated expression of genes associated with DNA methylation in the key FTO gene associated with obesity (Int J Obesity 2016;40:1079-88). It suggests that diet-induced metabolic changes can be transmitted from parent to offspring by mechanisms under epigenetic control.
Published studies from other research groups support the link between other of the cardiometabolic metabolic syndrome characteristics, epigenetic modifications, and circadian dysrhythmia including cardiovascular regulation and disease (Eur Heart J 2018;39[14]:2326-9), sleep loss and alterations in DNA methylation (Science Advances 2018;4[8]:eaar8590), and circadian dysrhythmia and fatty liver (Cell Metab 2012;15[6]:848-60). “In 2009, the FDA approved bromocriptine mesylate, a drug which has effects on circadian rhythm, for treatment of type 2 diabetes, suggesting its use in diabetes may have some role through the alteration of circadian rhythm,” continued Dr. Zimmet, who also is honorary president of the International Diabetes Federation. “Depression is also clearly linked to circadian rhythm and there is evidence from research and human studies that light therapy may be an effective treatment for type 2 diabetes and depression.”
Dr. Zimmet ended his presentation with a strong call for adding sleep apnea, fatty liver, and depression to the existing features of the metabolic syndrome “to encourage clinicians and researchers look at the picture of cardiometabolic risk much more broadly than as just a group of metabolic abnormalities,” he said. “We propose that these comorbidities be embraced within the definition of the cardiometabolic cluster and be renamed the ‘circadian syndrome.’ This cluster is now the main driver of the global chronic disease epidemic and its health burden. This is a disease of civilization – the result of the way we live.”
Dr. Zimmet reported having no disclosures.
EXPERT ANALYSIS FROM THE WCIRDC 2018
Comorbid TBI & PTSD raise the risk for sleep disturbances, pain
Veterans living with comorbid traumatic brain injury (TBI) and posttraumatic stress disorder were at increased risk for worse pain and sleep disturbances, reported Nadir M. Balba and colleagues at the VA Portland (Ore.) Health Care System.
The authors conducted a retrospective review of medical records at the VA Portland Health Care System (VAPORHCS) that evaluated 639 veterans who were referred to the VAPORHCS Sleep Disorders Clinic between May 2015 and November 2016. They wrote, “The purpose of this study was to determine whether Veterans with comorbid TBI and PTSD exhibit a higher prevalence of sleep disturbances (determined via self-report and objective polysomnography) and pain compared to Veterans with only TBI or PTSD.”
Patients were recruited to participate in the cross-sectional study, which included participation in an overnight sleep clinic as well as patient self-reported sleep quality, pain, and TBI and PTSD symptom severity. Sleep disturbances included insomnia, nightmares, sleep fragmentation, obstructive sleep apnea, and parasomnias. The survey tools used in the study included the Rivermead Post Concussion Questionnaire (RPCQ), the PTSD Checklist DSM-5 (PSTD-5), the Insomnia Severity Index (ISI), and the Functional Outcomes of Sleep Questionnaire-10 (FOSQ-10). Sleep studies were recorded using Polysmith version 9.0 and sleep staging was performed by a certified sleep technician and verified by a board-certified sleep medicine physician.
Patients were grouped into one of four trauma exposure classifications based on their prior history of trauma, including neither (n = 383), TBI (n = 67), PTSD (n = 126), and TBI+PTSD (n = 63).
Self-reported sleep disturbance, which was the worst among those with PTSD and those with comorbid TBI and PTSD, indicated that PTSD plays a more significant role in the occurrence of disturbed sleep than TBI, the researchers noted. “Participants in the TBI+PTSD and PTSD groups had significantly worse ISI scores (i.e., higher scores) compared to both the TBI and neither groups (P less than .001). Furthermore, participants in the TBI+PTSD and PTSD groups had significantly worse FOSQ-10 scores (ie, lower scores) compared to both the TBI and neither groups (P less than .001),” they wrote.
In terms of pain, patients with comorbid TBI and PTSD reported the greatest severity of pain, including more frequent headaches and worse photo and phono sensitivities. The TBI and PTSD groups, however, both scored significantly higher in their pain reports than those in the neither group, which suggests “that each of these conditions independently contributes to increased pain,” the authors observed. Ultimately, they cited multiple linear regression models, which attributed sleep disturbances and TBI symptom severity as the primary contributors to pain presentation.
“It is well established that sleep disturbances and pain are inextricably linked,” they said. The results of this study serve to validate that connection “but also suggest this link may be even stronger in those with comorbid TBI and PTSD,” they added.
The researchers cited self-report data as a possible study limitation. They also conceded that comorbid depression and substance use disorder could both play a role in further exacerbating sleep disturbance and pain.
Future research should evaluate how TBI and PTSD, along with other unidentified comorbid conditions, may work together in exacerbating symptoms so that more effective treatment interventions can be developed to address sleep and pain disturbance following multiple traumas.
The authors had no relevant financial disclosures to report.
SOURCE: Balba N et al. J Clin Sleep Med. 2018;14(11):1865-78.
Veterans living with comorbid traumatic brain injury (TBI) and posttraumatic stress disorder were at increased risk for worse pain and sleep disturbances, reported Nadir M. Balba and colleagues at the VA Portland (Ore.) Health Care System.
The authors conducted a retrospective review of medical records at the VA Portland Health Care System (VAPORHCS) that evaluated 639 veterans who were referred to the VAPORHCS Sleep Disorders Clinic between May 2015 and November 2016. They wrote, “The purpose of this study was to determine whether Veterans with comorbid TBI and PTSD exhibit a higher prevalence of sleep disturbances (determined via self-report and objective polysomnography) and pain compared to Veterans with only TBI or PTSD.”
Patients were recruited to participate in the cross-sectional study, which included participation in an overnight sleep clinic as well as patient self-reported sleep quality, pain, and TBI and PTSD symptom severity. Sleep disturbances included insomnia, nightmares, sleep fragmentation, obstructive sleep apnea, and parasomnias. The survey tools used in the study included the Rivermead Post Concussion Questionnaire (RPCQ), the PTSD Checklist DSM-5 (PSTD-5), the Insomnia Severity Index (ISI), and the Functional Outcomes of Sleep Questionnaire-10 (FOSQ-10). Sleep studies were recorded using Polysmith version 9.0 and sleep staging was performed by a certified sleep technician and verified by a board-certified sleep medicine physician.
Patients were grouped into one of four trauma exposure classifications based on their prior history of trauma, including neither (n = 383), TBI (n = 67), PTSD (n = 126), and TBI+PTSD (n = 63).
Self-reported sleep disturbance, which was the worst among those with PTSD and those with comorbid TBI and PTSD, indicated that PTSD plays a more significant role in the occurrence of disturbed sleep than TBI, the researchers noted. “Participants in the TBI+PTSD and PTSD groups had significantly worse ISI scores (i.e., higher scores) compared to both the TBI and neither groups (P less than .001). Furthermore, participants in the TBI+PTSD and PTSD groups had significantly worse FOSQ-10 scores (ie, lower scores) compared to both the TBI and neither groups (P less than .001),” they wrote.
In terms of pain, patients with comorbid TBI and PTSD reported the greatest severity of pain, including more frequent headaches and worse photo and phono sensitivities. The TBI and PTSD groups, however, both scored significantly higher in their pain reports than those in the neither group, which suggests “that each of these conditions independently contributes to increased pain,” the authors observed. Ultimately, they cited multiple linear regression models, which attributed sleep disturbances and TBI symptom severity as the primary contributors to pain presentation.
“It is well established that sleep disturbances and pain are inextricably linked,” they said. The results of this study serve to validate that connection “but also suggest this link may be even stronger in those with comorbid TBI and PTSD,” they added.
The researchers cited self-report data as a possible study limitation. They also conceded that comorbid depression and substance use disorder could both play a role in further exacerbating sleep disturbance and pain.
Future research should evaluate how TBI and PTSD, along with other unidentified comorbid conditions, may work together in exacerbating symptoms so that more effective treatment interventions can be developed to address sleep and pain disturbance following multiple traumas.
The authors had no relevant financial disclosures to report.
SOURCE: Balba N et al. J Clin Sleep Med. 2018;14(11):1865-78.
Veterans living with comorbid traumatic brain injury (TBI) and posttraumatic stress disorder were at increased risk for worse pain and sleep disturbances, reported Nadir M. Balba and colleagues at the VA Portland (Ore.) Health Care System.
The authors conducted a retrospective review of medical records at the VA Portland Health Care System (VAPORHCS) that evaluated 639 veterans who were referred to the VAPORHCS Sleep Disorders Clinic between May 2015 and November 2016. They wrote, “The purpose of this study was to determine whether Veterans with comorbid TBI and PTSD exhibit a higher prevalence of sleep disturbances (determined via self-report and objective polysomnography) and pain compared to Veterans with only TBI or PTSD.”
Patients were recruited to participate in the cross-sectional study, which included participation in an overnight sleep clinic as well as patient self-reported sleep quality, pain, and TBI and PTSD symptom severity. Sleep disturbances included insomnia, nightmares, sleep fragmentation, obstructive sleep apnea, and parasomnias. The survey tools used in the study included the Rivermead Post Concussion Questionnaire (RPCQ), the PTSD Checklist DSM-5 (PSTD-5), the Insomnia Severity Index (ISI), and the Functional Outcomes of Sleep Questionnaire-10 (FOSQ-10). Sleep studies were recorded using Polysmith version 9.0 and sleep staging was performed by a certified sleep technician and verified by a board-certified sleep medicine physician.
Patients were grouped into one of four trauma exposure classifications based on their prior history of trauma, including neither (n = 383), TBI (n = 67), PTSD (n = 126), and TBI+PTSD (n = 63).
Self-reported sleep disturbance, which was the worst among those with PTSD and those with comorbid TBI and PTSD, indicated that PTSD plays a more significant role in the occurrence of disturbed sleep than TBI, the researchers noted. “Participants in the TBI+PTSD and PTSD groups had significantly worse ISI scores (i.e., higher scores) compared to both the TBI and neither groups (P less than .001). Furthermore, participants in the TBI+PTSD and PTSD groups had significantly worse FOSQ-10 scores (ie, lower scores) compared to both the TBI and neither groups (P less than .001),” they wrote.
In terms of pain, patients with comorbid TBI and PTSD reported the greatest severity of pain, including more frequent headaches and worse photo and phono sensitivities. The TBI and PTSD groups, however, both scored significantly higher in their pain reports than those in the neither group, which suggests “that each of these conditions independently contributes to increased pain,” the authors observed. Ultimately, they cited multiple linear regression models, which attributed sleep disturbances and TBI symptom severity as the primary contributors to pain presentation.
“It is well established that sleep disturbances and pain are inextricably linked,” they said. The results of this study serve to validate that connection “but also suggest this link may be even stronger in those with comorbid TBI and PTSD,” they added.
The researchers cited self-report data as a possible study limitation. They also conceded that comorbid depression and substance use disorder could both play a role in further exacerbating sleep disturbance and pain.
Future research should evaluate how TBI and PTSD, along with other unidentified comorbid conditions, may work together in exacerbating symptoms so that more effective treatment interventions can be developed to address sleep and pain disturbance following multiple traumas.
The authors had no relevant financial disclosures to report.
SOURCE: Balba N et al. J Clin Sleep Med. 2018;14(11):1865-78.
FROM THE JOURNAL OF CLINICAL SLEEP MEDICINE
Key clinical point: The combination of traumatic brain injury and posttraumatic stress disorder is linked to a higher risk for sleep disturbances, pain, and light and noise sensitivities.
Major finding: Participants with comorbid TBI and PTSD and PTSD only had higher Insomnia Severity Index scores than those with TBI only or neither condition (P less than .001).
Study details: Retrospective medical record review.
Disclosures: The authors noted no relevant financial disclosures.
Source: Balba N et al. J Clin Sleep Med. 2018;14(11):1865-78.