User login
Teamwork guides cardio-rheumatology clinics that care for unique patient population
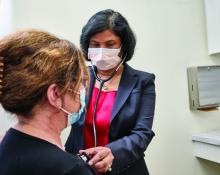
Clinical cardiologist Heba Wassif, MD, MPH, knows the value of working with her fellow rheumatologists, surgeons, and other clinicians to establish a care plan for her patients with cardiac conditions and autoimmune diseases.
She is the cofounder of the Cleveland Clinic’s new cardio-rheumatology program, which places an emphasis on multidisciplinary care. In her role, Dr. Wassif closely follows her patients, and if she sees any inflammation or any other condition that requires the rheumatologist, she reaches out to her colleagues to adjust medications if needed.
Collaboration with a rheumatologist was important when a patient with valvular disease was prepping for surgery. The patient was on significant immunosuppressants and the surgery had to be timed appropriately, accounting for any decreases in her immunosuppression, explained Dr. Wassif, director of inpatient clinical cardiology at Cleveland Clinic in Ohio.
Cardio-rheumatology programs are “the newest child” in a series of cardiology offshoots focusing on different populations. Cardio-oncology and cardio-obstetrics took off about 6 years ago, with cardio-rheumatology clinics and interested physicians rising in number over the last several years, Dr. Wassif noted.
The relationship between cardiovascular diseases and rheumatologic conditions is certainly recognized more often, “which means more literature is being published to discuss the link,” according to Rekha Mankad, MD, a trailblazer of this model of care. She directs the Women’s Heart Clinic at Mayo Clinic in Rochester, Minn., which was one of the earliest adopters of a cardio-rheumatology clinic.
Ten years ago, “nobody was talking about the link between rheumatologic conditions and cardiovascular disease,” Dr. Mankad said. “I’ve been asked to speak on this topic, and programs have asked me to speak about establishing cardio-rheumatology practices. So, there’s been an evolution as far as a recognition that these two conditions overlap.”
Patients have come to her independent of internal referrals, which means they have done Google searches on cardiology and rheumatology. “I think that it has made a splash, at least in the world of cardiology,” Dr. Mankad observed in an interview.
Other institutions such as NYU-Langone, Yale, Stanford, Brigham and Women’s Hospital in Boston, and Women’s College Hospital in Toronto have formed similar clinics whose focus is to address the specific needs of rheumatology patients with cardiac conditions through a teamwork approach.
Challenges of treating cardiac, rheumatologic conditions
The rise in clinics addresses the longstanding connection between autoimmune disorders and cardiac conditions.
Cardiologists have known that there is an element of inflammation that contributes to atherosclerosis, said Dr. Wassif, who has researched this topic extensively. A recent study she led found a strong association between rheumatic immune-mediated inflammatory diseases (IMIDs) and high risk of acute coronary syndrome in Medicare patients.
“This particular population has a very clear increased risk for cardiovascular conditions, including valve disease and heart failure,” she emphasized.
Patients with rheumatoid arthritis and lupus have up to a twofold and eightfold higher risk of heart disease, respectively, noted Michael S. Garshick, MD, a cardiovascular disease specialist who directs the cardio-rheumatology program at NYU-Langone Health, in New York. Cardiologists “have really developed an understanding that the immune system can impact the heart, and that there’s a need for people to understand the nuance behind how the immune system can affect them and what to do about it,” Dr. Garshick said.
Caring for patients with both afflictions comes with specific challenges. Many physicians are not well trained on managing and treating patients with these dual conditions.
The “lipid paradox,” in which lipids are reduced with active inflammation in some rheumatologic conditions, can make treatment more nuanced. In addition, the traditional ASCVD (atherosclerotic cardiovascular disease) score often underestimates the cardiovascular risk of these patients, noted cardiologist Margaret Furman, MD, MPH, assistant professor and codirector of Yale’s Cardio-Rheumatology Program, New Haven, Conn.
Newer biologic medications used to treat rheumatologic diseases can alter a patient’s lipid profile, she said in an interview.
“It can be difficult to assess each individual patient’s cardiovascular risk as their disease state and treatment can vary throughout their lifetime based on their degree of inflammation. The importance of aggressive lipid management is often underestimated,” Dr. Furman added.
Cardiology and rheumatology partnerships can address gaps in care of this unique group of patients, said Vaidehi R. Chowdhary, MBBS, MD, clinical chief of the Yale Section of Rheumatology, Allergy, and Immunology at Yale University.
“The role of the rheumatologist in this dyad is to educate patients on this risk, work toward adequate control of inflammation, and minimize use of medications that contribute to increased cardiovascular risks,” said Dr. Chowdhary, who cofounded Yale’s cardio-rheumatology program with Dr. Furman.
Cardiologists in turn can assert their knowledge about medications and their impact on lipids and inflammation, Dr. Wassif said.
Many anti-inflammatory therapies are now within the cardiologist’s purview, Dr. Garshick noted. “For example, specifically with pericarditis, there’s [Food and Drug Administration]–approved anti-inflammatories or biologics. We’re the ones who feel the most comfortable giving them right now.” Cardiologists quite often are consulted about medications that are efficacious in rheumatologic conditions but could negatively impact the cardiovascular system, such as Janus kinase inhibitors, he added.
‘Reading the tea leaves’
Each program has its own unique story. For the Cleveland Clinic, the concept of a cardio-rheumatology program began during the COVID-19 pandemic in 2020. Developing such a concept and gaining institutional acceptance is always a work in process, Dr. Wassif said. “It’s not that you decide one day that you’re going to build a center, and that center is going to come into fruition overnight. You first gauge interest within your division. Who are the individuals that are interested in this area?”
Cleveland Clinic’s center is seeking to build relations between medical disciplines while spotlighting the concept of cardio-rheumatology, said Dr. Wassif, who has been providing education within the clinic and at other health institutions to ensure that patients receive appropriate attention early.
NYU-Langone launched its program amid this heightened awareness that the immune system could affect atherosclerosis, “kind of reading of the tea leaves, so to speak,” Dr. Garshick said.
Several clinical trials served as a catalyst for this movement. “A lot of clinical cardiologists were never 100% convinced that targeting the immune system reduced cardiovascular disease,” he said. Then the CANTOS clinical trial came along and showed for the first time that a therapeutic monoclonal antibody targeting interleukin-1beta, a cytokine central to inflammatory response, could in fact reduce cardiovascular disease.
Trials like this, along with epidemiologic literature connecting the rheumatologic and the autoimmune conditions with cardiovascular disease, pushed this concept to the forefront, Dr. Garshick said.
The notion that a clinic could successfully address cardiac problems in patients with rheumatic diseases yielded promising returns at Women’s College Hospital in Toronto, according to a report presented at the 2018 American College of Rheumatology annual meeting. Researchers reported that patients with rheumatologic conditions who attended a cardio-rheumatology clinic at this center saw improvements in care. The clinic identified increased cardiovascular risk and early atherosclerosis, and 53.8% of patients altered their medications after being seen in the clinic.
A total of 39.7% and 32.1% received lipid lowering and antiplatelet therapies, respectively, and 14% received antihypertensive therapy. A small percentage were treated for heart failure or placed on lifelong anticoagulation therapy for atrial fibrillation, and one patient received a percutaneous coronary stent.
Ins and outs of the referral process
Initially designed for preventive cardiac risk assessment, Yale’s program evolved into a multidisciplinary, patient-centered approach for the management of complex cardiovascular conditions in patients with autoimmune rheumatologic diseases.
The program is open to anyone who carries a diagnosis of rheumatologic disease or has elevated inflammatory markers. “Every patient, regardless of the reason for the referral, receives a cardiovascular risk assessment,” Dr. Furman said.
Most referrals come from rheumatologists, although cardiology colleagues and pulmonologists have also sent referrals. A pulmonologist, for example, may want to rule out a cardiac cause to shortness of breath. The patient’s workup, care, and follow-up are based on the reason for referral.
“We are currently referring patients with established cardiac disease, traditional risk factors, or for better risk assessment for primary prevention of coronary artery disease,” Dr. Chowdhary said. “We communicate very frequently about medication changes, and patients are aware of goals of care from both sides.”
Dr. Furman works closely with several of the rheumatology specialists taking care of patients with rheumatoid arthritis, systemic lupus erythematosus, and scleroderma.
Rheumatology follows patients every 3-6 months or more frequently based on their disease activity.
Dr. Mankad uses her sleuthing skills at Mayo Clinic to determine what the patients need. If they come in for a preventive assessment, she looks more closely at their cardiovascular risks and may order additional imaging to look for subclinical atherosclerosis. “We’re more aggressive with statin therapy in this population because of that,” she said.
If it’s valve disease, she pays extra attention to the patients’ valves in the echocardiograms and follows them a bit more regularly than someone without a rheumatologic condition and valve disease.
For patients with heart failure signs or symptoms, “it depends on how symptomatic they are,” Dr. Mankad said. In some instances, she may look for evidence of heart failure with preserved ejection fraction in patients who have rheumatoid arthritis who happen to be short of breath. “There’s so many different manifestations that patients with rheumatologic conditions can have as far as what could be affected in the heart,” she noted.
Quite frequently, Dr. Mankad identifies subclinical disease in her patients with rheumatoid arthritis. “I’ve seen many patients whose risk scores would not dictate statin therapy. But I went looking for subclinical disease by either doing coronary assessment or carotid assessment and have found atherosclerosis that would be enough to warrant statin therapy.”
A personalized assessment to reduce cardiac risk
NYU-Langone’s program offers opportunities to educate patients about the link between cardiac and rheumatologic disease.
“Their rheumatologist or their dermatologist will say, ‘Hey, have you heard about the connection between psoriasis, psoriatic or rheumatoid arthritis, and heart disease and the risk of heart attack or stroke?’ ” Dr. Garshick said.
The patients will often say they know nothing about these connections and want to learn more about how to treat it.
“We’ll say, ‘we have someone here that can help you.’ They’ll send them to myself or other colleagues like me across the country. We’ll assess blood pressure, weight, lipids, hemoglobin A1c, and other serologic and oftentimes imaging biomarkers of cardiovascular risk.” The patients will receive a personalized assessment, listing things they can do to lower their risk, whether it’s diet, exercise, or lifestyle. “Many times it can involve medications to reduce heart disease risk,” said Dr. Garshick.
In some instances, a rheumatologist or dermatologist may be concerned about starting a patient on a specific medication for the disease such as a JAK inhibitor. “We’ll help assess their risk because there’s been a lot of literature out in the rheumatology world about the risk of JAK inhibitors and heart disease and blood clots,” said Dr. Garshick.
Dr. Garshick also sees patients with rheumatologic conditions who have a specific cardiovascular concern or complaint such as shortness of breath or chest pain. “We’ll work that up with a specific knowledge of the underlying immune condition and how that may impact their heart,” he said.
Advances in research
As they continue to see patients and devise specific care plans, developers of cardio-rheumatology programs have been supplementing their work with ongoing research.
Yale’s clinic is expanding this year to include a new attending physician, Attila Feher, MD, PhD, who has conducted research in autoimmunity and microcirculation using molecular imaging and multimodality imaging techniques. Prevalence of coronary microvascular dysfunction appears to be increased in this patient population, Dr. Furman said.
Dr. Wassif recently coauthored a paper that examined patients with underlying rheumatologic conditions who undergo valvular and aortic valve replacement. “To our surprise, there was really no difference between patients with autoimmune conditions and others with nonautoimmune conditions,” she said, adding that the study had its limitations.
Other work includes data on Medicare patients with ST- and non-ST-elevation myocardial infarctions who have an underlying autoimmune disorder. Dr. Wassif and her colleagues found that their long-term outcomes are worse than those of patients without these conditions. “It’s unclear if worse outcomes are related to complications of autoimmunity versus the extent of their underlying disease. This is a work in progress and certainly an area that is ripe for research.”
Dr. Garshick and other collaborators at NYU have been focusing on the endothelium, specifically platelet biology in patients with psoriasis, psoriatic arthritis, and lupus. “We’re about to start the same research with gout as well,” he said.
“The process we’re most interested in is understanding how these diseases impact the early stages of cholesterol. And the way we’re doing that is evaluating the vasculature, specifically the endothelium,” he said.
He has finished two clinical trials that evaluate how standard heart disease medications such as aspirin and statins impact or can potentially benefit patients with psoriasis and/or psoriatic arthritis. “We have a whole list of other trials in the pipeline with other institutions across the country.”
Through a grant, Dr. Mankad is assessing whether a PET scan could detect inflammation in the hearts of rheumatoid arthritis patients. “We’re looking to see if the reason these patients have heart failure later in life is because their heart muscle actually shows evidence of inflammation, even when they have no symptoms,” she explained.
Other tests such as echocardiogram and CT scans will be used to evaluate coronary disease in about 40-50 patients. The goal of using these multiple imaging tools is to find markers indicating that the heart is affected by rheumatoid arthritis, which may indicate a higher likelihood of developing heart failure, she said.
Clinics are popping up
Through these new clinics, some collaborations have emerged. Dr. Garshick works closely with Brigham and Women’s Hospital, which has a similar cardio-rheumatology program, run by Brittany Weber, MD, to exchange ideas, discuss challenging cases, and collaborate.
“There are a lot of clinics like us popping up across the country,” he observed. Every so often, he hears from other institutions that are interested in starting their own cardio-rheumatology programs. “They ask us: How do you start, what should we look for?”
It’s an education process for both patients and providers, Dr. Garshick emphasized. “I also think it’s a bandwidth issue. Many of our rheumatology and dermatology colleagues are acutely aware of the connection, but there may not be enough time at a clinic visit to really go in depth” with these dual conditions, he said.
NYU-Langone Health for the past several years has been holding a symposium to educate people on the cardio-rheumatology connection and treating inflammation in cardiovascular disease. This year’s symposium, held in conjunction with Brigham and Women’s Hospital, is scheduled for April 28. For more information, visit the course website: nyulmc.org/cvinflammationcme.
“What we’re trying to do is help [other institutions] get that bandwidth” to adequately help and serve these patients, he said.
Dr. Garshick has received consultant fees from Abbvie and Horizon therapeutics and an unrestricted research grant from Pfizer. No other sources had relevant financial disclosures.
Clinical cardiologist Heba Wassif, MD, MPH, knows the value of working with her fellow rheumatologists, surgeons, and other clinicians to establish a care plan for her patients with cardiac conditions and autoimmune diseases.
She is the cofounder of the Cleveland Clinic’s new cardio-rheumatology program, which places an emphasis on multidisciplinary care. In her role, Dr. Wassif closely follows her patients, and if she sees any inflammation or any other condition that requires the rheumatologist, she reaches out to her colleagues to adjust medications if needed.
Collaboration with a rheumatologist was important when a patient with valvular disease was prepping for surgery. The patient was on significant immunosuppressants and the surgery had to be timed appropriately, accounting for any decreases in her immunosuppression, explained Dr. Wassif, director of inpatient clinical cardiology at Cleveland Clinic in Ohio.
Cardio-rheumatology programs are “the newest child” in a series of cardiology offshoots focusing on different populations. Cardio-oncology and cardio-obstetrics took off about 6 years ago, with cardio-rheumatology clinics and interested physicians rising in number over the last several years, Dr. Wassif noted.
The relationship between cardiovascular diseases and rheumatologic conditions is certainly recognized more often, “which means more literature is being published to discuss the link,” according to Rekha Mankad, MD, a trailblazer of this model of care. She directs the Women’s Heart Clinic at Mayo Clinic in Rochester, Minn., which was one of the earliest adopters of a cardio-rheumatology clinic.
Ten years ago, “nobody was talking about the link between rheumatologic conditions and cardiovascular disease,” Dr. Mankad said. “I’ve been asked to speak on this topic, and programs have asked me to speak about establishing cardio-rheumatology practices. So, there’s been an evolution as far as a recognition that these two conditions overlap.”
Patients have come to her independent of internal referrals, which means they have done Google searches on cardiology and rheumatology. “I think that it has made a splash, at least in the world of cardiology,” Dr. Mankad observed in an interview.
Other institutions such as NYU-Langone, Yale, Stanford, Brigham and Women’s Hospital in Boston, and Women’s College Hospital in Toronto have formed similar clinics whose focus is to address the specific needs of rheumatology patients with cardiac conditions through a teamwork approach.
Challenges of treating cardiac, rheumatologic conditions
The rise in clinics addresses the longstanding connection between autoimmune disorders and cardiac conditions.
Cardiologists have known that there is an element of inflammation that contributes to atherosclerosis, said Dr. Wassif, who has researched this topic extensively. A recent study she led found a strong association between rheumatic immune-mediated inflammatory diseases (IMIDs) and high risk of acute coronary syndrome in Medicare patients.
“This particular population has a very clear increased risk for cardiovascular conditions, including valve disease and heart failure,” she emphasized.
Patients with rheumatoid arthritis and lupus have up to a twofold and eightfold higher risk of heart disease, respectively, noted Michael S. Garshick, MD, a cardiovascular disease specialist who directs the cardio-rheumatology program at NYU-Langone Health, in New York. Cardiologists “have really developed an understanding that the immune system can impact the heart, and that there’s a need for people to understand the nuance behind how the immune system can affect them and what to do about it,” Dr. Garshick said.
Caring for patients with both afflictions comes with specific challenges. Many physicians are not well trained on managing and treating patients with these dual conditions.
The “lipid paradox,” in which lipids are reduced with active inflammation in some rheumatologic conditions, can make treatment more nuanced. In addition, the traditional ASCVD (atherosclerotic cardiovascular disease) score often underestimates the cardiovascular risk of these patients, noted cardiologist Margaret Furman, MD, MPH, assistant professor and codirector of Yale’s Cardio-Rheumatology Program, New Haven, Conn.
Newer biologic medications used to treat rheumatologic diseases can alter a patient’s lipid profile, she said in an interview.
“It can be difficult to assess each individual patient’s cardiovascular risk as their disease state and treatment can vary throughout their lifetime based on their degree of inflammation. The importance of aggressive lipid management is often underestimated,” Dr. Furman added.
Cardiology and rheumatology partnerships can address gaps in care of this unique group of patients, said Vaidehi R. Chowdhary, MBBS, MD, clinical chief of the Yale Section of Rheumatology, Allergy, and Immunology at Yale University.
“The role of the rheumatologist in this dyad is to educate patients on this risk, work toward adequate control of inflammation, and minimize use of medications that contribute to increased cardiovascular risks,” said Dr. Chowdhary, who cofounded Yale’s cardio-rheumatology program with Dr. Furman.
Cardiologists in turn can assert their knowledge about medications and their impact on lipids and inflammation, Dr. Wassif said.
Many anti-inflammatory therapies are now within the cardiologist’s purview, Dr. Garshick noted. “For example, specifically with pericarditis, there’s [Food and Drug Administration]–approved anti-inflammatories or biologics. We’re the ones who feel the most comfortable giving them right now.” Cardiologists quite often are consulted about medications that are efficacious in rheumatologic conditions but could negatively impact the cardiovascular system, such as Janus kinase inhibitors, he added.
‘Reading the tea leaves’
Each program has its own unique story. For the Cleveland Clinic, the concept of a cardio-rheumatology program began during the COVID-19 pandemic in 2020. Developing such a concept and gaining institutional acceptance is always a work in process, Dr. Wassif said. “It’s not that you decide one day that you’re going to build a center, and that center is going to come into fruition overnight. You first gauge interest within your division. Who are the individuals that are interested in this area?”
Cleveland Clinic’s center is seeking to build relations between medical disciplines while spotlighting the concept of cardio-rheumatology, said Dr. Wassif, who has been providing education within the clinic and at other health institutions to ensure that patients receive appropriate attention early.
NYU-Langone launched its program amid this heightened awareness that the immune system could affect atherosclerosis, “kind of reading of the tea leaves, so to speak,” Dr. Garshick said.
Several clinical trials served as a catalyst for this movement. “A lot of clinical cardiologists were never 100% convinced that targeting the immune system reduced cardiovascular disease,” he said. Then the CANTOS clinical trial came along and showed for the first time that a therapeutic monoclonal antibody targeting interleukin-1beta, a cytokine central to inflammatory response, could in fact reduce cardiovascular disease.
Trials like this, along with epidemiologic literature connecting the rheumatologic and the autoimmune conditions with cardiovascular disease, pushed this concept to the forefront, Dr. Garshick said.
The notion that a clinic could successfully address cardiac problems in patients with rheumatic diseases yielded promising returns at Women’s College Hospital in Toronto, according to a report presented at the 2018 American College of Rheumatology annual meeting. Researchers reported that patients with rheumatologic conditions who attended a cardio-rheumatology clinic at this center saw improvements in care. The clinic identified increased cardiovascular risk and early atherosclerosis, and 53.8% of patients altered their medications after being seen in the clinic.
A total of 39.7% and 32.1% received lipid lowering and antiplatelet therapies, respectively, and 14% received antihypertensive therapy. A small percentage were treated for heart failure or placed on lifelong anticoagulation therapy for atrial fibrillation, and one patient received a percutaneous coronary stent.
Ins and outs of the referral process
Initially designed for preventive cardiac risk assessment, Yale’s program evolved into a multidisciplinary, patient-centered approach for the management of complex cardiovascular conditions in patients with autoimmune rheumatologic diseases.
The program is open to anyone who carries a diagnosis of rheumatologic disease or has elevated inflammatory markers. “Every patient, regardless of the reason for the referral, receives a cardiovascular risk assessment,” Dr. Furman said.
Most referrals come from rheumatologists, although cardiology colleagues and pulmonologists have also sent referrals. A pulmonologist, for example, may want to rule out a cardiac cause to shortness of breath. The patient’s workup, care, and follow-up are based on the reason for referral.
“We are currently referring patients with established cardiac disease, traditional risk factors, or for better risk assessment for primary prevention of coronary artery disease,” Dr. Chowdhary said. “We communicate very frequently about medication changes, and patients are aware of goals of care from both sides.”
Dr. Furman works closely with several of the rheumatology specialists taking care of patients with rheumatoid arthritis, systemic lupus erythematosus, and scleroderma.
Rheumatology follows patients every 3-6 months or more frequently based on their disease activity.
Dr. Mankad uses her sleuthing skills at Mayo Clinic to determine what the patients need. If they come in for a preventive assessment, she looks more closely at their cardiovascular risks and may order additional imaging to look for subclinical atherosclerosis. “We’re more aggressive with statin therapy in this population because of that,” she said.
If it’s valve disease, she pays extra attention to the patients’ valves in the echocardiograms and follows them a bit more regularly than someone without a rheumatologic condition and valve disease.
For patients with heart failure signs or symptoms, “it depends on how symptomatic they are,” Dr. Mankad said. In some instances, she may look for evidence of heart failure with preserved ejection fraction in patients who have rheumatoid arthritis who happen to be short of breath. “There’s so many different manifestations that patients with rheumatologic conditions can have as far as what could be affected in the heart,” she noted.
Quite frequently, Dr. Mankad identifies subclinical disease in her patients with rheumatoid arthritis. “I’ve seen many patients whose risk scores would not dictate statin therapy. But I went looking for subclinical disease by either doing coronary assessment or carotid assessment and have found atherosclerosis that would be enough to warrant statin therapy.”
A personalized assessment to reduce cardiac risk
NYU-Langone’s program offers opportunities to educate patients about the link between cardiac and rheumatologic disease.
“Their rheumatologist or their dermatologist will say, ‘Hey, have you heard about the connection between psoriasis, psoriatic or rheumatoid arthritis, and heart disease and the risk of heart attack or stroke?’ ” Dr. Garshick said.
The patients will often say they know nothing about these connections and want to learn more about how to treat it.
“We’ll say, ‘we have someone here that can help you.’ They’ll send them to myself or other colleagues like me across the country. We’ll assess blood pressure, weight, lipids, hemoglobin A1c, and other serologic and oftentimes imaging biomarkers of cardiovascular risk.” The patients will receive a personalized assessment, listing things they can do to lower their risk, whether it’s diet, exercise, or lifestyle. “Many times it can involve medications to reduce heart disease risk,” said Dr. Garshick.
In some instances, a rheumatologist or dermatologist may be concerned about starting a patient on a specific medication for the disease such as a JAK inhibitor. “We’ll help assess their risk because there’s been a lot of literature out in the rheumatology world about the risk of JAK inhibitors and heart disease and blood clots,” said Dr. Garshick.
Dr. Garshick also sees patients with rheumatologic conditions who have a specific cardiovascular concern or complaint such as shortness of breath or chest pain. “We’ll work that up with a specific knowledge of the underlying immune condition and how that may impact their heart,” he said.
Advances in research
As they continue to see patients and devise specific care plans, developers of cardio-rheumatology programs have been supplementing their work with ongoing research.
Yale’s clinic is expanding this year to include a new attending physician, Attila Feher, MD, PhD, who has conducted research in autoimmunity and microcirculation using molecular imaging and multimodality imaging techniques. Prevalence of coronary microvascular dysfunction appears to be increased in this patient population, Dr. Furman said.
Dr. Wassif recently coauthored a paper that examined patients with underlying rheumatologic conditions who undergo valvular and aortic valve replacement. “To our surprise, there was really no difference between patients with autoimmune conditions and others with nonautoimmune conditions,” she said, adding that the study had its limitations.
Other work includes data on Medicare patients with ST- and non-ST-elevation myocardial infarctions who have an underlying autoimmune disorder. Dr. Wassif and her colleagues found that their long-term outcomes are worse than those of patients without these conditions. “It’s unclear if worse outcomes are related to complications of autoimmunity versus the extent of their underlying disease. This is a work in progress and certainly an area that is ripe for research.”
Dr. Garshick and other collaborators at NYU have been focusing on the endothelium, specifically platelet biology in patients with psoriasis, psoriatic arthritis, and lupus. “We’re about to start the same research with gout as well,” he said.
“The process we’re most interested in is understanding how these diseases impact the early stages of cholesterol. And the way we’re doing that is evaluating the vasculature, specifically the endothelium,” he said.
He has finished two clinical trials that evaluate how standard heart disease medications such as aspirin and statins impact or can potentially benefit patients with psoriasis and/or psoriatic arthritis. “We have a whole list of other trials in the pipeline with other institutions across the country.”
Through a grant, Dr. Mankad is assessing whether a PET scan could detect inflammation in the hearts of rheumatoid arthritis patients. “We’re looking to see if the reason these patients have heart failure later in life is because their heart muscle actually shows evidence of inflammation, even when they have no symptoms,” she explained.
Other tests such as echocardiogram and CT scans will be used to evaluate coronary disease in about 40-50 patients. The goal of using these multiple imaging tools is to find markers indicating that the heart is affected by rheumatoid arthritis, which may indicate a higher likelihood of developing heart failure, she said.
Clinics are popping up
Through these new clinics, some collaborations have emerged. Dr. Garshick works closely with Brigham and Women’s Hospital, which has a similar cardio-rheumatology program, run by Brittany Weber, MD, to exchange ideas, discuss challenging cases, and collaborate.
“There are a lot of clinics like us popping up across the country,” he observed. Every so often, he hears from other institutions that are interested in starting their own cardio-rheumatology programs. “They ask us: How do you start, what should we look for?”
It’s an education process for both patients and providers, Dr. Garshick emphasized. “I also think it’s a bandwidth issue. Many of our rheumatology and dermatology colleagues are acutely aware of the connection, but there may not be enough time at a clinic visit to really go in depth” with these dual conditions, he said.
NYU-Langone Health for the past several years has been holding a symposium to educate people on the cardio-rheumatology connection and treating inflammation in cardiovascular disease. This year’s symposium, held in conjunction with Brigham and Women’s Hospital, is scheduled for April 28. For more information, visit the course website: nyulmc.org/cvinflammationcme.
“What we’re trying to do is help [other institutions] get that bandwidth” to adequately help and serve these patients, he said.
Dr. Garshick has received consultant fees from Abbvie and Horizon therapeutics and an unrestricted research grant from Pfizer. No other sources had relevant financial disclosures.
Clinical cardiologist Heba Wassif, MD, MPH, knows the value of working with her fellow rheumatologists, surgeons, and other clinicians to establish a care plan for her patients with cardiac conditions and autoimmune diseases.
She is the cofounder of the Cleveland Clinic’s new cardio-rheumatology program, which places an emphasis on multidisciplinary care. In her role, Dr. Wassif closely follows her patients, and if she sees any inflammation or any other condition that requires the rheumatologist, she reaches out to her colleagues to adjust medications if needed.
Collaboration with a rheumatologist was important when a patient with valvular disease was prepping for surgery. The patient was on significant immunosuppressants and the surgery had to be timed appropriately, accounting for any decreases in her immunosuppression, explained Dr. Wassif, director of inpatient clinical cardiology at Cleveland Clinic in Ohio.
Cardio-rheumatology programs are “the newest child” in a series of cardiology offshoots focusing on different populations. Cardio-oncology and cardio-obstetrics took off about 6 years ago, with cardio-rheumatology clinics and interested physicians rising in number over the last several years, Dr. Wassif noted.
The relationship between cardiovascular diseases and rheumatologic conditions is certainly recognized more often, “which means more literature is being published to discuss the link,” according to Rekha Mankad, MD, a trailblazer of this model of care. She directs the Women’s Heart Clinic at Mayo Clinic in Rochester, Minn., which was one of the earliest adopters of a cardio-rheumatology clinic.
Ten years ago, “nobody was talking about the link between rheumatologic conditions and cardiovascular disease,” Dr. Mankad said. “I’ve been asked to speak on this topic, and programs have asked me to speak about establishing cardio-rheumatology practices. So, there’s been an evolution as far as a recognition that these two conditions overlap.”
Patients have come to her independent of internal referrals, which means they have done Google searches on cardiology and rheumatology. “I think that it has made a splash, at least in the world of cardiology,” Dr. Mankad observed in an interview.
Other institutions such as NYU-Langone, Yale, Stanford, Brigham and Women’s Hospital in Boston, and Women’s College Hospital in Toronto have formed similar clinics whose focus is to address the specific needs of rheumatology patients with cardiac conditions through a teamwork approach.
Challenges of treating cardiac, rheumatologic conditions
The rise in clinics addresses the longstanding connection between autoimmune disorders and cardiac conditions.
Cardiologists have known that there is an element of inflammation that contributes to atherosclerosis, said Dr. Wassif, who has researched this topic extensively. A recent study she led found a strong association between rheumatic immune-mediated inflammatory diseases (IMIDs) and high risk of acute coronary syndrome in Medicare patients.
“This particular population has a very clear increased risk for cardiovascular conditions, including valve disease and heart failure,” she emphasized.
Patients with rheumatoid arthritis and lupus have up to a twofold and eightfold higher risk of heart disease, respectively, noted Michael S. Garshick, MD, a cardiovascular disease specialist who directs the cardio-rheumatology program at NYU-Langone Health, in New York. Cardiologists “have really developed an understanding that the immune system can impact the heart, and that there’s a need for people to understand the nuance behind how the immune system can affect them and what to do about it,” Dr. Garshick said.
Caring for patients with both afflictions comes with specific challenges. Many physicians are not well trained on managing and treating patients with these dual conditions.
The “lipid paradox,” in which lipids are reduced with active inflammation in some rheumatologic conditions, can make treatment more nuanced. In addition, the traditional ASCVD (atherosclerotic cardiovascular disease) score often underestimates the cardiovascular risk of these patients, noted cardiologist Margaret Furman, MD, MPH, assistant professor and codirector of Yale’s Cardio-Rheumatology Program, New Haven, Conn.
Newer biologic medications used to treat rheumatologic diseases can alter a patient’s lipid profile, she said in an interview.
“It can be difficult to assess each individual patient’s cardiovascular risk as their disease state and treatment can vary throughout their lifetime based on their degree of inflammation. The importance of aggressive lipid management is often underestimated,” Dr. Furman added.
Cardiology and rheumatology partnerships can address gaps in care of this unique group of patients, said Vaidehi R. Chowdhary, MBBS, MD, clinical chief of the Yale Section of Rheumatology, Allergy, and Immunology at Yale University.
“The role of the rheumatologist in this dyad is to educate patients on this risk, work toward adequate control of inflammation, and minimize use of medications that contribute to increased cardiovascular risks,” said Dr. Chowdhary, who cofounded Yale’s cardio-rheumatology program with Dr. Furman.
Cardiologists in turn can assert their knowledge about medications and their impact on lipids and inflammation, Dr. Wassif said.
Many anti-inflammatory therapies are now within the cardiologist’s purview, Dr. Garshick noted. “For example, specifically with pericarditis, there’s [Food and Drug Administration]–approved anti-inflammatories or biologics. We’re the ones who feel the most comfortable giving them right now.” Cardiologists quite often are consulted about medications that are efficacious in rheumatologic conditions but could negatively impact the cardiovascular system, such as Janus kinase inhibitors, he added.
‘Reading the tea leaves’
Each program has its own unique story. For the Cleveland Clinic, the concept of a cardio-rheumatology program began during the COVID-19 pandemic in 2020. Developing such a concept and gaining institutional acceptance is always a work in process, Dr. Wassif said. “It’s not that you decide one day that you’re going to build a center, and that center is going to come into fruition overnight. You first gauge interest within your division. Who are the individuals that are interested in this area?”
Cleveland Clinic’s center is seeking to build relations between medical disciplines while spotlighting the concept of cardio-rheumatology, said Dr. Wassif, who has been providing education within the clinic and at other health institutions to ensure that patients receive appropriate attention early.
NYU-Langone launched its program amid this heightened awareness that the immune system could affect atherosclerosis, “kind of reading of the tea leaves, so to speak,” Dr. Garshick said.
Several clinical trials served as a catalyst for this movement. “A lot of clinical cardiologists were never 100% convinced that targeting the immune system reduced cardiovascular disease,” he said. Then the CANTOS clinical trial came along and showed for the first time that a therapeutic monoclonal antibody targeting interleukin-1beta, a cytokine central to inflammatory response, could in fact reduce cardiovascular disease.
Trials like this, along with epidemiologic literature connecting the rheumatologic and the autoimmune conditions with cardiovascular disease, pushed this concept to the forefront, Dr. Garshick said.
The notion that a clinic could successfully address cardiac problems in patients with rheumatic diseases yielded promising returns at Women’s College Hospital in Toronto, according to a report presented at the 2018 American College of Rheumatology annual meeting. Researchers reported that patients with rheumatologic conditions who attended a cardio-rheumatology clinic at this center saw improvements in care. The clinic identified increased cardiovascular risk and early atherosclerosis, and 53.8% of patients altered their medications after being seen in the clinic.
A total of 39.7% and 32.1% received lipid lowering and antiplatelet therapies, respectively, and 14% received antihypertensive therapy. A small percentage were treated for heart failure or placed on lifelong anticoagulation therapy for atrial fibrillation, and one patient received a percutaneous coronary stent.
Ins and outs of the referral process
Initially designed for preventive cardiac risk assessment, Yale’s program evolved into a multidisciplinary, patient-centered approach for the management of complex cardiovascular conditions in patients with autoimmune rheumatologic diseases.
The program is open to anyone who carries a diagnosis of rheumatologic disease or has elevated inflammatory markers. “Every patient, regardless of the reason for the referral, receives a cardiovascular risk assessment,” Dr. Furman said.
Most referrals come from rheumatologists, although cardiology colleagues and pulmonologists have also sent referrals. A pulmonologist, for example, may want to rule out a cardiac cause to shortness of breath. The patient’s workup, care, and follow-up are based on the reason for referral.
“We are currently referring patients with established cardiac disease, traditional risk factors, or for better risk assessment for primary prevention of coronary artery disease,” Dr. Chowdhary said. “We communicate very frequently about medication changes, and patients are aware of goals of care from both sides.”
Dr. Furman works closely with several of the rheumatology specialists taking care of patients with rheumatoid arthritis, systemic lupus erythematosus, and scleroderma.
Rheumatology follows patients every 3-6 months or more frequently based on their disease activity.
Dr. Mankad uses her sleuthing skills at Mayo Clinic to determine what the patients need. If they come in for a preventive assessment, she looks more closely at their cardiovascular risks and may order additional imaging to look for subclinical atherosclerosis. “We’re more aggressive with statin therapy in this population because of that,” she said.
If it’s valve disease, she pays extra attention to the patients’ valves in the echocardiograms and follows them a bit more regularly than someone without a rheumatologic condition and valve disease.
For patients with heart failure signs or symptoms, “it depends on how symptomatic they are,” Dr. Mankad said. In some instances, she may look for evidence of heart failure with preserved ejection fraction in patients who have rheumatoid arthritis who happen to be short of breath. “There’s so many different manifestations that patients with rheumatologic conditions can have as far as what could be affected in the heart,” she noted.
Quite frequently, Dr. Mankad identifies subclinical disease in her patients with rheumatoid arthritis. “I’ve seen many patients whose risk scores would not dictate statin therapy. But I went looking for subclinical disease by either doing coronary assessment or carotid assessment and have found atherosclerosis that would be enough to warrant statin therapy.”
A personalized assessment to reduce cardiac risk
NYU-Langone’s program offers opportunities to educate patients about the link between cardiac and rheumatologic disease.
“Their rheumatologist or their dermatologist will say, ‘Hey, have you heard about the connection between psoriasis, psoriatic or rheumatoid arthritis, and heart disease and the risk of heart attack or stroke?’ ” Dr. Garshick said.
The patients will often say they know nothing about these connections and want to learn more about how to treat it.
“We’ll say, ‘we have someone here that can help you.’ They’ll send them to myself or other colleagues like me across the country. We’ll assess blood pressure, weight, lipids, hemoglobin A1c, and other serologic and oftentimes imaging biomarkers of cardiovascular risk.” The patients will receive a personalized assessment, listing things they can do to lower their risk, whether it’s diet, exercise, or lifestyle. “Many times it can involve medications to reduce heart disease risk,” said Dr. Garshick.
In some instances, a rheumatologist or dermatologist may be concerned about starting a patient on a specific medication for the disease such as a JAK inhibitor. “We’ll help assess their risk because there’s been a lot of literature out in the rheumatology world about the risk of JAK inhibitors and heart disease and blood clots,” said Dr. Garshick.
Dr. Garshick also sees patients with rheumatologic conditions who have a specific cardiovascular concern or complaint such as shortness of breath or chest pain. “We’ll work that up with a specific knowledge of the underlying immune condition and how that may impact their heart,” he said.
Advances in research
As they continue to see patients and devise specific care plans, developers of cardio-rheumatology programs have been supplementing their work with ongoing research.
Yale’s clinic is expanding this year to include a new attending physician, Attila Feher, MD, PhD, who has conducted research in autoimmunity and microcirculation using molecular imaging and multimodality imaging techniques. Prevalence of coronary microvascular dysfunction appears to be increased in this patient population, Dr. Furman said.
Dr. Wassif recently coauthored a paper that examined patients with underlying rheumatologic conditions who undergo valvular and aortic valve replacement. “To our surprise, there was really no difference between patients with autoimmune conditions and others with nonautoimmune conditions,” she said, adding that the study had its limitations.
Other work includes data on Medicare patients with ST- and non-ST-elevation myocardial infarctions who have an underlying autoimmune disorder. Dr. Wassif and her colleagues found that their long-term outcomes are worse than those of patients without these conditions. “It’s unclear if worse outcomes are related to complications of autoimmunity versus the extent of their underlying disease. This is a work in progress and certainly an area that is ripe for research.”
Dr. Garshick and other collaborators at NYU have been focusing on the endothelium, specifically platelet biology in patients with psoriasis, psoriatic arthritis, and lupus. “We’re about to start the same research with gout as well,” he said.
“The process we’re most interested in is understanding how these diseases impact the early stages of cholesterol. And the way we’re doing that is evaluating the vasculature, specifically the endothelium,” he said.
He has finished two clinical trials that evaluate how standard heart disease medications such as aspirin and statins impact or can potentially benefit patients with psoriasis and/or psoriatic arthritis. “We have a whole list of other trials in the pipeline with other institutions across the country.”
Through a grant, Dr. Mankad is assessing whether a PET scan could detect inflammation in the hearts of rheumatoid arthritis patients. “We’re looking to see if the reason these patients have heart failure later in life is because their heart muscle actually shows evidence of inflammation, even when they have no symptoms,” she explained.
Other tests such as echocardiogram and CT scans will be used to evaluate coronary disease in about 40-50 patients. The goal of using these multiple imaging tools is to find markers indicating that the heart is affected by rheumatoid arthritis, which may indicate a higher likelihood of developing heart failure, she said.
Clinics are popping up
Through these new clinics, some collaborations have emerged. Dr. Garshick works closely with Brigham and Women’s Hospital, which has a similar cardio-rheumatology program, run by Brittany Weber, MD, to exchange ideas, discuss challenging cases, and collaborate.
“There are a lot of clinics like us popping up across the country,” he observed. Every so often, he hears from other institutions that are interested in starting their own cardio-rheumatology programs. “They ask us: How do you start, what should we look for?”
It’s an education process for both patients and providers, Dr. Garshick emphasized. “I also think it’s a bandwidth issue. Many of our rheumatology and dermatology colleagues are acutely aware of the connection, but there may not be enough time at a clinic visit to really go in depth” with these dual conditions, he said.
NYU-Langone Health for the past several years has been holding a symposium to educate people on the cardio-rheumatology connection and treating inflammation in cardiovascular disease. This year’s symposium, held in conjunction with Brigham and Women’s Hospital, is scheduled for April 28. For more information, visit the course website: nyulmc.org/cvinflammationcme.
“What we’re trying to do is help [other institutions] get that bandwidth” to adequately help and serve these patients, he said.
Dr. Garshick has received consultant fees from Abbvie and Horizon therapeutics and an unrestricted research grant from Pfizer. No other sources had relevant financial disclosures.
Phil Robinson: Rheumatologist, colleague, huntsman spider rescuer
Helen Tanner remembers stealing glimpses of her husband, Philip (“Phil”) Robinson, MBChB, PhD, associate professor at the University of Queensland (Australia), catching and rehoming huge Huntsman spiders. Robinson made the extra effort because he didn’t want to hurt them; he wasn’t a big fan of the large spider with a potential leg span of 6 inches that’s commonly found in Australia, per the Australian Museum.
Robinson also relished taking his children, Eddie, 4, and Tommy, 7, on roller coaster rides, which they enjoyed, despite the experience typically giving him motion sickness, Tanner said.
“He would do anything to make the children happy,” she said. “His children meant the world to him.”
Robinson died Jan. 3 as a result of diffuse gastric adenocarcinoma, according to his wife, who added that it was a short, 2-week-long illness.
A leader of global effort to understand COVID-19 and rheumatic disease
Jinoos Yazdany, MD, MPH, chief of the division of rheumatology at Zuckerberg San Francisco General Hospital and professor of medicine at the University of California, San Francisco, described Robinson as “one of the hardest-working people I have ever known. ... [still] his deep love and dedication for his family and kids was always present.”
Robinson would wake up early, even on weekends, to lead international calls across multiple time zones, Yazdany said. “He was driven by a deep curiosity and an intense desire to generate scholarship that would help people with rheumatic diseases.”
Yazdany added that Robinson had a full research portfolio in gout and spondyloarthritis and a busy clinical practice.
She often caught glimpses of Robinson’s young children during Zoom calls. “He was also a talented baker and loved to bake with his kids, often posting pictures of his creations on social media,” Yazdany said.
A mutual colleague compiled some of Robinson’s baking successes. That includes “ ‘probably about to be locked down’ cookies” on July 17, 2021, and “Queensland lockdown cookies!!!” on July 2, 2021. Reuters reported on July 21, 2021, that Australia was witnessing an alarming increase in COVID-19 cases.
Robinson also worked his social media skills to rally support for the COVID-19 Global Rheumatology Alliance, according to an article published by his colleagues in The Rheumatologist. Yazdany collaborated with him in this effort.
Launched on March 12, 2020, the Global Rheumatology Alliance’s mission is to “collect, analyze, and disseminate information about COVID-19 and rheumatology to patients, physicians, and other relevant groups to improve the care of patients with rheumatic disease.” Robinson served as chair of governance and policy for the collaborative effort.
Inspired by a conversation on Twitter by Leonard Calabrese, DO, a rheumatologist at the Cleveland Clinic in Ohio, about an outcomes registry created by gastroenterologists specific to patients with inflammatory bowel disease, Robinson launched a discussion about a similar effort for rheumatology on Twitter, they write.
Along with colleagues and within a single Zoom conference call, Yazdany and Robinson had a plan to organize the registry, Robinson’s colleagues write.
Two projects of the Global Rheumatology Alliance are a health care provider–entered registry for providers to enter data about rheumatology patients with COVID-19 infections, and their COVID-19 Vax Survey, which is available in 12 languages, including English.
Yazdany had never met Robinson before she started working with him on the Global Rheumatology Alliance. They started chatting on Twitter, then moved to Zoom conference calls, and subsequently had weeks when they talked by phone and “emailed constantly,” she said.
“As I reflect on our initial interactions, I am struck by how brilliantly we got along and trusted each other,” Yazdany told this news organization. “We both liked to think big, believed in inclusive collaborations, and were committed to helping people with rheumatic diseases during a scary and uncertain time.”
Still, Yazdany noted that she and Phil brought different strengths to their collaborations. She brought her skills related to the technical aspects of research databases, while “Phil worked his magic in mobilizing friends and colleagues from all over the world,” she said. “He served as a wonderful leader, one whom people believed in and would follow.”
The two colleagues, who spent much of their collaborations over Zoom calls, email, and Slack, while living more than 7,000 miles apart, finally met in person at ACR Convergence 2022, which took place in Philadelphia that year. “It felt like the best kind of reunion with a dear friend,” Yazdany remembered.
A mentor who created a platform for ‘good people to do great things’
David Liew, MBBS, PhD, consultant rheumatologist and clinical pharmacologist at Austin Health in Melbourne, marveled at Robinson’s ability to “distill things simply and cleanly, with clarity but without losing detail.” Liew, who collaborated with Robinson throughout the COVID-19 pandemic, likens the experience to “jamming with [jazz musician John] Coltrane.”
“What I think was particularly remarkable was the capacity to not only have those thoughts himself, but to facilitate others to have that springboard,” said Liew, who added that Robinson “took enormous pleasure in facilitating others’ success.”
“I think the greatest joy he drew out of the COVID-19 Global Rheumatology Alliance, apart from facing up to the challenge that needed to be faced, was creating a platform for good people to do great things,” said Liew, who recalled one situation where Robinson gently challenged him. Looking back now, Liew can “now see he very clearly was laying me up, giving me the best chance to shine.”
He describes Robinson as “a deep soul who loved his wife and two sons enormously” and “a whiskey aficionado.” “[Whiskey] suited his contemplative style,” Liew recalled. “Some of my fondest conversations with him were over a whiskey, either in person or virtually, pondering the ‘big issues.’”
A ‘friend and a colleague and so much more’
Claire Barrett, MBBS, president of the Australian Rheumatology Association, described Robinson as a “friend and a colleague and so much more. ... [He was] someone who I worked, laughed, ate, drank, danced, and had fun with,” she told this news organization. They served together as volunteers for the Australian Rheumatology Association and its Queensland branch, as well as Arthritis Queensland; they were also colleagues at Metro North Hospital and Health Service in Brisbane, Queensland.
Robinson was her “go to” for insightful comment on a variety of topics, she said. That could be advice on managing a patient with difficult gout, challenging spondyloarthritis, or the best treatment for a patient with COVID-19.
“[Phil] was a friend I could ask about anything, knowing I would not be judged,” Barrett said. “His kids and our grandkids are similar ages, so we would swap stories and photos and laugh about how cute/funny/cuddly/busy/etc. they were. My heart is broken he won’t get the chance to enjoy their future and the excitement of having Phil continuing to be such an active dad.”
Tanner, Robinson’s wife, said, “he loved everything.” That included the academic side of medicine, and working out what was wrong with his patients and helping them get better. “He was dedicated to this,” she added.
“He also loved the camaraderie of the job – all the people he met and interacted with. [Phil] loved sharing his ideas for research and also discussing complex patients with colleagues. He was driven by finding the answers to problems and doing this as part of a team of researchers/clinicians. He wasn’t interested in personal success.”
Robinson received his medical degree from Otago Medical School in Dunedin, New Zealand, according to the University of Queensland. His specialty training in general and acute care medicine and rheumatology was completed in Wellington, New Zealand, and Dunedin. Robinson also achieved a PhD in human genetics at the University of Queensland Diamantina Institute and had a postdoctoral fellowship at the Queensland Brain Institute at the University of Queensland.
Before his death, he worked at the Royal Brisbane and Women’s Hospital in Herston, Queensland, and at St. Andrew’s War Memorial Hospital in Spring Hill in Brisbane.
A version of this article first appeared on Medscape.com.
Helen Tanner remembers stealing glimpses of her husband, Philip (“Phil”) Robinson, MBChB, PhD, associate professor at the University of Queensland (Australia), catching and rehoming huge Huntsman spiders. Robinson made the extra effort because he didn’t want to hurt them; he wasn’t a big fan of the large spider with a potential leg span of 6 inches that’s commonly found in Australia, per the Australian Museum.
Robinson also relished taking his children, Eddie, 4, and Tommy, 7, on roller coaster rides, which they enjoyed, despite the experience typically giving him motion sickness, Tanner said.
“He would do anything to make the children happy,” she said. “His children meant the world to him.”
Robinson died Jan. 3 as a result of diffuse gastric adenocarcinoma, according to his wife, who added that it was a short, 2-week-long illness.
A leader of global effort to understand COVID-19 and rheumatic disease
Jinoos Yazdany, MD, MPH, chief of the division of rheumatology at Zuckerberg San Francisco General Hospital and professor of medicine at the University of California, San Francisco, described Robinson as “one of the hardest-working people I have ever known. ... [still] his deep love and dedication for his family and kids was always present.”
Robinson would wake up early, even on weekends, to lead international calls across multiple time zones, Yazdany said. “He was driven by a deep curiosity and an intense desire to generate scholarship that would help people with rheumatic diseases.”
Yazdany added that Robinson had a full research portfolio in gout and spondyloarthritis and a busy clinical practice.
She often caught glimpses of Robinson’s young children during Zoom calls. “He was also a talented baker and loved to bake with his kids, often posting pictures of his creations on social media,” Yazdany said.
A mutual colleague compiled some of Robinson’s baking successes. That includes “ ‘probably about to be locked down’ cookies” on July 17, 2021, and “Queensland lockdown cookies!!!” on July 2, 2021. Reuters reported on July 21, 2021, that Australia was witnessing an alarming increase in COVID-19 cases.
Robinson also worked his social media skills to rally support for the COVID-19 Global Rheumatology Alliance, according to an article published by his colleagues in The Rheumatologist. Yazdany collaborated with him in this effort.
Launched on March 12, 2020, the Global Rheumatology Alliance’s mission is to “collect, analyze, and disseminate information about COVID-19 and rheumatology to patients, physicians, and other relevant groups to improve the care of patients with rheumatic disease.” Robinson served as chair of governance and policy for the collaborative effort.
Inspired by a conversation on Twitter by Leonard Calabrese, DO, a rheumatologist at the Cleveland Clinic in Ohio, about an outcomes registry created by gastroenterologists specific to patients with inflammatory bowel disease, Robinson launched a discussion about a similar effort for rheumatology on Twitter, they write.
Along with colleagues and within a single Zoom conference call, Yazdany and Robinson had a plan to organize the registry, Robinson’s colleagues write.
Two projects of the Global Rheumatology Alliance are a health care provider–entered registry for providers to enter data about rheumatology patients with COVID-19 infections, and their COVID-19 Vax Survey, which is available in 12 languages, including English.
Yazdany had never met Robinson before she started working with him on the Global Rheumatology Alliance. They started chatting on Twitter, then moved to Zoom conference calls, and subsequently had weeks when they talked by phone and “emailed constantly,” she said.
“As I reflect on our initial interactions, I am struck by how brilliantly we got along and trusted each other,” Yazdany told this news organization. “We both liked to think big, believed in inclusive collaborations, and were committed to helping people with rheumatic diseases during a scary and uncertain time.”
Still, Yazdany noted that she and Phil brought different strengths to their collaborations. She brought her skills related to the technical aspects of research databases, while “Phil worked his magic in mobilizing friends and colleagues from all over the world,” she said. “He served as a wonderful leader, one whom people believed in and would follow.”
The two colleagues, who spent much of their collaborations over Zoom calls, email, and Slack, while living more than 7,000 miles apart, finally met in person at ACR Convergence 2022, which took place in Philadelphia that year. “It felt like the best kind of reunion with a dear friend,” Yazdany remembered.
A mentor who created a platform for ‘good people to do great things’
David Liew, MBBS, PhD, consultant rheumatologist and clinical pharmacologist at Austin Health in Melbourne, marveled at Robinson’s ability to “distill things simply and cleanly, with clarity but without losing detail.” Liew, who collaborated with Robinson throughout the COVID-19 pandemic, likens the experience to “jamming with [jazz musician John] Coltrane.”
“What I think was particularly remarkable was the capacity to not only have those thoughts himself, but to facilitate others to have that springboard,” said Liew, who added that Robinson “took enormous pleasure in facilitating others’ success.”
“I think the greatest joy he drew out of the COVID-19 Global Rheumatology Alliance, apart from facing up to the challenge that needed to be faced, was creating a platform for good people to do great things,” said Liew, who recalled one situation where Robinson gently challenged him. Looking back now, Liew can “now see he very clearly was laying me up, giving me the best chance to shine.”
He describes Robinson as “a deep soul who loved his wife and two sons enormously” and “a whiskey aficionado.” “[Whiskey] suited his contemplative style,” Liew recalled. “Some of my fondest conversations with him were over a whiskey, either in person or virtually, pondering the ‘big issues.’”
A ‘friend and a colleague and so much more’
Claire Barrett, MBBS, president of the Australian Rheumatology Association, described Robinson as a “friend and a colleague and so much more. ... [He was] someone who I worked, laughed, ate, drank, danced, and had fun with,” she told this news organization. They served together as volunteers for the Australian Rheumatology Association and its Queensland branch, as well as Arthritis Queensland; they were also colleagues at Metro North Hospital and Health Service in Brisbane, Queensland.
Robinson was her “go to” for insightful comment on a variety of topics, she said. That could be advice on managing a patient with difficult gout, challenging spondyloarthritis, or the best treatment for a patient with COVID-19.
“[Phil] was a friend I could ask about anything, knowing I would not be judged,” Barrett said. “His kids and our grandkids are similar ages, so we would swap stories and photos and laugh about how cute/funny/cuddly/busy/etc. they were. My heart is broken he won’t get the chance to enjoy their future and the excitement of having Phil continuing to be such an active dad.”
Tanner, Robinson’s wife, said, “he loved everything.” That included the academic side of medicine, and working out what was wrong with his patients and helping them get better. “He was dedicated to this,” she added.
“He also loved the camaraderie of the job – all the people he met and interacted with. [Phil] loved sharing his ideas for research and also discussing complex patients with colleagues. He was driven by finding the answers to problems and doing this as part of a team of researchers/clinicians. He wasn’t interested in personal success.”
Robinson received his medical degree from Otago Medical School in Dunedin, New Zealand, according to the University of Queensland. His specialty training in general and acute care medicine and rheumatology was completed in Wellington, New Zealand, and Dunedin. Robinson also achieved a PhD in human genetics at the University of Queensland Diamantina Institute and had a postdoctoral fellowship at the Queensland Brain Institute at the University of Queensland.
Before his death, he worked at the Royal Brisbane and Women’s Hospital in Herston, Queensland, and at St. Andrew’s War Memorial Hospital in Spring Hill in Brisbane.
A version of this article first appeared on Medscape.com.
Helen Tanner remembers stealing glimpses of her husband, Philip (“Phil”) Robinson, MBChB, PhD, associate professor at the University of Queensland (Australia), catching and rehoming huge Huntsman spiders. Robinson made the extra effort because he didn’t want to hurt them; he wasn’t a big fan of the large spider with a potential leg span of 6 inches that’s commonly found in Australia, per the Australian Museum.
Robinson also relished taking his children, Eddie, 4, and Tommy, 7, on roller coaster rides, which they enjoyed, despite the experience typically giving him motion sickness, Tanner said.
“He would do anything to make the children happy,” she said. “His children meant the world to him.”
Robinson died Jan. 3 as a result of diffuse gastric adenocarcinoma, according to his wife, who added that it was a short, 2-week-long illness.
A leader of global effort to understand COVID-19 and rheumatic disease
Jinoos Yazdany, MD, MPH, chief of the division of rheumatology at Zuckerberg San Francisco General Hospital and professor of medicine at the University of California, San Francisco, described Robinson as “one of the hardest-working people I have ever known. ... [still] his deep love and dedication for his family and kids was always present.”
Robinson would wake up early, even on weekends, to lead international calls across multiple time zones, Yazdany said. “He was driven by a deep curiosity and an intense desire to generate scholarship that would help people with rheumatic diseases.”
Yazdany added that Robinson had a full research portfolio in gout and spondyloarthritis and a busy clinical practice.
She often caught glimpses of Robinson’s young children during Zoom calls. “He was also a talented baker and loved to bake with his kids, often posting pictures of his creations on social media,” Yazdany said.
A mutual colleague compiled some of Robinson’s baking successes. That includes “ ‘probably about to be locked down’ cookies” on July 17, 2021, and “Queensland lockdown cookies!!!” on July 2, 2021. Reuters reported on July 21, 2021, that Australia was witnessing an alarming increase in COVID-19 cases.
Robinson also worked his social media skills to rally support for the COVID-19 Global Rheumatology Alliance, according to an article published by his colleagues in The Rheumatologist. Yazdany collaborated with him in this effort.
Launched on March 12, 2020, the Global Rheumatology Alliance’s mission is to “collect, analyze, and disseminate information about COVID-19 and rheumatology to patients, physicians, and other relevant groups to improve the care of patients with rheumatic disease.” Robinson served as chair of governance and policy for the collaborative effort.
Inspired by a conversation on Twitter by Leonard Calabrese, DO, a rheumatologist at the Cleveland Clinic in Ohio, about an outcomes registry created by gastroenterologists specific to patients with inflammatory bowel disease, Robinson launched a discussion about a similar effort for rheumatology on Twitter, they write.
Along with colleagues and within a single Zoom conference call, Yazdany and Robinson had a plan to organize the registry, Robinson’s colleagues write.
Two projects of the Global Rheumatology Alliance are a health care provider–entered registry for providers to enter data about rheumatology patients with COVID-19 infections, and their COVID-19 Vax Survey, which is available in 12 languages, including English.
Yazdany had never met Robinson before she started working with him on the Global Rheumatology Alliance. They started chatting on Twitter, then moved to Zoom conference calls, and subsequently had weeks when they talked by phone and “emailed constantly,” she said.
“As I reflect on our initial interactions, I am struck by how brilliantly we got along and trusted each other,” Yazdany told this news organization. “We both liked to think big, believed in inclusive collaborations, and were committed to helping people with rheumatic diseases during a scary and uncertain time.”
Still, Yazdany noted that she and Phil brought different strengths to their collaborations. She brought her skills related to the technical aspects of research databases, while “Phil worked his magic in mobilizing friends and colleagues from all over the world,” she said. “He served as a wonderful leader, one whom people believed in and would follow.”
The two colleagues, who spent much of their collaborations over Zoom calls, email, and Slack, while living more than 7,000 miles apart, finally met in person at ACR Convergence 2022, which took place in Philadelphia that year. “It felt like the best kind of reunion with a dear friend,” Yazdany remembered.
A mentor who created a platform for ‘good people to do great things’
David Liew, MBBS, PhD, consultant rheumatologist and clinical pharmacologist at Austin Health in Melbourne, marveled at Robinson’s ability to “distill things simply and cleanly, with clarity but without losing detail.” Liew, who collaborated with Robinson throughout the COVID-19 pandemic, likens the experience to “jamming with [jazz musician John] Coltrane.”
“What I think was particularly remarkable was the capacity to not only have those thoughts himself, but to facilitate others to have that springboard,” said Liew, who added that Robinson “took enormous pleasure in facilitating others’ success.”
“I think the greatest joy he drew out of the COVID-19 Global Rheumatology Alliance, apart from facing up to the challenge that needed to be faced, was creating a platform for good people to do great things,” said Liew, who recalled one situation where Robinson gently challenged him. Looking back now, Liew can “now see he very clearly was laying me up, giving me the best chance to shine.”
He describes Robinson as “a deep soul who loved his wife and two sons enormously” and “a whiskey aficionado.” “[Whiskey] suited his contemplative style,” Liew recalled. “Some of my fondest conversations with him were over a whiskey, either in person or virtually, pondering the ‘big issues.’”
A ‘friend and a colleague and so much more’
Claire Barrett, MBBS, president of the Australian Rheumatology Association, described Robinson as a “friend and a colleague and so much more. ... [He was] someone who I worked, laughed, ate, drank, danced, and had fun with,” she told this news organization. They served together as volunteers for the Australian Rheumatology Association and its Queensland branch, as well as Arthritis Queensland; they were also colleagues at Metro North Hospital and Health Service in Brisbane, Queensland.
Robinson was her “go to” for insightful comment on a variety of topics, she said. That could be advice on managing a patient with difficult gout, challenging spondyloarthritis, or the best treatment for a patient with COVID-19.
“[Phil] was a friend I could ask about anything, knowing I would not be judged,” Barrett said. “His kids and our grandkids are similar ages, so we would swap stories and photos and laugh about how cute/funny/cuddly/busy/etc. they were. My heart is broken he won’t get the chance to enjoy their future and the excitement of having Phil continuing to be such an active dad.”
Tanner, Robinson’s wife, said, “he loved everything.” That included the academic side of medicine, and working out what was wrong with his patients and helping them get better. “He was dedicated to this,” she added.
“He also loved the camaraderie of the job – all the people he met and interacted with. [Phil] loved sharing his ideas for research and also discussing complex patients with colleagues. He was driven by finding the answers to problems and doing this as part of a team of researchers/clinicians. He wasn’t interested in personal success.”
Robinson received his medical degree from Otago Medical School in Dunedin, New Zealand, according to the University of Queensland. His specialty training in general and acute care medicine and rheumatology was completed in Wellington, New Zealand, and Dunedin. Robinson also achieved a PhD in human genetics at the University of Queensland Diamantina Institute and had a postdoctoral fellowship at the Queensland Brain Institute at the University of Queensland.
Before his death, he worked at the Royal Brisbane and Women’s Hospital in Herston, Queensland, and at St. Andrew’s War Memorial Hospital in Spring Hill in Brisbane.
A version of this article first appeared on Medscape.com.
Cardiovascular risk score multipliers suggested for rheumatic diseases
A re-evaluation of cardiovascular risk management guidelines intended for use by rheumatologists may be warranted based on findings from a recently published population-based study of the risks for 12 different cardiovascular disease outcomes in patients with autoimmune diseases.
“The notion that patients with rheumatic diseases are at increased risk of developing cardiovascular diseases has been ongoing for many years,” Nathalie Conrad, PhD, and coauthors wrote in a viewpoint article in Annals of the Rheumatic Diseases.
This has “sparked much debate concerning whether and when to initiate cardiovascular prevention therapies,” they said.
Dr. Conrad was first author on the population-based study published in The Lancet in August 2022 that used linked primary and secondary care records from datasets in the U.K. Clinical Practice Research Datalink involving individuals who were recently diagnosed with any of 19 different autoimmune diseases during an 18-year period stretching from 2000 to 2017 but free of cardiovascular disease until at least 12 months after incident autoimmune disease. “Every single autoimmune disorder we looked at was associated with increased cardiovascular risk,” Dr. Conrad, of the department of public health and primary care at Catholic University Leuven (Belgium), said in an interview.
Not only was the risk for cardiovascular disease increased for people with rheumatic diseases by an average of 68%, compared with people without rheumatic diseases, but also the whole spectrum of cardiovascular disorders was seen.
“We saw increases in thromboembolic diseases, degenerative heart diseases, and heart inflammation,” Dr. Conrad said.
Large datasets examined
The idea for the epidemiologic study came from mounting evidence for cardiovascular disease risk among people with autoimmune diseases but not enough to support the design of specific prevention measures.
Dr. Conrad’s Lancet study examined electronic health records of 446,449 individuals with autoimmune diseases and matched them to 2,102,830 individuals without autoimmune disease. This included 160,217 individuals with seven rheumatic diseases: rheumatoid arthritis, polymyalgia rheumatica, vasculitis, systemic lupus erythematosus, Sjögren’s syndrome, ankylosing spondylitis, and systemic sclerosis.
In addition to looking for any evidence of cardiovascular disease, Dr. Conrad and coauthors looked at 12 specific outcomes: atherosclerotic diseases, peripheral arterial disease, stroke or transient ischemic attack, heart failure, valve disorders, thromboembolic disease, atrial fibrillation or flutter, conduction system disease, supraventricular arrhythmias, aortic aneurysm, myocarditis and pericarditis, and infective endocarditis.
CV risk in rheumatic diseases
As might be expected, “greater magnitudes of risk” were seen for individuals with systemic lupus erythematosus and systemic sclerosis than for people in the general population, with the chances of cardiovascular disease being two to four times higher. But what perhaps wasn’t expected was that all rheumatic diseases carried an increased risk for heart or vascular-related problems.
Furthermore, the increased risk could not solely be accounted for by the presence of traditional risk factors, such as blood pressure, smoking, or obesity.
“The background here is that any context of systemic inflammation would be predicted to lead to an increased vascular risk,” Iain McInnes, MD, PhD, professor of medicine and rheumatology at the University of Glasgow, said in an interview. Dr. McInnes was a coauthor of the viewpoint article in Annals of the Rheumatic Diseases.
“The implication is that there may well be increased vascular risk across the whole range of immune-mediated inflammatory diseases,” he added. “We should not, however, infer the magnitude of risk will be the same for each disease.”
What is more intriguing, Dr. McInnes said, is that “we don’t know yet whether there’s one final common pathway that leads to the blood vessel being damaged or whether different diseases might contribute different pathways.”
He added: “A question for the future is to see what are those mechanisms that drive risk across different diseases? And the reason that matters, of course, is that we might want to think about the effectiveness of different therapeutic interventions.”
Determining cardiovascular risk
Dr. Conrad and associates in their viewpoint article suggested that an update to the European Alliance of Associations for Rheumatology guidelines for cardiovascular risk management of rheumatic and musculoskeletal diseases (RMDs) could tailor cardiovascular risk scores to certain diseases.
They suggested that the guidelines could consider a risk multiplier of 2.5 for systemic sclerosis, 2.0 for lupus, and 1.5 for any other rheumatic disease.
“We argue that [EULAR] recommendations should consider this new evidence of poorer cardiovascular health in numerous RMDs and envisage cardiovascular screening and associated prevention measures,” Dr. Conrad said.
While they recognize that risk multipliers aren’t perfect, “they are the best available option until personalized risk prediction tools are developed specifically for patients with RMDs.”
Addressing cardiovascular risk
As a former president of EULAR, Dr. McInnes was keen to point out that “EULAR’s recommendations are evidence based and are rigorously built on [standard operating procedures] that work and have stood the test of time. I’m quite sure that the members of relevant EULAR task forces will be looking at these data, but they’ll be looking at the whole range of literature to see whether change is necessary.”
Good-quality inflammatory disease control will certainly contribute to reducing vascular risk, “but we should not make the assumption that it will be sufficient,” he cautioned. “We still have to be very careful in addressing so called conventional risk factors, but in particular thinking about obesity and cardiometabolic syndrome to be sure that when those are present, that we detect them and we treat them appropriately.”
As to who is best placed to manage a patient’s cardiovascular risk profile, Dr. McInnes said: “I think the rheumatologist has a responsibility to make sure that as much of the patient’s disease spectrum is being treated as possible.”
“As a rheumatologist, I would like to know that those elements of a patient’s disease presentation are being addressed,” whether that is by a primary care physician, cardiologist, diabetologist, or other specialist involved in the optimal management of the patient.
Dr. Conrad acknowledged receiving support from the European Union’s Horizon 2020 Program, the European Society for Cardiology, and grant funding paid to her institution from the Belgian-based Research Foundation Flounders. She also acknowledged receipt of royalties in regard to the intellectual property of a home-monitoring system for heart failure paid to Oxford University Innovation. Dr. McInnes acknowledged financial relationships with many pharmaceutical companies.
*This article was updated 12/30/2022.
A re-evaluation of cardiovascular risk management guidelines intended for use by rheumatologists may be warranted based on findings from a recently published population-based study of the risks for 12 different cardiovascular disease outcomes in patients with autoimmune diseases.
“The notion that patients with rheumatic diseases are at increased risk of developing cardiovascular diseases has been ongoing for many years,” Nathalie Conrad, PhD, and coauthors wrote in a viewpoint article in Annals of the Rheumatic Diseases.
This has “sparked much debate concerning whether and when to initiate cardiovascular prevention therapies,” they said.
Dr. Conrad was first author on the population-based study published in The Lancet in August 2022 that used linked primary and secondary care records from datasets in the U.K. Clinical Practice Research Datalink involving individuals who were recently diagnosed with any of 19 different autoimmune diseases during an 18-year period stretching from 2000 to 2017 but free of cardiovascular disease until at least 12 months after incident autoimmune disease. “Every single autoimmune disorder we looked at was associated with increased cardiovascular risk,” Dr. Conrad, of the department of public health and primary care at Catholic University Leuven (Belgium), said in an interview.
Not only was the risk for cardiovascular disease increased for people with rheumatic diseases by an average of 68%, compared with people without rheumatic diseases, but also the whole spectrum of cardiovascular disorders was seen.
“We saw increases in thromboembolic diseases, degenerative heart diseases, and heart inflammation,” Dr. Conrad said.
Large datasets examined
The idea for the epidemiologic study came from mounting evidence for cardiovascular disease risk among people with autoimmune diseases but not enough to support the design of specific prevention measures.
Dr. Conrad’s Lancet study examined electronic health records of 446,449 individuals with autoimmune diseases and matched them to 2,102,830 individuals without autoimmune disease. This included 160,217 individuals with seven rheumatic diseases: rheumatoid arthritis, polymyalgia rheumatica, vasculitis, systemic lupus erythematosus, Sjögren’s syndrome, ankylosing spondylitis, and systemic sclerosis.
In addition to looking for any evidence of cardiovascular disease, Dr. Conrad and coauthors looked at 12 specific outcomes: atherosclerotic diseases, peripheral arterial disease, stroke or transient ischemic attack, heart failure, valve disorders, thromboembolic disease, atrial fibrillation or flutter, conduction system disease, supraventricular arrhythmias, aortic aneurysm, myocarditis and pericarditis, and infective endocarditis.
CV risk in rheumatic diseases
As might be expected, “greater magnitudes of risk” were seen for individuals with systemic lupus erythematosus and systemic sclerosis than for people in the general population, with the chances of cardiovascular disease being two to four times higher. But what perhaps wasn’t expected was that all rheumatic diseases carried an increased risk for heart or vascular-related problems.
Furthermore, the increased risk could not solely be accounted for by the presence of traditional risk factors, such as blood pressure, smoking, or obesity.
“The background here is that any context of systemic inflammation would be predicted to lead to an increased vascular risk,” Iain McInnes, MD, PhD, professor of medicine and rheumatology at the University of Glasgow, said in an interview. Dr. McInnes was a coauthor of the viewpoint article in Annals of the Rheumatic Diseases.
“The implication is that there may well be increased vascular risk across the whole range of immune-mediated inflammatory diseases,” he added. “We should not, however, infer the magnitude of risk will be the same for each disease.”
What is more intriguing, Dr. McInnes said, is that “we don’t know yet whether there’s one final common pathway that leads to the blood vessel being damaged or whether different diseases might contribute different pathways.”
He added: “A question for the future is to see what are those mechanisms that drive risk across different diseases? And the reason that matters, of course, is that we might want to think about the effectiveness of different therapeutic interventions.”
Determining cardiovascular risk
Dr. Conrad and associates in their viewpoint article suggested that an update to the European Alliance of Associations for Rheumatology guidelines for cardiovascular risk management of rheumatic and musculoskeletal diseases (RMDs) could tailor cardiovascular risk scores to certain diseases.
They suggested that the guidelines could consider a risk multiplier of 2.5 for systemic sclerosis, 2.0 for lupus, and 1.5 for any other rheumatic disease.
“We argue that [EULAR] recommendations should consider this new evidence of poorer cardiovascular health in numerous RMDs and envisage cardiovascular screening and associated prevention measures,” Dr. Conrad said.
While they recognize that risk multipliers aren’t perfect, “they are the best available option until personalized risk prediction tools are developed specifically for patients with RMDs.”
Addressing cardiovascular risk
As a former president of EULAR, Dr. McInnes was keen to point out that “EULAR’s recommendations are evidence based and are rigorously built on [standard operating procedures] that work and have stood the test of time. I’m quite sure that the members of relevant EULAR task forces will be looking at these data, but they’ll be looking at the whole range of literature to see whether change is necessary.”
Good-quality inflammatory disease control will certainly contribute to reducing vascular risk, “but we should not make the assumption that it will be sufficient,” he cautioned. “We still have to be very careful in addressing so called conventional risk factors, but in particular thinking about obesity and cardiometabolic syndrome to be sure that when those are present, that we detect them and we treat them appropriately.”
As to who is best placed to manage a patient’s cardiovascular risk profile, Dr. McInnes said: “I think the rheumatologist has a responsibility to make sure that as much of the patient’s disease spectrum is being treated as possible.”
“As a rheumatologist, I would like to know that those elements of a patient’s disease presentation are being addressed,” whether that is by a primary care physician, cardiologist, diabetologist, or other specialist involved in the optimal management of the patient.
Dr. Conrad acknowledged receiving support from the European Union’s Horizon 2020 Program, the European Society for Cardiology, and grant funding paid to her institution from the Belgian-based Research Foundation Flounders. She also acknowledged receipt of royalties in regard to the intellectual property of a home-monitoring system for heart failure paid to Oxford University Innovation. Dr. McInnes acknowledged financial relationships with many pharmaceutical companies.
*This article was updated 12/30/2022.
A re-evaluation of cardiovascular risk management guidelines intended for use by rheumatologists may be warranted based on findings from a recently published population-based study of the risks for 12 different cardiovascular disease outcomes in patients with autoimmune diseases.
“The notion that patients with rheumatic diseases are at increased risk of developing cardiovascular diseases has been ongoing for many years,” Nathalie Conrad, PhD, and coauthors wrote in a viewpoint article in Annals of the Rheumatic Diseases.
This has “sparked much debate concerning whether and when to initiate cardiovascular prevention therapies,” they said.
Dr. Conrad was first author on the population-based study published in The Lancet in August 2022 that used linked primary and secondary care records from datasets in the U.K. Clinical Practice Research Datalink involving individuals who were recently diagnosed with any of 19 different autoimmune diseases during an 18-year period stretching from 2000 to 2017 but free of cardiovascular disease until at least 12 months after incident autoimmune disease. “Every single autoimmune disorder we looked at was associated with increased cardiovascular risk,” Dr. Conrad, of the department of public health and primary care at Catholic University Leuven (Belgium), said in an interview.
Not only was the risk for cardiovascular disease increased for people with rheumatic diseases by an average of 68%, compared with people without rheumatic diseases, but also the whole spectrum of cardiovascular disorders was seen.
“We saw increases in thromboembolic diseases, degenerative heart diseases, and heart inflammation,” Dr. Conrad said.
Large datasets examined
The idea for the epidemiologic study came from mounting evidence for cardiovascular disease risk among people with autoimmune diseases but not enough to support the design of specific prevention measures.
Dr. Conrad’s Lancet study examined electronic health records of 446,449 individuals with autoimmune diseases and matched them to 2,102,830 individuals without autoimmune disease. This included 160,217 individuals with seven rheumatic diseases: rheumatoid arthritis, polymyalgia rheumatica, vasculitis, systemic lupus erythematosus, Sjögren’s syndrome, ankylosing spondylitis, and systemic sclerosis.
In addition to looking for any evidence of cardiovascular disease, Dr. Conrad and coauthors looked at 12 specific outcomes: atherosclerotic diseases, peripheral arterial disease, stroke or transient ischemic attack, heart failure, valve disorders, thromboembolic disease, atrial fibrillation or flutter, conduction system disease, supraventricular arrhythmias, aortic aneurysm, myocarditis and pericarditis, and infective endocarditis.
CV risk in rheumatic diseases
As might be expected, “greater magnitudes of risk” were seen for individuals with systemic lupus erythematosus and systemic sclerosis than for people in the general population, with the chances of cardiovascular disease being two to four times higher. But what perhaps wasn’t expected was that all rheumatic diseases carried an increased risk for heart or vascular-related problems.
Furthermore, the increased risk could not solely be accounted for by the presence of traditional risk factors, such as blood pressure, smoking, or obesity.
“The background here is that any context of systemic inflammation would be predicted to lead to an increased vascular risk,” Iain McInnes, MD, PhD, professor of medicine and rheumatology at the University of Glasgow, said in an interview. Dr. McInnes was a coauthor of the viewpoint article in Annals of the Rheumatic Diseases.
“The implication is that there may well be increased vascular risk across the whole range of immune-mediated inflammatory diseases,” he added. “We should not, however, infer the magnitude of risk will be the same for each disease.”
What is more intriguing, Dr. McInnes said, is that “we don’t know yet whether there’s one final common pathway that leads to the blood vessel being damaged or whether different diseases might contribute different pathways.”
He added: “A question for the future is to see what are those mechanisms that drive risk across different diseases? And the reason that matters, of course, is that we might want to think about the effectiveness of different therapeutic interventions.”
Determining cardiovascular risk
Dr. Conrad and associates in their viewpoint article suggested that an update to the European Alliance of Associations for Rheumatology guidelines for cardiovascular risk management of rheumatic and musculoskeletal diseases (RMDs) could tailor cardiovascular risk scores to certain diseases.
They suggested that the guidelines could consider a risk multiplier of 2.5 for systemic sclerosis, 2.0 for lupus, and 1.5 for any other rheumatic disease.
“We argue that [EULAR] recommendations should consider this new evidence of poorer cardiovascular health in numerous RMDs and envisage cardiovascular screening and associated prevention measures,” Dr. Conrad said.
While they recognize that risk multipliers aren’t perfect, “they are the best available option until personalized risk prediction tools are developed specifically for patients with RMDs.”
Addressing cardiovascular risk
As a former president of EULAR, Dr. McInnes was keen to point out that “EULAR’s recommendations are evidence based and are rigorously built on [standard operating procedures] that work and have stood the test of time. I’m quite sure that the members of relevant EULAR task forces will be looking at these data, but they’ll be looking at the whole range of literature to see whether change is necessary.”
Good-quality inflammatory disease control will certainly contribute to reducing vascular risk, “but we should not make the assumption that it will be sufficient,” he cautioned. “We still have to be very careful in addressing so called conventional risk factors, but in particular thinking about obesity and cardiometabolic syndrome to be sure that when those are present, that we detect them and we treat them appropriately.”
As to who is best placed to manage a patient’s cardiovascular risk profile, Dr. McInnes said: “I think the rheumatologist has a responsibility to make sure that as much of the patient’s disease spectrum is being treated as possible.”
“As a rheumatologist, I would like to know that those elements of a patient’s disease presentation are being addressed,” whether that is by a primary care physician, cardiologist, diabetologist, or other specialist involved in the optimal management of the patient.
Dr. Conrad acknowledged receiving support from the European Union’s Horizon 2020 Program, the European Society for Cardiology, and grant funding paid to her institution from the Belgian-based Research Foundation Flounders. She also acknowledged receipt of royalties in regard to the intellectual property of a home-monitoring system for heart failure paid to Oxford University Innovation. Dr. McInnes acknowledged financial relationships with many pharmaceutical companies.
*This article was updated 12/30/2022.
FROM ANNALS OF THE RHEUMATIC DISEASES
FDA approves Idacio as eighth adalimumab biosimilar in U.S.
A biosimilar drug to the tumor necrosis factor inhibitor adalimumab, marketed as Idacio (adalimumab-aacf), has been approved by the Food and Drug Administration for use in the United States, according to a press release from manufacturer Fresenius Kabi.
Idacio is a citrate-free, low-concentration formulation of adalimumab and is now approved for use for all but three of the indications that currently apply to the reference adalimumab product (Humira): rheumatoid arthritis, polyarticular juvenile idiopathic arthritis, psoriatic arthritis in adults, ankylosing spondylitis, Crohn’s disease in adults and children aged 6 years or older, ulcerative colitis in adults, and plaque psoriasis in adults. It does not apply to Humira’s indications for hidradenitis suppurativa, uveitis, or ulcerative colitis in pediatric patients aged 5 years and older.
Idacio is the eighth adalimumab biosimilar to be approved in the United States. Its approval was based on evidence of a similar profile of pharmacokinetics, safety, efficacy, and immunogenicity to Humira.
Idacio was first launched in 2019 and has been marketed in more than 37 countries worldwide, according to Fresenius Kabi. The U.S. launch is scheduled for July, and Idacio will be available as a self-administered prefilled syringe or prefilled pen.
A version of this article first appeared on Medscape.com.
A biosimilar drug to the tumor necrosis factor inhibitor adalimumab, marketed as Idacio (adalimumab-aacf), has been approved by the Food and Drug Administration for use in the United States, according to a press release from manufacturer Fresenius Kabi.
Idacio is a citrate-free, low-concentration formulation of adalimumab and is now approved for use for all but three of the indications that currently apply to the reference adalimumab product (Humira): rheumatoid arthritis, polyarticular juvenile idiopathic arthritis, psoriatic arthritis in adults, ankylosing spondylitis, Crohn’s disease in adults and children aged 6 years or older, ulcerative colitis in adults, and plaque psoriasis in adults. It does not apply to Humira’s indications for hidradenitis suppurativa, uveitis, or ulcerative colitis in pediatric patients aged 5 years and older.
Idacio is the eighth adalimumab biosimilar to be approved in the United States. Its approval was based on evidence of a similar profile of pharmacokinetics, safety, efficacy, and immunogenicity to Humira.
Idacio was first launched in 2019 and has been marketed in more than 37 countries worldwide, according to Fresenius Kabi. The U.S. launch is scheduled for July, and Idacio will be available as a self-administered prefilled syringe or prefilled pen.
A version of this article first appeared on Medscape.com.
A biosimilar drug to the tumor necrosis factor inhibitor adalimumab, marketed as Idacio (adalimumab-aacf), has been approved by the Food and Drug Administration for use in the United States, according to a press release from manufacturer Fresenius Kabi.
Idacio is a citrate-free, low-concentration formulation of adalimumab and is now approved for use for all but three of the indications that currently apply to the reference adalimumab product (Humira): rheumatoid arthritis, polyarticular juvenile idiopathic arthritis, psoriatic arthritis in adults, ankylosing spondylitis, Crohn’s disease in adults and children aged 6 years or older, ulcerative colitis in adults, and plaque psoriasis in adults. It does not apply to Humira’s indications for hidradenitis suppurativa, uveitis, or ulcerative colitis in pediatric patients aged 5 years and older.
Idacio is the eighth adalimumab biosimilar to be approved in the United States. Its approval was based on evidence of a similar profile of pharmacokinetics, safety, efficacy, and immunogenicity to Humira.
Idacio was first launched in 2019 and has been marketed in more than 37 countries worldwide, according to Fresenius Kabi. The U.S. launch is scheduled for July, and Idacio will be available as a self-administered prefilled syringe or prefilled pen.
A version of this article first appeared on Medscape.com.
Vaccination cuts long COVID risk for rheumatic disease patients
Patients with rheumatic disease are at least half as likely to develop long COVID after a SARS-CoV-2 infection if they have been fully vaccinated against COVID-19, according to research published in Annals of the Rheumatic Diseases (2022 Nov 28. doi: 10.1136/ard-2022-223439).
“Moreover, those who were vaccinated prior to getting COVID-19 had less pain and fatigue after their infection,” Zachary S. Wallace, MD, MSc, an assistant professor of medicine at Harvard Medical School, Boston, and a study author, said in an interview. “These findings reinforce the importance of vaccination in this population.”
Messaging around the value of COVID vaccination has been confusing for some with rheumatic disease “because our concern regarding a blunted response to vaccination has led many patients to think that they do not provide much benefit if they are on immunosuppression,” Dr. Wallace said. “In our cohort, which included many patients on immunosuppression of varying degrees, being vaccinated was quite beneficial.”
Leonard H. Calabrese, DO, director of the R.J. Fasenmyer Center for Clinical Immunology and a professor of medicine at the Cleveland Clinic, said in an interview that the study is an “extremely important contribution to our understanding of COVID-19 and its pattern of recovery in patients with immune-mediated inflammatory diseases [IMIDs].” Remaining unanswered questions are “whether patients with IMIDs develop more frequent PASC [post–acute sequelae of COVID-19] from COVID-19 and, if so, is it milder or more severe, and does it differ in its clinical phenotype?”
Long COVID risk assessed at 4 weeks and 3 months after infection
The researchers prospectively tracked 280 adult patients in the Mass General Brigham health care system in the greater Boston area who had systemic autoimmune rheumatic diseases and had an acute COVID-19 infection between March 2020 and July 2022. Patients were an average 53 years old, and most were White (82%) and female (80%). More than half (59%) had inflammatory arthritis, a quarter (24%) had connective tissue disease, and most others had a vasculitis condition or multiple conditions.
A total of 11% of patients were unvaccinated, 28% were partially vaccinated with one mRNA COVID-19 vaccine dose, and 41% were fully vaccinated with two mRNA vaccine doses or one Johnson & Johnson dose. The 116 fully vaccinated patients were considered to have a breakthrough infection while the other 164 were considered to have a nonbreakthrough infection. The breakthrough and nonbreakthrough groups were similar in terms of age, sex, race, ethnicity, smoking status, and type of rheumatic disease. Comorbidities were also similar, except obesity, which was more common in the non–breakthrough infection group (25%) than the breakthrough infection group (10%).
The researchers queried patients on their COVID-19 symptoms, how long symptoms lasted, treatments they received, and hospitalization details. COVID-19 symptoms assessed included fever, sore throat, new cough, nasal congestion/rhinorrhea, dyspnea, chest pain, rash, myalgia, fatigue/malaise, headache, nausea/vomiting, diarrhea, anosmia, dysgeusia, and joint pain.
Patients completed surveys about symptoms at 4 weeks and 3 months after infection. Long COVID, or PASC, was defined as any persistent symptom at the times assessed.
Vaccinated patients fared better across outcomes
At 4 weeks after infection, 41% of fully vaccinated patients had at least one persistent symptom, compared with 54% of unvaccinated or partially vaccinated patients (P = .04). At 3 months after infection, 21% of fully vaccinated patients had at least one persistent symptom, compared with 41% of unvaccinated or partially vaccinated patients (P < .0001).
Vaccinated patients were half as likely to have long COVID at 4 weeks after infection (adjusted odds ratio, 0.49) and 90% less likely to have long COVID 3 months after infection (aOR, 0.1), after adjustment for age, sex, race, comorbidities, and use of any of four immune-suppressing medications (anti-CD20 monoclonal antibodies, methotrexate, mycophenolate, or glucocorticoids).
Fully vaccinated patients with breakthrough infections had an average 21 additional days without symptoms during follow-up, compared with unvaccinated and partially vaccinated patients (P = .04).
Reduced risk of long COVID did not change for vaccinated patients after sensitivity analyses for those who did not receive nirmatrelvir/ritonavir (Paxlovid) or monoclonal antibodies, those who didn’t receive any COVID-19-related treatment, those who completed their questionnaires within 6 months after infection, and those who were not hospitalized.
“One important message is that among those who did get PASC, the severity appears similar among those with and without a breakthrough infection,” Dr. Wallace said. “This highlights the need for ongoing research to improve recognition, diagnosis, and treatment of PASC.”
Many more breakthrough infections (72%) than nonbreakthrough infections (2%) occurred during Omicron. The authors acknowledged that different variants might play a role in different long COVID risks but said such potential confounding is unlikely to fully explain the results.
“Even with data suggesting that the Omicron variants may be intrinsically less severe, vaccination still has an impact on severity of infection, rates of hospitalization, and other outcomes and thus may play a role in the risk of PASC,” lead author Naomi Patel, MD, an instructor at Harvard Medical School and a rheumatologist at Massachusetts General Hospital, said in an interview. “A study evaluating the proportions with PASC by vaccination status during the time in which a single variant is predominant, such as the early Omicron era, could help to better assess the more isolated impact of vaccination on PASC.”
Dr. Calabrese said he is convinced that Omicron infections are less likely to result in more severe forms of acute COVID than pre-Omicron infections, and he suspects Omicron infections are also less likely to result in long COVID, although less evidence currently supports this hypothesis.
Hospitalization was more common in unvaccinated/partly vaccinated patients than in vaccinated patients (27% vs. 5%; P = .001). Although pain and fatigue were lower in those with breakthrough infections, functional scores and health-related quality of life were similar in both groups.
Some symptoms significantly differed between vaccinated and unvaccinated/partly vaccinated groups, possibly caused partly by different variants. Nasal congestion was more common (73%) in those with breakthrough infections than in those with nonbreakthrough infections (46%; P < .0001). Those who were unvaccinated/partly vaccinated were significantly more likely to have loss of smell (46% vs. 22%) or taste (45% vs. 28%) or to have joint pain (11% vs. 4%).
Treatment with nirmatrelvir/ritonavir was also more common in vaccinated patients (12%) than in unvaccinated/partly vaccinated patients (1%; P < .0001), as was treatment with monoclonal antibodies (34% vs. 8%; P < .0001).
The study was limited by its low diversity and being at a single health care system, the authors said. Study coauthor Jeffrey A. Sparks, MD, MMSc, an assistant professor of medicine at Brigham and Women’s Hospital and Harvard Medical School, said in an interview that the group is planning additional studies as their cohort grows, including “investigating the relationships between COVID-19 and specific rheumatic diseases and immunomodulating medications, expansion of autoimmunity and systemic inflammation, and lung damage among specific patient populations.”
Dr. Calabrese said it will be important for follow-up study of the symptomatic patients to “determine how many of these patients will fit the clinical picture of long COVID or long-haul phenotypes over the months and years ahead, including documenting exertional malaise and quality of life.
This study only assessed patients who received zero, one, or two doses of a vaccine, but many patients with rheumatic disease today will likely have received booster doses. However, Dr. Calabrese said it would be difficult to quantify whether a third, fourth, or fifth dose offers additional protection from long-term COVID complications after full vaccination or hybrid vaccination.
The research was funded by the Rheumatology Research Foundation, the National Institutes of Health, the R. Bruce and Joan M. Mickey Research Scholar Fund, and the Llura Gund Award for Rheumatoid Arthritis Research and Care. Dr. Wallace has received research support from Bristol-Myers Squibb and Principia/Sanofi and consulting fees from Zenas BioPharma, Horizon, Sanofi, Shionogi, Viela Bio, and Medpace. Dr. Sparks has received research support from Bristol-Myers Squibb and consulting fees from AbbVie, Amgen, Boehringer Ingelheim, Bristol-Myers Squibb, Gilead, Inova Diagnostics, Janssen, Optum, and Pfizer. Dr. Patel has received consulting fees from FVC Health. Calabrese has consulted for Genentech, Sanofi-Regeneron, AstraZeneca, and GlaxoSmithKline.
A version of this article first appeared on Medscape.com.
Patients with rheumatic disease are at least half as likely to develop long COVID after a SARS-CoV-2 infection if they have been fully vaccinated against COVID-19, according to research published in Annals of the Rheumatic Diseases (2022 Nov 28. doi: 10.1136/ard-2022-223439).
“Moreover, those who were vaccinated prior to getting COVID-19 had less pain and fatigue after their infection,” Zachary S. Wallace, MD, MSc, an assistant professor of medicine at Harvard Medical School, Boston, and a study author, said in an interview. “These findings reinforce the importance of vaccination in this population.”
Messaging around the value of COVID vaccination has been confusing for some with rheumatic disease “because our concern regarding a blunted response to vaccination has led many patients to think that they do not provide much benefit if they are on immunosuppression,” Dr. Wallace said. “In our cohort, which included many patients on immunosuppression of varying degrees, being vaccinated was quite beneficial.”
Leonard H. Calabrese, DO, director of the R.J. Fasenmyer Center for Clinical Immunology and a professor of medicine at the Cleveland Clinic, said in an interview that the study is an “extremely important contribution to our understanding of COVID-19 and its pattern of recovery in patients with immune-mediated inflammatory diseases [IMIDs].” Remaining unanswered questions are “whether patients with IMIDs develop more frequent PASC [post–acute sequelae of COVID-19] from COVID-19 and, if so, is it milder or more severe, and does it differ in its clinical phenotype?”
Long COVID risk assessed at 4 weeks and 3 months after infection
The researchers prospectively tracked 280 adult patients in the Mass General Brigham health care system in the greater Boston area who had systemic autoimmune rheumatic diseases and had an acute COVID-19 infection between March 2020 and July 2022. Patients were an average 53 years old, and most were White (82%) and female (80%). More than half (59%) had inflammatory arthritis, a quarter (24%) had connective tissue disease, and most others had a vasculitis condition or multiple conditions.
A total of 11% of patients were unvaccinated, 28% were partially vaccinated with one mRNA COVID-19 vaccine dose, and 41% were fully vaccinated with two mRNA vaccine doses or one Johnson & Johnson dose. The 116 fully vaccinated patients were considered to have a breakthrough infection while the other 164 were considered to have a nonbreakthrough infection. The breakthrough and nonbreakthrough groups were similar in terms of age, sex, race, ethnicity, smoking status, and type of rheumatic disease. Comorbidities were also similar, except obesity, which was more common in the non–breakthrough infection group (25%) than the breakthrough infection group (10%).
The researchers queried patients on their COVID-19 symptoms, how long symptoms lasted, treatments they received, and hospitalization details. COVID-19 symptoms assessed included fever, sore throat, new cough, nasal congestion/rhinorrhea, dyspnea, chest pain, rash, myalgia, fatigue/malaise, headache, nausea/vomiting, diarrhea, anosmia, dysgeusia, and joint pain.
Patients completed surveys about symptoms at 4 weeks and 3 months after infection. Long COVID, or PASC, was defined as any persistent symptom at the times assessed.
Vaccinated patients fared better across outcomes
At 4 weeks after infection, 41% of fully vaccinated patients had at least one persistent symptom, compared with 54% of unvaccinated or partially vaccinated patients (P = .04). At 3 months after infection, 21% of fully vaccinated patients had at least one persistent symptom, compared with 41% of unvaccinated or partially vaccinated patients (P < .0001).
Vaccinated patients were half as likely to have long COVID at 4 weeks after infection (adjusted odds ratio, 0.49) and 90% less likely to have long COVID 3 months after infection (aOR, 0.1), after adjustment for age, sex, race, comorbidities, and use of any of four immune-suppressing medications (anti-CD20 monoclonal antibodies, methotrexate, mycophenolate, or glucocorticoids).
Fully vaccinated patients with breakthrough infections had an average 21 additional days without symptoms during follow-up, compared with unvaccinated and partially vaccinated patients (P = .04).
Reduced risk of long COVID did not change for vaccinated patients after sensitivity analyses for those who did not receive nirmatrelvir/ritonavir (Paxlovid) or monoclonal antibodies, those who didn’t receive any COVID-19-related treatment, those who completed their questionnaires within 6 months after infection, and those who were not hospitalized.
“One important message is that among those who did get PASC, the severity appears similar among those with and without a breakthrough infection,” Dr. Wallace said. “This highlights the need for ongoing research to improve recognition, diagnosis, and treatment of PASC.”
Many more breakthrough infections (72%) than nonbreakthrough infections (2%) occurred during Omicron. The authors acknowledged that different variants might play a role in different long COVID risks but said such potential confounding is unlikely to fully explain the results.
“Even with data suggesting that the Omicron variants may be intrinsically less severe, vaccination still has an impact on severity of infection, rates of hospitalization, and other outcomes and thus may play a role in the risk of PASC,” lead author Naomi Patel, MD, an instructor at Harvard Medical School and a rheumatologist at Massachusetts General Hospital, said in an interview. “A study evaluating the proportions with PASC by vaccination status during the time in which a single variant is predominant, such as the early Omicron era, could help to better assess the more isolated impact of vaccination on PASC.”
Dr. Calabrese said he is convinced that Omicron infections are less likely to result in more severe forms of acute COVID than pre-Omicron infections, and he suspects Omicron infections are also less likely to result in long COVID, although less evidence currently supports this hypothesis.
Hospitalization was more common in unvaccinated/partly vaccinated patients than in vaccinated patients (27% vs. 5%; P = .001). Although pain and fatigue were lower in those with breakthrough infections, functional scores and health-related quality of life were similar in both groups.
Some symptoms significantly differed between vaccinated and unvaccinated/partly vaccinated groups, possibly caused partly by different variants. Nasal congestion was more common (73%) in those with breakthrough infections than in those with nonbreakthrough infections (46%; P < .0001). Those who were unvaccinated/partly vaccinated were significantly more likely to have loss of smell (46% vs. 22%) or taste (45% vs. 28%) or to have joint pain (11% vs. 4%).
Treatment with nirmatrelvir/ritonavir was also more common in vaccinated patients (12%) than in unvaccinated/partly vaccinated patients (1%; P < .0001), as was treatment with monoclonal antibodies (34% vs. 8%; P < .0001).
The study was limited by its low diversity and being at a single health care system, the authors said. Study coauthor Jeffrey A. Sparks, MD, MMSc, an assistant professor of medicine at Brigham and Women’s Hospital and Harvard Medical School, said in an interview that the group is planning additional studies as their cohort grows, including “investigating the relationships between COVID-19 and specific rheumatic diseases and immunomodulating medications, expansion of autoimmunity and systemic inflammation, and lung damage among specific patient populations.”
Dr. Calabrese said it will be important for follow-up study of the symptomatic patients to “determine how many of these patients will fit the clinical picture of long COVID or long-haul phenotypes over the months and years ahead, including documenting exertional malaise and quality of life.
This study only assessed patients who received zero, one, or two doses of a vaccine, but many patients with rheumatic disease today will likely have received booster doses. However, Dr. Calabrese said it would be difficult to quantify whether a third, fourth, or fifth dose offers additional protection from long-term COVID complications after full vaccination or hybrid vaccination.
The research was funded by the Rheumatology Research Foundation, the National Institutes of Health, the R. Bruce and Joan M. Mickey Research Scholar Fund, and the Llura Gund Award for Rheumatoid Arthritis Research and Care. Dr. Wallace has received research support from Bristol-Myers Squibb and Principia/Sanofi and consulting fees from Zenas BioPharma, Horizon, Sanofi, Shionogi, Viela Bio, and Medpace. Dr. Sparks has received research support from Bristol-Myers Squibb and consulting fees from AbbVie, Amgen, Boehringer Ingelheim, Bristol-Myers Squibb, Gilead, Inova Diagnostics, Janssen, Optum, and Pfizer. Dr. Patel has received consulting fees from FVC Health. Calabrese has consulted for Genentech, Sanofi-Regeneron, AstraZeneca, and GlaxoSmithKline.
A version of this article first appeared on Medscape.com.
Patients with rheumatic disease are at least half as likely to develop long COVID after a SARS-CoV-2 infection if they have been fully vaccinated against COVID-19, according to research published in Annals of the Rheumatic Diseases (2022 Nov 28. doi: 10.1136/ard-2022-223439).
“Moreover, those who were vaccinated prior to getting COVID-19 had less pain and fatigue after their infection,” Zachary S. Wallace, MD, MSc, an assistant professor of medicine at Harvard Medical School, Boston, and a study author, said in an interview. “These findings reinforce the importance of vaccination in this population.”
Messaging around the value of COVID vaccination has been confusing for some with rheumatic disease “because our concern regarding a blunted response to vaccination has led many patients to think that they do not provide much benefit if they are on immunosuppression,” Dr. Wallace said. “In our cohort, which included many patients on immunosuppression of varying degrees, being vaccinated was quite beneficial.”
Leonard H. Calabrese, DO, director of the R.J. Fasenmyer Center for Clinical Immunology and a professor of medicine at the Cleveland Clinic, said in an interview that the study is an “extremely important contribution to our understanding of COVID-19 and its pattern of recovery in patients with immune-mediated inflammatory diseases [IMIDs].” Remaining unanswered questions are “whether patients with IMIDs develop more frequent PASC [post–acute sequelae of COVID-19] from COVID-19 and, if so, is it milder or more severe, and does it differ in its clinical phenotype?”
Long COVID risk assessed at 4 weeks and 3 months after infection
The researchers prospectively tracked 280 adult patients in the Mass General Brigham health care system in the greater Boston area who had systemic autoimmune rheumatic diseases and had an acute COVID-19 infection between March 2020 and July 2022. Patients were an average 53 years old, and most were White (82%) and female (80%). More than half (59%) had inflammatory arthritis, a quarter (24%) had connective tissue disease, and most others had a vasculitis condition or multiple conditions.
A total of 11% of patients were unvaccinated, 28% were partially vaccinated with one mRNA COVID-19 vaccine dose, and 41% were fully vaccinated with two mRNA vaccine doses or one Johnson & Johnson dose. The 116 fully vaccinated patients were considered to have a breakthrough infection while the other 164 were considered to have a nonbreakthrough infection. The breakthrough and nonbreakthrough groups were similar in terms of age, sex, race, ethnicity, smoking status, and type of rheumatic disease. Comorbidities were also similar, except obesity, which was more common in the non–breakthrough infection group (25%) than the breakthrough infection group (10%).
The researchers queried patients on their COVID-19 symptoms, how long symptoms lasted, treatments they received, and hospitalization details. COVID-19 symptoms assessed included fever, sore throat, new cough, nasal congestion/rhinorrhea, dyspnea, chest pain, rash, myalgia, fatigue/malaise, headache, nausea/vomiting, diarrhea, anosmia, dysgeusia, and joint pain.
Patients completed surveys about symptoms at 4 weeks and 3 months after infection. Long COVID, or PASC, was defined as any persistent symptom at the times assessed.
Vaccinated patients fared better across outcomes
At 4 weeks after infection, 41% of fully vaccinated patients had at least one persistent symptom, compared with 54% of unvaccinated or partially vaccinated patients (P = .04). At 3 months after infection, 21% of fully vaccinated patients had at least one persistent symptom, compared with 41% of unvaccinated or partially vaccinated patients (P < .0001).
Vaccinated patients were half as likely to have long COVID at 4 weeks after infection (adjusted odds ratio, 0.49) and 90% less likely to have long COVID 3 months after infection (aOR, 0.1), after adjustment for age, sex, race, comorbidities, and use of any of four immune-suppressing medications (anti-CD20 monoclonal antibodies, methotrexate, mycophenolate, or glucocorticoids).
Fully vaccinated patients with breakthrough infections had an average 21 additional days without symptoms during follow-up, compared with unvaccinated and partially vaccinated patients (P = .04).
Reduced risk of long COVID did not change for vaccinated patients after sensitivity analyses for those who did not receive nirmatrelvir/ritonavir (Paxlovid) or monoclonal antibodies, those who didn’t receive any COVID-19-related treatment, those who completed their questionnaires within 6 months after infection, and those who were not hospitalized.
“One important message is that among those who did get PASC, the severity appears similar among those with and without a breakthrough infection,” Dr. Wallace said. “This highlights the need for ongoing research to improve recognition, diagnosis, and treatment of PASC.”
Many more breakthrough infections (72%) than nonbreakthrough infections (2%) occurred during Omicron. The authors acknowledged that different variants might play a role in different long COVID risks but said such potential confounding is unlikely to fully explain the results.
“Even with data suggesting that the Omicron variants may be intrinsically less severe, vaccination still has an impact on severity of infection, rates of hospitalization, and other outcomes and thus may play a role in the risk of PASC,” lead author Naomi Patel, MD, an instructor at Harvard Medical School and a rheumatologist at Massachusetts General Hospital, said in an interview. “A study evaluating the proportions with PASC by vaccination status during the time in which a single variant is predominant, such as the early Omicron era, could help to better assess the more isolated impact of vaccination on PASC.”
Dr. Calabrese said he is convinced that Omicron infections are less likely to result in more severe forms of acute COVID than pre-Omicron infections, and he suspects Omicron infections are also less likely to result in long COVID, although less evidence currently supports this hypothesis.
Hospitalization was more common in unvaccinated/partly vaccinated patients than in vaccinated patients (27% vs. 5%; P = .001). Although pain and fatigue were lower in those with breakthrough infections, functional scores and health-related quality of life were similar in both groups.
Some symptoms significantly differed between vaccinated and unvaccinated/partly vaccinated groups, possibly caused partly by different variants. Nasal congestion was more common (73%) in those with breakthrough infections than in those with nonbreakthrough infections (46%; P < .0001). Those who were unvaccinated/partly vaccinated were significantly more likely to have loss of smell (46% vs. 22%) or taste (45% vs. 28%) or to have joint pain (11% vs. 4%).
Treatment with nirmatrelvir/ritonavir was also more common in vaccinated patients (12%) than in unvaccinated/partly vaccinated patients (1%; P < .0001), as was treatment with monoclonal antibodies (34% vs. 8%; P < .0001).
The study was limited by its low diversity and being at a single health care system, the authors said. Study coauthor Jeffrey A. Sparks, MD, MMSc, an assistant professor of medicine at Brigham and Women’s Hospital and Harvard Medical School, said in an interview that the group is planning additional studies as their cohort grows, including “investigating the relationships between COVID-19 and specific rheumatic diseases and immunomodulating medications, expansion of autoimmunity and systemic inflammation, and lung damage among specific patient populations.”
Dr. Calabrese said it will be important for follow-up study of the symptomatic patients to “determine how many of these patients will fit the clinical picture of long COVID or long-haul phenotypes over the months and years ahead, including documenting exertional malaise and quality of life.
This study only assessed patients who received zero, one, or two doses of a vaccine, but many patients with rheumatic disease today will likely have received booster doses. However, Dr. Calabrese said it would be difficult to quantify whether a third, fourth, or fifth dose offers additional protection from long-term COVID complications after full vaccination or hybrid vaccination.
The research was funded by the Rheumatology Research Foundation, the National Institutes of Health, the R. Bruce and Joan M. Mickey Research Scholar Fund, and the Llura Gund Award for Rheumatoid Arthritis Research and Care. Dr. Wallace has received research support from Bristol-Myers Squibb and Principia/Sanofi and consulting fees from Zenas BioPharma, Horizon, Sanofi, Shionogi, Viela Bio, and Medpace. Dr. Sparks has received research support from Bristol-Myers Squibb and consulting fees from AbbVie, Amgen, Boehringer Ingelheim, Bristol-Myers Squibb, Gilead, Inova Diagnostics, Janssen, Optum, and Pfizer. Dr. Patel has received consulting fees from FVC Health. Calabrese has consulted for Genentech, Sanofi-Regeneron, AstraZeneca, and GlaxoSmithKline.
A version of this article first appeared on Medscape.com.
FROM ANNALS OF THE RHEUMATIC DISEASES
Skinny-label biosimilars provide substantial savings to Medicare
Recent court rulings could put such saving under threat
Competition between five biologic drugs and their skinny-label biosimilars saved Medicare an estimated $1.5 billion during 2015-2020. But these savings accruing to Medicare and the availability of those and other biosimilars through skinny labeling is under threat from recent court rulings, according to a research letter published online in JAMA Internal Medicine.
The authors highlighted the need for such savings by noting that, while biologics comprise less than 5% of prescription drug use, their price tag amounts to about 40% of U.S. drug spending, Biologic manufacturers often delay the availability of biosimilars for additional years beyond the original patent expiration through further patents for supplemental indications. To provide a counterbalance, federal law allows the Food and Drug Administration to approve “skinny-label” generics and biosimilars that carve out patent-protected indications or regulatory exclusivities. But once a generic drug reaches the market through this process with a skinny label, it may often be substituted for indications that go beyond the ones listed on the skinny label. In fact, some state laws mandate that pharmacists substitute interchangeable generics for brand-name drugs, helping to decrease drug prices. In response to legal threats to the skinny-label pathway, Alexander C. Egilman and colleagues at Brigham and Women’s Hospital and Harvard Medical School, both in Boston, assessed the frequency of approval and marketing of skinny-label biosimilars from 2015 to 2021 and the resultant savings to Medicare.
The authors estimated annual Part B (clinician-administered) savings from skinny-label biosimilars through 2020 by comparing actual biologic and skinny-label biosimilar spending with estimated biologic spending without competition using the Medicare Dashboard. They assumed that the unit price of the biologic would increase at its 5-year compound annual growth rate prior to competition.
In that period, the FDA approved 33 biosimilars linked to 11 biologics. Among them, 22 (66.7%) had a skinny label. Of 21 biosimilars marketed before 2022, 13 (61.9%) were launched with a skinny label. Of the 8 biologics linked to these 21 biosimilars, 5 of the first-to-market biosimilars had skinny labels (bevacizumab, filgrastim, infliximab, pegfilgrastim, and rituximab), leading to earlier competition through 2021.
The estimated $1.5 billion in savings to Medicare from these skinny-label biosimilars over the 2015-2020 span represents 4.9% of the $30.2 billion that Medicare spent on the five biologics during this period. The researchers pointed out that once adalimumab (Humira) faces skinny-label biosimilar competition in 2023, savings will likely grow substantially.
In response to the research letter, an editor’s note by JAMA Internal Medicine Editorial Fellow Eric Ward, MD, and JAMA Internal Medicine Editor at Large and Online Editor Robert Steinbrook, MD, stated that, between 2015 and 2019, 24 (43%) of 56 brand-name drugs had competition from skinny-labeled generic formulations after first becoming available as generics.
The editors also referenced a JAMA Viewpoints article from 2021 that reviewed the most recent case challenging the skinny-label pathway in which GlaxoSmithKline sued Teva for its marketing of a skinny-label generic of the brand-name beta-blocker carvedilol (Coreg) that the plaintive claimed “induced physicians to prescribe carvedilol for indications that had been carved out by Teva’s skinny label, thus infringing GlaxoSmithKline’s patents.” A $235 million judgment against Teva was overturned by a district court and then reversed again by a Federal Circuit court that, after receiving criticism, reconsidered the case, and a panel affirmed the judgment against Teva.
“The Federal Circuit panel’s decision has the potential to put generic drugs that fail to adequately carve out indications from the brand name labeling at risk for damages related to infringement,” the authors wrote. Similar claims of infringement are being heard in other courts, they wrote, and they urged careful targeting of skinny-label carveouts, and suggest also that challenges to the arguments used against Teva focus on preservation of First Amendment rights as protection for lawful and accurate speech in drug labels.
“The legal uncertainties are likely to continue, as manufacturers pursue novel and complex strategies to protect the patents and regulatory exclusivities of brand-name drugs and biologics,” Dr. Ward and Dr. Steinbrook wrote, adding that “the path forward is for Congress to enact additional legislation that reaffirms and strengthens the permissibility of skinny labeling.”
The research letter’s corresponding author, Ameet Sarpatwari, PhD, JD, assistant professor at Harvard Medical School, and assistant director for the Harvard Program On Regulation, Therapeutics, And Law, echoed concerns over the Teva case in an interview. “There has certainly been concern that should the appellate decision stand, there will be a chilling effect. As the lone dissenter in that case noted, ‘no skinny-label generic is safe.’ I think many generic and biosimilar manufacturers are awaiting to see whether the Supreme Court will take the case.”
He added: “I do not believe the likelihood of skinny-label-supportive legislation making it through Congress will be greatly diminished in a divided Congress. Democrats and Republicans alike should seek to promote competition in the marketplace, which is what the skinny-labeling pathway accomplishes.”
The authors reported no relevant conflicts of interest. The research was funded by a grant from Arnold Ventures.
Recent court rulings could put such saving under threat
Recent court rulings could put such saving under threat
Competition between five biologic drugs and their skinny-label biosimilars saved Medicare an estimated $1.5 billion during 2015-2020. But these savings accruing to Medicare and the availability of those and other biosimilars through skinny labeling is under threat from recent court rulings, according to a research letter published online in JAMA Internal Medicine.
The authors highlighted the need for such savings by noting that, while biologics comprise less than 5% of prescription drug use, their price tag amounts to about 40% of U.S. drug spending, Biologic manufacturers often delay the availability of biosimilars for additional years beyond the original patent expiration through further patents for supplemental indications. To provide a counterbalance, federal law allows the Food and Drug Administration to approve “skinny-label” generics and biosimilars that carve out patent-protected indications or regulatory exclusivities. But once a generic drug reaches the market through this process with a skinny label, it may often be substituted for indications that go beyond the ones listed on the skinny label. In fact, some state laws mandate that pharmacists substitute interchangeable generics for brand-name drugs, helping to decrease drug prices. In response to legal threats to the skinny-label pathway, Alexander C. Egilman and colleagues at Brigham and Women’s Hospital and Harvard Medical School, both in Boston, assessed the frequency of approval and marketing of skinny-label biosimilars from 2015 to 2021 and the resultant savings to Medicare.
The authors estimated annual Part B (clinician-administered) savings from skinny-label biosimilars through 2020 by comparing actual biologic and skinny-label biosimilar spending with estimated biologic spending without competition using the Medicare Dashboard. They assumed that the unit price of the biologic would increase at its 5-year compound annual growth rate prior to competition.
In that period, the FDA approved 33 biosimilars linked to 11 biologics. Among them, 22 (66.7%) had a skinny label. Of 21 biosimilars marketed before 2022, 13 (61.9%) were launched with a skinny label. Of the 8 biologics linked to these 21 biosimilars, 5 of the first-to-market biosimilars had skinny labels (bevacizumab, filgrastim, infliximab, pegfilgrastim, and rituximab), leading to earlier competition through 2021.
The estimated $1.5 billion in savings to Medicare from these skinny-label biosimilars over the 2015-2020 span represents 4.9% of the $30.2 billion that Medicare spent on the five biologics during this period. The researchers pointed out that once adalimumab (Humira) faces skinny-label biosimilar competition in 2023, savings will likely grow substantially.
In response to the research letter, an editor’s note by JAMA Internal Medicine Editorial Fellow Eric Ward, MD, and JAMA Internal Medicine Editor at Large and Online Editor Robert Steinbrook, MD, stated that, between 2015 and 2019, 24 (43%) of 56 brand-name drugs had competition from skinny-labeled generic formulations after first becoming available as generics.
The editors also referenced a JAMA Viewpoints article from 2021 that reviewed the most recent case challenging the skinny-label pathway in which GlaxoSmithKline sued Teva for its marketing of a skinny-label generic of the brand-name beta-blocker carvedilol (Coreg) that the plaintive claimed “induced physicians to prescribe carvedilol for indications that had been carved out by Teva’s skinny label, thus infringing GlaxoSmithKline’s patents.” A $235 million judgment against Teva was overturned by a district court and then reversed again by a Federal Circuit court that, after receiving criticism, reconsidered the case, and a panel affirmed the judgment against Teva.
“The Federal Circuit panel’s decision has the potential to put generic drugs that fail to adequately carve out indications from the brand name labeling at risk for damages related to infringement,” the authors wrote. Similar claims of infringement are being heard in other courts, they wrote, and they urged careful targeting of skinny-label carveouts, and suggest also that challenges to the arguments used against Teva focus on preservation of First Amendment rights as protection for lawful and accurate speech in drug labels.
“The legal uncertainties are likely to continue, as manufacturers pursue novel and complex strategies to protect the patents and regulatory exclusivities of brand-name drugs and biologics,” Dr. Ward and Dr. Steinbrook wrote, adding that “the path forward is for Congress to enact additional legislation that reaffirms and strengthens the permissibility of skinny labeling.”
The research letter’s corresponding author, Ameet Sarpatwari, PhD, JD, assistant professor at Harvard Medical School, and assistant director for the Harvard Program On Regulation, Therapeutics, And Law, echoed concerns over the Teva case in an interview. “There has certainly been concern that should the appellate decision stand, there will be a chilling effect. As the lone dissenter in that case noted, ‘no skinny-label generic is safe.’ I think many generic and biosimilar manufacturers are awaiting to see whether the Supreme Court will take the case.”
He added: “I do not believe the likelihood of skinny-label-supportive legislation making it through Congress will be greatly diminished in a divided Congress. Democrats and Republicans alike should seek to promote competition in the marketplace, which is what the skinny-labeling pathway accomplishes.”
The authors reported no relevant conflicts of interest. The research was funded by a grant from Arnold Ventures.
Competition between five biologic drugs and their skinny-label biosimilars saved Medicare an estimated $1.5 billion during 2015-2020. But these savings accruing to Medicare and the availability of those and other biosimilars through skinny labeling is under threat from recent court rulings, according to a research letter published online in JAMA Internal Medicine.
The authors highlighted the need for such savings by noting that, while biologics comprise less than 5% of prescription drug use, their price tag amounts to about 40% of U.S. drug spending, Biologic manufacturers often delay the availability of biosimilars for additional years beyond the original patent expiration through further patents for supplemental indications. To provide a counterbalance, federal law allows the Food and Drug Administration to approve “skinny-label” generics and biosimilars that carve out patent-protected indications or regulatory exclusivities. But once a generic drug reaches the market through this process with a skinny label, it may often be substituted for indications that go beyond the ones listed on the skinny label. In fact, some state laws mandate that pharmacists substitute interchangeable generics for brand-name drugs, helping to decrease drug prices. In response to legal threats to the skinny-label pathway, Alexander C. Egilman and colleagues at Brigham and Women’s Hospital and Harvard Medical School, both in Boston, assessed the frequency of approval and marketing of skinny-label biosimilars from 2015 to 2021 and the resultant savings to Medicare.
The authors estimated annual Part B (clinician-administered) savings from skinny-label biosimilars through 2020 by comparing actual biologic and skinny-label biosimilar spending with estimated biologic spending without competition using the Medicare Dashboard. They assumed that the unit price of the biologic would increase at its 5-year compound annual growth rate prior to competition.
In that period, the FDA approved 33 biosimilars linked to 11 biologics. Among them, 22 (66.7%) had a skinny label. Of 21 biosimilars marketed before 2022, 13 (61.9%) were launched with a skinny label. Of the 8 biologics linked to these 21 biosimilars, 5 of the first-to-market biosimilars had skinny labels (bevacizumab, filgrastim, infliximab, pegfilgrastim, and rituximab), leading to earlier competition through 2021.
The estimated $1.5 billion in savings to Medicare from these skinny-label biosimilars over the 2015-2020 span represents 4.9% of the $30.2 billion that Medicare spent on the five biologics during this period. The researchers pointed out that once adalimumab (Humira) faces skinny-label biosimilar competition in 2023, savings will likely grow substantially.
In response to the research letter, an editor’s note by JAMA Internal Medicine Editorial Fellow Eric Ward, MD, and JAMA Internal Medicine Editor at Large and Online Editor Robert Steinbrook, MD, stated that, between 2015 and 2019, 24 (43%) of 56 brand-name drugs had competition from skinny-labeled generic formulations after first becoming available as generics.
The editors also referenced a JAMA Viewpoints article from 2021 that reviewed the most recent case challenging the skinny-label pathway in which GlaxoSmithKline sued Teva for its marketing of a skinny-label generic of the brand-name beta-blocker carvedilol (Coreg) that the plaintive claimed “induced physicians to prescribe carvedilol for indications that had been carved out by Teva’s skinny label, thus infringing GlaxoSmithKline’s patents.” A $235 million judgment against Teva was overturned by a district court and then reversed again by a Federal Circuit court that, after receiving criticism, reconsidered the case, and a panel affirmed the judgment against Teva.
“The Federal Circuit panel’s decision has the potential to put generic drugs that fail to adequately carve out indications from the brand name labeling at risk for damages related to infringement,” the authors wrote. Similar claims of infringement are being heard in other courts, they wrote, and they urged careful targeting of skinny-label carveouts, and suggest also that challenges to the arguments used against Teva focus on preservation of First Amendment rights as protection for lawful and accurate speech in drug labels.
“The legal uncertainties are likely to continue, as manufacturers pursue novel and complex strategies to protect the patents and regulatory exclusivities of brand-name drugs and biologics,” Dr. Ward and Dr. Steinbrook wrote, adding that “the path forward is for Congress to enact additional legislation that reaffirms and strengthens the permissibility of skinny labeling.”
The research letter’s corresponding author, Ameet Sarpatwari, PhD, JD, assistant professor at Harvard Medical School, and assistant director for the Harvard Program On Regulation, Therapeutics, And Law, echoed concerns over the Teva case in an interview. “There has certainly been concern that should the appellate decision stand, there will be a chilling effect. As the lone dissenter in that case noted, ‘no skinny-label generic is safe.’ I think many generic and biosimilar manufacturers are awaiting to see whether the Supreme Court will take the case.”
He added: “I do not believe the likelihood of skinny-label-supportive legislation making it through Congress will be greatly diminished in a divided Congress. Democrats and Republicans alike should seek to promote competition in the marketplace, which is what the skinny-labeling pathway accomplishes.”
The authors reported no relevant conflicts of interest. The research was funded by a grant from Arnold Ventures.
FROM JAMA INTERNAL MEDICINE
New ACR vaccination guideline: Take your best shot
PHILADELPHIA – The new American College of Rheumatology Guideline for Vaccinations in Patients with Rheumatic and Musculoskeletal Diseases (RMDs) emphasizes that both adult and pediatric patients should receive recommended vaccinations whenever possible.
But the guideline, currently in press, also offers recommendations about whether and when to withhold vaccines from patients with RMDs, such as avoiding the use of live attenuated virus vaccines in patients who are on immunosuppressive drug regimens, such as conventional synthetic disease-modifying antirheumatic drugs (DMARDs), biologic DMARDs, or targeted synthetic DMARDs.
The new consensus guideline was formulated with the understanding that patients with RMDs are at increased risk for vaccine-preventable infections and more serious complications from infections, compared with the general population.
However, the guideline also acknowledges that the immunogenicity and safety of vaccines may differ among patients with RMDs, and that, depending on the patient age and disease state, individuals may benefit from modified vaccine indications, schedules, or modified medication schedules, said guideline panel member Anne Bass, MD, a rheumatologist at Hospital for Special Surgery and a professor of clinical medicine at Weill Cornell Medicine in New York, who presented the guideline with other panel members in a session outlining the recommendations at the annual meeting of the ACR.
“In addition, vaccination recommendations – since much of it relates to medications – really applies across diseases, and so the ACR felt that, rather than having vaccine recommendations tacked onto the end of treatment guidelines for each individual disease, that the topic should be discussed or tackled as a whole,” she said.
The guideline does not cover vaccinations in patients taking nonsteroidal anti-inflammatory drugs because this class of agents has minimal or no impact on antibody responses to vaccines. The guideline also does not address vaccinations against COVID-19 infections since the rapidly changing formulations would make the recommendations obsolete before they were even published, and because the U.S. Centers for Disease Control and Prevention provides up-to-date guidance on COVID-19 vaccinations in patients with compromised immunity, she said.
Guiding principles
The overarching principles of the guideline are to give indicated vaccines to patients with RMD whenever possible and that any decision to hold medications before or after vaccination consider the dosage used, RMD disease activity, and the patient’s risk for vaccine-preventable infection.
The guideline also states that “shared decision-making with patients is a key component of any vaccination strategy.”
Panel member Clifton O. Bingham III, MD, professor of medicine at Johns Hopkins University in Baltimore, outlined expanded indications for vaccinations against influenza, pneumococcal infections, varicella zoster virus (VZV) and human papillomavirus (HPV).
Influenza
The guideline conditionally recommends that patients with RMD aged 65 years and older and adults older than age 18 years who are on immunosuppressive medications should receive either high-dose or adjuvanted influenza vaccination rather than regular-dose vaccines.
“It’s recognized that the high-dose or adjuvanted vaccinations may be unavailable for patients when they’re seen in your practice,” Dr. Bingham said,” and we came out with two additional statements within the guidelines that said that any flu vaccine is recommended over no flu vaccinations, because we do know that responses are elicited, and a flu vaccination today is preferred over a flu vaccination delay.”
Pneumococcal vaccination
The panelists strongly recommended that patients with RMD younger than age 65 years who are on immunosuppressive medication receive pneumococcal vaccinations.
The ACR guideline is in sync with those issued by the CDC’s Advisory Committee on Immunization Practices, Dr. Bingham said. He urged audience members to visit a CDC-ACIP web page for more information on who should receive pneumococcal vaccination and when.
Recombinant varicella zoster
The recommendations strongly support that patients aged 18 years and over who are on immunosuppressive therapies should receive the recombinant VZV vaccine (Shingrix).
HPV
A less robust, conditional recommendation is for patients with RMDs who are between the ages of 26 and 45 years and on immunosuppressive medications to receive the HPV vaccine (if they have not already received the vaccine).
Non-live attenuated vaccines
Kevin Winthrop, MD, MPH, professor of infectious diseases and public health at Oregon Health & Science University, Portland, summarized the recommendations for managing immunosuppressive therapies in patients scheduled to receive vaccinations using killed or nonactive antigens.
“In influenza season, don’t pass up the opportunity to vaccinate,” he said, adding, “if you can wait on rituximab dosing, do it, and if you can’t, go ahead and vaccinate.”
The guidelines also recommend a 2-week methotrexate hold at the time of influenza vaccination; other DMARD dosing changes are likely not necessary at the time of vaccination, “but this is an area of fervent study, and I think in a year or two we’ll have more experimental hold data with regard to other DMARDs,” Dr. Winthrop said.
For other nonlive attenuated vaccinations, recommendations are similar to those for influenza, except with more flexible timing because these vaccinations are not seasonal. When and how to hold methotrexate is still up in the air, he said.
Additionally, it’s recommended that vaccinations be delayed in patients on high-dose prednisone until the drug is tapered to below 20 mg per day, and ideally to less than 10 mg per day, he said.
Live-attenuated vaccines
The guideline conditionally recommends deferring live-attenuated vaccines in patients on immunosuppressive drugs. It also recommends holding these medications “for an appropriate period before” vaccination and for 4 weeks afterward.
“Although the evidence around conventional synthetic DMARDs and TNF inhibitors is reassuring in terms of their safety at the time of live attenuated vaccines, as you can see the number of studies is quite small, and so the voting panel conditionally recommend against administering live-attenuated virus vaccines to patients who are on conventional synthetics, biologic, or targeted DMARDs,” Dr. Bass said.
In utero exposures
Most women with RMD who have recently given birth will consult their general pediatricians rather than rheumatologists for infant vaccinations, but pediatricians may not be aware of the affect that in utero exposures to biologic DMARDs can have on vaccine safety and immunogenicity in infants, Dr, Bass said.
“It’s important that you, as a provider, give your recommendations regarding infant rotavirus vaccination after in utero exposure to the pregnant rheumatic disease patient prior to delivery, and let that patient know that this is something that they should share with their pediatrician to be,” she advised audience members.
Getting the message out
In an interview, session moderator and guidelines panelist Lisa F. Imundo, MD, director of the center for adolescent rheumatology at Columbia University in New York, noted that rheumatologists don’t usually have the full schedule of pediatric vaccinations in stock and often leave the decisions about what to give – and when – to general practitioners.
“Pediatric rheumatologists sometimes will give patients flu vaccinations because they’re a high-risk population of patients, and we want to make sure that they’re getting it in a timely manner,” she said.
In addition, because pneumococcal polysaccharide vaccines are not indicated in the general pediatric population, children on biologic DMARDs who have completed their standard series of pneumococcal conjugate vaccines (PCV13 or PVC15) are recommended to get a 23-valent pneumococcal polysaccharide vaccine, Dr. Imundo said.
She also noted that communication between pediatric rheumatologists and general practitioners about vaccine recommendations can be challenging.
“It’s a huge issue, figuring out how we’re going to communicate all of this information to our pediatric colleagues,” she said. “With individual patients, we may sometimes remind doctors, especially with our younger patients who haven’t gotten their live vaccines, that they really shouldn’t get live vaccines until they’re off medication or until we arrange holding medication for some period of time.”
She said that ACR vaccine committee members are working with infectious disease specialists and guideline developers for the American Academy of Pediatrics to ensure guidelines include the most important vaccination recommendations for pediatric patients with RMDs.
The development process for the guidelines was supported by the ACR. Dr. Bass reported no relevant disclosures, Dr. Bingham disclosed consulting activities, grant/research support, and royalties from various corporate entities. Dr. Winthrop disclosed consulting activities for and research funding from various companies. Dr. Imundo reported no relevant financial relationships.
PHILADELPHIA – The new American College of Rheumatology Guideline for Vaccinations in Patients with Rheumatic and Musculoskeletal Diseases (RMDs) emphasizes that both adult and pediatric patients should receive recommended vaccinations whenever possible.
But the guideline, currently in press, also offers recommendations about whether and when to withhold vaccines from patients with RMDs, such as avoiding the use of live attenuated virus vaccines in patients who are on immunosuppressive drug regimens, such as conventional synthetic disease-modifying antirheumatic drugs (DMARDs), biologic DMARDs, or targeted synthetic DMARDs.
The new consensus guideline was formulated with the understanding that patients with RMDs are at increased risk for vaccine-preventable infections and more serious complications from infections, compared with the general population.
However, the guideline also acknowledges that the immunogenicity and safety of vaccines may differ among patients with RMDs, and that, depending on the patient age and disease state, individuals may benefit from modified vaccine indications, schedules, or modified medication schedules, said guideline panel member Anne Bass, MD, a rheumatologist at Hospital for Special Surgery and a professor of clinical medicine at Weill Cornell Medicine in New York, who presented the guideline with other panel members in a session outlining the recommendations at the annual meeting of the ACR.
“In addition, vaccination recommendations – since much of it relates to medications – really applies across diseases, and so the ACR felt that, rather than having vaccine recommendations tacked onto the end of treatment guidelines for each individual disease, that the topic should be discussed or tackled as a whole,” she said.
The guideline does not cover vaccinations in patients taking nonsteroidal anti-inflammatory drugs because this class of agents has minimal or no impact on antibody responses to vaccines. The guideline also does not address vaccinations against COVID-19 infections since the rapidly changing formulations would make the recommendations obsolete before they were even published, and because the U.S. Centers for Disease Control and Prevention provides up-to-date guidance on COVID-19 vaccinations in patients with compromised immunity, she said.
Guiding principles
The overarching principles of the guideline are to give indicated vaccines to patients with RMD whenever possible and that any decision to hold medications before or after vaccination consider the dosage used, RMD disease activity, and the patient’s risk for vaccine-preventable infection.
The guideline also states that “shared decision-making with patients is a key component of any vaccination strategy.”
Panel member Clifton O. Bingham III, MD, professor of medicine at Johns Hopkins University in Baltimore, outlined expanded indications for vaccinations against influenza, pneumococcal infections, varicella zoster virus (VZV) and human papillomavirus (HPV).
Influenza
The guideline conditionally recommends that patients with RMD aged 65 years and older and adults older than age 18 years who are on immunosuppressive medications should receive either high-dose or adjuvanted influenza vaccination rather than regular-dose vaccines.
“It’s recognized that the high-dose or adjuvanted vaccinations may be unavailable for patients when they’re seen in your practice,” Dr. Bingham said,” and we came out with two additional statements within the guidelines that said that any flu vaccine is recommended over no flu vaccinations, because we do know that responses are elicited, and a flu vaccination today is preferred over a flu vaccination delay.”
Pneumococcal vaccination
The panelists strongly recommended that patients with RMD younger than age 65 years who are on immunosuppressive medication receive pneumococcal vaccinations.
The ACR guideline is in sync with those issued by the CDC’s Advisory Committee on Immunization Practices, Dr. Bingham said. He urged audience members to visit a CDC-ACIP web page for more information on who should receive pneumococcal vaccination and when.
Recombinant varicella zoster
The recommendations strongly support that patients aged 18 years and over who are on immunosuppressive therapies should receive the recombinant VZV vaccine (Shingrix).
HPV
A less robust, conditional recommendation is for patients with RMDs who are between the ages of 26 and 45 years and on immunosuppressive medications to receive the HPV vaccine (if they have not already received the vaccine).
Non-live attenuated vaccines
Kevin Winthrop, MD, MPH, professor of infectious diseases and public health at Oregon Health & Science University, Portland, summarized the recommendations for managing immunosuppressive therapies in patients scheduled to receive vaccinations using killed or nonactive antigens.
“In influenza season, don’t pass up the opportunity to vaccinate,” he said, adding, “if you can wait on rituximab dosing, do it, and if you can’t, go ahead and vaccinate.”
The guidelines also recommend a 2-week methotrexate hold at the time of influenza vaccination; other DMARD dosing changes are likely not necessary at the time of vaccination, “but this is an area of fervent study, and I think in a year or two we’ll have more experimental hold data with regard to other DMARDs,” Dr. Winthrop said.
For other nonlive attenuated vaccinations, recommendations are similar to those for influenza, except with more flexible timing because these vaccinations are not seasonal. When and how to hold methotrexate is still up in the air, he said.
Additionally, it’s recommended that vaccinations be delayed in patients on high-dose prednisone until the drug is tapered to below 20 mg per day, and ideally to less than 10 mg per day, he said.
Live-attenuated vaccines
The guideline conditionally recommends deferring live-attenuated vaccines in patients on immunosuppressive drugs. It also recommends holding these medications “for an appropriate period before” vaccination and for 4 weeks afterward.
“Although the evidence around conventional synthetic DMARDs and TNF inhibitors is reassuring in terms of their safety at the time of live attenuated vaccines, as you can see the number of studies is quite small, and so the voting panel conditionally recommend against administering live-attenuated virus vaccines to patients who are on conventional synthetics, biologic, or targeted DMARDs,” Dr. Bass said.
In utero exposures
Most women with RMD who have recently given birth will consult their general pediatricians rather than rheumatologists for infant vaccinations, but pediatricians may not be aware of the affect that in utero exposures to biologic DMARDs can have on vaccine safety and immunogenicity in infants, Dr, Bass said.
“It’s important that you, as a provider, give your recommendations regarding infant rotavirus vaccination after in utero exposure to the pregnant rheumatic disease patient prior to delivery, and let that patient know that this is something that they should share with their pediatrician to be,” she advised audience members.
Getting the message out
In an interview, session moderator and guidelines panelist Lisa F. Imundo, MD, director of the center for adolescent rheumatology at Columbia University in New York, noted that rheumatologists don’t usually have the full schedule of pediatric vaccinations in stock and often leave the decisions about what to give – and when – to general practitioners.
“Pediatric rheumatologists sometimes will give patients flu vaccinations because they’re a high-risk population of patients, and we want to make sure that they’re getting it in a timely manner,” she said.
In addition, because pneumococcal polysaccharide vaccines are not indicated in the general pediatric population, children on biologic DMARDs who have completed their standard series of pneumococcal conjugate vaccines (PCV13 or PVC15) are recommended to get a 23-valent pneumococcal polysaccharide vaccine, Dr. Imundo said.
She also noted that communication between pediatric rheumatologists and general practitioners about vaccine recommendations can be challenging.
“It’s a huge issue, figuring out how we’re going to communicate all of this information to our pediatric colleagues,” she said. “With individual patients, we may sometimes remind doctors, especially with our younger patients who haven’t gotten their live vaccines, that they really shouldn’t get live vaccines until they’re off medication or until we arrange holding medication for some period of time.”
She said that ACR vaccine committee members are working with infectious disease specialists and guideline developers for the American Academy of Pediatrics to ensure guidelines include the most important vaccination recommendations for pediatric patients with RMDs.
The development process for the guidelines was supported by the ACR. Dr. Bass reported no relevant disclosures, Dr. Bingham disclosed consulting activities, grant/research support, and royalties from various corporate entities. Dr. Winthrop disclosed consulting activities for and research funding from various companies. Dr. Imundo reported no relevant financial relationships.
PHILADELPHIA – The new American College of Rheumatology Guideline for Vaccinations in Patients with Rheumatic and Musculoskeletal Diseases (RMDs) emphasizes that both adult and pediatric patients should receive recommended vaccinations whenever possible.
But the guideline, currently in press, also offers recommendations about whether and when to withhold vaccines from patients with RMDs, such as avoiding the use of live attenuated virus vaccines in patients who are on immunosuppressive drug regimens, such as conventional synthetic disease-modifying antirheumatic drugs (DMARDs), biologic DMARDs, or targeted synthetic DMARDs.
The new consensus guideline was formulated with the understanding that patients with RMDs are at increased risk for vaccine-preventable infections and more serious complications from infections, compared with the general population.
However, the guideline also acknowledges that the immunogenicity and safety of vaccines may differ among patients with RMDs, and that, depending on the patient age and disease state, individuals may benefit from modified vaccine indications, schedules, or modified medication schedules, said guideline panel member Anne Bass, MD, a rheumatologist at Hospital for Special Surgery and a professor of clinical medicine at Weill Cornell Medicine in New York, who presented the guideline with other panel members in a session outlining the recommendations at the annual meeting of the ACR.
“In addition, vaccination recommendations – since much of it relates to medications – really applies across diseases, and so the ACR felt that, rather than having vaccine recommendations tacked onto the end of treatment guidelines for each individual disease, that the topic should be discussed or tackled as a whole,” she said.
The guideline does not cover vaccinations in patients taking nonsteroidal anti-inflammatory drugs because this class of agents has minimal or no impact on antibody responses to vaccines. The guideline also does not address vaccinations against COVID-19 infections since the rapidly changing formulations would make the recommendations obsolete before they were even published, and because the U.S. Centers for Disease Control and Prevention provides up-to-date guidance on COVID-19 vaccinations in patients with compromised immunity, she said.
Guiding principles
The overarching principles of the guideline are to give indicated vaccines to patients with RMD whenever possible and that any decision to hold medications before or after vaccination consider the dosage used, RMD disease activity, and the patient’s risk for vaccine-preventable infection.
The guideline also states that “shared decision-making with patients is a key component of any vaccination strategy.”
Panel member Clifton O. Bingham III, MD, professor of medicine at Johns Hopkins University in Baltimore, outlined expanded indications for vaccinations against influenza, pneumococcal infections, varicella zoster virus (VZV) and human papillomavirus (HPV).
Influenza
The guideline conditionally recommends that patients with RMD aged 65 years and older and adults older than age 18 years who are on immunosuppressive medications should receive either high-dose or adjuvanted influenza vaccination rather than regular-dose vaccines.
“It’s recognized that the high-dose or adjuvanted vaccinations may be unavailable for patients when they’re seen in your practice,” Dr. Bingham said,” and we came out with two additional statements within the guidelines that said that any flu vaccine is recommended over no flu vaccinations, because we do know that responses are elicited, and a flu vaccination today is preferred over a flu vaccination delay.”
Pneumococcal vaccination
The panelists strongly recommended that patients with RMD younger than age 65 years who are on immunosuppressive medication receive pneumococcal vaccinations.
The ACR guideline is in sync with those issued by the CDC’s Advisory Committee on Immunization Practices, Dr. Bingham said. He urged audience members to visit a CDC-ACIP web page for more information on who should receive pneumococcal vaccination and when.
Recombinant varicella zoster
The recommendations strongly support that patients aged 18 years and over who are on immunosuppressive therapies should receive the recombinant VZV vaccine (Shingrix).
HPV
A less robust, conditional recommendation is for patients with RMDs who are between the ages of 26 and 45 years and on immunosuppressive medications to receive the HPV vaccine (if they have not already received the vaccine).
Non-live attenuated vaccines
Kevin Winthrop, MD, MPH, professor of infectious diseases and public health at Oregon Health & Science University, Portland, summarized the recommendations for managing immunosuppressive therapies in patients scheduled to receive vaccinations using killed or nonactive antigens.
“In influenza season, don’t pass up the opportunity to vaccinate,” he said, adding, “if you can wait on rituximab dosing, do it, and if you can’t, go ahead and vaccinate.”
The guidelines also recommend a 2-week methotrexate hold at the time of influenza vaccination; other DMARD dosing changes are likely not necessary at the time of vaccination, “but this is an area of fervent study, and I think in a year or two we’ll have more experimental hold data with regard to other DMARDs,” Dr. Winthrop said.
For other nonlive attenuated vaccinations, recommendations are similar to those for influenza, except with more flexible timing because these vaccinations are not seasonal. When and how to hold methotrexate is still up in the air, he said.
Additionally, it’s recommended that vaccinations be delayed in patients on high-dose prednisone until the drug is tapered to below 20 mg per day, and ideally to less than 10 mg per day, he said.
Live-attenuated vaccines
The guideline conditionally recommends deferring live-attenuated vaccines in patients on immunosuppressive drugs. It also recommends holding these medications “for an appropriate period before” vaccination and for 4 weeks afterward.
“Although the evidence around conventional synthetic DMARDs and TNF inhibitors is reassuring in terms of their safety at the time of live attenuated vaccines, as you can see the number of studies is quite small, and so the voting panel conditionally recommend against administering live-attenuated virus vaccines to patients who are on conventional synthetics, biologic, or targeted DMARDs,” Dr. Bass said.
In utero exposures
Most women with RMD who have recently given birth will consult their general pediatricians rather than rheumatologists for infant vaccinations, but pediatricians may not be aware of the affect that in utero exposures to biologic DMARDs can have on vaccine safety and immunogenicity in infants, Dr, Bass said.
“It’s important that you, as a provider, give your recommendations regarding infant rotavirus vaccination after in utero exposure to the pregnant rheumatic disease patient prior to delivery, and let that patient know that this is something that they should share with their pediatrician to be,” she advised audience members.
Getting the message out
In an interview, session moderator and guidelines panelist Lisa F. Imundo, MD, director of the center for adolescent rheumatology at Columbia University in New York, noted that rheumatologists don’t usually have the full schedule of pediatric vaccinations in stock and often leave the decisions about what to give – and when – to general practitioners.
“Pediatric rheumatologists sometimes will give patients flu vaccinations because they’re a high-risk population of patients, and we want to make sure that they’re getting it in a timely manner,” she said.
In addition, because pneumococcal polysaccharide vaccines are not indicated in the general pediatric population, children on biologic DMARDs who have completed their standard series of pneumococcal conjugate vaccines (PCV13 or PVC15) are recommended to get a 23-valent pneumococcal polysaccharide vaccine, Dr. Imundo said.
She also noted that communication between pediatric rheumatologists and general practitioners about vaccine recommendations can be challenging.
“It’s a huge issue, figuring out how we’re going to communicate all of this information to our pediatric colleagues,” she said. “With individual patients, we may sometimes remind doctors, especially with our younger patients who haven’t gotten their live vaccines, that they really shouldn’t get live vaccines until they’re off medication or until we arrange holding medication for some period of time.”
She said that ACR vaccine committee members are working with infectious disease specialists and guideline developers for the American Academy of Pediatrics to ensure guidelines include the most important vaccination recommendations for pediatric patients with RMDs.
The development process for the guidelines was supported by the ACR. Dr. Bass reported no relevant disclosures, Dr. Bingham disclosed consulting activities, grant/research support, and royalties from various corporate entities. Dr. Winthrop disclosed consulting activities for and research funding from various companies. Dr. Imundo reported no relevant financial relationships.
AT ACR 2022
NSAIDs for spondyloarthritis may affect time to conception
PHILADELPHIA – Women with spondyloarthritis (SpA) who are desiring pregnancy may want to consider decreasing use or discontinuing use (with supervision) of nonsteroidal anti-inflammatory drugs before conception, new data suggest.
Researchers have found a connection between NSAID use and age and a significantly longer time to conception among women with spondyloarthritis. Sabrina Hamroun, MMed, with the rheumatology department at the University Hospital Cochin, Paris, presented the findings during a press conference at the annual meeting of the American College of Rheumatology.
SpA commonly affects women of childbearing age, but data are sparse regarding the effects of disease on fertility.
Patients in the study were taken from the French multicenter cohort GR2 from 2015 to June 2021.
Among the 207 patients with SpA in the cohort, 88 were selected for analysis of time to conception. Of these, 56 patients (63.6%) had a clinical pregnancy during follow-up.
Subfertility group took an average of 16 months to get pregnant
Subfertility was observed in 40 (45.4%) of the women, with an average time to conception of 16.1 months. A woman was considered subfertile if her time to conception was more than 12 months or if she did not become pregnant.
The average preconception Bath Ankylosing Spondylitis Disease Activity Index score was 2.9 (+/- 2.1), the authors noted. The average age of the participants was 32 years.
Twenty-three patients were treated with NSAIDs, eight with corticosteroids, 12 with conventional synthetic disease-modifying antirheumatic drugs, and 61 with biologics.
Researchers adjusted for factors including age, body mass index, disease duration and severity, smoking, form of SpA (axial, peripheral, or both), and medication in the preconception period.
They found significant associations between longer time to conception and age (hazard ratio, 1.22; 95% confidence interval, 1.08-1.40; P < .001), and a much higher hazard ratio with the use of NSAIDs during preconception (HR, 3.01; 95% CI, 2.15-3.85; P = .01).
Some data unavailable
Ms. Hamroun acknowledged that no data were available on the frequency of sexual intercourse or quality of life, factors that could affect time to conception. Women were asked when they discontinued contraceptive use and actively began trying to become pregnant.
She stated that information on the dose of NSAIDs used by the patients was incomplete, noting, “We were therefore unable to adjust the results of our statistical analyses on the dose used by patients.”
Additionally, because the study participants were patients at tertiary centers in France and had more severe disease, the results may not be generalizable to all women of childbearing age. Patients with less severe SpA are often managed in outpatient settings in France, she said.
When asked about alternatives to NSAIDs, Ms. Hamroun said that anti–tumor necrosis factor agents with low placental passage may be a good alternative “if a woman with long-standing difficulties to conceive needs a regular use of NSAIDs to control disease activity, in the absence of any other cause of subfertility.”
The patient’s age must also be considered, she noted.
“A therapeutic switch may be favored in a woman over 35 years of age, for example, whose fertility is already impaired by age,” Ms. Hamroun said.
As for the mechanism that might explain the effects of NSAIDs on conception, Ms. Hamroun said that prostaglandins are essential to ovulation and embryo implantation and explained that NSAIDs may work against ovulation and result in poor implantation (miscarriage) by blocking prostaglandins.
She pointed out that her results are in line with the ACR’s recommendation to discontinue NSAID use during the preconception period in women with SpA who are having difficulty conceiving.
Control before conception is important
Sinead Maguire, MD, a clinical and research fellow in the Spondylitis Program at Toronto (Ont.) Western Hospital who was not part of the study, said the study highlights the importance of optimizing disease control before conception.
“There are a number of things rheumatologists can do to support our SpA patients when they are trying to conceive,” she told this news organization. “One of the most important issues to address is ensuring their SpA is in remission and continues to remain so. For that reason, if a woman is requiring regular NSAIDs for symptom control, the results of this study might encourage me to consider a biologic agent sooner to ensure remission.”
She urged women who want to become pregnant to discuss medications with their rheumatologist before trying to conceive.
“It is very exciting to see studies such as this so that rheumatologists can provide answers to our patients’ questions with evidence-based advice,” she said.
Ms. Hamroun and several coauthors had no disclosures. Other coauthors disclosed relationships with companies including Merck/MSD, Novartis, Janssen, AbbVie/Abbott, Amgen, AstraZeneca, Biogen, Bristol-Myers Squibb, Galapagos, Eli Lilly, Novartis, and/or UCB. Dr. Maguire reports no relevant financial relationships.
A version of this article first appeared on Medscape.com.
PHILADELPHIA – Women with spondyloarthritis (SpA) who are desiring pregnancy may want to consider decreasing use or discontinuing use (with supervision) of nonsteroidal anti-inflammatory drugs before conception, new data suggest.
Researchers have found a connection between NSAID use and age and a significantly longer time to conception among women with spondyloarthritis. Sabrina Hamroun, MMed, with the rheumatology department at the University Hospital Cochin, Paris, presented the findings during a press conference at the annual meeting of the American College of Rheumatology.
SpA commonly affects women of childbearing age, but data are sparse regarding the effects of disease on fertility.
Patients in the study were taken from the French multicenter cohort GR2 from 2015 to June 2021.
Among the 207 patients with SpA in the cohort, 88 were selected for analysis of time to conception. Of these, 56 patients (63.6%) had a clinical pregnancy during follow-up.
Subfertility group took an average of 16 months to get pregnant
Subfertility was observed in 40 (45.4%) of the women, with an average time to conception of 16.1 months. A woman was considered subfertile if her time to conception was more than 12 months or if she did not become pregnant.
The average preconception Bath Ankylosing Spondylitis Disease Activity Index score was 2.9 (+/- 2.1), the authors noted. The average age of the participants was 32 years.
Twenty-three patients were treated with NSAIDs, eight with corticosteroids, 12 with conventional synthetic disease-modifying antirheumatic drugs, and 61 with biologics.
Researchers adjusted for factors including age, body mass index, disease duration and severity, smoking, form of SpA (axial, peripheral, or both), and medication in the preconception period.
They found significant associations between longer time to conception and age (hazard ratio, 1.22; 95% confidence interval, 1.08-1.40; P < .001), and a much higher hazard ratio with the use of NSAIDs during preconception (HR, 3.01; 95% CI, 2.15-3.85; P = .01).
Some data unavailable
Ms. Hamroun acknowledged that no data were available on the frequency of sexual intercourse or quality of life, factors that could affect time to conception. Women were asked when they discontinued contraceptive use and actively began trying to become pregnant.
She stated that information on the dose of NSAIDs used by the patients was incomplete, noting, “We were therefore unable to adjust the results of our statistical analyses on the dose used by patients.”
Additionally, because the study participants were patients at tertiary centers in France and had more severe disease, the results may not be generalizable to all women of childbearing age. Patients with less severe SpA are often managed in outpatient settings in France, she said.
When asked about alternatives to NSAIDs, Ms. Hamroun said that anti–tumor necrosis factor agents with low placental passage may be a good alternative “if a woman with long-standing difficulties to conceive needs a regular use of NSAIDs to control disease activity, in the absence of any other cause of subfertility.”
The patient’s age must also be considered, she noted.
“A therapeutic switch may be favored in a woman over 35 years of age, for example, whose fertility is already impaired by age,” Ms. Hamroun said.
As for the mechanism that might explain the effects of NSAIDs on conception, Ms. Hamroun said that prostaglandins are essential to ovulation and embryo implantation and explained that NSAIDs may work against ovulation and result in poor implantation (miscarriage) by blocking prostaglandins.
She pointed out that her results are in line with the ACR’s recommendation to discontinue NSAID use during the preconception period in women with SpA who are having difficulty conceiving.
Control before conception is important
Sinead Maguire, MD, a clinical and research fellow in the Spondylitis Program at Toronto (Ont.) Western Hospital who was not part of the study, said the study highlights the importance of optimizing disease control before conception.
“There are a number of things rheumatologists can do to support our SpA patients when they are trying to conceive,” she told this news organization. “One of the most important issues to address is ensuring their SpA is in remission and continues to remain so. For that reason, if a woman is requiring regular NSAIDs for symptom control, the results of this study might encourage me to consider a biologic agent sooner to ensure remission.”
She urged women who want to become pregnant to discuss medications with their rheumatologist before trying to conceive.
“It is very exciting to see studies such as this so that rheumatologists can provide answers to our patients’ questions with evidence-based advice,” she said.
Ms. Hamroun and several coauthors had no disclosures. Other coauthors disclosed relationships with companies including Merck/MSD, Novartis, Janssen, AbbVie/Abbott, Amgen, AstraZeneca, Biogen, Bristol-Myers Squibb, Galapagos, Eli Lilly, Novartis, and/or UCB. Dr. Maguire reports no relevant financial relationships.
A version of this article first appeared on Medscape.com.
PHILADELPHIA – Women with spondyloarthritis (SpA) who are desiring pregnancy may want to consider decreasing use or discontinuing use (with supervision) of nonsteroidal anti-inflammatory drugs before conception, new data suggest.
Researchers have found a connection between NSAID use and age and a significantly longer time to conception among women with spondyloarthritis. Sabrina Hamroun, MMed, with the rheumatology department at the University Hospital Cochin, Paris, presented the findings during a press conference at the annual meeting of the American College of Rheumatology.
SpA commonly affects women of childbearing age, but data are sparse regarding the effects of disease on fertility.
Patients in the study were taken from the French multicenter cohort GR2 from 2015 to June 2021.
Among the 207 patients with SpA in the cohort, 88 were selected for analysis of time to conception. Of these, 56 patients (63.6%) had a clinical pregnancy during follow-up.
Subfertility group took an average of 16 months to get pregnant
Subfertility was observed in 40 (45.4%) of the women, with an average time to conception of 16.1 months. A woman was considered subfertile if her time to conception was more than 12 months or if she did not become pregnant.
The average preconception Bath Ankylosing Spondylitis Disease Activity Index score was 2.9 (+/- 2.1), the authors noted. The average age of the participants was 32 years.
Twenty-three patients were treated with NSAIDs, eight with corticosteroids, 12 with conventional synthetic disease-modifying antirheumatic drugs, and 61 with biologics.
Researchers adjusted for factors including age, body mass index, disease duration and severity, smoking, form of SpA (axial, peripheral, or both), and medication in the preconception period.
They found significant associations between longer time to conception and age (hazard ratio, 1.22; 95% confidence interval, 1.08-1.40; P < .001), and a much higher hazard ratio with the use of NSAIDs during preconception (HR, 3.01; 95% CI, 2.15-3.85; P = .01).
Some data unavailable
Ms. Hamroun acknowledged that no data were available on the frequency of sexual intercourse or quality of life, factors that could affect time to conception. Women were asked when they discontinued contraceptive use and actively began trying to become pregnant.
She stated that information on the dose of NSAIDs used by the patients was incomplete, noting, “We were therefore unable to adjust the results of our statistical analyses on the dose used by patients.”
Additionally, because the study participants were patients at tertiary centers in France and had more severe disease, the results may not be generalizable to all women of childbearing age. Patients with less severe SpA are often managed in outpatient settings in France, she said.
When asked about alternatives to NSAIDs, Ms. Hamroun said that anti–tumor necrosis factor agents with low placental passage may be a good alternative “if a woman with long-standing difficulties to conceive needs a regular use of NSAIDs to control disease activity, in the absence of any other cause of subfertility.”
The patient’s age must also be considered, she noted.
“A therapeutic switch may be favored in a woman over 35 years of age, for example, whose fertility is already impaired by age,” Ms. Hamroun said.
As for the mechanism that might explain the effects of NSAIDs on conception, Ms. Hamroun said that prostaglandins are essential to ovulation and embryo implantation and explained that NSAIDs may work against ovulation and result in poor implantation (miscarriage) by blocking prostaglandins.
She pointed out that her results are in line with the ACR’s recommendation to discontinue NSAID use during the preconception period in women with SpA who are having difficulty conceiving.
Control before conception is important
Sinead Maguire, MD, a clinical and research fellow in the Spondylitis Program at Toronto (Ont.) Western Hospital who was not part of the study, said the study highlights the importance of optimizing disease control before conception.
“There are a number of things rheumatologists can do to support our SpA patients when they are trying to conceive,” she told this news organization. “One of the most important issues to address is ensuring their SpA is in remission and continues to remain so. For that reason, if a woman is requiring regular NSAIDs for symptom control, the results of this study might encourage me to consider a biologic agent sooner to ensure remission.”
She urged women who want to become pregnant to discuss medications with their rheumatologist before trying to conceive.
“It is very exciting to see studies such as this so that rheumatologists can provide answers to our patients’ questions with evidence-based advice,” she said.
Ms. Hamroun and several coauthors had no disclosures. Other coauthors disclosed relationships with companies including Merck/MSD, Novartis, Janssen, AbbVie/Abbott, Amgen, AstraZeneca, Biogen, Bristol-Myers Squibb, Galapagos, Eli Lilly, Novartis, and/or UCB. Dr. Maguire reports no relevant financial relationships.
A version of this article first appeared on Medscape.com.
AT ACR 2022
Retention rates high after biosimilar-to-biosimilar switch for inflammatory arthritis
PHILADELPHIA – When patients with inflammatory rheumatic diseases were switched from one biosimilar agent to another, treatment retention rates were high, investigators in Denmark reported.
The findings suggest patient-related factors rather than drug-related factors appear to determine whether patients will stay on the new drug, the researchers said.
One year after a Danish government-mandated switch from one infliximab (Remicade) biosimilar to another equally efficacious but less costly biosimilar, 83% of patients who had started therapy on a biosimilar (so-called “originator-naive” patients) stayed on the newly assigned therapy. And so did 92% of patients who had started on the original infliximab (“originator experienced”) before they were switched to one biosimilar and then another.
“In regards to potential baseline predictors, we found that treatment withdrawal was more frequent among originator-naive switchers and patients with higher baseline disease activity, especially [in] patient-reported outcomes, which may indicate that treatment-related outcomes may be more affected by patient-related rather than drug-related factors,” said lead author Hafsah Nabi, MD from the Danish biosimilar registry DANBIO and a PhD candidate at the Copenhagen Center for Arthritis Research.
Dr. Nabi reported the results in an oral abstract session at the annual meeting of the American College of Rheumatology.
Annual review of biologic agents
In Denmark, health authorities issue annual recommendations for the use of biologic agents. “And since patients receive this treatment free from the hospital, based on the tax system, the switches are made due to these cost considerations,” Dr. Nabi said in an interview.
To get the nod from Danish pharmaceutical regulators, pharmaceutical manufacturers submit drugs that have already been approved by the European Medicines Agency for consideration for treatment of specific indications, explained coauthor Merete Lund Hetland, MD, PhD, DMSc, from Rigshospitalet in Copenhagen.
“Those drugs that are then considered equally safe and effective are invited to this process where they will give their bid, and then the cheapest one will win,” she said.
The winning formulation will be able to capture about 80% of prescriptions for that indication for the coming year.
Awake at the switch
Dr. Nabi, Dr. Hetland, and colleagues studied how one such recent government-mandated switch from one biosimilar to another affected efficacy and patterns of care among patients with rheumatoid arthritis, psoriatic arthritis (PsA) and axial spondyloarthritis (axSpA).
To identify prior comorbidities, they drew data from the DANBIO registry, which is linked to patient specific but anonymous data from other comprehensive birth-to-death patient registries in Denmark.
They looked at all patients with RA, PsA, or axSpA who were switched from CT-P13 (Remsira, Inflectra) to GP1111 (Zessly) from April 1, 2019, to Feb. 1, 2020.
They identified a total of 1,605 patients, including 685 with RA, 314 with PsA, and 606 with axSpa. The median disease duration was 9 years, and 37% of all patients were in remission according to Clinical Disease Activity Index or Ankylosing Spondylitis Disease Activity Scale.
Of this group, 1,171 had started therapy on a biosimilar.
As noted above, 83% of patients who had never received original infliximab, and 92% of those who were originator experienced were still on the new biosimilar 1 year after the switch.
In a multivariate analysis controlling for demographic and clinical factors at baseline, the variables significantly associated with treatment withdrawal from the new biosimilar (GP11110) included previous Remicade exposure (hazard ratio, 0.36), methotrexate use (HR, 0.60), and patient-reported global visual analog scale (HR, 1.02).
Among all patients, disease activity was stable 6 months before and after the switch, Dr. Nabi said, although she did not show data to support it.
Patient education benefit
During the session, Jonathan Kay, MD, professor of rheumatology and chair of the division of rheumatology at the University of Massachusetts, Worcester, who was not involved the study, asked Dr. Nabi whether patients were educated about equivalent efficacy and safety of biosimilars prior to the switch. He noted that education prior to switching led to a much lower patient withdrawal rate in a similar switching study conducted in The Netherlands.
“In this study, we haven’t looked more specifically into the education and which strategies have been used prior to switching, and we also conclude in the study that there may be the presence of a nocebo effect, which can be handled by better educating the patients,” she replied.
The nocebo effect refers to the phenomenon in which a patient’s belief that a specific intervention may cause harm actually can lead to negative outcomes – in other words, the opposite of the placebo effect.
In an interview, Dr. Kay said that he is confident about the efficacy, safety, and equivalency of approved biosimilar agents.
“A biosimilar that has been reviewed and approved by a regulatory agency such as the [Food and Drug Administration or the [European Medicines Agency] should be equivalent in efficacy and comparable in safety and immunogenicity. I would be fully confident in switching from the reference product to the biosimilar,” he said.
Dr. Nabi reported that the study was partly funded by a research grant from Sandoz, the maker of GP1111. Dr. Hetland has disclosed grants from various companies, not including Sandoz. Dr. Kay disclosed consulting fees from various companies, not including Sandoz.
PHILADELPHIA – When patients with inflammatory rheumatic diseases were switched from one biosimilar agent to another, treatment retention rates were high, investigators in Denmark reported.
The findings suggest patient-related factors rather than drug-related factors appear to determine whether patients will stay on the new drug, the researchers said.
One year after a Danish government-mandated switch from one infliximab (Remicade) biosimilar to another equally efficacious but less costly biosimilar, 83% of patients who had started therapy on a biosimilar (so-called “originator-naive” patients) stayed on the newly assigned therapy. And so did 92% of patients who had started on the original infliximab (“originator experienced”) before they were switched to one biosimilar and then another.
“In regards to potential baseline predictors, we found that treatment withdrawal was more frequent among originator-naive switchers and patients with higher baseline disease activity, especially [in] patient-reported outcomes, which may indicate that treatment-related outcomes may be more affected by patient-related rather than drug-related factors,” said lead author Hafsah Nabi, MD from the Danish biosimilar registry DANBIO and a PhD candidate at the Copenhagen Center for Arthritis Research.
Dr. Nabi reported the results in an oral abstract session at the annual meeting of the American College of Rheumatology.
Annual review of biologic agents
In Denmark, health authorities issue annual recommendations for the use of biologic agents. “And since patients receive this treatment free from the hospital, based on the tax system, the switches are made due to these cost considerations,” Dr. Nabi said in an interview.
To get the nod from Danish pharmaceutical regulators, pharmaceutical manufacturers submit drugs that have already been approved by the European Medicines Agency for consideration for treatment of specific indications, explained coauthor Merete Lund Hetland, MD, PhD, DMSc, from Rigshospitalet in Copenhagen.
“Those drugs that are then considered equally safe and effective are invited to this process where they will give their bid, and then the cheapest one will win,” she said.
The winning formulation will be able to capture about 80% of prescriptions for that indication for the coming year.
Awake at the switch
Dr. Nabi, Dr. Hetland, and colleagues studied how one such recent government-mandated switch from one biosimilar to another affected efficacy and patterns of care among patients with rheumatoid arthritis, psoriatic arthritis (PsA) and axial spondyloarthritis (axSpA).
To identify prior comorbidities, they drew data from the DANBIO registry, which is linked to patient specific but anonymous data from other comprehensive birth-to-death patient registries in Denmark.
They looked at all patients with RA, PsA, or axSpA who were switched from CT-P13 (Remsira, Inflectra) to GP1111 (Zessly) from April 1, 2019, to Feb. 1, 2020.
They identified a total of 1,605 patients, including 685 with RA, 314 with PsA, and 606 with axSpa. The median disease duration was 9 years, and 37% of all patients were in remission according to Clinical Disease Activity Index or Ankylosing Spondylitis Disease Activity Scale.
Of this group, 1,171 had started therapy on a biosimilar.
As noted above, 83% of patients who had never received original infliximab, and 92% of those who were originator experienced were still on the new biosimilar 1 year after the switch.
In a multivariate analysis controlling for demographic and clinical factors at baseline, the variables significantly associated with treatment withdrawal from the new biosimilar (GP11110) included previous Remicade exposure (hazard ratio, 0.36), methotrexate use (HR, 0.60), and patient-reported global visual analog scale (HR, 1.02).
Among all patients, disease activity was stable 6 months before and after the switch, Dr. Nabi said, although she did not show data to support it.
Patient education benefit
During the session, Jonathan Kay, MD, professor of rheumatology and chair of the division of rheumatology at the University of Massachusetts, Worcester, who was not involved the study, asked Dr. Nabi whether patients were educated about equivalent efficacy and safety of biosimilars prior to the switch. He noted that education prior to switching led to a much lower patient withdrawal rate in a similar switching study conducted in The Netherlands.
“In this study, we haven’t looked more specifically into the education and which strategies have been used prior to switching, and we also conclude in the study that there may be the presence of a nocebo effect, which can be handled by better educating the patients,” she replied.
The nocebo effect refers to the phenomenon in which a patient’s belief that a specific intervention may cause harm actually can lead to negative outcomes – in other words, the opposite of the placebo effect.
In an interview, Dr. Kay said that he is confident about the efficacy, safety, and equivalency of approved biosimilar agents.
“A biosimilar that has been reviewed and approved by a regulatory agency such as the [Food and Drug Administration or the [European Medicines Agency] should be equivalent in efficacy and comparable in safety and immunogenicity. I would be fully confident in switching from the reference product to the biosimilar,” he said.
Dr. Nabi reported that the study was partly funded by a research grant from Sandoz, the maker of GP1111. Dr. Hetland has disclosed grants from various companies, not including Sandoz. Dr. Kay disclosed consulting fees from various companies, not including Sandoz.
PHILADELPHIA – When patients with inflammatory rheumatic diseases were switched from one biosimilar agent to another, treatment retention rates were high, investigators in Denmark reported.
The findings suggest patient-related factors rather than drug-related factors appear to determine whether patients will stay on the new drug, the researchers said.
One year after a Danish government-mandated switch from one infliximab (Remicade) biosimilar to another equally efficacious but less costly biosimilar, 83% of patients who had started therapy on a biosimilar (so-called “originator-naive” patients) stayed on the newly assigned therapy. And so did 92% of patients who had started on the original infliximab (“originator experienced”) before they were switched to one biosimilar and then another.
“In regards to potential baseline predictors, we found that treatment withdrawal was more frequent among originator-naive switchers and patients with higher baseline disease activity, especially [in] patient-reported outcomes, which may indicate that treatment-related outcomes may be more affected by patient-related rather than drug-related factors,” said lead author Hafsah Nabi, MD from the Danish biosimilar registry DANBIO and a PhD candidate at the Copenhagen Center for Arthritis Research.
Dr. Nabi reported the results in an oral abstract session at the annual meeting of the American College of Rheumatology.
Annual review of biologic agents
In Denmark, health authorities issue annual recommendations for the use of biologic agents. “And since patients receive this treatment free from the hospital, based on the tax system, the switches are made due to these cost considerations,” Dr. Nabi said in an interview.
To get the nod from Danish pharmaceutical regulators, pharmaceutical manufacturers submit drugs that have already been approved by the European Medicines Agency for consideration for treatment of specific indications, explained coauthor Merete Lund Hetland, MD, PhD, DMSc, from Rigshospitalet in Copenhagen.
“Those drugs that are then considered equally safe and effective are invited to this process where they will give their bid, and then the cheapest one will win,” she said.
The winning formulation will be able to capture about 80% of prescriptions for that indication for the coming year.
Awake at the switch
Dr. Nabi, Dr. Hetland, and colleagues studied how one such recent government-mandated switch from one biosimilar to another affected efficacy and patterns of care among patients with rheumatoid arthritis, psoriatic arthritis (PsA) and axial spondyloarthritis (axSpA).
To identify prior comorbidities, they drew data from the DANBIO registry, which is linked to patient specific but anonymous data from other comprehensive birth-to-death patient registries in Denmark.
They looked at all patients with RA, PsA, or axSpA who were switched from CT-P13 (Remsira, Inflectra) to GP1111 (Zessly) from April 1, 2019, to Feb. 1, 2020.
They identified a total of 1,605 patients, including 685 with RA, 314 with PsA, and 606 with axSpa. The median disease duration was 9 years, and 37% of all patients were in remission according to Clinical Disease Activity Index or Ankylosing Spondylitis Disease Activity Scale.
Of this group, 1,171 had started therapy on a biosimilar.
As noted above, 83% of patients who had never received original infliximab, and 92% of those who were originator experienced were still on the new biosimilar 1 year after the switch.
In a multivariate analysis controlling for demographic and clinical factors at baseline, the variables significantly associated with treatment withdrawal from the new biosimilar (GP11110) included previous Remicade exposure (hazard ratio, 0.36), methotrexate use (HR, 0.60), and patient-reported global visual analog scale (HR, 1.02).
Among all patients, disease activity was stable 6 months before and after the switch, Dr. Nabi said, although she did not show data to support it.
Patient education benefit
During the session, Jonathan Kay, MD, professor of rheumatology and chair of the division of rheumatology at the University of Massachusetts, Worcester, who was not involved the study, asked Dr. Nabi whether patients were educated about equivalent efficacy and safety of biosimilars prior to the switch. He noted that education prior to switching led to a much lower patient withdrawal rate in a similar switching study conducted in The Netherlands.
“In this study, we haven’t looked more specifically into the education and which strategies have been used prior to switching, and we also conclude in the study that there may be the presence of a nocebo effect, which can be handled by better educating the patients,” she replied.
The nocebo effect refers to the phenomenon in which a patient’s belief that a specific intervention may cause harm actually can lead to negative outcomes – in other words, the opposite of the placebo effect.
In an interview, Dr. Kay said that he is confident about the efficacy, safety, and equivalency of approved biosimilar agents.
“A biosimilar that has been reviewed and approved by a regulatory agency such as the [Food and Drug Administration or the [European Medicines Agency] should be equivalent in efficacy and comparable in safety and immunogenicity. I would be fully confident in switching from the reference product to the biosimilar,” he said.
Dr. Nabi reported that the study was partly funded by a research grant from Sandoz, the maker of GP1111. Dr. Hetland has disclosed grants from various companies, not including Sandoz. Dr. Kay disclosed consulting fees from various companies, not including Sandoz.
AT ACR 2022
Therapeutic drug monitoring pays off for arthritis patients
Therapeutic drug monitoring allowed patients with rheumatoid arthritis, psoriatic arthritis, and spondyloarthritis to reduce their dosage of tumor necrosis factor–alpha (TNF) inhibitors, based on data from 239 individuals.
Use of TNF-alpha inhibitors improves treatment response for many arthritis patients but dosage is rarely adjusted on an individual level, which may lead to unnecessary overdosing in some patients, Mogens Pfeiffer-Jensen, MD, of Aarhus (Denmark) University Hospital, and colleagues wrote.
Data from previous studies suggest that therapeutic drug monitoring (TDM) based on serum trough levels may allow for dose optimization and dose reduction in inflammatory bowel disease patients, but data in patients with arthritis are lacking, they wrote.
In a study published in the Scandinavian Journal of Rheumatology, the researchers recruited 99 patients with RA, 48 with psoriatic arthritis (PsA), and 92 with spondyloarthritis (SpA). The participants were randomized to standard care or standard care plus TDM. Serum trough levels were assessed at baseline and at every 4 months, and prescription changes or drug switches were implemented based on these levels. At baseline, 81 patients were being treated with infliximab (Remicade and biosimilars), 79 with etanercept (Enbrel), and 79 with adalimumab (Humira).
The primary endpoint was reduced drug prescription after 48 weeks.
Overall, TDM significantly reduced prescription of infliximab by 12% (P = .001) and prescription of etanercept by 15% (P = .01), compared with standard care. TDM also prolonged the interdosing intervals of etanercept by 235% (P = .02) and of adalimumab by 28% (P = .04), compared with standard care.
TDM patients taking infliximab had more frequent dose reduction and less frequent dose increases during and after the study when compared with patients who stayed with standard care; similar trends were seen with adalimumab. TDM also accelerated the switch to other biologics for patients on all three medications.
No significant differences occurred in adverse events or hospitalizations between the TDM and standard care patients.
Clinical composite scores (Disease Activity Score based on 28 joints with C-reactive protein) were reduced in patients with RA and PsA who were taking adalimumab and randomized to TDM, but no other clinical outcome differences were noted. Scores on the Health Assessment Questionnaire and global Visual Analog Scale for pain were significantly lower in patients in the TDM group who were taking infliximab and adalimumab, “indicating equally or superior sustained remission across diagnoses,” the researchers emphasized.
The findings were limited by several factors, including the variations in pathophysiology and open-label design. “However, since the TDM was based on an objective serum value and decision procedures were clear, we do not consider the potential of unconscious bias to outweigh the benefits of dose-changing abilities,” they wrote.
The researchers expressed surprise that the reduced use of TNF-alpha inhibitors did not significantly reduce adverse events or serious adverse events, compared with standard care, but they proposed that standard of care may have taken adverse events into account, because all patients had received prescriptions at least 3 months before the study.
As for clinical implications, the current costs of the biochemical assays necessary for TDM may be a barrier to implementing TDM as a standard part of daily clinical practice, the researchers added. However, the study was strengthened by the inclusion of patients with RA, PsA, and SpA, and is the first known to include patients receiving etanercept or adalimumab in an examination of TDM.
“Our data support TDM based solely on serum trough levels in [TNF-alpha inhibitors] with different pharmacokinetics as a future key player in personalized medicine for chronic rheumatoid diseases treated with biologics,” they concluded.
The study was supported by Spydspidspuljen, Region Midt, Denmark, and Department of Rheumatology, Aarhus University Hospital. The researchers had no financial conflicts to disclose.
Therapeutic drug monitoring allowed patients with rheumatoid arthritis, psoriatic arthritis, and spondyloarthritis to reduce their dosage of tumor necrosis factor–alpha (TNF) inhibitors, based on data from 239 individuals.
Use of TNF-alpha inhibitors improves treatment response for many arthritis patients but dosage is rarely adjusted on an individual level, which may lead to unnecessary overdosing in some patients, Mogens Pfeiffer-Jensen, MD, of Aarhus (Denmark) University Hospital, and colleagues wrote.
Data from previous studies suggest that therapeutic drug monitoring (TDM) based on serum trough levels may allow for dose optimization and dose reduction in inflammatory bowel disease patients, but data in patients with arthritis are lacking, they wrote.
In a study published in the Scandinavian Journal of Rheumatology, the researchers recruited 99 patients with RA, 48 with psoriatic arthritis (PsA), and 92 with spondyloarthritis (SpA). The participants were randomized to standard care or standard care plus TDM. Serum trough levels were assessed at baseline and at every 4 months, and prescription changes or drug switches were implemented based on these levels. At baseline, 81 patients were being treated with infliximab (Remicade and biosimilars), 79 with etanercept (Enbrel), and 79 with adalimumab (Humira).
The primary endpoint was reduced drug prescription after 48 weeks.
Overall, TDM significantly reduced prescription of infliximab by 12% (P = .001) and prescription of etanercept by 15% (P = .01), compared with standard care. TDM also prolonged the interdosing intervals of etanercept by 235% (P = .02) and of adalimumab by 28% (P = .04), compared with standard care.
TDM patients taking infliximab had more frequent dose reduction and less frequent dose increases during and after the study when compared with patients who stayed with standard care; similar trends were seen with adalimumab. TDM also accelerated the switch to other biologics for patients on all three medications.
No significant differences occurred in adverse events or hospitalizations between the TDM and standard care patients.
Clinical composite scores (Disease Activity Score based on 28 joints with C-reactive protein) were reduced in patients with RA and PsA who were taking adalimumab and randomized to TDM, but no other clinical outcome differences were noted. Scores on the Health Assessment Questionnaire and global Visual Analog Scale for pain were significantly lower in patients in the TDM group who were taking infliximab and adalimumab, “indicating equally or superior sustained remission across diagnoses,” the researchers emphasized.
The findings were limited by several factors, including the variations in pathophysiology and open-label design. “However, since the TDM was based on an objective serum value and decision procedures were clear, we do not consider the potential of unconscious bias to outweigh the benefits of dose-changing abilities,” they wrote.
The researchers expressed surprise that the reduced use of TNF-alpha inhibitors did not significantly reduce adverse events or serious adverse events, compared with standard care, but they proposed that standard of care may have taken adverse events into account, because all patients had received prescriptions at least 3 months before the study.
As for clinical implications, the current costs of the biochemical assays necessary for TDM may be a barrier to implementing TDM as a standard part of daily clinical practice, the researchers added. However, the study was strengthened by the inclusion of patients with RA, PsA, and SpA, and is the first known to include patients receiving etanercept or adalimumab in an examination of TDM.
“Our data support TDM based solely on serum trough levels in [TNF-alpha inhibitors] with different pharmacokinetics as a future key player in personalized medicine for chronic rheumatoid diseases treated with biologics,” they concluded.
The study was supported by Spydspidspuljen, Region Midt, Denmark, and Department of Rheumatology, Aarhus University Hospital. The researchers had no financial conflicts to disclose.
Therapeutic drug monitoring allowed patients with rheumatoid arthritis, psoriatic arthritis, and spondyloarthritis to reduce their dosage of tumor necrosis factor–alpha (TNF) inhibitors, based on data from 239 individuals.
Use of TNF-alpha inhibitors improves treatment response for many arthritis patients but dosage is rarely adjusted on an individual level, which may lead to unnecessary overdosing in some patients, Mogens Pfeiffer-Jensen, MD, of Aarhus (Denmark) University Hospital, and colleagues wrote.
Data from previous studies suggest that therapeutic drug monitoring (TDM) based on serum trough levels may allow for dose optimization and dose reduction in inflammatory bowel disease patients, but data in patients with arthritis are lacking, they wrote.
In a study published in the Scandinavian Journal of Rheumatology, the researchers recruited 99 patients with RA, 48 with psoriatic arthritis (PsA), and 92 with spondyloarthritis (SpA). The participants were randomized to standard care or standard care plus TDM. Serum trough levels were assessed at baseline and at every 4 months, and prescription changes or drug switches were implemented based on these levels. At baseline, 81 patients were being treated with infliximab (Remicade and biosimilars), 79 with etanercept (Enbrel), and 79 with adalimumab (Humira).
The primary endpoint was reduced drug prescription after 48 weeks.
Overall, TDM significantly reduced prescription of infliximab by 12% (P = .001) and prescription of etanercept by 15% (P = .01), compared with standard care. TDM also prolonged the interdosing intervals of etanercept by 235% (P = .02) and of adalimumab by 28% (P = .04), compared with standard care.
TDM patients taking infliximab had more frequent dose reduction and less frequent dose increases during and after the study when compared with patients who stayed with standard care; similar trends were seen with adalimumab. TDM also accelerated the switch to other biologics for patients on all three medications.
No significant differences occurred in adverse events or hospitalizations between the TDM and standard care patients.
Clinical composite scores (Disease Activity Score based on 28 joints with C-reactive protein) were reduced in patients with RA and PsA who were taking adalimumab and randomized to TDM, but no other clinical outcome differences were noted. Scores on the Health Assessment Questionnaire and global Visual Analog Scale for pain were significantly lower in patients in the TDM group who were taking infliximab and adalimumab, “indicating equally or superior sustained remission across diagnoses,” the researchers emphasized.
The findings were limited by several factors, including the variations in pathophysiology and open-label design. “However, since the TDM was based on an objective serum value and decision procedures were clear, we do not consider the potential of unconscious bias to outweigh the benefits of dose-changing abilities,” they wrote.
The researchers expressed surprise that the reduced use of TNF-alpha inhibitors did not significantly reduce adverse events or serious adverse events, compared with standard care, but they proposed that standard of care may have taken adverse events into account, because all patients had received prescriptions at least 3 months before the study.
As for clinical implications, the current costs of the biochemical assays necessary for TDM may be a barrier to implementing TDM as a standard part of daily clinical practice, the researchers added. However, the study was strengthened by the inclusion of patients with RA, PsA, and SpA, and is the first known to include patients receiving etanercept or adalimumab in an examination of TDM.
“Our data support TDM based solely on serum trough levels in [TNF-alpha inhibitors] with different pharmacokinetics as a future key player in personalized medicine for chronic rheumatoid diseases treated with biologics,” they concluded.
The study was supported by Spydspidspuljen, Region Midt, Denmark, and Department of Rheumatology, Aarhus University Hospital. The researchers had no financial conflicts to disclose.
FROM THE SCANDINAVIAN JOURNAL OF RHEUMATOLOGY