User login
Some women use prescription opioids during pregnancy
and almost a third of those women did not receive counseling from a provider on the effects of opioids on their unborn children, according to analysis from the Centers for Disease Control and Prevention.
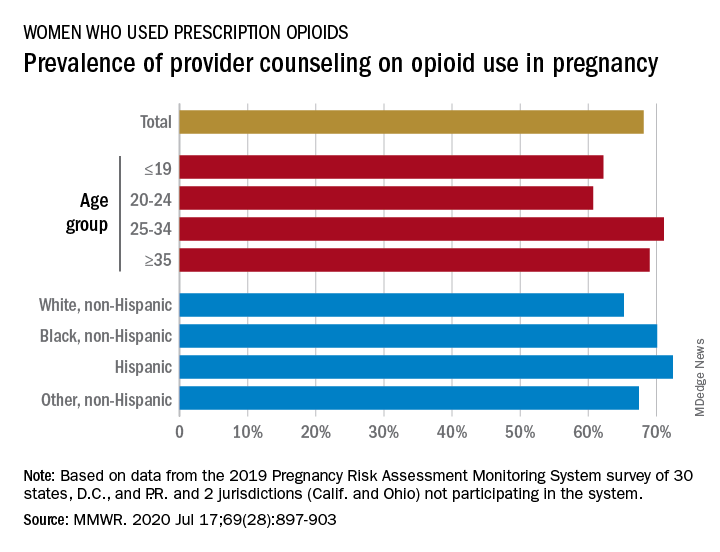
Data from the Pregnancy Risk Assessment Monitoring System 2019 survey show that 7% of the nearly 21,000 respondents reported using an opioid pain reliever during pregnancy, considerably lower than the fill rates of 14%-22% seen in studies of pharmacy dispensing, Jean Y. Ko, PhD, and associates at the CDC said in the Morbidity and Mortality Weekly Report.
In the current analysis, opioid use during pregnancy varied by age – the rate was highest, 10%, in those aged 19 years and under and dropped as age increased to 6% among those aged 35 and older – and by race/ethnicity – 9% of black women reported use, compared with 7% of Hispanics, 6% of whites, and 7% of all others, the investigators reported.
Use of prescription opioids was significantly higher for two specific groups. Women who smoked cigarettes during the last 3 months of their pregnancy had a 16% rate of opioid use, and those with depression during pregnancy had a rate of 13%, they said.
Physicians caring for pregnant women should seek to identify and address substance use and misuse, and mental health conditions such as depression, history of trauma, posttraumatic stress disorder, and anxiety, the CDC researchers pointed out.
The CDC and the American College of Obstetricians and Gynecologists both recommend that caregivers and patients also need to “discuss and carefully weigh risks and benefits when considering initiation of opioid therapy for chronic pain during pregnancy,” Dr. Ko and associates wrote.
That sort of counseling, however, was not always offered: 32% of the women with self-reported prescription opioid use during their pregnancy said that they had not been counseled about the drugs’ effect on an infant. Some variation was seen by age or race/ethnicity, but the differences were not significant, the researchers reported.
“Opioid prescribing consistent with clinical practice guidelines can ensure that patients, particularly those who are pregnant, have access to safer, more effective chronic pain treatment and reduce the number of persons at risk for opioid misuse, opioid use disorder, and overdose,” the investigators concluded.
Survey data from 32 jurisdictions (30 states, along with the District of Columbia and Puerto Rico) that participate in the monitoring system were included in the analysis, as were data from California and Ohio, which do not participate. All of the respondents had a live birth in the preceding 2-6 months, the researchers explained.
SOURCE: Ko JY et al. MMWR. 2020 Jul 17;69(28):897-903.
and almost a third of those women did not receive counseling from a provider on the effects of opioids on their unborn children, according to analysis from the Centers for Disease Control and Prevention.

Data from the Pregnancy Risk Assessment Monitoring System 2019 survey show that 7% of the nearly 21,000 respondents reported using an opioid pain reliever during pregnancy, considerably lower than the fill rates of 14%-22% seen in studies of pharmacy dispensing, Jean Y. Ko, PhD, and associates at the CDC said in the Morbidity and Mortality Weekly Report.
In the current analysis, opioid use during pregnancy varied by age – the rate was highest, 10%, in those aged 19 years and under and dropped as age increased to 6% among those aged 35 and older – and by race/ethnicity – 9% of black women reported use, compared with 7% of Hispanics, 6% of whites, and 7% of all others, the investigators reported.
Use of prescription opioids was significantly higher for two specific groups. Women who smoked cigarettes during the last 3 months of their pregnancy had a 16% rate of opioid use, and those with depression during pregnancy had a rate of 13%, they said.
Physicians caring for pregnant women should seek to identify and address substance use and misuse, and mental health conditions such as depression, history of trauma, posttraumatic stress disorder, and anxiety, the CDC researchers pointed out.
The CDC and the American College of Obstetricians and Gynecologists both recommend that caregivers and patients also need to “discuss and carefully weigh risks and benefits when considering initiation of opioid therapy for chronic pain during pregnancy,” Dr. Ko and associates wrote.
That sort of counseling, however, was not always offered: 32% of the women with self-reported prescription opioid use during their pregnancy said that they had not been counseled about the drugs’ effect on an infant. Some variation was seen by age or race/ethnicity, but the differences were not significant, the researchers reported.
“Opioid prescribing consistent with clinical practice guidelines can ensure that patients, particularly those who are pregnant, have access to safer, more effective chronic pain treatment and reduce the number of persons at risk for opioid misuse, opioid use disorder, and overdose,” the investigators concluded.
Survey data from 32 jurisdictions (30 states, along with the District of Columbia and Puerto Rico) that participate in the monitoring system were included in the analysis, as were data from California and Ohio, which do not participate. All of the respondents had a live birth in the preceding 2-6 months, the researchers explained.
SOURCE: Ko JY et al. MMWR. 2020 Jul 17;69(28):897-903.
and almost a third of those women did not receive counseling from a provider on the effects of opioids on their unborn children, according to analysis from the Centers for Disease Control and Prevention.

Data from the Pregnancy Risk Assessment Monitoring System 2019 survey show that 7% of the nearly 21,000 respondents reported using an opioid pain reliever during pregnancy, considerably lower than the fill rates of 14%-22% seen in studies of pharmacy dispensing, Jean Y. Ko, PhD, and associates at the CDC said in the Morbidity and Mortality Weekly Report.
In the current analysis, opioid use during pregnancy varied by age – the rate was highest, 10%, in those aged 19 years and under and dropped as age increased to 6% among those aged 35 and older – and by race/ethnicity – 9% of black women reported use, compared with 7% of Hispanics, 6% of whites, and 7% of all others, the investigators reported.
Use of prescription opioids was significantly higher for two specific groups. Women who smoked cigarettes during the last 3 months of their pregnancy had a 16% rate of opioid use, and those with depression during pregnancy had a rate of 13%, they said.
Physicians caring for pregnant women should seek to identify and address substance use and misuse, and mental health conditions such as depression, history of trauma, posttraumatic stress disorder, and anxiety, the CDC researchers pointed out.
The CDC and the American College of Obstetricians and Gynecologists both recommend that caregivers and patients also need to “discuss and carefully weigh risks and benefits when considering initiation of opioid therapy for chronic pain during pregnancy,” Dr. Ko and associates wrote.
That sort of counseling, however, was not always offered: 32% of the women with self-reported prescription opioid use during their pregnancy said that they had not been counseled about the drugs’ effect on an infant. Some variation was seen by age or race/ethnicity, but the differences were not significant, the researchers reported.
“Opioid prescribing consistent with clinical practice guidelines can ensure that patients, particularly those who are pregnant, have access to safer, more effective chronic pain treatment and reduce the number of persons at risk for opioid misuse, opioid use disorder, and overdose,” the investigators concluded.
Survey data from 32 jurisdictions (30 states, along with the District of Columbia and Puerto Rico) that participate in the monitoring system were included in the analysis, as were data from California and Ohio, which do not participate. All of the respondents had a live birth in the preceding 2-6 months, the researchers explained.
SOURCE: Ko JY et al. MMWR. 2020 Jul 17;69(28):897-903.
FROM MMWR
Limit customized compounded hormones to special circumstances
The use of compounded bioidentical hormone therapies should be limited to patients who are not able to use a hormone therapy product approved by the Food and Drug Administration for reasons of allergy or dosage, according to a new report from the National Academies of Sciences, Engineering, and Medicine.
In recent years, compounded bioidentical hormone therapies (cBHTs) have been “marketed as a personalized and natural approach to enhanced wellness using tailored preparations that address a myriad of symptoms, including those associated with menopause and aging,” wrote Donald R. Mattison, MD, of the University of Ottawa, and chair of the committee charged with producing the report, and colleagues.
Although both cBHTs and bioidentical hormone therapies (BHTs) contain hormones that are structurally and chemically identical to those in the human body, cBHTs have not undergone the safety, efficacy, and quality control tests of approved FDA products, according to the report.
In addition, cBHTs have no standardization when it comes to medication doses, and the products often are available in topicals such as creams or ointments, as well as pills or pellets. The lack of standards in dosing or form can contribute to the risk of overdose, the report emphasized.
Various cBTH products continue to be marketed to the public for age-related hormone symptoms including hot flashes associated with menopause and decreased muscle mass associated with decreased testosterone. However, cBHTs are not approved by the FDA in part because the individually mixed products are not tested to verify the amount of hormone that may be absorbed.
In response to the increased use of cBHTs, the National Academies convened a Committee on the Clinical Utility of Treating Patients with Compounded Bioidentical Hormone Replacement Therapy and commissioned a report.
The two typical reasons to prescribe cBHT are either to provide a medication in an alternate dose not available in approved products or to omit components of a medication to which a patient is allergic, according to the report.
The report includes an algorithm to help guide clinicians in prescribing FDA-approved products, including off-label use of approved products, before cBHT products. “There is a dearth of high-quality evidence ... available to establish whether cBHT preparations are safe or efficacious for their prescribed uses,” the report states.
Of note, the committee also found no guidelines to recommend the use of cBHT products as a substitute for off-label use of FDA-approved BHT products for patients with female sexual dysfunction or gender dysphoria, two conditions for which no FDA-approved BHT products exist.
“The North American Menopause Society applauds the efforts of the National Academies of Sciences, Engineering, and Medicine (NASEM) and endorses their recommendations on compounded bioidentical hormone therapy,” Stephanie S. Faubion, MD, medical director of The North American Menopause Society, wrote in a statement. “As a society, we remain committed to improving the care of midlife women through the promotion of evidence-based research, education, and clinical care.”
A report on the use of cBHTs was important at this time because of the widespread and largely unregulated use of these products with little data to support their safety and efficacy, Dr. Faubion said in an interview.
“There are no indications for use of custom compounded hormone therapy aside from an allergy to a component in the FDA-approved products or lack of availability of the needed dose, which would be exceedingly rare given the variety of forms and doses available with FDA-approved products,” she said.
Main concerns regarding the use of cBHTs are the lack of safety and efficacy data, Dr. Faubion emphasized. “Women believe these products are safer than FDA-approved products because they do not receive a package insert outlining potential risks as they do with FDA-approved products.” A lack of data and safety monitoring of cBHTs means that adverse effects are not monitored and reported, she said. Also, safety concerns persist regarding some forms of cBHTs such as pellets, which were specifically highlighted in the report.
Dr. Faubion said that she “absolutely” agrees with the report’s limited circumstances in which the used of cBHTs would be appropriate. “There are very few reasons why women would need to use compounded hormones instead of the FDA-approved versions, which are regulated for quality, efficacy and safety, readily available in the local pharmacy, and often covered by insurance.”
In terms of the future, “we need more education for women as consumers and for medical providers on this topic,” Dr. Faubion noted. Also, “clearly, there is a dearth of research on the true efficacy and safety of these compounded hormone therapy products.”
The statement from the National Academies crystallizes what experts have been saying for decades, according to Lubna Pal, MBBS, director of the menopause program at Yale University, New Haven, Conn.
The formal recommendations to limit the use of cBHTs “are not novel, but certainly needed,” and the statement “offers guidance regardless of your specialty,” Dr. Pal said in an interview.
There is often a disconnect between consumers’ understanding of compounding and the reality of safety concerns, she said. “We are in a tabloid era,” and education is key to guiding patients toward the FDA-approved treatments with safety data and demonstrated effectiveness, she said. “Safety should be the driving factor.” In compounded products, “there is no consistency that what you get today is the same as what you get tomorrow,” and the lack of standardization of cBHTs increases the risk for adverse events, she emphasized.
For patients with special needs such as allergies or other specialized dosing requirements, as noted in the National Academies statement, clinicians should discuss the options with patients and monitor them regularly to head off potential adverse events such as the development of uterine cancer, said Dr. Pal, who is a member of the Ob.Gyn. News editorial advisory board.
The research involved in creating the report was supported by the Food and Drug Administration.
Dr. Faubion had no financial conflicts to disclose. Dr. Pal had no relevant financial disclosures.
SOURCE: Mattison DR et al.; National Academies of Sciences, Engineering, and Medicine. The clinical utility of compounded bioidentical hormone therapy: A review of safety, effectiveness, and use. (Washington, DC: The National Academies Press. 2020.)
The use of compounded bioidentical hormone therapies should be limited to patients who are not able to use a hormone therapy product approved by the Food and Drug Administration for reasons of allergy or dosage, according to a new report from the National Academies of Sciences, Engineering, and Medicine.
In recent years, compounded bioidentical hormone therapies (cBHTs) have been “marketed as a personalized and natural approach to enhanced wellness using tailored preparations that address a myriad of symptoms, including those associated with menopause and aging,” wrote Donald R. Mattison, MD, of the University of Ottawa, and chair of the committee charged with producing the report, and colleagues.
Although both cBHTs and bioidentical hormone therapies (BHTs) contain hormones that are structurally and chemically identical to those in the human body, cBHTs have not undergone the safety, efficacy, and quality control tests of approved FDA products, according to the report.
In addition, cBHTs have no standardization when it comes to medication doses, and the products often are available in topicals such as creams or ointments, as well as pills or pellets. The lack of standards in dosing or form can contribute to the risk of overdose, the report emphasized.
Various cBTH products continue to be marketed to the public for age-related hormone symptoms including hot flashes associated with menopause and decreased muscle mass associated with decreased testosterone. However, cBHTs are not approved by the FDA in part because the individually mixed products are not tested to verify the amount of hormone that may be absorbed.
In response to the increased use of cBHTs, the National Academies convened a Committee on the Clinical Utility of Treating Patients with Compounded Bioidentical Hormone Replacement Therapy and commissioned a report.
The two typical reasons to prescribe cBHT are either to provide a medication in an alternate dose not available in approved products or to omit components of a medication to which a patient is allergic, according to the report.
The report includes an algorithm to help guide clinicians in prescribing FDA-approved products, including off-label use of approved products, before cBHT products. “There is a dearth of high-quality evidence ... available to establish whether cBHT preparations are safe or efficacious for their prescribed uses,” the report states.
Of note, the committee also found no guidelines to recommend the use of cBHT products as a substitute for off-label use of FDA-approved BHT products for patients with female sexual dysfunction or gender dysphoria, two conditions for which no FDA-approved BHT products exist.
“The North American Menopause Society applauds the efforts of the National Academies of Sciences, Engineering, and Medicine (NASEM) and endorses their recommendations on compounded bioidentical hormone therapy,” Stephanie S. Faubion, MD, medical director of The North American Menopause Society, wrote in a statement. “As a society, we remain committed to improving the care of midlife women through the promotion of evidence-based research, education, and clinical care.”
A report on the use of cBHTs was important at this time because of the widespread and largely unregulated use of these products with little data to support their safety and efficacy, Dr. Faubion said in an interview.
“There are no indications for use of custom compounded hormone therapy aside from an allergy to a component in the FDA-approved products or lack of availability of the needed dose, which would be exceedingly rare given the variety of forms and doses available with FDA-approved products,” she said.
Main concerns regarding the use of cBHTs are the lack of safety and efficacy data, Dr. Faubion emphasized. “Women believe these products are safer than FDA-approved products because they do not receive a package insert outlining potential risks as they do with FDA-approved products.” A lack of data and safety monitoring of cBHTs means that adverse effects are not monitored and reported, she said. Also, safety concerns persist regarding some forms of cBHTs such as pellets, which were specifically highlighted in the report.
Dr. Faubion said that she “absolutely” agrees with the report’s limited circumstances in which the used of cBHTs would be appropriate. “There are very few reasons why women would need to use compounded hormones instead of the FDA-approved versions, which are regulated for quality, efficacy and safety, readily available in the local pharmacy, and often covered by insurance.”
In terms of the future, “we need more education for women as consumers and for medical providers on this topic,” Dr. Faubion noted. Also, “clearly, there is a dearth of research on the true efficacy and safety of these compounded hormone therapy products.”
The statement from the National Academies crystallizes what experts have been saying for decades, according to Lubna Pal, MBBS, director of the menopause program at Yale University, New Haven, Conn.
The formal recommendations to limit the use of cBHTs “are not novel, but certainly needed,” and the statement “offers guidance regardless of your specialty,” Dr. Pal said in an interview.
There is often a disconnect between consumers’ understanding of compounding and the reality of safety concerns, she said. “We are in a tabloid era,” and education is key to guiding patients toward the FDA-approved treatments with safety data and demonstrated effectiveness, she said. “Safety should be the driving factor.” In compounded products, “there is no consistency that what you get today is the same as what you get tomorrow,” and the lack of standardization of cBHTs increases the risk for adverse events, she emphasized.
For patients with special needs such as allergies or other specialized dosing requirements, as noted in the National Academies statement, clinicians should discuss the options with patients and monitor them regularly to head off potential adverse events such as the development of uterine cancer, said Dr. Pal, who is a member of the Ob.Gyn. News editorial advisory board.
The research involved in creating the report was supported by the Food and Drug Administration.
Dr. Faubion had no financial conflicts to disclose. Dr. Pal had no relevant financial disclosures.
SOURCE: Mattison DR et al.; National Academies of Sciences, Engineering, and Medicine. The clinical utility of compounded bioidentical hormone therapy: A review of safety, effectiveness, and use. (Washington, DC: The National Academies Press. 2020.)
The use of compounded bioidentical hormone therapies should be limited to patients who are not able to use a hormone therapy product approved by the Food and Drug Administration for reasons of allergy or dosage, according to a new report from the National Academies of Sciences, Engineering, and Medicine.
In recent years, compounded bioidentical hormone therapies (cBHTs) have been “marketed as a personalized and natural approach to enhanced wellness using tailored preparations that address a myriad of symptoms, including those associated with menopause and aging,” wrote Donald R. Mattison, MD, of the University of Ottawa, and chair of the committee charged with producing the report, and colleagues.
Although both cBHTs and bioidentical hormone therapies (BHTs) contain hormones that are structurally and chemically identical to those in the human body, cBHTs have not undergone the safety, efficacy, and quality control tests of approved FDA products, according to the report.
In addition, cBHTs have no standardization when it comes to medication doses, and the products often are available in topicals such as creams or ointments, as well as pills or pellets. The lack of standards in dosing or form can contribute to the risk of overdose, the report emphasized.
Various cBTH products continue to be marketed to the public for age-related hormone symptoms including hot flashes associated with menopause and decreased muscle mass associated with decreased testosterone. However, cBHTs are not approved by the FDA in part because the individually mixed products are not tested to verify the amount of hormone that may be absorbed.
In response to the increased use of cBHTs, the National Academies convened a Committee on the Clinical Utility of Treating Patients with Compounded Bioidentical Hormone Replacement Therapy and commissioned a report.
The two typical reasons to prescribe cBHT are either to provide a medication in an alternate dose not available in approved products or to omit components of a medication to which a patient is allergic, according to the report.
The report includes an algorithm to help guide clinicians in prescribing FDA-approved products, including off-label use of approved products, before cBHT products. “There is a dearth of high-quality evidence ... available to establish whether cBHT preparations are safe or efficacious for their prescribed uses,” the report states.
Of note, the committee also found no guidelines to recommend the use of cBHT products as a substitute for off-label use of FDA-approved BHT products for patients with female sexual dysfunction or gender dysphoria, two conditions for which no FDA-approved BHT products exist.
“The North American Menopause Society applauds the efforts of the National Academies of Sciences, Engineering, and Medicine (NASEM) and endorses their recommendations on compounded bioidentical hormone therapy,” Stephanie S. Faubion, MD, medical director of The North American Menopause Society, wrote in a statement. “As a society, we remain committed to improving the care of midlife women through the promotion of evidence-based research, education, and clinical care.”
A report on the use of cBHTs was important at this time because of the widespread and largely unregulated use of these products with little data to support their safety and efficacy, Dr. Faubion said in an interview.
“There are no indications for use of custom compounded hormone therapy aside from an allergy to a component in the FDA-approved products or lack of availability of the needed dose, which would be exceedingly rare given the variety of forms and doses available with FDA-approved products,” she said.
Main concerns regarding the use of cBHTs are the lack of safety and efficacy data, Dr. Faubion emphasized. “Women believe these products are safer than FDA-approved products because they do not receive a package insert outlining potential risks as they do with FDA-approved products.” A lack of data and safety monitoring of cBHTs means that adverse effects are not monitored and reported, she said. Also, safety concerns persist regarding some forms of cBHTs such as pellets, which were specifically highlighted in the report.
Dr. Faubion said that she “absolutely” agrees with the report’s limited circumstances in which the used of cBHTs would be appropriate. “There are very few reasons why women would need to use compounded hormones instead of the FDA-approved versions, which are regulated for quality, efficacy and safety, readily available in the local pharmacy, and often covered by insurance.”
In terms of the future, “we need more education for women as consumers and for medical providers on this topic,” Dr. Faubion noted. Also, “clearly, there is a dearth of research on the true efficacy and safety of these compounded hormone therapy products.”
The statement from the National Academies crystallizes what experts have been saying for decades, according to Lubna Pal, MBBS, director of the menopause program at Yale University, New Haven, Conn.
The formal recommendations to limit the use of cBHTs “are not novel, but certainly needed,” and the statement “offers guidance regardless of your specialty,” Dr. Pal said in an interview.
There is often a disconnect between consumers’ understanding of compounding and the reality of safety concerns, she said. “We are in a tabloid era,” and education is key to guiding patients toward the FDA-approved treatments with safety data and demonstrated effectiveness, she said. “Safety should be the driving factor.” In compounded products, “there is no consistency that what you get today is the same as what you get tomorrow,” and the lack of standardization of cBHTs increases the risk for adverse events, she emphasized.
For patients with special needs such as allergies or other specialized dosing requirements, as noted in the National Academies statement, clinicians should discuss the options with patients and monitor them regularly to head off potential adverse events such as the development of uterine cancer, said Dr. Pal, who is a member of the Ob.Gyn. News editorial advisory board.
The research involved in creating the report was supported by the Food and Drug Administration.
Dr. Faubion had no financial conflicts to disclose. Dr. Pal had no relevant financial disclosures.
SOURCE: Mattison DR et al.; National Academies of Sciences, Engineering, and Medicine. The clinical utility of compounded bioidentical hormone therapy: A review of safety, effectiveness, and use. (Washington, DC: The National Academies Press. 2020.)
Cerclage in twin pregnancies reduces perinatal mortality in randomized trial
according to a randomized controlled trial. The trial, which was published in the American Journal of Obstetrics and Gynecology, included 30 patients at 8 centers. The investigators stopped the trial early because perinatal mortality occurred more often in the group that did not receive the intervention.
The research suggests that a combination of physical exam–indicated cerclage, indomethacin, and antibiotics decreased the incidence of spontaneous preterm birth and prolonged the period from diagnosis to delivery by an average of 5.6 weeks, compared with no cerclage.
“We’ve already incorporated this cerclage into our practice and have been able to offer this to pregnant mothers with twins with great success,” senior author Vincenzo Berghella, MD, said in a news release.
“These results have the potential to change practice and help many more women have healthy twin babies,” said Dr. Berghella, director of the division of maternal fetal medicine at Thomas Jefferson University in Philadelphia.
A shift in perspective
More research is needed to establish a standardized approach, but the trial should “open physicians’ perspectives to think about how, in selected cases and with the proper approach, cerclage can work well,” said Ozhan M. Turan, MD, PhD, director of the division of maternal and fetal medicine and director of fetal therapy and complex obstetric surgery at University of Maryland in Baltimore.
Although many physicians use cerclage for twin pregnancies in select situations, the practice is not well established. “If you look at the guidelines or books, mostly everyone thinks that doing a cerclage in twins is not a good idea,” Dr. Turan said in an interview.
In the present trial, the researchers controlled for many factors and carefully selected patients with no signs of preterm labor or infection. It is not simply a matter of saying, “Do the stitch,” he said. “But it is proven: if you select patients well and use the appropriate approach, then you could improve the outcome.”
The study is the first randomized controlled trial of physical exam–indicated cerclage focused on twins, according to its authors. It enrolled patients between July 2015 and July 2019. In the end, the researchers analyzed data from 30 pregnancies, rather than the originally intended 52. They stopped the trial after a data and safety monitoring board considered it “unethical to continue the study due to the considerable perinatal mortality in one of the arms ... and requested to unmask the arms of the study,” the researchers said.
Perinatal mortality occurred in 18% of neonates in the cerclage group (6 of 34), compared with 77% in the group without cerclage (20 of 26). All perinatal mortality cases were associated with delivery before 24 weeks.
“The small number of participants reflects how rare this condition is among all pregnancies,” first author Amanda Roman, MD, of the division of maternal fetal medicine at Thomas Jefferson University, Philadelphia, said in the news release. “But because women were randomized to treatment and nontreatment groups, the results are strong, as confirmed by the independent data and safety monitoring board.”
The researchers enrolled women with twin pregnancies and asymptomatic cervical dilation from 1 to 4 cm before 24 weeks. Exclusion criteria included monochorionic-monoamniotic pregnancy, selective fetal growth restriction, twin-twin transfusion syndrome, major fetal malformation, known genetic anomaly, placenta previa, signs of labor, or clinical chorioamnionitis.
In all, 17 women were randomized to cerclage and 13 to the no-cerclage group. Both groups had similar patient characteristics. About 93% of the twin gestations were diamniotic-dichorionic. Assisted reproductive technology was used by about 36% of the participants, and 20% had a history of singleton preterm birth. Four women assigned to cerclage did not undergo the procedure but were included in the intention-to-treat analysis. Two of the four patients had contraindications that occurred soon after randomization (rupture of amniotic membranes and vaginal bleeding), one had a friable cervix, and one declined cerclage after being randomized.
Spontaneous preterm birth before 34 weeks of gestation, the primary outcome, occurred in 12 of 17 women in the cerclage group and in all 13 women in the no-cerclage group (70% vs. 100%).
Trial to assess ultrasound indicated cerclage
“Expectant management with no cerclage is the current standard of care for these women,” Dr. Roman and coauthors wrote. “Despite small sample size, we were able to show a significant benefit to physical exam–indicated cerclage.”
Inability to place the cerclage in one patient due to friable cervix was the only intraoperative complication. “Larger cohorts in singleton pregnancies have informed a 10%-20% risk of intraoperative rupture of the membranes, cervical laceration, and bleeding during the procedure,” the researchers noted.
All women who received cerclage also received indomethacin and antibiotics, although these elements of management were not prespecified. Given the relatively small sample size, it is unclear what role factors such as indomethacin, which was administered to 82% of the cerclage group versus 31% of the no-cerclage group, and antibiotics may have played, said Dr. Turan.
Prospective studies may help clarify how the degree of cervical dilation, gestational age, use of progesterone, or surgical techniques may influence outcomes. In addition, the researchers are enrolling patients in another trial. That study aims to assess whether cerclage reduces the incidence of spontaneous preterm birth in asymptomatic women with twin gestations and cervical length of 15 mm or less diagnosed by transvaginal ultrasound between 16 and 24 weeks of gestation.
The study had no external financial support. The authors had no conflicts of interest. Dr. Turan said he had no relevant financial disclosures.
SOURCE: Roman A et al. Am J Obstet Gynecol. 2020 Jun. doi: 10.1016/j.ajog.2020.06.047.
according to a randomized controlled trial. The trial, which was published in the American Journal of Obstetrics and Gynecology, included 30 patients at 8 centers. The investigators stopped the trial early because perinatal mortality occurred more often in the group that did not receive the intervention.
The research suggests that a combination of physical exam–indicated cerclage, indomethacin, and antibiotics decreased the incidence of spontaneous preterm birth and prolonged the period from diagnosis to delivery by an average of 5.6 weeks, compared with no cerclage.
“We’ve already incorporated this cerclage into our practice and have been able to offer this to pregnant mothers with twins with great success,” senior author Vincenzo Berghella, MD, said in a news release.
“These results have the potential to change practice and help many more women have healthy twin babies,” said Dr. Berghella, director of the division of maternal fetal medicine at Thomas Jefferson University in Philadelphia.
A shift in perspective
More research is needed to establish a standardized approach, but the trial should “open physicians’ perspectives to think about how, in selected cases and with the proper approach, cerclage can work well,” said Ozhan M. Turan, MD, PhD, director of the division of maternal and fetal medicine and director of fetal therapy and complex obstetric surgery at University of Maryland in Baltimore.
Although many physicians use cerclage for twin pregnancies in select situations, the practice is not well established. “If you look at the guidelines or books, mostly everyone thinks that doing a cerclage in twins is not a good idea,” Dr. Turan said in an interview.
In the present trial, the researchers controlled for many factors and carefully selected patients with no signs of preterm labor or infection. It is not simply a matter of saying, “Do the stitch,” he said. “But it is proven: if you select patients well and use the appropriate approach, then you could improve the outcome.”
The study is the first randomized controlled trial of physical exam–indicated cerclage focused on twins, according to its authors. It enrolled patients between July 2015 and July 2019. In the end, the researchers analyzed data from 30 pregnancies, rather than the originally intended 52. They stopped the trial after a data and safety monitoring board considered it “unethical to continue the study due to the considerable perinatal mortality in one of the arms ... and requested to unmask the arms of the study,” the researchers said.
Perinatal mortality occurred in 18% of neonates in the cerclage group (6 of 34), compared with 77% in the group without cerclage (20 of 26). All perinatal mortality cases were associated with delivery before 24 weeks.
“The small number of participants reflects how rare this condition is among all pregnancies,” first author Amanda Roman, MD, of the division of maternal fetal medicine at Thomas Jefferson University, Philadelphia, said in the news release. “But because women were randomized to treatment and nontreatment groups, the results are strong, as confirmed by the independent data and safety monitoring board.”
The researchers enrolled women with twin pregnancies and asymptomatic cervical dilation from 1 to 4 cm before 24 weeks. Exclusion criteria included monochorionic-monoamniotic pregnancy, selective fetal growth restriction, twin-twin transfusion syndrome, major fetal malformation, known genetic anomaly, placenta previa, signs of labor, or clinical chorioamnionitis.
In all, 17 women were randomized to cerclage and 13 to the no-cerclage group. Both groups had similar patient characteristics. About 93% of the twin gestations were diamniotic-dichorionic. Assisted reproductive technology was used by about 36% of the participants, and 20% had a history of singleton preterm birth. Four women assigned to cerclage did not undergo the procedure but were included in the intention-to-treat analysis. Two of the four patients had contraindications that occurred soon after randomization (rupture of amniotic membranes and vaginal bleeding), one had a friable cervix, and one declined cerclage after being randomized.
Spontaneous preterm birth before 34 weeks of gestation, the primary outcome, occurred in 12 of 17 women in the cerclage group and in all 13 women in the no-cerclage group (70% vs. 100%).
Trial to assess ultrasound indicated cerclage
“Expectant management with no cerclage is the current standard of care for these women,” Dr. Roman and coauthors wrote. “Despite small sample size, we were able to show a significant benefit to physical exam–indicated cerclage.”
Inability to place the cerclage in one patient due to friable cervix was the only intraoperative complication. “Larger cohorts in singleton pregnancies have informed a 10%-20% risk of intraoperative rupture of the membranes, cervical laceration, and bleeding during the procedure,” the researchers noted.
All women who received cerclage also received indomethacin and antibiotics, although these elements of management were not prespecified. Given the relatively small sample size, it is unclear what role factors such as indomethacin, which was administered to 82% of the cerclage group versus 31% of the no-cerclage group, and antibiotics may have played, said Dr. Turan.
Prospective studies may help clarify how the degree of cervical dilation, gestational age, use of progesterone, or surgical techniques may influence outcomes. In addition, the researchers are enrolling patients in another trial. That study aims to assess whether cerclage reduces the incidence of spontaneous preterm birth in asymptomatic women with twin gestations and cervical length of 15 mm or less diagnosed by transvaginal ultrasound between 16 and 24 weeks of gestation.
The study had no external financial support. The authors had no conflicts of interest. Dr. Turan said he had no relevant financial disclosures.
SOURCE: Roman A et al. Am J Obstet Gynecol. 2020 Jun. doi: 10.1016/j.ajog.2020.06.047.
according to a randomized controlled trial. The trial, which was published in the American Journal of Obstetrics and Gynecology, included 30 patients at 8 centers. The investigators stopped the trial early because perinatal mortality occurred more often in the group that did not receive the intervention.
The research suggests that a combination of physical exam–indicated cerclage, indomethacin, and antibiotics decreased the incidence of spontaneous preterm birth and prolonged the period from diagnosis to delivery by an average of 5.6 weeks, compared with no cerclage.
“We’ve already incorporated this cerclage into our practice and have been able to offer this to pregnant mothers with twins with great success,” senior author Vincenzo Berghella, MD, said in a news release.
“These results have the potential to change practice and help many more women have healthy twin babies,” said Dr. Berghella, director of the division of maternal fetal medicine at Thomas Jefferson University in Philadelphia.
A shift in perspective
More research is needed to establish a standardized approach, but the trial should “open physicians’ perspectives to think about how, in selected cases and with the proper approach, cerclage can work well,” said Ozhan M. Turan, MD, PhD, director of the division of maternal and fetal medicine and director of fetal therapy and complex obstetric surgery at University of Maryland in Baltimore.
Although many physicians use cerclage for twin pregnancies in select situations, the practice is not well established. “If you look at the guidelines or books, mostly everyone thinks that doing a cerclage in twins is not a good idea,” Dr. Turan said in an interview.
In the present trial, the researchers controlled for many factors and carefully selected patients with no signs of preterm labor or infection. It is not simply a matter of saying, “Do the stitch,” he said. “But it is proven: if you select patients well and use the appropriate approach, then you could improve the outcome.”
The study is the first randomized controlled trial of physical exam–indicated cerclage focused on twins, according to its authors. It enrolled patients between July 2015 and July 2019. In the end, the researchers analyzed data from 30 pregnancies, rather than the originally intended 52. They stopped the trial after a data and safety monitoring board considered it “unethical to continue the study due to the considerable perinatal mortality in one of the arms ... and requested to unmask the arms of the study,” the researchers said.
Perinatal mortality occurred in 18% of neonates in the cerclage group (6 of 34), compared with 77% in the group without cerclage (20 of 26). All perinatal mortality cases were associated with delivery before 24 weeks.
“The small number of participants reflects how rare this condition is among all pregnancies,” first author Amanda Roman, MD, of the division of maternal fetal medicine at Thomas Jefferson University, Philadelphia, said in the news release. “But because women were randomized to treatment and nontreatment groups, the results are strong, as confirmed by the independent data and safety monitoring board.”
The researchers enrolled women with twin pregnancies and asymptomatic cervical dilation from 1 to 4 cm before 24 weeks. Exclusion criteria included monochorionic-monoamniotic pregnancy, selective fetal growth restriction, twin-twin transfusion syndrome, major fetal malformation, known genetic anomaly, placenta previa, signs of labor, or clinical chorioamnionitis.
In all, 17 women were randomized to cerclage and 13 to the no-cerclage group. Both groups had similar patient characteristics. About 93% of the twin gestations were diamniotic-dichorionic. Assisted reproductive technology was used by about 36% of the participants, and 20% had a history of singleton preterm birth. Four women assigned to cerclage did not undergo the procedure but were included in the intention-to-treat analysis. Two of the four patients had contraindications that occurred soon after randomization (rupture of amniotic membranes and vaginal bleeding), one had a friable cervix, and one declined cerclage after being randomized.
Spontaneous preterm birth before 34 weeks of gestation, the primary outcome, occurred in 12 of 17 women in the cerclage group and in all 13 women in the no-cerclage group (70% vs. 100%).
Trial to assess ultrasound indicated cerclage
“Expectant management with no cerclage is the current standard of care for these women,” Dr. Roman and coauthors wrote. “Despite small sample size, we were able to show a significant benefit to physical exam–indicated cerclage.”
Inability to place the cerclage in one patient due to friable cervix was the only intraoperative complication. “Larger cohorts in singleton pregnancies have informed a 10%-20% risk of intraoperative rupture of the membranes, cervical laceration, and bleeding during the procedure,” the researchers noted.
All women who received cerclage also received indomethacin and antibiotics, although these elements of management were not prespecified. Given the relatively small sample size, it is unclear what role factors such as indomethacin, which was administered to 82% of the cerclage group versus 31% of the no-cerclage group, and antibiotics may have played, said Dr. Turan.
Prospective studies may help clarify how the degree of cervical dilation, gestational age, use of progesterone, or surgical techniques may influence outcomes. In addition, the researchers are enrolling patients in another trial. That study aims to assess whether cerclage reduces the incidence of spontaneous preterm birth in asymptomatic women with twin gestations and cervical length of 15 mm or less diagnosed by transvaginal ultrasound between 16 and 24 weeks of gestation.
The study had no external financial support. The authors had no conflicts of interest. Dr. Turan said he had no relevant financial disclosures.
SOURCE: Roman A et al. Am J Obstet Gynecol. 2020 Jun. doi: 10.1016/j.ajog.2020.06.047.
FROM THE AMERICAN JOURNAL OF OBSTETRICS AND GYNECOLOGY
One-week postsurgical interval for voiding trial increases pass rate
Women who underwent vaginal prolapse surgery and did not immediately have a successful voiding trial were seven times more likely to pass their second voiding trial if their follow-up was 7 days after surgery instead of 4 days, according to a study in the American Journal of Obstetrics and Gynecology.
“This information is useful for setting expectations and for counseling patients on when it might be best to repeat a voiding trial in those with transient incomplete bladder emptying on the day of surgery, especially for those who may not live close to their surgeon, or for those who have difficulty traveling to the office,” said Jeffrey S. Schachar, MD, of Wake Forest Baptist Health in Winston-Salem, N.C., and colleagues. “Despite a higher rate of initial unsuccessful office voiding trials, however, the early group did have significantly fewer days with an indwelling transurethral catheter, as well as total catheterization days,” including self-catheterization.
The researchers note that rates of temporary use of catheters after surgery vary widely, from 12% to 83%, likely because no consensus exists on how long to wait for voiding trials and what constitutes a successful trial.
“It is critical to identify patients with incomplete bladder emptying in order to prevent pain, myogenic and neurogenic damage, ureteral reflux and bladder overdistension that may further impair voiding function,” the authors wrote. “However, extending bladder drainage beyond the necessary recovery period may be associated with higher rates of urinary tract infection (UTI) and patient bother.”
To learn more about the best duration for postoperative catheter use, the researchers enrolled 102 patients before they underwent vaginal prolapse surgery at Wake Forest Baptist Health and Cleveland Clinic Florida from February 2017 to November 2019. The 29 patients with a successful voiding trial within 6 hours after surgery left the study, and 5 others were excluded for needing longer vaginal packing.
The voiding trial involved helping the patient stand to drain the bladder via the catheter, backfilling the bladder with 300 mL of saline solution through the catheter, removing the catheter to give women 1 hour to urinate, and then measuring the postvoid residual with a catheter or ultrasound. At least 100 mL postvoid residual was considered persistent incomplete bladder emptying.
The 60 remaining patients who did not pass the initial voiding trial and opted to remain in the study received a transurethral indwelling catheter and were randomly assigned to return for a second voiding trial either 2-4 days after surgery (depending on day of the week) or 7 days after surgery. The groups were demographically and clinically similar, with predominantly white postmenopausal, non-smoking women with stage II or III multicompartment pelvic organ prolapse.
Women without successful trials could continue with the transurethral catheter or give themselves intermittent catheterizations with a follow-up schedule determined by their surgeon. The researchers then tracked the women for 6 weeks to determine the rate of unsuccessful repeat voiding trials.
Among the women who returned 2-4 days post surgery, 23% had unsuccessful follow-up voiding trials, compared with 3% in the group returning 7 days after surgery (relative risk = 7; P = .02). The researchers calculated that one case of persistent postoperative incomplete bladder emptying was prevented for every five patients who used a catheter for 7 days after surgery.
Kevin A. Ault, MD, professor of obstetrics and gynecology at the University of Kansas Medical Center in Kansas City, said the study was well done, although the findings were unsurprising. He said the clinical implication is straightforward – to wait a week before doing a second voiding trial.
“I suspect these findings match the clinical experience of many surgeons. It is always good to see a well-done clinical trial on a topic,” Dr Ault said in an interview. “The most notable finding is how this impacts patient counseling. Gynecologists should tell their patients that it will take a week with a catheter when this problem arises.”
“The main limitation is whether this finding can be extrapolated to other gynecological surgeries, such as hysterectomy,” said Dr. Ault, who was not involved in the study. “Urinary retention is likely less common after that surgery, but it is still bothersome to patients.”
Dr. Schachar and associates also reported that patients in the earlier group “used significantly more morphine dose equivalents within 24 hours of the office voiding trial than the late-voiding trial group, which was expected given the proximity to surgery” (3 vs. 0.38; P = .005). However, new postoperative pain medication prescriptions and refills were similar in both groups.
Secondary endpoints included UTI rates, total days with a catheter, and patient experience of discomfort with the catheter. The two groups of women reported similar levels of catheter bother, but there was a nonsignificant difference in UTI rates: 23% in the earlier group, compared with 7% in the later group (P = .07).
The early-voiding trial group had an average 5 days with an indwelling transurethral catheter, compared with a significantly different 7 days in the later group (P = .0007). The early group also had fewer total days with an indwelling transurethral catheter and self-catheterization (6 days), compared with the late group (7 days; P = .0013). No patients had persistent incomplete bladder emptying after 17 days post surgery.
“Being able to adequately predict which patients are more likely to have unsuccessful postoperative voiding trials allows surgeons to better counsel their patients and may guide clinical decisions,” Dr. Schachar and associates said. They acknowledged, however, that their study’s biggest weakness is the small enrollment, which led to larger confidence intervals related to relative risk differences between the groups.
The study did not use external funding. Four of the investigators received grant, research funding, or honoraria from one or many medical device or pharmaceutical companies. The remaining researchers had no disclosures. Dr. Ault said he had no relevant financial disclosures.
SOURCE: Schachar JS et al. Am J Obstet Gynecol. 2020 Jun. doi: 10.1016/j.ajog.2020.06.001.
Women who underwent vaginal prolapse surgery and did not immediately have a successful voiding trial were seven times more likely to pass their second voiding trial if their follow-up was 7 days after surgery instead of 4 days, according to a study in the American Journal of Obstetrics and Gynecology.
“This information is useful for setting expectations and for counseling patients on when it might be best to repeat a voiding trial in those with transient incomplete bladder emptying on the day of surgery, especially for those who may not live close to their surgeon, or for those who have difficulty traveling to the office,” said Jeffrey S. Schachar, MD, of Wake Forest Baptist Health in Winston-Salem, N.C., and colleagues. “Despite a higher rate of initial unsuccessful office voiding trials, however, the early group did have significantly fewer days with an indwelling transurethral catheter, as well as total catheterization days,” including self-catheterization.
The researchers note that rates of temporary use of catheters after surgery vary widely, from 12% to 83%, likely because no consensus exists on how long to wait for voiding trials and what constitutes a successful trial.
“It is critical to identify patients with incomplete bladder emptying in order to prevent pain, myogenic and neurogenic damage, ureteral reflux and bladder overdistension that may further impair voiding function,” the authors wrote. “However, extending bladder drainage beyond the necessary recovery period may be associated with higher rates of urinary tract infection (UTI) and patient bother.”
To learn more about the best duration for postoperative catheter use, the researchers enrolled 102 patients before they underwent vaginal prolapse surgery at Wake Forest Baptist Health and Cleveland Clinic Florida from February 2017 to November 2019. The 29 patients with a successful voiding trial within 6 hours after surgery left the study, and 5 others were excluded for needing longer vaginal packing.
The voiding trial involved helping the patient stand to drain the bladder via the catheter, backfilling the bladder with 300 mL of saline solution through the catheter, removing the catheter to give women 1 hour to urinate, and then measuring the postvoid residual with a catheter or ultrasound. At least 100 mL postvoid residual was considered persistent incomplete bladder emptying.
The 60 remaining patients who did not pass the initial voiding trial and opted to remain in the study received a transurethral indwelling catheter and were randomly assigned to return for a second voiding trial either 2-4 days after surgery (depending on day of the week) or 7 days after surgery. The groups were demographically and clinically similar, with predominantly white postmenopausal, non-smoking women with stage II or III multicompartment pelvic organ prolapse.
Women without successful trials could continue with the transurethral catheter or give themselves intermittent catheterizations with a follow-up schedule determined by their surgeon. The researchers then tracked the women for 6 weeks to determine the rate of unsuccessful repeat voiding trials.
Among the women who returned 2-4 days post surgery, 23% had unsuccessful follow-up voiding trials, compared with 3% in the group returning 7 days after surgery (relative risk = 7; P = .02). The researchers calculated that one case of persistent postoperative incomplete bladder emptying was prevented for every five patients who used a catheter for 7 days after surgery.
Kevin A. Ault, MD, professor of obstetrics and gynecology at the University of Kansas Medical Center in Kansas City, said the study was well done, although the findings were unsurprising. He said the clinical implication is straightforward – to wait a week before doing a second voiding trial.
“I suspect these findings match the clinical experience of many surgeons. It is always good to see a well-done clinical trial on a topic,” Dr Ault said in an interview. “The most notable finding is how this impacts patient counseling. Gynecologists should tell their patients that it will take a week with a catheter when this problem arises.”
“The main limitation is whether this finding can be extrapolated to other gynecological surgeries, such as hysterectomy,” said Dr. Ault, who was not involved in the study. “Urinary retention is likely less common after that surgery, but it is still bothersome to patients.”
Dr. Schachar and associates also reported that patients in the earlier group “used significantly more morphine dose equivalents within 24 hours of the office voiding trial than the late-voiding trial group, which was expected given the proximity to surgery” (3 vs. 0.38; P = .005). However, new postoperative pain medication prescriptions and refills were similar in both groups.
Secondary endpoints included UTI rates, total days with a catheter, and patient experience of discomfort with the catheter. The two groups of women reported similar levels of catheter bother, but there was a nonsignificant difference in UTI rates: 23% in the earlier group, compared with 7% in the later group (P = .07).
The early-voiding trial group had an average 5 days with an indwelling transurethral catheter, compared with a significantly different 7 days in the later group (P = .0007). The early group also had fewer total days with an indwelling transurethral catheter and self-catheterization (6 days), compared with the late group (7 days; P = .0013). No patients had persistent incomplete bladder emptying after 17 days post surgery.
“Being able to adequately predict which patients are more likely to have unsuccessful postoperative voiding trials allows surgeons to better counsel their patients and may guide clinical decisions,” Dr. Schachar and associates said. They acknowledged, however, that their study’s biggest weakness is the small enrollment, which led to larger confidence intervals related to relative risk differences between the groups.
The study did not use external funding. Four of the investigators received grant, research funding, or honoraria from one or many medical device or pharmaceutical companies. The remaining researchers had no disclosures. Dr. Ault said he had no relevant financial disclosures.
SOURCE: Schachar JS et al. Am J Obstet Gynecol. 2020 Jun. doi: 10.1016/j.ajog.2020.06.001.
Women who underwent vaginal prolapse surgery and did not immediately have a successful voiding trial were seven times more likely to pass their second voiding trial if their follow-up was 7 days after surgery instead of 4 days, according to a study in the American Journal of Obstetrics and Gynecology.
“This information is useful for setting expectations and for counseling patients on when it might be best to repeat a voiding trial in those with transient incomplete bladder emptying on the day of surgery, especially for those who may not live close to their surgeon, or for those who have difficulty traveling to the office,” said Jeffrey S. Schachar, MD, of Wake Forest Baptist Health in Winston-Salem, N.C., and colleagues. “Despite a higher rate of initial unsuccessful office voiding trials, however, the early group did have significantly fewer days with an indwelling transurethral catheter, as well as total catheterization days,” including self-catheterization.
The researchers note that rates of temporary use of catheters after surgery vary widely, from 12% to 83%, likely because no consensus exists on how long to wait for voiding trials and what constitutes a successful trial.
“It is critical to identify patients with incomplete bladder emptying in order to prevent pain, myogenic and neurogenic damage, ureteral reflux and bladder overdistension that may further impair voiding function,” the authors wrote. “However, extending bladder drainage beyond the necessary recovery period may be associated with higher rates of urinary tract infection (UTI) and patient bother.”
To learn more about the best duration for postoperative catheter use, the researchers enrolled 102 patients before they underwent vaginal prolapse surgery at Wake Forest Baptist Health and Cleveland Clinic Florida from February 2017 to November 2019. The 29 patients with a successful voiding trial within 6 hours after surgery left the study, and 5 others were excluded for needing longer vaginal packing.
The voiding trial involved helping the patient stand to drain the bladder via the catheter, backfilling the bladder with 300 mL of saline solution through the catheter, removing the catheter to give women 1 hour to urinate, and then measuring the postvoid residual with a catheter or ultrasound. At least 100 mL postvoid residual was considered persistent incomplete bladder emptying.
The 60 remaining patients who did not pass the initial voiding trial and opted to remain in the study received a transurethral indwelling catheter and were randomly assigned to return for a second voiding trial either 2-4 days after surgery (depending on day of the week) or 7 days after surgery. The groups were demographically and clinically similar, with predominantly white postmenopausal, non-smoking women with stage II or III multicompartment pelvic organ prolapse.
Women without successful trials could continue with the transurethral catheter or give themselves intermittent catheterizations with a follow-up schedule determined by their surgeon. The researchers then tracked the women for 6 weeks to determine the rate of unsuccessful repeat voiding trials.
Among the women who returned 2-4 days post surgery, 23% had unsuccessful follow-up voiding trials, compared with 3% in the group returning 7 days after surgery (relative risk = 7; P = .02). The researchers calculated that one case of persistent postoperative incomplete bladder emptying was prevented for every five patients who used a catheter for 7 days after surgery.
Kevin A. Ault, MD, professor of obstetrics and gynecology at the University of Kansas Medical Center in Kansas City, said the study was well done, although the findings were unsurprising. He said the clinical implication is straightforward – to wait a week before doing a second voiding trial.
“I suspect these findings match the clinical experience of many surgeons. It is always good to see a well-done clinical trial on a topic,” Dr Ault said in an interview. “The most notable finding is how this impacts patient counseling. Gynecologists should tell their patients that it will take a week with a catheter when this problem arises.”
“The main limitation is whether this finding can be extrapolated to other gynecological surgeries, such as hysterectomy,” said Dr. Ault, who was not involved in the study. “Urinary retention is likely less common after that surgery, but it is still bothersome to patients.”
Dr. Schachar and associates also reported that patients in the earlier group “used significantly more morphine dose equivalents within 24 hours of the office voiding trial than the late-voiding trial group, which was expected given the proximity to surgery” (3 vs. 0.38; P = .005). However, new postoperative pain medication prescriptions and refills were similar in both groups.
Secondary endpoints included UTI rates, total days with a catheter, and patient experience of discomfort with the catheter. The two groups of women reported similar levels of catheter bother, but there was a nonsignificant difference in UTI rates: 23% in the earlier group, compared with 7% in the later group (P = .07).
The early-voiding trial group had an average 5 days with an indwelling transurethral catheter, compared with a significantly different 7 days in the later group (P = .0007). The early group also had fewer total days with an indwelling transurethral catheter and self-catheterization (6 days), compared with the late group (7 days; P = .0013). No patients had persistent incomplete bladder emptying after 17 days post surgery.
“Being able to adequately predict which patients are more likely to have unsuccessful postoperative voiding trials allows surgeons to better counsel their patients and may guide clinical decisions,” Dr. Schachar and associates said. They acknowledged, however, that their study’s biggest weakness is the small enrollment, which led to larger confidence intervals related to relative risk differences between the groups.
The study did not use external funding. Four of the investigators received grant, research funding, or honoraria from one or many medical device or pharmaceutical companies. The remaining researchers had no disclosures. Dr. Ault said he had no relevant financial disclosures.
SOURCE: Schachar JS et al. Am J Obstet Gynecol. 2020 Jun. doi: 10.1016/j.ajog.2020.06.001.
FROM AMERICAN JOURNAL OF OBSTETRICS & GYNECOLOGY
Prior beta-blockers predict extra burden of heart failure in women with ACS
In the analysis of more than 13,000 patients with ACS and no history of cardiovascular (CV) disease, the women who had taken beta-blockers for hypertension showed about a one-third increased risk for heart failure (HF) at the time of their ACS presentation.
The difference between women and men was especially pronounced among patients with ST-segment elevation MI, compared with those with non-STEMI.
No such relationship between sex and risk for HF with ACS was observed among the larger portion of the cohort that had not previously been treated with beta-blockers, according to a report published July 13 in Hypertension, with lead author Raffaele Bugiardini, MD, University of Bologna (Italy).
Mortality at 30 days was sharply higher for patients with than without HF at their ACS presentation, by more than 600% for women and more than 800% for men.
“Our study provides robust evidence of an interaction between sex and beta-blocker therapy and suggests an increased risk of HF among women presenting with incident myocardial infarction,” Dr. Bugiardini said in an interview.
Given their novelty, “our findings raise strong concern about the appropriate role of beta-blockers in the therapy of hypertension in women with no prior history of cardiovascular diseases. Beta-blocker use may be an acute precipitant of heart failure in women presenting with incident ACS as first manifestation of coronary heart disease.” Dr. Bugiardini and colleagues wrote.
“There is one main implication for clinical practice. Discontinuing a beta-blocker in an otherwise healthy woman with hypertension and no prior CV disease is not harmful and could be wise,” Dr. Bugiardini said. “Blood pressure in women may be regulated in a safer way, such as using other medications and, of course, through diet and exercise.”
Rationale for the study
Men and women “differ with respect to the risk, causes, and prognosis of HF,” Dr. Bugiardini and colleagues wrote, and current guidelines “do not differentiate between the use of beta-blockers in men and in women.”
However, they proposed, “because prior trials and meta-analyses enrolled nearly five men for every woman, any differences in the effect of beta-blockers among women would have been concealed by the effect of beta-blocker therapy among men.”
The current study looked at data from October 2010 to July 2018 in the ISACS ARCHIVES, ISACS-TC, and the EMMACE-3X registries, covering 13,764 patients from 12 European countries who had a history of hypertension and presented with confirmed ACS.
Of the combined cohort, 2,590 (19%) had been treated with beta-blockers prior to their ACS presentation. They were similar to those without a history of beta-blocker use with respect to baseline features and use of other medications in an adjusted analysis.
In the group with prior beta-blocker use, 21.3% of the women and 16.7% of the men had HF of Killip class 2 or higher, a 4.6% absolute difference that worked out to a relative risk of 1.35 (95% confidence interval, 1.10-1.65).
The corresponding rates for women and men without prior beta-blocker use were 17.2% and 16.1%, respectively, for an absolute difference of only 1.1% and an RR of1.09 (95% CI, 0.97-1.21).
The interaction between sex and beta-blocker therapy for the HF outcome was significant (P < .034). An analysis that excluded patients in cardiogenic shock at their ACS presentation produced similar results.
In an analysis only of patients with STEMI, the RR for HF in women versus men was 1.44 (95% CI, 1.12-1.84) among those with a history of beta-blocker use, and 1.11 (95% CI, 0.98-1.26) among those who hadn’t used the drugs. The interaction between sex and beta-blocker use was significant (P = .033).
No such significant interaction was seen for the subgroup with non-STEMI as their index ACS (P = .14).
Heart failure at ACS was the most powerful observed predictor of 30-day mortality in women and in men in multivariate analysis; the odds ratios were 7.54 (95% CI, 5.78-9.83) and 9.62 (95% CI, 7.67-12.07), respectively.
“Our study underscores the importance of sex analyses in clinical research studies, which may provide further actionable data,” Dr. Bugiardini stated. “Failure to include both sexes in therapeutic studies is a missed opportunity to uncover underlying sex-specific risks. The adverse effect of beta-blocker therapy in women with hypertension is a sex-specific risk.”
Not just a male disease
Part of the study’s conclusions are “really not that surprising, because we have known for a long time that women who have an MI are much more likely to develop HF than men, and we also know that HF raises mortality after MI,” Ileana L. Pina, MD, MPH, Wayne State University, Detroit, said in an interview.
But what surprised her was that women taking beta-blockers were at greater risk for HF. “This association needs to be proven in a prospective study and confirmed in another dataset,” said Dr. Pina, who was not involved with the current study. “The most important message is to remember that HF is not just a ‘male’ disease and to pay attention to the symptoms of women and not discount or relegate them to anxiety or gastric problems.”
The study was observational, Dr. Bugiardini noted, so “the results may have some variance and need confirmation. However, a sex-stratified, randomized, controlled trial of beta-blocker therapy in patients with hypertension but no prior history of coronary heart disease or HF may not be considered ethical, since it would be designed to confirm risk … and not benefit.”
“Further observational studies may give confirmation,” he added. “In the meantime, the Food and Drug Administration should alert health care professionals of the adverse events associated with beta-blocker use in women with hypertension and no prior history of CV disease, [because] prescribing beta-blockers to a woman with hypertension means exposing her to unnecessary risk.”
Dr. Bugiardini and the other authors had no disclosures. Dr. Pina reported no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
In the analysis of more than 13,000 patients with ACS and no history of cardiovascular (CV) disease, the women who had taken beta-blockers for hypertension showed about a one-third increased risk for heart failure (HF) at the time of their ACS presentation.
The difference between women and men was especially pronounced among patients with ST-segment elevation MI, compared with those with non-STEMI.
No such relationship between sex and risk for HF with ACS was observed among the larger portion of the cohort that had not previously been treated with beta-blockers, according to a report published July 13 in Hypertension, with lead author Raffaele Bugiardini, MD, University of Bologna (Italy).
Mortality at 30 days was sharply higher for patients with than without HF at their ACS presentation, by more than 600% for women and more than 800% for men.
“Our study provides robust evidence of an interaction between sex and beta-blocker therapy and suggests an increased risk of HF among women presenting with incident myocardial infarction,” Dr. Bugiardini said in an interview.
Given their novelty, “our findings raise strong concern about the appropriate role of beta-blockers in the therapy of hypertension in women with no prior history of cardiovascular diseases. Beta-blocker use may be an acute precipitant of heart failure in women presenting with incident ACS as first manifestation of coronary heart disease.” Dr. Bugiardini and colleagues wrote.
“There is one main implication for clinical practice. Discontinuing a beta-blocker in an otherwise healthy woman with hypertension and no prior CV disease is not harmful and could be wise,” Dr. Bugiardini said. “Blood pressure in women may be regulated in a safer way, such as using other medications and, of course, through diet and exercise.”
Rationale for the study
Men and women “differ with respect to the risk, causes, and prognosis of HF,” Dr. Bugiardini and colleagues wrote, and current guidelines “do not differentiate between the use of beta-blockers in men and in women.”
However, they proposed, “because prior trials and meta-analyses enrolled nearly five men for every woman, any differences in the effect of beta-blockers among women would have been concealed by the effect of beta-blocker therapy among men.”
The current study looked at data from October 2010 to July 2018 in the ISACS ARCHIVES, ISACS-TC, and the EMMACE-3X registries, covering 13,764 patients from 12 European countries who had a history of hypertension and presented with confirmed ACS.
Of the combined cohort, 2,590 (19%) had been treated with beta-blockers prior to their ACS presentation. They were similar to those without a history of beta-blocker use with respect to baseline features and use of other medications in an adjusted analysis.
In the group with prior beta-blocker use, 21.3% of the women and 16.7% of the men had HF of Killip class 2 or higher, a 4.6% absolute difference that worked out to a relative risk of 1.35 (95% confidence interval, 1.10-1.65).
The corresponding rates for women and men without prior beta-blocker use were 17.2% and 16.1%, respectively, for an absolute difference of only 1.1% and an RR of1.09 (95% CI, 0.97-1.21).
The interaction between sex and beta-blocker therapy for the HF outcome was significant (P < .034). An analysis that excluded patients in cardiogenic shock at their ACS presentation produced similar results.
In an analysis only of patients with STEMI, the RR for HF in women versus men was 1.44 (95% CI, 1.12-1.84) among those with a history of beta-blocker use, and 1.11 (95% CI, 0.98-1.26) among those who hadn’t used the drugs. The interaction between sex and beta-blocker use was significant (P = .033).
No such significant interaction was seen for the subgroup with non-STEMI as their index ACS (P = .14).
Heart failure at ACS was the most powerful observed predictor of 30-day mortality in women and in men in multivariate analysis; the odds ratios were 7.54 (95% CI, 5.78-9.83) and 9.62 (95% CI, 7.67-12.07), respectively.
“Our study underscores the importance of sex analyses in clinical research studies, which may provide further actionable data,” Dr. Bugiardini stated. “Failure to include both sexes in therapeutic studies is a missed opportunity to uncover underlying sex-specific risks. The adverse effect of beta-blocker therapy in women with hypertension is a sex-specific risk.”
Not just a male disease
Part of the study’s conclusions are “really not that surprising, because we have known for a long time that women who have an MI are much more likely to develop HF than men, and we also know that HF raises mortality after MI,” Ileana L. Pina, MD, MPH, Wayne State University, Detroit, said in an interview.
But what surprised her was that women taking beta-blockers were at greater risk for HF. “This association needs to be proven in a prospective study and confirmed in another dataset,” said Dr. Pina, who was not involved with the current study. “The most important message is to remember that HF is not just a ‘male’ disease and to pay attention to the symptoms of women and not discount or relegate them to anxiety or gastric problems.”
The study was observational, Dr. Bugiardini noted, so “the results may have some variance and need confirmation. However, a sex-stratified, randomized, controlled trial of beta-blocker therapy in patients with hypertension but no prior history of coronary heart disease or HF may not be considered ethical, since it would be designed to confirm risk … and not benefit.”
“Further observational studies may give confirmation,” he added. “In the meantime, the Food and Drug Administration should alert health care professionals of the adverse events associated with beta-blocker use in women with hypertension and no prior history of CV disease, [because] prescribing beta-blockers to a woman with hypertension means exposing her to unnecessary risk.”
Dr. Bugiardini and the other authors had no disclosures. Dr. Pina reported no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
In the analysis of more than 13,000 patients with ACS and no history of cardiovascular (CV) disease, the women who had taken beta-blockers for hypertension showed about a one-third increased risk for heart failure (HF) at the time of their ACS presentation.
The difference between women and men was especially pronounced among patients with ST-segment elevation MI, compared with those with non-STEMI.
No such relationship between sex and risk for HF with ACS was observed among the larger portion of the cohort that had not previously been treated with beta-blockers, according to a report published July 13 in Hypertension, with lead author Raffaele Bugiardini, MD, University of Bologna (Italy).
Mortality at 30 days was sharply higher for patients with than without HF at their ACS presentation, by more than 600% for women and more than 800% for men.
“Our study provides robust evidence of an interaction between sex and beta-blocker therapy and suggests an increased risk of HF among women presenting with incident myocardial infarction,” Dr. Bugiardini said in an interview.
Given their novelty, “our findings raise strong concern about the appropriate role of beta-blockers in the therapy of hypertension in women with no prior history of cardiovascular diseases. Beta-blocker use may be an acute precipitant of heart failure in women presenting with incident ACS as first manifestation of coronary heart disease.” Dr. Bugiardini and colleagues wrote.
“There is one main implication for clinical practice. Discontinuing a beta-blocker in an otherwise healthy woman with hypertension and no prior CV disease is not harmful and could be wise,” Dr. Bugiardini said. “Blood pressure in women may be regulated in a safer way, such as using other medications and, of course, through diet and exercise.”
Rationale for the study
Men and women “differ with respect to the risk, causes, and prognosis of HF,” Dr. Bugiardini and colleagues wrote, and current guidelines “do not differentiate between the use of beta-blockers in men and in women.”
However, they proposed, “because prior trials and meta-analyses enrolled nearly five men for every woman, any differences in the effect of beta-blockers among women would have been concealed by the effect of beta-blocker therapy among men.”
The current study looked at data from October 2010 to July 2018 in the ISACS ARCHIVES, ISACS-TC, and the EMMACE-3X registries, covering 13,764 patients from 12 European countries who had a history of hypertension and presented with confirmed ACS.
Of the combined cohort, 2,590 (19%) had been treated with beta-blockers prior to their ACS presentation. They were similar to those without a history of beta-blocker use with respect to baseline features and use of other medications in an adjusted analysis.
In the group with prior beta-blocker use, 21.3% of the women and 16.7% of the men had HF of Killip class 2 or higher, a 4.6% absolute difference that worked out to a relative risk of 1.35 (95% confidence interval, 1.10-1.65).
The corresponding rates for women and men without prior beta-blocker use were 17.2% and 16.1%, respectively, for an absolute difference of only 1.1% and an RR of1.09 (95% CI, 0.97-1.21).
The interaction between sex and beta-blocker therapy for the HF outcome was significant (P < .034). An analysis that excluded patients in cardiogenic shock at their ACS presentation produced similar results.
In an analysis only of patients with STEMI, the RR for HF in women versus men was 1.44 (95% CI, 1.12-1.84) among those with a history of beta-blocker use, and 1.11 (95% CI, 0.98-1.26) among those who hadn’t used the drugs. The interaction between sex and beta-blocker use was significant (P = .033).
No such significant interaction was seen for the subgroup with non-STEMI as their index ACS (P = .14).
Heart failure at ACS was the most powerful observed predictor of 30-day mortality in women and in men in multivariate analysis; the odds ratios were 7.54 (95% CI, 5.78-9.83) and 9.62 (95% CI, 7.67-12.07), respectively.
“Our study underscores the importance of sex analyses in clinical research studies, which may provide further actionable data,” Dr. Bugiardini stated. “Failure to include both sexes in therapeutic studies is a missed opportunity to uncover underlying sex-specific risks. The adverse effect of beta-blocker therapy in women with hypertension is a sex-specific risk.”
Not just a male disease
Part of the study’s conclusions are “really not that surprising, because we have known for a long time that women who have an MI are much more likely to develop HF than men, and we also know that HF raises mortality after MI,” Ileana L. Pina, MD, MPH, Wayne State University, Detroit, said in an interview.
But what surprised her was that women taking beta-blockers were at greater risk for HF. “This association needs to be proven in a prospective study and confirmed in another dataset,” said Dr. Pina, who was not involved with the current study. “The most important message is to remember that HF is not just a ‘male’ disease and to pay attention to the symptoms of women and not discount or relegate them to anxiety or gastric problems.”
The study was observational, Dr. Bugiardini noted, so “the results may have some variance and need confirmation. However, a sex-stratified, randomized, controlled trial of beta-blocker therapy in patients with hypertension but no prior history of coronary heart disease or HF may not be considered ethical, since it would be designed to confirm risk … and not benefit.”
“Further observational studies may give confirmation,” he added. “In the meantime, the Food and Drug Administration should alert health care professionals of the adverse events associated with beta-blocker use in women with hypertension and no prior history of CV disease, [because] prescribing beta-blockers to a woman with hypertension means exposing her to unnecessary risk.”
Dr. Bugiardini and the other authors had no disclosures. Dr. Pina reported no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Novel program cuts weight retention after gestational diabetes
An online, lifestyle-based weight loss initiative known as the Balance After Baby (BAB) program is effective at reducing weight retention a year after birth among women with recent gestational diabetes.
Specifically, results of the study were positive in women of most ethnicities, bar those of a small group of Hispanic origin.
Jacinda Nicklas, MD, from the University of Colorado at Denver, Aurora, presented findings of the BAB trial during the virtual annual scientific sessions of the American Diabetes Association. She was coprincipal investigator alongside Ellen Seely, MD, from Brigham and Women’s Hospital, Boston.
“Looking at the entire population of women on the BAB program, there was a trend in weight loss from 6 weeks postpartum to 12 months (P = .09), and significantly less postpartum weight retention at 12 months (P = .04),” Dr. Nicklas said.
“Through this effect on postpartum weight retention, the BAB program has potential to delay or prevent development of type 2 diabetes in women with recent gestational diabetes, while the web-based, remote nature of the program is scalable and very relevant in current times,” she added. “However, the lack of efficacy in Hispanic women means it needs to be modified to be successful in this ethnic group.”
Frank Qian, MD, who also presented during the same session, said the BAB program has potential as a viable way of preventing both future pregnancy complications and the progression to overt type 2 diabetes in this high-risk population.
“Large-scale epidemiologic studies show us that weight gain from pregnancy is a major risk factor for long-term cardiometabolic risk, particularly for women with a history of gestational diabetes,” he observed. “In turn, it is critical to implement lifestyle interventions that can help women get as close to the weight they were before pregnancy as possible and keep that weight off.”
Postpartum weight retention a modifiable risk factor for type 2 diabetes
Current evidence shows that a large proportion of women who develop gestational diabetes go on to develop type 2 diabetes within 10 years and that women with a history of gestational diabetes are more likely to retain or gain weight postpartum.
Dr. Nicklas also pointed out that obesity and weight gain are the strongest modifiable risk factors for type 2 diabetes.
“We know from the Diabetes Prevention Program [DPP] that an intensive lifestyle program in women who had had gestational diabetes led to a 53% reduction in type 2 diabetes,” Dr. Nicklas noted.
However, she added there were barriers to adhering to the intensive DPP program – which required 16 one-on-one meetings in the first 24 weeks – including travel, as some participants lived quite remotely, or family responsibilities. Consequently, Dr. Nicklas and colleagues developed the BAB pilot trial, which involved web-based delivery with remote coaching.
The trial involved women with a history of gestational diabetes who were, on average, 7 weeks postpartum. The key outcome was weight at 12 months, compared with both 6-week postpartum weight and prepregnancy weight.
Based on encouraging results in the pilot trial – in which the intervention group showed significant weight loss from 6-week postpartum weight and in 12-month weight retention – a larger, two-site trial was initiated, the BAB Intervention randomized, controlled trial.
Outcome measures were the same as for the pilot study. The 181 participants were aged 18-45 years, had recent gestational diabetes, and had a mean prepregnancy body mass index of approximately 29 kg/m2. Around half were college educated, and 28% were from lower income households. Overall, 48% were white, 22% Asian, 17% African American, and 13% were of other ethnicities, with just over a third being Hispanic.
The initial study visit was at 6 weeks postpartum. Women were randomized to the behavioral intervention website plus a lifestyle coach group or to a control group that consisted of a website plus knowledge links.
The intervention website required women to complete some DPP-derived and bonus modules, and also featured action plans, tracked weight and steps, and had a direct link to contact their lifestyle coach. Follow-up visits were held at 6 and 12 months and A1c, waist circumference, and height/weight were measured. A total of 86% eligible women completed the 6- and 12-month visits.
Why didn’t the BAB program work in Hispanic women?
“The overall result showed that weight change from 6 weeks postpartum to 12 months revealed a slight gain in the control group of 1.3 pounds and a loss in the intervention group of 1.8 pounds, resulting in a between-group difference of 3.1 pounds [P = .09],” reported Dr. Nicklas. Adjustment for gestational weight gain and breastfeeding had no substantial effect.
When 12-month weight retention versus prepregnancy weight was assessed, the former was halved in participants in the BAB program.
The control group gained a mean of 10.1 pounds, and those in the intervention group gained a mean of 5.3 pounds, equivalent to a difference of 4.8 pounds (P = .04).
A prespecified analysis was conducted of 120 non-Hispanic women. At 12 months, weight retention, compared with prepregnancy weight showed an increase of 9 pounds in the control group versus 1.8 pounds in the intervention group (P = .01).
By comparison, in the small group of Hispanic women only, weight retention at 12 months compared to prepregnancy weight showed a 12.7-pound increase and a 13.3-pound increase in the control and intervention groups respectively, reported Dr. Nicklas.
Addressing the key question of why the BAB program was ineffective in Hispanic women, Dr. Nicklas said, “The literature tells us that low income Hispanic women are twice as likely to experience postpartum weight retention compared to white non-Hispanic women. But we also know that low-income Hispanic women generally engage less with interventions, and there is a higher acceptance of overweight among this ethnic group.”
The researchers hope to follow the women from their trial to determine who progresses to type 2 diabetes.
“Hispanic women are a high-risk population for gestational diabetes and type 2 diabetes, and we plan to identify the best options to help Hispanic women with a history of gestational diabetes prevent type 2 diabetes,” Dr. Nicklas said in an interview.
Dr. Qian also remarked on the differences observed in the weight loss outcomes for non-Hispanic versus Hispanic women, noting that it highlights the importance of studying lifestyle interventions in diverse populations. “Environmental and cultural factors that may differ across different racial or ethnic groups could impact the effectiveness of such interventions.
Dr. Nicklas and Dr. Qian have reported no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
An online, lifestyle-based weight loss initiative known as the Balance After Baby (BAB) program is effective at reducing weight retention a year after birth among women with recent gestational diabetes.
Specifically, results of the study were positive in women of most ethnicities, bar those of a small group of Hispanic origin.
Jacinda Nicklas, MD, from the University of Colorado at Denver, Aurora, presented findings of the BAB trial during the virtual annual scientific sessions of the American Diabetes Association. She was coprincipal investigator alongside Ellen Seely, MD, from Brigham and Women’s Hospital, Boston.
“Looking at the entire population of women on the BAB program, there was a trend in weight loss from 6 weeks postpartum to 12 months (P = .09), and significantly less postpartum weight retention at 12 months (P = .04),” Dr. Nicklas said.
“Through this effect on postpartum weight retention, the BAB program has potential to delay or prevent development of type 2 diabetes in women with recent gestational diabetes, while the web-based, remote nature of the program is scalable and very relevant in current times,” she added. “However, the lack of efficacy in Hispanic women means it needs to be modified to be successful in this ethnic group.”
Frank Qian, MD, who also presented during the same session, said the BAB program has potential as a viable way of preventing both future pregnancy complications and the progression to overt type 2 diabetes in this high-risk population.
“Large-scale epidemiologic studies show us that weight gain from pregnancy is a major risk factor for long-term cardiometabolic risk, particularly for women with a history of gestational diabetes,” he observed. “In turn, it is critical to implement lifestyle interventions that can help women get as close to the weight they were before pregnancy as possible and keep that weight off.”
Postpartum weight retention a modifiable risk factor for type 2 diabetes
Current evidence shows that a large proportion of women who develop gestational diabetes go on to develop type 2 diabetes within 10 years and that women with a history of gestational diabetes are more likely to retain or gain weight postpartum.
Dr. Nicklas also pointed out that obesity and weight gain are the strongest modifiable risk factors for type 2 diabetes.
“We know from the Diabetes Prevention Program [DPP] that an intensive lifestyle program in women who had had gestational diabetes led to a 53% reduction in type 2 diabetes,” Dr. Nicklas noted.
However, she added there were barriers to adhering to the intensive DPP program – which required 16 one-on-one meetings in the first 24 weeks – including travel, as some participants lived quite remotely, or family responsibilities. Consequently, Dr. Nicklas and colleagues developed the BAB pilot trial, which involved web-based delivery with remote coaching.
The trial involved women with a history of gestational diabetes who were, on average, 7 weeks postpartum. The key outcome was weight at 12 months, compared with both 6-week postpartum weight and prepregnancy weight.
Based on encouraging results in the pilot trial – in which the intervention group showed significant weight loss from 6-week postpartum weight and in 12-month weight retention – a larger, two-site trial was initiated, the BAB Intervention randomized, controlled trial.
Outcome measures were the same as for the pilot study. The 181 participants were aged 18-45 years, had recent gestational diabetes, and had a mean prepregnancy body mass index of approximately 29 kg/m2. Around half were college educated, and 28% were from lower income households. Overall, 48% were white, 22% Asian, 17% African American, and 13% were of other ethnicities, with just over a third being Hispanic.
The initial study visit was at 6 weeks postpartum. Women were randomized to the behavioral intervention website plus a lifestyle coach group or to a control group that consisted of a website plus knowledge links.
The intervention website required women to complete some DPP-derived and bonus modules, and also featured action plans, tracked weight and steps, and had a direct link to contact their lifestyle coach. Follow-up visits were held at 6 and 12 months and A1c, waist circumference, and height/weight were measured. A total of 86% eligible women completed the 6- and 12-month visits.
Why didn’t the BAB program work in Hispanic women?
“The overall result showed that weight change from 6 weeks postpartum to 12 months revealed a slight gain in the control group of 1.3 pounds and a loss in the intervention group of 1.8 pounds, resulting in a between-group difference of 3.1 pounds [P = .09],” reported Dr. Nicklas. Adjustment for gestational weight gain and breastfeeding had no substantial effect.
When 12-month weight retention versus prepregnancy weight was assessed, the former was halved in participants in the BAB program.
The control group gained a mean of 10.1 pounds, and those in the intervention group gained a mean of 5.3 pounds, equivalent to a difference of 4.8 pounds (P = .04).
A prespecified analysis was conducted of 120 non-Hispanic women. At 12 months, weight retention, compared with prepregnancy weight showed an increase of 9 pounds in the control group versus 1.8 pounds in the intervention group (P = .01).
By comparison, in the small group of Hispanic women only, weight retention at 12 months compared to prepregnancy weight showed a 12.7-pound increase and a 13.3-pound increase in the control and intervention groups respectively, reported Dr. Nicklas.
Addressing the key question of why the BAB program was ineffective in Hispanic women, Dr. Nicklas said, “The literature tells us that low income Hispanic women are twice as likely to experience postpartum weight retention compared to white non-Hispanic women. But we also know that low-income Hispanic women generally engage less with interventions, and there is a higher acceptance of overweight among this ethnic group.”
The researchers hope to follow the women from their trial to determine who progresses to type 2 diabetes.
“Hispanic women are a high-risk population for gestational diabetes and type 2 diabetes, and we plan to identify the best options to help Hispanic women with a history of gestational diabetes prevent type 2 diabetes,” Dr. Nicklas said in an interview.
Dr. Qian also remarked on the differences observed in the weight loss outcomes for non-Hispanic versus Hispanic women, noting that it highlights the importance of studying lifestyle interventions in diverse populations. “Environmental and cultural factors that may differ across different racial or ethnic groups could impact the effectiveness of such interventions.
Dr. Nicklas and Dr. Qian have reported no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
An online, lifestyle-based weight loss initiative known as the Balance After Baby (BAB) program is effective at reducing weight retention a year after birth among women with recent gestational diabetes.
Specifically, results of the study were positive in women of most ethnicities, bar those of a small group of Hispanic origin.
Jacinda Nicklas, MD, from the University of Colorado at Denver, Aurora, presented findings of the BAB trial during the virtual annual scientific sessions of the American Diabetes Association. She was coprincipal investigator alongside Ellen Seely, MD, from Brigham and Women’s Hospital, Boston.
“Looking at the entire population of women on the BAB program, there was a trend in weight loss from 6 weeks postpartum to 12 months (P = .09), and significantly less postpartum weight retention at 12 months (P = .04),” Dr. Nicklas said.
“Through this effect on postpartum weight retention, the BAB program has potential to delay or prevent development of type 2 diabetes in women with recent gestational diabetes, while the web-based, remote nature of the program is scalable and very relevant in current times,” she added. “However, the lack of efficacy in Hispanic women means it needs to be modified to be successful in this ethnic group.”
Frank Qian, MD, who also presented during the same session, said the BAB program has potential as a viable way of preventing both future pregnancy complications and the progression to overt type 2 diabetes in this high-risk population.
“Large-scale epidemiologic studies show us that weight gain from pregnancy is a major risk factor for long-term cardiometabolic risk, particularly for women with a history of gestational diabetes,” he observed. “In turn, it is critical to implement lifestyle interventions that can help women get as close to the weight they were before pregnancy as possible and keep that weight off.”
Postpartum weight retention a modifiable risk factor for type 2 diabetes
Current evidence shows that a large proportion of women who develop gestational diabetes go on to develop type 2 diabetes within 10 years and that women with a history of gestational diabetes are more likely to retain or gain weight postpartum.
Dr. Nicklas also pointed out that obesity and weight gain are the strongest modifiable risk factors for type 2 diabetes.
“We know from the Diabetes Prevention Program [DPP] that an intensive lifestyle program in women who had had gestational diabetes led to a 53% reduction in type 2 diabetes,” Dr. Nicklas noted.
However, she added there were barriers to adhering to the intensive DPP program – which required 16 one-on-one meetings in the first 24 weeks – including travel, as some participants lived quite remotely, or family responsibilities. Consequently, Dr. Nicklas and colleagues developed the BAB pilot trial, which involved web-based delivery with remote coaching.
The trial involved women with a history of gestational diabetes who were, on average, 7 weeks postpartum. The key outcome was weight at 12 months, compared with both 6-week postpartum weight and prepregnancy weight.
Based on encouraging results in the pilot trial – in which the intervention group showed significant weight loss from 6-week postpartum weight and in 12-month weight retention – a larger, two-site trial was initiated, the BAB Intervention randomized, controlled trial.
Outcome measures were the same as for the pilot study. The 181 participants were aged 18-45 years, had recent gestational diabetes, and had a mean prepregnancy body mass index of approximately 29 kg/m2. Around half were college educated, and 28% were from lower income households. Overall, 48% were white, 22% Asian, 17% African American, and 13% were of other ethnicities, with just over a third being Hispanic.
The initial study visit was at 6 weeks postpartum. Women were randomized to the behavioral intervention website plus a lifestyle coach group or to a control group that consisted of a website plus knowledge links.
The intervention website required women to complete some DPP-derived and bonus modules, and also featured action plans, tracked weight and steps, and had a direct link to contact their lifestyle coach. Follow-up visits were held at 6 and 12 months and A1c, waist circumference, and height/weight were measured. A total of 86% eligible women completed the 6- and 12-month visits.
Why didn’t the BAB program work in Hispanic women?
“The overall result showed that weight change from 6 weeks postpartum to 12 months revealed a slight gain in the control group of 1.3 pounds and a loss in the intervention group of 1.8 pounds, resulting in a between-group difference of 3.1 pounds [P = .09],” reported Dr. Nicklas. Adjustment for gestational weight gain and breastfeeding had no substantial effect.
When 12-month weight retention versus prepregnancy weight was assessed, the former was halved in participants in the BAB program.
The control group gained a mean of 10.1 pounds, and those in the intervention group gained a mean of 5.3 pounds, equivalent to a difference of 4.8 pounds (P = .04).
A prespecified analysis was conducted of 120 non-Hispanic women. At 12 months, weight retention, compared with prepregnancy weight showed an increase of 9 pounds in the control group versus 1.8 pounds in the intervention group (P = .01).
By comparison, in the small group of Hispanic women only, weight retention at 12 months compared to prepregnancy weight showed a 12.7-pound increase and a 13.3-pound increase in the control and intervention groups respectively, reported Dr. Nicklas.
Addressing the key question of why the BAB program was ineffective in Hispanic women, Dr. Nicklas said, “The literature tells us that low income Hispanic women are twice as likely to experience postpartum weight retention compared to white non-Hispanic women. But we also know that low-income Hispanic women generally engage less with interventions, and there is a higher acceptance of overweight among this ethnic group.”
The researchers hope to follow the women from their trial to determine who progresses to type 2 diabetes.
“Hispanic women are a high-risk population for gestational diabetes and type 2 diabetes, and we plan to identify the best options to help Hispanic women with a history of gestational diabetes prevent type 2 diabetes,” Dr. Nicklas said in an interview.
Dr. Qian also remarked on the differences observed in the weight loss outcomes for non-Hispanic versus Hispanic women, noting that it highlights the importance of studying lifestyle interventions in diverse populations. “Environmental and cultural factors that may differ across different racial or ethnic groups could impact the effectiveness of such interventions.
Dr. Nicklas and Dr. Qian have reported no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
FROM ADA 2020
Women suffer less NAFLD but more advanced fibrosis
Women have a lower risk of nonalcoholic fatty liver disease compared with men, but those who do develop the disease are significantly more likely than are men to develop advanced fibrosis, according to data from a meta-analysis of more than 62,000 individuals.
Sex disparity persists in most chronic liver diseases, with more cases and risk of progression reported in men, but the effect of sex on nonalcoholic fatty liver disease (NAFLD) remains unclear, wrote Maya Balakrishnan, MD, of Baylor College of Medicine, Houston, and colleagues. “Knowing whether and how [sex] influences the risk and severity of NAFLD is important for risk stratification, risk modification as well as prognostication,” they said.
In a study published in Clinical Gastroenterology and Hepatology, the researchers conducted a review and meta-analysis of 54 studies, including data from 62,239 patients with NAFLD, 5,428 with nonalcoholic steatohepatitis (NASH), and 6,444 with advanced NAFLD fibrosis.
Overall, women had a 19% lower risk of developing NAFLD compared with men (pooled risk ratio 0.81), a similar risk to men of developing NASH (RR, 1.00), and a 37% increased risk of advanced fibrosis (RR, 1.37) compared with men.
The risk of more severe disease in women increased with age. Among women aged 50 years and older, the risks of NASH and advanced fibrosis were significantly higher, at 17% and 56%, respectively (RR, 1.17 and RR, 1.56). The sex-specific prevalence of advanced fibrosis was not significantly different in patients younger than 50 years.
“Our findings of an increased prevalence of severe phenotypes of NAFLD – NASH and advanced fibrosis – among older women fits well into the current understanding of disease pathogenesis,” the researchers noted.
The findings were limited by several factors, including the cross-sectional nature and heterogeneity of the included studies and lack of data on possible contributions to NASH and NAFLD such as polycystic ovarian syndrome, cumulative use of hormone therapy, and pregnancy, the researchers noted.
However, the results were strengthened by the large patient population. “Given the higher risk of advanced fibrosis observed among women compared to men with NAFLD in our meta-analysis, it is plausible that cirrhosis and its complications may occur with greater frequency among women than in men,” the researchers said. Consequently, women older than 50 years with NAFLD should be evaluated frequently for advanced disease, they noted. In addition, “more focused and intensified efforts may be warranted to target lifestyle modifications and weight loss among young women with NAFLD, particularly in the presence of NASH and/or advanced fibrosis,” the researchers concluded.
Conducting the study at this time was important because of conjectures of sex-based differences in NAFLD prevalence and NAFLD progression, Dr. Balakrishnan said in an interview. “However, the findings from studies conducted across different study populations have been disparate. Therefore, it was important to perform a systematic review and meta-analysis to determine whether there are differences in NAFLD and NAFLD severity risk between the [sexes],” she said.
Dr. Balakrishnan said she was surprised by the higher risk of severe NASH fibrosis in women compared with men once NAFLD is established. “This was surprising and sets NAFLD apart from other highly prevalent chronic liver disease etiologies,” she said. “Other common liver diseases, for example hepatitis B and hepatitis C, tend to be more common among men and tend to progress more rapidly, and tend to be more severe among men compared to women,” she noted.
The take-home message for clinicians is that NAFLD is at least equally, if not more, aggressive in women compared with men, and should be evaluated with equal aggressiveness, Dr. Balakrishnan emphasized. “Moreover, in the future we may expect to see the burden of cirrhosis distributed more equally among women and men than we have to date. This has implications for liver disease screening and women’s health,” she said. The next steps for research are to determine the specific reasons for the higher risk of NAFLD fibrosis in women compared with men, she added.
The study was supported in part by the National Institutes of Health. The researchers had no financial conflicts to disclose.
SOURCE: Balakrishnan M et al. Clin Gastroenterol Hepatol. 2020 Apr 30. doi: 10.1016/j.cgh.2020.04.067.
Women have a lower risk of nonalcoholic fatty liver disease compared with men, but those who do develop the disease are significantly more likely than are men to develop advanced fibrosis, according to data from a meta-analysis of more than 62,000 individuals.
Sex disparity persists in most chronic liver diseases, with more cases and risk of progression reported in men, but the effect of sex on nonalcoholic fatty liver disease (NAFLD) remains unclear, wrote Maya Balakrishnan, MD, of Baylor College of Medicine, Houston, and colleagues. “Knowing whether and how [sex] influences the risk and severity of NAFLD is important for risk stratification, risk modification as well as prognostication,” they said.
In a study published in Clinical Gastroenterology and Hepatology, the researchers conducted a review and meta-analysis of 54 studies, including data from 62,239 patients with NAFLD, 5,428 with nonalcoholic steatohepatitis (NASH), and 6,444 with advanced NAFLD fibrosis.
Overall, women had a 19% lower risk of developing NAFLD compared with men (pooled risk ratio 0.81), a similar risk to men of developing NASH (RR, 1.00), and a 37% increased risk of advanced fibrosis (RR, 1.37) compared with men.
The risk of more severe disease in women increased with age. Among women aged 50 years and older, the risks of NASH and advanced fibrosis were significantly higher, at 17% and 56%, respectively (RR, 1.17 and RR, 1.56). The sex-specific prevalence of advanced fibrosis was not significantly different in patients younger than 50 years.
“Our findings of an increased prevalence of severe phenotypes of NAFLD – NASH and advanced fibrosis – among older women fits well into the current understanding of disease pathogenesis,” the researchers noted.
The findings were limited by several factors, including the cross-sectional nature and heterogeneity of the included studies and lack of data on possible contributions to NASH and NAFLD such as polycystic ovarian syndrome, cumulative use of hormone therapy, and pregnancy, the researchers noted.
However, the results were strengthened by the large patient population. “Given the higher risk of advanced fibrosis observed among women compared to men with NAFLD in our meta-analysis, it is plausible that cirrhosis and its complications may occur with greater frequency among women than in men,” the researchers said. Consequently, women older than 50 years with NAFLD should be evaluated frequently for advanced disease, they noted. In addition, “more focused and intensified efforts may be warranted to target lifestyle modifications and weight loss among young women with NAFLD, particularly in the presence of NASH and/or advanced fibrosis,” the researchers concluded.
Conducting the study at this time was important because of conjectures of sex-based differences in NAFLD prevalence and NAFLD progression, Dr. Balakrishnan said in an interview. “However, the findings from studies conducted across different study populations have been disparate. Therefore, it was important to perform a systematic review and meta-analysis to determine whether there are differences in NAFLD and NAFLD severity risk between the [sexes],” she said.
Dr. Balakrishnan said she was surprised by the higher risk of severe NASH fibrosis in women compared with men once NAFLD is established. “This was surprising and sets NAFLD apart from other highly prevalent chronic liver disease etiologies,” she said. “Other common liver diseases, for example hepatitis B and hepatitis C, tend to be more common among men and tend to progress more rapidly, and tend to be more severe among men compared to women,” she noted.
The take-home message for clinicians is that NAFLD is at least equally, if not more, aggressive in women compared with men, and should be evaluated with equal aggressiveness, Dr. Balakrishnan emphasized. “Moreover, in the future we may expect to see the burden of cirrhosis distributed more equally among women and men than we have to date. This has implications for liver disease screening and women’s health,” she said. The next steps for research are to determine the specific reasons for the higher risk of NAFLD fibrosis in women compared with men, she added.
The study was supported in part by the National Institutes of Health. The researchers had no financial conflicts to disclose.
SOURCE: Balakrishnan M et al. Clin Gastroenterol Hepatol. 2020 Apr 30. doi: 10.1016/j.cgh.2020.04.067.
Women have a lower risk of nonalcoholic fatty liver disease compared with men, but those who do develop the disease are significantly more likely than are men to develop advanced fibrosis, according to data from a meta-analysis of more than 62,000 individuals.
Sex disparity persists in most chronic liver diseases, with more cases and risk of progression reported in men, but the effect of sex on nonalcoholic fatty liver disease (NAFLD) remains unclear, wrote Maya Balakrishnan, MD, of Baylor College of Medicine, Houston, and colleagues. “Knowing whether and how [sex] influences the risk and severity of NAFLD is important for risk stratification, risk modification as well as prognostication,” they said.
In a study published in Clinical Gastroenterology and Hepatology, the researchers conducted a review and meta-analysis of 54 studies, including data from 62,239 patients with NAFLD, 5,428 with nonalcoholic steatohepatitis (NASH), and 6,444 with advanced NAFLD fibrosis.
Overall, women had a 19% lower risk of developing NAFLD compared with men (pooled risk ratio 0.81), a similar risk to men of developing NASH (RR, 1.00), and a 37% increased risk of advanced fibrosis (RR, 1.37) compared with men.
The risk of more severe disease in women increased with age. Among women aged 50 years and older, the risks of NASH and advanced fibrosis were significantly higher, at 17% and 56%, respectively (RR, 1.17 and RR, 1.56). The sex-specific prevalence of advanced fibrosis was not significantly different in patients younger than 50 years.
“Our findings of an increased prevalence of severe phenotypes of NAFLD – NASH and advanced fibrosis – among older women fits well into the current understanding of disease pathogenesis,” the researchers noted.
The findings were limited by several factors, including the cross-sectional nature and heterogeneity of the included studies and lack of data on possible contributions to NASH and NAFLD such as polycystic ovarian syndrome, cumulative use of hormone therapy, and pregnancy, the researchers noted.
However, the results were strengthened by the large patient population. “Given the higher risk of advanced fibrosis observed among women compared to men with NAFLD in our meta-analysis, it is plausible that cirrhosis and its complications may occur with greater frequency among women than in men,” the researchers said. Consequently, women older than 50 years with NAFLD should be evaluated frequently for advanced disease, they noted. In addition, “more focused and intensified efforts may be warranted to target lifestyle modifications and weight loss among young women with NAFLD, particularly in the presence of NASH and/or advanced fibrosis,” the researchers concluded.
Conducting the study at this time was important because of conjectures of sex-based differences in NAFLD prevalence and NAFLD progression, Dr. Balakrishnan said in an interview. “However, the findings from studies conducted across different study populations have been disparate. Therefore, it was important to perform a systematic review and meta-analysis to determine whether there are differences in NAFLD and NAFLD severity risk between the [sexes],” she said.
Dr. Balakrishnan said she was surprised by the higher risk of severe NASH fibrosis in women compared with men once NAFLD is established. “This was surprising and sets NAFLD apart from other highly prevalent chronic liver disease etiologies,” she said. “Other common liver diseases, for example hepatitis B and hepatitis C, tend to be more common among men and tend to progress more rapidly, and tend to be more severe among men compared to women,” she noted.
The take-home message for clinicians is that NAFLD is at least equally, if not more, aggressive in women compared with men, and should be evaluated with equal aggressiveness, Dr. Balakrishnan emphasized. “Moreover, in the future we may expect to see the burden of cirrhosis distributed more equally among women and men than we have to date. This has implications for liver disease screening and women’s health,” she said. The next steps for research are to determine the specific reasons for the higher risk of NAFLD fibrosis in women compared with men, she added.
The study was supported in part by the National Institutes of Health. The researchers had no financial conflicts to disclose.
SOURCE: Balakrishnan M et al. Clin Gastroenterol Hepatol. 2020 Apr 30. doi: 10.1016/j.cgh.2020.04.067.
FROM CLINICAL GASTROENTEROLOGY AND HEPATOLOGY
Heavy menstrual bleeding difficult to control in young patients with inherited platelet disorders
Physician consensus and a broadly effective treatment for heavy menstrual bleeding was not found among young patients with inherited platelet function disorders, according to the results of a retrospective chart review reported in the Journal of Pediatric and Adolescent Gynecology.
Heavy menstrual bleeding (HMB) in girls with inherited platelet function disorders (IPFD) can be difficult to control despite ongoing follow-up and treatment changes, reported Christine M. Pennesi, MD, of the University of Michigan, Ann Arbor, and colleagues.
They assessed 34 young women and girls (ages 9-25 years) diagnosed with IPFDs referred to gynecology and/or hematology at a tertiary care hospital between 2006 and 2018.
Billing codes were used to determine hormonal or nonhormonal treatments, and outcomes over a 1- to 2-year period were collected. The initial treatment was defined as the first treatment prescribed after referral. The primary outcome was treatment failure, defined as a change in treatment method because of continued bleeding.
The majority (56%) of patients failed initial treatment (n = 19); among all 34 individuals followed in the study, an average of 2.7 total treatments were required.
Six patients (18%) remained uncontrolled despite numerous treatment changes (mean treatment changes, four; range, two to seven), and two patients (6%) remained uncontrolled because of noncompliance with treatment.
Overall, the researchers identified a 18% failure rate of successfully treatment of HMB in young women and girls with IPFDs over a 2-year follow-up period.
Of the 26 women who achieved control of HMB within 2-year follow-up, 54% (n = 14) were on hormonal treatments, 27% (n = 7) on nonhormonal treatments, 12% (n = 3) on combined treatments, and 8% (n = 2) on no treatment at time of control, the authors stated.
“The heterogeneity in treatments that were described in this study, clearly demonstrate that, in selecting treatment methods for HMB in young women, other considerations are often in play. This includes patient preference and need for contraception. Some patients or parents may have personal or religious objections to hormonal methods or worry about hormones in this young age group,” the researchers speculated.
“Appropriate counseling in these patients should include that it would not be unexpected for a patient to need more than one treatment before control of bleeding is achieved. This may help to alleviate the fear of teenagers when continued bleeding occurs after starting their initial treatment,” Dr. Pennesi and colleagues concluded.
One of the authors participated in funded trials and received funding from several pharmaceutical companies. The others reported having no disclosures.
SOURCE: Pennesi CM et al. J Pediatr Adolesc Gynecol. 2020 Jun 22. doi: 10.1016/j.jpag.2020.06.019.
Physician consensus and a broadly effective treatment for heavy menstrual bleeding was not found among young patients with inherited platelet function disorders, according to the results of a retrospective chart review reported in the Journal of Pediatric and Adolescent Gynecology.
Heavy menstrual bleeding (HMB) in girls with inherited platelet function disorders (IPFD) can be difficult to control despite ongoing follow-up and treatment changes, reported Christine M. Pennesi, MD, of the University of Michigan, Ann Arbor, and colleagues.
They assessed 34 young women and girls (ages 9-25 years) diagnosed with IPFDs referred to gynecology and/or hematology at a tertiary care hospital between 2006 and 2018.
Billing codes were used to determine hormonal or nonhormonal treatments, and outcomes over a 1- to 2-year period were collected. The initial treatment was defined as the first treatment prescribed after referral. The primary outcome was treatment failure, defined as a change in treatment method because of continued bleeding.
The majority (56%) of patients failed initial treatment (n = 19); among all 34 individuals followed in the study, an average of 2.7 total treatments were required.
Six patients (18%) remained uncontrolled despite numerous treatment changes (mean treatment changes, four; range, two to seven), and two patients (6%) remained uncontrolled because of noncompliance with treatment.
Overall, the researchers identified a 18% failure rate of successfully treatment of HMB in young women and girls with IPFDs over a 2-year follow-up period.
Of the 26 women who achieved control of HMB within 2-year follow-up, 54% (n = 14) were on hormonal treatments, 27% (n = 7) on nonhormonal treatments, 12% (n = 3) on combined treatments, and 8% (n = 2) on no treatment at time of control, the authors stated.
“The heterogeneity in treatments that were described in this study, clearly demonstrate that, in selecting treatment methods for HMB in young women, other considerations are often in play. This includes patient preference and need for contraception. Some patients or parents may have personal or religious objections to hormonal methods or worry about hormones in this young age group,” the researchers speculated.
“Appropriate counseling in these patients should include that it would not be unexpected for a patient to need more than one treatment before control of bleeding is achieved. This may help to alleviate the fear of teenagers when continued bleeding occurs after starting their initial treatment,” Dr. Pennesi and colleagues concluded.
One of the authors participated in funded trials and received funding from several pharmaceutical companies. The others reported having no disclosures.
SOURCE: Pennesi CM et al. J Pediatr Adolesc Gynecol. 2020 Jun 22. doi: 10.1016/j.jpag.2020.06.019.
Physician consensus and a broadly effective treatment for heavy menstrual bleeding was not found among young patients with inherited platelet function disorders, according to the results of a retrospective chart review reported in the Journal of Pediatric and Adolescent Gynecology.
Heavy menstrual bleeding (HMB) in girls with inherited platelet function disorders (IPFD) can be difficult to control despite ongoing follow-up and treatment changes, reported Christine M. Pennesi, MD, of the University of Michigan, Ann Arbor, and colleagues.
They assessed 34 young women and girls (ages 9-25 years) diagnosed with IPFDs referred to gynecology and/or hematology at a tertiary care hospital between 2006 and 2018.
Billing codes were used to determine hormonal or nonhormonal treatments, and outcomes over a 1- to 2-year period were collected. The initial treatment was defined as the first treatment prescribed after referral. The primary outcome was treatment failure, defined as a change in treatment method because of continued bleeding.
The majority (56%) of patients failed initial treatment (n = 19); among all 34 individuals followed in the study, an average of 2.7 total treatments were required.
Six patients (18%) remained uncontrolled despite numerous treatment changes (mean treatment changes, four; range, two to seven), and two patients (6%) remained uncontrolled because of noncompliance with treatment.
Overall, the researchers identified a 18% failure rate of successfully treatment of HMB in young women and girls with IPFDs over a 2-year follow-up period.
Of the 26 women who achieved control of HMB within 2-year follow-up, 54% (n = 14) were on hormonal treatments, 27% (n = 7) on nonhormonal treatments, 12% (n = 3) on combined treatments, and 8% (n = 2) on no treatment at time of control, the authors stated.
“The heterogeneity in treatments that were described in this study, clearly demonstrate that, in selecting treatment methods for HMB in young women, other considerations are often in play. This includes patient preference and need for contraception. Some patients or parents may have personal or religious objections to hormonal methods or worry about hormones in this young age group,” the researchers speculated.
“Appropriate counseling in these patients should include that it would not be unexpected for a patient to need more than one treatment before control of bleeding is achieved. This may help to alleviate the fear of teenagers when continued bleeding occurs after starting their initial treatment,” Dr. Pennesi and colleagues concluded.
One of the authors participated in funded trials and received funding from several pharmaceutical companies. The others reported having no disclosures.
SOURCE: Pennesi CM et al. J Pediatr Adolesc Gynecol. 2020 Jun 22. doi: 10.1016/j.jpag.2020.06.019.
FROM THE JOURNAL OF PEDIATRIC AND ADOLESCENT GYNECOLOGY
Lipophilic statins linked to lower mortality in ovarian cancer
, findings from a large observational study suggest.
The study included 10,062 patients with epithelial ovarian cancer enrolled in the Finnish national cancer registry. There were 2,621 patients who were prescribed statins between 1995 and 2015, and 80% of them used lipophilic statins.
When compared with no statin use, any statin use was associated with a 40% reduction in ovarian cancer mortality (weighted hazard ratio, 0.60), and any use of lipophilic statins was associated with a 43% reduction in ovarian cancer mortality (wHR, 0.57).
Kala Visvanathan, MD, of Johns Hopkins University in Baltimore, and colleagues reported these findings in a poster at the AACR virtual meeting II.
Reductions in ovarian cancer mortality were observed in women who took simvastatin or atorvastatin (wHRs 0.24 and 0.20, respectively), the researchers found.
Lipophilic statin use also was associated with a reduction in ovarian cancer mortality across disease subtypes, although the magnitude of reduction varied. The hazard ratios were 0.60 for high-grade serous ovarian cancer, 0.50 for endometrioid ovarian cancer, 0.20 for clear cell ovarian cancer, 0.30 for mucinous ovarian cancer, and 0.27 for borderline disease.
Survival benefits were evident both in patients who started statins prior to their ovarian cancer diagnosis and in those who started statins after diagnosis.
Never-statin users had a median age of 62 years at baseline, and ever-statin users had a median age of 67 years. The median follow-up was 3.6 years and 5.5 years, respectively.
Data from the registry were linked to prescription claims, and a series of analyses were conducted to examine the association between pre- and postdiagnostic statin use and mortality. The findings were adjusted for age at diagnosis, stage, ovarian cancer subtype, treatments, year of diagnosis, and chronic disease medications. Adherence to statins was greater than 90%.
Implications and next steps
The idea of using statins for the treatment of ovarian cancer is appealing because of the promising survival data as well as the broad access, low cost, and tolerability of statins, Dr. Visvanathan said in a statement. About 28% of U.S. adults over age 40 routinely take statins for cholesterol control, and statins are widely used in other countries, she said.
“Our results support research to evaluate the repurposing of therapies that are well tolerated and inexpensive in order to help reduce the global cancer burden,” Dr. Visvanathan and colleagues wrote in their poster.
“Our results provide evidence in support of the evaluation of lipophilic statins, particularly atorvastatin and/or simvastatin, for the treatment of [epithelial ovarian cancer] in conjunction with existing therapies,” the researchers wrote. They added that these statins should be “evaluated in randomized clinical trials that include correlative endpoints.”
Further, the researchers argued that “the results are biologically plausible based on known mechanisms associated with statin use and highlight the fact that statins may be effective to treat more than one disease/outcome (i.e., high cholesterol, EOC [epithelial ovarian cancer], breast cancer).”
The results of this study are intriguing, according to James Yarmolinsky, MSc, of the University of Bristol, England. Mr. Yarmolinsky is the lead author of a case-control study that showed an association between genetically proxied 3-hydroxy-3-methylglutaryl coenzyme A (HMG-CoA) reductase inhibition and lower odds of developing epithelial ovarian cancer (JAMA. 2020;323[7]:646-655).
Mr. Yarmolinsky and colleagues found that HMG-CoA reductase inhibition equivalent to a 38.7-mg/dL reduction in low-density lipoprotein cholesterol was significantly associated with lower odds of epithelial ovarian cancer in the general population (odds ratio, 0.60) and among BRCA1/2 mutation carriers (hazard ratio, 0.69). The findings raised questions about whether a similar association would be seen with medications such as statins that inhibit HMG-CoA reductase.
“These findings linking statin use to lower ovarian cancer mortality are really interesting given our own research suggesting that these drugs may also lower women’s risk of developing this disease in the first place,” Mr. Yarmolinsky said.
“The survival rate for ovarian cancer remains the lowest among all gynecological cancers in the United States, so use of these medications in either a preventive or therapeutic context could offer an important approach for reducing disease burden,” he added. “If the findings reported by Visvanathan and colleagues can be shown to replicate in other large population-based studies, testing the efficacy of statins in a randomized clinical trial could provide definitive evidence of whether these medications lower ovarian cancer mortality.”
The Department of Defense and the Breast Cancer Research Foundation funded the current study. Dr. Visvanathan and Mr. Yarmolinsky reported no disclosures.
SOURCE: Visvanathan K et al. AACR 2020, Abstract 5782.
, findings from a large observational study suggest.
The study included 10,062 patients with epithelial ovarian cancer enrolled in the Finnish national cancer registry. There were 2,621 patients who were prescribed statins between 1995 and 2015, and 80% of them used lipophilic statins.
When compared with no statin use, any statin use was associated with a 40% reduction in ovarian cancer mortality (weighted hazard ratio, 0.60), and any use of lipophilic statins was associated with a 43% reduction in ovarian cancer mortality (wHR, 0.57).
Kala Visvanathan, MD, of Johns Hopkins University in Baltimore, and colleagues reported these findings in a poster at the AACR virtual meeting II.
Reductions in ovarian cancer mortality were observed in women who took simvastatin or atorvastatin (wHRs 0.24 and 0.20, respectively), the researchers found.
Lipophilic statin use also was associated with a reduction in ovarian cancer mortality across disease subtypes, although the magnitude of reduction varied. The hazard ratios were 0.60 for high-grade serous ovarian cancer, 0.50 for endometrioid ovarian cancer, 0.20 for clear cell ovarian cancer, 0.30 for mucinous ovarian cancer, and 0.27 for borderline disease.
Survival benefits were evident both in patients who started statins prior to their ovarian cancer diagnosis and in those who started statins after diagnosis.
Never-statin users had a median age of 62 years at baseline, and ever-statin users had a median age of 67 years. The median follow-up was 3.6 years and 5.5 years, respectively.
Data from the registry were linked to prescription claims, and a series of analyses were conducted to examine the association between pre- and postdiagnostic statin use and mortality. The findings were adjusted for age at diagnosis, stage, ovarian cancer subtype, treatments, year of diagnosis, and chronic disease medications. Adherence to statins was greater than 90%.
Implications and next steps
The idea of using statins for the treatment of ovarian cancer is appealing because of the promising survival data as well as the broad access, low cost, and tolerability of statins, Dr. Visvanathan said in a statement. About 28% of U.S. adults over age 40 routinely take statins for cholesterol control, and statins are widely used in other countries, she said.
“Our results support research to evaluate the repurposing of therapies that are well tolerated and inexpensive in order to help reduce the global cancer burden,” Dr. Visvanathan and colleagues wrote in their poster.
“Our results provide evidence in support of the evaluation of lipophilic statins, particularly atorvastatin and/or simvastatin, for the treatment of [epithelial ovarian cancer] in conjunction with existing therapies,” the researchers wrote. They added that these statins should be “evaluated in randomized clinical trials that include correlative endpoints.”
Further, the researchers argued that “the results are biologically plausible based on known mechanisms associated with statin use and highlight the fact that statins may be effective to treat more than one disease/outcome (i.e., high cholesterol, EOC [epithelial ovarian cancer], breast cancer).”
The results of this study are intriguing, according to James Yarmolinsky, MSc, of the University of Bristol, England. Mr. Yarmolinsky is the lead author of a case-control study that showed an association between genetically proxied 3-hydroxy-3-methylglutaryl coenzyme A (HMG-CoA) reductase inhibition and lower odds of developing epithelial ovarian cancer (JAMA. 2020;323[7]:646-655).
Mr. Yarmolinsky and colleagues found that HMG-CoA reductase inhibition equivalent to a 38.7-mg/dL reduction in low-density lipoprotein cholesterol was significantly associated with lower odds of epithelial ovarian cancer in the general population (odds ratio, 0.60) and among BRCA1/2 mutation carriers (hazard ratio, 0.69). The findings raised questions about whether a similar association would be seen with medications such as statins that inhibit HMG-CoA reductase.
“These findings linking statin use to lower ovarian cancer mortality are really interesting given our own research suggesting that these drugs may also lower women’s risk of developing this disease in the first place,” Mr. Yarmolinsky said.
“The survival rate for ovarian cancer remains the lowest among all gynecological cancers in the United States, so use of these medications in either a preventive or therapeutic context could offer an important approach for reducing disease burden,” he added. “If the findings reported by Visvanathan and colleagues can be shown to replicate in other large population-based studies, testing the efficacy of statins in a randomized clinical trial could provide definitive evidence of whether these medications lower ovarian cancer mortality.”
The Department of Defense and the Breast Cancer Research Foundation funded the current study. Dr. Visvanathan and Mr. Yarmolinsky reported no disclosures.
SOURCE: Visvanathan K et al. AACR 2020, Abstract 5782.
, findings from a large observational study suggest.
The study included 10,062 patients with epithelial ovarian cancer enrolled in the Finnish national cancer registry. There were 2,621 patients who were prescribed statins between 1995 and 2015, and 80% of them used lipophilic statins.
When compared with no statin use, any statin use was associated with a 40% reduction in ovarian cancer mortality (weighted hazard ratio, 0.60), and any use of lipophilic statins was associated with a 43% reduction in ovarian cancer mortality (wHR, 0.57).
Kala Visvanathan, MD, of Johns Hopkins University in Baltimore, and colleagues reported these findings in a poster at the AACR virtual meeting II.
Reductions in ovarian cancer mortality were observed in women who took simvastatin or atorvastatin (wHRs 0.24 and 0.20, respectively), the researchers found.
Lipophilic statin use also was associated with a reduction in ovarian cancer mortality across disease subtypes, although the magnitude of reduction varied. The hazard ratios were 0.60 for high-grade serous ovarian cancer, 0.50 for endometrioid ovarian cancer, 0.20 for clear cell ovarian cancer, 0.30 for mucinous ovarian cancer, and 0.27 for borderline disease.
Survival benefits were evident both in patients who started statins prior to their ovarian cancer diagnosis and in those who started statins after diagnosis.
Never-statin users had a median age of 62 years at baseline, and ever-statin users had a median age of 67 years. The median follow-up was 3.6 years and 5.5 years, respectively.
Data from the registry were linked to prescription claims, and a series of analyses were conducted to examine the association between pre- and postdiagnostic statin use and mortality. The findings were adjusted for age at diagnosis, stage, ovarian cancer subtype, treatments, year of diagnosis, and chronic disease medications. Adherence to statins was greater than 90%.
Implications and next steps
The idea of using statins for the treatment of ovarian cancer is appealing because of the promising survival data as well as the broad access, low cost, and tolerability of statins, Dr. Visvanathan said in a statement. About 28% of U.S. adults over age 40 routinely take statins for cholesterol control, and statins are widely used in other countries, she said.
“Our results support research to evaluate the repurposing of therapies that are well tolerated and inexpensive in order to help reduce the global cancer burden,” Dr. Visvanathan and colleagues wrote in their poster.
“Our results provide evidence in support of the evaluation of lipophilic statins, particularly atorvastatin and/or simvastatin, for the treatment of [epithelial ovarian cancer] in conjunction with existing therapies,” the researchers wrote. They added that these statins should be “evaluated in randomized clinical trials that include correlative endpoints.”
Further, the researchers argued that “the results are biologically plausible based on known mechanisms associated with statin use and highlight the fact that statins may be effective to treat more than one disease/outcome (i.e., high cholesterol, EOC [epithelial ovarian cancer], breast cancer).”
The results of this study are intriguing, according to James Yarmolinsky, MSc, of the University of Bristol, England. Mr. Yarmolinsky is the lead author of a case-control study that showed an association between genetically proxied 3-hydroxy-3-methylglutaryl coenzyme A (HMG-CoA) reductase inhibition and lower odds of developing epithelial ovarian cancer (JAMA. 2020;323[7]:646-655).
Mr. Yarmolinsky and colleagues found that HMG-CoA reductase inhibition equivalent to a 38.7-mg/dL reduction in low-density lipoprotein cholesterol was significantly associated with lower odds of epithelial ovarian cancer in the general population (odds ratio, 0.60) and among BRCA1/2 mutation carriers (hazard ratio, 0.69). The findings raised questions about whether a similar association would be seen with medications such as statins that inhibit HMG-CoA reductase.
“These findings linking statin use to lower ovarian cancer mortality are really interesting given our own research suggesting that these drugs may also lower women’s risk of developing this disease in the first place,” Mr. Yarmolinsky said.
“The survival rate for ovarian cancer remains the lowest among all gynecological cancers in the United States, so use of these medications in either a preventive or therapeutic context could offer an important approach for reducing disease burden,” he added. “If the findings reported by Visvanathan and colleagues can be shown to replicate in other large population-based studies, testing the efficacy of statins in a randomized clinical trial could provide definitive evidence of whether these medications lower ovarian cancer mortality.”
The Department of Defense and the Breast Cancer Research Foundation funded the current study. Dr. Visvanathan and Mr. Yarmolinsky reported no disclosures.
SOURCE: Visvanathan K et al. AACR 2020, Abstract 5782.
FROM AACR 2020
Pubovaginal sling during urethral diverticulectomy reduces stress incontinence
a large retrospective cohort study has found.
However, in 66% of cases in which the diverticulectomy alone was performed, women also saw their stress urinary incontinence (SUI) resolve.
For a study published online in the American Journal of Obstetrics and Gynecology, Sarah E. Bradley, MD, of Georgetown University in Washington and colleagues analyzed records for 485 urethral diverticulectomies performed at 11 institutions over a 16-year period. One-fifth of patients had an autologous fascial pubovaginal sling (PVS) placed at the time of surgery.
The concomitant sling was associated with a significantly greater reduction of SUI after adjustment for prior diverticulectomy, prior incontinence surgery, age, race, and parity (adjusted odds ratio, 2.27; 95% confidence interval, 1.02-5.03; P = .043).
However, 10% of women in the sling-treated group had recurrent UTI from 6 weeks after surgery, compared with 3% of those in the diverticulectomy-only group (P = .001). Even after adjustment for higher rates of UTI before surgery in the sling group, the odds of recurrent UTI still were higher with the concomitant sling. Women within the sling group also were more likely to experience urinary retention at more than 6 weeks after surgery (8% vs. 1%; P equal to .0001).
Dr. Bradley and her colleagues noted that theirs was the largest study to date evaluating postoperative SUI in patients undergoing diverticulectomy with and without a PVS, noting that many surgeons do not routinely offer the sling at the time of diverticulectomy.
They also acknowledged a selection bias in their study. “With the previously thought theoretical increased risk of the addition of PVS, it is likely that most providers would prefer only to offer this concomitant procedure to those with significantly bothersome SUI. Additionally, the majority of women that underwent PVS (83%) came from two of the 11 participating institutions,” the researchers wrote.
In an interview, Catherine A. Mathews, MD, of Wake Forest University in Winston Salem, N.C., argued for a different interpretation of the study’s results.
“The study was beautifully done and it’s an ideal subject for a review, but in some respect the authors missed the opportunity to highlight that there was a spontaneous resolution of stress incontinence symptoms in 66% of women who received diverticulectomy alone,” Dr. Matthews said, adding that this has important implications for medical decision-making and patient choice.
“Morbidity associated with the pubovaginal sling was very low in this study, probably because it was being done by very proficient surgeons, but in many centers it is higher,” Dr. Matthews said. Even with the overall low morbidity seen in the study, “there was still a significant price to pay” for some women in the pubovaginal sling–treated group. “Recurrent UTI can be challenging to manage in the long term, with antibiotic morbidity and significant symptom bother. For the patients with urinary retention, having to manage it with a catheter is a really awful.”
Dr. Matthews said that the study made a case for interval, rather than concomitant, sling placement in women undergoing urethral diverticulectomy. “If you have a patient who insists on addressing symptoms concomitantly, this study provides good information about the long-term likelihood of two complications: urinary retention and recurrent UTI,” she said. “The vast majority of patients that I’m counseling would choose not to have the sling because of these complications.” And while avoiding reoperation may seem a good reason to opt for the PVS during diverticulectomy, the sling was not associated with a decrease in reoperations, compared with diverticulectomy alone, she noted.
“As we can see in the study, diverticulectomy itself has a high impact on stress incontinence,” Dr. Matthews continued. “If you restore the urethral anatomy and wait for the urethra to heal, you have a very good chance that the incontinence resolves.” For those women who do not see resolution and whose symptoms are still severe enough to bother them, “you’d have the flexibility postoperatively to offer not only a pubovaginal sling, but a synthetic mesh sling or a urethral bulking procedure.”
Dr. Bradley and her colleagues reported no relevant financial disclosures. Dr. Matthews disclosed financial support from Boston Scientific and serving as an expert witness for Johnson & Johnson.
SOURCE: Bradley SE et al. Am J Obstet Gynecol. 2020. doi: 10.1016/j.ajog.2020.06.002.
a large retrospective cohort study has found.
However, in 66% of cases in which the diverticulectomy alone was performed, women also saw their stress urinary incontinence (SUI) resolve.
For a study published online in the American Journal of Obstetrics and Gynecology, Sarah E. Bradley, MD, of Georgetown University in Washington and colleagues analyzed records for 485 urethral diverticulectomies performed at 11 institutions over a 16-year period. One-fifth of patients had an autologous fascial pubovaginal sling (PVS) placed at the time of surgery.
The concomitant sling was associated with a significantly greater reduction of SUI after adjustment for prior diverticulectomy, prior incontinence surgery, age, race, and parity (adjusted odds ratio, 2.27; 95% confidence interval, 1.02-5.03; P = .043).
However, 10% of women in the sling-treated group had recurrent UTI from 6 weeks after surgery, compared with 3% of those in the diverticulectomy-only group (P = .001). Even after adjustment for higher rates of UTI before surgery in the sling group, the odds of recurrent UTI still were higher with the concomitant sling. Women within the sling group also were more likely to experience urinary retention at more than 6 weeks after surgery (8% vs. 1%; P equal to .0001).
Dr. Bradley and her colleagues noted that theirs was the largest study to date evaluating postoperative SUI in patients undergoing diverticulectomy with and without a PVS, noting that many surgeons do not routinely offer the sling at the time of diverticulectomy.
They also acknowledged a selection bias in their study. “With the previously thought theoretical increased risk of the addition of PVS, it is likely that most providers would prefer only to offer this concomitant procedure to those with significantly bothersome SUI. Additionally, the majority of women that underwent PVS (83%) came from two of the 11 participating institutions,” the researchers wrote.
In an interview, Catherine A. Mathews, MD, of Wake Forest University in Winston Salem, N.C., argued for a different interpretation of the study’s results.
“The study was beautifully done and it’s an ideal subject for a review, but in some respect the authors missed the opportunity to highlight that there was a spontaneous resolution of stress incontinence symptoms in 66% of women who received diverticulectomy alone,” Dr. Matthews said, adding that this has important implications for medical decision-making and patient choice.
“Morbidity associated with the pubovaginal sling was very low in this study, probably because it was being done by very proficient surgeons, but in many centers it is higher,” Dr. Matthews said. Even with the overall low morbidity seen in the study, “there was still a significant price to pay” for some women in the pubovaginal sling–treated group. “Recurrent UTI can be challenging to manage in the long term, with antibiotic morbidity and significant symptom bother. For the patients with urinary retention, having to manage it with a catheter is a really awful.”
Dr. Matthews said that the study made a case for interval, rather than concomitant, sling placement in women undergoing urethral diverticulectomy. “If you have a patient who insists on addressing symptoms concomitantly, this study provides good information about the long-term likelihood of two complications: urinary retention and recurrent UTI,” she said. “The vast majority of patients that I’m counseling would choose not to have the sling because of these complications.” And while avoiding reoperation may seem a good reason to opt for the PVS during diverticulectomy, the sling was not associated with a decrease in reoperations, compared with diverticulectomy alone, she noted.
“As we can see in the study, diverticulectomy itself has a high impact on stress incontinence,” Dr. Matthews continued. “If you restore the urethral anatomy and wait for the urethra to heal, you have a very good chance that the incontinence resolves.” For those women who do not see resolution and whose symptoms are still severe enough to bother them, “you’d have the flexibility postoperatively to offer not only a pubovaginal sling, but a synthetic mesh sling or a urethral bulking procedure.”
Dr. Bradley and her colleagues reported no relevant financial disclosures. Dr. Matthews disclosed financial support from Boston Scientific and serving as an expert witness for Johnson & Johnson.
SOURCE: Bradley SE et al. Am J Obstet Gynecol. 2020. doi: 10.1016/j.ajog.2020.06.002.
a large retrospective cohort study has found.
However, in 66% of cases in which the diverticulectomy alone was performed, women also saw their stress urinary incontinence (SUI) resolve.
For a study published online in the American Journal of Obstetrics and Gynecology, Sarah E. Bradley, MD, of Georgetown University in Washington and colleagues analyzed records for 485 urethral diverticulectomies performed at 11 institutions over a 16-year period. One-fifth of patients had an autologous fascial pubovaginal sling (PVS) placed at the time of surgery.
The concomitant sling was associated with a significantly greater reduction of SUI after adjustment for prior diverticulectomy, prior incontinence surgery, age, race, and parity (adjusted odds ratio, 2.27; 95% confidence interval, 1.02-5.03; P = .043).
However, 10% of women in the sling-treated group had recurrent UTI from 6 weeks after surgery, compared with 3% of those in the diverticulectomy-only group (P = .001). Even after adjustment for higher rates of UTI before surgery in the sling group, the odds of recurrent UTI still were higher with the concomitant sling. Women within the sling group also were more likely to experience urinary retention at more than 6 weeks after surgery (8% vs. 1%; P equal to .0001).
Dr. Bradley and her colleagues noted that theirs was the largest study to date evaluating postoperative SUI in patients undergoing diverticulectomy with and without a PVS, noting that many surgeons do not routinely offer the sling at the time of diverticulectomy.
They also acknowledged a selection bias in their study. “With the previously thought theoretical increased risk of the addition of PVS, it is likely that most providers would prefer only to offer this concomitant procedure to those with significantly bothersome SUI. Additionally, the majority of women that underwent PVS (83%) came from two of the 11 participating institutions,” the researchers wrote.
In an interview, Catherine A. Mathews, MD, of Wake Forest University in Winston Salem, N.C., argued for a different interpretation of the study’s results.
“The study was beautifully done and it’s an ideal subject for a review, but in some respect the authors missed the opportunity to highlight that there was a spontaneous resolution of stress incontinence symptoms in 66% of women who received diverticulectomy alone,” Dr. Matthews said, adding that this has important implications for medical decision-making and patient choice.
“Morbidity associated with the pubovaginal sling was very low in this study, probably because it was being done by very proficient surgeons, but in many centers it is higher,” Dr. Matthews said. Even with the overall low morbidity seen in the study, “there was still a significant price to pay” for some women in the pubovaginal sling–treated group. “Recurrent UTI can be challenging to manage in the long term, with antibiotic morbidity and significant symptom bother. For the patients with urinary retention, having to manage it with a catheter is a really awful.”
Dr. Matthews said that the study made a case for interval, rather than concomitant, sling placement in women undergoing urethral diverticulectomy. “If you have a patient who insists on addressing symptoms concomitantly, this study provides good information about the long-term likelihood of two complications: urinary retention and recurrent UTI,” she said. “The vast majority of patients that I’m counseling would choose not to have the sling because of these complications.” And while avoiding reoperation may seem a good reason to opt for the PVS during diverticulectomy, the sling was not associated with a decrease in reoperations, compared with diverticulectomy alone, she noted.
“As we can see in the study, diverticulectomy itself has a high impact on stress incontinence,” Dr. Matthews continued. “If you restore the urethral anatomy and wait for the urethra to heal, you have a very good chance that the incontinence resolves.” For those women who do not see resolution and whose symptoms are still severe enough to bother them, “you’d have the flexibility postoperatively to offer not only a pubovaginal sling, but a synthetic mesh sling or a urethral bulking procedure.”
Dr. Bradley and her colleagues reported no relevant financial disclosures. Dr. Matthews disclosed financial support from Boston Scientific and serving as an expert witness for Johnson & Johnson.
SOURCE: Bradley SE et al. Am J Obstet Gynecol. 2020. doi: 10.1016/j.ajog.2020.06.002.
FROM AMERICAN JOURNAL OF OBSTETRICS AND GYNECOLOGY






