User login
Official Newspaper of the American College of Surgeons
Big trials deflate off-pump CABG
SAN FRANCISCO – Off-pump coronary artery bypass graft surgery offered no advantages over on-pump CABG in any major endpoints at 1 year of follow-up in two major prospective randomized trials totaling more than 7,000 patients.
Among the key 1-year outcomes – which didn’t differ between off- and on-pump CABG patients in the GOPCABE and CORONARY trials – were death, MI, stroke, neurocognitive function, quality of life, renal failure, and repeat revascularization, investigators reported at the annual meeting of the American College of Cardiology.
A third randomized trial presented at the same session of the ACC meeting did find a significant outcome advantage favoring off-pump CABG in high-operative-risk patients at 1 year. However, experts discounted this Czech study because it was small, single center, reported only 30-day results, and the advantage found for off-pump surgery hinged on an outdated and inadequate definition of MI.
The new study findings signal a striking fall from grace for off-pump CABG. Not long ago, this technique, while controversial, was viewed by many as a progressive development within heart surgery, one that would revitalize a mature operation whose annual case numbers were declining in the face of stiff competition from percutaneous coronary intervention by cardiologists. Off-pump CABG was an innovation designed to avoid the perioperative complications related to aortic cross-clamping and the heart-lung machine, including the lingering neurocognitive dysfunction known informally in surgical circles as “pump head.”
However, the resounding lack of any demonstrable advantages for off-pump CABG in the two large trials presented in San Francisco left analysts scratching their heads as to the role remaining for this beating heart surgical technique, which is more difficult to learn and perform skillfully than on-pump bypass.
Speaking perhaps for many puzzled cardiologists in the packed audience in San Francisco, Dr. Christopher P. Cannon of Harvard Medical School, Boston, asked the surgeon-presenters of the CORONARY and GOPCABE trials, “Why would you want to do off-pump surgery if it doesn’t seem to help and it’s harder to do?”
Discussant Dr. Michael J. Mack, a cardiac surgeon, voiced a similar sentiment.
“I was an early advocate of off-pump surgery. But as a card-carrying off-pump bypass surgeon, it’s getting harder and harder for me to maintain enthusiasm for a potential benefit from this,” declared Dr. Mack, medical director of cardiovascular surgery for the Baylor Health Care System and director of cardiovascular research at the Heart Hospital in Plano, Tex.
He noted that the GOPCABE and CORONARY trials follow upon the earlier ROOBY (Randomized On/Off Bypass) trial, which actually showed worse outcomes in the off-pump group. ROOBY enrolled 2,203 Veterans Affairs patients, with the off-pump CABG group having a significantly higher 1-year rate of the primary composite endpoint comprising death, nonfatal MI, or repeat revascularization, along with worse graft patency (N. Engl. J. Med. 2009;361:1827-37).
GOPCABE and CORONARY were designed in part to answer critics of ROOBY, who have argued that the VA trial used insufficiently experienced off-pump CABG surgeons and featured a patient population at too low an operative risk to detect a signal of benefit favoring off-pump surgery.
The GOPCABE (German Off-Pump Coronary Artery Bypass Grafting in Elderly Patients) study involved 2,539 patients aged 75 years or older randomized at 12 German centers. A total of 60% of patients had triple-vessel disease, and no one was excluded from the trial because of left ventricular function or coronary artery anatomy.
Participating surgeons were highly experienced. Those who performed off-pump CABG in the study had previously done an average of 514 of them, while the on-pump surgeons had done an average of 1,378 of those operations.
“We wanted to have the best off-pump vs. the best on-pump surgeons, like in a competition,” explained Dr. Anno Diegeler, a surgeon at the Bad Neustadt (Germany) Heart Center.
He and his coinvestigators conducted GOPCABE because they believed it would be easier to show advantages for off-pump CABG in a population at high operative risk, such as elderly patients with many comorbidities. Indeed, the study hypothesis was that the off-pump group would show a robust 30% reduction in the primary endpoint, a composite of death, stroke, myocardial infarction, repeat revascularization, or new renal-replacement therapy at 1 year.
That didn’t happen. The 30-day rate of the primary endpoint was 7.8% in the off-pump group and 8.2% with on-pump CABG, while the 1-year rates were 13.1% and 14.0%, respectively. None of the individual components of the composite endpoint differed significantly between the groups, either.
Neurocognitive function wasn’t measured in GOPCABE, but it was in CORONARY (the CABG Off or On Pump Revascularization Study), which involved 4,752 randomized patients in 19 countries.
Dr. Andre Lamy presented the 1-year results. The primary endpoint was a composite of death, MI, stroke, or new renal failure requiring dialysis. The rate was 12.1% in patients in the off-pump group and similar at 13.3% in the on-pump group.
As in GOPCABE, the surgeons participating in CORONARY were highly proficient. They had to have at least 2 years’ experience as a staff cardiac surgeon and at least 100 prior cases of whichever operation they were assigned to. The vast majority met that standard for both procedures.
Quality of life and neurocognitive tests were conducted preoperatively, at discharge, at 30 days, and again at 1 year. Quality of life as assessed by the European Quality of Life-5 Dimensions (EQ-5D) questionnaire showed significant improvement over time in both study arms, with no difference between the two groups.
Nor were there any significant between-group differences in neurocognitive function as assessed using the Montreal Cognitive Assessment, the Digit Symbol Substitution Test, and the Trail Making Test Part B. However, neurocognitive testing was declared optional because it’s so time consuming, and many patients opted out. For example, only 1,273 of the original 4,752 patients returned to take the Montreal Cognitive Assessment at 1 year, noted Dr. Lamy, a heart surgeon at the Population Health Research Institute at McMaster University in Hamilton, Ont.
Discussant Dr. Bernard Gersh zeroed in on the incomplete neurocognitive testing.
“I think this is really a significant limitation. There’s a huge bias. If there’s any advantage to off-pump CABG, it may be in neurocognitive dysfunction,” commented Dr. Gersh, professor of medicine at the Mayo Clinic, Rochester, Minn.
In response to the discussants’ questioning as to where off-pump CABG fits into clinical practice in light of the disappointing CORONARY and GOPCABE findings, Dr. Diegeler said he remains convinced that some high-operative-risk patients – those with aortic calcification or other evidence of generalized vascular disease ‑ do benefit preferentially from off-pump surgery when performed by expert surgeons.
“It’s hard to prove that in a randomized clinical trial, though,” the surgeon conceded.
Dr. Lamy said a post hoc analysis of the CORONARY data showed that low-operative-risk patients as defined by a EuroScore of 0-2 tended to do better with on- than off-pump CABG, while the converse was true in those with moderate- or high-risk scores.
“In my personal practice now, my low-risk patients go on-pump and my moderate- and high-risk patients go off-pump,” he added.
Both surgeons were firmly in favor of off-pump CABG continuing to be available at expert centers so that surgeons can individualize their operation to suit a patient’s needs.
Dr. Jan Hlavicka presented the results of the PRAGUE-6 trial, in which 206 patients at high operative risk – a EuroScore of 6 or greater – were randomized to off- or on-pump CABG at Charles University, Prague. The operations were performed by five surgeons proficient in both procedures.
The 30-day primary composite endpoint comprising death, MI, stroke, or new renal failure requiring dialysis occurred in 20.6% of the on-pump group, compared with 9.2% of off-pump patients.
The off-pump group required significantly fewer RBC transfusions. However, there were no significant differences between the two groups in terms of average hospital length of stay, wound infection rates, or total hospital costs.
“We believe off-pump bypass in high-risk patients has a lower incidence of serious complications and is a safer means of direct revascularization in these patients,” concluded Dr. Hlavicka, a cardiac surgeon at the university.
Dr. Gersh noted that the only significant difference between the two groups in the individual components of the primary endpoint was in acute MI rates: 12.1% in the on- vs. 4.1% in the off-pump CABG group. He took issue with the Czech investigators’ use of the 2004 Society of Thoracic Surgeons definition of acute MI, which in the first 24 hours after surgery requires only an elevation of creatine kinase–MB at least five times the upper limit of normal with no need for new Q waves. That’s not sufficiently stringent. It surely captures many patients who don’t really have an acute MI. The data should be reanalyzed using a contemporary definition which requires new Q waves, he added.
The CORONARY trial was funded by the Canadian Institutes of Health Research; simultaneous with Dr. Lamy’s presentation of the data in San Francisco, the study was published online (N. Engl. J. Med. 2013 [doi:10.1056/NEJMoa1301228]).
The GOPCABE trial was sponsored by the German Society for Cardiovascular Surgery with funding support by Maquet. It was published online simultaneously with Dr. Diegeler’s ACC presentation (N. Engl. J. Med. 2013 [doi:10.1056/NEJMoa1211666]).
Dr. Hlavicka, Dr. Lamy, and Dr. Diegeler declared having no financial conflicts.
SAN FRANCISCO – Off-pump coronary artery bypass graft surgery offered no advantages over on-pump CABG in any major endpoints at 1 year of follow-up in two major prospective randomized trials totaling more than 7,000 patients.
Among the key 1-year outcomes – which didn’t differ between off- and on-pump CABG patients in the GOPCABE and CORONARY trials – were death, MI, stroke, neurocognitive function, quality of life, renal failure, and repeat revascularization, investigators reported at the annual meeting of the American College of Cardiology.
A third randomized trial presented at the same session of the ACC meeting did find a significant outcome advantage favoring off-pump CABG in high-operative-risk patients at 1 year. However, experts discounted this Czech study because it was small, single center, reported only 30-day results, and the advantage found for off-pump surgery hinged on an outdated and inadequate definition of MI.
The new study findings signal a striking fall from grace for off-pump CABG. Not long ago, this technique, while controversial, was viewed by many as a progressive development within heart surgery, one that would revitalize a mature operation whose annual case numbers were declining in the face of stiff competition from percutaneous coronary intervention by cardiologists. Off-pump CABG was an innovation designed to avoid the perioperative complications related to aortic cross-clamping and the heart-lung machine, including the lingering neurocognitive dysfunction known informally in surgical circles as “pump head.”
However, the resounding lack of any demonstrable advantages for off-pump CABG in the two large trials presented in San Francisco left analysts scratching their heads as to the role remaining for this beating heart surgical technique, which is more difficult to learn and perform skillfully than on-pump bypass.
Speaking perhaps for many puzzled cardiologists in the packed audience in San Francisco, Dr. Christopher P. Cannon of Harvard Medical School, Boston, asked the surgeon-presenters of the CORONARY and GOPCABE trials, “Why would you want to do off-pump surgery if it doesn’t seem to help and it’s harder to do?”
Discussant Dr. Michael J. Mack, a cardiac surgeon, voiced a similar sentiment.
“I was an early advocate of off-pump surgery. But as a card-carrying off-pump bypass surgeon, it’s getting harder and harder for me to maintain enthusiasm for a potential benefit from this,” declared Dr. Mack, medical director of cardiovascular surgery for the Baylor Health Care System and director of cardiovascular research at the Heart Hospital in Plano, Tex.
He noted that the GOPCABE and CORONARY trials follow upon the earlier ROOBY (Randomized On/Off Bypass) trial, which actually showed worse outcomes in the off-pump group. ROOBY enrolled 2,203 Veterans Affairs patients, with the off-pump CABG group having a significantly higher 1-year rate of the primary composite endpoint comprising death, nonfatal MI, or repeat revascularization, along with worse graft patency (N. Engl. J. Med. 2009;361:1827-37).
GOPCABE and CORONARY were designed in part to answer critics of ROOBY, who have argued that the VA trial used insufficiently experienced off-pump CABG surgeons and featured a patient population at too low an operative risk to detect a signal of benefit favoring off-pump surgery.
The GOPCABE (German Off-Pump Coronary Artery Bypass Grafting in Elderly Patients) study involved 2,539 patients aged 75 years or older randomized at 12 German centers. A total of 60% of patients had triple-vessel disease, and no one was excluded from the trial because of left ventricular function or coronary artery anatomy.
Participating surgeons were highly experienced. Those who performed off-pump CABG in the study had previously done an average of 514 of them, while the on-pump surgeons had done an average of 1,378 of those operations.
“We wanted to have the best off-pump vs. the best on-pump surgeons, like in a competition,” explained Dr. Anno Diegeler, a surgeon at the Bad Neustadt (Germany) Heart Center.
He and his coinvestigators conducted GOPCABE because they believed it would be easier to show advantages for off-pump CABG in a population at high operative risk, such as elderly patients with many comorbidities. Indeed, the study hypothesis was that the off-pump group would show a robust 30% reduction in the primary endpoint, a composite of death, stroke, myocardial infarction, repeat revascularization, or new renal-replacement therapy at 1 year.
That didn’t happen. The 30-day rate of the primary endpoint was 7.8% in the off-pump group and 8.2% with on-pump CABG, while the 1-year rates were 13.1% and 14.0%, respectively. None of the individual components of the composite endpoint differed significantly between the groups, either.
Neurocognitive function wasn’t measured in GOPCABE, but it was in CORONARY (the CABG Off or On Pump Revascularization Study), which involved 4,752 randomized patients in 19 countries.
Dr. Andre Lamy presented the 1-year results. The primary endpoint was a composite of death, MI, stroke, or new renal failure requiring dialysis. The rate was 12.1% in patients in the off-pump group and similar at 13.3% in the on-pump group.
As in GOPCABE, the surgeons participating in CORONARY were highly proficient. They had to have at least 2 years’ experience as a staff cardiac surgeon and at least 100 prior cases of whichever operation they were assigned to. The vast majority met that standard for both procedures.
Quality of life and neurocognitive tests were conducted preoperatively, at discharge, at 30 days, and again at 1 year. Quality of life as assessed by the European Quality of Life-5 Dimensions (EQ-5D) questionnaire showed significant improvement over time in both study arms, with no difference between the two groups.
Nor were there any significant between-group differences in neurocognitive function as assessed using the Montreal Cognitive Assessment, the Digit Symbol Substitution Test, and the Trail Making Test Part B. However, neurocognitive testing was declared optional because it’s so time consuming, and many patients opted out. For example, only 1,273 of the original 4,752 patients returned to take the Montreal Cognitive Assessment at 1 year, noted Dr. Lamy, a heart surgeon at the Population Health Research Institute at McMaster University in Hamilton, Ont.
Discussant Dr. Bernard Gersh zeroed in on the incomplete neurocognitive testing.
“I think this is really a significant limitation. There’s a huge bias. If there’s any advantage to off-pump CABG, it may be in neurocognitive dysfunction,” commented Dr. Gersh, professor of medicine at the Mayo Clinic, Rochester, Minn.
In response to the discussants’ questioning as to where off-pump CABG fits into clinical practice in light of the disappointing CORONARY and GOPCABE findings, Dr. Diegeler said he remains convinced that some high-operative-risk patients – those with aortic calcification or other evidence of generalized vascular disease ‑ do benefit preferentially from off-pump surgery when performed by expert surgeons.
“It’s hard to prove that in a randomized clinical trial, though,” the surgeon conceded.
Dr. Lamy said a post hoc analysis of the CORONARY data showed that low-operative-risk patients as defined by a EuroScore of 0-2 tended to do better with on- than off-pump CABG, while the converse was true in those with moderate- or high-risk scores.
“In my personal practice now, my low-risk patients go on-pump and my moderate- and high-risk patients go off-pump,” he added.
Both surgeons were firmly in favor of off-pump CABG continuing to be available at expert centers so that surgeons can individualize their operation to suit a patient’s needs.
Dr. Jan Hlavicka presented the results of the PRAGUE-6 trial, in which 206 patients at high operative risk – a EuroScore of 6 or greater – were randomized to off- or on-pump CABG at Charles University, Prague. The operations were performed by five surgeons proficient in both procedures.
The 30-day primary composite endpoint comprising death, MI, stroke, or new renal failure requiring dialysis occurred in 20.6% of the on-pump group, compared with 9.2% of off-pump patients.
The off-pump group required significantly fewer RBC transfusions. However, there were no significant differences between the two groups in terms of average hospital length of stay, wound infection rates, or total hospital costs.
“We believe off-pump bypass in high-risk patients has a lower incidence of serious complications and is a safer means of direct revascularization in these patients,” concluded Dr. Hlavicka, a cardiac surgeon at the university.
Dr. Gersh noted that the only significant difference between the two groups in the individual components of the primary endpoint was in acute MI rates: 12.1% in the on- vs. 4.1% in the off-pump CABG group. He took issue with the Czech investigators’ use of the 2004 Society of Thoracic Surgeons definition of acute MI, which in the first 24 hours after surgery requires only an elevation of creatine kinase–MB at least five times the upper limit of normal with no need for new Q waves. That’s not sufficiently stringent. It surely captures many patients who don’t really have an acute MI. The data should be reanalyzed using a contemporary definition which requires new Q waves, he added.
The CORONARY trial was funded by the Canadian Institutes of Health Research; simultaneous with Dr. Lamy’s presentation of the data in San Francisco, the study was published online (N. Engl. J. Med. 2013 [doi:10.1056/NEJMoa1301228]).
The GOPCABE trial was sponsored by the German Society for Cardiovascular Surgery with funding support by Maquet. It was published online simultaneously with Dr. Diegeler’s ACC presentation (N. Engl. J. Med. 2013 [doi:10.1056/NEJMoa1211666]).
Dr. Hlavicka, Dr. Lamy, and Dr. Diegeler declared having no financial conflicts.
SAN FRANCISCO – Off-pump coronary artery bypass graft surgery offered no advantages over on-pump CABG in any major endpoints at 1 year of follow-up in two major prospective randomized trials totaling more than 7,000 patients.
Among the key 1-year outcomes – which didn’t differ between off- and on-pump CABG patients in the GOPCABE and CORONARY trials – were death, MI, stroke, neurocognitive function, quality of life, renal failure, and repeat revascularization, investigators reported at the annual meeting of the American College of Cardiology.
A third randomized trial presented at the same session of the ACC meeting did find a significant outcome advantage favoring off-pump CABG in high-operative-risk patients at 1 year. However, experts discounted this Czech study because it was small, single center, reported only 30-day results, and the advantage found for off-pump surgery hinged on an outdated and inadequate definition of MI.
The new study findings signal a striking fall from grace for off-pump CABG. Not long ago, this technique, while controversial, was viewed by many as a progressive development within heart surgery, one that would revitalize a mature operation whose annual case numbers were declining in the face of stiff competition from percutaneous coronary intervention by cardiologists. Off-pump CABG was an innovation designed to avoid the perioperative complications related to aortic cross-clamping and the heart-lung machine, including the lingering neurocognitive dysfunction known informally in surgical circles as “pump head.”
However, the resounding lack of any demonstrable advantages for off-pump CABG in the two large trials presented in San Francisco left analysts scratching their heads as to the role remaining for this beating heart surgical technique, which is more difficult to learn and perform skillfully than on-pump bypass.
Speaking perhaps for many puzzled cardiologists in the packed audience in San Francisco, Dr. Christopher P. Cannon of Harvard Medical School, Boston, asked the surgeon-presenters of the CORONARY and GOPCABE trials, “Why would you want to do off-pump surgery if it doesn’t seem to help and it’s harder to do?”
Discussant Dr. Michael J. Mack, a cardiac surgeon, voiced a similar sentiment.
“I was an early advocate of off-pump surgery. But as a card-carrying off-pump bypass surgeon, it’s getting harder and harder for me to maintain enthusiasm for a potential benefit from this,” declared Dr. Mack, medical director of cardiovascular surgery for the Baylor Health Care System and director of cardiovascular research at the Heart Hospital in Plano, Tex.
He noted that the GOPCABE and CORONARY trials follow upon the earlier ROOBY (Randomized On/Off Bypass) trial, which actually showed worse outcomes in the off-pump group. ROOBY enrolled 2,203 Veterans Affairs patients, with the off-pump CABG group having a significantly higher 1-year rate of the primary composite endpoint comprising death, nonfatal MI, or repeat revascularization, along with worse graft patency (N. Engl. J. Med. 2009;361:1827-37).
GOPCABE and CORONARY were designed in part to answer critics of ROOBY, who have argued that the VA trial used insufficiently experienced off-pump CABG surgeons and featured a patient population at too low an operative risk to detect a signal of benefit favoring off-pump surgery.
The GOPCABE (German Off-Pump Coronary Artery Bypass Grafting in Elderly Patients) study involved 2,539 patients aged 75 years or older randomized at 12 German centers. A total of 60% of patients had triple-vessel disease, and no one was excluded from the trial because of left ventricular function or coronary artery anatomy.
Participating surgeons were highly experienced. Those who performed off-pump CABG in the study had previously done an average of 514 of them, while the on-pump surgeons had done an average of 1,378 of those operations.
“We wanted to have the best off-pump vs. the best on-pump surgeons, like in a competition,” explained Dr. Anno Diegeler, a surgeon at the Bad Neustadt (Germany) Heart Center.
He and his coinvestigators conducted GOPCABE because they believed it would be easier to show advantages for off-pump CABG in a population at high operative risk, such as elderly patients with many comorbidities. Indeed, the study hypothesis was that the off-pump group would show a robust 30% reduction in the primary endpoint, a composite of death, stroke, myocardial infarction, repeat revascularization, or new renal-replacement therapy at 1 year.
That didn’t happen. The 30-day rate of the primary endpoint was 7.8% in the off-pump group and 8.2% with on-pump CABG, while the 1-year rates were 13.1% and 14.0%, respectively. None of the individual components of the composite endpoint differed significantly between the groups, either.
Neurocognitive function wasn’t measured in GOPCABE, but it was in CORONARY (the CABG Off or On Pump Revascularization Study), which involved 4,752 randomized patients in 19 countries.
Dr. Andre Lamy presented the 1-year results. The primary endpoint was a composite of death, MI, stroke, or new renal failure requiring dialysis. The rate was 12.1% in patients in the off-pump group and similar at 13.3% in the on-pump group.
As in GOPCABE, the surgeons participating in CORONARY were highly proficient. They had to have at least 2 years’ experience as a staff cardiac surgeon and at least 100 prior cases of whichever operation they were assigned to. The vast majority met that standard for both procedures.
Quality of life and neurocognitive tests were conducted preoperatively, at discharge, at 30 days, and again at 1 year. Quality of life as assessed by the European Quality of Life-5 Dimensions (EQ-5D) questionnaire showed significant improvement over time in both study arms, with no difference between the two groups.
Nor were there any significant between-group differences in neurocognitive function as assessed using the Montreal Cognitive Assessment, the Digit Symbol Substitution Test, and the Trail Making Test Part B. However, neurocognitive testing was declared optional because it’s so time consuming, and many patients opted out. For example, only 1,273 of the original 4,752 patients returned to take the Montreal Cognitive Assessment at 1 year, noted Dr. Lamy, a heart surgeon at the Population Health Research Institute at McMaster University in Hamilton, Ont.
Discussant Dr. Bernard Gersh zeroed in on the incomplete neurocognitive testing.
“I think this is really a significant limitation. There’s a huge bias. If there’s any advantage to off-pump CABG, it may be in neurocognitive dysfunction,” commented Dr. Gersh, professor of medicine at the Mayo Clinic, Rochester, Minn.
In response to the discussants’ questioning as to where off-pump CABG fits into clinical practice in light of the disappointing CORONARY and GOPCABE findings, Dr. Diegeler said he remains convinced that some high-operative-risk patients – those with aortic calcification or other evidence of generalized vascular disease ‑ do benefit preferentially from off-pump surgery when performed by expert surgeons.
“It’s hard to prove that in a randomized clinical trial, though,” the surgeon conceded.
Dr. Lamy said a post hoc analysis of the CORONARY data showed that low-operative-risk patients as defined by a EuroScore of 0-2 tended to do better with on- than off-pump CABG, while the converse was true in those with moderate- or high-risk scores.
“In my personal practice now, my low-risk patients go on-pump and my moderate- and high-risk patients go off-pump,” he added.
Both surgeons were firmly in favor of off-pump CABG continuing to be available at expert centers so that surgeons can individualize their operation to suit a patient’s needs.
Dr. Jan Hlavicka presented the results of the PRAGUE-6 trial, in which 206 patients at high operative risk – a EuroScore of 6 or greater – were randomized to off- or on-pump CABG at Charles University, Prague. The operations were performed by five surgeons proficient in both procedures.
The 30-day primary composite endpoint comprising death, MI, stroke, or new renal failure requiring dialysis occurred in 20.6% of the on-pump group, compared with 9.2% of off-pump patients.
The off-pump group required significantly fewer RBC transfusions. However, there were no significant differences between the two groups in terms of average hospital length of stay, wound infection rates, or total hospital costs.
“We believe off-pump bypass in high-risk patients has a lower incidence of serious complications and is a safer means of direct revascularization in these patients,” concluded Dr. Hlavicka, a cardiac surgeon at the university.
Dr. Gersh noted that the only significant difference between the two groups in the individual components of the primary endpoint was in acute MI rates: 12.1% in the on- vs. 4.1% in the off-pump CABG group. He took issue with the Czech investigators’ use of the 2004 Society of Thoracic Surgeons definition of acute MI, which in the first 24 hours after surgery requires only an elevation of creatine kinase–MB at least five times the upper limit of normal with no need for new Q waves. That’s not sufficiently stringent. It surely captures many patients who don’t really have an acute MI. The data should be reanalyzed using a contemporary definition which requires new Q waves, he added.
The CORONARY trial was funded by the Canadian Institutes of Health Research; simultaneous with Dr. Lamy’s presentation of the data in San Francisco, the study was published online (N. Engl. J. Med. 2013 [doi:10.1056/NEJMoa1301228]).
The GOPCABE trial was sponsored by the German Society for Cardiovascular Surgery with funding support by Maquet. It was published online simultaneously with Dr. Diegeler’s ACC presentation (N. Engl. J. Med. 2013 [doi:10.1056/NEJMoa1211666]).
Dr. Hlavicka, Dr. Lamy, and Dr. Diegeler declared having no financial conflicts.
Major finding: Off-pump coronary artery bypass
graft surgery did not differ significantly from on-pump bypass in 1-year rates
of death, stroke, MI, or any other important outcomes in two large,
prospective, randomized trials.
Data source: The GOPCABE trial, in 2,539 randomized
patients; and the CORONARY trial, in 4,752 patients in 19 countries.
Disclosures: CORONARY was sponsored by the Canadian
Institutes of Health Research. GOPCABE was sponsored by the German Society for
Cardiovascular Surgery with funding support from Maquet. The presenters
reported having no financial conflicts.
Imaging agent approved for locating lymph nodes
A radioactive diagnostic imaging agent called Lymphoseek (technetium Tc 99m tilmanocept) Injection has been approved for locating lymph nodes in patients who have breast cancer or melanoma and are undergoing surgery to remove tumor-draining lymph nodes, the Food and Drug Administration announced.
Lymphoseek is the first new drug for lymph-node mapping to be approved in more than 30 years. Other FDA-approved drugs used for lymph-node mapping include sulfur colloid and isosulfan blue.
"To use Lymphoseek, doctors inject the drug into the tumor area and later, using a handheld radiation detector, find lymph nodes that have taken up Lymphoseek’s radioactivity," said Dr. Shaw Chen, deputy director of the Office of Drug Evaluation IV in the FDA’s Center for Drug Evaluation and Research.
Lymphoseek is marketed by Navidea Biopharmaceuticals. The manufacturer’s website notes that the ability to rapidly locate and biopsy sentinel nodes enables surgical management to be tailored specifically to each patient’s burden of disease.
In two clinical trials, 332 patients with melanoma or breast cancer were injected with Lymphoseek and blue dye. Surgeons subsequently removed suspected lymph nodes for pathologic examination. Confirmed lymph nodes were examined for their content of blue dye and Lymphoseek. The combination of Lymphoseek and blue dye localized most lymph nodes, although a notable number of nodes were localized only by Lymphoseek.
The most common side effects identified in clinical trials were pain and irritation at the injection site.
According to the manufacturer, a clinical trial involving patients with head and neck cancer is completing enrollment and is expected to be the subject of a future New Drug Application amendment. An initial Marketing Authorization Application filing in the European Union is anticipated by the end of 2012.
A radioactive diagnostic imaging agent called Lymphoseek (technetium Tc 99m tilmanocept) Injection has been approved for locating lymph nodes in patients who have breast cancer or melanoma and are undergoing surgery to remove tumor-draining lymph nodes, the Food and Drug Administration announced.
Lymphoseek is the first new drug for lymph-node mapping to be approved in more than 30 years. Other FDA-approved drugs used for lymph-node mapping include sulfur colloid and isosulfan blue.
"To use Lymphoseek, doctors inject the drug into the tumor area and later, using a handheld radiation detector, find lymph nodes that have taken up Lymphoseek’s radioactivity," said Dr. Shaw Chen, deputy director of the Office of Drug Evaluation IV in the FDA’s Center for Drug Evaluation and Research.
Lymphoseek is marketed by Navidea Biopharmaceuticals. The manufacturer’s website notes that the ability to rapidly locate and biopsy sentinel nodes enables surgical management to be tailored specifically to each patient’s burden of disease.
In two clinical trials, 332 patients with melanoma or breast cancer were injected with Lymphoseek and blue dye. Surgeons subsequently removed suspected lymph nodes for pathologic examination. Confirmed lymph nodes were examined for their content of blue dye and Lymphoseek. The combination of Lymphoseek and blue dye localized most lymph nodes, although a notable number of nodes were localized only by Lymphoseek.
The most common side effects identified in clinical trials were pain and irritation at the injection site.
According to the manufacturer, a clinical trial involving patients with head and neck cancer is completing enrollment and is expected to be the subject of a future New Drug Application amendment. An initial Marketing Authorization Application filing in the European Union is anticipated by the end of 2012.
A radioactive diagnostic imaging agent called Lymphoseek (technetium Tc 99m tilmanocept) Injection has been approved for locating lymph nodes in patients who have breast cancer or melanoma and are undergoing surgery to remove tumor-draining lymph nodes, the Food and Drug Administration announced.
Lymphoseek is the first new drug for lymph-node mapping to be approved in more than 30 years. Other FDA-approved drugs used for lymph-node mapping include sulfur colloid and isosulfan blue.
"To use Lymphoseek, doctors inject the drug into the tumor area and later, using a handheld radiation detector, find lymph nodes that have taken up Lymphoseek’s radioactivity," said Dr. Shaw Chen, deputy director of the Office of Drug Evaluation IV in the FDA’s Center for Drug Evaluation and Research.
Lymphoseek is marketed by Navidea Biopharmaceuticals. The manufacturer’s website notes that the ability to rapidly locate and biopsy sentinel nodes enables surgical management to be tailored specifically to each patient’s burden of disease.
In two clinical trials, 332 patients with melanoma or breast cancer were injected with Lymphoseek and blue dye. Surgeons subsequently removed suspected lymph nodes for pathologic examination. Confirmed lymph nodes were examined for their content of blue dye and Lymphoseek. The combination of Lymphoseek and blue dye localized most lymph nodes, although a notable number of nodes were localized only by Lymphoseek.
The most common side effects identified in clinical trials were pain and irritation at the injection site.
According to the manufacturer, a clinical trial involving patients with head and neck cancer is completing enrollment and is expected to be the subject of a future New Drug Application amendment. An initial Marketing Authorization Application filing in the European Union is anticipated by the end of 2012.
Gastric bypass shows best results for glycemic control
Gastric bypass leads to more significant and durable improvements in glycemic control than does sleeve gastrectomy or intensive medical therapy in moderately obese patients with type 2 diabetes, a prospective, randomized controlled trial has found.
Two years after the procedure, patients randomized to Roux-en-Y gastric bypass plus intensive medical therapy (IMT) had a mean HbA1c of 6.7 ± 1.2%, compared with 7.1 ± 0.8% for those who underwent sleeve gastrectomy and IMT, and 8.4 ± 2.3% for IMT alone, reported Dr Sangeeta R. Kashyap of the Cleveland Clinic and colleagues.
While both surgical procedures resulted in similar reductions in body weight, body mass index, and total body fat percentage at 24 months, gastric bypass resulted in the greatest absolute truncal fat reductions (–16% vs. –10%; P = .04), according to the findings, which were published online (Diabetes Care 2013 Feb. 25 [doi:10.2337/dc12-1596]).
The 2-year study enrolled a subset of 60 patients from the 1-year STAMPEDE trial, which evaluated the efficacy and safety of IMT alone – pharmacotherapy in conjunction with lifestyle interventions – or IMT combined with gastric bypass or sleeve gastrectomy. Of the 54 patients who completed the trial, the average age was 48.4 years, with a mean BMI of 36 kg/m2; most patients were taking at least three different diabetes medications.
The study extension examined the effects of the three treatment approaches on glucose control, pancreatic beta-cell function including insulin secretion and sensitivity, and body composition.
"Other long-term observational studies have documented greater relapse rates for glycemic control after gastric restrictive procedures such as sleeve gastrectomy, suggesting that surgical weight loss from enforced caloric restriction itself is insufficient to halt the disease," reported Dr Kashyap and colleagues. "Our results extend the findings from our initial 12-month report and suggest factors beyond weight loss that are specific to intestinal bypass patients help regulate glucose levels and restore pancreatic beta-cell function."
Gastric bypass was the only treatment to have any significant effects on pancreatic beta-cell function, with a median 5.8-fold (quartile 1: –7.00; quartile Q3: 11.29) increase in beta-cell function from baseline compared with only negligible increases for sleeve gastrectomy and IMT.
Researchers also observed a 2.7-fold (N = 9, 3.8 vs. 1.4; P < .001) increase in insulin sensitivity with gastric bypass among subjects not using insulin, compared with a 1.2-fold (N=10; 5.8 vs. 5.3) increase after sleeve gastrectomy, and no change in those randomized to IMT (2.6 vs. 2.4; P = NS).
"Bariatric surgery, particularly gastric bypass surgery, uniquely restores normal glucose tolerance and pancreatic [beta]-cell function, presumably by targeting the truncal fat that represents the core metabolic defect involved in diabetes pathogenesis," the researchers wrote.
Primary funding for the study came from Ethicon Endo-Surgery, with ancillary funding from the American Diabetes Association and the National Institutes of Health. The authors reported receiving research grants, consultations and honoraria from various companies and organizations, and one author received grants and honoraria from Ethicon Endo-Surgery as scientific advisory board member, consultant, and speaker.
Gastric bypass leads to more significant and durable improvements in glycemic control than does sleeve gastrectomy or intensive medical therapy in moderately obese patients with type 2 diabetes, a prospective, randomized controlled trial has found.
Two years after the procedure, patients randomized to Roux-en-Y gastric bypass plus intensive medical therapy (IMT) had a mean HbA1c of 6.7 ± 1.2%, compared with 7.1 ± 0.8% for those who underwent sleeve gastrectomy and IMT, and 8.4 ± 2.3% for IMT alone, reported Dr Sangeeta R. Kashyap of the Cleveland Clinic and colleagues.
While both surgical procedures resulted in similar reductions in body weight, body mass index, and total body fat percentage at 24 months, gastric bypass resulted in the greatest absolute truncal fat reductions (–16% vs. –10%; P = .04), according to the findings, which were published online (Diabetes Care 2013 Feb. 25 [doi:10.2337/dc12-1596]).
The 2-year study enrolled a subset of 60 patients from the 1-year STAMPEDE trial, which evaluated the efficacy and safety of IMT alone – pharmacotherapy in conjunction with lifestyle interventions – or IMT combined with gastric bypass or sleeve gastrectomy. Of the 54 patients who completed the trial, the average age was 48.4 years, with a mean BMI of 36 kg/m2; most patients were taking at least three different diabetes medications.
The study extension examined the effects of the three treatment approaches on glucose control, pancreatic beta-cell function including insulin secretion and sensitivity, and body composition.
"Other long-term observational studies have documented greater relapse rates for glycemic control after gastric restrictive procedures such as sleeve gastrectomy, suggesting that surgical weight loss from enforced caloric restriction itself is insufficient to halt the disease," reported Dr Kashyap and colleagues. "Our results extend the findings from our initial 12-month report and suggest factors beyond weight loss that are specific to intestinal bypass patients help regulate glucose levels and restore pancreatic beta-cell function."
Gastric bypass was the only treatment to have any significant effects on pancreatic beta-cell function, with a median 5.8-fold (quartile 1: –7.00; quartile Q3: 11.29) increase in beta-cell function from baseline compared with only negligible increases for sleeve gastrectomy and IMT.
Researchers also observed a 2.7-fold (N = 9, 3.8 vs. 1.4; P < .001) increase in insulin sensitivity with gastric bypass among subjects not using insulin, compared with a 1.2-fold (N=10; 5.8 vs. 5.3) increase after sleeve gastrectomy, and no change in those randomized to IMT (2.6 vs. 2.4; P = NS).
"Bariatric surgery, particularly gastric bypass surgery, uniquely restores normal glucose tolerance and pancreatic [beta]-cell function, presumably by targeting the truncal fat that represents the core metabolic defect involved in diabetes pathogenesis," the researchers wrote.
Primary funding for the study came from Ethicon Endo-Surgery, with ancillary funding from the American Diabetes Association and the National Institutes of Health. The authors reported receiving research grants, consultations and honoraria from various companies and organizations, and one author received grants and honoraria from Ethicon Endo-Surgery as scientific advisory board member, consultant, and speaker.
Gastric bypass leads to more significant and durable improvements in glycemic control than does sleeve gastrectomy or intensive medical therapy in moderately obese patients with type 2 diabetes, a prospective, randomized controlled trial has found.
Two years after the procedure, patients randomized to Roux-en-Y gastric bypass plus intensive medical therapy (IMT) had a mean HbA1c of 6.7 ± 1.2%, compared with 7.1 ± 0.8% for those who underwent sleeve gastrectomy and IMT, and 8.4 ± 2.3% for IMT alone, reported Dr Sangeeta R. Kashyap of the Cleveland Clinic and colleagues.
While both surgical procedures resulted in similar reductions in body weight, body mass index, and total body fat percentage at 24 months, gastric bypass resulted in the greatest absolute truncal fat reductions (–16% vs. –10%; P = .04), according to the findings, which were published online (Diabetes Care 2013 Feb. 25 [doi:10.2337/dc12-1596]).
The 2-year study enrolled a subset of 60 patients from the 1-year STAMPEDE trial, which evaluated the efficacy and safety of IMT alone – pharmacotherapy in conjunction with lifestyle interventions – or IMT combined with gastric bypass or sleeve gastrectomy. Of the 54 patients who completed the trial, the average age was 48.4 years, with a mean BMI of 36 kg/m2; most patients were taking at least three different diabetes medications.
The study extension examined the effects of the three treatment approaches on glucose control, pancreatic beta-cell function including insulin secretion and sensitivity, and body composition.
"Other long-term observational studies have documented greater relapse rates for glycemic control after gastric restrictive procedures such as sleeve gastrectomy, suggesting that surgical weight loss from enforced caloric restriction itself is insufficient to halt the disease," reported Dr Kashyap and colleagues. "Our results extend the findings from our initial 12-month report and suggest factors beyond weight loss that are specific to intestinal bypass patients help regulate glucose levels and restore pancreatic beta-cell function."
Gastric bypass was the only treatment to have any significant effects on pancreatic beta-cell function, with a median 5.8-fold (quartile 1: –7.00; quartile Q3: 11.29) increase in beta-cell function from baseline compared with only negligible increases for sleeve gastrectomy and IMT.
Researchers also observed a 2.7-fold (N = 9, 3.8 vs. 1.4; P < .001) increase in insulin sensitivity with gastric bypass among subjects not using insulin, compared with a 1.2-fold (N=10; 5.8 vs. 5.3) increase after sleeve gastrectomy, and no change in those randomized to IMT (2.6 vs. 2.4; P = NS).
"Bariatric surgery, particularly gastric bypass surgery, uniquely restores normal glucose tolerance and pancreatic [beta]-cell function, presumably by targeting the truncal fat that represents the core metabolic defect involved in diabetes pathogenesis," the researchers wrote.
Primary funding for the study came from Ethicon Endo-Surgery, with ancillary funding from the American Diabetes Association and the National Institutes of Health. The authors reported receiving research grants, consultations and honoraria from various companies and organizations, and one author received grants and honoraria from Ethicon Endo-Surgery as scientific advisory board member, consultant, and speaker.
Major finding: Gastric bypass beats sleeve gastrectomy and intensive medical therapy in type 2 diabetes management.
Data source: Prospective, randomized controlled trial in 60 moderately obese patients with type 2 diabetes.
Disclosures: Primary funding for the study came from Ethicon Endo-Surgery, with ancillary funding from the American Diabetes Association and the National Institutes of Health. The authors reported receiving research grants, consultations, and honoraria from various organizations, and one author received grants and honoraria from Ethicon Endo-Surgery as scientific advisory board member, consultant, and speaker.
No drop in employer-based health coverage in 2012
The percentage of Americans with employer-based health insurance was basically unchanged from 2011 to 2012, after dropping each of the previous 3 years, according to a survey published online Feb. 22 by Gallup.
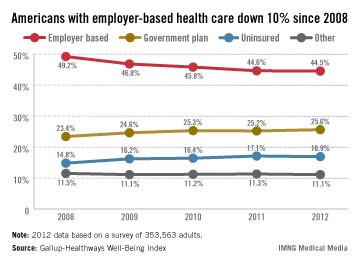
In 2012, 44.5% of the respondents reported getting coverage from their place of employment, compared with 44.6% in 2011. That figure had previously dropped from 49.2% in 2008, Gallup said.
Survey data from the Gallup-Healthways Well-Being Index show that an increasing percentage of people are covered by government-based plans: 25.6% in 2012, compared with 23.4% in 2008. The percentage of Americans who were uninsured dropped slightly – from 17.1% in 2011 to 16.9% in 2012 – but is still higher than in 2008. Those in the "other" category may be buying insurance themselves, Gallup noted.
The Well-Being Index results come from annual telephone interviews. For 2012, the random sample consisted of 353,563 adults in all 50 states and the District of Columbia.
The percentage of Americans with employer-based health insurance was basically unchanged from 2011 to 2012, after dropping each of the previous 3 years, according to a survey published online Feb. 22 by Gallup.
In 2012, 44.5% of the respondents reported getting coverage from their place of employment, compared with 44.6% in 2011. That figure had previously dropped from 49.2% in 2008, Gallup said.
Survey data from the Gallup-Healthways Well-Being Index show that an increasing percentage of people are covered by government-based plans: 25.6% in 2012, compared with 23.4% in 2008. The percentage of Americans who were uninsured dropped slightly – from 17.1% in 2011 to 16.9% in 2012 – but is still higher than in 2008. Those in the "other" category may be buying insurance themselves, Gallup noted.
The Well-Being Index results come from annual telephone interviews. For 2012, the random sample consisted of 353,563 adults in all 50 states and the District of Columbia.
The percentage of Americans with employer-based health insurance was basically unchanged from 2011 to 2012, after dropping each of the previous 3 years, according to a survey published online Feb. 22 by Gallup.
In 2012, 44.5% of the respondents reported getting coverage from their place of employment, compared with 44.6% in 2011. That figure had previously dropped from 49.2% in 2008, Gallup said.
Survey data from the Gallup-Healthways Well-Being Index show that an increasing percentage of people are covered by government-based plans: 25.6% in 2012, compared with 23.4% in 2008. The percentage of Americans who were uninsured dropped slightly – from 17.1% in 2011 to 16.9% in 2012 – but is still higher than in 2008. Those in the "other" category may be buying insurance themselves, Gallup noted.
The Well-Being Index results come from annual telephone interviews. For 2012, the random sample consisted of 353,563 adults in all 50 states and the District of Columbia.
Use of electronic cigarettes on the rise
More than twice as many adult smokers used electronic cigarettes in 2011 as in 2010, according to investigators from the Centers for Disease Control and Prevention.
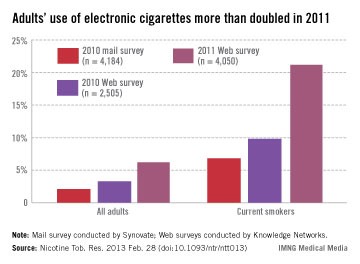
Approximately 21% of current smokers used electronic cigarettes at least once in 2011. The data come from Web-based surveys in 2010 and 2011 and one mail-based survey in 2010. For 2010, smokers’ use of e-cigarettes was 9.8% in the Web survey and 6.8% in the mail survey, said Brian A. King, Ph.D., and his associates at the CDC.
Among all adults, reported use of e-cigarettes was 6.2% in the 2011 survey, compared with 3.3% for the 2010 Web survey and 2.1% for the 2010 mail survey, they reported (Nicotine Tob. Res. 2013 Feb. 28 [doi:10.1093/ntr/ntt013]).
Among former smokers, e-cigarette use was 7.4% in the 2011 survey, 2.5% in the 2010 Web survey, and 0.6% in the 2010 mail survey. There was, however, "no significant difference in ever use of e-cigarettes ... by sex, age, race/ethnicity, education, income, or U.S. region" in any of the surveys, the investigators wrote.
Sample sizes were 4,050 for the 2011 Web survey, 2,505 for the 2010 Web survey, and 4,184 for the 2010 mail survey. Both Web-based surveys were conducted by Knowledge Networks; the mail-based survey was conducted by Synovate. Dr. King and his associates did not declare any financial conflicts of interest.
More than twice as many adult smokers used electronic cigarettes in 2011 as in 2010, according to investigators from the Centers for Disease Control and Prevention.
Approximately 21% of current smokers used electronic cigarettes at least once in 2011. The data come from Web-based surveys in 2010 and 2011 and one mail-based survey in 2010. For 2010, smokers’ use of e-cigarettes was 9.8% in the Web survey and 6.8% in the mail survey, said Brian A. King, Ph.D., and his associates at the CDC.
Among all adults, reported use of e-cigarettes was 6.2% in the 2011 survey, compared with 3.3% for the 2010 Web survey and 2.1% for the 2010 mail survey, they reported (Nicotine Tob. Res. 2013 Feb. 28 [doi:10.1093/ntr/ntt013]).
Among former smokers, e-cigarette use was 7.4% in the 2011 survey, 2.5% in the 2010 Web survey, and 0.6% in the 2010 mail survey. There was, however, "no significant difference in ever use of e-cigarettes ... by sex, age, race/ethnicity, education, income, or U.S. region" in any of the surveys, the investigators wrote.
Sample sizes were 4,050 for the 2011 Web survey, 2,505 for the 2010 Web survey, and 4,184 for the 2010 mail survey. Both Web-based surveys were conducted by Knowledge Networks; the mail-based survey was conducted by Synovate. Dr. King and his associates did not declare any financial conflicts of interest.
More than twice as many adult smokers used electronic cigarettes in 2011 as in 2010, according to investigators from the Centers for Disease Control and Prevention.
Approximately 21% of current smokers used electronic cigarettes at least once in 2011. The data come from Web-based surveys in 2010 and 2011 and one mail-based survey in 2010. For 2010, smokers’ use of e-cigarettes was 9.8% in the Web survey and 6.8% in the mail survey, said Brian A. King, Ph.D., and his associates at the CDC.
Among all adults, reported use of e-cigarettes was 6.2% in the 2011 survey, compared with 3.3% for the 2010 Web survey and 2.1% for the 2010 mail survey, they reported (Nicotine Tob. Res. 2013 Feb. 28 [doi:10.1093/ntr/ntt013]).
Among former smokers, e-cigarette use was 7.4% in the 2011 survey, 2.5% in the 2010 Web survey, and 0.6% in the 2010 mail survey. There was, however, "no significant difference in ever use of e-cigarettes ... by sex, age, race/ethnicity, education, income, or U.S. region" in any of the surveys, the investigators wrote.
Sample sizes were 4,050 for the 2011 Web survey, 2,505 for the 2010 Web survey, and 4,184 for the 2010 mail survey. Both Web-based surveys were conducted by Knowledge Networks; the mail-based survey was conducted by Synovate. Dr. King and his associates did not declare any financial conflicts of interest.
CMS audits EHR incentives – before paying them
NEW ORLEANS – Haven’t received your meaningful use incentive? Check your mail for an audit letter.
If in January you submitted an attestation of meaningful use of your electronic health record – with an eye to reaping the federal health IT incentive – an audit letter may be on its way to you.
A contractor for the Centers for Medicare and Medicaid Services began sending audit letters this week to randomly selected Medicare-eligible professionals and hospitals, Elizabeth Holland, a director of the HIT Initiatives Group in the agency’s Office of E-Health Standards and Services, said March 5 at the Healthcare Information and Management Systems Society annual conference. The audits could result in delays or ultimately, non-payment, she said.
"We have a fiduciary responsibility to make sure that we are paying appropriately," Ms. Holland explained, adding that providers who were not selected for the audit have already received their payments.
Audit letters are being sent by Figliozzi & Co. to Medicare-eligible hospitals and physicians. If recipients do not respond, "their payment will be held up until they respond and provide the documentation" to back up their attestation, Ms. Holland said. "If a certain amount of time goes by and they still don’t respond, they will not be getting a payment."
These prepayment audits follow on the heels of postpayment audits that the CMS began in July 2012. Under that program, Figliozzi & Co. audited Medicare-eligible professionals and states audited Medicaid-eligible professionals.
Ms. Holland said that more than 2,000 postpayment audits are underway; some are random and some are targeted. The data generated by the audits is, and will be, used to modify the agency’s approach to meaningful use. For instance, one goal is to see whether providers are appropriately reporting measures, she said.
CMS also has found that professionals do not have the proper documentation to support what they are attesting to. In the next month, the CMS will issue guidance on what documentation is needed, Ms. Holland said.
She presented data showing that so far, 161,890 eligible professionals – out of 527,200 who are eligible – have attested to meaningful use. Most of those (161,677) did so successfully. About 200 were not successful.
Of the 5,011 hospitals that are eligible, 2,653 have been successful. None failed.
On Twitter @aliciaault
NEW ORLEANS – Haven’t received your meaningful use incentive? Check your mail for an audit letter.
If in January you submitted an attestation of meaningful use of your electronic health record – with an eye to reaping the federal health IT incentive – an audit letter may be on its way to you.
A contractor for the Centers for Medicare and Medicaid Services began sending audit letters this week to randomly selected Medicare-eligible professionals and hospitals, Elizabeth Holland, a director of the HIT Initiatives Group in the agency’s Office of E-Health Standards and Services, said March 5 at the Healthcare Information and Management Systems Society annual conference. The audits could result in delays or ultimately, non-payment, she said.
"We have a fiduciary responsibility to make sure that we are paying appropriately," Ms. Holland explained, adding that providers who were not selected for the audit have already received their payments.
Audit letters are being sent by Figliozzi & Co. to Medicare-eligible hospitals and physicians. If recipients do not respond, "their payment will be held up until they respond and provide the documentation" to back up their attestation, Ms. Holland said. "If a certain amount of time goes by and they still don’t respond, they will not be getting a payment."
These prepayment audits follow on the heels of postpayment audits that the CMS began in July 2012. Under that program, Figliozzi & Co. audited Medicare-eligible professionals and states audited Medicaid-eligible professionals.
Ms. Holland said that more than 2,000 postpayment audits are underway; some are random and some are targeted. The data generated by the audits is, and will be, used to modify the agency’s approach to meaningful use. For instance, one goal is to see whether providers are appropriately reporting measures, she said.
CMS also has found that professionals do not have the proper documentation to support what they are attesting to. In the next month, the CMS will issue guidance on what documentation is needed, Ms. Holland said.
She presented data showing that so far, 161,890 eligible professionals – out of 527,200 who are eligible – have attested to meaningful use. Most of those (161,677) did so successfully. About 200 were not successful.
Of the 5,011 hospitals that are eligible, 2,653 have been successful. None failed.
On Twitter @aliciaault
NEW ORLEANS – Haven’t received your meaningful use incentive? Check your mail for an audit letter.
If in January you submitted an attestation of meaningful use of your electronic health record – with an eye to reaping the federal health IT incentive – an audit letter may be on its way to you.
A contractor for the Centers for Medicare and Medicaid Services began sending audit letters this week to randomly selected Medicare-eligible professionals and hospitals, Elizabeth Holland, a director of the HIT Initiatives Group in the agency’s Office of E-Health Standards and Services, said March 5 at the Healthcare Information and Management Systems Society annual conference. The audits could result in delays or ultimately, non-payment, she said.
"We have a fiduciary responsibility to make sure that we are paying appropriately," Ms. Holland explained, adding that providers who were not selected for the audit have already received their payments.
Audit letters are being sent by Figliozzi & Co. to Medicare-eligible hospitals and physicians. If recipients do not respond, "their payment will be held up until they respond and provide the documentation" to back up their attestation, Ms. Holland said. "If a certain amount of time goes by and they still don’t respond, they will not be getting a payment."
These prepayment audits follow on the heels of postpayment audits that the CMS began in July 2012. Under that program, Figliozzi & Co. audited Medicare-eligible professionals and states audited Medicaid-eligible professionals.
Ms. Holland said that more than 2,000 postpayment audits are underway; some are random and some are targeted. The data generated by the audits is, and will be, used to modify the agency’s approach to meaningful use. For instance, one goal is to see whether providers are appropriately reporting measures, she said.
CMS also has found that professionals do not have the proper documentation to support what they are attesting to. In the next month, the CMS will issue guidance on what documentation is needed, Ms. Holland said.
She presented data showing that so far, 161,890 eligible professionals – out of 527,200 who are eligible – have attested to meaningful use. Most of those (161,677) did so successfully. About 200 were not successful.
Of the 5,011 hospitals that are eligible, 2,653 have been successful. None failed.
On Twitter @aliciaault
AT THE HIMSS13 ANNUAL CONFERENCE
'Nightmare' CRE infections on the rise, CDC says
Between 2001 and 2011, the percentage of carbapenem-resistant Enterobacteriaceae infections reported by acute-care hospitals in the United States increased nearly fourfold, from 1.2% to 4.2%. More recent data from the first 6 months of 2102 suggest that the percentage of such infections is now slightly higher, at 4.6%.
The findings, which appear in a Vital Signs report released by the Centers for Disease Control and Prevention on March 5, are significant because CRE can kill up to 50% of patients who get bloodstream infections from them.
"It’s not often that our scientists come to me to say that we have a very serious problem and we need to sound an alarm," CDC Director Tom Frieden said during a related telephone press briefing. "But that’s exactly what we’re doing today. This Vital Signs is an early warning about a health care–associated infection that’s happening in hospitals and other inpatient medical facilities. The good news is that we now have an opportunity to prevent its further spread. The sooner we act, the less likely it will get out into the community."
For one component of the report, CDC researchers analyzed data from the National Healthcare Safety Network (NHSN) and its predecessor, the National Nosocomial Infections Surveillance system (NNIS), for the number of Enterobacteriaceae isolates; the percentage reported to be tested against carbapenems; and the percentage reported as carbapenem resistant in 2001 and in 2011. For another component, the researchers evaluated NHSN data for the number and percentage of facilities reporting CRE from a catheter-associated urinary tract infection or central line-associated bloodstream infection between January and June of 2012.
Of the CRE cases reported during the first half of 2012, about 18% occurred in long-term acute care hospitals and about 4% occurred in short-stay hospitals.
Dr. Frieden characterized CRE as a "nightmare bacteria" that poses a triple threat. "First, they’re resistant to all or nearly all antibiotics – even some of our last-resort drugs," he explained. "Second, they have high mortality rates. They kill up to half of people who get serious infections with them. And third, they can spread their resistance to other bacteria such as Escherichia coli and make E. coli resistant to those antibiotics also."
The risk of CRE infection is highest among patients who are receiving complex or long-term medical care, including those in short-stay hospitals or long-term acute care hospitals, or nursing homes. It’s commonly spread by people with unclean hands but "medical devices such as ventilators or catheters [also] increase the risk of life-threatening infection because they allow new bacteria to get deeply into a patient’s body," Dr. Frieden said.
According to the report, health care facilities in Northeastern states report the most cases of CRE, with 42 states reporting having had at least one patient test positive for the infection. In addition, one type of CRE, a resistant form of Klebsiella pneumoniae, demonstrated a nearly sevenfold increase between 2001 and 2011, jumping from 1.6% to 10.4%.
"That’s a very troubling increase," Dr. Frieden said. "In some of those places, these bacteria are now a routine challenge for patients and clinicians."
The good news, he continued, "is that we still have time to stop CRE. Many facilities can act now to prevent CRE from emerging or, if it has emerged, to control it. We need health care leaders, clinicians, and health care departments to act to prevent CRE, so it doesn’t become widespread and spread to the community." He listed six practical ways that health care providers can prevent CRE in their facilities:
• Know if your particular patient has CRE and request immediate alerts from your laboratory every time it identifies any patient with the infection.
• When receiving or transporting patients, make sure to ask or find out if the patient you’re receiving has CRE.
• Protect your patients from CRE by following contact and other precautions whenever you’re treating patients with CRE "so you don’t inadvertently spread their organism to someone else."
• Whenever possible, have specific rooms, equipment, and staff to care for CRE patients. "This reduces the chance that CRE will spread from one patient to others," he said.
• Remove temporary medical devices such as catheters as soon as possible.
• Prescribe antibiotics carefully. "Unfortunately, half of the antibiotics prescribed in this country are either unnecessary or inappropriate," Dr. Frieden said. "Overuse and misuse increases drug-resistant infections. That results in longer inpatient treatment, higher costs, and poorer patient outcomes."
These and other recommendations for hospitals, long-term acute care facilities, nursing homes, and health departments can be found in a CRE prevention toolkit released by the CDC in 2012. "We’re gratified to see that places that have implemented these tools have seen dramatic reductions in their CRE rates," Dr. Frieden said.
Authors of the report acknowledged at least three limitations of the data. First, they wrote, "antimicrobial susceptibility data reported to NNIS and NHSN were generated at individual institutions rather than [at] a central laboratory, and testing methodologies vary between facilities. Second, susceptibility interpretation is based on the recommended break points used when tested. Although carbapenem break points for Enterobacteriaceae were lowered in 2010 and might have influenced the increase in the percentage of isolates that were carbapenem-resistant, most laboratories would not have incorporated those changes by 2011. Finally, in some instances, complete susceptibility test results, particularly for carbapenems, were not reported to NNIS or NHSN, leading to a subset of isolates that were not included in these analyses."
The researchers reported having no relevant financial disclosures.
Between 2001 and 2011, the percentage of carbapenem-resistant Enterobacteriaceae infections reported by acute-care hospitals in the United States increased nearly fourfold, from 1.2% to 4.2%. More recent data from the first 6 months of 2102 suggest that the percentage of such infections is now slightly higher, at 4.6%.
The findings, which appear in a Vital Signs report released by the Centers for Disease Control and Prevention on March 5, are significant because CRE can kill up to 50% of patients who get bloodstream infections from them.
"It’s not often that our scientists come to me to say that we have a very serious problem and we need to sound an alarm," CDC Director Tom Frieden said during a related telephone press briefing. "But that’s exactly what we’re doing today. This Vital Signs is an early warning about a health care–associated infection that’s happening in hospitals and other inpatient medical facilities. The good news is that we now have an opportunity to prevent its further spread. The sooner we act, the less likely it will get out into the community."
For one component of the report, CDC researchers analyzed data from the National Healthcare Safety Network (NHSN) and its predecessor, the National Nosocomial Infections Surveillance system (NNIS), for the number of Enterobacteriaceae isolates; the percentage reported to be tested against carbapenems; and the percentage reported as carbapenem resistant in 2001 and in 2011. For another component, the researchers evaluated NHSN data for the number and percentage of facilities reporting CRE from a catheter-associated urinary tract infection or central line-associated bloodstream infection between January and June of 2012.
Of the CRE cases reported during the first half of 2012, about 18% occurred in long-term acute care hospitals and about 4% occurred in short-stay hospitals.
Dr. Frieden characterized CRE as a "nightmare bacteria" that poses a triple threat. "First, they’re resistant to all or nearly all antibiotics – even some of our last-resort drugs," he explained. "Second, they have high mortality rates. They kill up to half of people who get serious infections with them. And third, they can spread their resistance to other bacteria such as Escherichia coli and make E. coli resistant to those antibiotics also."
The risk of CRE infection is highest among patients who are receiving complex or long-term medical care, including those in short-stay hospitals or long-term acute care hospitals, or nursing homes. It’s commonly spread by people with unclean hands but "medical devices such as ventilators or catheters [also] increase the risk of life-threatening infection because they allow new bacteria to get deeply into a patient’s body," Dr. Frieden said.
According to the report, health care facilities in Northeastern states report the most cases of CRE, with 42 states reporting having had at least one patient test positive for the infection. In addition, one type of CRE, a resistant form of Klebsiella pneumoniae, demonstrated a nearly sevenfold increase between 2001 and 2011, jumping from 1.6% to 10.4%.
"That’s a very troubling increase," Dr. Frieden said. "In some of those places, these bacteria are now a routine challenge for patients and clinicians."
The good news, he continued, "is that we still have time to stop CRE. Many facilities can act now to prevent CRE from emerging or, if it has emerged, to control it. We need health care leaders, clinicians, and health care departments to act to prevent CRE, so it doesn’t become widespread and spread to the community." He listed six practical ways that health care providers can prevent CRE in their facilities:
• Know if your particular patient has CRE and request immediate alerts from your laboratory every time it identifies any patient with the infection.
• When receiving or transporting patients, make sure to ask or find out if the patient you’re receiving has CRE.
• Protect your patients from CRE by following contact and other precautions whenever you’re treating patients with CRE "so you don’t inadvertently spread their organism to someone else."
• Whenever possible, have specific rooms, equipment, and staff to care for CRE patients. "This reduces the chance that CRE will spread from one patient to others," he said.
• Remove temporary medical devices such as catheters as soon as possible.
• Prescribe antibiotics carefully. "Unfortunately, half of the antibiotics prescribed in this country are either unnecessary or inappropriate," Dr. Frieden said. "Overuse and misuse increases drug-resistant infections. That results in longer inpatient treatment, higher costs, and poorer patient outcomes."
These and other recommendations for hospitals, long-term acute care facilities, nursing homes, and health departments can be found in a CRE prevention toolkit released by the CDC in 2012. "We’re gratified to see that places that have implemented these tools have seen dramatic reductions in their CRE rates," Dr. Frieden said.
Authors of the report acknowledged at least three limitations of the data. First, they wrote, "antimicrobial susceptibility data reported to NNIS and NHSN were generated at individual institutions rather than [at] a central laboratory, and testing methodologies vary between facilities. Second, susceptibility interpretation is based on the recommended break points used when tested. Although carbapenem break points for Enterobacteriaceae were lowered in 2010 and might have influenced the increase in the percentage of isolates that were carbapenem-resistant, most laboratories would not have incorporated those changes by 2011. Finally, in some instances, complete susceptibility test results, particularly for carbapenems, were not reported to NNIS or NHSN, leading to a subset of isolates that were not included in these analyses."
The researchers reported having no relevant financial disclosures.
Between 2001 and 2011, the percentage of carbapenem-resistant Enterobacteriaceae infections reported by acute-care hospitals in the United States increased nearly fourfold, from 1.2% to 4.2%. More recent data from the first 6 months of 2102 suggest that the percentage of such infections is now slightly higher, at 4.6%.
The findings, which appear in a Vital Signs report released by the Centers for Disease Control and Prevention on March 5, are significant because CRE can kill up to 50% of patients who get bloodstream infections from them.
"It’s not often that our scientists come to me to say that we have a very serious problem and we need to sound an alarm," CDC Director Tom Frieden said during a related telephone press briefing. "But that’s exactly what we’re doing today. This Vital Signs is an early warning about a health care–associated infection that’s happening in hospitals and other inpatient medical facilities. The good news is that we now have an opportunity to prevent its further spread. The sooner we act, the less likely it will get out into the community."
For one component of the report, CDC researchers analyzed data from the National Healthcare Safety Network (NHSN) and its predecessor, the National Nosocomial Infections Surveillance system (NNIS), for the number of Enterobacteriaceae isolates; the percentage reported to be tested against carbapenems; and the percentage reported as carbapenem resistant in 2001 and in 2011. For another component, the researchers evaluated NHSN data for the number and percentage of facilities reporting CRE from a catheter-associated urinary tract infection or central line-associated bloodstream infection between January and June of 2012.
Of the CRE cases reported during the first half of 2012, about 18% occurred in long-term acute care hospitals and about 4% occurred in short-stay hospitals.
Dr. Frieden characterized CRE as a "nightmare bacteria" that poses a triple threat. "First, they’re resistant to all or nearly all antibiotics – even some of our last-resort drugs," he explained. "Second, they have high mortality rates. They kill up to half of people who get serious infections with them. And third, they can spread their resistance to other bacteria such as Escherichia coli and make E. coli resistant to those antibiotics also."
The risk of CRE infection is highest among patients who are receiving complex or long-term medical care, including those in short-stay hospitals or long-term acute care hospitals, or nursing homes. It’s commonly spread by people with unclean hands but "medical devices such as ventilators or catheters [also] increase the risk of life-threatening infection because they allow new bacteria to get deeply into a patient’s body," Dr. Frieden said.
According to the report, health care facilities in Northeastern states report the most cases of CRE, with 42 states reporting having had at least one patient test positive for the infection. In addition, one type of CRE, a resistant form of Klebsiella pneumoniae, demonstrated a nearly sevenfold increase between 2001 and 2011, jumping from 1.6% to 10.4%.
"That’s a very troubling increase," Dr. Frieden said. "In some of those places, these bacteria are now a routine challenge for patients and clinicians."
The good news, he continued, "is that we still have time to stop CRE. Many facilities can act now to prevent CRE from emerging or, if it has emerged, to control it. We need health care leaders, clinicians, and health care departments to act to prevent CRE, so it doesn’t become widespread and spread to the community." He listed six practical ways that health care providers can prevent CRE in their facilities:
• Know if your particular patient has CRE and request immediate alerts from your laboratory every time it identifies any patient with the infection.
• When receiving or transporting patients, make sure to ask or find out if the patient you’re receiving has CRE.
• Protect your patients from CRE by following contact and other precautions whenever you’re treating patients with CRE "so you don’t inadvertently spread their organism to someone else."
• Whenever possible, have specific rooms, equipment, and staff to care for CRE patients. "This reduces the chance that CRE will spread from one patient to others," he said.
• Remove temporary medical devices such as catheters as soon as possible.
• Prescribe antibiotics carefully. "Unfortunately, half of the antibiotics prescribed in this country are either unnecessary or inappropriate," Dr. Frieden said. "Overuse and misuse increases drug-resistant infections. That results in longer inpatient treatment, higher costs, and poorer patient outcomes."
These and other recommendations for hospitals, long-term acute care facilities, nursing homes, and health departments can be found in a CRE prevention toolkit released by the CDC in 2012. "We’re gratified to see that places that have implemented these tools have seen dramatic reductions in their CRE rates," Dr. Frieden said.
Authors of the report acknowledged at least three limitations of the data. First, they wrote, "antimicrobial susceptibility data reported to NNIS and NHSN were generated at individual institutions rather than [at] a central laboratory, and testing methodologies vary between facilities. Second, susceptibility interpretation is based on the recommended break points used when tested. Although carbapenem break points for Enterobacteriaceae were lowered in 2010 and might have influenced the increase in the percentage of isolates that were carbapenem-resistant, most laboratories would not have incorporated those changes by 2011. Finally, in some instances, complete susceptibility test results, particularly for carbapenems, were not reported to NNIS or NHSN, leading to a subset of isolates that were not included in these analyses."
The researchers reported having no relevant financial disclosures.
FROM CDC VITAL SIGNS
Major Finding: The percentage of carbapenem-resistant Enterobacteriaceae infections reported by acute-care hospitals in the United States increased from 1.2% in 2001 to 4.2% in 2011.
Data Source: CDC analysis of data from the National Healthcare Safety Network (NHSN) and its predecessor, the National Nosocomial Infections Surveillance system (NNIS).
Disclosures: The researchers reported having no relevant financial conflicts.
Surgical quality improvement: A call for federal support
The Patient Protection and Affordable Care Act defines an Accountable Care Organization (ACO) as a group of doctors, hospitals, or other health care providers that organize to "coordinate care to ensure that patients, especially the chronically ill, get the right care at the right time while avoiding unnecessary duplication of services and preventing medical errors". While this effort is getting a lot of national play, and funding through a ""shared savings" mechanism, it is curious to me that the discipline of surgery is left out of the mix-completely.
Because of the enormous size of the surgical undertaking in this country – more than 40 million surgical procedures are performed each year – I argue that the redesign of surgical care processes, with resulting improvement in quality and reduced cost, could match whatever savings come from a focus on the chronically ill. Surgeons need to lobby for a structural and funding mechanism similar to the ACO to support our quality improvement efforts.
In order to generate federal funding for surgical quality improvement, we need an established model – a population based model-that helps us to define best surgical practices, to improve coordination of care between the inpatient and outpatient settings, and between surgical specialists and our primary care colleagues. By analogy to the current ACO initiative, such a model would create great interest at the level of the Centers for Medicare & Medicaid Services.
In Michigan, we have developed a population-based model that effectively addresses these issues. The Michigan Surgical Quality Collaborative (MSQC) includes 52 primarily community based hospitals interested in improving quality. This organization is funded entirely by the dominant third party payer in the state, Blue Cross Blue Shield of Michigan (BCBSM). We identify best performers in a variety of areas, make site visits to the best performers in order to identify best practices, and then disseminate the resulting information to the rest of the collaborative. This is done via a website or quarterly in-person meetings. Last year, BCBSM announced that they had saved $89 million in avoidable costs by virtue of the improved results seen within MSQC over a 3-year period, a remarkable return on investment. The high level objectives of the ACO approach were met, quality improved, and costs went down, substantially, but in surgical care, not primary care.
Several surgical quality collaborative groups now exist similar to MSQC, such as the Surgical Care and Outcomes Assessment Program, the Vascular Study Group of New England, Northern New England Cardiovascular Consortium, Michigan Bariatric Surgery Collaborative, Michigan Urological Surgical Improvement Collaborative, and many more that are developing. These groups provide a rich source of information about how to actually improve quality, information that is not available from any other mechanism that I know of, but financial support for these efforts is shaky at best. The burden of financial support is usually at the level of the individual hospital, but as we all know, times are tough in hospitals, and long-term participation is not guaranteed. Support by the third-party payer, such as BCBSM in Michigan, has been very successful, but whether this approach would work broadly across the nation is not yet known.
If one accepts that we have a population-based model that works, what mechanism could we suggest for which CMS would provide ongoing financial support? One suggestion is that CMS could offer financial incentives to urge the existing ACOs to participate in a regional improvement collaborative. This would represent an expansion of the current concept, but would perhaps be easier, since it would build on an existing structure. Another suggestion is that CMS consider the regional quality improvement groups as ACOs in and of themselves, with savings from the shared savings model flowing to the group’s administrative center, and subsequent distribution to individual hospitals.
Whatever the mechanism, the main point is that surgeons, via our emerging population-based models of collaboration, have enormous, and seemingly unrecognized potential to "bend the cost curve." There is now no federal strategy that would support these efforts, but one is much needed. The American College of Surgeons should use its lobbying influence to argue this point.
Dr. Campbell is the Henry King Ransom Professor of Surgery, University of Michigan Health System, Ann Arbor.
The Patient Protection and Affordable Care Act defines an Accountable Care Organization (ACO) as a group of doctors, hospitals, or other health care providers that organize to "coordinate care to ensure that patients, especially the chronically ill, get the right care at the right time while avoiding unnecessary duplication of services and preventing medical errors". While this effort is getting a lot of national play, and funding through a ""shared savings" mechanism, it is curious to me that the discipline of surgery is left out of the mix-completely.
Because of the enormous size of the surgical undertaking in this country – more than 40 million surgical procedures are performed each year – I argue that the redesign of surgical care processes, with resulting improvement in quality and reduced cost, could match whatever savings come from a focus on the chronically ill. Surgeons need to lobby for a structural and funding mechanism similar to the ACO to support our quality improvement efforts.
In order to generate federal funding for surgical quality improvement, we need an established model – a population based model-that helps us to define best surgical practices, to improve coordination of care between the inpatient and outpatient settings, and between surgical specialists and our primary care colleagues. By analogy to the current ACO initiative, such a model would create great interest at the level of the Centers for Medicare & Medicaid Services.
In Michigan, we have developed a population-based model that effectively addresses these issues. The Michigan Surgical Quality Collaborative (MSQC) includes 52 primarily community based hospitals interested in improving quality. This organization is funded entirely by the dominant third party payer in the state, Blue Cross Blue Shield of Michigan (BCBSM). We identify best performers in a variety of areas, make site visits to the best performers in order to identify best practices, and then disseminate the resulting information to the rest of the collaborative. This is done via a website or quarterly in-person meetings. Last year, BCBSM announced that they had saved $89 million in avoidable costs by virtue of the improved results seen within MSQC over a 3-year period, a remarkable return on investment. The high level objectives of the ACO approach were met, quality improved, and costs went down, substantially, but in surgical care, not primary care.
Several surgical quality collaborative groups now exist similar to MSQC, such as the Surgical Care and Outcomes Assessment Program, the Vascular Study Group of New England, Northern New England Cardiovascular Consortium, Michigan Bariatric Surgery Collaborative, Michigan Urological Surgical Improvement Collaborative, and many more that are developing. These groups provide a rich source of information about how to actually improve quality, information that is not available from any other mechanism that I know of, but financial support for these efforts is shaky at best. The burden of financial support is usually at the level of the individual hospital, but as we all know, times are tough in hospitals, and long-term participation is not guaranteed. Support by the third-party payer, such as BCBSM in Michigan, has been very successful, but whether this approach would work broadly across the nation is not yet known.
If one accepts that we have a population-based model that works, what mechanism could we suggest for which CMS would provide ongoing financial support? One suggestion is that CMS could offer financial incentives to urge the existing ACOs to participate in a regional improvement collaborative. This would represent an expansion of the current concept, but would perhaps be easier, since it would build on an existing structure. Another suggestion is that CMS consider the regional quality improvement groups as ACOs in and of themselves, with savings from the shared savings model flowing to the group’s administrative center, and subsequent distribution to individual hospitals.
Whatever the mechanism, the main point is that surgeons, via our emerging population-based models of collaboration, have enormous, and seemingly unrecognized potential to "bend the cost curve." There is now no federal strategy that would support these efforts, but one is much needed. The American College of Surgeons should use its lobbying influence to argue this point.
Dr. Campbell is the Henry King Ransom Professor of Surgery, University of Michigan Health System, Ann Arbor.
The Patient Protection and Affordable Care Act defines an Accountable Care Organization (ACO) as a group of doctors, hospitals, or other health care providers that organize to "coordinate care to ensure that patients, especially the chronically ill, get the right care at the right time while avoiding unnecessary duplication of services and preventing medical errors". While this effort is getting a lot of national play, and funding through a ""shared savings" mechanism, it is curious to me that the discipline of surgery is left out of the mix-completely.
Because of the enormous size of the surgical undertaking in this country – more than 40 million surgical procedures are performed each year – I argue that the redesign of surgical care processes, with resulting improvement in quality and reduced cost, could match whatever savings come from a focus on the chronically ill. Surgeons need to lobby for a structural and funding mechanism similar to the ACO to support our quality improvement efforts.
In order to generate federal funding for surgical quality improvement, we need an established model – a population based model-that helps us to define best surgical practices, to improve coordination of care between the inpatient and outpatient settings, and between surgical specialists and our primary care colleagues. By analogy to the current ACO initiative, such a model would create great interest at the level of the Centers for Medicare & Medicaid Services.
In Michigan, we have developed a population-based model that effectively addresses these issues. The Michigan Surgical Quality Collaborative (MSQC) includes 52 primarily community based hospitals interested in improving quality. This organization is funded entirely by the dominant third party payer in the state, Blue Cross Blue Shield of Michigan (BCBSM). We identify best performers in a variety of areas, make site visits to the best performers in order to identify best practices, and then disseminate the resulting information to the rest of the collaborative. This is done via a website or quarterly in-person meetings. Last year, BCBSM announced that they had saved $89 million in avoidable costs by virtue of the improved results seen within MSQC over a 3-year period, a remarkable return on investment. The high level objectives of the ACO approach were met, quality improved, and costs went down, substantially, but in surgical care, not primary care.
Several surgical quality collaborative groups now exist similar to MSQC, such as the Surgical Care and Outcomes Assessment Program, the Vascular Study Group of New England, Northern New England Cardiovascular Consortium, Michigan Bariatric Surgery Collaborative, Michigan Urological Surgical Improvement Collaborative, and many more that are developing. These groups provide a rich source of information about how to actually improve quality, information that is not available from any other mechanism that I know of, but financial support for these efforts is shaky at best. The burden of financial support is usually at the level of the individual hospital, but as we all know, times are tough in hospitals, and long-term participation is not guaranteed. Support by the third-party payer, such as BCBSM in Michigan, has been very successful, but whether this approach would work broadly across the nation is not yet known.
If one accepts that we have a population-based model that works, what mechanism could we suggest for which CMS would provide ongoing financial support? One suggestion is that CMS could offer financial incentives to urge the existing ACOs to participate in a regional improvement collaborative. This would represent an expansion of the current concept, but would perhaps be easier, since it would build on an existing structure. Another suggestion is that CMS consider the regional quality improvement groups as ACOs in and of themselves, with savings from the shared savings model flowing to the group’s administrative center, and subsequent distribution to individual hospitals.
Whatever the mechanism, the main point is that surgeons, via our emerging population-based models of collaboration, have enormous, and seemingly unrecognized potential to "bend the cost curve." There is now no federal strategy that would support these efforts, but one is much needed. The American College of Surgeons should use its lobbying influence to argue this point.
Dr. Campbell is the Henry King Ransom Professor of Surgery, University of Michigan Health System, Ann Arbor.
ACS takes steps to lead the profession through the next 100 years
As we commemorate the American College of Surgeons’ Centennial, the leadership and staff continue to develop and engage in initiatives designed to ensure that members of the College are best positioned to continuing providing quality surgical care for the next 100 years.
With respect to advocacy and health policy, the College remains at the forefront of efforts to repeal and replace the flawed sustainable growth rate formula used to calculate Medicare physician payment. Importantly, the College has been working on a proposal to replace the SGR with a value-based update, which emphasizes that value = quality/cost.
The ACS is also looking at alternative methods of resolving medical liability claims. We hosted a Medical Liability Summit in October, and articles based on the presentations given at the meeting will be published in a special issue of the Bulletin this March.
Advocacy
In addition, the College has successfully nominated Fellows to serve on panels that are advising the government on quality of care issues. Last year, ACS nominee Stephen B. Edge, MD, FACS, was appointed co-chair of the National Quality Forum’s Cancer Maintenance Endorsement Project, and Frank Opelka, MD, FACS, Associate Medical Director of the Division of Advocacy and Health Policy, was appointed chair of the Physician Consortium for Performance Improvement.
Quality improvement
The College’s National Surgical Quality Improvement Program (ACS NSQIP®) continues to grow in size and stature. More than 500 hospitals now participate in ACS NSQIP, and data promulgated through this outcome measurement system were key in the completion of a project that the College conducted last year with the Joint Commission Center for Transforming Healthcare aimed at reducing colorectal surgical site infections. A total of 135 SSIs were averted at seven health care institutions that participated in the demonstration project, saving those facilities more $3.7 million. The Centers for Disease Control and ACS NSQIP also have entered into a formal agreement to study SSI. Additionally, approximately 30% of ACS NSQIP hospitals are participating in the Centers for Medicare & Medicaid Services public reporting pilot program, Hospital Compare.
The Rapid Quality Reporting System has enabled Commission on Cancer–accredited programs to better adhere to five NQF-endorsed quality performance measures. Recognizing the potential value of this program, CMS recently contracted with the ACS to implement RQRS at 11 cancer centers that support public reporting through Hospital Compare.
At press time, ACS leaders had completed visits to 12 cities to discuss how quality-improvement efforts enhance surgical patient safety and reduce costs as part of the College’s Inspiring Quality initiative. With the assistance of Weber Shandwick, we also released the video Driven, which explores how surgical leaders and other innovators over the past 100 years have inspired quality by advancing surgical care.
Education
The College continues to establish benchmarks and standards in surgical education. This past year, new emphasis was placed on helping surgeons and residents to navigate transitions in their careers. Perhaps the most significant example of our efforts to assist residents as they transition into practice is the development of fellowships for young surgeons who are leaving training and entering into general surgery practice. The College will begin pilot testing the ACS Transition to Practice Fellowship in General Surgery on July 1 at five institutions in regions of the United States that currently are underserved by general surgeons. Furthermore, the ACS is collaborating with the Association of Program Directors in Surgery and the Association for Surgical Education to develop a new Entering Surgery Resident Prep Curriculum.
Workforce issues
Rural areas feel the effects of general surgeon shortage most acutely. To ensure that the College is better able to address the challenges that rural surgical practice poses, the College established a new Advisory Council for Rural Surgery.
And, to help surgeons make smart decisions as the trend toward hospital employment for physicians continues to grow, the ACS developed a new resource titled Surgeons as Institutional Employees: A Strategic Look at the Dimensions of Surgeons as Employees of Hospitals. This document mailed with the February 2013 Bulletin.
Communications
The College continues to modernize its communications vehicles. In September 2012, the College released the first issue of the newly redesigned Bulletin and is now in the process of working with consultants to reconstruct the public website (www.facs.org) and to integrate the members’ portal into the main site. To increase the visibility of the College’s advocacy efforts, the ACS last summer launched a monthly e-newsletter, The ACS Advocate.
Looking forward
The ACS is continuing to develop proactive solutions to the challenges facing surgeons. Your continued input on how we can address your concerns is always welcome.
Dr. Hoyt is Executive Director of the American College of Surgeons.
As we commemorate the American College of Surgeons’ Centennial, the leadership and staff continue to develop and engage in initiatives designed to ensure that members of the College are best positioned to continuing providing quality surgical care for the next 100 years.
With respect to advocacy and health policy, the College remains at the forefront of efforts to repeal and replace the flawed sustainable growth rate formula used to calculate Medicare physician payment. Importantly, the College has been working on a proposal to replace the SGR with a value-based update, which emphasizes that value = quality/cost.
The ACS is also looking at alternative methods of resolving medical liability claims. We hosted a Medical Liability Summit in October, and articles based on the presentations given at the meeting will be published in a special issue of the Bulletin this March.
Advocacy
In addition, the College has successfully nominated Fellows to serve on panels that are advising the government on quality of care issues. Last year, ACS nominee Stephen B. Edge, MD, FACS, was appointed co-chair of the National Quality Forum’s Cancer Maintenance Endorsement Project, and Frank Opelka, MD, FACS, Associate Medical Director of the Division of Advocacy and Health Policy, was appointed chair of the Physician Consortium for Performance Improvement.
Quality improvement
The College’s National Surgical Quality Improvement Program (ACS NSQIP®) continues to grow in size and stature. More than 500 hospitals now participate in ACS NSQIP, and data promulgated through this outcome measurement system were key in the completion of a project that the College conducted last year with the Joint Commission Center for Transforming Healthcare aimed at reducing colorectal surgical site infections. A total of 135 SSIs were averted at seven health care institutions that participated in the demonstration project, saving those facilities more $3.7 million. The Centers for Disease Control and ACS NSQIP also have entered into a formal agreement to study SSI. Additionally, approximately 30% of ACS NSQIP hospitals are participating in the Centers for Medicare & Medicaid Services public reporting pilot program, Hospital Compare.
The Rapid Quality Reporting System has enabled Commission on Cancer–accredited programs to better adhere to five NQF-endorsed quality performance measures. Recognizing the potential value of this program, CMS recently contracted with the ACS to implement RQRS at 11 cancer centers that support public reporting through Hospital Compare.
At press time, ACS leaders had completed visits to 12 cities to discuss how quality-improvement efforts enhance surgical patient safety and reduce costs as part of the College’s Inspiring Quality initiative. With the assistance of Weber Shandwick, we also released the video Driven, which explores how surgical leaders and other innovators over the past 100 years have inspired quality by advancing surgical care.
Education
The College continues to establish benchmarks and standards in surgical education. This past year, new emphasis was placed on helping surgeons and residents to navigate transitions in their careers. Perhaps the most significant example of our efforts to assist residents as they transition into practice is the development of fellowships for young surgeons who are leaving training and entering into general surgery practice. The College will begin pilot testing the ACS Transition to Practice Fellowship in General Surgery on July 1 at five institutions in regions of the United States that currently are underserved by general surgeons. Furthermore, the ACS is collaborating with the Association of Program Directors in Surgery and the Association for Surgical Education to develop a new Entering Surgery Resident Prep Curriculum.
Workforce issues
Rural areas feel the effects of general surgeon shortage most acutely. To ensure that the College is better able to address the challenges that rural surgical practice poses, the College established a new Advisory Council for Rural Surgery.
And, to help surgeons make smart decisions as the trend toward hospital employment for physicians continues to grow, the ACS developed a new resource titled Surgeons as Institutional Employees: A Strategic Look at the Dimensions of Surgeons as Employees of Hospitals. This document mailed with the February 2013 Bulletin.
Communications
The College continues to modernize its communications vehicles. In September 2012, the College released the first issue of the newly redesigned Bulletin and is now in the process of working with consultants to reconstruct the public website (www.facs.org) and to integrate the members’ portal into the main site. To increase the visibility of the College’s advocacy efforts, the ACS last summer launched a monthly e-newsletter, The ACS Advocate.
Looking forward
The ACS is continuing to develop proactive solutions to the challenges facing surgeons. Your continued input on how we can address your concerns is always welcome.
Dr. Hoyt is Executive Director of the American College of Surgeons.
As we commemorate the American College of Surgeons’ Centennial, the leadership and staff continue to develop and engage in initiatives designed to ensure that members of the College are best positioned to continuing providing quality surgical care for the next 100 years.
With respect to advocacy and health policy, the College remains at the forefront of efforts to repeal and replace the flawed sustainable growth rate formula used to calculate Medicare physician payment. Importantly, the College has been working on a proposal to replace the SGR with a value-based update, which emphasizes that value = quality/cost.
The ACS is also looking at alternative methods of resolving medical liability claims. We hosted a Medical Liability Summit in October, and articles based on the presentations given at the meeting will be published in a special issue of the Bulletin this March.
Advocacy
In addition, the College has successfully nominated Fellows to serve on panels that are advising the government on quality of care issues. Last year, ACS nominee Stephen B. Edge, MD, FACS, was appointed co-chair of the National Quality Forum’s Cancer Maintenance Endorsement Project, and Frank Opelka, MD, FACS, Associate Medical Director of the Division of Advocacy and Health Policy, was appointed chair of the Physician Consortium for Performance Improvement.
Quality improvement
The College’s National Surgical Quality Improvement Program (ACS NSQIP®) continues to grow in size and stature. More than 500 hospitals now participate in ACS NSQIP, and data promulgated through this outcome measurement system were key in the completion of a project that the College conducted last year with the Joint Commission Center for Transforming Healthcare aimed at reducing colorectal surgical site infections. A total of 135 SSIs were averted at seven health care institutions that participated in the demonstration project, saving those facilities more $3.7 million. The Centers for Disease Control and ACS NSQIP also have entered into a formal agreement to study SSI. Additionally, approximately 30% of ACS NSQIP hospitals are participating in the Centers for Medicare & Medicaid Services public reporting pilot program, Hospital Compare.
The Rapid Quality Reporting System has enabled Commission on Cancer–accredited programs to better adhere to five NQF-endorsed quality performance measures. Recognizing the potential value of this program, CMS recently contracted with the ACS to implement RQRS at 11 cancer centers that support public reporting through Hospital Compare.
At press time, ACS leaders had completed visits to 12 cities to discuss how quality-improvement efforts enhance surgical patient safety and reduce costs as part of the College’s Inspiring Quality initiative. With the assistance of Weber Shandwick, we also released the video Driven, which explores how surgical leaders and other innovators over the past 100 years have inspired quality by advancing surgical care.
Education
The College continues to establish benchmarks and standards in surgical education. This past year, new emphasis was placed on helping surgeons and residents to navigate transitions in their careers. Perhaps the most significant example of our efforts to assist residents as they transition into practice is the development of fellowships for young surgeons who are leaving training and entering into general surgery practice. The College will begin pilot testing the ACS Transition to Practice Fellowship in General Surgery on July 1 at five institutions in regions of the United States that currently are underserved by general surgeons. Furthermore, the ACS is collaborating with the Association of Program Directors in Surgery and the Association for Surgical Education to develop a new Entering Surgery Resident Prep Curriculum.
Workforce issues
Rural areas feel the effects of general surgeon shortage most acutely. To ensure that the College is better able to address the challenges that rural surgical practice poses, the College established a new Advisory Council for Rural Surgery.
And, to help surgeons make smart decisions as the trend toward hospital employment for physicians continues to grow, the ACS developed a new resource titled Surgeons as Institutional Employees: A Strategic Look at the Dimensions of Surgeons as Employees of Hospitals. This document mailed with the February 2013 Bulletin.
Communications
The College continues to modernize its communications vehicles. In September 2012, the College released the first issue of the newly redesigned Bulletin and is now in the process of working with consultants to reconstruct the public website (www.facs.org) and to integrate the members’ portal into the main site. To increase the visibility of the College’s advocacy efforts, the ACS last summer launched a monthly e-newsletter, The ACS Advocate.
Looking forward
The ACS is continuing to develop proactive solutions to the challenges facing surgeons. Your continued input on how we can address your concerns is always welcome.
Dr. Hoyt is Executive Director of the American College of Surgeons.
Everolimus approval now includes prevention of liver transplant rejection
Food and Drug Administration approval of the immunosuppressant drug everolimus has been expanded to include prophylaxis of organ rejection in adults undergoing a liver transplant.
The approval, on Feb. 15, was announced by the manufacturer, Novartis. Everolimus, a mammalian target of rapamycin (mTOR) inhibitor, was first approved for prophylaxis of organ rejection in adults undergoing a kidney transplant in 2010, and is taken twice a day by mouth.
The approved indication states that in liver transplant recipients, everolimus is used in combination with the calcineurin inhibitor tacrolimus (at a reduced dose) and corticosteroids, and that it should be administered no earlier than 30 days post transplant. Safety and efficacy have not been established in pediatric patients, according to the prescribing information.
FDA approval was based on the 12-month results of a phase III, open-label international study of 719 liver transplant recipients (mean age, 54 years). At 12 months, the rate of the "efficacy failure" endpoint (defined as biopsy-proven acute rejection, graft loss, death, or loss to follow-up) was "comparable" between those patients treated with the reduced dose of everolimus, started 30 days after transplantation (9%), and the patients treated with the standard dose of tacrolimus, started 30 days after transplantation (13.6%), according to the Novartis statement and prescribing information. All patients were treated with corticosteroids.
Everolimus is the first mTOR inhibitor approved for liver transplant recipients, and is the first immunosuppressant approved by the FDA for use in liver transplant recipients in more than 10 years, according to Novartis.
Novartis markets everolimus in the United States as Zortress. It was approved in the European Union in the fourth quarter of 2012 for adult liver transplant recipients; it is marketed as Certican in Europe.
The new label is available here. Serious adverse events associated with everolimus should be reported to the FDA’s MedWatch program at 800-332-1088 or www.fda.gov/medwatch/.
Food and Drug Administration approval of the immunosuppressant drug everolimus has been expanded to include prophylaxis of organ rejection in adults undergoing a liver transplant.
The approval, on Feb. 15, was announced by the manufacturer, Novartis. Everolimus, a mammalian target of rapamycin (mTOR) inhibitor, was first approved for prophylaxis of organ rejection in adults undergoing a kidney transplant in 2010, and is taken twice a day by mouth.
The approved indication states that in liver transplant recipients, everolimus is used in combination with the calcineurin inhibitor tacrolimus (at a reduced dose) and corticosteroids, and that it should be administered no earlier than 30 days post transplant. Safety and efficacy have not been established in pediatric patients, according to the prescribing information.
FDA approval was based on the 12-month results of a phase III, open-label international study of 719 liver transplant recipients (mean age, 54 years). At 12 months, the rate of the "efficacy failure" endpoint (defined as biopsy-proven acute rejection, graft loss, death, or loss to follow-up) was "comparable" between those patients treated with the reduced dose of everolimus, started 30 days after transplantation (9%), and the patients treated with the standard dose of tacrolimus, started 30 days after transplantation (13.6%), according to the Novartis statement and prescribing information. All patients were treated with corticosteroids.
Everolimus is the first mTOR inhibitor approved for liver transplant recipients, and is the first immunosuppressant approved by the FDA for use in liver transplant recipients in more than 10 years, according to Novartis.
Novartis markets everolimus in the United States as Zortress. It was approved in the European Union in the fourth quarter of 2012 for adult liver transplant recipients; it is marketed as Certican in Europe.
The new label is available here. Serious adverse events associated with everolimus should be reported to the FDA’s MedWatch program at 800-332-1088 or www.fda.gov/medwatch/.
Food and Drug Administration approval of the immunosuppressant drug everolimus has been expanded to include prophylaxis of organ rejection in adults undergoing a liver transplant.
The approval, on Feb. 15, was announced by the manufacturer, Novartis. Everolimus, a mammalian target of rapamycin (mTOR) inhibitor, was first approved for prophylaxis of organ rejection in adults undergoing a kidney transplant in 2010, and is taken twice a day by mouth.
The approved indication states that in liver transplant recipients, everolimus is used in combination with the calcineurin inhibitor tacrolimus (at a reduced dose) and corticosteroids, and that it should be administered no earlier than 30 days post transplant. Safety and efficacy have not been established in pediatric patients, according to the prescribing information.
FDA approval was based on the 12-month results of a phase III, open-label international study of 719 liver transplant recipients (mean age, 54 years). At 12 months, the rate of the "efficacy failure" endpoint (defined as biopsy-proven acute rejection, graft loss, death, or loss to follow-up) was "comparable" between those patients treated with the reduced dose of everolimus, started 30 days after transplantation (9%), and the patients treated with the standard dose of tacrolimus, started 30 days after transplantation (13.6%), according to the Novartis statement and prescribing information. All patients were treated with corticosteroids.
Everolimus is the first mTOR inhibitor approved for liver transplant recipients, and is the first immunosuppressant approved by the FDA for use in liver transplant recipients in more than 10 years, according to Novartis.
Novartis markets everolimus in the United States as Zortress. It was approved in the European Union in the fourth quarter of 2012 for adult liver transplant recipients; it is marketed as Certican in Europe.
The new label is available here. Serious adverse events associated with everolimus should be reported to the FDA’s MedWatch program at 800-332-1088 or www.fda.gov/medwatch/.