User login
Alopecia Universalis Treated With Tofacitinib: The Role of JAK/STAT Inhibitors in Hair Regrowth
Alopecia areata (AA) is an autoimmune disease that immunopathogenetically is thought to be due to breakdown of the immune privilege of the proximal hair follicle during the anagen growth phase. Alopecia areata has been reported to have a lifetime prevalence of 1.7%.1 Recent studies have specifically identified cytotoxic CD8+ NKG2D+ T cells as being responsible for the activation of AA.2-4 Two interleukins—IL-2 and IL-15—have been implicated to be cytotoxic sensitizers allowing CD8+ T cells to secrete IFN-γ and recognize autoantigens via major histocompatibility complex class I.5,6 Janus kinases (JAKs) are enzymes that play major roles in many different molecular processes. Specifically, JAK1/3 has been determined to arbitrate IL-15 activation of receptors on CD8+ T cells.7 These cells then interact with CD4 T cells, mast cells, and other inflammatory cells to cause destruction of the hair follicle without damage to the keratinocyte and melanocyte stem cells, allowing for reversible yet relapsing hair loss.8
Treatment of AA is difficult, requiring patience and strict compliance while taking into account duration of disease, age at presentation, site involvement, patient expectations, cost and insurance coverage, prior therapies, and any comorbidities. At the time of this case, no US Food and Drug Administration–approved drug regimen existed for the treatment of AA, and, to date, no treatment is preventative.4 We present a case of a patient with alopecia universalis of 11 years’ duration that was refractory to intralesional triamcinolone, clobetasol, minoxidil, and UVB brush therapy yet was successfully treated with tofacitinib.
Case Report
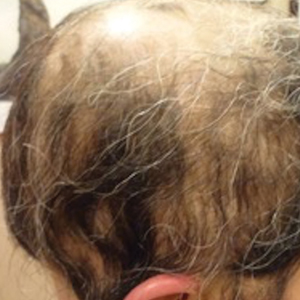
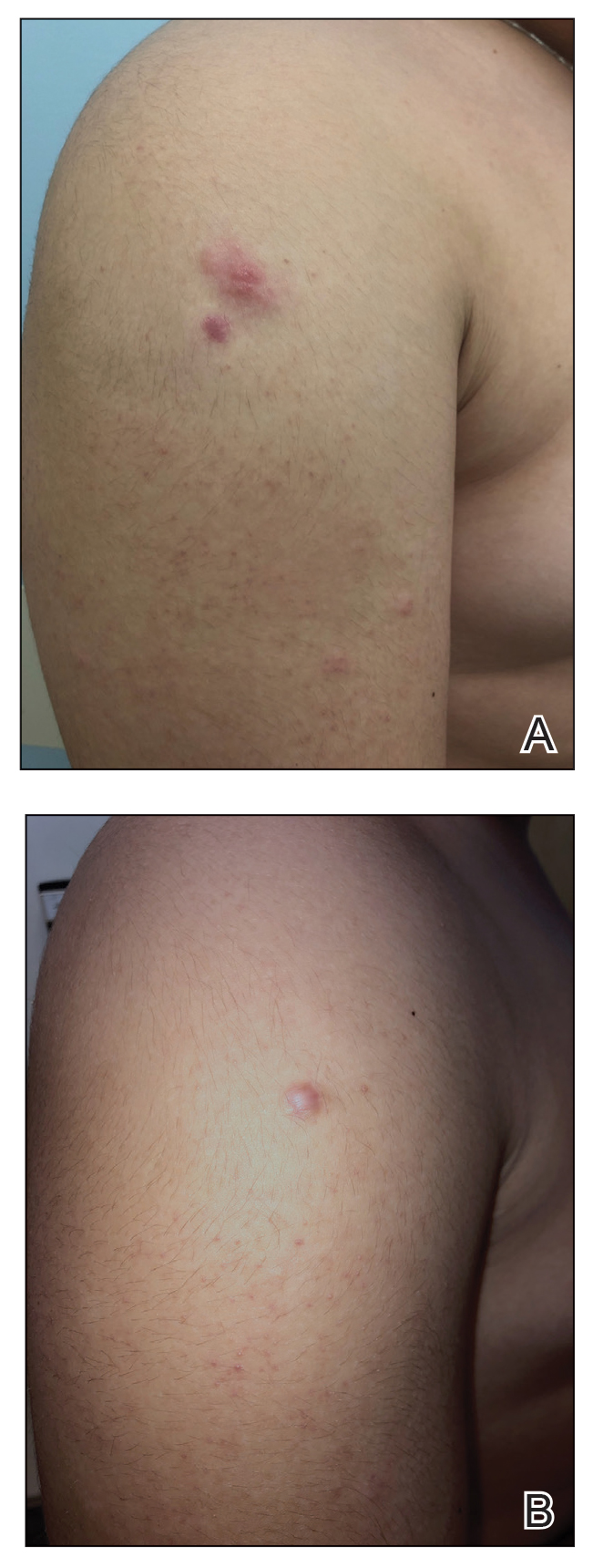
A 29-year-old otherwise-healthy woman presented to our clinic for treatment of alopecia universalis of 11 years’ duration that flared intermittently despite various treatments. Her medical history was unremarkable; however, she had a brother with alopecia universalis. She had no family history of any other autoimmune disorders. At the current presentation, the patient was known to have alopecia universalis with scant evidence of exclamation-point hairs on dermoscopy. Her treatment plan at this point consisted of intralesional triamcinolone to the active areas at 10 mg/mL every 4 weeks, plus clobetasol foam 0.05% at bedtime, minoxidil foam 5% at bedtime, and a UVB brush 3 times a week for 6 months before progressing to universalis type because of hair loss in the eyebrows and eyelashes. This treatment plan continued for 1 year with minimal improvement of the alopecia (Figure 1).
The patient was dissatisfied and wanted to discontinue therapy. Because these treatment options were exhausted with minimal benefit, the patient was then considered for treatment with tofacitinib. Baseline studies were performed, including purified protein derivative, complete blood cell count with differential, comprehensive metabolic panel, lipid profile, and liver function tests, all of which were within reference range. Insurance initially denied coverage of this therapy; a prior authorization was subsequently submitted and denied. A letter of medical necessity was then proposed, and approval for tofacitinib was finally granted. The patient was started on tofacitinib 5 mg twice daily and was monitored every 2 months with a complete blood cell count, comprehensive metabolic panel, lipid panels, and liver function tests. She had a platelet count of 112,000/μL (reference range, 150,000–450,000/μL) at baseline, and continued monitoring revealed a platelet count of 83,000 after 7 months of treatment. This platelet abnormality was evaluated by a hematologist and found to be within reference range; subsequent monitoring did not reveal any abnormalities.
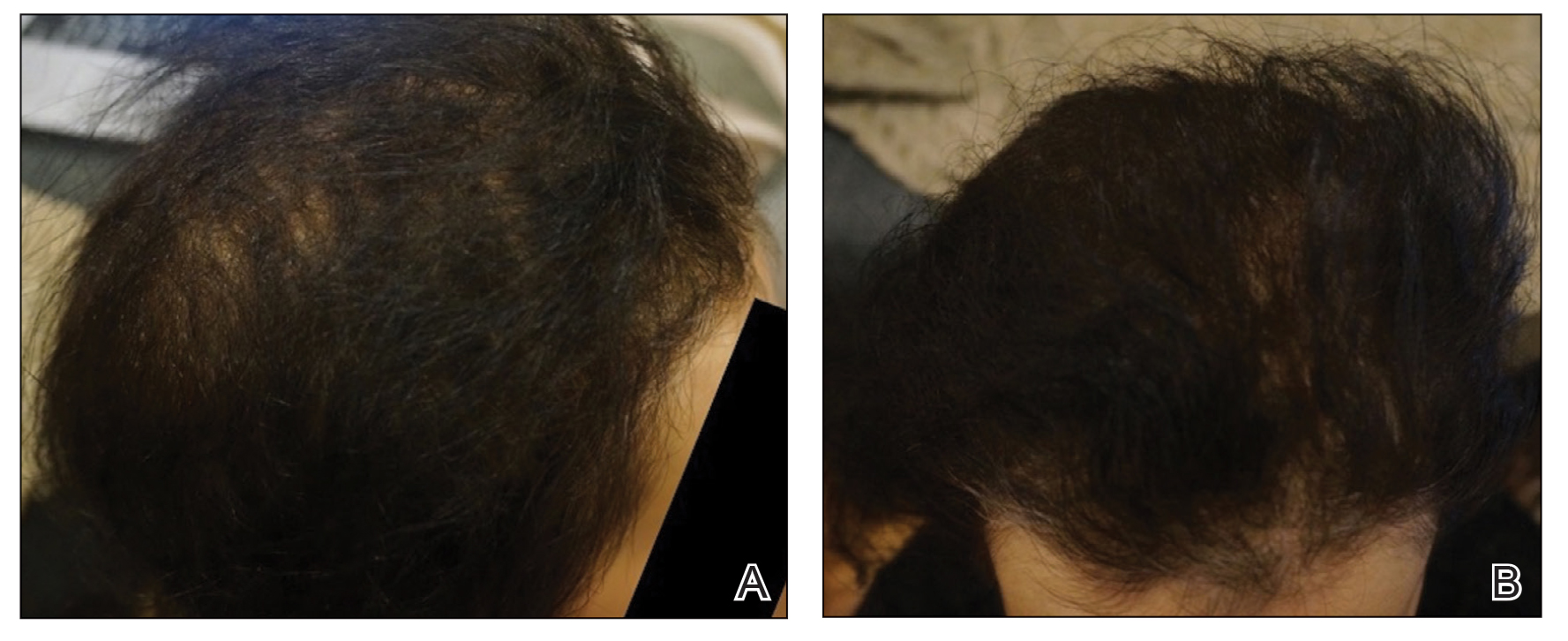
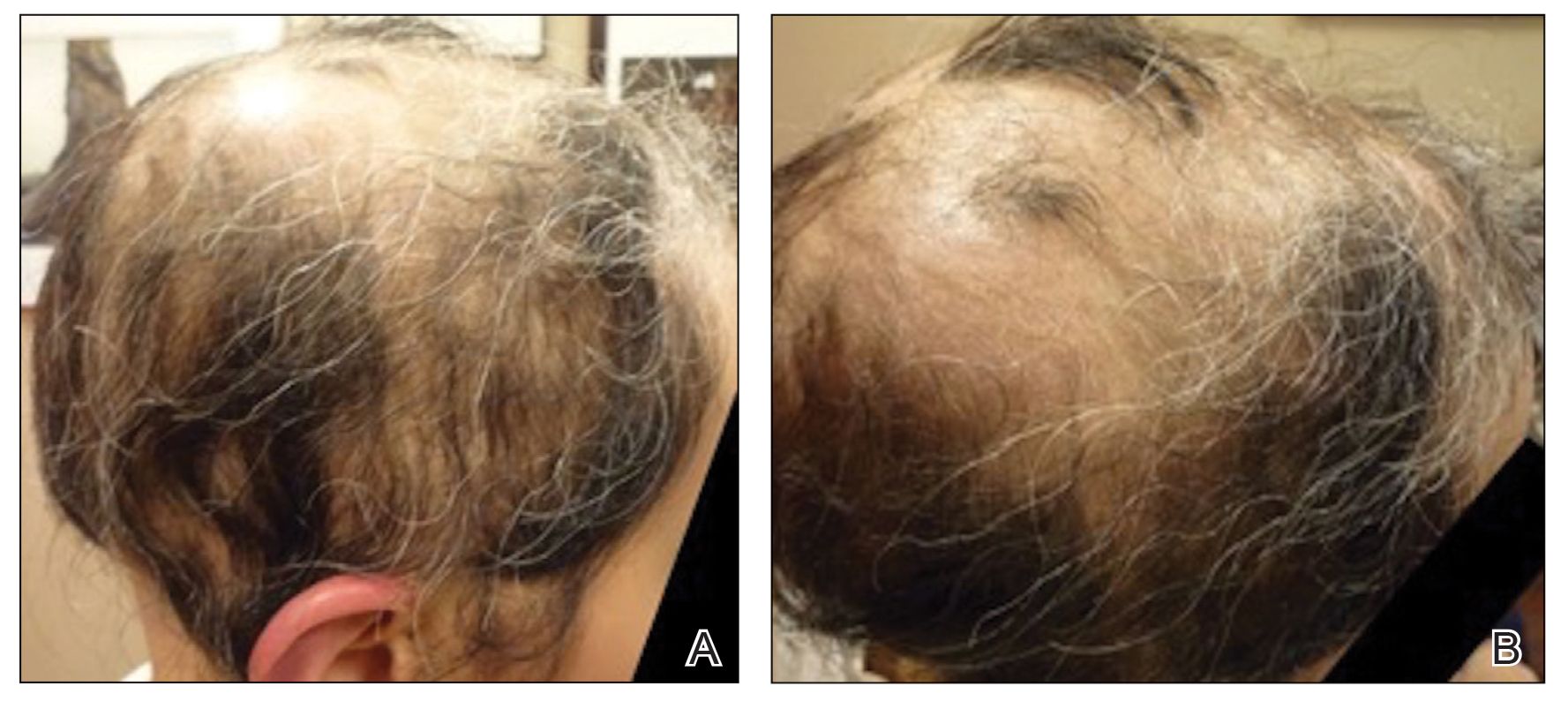
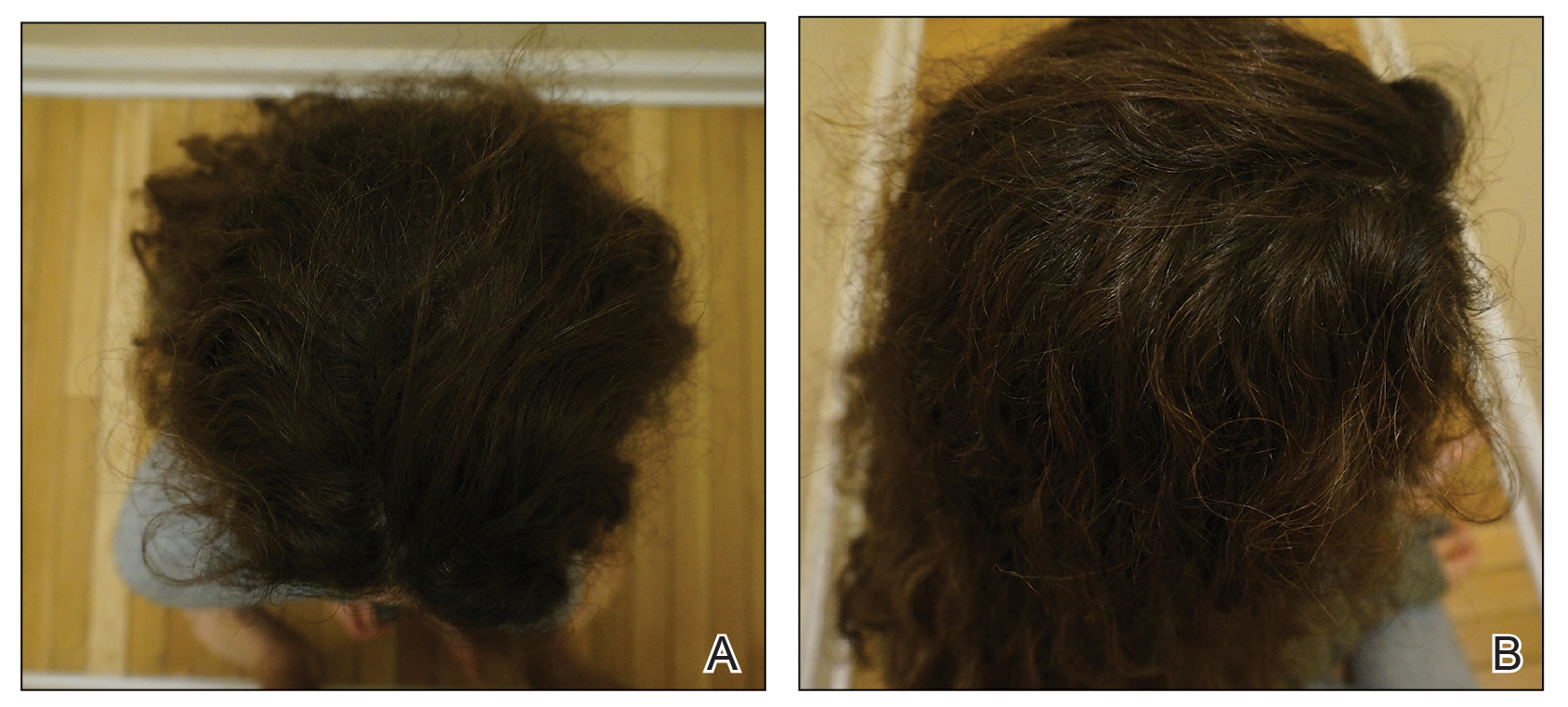
Initial hair growth on the scalp was diffuse with thin, white to light brown hairs in areas of hair loss at months 1 and 2, with progressive hair growth over months 3 to 7. Eyebrow hair growth was noted beginning at month 6. One year later, only hair regrowth occurred without any adverse events (Figure 2). After 5 years of treatment, the patient had a full head of thick hair (Figure 3). The tofacitinib dosage was 5 mg twice daily at initiation, and after 1 year increased to 10 mg twice daily. Her medical insurance subsequently changed and the regimen was adjusted to an 11-mg tablet and 5-mg tablet daily. She remained on this regimen with success.
Comment
Use of JAK Inhibitors—Reports and studies have shed light on the use and efficacy of JAK inhibitors in AA (Table).5-11 Tofacitinib is a selective JAK1/3 inhibitor that predominantly inhibits JAK3 but also inhibits JAK1, albeit to a lesser degree, which interferes with the JAK/STAT (signal transducer and activator of transcription) cascade responsible for the production, differentiation, and function of various B cells, T cells, and natural killer cells.2 Although it was developed for the management of allograft rejection, tofacitinib has made headway in rheumatology for treatment of patients with moderate to severe rheumatoid arthritis who are unable to take or are not responding to methotrexate.2 Since 2014, tofacitinib has been introduced to the therapeutic realm for AA but is not yet approved by the US Food and Drug Administration.3,4
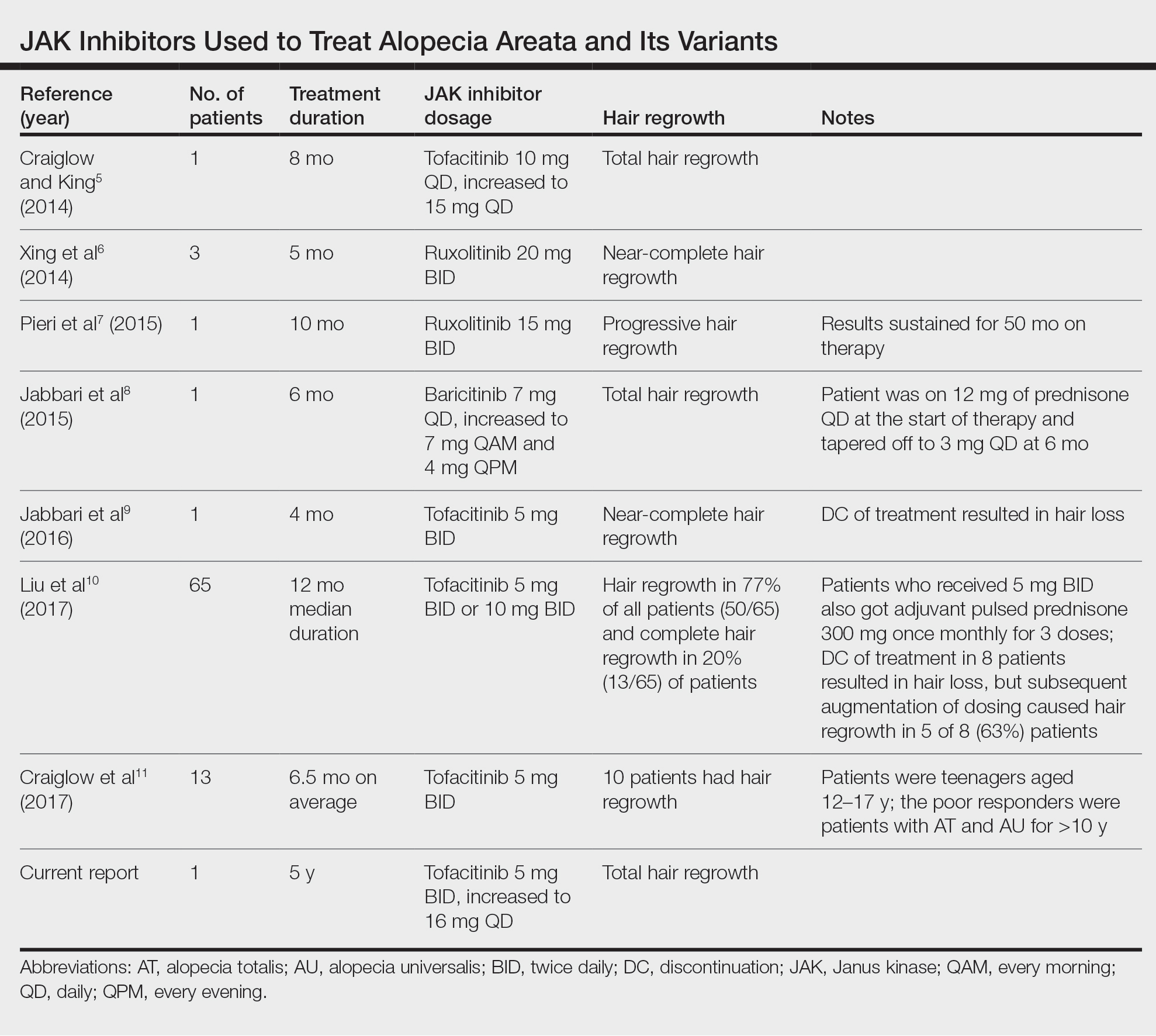
In 2014, Craiglow and King5 reported use of tofacitinib with dosages beginning at 10 mg/d and increasing to 15 mg/d in a patient with alopecia universalis and psoriasis. Total hair regrowth was noted after 8 months of therapy.5 Xing et al6 described 3 patients treated with ruxolitinib, a JAK1/2 inhibitor approved for the treatment of myelofibrosis, at an oral dose of 20 mg twice daily with near-complete hair regrowth after 5 months of treatment.6 Biopsies from lesions at baseline and after 3 months of therapy revealed a reduction in perifollicular T cells and in HLA class I and II expression in follicles.6 A patient in Italy with essential thrombocythemia and concurrent alopecia universalis was enrolled in a clinical trial with ruxolitinib and was treated with 15 mg twice daily. After 10 months of treatment, the patient had progressive hair regrowth that was sustained for more than 50 months of therapy.7 Baricitinib, a JAK1/2 inhibitor, was used in a 17-year-old adolescent boy to assess efficacy of the drug in
A recent retrospective study assessing response to tofacitinib in adults with AA (>40% hair loss), alopecia totalis, alopecia universalis, and stable or progressive diseases for at least 6 months determined a clinical response in 50 of 65 (77%) patients, with 13 patients exhibiting a complete response.10 Patients in this study were started on tofacitinib 5 mg twice daily with the addition of adjuvant pulsed prednisone (300 mg once monthly for 3 doses) with or without doubled dosing of tofacitinib if they had a halt in hair regrowth. This study demonstrated some benefit when pulsed prednisone was combined with the daily tofacitinib therapy. However, the study emphasized the importance of maintenance therapy, as 8 patients experienced hair loss with discontinuation after previously having hair regrowth; 5 (63%) of these patients experienced regrowth with augmentation of dosing or addition of adjuvant therapy.10
Another group of investigators assessed the efficacy of tofacitinib 5 mg in 13 adolescents aged 12 to 17 years, most with alopecia universalis (46% [6/13]); 10 of 13 (77%) patients responded to treatment with a mean duration of 6.5 months. The patients who had alopecia totalis and alopecia universalis for more than 10 years were poor responders to tofacitinib, and in fact, 1 of 13 (33%) patients in the study who did not respond to therapy had disease for 12 years.11 Therefore, starting tofacitinib either long-term or intermittently should be considered in children diagnosed early with severe AA, alopecia totalis, or alopecia universalis to prevent irreversible hair loss or progressive disease12,13; however, further data are required to assess efficacy and long-term benefits of this type of regimen.
Safety Profile—Widespread use of a medication is determined not only by its efficacy profile but also its safety profile. With any medication that exhibits immunosuppressive effects, adverse events must be considered and thoroughly discussed with patients and their primary care physicians. A prospective, open-label, single-arm trial examined the efficacy and safety of tofacitinib 5 mg twice daily in the treatment of AA and its more severe forms over 3 months.12 Of the 66 patients who completed the trial, 64% (42/66) exhibited a positive response to tofacitinib. Relapse was noted in 8.5 weeks after discontinuation of tofacitinib, reiterating the potential need for a maintenance regimen. In this study, 25.8% (17/66) of patients experienced infections as adverse events including (in decreasing order) upper respiratory tract infections, urinary tract infections, herpes zoster, conjunctivitis, bronchitis, mononucleosis, and paronychia. No reports of new or recurrent malignancy were noted. Other more constitutional adverse events were noted including headaches, abdominal pain, acne, diarrhea, fatigue, nausea, pruritus, hot flashes, cough, folliculitis, weight gain, dry eyes, and amenorrhea. One patient with a pre-existing liver condition experienced transaminitis that resolved with weight loss. There also were noted increases in low- and high-density lipoprotein levels.12 Our patient with baseline thrombocytopenia had mild drops in platelet count that subsequently stabilized and did not result in any bleeding abnormalities.
Duration of Therapy—Tofacitinib has demonstrated some preliminary success in the management of AA, but the appropriate duration of treatment requires further investigation. Our patient has been on tofacitinib for more than 5 years. She started at a total dosage of 10 mg/d, which increased to 16 mg/d. Initial dosing with maintenance regimens needs to be established for further widespread use to maximize benefit and minimize harm.
At what point do we decide to continue or stop treatment in patients who do not respond as expected or plateau? This is another critical question; our patient had periods of slowed growth and plateauing, but knowing the risks and benefits, she continued the medication and eventually experienced improved regrowth again.
Conclusion
Throughout the literature and in our patient, tofacitinib has demonstrated efficacy in treating AA. When other conventional therapies have failed, use of tofacitinib should be considered.
- Safavi KH, Muller SA, Suman VJ, et al. Incidence of alopecia areata in Olmstead County, Minnesota, 1975 through 1989. Mayo Clin Proc. 1995;70:628-633.
- Borazan NH, Furst DE. Nonsteroidal anti-inflammatory drugs, disease-modifying antirheumatic drugs, nonopioid analgesics, & drugs used in gout. In: Katzung BG, Trevor AJ, eds. Basic & Clinical Pharmacology. 13th ed. McGraw-Hill; 2015:618-642.
- Shapiro J. Current treatment of alopecia areata. J Investig Dermatol Symp Proc. 2013;16:S42-S44.
- Shapiro J. Dermatologic therapy: alopecia areata update. Dermatol Ther. 2011;24:301.
- Craiglow BG, King BA. Killing two birds with one stone: oral tofacitinib reverses alopecia universalis in a patient with plaque psoriasis. J Invest Dermatol. 2014;134:2988-2990.
- Xing L, Dai Z, Jabbari A, et al. Alopecia areata is driven by cytotoxic T lymphocytes and is reversed by JAK inhibition. Nat Med. 2014;20:1043-1049.
- Pieri L, Guglielmelli P, Vannucchi AM. Ruxolitinib-induced reversal of alopecia universalis in a patient with essential thrombocythemia. Am J Hematol. 2015;90:82-83.
- Jabbari A, Dai Z, Xing L, et al. Reversal of alopecia areata following treatment with the JAK1/2 inhibitor baricitinib. EbioMedicine. 2015;2:351-355.
- Jabbari A, Nguyen N, Cerise JE, et al. Treatment of an alopecia areata patient with tofacitinib results in regrowth of hair and changes in serum and skin biomarkers. Exp Dermatol. 2016;25:642-643.
- Liu LY, Craiglow BG, Dai F, et al. Tofacitinib for the treatment of severe alopecia areata and variants: a study of 90 patients. J Am Acad Dermatol. 2017;76:22-28.
- Craiglow BG, Liu LY, King BA. Tofacitinib for the treatment of alopecia areata and variants in adolescents. J Am Acad Dermatol. 2017;76:29-32.
- Kennedy Crispin M, Ko JM, Craiglow BG, et al. Safety and efficacy of the JAK inhibitor tofacitinib citrate in patients with alopecia areata. JCI Insight. 2016;1:E89776.
- Iorizzo M, Tosti A. Emerging drugs for alopecia areata: JAK inhibitors. Expert Opin Emerg Drugs. 2018;23:77-81.
Alopecia areata (AA) is an autoimmune disease that immunopathogenetically is thought to be due to breakdown of the immune privilege of the proximal hair follicle during the anagen growth phase. Alopecia areata has been reported to have a lifetime prevalence of 1.7%.1 Recent studies have specifically identified cytotoxic CD8+ NKG2D+ T cells as being responsible for the activation of AA.2-4 Two interleukins—IL-2 and IL-15—have been implicated to be cytotoxic sensitizers allowing CD8+ T cells to secrete IFN-γ and recognize autoantigens via major histocompatibility complex class I.5,6 Janus kinases (JAKs) are enzymes that play major roles in many different molecular processes. Specifically, JAK1/3 has been determined to arbitrate IL-15 activation of receptors on CD8+ T cells.7 These cells then interact with CD4 T cells, mast cells, and other inflammatory cells to cause destruction of the hair follicle without damage to the keratinocyte and melanocyte stem cells, allowing for reversible yet relapsing hair loss.8
Treatment of AA is difficult, requiring patience and strict compliance while taking into account duration of disease, age at presentation, site involvement, patient expectations, cost and insurance coverage, prior therapies, and any comorbidities. At the time of this case, no US Food and Drug Administration–approved drug regimen existed for the treatment of AA, and, to date, no treatment is preventative.4 We present a case of a patient with alopecia universalis of 11 years’ duration that was refractory to intralesional triamcinolone, clobetasol, minoxidil, and UVB brush therapy yet was successfully treated with tofacitinib.
Case Report
A 29-year-old otherwise-healthy woman presented to our clinic for treatment of alopecia universalis of 11 years’ duration that flared intermittently despite various treatments. Her medical history was unremarkable; however, she had a brother with alopecia universalis. She had no family history of any other autoimmune disorders. At the current presentation, the patient was known to have alopecia universalis with scant evidence of exclamation-point hairs on dermoscopy. Her treatment plan at this point consisted of intralesional triamcinolone to the active areas at 10 mg/mL every 4 weeks, plus clobetasol foam 0.05% at bedtime, minoxidil foam 5% at bedtime, and a UVB brush 3 times a week for 6 months before progressing to universalis type because of hair loss in the eyebrows and eyelashes. This treatment plan continued for 1 year with minimal improvement of the alopecia (Figure 1).

The patient was dissatisfied and wanted to discontinue therapy. Because these treatment options were exhausted with minimal benefit, the patient was then considered for treatment with tofacitinib. Baseline studies were performed, including purified protein derivative, complete blood cell count with differential, comprehensive metabolic panel, lipid profile, and liver function tests, all of which were within reference range. Insurance initially denied coverage of this therapy; a prior authorization was subsequently submitted and denied. A letter of medical necessity was then proposed, and approval for tofacitinib was finally granted. The patient was started on tofacitinib 5 mg twice daily and was monitored every 2 months with a complete blood cell count, comprehensive metabolic panel, lipid panels, and liver function tests. She had a platelet count of 112,000/μL (reference range, 150,000–450,000/μL) at baseline, and continued monitoring revealed a platelet count of 83,000 after 7 months of treatment. This platelet abnormality was evaluated by a hematologist and found to be within reference range; subsequent monitoring did not reveal any abnormalities.

Initial hair growth on the scalp was diffuse with thin, white to light brown hairs in areas of hair loss at months 1 and 2, with progressive hair growth over months 3 to 7. Eyebrow hair growth was noted beginning at month 6. One year later, only hair regrowth occurred without any adverse events (Figure 2). After 5 years of treatment, the patient had a full head of thick hair (Figure 3). The tofacitinib dosage was 5 mg twice daily at initiation, and after 1 year increased to 10 mg twice daily. Her medical insurance subsequently changed and the regimen was adjusted to an 11-mg tablet and 5-mg tablet daily. She remained on this regimen with success.

Comment
Use of JAK Inhibitors—Reports and studies have shed light on the use and efficacy of JAK inhibitors in AA (Table).5-11 Tofacitinib is a selective JAK1/3 inhibitor that predominantly inhibits JAK3 but also inhibits JAK1, albeit to a lesser degree, which interferes with the JAK/STAT (signal transducer and activator of transcription) cascade responsible for the production, differentiation, and function of various B cells, T cells, and natural killer cells.2 Although it was developed for the management of allograft rejection, tofacitinib has made headway in rheumatology for treatment of patients with moderate to severe rheumatoid arthritis who are unable to take or are not responding to methotrexate.2 Since 2014, tofacitinib has been introduced to the therapeutic realm for AA but is not yet approved by the US Food and Drug Administration.3,4

In 2014, Craiglow and King5 reported use of tofacitinib with dosages beginning at 10 mg/d and increasing to 15 mg/d in a patient with alopecia universalis and psoriasis. Total hair regrowth was noted after 8 months of therapy.5 Xing et al6 described 3 patients treated with ruxolitinib, a JAK1/2 inhibitor approved for the treatment of myelofibrosis, at an oral dose of 20 mg twice daily with near-complete hair regrowth after 5 months of treatment.6 Biopsies from lesions at baseline and after 3 months of therapy revealed a reduction in perifollicular T cells and in HLA class I and II expression in follicles.6 A patient in Italy with essential thrombocythemia and concurrent alopecia universalis was enrolled in a clinical trial with ruxolitinib and was treated with 15 mg twice daily. After 10 months of treatment, the patient had progressive hair regrowth that was sustained for more than 50 months of therapy.7 Baricitinib, a JAK1/2 inhibitor, was used in a 17-year-old adolescent boy to assess efficacy of the drug in
A recent retrospective study assessing response to tofacitinib in adults with AA (>40% hair loss), alopecia totalis, alopecia universalis, and stable or progressive diseases for at least 6 months determined a clinical response in 50 of 65 (77%) patients, with 13 patients exhibiting a complete response.10 Patients in this study were started on tofacitinib 5 mg twice daily with the addition of adjuvant pulsed prednisone (300 mg once monthly for 3 doses) with or without doubled dosing of tofacitinib if they had a halt in hair regrowth. This study demonstrated some benefit when pulsed prednisone was combined with the daily tofacitinib therapy. However, the study emphasized the importance of maintenance therapy, as 8 patients experienced hair loss with discontinuation after previously having hair regrowth; 5 (63%) of these patients experienced regrowth with augmentation of dosing or addition of adjuvant therapy.10
Another group of investigators assessed the efficacy of tofacitinib 5 mg in 13 adolescents aged 12 to 17 years, most with alopecia universalis (46% [6/13]); 10 of 13 (77%) patients responded to treatment with a mean duration of 6.5 months. The patients who had alopecia totalis and alopecia universalis for more than 10 years were poor responders to tofacitinib, and in fact, 1 of 13 (33%) patients in the study who did not respond to therapy had disease for 12 years.11 Therefore, starting tofacitinib either long-term or intermittently should be considered in children diagnosed early with severe AA, alopecia totalis, or alopecia universalis to prevent irreversible hair loss or progressive disease12,13; however, further data are required to assess efficacy and long-term benefits of this type of regimen.
Safety Profile—Widespread use of a medication is determined not only by its efficacy profile but also its safety profile. With any medication that exhibits immunosuppressive effects, adverse events must be considered and thoroughly discussed with patients and their primary care physicians. A prospective, open-label, single-arm trial examined the efficacy and safety of tofacitinib 5 mg twice daily in the treatment of AA and its more severe forms over 3 months.12 Of the 66 patients who completed the trial, 64% (42/66) exhibited a positive response to tofacitinib. Relapse was noted in 8.5 weeks after discontinuation of tofacitinib, reiterating the potential need for a maintenance regimen. In this study, 25.8% (17/66) of patients experienced infections as adverse events including (in decreasing order) upper respiratory tract infections, urinary tract infections, herpes zoster, conjunctivitis, bronchitis, mononucleosis, and paronychia. No reports of new or recurrent malignancy were noted. Other more constitutional adverse events were noted including headaches, abdominal pain, acne, diarrhea, fatigue, nausea, pruritus, hot flashes, cough, folliculitis, weight gain, dry eyes, and amenorrhea. One patient with a pre-existing liver condition experienced transaminitis that resolved with weight loss. There also were noted increases in low- and high-density lipoprotein levels.12 Our patient with baseline thrombocytopenia had mild drops in platelet count that subsequently stabilized and did not result in any bleeding abnormalities.
Duration of Therapy—Tofacitinib has demonstrated some preliminary success in the management of AA, but the appropriate duration of treatment requires further investigation. Our patient has been on tofacitinib for more than 5 years. She started at a total dosage of 10 mg/d, which increased to 16 mg/d. Initial dosing with maintenance regimens needs to be established for further widespread use to maximize benefit and minimize harm.
At what point do we decide to continue or stop treatment in patients who do not respond as expected or plateau? This is another critical question; our patient had periods of slowed growth and plateauing, but knowing the risks and benefits, she continued the medication and eventually experienced improved regrowth again.
Conclusion
Throughout the literature and in our patient, tofacitinib has demonstrated efficacy in treating AA. When other conventional therapies have failed, use of tofacitinib should be considered.
Alopecia areata (AA) is an autoimmune disease that immunopathogenetically is thought to be due to breakdown of the immune privilege of the proximal hair follicle during the anagen growth phase. Alopecia areata has been reported to have a lifetime prevalence of 1.7%.1 Recent studies have specifically identified cytotoxic CD8+ NKG2D+ T cells as being responsible for the activation of AA.2-4 Two interleukins—IL-2 and IL-15—have been implicated to be cytotoxic sensitizers allowing CD8+ T cells to secrete IFN-γ and recognize autoantigens via major histocompatibility complex class I.5,6 Janus kinases (JAKs) are enzymes that play major roles in many different molecular processes. Specifically, JAK1/3 has been determined to arbitrate IL-15 activation of receptors on CD8+ T cells.7 These cells then interact with CD4 T cells, mast cells, and other inflammatory cells to cause destruction of the hair follicle without damage to the keratinocyte and melanocyte stem cells, allowing for reversible yet relapsing hair loss.8
Treatment of AA is difficult, requiring patience and strict compliance while taking into account duration of disease, age at presentation, site involvement, patient expectations, cost and insurance coverage, prior therapies, and any comorbidities. At the time of this case, no US Food and Drug Administration–approved drug regimen existed for the treatment of AA, and, to date, no treatment is preventative.4 We present a case of a patient with alopecia universalis of 11 years’ duration that was refractory to intralesional triamcinolone, clobetasol, minoxidil, and UVB brush therapy yet was successfully treated with tofacitinib.
Case Report
A 29-year-old otherwise-healthy woman presented to our clinic for treatment of alopecia universalis of 11 years’ duration that flared intermittently despite various treatments. Her medical history was unremarkable; however, she had a brother with alopecia universalis. She had no family history of any other autoimmune disorders. At the current presentation, the patient was known to have alopecia universalis with scant evidence of exclamation-point hairs on dermoscopy. Her treatment plan at this point consisted of intralesional triamcinolone to the active areas at 10 mg/mL every 4 weeks, plus clobetasol foam 0.05% at bedtime, minoxidil foam 5% at bedtime, and a UVB brush 3 times a week for 6 months before progressing to universalis type because of hair loss in the eyebrows and eyelashes. This treatment plan continued for 1 year with minimal improvement of the alopecia (Figure 1).

The patient was dissatisfied and wanted to discontinue therapy. Because these treatment options were exhausted with minimal benefit, the patient was then considered for treatment with tofacitinib. Baseline studies were performed, including purified protein derivative, complete blood cell count with differential, comprehensive metabolic panel, lipid profile, and liver function tests, all of which were within reference range. Insurance initially denied coverage of this therapy; a prior authorization was subsequently submitted and denied. A letter of medical necessity was then proposed, and approval for tofacitinib was finally granted. The patient was started on tofacitinib 5 mg twice daily and was monitored every 2 months with a complete blood cell count, comprehensive metabolic panel, lipid panels, and liver function tests. She had a platelet count of 112,000/μL (reference range, 150,000–450,000/μL) at baseline, and continued monitoring revealed a platelet count of 83,000 after 7 months of treatment. This platelet abnormality was evaluated by a hematologist and found to be within reference range; subsequent monitoring did not reveal any abnormalities.

Initial hair growth on the scalp was diffuse with thin, white to light brown hairs in areas of hair loss at months 1 and 2, with progressive hair growth over months 3 to 7. Eyebrow hair growth was noted beginning at month 6. One year later, only hair regrowth occurred without any adverse events (Figure 2). After 5 years of treatment, the patient had a full head of thick hair (Figure 3). The tofacitinib dosage was 5 mg twice daily at initiation, and after 1 year increased to 10 mg twice daily. Her medical insurance subsequently changed and the regimen was adjusted to an 11-mg tablet and 5-mg tablet daily. She remained on this regimen with success.

Comment
Use of JAK Inhibitors—Reports and studies have shed light on the use and efficacy of JAK inhibitors in AA (Table).5-11 Tofacitinib is a selective JAK1/3 inhibitor that predominantly inhibits JAK3 but also inhibits JAK1, albeit to a lesser degree, which interferes with the JAK/STAT (signal transducer and activator of transcription) cascade responsible for the production, differentiation, and function of various B cells, T cells, and natural killer cells.2 Although it was developed for the management of allograft rejection, tofacitinib has made headway in rheumatology for treatment of patients with moderate to severe rheumatoid arthritis who are unable to take or are not responding to methotrexate.2 Since 2014, tofacitinib has been introduced to the therapeutic realm for AA but is not yet approved by the US Food and Drug Administration.3,4

In 2014, Craiglow and King5 reported use of tofacitinib with dosages beginning at 10 mg/d and increasing to 15 mg/d in a patient with alopecia universalis and psoriasis. Total hair regrowth was noted after 8 months of therapy.5 Xing et al6 described 3 patients treated with ruxolitinib, a JAK1/2 inhibitor approved for the treatment of myelofibrosis, at an oral dose of 20 mg twice daily with near-complete hair regrowth after 5 months of treatment.6 Biopsies from lesions at baseline and after 3 months of therapy revealed a reduction in perifollicular T cells and in HLA class I and II expression in follicles.6 A patient in Italy with essential thrombocythemia and concurrent alopecia universalis was enrolled in a clinical trial with ruxolitinib and was treated with 15 mg twice daily. After 10 months of treatment, the patient had progressive hair regrowth that was sustained for more than 50 months of therapy.7 Baricitinib, a JAK1/2 inhibitor, was used in a 17-year-old adolescent boy to assess efficacy of the drug in
A recent retrospective study assessing response to tofacitinib in adults with AA (>40% hair loss), alopecia totalis, alopecia universalis, and stable or progressive diseases for at least 6 months determined a clinical response in 50 of 65 (77%) patients, with 13 patients exhibiting a complete response.10 Patients in this study were started on tofacitinib 5 mg twice daily with the addition of adjuvant pulsed prednisone (300 mg once monthly for 3 doses) with or without doubled dosing of tofacitinib if they had a halt in hair regrowth. This study demonstrated some benefit when pulsed prednisone was combined with the daily tofacitinib therapy. However, the study emphasized the importance of maintenance therapy, as 8 patients experienced hair loss with discontinuation after previously having hair regrowth; 5 (63%) of these patients experienced regrowth with augmentation of dosing or addition of adjuvant therapy.10
Another group of investigators assessed the efficacy of tofacitinib 5 mg in 13 adolescents aged 12 to 17 years, most with alopecia universalis (46% [6/13]); 10 of 13 (77%) patients responded to treatment with a mean duration of 6.5 months. The patients who had alopecia totalis and alopecia universalis for more than 10 years were poor responders to tofacitinib, and in fact, 1 of 13 (33%) patients in the study who did not respond to therapy had disease for 12 years.11 Therefore, starting tofacitinib either long-term or intermittently should be considered in children diagnosed early with severe AA, alopecia totalis, or alopecia universalis to prevent irreversible hair loss or progressive disease12,13; however, further data are required to assess efficacy and long-term benefits of this type of regimen.
Safety Profile—Widespread use of a medication is determined not only by its efficacy profile but also its safety profile. With any medication that exhibits immunosuppressive effects, adverse events must be considered and thoroughly discussed with patients and their primary care physicians. A prospective, open-label, single-arm trial examined the efficacy and safety of tofacitinib 5 mg twice daily in the treatment of AA and its more severe forms over 3 months.12 Of the 66 patients who completed the trial, 64% (42/66) exhibited a positive response to tofacitinib. Relapse was noted in 8.5 weeks after discontinuation of tofacitinib, reiterating the potential need for a maintenance regimen. In this study, 25.8% (17/66) of patients experienced infections as adverse events including (in decreasing order) upper respiratory tract infections, urinary tract infections, herpes zoster, conjunctivitis, bronchitis, mononucleosis, and paronychia. No reports of new or recurrent malignancy were noted. Other more constitutional adverse events were noted including headaches, abdominal pain, acne, diarrhea, fatigue, nausea, pruritus, hot flashes, cough, folliculitis, weight gain, dry eyes, and amenorrhea. One patient with a pre-existing liver condition experienced transaminitis that resolved with weight loss. There also were noted increases in low- and high-density lipoprotein levels.12 Our patient with baseline thrombocytopenia had mild drops in platelet count that subsequently stabilized and did not result in any bleeding abnormalities.
Duration of Therapy—Tofacitinib has demonstrated some preliminary success in the management of AA, but the appropriate duration of treatment requires further investigation. Our patient has been on tofacitinib for more than 5 years. She started at a total dosage of 10 mg/d, which increased to 16 mg/d. Initial dosing with maintenance regimens needs to be established for further widespread use to maximize benefit and minimize harm.
At what point do we decide to continue or stop treatment in patients who do not respond as expected or plateau? This is another critical question; our patient had periods of slowed growth and plateauing, but knowing the risks and benefits, she continued the medication and eventually experienced improved regrowth again.
Conclusion
Throughout the literature and in our patient, tofacitinib has demonstrated efficacy in treating AA. When other conventional therapies have failed, use of tofacitinib should be considered.
- Safavi KH, Muller SA, Suman VJ, et al. Incidence of alopecia areata in Olmstead County, Minnesota, 1975 through 1989. Mayo Clin Proc. 1995;70:628-633.
- Borazan NH, Furst DE. Nonsteroidal anti-inflammatory drugs, disease-modifying antirheumatic drugs, nonopioid analgesics, & drugs used in gout. In: Katzung BG, Trevor AJ, eds. Basic & Clinical Pharmacology. 13th ed. McGraw-Hill; 2015:618-642.
- Shapiro J. Current treatment of alopecia areata. J Investig Dermatol Symp Proc. 2013;16:S42-S44.
- Shapiro J. Dermatologic therapy: alopecia areata update. Dermatol Ther. 2011;24:301.
- Craiglow BG, King BA. Killing two birds with one stone: oral tofacitinib reverses alopecia universalis in a patient with plaque psoriasis. J Invest Dermatol. 2014;134:2988-2990.
- Xing L, Dai Z, Jabbari A, et al. Alopecia areata is driven by cytotoxic T lymphocytes and is reversed by JAK inhibition. Nat Med. 2014;20:1043-1049.
- Pieri L, Guglielmelli P, Vannucchi AM. Ruxolitinib-induced reversal of alopecia universalis in a patient with essential thrombocythemia. Am J Hematol. 2015;90:82-83.
- Jabbari A, Dai Z, Xing L, et al. Reversal of alopecia areata following treatment with the JAK1/2 inhibitor baricitinib. EbioMedicine. 2015;2:351-355.
- Jabbari A, Nguyen N, Cerise JE, et al. Treatment of an alopecia areata patient with tofacitinib results in regrowth of hair and changes in serum and skin biomarkers. Exp Dermatol. 2016;25:642-643.
- Liu LY, Craiglow BG, Dai F, et al. Tofacitinib for the treatment of severe alopecia areata and variants: a study of 90 patients. J Am Acad Dermatol. 2017;76:22-28.
- Craiglow BG, Liu LY, King BA. Tofacitinib for the treatment of alopecia areata and variants in adolescents. J Am Acad Dermatol. 2017;76:29-32.
- Kennedy Crispin M, Ko JM, Craiglow BG, et al. Safety and efficacy of the JAK inhibitor tofacitinib citrate in patients with alopecia areata. JCI Insight. 2016;1:E89776.
- Iorizzo M, Tosti A. Emerging drugs for alopecia areata: JAK inhibitors. Expert Opin Emerg Drugs. 2018;23:77-81.
- Safavi KH, Muller SA, Suman VJ, et al. Incidence of alopecia areata in Olmstead County, Minnesota, 1975 through 1989. Mayo Clin Proc. 1995;70:628-633.
- Borazan NH, Furst DE. Nonsteroidal anti-inflammatory drugs, disease-modifying antirheumatic drugs, nonopioid analgesics, & drugs used in gout. In: Katzung BG, Trevor AJ, eds. Basic & Clinical Pharmacology. 13th ed. McGraw-Hill; 2015:618-642.
- Shapiro J. Current treatment of alopecia areata. J Investig Dermatol Symp Proc. 2013;16:S42-S44.
- Shapiro J. Dermatologic therapy: alopecia areata update. Dermatol Ther. 2011;24:301.
- Craiglow BG, King BA. Killing two birds with one stone: oral tofacitinib reverses alopecia universalis in a patient with plaque psoriasis. J Invest Dermatol. 2014;134:2988-2990.
- Xing L, Dai Z, Jabbari A, et al. Alopecia areata is driven by cytotoxic T lymphocytes and is reversed by JAK inhibition. Nat Med. 2014;20:1043-1049.
- Pieri L, Guglielmelli P, Vannucchi AM. Ruxolitinib-induced reversal of alopecia universalis in a patient with essential thrombocythemia. Am J Hematol. 2015;90:82-83.
- Jabbari A, Dai Z, Xing L, et al. Reversal of alopecia areata following treatment with the JAK1/2 inhibitor baricitinib. EbioMedicine. 2015;2:351-355.
- Jabbari A, Nguyen N, Cerise JE, et al. Treatment of an alopecia areata patient with tofacitinib results in regrowth of hair and changes in serum and skin biomarkers. Exp Dermatol. 2016;25:642-643.
- Liu LY, Craiglow BG, Dai F, et al. Tofacitinib for the treatment of severe alopecia areata and variants: a study of 90 patients. J Am Acad Dermatol. 2017;76:22-28.
- Craiglow BG, Liu LY, King BA. Tofacitinib for the treatment of alopecia areata and variants in adolescents. J Am Acad Dermatol. 2017;76:29-32.
- Kennedy Crispin M, Ko JM, Craiglow BG, et al. Safety and efficacy of the JAK inhibitor tofacitinib citrate in patients with alopecia areata. JCI Insight. 2016;1:E89776.
- Iorizzo M, Tosti A. Emerging drugs for alopecia areata: JAK inhibitors. Expert Opin Emerg Drugs. 2018;23:77-81.
Practice Points
- Janus kinase inhibitors target one of the cellular pathogeneses of alopecia areata.
- Janus kinase inhibitors may be an option for patients who have exhausted other treatment modalities for alopecia.
Pediatric Primary Cutaneous Marginal Zone Lymphoma Treated With Doxycycline
Case Report
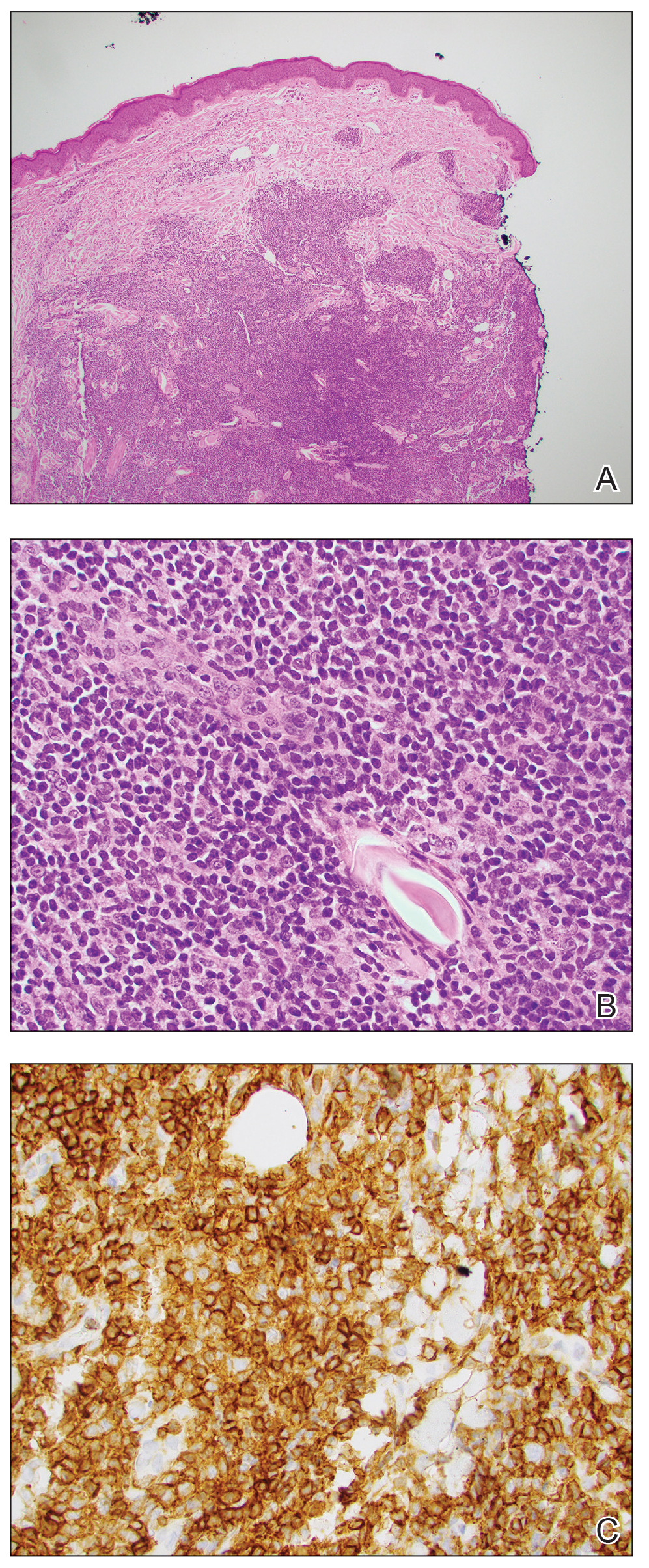
An otherwise healthy 13-year-old boy was referred to pediatric dermatology with multiple asymptomatic erythematous papules throughout the trunk and arms of 6 months’ duration. He denied fevers, night sweats, or weight loss. A punch biopsy revealed a dense atypical lymphoid infiltrate with follicular prominence extending periadnexally and perivascularly, which was most consistent with extranodal marginal zone lymphoma of mucosa-associated lymphoid tissue (Figures 1A and 1B). Cells were positive for Bcl-2, CD23, and CD20 (Figure 1C). Polymerase chain reaction analysis of the immunoglobulin heavy and κ chain gene rearrangements were positive, indicating the presence of a clonal B-cell expansion. The patient’s complete blood cell count, complete metabolic profile, serum lactate dehydrogenase, and erythrocyte sedimentation rate were within reference range. Lyme disease antibodies, Helicobacter pylori testing, thyroid function testing, thyroid antibodies, anti–Sjogren syndrome–related antigen A antibody, and anti–Sjogren syndrome–related antigen B were negative. Additionally, positron emission tomography (PET) with computed tomography (CT) revealed no abnormalities. He was diagnosed with stage T3b primary cutaneous marginal zone lymphoma (PCMZL) due to cutaneous involvement of 3 or more body regions.
The patient was started on clobetasol ointment 0.05% twice daily to the affected areas. After 2 months, he had progression of cutaneous disease, including increased number of lesions; erythema; and induration of lesions on the chest, back, and arms (Figure 2A) and was started on oral doxycycline 100 mg twice daily with subsequent notable improvement of the skin lesions at 2-week follow-up, including decreased erythema and induration of all lesions. He then received intralesional triamcinolone 20 mg/mL injections to 4 residual lesions; clobetasol ointment 0.05% twice daily was continued for the remaining lesions as needed for pruritus. He continued doxycycline for 4 months with further improvement of lesions (Figure 2B). Six months after discontinuing doxycycline, 2 small residual lesions remained on the left arm and back, but the patient did not develop any new or recurrent lesions.
Comment
Clinical Presentation—Primary cutaneous B-cell lymphomas include PCMZL, primary cutaneous follicle center lymphoma, and primary cutaneous large B-cell lymphoma. Primary cutaneous marginal zone lymphoma is an indolent extranodal B-cell lymphoma composed of small B cells, marginal zone cells, lymphoplasmacytoid cells, and mature plasma cells.1
Primary cutaneous marginal zone lymphoma typically presents in the fourth to sixth decades of life and is rare in children, with fewer than 40 cases in patients younger than 20 years.2 Amitay-Laish and colleagues2 reported 29 patients with pediatric PCMZL ranging in age from 1 to 19.5 years at diagnosis, with the majority of patients diagnosed after 10 years of age. Clinically, patients present with multifocal, erythematous to brown, dermal papules, plaques, and nodules most commonly distributed on the trunk and arms. A retrospective review of 11 pediatric patients with PCMZL over a median of 5.5 years demonstrated that the clinical presentation, histopathology, molecular findings, and prognosis of pediatric PCMZL appears similar to adult PCMZL.2 Cutaneous relapse is common, but extracutaneous spread is rare. The prognosis is excellent, with a disease-free survival rate of 93%.3
Diagnosis—The diagnosis of PCMZL requires histopathologic analysis of involved skin as well as exclusion of extracutaneous disease at the time of diagnosis during initial staging evaluation. Histologically there are nodular infiltrates of small lymphocytes in interfollicular compartments, reactive germinal centers, and clonality with monotypic immunoglobulin heavy chain genes.4 Laboratory workup should include complete blood cell count with differential, complete metabolic panel, and serum lactate dehydrogenase level. If lymphocytosis is present, flow cytometry of peripheral blood cells should be performed. Radiographic imaging with contrast-enhanced CT or PET/CT of the chest, abdomen, and pelvis should be performed for routine staging in most patients, with imaging of the neck recommended when cervical lymphadenopathy is detected.5 Patients with multifocal skin lesions should receive PET/CT to exclude systemic disease and assess lymph nodes. Bone marrow studies are not required for diagnosis.5,6
Associated Conditions—Systemic marginal zone lymphoma has been associated with autoimmune conditions, including Hashimoto thyroiditis and Sjögren syndrome; however, this association has not been shown in PCMZL and was not found in our patient.7,8 Borrelia-positive serology has been described in cases of PCMZL in Europe. The pathogenesis has been speculated to be due to chronic antigen stimulation related to the geographic distribution of Borrelia species.9 In endemic areas, Borrelia testing with serology or DNA testing of skin is recommended; however, there has been no strong correlation between Borrelia burgdorferi and PCMZL found in North America or Asia.9,10 Helicobacter pylori has been associated with gastric mucosal-associated lymphatic tissue lymphoma, which responds well to antibiotic therapy. However, an association between PCMZL and H pylori has not been well described.11
Management—Several treatment modalities have been attempted in patients with PCMZL with varying efficacy. Given the rarity of this disease, there is no standard therapy. Treatment options include radiation therapy, excision, topical steroids, intralesional steroids, intralesional rituximab, and antibiotics.2,12-14 Case reports of pediatric patients have demonstrated improvement with excision,15-19 intralesional steroids,20,21 intralesional rituximab,22 and clobetasol cream.23,24 In asymptomatic patients, watchful waiting often is employed given the overall indolent nature of PCMZL. Antibiotic therapy may be favored in Borrelia-positive cases. However, even in B burgdorferi–negative patients, there have been cases where there is response to antibiotics, particularly doxycycline.2,15,25 We elected for a trial of doxycycline in our patient based on these prior reports, along with the overall favorable side-effect profile of doxycycline for adolescents and our patient’s widespread cutaneous involvement.
Doxycycline is utilized in pediatric patients 8 years and older for numerous indications, including treatment of acne, Rocky Mountain spotted fever, and Lyme disease. Use of doxycycline in younger patients typically is avoided given the risk for dental enamel hypoplasia, tooth discoloration, and possible delays in skeletal development. Originally utilized for its antibacterial effects as an intracellular inhibitor of protein synthesis, doxycycline has been repurposed for oncologic therapies. It has been shown to have cytotoxic and antiproliferative properties in various cancer cells and also may inhibit leukemic cell migration.26 In PCMZL, doxycycline initially was utilized in Borrelia-positive patients in Europe and found to improve disease clearance.27 In patients without Borrelia infection, doxycycline is thought to enhance apoptosis through caspase-3 activation along with p53 and Bax upregulation.28
Intralesional triamcinolone alone may not be feasible in pediatric PCMZL patients because of widespread involvement, and doxycycline may be considered as a treatment option. Multiple low-risk treatment modalities may be used in conjunction to clear disease in pediatric patients, as demonstrated in our case.
Acknowledgment—We thank Ali Nael Amzajerdi, MD (Orange, California), for his contributions to the pathologic imaging in this report.
- Willemze R, Cerroni L, Kempf W, et al. The 2018 update of the WHO-EORTC classification for primary cutaneous lymphomas. Blood. 2019;133:1703-1714.
- Amitay-Laish I, Tavallaee M, Kim J, et al. Paediatric primary cutaneous marginal zone B-cell lymphoma: does it differ from its adult counterpart? Br J Dermatol. 2017;176:1010-1020.
- Servitje O, Muniesa C, Benavente Y, et al. Primary cutaneous marginal zone B-cell lymphoma: response to treatment and disease-free survival in a series of 137 patients. J Am Acad Dermatol. 2013;69:357-365.
- Vitiello P, Sica A, Ronchi A, et al. Primary cutaneous B-cell lymphomas: an update. Front Oncol. 2020;10:651.
- Tadiotto Cicogna G, Ferranti M, Alaibac M. Diagnostic workup of primary cutaneous B cell lymphomas: a clinician’s approach. Front Oncol. 2020;10:988.
- Willemze R, Hodak E, Zinzani PL, et al. Primary cutaneous lymphomas: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2013;24:149-154.
- Pereira FO, Graf H, Nomura LM, et al. Concomitant presentation of Hashimoto’s thyroiditis and maltoma of the thyroid in a twenty-year-old man with a rapidly growing mass in the neck. Thyroid. 2000;10:833-835.
- Ekström Smedby K, Vajdic CM, Falster M, et al. Autoimmune disorders and risk of non-Hodgkin lymphoma subtypes: a pooled analysis within the InterLymph Consortium. Blood. 2008;111:4029-4038.
- Slater DN. Borrelia burgdorferi-associated primary cutaneous B-cell lymphoma. Histopathology. 2001;38:73-77.
- Wood GS, Kamath NV, Guitart J, et al. Absence of Borrelia burgdorferi DNA in cutaneous B-cell lymphomas from the United States. J Cutan Pathol. 2001;28:502-507.
- Dalle S, Thomas L, Balme B, et al. Primary cutaneous marginal zone lymphoma. Crit Rev Oncol Hematol. 2010;74:156-162.
- Senff NJ, Noordijk EM, Kim YH, et al. European Organization for Research and Treatment of Cancer and International Society for Cutaneous Lymphoma consensus recommendations for the management of cutaneous B-cell lymphomas. Blood. 2008;112:1600-1609.
- Hamilton SN, Wai ES, Tan K, et al. Treatment and outcomes in patients with primary cutaneous B-cell lymphoma: the BC Cancer Agency experience. Int J Radiat Oncol Biol Phys. 2013;87:719-725.
- Peñate Y, Hernández-Machín B, Pérez-Méndez LI, et al. Intralesional rituximab in the treatment of indolent primary cutaneous B-cell lymphomas: an epidemiological observational multicentre study. The Spanish Working Group on Cutaneous Lymphoma. Br J Dermatol. 2012;167:174-179.
- Kempf W, Kazakov DV, Buechner SA, et al. Primary cutaneous marginal zone lymphoma in children: a report of 3 cases and review of the literature. Am J Dermatopathol. 2014;36:661-666.
- Ghatalia P, Porter J, Wroblewski D, et al. Primary cutaneous marginal zone lymphoma associated with juxta-articular fibrotic nodules in a teenager. J Cutan Pathol. 2013;40:477-484.
- Dargent JL, Devalck C, De Mey A, et al. Primary cutaneous marginal zone B-cell lymphoma of MALT type in a child. Pediatr Dev Pathol. 2006;9:468-473.
- Sroa N, Magro CM. Pediatric primary cutaneous marginal zone lymphoma: in association with chronic antihistamine use. J Cutan Pathol. 2006;33(suppl 2):1-5.
- Zambrano E, Mejıa-Mejıa O, Bifulco C, et al. Extranodal marginal zone B-cell lymphoma/maltoma of the lip in a child: case report and review of cutaneous lymphoid proliferations in childhood. Int J Surg Pathol. 2006;14:163-169.
- Kollipara R, Hans A, Hall J, et al. A case report of primary cutaneous marginal zone lymphoma treated with intralesional steroids. Dermatol Online J. 2015;21:13030/qt9s15929m.
- Skaljic M, Cotton CH, Reilly AF, et al. Complete resolution of primary cutaneous marginal zone B-cell lymphoma on the cheek of a 7-year-old boy with intralesional triamcinolone and tincture of time. Pediatr Dermatol. 2020;37:228-229.
- Park MY, Jung HJ, Park JE, et al. Pediatric primary cutaneous marginal zone B-cell lymphoma treated with intralesional rituximab. Eur J Dermatol. 2010;20:533-534.
- Amitay-Laish I, Feinmesser M, Ben-Amitai D, et al. Juvenile onset of primary low-grade cutaneous B-cell lymphoma. Br J Dermatol. 2009;161:140-147.
- Sharon V, Mecca PS, Steinherz PG, et al. Two pediatric cases of primary cutaneous B-cell lymphoma and review of the literature. Pediatr Dermatol. 2009;26:34-39.
- Jothishankar B, Di Raimondo C, Mueller L, et al. Primary cutaneous marginal zone lymphoma treated with doxycycline in a pediatric patient. Pediatr Dermatol. 2020;37:759-761.
- Markowska A, Kaysiewicz J, Markowska J, et al. Doxycycline, salinomycin, monensin and ivermectin repositioned as cancer drugs. Bioorg Med Chem Lett. 2019;29:1549-1554.
- Kutting B, Bonsmann G, Metze D, et al. Borrelia burgdorferi-associated primary cutaneous B-cell lymphoma: complete clearing of skin lesions after antibiotic pulse therapy or intralesional injection of interferon alfa-2a. J Am Acad Dermatol. 1997;36:311-314.
- Protasoni M, Kroon AM, Taanman JW. Mitochondria as oncotarget: a comparison between the tetracycline analogs doxycycline and COL-3. Oncotarget. 2018;9:33818-33831.
Case Report
An otherwise healthy 13-year-old boy was referred to pediatric dermatology with multiple asymptomatic erythematous papules throughout the trunk and arms of 6 months’ duration. He denied fevers, night sweats, or weight loss. A punch biopsy revealed a dense atypical lymphoid infiltrate with follicular prominence extending periadnexally and perivascularly, which was most consistent with extranodal marginal zone lymphoma of mucosa-associated lymphoid tissue (Figures 1A and 1B). Cells were positive for Bcl-2, CD23, and CD20 (Figure 1C). Polymerase chain reaction analysis of the immunoglobulin heavy and κ chain gene rearrangements were positive, indicating the presence of a clonal B-cell expansion. The patient’s complete blood cell count, complete metabolic profile, serum lactate dehydrogenase, and erythrocyte sedimentation rate were within reference range. Lyme disease antibodies, Helicobacter pylori testing, thyroid function testing, thyroid antibodies, anti–Sjogren syndrome–related antigen A antibody, and anti–Sjogren syndrome–related antigen B were negative. Additionally, positron emission tomography (PET) with computed tomography (CT) revealed no abnormalities. He was diagnosed with stage T3b primary cutaneous marginal zone lymphoma (PCMZL) due to cutaneous involvement of 3 or more body regions.

The patient was started on clobetasol ointment 0.05% twice daily to the affected areas. After 2 months, he had progression of cutaneous disease, including increased number of lesions; erythema; and induration of lesions on the chest, back, and arms (Figure 2A) and was started on oral doxycycline 100 mg twice daily with subsequent notable improvement of the skin lesions at 2-week follow-up, including decreased erythema and induration of all lesions. He then received intralesional triamcinolone 20 mg/mL injections to 4 residual lesions; clobetasol ointment 0.05% twice daily was continued for the remaining lesions as needed for pruritus. He continued doxycycline for 4 months with further improvement of lesions (Figure 2B). Six months after discontinuing doxycycline, 2 small residual lesions remained on the left arm and back, but the patient did not develop any new or recurrent lesions.

Comment
Clinical Presentation—Primary cutaneous B-cell lymphomas include PCMZL, primary cutaneous follicle center lymphoma, and primary cutaneous large B-cell lymphoma. Primary cutaneous marginal zone lymphoma is an indolent extranodal B-cell lymphoma composed of small B cells, marginal zone cells, lymphoplasmacytoid cells, and mature plasma cells.1
Primary cutaneous marginal zone lymphoma typically presents in the fourth to sixth decades of life and is rare in children, with fewer than 40 cases in patients younger than 20 years.2 Amitay-Laish and colleagues2 reported 29 patients with pediatric PCMZL ranging in age from 1 to 19.5 years at diagnosis, with the majority of patients diagnosed after 10 years of age. Clinically, patients present with multifocal, erythematous to brown, dermal papules, plaques, and nodules most commonly distributed on the trunk and arms. A retrospective review of 11 pediatric patients with PCMZL over a median of 5.5 years demonstrated that the clinical presentation, histopathology, molecular findings, and prognosis of pediatric PCMZL appears similar to adult PCMZL.2 Cutaneous relapse is common, but extracutaneous spread is rare. The prognosis is excellent, with a disease-free survival rate of 93%.3
Diagnosis—The diagnosis of PCMZL requires histopathologic analysis of involved skin as well as exclusion of extracutaneous disease at the time of diagnosis during initial staging evaluation. Histologically there are nodular infiltrates of small lymphocytes in interfollicular compartments, reactive germinal centers, and clonality with monotypic immunoglobulin heavy chain genes.4 Laboratory workup should include complete blood cell count with differential, complete metabolic panel, and serum lactate dehydrogenase level. If lymphocytosis is present, flow cytometry of peripheral blood cells should be performed. Radiographic imaging with contrast-enhanced CT or PET/CT of the chest, abdomen, and pelvis should be performed for routine staging in most patients, with imaging of the neck recommended when cervical lymphadenopathy is detected.5 Patients with multifocal skin lesions should receive PET/CT to exclude systemic disease and assess lymph nodes. Bone marrow studies are not required for diagnosis.5,6
Associated Conditions—Systemic marginal zone lymphoma has been associated with autoimmune conditions, including Hashimoto thyroiditis and Sjögren syndrome; however, this association has not been shown in PCMZL and was not found in our patient.7,8 Borrelia-positive serology has been described in cases of PCMZL in Europe. The pathogenesis has been speculated to be due to chronic antigen stimulation related to the geographic distribution of Borrelia species.9 In endemic areas, Borrelia testing with serology or DNA testing of skin is recommended; however, there has been no strong correlation between Borrelia burgdorferi and PCMZL found in North America or Asia.9,10 Helicobacter pylori has been associated with gastric mucosal-associated lymphatic tissue lymphoma, which responds well to antibiotic therapy. However, an association between PCMZL and H pylori has not been well described.11
Management—Several treatment modalities have been attempted in patients with PCMZL with varying efficacy. Given the rarity of this disease, there is no standard therapy. Treatment options include radiation therapy, excision, topical steroids, intralesional steroids, intralesional rituximab, and antibiotics.2,12-14 Case reports of pediatric patients have demonstrated improvement with excision,15-19 intralesional steroids,20,21 intralesional rituximab,22 and clobetasol cream.23,24 In asymptomatic patients, watchful waiting often is employed given the overall indolent nature of PCMZL. Antibiotic therapy may be favored in Borrelia-positive cases. However, even in B burgdorferi–negative patients, there have been cases where there is response to antibiotics, particularly doxycycline.2,15,25 We elected for a trial of doxycycline in our patient based on these prior reports, along with the overall favorable side-effect profile of doxycycline for adolescents and our patient’s widespread cutaneous involvement.
Doxycycline is utilized in pediatric patients 8 years and older for numerous indications, including treatment of acne, Rocky Mountain spotted fever, and Lyme disease. Use of doxycycline in younger patients typically is avoided given the risk for dental enamel hypoplasia, tooth discoloration, and possible delays in skeletal development. Originally utilized for its antibacterial effects as an intracellular inhibitor of protein synthesis, doxycycline has been repurposed for oncologic therapies. It has been shown to have cytotoxic and antiproliferative properties in various cancer cells and also may inhibit leukemic cell migration.26 In PCMZL, doxycycline initially was utilized in Borrelia-positive patients in Europe and found to improve disease clearance.27 In patients without Borrelia infection, doxycycline is thought to enhance apoptosis through caspase-3 activation along with p53 and Bax upregulation.28
Intralesional triamcinolone alone may not be feasible in pediatric PCMZL patients because of widespread involvement, and doxycycline may be considered as a treatment option. Multiple low-risk treatment modalities may be used in conjunction to clear disease in pediatric patients, as demonstrated in our case.
Acknowledgment—We thank Ali Nael Amzajerdi, MD (Orange, California), for his contributions to the pathologic imaging in this report.
Case Report
An otherwise healthy 13-year-old boy was referred to pediatric dermatology with multiple asymptomatic erythematous papules throughout the trunk and arms of 6 months’ duration. He denied fevers, night sweats, or weight loss. A punch biopsy revealed a dense atypical lymphoid infiltrate with follicular prominence extending periadnexally and perivascularly, which was most consistent with extranodal marginal zone lymphoma of mucosa-associated lymphoid tissue (Figures 1A and 1B). Cells were positive for Bcl-2, CD23, and CD20 (Figure 1C). Polymerase chain reaction analysis of the immunoglobulin heavy and κ chain gene rearrangements were positive, indicating the presence of a clonal B-cell expansion. The patient’s complete blood cell count, complete metabolic profile, serum lactate dehydrogenase, and erythrocyte sedimentation rate were within reference range. Lyme disease antibodies, Helicobacter pylori testing, thyroid function testing, thyroid antibodies, anti–Sjogren syndrome–related antigen A antibody, and anti–Sjogren syndrome–related antigen B were negative. Additionally, positron emission tomography (PET) with computed tomography (CT) revealed no abnormalities. He was diagnosed with stage T3b primary cutaneous marginal zone lymphoma (PCMZL) due to cutaneous involvement of 3 or more body regions.

The patient was started on clobetasol ointment 0.05% twice daily to the affected areas. After 2 months, he had progression of cutaneous disease, including increased number of lesions; erythema; and induration of lesions on the chest, back, and arms (Figure 2A) and was started on oral doxycycline 100 mg twice daily with subsequent notable improvement of the skin lesions at 2-week follow-up, including decreased erythema and induration of all lesions. He then received intralesional triamcinolone 20 mg/mL injections to 4 residual lesions; clobetasol ointment 0.05% twice daily was continued for the remaining lesions as needed for pruritus. He continued doxycycline for 4 months with further improvement of lesions (Figure 2B). Six months after discontinuing doxycycline, 2 small residual lesions remained on the left arm and back, but the patient did not develop any new or recurrent lesions.

Comment
Clinical Presentation—Primary cutaneous B-cell lymphomas include PCMZL, primary cutaneous follicle center lymphoma, and primary cutaneous large B-cell lymphoma. Primary cutaneous marginal zone lymphoma is an indolent extranodal B-cell lymphoma composed of small B cells, marginal zone cells, lymphoplasmacytoid cells, and mature plasma cells.1
Primary cutaneous marginal zone lymphoma typically presents in the fourth to sixth decades of life and is rare in children, with fewer than 40 cases in patients younger than 20 years.2 Amitay-Laish and colleagues2 reported 29 patients with pediatric PCMZL ranging in age from 1 to 19.5 years at diagnosis, with the majority of patients diagnosed after 10 years of age. Clinically, patients present with multifocal, erythematous to brown, dermal papules, plaques, and nodules most commonly distributed on the trunk and arms. A retrospective review of 11 pediatric patients with PCMZL over a median of 5.5 years demonstrated that the clinical presentation, histopathology, molecular findings, and prognosis of pediatric PCMZL appears similar to adult PCMZL.2 Cutaneous relapse is common, but extracutaneous spread is rare. The prognosis is excellent, with a disease-free survival rate of 93%.3
Diagnosis—The diagnosis of PCMZL requires histopathologic analysis of involved skin as well as exclusion of extracutaneous disease at the time of diagnosis during initial staging evaluation. Histologically there are nodular infiltrates of small lymphocytes in interfollicular compartments, reactive germinal centers, and clonality with monotypic immunoglobulin heavy chain genes.4 Laboratory workup should include complete blood cell count with differential, complete metabolic panel, and serum lactate dehydrogenase level. If lymphocytosis is present, flow cytometry of peripheral blood cells should be performed. Radiographic imaging with contrast-enhanced CT or PET/CT of the chest, abdomen, and pelvis should be performed for routine staging in most patients, with imaging of the neck recommended when cervical lymphadenopathy is detected.5 Patients with multifocal skin lesions should receive PET/CT to exclude systemic disease and assess lymph nodes. Bone marrow studies are not required for diagnosis.5,6
Associated Conditions—Systemic marginal zone lymphoma has been associated with autoimmune conditions, including Hashimoto thyroiditis and Sjögren syndrome; however, this association has not been shown in PCMZL and was not found in our patient.7,8 Borrelia-positive serology has been described in cases of PCMZL in Europe. The pathogenesis has been speculated to be due to chronic antigen stimulation related to the geographic distribution of Borrelia species.9 In endemic areas, Borrelia testing with serology or DNA testing of skin is recommended; however, there has been no strong correlation between Borrelia burgdorferi and PCMZL found in North America or Asia.9,10 Helicobacter pylori has been associated with gastric mucosal-associated lymphatic tissue lymphoma, which responds well to antibiotic therapy. However, an association between PCMZL and H pylori has not been well described.11
Management—Several treatment modalities have been attempted in patients with PCMZL with varying efficacy. Given the rarity of this disease, there is no standard therapy. Treatment options include radiation therapy, excision, topical steroids, intralesional steroids, intralesional rituximab, and antibiotics.2,12-14 Case reports of pediatric patients have demonstrated improvement with excision,15-19 intralesional steroids,20,21 intralesional rituximab,22 and clobetasol cream.23,24 In asymptomatic patients, watchful waiting often is employed given the overall indolent nature of PCMZL. Antibiotic therapy may be favored in Borrelia-positive cases. However, even in B burgdorferi–negative patients, there have been cases where there is response to antibiotics, particularly doxycycline.2,15,25 We elected for a trial of doxycycline in our patient based on these prior reports, along with the overall favorable side-effect profile of doxycycline for adolescents and our patient’s widespread cutaneous involvement.
Doxycycline is utilized in pediatric patients 8 years and older for numerous indications, including treatment of acne, Rocky Mountain spotted fever, and Lyme disease. Use of doxycycline in younger patients typically is avoided given the risk for dental enamel hypoplasia, tooth discoloration, and possible delays in skeletal development. Originally utilized for its antibacterial effects as an intracellular inhibitor of protein synthesis, doxycycline has been repurposed for oncologic therapies. It has been shown to have cytotoxic and antiproliferative properties in various cancer cells and also may inhibit leukemic cell migration.26 In PCMZL, doxycycline initially was utilized in Borrelia-positive patients in Europe and found to improve disease clearance.27 In patients without Borrelia infection, doxycycline is thought to enhance apoptosis through caspase-3 activation along with p53 and Bax upregulation.28
Intralesional triamcinolone alone may not be feasible in pediatric PCMZL patients because of widespread involvement, and doxycycline may be considered as a treatment option. Multiple low-risk treatment modalities may be used in conjunction to clear disease in pediatric patients, as demonstrated in our case.
Acknowledgment—We thank Ali Nael Amzajerdi, MD (Orange, California), for his contributions to the pathologic imaging in this report.
- Willemze R, Cerroni L, Kempf W, et al. The 2018 update of the WHO-EORTC classification for primary cutaneous lymphomas. Blood. 2019;133:1703-1714.
- Amitay-Laish I, Tavallaee M, Kim J, et al. Paediatric primary cutaneous marginal zone B-cell lymphoma: does it differ from its adult counterpart? Br J Dermatol. 2017;176:1010-1020.
- Servitje O, Muniesa C, Benavente Y, et al. Primary cutaneous marginal zone B-cell lymphoma: response to treatment and disease-free survival in a series of 137 patients. J Am Acad Dermatol. 2013;69:357-365.
- Vitiello P, Sica A, Ronchi A, et al. Primary cutaneous B-cell lymphomas: an update. Front Oncol. 2020;10:651.
- Tadiotto Cicogna G, Ferranti M, Alaibac M. Diagnostic workup of primary cutaneous B cell lymphomas: a clinician’s approach. Front Oncol. 2020;10:988.
- Willemze R, Hodak E, Zinzani PL, et al. Primary cutaneous lymphomas: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2013;24:149-154.
- Pereira FO, Graf H, Nomura LM, et al. Concomitant presentation of Hashimoto’s thyroiditis and maltoma of the thyroid in a twenty-year-old man with a rapidly growing mass in the neck. Thyroid. 2000;10:833-835.
- Ekström Smedby K, Vajdic CM, Falster M, et al. Autoimmune disorders and risk of non-Hodgkin lymphoma subtypes: a pooled analysis within the InterLymph Consortium. Blood. 2008;111:4029-4038.
- Slater DN. Borrelia burgdorferi-associated primary cutaneous B-cell lymphoma. Histopathology. 2001;38:73-77.
- Wood GS, Kamath NV, Guitart J, et al. Absence of Borrelia burgdorferi DNA in cutaneous B-cell lymphomas from the United States. J Cutan Pathol. 2001;28:502-507.
- Dalle S, Thomas L, Balme B, et al. Primary cutaneous marginal zone lymphoma. Crit Rev Oncol Hematol. 2010;74:156-162.
- Senff NJ, Noordijk EM, Kim YH, et al. European Organization for Research and Treatment of Cancer and International Society for Cutaneous Lymphoma consensus recommendations for the management of cutaneous B-cell lymphomas. Blood. 2008;112:1600-1609.
- Hamilton SN, Wai ES, Tan K, et al. Treatment and outcomes in patients with primary cutaneous B-cell lymphoma: the BC Cancer Agency experience. Int J Radiat Oncol Biol Phys. 2013;87:719-725.
- Peñate Y, Hernández-Machín B, Pérez-Méndez LI, et al. Intralesional rituximab in the treatment of indolent primary cutaneous B-cell lymphomas: an epidemiological observational multicentre study. The Spanish Working Group on Cutaneous Lymphoma. Br J Dermatol. 2012;167:174-179.
- Kempf W, Kazakov DV, Buechner SA, et al. Primary cutaneous marginal zone lymphoma in children: a report of 3 cases and review of the literature. Am J Dermatopathol. 2014;36:661-666.
- Ghatalia P, Porter J, Wroblewski D, et al. Primary cutaneous marginal zone lymphoma associated with juxta-articular fibrotic nodules in a teenager. J Cutan Pathol. 2013;40:477-484.
- Dargent JL, Devalck C, De Mey A, et al. Primary cutaneous marginal zone B-cell lymphoma of MALT type in a child. Pediatr Dev Pathol. 2006;9:468-473.
- Sroa N, Magro CM. Pediatric primary cutaneous marginal zone lymphoma: in association with chronic antihistamine use. J Cutan Pathol. 2006;33(suppl 2):1-5.
- Zambrano E, Mejıa-Mejıa O, Bifulco C, et al. Extranodal marginal zone B-cell lymphoma/maltoma of the lip in a child: case report and review of cutaneous lymphoid proliferations in childhood. Int J Surg Pathol. 2006;14:163-169.
- Kollipara R, Hans A, Hall J, et al. A case report of primary cutaneous marginal zone lymphoma treated with intralesional steroids. Dermatol Online J. 2015;21:13030/qt9s15929m.
- Skaljic M, Cotton CH, Reilly AF, et al. Complete resolution of primary cutaneous marginal zone B-cell lymphoma on the cheek of a 7-year-old boy with intralesional triamcinolone and tincture of time. Pediatr Dermatol. 2020;37:228-229.
- Park MY, Jung HJ, Park JE, et al. Pediatric primary cutaneous marginal zone B-cell lymphoma treated with intralesional rituximab. Eur J Dermatol. 2010;20:533-534.
- Amitay-Laish I, Feinmesser M, Ben-Amitai D, et al. Juvenile onset of primary low-grade cutaneous B-cell lymphoma. Br J Dermatol. 2009;161:140-147.
- Sharon V, Mecca PS, Steinherz PG, et al. Two pediatric cases of primary cutaneous B-cell lymphoma and review of the literature. Pediatr Dermatol. 2009;26:34-39.
- Jothishankar B, Di Raimondo C, Mueller L, et al. Primary cutaneous marginal zone lymphoma treated with doxycycline in a pediatric patient. Pediatr Dermatol. 2020;37:759-761.
- Markowska A, Kaysiewicz J, Markowska J, et al. Doxycycline, salinomycin, monensin and ivermectin repositioned as cancer drugs. Bioorg Med Chem Lett. 2019;29:1549-1554.
- Kutting B, Bonsmann G, Metze D, et al. Borrelia burgdorferi-associated primary cutaneous B-cell lymphoma: complete clearing of skin lesions after antibiotic pulse therapy or intralesional injection of interferon alfa-2a. J Am Acad Dermatol. 1997;36:311-314.
- Protasoni M, Kroon AM, Taanman JW. Mitochondria as oncotarget: a comparison between the tetracycline analogs doxycycline and COL-3. Oncotarget. 2018;9:33818-33831.
- Willemze R, Cerroni L, Kempf W, et al. The 2018 update of the WHO-EORTC classification for primary cutaneous lymphomas. Blood. 2019;133:1703-1714.
- Amitay-Laish I, Tavallaee M, Kim J, et al. Paediatric primary cutaneous marginal zone B-cell lymphoma: does it differ from its adult counterpart? Br J Dermatol. 2017;176:1010-1020.
- Servitje O, Muniesa C, Benavente Y, et al. Primary cutaneous marginal zone B-cell lymphoma: response to treatment and disease-free survival in a series of 137 patients. J Am Acad Dermatol. 2013;69:357-365.
- Vitiello P, Sica A, Ronchi A, et al. Primary cutaneous B-cell lymphomas: an update. Front Oncol. 2020;10:651.
- Tadiotto Cicogna G, Ferranti M, Alaibac M. Diagnostic workup of primary cutaneous B cell lymphomas: a clinician’s approach. Front Oncol. 2020;10:988.
- Willemze R, Hodak E, Zinzani PL, et al. Primary cutaneous lymphomas: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2013;24:149-154.
- Pereira FO, Graf H, Nomura LM, et al. Concomitant presentation of Hashimoto’s thyroiditis and maltoma of the thyroid in a twenty-year-old man with a rapidly growing mass in the neck. Thyroid. 2000;10:833-835.
- Ekström Smedby K, Vajdic CM, Falster M, et al. Autoimmune disorders and risk of non-Hodgkin lymphoma subtypes: a pooled analysis within the InterLymph Consortium. Blood. 2008;111:4029-4038.
- Slater DN. Borrelia burgdorferi-associated primary cutaneous B-cell lymphoma. Histopathology. 2001;38:73-77.
- Wood GS, Kamath NV, Guitart J, et al. Absence of Borrelia burgdorferi DNA in cutaneous B-cell lymphomas from the United States. J Cutan Pathol. 2001;28:502-507.
- Dalle S, Thomas L, Balme B, et al. Primary cutaneous marginal zone lymphoma. Crit Rev Oncol Hematol. 2010;74:156-162.
- Senff NJ, Noordijk EM, Kim YH, et al. European Organization for Research and Treatment of Cancer and International Society for Cutaneous Lymphoma consensus recommendations for the management of cutaneous B-cell lymphomas. Blood. 2008;112:1600-1609.
- Hamilton SN, Wai ES, Tan K, et al. Treatment and outcomes in patients with primary cutaneous B-cell lymphoma: the BC Cancer Agency experience. Int J Radiat Oncol Biol Phys. 2013;87:719-725.
- Peñate Y, Hernández-Machín B, Pérez-Méndez LI, et al. Intralesional rituximab in the treatment of indolent primary cutaneous B-cell lymphomas: an epidemiological observational multicentre study. The Spanish Working Group on Cutaneous Lymphoma. Br J Dermatol. 2012;167:174-179.
- Kempf W, Kazakov DV, Buechner SA, et al. Primary cutaneous marginal zone lymphoma in children: a report of 3 cases and review of the literature. Am J Dermatopathol. 2014;36:661-666.
- Ghatalia P, Porter J, Wroblewski D, et al. Primary cutaneous marginal zone lymphoma associated with juxta-articular fibrotic nodules in a teenager. J Cutan Pathol. 2013;40:477-484.
- Dargent JL, Devalck C, De Mey A, et al. Primary cutaneous marginal zone B-cell lymphoma of MALT type in a child. Pediatr Dev Pathol. 2006;9:468-473.
- Sroa N, Magro CM. Pediatric primary cutaneous marginal zone lymphoma: in association with chronic antihistamine use. J Cutan Pathol. 2006;33(suppl 2):1-5.
- Zambrano E, Mejıa-Mejıa O, Bifulco C, et al. Extranodal marginal zone B-cell lymphoma/maltoma of the lip in a child: case report and review of cutaneous lymphoid proliferations in childhood. Int J Surg Pathol. 2006;14:163-169.
- Kollipara R, Hans A, Hall J, et al. A case report of primary cutaneous marginal zone lymphoma treated with intralesional steroids. Dermatol Online J. 2015;21:13030/qt9s15929m.
- Skaljic M, Cotton CH, Reilly AF, et al. Complete resolution of primary cutaneous marginal zone B-cell lymphoma on the cheek of a 7-year-old boy with intralesional triamcinolone and tincture of time. Pediatr Dermatol. 2020;37:228-229.
- Park MY, Jung HJ, Park JE, et al. Pediatric primary cutaneous marginal zone B-cell lymphoma treated with intralesional rituximab. Eur J Dermatol. 2010;20:533-534.
- Amitay-Laish I, Feinmesser M, Ben-Amitai D, et al. Juvenile onset of primary low-grade cutaneous B-cell lymphoma. Br J Dermatol. 2009;161:140-147.
- Sharon V, Mecca PS, Steinherz PG, et al. Two pediatric cases of primary cutaneous B-cell lymphoma and review of the literature. Pediatr Dermatol. 2009;26:34-39.
- Jothishankar B, Di Raimondo C, Mueller L, et al. Primary cutaneous marginal zone lymphoma treated with doxycycline in a pediatric patient. Pediatr Dermatol. 2020;37:759-761.
- Markowska A, Kaysiewicz J, Markowska J, et al. Doxycycline, salinomycin, monensin and ivermectin repositioned as cancer drugs. Bioorg Med Chem Lett. 2019;29:1549-1554.
- Kutting B, Bonsmann G, Metze D, et al. Borrelia burgdorferi-associated primary cutaneous B-cell lymphoma: complete clearing of skin lesions after antibiotic pulse therapy or intralesional injection of interferon alfa-2a. J Am Acad Dermatol. 1997;36:311-314.
- Protasoni M, Kroon AM, Taanman JW. Mitochondria as oncotarget: a comparison between the tetracycline analogs doxycycline and COL-3. Oncotarget. 2018;9:33818-33831.
Practice Points
- When skin biopsy reveals marginal zone lymphoma, laboratory workup should include a complete blood cell count, chemistry, and serum lactate dehydrogenase levels. If lymphocytosis is present, flow cytometry of peripheral blood cells should be performed.
- For patients with multifocal skin lesions, positive emission tomography with computed tomography is utilized to exclude systemic disease and assess lymph node involvement.
- Treatments for primary cutaneous marginal zone lymphoma include excision, topical steroids, intralesional steroids, intralesional rituximab, radiation therapy, and antibiotics.
- Doxycycline can be considered as a treatment option for pediatric patients with widespread cutaneous involvement.
Case Q: How soon after taking emergency contraception can a patient begin hormonal contraception?
Individuals spend close to half of their lives preventing, or planning for, pregnancy. As such, contraception plays a major role in patient-provider interactions. Contraception counseling and management is a common scenario encountered in the general gynecologist’s practice. Luckily, we have two evidence-based guidelines developed by the US Centers for Disease Control and Prevention (CDC) that support the provision of contraceptive care:
- US Medical Eligibility for Contraceptive Use (US-MEC),1 which provides guidance on which patients can safely use a method
- US Selected Practice Recommendations for Contraceptive Use (US-SPR),2 which provides method-specific guidance on how to use a method (including how to: initiate or start a method; manage adherence issues, such as a missed pill, etc; and manage common issues like breakthrough bleeding). Both of these guidelines are updated routinely and are publicly available online or for free, through smartphone applications.
While most contraceptive care is straightforward, there are circumstances that require additional consideration. In this 3-part series we review 3 clinical cases, existing evidence to guide management decisions, and our recommendations. In part 1, we focus on restarting hormonal contraception after ulipristal acetate administration. In parts 2 and 3, we will discuss removal of a nonpalpable contraceptive implant and the consideration of a levonorgestrel-releasing intrauterine device (LNG-IUD) for emergency contraception.
- After using ulipristal acetate for emergency contraception, advise patients to wait at least 5 days to initiate hormonal contraception and about the importance of abstaining or using a back-up method for another 7 days with the start of their hormonal contraceptive method
CASE Meeting emergency and follow-up contraception needs
A 27-year-old woman (G0) presents to you after having unprotected intercourse 4 days ago. She does not formally track her menstrual cycles and is unsure when her last menstrual period was. She is not using contraception but is interested in starting a method. After counseling, she elects to take a dose of oral ulipristal acetate (UPA; Ella) now for emergency contraception and would like to start a combined oral contraceptive (COC) pill moving forward.
How soon after taking UPA should you tell her to start the combined hormonal pill?
Effectiveness of hormonal contraception following UPA
UPA does not appear to decrease the efficacy of COCs when started around the same time. However, immediately starting a hormonal contraceptive can decrease the effectiveness of UPA, and as such, it is recommended to take UPA and then abstain or use a backup method for 7 days before initiating a hormonal contraceptive method.1 By obtaining some additional information from your patient and with the use of shared decision making, though, your patient may be able to start their contraceptive method earlier than 5 days after UPA.
What is UPA
UPA is a progesterone receptor modulator used for emergency contraception intenhded to prevent pregnancy after unprotected intercourse or contraceptive failure.3 It works by delaying follicular rupture at least 5 days, if taken before the peak of the luteinizing hormone (LH) surge. If taken after that timeframe, it does not work. Since UPA competes for the progesterone receptor, there is a concern that the effectiveness of UPA may be decreased if a progestin-containing form of contraception is started immediately after taking UPA, or vice versa.4 Several studies have now specifically looked at the interaction between UPA and progestin-containing contraceptives, including at how UPA is impacted by the contraceptive method, and conversely, how the contraceptive method is impacted by UPA.5-8
Data on types of hormonal contraception. Brache and colleagues demonstrated that UPA users who started a desogestrel progestin-only pill (DSG POP) the next day had higher rates of ovulation within 5 days of taking UPA (45%), compared with those who the next day started a placebo pill (3%).6 This type of progestin-only pill is not available in the United States.
A study by Edelman and colleagues demonstrated similar findings in those starting a COC pill containing estrogen and progestin. When taking a COC two days after UPA use, more participants had evidence of follicular rupture in less than 5 days.5 It should be noted that these studies focused on ovulation, which—while necessary for conception to occur—is a surrogate biomarker for pregnancy risk. Additional studies have looked at the impact of UPA on the COC and have not found that UPA impacts ovulation suppression of the COC with its initiation or use.8
Considering unprotected intercourse and UPA timing. Of course, the risk of pregnancy is reliant on cycle timing plus the presence of viable sperm in the reproductive tract. Sperm have been shown to only be viable in the reproductive tract for 5 days, which could result in fertilization and subsequent pregnancy. Longevity of an egg is much shorter, at 12 to 24 hours after ovulation. For this patient, her exposure was 4 days ago, but sperm are only viable for approximately 5 days—she could consider taking the UPA now and then starting a COC earlier than 5 days since she only needs an extra day or two of protection from the UPA from the sperm in her reproductive tract. Your patient’s involvement in this decision making is paramount, as only they can prioritize their desire to avoid pregnancy from their recent act of unprotected intercourse versus their immediate needs for starting their method of contraception. It is important that individuals abstain from sexual activity or use an additional back-up method during the first 7 days of starting their method of contraception.
Continue to: Counseling considerations for the case patient...
Counseling considerations for the case patient
For a patient planning to start or resume a hormonal contraceptive method after taking UPA, the waiting period recommended by the CDC (5 days) is most beneficial for patients who are uncertain about their menstrual cycle timing in relation to the act of unprotected intercourse that already occurred and need to prioritize maximum effectiveness of emergency contraception.
Patients with unsure cycle-sex timing planning to self-start or resume a short-term hormonal contraceptive method (eg, pills, patches, or rings), should be counseled to wait 5 days after the most recent act of unprotected sex, before taking their hormonal contraceptive method.7 Patients with unsure cycle-sex timing planning to use provider-dependent hormonal contraceptive methods (eg, those requiring a prescription, including a progestin-contraceptive implant or depot medroxyprogesterone acetate) should also be counseled to wait. Timing of levonorgestrel and copper intrauterine devices are addressed in part 3 of this series.
However, if your patient has a good understanding of their menstrual cycle, and the primary concern is exposure from subsequent sexual encounters and not the recent unprotected intercourse, it is advisable to provide UPA and immediately initiate a contraceptive method. One of the primary reasons for emergency contraception failure is that its effectiveness is limited to the most recent act of unprotected sexual intercourse and does not extend to subsequent acts throughout the month.
For these patients with sure cycle-sex timing who are planning to start or resume short-or long-term contraceptive methods, and whose primary concern is to prevent pregnancy risk from subsequent sexual encounters, immediately initiating a contraceptive method is advisable. For provider-dependent methods, we must weigh the risk of unintended pregnancy from the act of intercourse that already occurred (and the potential to increase that risk by initiating a method that could compromise UPA efficacy) versus the future risk of pregnancy if the patient cannot return for a contraception visit.7
In short, starting the contraceptive method at the time of UPA use can be considered after shared decision making with the patient and understanding what their primary concerns are.
Important point
Counsel on using backup barrier contraception after UPA
Oral emergency contraception only covers that one act of unprotected intercourse and does not continue to protect a patient from pregnancy for the rest of their cycle. When taken before ovulation, UPA works by delaying follicular development and rupture for at least 5 days. Patients who continue to have unprotected intercourse after taking UPA are at a high risk of an unintended pregnancy from this ‘stalled’ follicle that will eventually ovulate. Follicular maturation resumes after UPA’s effects wane, and the patient is primed for ovulation (and therefore unintended pregnancy) if ongoing unprotected intercourse occurs for the rest of their cycle.
Therefore, it is important to counsel patients on the need, if they do not desire a pregnancy, to abstain or start a method of contraception.
Final question
What about starting or resuming non–hormonal contraceptive methods?
Non-hormonal contraceptive methods can be started immediately with UPA use.1
CASE Resolved
After shared decision making, the patient decides to start using the COC pill. You prescribe her both UPA for emergency contraception and a combined hormonal contraceptive pill. Given her unsure cycle-sex timing, she expresses to you that her most important priority is preventing unintended pregnancy. You counsel her to set a reminder on her phone to start taking the pill 5 days from her most recent act of unprotected intercourse. You also counsel her to use a back-up barrier method of contraception for 7 days after starting her COC pill. ●

- Curtis KM, Jatlaoui TC, Tepper NK, et al. U.S. Selected Practice Recommendations for Contraceptive Use, 2016. Morb Mortal Wkly Rep. 2016;65:1-66. https://doi .org/10.15585/mmwr.rr6504a1
- Centers for Disease Control and Prevention. National Center for Chronic Disease Prevention and Health Promotion, Division of Reproductive Health. US Selected Practice Recommendations for Contraceptive Use (US-SPR). Accessed October 11, 2023. https://www.cdc.gov/reproductivehealth /contraception/mmwr/spr/summary.html
- Ella [package insert]. Charleston, SC; Afaxys, Inc. 2014.
- Salcedo J, Rodriguez MI, Curtis KM, et al. When can a woman resume or initiate contraception after taking emergency contraceptive pills? A systematic review. Contraception. 2013;87:602-604. https://doi.org/10.1016 /j.contraception.2012.08.013
- Edelman AB, Jensen JT, McCrimmon S, et al. Combined oral contraceptive interference with the ability of ulipristal acetate to delay ovulation: a prospective cohort study. Contraception. 2018;98:463-466. doi: 10.1016/j.contraception.2018.08.003
- Brache V, Cochon L, Duijkers IJM, et al. A prospective, randomized, pharmacodynamic study of quick-starting a desogestrel progestin-only pill following ulipristal acetate for emergency contraception. Hum Reprod Oxf Engl. 2015;30:2785-2793. https://doi.org/10.1093/humrep /dev241
- Cameron ST, Berger C, Michie L, et al. The effects on ovarian activity of ulipristal acetate when ‘quickstarting’ a combined oral contraceptive pill: a prospective, randomized, doubleblind parallel-arm, placebo-controlled study. Hum Reprod. 2015;30:1566-1572. doi: 10.1093/humrep/dev115
- Banh C, Rautenberg T, Diujkers I, et al. The effects on ovarian activity of delaying versus immediately restarting combined oral contraception after missing three pills and taking ulipristal acetate 30 mg. Contraception. 2020;102:145-151. doi: 10.1016/j.contraception.2020.05.013
- American Society for Emergency Contraception. Providing ongoing hormonal contraception after use of emergency contraceptive pills. September 2016. Accessed October 11, 2023. chrome-extension://efaidnbmnnnibpcajpcglclefindmkaj /https://www.americansocietyforec.org/_files/ugd/7f2e0b _ff1bc90bea204644ba28d1b0e6a6a6a8.pdf
Individuals spend close to half of their lives preventing, or planning for, pregnancy. As such, contraception plays a major role in patient-provider interactions. Contraception counseling and management is a common scenario encountered in the general gynecologist’s practice. Luckily, we have two evidence-based guidelines developed by the US Centers for Disease Control and Prevention (CDC) that support the provision of contraceptive care:
- US Medical Eligibility for Contraceptive Use (US-MEC),1 which provides guidance on which patients can safely use a method
- US Selected Practice Recommendations for Contraceptive Use (US-SPR),2 which provides method-specific guidance on how to use a method (including how to: initiate or start a method; manage adherence issues, such as a missed pill, etc; and manage common issues like breakthrough bleeding). Both of these guidelines are updated routinely and are publicly available online or for free, through smartphone applications.
While most contraceptive care is straightforward, there are circumstances that require additional consideration. In this 3-part series we review 3 clinical cases, existing evidence to guide management decisions, and our recommendations. In part 1, we focus on restarting hormonal contraception after ulipristal acetate administration. In parts 2 and 3, we will discuss removal of a nonpalpable contraceptive implant and the consideration of a levonorgestrel-releasing intrauterine device (LNG-IUD) for emergency contraception.
- After using ulipristal acetate for emergency contraception, advise patients to wait at least 5 days to initiate hormonal contraception and about the importance of abstaining or using a back-up method for another 7 days with the start of their hormonal contraceptive method
CASE Meeting emergency and follow-up contraception needs
A 27-year-old woman (G0) presents to you after having unprotected intercourse 4 days ago. She does not formally track her menstrual cycles and is unsure when her last menstrual period was. She is not using contraception but is interested in starting a method. After counseling, she elects to take a dose of oral ulipristal acetate (UPA; Ella) now for emergency contraception and would like to start a combined oral contraceptive (COC) pill moving forward.
How soon after taking UPA should you tell her to start the combined hormonal pill?
Effectiveness of hormonal contraception following UPA
UPA does not appear to decrease the efficacy of COCs when started around the same time. However, immediately starting a hormonal contraceptive can decrease the effectiveness of UPA, and as such, it is recommended to take UPA and then abstain or use a backup method for 7 days before initiating a hormonal contraceptive method.1 By obtaining some additional information from your patient and with the use of shared decision making, though, your patient may be able to start their contraceptive method earlier than 5 days after UPA.
What is UPA
UPA is a progesterone receptor modulator used for emergency contraception intenhded to prevent pregnancy after unprotected intercourse or contraceptive failure.3 It works by delaying follicular rupture at least 5 days, if taken before the peak of the luteinizing hormone (LH) surge. If taken after that timeframe, it does not work. Since UPA competes for the progesterone receptor, there is a concern that the effectiveness of UPA may be decreased if a progestin-containing form of contraception is started immediately after taking UPA, or vice versa.4 Several studies have now specifically looked at the interaction between UPA and progestin-containing contraceptives, including at how UPA is impacted by the contraceptive method, and conversely, how the contraceptive method is impacted by UPA.5-8
Data on types of hormonal contraception. Brache and colleagues demonstrated that UPA users who started a desogestrel progestin-only pill (DSG POP) the next day had higher rates of ovulation within 5 days of taking UPA (45%), compared with those who the next day started a placebo pill (3%).6 This type of progestin-only pill is not available in the United States.
A study by Edelman and colleagues demonstrated similar findings in those starting a COC pill containing estrogen and progestin. When taking a COC two days after UPA use, more participants had evidence of follicular rupture in less than 5 days.5 It should be noted that these studies focused on ovulation, which—while necessary for conception to occur—is a surrogate biomarker for pregnancy risk. Additional studies have looked at the impact of UPA on the COC and have not found that UPA impacts ovulation suppression of the COC with its initiation or use.8
Considering unprotected intercourse and UPA timing. Of course, the risk of pregnancy is reliant on cycle timing plus the presence of viable sperm in the reproductive tract. Sperm have been shown to only be viable in the reproductive tract for 5 days, which could result in fertilization and subsequent pregnancy. Longevity of an egg is much shorter, at 12 to 24 hours after ovulation. For this patient, her exposure was 4 days ago, but sperm are only viable for approximately 5 days—she could consider taking the UPA now and then starting a COC earlier than 5 days since she only needs an extra day or two of protection from the UPA from the sperm in her reproductive tract. Your patient’s involvement in this decision making is paramount, as only they can prioritize their desire to avoid pregnancy from their recent act of unprotected intercourse versus their immediate needs for starting their method of contraception. It is important that individuals abstain from sexual activity or use an additional back-up method during the first 7 days of starting their method of contraception.
Continue to: Counseling considerations for the case patient...
Counseling considerations for the case patient
For a patient planning to start or resume a hormonal contraceptive method after taking UPA, the waiting period recommended by the CDC (5 days) is most beneficial for patients who are uncertain about their menstrual cycle timing in relation to the act of unprotected intercourse that already occurred and need to prioritize maximum effectiveness of emergency contraception.
Patients with unsure cycle-sex timing planning to self-start or resume a short-term hormonal contraceptive method (eg, pills, patches, or rings), should be counseled to wait 5 days after the most recent act of unprotected sex, before taking their hormonal contraceptive method.7 Patients with unsure cycle-sex timing planning to use provider-dependent hormonal contraceptive methods (eg, those requiring a prescription, including a progestin-contraceptive implant or depot medroxyprogesterone acetate) should also be counseled to wait. Timing of levonorgestrel and copper intrauterine devices are addressed in part 3 of this series.
However, if your patient has a good understanding of their menstrual cycle, and the primary concern is exposure from subsequent sexual encounters and not the recent unprotected intercourse, it is advisable to provide UPA and immediately initiate a contraceptive method. One of the primary reasons for emergency contraception failure is that its effectiveness is limited to the most recent act of unprotected sexual intercourse and does not extend to subsequent acts throughout the month.
For these patients with sure cycle-sex timing who are planning to start or resume short-or long-term contraceptive methods, and whose primary concern is to prevent pregnancy risk from subsequent sexual encounters, immediately initiating a contraceptive method is advisable. For provider-dependent methods, we must weigh the risk of unintended pregnancy from the act of intercourse that already occurred (and the potential to increase that risk by initiating a method that could compromise UPA efficacy) versus the future risk of pregnancy if the patient cannot return for a contraception visit.7
In short, starting the contraceptive method at the time of UPA use can be considered after shared decision making with the patient and understanding what their primary concerns are.
Important point
Counsel on using backup barrier contraception after UPA
Oral emergency contraception only covers that one act of unprotected intercourse and does not continue to protect a patient from pregnancy for the rest of their cycle. When taken before ovulation, UPA works by delaying follicular development and rupture for at least 5 days. Patients who continue to have unprotected intercourse after taking UPA are at a high risk of an unintended pregnancy from this ‘stalled’ follicle that will eventually ovulate. Follicular maturation resumes after UPA’s effects wane, and the patient is primed for ovulation (and therefore unintended pregnancy) if ongoing unprotected intercourse occurs for the rest of their cycle.
Therefore, it is important to counsel patients on the need, if they do not desire a pregnancy, to abstain or start a method of contraception.
Final question
What about starting or resuming non–hormonal contraceptive methods?
Non-hormonal contraceptive methods can be started immediately with UPA use.1
CASE Resolved
After shared decision making, the patient decides to start using the COC pill. You prescribe her both UPA for emergency contraception and a combined hormonal contraceptive pill. Given her unsure cycle-sex timing, she expresses to you that her most important priority is preventing unintended pregnancy. You counsel her to set a reminder on her phone to start taking the pill 5 days from her most recent act of unprotected intercourse. You also counsel her to use a back-up barrier method of contraception for 7 days after starting her COC pill. ●

Individuals spend close to half of their lives preventing, or planning for, pregnancy. As such, contraception plays a major role in patient-provider interactions. Contraception counseling and management is a common scenario encountered in the general gynecologist’s practice. Luckily, we have two evidence-based guidelines developed by the US Centers for Disease Control and Prevention (CDC) that support the provision of contraceptive care:
- US Medical Eligibility for Contraceptive Use (US-MEC),1 which provides guidance on which patients can safely use a method
- US Selected Practice Recommendations for Contraceptive Use (US-SPR),2 which provides method-specific guidance on how to use a method (including how to: initiate or start a method; manage adherence issues, such as a missed pill, etc; and manage common issues like breakthrough bleeding). Both of these guidelines are updated routinely and are publicly available online or for free, through smartphone applications.
While most contraceptive care is straightforward, there are circumstances that require additional consideration. In this 3-part series we review 3 clinical cases, existing evidence to guide management decisions, and our recommendations. In part 1, we focus on restarting hormonal contraception after ulipristal acetate administration. In parts 2 and 3, we will discuss removal of a nonpalpable contraceptive implant and the consideration of a levonorgestrel-releasing intrauterine device (LNG-IUD) for emergency contraception.
- After using ulipristal acetate for emergency contraception, advise patients to wait at least 5 days to initiate hormonal contraception and about the importance of abstaining or using a back-up method for another 7 days with the start of their hormonal contraceptive method
CASE Meeting emergency and follow-up contraception needs
A 27-year-old woman (G0) presents to you after having unprotected intercourse 4 days ago. She does not formally track her menstrual cycles and is unsure when her last menstrual period was. She is not using contraception but is interested in starting a method. After counseling, she elects to take a dose of oral ulipristal acetate (UPA; Ella) now for emergency contraception and would like to start a combined oral contraceptive (COC) pill moving forward.
How soon after taking UPA should you tell her to start the combined hormonal pill?
Effectiveness of hormonal contraception following UPA
UPA does not appear to decrease the efficacy of COCs when started around the same time. However, immediately starting a hormonal contraceptive can decrease the effectiveness of UPA, and as such, it is recommended to take UPA and then abstain or use a backup method for 7 days before initiating a hormonal contraceptive method.1 By obtaining some additional information from your patient and with the use of shared decision making, though, your patient may be able to start their contraceptive method earlier than 5 days after UPA.
What is UPA
UPA is a progesterone receptor modulator used for emergency contraception intenhded to prevent pregnancy after unprotected intercourse or contraceptive failure.3 It works by delaying follicular rupture at least 5 days, if taken before the peak of the luteinizing hormone (LH) surge. If taken after that timeframe, it does not work. Since UPA competes for the progesterone receptor, there is a concern that the effectiveness of UPA may be decreased if a progestin-containing form of contraception is started immediately after taking UPA, or vice versa.4 Several studies have now specifically looked at the interaction between UPA and progestin-containing contraceptives, including at how UPA is impacted by the contraceptive method, and conversely, how the contraceptive method is impacted by UPA.5-8
Data on types of hormonal contraception. Brache and colleagues demonstrated that UPA users who started a desogestrel progestin-only pill (DSG POP) the next day had higher rates of ovulation within 5 days of taking UPA (45%), compared with those who the next day started a placebo pill (3%).6 This type of progestin-only pill is not available in the United States.
A study by Edelman and colleagues demonstrated similar findings in those starting a COC pill containing estrogen and progestin. When taking a COC two days after UPA use, more participants had evidence of follicular rupture in less than 5 days.5 It should be noted that these studies focused on ovulation, which—while necessary for conception to occur—is a surrogate biomarker for pregnancy risk. Additional studies have looked at the impact of UPA on the COC and have not found that UPA impacts ovulation suppression of the COC with its initiation or use.8
Considering unprotected intercourse and UPA timing. Of course, the risk of pregnancy is reliant on cycle timing plus the presence of viable sperm in the reproductive tract. Sperm have been shown to only be viable in the reproductive tract for 5 days, which could result in fertilization and subsequent pregnancy. Longevity of an egg is much shorter, at 12 to 24 hours after ovulation. For this patient, her exposure was 4 days ago, but sperm are only viable for approximately 5 days—she could consider taking the UPA now and then starting a COC earlier than 5 days since she only needs an extra day or two of protection from the UPA from the sperm in her reproductive tract. Your patient’s involvement in this decision making is paramount, as only they can prioritize their desire to avoid pregnancy from their recent act of unprotected intercourse versus their immediate needs for starting their method of contraception. It is important that individuals abstain from sexual activity or use an additional back-up method during the first 7 days of starting their method of contraception.
Continue to: Counseling considerations for the case patient...
Counseling considerations for the case patient
For a patient planning to start or resume a hormonal contraceptive method after taking UPA, the waiting period recommended by the CDC (5 days) is most beneficial for patients who are uncertain about their menstrual cycle timing in relation to the act of unprotected intercourse that already occurred and need to prioritize maximum effectiveness of emergency contraception.
Patients with unsure cycle-sex timing planning to self-start or resume a short-term hormonal contraceptive method (eg, pills, patches, or rings), should be counseled to wait 5 days after the most recent act of unprotected sex, before taking their hormonal contraceptive method.7 Patients with unsure cycle-sex timing planning to use provider-dependent hormonal contraceptive methods (eg, those requiring a prescription, including a progestin-contraceptive implant or depot medroxyprogesterone acetate) should also be counseled to wait. Timing of levonorgestrel and copper intrauterine devices are addressed in part 3 of this series.
However, if your patient has a good understanding of their menstrual cycle, and the primary concern is exposure from subsequent sexual encounters and not the recent unprotected intercourse, it is advisable to provide UPA and immediately initiate a contraceptive method. One of the primary reasons for emergency contraception failure is that its effectiveness is limited to the most recent act of unprotected sexual intercourse and does not extend to subsequent acts throughout the month.
For these patients with sure cycle-sex timing who are planning to start or resume short-or long-term contraceptive methods, and whose primary concern is to prevent pregnancy risk from subsequent sexual encounters, immediately initiating a contraceptive method is advisable. For provider-dependent methods, we must weigh the risk of unintended pregnancy from the act of intercourse that already occurred (and the potential to increase that risk by initiating a method that could compromise UPA efficacy) versus the future risk of pregnancy if the patient cannot return for a contraception visit.7
In short, starting the contraceptive method at the time of UPA use can be considered after shared decision making with the patient and understanding what their primary concerns are.
Important point
Counsel on using backup barrier contraception after UPA
Oral emergency contraception only covers that one act of unprotected intercourse and does not continue to protect a patient from pregnancy for the rest of their cycle. When taken before ovulation, UPA works by delaying follicular development and rupture for at least 5 days. Patients who continue to have unprotected intercourse after taking UPA are at a high risk of an unintended pregnancy from this ‘stalled’ follicle that will eventually ovulate. Follicular maturation resumes after UPA’s effects wane, and the patient is primed for ovulation (and therefore unintended pregnancy) if ongoing unprotected intercourse occurs for the rest of their cycle.
Therefore, it is important to counsel patients on the need, if they do not desire a pregnancy, to abstain or start a method of contraception.
Final question
What about starting or resuming non–hormonal contraceptive methods?
Non-hormonal contraceptive methods can be started immediately with UPA use.1
CASE Resolved
After shared decision making, the patient decides to start using the COC pill. You prescribe her both UPA for emergency contraception and a combined hormonal contraceptive pill. Given her unsure cycle-sex timing, she expresses to you that her most important priority is preventing unintended pregnancy. You counsel her to set a reminder on her phone to start taking the pill 5 days from her most recent act of unprotected intercourse. You also counsel her to use a back-up barrier method of contraception for 7 days after starting her COC pill. ●

- Curtis KM, Jatlaoui TC, Tepper NK, et al. U.S. Selected Practice Recommendations for Contraceptive Use, 2016. Morb Mortal Wkly Rep. 2016;65:1-66. https://doi .org/10.15585/mmwr.rr6504a1
- Centers for Disease Control and Prevention. National Center for Chronic Disease Prevention and Health Promotion, Division of Reproductive Health. US Selected Practice Recommendations for Contraceptive Use (US-SPR). Accessed October 11, 2023. https://www.cdc.gov/reproductivehealth /contraception/mmwr/spr/summary.html
- Ella [package insert]. Charleston, SC; Afaxys, Inc. 2014.
- Salcedo J, Rodriguez MI, Curtis KM, et al. When can a woman resume or initiate contraception after taking emergency contraceptive pills? A systematic review. Contraception. 2013;87:602-604. https://doi.org/10.1016 /j.contraception.2012.08.013
- Edelman AB, Jensen JT, McCrimmon S, et al. Combined oral contraceptive interference with the ability of ulipristal acetate to delay ovulation: a prospective cohort study. Contraception. 2018;98:463-466. doi: 10.1016/j.contraception.2018.08.003
- Brache V, Cochon L, Duijkers IJM, et al. A prospective, randomized, pharmacodynamic study of quick-starting a desogestrel progestin-only pill following ulipristal acetate for emergency contraception. Hum Reprod Oxf Engl. 2015;30:2785-2793. https://doi.org/10.1093/humrep /dev241
- Cameron ST, Berger C, Michie L, et al. The effects on ovarian activity of ulipristal acetate when ‘quickstarting’ a combined oral contraceptive pill: a prospective, randomized, doubleblind parallel-arm, placebo-controlled study. Hum Reprod. 2015;30:1566-1572. doi: 10.1093/humrep/dev115
- Banh C, Rautenberg T, Diujkers I, et al. The effects on ovarian activity of delaying versus immediately restarting combined oral contraception after missing three pills and taking ulipristal acetate 30 mg. Contraception. 2020;102:145-151. doi: 10.1016/j.contraception.2020.05.013
- American Society for Emergency Contraception. Providing ongoing hormonal contraception after use of emergency contraceptive pills. September 2016. Accessed October 11, 2023. chrome-extension://efaidnbmnnnibpcajpcglclefindmkaj /https://www.americansocietyforec.org/_files/ugd/7f2e0b _ff1bc90bea204644ba28d1b0e6a6a6a8.pdf
- Curtis KM, Jatlaoui TC, Tepper NK, et al. U.S. Selected Practice Recommendations for Contraceptive Use, 2016. Morb Mortal Wkly Rep. 2016;65:1-66. https://doi .org/10.15585/mmwr.rr6504a1
- Centers for Disease Control and Prevention. National Center for Chronic Disease Prevention and Health Promotion, Division of Reproductive Health. US Selected Practice Recommendations for Contraceptive Use (US-SPR). Accessed October 11, 2023. https://www.cdc.gov/reproductivehealth /contraception/mmwr/spr/summary.html
- Ella [package insert]. Charleston, SC; Afaxys, Inc. 2014.
- Salcedo J, Rodriguez MI, Curtis KM, et al. When can a woman resume or initiate contraception after taking emergency contraceptive pills? A systematic review. Contraception. 2013;87:602-604. https://doi.org/10.1016 /j.contraception.2012.08.013
- Edelman AB, Jensen JT, McCrimmon S, et al. Combined oral contraceptive interference with the ability of ulipristal acetate to delay ovulation: a prospective cohort study. Contraception. 2018;98:463-466. doi: 10.1016/j.contraception.2018.08.003
- Brache V, Cochon L, Duijkers IJM, et al. A prospective, randomized, pharmacodynamic study of quick-starting a desogestrel progestin-only pill following ulipristal acetate for emergency contraception. Hum Reprod Oxf Engl. 2015;30:2785-2793. https://doi.org/10.1093/humrep /dev241
- Cameron ST, Berger C, Michie L, et al. The effects on ovarian activity of ulipristal acetate when ‘quickstarting’ a combined oral contraceptive pill: a prospective, randomized, doubleblind parallel-arm, placebo-controlled study. Hum Reprod. 2015;30:1566-1572. doi: 10.1093/humrep/dev115
- Banh C, Rautenberg T, Diujkers I, et al. The effects on ovarian activity of delaying versus immediately restarting combined oral contraception after missing three pills and taking ulipristal acetate 30 mg. Contraception. 2020;102:145-151. doi: 10.1016/j.contraception.2020.05.013
- American Society for Emergency Contraception. Providing ongoing hormonal contraception after use of emergency contraceptive pills. September 2016. Accessed October 11, 2023. chrome-extension://efaidnbmnnnibpcajpcglclefindmkaj /https://www.americansocietyforec.org/_files/ugd/7f2e0b _ff1bc90bea204644ba28d1b0e6a6a6a8.pdf
RSV vaccination during pregnancy: Finally ready for prime time
A 28-year-old primigravid woman at 30 weeks’ gestation inquires about the new vaccine to protect her newborn baby against respiratory syncytial virus infection (RSV). Her neighbor’s daughter recently was hospitalized for the treatment of RSV, and she is understandably concerned about her own newborn. The patient is healthy, and she has never had any serious respiratory infection. She is taking no medications other than prenatal vitamins.
What advice should you give her?
If you decide to administer this vaccine, what is the appropriate timing of administration?
Are there any maternal or fetal safety concerns related to use of this vaccine in pregnancy?
Respiratory syncytial virus (RSV) is a member of the Paramyxoviridae family. It is an enveloped, single-stranded RNA virus that is 150-300 nm in size. The virus codes for 10 virus-specific proteins. The 2 most important are the G protein, which enables the virus to attach to host cells, and the F protein, which facilitates the entry of the virus into the host cell by fusing the host and viral membranes. Two distinct subtypes exist: A and B. There is genetic variation within each subtype and between subtypes. These subtle genetic variations create the potential for reinfections, and hence, research has focused on development of a vaccine that covers both subtypes.1
RSV is the most common cause of acute lower respiratory tract infection in infants younger than 6 months of age. In these children, RSV is one of the most prominent causes of death, with mortality particularly marked in low- and middle-resource countries as well as in children who were born premature and/or who are immunocompromised. RSV has its greatest impact during winter epidemics in temperate climates and during the rainy seasons in tropical climates. The virus rarely is encountered in the summer.1 Among young children, RSV primarily is transmitted via close contact with contaminated fingers or fomites and by self-inoculation of the conjunctiva or anterior nares. The incubation period of the infection is 4 to 6 days, and viral shedding may persist for 2 weeks or longer. Most patients gradually recover within 1 to 2 weeks.1 Adults who contract RSV usually have symptoms suggestive of a common cold; however, in older adults or those who have comorbidities, serious and potentially life-threatening lower respiratory tract infections may develop.
Recently, there have been 2 main approaches to the prevention and treatment of RSV in infants. One has been the development of monoclonal antibodies such as motavizumab, palivizumab, and nirsevimab. The other has been the development of a vaccine that could be administered to pregnant women and which could provide protection for the neonate in the early months of life.2,3
In late August 2023, the US Food and Drug Administration (FDA) announced the approval of a new bivalent RSV prefusion F vaccine (ABRYSVO, Pfizer) intended for administration to pregnant women.4 Of note, previous efforts to develop whole-virus vaccines either have been ineffective or have potentiated the disease in infants who became infected; development of an effective vaccine had eluded scientists and clinicians for nearly 50 years.2 Thus, the new vaccine that targets the F protein of the virus represents a major and welcomed breakthrough.
This article reviews the 3 most recent investigations that preceded the ultimate approval of this vaccine and discusses specific logistical issues related to vaccine administration.
Continue to: First step toward vaccine approval...
First step toward vaccine approval
Madhi and colleagues5 were among the first to conduct a large well-designed study to evaluate the effectiveness of maternal vaccination in preventing neonatal infection in the first few months of life. The authors enrolled more than 4,500 healthy pregnant women at 28 to 36 weeks of gestation and assigned them to receive either a single intramuscular dose of an RSV fusion (F) protein vaccine or placebo in a ratio of 2:1. The primary end point was a “medically significant lower respiratory tract infection” within the first 90 days of life. The percentage of infants who met the primary end point was low in both groups: 1.5% in the vaccine group and 2.4% in the placebo group (efficacy 39.4%). The efficacy of the vaccine in preventing lower respiratory tract infection with severe hypoxemia was 48.3% and 44.4% in preventing hospitalization. Although there were differences between the 2 groups, they did not meet the prespecified success criterion for efficacy. Vaccine recipients had more local injection site reactions (40.7% vs 9.9%); however, there was no difference in the frequency of other adverse effects.
Intermediate step: Continued assessment of vaccine safety and immunogenicity
The next important step in the development of the RSV vaccine was a study by Simoes et al,6 who conducted a phase 2b trial to determine the safety and immunogenicity of the RSVpreF vaccine. The authors randomly assigned pregnant women at 24 to 36 weeks of gestation to receive either 120 or 240 µg of RSVpreF vaccine or placebo. The key endpoints were the following: maternal and infant safety; the maternal-to-infant transplacental transfer ratio; and the presence of RSV A, B, and combined A/B neutralizing antibody in maternal serum and umbilical cord blood at delivery. The authors conducted a planned interim analysis that included 327 mothers who received the vaccine. The incidence of adverse effects was similar in mothers and infants in the vaccine compared with the placebo group. None of the adverse effects were judged to be serious. The transplacental neutralizing antibody transfer ratios ranged from 1.4 to 2.1 across a range of gestational ages. The vaccine elicited meaningful neutralizing titers of antibody in maternal serum even up to 7 weeks after immunization. The levels of neutralizing antibodies in umbilical cord blood did not vary substantially with respect to gestational age. A post hoc analysis showed that the transferred antibodies prevented medically-attended RSV-associated lower respiratory tract illnesses in the infants.
Final step: Convincing proof of efficacy
The most recent of the 3 studies, and the one that had the greatest impact in convincing the FDA to approve the vaccine, was the report by Kampmann and colleagues.7 The authors conducted a phase 3 prospective, randomized, double-blind trial in 18 different countries over 4 RSV seasons: 2 in the northern hemisphere and 2 in the southern hemisphere. They enrolled healthy pregnant women with singleton gestations at 24 to 36 weeks of gestation and assigned them in a 1:1 ratio to a single intramuscular injection of 120 µg of a bivalent RSV prefusion F protein-based (RSVpreF) vaccine or placebo. They excluded patients with any recognized risk factor for an adverse pregnancy outcome, including preterm labor. The 2 primary efficacy endpoints were a medically-attended severe RSV–lower respiratory tract infection and any medically attended RSV-associated lower respiratory tract illness in infants within 90, 120, 150, and 180 days after birth.
The efficacy of the vaccine in preventing severe lower respiratory tract illness within 90 days of delivery was 81.8% (99.5% confidence interval [CI], 40.6–96.3). The efficacy within 180 days of delivery was 69.4% (97.58% CI, 44.3–84.1). These differences reached the study’s pre-established statistical criteria for success. The overall rate of lower respiratory tract infections was not significantly different. The frequencies of adverse effects in mothers and infants were similar in the vaccine and placebo groups. In particular, the frequency of preterm delivery in the vaccine group was 0.8%, compared with 0.6% in the placebo group (P = NS).
In previous reports to the FDA,4 the frequency rate of preterm delivery in RSV vaccine recipients was slightly increased in vaccine recipients compared with patients who received placebo. The difference among the groups was too small to infer a causal relationship; however, as a condition of vaccine approval, the FDA has required Pfizer to conduct a postmarketing study to be certain that administration of the vaccine does not increase the risk for preterm delivery.
Practical details
The new vaccine is a bivalent recombinant vaccine that elicits a robust antibody response against the F (fusion) protein of the virus. In addition to the F antigen, the vaccine contains the following buffer ingredients: tromethamine, sucrose, mannitol, polysorbate, and sodium chloride.8 There are no preservatives in the vaccine.
The vaccine should be administered in a single, 0.5 mL, intramuscular injection at 32 to 36 weeks of gestation. Patients who are allergic to any of the components of the vaccine should not be vaccinated. Patients with a mild upper respiratory tract infection may receive the vaccine. Administration should be delayed in patients who are moderately to severely ill. The vaccine may be administered at the same time as other vaccines, such as influenza or Tdap.
The most common side effects of the vaccine are local injection site reactions, such as pain, redness, or swelling. Some patients may experience mild systemic manifestations, including fatigue, fever, headache, nausea, diarrhea, arthralgias, and myalgias. According to the Centers for Disease Control and Prevention, the approximate wholesale acquisition cost of the vaccine is $320 for 1 injection.
CASE Resolution
This patient is healthy and has no contraindication to the new RSV vaccine. According to the FDA, the optimal time for administration of the vaccine is 32 to 36 weeks of gestation. The patient should anticipate very few side effects following the vaccination, and the vaccine has approximately 80% efficacy in preventing severe lower respiratory tract infection in her neonate. ●
- RSV is the most common cause of acute lower respiratory tract infection in infants younger than 6 months of age.
- In low- and middle-resource countries, RSV is a leading cause of infant death.
- In late August 2023, the FDA approved the first RSV vaccine that can be administered to pregnant women to provide protection for the infant in the first few months of life.
- The vaccine specifically targets the F protein of the virus, a protein which is essential for facilitating fusion between the viral and host cell membranes, resulting in penetration of the virus into the host cell.
- The vaccine should be administered as a single intramuscular injection at 32 to 36 weeks’ gestation.
- The vaccine is approximately 82% effective in preventing severe lower respiratory tract infection in infants within the first 6 months of life.
- To exercise an abundance of caution, because of a possible association between administration of the vaccine and an increased risk for preterm delivery, vaccination should be delayed until 36 weeks in patients clearly identified as at-risk for preterm delivery.
- Dolin R. Common viral respiratory infections. In, Isselbacher KJ, Braunwald E, Wilson JD, et al, eds. Harrison’s Principles of Internal Medicine. 13th ed. McGraw-Hill; 1994:805-806.
- Mazur N, Terstappen J, Baral R, et al. Respiratory syncytial virus prevention within reach: the vaccine and monoclonal antibody landscape. Lancet Infect Dis. 2023;23:E2-E21.
- Hammitt LL, Dagan R, Yuan Y, et al. Nirsevimab for prevention of RSV in healthy late-preterm and term infants. N Engl J Med. 2022;386:837-846.
- US Food and Drug Administration News Release. August 21, 2023. Accessed October 26, 2023. https://www.fda.gov/news -events/press-announcements/fda-approves-first-vaccine -pregnant-individuals-prevent-rsv-infants
- Madhi SA, Polack FP, Piedra PA, et al. Respiratory syncytial virus vaccination during pregnancy and effects in infants. N Engl J Med. 2020;383:426-439.
- Simoes EAF, Center KJ, Tita ATN, et al. Prefusion F proteinbased respiratory syncytial virus immunization in pregnancy. N Eng J Med. 2022;386:1615-1626.
- Kampmann B, Madhi SA, Munjal I, et al. Bivalent prefusion F vaccine in pregnancy to prevent RSV illness in infants. N Engl J Med. 2023;388:1451-1464.
- Centers for Disease Control and Prevention. Vaccine Information Statement. Respiratory Syncytial Virus (RSV) Vaccine VIS. October 19, 2023. Accessed October 26, 2023. https://www. cdc.gov/vaccines/hcp/vis/vis-statements/rsv.html
A 28-year-old primigravid woman at 30 weeks’ gestation inquires about the new vaccine to protect her newborn baby against respiratory syncytial virus infection (RSV). Her neighbor’s daughter recently was hospitalized for the treatment of RSV, and she is understandably concerned about her own newborn. The patient is healthy, and she has never had any serious respiratory infection. She is taking no medications other than prenatal vitamins.
What advice should you give her?
If you decide to administer this vaccine, what is the appropriate timing of administration?
Are there any maternal or fetal safety concerns related to use of this vaccine in pregnancy?
Respiratory syncytial virus (RSV) is a member of the Paramyxoviridae family. It is an enveloped, single-stranded RNA virus that is 150-300 nm in size. The virus codes for 10 virus-specific proteins. The 2 most important are the G protein, which enables the virus to attach to host cells, and the F protein, which facilitates the entry of the virus into the host cell by fusing the host and viral membranes. Two distinct subtypes exist: A and B. There is genetic variation within each subtype and between subtypes. These subtle genetic variations create the potential for reinfections, and hence, research has focused on development of a vaccine that covers both subtypes.1
RSV is the most common cause of acute lower respiratory tract infection in infants younger than 6 months of age. In these children, RSV is one of the most prominent causes of death, with mortality particularly marked in low- and middle-resource countries as well as in children who were born premature and/or who are immunocompromised. RSV has its greatest impact during winter epidemics in temperate climates and during the rainy seasons in tropical climates. The virus rarely is encountered in the summer.1 Among young children, RSV primarily is transmitted via close contact with contaminated fingers or fomites and by self-inoculation of the conjunctiva or anterior nares. The incubation period of the infection is 4 to 6 days, and viral shedding may persist for 2 weeks or longer. Most patients gradually recover within 1 to 2 weeks.1 Adults who contract RSV usually have symptoms suggestive of a common cold; however, in older adults or those who have comorbidities, serious and potentially life-threatening lower respiratory tract infections may develop.
Recently, there have been 2 main approaches to the prevention and treatment of RSV in infants. One has been the development of monoclonal antibodies such as motavizumab, palivizumab, and nirsevimab. The other has been the development of a vaccine that could be administered to pregnant women and which could provide protection for the neonate in the early months of life.2,3
In late August 2023, the US Food and Drug Administration (FDA) announced the approval of a new bivalent RSV prefusion F vaccine (ABRYSVO, Pfizer) intended for administration to pregnant women.4 Of note, previous efforts to develop whole-virus vaccines either have been ineffective or have potentiated the disease in infants who became infected; development of an effective vaccine had eluded scientists and clinicians for nearly 50 years.2 Thus, the new vaccine that targets the F protein of the virus represents a major and welcomed breakthrough.
This article reviews the 3 most recent investigations that preceded the ultimate approval of this vaccine and discusses specific logistical issues related to vaccine administration.
Continue to: First step toward vaccine approval...
First step toward vaccine approval
Madhi and colleagues5 were among the first to conduct a large well-designed study to evaluate the effectiveness of maternal vaccination in preventing neonatal infection in the first few months of life. The authors enrolled more than 4,500 healthy pregnant women at 28 to 36 weeks of gestation and assigned them to receive either a single intramuscular dose of an RSV fusion (F) protein vaccine or placebo in a ratio of 2:1. The primary end point was a “medically significant lower respiratory tract infection” within the first 90 days of life. The percentage of infants who met the primary end point was low in both groups: 1.5% in the vaccine group and 2.4% in the placebo group (efficacy 39.4%). The efficacy of the vaccine in preventing lower respiratory tract infection with severe hypoxemia was 48.3% and 44.4% in preventing hospitalization. Although there were differences between the 2 groups, they did not meet the prespecified success criterion for efficacy. Vaccine recipients had more local injection site reactions (40.7% vs 9.9%); however, there was no difference in the frequency of other adverse effects.
Intermediate step: Continued assessment of vaccine safety and immunogenicity
The next important step in the development of the RSV vaccine was a study by Simoes et al,6 who conducted a phase 2b trial to determine the safety and immunogenicity of the RSVpreF vaccine. The authors randomly assigned pregnant women at 24 to 36 weeks of gestation to receive either 120 or 240 µg of RSVpreF vaccine or placebo. The key endpoints were the following: maternal and infant safety; the maternal-to-infant transplacental transfer ratio; and the presence of RSV A, B, and combined A/B neutralizing antibody in maternal serum and umbilical cord blood at delivery. The authors conducted a planned interim analysis that included 327 mothers who received the vaccine. The incidence of adverse effects was similar in mothers and infants in the vaccine compared with the placebo group. None of the adverse effects were judged to be serious. The transplacental neutralizing antibody transfer ratios ranged from 1.4 to 2.1 across a range of gestational ages. The vaccine elicited meaningful neutralizing titers of antibody in maternal serum even up to 7 weeks after immunization. The levels of neutralizing antibodies in umbilical cord blood did not vary substantially with respect to gestational age. A post hoc analysis showed that the transferred antibodies prevented medically-attended RSV-associated lower respiratory tract illnesses in the infants.
Final step: Convincing proof of efficacy
The most recent of the 3 studies, and the one that had the greatest impact in convincing the FDA to approve the vaccine, was the report by Kampmann and colleagues.7 The authors conducted a phase 3 prospective, randomized, double-blind trial in 18 different countries over 4 RSV seasons: 2 in the northern hemisphere and 2 in the southern hemisphere. They enrolled healthy pregnant women with singleton gestations at 24 to 36 weeks of gestation and assigned them in a 1:1 ratio to a single intramuscular injection of 120 µg of a bivalent RSV prefusion F protein-based (RSVpreF) vaccine or placebo. They excluded patients with any recognized risk factor for an adverse pregnancy outcome, including preterm labor. The 2 primary efficacy endpoints were a medically-attended severe RSV–lower respiratory tract infection and any medically attended RSV-associated lower respiratory tract illness in infants within 90, 120, 150, and 180 days after birth.
The efficacy of the vaccine in preventing severe lower respiratory tract illness within 90 days of delivery was 81.8% (99.5% confidence interval [CI], 40.6–96.3). The efficacy within 180 days of delivery was 69.4% (97.58% CI, 44.3–84.1). These differences reached the study’s pre-established statistical criteria for success. The overall rate of lower respiratory tract infections was not significantly different. The frequencies of adverse effects in mothers and infants were similar in the vaccine and placebo groups. In particular, the frequency of preterm delivery in the vaccine group was 0.8%, compared with 0.6% in the placebo group (P = NS).
In previous reports to the FDA,4 the frequency rate of preterm delivery in RSV vaccine recipients was slightly increased in vaccine recipients compared with patients who received placebo. The difference among the groups was too small to infer a causal relationship; however, as a condition of vaccine approval, the FDA has required Pfizer to conduct a postmarketing study to be certain that administration of the vaccine does not increase the risk for preterm delivery.
Practical details
The new vaccine is a bivalent recombinant vaccine that elicits a robust antibody response against the F (fusion) protein of the virus. In addition to the F antigen, the vaccine contains the following buffer ingredients: tromethamine, sucrose, mannitol, polysorbate, and sodium chloride.8 There are no preservatives in the vaccine.
The vaccine should be administered in a single, 0.5 mL, intramuscular injection at 32 to 36 weeks of gestation. Patients who are allergic to any of the components of the vaccine should not be vaccinated. Patients with a mild upper respiratory tract infection may receive the vaccine. Administration should be delayed in patients who are moderately to severely ill. The vaccine may be administered at the same time as other vaccines, such as influenza or Tdap.
The most common side effects of the vaccine are local injection site reactions, such as pain, redness, or swelling. Some patients may experience mild systemic manifestations, including fatigue, fever, headache, nausea, diarrhea, arthralgias, and myalgias. According to the Centers for Disease Control and Prevention, the approximate wholesale acquisition cost of the vaccine is $320 for 1 injection.
CASE Resolution
This patient is healthy and has no contraindication to the new RSV vaccine. According to the FDA, the optimal time for administration of the vaccine is 32 to 36 weeks of gestation. The patient should anticipate very few side effects following the vaccination, and the vaccine has approximately 80% efficacy in preventing severe lower respiratory tract infection in her neonate. ●
- RSV is the most common cause of acute lower respiratory tract infection in infants younger than 6 months of age.
- In low- and middle-resource countries, RSV is a leading cause of infant death.
- In late August 2023, the FDA approved the first RSV vaccine that can be administered to pregnant women to provide protection for the infant in the first few months of life.
- The vaccine specifically targets the F protein of the virus, a protein which is essential for facilitating fusion between the viral and host cell membranes, resulting in penetration of the virus into the host cell.
- The vaccine should be administered as a single intramuscular injection at 32 to 36 weeks’ gestation.
- The vaccine is approximately 82% effective in preventing severe lower respiratory tract infection in infants within the first 6 months of life.
- To exercise an abundance of caution, because of a possible association between administration of the vaccine and an increased risk for preterm delivery, vaccination should be delayed until 36 weeks in patients clearly identified as at-risk for preterm delivery.
A 28-year-old primigravid woman at 30 weeks’ gestation inquires about the new vaccine to protect her newborn baby against respiratory syncytial virus infection (RSV). Her neighbor’s daughter recently was hospitalized for the treatment of RSV, and she is understandably concerned about her own newborn. The patient is healthy, and she has never had any serious respiratory infection. She is taking no medications other than prenatal vitamins.
What advice should you give her?
If you decide to administer this vaccine, what is the appropriate timing of administration?
Are there any maternal or fetal safety concerns related to use of this vaccine in pregnancy?
Respiratory syncytial virus (RSV) is a member of the Paramyxoviridae family. It is an enveloped, single-stranded RNA virus that is 150-300 nm in size. The virus codes for 10 virus-specific proteins. The 2 most important are the G protein, which enables the virus to attach to host cells, and the F protein, which facilitates the entry of the virus into the host cell by fusing the host and viral membranes. Two distinct subtypes exist: A and B. There is genetic variation within each subtype and between subtypes. These subtle genetic variations create the potential for reinfections, and hence, research has focused on development of a vaccine that covers both subtypes.1
RSV is the most common cause of acute lower respiratory tract infection in infants younger than 6 months of age. In these children, RSV is one of the most prominent causes of death, with mortality particularly marked in low- and middle-resource countries as well as in children who were born premature and/or who are immunocompromised. RSV has its greatest impact during winter epidemics in temperate climates and during the rainy seasons in tropical climates. The virus rarely is encountered in the summer.1 Among young children, RSV primarily is transmitted via close contact with contaminated fingers or fomites and by self-inoculation of the conjunctiva or anterior nares. The incubation period of the infection is 4 to 6 days, and viral shedding may persist for 2 weeks or longer. Most patients gradually recover within 1 to 2 weeks.1 Adults who contract RSV usually have symptoms suggestive of a common cold; however, in older adults or those who have comorbidities, serious and potentially life-threatening lower respiratory tract infections may develop.
Recently, there have been 2 main approaches to the prevention and treatment of RSV in infants. One has been the development of monoclonal antibodies such as motavizumab, palivizumab, and nirsevimab. The other has been the development of a vaccine that could be administered to pregnant women and which could provide protection for the neonate in the early months of life.2,3
In late August 2023, the US Food and Drug Administration (FDA) announced the approval of a new bivalent RSV prefusion F vaccine (ABRYSVO, Pfizer) intended for administration to pregnant women.4 Of note, previous efforts to develop whole-virus vaccines either have been ineffective or have potentiated the disease in infants who became infected; development of an effective vaccine had eluded scientists and clinicians for nearly 50 years.2 Thus, the new vaccine that targets the F protein of the virus represents a major and welcomed breakthrough.
This article reviews the 3 most recent investigations that preceded the ultimate approval of this vaccine and discusses specific logistical issues related to vaccine administration.
Continue to: First step toward vaccine approval...
First step toward vaccine approval
Madhi and colleagues5 were among the first to conduct a large well-designed study to evaluate the effectiveness of maternal vaccination in preventing neonatal infection in the first few months of life. The authors enrolled more than 4,500 healthy pregnant women at 28 to 36 weeks of gestation and assigned them to receive either a single intramuscular dose of an RSV fusion (F) protein vaccine or placebo in a ratio of 2:1. The primary end point was a “medically significant lower respiratory tract infection” within the first 90 days of life. The percentage of infants who met the primary end point was low in both groups: 1.5% in the vaccine group and 2.4% in the placebo group (efficacy 39.4%). The efficacy of the vaccine in preventing lower respiratory tract infection with severe hypoxemia was 48.3% and 44.4% in preventing hospitalization. Although there were differences between the 2 groups, they did not meet the prespecified success criterion for efficacy. Vaccine recipients had more local injection site reactions (40.7% vs 9.9%); however, there was no difference in the frequency of other adverse effects.
Intermediate step: Continued assessment of vaccine safety and immunogenicity
The next important step in the development of the RSV vaccine was a study by Simoes et al,6 who conducted a phase 2b trial to determine the safety and immunogenicity of the RSVpreF vaccine. The authors randomly assigned pregnant women at 24 to 36 weeks of gestation to receive either 120 or 240 µg of RSVpreF vaccine or placebo. The key endpoints were the following: maternal and infant safety; the maternal-to-infant transplacental transfer ratio; and the presence of RSV A, B, and combined A/B neutralizing antibody in maternal serum and umbilical cord blood at delivery. The authors conducted a planned interim analysis that included 327 mothers who received the vaccine. The incidence of adverse effects was similar in mothers and infants in the vaccine compared with the placebo group. None of the adverse effects were judged to be serious. The transplacental neutralizing antibody transfer ratios ranged from 1.4 to 2.1 across a range of gestational ages. The vaccine elicited meaningful neutralizing titers of antibody in maternal serum even up to 7 weeks after immunization. The levels of neutralizing antibodies in umbilical cord blood did not vary substantially with respect to gestational age. A post hoc analysis showed that the transferred antibodies prevented medically-attended RSV-associated lower respiratory tract illnesses in the infants.
Final step: Convincing proof of efficacy
The most recent of the 3 studies, and the one that had the greatest impact in convincing the FDA to approve the vaccine, was the report by Kampmann and colleagues.7 The authors conducted a phase 3 prospective, randomized, double-blind trial in 18 different countries over 4 RSV seasons: 2 in the northern hemisphere and 2 in the southern hemisphere. They enrolled healthy pregnant women with singleton gestations at 24 to 36 weeks of gestation and assigned them in a 1:1 ratio to a single intramuscular injection of 120 µg of a bivalent RSV prefusion F protein-based (RSVpreF) vaccine or placebo. They excluded patients with any recognized risk factor for an adverse pregnancy outcome, including preterm labor. The 2 primary efficacy endpoints were a medically-attended severe RSV–lower respiratory tract infection and any medically attended RSV-associated lower respiratory tract illness in infants within 90, 120, 150, and 180 days after birth.
The efficacy of the vaccine in preventing severe lower respiratory tract illness within 90 days of delivery was 81.8% (99.5% confidence interval [CI], 40.6–96.3). The efficacy within 180 days of delivery was 69.4% (97.58% CI, 44.3–84.1). These differences reached the study’s pre-established statistical criteria for success. The overall rate of lower respiratory tract infections was not significantly different. The frequencies of adverse effects in mothers and infants were similar in the vaccine and placebo groups. In particular, the frequency of preterm delivery in the vaccine group was 0.8%, compared with 0.6% in the placebo group (P = NS).
In previous reports to the FDA,4 the frequency rate of preterm delivery in RSV vaccine recipients was slightly increased in vaccine recipients compared with patients who received placebo. The difference among the groups was too small to infer a causal relationship; however, as a condition of vaccine approval, the FDA has required Pfizer to conduct a postmarketing study to be certain that administration of the vaccine does not increase the risk for preterm delivery.
Practical details
The new vaccine is a bivalent recombinant vaccine that elicits a robust antibody response against the F (fusion) protein of the virus. In addition to the F antigen, the vaccine contains the following buffer ingredients: tromethamine, sucrose, mannitol, polysorbate, and sodium chloride.8 There are no preservatives in the vaccine.
The vaccine should be administered in a single, 0.5 mL, intramuscular injection at 32 to 36 weeks of gestation. Patients who are allergic to any of the components of the vaccine should not be vaccinated. Patients with a mild upper respiratory tract infection may receive the vaccine. Administration should be delayed in patients who are moderately to severely ill. The vaccine may be administered at the same time as other vaccines, such as influenza or Tdap.
The most common side effects of the vaccine are local injection site reactions, such as pain, redness, or swelling. Some patients may experience mild systemic manifestations, including fatigue, fever, headache, nausea, diarrhea, arthralgias, and myalgias. According to the Centers for Disease Control and Prevention, the approximate wholesale acquisition cost of the vaccine is $320 for 1 injection.
CASE Resolution
This patient is healthy and has no contraindication to the new RSV vaccine. According to the FDA, the optimal time for administration of the vaccine is 32 to 36 weeks of gestation. The patient should anticipate very few side effects following the vaccination, and the vaccine has approximately 80% efficacy in preventing severe lower respiratory tract infection in her neonate. ●
- RSV is the most common cause of acute lower respiratory tract infection in infants younger than 6 months of age.
- In low- and middle-resource countries, RSV is a leading cause of infant death.
- In late August 2023, the FDA approved the first RSV vaccine that can be administered to pregnant women to provide protection for the infant in the first few months of life.
- The vaccine specifically targets the F protein of the virus, a protein which is essential for facilitating fusion between the viral and host cell membranes, resulting in penetration of the virus into the host cell.
- The vaccine should be administered as a single intramuscular injection at 32 to 36 weeks’ gestation.
- The vaccine is approximately 82% effective in preventing severe lower respiratory tract infection in infants within the first 6 months of life.
- To exercise an abundance of caution, because of a possible association between administration of the vaccine and an increased risk for preterm delivery, vaccination should be delayed until 36 weeks in patients clearly identified as at-risk for preterm delivery.
- Dolin R. Common viral respiratory infections. In, Isselbacher KJ, Braunwald E, Wilson JD, et al, eds. Harrison’s Principles of Internal Medicine. 13th ed. McGraw-Hill; 1994:805-806.
- Mazur N, Terstappen J, Baral R, et al. Respiratory syncytial virus prevention within reach: the vaccine and monoclonal antibody landscape. Lancet Infect Dis. 2023;23:E2-E21.
- Hammitt LL, Dagan R, Yuan Y, et al. Nirsevimab for prevention of RSV in healthy late-preterm and term infants. N Engl J Med. 2022;386:837-846.
- US Food and Drug Administration News Release. August 21, 2023. Accessed October 26, 2023. https://www.fda.gov/news -events/press-announcements/fda-approves-first-vaccine -pregnant-individuals-prevent-rsv-infants
- Madhi SA, Polack FP, Piedra PA, et al. Respiratory syncytial virus vaccination during pregnancy and effects in infants. N Engl J Med. 2020;383:426-439.
- Simoes EAF, Center KJ, Tita ATN, et al. Prefusion F proteinbased respiratory syncytial virus immunization in pregnancy. N Eng J Med. 2022;386:1615-1626.
- Kampmann B, Madhi SA, Munjal I, et al. Bivalent prefusion F vaccine in pregnancy to prevent RSV illness in infants. N Engl J Med. 2023;388:1451-1464.
- Centers for Disease Control and Prevention. Vaccine Information Statement. Respiratory Syncytial Virus (RSV) Vaccine VIS. October 19, 2023. Accessed October 26, 2023. https://www. cdc.gov/vaccines/hcp/vis/vis-statements/rsv.html
- Dolin R. Common viral respiratory infections. In, Isselbacher KJ, Braunwald E, Wilson JD, et al, eds. Harrison’s Principles of Internal Medicine. 13th ed. McGraw-Hill; 1994:805-806.
- Mazur N, Terstappen J, Baral R, et al. Respiratory syncytial virus prevention within reach: the vaccine and monoclonal antibody landscape. Lancet Infect Dis. 2023;23:E2-E21.
- Hammitt LL, Dagan R, Yuan Y, et al. Nirsevimab for prevention of RSV in healthy late-preterm and term infants. N Engl J Med. 2022;386:837-846.
- US Food and Drug Administration News Release. August 21, 2023. Accessed October 26, 2023. https://www.fda.gov/news -events/press-announcements/fda-approves-first-vaccine -pregnant-individuals-prevent-rsv-infants
- Madhi SA, Polack FP, Piedra PA, et al. Respiratory syncytial virus vaccination during pregnancy and effects in infants. N Engl J Med. 2020;383:426-439.
- Simoes EAF, Center KJ, Tita ATN, et al. Prefusion F proteinbased respiratory syncytial virus immunization in pregnancy. N Eng J Med. 2022;386:1615-1626.
- Kampmann B, Madhi SA, Munjal I, et al. Bivalent prefusion F vaccine in pregnancy to prevent RSV illness in infants. N Engl J Med. 2023;388:1451-1464.
- Centers for Disease Control and Prevention. Vaccine Information Statement. Respiratory Syncytial Virus (RSV) Vaccine VIS. October 19, 2023. Accessed October 26, 2023. https://www. cdc.gov/vaccines/hcp/vis/vis-statements/rsv.html
Essential oils: How safe? How effective?
Essential oils (EOs), which are concentrated plant-based oils, have become ubiquitous over the past decade. Given the far reach of EOs and their longtime use in traditional, complementary, alternative, and integrative medicine, it is imperative that clinicians have some knowledge of the potential benefits, risks, and overall efficacy.
Commonly used for aromatic benefits (aromatherapy), EOs are now also incorporated into a multitude of products promoting health and wellness. EOs are sold as individual products and can be a component in consumer goods such as cosmetics, body care/hygiene/beauty products, laundry detergents, insect repellents, over-the-counter medications, and food.
The review that follows presents the most current evidence available. With that said, it’s important to keep in mind some caveats that relate to this evidence. First, the studies cited tend to have a small sample size. Second, a majority of these studies were conducted in countries where there appears to be a significant culture of EO use, which could contribute to confirmation bias. Finally, in a number of the studies, there is concern for publication bias as well as a discrepancy between calculated statistical significance and actual clinical relevance.

What are essential oils?
EOs generally are made by extracting the oil from leaves, bark, flowers, seeds/fruit, rinds, and/or roots by steaming or pressing parts of a plant. It can take several pounds of plant material to produce a single bottle of EO, which usually contains ≥ 15 to 30 mL (.5 to 1 oz).1
Some commonly used EOs in the United States are lavender, peppermint, rose, clary sage, tea tree, eucalyptus, and citrus; however, there are approximately 300 EOs available.2 EOs are used most often via topical application, inhalation, or ingestion.
As with any botanical agent, EOs are complex substances often containing a multitude of chemical compounds.1 Because of the complex makeup of EOs, which often contain up to 100 volatile organic compounds, and their wide-ranging potential effects, applying the scientific method to study effectiveness poses a challenge that has limited their adoption in evidence-based practice.2
Availability and cost. EOs can be purchased at large retailers (eg, grocery stores, drug stores) and smaller health food stores, as well as on the Internet. Various EO vehicles, such as inhalers and topical creams, also can be purchased at these stores.
Continue to: The cost varies...
The cost varies enormously by manufacturer and type of plant used to make the EO. Common EOs such as peppermint and lavender oil generally cost $10 to $25, while rarer plant oils can cost $80 or more per bottle.
How safe are essential oils?
Patients may assume EOs are harmless because they are derived from natural plants and have been used medicinally for centuries. However, care must be taken with their use.
The safest way to use EOs is topically, although due to their highly concentrated nature, EOs should be diluted in an unscented neutral carrier oil such as coconut, jojoba, olive, or sweet almond.3 Ingestion of certain oils can cause hepatotoxicity, seizures, and even death.3 In fact, patients should speak with a knowledgeable physician before purchasing any oral EO capsules.
Whether used topically or ingested, all EOs carry risk for skin irritation and allergic reactions, and oral ingestion may result in some negative gastrointestinal (GI) adverse effects.4 A case report of 3 patients published in 2007 identified the potential for lavender and tea tree EOs to be endocrine disruptors.5
Inhalation of EOs may be harmful, as they emit many volatile organic compounds, some of which are considered potentially hazardous.6 At this time, there is insufficient evidence regarding inhaled EOs and their direct connection to respiratory health. It is reasonable to suggest, however, that the prolonged use of EOs and their use by patients who have lung conditions such as asthma or COPD should be avoided.7
Continue to: How are quality and purity assessed?
How are quality and purity assessed?
Like other dietary supplements, EOs are not regulated. No US regulatory agencies (eg, the US Food and Drug Administration [FDA] or Department of Agriculture [USDA]) certify or approve EOs for quality and purity. Bottles labeled with “QAI” for Quality Assurance International or “USDA Organic” will ensure the plant constituents used in the EO are from organic farming but do not attest to quality or purity.
Manufacturers commonly use marketing terms such as “therapeutic grade” or “pure” to sell products, but again, these terms do not reflect the product’s quality or purity. A labeled single EO may contain contaminants, alcohol, or additional ingredients.7 When choosing to use EOs, identifying reputable brands is essential; one resource is the independent testing organization ConsumerLab.com.
It is important to assess the manufacturer and read ingredient labels before purchasing an EO to understand what the product contains. Reputable companies will identify the plant ingredient, usually by the formal Latin binomial name, and explain the extraction process. A more certain way to assess the quality and purity of an EO is to ask the manufacturer to provide a certificate of analysis and gas chromatography/mass spectroscopy (GC/MS) data for the specific product. Some manufacturers offer GC/MS test results on their website Quality page.8 Others have detailed information on quality and testing, and GC/MS test reports can be obtained.9 Yet another manufacturer has test results on a product page matching reports to batch codes.10
Which conditions have evidence of benefit from essential oils?
EOs currently are being studied for treatment of many conditions—including pain, GI disorders, behavioral health disorders, and women’s health issues. The TABLE summarizes the conditions treated, outcomes, and practical applications of EOs.11-44
Pain
Headache. As an adjunct to available medications and procedures for headache treatment, EOs are one of the nonpharmacologic modalities that patients and clinicians have at their disposal for both migraine and tension-type headaches. A systematic review of 19 randomized controlled trials (RCTs) examining the effects of herbal ingredients for the acute treatment or prophylaxis of migraines found certain topically applied or inhaled EOs, such as peppermint and chamomile, to be effective for migraine pain alleviation; however, topically applied rose oil was not effective.11-13 Note: “topical application” in these studies implies application of the EO to ≥ 1 of the following areas: temples, forehead, behind ears, or above upper lip/below the nose.
Continue to: One RCT with 120 patients...
One RCT with 120 patients evaluated diluted intranasal peppermint oil and found that it reduced migraine intensity at similar rates to intranasal lidocaine.13 In this study, patients were randomized to receive one of the following: 4% lidocaine, 1.5% peppermint EO, or placebo. Two drops of the intranasal intervention were self-administered while the patient was in a supine position with their head suspended off the edge of the surface on which they were lying. They were instructed to stay in this position for at least 30 seconds after administration.
With regard to tension headache treatment, there is limited literature on the use of EOs. One study found that a preparation of peppermint oil applied topically to the temples and forehead of study participants resulted in significant analgesic effect.14
Fibromyalgia. Usual treatments for fibromyalgia include exercise, antidepressant and anticonvulsant medications, and stress management. Evidence also supports the use of inhaled and topically applied (with and without massage) lavender oil to improve symptoms.26 Positive effects may be related to the analgesic, anti-inflammatory, sleep-regulating, and anxiety-reducing effects of the major volatile compounds contained in lavender oil.
In one RCT with 42 patients with fibromyalgia, the use of inhaled lavender oil was shown to increase the perception of well-being (assessed on the validated SF-36 Health Survey Questionnaire) after 4 weeks.27 In this study, the patient applied 3 drops of an oil mixture, comprising 1 mL lavender EO and 10 mL of fixed neutral base oil, to the wrist and inhaled for 10 minutes before going to bed.
The use of a topical oil blend labeled “Oil 24” (containing camphor, rosemary, eucalyptus, peppermint, aloe vera, and lemon/orange) also has been shown to be more effective than placebo in managing fibromyalgia symptoms. A randomized controlled pilot study of 153 participants found that regular application of Oil 24 improved scores on pain scales and the Fibromyalgia Impact Questionnaire.28
Continue to: GI disorders
GI disorders
Irritable bowel syndrome. Peppermint oil relaxes GI smooth muscle, which has led to investigation of its use in irritable bowel syndrome (IBS) symptom amelioration.17 One meta-analysis including 12 RCTs with 835 patients with undifferentiated IBS found that orally ingested peppermint EO capsules reduced patient-reported symptoms of either abdominal pain or global symptoms.18
One study utilized the Total IBS Symptom Score to evaluate symptom reduction in patients with IBS-D (with diarrhea) and IBS-M (mixed) using 180-mg peppermint EO capsules ingested 3 times daily. There was a significant improvement in abdominal pain/discomfort, bloating/distension, pain at evacuation, and bowel urgency.19 A reduction in symptoms was observed after the first 24 hours of treatment and at the end of the 4-week treatment period.
In another study, among the 190 patients meeting Rome IV criteria for general (nonspecific) IBS who were treated with 182-mg peppermint EO capsules, no statistically significant reduction in overall symptom relief was found (based on outcome measures by the FDA and European Medicines Agency). However, in a secondary outcome analysis, peppermint oil produced greater improvements than placebo for the alleviation of abdominal pain, discomfort, and general IBS severity.20
Chemotherapy-induced nausea and vomiting. Patients with cancer undergoing chemotherapy often explore integrative medicine approaches, including aromatherapy, to ameliorate adverse effects and improve quality of life.38 A few small studies have shown potential for the use of inhaled ginger oil to reduce nausea and vomiting severity and improve health-related quality-of-life measures in these patients.
For example, a study with 60 participants found that inhaling ginger EO for 10 minutes was beneficial for reducing both nausea and vomiting.39 A single-blind, controlled, randomized crossover study of 60 patients with breast cancer receiving chemotherapy showed that ginger EO inhaled 3 times per day for 2 minutes at a time can decrease the severity of nausea but had no effect on vomiting. The same study showed that health-related quality of life improved with the ginger oil treatment.40
Continue to: Other EOs such as cardamom...
Other EOs such as cardamom and peppermint show promise as an adjunctive treatment for chemotherapy-induced nausea and vomiting as well.38
Postoperative nausea. A 2013 randomized trial of 303 patients examined the use of ginger EO, a blend of EOs (including ginger, spearmint, peppermint, and cardamom), and isopropyl alcohol. Both the single EO and EO blend significantly reduced the symptom of nausea. The number of antiemetic medications requested by patients receiving an EO also was significantly reduced compared to those receiving saline.15
The use of EOs to reduce nausea after cardiac operations was reviewed in an RCT of 60 surgical candidates using 10% peppermint oil via nebulization for 10 minutes.16 This technique was effective in reducing nausea during cardiac postoperative periods. Although the evidence for the use of EOs for postoperative nausea is not robust, it may be a useful and generally safe approach for this common issue.
Behavioral health
Insomnia. EOs have been used as a treatment for insomnia traditionally and in complementary, alternative, and integrative medicine. A 2014 systematic review of 15 quantitative studies, including 11 RCTs, evaluated the hypnotic effects of EOs through inhalation, finding the strongest evidence for lavender, jasmine, and peppermint oils.29 The majority of the studies in the systematic review used the Pittsburgh Sleep Quality Index (PSQI) to evaluate EO effectiveness. A more recent 2021 systematic review and meta-analysis that evaluated 34 RCTs found that inhalation of EOs, most notably lavender aromatherapy, is effective in improving sleep problems such as insomnia.30
Findings from multiple smaller RCTs were consistent with those of the aforementioned systematic reviews. For example, in a well-conducted parallel randomized double-blind placebo-controlled trial of 100 people using orally ingested lemon verbena, the authors concluded that this intervention can be a complementary therapy for improving sleep quality and reducing insomnia severity.31 Another RCT with 60 participants evaluated an inhaled EO blend (lemon, eucalyptus, tea tree, and peppermint) over 4 weeks and found lowered perceived stress and depression as well as better sleep quality, but no influence on objective physiologic data such as stress indices or immune states.32
Continue to: In a 2020 randomized crossover...
In a 2020 randomized crossover placebocontrolled trial of 37 participants with diabetes reporting insomnia, inhaled lavender improved sleep quality and quantity, quality of life, and mood but not physiologic or metabolic measures, such as fasting glucose.33 Findings were similar in a cohort of cardiac rehabilitation patients (n = 37) who were treated with either an inhaled combination of lavender, bergamot, and ylang ylang, or placebo; cotton balls infused with the intervention oil or placebo oil were placed at the patient’s bedside for 5 nights. Sleep quality of participants receiving intervention oil was significantly better than the sleep quality of participants receiving the placebo oil as measured by participant completion of the PSQI.34
Anxiety is a common disorder that can be managed with nonpharmacologic treatments such as yoga, deep breathing, meditation, and EO therapy.21,22 In a systematic review and meta-analysis, the inhaled and topical use (with or without massage) of lavender EO was shown to improve psychological and physical manifestations of anxiety.23 Lavender EO is purported to affect the parasympathetic nervous system via anxiolytic, sedative, analgesic, and anticonvulsant properties.24 One systematic review and meta-analysis evaluating the anxiolytic effect of both inhaled and topical lavender EO found improvement in several biomarkers and physiologic data including blood pressure, heart rate, and cortisol levels, as well as a reduction in self-reported levels of anxiety, compared with placebo.25
Anxiety related to dental procedures is another area of study for the use of EOs. Two RCTs demonstrate statistically significant improvement in anxiety-related physiologic markers such as heart rate, blood pressure, and salivary cortisol levels in children who inhaled lavender EO during dental procedures.41,42 In 1 of the RCTs, the intervention was described as 3 drops of 100% lavender EO applied to a cloth and inhaled over the course of 3 minutes.41 Additionally, 2 studies found that orange EO was beneficial for dental procedure–induced anxiety, reducing pulse rates, cortisol levels, and self-reported anxiety.43,44
Dementia-related behavioral disturbances. A small, poorly designed study examining 2 EO blends—rosemary with lemon and lavender with orange—found some potential for improving cognitive function, especially in patients with Alzheimer disease.45 A Cochrane review of 13 RCTs totaling 708 patients concluded that it is not certain from the available evidence that EO therapy benefits patients with dementia in long-term-care facilities and hospital wards.46 Given that reporting of adverse events in the trials was poor, it is not possible to make conclusions about the risk vs benefit of EO therapy in this population.
Women’s health
Dysmenorrhea.
Continue to: In a randomized, double-blind clinical trial...
In a randomized, double-blind clinical trial of 48 women, a cream-based blend of lavender, clary sage, and marjoram EO (used topically in a 2:1:1 ratio diluted in unscented cream at 3% concentration and applied daily via abdominal massage) reduced participants’ reported menstrual pain symptoms and duration of pain.36 In a meta-analysis of 6 studies, topical abdominal application of EO (mainly lavender with or without other oils) with massage showed superiority over massage with placebo oils in reducing menstrual pain.37 A reduction in pain, mood symptoms, and fatigue in women with premenstrual symptoms was seen in an RCT of 77 patients using 3 drops of inhaled lavender EO.47
Labor. There is limited evidence for the use of EOs during labor. In an RCT of 104 women, patient-selected diffused EOs, including lavender, rose geranium, citrus, or jasmine, were found to help lower pain scores during the latent and early active phase of labor. There were no differences in labor augmentation, length of labor, perinatal outcomes, or need for additional pain medication.48
Other uses
Antimicrobial support. Some common EOs that have demonstrated antimicrobial properties are oregano, thyme, clove, lavender, clary sage, garlic, and cinnamon.49,50 Topical lemongrass and tea tree EOs have shown some degree of efficacy as an alternative treatment for acne, decolonization of methicillin-resistant Staphylococcus aureus, and superficial fungal infections.51 Support for an oral mixture of EOs labeled Myrtol (containing eucalyptus, citrus myrtle, and lavender) for viral acute bronchitis and sinusitis was found in a review of 7 studies.52 More research needs to be done before clear recommendations can be made on the use of EOs as antimicrobials, but the current data are encouraging.
Insect repellent. Reviews of the insect-repellent properties of EOs have shown promise and are in the public’s interest due to increasing awareness of the potential health and environmental hazards of synthetic repellents.53 Individual compounds present in EOs such as citronella/lemongrass, basil, and eucalyptus species demonstrate high repellent activity.54 Since EOs require frequent reapplication for efficacy due to their highly volatile nature, scientists are currently developing a means to prolong their protection time through cream-based formulations.55
The bottom line
Because of the ubiquity of EOs, family physicians will undoubtedly be asked about them by patients, and it would be beneficial to feel comfortable discussing their most common uses. For most adult patients, the topical and periodic inhaled usage of EOs is generally safe.56
There is existing evidence of efficacy for a number of EOs, most strongly for lavender and peppermint. Future research into EOs should include higher-powered and higher-quality studies in order to provide more conclusive evidence regarding the continued use of EOs for many common conditions. More evidence-based information on dosing, application/use regimens, and safety in long-term use also will help providers better instruct patients on how to utilize EOs effectively and safely.
CORRESPONDENCE
Pooja Amy Shah, MD, Columbia University College of Physicians & Surgeons, 610 West 158th Street, New York, NY 10032; [email protected]
1. Butnariu M, Sarac I. Essential oils from plants. J Biotechnol Biomed Sci. 2018;1:35-43. doi: 10.14302/issn.2576-6694.jbbs-18-2489
2. Singh B, Sellam P, Majumder, J, et al. Floral essential oils : importance and uses for mankind. HortFlora Res Spectr. 2014;3:7-13. www.academia.edu/6707801/Floral_essential_oils_Importance_and_uses_for_mankind
3. Posadzki P, Alotaibi A, Ernst E. Adverse effects of aromatherapy: a systematic review of case reports and case series. Int J Risk Saf Med. 2012;24:147-161. doi: 10.3233/JRS-2012-0568
4. Sharmeen JB, Mahomoodally FM, Zengin G, et al. Essential oils as natural sources of fragrance compounds for cosmetics and cosmeceuticals. Molecules. 2021;26:666. doi: 10.3390/molecules26030666
5. Henley DV, Lipson N, Korach KS, et al. Prepubertal gynecomastia linked to lavender and tea tree oils. N Engl J Med. 2007;356:479-485. doi: 10.1056/NEJMoa064725
6. Nematollahi N, Weinberg JL, Flattery J, et al. Volatile chemical emissions from essential oils with therapeutic claims. Air Qual Atmosphere Health. 2021;14:365-369. doi: 10.1007/s11869-020-00941-4
7. Balekian D, Long A. Essential oil diffusers and asthma. Published February 24, 2020. Accessed September 22, 2023. www.aaaai.org/Allergist-Resources/Ask-the-Expert/Answers/Old-Ask-the-Experts/oil-diffusers-asthma
8. Aura Cacia. Quality. Accessed September 22, 2023. www.auracacia.com/quality
9. Now. Essential oil identity & purity testing. Accessed September 22, 2023. www.nowfoods.com/quality-safety/essential-oil-identity-purity-testing
10. Aura Cacia. GCMS documents. Accessed September 22, 2023. www.auracacia.com/aura-cacia-gcms-documents
11. Lopresti AL, Smith SJ, Drummond PD. Herbal treatments for migraine: a systematic review of randomised-controlled studies. Phytother Res. 2020;34:2493-2517. doi: 10.1002/ptr.6701
12. Niazi M, Hashempur MH, Taghizadeh M, et al. Efficacy of topical Rose (Rosa damascena Mill.) oil for migraine headache: A randomized double-blinded placebo-controlled cross-over trial. Complement Ther Med. 2017;34:35-41. doi: 10.1016/j.ctim. 2017.07.009
13. Rafieian-Kopaei M, Hasanpour-Dehkordi A, Lorigooini Z, et al. Comparing the effect of intranasal lidocaine 4% with peppermint essential oil drop 1.5% on migraine attacks: a double-blind clinical trial. Int J Prev Med. 2019;10:121. doi: 10.4103/ijpvm.IJPVM_530_17
14. Göbel H, Fresenius J, Heinze A, et al. [Effectiveness of Oleum menthae piperitae and paracetamol in therapy of headache of the tension type]. Nervenarzt. 1996;67:672-681. doi: 10.1007/s001150050040
15. Hunt R, Dienemann J, Norton HJ, et al. Aromatherapy as treatment for postoperative nausea: a randomized trial. Anesth Analg. 2013;117:597-604. doi: 10.1213/ANE.0b013e31824a0b1c
16. Maghami M, Afazel MR, Azizi-Fini I, et al. The effect of aromatherapy with peppermint essential oil on nausea and vomiting after cardiac surgery: a randomized clinical trial. Complement Ther Clin Pract. 2020;40:101199. doi: 10.1016/j.ctcp.2020.101199
17. Hills JM, Aaronson PI. The mechanism of action of peppermint oil on gastrointestinal smooth muscle. An analysis using patch clamp electrophysiology and isolated tissue pharmacology in rabbit and guinea pig. Gastroenterology. 1991;101:55-65. doi: 10.1016/0016-5085(91)90459-x
18. Alammar N, Wang L, Saberi B, et al. The impact of peppermint oil on the irritable bowel syndrome: a meta-analysis of the pooled clinical data. BMC Complement Altern Med. 2019;19:21. doi: 10.1186/s12906-018-2409-0
19. Cash BD, Epstein MS, Shah SM. A novel delivery system of peppermint oil is an effective therapy for irritable bowel syndrome symptoms. Dig Dis Sci. 2016;61:560-571. doi: 10.1007/s10620-015-3858-7
20. Weerts ZZRM, Masclee AAM, Witteman BJM, et al. Efficacy and safety of peppermint oil in a randomized, double-blind trial of patients with irritable bowel syndrome. Gastroenterology. 2020;158:123-136. doi: 10.1053/j.gastro.2019.08.026
21. Ma X, Yue ZQ, Gong ZQ, et al. The effect of diaphragmatic breathing on attention, negative affect and stress in healthy adults. Front Psychol. 2017;8:874. doi: 10.3389/fpsyg.2017.00874
22. Cabral P, Meyer HB, Ames D. Effectiveness of yoga therapy as a complementary treatment for major psychiatric disorders: a meta-analysis. Prim Care Companion CNS Disord. Published July 7, 2011. doi: 10.4088/PCC.10r01068
23. Donelli D, Antonelli M, Bellinazzi C, et ala. Effects of lavender on anxiety: systematic review and meta-analysis. Phytomedicine Int J Phytother Phytopharm. 2019;65:153099. doi: 10.1016/j.phymed.2019.153099
24. Koulivand PH, Khaleghi Ghadiri M, Gorji A. Lavender and the nervous system. Evid Based Complement Alternat Med. 2013;2013:1-10. doi: 10.1155/2013/681304
25. Kang HJ, Nam ES, Lee Y, et al. How strong is the evidence for the anxiolytic efficacy of lavender? Systematic review and meta-analysis of randomized controlled trials. Asian Nurs Res. 2019;13:295-305. doi: 10.1016/j.anr.2019.11.003
26. Barão Paixão VL, Freire de Carvalho J. Essential oil therapy in rheumatic diseases: a systematic review. Complement Ther Clin Pract. 2021;43:101391. doi: 10.1016/j.ctcp.2021.101391
27. Yasa Ozturk G, Bashan I. The effect of aromatherapy with lavender oil on the health-related quality of life in patients with fibromyalgia. J Food Qual. 2021;2021:1-5. doi: 10.1155/2021/9938630
28. Ko GD, Hum A, Traitses G, et al. Effects of topical O24 essential oils on patients with fibromyalgia syndrome: a randomized, placebo controlled pilot study. J Musculoskelet Pain. 2007;15:11-19. doi: 10.1300/J094v15n01_03
29. Lillehei AS, Halcon LL. A systematic review of the effect of inhaled essential oils on sleep. J Altern Complement Med. 2014;20:441-451. doi: 10.1089/acm.2013.0311
30. Cheong MJ, Kim S, Kim JS, et al. A systematic literature review and meta-analysis of the clinical effects of aroma inhalation therapy on sleep problems. Medicine (Baltimore). 2021;100:e24652. doi: 10.1097/MD.0000000000024652
31. Afrasiabian F, Mirabzadeh Ardakani M, Rahmani K, et al. Aloysia citriodora Paláu (lemon verbena) for insomnia patients: a randomized, double-blind, placebo-controlled clinical trial of efficacy and safety. Phytother Res PTR. 2019;33:350-359. doi: 10.1002/ptr.6228
32. Lee M, Lim S, Song JA, et al. The effects of aromatherapy essential oil inhalation on stress, sleep quality and immunity in healthy adults: randomized controlled trial. Eur J Integr Med. 2017;12:79-86. doi: 10.1016/j.eujim.2017.04.009
33. Nasiri Lari Z, Hajimonfarednejad M, Riasatian M, et al. Efficacy of inhaled Lavandula angustifolia Mill. Essential oil on sleep quality, quality of life and metabolic control in patients with diabetes mellitus type II and insomnia. J Ethnopharmacol. 2020;251:112560. doi: 10.1016/j.jep.2020.112560
34. McDonnell B, Newcomb P. Trial of essential oils to improve sleep for patients in cardiac rehabilitation. J Altern Complement Med N Y N. 2019;25:1193-1199. doi: 10.1089/acm.2019.0222
35. Song JA, Lee MK, Min E, et al. Effects of aromatherapy on dysmenorrhea: a systematic review and meta-analysis. Int J Nurs Stud. 2018;84:1-11. doi: 10.1016/j.ijnurstu.2018.01.016
36. Ou MC, Hsu TF, Lai AC, et al. Pain relief assessment by aromatic essential oil massage on outpatients with primary dysmenorrhea: a randomized, double-blind clinical trial: PD pain relief by aromatic oil massage. J Obstet Gynaecol Res. 2012;38:817-822. doi: 10.1111/j.1447-0756.2011.01802.x
37. Sut N, Kahyaoglu-Sut H. Effect of aromatherapy massage on pain in primary dysmenorrhea: a meta-analysis. Complement Ther Clin Pract. 2017;27:5-10. doi: 10.1016/j.ctcp.2017.01.001
38. Keyhanmehr AS, Kolouri S, Heydarirad G, et al. Aromatherapy for the management of cancer complications: a narrative review. Complement Ther Clin Pract. 2018;31:175-180. doi: 10.1016/j.ctcp.2018.02.009
39. Sriningsih I, Elisa E, Lestari KP. Aromatherapy ginger use in patients with nausea & vomiting on post cervical cancer chemotherapy. KEMAS J Kesehat Masy. 2017;13:59-68. doi: 10.15294/kemas.v13i1.5367
40. Lua PL, Salihah N, Mazlan N. Effects of inhaled ginger aromatherapy on chemotherapy-induced nausea and vomiting and health-related quality of life in women with breast cancer. Complement Ther Med. 2015;23:396-404. doi: 10.1016/j.ctim.2015.03.009
41. Arslan I, Aydinoglu S, Karan NB. Can lavender oil inhalation help to overcome dental anxiety and pain in children? A randomized clinical trial. Eur J Pediatr. 2020;179:985-992. doi: 10.1007/s00431-020-03595-7
42. Ghaderi F, Solhjou N. The effects of lavender aromatherapy on stress and pain perception in children during dental treatment: a randomized clinical trial. Complement Ther Clin Pract. 2020;40:101182. doi: 10.1016/j.ctcp.2020.101182
43. Jafarzadeh M, Arman S, Pour FF. Effect of aromatherapy with orange essential oil on salivary cortisol and pulse rate in children during dental treatment: a randomized controlled clinical trial. Adv Biomed Res. 2013;2:10. doi: 10.4103/2277-9175.107968
44. Lehrner J, Eckersberger C, Walla P, et al. Ambient odor of orange in a dental office reduces anxiety and improves mood in female patients. Physiol Behav. 2000;71:83-86. doi: 10.1016/S0031-9384(00)00308-5
45. Jimbo D, Kimura Y, Taniguchi M, et al. Effect of aromatherapy on patients with Alzheimer’s disease. Psychogeriatrics. 2009;9:173-179. doi: 10.1111/j.1479-8301.2009.00299.x
46. Ball EL, Owen-Booth B, Gray A, et al. Aromatherapy for dementia. Cochrane Database Syst Rev. 2020;(8). doi: 10.1002/14651858.CD003150.pub3
47. Uzunçakmak T, Ayaz Alkaya S. Effect of aromatherapy on coping with premenstrual syndrome: a randomized controlled trial. Complement Ther Med. 2018;36:63-67. doi: 10.1016/j.ctim.2017.11.022
48. Tanvisut R, Traisrisilp K, Tongsong T. Efficacy of aromatherapy for reducing pain during labor: a randomized controlled trial. Arch Gynecol Obstet. 2018;297:1145-1150. doi: 10.1007/s00404-018-4700-1
49. Ramsey JT, Shropshire BC, Nagy TR, et al. Essential oils and health. Yale J Biol Med. 2020;93:291-305.
50. Puškárová A, Bučková M, Kraková L, et al. The antibacterial and antifungal activity of six essential oils and their cyto/genotoxicity to human HEL 12469 cells. Sci Rep. 2017;7:8211. doi: 10.1038/s41598-017-08673-9
51. Deyno S, Mtewa AG, Abebe A, et al. Essential oils as topical anti-infective agents: a systematic review and meta-analysis. Complement Ther Med. 2019;47:102224. doi: 10.1016/j.ctim.2019.102224
52. Prall S, Bowles EJ, Bennett K, et al. Effects of essential oils on symptoms and course (duration and severity) of viral respiratory infections in humans: a rapid review. Adv Integr Med. 2020;7:218-221. doi: 10.1016/j.aimed.2020.07.005
53. Weeks JA, Guiney PD, Nikiforov AI. Assessment of the environmental fate and ecotoxicity of N,N-diethyl-m-toluamide (DEET). Integr Environ Assess Manag. 2012;8:120-134. doi: 10.1002/ieam.1246
54. Nerio LS, Olivero-Verbel J, Stashenko E. Repellent activity of essential oils: a review. Bioresour Technol. 2010;101:372-378. doi: 10.1016/j.biortech.2009.07.048
55. Lee MY. Essential oils as repellents against arthropods. BioMed Res Int. 2018;2018:6860271. doi: 10.1155/2018/6860271
56. Göbel H, Heinze A, Heinze-Kuhn K, et al. [Peppermint oil in the acute treatment of tension-type headache]. Schmerz Berl Ger. 2016;30:295-310. doi: 10.1007/s00482-016-0109-6
Essential oils (EOs), which are concentrated plant-based oils, have become ubiquitous over the past decade. Given the far reach of EOs and their longtime use in traditional, complementary, alternative, and integrative medicine, it is imperative that clinicians have some knowledge of the potential benefits, risks, and overall efficacy.
Commonly used for aromatic benefits (aromatherapy), EOs are now also incorporated into a multitude of products promoting health and wellness. EOs are sold as individual products and can be a component in consumer goods such as cosmetics, body care/hygiene/beauty products, laundry detergents, insect repellents, over-the-counter medications, and food.
The review that follows presents the most current evidence available. With that said, it’s important to keep in mind some caveats that relate to this evidence. First, the studies cited tend to have a small sample size. Second, a majority of these studies were conducted in countries where there appears to be a significant culture of EO use, which could contribute to confirmation bias. Finally, in a number of the studies, there is concern for publication bias as well as a discrepancy between calculated statistical significance and actual clinical relevance.

What are essential oils?
EOs generally are made by extracting the oil from leaves, bark, flowers, seeds/fruit, rinds, and/or roots by steaming or pressing parts of a plant. It can take several pounds of plant material to produce a single bottle of EO, which usually contains ≥ 15 to 30 mL (.5 to 1 oz).1
Some commonly used EOs in the United States are lavender, peppermint, rose, clary sage, tea tree, eucalyptus, and citrus; however, there are approximately 300 EOs available.2 EOs are used most often via topical application, inhalation, or ingestion.
As with any botanical agent, EOs are complex substances often containing a multitude of chemical compounds.1 Because of the complex makeup of EOs, which often contain up to 100 volatile organic compounds, and their wide-ranging potential effects, applying the scientific method to study effectiveness poses a challenge that has limited their adoption in evidence-based practice.2
Availability and cost. EOs can be purchased at large retailers (eg, grocery stores, drug stores) and smaller health food stores, as well as on the Internet. Various EO vehicles, such as inhalers and topical creams, also can be purchased at these stores.
Continue to: The cost varies...
The cost varies enormously by manufacturer and type of plant used to make the EO. Common EOs such as peppermint and lavender oil generally cost $10 to $25, while rarer plant oils can cost $80 or more per bottle.
How safe are essential oils?
Patients may assume EOs are harmless because they are derived from natural plants and have been used medicinally for centuries. However, care must be taken with their use.
The safest way to use EOs is topically, although due to their highly concentrated nature, EOs should be diluted in an unscented neutral carrier oil such as coconut, jojoba, olive, or sweet almond.3 Ingestion of certain oils can cause hepatotoxicity, seizures, and even death.3 In fact, patients should speak with a knowledgeable physician before purchasing any oral EO capsules.
Whether used topically or ingested, all EOs carry risk for skin irritation and allergic reactions, and oral ingestion may result in some negative gastrointestinal (GI) adverse effects.4 A case report of 3 patients published in 2007 identified the potential for lavender and tea tree EOs to be endocrine disruptors.5
Inhalation of EOs may be harmful, as they emit many volatile organic compounds, some of which are considered potentially hazardous.6 At this time, there is insufficient evidence regarding inhaled EOs and their direct connection to respiratory health. It is reasonable to suggest, however, that the prolonged use of EOs and their use by patients who have lung conditions such as asthma or COPD should be avoided.7
Continue to: How are quality and purity assessed?
How are quality and purity assessed?
Like other dietary supplements, EOs are not regulated. No US regulatory agencies (eg, the US Food and Drug Administration [FDA] or Department of Agriculture [USDA]) certify or approve EOs for quality and purity. Bottles labeled with “QAI” for Quality Assurance International or “USDA Organic” will ensure the plant constituents used in the EO are from organic farming but do not attest to quality or purity.
Manufacturers commonly use marketing terms such as “therapeutic grade” or “pure” to sell products, but again, these terms do not reflect the product’s quality or purity. A labeled single EO may contain contaminants, alcohol, or additional ingredients.7 When choosing to use EOs, identifying reputable brands is essential; one resource is the independent testing organization ConsumerLab.com.
It is important to assess the manufacturer and read ingredient labels before purchasing an EO to understand what the product contains. Reputable companies will identify the plant ingredient, usually by the formal Latin binomial name, and explain the extraction process. A more certain way to assess the quality and purity of an EO is to ask the manufacturer to provide a certificate of analysis and gas chromatography/mass spectroscopy (GC/MS) data for the specific product. Some manufacturers offer GC/MS test results on their website Quality page.8 Others have detailed information on quality and testing, and GC/MS test reports can be obtained.9 Yet another manufacturer has test results on a product page matching reports to batch codes.10
Which conditions have evidence of benefit from essential oils?
EOs currently are being studied for treatment of many conditions—including pain, GI disorders, behavioral health disorders, and women’s health issues. The TABLE summarizes the conditions treated, outcomes, and practical applications of EOs.11-44

Pain
Headache. As an adjunct to available medications and procedures for headache treatment, EOs are one of the nonpharmacologic modalities that patients and clinicians have at their disposal for both migraine and tension-type headaches. A systematic review of 19 randomized controlled trials (RCTs) examining the effects of herbal ingredients for the acute treatment or prophylaxis of migraines found certain topically applied or inhaled EOs, such as peppermint and chamomile, to be effective for migraine pain alleviation; however, topically applied rose oil was not effective.11-13 Note: “topical application” in these studies implies application of the EO to ≥ 1 of the following areas: temples, forehead, behind ears, or above upper lip/below the nose.
Continue to: One RCT with 120 patients...
One RCT with 120 patients evaluated diluted intranasal peppermint oil and found that it reduced migraine intensity at similar rates to intranasal lidocaine.13 In this study, patients were randomized to receive one of the following: 4% lidocaine, 1.5% peppermint EO, or placebo. Two drops of the intranasal intervention were self-administered while the patient was in a supine position with their head suspended off the edge of the surface on which they were lying. They were instructed to stay in this position for at least 30 seconds after administration.
With regard to tension headache treatment, there is limited literature on the use of EOs. One study found that a preparation of peppermint oil applied topically to the temples and forehead of study participants resulted in significant analgesic effect.14
Fibromyalgia. Usual treatments for fibromyalgia include exercise, antidepressant and anticonvulsant medications, and stress management. Evidence also supports the use of inhaled and topically applied (with and without massage) lavender oil to improve symptoms.26 Positive effects may be related to the analgesic, anti-inflammatory, sleep-regulating, and anxiety-reducing effects of the major volatile compounds contained in lavender oil.
In one RCT with 42 patients with fibromyalgia, the use of inhaled lavender oil was shown to increase the perception of well-being (assessed on the validated SF-36 Health Survey Questionnaire) after 4 weeks.27 In this study, the patient applied 3 drops of an oil mixture, comprising 1 mL lavender EO and 10 mL of fixed neutral base oil, to the wrist and inhaled for 10 minutes before going to bed.
The use of a topical oil blend labeled “Oil 24” (containing camphor, rosemary, eucalyptus, peppermint, aloe vera, and lemon/orange) also has been shown to be more effective than placebo in managing fibromyalgia symptoms. A randomized controlled pilot study of 153 participants found that regular application of Oil 24 improved scores on pain scales and the Fibromyalgia Impact Questionnaire.28
Continue to: GI disorders
GI disorders
Irritable bowel syndrome. Peppermint oil relaxes GI smooth muscle, which has led to investigation of its use in irritable bowel syndrome (IBS) symptom amelioration.17 One meta-analysis including 12 RCTs with 835 patients with undifferentiated IBS found that orally ingested peppermint EO capsules reduced patient-reported symptoms of either abdominal pain or global symptoms.18
One study utilized the Total IBS Symptom Score to evaluate symptom reduction in patients with IBS-D (with diarrhea) and IBS-M (mixed) using 180-mg peppermint EO capsules ingested 3 times daily. There was a significant improvement in abdominal pain/discomfort, bloating/distension, pain at evacuation, and bowel urgency.19 A reduction in symptoms was observed after the first 24 hours of treatment and at the end of the 4-week treatment period.
In another study, among the 190 patients meeting Rome IV criteria for general (nonspecific) IBS who were treated with 182-mg peppermint EO capsules, no statistically significant reduction in overall symptom relief was found (based on outcome measures by the FDA and European Medicines Agency). However, in a secondary outcome analysis, peppermint oil produced greater improvements than placebo for the alleviation of abdominal pain, discomfort, and general IBS severity.20
Chemotherapy-induced nausea and vomiting. Patients with cancer undergoing chemotherapy often explore integrative medicine approaches, including aromatherapy, to ameliorate adverse effects and improve quality of life.38 A few small studies have shown potential for the use of inhaled ginger oil to reduce nausea and vomiting severity and improve health-related quality-of-life measures in these patients.
For example, a study with 60 participants found that inhaling ginger EO for 10 minutes was beneficial for reducing both nausea and vomiting.39 A single-blind, controlled, randomized crossover study of 60 patients with breast cancer receiving chemotherapy showed that ginger EO inhaled 3 times per day for 2 minutes at a time can decrease the severity of nausea but had no effect on vomiting. The same study showed that health-related quality of life improved with the ginger oil treatment.40
Continue to: Other EOs such as cardamom...
Other EOs such as cardamom and peppermint show promise as an adjunctive treatment for chemotherapy-induced nausea and vomiting as well.38
Postoperative nausea. A 2013 randomized trial of 303 patients examined the use of ginger EO, a blend of EOs (including ginger, spearmint, peppermint, and cardamom), and isopropyl alcohol. Both the single EO and EO blend significantly reduced the symptom of nausea. The number of antiemetic medications requested by patients receiving an EO also was significantly reduced compared to those receiving saline.15
The use of EOs to reduce nausea after cardiac operations was reviewed in an RCT of 60 surgical candidates using 10% peppermint oil via nebulization for 10 minutes.16 This technique was effective in reducing nausea during cardiac postoperative periods. Although the evidence for the use of EOs for postoperative nausea is not robust, it may be a useful and generally safe approach for this common issue.
Behavioral health
Insomnia. EOs have been used as a treatment for insomnia traditionally and in complementary, alternative, and integrative medicine. A 2014 systematic review of 15 quantitative studies, including 11 RCTs, evaluated the hypnotic effects of EOs through inhalation, finding the strongest evidence for lavender, jasmine, and peppermint oils.29 The majority of the studies in the systematic review used the Pittsburgh Sleep Quality Index (PSQI) to evaluate EO effectiveness. A more recent 2021 systematic review and meta-analysis that evaluated 34 RCTs found that inhalation of EOs, most notably lavender aromatherapy, is effective in improving sleep problems such as insomnia.30
Findings from multiple smaller RCTs were consistent with those of the aforementioned systematic reviews. For example, in a well-conducted parallel randomized double-blind placebo-controlled trial of 100 people using orally ingested lemon verbena, the authors concluded that this intervention can be a complementary therapy for improving sleep quality and reducing insomnia severity.31 Another RCT with 60 participants evaluated an inhaled EO blend (lemon, eucalyptus, tea tree, and peppermint) over 4 weeks and found lowered perceived stress and depression as well as better sleep quality, but no influence on objective physiologic data such as stress indices or immune states.32
Continue to: In a 2020 randomized crossover...
In a 2020 randomized crossover placebocontrolled trial of 37 participants with diabetes reporting insomnia, inhaled lavender improved sleep quality and quantity, quality of life, and mood but not physiologic or metabolic measures, such as fasting glucose.33 Findings were similar in a cohort of cardiac rehabilitation patients (n = 37) who were treated with either an inhaled combination of lavender, bergamot, and ylang ylang, or placebo; cotton balls infused with the intervention oil or placebo oil were placed at the patient’s bedside for 5 nights. Sleep quality of participants receiving intervention oil was significantly better than the sleep quality of participants receiving the placebo oil as measured by participant completion of the PSQI.34
Anxiety is a common disorder that can be managed with nonpharmacologic treatments such as yoga, deep breathing, meditation, and EO therapy.21,22 In a systematic review and meta-analysis, the inhaled and topical use (with or without massage) of lavender EO was shown to improve psychological and physical manifestations of anxiety.23 Lavender EO is purported to affect the parasympathetic nervous system via anxiolytic, sedative, analgesic, and anticonvulsant properties.24 One systematic review and meta-analysis evaluating the anxiolytic effect of both inhaled and topical lavender EO found improvement in several biomarkers and physiologic data including blood pressure, heart rate, and cortisol levels, as well as a reduction in self-reported levels of anxiety, compared with placebo.25
Anxiety related to dental procedures is another area of study for the use of EOs. Two RCTs demonstrate statistically significant improvement in anxiety-related physiologic markers such as heart rate, blood pressure, and salivary cortisol levels in children who inhaled lavender EO during dental procedures.41,42 In 1 of the RCTs, the intervention was described as 3 drops of 100% lavender EO applied to a cloth and inhaled over the course of 3 minutes.41 Additionally, 2 studies found that orange EO was beneficial for dental procedure–induced anxiety, reducing pulse rates, cortisol levels, and self-reported anxiety.43,44
Dementia-related behavioral disturbances. A small, poorly designed study examining 2 EO blends—rosemary with lemon and lavender with orange—found some potential for improving cognitive function, especially in patients with Alzheimer disease.45 A Cochrane review of 13 RCTs totaling 708 patients concluded that it is not certain from the available evidence that EO therapy benefits patients with dementia in long-term-care facilities and hospital wards.46 Given that reporting of adverse events in the trials was poor, it is not possible to make conclusions about the risk vs benefit of EO therapy in this population.
Women’s health
Dysmenorrhea.
Continue to: In a randomized, double-blind clinical trial...
In a randomized, double-blind clinical trial of 48 women, a cream-based blend of lavender, clary sage, and marjoram EO (used topically in a 2:1:1 ratio diluted in unscented cream at 3% concentration and applied daily via abdominal massage) reduced participants’ reported menstrual pain symptoms and duration of pain.36 In a meta-analysis of 6 studies, topical abdominal application of EO (mainly lavender with or without other oils) with massage showed superiority over massage with placebo oils in reducing menstrual pain.37 A reduction in pain, mood symptoms, and fatigue in women with premenstrual symptoms was seen in an RCT of 77 patients using 3 drops of inhaled lavender EO.47
Labor. There is limited evidence for the use of EOs during labor. In an RCT of 104 women, patient-selected diffused EOs, including lavender, rose geranium, citrus, or jasmine, were found to help lower pain scores during the latent and early active phase of labor. There were no differences in labor augmentation, length of labor, perinatal outcomes, or need for additional pain medication.48
Other uses
Antimicrobial support. Some common EOs that have demonstrated antimicrobial properties are oregano, thyme, clove, lavender, clary sage, garlic, and cinnamon.49,50 Topical lemongrass and tea tree EOs have shown some degree of efficacy as an alternative treatment for acne, decolonization of methicillin-resistant Staphylococcus aureus, and superficial fungal infections.51 Support for an oral mixture of EOs labeled Myrtol (containing eucalyptus, citrus myrtle, and lavender) for viral acute bronchitis and sinusitis was found in a review of 7 studies.52 More research needs to be done before clear recommendations can be made on the use of EOs as antimicrobials, but the current data are encouraging.
Insect repellent. Reviews of the insect-repellent properties of EOs have shown promise and are in the public’s interest due to increasing awareness of the potential health and environmental hazards of synthetic repellents.53 Individual compounds present in EOs such as citronella/lemongrass, basil, and eucalyptus species demonstrate high repellent activity.54 Since EOs require frequent reapplication for efficacy due to their highly volatile nature, scientists are currently developing a means to prolong their protection time through cream-based formulations.55
The bottom line
Because of the ubiquity of EOs, family physicians will undoubtedly be asked about them by patients, and it would be beneficial to feel comfortable discussing their most common uses. For most adult patients, the topical and periodic inhaled usage of EOs is generally safe.56
There is existing evidence of efficacy for a number of EOs, most strongly for lavender and peppermint. Future research into EOs should include higher-powered and higher-quality studies in order to provide more conclusive evidence regarding the continued use of EOs for many common conditions. More evidence-based information on dosing, application/use regimens, and safety in long-term use also will help providers better instruct patients on how to utilize EOs effectively and safely.
CORRESPONDENCE
Pooja Amy Shah, MD, Columbia University College of Physicians & Surgeons, 610 West 158th Street, New York, NY 10032; [email protected]
Essential oils (EOs), which are concentrated plant-based oils, have become ubiquitous over the past decade. Given the far reach of EOs and their longtime use in traditional, complementary, alternative, and integrative medicine, it is imperative that clinicians have some knowledge of the potential benefits, risks, and overall efficacy.
Commonly used for aromatic benefits (aromatherapy), EOs are now also incorporated into a multitude of products promoting health and wellness. EOs are sold as individual products and can be a component in consumer goods such as cosmetics, body care/hygiene/beauty products, laundry detergents, insect repellents, over-the-counter medications, and food.
The review that follows presents the most current evidence available. With that said, it’s important to keep in mind some caveats that relate to this evidence. First, the studies cited tend to have a small sample size. Second, a majority of these studies were conducted in countries where there appears to be a significant culture of EO use, which could contribute to confirmation bias. Finally, in a number of the studies, there is concern for publication bias as well as a discrepancy between calculated statistical significance and actual clinical relevance.

What are essential oils?
EOs generally are made by extracting the oil from leaves, bark, flowers, seeds/fruit, rinds, and/or roots by steaming or pressing parts of a plant. It can take several pounds of plant material to produce a single bottle of EO, which usually contains ≥ 15 to 30 mL (.5 to 1 oz).1
Some commonly used EOs in the United States are lavender, peppermint, rose, clary sage, tea tree, eucalyptus, and citrus; however, there are approximately 300 EOs available.2 EOs are used most often via topical application, inhalation, or ingestion.
As with any botanical agent, EOs are complex substances often containing a multitude of chemical compounds.1 Because of the complex makeup of EOs, which often contain up to 100 volatile organic compounds, and their wide-ranging potential effects, applying the scientific method to study effectiveness poses a challenge that has limited their adoption in evidence-based practice.2
Availability and cost. EOs can be purchased at large retailers (eg, grocery stores, drug stores) and smaller health food stores, as well as on the Internet. Various EO vehicles, such as inhalers and topical creams, also can be purchased at these stores.
Continue to: The cost varies...
The cost varies enormously by manufacturer and type of plant used to make the EO. Common EOs such as peppermint and lavender oil generally cost $10 to $25, while rarer plant oils can cost $80 or more per bottle.
How safe are essential oils?
Patients may assume EOs are harmless because they are derived from natural plants and have been used medicinally for centuries. However, care must be taken with their use.
The safest way to use EOs is topically, although due to their highly concentrated nature, EOs should be diluted in an unscented neutral carrier oil such as coconut, jojoba, olive, or sweet almond.3 Ingestion of certain oils can cause hepatotoxicity, seizures, and even death.3 In fact, patients should speak with a knowledgeable physician before purchasing any oral EO capsules.
Whether used topically or ingested, all EOs carry risk for skin irritation and allergic reactions, and oral ingestion may result in some negative gastrointestinal (GI) adverse effects.4 A case report of 3 patients published in 2007 identified the potential for lavender and tea tree EOs to be endocrine disruptors.5
Inhalation of EOs may be harmful, as they emit many volatile organic compounds, some of which are considered potentially hazardous.6 At this time, there is insufficient evidence regarding inhaled EOs and their direct connection to respiratory health. It is reasonable to suggest, however, that the prolonged use of EOs and their use by patients who have lung conditions such as asthma or COPD should be avoided.7
Continue to: How are quality and purity assessed?
How are quality and purity assessed?
Like other dietary supplements, EOs are not regulated. No US regulatory agencies (eg, the US Food and Drug Administration [FDA] or Department of Agriculture [USDA]) certify or approve EOs for quality and purity. Bottles labeled with “QAI” for Quality Assurance International or “USDA Organic” will ensure the plant constituents used in the EO are from organic farming but do not attest to quality or purity.
Manufacturers commonly use marketing terms such as “therapeutic grade” or “pure” to sell products, but again, these terms do not reflect the product’s quality or purity. A labeled single EO may contain contaminants, alcohol, or additional ingredients.7 When choosing to use EOs, identifying reputable brands is essential; one resource is the independent testing organization ConsumerLab.com.
It is important to assess the manufacturer and read ingredient labels before purchasing an EO to understand what the product contains. Reputable companies will identify the plant ingredient, usually by the formal Latin binomial name, and explain the extraction process. A more certain way to assess the quality and purity of an EO is to ask the manufacturer to provide a certificate of analysis and gas chromatography/mass spectroscopy (GC/MS) data for the specific product. Some manufacturers offer GC/MS test results on their website Quality page.8 Others have detailed information on quality and testing, and GC/MS test reports can be obtained.9 Yet another manufacturer has test results on a product page matching reports to batch codes.10
Which conditions have evidence of benefit from essential oils?
EOs currently are being studied for treatment of many conditions—including pain, GI disorders, behavioral health disorders, and women’s health issues. The TABLE summarizes the conditions treated, outcomes, and practical applications of EOs.11-44

Pain
Headache. As an adjunct to available medications and procedures for headache treatment, EOs are one of the nonpharmacologic modalities that patients and clinicians have at their disposal for both migraine and tension-type headaches. A systematic review of 19 randomized controlled trials (RCTs) examining the effects of herbal ingredients for the acute treatment or prophylaxis of migraines found certain topically applied or inhaled EOs, such as peppermint and chamomile, to be effective for migraine pain alleviation; however, topically applied rose oil was not effective.11-13 Note: “topical application” in these studies implies application of the EO to ≥ 1 of the following areas: temples, forehead, behind ears, or above upper lip/below the nose.
Continue to: One RCT with 120 patients...
One RCT with 120 patients evaluated diluted intranasal peppermint oil and found that it reduced migraine intensity at similar rates to intranasal lidocaine.13 In this study, patients were randomized to receive one of the following: 4% lidocaine, 1.5% peppermint EO, or placebo. Two drops of the intranasal intervention were self-administered while the patient was in a supine position with their head suspended off the edge of the surface on which they were lying. They were instructed to stay in this position for at least 30 seconds after administration.
With regard to tension headache treatment, there is limited literature on the use of EOs. One study found that a preparation of peppermint oil applied topically to the temples and forehead of study participants resulted in significant analgesic effect.14
Fibromyalgia. Usual treatments for fibromyalgia include exercise, antidepressant and anticonvulsant medications, and stress management. Evidence also supports the use of inhaled and topically applied (with and without massage) lavender oil to improve symptoms.26 Positive effects may be related to the analgesic, anti-inflammatory, sleep-regulating, and anxiety-reducing effects of the major volatile compounds contained in lavender oil.
In one RCT with 42 patients with fibromyalgia, the use of inhaled lavender oil was shown to increase the perception of well-being (assessed on the validated SF-36 Health Survey Questionnaire) after 4 weeks.27 In this study, the patient applied 3 drops of an oil mixture, comprising 1 mL lavender EO and 10 mL of fixed neutral base oil, to the wrist and inhaled for 10 minutes before going to bed.
The use of a topical oil blend labeled “Oil 24” (containing camphor, rosemary, eucalyptus, peppermint, aloe vera, and lemon/orange) also has been shown to be more effective than placebo in managing fibromyalgia symptoms. A randomized controlled pilot study of 153 participants found that regular application of Oil 24 improved scores on pain scales and the Fibromyalgia Impact Questionnaire.28
Continue to: GI disorders
GI disorders
Irritable bowel syndrome. Peppermint oil relaxes GI smooth muscle, which has led to investigation of its use in irritable bowel syndrome (IBS) symptom amelioration.17 One meta-analysis including 12 RCTs with 835 patients with undifferentiated IBS found that orally ingested peppermint EO capsules reduced patient-reported symptoms of either abdominal pain or global symptoms.18
One study utilized the Total IBS Symptom Score to evaluate symptom reduction in patients with IBS-D (with diarrhea) and IBS-M (mixed) using 180-mg peppermint EO capsules ingested 3 times daily. There was a significant improvement in abdominal pain/discomfort, bloating/distension, pain at evacuation, and bowel urgency.19 A reduction in symptoms was observed after the first 24 hours of treatment and at the end of the 4-week treatment period.
In another study, among the 190 patients meeting Rome IV criteria for general (nonspecific) IBS who were treated with 182-mg peppermint EO capsules, no statistically significant reduction in overall symptom relief was found (based on outcome measures by the FDA and European Medicines Agency). However, in a secondary outcome analysis, peppermint oil produced greater improvements than placebo for the alleviation of abdominal pain, discomfort, and general IBS severity.20
Chemotherapy-induced nausea and vomiting. Patients with cancer undergoing chemotherapy often explore integrative medicine approaches, including aromatherapy, to ameliorate adverse effects and improve quality of life.38 A few small studies have shown potential for the use of inhaled ginger oil to reduce nausea and vomiting severity and improve health-related quality-of-life measures in these patients.
For example, a study with 60 participants found that inhaling ginger EO for 10 minutes was beneficial for reducing both nausea and vomiting.39 A single-blind, controlled, randomized crossover study of 60 patients with breast cancer receiving chemotherapy showed that ginger EO inhaled 3 times per day for 2 minutes at a time can decrease the severity of nausea but had no effect on vomiting. The same study showed that health-related quality of life improved with the ginger oil treatment.40
Continue to: Other EOs such as cardamom...
Other EOs such as cardamom and peppermint show promise as an adjunctive treatment for chemotherapy-induced nausea and vomiting as well.38
Postoperative nausea. A 2013 randomized trial of 303 patients examined the use of ginger EO, a blend of EOs (including ginger, spearmint, peppermint, and cardamom), and isopropyl alcohol. Both the single EO and EO blend significantly reduced the symptom of nausea. The number of antiemetic medications requested by patients receiving an EO also was significantly reduced compared to those receiving saline.15
The use of EOs to reduce nausea after cardiac operations was reviewed in an RCT of 60 surgical candidates using 10% peppermint oil via nebulization for 10 minutes.16 This technique was effective in reducing nausea during cardiac postoperative periods. Although the evidence for the use of EOs for postoperative nausea is not robust, it may be a useful and generally safe approach for this common issue.
Behavioral health
Insomnia. EOs have been used as a treatment for insomnia traditionally and in complementary, alternative, and integrative medicine. A 2014 systematic review of 15 quantitative studies, including 11 RCTs, evaluated the hypnotic effects of EOs through inhalation, finding the strongest evidence for lavender, jasmine, and peppermint oils.29 The majority of the studies in the systematic review used the Pittsburgh Sleep Quality Index (PSQI) to evaluate EO effectiveness. A more recent 2021 systematic review and meta-analysis that evaluated 34 RCTs found that inhalation of EOs, most notably lavender aromatherapy, is effective in improving sleep problems such as insomnia.30
Findings from multiple smaller RCTs were consistent with those of the aforementioned systematic reviews. For example, in a well-conducted parallel randomized double-blind placebo-controlled trial of 100 people using orally ingested lemon verbena, the authors concluded that this intervention can be a complementary therapy for improving sleep quality and reducing insomnia severity.31 Another RCT with 60 participants evaluated an inhaled EO blend (lemon, eucalyptus, tea tree, and peppermint) over 4 weeks and found lowered perceived stress and depression as well as better sleep quality, but no influence on objective physiologic data such as stress indices or immune states.32
Continue to: In a 2020 randomized crossover...
In a 2020 randomized crossover placebocontrolled trial of 37 participants with diabetes reporting insomnia, inhaled lavender improved sleep quality and quantity, quality of life, and mood but not physiologic or metabolic measures, such as fasting glucose.33 Findings were similar in a cohort of cardiac rehabilitation patients (n = 37) who were treated with either an inhaled combination of lavender, bergamot, and ylang ylang, or placebo; cotton balls infused with the intervention oil or placebo oil were placed at the patient’s bedside for 5 nights. Sleep quality of participants receiving intervention oil was significantly better than the sleep quality of participants receiving the placebo oil as measured by participant completion of the PSQI.34
Anxiety is a common disorder that can be managed with nonpharmacologic treatments such as yoga, deep breathing, meditation, and EO therapy.21,22 In a systematic review and meta-analysis, the inhaled and topical use (with or without massage) of lavender EO was shown to improve psychological and physical manifestations of anxiety.23 Lavender EO is purported to affect the parasympathetic nervous system via anxiolytic, sedative, analgesic, and anticonvulsant properties.24 One systematic review and meta-analysis evaluating the anxiolytic effect of both inhaled and topical lavender EO found improvement in several biomarkers and physiologic data including blood pressure, heart rate, and cortisol levels, as well as a reduction in self-reported levels of anxiety, compared with placebo.25
Anxiety related to dental procedures is another area of study for the use of EOs. Two RCTs demonstrate statistically significant improvement in anxiety-related physiologic markers such as heart rate, blood pressure, and salivary cortisol levels in children who inhaled lavender EO during dental procedures.41,42 In 1 of the RCTs, the intervention was described as 3 drops of 100% lavender EO applied to a cloth and inhaled over the course of 3 minutes.41 Additionally, 2 studies found that orange EO was beneficial for dental procedure–induced anxiety, reducing pulse rates, cortisol levels, and self-reported anxiety.43,44
Dementia-related behavioral disturbances. A small, poorly designed study examining 2 EO blends—rosemary with lemon and lavender with orange—found some potential for improving cognitive function, especially in patients with Alzheimer disease.45 A Cochrane review of 13 RCTs totaling 708 patients concluded that it is not certain from the available evidence that EO therapy benefits patients with dementia in long-term-care facilities and hospital wards.46 Given that reporting of adverse events in the trials was poor, it is not possible to make conclusions about the risk vs benefit of EO therapy in this population.
Women’s health
Dysmenorrhea.
Continue to: In a randomized, double-blind clinical trial...
In a randomized, double-blind clinical trial of 48 women, a cream-based blend of lavender, clary sage, and marjoram EO (used topically in a 2:1:1 ratio diluted in unscented cream at 3% concentration and applied daily via abdominal massage) reduced participants’ reported menstrual pain symptoms and duration of pain.36 In a meta-analysis of 6 studies, topical abdominal application of EO (mainly lavender with or without other oils) with massage showed superiority over massage with placebo oils in reducing menstrual pain.37 A reduction in pain, mood symptoms, and fatigue in women with premenstrual symptoms was seen in an RCT of 77 patients using 3 drops of inhaled lavender EO.47
Labor. There is limited evidence for the use of EOs during labor. In an RCT of 104 women, patient-selected diffused EOs, including lavender, rose geranium, citrus, or jasmine, were found to help lower pain scores during the latent and early active phase of labor. There were no differences in labor augmentation, length of labor, perinatal outcomes, or need for additional pain medication.48
Other uses
Antimicrobial support. Some common EOs that have demonstrated antimicrobial properties are oregano, thyme, clove, lavender, clary sage, garlic, and cinnamon.49,50 Topical lemongrass and tea tree EOs have shown some degree of efficacy as an alternative treatment for acne, decolonization of methicillin-resistant Staphylococcus aureus, and superficial fungal infections.51 Support for an oral mixture of EOs labeled Myrtol (containing eucalyptus, citrus myrtle, and lavender) for viral acute bronchitis and sinusitis was found in a review of 7 studies.52 More research needs to be done before clear recommendations can be made on the use of EOs as antimicrobials, but the current data are encouraging.
Insect repellent. Reviews of the insect-repellent properties of EOs have shown promise and are in the public’s interest due to increasing awareness of the potential health and environmental hazards of synthetic repellents.53 Individual compounds present in EOs such as citronella/lemongrass, basil, and eucalyptus species demonstrate high repellent activity.54 Since EOs require frequent reapplication for efficacy due to their highly volatile nature, scientists are currently developing a means to prolong their protection time through cream-based formulations.55
The bottom line
Because of the ubiquity of EOs, family physicians will undoubtedly be asked about them by patients, and it would be beneficial to feel comfortable discussing their most common uses. For most adult patients, the topical and periodic inhaled usage of EOs is generally safe.56
There is existing evidence of efficacy for a number of EOs, most strongly for lavender and peppermint. Future research into EOs should include higher-powered and higher-quality studies in order to provide more conclusive evidence regarding the continued use of EOs for many common conditions. More evidence-based information on dosing, application/use regimens, and safety in long-term use also will help providers better instruct patients on how to utilize EOs effectively and safely.
CORRESPONDENCE
Pooja Amy Shah, MD, Columbia University College of Physicians & Surgeons, 610 West 158th Street, New York, NY 10032; [email protected]
1. Butnariu M, Sarac I. Essential oils from plants. J Biotechnol Biomed Sci. 2018;1:35-43. doi: 10.14302/issn.2576-6694.jbbs-18-2489
2. Singh B, Sellam P, Majumder, J, et al. Floral essential oils : importance and uses for mankind. HortFlora Res Spectr. 2014;3:7-13. www.academia.edu/6707801/Floral_essential_oils_Importance_and_uses_for_mankind
3. Posadzki P, Alotaibi A, Ernst E. Adverse effects of aromatherapy: a systematic review of case reports and case series. Int J Risk Saf Med. 2012;24:147-161. doi: 10.3233/JRS-2012-0568
4. Sharmeen JB, Mahomoodally FM, Zengin G, et al. Essential oils as natural sources of fragrance compounds for cosmetics and cosmeceuticals. Molecules. 2021;26:666. doi: 10.3390/molecules26030666
5. Henley DV, Lipson N, Korach KS, et al. Prepubertal gynecomastia linked to lavender and tea tree oils. N Engl J Med. 2007;356:479-485. doi: 10.1056/NEJMoa064725
6. Nematollahi N, Weinberg JL, Flattery J, et al. Volatile chemical emissions from essential oils with therapeutic claims. Air Qual Atmosphere Health. 2021;14:365-369. doi: 10.1007/s11869-020-00941-4
7. Balekian D, Long A. Essential oil diffusers and asthma. Published February 24, 2020. Accessed September 22, 2023. www.aaaai.org/Allergist-Resources/Ask-the-Expert/Answers/Old-Ask-the-Experts/oil-diffusers-asthma
8. Aura Cacia. Quality. Accessed September 22, 2023. www.auracacia.com/quality
9. Now. Essential oil identity & purity testing. Accessed September 22, 2023. www.nowfoods.com/quality-safety/essential-oil-identity-purity-testing
10. Aura Cacia. GCMS documents. Accessed September 22, 2023. www.auracacia.com/aura-cacia-gcms-documents
11. Lopresti AL, Smith SJ, Drummond PD. Herbal treatments for migraine: a systematic review of randomised-controlled studies. Phytother Res. 2020;34:2493-2517. doi: 10.1002/ptr.6701
12. Niazi M, Hashempur MH, Taghizadeh M, et al. Efficacy of topical Rose (Rosa damascena Mill.) oil for migraine headache: A randomized double-blinded placebo-controlled cross-over trial. Complement Ther Med. 2017;34:35-41. doi: 10.1016/j.ctim. 2017.07.009
13. Rafieian-Kopaei M, Hasanpour-Dehkordi A, Lorigooini Z, et al. Comparing the effect of intranasal lidocaine 4% with peppermint essential oil drop 1.5% on migraine attacks: a double-blind clinical trial. Int J Prev Med. 2019;10:121. doi: 10.4103/ijpvm.IJPVM_530_17
14. Göbel H, Fresenius J, Heinze A, et al. [Effectiveness of Oleum menthae piperitae and paracetamol in therapy of headache of the tension type]. Nervenarzt. 1996;67:672-681. doi: 10.1007/s001150050040
15. Hunt R, Dienemann J, Norton HJ, et al. Aromatherapy as treatment for postoperative nausea: a randomized trial. Anesth Analg. 2013;117:597-604. doi: 10.1213/ANE.0b013e31824a0b1c
16. Maghami M, Afazel MR, Azizi-Fini I, et al. The effect of aromatherapy with peppermint essential oil on nausea and vomiting after cardiac surgery: a randomized clinical trial. Complement Ther Clin Pract. 2020;40:101199. doi: 10.1016/j.ctcp.2020.101199
17. Hills JM, Aaronson PI. The mechanism of action of peppermint oil on gastrointestinal smooth muscle. An analysis using patch clamp electrophysiology and isolated tissue pharmacology in rabbit and guinea pig. Gastroenterology. 1991;101:55-65. doi: 10.1016/0016-5085(91)90459-x
18. Alammar N, Wang L, Saberi B, et al. The impact of peppermint oil on the irritable bowel syndrome: a meta-analysis of the pooled clinical data. BMC Complement Altern Med. 2019;19:21. doi: 10.1186/s12906-018-2409-0
19. Cash BD, Epstein MS, Shah SM. A novel delivery system of peppermint oil is an effective therapy for irritable bowel syndrome symptoms. Dig Dis Sci. 2016;61:560-571. doi: 10.1007/s10620-015-3858-7
20. Weerts ZZRM, Masclee AAM, Witteman BJM, et al. Efficacy and safety of peppermint oil in a randomized, double-blind trial of patients with irritable bowel syndrome. Gastroenterology. 2020;158:123-136. doi: 10.1053/j.gastro.2019.08.026
21. Ma X, Yue ZQ, Gong ZQ, et al. The effect of diaphragmatic breathing on attention, negative affect and stress in healthy adults. Front Psychol. 2017;8:874. doi: 10.3389/fpsyg.2017.00874
22. Cabral P, Meyer HB, Ames D. Effectiveness of yoga therapy as a complementary treatment for major psychiatric disorders: a meta-analysis. Prim Care Companion CNS Disord. Published July 7, 2011. doi: 10.4088/PCC.10r01068
23. Donelli D, Antonelli M, Bellinazzi C, et ala. Effects of lavender on anxiety: systematic review and meta-analysis. Phytomedicine Int J Phytother Phytopharm. 2019;65:153099. doi: 10.1016/j.phymed.2019.153099
24. Koulivand PH, Khaleghi Ghadiri M, Gorji A. Lavender and the nervous system. Evid Based Complement Alternat Med. 2013;2013:1-10. doi: 10.1155/2013/681304
25. Kang HJ, Nam ES, Lee Y, et al. How strong is the evidence for the anxiolytic efficacy of lavender? Systematic review and meta-analysis of randomized controlled trials. Asian Nurs Res. 2019;13:295-305. doi: 10.1016/j.anr.2019.11.003
26. Barão Paixão VL, Freire de Carvalho J. Essential oil therapy in rheumatic diseases: a systematic review. Complement Ther Clin Pract. 2021;43:101391. doi: 10.1016/j.ctcp.2021.101391
27. Yasa Ozturk G, Bashan I. The effect of aromatherapy with lavender oil on the health-related quality of life in patients with fibromyalgia. J Food Qual. 2021;2021:1-5. doi: 10.1155/2021/9938630
28. Ko GD, Hum A, Traitses G, et al. Effects of topical O24 essential oils on patients with fibromyalgia syndrome: a randomized, placebo controlled pilot study. J Musculoskelet Pain. 2007;15:11-19. doi: 10.1300/J094v15n01_03
29. Lillehei AS, Halcon LL. A systematic review of the effect of inhaled essential oils on sleep. J Altern Complement Med. 2014;20:441-451. doi: 10.1089/acm.2013.0311
30. Cheong MJ, Kim S, Kim JS, et al. A systematic literature review and meta-analysis of the clinical effects of aroma inhalation therapy on sleep problems. Medicine (Baltimore). 2021;100:e24652. doi: 10.1097/MD.0000000000024652
31. Afrasiabian F, Mirabzadeh Ardakani M, Rahmani K, et al. Aloysia citriodora Paláu (lemon verbena) for insomnia patients: a randomized, double-blind, placebo-controlled clinical trial of efficacy and safety. Phytother Res PTR. 2019;33:350-359. doi: 10.1002/ptr.6228
32. Lee M, Lim S, Song JA, et al. The effects of aromatherapy essential oil inhalation on stress, sleep quality and immunity in healthy adults: randomized controlled trial. Eur J Integr Med. 2017;12:79-86. doi: 10.1016/j.eujim.2017.04.009
33. Nasiri Lari Z, Hajimonfarednejad M, Riasatian M, et al. Efficacy of inhaled Lavandula angustifolia Mill. Essential oil on sleep quality, quality of life and metabolic control in patients with diabetes mellitus type II and insomnia. J Ethnopharmacol. 2020;251:112560. doi: 10.1016/j.jep.2020.112560
34. McDonnell B, Newcomb P. Trial of essential oils to improve sleep for patients in cardiac rehabilitation. J Altern Complement Med N Y N. 2019;25:1193-1199. doi: 10.1089/acm.2019.0222
35. Song JA, Lee MK, Min E, et al. Effects of aromatherapy on dysmenorrhea: a systematic review and meta-analysis. Int J Nurs Stud. 2018;84:1-11. doi: 10.1016/j.ijnurstu.2018.01.016
36. Ou MC, Hsu TF, Lai AC, et al. Pain relief assessment by aromatic essential oil massage on outpatients with primary dysmenorrhea: a randomized, double-blind clinical trial: PD pain relief by aromatic oil massage. J Obstet Gynaecol Res. 2012;38:817-822. doi: 10.1111/j.1447-0756.2011.01802.x
37. Sut N, Kahyaoglu-Sut H. Effect of aromatherapy massage on pain in primary dysmenorrhea: a meta-analysis. Complement Ther Clin Pract. 2017;27:5-10. doi: 10.1016/j.ctcp.2017.01.001
38. Keyhanmehr AS, Kolouri S, Heydarirad G, et al. Aromatherapy for the management of cancer complications: a narrative review. Complement Ther Clin Pract. 2018;31:175-180. doi: 10.1016/j.ctcp.2018.02.009
39. Sriningsih I, Elisa E, Lestari KP. Aromatherapy ginger use in patients with nausea & vomiting on post cervical cancer chemotherapy. KEMAS J Kesehat Masy. 2017;13:59-68. doi: 10.15294/kemas.v13i1.5367
40. Lua PL, Salihah N, Mazlan N. Effects of inhaled ginger aromatherapy on chemotherapy-induced nausea and vomiting and health-related quality of life in women with breast cancer. Complement Ther Med. 2015;23:396-404. doi: 10.1016/j.ctim.2015.03.009
41. Arslan I, Aydinoglu S, Karan NB. Can lavender oil inhalation help to overcome dental anxiety and pain in children? A randomized clinical trial. Eur J Pediatr. 2020;179:985-992. doi: 10.1007/s00431-020-03595-7
42. Ghaderi F, Solhjou N. The effects of lavender aromatherapy on stress and pain perception in children during dental treatment: a randomized clinical trial. Complement Ther Clin Pract. 2020;40:101182. doi: 10.1016/j.ctcp.2020.101182
43. Jafarzadeh M, Arman S, Pour FF. Effect of aromatherapy with orange essential oil on salivary cortisol and pulse rate in children during dental treatment: a randomized controlled clinical trial. Adv Biomed Res. 2013;2:10. doi: 10.4103/2277-9175.107968
44. Lehrner J, Eckersberger C, Walla P, et al. Ambient odor of orange in a dental office reduces anxiety and improves mood in female patients. Physiol Behav. 2000;71:83-86. doi: 10.1016/S0031-9384(00)00308-5
45. Jimbo D, Kimura Y, Taniguchi M, et al. Effect of aromatherapy on patients with Alzheimer’s disease. Psychogeriatrics. 2009;9:173-179. doi: 10.1111/j.1479-8301.2009.00299.x
46. Ball EL, Owen-Booth B, Gray A, et al. Aromatherapy for dementia. Cochrane Database Syst Rev. 2020;(8). doi: 10.1002/14651858.CD003150.pub3
47. Uzunçakmak T, Ayaz Alkaya S. Effect of aromatherapy on coping with premenstrual syndrome: a randomized controlled trial. Complement Ther Med. 2018;36:63-67. doi: 10.1016/j.ctim.2017.11.022
48. Tanvisut R, Traisrisilp K, Tongsong T. Efficacy of aromatherapy for reducing pain during labor: a randomized controlled trial. Arch Gynecol Obstet. 2018;297:1145-1150. doi: 10.1007/s00404-018-4700-1
49. Ramsey JT, Shropshire BC, Nagy TR, et al. Essential oils and health. Yale J Biol Med. 2020;93:291-305.
50. Puškárová A, Bučková M, Kraková L, et al. The antibacterial and antifungal activity of six essential oils and their cyto/genotoxicity to human HEL 12469 cells. Sci Rep. 2017;7:8211. doi: 10.1038/s41598-017-08673-9
51. Deyno S, Mtewa AG, Abebe A, et al. Essential oils as topical anti-infective agents: a systematic review and meta-analysis. Complement Ther Med. 2019;47:102224. doi: 10.1016/j.ctim.2019.102224
52. Prall S, Bowles EJ, Bennett K, et al. Effects of essential oils on symptoms and course (duration and severity) of viral respiratory infections in humans: a rapid review. Adv Integr Med. 2020;7:218-221. doi: 10.1016/j.aimed.2020.07.005
53. Weeks JA, Guiney PD, Nikiforov AI. Assessment of the environmental fate and ecotoxicity of N,N-diethyl-m-toluamide (DEET). Integr Environ Assess Manag. 2012;8:120-134. doi: 10.1002/ieam.1246
54. Nerio LS, Olivero-Verbel J, Stashenko E. Repellent activity of essential oils: a review. Bioresour Technol. 2010;101:372-378. doi: 10.1016/j.biortech.2009.07.048
55. Lee MY. Essential oils as repellents against arthropods. BioMed Res Int. 2018;2018:6860271. doi: 10.1155/2018/6860271
56. Göbel H, Heinze A, Heinze-Kuhn K, et al. [Peppermint oil in the acute treatment of tension-type headache]. Schmerz Berl Ger. 2016;30:295-310. doi: 10.1007/s00482-016-0109-6
1. Butnariu M, Sarac I. Essential oils from plants. J Biotechnol Biomed Sci. 2018;1:35-43. doi: 10.14302/issn.2576-6694.jbbs-18-2489
2. Singh B, Sellam P, Majumder, J, et al. Floral essential oils : importance and uses for mankind. HortFlora Res Spectr. 2014;3:7-13. www.academia.edu/6707801/Floral_essential_oils_Importance_and_uses_for_mankind
3. Posadzki P, Alotaibi A, Ernst E. Adverse effects of aromatherapy: a systematic review of case reports and case series. Int J Risk Saf Med. 2012;24:147-161. doi: 10.3233/JRS-2012-0568
4. Sharmeen JB, Mahomoodally FM, Zengin G, et al. Essential oils as natural sources of fragrance compounds for cosmetics and cosmeceuticals. Molecules. 2021;26:666. doi: 10.3390/molecules26030666
5. Henley DV, Lipson N, Korach KS, et al. Prepubertal gynecomastia linked to lavender and tea tree oils. N Engl J Med. 2007;356:479-485. doi: 10.1056/NEJMoa064725
6. Nematollahi N, Weinberg JL, Flattery J, et al. Volatile chemical emissions from essential oils with therapeutic claims. Air Qual Atmosphere Health. 2021;14:365-369. doi: 10.1007/s11869-020-00941-4
7. Balekian D, Long A. Essential oil diffusers and asthma. Published February 24, 2020. Accessed September 22, 2023. www.aaaai.org/Allergist-Resources/Ask-the-Expert/Answers/Old-Ask-the-Experts/oil-diffusers-asthma
8. Aura Cacia. Quality. Accessed September 22, 2023. www.auracacia.com/quality
9. Now. Essential oil identity & purity testing. Accessed September 22, 2023. www.nowfoods.com/quality-safety/essential-oil-identity-purity-testing
10. Aura Cacia. GCMS documents. Accessed September 22, 2023. www.auracacia.com/aura-cacia-gcms-documents
11. Lopresti AL, Smith SJ, Drummond PD. Herbal treatments for migraine: a systematic review of randomised-controlled studies. Phytother Res. 2020;34:2493-2517. doi: 10.1002/ptr.6701
12. Niazi M, Hashempur MH, Taghizadeh M, et al. Efficacy of topical Rose (Rosa damascena Mill.) oil for migraine headache: A randomized double-blinded placebo-controlled cross-over trial. Complement Ther Med. 2017;34:35-41. doi: 10.1016/j.ctim. 2017.07.009
13. Rafieian-Kopaei M, Hasanpour-Dehkordi A, Lorigooini Z, et al. Comparing the effect of intranasal lidocaine 4% with peppermint essential oil drop 1.5% on migraine attacks: a double-blind clinical trial. Int J Prev Med. 2019;10:121. doi: 10.4103/ijpvm.IJPVM_530_17
14. Göbel H, Fresenius J, Heinze A, et al. [Effectiveness of Oleum menthae piperitae and paracetamol in therapy of headache of the tension type]. Nervenarzt. 1996;67:672-681. doi: 10.1007/s001150050040
15. Hunt R, Dienemann J, Norton HJ, et al. Aromatherapy as treatment for postoperative nausea: a randomized trial. Anesth Analg. 2013;117:597-604. doi: 10.1213/ANE.0b013e31824a0b1c
16. Maghami M, Afazel MR, Azizi-Fini I, et al. The effect of aromatherapy with peppermint essential oil on nausea and vomiting after cardiac surgery: a randomized clinical trial. Complement Ther Clin Pract. 2020;40:101199. doi: 10.1016/j.ctcp.2020.101199
17. Hills JM, Aaronson PI. The mechanism of action of peppermint oil on gastrointestinal smooth muscle. An analysis using patch clamp electrophysiology and isolated tissue pharmacology in rabbit and guinea pig. Gastroenterology. 1991;101:55-65. doi: 10.1016/0016-5085(91)90459-x
18. Alammar N, Wang L, Saberi B, et al. The impact of peppermint oil on the irritable bowel syndrome: a meta-analysis of the pooled clinical data. BMC Complement Altern Med. 2019;19:21. doi: 10.1186/s12906-018-2409-0
19. Cash BD, Epstein MS, Shah SM. A novel delivery system of peppermint oil is an effective therapy for irritable bowel syndrome symptoms. Dig Dis Sci. 2016;61:560-571. doi: 10.1007/s10620-015-3858-7
20. Weerts ZZRM, Masclee AAM, Witteman BJM, et al. Efficacy and safety of peppermint oil in a randomized, double-blind trial of patients with irritable bowel syndrome. Gastroenterology. 2020;158:123-136. doi: 10.1053/j.gastro.2019.08.026
21. Ma X, Yue ZQ, Gong ZQ, et al. The effect of diaphragmatic breathing on attention, negative affect and stress in healthy adults. Front Psychol. 2017;8:874. doi: 10.3389/fpsyg.2017.00874
22. Cabral P, Meyer HB, Ames D. Effectiveness of yoga therapy as a complementary treatment for major psychiatric disorders: a meta-analysis. Prim Care Companion CNS Disord. Published July 7, 2011. doi: 10.4088/PCC.10r01068
23. Donelli D, Antonelli M, Bellinazzi C, et ala. Effects of lavender on anxiety: systematic review and meta-analysis. Phytomedicine Int J Phytother Phytopharm. 2019;65:153099. doi: 10.1016/j.phymed.2019.153099
24. Koulivand PH, Khaleghi Ghadiri M, Gorji A. Lavender and the nervous system. Evid Based Complement Alternat Med. 2013;2013:1-10. doi: 10.1155/2013/681304
25. Kang HJ, Nam ES, Lee Y, et al. How strong is the evidence for the anxiolytic efficacy of lavender? Systematic review and meta-analysis of randomized controlled trials. Asian Nurs Res. 2019;13:295-305. doi: 10.1016/j.anr.2019.11.003
26. Barão Paixão VL, Freire de Carvalho J. Essential oil therapy in rheumatic diseases: a systematic review. Complement Ther Clin Pract. 2021;43:101391. doi: 10.1016/j.ctcp.2021.101391
27. Yasa Ozturk G, Bashan I. The effect of aromatherapy with lavender oil on the health-related quality of life in patients with fibromyalgia. J Food Qual. 2021;2021:1-5. doi: 10.1155/2021/9938630
28. Ko GD, Hum A, Traitses G, et al. Effects of topical O24 essential oils on patients with fibromyalgia syndrome: a randomized, placebo controlled pilot study. J Musculoskelet Pain. 2007;15:11-19. doi: 10.1300/J094v15n01_03
29. Lillehei AS, Halcon LL. A systematic review of the effect of inhaled essential oils on sleep. J Altern Complement Med. 2014;20:441-451. doi: 10.1089/acm.2013.0311
30. Cheong MJ, Kim S, Kim JS, et al. A systematic literature review and meta-analysis of the clinical effects of aroma inhalation therapy on sleep problems. Medicine (Baltimore). 2021;100:e24652. doi: 10.1097/MD.0000000000024652
31. Afrasiabian F, Mirabzadeh Ardakani M, Rahmani K, et al. Aloysia citriodora Paláu (lemon verbena) for insomnia patients: a randomized, double-blind, placebo-controlled clinical trial of efficacy and safety. Phytother Res PTR. 2019;33:350-359. doi: 10.1002/ptr.6228
32. Lee M, Lim S, Song JA, et al. The effects of aromatherapy essential oil inhalation on stress, sleep quality and immunity in healthy adults: randomized controlled trial. Eur J Integr Med. 2017;12:79-86. doi: 10.1016/j.eujim.2017.04.009
33. Nasiri Lari Z, Hajimonfarednejad M, Riasatian M, et al. Efficacy of inhaled Lavandula angustifolia Mill. Essential oil on sleep quality, quality of life and metabolic control in patients with diabetes mellitus type II and insomnia. J Ethnopharmacol. 2020;251:112560. doi: 10.1016/j.jep.2020.112560
34. McDonnell B, Newcomb P. Trial of essential oils to improve sleep for patients in cardiac rehabilitation. J Altern Complement Med N Y N. 2019;25:1193-1199. doi: 10.1089/acm.2019.0222
35. Song JA, Lee MK, Min E, et al. Effects of aromatherapy on dysmenorrhea: a systematic review and meta-analysis. Int J Nurs Stud. 2018;84:1-11. doi: 10.1016/j.ijnurstu.2018.01.016
36. Ou MC, Hsu TF, Lai AC, et al. Pain relief assessment by aromatic essential oil massage on outpatients with primary dysmenorrhea: a randomized, double-blind clinical trial: PD pain relief by aromatic oil massage. J Obstet Gynaecol Res. 2012;38:817-822. doi: 10.1111/j.1447-0756.2011.01802.x
37. Sut N, Kahyaoglu-Sut H. Effect of aromatherapy massage on pain in primary dysmenorrhea: a meta-analysis. Complement Ther Clin Pract. 2017;27:5-10. doi: 10.1016/j.ctcp.2017.01.001
38. Keyhanmehr AS, Kolouri S, Heydarirad G, et al. Aromatherapy for the management of cancer complications: a narrative review. Complement Ther Clin Pract. 2018;31:175-180. doi: 10.1016/j.ctcp.2018.02.009
39. Sriningsih I, Elisa E, Lestari KP. Aromatherapy ginger use in patients with nausea & vomiting on post cervical cancer chemotherapy. KEMAS J Kesehat Masy. 2017;13:59-68. doi: 10.15294/kemas.v13i1.5367
40. Lua PL, Salihah N, Mazlan N. Effects of inhaled ginger aromatherapy on chemotherapy-induced nausea and vomiting and health-related quality of life in women with breast cancer. Complement Ther Med. 2015;23:396-404. doi: 10.1016/j.ctim.2015.03.009
41. Arslan I, Aydinoglu S, Karan NB. Can lavender oil inhalation help to overcome dental anxiety and pain in children? A randomized clinical trial. Eur J Pediatr. 2020;179:985-992. doi: 10.1007/s00431-020-03595-7
42. Ghaderi F, Solhjou N. The effects of lavender aromatherapy on stress and pain perception in children during dental treatment: a randomized clinical trial. Complement Ther Clin Pract. 2020;40:101182. doi: 10.1016/j.ctcp.2020.101182
43. Jafarzadeh M, Arman S, Pour FF. Effect of aromatherapy with orange essential oil on salivary cortisol and pulse rate in children during dental treatment: a randomized controlled clinical trial. Adv Biomed Res. 2013;2:10. doi: 10.4103/2277-9175.107968
44. Lehrner J, Eckersberger C, Walla P, et al. Ambient odor of orange in a dental office reduces anxiety and improves mood in female patients. Physiol Behav. 2000;71:83-86. doi: 10.1016/S0031-9384(00)00308-5
45. Jimbo D, Kimura Y, Taniguchi M, et al. Effect of aromatherapy on patients with Alzheimer’s disease. Psychogeriatrics. 2009;9:173-179. doi: 10.1111/j.1479-8301.2009.00299.x
46. Ball EL, Owen-Booth B, Gray A, et al. Aromatherapy for dementia. Cochrane Database Syst Rev. 2020;(8). doi: 10.1002/14651858.CD003150.pub3
47. Uzunçakmak T, Ayaz Alkaya S. Effect of aromatherapy on coping with premenstrual syndrome: a randomized controlled trial. Complement Ther Med. 2018;36:63-67. doi: 10.1016/j.ctim.2017.11.022
48. Tanvisut R, Traisrisilp K, Tongsong T. Efficacy of aromatherapy for reducing pain during labor: a randomized controlled trial. Arch Gynecol Obstet. 2018;297:1145-1150. doi: 10.1007/s00404-018-4700-1
49. Ramsey JT, Shropshire BC, Nagy TR, et al. Essential oils and health. Yale J Biol Med. 2020;93:291-305.
50. Puškárová A, Bučková M, Kraková L, et al. The antibacterial and antifungal activity of six essential oils and their cyto/genotoxicity to human HEL 12469 cells. Sci Rep. 2017;7:8211. doi: 10.1038/s41598-017-08673-9
51. Deyno S, Mtewa AG, Abebe A, et al. Essential oils as topical anti-infective agents: a systematic review and meta-analysis. Complement Ther Med. 2019;47:102224. doi: 10.1016/j.ctim.2019.102224
52. Prall S, Bowles EJ, Bennett K, et al. Effects of essential oils on symptoms and course (duration and severity) of viral respiratory infections in humans: a rapid review. Adv Integr Med. 2020;7:218-221. doi: 10.1016/j.aimed.2020.07.005
53. Weeks JA, Guiney PD, Nikiforov AI. Assessment of the environmental fate and ecotoxicity of N,N-diethyl-m-toluamide (DEET). Integr Environ Assess Manag. 2012;8:120-134. doi: 10.1002/ieam.1246
54. Nerio LS, Olivero-Verbel J, Stashenko E. Repellent activity of essential oils: a review. Bioresour Technol. 2010;101:372-378. doi: 10.1016/j.biortech.2009.07.048
55. Lee MY. Essential oils as repellents against arthropods. BioMed Res Int. 2018;2018:6860271. doi: 10.1155/2018/6860271
56. Göbel H, Heinze A, Heinze-Kuhn K, et al. [Peppermint oil in the acute treatment of tension-type headache]. Schmerz Berl Ger. 2016;30:295-310. doi: 10.1007/s00482-016-0109-6
PRACTICE RECOMMENDATIONS
› Utilize lavender essential oil as an adjunctive treatment for fibromyalgia, dysmenorrhea, anxiety, and insomnia symptoms. B
› Recommend peppermint essential oil as an adjunctive treatment for irritable bowel syndrome, chemotherapy-induced nausea, and headache. B
Strength of recommendation (SOR)
A Good-quality patient-oriented evidence
B Inconsistent or limited-quality patient-oriented evidence
C Consensus, usual practice, opinion, disease-oriented evidence, case series
Pustular Eruption on the Face
The Diagnosis: Eczema Herpeticum
The patient’s condition with worsening facial edema and notable pain prompted a bedside Tzanck smear using a sample from the base of a deroofed forehead vesicle. In addition, a swab of a deroofed lesion was sent for herpes simplex virus and varicella-zoster virus (VZV) polymerase chain reaction (PCR) testing. The Tzanck smear demonstrated ballooning multinucleated syncytial giant cells and eosinophilic inclusion bodies (Figure), which are characteristic of certain herpesviruses including herpes simplex virus and VZV. He was started on intravenous acyclovir while PCR results were pending; the PCR test later confirmed positivity for herpes simplex virus type 1. Treatment was transitioned to oral valacyclovir once the lesions started crusting over. Notable healing and epithelialization of the lesions occurred during his hospital stay, and he was discharged home 5 days after starting treatment. He was counseled on autoinoculation, advised that he was considered infectious until all lesions had crusted over, and encouraged to employ frequent handwashing. Complete resolution of eczema herpeticum (EH) was noted at 3-week follow-up.
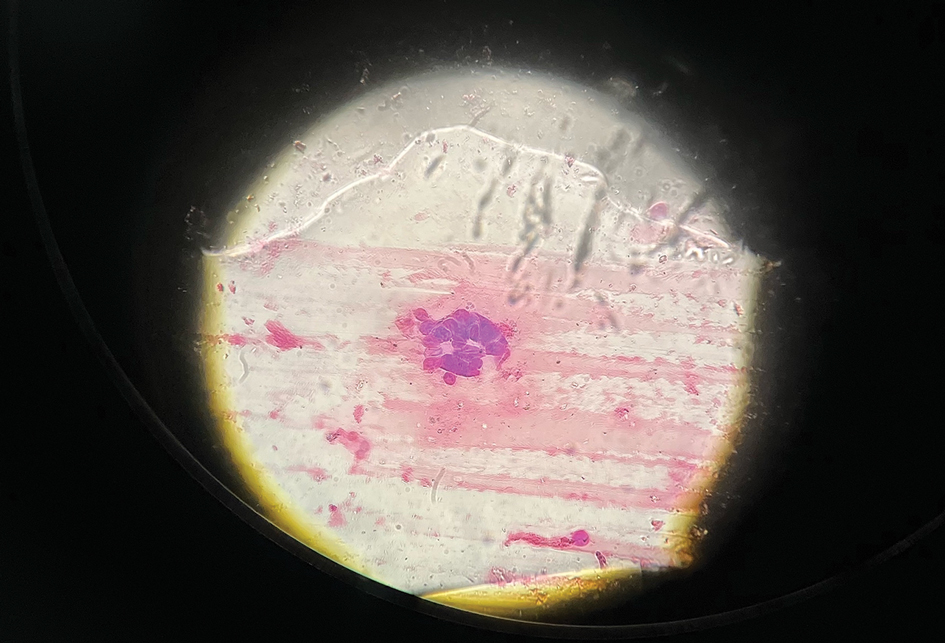
Eczema herpeticum (also known as Kaposi varicelliform eruption) is a potentially life-threatening disseminated cutaneous infection caused by herpes simplex virus types 1 and 2 in patients with pre-existing skin disease.1 It typically presents as a complication of atopic dermatitis (AD) but also has been identified as a rare complication in other conditions that disrupt the normal skin barrier, including mycosis fungoides, pemphigus foliaceus, pemphigus vulgaris, Darier disease, pityriasis rubra pilaris, contact dermatitis, and seborrheic dermatitis.1-4
The pathogenesis of EH is multifactorial. Disruption of the stratum corneum; impaired natural killer cell function; early-onset, untreated, or severe AD; disrupted skin microbiota with skewed colonization by Staphylococcus aureus; immunosuppressive AD therapies such as calcineurin inhibitors; eosinophilia; and helper T cell (TH2) cytokine predominance all have been suggested to play a role in the development of EH.5-8
As seen in our patient, EH presents with a sudden eruption of painful or pruritic, grouped, monomorphic, domeshaped vesicles with background swelling and erythema typically on the head, neck, and trunk. Vesicles then progress to punched-out erosions with overlying hemorrhagic crusting that can coalesce to form large denuded areas susceptible to superinfection with bacteria.9 Other accompanying symptoms include high fever, chills, malaise, and lymphadenopathy. Associated inflammation, classically described as erythema, may be difficult to discern in patients with darker skin and appears as hyperpigmentation; therefore, identification of clusters of monomorphic vesicles in areas of pre-existing dermatitis is particularly important for clinical diagnosis in people with darker skin types.
Various tests are available to confirm diagnosis in ambiguous cases. Bedside Tzanck smears can be performed rapidly and are considered positive if characteristic multinucleated giant cells are noted; however, they do not differentiate between the various herpesviruses. Direct fluorescent antibody testing of scraped lesions and viral cultures of swabbed vesicular fluid are equally effective in distinguishing between herpes simplex virus type 1, herpes simplex virus type 2, and VZV; PCR confirms the diagnosis with high specificity and sensitivity.10
In our patient, the initial differential diagnosis included EH, acute generalized exanthematous pustulosis, allergic contact dermatitis, and Orthopoxvirus infection. The positive Tzanck smear reduced the likelihood of a nonviral etiology. Additionally, worsening of the rash despite discontinuation of medications and utilization of topical steroids argued against acute generalized exanthematous pustulosis and allergic contact dermatitis. The laboratory findings reduced the likelihood of drug reaction with eosinophilia and systemic symptoms (DRESS) syndrome, and PCR findings ultimately ruled out Orthopoxvirus infections. Additional differential diagnoses for EH include dermatitis herpetiformis; primary VZV infection; hand, foot, and mouth disease; disseminated zoster infection; disseminated molluscum contagiosum; and eczema coxsackium.
Complications of EH include scarring; herpetic keratitis due to corneal infection, which if left untreated can progress to blindness; and rarely death due to multiorgan failure or septicemia.11 The traditional smallpox vaccine (ACAM2000) is contraindicated in patients with AD and EH, even when AD is in remission. These patients should avoid contact with recently vaccinated individuals.12 An alternative vaccine—Jynneos (Bavarian Nordic)—is available for these patients and their family members.13 Clinicians should be aware of this guideline, especially given the recent mpox (monkeypox) outbreaks.
Mild cases of EH are more common, may sometimes go unnoticed, and self-resolve in healthy patients. Severe cases may require systemic antiviral therapy. Acyclovir and its prodrug valacyclovir are standard treatments for EH. Alternatively, foscarnet or cidofovir can be used in the treatment of acyclovir-resistant thymidine kinase– deficient herpes simplex virus and other acyclovirresistant cases.14 Any secondary bacterial superinfections, usually due to staphylococcal or streptococcal bacteria, should be treated with antibiotics. A thorough ophthalmologic evaluation should be performed for patients with periocular involvement of EH. Empiric treatment should be started immediately, given a relative low toxicity of systemic antiviral therapy and high morbidity and mortality associated with untreated widespread EH.
It is important to maintain a high index of clinical suspicion for EH, especially in patients with pre-existing conditions such as AD who present with systemic symptoms and facial vesicles, pustules, or erosions to ensure prompt diagnosis and appropriate treatment.
- Baaniya B, Agrawal S. Kaposi varicelliform eruption in a patient with pemphigus vulgaris: a case report and review of the literature. Case Rep Dermatol Med. 2020;2020:6695342. doi:10.1155/2020/6695342
- Tayabali K, Pothiwalla H, Lowitt M. Eczema herpeticum in Darier’s disease: a topical storm. J Community Hosp Intern Med Perspect. 2019;9:347. doi:10.1080/20009666.2019.1650590
- Cavalié M, Giacchero D, Cardot-Leccia N, et al. Kaposi’s varicelliform eruption in a patient with pityriasis rubra pilaris (pityriasis rubra pilaris herpeticum). J Eur Acad Dermatol Venereol. 2013;27:1585-1586. doi:10.1111/JDV.12120
- Lee GH, Kim YM, Lee SY, et al. A case of eczema herpeticum with Hailey-Hailey disease. Ann Dermatol. 2009;21:311-314. doi:10.5021/ad.2009.21.3.311
- Seegräber M, Worm M, Werfel T, et al. Recurrent eczema herpeticum— a retrospective European multicenter study evaluating the clinical characteristics of eczema herpeticum cases in atopic dermatitis patients. J Eur Acad Dermatol Venereol. 2020;34:1074-1079. doi:10.1111/JDV.16090
- Kawakami Y, Ando T, Lee J-R, et al. Defective natural killer cell activity in a mouse model of eczema herpeticum. J Allergy Clin Immunol. 2017;139:997-1006.e10. doi:10.1016/j.jaci.2016.06.034
- Beck L, Latchney L, Zaccaro D, et al. Biomarkers of disease severity and Th2 polarity are predictors of risk for eczema herpeticum. J Allergy Clin Immunol. 2008;121:S37-S37. doi:10.1016/j.jaci.2007.12.152
- Kim M, Jung M, Hong SP, et al. Topical calcineurin inhibitors compromise stratum corneum integrity, epidermal permeability and antimicrobial barrier function. Exp Dermatol. 2010; 19:501-510. doi:10.1111/J.1600-0625.2009.00941.X
- Karray M, Kwan E, Souissi A. Kaposi varicelliform eruption. StatPearls [Internet]. StatPearls Publishing; 2023. https://www.ncbi.nlm.nih.gov/books/NBK482432/
- Dominguez SR, Pretty K, Hengartner R, et al. Comparison of herpes simplex virus PCR with culture for virus detection in multisource surface swab specimens from neonates [published online September 25, 2018]. J Clin Microbiol. doi:10.1128/JCM.00632-18
- Feye F, De Halleux C, Gillet JB, et al. Exacerbation of atopic dermatitis in the emergency department. Eur J Emerg Med. 2004;11:49-52. doi:10.1097/00063110-200412000-00014
- Casey C, Vellozzi C, Mootrey GT, et al; Vaccinia Case Definition Development Working Group; Advisory Committee on Immunization Practices-Armed Forces Epidemiological Board Smallpox Vaccine Safety Working Group. Surveillance guidelines for smallpox vaccine (vaccinia) adverse reactions. MMWR Recomm Rep. 2006;55:1-16.
- Rao AK, Petersen BW, Whitehill F, et al. Use of JYNNEOS (Smallpox and Monkeypox Vaccine, Live, Nonreplicating) for preexposure vaccination of persons at risk for occupational exposure to orthopoxviruses: recommendations of the Advisory Committee on Immunization Practices—United States, 2022. MMWR Morb Mortal Wkly Rep. 2022;71:734-742. doi:10.15585 /MMWR.MM7122E1
- Piret J, Boivin G. Resistance of herpes simplex viruses to nucleoside analogues: mechanisms, prevalence, and management. Antimicrob Agents Chemother. 2011;55:459. doi:10.1128/AAC.00615-10
The Diagnosis: Eczema Herpeticum
The patient’s condition with worsening facial edema and notable pain prompted a bedside Tzanck smear using a sample from the base of a deroofed forehead vesicle. In addition, a swab of a deroofed lesion was sent for herpes simplex virus and varicella-zoster virus (VZV) polymerase chain reaction (PCR) testing. The Tzanck smear demonstrated ballooning multinucleated syncytial giant cells and eosinophilic inclusion bodies (Figure), which are characteristic of certain herpesviruses including herpes simplex virus and VZV. He was started on intravenous acyclovir while PCR results were pending; the PCR test later confirmed positivity for herpes simplex virus type 1. Treatment was transitioned to oral valacyclovir once the lesions started crusting over. Notable healing and epithelialization of the lesions occurred during his hospital stay, and he was discharged home 5 days after starting treatment. He was counseled on autoinoculation, advised that he was considered infectious until all lesions had crusted over, and encouraged to employ frequent handwashing. Complete resolution of eczema herpeticum (EH) was noted at 3-week follow-up.

Eczema herpeticum (also known as Kaposi varicelliform eruption) is a potentially life-threatening disseminated cutaneous infection caused by herpes simplex virus types 1 and 2 in patients with pre-existing skin disease.1 It typically presents as a complication of atopic dermatitis (AD) but also has been identified as a rare complication in other conditions that disrupt the normal skin barrier, including mycosis fungoides, pemphigus foliaceus, pemphigus vulgaris, Darier disease, pityriasis rubra pilaris, contact dermatitis, and seborrheic dermatitis.1-4
The pathogenesis of EH is multifactorial. Disruption of the stratum corneum; impaired natural killer cell function; early-onset, untreated, or severe AD; disrupted skin microbiota with skewed colonization by Staphylococcus aureus; immunosuppressive AD therapies such as calcineurin inhibitors; eosinophilia; and helper T cell (TH2) cytokine predominance all have been suggested to play a role in the development of EH.5-8
As seen in our patient, EH presents with a sudden eruption of painful or pruritic, grouped, monomorphic, domeshaped vesicles with background swelling and erythema typically on the head, neck, and trunk. Vesicles then progress to punched-out erosions with overlying hemorrhagic crusting that can coalesce to form large denuded areas susceptible to superinfection with bacteria.9 Other accompanying symptoms include high fever, chills, malaise, and lymphadenopathy. Associated inflammation, classically described as erythema, may be difficult to discern in patients with darker skin and appears as hyperpigmentation; therefore, identification of clusters of monomorphic vesicles in areas of pre-existing dermatitis is particularly important for clinical diagnosis in people with darker skin types.
Various tests are available to confirm diagnosis in ambiguous cases. Bedside Tzanck smears can be performed rapidly and are considered positive if characteristic multinucleated giant cells are noted; however, they do not differentiate between the various herpesviruses. Direct fluorescent antibody testing of scraped lesions and viral cultures of swabbed vesicular fluid are equally effective in distinguishing between herpes simplex virus type 1, herpes simplex virus type 2, and VZV; PCR confirms the diagnosis with high specificity and sensitivity.10
In our patient, the initial differential diagnosis included EH, acute generalized exanthematous pustulosis, allergic contact dermatitis, and Orthopoxvirus infection. The positive Tzanck smear reduced the likelihood of a nonviral etiology. Additionally, worsening of the rash despite discontinuation of medications and utilization of topical steroids argued against acute generalized exanthematous pustulosis and allergic contact dermatitis. The laboratory findings reduced the likelihood of drug reaction with eosinophilia and systemic symptoms (DRESS) syndrome, and PCR findings ultimately ruled out Orthopoxvirus infections. Additional differential diagnoses for EH include dermatitis herpetiformis; primary VZV infection; hand, foot, and mouth disease; disseminated zoster infection; disseminated molluscum contagiosum; and eczema coxsackium.
Complications of EH include scarring; herpetic keratitis due to corneal infection, which if left untreated can progress to blindness; and rarely death due to multiorgan failure or septicemia.11 The traditional smallpox vaccine (ACAM2000) is contraindicated in patients with AD and EH, even when AD is in remission. These patients should avoid contact with recently vaccinated individuals.12 An alternative vaccine—Jynneos (Bavarian Nordic)—is available for these patients and their family members.13 Clinicians should be aware of this guideline, especially given the recent mpox (monkeypox) outbreaks.
Mild cases of EH are more common, may sometimes go unnoticed, and self-resolve in healthy patients. Severe cases may require systemic antiviral therapy. Acyclovir and its prodrug valacyclovir are standard treatments for EH. Alternatively, foscarnet or cidofovir can be used in the treatment of acyclovir-resistant thymidine kinase– deficient herpes simplex virus and other acyclovirresistant cases.14 Any secondary bacterial superinfections, usually due to staphylococcal or streptococcal bacteria, should be treated with antibiotics. A thorough ophthalmologic evaluation should be performed for patients with periocular involvement of EH. Empiric treatment should be started immediately, given a relative low toxicity of systemic antiviral therapy and high morbidity and mortality associated with untreated widespread EH.
It is important to maintain a high index of clinical suspicion for EH, especially in patients with pre-existing conditions such as AD who present with systemic symptoms and facial vesicles, pustules, or erosions to ensure prompt diagnosis and appropriate treatment.
The Diagnosis: Eczema Herpeticum
The patient’s condition with worsening facial edema and notable pain prompted a bedside Tzanck smear using a sample from the base of a deroofed forehead vesicle. In addition, a swab of a deroofed lesion was sent for herpes simplex virus and varicella-zoster virus (VZV) polymerase chain reaction (PCR) testing. The Tzanck smear demonstrated ballooning multinucleated syncytial giant cells and eosinophilic inclusion bodies (Figure), which are characteristic of certain herpesviruses including herpes simplex virus and VZV. He was started on intravenous acyclovir while PCR results were pending; the PCR test later confirmed positivity for herpes simplex virus type 1. Treatment was transitioned to oral valacyclovir once the lesions started crusting over. Notable healing and epithelialization of the lesions occurred during his hospital stay, and he was discharged home 5 days after starting treatment. He was counseled on autoinoculation, advised that he was considered infectious until all lesions had crusted over, and encouraged to employ frequent handwashing. Complete resolution of eczema herpeticum (EH) was noted at 3-week follow-up.

Eczema herpeticum (also known as Kaposi varicelliform eruption) is a potentially life-threatening disseminated cutaneous infection caused by herpes simplex virus types 1 and 2 in patients with pre-existing skin disease.1 It typically presents as a complication of atopic dermatitis (AD) but also has been identified as a rare complication in other conditions that disrupt the normal skin barrier, including mycosis fungoides, pemphigus foliaceus, pemphigus vulgaris, Darier disease, pityriasis rubra pilaris, contact dermatitis, and seborrheic dermatitis.1-4
The pathogenesis of EH is multifactorial. Disruption of the stratum corneum; impaired natural killer cell function; early-onset, untreated, or severe AD; disrupted skin microbiota with skewed colonization by Staphylococcus aureus; immunosuppressive AD therapies such as calcineurin inhibitors; eosinophilia; and helper T cell (TH2) cytokine predominance all have been suggested to play a role in the development of EH.5-8
As seen in our patient, EH presents with a sudden eruption of painful or pruritic, grouped, monomorphic, domeshaped vesicles with background swelling and erythema typically on the head, neck, and trunk. Vesicles then progress to punched-out erosions with overlying hemorrhagic crusting that can coalesce to form large denuded areas susceptible to superinfection with bacteria.9 Other accompanying symptoms include high fever, chills, malaise, and lymphadenopathy. Associated inflammation, classically described as erythema, may be difficult to discern in patients with darker skin and appears as hyperpigmentation; therefore, identification of clusters of monomorphic vesicles in areas of pre-existing dermatitis is particularly important for clinical diagnosis in people with darker skin types.
Various tests are available to confirm diagnosis in ambiguous cases. Bedside Tzanck smears can be performed rapidly and are considered positive if characteristic multinucleated giant cells are noted; however, they do not differentiate between the various herpesviruses. Direct fluorescent antibody testing of scraped lesions and viral cultures of swabbed vesicular fluid are equally effective in distinguishing between herpes simplex virus type 1, herpes simplex virus type 2, and VZV; PCR confirms the diagnosis with high specificity and sensitivity.10
In our patient, the initial differential diagnosis included EH, acute generalized exanthematous pustulosis, allergic contact dermatitis, and Orthopoxvirus infection. The positive Tzanck smear reduced the likelihood of a nonviral etiology. Additionally, worsening of the rash despite discontinuation of medications and utilization of topical steroids argued against acute generalized exanthematous pustulosis and allergic contact dermatitis. The laboratory findings reduced the likelihood of drug reaction with eosinophilia and systemic symptoms (DRESS) syndrome, and PCR findings ultimately ruled out Orthopoxvirus infections. Additional differential diagnoses for EH include dermatitis herpetiformis; primary VZV infection; hand, foot, and mouth disease; disseminated zoster infection; disseminated molluscum contagiosum; and eczema coxsackium.
Complications of EH include scarring; herpetic keratitis due to corneal infection, which if left untreated can progress to blindness; and rarely death due to multiorgan failure or septicemia.11 The traditional smallpox vaccine (ACAM2000) is contraindicated in patients with AD and EH, even when AD is in remission. These patients should avoid contact with recently vaccinated individuals.12 An alternative vaccine—Jynneos (Bavarian Nordic)—is available for these patients and their family members.13 Clinicians should be aware of this guideline, especially given the recent mpox (monkeypox) outbreaks.
Mild cases of EH are more common, may sometimes go unnoticed, and self-resolve in healthy patients. Severe cases may require systemic antiviral therapy. Acyclovir and its prodrug valacyclovir are standard treatments for EH. Alternatively, foscarnet or cidofovir can be used in the treatment of acyclovir-resistant thymidine kinase– deficient herpes simplex virus and other acyclovirresistant cases.14 Any secondary bacterial superinfections, usually due to staphylococcal or streptococcal bacteria, should be treated with antibiotics. A thorough ophthalmologic evaluation should be performed for patients with periocular involvement of EH. Empiric treatment should be started immediately, given a relative low toxicity of systemic antiviral therapy and high morbidity and mortality associated with untreated widespread EH.
It is important to maintain a high index of clinical suspicion for EH, especially in patients with pre-existing conditions such as AD who present with systemic symptoms and facial vesicles, pustules, or erosions to ensure prompt diagnosis and appropriate treatment.
- Baaniya B, Agrawal S. Kaposi varicelliform eruption in a patient with pemphigus vulgaris: a case report and review of the literature. Case Rep Dermatol Med. 2020;2020:6695342. doi:10.1155/2020/6695342
- Tayabali K, Pothiwalla H, Lowitt M. Eczema herpeticum in Darier’s disease: a topical storm. J Community Hosp Intern Med Perspect. 2019;9:347. doi:10.1080/20009666.2019.1650590
- Cavalié M, Giacchero D, Cardot-Leccia N, et al. Kaposi’s varicelliform eruption in a patient with pityriasis rubra pilaris (pityriasis rubra pilaris herpeticum). J Eur Acad Dermatol Venereol. 2013;27:1585-1586. doi:10.1111/JDV.12120
- Lee GH, Kim YM, Lee SY, et al. A case of eczema herpeticum with Hailey-Hailey disease. Ann Dermatol. 2009;21:311-314. doi:10.5021/ad.2009.21.3.311
- Seegräber M, Worm M, Werfel T, et al. Recurrent eczema herpeticum— a retrospective European multicenter study evaluating the clinical characteristics of eczema herpeticum cases in atopic dermatitis patients. J Eur Acad Dermatol Venereol. 2020;34:1074-1079. doi:10.1111/JDV.16090
- Kawakami Y, Ando T, Lee J-R, et al. Defective natural killer cell activity in a mouse model of eczema herpeticum. J Allergy Clin Immunol. 2017;139:997-1006.e10. doi:10.1016/j.jaci.2016.06.034
- Beck L, Latchney L, Zaccaro D, et al. Biomarkers of disease severity and Th2 polarity are predictors of risk for eczema herpeticum. J Allergy Clin Immunol. 2008;121:S37-S37. doi:10.1016/j.jaci.2007.12.152
- Kim M, Jung M, Hong SP, et al. Topical calcineurin inhibitors compromise stratum corneum integrity, epidermal permeability and antimicrobial barrier function. Exp Dermatol. 2010; 19:501-510. doi:10.1111/J.1600-0625.2009.00941.X
- Karray M, Kwan E, Souissi A. Kaposi varicelliform eruption. StatPearls [Internet]. StatPearls Publishing; 2023. https://www.ncbi.nlm.nih.gov/books/NBK482432/
- Dominguez SR, Pretty K, Hengartner R, et al. Comparison of herpes simplex virus PCR with culture for virus detection in multisource surface swab specimens from neonates [published online September 25, 2018]. J Clin Microbiol. doi:10.1128/JCM.00632-18
- Feye F, De Halleux C, Gillet JB, et al. Exacerbation of atopic dermatitis in the emergency department. Eur J Emerg Med. 2004;11:49-52. doi:10.1097/00063110-200412000-00014
- Casey C, Vellozzi C, Mootrey GT, et al; Vaccinia Case Definition Development Working Group; Advisory Committee on Immunization Practices-Armed Forces Epidemiological Board Smallpox Vaccine Safety Working Group. Surveillance guidelines for smallpox vaccine (vaccinia) adverse reactions. MMWR Recomm Rep. 2006;55:1-16.
- Rao AK, Petersen BW, Whitehill F, et al. Use of JYNNEOS (Smallpox and Monkeypox Vaccine, Live, Nonreplicating) for preexposure vaccination of persons at risk for occupational exposure to orthopoxviruses: recommendations of the Advisory Committee on Immunization Practices—United States, 2022. MMWR Morb Mortal Wkly Rep. 2022;71:734-742. doi:10.15585 /MMWR.MM7122E1
- Piret J, Boivin G. Resistance of herpes simplex viruses to nucleoside analogues: mechanisms, prevalence, and management. Antimicrob Agents Chemother. 2011;55:459. doi:10.1128/AAC.00615-10
- Baaniya B, Agrawal S. Kaposi varicelliform eruption in a patient with pemphigus vulgaris: a case report and review of the literature. Case Rep Dermatol Med. 2020;2020:6695342. doi:10.1155/2020/6695342
- Tayabali K, Pothiwalla H, Lowitt M. Eczema herpeticum in Darier’s disease: a topical storm. J Community Hosp Intern Med Perspect. 2019;9:347. doi:10.1080/20009666.2019.1650590
- Cavalié M, Giacchero D, Cardot-Leccia N, et al. Kaposi’s varicelliform eruption in a patient with pityriasis rubra pilaris (pityriasis rubra pilaris herpeticum). J Eur Acad Dermatol Venereol. 2013;27:1585-1586. doi:10.1111/JDV.12120
- Lee GH, Kim YM, Lee SY, et al. A case of eczema herpeticum with Hailey-Hailey disease. Ann Dermatol. 2009;21:311-314. doi:10.5021/ad.2009.21.3.311
- Seegräber M, Worm M, Werfel T, et al. Recurrent eczema herpeticum— a retrospective European multicenter study evaluating the clinical characteristics of eczema herpeticum cases in atopic dermatitis patients. J Eur Acad Dermatol Venereol. 2020;34:1074-1079. doi:10.1111/JDV.16090
- Kawakami Y, Ando T, Lee J-R, et al. Defective natural killer cell activity in a mouse model of eczema herpeticum. J Allergy Clin Immunol. 2017;139:997-1006.e10. doi:10.1016/j.jaci.2016.06.034
- Beck L, Latchney L, Zaccaro D, et al. Biomarkers of disease severity and Th2 polarity are predictors of risk for eczema herpeticum. J Allergy Clin Immunol. 2008;121:S37-S37. doi:10.1016/j.jaci.2007.12.152
- Kim M, Jung M, Hong SP, et al. Topical calcineurin inhibitors compromise stratum corneum integrity, epidermal permeability and antimicrobial barrier function. Exp Dermatol. 2010; 19:501-510. doi:10.1111/J.1600-0625.2009.00941.X
- Karray M, Kwan E, Souissi A. Kaposi varicelliform eruption. StatPearls [Internet]. StatPearls Publishing; 2023. https://www.ncbi.nlm.nih.gov/books/NBK482432/
- Dominguez SR, Pretty K, Hengartner R, et al. Comparison of herpes simplex virus PCR with culture for virus detection in multisource surface swab specimens from neonates [published online September 25, 2018]. J Clin Microbiol. doi:10.1128/JCM.00632-18
- Feye F, De Halleux C, Gillet JB, et al. Exacerbation of atopic dermatitis in the emergency department. Eur J Emerg Med. 2004;11:49-52. doi:10.1097/00063110-200412000-00014
- Casey C, Vellozzi C, Mootrey GT, et al; Vaccinia Case Definition Development Working Group; Advisory Committee on Immunization Practices-Armed Forces Epidemiological Board Smallpox Vaccine Safety Working Group. Surveillance guidelines for smallpox vaccine (vaccinia) adverse reactions. MMWR Recomm Rep. 2006;55:1-16.
- Rao AK, Petersen BW, Whitehill F, et al. Use of JYNNEOS (Smallpox and Monkeypox Vaccine, Live, Nonreplicating) for preexposure vaccination of persons at risk for occupational exposure to orthopoxviruses: recommendations of the Advisory Committee on Immunization Practices—United States, 2022. MMWR Morb Mortal Wkly Rep. 2022;71:734-742. doi:10.15585 /MMWR.MM7122E1
- Piret J, Boivin G. Resistance of herpes simplex viruses to nucleoside analogues: mechanisms, prevalence, and management. Antimicrob Agents Chemother. 2011;55:459. doi:10.1128/AAC.00615-10
A 52-year-old man developed a sudden eruption of small pustules on background erythema and edema covering the forehead, nasal bridge, periorbital region, cheeks, and perioral region on day 3 of hospitalization in the intensive care unit for management of septic shock secondary to a complicated urinary tract infection. He had a medical history of benign prostatic hyperplasia, sarcoidosis, and atopic dermatitis. He initially presented to the emergency department with fever, chills, and dysuria of 2 days’ duration. Because he received ceftriaxone, vancomycin, ciprofloxacin, and tamsulosin while hospitalized for the infection, the primary medical team suspected a drug reaction and empirically started applying hydrocortisone cream 2.5%. The rash continued to spread over the ensuing day, prompting a dermatology consultation to rule out a drug eruption and to help guide further management. The patient was in substantial distress and pain. Physical examination revealed numerous discrete and confluent monomorphic pustules on background erythema with faint collarettes of scale covering most of the face. Substantial periorbital and facial edema forced the eyes closed. There was no mucous membrane involvement. A review of systems was negative for dyspnea and dysphagia, and the rash was not present elsewhere on the body. Ophthalmologic evaluation revealed no ocular involvement or vision changes. Laboratory studies demonstrated neutrophilia (17.27×109 cells/L [reference range, 2.0–6.9×109 cells/L]). The eosinophil count, blood urea nitrogen/creatinine, and liver function tests were within reference range.
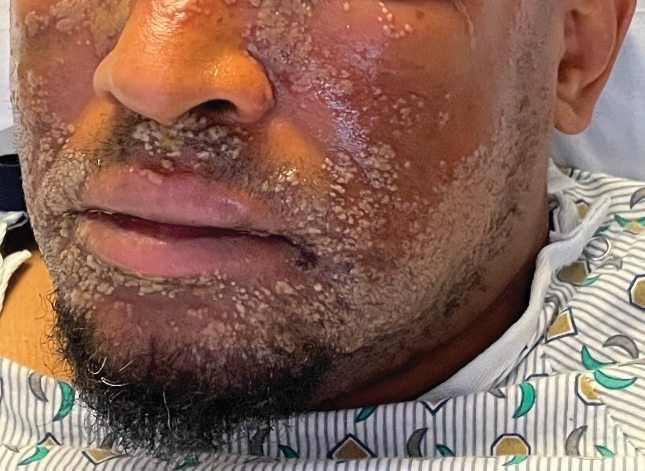
Papules on lip
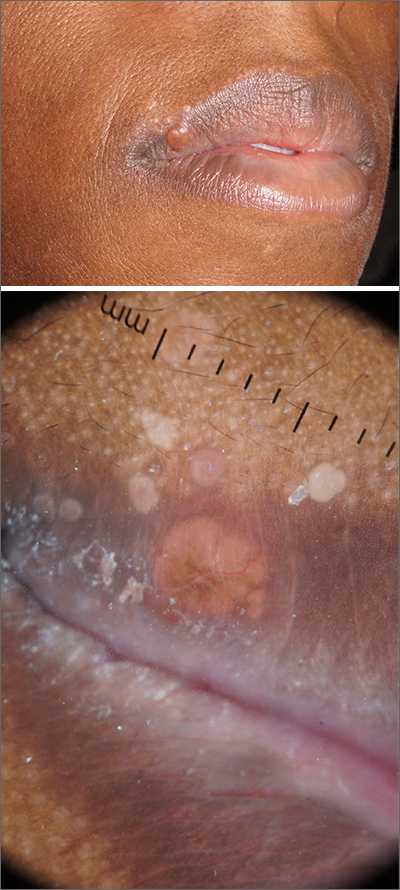
Pathology showed noncaseating granulomas consistent with cutaneous sarcoidosis. Based on these biopsy findings, a chest x-ray was ordered, and it confirmed a pulmonary sarcoid. A multidisciplinary work-up (including cardiac evaluation, continued rheumatologic care, and evaluation by Hematology) addressed this new finding.
Sarcoidosis is a multisystem inflammatory disorder characterized by the development of granulomas that can arise in any organ, but frequently involve the skin and lungs. Patients with cutaneous disease develop smooth skin lesions, including flesh-colored to pink or brown papules on the face. Genetic and environmental factors are both thought to contribute to the disease.
Race is a significant factor in the development of disease. Hispanic and Asian patients are significantly less likely to develop the disease compared to White or Black patients. In the Black Women’s Health Study, incidence in Black women was 71 per 100,000.1 Women are more likely to be affected than men.1
Many patients with sarcoidosis have a mild course, but for others the disease may progress on the skin or include pulmonary, renal, neurologic, or cardiac disease. Sometimes sarcoidosis is fatal. Recurrence can occur at any point later in life. Race influences disease severity as well as incidence, with hospitalization being 9 times as likely in Black patients compared with White patients.2 One recent study puts sarcoidosis mortality rates for Black women at 10 per million compared with 3 per million in Black men, and 1 per million in White women or men.3
Patients with disease limited to the skin may be treated with topical steroids such as clobetasol 0.05% cream or ointment or intralesional triamcinolone 10 mg/mL injected into affected lesions every 2 to 4 weeks. With pulmonary or other systemic disease, treatment may include various disease-modifying agents including prednisone, methotrexate, hydroxychloroquine, and TNF-alpha inhibitors. Because of the long-term adverse effects of systemic steroids, these agents are reserved for instances when pulmonary function is significantly impacted.
This patient had a reassuring cardiac and hematology work-up. Her pulmonary function was impacted sufficiently enough that her pulmonologist added a course of prednisone 10 mg daily tapered over 6 weeks. She had been on hydroxychloroquine 200 mg twice daily prior to the diagnosis of sarcoidosis for presumed mixed connective tissue disease and was continued on it for sarcoidosis after completing the prednisone taper. With these treatments, her facial lesions cleared and her breathing symptoms and fatigue improved. She remains under surveillance with a multidisciplinary team.
Photos and text for Photo Rounds Friday courtesy of Jonathan Karnes, MD (copyright retained). Dr. Karnes is the medical director of MDFMR Dermatology Services, Augusta, ME.
1. Cozier Y, Berman J, Palmer J, et al. Sarcoidosis in black women in the United States: data from the Black Women's Health Study. Chest. 2011;139:144-150. doi: 10.1378/chest.10-0413
2. Foreman MG, Mannino DM, Kamugisha L, et al. Hospitalization for patients with sarcoidosis: 1979-2000. Sarcoidosis Vasc Diffuse Lung Dis. 2006;23:124-129.
3. Mirsaeidi M, Machado R, Schraufnagel D, et al. Racial difference in sarcoidosis mortality in the United States. Chest. 2015; 147: 438-449. doi: 10.1378/chest.14-1120

Pathology showed noncaseating granulomas consistent with cutaneous sarcoidosis. Based on these biopsy findings, a chest x-ray was ordered, and it confirmed a pulmonary sarcoid. A multidisciplinary work-up (including cardiac evaluation, continued rheumatologic care, and evaluation by Hematology) addressed this new finding.
Sarcoidosis is a multisystem inflammatory disorder characterized by the development of granulomas that can arise in any organ, but frequently involve the skin and lungs. Patients with cutaneous disease develop smooth skin lesions, including flesh-colored to pink or brown papules on the face. Genetic and environmental factors are both thought to contribute to the disease.
Race is a significant factor in the development of disease. Hispanic and Asian patients are significantly less likely to develop the disease compared to White or Black patients. In the Black Women’s Health Study, incidence in Black women was 71 per 100,000.1 Women are more likely to be affected than men.1
Many patients with sarcoidosis have a mild course, but for others the disease may progress on the skin or include pulmonary, renal, neurologic, or cardiac disease. Sometimes sarcoidosis is fatal. Recurrence can occur at any point later in life. Race influences disease severity as well as incidence, with hospitalization being 9 times as likely in Black patients compared with White patients.2 One recent study puts sarcoidosis mortality rates for Black women at 10 per million compared with 3 per million in Black men, and 1 per million in White women or men.3
Patients with disease limited to the skin may be treated with topical steroids such as clobetasol 0.05% cream or ointment or intralesional triamcinolone 10 mg/mL injected into affected lesions every 2 to 4 weeks. With pulmonary or other systemic disease, treatment may include various disease-modifying agents including prednisone, methotrexate, hydroxychloroquine, and TNF-alpha inhibitors. Because of the long-term adverse effects of systemic steroids, these agents are reserved for instances when pulmonary function is significantly impacted.
This patient had a reassuring cardiac and hematology work-up. Her pulmonary function was impacted sufficiently enough that her pulmonologist added a course of prednisone 10 mg daily tapered over 6 weeks. She had been on hydroxychloroquine 200 mg twice daily prior to the diagnosis of sarcoidosis for presumed mixed connective tissue disease and was continued on it for sarcoidosis after completing the prednisone taper. With these treatments, her facial lesions cleared and her breathing symptoms and fatigue improved. She remains under surveillance with a multidisciplinary team.
Photos and text for Photo Rounds Friday courtesy of Jonathan Karnes, MD (copyright retained). Dr. Karnes is the medical director of MDFMR Dermatology Services, Augusta, ME.

Pathology showed noncaseating granulomas consistent with cutaneous sarcoidosis. Based on these biopsy findings, a chest x-ray was ordered, and it confirmed a pulmonary sarcoid. A multidisciplinary work-up (including cardiac evaluation, continued rheumatologic care, and evaluation by Hematology) addressed this new finding.
Sarcoidosis is a multisystem inflammatory disorder characterized by the development of granulomas that can arise in any organ, but frequently involve the skin and lungs. Patients with cutaneous disease develop smooth skin lesions, including flesh-colored to pink or brown papules on the face. Genetic and environmental factors are both thought to contribute to the disease.
Race is a significant factor in the development of disease. Hispanic and Asian patients are significantly less likely to develop the disease compared to White or Black patients. In the Black Women’s Health Study, incidence in Black women was 71 per 100,000.1 Women are more likely to be affected than men.1
Many patients with sarcoidosis have a mild course, but for others the disease may progress on the skin or include pulmonary, renal, neurologic, or cardiac disease. Sometimes sarcoidosis is fatal. Recurrence can occur at any point later in life. Race influences disease severity as well as incidence, with hospitalization being 9 times as likely in Black patients compared with White patients.2 One recent study puts sarcoidosis mortality rates for Black women at 10 per million compared with 3 per million in Black men, and 1 per million in White women or men.3
Patients with disease limited to the skin may be treated with topical steroids such as clobetasol 0.05% cream or ointment or intralesional triamcinolone 10 mg/mL injected into affected lesions every 2 to 4 weeks. With pulmonary or other systemic disease, treatment may include various disease-modifying agents including prednisone, methotrexate, hydroxychloroquine, and TNF-alpha inhibitors. Because of the long-term adverse effects of systemic steroids, these agents are reserved for instances when pulmonary function is significantly impacted.
This patient had a reassuring cardiac and hematology work-up. Her pulmonary function was impacted sufficiently enough that her pulmonologist added a course of prednisone 10 mg daily tapered over 6 weeks. She had been on hydroxychloroquine 200 mg twice daily prior to the diagnosis of sarcoidosis for presumed mixed connective tissue disease and was continued on it for sarcoidosis after completing the prednisone taper. With these treatments, her facial lesions cleared and her breathing symptoms and fatigue improved. She remains under surveillance with a multidisciplinary team.
Photos and text for Photo Rounds Friday courtesy of Jonathan Karnes, MD (copyright retained). Dr. Karnes is the medical director of MDFMR Dermatology Services, Augusta, ME.
1. Cozier Y, Berman J, Palmer J, et al. Sarcoidosis in black women in the United States: data from the Black Women's Health Study. Chest. 2011;139:144-150. doi: 10.1378/chest.10-0413
2. Foreman MG, Mannino DM, Kamugisha L, et al. Hospitalization for patients with sarcoidosis: 1979-2000. Sarcoidosis Vasc Diffuse Lung Dis. 2006;23:124-129.
3. Mirsaeidi M, Machado R, Schraufnagel D, et al. Racial difference in sarcoidosis mortality in the United States. Chest. 2015; 147: 438-449. doi: 10.1378/chest.14-1120
1. Cozier Y, Berman J, Palmer J, et al. Sarcoidosis in black women in the United States: data from the Black Women's Health Study. Chest. 2011;139:144-150. doi: 10.1378/chest.10-0413
2. Foreman MG, Mannino DM, Kamugisha L, et al. Hospitalization for patients with sarcoidosis: 1979-2000. Sarcoidosis Vasc Diffuse Lung Dis. 2006;23:124-129.
3. Mirsaeidi M, Machado R, Schraufnagel D, et al. Racial difference in sarcoidosis mortality in the United States. Chest. 2015; 147: 438-449. doi: 10.1378/chest.14-1120
Author Q&A: GI needs better parental leave policies
The authors note that this article can serve as a roadmap for institutions and practices to create a parental leave policy and return-to-work environment that attracts talent and supports a diverse and thriving workforce.
Despite a joint statement by the four main gastroenterology societies more than 25 years ago, few structural changes have been implemented to mandate a minimum of 12 weeks of parental leave for gastroenterologists.
We asked one of the article’s authors, Dr. Lauren D. Feld, a few questions about the motivation behind this article and the movement at large.
Q: What motivated you and your coauthors to write this article?
A: “It was a pleasure working with my coauthors – an incredible team of gender equity experts – Drs. Amy S. Oxentenko, Dawn Sears, Aline Charabaty, Loren G. Rabinowitz, and Julie K. Silver.
I’m grateful to Dr. May and Dr. Quezada for the invitation to write about the important topic of creating family-friendly work environments. My coauthors and I have noticed increasing support for women in gastroenterology.”
Q: Why is this issue important?
A: “Nationwide, women are leaving clinical and academic medicine at alarming rates. The incompatibility of parenthood with a traditional medical career has been identified as a major driver of retention issues across specialties. In addition to impacting retention, incompatibility with pregnancy and parenthood also impacts recruitment. Survey studies of internal medicine residents have identified concerns about family life as a major barrier to choosing gastroenterology as a specialty. Women in medicine have worked too hard to get to where they are to be excluded from or driven out of our field.
Beyond the impact on the physicians, there is a major impact on patients. Studies have identified that women patients’ preference for a woman endoscopist as well as the difficulty in finding women endoscopists has created a barrier to colon cancer screening for women. Areas of research have also gone understudied because they primarily impact women patients. We must work towards equity for the benefit of both physicians and patients.”
Q: What actions can practicing GI doctors take now to help support better parental leave and return to work policies?
A: “Start by reviewing this article and asking your human resources for your employer’s policies in this area. If your employer doesn’t have a parental leave policy, or if their policy is inadequate, discuss the importance of this with your leadership. Describing the cost impact of physicians leaving practice is a good way to justify the cost investment to support family friendly policies.”
The authors recommend policies outlined in the paper be consistent across genders with attention to equity for the LGBTQ+ community. The blueprint for parental leave and return to work department policies includes:
- Specific policies to support physicians during pregnancy, including endoscopy ergonomic accommodations.
- Components of a parental leave policy such as duration and adjusted RVUs to account for leave.
- Coverage models to consider during leave.
- How to create a family friendly return to work, including modified overnight call during postpartum and autonomy over schedule.
The authors note that this article can serve as a roadmap for institutions and practices to create a parental leave policy and return-to-work environment that attracts talent and supports a diverse and thriving workforce.
Despite a joint statement by the four main gastroenterology societies more than 25 years ago, few structural changes have been implemented to mandate a minimum of 12 weeks of parental leave for gastroenterologists.
We asked one of the article’s authors, Dr. Lauren D. Feld, a few questions about the motivation behind this article and the movement at large.
Q: What motivated you and your coauthors to write this article?
A: “It was a pleasure working with my coauthors – an incredible team of gender equity experts – Drs. Amy S. Oxentenko, Dawn Sears, Aline Charabaty, Loren G. Rabinowitz, and Julie K. Silver.
I’m grateful to Dr. May and Dr. Quezada for the invitation to write about the important topic of creating family-friendly work environments. My coauthors and I have noticed increasing support for women in gastroenterology.”
Q: Why is this issue important?
A: “Nationwide, women are leaving clinical and academic medicine at alarming rates. The incompatibility of parenthood with a traditional medical career has been identified as a major driver of retention issues across specialties. In addition to impacting retention, incompatibility with pregnancy and parenthood also impacts recruitment. Survey studies of internal medicine residents have identified concerns about family life as a major barrier to choosing gastroenterology as a specialty. Women in medicine have worked too hard to get to where they are to be excluded from or driven out of our field.
Beyond the impact on the physicians, there is a major impact on patients. Studies have identified that women patients’ preference for a woman endoscopist as well as the difficulty in finding women endoscopists has created a barrier to colon cancer screening for women. Areas of research have also gone understudied because they primarily impact women patients. We must work towards equity for the benefit of both physicians and patients.”
Q: What actions can practicing GI doctors take now to help support better parental leave and return to work policies?
A: “Start by reviewing this article and asking your human resources for your employer’s policies in this area. If your employer doesn’t have a parental leave policy, or if their policy is inadequate, discuss the importance of this with your leadership. Describing the cost impact of physicians leaving practice is a good way to justify the cost investment to support family friendly policies.”
The authors recommend policies outlined in the paper be consistent across genders with attention to equity for the LGBTQ+ community. The blueprint for parental leave and return to work department policies includes:
- Specific policies to support physicians during pregnancy, including endoscopy ergonomic accommodations.
- Components of a parental leave policy such as duration and adjusted RVUs to account for leave.
- Coverage models to consider during leave.
- How to create a family friendly return to work, including modified overnight call during postpartum and autonomy over schedule.
The authors note that this article can serve as a roadmap for institutions and practices to create a parental leave policy and return-to-work environment that attracts talent and supports a diverse and thriving workforce.
Despite a joint statement by the four main gastroenterology societies more than 25 years ago, few structural changes have been implemented to mandate a minimum of 12 weeks of parental leave for gastroenterologists.
We asked one of the article’s authors, Dr. Lauren D. Feld, a few questions about the motivation behind this article and the movement at large.
Q: What motivated you and your coauthors to write this article?
A: “It was a pleasure working with my coauthors – an incredible team of gender equity experts – Drs. Amy S. Oxentenko, Dawn Sears, Aline Charabaty, Loren G. Rabinowitz, and Julie K. Silver.
I’m grateful to Dr. May and Dr. Quezada for the invitation to write about the important topic of creating family-friendly work environments. My coauthors and I have noticed increasing support for women in gastroenterology.”
Q: Why is this issue important?
A: “Nationwide, women are leaving clinical and academic medicine at alarming rates. The incompatibility of parenthood with a traditional medical career has been identified as a major driver of retention issues across specialties. In addition to impacting retention, incompatibility with pregnancy and parenthood also impacts recruitment. Survey studies of internal medicine residents have identified concerns about family life as a major barrier to choosing gastroenterology as a specialty. Women in medicine have worked too hard to get to where they are to be excluded from or driven out of our field.
Beyond the impact on the physicians, there is a major impact on patients. Studies have identified that women patients’ preference for a woman endoscopist as well as the difficulty in finding women endoscopists has created a barrier to colon cancer screening for women. Areas of research have also gone understudied because they primarily impact women patients. We must work towards equity for the benefit of both physicians and patients.”
Q: What actions can practicing GI doctors take now to help support better parental leave and return to work policies?
A: “Start by reviewing this article and asking your human resources for your employer’s policies in this area. If your employer doesn’t have a parental leave policy, or if their policy is inadequate, discuss the importance of this with your leadership. Describing the cost impact of physicians leaving practice is a good way to justify the cost investment to support family friendly policies.”
The authors recommend policies outlined in the paper be consistent across genders with attention to equity for the LGBTQ+ community. The blueprint for parental leave and return to work department policies includes:
- Specific policies to support physicians during pregnancy, including endoscopy ergonomic accommodations.
- Components of a parental leave policy such as duration and adjusted RVUs to account for leave.
- Coverage models to consider during leave.
- How to create a family friendly return to work, including modified overnight call during postpartum and autonomy over schedule.
Commentary: Examining Inpatient Admission, Hypothyroidism, and Vestibular Migraine, November 2023
Inpatient headache admissions are often considered a last-case scenario option for patients with chronic refractory migraine. Initially described by Raskin,1 the admission typically consists of repetitive infusions of dihydroergotamine (DHE) with a pretreatment of antihistamine, neuroleptic, and other antinausea medications in addition to anti-inflammatory or steroid medications, IV fluids, and magnesium. Often this is superimposed on a continuous infusion of lidocaine or ketamine. One common concern is whether the use of DHE is safe for patients at a higher risk for vascular disease. Wang and colleagues reported on the incidence of cardiovascular adverse events in this population receiving repetitive intravenous DHE.
They present findings based on the Jefferson Headache Center (Philadelphia, Pennsylvania) Inpatient Headache Protocol, looking at patients admitted from January through October 2019. Of the 347 patients admitted during this period, 64 were identified as having either an elevated or low risk for atherosclerotic vascular disease. The degree of vascular risk was determined on the basis of an atherosclerotic cardiovascular disease calculation, using body mass index values, cholesterol level, and mean arterial blood pressure readings, as well as smoking and diabetes history. A score < 5.0% was designated low risk, while elevated risk included scores up to 20%. DHE was not offered to patients with a history of moderate to severe ischemic heart disease, coronary vasospasm, peripheral artery disease, Raynaud phenomenon, or ischemic stroke.
The primary outcome was treatment effectiveness, determined by an 11-point pain scale; secondary outcomes included tolerability, as defined by change in the patient's QTc (corrected QT interval) based on their daily ECG monitoring, the incidence of chest pain or shortness of breath, and whether DHE needed to be tapered or discontinued. The researchers noted that the elevated-vascular-risk group had fewer patients receiving the maximum dose of DHE and receiving less DHE over the course of their admission as compared with the low-risk group. They also reported lower response rates and less freedom from pain after admission. No clinically significant adverse events were noted in either group, and only three patients had sustained ECG changes from baseline.
DHE remains an effective treatment for the most chronic and refractory migraine cases, and it can be provided safely and effectively in an appropriately monitored setting. Although there still are contraindications for receiving DHE, those who can receive it may benefit significantly. Ideally, this should be done with cardiac clearance if there is any doubt regarding the vascular risk for any individual patient.
The frequency and severity of migraine can fluctuate due to a myriad of factors. When faced with worsening migraine, most healthcare providers ask their patients about specific triggers or other potential causes that may have led to the recent subacute worsening. Many healthcare providers will also investigate further, and when appropriate, order serum lab testing to determine whether any potential metabolic derangement, vitamin deficiency, or other abnormality could be contributing. Subclinical hypothyroidism is a common finding when investigating for these potential causes of worsening migraine, and often our internal medicine or endocrinology colleagues will discount these findings as "borderline" or still within normal limits. Dev and colleagues sought to determine whether low-dose thyroid replacement was beneficial for migraine prevention in this situation.
This study defined subclinical hypothyroidism as a thyroid-stimulating hormone (TSH) level of 4.5-10.0 mIU/L with a normal free thyroxine (T4), measured twice within 6 weeks for confirmation. Patients with prior thyroid disease were excluded from the study. Participants were randomly assigned to take 25 μg levothyroxine supplementation or placebo and were allowed to continue their migraine treatments. The primary outcomes were reduction in headache duration, frequency, severity, and Migraine Disability Assessment (MIDAS) score after 3 months.
A total of 87 patients with migraine and subclinical hypothyroidism were recruited, and the investigators noted a statistically significant improvement in all parameters (migraine frequency, severity, duration, MIDAS score) after 3 months of low-dose thyroid supplementation. This was maintained at a 3-month follow-up visit as well. TSH levels normalized in 87% of participants after repeat testing 3 months after the intervention.
When evaluating patients with worsening migraine who had been stable, it is wise to consider subclinical hypothyroidism as a potential etiology. Low-dose thyroid hormone supplementation appears to be well tolerated and effective for normalizing both TSH levels and, importantly, the migrainous exacerbation. Clinicians often will start or add preventive medications, ignoring the reason that there was an exacerbation in the first place, even though many of our colleagues choose not to treat this degree of hypothyroidism.
Many variants of migraine are better recognized and understood now; chief among these is vestibular migraine (VM). VM is a migraine subtype characterized by frequent or near-constant vestibular symptoms (vertigo, lightheadedness, disequilibrium, or rocking) with superimposed headache symptoms with some migrainous features. VM is generally considered to be more difficult to treat and more treatment-refractory than episodic or chronic migraine without vestibular symptoms. There are few treatments that are specific for this variant of migraine. Chen and colleagues sought to better understand the evidence of specific treatments for VM via meta-analysis.
Only randomized controlled trials were included in this meta-analysis; seven studies that specifically recruited patients with vestibular migraine using International Classification of Headache Disorders (ICHD) criteria were included. The outcomes of the studies were changes in frequency or severity of vestibular migraine attacks. The studies that were included were published from 2014 to 2022 and comprised multiple treatment comparisons, including metoprolol, venlafaxine, valproic acid, propranolol, flunarizine, a probiotic, and relaxation techniques.
Three interventions were noted to be significantly beneficial for VM prevention, specifically a decrease in migraine frequency: valproic acid, propranolol, and venlafaxine. Valproic acid yielded the greatest decrease in VM frequency among all interventions. None of the interventions were associated with improvement in VM severity, and none of the treatments were associated with significantly different adverse-event and dropout rates.
VM is widely thought to be underdiagnosed and should be considered more frequently. This includes situations in which the headache component of the patient's complaints is relatively mild but still associated with features of migraines, such as sensitivities to light and sound, nausea, or unilateral presentations of pain. There remain very few high-quality VM studies, but this meta-analysis should highlight potential treatment options and raise the profile for this diagnosis in order for further trials to be performed.
Additional Reference
1. Raskin NH. Repetitive intravenous dihydroergotamine as therapy for intractable migraine. Neurology. 1986;36(7):995-997. doi: 10.1212/WNL.36.7.995
Inpatient headache admissions are often considered a last-case scenario option for patients with chronic refractory migraine. Initially described by Raskin,1 the admission typically consists of repetitive infusions of dihydroergotamine (DHE) with a pretreatment of antihistamine, neuroleptic, and other antinausea medications in addition to anti-inflammatory or steroid medications, IV fluids, and magnesium. Often this is superimposed on a continuous infusion of lidocaine or ketamine. One common concern is whether the use of DHE is safe for patients at a higher risk for vascular disease. Wang and colleagues reported on the incidence of cardiovascular adverse events in this population receiving repetitive intravenous DHE.
They present findings based on the Jefferson Headache Center (Philadelphia, Pennsylvania) Inpatient Headache Protocol, looking at patients admitted from January through October 2019. Of the 347 patients admitted during this period, 64 were identified as having either an elevated or low risk for atherosclerotic vascular disease. The degree of vascular risk was determined on the basis of an atherosclerotic cardiovascular disease calculation, using body mass index values, cholesterol level, and mean arterial blood pressure readings, as well as smoking and diabetes history. A score < 5.0% was designated low risk, while elevated risk included scores up to 20%. DHE was not offered to patients with a history of moderate to severe ischemic heart disease, coronary vasospasm, peripheral artery disease, Raynaud phenomenon, or ischemic stroke.
The primary outcome was treatment effectiveness, determined by an 11-point pain scale; secondary outcomes included tolerability, as defined by change in the patient's QTc (corrected QT interval) based on their daily ECG monitoring, the incidence of chest pain or shortness of breath, and whether DHE needed to be tapered or discontinued. The researchers noted that the elevated-vascular-risk group had fewer patients receiving the maximum dose of DHE and receiving less DHE over the course of their admission as compared with the low-risk group. They also reported lower response rates and less freedom from pain after admission. No clinically significant adverse events were noted in either group, and only three patients had sustained ECG changes from baseline.
DHE remains an effective treatment for the most chronic and refractory migraine cases, and it can be provided safely and effectively in an appropriately monitored setting. Although there still are contraindications for receiving DHE, those who can receive it may benefit significantly. Ideally, this should be done with cardiac clearance if there is any doubt regarding the vascular risk for any individual patient.
The frequency and severity of migraine can fluctuate due to a myriad of factors. When faced with worsening migraine, most healthcare providers ask their patients about specific triggers or other potential causes that may have led to the recent subacute worsening. Many healthcare providers will also investigate further, and when appropriate, order serum lab testing to determine whether any potential metabolic derangement, vitamin deficiency, or other abnormality could be contributing. Subclinical hypothyroidism is a common finding when investigating for these potential causes of worsening migraine, and often our internal medicine or endocrinology colleagues will discount these findings as "borderline" or still within normal limits. Dev and colleagues sought to determine whether low-dose thyroid replacement was beneficial for migraine prevention in this situation.
This study defined subclinical hypothyroidism as a thyroid-stimulating hormone (TSH) level of 4.5-10.0 mIU/L with a normal free thyroxine (T4), measured twice within 6 weeks for confirmation. Patients with prior thyroid disease were excluded from the study. Participants were randomly assigned to take 25 μg levothyroxine supplementation or placebo and were allowed to continue their migraine treatments. The primary outcomes were reduction in headache duration, frequency, severity, and Migraine Disability Assessment (MIDAS) score after 3 months.
A total of 87 patients with migraine and subclinical hypothyroidism were recruited, and the investigators noted a statistically significant improvement in all parameters (migraine frequency, severity, duration, MIDAS score) after 3 months of low-dose thyroid supplementation. This was maintained at a 3-month follow-up visit as well. TSH levels normalized in 87% of participants after repeat testing 3 months after the intervention.
When evaluating patients with worsening migraine who had been stable, it is wise to consider subclinical hypothyroidism as a potential etiology. Low-dose thyroid hormone supplementation appears to be well tolerated and effective for normalizing both TSH levels and, importantly, the migrainous exacerbation. Clinicians often will start or add preventive medications, ignoring the reason that there was an exacerbation in the first place, even though many of our colleagues choose not to treat this degree of hypothyroidism.
Many variants of migraine are better recognized and understood now; chief among these is vestibular migraine (VM). VM is a migraine subtype characterized by frequent or near-constant vestibular symptoms (vertigo, lightheadedness, disequilibrium, or rocking) with superimposed headache symptoms with some migrainous features. VM is generally considered to be more difficult to treat and more treatment-refractory than episodic or chronic migraine without vestibular symptoms. There are few treatments that are specific for this variant of migraine. Chen and colleagues sought to better understand the evidence of specific treatments for VM via meta-analysis.
Only randomized controlled trials were included in this meta-analysis; seven studies that specifically recruited patients with vestibular migraine using International Classification of Headache Disorders (ICHD) criteria were included. The outcomes of the studies were changes in frequency or severity of vestibular migraine attacks. The studies that were included were published from 2014 to 2022 and comprised multiple treatment comparisons, including metoprolol, venlafaxine, valproic acid, propranolol, flunarizine, a probiotic, and relaxation techniques.
Three interventions were noted to be significantly beneficial for VM prevention, specifically a decrease in migraine frequency: valproic acid, propranolol, and venlafaxine. Valproic acid yielded the greatest decrease in VM frequency among all interventions. None of the interventions were associated with improvement in VM severity, and none of the treatments were associated with significantly different adverse-event and dropout rates.
VM is widely thought to be underdiagnosed and should be considered more frequently. This includes situations in which the headache component of the patient's complaints is relatively mild but still associated with features of migraines, such as sensitivities to light and sound, nausea, or unilateral presentations of pain. There remain very few high-quality VM studies, but this meta-analysis should highlight potential treatment options and raise the profile for this diagnosis in order for further trials to be performed.
Additional Reference
1. Raskin NH. Repetitive intravenous dihydroergotamine as therapy for intractable migraine. Neurology. 1986;36(7):995-997. doi: 10.1212/WNL.36.7.995
Inpatient headache admissions are often considered a last-case scenario option for patients with chronic refractory migraine. Initially described by Raskin,1 the admission typically consists of repetitive infusions of dihydroergotamine (DHE) with a pretreatment of antihistamine, neuroleptic, and other antinausea medications in addition to anti-inflammatory or steroid medications, IV fluids, and magnesium. Often this is superimposed on a continuous infusion of lidocaine or ketamine. One common concern is whether the use of DHE is safe for patients at a higher risk for vascular disease. Wang and colleagues reported on the incidence of cardiovascular adverse events in this population receiving repetitive intravenous DHE.
They present findings based on the Jefferson Headache Center (Philadelphia, Pennsylvania) Inpatient Headache Protocol, looking at patients admitted from January through October 2019. Of the 347 patients admitted during this period, 64 were identified as having either an elevated or low risk for atherosclerotic vascular disease. The degree of vascular risk was determined on the basis of an atherosclerotic cardiovascular disease calculation, using body mass index values, cholesterol level, and mean arterial blood pressure readings, as well as smoking and diabetes history. A score < 5.0% was designated low risk, while elevated risk included scores up to 20%. DHE was not offered to patients with a history of moderate to severe ischemic heart disease, coronary vasospasm, peripheral artery disease, Raynaud phenomenon, or ischemic stroke.
The primary outcome was treatment effectiveness, determined by an 11-point pain scale; secondary outcomes included tolerability, as defined by change in the patient's QTc (corrected QT interval) based on their daily ECG monitoring, the incidence of chest pain or shortness of breath, and whether DHE needed to be tapered or discontinued. The researchers noted that the elevated-vascular-risk group had fewer patients receiving the maximum dose of DHE and receiving less DHE over the course of their admission as compared with the low-risk group. They also reported lower response rates and less freedom from pain after admission. No clinically significant adverse events were noted in either group, and only three patients had sustained ECG changes from baseline.
DHE remains an effective treatment for the most chronic and refractory migraine cases, and it can be provided safely and effectively in an appropriately monitored setting. Although there still are contraindications for receiving DHE, those who can receive it may benefit significantly. Ideally, this should be done with cardiac clearance if there is any doubt regarding the vascular risk for any individual patient.
The frequency and severity of migraine can fluctuate due to a myriad of factors. When faced with worsening migraine, most healthcare providers ask their patients about specific triggers or other potential causes that may have led to the recent subacute worsening. Many healthcare providers will also investigate further, and when appropriate, order serum lab testing to determine whether any potential metabolic derangement, vitamin deficiency, or other abnormality could be contributing. Subclinical hypothyroidism is a common finding when investigating for these potential causes of worsening migraine, and often our internal medicine or endocrinology colleagues will discount these findings as "borderline" or still within normal limits. Dev and colleagues sought to determine whether low-dose thyroid replacement was beneficial for migraine prevention in this situation.
This study defined subclinical hypothyroidism as a thyroid-stimulating hormone (TSH) level of 4.5-10.0 mIU/L with a normal free thyroxine (T4), measured twice within 6 weeks for confirmation. Patients with prior thyroid disease were excluded from the study. Participants were randomly assigned to take 25 μg levothyroxine supplementation or placebo and were allowed to continue their migraine treatments. The primary outcomes were reduction in headache duration, frequency, severity, and Migraine Disability Assessment (MIDAS) score after 3 months.
A total of 87 patients with migraine and subclinical hypothyroidism were recruited, and the investigators noted a statistically significant improvement in all parameters (migraine frequency, severity, duration, MIDAS score) after 3 months of low-dose thyroid supplementation. This was maintained at a 3-month follow-up visit as well. TSH levels normalized in 87% of participants after repeat testing 3 months after the intervention.
When evaluating patients with worsening migraine who had been stable, it is wise to consider subclinical hypothyroidism as a potential etiology. Low-dose thyroid hormone supplementation appears to be well tolerated and effective for normalizing both TSH levels and, importantly, the migrainous exacerbation. Clinicians often will start or add preventive medications, ignoring the reason that there was an exacerbation in the first place, even though many of our colleagues choose not to treat this degree of hypothyroidism.
Many variants of migraine are better recognized and understood now; chief among these is vestibular migraine (VM). VM is a migraine subtype characterized by frequent or near-constant vestibular symptoms (vertigo, lightheadedness, disequilibrium, or rocking) with superimposed headache symptoms with some migrainous features. VM is generally considered to be more difficult to treat and more treatment-refractory than episodic or chronic migraine without vestibular symptoms. There are few treatments that are specific for this variant of migraine. Chen and colleagues sought to better understand the evidence of specific treatments for VM via meta-analysis.
Only randomized controlled trials were included in this meta-analysis; seven studies that specifically recruited patients with vestibular migraine using International Classification of Headache Disorders (ICHD) criteria were included. The outcomes of the studies were changes in frequency or severity of vestibular migraine attacks. The studies that were included were published from 2014 to 2022 and comprised multiple treatment comparisons, including metoprolol, venlafaxine, valproic acid, propranolol, flunarizine, a probiotic, and relaxation techniques.
Three interventions were noted to be significantly beneficial for VM prevention, specifically a decrease in migraine frequency: valproic acid, propranolol, and venlafaxine. Valproic acid yielded the greatest decrease in VM frequency among all interventions. None of the interventions were associated with improvement in VM severity, and none of the treatments were associated with significantly different adverse-event and dropout rates.
VM is widely thought to be underdiagnosed and should be considered more frequently. This includes situations in which the headache component of the patient's complaints is relatively mild but still associated with features of migraines, such as sensitivities to light and sound, nausea, or unilateral presentations of pain. There remain very few high-quality VM studies, but this meta-analysis should highlight potential treatment options and raise the profile for this diagnosis in order for further trials to be performed.
Additional Reference
1. Raskin NH. Repetitive intravenous dihydroergotamine as therapy for intractable migraine. Neurology. 1986;36(7):995-997. doi: 10.1212/WNL.36.7.995
Forgetfulness and mood fluctuations
This patient's symptoms go beyond just memory problems: She has difficulty with daily tasks, shows behavioral changes, and has significant communication difficulties — symptoms not found in mild cognitive impairment. While the patient has some behavioral changes, she does not exhibit the pronounced personality changes typical of frontotemporal dementia. Finally, the patient's cognitive decline is gradual and consistent without the stepwise progression typical of vascular dementia. Given the comprehensive presentation of the patient's symptoms and the results of her clinical investigations, middle-stage Alzheimer's disease is the most fitting diagnosis.
Alzheimer's disease is a progressive and irreversible brain disorder that affects memory, behavior, and cognitive skills. This condition causes the degeneration and death of brain cells, leading to various cognitive issues. Alzheimer's disease is the most common cause of dementia and accounts for 60%-80% of dementia cases. Although the exact cause is unknown, it is believed to result from genetic, lifestyle, and environmental factors. Alzheimer's disease progresses through stages — mild (early stage), moderate (middle stage), and severe (late stage) — and each stage has different signs and symptoms.
Alzheimer's disease is commonly observed in individuals 65 years or older, as age is the most significant risk factor. Another risk factor for Alzheimer's disease is family history; individuals who have parents or siblings with Alzheimer's disease are more likely to develop the disease. The risk increases with the number of family members diagnosed with the disease. Genetics also contribute to the development of Alzheimer's disease. Genes for developing Alzheimer's disease have been classified as deterministic and risk genes, which imply that they can cause the disease or increase the risk of developing it; however, the deterministic gene, which almost guarantees the occurrence of Alzheimer's, is rare and is found in less than 1% of cases. Experiencing a head injury is also a possible risk factor for Alzheimer's disease.
Accurate diagnosis of Alzheimer's disease requires a thorough history and physical examination. Gathering information from the patient's family and caregivers is important because some patients may not be aware of their condition. It is common for Alzheimer's disease patients to experience "sundowning," which causes confusion, agitation, and behavioral issues in the evening. A comprehensive physical examination, including a detailed neurologic and mental status exam, is necessary to determine the stage of the disease and rule out other conditions. Typically, the neurologic exam of Alzheimer's disease patients is normal.
Volumetric MRI is a recent technique that allows precise measurement of changes in brain volume. In Alzheimer's disease, shrinkage in the medial temporal lobe is visible through volumetric MRI. However, hippocampal atrophy is also a normal part of age-related memory decline, which raises doubts about the appropriateness of using volumetric MRI for early detection of Alzheimer's disease. The full potential of volumetric MRI in aiding the diagnosis of Alzheimer's disease is yet to be fully established.
Alzheimer's disease has no known cure, and treatment options are limited to addressing symptoms. Currently, three types of drugs are approved for treating the moderate or severe stages of the disease: cholinesterase inhibitors, partial N-methyl D-aspartate (NMDA) antagonists, and amyloid-directed antibodies. Cholinesterase inhibitors increase acetylcholine levels, a chemical crucial for cognitive functions such as memory and learning. NMDA antagonists (memantine) blocks NMDA receptors whose overactivation is implicated in Alzheimer's disease and related to synaptic dysfunction. Antiamyloid monoclonal antibodies bind to and promote the clearance of amyloid-beta peptides, thereby reducing amyloid plaques in the brain, which are associated with Alzheimer's disease.
Jasvinder Chawla, MD, Professor of Neurology, Loyola University Medical Center, Maywood; Director, Clinical Neurophysiology Lab, Department of Neurology, Hines VA Hospital, Hines, IL.
Jasvinder Chawla, MD, has disclosed no relevant financial relationships.
Image Quizzes are fictional or fictionalized clinical scenarios intended to provide evidence-based educational takeaways.
This patient's symptoms go beyond just memory problems: She has difficulty with daily tasks, shows behavioral changes, and has significant communication difficulties — symptoms not found in mild cognitive impairment. While the patient has some behavioral changes, she does not exhibit the pronounced personality changes typical of frontotemporal dementia. Finally, the patient's cognitive decline is gradual and consistent without the stepwise progression typical of vascular dementia. Given the comprehensive presentation of the patient's symptoms and the results of her clinical investigations, middle-stage Alzheimer's disease is the most fitting diagnosis.
Alzheimer's disease is a progressive and irreversible brain disorder that affects memory, behavior, and cognitive skills. This condition causes the degeneration and death of brain cells, leading to various cognitive issues. Alzheimer's disease is the most common cause of dementia and accounts for 60%-80% of dementia cases. Although the exact cause is unknown, it is believed to result from genetic, lifestyle, and environmental factors. Alzheimer's disease progresses through stages — mild (early stage), moderate (middle stage), and severe (late stage) — and each stage has different signs and symptoms.
Alzheimer's disease is commonly observed in individuals 65 years or older, as age is the most significant risk factor. Another risk factor for Alzheimer's disease is family history; individuals who have parents or siblings with Alzheimer's disease are more likely to develop the disease. The risk increases with the number of family members diagnosed with the disease. Genetics also contribute to the development of Alzheimer's disease. Genes for developing Alzheimer's disease have been classified as deterministic and risk genes, which imply that they can cause the disease or increase the risk of developing it; however, the deterministic gene, which almost guarantees the occurrence of Alzheimer's, is rare and is found in less than 1% of cases. Experiencing a head injury is also a possible risk factor for Alzheimer's disease.
Accurate diagnosis of Alzheimer's disease requires a thorough history and physical examination. Gathering information from the patient's family and caregivers is important because some patients may not be aware of their condition. It is common for Alzheimer's disease patients to experience "sundowning," which causes confusion, agitation, and behavioral issues in the evening. A comprehensive physical examination, including a detailed neurologic and mental status exam, is necessary to determine the stage of the disease and rule out other conditions. Typically, the neurologic exam of Alzheimer's disease patients is normal.
Volumetric MRI is a recent technique that allows precise measurement of changes in brain volume. In Alzheimer's disease, shrinkage in the medial temporal lobe is visible through volumetric MRI. However, hippocampal atrophy is also a normal part of age-related memory decline, which raises doubts about the appropriateness of using volumetric MRI for early detection of Alzheimer's disease. The full potential of volumetric MRI in aiding the diagnosis of Alzheimer's disease is yet to be fully established.
Alzheimer's disease has no known cure, and treatment options are limited to addressing symptoms. Currently, three types of drugs are approved for treating the moderate or severe stages of the disease: cholinesterase inhibitors, partial N-methyl D-aspartate (NMDA) antagonists, and amyloid-directed antibodies. Cholinesterase inhibitors increase acetylcholine levels, a chemical crucial for cognitive functions such as memory and learning. NMDA antagonists (memantine) blocks NMDA receptors whose overactivation is implicated in Alzheimer's disease and related to synaptic dysfunction. Antiamyloid monoclonal antibodies bind to and promote the clearance of amyloid-beta peptides, thereby reducing amyloid plaques in the brain, which are associated with Alzheimer's disease.
Jasvinder Chawla, MD, Professor of Neurology, Loyola University Medical Center, Maywood; Director, Clinical Neurophysiology Lab, Department of Neurology, Hines VA Hospital, Hines, IL.
Jasvinder Chawla, MD, has disclosed no relevant financial relationships.
Image Quizzes are fictional or fictionalized clinical scenarios intended to provide evidence-based educational takeaways.
This patient's symptoms go beyond just memory problems: She has difficulty with daily tasks, shows behavioral changes, and has significant communication difficulties — symptoms not found in mild cognitive impairment. While the patient has some behavioral changes, she does not exhibit the pronounced personality changes typical of frontotemporal dementia. Finally, the patient's cognitive decline is gradual and consistent without the stepwise progression typical of vascular dementia. Given the comprehensive presentation of the patient's symptoms and the results of her clinical investigations, middle-stage Alzheimer's disease is the most fitting diagnosis.
Alzheimer's disease is a progressive and irreversible brain disorder that affects memory, behavior, and cognitive skills. This condition causes the degeneration and death of brain cells, leading to various cognitive issues. Alzheimer's disease is the most common cause of dementia and accounts for 60%-80% of dementia cases. Although the exact cause is unknown, it is believed to result from genetic, lifestyle, and environmental factors. Alzheimer's disease progresses through stages — mild (early stage), moderate (middle stage), and severe (late stage) — and each stage has different signs and symptoms.
Alzheimer's disease is commonly observed in individuals 65 years or older, as age is the most significant risk factor. Another risk factor for Alzheimer's disease is family history; individuals who have parents or siblings with Alzheimer's disease are more likely to develop the disease. The risk increases with the number of family members diagnosed with the disease. Genetics also contribute to the development of Alzheimer's disease. Genes for developing Alzheimer's disease have been classified as deterministic and risk genes, which imply that they can cause the disease or increase the risk of developing it; however, the deterministic gene, which almost guarantees the occurrence of Alzheimer's, is rare and is found in less than 1% of cases. Experiencing a head injury is also a possible risk factor for Alzheimer's disease.
Accurate diagnosis of Alzheimer's disease requires a thorough history and physical examination. Gathering information from the patient's family and caregivers is important because some patients may not be aware of their condition. It is common for Alzheimer's disease patients to experience "sundowning," which causes confusion, agitation, and behavioral issues in the evening. A comprehensive physical examination, including a detailed neurologic and mental status exam, is necessary to determine the stage of the disease and rule out other conditions. Typically, the neurologic exam of Alzheimer's disease patients is normal.
Volumetric MRI is a recent technique that allows precise measurement of changes in brain volume. In Alzheimer's disease, shrinkage in the medial temporal lobe is visible through volumetric MRI. However, hippocampal atrophy is also a normal part of age-related memory decline, which raises doubts about the appropriateness of using volumetric MRI for early detection of Alzheimer's disease. The full potential of volumetric MRI in aiding the diagnosis of Alzheimer's disease is yet to be fully established.
Alzheimer's disease has no known cure, and treatment options are limited to addressing symptoms. Currently, three types of drugs are approved for treating the moderate or severe stages of the disease: cholinesterase inhibitors, partial N-methyl D-aspartate (NMDA) antagonists, and amyloid-directed antibodies. Cholinesterase inhibitors increase acetylcholine levels, a chemical crucial for cognitive functions such as memory and learning. NMDA antagonists (memantine) blocks NMDA receptors whose overactivation is implicated in Alzheimer's disease and related to synaptic dysfunction. Antiamyloid monoclonal antibodies bind to and promote the clearance of amyloid-beta peptides, thereby reducing amyloid plaques in the brain, which are associated with Alzheimer's disease.
Jasvinder Chawla, MD, Professor of Neurology, Loyola University Medical Center, Maywood; Director, Clinical Neurophysiology Lab, Department of Neurology, Hines VA Hospital, Hines, IL.
Jasvinder Chawla, MD, has disclosed no relevant financial relationships.
Image Quizzes are fictional or fictionalized clinical scenarios intended to provide evidence-based educational takeaways.
The patient is a 72-year-old retired schoolteacher accompanied by her daughter. Over the past year, her family has become increasingly concerned about her forgetfulness, mood fluctuations, and challenges in performing daily activities. The patient often forgets her grandchildren's names and struggles to recall significant recent events. She frequently misplaces household items and has missed several appointments. During her consultation, she has difficulty finding the right words, often repeats herself, and seems to lose track of the conversation. Her daughter shared concerning incidents, such as the patient wearing heavy sweaters during hot summer days and falling victim to a phone scam, which was uncharacteristic of her previous discerning nature. Additionally, the patient has become more reclusive, avoiding the social gatherings she once loved. She occasionally exhibits signs of agitation, especially in the evening. She has also stopped cooking as a result of instances of forgetting to turn off the stove and has had challenges managing her finances, leading to unpaid bills. A thorough neurologic exam is performed and is normal. Coronal T1-weighted MRI reveals hippocampal atrophy, particularly on the right side.