User login
In critically ill patients, dalteparin is more cost-effective for VTE prevention
The low molecular weight heparin dalteparin and unfractionated heparin are associated with similar rates of thrombosis and major bleeding, but dalteparin is associated with lower rates of pulmonary embolus and heparin-induced thrombocytopenia, based on results from a prospective randomized study.
Given for prevention of venous thromboembolism, median hospital costs per patient were $39,508 for dalteparin users and $40,805 for unfractionated heparin users. Dalteparin remained the least costly strategy until its acquisition costs rose from $8 per dose to $179, as reported online 1 November in the Journal of the American Medical Association [doi:10.1001/jama.2014.15101].
The economic analysis—conducted alongside the multi-centre, randomized PROTECT trial in 2344 critically-ill medical-surgical patients— showed no matter how low the acquisition cost of unfractionated heparin, there was no threshold that favored that form of prophylaxis, according to data also presented at the Critical Care Canada Forum.
“From a health care payer perspective, VTE prophylaxis with the LMWH [low molecular weight heparin] dalteparin in critically ill medical-surgical patients was more effective and had similar or lower costs than the use of UFH [unfractionated heparin],” wrote Dr. Robert A. Fowler, from the Sunnybrook Health Sciences Centre, University of Toronto, and colleagues.The E-PROTECT study was funded by the Heart and Stroke Foundation (Ontario, Canada), the University of Toronto, and the Canadian Intensive Care Foundation. PROTECT was funded by the Canadian Institutes of Health Research, the Heart and Stroke Foundation (Canada), and the Australian and New Zealand College of Anesthetists Research Foundation. Some authors reported fees, support, and consultancies from the pharmaceutical industry.
The low molecular weight heparin dalteparin and unfractionated heparin are associated with similar rates of thrombosis and major bleeding, but dalteparin is associated with lower rates of pulmonary embolus and heparin-induced thrombocytopenia, based on results from a prospective randomized study.
Given for prevention of venous thromboembolism, median hospital costs per patient were $39,508 for dalteparin users and $40,805 for unfractionated heparin users. Dalteparin remained the least costly strategy until its acquisition costs rose from $8 per dose to $179, as reported online 1 November in the Journal of the American Medical Association [doi:10.1001/jama.2014.15101].
The economic analysis—conducted alongside the multi-centre, randomized PROTECT trial in 2344 critically-ill medical-surgical patients— showed no matter how low the acquisition cost of unfractionated heparin, there was no threshold that favored that form of prophylaxis, according to data also presented at the Critical Care Canada Forum.
“From a health care payer perspective, VTE prophylaxis with the LMWH [low molecular weight heparin] dalteparin in critically ill medical-surgical patients was more effective and had similar or lower costs than the use of UFH [unfractionated heparin],” wrote Dr. Robert A. Fowler, from the Sunnybrook Health Sciences Centre, University of Toronto, and colleagues.The E-PROTECT study was funded by the Heart and Stroke Foundation (Ontario, Canada), the University of Toronto, and the Canadian Intensive Care Foundation. PROTECT was funded by the Canadian Institutes of Health Research, the Heart and Stroke Foundation (Canada), and the Australian and New Zealand College of Anesthetists Research Foundation. Some authors reported fees, support, and consultancies from the pharmaceutical industry.
The low molecular weight heparin dalteparin and unfractionated heparin are associated with similar rates of thrombosis and major bleeding, but dalteparin is associated with lower rates of pulmonary embolus and heparin-induced thrombocytopenia, based on results from a prospective randomized study.
Given for prevention of venous thromboembolism, median hospital costs per patient were $39,508 for dalteparin users and $40,805 for unfractionated heparin users. Dalteparin remained the least costly strategy until its acquisition costs rose from $8 per dose to $179, as reported online 1 November in the Journal of the American Medical Association [doi:10.1001/jama.2014.15101].
The economic analysis—conducted alongside the multi-centre, randomized PROTECT trial in 2344 critically-ill medical-surgical patients— showed no matter how low the acquisition cost of unfractionated heparin, there was no threshold that favored that form of prophylaxis, according to data also presented at the Critical Care Canada Forum.
“From a health care payer perspective, VTE prophylaxis with the LMWH [low molecular weight heparin] dalteparin in critically ill medical-surgical patients was more effective and had similar or lower costs than the use of UFH [unfractionated heparin],” wrote Dr. Robert A. Fowler, from the Sunnybrook Health Sciences Centre, University of Toronto, and colleagues.The E-PROTECT study was funded by the Heart and Stroke Foundation (Ontario, Canada), the University of Toronto, and the Canadian Intensive Care Foundation. PROTECT was funded by the Canadian Institutes of Health Research, the Heart and Stroke Foundation (Canada), and the Australian and New Zealand College of Anesthetists Research Foundation. Some authors reported fees, support, and consultancies from the pharmaceutical industry.
FROM JAMA
Key clinical point: Dalteparin is more cost-effective than unfractionated heparin in the prevention of venous thromboembolism.
Major finding: Dalteparin is as effective as unfractionated heparin in reducing thrombosis, for the same cost, but with less pulmonary embolus and heparin-induced thrombocytopenia.
Data source: Economic analysis of a prospective randomized controlled trial of low molecular weight heparin dalteparin versus unfractionated heparin in 2344 critically-ill medical-surgical patients
Disclosures: The E-PROTECT study was funded by the Heart and Stroke Foundation (Ontario, Canada), the University of Toronto, and the Canadian Intensive Care Foundation. PROTECT was funded by the Canadian Institutes of Health Research, the Heart and Stroke Foundation (Canada), and the Australian and New Zealand College of Anesthetists Research Foundation. Some authors reported fees, support, and consultancies to the pharmaceutical industry.
Androgen-deprivation therapy linked to cardiac death
Androgen-deprivation therapy for prostate cancer may increase the risk of cardiac death among men who have congestive heart failure or who have experienced a previous myocardial infarction, a retrospective cohort study has found.
The study of 5,077 men, whose prostate cancer was treated with brachytherapy with or without neoadjuvant androgen-deprivation therapy (ADT), showed that ADT was associated with a greater-than-threefold increase in the risk of cardiac specific mortality among men with heart failure or myocardial infarction (adjusted hazard ratio, 3.28; 95% confidence interval, 1.01-10.64; P = .048), representing a 5% increase in absolute risk over 5 years.
However, researchers saw no association between ADT and cardiac death among men with no cardiac risk factors, or those with diabetes mellitus, hypertension, or hypercholesterolemia, according to a paper published online Oct. 29 in BJU International (2014 [doi:10.1111/bju.12905]).
“These results add vital detail to past work that showed that the delivery of ADT to men with moderate to severe comorbidity is associated with no survival benefit, and that delivery of ADT to men with [HF] or past MI is associated with increased risk of death from any cause,” wrote Dr. David R. Ziehr from Harvard Medical School, Boston, and his colleagues.
The study was supported by Fitz’s CancerWarriors, David and Cynthia Chapin, the Prostate Cancer Foundation, Hugh Simons in honor of Frank and Anne Simons, and a grant from an anonymous family foundation. Some authors declared consultancies, lectureships, and advisory board positions for pharmaceutical companies.
Androgen-deprivation therapy for prostate cancer may increase the risk of cardiac death among men who have congestive heart failure or who have experienced a previous myocardial infarction, a retrospective cohort study has found.
The study of 5,077 men, whose prostate cancer was treated with brachytherapy with or without neoadjuvant androgen-deprivation therapy (ADT), showed that ADT was associated with a greater-than-threefold increase in the risk of cardiac specific mortality among men with heart failure or myocardial infarction (adjusted hazard ratio, 3.28; 95% confidence interval, 1.01-10.64; P = .048), representing a 5% increase in absolute risk over 5 years.
However, researchers saw no association between ADT and cardiac death among men with no cardiac risk factors, or those with diabetes mellitus, hypertension, or hypercholesterolemia, according to a paper published online Oct. 29 in BJU International (2014 [doi:10.1111/bju.12905]).
“These results add vital detail to past work that showed that the delivery of ADT to men with moderate to severe comorbidity is associated with no survival benefit, and that delivery of ADT to men with [HF] or past MI is associated with increased risk of death from any cause,” wrote Dr. David R. Ziehr from Harvard Medical School, Boston, and his colleagues.
The study was supported by Fitz’s CancerWarriors, David and Cynthia Chapin, the Prostate Cancer Foundation, Hugh Simons in honor of Frank and Anne Simons, and a grant from an anonymous family foundation. Some authors declared consultancies, lectureships, and advisory board positions for pharmaceutical companies.
Androgen-deprivation therapy for prostate cancer may increase the risk of cardiac death among men who have congestive heart failure or who have experienced a previous myocardial infarction, a retrospective cohort study has found.
The study of 5,077 men, whose prostate cancer was treated with brachytherapy with or without neoadjuvant androgen-deprivation therapy (ADT), showed that ADT was associated with a greater-than-threefold increase in the risk of cardiac specific mortality among men with heart failure or myocardial infarction (adjusted hazard ratio, 3.28; 95% confidence interval, 1.01-10.64; P = .048), representing a 5% increase in absolute risk over 5 years.
However, researchers saw no association between ADT and cardiac death among men with no cardiac risk factors, or those with diabetes mellitus, hypertension, or hypercholesterolemia, according to a paper published online Oct. 29 in BJU International (2014 [doi:10.1111/bju.12905]).
“These results add vital detail to past work that showed that the delivery of ADT to men with moderate to severe comorbidity is associated with no survival benefit, and that delivery of ADT to men with [HF] or past MI is associated with increased risk of death from any cause,” wrote Dr. David R. Ziehr from Harvard Medical School, Boston, and his colleagues.
The study was supported by Fitz’s CancerWarriors, David and Cynthia Chapin, the Prostate Cancer Foundation, Hugh Simons in honor of Frank and Anne Simons, and a grant from an anonymous family foundation. Some authors declared consultancies, lectureships, and advisory board positions for pharmaceutical companies.
FROM BJU INTERNATIONAL
Key clinical point: Androgen-deprivation therapy may increase in the risk of cardiac death in men with heart failure or prior myocardial infarction.
Major finding: Men with heart failure or a prior myocardial infarction who were treated with androgen-deprivation therapy showed a threefold increase in the risk of cardiac death.
Data source: Retrospective cohort study of 5,077 men with prostate cancer treated with androgen-deprivation therapy.
Disclosures: The study was supported by Fitz’s CancerWarriors, David and Cynthia Chapin, the Prostate Cancer Foundation, Hugh Simons in honor of Frank and Anne Simons, and a grant from an anonymous family foundation. Some authors declared consultancies, lectureships, and advisory board positions for pharmaceutical companies.
Metformin linked to less intensification of diabetes treatment
First-line treatment with metformin for diabetes is associated with significantly less intensification of treatment, and fewer short-term adverse outcomes such as cardiovascular events, emergency department visits, and hypoglycemia, compared with other oral glucose-lowering medications.
A retrospective cohort study of 15,516 patients prescribed an oral glucose-lowering medication found that 25% of patients initially prescribed metformin required a second oral agent, compared with 37% of patients prescribed a sulfonylurea, 40% of those prescribed a thiazolidinedione, and 36% of those prescribed a dipeptidyl peptidase–4 inhibitor, according to a retrospective study published online Oct. 27 (JAMA Intern. Med. 2014 Oct. 27 [doi: 10.1001/jamainternmed.2014.5294]).
While numerous guidelines recommend metformin as the first-line choice of glucose-lowering agents, only 58% of patients started therapy with metformin, with a sulfonylurea the second-most-popular choice, despite its increased risk of adverse cardiovascular events.
“Because underuse of metformin may lead to important harms and costs in the treatment of patients with diabetes, multilevel interventions to increase prescribing quality may be needed,” wrote Dr. Seth A. Berkowitz of Harvard University, Boston, and his colleagues.
The study was supported by an unrestricted grant from CVS Health to Brigham and Women’s Hospital, Boston. One author declared funding from an Institutional National Research Service Award, the Ryoichi Sasakawa Fellowship Fund, and Massachusetts General Hospital. There were no other conflicts of interest declared.
Although the superiority of metformin as a first-line monotherapy is not news, the study investigators’ choice of treatment intensification as its primary outcome was novel, and highlighted something of greater significance to patients than to physicians.
Patients consider treatment intensification as a result of failure. Reframing the addition of medication as a necessary step for wellness and health maintenance may go a long way toward patient acceptance of intensification as an unfortunate but necessary part of good self-care.
Dr. Jodi B. Segal and Dr. Nisa M. Maruthur are with the department of medicine at Johns Hopkins University, Baltimore. These comments are taken from their editorial accompanying Dr. Berkowitz’s article (JAMA Intern. Med. 2014 [doi: 10.1001/jamainternmed.2014.4296]). They reported having no relevant disclosures.
Although the superiority of metformin as a first-line monotherapy is not news, the study investigators’ choice of treatment intensification as its primary outcome was novel, and highlighted something of greater significance to patients than to physicians.
Patients consider treatment intensification as a result of failure. Reframing the addition of medication as a necessary step for wellness and health maintenance may go a long way toward patient acceptance of intensification as an unfortunate but necessary part of good self-care.
Dr. Jodi B. Segal and Dr. Nisa M. Maruthur are with the department of medicine at Johns Hopkins University, Baltimore. These comments are taken from their editorial accompanying Dr. Berkowitz’s article (JAMA Intern. Med. 2014 [doi: 10.1001/jamainternmed.2014.4296]). They reported having no relevant disclosures.
Although the superiority of metformin as a first-line monotherapy is not news, the study investigators’ choice of treatment intensification as its primary outcome was novel, and highlighted something of greater significance to patients than to physicians.
Patients consider treatment intensification as a result of failure. Reframing the addition of medication as a necessary step for wellness and health maintenance may go a long way toward patient acceptance of intensification as an unfortunate but necessary part of good self-care.
Dr. Jodi B. Segal and Dr. Nisa M. Maruthur are with the department of medicine at Johns Hopkins University, Baltimore. These comments are taken from their editorial accompanying Dr. Berkowitz’s article (JAMA Intern. Med. 2014 [doi: 10.1001/jamainternmed.2014.4296]). They reported having no relevant disclosures.
First-line treatment with metformin for diabetes is associated with significantly less intensification of treatment, and fewer short-term adverse outcomes such as cardiovascular events, emergency department visits, and hypoglycemia, compared with other oral glucose-lowering medications.
A retrospective cohort study of 15,516 patients prescribed an oral glucose-lowering medication found that 25% of patients initially prescribed metformin required a second oral agent, compared with 37% of patients prescribed a sulfonylurea, 40% of those prescribed a thiazolidinedione, and 36% of those prescribed a dipeptidyl peptidase–4 inhibitor, according to a retrospective study published online Oct. 27 (JAMA Intern. Med. 2014 Oct. 27 [doi: 10.1001/jamainternmed.2014.5294]).
While numerous guidelines recommend metformin as the first-line choice of glucose-lowering agents, only 58% of patients started therapy with metformin, with a sulfonylurea the second-most-popular choice, despite its increased risk of adverse cardiovascular events.
“Because underuse of metformin may lead to important harms and costs in the treatment of patients with diabetes, multilevel interventions to increase prescribing quality may be needed,” wrote Dr. Seth A. Berkowitz of Harvard University, Boston, and his colleagues.
The study was supported by an unrestricted grant from CVS Health to Brigham and Women’s Hospital, Boston. One author declared funding from an Institutional National Research Service Award, the Ryoichi Sasakawa Fellowship Fund, and Massachusetts General Hospital. There were no other conflicts of interest declared.
First-line treatment with metformin for diabetes is associated with significantly less intensification of treatment, and fewer short-term adverse outcomes such as cardiovascular events, emergency department visits, and hypoglycemia, compared with other oral glucose-lowering medications.
A retrospective cohort study of 15,516 patients prescribed an oral glucose-lowering medication found that 25% of patients initially prescribed metformin required a second oral agent, compared with 37% of patients prescribed a sulfonylurea, 40% of those prescribed a thiazolidinedione, and 36% of those prescribed a dipeptidyl peptidase–4 inhibitor, according to a retrospective study published online Oct. 27 (JAMA Intern. Med. 2014 Oct. 27 [doi: 10.1001/jamainternmed.2014.5294]).
While numerous guidelines recommend metformin as the first-line choice of glucose-lowering agents, only 58% of patients started therapy with metformin, with a sulfonylurea the second-most-popular choice, despite its increased risk of adverse cardiovascular events.
“Because underuse of metformin may lead to important harms and costs in the treatment of patients with diabetes, multilevel interventions to increase prescribing quality may be needed,” wrote Dr. Seth A. Berkowitz of Harvard University, Boston, and his colleagues.
The study was supported by an unrestricted grant from CVS Health to Brigham and Women’s Hospital, Boston. One author declared funding from an Institutional National Research Service Award, the Ryoichi Sasakawa Fellowship Fund, and Massachusetts General Hospital. There were no other conflicts of interest declared.
FROM JAMA INTERNAL MEDICINE
Key clinical point: Metformin as first-line therapy is associated with lower rates of progression, compared with other oral glucose-lowering medications.
Major finding: Around one-quarter of patients started on metformin progressed to a second agent, compared with 37% of patients prescribed sulfonylurea.
Data source: Retrospective cohort study of 15,516 patients prescribed an oral glucose-lowering agent.
Disclosures: The study was supported by an unrestricted grant from CVS Health to Brigham and Women’s Hospital, Boston. One author declared funding from an Institutional National Research Service Award, the Ryoichi Sasakawa Fellowship Fund, and Massachusetts General Hospital. There were no other conflicts of interest declared.
Questions raised over intrapartum antibiotics to prevent neonatal streptococcus B
The introduction of antibiotic prophylaxis in the Netherlands to prevent invasive neonatal group B streptococcus infection has failed to achieve a decrease in the incidence of infection, prompting calls for guidelines to be reassessed and alternative prevention methods considered.
Analysis of nationwide surveillance data from 1987 to 2011 showed that the incidence of invasive group B streptococcus in infants aged 3 months and under actually increased from 0.20 per 1,000 live births to 0.32 per 1,000 (P < .0001), while the incidence of early-onset disease increased from 0.11 per 1,000 live births to 0.19 per 1,000 (P < .0001).
Researchers also noted an increase in cases of disease caused by clonal complex 17 – which has been associated with invasive disease in neonates – and fewer cases caused by clonal complex 19, as well as a nonsignificant increase in the incidence of invasive Escherichia coli infection after the 1999 implementation of intravenous antibiotic prophylaxis during labor.
“Our findings offer no explanation for the increase in incidence over the past 25 years in Netherlands, [however] possible explanations include changes in the host, medical practice, increased submission of isolates to the National Laboratory, or the pathogen itself,” wrote Dr. Vincent Bekker, from the Emma Children’s Hospital, Academic Medical Center, Amsterdam, and colleagues (Lancet Infect. Dis. 2014 Oct. 19).
In an accompanying editorial, Dr. Shannon D. Manning of Michigan State University, East Lansing, pointed out that the results ran counter to those of other studies, such as one from the United States which showed that intrapartum antibiotic prophylaxis was associated with a 65% reduction in early-onset disease.
“The findings of Bekker and colleagues confirm that group B streptococcus disease in babies remains a global public health concern and show the importance of continuous surveillance in different geographic locations,” Dr. Manning noted.
The study was supported by the National Institute of Public Health and the Environment. There were no other conflicts of interest declared.
The introduction of antibiotic prophylaxis in the Netherlands to prevent invasive neonatal group B streptococcus infection has failed to achieve a decrease in the incidence of infection, prompting calls for guidelines to be reassessed and alternative prevention methods considered.
Analysis of nationwide surveillance data from 1987 to 2011 showed that the incidence of invasive group B streptococcus in infants aged 3 months and under actually increased from 0.20 per 1,000 live births to 0.32 per 1,000 (P < .0001), while the incidence of early-onset disease increased from 0.11 per 1,000 live births to 0.19 per 1,000 (P < .0001).
Researchers also noted an increase in cases of disease caused by clonal complex 17 – which has been associated with invasive disease in neonates – and fewer cases caused by clonal complex 19, as well as a nonsignificant increase in the incidence of invasive Escherichia coli infection after the 1999 implementation of intravenous antibiotic prophylaxis during labor.
“Our findings offer no explanation for the increase in incidence over the past 25 years in Netherlands, [however] possible explanations include changes in the host, medical practice, increased submission of isolates to the National Laboratory, or the pathogen itself,” wrote Dr. Vincent Bekker, from the Emma Children’s Hospital, Academic Medical Center, Amsterdam, and colleagues (Lancet Infect. Dis. 2014 Oct. 19).
In an accompanying editorial, Dr. Shannon D. Manning of Michigan State University, East Lansing, pointed out that the results ran counter to those of other studies, such as one from the United States which showed that intrapartum antibiotic prophylaxis was associated with a 65% reduction in early-onset disease.
“The findings of Bekker and colleagues confirm that group B streptococcus disease in babies remains a global public health concern and show the importance of continuous surveillance in different geographic locations,” Dr. Manning noted.
The study was supported by the National Institute of Public Health and the Environment. There were no other conflicts of interest declared.
The introduction of antibiotic prophylaxis in the Netherlands to prevent invasive neonatal group B streptococcus infection has failed to achieve a decrease in the incidence of infection, prompting calls for guidelines to be reassessed and alternative prevention methods considered.
Analysis of nationwide surveillance data from 1987 to 2011 showed that the incidence of invasive group B streptococcus in infants aged 3 months and under actually increased from 0.20 per 1,000 live births to 0.32 per 1,000 (P < .0001), while the incidence of early-onset disease increased from 0.11 per 1,000 live births to 0.19 per 1,000 (P < .0001).
Researchers also noted an increase in cases of disease caused by clonal complex 17 – which has been associated with invasive disease in neonates – and fewer cases caused by clonal complex 19, as well as a nonsignificant increase in the incidence of invasive Escherichia coli infection after the 1999 implementation of intravenous antibiotic prophylaxis during labor.
“Our findings offer no explanation for the increase in incidence over the past 25 years in Netherlands, [however] possible explanations include changes in the host, medical practice, increased submission of isolates to the National Laboratory, or the pathogen itself,” wrote Dr. Vincent Bekker, from the Emma Children’s Hospital, Academic Medical Center, Amsterdam, and colleagues (Lancet Infect. Dis. 2014 Oct. 19).
In an accompanying editorial, Dr. Shannon D. Manning of Michigan State University, East Lansing, pointed out that the results ran counter to those of other studies, such as one from the United States which showed that intrapartum antibiotic prophylaxis was associated with a 65% reduction in early-onset disease.
“The findings of Bekker and colleagues confirm that group B streptococcus disease in babies remains a global public health concern and show the importance of continuous surveillance in different geographic locations,” Dr. Manning noted.
The study was supported by the National Institute of Public Health and the Environment. There were no other conflicts of interest declared.
FROM THE LANCET INFECTIOUS DISEASES
Key clinical point: Dutch data show an increase in the incidence of invasive neonatal group B streptococcus infection despite intrapartum antibiotic prophylaxis.
Major finding: The incidence of invasive neonatal group B streptococcus infection has increased in the Netherlands since the introduction of intrapartum antibiotic prophylaxis.
Data source: Analysis of population-wide surveillance data.
Disclosures: The study was supported by the National Institute of Public Health and the Environment. There were no other conflicts of interest declared.
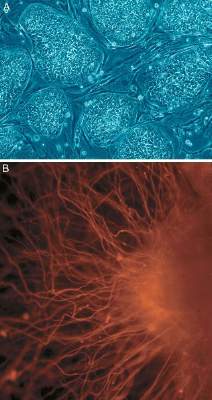
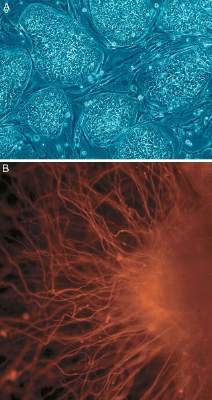
Embryonic Stem Cells Improved Vision in Macular Degeneration
In a first for human embryonic stem cell therapy, use of the pluripotent cells to treat macular dystrophy and macular degeneration significantly improved visual acuity without adverse proliferation, rejection, or serious systemic safety issues for a median of nearly 2 years after transplantation.
“The results of this study provide the first evidence of the medium-term to long-term safety, graft survival, and possible biological activity of pluripotent stem cell progeny in individuals with any disease,” wrote Dr. Steven D. Schwartz, from the Jules Stein Eye Institute Retina Division, Los Angeles, and colleagues.
“Our results suggest that hESC-derived cells could provide a potentially safe new source of cells for the treatment of a variety of unmet medical conditions caused by tissue loss or dysfunction,” the authors noted.
In two prospective phase I/II studies using human embryonic stem cell therapy (hESC)-derived retinal pigment epithelium – one involving nine patients with Stargardt’s macular dystrophy, and the second in nine patients with atrophic age-related macular degeneration – 13 (72%) patients showed an increase in subretinal pigmentation after transplantation of the cells into the subretinal space.
Six months after the transplant, patients with age-related macular degeneration showed improvements in visual acuity by at least 15 letters in four eyes (which the authors described as a clinically significant measure of improvement) and 11-14 letters in two eyes. Visual acuity remained stable (equal to or less than 10 letters) in three eyes, according to a paper published online in the Oct. 15 issue of the Lancet.
In the patients with Stargardt’s macular dystrophy, visual acuity data from eight patients at the 6-month follow up showed improvement by at least 15 letters in three eyes, stable in four eyes, and decreased by 11 letters in one eye (Lancet 2014, Oct. 15 [http://dx.doi.org/10.1016/S0140-6736(14)61376-3]).
Four treated eyes developed visually significant progression of cataracts. One patient with Stargardt’s macular dystrophy developed severe vitreous cavity inflammation consistent with acute postoperative endophthalmitis in one eye within 4 days of surgery, while another eye developed vitreous inflammation. All of those adverse events are associated with vitreoretinal surgery and immunosuppression.
“Additionally, there were no occurrences of adverse proliferation, growth of transplanted tissue at the injection site that was suggestive of a teratoma, ectopic tissue (nonretinal pigment epithelium), or other significant ocular adverse safety issues related to the hESC-retinal pigment epithelium cells in any patient during the observation period,” the researchers wrote.
In the eyes with age-related macular degeneration that did not develop cataracts in the first 6 months of follow-up, researchers noted a median improvement in visual acuity of 16 letters at 6 months, and 14 letters at 12 months, compared with no improvements at 6 months or 12 months in the untreated fellow eyes.
The treatment was delivered to sites with native but compromised retinal pigment epithelium and overlying photoreceptors, with the goal of improving the chances of transplant integration and photoreceptor cell rescue.
Patients from each study were divided into three dose cohorts receiving 50,000, 100,000, and 150,000 cells, and all received immunosuppressive therapy with tacrolimus and mycophenolate mofetil, from 1 week before the surgical procedure until 12 weeks after it.
The work is a “major accomplishment” that has already facilitated other trials of human embryonic stem cell therapy, noted Dr. Anthony Atala of the Wake Forest Institute for Regenerative Medicine, Winston-Salem, N.C., in an accompanying editorial (Lancet 2014, Oct. 15 [http://dx.doi.org/10.1016/S0140-6736(14)61820-1]).
“Now, we have follow-up that extends to longer than 3 years in patients treated with hESC-derived stem cells, showing both safety and apparent efficacy,” he wrote.
Advanced Cell Technology funded the study. Dr. Schwartz has received research support and consulting fees from Alcon, Allergan, Avalanche, Bausch and Lomb, Genentech, and Regeneron. Six of the study authors were employees of Advanced Cell Technology. The editorial author declared no conflicts of interest.
In a first for human embryonic stem cell therapy, use of the pluripotent cells to treat macular dystrophy and macular degeneration significantly improved visual acuity without adverse proliferation, rejection, or serious systemic safety issues for a median of nearly 2 years after transplantation.
“The results of this study provide the first evidence of the medium-term to long-term safety, graft survival, and possible biological activity of pluripotent stem cell progeny in individuals with any disease,” wrote Dr. Steven D. Schwartz, from the Jules Stein Eye Institute Retina Division, Los Angeles, and colleagues.
“Our results suggest that hESC-derived cells could provide a potentially safe new source of cells for the treatment of a variety of unmet medical conditions caused by tissue loss or dysfunction,” the authors noted.
In two prospective phase I/II studies using human embryonic stem cell therapy (hESC)-derived retinal pigment epithelium – one involving nine patients with Stargardt’s macular dystrophy, and the second in nine patients with atrophic age-related macular degeneration – 13 (72%) patients showed an increase in subretinal pigmentation after transplantation of the cells into the subretinal space.
Six months after the transplant, patients with age-related macular degeneration showed improvements in visual acuity by at least 15 letters in four eyes (which the authors described as a clinically significant measure of improvement) and 11-14 letters in two eyes. Visual acuity remained stable (equal to or less than 10 letters) in three eyes, according to a paper published online in the Oct. 15 issue of the Lancet.
In the patients with Stargardt’s macular dystrophy, visual acuity data from eight patients at the 6-month follow up showed improvement by at least 15 letters in three eyes, stable in four eyes, and decreased by 11 letters in one eye (Lancet 2014, Oct. 15 [http://dx.doi.org/10.1016/S0140-6736(14)61376-3]).
Four treated eyes developed visually significant progression of cataracts. One patient with Stargardt’s macular dystrophy developed severe vitreous cavity inflammation consistent with acute postoperative endophthalmitis in one eye within 4 days of surgery, while another eye developed vitreous inflammation. All of those adverse events are associated with vitreoretinal surgery and immunosuppression.
“Additionally, there were no occurrences of adverse proliferation, growth of transplanted tissue at the injection site that was suggestive of a teratoma, ectopic tissue (nonretinal pigment epithelium), or other significant ocular adverse safety issues related to the hESC-retinal pigment epithelium cells in any patient during the observation period,” the researchers wrote.
In the eyes with age-related macular degeneration that did not develop cataracts in the first 6 months of follow-up, researchers noted a median improvement in visual acuity of 16 letters at 6 months, and 14 letters at 12 months, compared with no improvements at 6 months or 12 months in the untreated fellow eyes.
The treatment was delivered to sites with native but compromised retinal pigment epithelium and overlying photoreceptors, with the goal of improving the chances of transplant integration and photoreceptor cell rescue.
Patients from each study were divided into three dose cohorts receiving 50,000, 100,000, and 150,000 cells, and all received immunosuppressive therapy with tacrolimus and mycophenolate mofetil, from 1 week before the surgical procedure until 12 weeks after it.
The work is a “major accomplishment” that has already facilitated other trials of human embryonic stem cell therapy, noted Dr. Anthony Atala of the Wake Forest Institute for Regenerative Medicine, Winston-Salem, N.C., in an accompanying editorial (Lancet 2014, Oct. 15 [http://dx.doi.org/10.1016/S0140-6736(14)61820-1]).
“Now, we have follow-up that extends to longer than 3 years in patients treated with hESC-derived stem cells, showing both safety and apparent efficacy,” he wrote.
Advanced Cell Technology funded the study. Dr. Schwartz has received research support and consulting fees from Alcon, Allergan, Avalanche, Bausch and Lomb, Genentech, and Regeneron. Six of the study authors were employees of Advanced Cell Technology. The editorial author declared no conflicts of interest.
In a first for human embryonic stem cell therapy, use of the pluripotent cells to treat macular dystrophy and macular degeneration significantly improved visual acuity without adverse proliferation, rejection, or serious systemic safety issues for a median of nearly 2 years after transplantation.
“The results of this study provide the first evidence of the medium-term to long-term safety, graft survival, and possible biological activity of pluripotent stem cell progeny in individuals with any disease,” wrote Dr. Steven D. Schwartz, from the Jules Stein Eye Institute Retina Division, Los Angeles, and colleagues.
“Our results suggest that hESC-derived cells could provide a potentially safe new source of cells for the treatment of a variety of unmet medical conditions caused by tissue loss or dysfunction,” the authors noted.
In two prospective phase I/II studies using human embryonic stem cell therapy (hESC)-derived retinal pigment epithelium – one involving nine patients with Stargardt’s macular dystrophy, and the second in nine patients with atrophic age-related macular degeneration – 13 (72%) patients showed an increase in subretinal pigmentation after transplantation of the cells into the subretinal space.
Six months after the transplant, patients with age-related macular degeneration showed improvements in visual acuity by at least 15 letters in four eyes (which the authors described as a clinically significant measure of improvement) and 11-14 letters in two eyes. Visual acuity remained stable (equal to or less than 10 letters) in three eyes, according to a paper published online in the Oct. 15 issue of the Lancet.
In the patients with Stargardt’s macular dystrophy, visual acuity data from eight patients at the 6-month follow up showed improvement by at least 15 letters in three eyes, stable in four eyes, and decreased by 11 letters in one eye (Lancet 2014, Oct. 15 [http://dx.doi.org/10.1016/S0140-6736(14)61376-3]).
Four treated eyes developed visually significant progression of cataracts. One patient with Stargardt’s macular dystrophy developed severe vitreous cavity inflammation consistent with acute postoperative endophthalmitis in one eye within 4 days of surgery, while another eye developed vitreous inflammation. All of those adverse events are associated with vitreoretinal surgery and immunosuppression.
“Additionally, there were no occurrences of adverse proliferation, growth of transplanted tissue at the injection site that was suggestive of a teratoma, ectopic tissue (nonretinal pigment epithelium), or other significant ocular adverse safety issues related to the hESC-retinal pigment epithelium cells in any patient during the observation period,” the researchers wrote.
In the eyes with age-related macular degeneration that did not develop cataracts in the first 6 months of follow-up, researchers noted a median improvement in visual acuity of 16 letters at 6 months, and 14 letters at 12 months, compared with no improvements at 6 months or 12 months in the untreated fellow eyes.
The treatment was delivered to sites with native but compromised retinal pigment epithelium and overlying photoreceptors, with the goal of improving the chances of transplant integration and photoreceptor cell rescue.
Patients from each study were divided into three dose cohorts receiving 50,000, 100,000, and 150,000 cells, and all received immunosuppressive therapy with tacrolimus and mycophenolate mofetil, from 1 week before the surgical procedure until 12 weeks after it.
The work is a “major accomplishment” that has already facilitated other trials of human embryonic stem cell therapy, noted Dr. Anthony Atala of the Wake Forest Institute for Regenerative Medicine, Winston-Salem, N.C., in an accompanying editorial (Lancet 2014, Oct. 15 [http://dx.doi.org/10.1016/S0140-6736(14)61820-1]).
“Now, we have follow-up that extends to longer than 3 years in patients treated with hESC-derived stem cells, showing both safety and apparent efficacy,” he wrote.
Advanced Cell Technology funded the study. Dr. Schwartz has received research support and consulting fees from Alcon, Allergan, Avalanche, Bausch and Lomb, Genentech, and Regeneron. Six of the study authors were employees of Advanced Cell Technology. The editorial author declared no conflicts of interest.
FROM THE LANCET
Embryonic stem cells improved vision in macular degeneration
In a first for human embryonic stem cell therapy, use of the pluripotent cells to treat macular dystrophy and macular degeneration significantly improved visual acuity without adverse proliferation, rejection, or serious systemic safety issues for a median of nearly 2 years after transplantation.
“The results of this study provide the first evidence of the medium-term to long-term safety, graft survival, and possible biological activity of pluripotent stem cell progeny in individuals with any disease,” wrote Dr. Steven D. Schwartz, from the Jules Stein Eye Institute Retina Division, Los Angeles, and colleagues.
“Our results suggest that hESC-derived cells could provide a potentially safe new source of cells for the treatment of a variety of unmet medical conditions caused by tissue loss or dysfunction,” the authors noted.
In two prospective phase I/II studies using human embryonic stem cell therapy (hESC)-derived retinal pigment epithelium – one involving nine patients with Stargardt’s macular dystrophy, and the second in nine patients with atrophic age-related macular degeneration – 13 (72%) patients showed an increase in subretinal pigmentation after transplantation of the cells into the subretinal space.
Six months after the transplant, patients with age-related macular degeneration showed improvements in visual acuity by at least 15 letters in four eyes (which the authors described as a clinically significant measure of improvement) and 11-14 letters in two eyes. Visual acuity remained stable (equal to or less than 10 letters) in three eyes, according to a paper published online in the Oct. 15 issue of the Lancet.
In the patients with Stargardt’s macular dystrophy, visual acuity data from eight patients at the 6-month follow up showed improvement by at least 15 letters in three eyes, stable in four eyes, and decreased by 11 letters in one eye (Lancet 2014, Oct. 15 [http://dx.doi.org/10.1016/S0140-6736(14)61376-3]).
Four treated eyes developed visually significant progression of cataracts. One patient with Stargardt’s macular dystrophy developed severe vitreous cavity inflammation consistent with acute postoperative endophthalmitis in one eye within 4 days of surgery, while another eye developed vitreous inflammation. All of those adverse events are associated with vitreoretinal surgery and immunosuppression.
“Additionally, there were no occurrences of adverse proliferation, growth of transplanted tissue at the injection site that was suggestive of a teratoma, ectopic tissue (nonretinal pigment epithelium), or other significant ocular adverse safety issues related to the hESC-retinal pigment epithelium cells in any patient during the observation period,” the researchers wrote.
In the eyes with age-related macular degeneration that did not develop cataracts in the first 6 months of follow-up, researchers noted a median improvement in visual acuity of 16 letters at 6 months, and 14 letters at 12 months, compared with no improvements at 6 months or 12 months in the untreated fellow eyes.
The treatment was delivered to sites with native but compromised retinal pigment epithelium and overlying photoreceptors, with the goal of improving the chances of transplant integration and photoreceptor cell rescue.
Patients from each study were divided into three dose cohorts receiving 50,000, 100,000, and 150,000 cells, and all received immunosuppressive therapy with tacrolimus and mycophenolate mofetil, from 1 week before the surgical procedure until 12 weeks after it.
The work is a “major accomplishment” that has already facilitated other trials of human embryonic stem cell therapy, noted Dr. Anthony Atala of the Wake Forest Institute for Regenerative Medicine, Winston-Salem, N.C., in an accompanying editorial (Lancet 2014, Oct. 15 [http://dx.doi.org/10.1016/S0140-6736(14)61820-1]).
“Now, we have follow-up that extends to longer than 3 years in patients treated with hESC-derived stem cells, showing both safety and apparent efficacy,” he wrote.
Advanced Cell Technology funded the study. Dr. Schwartz has received research support and consulting fees from Alcon, Allergan, Avalanche, Bausch and Lomb, Genentech, and Regeneron. Six of the study authors were employees of Advanced Cell Technology. The editorial author declared no conflicts of interest.
In a first for human embryonic stem cell therapy, use of the pluripotent cells to treat macular dystrophy and macular degeneration significantly improved visual acuity without adverse proliferation, rejection, or serious systemic safety issues for a median of nearly 2 years after transplantation.
“The results of this study provide the first evidence of the medium-term to long-term safety, graft survival, and possible biological activity of pluripotent stem cell progeny in individuals with any disease,” wrote Dr. Steven D. Schwartz, from the Jules Stein Eye Institute Retina Division, Los Angeles, and colleagues.
“Our results suggest that hESC-derived cells could provide a potentially safe new source of cells for the treatment of a variety of unmet medical conditions caused by tissue loss or dysfunction,” the authors noted.
In two prospective phase I/II studies using human embryonic stem cell therapy (hESC)-derived retinal pigment epithelium – one involving nine patients with Stargardt’s macular dystrophy, and the second in nine patients with atrophic age-related macular degeneration – 13 (72%) patients showed an increase in subretinal pigmentation after transplantation of the cells into the subretinal space.
Six months after the transplant, patients with age-related macular degeneration showed improvements in visual acuity by at least 15 letters in four eyes (which the authors described as a clinically significant measure of improvement) and 11-14 letters in two eyes. Visual acuity remained stable (equal to or less than 10 letters) in three eyes, according to a paper published online in the Oct. 15 issue of the Lancet.
In the patients with Stargardt’s macular dystrophy, visual acuity data from eight patients at the 6-month follow up showed improvement by at least 15 letters in three eyes, stable in four eyes, and decreased by 11 letters in one eye (Lancet 2014, Oct. 15 [http://dx.doi.org/10.1016/S0140-6736(14)61376-3]).
Four treated eyes developed visually significant progression of cataracts. One patient with Stargardt’s macular dystrophy developed severe vitreous cavity inflammation consistent with acute postoperative endophthalmitis in one eye within 4 days of surgery, while another eye developed vitreous inflammation. All of those adverse events are associated with vitreoretinal surgery and immunosuppression.
“Additionally, there were no occurrences of adverse proliferation, growth of transplanted tissue at the injection site that was suggestive of a teratoma, ectopic tissue (nonretinal pigment epithelium), or other significant ocular adverse safety issues related to the hESC-retinal pigment epithelium cells in any patient during the observation period,” the researchers wrote.
In the eyes with age-related macular degeneration that did not develop cataracts in the first 6 months of follow-up, researchers noted a median improvement in visual acuity of 16 letters at 6 months, and 14 letters at 12 months, compared with no improvements at 6 months or 12 months in the untreated fellow eyes.
The treatment was delivered to sites with native but compromised retinal pigment epithelium and overlying photoreceptors, with the goal of improving the chances of transplant integration and photoreceptor cell rescue.
Patients from each study were divided into three dose cohorts receiving 50,000, 100,000, and 150,000 cells, and all received immunosuppressive therapy with tacrolimus and mycophenolate mofetil, from 1 week before the surgical procedure until 12 weeks after it.
The work is a “major accomplishment” that has already facilitated other trials of human embryonic stem cell therapy, noted Dr. Anthony Atala of the Wake Forest Institute for Regenerative Medicine, Winston-Salem, N.C., in an accompanying editorial (Lancet 2014, Oct. 15 [http://dx.doi.org/10.1016/S0140-6736(14)61820-1]).
“Now, we have follow-up that extends to longer than 3 years in patients treated with hESC-derived stem cells, showing both safety and apparent efficacy,” he wrote.
Advanced Cell Technology funded the study. Dr. Schwartz has received research support and consulting fees from Alcon, Allergan, Avalanche, Bausch and Lomb, Genentech, and Regeneron. Six of the study authors were employees of Advanced Cell Technology. The editorial author declared no conflicts of interest.
In a first for human embryonic stem cell therapy, use of the pluripotent cells to treat macular dystrophy and macular degeneration significantly improved visual acuity without adverse proliferation, rejection, or serious systemic safety issues for a median of nearly 2 years after transplantation.
“The results of this study provide the first evidence of the medium-term to long-term safety, graft survival, and possible biological activity of pluripotent stem cell progeny in individuals with any disease,” wrote Dr. Steven D. Schwartz, from the Jules Stein Eye Institute Retina Division, Los Angeles, and colleagues.
“Our results suggest that hESC-derived cells could provide a potentially safe new source of cells for the treatment of a variety of unmet medical conditions caused by tissue loss or dysfunction,” the authors noted.
In two prospective phase I/II studies using human embryonic stem cell therapy (hESC)-derived retinal pigment epithelium – one involving nine patients with Stargardt’s macular dystrophy, and the second in nine patients with atrophic age-related macular degeneration – 13 (72%) patients showed an increase in subretinal pigmentation after transplantation of the cells into the subretinal space.
Six months after the transplant, patients with age-related macular degeneration showed improvements in visual acuity by at least 15 letters in four eyes (which the authors described as a clinically significant measure of improvement) and 11-14 letters in two eyes. Visual acuity remained stable (equal to or less than 10 letters) in three eyes, according to a paper published online in the Oct. 15 issue of the Lancet.
In the patients with Stargardt’s macular dystrophy, visual acuity data from eight patients at the 6-month follow up showed improvement by at least 15 letters in three eyes, stable in four eyes, and decreased by 11 letters in one eye (Lancet 2014, Oct. 15 [http://dx.doi.org/10.1016/S0140-6736(14)61376-3]).
Four treated eyes developed visually significant progression of cataracts. One patient with Stargardt’s macular dystrophy developed severe vitreous cavity inflammation consistent with acute postoperative endophthalmitis in one eye within 4 days of surgery, while another eye developed vitreous inflammation. All of those adverse events are associated with vitreoretinal surgery and immunosuppression.
“Additionally, there were no occurrences of adverse proliferation, growth of transplanted tissue at the injection site that was suggestive of a teratoma, ectopic tissue (nonretinal pigment epithelium), or other significant ocular adverse safety issues related to the hESC-retinal pigment epithelium cells in any patient during the observation period,” the researchers wrote.
In the eyes with age-related macular degeneration that did not develop cataracts in the first 6 months of follow-up, researchers noted a median improvement in visual acuity of 16 letters at 6 months, and 14 letters at 12 months, compared with no improvements at 6 months or 12 months in the untreated fellow eyes.
The treatment was delivered to sites with native but compromised retinal pigment epithelium and overlying photoreceptors, with the goal of improving the chances of transplant integration and photoreceptor cell rescue.
Patients from each study were divided into three dose cohorts receiving 50,000, 100,000, and 150,000 cells, and all received immunosuppressive therapy with tacrolimus and mycophenolate mofetil, from 1 week before the surgical procedure until 12 weeks after it.
The work is a “major accomplishment” that has already facilitated other trials of human embryonic stem cell therapy, noted Dr. Anthony Atala of the Wake Forest Institute for Regenerative Medicine, Winston-Salem, N.C., in an accompanying editorial (Lancet 2014, Oct. 15 [http://dx.doi.org/10.1016/S0140-6736(14)61820-1]).
“Now, we have follow-up that extends to longer than 3 years in patients treated with hESC-derived stem cells, showing both safety and apparent efficacy,” he wrote.
Advanced Cell Technology funded the study. Dr. Schwartz has received research support and consulting fees from Alcon, Allergan, Avalanche, Bausch and Lomb, Genentech, and Regeneron. Six of the study authors were employees of Advanced Cell Technology. The editorial author declared no conflicts of interest.
FROM THE LANCET
Key clinical point: Human embryonic stem cell transplants achieved clinically significant improvements in visual acuity.
Major finding: Nearly three-quarters of patients with macular degeneration or dystrophy showed increases in subretinal pigmentation.
Data source: Two prospective phase I/II trials of human embryonic stem cell therapy (hESC)–derived retinal pigment epithelium in nine patients with atrophic age-related macular degeneration and nine with Stargardt’s macular dystrophy.
Disclosures: Advanced Cell Technology funded the study. Dr. Schwartz has received research support and consulting fees from Alcon, Allergan, Avalanche, Bausch and Lomb, Genentech, and Regeneron. Six of the study authors were employees of Advanced Cell Technology. The editorial author declared no conflicts of interest.
Inflammatory rheumatic diseases raise venous thromboembolism risk
Individuals with inflammatory rheumatic diseases such as inflammatory arthritis, vasculitis, and connective tissue diseases, have a threefold increase in the risk of venous thromboembolism, compared with the general population, according to a meta-analysis.
The meta-analysis of 25 studies – 10 of which included patients with rheumatoid arthritis (RA) – found those with RA were more than twice as likely to develop deep vein thrombosis or a pulmonary embolism, compared with an age- and sex-matched individuals who had other comorbidities such as diabetes, peripheral vascular disease/coronary artery disease, and malignancy (OR, 2.23; 95% confidence interval, 2.02-2.47). The RA patients had a cumulative venous thromboembolism (VTE) incidence of 2.18% (Arthritis Res. Ther. 2014;16:435 [doi:10.1186/s13075-014-0435-y]).
Ten studies comprising 54,697 patients with systemic lupus erythematosus showed a cumulative thrombosis incidence of 7.29% (95% CI, 5.82%-8.75%). Other diseases for which the investigators calculated cumulative incidence rates of VTE, based on four studies apiece, were Sjögren’s syndrome (2.18%; 95% CI, 0.79%-3.57%), inflammatory myositis (4.03%; 95% CI, 2.38%-5.67%), Antineutrophil cytoplasmic antibody vasculitis (7.97%; 95% CI, 5.67%-10.28%), and systemic sclerosis (3.13%; 95% CI, 1.73%-4.52%).
“We believe that the increased VTE risk is associated with the activity of the inflammatory diseases, rather than with the treatments used for controlling the disease,” wrote Dr. Jason Lee of the University of Western Ontario, London, and Dr. Janet Pope, of the division of rheumatology at St. Joseph’s Health Care, London, Ont.
The authors said that they had no conflicts of interest.
Individuals with inflammatory rheumatic diseases such as inflammatory arthritis, vasculitis, and connective tissue diseases, have a threefold increase in the risk of venous thromboembolism, compared with the general population, according to a meta-analysis.
The meta-analysis of 25 studies – 10 of which included patients with rheumatoid arthritis (RA) – found those with RA were more than twice as likely to develop deep vein thrombosis or a pulmonary embolism, compared with an age- and sex-matched individuals who had other comorbidities such as diabetes, peripheral vascular disease/coronary artery disease, and malignancy (OR, 2.23; 95% confidence interval, 2.02-2.47). The RA patients had a cumulative venous thromboembolism (VTE) incidence of 2.18% (Arthritis Res. Ther. 2014;16:435 [doi:10.1186/s13075-014-0435-y]).
Ten studies comprising 54,697 patients with systemic lupus erythematosus showed a cumulative thrombosis incidence of 7.29% (95% CI, 5.82%-8.75%). Other diseases for which the investigators calculated cumulative incidence rates of VTE, based on four studies apiece, were Sjögren’s syndrome (2.18%; 95% CI, 0.79%-3.57%), inflammatory myositis (4.03%; 95% CI, 2.38%-5.67%), Antineutrophil cytoplasmic antibody vasculitis (7.97%; 95% CI, 5.67%-10.28%), and systemic sclerosis (3.13%; 95% CI, 1.73%-4.52%).
“We believe that the increased VTE risk is associated with the activity of the inflammatory diseases, rather than with the treatments used for controlling the disease,” wrote Dr. Jason Lee of the University of Western Ontario, London, and Dr. Janet Pope, of the division of rheumatology at St. Joseph’s Health Care, London, Ont.
The authors said that they had no conflicts of interest.
Individuals with inflammatory rheumatic diseases such as inflammatory arthritis, vasculitis, and connective tissue diseases, have a threefold increase in the risk of venous thromboembolism, compared with the general population, according to a meta-analysis.
The meta-analysis of 25 studies – 10 of which included patients with rheumatoid arthritis (RA) – found those with RA were more than twice as likely to develop deep vein thrombosis or a pulmonary embolism, compared with an age- and sex-matched individuals who had other comorbidities such as diabetes, peripheral vascular disease/coronary artery disease, and malignancy (OR, 2.23; 95% confidence interval, 2.02-2.47). The RA patients had a cumulative venous thromboembolism (VTE) incidence of 2.18% (Arthritis Res. Ther. 2014;16:435 [doi:10.1186/s13075-014-0435-y]).
Ten studies comprising 54,697 patients with systemic lupus erythematosus showed a cumulative thrombosis incidence of 7.29% (95% CI, 5.82%-8.75%). Other diseases for which the investigators calculated cumulative incidence rates of VTE, based on four studies apiece, were Sjögren’s syndrome (2.18%; 95% CI, 0.79%-3.57%), inflammatory myositis (4.03%; 95% CI, 2.38%-5.67%), Antineutrophil cytoplasmic antibody vasculitis (7.97%; 95% CI, 5.67%-10.28%), and systemic sclerosis (3.13%; 95% CI, 1.73%-4.52%).
“We believe that the increased VTE risk is associated with the activity of the inflammatory diseases, rather than with the treatments used for controlling the disease,” wrote Dr. Jason Lee of the University of Western Ontario, London, and Dr. Janet Pope, of the division of rheumatology at St. Joseph’s Health Care, London, Ont.
The authors said that they had no conflicts of interest.
FROM ARTHRITIS RESEARCH & THERAPY
Key clinical point: There is strong evidence for an elevated baseline risk of VTE in patients with inflammatory rheumatic diseases.
Major finding: Patients with rheumatoid arthritis are more than twice as likely to develop deep vein thrombosis or a pulmonary embolism, compared with an age- and sex-matched patients.
Data source: Meta-analysis of 25 studies.
Disclosures: No conflicts of interest were declared.
Combination therapy best in RA regardless of prognosis
Rheumatoid arthritis patients with differing prognoses achieved similar improvements in clinical outcomes and functional improvements with initial combination therapy but fared differently when given initial monotherapy, according to the results of a post-hoc analysis of a randomized controlled trial.
The results challenge a recent push to tailor treatment strategy to risk stratification, argued Dr. Iris Markusse of the department of rheumatology at Leiden (the Netherlands) University Medical Center and her coauthors.
The researchers examined the treatment outcomes of 508 patients with rheumatoid arthritis in a post-hoc analysis of the BEST study in which patients were randomized to one of four treatment groups: sequential monotherapy starting with methotrexate, step-up combination therapy, initial combination therapy with prednisone, or initial combination with infliximab. For this post-hoc analysis of 1-year results, the investigators combined the first two groups that both started with methotrexate monotherapy because they had very similar disease outcomes during the first year of follow-up. The third and fourth groups that received initial combination therapy were combined as well.
The results were analyzed according to whether the patients were classified by disease characteristics and radiographic progression as having a poor or nonpoor prognosis. The researchers defined poor prognosis – 417 of the 508 patients – by the presence of at least three of four poor prognostic factors: Disease Activity Score of 3.7 or greater, swollen joint count of 10 or higher, four or more erosions, and positive results for both rheumatoid factor and anti-citrullinated peptide autoantibodies.
They found little difference between the two prognostic groups, with both showing significantly higher rates of remission and functional improvements with combination therapy than monotherapy, and the differences were preserved at 1 year. Among poor-prognosis patients, 93% achieved an American College of Rheumatology 20 (ACR 20) response at 1 year, compared with 80% of those on initial monotherapy. In the non–poor prognosis patients, initial combination therapy proved significantly better than initial monotherapy at achieving ACR20 response (85% vs. 72%).
Both poor- and non–poor prognosis groups had similar improvements in Health Assessment Questionnaire scores, and there was no significant differences in the toxicity of the treatment approaches in the different prognosis groups based on the numbers of reported adverse events (Arthritis Res. Ther. 2014 Sept. 25 [doi:10.1186/s13075-014-0430-3]).
“These results suggest that prognostic factors associated with future radiographic damage progression contribute little to predict early clinical response to initial treatment, and therefore in our opinion tailored treatment based on prognosis as suggested by the EULAR [European League Against Rheumatism] guidelines is currently not feasible,” Dr. Markusse and her associates wrote.
The study was supported by grants from the Dutch Insurance Companies, Schering-Plough, and Janssen. The authors declared having no other conflicts of interest.
Rheumatoid arthritis patients with differing prognoses achieved similar improvements in clinical outcomes and functional improvements with initial combination therapy but fared differently when given initial monotherapy, according to the results of a post-hoc analysis of a randomized controlled trial.
The results challenge a recent push to tailor treatment strategy to risk stratification, argued Dr. Iris Markusse of the department of rheumatology at Leiden (the Netherlands) University Medical Center and her coauthors.
The researchers examined the treatment outcomes of 508 patients with rheumatoid arthritis in a post-hoc analysis of the BEST study in which patients were randomized to one of four treatment groups: sequential monotherapy starting with methotrexate, step-up combination therapy, initial combination therapy with prednisone, or initial combination with infliximab. For this post-hoc analysis of 1-year results, the investigators combined the first two groups that both started with methotrexate monotherapy because they had very similar disease outcomes during the first year of follow-up. The third and fourth groups that received initial combination therapy were combined as well.
The results were analyzed according to whether the patients were classified by disease characteristics and radiographic progression as having a poor or nonpoor prognosis. The researchers defined poor prognosis – 417 of the 508 patients – by the presence of at least three of four poor prognostic factors: Disease Activity Score of 3.7 or greater, swollen joint count of 10 or higher, four or more erosions, and positive results for both rheumatoid factor and anti-citrullinated peptide autoantibodies.
They found little difference between the two prognostic groups, with both showing significantly higher rates of remission and functional improvements with combination therapy than monotherapy, and the differences were preserved at 1 year. Among poor-prognosis patients, 93% achieved an American College of Rheumatology 20 (ACR 20) response at 1 year, compared with 80% of those on initial monotherapy. In the non–poor prognosis patients, initial combination therapy proved significantly better than initial monotherapy at achieving ACR20 response (85% vs. 72%).
Both poor- and non–poor prognosis groups had similar improvements in Health Assessment Questionnaire scores, and there was no significant differences in the toxicity of the treatment approaches in the different prognosis groups based on the numbers of reported adverse events (Arthritis Res. Ther. 2014 Sept. 25 [doi:10.1186/s13075-014-0430-3]).
“These results suggest that prognostic factors associated with future radiographic damage progression contribute little to predict early clinical response to initial treatment, and therefore in our opinion tailored treatment based on prognosis as suggested by the EULAR [European League Against Rheumatism] guidelines is currently not feasible,” Dr. Markusse and her associates wrote.
The study was supported by grants from the Dutch Insurance Companies, Schering-Plough, and Janssen. The authors declared having no other conflicts of interest.
Rheumatoid arthritis patients with differing prognoses achieved similar improvements in clinical outcomes and functional improvements with initial combination therapy but fared differently when given initial monotherapy, according to the results of a post-hoc analysis of a randomized controlled trial.
The results challenge a recent push to tailor treatment strategy to risk stratification, argued Dr. Iris Markusse of the department of rheumatology at Leiden (the Netherlands) University Medical Center and her coauthors.
The researchers examined the treatment outcomes of 508 patients with rheumatoid arthritis in a post-hoc analysis of the BEST study in which patients were randomized to one of four treatment groups: sequential monotherapy starting with methotrexate, step-up combination therapy, initial combination therapy with prednisone, or initial combination with infliximab. For this post-hoc analysis of 1-year results, the investigators combined the first two groups that both started with methotrexate monotherapy because they had very similar disease outcomes during the first year of follow-up. The third and fourth groups that received initial combination therapy were combined as well.
The results were analyzed according to whether the patients were classified by disease characteristics and radiographic progression as having a poor or nonpoor prognosis. The researchers defined poor prognosis – 417 of the 508 patients – by the presence of at least three of four poor prognostic factors: Disease Activity Score of 3.7 or greater, swollen joint count of 10 or higher, four or more erosions, and positive results for both rheumatoid factor and anti-citrullinated peptide autoantibodies.
They found little difference between the two prognostic groups, with both showing significantly higher rates of remission and functional improvements with combination therapy than monotherapy, and the differences were preserved at 1 year. Among poor-prognosis patients, 93% achieved an American College of Rheumatology 20 (ACR 20) response at 1 year, compared with 80% of those on initial monotherapy. In the non–poor prognosis patients, initial combination therapy proved significantly better than initial monotherapy at achieving ACR20 response (85% vs. 72%).
Both poor- and non–poor prognosis groups had similar improvements in Health Assessment Questionnaire scores, and there was no significant differences in the toxicity of the treatment approaches in the different prognosis groups based on the numbers of reported adverse events (Arthritis Res. Ther. 2014 Sept. 25 [doi:10.1186/s13075-014-0430-3]).
“These results suggest that prognostic factors associated with future radiographic damage progression contribute little to predict early clinical response to initial treatment, and therefore in our opinion tailored treatment based on prognosis as suggested by the EULAR [European League Against Rheumatism] guidelines is currently not feasible,” Dr. Markusse and her associates wrote.
The study was supported by grants from the Dutch Insurance Companies, Schering-Plough, and Janssen. The authors declared having no other conflicts of interest.
FROM ARTHRITIS RESEARCH & THERAPY
Key clinical point: Rheumatoid arthritis patients achieve greater benefits from initial combination therapy, regardless of their disease prognosis.
Major finding: Among poor-prognosis patients, 93% achieved an ACR 20 response at 1 year, compared with 80% of those on initial monotherapy, and more non–poor prognosis patients on initial combination treatment also achieved an ACR20 response (85% vs. 72%).
Data source: A post-hoc analysis of the randomized controlled BEST trial in 508 patients with rheumatoid arthritis.
Disclosures: The study was supported by grants from the Dutch Insurance Companies, Schering-Plough, and Janssen. The authors declared having no other conflicts of interest.
Schizophrenia linked with dyslipidemia, hypertension, and smoking
First-episode schizophrenia spectrum disorders are associated with a significantly higher prevalence of metabolic risk factors, such as smoking, dyslipidemia, and elevated body mass, compared with the general population. Those risk factors are tied to illness duration and are exacerbated by antipsychotic therapy, data from a cross-sectional study of patients with the disorders show.
Among the 394 patients with cardiometabolic data, about half smoked cigarettes, 39.9% had prehypertension, 10.0% had hypertension, and 13.2% had metabolic syndrome, and 48.3% were obese or overweight – a figure similar to that found among a similarly aged U.S. population – while 56.5% had dyslipidemia, which reflects the incidence in a general population 15-20 years older.
Duration of psychiatric illness was significantly associated with higher body mass index, fat mass, fat percentage, and waist circumference. Antipsychotic treatment was linked to higher measures of non-HDL-C, triglyceride levels, and triglyceride to HDL-C ratios but lower HDL-C measures and systolic blood pressure, according to a paper published online Oct. 8 in JAMA Psychiatry ([doi:10.1001/jamapsychiatry.2014.1314]).
“In patients with [first-episode schizophrenia], cardiometabolic risk factors and abnormalities are present early in the illness and likely related to the underlying illness, unhealthy lifestyle, and antipsychotic medications, which interact with each other,” wrote Dr. Christoph U. Correll, professor, psychiatry and molecular medicine at the Hofstra University, Hempstead, N.Y., and his colleagues. “Prevention of and early interventions for psychiatric illness with lower-risk agents, routine antipsychotic adverse effect monitoring, and smoking cessation interventions are needed from the earliest illness phases.”
The study was supported by the National Institute of Mental Health, and the American Recovery and Reinvestment Act. Dr. Correll disclosed acting as a consultant and/or adviser to or receiving honoraria from several companies and entities, including numerous makers of antipsychotics.
First-episode schizophrenia spectrum disorders are associated with a significantly higher prevalence of metabolic risk factors, such as smoking, dyslipidemia, and elevated body mass, compared with the general population. Those risk factors are tied to illness duration and are exacerbated by antipsychotic therapy, data from a cross-sectional study of patients with the disorders show.
Among the 394 patients with cardiometabolic data, about half smoked cigarettes, 39.9% had prehypertension, 10.0% had hypertension, and 13.2% had metabolic syndrome, and 48.3% were obese or overweight – a figure similar to that found among a similarly aged U.S. population – while 56.5% had dyslipidemia, which reflects the incidence in a general population 15-20 years older.
Duration of psychiatric illness was significantly associated with higher body mass index, fat mass, fat percentage, and waist circumference. Antipsychotic treatment was linked to higher measures of non-HDL-C, triglyceride levels, and triglyceride to HDL-C ratios but lower HDL-C measures and systolic blood pressure, according to a paper published online Oct. 8 in JAMA Psychiatry ([doi:10.1001/jamapsychiatry.2014.1314]).
“In patients with [first-episode schizophrenia], cardiometabolic risk factors and abnormalities are present early in the illness and likely related to the underlying illness, unhealthy lifestyle, and antipsychotic medications, which interact with each other,” wrote Dr. Christoph U. Correll, professor, psychiatry and molecular medicine at the Hofstra University, Hempstead, N.Y., and his colleagues. “Prevention of and early interventions for psychiatric illness with lower-risk agents, routine antipsychotic adverse effect monitoring, and smoking cessation interventions are needed from the earliest illness phases.”
The study was supported by the National Institute of Mental Health, and the American Recovery and Reinvestment Act. Dr. Correll disclosed acting as a consultant and/or adviser to or receiving honoraria from several companies and entities, including numerous makers of antipsychotics.
First-episode schizophrenia spectrum disorders are associated with a significantly higher prevalence of metabolic risk factors, such as smoking, dyslipidemia, and elevated body mass, compared with the general population. Those risk factors are tied to illness duration and are exacerbated by antipsychotic therapy, data from a cross-sectional study of patients with the disorders show.
Among the 394 patients with cardiometabolic data, about half smoked cigarettes, 39.9% had prehypertension, 10.0% had hypertension, and 13.2% had metabolic syndrome, and 48.3% were obese or overweight – a figure similar to that found among a similarly aged U.S. population – while 56.5% had dyslipidemia, which reflects the incidence in a general population 15-20 years older.
Duration of psychiatric illness was significantly associated with higher body mass index, fat mass, fat percentage, and waist circumference. Antipsychotic treatment was linked to higher measures of non-HDL-C, triglyceride levels, and triglyceride to HDL-C ratios but lower HDL-C measures and systolic blood pressure, according to a paper published online Oct. 8 in JAMA Psychiatry ([doi:10.1001/jamapsychiatry.2014.1314]).
“In patients with [first-episode schizophrenia], cardiometabolic risk factors and abnormalities are present early in the illness and likely related to the underlying illness, unhealthy lifestyle, and antipsychotic medications, which interact with each other,” wrote Dr. Christoph U. Correll, professor, psychiatry and molecular medicine at the Hofstra University, Hempstead, N.Y., and his colleagues. “Prevention of and early interventions for psychiatric illness with lower-risk agents, routine antipsychotic adverse effect monitoring, and smoking cessation interventions are needed from the earliest illness phases.”
The study was supported by the National Institute of Mental Health, and the American Recovery and Reinvestment Act. Dr. Correll disclosed acting as a consultant and/or adviser to or receiving honoraria from several companies and entities, including numerous makers of antipsychotics.
FROM JAMA PSYCHIATRY
Key clinical point: Schizophrenia diagnosis and treatment are associated with elevated metabolic risk factors.
Major finding: More than half of patients with first-episode schizophrenia have dyslipidemia.
Data source: Cross-sectional study of 404 patients with first-episode schizophrenia spectrum disorder.
Disclosures: The study was supported by the National Institute of Mental Health, and the American Recovery and Reinvestment Act. Dr. Correll disclosed acting as a consultant and/or adviser to or receiving honoraria from several companies and entities, including numerous makers of antipsychotics.
Position statement on obesity and cancer issued by the American Society of Clinical Oncology
Oncologists are increasingly being called on to address obesity in cancer patients as the rates of obesity are increasing and more cancer patients are becoming cancer survivors, according to a position statement on obesity and cancer from the American Society of Clinical Oncology.
Obesity appears to have negative downstream effects for cancer risk, recurrence, and mortality. As many as 84,000 cancer diagnoses each year are attributed to obesity, and overweight and obesity are implicated in 15%-20% of total cancer-related mortality,” wrote Dr. Jennifer A. Ligibel from the Dana-Farber Cancer Institute, Boston, and her colleagues in the statement, which calls for increased education, clinical guidance, research, and advocacy on the issue.
Evidence suggests that obese individuals have a significantly worse prognosis than do nonobese patients. One meta-analysis found a 75% increase in mortality in premenopausal women who were obese at the time of breast cancer diagnosis, compared with normal-weight patients. Further, obesity can interfere with the delivery of cancer therapy and is associated with increases in the risk of second primary malignancies, according to the paper published Oct.1 in the Journal of Clinical Oncology.
Dr. Ligibel and her colleagues said many individuals initiate positive health behavior changes after a cancer diagnosis, and such a point can therefore serve as a “teachable moment” that may offer the chance to motivate individuals to implement lifestyle and behavioral changes.
The statement advised assessing patients’ weight by calculating BMI and referring patients to appropriate services such as oncology nutrition, rehabilitation medicine, and exercise physiology. Oncologists also are encouraged to lead by example and to share their personal experiences at attempts to lose weight and to increase physical activity. Participating in community events focused on physical activity and healthy living “can also reinforce the importance of health lifestyle behaviors as part of a a comprehensive cancer-control strategy,” the statement said.
ASCO also announced that it will host a summit to assemble groups needed to conduct lifestyle intervention research in cancer survivors and urged studies of lifestyle changes on outcomes in patients with early-stage cancers.
Additionally, ASCO has developed a toolkit to assist in addressing weight management, including strategies for improving nutrition and physical activity as well as insurance coverage for weight-management services.
The authors declared no conflicts of interest.
Oncologists are increasingly being called on to address obesity in cancer patients as the rates of obesity are increasing and more cancer patients are becoming cancer survivors, according to a position statement on obesity and cancer from the American Society of Clinical Oncology.
Obesity appears to have negative downstream effects for cancer risk, recurrence, and mortality. As many as 84,000 cancer diagnoses each year are attributed to obesity, and overweight and obesity are implicated in 15%-20% of total cancer-related mortality,” wrote Dr. Jennifer A. Ligibel from the Dana-Farber Cancer Institute, Boston, and her colleagues in the statement, which calls for increased education, clinical guidance, research, and advocacy on the issue.
Evidence suggests that obese individuals have a significantly worse prognosis than do nonobese patients. One meta-analysis found a 75% increase in mortality in premenopausal women who were obese at the time of breast cancer diagnosis, compared with normal-weight patients. Further, obesity can interfere with the delivery of cancer therapy and is associated with increases in the risk of second primary malignancies, according to the paper published Oct.1 in the Journal of Clinical Oncology.
Dr. Ligibel and her colleagues said many individuals initiate positive health behavior changes after a cancer diagnosis, and such a point can therefore serve as a “teachable moment” that may offer the chance to motivate individuals to implement lifestyle and behavioral changes.
The statement advised assessing patients’ weight by calculating BMI and referring patients to appropriate services such as oncology nutrition, rehabilitation medicine, and exercise physiology. Oncologists also are encouraged to lead by example and to share their personal experiences at attempts to lose weight and to increase physical activity. Participating in community events focused on physical activity and healthy living “can also reinforce the importance of health lifestyle behaviors as part of a a comprehensive cancer-control strategy,” the statement said.
ASCO also announced that it will host a summit to assemble groups needed to conduct lifestyle intervention research in cancer survivors and urged studies of lifestyle changes on outcomes in patients with early-stage cancers.
Additionally, ASCO has developed a toolkit to assist in addressing weight management, including strategies for improving nutrition and physical activity as well as insurance coverage for weight-management services.
The authors declared no conflicts of interest.
Oncologists are increasingly being called on to address obesity in cancer patients as the rates of obesity are increasing and more cancer patients are becoming cancer survivors, according to a position statement on obesity and cancer from the American Society of Clinical Oncology.
Obesity appears to have negative downstream effects for cancer risk, recurrence, and mortality. As many as 84,000 cancer diagnoses each year are attributed to obesity, and overweight and obesity are implicated in 15%-20% of total cancer-related mortality,” wrote Dr. Jennifer A. Ligibel from the Dana-Farber Cancer Institute, Boston, and her colleagues in the statement, which calls for increased education, clinical guidance, research, and advocacy on the issue.
Evidence suggests that obese individuals have a significantly worse prognosis than do nonobese patients. One meta-analysis found a 75% increase in mortality in premenopausal women who were obese at the time of breast cancer diagnosis, compared with normal-weight patients. Further, obesity can interfere with the delivery of cancer therapy and is associated with increases in the risk of second primary malignancies, according to the paper published Oct.1 in the Journal of Clinical Oncology.
Dr. Ligibel and her colleagues said many individuals initiate positive health behavior changes after a cancer diagnosis, and such a point can therefore serve as a “teachable moment” that may offer the chance to motivate individuals to implement lifestyle and behavioral changes.
The statement advised assessing patients’ weight by calculating BMI and referring patients to appropriate services such as oncology nutrition, rehabilitation medicine, and exercise physiology. Oncologists also are encouraged to lead by example and to share their personal experiences at attempts to lose weight and to increase physical activity. Participating in community events focused on physical activity and healthy living “can also reinforce the importance of health lifestyle behaviors as part of a a comprehensive cancer-control strategy,” the statement said.
ASCO also announced that it will host a summit to assemble groups needed to conduct lifestyle intervention research in cancer survivors and urged studies of lifestyle changes on outcomes in patients with early-stage cancers.
Additionally, ASCO has developed a toolkit to assist in addressing weight management, including strategies for improving nutrition and physical activity as well as insurance coverage for weight-management services.
The authors declared no conflicts of interest.