User login
Racial, geographic, insurance disparities in endometrial cancer outcomes
MELBOURNE – Residents of the mountain region of the United States are significantly less likely to receive postoperative treatment for advanced endometrial cancer, compared with residents of the Northeast or Midwest, according to an oral presentation made at the biennial meeting of the International Gynecologic Cancer Society.
Retrospective analysis of data from 228,511 patients registered in the National Cancer Data Base – 15.5% of whom were stage III or IV – showed that women living in the mountain region and the Midwest were also significantly more likely to be diagnosed with advanced stage disease than those living in the South (hazard ratio 1.29 and 1.27 respectively, P < .0001)
Researchers also found having Medicaid insurance significantly increased the likelihood of being diagnosed with stage III-IV endometrial cancer (HR 1.51, P < .0001) and decreased overall survival, compared with individuals with private insurance or in managed care (HR 0.85).
“The aim of this study was to estimate the contribution of race and ethnicity, socioeconomic factors such as education and income, as well as place of residence on eventual cancer outcome,” said Dr. Sean Dowdy, professor and chair of gynecologic surgery at the Mayo Clinic.
“Specifically we looked at three questions: one was the factors that are associated with advanced stage at diagnosis, stage III-IV; whether patients received postsurgical treatment when they had advanced disease; and also their overall survival,” Dr. Dowdy told the conference.
While the study initially found that African American women were significantly more likely to be diagnosed with advanced disease than white women, this difference was no longer statistically significant after adjusting for other factors, such as the fourfold higher incidence of serous cancer in African American women.
African American women also had a reduced overall survival, compared with white women, although the study did not find any significant impact of race on treatment rates or the likelihood of being treated at a high-volume facility.
“I expected African Americans to have worse access, and to be diagnosed with advanced-stage disease, and I thought that that would be the reason for their reduced survival, but we actually didn’t see that,” Dr. Dowdy said in an interview.
“The other possibility is, there is something unique about African Americans, about their tumors or about their host response, so maybe their immunological response or something like that may impact their survival.”
The study also observed reduced survival among patients treated at low-volume facilities – although this represented a relatively small number of patients – and noted a twofold increase in reduced survival among patients aged over 80 years.
“In the age category, it’s not surprising that patients were less likely to be treated, but it’s surprising the magnitude of this, so patients that were 90 years were ten times less likely to be treated than patients who were in their 50s,” Dr. Dowdy told the audience.
Commenting on the negative impact of Medicaid and Medicare insurance, Dr. Dowdy pointed out that the study predated the introduction of the Affordable Care Act, and it was too early to tell what impact that might have on the outcomes for those patients without private insurance.
However he also called for more research into the racial disparities in overall survival.
“If the problem is access, then we need to put money into education, and we need to maybe change the system so that it’s more accessible,” Dr. Dowdy said.
“If it’s a biology thing then we need to figure out what that is and figure out whether there are specific treatments that we should be using in these patients.”
The study was supported by the Mayo Clinic, and there were no conflicts of interest declared.
MELBOURNE – Residents of the mountain region of the United States are significantly less likely to receive postoperative treatment for advanced endometrial cancer, compared with residents of the Northeast or Midwest, according to an oral presentation made at the biennial meeting of the International Gynecologic Cancer Society.
Retrospective analysis of data from 228,511 patients registered in the National Cancer Data Base – 15.5% of whom were stage III or IV – showed that women living in the mountain region and the Midwest were also significantly more likely to be diagnosed with advanced stage disease than those living in the South (hazard ratio 1.29 and 1.27 respectively, P < .0001)
Researchers also found having Medicaid insurance significantly increased the likelihood of being diagnosed with stage III-IV endometrial cancer (HR 1.51, P < .0001) and decreased overall survival, compared with individuals with private insurance or in managed care (HR 0.85).
“The aim of this study was to estimate the contribution of race and ethnicity, socioeconomic factors such as education and income, as well as place of residence on eventual cancer outcome,” said Dr. Sean Dowdy, professor and chair of gynecologic surgery at the Mayo Clinic.
“Specifically we looked at three questions: one was the factors that are associated with advanced stage at diagnosis, stage III-IV; whether patients received postsurgical treatment when they had advanced disease; and also their overall survival,” Dr. Dowdy told the conference.
While the study initially found that African American women were significantly more likely to be diagnosed with advanced disease than white women, this difference was no longer statistically significant after adjusting for other factors, such as the fourfold higher incidence of serous cancer in African American women.
African American women also had a reduced overall survival, compared with white women, although the study did not find any significant impact of race on treatment rates or the likelihood of being treated at a high-volume facility.
“I expected African Americans to have worse access, and to be diagnosed with advanced-stage disease, and I thought that that would be the reason for their reduced survival, but we actually didn’t see that,” Dr. Dowdy said in an interview.
“The other possibility is, there is something unique about African Americans, about their tumors or about their host response, so maybe their immunological response or something like that may impact their survival.”
The study also observed reduced survival among patients treated at low-volume facilities – although this represented a relatively small number of patients – and noted a twofold increase in reduced survival among patients aged over 80 years.
“In the age category, it’s not surprising that patients were less likely to be treated, but it’s surprising the magnitude of this, so patients that were 90 years were ten times less likely to be treated than patients who were in their 50s,” Dr. Dowdy told the audience.
Commenting on the negative impact of Medicaid and Medicare insurance, Dr. Dowdy pointed out that the study predated the introduction of the Affordable Care Act, and it was too early to tell what impact that might have on the outcomes for those patients without private insurance.
However he also called for more research into the racial disparities in overall survival.
“If the problem is access, then we need to put money into education, and we need to maybe change the system so that it’s more accessible,” Dr. Dowdy said.
“If it’s a biology thing then we need to figure out what that is and figure out whether there are specific treatments that we should be using in these patients.”
The study was supported by the Mayo Clinic, and there were no conflicts of interest declared.
MELBOURNE – Residents of the mountain region of the United States are significantly less likely to receive postoperative treatment for advanced endometrial cancer, compared with residents of the Northeast or Midwest, according to an oral presentation made at the biennial meeting of the International Gynecologic Cancer Society.
Retrospective analysis of data from 228,511 patients registered in the National Cancer Data Base – 15.5% of whom were stage III or IV – showed that women living in the mountain region and the Midwest were also significantly more likely to be diagnosed with advanced stage disease than those living in the South (hazard ratio 1.29 and 1.27 respectively, P < .0001)
Researchers also found having Medicaid insurance significantly increased the likelihood of being diagnosed with stage III-IV endometrial cancer (HR 1.51, P < .0001) and decreased overall survival, compared with individuals with private insurance or in managed care (HR 0.85).
“The aim of this study was to estimate the contribution of race and ethnicity, socioeconomic factors such as education and income, as well as place of residence on eventual cancer outcome,” said Dr. Sean Dowdy, professor and chair of gynecologic surgery at the Mayo Clinic.
“Specifically we looked at three questions: one was the factors that are associated with advanced stage at diagnosis, stage III-IV; whether patients received postsurgical treatment when they had advanced disease; and also their overall survival,” Dr. Dowdy told the conference.
While the study initially found that African American women were significantly more likely to be diagnosed with advanced disease than white women, this difference was no longer statistically significant after adjusting for other factors, such as the fourfold higher incidence of serous cancer in African American women.
African American women also had a reduced overall survival, compared with white women, although the study did not find any significant impact of race on treatment rates or the likelihood of being treated at a high-volume facility.
“I expected African Americans to have worse access, and to be diagnosed with advanced-stage disease, and I thought that that would be the reason for their reduced survival, but we actually didn’t see that,” Dr. Dowdy said in an interview.
“The other possibility is, there is something unique about African Americans, about their tumors or about their host response, so maybe their immunological response or something like that may impact their survival.”
The study also observed reduced survival among patients treated at low-volume facilities – although this represented a relatively small number of patients – and noted a twofold increase in reduced survival among patients aged over 80 years.
“In the age category, it’s not surprising that patients were less likely to be treated, but it’s surprising the magnitude of this, so patients that were 90 years were ten times less likely to be treated than patients who were in their 50s,” Dr. Dowdy told the audience.
Commenting on the negative impact of Medicaid and Medicare insurance, Dr. Dowdy pointed out that the study predated the introduction of the Affordable Care Act, and it was too early to tell what impact that might have on the outcomes for those patients without private insurance.
However he also called for more research into the racial disparities in overall survival.
“If the problem is access, then we need to put money into education, and we need to maybe change the system so that it’s more accessible,” Dr. Dowdy said.
“If it’s a biology thing then we need to figure out what that is and figure out whether there are specific treatments that we should be using in these patients.”
The study was supported by the Mayo Clinic, and there were no conflicts of interest declared.
AT IGCS 2014
Key clinical point: Geographic location and insurance status have a significant impact on stage at diagnosis, rates of postoperative treatment, and overall survival among women with endometrial cancer.
Major finding: Women with Medicaid insurance have a 51% increase in the likelihood of being diagnosed with stage III-IV endometrial cancer, compared with the privately insured.
Data source: Retrospective analysis of data from 228,511 patients registered in the National Cancer Database.
Disclosures: The study was supported by the Mayo Clinic. There were no conflicts of interest declared.
Bevacizumab plus fosbretabulin improves PFS* in ovarian cancer
MELBOURNE – The combination of the antiangiogenesis drug bevacizumab and the vascular-disrupting agent fosbretabulin is superior to bevacizumab alone in the treatment of recurrent ovarian, tubal, and peritoneal cancer, according to an oral presentation made at the biennial meeting of the International Gynecologic Cancer Society.
A randomized, open-label, phase II study in 107 patients found a significant improvement in progression-free survival among patients treated with the combination of bevacizumab and fosbretabulin versus bevacizumab alone (hazard ratio, 0.685; P = .049), with a median improvement of 2.5 months.
Researchers observed a greater improvement in progression-free survival among patients with platinum-resistant disease, with a 3.3-month improvement (HR, 0.57; P = .01), but the difference in platinum-sensitive patients failed to reach statistical significance.
Patients in the combination arm also showed a higher response rate than those in the single-therapy arm.
Presenter Dr. Bradley Monk said the challenge with using antiangiogenesis agents alone was that they only dealt with new blood vessel growth.
“The Achilles’ heel of angiogenesis is, what about the blood vessels that are already there?” said Dr. Monk, director of gynecological oncology at the University of Arizona Cancer Center and Dignity Health, both in Phoenix.
Dr. Monk said antivascular agents such as fosbretabulin collapse the established tumor blood vessels, causing mechanistic obstructions that lead to tumor necrosis, and because of their tolerability, they can be combined with chemotherapy and antiangiogenesis agents.
While this approach has been considered before, not all vascular-disrupting agents were suitable.
“There are different types of vascular-disrupting agents. They’re not all the same, and, in fact, there are vascular-disrupting agents that have not worked,” Dr. Monk said in an interview.
He told the conference that fosbretabulin is a potent and reversible tubulin depolymerizing agent, targeting the tubulin cytoskeleton that is so important in tumor-associated blood vessels, which lack smooth muscle and pericyte coverage and rely on tubulin to maintain their structure.
The combination approach was associated with a significantly higher incidence of adverse events, compared with bevacizumab alone, and 16.7% of patients in the combination arm came off the study because of adverse events, compared with 5.7% of patients in the monotherapy arm.
There were more cases of grade 3 hypertension in the combination arm than in the monotherapy arm, which Dr. Monk said was a known side effect of fosbretabulin.
There was one grade 4 hypertension event in the combination arm, and one grade 4 metabolism-related adverse event in the bevacizumab-alone arm.
Data on overall survival were not mature, but Dr. Monk said that based on these results, the combination of fosbretabulin and bevacizumab warranted further study in ovarian cancer.
The study was sponsored by the National Cancer Institute. One author is an employee of fosbretabulin manufacturer Oxigene. The presenter declared expenses, consultancies, and research funding from several pharmaceutical companies, including Oxigene.
*Correction, 11/10/2014: A previous version of the headline did not clearly state that the improvement was in progression free survival.
MELBOURNE – The combination of the antiangiogenesis drug bevacizumab and the vascular-disrupting agent fosbretabulin is superior to bevacizumab alone in the treatment of recurrent ovarian, tubal, and peritoneal cancer, according to an oral presentation made at the biennial meeting of the International Gynecologic Cancer Society.
A randomized, open-label, phase II study in 107 patients found a significant improvement in progression-free survival among patients treated with the combination of bevacizumab and fosbretabulin versus bevacizumab alone (hazard ratio, 0.685; P = .049), with a median improvement of 2.5 months.
Researchers observed a greater improvement in progression-free survival among patients with platinum-resistant disease, with a 3.3-month improvement (HR, 0.57; P = .01), but the difference in platinum-sensitive patients failed to reach statistical significance.
Patients in the combination arm also showed a higher response rate than those in the single-therapy arm.
Presenter Dr. Bradley Monk said the challenge with using antiangiogenesis agents alone was that they only dealt with new blood vessel growth.
“The Achilles’ heel of angiogenesis is, what about the blood vessels that are already there?” said Dr. Monk, director of gynecological oncology at the University of Arizona Cancer Center and Dignity Health, both in Phoenix.
Dr. Monk said antivascular agents such as fosbretabulin collapse the established tumor blood vessels, causing mechanistic obstructions that lead to tumor necrosis, and because of their tolerability, they can be combined with chemotherapy and antiangiogenesis agents.
While this approach has been considered before, not all vascular-disrupting agents were suitable.
“There are different types of vascular-disrupting agents. They’re not all the same, and, in fact, there are vascular-disrupting agents that have not worked,” Dr. Monk said in an interview.
He told the conference that fosbretabulin is a potent and reversible tubulin depolymerizing agent, targeting the tubulin cytoskeleton that is so important in tumor-associated blood vessels, which lack smooth muscle and pericyte coverage and rely on tubulin to maintain their structure.
The combination approach was associated with a significantly higher incidence of adverse events, compared with bevacizumab alone, and 16.7% of patients in the combination arm came off the study because of adverse events, compared with 5.7% of patients in the monotherapy arm.
There were more cases of grade 3 hypertension in the combination arm than in the monotherapy arm, which Dr. Monk said was a known side effect of fosbretabulin.
There was one grade 4 hypertension event in the combination arm, and one grade 4 metabolism-related adverse event in the bevacizumab-alone arm.
Data on overall survival were not mature, but Dr. Monk said that based on these results, the combination of fosbretabulin and bevacizumab warranted further study in ovarian cancer.
The study was sponsored by the National Cancer Institute. One author is an employee of fosbretabulin manufacturer Oxigene. The presenter declared expenses, consultancies, and research funding from several pharmaceutical companies, including Oxigene.
*Correction, 11/10/2014: A previous version of the headline did not clearly state that the improvement was in progression free survival.
MELBOURNE – The combination of the antiangiogenesis drug bevacizumab and the vascular-disrupting agent fosbretabulin is superior to bevacizumab alone in the treatment of recurrent ovarian, tubal, and peritoneal cancer, according to an oral presentation made at the biennial meeting of the International Gynecologic Cancer Society.
A randomized, open-label, phase II study in 107 patients found a significant improvement in progression-free survival among patients treated with the combination of bevacizumab and fosbretabulin versus bevacizumab alone (hazard ratio, 0.685; P = .049), with a median improvement of 2.5 months.
Researchers observed a greater improvement in progression-free survival among patients with platinum-resistant disease, with a 3.3-month improvement (HR, 0.57; P = .01), but the difference in platinum-sensitive patients failed to reach statistical significance.
Patients in the combination arm also showed a higher response rate than those in the single-therapy arm.
Presenter Dr. Bradley Monk said the challenge with using antiangiogenesis agents alone was that they only dealt with new blood vessel growth.
“The Achilles’ heel of angiogenesis is, what about the blood vessels that are already there?” said Dr. Monk, director of gynecological oncology at the University of Arizona Cancer Center and Dignity Health, both in Phoenix.
Dr. Monk said antivascular agents such as fosbretabulin collapse the established tumor blood vessels, causing mechanistic obstructions that lead to tumor necrosis, and because of their tolerability, they can be combined with chemotherapy and antiangiogenesis agents.
While this approach has been considered before, not all vascular-disrupting agents were suitable.
“There are different types of vascular-disrupting agents. They’re not all the same, and, in fact, there are vascular-disrupting agents that have not worked,” Dr. Monk said in an interview.
He told the conference that fosbretabulin is a potent and reversible tubulin depolymerizing agent, targeting the tubulin cytoskeleton that is so important in tumor-associated blood vessels, which lack smooth muscle and pericyte coverage and rely on tubulin to maintain their structure.
The combination approach was associated with a significantly higher incidence of adverse events, compared with bevacizumab alone, and 16.7% of patients in the combination arm came off the study because of adverse events, compared with 5.7% of patients in the monotherapy arm.
There were more cases of grade 3 hypertension in the combination arm than in the monotherapy arm, which Dr. Monk said was a known side effect of fosbretabulin.
There was one grade 4 hypertension event in the combination arm, and one grade 4 metabolism-related adverse event in the bevacizumab-alone arm.
Data on overall survival were not mature, but Dr. Monk said that based on these results, the combination of fosbretabulin and bevacizumab warranted further study in ovarian cancer.
The study was sponsored by the National Cancer Institute. One author is an employee of fosbretabulin manufacturer Oxigene. The presenter declared expenses, consultancies, and research funding from several pharmaceutical companies, including Oxigene.
*Correction, 11/10/2014: A previous version of the headline did not clearly state that the improvement was in progression free survival.
AT IGCS 2014
Key clinical point: The combination of bevacizumab and fosbretabulin significantly improves progression-free survival, compared with bevacizumab alone.
Major finding: The combination of bevacizumab and fosbretabulin achieved a 2.5-month improvement in progression-free survival.
Data source: Prospective, randomized, open-label phase II study in 107 patients with recurrent ovarian, tubal, and peritoneal cancer.
Disclosures: The study was sponsored by the National Cancer Institute. One author is an employee of fosbretabulin manufacturer Oxigene. The presenter declared expenses, consultancies, and research funding from several pharmaceutical companies, including Oxigene.
Elevated branched-chain amino acids may predict pancreatic cancer
Elevated plasma levels of three branched-chain amino acids may predict the development of pancreatic ductal adenocarcinoma at least 2 years before diagnosis, and well before the onset of clinical cachexia, new data suggest.
Researchers retrospectively examined blood samples taken in four large prospective cohort studies, including the Nurses Health Study and the Women’s Health Initiative, and found significant associations between the levels of 15 metabolites and future diagnosis of pancreatic cancer.
In particular, elevated plasma levels of isoleucine, leucine, and valine were highly predictive of later pancreatic cancer, as patients in the top quintile of increasing levels had at least a twofold increase in the risk of pancreatic cancer, compared with those in the bottom quintile, according to a paper published online in Nature Medicine.
Researchers found that the associations between elevated branched-chain amino acids and future cancer diagnosis were most significant between 2 and 5 years before the diagnosis (Nature Med. 2014;20:1193-8 [doi:10.1038/nm.3686]).
Previous studies attempting to single out metabolite biomarkers predicting future risk of cancer have relied on a cross-sectional design, comparing affected patients with cancer-free individuals, which researchers said was likely to be complicated by the impact of advanced disease on circulating metabolite profiles.
“This is particularly true for patients with pancreatic cancer, who commonly have significant anorexia, weight loss, and pancreatic insufficiency at the time of diagnosis,” wrote Jared R. Mayers, Ph.D., from the Koch Institute for Integrative Cancer Research at MIT, Boston,and his colleagues.
“Increased muscle catabolism represents one aspect of cancer-associated cachexia, a wasting syndrome frequently affecting patients with advanced PDAC [pancreatic ductal adenocarcinoma] and contributing to worse outcomes.”“Our findings, however, suggest that protein breakdown begins much earlier than previously appreciated and predates onset of clinical cachexia.”
Elevated circulating branched-chain amino acids are known to be associated with a future risk of diabetes, and type 2 diabetes is also a predisposing factor for pancreatic cancer.
However, researchers found the results remained unchanged after excluding subjects who had diabetes at baseline, suggesting that the findings were not simply indicating the prevalent diabetes associated with the later development of pancreatic cancer.
The study showed circulating branched-chain amino acids (BCAA) were also modestly correlated with obesity and glucose intolerance; however, the relationship with future pancreatic cancer risk persisted after accounting for those markers.
Researchers also tested the possibility that occult pancreatic cancer may have already been present during the periods showing the strongest correlations between elevated BCAAs and later cancer risk, by conducting a prospective serial blood sampling study in mice genetically predisposed to pancreatic cancer.
They found the mice – which have a median survival of 21 weeks – initially showed similar levels of BCAAs to normal mice, but these increased significantly from 15 to 17 weeks before death.
“These data suggest circulating BCAA elevations accompany early PDAC,” the researchers wrote.
The study was supported by the Howard Hughes Medical Institute, the Lustgarten Foundation, and grants from the National Institutes of Health, the Nestle Research Center, the Robert T. and Judith B. Hale Fund for Pancreatic Cancer, Perry S. Levy Fund for Gastrointestinal Cancer Research, the Pappas Family Research Fund for Pancreatic Cancer, the Burroughs Wellcome Fund, the Damon Runyon Cancer Research Foundation, the Smith Family and the Stern Family, the American Society of Clinical Oncology Conquer Cancer Foundation, the Howard Hughes Medical Institute and Promises for Purple. There were no conflicts of interest declared.
Elevated plasma levels of three branched-chain amino acids may predict the development of pancreatic ductal adenocarcinoma at least 2 years before diagnosis, and well before the onset of clinical cachexia, new data suggest.
Researchers retrospectively examined blood samples taken in four large prospective cohort studies, including the Nurses Health Study and the Women’s Health Initiative, and found significant associations between the levels of 15 metabolites and future diagnosis of pancreatic cancer.
In particular, elevated plasma levels of isoleucine, leucine, and valine were highly predictive of later pancreatic cancer, as patients in the top quintile of increasing levels had at least a twofold increase in the risk of pancreatic cancer, compared with those in the bottom quintile, according to a paper published online in Nature Medicine.
Researchers found that the associations between elevated branched-chain amino acids and future cancer diagnosis were most significant between 2 and 5 years before the diagnosis (Nature Med. 2014;20:1193-8 [doi:10.1038/nm.3686]).
Previous studies attempting to single out metabolite biomarkers predicting future risk of cancer have relied on a cross-sectional design, comparing affected patients with cancer-free individuals, which researchers said was likely to be complicated by the impact of advanced disease on circulating metabolite profiles.
“This is particularly true for patients with pancreatic cancer, who commonly have significant anorexia, weight loss, and pancreatic insufficiency at the time of diagnosis,” wrote Jared R. Mayers, Ph.D., from the Koch Institute for Integrative Cancer Research at MIT, Boston,and his colleagues.
“Increased muscle catabolism represents one aspect of cancer-associated cachexia, a wasting syndrome frequently affecting patients with advanced PDAC [pancreatic ductal adenocarcinoma] and contributing to worse outcomes.”“Our findings, however, suggest that protein breakdown begins much earlier than previously appreciated and predates onset of clinical cachexia.”
Elevated circulating branched-chain amino acids are known to be associated with a future risk of diabetes, and type 2 diabetes is also a predisposing factor for pancreatic cancer.
However, researchers found the results remained unchanged after excluding subjects who had diabetes at baseline, suggesting that the findings were not simply indicating the prevalent diabetes associated with the later development of pancreatic cancer.
The study showed circulating branched-chain amino acids (BCAA) were also modestly correlated with obesity and glucose intolerance; however, the relationship with future pancreatic cancer risk persisted after accounting for those markers.
Researchers also tested the possibility that occult pancreatic cancer may have already been present during the periods showing the strongest correlations between elevated BCAAs and later cancer risk, by conducting a prospective serial blood sampling study in mice genetically predisposed to pancreatic cancer.
They found the mice – which have a median survival of 21 weeks – initially showed similar levels of BCAAs to normal mice, but these increased significantly from 15 to 17 weeks before death.
“These data suggest circulating BCAA elevations accompany early PDAC,” the researchers wrote.
The study was supported by the Howard Hughes Medical Institute, the Lustgarten Foundation, and grants from the National Institutes of Health, the Nestle Research Center, the Robert T. and Judith B. Hale Fund for Pancreatic Cancer, Perry S. Levy Fund for Gastrointestinal Cancer Research, the Pappas Family Research Fund for Pancreatic Cancer, the Burroughs Wellcome Fund, the Damon Runyon Cancer Research Foundation, the Smith Family and the Stern Family, the American Society of Clinical Oncology Conquer Cancer Foundation, the Howard Hughes Medical Institute and Promises for Purple. There were no conflicts of interest declared.
Elevated plasma levels of three branched-chain amino acids may predict the development of pancreatic ductal adenocarcinoma at least 2 years before diagnosis, and well before the onset of clinical cachexia, new data suggest.
Researchers retrospectively examined blood samples taken in four large prospective cohort studies, including the Nurses Health Study and the Women’s Health Initiative, and found significant associations between the levels of 15 metabolites and future diagnosis of pancreatic cancer.
In particular, elevated plasma levels of isoleucine, leucine, and valine were highly predictive of later pancreatic cancer, as patients in the top quintile of increasing levels had at least a twofold increase in the risk of pancreatic cancer, compared with those in the bottom quintile, according to a paper published online in Nature Medicine.
Researchers found that the associations between elevated branched-chain amino acids and future cancer diagnosis were most significant between 2 and 5 years before the diagnosis (Nature Med. 2014;20:1193-8 [doi:10.1038/nm.3686]).
Previous studies attempting to single out metabolite biomarkers predicting future risk of cancer have relied on a cross-sectional design, comparing affected patients with cancer-free individuals, which researchers said was likely to be complicated by the impact of advanced disease on circulating metabolite profiles.
“This is particularly true for patients with pancreatic cancer, who commonly have significant anorexia, weight loss, and pancreatic insufficiency at the time of diagnosis,” wrote Jared R. Mayers, Ph.D., from the Koch Institute for Integrative Cancer Research at MIT, Boston,and his colleagues.
“Increased muscle catabolism represents one aspect of cancer-associated cachexia, a wasting syndrome frequently affecting patients with advanced PDAC [pancreatic ductal adenocarcinoma] and contributing to worse outcomes.”“Our findings, however, suggest that protein breakdown begins much earlier than previously appreciated and predates onset of clinical cachexia.”
Elevated circulating branched-chain amino acids are known to be associated with a future risk of diabetes, and type 2 diabetes is also a predisposing factor for pancreatic cancer.
However, researchers found the results remained unchanged after excluding subjects who had diabetes at baseline, suggesting that the findings were not simply indicating the prevalent diabetes associated with the later development of pancreatic cancer.
The study showed circulating branched-chain amino acids (BCAA) were also modestly correlated with obesity and glucose intolerance; however, the relationship with future pancreatic cancer risk persisted after accounting for those markers.
Researchers also tested the possibility that occult pancreatic cancer may have already been present during the periods showing the strongest correlations between elevated BCAAs and later cancer risk, by conducting a prospective serial blood sampling study in mice genetically predisposed to pancreatic cancer.
They found the mice – which have a median survival of 21 weeks – initially showed similar levels of BCAAs to normal mice, but these increased significantly from 15 to 17 weeks before death.
“These data suggest circulating BCAA elevations accompany early PDAC,” the researchers wrote.
The study was supported by the Howard Hughes Medical Institute, the Lustgarten Foundation, and grants from the National Institutes of Health, the Nestle Research Center, the Robert T. and Judith B. Hale Fund for Pancreatic Cancer, Perry S. Levy Fund for Gastrointestinal Cancer Research, the Pappas Family Research Fund for Pancreatic Cancer, the Burroughs Wellcome Fund, the Damon Runyon Cancer Research Foundation, the Smith Family and the Stern Family, the American Society of Clinical Oncology Conquer Cancer Foundation, the Howard Hughes Medical Institute and Promises for Purple. There were no conflicts of interest declared.
FROM NATURE MEDICINE
Key clinical point: Elevated plasma levels of three branched-chain amino acids may predict the development of pancreatic cancer at least 2 years before diagnosis.
Major finding: Individuals in the highest quintile of plasma isoleucine, leucine, and valine have a twofold increase in the risk of pancreatic cancer.
Data source: Retrospective analysis of data from four large prospective cohort studies.
Disclosures: The study was supported by the Howard Hughes Medical Institute, the Lustgarten Foundation, and grants from the National Institutes of Health, the Nestle Research Center, the Robert T. and Judith B. Hale Fund for Pancreatic Cancer, Perry S. Levy Fund for Gastrointestinal Cancer Research, the Pappas Family Research Fund for Pancreatic Cancer, the Burroughs Wellcome Fund, the Damon Runyon Cancer Research Foundation, the Smith Family and the Stern Family, the American Society of Clinical Oncology Conquer Cancer Foundation, the Howard Hughes Medical Institute, and Promises for Purple. There were no conflicts of interest declared.
Adverse events remain rare for noninvasive and minimally invasive cosmetic procedures
Noninvasive and minimally invasive cosmetic dermatology procedures – such as laser treatments, neurotoxin injections, and soft tissue augmentation – are associated with an aggregate adverse event rate of less than 1% when performed by experienced, board-certified dermatologists, based on data from a multicenter prospective cohort study of more than 20,000 procedures.
Researchers used a web-based data collection system to record relevant procedures and adverse events at eight private and institutional dermatology outpatient clinical practices. They identified a total of 48 adverse events from 20,399 procedures, representing an overall adverse event rate of 0.24%.
The most common adverse events were hyperpigmentation after laser or energy treatments and lumps, nodules, or beading after treatment with neurotoxins or fillers, with adverse events most commonly occurring after procedures on the cheeks, nasolabial folds, and eyelids. No serious adverse events were reported.
“Patients seeking such [noninvasive and minimally invasive] procedures can be reassured that, at least in the hands of trained board-certified dermatologists, they pose minimal risk,” wrote Dr. Murad Alam of Northwestern University, Chicago, and associates. The findings were published online November 5 in JAMA Dermatology (doi:10.1001/jamadermatol.2014.2494).
The study was supported by departmental research funds from the Feinberg School of Medicine, Northwestern University. Dr. Alam declared consultancies with Amway and Leo Pharma, and has been principal investigator on studies funded in part by Allergan, Bioform, Medicis, and Ulthera. There were no other conflicts of interest declared.
Noninvasive and minimally invasive cosmetic dermatology procedures – such as laser treatments, neurotoxin injections, and soft tissue augmentation – are associated with an aggregate adverse event rate of less than 1% when performed by experienced, board-certified dermatologists, based on data from a multicenter prospective cohort study of more than 20,000 procedures.
Researchers used a web-based data collection system to record relevant procedures and adverse events at eight private and institutional dermatology outpatient clinical practices. They identified a total of 48 adverse events from 20,399 procedures, representing an overall adverse event rate of 0.24%.
The most common adverse events were hyperpigmentation after laser or energy treatments and lumps, nodules, or beading after treatment with neurotoxins or fillers, with adverse events most commonly occurring after procedures on the cheeks, nasolabial folds, and eyelids. No serious adverse events were reported.
“Patients seeking such [noninvasive and minimally invasive] procedures can be reassured that, at least in the hands of trained board-certified dermatologists, they pose minimal risk,” wrote Dr. Murad Alam of Northwestern University, Chicago, and associates. The findings were published online November 5 in JAMA Dermatology (doi:10.1001/jamadermatol.2014.2494).
The study was supported by departmental research funds from the Feinberg School of Medicine, Northwestern University. Dr. Alam declared consultancies with Amway and Leo Pharma, and has been principal investigator on studies funded in part by Allergan, Bioform, Medicis, and Ulthera. There were no other conflicts of interest declared.
Noninvasive and minimally invasive cosmetic dermatology procedures – such as laser treatments, neurotoxin injections, and soft tissue augmentation – are associated with an aggregate adverse event rate of less than 1% when performed by experienced, board-certified dermatologists, based on data from a multicenter prospective cohort study of more than 20,000 procedures.
Researchers used a web-based data collection system to record relevant procedures and adverse events at eight private and institutional dermatology outpatient clinical practices. They identified a total of 48 adverse events from 20,399 procedures, representing an overall adverse event rate of 0.24%.
The most common adverse events were hyperpigmentation after laser or energy treatments and lumps, nodules, or beading after treatment with neurotoxins or fillers, with adverse events most commonly occurring after procedures on the cheeks, nasolabial folds, and eyelids. No serious adverse events were reported.
“Patients seeking such [noninvasive and minimally invasive] procedures can be reassured that, at least in the hands of trained board-certified dermatologists, they pose minimal risk,” wrote Dr. Murad Alam of Northwestern University, Chicago, and associates. The findings were published online November 5 in JAMA Dermatology (doi:10.1001/jamadermatol.2014.2494).
The study was supported by departmental research funds from the Feinberg School of Medicine, Northwestern University. Dr. Alam declared consultancies with Amway and Leo Pharma, and has been principal investigator on studies funded in part by Allergan, Bioform, Medicis, and Ulthera. There were no other conflicts of interest declared.
FROM JAMA DERMATOLOGY
Key clinical point: Noninvasive and minimally invasive cosmetic dermatology procedures are safe when performed by experienced dermatologists.
Major finding: Noninvasive and minimally invasive cosmetic dermatology procedures were associated with a 0.24% adverse event rate.
Data source: Multicenter prospective cohort study of 20,399 procedures at eight institutions.
Disclosures: The study was supported by departmental research funds from the Feinberg School of Medicine, Northwestern University. Dr. Alam declared consultancies with Amway and Leo Pharma, and has been principal investigator on studies funded in part by Allergan, Bioform, Medicis, and Ulthera. There were no other conflicts of interest declared.
Colorectal cancer on the increase in young adults
Colorectal cancer rates have declined in both men and women since 1975, particularly in those over age 75 years, but there has also been a significant increase in the incidence among those aged 20-34 years, a retrospective cohort study shows.
Researchers analyzed data from 393,241 patients registered in the Surveillance, Epidemiology, and End Results (SEER) colorectal cancer registry from 1975 to 2010, finding the overall, age-adjusted incidence of colorectal cancer decreased by 0.92% during that time.
They observed a steady decline in the incidence of colorectal cancer in the over 50 age group, with a 1.15% decrease in those aged over 75 years, while in those aged 20-34 years, there was a nearly 2% increase, according to data published online November 5 in JAMA Surgery (doi:10.1001/jamasurg.2014.1756).
“At the present rate, the incidence rate for young patients with newly diagnosed colon or rectal cancer will nearly double by 2030, while it will similarly decline by more than one-third among patients older than the screening age of 50 years,” wrote Dr. Christina E. Bailey of the University of Texas M.D. Anderson Cancer Center, Houston, and colleagues.
The study was supported by grants from the National Institutes of Health and National Cancer Institute. There were no conflicts of interest declared.
Colorectal cancer rates have declined in both men and women since 1975, particularly in those over age 75 years, but there has also been a significant increase in the incidence among those aged 20-34 years, a retrospective cohort study shows.
Researchers analyzed data from 393,241 patients registered in the Surveillance, Epidemiology, and End Results (SEER) colorectal cancer registry from 1975 to 2010, finding the overall, age-adjusted incidence of colorectal cancer decreased by 0.92% during that time.
They observed a steady decline in the incidence of colorectal cancer in the over 50 age group, with a 1.15% decrease in those aged over 75 years, while in those aged 20-34 years, there was a nearly 2% increase, according to data published online November 5 in JAMA Surgery (doi:10.1001/jamasurg.2014.1756).
“At the present rate, the incidence rate for young patients with newly diagnosed colon or rectal cancer will nearly double by 2030, while it will similarly decline by more than one-third among patients older than the screening age of 50 years,” wrote Dr. Christina E. Bailey of the University of Texas M.D. Anderson Cancer Center, Houston, and colleagues.
The study was supported by grants from the National Institutes of Health and National Cancer Institute. There were no conflicts of interest declared.
Colorectal cancer rates have declined in both men and women since 1975, particularly in those over age 75 years, but there has also been a significant increase in the incidence among those aged 20-34 years, a retrospective cohort study shows.
Researchers analyzed data from 393,241 patients registered in the Surveillance, Epidemiology, and End Results (SEER) colorectal cancer registry from 1975 to 2010, finding the overall, age-adjusted incidence of colorectal cancer decreased by 0.92% during that time.
They observed a steady decline in the incidence of colorectal cancer in the over 50 age group, with a 1.15% decrease in those aged over 75 years, while in those aged 20-34 years, there was a nearly 2% increase, according to data published online November 5 in JAMA Surgery (doi:10.1001/jamasurg.2014.1756).
“At the present rate, the incidence rate for young patients with newly diagnosed colon or rectal cancer will nearly double by 2030, while it will similarly decline by more than one-third among patients older than the screening age of 50 years,” wrote Dr. Christina E. Bailey of the University of Texas M.D. Anderson Cancer Center, Houston, and colleagues.
The study was supported by grants from the National Institutes of Health and National Cancer Institute. There were no conflicts of interest declared.
FROM JAMA SURGERY
Key clinical point: Colorectal cancer rates have declined steadily since 1975 in those over 50, but have increased in the 20- to 34-year-old age group.
Major finding: Colorectal cancer incidence has increased nearly 2% in those aged 20-34 years.
Data source: Retrospective cohort study of data from 393,241 patients registered in the Surveillance, Epidemiology, and End Results (SEER) colorectal cancer registry.
Disclosures: The study was supported by grants from the National Institutes of Health and National Cancer Institute. There were no conflicts of interest declared.
Tetravalent dengue vaccine shown to be effective and safe in children
The phase III trial of a tetravalent dengue vaccine among children in Latin America has shown the vaccine to be protective against the virus, with significant reductions in the risk of hospitalization with dengue.
The placebo-controlled trial of a three-dose vaccine schedule among 20,869 children living in dengue-endemic regions showed a vaccine efficacy of 60.8% in the per-protocol analysis and 64.7% in the intention-to-treat population of individuals who received at least one injection, researchers said at the annual meeting of the American Society of Tropical Medicine and Hygiene.
The vaccine had an efficacy of 95.5% in preventing severe dengue and 80.3% efficacy in preventing hospitalization, while also showing efficacy against serotypes 1, 2, 3, and 4 and a rate of adverse events similar to that of the placebo group, according to a paper published simultaneously online in the New England Journal of Medicine (2014 Nov. 3 [doi:10.1056/NEJMoa1411037]).
“Overall, the results of our study and the Asian study provide a consistent picture of the efficacy and safety of this dengue vaccine after 25 months of active surveillance in 10 countries among different populations (including a variety of ages and ethnic backgrounds) over different seasons with different circulating serotypes and levels of endemicity,” wrote Dr. Luis Villar of the Universidad Industrial de Santander, Colombia, and colleagues.
The study was supported by Sanofi Pasteur. Some authors were employees of Sanofi Pasteur, and others declared institutional and research grants and honoraria from the study sponsor and other pharmaceutical companies.
The phase III trial of a tetravalent dengue vaccine among children in Latin America has shown the vaccine to be protective against the virus, with significant reductions in the risk of hospitalization with dengue.
The placebo-controlled trial of a three-dose vaccine schedule among 20,869 children living in dengue-endemic regions showed a vaccine efficacy of 60.8% in the per-protocol analysis and 64.7% in the intention-to-treat population of individuals who received at least one injection, researchers said at the annual meeting of the American Society of Tropical Medicine and Hygiene.
The vaccine had an efficacy of 95.5% in preventing severe dengue and 80.3% efficacy in preventing hospitalization, while also showing efficacy against serotypes 1, 2, 3, and 4 and a rate of adverse events similar to that of the placebo group, according to a paper published simultaneously online in the New England Journal of Medicine (2014 Nov. 3 [doi:10.1056/NEJMoa1411037]).
“Overall, the results of our study and the Asian study provide a consistent picture of the efficacy and safety of this dengue vaccine after 25 months of active surveillance in 10 countries among different populations (including a variety of ages and ethnic backgrounds) over different seasons with different circulating serotypes and levels of endemicity,” wrote Dr. Luis Villar of the Universidad Industrial de Santander, Colombia, and colleagues.
The study was supported by Sanofi Pasteur. Some authors were employees of Sanofi Pasteur, and others declared institutional and research grants and honoraria from the study sponsor and other pharmaceutical companies.
The phase III trial of a tetravalent dengue vaccine among children in Latin America has shown the vaccine to be protective against the virus, with significant reductions in the risk of hospitalization with dengue.
The placebo-controlled trial of a three-dose vaccine schedule among 20,869 children living in dengue-endemic regions showed a vaccine efficacy of 60.8% in the per-protocol analysis and 64.7% in the intention-to-treat population of individuals who received at least one injection, researchers said at the annual meeting of the American Society of Tropical Medicine and Hygiene.
The vaccine had an efficacy of 95.5% in preventing severe dengue and 80.3% efficacy in preventing hospitalization, while also showing efficacy against serotypes 1, 2, 3, and 4 and a rate of adverse events similar to that of the placebo group, according to a paper published simultaneously online in the New England Journal of Medicine (2014 Nov. 3 [doi:10.1056/NEJMoa1411037]).
“Overall, the results of our study and the Asian study provide a consistent picture of the efficacy and safety of this dengue vaccine after 25 months of active surveillance in 10 countries among different populations (including a variety of ages and ethnic backgrounds) over different seasons with different circulating serotypes and levels of endemicity,” wrote Dr. Luis Villar of the Universidad Industrial de Santander, Colombia, and colleagues.
The study was supported by Sanofi Pasteur. Some authors were employees of Sanofi Pasteur, and others declared institutional and research grants and honoraria from the study sponsor and other pharmaceutical companies.
FROM ASTMH 2014
Key clinical point: A tetravalent dengue vaccine reduced the incidence of dengue, including severe infection, and hospitalizations.
Major finding: The study showed a vaccine efficacy of 60.8% in the per-protocol analysis and 64.7% in the intention-to-treat population.
Data source: Randomized, blinded, placebo-controlled phase III trial in 20,869 children.
Disclosures: The study was supported by Sanofi Pasteur. Some authors were employees of Sanofi Pasteur, and others declared institutional and research grants and honoraria from the study sponsor and other pharmaceutical companies.
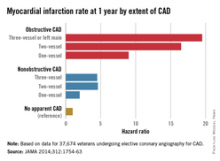
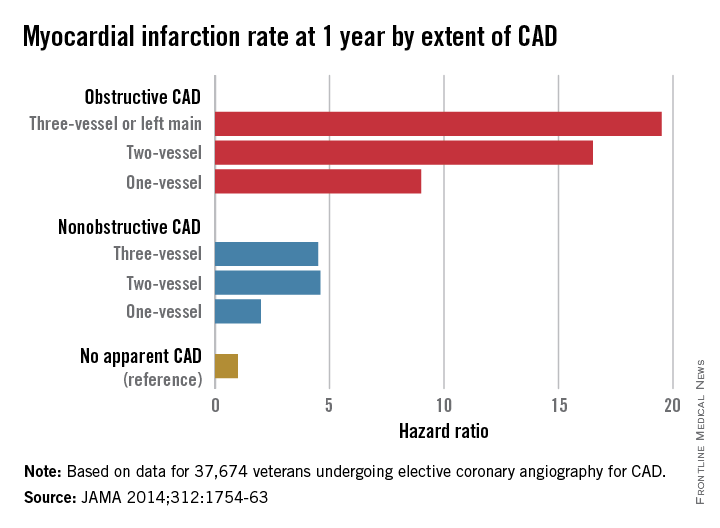
Nonobstructive CAD increases risk of MI and death
Patients with nonobstructive coronary artery disease are at significantly greater risk of myocardial infarction and all-cause mortality than are those with no CAD, and research is needed to explore risk mitigation methods in this group, say the authors of a retrospective cohort study.
The study of 37,674 U.S. veterans undergoing elective coronary angiography for CAD found patients with one-vessel nonobstructive CAD were at double the risk of a MI within 1 year, compared with those with no apparent CAD (see chart), and showed a 60% increase in mortality among individuals with three-vessel nonobstructive CAD.
“The 1-year MI risk progressively increased by CAD extent, rather than abruptly increasing between nonobstructive and obstructive CAD,” wrote Dr. Thomas M. Maddox of the Denver Veterans Affairs Medical Center and his colleagues, in an article published online Nov. 4 (JAMA 2014;312:1754-63 [doi:10.1001/jama.2014.14681]). [However] empirical evidence is lacking as to whether these patients benefit from the prevention therapies recommended for their obstructive CAD counterparts.”
The study was supported by the Department of Veterans Affairs Office of Information and Analytics. Authors declared a variety of funding, directorships, and committee positions in public and private industry.
Patients with nonobstructive coronary artery disease are at significantly greater risk of myocardial infarction and all-cause mortality than are those with no CAD, and research is needed to explore risk mitigation methods in this group, say the authors of a retrospective cohort study.
The study of 37,674 U.S. veterans undergoing elective coronary angiography for CAD found patients with one-vessel nonobstructive CAD were at double the risk of a MI within 1 year, compared with those with no apparent CAD (see chart), and showed a 60% increase in mortality among individuals with three-vessel nonobstructive CAD.
“The 1-year MI risk progressively increased by CAD extent, rather than abruptly increasing between nonobstructive and obstructive CAD,” wrote Dr. Thomas M. Maddox of the Denver Veterans Affairs Medical Center and his colleagues, in an article published online Nov. 4 (JAMA 2014;312:1754-63 [doi:10.1001/jama.2014.14681]). [However] empirical evidence is lacking as to whether these patients benefit from the prevention therapies recommended for their obstructive CAD counterparts.”
The study was supported by the Department of Veterans Affairs Office of Information and Analytics. Authors declared a variety of funding, directorships, and committee positions in public and private industry.
Patients with nonobstructive coronary artery disease are at significantly greater risk of myocardial infarction and all-cause mortality than are those with no CAD, and research is needed to explore risk mitigation methods in this group, say the authors of a retrospective cohort study.
The study of 37,674 U.S. veterans undergoing elective coronary angiography for CAD found patients with one-vessel nonobstructive CAD were at double the risk of a MI within 1 year, compared with those with no apparent CAD (see chart), and showed a 60% increase in mortality among individuals with three-vessel nonobstructive CAD.
“The 1-year MI risk progressively increased by CAD extent, rather than abruptly increasing between nonobstructive and obstructive CAD,” wrote Dr. Thomas M. Maddox of the Denver Veterans Affairs Medical Center and his colleagues, in an article published online Nov. 4 (JAMA 2014;312:1754-63 [doi:10.1001/jama.2014.14681]). [However] empirical evidence is lacking as to whether these patients benefit from the prevention therapies recommended for their obstructive CAD counterparts.”
The study was supported by the Department of Veterans Affairs Office of Information and Analytics. Authors declared a variety of funding, directorships, and committee positions in public and private industry.
FROM JAMA
Key clinical point: Nonobstructive coronary artery disease is associated with a significant increase in the risk of myocardial infarction and death.
Major finding: Patients with nonobstructive coronary artery disease have between 2.0 and 4.6 times the MI risk than do those without CAD.
Data source: Retrospective cohort study of 37,674 U.S. veterans undergoing elective coronary angiography for coronary artery disease.
Disclosures: The study was supported by the Department of Veterans Affairs Office of Information and Analytics. Authors declared a variety of funding, directorships, and committee positions in public and private industry.
Sargramostim addition improves survival in metastatic melanoma
The addition of sargramostim to ipilimumab therapy for unresectable stage III or IV melanoma may improve overall survival and toxicity, but it does not appear to affect progression-free survival, results from a phase II randomized trial suggest.
Patients randomized to receive ipilimumab plus sargramostim showed significantly lower mortality at 1 year, compared with those treated with ipilimumab alone (hazard ratio, 0.64), and they experienced significantly fewer grade 3 or above treatment-related toxicities, particularly gastrointestinal toxicities such as colonic perforation.
“The improved toxicity profile must be considered as contributing to the improved survival even in light of the survival advantage remaining when patients who discontinued therapy due to toxicity are excluded,” wrote Dr. F. Stephen Hodi of the Dana-Farber Cancer Institute, Boston, and colleagues.
Researchers stressed that the study, which enrolled a total of 245 patients, is ongoing, and that the findings need to be confirmed in larger studies with longer follow-up, according to a paper published online Nov. 4 (JAMA 2014 [doi:10.1001/jama.2014.13943].
The study was supported by Public Health Service grants, the Eastern Cooperative Oncology Group, the U.S. National Cancer Institute, the U.S. National Institutes of Health, the U.S. Department of Health & Human Services, Bristol-Myers Squibb, and Genzyme. Some authors reported grants, personal fees, and support from pharmaceutical companies, including Bristol-Myers Squibb and Genzyme.
The addition of sargramostim to ipilimumab therapy for unresectable stage III or IV melanoma may improve overall survival and toxicity, but it does not appear to affect progression-free survival, results from a phase II randomized trial suggest.
Patients randomized to receive ipilimumab plus sargramostim showed significantly lower mortality at 1 year, compared with those treated with ipilimumab alone (hazard ratio, 0.64), and they experienced significantly fewer grade 3 or above treatment-related toxicities, particularly gastrointestinal toxicities such as colonic perforation.
“The improved toxicity profile must be considered as contributing to the improved survival even in light of the survival advantage remaining when patients who discontinued therapy due to toxicity are excluded,” wrote Dr. F. Stephen Hodi of the Dana-Farber Cancer Institute, Boston, and colleagues.
Researchers stressed that the study, which enrolled a total of 245 patients, is ongoing, and that the findings need to be confirmed in larger studies with longer follow-up, according to a paper published online Nov. 4 (JAMA 2014 [doi:10.1001/jama.2014.13943].
The study was supported by Public Health Service grants, the Eastern Cooperative Oncology Group, the U.S. National Cancer Institute, the U.S. National Institutes of Health, the U.S. Department of Health & Human Services, Bristol-Myers Squibb, and Genzyme. Some authors reported grants, personal fees, and support from pharmaceutical companies, including Bristol-Myers Squibb and Genzyme.
The addition of sargramostim to ipilimumab therapy for unresectable stage III or IV melanoma may improve overall survival and toxicity, but it does not appear to affect progression-free survival, results from a phase II randomized trial suggest.
Patients randomized to receive ipilimumab plus sargramostim showed significantly lower mortality at 1 year, compared with those treated with ipilimumab alone (hazard ratio, 0.64), and they experienced significantly fewer grade 3 or above treatment-related toxicities, particularly gastrointestinal toxicities such as colonic perforation.
“The improved toxicity profile must be considered as contributing to the improved survival even in light of the survival advantage remaining when patients who discontinued therapy due to toxicity are excluded,” wrote Dr. F. Stephen Hodi of the Dana-Farber Cancer Institute, Boston, and colleagues.
Researchers stressed that the study, which enrolled a total of 245 patients, is ongoing, and that the findings need to be confirmed in larger studies with longer follow-up, according to a paper published online Nov. 4 (JAMA 2014 [doi:10.1001/jama.2014.13943].
The study was supported by Public Health Service grants, the Eastern Cooperative Oncology Group, the U.S. National Cancer Institute, the U.S. National Institutes of Health, the U.S. Department of Health & Human Services, Bristol-Myers Squibb, and Genzyme. Some authors reported grants, personal fees, and support from pharmaceutical companies, including Bristol-Myers Squibb and Genzyme.
FROM JAMA
Key clinical point: Sargramostim plus ipilimumab improves overall survival and toxicity profile, compared with ipilimumab alone.
Major finding: Patients treated with sargramostim plus ipilimumab showed a 36% reduction in mortality at one year, compared with those on ipilimumab alone.
Data source: Randomized controlled trial in 245 patients with unresectable stage III or IV melanoma.
Disclosures: The study was supported by Public Health Service grants, the Eastern Cooperative Oncology Group, the U.S. National Cancer Institute, the U.S. National Institutes of Health, the U.S. Department of Health and Human Services, Bristol-Myers Squibb, and Genzyme. Some authors reported grants, personal fees, and support from pharmaceutical companies, including Bristol-Myers Squibb and Genzyme.
Atrial fibrillation doubles silent cerebral infarct risk
Atrial fibrillation is associated with a more than twofold increase in the risk of silent cerebral infarctions, even in patients with no history of symptomatic stroke, a systematic review and meta-analysis has found.
The analysis of 11 studies involving 5,317 adults with atrial fibrillation (AF) but no history of stroke or prosthetic valves showed a significant increase in the risk of silent cerebral infarctions (odds ratio, 2.62), independent of whether their atrial fibrillation was paroxysmal or persistent, according to meta-analysis published online Nov. 3 in the Annals of Internal Medicine [doi:10.7326/M14-0538].
“Although SCIs [silent cerebral infarctions] do not present with acute stroke symptoms, they have been reported to be associated with more than three- and twofold increases in the risk for symptomatic stroke and dementia, respectively,” wrote Dr. Shadi Kalantarian and colleagues from the Institute for Heart Vascular and Stroke Care and Massachusetts General Hospital.
“Consequently, the higher prevalence of SCI in patients with AF may put this population at a greater risk for cognitive impairment, future stroke, and disability.”
The study was funded by the Deane Institute for Integrative Research in Atrial Fibrillation and Stroke, Massachusetts General Hospital, and the Harvard Catalyst and the Harvard Clinical and Translational Science Center. Two authors declared grants and personal fees from private industry.
Atrial fibrillation is associated with a more than twofold increase in the risk of silent cerebral infarctions, even in patients with no history of symptomatic stroke, a systematic review and meta-analysis has found.
The analysis of 11 studies involving 5,317 adults with atrial fibrillation (AF) but no history of stroke or prosthetic valves showed a significant increase in the risk of silent cerebral infarctions (odds ratio, 2.62), independent of whether their atrial fibrillation was paroxysmal or persistent, according to meta-analysis published online Nov. 3 in the Annals of Internal Medicine [doi:10.7326/M14-0538].
“Although SCIs [silent cerebral infarctions] do not present with acute stroke symptoms, they have been reported to be associated with more than three- and twofold increases in the risk for symptomatic stroke and dementia, respectively,” wrote Dr. Shadi Kalantarian and colleagues from the Institute for Heart Vascular and Stroke Care and Massachusetts General Hospital.
“Consequently, the higher prevalence of SCI in patients with AF may put this population at a greater risk for cognitive impairment, future stroke, and disability.”
The study was funded by the Deane Institute for Integrative Research in Atrial Fibrillation and Stroke, Massachusetts General Hospital, and the Harvard Catalyst and the Harvard Clinical and Translational Science Center. Two authors declared grants and personal fees from private industry.
Atrial fibrillation is associated with a more than twofold increase in the risk of silent cerebral infarctions, even in patients with no history of symptomatic stroke, a systematic review and meta-analysis has found.
The analysis of 11 studies involving 5,317 adults with atrial fibrillation (AF) but no history of stroke or prosthetic valves showed a significant increase in the risk of silent cerebral infarctions (odds ratio, 2.62), independent of whether their atrial fibrillation was paroxysmal or persistent, according to meta-analysis published online Nov. 3 in the Annals of Internal Medicine [doi:10.7326/M14-0538].
“Although SCIs [silent cerebral infarctions] do not present with acute stroke symptoms, they have been reported to be associated with more than three- and twofold increases in the risk for symptomatic stroke and dementia, respectively,” wrote Dr. Shadi Kalantarian and colleagues from the Institute for Heart Vascular and Stroke Care and Massachusetts General Hospital.
“Consequently, the higher prevalence of SCI in patients with AF may put this population at a greater risk for cognitive impairment, future stroke, and disability.”
The study was funded by the Deane Institute for Integrative Research in Atrial Fibrillation and Stroke, Massachusetts General Hospital, and the Harvard Catalyst and the Harvard Clinical and Translational Science Center. Two authors declared grants and personal fees from private industry.
FROM ANNALS OF INTERNAL MEDICINE
Key clinical point: Atrial fibrillation significantly increases the risk of silent cerebral infarction even in patients with no history of stroke.
Major finding: Patients with atrial fibrillation had a more than twofold increase in the risk of silent cerebral infarction.
Data source: Systematic review and meta-analysis of 11 studies involving 5,317 patients with atrial fibrillation but no history of stroke.
Disclosures: The study was funded by the Deane Institute for Integrative Research in Atrial Fibrillation and Stroke, Massachusetts General Hospital, and the Harvard Catalyst and the Harvard Clinical and Translational Science Center. Two authors declared grants and personal fees from private industry.
Oxaliplatin noninferior to cisplatin in advanced gastric cancer
Oxaliplatin plus S-1 is as effective as cisplatin plus S-1 for the treatment of advanced gastric cancer in chemotherapy-naive patients and is associated with fewer serious adverse events, a noninferiority trial has shown.
The randomized, open-label, phase III study in 685 patients found that oxaliplatin plus S-1 achieved noninferiority in progression-free survival, compared with cisplatin plus S-1 (5.5 versus 5.4 months; hazard ratio, 1.004; 95% confidence interval, 0.840-1.199), with a similar duration of overall survival, according to a paper published online Oct. 14 (Ann. Oncol. 2014 [doi:10.1093/annonc/mdu472]).
Researchers also observed significantly more cases of grade 3 or worse leukopenia, neutropenia, anemia, febrile neutropenia, hyponatremia, and sensory neuropathy among patients treated with cisplatin plus S-1.
“Renal function is likely impaired during treatment with a cisplatin-containing regimen, even when adequate hydration to prevent renal toxicity is provided, while oxaliplatin does not affect renal function [and] these were the probable reasons underlying the favorable results of SOX [oxaliplatin plus S-1] in elderly patients or patients with renal dysfunction,” wrote Dr. Yasuhide Yamada of the National Cancer Center Hospital, Tokyo, and colleagues.
The study was funded by Yakult Honsha. Some authors declared honoraria, travel grants, consultancies, and funding from the study sponsor and other pharmaceutical companies.
Oxaliplatin plus S-1 is as effective as cisplatin plus S-1 for the treatment of advanced gastric cancer in chemotherapy-naive patients and is associated with fewer serious adverse events, a noninferiority trial has shown.
The randomized, open-label, phase III study in 685 patients found that oxaliplatin plus S-1 achieved noninferiority in progression-free survival, compared with cisplatin plus S-1 (5.5 versus 5.4 months; hazard ratio, 1.004; 95% confidence interval, 0.840-1.199), with a similar duration of overall survival, according to a paper published online Oct. 14 (Ann. Oncol. 2014 [doi:10.1093/annonc/mdu472]).
Researchers also observed significantly more cases of grade 3 or worse leukopenia, neutropenia, anemia, febrile neutropenia, hyponatremia, and sensory neuropathy among patients treated with cisplatin plus S-1.
“Renal function is likely impaired during treatment with a cisplatin-containing regimen, even when adequate hydration to prevent renal toxicity is provided, while oxaliplatin does not affect renal function [and] these were the probable reasons underlying the favorable results of SOX [oxaliplatin plus S-1] in elderly patients or patients with renal dysfunction,” wrote Dr. Yasuhide Yamada of the National Cancer Center Hospital, Tokyo, and colleagues.
The study was funded by Yakult Honsha. Some authors declared honoraria, travel grants, consultancies, and funding from the study sponsor and other pharmaceutical companies.
Oxaliplatin plus S-1 is as effective as cisplatin plus S-1 for the treatment of advanced gastric cancer in chemotherapy-naive patients and is associated with fewer serious adverse events, a noninferiority trial has shown.
The randomized, open-label, phase III study in 685 patients found that oxaliplatin plus S-1 achieved noninferiority in progression-free survival, compared with cisplatin plus S-1 (5.5 versus 5.4 months; hazard ratio, 1.004; 95% confidence interval, 0.840-1.199), with a similar duration of overall survival, according to a paper published online Oct. 14 (Ann. Oncol. 2014 [doi:10.1093/annonc/mdu472]).
Researchers also observed significantly more cases of grade 3 or worse leukopenia, neutropenia, anemia, febrile neutropenia, hyponatremia, and sensory neuropathy among patients treated with cisplatin plus S-1.
“Renal function is likely impaired during treatment with a cisplatin-containing regimen, even when adequate hydration to prevent renal toxicity is provided, while oxaliplatin does not affect renal function [and] these were the probable reasons underlying the favorable results of SOX [oxaliplatin plus S-1] in elderly patients or patients with renal dysfunction,” wrote Dr. Yasuhide Yamada of the National Cancer Center Hospital, Tokyo, and colleagues.
The study was funded by Yakult Honsha. Some authors declared honoraria, travel grants, consultancies, and funding from the study sponsor and other pharmaceutical companies.
FROM ANNALS OF ONCOLOGY
Key clinical point: Oxaliplatin plus S-1 is as effective as cisplatin plus S-1 for the treatment of advanced gastric cancer in chemotherapy-naive patients and is associated with fewer serious adverse events.
Major finding: Oxaliplatin plus S-1 achieves similar duration of progression-free survival and overall survival to cisplatin plus S-1.
Data source: Randomized, open-label, phase III study in 685 patients with chemotherapy-naive advanced gastric cancer.
Disclosures: The study was funded by Yakult Honsha. Some authors declared honoraria, travel grants, consultancies, and funding from the study sponsor and other pharmaceutical companies.