User login
ID experts urge widespread flu vaccination for 2018-2019 season
WASHINGTON – The flu vaccine may not be perfect, but it can reduce the severity of illness and curb the risk of spreading the disease to others, William Schaffner, MD, emphasized at a press conference held by the National Foundation for Infectious Diseases.
“Give the vaccine credit for softening the blow,” said Dr. Schaffner, medical director of NFID and a professor at Vanderbilt University in Nashville.
Dr. Schaffner and a panel of experts including U.S. Surgeon General Jerome M. Adams, MD, encouraged the public and the health care community to follow recommendation from the Centers for Disease Control & Prevention that everyone aged 6 months and older receive an influenza vaccine.
Dr. Schaffner shared recent data showing that complications from the flu don’t stop when the acute illness resolves. Acute influenza causes a whole-body inflammatory reaction, and consequently “there is an increased risk of heart attack and stroke during the 2-4 weeks of recovery from acute influenza,” he said. In addition, older adults who experience acute flu and are already frail may never regain their pre-flu level of function, as the flu can start a “domino effect of decline and disability.”
Despite last year’s severe flu season that included 180 deaths in children, vaccination remains the most effective protection against the flu, Dr. Adams said.
This year, between 163 million and 168 million doses of vaccine will be available in the United States. The vaccine is available in a range of settings including doctors’ offices, pharmacies, grocery stores, and workplaces, said Dr. Adams.
Flu vaccine choices this year include a return of the live-attenuated influenza vaccine (LAIV) given via nasal spray, along with the standard influenza vaccine that includes either three influenza viruses (trivalent, with two influenza A and one influenza B) or four influenza viruses (quadrivalent, with two influenza A and two influenza B). Other options are adjuvanted vaccine and high-dose vaccine for adults aged 65 years and older, and a cell-based and recombinant vaccine as alternatives to egg-based vaccines.
Dr. Adams emphasized the importance of healthy people getting vaccinated to protect the community. “All the people who died from the flu caught it from someone else,” he said.
The message to health care providers remains the same: Recommend the flu vaccine to patients at every opportunity, and lead by example and get vaccinated yourself, Dr. Adams said. He noted this year’s strategies to promote flu vaccination on social media, and encouraged clinicians to recommend the flu shot to their patients and to showcase their own shots via the #FightFlu hashtag.
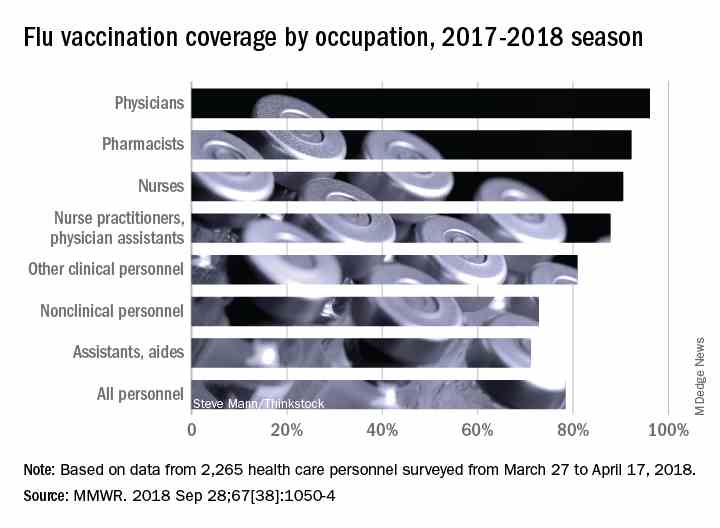
Vaccination among health care personnel last year was approximately 78%, which is a plateau over the past several years (MMWR 2018; 67:1050-54).
Be prepared to offer antivirals to patients as appropriate, and to promote the pneumococcal vaccine to eligible older adults as well, to protect not only themselves, but their contacts and the community, Dr. Adams emphasized. Currently approved antiviral drugs recommended for the 2018-2019 flu season: oseltamivir, zanamivir, and peramivir.
Wendy Sue Swanson, MD, of Seattle Children’s Hospital, stressed the importance of flu vaccination for all children, given their ability to spread viral infections. She noted a concerning 2% drop in vaccinations for children aged 6 months to 4 years, although vaccination coverage in this group was highest among children overall, at approximately 68% last season.
Last year, approximately 80% of the child deaths from flu occurred in unvaccinated children, but the vaccine has been shown to reduce the likelihood of hospitalization or death even if a child does become ill, Dr. Swanson said.
Laura E. Riley, MD, of Weill Cornell Medical Center, noted that vaccination of pregnant women has plateaued in recent years, and was 49% last year. “Our goal is 80% plus,” she said. Data show that pregnant women who received flu vaccination were 40% less likely to be hospitalized for the flu, she noted. The American College of Obstetricians and Gynecologists recommends flu vaccination as safe during any trimester, and valuable to both mothers and newborns because it provides protective antibodies during the first 6 months of life before babies can receive their own vaccinations, Dr. Riley said.
More information about this year’s flu season is available from the CDC and NFID.
WASHINGTON – The flu vaccine may not be perfect, but it can reduce the severity of illness and curb the risk of spreading the disease to others, William Schaffner, MD, emphasized at a press conference held by the National Foundation for Infectious Diseases.

“Give the vaccine credit for softening the blow,” said Dr. Schaffner, medical director of NFID and a professor at Vanderbilt University in Nashville.
Dr. Schaffner and a panel of experts including U.S. Surgeon General Jerome M. Adams, MD, encouraged the public and the health care community to follow recommendation from the Centers for Disease Control & Prevention that everyone aged 6 months and older receive an influenza vaccine.
Dr. Schaffner shared recent data showing that complications from the flu don’t stop when the acute illness resolves. Acute influenza causes a whole-body inflammatory reaction, and consequently “there is an increased risk of heart attack and stroke during the 2-4 weeks of recovery from acute influenza,” he said. In addition, older adults who experience acute flu and are already frail may never regain their pre-flu level of function, as the flu can start a “domino effect of decline and disability.”
Despite last year’s severe flu season that included 180 deaths in children, vaccination remains the most effective protection against the flu, Dr. Adams said.
This year, between 163 million and 168 million doses of vaccine will be available in the United States. The vaccine is available in a range of settings including doctors’ offices, pharmacies, grocery stores, and workplaces, said Dr. Adams.
Flu vaccine choices this year include a return of the live-attenuated influenza vaccine (LAIV) given via nasal spray, along with the standard influenza vaccine that includes either three influenza viruses (trivalent, with two influenza A and one influenza B) or four influenza viruses (quadrivalent, with two influenza A and two influenza B). Other options are adjuvanted vaccine and high-dose vaccine for adults aged 65 years and older, and a cell-based and recombinant vaccine as alternatives to egg-based vaccines.
Dr. Adams emphasized the importance of healthy people getting vaccinated to protect the community. “All the people who died from the flu caught it from someone else,” he said.
The message to health care providers remains the same: Recommend the flu vaccine to patients at every opportunity, and lead by example and get vaccinated yourself, Dr. Adams said. He noted this year’s strategies to promote flu vaccination on social media, and encouraged clinicians to recommend the flu shot to their patients and to showcase their own shots via the #FightFlu hashtag.
Vaccination among health care personnel last year was approximately 78%, which is a plateau over the past several years (MMWR 2018; 67:1050-54).
Be prepared to offer antivirals to patients as appropriate, and to promote the pneumococcal vaccine to eligible older adults as well, to protect not only themselves, but their contacts and the community, Dr. Adams emphasized. Currently approved antiviral drugs recommended for the 2018-2019 flu season: oseltamivir, zanamivir, and peramivir.
Wendy Sue Swanson, MD, of Seattle Children’s Hospital, stressed the importance of flu vaccination for all children, given their ability to spread viral infections. She noted a concerning 2% drop in vaccinations for children aged 6 months to 4 years, although vaccination coverage in this group was highest among children overall, at approximately 68% last season.
Last year, approximately 80% of the child deaths from flu occurred in unvaccinated children, but the vaccine has been shown to reduce the likelihood of hospitalization or death even if a child does become ill, Dr. Swanson said.
Laura E. Riley, MD, of Weill Cornell Medical Center, noted that vaccination of pregnant women has plateaued in recent years, and was 49% last year. “Our goal is 80% plus,” she said. Data show that pregnant women who received flu vaccination were 40% less likely to be hospitalized for the flu, she noted. The American College of Obstetricians and Gynecologists recommends flu vaccination as safe during any trimester, and valuable to both mothers and newborns because it provides protective antibodies during the first 6 months of life before babies can receive their own vaccinations, Dr. Riley said.
More information about this year’s flu season is available from the CDC and NFID.
WASHINGTON – The flu vaccine may not be perfect, but it can reduce the severity of illness and curb the risk of spreading the disease to others, William Schaffner, MD, emphasized at a press conference held by the National Foundation for Infectious Diseases.

“Give the vaccine credit for softening the blow,” said Dr. Schaffner, medical director of NFID and a professor at Vanderbilt University in Nashville.
Dr. Schaffner and a panel of experts including U.S. Surgeon General Jerome M. Adams, MD, encouraged the public and the health care community to follow recommendation from the Centers for Disease Control & Prevention that everyone aged 6 months and older receive an influenza vaccine.
Dr. Schaffner shared recent data showing that complications from the flu don’t stop when the acute illness resolves. Acute influenza causes a whole-body inflammatory reaction, and consequently “there is an increased risk of heart attack and stroke during the 2-4 weeks of recovery from acute influenza,” he said. In addition, older adults who experience acute flu and are already frail may never regain their pre-flu level of function, as the flu can start a “domino effect of decline and disability.”
Despite last year’s severe flu season that included 180 deaths in children, vaccination remains the most effective protection against the flu, Dr. Adams said.
This year, between 163 million and 168 million doses of vaccine will be available in the United States. The vaccine is available in a range of settings including doctors’ offices, pharmacies, grocery stores, and workplaces, said Dr. Adams.
Flu vaccine choices this year include a return of the live-attenuated influenza vaccine (LAIV) given via nasal spray, along with the standard influenza vaccine that includes either three influenza viruses (trivalent, with two influenza A and one influenza B) or four influenza viruses (quadrivalent, with two influenza A and two influenza B). Other options are adjuvanted vaccine and high-dose vaccine for adults aged 65 years and older, and a cell-based and recombinant vaccine as alternatives to egg-based vaccines.
Dr. Adams emphasized the importance of healthy people getting vaccinated to protect the community. “All the people who died from the flu caught it from someone else,” he said.
The message to health care providers remains the same: Recommend the flu vaccine to patients at every opportunity, and lead by example and get vaccinated yourself, Dr. Adams said. He noted this year’s strategies to promote flu vaccination on social media, and encouraged clinicians to recommend the flu shot to their patients and to showcase their own shots via the #FightFlu hashtag.
Vaccination among health care personnel last year was approximately 78%, which is a plateau over the past several years (MMWR 2018; 67:1050-54).
Be prepared to offer antivirals to patients as appropriate, and to promote the pneumococcal vaccine to eligible older adults as well, to protect not only themselves, but their contacts and the community, Dr. Adams emphasized. Currently approved antiviral drugs recommended for the 2018-2019 flu season: oseltamivir, zanamivir, and peramivir.
Wendy Sue Swanson, MD, of Seattle Children’s Hospital, stressed the importance of flu vaccination for all children, given their ability to spread viral infections. She noted a concerning 2% drop in vaccinations for children aged 6 months to 4 years, although vaccination coverage in this group was highest among children overall, at approximately 68% last season.
Last year, approximately 80% of the child deaths from flu occurred in unvaccinated children, but the vaccine has been shown to reduce the likelihood of hospitalization or death even if a child does become ill, Dr. Swanson said.
Laura E. Riley, MD, of Weill Cornell Medical Center, noted that vaccination of pregnant women has plateaued in recent years, and was 49% last year. “Our goal is 80% plus,” she said. Data show that pregnant women who received flu vaccination were 40% less likely to be hospitalized for the flu, she noted. The American College of Obstetricians and Gynecologists recommends flu vaccination as safe during any trimester, and valuable to both mothers and newborns because it provides protective antibodies during the first 6 months of life before babies can receive their own vaccinations, Dr. Riley said.
More information about this year’s flu season is available from the CDC and NFID.
FROM AN NFID PRESS CONFERENCE
Gestational weight outside guidelines adversely affects mothers, babies
Gestational weight gain above or below the level recommended by the Institute of Medicine (IOM) guidelines resulted in significantly worse outcomes for mothers and babies, according to data from nearly 30,000 women.
Previous studies of the relationship between gestational weight gain and maternal and neonatal outcomes have been limited by “small sample sizes, single sites, restricted reporting of outcomes, and a lack of racial-ethnic diversity,” Michelle A. Kominiarek, MD, of Northwestern University in Chicago and her colleagues wrote . To determine the effects of gestational weight gain on a large and more diverse population, the researchers conducted a secondary analysis of the Eunice Kennedy Shriver National Institute of Child Health and Human Development (NICHD) Maternal-Fetal Medicine Units Network’s Assessment of Perinatal Excellence study. The findings were published in Obstetrics & Gynecology.
Gestational weight gain above the amount recommended by IOM guidelines was significantly associated with adverse outcomes in neonates, including macrosomia (adjusted odds ratio, 2.66), shoulder dystocia (aOR, 1.74), and neonatal hypoglycemia (aOR, 1.60).
In further multivariate analysis, adverse maternal outcomes associated with gestational weight gain above that recommended by the guidelines included hypertensive diseases of pregnancy for any parity (aOR, 1.84) and increased risk of cesarean delivery in nulliparous and multiparous women (aORs, 1.44 and 1.26, respectively).
Gestational weight gain below the recommended amount was associated with both spontaneous (aOR, 1.50) and indicated (aOR, 1.34) preterm birth. Weight gain above the guidelines was associated with a greater risk of indicated preterm birth only (aOR, 1.24).
The study population included 29,861 women at 25 hospitals over a 3-year period. Of these, 51% had gestational weight gains above the amount recommended by the IOM guidelines and 21% had gestational weight gains below it. The researchers calculated gestational weight gain by subtracting prepregnancy weight from delivery weight or, if prepregnancy weight was not available, by subtracting weight at the first prenatal visit at 13 weeks of gestation or earlier from delivery weight.
The study findings were limited by the use of self-reported prepregnancy weight and the possible effects of changes to the guidelines with respect to obese patients, the researchers said. However, the results support those from previous studies, and the “noted strengths include analysis of 29,861 women representative of the United States with rigorous ascertainment of outcomes and calculation of gestational weight gain to account for the wide range of gestational ages at delivery,” Dr. Kominiarek and her associates wrote.
Overall, the data support efforts to educate women on health behaviors and how gestational weight gain affects them and their infants, and additional research is needed to help women meet their goals for appropriate gestational weight, the researchers concluded.
SOURCE: Kominiarek MA et al. Obstet Gynecol. 2018 Oct;132(4):875-81.
“We are struggling with an obesity epidemic in this country, and pregnancy accounts for a risk time for women to gain excessive weight,” Martina L. Badell, MD, said in an interview. “This is a very well-designed large study which attempted to systematically evaluate the adverse perinatal outcomes associated with inappropriate weight gain in pregnancy across a diverse group of women.”
She emphasized that “the take home message is the importance of counseling regarding weight gain in pregnancy and monitoring it closely in real time as the associated risks are significant and potentially avoidable. The first step to solving a problem is adequately quantifying it, and this study does just that. The next step is giving this information to pregnant women along with making weight gain a part of the discussion prior to pregnancy and at every prenatal visit.”
Dr. Badell added, “Ideally, the weight gain for an individual pregnant women would be tracked and discussed with her during each prenatal visit. If she is below or above the recommendations, the risks associated with this could be discussed along with strategies to get/stay on track. In an ideal world, women struggling with weight gain goals in pregnancy would have access to a dietitian. However, in reality, ob.gyn. offices will likely need to come up with patient education handouts or staff education.”
Another useful avenue for research would be assessing the effects of patient education, Dr. Badell said. “The next best step would be implementing a study to assess if education of women during pregnancy about their individual weight gain at each visit and discussion regarding perinatal risks affects ultimate weight gain and reduces risks. Additionally, education could begin in the preconception phase as this knowledge is likely important even prior to pregnancy. Finally, studies are needed on interventions such as working with dietitians or patient education classes once a woman has been identified as not being within weight gain goals to evaluate if these can alter weight gain and improve outcomes.”
Dr. Badell is a maternal-fetal medicine specialist in the department of gynecology and obstetrics at Emory University, Atlanta. She was asked to comment on the findings of Kominiarek MA et al. Dr. Badell had no relevant financial conflicts to disclose.
“We are struggling with an obesity epidemic in this country, and pregnancy accounts for a risk time for women to gain excessive weight,” Martina L. Badell, MD, said in an interview. “This is a very well-designed large study which attempted to systematically evaluate the adverse perinatal outcomes associated with inappropriate weight gain in pregnancy across a diverse group of women.”
She emphasized that “the take home message is the importance of counseling regarding weight gain in pregnancy and monitoring it closely in real time as the associated risks are significant and potentially avoidable. The first step to solving a problem is adequately quantifying it, and this study does just that. The next step is giving this information to pregnant women along with making weight gain a part of the discussion prior to pregnancy and at every prenatal visit.”
Dr. Badell added, “Ideally, the weight gain for an individual pregnant women would be tracked and discussed with her during each prenatal visit. If she is below or above the recommendations, the risks associated with this could be discussed along with strategies to get/stay on track. In an ideal world, women struggling with weight gain goals in pregnancy would have access to a dietitian. However, in reality, ob.gyn. offices will likely need to come up with patient education handouts or staff education.”
Another useful avenue for research would be assessing the effects of patient education, Dr. Badell said. “The next best step would be implementing a study to assess if education of women during pregnancy about their individual weight gain at each visit and discussion regarding perinatal risks affects ultimate weight gain and reduces risks. Additionally, education could begin in the preconception phase as this knowledge is likely important even prior to pregnancy. Finally, studies are needed on interventions such as working with dietitians or patient education classes once a woman has been identified as not being within weight gain goals to evaluate if these can alter weight gain and improve outcomes.”
Dr. Badell is a maternal-fetal medicine specialist in the department of gynecology and obstetrics at Emory University, Atlanta. She was asked to comment on the findings of Kominiarek MA et al. Dr. Badell had no relevant financial conflicts to disclose.
“We are struggling with an obesity epidemic in this country, and pregnancy accounts for a risk time for women to gain excessive weight,” Martina L. Badell, MD, said in an interview. “This is a very well-designed large study which attempted to systematically evaluate the adverse perinatal outcomes associated with inappropriate weight gain in pregnancy across a diverse group of women.”
She emphasized that “the take home message is the importance of counseling regarding weight gain in pregnancy and monitoring it closely in real time as the associated risks are significant and potentially avoidable. The first step to solving a problem is adequately quantifying it, and this study does just that. The next step is giving this information to pregnant women along with making weight gain a part of the discussion prior to pregnancy and at every prenatal visit.”
Dr. Badell added, “Ideally, the weight gain for an individual pregnant women would be tracked and discussed with her during each prenatal visit. If she is below or above the recommendations, the risks associated with this could be discussed along with strategies to get/stay on track. In an ideal world, women struggling with weight gain goals in pregnancy would have access to a dietitian. However, in reality, ob.gyn. offices will likely need to come up with patient education handouts or staff education.”
Another useful avenue for research would be assessing the effects of patient education, Dr. Badell said. “The next best step would be implementing a study to assess if education of women during pregnancy about their individual weight gain at each visit and discussion regarding perinatal risks affects ultimate weight gain and reduces risks. Additionally, education could begin in the preconception phase as this knowledge is likely important even prior to pregnancy. Finally, studies are needed on interventions such as working with dietitians or patient education classes once a woman has been identified as not being within weight gain goals to evaluate if these can alter weight gain and improve outcomes.”
Dr. Badell is a maternal-fetal medicine specialist in the department of gynecology and obstetrics at Emory University, Atlanta. She was asked to comment on the findings of Kominiarek MA et al. Dr. Badell had no relevant financial conflicts to disclose.
Gestational weight gain above or below the level recommended by the Institute of Medicine (IOM) guidelines resulted in significantly worse outcomes for mothers and babies, according to data from nearly 30,000 women.
Previous studies of the relationship between gestational weight gain and maternal and neonatal outcomes have been limited by “small sample sizes, single sites, restricted reporting of outcomes, and a lack of racial-ethnic diversity,” Michelle A. Kominiarek, MD, of Northwestern University in Chicago and her colleagues wrote . To determine the effects of gestational weight gain on a large and more diverse population, the researchers conducted a secondary analysis of the Eunice Kennedy Shriver National Institute of Child Health and Human Development (NICHD) Maternal-Fetal Medicine Units Network’s Assessment of Perinatal Excellence study. The findings were published in Obstetrics & Gynecology.
Gestational weight gain above the amount recommended by IOM guidelines was significantly associated with adverse outcomes in neonates, including macrosomia (adjusted odds ratio, 2.66), shoulder dystocia (aOR, 1.74), and neonatal hypoglycemia (aOR, 1.60).
In further multivariate analysis, adverse maternal outcomes associated with gestational weight gain above that recommended by the guidelines included hypertensive diseases of pregnancy for any parity (aOR, 1.84) and increased risk of cesarean delivery in nulliparous and multiparous women (aORs, 1.44 and 1.26, respectively).
Gestational weight gain below the recommended amount was associated with both spontaneous (aOR, 1.50) and indicated (aOR, 1.34) preterm birth. Weight gain above the guidelines was associated with a greater risk of indicated preterm birth only (aOR, 1.24).
The study population included 29,861 women at 25 hospitals over a 3-year period. Of these, 51% had gestational weight gains above the amount recommended by the IOM guidelines and 21% had gestational weight gains below it. The researchers calculated gestational weight gain by subtracting prepregnancy weight from delivery weight or, if prepregnancy weight was not available, by subtracting weight at the first prenatal visit at 13 weeks of gestation or earlier from delivery weight.
The study findings were limited by the use of self-reported prepregnancy weight and the possible effects of changes to the guidelines with respect to obese patients, the researchers said. However, the results support those from previous studies, and the “noted strengths include analysis of 29,861 women representative of the United States with rigorous ascertainment of outcomes and calculation of gestational weight gain to account for the wide range of gestational ages at delivery,” Dr. Kominiarek and her associates wrote.
Overall, the data support efforts to educate women on health behaviors and how gestational weight gain affects them and their infants, and additional research is needed to help women meet their goals for appropriate gestational weight, the researchers concluded.
SOURCE: Kominiarek MA et al. Obstet Gynecol. 2018 Oct;132(4):875-81.
Gestational weight gain above or below the level recommended by the Institute of Medicine (IOM) guidelines resulted in significantly worse outcomes for mothers and babies, according to data from nearly 30,000 women.
Previous studies of the relationship between gestational weight gain and maternal and neonatal outcomes have been limited by “small sample sizes, single sites, restricted reporting of outcomes, and a lack of racial-ethnic diversity,” Michelle A. Kominiarek, MD, of Northwestern University in Chicago and her colleagues wrote . To determine the effects of gestational weight gain on a large and more diverse population, the researchers conducted a secondary analysis of the Eunice Kennedy Shriver National Institute of Child Health and Human Development (NICHD) Maternal-Fetal Medicine Units Network’s Assessment of Perinatal Excellence study. The findings were published in Obstetrics & Gynecology.
Gestational weight gain above the amount recommended by IOM guidelines was significantly associated with adverse outcomes in neonates, including macrosomia (adjusted odds ratio, 2.66), shoulder dystocia (aOR, 1.74), and neonatal hypoglycemia (aOR, 1.60).
In further multivariate analysis, adverse maternal outcomes associated with gestational weight gain above that recommended by the guidelines included hypertensive diseases of pregnancy for any parity (aOR, 1.84) and increased risk of cesarean delivery in nulliparous and multiparous women (aORs, 1.44 and 1.26, respectively).
Gestational weight gain below the recommended amount was associated with both spontaneous (aOR, 1.50) and indicated (aOR, 1.34) preterm birth. Weight gain above the guidelines was associated with a greater risk of indicated preterm birth only (aOR, 1.24).
The study population included 29,861 women at 25 hospitals over a 3-year period. Of these, 51% had gestational weight gains above the amount recommended by the IOM guidelines and 21% had gestational weight gains below it. The researchers calculated gestational weight gain by subtracting prepregnancy weight from delivery weight or, if prepregnancy weight was not available, by subtracting weight at the first prenatal visit at 13 weeks of gestation or earlier from delivery weight.
The study findings were limited by the use of self-reported prepregnancy weight and the possible effects of changes to the guidelines with respect to obese patients, the researchers said. However, the results support those from previous studies, and the “noted strengths include analysis of 29,861 women representative of the United States with rigorous ascertainment of outcomes and calculation of gestational weight gain to account for the wide range of gestational ages at delivery,” Dr. Kominiarek and her associates wrote.
Overall, the data support efforts to educate women on health behaviors and how gestational weight gain affects them and their infants, and additional research is needed to help women meet their goals for appropriate gestational weight, the researchers concluded.
SOURCE: Kominiarek MA et al. Obstet Gynecol. 2018 Oct;132(4):875-81.
FROM OBSTETRICS & GYNECOLOGY
Key clinical point: Gestational weight gain or loss is a significant risk factor for adverse maternal and neonatal outcomes.
Major finding: Gestational weight gain above the recommended amount was significantly associated with adverse outcomes in neonates, including macrosomia (adjusted odds ratio, 2.66), shoulder dystocia (aOR, 1.74), and neonatal hypoglycemia (aOR, 1.60).
Study details: The data came from 29,861 women who delivered at 25 hospitals across the United States on randomly selected days during a 3-year period.
Disclosures: The researchers had no financial conflicts to disclose. The study was supported in part by various grants from the Eunice Kennedy Shriver National Institute of Child Health and Human Development and the National Center for Research Resources.
Source: Kominiarek MA et al. Obstet Gynecol. 2018 Oct;132(4):875-81.
FDA review supports Nuplazid’s safety
Pimavanserin (Nuplazid) remains an acceptable treatment for the hallucinations and delusions associated with Parkinson’s disease, according to a statement issued by the Food and Drug Administration after the agency conducted a postmarketing review of deaths and serious adverse events associated with the drug.
“Based on an analysis of all available data, FDA did not identify any new or unexpected safety findings” associated with the drug, according to the Sept. 20 statement.
However, the FDA researchers identified prescribing patterns that might increase the risk of serious adverse events, such as the concomitant use of pimavanserin and other antipsychotic drugs or drugs that can cause QT prolongation. The QT prolongation risk is listed on the drug label, which also includes a Boxed Warning about increased mortality risk in elderly patients.
The FDA statement reminds clinicians to know the risks described in the label and to be aware that no other antipsychotics are currently approved for psychosis in Parkinson’s patients.
The review was prompted by the number of reports of serious adverse events and deaths associated with pimavanserin, based on data obtained from multiple sources including the FDA Adverse Event Reporting System (FAERS), drug utilization data, safety data from the new drug application, the sponsor’s Periodic Adverse Drug Experience Reports, the sponsor’s analysis of fatal adverse event reports, and data from published medical literature.
When conducting the review, the FDA considered several factors, including the fact that Parkinson’s disease patients have higher mortality in general because of older age, advanced disease, and other medical comorbidities. In addition, pimavanserin adverse events and deaths are more likely to be reported because the drug is distributed mainly through a patient-support program and specialty pharmacy. The FDA also found no pattern suggestive of a drug effect on causes of death in patients whose deaths were reported through FAERS.
“Overall, the postmarketing data were consistent with the safety data obtained from the premarketing controlled clinical trials of Nuplazid for Parkinson’s disease psychosis,” according to the FDA statement.
Pimavanserin (Nuplazid) remains an acceptable treatment for the hallucinations and delusions associated with Parkinson’s disease, according to a statement issued by the Food and Drug Administration after the agency conducted a postmarketing review of deaths and serious adverse events associated with the drug.
“Based on an analysis of all available data, FDA did not identify any new or unexpected safety findings” associated with the drug, according to the Sept. 20 statement.
However, the FDA researchers identified prescribing patterns that might increase the risk of serious adverse events, such as the concomitant use of pimavanserin and other antipsychotic drugs or drugs that can cause QT prolongation. The QT prolongation risk is listed on the drug label, which also includes a Boxed Warning about increased mortality risk in elderly patients.
The FDA statement reminds clinicians to know the risks described in the label and to be aware that no other antipsychotics are currently approved for psychosis in Parkinson’s patients.
The review was prompted by the number of reports of serious adverse events and deaths associated with pimavanserin, based on data obtained from multiple sources including the FDA Adverse Event Reporting System (FAERS), drug utilization data, safety data from the new drug application, the sponsor’s Periodic Adverse Drug Experience Reports, the sponsor’s analysis of fatal adverse event reports, and data from published medical literature.
When conducting the review, the FDA considered several factors, including the fact that Parkinson’s disease patients have higher mortality in general because of older age, advanced disease, and other medical comorbidities. In addition, pimavanserin adverse events and deaths are more likely to be reported because the drug is distributed mainly through a patient-support program and specialty pharmacy. The FDA also found no pattern suggestive of a drug effect on causes of death in patients whose deaths were reported through FAERS.
“Overall, the postmarketing data were consistent with the safety data obtained from the premarketing controlled clinical trials of Nuplazid for Parkinson’s disease psychosis,” according to the FDA statement.
Pimavanserin (Nuplazid) remains an acceptable treatment for the hallucinations and delusions associated with Parkinson’s disease, according to a statement issued by the Food and Drug Administration after the agency conducted a postmarketing review of deaths and serious adverse events associated with the drug.
“Based on an analysis of all available data, FDA did not identify any new or unexpected safety findings” associated with the drug, according to the Sept. 20 statement.
However, the FDA researchers identified prescribing patterns that might increase the risk of serious adverse events, such as the concomitant use of pimavanserin and other antipsychotic drugs or drugs that can cause QT prolongation. The QT prolongation risk is listed on the drug label, which also includes a Boxed Warning about increased mortality risk in elderly patients.
The FDA statement reminds clinicians to know the risks described in the label and to be aware that no other antipsychotics are currently approved for psychosis in Parkinson’s patients.
The review was prompted by the number of reports of serious adverse events and deaths associated with pimavanserin, based on data obtained from multiple sources including the FDA Adverse Event Reporting System (FAERS), drug utilization data, safety data from the new drug application, the sponsor’s Periodic Adverse Drug Experience Reports, the sponsor’s analysis of fatal adverse event reports, and data from published medical literature.
When conducting the review, the FDA considered several factors, including the fact that Parkinson’s disease patients have higher mortality in general because of older age, advanced disease, and other medical comorbidities. In addition, pimavanserin adverse events and deaths are more likely to be reported because the drug is distributed mainly through a patient-support program and specialty pharmacy. The FDA also found no pattern suggestive of a drug effect on causes of death in patients whose deaths were reported through FAERS.
“Overall, the postmarketing data were consistent with the safety data obtained from the premarketing controlled clinical trials of Nuplazid for Parkinson’s disease psychosis,” according to the FDA statement.
Opioid use predicts longer sleep latency in chronic pain patients
Patients with chronic pain who took opioids reported significantly more difficulty falling asleep, compared with those who didn’t use opioids, a study of 144 adults has found.
“Identification of factors that influence insomnia symptoms among adults with chronic pain may inform prevention and treatment efforts for both disorders,” wrote Mary Beth Miller, PhD, of the University of Missouri School of Medicine, Columbia, and her colleagues.
To identify the potential impact of opioid use on sleep among chronic pain patients, the researchers recruited adults reporting symptoms of both insomnia and fibromyalgia. The average age of the participants was 52 years, and 95% were women. The study findings were published in Sleep Medicine.
The participants completed sleep diaries for 14 days, during which they recorded data including when they went to bed, how long it took them to fall asleep (SOL), how often they woke up during the night and how long they stayed awake (WASO), what time they woke up, and what time they got out of bed. Patients also reported “yes” or “no” on opioid use and their dosage each day and rated their pain on a scale of 0-100 each night before retiring.
The study participants wore wrist actigraphy devices for baseline assessment and underwent 1 night of ambulatory polysomnography.
The researchers used a multiple regression model to examine how pain intensity affected the association between opioid use and insomnia.
Overall, “opioid use was not associated with improvements in insomnia symptoms across any level of pain intensity, and was associated with worse insomnia symptoms among those reporting less intense pain,” the researchers said.
Opioid use was associated with significantly longer time to sleep onset in participants with low levels of pain (P = .02) but not among those with moderate to high levels; average sleep onset latency appeared unaffected by pain level among participants who did not use opioids.
The study findings were limited by several factors including the small number of male participants, the use of paper forms for the sleep diaries, which prevented confirmation of timely reporting, and the cross-sectional nature of the analysis, the researchers noted. However, from a clinical perspective, the “findings suggest that it may be important to advise patients reporting symptoms of insomnia about the risks of extending time in bed when providing them with opioid pain medication and that the use of behavioral or cognitive-behavioral treatment for insomnia may be recommended,” they said. The researchers also recommended that future studies address the longitudinal associations between opioid use and insomnia.
The researchers had no financial conflicts to disclose.
SOURCE: Miller M et al., Sleep Med. 2018; doi: 10.1016/j.sleep.2018.08.015.
Patients with chronic pain who took opioids reported significantly more difficulty falling asleep, compared with those who didn’t use opioids, a study of 144 adults has found.
“Identification of factors that influence insomnia symptoms among adults with chronic pain may inform prevention and treatment efforts for both disorders,” wrote Mary Beth Miller, PhD, of the University of Missouri School of Medicine, Columbia, and her colleagues.
To identify the potential impact of opioid use on sleep among chronic pain patients, the researchers recruited adults reporting symptoms of both insomnia and fibromyalgia. The average age of the participants was 52 years, and 95% were women. The study findings were published in Sleep Medicine.
The participants completed sleep diaries for 14 days, during which they recorded data including when they went to bed, how long it took them to fall asleep (SOL), how often they woke up during the night and how long they stayed awake (WASO), what time they woke up, and what time they got out of bed. Patients also reported “yes” or “no” on opioid use and their dosage each day and rated their pain on a scale of 0-100 each night before retiring.
The study participants wore wrist actigraphy devices for baseline assessment and underwent 1 night of ambulatory polysomnography.
The researchers used a multiple regression model to examine how pain intensity affected the association between opioid use and insomnia.
Overall, “opioid use was not associated with improvements in insomnia symptoms across any level of pain intensity, and was associated with worse insomnia symptoms among those reporting less intense pain,” the researchers said.
Opioid use was associated with significantly longer time to sleep onset in participants with low levels of pain (P = .02) but not among those with moderate to high levels; average sleep onset latency appeared unaffected by pain level among participants who did not use opioids.
The study findings were limited by several factors including the small number of male participants, the use of paper forms for the sleep diaries, which prevented confirmation of timely reporting, and the cross-sectional nature of the analysis, the researchers noted. However, from a clinical perspective, the “findings suggest that it may be important to advise patients reporting symptoms of insomnia about the risks of extending time in bed when providing them with opioid pain medication and that the use of behavioral or cognitive-behavioral treatment for insomnia may be recommended,” they said. The researchers also recommended that future studies address the longitudinal associations between opioid use and insomnia.
The researchers had no financial conflicts to disclose.
SOURCE: Miller M et al., Sleep Med. 2018; doi: 10.1016/j.sleep.2018.08.015.
Patients with chronic pain who took opioids reported significantly more difficulty falling asleep, compared with those who didn’t use opioids, a study of 144 adults has found.
“Identification of factors that influence insomnia symptoms among adults with chronic pain may inform prevention and treatment efforts for both disorders,” wrote Mary Beth Miller, PhD, of the University of Missouri School of Medicine, Columbia, and her colleagues.
To identify the potential impact of opioid use on sleep among chronic pain patients, the researchers recruited adults reporting symptoms of both insomnia and fibromyalgia. The average age of the participants was 52 years, and 95% were women. The study findings were published in Sleep Medicine.
The participants completed sleep diaries for 14 days, during which they recorded data including when they went to bed, how long it took them to fall asleep (SOL), how often they woke up during the night and how long they stayed awake (WASO), what time they woke up, and what time they got out of bed. Patients also reported “yes” or “no” on opioid use and their dosage each day and rated their pain on a scale of 0-100 each night before retiring.
The study participants wore wrist actigraphy devices for baseline assessment and underwent 1 night of ambulatory polysomnography.
The researchers used a multiple regression model to examine how pain intensity affected the association between opioid use and insomnia.
Overall, “opioid use was not associated with improvements in insomnia symptoms across any level of pain intensity, and was associated with worse insomnia symptoms among those reporting less intense pain,” the researchers said.
Opioid use was associated with significantly longer time to sleep onset in participants with low levels of pain (P = .02) but not among those with moderate to high levels; average sleep onset latency appeared unaffected by pain level among participants who did not use opioids.
The study findings were limited by several factors including the small number of male participants, the use of paper forms for the sleep diaries, which prevented confirmation of timely reporting, and the cross-sectional nature of the analysis, the researchers noted. However, from a clinical perspective, the “findings suggest that it may be important to advise patients reporting symptoms of insomnia about the risks of extending time in bed when providing them with opioid pain medication and that the use of behavioral or cognitive-behavioral treatment for insomnia may be recommended,” they said. The researchers also recommended that future studies address the longitudinal associations between opioid use and insomnia.
The researchers had no financial conflicts to disclose.
SOURCE: Miller M et al., Sleep Med. 2018; doi: 10.1016/j.sleep.2018.08.015.
FROM SLEEP MEDICINE
Key clinical point: Adults taking opioids for chronic pain reported more difficulty falling asleep, compared with those who didn’t use opioids.
Major finding: Approximately one-third of adults with chronic pain reported using an opioid.
Study details: The data come from 144 adults who reported both insomnia and fibromyalgia.
Disclosures: The researchers had no financial conflicts to disclose.
Source: Miller M et al. Sleep Medicine. 2018. doi: 10.1016/j.sleep.2018.08.015.
Task force advises behavioral intervention for obese adults
The U.S. Preventive Services Task Force advises clinicians to refer or offer intensive behavioral weight-loss interventions to obese adults, according to an updated recommendation statement published in JAMA.
Obesity affects more than one-third of U.S. adults, according to federal statistics. It carries increased risk for comorbidities including heart disease, diabetes, and various cancers, as well as increased risk of death among adults younger than 65 years, noted lead author Susan J. Curry, PhD, of the University of Iowa, Iowa City, and members of the Task Force.
The B recommendation applies to obese adults; obesity was defined as a body mass index of 30 kg/m2 or higher. The evidence review focused on interventions for weight loss and weight maintenance that could be provided in primary care or referred from primary care, such as nutrition counseling, exercise strategies, and goal setting.
The Task Force found adequate evidence that behavior-based weight-loss interventions improved weight, reduced incidence of type 2 diabetes, and helped maintain weight loss after interventions ended.
The Task Force found small to no evidence of harm associated with any of the behavioral weight-loss interventions, which included group sessions, personal sessions, print-based interventions, and technology-based interventions (such as text messages). Although interventions that combined drug therapy with behavioral intervention resulted in greater weight loss over 12-18 months, compared with behavioral interventions alone, the attrition rates were high and data on weight-loss maintenance after discontinuation of the drugs were limited, the Task Force noted.
“As a result, the USPSTF encourages clinicians to promote behavioral interventions as the primary focus of effective interventions for weight loss in adults,” they wrote.
The Task Force acknowledged the need for future research in subgroups and to explore whether factors such as genetics and untreated conditions are barriers to behavior-based weight loss interventions.
In the evidence review published in JAMA, Erin S. LeBlanc, MD, of Kaiser Permanente in Portland, Ore., and her colleagues reviewed data from 122 randomized, controlled trials including more than 62,000 persons and 2 observational studies including more than 209,000 persons.
The researchers found behavioral interventions were associated with greater weight loss and less risk of developing diabetes, compared with control interventions.
Intensive behavioral interventions included counseling patients about healthy eating, encouraging physical activity, setting weight and health goals, and assisting with weight monitoring. The interventions ranged from text messaging to in-person sessions for individuals or groups. The average absolute weight loss in the trials included in the review ranged from –0.5 kg to –9.3 kg (–1.1 lb to –20.5 lb) for intervention patients and from +1.4 kg to –5.6 kg (+3.1 lb to –12.3 lb) in controls.
Limitations of the review included a lack of data on population subgroups and a lack of long-term data on weight and health outcomes, the researchers noted. However, the results support the value of behavior-based therapy for obesity treatment.
The final recommendation is consistent with the 2018 draft recommendation and updates the 2012 final recommendation on obesity management.
The researchers and Task Force members had no relevant financial conflicts to disclose.
SOURCE: U.S. Preventive Services Task Force. JAMA. 2018;320(11):1163-71. doi: 10.1001/jama.2018.13022.
For most primary care clinicians, referring obese patients for more advanced behavioral therapy will be the most practical integration of the recommendation, Susan Z. Yanovski, MD, wrote in an accompanying editorial. Clinicians with training in motivational interviewing or counseling may help assess a patient’s readiness for treatment, but even being familiar with weight-management resources in the community can help patients find the right fit.
“Clinicians can do their patients a great service by showing respect for their patients’ struggles with weight management, screening for obesity-related comorbidities, and providing treatment for identified conditions regardless of the patient’s motivation for, or success with, weight-loss treatment,” she said.
Dr. Yanovski noted that pharmacotherapy options have increased since the 2012 recommendations, when orlistat was the only approved drug for long-term treatment of obesity. Five medications are currently available for this indication.
The USPSTF review was limited in scope for both drug and behavior therapy, noted Dr. Yanovski. “Because the recommendations are meant to apply to adults without diseases for which weight loss is part of disease management, some large and long-term clinical trials conducted among patients with type 2 diabetes or cardiovascular disease were not included.”
Another limitation was the exclusion of surgical treatments as being outside the primary care setting, but bariatric surgery remains a viable option for many patients, especially for prevention or resolution of type 2 diabetes. Primary care clinicians are in a position to identify patients who might benefit and to provide referrals to surgeons if appropriate, she wrote.
Dr. Yanovski agreed with the recommendations but concluded that early strategies to prevent obesity should not be neglected. “Research to develop effective prevention strategies throughout the life course, including infancy and early childhood, could ultimately decrease the number of adults who must confront the difficult challenge of losing excess weight.”
Dr. Yanovski is affiliated with the National Institute of Diabetes and Digestive and Kidney Diseases. She disclosed that her spouse has received research funding from Zafgen and Rhythm Pharmaceuticals for studies of investigational products to treat obesity. Her comments are summarized from an editorial accompanying the articles by Curry SJ et al. and LeBlanc ES et al. (JAMA. 2018;320[11]:1111-3).
For most primary care clinicians, referring obese patients for more advanced behavioral therapy will be the most practical integration of the recommendation, Susan Z. Yanovski, MD, wrote in an accompanying editorial. Clinicians with training in motivational interviewing or counseling may help assess a patient’s readiness for treatment, but even being familiar with weight-management resources in the community can help patients find the right fit.
“Clinicians can do their patients a great service by showing respect for their patients’ struggles with weight management, screening for obesity-related comorbidities, and providing treatment for identified conditions regardless of the patient’s motivation for, or success with, weight-loss treatment,” she said.
Dr. Yanovski noted that pharmacotherapy options have increased since the 2012 recommendations, when orlistat was the only approved drug for long-term treatment of obesity. Five medications are currently available for this indication.
The USPSTF review was limited in scope for both drug and behavior therapy, noted Dr. Yanovski. “Because the recommendations are meant to apply to adults without diseases for which weight loss is part of disease management, some large and long-term clinical trials conducted among patients with type 2 diabetes or cardiovascular disease were not included.”
Another limitation was the exclusion of surgical treatments as being outside the primary care setting, but bariatric surgery remains a viable option for many patients, especially for prevention or resolution of type 2 diabetes. Primary care clinicians are in a position to identify patients who might benefit and to provide referrals to surgeons if appropriate, she wrote.
Dr. Yanovski agreed with the recommendations but concluded that early strategies to prevent obesity should not be neglected. “Research to develop effective prevention strategies throughout the life course, including infancy and early childhood, could ultimately decrease the number of adults who must confront the difficult challenge of losing excess weight.”
Dr. Yanovski is affiliated with the National Institute of Diabetes and Digestive and Kidney Diseases. She disclosed that her spouse has received research funding from Zafgen and Rhythm Pharmaceuticals for studies of investigational products to treat obesity. Her comments are summarized from an editorial accompanying the articles by Curry SJ et al. and LeBlanc ES et al. (JAMA. 2018;320[11]:1111-3).
For most primary care clinicians, referring obese patients for more advanced behavioral therapy will be the most practical integration of the recommendation, Susan Z. Yanovski, MD, wrote in an accompanying editorial. Clinicians with training in motivational interviewing or counseling may help assess a patient’s readiness for treatment, but even being familiar with weight-management resources in the community can help patients find the right fit.
“Clinicians can do their patients a great service by showing respect for their patients’ struggles with weight management, screening for obesity-related comorbidities, and providing treatment for identified conditions regardless of the patient’s motivation for, or success with, weight-loss treatment,” she said.
Dr. Yanovski noted that pharmacotherapy options have increased since the 2012 recommendations, when orlistat was the only approved drug for long-term treatment of obesity. Five medications are currently available for this indication.
The USPSTF review was limited in scope for both drug and behavior therapy, noted Dr. Yanovski. “Because the recommendations are meant to apply to adults without diseases for which weight loss is part of disease management, some large and long-term clinical trials conducted among patients with type 2 diabetes or cardiovascular disease were not included.”
Another limitation was the exclusion of surgical treatments as being outside the primary care setting, but bariatric surgery remains a viable option for many patients, especially for prevention or resolution of type 2 diabetes. Primary care clinicians are in a position to identify patients who might benefit and to provide referrals to surgeons if appropriate, she wrote.
Dr. Yanovski agreed with the recommendations but concluded that early strategies to prevent obesity should not be neglected. “Research to develop effective prevention strategies throughout the life course, including infancy and early childhood, could ultimately decrease the number of adults who must confront the difficult challenge of losing excess weight.”
Dr. Yanovski is affiliated with the National Institute of Diabetes and Digestive and Kidney Diseases. She disclosed that her spouse has received research funding from Zafgen and Rhythm Pharmaceuticals for studies of investigational products to treat obesity. Her comments are summarized from an editorial accompanying the articles by Curry SJ et al. and LeBlanc ES et al. (JAMA. 2018;320[11]:1111-3).
The U.S. Preventive Services Task Force advises clinicians to refer or offer intensive behavioral weight-loss interventions to obese adults, according to an updated recommendation statement published in JAMA.
Obesity affects more than one-third of U.S. adults, according to federal statistics. It carries increased risk for comorbidities including heart disease, diabetes, and various cancers, as well as increased risk of death among adults younger than 65 years, noted lead author Susan J. Curry, PhD, of the University of Iowa, Iowa City, and members of the Task Force.
The B recommendation applies to obese adults; obesity was defined as a body mass index of 30 kg/m2 or higher. The evidence review focused on interventions for weight loss and weight maintenance that could be provided in primary care or referred from primary care, such as nutrition counseling, exercise strategies, and goal setting.
The Task Force found adequate evidence that behavior-based weight-loss interventions improved weight, reduced incidence of type 2 diabetes, and helped maintain weight loss after interventions ended.
The Task Force found small to no evidence of harm associated with any of the behavioral weight-loss interventions, which included group sessions, personal sessions, print-based interventions, and technology-based interventions (such as text messages). Although interventions that combined drug therapy with behavioral intervention resulted in greater weight loss over 12-18 months, compared with behavioral interventions alone, the attrition rates were high and data on weight-loss maintenance after discontinuation of the drugs were limited, the Task Force noted.
“As a result, the USPSTF encourages clinicians to promote behavioral interventions as the primary focus of effective interventions for weight loss in adults,” they wrote.
The Task Force acknowledged the need for future research in subgroups and to explore whether factors such as genetics and untreated conditions are barriers to behavior-based weight loss interventions.
In the evidence review published in JAMA, Erin S. LeBlanc, MD, of Kaiser Permanente in Portland, Ore., and her colleagues reviewed data from 122 randomized, controlled trials including more than 62,000 persons and 2 observational studies including more than 209,000 persons.
The researchers found behavioral interventions were associated with greater weight loss and less risk of developing diabetes, compared with control interventions.
Intensive behavioral interventions included counseling patients about healthy eating, encouraging physical activity, setting weight and health goals, and assisting with weight monitoring. The interventions ranged from text messaging to in-person sessions for individuals or groups. The average absolute weight loss in the trials included in the review ranged from –0.5 kg to –9.3 kg (–1.1 lb to –20.5 lb) for intervention patients and from +1.4 kg to –5.6 kg (+3.1 lb to –12.3 lb) in controls.
Limitations of the review included a lack of data on population subgroups and a lack of long-term data on weight and health outcomes, the researchers noted. However, the results support the value of behavior-based therapy for obesity treatment.
The final recommendation is consistent with the 2018 draft recommendation and updates the 2012 final recommendation on obesity management.
The researchers and Task Force members had no relevant financial conflicts to disclose.
SOURCE: U.S. Preventive Services Task Force. JAMA. 2018;320(11):1163-71. doi: 10.1001/jama.2018.13022.
The U.S. Preventive Services Task Force advises clinicians to refer or offer intensive behavioral weight-loss interventions to obese adults, according to an updated recommendation statement published in JAMA.
Obesity affects more than one-third of U.S. adults, according to federal statistics. It carries increased risk for comorbidities including heart disease, diabetes, and various cancers, as well as increased risk of death among adults younger than 65 years, noted lead author Susan J. Curry, PhD, of the University of Iowa, Iowa City, and members of the Task Force.
The B recommendation applies to obese adults; obesity was defined as a body mass index of 30 kg/m2 or higher. The evidence review focused on interventions for weight loss and weight maintenance that could be provided in primary care or referred from primary care, such as nutrition counseling, exercise strategies, and goal setting.
The Task Force found adequate evidence that behavior-based weight-loss interventions improved weight, reduced incidence of type 2 diabetes, and helped maintain weight loss after interventions ended.
The Task Force found small to no evidence of harm associated with any of the behavioral weight-loss interventions, which included group sessions, personal sessions, print-based interventions, and technology-based interventions (such as text messages). Although interventions that combined drug therapy with behavioral intervention resulted in greater weight loss over 12-18 months, compared with behavioral interventions alone, the attrition rates were high and data on weight-loss maintenance after discontinuation of the drugs were limited, the Task Force noted.
“As a result, the USPSTF encourages clinicians to promote behavioral interventions as the primary focus of effective interventions for weight loss in adults,” they wrote.
The Task Force acknowledged the need for future research in subgroups and to explore whether factors such as genetics and untreated conditions are barriers to behavior-based weight loss interventions.
In the evidence review published in JAMA, Erin S. LeBlanc, MD, of Kaiser Permanente in Portland, Ore., and her colleagues reviewed data from 122 randomized, controlled trials including more than 62,000 persons and 2 observational studies including more than 209,000 persons.
The researchers found behavioral interventions were associated with greater weight loss and less risk of developing diabetes, compared with control interventions.
Intensive behavioral interventions included counseling patients about healthy eating, encouraging physical activity, setting weight and health goals, and assisting with weight monitoring. The interventions ranged from text messaging to in-person sessions for individuals or groups. The average absolute weight loss in the trials included in the review ranged from –0.5 kg to –9.3 kg (–1.1 lb to –20.5 lb) for intervention patients and from +1.4 kg to –5.6 kg (+3.1 lb to –12.3 lb) in controls.
Limitations of the review included a lack of data on population subgroups and a lack of long-term data on weight and health outcomes, the researchers noted. However, the results support the value of behavior-based therapy for obesity treatment.
The final recommendation is consistent with the 2018 draft recommendation and updates the 2012 final recommendation on obesity management.
The researchers and Task Force members had no relevant financial conflicts to disclose.
SOURCE: U.S. Preventive Services Task Force. JAMA. 2018;320(11):1163-71. doi: 10.1001/jama.2018.13022.
FROM JAMA
One-step gestational diabetes screening doesn’t improve outcomes
according to data from a before-and-after cohort study of women in the state of Washington.
The one-step test, a 75-g 2-hour oral glucose tolerance test (OGTT), was recommended for all pregnant women in 2010, although the traditional two-step test – a 50-g screening glucose challenge test followed by a 100-g 3-hour OGTT – remains widely used, wrote Gaia Pocobelli, PhD, of Kaiser Permanente Washington Health Research Institute, Seattle, and her colleagues. “No randomized trial has been published comparing outcomes of the two approaches.”
In a study published in Obstetrics & Gynecology, the researchers compared data from 23,257 women who received prenatal care in Washington State between January 2009 and December 2014, including 8,363 women who received care before the guideline change, 4,103 who received care during a transition period, and 10,791 after the guideline change. Approximately 60% of the women received care from clinicians internal to Kaiser Permanente; 40% received care from external providers. Most (87%) of the internal clinicians switched to the one-step approach, the researchers said. Only 5% of external providers did so.
Overall, adopting the one-step approach was associated with a 41% increase in the diagnosis of GDM without improved maternal or neonatal outcomes, the researchers noted.
The incidence of GDM increased from 7% before the guideline change to 11% afterward for women seen by internal providers. For women seen by external providers, gestational diabetes incidence increased from 10% to 11%.
For women seen by internal providers, the use of insulin increased from 1% before the guideline change to 4% afterward; for women seen by external providers, use of insulin increased from 1.3% to 1.4% (change between the groups P less than .001).
In addition, women seen by internal providers were more likely to undergo induction of labor after the guideline change (25% to 29%), while labor induction decreased for women seen by external providers (31% to 29%) for a relative risk of 1.2.
Neonatal hypoglycemia increased from 1% to 2% among women seen by internal providers, but decreased slightly from 2.4% to 2.1% for women seen by external providers, for a relative risk of 1.77.
There were no significant differences between the women seen by internal and external providers in risk of primary cesarean section, large for gestational age, small for gestational age, or neonatal ICU admission.
The main limitation of the study was the potential confounding variables including maternal diet and exercise, and possible underreporting of risk factors such as smoking, the researchers noted. However, the results were strengthened by the large study population, and the results “do not suggest a benefit of adopting the one-step over the two-step approach.
“Kaiser Permanente Washington has revised [its] guidelines to return to a two-step process. We recommend that any health care system considering switching to the one-step approach incorporate a rigorous evaluation of changes in maternal and neonatal outcomes,” Dr. Pocobelli and her associates added.
Dr. Pocobelli disclosed funding from Jazz Pharmaceuticals for work unrelated to this study. The study was supported in part by a grant from the Group Health Foundation Momentum Fund.
Diabetes is a significant global public health concern, but is especially problematic for women of reproductive age because diabetes in pregnancy can cause significant health complications for the mother and baby. Gestational diabetes mellitus (GDM) affects up to 10% of pregnancies in the United States annually, and is associated with perinatal loss, operative delivery, macrosomia, hypoglycemia, respiratory distress syndrome, and metabolic derangements for the offspring. For the mother, GDM is associated with hypertensive disorders, infections, hydramnios, and increased risk for developing type 2 diabetes later in life. As the incidence of GDM continues to rise, studies examining how to reduce, manage or prevent this condition become increasingly important.
The authors’ conclusions, that adopting the one-step approach increased the number of women with diagnosed GDM but did not significantly improve maternal or neonatal outcomes, are not surprising. Since the initial publication of the Hyperglycemia and Adverse Pregnancy Outcome Study, upon which the International Association of the Diabetes in Pregnancy Study Groups based its recommendations to go to a one-step approach, much debate has ensued about the best method to diagnose GDM. Indeed, the National Institutes of Health convened a consensus panel to review the literature and determine whether the one-step approach should be universally adopted (the panel concluded that more information was needed, and that the current two-step approach should continue to be used).
As the authors concede, studies have shown conflicting results, and no large-scale randomized controlled trial has been conducted to date. However, the literature does not bear out the idea that the one-step approach is truly better. The current study, although including a significant number of women and a reasonable control group, only serves as yet another study to reinforce what has previously been published.
I would agree with the researchers’ conclusions that the one-step approach is not necessarily beneficial. Although the one-step approach may identify a subset of patients who might not otherwise be diagnosed with GDM, it still remains unclear whether the outcomes for these patients will be improved. Furthermore, additional testing, need for insulin or other oral antidiabetic medications, etc., would result in additional stress to the patient and the health care system. Based on the authors’ findings, and results of other studies, it remains to be determined if the effort (diagnosing additional patients with GDM) is justified medically, economically, or otherwise.
As ob.gyns., we must continually ask ourselves: “By not doing something, are we causing harm to our patients?” If we change the diagnostic criteria for GDM, thereby increasing the number of women with the condition who would then require additional care, medications, and, potentially, more complex decisions around timing and mode of delivery, we need to be certain that we are not doing harm. This, and other studies examining the use of the one- versus two-step approach have yet to demonstrate, unequivocally, that changing the criteria reduces harm, and, perhaps, might – unintentionally – cause more.
As the study authors and the NIH consensus panel concluded, more rigorous evaluation is needed; that is, a large, multicenter randomized controlled trial that examines not only the benefits during pregnancy but also the long-term benefits to women and their children.
E. Albert Reece, MD, PhD, MBA, who specializes in maternal-fetal medicine, is vice president for medical affairs at the University of Maryland, Baltimore, as well as the John Z. and Akiko K. Bowers Distinguished Professor and dean of the school of medicine. He provided commentary on the study by Pocobelli et al. Dr. Reece said he had no relevant financial disclosures.
Diabetes is a significant global public health concern, but is especially problematic for women of reproductive age because diabetes in pregnancy can cause significant health complications for the mother and baby. Gestational diabetes mellitus (GDM) affects up to 10% of pregnancies in the United States annually, and is associated with perinatal loss, operative delivery, macrosomia, hypoglycemia, respiratory distress syndrome, and metabolic derangements for the offspring. For the mother, GDM is associated with hypertensive disorders, infections, hydramnios, and increased risk for developing type 2 diabetes later in life. As the incidence of GDM continues to rise, studies examining how to reduce, manage or prevent this condition become increasingly important.
The authors’ conclusions, that adopting the one-step approach increased the number of women with diagnosed GDM but did not significantly improve maternal or neonatal outcomes, are not surprising. Since the initial publication of the Hyperglycemia and Adverse Pregnancy Outcome Study, upon which the International Association of the Diabetes in Pregnancy Study Groups based its recommendations to go to a one-step approach, much debate has ensued about the best method to diagnose GDM. Indeed, the National Institutes of Health convened a consensus panel to review the literature and determine whether the one-step approach should be universally adopted (the panel concluded that more information was needed, and that the current two-step approach should continue to be used).
As the authors concede, studies have shown conflicting results, and no large-scale randomized controlled trial has been conducted to date. However, the literature does not bear out the idea that the one-step approach is truly better. The current study, although including a significant number of women and a reasonable control group, only serves as yet another study to reinforce what has previously been published.
I would agree with the researchers’ conclusions that the one-step approach is not necessarily beneficial. Although the one-step approach may identify a subset of patients who might not otherwise be diagnosed with GDM, it still remains unclear whether the outcomes for these patients will be improved. Furthermore, additional testing, need for insulin or other oral antidiabetic medications, etc., would result in additional stress to the patient and the health care system. Based on the authors’ findings, and results of other studies, it remains to be determined if the effort (diagnosing additional patients with GDM) is justified medically, economically, or otherwise.
As ob.gyns., we must continually ask ourselves: “By not doing something, are we causing harm to our patients?” If we change the diagnostic criteria for GDM, thereby increasing the number of women with the condition who would then require additional care, medications, and, potentially, more complex decisions around timing and mode of delivery, we need to be certain that we are not doing harm. This, and other studies examining the use of the one- versus two-step approach have yet to demonstrate, unequivocally, that changing the criteria reduces harm, and, perhaps, might – unintentionally – cause more.
As the study authors and the NIH consensus panel concluded, more rigorous evaluation is needed; that is, a large, multicenter randomized controlled trial that examines not only the benefits during pregnancy but also the long-term benefits to women and their children.
E. Albert Reece, MD, PhD, MBA, who specializes in maternal-fetal medicine, is vice president for medical affairs at the University of Maryland, Baltimore, as well as the John Z. and Akiko K. Bowers Distinguished Professor and dean of the school of medicine. He provided commentary on the study by Pocobelli et al. Dr. Reece said he had no relevant financial disclosures.
Diabetes is a significant global public health concern, but is especially problematic for women of reproductive age because diabetes in pregnancy can cause significant health complications for the mother and baby. Gestational diabetes mellitus (GDM) affects up to 10% of pregnancies in the United States annually, and is associated with perinatal loss, operative delivery, macrosomia, hypoglycemia, respiratory distress syndrome, and metabolic derangements for the offspring. For the mother, GDM is associated with hypertensive disorders, infections, hydramnios, and increased risk for developing type 2 diabetes later in life. As the incidence of GDM continues to rise, studies examining how to reduce, manage or prevent this condition become increasingly important.
The authors’ conclusions, that adopting the one-step approach increased the number of women with diagnosed GDM but did not significantly improve maternal or neonatal outcomes, are not surprising. Since the initial publication of the Hyperglycemia and Adverse Pregnancy Outcome Study, upon which the International Association of the Diabetes in Pregnancy Study Groups based its recommendations to go to a one-step approach, much debate has ensued about the best method to diagnose GDM. Indeed, the National Institutes of Health convened a consensus panel to review the literature and determine whether the one-step approach should be universally adopted (the panel concluded that more information was needed, and that the current two-step approach should continue to be used).
As the authors concede, studies have shown conflicting results, and no large-scale randomized controlled trial has been conducted to date. However, the literature does not bear out the idea that the one-step approach is truly better. The current study, although including a significant number of women and a reasonable control group, only serves as yet another study to reinforce what has previously been published.
I would agree with the researchers’ conclusions that the one-step approach is not necessarily beneficial. Although the one-step approach may identify a subset of patients who might not otherwise be diagnosed with GDM, it still remains unclear whether the outcomes for these patients will be improved. Furthermore, additional testing, need for insulin or other oral antidiabetic medications, etc., would result in additional stress to the patient and the health care system. Based on the authors’ findings, and results of other studies, it remains to be determined if the effort (diagnosing additional patients with GDM) is justified medically, economically, or otherwise.
As ob.gyns., we must continually ask ourselves: “By not doing something, are we causing harm to our patients?” If we change the diagnostic criteria for GDM, thereby increasing the number of women with the condition who would then require additional care, medications, and, potentially, more complex decisions around timing and mode of delivery, we need to be certain that we are not doing harm. This, and other studies examining the use of the one- versus two-step approach have yet to demonstrate, unequivocally, that changing the criteria reduces harm, and, perhaps, might – unintentionally – cause more.
As the study authors and the NIH consensus panel concluded, more rigorous evaluation is needed; that is, a large, multicenter randomized controlled trial that examines not only the benefits during pregnancy but also the long-term benefits to women and their children.
E. Albert Reece, MD, PhD, MBA, who specializes in maternal-fetal medicine, is vice president for medical affairs at the University of Maryland, Baltimore, as well as the John Z. and Akiko K. Bowers Distinguished Professor and dean of the school of medicine. He provided commentary on the study by Pocobelli et al. Dr. Reece said he had no relevant financial disclosures.
according to data from a before-and-after cohort study of women in the state of Washington.
The one-step test, a 75-g 2-hour oral glucose tolerance test (OGTT), was recommended for all pregnant women in 2010, although the traditional two-step test – a 50-g screening glucose challenge test followed by a 100-g 3-hour OGTT – remains widely used, wrote Gaia Pocobelli, PhD, of Kaiser Permanente Washington Health Research Institute, Seattle, and her colleagues. “No randomized trial has been published comparing outcomes of the two approaches.”
In a study published in Obstetrics & Gynecology, the researchers compared data from 23,257 women who received prenatal care in Washington State between January 2009 and December 2014, including 8,363 women who received care before the guideline change, 4,103 who received care during a transition period, and 10,791 after the guideline change. Approximately 60% of the women received care from clinicians internal to Kaiser Permanente; 40% received care from external providers. Most (87%) of the internal clinicians switched to the one-step approach, the researchers said. Only 5% of external providers did so.
Overall, adopting the one-step approach was associated with a 41% increase in the diagnosis of GDM without improved maternal or neonatal outcomes, the researchers noted.
The incidence of GDM increased from 7% before the guideline change to 11% afterward for women seen by internal providers. For women seen by external providers, gestational diabetes incidence increased from 10% to 11%.
For women seen by internal providers, the use of insulin increased from 1% before the guideline change to 4% afterward; for women seen by external providers, use of insulin increased from 1.3% to 1.4% (change between the groups P less than .001).
In addition, women seen by internal providers were more likely to undergo induction of labor after the guideline change (25% to 29%), while labor induction decreased for women seen by external providers (31% to 29%) for a relative risk of 1.2.
Neonatal hypoglycemia increased from 1% to 2% among women seen by internal providers, but decreased slightly from 2.4% to 2.1% for women seen by external providers, for a relative risk of 1.77.
There were no significant differences between the women seen by internal and external providers in risk of primary cesarean section, large for gestational age, small for gestational age, or neonatal ICU admission.
The main limitation of the study was the potential confounding variables including maternal diet and exercise, and possible underreporting of risk factors such as smoking, the researchers noted. However, the results were strengthened by the large study population, and the results “do not suggest a benefit of adopting the one-step over the two-step approach.
“Kaiser Permanente Washington has revised [its] guidelines to return to a two-step process. We recommend that any health care system considering switching to the one-step approach incorporate a rigorous evaluation of changes in maternal and neonatal outcomes,” Dr. Pocobelli and her associates added.
Dr. Pocobelli disclosed funding from Jazz Pharmaceuticals for work unrelated to this study. The study was supported in part by a grant from the Group Health Foundation Momentum Fund.
according to data from a before-and-after cohort study of women in the state of Washington.
The one-step test, a 75-g 2-hour oral glucose tolerance test (OGTT), was recommended for all pregnant women in 2010, although the traditional two-step test – a 50-g screening glucose challenge test followed by a 100-g 3-hour OGTT – remains widely used, wrote Gaia Pocobelli, PhD, of Kaiser Permanente Washington Health Research Institute, Seattle, and her colleagues. “No randomized trial has been published comparing outcomes of the two approaches.”
In a study published in Obstetrics & Gynecology, the researchers compared data from 23,257 women who received prenatal care in Washington State between January 2009 and December 2014, including 8,363 women who received care before the guideline change, 4,103 who received care during a transition period, and 10,791 after the guideline change. Approximately 60% of the women received care from clinicians internal to Kaiser Permanente; 40% received care from external providers. Most (87%) of the internal clinicians switched to the one-step approach, the researchers said. Only 5% of external providers did so.
Overall, adopting the one-step approach was associated with a 41% increase in the diagnosis of GDM without improved maternal or neonatal outcomes, the researchers noted.
The incidence of GDM increased from 7% before the guideline change to 11% afterward for women seen by internal providers. For women seen by external providers, gestational diabetes incidence increased from 10% to 11%.
For women seen by internal providers, the use of insulin increased from 1% before the guideline change to 4% afterward; for women seen by external providers, use of insulin increased from 1.3% to 1.4% (change between the groups P less than .001).
In addition, women seen by internal providers were more likely to undergo induction of labor after the guideline change (25% to 29%), while labor induction decreased for women seen by external providers (31% to 29%) for a relative risk of 1.2.
Neonatal hypoglycemia increased from 1% to 2% among women seen by internal providers, but decreased slightly from 2.4% to 2.1% for women seen by external providers, for a relative risk of 1.77.
There were no significant differences between the women seen by internal and external providers in risk of primary cesarean section, large for gestational age, small for gestational age, or neonatal ICU admission.
The main limitation of the study was the potential confounding variables including maternal diet and exercise, and possible underreporting of risk factors such as smoking, the researchers noted. However, the results were strengthened by the large study population, and the results “do not suggest a benefit of adopting the one-step over the two-step approach.
“Kaiser Permanente Washington has revised [its] guidelines to return to a two-step process. We recommend that any health care system considering switching to the one-step approach incorporate a rigorous evaluation of changes in maternal and neonatal outcomes,” Dr. Pocobelli and her associates added.
Dr. Pocobelli disclosed funding from Jazz Pharmaceuticals for work unrelated to this study. The study was supported in part by a grant from the Group Health Foundation Momentum Fund.
FROM OBSTETRICS & GYNECOLOGY
Key clinical point: Increased diagnoses of gestational diabetes did not significantly improve maternal or fetal outcomes.
Major finding: Adoption of a one-step screening process for gestational diabetes increased diagnoses by 41%.
Study details: The data come from a before-and-after cohort study with a population of 23,257 women.
Disclosures: Dr. Pocobelli disclosed funding from Jazz Pharmaceuticals for work unrelated to this study. The study was supported in part by a grant from the Group Health Foundation Momentum Fund.
Source: Pocobelli G et al. Obstet Gynecol. 2018;132:859-67.
FDA attacks antibiotic resistance with new strategy
WASHINGTON – A strategy combining stewardship and science is needed to help combat antimicrobial resistance, and updated plans from the U.S. Food and Drug Administration include four key components to address all aspects of product development and use, FDA commissioner Scott Gottlieb, MD, said in a press briefing in Washington on Sept. 14.
“The FDA plays a unique role in advancing human and animal health” that provides a unique vantage point for coordinating all aspects of product development and application, he said.
The FDA’s comprehensive approach to the challenge of antimicrobial resistance (AMR) includes:
- Facilitating product development.
- Promoting antimicrobial stewardship.
- Supporting the development of new tools for surveillance.
- Advancing scientific initiatives, including research for the development of alternative treatments.
The FDA’s product development plan to combat AMR includes the creation of incentives for companies to develop new antibiotic products and create a robust pipeline, which is a challenge because of the lack of immediate economic gain, Dr. Gottlieb said.
“It necessary to change the perception that the costs and risks of antibiotic innovation are too high relative to their expected gains,” he emphasized.
Strategies to incentivize companies include fast track designation, priority review, and breakthrough therapy designation. In addition, the Limited Population Pathway for Antibacterial and Antifungal Drugs (LPAD) is designed to promote development of antimicrobial drugs for limited and underserved populations, Dr. Gottlieb said. The FDA plan also calls for pursuing reimbursement options with the Centers for Medicare & Medicaid Services.
Promoting antimicrobial stewardship remains an ongoing element of the FDA’s plan to reduce AMR. In conjunction with the release of the FDA’s updated approach to AMR, the FDA’s Center for Veterinary Medicine CVM released a 5-year action plan to promote and support antimicrobial stewardship in not only the agricultural arena, but in companion animals as well.
The FDA plans to bring all antimicrobials of medical importance that are approved for use in animals under the oversight of CVM, which will pursue the improve labeling on antimicrobial drugs used in the feed and water of food-producing animals, including defining durations of use, Dr. Gottlieb noted.
Supporting the development and improvement of surveillance tools is “essential to understanding the drivers of resistance in human and veterinary settings and formulating appropriate responses” to outbreaks, Dr. Gottlieb said.
To help meet this goal, the FDA will expand sampling via the National Antimicrobial Resistance Monitoring System (NARMS) database, he said. Other surveillance goals include supporting genomics research and expanding AMR monitoring to include pathogens associated with animal feed and companion animals, he added.
As part of the final component of the FDA’s AMR strategy to advance scientific initiatives, the FDA has released a new Request for Information “to obtain additional, external input on how best to develop an annual list of regulatory science initiatives specific for antimicrobial products,” Dr. Gottlieb announced. The FDA intends to use the information gained from clinicians and others in its creation of guidance documents and recommendations to streamline the antibiotic development process. He also cited the FDA’s ongoing support of partnerships with public and private organizations such as the Clinical Trials Transformation Initiative, which focuses on drug development for severe bacterial infections with current unmet medical need.
“We need to harness science and policy to help our public health systems and researchers become nimbler in the battle against drug-resistant pathogens,” Dr. Gottlieb concluded.
In a panel discussion following the briefing, several experts offered perspective on the FDA’s goals and on the challenges of AMR.
William Flynn, DVM, deputy director of science policy for the Center of Veterinary Medicine, noted some goals for reducing the use of antibiotics in the veterinary arena.
“We are trying to focus on the driver: What are the disease conditions that drive use of the product,” he said. Ideally, better management of disease conditions can reduce reliance on antibiotics, he added.
Also in the panel discussion, Steven Gitterman, MD, deputy director of the division of microbiology devices at the Center for Devices and Radiological Health, emphasized the value of sustainable trial databases so AMR research can continue on an ongoing basis. Finally, Carolyn Wilson, PhD, associate director of research at the Center for Biologics Evaluation and Research, noted that the FDA’s research and development efforts include antibiotic alternatives, including live biotherapeutic products, fecal microbiota transplantation, and bacteriophage therapy.
Visit www.fda.gov for a transcript of Dr. Gottlieb’s talk, and for the updated FDA website page with more details on the agency’s plans to combat antimicrobial resistance.
Dr. Gottlieb and the panelists had no financial conflicts to disclose.
WASHINGTON – A strategy combining stewardship and science is needed to help combat antimicrobial resistance, and updated plans from the U.S. Food and Drug Administration include four key components to address all aspects of product development and use, FDA commissioner Scott Gottlieb, MD, said in a press briefing in Washington on Sept. 14.
“The FDA plays a unique role in advancing human and animal health” that provides a unique vantage point for coordinating all aspects of product development and application, he said.
The FDA’s comprehensive approach to the challenge of antimicrobial resistance (AMR) includes:
- Facilitating product development.
- Promoting antimicrobial stewardship.
- Supporting the development of new tools for surveillance.
- Advancing scientific initiatives, including research for the development of alternative treatments.
The FDA’s product development plan to combat AMR includes the creation of incentives for companies to develop new antibiotic products and create a robust pipeline, which is a challenge because of the lack of immediate economic gain, Dr. Gottlieb said.
“It necessary to change the perception that the costs and risks of antibiotic innovation are too high relative to their expected gains,” he emphasized.
Strategies to incentivize companies include fast track designation, priority review, and breakthrough therapy designation. In addition, the Limited Population Pathway for Antibacterial and Antifungal Drugs (LPAD) is designed to promote development of antimicrobial drugs for limited and underserved populations, Dr. Gottlieb said. The FDA plan also calls for pursuing reimbursement options with the Centers for Medicare & Medicaid Services.
Promoting antimicrobial stewardship remains an ongoing element of the FDA’s plan to reduce AMR. In conjunction with the release of the FDA’s updated approach to AMR, the FDA’s Center for Veterinary Medicine CVM released a 5-year action plan to promote and support antimicrobial stewardship in not only the agricultural arena, but in companion animals as well.
The FDA plans to bring all antimicrobials of medical importance that are approved for use in animals under the oversight of CVM, which will pursue the improve labeling on antimicrobial drugs used in the feed and water of food-producing animals, including defining durations of use, Dr. Gottlieb noted.
Supporting the development and improvement of surveillance tools is “essential to understanding the drivers of resistance in human and veterinary settings and formulating appropriate responses” to outbreaks, Dr. Gottlieb said.
To help meet this goal, the FDA will expand sampling via the National Antimicrobial Resistance Monitoring System (NARMS) database, he said. Other surveillance goals include supporting genomics research and expanding AMR monitoring to include pathogens associated with animal feed and companion animals, he added.
As part of the final component of the FDA’s AMR strategy to advance scientific initiatives, the FDA has released a new Request for Information “to obtain additional, external input on how best to develop an annual list of regulatory science initiatives specific for antimicrobial products,” Dr. Gottlieb announced. The FDA intends to use the information gained from clinicians and others in its creation of guidance documents and recommendations to streamline the antibiotic development process. He also cited the FDA’s ongoing support of partnerships with public and private organizations such as the Clinical Trials Transformation Initiative, which focuses on drug development for severe bacterial infections with current unmet medical need.
“We need to harness science and policy to help our public health systems and researchers become nimbler in the battle against drug-resistant pathogens,” Dr. Gottlieb concluded.
In a panel discussion following the briefing, several experts offered perspective on the FDA’s goals and on the challenges of AMR.
William Flynn, DVM, deputy director of science policy for the Center of Veterinary Medicine, noted some goals for reducing the use of antibiotics in the veterinary arena.
“We are trying to focus on the driver: What are the disease conditions that drive use of the product,” he said. Ideally, better management of disease conditions can reduce reliance on antibiotics, he added.
Also in the panel discussion, Steven Gitterman, MD, deputy director of the division of microbiology devices at the Center for Devices and Radiological Health, emphasized the value of sustainable trial databases so AMR research can continue on an ongoing basis. Finally, Carolyn Wilson, PhD, associate director of research at the Center for Biologics Evaluation and Research, noted that the FDA’s research and development efforts include antibiotic alternatives, including live biotherapeutic products, fecal microbiota transplantation, and bacteriophage therapy.
Visit www.fda.gov for a transcript of Dr. Gottlieb’s talk, and for the updated FDA website page with more details on the agency’s plans to combat antimicrobial resistance.
Dr. Gottlieb and the panelists had no financial conflicts to disclose.
WASHINGTON – A strategy combining stewardship and science is needed to help combat antimicrobial resistance, and updated plans from the U.S. Food and Drug Administration include four key components to address all aspects of product development and use, FDA commissioner Scott Gottlieb, MD, said in a press briefing in Washington on Sept. 14.
“The FDA plays a unique role in advancing human and animal health” that provides a unique vantage point for coordinating all aspects of product development and application, he said.
The FDA’s comprehensive approach to the challenge of antimicrobial resistance (AMR) includes:
- Facilitating product development.
- Promoting antimicrobial stewardship.
- Supporting the development of new tools for surveillance.
- Advancing scientific initiatives, including research for the development of alternative treatments.
The FDA’s product development plan to combat AMR includes the creation of incentives for companies to develop new antibiotic products and create a robust pipeline, which is a challenge because of the lack of immediate economic gain, Dr. Gottlieb said.
“It necessary to change the perception that the costs and risks of antibiotic innovation are too high relative to their expected gains,” he emphasized.
Strategies to incentivize companies include fast track designation, priority review, and breakthrough therapy designation. In addition, the Limited Population Pathway for Antibacterial and Antifungal Drugs (LPAD) is designed to promote development of antimicrobial drugs for limited and underserved populations, Dr. Gottlieb said. The FDA plan also calls for pursuing reimbursement options with the Centers for Medicare & Medicaid Services.
Promoting antimicrobial stewardship remains an ongoing element of the FDA’s plan to reduce AMR. In conjunction with the release of the FDA’s updated approach to AMR, the FDA’s Center for Veterinary Medicine CVM released a 5-year action plan to promote and support antimicrobial stewardship in not only the agricultural arena, but in companion animals as well.
The FDA plans to bring all antimicrobials of medical importance that are approved for use in animals under the oversight of CVM, which will pursue the improve labeling on antimicrobial drugs used in the feed and water of food-producing animals, including defining durations of use, Dr. Gottlieb noted.
Supporting the development and improvement of surveillance tools is “essential to understanding the drivers of resistance in human and veterinary settings and formulating appropriate responses” to outbreaks, Dr. Gottlieb said.
To help meet this goal, the FDA will expand sampling via the National Antimicrobial Resistance Monitoring System (NARMS) database, he said. Other surveillance goals include supporting genomics research and expanding AMR monitoring to include pathogens associated with animal feed and companion animals, he added.
As part of the final component of the FDA’s AMR strategy to advance scientific initiatives, the FDA has released a new Request for Information “to obtain additional, external input on how best to develop an annual list of regulatory science initiatives specific for antimicrobial products,” Dr. Gottlieb announced. The FDA intends to use the information gained from clinicians and others in its creation of guidance documents and recommendations to streamline the antibiotic development process. He also cited the FDA’s ongoing support of partnerships with public and private organizations such as the Clinical Trials Transformation Initiative, which focuses on drug development for severe bacterial infections with current unmet medical need.
“We need to harness science and policy to help our public health systems and researchers become nimbler in the battle against drug-resistant pathogens,” Dr. Gottlieb concluded.
In a panel discussion following the briefing, several experts offered perspective on the FDA’s goals and on the challenges of AMR.
William Flynn, DVM, deputy director of science policy for the Center of Veterinary Medicine, noted some goals for reducing the use of antibiotics in the veterinary arena.
“We are trying to focus on the driver: What are the disease conditions that drive use of the product,” he said. Ideally, better management of disease conditions can reduce reliance on antibiotics, he added.
Also in the panel discussion, Steven Gitterman, MD, deputy director of the division of microbiology devices at the Center for Devices and Radiological Health, emphasized the value of sustainable trial databases so AMR research can continue on an ongoing basis. Finally, Carolyn Wilson, PhD, associate director of research at the Center for Biologics Evaluation and Research, noted that the FDA’s research and development efforts include antibiotic alternatives, including live biotherapeutic products, fecal microbiota transplantation, and bacteriophage therapy.
Visit www.fda.gov for a transcript of Dr. Gottlieb’s talk, and for the updated FDA website page with more details on the agency’s plans to combat antimicrobial resistance.
Dr. Gottlieb and the panelists had no financial conflicts to disclose.
GERD patients who fail PPI often have functional heartburn or hypersensitivity
Abnormal pH results were similar in patients with gastroesophageal reflux disease (GERD) who improved or failed to improve on a once-daily dose of a proton pump inhibitor (PPI), but 75% of patients who failed treatment demonstrated either functional heartburn or reflux hypersensitivity, based on data from 29 adults.
Previous research on PPI failure in GERD patients has focused on twice-daily doses; “the purpose of the study was to compare impedance-pH parameters between patients who failed versus those who responded to PPI once daily,” wrote Jason Abdallah, MD, of Case Western Reserve University in Cleveland and colleagues.
In a study published in Clinical Gastroenterology and Hepatology, the investigators reviewed data from adults diagnosed with GERD who were treated with PPI therapy. The 16 who reported heartburn and/or regurgitation at least twice a week for 3 months while on a standard, once-daily PPI dose were classified as the failure group. The 13 patients who reported complete symptom resolution for at least 4 weeks while on the same standard dose were classified as the success group.
Most of the patients in the PPI-failure group (75%) were found to have either functional heartburn or reflux hypersensitivity with GERD. Impedance and pH parameters did not differ significantly between the PPI-failure and -success group, the researchers noted. Abnormal pH test results were similar between the groups, occurring in four of the patients who were successfully treated with PPI (31%) and four of the patients who failed PPI treatment (25%).
All patients completed the Short-Form 36 (SF-36) and GERD Health-Related Quality of Life (GERD-HRQL) questionnaires, and all underwent upper endoscopy and combined 24-hour esophageal impedance and pH monitoring within 2-4 weeks of study enrollment and while following their PPI treatment plans. There were no significant differences in demographic characteristics between the success and failure groups; the mean ages were 55 years and 47 years, respectively.
The patients in the success group averaged higher scores on the SF-36 than the failure group, but the difference was not significant. On the GERD-HRQL, treatment-failure patients reported that overall heartburn and either heartburn or bloating while lying down were the symptoms they found most annoying on a daily basis.
Among the treatment-failure patients, 10 (62%) had normal acid exposure and negative symptom-reflux association, 2 patients (13%) had normal acid exposure and positive symptom-reflux association, and 4 patients (25%) had abnormal esophageal acid exposure. Patients in the treatment failure group reported a total of 315 episodes of either heartburn or regurgitation.
Endoscopy findings were normal in most of the patients in both groups; 81% of the treatment-failure patients and 69% of the treatment-success patients had normal upper endoscopy findings. Abnormal findings in the treatment-success group included one case of erosive esophagitis, two cases of Barrett’s esophagus, three cases of nonobstructive Schatzki rings, and five cases of hiatal hernia. Abnormal findings in the treatment-failure group included two cases of Schatzki rings, one case of esophageal stricture, and three cases of hiatal hernia.
The total number of reflux events was similar between the groups; 1,279 in the treatment-failure group and 1,099 in the treatment-success group, with the number of reflux events per patient averaging 80 and 84, respectively.
“Our results support the hypothesis that PPI failure is primarily driven by esophageal hypersensitivity,” the researchers noted. The similarity in impedance and reflux “implies that the shift to nonacidic reflux is a general PPI phenomenon, as opposed to being unique to PPI-failure patients,” they said.
The study was limited by the small patient population, but the results provide some insight into refractory GERD and suggest that patients who fail to respond to once-daily PPI might benefit from a neuromodulator, as well as psychological interventions including cognitive-behavioral therapy, hypnotherapy, relaxation techniques, mindfulness, and biofeedback, the researchers concluded.
Dr. Abdullah had no financial conflicts to disclose; a coauthor disclosed relationships with companies including Ironwood Pharmaceuticals, Mederi Therapeutics, and Ethicon Pharmaceuticals.
SOURCE: Abdallah J et al. Clin Gastroenterol Hepatol. 2018; doi: 10.1016/j.cgh.2018.06.018.
Abnormal pH results were similar in patients with gastroesophageal reflux disease (GERD) who improved or failed to improve on a once-daily dose of a proton pump inhibitor (PPI), but 75% of patients who failed treatment demonstrated either functional heartburn or reflux hypersensitivity, based on data from 29 adults.
Previous research on PPI failure in GERD patients has focused on twice-daily doses; “the purpose of the study was to compare impedance-pH parameters between patients who failed versus those who responded to PPI once daily,” wrote Jason Abdallah, MD, of Case Western Reserve University in Cleveland and colleagues.
In a study published in Clinical Gastroenterology and Hepatology, the investigators reviewed data from adults diagnosed with GERD who were treated with PPI therapy. The 16 who reported heartburn and/or regurgitation at least twice a week for 3 months while on a standard, once-daily PPI dose were classified as the failure group. The 13 patients who reported complete symptom resolution for at least 4 weeks while on the same standard dose were classified as the success group.
Most of the patients in the PPI-failure group (75%) were found to have either functional heartburn or reflux hypersensitivity with GERD. Impedance and pH parameters did not differ significantly between the PPI-failure and -success group, the researchers noted. Abnormal pH test results were similar between the groups, occurring in four of the patients who were successfully treated with PPI (31%) and four of the patients who failed PPI treatment (25%).
All patients completed the Short-Form 36 (SF-36) and GERD Health-Related Quality of Life (GERD-HRQL) questionnaires, and all underwent upper endoscopy and combined 24-hour esophageal impedance and pH monitoring within 2-4 weeks of study enrollment and while following their PPI treatment plans. There were no significant differences in demographic characteristics between the success and failure groups; the mean ages were 55 years and 47 years, respectively.
The patients in the success group averaged higher scores on the SF-36 than the failure group, but the difference was not significant. On the GERD-HRQL, treatment-failure patients reported that overall heartburn and either heartburn or bloating while lying down were the symptoms they found most annoying on a daily basis.
Among the treatment-failure patients, 10 (62%) had normal acid exposure and negative symptom-reflux association, 2 patients (13%) had normal acid exposure and positive symptom-reflux association, and 4 patients (25%) had abnormal esophageal acid exposure. Patients in the treatment failure group reported a total of 315 episodes of either heartburn or regurgitation.
Endoscopy findings were normal in most of the patients in both groups; 81% of the treatment-failure patients and 69% of the treatment-success patients had normal upper endoscopy findings. Abnormal findings in the treatment-success group included one case of erosive esophagitis, two cases of Barrett’s esophagus, three cases of nonobstructive Schatzki rings, and five cases of hiatal hernia. Abnormal findings in the treatment-failure group included two cases of Schatzki rings, one case of esophageal stricture, and three cases of hiatal hernia.
The total number of reflux events was similar between the groups; 1,279 in the treatment-failure group and 1,099 in the treatment-success group, with the number of reflux events per patient averaging 80 and 84, respectively.
“Our results support the hypothesis that PPI failure is primarily driven by esophageal hypersensitivity,” the researchers noted. The similarity in impedance and reflux “implies that the shift to nonacidic reflux is a general PPI phenomenon, as opposed to being unique to PPI-failure patients,” they said.
The study was limited by the small patient population, but the results provide some insight into refractory GERD and suggest that patients who fail to respond to once-daily PPI might benefit from a neuromodulator, as well as psychological interventions including cognitive-behavioral therapy, hypnotherapy, relaxation techniques, mindfulness, and biofeedback, the researchers concluded.
Dr. Abdullah had no financial conflicts to disclose; a coauthor disclosed relationships with companies including Ironwood Pharmaceuticals, Mederi Therapeutics, and Ethicon Pharmaceuticals.
SOURCE: Abdallah J et al. Clin Gastroenterol Hepatol. 2018; doi: 10.1016/j.cgh.2018.06.018.
Abnormal pH results were similar in patients with gastroesophageal reflux disease (GERD) who improved or failed to improve on a once-daily dose of a proton pump inhibitor (PPI), but 75% of patients who failed treatment demonstrated either functional heartburn or reflux hypersensitivity, based on data from 29 adults.
Previous research on PPI failure in GERD patients has focused on twice-daily doses; “the purpose of the study was to compare impedance-pH parameters between patients who failed versus those who responded to PPI once daily,” wrote Jason Abdallah, MD, of Case Western Reserve University in Cleveland and colleagues.
In a study published in Clinical Gastroenterology and Hepatology, the investigators reviewed data from adults diagnosed with GERD who were treated with PPI therapy. The 16 who reported heartburn and/or regurgitation at least twice a week for 3 months while on a standard, once-daily PPI dose were classified as the failure group. The 13 patients who reported complete symptom resolution for at least 4 weeks while on the same standard dose were classified as the success group.
Most of the patients in the PPI-failure group (75%) were found to have either functional heartburn or reflux hypersensitivity with GERD. Impedance and pH parameters did not differ significantly between the PPI-failure and -success group, the researchers noted. Abnormal pH test results were similar between the groups, occurring in four of the patients who were successfully treated with PPI (31%) and four of the patients who failed PPI treatment (25%).
All patients completed the Short-Form 36 (SF-36) and GERD Health-Related Quality of Life (GERD-HRQL) questionnaires, and all underwent upper endoscopy and combined 24-hour esophageal impedance and pH monitoring within 2-4 weeks of study enrollment and while following their PPI treatment plans. There were no significant differences in demographic characteristics between the success and failure groups; the mean ages were 55 years and 47 years, respectively.
The patients in the success group averaged higher scores on the SF-36 than the failure group, but the difference was not significant. On the GERD-HRQL, treatment-failure patients reported that overall heartburn and either heartburn or bloating while lying down were the symptoms they found most annoying on a daily basis.
Among the treatment-failure patients, 10 (62%) had normal acid exposure and negative symptom-reflux association, 2 patients (13%) had normal acid exposure and positive symptom-reflux association, and 4 patients (25%) had abnormal esophageal acid exposure. Patients in the treatment failure group reported a total of 315 episodes of either heartburn or regurgitation.
Endoscopy findings were normal in most of the patients in both groups; 81% of the treatment-failure patients and 69% of the treatment-success patients had normal upper endoscopy findings. Abnormal findings in the treatment-success group included one case of erosive esophagitis, two cases of Barrett’s esophagus, three cases of nonobstructive Schatzki rings, and five cases of hiatal hernia. Abnormal findings in the treatment-failure group included two cases of Schatzki rings, one case of esophageal stricture, and three cases of hiatal hernia.
The total number of reflux events was similar between the groups; 1,279 in the treatment-failure group and 1,099 in the treatment-success group, with the number of reflux events per patient averaging 80 and 84, respectively.
“Our results support the hypothesis that PPI failure is primarily driven by esophageal hypersensitivity,” the researchers noted. The similarity in impedance and reflux “implies that the shift to nonacidic reflux is a general PPI phenomenon, as opposed to being unique to PPI-failure patients,” they said.
The study was limited by the small patient population, but the results provide some insight into refractory GERD and suggest that patients who fail to respond to once-daily PPI might benefit from a neuromodulator, as well as psychological interventions including cognitive-behavioral therapy, hypnotherapy, relaxation techniques, mindfulness, and biofeedback, the researchers concluded.
Dr. Abdullah had no financial conflicts to disclose; a coauthor disclosed relationships with companies including Ironwood Pharmaceuticals, Mederi Therapeutics, and Ethicon Pharmaceuticals.
SOURCE: Abdallah J et al. Clin Gastroenterol Hepatol. 2018; doi: 10.1016/j.cgh.2018.06.018.
FROM CLINICAL GASTROENTEROLOGY AND HEPATOLOGY
Key clinical point: PPI failure in GERD patients appears to be driven by esophageal hypersensitivity, not significantly associated with reflux.
Major finding: Most (75%) of the patients who failed PPI treatment had heartburn or reflux hypersensitivity with GERD.
Study details: The data come from a prospective cohort study of 29 adults with GERD.
Disclosures: Dr. Abdullah had no financial conflicts to disclose; a coauthor disclosed relationships with companies including Ironwood Pharmaceuticals, Mederi Therapeutics, and Ethicon Pharmaceuticals.
Source: Abdullah J et al. Clin Gastroenterol Hepatol. 2018; doi: 10.1016/j.cgh.2018.06.018.
Psoriatic arthritis activity spikes briefly postpartum
Disease activity for women with psoriatic arthritis seeking pregnancy was relatively stable through 1 year after delivery, but there was a significant jump at 6 months postpartum follow-up, based on data from a prospective study of more than 100 patients.
Previous research has described rheumatoid arthritis remission during pregnancy, but the symptoms of psoriatic arthritis (PsA) before, during, and after pregnancy have not been well studied, wrote Kristin Ursin, MD, of Trondheim (Norway) University Hospital, and her colleagues.
In a study published in Arthritis Care & Research, the investigators reviewed data from 108 pregnancies in 103 women with PsA who were diagnosed between January 2006 and October 2017.
The participants were enrolled in a Norwegian nationwide registry that followed women with inflammatory diseases from preconception through 1 year after delivery. Disease activity was measured using the DAS28-CRP (28-Joint Disease Activity Score with C-reactive Protein). Participants were assessed at seven time points: before pregnancy, during each trimester, and at 6 weeks, 6 months, and 12 months after delivery.
Although approximately 75% of the women had stable disease activity throughout the study period, activity spiked at 6 months after delivery; DAS28-CRP scores at 6 months post partum were significantly higher than scores at 6 weeks post partum (2.71 vs. 2.45, respectively; P = .016).
The researchers conducted an additional analysis of the potential role of tumor necrosis factor inhibitor use and found that women taking a TNFi had significantly lower disease activity during pregnancy, compared with women not taking a TNFi; mean DAS28-CRP scores at 6 months post partum for these groups were 2.22 and 2.72, respectively (P = .043).
The study was limited by the use of DAS28-CRP as the main measure of disease activity; the index does not include potentially affected distal interphalangeal joints. In addition, not all the participants were assessed at each of the seven time points. However, the results suggest that most pregnant women with PsA experience low levels of disease activity, the researchers said. “Future research on pregnancy in women with PsA should include extended joint count (66/68 joints), and assessment of dactylitis, entheses, axial skeleton, and psoriasis,” they added.
The researchers had no financial conflicts to disclose. The study was funded by the department of rheumatology at Trondheim University Hospital and the Research Fund of the Norwegian organization for people with rheumatic disease.
SOURCE: Ursin K et al. Arthritis Care Res. 2018 Sep 7. doi: 10.1002/acr.23747.
Disease activity for women with psoriatic arthritis seeking pregnancy was relatively stable through 1 year after delivery, but there was a significant jump at 6 months postpartum follow-up, based on data from a prospective study of more than 100 patients.
Previous research has described rheumatoid arthritis remission during pregnancy, but the symptoms of psoriatic arthritis (PsA) before, during, and after pregnancy have not been well studied, wrote Kristin Ursin, MD, of Trondheim (Norway) University Hospital, and her colleagues.
In a study published in Arthritis Care & Research, the investigators reviewed data from 108 pregnancies in 103 women with PsA who were diagnosed between January 2006 and October 2017.
The participants were enrolled in a Norwegian nationwide registry that followed women with inflammatory diseases from preconception through 1 year after delivery. Disease activity was measured using the DAS28-CRP (28-Joint Disease Activity Score with C-reactive Protein). Participants were assessed at seven time points: before pregnancy, during each trimester, and at 6 weeks, 6 months, and 12 months after delivery.
Although approximately 75% of the women had stable disease activity throughout the study period, activity spiked at 6 months after delivery; DAS28-CRP scores at 6 months post partum were significantly higher than scores at 6 weeks post partum (2.71 vs. 2.45, respectively; P = .016).
The researchers conducted an additional analysis of the potential role of tumor necrosis factor inhibitor use and found that women taking a TNFi had significantly lower disease activity during pregnancy, compared with women not taking a TNFi; mean DAS28-CRP scores at 6 months post partum for these groups were 2.22 and 2.72, respectively (P = .043).
The study was limited by the use of DAS28-CRP as the main measure of disease activity; the index does not include potentially affected distal interphalangeal joints. In addition, not all the participants were assessed at each of the seven time points. However, the results suggest that most pregnant women with PsA experience low levels of disease activity, the researchers said. “Future research on pregnancy in women with PsA should include extended joint count (66/68 joints), and assessment of dactylitis, entheses, axial skeleton, and psoriasis,” they added.
The researchers had no financial conflicts to disclose. The study was funded by the department of rheumatology at Trondheim University Hospital and the Research Fund of the Norwegian organization for people with rheumatic disease.
SOURCE: Ursin K et al. Arthritis Care Res. 2018 Sep 7. doi: 10.1002/acr.23747.
Disease activity for women with psoriatic arthritis seeking pregnancy was relatively stable through 1 year after delivery, but there was a significant jump at 6 months postpartum follow-up, based on data from a prospective study of more than 100 patients.
Previous research has described rheumatoid arthritis remission during pregnancy, but the symptoms of psoriatic arthritis (PsA) before, during, and after pregnancy have not been well studied, wrote Kristin Ursin, MD, of Trondheim (Norway) University Hospital, and her colleagues.
In a study published in Arthritis Care & Research, the investigators reviewed data from 108 pregnancies in 103 women with PsA who were diagnosed between January 2006 and October 2017.
The participants were enrolled in a Norwegian nationwide registry that followed women with inflammatory diseases from preconception through 1 year after delivery. Disease activity was measured using the DAS28-CRP (28-Joint Disease Activity Score with C-reactive Protein). Participants were assessed at seven time points: before pregnancy, during each trimester, and at 6 weeks, 6 months, and 12 months after delivery.
Although approximately 75% of the women had stable disease activity throughout the study period, activity spiked at 6 months after delivery; DAS28-CRP scores at 6 months post partum were significantly higher than scores at 6 weeks post partum (2.71 vs. 2.45, respectively; P = .016).
The researchers conducted an additional analysis of the potential role of tumor necrosis factor inhibitor use and found that women taking a TNFi had significantly lower disease activity during pregnancy, compared with women not taking a TNFi; mean DAS28-CRP scores at 6 months post partum for these groups were 2.22 and 2.72, respectively (P = .043).
The study was limited by the use of DAS28-CRP as the main measure of disease activity; the index does not include potentially affected distal interphalangeal joints. In addition, not all the participants were assessed at each of the seven time points. However, the results suggest that most pregnant women with PsA experience low levels of disease activity, the researchers said. “Future research on pregnancy in women with PsA should include extended joint count (66/68 joints), and assessment of dactylitis, entheses, axial skeleton, and psoriasis,” they added.
The researchers had no financial conflicts to disclose. The study was funded by the department of rheumatology at Trondheim University Hospital and the Research Fund of the Norwegian organization for people with rheumatic disease.
SOURCE: Ursin K et al. Arthritis Care Res. 2018 Sep 7. doi: 10.1002/acr.23747.
FROM ARTHRITIS CARE & RESEARCH
Key clinical point: Disease activity for pregnant women with PsA decreased during pregnancy, increased significantly by 6 months post partum, and returned to baseline by 12 months post partum.
Major finding: The average DAS28-CRP was 2.71 at 6 months vs. 2.45 at 6 weeks (P = .016).
Study details: The data come from 108 women with 103 pregnancies who were part of a national registry in Norway.
Disclosures: The researchers had no financial conflicts to disclose. The study was funded by the department of rheumatology at Trondheim University Hospital and the Research Fund of the Norwegian organization for people with rheumatic disease.
Source: Ursin K et al. Arthritis Care Res. 2018 Sep 7. doi: 10.1002/acr.23747.
Global incidence of rosacea estimated to be over 5%
The global incidence of rosacea among adults is approximately 5%, based on data from a meta-analysis and systematic review.
The National Rosacea Society Expert Committee recently updated its phenotype-based classification system, but the global prevalence and incidence of rosacea remain unknown, wrote Lise Gether, MD, of the University of Copenhagen, Denmark, and her colleagues.
In a review published in the British Journal of Dermatology, the researchers examined the prevalence of rosacea among dermatology patients and in the general population worldwide by analyzing 32 studies including 41 populations and 26,538,319 individuals. The study populations comprised 22 from Europe, 9 from North America, 4 from Asia, 3 from South America, and 3 from Africa. Of the 32 studies, 18 included the general population and 14 included only dermatology patients.
Overall, the pooled proportion of individuals with rosacea was 5.46% in the general population and 2.39% among dermatology patients. Of note, the pooled proportions varied when the studies were grouped by diagnostic method: self-diagnosis (9.7%), physician diagnosis (5.5%), and health care database estimate (1.05%).
Rosacea prevalence by age was highest among individuals aged 45-60 years, but “based on the available data, it was not possible to make useful stratified estimates,” the researchers said.
The researchers estimated rosacea prevalence by gender using data from the 10 studies that reported numbers from both genders. Based on a population of 5,601,642 women and 3,529,559 men, the pooled proportions were similar; 5.4% for women and 3.9% for men.
“The methods used to study the prevalence of rosacea are of great importance as misclassification may be a concern,” the researchers noted. Individuals with mild to moderate rosacea might not seek medical treatment, which might contribute to the low prevalence from health care database estimates. Conversely, the high prevalence with self-reports might suggest a lack of specificity in the questionnaires.
There were no external funding sources. Dr. Gether had no relevant financial disclosures. Coauthors disclosed relationships with companies including Galderma, Pfizer, Eli Lilly, Novartis, and Janssen.
SOURCE: Gether L et al. Br J Dermatol. 2018. doi: 10.1111/bjd.16481
The global incidence of rosacea among adults is approximately 5%, based on data from a meta-analysis and systematic review.
The National Rosacea Society Expert Committee recently updated its phenotype-based classification system, but the global prevalence and incidence of rosacea remain unknown, wrote Lise Gether, MD, of the University of Copenhagen, Denmark, and her colleagues.
In a review published in the British Journal of Dermatology, the researchers examined the prevalence of rosacea among dermatology patients and in the general population worldwide by analyzing 32 studies including 41 populations and 26,538,319 individuals. The study populations comprised 22 from Europe, 9 from North America, 4 from Asia, 3 from South America, and 3 from Africa. Of the 32 studies, 18 included the general population and 14 included only dermatology patients.
Overall, the pooled proportion of individuals with rosacea was 5.46% in the general population and 2.39% among dermatology patients. Of note, the pooled proportions varied when the studies were grouped by diagnostic method: self-diagnosis (9.7%), physician diagnosis (5.5%), and health care database estimate (1.05%).
Rosacea prevalence by age was highest among individuals aged 45-60 years, but “based on the available data, it was not possible to make useful stratified estimates,” the researchers said.
The researchers estimated rosacea prevalence by gender using data from the 10 studies that reported numbers from both genders. Based on a population of 5,601,642 women and 3,529,559 men, the pooled proportions were similar; 5.4% for women and 3.9% for men.
“The methods used to study the prevalence of rosacea are of great importance as misclassification may be a concern,” the researchers noted. Individuals with mild to moderate rosacea might not seek medical treatment, which might contribute to the low prevalence from health care database estimates. Conversely, the high prevalence with self-reports might suggest a lack of specificity in the questionnaires.
There were no external funding sources. Dr. Gether had no relevant financial disclosures. Coauthors disclosed relationships with companies including Galderma, Pfizer, Eli Lilly, Novartis, and Janssen.
SOURCE: Gether L et al. Br J Dermatol. 2018. doi: 10.1111/bjd.16481
The global incidence of rosacea among adults is approximately 5%, based on data from a meta-analysis and systematic review.
The National Rosacea Society Expert Committee recently updated its phenotype-based classification system, but the global prevalence and incidence of rosacea remain unknown, wrote Lise Gether, MD, of the University of Copenhagen, Denmark, and her colleagues.
In a review published in the British Journal of Dermatology, the researchers examined the prevalence of rosacea among dermatology patients and in the general population worldwide by analyzing 32 studies including 41 populations and 26,538,319 individuals. The study populations comprised 22 from Europe, 9 from North America, 4 from Asia, 3 from South America, and 3 from Africa. Of the 32 studies, 18 included the general population and 14 included only dermatology patients.
Overall, the pooled proportion of individuals with rosacea was 5.46% in the general population and 2.39% among dermatology patients. Of note, the pooled proportions varied when the studies were grouped by diagnostic method: self-diagnosis (9.7%), physician diagnosis (5.5%), and health care database estimate (1.05%).
Rosacea prevalence by age was highest among individuals aged 45-60 years, but “based on the available data, it was not possible to make useful stratified estimates,” the researchers said.
The researchers estimated rosacea prevalence by gender using data from the 10 studies that reported numbers from both genders. Based on a population of 5,601,642 women and 3,529,559 men, the pooled proportions were similar; 5.4% for women and 3.9% for men.
“The methods used to study the prevalence of rosacea are of great importance as misclassification may be a concern,” the researchers noted. Individuals with mild to moderate rosacea might not seek medical treatment, which might contribute to the low prevalence from health care database estimates. Conversely, the high prevalence with self-reports might suggest a lack of specificity in the questionnaires.
There were no external funding sources. Dr. Gether had no relevant financial disclosures. Coauthors disclosed relationships with companies including Galderma, Pfizer, Eli Lilly, Novartis, and Janssen.
SOURCE: Gether L et al. Br J Dermatol. 2018. doi: 10.1111/bjd.16481
FROM THE BRITISH JOURNAL OF DERMATOLOGY
Key clinical point: Rosacea occurs worldwide in both men and women, but diagnosis remains inconsistent.
Major finding:
Study details: A meta-analysis of 32 studies and 26,538,319 individuals.
Disclosures: There were no external funding sources. Dr. Gether had no relevant financial disclosures. Coauthors disclosed relationships with companies including Galderma, Pfizer, Eli Lilly, Novartis, and Janssen.
Source: Gether L et al. Br J Dermatol. 2018. doi: 10.1111/bjd.16481.