User login
Linezolid succeeds against gram-positive bacterial infections in ICU
Approximately 80% of patients in intensive care showed clinical improvement in gram-positive bacterial infections after treatment with linezolid, based on data from more than 300 individuals.
Bacterial infections remain a challenge in the management of critically ill patients, as many gram-positive pathogens have become resistant to multiple drug options, Aijia Ma, MD, of West China Hospital of Sichuan University, and colleagues wrote.
Linezolid has demonstrated effectiveness against MRSA and skin and soft-tissue infections (SSTIs), but its use in critically ill patients with gram-positive infections in the ICU has not been characterized, they said.
In a multicenter, real-world study published in the Journal of Intensive Medicine, the researchers reviewed data from 52 hospitals between June 2018 and December 2019. The study population included 366 patients admitted to the ICU with a clinical or laboratory diagnosis of a gram-positive bacterial infection. Patients were treated with linezolid injections (200 mg/100 mL) and followed up once a day until 48 hours after discontinuing therapy, transferring out of the ICU, or death. Most of the patients (243) were older than 65 years; 90 were aged 18-65 years, and 30 were younger than 18 years. Approximately two-thirds (67%) were men. The primary outcome of clinical efficacy was success (cured or improved).
Linezolid was used as second-line and first-line treatment in 232 (63.4%) and 134 (36.6%) patients, respectively. The most common isolated strain was Staphylococcus aureus (31% MRSA; 12.6% methicillin-susceptible S. aureus [MSSA]) followed by Enterococci (6.7% vancomycin resistant, 9.2% vancomycin susceptible) and Streptococcus pneumoniae (3.4% multidrug resistant, 1.7% non–multidrug resistant).
Overall, 82.2% of patients met the criteria for clinical success; 34 (9.3%) were cured and 267 (73%) improved. Clinical success rates for first-line and second-line linezolid therapy were 79.9% and 83.6%, respectively. Failure rates for linezolid were higher for second-line versus first-line treatment (9.5% vs. 5.2%).
The clinical success rate was highest against MSSA (93.3%), followed by MRSA (83.8%). The average daily linezolid dose was 1,109 mg, and the mean treatment time was 5.1 days.
A total of eight patients (2.2%) reported linezolid-related adverse events, and four patients discontinued the medication because of them; none reported treatment-related serious adverse events. The low incidence of thrombocytopenia in the current study (two patients), compared with previous studies may have been related to avoidance of linezolid for at-risk patients as determined by clinicians, and the relatively short duration of linezolid use, the researchers wrote.
The study findings were limited by several factors, including the observational design and inability to compare the efficacy of different drugs; the small sample size; and the lack of data on drugs used prior to ICU admission, the researchers noted. Other limitations included the low detection rate of gram-positive bacteria and potential underreporting of adverse events.
However, the although clinicians will need to pay close attention to possible side effects and evaluate patient conditions on an individual basis before using linezolid in the clinic, they concluded.
The study was supported by grants from West China Hospital of Sichuan University. The researchers reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Approximately 80% of patients in intensive care showed clinical improvement in gram-positive bacterial infections after treatment with linezolid, based on data from more than 300 individuals.
Bacterial infections remain a challenge in the management of critically ill patients, as many gram-positive pathogens have become resistant to multiple drug options, Aijia Ma, MD, of West China Hospital of Sichuan University, and colleagues wrote.
Linezolid has demonstrated effectiveness against MRSA and skin and soft-tissue infections (SSTIs), but its use in critically ill patients with gram-positive infections in the ICU has not been characterized, they said.
In a multicenter, real-world study published in the Journal of Intensive Medicine, the researchers reviewed data from 52 hospitals between June 2018 and December 2019. The study population included 366 patients admitted to the ICU with a clinical or laboratory diagnosis of a gram-positive bacterial infection. Patients were treated with linezolid injections (200 mg/100 mL) and followed up once a day until 48 hours after discontinuing therapy, transferring out of the ICU, or death. Most of the patients (243) were older than 65 years; 90 were aged 18-65 years, and 30 were younger than 18 years. Approximately two-thirds (67%) were men. The primary outcome of clinical efficacy was success (cured or improved).
Linezolid was used as second-line and first-line treatment in 232 (63.4%) and 134 (36.6%) patients, respectively. The most common isolated strain was Staphylococcus aureus (31% MRSA; 12.6% methicillin-susceptible S. aureus [MSSA]) followed by Enterococci (6.7% vancomycin resistant, 9.2% vancomycin susceptible) and Streptococcus pneumoniae (3.4% multidrug resistant, 1.7% non–multidrug resistant).
Overall, 82.2% of patients met the criteria for clinical success; 34 (9.3%) were cured and 267 (73%) improved. Clinical success rates for first-line and second-line linezolid therapy were 79.9% and 83.6%, respectively. Failure rates for linezolid were higher for second-line versus first-line treatment (9.5% vs. 5.2%).
The clinical success rate was highest against MSSA (93.3%), followed by MRSA (83.8%). The average daily linezolid dose was 1,109 mg, and the mean treatment time was 5.1 days.
A total of eight patients (2.2%) reported linezolid-related adverse events, and four patients discontinued the medication because of them; none reported treatment-related serious adverse events. The low incidence of thrombocytopenia in the current study (two patients), compared with previous studies may have been related to avoidance of linezolid for at-risk patients as determined by clinicians, and the relatively short duration of linezolid use, the researchers wrote.
The study findings were limited by several factors, including the observational design and inability to compare the efficacy of different drugs; the small sample size; and the lack of data on drugs used prior to ICU admission, the researchers noted. Other limitations included the low detection rate of gram-positive bacteria and potential underreporting of adverse events.
However, the although clinicians will need to pay close attention to possible side effects and evaluate patient conditions on an individual basis before using linezolid in the clinic, they concluded.
The study was supported by grants from West China Hospital of Sichuan University. The researchers reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Approximately 80% of patients in intensive care showed clinical improvement in gram-positive bacterial infections after treatment with linezolid, based on data from more than 300 individuals.
Bacterial infections remain a challenge in the management of critically ill patients, as many gram-positive pathogens have become resistant to multiple drug options, Aijia Ma, MD, of West China Hospital of Sichuan University, and colleagues wrote.
Linezolid has demonstrated effectiveness against MRSA and skin and soft-tissue infections (SSTIs), but its use in critically ill patients with gram-positive infections in the ICU has not been characterized, they said.
In a multicenter, real-world study published in the Journal of Intensive Medicine, the researchers reviewed data from 52 hospitals between June 2018 and December 2019. The study population included 366 patients admitted to the ICU with a clinical or laboratory diagnosis of a gram-positive bacterial infection. Patients were treated with linezolid injections (200 mg/100 mL) and followed up once a day until 48 hours after discontinuing therapy, transferring out of the ICU, or death. Most of the patients (243) were older than 65 years; 90 were aged 18-65 years, and 30 were younger than 18 years. Approximately two-thirds (67%) were men. The primary outcome of clinical efficacy was success (cured or improved).
Linezolid was used as second-line and first-line treatment in 232 (63.4%) and 134 (36.6%) patients, respectively. The most common isolated strain was Staphylococcus aureus (31% MRSA; 12.6% methicillin-susceptible S. aureus [MSSA]) followed by Enterococci (6.7% vancomycin resistant, 9.2% vancomycin susceptible) and Streptococcus pneumoniae (3.4% multidrug resistant, 1.7% non–multidrug resistant).
Overall, 82.2% of patients met the criteria for clinical success; 34 (9.3%) were cured and 267 (73%) improved. Clinical success rates for first-line and second-line linezolid therapy were 79.9% and 83.6%, respectively. Failure rates for linezolid were higher for second-line versus first-line treatment (9.5% vs. 5.2%).
The clinical success rate was highest against MSSA (93.3%), followed by MRSA (83.8%). The average daily linezolid dose was 1,109 mg, and the mean treatment time was 5.1 days.
A total of eight patients (2.2%) reported linezolid-related adverse events, and four patients discontinued the medication because of them; none reported treatment-related serious adverse events. The low incidence of thrombocytopenia in the current study (two patients), compared with previous studies may have been related to avoidance of linezolid for at-risk patients as determined by clinicians, and the relatively short duration of linezolid use, the researchers wrote.
The study findings were limited by several factors, including the observational design and inability to compare the efficacy of different drugs; the small sample size; and the lack of data on drugs used prior to ICU admission, the researchers noted. Other limitations included the low detection rate of gram-positive bacteria and potential underreporting of adverse events.
However, the although clinicians will need to pay close attention to possible side effects and evaluate patient conditions on an individual basis before using linezolid in the clinic, they concluded.
The study was supported by grants from West China Hospital of Sichuan University. The researchers reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM THE JOURNAL OF INTENSIVE MEDICINE
Hypertension heightens risk for severe COVID-19, even in the fully vaxxed
Adults with hypertension who were vaccinated for COVID-19 with at least one booster were more than twice as likely as vaccinated and boosted individuals without hypertension to be hospitalized for severe COVID-19, according to data from more than 900 individuals.
“We were surprised to learn that many people who were hospitalized with COVID-19 had hypertension and no other risk factors,” said Susan Cheng, MD, MPH, director of the Institute for Research on Healthy Aging in the department of cardiology at the Smidt Heart Institute, Los Angeles, and a senior author of the study. “This is concerning when you consider that almost half of American adults have high blood pressure.”
COVID-19 vaccines demonstrated ability to reduce death and some of the most severe side effects from the infection in the early stages of the pandemic. Although the Omicron surge prompted recommendations for a third mRNA vaccine dose, “a proportion of individuals who received three mRNA vaccine doses still required hospitalization for COVID-19 during the Omicron surge,” and the characteristics associated with severe illness in vaccinated and boosted patients have not been explored, Joseph Ebinger, MD, of Cedars-Sinai Medical Center, Los Angeles, and colleagues wrote.
Previous research has shown an association between high blood pressure an increased risk for developing severe COVID-19 compared to several other chronic health conditions, including kidney disease, type 2 diabetes, chronic obstructive pulmonary disease, and heart failure, the researchers noted.
In a study published in Hypertension, the researchers identified 912 adults who received at least three doses of mRNA COVID-19 vaccine and were later diagnosed with COVID-19 during the surge in infections from the Omicron variant between December 2021 and April 2022.
A total of 145 of the individuals were hospitalized (16%); of these, 125 (86%) had hypertension.
Patients with hypertension were the most likely to be hospitalized, with an odds ratio of 2.9. In addition to high blood pressure, factors including older age (OR, 1.3), chronic kidney disease (OR, 2.2), prior myocardial infarction or heart failure (OR, 2.2), and longer time since the last vaccination and COVID-19 infection were associated with increased risk of hospitalization in a multivariate analysis.
However, the increased risk of severe illness and hospitalization associated with high blood pressure persisted, with an OR of 2.6, in the absence of comorbid conditions such as type 2 diabetes, kidney disease, and heart failure, the researchers emphasized.
“Although the mechanism for hypertension-associated COVID-19 risk remains unclear, prior studies have identified delayed SARS-CoV-2 viral clearance and prolonged inflammatory response among hypertensive patients, which may contribute to greater disease severity,” they wrote.
The findings were limited by several factors, including the use of data from a single center and lack of information on which Omicron variants and subvariants were behind the infections, the researchers noted.
However, the results highlight the need for more research on how to reduce the risks of severe COVID-19 in vulnerable populations, and on the mechanism for a potential connection between high blood pressure and severe COVID-19, they said.
Given the high prevalence of hypertension worldwide, increased understanding of the hypertension-specific risks and identification of individual and population-level risk reduction strategies will be important to the transition of COVID-19 from pandemic to endemic, they concluded.
Omicron changes the game
“When the pandemic initially started, many conditions were seen to increase risk for more severe COVID illness, and hypertension was one of those factors – and then things changed,” lead author Dr. Ebinger said in an interview. “First, vaccines arrived on the scene and substantially reduced risk of severe COVID for everyone who received them. Second, Omicron arrived and, while more transmissible, this variant has been less likely to cause severe COVID. On the one hand, we have vaccines and boosters that we want to think of as ‘the great equalizer’ when it comes to preexisting conditions. On the other hand, we have a dominant set of SARS-CoV-2 subvariants that seem less virulent in most people.
“Taken together, we have been hoping and even assuming that we have been doing pretty well with minimizing risks. Unfortunately, our study results indicate this is not exactly the case,” he said.
“Although vaccines and boosters appear to have equalized or minimized the risks of severe COVID for some people, this has not happened for others – even in the setting of the milder Omicron variant. Of individuals who were fully vaccinated and boosted, having hypertension increased the odds of needing to be hospitalized after getting infected with Omicron by 2.6-fold, even when accounting for or in the absence of having any major chronic disease that might otherwise predispose to more severe COVID-19 illness,” Dr. Ebinger added.
“So, while the originally seen risks of having obesity or diabetes seem to have been minimized during this current era of pandemic, the risk of having hypertension has persisted. We found this both surprising and concerning, because hypertension is very common and present in over half of people over age 50.”
Surprisingly, “we found that a fair number of people, even after being fully vaccinated plus a having gotten a booster, will not only catch Omicron but get sick enough to need hospital care,” Dr. Ebinger emphasized. “Moreover, it is not just older adults with major comorbid conditions who are vulnerable. Our data show that this can happen to an adult of any age and especially if a person has only hypertension and otherwise no major chronic disease.”
The first takeaway message for clinicians at this time is to raise awareness, Dr. Ebinger stressed in the interview. “We need to raise understanding around the fact that receiving three doses of vaccine may not prevent severe COVID-19 illness in everyone, even when the circulating viral variant is presumed to be causing only mild disease in most people. Moreover, the people who are most at risk are not whom we might think they are. They are not the sickest of the sick. They include people who might not have major conditions such as heart disease or kidney disease, but they do have hypertension.”
Second, “we need more research to understand out why there is this link between hypertension and excess risk for the more severe forms of COVID-19, despite it arising from a supposedly milder variant,” said Dr. Ebinger.
“Third, we need to determine how to reduce these risks, whether through more tailored vaccine regimens or novel therapeutics or a combination approach,” he said.
Looking ahead, “the biological mechanism underpinning the association between hypertension and severe COVID-19 remains underexplored. Future work should focus on understanding the factors linking hypertension to severe COVID-19, as this may elucidate both information on how SARS-CoV-2 effects the body and potential targets for intervention,” Dr. Ebinger added.
The study was supported in part by Cedars-Sinai Medical Center, the Erika J. Glazer Family Foundation and the National Institutes of Health. The researchers had no financial conflicts to disclose.
Adults with hypertension who were vaccinated for COVID-19 with at least one booster were more than twice as likely as vaccinated and boosted individuals without hypertension to be hospitalized for severe COVID-19, according to data from more than 900 individuals.
“We were surprised to learn that many people who were hospitalized with COVID-19 had hypertension and no other risk factors,” said Susan Cheng, MD, MPH, director of the Institute for Research on Healthy Aging in the department of cardiology at the Smidt Heart Institute, Los Angeles, and a senior author of the study. “This is concerning when you consider that almost half of American adults have high blood pressure.”
COVID-19 vaccines demonstrated ability to reduce death and some of the most severe side effects from the infection in the early stages of the pandemic. Although the Omicron surge prompted recommendations for a third mRNA vaccine dose, “a proportion of individuals who received three mRNA vaccine doses still required hospitalization for COVID-19 during the Omicron surge,” and the characteristics associated with severe illness in vaccinated and boosted patients have not been explored, Joseph Ebinger, MD, of Cedars-Sinai Medical Center, Los Angeles, and colleagues wrote.
Previous research has shown an association between high blood pressure an increased risk for developing severe COVID-19 compared to several other chronic health conditions, including kidney disease, type 2 diabetes, chronic obstructive pulmonary disease, and heart failure, the researchers noted.
In a study published in Hypertension, the researchers identified 912 adults who received at least three doses of mRNA COVID-19 vaccine and were later diagnosed with COVID-19 during the surge in infections from the Omicron variant between December 2021 and April 2022.
A total of 145 of the individuals were hospitalized (16%); of these, 125 (86%) had hypertension.
Patients with hypertension were the most likely to be hospitalized, with an odds ratio of 2.9. In addition to high blood pressure, factors including older age (OR, 1.3), chronic kidney disease (OR, 2.2), prior myocardial infarction or heart failure (OR, 2.2), and longer time since the last vaccination and COVID-19 infection were associated with increased risk of hospitalization in a multivariate analysis.
However, the increased risk of severe illness and hospitalization associated with high blood pressure persisted, with an OR of 2.6, in the absence of comorbid conditions such as type 2 diabetes, kidney disease, and heart failure, the researchers emphasized.
“Although the mechanism for hypertension-associated COVID-19 risk remains unclear, prior studies have identified delayed SARS-CoV-2 viral clearance and prolonged inflammatory response among hypertensive patients, which may contribute to greater disease severity,” they wrote.
The findings were limited by several factors, including the use of data from a single center and lack of information on which Omicron variants and subvariants were behind the infections, the researchers noted.
However, the results highlight the need for more research on how to reduce the risks of severe COVID-19 in vulnerable populations, and on the mechanism for a potential connection between high blood pressure and severe COVID-19, they said.
Given the high prevalence of hypertension worldwide, increased understanding of the hypertension-specific risks and identification of individual and population-level risk reduction strategies will be important to the transition of COVID-19 from pandemic to endemic, they concluded.
Omicron changes the game
“When the pandemic initially started, many conditions were seen to increase risk for more severe COVID illness, and hypertension was one of those factors – and then things changed,” lead author Dr. Ebinger said in an interview. “First, vaccines arrived on the scene and substantially reduced risk of severe COVID for everyone who received them. Second, Omicron arrived and, while more transmissible, this variant has been less likely to cause severe COVID. On the one hand, we have vaccines and boosters that we want to think of as ‘the great equalizer’ when it comes to preexisting conditions. On the other hand, we have a dominant set of SARS-CoV-2 subvariants that seem less virulent in most people.
“Taken together, we have been hoping and even assuming that we have been doing pretty well with minimizing risks. Unfortunately, our study results indicate this is not exactly the case,” he said.
“Although vaccines and boosters appear to have equalized or minimized the risks of severe COVID for some people, this has not happened for others – even in the setting of the milder Omicron variant. Of individuals who were fully vaccinated and boosted, having hypertension increased the odds of needing to be hospitalized after getting infected with Omicron by 2.6-fold, even when accounting for or in the absence of having any major chronic disease that might otherwise predispose to more severe COVID-19 illness,” Dr. Ebinger added.
“So, while the originally seen risks of having obesity or diabetes seem to have been minimized during this current era of pandemic, the risk of having hypertension has persisted. We found this both surprising and concerning, because hypertension is very common and present in over half of people over age 50.”
Surprisingly, “we found that a fair number of people, even after being fully vaccinated plus a having gotten a booster, will not only catch Omicron but get sick enough to need hospital care,” Dr. Ebinger emphasized. “Moreover, it is not just older adults with major comorbid conditions who are vulnerable. Our data show that this can happen to an adult of any age and especially if a person has only hypertension and otherwise no major chronic disease.”
The first takeaway message for clinicians at this time is to raise awareness, Dr. Ebinger stressed in the interview. “We need to raise understanding around the fact that receiving three doses of vaccine may not prevent severe COVID-19 illness in everyone, even when the circulating viral variant is presumed to be causing only mild disease in most people. Moreover, the people who are most at risk are not whom we might think they are. They are not the sickest of the sick. They include people who might not have major conditions such as heart disease or kidney disease, but they do have hypertension.”
Second, “we need more research to understand out why there is this link between hypertension and excess risk for the more severe forms of COVID-19, despite it arising from a supposedly milder variant,” said Dr. Ebinger.
“Third, we need to determine how to reduce these risks, whether through more tailored vaccine regimens or novel therapeutics or a combination approach,” he said.
Looking ahead, “the biological mechanism underpinning the association between hypertension and severe COVID-19 remains underexplored. Future work should focus on understanding the factors linking hypertension to severe COVID-19, as this may elucidate both information on how SARS-CoV-2 effects the body and potential targets for intervention,” Dr. Ebinger added.
The study was supported in part by Cedars-Sinai Medical Center, the Erika J. Glazer Family Foundation and the National Institutes of Health. The researchers had no financial conflicts to disclose.
Adults with hypertension who were vaccinated for COVID-19 with at least one booster were more than twice as likely as vaccinated and boosted individuals without hypertension to be hospitalized for severe COVID-19, according to data from more than 900 individuals.
“We were surprised to learn that many people who were hospitalized with COVID-19 had hypertension and no other risk factors,” said Susan Cheng, MD, MPH, director of the Institute for Research on Healthy Aging in the department of cardiology at the Smidt Heart Institute, Los Angeles, and a senior author of the study. “This is concerning when you consider that almost half of American adults have high blood pressure.”
COVID-19 vaccines demonstrated ability to reduce death and some of the most severe side effects from the infection in the early stages of the pandemic. Although the Omicron surge prompted recommendations for a third mRNA vaccine dose, “a proportion of individuals who received three mRNA vaccine doses still required hospitalization for COVID-19 during the Omicron surge,” and the characteristics associated with severe illness in vaccinated and boosted patients have not been explored, Joseph Ebinger, MD, of Cedars-Sinai Medical Center, Los Angeles, and colleagues wrote.
Previous research has shown an association between high blood pressure an increased risk for developing severe COVID-19 compared to several other chronic health conditions, including kidney disease, type 2 diabetes, chronic obstructive pulmonary disease, and heart failure, the researchers noted.
In a study published in Hypertension, the researchers identified 912 adults who received at least three doses of mRNA COVID-19 vaccine and were later diagnosed with COVID-19 during the surge in infections from the Omicron variant between December 2021 and April 2022.
A total of 145 of the individuals were hospitalized (16%); of these, 125 (86%) had hypertension.
Patients with hypertension were the most likely to be hospitalized, with an odds ratio of 2.9. In addition to high blood pressure, factors including older age (OR, 1.3), chronic kidney disease (OR, 2.2), prior myocardial infarction or heart failure (OR, 2.2), and longer time since the last vaccination and COVID-19 infection were associated with increased risk of hospitalization in a multivariate analysis.
However, the increased risk of severe illness and hospitalization associated with high blood pressure persisted, with an OR of 2.6, in the absence of comorbid conditions such as type 2 diabetes, kidney disease, and heart failure, the researchers emphasized.
“Although the mechanism for hypertension-associated COVID-19 risk remains unclear, prior studies have identified delayed SARS-CoV-2 viral clearance and prolonged inflammatory response among hypertensive patients, which may contribute to greater disease severity,” they wrote.
The findings were limited by several factors, including the use of data from a single center and lack of information on which Omicron variants and subvariants were behind the infections, the researchers noted.
However, the results highlight the need for more research on how to reduce the risks of severe COVID-19 in vulnerable populations, and on the mechanism for a potential connection between high blood pressure and severe COVID-19, they said.
Given the high prevalence of hypertension worldwide, increased understanding of the hypertension-specific risks and identification of individual and population-level risk reduction strategies will be important to the transition of COVID-19 from pandemic to endemic, they concluded.
Omicron changes the game
“When the pandemic initially started, many conditions were seen to increase risk for more severe COVID illness, and hypertension was one of those factors – and then things changed,” lead author Dr. Ebinger said in an interview. “First, vaccines arrived on the scene and substantially reduced risk of severe COVID for everyone who received them. Second, Omicron arrived and, while more transmissible, this variant has been less likely to cause severe COVID. On the one hand, we have vaccines and boosters that we want to think of as ‘the great equalizer’ when it comes to preexisting conditions. On the other hand, we have a dominant set of SARS-CoV-2 subvariants that seem less virulent in most people.
“Taken together, we have been hoping and even assuming that we have been doing pretty well with minimizing risks. Unfortunately, our study results indicate this is not exactly the case,” he said.
“Although vaccines and boosters appear to have equalized or minimized the risks of severe COVID for some people, this has not happened for others – even in the setting of the milder Omicron variant. Of individuals who were fully vaccinated and boosted, having hypertension increased the odds of needing to be hospitalized after getting infected with Omicron by 2.6-fold, even when accounting for or in the absence of having any major chronic disease that might otherwise predispose to more severe COVID-19 illness,” Dr. Ebinger added.
“So, while the originally seen risks of having obesity or diabetes seem to have been minimized during this current era of pandemic, the risk of having hypertension has persisted. We found this both surprising and concerning, because hypertension is very common and present in over half of people over age 50.”
Surprisingly, “we found that a fair number of people, even after being fully vaccinated plus a having gotten a booster, will not only catch Omicron but get sick enough to need hospital care,” Dr. Ebinger emphasized. “Moreover, it is not just older adults with major comorbid conditions who are vulnerable. Our data show that this can happen to an adult of any age and especially if a person has only hypertension and otherwise no major chronic disease.”
The first takeaway message for clinicians at this time is to raise awareness, Dr. Ebinger stressed in the interview. “We need to raise understanding around the fact that receiving three doses of vaccine may not prevent severe COVID-19 illness in everyone, even when the circulating viral variant is presumed to be causing only mild disease in most people. Moreover, the people who are most at risk are not whom we might think they are. They are not the sickest of the sick. They include people who might not have major conditions such as heart disease or kidney disease, but they do have hypertension.”
Second, “we need more research to understand out why there is this link between hypertension and excess risk for the more severe forms of COVID-19, despite it arising from a supposedly milder variant,” said Dr. Ebinger.
“Third, we need to determine how to reduce these risks, whether through more tailored vaccine regimens or novel therapeutics or a combination approach,” he said.
Looking ahead, “the biological mechanism underpinning the association between hypertension and severe COVID-19 remains underexplored. Future work should focus on understanding the factors linking hypertension to severe COVID-19, as this may elucidate both information on how SARS-CoV-2 effects the body and potential targets for intervention,” Dr. Ebinger added.
The study was supported in part by Cedars-Sinai Medical Center, the Erika J. Glazer Family Foundation and the National Institutes of Health. The researchers had no financial conflicts to disclose.
FROM HYPERTENSION
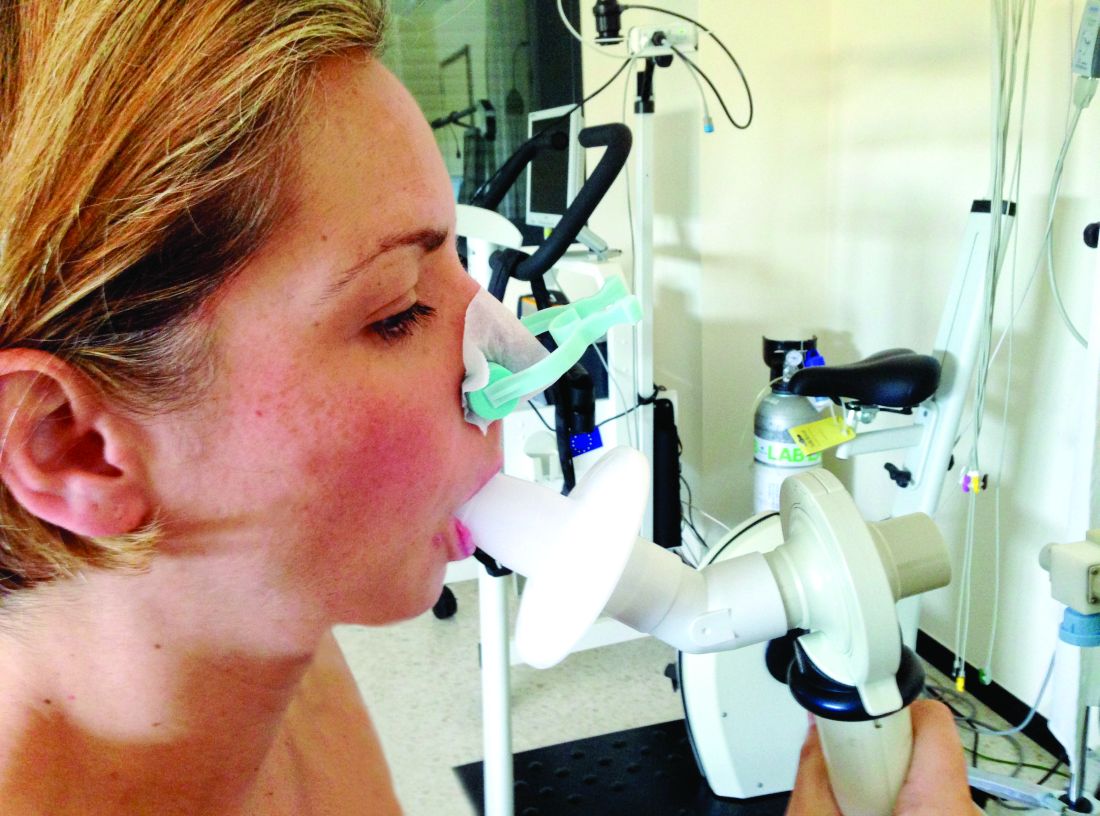
Race-specific spirometry may miss emphysema diagnoses
An overreliance on spirometry to identify emphysema led to missed cases in Black individuals, particularly men, based on a secondary data analysis of 2,674 people.
“Over the last few years, there has been growing debate around the use of race adjustment in diagnostic algorithms and equations commonly used in medicine,” lead author Gabrielle Yi-Hui Liu, MD, said in an interview. “Whereas, previously it was common to accept racial or ethnic differences in clinical measures and outcomes as inherent differences among populations, there is now more recognition of how racism, socioeconomic status, and environmental exposures can cause these racial differences. Our initial interest in this study was to examine how the use of race-specific spirometry reference equations, and the use of spirometry in general, may be contributing to racial disparities.”
“Previous studies have suggested that the use of race-specific equations in spirometry can exacerbate racial inequities in healthcare outcomes by under-recognition of early disease in Black adults, and this study adds to that evidence,” said Suman Pal, MBBS, of the University of New Mexico, Albuquerque, in an interview.
“By examining the crucial ways in which systemic factors in medicine, such as race-specific equations, exacerbate racial inequities in healthcare, this study is a timely analysis in a moment of national reckoning of structural racism,” said Dr. Pal, who was not involved in the study.
In a study published in Annals of Internal Medicine, Dr. Liu and colleagues at Northwestern University, Chicago, conducted a secondary analysis of data from the CARDIA Lung study (Coronary Artery Risk Development In Young Adults).
The primary outcome of the study was the prevalence of emphysema among participants with various measures of normal spirometry results, stratified by sex and race. The normal results included an forced expiratory volume in 1 second (FEV1)–forced vital capacity (FVC) ratio greater than or equal to 0.7 or greater than or equal to the lower limit of normal. The participants also were stratified by FEV1 percent predicted, using race-specific reference equations, for FEV1 between 80% and 99% of predicted, or an FEV1 between 100% and 120% of predicted.
The study population included 485 Black men, 762 Black women, 659 White men, and 768 White women who received both a CT scan (in 2010-2011) and spirometry (obtained in 2015-2016) in the CARDIA study. The mean age of the participants at the spirometry exam was 55 years.
A total of 5.3% of the participants had emphysema after stratifying by FEV1-FVC ratio. The prevalence was significantly higher for Black men, compared with White men (12.3% vs. 4.0%; relative risk, 3.0), and for Black women, compared with White women (5.0% vs. 2.6%; RR, 1.9).
The association between Black race and emphysema risk persisted but decreased when the researchers used a race-neutral estimate.
When the participants were stratified by race-specific FEV1 percent predicted, 6.5% of individuals with a race-specific FEV1 between 80% and 99% had emphysema. After controlling for factors including age and smoking, emphysema was significantly more prevalent in Black men versus White men (15.5% vs. 4.0%) and in Black women, compared with White women (6.6% vs. 3.4%).
The racial difference persisted in men with a race-specific FEV1 between 100% and 120% of predicted. Of these, 4.0% had emphysema. The prevalence was significantly higher in Black men, compared with White men (13.9% vs. 2.2%), but similar between Black women and White women (2.6% vs. 2.0%).
The use of race-neutral equations reduced, but did not eliminate, these disparities, the researchers said.
The findings were limited by the lack of CT imaging data from the same visit as the final spirometry collection, the researchers noted. “Given that imaging was obtained 5 years before spirometry and emphysema is an irreversible finding, this may have led to an overall underestimation of the prevalence of emphysema.”
Spirometry alone misses cases
“We were surprised by the substantial rates of emphysema we saw among Black men in our cohort with normal spirometry,” Dr. Liu said in an interview. “We did not expect to find than more than one in eight Black men with an FEV1 between 100% and 120% predicted would have emphysema – a rate more than six times higher than White men with the same range of FEV1.”
“One takeaway is that we are likely missing a lot of people with impaired respiratory health or true lung disease by only using spirometry to diagnose COPD,” said Dr. Liu. In clinical practice, “physicians should consider ordering CT scans on patients with normal spirometry who have respiratory symptoms such as cough or shortness of breath. If emphysema is found, physicians should discuss mitigating any potential risk factors and consider the use of COPD medications such as inhalers.
“Our findings also support using race-neutral reference equations to interpret spirometry instead of race-specific equations. Racial disparities in rates of emphysema among those with ‘normal’ FEV1 [between 80% and 120% predicted], were attenuated or eliminated when race-neutral equations were used to calculate FEV1. This suggests that race-specific equations are normalizing worse lung health in Black adults,” Dr. Liu explained.
“We need to continue research into additional tools that can be used to assess respiratory health and diagnose COPD, while keeping in mind how these tools may affect racial disparities,” said Dr. Liu. “Our study suggests that our reliance on spirometry measures such as FEV1/FVC ratio and FEV1 is missing a number of people with respiratory symptoms and CT evidence of lung disease, and that this is disproportionately affecting Black adults in the United States.” Looking ahead, “it is important to find better tools to identify people with impaired respiratory health or early manifestations of disease so we can intercept chronic lung disease before it becomes clinically apparent and patients have sustained significant lung damage.”
The CARDIA study was supported by the National Heart, Lung, and Blood Institute in collaboration with the University of Alabama at Birmingham, Northwestern University, the University of Minnesota, and the Kaiser Foundation Research Institute. Dr. Liu was supported by a grant from the National Institutes of Health. The researchers had no financial conflicts to disclose. Dr. Pal had no financial conflicts to disclose.
*This article was updated 7/22/2022.
An overreliance on spirometry to identify emphysema led to missed cases in Black individuals, particularly men, based on a secondary data analysis of 2,674 people.
“Over the last few years, there has been growing debate around the use of race adjustment in diagnostic algorithms and equations commonly used in medicine,” lead author Gabrielle Yi-Hui Liu, MD, said in an interview. “Whereas, previously it was common to accept racial or ethnic differences in clinical measures and outcomes as inherent differences among populations, there is now more recognition of how racism, socioeconomic status, and environmental exposures can cause these racial differences. Our initial interest in this study was to examine how the use of race-specific spirometry reference equations, and the use of spirometry in general, may be contributing to racial disparities.”
“Previous studies have suggested that the use of race-specific equations in spirometry can exacerbate racial inequities in healthcare outcomes by under-recognition of early disease in Black adults, and this study adds to that evidence,” said Suman Pal, MBBS, of the University of New Mexico, Albuquerque, in an interview.
“By examining the crucial ways in which systemic factors in medicine, such as race-specific equations, exacerbate racial inequities in healthcare, this study is a timely analysis in a moment of national reckoning of structural racism,” said Dr. Pal, who was not involved in the study.
In a study published in Annals of Internal Medicine, Dr. Liu and colleagues at Northwestern University, Chicago, conducted a secondary analysis of data from the CARDIA Lung study (Coronary Artery Risk Development In Young Adults).
The primary outcome of the study was the prevalence of emphysema among participants with various measures of normal spirometry results, stratified by sex and race. The normal results included an forced expiratory volume in 1 second (FEV1)–forced vital capacity (FVC) ratio greater than or equal to 0.7 or greater than or equal to the lower limit of normal. The participants also were stratified by FEV1 percent predicted, using race-specific reference equations, for FEV1 between 80% and 99% of predicted, or an FEV1 between 100% and 120% of predicted.
The study population included 485 Black men, 762 Black women, 659 White men, and 768 White women who received both a CT scan (in 2010-2011) and spirometry (obtained in 2015-2016) in the CARDIA study. The mean age of the participants at the spirometry exam was 55 years.
A total of 5.3% of the participants had emphysema after stratifying by FEV1-FVC ratio. The prevalence was significantly higher for Black men, compared with White men (12.3% vs. 4.0%; relative risk, 3.0), and for Black women, compared with White women (5.0% vs. 2.6%; RR, 1.9).
The association between Black race and emphysema risk persisted but decreased when the researchers used a race-neutral estimate.
When the participants were stratified by race-specific FEV1 percent predicted, 6.5% of individuals with a race-specific FEV1 between 80% and 99% had emphysema. After controlling for factors including age and smoking, emphysema was significantly more prevalent in Black men versus White men (15.5% vs. 4.0%) and in Black women, compared with White women (6.6% vs. 3.4%).
The racial difference persisted in men with a race-specific FEV1 between 100% and 120% of predicted. Of these, 4.0% had emphysema. The prevalence was significantly higher in Black men, compared with White men (13.9% vs. 2.2%), but similar between Black women and White women (2.6% vs. 2.0%).
The use of race-neutral equations reduced, but did not eliminate, these disparities, the researchers said.
The findings were limited by the lack of CT imaging data from the same visit as the final spirometry collection, the researchers noted. “Given that imaging was obtained 5 years before spirometry and emphysema is an irreversible finding, this may have led to an overall underestimation of the prevalence of emphysema.”
Spirometry alone misses cases
“We were surprised by the substantial rates of emphysema we saw among Black men in our cohort with normal spirometry,” Dr. Liu said in an interview. “We did not expect to find than more than one in eight Black men with an FEV1 between 100% and 120% predicted would have emphysema – a rate more than six times higher than White men with the same range of FEV1.”
“One takeaway is that we are likely missing a lot of people with impaired respiratory health or true lung disease by only using spirometry to diagnose COPD,” said Dr. Liu. In clinical practice, “physicians should consider ordering CT scans on patients with normal spirometry who have respiratory symptoms such as cough or shortness of breath. If emphysema is found, physicians should discuss mitigating any potential risk factors and consider the use of COPD medications such as inhalers.
“Our findings also support using race-neutral reference equations to interpret spirometry instead of race-specific equations. Racial disparities in rates of emphysema among those with ‘normal’ FEV1 [between 80% and 120% predicted], were attenuated or eliminated when race-neutral equations were used to calculate FEV1. This suggests that race-specific equations are normalizing worse lung health in Black adults,” Dr. Liu explained.
“We need to continue research into additional tools that can be used to assess respiratory health and diagnose COPD, while keeping in mind how these tools may affect racial disparities,” said Dr. Liu. “Our study suggests that our reliance on spirometry measures such as FEV1/FVC ratio and FEV1 is missing a number of people with respiratory symptoms and CT evidence of lung disease, and that this is disproportionately affecting Black adults in the United States.” Looking ahead, “it is important to find better tools to identify people with impaired respiratory health or early manifestations of disease so we can intercept chronic lung disease before it becomes clinically apparent and patients have sustained significant lung damage.”
The CARDIA study was supported by the National Heart, Lung, and Blood Institute in collaboration with the University of Alabama at Birmingham, Northwestern University, the University of Minnesota, and the Kaiser Foundation Research Institute. Dr. Liu was supported by a grant from the National Institutes of Health. The researchers had no financial conflicts to disclose. Dr. Pal had no financial conflicts to disclose.
*This article was updated 7/22/2022.
An overreliance on spirometry to identify emphysema led to missed cases in Black individuals, particularly men, based on a secondary data analysis of 2,674 people.
“Over the last few years, there has been growing debate around the use of race adjustment in diagnostic algorithms and equations commonly used in medicine,” lead author Gabrielle Yi-Hui Liu, MD, said in an interview. “Whereas, previously it was common to accept racial or ethnic differences in clinical measures and outcomes as inherent differences among populations, there is now more recognition of how racism, socioeconomic status, and environmental exposures can cause these racial differences. Our initial interest in this study was to examine how the use of race-specific spirometry reference equations, and the use of spirometry in general, may be contributing to racial disparities.”
“Previous studies have suggested that the use of race-specific equations in spirometry can exacerbate racial inequities in healthcare outcomes by under-recognition of early disease in Black adults, and this study adds to that evidence,” said Suman Pal, MBBS, of the University of New Mexico, Albuquerque, in an interview.
“By examining the crucial ways in which systemic factors in medicine, such as race-specific equations, exacerbate racial inequities in healthcare, this study is a timely analysis in a moment of national reckoning of structural racism,” said Dr. Pal, who was not involved in the study.
In a study published in Annals of Internal Medicine, Dr. Liu and colleagues at Northwestern University, Chicago, conducted a secondary analysis of data from the CARDIA Lung study (Coronary Artery Risk Development In Young Adults).
The primary outcome of the study was the prevalence of emphysema among participants with various measures of normal spirometry results, stratified by sex and race. The normal results included an forced expiratory volume in 1 second (FEV1)–forced vital capacity (FVC) ratio greater than or equal to 0.7 or greater than or equal to the lower limit of normal. The participants also were stratified by FEV1 percent predicted, using race-specific reference equations, for FEV1 between 80% and 99% of predicted, or an FEV1 between 100% and 120% of predicted.
The study population included 485 Black men, 762 Black women, 659 White men, and 768 White women who received both a CT scan (in 2010-2011) and spirometry (obtained in 2015-2016) in the CARDIA study. The mean age of the participants at the spirometry exam was 55 years.
A total of 5.3% of the participants had emphysema after stratifying by FEV1-FVC ratio. The prevalence was significantly higher for Black men, compared with White men (12.3% vs. 4.0%; relative risk, 3.0), and for Black women, compared with White women (5.0% vs. 2.6%; RR, 1.9).
The association between Black race and emphysema risk persisted but decreased when the researchers used a race-neutral estimate.
When the participants were stratified by race-specific FEV1 percent predicted, 6.5% of individuals with a race-specific FEV1 between 80% and 99% had emphysema. After controlling for factors including age and smoking, emphysema was significantly more prevalent in Black men versus White men (15.5% vs. 4.0%) and in Black women, compared with White women (6.6% vs. 3.4%).
The racial difference persisted in men with a race-specific FEV1 between 100% and 120% of predicted. Of these, 4.0% had emphysema. The prevalence was significantly higher in Black men, compared with White men (13.9% vs. 2.2%), but similar between Black women and White women (2.6% vs. 2.0%).
The use of race-neutral equations reduced, but did not eliminate, these disparities, the researchers said.
The findings were limited by the lack of CT imaging data from the same visit as the final spirometry collection, the researchers noted. “Given that imaging was obtained 5 years before spirometry and emphysema is an irreversible finding, this may have led to an overall underestimation of the prevalence of emphysema.”
Spirometry alone misses cases
“We were surprised by the substantial rates of emphysema we saw among Black men in our cohort with normal spirometry,” Dr. Liu said in an interview. “We did not expect to find than more than one in eight Black men with an FEV1 between 100% and 120% predicted would have emphysema – a rate more than six times higher than White men with the same range of FEV1.”
“One takeaway is that we are likely missing a lot of people with impaired respiratory health or true lung disease by only using spirometry to diagnose COPD,” said Dr. Liu. In clinical practice, “physicians should consider ordering CT scans on patients with normal spirometry who have respiratory symptoms such as cough or shortness of breath. If emphysema is found, physicians should discuss mitigating any potential risk factors and consider the use of COPD medications such as inhalers.
“Our findings also support using race-neutral reference equations to interpret spirometry instead of race-specific equations. Racial disparities in rates of emphysema among those with ‘normal’ FEV1 [between 80% and 120% predicted], were attenuated or eliminated when race-neutral equations were used to calculate FEV1. This suggests that race-specific equations are normalizing worse lung health in Black adults,” Dr. Liu explained.
“We need to continue research into additional tools that can be used to assess respiratory health and diagnose COPD, while keeping in mind how these tools may affect racial disparities,” said Dr. Liu. “Our study suggests that our reliance on spirometry measures such as FEV1/FVC ratio and FEV1 is missing a number of people with respiratory symptoms and CT evidence of lung disease, and that this is disproportionately affecting Black adults in the United States.” Looking ahead, “it is important to find better tools to identify people with impaired respiratory health or early manifestations of disease so we can intercept chronic lung disease before it becomes clinically apparent and patients have sustained significant lung damage.”
The CARDIA study was supported by the National Heart, Lung, and Blood Institute in collaboration with the University of Alabama at Birmingham, Northwestern University, the University of Minnesota, and the Kaiser Foundation Research Institute. Dr. Liu was supported by a grant from the National Institutes of Health. The researchers had no financial conflicts to disclose. Dr. Pal had no financial conflicts to disclose.
*This article was updated 7/22/2022.
FROM ANNALS OF INTERNAL MEDICINE
Berdazimer gel beats vehicle for molluscum contagiosum in phase 3 study
Treatment with .
Molluscum contagiosum (MC) remains a common infection that, despite being self-limiting, may persist for months or years, and is associated with quality of life concerns and the need for ongoing therapy, wrote John C. Browning, MD, of Texas Dermatology and Laser Specialists, San Antonio, and colleagues, who conducted the phase 3 randomized study.
The infection is most common in children aged 1-14 years, and treatment may be needed in part to avoid infecting peers and family members, they said. No treatments for molluscum are currently approved by the Food and Drug Administration.
In the study, which was published in JAMA Dermatology, the researchers randomized 444 patients to berdazimer gel 10.3% and 447 to a placebo gel, applied once daily in a thin layer on all MC lesions for 12 weeks. The study was conducted at 55 clinics across the United States between Sept. 1, 2020, and July 21, 2021. The mean age of the patients was about 6.5 years (range was 0.9-49 years), and about 85% were White. Participants had 3-70 raised MC lesions; those with sexually transmitted MC or MC in the periocular area were excluded. The primary endpoint was complete clearance of MC lesions after 12 weeks of treatment. At 12 weeks, significantly more patients treated with berdazimer gel achieved complete clearance than those on vehicle (32.4% vs. 19.7%; P < .001). A total of 64 (14.4%) patients in the berdazimer group discontinued treatment because of MC clearance, compared with 40 patients (8.9%) in the vehicle group.
Most adverse events were mild or moderate, and rates of adverse events resulting in treatment discontinuation were low overall for both groups; the most common adverse events were application-site pain and erythema, which were mostly mild. Overall, 4.1% of berdazimer-treated patients and 0.7% of placebo patients discontinued the study because of adverse events.
The study findings were limited by several factors, including the small number of patients in subgroups for race, ethnicity, and age; and the lack of data on patients with sexually transmitted MC and on concomitant use with other topical MC therapies, the researchers noted.
However, the results represent the largest randomized clinical trial of berdazimer 10.3% to date, and support its potential as a first-line therapy for MC patients aged 6 months and older, according to the authors. “Berdazimer is under consideration as a first in-class therapeutic agent for MC and may provide a topical prescription alternative to other therapies used for this highly contagious and psychosocially challenging skin condition,” they said.
Having a reliable, steroid-free, safe, and efficacious medication to treat molluscum in the pediatric population, as early as age 6 months, that can be used at home would “change the whole therapeutic paradigm,” one of the study authors, Adelaide Hebert, MD, said in an interview at the Society for Pediatric Dermatology annual meeting in July, where she presented phase 2 data on berdazimer gel. “This is a common problem and the rate of infections among siblings if it goes untreated is 41%. Affected kids have a sense of isolation; they don’t get invited to swimming parties.”
The lack of a safe and effective topical therapy “has been challenging,” added Dr. Hebert, professor of dermatology and pediatrics, and chief of pediatric dermatology at the University of Texas, Houston. She noted that treatments that have been used but have not been successful include imiquimod. “I’m not impressed with tretinoin,” although it is prescribed for MC, and the most common treatment prescribed by pediatricians for molluscum – mupirocin – is “usually not effective,” she said.
Another MC treatment in trials
Another investigative treatment for molluscum contagiosum, VP-102, a drug-device combination of cantharidin 0.7% administered through a single-use precision applicator, has been evaluated in phase 3 studies of patients with MC aged 2 years and older. The results of two phase 3 studies were published in 2020.
In May 2022, Verrica Pharmaceuticals, which is developing VP-102, announced that Food and Drug Administration approval had been delayed because of deficiencies identified at a contract manufacturing organization, and that the company was working with the agency to bring VP-102 to the market as soon as possible.
A step in the right direction
Although MC is self-resolving, cases last an average of 13.5 months, and “many families look to fast-forward their child’s experience with the infection,” Vikash S. Oza, MD, a pediatric dermatologist at New York University, New York, wrote in an editorial that accompanied the berdazimer study.
“To truly create a paradigm shift in the decision to treat MC, a therapeutic treatment would need to be developed that would lead to resolution of the infection over a short time frame (ideally, weeks) with minimal discomfort,” Dr. Oza noted. “Both VP-102 and berdazimer gel, 10.3%, have the potential to be the first-ever MC therapies approved by the U.S. Food and Drug Administration,” and families seeking to reduce MC in visible areas would welcome this option for a home therapy, he said.
However, Dr. Oza emphasized that potential barriers to widespread use of these therapies include whether the efficacy can be maintained in patients who fail to comply with daily application, and the ongoing need for office-based therapy to manage sexually transmitted MC in adults and periocular and perianal MC in children. The study was funded by Novan. Lead author Dr. Browning disclosed grants from Novan during the conduct of the study; Dr. Hebert reported grants from the University of Texas Health Science Center McGovern Medical School-Houston during the conduct of the study. Disclosures of other authors included having reported equity in Novan during the conduct of the study and receiving a grant from Novan. Dr. Oza had no financial conflicts to disclose.
Treatment with .
Molluscum contagiosum (MC) remains a common infection that, despite being self-limiting, may persist for months or years, and is associated with quality of life concerns and the need for ongoing therapy, wrote John C. Browning, MD, of Texas Dermatology and Laser Specialists, San Antonio, and colleagues, who conducted the phase 3 randomized study.
The infection is most common in children aged 1-14 years, and treatment may be needed in part to avoid infecting peers and family members, they said. No treatments for molluscum are currently approved by the Food and Drug Administration.
In the study, which was published in JAMA Dermatology, the researchers randomized 444 patients to berdazimer gel 10.3% and 447 to a placebo gel, applied once daily in a thin layer on all MC lesions for 12 weeks. The study was conducted at 55 clinics across the United States between Sept. 1, 2020, and July 21, 2021. The mean age of the patients was about 6.5 years (range was 0.9-49 years), and about 85% were White. Participants had 3-70 raised MC lesions; those with sexually transmitted MC or MC in the periocular area were excluded. The primary endpoint was complete clearance of MC lesions after 12 weeks of treatment. At 12 weeks, significantly more patients treated with berdazimer gel achieved complete clearance than those on vehicle (32.4% vs. 19.7%; P < .001). A total of 64 (14.4%) patients in the berdazimer group discontinued treatment because of MC clearance, compared with 40 patients (8.9%) in the vehicle group.
Most adverse events were mild or moderate, and rates of adverse events resulting in treatment discontinuation were low overall for both groups; the most common adverse events were application-site pain and erythema, which were mostly mild. Overall, 4.1% of berdazimer-treated patients and 0.7% of placebo patients discontinued the study because of adverse events.
The study findings were limited by several factors, including the small number of patients in subgroups for race, ethnicity, and age; and the lack of data on patients with sexually transmitted MC and on concomitant use with other topical MC therapies, the researchers noted.
However, the results represent the largest randomized clinical trial of berdazimer 10.3% to date, and support its potential as a first-line therapy for MC patients aged 6 months and older, according to the authors. “Berdazimer is under consideration as a first in-class therapeutic agent for MC and may provide a topical prescription alternative to other therapies used for this highly contagious and psychosocially challenging skin condition,” they said.
Having a reliable, steroid-free, safe, and efficacious medication to treat molluscum in the pediatric population, as early as age 6 months, that can be used at home would “change the whole therapeutic paradigm,” one of the study authors, Adelaide Hebert, MD, said in an interview at the Society for Pediatric Dermatology annual meeting in July, where she presented phase 2 data on berdazimer gel. “This is a common problem and the rate of infections among siblings if it goes untreated is 41%. Affected kids have a sense of isolation; they don’t get invited to swimming parties.”
The lack of a safe and effective topical therapy “has been challenging,” added Dr. Hebert, professor of dermatology and pediatrics, and chief of pediatric dermatology at the University of Texas, Houston. She noted that treatments that have been used but have not been successful include imiquimod. “I’m not impressed with tretinoin,” although it is prescribed for MC, and the most common treatment prescribed by pediatricians for molluscum – mupirocin – is “usually not effective,” she said.
Another MC treatment in trials
Another investigative treatment for molluscum contagiosum, VP-102, a drug-device combination of cantharidin 0.7% administered through a single-use precision applicator, has been evaluated in phase 3 studies of patients with MC aged 2 years and older. The results of two phase 3 studies were published in 2020.
In May 2022, Verrica Pharmaceuticals, which is developing VP-102, announced that Food and Drug Administration approval had been delayed because of deficiencies identified at a contract manufacturing organization, and that the company was working with the agency to bring VP-102 to the market as soon as possible.
A step in the right direction
Although MC is self-resolving, cases last an average of 13.5 months, and “many families look to fast-forward their child’s experience with the infection,” Vikash S. Oza, MD, a pediatric dermatologist at New York University, New York, wrote in an editorial that accompanied the berdazimer study.
“To truly create a paradigm shift in the decision to treat MC, a therapeutic treatment would need to be developed that would lead to resolution of the infection over a short time frame (ideally, weeks) with minimal discomfort,” Dr. Oza noted. “Both VP-102 and berdazimer gel, 10.3%, have the potential to be the first-ever MC therapies approved by the U.S. Food and Drug Administration,” and families seeking to reduce MC in visible areas would welcome this option for a home therapy, he said.
However, Dr. Oza emphasized that potential barriers to widespread use of these therapies include whether the efficacy can be maintained in patients who fail to comply with daily application, and the ongoing need for office-based therapy to manage sexually transmitted MC in adults and periocular and perianal MC in children. The study was funded by Novan. Lead author Dr. Browning disclosed grants from Novan during the conduct of the study; Dr. Hebert reported grants from the University of Texas Health Science Center McGovern Medical School-Houston during the conduct of the study. Disclosures of other authors included having reported equity in Novan during the conduct of the study and receiving a grant from Novan. Dr. Oza had no financial conflicts to disclose.
Treatment with .
Molluscum contagiosum (MC) remains a common infection that, despite being self-limiting, may persist for months or years, and is associated with quality of life concerns and the need for ongoing therapy, wrote John C. Browning, MD, of Texas Dermatology and Laser Specialists, San Antonio, and colleagues, who conducted the phase 3 randomized study.
The infection is most common in children aged 1-14 years, and treatment may be needed in part to avoid infecting peers and family members, they said. No treatments for molluscum are currently approved by the Food and Drug Administration.
In the study, which was published in JAMA Dermatology, the researchers randomized 444 patients to berdazimer gel 10.3% and 447 to a placebo gel, applied once daily in a thin layer on all MC lesions for 12 weeks. The study was conducted at 55 clinics across the United States between Sept. 1, 2020, and July 21, 2021. The mean age of the patients was about 6.5 years (range was 0.9-49 years), and about 85% were White. Participants had 3-70 raised MC lesions; those with sexually transmitted MC or MC in the periocular area were excluded. The primary endpoint was complete clearance of MC lesions after 12 weeks of treatment. At 12 weeks, significantly more patients treated with berdazimer gel achieved complete clearance than those on vehicle (32.4% vs. 19.7%; P < .001). A total of 64 (14.4%) patients in the berdazimer group discontinued treatment because of MC clearance, compared with 40 patients (8.9%) in the vehicle group.
Most adverse events were mild or moderate, and rates of adverse events resulting in treatment discontinuation were low overall for both groups; the most common adverse events were application-site pain and erythema, which were mostly mild. Overall, 4.1% of berdazimer-treated patients and 0.7% of placebo patients discontinued the study because of adverse events.
The study findings were limited by several factors, including the small number of patients in subgroups for race, ethnicity, and age; and the lack of data on patients with sexually transmitted MC and on concomitant use with other topical MC therapies, the researchers noted.
However, the results represent the largest randomized clinical trial of berdazimer 10.3% to date, and support its potential as a first-line therapy for MC patients aged 6 months and older, according to the authors. “Berdazimer is under consideration as a first in-class therapeutic agent for MC and may provide a topical prescription alternative to other therapies used for this highly contagious and psychosocially challenging skin condition,” they said.
Having a reliable, steroid-free, safe, and efficacious medication to treat molluscum in the pediatric population, as early as age 6 months, that can be used at home would “change the whole therapeutic paradigm,” one of the study authors, Adelaide Hebert, MD, said in an interview at the Society for Pediatric Dermatology annual meeting in July, where she presented phase 2 data on berdazimer gel. “This is a common problem and the rate of infections among siblings if it goes untreated is 41%. Affected kids have a sense of isolation; they don’t get invited to swimming parties.”
The lack of a safe and effective topical therapy “has been challenging,” added Dr. Hebert, professor of dermatology and pediatrics, and chief of pediatric dermatology at the University of Texas, Houston. She noted that treatments that have been used but have not been successful include imiquimod. “I’m not impressed with tretinoin,” although it is prescribed for MC, and the most common treatment prescribed by pediatricians for molluscum – mupirocin – is “usually not effective,” she said.
Another MC treatment in trials
Another investigative treatment for molluscum contagiosum, VP-102, a drug-device combination of cantharidin 0.7% administered through a single-use precision applicator, has been evaluated in phase 3 studies of patients with MC aged 2 years and older. The results of two phase 3 studies were published in 2020.
In May 2022, Verrica Pharmaceuticals, which is developing VP-102, announced that Food and Drug Administration approval had been delayed because of deficiencies identified at a contract manufacturing organization, and that the company was working with the agency to bring VP-102 to the market as soon as possible.
A step in the right direction
Although MC is self-resolving, cases last an average of 13.5 months, and “many families look to fast-forward their child’s experience with the infection,” Vikash S. Oza, MD, a pediatric dermatologist at New York University, New York, wrote in an editorial that accompanied the berdazimer study.
“To truly create a paradigm shift in the decision to treat MC, a therapeutic treatment would need to be developed that would lead to resolution of the infection over a short time frame (ideally, weeks) with minimal discomfort,” Dr. Oza noted. “Both VP-102 and berdazimer gel, 10.3%, have the potential to be the first-ever MC therapies approved by the U.S. Food and Drug Administration,” and families seeking to reduce MC in visible areas would welcome this option for a home therapy, he said.
However, Dr. Oza emphasized that potential barriers to widespread use of these therapies include whether the efficacy can be maintained in patients who fail to comply with daily application, and the ongoing need for office-based therapy to manage sexually transmitted MC in adults and periocular and perianal MC in children. The study was funded by Novan. Lead author Dr. Browning disclosed grants from Novan during the conduct of the study; Dr. Hebert reported grants from the University of Texas Health Science Center McGovern Medical School-Houston during the conduct of the study. Disclosures of other authors included having reported equity in Novan during the conduct of the study and receiving a grant from Novan. Dr. Oza had no financial conflicts to disclose.
FROM JAMA DERMATOLOGY
Moderate drinking shows more benefit for older vs. younger adults
The health risks and benefits of moderate alcohol consumption are complex and remain a hot topic of debate. The data suggest that small amounts of alcohol may reduce the risk of certain health outcomes over time, but increase the risk of others, wrote Dana Bryazka, MS, a researcher at the Institute for Health Metrics and Evaluation (IHME) at the University of Washington, Seattle, and colleagues, in a paper published in the Lancet.
“The amount of alcohol that minimizes health loss is likely to depend on the distribution of underlying causes of disease burden in a given population. Since this distribution varies widely by geography, age, sex, and time, the level of alcohol consumption associated with the lowest risk to health would depend on the age structure and disease composition of that population,” the researchers wrote.
“We estimate that 1.78 million people worldwide died due to alcohol use in 2020,” Ms. Bryazka said in an interview. “It is important that alcohol consumption guidelines and policies are updated to minimize this harm, particularly in the populations at greatest risk,” she said.
“Existing alcohol consumption guidelines frequently vary by sex, with higher consumption thresholds set for males compared to females. Interestingly, with the currently available data we do not see evidence that risk of alcohol use varies by sex,” she noted.
Methods and results
In the study, the researchers conducted a systematic analysis of burden-weighted dose-response relative risk curves across 22 health outcomes. They used disease rates from the Global Burden of Diseases, Injuries, and Risk Factors Study (GBD) 2020 for the years 1990-2020 for 21 regions, including 204 countries and territories. The data were analyzed by 5-year age group, sex, and year for individuals aged 15-95 years and older. The researchers estimated the theoretical minimum risk exposure level (TMREL) and nondrinker equivalent (NDE), meaning the amount of alcohol at which the health risk equals that of a nondrinker.
One standard drink was defined as 10 g of pure alcohol, equivalent to a small glass of red wine (100 mL or 3.4 fluid ounces) at 13% alcohol by volume, a can or bottle of beer (375 mL or 12 fluid ounces) at 3.5% alcohol by volume, or a shot of whiskey or other spirits (30 mL or 1.0 fluid ounces) at 40% alcohol by volume.
Overall, the TMREL was low regardless of age, sex, time, or geography, and varied from 0 to 1.87 standard drinks per day. However, it was lowest for males aged 15-39 years (0.136 drinks per day) and only slightly higher for females aged 15-39 (0.273), representing 1-2 tenths of a standard drink.
For adults aged 40 and older without any underlying health conditions, drinking a small amount of alcohol may provide some benefits, such as reducing the risk of ischemic heart disease, stroke, and diabetes, the researchers noted. In general, for individuals aged 40-64 years, TMRELs ranged from about half a standard drink per day (0.527 drinks for males and 0.562 standard drinks per day for females) to almost two standard drinks (1.69 standard drinks per day for males and 1.82 for females). For those older than 65 years, the TMRELs represented just over 3 standard drinks per day (3.19 for males and 3.51 for females). For individuals aged 40 years and older, the distribution of disease burden varied by region, but was J-shaped across all regions, the researchers noted.
The researchers also found that those individuals consuming harmful amounts of alcohol were most likely to be aged 15-39 (59.1%) and male (76.9%).
The study findings were limited by several factors including the observational design and lack of data on drinking patterns, such as binge drinking, the researchers noted. Other limitations include the lack of data reflecting patterns of alcohol consumption during the COVID-19 pandemic, and exclusion of outcomes often associated with alcohol use, such as depression, anxiety, and dementia, that might reduce estimates of TMREL and NDE.
However, the results add to the ongoing discussion of the relationship between moderate alcohol consumption and health, the researchers said.
“The findings of this study support the development of tailored guidelines and recommendations on alcohol consumption by age and across regions and highlight that existing low consumption thresholds are too high for younger populations in all regions,” they concluded.
Consider individual factors when counseling patients
The takeaway message for primary care is that alcohol consumed in moderation can reduce the risk of ischemic heart disease, stroke, and diabetes, Ms. Bryazka noted. “However, it also increases the risk of many cancers, intentional and unintentional injuries, and infectious diseases like tuberculosis,” she said. “Of these health outcomes, young people are most likely to experience injuries, and as a result, we find that there are significant health risks associated with consuming alcohol for young people. Among older individuals, the relative proportions of these outcomes vary by geography, and so do the risks associated with consuming alcohol,” she explained.
“Importantly, our analysis was conducted at the population level; when evaluating risk at the individual level, it is also important to consider other factors such as the presence of comorbidities and interactions between alcohol and medications,” she emphasized.
Health and alcohol interaction is complicated
“These findings seemingly contradict a previous [Global Burden of Diseases, Injuries, and Risk Factors Study] estimate published in The Lancet, which emphasized that any alcohol use, regardless of amount, leads to health loss across populations,” wrote Robyn Burton, PhD, and Nick Sheron, MD, both of King’s College, London, in an accompanying comment.
However, the novel methods of weighting relative risk curves according to levels of underlying disease drive the difference in results, along with disaggregated estimates by age, sex, and region, they said.
“Across most geographical regions in this latest analysis, injuries accounted for most alcohol-related harm in younger age groups. This led to a minimum risk level of zero, or very close to zero, among individuals aged 15-39 years across all geographical regions,” which is lower than the level for older adults because of the shift in alcohol-related disease burden towards cardiovascular disease and cancers, they said. “This highlights the need to consider existing rates of disease in a population when trying to determine the total harm posed by alcohol,” the commentators wrote.
In an additional commentary, Tony Rao, MD, a visiting clinical research fellow in psychiatry at King’s College, London, noted that “the elephant in the room with this study is the interpretation of risk based on outcomes for cardiovascular disease – particularly in older people. We know that any purported health benefits from alcohol on the heart and circulation are balanced out by the increased risk from other conditions such as cancer, liver disease, and mental disorders such as depression and dementia,” Dr. Rao said. “If we are to simply draw the conclusion that older people should continue or start drinking small amounts because it protects against diseases affecting heart and circulation – which still remains controversial – other lifestyle changes or the use of drugs targeted at individual cardiovascular disorders seem like a less harmful way of improving health and wellbeing.”
Data can guide clinical practice
No previous study has examined the effect of the theoretical minimum risk of alcohol consumption by geography, age, sex, and time in the context of background disease rates, said Noel Deep, MD, in an interview.
“This study enabled the researchers to quantify the proportion of the population that consumed alcohol in amounts that exceeded the thresholds by location, age, sex, and year, and this can serve as a guide in our efforts to target the control of alcohol intake by individuals,” said Dr. Deep, a general internist in private practice in Antigo, Wisc. He also serves as chief medical officer and a staff physician at Aspirus Langlade Hospital in Antigo.
The first take-home message for clinicians is that even low levels of alcohol consumption can have deleterious effects on the health of patients, and patients should be advised accordingly based on the prevalence of diseases in that community and geographic area, Dr. Deep said. “Secondly, clinicians should also consider the risk of alcohol consumption on all forms of health impacts in a given population rather than just focusing on alcohol-related health conditions,” he added.
“This study provides us with the data to tailor our efforts in educating the clinicians and the public about the relationship between alcohol consumption and disease outcomes based on the observed disease rates in each population,” Dr. Deep explained. “The data should provide another reason for physicians to advise their younger patients, especially the younger males, to avoid or minimize alcohol use,” he said. The data also can help clinicians formulate public health messaging and community education to reduce harmful alcohol use, he added.
As for additional research, Dr. Deep said he would like to see data on the difference in the health-related effects of alcohol in binge-drinkers vs. those who regularly consume alcohol on a daily basis. “It would probably also be helpful to figure out what type of alcohol is being studied and the quality of the alcohol,” he said.
The study was supported by the Bill and Melinda Gates Foundation. Ms. Bryazka and colleagues had no financial conflicts to disclose. Dr. Burton disclosed serving as a consultant to the World Health Organization European Office for the Prevention and Control of Noncommunicable Diseases. Dr. Sheron had no financial conflicts to disclose. Dr. Deep had no financial conflicts to disclose, but serves on the Editorial Advisory Board of Internal Medicine News.
The study was supported by the Bill and Melinda Gates Foundation.
The health risks and benefits of moderate alcohol consumption are complex and remain a hot topic of debate. The data suggest that small amounts of alcohol may reduce the risk of certain health outcomes over time, but increase the risk of others, wrote Dana Bryazka, MS, a researcher at the Institute for Health Metrics and Evaluation (IHME) at the University of Washington, Seattle, and colleagues, in a paper published in the Lancet.
“The amount of alcohol that minimizes health loss is likely to depend on the distribution of underlying causes of disease burden in a given population. Since this distribution varies widely by geography, age, sex, and time, the level of alcohol consumption associated with the lowest risk to health would depend on the age structure and disease composition of that population,” the researchers wrote.
“We estimate that 1.78 million people worldwide died due to alcohol use in 2020,” Ms. Bryazka said in an interview. “It is important that alcohol consumption guidelines and policies are updated to minimize this harm, particularly in the populations at greatest risk,” she said.
“Existing alcohol consumption guidelines frequently vary by sex, with higher consumption thresholds set for males compared to females. Interestingly, with the currently available data we do not see evidence that risk of alcohol use varies by sex,” she noted.
Methods and results
In the study, the researchers conducted a systematic analysis of burden-weighted dose-response relative risk curves across 22 health outcomes. They used disease rates from the Global Burden of Diseases, Injuries, and Risk Factors Study (GBD) 2020 for the years 1990-2020 for 21 regions, including 204 countries and territories. The data were analyzed by 5-year age group, sex, and year for individuals aged 15-95 years and older. The researchers estimated the theoretical minimum risk exposure level (TMREL) and nondrinker equivalent (NDE), meaning the amount of alcohol at which the health risk equals that of a nondrinker.
One standard drink was defined as 10 g of pure alcohol, equivalent to a small glass of red wine (100 mL or 3.4 fluid ounces) at 13% alcohol by volume, a can or bottle of beer (375 mL or 12 fluid ounces) at 3.5% alcohol by volume, or a shot of whiskey or other spirits (30 mL or 1.0 fluid ounces) at 40% alcohol by volume.
Overall, the TMREL was low regardless of age, sex, time, or geography, and varied from 0 to 1.87 standard drinks per day. However, it was lowest for males aged 15-39 years (0.136 drinks per day) and only slightly higher for females aged 15-39 (0.273), representing 1-2 tenths of a standard drink.
For adults aged 40 and older without any underlying health conditions, drinking a small amount of alcohol may provide some benefits, such as reducing the risk of ischemic heart disease, stroke, and diabetes, the researchers noted. In general, for individuals aged 40-64 years, TMRELs ranged from about half a standard drink per day (0.527 drinks for males and 0.562 standard drinks per day for females) to almost two standard drinks (1.69 standard drinks per day for males and 1.82 for females). For those older than 65 years, the TMRELs represented just over 3 standard drinks per day (3.19 for males and 3.51 for females). For individuals aged 40 years and older, the distribution of disease burden varied by region, but was J-shaped across all regions, the researchers noted.
The researchers also found that those individuals consuming harmful amounts of alcohol were most likely to be aged 15-39 (59.1%) and male (76.9%).
The study findings were limited by several factors including the observational design and lack of data on drinking patterns, such as binge drinking, the researchers noted. Other limitations include the lack of data reflecting patterns of alcohol consumption during the COVID-19 pandemic, and exclusion of outcomes often associated with alcohol use, such as depression, anxiety, and dementia, that might reduce estimates of TMREL and NDE.
However, the results add to the ongoing discussion of the relationship between moderate alcohol consumption and health, the researchers said.
“The findings of this study support the development of tailored guidelines and recommendations on alcohol consumption by age and across regions and highlight that existing low consumption thresholds are too high for younger populations in all regions,” they concluded.
Consider individual factors when counseling patients
The takeaway message for primary care is that alcohol consumed in moderation can reduce the risk of ischemic heart disease, stroke, and diabetes, Ms. Bryazka noted. “However, it also increases the risk of many cancers, intentional and unintentional injuries, and infectious diseases like tuberculosis,” she said. “Of these health outcomes, young people are most likely to experience injuries, and as a result, we find that there are significant health risks associated with consuming alcohol for young people. Among older individuals, the relative proportions of these outcomes vary by geography, and so do the risks associated with consuming alcohol,” she explained.
“Importantly, our analysis was conducted at the population level; when evaluating risk at the individual level, it is also important to consider other factors such as the presence of comorbidities and interactions between alcohol and medications,” she emphasized.
Health and alcohol interaction is complicated
“These findings seemingly contradict a previous [Global Burden of Diseases, Injuries, and Risk Factors Study] estimate published in The Lancet, which emphasized that any alcohol use, regardless of amount, leads to health loss across populations,” wrote Robyn Burton, PhD, and Nick Sheron, MD, both of King’s College, London, in an accompanying comment.
However, the novel methods of weighting relative risk curves according to levels of underlying disease drive the difference in results, along with disaggregated estimates by age, sex, and region, they said.
“Across most geographical regions in this latest analysis, injuries accounted for most alcohol-related harm in younger age groups. This led to a minimum risk level of zero, or very close to zero, among individuals aged 15-39 years across all geographical regions,” which is lower than the level for older adults because of the shift in alcohol-related disease burden towards cardiovascular disease and cancers, they said. “This highlights the need to consider existing rates of disease in a population when trying to determine the total harm posed by alcohol,” the commentators wrote.
In an additional commentary, Tony Rao, MD, a visiting clinical research fellow in psychiatry at King’s College, London, noted that “the elephant in the room with this study is the interpretation of risk based on outcomes for cardiovascular disease – particularly in older people. We know that any purported health benefits from alcohol on the heart and circulation are balanced out by the increased risk from other conditions such as cancer, liver disease, and mental disorders such as depression and dementia,” Dr. Rao said. “If we are to simply draw the conclusion that older people should continue or start drinking small amounts because it protects against diseases affecting heart and circulation – which still remains controversial – other lifestyle changes or the use of drugs targeted at individual cardiovascular disorders seem like a less harmful way of improving health and wellbeing.”
Data can guide clinical practice
No previous study has examined the effect of the theoretical minimum risk of alcohol consumption by geography, age, sex, and time in the context of background disease rates, said Noel Deep, MD, in an interview.
“This study enabled the researchers to quantify the proportion of the population that consumed alcohol in amounts that exceeded the thresholds by location, age, sex, and year, and this can serve as a guide in our efforts to target the control of alcohol intake by individuals,” said Dr. Deep, a general internist in private practice in Antigo, Wisc. He also serves as chief medical officer and a staff physician at Aspirus Langlade Hospital in Antigo.
The first take-home message for clinicians is that even low levels of alcohol consumption can have deleterious effects on the health of patients, and patients should be advised accordingly based on the prevalence of diseases in that community and geographic area, Dr. Deep said. “Secondly, clinicians should also consider the risk of alcohol consumption on all forms of health impacts in a given population rather than just focusing on alcohol-related health conditions,” he added.
“This study provides us with the data to tailor our efforts in educating the clinicians and the public about the relationship between alcohol consumption and disease outcomes based on the observed disease rates in each population,” Dr. Deep explained. “The data should provide another reason for physicians to advise their younger patients, especially the younger males, to avoid or minimize alcohol use,” he said. The data also can help clinicians formulate public health messaging and community education to reduce harmful alcohol use, he added.
As for additional research, Dr. Deep said he would like to see data on the difference in the health-related effects of alcohol in binge-drinkers vs. those who regularly consume alcohol on a daily basis. “It would probably also be helpful to figure out what type of alcohol is being studied and the quality of the alcohol,” he said.
The study was supported by the Bill and Melinda Gates Foundation. Ms. Bryazka and colleagues had no financial conflicts to disclose. Dr. Burton disclosed serving as a consultant to the World Health Organization European Office for the Prevention and Control of Noncommunicable Diseases. Dr. Sheron had no financial conflicts to disclose. Dr. Deep had no financial conflicts to disclose, but serves on the Editorial Advisory Board of Internal Medicine News.
The study was supported by the Bill and Melinda Gates Foundation.
The health risks and benefits of moderate alcohol consumption are complex and remain a hot topic of debate. The data suggest that small amounts of alcohol may reduce the risk of certain health outcomes over time, but increase the risk of others, wrote Dana Bryazka, MS, a researcher at the Institute for Health Metrics and Evaluation (IHME) at the University of Washington, Seattle, and colleagues, in a paper published in the Lancet.
“The amount of alcohol that minimizes health loss is likely to depend on the distribution of underlying causes of disease burden in a given population. Since this distribution varies widely by geography, age, sex, and time, the level of alcohol consumption associated with the lowest risk to health would depend on the age structure and disease composition of that population,” the researchers wrote.
“We estimate that 1.78 million people worldwide died due to alcohol use in 2020,” Ms. Bryazka said in an interview. “It is important that alcohol consumption guidelines and policies are updated to minimize this harm, particularly in the populations at greatest risk,” she said.
“Existing alcohol consumption guidelines frequently vary by sex, with higher consumption thresholds set for males compared to females. Interestingly, with the currently available data we do not see evidence that risk of alcohol use varies by sex,” she noted.
Methods and results
In the study, the researchers conducted a systematic analysis of burden-weighted dose-response relative risk curves across 22 health outcomes. They used disease rates from the Global Burden of Diseases, Injuries, and Risk Factors Study (GBD) 2020 for the years 1990-2020 for 21 regions, including 204 countries and territories. The data were analyzed by 5-year age group, sex, and year for individuals aged 15-95 years and older. The researchers estimated the theoretical minimum risk exposure level (TMREL) and nondrinker equivalent (NDE), meaning the amount of alcohol at which the health risk equals that of a nondrinker.
One standard drink was defined as 10 g of pure alcohol, equivalent to a small glass of red wine (100 mL or 3.4 fluid ounces) at 13% alcohol by volume, a can or bottle of beer (375 mL or 12 fluid ounces) at 3.5% alcohol by volume, or a shot of whiskey or other spirits (30 mL or 1.0 fluid ounces) at 40% alcohol by volume.
Overall, the TMREL was low regardless of age, sex, time, or geography, and varied from 0 to 1.87 standard drinks per day. However, it was lowest for males aged 15-39 years (0.136 drinks per day) and only slightly higher for females aged 15-39 (0.273), representing 1-2 tenths of a standard drink.
For adults aged 40 and older without any underlying health conditions, drinking a small amount of alcohol may provide some benefits, such as reducing the risk of ischemic heart disease, stroke, and diabetes, the researchers noted. In general, for individuals aged 40-64 years, TMRELs ranged from about half a standard drink per day (0.527 drinks for males and 0.562 standard drinks per day for females) to almost two standard drinks (1.69 standard drinks per day for males and 1.82 for females). For those older than 65 years, the TMRELs represented just over 3 standard drinks per day (3.19 for males and 3.51 for females). For individuals aged 40 years and older, the distribution of disease burden varied by region, but was J-shaped across all regions, the researchers noted.
The researchers also found that those individuals consuming harmful amounts of alcohol were most likely to be aged 15-39 (59.1%) and male (76.9%).
The study findings were limited by several factors including the observational design and lack of data on drinking patterns, such as binge drinking, the researchers noted. Other limitations include the lack of data reflecting patterns of alcohol consumption during the COVID-19 pandemic, and exclusion of outcomes often associated with alcohol use, such as depression, anxiety, and dementia, that might reduce estimates of TMREL and NDE.
However, the results add to the ongoing discussion of the relationship between moderate alcohol consumption and health, the researchers said.
“The findings of this study support the development of tailored guidelines and recommendations on alcohol consumption by age and across regions and highlight that existing low consumption thresholds are too high for younger populations in all regions,” they concluded.
Consider individual factors when counseling patients
The takeaway message for primary care is that alcohol consumed in moderation can reduce the risk of ischemic heart disease, stroke, and diabetes, Ms. Bryazka noted. “However, it also increases the risk of many cancers, intentional and unintentional injuries, and infectious diseases like tuberculosis,” she said. “Of these health outcomes, young people are most likely to experience injuries, and as a result, we find that there are significant health risks associated with consuming alcohol for young people. Among older individuals, the relative proportions of these outcomes vary by geography, and so do the risks associated with consuming alcohol,” she explained.
“Importantly, our analysis was conducted at the population level; when evaluating risk at the individual level, it is also important to consider other factors such as the presence of comorbidities and interactions between alcohol and medications,” she emphasized.
Health and alcohol interaction is complicated
“These findings seemingly contradict a previous [Global Burden of Diseases, Injuries, and Risk Factors Study] estimate published in The Lancet, which emphasized that any alcohol use, regardless of amount, leads to health loss across populations,” wrote Robyn Burton, PhD, and Nick Sheron, MD, both of King’s College, London, in an accompanying comment.
However, the novel methods of weighting relative risk curves according to levels of underlying disease drive the difference in results, along with disaggregated estimates by age, sex, and region, they said.
“Across most geographical regions in this latest analysis, injuries accounted for most alcohol-related harm in younger age groups. This led to a minimum risk level of zero, or very close to zero, among individuals aged 15-39 years across all geographical regions,” which is lower than the level for older adults because of the shift in alcohol-related disease burden towards cardiovascular disease and cancers, they said. “This highlights the need to consider existing rates of disease in a population when trying to determine the total harm posed by alcohol,” the commentators wrote.
In an additional commentary, Tony Rao, MD, a visiting clinical research fellow in psychiatry at King’s College, London, noted that “the elephant in the room with this study is the interpretation of risk based on outcomes for cardiovascular disease – particularly in older people. We know that any purported health benefits from alcohol on the heart and circulation are balanced out by the increased risk from other conditions such as cancer, liver disease, and mental disorders such as depression and dementia,” Dr. Rao said. “If we are to simply draw the conclusion that older people should continue or start drinking small amounts because it protects against diseases affecting heart and circulation – which still remains controversial – other lifestyle changes or the use of drugs targeted at individual cardiovascular disorders seem like a less harmful way of improving health and wellbeing.”
Data can guide clinical practice
No previous study has examined the effect of the theoretical minimum risk of alcohol consumption by geography, age, sex, and time in the context of background disease rates, said Noel Deep, MD, in an interview.
“This study enabled the researchers to quantify the proportion of the population that consumed alcohol in amounts that exceeded the thresholds by location, age, sex, and year, and this can serve as a guide in our efforts to target the control of alcohol intake by individuals,” said Dr. Deep, a general internist in private practice in Antigo, Wisc. He also serves as chief medical officer and a staff physician at Aspirus Langlade Hospital in Antigo.
The first take-home message for clinicians is that even low levels of alcohol consumption can have deleterious effects on the health of patients, and patients should be advised accordingly based on the prevalence of diseases in that community and geographic area, Dr. Deep said. “Secondly, clinicians should also consider the risk of alcohol consumption on all forms of health impacts in a given population rather than just focusing on alcohol-related health conditions,” he added.
“This study provides us with the data to tailor our efforts in educating the clinicians and the public about the relationship between alcohol consumption and disease outcomes based on the observed disease rates in each population,” Dr. Deep explained. “The data should provide another reason for physicians to advise their younger patients, especially the younger males, to avoid or minimize alcohol use,” he said. The data also can help clinicians formulate public health messaging and community education to reduce harmful alcohol use, he added.
As for additional research, Dr. Deep said he would like to see data on the difference in the health-related effects of alcohol in binge-drinkers vs. those who regularly consume alcohol on a daily basis. “It would probably also be helpful to figure out what type of alcohol is being studied and the quality of the alcohol,” he said.
The study was supported by the Bill and Melinda Gates Foundation. Ms. Bryazka and colleagues had no financial conflicts to disclose. Dr. Burton disclosed serving as a consultant to the World Health Organization European Office for the Prevention and Control of Noncommunicable Diseases. Dr. Sheron had no financial conflicts to disclose. Dr. Deep had no financial conflicts to disclose, but serves on the Editorial Advisory Board of Internal Medicine News.
The study was supported by the Bill and Melinda Gates Foundation.
FROM THE LANCET
Inflammatory profiles impact major depressive disorder
Early onset of disease in patients with major depressive disorder may be linked to a specific inflammatory profile, based on data from 234 individuals.
Major depressive disorder (MDD) remains common, and evidence suggests that it is increasing among younger individuals, but data on early-onset MDD in adults are limited, Ana Paula Anzolin, a graduate student at the Universidade Federal do Rio Grande do Sul, Porto Alegre, Brazil, and colleagues wrote.
Although previous studies have shown abnormal cytokine production in patients with MDD, the impact of inflammation on MDD and disease onset and progression remains unclear, they said.
In a study published in Psychiatry Research, the authors identified outpatients aged 18-85 years with confirmed MDD and scores of at least 8 on the HAM-D scale who were undergoing treatment at a single center. Early onset was defined as a diagnosis of MDD before age 30 years (99 patients) and late onset was defined as a diagnosis at age 30 years and older (135 patients). The researchers measured levels of interleukin-6, IL-1 beta, IL-10, and tumor necrosis factor alpha (TNF-alpha).
Overall, the level of cytokine profiles in early- versus late-onset disease was significantly higher for IL-1B and TNF-alpha (P < .001 for both). The significant difference between early- and late-onset disease remained regardless of comorbidity with autoimmune diseases, the researchers noted.
IL-6 levels were higher in the early-onset group and IL-10 levels were higher in the late-onset group, but these differences were not significant.
the researchers wrote.
The results also support findings from previous studies that suggest a divergence between early- and late adult–onset depression, they said. More research on early-onset MDD in adults is needed, as these patients tend to have more severe symptoms, more medical and psychiatric comorbidities, and an increased risk of depressive episodes and suicide attempts.
The study findings were limited by several factors including the lack of a control group, the retrospective assessment of disease onset, and the limited cytokines studied, which do not reflect changes in the entire immune network response, the researchers noted.
However, the study is the first known to examine the association of serum cytokines and early- and late-onset MDD in adults, and the results support the use of IL-1B and TNF-alpha as potential treatment targets in the development of new therapies for MDD, they concluded.
The study was supported by the Fundo de Incentivo à Pesquisa – Hospital de Clínicas de Porto Alegre, the Conselho Nacional de Desenvolvimento Científico e Tecnológico, and the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior. The researchers had no financial conflicts to disclose.
Early onset of disease in patients with major depressive disorder may be linked to a specific inflammatory profile, based on data from 234 individuals.
Major depressive disorder (MDD) remains common, and evidence suggests that it is increasing among younger individuals, but data on early-onset MDD in adults are limited, Ana Paula Anzolin, a graduate student at the Universidade Federal do Rio Grande do Sul, Porto Alegre, Brazil, and colleagues wrote.
Although previous studies have shown abnormal cytokine production in patients with MDD, the impact of inflammation on MDD and disease onset and progression remains unclear, they said.
In a study published in Psychiatry Research, the authors identified outpatients aged 18-85 years with confirmed MDD and scores of at least 8 on the HAM-D scale who were undergoing treatment at a single center. Early onset was defined as a diagnosis of MDD before age 30 years (99 patients) and late onset was defined as a diagnosis at age 30 years and older (135 patients). The researchers measured levels of interleukin-6, IL-1 beta, IL-10, and tumor necrosis factor alpha (TNF-alpha).
Overall, the level of cytokine profiles in early- versus late-onset disease was significantly higher for IL-1B and TNF-alpha (P < .001 for both). The significant difference between early- and late-onset disease remained regardless of comorbidity with autoimmune diseases, the researchers noted.
IL-6 levels were higher in the early-onset group and IL-10 levels were higher in the late-onset group, but these differences were not significant.
the researchers wrote.
The results also support findings from previous studies that suggest a divergence between early- and late adult–onset depression, they said. More research on early-onset MDD in adults is needed, as these patients tend to have more severe symptoms, more medical and psychiatric comorbidities, and an increased risk of depressive episodes and suicide attempts.
The study findings were limited by several factors including the lack of a control group, the retrospective assessment of disease onset, and the limited cytokines studied, which do not reflect changes in the entire immune network response, the researchers noted.
However, the study is the first known to examine the association of serum cytokines and early- and late-onset MDD in adults, and the results support the use of IL-1B and TNF-alpha as potential treatment targets in the development of new therapies for MDD, they concluded.
The study was supported by the Fundo de Incentivo à Pesquisa – Hospital de Clínicas de Porto Alegre, the Conselho Nacional de Desenvolvimento Científico e Tecnológico, and the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior. The researchers had no financial conflicts to disclose.
Early onset of disease in patients with major depressive disorder may be linked to a specific inflammatory profile, based on data from 234 individuals.
Major depressive disorder (MDD) remains common, and evidence suggests that it is increasing among younger individuals, but data on early-onset MDD in adults are limited, Ana Paula Anzolin, a graduate student at the Universidade Federal do Rio Grande do Sul, Porto Alegre, Brazil, and colleagues wrote.
Although previous studies have shown abnormal cytokine production in patients with MDD, the impact of inflammation on MDD and disease onset and progression remains unclear, they said.
In a study published in Psychiatry Research, the authors identified outpatients aged 18-85 years with confirmed MDD and scores of at least 8 on the HAM-D scale who were undergoing treatment at a single center. Early onset was defined as a diagnosis of MDD before age 30 years (99 patients) and late onset was defined as a diagnosis at age 30 years and older (135 patients). The researchers measured levels of interleukin-6, IL-1 beta, IL-10, and tumor necrosis factor alpha (TNF-alpha).
Overall, the level of cytokine profiles in early- versus late-onset disease was significantly higher for IL-1B and TNF-alpha (P < .001 for both). The significant difference between early- and late-onset disease remained regardless of comorbidity with autoimmune diseases, the researchers noted.
IL-6 levels were higher in the early-onset group and IL-10 levels were higher in the late-onset group, but these differences were not significant.
the researchers wrote.
The results also support findings from previous studies that suggest a divergence between early- and late adult–onset depression, they said. More research on early-onset MDD in adults is needed, as these patients tend to have more severe symptoms, more medical and psychiatric comorbidities, and an increased risk of depressive episodes and suicide attempts.
The study findings were limited by several factors including the lack of a control group, the retrospective assessment of disease onset, and the limited cytokines studied, which do not reflect changes in the entire immune network response, the researchers noted.
However, the study is the first known to examine the association of serum cytokines and early- and late-onset MDD in adults, and the results support the use of IL-1B and TNF-alpha as potential treatment targets in the development of new therapies for MDD, they concluded.
The study was supported by the Fundo de Incentivo à Pesquisa – Hospital de Clínicas de Porto Alegre, the Conselho Nacional de Desenvolvimento Científico e Tecnológico, and the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior. The researchers had no financial conflicts to disclose.
FROM PSYCHIATRY RESEARCH
Uveitis in juvenile arthritis patients persists into midlife
Active uveitis remained in 43.4% of juvenile idiopathic arthritis (JIA) patients up to 40 years after a diagnosis, based on data from 30 individuals.
Uveitis occurs in approximately 10%-20% of patients with JIA, but data on the long-term activity and prevalence are limited, although previous studies suggest that uveitis can persist into adulthood, wrote Dr. Angelika Skarin of Skåne University in Lund, Sweden, and colleagues.
In a study published in Pediatric Rheumatology, the researchers reviewed ophthalmic records from 30 JIA patients at a mean of 40.7 years after uveitis onset. They compared these records to data collected from the same patient population at a mean of 7.2 and 24.0 years after onset. In the previous follow-up studies, 49% of the patients had active uveitis at 24 years, and the prevalence of cataracts and glaucoma increased between the 7-year and 24-year assessments.
In the current study, 43.4% of the population had active uveitis at the 40-year follow-up, which corresponded to 23.6% of the original study cohort. The mean age of the participants overall was 46.9 years, the mean duration of joint disease was 42.99 years, and the mean time from onset of uveitis was 40.7 years.
In addition, 66.6% of the patients in the current study had cataracts or had undergone cataract surgery in one or both eyes, and 40.0% had glaucoma.
By the time of the current study, of the original cohort of 55 individuals, 11 were deceased; rheumatic disease was declared the main cause in four patients and a contributing factor in three others.
Potential drivers of the earliest cases of glaucoma and ocular hypertension (G/OH) include increased intraocular pressure as a result of topical corticosteroid treatment, the researchers noted in their discussion. However, G/OH occurring later than the 7-year follow-up was “more likely to be the type observed in many patients with long-standing chronic uveitis, where a gradual increase in intraocular pressure is assumed to be caused by impaired aqueous outflow,” they said.
Only 4 of the 30 patients did not have regular ophthalmology visits, which suggests a study population with ocular symptoms or concerns about their eyesight, the researchers wrote. “The fact that 13% of our original cohort were reported to have severe visual impairment or worse in both eyes at any of the three follow-ups is noteworthy,” compared to reports of visual impairment of less than 0.5% in a German study in the general population for similar ages.
The findings were limited by several factors, including the retrospective design, small study population, and lack of data on 25 of the original 55-member study cohort, which may reduce the reliability of the current study, the researchers noted. However, the results reflect data from previous studies and support the need for JIA patients to continue regular ophthalmic checkups throughout life, they concluded.
The study was supported by Stiftelsen för Synskadade i f.d. Malmöhus län, Sweden, Skånes Universitetssjukhus Stiftelser och Donationer, Ögonfonden, and the Swedish Society of Medicine. The researchers had no financial conflicts to disclose.
Active uveitis remained in 43.4% of juvenile idiopathic arthritis (JIA) patients up to 40 years after a diagnosis, based on data from 30 individuals.
Uveitis occurs in approximately 10%-20% of patients with JIA, but data on the long-term activity and prevalence are limited, although previous studies suggest that uveitis can persist into adulthood, wrote Dr. Angelika Skarin of Skåne University in Lund, Sweden, and colleagues.
In a study published in Pediatric Rheumatology, the researchers reviewed ophthalmic records from 30 JIA patients at a mean of 40.7 years after uveitis onset. They compared these records to data collected from the same patient population at a mean of 7.2 and 24.0 years after onset. In the previous follow-up studies, 49% of the patients had active uveitis at 24 years, and the prevalence of cataracts and glaucoma increased between the 7-year and 24-year assessments.
In the current study, 43.4% of the population had active uveitis at the 40-year follow-up, which corresponded to 23.6% of the original study cohort. The mean age of the participants overall was 46.9 years, the mean duration of joint disease was 42.99 years, and the mean time from onset of uveitis was 40.7 years.
In addition, 66.6% of the patients in the current study had cataracts or had undergone cataract surgery in one or both eyes, and 40.0% had glaucoma.
By the time of the current study, of the original cohort of 55 individuals, 11 were deceased; rheumatic disease was declared the main cause in four patients and a contributing factor in three others.
Potential drivers of the earliest cases of glaucoma and ocular hypertension (G/OH) include increased intraocular pressure as a result of topical corticosteroid treatment, the researchers noted in their discussion. However, G/OH occurring later than the 7-year follow-up was “more likely to be the type observed in many patients with long-standing chronic uveitis, where a gradual increase in intraocular pressure is assumed to be caused by impaired aqueous outflow,” they said.
Only 4 of the 30 patients did not have regular ophthalmology visits, which suggests a study population with ocular symptoms or concerns about their eyesight, the researchers wrote. “The fact that 13% of our original cohort were reported to have severe visual impairment or worse in both eyes at any of the three follow-ups is noteworthy,” compared to reports of visual impairment of less than 0.5% in a German study in the general population for similar ages.
The findings were limited by several factors, including the retrospective design, small study population, and lack of data on 25 of the original 55-member study cohort, which may reduce the reliability of the current study, the researchers noted. However, the results reflect data from previous studies and support the need for JIA patients to continue regular ophthalmic checkups throughout life, they concluded.
The study was supported by Stiftelsen för Synskadade i f.d. Malmöhus län, Sweden, Skånes Universitetssjukhus Stiftelser och Donationer, Ögonfonden, and the Swedish Society of Medicine. The researchers had no financial conflicts to disclose.
Active uveitis remained in 43.4% of juvenile idiopathic arthritis (JIA) patients up to 40 years after a diagnosis, based on data from 30 individuals.
Uveitis occurs in approximately 10%-20% of patients with JIA, but data on the long-term activity and prevalence are limited, although previous studies suggest that uveitis can persist into adulthood, wrote Dr. Angelika Skarin of Skåne University in Lund, Sweden, and colleagues.
In a study published in Pediatric Rheumatology, the researchers reviewed ophthalmic records from 30 JIA patients at a mean of 40.7 years after uveitis onset. They compared these records to data collected from the same patient population at a mean of 7.2 and 24.0 years after onset. In the previous follow-up studies, 49% of the patients had active uveitis at 24 years, and the prevalence of cataracts and glaucoma increased between the 7-year and 24-year assessments.
In the current study, 43.4% of the population had active uveitis at the 40-year follow-up, which corresponded to 23.6% of the original study cohort. The mean age of the participants overall was 46.9 years, the mean duration of joint disease was 42.99 years, and the mean time from onset of uveitis was 40.7 years.
In addition, 66.6% of the patients in the current study had cataracts or had undergone cataract surgery in one or both eyes, and 40.0% had glaucoma.
By the time of the current study, of the original cohort of 55 individuals, 11 were deceased; rheumatic disease was declared the main cause in four patients and a contributing factor in three others.
Potential drivers of the earliest cases of glaucoma and ocular hypertension (G/OH) include increased intraocular pressure as a result of topical corticosteroid treatment, the researchers noted in their discussion. However, G/OH occurring later than the 7-year follow-up was “more likely to be the type observed in many patients with long-standing chronic uveitis, where a gradual increase in intraocular pressure is assumed to be caused by impaired aqueous outflow,” they said.
Only 4 of the 30 patients did not have regular ophthalmology visits, which suggests a study population with ocular symptoms or concerns about their eyesight, the researchers wrote. “The fact that 13% of our original cohort were reported to have severe visual impairment or worse in both eyes at any of the three follow-ups is noteworthy,” compared to reports of visual impairment of less than 0.5% in a German study in the general population for similar ages.
The findings were limited by several factors, including the retrospective design, small study population, and lack of data on 25 of the original 55-member study cohort, which may reduce the reliability of the current study, the researchers noted. However, the results reflect data from previous studies and support the need for JIA patients to continue regular ophthalmic checkups throughout life, they concluded.
The study was supported by Stiftelsen för Synskadade i f.d. Malmöhus län, Sweden, Skånes Universitetssjukhus Stiftelser och Donationer, Ögonfonden, and the Swedish Society of Medicine. The researchers had no financial conflicts to disclose.
FROM PEDIATRIC RHEUMATOLOGY
Medical management of miscarriage curbs costs and maintains quality of care
Medical management of early pregnancy loss costs less and offers similar quality of life to uterine aspiration, based on data from an analytical model.
Early pregnancy loss (EPL) occurs in more than 1 million women in the United States each year, and many patients are diagnosed before they show symptoms, wrote Divyah Nagendra, MD, of Cambridge Health Alliance, Mass., and colleagues.
A 2018 study showed that medical management of EPL with mifepristone added to misoprostol increased effectiveness and reduced the need for additional medication or subsequent uterine procedures, but the cost of mifepristone is perceived as a barrier, and the cost-effectiveness of its use, compared with surgical or expectant management, has not been well studied, the researchers noted.
“We already know that adding mifepristone to the medical management of early pregnancy loss increases the effectiveness of the regimen,” coauthor Courtney A. Schreiber, MD, of the University of Pennsylvania, Philadelphia, said in an interview. “Procedural uterine aspiration is highly effective as well, so patients and providers may consider the cost when deciding on a treatment strategy,” she added.
“If medication management is preferred by many patients, decreases the need to access in-person clinical care during a pandemic, and is found to be cost-effective, clinicians and policymakers should increase efforts to improve mifepristone availability and reduce access burdens,” the researchers wrote.
In a study published in the American Journal of Obstetrics & Gynecology, the researchers created a decision-analytic model using data from published literature and the Pregnancy Failure Regiments Trial (PreFaiR) to compare office-based uterine aspiration to medical management with mifepristone pretreatment followed by misoprostol for EPL.
The PrFaiR study randomized 300 women who experienced EPL before 12 weeks’ gestation to medication management with 800 mcg misoprostol vaginally, with or without pretreatment of 200 mg mifeprestone orally. The average age of the participants was 30.7 years, and demographics were similar between the groups.
The researchers used the PrFaiR data for medical management and patient-level data from published literature for uterine aspiration.
The primary outcome was the cost per quality-adjusted life year (QALY) gained. QALY was based on a modified utility score from the published literature. Effectiveness was based on QALY gained and the rate of complete expulsion of the gestational sac without additional intervention.
Overall, the mean costs per person were significantly higher for uterine aspiration, compared with medical management ($828 vs. $661, P = .004). Uterine aspiration was significantly more effective for complete gestational sac expulsion (97.3% vs. 83.8%, P = .0001). However, the QALYs were significantly higher for medical management, compared with uterine aspiration (0.082 vs. 0.079, P < .0001).
Cost-effectiveness was greater for medical management from a health care sector perspective, with lower costs and higher QALYs than uterine aspiration, the researchers noted.
They also evaluated the effect of mifepristone pretreatment on cost-effectiveness and found that medical management would remain cost effective, compared with uterine aspiration even if uterine aspiration procedures decreased in cost and mifepristone increased in cost, and even if medication management had a decreased completion rate and utility score, compared with uterine aspiration.
“Our analysis demonstrates that the incremental cost-effectiveness ratio (ICER) for medical management is well below the maximum willingness-to-pay threshold of approximately $100,000 per QALY gained,” the researchers wrote in their discussion of the findings.
Potential savings, uncertain access
Despite the potential savings and patient benefits, access to mifepristone remains a barrier, the researchers said.
Although the FDA lifted some restrictions on mifepristone in 2021 in the wake of the ongoing COVID-19 pandemic, the effect of new abortion-related restrictions remains to be seen.
The study findings were limited by several factors, including the use of 2018 National Medicare reimbursement rates to calculate costs, though actual costs vary by region and payer, the researchers noted. Other limitations include variations in cost of mifepristone by region and time and the differences in data sources between the uterine aspiration and medical management groups. More research is needed to assess QALYs for early pregnancy loss to establish standard measures and to assess the societal perspective of ESL as well as the health care perspective, they added.
However, the current results support medical management of EPL with mifepristone pretreatment followed by misoprostol as a “high-value care alternative” to office-based uterine aspiration, they said. “Increasing access to mifepristone and eliminating unnecessary restrictions will improve early pregnancy care,” they concluded.
“Given how effective procedural management is, we were slightly surprised that medical management remains cost effective,” Dr. Schreiber said in an interview.
Looking ahead in the wake of new restrictions on use for abortion, “patients may have difficulty accessing either medical or procedural management for early pregnancy loss,” Dr. Schreiber noted. “We support the accessibility of all evidence-based care and hope that our data will help overcome perceived financial barriers,” she said. Additional research needs include improved implementation and access to evidence-based early pregnancy loss care, she added.
Reasons to lift regulations
“Given the recent overturning of Roe v. Wade, any medications that are associated with abortion have increased scrutiny, especially mifepristone and misoprostol, even though these medications are also used for managing early pregnancy loss,” Sarah W. Prager, MD, of the University of Washington, Seattle, said in an interview. “Demonstrating that medication management of EPL with mifepristone/misoprostol is less expensive and has increased QALYs associated with it is yet another reason to deregulate mifepristone so it can also be more accessible for management of EPL,” said Dr. Prager, who was not involved in the study.
Dr. Prager said she was not surprised by the findings, as effective medication should be less expensive than a procedure. “I would caution that the increased QALYs found in this study should not be interpreted as a reason to restrict surgical management of EPL but to increase access also to medication management, even though medication has a slightly lower rate of complete gestational sac expulsion,” she noted. “Mode of management should be up to the patient, unless there is a clear medical reason for one or the other.”
Going forward, “the FDA has it in its power to remove the REMS, which would immediately make mifepristone a medication that can be prescribed through a pharmacy and therefore much more available,” said Dr. Prager. “Restrictions for both medication and surgical management of EPL will likely increase in states where abortion is illegal, and it could possibly lead to patients having less choice as to mode of management,” she explained.
“There are many studies showing that all modes of EPL management are safe and effective and should be supported with respect to patient choice,” Dr. Prager noted. “There are also substantial data supporting the overall safety of mifepristone, and there are no scientific or medical data suggesting the REMS increases safety in any way. Frankly, there are no good, evidence-based reasons to continue to keep the REMS in place,” she said.
The study was supported by the National Institute of Child Health and Human Development of the National Institutes of Health and a Society of Family Planning Research Fund Midcareer Mentor Award. The researchers had no financial conflicts to disclose. Dr. Prager had no financial conflicts to disclose and serves on the Editorial Advisory Board of Ob.Gyn. News.
Medical management of early pregnancy loss costs less and offers similar quality of life to uterine aspiration, based on data from an analytical model.
Early pregnancy loss (EPL) occurs in more than 1 million women in the United States each year, and many patients are diagnosed before they show symptoms, wrote Divyah Nagendra, MD, of Cambridge Health Alliance, Mass., and colleagues.
A 2018 study showed that medical management of EPL with mifepristone added to misoprostol increased effectiveness and reduced the need for additional medication or subsequent uterine procedures, but the cost of mifepristone is perceived as a barrier, and the cost-effectiveness of its use, compared with surgical or expectant management, has not been well studied, the researchers noted.
“We already know that adding mifepristone to the medical management of early pregnancy loss increases the effectiveness of the regimen,” coauthor Courtney A. Schreiber, MD, of the University of Pennsylvania, Philadelphia, said in an interview. “Procedural uterine aspiration is highly effective as well, so patients and providers may consider the cost when deciding on a treatment strategy,” she added.
“If medication management is preferred by many patients, decreases the need to access in-person clinical care during a pandemic, and is found to be cost-effective, clinicians and policymakers should increase efforts to improve mifepristone availability and reduce access burdens,” the researchers wrote.
In a study published in the American Journal of Obstetrics & Gynecology, the researchers created a decision-analytic model using data from published literature and the Pregnancy Failure Regiments Trial (PreFaiR) to compare office-based uterine aspiration to medical management with mifepristone pretreatment followed by misoprostol for EPL.
The PrFaiR study randomized 300 women who experienced EPL before 12 weeks’ gestation to medication management with 800 mcg misoprostol vaginally, with or without pretreatment of 200 mg mifeprestone orally. The average age of the participants was 30.7 years, and demographics were similar between the groups.
The researchers used the PrFaiR data for medical management and patient-level data from published literature for uterine aspiration.
The primary outcome was the cost per quality-adjusted life year (QALY) gained. QALY was based on a modified utility score from the published literature. Effectiveness was based on QALY gained and the rate of complete expulsion of the gestational sac without additional intervention.
Overall, the mean costs per person were significantly higher for uterine aspiration, compared with medical management ($828 vs. $661, P = .004). Uterine aspiration was significantly more effective for complete gestational sac expulsion (97.3% vs. 83.8%, P = .0001). However, the QALYs were significantly higher for medical management, compared with uterine aspiration (0.082 vs. 0.079, P < .0001).
Cost-effectiveness was greater for medical management from a health care sector perspective, with lower costs and higher QALYs than uterine aspiration, the researchers noted.
They also evaluated the effect of mifepristone pretreatment on cost-effectiveness and found that medical management would remain cost effective, compared with uterine aspiration even if uterine aspiration procedures decreased in cost and mifepristone increased in cost, and even if medication management had a decreased completion rate and utility score, compared with uterine aspiration.
“Our analysis demonstrates that the incremental cost-effectiveness ratio (ICER) for medical management is well below the maximum willingness-to-pay threshold of approximately $100,000 per QALY gained,” the researchers wrote in their discussion of the findings.
Potential savings, uncertain access
Despite the potential savings and patient benefits, access to mifepristone remains a barrier, the researchers said.
Although the FDA lifted some restrictions on mifepristone in 2021 in the wake of the ongoing COVID-19 pandemic, the effect of new abortion-related restrictions remains to be seen.
The study findings were limited by several factors, including the use of 2018 National Medicare reimbursement rates to calculate costs, though actual costs vary by region and payer, the researchers noted. Other limitations include variations in cost of mifepristone by region and time and the differences in data sources between the uterine aspiration and medical management groups. More research is needed to assess QALYs for early pregnancy loss to establish standard measures and to assess the societal perspective of ESL as well as the health care perspective, they added.
However, the current results support medical management of EPL with mifepristone pretreatment followed by misoprostol as a “high-value care alternative” to office-based uterine aspiration, they said. “Increasing access to mifepristone and eliminating unnecessary restrictions will improve early pregnancy care,” they concluded.
“Given how effective procedural management is, we were slightly surprised that medical management remains cost effective,” Dr. Schreiber said in an interview.
Looking ahead in the wake of new restrictions on use for abortion, “patients may have difficulty accessing either medical or procedural management for early pregnancy loss,” Dr. Schreiber noted. “We support the accessibility of all evidence-based care and hope that our data will help overcome perceived financial barriers,” she said. Additional research needs include improved implementation and access to evidence-based early pregnancy loss care, she added.
Reasons to lift regulations
“Given the recent overturning of Roe v. Wade, any medications that are associated with abortion have increased scrutiny, especially mifepristone and misoprostol, even though these medications are also used for managing early pregnancy loss,” Sarah W. Prager, MD, of the University of Washington, Seattle, said in an interview. “Demonstrating that medication management of EPL with mifepristone/misoprostol is less expensive and has increased QALYs associated with it is yet another reason to deregulate mifepristone so it can also be more accessible for management of EPL,” said Dr. Prager, who was not involved in the study.
Dr. Prager said she was not surprised by the findings, as effective medication should be less expensive than a procedure. “I would caution that the increased QALYs found in this study should not be interpreted as a reason to restrict surgical management of EPL but to increase access also to medication management, even though medication has a slightly lower rate of complete gestational sac expulsion,” she noted. “Mode of management should be up to the patient, unless there is a clear medical reason for one or the other.”
Going forward, “the FDA has it in its power to remove the REMS, which would immediately make mifepristone a medication that can be prescribed through a pharmacy and therefore much more available,” said Dr. Prager. “Restrictions for both medication and surgical management of EPL will likely increase in states where abortion is illegal, and it could possibly lead to patients having less choice as to mode of management,” she explained.
“There are many studies showing that all modes of EPL management are safe and effective and should be supported with respect to patient choice,” Dr. Prager noted. “There are also substantial data supporting the overall safety of mifepristone, and there are no scientific or medical data suggesting the REMS increases safety in any way. Frankly, there are no good, evidence-based reasons to continue to keep the REMS in place,” she said.
The study was supported by the National Institute of Child Health and Human Development of the National Institutes of Health and a Society of Family Planning Research Fund Midcareer Mentor Award. The researchers had no financial conflicts to disclose. Dr. Prager had no financial conflicts to disclose and serves on the Editorial Advisory Board of Ob.Gyn. News.
Medical management of early pregnancy loss costs less and offers similar quality of life to uterine aspiration, based on data from an analytical model.
Early pregnancy loss (EPL) occurs in more than 1 million women in the United States each year, and many patients are diagnosed before they show symptoms, wrote Divyah Nagendra, MD, of Cambridge Health Alliance, Mass., and colleagues.
A 2018 study showed that medical management of EPL with mifepristone added to misoprostol increased effectiveness and reduced the need for additional medication or subsequent uterine procedures, but the cost of mifepristone is perceived as a barrier, and the cost-effectiveness of its use, compared with surgical or expectant management, has not been well studied, the researchers noted.
“We already know that adding mifepristone to the medical management of early pregnancy loss increases the effectiveness of the regimen,” coauthor Courtney A. Schreiber, MD, of the University of Pennsylvania, Philadelphia, said in an interview. “Procedural uterine aspiration is highly effective as well, so patients and providers may consider the cost when deciding on a treatment strategy,” she added.
“If medication management is preferred by many patients, decreases the need to access in-person clinical care during a pandemic, and is found to be cost-effective, clinicians and policymakers should increase efforts to improve mifepristone availability and reduce access burdens,” the researchers wrote.
In a study published in the American Journal of Obstetrics & Gynecology, the researchers created a decision-analytic model using data from published literature and the Pregnancy Failure Regiments Trial (PreFaiR) to compare office-based uterine aspiration to medical management with mifepristone pretreatment followed by misoprostol for EPL.
The PrFaiR study randomized 300 women who experienced EPL before 12 weeks’ gestation to medication management with 800 mcg misoprostol vaginally, with or without pretreatment of 200 mg mifeprestone orally. The average age of the participants was 30.7 years, and demographics were similar between the groups.
The researchers used the PrFaiR data for medical management and patient-level data from published literature for uterine aspiration.
The primary outcome was the cost per quality-adjusted life year (QALY) gained. QALY was based on a modified utility score from the published literature. Effectiveness was based on QALY gained and the rate of complete expulsion of the gestational sac without additional intervention.
Overall, the mean costs per person were significantly higher for uterine aspiration, compared with medical management ($828 vs. $661, P = .004). Uterine aspiration was significantly more effective for complete gestational sac expulsion (97.3% vs. 83.8%, P = .0001). However, the QALYs were significantly higher for medical management, compared with uterine aspiration (0.082 vs. 0.079, P < .0001).
Cost-effectiveness was greater for medical management from a health care sector perspective, with lower costs and higher QALYs than uterine aspiration, the researchers noted.
They also evaluated the effect of mifepristone pretreatment on cost-effectiveness and found that medical management would remain cost effective, compared with uterine aspiration even if uterine aspiration procedures decreased in cost and mifepristone increased in cost, and even if medication management had a decreased completion rate and utility score, compared with uterine aspiration.
“Our analysis demonstrates that the incremental cost-effectiveness ratio (ICER) for medical management is well below the maximum willingness-to-pay threshold of approximately $100,000 per QALY gained,” the researchers wrote in their discussion of the findings.
Potential savings, uncertain access
Despite the potential savings and patient benefits, access to mifepristone remains a barrier, the researchers said.
Although the FDA lifted some restrictions on mifepristone in 2021 in the wake of the ongoing COVID-19 pandemic, the effect of new abortion-related restrictions remains to be seen.
The study findings were limited by several factors, including the use of 2018 National Medicare reimbursement rates to calculate costs, though actual costs vary by region and payer, the researchers noted. Other limitations include variations in cost of mifepristone by region and time and the differences in data sources between the uterine aspiration and medical management groups. More research is needed to assess QALYs for early pregnancy loss to establish standard measures and to assess the societal perspective of ESL as well as the health care perspective, they added.
However, the current results support medical management of EPL with mifepristone pretreatment followed by misoprostol as a “high-value care alternative” to office-based uterine aspiration, they said. “Increasing access to mifepristone and eliminating unnecessary restrictions will improve early pregnancy care,” they concluded.
“Given how effective procedural management is, we were slightly surprised that medical management remains cost effective,” Dr. Schreiber said in an interview.
Looking ahead in the wake of new restrictions on use for abortion, “patients may have difficulty accessing either medical or procedural management for early pregnancy loss,” Dr. Schreiber noted. “We support the accessibility of all evidence-based care and hope that our data will help overcome perceived financial barriers,” she said. Additional research needs include improved implementation and access to evidence-based early pregnancy loss care, she added.
Reasons to lift regulations
“Given the recent overturning of Roe v. Wade, any medications that are associated with abortion have increased scrutiny, especially mifepristone and misoprostol, even though these medications are also used for managing early pregnancy loss,” Sarah W. Prager, MD, of the University of Washington, Seattle, said in an interview. “Demonstrating that medication management of EPL with mifepristone/misoprostol is less expensive and has increased QALYs associated with it is yet another reason to deregulate mifepristone so it can also be more accessible for management of EPL,” said Dr. Prager, who was not involved in the study.
Dr. Prager said she was not surprised by the findings, as effective medication should be less expensive than a procedure. “I would caution that the increased QALYs found in this study should not be interpreted as a reason to restrict surgical management of EPL but to increase access also to medication management, even though medication has a slightly lower rate of complete gestational sac expulsion,” she noted. “Mode of management should be up to the patient, unless there is a clear medical reason for one or the other.”
Going forward, “the FDA has it in its power to remove the REMS, which would immediately make mifepristone a medication that can be prescribed through a pharmacy and therefore much more available,” said Dr. Prager. “Restrictions for both medication and surgical management of EPL will likely increase in states where abortion is illegal, and it could possibly lead to patients having less choice as to mode of management,” she explained.
“There are many studies showing that all modes of EPL management are safe and effective and should be supported with respect to patient choice,” Dr. Prager noted. “There are also substantial data supporting the overall safety of mifepristone, and there are no scientific or medical data suggesting the REMS increases safety in any way. Frankly, there are no good, evidence-based reasons to continue to keep the REMS in place,” she said.
The study was supported by the National Institute of Child Health and Human Development of the National Institutes of Health and a Society of Family Planning Research Fund Midcareer Mentor Award. The researchers had no financial conflicts to disclose. Dr. Prager had no financial conflicts to disclose and serves on the Editorial Advisory Board of Ob.Gyn. News.
FROM THE AMERICAN JOURNAL OF OBSTETRICS & GYNECOLOGY
Biologics reduce exacerbations in severe asthma
, based on data from more than 2,000 individuals.
The development of biologics to target specific inflammatory pathways “has transformed the management of uncontrolled SA,” but data on the real-world use of biologics in severe asthma patients treated by subspecialists are limited, wrote Reynold A. Panettieri, Jr., MD, of Rutgers, State University of New Jersey, New Brunswick, and colleagues.
In a study published in the Annals of Allergy, Asthma & Immunology, the researchers reviewed data from CHRONICLE, an ongoing, prospective, real-world noninterventional study of adults aged 18 years and older with severe asthma in the United States.
The study population included 2,847 patients enrolled in the CHRONICLE study between February 2018 and February 2021; 68.8% were women, 74.6% were White. The patients ranged in age from 18 to 89 years, with a mean age of 54.2 years.
Biologic use was defined as patients who started or had ongoing use of biologics between 12 months before enrollment and the patient’s most recent data collection. Switches were defined as stopping one biologic and starting another within 6 months; stops were defined as discontinuing a biologic without switching to another within 6 months. A total of 66% of the patients were using biologics at the time of study enrollment. The most common biologic was omalizumab (47%), followed by benralizumab (27%), mepolizumab (26%), and dupilumab (18%).
Overall, 89% of the patients had ongoing biologic use, 16% had biologic switches, and 13% had stops.
Patients who started biologics or switched biologics had significant reductions in asthma exacerbations at 6 months, compared with nonbiologic users of 58% (1.80 vs. 0.76 per patient-year) and 49% (1.47 vs. 0.75 per patient-year), respectively (P < .001 for both). Asthma exacerbations declined by 70% among biologics users for whom data were available for 12 months before and 12 months after starting biologics.
Exacerbations decreased at 6 months after biologic initiation across all subgroups of patients, notably patients with pre-biologic FEV1 < 80% and patients with FEV1 ≥ 80% (66% and 53%, respectively); never smokers and current/former smokers (63% and 50%, respectively); and patients with COPD and without COPD (58% and 52%, respectively).
The researchers also found a greater reduction in exacerbations among patients who switched from anti-IgE therapy to anti–IL-5/IL-5R/IL-4R therapy, compared with those who switched among anti–IL-5/IL-5R/IL-4R therapies (58% vs. 46%).
Patients who stopped or switched biologics appeared to have more severe or treatment-refractory disease than those with ongoing biologic use, the researchers noted. The most common reason for stopping or switching was worsening symptoms.
The study findings were limited by several factors, including the focus only on adults in the United States with subspecialist-treated SA, which may limit generalizability to children or other populations, the researchers noted. Other limitations included the variation in clinical decisions and insurance coverage and the inability to conduct longitudinal assessments, they said.
The results demonstrate that starting or switching biologics was consistently associated with fewer exacerbations in severe asthma. However, more research is needed to determine why some patients were not receiving biologics because they were not considered clinically eligible by their subspecialist health care providers, the researchers concluded.
The current study and the CHRONICLE study were supported by AstraZeneca. Lead author Dr. Panettieri disclosed serving on the advisory boards for and receiving grant support from AstraZeneca, Sanofi, Genentech, Regeneron, and Novartis.
, based on data from more than 2,000 individuals.
The development of biologics to target specific inflammatory pathways “has transformed the management of uncontrolled SA,” but data on the real-world use of biologics in severe asthma patients treated by subspecialists are limited, wrote Reynold A. Panettieri, Jr., MD, of Rutgers, State University of New Jersey, New Brunswick, and colleagues.
In a study published in the Annals of Allergy, Asthma & Immunology, the researchers reviewed data from CHRONICLE, an ongoing, prospective, real-world noninterventional study of adults aged 18 years and older with severe asthma in the United States.
The study population included 2,847 patients enrolled in the CHRONICLE study between February 2018 and February 2021; 68.8% were women, 74.6% were White. The patients ranged in age from 18 to 89 years, with a mean age of 54.2 years.
Biologic use was defined as patients who started or had ongoing use of biologics between 12 months before enrollment and the patient’s most recent data collection. Switches were defined as stopping one biologic and starting another within 6 months; stops were defined as discontinuing a biologic without switching to another within 6 months. A total of 66% of the patients were using biologics at the time of study enrollment. The most common biologic was omalizumab (47%), followed by benralizumab (27%), mepolizumab (26%), and dupilumab (18%).
Overall, 89% of the patients had ongoing biologic use, 16% had biologic switches, and 13% had stops.
Patients who started biologics or switched biologics had significant reductions in asthma exacerbations at 6 months, compared with nonbiologic users of 58% (1.80 vs. 0.76 per patient-year) and 49% (1.47 vs. 0.75 per patient-year), respectively (P < .001 for both). Asthma exacerbations declined by 70% among biologics users for whom data were available for 12 months before and 12 months after starting biologics.
Exacerbations decreased at 6 months after biologic initiation across all subgroups of patients, notably patients with pre-biologic FEV1 < 80% and patients with FEV1 ≥ 80% (66% and 53%, respectively); never smokers and current/former smokers (63% and 50%, respectively); and patients with COPD and without COPD (58% and 52%, respectively).
The researchers also found a greater reduction in exacerbations among patients who switched from anti-IgE therapy to anti–IL-5/IL-5R/IL-4R therapy, compared with those who switched among anti–IL-5/IL-5R/IL-4R therapies (58% vs. 46%).
Patients who stopped or switched biologics appeared to have more severe or treatment-refractory disease than those with ongoing biologic use, the researchers noted. The most common reason for stopping or switching was worsening symptoms.
The study findings were limited by several factors, including the focus only on adults in the United States with subspecialist-treated SA, which may limit generalizability to children or other populations, the researchers noted. Other limitations included the variation in clinical decisions and insurance coverage and the inability to conduct longitudinal assessments, they said.
The results demonstrate that starting or switching biologics was consistently associated with fewer exacerbations in severe asthma. However, more research is needed to determine why some patients were not receiving biologics because they were not considered clinically eligible by their subspecialist health care providers, the researchers concluded.
The current study and the CHRONICLE study were supported by AstraZeneca. Lead author Dr. Panettieri disclosed serving on the advisory boards for and receiving grant support from AstraZeneca, Sanofi, Genentech, Regeneron, and Novartis.
, based on data from more than 2,000 individuals.
The development of biologics to target specific inflammatory pathways “has transformed the management of uncontrolled SA,” but data on the real-world use of biologics in severe asthma patients treated by subspecialists are limited, wrote Reynold A. Panettieri, Jr., MD, of Rutgers, State University of New Jersey, New Brunswick, and colleagues.
In a study published in the Annals of Allergy, Asthma & Immunology, the researchers reviewed data from CHRONICLE, an ongoing, prospective, real-world noninterventional study of adults aged 18 years and older with severe asthma in the United States.
The study population included 2,847 patients enrolled in the CHRONICLE study between February 2018 and February 2021; 68.8% were women, 74.6% were White. The patients ranged in age from 18 to 89 years, with a mean age of 54.2 years.
Biologic use was defined as patients who started or had ongoing use of biologics between 12 months before enrollment and the patient’s most recent data collection. Switches were defined as stopping one biologic and starting another within 6 months; stops were defined as discontinuing a biologic without switching to another within 6 months. A total of 66% of the patients were using biologics at the time of study enrollment. The most common biologic was omalizumab (47%), followed by benralizumab (27%), mepolizumab (26%), and dupilumab (18%).
Overall, 89% of the patients had ongoing biologic use, 16% had biologic switches, and 13% had stops.
Patients who started biologics or switched biologics had significant reductions in asthma exacerbations at 6 months, compared with nonbiologic users of 58% (1.80 vs. 0.76 per patient-year) and 49% (1.47 vs. 0.75 per patient-year), respectively (P < .001 for both). Asthma exacerbations declined by 70% among biologics users for whom data were available for 12 months before and 12 months after starting biologics.
Exacerbations decreased at 6 months after biologic initiation across all subgroups of patients, notably patients with pre-biologic FEV1 < 80% and patients with FEV1 ≥ 80% (66% and 53%, respectively); never smokers and current/former smokers (63% and 50%, respectively); and patients with COPD and without COPD (58% and 52%, respectively).
The researchers also found a greater reduction in exacerbations among patients who switched from anti-IgE therapy to anti–IL-5/IL-5R/IL-4R therapy, compared with those who switched among anti–IL-5/IL-5R/IL-4R therapies (58% vs. 46%).
Patients who stopped or switched biologics appeared to have more severe or treatment-refractory disease than those with ongoing biologic use, the researchers noted. The most common reason for stopping or switching was worsening symptoms.
The study findings were limited by several factors, including the focus only on adults in the United States with subspecialist-treated SA, which may limit generalizability to children or other populations, the researchers noted. Other limitations included the variation in clinical decisions and insurance coverage and the inability to conduct longitudinal assessments, they said.
The results demonstrate that starting or switching biologics was consistently associated with fewer exacerbations in severe asthma. However, more research is needed to determine why some patients were not receiving biologics because they were not considered clinically eligible by their subspecialist health care providers, the researchers concluded.
The current study and the CHRONICLE study were supported by AstraZeneca. Lead author Dr. Panettieri disclosed serving on the advisory boards for and receiving grant support from AstraZeneca, Sanofi, Genentech, Regeneron, and Novartis.
FROM THE ANNALS OF ALLERGY, ASTHMA & IMMUNOLOGY
COPD predicts hospital readmission after fractures
Chronic obstructive pulmonary disease (COPD) was among the significant predictors of hospital readmission in older adults with fractures, based on data from nearly 400 individuals.
Fractures in the elderly remain a major health concern, and readmissions are common; however, Lara Cristina da Cunha Guimarães, MSN, of State School of Public Health Candido Santiago, State Department of Health of Goiás (Brazil), and colleagues wrote.
Previous research suggests that readmissions risk may be greater in patients with preadmission conditions including pulmonary and cardiac disease, history of stroke and other neurological conditions, and other factors associated with aging in general, they said.
In a study published in the journal Injury , the researchers reviewed data from 376 adults aged 60 years and older in a trauma referral hospital in Brazil who had suffered fractures and were hospitalized between Sept. 1, 2016, and Feb. 28, 2017. The primary outcome was readmission up to one year after discharge from the initial hospitalization for fracture.
Approximately half of the patients experienced femur fractures (53.2%), and the most frequent cause was falling from standing height (72.9%). The overall incidence of readmission was 20.7%. A total of 30.5% of readmissions were related to the fracture, and surgical-site infections were the most common cause of fracture-related complications.
More than half (58.3%) of the readmissions were related to clinical complications.
In a multivariate analysis, several clinical factors not related to fractures were independently associated with readmission, including a previous diagnosis of COPD, age between 60 and 69 years, a fracture of the femur, and delirium at the time of the first hospitalization for fracture.
Pneumonia was the most frequent cause of clinical complications, reflecting data from other recent studies, the researchers noted. “Elderly people with COPD are more susceptible to infections, such as pneumonia, which was a cause of frequent readmissions in the population studied. The presence of COPD can contribute to imbalance in the pulmonary microbiome, mucus production and persistent inflammation of the airways, and structural damage, which increases exposure of the pulmonary mucosa to pathogens.” COPD also can be associated with cardiovascular, mental, and musculoskeletal diseases that can further complicate and delay recovery from fractures.
The study findings were limited by the potential for incomplete information in medical records. However, the results indicate a range of causes and conditions associated with hospital readmission after fractures in older adults, they said. Recognizing these factors can guide plans for transitions from hospital to home care to reduce complications and readmissions.
The study received no outside funding. The researchers had no financial conflicts to disclose.
Chronic obstructive pulmonary disease (COPD) was among the significant predictors of hospital readmission in older adults with fractures, based on data from nearly 400 individuals.
Fractures in the elderly remain a major health concern, and readmissions are common; however, Lara Cristina da Cunha Guimarães, MSN, of State School of Public Health Candido Santiago, State Department of Health of Goiás (Brazil), and colleagues wrote.
Previous research suggests that readmissions risk may be greater in patients with preadmission conditions including pulmonary and cardiac disease, history of stroke and other neurological conditions, and other factors associated with aging in general, they said.
In a study published in the journal Injury , the researchers reviewed data from 376 adults aged 60 years and older in a trauma referral hospital in Brazil who had suffered fractures and were hospitalized between Sept. 1, 2016, and Feb. 28, 2017. The primary outcome was readmission up to one year after discharge from the initial hospitalization for fracture.
Approximately half of the patients experienced femur fractures (53.2%), and the most frequent cause was falling from standing height (72.9%). The overall incidence of readmission was 20.7%. A total of 30.5% of readmissions were related to the fracture, and surgical-site infections were the most common cause of fracture-related complications.
More than half (58.3%) of the readmissions were related to clinical complications.
In a multivariate analysis, several clinical factors not related to fractures were independently associated with readmission, including a previous diagnosis of COPD, age between 60 and 69 years, a fracture of the femur, and delirium at the time of the first hospitalization for fracture.
Pneumonia was the most frequent cause of clinical complications, reflecting data from other recent studies, the researchers noted. “Elderly people with COPD are more susceptible to infections, such as pneumonia, which was a cause of frequent readmissions in the population studied. The presence of COPD can contribute to imbalance in the pulmonary microbiome, mucus production and persistent inflammation of the airways, and structural damage, which increases exposure of the pulmonary mucosa to pathogens.” COPD also can be associated with cardiovascular, mental, and musculoskeletal diseases that can further complicate and delay recovery from fractures.
The study findings were limited by the potential for incomplete information in medical records. However, the results indicate a range of causes and conditions associated with hospital readmission after fractures in older adults, they said. Recognizing these factors can guide plans for transitions from hospital to home care to reduce complications and readmissions.
The study received no outside funding. The researchers had no financial conflicts to disclose.
Chronic obstructive pulmonary disease (COPD) was among the significant predictors of hospital readmission in older adults with fractures, based on data from nearly 400 individuals.
Fractures in the elderly remain a major health concern, and readmissions are common; however, Lara Cristina da Cunha Guimarães, MSN, of State School of Public Health Candido Santiago, State Department of Health of Goiás (Brazil), and colleagues wrote.
Previous research suggests that readmissions risk may be greater in patients with preadmission conditions including pulmonary and cardiac disease, history of stroke and other neurological conditions, and other factors associated with aging in general, they said.
In a study published in the journal Injury , the researchers reviewed data from 376 adults aged 60 years and older in a trauma referral hospital in Brazil who had suffered fractures and were hospitalized between Sept. 1, 2016, and Feb. 28, 2017. The primary outcome was readmission up to one year after discharge from the initial hospitalization for fracture.
Approximately half of the patients experienced femur fractures (53.2%), and the most frequent cause was falling from standing height (72.9%). The overall incidence of readmission was 20.7%. A total of 30.5% of readmissions were related to the fracture, and surgical-site infections were the most common cause of fracture-related complications.
More than half (58.3%) of the readmissions were related to clinical complications.
In a multivariate analysis, several clinical factors not related to fractures were independently associated with readmission, including a previous diagnosis of COPD, age between 60 and 69 years, a fracture of the femur, and delirium at the time of the first hospitalization for fracture.
Pneumonia was the most frequent cause of clinical complications, reflecting data from other recent studies, the researchers noted. “Elderly people with COPD are more susceptible to infections, such as pneumonia, which was a cause of frequent readmissions in the population studied. The presence of COPD can contribute to imbalance in the pulmonary microbiome, mucus production and persistent inflammation of the airways, and structural damage, which increases exposure of the pulmonary mucosa to pathogens.” COPD also can be associated with cardiovascular, mental, and musculoskeletal diseases that can further complicate and delay recovery from fractures.
The study findings were limited by the potential for incomplete information in medical records. However, the results indicate a range of causes and conditions associated with hospital readmission after fractures in older adults, they said. Recognizing these factors can guide plans for transitions from hospital to home care to reduce complications and readmissions.
The study received no outside funding. The researchers had no financial conflicts to disclose.
FROM INJURY