User login
Lucas Franki is an associate editor for MDedge News, and has been with the company since 2014. He has a BA in English from Penn State University and is an Eagle Scout.
FDA approves alectinib as frontline therapy for ALK-positive metastatic NSCLC
The Food and Drug Administration has approved frontline alectinib for the treatment of anaplastic lymphoma kinase (ALK)–positive metastatic non–small cell lung cancer (NSCLC) that has been detected by an FDA-approved test.
The drug previously received accelerated approval for treatment of patients with ALK-positive metastatic NSCLC whose disease progressed while receiving crizotinib or who were intolerant of that treatment.
There was also a lower incidence of progression to the central nervous system as first site of disease progression with alectinib: Incidence of progression to the CNS was 12% for patients receiving alectinib and 45% for patients receiving crizotinib.
The most common adverse events in patients receiving alectinib were fatigue, constipation, edema, myalgia, and anemia. Serious adverse events occurred in 28% of patients. Adverse events resulting in discontinuation occurred in 11% of patients.
“All patients in the trial were required to have evidence of ALK-rearrangement identified by the VENTANA ALK (D5F3) CDx Assay performed through central laboratory testing,” the FDA said in a press statement.
The recommended dose is 600 mg orally taken twice daily with food.
The Food and Drug Administration has approved frontline alectinib for the treatment of anaplastic lymphoma kinase (ALK)–positive metastatic non–small cell lung cancer (NSCLC) that has been detected by an FDA-approved test.
The drug previously received accelerated approval for treatment of patients with ALK-positive metastatic NSCLC whose disease progressed while receiving crizotinib or who were intolerant of that treatment.
There was also a lower incidence of progression to the central nervous system as first site of disease progression with alectinib: Incidence of progression to the CNS was 12% for patients receiving alectinib and 45% for patients receiving crizotinib.
The most common adverse events in patients receiving alectinib were fatigue, constipation, edema, myalgia, and anemia. Serious adverse events occurred in 28% of patients. Adverse events resulting in discontinuation occurred in 11% of patients.
“All patients in the trial were required to have evidence of ALK-rearrangement identified by the VENTANA ALK (D5F3) CDx Assay performed through central laboratory testing,” the FDA said in a press statement.
The recommended dose is 600 mg orally taken twice daily with food.
The Food and Drug Administration has approved frontline alectinib for the treatment of anaplastic lymphoma kinase (ALK)–positive metastatic non–small cell lung cancer (NSCLC) that has been detected by an FDA-approved test.
The drug previously received accelerated approval for treatment of patients with ALK-positive metastatic NSCLC whose disease progressed while receiving crizotinib or who were intolerant of that treatment.
There was also a lower incidence of progression to the central nervous system as first site of disease progression with alectinib: Incidence of progression to the CNS was 12% for patients receiving alectinib and 45% for patients receiving crizotinib.
The most common adverse events in patients receiving alectinib were fatigue, constipation, edema, myalgia, and anemia. Serious adverse events occurred in 28% of patients. Adverse events resulting in discontinuation occurred in 11% of patients.
“All patients in the trial were required to have evidence of ALK-rearrangement identified by the VENTANA ALK (D5F3) CDx Assay performed through central laboratory testing,” the FDA said in a press statement.
The recommended dose is 600 mg orally taken twice daily with food.
Case study suggests maternal inheritance of PBC susceptibility
A case report involving a family with four sisters with primary biliary cholangitis (PBC) suggests that there may be a maternal inheritance of susceptibility, according to Saeam Shin, MD, and associates at Hallym University in Seoul, South Korea.
In the first case, a 56-year-old woman was diagnosed with PBC, and afterwards, her three sisters, her brother, and her half-sister born to a different mother were evaluated for PBC as well. The second and fourth sisters showed no symptoms, but they were antimitochondrial-antibody (AMA) positive and were diagnosed with PBC. The third sister had been admitted to a different hospital for acute hepatitis of unknown origin – after receiving a positive AMA test, she also was diagnosed with PBC.
The brother and half-sister evaluated for PBC showed no symptoms and had negative AMA tests. The four sisters diagnosed showed good response to ursodeoxycholic acid in liver biochemistry tests and have continued on that medication without complication.
“If one patient is diagnosed with PBC, screening with AMA and liver function tests should be recommended to other family members for the early detection and management of this condition, especially for female relatives,” the investigators wrote.
Find the full case report in the World Journal of Gastroenterology (doi: 10.3748/wjg.v23.i39.7191).
A case report involving a family with four sisters with primary biliary cholangitis (PBC) suggests that there may be a maternal inheritance of susceptibility, according to Saeam Shin, MD, and associates at Hallym University in Seoul, South Korea.
In the first case, a 56-year-old woman was diagnosed with PBC, and afterwards, her three sisters, her brother, and her half-sister born to a different mother were evaluated for PBC as well. The second and fourth sisters showed no symptoms, but they were antimitochondrial-antibody (AMA) positive and were diagnosed with PBC. The third sister had been admitted to a different hospital for acute hepatitis of unknown origin – after receiving a positive AMA test, she also was diagnosed with PBC.
The brother and half-sister evaluated for PBC showed no symptoms and had negative AMA tests. The four sisters diagnosed showed good response to ursodeoxycholic acid in liver biochemistry tests and have continued on that medication without complication.
“If one patient is diagnosed with PBC, screening with AMA and liver function tests should be recommended to other family members for the early detection and management of this condition, especially for female relatives,” the investigators wrote.
Find the full case report in the World Journal of Gastroenterology (doi: 10.3748/wjg.v23.i39.7191).
A case report involving a family with four sisters with primary biliary cholangitis (PBC) suggests that there may be a maternal inheritance of susceptibility, according to Saeam Shin, MD, and associates at Hallym University in Seoul, South Korea.
In the first case, a 56-year-old woman was diagnosed with PBC, and afterwards, her three sisters, her brother, and her half-sister born to a different mother were evaluated for PBC as well. The second and fourth sisters showed no symptoms, but they were antimitochondrial-antibody (AMA) positive and were diagnosed with PBC. The third sister had been admitted to a different hospital for acute hepatitis of unknown origin – after receiving a positive AMA test, she also was diagnosed with PBC.
The brother and half-sister evaluated for PBC showed no symptoms and had negative AMA tests. The four sisters diagnosed showed good response to ursodeoxycholic acid in liver biochemistry tests and have continued on that medication without complication.
“If one patient is diagnosed with PBC, screening with AMA and liver function tests should be recommended to other family members for the early detection and management of this condition, especially for female relatives,” the investigators wrote.
Find the full case report in the World Journal of Gastroenterology (doi: 10.3748/wjg.v23.i39.7191).
FROM THE WORLD JOURNAL OF GASTROENTEROLOGY
U.S. judge orders Philips to cease AED manufacturing
A U.S. District Judge has ordered Philips North America, as well as two Philips officers, to cease manufacturing and distribution of automatic external defibrillators (AEDs) until they can comply with federal regulations in a consent decree, according to a statement from the Food and Drug Administration.
In a complaint filed with the decree, Philips North America in Andover, Mass., which operates as Philips Medical Systems and Philips Healthcare, sold compromised automatic external defibrillators and Q-CPR Meters in violation of current Federal Food, Drug and Cosmetic (FD&C) Act good manufacturing practice requirements. The injunction also applies to Carla Kriwet and Ojas Buch of the Patient Care and Monitoring Solutions business group, according to the statement.
“AEDs are life-saving tools and are designed to be used by the general public or professionals in an emergency. People rely on these devices to work when needed. By not adequately addressing corrective and preventative actions with their AEDs in a timely manner, Philips distributed adulterated products that put people at risk,” Melinda Plaisier, associate commissioner for regulatory affairs at the FDA said in the press release.
In an Oct. 11 statement, Carla Kriwet, head of Connected Care & Health Informatics at Royal Philips, said “We are committed to delivering high-quality, innovative products and solutions, and we take this matter very seriously. We are fully prepared to fulfill the terms of the decree, and we hope to resume the suspended defibrillator production in the course of 2018.”
Ms. Kriwet added that in the past several years Philips has made significant investments in its quality procedures and leadership.
The company recommends that Philips defibrillators currently in use by customers should remain in use, and should not be taken out of service as Philips has no reason to believe they pose a risk to patients.
A U.S. District Judge has ordered Philips North America, as well as two Philips officers, to cease manufacturing and distribution of automatic external defibrillators (AEDs) until they can comply with federal regulations in a consent decree, according to a statement from the Food and Drug Administration.
In a complaint filed with the decree, Philips North America in Andover, Mass., which operates as Philips Medical Systems and Philips Healthcare, sold compromised automatic external defibrillators and Q-CPR Meters in violation of current Federal Food, Drug and Cosmetic (FD&C) Act good manufacturing practice requirements. The injunction also applies to Carla Kriwet and Ojas Buch of the Patient Care and Monitoring Solutions business group, according to the statement.
“AEDs are life-saving tools and are designed to be used by the general public or professionals in an emergency. People rely on these devices to work when needed. By not adequately addressing corrective and preventative actions with their AEDs in a timely manner, Philips distributed adulterated products that put people at risk,” Melinda Plaisier, associate commissioner for regulatory affairs at the FDA said in the press release.
In an Oct. 11 statement, Carla Kriwet, head of Connected Care & Health Informatics at Royal Philips, said “We are committed to delivering high-quality, innovative products and solutions, and we take this matter very seriously. We are fully prepared to fulfill the terms of the decree, and we hope to resume the suspended defibrillator production in the course of 2018.”
Ms. Kriwet added that in the past several years Philips has made significant investments in its quality procedures and leadership.
The company recommends that Philips defibrillators currently in use by customers should remain in use, and should not be taken out of service as Philips has no reason to believe they pose a risk to patients.
A U.S. District Judge has ordered Philips North America, as well as two Philips officers, to cease manufacturing and distribution of automatic external defibrillators (AEDs) until they can comply with federal regulations in a consent decree, according to a statement from the Food and Drug Administration.
In a complaint filed with the decree, Philips North America in Andover, Mass., which operates as Philips Medical Systems and Philips Healthcare, sold compromised automatic external defibrillators and Q-CPR Meters in violation of current Federal Food, Drug and Cosmetic (FD&C) Act good manufacturing practice requirements. The injunction also applies to Carla Kriwet and Ojas Buch of the Patient Care and Monitoring Solutions business group, according to the statement.
“AEDs are life-saving tools and are designed to be used by the general public or professionals in an emergency. People rely on these devices to work when needed. By not adequately addressing corrective and preventative actions with their AEDs in a timely manner, Philips distributed adulterated products that put people at risk,” Melinda Plaisier, associate commissioner for regulatory affairs at the FDA said in the press release.
In an Oct. 11 statement, Carla Kriwet, head of Connected Care & Health Informatics at Royal Philips, said “We are committed to delivering high-quality, innovative products and solutions, and we take this matter very seriously. We are fully prepared to fulfill the terms of the decree, and we hope to resume the suspended defibrillator production in the course of 2018.”
Ms. Kriwet added that in the past several years Philips has made significant investments in its quality procedures and leadership.
The company recommends that Philips defibrillators currently in use by customers should remain in use, and should not be taken out of service as Philips has no reason to believe they pose a risk to patients.
Fetal fibronectin may be underused in preterm birth detection
Only a small percentage of pregnant women with symptoms of preterm labor who are admitted to emergency departments or labor and delivery units receive fetal fibronectin testing, suggesting the test may not be fully utilized, according to results from a retrospective study.
Using data collected from the Medical Outcomes Research for Effectiveness and Economics Registry, 23,062 patients were included in the study, of whom just 12% received fetal fibronectin (fFN) testing, according to Sean C. Blackwell, MD, of the department of obstetrics, gynecology and reproductive sciences at the University of Texas, Houston, and his associates (Clinicoecon Outcomes Res. 2017 Oct 3;9:585-94. doi: 10.2147/CEOR.S141061).
The rate of fFN testing was even lower – 4.2% – among women who were discharged home but gave birth within 3 days, compared with the testing rate of 16.7% in women who were discharged and did not give birth within 3 days, which suggests “that there may be opportunities to improve the care management of these patients with the use of such screening tools as fFN testing,” the researchers wrote.
Patients who resided in the Northeast were less likely to receive fFN testing, while patients in the West were slightly more likely to receive testing. Patients with more all-cause physician visits and who had received transvaginal ultrasound also were more likely to receive fFN testing, they reported.
“Additional research is needed to determine how to use quantitative fFN testing tools currently under development as part of screening for risk of [preterm labor] allowing physicians the opportunity to better understand patient risk factors and tailor interventions to optimize pre- and perinatal care for the woman and her neonate,” Dr. Blackwell and his associates wrote.
Three of the study authors are employees of Avalere Health, which received funding from Hologic Inc., to support the study. Another author is an employee of Hologic, which produces fFN tests. The authors had no other disclosures.
Only a small percentage of pregnant women with symptoms of preterm labor who are admitted to emergency departments or labor and delivery units receive fetal fibronectin testing, suggesting the test may not be fully utilized, according to results from a retrospective study.
Using data collected from the Medical Outcomes Research for Effectiveness and Economics Registry, 23,062 patients were included in the study, of whom just 12% received fetal fibronectin (fFN) testing, according to Sean C. Blackwell, MD, of the department of obstetrics, gynecology and reproductive sciences at the University of Texas, Houston, and his associates (Clinicoecon Outcomes Res. 2017 Oct 3;9:585-94. doi: 10.2147/CEOR.S141061).
The rate of fFN testing was even lower – 4.2% – among women who were discharged home but gave birth within 3 days, compared with the testing rate of 16.7% in women who were discharged and did not give birth within 3 days, which suggests “that there may be opportunities to improve the care management of these patients with the use of such screening tools as fFN testing,” the researchers wrote.
Patients who resided in the Northeast were less likely to receive fFN testing, while patients in the West were slightly more likely to receive testing. Patients with more all-cause physician visits and who had received transvaginal ultrasound also were more likely to receive fFN testing, they reported.
“Additional research is needed to determine how to use quantitative fFN testing tools currently under development as part of screening for risk of [preterm labor] allowing physicians the opportunity to better understand patient risk factors and tailor interventions to optimize pre- and perinatal care for the woman and her neonate,” Dr. Blackwell and his associates wrote.
Three of the study authors are employees of Avalere Health, which received funding from Hologic Inc., to support the study. Another author is an employee of Hologic, which produces fFN tests. The authors had no other disclosures.
Only a small percentage of pregnant women with symptoms of preterm labor who are admitted to emergency departments or labor and delivery units receive fetal fibronectin testing, suggesting the test may not be fully utilized, according to results from a retrospective study.
Using data collected from the Medical Outcomes Research for Effectiveness and Economics Registry, 23,062 patients were included in the study, of whom just 12% received fetal fibronectin (fFN) testing, according to Sean C. Blackwell, MD, of the department of obstetrics, gynecology and reproductive sciences at the University of Texas, Houston, and his associates (Clinicoecon Outcomes Res. 2017 Oct 3;9:585-94. doi: 10.2147/CEOR.S141061).
The rate of fFN testing was even lower – 4.2% – among women who were discharged home but gave birth within 3 days, compared with the testing rate of 16.7% in women who were discharged and did not give birth within 3 days, which suggests “that there may be opportunities to improve the care management of these patients with the use of such screening tools as fFN testing,” the researchers wrote.
Patients who resided in the Northeast were less likely to receive fFN testing, while patients in the West were slightly more likely to receive testing. Patients with more all-cause physician visits and who had received transvaginal ultrasound also were more likely to receive fFN testing, they reported.
“Additional research is needed to determine how to use quantitative fFN testing tools currently under development as part of screening for risk of [preterm labor] allowing physicians the opportunity to better understand patient risk factors and tailor interventions to optimize pre- and perinatal care for the woman and her neonate,” Dr. Blackwell and his associates wrote.
Three of the study authors are employees of Avalere Health, which received funding from Hologic Inc., to support the study. Another author is an employee of Hologic, which produces fFN tests. The authors had no other disclosures.
FROM CLINICOECONOMICS AND OUTCOMES RESEARCH
Key clinical point:
Major finding: Pregnant women with symptoms of preterm labor received fFN testing at an overall rate of 12%.
Data source: A total of 23,062 patients from the Medical Outcomes Research for Effectiveness and Economics Registry.
Disclosures: Three of the study authors are employees of Avalere Health, which received funding from Hologic Inc., to support the study. Another author is an employee of Hologic, which produces fFN tests. The authors had no other disclosures.
Driving ability of Parkinson’s patients can decline quickly
according to a longitudinal cohort study conducted by Urgun Uc, MD, and associates.
While some patients with Parkinson’s disease who took part in a test of driving skills on a standardized driving course performed significantly worse at a baseline assessment than did healthy control patients, the patients who performed at a similar level as the control group made a significantly higher number of driving mistakes 2 years later on a follow-up evaluation.
Among the original 67 Parkinson’s disease patients, the 28 who returned for repeat testing at 2 years had performed just as well at baseline as the 69 control group returnees. But at the 2-year follow-up, the Parkinson’s disease group made an average of 49.7 mistakes, while the control group made 34.6 mistakes.
In addition to an increase in overall driving errors, Parkinson’s disease patients also made a significantly higher number of lane observance, overtaking, turning, miscellaneous, and serious errors after 2 years than did the control group. Risk factors for worse performance after 2 years included greater error count and worse visual acuity at baseline, and worsening of global cognition, Unified Parkinson’s Disease Rating Scale scores, executive function, visual-processing speed, and attention.
“Health care providers for patients with Parkinson’s disease should routinely inquire about driving status and make necessary referrals for evaluation of driving fitness as needed. Further research is needed to determine if improvement of underlying impairments in visual perception, executive function, and motor abilities through physical exercise and cognitive training can preserve driving ability in Parkinson’s disease for a longer time,” the investigators concluded.
Read the full study in Neurology (doi: 10.1212/WNL.0000000000004629).
according to a longitudinal cohort study conducted by Urgun Uc, MD, and associates.
While some patients with Parkinson’s disease who took part in a test of driving skills on a standardized driving course performed significantly worse at a baseline assessment than did healthy control patients, the patients who performed at a similar level as the control group made a significantly higher number of driving mistakes 2 years later on a follow-up evaluation.
Among the original 67 Parkinson’s disease patients, the 28 who returned for repeat testing at 2 years had performed just as well at baseline as the 69 control group returnees. But at the 2-year follow-up, the Parkinson’s disease group made an average of 49.7 mistakes, while the control group made 34.6 mistakes.
In addition to an increase in overall driving errors, Parkinson’s disease patients also made a significantly higher number of lane observance, overtaking, turning, miscellaneous, and serious errors after 2 years than did the control group. Risk factors for worse performance after 2 years included greater error count and worse visual acuity at baseline, and worsening of global cognition, Unified Parkinson’s Disease Rating Scale scores, executive function, visual-processing speed, and attention.
“Health care providers for patients with Parkinson’s disease should routinely inquire about driving status and make necessary referrals for evaluation of driving fitness as needed. Further research is needed to determine if improvement of underlying impairments in visual perception, executive function, and motor abilities through physical exercise and cognitive training can preserve driving ability in Parkinson’s disease for a longer time,” the investigators concluded.
Read the full study in Neurology (doi: 10.1212/WNL.0000000000004629).
according to a longitudinal cohort study conducted by Urgun Uc, MD, and associates.
While some patients with Parkinson’s disease who took part in a test of driving skills on a standardized driving course performed significantly worse at a baseline assessment than did healthy control patients, the patients who performed at a similar level as the control group made a significantly higher number of driving mistakes 2 years later on a follow-up evaluation.
Among the original 67 Parkinson’s disease patients, the 28 who returned for repeat testing at 2 years had performed just as well at baseline as the 69 control group returnees. But at the 2-year follow-up, the Parkinson’s disease group made an average of 49.7 mistakes, while the control group made 34.6 mistakes.
In addition to an increase in overall driving errors, Parkinson’s disease patients also made a significantly higher number of lane observance, overtaking, turning, miscellaneous, and serious errors after 2 years than did the control group. Risk factors for worse performance after 2 years included greater error count and worse visual acuity at baseline, and worsening of global cognition, Unified Parkinson’s Disease Rating Scale scores, executive function, visual-processing speed, and attention.
“Health care providers for patients with Parkinson’s disease should routinely inquire about driving status and make necessary referrals for evaluation of driving fitness as needed. Further research is needed to determine if improvement of underlying impairments in visual perception, executive function, and motor abilities through physical exercise and cognitive training can preserve driving ability in Parkinson’s disease for a longer time,” the investigators concluded.
Read the full study in Neurology (doi: 10.1212/WNL.0000000000004629).
FROM NEUROLOGY
JZP-110 improves excessive sleepiness in OSA patients
The selective dopamine norepinephrine reuptake inhibitor JZP-110 was effective at treating excessive sleepiness in obstructive sleep apnea patients, according to an abstract on a study to be presented at the CHEST annual meeting.
In a 12-week, phase 3 trial, adult patients with obstructive sleep apnea were randomized to receive placebo or once-daily JZP-110 at dosages of 37.5 mg, 75 mg, 150 mg, or 300 mg. A total of 459 patients were included in the final analysis.
At the end of the trial, the mean reduction from baseline on a maintenance of wakefulness test was 0.2 minutes for patients in the placebo group, 4.7 minutes in the 37.5-mg group, 9.1 minutes in the 75-mg group, 11 minutes in the 150-mg group, and 13 minutes in the 300-mg group. Mean changes in Epworth Sleepiness Scale scores were –3.3 for patients in the placebo group, –5.1 in the 37.5-mg group, –5 in the 75-mg group, –7.7 in the 150-mg group, and –7.9 in the 300-mg group.
A significantly higher rate of patients in the 75-mg (72.4%), 150-mg (89.7%), and 300-mg (88.7%) groups improved on the Patient Global Impression of Change scale, compared with patients in the placebo (49.1%) and 37.5-mg (55.4%) groups.
Just under 68% of patients who received JZP-110 experienced at least one adverse event, compared with 47.9% of patients who received placebo. The most common adverse events were headache, nausea, decreased appetite, and anxiety. Six serious adverse events were reported over the study period, but none was related to JZP-110.
The study is scheduled to be presented on Sunday, Oct. 29, from 1:30 p.m. to 1:45 p.m. in Room 601A of the Toronto Convention Centre South Building as part of the “Obstructive Sleep Apnea: Insights & Management” session, which will run from 1:30 p.m. to 3 p.m.
The selective dopamine norepinephrine reuptake inhibitor JZP-110 was effective at treating excessive sleepiness in obstructive sleep apnea patients, according to an abstract on a study to be presented at the CHEST annual meeting.
In a 12-week, phase 3 trial, adult patients with obstructive sleep apnea were randomized to receive placebo or once-daily JZP-110 at dosages of 37.5 mg, 75 mg, 150 mg, or 300 mg. A total of 459 patients were included in the final analysis.
At the end of the trial, the mean reduction from baseline on a maintenance of wakefulness test was 0.2 minutes for patients in the placebo group, 4.7 minutes in the 37.5-mg group, 9.1 minutes in the 75-mg group, 11 minutes in the 150-mg group, and 13 minutes in the 300-mg group. Mean changes in Epworth Sleepiness Scale scores were –3.3 for patients in the placebo group, –5.1 in the 37.5-mg group, –5 in the 75-mg group, –7.7 in the 150-mg group, and –7.9 in the 300-mg group.
A significantly higher rate of patients in the 75-mg (72.4%), 150-mg (89.7%), and 300-mg (88.7%) groups improved on the Patient Global Impression of Change scale, compared with patients in the placebo (49.1%) and 37.5-mg (55.4%) groups.
Just under 68% of patients who received JZP-110 experienced at least one adverse event, compared with 47.9% of patients who received placebo. The most common adverse events were headache, nausea, decreased appetite, and anxiety. Six serious adverse events were reported over the study period, but none was related to JZP-110.
The study is scheduled to be presented on Sunday, Oct. 29, from 1:30 p.m. to 1:45 p.m. in Room 601A of the Toronto Convention Centre South Building as part of the “Obstructive Sleep Apnea: Insights & Management” session, which will run from 1:30 p.m. to 3 p.m.
The selective dopamine norepinephrine reuptake inhibitor JZP-110 was effective at treating excessive sleepiness in obstructive sleep apnea patients, according to an abstract on a study to be presented at the CHEST annual meeting.
In a 12-week, phase 3 trial, adult patients with obstructive sleep apnea were randomized to receive placebo or once-daily JZP-110 at dosages of 37.5 mg, 75 mg, 150 mg, or 300 mg. A total of 459 patients were included in the final analysis.
At the end of the trial, the mean reduction from baseline on a maintenance of wakefulness test was 0.2 minutes for patients in the placebo group, 4.7 minutes in the 37.5-mg group, 9.1 minutes in the 75-mg group, 11 minutes in the 150-mg group, and 13 minutes in the 300-mg group. Mean changes in Epworth Sleepiness Scale scores were –3.3 for patients in the placebo group, –5.1 in the 37.5-mg group, –5 in the 75-mg group, –7.7 in the 150-mg group, and –7.9 in the 300-mg group.
A significantly higher rate of patients in the 75-mg (72.4%), 150-mg (89.7%), and 300-mg (88.7%) groups improved on the Patient Global Impression of Change scale, compared with patients in the placebo (49.1%) and 37.5-mg (55.4%) groups.
Just under 68% of patients who received JZP-110 experienced at least one adverse event, compared with 47.9% of patients who received placebo. The most common adverse events were headache, nausea, decreased appetite, and anxiety. Six serious adverse events were reported over the study period, but none was related to JZP-110.
The study is scheduled to be presented on Sunday, Oct. 29, from 1:30 p.m. to 1:45 p.m. in Room 601A of the Toronto Convention Centre South Building as part of the “Obstructive Sleep Apnea: Insights & Management” session, which will run from 1:30 p.m. to 3 p.m.
FROM CHEST 2017
Sepsis response team does not improve mortality/organ dysfunction
A sepsis response team did not have a positive effect on mortality or organ dysfunction in septic patients, compared with standard treatment by a primary care team, according to a study abstract scheduled to be presented at CHEST 2017.
Compared with the primary care team, the sepsis team was more likely to intervene on patients with a quick Sepsis-Related Organ Failure Assessment score greater than 1 (33.8% vs. 22.8%), change or initiate antibiotics within 3 hours (64.6% vs. 37.2%), and obtain blood cultures on time (66.4% vs 45.2%). An additional difference between the two groups was that the sepsis team had better compliance with the 3-hour bundle (15.2% vs 8.4%).
Despite the sepsis team’s higher level of compliance with certain protocols, the combined outcome measure of mortality and organ dysfunction within 28 days was not significantly higher for patients treated by the sepsis team (11.3% vs. 9.8%; P = .6). In fact, there was at least one downside to being treated by the sepsis team, which was having a 14% longer hospital stay.
Chhaya Patel, MD, is scheduled to present the abstract on Sun., Oct. 29th, at 2:30-2:45 p.m. in Convention Center – 602B. This research is part of the Sepsis & Septic Shock session at the CHEST annual meeting, which will run from 1:30 to 3:00 p.m.
A sepsis response team did not have a positive effect on mortality or organ dysfunction in septic patients, compared with standard treatment by a primary care team, according to a study abstract scheduled to be presented at CHEST 2017.
Compared with the primary care team, the sepsis team was more likely to intervene on patients with a quick Sepsis-Related Organ Failure Assessment score greater than 1 (33.8% vs. 22.8%), change or initiate antibiotics within 3 hours (64.6% vs. 37.2%), and obtain blood cultures on time (66.4% vs 45.2%). An additional difference between the two groups was that the sepsis team had better compliance with the 3-hour bundle (15.2% vs 8.4%).
Despite the sepsis team’s higher level of compliance with certain protocols, the combined outcome measure of mortality and organ dysfunction within 28 days was not significantly higher for patients treated by the sepsis team (11.3% vs. 9.8%; P = .6). In fact, there was at least one downside to being treated by the sepsis team, which was having a 14% longer hospital stay.
Chhaya Patel, MD, is scheduled to present the abstract on Sun., Oct. 29th, at 2:30-2:45 p.m. in Convention Center – 602B. This research is part of the Sepsis & Septic Shock session at the CHEST annual meeting, which will run from 1:30 to 3:00 p.m.
A sepsis response team did not have a positive effect on mortality or organ dysfunction in septic patients, compared with standard treatment by a primary care team, according to a study abstract scheduled to be presented at CHEST 2017.
Compared with the primary care team, the sepsis team was more likely to intervene on patients with a quick Sepsis-Related Organ Failure Assessment score greater than 1 (33.8% vs. 22.8%), change or initiate antibiotics within 3 hours (64.6% vs. 37.2%), and obtain blood cultures on time (66.4% vs 45.2%). An additional difference between the two groups was that the sepsis team had better compliance with the 3-hour bundle (15.2% vs 8.4%).
Despite the sepsis team’s higher level of compliance with certain protocols, the combined outcome measure of mortality and organ dysfunction within 28 days was not significantly higher for patients treated by the sepsis team (11.3% vs. 9.8%; P = .6). In fact, there was at least one downside to being treated by the sepsis team, which was having a 14% longer hospital stay.
Chhaya Patel, MD, is scheduled to present the abstract on Sun., Oct. 29th, at 2:30-2:45 p.m. in Convention Center – 602B. This research is part of the Sepsis & Septic Shock session at the CHEST annual meeting, which will run from 1:30 to 3:00 p.m.
FROM CHEST 2017
Robotic-assisted pulmonary lobectomy effective for large tumors
Robotic-assisted pulmonary lobectomy is a safe and effective way to remove large tumors in patients with non–small cell lung cancer (NSCLC), according to the abstract of a study scheduled to be presented at the CHEST annual meeting.
The study covers a retrospective analysis of 345 NSCLC patients with tumors who underwent robotic-assisted pulmonary lobectomy performed by one surgeon from September 2010 through August 2016. The participants were grouped into the following three cohorts: patients with tumors less than 5 cm in diameter, patients with tumors from 5 to 7 cm, and patients with tumors larger than 7 cm. The researchers excluded patients with pulmonary metastases or benign lesions from the study.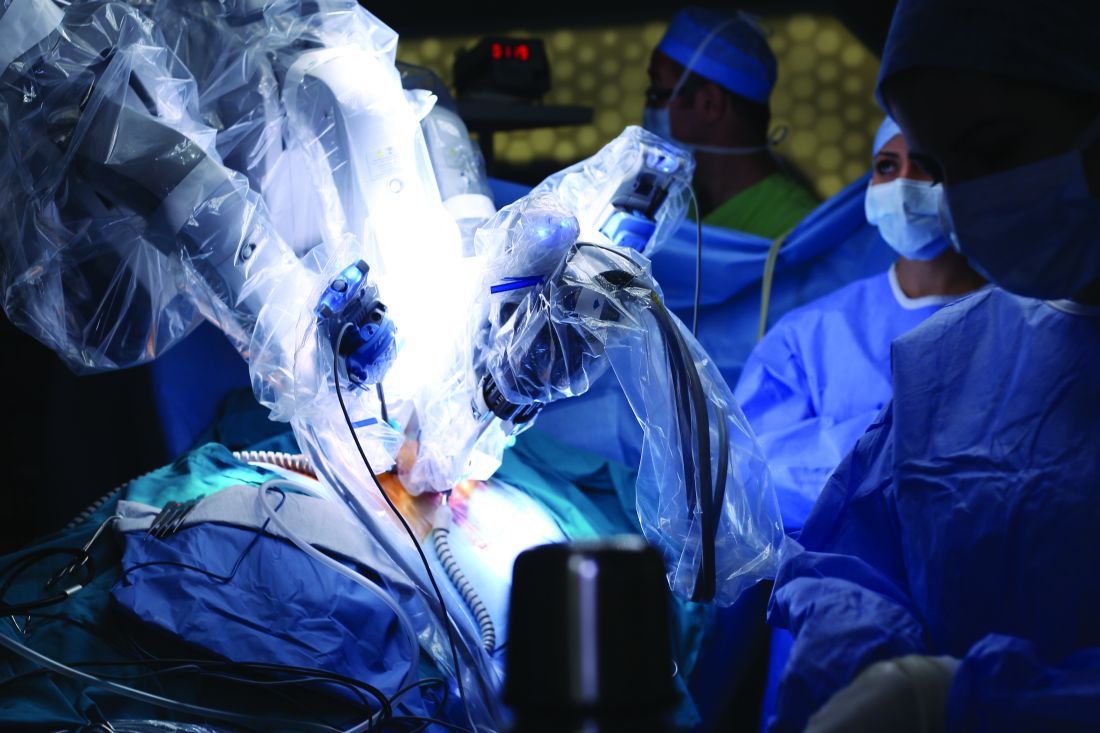
Patients with smaller tumors were more likely to have simple lobectomy or lobectomy plus wedge, while patients with larger tumors were more likely to require lobectomy with chest wall resection. Increased tumor size was also associated with increased intraoperative estimated blood loss, skin-to-skin operative time, hospital length of stay, and overall conversion to open lobectomy.
There was no association found between tumor size and increased overall intraoperative or postoperative complications, or in-hospital mortality.
Nirav Patel, MD, FCCP, of the Tampa Bay Sleep Center is scheduled to present his abstract on Sunday Oct. 29th, between 2:15 and 2:30 p.m. in Convention Center – 606. Dr. Patel’s research is part of the Cardiothoracic Surgery session, running from 1:30 p.m. to 3:00 p.m. at the CHEST annual meeting.
Robotic-assisted pulmonary lobectomy is a safe and effective way to remove large tumors in patients with non–small cell lung cancer (NSCLC), according to the abstract of a study scheduled to be presented at the CHEST annual meeting.
The study covers a retrospective analysis of 345 NSCLC patients with tumors who underwent robotic-assisted pulmonary lobectomy performed by one surgeon from September 2010 through August 2016. The participants were grouped into the following three cohorts: patients with tumors less than 5 cm in diameter, patients with tumors from 5 to 7 cm, and patients with tumors larger than 7 cm. The researchers excluded patients with pulmonary metastases or benign lesions from the study.
Patients with smaller tumors were more likely to have simple lobectomy or lobectomy plus wedge, while patients with larger tumors were more likely to require lobectomy with chest wall resection. Increased tumor size was also associated with increased intraoperative estimated blood loss, skin-to-skin operative time, hospital length of stay, and overall conversion to open lobectomy.
There was no association found between tumor size and increased overall intraoperative or postoperative complications, or in-hospital mortality.
Nirav Patel, MD, FCCP, of the Tampa Bay Sleep Center is scheduled to present his abstract on Sunday Oct. 29th, between 2:15 and 2:30 p.m. in Convention Center – 606. Dr. Patel’s research is part of the Cardiothoracic Surgery session, running from 1:30 p.m. to 3:00 p.m. at the CHEST annual meeting.
Robotic-assisted pulmonary lobectomy is a safe and effective way to remove large tumors in patients with non–small cell lung cancer (NSCLC), according to the abstract of a study scheduled to be presented at the CHEST annual meeting.
The study covers a retrospective analysis of 345 NSCLC patients with tumors who underwent robotic-assisted pulmonary lobectomy performed by one surgeon from September 2010 through August 2016. The participants were grouped into the following three cohorts: patients with tumors less than 5 cm in diameter, patients with tumors from 5 to 7 cm, and patients with tumors larger than 7 cm. The researchers excluded patients with pulmonary metastases or benign lesions from the study.
Patients with smaller tumors were more likely to have simple lobectomy or lobectomy plus wedge, while patients with larger tumors were more likely to require lobectomy with chest wall resection. Increased tumor size was also associated with increased intraoperative estimated blood loss, skin-to-skin operative time, hospital length of stay, and overall conversion to open lobectomy.
There was no association found between tumor size and increased overall intraoperative or postoperative complications, or in-hospital mortality.
Nirav Patel, MD, FCCP, of the Tampa Bay Sleep Center is scheduled to present his abstract on Sunday Oct. 29th, between 2:15 and 2:30 p.m. in Convention Center – 606. Dr. Patel’s research is part of the Cardiothoracic Surgery session, running from 1:30 p.m. to 3:00 p.m. at the CHEST annual meeting.
FROM CHEST 2017
Near-fatal asthma treated effectively by ECMO
Extracorporeal membrane oxygenation (ECMO) is an effective way to treat near fatal asthma, but physicians must remember the risk of complications, according to an abstract on a study scheduled to be presented at CHEST 2017.
The study covers a retrospective analysis of 371 children with asthma who were treated with ECMO; it used data collected by the Extracorporeal Life Support Organization registry from 1988 to 2016. The median age of the children in the study was 7.5 years; the participant group was 43% white and 39% black, as well as 56% male, according to the abstract, which is mentioned in the program for the CHEST annual meeting.
About 80% of children experienced at least one complication, with 20% experiencing three or more. Children who had three or more complications were significantly less likely to experience lung recovery.
Of the children who received VV cannulation, 90% experienced lung recovery, whereas only 69% of children who received VA cannulation recovered. (P less than .0001). VA cannulation was also associated with a higher risk of neurological complications, while those who received VV cannulation were significantly more likely to survive.
The abstract is scheduled to be presented on Sunday Oct. 29 from 2:30 p.m. to 2:45 p.m. in Room 603 of Toronto Convention Centre South Building as part of the Acute Lung Injury & Respiratory Failure session, which will run from 1:30 p.m. to 3 p.m.
Extracorporeal membrane oxygenation (ECMO) is an effective way to treat near fatal asthma, but physicians must remember the risk of complications, according to an abstract on a study scheduled to be presented at CHEST 2017.
The study covers a retrospective analysis of 371 children with asthma who were treated with ECMO; it used data collected by the Extracorporeal Life Support Organization registry from 1988 to 2016. The median age of the children in the study was 7.5 years; the participant group was 43% white and 39% black, as well as 56% male, according to the abstract, which is mentioned in the program for the CHEST annual meeting.
About 80% of children experienced at least one complication, with 20% experiencing three or more. Children who had three or more complications were significantly less likely to experience lung recovery.
Of the children who received VV cannulation, 90% experienced lung recovery, whereas only 69% of children who received VA cannulation recovered. (P less than .0001). VA cannulation was also associated with a higher risk of neurological complications, while those who received VV cannulation were significantly more likely to survive.
The abstract is scheduled to be presented on Sunday Oct. 29 from 2:30 p.m. to 2:45 p.m. in Room 603 of Toronto Convention Centre South Building as part of the Acute Lung Injury & Respiratory Failure session, which will run from 1:30 p.m. to 3 p.m.
Extracorporeal membrane oxygenation (ECMO) is an effective way to treat near fatal asthma, but physicians must remember the risk of complications, according to an abstract on a study scheduled to be presented at CHEST 2017.
The study covers a retrospective analysis of 371 children with asthma who were treated with ECMO; it used data collected by the Extracorporeal Life Support Organization registry from 1988 to 2016. The median age of the children in the study was 7.5 years; the participant group was 43% white and 39% black, as well as 56% male, according to the abstract, which is mentioned in the program for the CHEST annual meeting.
About 80% of children experienced at least one complication, with 20% experiencing three or more. Children who had three or more complications were significantly less likely to experience lung recovery.
Of the children who received VV cannulation, 90% experienced lung recovery, whereas only 69% of children who received VA cannulation recovered. (P less than .0001). VA cannulation was also associated with a higher risk of neurological complications, while those who received VV cannulation were significantly more likely to survive.
The abstract is scheduled to be presented on Sunday Oct. 29 from 2:30 p.m. to 2:45 p.m. in Room 603 of Toronto Convention Centre South Building as part of the Acute Lung Injury & Respiratory Failure session, which will run from 1:30 p.m. to 3 p.m.
Bilirubin levels associated with transplant-free survival in PBC patients
Normal serum bilirubin concentrations in patients with primary biliary cholangitis (PBC) were associated with improved odds of transplant-free survival, according to a study presented at the annual meeting of the American Association for the Study of Liver Diseases.
In a retrospective analysis of data from the Global PBC Study group, PBC patients who had bilirubin levels between normal and the upper limit of normal at baseline (n = 2,795), at 1 year (n = 3,082), at 3 years (n = 1,657), or at 5 years (n = 1,339) were included in the study. Both ursodeoxycholic acid–treated and untreated patients were included, according to Carla Murillo Perez of Toronto General Hospital and her associates.
Each cohort was organized into quartiles, with Q1 having the lowest bilirubin levels and Q4 having the highest. In the baseline cohort, 5-year transplant-free survival rates were 97% in Q1, 95% in Q2, 96% in Q3, and 91% in Q4; similarly improved odds for transplant-free survival in lower quartiles were seen in the later cohorts.
Higher bilirubin (per 0.1 × upper limit of normal increase) was associated with an increased chance for death or transplantation, with hazard ratios of 1.14 in the baseline cohort, 1.21 in the 1-year cohort, 1.19 in the 3-year cohort, and 1.17 in the 5-year cohort, Ms. Perez and her associates said.
Dr. Cyriel Ponsioen, Dr. Christophe Corpechot, Dr. Marlyn Mayo, Dr. Annarosa Floreani, Dr. Albert Pares, Dr. Frederik Nevens, Dr. Kris Kowdley, Dr. Tony Bruns, Dr. Gideon Hirschfield, Dr. Keith Lindor, and Dr. Harry Janssen reported conflicts of interest.
Normal serum bilirubin concentrations in patients with primary biliary cholangitis (PBC) were associated with improved odds of transplant-free survival, according to a study presented at the annual meeting of the American Association for the Study of Liver Diseases.
In a retrospective analysis of data from the Global PBC Study group, PBC patients who had bilirubin levels between normal and the upper limit of normal at baseline (n = 2,795), at 1 year (n = 3,082), at 3 years (n = 1,657), or at 5 years (n = 1,339) were included in the study. Both ursodeoxycholic acid–treated and untreated patients were included, according to Carla Murillo Perez of Toronto General Hospital and her associates.
Each cohort was organized into quartiles, with Q1 having the lowest bilirubin levels and Q4 having the highest. In the baseline cohort, 5-year transplant-free survival rates were 97% in Q1, 95% in Q2, 96% in Q3, and 91% in Q4; similarly improved odds for transplant-free survival in lower quartiles were seen in the later cohorts.
Higher bilirubin (per 0.1 × upper limit of normal increase) was associated with an increased chance for death or transplantation, with hazard ratios of 1.14 in the baseline cohort, 1.21 in the 1-year cohort, 1.19 in the 3-year cohort, and 1.17 in the 5-year cohort, Ms. Perez and her associates said.
Dr. Cyriel Ponsioen, Dr. Christophe Corpechot, Dr. Marlyn Mayo, Dr. Annarosa Floreani, Dr. Albert Pares, Dr. Frederik Nevens, Dr. Kris Kowdley, Dr. Tony Bruns, Dr. Gideon Hirschfield, Dr. Keith Lindor, and Dr. Harry Janssen reported conflicts of interest.
Normal serum bilirubin concentrations in patients with primary biliary cholangitis (PBC) were associated with improved odds of transplant-free survival, according to a study presented at the annual meeting of the American Association for the Study of Liver Diseases.
In a retrospective analysis of data from the Global PBC Study group, PBC patients who had bilirubin levels between normal and the upper limit of normal at baseline (n = 2,795), at 1 year (n = 3,082), at 3 years (n = 1,657), or at 5 years (n = 1,339) were included in the study. Both ursodeoxycholic acid–treated and untreated patients were included, according to Carla Murillo Perez of Toronto General Hospital and her associates.
Each cohort was organized into quartiles, with Q1 having the lowest bilirubin levels and Q4 having the highest. In the baseline cohort, 5-year transplant-free survival rates were 97% in Q1, 95% in Q2, 96% in Q3, and 91% in Q4; similarly improved odds for transplant-free survival in lower quartiles were seen in the later cohorts.
Higher bilirubin (per 0.1 × upper limit of normal increase) was associated with an increased chance for death or transplantation, with hazard ratios of 1.14 in the baseline cohort, 1.21 in the 1-year cohort, 1.19 in the 3-year cohort, and 1.17 in the 5-year cohort, Ms. Perez and her associates said.
Dr. Cyriel Ponsioen, Dr. Christophe Corpechot, Dr. Marlyn Mayo, Dr. Annarosa Floreani, Dr. Albert Pares, Dr. Frederik Nevens, Dr. Kris Kowdley, Dr. Tony Bruns, Dr. Gideon Hirschfield, Dr. Keith Lindor, and Dr. Harry Janssen reported conflicts of interest.
FROM THE LIVER MEETING 2017
Key clinical point:
Major finding: In a baseline cohort, 5-year transplant-free survival rates were 97% in patients with the lowest bilirubin levels and 91% in patients with the highest.
Data source: A retrospective analysis of data from the Global PBC Study group database.
Disclosures: Dr. Cyriel Ponsioen, Dr. Christophe Corpechot, Dr. Marlyn Mayo, Dr. Annarosa Floreani, Dr. Albert Pares, Dr. Frederik Nevens, Dr. Kris Kowdley, Dr. Tony Bruns, Dr. Gideon Hirschfield, Dr. Keith Lindor, and Dr. Harry Janssen reported conflicts of interest.