User login
Radiotherapy planning scans reveal breast cancer patients’ CVD risk
Radiotherapy planning scans may be a rich untapped source of information for estimating the risk of cardiovascular disease (CVD) in breast cancer patients, a large study suggests.
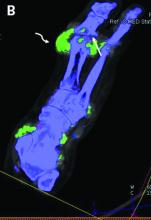
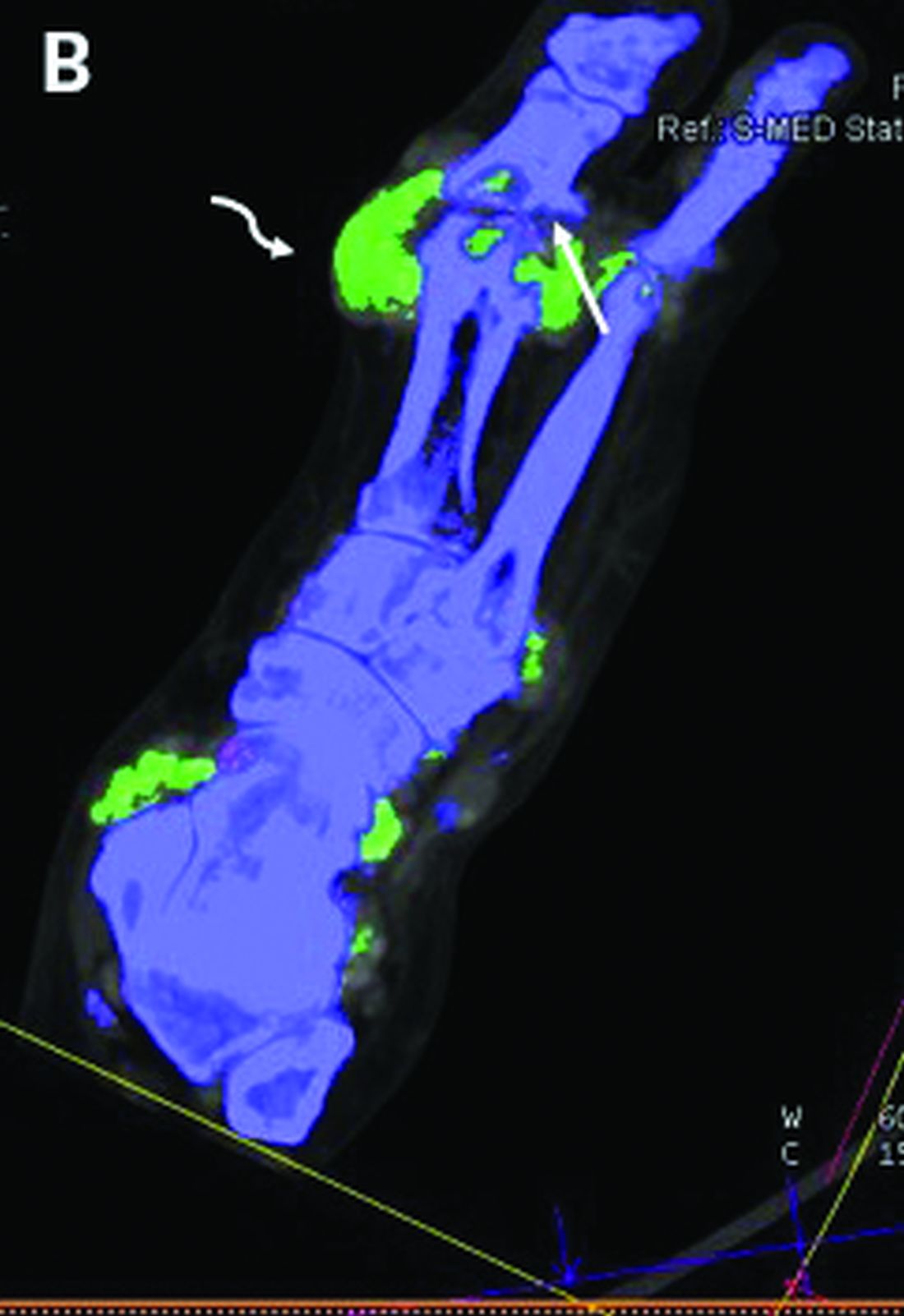
Researchers found that breast cancer patients with a coronary artery calcifications (CAC) score exceeding 400 had nearly four times the adjusted risk of fatal and nonfatal CVD events when compared with patients who had a CAC score of 0.
Patients with scores exceeding 400 also had more than eight times the risk of coronary heart disease events. The associations were especially strong in the subset of patients who received anthracycline-containing chemotherapy.
Helena Verkooijen, MD, PhD, of University Medical Center Utrecht (the Netherlands) presented these findings at the 12th European Breast Cancer Conference.
Dr. Verkooijen noted that, over the past 50 years, breast cancer has dramatically declined as a cause of death among breast cancer survivors, while CVD has continued to account for about 20% of the total deaths in this population.
CACs are sometimes incidentally seen in radiotherapy planning CT scans. “Right now, this information is not often used for patient stratification or informing patients about their cardiovascular risk, and this is a pity, because we know that it is an independent risk factor, and, often, the presence of calcifications can occur in the absence of other cardiovascular risk factors,” Dr. Verkooijen said.
Study details
Dr. Verkooijen and and colleagues from the Bragataston Study Group retrospectively studied 15,919 breast cancer patients who had radiotherapy planning CT scans during 2004-2016 at three Dutch institutions.
The researchers used an automated deep-learning algorithm (described in Radiology) to detect and quantify coronary calcium in planning CT scans and calculate CAC scores, classifying them into five categories.
The median follow-up was 51.6 months. Most women (70%) did not have any calcium detected in their coronary arteries (CAC score of 0), while 3% fell into the highest category (CAC score of >400).
The incidence of nonfatal and fatal CVD events increased with CAC score:
- 5.1% with a score of 0.
- 8.5% with a score of 1-10.
- 13.5% with a score of 11-100.
- 17.6% with a score of 101-400.
- 28.0% with a score greater than 400.
In analyses adjusted for age, laterality of radiation, and receipt of cardiotoxic agents – anthracyclines and trastuzumab – women with a score exceeding 400 had sharply elevated adjusted risks of CVD events (hazard ratio, 3.7), of coronary heart disease events specifically (HR, 8.2), and of death from any cause (HR, 2.8), when compared with peers who had a CAC score of 0.
On further scrutiny of CVD events, the pattern was similar regardless of whether radiation was left- or right-sided. However, the association was stronger among women who received anthracyclines as compared with counterparts who did not, with a nearly six-fold higher risk for those with highest versus lowest CAC scores.
When the women were surveyed, nearly 90% said they wanted to be informed about their CAC score and associated CVD risk, even in the absence of evidence-based risk reduction strategies.
Applying the results
“We believe that this is the first time that anyone has conducted a study on this topic on a scale like this, and we show that it is possible to relatively easily identify women at a very high risk of CVD,” Dr. Verkooijen said. “But what do we do with this information, because these scans are not made to answer this question. … This is information that we get that we haven’t really requested. I think we should only use this information when we have really shown that we can help patients reduce their risk of cardiovascular disease.”
To that end, Dr. Verkooijen and colleagues are planning additional research that will look at the potential benefit of referring high-risk patients for cardioprevention strategies and at the role of using the CAC score to personalize treatment strategies.
“This is an interesting and novel approach to predicting cardiac events for patients undergoing breast cancer treatment,” Meena S. Moran, MD, of Yale University in New Haven, Conn., commented in an interview.
The approach would likely be feasible in typical practice with widespread availability of the automated algorithm and might even alter treatment planning in real time, she said. “From the standpoint of radiation oncology, it would mean running the software to generate a CAC score, which would allow for modifications in decision-making during treatment planning, such as whether or not to include the internal mammary nodal chain in a patient who may be in the ‘gray zone’ for regional nodal radiation. For example, if a patient has a high CAC score, plus if they have received (or are receiving) cardiotoxic drugs, radiation oncologists can use that information as an additional factor to consider in the decision-making of whether or not to include the internal mammary chain, which inevitably can increase the dose delivered to the heart,” Dr. Moran elaborated.
Dr. Verkooijen’s study was supported by the Dutch Cancer Society, the European Commission, the Dutch Digestive Foundation, the Netherlands Organisation for Scientific Research, and Elekta. Dr. Verkooijen and Dr. Moran disclosed no conflicts of interest.
SOURCE: Gal R et al. EBCC-12 Virtual Congress, Abstract 7.
Radiotherapy planning scans may be a rich untapped source of information for estimating the risk of cardiovascular disease (CVD) in breast cancer patients, a large study suggests.
Researchers found that breast cancer patients with a coronary artery calcifications (CAC) score exceeding 400 had nearly four times the adjusted risk of fatal and nonfatal CVD events when compared with patients who had a CAC score of 0.
Patients with scores exceeding 400 also had more than eight times the risk of coronary heart disease events. The associations were especially strong in the subset of patients who received anthracycline-containing chemotherapy.
Helena Verkooijen, MD, PhD, of University Medical Center Utrecht (the Netherlands) presented these findings at the 12th European Breast Cancer Conference.
Dr. Verkooijen noted that, over the past 50 years, breast cancer has dramatically declined as a cause of death among breast cancer survivors, while CVD has continued to account for about 20% of the total deaths in this population.
CACs are sometimes incidentally seen in radiotherapy planning CT scans. “Right now, this information is not often used for patient stratification or informing patients about their cardiovascular risk, and this is a pity, because we know that it is an independent risk factor, and, often, the presence of calcifications can occur in the absence of other cardiovascular risk factors,” Dr. Verkooijen said.
Study details
Dr. Verkooijen and and colleagues from the Bragataston Study Group retrospectively studied 15,919 breast cancer patients who had radiotherapy planning CT scans during 2004-2016 at three Dutch institutions.
The researchers used an automated deep-learning algorithm (described in Radiology) to detect and quantify coronary calcium in planning CT scans and calculate CAC scores, classifying them into five categories.
The median follow-up was 51.6 months. Most women (70%) did not have any calcium detected in their coronary arteries (CAC score of 0), while 3% fell into the highest category (CAC score of >400).
The incidence of nonfatal and fatal CVD events increased with CAC score:
- 5.1% with a score of 0.
- 8.5% with a score of 1-10.
- 13.5% with a score of 11-100.
- 17.6% with a score of 101-400.
- 28.0% with a score greater than 400.
In analyses adjusted for age, laterality of radiation, and receipt of cardiotoxic agents – anthracyclines and trastuzumab – women with a score exceeding 400 had sharply elevated adjusted risks of CVD events (hazard ratio, 3.7), of coronary heart disease events specifically (HR, 8.2), and of death from any cause (HR, 2.8), when compared with peers who had a CAC score of 0.
On further scrutiny of CVD events, the pattern was similar regardless of whether radiation was left- or right-sided. However, the association was stronger among women who received anthracyclines as compared with counterparts who did not, with a nearly six-fold higher risk for those with highest versus lowest CAC scores.
When the women were surveyed, nearly 90% said they wanted to be informed about their CAC score and associated CVD risk, even in the absence of evidence-based risk reduction strategies.
Applying the results
“We believe that this is the first time that anyone has conducted a study on this topic on a scale like this, and we show that it is possible to relatively easily identify women at a very high risk of CVD,” Dr. Verkooijen said. “But what do we do with this information, because these scans are not made to answer this question. … This is information that we get that we haven’t really requested. I think we should only use this information when we have really shown that we can help patients reduce their risk of cardiovascular disease.”
To that end, Dr. Verkooijen and colleagues are planning additional research that will look at the potential benefit of referring high-risk patients for cardioprevention strategies and at the role of using the CAC score to personalize treatment strategies.
“This is an interesting and novel approach to predicting cardiac events for patients undergoing breast cancer treatment,” Meena S. Moran, MD, of Yale University in New Haven, Conn., commented in an interview.
The approach would likely be feasible in typical practice with widespread availability of the automated algorithm and might even alter treatment planning in real time, she said. “From the standpoint of radiation oncology, it would mean running the software to generate a CAC score, which would allow for modifications in decision-making during treatment planning, such as whether or not to include the internal mammary nodal chain in a patient who may be in the ‘gray zone’ for regional nodal radiation. For example, if a patient has a high CAC score, plus if they have received (or are receiving) cardiotoxic drugs, radiation oncologists can use that information as an additional factor to consider in the decision-making of whether or not to include the internal mammary chain, which inevitably can increase the dose delivered to the heart,” Dr. Moran elaborated.
Dr. Verkooijen’s study was supported by the Dutch Cancer Society, the European Commission, the Dutch Digestive Foundation, the Netherlands Organisation for Scientific Research, and Elekta. Dr. Verkooijen and Dr. Moran disclosed no conflicts of interest.
SOURCE: Gal R et al. EBCC-12 Virtual Congress, Abstract 7.
Radiotherapy planning scans may be a rich untapped source of information for estimating the risk of cardiovascular disease (CVD) in breast cancer patients, a large study suggests.
Researchers found that breast cancer patients with a coronary artery calcifications (CAC) score exceeding 400 had nearly four times the adjusted risk of fatal and nonfatal CVD events when compared with patients who had a CAC score of 0.
Patients with scores exceeding 400 also had more than eight times the risk of coronary heart disease events. The associations were especially strong in the subset of patients who received anthracycline-containing chemotherapy.
Helena Verkooijen, MD, PhD, of University Medical Center Utrecht (the Netherlands) presented these findings at the 12th European Breast Cancer Conference.
Dr. Verkooijen noted that, over the past 50 years, breast cancer has dramatically declined as a cause of death among breast cancer survivors, while CVD has continued to account for about 20% of the total deaths in this population.
CACs are sometimes incidentally seen in radiotherapy planning CT scans. “Right now, this information is not often used for patient stratification or informing patients about their cardiovascular risk, and this is a pity, because we know that it is an independent risk factor, and, often, the presence of calcifications can occur in the absence of other cardiovascular risk factors,” Dr. Verkooijen said.
Study details
Dr. Verkooijen and and colleagues from the Bragataston Study Group retrospectively studied 15,919 breast cancer patients who had radiotherapy planning CT scans during 2004-2016 at three Dutch institutions.
The researchers used an automated deep-learning algorithm (described in Radiology) to detect and quantify coronary calcium in planning CT scans and calculate CAC scores, classifying them into five categories.
The median follow-up was 51.6 months. Most women (70%) did not have any calcium detected in their coronary arteries (CAC score of 0), while 3% fell into the highest category (CAC score of >400).
The incidence of nonfatal and fatal CVD events increased with CAC score:
- 5.1% with a score of 0.
- 8.5% with a score of 1-10.
- 13.5% with a score of 11-100.
- 17.6% with a score of 101-400.
- 28.0% with a score greater than 400.
In analyses adjusted for age, laterality of radiation, and receipt of cardiotoxic agents – anthracyclines and trastuzumab – women with a score exceeding 400 had sharply elevated adjusted risks of CVD events (hazard ratio, 3.7), of coronary heart disease events specifically (HR, 8.2), and of death from any cause (HR, 2.8), when compared with peers who had a CAC score of 0.
On further scrutiny of CVD events, the pattern was similar regardless of whether radiation was left- or right-sided. However, the association was stronger among women who received anthracyclines as compared with counterparts who did not, with a nearly six-fold higher risk for those with highest versus lowest CAC scores.
When the women were surveyed, nearly 90% said they wanted to be informed about their CAC score and associated CVD risk, even in the absence of evidence-based risk reduction strategies.
Applying the results
“We believe that this is the first time that anyone has conducted a study on this topic on a scale like this, and we show that it is possible to relatively easily identify women at a very high risk of CVD,” Dr. Verkooijen said. “But what do we do with this information, because these scans are not made to answer this question. … This is information that we get that we haven’t really requested. I think we should only use this information when we have really shown that we can help patients reduce their risk of cardiovascular disease.”
To that end, Dr. Verkooijen and colleagues are planning additional research that will look at the potential benefit of referring high-risk patients for cardioprevention strategies and at the role of using the CAC score to personalize treatment strategies.
“This is an interesting and novel approach to predicting cardiac events for patients undergoing breast cancer treatment,” Meena S. Moran, MD, of Yale University in New Haven, Conn., commented in an interview.
The approach would likely be feasible in typical practice with widespread availability of the automated algorithm and might even alter treatment planning in real time, she said. “From the standpoint of radiation oncology, it would mean running the software to generate a CAC score, which would allow for modifications in decision-making during treatment planning, such as whether or not to include the internal mammary nodal chain in a patient who may be in the ‘gray zone’ for regional nodal radiation. For example, if a patient has a high CAC score, plus if they have received (or are receiving) cardiotoxic drugs, radiation oncologists can use that information as an additional factor to consider in the decision-making of whether or not to include the internal mammary chain, which inevitably can increase the dose delivered to the heart,” Dr. Moran elaborated.
Dr. Verkooijen’s study was supported by the Dutch Cancer Society, the European Commission, the Dutch Digestive Foundation, the Netherlands Organisation for Scientific Research, and Elekta. Dr. Verkooijen and Dr. Moran disclosed no conflicts of interest.
SOURCE: Gal R et al. EBCC-12 Virtual Congress, Abstract 7.
FROM EBCC-12 VIRTUAL CONFERENCE
Gene signature may improve prognostication in ovarian cancer
according to a study published in Annals of Oncology.
“Gene expression signature tests for prognosis are available for other cancers, such as breast cancer, and these help with treatment decisions, but no such tests are available for ovarian cancer,” senior investigator Susan J. Ramus, PhD, of Lowy Cancer Research Centre, University of NSW Sydney, commented in an interview.
Dr. Ramus and associates developed and validated their 101-gene expression signature using pretreatment tumor tissue from 3,769 women with high-grade serous ovarian cancer treated on 21 studies.
The investigators found this signature, called OTTA-SPOT (Ovarian Tumor Tissue Analysis Consortium–Stratified Prognosis of Ovarian Tumors), performed well at stratifying women according to overall survival. Median overall survival times ranged from about 2 years for patients in the top quintile of scores to more than 9 years for patients in the bottom quintile.
Moreover, OTTA-SPOT significantly improved prognostication when added to age and stage.
“This tumor test works on formalin-fixed, paraffin-embedded tumors, as collected routinely in clinical practice,” Dr. Ramus noted. “Women predicted to have poor survival using current treatments could be included in clinical trials to rapidly get alternative treatment. Many of the genes included in this test are targets of known drugs, so this information could lead to alternative targeted treatments.
“This test is not ready for routine clinical care yet,” she added. “The next step would be to include this signature as part of a clinical trial. If patients predicted to have poor survival are given alternative treatments that improve their survival, then the test could be included in treatment decisions.”
Study details
Dr. Ramus and colleagues began this work by measuring tumor expression of 513 genes selected via meta-analysis. The team then developed a gene expression assay and a prognostic signature for overall survival, which they trained on tumors from 2,702 women in 15 studies and validated on an independent set of tumors from 1,067 women in 6 studies.
In analyses adjusted for covariates, expression levels of 276 genes were associated with overall survival. The signature with the best prognostic performance contained 101 genes that were enriched in pathways having treatment implications, such as pathways involved in immune response, mitosis, and homologous recombination repair.
Adding the signature to age and stage alone improved prediction of 2- and 5-year overall survival. The area under the curve increased from 0.61 to 0.69 for 2-year overall survival and from 0.62 to 0.75 for 5-year overall survival (with nonoverlapping 95% confidence intervals for 5-year survival).
Each standard deviation increase in the gene expression score was associated with a more than doubling of the risk of death (hazard ratio, 2.35; P < .001).
The median overall survival by gene expression score quintile was 9.5 years for patients in the first quintile, 5.4 years for patients in the second, 3.8 years for patients in the third, 3.2 years for patients in the fourth, and 2.3 years for patients in the fifth.
This study was funded by the National Institutes of Health/National Cancer Institute, the Canadian Institutes for Health Research, and the Department of Defense Ovarian Cancer Research Program. Some of the authors disclosed financial relationships with a range of companies. Dr. Ramus disclosed no conflicts of interest.
SOURCE: Millstein J et al. Ann Oncol. 2020 Sep;31(9):1240-50.
according to a study published in Annals of Oncology.
“Gene expression signature tests for prognosis are available for other cancers, such as breast cancer, and these help with treatment decisions, but no such tests are available for ovarian cancer,” senior investigator Susan J. Ramus, PhD, of Lowy Cancer Research Centre, University of NSW Sydney, commented in an interview.
Dr. Ramus and associates developed and validated their 101-gene expression signature using pretreatment tumor tissue from 3,769 women with high-grade serous ovarian cancer treated on 21 studies.
The investigators found this signature, called OTTA-SPOT (Ovarian Tumor Tissue Analysis Consortium–Stratified Prognosis of Ovarian Tumors), performed well at stratifying women according to overall survival. Median overall survival times ranged from about 2 years for patients in the top quintile of scores to more than 9 years for patients in the bottom quintile.
Moreover, OTTA-SPOT significantly improved prognostication when added to age and stage.
“This tumor test works on formalin-fixed, paraffin-embedded tumors, as collected routinely in clinical practice,” Dr. Ramus noted. “Women predicted to have poor survival using current treatments could be included in clinical trials to rapidly get alternative treatment. Many of the genes included in this test are targets of known drugs, so this information could lead to alternative targeted treatments.
“This test is not ready for routine clinical care yet,” she added. “The next step would be to include this signature as part of a clinical trial. If patients predicted to have poor survival are given alternative treatments that improve their survival, then the test could be included in treatment decisions.”
Study details
Dr. Ramus and colleagues began this work by measuring tumor expression of 513 genes selected via meta-analysis. The team then developed a gene expression assay and a prognostic signature for overall survival, which they trained on tumors from 2,702 women in 15 studies and validated on an independent set of tumors from 1,067 women in 6 studies.
In analyses adjusted for covariates, expression levels of 276 genes were associated with overall survival. The signature with the best prognostic performance contained 101 genes that were enriched in pathways having treatment implications, such as pathways involved in immune response, mitosis, and homologous recombination repair.
Adding the signature to age and stage alone improved prediction of 2- and 5-year overall survival. The area under the curve increased from 0.61 to 0.69 for 2-year overall survival and from 0.62 to 0.75 for 5-year overall survival (with nonoverlapping 95% confidence intervals for 5-year survival).
Each standard deviation increase in the gene expression score was associated with a more than doubling of the risk of death (hazard ratio, 2.35; P < .001).
The median overall survival by gene expression score quintile was 9.5 years for patients in the first quintile, 5.4 years for patients in the second, 3.8 years for patients in the third, 3.2 years for patients in the fourth, and 2.3 years for patients in the fifth.
This study was funded by the National Institutes of Health/National Cancer Institute, the Canadian Institutes for Health Research, and the Department of Defense Ovarian Cancer Research Program. Some of the authors disclosed financial relationships with a range of companies. Dr. Ramus disclosed no conflicts of interest.
SOURCE: Millstein J et al. Ann Oncol. 2020 Sep;31(9):1240-50.
according to a study published in Annals of Oncology.
“Gene expression signature tests for prognosis are available for other cancers, such as breast cancer, and these help with treatment decisions, but no such tests are available for ovarian cancer,” senior investigator Susan J. Ramus, PhD, of Lowy Cancer Research Centre, University of NSW Sydney, commented in an interview.
Dr. Ramus and associates developed and validated their 101-gene expression signature using pretreatment tumor tissue from 3,769 women with high-grade serous ovarian cancer treated on 21 studies.
The investigators found this signature, called OTTA-SPOT (Ovarian Tumor Tissue Analysis Consortium–Stratified Prognosis of Ovarian Tumors), performed well at stratifying women according to overall survival. Median overall survival times ranged from about 2 years for patients in the top quintile of scores to more than 9 years for patients in the bottom quintile.
Moreover, OTTA-SPOT significantly improved prognostication when added to age and stage.
“This tumor test works on formalin-fixed, paraffin-embedded tumors, as collected routinely in clinical practice,” Dr. Ramus noted. “Women predicted to have poor survival using current treatments could be included in clinical trials to rapidly get alternative treatment. Many of the genes included in this test are targets of known drugs, so this information could lead to alternative targeted treatments.
“This test is not ready for routine clinical care yet,” she added. “The next step would be to include this signature as part of a clinical trial. If patients predicted to have poor survival are given alternative treatments that improve their survival, then the test could be included in treatment decisions.”
Study details
Dr. Ramus and colleagues began this work by measuring tumor expression of 513 genes selected via meta-analysis. The team then developed a gene expression assay and a prognostic signature for overall survival, which they trained on tumors from 2,702 women in 15 studies and validated on an independent set of tumors from 1,067 women in 6 studies.
In analyses adjusted for covariates, expression levels of 276 genes were associated with overall survival. The signature with the best prognostic performance contained 101 genes that were enriched in pathways having treatment implications, such as pathways involved in immune response, mitosis, and homologous recombination repair.
Adding the signature to age and stage alone improved prediction of 2- and 5-year overall survival. The area under the curve increased from 0.61 to 0.69 for 2-year overall survival and from 0.62 to 0.75 for 5-year overall survival (with nonoverlapping 95% confidence intervals for 5-year survival).
Each standard deviation increase in the gene expression score was associated with a more than doubling of the risk of death (hazard ratio, 2.35; P < .001).
The median overall survival by gene expression score quintile was 9.5 years for patients in the first quintile, 5.4 years for patients in the second, 3.8 years for patients in the third, 3.2 years for patients in the fourth, and 2.3 years for patients in the fifth.
This study was funded by the National Institutes of Health/National Cancer Institute, the Canadian Institutes for Health Research, and the Department of Defense Ovarian Cancer Research Program. Some of the authors disclosed financial relationships with a range of companies. Dr. Ramus disclosed no conflicts of interest.
SOURCE: Millstein J et al. Ann Oncol. 2020 Sep;31(9):1240-50.
FROM ANNALS OF ONCOLOGY
Delaying RT for higher-risk prostate cancer found safe
A study of more than 60,000 prostate cancer patients suggests it is safe to delay radiation therapy (RT) for at least 6 months for localized higher-risk disease being treated with androgen deprivation therapy.
These findings are relevant to oncology care in the COVID-19 era, as the pandemic has complicated delivery of radiation therapy (RT) in several ways, the study authors wrote in JAMA Oncology.
“Daily hospital trips for RT create many possible points of COVID-19 transmission, and patients with cancer are at high risk of COVID-19 mortality,” Edward Christopher Dee, a research fellow at Dana-Farber Cancer Institute in Boston, and colleagues wrote.
To assess the safety of delaying RT, the investigators analyzed National Cancer Database data for 63,858 men with localized but unfavorable intermediate-risk, high-risk, or very-high-risk prostate cancer diagnosed during 2004-2014 and managed with external beam RT and androgen deprivation therapy (ADT).
Only 5.6% of patients (n = 3,572) initiated their RT 0-60 days before starting ADT. Another 36.3% (n = 23,207) initiated RT 1-60 days after starting ADT, 47.4% (n = 30,285) initiated RT 61-120 days after starting ADT, and 10.6% (n = 6,794) initiated RT 121-180 days after starting ADT.
The investigators found that 10-year overall survival rates were similar regardless of when patients started RT.
Multivariate analysis in the unfavorable intermediate-risk group showed that, relative to peers who started RT before ADT, men initiating RT later did not have significantly poorer overall survival, regardless of whether RT was initiated 1-60 days after starting ADT (hazard ratio for death, 1.03; P = .64), 61-120 days after (HR, 0.95; P = .42), or 121-180 days after (HR, 0.99; P = .90).
Findings were similar in the combined high-risk and very-high-risk group, with no significant elevation of mortality risk for patients initiating RT 1-60 days after starting ADT (HR, 1.07; P = .12), 61-120 days after (HR, 1.04; P = .36), or 121-180 days after (HR, 1.07; P = .17).
“These results validate the findings of two prior randomized trials and possibly justify the delay of prostate RT for patients currently receiving ADT until COVID-19 infection rates in the community and hospitals are lower,” the authors wrote.
Despite the fairly short follow-up period and other study limitations, “if COVID-19 outbreaks continue to occur sporadically during the coming months to years, these data could allow future flexibility about the timing of RT initiation,” the authors concluded.
Experts weigh in
“Overall, this study is asking a good question given the COVID situation and the fact that many providers are delaying RT due to COVID concerns of patients and providers,” Colleen A. Lawton, MD, of the Medical College of Wisconsin, Milwaukee, commented in an interview.
At the same time, Dr. Lawton cautioned about oversimplifying the issue, noting that results of the Radiation Therapy Oncology Group (RTOG) 9413 trial suggest important interactions between the anatomic extent of RT and the timing of ADT on outcomes (Int J Radiat Oncol Biol Phys. 2007 Nov 1;69[3]:646-55).
“I have certainly delayed some of my own patients with ADT during the COVID pandemic,” she reported. “No one knows what the maximum acceptable delay should be. A few months is likely not a problem, and a year is probably too much, but scientifically, we just don’t know.”
The interplay of volume irradiated and ADT timing is relevant here, agreed Mack Roach III, MD, of University of California, San Francisco.
In addition, the study did not address why ADT was given when it was, the duration of this therapy, and endpoints other than overall survival (such as prostate-specific antigen failure rate) that may better reflect the effectiveness of cancer treatment.
“Yes, delays are safe for patients on ADT, but not for the reasons stated. A more appropriate source of data is RTOG 9910, which compared 28 versus 8 weeks of ADT prior to RT for mostly intermediate-risk prostate cancer patients with comparable results,” Dr. Roach noted (J Clin Oncol. 2015 Feb 1;33[4]:332-9).
“Delay duration should be based on the risk of disease, but 6 months is probably safe, especially if on ADT,” he said.
Michael J. Zelefsky, MD, of Memorial Sloan Kettering Cancer Center in New York, said he agreed with the investigators’ main conclusions. “Once ADT suppression is achieved, maintaining patients on this regimen for 6 months would not likely lead to the development of a castrate-resistant state where radiotherapy would be less effective,” he elaborated.
However, limitations of the database used preclude conclusions about the safety of longer delays or the impact on other outcomes, he cautioned.
“This study provides further support to the accepted notion that delays of up to 6 months prior to initiation of planned prostate radiation would be safe and appropriate, especially where concerns of COVID outbreaks may present significant logistic challenges and concerns for the patient, who needs to commit to a course of daily radiation treatments, which could span for 5-8 weeks,” Dr. Zelefsky said.
“We have, in fact, adopted this approach in our clinics during the COVID outbreaks in New York,” he reported. “Most of our patients with unfavorable intermediate- or high-risk disease were initiated on ADT planned for at least 4-6 months before the radiotherapy was initiated. In addition, for these reasons, our preference has been to also offer such patients, if feasible, an ultrahypofractionated treatment course where the radiotherapy course is completed in five fractions over 1-2 weeks.”
This research was funded by the National Institutes of Health. The authors disclosed various grants and personal fees outside the submitted work. Dr. Lawton disclosed that she was a coauthor on RTOG 9413. Dr. Roach and Dr. Zelefsky disclosed no relevant conflicts of interest.
SOURCE: Dee EC et al. JAMA Oncol. 2020 Aug 13. doi: 10.1001/jamaoncol.2020.3545.
A study of more than 60,000 prostate cancer patients suggests it is safe to delay radiation therapy (RT) for at least 6 months for localized higher-risk disease being treated with androgen deprivation therapy.
These findings are relevant to oncology care in the COVID-19 era, as the pandemic has complicated delivery of radiation therapy (RT) in several ways, the study authors wrote in JAMA Oncology.
“Daily hospital trips for RT create many possible points of COVID-19 transmission, and patients with cancer are at high risk of COVID-19 mortality,” Edward Christopher Dee, a research fellow at Dana-Farber Cancer Institute in Boston, and colleagues wrote.
To assess the safety of delaying RT, the investigators analyzed National Cancer Database data for 63,858 men with localized but unfavorable intermediate-risk, high-risk, or very-high-risk prostate cancer diagnosed during 2004-2014 and managed with external beam RT and androgen deprivation therapy (ADT).
Only 5.6% of patients (n = 3,572) initiated their RT 0-60 days before starting ADT. Another 36.3% (n = 23,207) initiated RT 1-60 days after starting ADT, 47.4% (n = 30,285) initiated RT 61-120 days after starting ADT, and 10.6% (n = 6,794) initiated RT 121-180 days after starting ADT.
The investigators found that 10-year overall survival rates were similar regardless of when patients started RT.
Multivariate analysis in the unfavorable intermediate-risk group showed that, relative to peers who started RT before ADT, men initiating RT later did not have significantly poorer overall survival, regardless of whether RT was initiated 1-60 days after starting ADT (hazard ratio for death, 1.03; P = .64), 61-120 days after (HR, 0.95; P = .42), or 121-180 days after (HR, 0.99; P = .90).
Findings were similar in the combined high-risk and very-high-risk group, with no significant elevation of mortality risk for patients initiating RT 1-60 days after starting ADT (HR, 1.07; P = .12), 61-120 days after (HR, 1.04; P = .36), or 121-180 days after (HR, 1.07; P = .17).
“These results validate the findings of two prior randomized trials and possibly justify the delay of prostate RT for patients currently receiving ADT until COVID-19 infection rates in the community and hospitals are lower,” the authors wrote.
Despite the fairly short follow-up period and other study limitations, “if COVID-19 outbreaks continue to occur sporadically during the coming months to years, these data could allow future flexibility about the timing of RT initiation,” the authors concluded.
Experts weigh in
“Overall, this study is asking a good question given the COVID situation and the fact that many providers are delaying RT due to COVID concerns of patients and providers,” Colleen A. Lawton, MD, of the Medical College of Wisconsin, Milwaukee, commented in an interview.
At the same time, Dr. Lawton cautioned about oversimplifying the issue, noting that results of the Radiation Therapy Oncology Group (RTOG) 9413 trial suggest important interactions between the anatomic extent of RT and the timing of ADT on outcomes (Int J Radiat Oncol Biol Phys. 2007 Nov 1;69[3]:646-55).
“I have certainly delayed some of my own patients with ADT during the COVID pandemic,” she reported. “No one knows what the maximum acceptable delay should be. A few months is likely not a problem, and a year is probably too much, but scientifically, we just don’t know.”
The interplay of volume irradiated and ADT timing is relevant here, agreed Mack Roach III, MD, of University of California, San Francisco.
In addition, the study did not address why ADT was given when it was, the duration of this therapy, and endpoints other than overall survival (such as prostate-specific antigen failure rate) that may better reflect the effectiveness of cancer treatment.
“Yes, delays are safe for patients on ADT, but not for the reasons stated. A more appropriate source of data is RTOG 9910, which compared 28 versus 8 weeks of ADT prior to RT for mostly intermediate-risk prostate cancer patients with comparable results,” Dr. Roach noted (J Clin Oncol. 2015 Feb 1;33[4]:332-9).
“Delay duration should be based on the risk of disease, but 6 months is probably safe, especially if on ADT,” he said.
Michael J. Zelefsky, MD, of Memorial Sloan Kettering Cancer Center in New York, said he agreed with the investigators’ main conclusions. “Once ADT suppression is achieved, maintaining patients on this regimen for 6 months would not likely lead to the development of a castrate-resistant state where radiotherapy would be less effective,” he elaborated.
However, limitations of the database used preclude conclusions about the safety of longer delays or the impact on other outcomes, he cautioned.
“This study provides further support to the accepted notion that delays of up to 6 months prior to initiation of planned prostate radiation would be safe and appropriate, especially where concerns of COVID outbreaks may present significant logistic challenges and concerns for the patient, who needs to commit to a course of daily radiation treatments, which could span for 5-8 weeks,” Dr. Zelefsky said.
“We have, in fact, adopted this approach in our clinics during the COVID outbreaks in New York,” he reported. “Most of our patients with unfavorable intermediate- or high-risk disease were initiated on ADT planned for at least 4-6 months before the radiotherapy was initiated. In addition, for these reasons, our preference has been to also offer such patients, if feasible, an ultrahypofractionated treatment course where the radiotherapy course is completed in five fractions over 1-2 weeks.”
This research was funded by the National Institutes of Health. The authors disclosed various grants and personal fees outside the submitted work. Dr. Lawton disclosed that she was a coauthor on RTOG 9413. Dr. Roach and Dr. Zelefsky disclosed no relevant conflicts of interest.
SOURCE: Dee EC et al. JAMA Oncol. 2020 Aug 13. doi: 10.1001/jamaoncol.2020.3545.
A study of more than 60,000 prostate cancer patients suggests it is safe to delay radiation therapy (RT) for at least 6 months for localized higher-risk disease being treated with androgen deprivation therapy.
These findings are relevant to oncology care in the COVID-19 era, as the pandemic has complicated delivery of radiation therapy (RT) in several ways, the study authors wrote in JAMA Oncology.
“Daily hospital trips for RT create many possible points of COVID-19 transmission, and patients with cancer are at high risk of COVID-19 mortality,” Edward Christopher Dee, a research fellow at Dana-Farber Cancer Institute in Boston, and colleagues wrote.
To assess the safety of delaying RT, the investigators analyzed National Cancer Database data for 63,858 men with localized but unfavorable intermediate-risk, high-risk, or very-high-risk prostate cancer diagnosed during 2004-2014 and managed with external beam RT and androgen deprivation therapy (ADT).
Only 5.6% of patients (n = 3,572) initiated their RT 0-60 days before starting ADT. Another 36.3% (n = 23,207) initiated RT 1-60 days after starting ADT, 47.4% (n = 30,285) initiated RT 61-120 days after starting ADT, and 10.6% (n = 6,794) initiated RT 121-180 days after starting ADT.
The investigators found that 10-year overall survival rates were similar regardless of when patients started RT.
Multivariate analysis in the unfavorable intermediate-risk group showed that, relative to peers who started RT before ADT, men initiating RT later did not have significantly poorer overall survival, regardless of whether RT was initiated 1-60 days after starting ADT (hazard ratio for death, 1.03; P = .64), 61-120 days after (HR, 0.95; P = .42), or 121-180 days after (HR, 0.99; P = .90).
Findings were similar in the combined high-risk and very-high-risk group, with no significant elevation of mortality risk for patients initiating RT 1-60 days after starting ADT (HR, 1.07; P = .12), 61-120 days after (HR, 1.04; P = .36), or 121-180 days after (HR, 1.07; P = .17).
“These results validate the findings of two prior randomized trials and possibly justify the delay of prostate RT for patients currently receiving ADT until COVID-19 infection rates in the community and hospitals are lower,” the authors wrote.
Despite the fairly short follow-up period and other study limitations, “if COVID-19 outbreaks continue to occur sporadically during the coming months to years, these data could allow future flexibility about the timing of RT initiation,” the authors concluded.
Experts weigh in
“Overall, this study is asking a good question given the COVID situation and the fact that many providers are delaying RT due to COVID concerns of patients and providers,” Colleen A. Lawton, MD, of the Medical College of Wisconsin, Milwaukee, commented in an interview.
At the same time, Dr. Lawton cautioned about oversimplifying the issue, noting that results of the Radiation Therapy Oncology Group (RTOG) 9413 trial suggest important interactions between the anatomic extent of RT and the timing of ADT on outcomes (Int J Radiat Oncol Biol Phys. 2007 Nov 1;69[3]:646-55).
“I have certainly delayed some of my own patients with ADT during the COVID pandemic,” she reported. “No one knows what the maximum acceptable delay should be. A few months is likely not a problem, and a year is probably too much, but scientifically, we just don’t know.”
The interplay of volume irradiated and ADT timing is relevant here, agreed Mack Roach III, MD, of University of California, San Francisco.
In addition, the study did not address why ADT was given when it was, the duration of this therapy, and endpoints other than overall survival (such as prostate-specific antigen failure rate) that may better reflect the effectiveness of cancer treatment.
“Yes, delays are safe for patients on ADT, but not for the reasons stated. A more appropriate source of data is RTOG 9910, which compared 28 versus 8 weeks of ADT prior to RT for mostly intermediate-risk prostate cancer patients with comparable results,” Dr. Roach noted (J Clin Oncol. 2015 Feb 1;33[4]:332-9).
“Delay duration should be based on the risk of disease, but 6 months is probably safe, especially if on ADT,” he said.
Michael J. Zelefsky, MD, of Memorial Sloan Kettering Cancer Center in New York, said he agreed with the investigators’ main conclusions. “Once ADT suppression is achieved, maintaining patients on this regimen for 6 months would not likely lead to the development of a castrate-resistant state where radiotherapy would be less effective,” he elaborated.
However, limitations of the database used preclude conclusions about the safety of longer delays or the impact on other outcomes, he cautioned.
“This study provides further support to the accepted notion that delays of up to 6 months prior to initiation of planned prostate radiation would be safe and appropriate, especially where concerns of COVID outbreaks may present significant logistic challenges and concerns for the patient, who needs to commit to a course of daily radiation treatments, which could span for 5-8 weeks,” Dr. Zelefsky said.
“We have, in fact, adopted this approach in our clinics during the COVID outbreaks in New York,” he reported. “Most of our patients with unfavorable intermediate- or high-risk disease were initiated on ADT planned for at least 4-6 months before the radiotherapy was initiated. In addition, for these reasons, our preference has been to also offer such patients, if feasible, an ultrahypofractionated treatment course where the radiotherapy course is completed in five fractions over 1-2 weeks.”
This research was funded by the National Institutes of Health. The authors disclosed various grants and personal fees outside the submitted work. Dr. Lawton disclosed that she was a coauthor on RTOG 9413. Dr. Roach and Dr. Zelefsky disclosed no relevant conflicts of interest.
SOURCE: Dee EC et al. JAMA Oncol. 2020 Aug 13. doi: 10.1001/jamaoncol.2020.3545.
FROM JAMA ONCOLOGY
Incidence, prognosis of second lung cancers support long-term surveillance
Second lung cancers occurring up to a decade after the first are on the rise, but their prognosis is similar – especially when detected early – which supports long-term surveillance in survivors, finds a large population-based study.
Although guidelines recommend continued annual low-dose CT scan surveillance extending beyond 4 years for this population based on expert consensus, long-term evidence of benefit is lacking.
Investigators led by John M. Varlotto, MD, a radiation oncologist at the University of Massachusetts Medical Center, Worcester, analyzed Surveillance, Epidemiology & End Results (SEER) data for more than 58,000 patients with first and sometimes second non–small cell lung cancers initially treated by surgical resection.
Study results reported in Lung Cancer showed that the age-adjusted incidence of second lung cancers occurring 4-10 years after the first lung cancer rose sharply during the 1985-2014 study period, driven by a large uptick in women patients.
Among all patients, second lung cancers had similar overall survival as first lung cancers, but poorer lung cancer–specific survival. However, among the subset of patients having early-stage resectable disease (tumors measuring less than 4 cm with negative nodes), both outcomes were statistically indistinguishable.
“Because our investigation noted that the overall survival of patients undergoing a second lung cancer operation was similar to those patients undergoing a first operation, and because there is a rising rate of second lung cancer in lung cancer survivors, we feel that continued surveillance beyond the 4-year interval as recommended by the American Association for Thoracic Surgery as well as the [National Comprehensive Cancer Network] guidelines would be beneficial to long-term survivors of early-stage lung cancer,” Dr. Varlotto and coinvestigators wrote.
“The recent results from recent lung cancer screening studies demonstrate that females may benefit preferentially from screening … and our study suggests that these preferential benefits of increased CT scan surveillance may extend to females who are long-term survivors of lung cancer as well,” they added.
Findings in context
“As this is an observational study, it is challenging to understand what is driving the rise in prevalence of second lung cancers,” Mara Antonoff, MD, of The University of Texas MD Anderson Cancer Center in Houston commented in an interview.
“Overall, the findings are very important, as they suggest that we should continue to perform surveillance imaging for patients beyond recommended guidelines, which may allow us to achieve better survival outcomes for those individuals who develop a second lung cancer years after the first lung cancer,” she agreed.
“Just as lung cancer screening is important to identifying lung cancers at an earlier stage when they are more easily treatable and more likely to be cured, surveillance after an initial treatment for lung cancer would allow a diagnosis of second lung cancers at an earlier stage, so the patients can again achieve durable cure,” Dr. Antonoff concluded.
Study details
For the study, Dr. Varlotto and coinvestigators used data from SEER-13 and SEER-18 to identify patients with a lung cancer diagnosis during 1998-2013, and data from SEER-9, covering the years 1985-2014, to calculate rates of second cancers occurring 4-10 years after a first lung cancer.
Analyses were based on 58,758 patients with a surgically resected first primary lung cancer (55.9% with early-stage disease) and 384 patients with a surgically resected second primary lung cancer (77.6% with early-stage disease). Median follow-up was 76 months for the former and 46 months for the latter.
Results showed that in the 4-10 years after a first lung cancer diagnosis, the age-adjusted incidence of second lung cancers rose by study year but remained less than that of all other second cancers combined until the mid-2000s. Among women, incidence started rising sharply in 2001 and significantly exceeded that of all other second cancers starting in 2005.
In the entire population of study patients, propensity-adjusted analyses showed that second lung cancers were similar to first lung cancers on overall survival (P = .1726) but had worse lung cancer–specific survival (P = .0143). However, in the subset of patients with early-stage resectable disease, second and first lung cancers were similar on both overall survival (P = .3872) and lung cancer–specific survival (P = .1276).
Dr. Varlotto disclosed that he had no conflicts of interest. The study was funded by the Department of Radiation Oncology, University of Massachusetts. Dr. Antonoff disclosed that she had no relevant conflicts of interest.
SOURCE: Varlotto JM et al. Lung Cancer. 2020;147:115-122.
Second lung cancers occurring up to a decade after the first are on the rise, but their prognosis is similar – especially when detected early – which supports long-term surveillance in survivors, finds a large population-based study.
Although guidelines recommend continued annual low-dose CT scan surveillance extending beyond 4 years for this population based on expert consensus, long-term evidence of benefit is lacking.
Investigators led by John M. Varlotto, MD, a radiation oncologist at the University of Massachusetts Medical Center, Worcester, analyzed Surveillance, Epidemiology & End Results (SEER) data for more than 58,000 patients with first and sometimes second non–small cell lung cancers initially treated by surgical resection.
Study results reported in Lung Cancer showed that the age-adjusted incidence of second lung cancers occurring 4-10 years after the first lung cancer rose sharply during the 1985-2014 study period, driven by a large uptick in women patients.
Among all patients, second lung cancers had similar overall survival as first lung cancers, but poorer lung cancer–specific survival. However, among the subset of patients having early-stage resectable disease (tumors measuring less than 4 cm with negative nodes), both outcomes were statistically indistinguishable.
“Because our investigation noted that the overall survival of patients undergoing a second lung cancer operation was similar to those patients undergoing a first operation, and because there is a rising rate of second lung cancer in lung cancer survivors, we feel that continued surveillance beyond the 4-year interval as recommended by the American Association for Thoracic Surgery as well as the [National Comprehensive Cancer Network] guidelines would be beneficial to long-term survivors of early-stage lung cancer,” Dr. Varlotto and coinvestigators wrote.
“The recent results from recent lung cancer screening studies demonstrate that females may benefit preferentially from screening … and our study suggests that these preferential benefits of increased CT scan surveillance may extend to females who are long-term survivors of lung cancer as well,” they added.
Findings in context
“As this is an observational study, it is challenging to understand what is driving the rise in prevalence of second lung cancers,” Mara Antonoff, MD, of The University of Texas MD Anderson Cancer Center in Houston commented in an interview.
“Overall, the findings are very important, as they suggest that we should continue to perform surveillance imaging for patients beyond recommended guidelines, which may allow us to achieve better survival outcomes for those individuals who develop a second lung cancer years after the first lung cancer,” she agreed.
“Just as lung cancer screening is important to identifying lung cancers at an earlier stage when they are more easily treatable and more likely to be cured, surveillance after an initial treatment for lung cancer would allow a diagnosis of second lung cancers at an earlier stage, so the patients can again achieve durable cure,” Dr. Antonoff concluded.
Study details
For the study, Dr. Varlotto and coinvestigators used data from SEER-13 and SEER-18 to identify patients with a lung cancer diagnosis during 1998-2013, and data from SEER-9, covering the years 1985-2014, to calculate rates of second cancers occurring 4-10 years after a first lung cancer.
Analyses were based on 58,758 patients with a surgically resected first primary lung cancer (55.9% with early-stage disease) and 384 patients with a surgically resected second primary lung cancer (77.6% with early-stage disease). Median follow-up was 76 months for the former and 46 months for the latter.
Results showed that in the 4-10 years after a first lung cancer diagnosis, the age-adjusted incidence of second lung cancers rose by study year but remained less than that of all other second cancers combined until the mid-2000s. Among women, incidence started rising sharply in 2001 and significantly exceeded that of all other second cancers starting in 2005.
In the entire population of study patients, propensity-adjusted analyses showed that second lung cancers were similar to first lung cancers on overall survival (P = .1726) but had worse lung cancer–specific survival (P = .0143). However, in the subset of patients with early-stage resectable disease, second and first lung cancers were similar on both overall survival (P = .3872) and lung cancer–specific survival (P = .1276).
Dr. Varlotto disclosed that he had no conflicts of interest. The study was funded by the Department of Radiation Oncology, University of Massachusetts. Dr. Antonoff disclosed that she had no relevant conflicts of interest.
SOURCE: Varlotto JM et al. Lung Cancer. 2020;147:115-122.
Second lung cancers occurring up to a decade after the first are on the rise, but their prognosis is similar – especially when detected early – which supports long-term surveillance in survivors, finds a large population-based study.
Although guidelines recommend continued annual low-dose CT scan surveillance extending beyond 4 years for this population based on expert consensus, long-term evidence of benefit is lacking.
Investigators led by John M. Varlotto, MD, a radiation oncologist at the University of Massachusetts Medical Center, Worcester, analyzed Surveillance, Epidemiology & End Results (SEER) data for more than 58,000 patients with first and sometimes second non–small cell lung cancers initially treated by surgical resection.
Study results reported in Lung Cancer showed that the age-adjusted incidence of second lung cancers occurring 4-10 years after the first lung cancer rose sharply during the 1985-2014 study period, driven by a large uptick in women patients.
Among all patients, second lung cancers had similar overall survival as first lung cancers, but poorer lung cancer–specific survival. However, among the subset of patients having early-stage resectable disease (tumors measuring less than 4 cm with negative nodes), both outcomes were statistically indistinguishable.
“Because our investigation noted that the overall survival of patients undergoing a second lung cancer operation was similar to those patients undergoing a first operation, and because there is a rising rate of second lung cancer in lung cancer survivors, we feel that continued surveillance beyond the 4-year interval as recommended by the American Association for Thoracic Surgery as well as the [National Comprehensive Cancer Network] guidelines would be beneficial to long-term survivors of early-stage lung cancer,” Dr. Varlotto and coinvestigators wrote.
“The recent results from recent lung cancer screening studies demonstrate that females may benefit preferentially from screening … and our study suggests that these preferential benefits of increased CT scan surveillance may extend to females who are long-term survivors of lung cancer as well,” they added.
Findings in context
“As this is an observational study, it is challenging to understand what is driving the rise in prevalence of second lung cancers,” Mara Antonoff, MD, of The University of Texas MD Anderson Cancer Center in Houston commented in an interview.
“Overall, the findings are very important, as they suggest that we should continue to perform surveillance imaging for patients beyond recommended guidelines, which may allow us to achieve better survival outcomes for those individuals who develop a second lung cancer years after the first lung cancer,” she agreed.
“Just as lung cancer screening is important to identifying lung cancers at an earlier stage when they are more easily treatable and more likely to be cured, surveillance after an initial treatment for lung cancer would allow a diagnosis of second lung cancers at an earlier stage, so the patients can again achieve durable cure,” Dr. Antonoff concluded.
Study details
For the study, Dr. Varlotto and coinvestigators used data from SEER-13 and SEER-18 to identify patients with a lung cancer diagnosis during 1998-2013, and data from SEER-9, covering the years 1985-2014, to calculate rates of second cancers occurring 4-10 years after a first lung cancer.
Analyses were based on 58,758 patients with a surgically resected first primary lung cancer (55.9% with early-stage disease) and 384 patients with a surgically resected second primary lung cancer (77.6% with early-stage disease). Median follow-up was 76 months for the former and 46 months for the latter.
Results showed that in the 4-10 years after a first lung cancer diagnosis, the age-adjusted incidence of second lung cancers rose by study year but remained less than that of all other second cancers combined until the mid-2000s. Among women, incidence started rising sharply in 2001 and significantly exceeded that of all other second cancers starting in 2005.
In the entire population of study patients, propensity-adjusted analyses showed that second lung cancers were similar to first lung cancers on overall survival (P = .1726) but had worse lung cancer–specific survival (P = .0143). However, in the subset of patients with early-stage resectable disease, second and first lung cancers were similar on both overall survival (P = .3872) and lung cancer–specific survival (P = .1276).
Dr. Varlotto disclosed that he had no conflicts of interest. The study was funded by the Department of Radiation Oncology, University of Massachusetts. Dr. Antonoff disclosed that she had no relevant conflicts of interest.
SOURCE: Varlotto JM et al. Lung Cancer. 2020;147:115-122.
FROM LUNG CANCER
Early screening may halve breast cancer mortality in childhood cancer survivors
Two strategies – annual mammography with MRI and annual MRI alone – at least halved breast cancer mortality when started at the ages of 25 or 30 years.
Jennifer M. Yeh, PhD, of Harvard Medical School in Boston and colleagues reported these results in the Annals of Internal Medicine.
When cost was also considered, 30 years emerged as the preferred starting age, dropping the incremental cost-effectiveness ratio (ICER) below the generally accepted threshold of $100,000 per quality-adjusted life-year gained.
“Our findings underscore the importance of making sure that young women previously treated with chest radiation are informed about their elevated breast cancer risk and the benefits of routine screening. Both primary care providers and oncologists who care for survivors should discuss breast cancer screening with these patients,” Dr. Yeh and colleagues wrote.
“Screening guidelines should emphasize the importance of MRI screening (with or without mammography) among survivors,” the authors recommended. “Our findings also highlight the importance of ensuring that survivors have access to health insurance coverage for MRI screening.”
Implications for awareness, coverage
“My hope is that, by showing the significantly decreased risk of death associated with early breast cancer screening, with harm-benefit ratios considerably lower than benchmarks for average-risk women, this study will help health insurance companies see the benefit in covering early screening for at-risk survivors,” commented Karen E. Effinger, MD, of Emory University, Atlanta, and the Aflac Cancer & Blood Disorders Center at Children’s Healthcare of Atlanta.
“In many survivors, the cost of current screening [as recommended by] guidelines is prohibitive,” added Dr. Effinger, who was not involved in the current study.
The main concern regarding the study’s findings is generalizability to the contemporary era, given the use of a cohort diagnosed and treated decades ago and changes in radiation techniques and dosing since then, she noted in an interview. This limitation was addressed in a sensitivity analysis that halved the women’s base-case lifetime risk of breast cancer and still netted similar results.
“However, it will take many years to determine the true risk reduction of our current treatment strategies,” Dr. Effinger acknowledged.
“It is crucial that we improve our education of both survivors and our colleagues who care for these survivors, especially in regard to risk of subsequent malignancies and the benefits of screening,” Dr. Effinger maintained. “While many people are aware of the risk of breast cancer associated with BRCA mutations, the increased risk in survivors of childhood cancer is not as recognized by nononcologists. This study reinforces that increasing this awareness can save lives.”
In educating their patients about preventive care, health care providers must strike “a fine balance between discussing the risks and benefits of screening without provoking significant anxiety,” she concluded. “It is important for survivors to establish care with a primary care provider in order to develop trust and receive the guidance they need to decrease the risk of early mortality.”
Study details
Dr. Yeh and colleagues developed models to compare outcomes with various screening strategies among women aged 20 years who had received chest radiotherapy for childhood cancer during 1970-1986. The women had been diagnosed with Hodgkin lymphoma (55%), Wilms tumor (12%), non-Hodgkin lymphoma (8%), and other cancers.
The investigators conducted their analysis using data from the Childhood Cancer Survivor Study and other published sources, a lifetime time horizon, and a payer perspective.
The team assessed three strategies: no screening; digital mammography with MRI screening starting at 25 years of age (the current Children’s Oncology Group recommendation), 30 years, or 35 years and continuing to 74 years of age; and MRI only starting at age 25, 30, or 35 years and continuing to age 74 years.
The main study results showed that, without screening, women who had received chest radiation for childhood cancer had a 10%-11% lifetime risk of breast cancer mortality across models.
Relative to no screening, starting at age 25 years, the largest share of deaths was averted with the strategy of annual mammography with MRI – 56.3%-71.2% – or with the strategy of annual MRI alone – 55.7%-62.0%.
These two strategies also yielded the most screening tests, as well as the most false-positive test results and benign biopsy results.
For women who started screening at age 25, there were 4,188-4,879 false-positive test results per 1,000 women for mammography plus MRI and 3,283-3,764 false-positive results per 1,000 women for MRI alone.
For women who started screening at age 25, there were 1,340-1,561 benign biopsy results per 1,000 women for mammography plus MRI and 1,248-1,430 benign results per 1,000 women for MRI alone.
After cost was factored in, beginning screening at age 30 emerged as the preferred strategy to achieve an ICER threshold of less than $100,000 per quality-adjusted life-year gained.
When started at 30 years of age, annual mammography with MRI averted 54.7%-68.8% of breast cancer deaths, with an ICER of $25,400-$113,200 per quality-adjusted life-year gained. Annual MRI alone averted 54.0%-60.0% of breast cancer deaths, with an ICER of $21,800-$50,580 per quality-adjusted life-year gained.
This research was supported by grants from the National Cancer Institute, American Cancer Society, and American Lebanese Syrian Associated Charities. The authors disclosed relationships with GE Healthcare and Biovector. Dr. Effinger disclosed no relevant conflicts of interest.
SOURCE: Yeh JM et al. Ann Intern Med. 2020 Jul 7. doi: 10.7326/M19-3481.
Two strategies – annual mammography with MRI and annual MRI alone – at least halved breast cancer mortality when started at the ages of 25 or 30 years.
Jennifer M. Yeh, PhD, of Harvard Medical School in Boston and colleagues reported these results in the Annals of Internal Medicine.
When cost was also considered, 30 years emerged as the preferred starting age, dropping the incremental cost-effectiveness ratio (ICER) below the generally accepted threshold of $100,000 per quality-adjusted life-year gained.
“Our findings underscore the importance of making sure that young women previously treated with chest radiation are informed about their elevated breast cancer risk and the benefits of routine screening. Both primary care providers and oncologists who care for survivors should discuss breast cancer screening with these patients,” Dr. Yeh and colleagues wrote.
“Screening guidelines should emphasize the importance of MRI screening (with or without mammography) among survivors,” the authors recommended. “Our findings also highlight the importance of ensuring that survivors have access to health insurance coverage for MRI screening.”
Implications for awareness, coverage
“My hope is that, by showing the significantly decreased risk of death associated with early breast cancer screening, with harm-benefit ratios considerably lower than benchmarks for average-risk women, this study will help health insurance companies see the benefit in covering early screening for at-risk survivors,” commented Karen E. Effinger, MD, of Emory University, Atlanta, and the Aflac Cancer & Blood Disorders Center at Children’s Healthcare of Atlanta.
“In many survivors, the cost of current screening [as recommended by] guidelines is prohibitive,” added Dr. Effinger, who was not involved in the current study.
The main concern regarding the study’s findings is generalizability to the contemporary era, given the use of a cohort diagnosed and treated decades ago and changes in radiation techniques and dosing since then, she noted in an interview. This limitation was addressed in a sensitivity analysis that halved the women’s base-case lifetime risk of breast cancer and still netted similar results.
“However, it will take many years to determine the true risk reduction of our current treatment strategies,” Dr. Effinger acknowledged.
“It is crucial that we improve our education of both survivors and our colleagues who care for these survivors, especially in regard to risk of subsequent malignancies and the benefits of screening,” Dr. Effinger maintained. “While many people are aware of the risk of breast cancer associated with BRCA mutations, the increased risk in survivors of childhood cancer is not as recognized by nononcologists. This study reinforces that increasing this awareness can save lives.”
In educating their patients about preventive care, health care providers must strike “a fine balance between discussing the risks and benefits of screening without provoking significant anxiety,” she concluded. “It is important for survivors to establish care with a primary care provider in order to develop trust and receive the guidance they need to decrease the risk of early mortality.”
Study details
Dr. Yeh and colleagues developed models to compare outcomes with various screening strategies among women aged 20 years who had received chest radiotherapy for childhood cancer during 1970-1986. The women had been diagnosed with Hodgkin lymphoma (55%), Wilms tumor (12%), non-Hodgkin lymphoma (8%), and other cancers.
The investigators conducted their analysis using data from the Childhood Cancer Survivor Study and other published sources, a lifetime time horizon, and a payer perspective.
The team assessed three strategies: no screening; digital mammography with MRI screening starting at 25 years of age (the current Children’s Oncology Group recommendation), 30 years, or 35 years and continuing to 74 years of age; and MRI only starting at age 25, 30, or 35 years and continuing to age 74 years.
The main study results showed that, without screening, women who had received chest radiation for childhood cancer had a 10%-11% lifetime risk of breast cancer mortality across models.
Relative to no screening, starting at age 25 years, the largest share of deaths was averted with the strategy of annual mammography with MRI – 56.3%-71.2% – or with the strategy of annual MRI alone – 55.7%-62.0%.
These two strategies also yielded the most screening tests, as well as the most false-positive test results and benign biopsy results.
For women who started screening at age 25, there were 4,188-4,879 false-positive test results per 1,000 women for mammography plus MRI and 3,283-3,764 false-positive results per 1,000 women for MRI alone.
For women who started screening at age 25, there were 1,340-1,561 benign biopsy results per 1,000 women for mammography plus MRI and 1,248-1,430 benign results per 1,000 women for MRI alone.
After cost was factored in, beginning screening at age 30 emerged as the preferred strategy to achieve an ICER threshold of less than $100,000 per quality-adjusted life-year gained.
When started at 30 years of age, annual mammography with MRI averted 54.7%-68.8% of breast cancer deaths, with an ICER of $25,400-$113,200 per quality-adjusted life-year gained. Annual MRI alone averted 54.0%-60.0% of breast cancer deaths, with an ICER of $21,800-$50,580 per quality-adjusted life-year gained.
This research was supported by grants from the National Cancer Institute, American Cancer Society, and American Lebanese Syrian Associated Charities. The authors disclosed relationships with GE Healthcare and Biovector. Dr. Effinger disclosed no relevant conflicts of interest.
SOURCE: Yeh JM et al. Ann Intern Med. 2020 Jul 7. doi: 10.7326/M19-3481.
Two strategies – annual mammography with MRI and annual MRI alone – at least halved breast cancer mortality when started at the ages of 25 or 30 years.
Jennifer M. Yeh, PhD, of Harvard Medical School in Boston and colleagues reported these results in the Annals of Internal Medicine.
When cost was also considered, 30 years emerged as the preferred starting age, dropping the incremental cost-effectiveness ratio (ICER) below the generally accepted threshold of $100,000 per quality-adjusted life-year gained.
“Our findings underscore the importance of making sure that young women previously treated with chest radiation are informed about their elevated breast cancer risk and the benefits of routine screening. Both primary care providers and oncologists who care for survivors should discuss breast cancer screening with these patients,” Dr. Yeh and colleagues wrote.
“Screening guidelines should emphasize the importance of MRI screening (with or without mammography) among survivors,” the authors recommended. “Our findings also highlight the importance of ensuring that survivors have access to health insurance coverage for MRI screening.”
Implications for awareness, coverage
“My hope is that, by showing the significantly decreased risk of death associated with early breast cancer screening, with harm-benefit ratios considerably lower than benchmarks for average-risk women, this study will help health insurance companies see the benefit in covering early screening for at-risk survivors,” commented Karen E. Effinger, MD, of Emory University, Atlanta, and the Aflac Cancer & Blood Disorders Center at Children’s Healthcare of Atlanta.
“In many survivors, the cost of current screening [as recommended by] guidelines is prohibitive,” added Dr. Effinger, who was not involved in the current study.
The main concern regarding the study’s findings is generalizability to the contemporary era, given the use of a cohort diagnosed and treated decades ago and changes in radiation techniques and dosing since then, she noted in an interview. This limitation was addressed in a sensitivity analysis that halved the women’s base-case lifetime risk of breast cancer and still netted similar results.
“However, it will take many years to determine the true risk reduction of our current treatment strategies,” Dr. Effinger acknowledged.
“It is crucial that we improve our education of both survivors and our colleagues who care for these survivors, especially in regard to risk of subsequent malignancies and the benefits of screening,” Dr. Effinger maintained. “While many people are aware of the risk of breast cancer associated with BRCA mutations, the increased risk in survivors of childhood cancer is not as recognized by nononcologists. This study reinforces that increasing this awareness can save lives.”
In educating their patients about preventive care, health care providers must strike “a fine balance between discussing the risks and benefits of screening without provoking significant anxiety,” she concluded. “It is important for survivors to establish care with a primary care provider in order to develop trust and receive the guidance they need to decrease the risk of early mortality.”
Study details
Dr. Yeh and colleagues developed models to compare outcomes with various screening strategies among women aged 20 years who had received chest radiotherapy for childhood cancer during 1970-1986. The women had been diagnosed with Hodgkin lymphoma (55%), Wilms tumor (12%), non-Hodgkin lymphoma (8%), and other cancers.
The investigators conducted their analysis using data from the Childhood Cancer Survivor Study and other published sources, a lifetime time horizon, and a payer perspective.
The team assessed three strategies: no screening; digital mammography with MRI screening starting at 25 years of age (the current Children’s Oncology Group recommendation), 30 years, or 35 years and continuing to 74 years of age; and MRI only starting at age 25, 30, or 35 years and continuing to age 74 years.
The main study results showed that, without screening, women who had received chest radiation for childhood cancer had a 10%-11% lifetime risk of breast cancer mortality across models.
Relative to no screening, starting at age 25 years, the largest share of deaths was averted with the strategy of annual mammography with MRI – 56.3%-71.2% – or with the strategy of annual MRI alone – 55.7%-62.0%.
These two strategies also yielded the most screening tests, as well as the most false-positive test results and benign biopsy results.
For women who started screening at age 25, there were 4,188-4,879 false-positive test results per 1,000 women for mammography plus MRI and 3,283-3,764 false-positive results per 1,000 women for MRI alone.
For women who started screening at age 25, there were 1,340-1,561 benign biopsy results per 1,000 women for mammography plus MRI and 1,248-1,430 benign results per 1,000 women for MRI alone.
After cost was factored in, beginning screening at age 30 emerged as the preferred strategy to achieve an ICER threshold of less than $100,000 per quality-adjusted life-year gained.
When started at 30 years of age, annual mammography with MRI averted 54.7%-68.8% of breast cancer deaths, with an ICER of $25,400-$113,200 per quality-adjusted life-year gained. Annual MRI alone averted 54.0%-60.0% of breast cancer deaths, with an ICER of $21,800-$50,580 per quality-adjusted life-year gained.
This research was supported by grants from the National Cancer Institute, American Cancer Society, and American Lebanese Syrian Associated Charities. The authors disclosed relationships with GE Healthcare and Biovector. Dr. Effinger disclosed no relevant conflicts of interest.
SOURCE: Yeh JM et al. Ann Intern Med. 2020 Jul 7. doi: 10.7326/M19-3481.
FROM ANNALS OF INTERNAL MEDICINE
Trio of antibodies may enable earlier diagnosis of axSpA
Three autoantibodies to newly discovered axial spondyloarthritis peptides may improve early diagnosis of the disease, according to a cross-sectional cohort study reported in Arthritis & Rheumatology.
The Assessment in SpondyloArthritis International Society (ASAS) classification criteria were not intended for diagnosis and do not differentiate well between patients with early axial spondyloarthritis (axSpA) and patients with nonspecific chronic low back pain, note the investigators, who conducted their research under senior investigator Veerle Somers, PhD, professor of molecular biology at Hasselt (Belgium) University and vice dean of the School of Life Sciences at Transnationale Universiteit Limburg, also in Hasselt.
“Therefore, for many patients, axSpA diagnosis may be challenging and is often delayed by several years after the occurrence of first clinical symptoms, posing a problem for early treatment initiation,” they wrote.
The investigators used plasma samples from patients with early disease and an axSpA complementary DNA phage display library developed with synovial tissue to screen for IgG antibodies that displayed significantly higher reactivity to plasma pools from the early axSpA patients than healthy controls.
They then assessed presence of the antibodies with enzyme-linked immunosorbent assays in a mixed cohort (76 patients with early axSpA having mean disease duration of 2.8 years, 75 control patients with nonspecific chronic low back pain, 60 patients with RA, and 94 healthy controls) and in an axSpA-only cohort (174 patients, 79 of whom had early disease with mean disease duration of 1.4 years).
Screening identified antibodies to nine novel peptides – eight peptides showing partial homology to human proteins and one novel axSpA autoantigen, double homeobox protein 4 (DUX4) – that were more commonly present in patients with early axSpA than in healthy controls, Dr. Somers and coinvestigators reported.
Subsequent analyses focused on the three antibodies having the highest positive likelihood ratios for differentiating axSpA from chronic low back pain.
Some 14.2% of the combined group of all patients with early axSpA had at least one antibody in this panel, compared with just 5.3% of the patients with chronic low back pain (P = .0484), corresponding to 95% specificity.
Prevalence did not differ significantly from that in patients with RA (10.0%; P = .5025) or healthy controls (8.4%; P = .2292).
The positive likelihood ratio for confirming early axSpA using the three antibodies was 2.7, on par with the historical ratio of 2.5 seen for C-reactive protein (CRP), the currently used laboratory marker, the investigators noted.
Among the patients with chronic low back pain, the posttest probability for axSpA increased from 79% with presence of inflammatory back pain and positive test results for HLA-B27 and CRP to 91% with addition of testing for the three antibodies.
The researchers proposed that, “in combination with other laboratory markers such as HLA-B27 and CRP, antibodies against our [three peptides] ... could provide a novel tool for the diagnosis of a subset of axSpA patients,” but the three-peptide panel needs to be studied more in larger cohorts of early axSpA patients and controls with low back pain.
Findings in context
“The authors did a number of steps laudably,” James T. Rosenbaum, MD, chair of the division of arthritis and rheumatic diseases and the Edward E. Rosenbaum Professor of Inflammation Research at Oregon Health & Science University, Portland, commented in an interview. Specifically, they used a variety of appropriate controls, had discovery and validation sets, achieved a fairly good sample size, and applied the phage library technique.
“Despite this technological tour de force and the need for a sensitive and specific blood test to diagnose nonradiographic axSpA, this study is preliminary,” he cautioned. “For example, the authors found antibodies to DUX4 in 8% of axSpA patients versus 3% of healthy controls, 4% of patients with chronic low back pain, and 7% with RA. It took a combination of antigens to enhance the diagnostic accuracy of the ASAS criteria to diagnose axSpA. For each antigen that was studied, more than 80% of the axSpA patients had no detectable antibodies.”
Importantly, rheumatic diseases are often immune mediated without being autoimmune, calling into question the role of the antibodies, according to Dr. Rosenbaum.
“Even if further studies validate these observations, additional research needs to be done to support the concept that these antibodies cause disease as opposed to being mere epiphenomena as is suggested by the low prevalence,” he concluded. “Current hypotheses as to the cause of ankylosing spondylitis now point to the microbiome and autoinflammatory rather than autoimmune pathways, but the jury is still out.”
Dr. Somers and three coauthors disclosed having a patent pending on the markers. The study was funded by a personal grant from the Agency for Innovation by Science and Technology Flanders. Dr. Rosenbaum disclosed that he consults for AbbVie, Gilead, Novartis, Pfizer, Roche, and UCB.
SOURCE: Quaden D et al. Arthritis Rheumatol. 2020 Jul 8. doi: 10.1002/art.41427.
Three autoantibodies to newly discovered axial spondyloarthritis peptides may improve early diagnosis of the disease, according to a cross-sectional cohort study reported in Arthritis & Rheumatology.
The Assessment in SpondyloArthritis International Society (ASAS) classification criteria were not intended for diagnosis and do not differentiate well between patients with early axial spondyloarthritis (axSpA) and patients with nonspecific chronic low back pain, note the investigators, who conducted their research under senior investigator Veerle Somers, PhD, professor of molecular biology at Hasselt (Belgium) University and vice dean of the School of Life Sciences at Transnationale Universiteit Limburg, also in Hasselt.
“Therefore, for many patients, axSpA diagnosis may be challenging and is often delayed by several years after the occurrence of first clinical symptoms, posing a problem for early treatment initiation,” they wrote.
The investigators used plasma samples from patients with early disease and an axSpA complementary DNA phage display library developed with synovial tissue to screen for IgG antibodies that displayed significantly higher reactivity to plasma pools from the early axSpA patients than healthy controls.
They then assessed presence of the antibodies with enzyme-linked immunosorbent assays in a mixed cohort (76 patients with early axSpA having mean disease duration of 2.8 years, 75 control patients with nonspecific chronic low back pain, 60 patients with RA, and 94 healthy controls) and in an axSpA-only cohort (174 patients, 79 of whom had early disease with mean disease duration of 1.4 years).
Screening identified antibodies to nine novel peptides – eight peptides showing partial homology to human proteins and one novel axSpA autoantigen, double homeobox protein 4 (DUX4) – that were more commonly present in patients with early axSpA than in healthy controls, Dr. Somers and coinvestigators reported.
Subsequent analyses focused on the three antibodies having the highest positive likelihood ratios for differentiating axSpA from chronic low back pain.
Some 14.2% of the combined group of all patients with early axSpA had at least one antibody in this panel, compared with just 5.3% of the patients with chronic low back pain (P = .0484), corresponding to 95% specificity.
Prevalence did not differ significantly from that in patients with RA (10.0%; P = .5025) or healthy controls (8.4%; P = .2292).
The positive likelihood ratio for confirming early axSpA using the three antibodies was 2.7, on par with the historical ratio of 2.5 seen for C-reactive protein (CRP), the currently used laboratory marker, the investigators noted.
Among the patients with chronic low back pain, the posttest probability for axSpA increased from 79% with presence of inflammatory back pain and positive test results for HLA-B27 and CRP to 91% with addition of testing for the three antibodies.
The researchers proposed that, “in combination with other laboratory markers such as HLA-B27 and CRP, antibodies against our [three peptides] ... could provide a novel tool for the diagnosis of a subset of axSpA patients,” but the three-peptide panel needs to be studied more in larger cohorts of early axSpA patients and controls with low back pain.
Findings in context
“The authors did a number of steps laudably,” James T. Rosenbaum, MD, chair of the division of arthritis and rheumatic diseases and the Edward E. Rosenbaum Professor of Inflammation Research at Oregon Health & Science University, Portland, commented in an interview. Specifically, they used a variety of appropriate controls, had discovery and validation sets, achieved a fairly good sample size, and applied the phage library technique.
“Despite this technological tour de force and the need for a sensitive and specific blood test to diagnose nonradiographic axSpA, this study is preliminary,” he cautioned. “For example, the authors found antibodies to DUX4 in 8% of axSpA patients versus 3% of healthy controls, 4% of patients with chronic low back pain, and 7% with RA. It took a combination of antigens to enhance the diagnostic accuracy of the ASAS criteria to diagnose axSpA. For each antigen that was studied, more than 80% of the axSpA patients had no detectable antibodies.”
Importantly, rheumatic diseases are often immune mediated without being autoimmune, calling into question the role of the antibodies, according to Dr. Rosenbaum.
“Even if further studies validate these observations, additional research needs to be done to support the concept that these antibodies cause disease as opposed to being mere epiphenomena as is suggested by the low prevalence,” he concluded. “Current hypotheses as to the cause of ankylosing spondylitis now point to the microbiome and autoinflammatory rather than autoimmune pathways, but the jury is still out.”
Dr. Somers and three coauthors disclosed having a patent pending on the markers. The study was funded by a personal grant from the Agency for Innovation by Science and Technology Flanders. Dr. Rosenbaum disclosed that he consults for AbbVie, Gilead, Novartis, Pfizer, Roche, and UCB.
SOURCE: Quaden D et al. Arthritis Rheumatol. 2020 Jul 8. doi: 10.1002/art.41427.
Three autoantibodies to newly discovered axial spondyloarthritis peptides may improve early diagnosis of the disease, according to a cross-sectional cohort study reported in Arthritis & Rheumatology.
The Assessment in SpondyloArthritis International Society (ASAS) classification criteria were not intended for diagnosis and do not differentiate well between patients with early axial spondyloarthritis (axSpA) and patients with nonspecific chronic low back pain, note the investigators, who conducted their research under senior investigator Veerle Somers, PhD, professor of molecular biology at Hasselt (Belgium) University and vice dean of the School of Life Sciences at Transnationale Universiteit Limburg, also in Hasselt.
“Therefore, for many patients, axSpA diagnosis may be challenging and is often delayed by several years after the occurrence of first clinical symptoms, posing a problem for early treatment initiation,” they wrote.
The investigators used plasma samples from patients with early disease and an axSpA complementary DNA phage display library developed with synovial tissue to screen for IgG antibodies that displayed significantly higher reactivity to plasma pools from the early axSpA patients than healthy controls.
They then assessed presence of the antibodies with enzyme-linked immunosorbent assays in a mixed cohort (76 patients with early axSpA having mean disease duration of 2.8 years, 75 control patients with nonspecific chronic low back pain, 60 patients with RA, and 94 healthy controls) and in an axSpA-only cohort (174 patients, 79 of whom had early disease with mean disease duration of 1.4 years).
Screening identified antibodies to nine novel peptides – eight peptides showing partial homology to human proteins and one novel axSpA autoantigen, double homeobox protein 4 (DUX4) – that were more commonly present in patients with early axSpA than in healthy controls, Dr. Somers and coinvestigators reported.
Subsequent analyses focused on the three antibodies having the highest positive likelihood ratios for differentiating axSpA from chronic low back pain.
Some 14.2% of the combined group of all patients with early axSpA had at least one antibody in this panel, compared with just 5.3% of the patients with chronic low back pain (P = .0484), corresponding to 95% specificity.
Prevalence did not differ significantly from that in patients with RA (10.0%; P = .5025) or healthy controls (8.4%; P = .2292).
The positive likelihood ratio for confirming early axSpA using the three antibodies was 2.7, on par with the historical ratio of 2.5 seen for C-reactive protein (CRP), the currently used laboratory marker, the investigators noted.
Among the patients with chronic low back pain, the posttest probability for axSpA increased from 79% with presence of inflammatory back pain and positive test results for HLA-B27 and CRP to 91% with addition of testing for the three antibodies.
The researchers proposed that, “in combination with other laboratory markers such as HLA-B27 and CRP, antibodies against our [three peptides] ... could provide a novel tool for the diagnosis of a subset of axSpA patients,” but the three-peptide panel needs to be studied more in larger cohorts of early axSpA patients and controls with low back pain.
Findings in context
“The authors did a number of steps laudably,” James T. Rosenbaum, MD, chair of the division of arthritis and rheumatic diseases and the Edward E. Rosenbaum Professor of Inflammation Research at Oregon Health & Science University, Portland, commented in an interview. Specifically, they used a variety of appropriate controls, had discovery and validation sets, achieved a fairly good sample size, and applied the phage library technique.
“Despite this technological tour de force and the need for a sensitive and specific blood test to diagnose nonradiographic axSpA, this study is preliminary,” he cautioned. “For example, the authors found antibodies to DUX4 in 8% of axSpA patients versus 3% of healthy controls, 4% of patients with chronic low back pain, and 7% with RA. It took a combination of antigens to enhance the diagnostic accuracy of the ASAS criteria to diagnose axSpA. For each antigen that was studied, more than 80% of the axSpA patients had no detectable antibodies.”
Importantly, rheumatic diseases are often immune mediated without being autoimmune, calling into question the role of the antibodies, according to Dr. Rosenbaum.
“Even if further studies validate these observations, additional research needs to be done to support the concept that these antibodies cause disease as opposed to being mere epiphenomena as is suggested by the low prevalence,” he concluded. “Current hypotheses as to the cause of ankylosing spondylitis now point to the microbiome and autoinflammatory rather than autoimmune pathways, but the jury is still out.”
Dr. Somers and three coauthors disclosed having a patent pending on the markers. The study was funded by a personal grant from the Agency for Innovation by Science and Technology Flanders. Dr. Rosenbaum disclosed that he consults for AbbVie, Gilead, Novartis, Pfizer, Roche, and UCB.
SOURCE: Quaden D et al. Arthritis Rheumatol. 2020 Jul 8. doi: 10.1002/art.41427.
FROM ARTHRITIS & RHEUMATOLOGY
DECT has mixed performance in differentiating gout vs. CPPD
Dual-energy computed tomography (DECT) appears to have limited utility for differentiating between gout and calcium pyrophosphate deposition disease (CPPD), according to a German prospective cohort study. Findings were reported at the annual European Congress of Rheumatology, held online this year because of COVID-19.
“Differentiation of gout and pseudogout, or CPPD, is sometimes difficult,” said presenting investigator Valentin S. Schäfer, MD, associate professor of internal medicine and head of the department of rheumatology and clinical immunology at University Hospital Bonn (Germany).
“Arthrocentesis and subsequent polarization microscopy remains the gold standard,” he noted. “Novel diagnostic approaches, such as DECT, have recently been validated for gout, but limited data [are] available on the use of DECT in patients with CPPD.”
The investigators studied 30 patients: 22 with suspected gout and 8 with suspected CPPD. All underwent arthrocentesis with subsequent polarization microscopy for definitive diagnosis, plus clinical examination, ultrasound examination, conventional radiography, DECT, and assessment of 12 laboratory parameters.
For diagnosis of gout, DECT had a sensitivity and specificity of 59.1% and 100%, respectively, Dr. Schäfer reported, noting that this sensitivity falls considerably short of the 90% previously reported for gout.
Corresponding sensitivity and specificity were 90.9% and 75% for ultrasound, 58.8% and 100% for conventional radiography, and 81.8% and 87.5% for the rheumatologists’ suspected clinical diagnosis.
For diagnosis of CPPD, DECT had sensitivity of 37.5% and specificity of 81.8%. Corresponding values were 87.5% and 91% for ultrasound, 0% and 94.1% for conventional radiography, and 75.0% and 100% for suspected clinical diagnosis.
None of the 12 laboratory parameters studied – uric acid, C-reactive protein, organic phosphate, and leukocytes, among others – significantly differentiated between conditions.
“Both ultrasound and suspected clinical diagnosis had higher sensitivities than DECT for gout and CPPD,” Dr. Schäfer concluded. “Further studies with larger patient cohorts and perhaps modified scan protocols are needed in order to determine the diagnostic utility of DECT in CPPD.”
Findings in context
“Noninvasive, accurate methods for distinguishing between gout and CPPD will improve clinical care,” Sara K. Tedeschi, MD, MPH, predicted in an interview.
“Arthrocentesis is painful in an acutely inflamed joint, can be difficult to perform on small joints, and is underutilized in clinical practice,” she elaborated. And ultrasound is operator dependent and does not quantify crystal volume in and around the joint.
The question addressed by the study is therefore clinically relevant, according to Dr. Tedeschi, a rheumatologist at Brigham and Women’s Hospital and assistant professor of medicine at Harvard Medical School, Boston.
However, among the patients with CPPD, the study did not report specific phenotypes (acute inflammatory arthritis, chronic inflammatory arthritis, and osteoarthritis with calcium pyrophosphate deposits), she noted. “It is difficult to draw conclusions about the sensitivity or specificity of DECT for CPPD without this information, especially among just 8 CPPD patients.”
In addition, among the patients with gout, the proportion having new-onset disease with flare duration less than 6 weeks and the proportion with tophi were unknown, both of which affected DECT sensitivity in the previous study that reported 90% sensitivity. “Based on the 95% confidence interval in the present study, it is possible that with a larger sample size, DECT sensitivity for gout would have been higher,” she pointed out. “We also do not know the DECT software settings, which impact DECT interpretation as positive or negative for the crystal of interest.”
Finally, “it would be relevant to know what joints were aspirated and imaged in each group,” Dr. Tedeschi said. “For example, if the first metatarsophalangeal (MTP) joint was aspirated and imaged for half of the gout patients but for none of the CPPD patients, that may affect the study interpretation.”
The study did not receive any specific funding. Dr. Schäfer disclosed a variety of financial relationships with multiple pharmaceutical companies. Dr. Tedeschi disclosed receiving grant support from the National Institutes of Health to study imaging modalities for CPPD, and being first author on a study comparing the sensitivity of DECT, ultrasound, and x-ray for acute CPP crystal arthritis.
SOURCE: Kravchenko D et al. Ann Rheum Dis. 2020 Jun;79[suppl 1]:196.
Dual-energy computed tomography (DECT) appears to have limited utility for differentiating between gout and calcium pyrophosphate deposition disease (CPPD), according to a German prospective cohort study. Findings were reported at the annual European Congress of Rheumatology, held online this year because of COVID-19.
“Differentiation of gout and pseudogout, or CPPD, is sometimes difficult,” said presenting investigator Valentin S. Schäfer, MD, associate professor of internal medicine and head of the department of rheumatology and clinical immunology at University Hospital Bonn (Germany).
“Arthrocentesis and subsequent polarization microscopy remains the gold standard,” he noted. “Novel diagnostic approaches, such as DECT, have recently been validated for gout, but limited data [are] available on the use of DECT in patients with CPPD.”
The investigators studied 30 patients: 22 with suspected gout and 8 with suspected CPPD. All underwent arthrocentesis with subsequent polarization microscopy for definitive diagnosis, plus clinical examination, ultrasound examination, conventional radiography, DECT, and assessment of 12 laboratory parameters.
For diagnosis of gout, DECT had a sensitivity and specificity of 59.1% and 100%, respectively, Dr. Schäfer reported, noting that this sensitivity falls considerably short of the 90% previously reported for gout.
Corresponding sensitivity and specificity were 90.9% and 75% for ultrasound, 58.8% and 100% for conventional radiography, and 81.8% and 87.5% for the rheumatologists’ suspected clinical diagnosis.
For diagnosis of CPPD, DECT had sensitivity of 37.5% and specificity of 81.8%. Corresponding values were 87.5% and 91% for ultrasound, 0% and 94.1% for conventional radiography, and 75.0% and 100% for suspected clinical diagnosis.
None of the 12 laboratory parameters studied – uric acid, C-reactive protein, organic phosphate, and leukocytes, among others – significantly differentiated between conditions.
“Both ultrasound and suspected clinical diagnosis had higher sensitivities than DECT for gout and CPPD,” Dr. Schäfer concluded. “Further studies with larger patient cohorts and perhaps modified scan protocols are needed in order to determine the diagnostic utility of DECT in CPPD.”
Findings in context
“Noninvasive, accurate methods for distinguishing between gout and CPPD will improve clinical care,” Sara K. Tedeschi, MD, MPH, predicted in an interview.
“Arthrocentesis is painful in an acutely inflamed joint, can be difficult to perform on small joints, and is underutilized in clinical practice,” she elaborated. And ultrasound is operator dependent and does not quantify crystal volume in and around the joint.
The question addressed by the study is therefore clinically relevant, according to Dr. Tedeschi, a rheumatologist at Brigham and Women’s Hospital and assistant professor of medicine at Harvard Medical School, Boston.
However, among the patients with CPPD, the study did not report specific phenotypes (acute inflammatory arthritis, chronic inflammatory arthritis, and osteoarthritis with calcium pyrophosphate deposits), she noted. “It is difficult to draw conclusions about the sensitivity or specificity of DECT for CPPD without this information, especially among just 8 CPPD patients.”
In addition, among the patients with gout, the proportion having new-onset disease with flare duration less than 6 weeks and the proportion with tophi were unknown, both of which affected DECT sensitivity in the previous study that reported 90% sensitivity. “Based on the 95% confidence interval in the present study, it is possible that with a larger sample size, DECT sensitivity for gout would have been higher,” she pointed out. “We also do not know the DECT software settings, which impact DECT interpretation as positive or negative for the crystal of interest.”
Finally, “it would be relevant to know what joints were aspirated and imaged in each group,” Dr. Tedeschi said. “For example, if the first metatarsophalangeal (MTP) joint was aspirated and imaged for half of the gout patients but for none of the CPPD patients, that may affect the study interpretation.”
The study did not receive any specific funding. Dr. Schäfer disclosed a variety of financial relationships with multiple pharmaceutical companies. Dr. Tedeschi disclosed receiving grant support from the National Institutes of Health to study imaging modalities for CPPD, and being first author on a study comparing the sensitivity of DECT, ultrasound, and x-ray for acute CPP crystal arthritis.
SOURCE: Kravchenko D et al. Ann Rheum Dis. 2020 Jun;79[suppl 1]:196.
Dual-energy computed tomography (DECT) appears to have limited utility for differentiating between gout and calcium pyrophosphate deposition disease (CPPD), according to a German prospective cohort study. Findings were reported at the annual European Congress of Rheumatology, held online this year because of COVID-19.
“Differentiation of gout and pseudogout, or CPPD, is sometimes difficult,” said presenting investigator Valentin S. Schäfer, MD, associate professor of internal medicine and head of the department of rheumatology and clinical immunology at University Hospital Bonn (Germany).
“Arthrocentesis and subsequent polarization microscopy remains the gold standard,” he noted. “Novel diagnostic approaches, such as DECT, have recently been validated for gout, but limited data [are] available on the use of DECT in patients with CPPD.”
The investigators studied 30 patients: 22 with suspected gout and 8 with suspected CPPD. All underwent arthrocentesis with subsequent polarization microscopy for definitive diagnosis, plus clinical examination, ultrasound examination, conventional radiography, DECT, and assessment of 12 laboratory parameters.
For diagnosis of gout, DECT had a sensitivity and specificity of 59.1% and 100%, respectively, Dr. Schäfer reported, noting that this sensitivity falls considerably short of the 90% previously reported for gout.
Corresponding sensitivity and specificity were 90.9% and 75% for ultrasound, 58.8% and 100% for conventional radiography, and 81.8% and 87.5% for the rheumatologists’ suspected clinical diagnosis.
For diagnosis of CPPD, DECT had sensitivity of 37.5% and specificity of 81.8%. Corresponding values were 87.5% and 91% for ultrasound, 0% and 94.1% for conventional radiography, and 75.0% and 100% for suspected clinical diagnosis.
None of the 12 laboratory parameters studied – uric acid, C-reactive protein, organic phosphate, and leukocytes, among others – significantly differentiated between conditions.
“Both ultrasound and suspected clinical diagnosis had higher sensitivities than DECT for gout and CPPD,” Dr. Schäfer concluded. “Further studies with larger patient cohorts and perhaps modified scan protocols are needed in order to determine the diagnostic utility of DECT in CPPD.”
Findings in context
“Noninvasive, accurate methods for distinguishing between gout and CPPD will improve clinical care,” Sara K. Tedeschi, MD, MPH, predicted in an interview.
“Arthrocentesis is painful in an acutely inflamed joint, can be difficult to perform on small joints, and is underutilized in clinical practice,” she elaborated. And ultrasound is operator dependent and does not quantify crystal volume in and around the joint.
The question addressed by the study is therefore clinically relevant, according to Dr. Tedeschi, a rheumatologist at Brigham and Women’s Hospital and assistant professor of medicine at Harvard Medical School, Boston.
However, among the patients with CPPD, the study did not report specific phenotypes (acute inflammatory arthritis, chronic inflammatory arthritis, and osteoarthritis with calcium pyrophosphate deposits), she noted. “It is difficult to draw conclusions about the sensitivity or specificity of DECT for CPPD without this information, especially among just 8 CPPD patients.”
In addition, among the patients with gout, the proportion having new-onset disease with flare duration less than 6 weeks and the proportion with tophi were unknown, both of which affected DECT sensitivity in the previous study that reported 90% sensitivity. “Based on the 95% confidence interval in the present study, it is possible that with a larger sample size, DECT sensitivity for gout would have been higher,” she pointed out. “We also do not know the DECT software settings, which impact DECT interpretation as positive or negative for the crystal of interest.”
Finally, “it would be relevant to know what joints were aspirated and imaged in each group,” Dr. Tedeschi said. “For example, if the first metatarsophalangeal (MTP) joint was aspirated and imaged for half of the gout patients but for none of the CPPD patients, that may affect the study interpretation.”
The study did not receive any specific funding. Dr. Schäfer disclosed a variety of financial relationships with multiple pharmaceutical companies. Dr. Tedeschi disclosed receiving grant support from the National Institutes of Health to study imaging modalities for CPPD, and being first author on a study comparing the sensitivity of DECT, ultrasound, and x-ray for acute CPP crystal arthritis.
SOURCE: Kravchenko D et al. Ann Rheum Dis. 2020 Jun;79[suppl 1]:196.
FROM THE EULAR 2020 E-CONGRESS
New registry focuses on rheumatic immune-related AEs of cancer therapy
Its first findings were reported at the annual European Congress of Rheumatology, held online this year due to COVID-19.
“We have limited knowledge on the interrelationships between malignant and rheumatic diseases on both the clinical and molecular level, and we have a large unmet need for management guidelines in the case of the coincidence of both disease entities,” noted lead author Karolina Benesova, MD, of the department of hematology, oncology, and rheumatology at University Hospital Heidelberg (Germany).
The TRheuMa registry – Therapy-Induced Rheumatic Symptoms in Patients with Malignancy – is one of three registries in a multicenter observational project exploring various contexts between malignant and rheumatic diseases. Over its first 22 months, the registry recruited 69 patients having rheumatic symptoms as a result of immune checkpoint inhibitor therapy or other cancer therapies.
Registry findings
The largest shares of patients had non–small cell lung cancer (38%) or melanoma (33%), Dr. Benesova reported. The immune checkpoint inhibitors most commonly received were pembrolizumab (Keytruda), nivolumab (Opdivo), and ipilimumab (Yervoy).
The immune-related adverse events usually presented with symptoms of de novo spondyloarthritis or psoriatic arthritis (42%), late-onset RA (17%), or polymyalgia rheumatica (14%). But 16% of the patients were experiencing a flare of a preexisting rheumatic and musculoskeletal disease.
Laboratory findings differed somewhat from those of classical rheumatic and musculoskeletal diseases, according to Dr. Benesova. Specific findings were rare; in particular, most patients did not have detectable autoantibodies. However, 76% had an elevated C-reactive protein level and 39% had an elevated soluble CD25 level. In addition, nearly all patients (96%) undergoing joint ultrasound had pathologic findings.
“Based on our experiences from interdisciplinary care together with our local oncologists, we have developed a therapeutic algorithm for rheumatic immune-related adverse events,” she reported, noting that the algorithm is consistent with recently published recommendations in this area.
The large majority of patients were adequately treated with prednisone at a dose greater than 10 mg (40%) or at a dose of 10 mg or less with or without an NSAID (40%), while some received NSAID monotherapy (14%).
“We have a growing proportion of patients on conventional or biological [disease-modifying antirheumatic drugs],” Dr. Benesova noted. “These are mostly patients with preexisting rheumatic and musculoskeletal disease or highly suspected de novo classical rheumatic and musculoskeletal disease under checkpoint inhibitor therapy.”
Patients with melanoma having a rheumatic immune-related adverse event had a better response to their therapy than historical counterparts who did not have such events: 39% of the former had a complete response, relative to merely 4% of the latter.
Only a small proportion of patients overall (9%) had to discontinue immune checkpoint inhibitor therapy because of their adverse event, and some of them may be eligible for rechallenge if their cancer progresses, Dr. Benesova noted.
“There is still a lot to be done,” she stated, such as better elucidating the nature of these adverse events [whether transient side effects or a triggering of chronic rheumatic and musculoskeletal diseases], the need for a defensive treatment strategy, and the advisability of closer monitoring of high-risk patients given immune checkpoint inhibitors. “We are aiming at solving these questions in the next few years,” she concluded.
Findings in context
“Registries are important to gain prospective data on patient outcomes,” Sabina Sandigursky, MD, an instructor in the division of rheumatology at the Laura and Isaac Perlmutter Cancer Center at New York University, commented in an interview. “One must be careful, while interpreting these data, especially since they are not randomized, controlled trials.”
Patterns may differ at other centers, too, she pointed out. “The German registry reported a predominance of spondyloarthritis-like disease; however, our patients have a predominance of small-joint involvement. It is unclear what accounts for this difference.”
Individual institutions in North America are similarly collecting data on this patient population, with efforts underway to compile those data to provide a larger picture, according to Dr. Sandigursky.
“Many of the syndromes that we consider to be rheumatic immune-related adverse events have been well described by groups from the U.S., Canada, Australia, and European Union,” she concluded. “From this registry, we can observe how patients are being treated in real time since this information is largely consensus based.”
The study did not receive any specific funding. Dr. Benesova disclosed grant/research support from AbbVie, Novartis, Rheumaliga Baden-Wurttemberg, and the University of Heidelberg, and consultancies, speaker fees, and/or travel reimbursements from AbbVie, Bristol-Myers Squibb, Gilead, Janssen, Medac, Merck Sharp & Dohme, Mundipharma, Novartis, Pfizer, Roche, and UCB. Some of her coauthors also disclosed financial relationships with industry. Dr. Sandigursky disclosed having no relevant conflicts of interest.
SOURCE: Benesova K et al. Ann Rheum Dis 2020;79[suppl 1]:168-9, Abstract OP0270.
Its first findings were reported at the annual European Congress of Rheumatology, held online this year due to COVID-19.
“We have limited knowledge on the interrelationships between malignant and rheumatic diseases on both the clinical and molecular level, and we have a large unmet need for management guidelines in the case of the coincidence of both disease entities,” noted lead author Karolina Benesova, MD, of the department of hematology, oncology, and rheumatology at University Hospital Heidelberg (Germany).
The TRheuMa registry – Therapy-Induced Rheumatic Symptoms in Patients with Malignancy – is one of three registries in a multicenter observational project exploring various contexts between malignant and rheumatic diseases. Over its first 22 months, the registry recruited 69 patients having rheumatic symptoms as a result of immune checkpoint inhibitor therapy or other cancer therapies.
Registry findings
The largest shares of patients had non–small cell lung cancer (38%) or melanoma (33%), Dr. Benesova reported. The immune checkpoint inhibitors most commonly received were pembrolizumab (Keytruda), nivolumab (Opdivo), and ipilimumab (Yervoy).
The immune-related adverse events usually presented with symptoms of de novo spondyloarthritis or psoriatic arthritis (42%), late-onset RA (17%), or polymyalgia rheumatica (14%). But 16% of the patients were experiencing a flare of a preexisting rheumatic and musculoskeletal disease.
Laboratory findings differed somewhat from those of classical rheumatic and musculoskeletal diseases, according to Dr. Benesova. Specific findings were rare; in particular, most patients did not have detectable autoantibodies. However, 76% had an elevated C-reactive protein level and 39% had an elevated soluble CD25 level. In addition, nearly all patients (96%) undergoing joint ultrasound had pathologic findings.
“Based on our experiences from interdisciplinary care together with our local oncologists, we have developed a therapeutic algorithm for rheumatic immune-related adverse events,” she reported, noting that the algorithm is consistent with recently published recommendations in this area.
The large majority of patients were adequately treated with prednisone at a dose greater than 10 mg (40%) or at a dose of 10 mg or less with or without an NSAID (40%), while some received NSAID monotherapy (14%).
“We have a growing proportion of patients on conventional or biological [disease-modifying antirheumatic drugs],” Dr. Benesova noted. “These are mostly patients with preexisting rheumatic and musculoskeletal disease or highly suspected de novo classical rheumatic and musculoskeletal disease under checkpoint inhibitor therapy.”
Patients with melanoma having a rheumatic immune-related adverse event had a better response to their therapy than historical counterparts who did not have such events: 39% of the former had a complete response, relative to merely 4% of the latter.
Only a small proportion of patients overall (9%) had to discontinue immune checkpoint inhibitor therapy because of their adverse event, and some of them may be eligible for rechallenge if their cancer progresses, Dr. Benesova noted.
“There is still a lot to be done,” she stated, such as better elucidating the nature of these adverse events [whether transient side effects or a triggering of chronic rheumatic and musculoskeletal diseases], the need for a defensive treatment strategy, and the advisability of closer monitoring of high-risk patients given immune checkpoint inhibitors. “We are aiming at solving these questions in the next few years,” she concluded.
Findings in context
“Registries are important to gain prospective data on patient outcomes,” Sabina Sandigursky, MD, an instructor in the division of rheumatology at the Laura and Isaac Perlmutter Cancer Center at New York University, commented in an interview. “One must be careful, while interpreting these data, especially since they are not randomized, controlled trials.”
Patterns may differ at other centers, too, she pointed out. “The German registry reported a predominance of spondyloarthritis-like disease; however, our patients have a predominance of small-joint involvement. It is unclear what accounts for this difference.”
Individual institutions in North America are similarly collecting data on this patient population, with efforts underway to compile those data to provide a larger picture, according to Dr. Sandigursky.
“Many of the syndromes that we consider to be rheumatic immune-related adverse events have been well described by groups from the U.S., Canada, Australia, and European Union,” she concluded. “From this registry, we can observe how patients are being treated in real time since this information is largely consensus based.”
The study did not receive any specific funding. Dr. Benesova disclosed grant/research support from AbbVie, Novartis, Rheumaliga Baden-Wurttemberg, and the University of Heidelberg, and consultancies, speaker fees, and/or travel reimbursements from AbbVie, Bristol-Myers Squibb, Gilead, Janssen, Medac, Merck Sharp & Dohme, Mundipharma, Novartis, Pfizer, Roche, and UCB. Some of her coauthors also disclosed financial relationships with industry. Dr. Sandigursky disclosed having no relevant conflicts of interest.
SOURCE: Benesova K et al. Ann Rheum Dis 2020;79[suppl 1]:168-9, Abstract OP0270.
Its first findings were reported at the annual European Congress of Rheumatology, held online this year due to COVID-19.
“We have limited knowledge on the interrelationships between malignant and rheumatic diseases on both the clinical and molecular level, and we have a large unmet need for management guidelines in the case of the coincidence of both disease entities,” noted lead author Karolina Benesova, MD, of the department of hematology, oncology, and rheumatology at University Hospital Heidelberg (Germany).
The TRheuMa registry – Therapy-Induced Rheumatic Symptoms in Patients with Malignancy – is one of three registries in a multicenter observational project exploring various contexts between malignant and rheumatic diseases. Over its first 22 months, the registry recruited 69 patients having rheumatic symptoms as a result of immune checkpoint inhibitor therapy or other cancer therapies.
Registry findings
The largest shares of patients had non–small cell lung cancer (38%) or melanoma (33%), Dr. Benesova reported. The immune checkpoint inhibitors most commonly received were pembrolizumab (Keytruda), nivolumab (Opdivo), and ipilimumab (Yervoy).
The immune-related adverse events usually presented with symptoms of de novo spondyloarthritis or psoriatic arthritis (42%), late-onset RA (17%), or polymyalgia rheumatica (14%). But 16% of the patients were experiencing a flare of a preexisting rheumatic and musculoskeletal disease.
Laboratory findings differed somewhat from those of classical rheumatic and musculoskeletal diseases, according to Dr. Benesova. Specific findings were rare; in particular, most patients did not have detectable autoantibodies. However, 76% had an elevated C-reactive protein level and 39% had an elevated soluble CD25 level. In addition, nearly all patients (96%) undergoing joint ultrasound had pathologic findings.
“Based on our experiences from interdisciplinary care together with our local oncologists, we have developed a therapeutic algorithm for rheumatic immune-related adverse events,” she reported, noting that the algorithm is consistent with recently published recommendations in this area.
The large majority of patients were adequately treated with prednisone at a dose greater than 10 mg (40%) or at a dose of 10 mg or less with or without an NSAID (40%), while some received NSAID monotherapy (14%).
“We have a growing proportion of patients on conventional or biological [disease-modifying antirheumatic drugs],” Dr. Benesova noted. “These are mostly patients with preexisting rheumatic and musculoskeletal disease or highly suspected de novo classical rheumatic and musculoskeletal disease under checkpoint inhibitor therapy.”
Patients with melanoma having a rheumatic immune-related adverse event had a better response to their therapy than historical counterparts who did not have such events: 39% of the former had a complete response, relative to merely 4% of the latter.
Only a small proportion of patients overall (9%) had to discontinue immune checkpoint inhibitor therapy because of their adverse event, and some of them may be eligible for rechallenge if their cancer progresses, Dr. Benesova noted.
“There is still a lot to be done,” she stated, such as better elucidating the nature of these adverse events [whether transient side effects or a triggering of chronic rheumatic and musculoskeletal diseases], the need for a defensive treatment strategy, and the advisability of closer monitoring of high-risk patients given immune checkpoint inhibitors. “We are aiming at solving these questions in the next few years,” she concluded.
Findings in context
“Registries are important to gain prospective data on patient outcomes,” Sabina Sandigursky, MD, an instructor in the division of rheumatology at the Laura and Isaac Perlmutter Cancer Center at New York University, commented in an interview. “One must be careful, while interpreting these data, especially since they are not randomized, controlled trials.”
Patterns may differ at other centers, too, she pointed out. “The German registry reported a predominance of spondyloarthritis-like disease; however, our patients have a predominance of small-joint involvement. It is unclear what accounts for this difference.”
Individual institutions in North America are similarly collecting data on this patient population, with efforts underway to compile those data to provide a larger picture, according to Dr. Sandigursky.
“Many of the syndromes that we consider to be rheumatic immune-related adverse events have been well described by groups from the U.S., Canada, Australia, and European Union,” she concluded. “From this registry, we can observe how patients are being treated in real time since this information is largely consensus based.”
The study did not receive any specific funding. Dr. Benesova disclosed grant/research support from AbbVie, Novartis, Rheumaliga Baden-Wurttemberg, and the University of Heidelberg, and consultancies, speaker fees, and/or travel reimbursements from AbbVie, Bristol-Myers Squibb, Gilead, Janssen, Medac, Merck Sharp & Dohme, Mundipharma, Novartis, Pfizer, Roche, and UCB. Some of her coauthors also disclosed financial relationships with industry. Dr. Sandigursky disclosed having no relevant conflicts of interest.
SOURCE: Benesova K et al. Ann Rheum Dis 2020;79[suppl 1]:168-9, Abstract OP0270.
FROM THE EULAR 2020 E-CONGRESS
Studies give new insight on starting, stopping etanercept in nonradiographic axSpA
The results from a pair of clinical trials should help to take the guesswork out of starting and stopping the tumor necrosis factor inhibitor etanercept (Enbrel) in patients with nonradiographic axial spondyloarthritis (nr-axSpA). The trials were reported at the annual European Congress of Rheumatology, held online this year due to COVID-19.
Optimal use of etanercept in this disease is still being defined, according to the investigators. Its effects, if any, when given very early in the disease course is unclear, and guidance is conflicting when it comes to stopping the drug after inactive disease is achieved.
In the Dutch randomized controlled PrevAS trial of 80 patients with suspected very early nr-axSpA, initiating etanercept instead of placebo did not significantly improve the odds of achieving a 20% improvement in Assessment of Spondyloarthritis International Society (ASAS 20) response criteria at week 16.
And in the multinational, open-label, phase 4 RE-EMBARK trial, three-quarters of the 119 patients with nr-axSpA who achieved inactive disease on etanercept and stopped the drug experienced a flare within 40 weeks. However, the majority were able to regain disease inactivity after restarting the drug.
Findings in context
“We all have some patients like this [PrevAS population] where we strongly believe they have axial spondyloarthritis but do not fully qualify,” Nigil Haroon MD, PhD, said in an interview. “From a clinical decision-making process, we may diagnose these patients with axial spondyloarthritis, but due to restrictions in access to medications, we have difficulty accessing biologic medications for them. Hence, this study has practical implications.”
“It has already been shown in other, much larger studies that, even in patients who satisfy the criteria of axial spondyloarthritis, those who are MRI and CRP [C-reactive protein] negative are unlikely to respond, so the results are not surprising,” commented Dr. Haroon, who is codirector of the spondylitis program at the University Health Network and associate professor of medicine and rheumatology at the University of Toronto.
Although intended to be a population with suspected very early disease, several of the PrevAS patients would have met ASAS criteria for the disease at baseline, Dr. Haroon cautioned. In addition, the small sample size precluded subgroup analyses.
“The overall conclusion should be, this is a negative study, rather than state there was a trend to better improvement on etanercept. Although there are practical implications, as mentioned, I don’t think this study, with the numbers and the results presented, will change clinical practice,” he said.
The question of stopping biologics in nr-axSpA was previously addressed in the ABILITY-3 randomized trial of adalimumab (Humira), which found that flares were significantly more common with stopping versus continuing the drug and only about half of patients were able to get back in remission by restarting the drug, according to Dr. Haroon.
However, the RE-EMBARK and ABILITY-3 studies differed in both design and patient population, he noted. For example, the mean disease duration was only about 2 years in the former study, compared with 7 years in the latter.
The initial 59% rate of attaining inactive disease on etanercept in RE-EMBARK was “impressive,” Dr. Haroon said, “but as this was an open-label study, higher values are expected.”
“The message in both studies is that stopping biologics completely is not a good idea as the majority of patients, 70%-75%, will relapse within a short period,” he concluded. “However, it should be kept in mind that these [RE-EMBARK] patients received biologic only for a short 24-week period. This study does not answer the question of whether nonradiographic axial spondyloarthritis patients with sustained inactive disease can be taken off biologics abruptly without a taper over time.”
Details of the studies
In the PrevAS trial, Tamara Rusman, a PhD candidate in Rheumatology at the VU University Medical Center Amsterdam and coinvestigators studied patients meeting Calin criteria for inflammatory back pain who had high disease activity plus either HLA-B27 positivity with at least one feature of axial spondyloarthritis or HLA-B27 negativity with two features.
This population is of interest because “most studies have included only patients with nonradiographic axial spondyloarthritis with a positive MRI of the sacroiliac joints and/or an elevated C-reactive protein level,” she noted.
Results showed that, during 16 weeks of treatment, etanercept users had a nonsignficantly higher rate of achieving an ASAS 20 response with etanercept versus placebo users (17% vs. 11%; hazard ratio, 2.1; P = .2). The etanercept group also had a somewhat higher rate of response as defined by the Ankylosing Spondylitis Disease Activity Score CRP (ASDAS-CRP) criterion (25% vs. 13%; hazard ratio, 1.1; P = .8).
“Based on these data, early treatment in inflammatory back pain patients prone to develop axial spondyloarthritis seems not to be useful,” Ms. Rusman concluded. “However, monitoring of these patients should be continued since they remain a risk group for developing axial spondyloarthritis.”
In the RE-EMBARK trial, investigators led by Filip Van den Bosch, MD, PhD, Rheumatology Head-of-Clinic at Ghent (Belgium) University Hospital, started with a cohort of 208 patients with nr-axSpA who were given etanercept and background NSAIDs for 24 weeks.
“Current guidelines do not agree on whether a TNF-blocking agent or another biological DMARD should be tapered once a status of low disease activity or remission is achieved,” he noted.
Overall, 59% of the patients achieved inactive disease (defined as an ASDAS-CRP < 1.3) and discontinued etanercept.
During the next 40 weeks, 24% of these patients maintained inactive disease with only the background NSAID therapy. Among the 75% who experienced a flare, defined as an ASDAS with erythrocyte sedimentation rate (ASDAS-ESR) score of 2.1 or greater, the median time to flare was 16.1 weeks. Fully 62% of this group were able to regain disease inactivity within 12 weeks of restarting etanercept.
In a comparative analysis, relative to the RE-EMBARK patients discontinuing etanercept, similar patients who continued etanercept on the companion EMBARK trial had a longer time to flare (P < .0001) and an 85% lower risk of this outcome.
“There were no new safety signals identified, and as expected, the number of treatment-emergent adverse events dropped during the drug-free period and, interestingly, remained stable over retreatment,” Dr. Van den Bosch noted.
“Temporarily discontinuing etanercept may be an option for some patients with stable inactive nonradiographic axial spondyloarthritis,” he concluded.
The PrevAS trial was financially supported by Pfizer and ReumaNederland. Ms. Rusman declared no relevant conflicts of interest; four coauthors reported financial relationship(s) with Pfizer and other pharmaceutical companies. The RE-EMBARK trial was sponsored by Pfizer. Dr. Van den Bosch disclosed receiving grant/research support from AbbVie, Merck, and UCB, and consulting fees from AbbVie, Bristol-Myers Squibb, Celgene, Eli Lilly, Galapagos, Gilead, Janssen, Novartis, Pfizer, and UCB. Four coauthors reported financial ties to Pfizer and other pharmaceutical companies, and five coauthors were employees and shareholders of Pfizer.
SOURCES: Rusman T et al. Ann Rheum Dis. 2020;79[suppl 1]:72-3; and Van den Bosch F et al. Ann Rheum Dis. 2020;79[suppl 1]:70.
The results from a pair of clinical trials should help to take the guesswork out of starting and stopping the tumor necrosis factor inhibitor etanercept (Enbrel) in patients with nonradiographic axial spondyloarthritis (nr-axSpA). The trials were reported at the annual European Congress of Rheumatology, held online this year due to COVID-19.
Optimal use of etanercept in this disease is still being defined, according to the investigators. Its effects, if any, when given very early in the disease course is unclear, and guidance is conflicting when it comes to stopping the drug after inactive disease is achieved.
In the Dutch randomized controlled PrevAS trial of 80 patients with suspected very early nr-axSpA, initiating etanercept instead of placebo did not significantly improve the odds of achieving a 20% improvement in Assessment of Spondyloarthritis International Society (ASAS 20) response criteria at week 16.
And in the multinational, open-label, phase 4 RE-EMBARK trial, three-quarters of the 119 patients with nr-axSpA who achieved inactive disease on etanercept and stopped the drug experienced a flare within 40 weeks. However, the majority were able to regain disease inactivity after restarting the drug.
Findings in context
“We all have some patients like this [PrevAS population] where we strongly believe they have axial spondyloarthritis but do not fully qualify,” Nigil Haroon MD, PhD, said in an interview. “From a clinical decision-making process, we may diagnose these patients with axial spondyloarthritis, but due to restrictions in access to medications, we have difficulty accessing biologic medications for them. Hence, this study has practical implications.”
“It has already been shown in other, much larger studies that, even in patients who satisfy the criteria of axial spondyloarthritis, those who are MRI and CRP [C-reactive protein] negative are unlikely to respond, so the results are not surprising,” commented Dr. Haroon, who is codirector of the spondylitis program at the University Health Network and associate professor of medicine and rheumatology at the University of Toronto.
Although intended to be a population with suspected very early disease, several of the PrevAS patients would have met ASAS criteria for the disease at baseline, Dr. Haroon cautioned. In addition, the small sample size precluded subgroup analyses.
“The overall conclusion should be, this is a negative study, rather than state there was a trend to better improvement on etanercept. Although there are practical implications, as mentioned, I don’t think this study, with the numbers and the results presented, will change clinical practice,” he said.
The question of stopping biologics in nr-axSpA was previously addressed in the ABILITY-3 randomized trial of adalimumab (Humira), which found that flares were significantly more common with stopping versus continuing the drug and only about half of patients were able to get back in remission by restarting the drug, according to Dr. Haroon.
However, the RE-EMBARK and ABILITY-3 studies differed in both design and patient population, he noted. For example, the mean disease duration was only about 2 years in the former study, compared with 7 years in the latter.
The initial 59% rate of attaining inactive disease on etanercept in RE-EMBARK was “impressive,” Dr. Haroon said, “but as this was an open-label study, higher values are expected.”
“The message in both studies is that stopping biologics completely is not a good idea as the majority of patients, 70%-75%, will relapse within a short period,” he concluded. “However, it should be kept in mind that these [RE-EMBARK] patients received biologic only for a short 24-week period. This study does not answer the question of whether nonradiographic axial spondyloarthritis patients with sustained inactive disease can be taken off biologics abruptly without a taper over time.”
Details of the studies
In the PrevAS trial, Tamara Rusman, a PhD candidate in Rheumatology at the VU University Medical Center Amsterdam and coinvestigators studied patients meeting Calin criteria for inflammatory back pain who had high disease activity plus either HLA-B27 positivity with at least one feature of axial spondyloarthritis or HLA-B27 negativity with two features.
This population is of interest because “most studies have included only patients with nonradiographic axial spondyloarthritis with a positive MRI of the sacroiliac joints and/or an elevated C-reactive protein level,” she noted.
Results showed that, during 16 weeks of treatment, etanercept users had a nonsignficantly higher rate of achieving an ASAS 20 response with etanercept versus placebo users (17% vs. 11%; hazard ratio, 2.1; P = .2). The etanercept group also had a somewhat higher rate of response as defined by the Ankylosing Spondylitis Disease Activity Score CRP (ASDAS-CRP) criterion (25% vs. 13%; hazard ratio, 1.1; P = .8).
“Based on these data, early treatment in inflammatory back pain patients prone to develop axial spondyloarthritis seems not to be useful,” Ms. Rusman concluded. “However, monitoring of these patients should be continued since they remain a risk group for developing axial spondyloarthritis.”
In the RE-EMBARK trial, investigators led by Filip Van den Bosch, MD, PhD, Rheumatology Head-of-Clinic at Ghent (Belgium) University Hospital, started with a cohort of 208 patients with nr-axSpA who were given etanercept and background NSAIDs for 24 weeks.
“Current guidelines do not agree on whether a TNF-blocking agent or another biological DMARD should be tapered once a status of low disease activity or remission is achieved,” he noted.
Overall, 59% of the patients achieved inactive disease (defined as an ASDAS-CRP < 1.3) and discontinued etanercept.
During the next 40 weeks, 24% of these patients maintained inactive disease with only the background NSAID therapy. Among the 75% who experienced a flare, defined as an ASDAS with erythrocyte sedimentation rate (ASDAS-ESR) score of 2.1 or greater, the median time to flare was 16.1 weeks. Fully 62% of this group were able to regain disease inactivity within 12 weeks of restarting etanercept.
In a comparative analysis, relative to the RE-EMBARK patients discontinuing etanercept, similar patients who continued etanercept on the companion EMBARK trial had a longer time to flare (P < .0001) and an 85% lower risk of this outcome.
“There were no new safety signals identified, and as expected, the number of treatment-emergent adverse events dropped during the drug-free period and, interestingly, remained stable over retreatment,” Dr. Van den Bosch noted.
“Temporarily discontinuing etanercept may be an option for some patients with stable inactive nonradiographic axial spondyloarthritis,” he concluded.
The PrevAS trial was financially supported by Pfizer and ReumaNederland. Ms. Rusman declared no relevant conflicts of interest; four coauthors reported financial relationship(s) with Pfizer and other pharmaceutical companies. The RE-EMBARK trial was sponsored by Pfizer. Dr. Van den Bosch disclosed receiving grant/research support from AbbVie, Merck, and UCB, and consulting fees from AbbVie, Bristol-Myers Squibb, Celgene, Eli Lilly, Galapagos, Gilead, Janssen, Novartis, Pfizer, and UCB. Four coauthors reported financial ties to Pfizer and other pharmaceutical companies, and five coauthors were employees and shareholders of Pfizer.
SOURCES: Rusman T et al. Ann Rheum Dis. 2020;79[suppl 1]:72-3; and Van den Bosch F et al. Ann Rheum Dis. 2020;79[suppl 1]:70.
The results from a pair of clinical trials should help to take the guesswork out of starting and stopping the tumor necrosis factor inhibitor etanercept (Enbrel) in patients with nonradiographic axial spondyloarthritis (nr-axSpA). The trials were reported at the annual European Congress of Rheumatology, held online this year due to COVID-19.
Optimal use of etanercept in this disease is still being defined, according to the investigators. Its effects, if any, when given very early in the disease course is unclear, and guidance is conflicting when it comes to stopping the drug after inactive disease is achieved.
In the Dutch randomized controlled PrevAS trial of 80 patients with suspected very early nr-axSpA, initiating etanercept instead of placebo did not significantly improve the odds of achieving a 20% improvement in Assessment of Spondyloarthritis International Society (ASAS 20) response criteria at week 16.
And in the multinational, open-label, phase 4 RE-EMBARK trial, three-quarters of the 119 patients with nr-axSpA who achieved inactive disease on etanercept and stopped the drug experienced a flare within 40 weeks. However, the majority were able to regain disease inactivity after restarting the drug.
Findings in context
“We all have some patients like this [PrevAS population] where we strongly believe they have axial spondyloarthritis but do not fully qualify,” Nigil Haroon MD, PhD, said in an interview. “From a clinical decision-making process, we may diagnose these patients with axial spondyloarthritis, but due to restrictions in access to medications, we have difficulty accessing biologic medications for them. Hence, this study has practical implications.”
“It has already been shown in other, much larger studies that, even in patients who satisfy the criteria of axial spondyloarthritis, those who are MRI and CRP [C-reactive protein] negative are unlikely to respond, so the results are not surprising,” commented Dr. Haroon, who is codirector of the spondylitis program at the University Health Network and associate professor of medicine and rheumatology at the University of Toronto.
Although intended to be a population with suspected very early disease, several of the PrevAS patients would have met ASAS criteria for the disease at baseline, Dr. Haroon cautioned. In addition, the small sample size precluded subgroup analyses.
“The overall conclusion should be, this is a negative study, rather than state there was a trend to better improvement on etanercept. Although there are practical implications, as mentioned, I don’t think this study, with the numbers and the results presented, will change clinical practice,” he said.
The question of stopping biologics in nr-axSpA was previously addressed in the ABILITY-3 randomized trial of adalimumab (Humira), which found that flares were significantly more common with stopping versus continuing the drug and only about half of patients were able to get back in remission by restarting the drug, according to Dr. Haroon.
However, the RE-EMBARK and ABILITY-3 studies differed in both design and patient population, he noted. For example, the mean disease duration was only about 2 years in the former study, compared with 7 years in the latter.
The initial 59% rate of attaining inactive disease on etanercept in RE-EMBARK was “impressive,” Dr. Haroon said, “but as this was an open-label study, higher values are expected.”
“The message in both studies is that stopping biologics completely is not a good idea as the majority of patients, 70%-75%, will relapse within a short period,” he concluded. “However, it should be kept in mind that these [RE-EMBARK] patients received biologic only for a short 24-week period. This study does not answer the question of whether nonradiographic axial spondyloarthritis patients with sustained inactive disease can be taken off biologics abruptly without a taper over time.”
Details of the studies
In the PrevAS trial, Tamara Rusman, a PhD candidate in Rheumatology at the VU University Medical Center Amsterdam and coinvestigators studied patients meeting Calin criteria for inflammatory back pain who had high disease activity plus either HLA-B27 positivity with at least one feature of axial spondyloarthritis or HLA-B27 negativity with two features.
This population is of interest because “most studies have included only patients with nonradiographic axial spondyloarthritis with a positive MRI of the sacroiliac joints and/or an elevated C-reactive protein level,” she noted.
Results showed that, during 16 weeks of treatment, etanercept users had a nonsignficantly higher rate of achieving an ASAS 20 response with etanercept versus placebo users (17% vs. 11%; hazard ratio, 2.1; P = .2). The etanercept group also had a somewhat higher rate of response as defined by the Ankylosing Spondylitis Disease Activity Score CRP (ASDAS-CRP) criterion (25% vs. 13%; hazard ratio, 1.1; P = .8).
“Based on these data, early treatment in inflammatory back pain patients prone to develop axial spondyloarthritis seems not to be useful,” Ms. Rusman concluded. “However, monitoring of these patients should be continued since they remain a risk group for developing axial spondyloarthritis.”
In the RE-EMBARK trial, investigators led by Filip Van den Bosch, MD, PhD, Rheumatology Head-of-Clinic at Ghent (Belgium) University Hospital, started with a cohort of 208 patients with nr-axSpA who were given etanercept and background NSAIDs for 24 weeks.
“Current guidelines do not agree on whether a TNF-blocking agent or another biological DMARD should be tapered once a status of low disease activity or remission is achieved,” he noted.
Overall, 59% of the patients achieved inactive disease (defined as an ASDAS-CRP < 1.3) and discontinued etanercept.
During the next 40 weeks, 24% of these patients maintained inactive disease with only the background NSAID therapy. Among the 75% who experienced a flare, defined as an ASDAS with erythrocyte sedimentation rate (ASDAS-ESR) score of 2.1 or greater, the median time to flare was 16.1 weeks. Fully 62% of this group were able to regain disease inactivity within 12 weeks of restarting etanercept.
In a comparative analysis, relative to the RE-EMBARK patients discontinuing etanercept, similar patients who continued etanercept on the companion EMBARK trial had a longer time to flare (P < .0001) and an 85% lower risk of this outcome.
“There were no new safety signals identified, and as expected, the number of treatment-emergent adverse events dropped during the drug-free period and, interestingly, remained stable over retreatment,” Dr. Van den Bosch noted.
“Temporarily discontinuing etanercept may be an option for some patients with stable inactive nonradiographic axial spondyloarthritis,” he concluded.
The PrevAS trial was financially supported by Pfizer and ReumaNederland. Ms. Rusman declared no relevant conflicts of interest; four coauthors reported financial relationship(s) with Pfizer and other pharmaceutical companies. The RE-EMBARK trial was sponsored by Pfizer. Dr. Van den Bosch disclosed receiving grant/research support from AbbVie, Merck, and UCB, and consulting fees from AbbVie, Bristol-Myers Squibb, Celgene, Eli Lilly, Galapagos, Gilead, Janssen, Novartis, Pfizer, and UCB. Four coauthors reported financial ties to Pfizer and other pharmaceutical companies, and five coauthors were employees and shareholders of Pfizer.
SOURCES: Rusman T et al. Ann Rheum Dis. 2020;79[suppl 1]:72-3; and Van den Bosch F et al. Ann Rheum Dis. 2020;79[suppl 1]:70.
FROM THE EULAR 2020 E-CONGRESS
Key clinical point: In nonradiographic axial spondyloarthritis (nr-axSpA), etanercept does not have significant benefit by 16 weeks when started in very early disease, and the majority of patients who achieved inactive disease on the drug and then stopped it experienced a flare within 40 weeks.
Major finding: Patients with suspected very early disease who took etanercept did not have a significantly greater rate of achieving a 20% improvement in Assessment of Spondyloarthritis International Society (ASAS 20) response criteria at week 16 than did those taking placebo (17% vs. 11%; hazard ratio, 2.1; P = .2). In a separate trial, 75% of patients who achieved inactive disease with etanercept and then stopped the drug had a flare within 40 weeks, but 62% of this group were able to regain disease inactivity within 12 weeks of restarting etanercept.
Study details: A randomized, placebo-controlled PrevAS trial involved 80 patients with suspected very early nr-axSpA who started either etanercept or placebo, and the multicenter, open-label, phase 4 RE-EMBARK trial involved 119 patients achieving inactive nr-axSpA on etanercept.
Disclosures: The PrevAS trial was financially supported by Pfizer and ReumaNederland. Ms. Rusman declared no relevant conflicts of interest; four coauthors reported financial relationship(s) with Pfizer and other pharmaceutical companies. The RE-EMBARK trial was sponsored by Pfizer. Dr. Van den Bosch disclosed receiving grant/research support from AbbVie, Merck, and UCB and consulting fees from AbbVie, Bristol-Myers Squibb, Celgene, Eli Lilly, Galapagos, Gilead, Janssen, Novartis, Pfizer, and UCB. Four coauthors reported financial ties to Pfizer and other pharmaceutical companies, and five coauthors were employees and shareholders of Pfizer.
Sources: Rusman T et al. Ann Rheum Dis. 2020;79[suppl 1]:72-3; and Van den Bosch F et al. Ann Rheum Dis. 2020;79[suppl 1]:70.
No link seen between methotrexate, interstitial lung disease in RA
Patients with rheumatoid arthritis (RA) have an elevated risk of interstitial lung disease (ILD), but methotrexate does not accentuate that risk and may in fact be protective, new data show. These were among key findings of a pair of studies reported at the annual European Congress of Rheumatology, held online this year due to COVID-19.
Although a guideline-recommended cornerstone in the management of RA, methotrexate has been associated with both hypersensitivity pneumonitis and diffuse lung disease. However, its involvement in the development of ILD among patients with RA is unclear.
A Danish study of more than 30,000 RA patients reported at the congress found that their risk of ILD was about three to five times that of the general population. However, risk did not differ significantly whether they had filled a methotrexate prescription or not.
In addition, a multinational case-control study of more than 1,000 RA patients also reported at the congress found that, compared with never-users of methotrexate, ever-users actually had a 59% lower likelihood of developing ILD.
However, both studies were limited by their retrospective design, Elizabeth R. Volkmann, MD, codirector of the connective tissue disease–related interstitial lung disease program at the University of California, Los Angeles, cautioned in an interview. Hence, there was likely systematic bias and confounding.
“I would interpret the conclusions of both studies with caution,” she maintained. “To understand how a particular intervention, such as methotrexate use, affects the outcome of ILD development, a prospective design is needed, which adequately adjusts for known ILD risk factors, such as male sex and smoking.”
As to whether the new findings are practice changing and how they might affect patient counseling, “the answers to these questions are not straightforward and depend on other patient-related factors,” according to Dr. Volkmann.
Danish nationwide study
René Cordtz, MD, a clinical assistant at the Center for Rheumatology and Spine Diseases, Rigshospitalet‐Gentofte, Copenhagen, and colleagues conducted a nationwide population-based cohort study using registry data from 1997 to 2015 to assess lung disease among patients with RA by prescriptions filled.
Results based on 30,512 RA patients showed that, compared with peers filling no methotrexate prescriptions, patients filling at least one did not have a significantly elevated risk of ILD at either 1 year of follow-up (hazard ratio, 1.03) or 5 years of follow-up (HR, 1.00). (Findings were similar for sulfasalazine, with respective nonsignificant HRs of 0.88 and 1.14.)
In addition, patients with RA had a similarly sharply elevated 5-year risk of ILD relative to the general population regardless of whether they had filled neither methotrexate nor sulfasalazine prescriptions (standardized incidence ratio, 3.38) or had filled prescriptions for methotrexate only (SIR, 3.63), sulfasalazine only (SIR, 4.12), or both (SIR, 5.45).
“RA patients have an increased risk of ILD, compared to the general population, which was not surprising, but very importantly, that risk was not further exacerbated in those treated with methotrexate,” Dr. Cordtz concluded. “We do acknowledge that purchasing your medicine is different from taking your medicine, which is why we found it extra reassuring that when requiring at least two methotrexate prescriptions to be considered exposed, it did not change our results.”
Multinational study
Pierre-Antoine Juge, MD, a rheumatologist at Bichat-Claude Bernard Hospital, Paris, and colleagues performed a case-control study among 482 RA patients with ILD and 741 RA patients without ILD in three cohorts: a French discovery cohort, a multinational (Brazilian, Italian, Mexican, United Kingdom, and United States) replication cohort, and a combined cohort. Those with methotrexate hypersensitivity pneumonitis were excluded.
Results showed that relative to peers without ILD, patients with ILD had a lower prevalence of ever having used methotrexate and had received a lower cumulative methotrexate dose, findings that were consistent across all three cohorts.
Methotrexate ever-use was associated with a significantly lower adjusted likelihood of ILD in the discovery cohort (odds ratio, 0.46), the replication cohort (OR, 0.38), and the combined cohort (OR, 0.41). Furthermore, ever-users were less commonly represented among patients with ILD regardless of chest high-resolution CT pattern (usual interstitial pneumonia pattern vs. not).
Finally, methotrexate use appeared to delay the adjusted time to onset of ILD by 3.5 years in the discovery cohort (P = .001), by 3.2 years in the replication cohort (P < .0001), and by 3.5 years in the combined cohort (P < .0001).
“Outside of methotrexate hypersensitivity pneumonitis, methotrexate was not a risk factor for RA-associated ILD in our study. We observed an inverse relationship that was similar whatever the high-resolution CT pattern,” Dr. Juge commented. “But this possible protective effect should be confirmed through a dedicated prospective, randomized, controlled trial.”
“Methotrexate should not be considered as a causal factor for RA-associated ILD, and its [discontinuation] should be discussed through a multidisciplinary discussion,” he recommended. In addition, “this study does not investigate the impact of methotrexate use on RA-associated ILD prognosis.”
The Danish study did not receive any specific funding, and none of its authors reported having any financial disclosures. The multinational study did not receive any specific funding. Dr. Juge disclosed that he had no relevant conflicts of interest, but many of his coauthors reported financial relationships with industry. Dr. Volkmann disclosed consulting for Boehringer Ingelheim and Forbius, and receiving grant support from Forbius and Corbus.
SOURCES: Cordtz R et al. Ann Rheum Dis. 2020;79[suppl 1]:147-8, Abstract OP0232; Juge P-A et al. Ann Rheum Dis. 2020;79[suppl 1]:25, Abstract OP0236.
Patients with rheumatoid arthritis (RA) have an elevated risk of interstitial lung disease (ILD), but methotrexate does not accentuate that risk and may in fact be protective, new data show. These were among key findings of a pair of studies reported at the annual European Congress of Rheumatology, held online this year due to COVID-19.
Although a guideline-recommended cornerstone in the management of RA, methotrexate has been associated with both hypersensitivity pneumonitis and diffuse lung disease. However, its involvement in the development of ILD among patients with RA is unclear.
A Danish study of more than 30,000 RA patients reported at the congress found that their risk of ILD was about three to five times that of the general population. However, risk did not differ significantly whether they had filled a methotrexate prescription or not.
In addition, a multinational case-control study of more than 1,000 RA patients also reported at the congress found that, compared with never-users of methotrexate, ever-users actually had a 59% lower likelihood of developing ILD.
However, both studies were limited by their retrospective design, Elizabeth R. Volkmann, MD, codirector of the connective tissue disease–related interstitial lung disease program at the University of California, Los Angeles, cautioned in an interview. Hence, there was likely systematic bias and confounding.
“I would interpret the conclusions of both studies with caution,” she maintained. “To understand how a particular intervention, such as methotrexate use, affects the outcome of ILD development, a prospective design is needed, which adequately adjusts for known ILD risk factors, such as male sex and smoking.”
As to whether the new findings are practice changing and how they might affect patient counseling, “the answers to these questions are not straightforward and depend on other patient-related factors,” according to Dr. Volkmann.
Danish nationwide study
René Cordtz, MD, a clinical assistant at the Center for Rheumatology and Spine Diseases, Rigshospitalet‐Gentofte, Copenhagen, and colleagues conducted a nationwide population-based cohort study using registry data from 1997 to 2015 to assess lung disease among patients with RA by prescriptions filled.
Results based on 30,512 RA patients showed that, compared with peers filling no methotrexate prescriptions, patients filling at least one did not have a significantly elevated risk of ILD at either 1 year of follow-up (hazard ratio, 1.03) or 5 years of follow-up (HR, 1.00). (Findings were similar for sulfasalazine, with respective nonsignificant HRs of 0.88 and 1.14.)
In addition, patients with RA had a similarly sharply elevated 5-year risk of ILD relative to the general population regardless of whether they had filled neither methotrexate nor sulfasalazine prescriptions (standardized incidence ratio, 3.38) or had filled prescriptions for methotrexate only (SIR, 3.63), sulfasalazine only (SIR, 4.12), or both (SIR, 5.45).
“RA patients have an increased risk of ILD, compared to the general population, which was not surprising, but very importantly, that risk was not further exacerbated in those treated with methotrexate,” Dr. Cordtz concluded. “We do acknowledge that purchasing your medicine is different from taking your medicine, which is why we found it extra reassuring that when requiring at least two methotrexate prescriptions to be considered exposed, it did not change our results.”
Multinational study
Pierre-Antoine Juge, MD, a rheumatologist at Bichat-Claude Bernard Hospital, Paris, and colleagues performed a case-control study among 482 RA patients with ILD and 741 RA patients without ILD in three cohorts: a French discovery cohort, a multinational (Brazilian, Italian, Mexican, United Kingdom, and United States) replication cohort, and a combined cohort. Those with methotrexate hypersensitivity pneumonitis were excluded.
Results showed that relative to peers without ILD, patients with ILD had a lower prevalence of ever having used methotrexate and had received a lower cumulative methotrexate dose, findings that were consistent across all three cohorts.
Methotrexate ever-use was associated with a significantly lower adjusted likelihood of ILD in the discovery cohort (odds ratio, 0.46), the replication cohort (OR, 0.38), and the combined cohort (OR, 0.41). Furthermore, ever-users were less commonly represented among patients with ILD regardless of chest high-resolution CT pattern (usual interstitial pneumonia pattern vs. not).
Finally, methotrexate use appeared to delay the adjusted time to onset of ILD by 3.5 years in the discovery cohort (P = .001), by 3.2 years in the replication cohort (P < .0001), and by 3.5 years in the combined cohort (P < .0001).
“Outside of methotrexate hypersensitivity pneumonitis, methotrexate was not a risk factor for RA-associated ILD in our study. We observed an inverse relationship that was similar whatever the high-resolution CT pattern,” Dr. Juge commented. “But this possible protective effect should be confirmed through a dedicated prospective, randomized, controlled trial.”
“Methotrexate should not be considered as a causal factor for RA-associated ILD, and its [discontinuation] should be discussed through a multidisciplinary discussion,” he recommended. In addition, “this study does not investigate the impact of methotrexate use on RA-associated ILD prognosis.”
The Danish study did not receive any specific funding, and none of its authors reported having any financial disclosures. The multinational study did not receive any specific funding. Dr. Juge disclosed that he had no relevant conflicts of interest, but many of his coauthors reported financial relationships with industry. Dr. Volkmann disclosed consulting for Boehringer Ingelheim and Forbius, and receiving grant support from Forbius and Corbus.
SOURCES: Cordtz R et al. Ann Rheum Dis. 2020;79[suppl 1]:147-8, Abstract OP0232; Juge P-A et al. Ann Rheum Dis. 2020;79[suppl 1]:25, Abstract OP0236.
Patients with rheumatoid arthritis (RA) have an elevated risk of interstitial lung disease (ILD), but methotrexate does not accentuate that risk and may in fact be protective, new data show. These were among key findings of a pair of studies reported at the annual European Congress of Rheumatology, held online this year due to COVID-19.
Although a guideline-recommended cornerstone in the management of RA, methotrexate has been associated with both hypersensitivity pneumonitis and diffuse lung disease. However, its involvement in the development of ILD among patients with RA is unclear.
A Danish study of more than 30,000 RA patients reported at the congress found that their risk of ILD was about three to five times that of the general population. However, risk did not differ significantly whether they had filled a methotrexate prescription or not.
In addition, a multinational case-control study of more than 1,000 RA patients also reported at the congress found that, compared with never-users of methotrexate, ever-users actually had a 59% lower likelihood of developing ILD.
However, both studies were limited by their retrospective design, Elizabeth R. Volkmann, MD, codirector of the connective tissue disease–related interstitial lung disease program at the University of California, Los Angeles, cautioned in an interview. Hence, there was likely systematic bias and confounding.
“I would interpret the conclusions of both studies with caution,” she maintained. “To understand how a particular intervention, such as methotrexate use, affects the outcome of ILD development, a prospective design is needed, which adequately adjusts for known ILD risk factors, such as male sex and smoking.”
As to whether the new findings are practice changing and how they might affect patient counseling, “the answers to these questions are not straightforward and depend on other patient-related factors,” according to Dr. Volkmann.
Danish nationwide study
René Cordtz, MD, a clinical assistant at the Center for Rheumatology and Spine Diseases, Rigshospitalet‐Gentofte, Copenhagen, and colleagues conducted a nationwide population-based cohort study using registry data from 1997 to 2015 to assess lung disease among patients with RA by prescriptions filled.
Results based on 30,512 RA patients showed that, compared with peers filling no methotrexate prescriptions, patients filling at least one did not have a significantly elevated risk of ILD at either 1 year of follow-up (hazard ratio, 1.03) or 5 years of follow-up (HR, 1.00). (Findings were similar for sulfasalazine, with respective nonsignificant HRs of 0.88 and 1.14.)
In addition, patients with RA had a similarly sharply elevated 5-year risk of ILD relative to the general population regardless of whether they had filled neither methotrexate nor sulfasalazine prescriptions (standardized incidence ratio, 3.38) or had filled prescriptions for methotrexate only (SIR, 3.63), sulfasalazine only (SIR, 4.12), or both (SIR, 5.45).
“RA patients have an increased risk of ILD, compared to the general population, which was not surprising, but very importantly, that risk was not further exacerbated in those treated with methotrexate,” Dr. Cordtz concluded. “We do acknowledge that purchasing your medicine is different from taking your medicine, which is why we found it extra reassuring that when requiring at least two methotrexate prescriptions to be considered exposed, it did not change our results.”
Multinational study
Pierre-Antoine Juge, MD, a rheumatologist at Bichat-Claude Bernard Hospital, Paris, and colleagues performed a case-control study among 482 RA patients with ILD and 741 RA patients without ILD in three cohorts: a French discovery cohort, a multinational (Brazilian, Italian, Mexican, United Kingdom, and United States) replication cohort, and a combined cohort. Those with methotrexate hypersensitivity pneumonitis were excluded.
Results showed that relative to peers without ILD, patients with ILD had a lower prevalence of ever having used methotrexate and had received a lower cumulative methotrexate dose, findings that were consistent across all three cohorts.
Methotrexate ever-use was associated with a significantly lower adjusted likelihood of ILD in the discovery cohort (odds ratio, 0.46), the replication cohort (OR, 0.38), and the combined cohort (OR, 0.41). Furthermore, ever-users were less commonly represented among patients with ILD regardless of chest high-resolution CT pattern (usual interstitial pneumonia pattern vs. not).
Finally, methotrexate use appeared to delay the adjusted time to onset of ILD by 3.5 years in the discovery cohort (P = .001), by 3.2 years in the replication cohort (P < .0001), and by 3.5 years in the combined cohort (P < .0001).
“Outside of methotrexate hypersensitivity pneumonitis, methotrexate was not a risk factor for RA-associated ILD in our study. We observed an inverse relationship that was similar whatever the high-resolution CT pattern,” Dr. Juge commented. “But this possible protective effect should be confirmed through a dedicated prospective, randomized, controlled trial.”
“Methotrexate should not be considered as a causal factor for RA-associated ILD, and its [discontinuation] should be discussed through a multidisciplinary discussion,” he recommended. In addition, “this study does not investigate the impact of methotrexate use on RA-associated ILD prognosis.”
The Danish study did not receive any specific funding, and none of its authors reported having any financial disclosures. The multinational study did not receive any specific funding. Dr. Juge disclosed that he had no relevant conflicts of interest, but many of his coauthors reported financial relationships with industry. Dr. Volkmann disclosed consulting for Boehringer Ingelheim and Forbius, and receiving grant support from Forbius and Corbus.
SOURCES: Cordtz R et al. Ann Rheum Dis. 2020;79[suppl 1]:147-8, Abstract OP0232; Juge P-A et al. Ann Rheum Dis. 2020;79[suppl 1]:25, Abstract OP0236.
FROM THE EULAR 2020 E-CONGRESS