User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'main-prefix')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
div[contains(@class, 'view-medstat-quiz-listing-panes')]
div[contains(@class, 'pane-article-sidebar-latest-news')]
COVID-19: U.S. sets new weekly high in children
the American Academy of Pediatrics announced Nov. 2.
For the week, over 61,000 cases were reported in children, bringing the number of COVID-19 cases for the month of October to nearly 200,000 and the total since the start of the pandemic to over 853,000, the AAP and the Children’s Hospital Association said in their weekly report.
“These numbers reflect a disturbing increase in cases throughout most of the United States in all populations, especially among young adults,” Yvonne Maldonado, MD, chair of the AAP Committee on Infectious Diseases, said in a separate statement. “We are entering a heightened wave of infections around the country. We would encourage family holiday gatherings to be avoided if possible, especially if there are high-risk individuals in the household.”
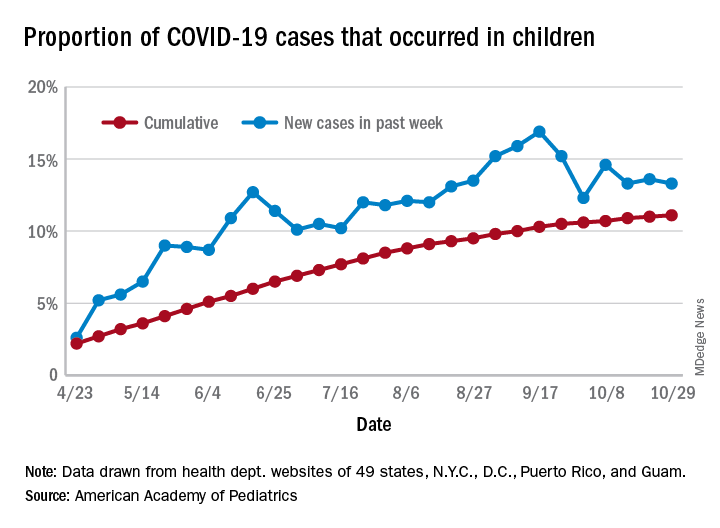
For the week ending Oct. 29, children represented 13.3% of all cases, possibly constituting a minitrend of stability over the past 3 weeks. For the full length of the pandemic, 11.1% of all COVID-19 cases have occurred in children, although severe illness is much less common: 1.7% of all hospitalizations (data from 24 states and New York City) and 0.06% of all deaths (data from 42 states and New York City), the AAP and CHA report said.
Other data show that 1,134 per 100,000 children in the United States have been infected by the coronavirus, up from 1,053 the previous week, with state rates ranging from 221 per 100,000 in Vermont to 3,321 in North Dakota. In Wyoming, 25.5% of all COVID-19 cases have occurred in children, the highest of any state, while New Jersey has the lowest rate at 4.9%, the AAP/CHA report showed.
In the 10 states making testing data available, children represent the lowest percentage of tests in Iowa (5.0%) and the highest in Indiana (16.9%). Iowa, however, has the highest positivity rate for children at 14.6%, along with Nevada, while West Virginia has the lowest at 3.6%, the AAP and CHA said in the report.
These numbers, however, may not be telling the whole story. “The number of reported COVID-19 cases in children is likely an undercount because children’s symptoms are often mild and they may not be tested for every illness,” the AAP said in its statement.
“We urge policy makers to listen to doctors and public health experts rather than level baseless accusations against them. Physicians, nurses and other health care professionals have put their lives on the line to protect our communities. We can all do our part to protect them, and our communities, by wearing masks, practicing physical distancing, and getting our flu immunizations,” AAP President Sally Goza, MD, said in the AAP statement.
the American Academy of Pediatrics announced Nov. 2.

For the week, over 61,000 cases were reported in children, bringing the number of COVID-19 cases for the month of October to nearly 200,000 and the total since the start of the pandemic to over 853,000, the AAP and the Children’s Hospital Association said in their weekly report.
“These numbers reflect a disturbing increase in cases throughout most of the United States in all populations, especially among young adults,” Yvonne Maldonado, MD, chair of the AAP Committee on Infectious Diseases, said in a separate statement. “We are entering a heightened wave of infections around the country. We would encourage family holiday gatherings to be avoided if possible, especially if there are high-risk individuals in the household.”
For the week ending Oct. 29, children represented 13.3% of all cases, possibly constituting a minitrend of stability over the past 3 weeks. For the full length of the pandemic, 11.1% of all COVID-19 cases have occurred in children, although severe illness is much less common: 1.7% of all hospitalizations (data from 24 states and New York City) and 0.06% of all deaths (data from 42 states and New York City), the AAP and CHA report said.
Other data show that 1,134 per 100,000 children in the United States have been infected by the coronavirus, up from 1,053 the previous week, with state rates ranging from 221 per 100,000 in Vermont to 3,321 in North Dakota. In Wyoming, 25.5% of all COVID-19 cases have occurred in children, the highest of any state, while New Jersey has the lowest rate at 4.9%, the AAP/CHA report showed.
In the 10 states making testing data available, children represent the lowest percentage of tests in Iowa (5.0%) and the highest in Indiana (16.9%). Iowa, however, has the highest positivity rate for children at 14.6%, along with Nevada, while West Virginia has the lowest at 3.6%, the AAP and CHA said in the report.
These numbers, however, may not be telling the whole story. “The number of reported COVID-19 cases in children is likely an undercount because children’s symptoms are often mild and they may not be tested for every illness,” the AAP said in its statement.
“We urge policy makers to listen to doctors and public health experts rather than level baseless accusations against them. Physicians, nurses and other health care professionals have put their lives on the line to protect our communities. We can all do our part to protect them, and our communities, by wearing masks, practicing physical distancing, and getting our flu immunizations,” AAP President Sally Goza, MD, said in the AAP statement.
the American Academy of Pediatrics announced Nov. 2.

For the week, over 61,000 cases were reported in children, bringing the number of COVID-19 cases for the month of October to nearly 200,000 and the total since the start of the pandemic to over 853,000, the AAP and the Children’s Hospital Association said in their weekly report.
“These numbers reflect a disturbing increase in cases throughout most of the United States in all populations, especially among young adults,” Yvonne Maldonado, MD, chair of the AAP Committee on Infectious Diseases, said in a separate statement. “We are entering a heightened wave of infections around the country. We would encourage family holiday gatherings to be avoided if possible, especially if there are high-risk individuals in the household.”
For the week ending Oct. 29, children represented 13.3% of all cases, possibly constituting a minitrend of stability over the past 3 weeks. For the full length of the pandemic, 11.1% of all COVID-19 cases have occurred in children, although severe illness is much less common: 1.7% of all hospitalizations (data from 24 states and New York City) and 0.06% of all deaths (data from 42 states and New York City), the AAP and CHA report said.
Other data show that 1,134 per 100,000 children in the United States have been infected by the coronavirus, up from 1,053 the previous week, with state rates ranging from 221 per 100,000 in Vermont to 3,321 in North Dakota. In Wyoming, 25.5% of all COVID-19 cases have occurred in children, the highest of any state, while New Jersey has the lowest rate at 4.9%, the AAP/CHA report showed.
In the 10 states making testing data available, children represent the lowest percentage of tests in Iowa (5.0%) and the highest in Indiana (16.9%). Iowa, however, has the highest positivity rate for children at 14.6%, along with Nevada, while West Virginia has the lowest at 3.6%, the AAP and CHA said in the report.
These numbers, however, may not be telling the whole story. “The number of reported COVID-19 cases in children is likely an undercount because children’s symptoms are often mild and they may not be tested for every illness,” the AAP said in its statement.
“We urge policy makers to listen to doctors and public health experts rather than level baseless accusations against them. Physicians, nurses and other health care professionals have put their lives on the line to protect our communities. We can all do our part to protect them, and our communities, by wearing masks, practicing physical distancing, and getting our flu immunizations,” AAP President Sally Goza, MD, said in the AAP statement.
HF an added risk in COVID-19, regardless of ejection fraction
People with a history of heart failure – no matter the type – face more complications and death than their peers without HF once hospitalized with COVID-19, a new observational study shows.
A history of HF was associated with a near doubling risk of in-hospital mortality and ICU care and more than a tripling risk of mechanical ventilation despite adjustment for 18 factors including race, obesity, diabetes, previous treatment with renin-angiotensin-aldosterone system (RAAS) inhibitors, and severity of illness.
Adverse outcomes were high regardless of whether patients had HF with a preserved, mid-range, or reduced left ventricular ejection fraction (HFpEF/HFmrEF/HFrEF).
“That for me was the real zinger,” senior author Anuradha Lala, MD, said in an interview . “Because as clinicians, oftentimes, and wrongly so, we think this person has preserved ejection fraction, so they’re not needing my heart failure expertise as much as someone with heart failure with reduced ejection fraction.”
In the peak of the pandemic, that may have meant triaging patients with HFpEF to a regular floor, whereas those with HFrEF were seen by the specialist team.
“What this alerted me to is to take heart failure as a diagnosis very seriously, regardless of ejection fraction, and that is very much in line with all of the emerging data about heart failure with preserved ejection fraction,” said Dr. Lala, from the Icahn School of Medicine at Mount Sinai, New York.
“Now when I see patients in the clinic, I incorporate part of our visit to talking about what they are doing to prevent COVID, which I really wasn’t doing before. It was like ‘Oh yeah, what crazy times we’re dealing with’ and then addressing their heart failure as I normally would,” she said. “But now, interwoven into every visit is: Are you wearing a mask, what’s your social distancing policy, who are you living with at home, has anyone at home or who you’ve interacted with been sick? I’m asking those questions just as a knee-jerk reaction for these patients because I know the repercussions. We have to keep in mind these are observational studies, so I can’t prove causality but these are observations that are, nonetheless, quite robust.”
Although cardiovascular disease, including HF, is recognized as a risk factor for worse outcomes in COVID-19 patients, data are sparse on the clinical course and prognosis of patients with preexisting HF.
“I would have expected that there would have been a gradation of risk from the people with very low ejection fractions up into the normal range, but here it didn’t seem to matter at all. So that’s an important point that bad outcomes were independent of ejection fraction,” commented Lee Goldberg, MD, professor of medicine and chief of advanced heart failure and cardiac transplant at the University of Pennsylvania, Philadelphia.
The study also validated that there is no association between use of RAAS inhibitors and bad outcomes in patients with COVID-19, he said.
Although this has been demonstrated in several studies, concerns were raised early in the pandemic that ACE inhibitors and angiotensin receptor blockers could facilitate infection with SARS-CoV-2 and increase the risk of severe or lethal COVID-19.
“For most clinicians that question has been put to bed, but we’re still getting patients that will ask during office visits ‘Is it safe for me to stay on?’ They still have that doubt [about] ‘Are we doing the right thing?’ ” Dr. Goldberg said.
“We can reassure them now. A lot of us are able to say there’s nothing to that, we’re very clear about this, stay on the meds. If anything, there’s data that suggest actually it may be better to be on an ACE inhibitor; that the hospitalizations were shorter and the outcomes were a little bit better.”
For the current study, published online Oct. 28 in the Journal of the American College of Cardiology, the investigators analyzed 6,439 patients admitted for COVID-19 at one of five Mount Sinai Health System hospitals in New York between Feb. 27 and June 26. Their mean age was 65.3 years, 45% were women, and one-third were treated with RAAS inhibitors before admission.
Using ICD-9/10 codes and individual chart review, HF was identified in 422 patients (6.6%), of which 250 patients had HFpEF (≥50%), 44 had HFmrEF (41%-49%), and 128 had HFrEF (≤40%).
Patients with HFpEF were older, more frequently women with a higher body mass index and history of lung disease than patients with HFrEF, whereas those with HFmrEF fell in between.
The HFpEF group was also treated with hydroxychloroquine or macrolides and noninvasive ventilation more frequently than the other two groups, whereas antiplatelet and neurohormonal therapies were more common in the HFrEF group.
Patients with a history of HF had significantly longer hospital stays than those without HF (8 days vs. 6 days), increased need for intubation (22.8% vs. 11.9%) and ICU care (23.2% vs. 16.6%), and worse in-hospital mortality (40% vs. 24.9%).
After multivariable regression adjustment, HF persisted as an independent risk factor for ICU care (odds ratio, 1.71; 95% CI, 1.25-2.34), intubation and mechanical ventilation (OR, 3.64; 95% CI, 2.56-5.16), and in-hospital mortality (OR, 1.88; 95% CI, 1.27-2.78).
“I knew to expect higher rates of adverse outcomes but I didn’t expect it to be nearly a twofold increase,” Dr. Lala said. “I thought that was pretty powerful.”
No significant differences were seen across LVEF categories in length of stay, need for ICU care, intubation and mechanical ventilation, acute kidney injury, shock, thromboembolic events, arrhythmias, or 30-day readmission rates.
However, cardiogenic shock (7.8% vs. 2.3% vs. 2%) and HF-related causes for 30-day readmissions (47.1% vs. 0% vs. 8.6%) were significantly higher in patients with HFrEF than in those with HFmrEF or HFpEF.
Also, mortality was lower in those with HFmrEF (22.7%) than with HFrEF (38.3%) and HFpEF (44%). The group was small but the “results suggested that patients with HFmrEF could have a better prognosis, because they can represent a distinct and more favorable HF phenotype,” the authors wrote.
The statistical testing didn’t show much difference and the patient numbers were very small, noted Dr. Goldberg. “So they might be overreaching a little bit there.”
“To me, the take-home message is that just having the phenotype of heart failure, regardless of EF, is associated with bad outcomes and we need to be vigilant on two fronts,” he said. “We really need to be doing prevention in the folks with heart failure because if they get COVID their outcomes are not going to be as good. Second, as clinicians, if we see a patient presenting with COVID who has a history of heart failure we may want to be much more vigilant with that individual than we might otherwise be. So I think there’s something to be said for kind of risk-stratifying people in that way.”
Dr. Goldberg pointed out that the study had many “amazing strengths,” including a large, racially diverse population, direct chart review to identify HF patients, and capturing a patient’s specific HF phenotype.
Weaknesses are that it was a single-center study, so the biases of how these patients were treated are not easily controlled for, he said. “We also don’t know when the hospital system was very strained as they were making some decisions: Were the older patients who had advanced heart and lung disease ultimately less aggressively treated because they felt they wouldn’t survive?”
Dr. Lala has received personal fees from Zoll, outside the submitted work. Dr. Goldberg reported research funding with Respicardia and consulting fees from Abbott.
This article first appeared on Medscape.com.
People with a history of heart failure – no matter the type – face more complications and death than their peers without HF once hospitalized with COVID-19, a new observational study shows.
A history of HF was associated with a near doubling risk of in-hospital mortality and ICU care and more than a tripling risk of mechanical ventilation despite adjustment for 18 factors including race, obesity, diabetes, previous treatment with renin-angiotensin-aldosterone system (RAAS) inhibitors, and severity of illness.
Adverse outcomes were high regardless of whether patients had HF with a preserved, mid-range, or reduced left ventricular ejection fraction (HFpEF/HFmrEF/HFrEF).
“That for me was the real zinger,” senior author Anuradha Lala, MD, said in an interview . “Because as clinicians, oftentimes, and wrongly so, we think this person has preserved ejection fraction, so they’re not needing my heart failure expertise as much as someone with heart failure with reduced ejection fraction.”
In the peak of the pandemic, that may have meant triaging patients with HFpEF to a regular floor, whereas those with HFrEF were seen by the specialist team.
“What this alerted me to is to take heart failure as a diagnosis very seriously, regardless of ejection fraction, and that is very much in line with all of the emerging data about heart failure with preserved ejection fraction,” said Dr. Lala, from the Icahn School of Medicine at Mount Sinai, New York.
“Now when I see patients in the clinic, I incorporate part of our visit to talking about what they are doing to prevent COVID, which I really wasn’t doing before. It was like ‘Oh yeah, what crazy times we’re dealing with’ and then addressing their heart failure as I normally would,” she said. “But now, interwoven into every visit is: Are you wearing a mask, what’s your social distancing policy, who are you living with at home, has anyone at home or who you’ve interacted with been sick? I’m asking those questions just as a knee-jerk reaction for these patients because I know the repercussions. We have to keep in mind these are observational studies, so I can’t prove causality but these are observations that are, nonetheless, quite robust.”
Although cardiovascular disease, including HF, is recognized as a risk factor for worse outcomes in COVID-19 patients, data are sparse on the clinical course and prognosis of patients with preexisting HF.
“I would have expected that there would have been a gradation of risk from the people with very low ejection fractions up into the normal range, but here it didn’t seem to matter at all. So that’s an important point that bad outcomes were independent of ejection fraction,” commented Lee Goldberg, MD, professor of medicine and chief of advanced heart failure and cardiac transplant at the University of Pennsylvania, Philadelphia.
The study also validated that there is no association between use of RAAS inhibitors and bad outcomes in patients with COVID-19, he said.
Although this has been demonstrated in several studies, concerns were raised early in the pandemic that ACE inhibitors and angiotensin receptor blockers could facilitate infection with SARS-CoV-2 and increase the risk of severe or lethal COVID-19.
“For most clinicians that question has been put to bed, but we’re still getting patients that will ask during office visits ‘Is it safe for me to stay on?’ They still have that doubt [about] ‘Are we doing the right thing?’ ” Dr. Goldberg said.
“We can reassure them now. A lot of us are able to say there’s nothing to that, we’re very clear about this, stay on the meds. If anything, there’s data that suggest actually it may be better to be on an ACE inhibitor; that the hospitalizations were shorter and the outcomes were a little bit better.”
For the current study, published online Oct. 28 in the Journal of the American College of Cardiology, the investigators analyzed 6,439 patients admitted for COVID-19 at one of five Mount Sinai Health System hospitals in New York between Feb. 27 and June 26. Their mean age was 65.3 years, 45% were women, and one-third were treated with RAAS inhibitors before admission.
Using ICD-9/10 codes and individual chart review, HF was identified in 422 patients (6.6%), of which 250 patients had HFpEF (≥50%), 44 had HFmrEF (41%-49%), and 128 had HFrEF (≤40%).
Patients with HFpEF were older, more frequently women with a higher body mass index and history of lung disease than patients with HFrEF, whereas those with HFmrEF fell in between.
The HFpEF group was also treated with hydroxychloroquine or macrolides and noninvasive ventilation more frequently than the other two groups, whereas antiplatelet and neurohormonal therapies were more common in the HFrEF group.
Patients with a history of HF had significantly longer hospital stays than those without HF (8 days vs. 6 days), increased need for intubation (22.8% vs. 11.9%) and ICU care (23.2% vs. 16.6%), and worse in-hospital mortality (40% vs. 24.9%).
After multivariable regression adjustment, HF persisted as an independent risk factor for ICU care (odds ratio, 1.71; 95% CI, 1.25-2.34), intubation and mechanical ventilation (OR, 3.64; 95% CI, 2.56-5.16), and in-hospital mortality (OR, 1.88; 95% CI, 1.27-2.78).
“I knew to expect higher rates of adverse outcomes but I didn’t expect it to be nearly a twofold increase,” Dr. Lala said. “I thought that was pretty powerful.”
No significant differences were seen across LVEF categories in length of stay, need for ICU care, intubation and mechanical ventilation, acute kidney injury, shock, thromboembolic events, arrhythmias, or 30-day readmission rates.
However, cardiogenic shock (7.8% vs. 2.3% vs. 2%) and HF-related causes for 30-day readmissions (47.1% vs. 0% vs. 8.6%) were significantly higher in patients with HFrEF than in those with HFmrEF or HFpEF.
Also, mortality was lower in those with HFmrEF (22.7%) than with HFrEF (38.3%) and HFpEF (44%). The group was small but the “results suggested that patients with HFmrEF could have a better prognosis, because they can represent a distinct and more favorable HF phenotype,” the authors wrote.
The statistical testing didn’t show much difference and the patient numbers were very small, noted Dr. Goldberg. “So they might be overreaching a little bit there.”
“To me, the take-home message is that just having the phenotype of heart failure, regardless of EF, is associated with bad outcomes and we need to be vigilant on two fronts,” he said. “We really need to be doing prevention in the folks with heart failure because if they get COVID their outcomes are not going to be as good. Second, as clinicians, if we see a patient presenting with COVID who has a history of heart failure we may want to be much more vigilant with that individual than we might otherwise be. So I think there’s something to be said for kind of risk-stratifying people in that way.”
Dr. Goldberg pointed out that the study had many “amazing strengths,” including a large, racially diverse population, direct chart review to identify HF patients, and capturing a patient’s specific HF phenotype.
Weaknesses are that it was a single-center study, so the biases of how these patients were treated are not easily controlled for, he said. “We also don’t know when the hospital system was very strained as they were making some decisions: Were the older patients who had advanced heart and lung disease ultimately less aggressively treated because they felt they wouldn’t survive?”
Dr. Lala has received personal fees from Zoll, outside the submitted work. Dr. Goldberg reported research funding with Respicardia and consulting fees from Abbott.
This article first appeared on Medscape.com.
People with a history of heart failure – no matter the type – face more complications and death than their peers without HF once hospitalized with COVID-19, a new observational study shows.
A history of HF was associated with a near doubling risk of in-hospital mortality and ICU care and more than a tripling risk of mechanical ventilation despite adjustment for 18 factors including race, obesity, diabetes, previous treatment with renin-angiotensin-aldosterone system (RAAS) inhibitors, and severity of illness.
Adverse outcomes were high regardless of whether patients had HF with a preserved, mid-range, or reduced left ventricular ejection fraction (HFpEF/HFmrEF/HFrEF).
“That for me was the real zinger,” senior author Anuradha Lala, MD, said in an interview . “Because as clinicians, oftentimes, and wrongly so, we think this person has preserved ejection fraction, so they’re not needing my heart failure expertise as much as someone with heart failure with reduced ejection fraction.”
In the peak of the pandemic, that may have meant triaging patients with HFpEF to a regular floor, whereas those with HFrEF were seen by the specialist team.
“What this alerted me to is to take heart failure as a diagnosis very seriously, regardless of ejection fraction, and that is very much in line with all of the emerging data about heart failure with preserved ejection fraction,” said Dr. Lala, from the Icahn School of Medicine at Mount Sinai, New York.
“Now when I see patients in the clinic, I incorporate part of our visit to talking about what they are doing to prevent COVID, which I really wasn’t doing before. It was like ‘Oh yeah, what crazy times we’re dealing with’ and then addressing their heart failure as I normally would,” she said. “But now, interwoven into every visit is: Are you wearing a mask, what’s your social distancing policy, who are you living with at home, has anyone at home or who you’ve interacted with been sick? I’m asking those questions just as a knee-jerk reaction for these patients because I know the repercussions. We have to keep in mind these are observational studies, so I can’t prove causality but these are observations that are, nonetheless, quite robust.”
Although cardiovascular disease, including HF, is recognized as a risk factor for worse outcomes in COVID-19 patients, data are sparse on the clinical course and prognosis of patients with preexisting HF.
“I would have expected that there would have been a gradation of risk from the people with very low ejection fractions up into the normal range, but here it didn’t seem to matter at all. So that’s an important point that bad outcomes were independent of ejection fraction,” commented Lee Goldberg, MD, professor of medicine and chief of advanced heart failure and cardiac transplant at the University of Pennsylvania, Philadelphia.
The study also validated that there is no association between use of RAAS inhibitors and bad outcomes in patients with COVID-19, he said.
Although this has been demonstrated in several studies, concerns were raised early in the pandemic that ACE inhibitors and angiotensin receptor blockers could facilitate infection with SARS-CoV-2 and increase the risk of severe or lethal COVID-19.
“For most clinicians that question has been put to bed, but we’re still getting patients that will ask during office visits ‘Is it safe for me to stay on?’ They still have that doubt [about] ‘Are we doing the right thing?’ ” Dr. Goldberg said.
“We can reassure them now. A lot of us are able to say there’s nothing to that, we’re very clear about this, stay on the meds. If anything, there’s data that suggest actually it may be better to be on an ACE inhibitor; that the hospitalizations were shorter and the outcomes were a little bit better.”
For the current study, published online Oct. 28 in the Journal of the American College of Cardiology, the investigators analyzed 6,439 patients admitted for COVID-19 at one of five Mount Sinai Health System hospitals in New York between Feb. 27 and June 26. Their mean age was 65.3 years, 45% were women, and one-third were treated with RAAS inhibitors before admission.
Using ICD-9/10 codes and individual chart review, HF was identified in 422 patients (6.6%), of which 250 patients had HFpEF (≥50%), 44 had HFmrEF (41%-49%), and 128 had HFrEF (≤40%).
Patients with HFpEF were older, more frequently women with a higher body mass index and history of lung disease than patients with HFrEF, whereas those with HFmrEF fell in between.
The HFpEF group was also treated with hydroxychloroquine or macrolides and noninvasive ventilation more frequently than the other two groups, whereas antiplatelet and neurohormonal therapies were more common in the HFrEF group.
Patients with a history of HF had significantly longer hospital stays than those without HF (8 days vs. 6 days), increased need for intubation (22.8% vs. 11.9%) and ICU care (23.2% vs. 16.6%), and worse in-hospital mortality (40% vs. 24.9%).
After multivariable regression adjustment, HF persisted as an independent risk factor for ICU care (odds ratio, 1.71; 95% CI, 1.25-2.34), intubation and mechanical ventilation (OR, 3.64; 95% CI, 2.56-5.16), and in-hospital mortality (OR, 1.88; 95% CI, 1.27-2.78).
“I knew to expect higher rates of adverse outcomes but I didn’t expect it to be nearly a twofold increase,” Dr. Lala said. “I thought that was pretty powerful.”
No significant differences were seen across LVEF categories in length of stay, need for ICU care, intubation and mechanical ventilation, acute kidney injury, shock, thromboembolic events, arrhythmias, or 30-day readmission rates.
However, cardiogenic shock (7.8% vs. 2.3% vs. 2%) and HF-related causes for 30-day readmissions (47.1% vs. 0% vs. 8.6%) were significantly higher in patients with HFrEF than in those with HFmrEF or HFpEF.
Also, mortality was lower in those with HFmrEF (22.7%) than with HFrEF (38.3%) and HFpEF (44%). The group was small but the “results suggested that patients with HFmrEF could have a better prognosis, because they can represent a distinct and more favorable HF phenotype,” the authors wrote.
The statistical testing didn’t show much difference and the patient numbers were very small, noted Dr. Goldberg. “So they might be overreaching a little bit there.”
“To me, the take-home message is that just having the phenotype of heart failure, regardless of EF, is associated with bad outcomes and we need to be vigilant on two fronts,” he said. “We really need to be doing prevention in the folks with heart failure because if they get COVID their outcomes are not going to be as good. Second, as clinicians, if we see a patient presenting with COVID who has a history of heart failure we may want to be much more vigilant with that individual than we might otherwise be. So I think there’s something to be said for kind of risk-stratifying people in that way.”
Dr. Goldberg pointed out that the study had many “amazing strengths,” including a large, racially diverse population, direct chart review to identify HF patients, and capturing a patient’s specific HF phenotype.
Weaknesses are that it was a single-center study, so the biases of how these patients were treated are not easily controlled for, he said. “We also don’t know when the hospital system was very strained as they were making some decisions: Were the older patients who had advanced heart and lung disease ultimately less aggressively treated because they felt they wouldn’t survive?”
Dr. Lala has received personal fees from Zoll, outside the submitted work. Dr. Goldberg reported research funding with Respicardia and consulting fees from Abbott.
This article first appeared on Medscape.com.
Biologics may protect psoriasis patients against severe COVID-19
presented at the virtual annual congress of the European Academy of Dermatology and Venereology.
“Biologics seem to be very protective against severe, poor-prognosis COVID-19, but they do not prevent infection with the virus,” reported Giovanni Damiani, MD, a dermatologist at the University of Milan.
This apparent protective effect of biologic agents against severe and even fatal COVID-19 is all the more impressive because the psoriasis patients included in the Italian study – as is true of those elsewhere throughout the world – had relatively high rates of obesity, smoking, and chronic obstructive pulmonary disease, known risk factors for severe COVID-19, he added.
He presented a case-control study including 1,193 adult psoriasis patients on biologics or apremilast (Otezla) at Milan’s San Donato Hospital during the period from Feb. 21 to April 9, 2020. The control group comprised more than 10 million individuals, the entire adult population of the Lombardy region, of which Milan is the capital. This was the hardest-hit area in all of Italy during the first wave of COVID-19.
Twenty-two of the 1,193 psoriasis patients experienced confirmed COVID-19 during the study period. Seventeen were quarantined at home because their disease was mild. Five were hospitalized. But no psoriasis patients were placed in intensive care, and none died.
Psoriasis patients on biologics were significantly more likely than the general Lombardian population to test positive for COVID-19, with an unadjusted odds ratio of 3.43. They were at 9.05-fold increased risk of home quarantine for mild disease, and at 3.59-fold greater risk than controls for hospitalization for COVID-19. However, they were not at significantly increased risk of ICU admission. And while they actually had a 59% relative risk reduction for death, this didn’t achieve statistical significance.
Forty-five percent of the psoriasis patients were on an interleukin-17 (IL-17) inhibitor, 22% were on a tumor necrosis factor–alpha inhibitor, and 20% were taking an IL-12/23 inhibitor. Of note, none of 77 patients on apremilast developed COVID-19, even though it is widely considered a less potent psoriasis therapy than the injectable monoclonal antibody biologics.
The French experience
Anne-Claire Fougerousse, MD, and her French coinvestigators conducted a study designed to address a different question: Is it safe to start psoriasis patients on biologics or older conventional systemic agents such as methotrexate during the pandemic?
She presented a French national cross-sectional study of 1,418 adult psoriasis patients on a biologic or standard systemic therapy during a snapshot in time near the peak of the first wave of the pandemic in France: the period from April 27 to May 7, 2020. The group included 1,188 psoriasis patients on maintenance therapy and 230 who had initiated systemic treatment within the past 4 months. More than one-third of the patients had at least one risk factor for severe COVID-19.
Although testing wasn’t available to confirm all cases, 54 patients developed probable COVID-19 during the study period. Only five required hospitalization. None died. The two hospitalized psoriasis patients admitted to an ICU had obesity as a risk factor for severe COVID-19, as did another of the five hospitalized patients, reported Dr. Fougerousse, a dermatologist at the Bégin Military Teaching Hospital in Saint-Mandé, France. Hospitalization for COVID-19 was required in 0.43% of the French treatment initiators, not significantly different from the 0.34% rate in patients on maintenance systemic therapy. A study limitation was the lack of a control group.
Nonetheless, the data did answer the investigators’ main question: “This is the first data showing no increased incidence of severe COVID-19 in psoriasis patients receiving systemic therapy in the treatment initiation period compared to those on maintenance therapy. This may now allow physicians to initiate conventional systemic or biologic therapy in patients with severe psoriasis on a case-by-case basis in the context of the persistent COVID-19 pandemic,” Dr. Fougerousse concluded.
Proposed mechanism of benefit
The Italian study findings that biologics boost the risk of infection with the SARS-CoV-2 virus in psoriasis patients while potentially protecting them against ICU admission and death are backed by a biologically plausible albeit as yet unproven mechanism of action, Dr. Damiani asserted.
He elaborated: A vast body of high-quality clinical trials data demonstrates that these targeted immunosuppressive agents are associated with modestly increased risk of viral infections, including both skin and respiratory tract infections. So there is no reason to suppose these agents would offer protection against the first phase of COVID-19, involving SARS-CoV-2 infection, nor protect against the second (pulmonary phase), whose hallmarks are dyspnea with or without hypoxia. But progression to the third phase, involving hyperinflammation and hypercoagulation – dubbed the cytokine storm – could be a different matter.
“Of particular interest was that our patients on IL-17 inhibitors displayed a really great outcome. Interleukin-17 has procoagulant and prothrombotic effects, organizes bronchoalveolar remodeling, has a profibrotic effect, induces mitochondrial dysfunction, and encourages dendritic cell migration in peribronchial lymph nodes. Therefore, by antagonizing this interleukin, we may have a better prognosis, although further studies are needed to be certain,” Dr. Damiani commented.
Publication of his preliminary findings drew the attention of a group of highly respected thought leaders in psoriasis, including James G. Krueger, MD, head of the laboratory for investigative dermatology and codirector of the center for clinical and investigative science at Rockefeller University, New York.
The Italian report prompted them to analyze data from the phase 4, double-blind, randomized ObePso-S study investigating the effects of the IL-17 inhibitor secukinumab (Cosentyx) on systemic inflammatory markers and gene expression in psoriasis patients. The investigators demonstrated that IL-17–mediated inflammation in psoriasis patients was associated with increased expression of the angiotensin-converting enzyme 2 (ACE2) receptor in lesional skin, and that treatment with secukinumab dropped ACE2 expression to levels seen in nonlesional skin. Given that ACE2 is the chief portal of entry for SARS-CoV-2 and that IL-17 exerts systemic proinflammatory effects, it’s plausible that inhibition of IL-17–mediated inflammation via dampening of ACE2 expression in noncutaneous epithelia “could prove to be advantageous in patients with psoriasis who are at risk for SARS-CoV-2 infection,” according to Dr. Krueger and his coinvestigators in the Journal of Allergy and Clinical Immunology.
Dr. Damiani and Dr. Fougerousse reported having no financial conflicts regarding their studies. The secukinumab/ACE2 receptor study was funded by Novartis.
presented at the virtual annual congress of the European Academy of Dermatology and Venereology.
“Biologics seem to be very protective against severe, poor-prognosis COVID-19, but they do not prevent infection with the virus,” reported Giovanni Damiani, MD, a dermatologist at the University of Milan.
This apparent protective effect of biologic agents against severe and even fatal COVID-19 is all the more impressive because the psoriasis patients included in the Italian study – as is true of those elsewhere throughout the world – had relatively high rates of obesity, smoking, and chronic obstructive pulmonary disease, known risk factors for severe COVID-19, he added.
He presented a case-control study including 1,193 adult psoriasis patients on biologics or apremilast (Otezla) at Milan’s San Donato Hospital during the period from Feb. 21 to April 9, 2020. The control group comprised more than 10 million individuals, the entire adult population of the Lombardy region, of which Milan is the capital. This was the hardest-hit area in all of Italy during the first wave of COVID-19.
Twenty-two of the 1,193 psoriasis patients experienced confirmed COVID-19 during the study period. Seventeen were quarantined at home because their disease was mild. Five were hospitalized. But no psoriasis patients were placed in intensive care, and none died.
Psoriasis patients on biologics were significantly more likely than the general Lombardian population to test positive for COVID-19, with an unadjusted odds ratio of 3.43. They were at 9.05-fold increased risk of home quarantine for mild disease, and at 3.59-fold greater risk than controls for hospitalization for COVID-19. However, they were not at significantly increased risk of ICU admission. And while they actually had a 59% relative risk reduction for death, this didn’t achieve statistical significance.
Forty-five percent of the psoriasis patients were on an interleukin-17 (IL-17) inhibitor, 22% were on a tumor necrosis factor–alpha inhibitor, and 20% were taking an IL-12/23 inhibitor. Of note, none of 77 patients on apremilast developed COVID-19, even though it is widely considered a less potent psoriasis therapy than the injectable monoclonal antibody biologics.
The French experience
Anne-Claire Fougerousse, MD, and her French coinvestigators conducted a study designed to address a different question: Is it safe to start psoriasis patients on biologics or older conventional systemic agents such as methotrexate during the pandemic?
She presented a French national cross-sectional study of 1,418 adult psoriasis patients on a biologic or standard systemic therapy during a snapshot in time near the peak of the first wave of the pandemic in France: the period from April 27 to May 7, 2020. The group included 1,188 psoriasis patients on maintenance therapy and 230 who had initiated systemic treatment within the past 4 months. More than one-third of the patients had at least one risk factor for severe COVID-19.
Although testing wasn’t available to confirm all cases, 54 patients developed probable COVID-19 during the study period. Only five required hospitalization. None died. The two hospitalized psoriasis patients admitted to an ICU had obesity as a risk factor for severe COVID-19, as did another of the five hospitalized patients, reported Dr. Fougerousse, a dermatologist at the Bégin Military Teaching Hospital in Saint-Mandé, France. Hospitalization for COVID-19 was required in 0.43% of the French treatment initiators, not significantly different from the 0.34% rate in patients on maintenance systemic therapy. A study limitation was the lack of a control group.
Nonetheless, the data did answer the investigators’ main question: “This is the first data showing no increased incidence of severe COVID-19 in psoriasis patients receiving systemic therapy in the treatment initiation period compared to those on maintenance therapy. This may now allow physicians to initiate conventional systemic or biologic therapy in patients with severe psoriasis on a case-by-case basis in the context of the persistent COVID-19 pandemic,” Dr. Fougerousse concluded.
Proposed mechanism of benefit
The Italian study findings that biologics boost the risk of infection with the SARS-CoV-2 virus in psoriasis patients while potentially protecting them against ICU admission and death are backed by a biologically plausible albeit as yet unproven mechanism of action, Dr. Damiani asserted.
He elaborated: A vast body of high-quality clinical trials data demonstrates that these targeted immunosuppressive agents are associated with modestly increased risk of viral infections, including both skin and respiratory tract infections. So there is no reason to suppose these agents would offer protection against the first phase of COVID-19, involving SARS-CoV-2 infection, nor protect against the second (pulmonary phase), whose hallmarks are dyspnea with or without hypoxia. But progression to the third phase, involving hyperinflammation and hypercoagulation – dubbed the cytokine storm – could be a different matter.
“Of particular interest was that our patients on IL-17 inhibitors displayed a really great outcome. Interleukin-17 has procoagulant and prothrombotic effects, organizes bronchoalveolar remodeling, has a profibrotic effect, induces mitochondrial dysfunction, and encourages dendritic cell migration in peribronchial lymph nodes. Therefore, by antagonizing this interleukin, we may have a better prognosis, although further studies are needed to be certain,” Dr. Damiani commented.
Publication of his preliminary findings drew the attention of a group of highly respected thought leaders in psoriasis, including James G. Krueger, MD, head of the laboratory for investigative dermatology and codirector of the center for clinical and investigative science at Rockefeller University, New York.
The Italian report prompted them to analyze data from the phase 4, double-blind, randomized ObePso-S study investigating the effects of the IL-17 inhibitor secukinumab (Cosentyx) on systemic inflammatory markers and gene expression in psoriasis patients. The investigators demonstrated that IL-17–mediated inflammation in psoriasis patients was associated with increased expression of the angiotensin-converting enzyme 2 (ACE2) receptor in lesional skin, and that treatment with secukinumab dropped ACE2 expression to levels seen in nonlesional skin. Given that ACE2 is the chief portal of entry for SARS-CoV-2 and that IL-17 exerts systemic proinflammatory effects, it’s plausible that inhibition of IL-17–mediated inflammation via dampening of ACE2 expression in noncutaneous epithelia “could prove to be advantageous in patients with psoriasis who are at risk for SARS-CoV-2 infection,” according to Dr. Krueger and his coinvestigators in the Journal of Allergy and Clinical Immunology.
Dr. Damiani and Dr. Fougerousse reported having no financial conflicts regarding their studies. The secukinumab/ACE2 receptor study was funded by Novartis.
presented at the virtual annual congress of the European Academy of Dermatology and Venereology.
“Biologics seem to be very protective against severe, poor-prognosis COVID-19, but they do not prevent infection with the virus,” reported Giovanni Damiani, MD, a dermatologist at the University of Milan.
This apparent protective effect of biologic agents against severe and even fatal COVID-19 is all the more impressive because the psoriasis patients included in the Italian study – as is true of those elsewhere throughout the world – had relatively high rates of obesity, smoking, and chronic obstructive pulmonary disease, known risk factors for severe COVID-19, he added.
He presented a case-control study including 1,193 adult psoriasis patients on biologics or apremilast (Otezla) at Milan’s San Donato Hospital during the period from Feb. 21 to April 9, 2020. The control group comprised more than 10 million individuals, the entire adult population of the Lombardy region, of which Milan is the capital. This was the hardest-hit area in all of Italy during the first wave of COVID-19.
Twenty-two of the 1,193 psoriasis patients experienced confirmed COVID-19 during the study period. Seventeen were quarantined at home because their disease was mild. Five were hospitalized. But no psoriasis patients were placed in intensive care, and none died.
Psoriasis patients on biologics were significantly more likely than the general Lombardian population to test positive for COVID-19, with an unadjusted odds ratio of 3.43. They were at 9.05-fold increased risk of home quarantine for mild disease, and at 3.59-fold greater risk than controls for hospitalization for COVID-19. However, they were not at significantly increased risk of ICU admission. And while they actually had a 59% relative risk reduction for death, this didn’t achieve statistical significance.
Forty-five percent of the psoriasis patients were on an interleukin-17 (IL-17) inhibitor, 22% were on a tumor necrosis factor–alpha inhibitor, and 20% were taking an IL-12/23 inhibitor. Of note, none of 77 patients on apremilast developed COVID-19, even though it is widely considered a less potent psoriasis therapy than the injectable monoclonal antibody biologics.
The French experience
Anne-Claire Fougerousse, MD, and her French coinvestigators conducted a study designed to address a different question: Is it safe to start psoriasis patients on biologics or older conventional systemic agents such as methotrexate during the pandemic?
She presented a French national cross-sectional study of 1,418 adult psoriasis patients on a biologic or standard systemic therapy during a snapshot in time near the peak of the first wave of the pandemic in France: the period from April 27 to May 7, 2020. The group included 1,188 psoriasis patients on maintenance therapy and 230 who had initiated systemic treatment within the past 4 months. More than one-third of the patients had at least one risk factor for severe COVID-19.
Although testing wasn’t available to confirm all cases, 54 patients developed probable COVID-19 during the study period. Only five required hospitalization. None died. The two hospitalized psoriasis patients admitted to an ICU had obesity as a risk factor for severe COVID-19, as did another of the five hospitalized patients, reported Dr. Fougerousse, a dermatologist at the Bégin Military Teaching Hospital in Saint-Mandé, France. Hospitalization for COVID-19 was required in 0.43% of the French treatment initiators, not significantly different from the 0.34% rate in patients on maintenance systemic therapy. A study limitation was the lack of a control group.
Nonetheless, the data did answer the investigators’ main question: “This is the first data showing no increased incidence of severe COVID-19 in psoriasis patients receiving systemic therapy in the treatment initiation period compared to those on maintenance therapy. This may now allow physicians to initiate conventional systemic or biologic therapy in patients with severe psoriasis on a case-by-case basis in the context of the persistent COVID-19 pandemic,” Dr. Fougerousse concluded.
Proposed mechanism of benefit
The Italian study findings that biologics boost the risk of infection with the SARS-CoV-2 virus in psoriasis patients while potentially protecting them against ICU admission and death are backed by a biologically plausible albeit as yet unproven mechanism of action, Dr. Damiani asserted.
He elaborated: A vast body of high-quality clinical trials data demonstrates that these targeted immunosuppressive agents are associated with modestly increased risk of viral infections, including both skin and respiratory tract infections. So there is no reason to suppose these agents would offer protection against the first phase of COVID-19, involving SARS-CoV-2 infection, nor protect against the second (pulmonary phase), whose hallmarks are dyspnea with or without hypoxia. But progression to the third phase, involving hyperinflammation and hypercoagulation – dubbed the cytokine storm – could be a different matter.
“Of particular interest was that our patients on IL-17 inhibitors displayed a really great outcome. Interleukin-17 has procoagulant and prothrombotic effects, organizes bronchoalveolar remodeling, has a profibrotic effect, induces mitochondrial dysfunction, and encourages dendritic cell migration in peribronchial lymph nodes. Therefore, by antagonizing this interleukin, we may have a better prognosis, although further studies are needed to be certain,” Dr. Damiani commented.
Publication of his preliminary findings drew the attention of a group of highly respected thought leaders in psoriasis, including James G. Krueger, MD, head of the laboratory for investigative dermatology and codirector of the center for clinical and investigative science at Rockefeller University, New York.
The Italian report prompted them to analyze data from the phase 4, double-blind, randomized ObePso-S study investigating the effects of the IL-17 inhibitor secukinumab (Cosentyx) on systemic inflammatory markers and gene expression in psoriasis patients. The investigators demonstrated that IL-17–mediated inflammation in psoriasis patients was associated with increased expression of the angiotensin-converting enzyme 2 (ACE2) receptor in lesional skin, and that treatment with secukinumab dropped ACE2 expression to levels seen in nonlesional skin. Given that ACE2 is the chief portal of entry for SARS-CoV-2 and that IL-17 exerts systemic proinflammatory effects, it’s plausible that inhibition of IL-17–mediated inflammation via dampening of ACE2 expression in noncutaneous epithelia “could prove to be advantageous in patients with psoriasis who are at risk for SARS-CoV-2 infection,” according to Dr. Krueger and his coinvestigators in the Journal of Allergy and Clinical Immunology.
Dr. Damiani and Dr. Fougerousse reported having no financial conflicts regarding their studies. The secukinumab/ACE2 receptor study was funded by Novartis.
FROM THE EADV CONGRESS
Updated heart failure measures add newer meds
Safety measures for lab monitoring of mineralocorticoid receptor agonist therapy, performance measures for sacubitril/valsartan, cardiac resynchronization therapy and titration of medications, and quality measures based on patient-reported outcomes are among the updates the joint task force of the American College of Cardiology and the American Heart Association have made to performance and quality measures for managing adults with heart failure.
The revisions, published online Nov. 2 in the Journal of the American College of Cardiology, update the 2011 ACC/AHA heart failure measure set, writing committee vice chair Gregg C. Fonarow, MD, said in an interview. The 2011 measure set predates the 2015 approval of the angiotensin receptor neprilysin inhibitor (ARNI) sacubitril/valsartan for heart failure in adults.
Measures stress dosages, strength of evidence
“For the first time the heart failure performance measure sets also focus on not just the use of guideline-recommended medication at any dose, but on utilizing the doses that are evidence-based and guideline recommended so long as they are well tolerated,” said Dr. Fonarow, interim chief of cardiology at the University of California, Los Angeles. “The measure set now includes assessment of patients being treated with doses of medications at 50% or greater of target dose in the absence of contraindications or documented intolerance.”
The update includes seven new performance measures, two quality measures, and one structural measure. The performance measures come from the strongest recommendations – that is, a class of recommendation of 1 (strong) or 3 (no benefit or harmful, process to be avoided) – in the 2017 ACC/AHA/Heart Failure Society of American heart failure guideline update published in Circulation.
In addition to the 2017 update, the writing committee also reviewed existing performance measures. “Those management strategies, diagnostic testing, medications, and devices with the strongest evidence and highest level of guideline recommendations were further considered for inclusion in the performance measure set,” Dr. Fonarow said. “The measures went through extensive review by peer reviewers and approval from the organizations represented.”
Specifically, the update includes measures for monitoring serum potassium after starting mineralocorticoid receptor antagonists therapy, and cardiac resynchronization therapy for patients with heart failure with reduced ejection fraction already on guideline-directed therapy. “This therapy can significantly improve functional capacity and outcomes in appropriately selected patients,” Dr. Fonarow said.
New and retired measures
The update adds two performance measures for titration of medications based on dose, either reaching 50% of the recommended dose for a variety of medications, including ARNI, or documenting that the dose wasn’t tolerated for other reason for not using the dose.
The new structural measure calls for facility participation in a heart failure registry. The revised measure set now consists of 18 measures in all.
The update retired one measure from the 2011 set: left ventricular ejection fraction assessment for inpatients. The committee cited its use above 97% as the reason, but LVEF in outpatients remains a measure.
The following tree measures have been revised:
- Patient self-care education has moved from performance measure to quality measure because of concerns about the accuracy of self-care education documentation and limited evidence of improved outcomes with better documentation.
- ACE inhibitor or angiotensin receptor blocker therapy for left ventricular systolic dysfunction adds ARNI therapy to align with the 2017 ACC/AHA/HFSA update.
- Postdischarge appointments shifts from performance to quality measure and include a 7-day limit.
Measures future research should focus on, noted Dr. Fonarow, include the use of sodium glucose cotransporter 2 (SGLT2) inhibitors for heart failure, including in patients without diabetes. “Since the ACC/AHA heart failure guidelines had not yet been updated to recommend these therapies they could not be included in this performance measure set,” he said.
He also said “an urgent need” exists for further research into treatments for heart failure with preserved ejection fraction along with optimal implementation strategies.
“If these ACC/AHA heart failure performance measures were applied in all settings in which patients with heart failure in the United States are being cared for, and optimal and equitable conformity with each of these measures were achieved, over 100,000 lives a year of patients with heart failure could be saved,” he said. “There’s in an urgent need to measure and improve heart failure care quality.”
Dr. Fonarow reported financial relationships with Abbott, Amgen, AstraZeneca, CHF Solutions, Janssen, Medtronic, Merck, and Novartis.
SOURCE: American College of Cardiology/American Heart Association Task Force on Performance Measures. J Am Coll Cardiol. 2020 Nov 2;76:2527-64.
Safety measures for lab monitoring of mineralocorticoid receptor agonist therapy, performance measures for sacubitril/valsartan, cardiac resynchronization therapy and titration of medications, and quality measures based on patient-reported outcomes are among the updates the joint task force of the American College of Cardiology and the American Heart Association have made to performance and quality measures for managing adults with heart failure.
The revisions, published online Nov. 2 in the Journal of the American College of Cardiology, update the 2011 ACC/AHA heart failure measure set, writing committee vice chair Gregg C. Fonarow, MD, said in an interview. The 2011 measure set predates the 2015 approval of the angiotensin receptor neprilysin inhibitor (ARNI) sacubitril/valsartan for heart failure in adults.
Measures stress dosages, strength of evidence
“For the first time the heart failure performance measure sets also focus on not just the use of guideline-recommended medication at any dose, but on utilizing the doses that are evidence-based and guideline recommended so long as they are well tolerated,” said Dr. Fonarow, interim chief of cardiology at the University of California, Los Angeles. “The measure set now includes assessment of patients being treated with doses of medications at 50% or greater of target dose in the absence of contraindications or documented intolerance.”
The update includes seven new performance measures, two quality measures, and one structural measure. The performance measures come from the strongest recommendations – that is, a class of recommendation of 1 (strong) or 3 (no benefit or harmful, process to be avoided) – in the 2017 ACC/AHA/Heart Failure Society of American heart failure guideline update published in Circulation.
In addition to the 2017 update, the writing committee also reviewed existing performance measures. “Those management strategies, diagnostic testing, medications, and devices with the strongest evidence and highest level of guideline recommendations were further considered for inclusion in the performance measure set,” Dr. Fonarow said. “The measures went through extensive review by peer reviewers and approval from the organizations represented.”
Specifically, the update includes measures for monitoring serum potassium after starting mineralocorticoid receptor antagonists therapy, and cardiac resynchronization therapy for patients with heart failure with reduced ejection fraction already on guideline-directed therapy. “This therapy can significantly improve functional capacity and outcomes in appropriately selected patients,” Dr. Fonarow said.
New and retired measures
The update adds two performance measures for titration of medications based on dose, either reaching 50% of the recommended dose for a variety of medications, including ARNI, or documenting that the dose wasn’t tolerated for other reason for not using the dose.
The new structural measure calls for facility participation in a heart failure registry. The revised measure set now consists of 18 measures in all.
The update retired one measure from the 2011 set: left ventricular ejection fraction assessment for inpatients. The committee cited its use above 97% as the reason, but LVEF in outpatients remains a measure.
The following tree measures have been revised:
- Patient self-care education has moved from performance measure to quality measure because of concerns about the accuracy of self-care education documentation and limited evidence of improved outcomes with better documentation.
- ACE inhibitor or angiotensin receptor blocker therapy for left ventricular systolic dysfunction adds ARNI therapy to align with the 2017 ACC/AHA/HFSA update.
- Postdischarge appointments shifts from performance to quality measure and include a 7-day limit.
Measures future research should focus on, noted Dr. Fonarow, include the use of sodium glucose cotransporter 2 (SGLT2) inhibitors for heart failure, including in patients without diabetes. “Since the ACC/AHA heart failure guidelines had not yet been updated to recommend these therapies they could not be included in this performance measure set,” he said.
He also said “an urgent need” exists for further research into treatments for heart failure with preserved ejection fraction along with optimal implementation strategies.
“If these ACC/AHA heart failure performance measures were applied in all settings in which patients with heart failure in the United States are being cared for, and optimal and equitable conformity with each of these measures were achieved, over 100,000 lives a year of patients with heart failure could be saved,” he said. “There’s in an urgent need to measure and improve heart failure care quality.”
Dr. Fonarow reported financial relationships with Abbott, Amgen, AstraZeneca, CHF Solutions, Janssen, Medtronic, Merck, and Novartis.
SOURCE: American College of Cardiology/American Heart Association Task Force on Performance Measures. J Am Coll Cardiol. 2020 Nov 2;76:2527-64.
Safety measures for lab monitoring of mineralocorticoid receptor agonist therapy, performance measures for sacubitril/valsartan, cardiac resynchronization therapy and titration of medications, and quality measures based on patient-reported outcomes are among the updates the joint task force of the American College of Cardiology and the American Heart Association have made to performance and quality measures for managing adults with heart failure.
The revisions, published online Nov. 2 in the Journal of the American College of Cardiology, update the 2011 ACC/AHA heart failure measure set, writing committee vice chair Gregg C. Fonarow, MD, said in an interview. The 2011 measure set predates the 2015 approval of the angiotensin receptor neprilysin inhibitor (ARNI) sacubitril/valsartan for heart failure in adults.
Measures stress dosages, strength of evidence
“For the first time the heart failure performance measure sets also focus on not just the use of guideline-recommended medication at any dose, but on utilizing the doses that are evidence-based and guideline recommended so long as they are well tolerated,” said Dr. Fonarow, interim chief of cardiology at the University of California, Los Angeles. “The measure set now includes assessment of patients being treated with doses of medications at 50% or greater of target dose in the absence of contraindications or documented intolerance.”
The update includes seven new performance measures, two quality measures, and one structural measure. The performance measures come from the strongest recommendations – that is, a class of recommendation of 1 (strong) or 3 (no benefit or harmful, process to be avoided) – in the 2017 ACC/AHA/Heart Failure Society of American heart failure guideline update published in Circulation.
In addition to the 2017 update, the writing committee also reviewed existing performance measures. “Those management strategies, diagnostic testing, medications, and devices with the strongest evidence and highest level of guideline recommendations were further considered for inclusion in the performance measure set,” Dr. Fonarow said. “The measures went through extensive review by peer reviewers and approval from the organizations represented.”
Specifically, the update includes measures for monitoring serum potassium after starting mineralocorticoid receptor antagonists therapy, and cardiac resynchronization therapy for patients with heart failure with reduced ejection fraction already on guideline-directed therapy. “This therapy can significantly improve functional capacity and outcomes in appropriately selected patients,” Dr. Fonarow said.
New and retired measures
The update adds two performance measures for titration of medications based on dose, either reaching 50% of the recommended dose for a variety of medications, including ARNI, or documenting that the dose wasn’t tolerated for other reason for not using the dose.
The new structural measure calls for facility participation in a heart failure registry. The revised measure set now consists of 18 measures in all.
The update retired one measure from the 2011 set: left ventricular ejection fraction assessment for inpatients. The committee cited its use above 97% as the reason, but LVEF in outpatients remains a measure.
The following tree measures have been revised:
- Patient self-care education has moved from performance measure to quality measure because of concerns about the accuracy of self-care education documentation and limited evidence of improved outcomes with better documentation.
- ACE inhibitor or angiotensin receptor blocker therapy for left ventricular systolic dysfunction adds ARNI therapy to align with the 2017 ACC/AHA/HFSA update.
- Postdischarge appointments shifts from performance to quality measure and include a 7-day limit.
Measures future research should focus on, noted Dr. Fonarow, include the use of sodium glucose cotransporter 2 (SGLT2) inhibitors for heart failure, including in patients without diabetes. “Since the ACC/AHA heart failure guidelines had not yet been updated to recommend these therapies they could not be included in this performance measure set,” he said.
He also said “an urgent need” exists for further research into treatments for heart failure with preserved ejection fraction along with optimal implementation strategies.
“If these ACC/AHA heart failure performance measures were applied in all settings in which patients with heart failure in the United States are being cared for, and optimal and equitable conformity with each of these measures were achieved, over 100,000 lives a year of patients with heart failure could be saved,” he said. “There’s in an urgent need to measure and improve heart failure care quality.”
Dr. Fonarow reported financial relationships with Abbott, Amgen, AstraZeneca, CHF Solutions, Janssen, Medtronic, Merck, and Novartis.
SOURCE: American College of Cardiology/American Heart Association Task Force on Performance Measures. J Am Coll Cardiol. 2020 Nov 2;76:2527-64.
FROM THE JOURNAL OF THE AMERICAN COLLEGE OF CARDIOLOGY
Health sector has spent $464 million on lobbying in 2020
, according to the Center for Responsive Politics.
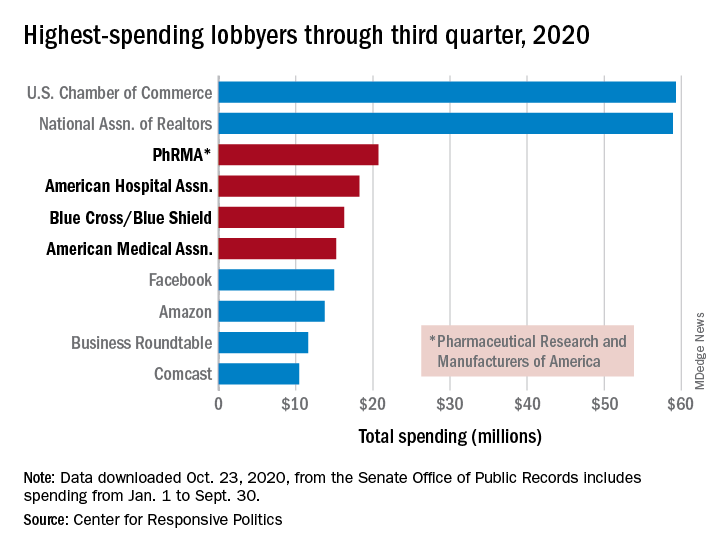
PhRMA spent $20.7 million on lobbying through the end of September, good enough for third on the overall list of U.S. companies and organizations. Three other members of the health sector made the top 10: the American Hospital Association ($18.3 million), BlueCross/BlueShield ($16.3 million), and the American Medical Association ($15.2 million), the center reported.
Total spending by the health sector was $464 million from Jan. 1 to Sept. 30, topping the finance/insurance/real estate sector at $403 million, and miscellaneous business at $371 million. Miscellaneous business is the home of the U.S. Chamber of Commerce, the annual leader in such spending for the last 20 years, based on data from the Senate Office of Public Records.
The largest share of health sector spending came from pharmaceuticals/health products, with a total of almost $233 million, just slightly more than the sector’s four other constituents combined: hospitals/nursing homes ($80 million), health services/HMOs ($75 million), health professionals ($67 million), and miscellaneous health ($9.5 million), the center said on OpenSecrets.org.
Taking one step down from the sector level, that $233 million made pharmaceuticals/health products the highest spending of about 100 industries in 2020, nearly doubling the efforts of electronics manufacturing and equipment ($118 million), which came a distant second. Hospitals/nursing homes was eighth on the industry list, the center noted.
, according to the Center for Responsive Politics.

PhRMA spent $20.7 million on lobbying through the end of September, good enough for third on the overall list of U.S. companies and organizations. Three other members of the health sector made the top 10: the American Hospital Association ($18.3 million), BlueCross/BlueShield ($16.3 million), and the American Medical Association ($15.2 million), the center reported.
Total spending by the health sector was $464 million from Jan. 1 to Sept. 30, topping the finance/insurance/real estate sector at $403 million, and miscellaneous business at $371 million. Miscellaneous business is the home of the U.S. Chamber of Commerce, the annual leader in such spending for the last 20 years, based on data from the Senate Office of Public Records.
The largest share of health sector spending came from pharmaceuticals/health products, with a total of almost $233 million, just slightly more than the sector’s four other constituents combined: hospitals/nursing homes ($80 million), health services/HMOs ($75 million), health professionals ($67 million), and miscellaneous health ($9.5 million), the center said on OpenSecrets.org.
Taking one step down from the sector level, that $233 million made pharmaceuticals/health products the highest spending of about 100 industries in 2020, nearly doubling the efforts of electronics manufacturing and equipment ($118 million), which came a distant second. Hospitals/nursing homes was eighth on the industry list, the center noted.
, according to the Center for Responsive Politics.

PhRMA spent $20.7 million on lobbying through the end of September, good enough for third on the overall list of U.S. companies and organizations. Three other members of the health sector made the top 10: the American Hospital Association ($18.3 million), BlueCross/BlueShield ($16.3 million), and the American Medical Association ($15.2 million), the center reported.
Total spending by the health sector was $464 million from Jan. 1 to Sept. 30, topping the finance/insurance/real estate sector at $403 million, and miscellaneous business at $371 million. Miscellaneous business is the home of the U.S. Chamber of Commerce, the annual leader in such spending for the last 20 years, based on data from the Senate Office of Public Records.
The largest share of health sector spending came from pharmaceuticals/health products, with a total of almost $233 million, just slightly more than the sector’s four other constituents combined: hospitals/nursing homes ($80 million), health services/HMOs ($75 million), health professionals ($67 million), and miscellaneous health ($9.5 million), the center said on OpenSecrets.org.
Taking one step down from the sector level, that $233 million made pharmaceuticals/health products the highest spending of about 100 industries in 2020, nearly doubling the efforts of electronics manufacturing and equipment ($118 million), which came a distant second. Hospitals/nursing homes was eighth on the industry list, the center noted.
COMPARE CRUSH: Crushed prehospital prasugrel misses mark in STEMI
Giving crushed prasugrel (Effient) to patients with ST-segment elevation myocardial infarction (STEMI) en route to a planned primary percutaneous coronary intervention (PCI) does not improve reperfusion rates, results of the COMPARE CRUSH trial show.
Patients assigned to prasugrel as crushed or integral tablets had similar rates of the study’s co-primary endpoints of thrombolysis in myocardial infarction (TIMI) 3 flow in the infarct-related artery at first angiography (31% vs. 32.7%; P = .64) and complete ST-segment resolution 1 hour post PCI (59.9% vs. 57.3%; P = .55).
“These findings hold in spite of the fact that crushed tablets of prasugrel led to more potent platelet inhibition compared with integral tablets,” said study author Georgios Vlachojannis, MD, PhD, University Medical Center Utrecht, the Netherlands.
“Whether faster and more potent antiplatelet therapy can improve coronary reperfusion in contemporary STEMI treatment regimen warrants further investigation.”
The results were reported in a late-breaking clinical science session at the Transcatheter Cardiovascular Therapeutics virtual annual meeting and published simultaneously in the journal Circulation. The meeting was sponsored by the Cardiovascular Research Foundation.
Fibrinolytics and glycoprotein IIb/IIIa inhibitors have demonstrated improved coronary reperfusion and outcomes when given pre hospital. Prior studies have also shown that early administration of a crushed P2Y12 inhibitor increases bioavailability and speeds platelet inhibition in STEMI patients, Dr. Vlachojannis noted.
However, the large randomized ATLANTIC trial, which compared prehospital to cath lab administration of crushed or integral ticagrelor (Brilinta), also found no difference in either TIMI flow in the infarct-related artery or ST-segment resolution.
Between November 2017 and March 2020, the investigator-initiated COMPARE CRUSH trial randomly allocated 727 STEMI patients (mean age, 62 years; 23% female) undergoing primary PCI to receive in the ambulance a 60-mg loading dose of prasugrel as either crushed or integral tablets.
The median time from onset of symptoms to first medical contact was 59 minutes, from first medical contact to study treatment 22 minutes, and from study treatment to primary PCI 57 minutes. These times did not differ between groups.
Platelet reactivity at the beginning of coronary angiography was significantly lower in the crushed group than in the integral group (P2Y12 reactivity units 192 vs. 227; P < .01). This resulted in significantly fewer patients in the crushed group with high platelet reactivity, defined as P2Y12 reactivity units >208, prior to the start of PCI (43.3% vs. 62.6%; P < .01).
There was no difference between the crushed and integral groups in the primary safety endpoint of TIMI major and BARC type 3 or higher bleeding within 48 hours after study treatment (0.4% vs 0.7%).
Death, MI, stroke, and urgent revascularization rates were also similar between groups during index hospitalization and at 30 days. Definite stent thrombosis occurred in one patient in the crushed group and two patients in the integral group.
In an exploratory analysis, the co-primary endpoint results were consistent across multiple subgroups, although there was a trend toward greater benefit on TIMI 3 flow in the crushed tablet group in patients older than age 75 years (P for interaction = .04), presenting with anterior infarction (P for interaction = .03), or with a history of prior PCI (P for interaction < .01).
“However, these results should be regarded as hypothesis-generating,” the authors wrote. “Opioids use in the ambulance was remarkably low in our study compared with the ATLANTIC trial, which might explain that we did not observe any significant interaction.”
Notably, morphine was used in half the ATLANTIC patients and was thought to have possibly delayed the absorption of ticagrelor.
During discussion following the presentation, Sunil V. Rao, MD, Duke University Medical Center, Durham, N.C., asked: “Based on what you found, which is really no clinical advantage but no safety issue either, are you having your patients with ST-segment MI administering crushed prasugrel now?”
Dr. Vlachojannis said they didn’t see any clinical impact but reiterated that high platelet reactivity was reduced by one-third. “If this now translates into a safer primary PCI procedure, we can’t say. The study wasn’t powered for this kind of endpoint. Is this enough to give you a recommendation, Sunil, I’m not sure.”
“What we know with COMPARE CRUSH, and this is important, is that we tried to give the medication as soon as possible and tried to give this medication in a formulation which has the most favorable pharmacodynamics profile, and we still see it’s not doing the job,” he added.
Fellow panelist Philippe Gabriel Steg, MD, Imperial College London, questioned whether treatment time may play a role in teasing out the relatively modest differences that platelet reactivity may have on clinical outcomes.
Dr. Vlachojannis said the time from symptom onset to first medical contact was very fast and similar to that in the ATLANTIC trial. “The short time intervals have certainly influenced the outcomes.”
Panelist Marco Valgimigli, MD, PhD, University Hospital Bern, Switzerland, followed up on the morphine issue, asking whether the investigators tested for an interaction between morphine or opioid use and platelet reactivity at the time of PCI.
“We haven’t looked into this but you probably have the ON-TIME 3 data in your mind when you’re asking this, where crushed ticagrelor given in the ambulance didn’t influence platelet reactivity at the time point of PCI,” Dr. Vlachojannis said. “We are going to look further into the data and certainly the platelet reactivity analysis is going to be very interesting in this data set.”
The study was an investigator-initiated trial sponsored by Maasstad Cardiovascular Research B.V. with unrestricted grants from Shanghai MicroPort Medical and Daiichi Sankyo. Dr. Vlachojannis declared receiving consulting fees from AstraZeneca, and research grants from Daiichi Sankyo and Shanghai MicroPort.
A version of this article originally appeared on Medscape.com.
Giving crushed prasugrel (Effient) to patients with ST-segment elevation myocardial infarction (STEMI) en route to a planned primary percutaneous coronary intervention (PCI) does not improve reperfusion rates, results of the COMPARE CRUSH trial show.
Patients assigned to prasugrel as crushed or integral tablets had similar rates of the study’s co-primary endpoints of thrombolysis in myocardial infarction (TIMI) 3 flow in the infarct-related artery at first angiography (31% vs. 32.7%; P = .64) and complete ST-segment resolution 1 hour post PCI (59.9% vs. 57.3%; P = .55).
“These findings hold in spite of the fact that crushed tablets of prasugrel led to more potent platelet inhibition compared with integral tablets,” said study author Georgios Vlachojannis, MD, PhD, University Medical Center Utrecht, the Netherlands.
“Whether faster and more potent antiplatelet therapy can improve coronary reperfusion in contemporary STEMI treatment regimen warrants further investigation.”
The results were reported in a late-breaking clinical science session at the Transcatheter Cardiovascular Therapeutics virtual annual meeting and published simultaneously in the journal Circulation. The meeting was sponsored by the Cardiovascular Research Foundation.
Fibrinolytics and glycoprotein IIb/IIIa inhibitors have demonstrated improved coronary reperfusion and outcomes when given pre hospital. Prior studies have also shown that early administration of a crushed P2Y12 inhibitor increases bioavailability and speeds platelet inhibition in STEMI patients, Dr. Vlachojannis noted.
However, the large randomized ATLANTIC trial, which compared prehospital to cath lab administration of crushed or integral ticagrelor (Brilinta), also found no difference in either TIMI flow in the infarct-related artery or ST-segment resolution.
Between November 2017 and March 2020, the investigator-initiated COMPARE CRUSH trial randomly allocated 727 STEMI patients (mean age, 62 years; 23% female) undergoing primary PCI to receive in the ambulance a 60-mg loading dose of prasugrel as either crushed or integral tablets.
The median time from onset of symptoms to first medical contact was 59 minutes, from first medical contact to study treatment 22 minutes, and from study treatment to primary PCI 57 minutes. These times did not differ between groups.
Platelet reactivity at the beginning of coronary angiography was significantly lower in the crushed group than in the integral group (P2Y12 reactivity units 192 vs. 227; P < .01). This resulted in significantly fewer patients in the crushed group with high platelet reactivity, defined as P2Y12 reactivity units >208, prior to the start of PCI (43.3% vs. 62.6%; P < .01).
There was no difference between the crushed and integral groups in the primary safety endpoint of TIMI major and BARC type 3 or higher bleeding within 48 hours after study treatment (0.4% vs 0.7%).
Death, MI, stroke, and urgent revascularization rates were also similar between groups during index hospitalization and at 30 days. Definite stent thrombosis occurred in one patient in the crushed group and two patients in the integral group.
In an exploratory analysis, the co-primary endpoint results were consistent across multiple subgroups, although there was a trend toward greater benefit on TIMI 3 flow in the crushed tablet group in patients older than age 75 years (P for interaction = .04), presenting with anterior infarction (P for interaction = .03), or with a history of prior PCI (P for interaction < .01).
“However, these results should be regarded as hypothesis-generating,” the authors wrote. “Opioids use in the ambulance was remarkably low in our study compared with the ATLANTIC trial, which might explain that we did not observe any significant interaction.”
Notably, morphine was used in half the ATLANTIC patients and was thought to have possibly delayed the absorption of ticagrelor.
During discussion following the presentation, Sunil V. Rao, MD, Duke University Medical Center, Durham, N.C., asked: “Based on what you found, which is really no clinical advantage but no safety issue either, are you having your patients with ST-segment MI administering crushed prasugrel now?”
Dr. Vlachojannis said they didn’t see any clinical impact but reiterated that high platelet reactivity was reduced by one-third. “If this now translates into a safer primary PCI procedure, we can’t say. The study wasn’t powered for this kind of endpoint. Is this enough to give you a recommendation, Sunil, I’m not sure.”
“What we know with COMPARE CRUSH, and this is important, is that we tried to give the medication as soon as possible and tried to give this medication in a formulation which has the most favorable pharmacodynamics profile, and we still see it’s not doing the job,” he added.
Fellow panelist Philippe Gabriel Steg, MD, Imperial College London, questioned whether treatment time may play a role in teasing out the relatively modest differences that platelet reactivity may have on clinical outcomes.
Dr. Vlachojannis said the time from symptom onset to first medical contact was very fast and similar to that in the ATLANTIC trial. “The short time intervals have certainly influenced the outcomes.”
Panelist Marco Valgimigli, MD, PhD, University Hospital Bern, Switzerland, followed up on the morphine issue, asking whether the investigators tested for an interaction between morphine or opioid use and platelet reactivity at the time of PCI.
“We haven’t looked into this but you probably have the ON-TIME 3 data in your mind when you’re asking this, where crushed ticagrelor given in the ambulance didn’t influence platelet reactivity at the time point of PCI,” Dr. Vlachojannis said. “We are going to look further into the data and certainly the platelet reactivity analysis is going to be very interesting in this data set.”
The study was an investigator-initiated trial sponsored by Maasstad Cardiovascular Research B.V. with unrestricted grants from Shanghai MicroPort Medical and Daiichi Sankyo. Dr. Vlachojannis declared receiving consulting fees from AstraZeneca, and research grants from Daiichi Sankyo and Shanghai MicroPort.
A version of this article originally appeared on Medscape.com.
Giving crushed prasugrel (Effient) to patients with ST-segment elevation myocardial infarction (STEMI) en route to a planned primary percutaneous coronary intervention (PCI) does not improve reperfusion rates, results of the COMPARE CRUSH trial show.
Patients assigned to prasugrel as crushed or integral tablets had similar rates of the study’s co-primary endpoints of thrombolysis in myocardial infarction (TIMI) 3 flow in the infarct-related artery at first angiography (31% vs. 32.7%; P = .64) and complete ST-segment resolution 1 hour post PCI (59.9% vs. 57.3%; P = .55).
“These findings hold in spite of the fact that crushed tablets of prasugrel led to more potent platelet inhibition compared with integral tablets,” said study author Georgios Vlachojannis, MD, PhD, University Medical Center Utrecht, the Netherlands.
“Whether faster and more potent antiplatelet therapy can improve coronary reperfusion in contemporary STEMI treatment regimen warrants further investigation.”
The results were reported in a late-breaking clinical science session at the Transcatheter Cardiovascular Therapeutics virtual annual meeting and published simultaneously in the journal Circulation. The meeting was sponsored by the Cardiovascular Research Foundation.
Fibrinolytics and glycoprotein IIb/IIIa inhibitors have demonstrated improved coronary reperfusion and outcomes when given pre hospital. Prior studies have also shown that early administration of a crushed P2Y12 inhibitor increases bioavailability and speeds platelet inhibition in STEMI patients, Dr. Vlachojannis noted.
However, the large randomized ATLANTIC trial, which compared prehospital to cath lab administration of crushed or integral ticagrelor (Brilinta), also found no difference in either TIMI flow in the infarct-related artery or ST-segment resolution.
Between November 2017 and March 2020, the investigator-initiated COMPARE CRUSH trial randomly allocated 727 STEMI patients (mean age, 62 years; 23% female) undergoing primary PCI to receive in the ambulance a 60-mg loading dose of prasugrel as either crushed or integral tablets.
The median time from onset of symptoms to first medical contact was 59 minutes, from first medical contact to study treatment 22 minutes, and from study treatment to primary PCI 57 minutes. These times did not differ between groups.
Platelet reactivity at the beginning of coronary angiography was significantly lower in the crushed group than in the integral group (P2Y12 reactivity units 192 vs. 227; P < .01). This resulted in significantly fewer patients in the crushed group with high platelet reactivity, defined as P2Y12 reactivity units >208, prior to the start of PCI (43.3% vs. 62.6%; P < .01).
There was no difference between the crushed and integral groups in the primary safety endpoint of TIMI major and BARC type 3 or higher bleeding within 48 hours after study treatment (0.4% vs 0.7%).
Death, MI, stroke, and urgent revascularization rates were also similar between groups during index hospitalization and at 30 days. Definite stent thrombosis occurred in one patient in the crushed group and two patients in the integral group.
In an exploratory analysis, the co-primary endpoint results were consistent across multiple subgroups, although there was a trend toward greater benefit on TIMI 3 flow in the crushed tablet group in patients older than age 75 years (P for interaction = .04), presenting with anterior infarction (P for interaction = .03), or with a history of prior PCI (P for interaction < .01).
“However, these results should be regarded as hypothesis-generating,” the authors wrote. “Opioids use in the ambulance was remarkably low in our study compared with the ATLANTIC trial, which might explain that we did not observe any significant interaction.”
Notably, morphine was used in half the ATLANTIC patients and was thought to have possibly delayed the absorption of ticagrelor.
During discussion following the presentation, Sunil V. Rao, MD, Duke University Medical Center, Durham, N.C., asked: “Based on what you found, which is really no clinical advantage but no safety issue either, are you having your patients with ST-segment MI administering crushed prasugrel now?”
Dr. Vlachojannis said they didn’t see any clinical impact but reiterated that high platelet reactivity was reduced by one-third. “If this now translates into a safer primary PCI procedure, we can’t say. The study wasn’t powered for this kind of endpoint. Is this enough to give you a recommendation, Sunil, I’m not sure.”
“What we know with COMPARE CRUSH, and this is important, is that we tried to give the medication as soon as possible and tried to give this medication in a formulation which has the most favorable pharmacodynamics profile, and we still see it’s not doing the job,” he added.
Fellow panelist Philippe Gabriel Steg, MD, Imperial College London, questioned whether treatment time may play a role in teasing out the relatively modest differences that platelet reactivity may have on clinical outcomes.
Dr. Vlachojannis said the time from symptom onset to first medical contact was very fast and similar to that in the ATLANTIC trial. “The short time intervals have certainly influenced the outcomes.”
Panelist Marco Valgimigli, MD, PhD, University Hospital Bern, Switzerland, followed up on the morphine issue, asking whether the investigators tested for an interaction between morphine or opioid use and platelet reactivity at the time of PCI.
“We haven’t looked into this but you probably have the ON-TIME 3 data in your mind when you’re asking this, where crushed ticagrelor given in the ambulance didn’t influence platelet reactivity at the time point of PCI,” Dr. Vlachojannis said. “We are going to look further into the data and certainly the platelet reactivity analysis is going to be very interesting in this data set.”
The study was an investigator-initiated trial sponsored by Maasstad Cardiovascular Research B.V. with unrestricted grants from Shanghai MicroPort Medical and Daiichi Sankyo. Dr. Vlachojannis declared receiving consulting fees from AstraZeneca, and research grants from Daiichi Sankyo and Shanghai MicroPort.
A version of this article originally appeared on Medscape.com.
Med student’s cardiac crisis a COVID-era medical mystery
Within minutes of her arrival at Community North Hospital in Indianapolis, Ramya Yeleti’s vital signs plummeted; her pulse was at 45 beats per minute and her ejection fraction was hovering near 10%. “I definitely thought there was a chance I would close my eyes and never open them again, but I only had a few seconds to process that,” she recalled. Then everything went black. Ramya fell unconscious as shock pads were positioned and a swarm of clinicians prepared to insert an Impella heart pump through a catheter into her aorta.
The third-year medical student and aspiring psychiatrist had been doing in-person neurology rotations in July when she began to experience fever and uncontrolled vomiting. Her initial thought was that she must have caught the flu from a patient.
After all, Ramya, along with her father Ram Yeleti, MD, mother Indira, and twin sister Divya, had all weathered COVID-19 in previous months and later tested positive for SARS-CoV-2 antibodies. The only family member who had been spared was her younger brother Rohith.
Indira suffered a severe case, requiring ICU care for 2 days but no ventilator; the others experienced mostly mild symptoms. Ramya — who was studying for her third-year board exams after classes at Marian University College of Osteopathic Medicine in Indianapolis went virtual in March — was left with lingering fatigue; however, her cough and muscle aches abated and her sense of taste and smell returned. When she started rotations, she thought her life was getting back to normal.
Ramya’s flu symptoms did not improve. A university-mandated rapid COVID test came back negative, but 2 more days of vomiting started to worry both her and her father, who is a cardiologist and chief physician executive at Community Health Network in Indianapolis. After Ramya felt some chest pain, she asked her father to listen to her heart. All sounded normal, and Ram prescribed ondansetron for her nausea.
But the antiemetic didn’t work, and by the next morning both father and daughter were convinced that they needed to head to the emergency department.
“I wanted to double-check if I was missing something about her being dehydrated,” Ram told Medscape Medical News. “Several things can cause protracted nausea, like hepatitis, appendicitis, or another infection. I feel terribly guilty I didn’t realize she had a heart condition.”
A surprising turn for the worst
Ramya’s subtle symptoms quickly gave way to the dramatic cardiac crisis that unfolded just after her arrival at Community North. “Her EKG looked absolutely horrendous, like a 75-year-old having a heart attack,” Ram said.
As a cardiologist, he knew his daughter’s situation was growing dire when he heard physicians shouting that the Impella wasn’t working and she needed extracorporeal membrane oxygenation (ECMO).
“At that point, I didn’t think she’d survive,” her father recalled. “We had 10 physicians in the room who worked on her for 5 hours to get her stabilized.”
“It was especially traumatic because, obviously, I knew exactly what was happening,” he added. “You can’t sugarcoat anything.”
After being connected to the heart–lung equipment, Ramya was transferred to IU Health Methodist Hospital, also in Indianapolis, where she was tested again for COVID-19. Unlike the rapid test administered just days earlier, the PCR assay came back positive.
“I knew she had acute myocarditis, but coronavirus never crossed my mind,” said Ram.
“As we were dealing with her heart, we were also dealing with this challenge: she was coming back positive for COVID-19 again,” said Roopa Rao, MD, the heart failure transplant cardiologist at IU Health who treated Ramya.
“We weren’t sure whether we were dealing with an active infection or dead virus” from her previous infection, Rao said, “so we started treating her like she had active COVID-19 and gave her remdesivir, convalescent plasma, and steroids, which was the protocol in our hospital.”
A biopsy of Ramya’s heart tissue, along with blood tests, indicated a past parvovirus infection. It’s possible that Ramya’s previous coronavirus infection made her susceptible to heart damage from a newer parvovirus infection, said Rao. Either virus, or both together, could have been responsible for the calamity.
Although it was unheard of during Ramya’s cardiac crisis in early August, evolving evidence now raises the possibility that she is one of a handful of people in the world to be reinfected with SARS-CoV-2. Also emerging are cases of COVID-related myocarditis and other extreme heart complications, particularly in young people.
“At the time, it wasn’t really clear if people could have another infection so quickly,” Rao told Medscape Medical News. “It is possible she is one of these rare individuals to have COVID-19 twice. I’m hoping at some point we will have some clarity.”
“I would favor a coinfection as probably the triggering factor for her sickness,” she said. “It may take some time, but like any other disease — and it doesn’t look like COVID will go away magically — I hope we’ll have some answers down the road.”
Another wrinkle
The next 48 hours brought astonishing news: Ramya’s heart function had rebounded to nearly normal, and her ejection fraction increased to about 45%. Heart transplantation wouldn’t be necessary, although Rao stood poised to follow through if ECMO only sustained, rather than improved, Ramya’s prognosis.
“Ramya was so sick that if she didn’t recover, the only option would be a heart transplant,” said Rao. “But we wanted to do everything to keep that heart.”
After steroid and COVID treatment, Ramya’s heart started to come back. “It didn’t make sense to me,” said Rao. “I don’t know what helped. If we hadn’t done ECMO, her heart probably wouldn’t have recovered, so I would say we have to support these patients and give them time for the heart to recover, even to the point of ECMO.”
Despite the good news, Ramya’s survival still hung in the balance. When she was disconnected from ECMO, clinicians discovered that the Impella device had caused a rare complication, damaging her mitral valve. The valve could be repaired surgically, but both Rao and Ram felt great trepidation at the prospect of cardiopulmonary bypass during the open-heart procedure.
“They would need to stop her heart and restart it, and I was concerned it would not restart,” Ram explained. “I didn’t like the idea of open-heart surgery, but my biggest fear was she was not going to survive it because of a really fresh, sick heart.”
The cardiologists’ fears did, in fact, come to pass: it took an hour to coax Ramya’s heart back at the end of surgery. But, just as the surgeon was preparing to reconnect Ramya to ECMO in desperation, “her heart recovered again,” Rao reported.
“Some things you never forget in life,” she said. “I can’t describe how everyone in the OR felt, all taking care of her. I told Ramya, ‘you are a fighter’.”
New strength
Six days would pass before Ramya woke up and learned of the astounding series of events that saved her. She knew “something was really wrong” because of the incision at the center of her chest, but learning she’d been on ECMO and the heart transplant list drove home how close to death she’d actually come.
“Most people don’t get off ECMO; they die on it,” she said. “And the chances of dying on the heart transplant list are very high. It was very strange to me that this was my story all of a sudden, when a week and a half earlier I was on rotation.”
Ongoing physical therapy over the past 3 months has transformed Ramya from a state of profound physical weakness to a place of relative strength. The now-fourth-year med student is turning 26 in November and is hungry to restart in-person rotations. Her downtime has been filled in part with researching myocarditis and collaborating with Rao on her own case study for journal publication.
But the mental trauma from her experience has girded her in ways she knows will make her stronger personally and professionally in the years ahead.
“It’s still very hard. I’m still recovering,” she acknowledged. “I described it to my therapist as an invisible wound on my brain.”
“When I came out of the hospital, I still had ECMO wounds, deep gashes on my legs that affected how fast and how long I could walk,” she said. “I felt like the same thing was going on my brain — a huge cut no one could see.”
Her intention to specialize in psychiatry has become more pressing now that Ramya has realized the impact of trauma on mental health.
“My body failing me was awful, but I could handle it,” she said. “Losing any part of my mind would have been way worse. I want to take care of that in my patients.”
This article first appeared on Medscape.com.
Within minutes of her arrival at Community North Hospital in Indianapolis, Ramya Yeleti’s vital signs plummeted; her pulse was at 45 beats per minute and her ejection fraction was hovering near 10%. “I definitely thought there was a chance I would close my eyes and never open them again, but I only had a few seconds to process that,” she recalled. Then everything went black. Ramya fell unconscious as shock pads were positioned and a swarm of clinicians prepared to insert an Impella heart pump through a catheter into her aorta.
The third-year medical student and aspiring psychiatrist had been doing in-person neurology rotations in July when she began to experience fever and uncontrolled vomiting. Her initial thought was that she must have caught the flu from a patient.
After all, Ramya, along with her father Ram Yeleti, MD, mother Indira, and twin sister Divya, had all weathered COVID-19 in previous months and later tested positive for SARS-CoV-2 antibodies. The only family member who had been spared was her younger brother Rohith.
Indira suffered a severe case, requiring ICU care for 2 days but no ventilator; the others experienced mostly mild symptoms. Ramya — who was studying for her third-year board exams after classes at Marian University College of Osteopathic Medicine in Indianapolis went virtual in March — was left with lingering fatigue; however, her cough and muscle aches abated and her sense of taste and smell returned. When she started rotations, she thought her life was getting back to normal.
Ramya’s flu symptoms did not improve. A university-mandated rapid COVID test came back negative, but 2 more days of vomiting started to worry both her and her father, who is a cardiologist and chief physician executive at Community Health Network in Indianapolis. After Ramya felt some chest pain, she asked her father to listen to her heart. All sounded normal, and Ram prescribed ondansetron for her nausea.
But the antiemetic didn’t work, and by the next morning both father and daughter were convinced that they needed to head to the emergency department.
“I wanted to double-check if I was missing something about her being dehydrated,” Ram told Medscape Medical News. “Several things can cause protracted nausea, like hepatitis, appendicitis, or another infection. I feel terribly guilty I didn’t realize she had a heart condition.”
A surprising turn for the worst
Ramya’s subtle symptoms quickly gave way to the dramatic cardiac crisis that unfolded just after her arrival at Community North. “Her EKG looked absolutely horrendous, like a 75-year-old having a heart attack,” Ram said.
As a cardiologist, he knew his daughter’s situation was growing dire when he heard physicians shouting that the Impella wasn’t working and she needed extracorporeal membrane oxygenation (ECMO).
“At that point, I didn’t think she’d survive,” her father recalled. “We had 10 physicians in the room who worked on her for 5 hours to get her stabilized.”
“It was especially traumatic because, obviously, I knew exactly what was happening,” he added. “You can’t sugarcoat anything.”
After being connected to the heart–lung equipment, Ramya was transferred to IU Health Methodist Hospital, also in Indianapolis, where she was tested again for COVID-19. Unlike the rapid test administered just days earlier, the PCR assay came back positive.
“I knew she had acute myocarditis, but coronavirus never crossed my mind,” said Ram.
“As we were dealing with her heart, we were also dealing with this challenge: she was coming back positive for COVID-19 again,” said Roopa Rao, MD, the heart failure transplant cardiologist at IU Health who treated Ramya.
“We weren’t sure whether we were dealing with an active infection or dead virus” from her previous infection, Rao said, “so we started treating her like she had active COVID-19 and gave her remdesivir, convalescent plasma, and steroids, which was the protocol in our hospital.”
A biopsy of Ramya’s heart tissue, along with blood tests, indicated a past parvovirus infection. It’s possible that Ramya’s previous coronavirus infection made her susceptible to heart damage from a newer parvovirus infection, said Rao. Either virus, or both together, could have been responsible for the calamity.
Although it was unheard of during Ramya’s cardiac crisis in early August, evolving evidence now raises the possibility that she is one of a handful of people in the world to be reinfected with SARS-CoV-2. Also emerging are cases of COVID-related myocarditis and other extreme heart complications, particularly in young people.
“At the time, it wasn’t really clear if people could have another infection so quickly,” Rao told Medscape Medical News. “It is possible she is one of these rare individuals to have COVID-19 twice. I’m hoping at some point we will have some clarity.”
“I would favor a coinfection as probably the triggering factor for her sickness,” she said. “It may take some time, but like any other disease — and it doesn’t look like COVID will go away magically — I hope we’ll have some answers down the road.”
Another wrinkle
The next 48 hours brought astonishing news: Ramya’s heart function had rebounded to nearly normal, and her ejection fraction increased to about 45%. Heart transplantation wouldn’t be necessary, although Rao stood poised to follow through if ECMO only sustained, rather than improved, Ramya’s prognosis.
“Ramya was so sick that if she didn’t recover, the only option would be a heart transplant,” said Rao. “But we wanted to do everything to keep that heart.”
After steroid and COVID treatment, Ramya’s heart started to come back. “It didn’t make sense to me,” said Rao. “I don’t know what helped. If we hadn’t done ECMO, her heart probably wouldn’t have recovered, so I would say we have to support these patients and give them time for the heart to recover, even to the point of ECMO.”
Despite the good news, Ramya’s survival still hung in the balance. When she was disconnected from ECMO, clinicians discovered that the Impella device had caused a rare complication, damaging her mitral valve. The valve could be repaired surgically, but both Rao and Ram felt great trepidation at the prospect of cardiopulmonary bypass during the open-heart procedure.
“They would need to stop her heart and restart it, and I was concerned it would not restart,” Ram explained. “I didn’t like the idea of open-heart surgery, but my biggest fear was she was not going to survive it because of a really fresh, sick heart.”
The cardiologists’ fears did, in fact, come to pass: it took an hour to coax Ramya’s heart back at the end of surgery. But, just as the surgeon was preparing to reconnect Ramya to ECMO in desperation, “her heart recovered again,” Rao reported.
“Some things you never forget in life,” she said. “I can’t describe how everyone in the OR felt, all taking care of her. I told Ramya, ‘you are a fighter’.”
New strength
Six days would pass before Ramya woke up and learned of the astounding series of events that saved her. She knew “something was really wrong” because of the incision at the center of her chest, but learning she’d been on ECMO and the heart transplant list drove home how close to death she’d actually come.
“Most people don’t get off ECMO; they die on it,” she said. “And the chances of dying on the heart transplant list are very high. It was very strange to me that this was my story all of a sudden, when a week and a half earlier I was on rotation.”
Ongoing physical therapy over the past 3 months has transformed Ramya from a state of profound physical weakness to a place of relative strength. The now-fourth-year med student is turning 26 in November and is hungry to restart in-person rotations. Her downtime has been filled in part with researching myocarditis and collaborating with Rao on her own case study for journal publication.
But the mental trauma from her experience has girded her in ways she knows will make her stronger personally and professionally in the years ahead.
“It’s still very hard. I’m still recovering,” she acknowledged. “I described it to my therapist as an invisible wound on my brain.”
“When I came out of the hospital, I still had ECMO wounds, deep gashes on my legs that affected how fast and how long I could walk,” she said. “I felt like the same thing was going on my brain — a huge cut no one could see.”
Her intention to specialize in psychiatry has become more pressing now that Ramya has realized the impact of trauma on mental health.
“My body failing me was awful, but I could handle it,” she said. “Losing any part of my mind would have been way worse. I want to take care of that in my patients.”
This article first appeared on Medscape.com.
Within minutes of her arrival at Community North Hospital in Indianapolis, Ramya Yeleti’s vital signs plummeted; her pulse was at 45 beats per minute and her ejection fraction was hovering near 10%. “I definitely thought there was a chance I would close my eyes and never open them again, but I only had a few seconds to process that,” she recalled. Then everything went black. Ramya fell unconscious as shock pads were positioned and a swarm of clinicians prepared to insert an Impella heart pump through a catheter into her aorta.
The third-year medical student and aspiring psychiatrist had been doing in-person neurology rotations in July when she began to experience fever and uncontrolled vomiting. Her initial thought was that she must have caught the flu from a patient.
After all, Ramya, along with her father Ram Yeleti, MD, mother Indira, and twin sister Divya, had all weathered COVID-19 in previous months and later tested positive for SARS-CoV-2 antibodies. The only family member who had been spared was her younger brother Rohith.
Indira suffered a severe case, requiring ICU care for 2 days but no ventilator; the others experienced mostly mild symptoms. Ramya — who was studying for her third-year board exams after classes at Marian University College of Osteopathic Medicine in Indianapolis went virtual in March — was left with lingering fatigue; however, her cough and muscle aches abated and her sense of taste and smell returned. When she started rotations, she thought her life was getting back to normal.
Ramya’s flu symptoms did not improve. A university-mandated rapid COVID test came back negative, but 2 more days of vomiting started to worry both her and her father, who is a cardiologist and chief physician executive at Community Health Network in Indianapolis. After Ramya felt some chest pain, she asked her father to listen to her heart. All sounded normal, and Ram prescribed ondansetron for her nausea.
But the antiemetic didn’t work, and by the next morning both father and daughter were convinced that they needed to head to the emergency department.
“I wanted to double-check if I was missing something about her being dehydrated,” Ram told Medscape Medical News. “Several things can cause protracted nausea, like hepatitis, appendicitis, or another infection. I feel terribly guilty I didn’t realize she had a heart condition.”
A surprising turn for the worst
Ramya’s subtle symptoms quickly gave way to the dramatic cardiac crisis that unfolded just after her arrival at Community North. “Her EKG looked absolutely horrendous, like a 75-year-old having a heart attack,” Ram said.
As a cardiologist, he knew his daughter’s situation was growing dire when he heard physicians shouting that the Impella wasn’t working and she needed extracorporeal membrane oxygenation (ECMO).
“At that point, I didn’t think she’d survive,” her father recalled. “We had 10 physicians in the room who worked on her for 5 hours to get her stabilized.”
“It was especially traumatic because, obviously, I knew exactly what was happening,” he added. “You can’t sugarcoat anything.”
After being connected to the heart–lung equipment, Ramya was transferred to IU Health Methodist Hospital, also in Indianapolis, where she was tested again for COVID-19. Unlike the rapid test administered just days earlier, the PCR assay came back positive.
“I knew she had acute myocarditis, but coronavirus never crossed my mind,” said Ram.
“As we were dealing with her heart, we were also dealing with this challenge: she was coming back positive for COVID-19 again,” said Roopa Rao, MD, the heart failure transplant cardiologist at IU Health who treated Ramya.
“We weren’t sure whether we were dealing with an active infection or dead virus” from her previous infection, Rao said, “so we started treating her like she had active COVID-19 and gave her remdesivir, convalescent plasma, and steroids, which was the protocol in our hospital.”
A biopsy of Ramya’s heart tissue, along with blood tests, indicated a past parvovirus infection. It’s possible that Ramya’s previous coronavirus infection made her susceptible to heart damage from a newer parvovirus infection, said Rao. Either virus, or both together, could have been responsible for the calamity.
Although it was unheard of during Ramya’s cardiac crisis in early August, evolving evidence now raises the possibility that she is one of a handful of people in the world to be reinfected with SARS-CoV-2. Also emerging are cases of COVID-related myocarditis and other extreme heart complications, particularly in young people.
“At the time, it wasn’t really clear if people could have another infection so quickly,” Rao told Medscape Medical News. “It is possible she is one of these rare individuals to have COVID-19 twice. I’m hoping at some point we will have some clarity.”
“I would favor a coinfection as probably the triggering factor for her sickness,” she said. “It may take some time, but like any other disease — and it doesn’t look like COVID will go away magically — I hope we’ll have some answers down the road.”
Another wrinkle
The next 48 hours brought astonishing news: Ramya’s heart function had rebounded to nearly normal, and her ejection fraction increased to about 45%. Heart transplantation wouldn’t be necessary, although Rao stood poised to follow through if ECMO only sustained, rather than improved, Ramya’s prognosis.
“Ramya was so sick that if she didn’t recover, the only option would be a heart transplant,” said Rao. “But we wanted to do everything to keep that heart.”
After steroid and COVID treatment, Ramya’s heart started to come back. “It didn’t make sense to me,” said Rao. “I don’t know what helped. If we hadn’t done ECMO, her heart probably wouldn’t have recovered, so I would say we have to support these patients and give them time for the heart to recover, even to the point of ECMO.”
Despite the good news, Ramya’s survival still hung in the balance. When she was disconnected from ECMO, clinicians discovered that the Impella device had caused a rare complication, damaging her mitral valve. The valve could be repaired surgically, but both Rao and Ram felt great trepidation at the prospect of cardiopulmonary bypass during the open-heart procedure.
“They would need to stop her heart and restart it, and I was concerned it would not restart,” Ram explained. “I didn’t like the idea of open-heart surgery, but my biggest fear was she was not going to survive it because of a really fresh, sick heart.”
The cardiologists’ fears did, in fact, come to pass: it took an hour to coax Ramya’s heart back at the end of surgery. But, just as the surgeon was preparing to reconnect Ramya to ECMO in desperation, “her heart recovered again,” Rao reported.
“Some things you never forget in life,” she said. “I can’t describe how everyone in the OR felt, all taking care of her. I told Ramya, ‘you are a fighter’.”
New strength
Six days would pass before Ramya woke up and learned of the astounding series of events that saved her. She knew “something was really wrong” because of the incision at the center of her chest, but learning she’d been on ECMO and the heart transplant list drove home how close to death she’d actually come.
“Most people don’t get off ECMO; they die on it,” she said. “And the chances of dying on the heart transplant list are very high. It was very strange to me that this was my story all of a sudden, when a week and a half earlier I was on rotation.”
Ongoing physical therapy over the past 3 months has transformed Ramya from a state of profound physical weakness to a place of relative strength. The now-fourth-year med student is turning 26 in November and is hungry to restart in-person rotations. Her downtime has been filled in part with researching myocarditis and collaborating with Rao on her own case study for journal publication.
But the mental trauma from her experience has girded her in ways she knows will make her stronger personally and professionally in the years ahead.
“It’s still very hard. I’m still recovering,” she acknowledged. “I described it to my therapist as an invisible wound on my brain.”
“When I came out of the hospital, I still had ECMO wounds, deep gashes on my legs that affected how fast and how long I could walk,” she said. “I felt like the same thing was going on my brain — a huge cut no one could see.”
Her intention to specialize in psychiatry has become more pressing now that Ramya has realized the impact of trauma on mental health.
“My body failing me was awful, but I could handle it,” she said. “Losing any part of my mind would have been way worse. I want to take care of that in my patients.”
This article first appeared on Medscape.com.
About 17% of COVID-19 survivors retest positive in follow-up study
For reasons unknown, about one in six people who recovered from COVID-19 subsequently retested positive at least 2 weeks later, researchers reported in a study in Italy.
Sore throat and rhinitis were the only symptoms associated with a positive result. “Patients who continued to have respiratory symptoms, especially, were more likely to have a new positive test result,” lead author Francesco Landi, MD, PhD, said in an interview.
“This suggests the persistence of respiratory symptoms should not be underestimated and should be adequately assessed in all patients considered recovered from COVID-19,” he said.
“The study results are interesting,” Akiko Iwasaki, PhD, an immunobiologist at Yale University and the Howard Hughes Medical Institute, both in New Haven, Conn.,, said in an interview. “There are other reports of RNA detection postdischarge, but this study ... found that only two symptoms out of many – sore throat and rhinitis – were higher in those with PCR [polymerase chain reaction]-positive status.”
The study was published online Sept. 18 in the American Journal of Preventive Medicine.
The findings could carry important implications for people who continue to be symptomatic. “It is reasonable to be cautious and avoid close contact with others, wear a face mask, and possibly undergo an additional nasopharyngeal swab,” said Dr. Landi, associate professor of internal medicine at Catholic University of the Sacred Heart in Rome.
“One of most interesting findings is that persistent symptoms do not correlate with PCR positivity, suggesting that symptoms are in many cases not due to ongoing viral replication,” Jonathan Karn, PhD, professor and chair of the department of molecular biology and microbiology at Case Western Reserve University, Cleveland, said in an interview.
“The key technical problem, which they have discussed, is that a viral RNA signal in the PCR assay does not necessarily mean that infectious virus is present,” Dr. Karn said. He added that new comprehensive viral RNA analyses would be needed to answer this question.
Official COVID-19 recovery
To identify risk factors and COVID-19 survivors more likely to retest positive, Dr. Landi and members of the Gemelli Against COVID-19 Post-Acute Care Study Group evaluated 131 people after hospital discharge.
All participants met World Health Organization criteria for release from isolation, including two negative test results at least 24 hours apart, and were studied between April 21 and May 21. Mean age was 56 and 39% were women. Only a slightly higher mean body mass index of 27.6 kg/m2 in the positive group versus 25.9 in the negative group, was significant.
Although 51% of survivors reported fatigue, 44% had dyspnea, and 17% were coughing, the rates did not differ significantly between groups. In contrast, 18% of positive survivors and 4% of negative survivors had a sore throat (P = .04), and 27% versus 12%, respectively, reported rhinitis (P = .05).
People returned for follow-up visits a mean 17 days after the second negative swab test.
Asymptomatic COVID-19 carriers
“These findings indicate that a noteworthy rate of recovered patients with COVID-19 could still be asymptomatic carriers of the virus,” the researchers noted in the paper. “Even in the absence of specific guidelines, the 22 patients who tested positive for COVID-19 again were suggested to quarantine for a second time.”
No family member or close contact of the positive survivors reported SARS-CoV-2 infection. All patients continued to wear masks and observe social distancing recommendations, which makes it “very difficult to affirm whether these patients were really contagious,” the researchers noted.
Next steps
Evaluating all COVID-19 survivors to identify any who retest positive “will be a crucial contribution to a better understanding of both the natural history of COVID-19 as well as the public health implications of viral shedding,” the authors wrote.
One study limitation is that the reverse transcriptase–PCR test reveals genetic sequences specific to COVID-19. “It is important to underline that this is not a viral culture and cannot determine whether the virus is viable and transmissible,” the researchers noted.
“In this respect, we are trying to better understand if the persistence of long-time positive [reverse transcriptase]–PCR test for COVID-19 is really correlated to a potential contagiousness,” they added.
Dr. Landi and colleagues said their findings should be considered preliminary, and larger data samples are warranted to validate the results.
Dr. Landi and Dr. Karn disclosed no relevant financial relationships. Dr. Iwasaki disclosed a research grant from Condair, a 5% or greater equity interest in RIGImmune, and income of $250 or more from PureTec.
A version of this article originally appeared on Medscape.com.
For reasons unknown, about one in six people who recovered from COVID-19 subsequently retested positive at least 2 weeks later, researchers reported in a study in Italy.
Sore throat and rhinitis were the only symptoms associated with a positive result. “Patients who continued to have respiratory symptoms, especially, were more likely to have a new positive test result,” lead author Francesco Landi, MD, PhD, said in an interview.
“This suggests the persistence of respiratory symptoms should not be underestimated and should be adequately assessed in all patients considered recovered from COVID-19,” he said.
“The study results are interesting,” Akiko Iwasaki, PhD, an immunobiologist at Yale University and the Howard Hughes Medical Institute, both in New Haven, Conn.,, said in an interview. “There are other reports of RNA detection postdischarge, but this study ... found that only two symptoms out of many – sore throat and rhinitis – were higher in those with PCR [polymerase chain reaction]-positive status.”
The study was published online Sept. 18 in the American Journal of Preventive Medicine.
The findings could carry important implications for people who continue to be symptomatic. “It is reasonable to be cautious and avoid close contact with others, wear a face mask, and possibly undergo an additional nasopharyngeal swab,” said Dr. Landi, associate professor of internal medicine at Catholic University of the Sacred Heart in Rome.
“One of most interesting findings is that persistent symptoms do not correlate with PCR positivity, suggesting that symptoms are in many cases not due to ongoing viral replication,” Jonathan Karn, PhD, professor and chair of the department of molecular biology and microbiology at Case Western Reserve University, Cleveland, said in an interview.
“The key technical problem, which they have discussed, is that a viral RNA signal in the PCR assay does not necessarily mean that infectious virus is present,” Dr. Karn said. He added that new comprehensive viral RNA analyses would be needed to answer this question.
Official COVID-19 recovery
To identify risk factors and COVID-19 survivors more likely to retest positive, Dr. Landi and members of the Gemelli Against COVID-19 Post-Acute Care Study Group evaluated 131 people after hospital discharge.
All participants met World Health Organization criteria for release from isolation, including two negative test results at least 24 hours apart, and were studied between April 21 and May 21. Mean age was 56 and 39% were women. Only a slightly higher mean body mass index of 27.6 kg/m2 in the positive group versus 25.9 in the negative group, was significant.
Although 51% of survivors reported fatigue, 44% had dyspnea, and 17% were coughing, the rates did not differ significantly between groups. In contrast, 18% of positive survivors and 4% of negative survivors had a sore throat (P = .04), and 27% versus 12%, respectively, reported rhinitis (P = .05).
People returned for follow-up visits a mean 17 days after the second negative swab test.
Asymptomatic COVID-19 carriers
“These findings indicate that a noteworthy rate of recovered patients with COVID-19 could still be asymptomatic carriers of the virus,” the researchers noted in the paper. “Even in the absence of specific guidelines, the 22 patients who tested positive for COVID-19 again were suggested to quarantine for a second time.”
No family member or close contact of the positive survivors reported SARS-CoV-2 infection. All patients continued to wear masks and observe social distancing recommendations, which makes it “very difficult to affirm whether these patients were really contagious,” the researchers noted.
Next steps
Evaluating all COVID-19 survivors to identify any who retest positive “will be a crucial contribution to a better understanding of both the natural history of COVID-19 as well as the public health implications of viral shedding,” the authors wrote.
One study limitation is that the reverse transcriptase–PCR test reveals genetic sequences specific to COVID-19. “It is important to underline that this is not a viral culture and cannot determine whether the virus is viable and transmissible,” the researchers noted.
“In this respect, we are trying to better understand if the persistence of long-time positive [reverse transcriptase]–PCR test for COVID-19 is really correlated to a potential contagiousness,” they added.
Dr. Landi and colleagues said their findings should be considered preliminary, and larger data samples are warranted to validate the results.
Dr. Landi and Dr. Karn disclosed no relevant financial relationships. Dr. Iwasaki disclosed a research grant from Condair, a 5% or greater equity interest in RIGImmune, and income of $250 or more from PureTec.
A version of this article originally appeared on Medscape.com.
For reasons unknown, about one in six people who recovered from COVID-19 subsequently retested positive at least 2 weeks later, researchers reported in a study in Italy.
Sore throat and rhinitis were the only symptoms associated with a positive result. “Patients who continued to have respiratory symptoms, especially, were more likely to have a new positive test result,” lead author Francesco Landi, MD, PhD, said in an interview.
“This suggests the persistence of respiratory symptoms should not be underestimated and should be adequately assessed in all patients considered recovered from COVID-19,” he said.
“The study results are interesting,” Akiko Iwasaki, PhD, an immunobiologist at Yale University and the Howard Hughes Medical Institute, both in New Haven, Conn.,, said in an interview. “There are other reports of RNA detection postdischarge, but this study ... found that only two symptoms out of many – sore throat and rhinitis – were higher in those with PCR [polymerase chain reaction]-positive status.”
The study was published online Sept. 18 in the American Journal of Preventive Medicine.
The findings could carry important implications for people who continue to be symptomatic. “It is reasonable to be cautious and avoid close contact with others, wear a face mask, and possibly undergo an additional nasopharyngeal swab,” said Dr. Landi, associate professor of internal medicine at Catholic University of the Sacred Heart in Rome.
“One of most interesting findings is that persistent symptoms do not correlate with PCR positivity, suggesting that symptoms are in many cases not due to ongoing viral replication,” Jonathan Karn, PhD, professor and chair of the department of molecular biology and microbiology at Case Western Reserve University, Cleveland, said in an interview.
“The key technical problem, which they have discussed, is that a viral RNA signal in the PCR assay does not necessarily mean that infectious virus is present,” Dr. Karn said. He added that new comprehensive viral RNA analyses would be needed to answer this question.
Official COVID-19 recovery
To identify risk factors and COVID-19 survivors more likely to retest positive, Dr. Landi and members of the Gemelli Against COVID-19 Post-Acute Care Study Group evaluated 131 people after hospital discharge.
All participants met World Health Organization criteria for release from isolation, including two negative test results at least 24 hours apart, and were studied between April 21 and May 21. Mean age was 56 and 39% were women. Only a slightly higher mean body mass index of 27.6 kg/m2 in the positive group versus 25.9 in the negative group, was significant.
Although 51% of survivors reported fatigue, 44% had dyspnea, and 17% were coughing, the rates did not differ significantly between groups. In contrast, 18% of positive survivors and 4% of negative survivors had a sore throat (P = .04), and 27% versus 12%, respectively, reported rhinitis (P = .05).
People returned for follow-up visits a mean 17 days after the second negative swab test.
Asymptomatic COVID-19 carriers
“These findings indicate that a noteworthy rate of recovered patients with COVID-19 could still be asymptomatic carriers of the virus,” the researchers noted in the paper. “Even in the absence of specific guidelines, the 22 patients who tested positive for COVID-19 again were suggested to quarantine for a second time.”
No family member or close contact of the positive survivors reported SARS-CoV-2 infection. All patients continued to wear masks and observe social distancing recommendations, which makes it “very difficult to affirm whether these patients were really contagious,” the researchers noted.
Next steps
Evaluating all COVID-19 survivors to identify any who retest positive “will be a crucial contribution to a better understanding of both the natural history of COVID-19 as well as the public health implications of viral shedding,” the authors wrote.
One study limitation is that the reverse transcriptase–PCR test reveals genetic sequences specific to COVID-19. “It is important to underline that this is not a viral culture and cannot determine whether the virus is viable and transmissible,” the researchers noted.
“In this respect, we are trying to better understand if the persistence of long-time positive [reverse transcriptase]–PCR test for COVID-19 is really correlated to a potential contagiousness,” they added.
Dr. Landi and colleagues said their findings should be considered preliminary, and larger data samples are warranted to validate the results.
Dr. Landi and Dr. Karn disclosed no relevant financial relationships. Dr. Iwasaki disclosed a research grant from Condair, a 5% or greater equity interest in RIGImmune, and income of $250 or more from PureTec.
A version of this article originally appeared on Medscape.com.
CDC panel takes on COVID vaccine rollout, risks, and side effects
Federal advisers who will help determine which Americans get the first COVID vaccines took an in-depth look Oct. 30 at the challenges they face in selecting priority groups.
The Advisory Committee on Immunization Practices (ACIP) of the Centers for Disease Control and Prevention (CDC) will face two key decisions once a COVID vaccine wins clearance from the US Food and Drug Administration (FDA).
ACIP will need to decide whether to recommend its use in adults (the age group in which vaccines are currently being tested). The group will also need to offer direction on which groups should get priority in vaccine allocation, inasmuch as early supplies will not be sufficient to vaccinate everyone.
At the Oct. 30 meeting, CDC’s Kathleen Dooling, MD, MPH, suggested that ACIP plan on tackling these issues as two separate questions when it comes time to weigh in on an approved vaccine. Although there was no formal vote among ACIP members at the meeting, Dooling’s proposal for tackling a future recommendation in a two-part fashion drew positive feedback.
ACIP member Katherine A. Poehling, MD, MPH, suggested that the panel and CDC be ready to reexamine the situation frequently regarding COVID vaccination. “Perhaps we could think about reviewing data on a monthly basis and updating the recommendation, so that we can account for the concerns and balance both the benefits and the [potential] harm,” Poehling said.
Dooling agreed. “Both the vaccine recommendation and allocation will be revisited in what is a very dynamic situation,” Dooling replied to Poehling. “So all new evidence will be brought to ACIP, and certainly the allocation as vaccine distribution proceeds will need to be adjusted accordingly.”
Ethics and limited evidence
During the meeting, ACIP members repeatedly expressed discomfort with the prospect of having to weigh in on widespread use of COVID vaccines on the basis of limited evidence.
Within months, FDA may opt for a special clearance, known as an emergency use authorization (EUA), for one or more of the experimental COVID vaccines now in advanced testing. Many of FDA’s past EUA clearances were granted for test kits. For those EUA approvals, the agency considered risks of false results but not longer-term, direct harm to patients from these products.
With a COVID vaccine, there will be strong pressure to distribute doses as quickly as possible with the hope of curbing the pandemic, which has already led to more than 229,000 deaths in the United States alone and has disrupted lives and economies around the world. But questions will persist about the possibility of serious complications from these vaccines, ACIP members noted.
“My personal struggle is the ethical side and how to balance these two,” said ACIP member Robert L. Atmar, MD, of Baylor College of Medicine, Houston, Texas, who noted that he expects his fellow panelists to share this concern.
Currently, four experimental COVID vaccines likely to be used in the United States have advanced to phase 3 testing. Pfizer Inc and BioNtech have enrolled more than 42,000 participants in a test of their candidate, BNT162b2 vaccine, and rival Moderna has enrolled about 30,000 participants in a test of its mRNA-1273 vaccine, CDC staff said.
The other two advanced COVID vaccine candidates have overcome recent hurdles. AstraZeneca Plc on Oct. 23 announced that FDA had removed a hold on the testing of its AZD1222 vaccine candidate; the trial will enroll approximately 30,000 people. Johnson & Johnson’s Janssen unit also announced that day the lifting of a safety pause for its Ad26.COV2.S vaccine; the phase 3 trial for that vaccine will enroll approximately 60,000 volunteers. Federal agencies, states, and territories have developed plans for future distribution of COVID vaccines, CDC staff said in briefing materials for today’s ACIP meeting.
Several ACIP members raised many of the same concerns that members of an FDA advisory committee raised at a meeting earlier in October. ACIP and FDA advisers honed in on the FDA’s decision to set a median follow-up duration of 2 months in phase 3 trials in connection with expected EUA applications for COVID-19 vaccines.
“I struggle with following people for 2 months after their second vaccination as a time point to start making final decisions about safety,” said ACIP member Sharon E. Frey, MD, a professor at St. Louis University School of Medicine, St. Louis, Missouri. “I just want to put that out there.”
Medical front line, then who?
There is consensus that healthcare workers be in the first stage ― Phase 1 ― of distribution. That recommendation was made in a report from the National Academies of Sciences, Engineering, and Medicine (NASEM). Phase 1A would include first responders; Phase 1B might include people of all ages who have two or more comorbidities that put them at significantly higher risk for COVID-19 or death, as well as older adults living in congregate or overcrowded settings, the NASEM report said.
A presentation from the CDC’s Matthew Biggerstaff, ScD, MPH, underscored challenges in distributing what are expected to be limited initial supplies of COVID vaccines.
Biggerstaff showed several scenarios the CDC’s Data, Analytics, and Modeling Task Force had studied. The initial allocation of vaccines would be for healthcare workers, followed by what the CDC called Phase 1B.
Choices for a rollout may include next giving COVID vaccines to people at high risk, such as persons who have one or more chronic medical conditions, including heart disease, diabetes, kidney disease, or obesity. Other options for the rollout could be to vaccinate people aged 65 years and older or essential workers whose employment puts them in contact with the public, thus raising the risk of contracting the virus.
The CDC’s research found that the greatest impact in preventing death was to initially vaccinate adults aged 65 and older in Phase 1B. The agency staff described this approach as likely to result in an about “1 to 11% increase in averted deaths across the scenarios.”
Initially vaccinating essential workers or high-risk adults in Phase 1B would avert the most infections. The agency staff described this approach as yielding about “1 to 5% increase in averted infections across the scenarios,” Biggerstaff said during his presentation.
The following are other findings of the CDC staff:
The earlier the vaccine rollout relative to increasing transmission, the greater the averted percentage and differences between the strategies.
Differences were not substantial in some scenarios.
The need to continue efforts to slow the spread of COVID-19 should be emphasized.
Adverse effects
ACIP members also heard about strategies for tracking potential side effects of future vaccines. A presentation by Tom Shimabukuro, MD, MPH, MBA, from the CDC’s COVID-19 Vaccine Task Force/Vaccine Safety Team, included details about a new smartphone-based active surveillance program for COVID-19 vaccine safety.
Known as v-safe, this system would use Web-based survey monitoring and incorporate text messaging. It would conduct electronic health checks on vaccine recipients, which would occur daily during the first week post vaccination and weekly thereafter for 6 weeks from the time of vaccination.
Clinicians “can play an important role in helping CDC enroll patients in v-safe at the time of vaccination,” Shimabukuro noted in his presentation. This would add another task, though, for clinicians, the CDC staff noted.
Pregnancy and breastfeeding are special concerns
Of special concern with the rollout of a COVID vaccine are recommendations regarding pregnancy and breastfeeding. Women constitute about 75% of the healthcare workforce, CDC staff noted.
At the time the initial ACIP COVID vaccination recommendations are made, there could be approximately 330,000 healthcare personnel who are pregnant or who have recently given birth. Available data indicate potentially increased risks for severe maternal illness and preterm birth associated with SARS-CoV-2 infection, said CDC’s Megan Wallace, DrPH, MPH, in a presentation for the Friday meeting.
In an Oct. 27 letter to ACIP, Chair Jose Romero, the American College of Obstetricians and Gynecologists (ACOG), urged the panel to ensure that pregnant women and new mothers in the healthcare workforce have priority access to a COVID vaccine. Pregnant and lactating women were “noticeably and alarmingly absent from the NASEM vaccine allocation plan for COVID-19,” wrote Christopher M. Zahn, MD, vice president for practice activities at ACOG, in the letter to Romero.
“ACOG urges ACIP to incorporate pregnant and lactating women clearly and explicitly into its COVID-19 vaccine allocation and prioritization framework,” Zahn wrote. “Should an Emergency Use Authorization be executed for one or more COVID-19 vaccines and provide a permissive recommendation for pregnant and lactating women, pregnant health care workers, pregnant first responders, and pregnant individuals with underlying conditions should be prioritized for vaccination alongside their non-pregnant peers.”
This article first appeared on Medscape.com.
Federal advisers who will help determine which Americans get the first COVID vaccines took an in-depth look Oct. 30 at the challenges they face in selecting priority groups.
The Advisory Committee on Immunization Practices (ACIP) of the Centers for Disease Control and Prevention (CDC) will face two key decisions once a COVID vaccine wins clearance from the US Food and Drug Administration (FDA).
ACIP will need to decide whether to recommend its use in adults (the age group in which vaccines are currently being tested). The group will also need to offer direction on which groups should get priority in vaccine allocation, inasmuch as early supplies will not be sufficient to vaccinate everyone.
At the Oct. 30 meeting, CDC’s Kathleen Dooling, MD, MPH, suggested that ACIP plan on tackling these issues as two separate questions when it comes time to weigh in on an approved vaccine. Although there was no formal vote among ACIP members at the meeting, Dooling’s proposal for tackling a future recommendation in a two-part fashion drew positive feedback.
ACIP member Katherine A. Poehling, MD, MPH, suggested that the panel and CDC be ready to reexamine the situation frequently regarding COVID vaccination. “Perhaps we could think about reviewing data on a monthly basis and updating the recommendation, so that we can account for the concerns and balance both the benefits and the [potential] harm,” Poehling said.
Dooling agreed. “Both the vaccine recommendation and allocation will be revisited in what is a very dynamic situation,” Dooling replied to Poehling. “So all new evidence will be brought to ACIP, and certainly the allocation as vaccine distribution proceeds will need to be adjusted accordingly.”
Ethics and limited evidence
During the meeting, ACIP members repeatedly expressed discomfort with the prospect of having to weigh in on widespread use of COVID vaccines on the basis of limited evidence.
Within months, FDA may opt for a special clearance, known as an emergency use authorization (EUA), for one or more of the experimental COVID vaccines now in advanced testing. Many of FDA’s past EUA clearances were granted for test kits. For those EUA approvals, the agency considered risks of false results but not longer-term, direct harm to patients from these products.
With a COVID vaccine, there will be strong pressure to distribute doses as quickly as possible with the hope of curbing the pandemic, which has already led to more than 229,000 deaths in the United States alone and has disrupted lives and economies around the world. But questions will persist about the possibility of serious complications from these vaccines, ACIP members noted.
“My personal struggle is the ethical side and how to balance these two,” said ACIP member Robert L. Atmar, MD, of Baylor College of Medicine, Houston, Texas, who noted that he expects his fellow panelists to share this concern.
Currently, four experimental COVID vaccines likely to be used in the United States have advanced to phase 3 testing. Pfizer Inc and BioNtech have enrolled more than 42,000 participants in a test of their candidate, BNT162b2 vaccine, and rival Moderna has enrolled about 30,000 participants in a test of its mRNA-1273 vaccine, CDC staff said.
The other two advanced COVID vaccine candidates have overcome recent hurdles. AstraZeneca Plc on Oct. 23 announced that FDA had removed a hold on the testing of its AZD1222 vaccine candidate; the trial will enroll approximately 30,000 people. Johnson & Johnson’s Janssen unit also announced that day the lifting of a safety pause for its Ad26.COV2.S vaccine; the phase 3 trial for that vaccine will enroll approximately 60,000 volunteers. Federal agencies, states, and territories have developed plans for future distribution of COVID vaccines, CDC staff said in briefing materials for today’s ACIP meeting.
Several ACIP members raised many of the same concerns that members of an FDA advisory committee raised at a meeting earlier in October. ACIP and FDA advisers honed in on the FDA’s decision to set a median follow-up duration of 2 months in phase 3 trials in connection with expected EUA applications for COVID-19 vaccines.
“I struggle with following people for 2 months after their second vaccination as a time point to start making final decisions about safety,” said ACIP member Sharon E. Frey, MD, a professor at St. Louis University School of Medicine, St. Louis, Missouri. “I just want to put that out there.”
Medical front line, then who?
There is consensus that healthcare workers be in the first stage ― Phase 1 ― of distribution. That recommendation was made in a report from the National Academies of Sciences, Engineering, and Medicine (NASEM). Phase 1A would include first responders; Phase 1B might include people of all ages who have two or more comorbidities that put them at significantly higher risk for COVID-19 or death, as well as older adults living in congregate or overcrowded settings, the NASEM report said.
A presentation from the CDC’s Matthew Biggerstaff, ScD, MPH, underscored challenges in distributing what are expected to be limited initial supplies of COVID vaccines.
Biggerstaff showed several scenarios the CDC’s Data, Analytics, and Modeling Task Force had studied. The initial allocation of vaccines would be for healthcare workers, followed by what the CDC called Phase 1B.
Choices for a rollout may include next giving COVID vaccines to people at high risk, such as persons who have one or more chronic medical conditions, including heart disease, diabetes, kidney disease, or obesity. Other options for the rollout could be to vaccinate people aged 65 years and older or essential workers whose employment puts them in contact with the public, thus raising the risk of contracting the virus.
The CDC’s research found that the greatest impact in preventing death was to initially vaccinate adults aged 65 and older in Phase 1B. The agency staff described this approach as likely to result in an about “1 to 11% increase in averted deaths across the scenarios.”
Initially vaccinating essential workers or high-risk adults in Phase 1B would avert the most infections. The agency staff described this approach as yielding about “1 to 5% increase in averted infections across the scenarios,” Biggerstaff said during his presentation.
The following are other findings of the CDC staff:
The earlier the vaccine rollout relative to increasing transmission, the greater the averted percentage and differences between the strategies.
Differences were not substantial in some scenarios.
The need to continue efforts to slow the spread of COVID-19 should be emphasized.
Adverse effects
ACIP members also heard about strategies for tracking potential side effects of future vaccines. A presentation by Tom Shimabukuro, MD, MPH, MBA, from the CDC’s COVID-19 Vaccine Task Force/Vaccine Safety Team, included details about a new smartphone-based active surveillance program for COVID-19 vaccine safety.
Known as v-safe, this system would use Web-based survey monitoring and incorporate text messaging. It would conduct electronic health checks on vaccine recipients, which would occur daily during the first week post vaccination and weekly thereafter for 6 weeks from the time of vaccination.
Clinicians “can play an important role in helping CDC enroll patients in v-safe at the time of vaccination,” Shimabukuro noted in his presentation. This would add another task, though, for clinicians, the CDC staff noted.
Pregnancy and breastfeeding are special concerns
Of special concern with the rollout of a COVID vaccine are recommendations regarding pregnancy and breastfeeding. Women constitute about 75% of the healthcare workforce, CDC staff noted.
At the time the initial ACIP COVID vaccination recommendations are made, there could be approximately 330,000 healthcare personnel who are pregnant or who have recently given birth. Available data indicate potentially increased risks for severe maternal illness and preterm birth associated with SARS-CoV-2 infection, said CDC’s Megan Wallace, DrPH, MPH, in a presentation for the Friday meeting.
In an Oct. 27 letter to ACIP, Chair Jose Romero, the American College of Obstetricians and Gynecologists (ACOG), urged the panel to ensure that pregnant women and new mothers in the healthcare workforce have priority access to a COVID vaccine. Pregnant and lactating women were “noticeably and alarmingly absent from the NASEM vaccine allocation plan for COVID-19,” wrote Christopher M. Zahn, MD, vice president for practice activities at ACOG, in the letter to Romero.
“ACOG urges ACIP to incorporate pregnant and lactating women clearly and explicitly into its COVID-19 vaccine allocation and prioritization framework,” Zahn wrote. “Should an Emergency Use Authorization be executed for one or more COVID-19 vaccines and provide a permissive recommendation for pregnant and lactating women, pregnant health care workers, pregnant first responders, and pregnant individuals with underlying conditions should be prioritized for vaccination alongside their non-pregnant peers.”
This article first appeared on Medscape.com.
Federal advisers who will help determine which Americans get the first COVID vaccines took an in-depth look Oct. 30 at the challenges they face in selecting priority groups.
The Advisory Committee on Immunization Practices (ACIP) of the Centers for Disease Control and Prevention (CDC) will face two key decisions once a COVID vaccine wins clearance from the US Food and Drug Administration (FDA).
ACIP will need to decide whether to recommend its use in adults (the age group in which vaccines are currently being tested). The group will also need to offer direction on which groups should get priority in vaccine allocation, inasmuch as early supplies will not be sufficient to vaccinate everyone.
At the Oct. 30 meeting, CDC’s Kathleen Dooling, MD, MPH, suggested that ACIP plan on tackling these issues as two separate questions when it comes time to weigh in on an approved vaccine. Although there was no formal vote among ACIP members at the meeting, Dooling’s proposal for tackling a future recommendation in a two-part fashion drew positive feedback.
ACIP member Katherine A. Poehling, MD, MPH, suggested that the panel and CDC be ready to reexamine the situation frequently regarding COVID vaccination. “Perhaps we could think about reviewing data on a monthly basis and updating the recommendation, so that we can account for the concerns and balance both the benefits and the [potential] harm,” Poehling said.
Dooling agreed. “Both the vaccine recommendation and allocation will be revisited in what is a very dynamic situation,” Dooling replied to Poehling. “So all new evidence will be brought to ACIP, and certainly the allocation as vaccine distribution proceeds will need to be adjusted accordingly.”
Ethics and limited evidence
During the meeting, ACIP members repeatedly expressed discomfort with the prospect of having to weigh in on widespread use of COVID vaccines on the basis of limited evidence.
Within months, FDA may opt for a special clearance, known as an emergency use authorization (EUA), for one or more of the experimental COVID vaccines now in advanced testing. Many of FDA’s past EUA clearances were granted for test kits. For those EUA approvals, the agency considered risks of false results but not longer-term, direct harm to patients from these products.
With a COVID vaccine, there will be strong pressure to distribute doses as quickly as possible with the hope of curbing the pandemic, which has already led to more than 229,000 deaths in the United States alone and has disrupted lives and economies around the world. But questions will persist about the possibility of serious complications from these vaccines, ACIP members noted.
“My personal struggle is the ethical side and how to balance these two,” said ACIP member Robert L. Atmar, MD, of Baylor College of Medicine, Houston, Texas, who noted that he expects his fellow panelists to share this concern.
Currently, four experimental COVID vaccines likely to be used in the United States have advanced to phase 3 testing. Pfizer Inc and BioNtech have enrolled more than 42,000 participants in a test of their candidate, BNT162b2 vaccine, and rival Moderna has enrolled about 30,000 participants in a test of its mRNA-1273 vaccine, CDC staff said.
The other two advanced COVID vaccine candidates have overcome recent hurdles. AstraZeneca Plc on Oct. 23 announced that FDA had removed a hold on the testing of its AZD1222 vaccine candidate; the trial will enroll approximately 30,000 people. Johnson & Johnson’s Janssen unit also announced that day the lifting of a safety pause for its Ad26.COV2.S vaccine; the phase 3 trial for that vaccine will enroll approximately 60,000 volunteers. Federal agencies, states, and territories have developed plans for future distribution of COVID vaccines, CDC staff said in briefing materials for today’s ACIP meeting.
Several ACIP members raised many of the same concerns that members of an FDA advisory committee raised at a meeting earlier in October. ACIP and FDA advisers honed in on the FDA’s decision to set a median follow-up duration of 2 months in phase 3 trials in connection with expected EUA applications for COVID-19 vaccines.
“I struggle with following people for 2 months after their second vaccination as a time point to start making final decisions about safety,” said ACIP member Sharon E. Frey, MD, a professor at St. Louis University School of Medicine, St. Louis, Missouri. “I just want to put that out there.”
Medical front line, then who?
There is consensus that healthcare workers be in the first stage ― Phase 1 ― of distribution. That recommendation was made in a report from the National Academies of Sciences, Engineering, and Medicine (NASEM). Phase 1A would include first responders; Phase 1B might include people of all ages who have two or more comorbidities that put them at significantly higher risk for COVID-19 or death, as well as older adults living in congregate or overcrowded settings, the NASEM report said.
A presentation from the CDC’s Matthew Biggerstaff, ScD, MPH, underscored challenges in distributing what are expected to be limited initial supplies of COVID vaccines.
Biggerstaff showed several scenarios the CDC’s Data, Analytics, and Modeling Task Force had studied. The initial allocation of vaccines would be for healthcare workers, followed by what the CDC called Phase 1B.
Choices for a rollout may include next giving COVID vaccines to people at high risk, such as persons who have one or more chronic medical conditions, including heart disease, diabetes, kidney disease, or obesity. Other options for the rollout could be to vaccinate people aged 65 years and older or essential workers whose employment puts them in contact with the public, thus raising the risk of contracting the virus.
The CDC’s research found that the greatest impact in preventing death was to initially vaccinate adults aged 65 and older in Phase 1B. The agency staff described this approach as likely to result in an about “1 to 11% increase in averted deaths across the scenarios.”
Initially vaccinating essential workers or high-risk adults in Phase 1B would avert the most infections. The agency staff described this approach as yielding about “1 to 5% increase in averted infections across the scenarios,” Biggerstaff said during his presentation.
The following are other findings of the CDC staff:
The earlier the vaccine rollout relative to increasing transmission, the greater the averted percentage and differences between the strategies.
Differences were not substantial in some scenarios.
The need to continue efforts to slow the spread of COVID-19 should be emphasized.
Adverse effects
ACIP members also heard about strategies for tracking potential side effects of future vaccines. A presentation by Tom Shimabukuro, MD, MPH, MBA, from the CDC’s COVID-19 Vaccine Task Force/Vaccine Safety Team, included details about a new smartphone-based active surveillance program for COVID-19 vaccine safety.
Known as v-safe, this system would use Web-based survey monitoring and incorporate text messaging. It would conduct electronic health checks on vaccine recipients, which would occur daily during the first week post vaccination and weekly thereafter for 6 weeks from the time of vaccination.
Clinicians “can play an important role in helping CDC enroll patients in v-safe at the time of vaccination,” Shimabukuro noted in his presentation. This would add another task, though, for clinicians, the CDC staff noted.
Pregnancy and breastfeeding are special concerns
Of special concern with the rollout of a COVID vaccine are recommendations regarding pregnancy and breastfeeding. Women constitute about 75% of the healthcare workforce, CDC staff noted.
At the time the initial ACIP COVID vaccination recommendations are made, there could be approximately 330,000 healthcare personnel who are pregnant or who have recently given birth. Available data indicate potentially increased risks for severe maternal illness and preterm birth associated with SARS-CoV-2 infection, said CDC’s Megan Wallace, DrPH, MPH, in a presentation for the Friday meeting.
In an Oct. 27 letter to ACIP, Chair Jose Romero, the American College of Obstetricians and Gynecologists (ACOG), urged the panel to ensure that pregnant women and new mothers in the healthcare workforce have priority access to a COVID vaccine. Pregnant and lactating women were “noticeably and alarmingly absent from the NASEM vaccine allocation plan for COVID-19,” wrote Christopher M. Zahn, MD, vice president for practice activities at ACOG, in the letter to Romero.
“ACOG urges ACIP to incorporate pregnant and lactating women clearly and explicitly into its COVID-19 vaccine allocation and prioritization framework,” Zahn wrote. “Should an Emergency Use Authorization be executed for one or more COVID-19 vaccines and provide a permissive recommendation for pregnant and lactating women, pregnant health care workers, pregnant first responders, and pregnant individuals with underlying conditions should be prioritized for vaccination alongside their non-pregnant peers.”
This article first appeared on Medscape.com.
Is the tide turning on the ‘grubby’ affair of EXCEL and the European guidelines?
“I disapprove of what you say, but I will defend to the death your right to say it.” The choice of the secretary general of the European Association for Cardio-Thoracic Surgery to open with this quote was the first hint that the next presentation at the 2019 annual meeting would be anything but dull. The session chair followed with a reminder to keep the discussion polite and civil.
Presenter David Taggart, MD, PhD, did not disappoint. The professor of cardiovascular surgery at the University of Oxford (England) began with the announcement that he had withdrawn his name from a recent paper in the New England Journal of Medicine. He then proceeded to accuse his coinvestigators of misrepresenting the findings of a major clinical trial.
Dr. Taggart was chair of the surgical committee for the Abbott-sponsored EXCEL trial, which compared two procedures for patients who had blockages in their left main coronary artery: percutaneous coronary intervention (PCI) using coronary stents, and coronary artery bypass graft surgery (CABG). The investigators designed the trial to compare outcomes for the two treatments using a composite endpoint of death, stroke, and MI. The 3-year follow-up data had been published in NEJM without controversy – or, at least, without public controversy.
But when it came time to publish the 5-year follow-up, there was a significantly higher rate of death in the stent group, and both Dr. Taggart and the journal editors were concerned that this finding was being downplayed in the manuscript.
In their comments to the authors, the journal editors had recommended including the mortality difference (unless clearly trivial) ‘”in the concluding statement in the final paragraph.” Yet, the concluding statement of the published paper read that there “was no significant difference between PCI and CABG.”
In Dr. Taggart’s view, that claim was dangerous for patients, and so he was left with no choice but to remove himself as an author, a first for the academic with over 300 scientific papers to his name.
Earlier publications from the EXCEL trial had influenced European treatment guidelines. But subsequent allegations of misconduct and hidden data spurred the EACTS to repudiate those guidelines out of concern “that some results in the EXCEL trial appear to have been concealed and that some patients may therefore have received the wrong clinical advice.”
The controversy pitted cardiothoracic surgeons against interventional cardiologists, who were seen as increasingly encroaching on the surgeons’ turf. Dr. Taggart was a long-time critic of the subspecialty.
Surgeons demanded an independent analysis of the EXCEL trial data – a demand that the investigators have yet to satisfy. Dr. Taggart was the first to speak publicly, but others had major reservations about the trial reporting and conduct years earlier.
Mortality data held back
One such person was Lars Wallentin, MD, a professor of cardiology at Uppsala (Sweden) University Hospital, who chaired the independent committee that monitored the safety and scientific validity of the EXCEL trial.
The committee, known as the data and safety monitoring board (DSMB), received a report on March 23, 2016, that showed that increasingly more patients who had received stents were dying, compared with the group of patients that had undergone CABG. A graph of the survival curves showed the gap between the two groups widening after 3 years (Figure 1).
By September of that year, Dr. Wallentin and other members of the DSMB were anxious to share the concerning mortality difference with the broader medical community.
They were aware that EACTS and the European Society of Cardiology had started the process of updating their guidelines on myocardial revascularization, and were keen for the guideline writing committee to see all of the data.
Meanwhile, the trial investigators, led by principal investigator Gregg Stone, MD, then at New York–Presbyterian Hospital and Columbia University Medical Center, were preparing to publish a report of the 3-year outcomes. Recruitment for EXCEL started in September 2010, so at the time of the 3-year analysis in 2016, some patients had been followed up for over 5 years. But the data, published in NEJM in October 2016, were capped at 3 years (Figure 2). It didn’t show the widening gap in late mortality that Dr. Wallentin and the rest of the DSMB had seen.
When asked about this, the investigators said they were transparent about their plans to cap the data at 3 years in an amendment to the study protocol. Stone’s coprincipal investigators were interventional cardiologist Patrick Serruys, MD, then of Imperial College London; and two surgeons: Joseph Sabik, MD, then of the Cleveland Clinic Foundation, and A. Pieter Kappetein, MD, PhD, then at Erasmus Medical Center, Rotterdam. The four principal investigators all declared financial payments from stent manufacturers either to themselves or their institutions.
Study sponsor Abbott has distanced itself from the decisions made and has referred all questions about the trial to the EXCEL investigators. Charles Simonton, chief medical officer at Abbott (now at Abiomed) was a coauthor on both the 3- and 5-year papers. Dr. Wallentin believes that the sponsor must have been aware of the DSMB’s concerns.
Continuing DSMB concerns
A year later, the DSMB was still troubled. Dr. Wallentin emailed Dr. Stone in September 2017 asking for an updated analysis of the mortality data without any capping in time.
Dr. Wallentin added that he didn’t think that unblinding the mortality results would be an issue at that stage because these were late deaths in a trial where the interventions were long completed. But, he warned, “it might be very concerning if, in the future, suspicions were raised that already available information on mortality was withheld from the cardiology and thoracic surgery community.”
The investigators took a month to respond. They declined the request, saying that the trial was not statistically powered to measure mortality. In his email to Dr. Wallentin, Dr. Stone stressed that they were committed to complete disclosure of all of the EXCEL data and that the responsible time point to unblind was after 4 years. His coprincipal investigators (Dr. Serruys, Dr. Sabik, and Dr. Kappetein) as well as EXCEL statistical committee chair Stuart Pocock, PhD, and Mr. Simonton were all copied on the email.
Dr. Wallentin deferred to the principal investigators’ arguments.
Missing MI data
Death was not the only outcome of the EXCEL trial to draw scrutiny.
The EXCEL investigators used a unique definition of MI that was almost exclusively based on a rise in the cardiac biomarker CK-MB. This protocol definition of MI was later adapted into the Society for Cardiovascular Angiography and Interventions definition in a paper coauthored by Dr. Stone. The investigators agreed to also measure MIs that met the more commonly used Third Universal Definition as a secondary endpoint. The Third Universal Definition of Myocardial Infarction uses a change in biomarkers – preferably troponin or alternatively CK-MB – coupled with other clinical signs.
It is standard practice to report secondary endpoints in any analysis of the main findings of a study. Yet, the EXCEL investigators did not report the universal definition of MI in either the 3-year or 5-year publications.
This is critical because MI according to one definition may not count according to the other, and the final tally could tip the trial results positive, negative, or neutral for coronary stents.
In Dr. Taggart’s opinion, the protocol definition puts CABG at a disadvantage because it uses the same biomarker threshold for procedural-related MI for both PCI and CABG. Because surgery involves more manipulation of the heart, cardiac enzyme levels will naturally be higher after CABG than PCI. These procedure-related enzyme elevations are not “true clinical MIs,” according to Dr. Taggart and others.
Late last year, a dataset containing the 3-year follow-up of EXCEL, including the information on the universal definition of MI, was leaked to the BBC. Working with biostatisticians, the BBC confirmed that according to this definition, there were more MIs in the stent group.
Originally, the investigators disputed the finding, calling the BBC data “imaginary.” They claimed that they were unable to calculate a rate of MI according to the universal definition because they lacked routine collection of troponins, although the universal definition also allows use of CK-MB. They have since published an analysis of 5-year MI data according to the universal definition, which showed twice the rate of MI in the PCI group.
From the leaked data, the BBC calculated the main composite endpoint of death, stroke, and MI using the universal definition of MI. Now the results swung in favor of CABG.
Impact on guidelines
None of this was known at the time the European cardiology societies convened a committee to write their new guidelines on myocardial revascularization. The writing panel disagreed about whether PCI and CABG were equivalent for patients with left main coronary artery disease (CAD).
Besides EXCEL, another study, the NOBLE trial, compared PCI and CABG in left main CAD and came to opposite conclusions – conclusions that matched the leaked data. In that trial, European investigators chose a slightly different primary endpoint: a composite of death, MI, stroke, and the need for a repeat procedure. They used the universal definition of MI exclusively, and notably, they omitted procedural MI from their clinical event count. The results, published at the same time as the EXCEL 3-year findings, suggested that CABG was better.
Given the discrepant findings of two large trials, the guideline committee considered all of the available data comparing the two methods of revascularization for left main CAD. But even then, things weren’t clear-cut. One draft meta-analysis, supported by the National Institute for Health Research, suggested that results were worse for first- and second-generation drug-eluting coronary stents – including those used in EXCEL – compared with surgery.
Another meta-analysis, later published in The Lancet, drew a different conclusion and found that PCI was just as good as surgery. The main author, Stuart Head, a cardiothoracic surgeon on the ESC/EACTS guideline committee, was a research fellow with EXCEL investigator Dr. Kappetein at Erasmus. EXCEL investigators Dr. Stone, Dr. Kappetein, and Dr. Serruys were coauthors of the Lancet meta-analysis.
There was heated discussion about the committee’s draft recommendations, which gave both CABG and PCI a Class IA recommendation in patients with left main CAD and low anatomical complexity. In October 2017, the ESC commissioned an anonymous external reviewer to weigh in. James Brophy, MD, PhD, a cardiologist and professor of medicine and epidemiology at McGill University, Montreal, confirmed that he was the reviewer after he published an updated version in June 2020.
Looking at all of the data available at the time comparing the procedures for left main CAD, Dr. Brophy’s analysis suggested a 73% chance that the excess in death, stroke, or MI represents at least two excess events per 100 patients treated with PCI rather than CABG.
Dr. Brophy thought that most patients would find these differences clinically meaningful and advised against giving both procedures the same class of recommendation. He was also concerned that many readers will skip to the summary recommendation table without reading the entire guideline document.
“I feel this is misleading in its present form,” he wrote in 2017.
Despite Dr. Brophy’s review, the guideline committee stuck with its original recommendations. The final 2018 ESC/EACTS Guidelines on myocardial revascularization gave equal weight to both CABG and PCI in patients with left main CAD and low anatomical complexity. In contrast, US guidelines do not put PCI and CABG on the same footing for any group of patients with left main CAD.
The lead author of the ESC/EACTS guidelines section on left main disease, and around a third of those on the writing task force, all declared financial payments from stent manufacturers either to themselves or their institutions. The EXCEL principal investigator, Dr. Kappetein, was secretary general of EACTS and oversaw the guidelines process for the surgical organization. He left to work for Medtronic midway through the process and was later joined there by his former research fellow, Stuart Head.
Dr. Brophy said in an interview that given the final guideline recommendations, he assumed that the committee had other reviews and went with the majority opinion.
But not everyone involved in the guidelines saw Dr. Brophy’s review. Nick Freemantle, a statistical reviewer appointed by EACTS, expected to see it but didn’t. This omission calls into question the neutrality of the whole process, in his view.
Mr. Freemantle believes that the deck was stacked so that he only saw the pieces of evidence that supported the conclusions that were already decided and that he was not shown “the bits that don’t fit that neatly.”
“And without that narrative, it all feels a bit grubby, to be honest,” he said.
Professor Barbara Casadei, ESC president, disputed this, saying that the guidelines were approved by all surgical members, including the EACTS council.
Missing from Dr. Brophy’s review were the later data from EXCEL. As he had told the DSMB in 2017, Stone presented the 4-year data from EXCEL at the TCT conference in September 2018. At this point, the analysis showed that 10.3% of people had died after PCI and 7.4% after CABG.
But this presentation was not given much prominence at the conference, which Dr. Stone organized, and occurred during a didactic session in a small room rather than on one of the main stages where the 3-year data from EXCEL were announced with much fanfare. The presentation also took place 3 weeks after the European guidelines were published.
Surgeons withdraw support
After the BBC report last year that the universal definition of MI data had been collected but not published in the 3-year follow-up manuscript, and showed more MI in the PCI group than the protocol definition, the EACTS withdrew its support for the guidelines. The ESC continued to uphold the guidelines «until there is robust scientific evidence (as opposed to allegations) indicating we should do otherwise,” said Ms. Casadei.
A spokesperson for NEJM said the journal stood by the EXCEL papers because “there is no credible harm to patients from the publication of the paper and accurate reporting of trial results.” NEJM has since conducted a review and published a series of letters in response. The letters have reinvigorated rather than appeased the dissenters, as reported by Medscape.
A number of cardiologists and researchers started a petition on change.org to revise the EACTS/ESC left main CAD guidelines, and surgical societies across the globe have written to the editor of NEJM asking him to retract or amend the EXCEL papers.
This has not happened. The journal’s editor maintains that the letters containing the analyses are “sufficient information” to allow readers and guideline authors to “evaluate the trial findings.”
Dr. Taggart was dismissive of that response. “There is still no recognition or acknowledgment that failure to publish these data in 2016 ‘misled’ the guideline writers for the ESC/EACTS guidelines, and there is still no formal correction of the 2016 and 2019 NEJM manuscripts.”
Over a year after the BBC received the leaked data, the EXCEL investigators published an analysis of the primary outcome using the universal definition of MI data in the Journal of the American College of Cardiology.
It shows 141 events in the PCI arm, compared with 102 in the CABG arm. The investigators acknowledge that the rates of procedural MI differ depending on the definition used. According to their analysis, the protocol definition was predictive of mortality after both treatments, whereas the universal definition of procedural MI was predictive of mortality only after CABG. Not everyone agrees with this interpretation, and an accompanying editorial questioned these conclusions.
For Dr. Wallentin, it’s a relief that these data are in the public domain so that their interpretation and clinical consequences can be “openly discussed.” He hoped that the whole experience will result in something constructive and useful for the future.
As for the guidelines, the tide may be turning.
In a joint statement with EACTS on Oct. 6, 2020, the ESC agreed to review its guidelines for left main disease in the light of emerging, longer-term outcome data from the trials of CABG versus PCI.
Dr. Taggart has no regrets about speaking out despite this being “an exceedingly painful and bruising experience.”
The saga, he said, “reflects very badly on our specialty, the investigators, industry, and the world’s ‘leading’ medical journal.”
This article first appeared on Medscape.com.
“I disapprove of what you say, but I will defend to the death your right to say it.” The choice of the secretary general of the European Association for Cardio-Thoracic Surgery to open with this quote was the first hint that the next presentation at the 2019 annual meeting would be anything but dull. The session chair followed with a reminder to keep the discussion polite and civil.
Presenter David Taggart, MD, PhD, did not disappoint. The professor of cardiovascular surgery at the University of Oxford (England) began with the announcement that he had withdrawn his name from a recent paper in the New England Journal of Medicine. He then proceeded to accuse his coinvestigators of misrepresenting the findings of a major clinical trial.
Dr. Taggart was chair of the surgical committee for the Abbott-sponsored EXCEL trial, which compared two procedures for patients who had blockages in their left main coronary artery: percutaneous coronary intervention (PCI) using coronary stents, and coronary artery bypass graft surgery (CABG). The investigators designed the trial to compare outcomes for the two treatments using a composite endpoint of death, stroke, and MI. The 3-year follow-up data had been published in NEJM without controversy – or, at least, without public controversy.
But when it came time to publish the 5-year follow-up, there was a significantly higher rate of death in the stent group, and both Dr. Taggart and the journal editors were concerned that this finding was being downplayed in the manuscript.
In their comments to the authors, the journal editors had recommended including the mortality difference (unless clearly trivial) ‘”in the concluding statement in the final paragraph.” Yet, the concluding statement of the published paper read that there “was no significant difference between PCI and CABG.”
In Dr. Taggart’s view, that claim was dangerous for patients, and so he was left with no choice but to remove himself as an author, a first for the academic with over 300 scientific papers to his name.
Earlier publications from the EXCEL trial had influenced European treatment guidelines. But subsequent allegations of misconduct and hidden data spurred the EACTS to repudiate those guidelines out of concern “that some results in the EXCEL trial appear to have been concealed and that some patients may therefore have received the wrong clinical advice.”
The controversy pitted cardiothoracic surgeons against interventional cardiologists, who were seen as increasingly encroaching on the surgeons’ turf. Dr. Taggart was a long-time critic of the subspecialty.
Surgeons demanded an independent analysis of the EXCEL trial data – a demand that the investigators have yet to satisfy. Dr. Taggart was the first to speak publicly, but others had major reservations about the trial reporting and conduct years earlier.
Mortality data held back
One such person was Lars Wallentin, MD, a professor of cardiology at Uppsala (Sweden) University Hospital, who chaired the independent committee that monitored the safety and scientific validity of the EXCEL trial.
The committee, known as the data and safety monitoring board (DSMB), received a report on March 23, 2016, that showed that increasingly more patients who had received stents were dying, compared with the group of patients that had undergone CABG. A graph of the survival curves showed the gap between the two groups widening after 3 years (Figure 1).
By September of that year, Dr. Wallentin and other members of the DSMB were anxious to share the concerning mortality difference with the broader medical community.
They were aware that EACTS and the European Society of Cardiology had started the process of updating their guidelines on myocardial revascularization, and were keen for the guideline writing committee to see all of the data.
Meanwhile, the trial investigators, led by principal investigator Gregg Stone, MD, then at New York–Presbyterian Hospital and Columbia University Medical Center, were preparing to publish a report of the 3-year outcomes. Recruitment for EXCEL started in September 2010, so at the time of the 3-year analysis in 2016, some patients had been followed up for over 5 years. But the data, published in NEJM in October 2016, were capped at 3 years (Figure 2). It didn’t show the widening gap in late mortality that Dr. Wallentin and the rest of the DSMB had seen.
When asked about this, the investigators said they were transparent about their plans to cap the data at 3 years in an amendment to the study protocol. Stone’s coprincipal investigators were interventional cardiologist Patrick Serruys, MD, then of Imperial College London; and two surgeons: Joseph Sabik, MD, then of the Cleveland Clinic Foundation, and A. Pieter Kappetein, MD, PhD, then at Erasmus Medical Center, Rotterdam. The four principal investigators all declared financial payments from stent manufacturers either to themselves or their institutions.
Study sponsor Abbott has distanced itself from the decisions made and has referred all questions about the trial to the EXCEL investigators. Charles Simonton, chief medical officer at Abbott (now at Abiomed) was a coauthor on both the 3- and 5-year papers. Dr. Wallentin believes that the sponsor must have been aware of the DSMB’s concerns.
Continuing DSMB concerns
A year later, the DSMB was still troubled. Dr. Wallentin emailed Dr. Stone in September 2017 asking for an updated analysis of the mortality data without any capping in time.
Dr. Wallentin added that he didn’t think that unblinding the mortality results would be an issue at that stage because these were late deaths in a trial where the interventions were long completed. But, he warned, “it might be very concerning if, in the future, suspicions were raised that already available information on mortality was withheld from the cardiology and thoracic surgery community.”
The investigators took a month to respond. They declined the request, saying that the trial was not statistically powered to measure mortality. In his email to Dr. Wallentin, Dr. Stone stressed that they were committed to complete disclosure of all of the EXCEL data and that the responsible time point to unblind was after 4 years. His coprincipal investigators (Dr. Serruys, Dr. Sabik, and Dr. Kappetein) as well as EXCEL statistical committee chair Stuart Pocock, PhD, and Mr. Simonton were all copied on the email.
Dr. Wallentin deferred to the principal investigators’ arguments.
Missing MI data
Death was not the only outcome of the EXCEL trial to draw scrutiny.
The EXCEL investigators used a unique definition of MI that was almost exclusively based on a rise in the cardiac biomarker CK-MB. This protocol definition of MI was later adapted into the Society for Cardiovascular Angiography and Interventions definition in a paper coauthored by Dr. Stone. The investigators agreed to also measure MIs that met the more commonly used Third Universal Definition as a secondary endpoint. The Third Universal Definition of Myocardial Infarction uses a change in biomarkers – preferably troponin or alternatively CK-MB – coupled with other clinical signs.
It is standard practice to report secondary endpoints in any analysis of the main findings of a study. Yet, the EXCEL investigators did not report the universal definition of MI in either the 3-year or 5-year publications.
This is critical because MI according to one definition may not count according to the other, and the final tally could tip the trial results positive, negative, or neutral for coronary stents.
In Dr. Taggart’s opinion, the protocol definition puts CABG at a disadvantage because it uses the same biomarker threshold for procedural-related MI for both PCI and CABG. Because surgery involves more manipulation of the heart, cardiac enzyme levels will naturally be higher after CABG than PCI. These procedure-related enzyme elevations are not “true clinical MIs,” according to Dr. Taggart and others.
Late last year, a dataset containing the 3-year follow-up of EXCEL, including the information on the universal definition of MI, was leaked to the BBC. Working with biostatisticians, the BBC confirmed that according to this definition, there were more MIs in the stent group.
Originally, the investigators disputed the finding, calling the BBC data “imaginary.” They claimed that they were unable to calculate a rate of MI according to the universal definition because they lacked routine collection of troponins, although the universal definition also allows use of CK-MB. They have since published an analysis of 5-year MI data according to the universal definition, which showed twice the rate of MI in the PCI group.
From the leaked data, the BBC calculated the main composite endpoint of death, stroke, and MI using the universal definition of MI. Now the results swung in favor of CABG.
Impact on guidelines
None of this was known at the time the European cardiology societies convened a committee to write their new guidelines on myocardial revascularization. The writing panel disagreed about whether PCI and CABG were equivalent for patients with left main coronary artery disease (CAD).
Besides EXCEL, another study, the NOBLE trial, compared PCI and CABG in left main CAD and came to opposite conclusions – conclusions that matched the leaked data. In that trial, European investigators chose a slightly different primary endpoint: a composite of death, MI, stroke, and the need for a repeat procedure. They used the universal definition of MI exclusively, and notably, they omitted procedural MI from their clinical event count. The results, published at the same time as the EXCEL 3-year findings, suggested that CABG was better.
Given the discrepant findings of two large trials, the guideline committee considered all of the available data comparing the two methods of revascularization for left main CAD. But even then, things weren’t clear-cut. One draft meta-analysis, supported by the National Institute for Health Research, suggested that results were worse for first- and second-generation drug-eluting coronary stents – including those used in EXCEL – compared with surgery.
Another meta-analysis, later published in The Lancet, drew a different conclusion and found that PCI was just as good as surgery. The main author, Stuart Head, a cardiothoracic surgeon on the ESC/EACTS guideline committee, was a research fellow with EXCEL investigator Dr. Kappetein at Erasmus. EXCEL investigators Dr. Stone, Dr. Kappetein, and Dr. Serruys were coauthors of the Lancet meta-analysis.
There was heated discussion about the committee’s draft recommendations, which gave both CABG and PCI a Class IA recommendation in patients with left main CAD and low anatomical complexity. In October 2017, the ESC commissioned an anonymous external reviewer to weigh in. James Brophy, MD, PhD, a cardiologist and professor of medicine and epidemiology at McGill University, Montreal, confirmed that he was the reviewer after he published an updated version in June 2020.
Looking at all of the data available at the time comparing the procedures for left main CAD, Dr. Brophy’s analysis suggested a 73% chance that the excess in death, stroke, or MI represents at least two excess events per 100 patients treated with PCI rather than CABG.
Dr. Brophy thought that most patients would find these differences clinically meaningful and advised against giving both procedures the same class of recommendation. He was also concerned that many readers will skip to the summary recommendation table without reading the entire guideline document.
“I feel this is misleading in its present form,” he wrote in 2017.
Despite Dr. Brophy’s review, the guideline committee stuck with its original recommendations. The final 2018 ESC/EACTS Guidelines on myocardial revascularization gave equal weight to both CABG and PCI in patients with left main CAD and low anatomical complexity. In contrast, US guidelines do not put PCI and CABG on the same footing for any group of patients with left main CAD.
The lead author of the ESC/EACTS guidelines section on left main disease, and around a third of those on the writing task force, all declared financial payments from stent manufacturers either to themselves or their institutions. The EXCEL principal investigator, Dr. Kappetein, was secretary general of EACTS and oversaw the guidelines process for the surgical organization. He left to work for Medtronic midway through the process and was later joined there by his former research fellow, Stuart Head.
Dr. Brophy said in an interview that given the final guideline recommendations, he assumed that the committee had other reviews and went with the majority opinion.
But not everyone involved in the guidelines saw Dr. Brophy’s review. Nick Freemantle, a statistical reviewer appointed by EACTS, expected to see it but didn’t. This omission calls into question the neutrality of the whole process, in his view.
Mr. Freemantle believes that the deck was stacked so that he only saw the pieces of evidence that supported the conclusions that were already decided and that he was not shown “the bits that don’t fit that neatly.”
“And without that narrative, it all feels a bit grubby, to be honest,” he said.
Professor Barbara Casadei, ESC president, disputed this, saying that the guidelines were approved by all surgical members, including the EACTS council.
Missing from Dr. Brophy’s review were the later data from EXCEL. As he had told the DSMB in 2017, Stone presented the 4-year data from EXCEL at the TCT conference in September 2018. At this point, the analysis showed that 10.3% of people had died after PCI and 7.4% after CABG.
But this presentation was not given much prominence at the conference, which Dr. Stone organized, and occurred during a didactic session in a small room rather than on one of the main stages where the 3-year data from EXCEL were announced with much fanfare. The presentation also took place 3 weeks after the European guidelines were published.
Surgeons withdraw support
After the BBC report last year that the universal definition of MI data had been collected but not published in the 3-year follow-up manuscript, and showed more MI in the PCI group than the protocol definition, the EACTS withdrew its support for the guidelines. The ESC continued to uphold the guidelines «until there is robust scientific evidence (as opposed to allegations) indicating we should do otherwise,” said Ms. Casadei.
A spokesperson for NEJM said the journal stood by the EXCEL papers because “there is no credible harm to patients from the publication of the paper and accurate reporting of trial results.” NEJM has since conducted a review and published a series of letters in response. The letters have reinvigorated rather than appeased the dissenters, as reported by Medscape.
A number of cardiologists and researchers started a petition on change.org to revise the EACTS/ESC left main CAD guidelines, and surgical societies across the globe have written to the editor of NEJM asking him to retract or amend the EXCEL papers.
This has not happened. The journal’s editor maintains that the letters containing the analyses are “sufficient information” to allow readers and guideline authors to “evaluate the trial findings.”
Dr. Taggart was dismissive of that response. “There is still no recognition or acknowledgment that failure to publish these data in 2016 ‘misled’ the guideline writers for the ESC/EACTS guidelines, and there is still no formal correction of the 2016 and 2019 NEJM manuscripts.”
Over a year after the BBC received the leaked data, the EXCEL investigators published an analysis of the primary outcome using the universal definition of MI data in the Journal of the American College of Cardiology.
It shows 141 events in the PCI arm, compared with 102 in the CABG arm. The investigators acknowledge that the rates of procedural MI differ depending on the definition used. According to their analysis, the protocol definition was predictive of mortality after both treatments, whereas the universal definition of procedural MI was predictive of mortality only after CABG. Not everyone agrees with this interpretation, and an accompanying editorial questioned these conclusions.
For Dr. Wallentin, it’s a relief that these data are in the public domain so that their interpretation and clinical consequences can be “openly discussed.” He hoped that the whole experience will result in something constructive and useful for the future.
As for the guidelines, the tide may be turning.
In a joint statement with EACTS on Oct. 6, 2020, the ESC agreed to review its guidelines for left main disease in the light of emerging, longer-term outcome data from the trials of CABG versus PCI.
Dr. Taggart has no regrets about speaking out despite this being “an exceedingly painful and bruising experience.”
The saga, he said, “reflects very badly on our specialty, the investigators, industry, and the world’s ‘leading’ medical journal.”
This article first appeared on Medscape.com.
“I disapprove of what you say, but I will defend to the death your right to say it.” The choice of the secretary general of the European Association for Cardio-Thoracic Surgery to open with this quote was the first hint that the next presentation at the 2019 annual meeting would be anything but dull. The session chair followed with a reminder to keep the discussion polite and civil.
Presenter David Taggart, MD, PhD, did not disappoint. The professor of cardiovascular surgery at the University of Oxford (England) began with the announcement that he had withdrawn his name from a recent paper in the New England Journal of Medicine. He then proceeded to accuse his coinvestigators of misrepresenting the findings of a major clinical trial.
Dr. Taggart was chair of the surgical committee for the Abbott-sponsored EXCEL trial, which compared two procedures for patients who had blockages in their left main coronary artery: percutaneous coronary intervention (PCI) using coronary stents, and coronary artery bypass graft surgery (CABG). The investigators designed the trial to compare outcomes for the two treatments using a composite endpoint of death, stroke, and MI. The 3-year follow-up data had been published in NEJM without controversy – or, at least, without public controversy.
But when it came time to publish the 5-year follow-up, there was a significantly higher rate of death in the stent group, and both Dr. Taggart and the journal editors were concerned that this finding was being downplayed in the manuscript.
In their comments to the authors, the journal editors had recommended including the mortality difference (unless clearly trivial) ‘”in the concluding statement in the final paragraph.” Yet, the concluding statement of the published paper read that there “was no significant difference between PCI and CABG.”
In Dr. Taggart’s view, that claim was dangerous for patients, and so he was left with no choice but to remove himself as an author, a first for the academic with over 300 scientific papers to his name.
Earlier publications from the EXCEL trial had influenced European treatment guidelines. But subsequent allegations of misconduct and hidden data spurred the EACTS to repudiate those guidelines out of concern “that some results in the EXCEL trial appear to have been concealed and that some patients may therefore have received the wrong clinical advice.”
The controversy pitted cardiothoracic surgeons against interventional cardiologists, who were seen as increasingly encroaching on the surgeons’ turf. Dr. Taggart was a long-time critic of the subspecialty.
Surgeons demanded an independent analysis of the EXCEL trial data – a demand that the investigators have yet to satisfy. Dr. Taggart was the first to speak publicly, but others had major reservations about the trial reporting and conduct years earlier.
Mortality data held back
One such person was Lars Wallentin, MD, a professor of cardiology at Uppsala (Sweden) University Hospital, who chaired the independent committee that monitored the safety and scientific validity of the EXCEL trial.
The committee, known as the data and safety monitoring board (DSMB), received a report on March 23, 2016, that showed that increasingly more patients who had received stents were dying, compared with the group of patients that had undergone CABG. A graph of the survival curves showed the gap between the two groups widening after 3 years (Figure 1).
By September of that year, Dr. Wallentin and other members of the DSMB were anxious to share the concerning mortality difference with the broader medical community.
They were aware that EACTS and the European Society of Cardiology had started the process of updating their guidelines on myocardial revascularization, and were keen for the guideline writing committee to see all of the data.
Meanwhile, the trial investigators, led by principal investigator Gregg Stone, MD, then at New York–Presbyterian Hospital and Columbia University Medical Center, were preparing to publish a report of the 3-year outcomes. Recruitment for EXCEL started in September 2010, so at the time of the 3-year analysis in 2016, some patients had been followed up for over 5 years. But the data, published in NEJM in October 2016, were capped at 3 years (Figure 2). It didn’t show the widening gap in late mortality that Dr. Wallentin and the rest of the DSMB had seen.
When asked about this, the investigators said they were transparent about their plans to cap the data at 3 years in an amendment to the study protocol. Stone’s coprincipal investigators were interventional cardiologist Patrick Serruys, MD, then of Imperial College London; and two surgeons: Joseph Sabik, MD, then of the Cleveland Clinic Foundation, and A. Pieter Kappetein, MD, PhD, then at Erasmus Medical Center, Rotterdam. The four principal investigators all declared financial payments from stent manufacturers either to themselves or their institutions.
Study sponsor Abbott has distanced itself from the decisions made and has referred all questions about the trial to the EXCEL investigators. Charles Simonton, chief medical officer at Abbott (now at Abiomed) was a coauthor on both the 3- and 5-year papers. Dr. Wallentin believes that the sponsor must have been aware of the DSMB’s concerns.
Continuing DSMB concerns
A year later, the DSMB was still troubled. Dr. Wallentin emailed Dr. Stone in September 2017 asking for an updated analysis of the mortality data without any capping in time.
Dr. Wallentin added that he didn’t think that unblinding the mortality results would be an issue at that stage because these were late deaths in a trial where the interventions were long completed. But, he warned, “it might be very concerning if, in the future, suspicions were raised that already available information on mortality was withheld from the cardiology and thoracic surgery community.”
The investigators took a month to respond. They declined the request, saying that the trial was not statistically powered to measure mortality. In his email to Dr. Wallentin, Dr. Stone stressed that they were committed to complete disclosure of all of the EXCEL data and that the responsible time point to unblind was after 4 years. His coprincipal investigators (Dr. Serruys, Dr. Sabik, and Dr. Kappetein) as well as EXCEL statistical committee chair Stuart Pocock, PhD, and Mr. Simonton were all copied on the email.
Dr. Wallentin deferred to the principal investigators’ arguments.
Missing MI data
Death was not the only outcome of the EXCEL trial to draw scrutiny.
The EXCEL investigators used a unique definition of MI that was almost exclusively based on a rise in the cardiac biomarker CK-MB. This protocol definition of MI was later adapted into the Society for Cardiovascular Angiography and Interventions definition in a paper coauthored by Dr. Stone. The investigators agreed to also measure MIs that met the more commonly used Third Universal Definition as a secondary endpoint. The Third Universal Definition of Myocardial Infarction uses a change in biomarkers – preferably troponin or alternatively CK-MB – coupled with other clinical signs.
It is standard practice to report secondary endpoints in any analysis of the main findings of a study. Yet, the EXCEL investigators did not report the universal definition of MI in either the 3-year or 5-year publications.
This is critical because MI according to one definition may not count according to the other, and the final tally could tip the trial results positive, negative, or neutral for coronary stents.
In Dr. Taggart’s opinion, the protocol definition puts CABG at a disadvantage because it uses the same biomarker threshold for procedural-related MI for both PCI and CABG. Because surgery involves more manipulation of the heart, cardiac enzyme levels will naturally be higher after CABG than PCI. These procedure-related enzyme elevations are not “true clinical MIs,” according to Dr. Taggart and others.
Late last year, a dataset containing the 3-year follow-up of EXCEL, including the information on the universal definition of MI, was leaked to the BBC. Working with biostatisticians, the BBC confirmed that according to this definition, there were more MIs in the stent group.
Originally, the investigators disputed the finding, calling the BBC data “imaginary.” They claimed that they were unable to calculate a rate of MI according to the universal definition because they lacked routine collection of troponins, although the universal definition also allows use of CK-MB. They have since published an analysis of 5-year MI data according to the universal definition, which showed twice the rate of MI in the PCI group.
From the leaked data, the BBC calculated the main composite endpoint of death, stroke, and MI using the universal definition of MI. Now the results swung in favor of CABG.
Impact on guidelines
None of this was known at the time the European cardiology societies convened a committee to write their new guidelines on myocardial revascularization. The writing panel disagreed about whether PCI and CABG were equivalent for patients with left main coronary artery disease (CAD).
Besides EXCEL, another study, the NOBLE trial, compared PCI and CABG in left main CAD and came to opposite conclusions – conclusions that matched the leaked data. In that trial, European investigators chose a slightly different primary endpoint: a composite of death, MI, stroke, and the need for a repeat procedure. They used the universal definition of MI exclusively, and notably, they omitted procedural MI from their clinical event count. The results, published at the same time as the EXCEL 3-year findings, suggested that CABG was better.
Given the discrepant findings of two large trials, the guideline committee considered all of the available data comparing the two methods of revascularization for left main CAD. But even then, things weren’t clear-cut. One draft meta-analysis, supported by the National Institute for Health Research, suggested that results were worse for first- and second-generation drug-eluting coronary stents – including those used in EXCEL – compared with surgery.
Another meta-analysis, later published in The Lancet, drew a different conclusion and found that PCI was just as good as surgery. The main author, Stuart Head, a cardiothoracic surgeon on the ESC/EACTS guideline committee, was a research fellow with EXCEL investigator Dr. Kappetein at Erasmus. EXCEL investigators Dr. Stone, Dr. Kappetein, and Dr. Serruys were coauthors of the Lancet meta-analysis.
There was heated discussion about the committee’s draft recommendations, which gave both CABG and PCI a Class IA recommendation in patients with left main CAD and low anatomical complexity. In October 2017, the ESC commissioned an anonymous external reviewer to weigh in. James Brophy, MD, PhD, a cardiologist and professor of medicine and epidemiology at McGill University, Montreal, confirmed that he was the reviewer after he published an updated version in June 2020.
Looking at all of the data available at the time comparing the procedures for left main CAD, Dr. Brophy’s analysis suggested a 73% chance that the excess in death, stroke, or MI represents at least two excess events per 100 patients treated with PCI rather than CABG.
Dr. Brophy thought that most patients would find these differences clinically meaningful and advised against giving both procedures the same class of recommendation. He was also concerned that many readers will skip to the summary recommendation table without reading the entire guideline document.
“I feel this is misleading in its present form,” he wrote in 2017.
Despite Dr. Brophy’s review, the guideline committee stuck with its original recommendations. The final 2018 ESC/EACTS Guidelines on myocardial revascularization gave equal weight to both CABG and PCI in patients with left main CAD and low anatomical complexity. In contrast, US guidelines do not put PCI and CABG on the same footing for any group of patients with left main CAD.
The lead author of the ESC/EACTS guidelines section on left main disease, and around a third of those on the writing task force, all declared financial payments from stent manufacturers either to themselves or their institutions. The EXCEL principal investigator, Dr. Kappetein, was secretary general of EACTS and oversaw the guidelines process for the surgical organization. He left to work for Medtronic midway through the process and was later joined there by his former research fellow, Stuart Head.
Dr. Brophy said in an interview that given the final guideline recommendations, he assumed that the committee had other reviews and went with the majority opinion.
But not everyone involved in the guidelines saw Dr. Brophy’s review. Nick Freemantle, a statistical reviewer appointed by EACTS, expected to see it but didn’t. This omission calls into question the neutrality of the whole process, in his view.
Mr. Freemantle believes that the deck was stacked so that he only saw the pieces of evidence that supported the conclusions that were already decided and that he was not shown “the bits that don’t fit that neatly.”
“And without that narrative, it all feels a bit grubby, to be honest,” he said.
Professor Barbara Casadei, ESC president, disputed this, saying that the guidelines were approved by all surgical members, including the EACTS council.
Missing from Dr. Brophy’s review were the later data from EXCEL. As he had told the DSMB in 2017, Stone presented the 4-year data from EXCEL at the TCT conference in September 2018. At this point, the analysis showed that 10.3% of people had died after PCI and 7.4% after CABG.
But this presentation was not given much prominence at the conference, which Dr. Stone organized, and occurred during a didactic session in a small room rather than on one of the main stages where the 3-year data from EXCEL were announced with much fanfare. The presentation also took place 3 weeks after the European guidelines were published.
Surgeons withdraw support
After the BBC report last year that the universal definition of MI data had been collected but not published in the 3-year follow-up manuscript, and showed more MI in the PCI group than the protocol definition, the EACTS withdrew its support for the guidelines. The ESC continued to uphold the guidelines «until there is robust scientific evidence (as opposed to allegations) indicating we should do otherwise,” said Ms. Casadei.
A spokesperson for NEJM said the journal stood by the EXCEL papers because “there is no credible harm to patients from the publication of the paper and accurate reporting of trial results.” NEJM has since conducted a review and published a series of letters in response. The letters have reinvigorated rather than appeased the dissenters, as reported by Medscape.
A number of cardiologists and researchers started a petition on change.org to revise the EACTS/ESC left main CAD guidelines, and surgical societies across the globe have written to the editor of NEJM asking him to retract or amend the EXCEL papers.
This has not happened. The journal’s editor maintains that the letters containing the analyses are “sufficient information” to allow readers and guideline authors to “evaluate the trial findings.”
Dr. Taggart was dismissive of that response. “There is still no recognition or acknowledgment that failure to publish these data in 2016 ‘misled’ the guideline writers for the ESC/EACTS guidelines, and there is still no formal correction of the 2016 and 2019 NEJM manuscripts.”
Over a year after the BBC received the leaked data, the EXCEL investigators published an analysis of the primary outcome using the universal definition of MI data in the Journal of the American College of Cardiology.
It shows 141 events in the PCI arm, compared with 102 in the CABG arm. The investigators acknowledge that the rates of procedural MI differ depending on the definition used. According to their analysis, the protocol definition was predictive of mortality after both treatments, whereas the universal definition of procedural MI was predictive of mortality only after CABG. Not everyone agrees with this interpretation, and an accompanying editorial questioned these conclusions.
For Dr. Wallentin, it’s a relief that these data are in the public domain so that their interpretation and clinical consequences can be “openly discussed.” He hoped that the whole experience will result in something constructive and useful for the future.
As for the guidelines, the tide may be turning.
In a joint statement with EACTS on Oct. 6, 2020, the ESC agreed to review its guidelines for left main disease in the light of emerging, longer-term outcome data from the trials of CABG versus PCI.
Dr. Taggart has no regrets about speaking out despite this being “an exceedingly painful and bruising experience.”
The saga, he said, “reflects very badly on our specialty, the investigators, industry, and the world’s ‘leading’ medical journal.”
This article first appeared on Medscape.com.