User login
Welcome to Current Psychiatry, a leading source of information, online and in print, for practitioners of psychiatry and its related subspecialties, including addiction psychiatry, child and adolescent psychiatry, and geriatric psychiatry. This Web site contains evidence-based reviews of the prevention, diagnosis, and treatment of mental illness and psychological disorders; case reports; updates on psychopharmacology; news about the specialty of psychiatry; pearls for practice; and other topics of interest and use to this audience.
Dear Drupal User: You're seeing this because you're logged in to Drupal, and not redirected to MDedge.com/psychiatry.
Depression
adolescent depression
adolescent major depressive disorder
adolescent schizophrenia
adolescent with major depressive disorder
animals
autism
baby
brexpiprazole
child
child bipolar
child depression
child schizophrenia
children with bipolar disorder
children with depression
children with major depressive disorder
compulsive behaviors
cure
elderly bipolar
elderly depression
elderly major depressive disorder
elderly schizophrenia
elderly with dementia
first break
first episode
gambling
gaming
geriatric depression
geriatric major depressive disorder
geriatric schizophrenia
infant
kid
major depressive disorder
major depressive disorder in adolescents
major depressive disorder in children
parenting
pediatric
pediatric bipolar
pediatric depression
pediatric major depressive disorder
pediatric schizophrenia
pregnancy
pregnant
rexulti
skin care
teen
wine
section[contains(@class, 'nav-hidden')]
footer[@id='footer']
div[contains(@class, 'pane-pub-article-current-psychiatry')]
div[contains(@class, 'pane-pub-home-current-psychiatry')]
div[contains(@class, 'pane-pub-topic-current-psychiatry')]
div[contains(@class, 'panel-panel-inner')]
div[contains(@class, 'pane-node-field-article-topics')]
section[contains(@class, 'footer-nav-section-wrapper')]
Drug-induced progressive multifocal leukoencephalopathy: Rare but serious
Mr. P, age 67, presents to the clinic with vision changes and memory loss following a fall in his home due to limb weakness. Six years ago, his care team diagnosed him with rheumatoid arthritis (RA). Mr. P’s current medication regimen includes methotrexate 20 mg once weekly and etanercept 50 mg once weekly, and he has been stable on this plan for 3 years. Mr. P also was recently diagnosed with major depressive disorder (MDD), but has not yet started treatment. Following a complete workup, an MRI of Mr. P’s brain revealed white matter demyelination. Due to these findings, he is scheduled for a brain biopsy, which confirms a diagnosis of progressive multifocal leukoencephalopathy (PML).
PML is a demyelinating disease of the central nervous system caused by the John Cunningham virus (JCV), or JC polyomavirus, named for the first patient identified to have contracted the virus.1 Asymptomatic infection of JCV often occurs in childhood, and antibodies are found in ≤70% of healthy adults. In most individuals, JCV remains latent in the kidneys and lymphoid organs, but immunosuppression can cause it to reactivate.2
JCV infects oligodendrocytes, astrocytes, and neurons, which results in white matter demyelination. Due to this demyelination, individuals can experience visual field defects, speech disturbances, ataxia, paresthesia, and cognitive impairments.2 Limb weakness presents in 60% of patients with PML, visual disturbances in 20%, and gait disturbances in 65%.3 Progression of these symptoms can lead to a more severe clinical presentation, including focal seizures in ≤10% of patients, and the mortality rate is 30% to 50%.3 Patients with comorbid HIV have a mortality rate ≤90%.2
Currently, there are no biomarkers that can identify PML in its early stages. A PML diagnosis is typically based on the patient’s clinical presentation, radiological imaging, and detection of JCV DNA. A brain biopsy is the gold standard for PML diagnosis.1
Interestingly, data suggest that glial cells harboring JCV in the brain express receptors for serotonin and dopamine.4 Researchers pinpointed 5HT2A receptors as JCV entry points into cells, and theorized that medications competing for binding, such as certain psychotropic agents, might decrease JCV entry. Cells lacking the 5HT2A receptor have shown immunity to JCV infection and the ability of cells to be infected was restored through transfection of 5HT2A receptors.4
Immunosuppressant medications can cause PML
PML was initially seen in individuals with conditions that cause immunosuppression, such as malignancies and HIV. However, “drug-induced PML” refers to cases in which drug-induced immunosuppression creates an environment that allows JCV to reactivate and disseminate back into the CNS.4 It is important to emphasize that drug-induced PML is a very rare effect of certain immunosuppressant medications. Medications that can weaken the immune system include glucocorticoids, monoclonal antibodies, alkylating agents, purine analogues, antimetabolites, and immunosuppressants (Table).1

These medications are used to treat conditions such as multiple sclerosis, RA, psoriatic arthritis, and lupus. Although drug-induced PML can result from the use of any of these agents, the highest incidence (1%) is found with natalizumab. Rates of incidence with other agents are either unknown or as low as .002%.1 Evidence suggests that the risk for PML increases with the duration of therapy.5
Continue to: Management
Management: Stop the offending agent, restore immune function
Specific pharmacologic treatments for PML are lacking. Management of drug-induced PML starts with discontinuing the offending agent. Restoring immune function has been found to be the most effective approach to treat PML.3 Restoration is possible through interleukin-2 (IL-2), IL-7, and T-cell infusions. Other treatment options are theoretical and include the development of a JCV vaccine to stimulate host response, plasma exchange to remove the medication from the host, and antiviral therapy targeting JCV replication. Diclofenac, isotretinoin, and mefloquine can inhibit JCV replication.3
Based on the theory that JCV requires 5HT2A receptors for entry into cells, researchers have studied medications that block this receptor as a treatment for PML. The first-generation antipsychotic chlorpromazine did not show benefit when combined with cidofovir, a replication inhibitor.3 Antipsychotics agents such as ziprasidone and olanzapine have shown in vitro inhibition of JCV, while risperidone has mixed results, with 1 trial failing to find a difference on JCV in fetal glial cells.3 Second-generation antipsychotics may be the preferred option due to more potent antagonism of the 5HT2A receptors and fewer adverse effects compared to agents such as chlorpromazine.4 The antidepressant mirtazapine has shown to have promising results, with evidence indicating that earlier initiation is more beneficial.3 Overall, data involving the use of medications that act on the 5HT2A receptor are mixed. Recent data suggest that JCV might enter cells independent of 5HT2A receptors; however, more research in this area is needed.2
The best strategy for treating drug-induced PML has not yet been determined. While combination therapy is thought to be more successful than monotherapy, ultimately, it depends on the patient’s immune response. If a psychotropic medication is chosen as adjunct treatment for drug-induced PML, it is prudent to assess the patient’s entire clinical picture to determine the specific indication for therapy (ie, treating symptomatology or drug-induced PML).
CASE CONTINUED
Following diagnosis, Mr. P is provided supportive therapy, and his care team discontinues methotrexate and etanercept. Although data are mixed on the efficacy of medications that work on 5HT2A receptors, because Mr. P was recently diagnosed with MDD, he is started on mirtazapine 15 mg/d at night in an attempt to manage both MDD and PML. It is possible that his depressive symptoms developed as a result of drug-induced PML rather than major depressive disorder. Discontinuing methotrexate and etanercept stabilizes Mr. P’s PML symptoms but leads to an exacerbation of his RA symptoms. Mr. P is initiated on hyd
Related Resources
- Castle D, Robertson NP. Treatment of progressive multifocal leukoencephalopathy. J Neurol. 2019;266(10):2587-2589. doi:10.1007/s00415-019-09501-y
Drug Brand Names
Abatacept • Orencia
Adalimumab • Humira
Alemtuzumab • Campath
Azathioprine • Azasan, Imuran
Basiliximab • Simulect
Belimumab • Benlysta
Bevacizumab • Avastin
Brentuximab vedotin • Adcetris
Cetuximab • Erbitux
Chlorpromazine • Thorazine, Largactil
Cidofovir • Vistide
Cladribine • Mavenclad
Cyclophosphamide • Cytoxan
Cyclosporine • Gengraf, Neoral
Dacarbazine • DTIC-Dome
Diclofenac • Cambia, Zorvolex
Dimethyl fumarate • Tecfidera
Etanercept • Enbrel
Fingolimod • Gilenya
Fludarabine • Fludara
Hydroxychloroquine • Plaquenil
Ibritumomab tiuxetan • Zevalin
Infliximab • Avsola, Inflectra
Isotretinoin • Absorica, Claravis
Mefloquine • Lariam
Methotrexate • Rheumatrex, Trexall
Mirtazapine • Remeron
Mitoxantrone • Novantrone
Muromonab-CD3 • Orthoclone OKT3
Mycophenolate mofetil • CellCept
Natalizumab • Tysabri
Nelarabine • Arranon
Obinutuzumab • Gazyva
Olanzapine • Zyprexa
Risperidone • Risperdal
Tacrolimus • Prograf
Vincristine • Vincasar PFS
Ziprasidone • Geodon
1. Yukitake M. Drug-induced progressive multifocal leukoencephalopathy in multiple sclerosis: a comprehensive review. Clin Exp Neuroimmunol. 2018;9(1):37-47. doi:10.1111/cen3.12440
2. Alstadhaug KB, Myhr KM, Rinaldo CH. Progressive multifocal leukoencephalopathy. Tidsskr Nor Laegeforen. 2017;137(23-24):10.4045/tidsskr.16.1092. doi:10.4045/tidsskr.16.1092
3. Williamson EML, Berger JR. Diagnosis and treatment of progressive multifocal leukoencephalopathy associated with multiple sclerosis therapies. Neurotherapeutics. 2017;14(4):961-973. doi:10.1007/s13311-017-0570-7
4. Altschuler EL, Kast RE. The atypical antipsychotic agents ziprasidone, risperidone and olanzapine as treatment for and prophylaxis against progressive multifocal leukoencephalopathy. Med Hypotheses. 2005;65(3):585-586.
5. Vinhas de Souza M, Keller-Stanislawski B, Blake K, et al. Drug-induced PML: a global agenda for a global challenge. Clin Pharmacol Ther. 2012;91(4):747-750. doi:10.1038/clpt.2012.4
Mr. P, age 67, presents to the clinic with vision changes and memory loss following a fall in his home due to limb weakness. Six years ago, his care team diagnosed him with rheumatoid arthritis (RA). Mr. P’s current medication regimen includes methotrexate 20 mg once weekly and etanercept 50 mg once weekly, and he has been stable on this plan for 3 years. Mr. P also was recently diagnosed with major depressive disorder (MDD), but has not yet started treatment. Following a complete workup, an MRI of Mr. P’s brain revealed white matter demyelination. Due to these findings, he is scheduled for a brain biopsy, which confirms a diagnosis of progressive multifocal leukoencephalopathy (PML).
PML is a demyelinating disease of the central nervous system caused by the John Cunningham virus (JCV), or JC polyomavirus, named for the first patient identified to have contracted the virus.1 Asymptomatic infection of JCV often occurs in childhood, and antibodies are found in ≤70% of healthy adults. In most individuals, JCV remains latent in the kidneys and lymphoid organs, but immunosuppression can cause it to reactivate.2
JCV infects oligodendrocytes, astrocytes, and neurons, which results in white matter demyelination. Due to this demyelination, individuals can experience visual field defects, speech disturbances, ataxia, paresthesia, and cognitive impairments.2 Limb weakness presents in 60% of patients with PML, visual disturbances in 20%, and gait disturbances in 65%.3 Progression of these symptoms can lead to a more severe clinical presentation, including focal seizures in ≤10% of patients, and the mortality rate is 30% to 50%.3 Patients with comorbid HIV have a mortality rate ≤90%.2
Currently, there are no biomarkers that can identify PML in its early stages. A PML diagnosis is typically based on the patient’s clinical presentation, radiological imaging, and detection of JCV DNA. A brain biopsy is the gold standard for PML diagnosis.1
Interestingly, data suggest that glial cells harboring JCV in the brain express receptors for serotonin and dopamine.4 Researchers pinpointed 5HT2A receptors as JCV entry points into cells, and theorized that medications competing for binding, such as certain psychotropic agents, might decrease JCV entry. Cells lacking the 5HT2A receptor have shown immunity to JCV infection and the ability of cells to be infected was restored through transfection of 5HT2A receptors.4
Immunosuppressant medications can cause PML
PML was initially seen in individuals with conditions that cause immunosuppression, such as malignancies and HIV. However, “drug-induced PML” refers to cases in which drug-induced immunosuppression creates an environment that allows JCV to reactivate and disseminate back into the CNS.4 It is important to emphasize that drug-induced PML is a very rare effect of certain immunosuppressant medications. Medications that can weaken the immune system include glucocorticoids, monoclonal antibodies, alkylating agents, purine analogues, antimetabolites, and immunosuppressants (Table).1

These medications are used to treat conditions such as multiple sclerosis, RA, psoriatic arthritis, and lupus. Although drug-induced PML can result from the use of any of these agents, the highest incidence (1%) is found with natalizumab. Rates of incidence with other agents are either unknown or as low as .002%.1 Evidence suggests that the risk for PML increases with the duration of therapy.5
Continue to: Management
Management: Stop the offending agent, restore immune function
Specific pharmacologic treatments for PML are lacking. Management of drug-induced PML starts with discontinuing the offending agent. Restoring immune function has been found to be the most effective approach to treat PML.3 Restoration is possible through interleukin-2 (IL-2), IL-7, and T-cell infusions. Other treatment options are theoretical and include the development of a JCV vaccine to stimulate host response, plasma exchange to remove the medication from the host, and antiviral therapy targeting JCV replication. Diclofenac, isotretinoin, and mefloquine can inhibit JCV replication.3
Based on the theory that JCV requires 5HT2A receptors for entry into cells, researchers have studied medications that block this receptor as a treatment for PML. The first-generation antipsychotic chlorpromazine did not show benefit when combined with cidofovir, a replication inhibitor.3 Antipsychotics agents such as ziprasidone and olanzapine have shown in vitro inhibition of JCV, while risperidone has mixed results, with 1 trial failing to find a difference on JCV in fetal glial cells.3 Second-generation antipsychotics may be the preferred option due to more potent antagonism of the 5HT2A receptors and fewer adverse effects compared to agents such as chlorpromazine.4 The antidepressant mirtazapine has shown to have promising results, with evidence indicating that earlier initiation is more beneficial.3 Overall, data involving the use of medications that act on the 5HT2A receptor are mixed. Recent data suggest that JCV might enter cells independent of 5HT2A receptors; however, more research in this area is needed.2
The best strategy for treating drug-induced PML has not yet been determined. While combination therapy is thought to be more successful than monotherapy, ultimately, it depends on the patient’s immune response. If a psychotropic medication is chosen as adjunct treatment for drug-induced PML, it is prudent to assess the patient’s entire clinical picture to determine the specific indication for therapy (ie, treating symptomatology or drug-induced PML).
CASE CONTINUED
Following diagnosis, Mr. P is provided supportive therapy, and his care team discontinues methotrexate and etanercept. Although data are mixed on the efficacy of medications that work on 5HT2A receptors, because Mr. P was recently diagnosed with MDD, he is started on mirtazapine 15 mg/d at night in an attempt to manage both MDD and PML. It is possible that his depressive symptoms developed as a result of drug-induced PML rather than major depressive disorder. Discontinuing methotrexate and etanercept stabilizes Mr. P’s PML symptoms but leads to an exacerbation of his RA symptoms. Mr. P is initiated on hyd
Related Resources
- Castle D, Robertson NP. Treatment of progressive multifocal leukoencephalopathy. J Neurol. 2019;266(10):2587-2589. doi:10.1007/s00415-019-09501-y
Drug Brand Names
Abatacept • Orencia
Adalimumab • Humira
Alemtuzumab • Campath
Azathioprine • Azasan, Imuran
Basiliximab • Simulect
Belimumab • Benlysta
Bevacizumab • Avastin
Brentuximab vedotin • Adcetris
Cetuximab • Erbitux
Chlorpromazine • Thorazine, Largactil
Cidofovir • Vistide
Cladribine • Mavenclad
Cyclophosphamide • Cytoxan
Cyclosporine • Gengraf, Neoral
Dacarbazine • DTIC-Dome
Diclofenac • Cambia, Zorvolex
Dimethyl fumarate • Tecfidera
Etanercept • Enbrel
Fingolimod • Gilenya
Fludarabine • Fludara
Hydroxychloroquine • Plaquenil
Ibritumomab tiuxetan • Zevalin
Infliximab • Avsola, Inflectra
Isotretinoin • Absorica, Claravis
Mefloquine • Lariam
Methotrexate • Rheumatrex, Trexall
Mirtazapine • Remeron
Mitoxantrone • Novantrone
Muromonab-CD3 • Orthoclone OKT3
Mycophenolate mofetil • CellCept
Natalizumab • Tysabri
Nelarabine • Arranon
Obinutuzumab • Gazyva
Olanzapine • Zyprexa
Risperidone • Risperdal
Tacrolimus • Prograf
Vincristine • Vincasar PFS
Ziprasidone • Geodon
Mr. P, age 67, presents to the clinic with vision changes and memory loss following a fall in his home due to limb weakness. Six years ago, his care team diagnosed him with rheumatoid arthritis (RA). Mr. P’s current medication regimen includes methotrexate 20 mg once weekly and etanercept 50 mg once weekly, and he has been stable on this plan for 3 years. Mr. P also was recently diagnosed with major depressive disorder (MDD), but has not yet started treatment. Following a complete workup, an MRI of Mr. P’s brain revealed white matter demyelination. Due to these findings, he is scheduled for a brain biopsy, which confirms a diagnosis of progressive multifocal leukoencephalopathy (PML).
PML is a demyelinating disease of the central nervous system caused by the John Cunningham virus (JCV), or JC polyomavirus, named for the first patient identified to have contracted the virus.1 Asymptomatic infection of JCV often occurs in childhood, and antibodies are found in ≤70% of healthy adults. In most individuals, JCV remains latent in the kidneys and lymphoid organs, but immunosuppression can cause it to reactivate.2
JCV infects oligodendrocytes, astrocytes, and neurons, which results in white matter demyelination. Due to this demyelination, individuals can experience visual field defects, speech disturbances, ataxia, paresthesia, and cognitive impairments.2 Limb weakness presents in 60% of patients with PML, visual disturbances in 20%, and gait disturbances in 65%.3 Progression of these symptoms can lead to a more severe clinical presentation, including focal seizures in ≤10% of patients, and the mortality rate is 30% to 50%.3 Patients with comorbid HIV have a mortality rate ≤90%.2
Currently, there are no biomarkers that can identify PML in its early stages. A PML diagnosis is typically based on the patient’s clinical presentation, radiological imaging, and detection of JCV DNA. A brain biopsy is the gold standard for PML diagnosis.1
Interestingly, data suggest that glial cells harboring JCV in the brain express receptors for serotonin and dopamine.4 Researchers pinpointed 5HT2A receptors as JCV entry points into cells, and theorized that medications competing for binding, such as certain psychotropic agents, might decrease JCV entry. Cells lacking the 5HT2A receptor have shown immunity to JCV infection and the ability of cells to be infected was restored through transfection of 5HT2A receptors.4
Immunosuppressant medications can cause PML
PML was initially seen in individuals with conditions that cause immunosuppression, such as malignancies and HIV. However, “drug-induced PML” refers to cases in which drug-induced immunosuppression creates an environment that allows JCV to reactivate and disseminate back into the CNS.4 It is important to emphasize that drug-induced PML is a very rare effect of certain immunosuppressant medications. Medications that can weaken the immune system include glucocorticoids, monoclonal antibodies, alkylating agents, purine analogues, antimetabolites, and immunosuppressants (Table).1

These medications are used to treat conditions such as multiple sclerosis, RA, psoriatic arthritis, and lupus. Although drug-induced PML can result from the use of any of these agents, the highest incidence (1%) is found with natalizumab. Rates of incidence with other agents are either unknown or as low as .002%.1 Evidence suggests that the risk for PML increases with the duration of therapy.5
Continue to: Management
Management: Stop the offending agent, restore immune function
Specific pharmacologic treatments for PML are lacking. Management of drug-induced PML starts with discontinuing the offending agent. Restoring immune function has been found to be the most effective approach to treat PML.3 Restoration is possible through interleukin-2 (IL-2), IL-7, and T-cell infusions. Other treatment options are theoretical and include the development of a JCV vaccine to stimulate host response, plasma exchange to remove the medication from the host, and antiviral therapy targeting JCV replication. Diclofenac, isotretinoin, and mefloquine can inhibit JCV replication.3
Based on the theory that JCV requires 5HT2A receptors for entry into cells, researchers have studied medications that block this receptor as a treatment for PML. The first-generation antipsychotic chlorpromazine did not show benefit when combined with cidofovir, a replication inhibitor.3 Antipsychotics agents such as ziprasidone and olanzapine have shown in vitro inhibition of JCV, while risperidone has mixed results, with 1 trial failing to find a difference on JCV in fetal glial cells.3 Second-generation antipsychotics may be the preferred option due to more potent antagonism of the 5HT2A receptors and fewer adverse effects compared to agents such as chlorpromazine.4 The antidepressant mirtazapine has shown to have promising results, with evidence indicating that earlier initiation is more beneficial.3 Overall, data involving the use of medications that act on the 5HT2A receptor are mixed. Recent data suggest that JCV might enter cells independent of 5HT2A receptors; however, more research in this area is needed.2
The best strategy for treating drug-induced PML has not yet been determined. While combination therapy is thought to be more successful than monotherapy, ultimately, it depends on the patient’s immune response. If a psychotropic medication is chosen as adjunct treatment for drug-induced PML, it is prudent to assess the patient’s entire clinical picture to determine the specific indication for therapy (ie, treating symptomatology or drug-induced PML).
CASE CONTINUED
Following diagnosis, Mr. P is provided supportive therapy, and his care team discontinues methotrexate and etanercept. Although data are mixed on the efficacy of medications that work on 5HT2A receptors, because Mr. P was recently diagnosed with MDD, he is started on mirtazapine 15 mg/d at night in an attempt to manage both MDD and PML. It is possible that his depressive symptoms developed as a result of drug-induced PML rather than major depressive disorder. Discontinuing methotrexate and etanercept stabilizes Mr. P’s PML symptoms but leads to an exacerbation of his RA symptoms. Mr. P is initiated on hyd
Related Resources
- Castle D, Robertson NP. Treatment of progressive multifocal leukoencephalopathy. J Neurol. 2019;266(10):2587-2589. doi:10.1007/s00415-019-09501-y
Drug Brand Names
Abatacept • Orencia
Adalimumab • Humira
Alemtuzumab • Campath
Azathioprine • Azasan, Imuran
Basiliximab • Simulect
Belimumab • Benlysta
Bevacizumab • Avastin
Brentuximab vedotin • Adcetris
Cetuximab • Erbitux
Chlorpromazine • Thorazine, Largactil
Cidofovir • Vistide
Cladribine • Mavenclad
Cyclophosphamide • Cytoxan
Cyclosporine • Gengraf, Neoral
Dacarbazine • DTIC-Dome
Diclofenac • Cambia, Zorvolex
Dimethyl fumarate • Tecfidera
Etanercept • Enbrel
Fingolimod • Gilenya
Fludarabine • Fludara
Hydroxychloroquine • Plaquenil
Ibritumomab tiuxetan • Zevalin
Infliximab • Avsola, Inflectra
Isotretinoin • Absorica, Claravis
Mefloquine • Lariam
Methotrexate • Rheumatrex, Trexall
Mirtazapine • Remeron
Mitoxantrone • Novantrone
Muromonab-CD3 • Orthoclone OKT3
Mycophenolate mofetil • CellCept
Natalizumab • Tysabri
Nelarabine • Arranon
Obinutuzumab • Gazyva
Olanzapine • Zyprexa
Risperidone • Risperdal
Tacrolimus • Prograf
Vincristine • Vincasar PFS
Ziprasidone • Geodon
1. Yukitake M. Drug-induced progressive multifocal leukoencephalopathy in multiple sclerosis: a comprehensive review. Clin Exp Neuroimmunol. 2018;9(1):37-47. doi:10.1111/cen3.12440
2. Alstadhaug KB, Myhr KM, Rinaldo CH. Progressive multifocal leukoencephalopathy. Tidsskr Nor Laegeforen. 2017;137(23-24):10.4045/tidsskr.16.1092. doi:10.4045/tidsskr.16.1092
3. Williamson EML, Berger JR. Diagnosis and treatment of progressive multifocal leukoencephalopathy associated with multiple sclerosis therapies. Neurotherapeutics. 2017;14(4):961-973. doi:10.1007/s13311-017-0570-7
4. Altschuler EL, Kast RE. The atypical antipsychotic agents ziprasidone, risperidone and olanzapine as treatment for and prophylaxis against progressive multifocal leukoencephalopathy. Med Hypotheses. 2005;65(3):585-586.
5. Vinhas de Souza M, Keller-Stanislawski B, Blake K, et al. Drug-induced PML: a global agenda for a global challenge. Clin Pharmacol Ther. 2012;91(4):747-750. doi:10.1038/clpt.2012.4
1. Yukitake M. Drug-induced progressive multifocal leukoencephalopathy in multiple sclerosis: a comprehensive review. Clin Exp Neuroimmunol. 2018;9(1):37-47. doi:10.1111/cen3.12440
2. Alstadhaug KB, Myhr KM, Rinaldo CH. Progressive multifocal leukoencephalopathy. Tidsskr Nor Laegeforen. 2017;137(23-24):10.4045/tidsskr.16.1092. doi:10.4045/tidsskr.16.1092
3. Williamson EML, Berger JR. Diagnosis and treatment of progressive multifocal leukoencephalopathy associated with multiple sclerosis therapies. Neurotherapeutics. 2017;14(4):961-973. doi:10.1007/s13311-017-0570-7
4. Altschuler EL, Kast RE. The atypical antipsychotic agents ziprasidone, risperidone and olanzapine as treatment for and prophylaxis against progressive multifocal leukoencephalopathy. Med Hypotheses. 2005;65(3):585-586.
5. Vinhas de Souza M, Keller-Stanislawski B, Blake K, et al. Drug-induced PML: a global agenda for a global challenge. Clin Pharmacol Ther. 2012;91(4):747-750. doi:10.1038/clpt.2012.4
Hold or not to hold: Navigating involuntary commitment
CASE Depressed and suicidal
Police arrive at the home of Mr. H, age 50, after his wife calls 911. She reports he has depression and she saw him in bed brandishing a firearm as if he wanted to hurt himself. Upon arrival, the officers enter the house and find Mr. H in bed without a firearm. Mr. H says little to the officers about the alleged events, but acknowledges he has depression and is willing to go the hospital for further evaluation. Neither his wife nor the officers locate a firearm in the home.
EVALUATION Emergency detention
In the emergency department (ED), Mr. H’s laboratory results and physical examination findings are normal. He acknowledges feeling depressed over the past 2 weeks. Though he cannot identify any precipitants, he says he has experienced anhedonia, lack of appetite, decreased energy, and changes in his sleep patterns. When asked about the day’s events concerning the firearm, Mr. H becomes guarded and does not give a clear answer regarding having thoughts of suicide.
The evaluating psychiatrist obtains collateral from Mr. H’s wife and reviews his medical records. There are no active prescriptions on file and the psychiatrist notices that last year there was a suicide attempt involving a firearm. Following that episode, Mr. H was hospitalized, treated with sertraline 50 mg/d, and discharged with a diagnosis of major depressive disorder. There was no legal or substance abuse history.
In the ED, the psychiatrist conducts a psychiatric evaluation, including a suicide risk assessment, and determines Mr. H is at imminent risk of ending his life. Mr. H’s psychiatrist explains there are 2 treatment options: to be admitted to the hospital or to be discharged. The psychiatrist recommends hospital admission to Mr. H for his safety and stabilization. Mr. H says he prefers to return home.
Because the psychiatrist believes Mr. H is at imminent risk of ending his life and there is no less restrictive setting for treatment, he implements an emergency detention. In Ohio, this allows Mr. H to be held in the hospital for no more than 3 court days in accordance with state law. Before Mr. H’s emergency detention periods ends, the psychiatrist will need to decide whether Mr. H can be safely discharged. If the psychiatrist determines that Mr. H still needs treatment, the court will be petitioned for a civil commitment hearing.
[polldaddy:11189291]
The author’s observations
In some cases, courts allow information a psychiatrist does not directly obtain from a patient to be admitted as testimony in a civil commitment hearing. However, some jurisdictions consider sources of information not obtained directly from the patient as hearsay and thus inadmissible.1 Though each source listed may provide credible information that could be presented at a hearing, the psychiatrist should discuss with the patient the information obtained from these sources to ensure it is admissable.2 A discussion with Mr. H about the factors that led to his hospital arrival will avoid the psychiatrist’s reliance on what another person has heard or seen when providing testimony. Even when a psychiatrist is not faced with an issue of admissibility, caution must be taken with third-party reports.3
TREATMENT Civil commitment hearing
Before the emergency detention period expires, Mr. H’s psychiatrist determines that he remains at imminent risk of self-harm. To continue hospitalization, the psychiatrist files a petition for civil commitment and testifies at the commitment hearing. He reports that Mr. H suffers from a substantial mood disorder that grossly impairs his judgment and behavior. The psychiatrist also testifies that the least restrictive environment for treatment continues to be inpatient hospitalization, because Mr. H is still at imminent risk of harming himself.
Continue to: Following the psychiatrist's...
Following the psychiatrist’s testimony, the magistrate finds that Mr. H is a mentally ill person subject to hospitalization given his mood disorder that grossly impairs his judgment and behavior. The magistrate orders that Mr. H be civilly committed to the hospital.
[polldaddy:11189293]
The author’s observations
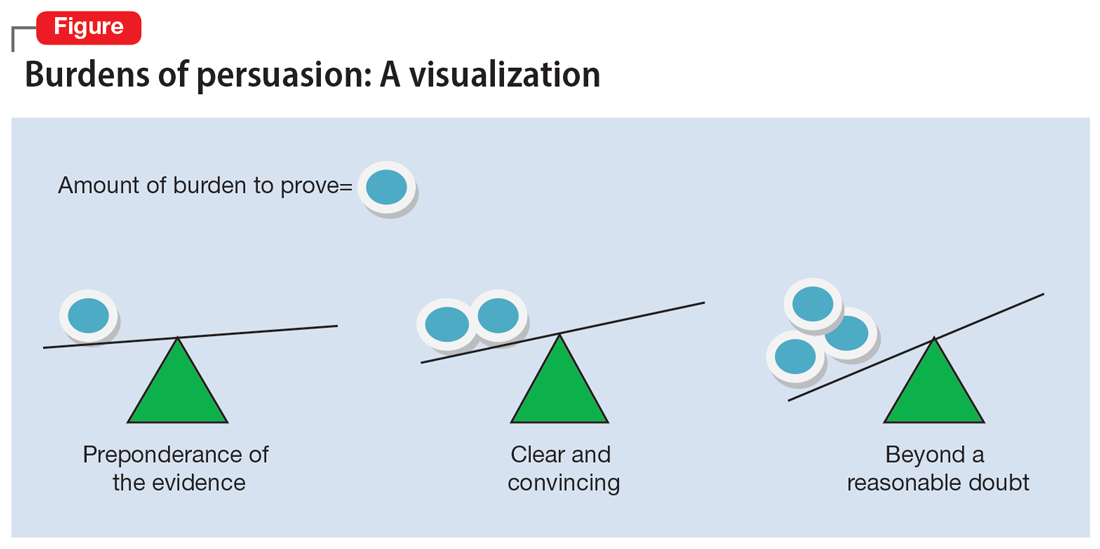
The psychiatrist’s testimony mirrors the language regarding civil commitment in the Ohio Revised Code.4 Other elements considered for mental illness in Ohio are a substantial disorder of memory, thought, orientation, or perception that grossly impairs one’s capacity to recognize reality or meet the demands of life.4 The definition of what constitutes a mental disorder varies by state, but the burden of persuasion—the standard by which the court must be convinced—is generally uniform.5 In the 1979 case Addington v Texas, the United States Supreme Court concluded that in a civil commitment hearing, the minimum standard of proof for involuntary commitment must be clear and convincing evidence.6 Neither medical certainty nor substantial probability are burdens of persuasions.6 Instead, these terms may be presented in a forensic report when an examiner outlines their opinion. Table 1 and the Figure provide more detail on burdens of persuasion.

TREATMENT Civil commitment and patient rights
At a regularly scheduled treatment team meeting, the team informs Mr. H that he has been civilly committed for further treatment. Mr. H becomes upset and tells the team the decision is a complete violation of his rights. After a long rant, Mr. H walks out of the room, saying, “I did not even know when this hearing was.” A member of the treatment team becomes concerned that Mr. H may not have been notified of the hearing.
[polldaddy:11189294]
The author’s observations
It is not clear if Mr. H had been notified of his civil commitment hearing. If Mr. H had not been notified, his rights would have been compromised. Lessard v Schmidt (1972) outlined that individuals involved in a civil commitment hearing should be afforded the same rights as those involved in criminal proceedings.7 Mr. H should have been notified of his hearing and afforded the opportunity to have the assignment of counsel, the right to appear, the right to testify, the right to present witnesses and other evidence, and the right to confront witnesses.
Without notification of the hearing, the only right that would have remained intact for Mr. H would have been the assignment of counsel in his absence and without his knowledge. If Mr. H had been notified of the hearing and did not want to attend, yet still desired legal counsel, he could have waived his presence voluntarily after discussing this option with his attorney.8,9
Continue to: OUTCOME Stabilization and discharge
OUTCOME Stabilization and discharge
During his 10-day stay, Mr. H is treated with sertraline 50 mg/d and engages in individual and group therapy. He shows noticeable improvement in his depressive symptoms and reports having no thoughts of suicide or self-harm. The treatment team determines it is appropriate to discharge him home (the firearm was never found) and involves his wife in safety planning and follow-up care. On the day of his discharge, Mr. H reflects on his treatment and civil commitment. He says, “I did not know a judge could order me to be hospitalized.”
[polldaddy:11189297]
The author’s observations
The physician’s decision to pursue civil commitment is best described by the legal doctrines of police powers and parens patriae. Other relevant ethical principles are described in Table 2.10

Though ethical principles may play a role in civil commitment, parens patriae and police powers is the answer with respect to the State.11Parens patriae is Latin for the “parent of the country” and grants the State the power to protect those residents who are most vulnerable. Police power is the authority of the State to enact and enforce rules that limit the rights of individuals for the greater good of ensuring health, safety, and welfare of all citizens.
Bottom Line
Psychiatrists are entrusted with recognizing when a patient, due to mental illness, is a danger to themselves or others and in need of treatment. After an emergency detention period, if the patient remains a danger to themselves or others and does not want to voluntarily receive treatment, a court hearing is required. As an expert witness, the treating psychiatrist should know the factors of law in their jurisdiction that determine civil commitment.
Related Resources
- Extreme Risk Protection Orders. Johns Hopkins Bloomberg School of Public Health. https://www.jhsph.edu/research/ centers-and-institutes/johns-hopkins-center-for-gun-violenceprevention-and-policy/research/extreme-risk-protectionorders/
- Gutheil TG. The Psychiatrist as Expert Witness. 2nd ed. American Psychiatric Association Publishing; 2009.
- Landmark Cases 2014. American Academy of Psychiatry and the Law. https://www.aapl.org/landmark-cases
Drug Brand Names
Sertraline • Zoloft
1. Pinals DA, Mossman D. Evaluation for Civil Commitment. Oxford University Press; 2012.
2. Thatcher BT, Mossman D. Testifying for civil commitment: help unwilling patients get the treatment they need. Current Psychiatry. 2009;8(11):51-56.
3. Marett CP, Mossman D. What is your liability for involuntary commitment based on faulty information? Current Psychiatry. 2017;16(3):21-25,33.
4. Ohio Rev Code § 5122.01 (2018).
5. The Burden of Proof. University of Minnesota. Accessed January 23, 2022. https://open.lib.umn.edu/criminallaw/chapter/2-4-the-burden-of-proof/
6. Gold LH, Frierson RL, eds. The American Psychiatric Association Publishing Textbook of Forensic Psychiatry. 3rd ed. American Psychiatric Association Publishing; 2018.
7. Gold LH, Frierson RL, eds. The American Psychiatric Association Publishing Textbook of Suicide Assessment and Management. 3rd ed. American Psychiatric Association Publishing; 2020.
8. Cook J. Good lawyering and bad role models: the role of respondent’s counsel in a civil commitment hearing. Georgetown Journal of Legal Ethics. 2000;14(1):179-195.
9. Ferris CE. The search for due process in civil commitment hearings: how procedural realities have altered substantive standards. Vanderbilt Law Rev. 2008;61(3):959-981.
10. Substance Abuse and Mental Health Services Administration. Civil Commitment and the Mental Health Care Continuum: Historical Trends and Principles for Law and Practice. 2019. Accessed January 23, 2022. https://www.samhsa.gov/resource/ebp/civil-commitment-mental-health-care-continuum-historical-trends-principles-law
11. Melton GB, Petrila J, Poythress NG, et al. Psychological Evaluations for the Courts: A Handbook for Mental Health Profession. 4th ed. Guilford Press; 2018.
CASE Depressed and suicidal
Police arrive at the home of Mr. H, age 50, after his wife calls 911. She reports he has depression and she saw him in bed brandishing a firearm as if he wanted to hurt himself. Upon arrival, the officers enter the house and find Mr. H in bed without a firearm. Mr. H says little to the officers about the alleged events, but acknowledges he has depression and is willing to go the hospital for further evaluation. Neither his wife nor the officers locate a firearm in the home.
EVALUATION Emergency detention
In the emergency department (ED), Mr. H’s laboratory results and physical examination findings are normal. He acknowledges feeling depressed over the past 2 weeks. Though he cannot identify any precipitants, he says he has experienced anhedonia, lack of appetite, decreased energy, and changes in his sleep patterns. When asked about the day’s events concerning the firearm, Mr. H becomes guarded and does not give a clear answer regarding having thoughts of suicide.
The evaluating psychiatrist obtains collateral from Mr. H’s wife and reviews his medical records. There are no active prescriptions on file and the psychiatrist notices that last year there was a suicide attempt involving a firearm. Following that episode, Mr. H was hospitalized, treated with sertraline 50 mg/d, and discharged with a diagnosis of major depressive disorder. There was no legal or substance abuse history.
In the ED, the psychiatrist conducts a psychiatric evaluation, including a suicide risk assessment, and determines Mr. H is at imminent risk of ending his life. Mr. H’s psychiatrist explains there are 2 treatment options: to be admitted to the hospital or to be discharged. The psychiatrist recommends hospital admission to Mr. H for his safety and stabilization. Mr. H says he prefers to return home.
Because the psychiatrist believes Mr. H is at imminent risk of ending his life and there is no less restrictive setting for treatment, he implements an emergency detention. In Ohio, this allows Mr. H to be held in the hospital for no more than 3 court days in accordance with state law. Before Mr. H’s emergency detention periods ends, the psychiatrist will need to decide whether Mr. H can be safely discharged. If the psychiatrist determines that Mr. H still needs treatment, the court will be petitioned for a civil commitment hearing.
[polldaddy:11189291]
The author’s observations
In some cases, courts allow information a psychiatrist does not directly obtain from a patient to be admitted as testimony in a civil commitment hearing. However, some jurisdictions consider sources of information not obtained directly from the patient as hearsay and thus inadmissible.1 Though each source listed may provide credible information that could be presented at a hearing, the psychiatrist should discuss with the patient the information obtained from these sources to ensure it is admissable.2 A discussion with Mr. H about the factors that led to his hospital arrival will avoid the psychiatrist’s reliance on what another person has heard or seen when providing testimony. Even when a psychiatrist is not faced with an issue of admissibility, caution must be taken with third-party reports.3
TREATMENT Civil commitment hearing
Before the emergency detention period expires, Mr. H’s psychiatrist determines that he remains at imminent risk of self-harm. To continue hospitalization, the psychiatrist files a petition for civil commitment and testifies at the commitment hearing. He reports that Mr. H suffers from a substantial mood disorder that grossly impairs his judgment and behavior. The psychiatrist also testifies that the least restrictive environment for treatment continues to be inpatient hospitalization, because Mr. H is still at imminent risk of harming himself.
Continue to: Following the psychiatrist's...
Following the psychiatrist’s testimony, the magistrate finds that Mr. H is a mentally ill person subject to hospitalization given his mood disorder that grossly impairs his judgment and behavior. The magistrate orders that Mr. H be civilly committed to the hospital.
[polldaddy:11189293]
The author’s observations
The psychiatrist’s testimony mirrors the language regarding civil commitment in the Ohio Revised Code.4 Other elements considered for mental illness in Ohio are a substantial disorder of memory, thought, orientation, or perception that grossly impairs one’s capacity to recognize reality or meet the demands of life.4 The definition of what constitutes a mental disorder varies by state, but the burden of persuasion—the standard by which the court must be convinced—is generally uniform.5 In the 1979 case Addington v Texas, the United States Supreme Court concluded that in a civil commitment hearing, the minimum standard of proof for involuntary commitment must be clear and convincing evidence.6 Neither medical certainty nor substantial probability are burdens of persuasions.6 Instead, these terms may be presented in a forensic report when an examiner outlines their opinion. Table 1 and the Figure provide more detail on burdens of persuasion.

TREATMENT Civil commitment and patient rights
At a regularly scheduled treatment team meeting, the team informs Mr. H that he has been civilly committed for further treatment. Mr. H becomes upset and tells the team the decision is a complete violation of his rights. After a long rant, Mr. H walks out of the room, saying, “I did not even know when this hearing was.” A member of the treatment team becomes concerned that Mr. H may not have been notified of the hearing.

[polldaddy:11189294]
The author’s observations
It is not clear if Mr. H had been notified of his civil commitment hearing. If Mr. H had not been notified, his rights would have been compromised. Lessard v Schmidt (1972) outlined that individuals involved in a civil commitment hearing should be afforded the same rights as those involved in criminal proceedings.7 Mr. H should have been notified of his hearing and afforded the opportunity to have the assignment of counsel, the right to appear, the right to testify, the right to present witnesses and other evidence, and the right to confront witnesses.
Without notification of the hearing, the only right that would have remained intact for Mr. H would have been the assignment of counsel in his absence and without his knowledge. If Mr. H had been notified of the hearing and did not want to attend, yet still desired legal counsel, he could have waived his presence voluntarily after discussing this option with his attorney.8,9
Continue to: OUTCOME Stabilization and discharge
OUTCOME Stabilization and discharge
During his 10-day stay, Mr. H is treated with sertraline 50 mg/d and engages in individual and group therapy. He shows noticeable improvement in his depressive symptoms and reports having no thoughts of suicide or self-harm. The treatment team determines it is appropriate to discharge him home (the firearm was never found) and involves his wife in safety planning and follow-up care. On the day of his discharge, Mr. H reflects on his treatment and civil commitment. He says, “I did not know a judge could order me to be hospitalized.”
[polldaddy:11189297]
The author’s observations
The physician’s decision to pursue civil commitment is best described by the legal doctrines of police powers and parens patriae. Other relevant ethical principles are described in Table 2.10

Though ethical principles may play a role in civil commitment, parens patriae and police powers is the answer with respect to the State.11Parens patriae is Latin for the “parent of the country” and grants the State the power to protect those residents who are most vulnerable. Police power is the authority of the State to enact and enforce rules that limit the rights of individuals for the greater good of ensuring health, safety, and welfare of all citizens.
Bottom Line
Psychiatrists are entrusted with recognizing when a patient, due to mental illness, is a danger to themselves or others and in need of treatment. After an emergency detention period, if the patient remains a danger to themselves or others and does not want to voluntarily receive treatment, a court hearing is required. As an expert witness, the treating psychiatrist should know the factors of law in their jurisdiction that determine civil commitment.
Related Resources
- Extreme Risk Protection Orders. Johns Hopkins Bloomberg School of Public Health. https://www.jhsph.edu/research/ centers-and-institutes/johns-hopkins-center-for-gun-violenceprevention-and-policy/research/extreme-risk-protectionorders/
- Gutheil TG. The Psychiatrist as Expert Witness. 2nd ed. American Psychiatric Association Publishing; 2009.
- Landmark Cases 2014. American Academy of Psychiatry and the Law. https://www.aapl.org/landmark-cases
Drug Brand Names
Sertraline • Zoloft
CASE Depressed and suicidal
Police arrive at the home of Mr. H, age 50, after his wife calls 911. She reports he has depression and she saw him in bed brandishing a firearm as if he wanted to hurt himself. Upon arrival, the officers enter the house and find Mr. H in bed without a firearm. Mr. H says little to the officers about the alleged events, but acknowledges he has depression and is willing to go the hospital for further evaluation. Neither his wife nor the officers locate a firearm in the home.
EVALUATION Emergency detention
In the emergency department (ED), Mr. H’s laboratory results and physical examination findings are normal. He acknowledges feeling depressed over the past 2 weeks. Though he cannot identify any precipitants, he says he has experienced anhedonia, lack of appetite, decreased energy, and changes in his sleep patterns. When asked about the day’s events concerning the firearm, Mr. H becomes guarded and does not give a clear answer regarding having thoughts of suicide.
The evaluating psychiatrist obtains collateral from Mr. H’s wife and reviews his medical records. There are no active prescriptions on file and the psychiatrist notices that last year there was a suicide attempt involving a firearm. Following that episode, Mr. H was hospitalized, treated with sertraline 50 mg/d, and discharged with a diagnosis of major depressive disorder. There was no legal or substance abuse history.
In the ED, the psychiatrist conducts a psychiatric evaluation, including a suicide risk assessment, and determines Mr. H is at imminent risk of ending his life. Mr. H’s psychiatrist explains there are 2 treatment options: to be admitted to the hospital or to be discharged. The psychiatrist recommends hospital admission to Mr. H for his safety and stabilization. Mr. H says he prefers to return home.
Because the psychiatrist believes Mr. H is at imminent risk of ending his life and there is no less restrictive setting for treatment, he implements an emergency detention. In Ohio, this allows Mr. H to be held in the hospital for no more than 3 court days in accordance with state law. Before Mr. H’s emergency detention periods ends, the psychiatrist will need to decide whether Mr. H can be safely discharged. If the psychiatrist determines that Mr. H still needs treatment, the court will be petitioned for a civil commitment hearing.
[polldaddy:11189291]
The author’s observations
In some cases, courts allow information a psychiatrist does not directly obtain from a patient to be admitted as testimony in a civil commitment hearing. However, some jurisdictions consider sources of information not obtained directly from the patient as hearsay and thus inadmissible.1 Though each source listed may provide credible information that could be presented at a hearing, the psychiatrist should discuss with the patient the information obtained from these sources to ensure it is admissable.2 A discussion with Mr. H about the factors that led to his hospital arrival will avoid the psychiatrist’s reliance on what another person has heard or seen when providing testimony. Even when a psychiatrist is not faced with an issue of admissibility, caution must be taken with third-party reports.3
TREATMENT Civil commitment hearing
Before the emergency detention period expires, Mr. H’s psychiatrist determines that he remains at imminent risk of self-harm. To continue hospitalization, the psychiatrist files a petition for civil commitment and testifies at the commitment hearing. He reports that Mr. H suffers from a substantial mood disorder that grossly impairs his judgment and behavior. The psychiatrist also testifies that the least restrictive environment for treatment continues to be inpatient hospitalization, because Mr. H is still at imminent risk of harming himself.
Continue to: Following the psychiatrist's...
Following the psychiatrist’s testimony, the magistrate finds that Mr. H is a mentally ill person subject to hospitalization given his mood disorder that grossly impairs his judgment and behavior. The magistrate orders that Mr. H be civilly committed to the hospital.
[polldaddy:11189293]
The author’s observations
The psychiatrist’s testimony mirrors the language regarding civil commitment in the Ohio Revised Code.4 Other elements considered for mental illness in Ohio are a substantial disorder of memory, thought, orientation, or perception that grossly impairs one’s capacity to recognize reality or meet the demands of life.4 The definition of what constitutes a mental disorder varies by state, but the burden of persuasion—the standard by which the court must be convinced—is generally uniform.5 In the 1979 case Addington v Texas, the United States Supreme Court concluded that in a civil commitment hearing, the minimum standard of proof for involuntary commitment must be clear and convincing evidence.6 Neither medical certainty nor substantial probability are burdens of persuasions.6 Instead, these terms may be presented in a forensic report when an examiner outlines their opinion. Table 1 and the Figure provide more detail on burdens of persuasion.

TREATMENT Civil commitment and patient rights
At a regularly scheduled treatment team meeting, the team informs Mr. H that he has been civilly committed for further treatment. Mr. H becomes upset and tells the team the decision is a complete violation of his rights. After a long rant, Mr. H walks out of the room, saying, “I did not even know when this hearing was.” A member of the treatment team becomes concerned that Mr. H may not have been notified of the hearing.

[polldaddy:11189294]
The author’s observations
It is not clear if Mr. H had been notified of his civil commitment hearing. If Mr. H had not been notified, his rights would have been compromised. Lessard v Schmidt (1972) outlined that individuals involved in a civil commitment hearing should be afforded the same rights as those involved in criminal proceedings.7 Mr. H should have been notified of his hearing and afforded the opportunity to have the assignment of counsel, the right to appear, the right to testify, the right to present witnesses and other evidence, and the right to confront witnesses.
Without notification of the hearing, the only right that would have remained intact for Mr. H would have been the assignment of counsel in his absence and without his knowledge. If Mr. H had been notified of the hearing and did not want to attend, yet still desired legal counsel, he could have waived his presence voluntarily after discussing this option with his attorney.8,9
Continue to: OUTCOME Stabilization and discharge
OUTCOME Stabilization and discharge
During his 10-day stay, Mr. H is treated with sertraline 50 mg/d and engages in individual and group therapy. He shows noticeable improvement in his depressive symptoms and reports having no thoughts of suicide or self-harm. The treatment team determines it is appropriate to discharge him home (the firearm was never found) and involves his wife in safety planning and follow-up care. On the day of his discharge, Mr. H reflects on his treatment and civil commitment. He says, “I did not know a judge could order me to be hospitalized.”
[polldaddy:11189297]
The author’s observations
The physician’s decision to pursue civil commitment is best described by the legal doctrines of police powers and parens patriae. Other relevant ethical principles are described in Table 2.10

Though ethical principles may play a role in civil commitment, parens patriae and police powers is the answer with respect to the State.11Parens patriae is Latin for the “parent of the country” and grants the State the power to protect those residents who are most vulnerable. Police power is the authority of the State to enact and enforce rules that limit the rights of individuals for the greater good of ensuring health, safety, and welfare of all citizens.
Bottom Line
Psychiatrists are entrusted with recognizing when a patient, due to mental illness, is a danger to themselves or others and in need of treatment. After an emergency detention period, if the patient remains a danger to themselves or others and does not want to voluntarily receive treatment, a court hearing is required. As an expert witness, the treating psychiatrist should know the factors of law in their jurisdiction that determine civil commitment.
Related Resources
- Extreme Risk Protection Orders. Johns Hopkins Bloomberg School of Public Health. https://www.jhsph.edu/research/ centers-and-institutes/johns-hopkins-center-for-gun-violenceprevention-and-policy/research/extreme-risk-protectionorders/
- Gutheil TG. The Psychiatrist as Expert Witness. 2nd ed. American Psychiatric Association Publishing; 2009.
- Landmark Cases 2014. American Academy of Psychiatry and the Law. https://www.aapl.org/landmark-cases
Drug Brand Names
Sertraline • Zoloft
1. Pinals DA, Mossman D. Evaluation for Civil Commitment. Oxford University Press; 2012.
2. Thatcher BT, Mossman D. Testifying for civil commitment: help unwilling patients get the treatment they need. Current Psychiatry. 2009;8(11):51-56.
3. Marett CP, Mossman D. What is your liability for involuntary commitment based on faulty information? Current Psychiatry. 2017;16(3):21-25,33.
4. Ohio Rev Code § 5122.01 (2018).
5. The Burden of Proof. University of Minnesota. Accessed January 23, 2022. https://open.lib.umn.edu/criminallaw/chapter/2-4-the-burden-of-proof/
6. Gold LH, Frierson RL, eds. The American Psychiatric Association Publishing Textbook of Forensic Psychiatry. 3rd ed. American Psychiatric Association Publishing; 2018.
7. Gold LH, Frierson RL, eds. The American Psychiatric Association Publishing Textbook of Suicide Assessment and Management. 3rd ed. American Psychiatric Association Publishing; 2020.
8. Cook J. Good lawyering and bad role models: the role of respondent’s counsel in a civil commitment hearing. Georgetown Journal of Legal Ethics. 2000;14(1):179-195.
9. Ferris CE. The search for due process in civil commitment hearings: how procedural realities have altered substantive standards. Vanderbilt Law Rev. 2008;61(3):959-981.
10. Substance Abuse and Mental Health Services Administration. Civil Commitment and the Mental Health Care Continuum: Historical Trends and Principles for Law and Practice. 2019. Accessed January 23, 2022. https://www.samhsa.gov/resource/ebp/civil-commitment-mental-health-care-continuum-historical-trends-principles-law
11. Melton GB, Petrila J, Poythress NG, et al. Psychological Evaluations for the Courts: A Handbook for Mental Health Profession. 4th ed. Guilford Press; 2018.
1. Pinals DA, Mossman D. Evaluation for Civil Commitment. Oxford University Press; 2012.
2. Thatcher BT, Mossman D. Testifying for civil commitment: help unwilling patients get the treatment they need. Current Psychiatry. 2009;8(11):51-56.
3. Marett CP, Mossman D. What is your liability for involuntary commitment based on faulty information? Current Psychiatry. 2017;16(3):21-25,33.
4. Ohio Rev Code § 5122.01 (2018).
5. The Burden of Proof. University of Minnesota. Accessed January 23, 2022. https://open.lib.umn.edu/criminallaw/chapter/2-4-the-burden-of-proof/
6. Gold LH, Frierson RL, eds. The American Psychiatric Association Publishing Textbook of Forensic Psychiatry. 3rd ed. American Psychiatric Association Publishing; 2018.
7. Gold LH, Frierson RL, eds. The American Psychiatric Association Publishing Textbook of Suicide Assessment and Management. 3rd ed. American Psychiatric Association Publishing; 2020.
8. Cook J. Good lawyering and bad role models: the role of respondent’s counsel in a civil commitment hearing. Georgetown Journal of Legal Ethics. 2000;14(1):179-195.
9. Ferris CE. The search for due process in civil commitment hearings: how procedural realities have altered substantive standards. Vanderbilt Law Rev. 2008;61(3):959-981.
10. Substance Abuse and Mental Health Services Administration. Civil Commitment and the Mental Health Care Continuum: Historical Trends and Principles for Law and Practice. 2019. Accessed January 23, 2022. https://www.samhsa.gov/resource/ebp/civil-commitment-mental-health-care-continuum-historical-trends-principles-law
11. Melton GB, Petrila J, Poythress NG, et al. Psychological Evaluations for the Courts: A Handbook for Mental Health Profession. 4th ed. Guilford Press; 2018.
Preparing patients with serious mental illness for extreme HEAT
Climate change is causing intense heat waves that threaten human health across the globe.
A confluence of factors increases risk
Thermoregulatory dysfunction is thought to be intrinsic to patients with schizophrenia partly due to dysregulated dopaminergic neurotransmission.2 This is compounded by these patients’ higher burden of chronic medical comorbidities such as cardiovascular and respiratory illnesses, which together with psychotropic (ie, antipsychotics, antidepressants, lithium, benzodiazepines) and medical medications (ie, certain antihypertensives, diuretics, treatment for urinary incontinence) further disrupt the body’s cooling strategies and increase vulnerability to heat-related illnesses.1,3 Antipsychotics commonly prescribed to patients with SMI increase hyperthermia risk largely by 2 mechanisms: central and peripheral thermal dysregulation, and anticholinergic properties (ie, olanzapine, clozapine, chlorpromazine).2,3 Other anticholinergic medications prescribed to treat extrapyramidal symptoms (ie, diphenhydramine, benztropine, trihexyphenidyl), anxiety, depression, and insomnia (ie, paroxetine, trazodone, doxepin) further add insult to injury because they impair sweating, which decreases the body’s ability to eliminate heat through evaporation.2,3 Additionally, high temperature exacerbates psychiatric symptoms in patients with SMI, resulting in increased hospitalizations and emergency department visits.
How to keep patients safe
The acronym HEAT provides a framework that psychiatrists can use to highlight the importance of planning for heat waves in their institution and guiding discussions with individual patients about heat-related illnesses (Table 1).

Help the health care system where you work plan and prepare for heat waves. In-service training in mental health settings such as outpatient clinics, shelters, group homes, and residential programs can help staff identify patients at particular risk and reinforce key prevention messages.
Educate patients and their caregivers on strategies for preventing heat-related illness. Informational materials can be distributed in clinics, residential settings, and day programs. A 1-page downloadable pamphlet available at https://smiadviser.org/wp-content/uploads/2022/08/SMI-Heat-Stroke-ver1.0-FINAL.pdf summarizes key prevention messages of staying hydrated, staying cool, and staying safe.
Assess personalized heat-related risks. Inquire about patients’ daily activities, access to air conditioning, and water intake. Minimize the use of anticholinergic medications. Identify who patients can turn to for assistance, especially for those who struggle with cognitive impairment and social isolation.
Teach patients, caregivers, and staff the signs and symptoms of heat exhaustion and heat stroke and how to respond in such situations.
HEAT focuses psychiatric clinicians on preparing and protecting patients with SMI against dangerous heat waves. Clinicians can take a proactive leadership role in disseminating basic principles of heat-related illness prevention and heat-wave toolkits by using resources available from organizations such as the Climate Psychiatry Alliance (Table 2). They can also initiate advocacy efforts to raise awareness about the elevated risks of heat-related illnesses in this vulnerable population.

1. Schmeltz MT, Gamble JL. Risk characterization of hospitalizations for mental illness and/or behavioral disorders with concurrent heat-related illness. PLoS One. 2017;12(10):e0186509. doi:10.1371/journal.pone.0186509
2. Lee CP, Chen PJ, Chang CM. Heat stroke during treatment with olanzapine, trihexyphenidyl, and trazodone in a patient with schizophrenia. Acta Neuropsychiatrica. 2015;27(6):380-385.
3. Bongers KS, Salahudeen MS, Peterson GM. Drug-associated non-pyrogenic hyperthermia: a narrative review. Eur J Clin Pharmacol. 2020;76(1):9-16.
Climate change is causing intense heat waves that threaten human health across the globe.
A confluence of factors increases risk
Thermoregulatory dysfunction is thought to be intrinsic to patients with schizophrenia partly due to dysregulated dopaminergic neurotransmission.2 This is compounded by these patients’ higher burden of chronic medical comorbidities such as cardiovascular and respiratory illnesses, which together with psychotropic (ie, antipsychotics, antidepressants, lithium, benzodiazepines) and medical medications (ie, certain antihypertensives, diuretics, treatment for urinary incontinence) further disrupt the body’s cooling strategies and increase vulnerability to heat-related illnesses.1,3 Antipsychotics commonly prescribed to patients with SMI increase hyperthermia risk largely by 2 mechanisms: central and peripheral thermal dysregulation, and anticholinergic properties (ie, olanzapine, clozapine, chlorpromazine).2,3 Other anticholinergic medications prescribed to treat extrapyramidal symptoms (ie, diphenhydramine, benztropine, trihexyphenidyl), anxiety, depression, and insomnia (ie, paroxetine, trazodone, doxepin) further add insult to injury because they impair sweating, which decreases the body’s ability to eliminate heat through evaporation.2,3 Additionally, high temperature exacerbates psychiatric symptoms in patients with SMI, resulting in increased hospitalizations and emergency department visits.
How to keep patients safe
The acronym HEAT provides a framework that psychiatrists can use to highlight the importance of planning for heat waves in their institution and guiding discussions with individual patients about heat-related illnesses (Table 1).

Help the health care system where you work plan and prepare for heat waves. In-service training in mental health settings such as outpatient clinics, shelters, group homes, and residential programs can help staff identify patients at particular risk and reinforce key prevention messages.
Educate patients and their caregivers on strategies for preventing heat-related illness. Informational materials can be distributed in clinics, residential settings, and day programs. A 1-page downloadable pamphlet available at https://smiadviser.org/wp-content/uploads/2022/08/SMI-Heat-Stroke-ver1.0-FINAL.pdf summarizes key prevention messages of staying hydrated, staying cool, and staying safe.
Assess personalized heat-related risks. Inquire about patients’ daily activities, access to air conditioning, and water intake. Minimize the use of anticholinergic medications. Identify who patients can turn to for assistance, especially for those who struggle with cognitive impairment and social isolation.
Teach patients, caregivers, and staff the signs and symptoms of heat exhaustion and heat stroke and how to respond in such situations.
HEAT focuses psychiatric clinicians on preparing and protecting patients with SMI against dangerous heat waves. Clinicians can take a proactive leadership role in disseminating basic principles of heat-related illness prevention and heat-wave toolkits by using resources available from organizations such as the Climate Psychiatry Alliance (Table 2). They can also initiate advocacy efforts to raise awareness about the elevated risks of heat-related illnesses in this vulnerable population.

Climate change is causing intense heat waves that threaten human health across the globe.
A confluence of factors increases risk
Thermoregulatory dysfunction is thought to be intrinsic to patients with schizophrenia partly due to dysregulated dopaminergic neurotransmission.2 This is compounded by these patients’ higher burden of chronic medical comorbidities such as cardiovascular and respiratory illnesses, which together with psychotropic (ie, antipsychotics, antidepressants, lithium, benzodiazepines) and medical medications (ie, certain antihypertensives, diuretics, treatment for urinary incontinence) further disrupt the body’s cooling strategies and increase vulnerability to heat-related illnesses.1,3 Antipsychotics commonly prescribed to patients with SMI increase hyperthermia risk largely by 2 mechanisms: central and peripheral thermal dysregulation, and anticholinergic properties (ie, olanzapine, clozapine, chlorpromazine).2,3 Other anticholinergic medications prescribed to treat extrapyramidal symptoms (ie, diphenhydramine, benztropine, trihexyphenidyl), anxiety, depression, and insomnia (ie, paroxetine, trazodone, doxepin) further add insult to injury because they impair sweating, which decreases the body’s ability to eliminate heat through evaporation.2,3 Additionally, high temperature exacerbates psychiatric symptoms in patients with SMI, resulting in increased hospitalizations and emergency department visits.
How to keep patients safe
The acronym HEAT provides a framework that psychiatrists can use to highlight the importance of planning for heat waves in their institution and guiding discussions with individual patients about heat-related illnesses (Table 1).

Help the health care system where you work plan and prepare for heat waves. In-service training in mental health settings such as outpatient clinics, shelters, group homes, and residential programs can help staff identify patients at particular risk and reinforce key prevention messages.
Educate patients and their caregivers on strategies for preventing heat-related illness. Informational materials can be distributed in clinics, residential settings, and day programs. A 1-page downloadable pamphlet available at https://smiadviser.org/wp-content/uploads/2022/08/SMI-Heat-Stroke-ver1.0-FINAL.pdf summarizes key prevention messages of staying hydrated, staying cool, and staying safe.
Assess personalized heat-related risks. Inquire about patients’ daily activities, access to air conditioning, and water intake. Minimize the use of anticholinergic medications. Identify who patients can turn to for assistance, especially for those who struggle with cognitive impairment and social isolation.
Teach patients, caregivers, and staff the signs and symptoms of heat exhaustion and heat stroke and how to respond in such situations.
HEAT focuses psychiatric clinicians on preparing and protecting patients with SMI against dangerous heat waves. Clinicians can take a proactive leadership role in disseminating basic principles of heat-related illness prevention and heat-wave toolkits by using resources available from organizations such as the Climate Psychiatry Alliance (Table 2). They can also initiate advocacy efforts to raise awareness about the elevated risks of heat-related illnesses in this vulnerable population.

1. Schmeltz MT, Gamble JL. Risk characterization of hospitalizations for mental illness and/or behavioral disorders with concurrent heat-related illness. PLoS One. 2017;12(10):e0186509. doi:10.1371/journal.pone.0186509
2. Lee CP, Chen PJ, Chang CM. Heat stroke during treatment with olanzapine, trihexyphenidyl, and trazodone in a patient with schizophrenia. Acta Neuropsychiatrica. 2015;27(6):380-385.
3. Bongers KS, Salahudeen MS, Peterson GM. Drug-associated non-pyrogenic hyperthermia: a narrative review. Eur J Clin Pharmacol. 2020;76(1):9-16.
1. Schmeltz MT, Gamble JL. Risk characterization of hospitalizations for mental illness and/or behavioral disorders with concurrent heat-related illness. PLoS One. 2017;12(10):e0186509. doi:10.1371/journal.pone.0186509
2. Lee CP, Chen PJ, Chang CM. Heat stroke during treatment with olanzapine, trihexyphenidyl, and trazodone in a patient with schizophrenia. Acta Neuropsychiatrica. 2015;27(6):380-385.
3. Bongers KS, Salahudeen MS, Peterson GM. Drug-associated non-pyrogenic hyperthermia: a narrative review. Eur J Clin Pharmacol. 2020;76(1):9-16.
Lithium for bipolar disorder: Which patients will respond?
Though Cade discovered it 70 years ago, lithium is still considered the gold standard treatment for preventing manic and depressive phases of bipolar disorder (BD). In addition to its primary indication as a mood stabilizer, lithium has demonstrated efficacy as an augmenting medication for unipolar major depressive disorder.1 While lithium is a first-line agent for BD, it does not improve symptoms in every patient. In a 2004 meta-analysis of 5 randomized controlled trials of patients with BD, Geddes et al2 found lithium was more effective than placebo in preventing the recurrence of mania, with 60% in the lithium group remaining stable compared to 40% in the placebo group. Being able to predict which patients will respond to lithium is crucial to prevent unnecessary exposure to lithium, which can produce significant adverse effects, including somnolence, nausea, diarrhea, and hypothyroidism.2
Several studies have investigated various clinical factors that might predict which patients with BD will respond to lithium. In a review, Kleindienst et al3 highlighted 3 factors that predicted a positive response to lithium:
- fewer hospitalizations prior to treatment
- an episodic course characterized sequentially by mania, depression, and then euthymia
- a later age (>50) at onset of BD.
Recent studies and reviews have isolated additional positive predictors, including having a family history of BD and a shorter duration of illness before receiving lithium, as well as negative predictors, such as rapid cycling, a large number of previous hospitalizations, a depression/mania/euthymia pattern, mood-incongruent psychotic features, and the presence of residual symptoms between mood episodes.3,4
The Table provides a list of probable and possible positive and negative predictors for therapeutic response to lithium in patients with BD.3-6 While relevant, the factors listed as possible predictors may not carry as much influence on lithium responsivity as those categorized as probable predictors.

Because of heterogeneity among studies, clinicians should consider their patient’s presentation as a whole, rather than basing medication choice on independent factors. Ultimately, more studies are required to fully determine the most relevant clinical parameters for lithium response. Overall, however, it appears these clinical factors could be extremely useful to guide psychiatrists in the optimal use of lithium while caring for patients with BD.
1. Crossley NA, Bauer M. Acceleration and augmentation of antidepressants with lithium for depressive disorders: two meta-analyses of randomized, placebo-controlled trials. J Clin Psychiatry. 2007;68(6):935-940.
2. Geddes JR, Burgess S, Hawton K, et al. Long-term lithium therapy for bipolar disorder: systematic review and meta-analysis of randomized controlled trials. Am J Psychiatry. 2004;1m61(2):217-222.
3. Kleindienst N, Engel RR, Greil W. Which clinical factors predict response to prophylactic lithium? A systematic review for bipolar disorders. Bipolar Disord. 2005;7(5):404-417.
4. Kleindienst N, Engel RR, Greil W. Psychosocial and demographic factors associated with response to prophylactic lithium: a systematic review for bipolar disorders. Psychol Med. 2005;35(12):1685-1694.
5. Hui TP, Kandola A, Shen L, et al. A systematic review and meta-analysis of clinical predictors of lithium response in bipolar disorder. Acta Psychiatr Scand. 2019;140(2):94-115.
6. Grillault Laroche D, Etain B, Severus E, et al. Socio-demographic and clinical predictors of outcome to long-term treatment with lithium in bipolar disorders: a systematic review of the contemporary literature and recommendations from the ISBD/IGSLI Task Force on treatment with lithium. Int J Bipolar Disord. 2020;8(1):40.
Though Cade discovered it 70 years ago, lithium is still considered the gold standard treatment for preventing manic and depressive phases of bipolar disorder (BD). In addition to its primary indication as a mood stabilizer, lithium has demonstrated efficacy as an augmenting medication for unipolar major depressive disorder.1 While lithium is a first-line agent for BD, it does not improve symptoms in every patient. In a 2004 meta-analysis of 5 randomized controlled trials of patients with BD, Geddes et al2 found lithium was more effective than placebo in preventing the recurrence of mania, with 60% in the lithium group remaining stable compared to 40% in the placebo group. Being able to predict which patients will respond to lithium is crucial to prevent unnecessary exposure to lithium, which can produce significant adverse effects, including somnolence, nausea, diarrhea, and hypothyroidism.2
Several studies have investigated various clinical factors that might predict which patients with BD will respond to lithium. In a review, Kleindienst et al3 highlighted 3 factors that predicted a positive response to lithium:
- fewer hospitalizations prior to treatment
- an episodic course characterized sequentially by mania, depression, and then euthymia
- a later age (>50) at onset of BD.
Recent studies and reviews have isolated additional positive predictors, including having a family history of BD and a shorter duration of illness before receiving lithium, as well as negative predictors, such as rapid cycling, a large number of previous hospitalizations, a depression/mania/euthymia pattern, mood-incongruent psychotic features, and the presence of residual symptoms between mood episodes.3,4
The Table provides a list of probable and possible positive and negative predictors for therapeutic response to lithium in patients with BD.3-6 While relevant, the factors listed as possible predictors may not carry as much influence on lithium responsivity as those categorized as probable predictors.

Because of heterogeneity among studies, clinicians should consider their patient’s presentation as a whole, rather than basing medication choice on independent factors. Ultimately, more studies are required to fully determine the most relevant clinical parameters for lithium response. Overall, however, it appears these clinical factors could be extremely useful to guide psychiatrists in the optimal use of lithium while caring for patients with BD.
Though Cade discovered it 70 years ago, lithium is still considered the gold standard treatment for preventing manic and depressive phases of bipolar disorder (BD). In addition to its primary indication as a mood stabilizer, lithium has demonstrated efficacy as an augmenting medication for unipolar major depressive disorder.1 While lithium is a first-line agent for BD, it does not improve symptoms in every patient. In a 2004 meta-analysis of 5 randomized controlled trials of patients with BD, Geddes et al2 found lithium was more effective than placebo in preventing the recurrence of mania, with 60% in the lithium group remaining stable compared to 40% in the placebo group. Being able to predict which patients will respond to lithium is crucial to prevent unnecessary exposure to lithium, which can produce significant adverse effects, including somnolence, nausea, diarrhea, and hypothyroidism.2
Several studies have investigated various clinical factors that might predict which patients with BD will respond to lithium. In a review, Kleindienst et al3 highlighted 3 factors that predicted a positive response to lithium:
- fewer hospitalizations prior to treatment
- an episodic course characterized sequentially by mania, depression, and then euthymia
- a later age (>50) at onset of BD.
Recent studies and reviews have isolated additional positive predictors, including having a family history of BD and a shorter duration of illness before receiving lithium, as well as negative predictors, such as rapid cycling, a large number of previous hospitalizations, a depression/mania/euthymia pattern, mood-incongruent psychotic features, and the presence of residual symptoms between mood episodes.3,4
The Table provides a list of probable and possible positive and negative predictors for therapeutic response to lithium in patients with BD.3-6 While relevant, the factors listed as possible predictors may not carry as much influence on lithium responsivity as those categorized as probable predictors.

Because of heterogeneity among studies, clinicians should consider their patient’s presentation as a whole, rather than basing medication choice on independent factors. Ultimately, more studies are required to fully determine the most relevant clinical parameters for lithium response. Overall, however, it appears these clinical factors could be extremely useful to guide psychiatrists in the optimal use of lithium while caring for patients with BD.
1. Crossley NA, Bauer M. Acceleration and augmentation of antidepressants with lithium for depressive disorders: two meta-analyses of randomized, placebo-controlled trials. J Clin Psychiatry. 2007;68(6):935-940.
2. Geddes JR, Burgess S, Hawton K, et al. Long-term lithium therapy for bipolar disorder: systematic review and meta-analysis of randomized controlled trials. Am J Psychiatry. 2004;1m61(2):217-222.
3. Kleindienst N, Engel RR, Greil W. Which clinical factors predict response to prophylactic lithium? A systematic review for bipolar disorders. Bipolar Disord. 2005;7(5):404-417.
4. Kleindienst N, Engel RR, Greil W. Psychosocial and demographic factors associated with response to prophylactic lithium: a systematic review for bipolar disorders. Psychol Med. 2005;35(12):1685-1694.
5. Hui TP, Kandola A, Shen L, et al. A systematic review and meta-analysis of clinical predictors of lithium response in bipolar disorder. Acta Psychiatr Scand. 2019;140(2):94-115.
6. Grillault Laroche D, Etain B, Severus E, et al. Socio-demographic and clinical predictors of outcome to long-term treatment with lithium in bipolar disorders: a systematic review of the contemporary literature and recommendations from the ISBD/IGSLI Task Force on treatment with lithium. Int J Bipolar Disord. 2020;8(1):40.
1. Crossley NA, Bauer M. Acceleration and augmentation of antidepressants with lithium for depressive disorders: two meta-analyses of randomized, placebo-controlled trials. J Clin Psychiatry. 2007;68(6):935-940.
2. Geddes JR, Burgess S, Hawton K, et al. Long-term lithium therapy for bipolar disorder: systematic review and meta-analysis of randomized controlled trials. Am J Psychiatry. 2004;1m61(2):217-222.
3. Kleindienst N, Engel RR, Greil W. Which clinical factors predict response to prophylactic lithium? A systematic review for bipolar disorders. Bipolar Disord. 2005;7(5):404-417.
4. Kleindienst N, Engel RR, Greil W. Psychosocial and demographic factors associated with response to prophylactic lithium: a systematic review for bipolar disorders. Psychol Med. 2005;35(12):1685-1694.
5. Hui TP, Kandola A, Shen L, et al. A systematic review and meta-analysis of clinical predictors of lithium response in bipolar disorder. Acta Psychiatr Scand. 2019;140(2):94-115.
6. Grillault Laroche D, Etain B, Severus E, et al. Socio-demographic and clinical predictors of outcome to long-term treatment with lithium in bipolar disorders: a systematic review of the contemporary literature and recommendations from the ISBD/IGSLI Task Force on treatment with lithium. Int J Bipolar Disord. 2020;8(1):40.
Melatonin as a sleep aid: Are you prescribing it correctly?
Difficulty achieving regular restorative sleep is a common symptom of many psychiatric illnesses and can pose a pharmaceutical challenge, particularly for patients who have contraindications to benzodiazepines or sedative-hypnotics. Melatonin is commonly used to treat insomnia and circadian rhythm disorders in hospitalized patients because it is largely considered safe, nonhabit forming, unlikely to interact with other medications, and possibly protective against delirium.1 We support its short-term use in patients with sleep disruption, even if they do not meet the diagnostic criteria for insomnia or a circadian rhythm sleep-wake disorder. However, this use should be guided by consideration of the known physiological actions of melatonin, and not by an assumption that it acts as a simple sedative-hypnotic.
How melatonin works
Melatonin is an endogenous neurohormone involved in circadian rhythm regulation (sleep/wake regulation), a fundamental process in the functioning of the CNS and in the development of psychiatric disorders.2 Melatonin is commonly described as a sleep-promoting neurotransmitter, but it is more accurately described as a “darkness hormone.”3 With an onset at dusk and offset at sunrise, melatonin is the signal for biological night, not the signal for sleep. Melanopsin-containing retina neurons sensitive to blue light sense the diminishing light of the evening and communicate this cue to the brain’s master clock in the suprachiasmatic nucleus (SCN) of the hypothalamus (via the retinohypothalamic pathway). The SCN then releases its inhibition on the pineal gland, allowing it to release melatonin into the bloodstream and CSF. The timing of this release is known as the dim-light melatonin onset (DLMO).
Selecting the optimal timing and dose
Studies in laboratory and home settings have consistently shown that the DLMO precedes the onset of sleep by approximately 2 to 4 hours.4 Thus, we recommend scheduling melatonin administration for 2 to 4 hours before the intended bedtime.
Lower doses better replicate physiological levels of melatonin. A lower dose is also less likely to lead to a compromise of the entrainment process and the induction of a delayed sleep phase due to the lingering presence of melatonin, or the phase-delaying effects of a strong melatonin signal much later than the ideal DLMO. Giving higher doses at bedtime will induce sleep but may cause a circadian phase delay, effectively “jet lagging” the patient. We recommend prescribing low-dose melatonin (LDM; 0.5 to 1 mg) 2 to 4 hours before the intended bedtime rather than higher doses (≥5 mg) given at bedtime as is common practice and recommended by many melatonin manufacturers. LDM better simulates the natural release and function of melatonin and avoids potential adverse circadian phase delays. T
1. Joseph SG. Melatonin supplementation for the prevention of hospital-associated delirium. Ment Health Clin. 2018;7(4):143-146. doi:10.9740/mhc.2017.07.143
2. Arendt J, Skene DJ. Melatonin as a chronobiotic. Sleep Med Rev. 2005;9(1):25-39. doi:10.1016/j.smrv.2004.05.002
3. Tallavajhula S, Rodgers JJ, Slater JD. Sleep and sleep-wake disorders. In: Arciniengas DB, Yudofsky SC, Hales RE, eds. Textbook of Neuropsychiatry and Clinical Neurosciences. American Psychiatric Association Publishing; 2018:373-393.
4. Sletten TL, Vincenzi S, Redman JR, et al. Timing of sleep and its relationship with the endogenous melatonin rhythm. Front Neurol. 2010;1:137. doi:10.3389/fneur.2010.00137
Difficulty achieving regular restorative sleep is a common symptom of many psychiatric illnesses and can pose a pharmaceutical challenge, particularly for patients who have contraindications to benzodiazepines or sedative-hypnotics. Melatonin is commonly used to treat insomnia and circadian rhythm disorders in hospitalized patients because it is largely considered safe, nonhabit forming, unlikely to interact with other medications, and possibly protective against delirium.1 We support its short-term use in patients with sleep disruption, even if they do not meet the diagnostic criteria for insomnia or a circadian rhythm sleep-wake disorder. However, this use should be guided by consideration of the known physiological actions of melatonin, and not by an assumption that it acts as a simple sedative-hypnotic.
How melatonin works
Melatonin is an endogenous neurohormone involved in circadian rhythm regulation (sleep/wake regulation), a fundamental process in the functioning of the CNS and in the development of psychiatric disorders.2 Melatonin is commonly described as a sleep-promoting neurotransmitter, but it is more accurately described as a “darkness hormone.”3 With an onset at dusk and offset at sunrise, melatonin is the signal for biological night, not the signal for sleep. Melanopsin-containing retina neurons sensitive to blue light sense the diminishing light of the evening and communicate this cue to the brain’s master clock in the suprachiasmatic nucleus (SCN) of the hypothalamus (via the retinohypothalamic pathway). The SCN then releases its inhibition on the pineal gland, allowing it to release melatonin into the bloodstream and CSF. The timing of this release is known as the dim-light melatonin onset (DLMO).
Selecting the optimal timing and dose
Studies in laboratory and home settings have consistently shown that the DLMO precedes the onset of sleep by approximately 2 to 4 hours.4 Thus, we recommend scheduling melatonin administration for 2 to 4 hours before the intended bedtime.
Lower doses better replicate physiological levels of melatonin. A lower dose is also less likely to lead to a compromise of the entrainment process and the induction of a delayed sleep phase due to the lingering presence of melatonin, or the phase-delaying effects of a strong melatonin signal much later than the ideal DLMO. Giving higher doses at bedtime will induce sleep but may cause a circadian phase delay, effectively “jet lagging” the patient. We recommend prescribing low-dose melatonin (LDM; 0.5 to 1 mg) 2 to 4 hours before the intended bedtime rather than higher doses (≥5 mg) given at bedtime as is common practice and recommended by many melatonin manufacturers. LDM better simulates the natural release and function of melatonin and avoids potential adverse circadian phase delays. T
Difficulty achieving regular restorative sleep is a common symptom of many psychiatric illnesses and can pose a pharmaceutical challenge, particularly for patients who have contraindications to benzodiazepines or sedative-hypnotics. Melatonin is commonly used to treat insomnia and circadian rhythm disorders in hospitalized patients because it is largely considered safe, nonhabit forming, unlikely to interact with other medications, and possibly protective against delirium.1 We support its short-term use in patients with sleep disruption, even if they do not meet the diagnostic criteria for insomnia or a circadian rhythm sleep-wake disorder. However, this use should be guided by consideration of the known physiological actions of melatonin, and not by an assumption that it acts as a simple sedative-hypnotic.
How melatonin works
Melatonin is an endogenous neurohormone involved in circadian rhythm regulation (sleep/wake regulation), a fundamental process in the functioning of the CNS and in the development of psychiatric disorders.2 Melatonin is commonly described as a sleep-promoting neurotransmitter, but it is more accurately described as a “darkness hormone.”3 With an onset at dusk and offset at sunrise, melatonin is the signal for biological night, not the signal for sleep. Melanopsin-containing retina neurons sensitive to blue light sense the diminishing light of the evening and communicate this cue to the brain’s master clock in the suprachiasmatic nucleus (SCN) of the hypothalamus (via the retinohypothalamic pathway). The SCN then releases its inhibition on the pineal gland, allowing it to release melatonin into the bloodstream and CSF. The timing of this release is known as the dim-light melatonin onset (DLMO).
Selecting the optimal timing and dose
Studies in laboratory and home settings have consistently shown that the DLMO precedes the onset of sleep by approximately 2 to 4 hours.4 Thus, we recommend scheduling melatonin administration for 2 to 4 hours before the intended bedtime.
Lower doses better replicate physiological levels of melatonin. A lower dose is also less likely to lead to a compromise of the entrainment process and the induction of a delayed sleep phase due to the lingering presence of melatonin, or the phase-delaying effects of a strong melatonin signal much later than the ideal DLMO. Giving higher doses at bedtime will induce sleep but may cause a circadian phase delay, effectively “jet lagging” the patient. We recommend prescribing low-dose melatonin (LDM; 0.5 to 1 mg) 2 to 4 hours before the intended bedtime rather than higher doses (≥5 mg) given at bedtime as is common practice and recommended by many melatonin manufacturers. LDM better simulates the natural release and function of melatonin and avoids potential adverse circadian phase delays. T
1. Joseph SG. Melatonin supplementation for the prevention of hospital-associated delirium. Ment Health Clin. 2018;7(4):143-146. doi:10.9740/mhc.2017.07.143
2. Arendt J, Skene DJ. Melatonin as a chronobiotic. Sleep Med Rev. 2005;9(1):25-39. doi:10.1016/j.smrv.2004.05.002
3. Tallavajhula S, Rodgers JJ, Slater JD. Sleep and sleep-wake disorders. In: Arciniengas DB, Yudofsky SC, Hales RE, eds. Textbook of Neuropsychiatry and Clinical Neurosciences. American Psychiatric Association Publishing; 2018:373-393.
4. Sletten TL, Vincenzi S, Redman JR, et al. Timing of sleep and its relationship with the endogenous melatonin rhythm. Front Neurol. 2010;1:137. doi:10.3389/fneur.2010.00137
1. Joseph SG. Melatonin supplementation for the prevention of hospital-associated delirium. Ment Health Clin. 2018;7(4):143-146. doi:10.9740/mhc.2017.07.143
2. Arendt J, Skene DJ. Melatonin as a chronobiotic. Sleep Med Rev. 2005;9(1):25-39. doi:10.1016/j.smrv.2004.05.002
3. Tallavajhula S, Rodgers JJ, Slater JD. Sleep and sleep-wake disorders. In: Arciniengas DB, Yudofsky SC, Hales RE, eds. Textbook of Neuropsychiatry and Clinical Neurosciences. American Psychiatric Association Publishing; 2018:373-393.
4. Sletten TL, Vincenzi S, Redman JR, et al. Timing of sleep and its relationship with the endogenous melatonin rhythm. Front Neurol. 2010;1:137. doi:10.3389/fneur.2010.00137
Proposal for a new diagnosis: Acute anxiety disorder
Editor’s note: Readers’ Forum is a department for correspondence from readers that is not in response to articles published in
Mr. F, age 42, says he has always been a very anxious person and has chronically found his worrying to negatively affect his life. He says that over the last month his anxiety has been “off the charts” and he is worrying “24/7” due to taking on new responsibilities at his job and his son being diagnosed with lupus. He says his constant worrying is significantly impairing his ability to focus at his job, and he is considering taking a mental health leave from work. His wife reports that she is extremely frustrated because Mr. F has been isolating himself from family and friends; he admits this is true and attributes it to being preoccupied by his worries.
Mr. F endorses chronic insomnia, muscle tension, and irritability associated with anxiety; these have all substantially worsened over the last month. He admits that recently he has occasionally thought it would be easier if he weren’t alive. Mr. F denies having problems with his energy or motivation levels and insists that he generally feels very anxious, but not depressed. He says he drinks 1 alcoholic drink per week and denies any other substance use. Mr. F is overweight and has slightly elevated cholesterol but denies any other health conditions. He takes melatonin to help him sleep but does not take any prescribed medications.
Although this vignette provides limited details, on the surface it appears that Mr. F is experiencing an exacerbation of chronic generalized anxiety disorder (GAD). However, in this article, I propose establishing a new diagnosis: “acute anxiety disorder,” which would encapsulate severe exacerbations of a pre-existing anxiety disorder. Among the patients I have encountered for whom this diagnosis would fit, most have pre-existing GAD or panic disorder.
A look at the differential diagnosis
It is important to determine whether Mr. F is using any substances or has a medical condition that could be contributing to his anxiety. Other psychiatric diagnoses that could be considered include:
Adjustment disorder. This diagnosis would make sense if Mr. F didn’t have an apparent chronic history of symptoms that meet criteria for GAD.
Major depressive disorder with anxious distress. Many patients experiencing a major depressive episode meet the criteria for the specifier “with anxious distress,” even those who do not have a comorbid anxiety disorder.1 However, it is not evident from this vignette that Mr. F is experiencing a major depressive episode.
Continue to: Panic disorder and GAD...
Panic disorder and GAD. It is possible for a patient with GAD to develop panic disorder, which, at times, occurs after experiencing significant life stressors. Panic disorder requires the presence of recurrent panic attacks. Mr. F describes experiencing chronic, intense symptoms of anxiety rather than the discreet episodes of acute symptoms that characterize panic attacks.
Acute stress disorder. This diagnosis involves psychological symptoms that occur in response to exposure to actual or threatened death, serious injury, or sexual violation. Mr. F was not exposed to any of these stressors.
Why this new diagnosis would be helpful
A new diagnosis, acute anxiety disorder, would indicate that a patient is currently experiencing an acute exacerbation of a chronic anxiety disorder that is leading to a significant decrease in their baseline functioning. My proposed criteria for acute anxiety disorder appear in the Table. Here are some reasons this diagnosis would be helpful:

Signifier of severity. Anxiety disorders such as GAD are generally not considered severe conditions and not considered to fall under the rubric of SPMI (severe and persistent mental illness).2 Posttraumatic stress disorder is the anxiety disorder–like condition most often found in the SPMI category. A diagnosis of acute anxiety disorder would indicate a patient is experiencing an episode of anxiety that is distinct from their chronic anxiety condition due to its severe impact on functional capabilities. Acute anxiety disorder would certainly not qualify as a “SPMI diagnosis” that would facilitate someone being considered eligible for supplemental security income, but it might be a legitimate justification for someone to receive short-term disability.
Treatment approach. The pharmacologic treatment of anxiety disorders usually involves a selective serotonin reuptake inhibitor (SSRI) or serotonin-norepinephrine reuptake inhibitor (SNRI). However, these medications can sometimes briefly increase anxiety when they are started. Individuals with acute anxiety are the most vulnerable to the possibility of experiencing increased anxiety when starting an SSRI or SNRI and may benefit from a slower titration of these medications. In light of this and the length of time required for SSRIs or SNRIs to exert a positive effect (typically a few weeks), patients with acute anxiety are best served by treatment with a medication with an immediate onset of action, such as a benzodiazepine or a sleep medication (eg, zolpidem). Benzodiazepines and hypnotics such as zolpidem are best prescribed for as-needed use because they carry a risk of dependence. One might consider prescribing mirtazapine or pregabalin (both of which are used off-label to treat anxiety) because these medications also have a relatively rapid onset of action and can treat both anxiety and insomnia (particularly mirtazapine).
Research considerations. It would be helpful to study which treatments are most effective for the subset of patients who experience acute anxiety disorder as I define it. Perhaps psychotherapy treatment protocols could be adapted or created. Treatment with esketamine or IV ketamine might be further studied as a treatment for acute anxiety because some evidence suggests ketamine is efficacious for this indication.3
1. Otsubo T, Hokama C, Sano N, et al. How significant is the assessment of the DSM-5 ‘anxious distress’ specifier in patients with major depressive disorder without comorbid anxiety disorders in the continuation/maintenance phase? Int J Psychiatry Clin Pract. 2021;25(4):385-392. doi:10.1080/13651501.2021.1907415
2. Butler H, O’Brien AJ. Access to specialist palliative care services by people with severe and persistent mental illness: a retrospective cohort study. Int J Ment Health Nurs. 2018;27(2):737-746. doi:10.1111/inm.12360
3. Glue P, Neehoff SM, Medlicott NJ, et al. Safety and efficacy of maintenance ketamine treatment in patients with treatment-refractory generalised anxiety and social anxiety disorders. J Psychopharmacol. 2018;32(6):663-667. doi:10.1177/0269881118762073
Editor’s note: Readers’ Forum is a department for correspondence from readers that is not in response to articles published in
Mr. F, age 42, says he has always been a very anxious person and has chronically found his worrying to negatively affect his life. He says that over the last month his anxiety has been “off the charts” and he is worrying “24/7” due to taking on new responsibilities at his job and his son being diagnosed with lupus. He says his constant worrying is significantly impairing his ability to focus at his job, and he is considering taking a mental health leave from work. His wife reports that she is extremely frustrated because Mr. F has been isolating himself from family and friends; he admits this is true and attributes it to being preoccupied by his worries.
Mr. F endorses chronic insomnia, muscle tension, and irritability associated with anxiety; these have all substantially worsened over the last month. He admits that recently he has occasionally thought it would be easier if he weren’t alive. Mr. F denies having problems with his energy or motivation levels and insists that he generally feels very anxious, but not depressed. He says he drinks 1 alcoholic drink per week and denies any other substance use. Mr. F is overweight and has slightly elevated cholesterol but denies any other health conditions. He takes melatonin to help him sleep but does not take any prescribed medications.
Although this vignette provides limited details, on the surface it appears that Mr. F is experiencing an exacerbation of chronic generalized anxiety disorder (GAD). However, in this article, I propose establishing a new diagnosis: “acute anxiety disorder,” which would encapsulate severe exacerbations of a pre-existing anxiety disorder. Among the patients I have encountered for whom this diagnosis would fit, most have pre-existing GAD or panic disorder.
A look at the differential diagnosis
It is important to determine whether Mr. F is using any substances or has a medical condition that could be contributing to his anxiety. Other psychiatric diagnoses that could be considered include:
Adjustment disorder. This diagnosis would make sense if Mr. F didn’t have an apparent chronic history of symptoms that meet criteria for GAD.
Major depressive disorder with anxious distress. Many patients experiencing a major depressive episode meet the criteria for the specifier “with anxious distress,” even those who do not have a comorbid anxiety disorder.1 However, it is not evident from this vignette that Mr. F is experiencing a major depressive episode.
Continue to: Panic disorder and GAD...
Panic disorder and GAD. It is possible for a patient with GAD to develop panic disorder, which, at times, occurs after experiencing significant life stressors. Panic disorder requires the presence of recurrent panic attacks. Mr. F describes experiencing chronic, intense symptoms of anxiety rather than the discreet episodes of acute symptoms that characterize panic attacks.
Acute stress disorder. This diagnosis involves psychological symptoms that occur in response to exposure to actual or threatened death, serious injury, or sexual violation. Mr. F was not exposed to any of these stressors.
Why this new diagnosis would be helpful
A new diagnosis, acute anxiety disorder, would indicate that a patient is currently experiencing an acute exacerbation of a chronic anxiety disorder that is leading to a significant decrease in their baseline functioning. My proposed criteria for acute anxiety disorder appear in the Table. Here are some reasons this diagnosis would be helpful:

Signifier of severity. Anxiety disorders such as GAD are generally not considered severe conditions and not considered to fall under the rubric of SPMI (severe and persistent mental illness).2 Posttraumatic stress disorder is the anxiety disorder–like condition most often found in the SPMI category. A diagnosis of acute anxiety disorder would indicate a patient is experiencing an episode of anxiety that is distinct from their chronic anxiety condition due to its severe impact on functional capabilities. Acute anxiety disorder would certainly not qualify as a “SPMI diagnosis” that would facilitate someone being considered eligible for supplemental security income, but it might be a legitimate justification for someone to receive short-term disability.
Treatment approach. The pharmacologic treatment of anxiety disorders usually involves a selective serotonin reuptake inhibitor (SSRI) or serotonin-norepinephrine reuptake inhibitor (SNRI). However, these medications can sometimes briefly increase anxiety when they are started. Individuals with acute anxiety are the most vulnerable to the possibility of experiencing increased anxiety when starting an SSRI or SNRI and may benefit from a slower titration of these medications. In light of this and the length of time required for SSRIs or SNRIs to exert a positive effect (typically a few weeks), patients with acute anxiety are best served by treatment with a medication with an immediate onset of action, such as a benzodiazepine or a sleep medication (eg, zolpidem). Benzodiazepines and hypnotics such as zolpidem are best prescribed for as-needed use because they carry a risk of dependence. One might consider prescribing mirtazapine or pregabalin (both of which are used off-label to treat anxiety) because these medications also have a relatively rapid onset of action and can treat both anxiety and insomnia (particularly mirtazapine).
Research considerations. It would be helpful to study which treatments are most effective for the subset of patients who experience acute anxiety disorder as I define it. Perhaps psychotherapy treatment protocols could be adapted or created. Treatment with esketamine or IV ketamine might be further studied as a treatment for acute anxiety because some evidence suggests ketamine is efficacious for this indication.3
Editor’s note: Readers’ Forum is a department for correspondence from readers that is not in response to articles published in
Mr. F, age 42, says he has always been a very anxious person and has chronically found his worrying to negatively affect his life. He says that over the last month his anxiety has been “off the charts” and he is worrying “24/7” due to taking on new responsibilities at his job and his son being diagnosed with lupus. He says his constant worrying is significantly impairing his ability to focus at his job, and he is considering taking a mental health leave from work. His wife reports that she is extremely frustrated because Mr. F has been isolating himself from family and friends; he admits this is true and attributes it to being preoccupied by his worries.
Mr. F endorses chronic insomnia, muscle tension, and irritability associated with anxiety; these have all substantially worsened over the last month. He admits that recently he has occasionally thought it would be easier if he weren’t alive. Mr. F denies having problems with his energy or motivation levels and insists that he generally feels very anxious, but not depressed. He says he drinks 1 alcoholic drink per week and denies any other substance use. Mr. F is overweight and has slightly elevated cholesterol but denies any other health conditions. He takes melatonin to help him sleep but does not take any prescribed medications.
Although this vignette provides limited details, on the surface it appears that Mr. F is experiencing an exacerbation of chronic generalized anxiety disorder (GAD). However, in this article, I propose establishing a new diagnosis: “acute anxiety disorder,” which would encapsulate severe exacerbations of a pre-existing anxiety disorder. Among the patients I have encountered for whom this diagnosis would fit, most have pre-existing GAD or panic disorder.
A look at the differential diagnosis
It is important to determine whether Mr. F is using any substances or has a medical condition that could be contributing to his anxiety. Other psychiatric diagnoses that could be considered include:
Adjustment disorder. This diagnosis would make sense if Mr. F didn’t have an apparent chronic history of symptoms that meet criteria for GAD.
Major depressive disorder with anxious distress. Many patients experiencing a major depressive episode meet the criteria for the specifier “with anxious distress,” even those who do not have a comorbid anxiety disorder.1 However, it is not evident from this vignette that Mr. F is experiencing a major depressive episode.
Continue to: Panic disorder and GAD...
Panic disorder and GAD. It is possible for a patient with GAD to develop panic disorder, which, at times, occurs after experiencing significant life stressors. Panic disorder requires the presence of recurrent panic attacks. Mr. F describes experiencing chronic, intense symptoms of anxiety rather than the discreet episodes of acute symptoms that characterize panic attacks.
Acute stress disorder. This diagnosis involves psychological symptoms that occur in response to exposure to actual or threatened death, serious injury, or sexual violation. Mr. F was not exposed to any of these stressors.
Why this new diagnosis would be helpful
A new diagnosis, acute anxiety disorder, would indicate that a patient is currently experiencing an acute exacerbation of a chronic anxiety disorder that is leading to a significant decrease in their baseline functioning. My proposed criteria for acute anxiety disorder appear in the Table. Here are some reasons this diagnosis would be helpful:

Signifier of severity. Anxiety disorders such as GAD are generally not considered severe conditions and not considered to fall under the rubric of SPMI (severe and persistent mental illness).2 Posttraumatic stress disorder is the anxiety disorder–like condition most often found in the SPMI category. A diagnosis of acute anxiety disorder would indicate a patient is experiencing an episode of anxiety that is distinct from their chronic anxiety condition due to its severe impact on functional capabilities. Acute anxiety disorder would certainly not qualify as a “SPMI diagnosis” that would facilitate someone being considered eligible for supplemental security income, but it might be a legitimate justification for someone to receive short-term disability.
Treatment approach. The pharmacologic treatment of anxiety disorders usually involves a selective serotonin reuptake inhibitor (SSRI) or serotonin-norepinephrine reuptake inhibitor (SNRI). However, these medications can sometimes briefly increase anxiety when they are started. Individuals with acute anxiety are the most vulnerable to the possibility of experiencing increased anxiety when starting an SSRI or SNRI and may benefit from a slower titration of these medications. In light of this and the length of time required for SSRIs or SNRIs to exert a positive effect (typically a few weeks), patients with acute anxiety are best served by treatment with a medication with an immediate onset of action, such as a benzodiazepine or a sleep medication (eg, zolpidem). Benzodiazepines and hypnotics such as zolpidem are best prescribed for as-needed use because they carry a risk of dependence. One might consider prescribing mirtazapine or pregabalin (both of which are used off-label to treat anxiety) because these medications also have a relatively rapid onset of action and can treat both anxiety and insomnia (particularly mirtazapine).
Research considerations. It would be helpful to study which treatments are most effective for the subset of patients who experience acute anxiety disorder as I define it. Perhaps psychotherapy treatment protocols could be adapted or created. Treatment with esketamine or IV ketamine might be further studied as a treatment for acute anxiety because some evidence suggests ketamine is efficacious for this indication.3
1. Otsubo T, Hokama C, Sano N, et al. How significant is the assessment of the DSM-5 ‘anxious distress’ specifier in patients with major depressive disorder without comorbid anxiety disorders in the continuation/maintenance phase? Int J Psychiatry Clin Pract. 2021;25(4):385-392. doi:10.1080/13651501.2021.1907415
2. Butler H, O’Brien AJ. Access to specialist palliative care services by people with severe and persistent mental illness: a retrospective cohort study. Int J Ment Health Nurs. 2018;27(2):737-746. doi:10.1111/inm.12360
3. Glue P, Neehoff SM, Medlicott NJ, et al. Safety and efficacy of maintenance ketamine treatment in patients with treatment-refractory generalised anxiety and social anxiety disorders. J Psychopharmacol. 2018;32(6):663-667. doi:10.1177/0269881118762073
1. Otsubo T, Hokama C, Sano N, et al. How significant is the assessment of the DSM-5 ‘anxious distress’ specifier in patients with major depressive disorder without comorbid anxiety disorders in the continuation/maintenance phase? Int J Psychiatry Clin Pract. 2021;25(4):385-392. doi:10.1080/13651501.2021.1907415
2. Butler H, O’Brien AJ. Access to specialist palliative care services by people with severe and persistent mental illness: a retrospective cohort study. Int J Ment Health Nurs. 2018;27(2):737-746. doi:10.1111/inm.12360
3. Glue P, Neehoff SM, Medlicott NJ, et al. Safety and efficacy of maintenance ketamine treatment in patients with treatment-refractory generalised anxiety and social anxiety disorders. J Psychopharmacol. 2018;32(6):663-667. doi:10.1177/0269881118762073
Neurosurgical treatment of OCD: Patient selection, safety, and access
Obsessive-compulsive disorder (OCD) is typically a severe, chronic illness in which patients have recurrent, unwanted thoughts, urges, and compulsions.1 It causes significant morbidity and lost potential over time, and is the world’s 10th-most disabling disorder in terms of lost income and decreased quality of life, and the fifth-most disabling mental health condition.2 Patients with OCD (and their clinicians) are often desperate for an efficacious treatment, but we must ensure that those who are not helped by traditional psychotherapeutic and/or pharmacologic treatments are appropriate for safe neurosurgical intervention.
Pros and cons of neurosurgical therapies
Most patients with OCD are effectively treated with cognitive-behavioral therapy and pharmacotherapy in the form of selective serotonin reuptake inhibitors, clomipramine, or second-generation antipsychotics. However, up to 5% of individuals with OCD will have symptoms refractory to these traditional therapies.3 These cases require more aggressive forms of therapy, including radiofrequency ablation surgeries and deep brain stimulation (DBS). The efficacy of both therapies is similar at 40% to 60%.4,5 While these treatments can be life-changing for patients fortunate to receive them, they are not without issue.
Only a limited number of institutions offer these neurosurgical techniques, and for many patients, those locations may be inaccessible. Patients may not experience relief simply due to where they live, difficult logistics, and the high cost requisite to receive care. If fortunate enough to live near a participating institution or have the means to travel to one, the patient and clinician must then choose the best option based on the nuances of the patient’s situation.
Ablation techniques, such as gamma knife or magnetic resonance–guided ultrasound, are simpler and more cost-effective. A drawback of this approach, however, is that it is irreversible. Lesioned structures are irreparable, as are the adverse effects of the surgery, which, while rare, may include a persistent minimally conscious state or necrotic cysts.4 A benefit of this approach is that there is no need for lengthy follow-up as seen with DBS.
DBS is more complicated. In addition to having to undergo an open neurosurgical procedure, these patients require long-term follow-up and monitoring. A positive aspect is the device can be turned off or removed. However, the amount of follow-up and adjustments is significant. These patients need access to clinicians skilled in DBS device management.
Finally, we must consider the chronically ill patient’s perspective after successful treatment. While the patient’s symptoms may improve, their lives and identities likely developed around their symptoms. Bosanac et al6 describe this reality well in a case study in which a patient with OCD was “burdened with normality” after successful DBS treatment. He was finally able to work, build meaningful relationships, and approach previously unattainable social milestones. This was an overwhelming experience for him, and he and his family needed guidance into the world in which most of us find comfort.
As ablation techniques, DBS, and other cutting-edge therapies for OCD come to the forefront of modern care, clinicians must remember to keep patient safety first. Verify follow-up care before committing patients to invasive and irreversible treatments. While general access is currently poor, participating institutions should consider advertising and communicating that there is an accessible network available for these chronically ill individuals.
1. Ruscio AM, Stein DJ, Chiu WT, et al. The epidemiology of obsessive-compulsive disorder in the National Comorbidity Survey Replication. Mol Psychiatry. 2010;15(1):53-63.
2. World Health Organization. The Global Burden of Disease: 2004 Update. World Health Organization; 2008.
3. Jenike MA, Rauch SL. Managing the patient with treatment-resistant obsessive compulsive disorder: current strategies. J Clin Psychiatry. 1994;55 Suppl:11-17.
4. Rasmussen SA, Noren G, Greenberg BD, et al. Gamma ventral capsulotomy in intractable obsessive-compulsive disorder. Biol Psychiatry. 2018;84(5):355-364.
5. Kumar KK, Appelboom, G, Lamsam L, et al. Comparative effectiveness of neuroablation and deep brain stimulation for treatment-resistant obsessive-compulsive disorder: a meta-analytic study. J Neurol Neurosurg Psychiatry. 2019;90(4):469-473.
6. Bosanac P, Hamilton BE, Lucak J, et al. Identity challenges and ‘burden of normality’ after DBS for severe OCD: a narrative case study. BMC Psychiatry. 2018;18(1):186.
Obsessive-compulsive disorder (OCD) is typically a severe, chronic illness in which patients have recurrent, unwanted thoughts, urges, and compulsions.1 It causes significant morbidity and lost potential over time, and is the world’s 10th-most disabling disorder in terms of lost income and decreased quality of life, and the fifth-most disabling mental health condition.2 Patients with OCD (and their clinicians) are often desperate for an efficacious treatment, but we must ensure that those who are not helped by traditional psychotherapeutic and/or pharmacologic treatments are appropriate for safe neurosurgical intervention.
Pros and cons of neurosurgical therapies
Most patients with OCD are effectively treated with cognitive-behavioral therapy and pharmacotherapy in the form of selective serotonin reuptake inhibitors, clomipramine, or second-generation antipsychotics. However, up to 5% of individuals with OCD will have symptoms refractory to these traditional therapies.3 These cases require more aggressive forms of therapy, including radiofrequency ablation surgeries and deep brain stimulation (DBS). The efficacy of both therapies is similar at 40% to 60%.4,5 While these treatments can be life-changing for patients fortunate to receive them, they are not without issue.
Only a limited number of institutions offer these neurosurgical techniques, and for many patients, those locations may be inaccessible. Patients may not experience relief simply due to where they live, difficult logistics, and the high cost requisite to receive care. If fortunate enough to live near a participating institution or have the means to travel to one, the patient and clinician must then choose the best option based on the nuances of the patient’s situation.
Ablation techniques, such as gamma knife or magnetic resonance–guided ultrasound, are simpler and more cost-effective. A drawback of this approach, however, is that it is irreversible. Lesioned structures are irreparable, as are the adverse effects of the surgery, which, while rare, may include a persistent minimally conscious state or necrotic cysts.4 A benefit of this approach is that there is no need for lengthy follow-up as seen with DBS.
DBS is more complicated. In addition to having to undergo an open neurosurgical procedure, these patients require long-term follow-up and monitoring. A positive aspect is the device can be turned off or removed. However, the amount of follow-up and adjustments is significant. These patients need access to clinicians skilled in DBS device management.
Finally, we must consider the chronically ill patient’s perspective after successful treatment. While the patient’s symptoms may improve, their lives and identities likely developed around their symptoms. Bosanac et al6 describe this reality well in a case study in which a patient with OCD was “burdened with normality” after successful DBS treatment. He was finally able to work, build meaningful relationships, and approach previously unattainable social milestones. This was an overwhelming experience for him, and he and his family needed guidance into the world in which most of us find comfort.
As ablation techniques, DBS, and other cutting-edge therapies for OCD come to the forefront of modern care, clinicians must remember to keep patient safety first. Verify follow-up care before committing patients to invasive and irreversible treatments. While general access is currently poor, participating institutions should consider advertising and communicating that there is an accessible network available for these chronically ill individuals.
Obsessive-compulsive disorder (OCD) is typically a severe, chronic illness in which patients have recurrent, unwanted thoughts, urges, and compulsions.1 It causes significant morbidity and lost potential over time, and is the world’s 10th-most disabling disorder in terms of lost income and decreased quality of life, and the fifth-most disabling mental health condition.2 Patients with OCD (and their clinicians) are often desperate for an efficacious treatment, but we must ensure that those who are not helped by traditional psychotherapeutic and/or pharmacologic treatments are appropriate for safe neurosurgical intervention.
Pros and cons of neurosurgical therapies
Most patients with OCD are effectively treated with cognitive-behavioral therapy and pharmacotherapy in the form of selective serotonin reuptake inhibitors, clomipramine, or second-generation antipsychotics. However, up to 5% of individuals with OCD will have symptoms refractory to these traditional therapies.3 These cases require more aggressive forms of therapy, including radiofrequency ablation surgeries and deep brain stimulation (DBS). The efficacy of both therapies is similar at 40% to 60%.4,5 While these treatments can be life-changing for patients fortunate to receive them, they are not without issue.
Only a limited number of institutions offer these neurosurgical techniques, and for many patients, those locations may be inaccessible. Patients may not experience relief simply due to where they live, difficult logistics, and the high cost requisite to receive care. If fortunate enough to live near a participating institution or have the means to travel to one, the patient and clinician must then choose the best option based on the nuances of the patient’s situation.
Ablation techniques, such as gamma knife or magnetic resonance–guided ultrasound, are simpler and more cost-effective. A drawback of this approach, however, is that it is irreversible. Lesioned structures are irreparable, as are the adverse effects of the surgery, which, while rare, may include a persistent minimally conscious state or necrotic cysts.4 A benefit of this approach is that there is no need for lengthy follow-up as seen with DBS.
DBS is more complicated. In addition to having to undergo an open neurosurgical procedure, these patients require long-term follow-up and monitoring. A positive aspect is the device can be turned off or removed. However, the amount of follow-up and adjustments is significant. These patients need access to clinicians skilled in DBS device management.
Finally, we must consider the chronically ill patient’s perspective after successful treatment. While the patient’s symptoms may improve, their lives and identities likely developed around their symptoms. Bosanac et al6 describe this reality well in a case study in which a patient with OCD was “burdened with normality” after successful DBS treatment. He was finally able to work, build meaningful relationships, and approach previously unattainable social milestones. This was an overwhelming experience for him, and he and his family needed guidance into the world in which most of us find comfort.
As ablation techniques, DBS, and other cutting-edge therapies for OCD come to the forefront of modern care, clinicians must remember to keep patient safety first. Verify follow-up care before committing patients to invasive and irreversible treatments. While general access is currently poor, participating institutions should consider advertising and communicating that there is an accessible network available for these chronically ill individuals.
1. Ruscio AM, Stein DJ, Chiu WT, et al. The epidemiology of obsessive-compulsive disorder in the National Comorbidity Survey Replication. Mol Psychiatry. 2010;15(1):53-63.
2. World Health Organization. The Global Burden of Disease: 2004 Update. World Health Organization; 2008.
3. Jenike MA, Rauch SL. Managing the patient with treatment-resistant obsessive compulsive disorder: current strategies. J Clin Psychiatry. 1994;55 Suppl:11-17.
4. Rasmussen SA, Noren G, Greenberg BD, et al. Gamma ventral capsulotomy in intractable obsessive-compulsive disorder. Biol Psychiatry. 2018;84(5):355-364.
5. Kumar KK, Appelboom, G, Lamsam L, et al. Comparative effectiveness of neuroablation and deep brain stimulation for treatment-resistant obsessive-compulsive disorder: a meta-analytic study. J Neurol Neurosurg Psychiatry. 2019;90(4):469-473.
6. Bosanac P, Hamilton BE, Lucak J, et al. Identity challenges and ‘burden of normality’ after DBS for severe OCD: a narrative case study. BMC Psychiatry. 2018;18(1):186.
1. Ruscio AM, Stein DJ, Chiu WT, et al. The epidemiology of obsessive-compulsive disorder in the National Comorbidity Survey Replication. Mol Psychiatry. 2010;15(1):53-63.
2. World Health Organization. The Global Burden of Disease: 2004 Update. World Health Organization; 2008.
3. Jenike MA, Rauch SL. Managing the patient with treatment-resistant obsessive compulsive disorder: current strategies. J Clin Psychiatry. 1994;55 Suppl:11-17.
4. Rasmussen SA, Noren G, Greenberg BD, et al. Gamma ventral capsulotomy in intractable obsessive-compulsive disorder. Biol Psychiatry. 2018;84(5):355-364.
5. Kumar KK, Appelboom, G, Lamsam L, et al. Comparative effectiveness of neuroablation and deep brain stimulation for treatment-resistant obsessive-compulsive disorder: a meta-analytic study. J Neurol Neurosurg Psychiatry. 2019;90(4):469-473.
6. Bosanac P, Hamilton BE, Lucak J, et al. Identity challenges and ‘burden of normality’ after DBS for severe OCD: a narrative case study. BMC Psychiatry. 2018;18(1):186.
‘Med check’ appointments: How to minimize your malpractice risk
Medical malpractice claims can arise in any type of health care setting. The purpose of this article is to discuss the risk of medical malpractice suits in the context of brief “med checks,” which are 15- to 20-minute follow-up appointments for psychiatric outpatient medication management. Similar issues arise in brief new patient and transfer visits.
Malpractice hinges on ‘reasonableness’
Malpractice is an allegation of professional negligence.1 More specifically, it is an allegation that a clinician violated an existing duty by deviating from the standard of care, and that deviation caused damages.2 Medical malpractice claims involve questions about whether there was a deviation from the standard of care (whether the clinician failed to exercise a reasonable degree of skill and care given the context of the situation) and whether there was causation (whether a deviation caused a patient’s damages).3 These are fact-based determinations. Thus, the legal resolution of a malpractice claim is based on the facts of each specific case.
The advisability of 15-minute med checks and the associated limitation on a clinician’s ability to provide talk therapy are beyond the scope of this article. What is clear, however, is that not all brief med check appointments are created equal. Their safety and efficacy are dictated by the milieu in which they exist.
Practically speaking, although many factors need to be considered, the standard of care in a medical malpractice lawsuit is based on reasonableness.4-6 One strategy to proactively manage your malpractice risk is to consider—either for your existing job or before accepting a new position—whether your agency’s setup for brief med checks will allow you to practice reasonably. This article provides information to help you answer this question and describes a hypothetical case vignette to illustrate how certain factors might help lower the chances of facing a malpractice suit.
Established patients
In med check appointments for established patients, consider the patient population, the availability of pre- and postvisit support services, and contingency plans (Table).

Different patient populations require different levels of treatment. Consider, for example, a patient with anxiety and trauma who is actively engaged with a therapist who works at the same agency as their psychiatrist, where the medication management appointments are solely for selective serotonin reuptake inhibitor refills. Compare that to a dual-diagnosis patient—with a psychotic and substance use disorder—who has had poor medication compliance and frequent rehospitalizations. The first patient is more likely to be reasonably managed in a 15-minute med check. The second patient would need significantly more pre- and postvisit support services. This consideration is relevant from a clinical perspective, and if a bad outcome occurs, from a malpractice perspective. Patient populations are not homogeneous; the reasonableness of a clinician’s actions during a brief med check visit depends on the specific patient.
Pre- and postvisit support services vary greatly from clinic to clinic. They range from clerical support (eg, calling a pharmacy to ensure that a patient’s medication is available for same-day pickup) to nursing support (eg, an injection nurse who is on site and can immediately provide a patient with a missed injection) to case manager support (eg, a case manager to facilitate coordination of care, such as by having a patient fill out record releases and then working to ensure that relevant hospital records are received prior to the next visit). The real-world availability of these services can determine the feasibility of safely conducting a 15-minute med check visit.
Continue to: Regardless of the patient population...
Regardless of the patient population, unexpected situations will arise. It could be a patient with posttraumatic stress disorder who was recently retraumatized and is in the midst of disclosing this new trauma at the end of a 15-minute visit. Or it could be a patient with dual diagnoses who comes to the agency intoxicated and manic, describing a plan to kill his neighbor with a shotgun. A clinician’s ability to meet the standard of care, and act reasonably within the confines of a brief med check structure, can thus depend on whether there are means of adequately managing such emergent situations.
Some clinics have fairly high no-show rates. Leaving no-show slots open for administrative time can provide a means of managing emergent situations. If, however, they are automatically rebooked with walk-ins, brief visits become more challenging. Thus, when assessing contingency plan logistics, consider the no-show rate, what happens when there are no-shows, how many other clinicians are available on a given day, and whether staff is available to provide support (eg, sitting with a patient while waiting for an ambulance).
New and transfer patients
Brief visits for new or transfer patients require the same assessment described above. However, there are additional considerations regarding previsit support services. Some clinics use clinical social workers to perform intake evaluations before a new patient sees the psychiatrist. A high-quality intake evaluation can allow a psychiatrist to focus, in a shorter amount of time, on a patient’s medication needs. An additional time saver is having support staff who will obtain relevant medical records before a patient’s first psychiatric visit. Such actions can greatly increase the efficacy of a new patient appointment for the prescribing clinician.
The reliability of and level of detail assessed in prior evaluations can be particularly relevant when considering a job providing coverage as locum tenens, when all patients will be new to you. Unfortunately, if you are not employed at a clinic, it can be hard to assess this ahead of time. If you know colleagues in the area where you are considering taking a locum position, ask for their opinions about the quality of work at the agency.
Case vignette
Mr. J is a 30-year-old man with schizoaffective disorder. For several years, he has been followed once every 4 weeks at the local clinic. During the first year of treatment, he had numerous hospitalizations due to medication noncompliance, psychotic episodes, and threats of violence against his mother. For the past year, he had been stable on the same dose of an oral antipsychotic medication (risperidone 2 mg twice a day). Then he stopped taking his medication, became increasingly psychotic, and, while holding a butcher knife, threatened to kill his mother. His mother called 911 and Mr. J was hospitalized.
Continue to: While in the hospital...
While in the hospital, Mr. J was restarted on risperidone 2 mg twice a day, and lithium 600 mg twice a day was added. As part of discharge planning, the hospital social worker set up an outpatient appointment with Dr. R, Mr. J’s treating psychiatrist at the clinic. That appointment was scheduled as a 15-minute med check. At the visit, Dr. R did not have or try to obtain a copy of the hospital discharge summary. Mr. J told Dr. R that he had been hospitalized because he had run out of his oral antipsychotic, and that it had been restarted during the hospitalization. Dr. R—who did not know about the recent incident involving a butcher knife or the subsequent medication changes—continued Mr. J’s risperidone, but did not continue his lithium because she did not know it had been added.
Dr. R scheduled a 4-week follow-up visit for Mr. J. Then she went on maternity leave. Because the agency was short-staffed, they hired Dr. C—a locum tenens—to see all of Dr. R’s established patients in 15-minute time slots.
At their first visit, Mr. J told Dr. C that he was gaining too much weight from his antipsychotic and wanted to know if it would be OK to decrease the dose. Dr. C reviewed Dr. R’s last office note but, due to limited time, did not review any other notes. Although Dr. C had 2 no-shows that day, the clinic had a policy that required Dr. C to see walk-ins whenever there was a no-show.
Dr. C did not know of Mr. J’s threats of violence or the medication changes associated with his recent hospitalization (they were not referenced in Dr. R’s last note). Dr. C decreased the dose of Mr. J’s risperidone from 2 mg twice a day to 0.5 mg twice a day. He did not do a violence risk assessment. Two weeks after the visit with Dr. C, Mr. J, who had become increasingly depressed and psychotic, killed his mother and died by suicide.
The estates of Mr. J and his mother filed a medical malpractice lawsuit against Dr. R and Dr. C. Both psychiatrists had a duty to Mr. J. Whether there was a duty to Mr. J’s mother would depend in part on the state’s duty to protect laws. Either way, the malpractice case would hinge on whether the psychiatrists’ conduct fell below the standard of care.
Continue to: In this case...
In this case, the critical issues were Dr. R’s failure to obtain and review the recent hospital records and Dr. C’s decision to decrease the antipsychotic dose. Of particular concern is Dr. C’s decision to decrease the antipsychotic dose without reviewing more information from past records, and the resultant failure to perform a violence risk assessment. These deviations cannot be blamed entirely on the brevity of the med check appointment. They could happen in a clinic that allotted longer time periods for follow-up visits, but they are, however, more likely to occur in a short med check appointment due to time constraints.
The likelihood of these errors could have been reduced by additional support services, as well as more time for Dr. C to see each patient who was new to him. For example, if there had been a support person available to obtain hospital records prior to the postdischarge appointment, Dr. R and Dr. C would have been more likely to be aware of the violent threat associated with Mr. J’s hospitalization. Additionally, if the busy clinicians had contingency plans to assess complicated patients, such as the ability to use no-show time to deal with difficult situations, Dr. C could have taken more time to review past records.
Bottom Line
When working in a setting that involves brief med check appointments, assess the agency structure, and whether it will allow you to practice reasonably. This will be relevant clinically and may reduce the risk of malpractice lawsuits. Reasonableness of a clinician’s actions is a fact-specific question and is influenced by multiple factors, including the patient population, the availability and quality of an agency’s support services, and contingency plans.
Related Resources
- Mossman D. Successfully navigating the 15-minute ‘med check.’ Current Psychiatry. 2010;9(6):40-43.
- Olfson M, Marcus SC. National trends in outpatient psychotherapy. Am J Psychiatry. 2010;167(12):1456-1463.
Drug Brand Names
Lithium • Eskalith, Lithobid
Risperidone • Risperdal
1. Malpractice. In: Garner BA, ed. Black’s Law Dictionary. 11th ed. Thomson West; 2019:1148.
2. Frierson RL, Joshi KG. Malpractice law and psychiatry: an overview. Focus. 2019;17:332-336. doi:10.1176/appi.focus.20190017
3. Negligence Based Claims. In: Boumil MM, Hattis PA, eds. Medical Liability in a Nutshell. 4th ed. West Academic Publishing; 2017:43-88
4. Peters PG. The quiet demise of deference to custom: malpractice law at the millennium. Washington and Lee Law Review. 2000;57(1):163-205. Accessed July 8, 2022. https://scholarlycommons.law.wlu.edu/wlulr/vol57/iss1/5
5. Simon RI. Standard-of-care testimony: best practices or reasonable care? J Am Acad Psychiatry Law. 2005;33(1):8-11. Accessed July 8, 2022. http://jaapl.org/content/33/1/8
6. Behrens SA. Call in Houdini: the time has come to be released from the geographic straightjacket known as the locality rule. Drake Law Review. 2008; 56(3):753-790. Accessed June 20, 2022. https://lawreviewdrake.files.wordpress.com/2015/06/lrvol56-3_behrens.pdf
Medical malpractice claims can arise in any type of health care setting. The purpose of this article is to discuss the risk of medical malpractice suits in the context of brief “med checks,” which are 15- to 20-minute follow-up appointments for psychiatric outpatient medication management. Similar issues arise in brief new patient and transfer visits.
Malpractice hinges on ‘reasonableness’
Malpractice is an allegation of professional negligence.1 More specifically, it is an allegation that a clinician violated an existing duty by deviating from the standard of care, and that deviation caused damages.2 Medical malpractice claims involve questions about whether there was a deviation from the standard of care (whether the clinician failed to exercise a reasonable degree of skill and care given the context of the situation) and whether there was causation (whether a deviation caused a patient’s damages).3 These are fact-based determinations. Thus, the legal resolution of a malpractice claim is based on the facts of each specific case.
The advisability of 15-minute med checks and the associated limitation on a clinician’s ability to provide talk therapy are beyond the scope of this article. What is clear, however, is that not all brief med check appointments are created equal. Their safety and efficacy are dictated by the milieu in which they exist.
Practically speaking, although many factors need to be considered, the standard of care in a medical malpractice lawsuit is based on reasonableness.4-6 One strategy to proactively manage your malpractice risk is to consider—either for your existing job or before accepting a new position—whether your agency’s setup for brief med checks will allow you to practice reasonably. This article provides information to help you answer this question and describes a hypothetical case vignette to illustrate how certain factors might help lower the chances of facing a malpractice suit.
Established patients
In med check appointments for established patients, consider the patient population, the availability of pre- and postvisit support services, and contingency plans (Table).

Different patient populations require different levels of treatment. Consider, for example, a patient with anxiety and trauma who is actively engaged with a therapist who works at the same agency as their psychiatrist, where the medication management appointments are solely for selective serotonin reuptake inhibitor refills. Compare that to a dual-diagnosis patient—with a psychotic and substance use disorder—who has had poor medication compliance and frequent rehospitalizations. The first patient is more likely to be reasonably managed in a 15-minute med check. The second patient would need significantly more pre- and postvisit support services. This consideration is relevant from a clinical perspective, and if a bad outcome occurs, from a malpractice perspective. Patient populations are not homogeneous; the reasonableness of a clinician’s actions during a brief med check visit depends on the specific patient.
Pre- and postvisit support services vary greatly from clinic to clinic. They range from clerical support (eg, calling a pharmacy to ensure that a patient’s medication is available for same-day pickup) to nursing support (eg, an injection nurse who is on site and can immediately provide a patient with a missed injection) to case manager support (eg, a case manager to facilitate coordination of care, such as by having a patient fill out record releases and then working to ensure that relevant hospital records are received prior to the next visit). The real-world availability of these services can determine the feasibility of safely conducting a 15-minute med check visit.
Continue to: Regardless of the patient population...
Regardless of the patient population, unexpected situations will arise. It could be a patient with posttraumatic stress disorder who was recently retraumatized and is in the midst of disclosing this new trauma at the end of a 15-minute visit. Or it could be a patient with dual diagnoses who comes to the agency intoxicated and manic, describing a plan to kill his neighbor with a shotgun. A clinician’s ability to meet the standard of care, and act reasonably within the confines of a brief med check structure, can thus depend on whether there are means of adequately managing such emergent situations.
Some clinics have fairly high no-show rates. Leaving no-show slots open for administrative time can provide a means of managing emergent situations. If, however, they are automatically rebooked with walk-ins, brief visits become more challenging. Thus, when assessing contingency plan logistics, consider the no-show rate, what happens when there are no-shows, how many other clinicians are available on a given day, and whether staff is available to provide support (eg, sitting with a patient while waiting for an ambulance).
New and transfer patients
Brief visits for new or transfer patients require the same assessment described above. However, there are additional considerations regarding previsit support services. Some clinics use clinical social workers to perform intake evaluations before a new patient sees the psychiatrist. A high-quality intake evaluation can allow a psychiatrist to focus, in a shorter amount of time, on a patient’s medication needs. An additional time saver is having support staff who will obtain relevant medical records before a patient’s first psychiatric visit. Such actions can greatly increase the efficacy of a new patient appointment for the prescribing clinician.
The reliability of and level of detail assessed in prior evaluations can be particularly relevant when considering a job providing coverage as locum tenens, when all patients will be new to you. Unfortunately, if you are not employed at a clinic, it can be hard to assess this ahead of time. If you know colleagues in the area where you are considering taking a locum position, ask for their opinions about the quality of work at the agency.
Case vignette
Mr. J is a 30-year-old man with schizoaffective disorder. For several years, he has been followed once every 4 weeks at the local clinic. During the first year of treatment, he had numerous hospitalizations due to medication noncompliance, psychotic episodes, and threats of violence against his mother. For the past year, he had been stable on the same dose of an oral antipsychotic medication (risperidone 2 mg twice a day). Then he stopped taking his medication, became increasingly psychotic, and, while holding a butcher knife, threatened to kill his mother. His mother called 911 and Mr. J was hospitalized.
Continue to: While in the hospital...
While in the hospital, Mr. J was restarted on risperidone 2 mg twice a day, and lithium 600 mg twice a day was added. As part of discharge planning, the hospital social worker set up an outpatient appointment with Dr. R, Mr. J’s treating psychiatrist at the clinic. That appointment was scheduled as a 15-minute med check. At the visit, Dr. R did not have or try to obtain a copy of the hospital discharge summary. Mr. J told Dr. R that he had been hospitalized because he had run out of his oral antipsychotic, and that it had been restarted during the hospitalization. Dr. R—who did not know about the recent incident involving a butcher knife or the subsequent medication changes—continued Mr. J’s risperidone, but did not continue his lithium because she did not know it had been added.
Dr. R scheduled a 4-week follow-up visit for Mr. J. Then she went on maternity leave. Because the agency was short-staffed, they hired Dr. C—a locum tenens—to see all of Dr. R’s established patients in 15-minute time slots.
At their first visit, Mr. J told Dr. C that he was gaining too much weight from his antipsychotic and wanted to know if it would be OK to decrease the dose. Dr. C reviewed Dr. R’s last office note but, due to limited time, did not review any other notes. Although Dr. C had 2 no-shows that day, the clinic had a policy that required Dr. C to see walk-ins whenever there was a no-show.
Dr. C did not know of Mr. J’s threats of violence or the medication changes associated with his recent hospitalization (they were not referenced in Dr. R’s last note). Dr. C decreased the dose of Mr. J’s risperidone from 2 mg twice a day to 0.5 mg twice a day. He did not do a violence risk assessment. Two weeks after the visit with Dr. C, Mr. J, who had become increasingly depressed and psychotic, killed his mother and died by suicide.
The estates of Mr. J and his mother filed a medical malpractice lawsuit against Dr. R and Dr. C. Both psychiatrists had a duty to Mr. J. Whether there was a duty to Mr. J’s mother would depend in part on the state’s duty to protect laws. Either way, the malpractice case would hinge on whether the psychiatrists’ conduct fell below the standard of care.
Continue to: In this case...
In this case, the critical issues were Dr. R’s failure to obtain and review the recent hospital records and Dr. C’s decision to decrease the antipsychotic dose. Of particular concern is Dr. C’s decision to decrease the antipsychotic dose without reviewing more information from past records, and the resultant failure to perform a violence risk assessment. These deviations cannot be blamed entirely on the brevity of the med check appointment. They could happen in a clinic that allotted longer time periods for follow-up visits, but they are, however, more likely to occur in a short med check appointment due to time constraints.
The likelihood of these errors could have been reduced by additional support services, as well as more time for Dr. C to see each patient who was new to him. For example, if there had been a support person available to obtain hospital records prior to the postdischarge appointment, Dr. R and Dr. C would have been more likely to be aware of the violent threat associated with Mr. J’s hospitalization. Additionally, if the busy clinicians had contingency plans to assess complicated patients, such as the ability to use no-show time to deal with difficult situations, Dr. C could have taken more time to review past records.
Bottom Line
When working in a setting that involves brief med check appointments, assess the agency structure, and whether it will allow you to practice reasonably. This will be relevant clinically and may reduce the risk of malpractice lawsuits. Reasonableness of a clinician’s actions is a fact-specific question and is influenced by multiple factors, including the patient population, the availability and quality of an agency’s support services, and contingency plans.
Related Resources
- Mossman D. Successfully navigating the 15-minute ‘med check.’ Current Psychiatry. 2010;9(6):40-43.
- Olfson M, Marcus SC. National trends in outpatient psychotherapy. Am J Psychiatry. 2010;167(12):1456-1463.
Drug Brand Names
Lithium • Eskalith, Lithobid
Risperidone • Risperdal
Medical malpractice claims can arise in any type of health care setting. The purpose of this article is to discuss the risk of medical malpractice suits in the context of brief “med checks,” which are 15- to 20-minute follow-up appointments for psychiatric outpatient medication management. Similar issues arise in brief new patient and transfer visits.
Malpractice hinges on ‘reasonableness’
Malpractice is an allegation of professional negligence.1 More specifically, it is an allegation that a clinician violated an existing duty by deviating from the standard of care, and that deviation caused damages.2 Medical malpractice claims involve questions about whether there was a deviation from the standard of care (whether the clinician failed to exercise a reasonable degree of skill and care given the context of the situation) and whether there was causation (whether a deviation caused a patient’s damages).3 These are fact-based determinations. Thus, the legal resolution of a malpractice claim is based on the facts of each specific case.
The advisability of 15-minute med checks and the associated limitation on a clinician’s ability to provide talk therapy are beyond the scope of this article. What is clear, however, is that not all brief med check appointments are created equal. Their safety and efficacy are dictated by the milieu in which they exist.
Practically speaking, although many factors need to be considered, the standard of care in a medical malpractice lawsuit is based on reasonableness.4-6 One strategy to proactively manage your malpractice risk is to consider—either for your existing job or before accepting a new position—whether your agency’s setup for brief med checks will allow you to practice reasonably. This article provides information to help you answer this question and describes a hypothetical case vignette to illustrate how certain factors might help lower the chances of facing a malpractice suit.
Established patients
In med check appointments for established patients, consider the patient population, the availability of pre- and postvisit support services, and contingency plans (Table).

Different patient populations require different levels of treatment. Consider, for example, a patient with anxiety and trauma who is actively engaged with a therapist who works at the same agency as their psychiatrist, where the medication management appointments are solely for selective serotonin reuptake inhibitor refills. Compare that to a dual-diagnosis patient—with a psychotic and substance use disorder—who has had poor medication compliance and frequent rehospitalizations. The first patient is more likely to be reasonably managed in a 15-minute med check. The second patient would need significantly more pre- and postvisit support services. This consideration is relevant from a clinical perspective, and if a bad outcome occurs, from a malpractice perspective. Patient populations are not homogeneous; the reasonableness of a clinician’s actions during a brief med check visit depends on the specific patient.
Pre- and postvisit support services vary greatly from clinic to clinic. They range from clerical support (eg, calling a pharmacy to ensure that a patient’s medication is available for same-day pickup) to nursing support (eg, an injection nurse who is on site and can immediately provide a patient with a missed injection) to case manager support (eg, a case manager to facilitate coordination of care, such as by having a patient fill out record releases and then working to ensure that relevant hospital records are received prior to the next visit). The real-world availability of these services can determine the feasibility of safely conducting a 15-minute med check visit.
Continue to: Regardless of the patient population...
Regardless of the patient population, unexpected situations will arise. It could be a patient with posttraumatic stress disorder who was recently retraumatized and is in the midst of disclosing this new trauma at the end of a 15-minute visit. Or it could be a patient with dual diagnoses who comes to the agency intoxicated and manic, describing a plan to kill his neighbor with a shotgun. A clinician’s ability to meet the standard of care, and act reasonably within the confines of a brief med check structure, can thus depend on whether there are means of adequately managing such emergent situations.
Some clinics have fairly high no-show rates. Leaving no-show slots open for administrative time can provide a means of managing emergent situations. If, however, they are automatically rebooked with walk-ins, brief visits become more challenging. Thus, when assessing contingency plan logistics, consider the no-show rate, what happens when there are no-shows, how many other clinicians are available on a given day, and whether staff is available to provide support (eg, sitting with a patient while waiting for an ambulance).
New and transfer patients
Brief visits for new or transfer patients require the same assessment described above. However, there are additional considerations regarding previsit support services. Some clinics use clinical social workers to perform intake evaluations before a new patient sees the psychiatrist. A high-quality intake evaluation can allow a psychiatrist to focus, in a shorter amount of time, on a patient’s medication needs. An additional time saver is having support staff who will obtain relevant medical records before a patient’s first psychiatric visit. Such actions can greatly increase the efficacy of a new patient appointment for the prescribing clinician.
The reliability of and level of detail assessed in prior evaluations can be particularly relevant when considering a job providing coverage as locum tenens, when all patients will be new to you. Unfortunately, if you are not employed at a clinic, it can be hard to assess this ahead of time. If you know colleagues in the area where you are considering taking a locum position, ask for their opinions about the quality of work at the agency.
Case vignette
Mr. J is a 30-year-old man with schizoaffective disorder. For several years, he has been followed once every 4 weeks at the local clinic. During the first year of treatment, he had numerous hospitalizations due to medication noncompliance, psychotic episodes, and threats of violence against his mother. For the past year, he had been stable on the same dose of an oral antipsychotic medication (risperidone 2 mg twice a day). Then he stopped taking his medication, became increasingly psychotic, and, while holding a butcher knife, threatened to kill his mother. His mother called 911 and Mr. J was hospitalized.
Continue to: While in the hospital...
While in the hospital, Mr. J was restarted on risperidone 2 mg twice a day, and lithium 600 mg twice a day was added. As part of discharge planning, the hospital social worker set up an outpatient appointment with Dr. R, Mr. J’s treating psychiatrist at the clinic. That appointment was scheduled as a 15-minute med check. At the visit, Dr. R did not have or try to obtain a copy of the hospital discharge summary. Mr. J told Dr. R that he had been hospitalized because he had run out of his oral antipsychotic, and that it had been restarted during the hospitalization. Dr. R—who did not know about the recent incident involving a butcher knife or the subsequent medication changes—continued Mr. J’s risperidone, but did not continue his lithium because she did not know it had been added.
Dr. R scheduled a 4-week follow-up visit for Mr. J. Then she went on maternity leave. Because the agency was short-staffed, they hired Dr. C—a locum tenens—to see all of Dr. R’s established patients in 15-minute time slots.
At their first visit, Mr. J told Dr. C that he was gaining too much weight from his antipsychotic and wanted to know if it would be OK to decrease the dose. Dr. C reviewed Dr. R’s last office note but, due to limited time, did not review any other notes. Although Dr. C had 2 no-shows that day, the clinic had a policy that required Dr. C to see walk-ins whenever there was a no-show.
Dr. C did not know of Mr. J’s threats of violence or the medication changes associated with his recent hospitalization (they were not referenced in Dr. R’s last note). Dr. C decreased the dose of Mr. J’s risperidone from 2 mg twice a day to 0.5 mg twice a day. He did not do a violence risk assessment. Two weeks after the visit with Dr. C, Mr. J, who had become increasingly depressed and psychotic, killed his mother and died by suicide.
The estates of Mr. J and his mother filed a medical malpractice lawsuit against Dr. R and Dr. C. Both psychiatrists had a duty to Mr. J. Whether there was a duty to Mr. J’s mother would depend in part on the state’s duty to protect laws. Either way, the malpractice case would hinge on whether the psychiatrists’ conduct fell below the standard of care.
Continue to: In this case...
In this case, the critical issues were Dr. R’s failure to obtain and review the recent hospital records and Dr. C’s decision to decrease the antipsychotic dose. Of particular concern is Dr. C’s decision to decrease the antipsychotic dose without reviewing more information from past records, and the resultant failure to perform a violence risk assessment. These deviations cannot be blamed entirely on the brevity of the med check appointment. They could happen in a clinic that allotted longer time periods for follow-up visits, but they are, however, more likely to occur in a short med check appointment due to time constraints.
The likelihood of these errors could have been reduced by additional support services, as well as more time for Dr. C to see each patient who was new to him. For example, if there had been a support person available to obtain hospital records prior to the postdischarge appointment, Dr. R and Dr. C would have been more likely to be aware of the violent threat associated with Mr. J’s hospitalization. Additionally, if the busy clinicians had contingency plans to assess complicated patients, such as the ability to use no-show time to deal with difficult situations, Dr. C could have taken more time to review past records.
Bottom Line
When working in a setting that involves brief med check appointments, assess the agency structure, and whether it will allow you to practice reasonably. This will be relevant clinically and may reduce the risk of malpractice lawsuits. Reasonableness of a clinician’s actions is a fact-specific question and is influenced by multiple factors, including the patient population, the availability and quality of an agency’s support services, and contingency plans.
Related Resources
- Mossman D. Successfully navigating the 15-minute ‘med check.’ Current Psychiatry. 2010;9(6):40-43.
- Olfson M, Marcus SC. National trends in outpatient psychotherapy. Am J Psychiatry. 2010;167(12):1456-1463.
Drug Brand Names
Lithium • Eskalith, Lithobid
Risperidone • Risperdal
1. Malpractice. In: Garner BA, ed. Black’s Law Dictionary. 11th ed. Thomson West; 2019:1148.
2. Frierson RL, Joshi KG. Malpractice law and psychiatry: an overview. Focus. 2019;17:332-336. doi:10.1176/appi.focus.20190017
3. Negligence Based Claims. In: Boumil MM, Hattis PA, eds. Medical Liability in a Nutshell. 4th ed. West Academic Publishing; 2017:43-88
4. Peters PG. The quiet demise of deference to custom: malpractice law at the millennium. Washington and Lee Law Review. 2000;57(1):163-205. Accessed July 8, 2022. https://scholarlycommons.law.wlu.edu/wlulr/vol57/iss1/5
5. Simon RI. Standard-of-care testimony: best practices or reasonable care? J Am Acad Psychiatry Law. 2005;33(1):8-11. Accessed July 8, 2022. http://jaapl.org/content/33/1/8
6. Behrens SA. Call in Houdini: the time has come to be released from the geographic straightjacket known as the locality rule. Drake Law Review. 2008; 56(3):753-790. Accessed June 20, 2022. https://lawreviewdrake.files.wordpress.com/2015/06/lrvol56-3_behrens.pdf
1. Malpractice. In: Garner BA, ed. Black’s Law Dictionary. 11th ed. Thomson West; 2019:1148.
2. Frierson RL, Joshi KG. Malpractice law and psychiatry: an overview. Focus. 2019;17:332-336. doi:10.1176/appi.focus.20190017
3. Negligence Based Claims. In: Boumil MM, Hattis PA, eds. Medical Liability in a Nutshell. 4th ed. West Academic Publishing; 2017:43-88
4. Peters PG. The quiet demise of deference to custom: malpractice law at the millennium. Washington and Lee Law Review. 2000;57(1):163-205. Accessed July 8, 2022. https://scholarlycommons.law.wlu.edu/wlulr/vol57/iss1/5
5. Simon RI. Standard-of-care testimony: best practices or reasonable care? J Am Acad Psychiatry Law. 2005;33(1):8-11. Accessed July 8, 2022. http://jaapl.org/content/33/1/8
6. Behrens SA. Call in Houdini: the time has come to be released from the geographic straightjacket known as the locality rule. Drake Law Review. 2008; 56(3):753-790. Accessed June 20, 2022. https://lawreviewdrake.files.wordpress.com/2015/06/lrvol56-3_behrens.pdf
Risk factors for nonsuicidal self-injury: A review of the evidence
Nonsuicidal self-injury (NSSI) is the direct and deliberate destruction of body tissue without intent to die.1 Common forms of NSSI include cutting, burning, scraping/scratching skin, biting, hitting, and interfering with wound healing.2 Functional theories suggest that NSSI temporarily alleviates overwhelming negative emotions and can produce feelings of relief, resulting in a reinforcing effect.3
NSSI has been shown to be a risk factor for future suicide attempts.4 A 2018 study found that NSSI is associated with an increased risk of subsequent suicidal ideation (odds ratio [OR] 2.8), suicide plan (OR 3.0), and suicide attempt (OR 5.5).5 NSSI is also associated with individuals who had suicidal ideation and formed a suicide plan, and individuals who had a suicide plan and attempted suicide (ORs 1.7 to 2.1).5 Another study found that 70% of adolescents who engage in NSSI have attempted suicide during their lifetime, and 55% have multiple attempts.6
Given the overlap between suicide attempts and NSSI, performing a thorough suicide risk assessment (which is beyond the scope of this article) is crucial. This article describes the static and dynamic risk factors for NSSI in adolescents and adults, which can help us perform a suicide risk assessment and allow us to formulate an appropriate treatment plan that includes safety-based interventions.

NSSI risk factors for adolescents
From developing sexual identity and undergoing puberty to achieving increased independence from their parents and developing a sense of autonomy, adolescents undergo many biological, psychological, and social changes before reaching adulthood.7 Data suggest that NSSI often begins in adolescence, with a typical onset at age 13 or 14.3 Community studies show that one-third to one-half of adolescents in the United States have engaged in NSSI.8,9 Previously, NSSI during adolescence was associated with 3 major diagnostic categories: eating disorders, developmental disabilities, and borderline personality disorder (BPD).10 However, recent data suggest that NSSI is also common outside of these categories. Here we describe static and dynamic risk factors for NSSI in adolescents (Table 111-42). Table 211-42 summarizes the studies of NSSI in adolescents that we reviewed.
Static risk factors
Female adolescents and adults engage in NSSI at higher rates than males. The difference is larger in clinical populations compared to the general population.11
A large portion of research about NSSI has been conducted in studies in which the majority of participants were White.12 Most studies report a higher prevalence of NSSI among non-Hispanic White youth,13 but some suggest other ethnic groups may also experience high rates of self-harm and NSSI.13-15 Several studies have demonstrated high rates of self-harm among South Asian adult females compared with White adult females, but this difference may be less pronounced in adolescents.14 One study in the United Kingdom found that White females age 10 to 14 had higher rates of self-harm compared to South Asian females,14 while another found that risk and rates of self-harm in young South Asian people varied by city and country of origin.15 Young Black females15 and young Black males13 also may be at an increased risk of self-harm. One review found that Black females were more likely to self-harm than Asian or White groups.15
Several studies suggest that sexual minority adolescents (SMA) (eg, lesbian, gay, bisexual, transgender, queer) are at greater risk for NSSI than heterosexual adolescents.16 SMA have been shown to engage in a significantly greater frequency of NSSI and more types of NSSI than heterosexual adolescents.16 Furthermore, on the Inventory of Statements about Self-Injury, SMA self-reported using NSSI for intrapersonal functions (eg, for affect regulation, antisuicide, self-punishment) significantly greater than their heterosexual peers; however, there were no significant differences between the 2 groups on interpersonal functions (eg, autonomy, interpersonal boundaries, peer bonding, sensation-seeking).16
Continue to: Transgender and gender nonconfirming...
Transgender and gender nonconfirming (GNC) youth are at a particularly high risk for NSSI; 30% to 45.5% of transgender adolescents report self-injury.17 Factors shown to distinguish transgender/GNC youth who engage in NSSI from those who do not include having a mental health problem, depression, running away from home, substance use, lower self-esteem/greater self-criticism, experiencing transphobia victimization, and having more interpersonal problems.18,19 Among transgender/GNC youth, those whose biological sex is female are more likely to report NSSI than those whose biological sex is male (ie, transgendered adolescent males are more likely to report NSSI than transgendered adolescent females).18,19
Most forms of childhood maltreatment have been associated with NSSI. In a recently published review, Liu et al20 found that childhood maltreatment (including sexual abuse, physical abuse, emotional abuse, and physical neglect) was associated with an increased risk for NSSI. However, conflicting evidence suggests that when confounders are removed, only childhood emotional abuse was directly associated with NSSI.21 Current evidence is modest for childhood emotional neglect as a risk factor for NSSI.20
Increasing research is investigating the biological processes that may be implicated in NSSI. Some studies suggest that endogenous opioids,22 monoamine neurotransmitters,22 and the hypothalamic-pituitary-adrenal (HPA) axis23 may play a role in NSSI. Compared to healthy controls, adolescents engaging in NSSI have been shown to have lower pain intensity (P = .036), higher pain thresholds (P = .040), and lower beta-endorphins (endogenous opioid hormones involved in mediating stress and pain) (P = .002).24 There may be alterations in the HPA axis among adolescents who engage in NSSI, more specifically stronger cortisol awakening responses.23 Both functional and standard MRI have been used to study the neurobiology of NSSI. One study demonstrated differences in functional connectivity between brain areas linked to neuroregulation of emotions in adolescents who engage in NSSI,25 while another found volume reduction in the insula of these adolescents, which suggests a possible neurobiological reason for impulsivity and the increased risk of suicidal behavior.26
Dynamic risk factors
Research has repeatedly shown bullying is a risk factor for NSSI.27 One study found that younger children who were victimized reported significantly more NSSI than older children.28 New data suggest that perpetrators of bullying are also at risk for deliberate self-harm behavior (SHB), which this study defined as a behavior that is intended to cause self-harm but without suicidal intent and having a nonfatal outcome.29 Victims of cyberbullying also are at a greater risk for self-harm, suicidal behaviors, and suicide attempt.30 To a lesser extent, cyberbullying perpetrators are at greater risk for suicidal behaviors and suicidal ideation.30 Bullying is a risk factor for NSSI not only in adolescence, but also in adulthood. Lereya et al31 found that victims of bullying in childhood and early adolescence were more likely to have mental health problems (including anxiety and depression) and more likely to engage in SHB—which this study defined as hurting oneself on purpose in any way—as adults.
The effects of internet use on adolescents’ mental health also has been investigated. A recent review that explored the relationship between all types of internet use (general use, internet addiction, social media, self-harm websites, forums, etc) and SHB/suicidal behavior found that young people with internet addiction, high levels of internet use, and a tendency to view websites with self-harm or suicidal content were at higher risk of engaging in SHB/suicidal behavior.32 This study did not use a specific definition for SHB or suicidal behavior.32
Continue to: Membership in certain youth...
Membership in certain youth subcultures (eg, emo or goth) has been evaluated as potential risk factors for depression and deliberate self-harm. Bowes et al33 found that for each unit increase in goth affiliation (not at all, not very much, somewhat, more than somewhat, very much), youth were 1.52 times more likely to engage in SHB; these researchers also reported a dose-response association between goth identification and future SHB. This study asked participants if they have ever tried to harm or hurt themselves in any manner, but did not distinguish between individuals who had harmed themselves with and without suicidal intent.33
Personality traits such as impulsiveness and loneliness have been linked to NSSI among adolescents.34,35 A recent study found that adolescents who met the proposed DSM-5 diagnostic criteria for NSSI scored higher on the Barratt Impulsiveness Scale, specifically in measures of:
- motor impulsiveness (ie, acting without thinking)
- attentional impulsiveness (ie, making decisions quickly)
- impulsiveness due to lack of planning (ie, failure to plan for the future).34
This study also found that adolescents who identified as being lonely based on scores on the Brazilian Loneliness Scale were at a higher risk for NSSI.34
A recent systematic review (32 studies) and meta-analysis (9 studies) found that school absenteeism was associated with a risk of self-harm (pooled aOR 1.37, P = .01) and suicidal ideation (pooled aOR 1.20, P = .03).36 This study suggested that school absenteeism, an important marker of social exclusion, was associated with both SHB and suicidal ideation in young people.36 It defined SHB as any act of self-injury or self-poisoning, regardless of intent.36
Finally, family-related factors have been associated with an increased risk of NSSI. One study of 11,814 children age 9 and 10 revealed that high family conflict (OR 1.09; 95% CI, 1.05 to 1.14) and low parental monitoring (OR 0.95; 95% CI, 0.93 to 0.98) were associated with NSSI.37 A smaller, community-based study found that adolescents with NSSI reported significantly less maternal support and warmth than nonclinical controls, but a cause-and-effect relationship has not yet been determined.38 Parental history alone may influence adolescents’ risk of NSSI. A study that included nearly 76,000 youth found that adolescents with perceived parental alcohol problems had higher odds of self-injury, suicidal ideation, and suicide attempts.39 Adolescents exposed to maternal or paternal adversities were also at a higher risk of self-harm (hazard ratio 1.5 to 5.4 among males, 1.7 to 3.9 among females).40
Continue to: NSSI risk factors for adults
NSSI risk factors for adults
Although data regarding the prevalence of NSSI in adults are lacking, available studies report a 12-month prevalence of 0.9%2 and a lifetime prevalence of 5.5% to 5.9%.43 There is a significant overlap in risk factors for NSSI in adolescent and adult populations, but there are also many important differences. The static and dynamic risk factors for NSSI in adults are described in Table 3.44-66 Table 444-66 summarizes the studies of NSSI in adults that we reviewed.
Static risk factors
Research findings regarding the prevalence of NSSI based on gender are varied. For years, it has been believed that women are more likely to engage in NSSI than men. Recent meta-analyses that have examined this relationship closely found that the gender difference is larger for clinical samples compared to community samples and more pronounced in younger individuals.11
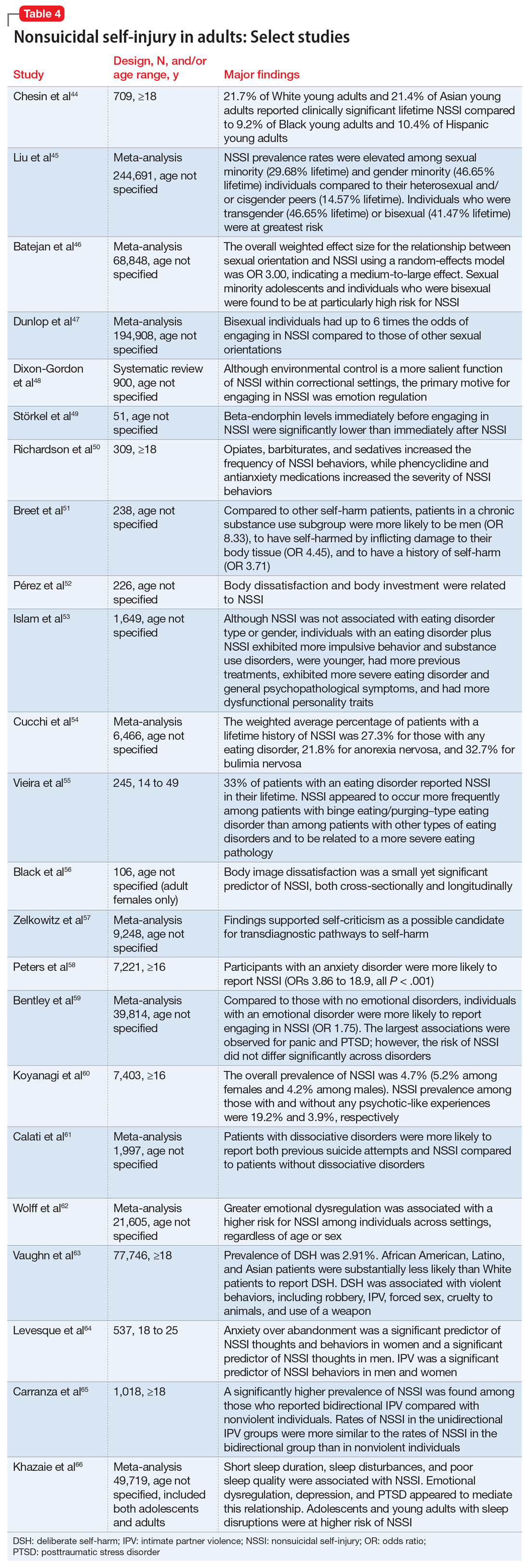
As is the case with adolescents, there may be ethnic variations in rates of self-harm and NSSI among adults. A 2013 study by Chesin et al44 found that Asian and White young adults experience higher rates of NSSI than their Hispanic and Black counterparts. Evidence suggests that relative rates of self-harm for older South Asian adults are lower than in older White adults.15
Compared to heterosexual or cisgender individuals, members of sexual and gender minorities have a higher past-year and lifetime prevalence of NSSI.45 One study found that the weighted effect size between sexual orientation and NSSI had an OR of 3 (95% CI, 2.46 to 3.66), indicating a medium-to-large effect.46 Bisexual and transgender individuals appear to be at the highest risk for NSSI when compared to members of other sexual and gender minority groups.45 One review that included mostly cross-sectional studies found that individuals identifying as bisexual had up to 6 times the odds of engaging in NSSI when compared to those of other sexual orientations.47
Incarceration is a risk factor for NSSI. The rates of NSSI in criminal justice settings are higher (up to 61%) than in the general adult population (approximately 4%).48 Recent research found that NSSI serves similar functions in correctional and non-correctional settings, primarily to regulate emotions.48 However, there is also evidence of higher rates of NSSI being motivated by an attempt to influence the environment (ie, engaging in NSSI in order to be transferred to another prison unit) compared to NSSI in community settings.48
Continue to: Though less robust than data...
Though less robust than data published regarding adolescents, the role of biological processes in adults engaging in NSSI has also been studied. A 2021 study by Störkel et al49 found that levels of salivary beta-endorphins were significantly lower in adults immediately before engaging in NSSI compared to after NSSI. Furthermore, adults who engage in NSSI have lower levels of met-enkephalin (P < .01), an opioid growth factor, compared to adults who have never engaged in NSSI.22
Dynamic risk factors
Individuals who engage in NSSI often report substance use, but there is little data on whether substance use is an independent risk factor for NSSI. Although limited, recent evidence suggests illicit substance use in both adolescents41 and adults50 increases risk for NSSI. Richardson et al50 found that the use of barbiturates, opiates, and sedatives significantly increased the frequency of NSSI, whereas use of marijuana, phencyclidine, and medications used to treat anxiety significantly increased the severity of NSSI. A smaller study conducted in South Africa found that individuals who engage in substance use and NSSI were more likely to be male (P < .001).51
Eating disorders and NSSI are highly comorbid.52 The lifetime prevalence of NSSI among individuals with eating disorders ranges from 20.6%to 37.1%.52,53 Results are inconsistent regarding which eating disorders (if any) are greater risk factors for NSSI. One study found that the prevalence of NSSI in patients with bulimia nervosa was 32.7% (95% CI, 26.9% to 39.1%) vs 21.8% in patients with anorexia nervosa (95% CI, 18.5% to 25.6%).54 Another study found that individuals with binge eating/purging–type eating disorders reported engaging in NSSI more frequently than those with other types of eating disorders.55 Among patients with eating disorders who reported NSSI, risk factors included younger age of onset, more negative self-evaluation, more impulsive behavior, concomitant substance use, history of suicide attempts, childhood abuse, and peer aggression.53,55 Body image dissatisfaction and self-criticism, even in individuals not formally diagnosed with an eating disorder, are small but significant predictors of NSSI.56,57
Mood disorders have also been linked to NSSI.58,59 Anxiety disorders (including generalized anxiety disorder, social phobia, panic disorder, and agoraphobia) as well as anxiety-related disorders such as obsessive-compulsive disorder have been significantly associated with NSSI (P < .001), but this relationship decreased in strength when mood instability was removed as a confounder.58 Among patients with anxiety and anxiety-related disorders, panic disorder and posttraumatic stress disorder (PTSD) have shown the strongest association with NSSI, with pooled aORs of 2.67 and 2.06, respectively.59
Recent studies have examined the association of other mental health disorders and symptoms with NSSI, including psychosis60 and dissociative symptoms.61 One study found that paranoia, thought control, and auditory hallucinations were significantly associated with NSSI60; however, after controlling for concomitant BPD, only paranoia was significantly associated with NSSI.60 Individuals diagnosed with dissociative disorders were more likely than patients without such disorders to endorse NSSI and suicide attempts.61
Continue to: Emotional dysregulation...
Emotional dysregulation (EDR)—defined as difficulty understanding, recognizing, and managing one’s emotions—has been researched extensively in relation to NSSI.62 A recent review that included studies of both adolescents and adults reported a significant association between EDR and NSSI, with an OR of 2.40 (95% CI, 2.01 to 2.86).62 A larger effect size was observed between EDR and lifetime NSSI (OR 3.21; 95% CI, 2.63 to 3.91) compared to past-year NSSI (OR 2.32; 95% CI, 1.84 to 2.92).62 Patient age, sex, and sample type (clinical vs community) were not significant moderators of strength between the reported associations.62
Studies examining intimate partner violence (IPV) and NSSI have found that young adults who engage in IPV (both as victims and as perpetrators) are more likely to report NSSI.63-65 Researchers have proposed that anxiety over abandonment may explain this relationship.64 A recent study found that individuals with bidirectional IPV (ie, both victimization and perpetration) engaged in NSSI at a higher prevalence than those engaging in unidirectional IPV or no IPV.65 This suggests that relationship violence in general (rather than just being a victim of IPV) may be a risk factor for NSSI.65
Finally, studies suggest that adolescents and adults who have sleep problems (insomnia, short sleep duration, long sleep onset latency, waking after sleep onset, and poor quality sleep) are more likely to report self-harm or NSSI than those without sleep problems.42,66 In adults, this relationship is partially mediated by depressive symptoms, EDR, and PTSD.66 In adolescents, depressive symptoms are a mediator for this relationship.42
Bottom Line
Nonsuicidal self-injury (NSSI) is a significant health concern due to its association with suicide attempts. Although there are similarities in NSSI risk factors between adolescents and adults, there are also important differences. Understanding these differences is necessary to develop appropriate treatment plans.
Related Resources
- American Foundation for Suicide Prevention. https://afsp.org/
- Cipriano A, Cella S, Cotrufo P. Nonsuicidal self-injury: a systematic review. Front Psych. 2017;8:1946. doi:10.3389/ fpsyg.2017.01946
- Gold LH, Frierson RL, eds. Textbook of Suicide Risk Assessment and Management. 3rd ed. American Psychiatric Association Publishing; 2020.
1. Nock MK. Self-injury. Annu Rev Clin Psychol. 2010;6:339-363.
2. Klonsky ED. Non-suicidal self-injury in United States adults: prevalence, sociodemographics, topography and functions. Psychol Med. 2011;41(9):1981-1986.
3. Klonsky ED. Nonsuicidal self-injury: what we know, and what we need to know. Can J Psychiatry. 2014;59(11):565-568.
4. Wilkinson P, Kelvin R, Roberts C, et al. Clinical and psychosocial predictors of suicide attempts and nonsuicidal self-injury in the Adolescent Depression Antidepressants and Psychotherapy Trial (ADAPT). Am J Psychiatry. 2011;168(5):495-501.
5. Kiekens G, Hasking P, Boyes M, et al. The associations between non-suicidal self-injury and first onset suicidal thoughts and behaviors. J Affect Disord. 2018;239:171-179.
6. Nock MK, Joiner TE, Gordon KH, et al. Non-suicidal self-injury among adolescents: diagnostic correlates and relation to suicide attempts. Psychiatry Res. 2006;144(1):65-72.
7. Christie D, Viner R. Adolescent development. BMJ. 2005;330(7486):301-304.
8. Yates TM, Tracy AJ, Luthar SS. Nonsuicidal self-injury among “privileged” youths: longitudinal and cross-sectional approaches to developmental process. J Consult Clin Psychol. 2008;76(1):52-62.
9. Lloyd-Richardson EE, Perrine N, Dierker L, et al. Characteristics and functions of non-suicidal self-injury in a community sample of adolescents. Psychol Med. 2007;37(8):1183-1192.
10. Peterson J, Freedenthal S, Sheldon C, et al. Nonsuicidal self injury in adolescents. Psychiatry(Edgmont). 2008;5(11):20-26.
11. Bresin K, Schoenleber M. Gender differences in the prevalence of nonsuicidal self-injury: a meta-analysis. Clin Psychol Rev. 2015;38:55-64.
12. Gholamrezaei M, Stefano JD, Heath NL. Nonsuicidal self-injury across cultures and ethnic and racial minorities: a review. Int J Psychol. 2015;52(4):316-326.
13. Rojas-Velasquez DA, Pluhar EI, Burns PA, et al. Nonsuicidal self-injury among African American and Hispanic adolescents and young adults: a systematic review. Prev Sci. 2021;22:367-377.
14. Bhui K, McKenzie K, Rasul F. Rates, risk factors & methods of self harm among minority ethnic groups in the UK: a systematic review. BMC Public Health. 2007;7:336.
15. Cooper J, Murphy E, Webb R, et al. Ethnic differences in self-harm, rates, characteristics and service provision: three-city cohort study. Br J Psychiatry. 2010;197(3):212-218.
16. Peters JR, Mereish EH, Krek MA, et al. Sexual orientation differences in non-suicidal self-injury, suicidality, and psychosocial factors among an inpatient psychiatric sample of adolescents. Psychiatry Res. 2020;284:112664.
17. Connolly MD, Zervos MJ, Barone 2nd CJ, et al. The mental health of transgender youth: advances in understanding. J Adolesc Health. 2016;59(5):489-495.
18. Taliaferro LA, McMorris BJ, Rider GN, et al. Risk and protective factors for self-harm in a population-based sample of transgender youth. Archives Suicide Res. 2019;23(2):203-221.
19. Arcelus J, Claes L, Witcomb GL, et al. Risk factors for non-suicidal self-injury among trans youth. J Sex Med. 2016;13(3):402-412.
20. Liu RT, Scopelliti KM, Pittman SK, et al. Childhood maltreatment and non-suicidal self-injury: a systematic review and meta-analysis. Lancet Psychiatry. 2018;5(1):51-64.
21. Thomassin K, Shaffer A, Madden A, et al. Specificity of childhood maltreatment and emotion deficit in nonsuicidal self-injury in an inpatient sample of youth. Psychiatry Res. 2016;244:103-108.
22. Stanley B, Sher L, Wilson S, et al. Non-suicidal self-injurious behavior, endogenous opioids and monoamine neurotransmitters. J Affect Disord. 2010;124(1-2):134-140.
23. Reichl C, Heyer A, Brunner R, et al. Hypothalamic-pituitary-adrenal axis, childhood adversity and adolescent nonsuicidal self-injury. Psychoneuroendocrinology. 2016;74:203-211.
24. van der Venne P, Balint A, Drews E, et al. Pain sensitivity and plasma beta-endorphin in adolescent non-suicidal self-injury. J Affect Disord. 2021;278:199-209.
25. Osuch E, Ford K, Wrath A, et al. Functional MRI of pain application in youth who engaged in repetitive non-suicidal self-injury vs. psychiatric controls. Psychiatry Res. 2014;223(2):104-112.
26. Ando A, Reichl C, Scheu F, et al. Regional grey matter volume reduction in adolescents engaging in non-suicidal self-injury. Psychiatry Res Neuroimaging. 2018;280:48-55.
27. Karanikola MNK, Lyberg A, Holm A-L, et al. The association between deliberate self-harm and school bullying victimization and the mediating effect of depressive symptoms and self-stigma: a systematic review. BioMed Res Int. 2018;4745791. doi: 10.1155/2018/4745791
28. van Geel M, Goemans A, Vedder P. A meta-analysis on the relation between peer victimization and adolescent non-suicidal self-injury. Psychiatry Res. 2015;230(2):364-368.
29. Heerde JA, Hemphill SA. Are bullying perpetration and victimization associated with adolescent deliberate self-harm? A meta-analysis. Arch Suicide Res. 2019;23(3):353-381.
30. John A, Glendenning AC, Marchant A, et al. Self-harm, suicidal behaviours, and cyberbullying in children and young people: systematic review. J Med Internet Res. 2018;20(4):e129. doi: 10.2196/jmir.9044
31. Lereya ST, Copeland WE, Costello EJ, et al. Adult mental health consequences of peer bullying and maltreatment in childhood: two cohorts in two countries. Lancet Psychiatry. 2015;2(6):524-531.
32. Marchant A, Hawton K, Stewart A, et al. A systematic review of the relationship between internet use, self-harm and suicidal behaviour in young people: the good, the bad and the unknown. PLoS One. 2017;12(8):e0181722. doi: 10.1371/journal.pone.0181722
33. Bowes L, Carnegie R, Pearson R, et al. Risk of depression and self-harm in teenagers identifying with goth subculture: a longitudinal cohort study. Lancet Psychiatry. 2015;2(9):793-800.
34. Costa RPO, Peixoto ALRP, Lucas CCA, et al. Profile of non-suicidal self-injury in adolescents: interface with impulsiveness and loneliness. J Pediatr (Rio J). 2021;97(2):184-190.
35. McHugh CM, Lee RSC, Hermens DF, et al. Impulsivity in the self-harm and suicidal behavior of young people: a systematic review and meta-analysis. J Psychiatr Res. 2019;116:51-60.
36. Epstein S, Roberts E, Sedgwick R, et al. School absenteeism as a risk factor for self-harm and suicidal ideation in children and adolescents: a systematic review and meta-analysis. Eur Child Adolesc Psychiatry. 2020;29(9):1175-1194.
37. DeVille DC, Whalen D, Breslin FJ, et al. Prevalence and family-related factors associated with suicidal ideation, suicide attempts, and self-injury in children aged 9 to 10 years. JAMA Netw Open. 2020;3(2):e1920956. doi: 10.1001/jamanetworkopen.2019.20956
38. Tschan T, Schmid M, In-Albon T. Parenting behavior in families of female adolescents with nonsuicidal self-injury in comparison to a clinical and a nonclinical control group. Child Adolesc Psychiatry Ment Health. 2015;9:17.
39. Pisinger V, Hawton K, Tolstrup JS. Self-injury and suicide behavior among young people with perceived parental alcohol problems in Denmark: a school-based survey. Eur Child Adolesc Psychiatry. 2018;27(2):201-208.
40. Pitkänen J, Remes H, Aaltonen M, et al. Experience of maternal and paternal adversities in childhood as determinants of self-harm in adolescence and young adulthood. J Epidemiol Community Health. 2019;73(11):1040-1046.
41. Monto MA, McRee N, Deryck FS. Nonsuicidal self-injury among a representative sample of US adolescents, 2015. Am J Public Health. 2018;108(8):1042-1048.
42. Hysing M, Sivertsen B, Stormark KM, et al. Sleep problems and self-harm in adolescence. Br J Psychiatry. 2015;207(4):306-312.
43. Swannell SV, Martin GE, Page A, et al. Prevalence of nonsuicidal self-injury in nonclinical samples: systematic review, meta-analysis and meta-regression. Suicide Life Threat Behav. 2014;44(3):273-303.
44. Chesin M, Moster A, Jeglic E. Non-suicidal self-injury among ethnically and racially diverse emerging adults: do factors unique to the minority experience matter? Current Psychology. 2013;32:318-328.
45. Liu RT, Sheehan AE, Walsh RFL, et al. Prevalence and correlates of non-suicidal self-injury among lesbian, gay, bisexual, and transgender individuals: a systematic review and meta-analysis. Clin Psychol Rev. 2019;74:101-783. doi:10.1016/j.cpr.2019.101783
46. Batejan KL, Jarvi SM, Swenson LP. Sexual orientation and non-suicidal self-injury: a meta-analytic review. Arch Suicide Res. 2015;19(2):131-150.
47. Dunlop BJ, Hartley S, Oladokun O, et al. Bisexuality and non-suicidal self-injury (NSSI): a narrative synthesis of associated variables and a meta-analysis of risk. J Affect Disord. 2020;276:1159-1172.
48. Dixon-Gordon K, Harrison N, Roesch R. Non-suicidal self-injury within offender populations: a systematic review. Int J Forensic Ment Health. 2012;11(1):33-50.
49. Störkel LM, Karabatsiakis A, Hepp K, et al. Salivary beta-endorphin in nonsuicidal self-injury: an ambulatory assessment study. Neuropsychopharmacology. 2021;46(7):1357-1363.
50. Richardson E, DePue MK, Therriault DJ, et al. The influence of substance use on engagement in non-suicidal self-injury (NSI) in adults. Subst Use Misuse. 2020;55(1):89-94.
51. Breet E, Bantjes J, Lewis I. Chronic substance use and self-harm in a primary health care setting. Afr J Prim Health Care Fam Med. 2018;10(1):e1-e9. doi: 10.4102/phcfm.v10i1.1544
52. Pérez S, Marco JH, Cañabate M. Non-suicidal self-injury in patients with eating disorders: prevalence, forms, functions, and body image correlates. Compr Psychiatry. 2018;84:32-38.
53. Islam MA, Steiger H, Jimenez-Murcia S, et al. Non-suicidal self-injury in different eating disorder types: relevance of personality traits and gender. Eur Eat Disord Rev. 2015;23(6):553-560.
54. Cucchi A, Ryan D, Konstantakopoulos G, et al. Lifetime prevalence of non-suicidal self-injury in patients with eating disorders: a systematic review and meta-analysis. Psychol Med. 2016;46(7):1345-1358.
55. Vieira AI, Machado BC, Machado PPP, et al. Putative risk factors for non-suicidal self-injury in eating disorders. Eur Eat Disord Rev. 2017;25(6):544-550.
56. Black EB, Garratt M, Beccaria G, et al. Body image as a predictor of nonsuicidal self-injury in women: a longitudinal study. Compr Psychiatry. 2019;88:83-89.
57. Zelkowitz RL, Cole DA. Self-criticism as a transdiagnostic process in nonsuicidal self-injury and disordered eating: systematic review and meta-analysis. Suicide Life Threat Behav. 2019;49(1):310-327.
58. Peters EM, Bowen R, Balbuena L. Mood instability contributes to impulsivity, non-suicidal self-injury, and binge eating/purging in people with anxiety disorders. Psychol Psychother. 2019;92(3):422-438.
59. Bentley KH, Cassiello-Robbins CF, Vittorio L, et al. The association between nonsuicidal self-injury and the emotional disorders: a meta-analytic review. Clin Psychol Rev. 2015;37:72-88.
60. Koyanagi A, Stickley A, Haro JM. Psychotic-like experiences and nonsuicidal self-injury in England: results from a national survey [corrected]. PLoS One. 2015;10(12):e0145533. doi: 10.1371/journal.pone.0145533
61. Calati R, Bensassi I, Courtet P. The link between dissociation and both suicide attempts and non-suicidal self-injury: meta-analyses. Psychiatry Res. 2017;251:103-114.
62. Wolff JC, Thompson E, Thomas SA, et al. Emotion dysregulation and non-suicidal self-injury: a systematic review and meta-analysis. Eur Psychiatry. 2019;59:25-36.
63. Vaughn MG, Salas-Wright CP, DeLisi M, et al. Deliberate self-harm and the nexus of violence, victimization, and mental health problems in the United States. Psychiatry Res. 2015;225(3):588-595.
64. Levesque C, Lafontaine M-F, Bureau J-F, et al. The influence of romantic attachment and intimate partner violence on nonsuicidal self-injury in young adults. J Youth Adolesc. 2010;39(5):474-483.
65. Carranza AB, Wallis CRD, Jonnson MR, et al. Nonsuicidal self-injury and intimate partner violence: directionality of violence and motives for self-injury. J Interpers Violence. 2020;886260520922372. doi: 10.1177/0886260520922372
66. Khazaie H, Zakiei A, McCall WV, et al. Relationship between sleep problems and self-injury: a systematic review. Behav Sleep Med. 2020;1-16. doi: 10.1080/15402002.2020.1822360
Nonsuicidal self-injury (NSSI) is the direct and deliberate destruction of body tissue without intent to die.1 Common forms of NSSI include cutting, burning, scraping/scratching skin, biting, hitting, and interfering with wound healing.2 Functional theories suggest that NSSI temporarily alleviates overwhelming negative emotions and can produce feelings of relief, resulting in a reinforcing effect.3
NSSI has been shown to be a risk factor for future suicide attempts.4 A 2018 study found that NSSI is associated with an increased risk of subsequent suicidal ideation (odds ratio [OR] 2.8), suicide plan (OR 3.0), and suicide attempt (OR 5.5).5 NSSI is also associated with individuals who had suicidal ideation and formed a suicide plan, and individuals who had a suicide plan and attempted suicide (ORs 1.7 to 2.1).5 Another study found that 70% of adolescents who engage in NSSI have attempted suicide during their lifetime, and 55% have multiple attempts.6
Given the overlap between suicide attempts and NSSI, performing a thorough suicide risk assessment (which is beyond the scope of this article) is crucial. This article describes the static and dynamic risk factors for NSSI in adolescents and adults, which can help us perform a suicide risk assessment and allow us to formulate an appropriate treatment plan that includes safety-based interventions.

NSSI risk factors for adolescents
From developing sexual identity and undergoing puberty to achieving increased independence from their parents and developing a sense of autonomy, adolescents undergo many biological, psychological, and social changes before reaching adulthood.7 Data suggest that NSSI often begins in adolescence, with a typical onset at age 13 or 14.3 Community studies show that one-third to one-half of adolescents in the United States have engaged in NSSI.8,9 Previously, NSSI during adolescence was associated with 3 major diagnostic categories: eating disorders, developmental disabilities, and borderline personality disorder (BPD).10 However, recent data suggest that NSSI is also common outside of these categories. Here we describe static and dynamic risk factors for NSSI in adolescents (Table 111-42). Table 211-42 summarizes the studies of NSSI in adolescents that we reviewed.

Static risk factors
Female adolescents and adults engage in NSSI at higher rates than males. The difference is larger in clinical populations compared to the general population.11
A large portion of research about NSSI has been conducted in studies in which the majority of participants were White.12 Most studies report a higher prevalence of NSSI among non-Hispanic White youth,13 but some suggest other ethnic groups may also experience high rates of self-harm and NSSI.13-15 Several studies have demonstrated high rates of self-harm among South Asian adult females compared with White adult females, but this difference may be less pronounced in adolescents.14 One study in the United Kingdom found that White females age 10 to 14 had higher rates of self-harm compared to South Asian females,14 while another found that risk and rates of self-harm in young South Asian people varied by city and country of origin.15 Young Black females15 and young Black males13 also may be at an increased risk of self-harm. One review found that Black females were more likely to self-harm than Asian or White groups.15
Several studies suggest that sexual minority adolescents (SMA) (eg, lesbian, gay, bisexual, transgender, queer) are at greater risk for NSSI than heterosexual adolescents.16 SMA have been shown to engage in a significantly greater frequency of NSSI and more types of NSSI than heterosexual adolescents.16 Furthermore, on the Inventory of Statements about Self-Injury, SMA self-reported using NSSI for intrapersonal functions (eg, for affect regulation, antisuicide, self-punishment) significantly greater than their heterosexual peers; however, there were no significant differences between the 2 groups on interpersonal functions (eg, autonomy, interpersonal boundaries, peer bonding, sensation-seeking).16
Continue to: Transgender and gender nonconfirming...
Transgender and gender nonconfirming (GNC) youth are at a particularly high risk for NSSI; 30% to 45.5% of transgender adolescents report self-injury.17 Factors shown to distinguish transgender/GNC youth who engage in NSSI from those who do not include having a mental health problem, depression, running away from home, substance use, lower self-esteem/greater self-criticism, experiencing transphobia victimization, and having more interpersonal problems.18,19 Among transgender/GNC youth, those whose biological sex is female are more likely to report NSSI than those whose biological sex is male (ie, transgendered adolescent males are more likely to report NSSI than transgendered adolescent females).18,19
Most forms of childhood maltreatment have been associated with NSSI. In a recently published review, Liu et al20 found that childhood maltreatment (including sexual abuse, physical abuse, emotional abuse, and physical neglect) was associated with an increased risk for NSSI. However, conflicting evidence suggests that when confounders are removed, only childhood emotional abuse was directly associated with NSSI.21 Current evidence is modest for childhood emotional neglect as a risk factor for NSSI.20
Increasing research is investigating the biological processes that may be implicated in NSSI. Some studies suggest that endogenous opioids,22 monoamine neurotransmitters,22 and the hypothalamic-pituitary-adrenal (HPA) axis23 may play a role in NSSI. Compared to healthy controls, adolescents engaging in NSSI have been shown to have lower pain intensity (P = .036), higher pain thresholds (P = .040), and lower beta-endorphins (endogenous opioid hormones involved in mediating stress and pain) (P = .002).24 There may be alterations in the HPA axis among adolescents who engage in NSSI, more specifically stronger cortisol awakening responses.23 Both functional and standard MRI have been used to study the neurobiology of NSSI. One study demonstrated differences in functional connectivity between brain areas linked to neuroregulation of emotions in adolescents who engage in NSSI,25 while another found volume reduction in the insula of these adolescents, which suggests a possible neurobiological reason for impulsivity and the increased risk of suicidal behavior.26
Dynamic risk factors
Research has repeatedly shown bullying is a risk factor for NSSI.27 One study found that younger children who were victimized reported significantly more NSSI than older children.28 New data suggest that perpetrators of bullying are also at risk for deliberate self-harm behavior (SHB), which this study defined as a behavior that is intended to cause self-harm but without suicidal intent and having a nonfatal outcome.29 Victims of cyberbullying also are at a greater risk for self-harm, suicidal behaviors, and suicide attempt.30 To a lesser extent, cyberbullying perpetrators are at greater risk for suicidal behaviors and suicidal ideation.30 Bullying is a risk factor for NSSI not only in adolescence, but also in adulthood. Lereya et al31 found that victims of bullying in childhood and early adolescence were more likely to have mental health problems (including anxiety and depression) and more likely to engage in SHB—which this study defined as hurting oneself on purpose in any way—as adults.
The effects of internet use on adolescents’ mental health also has been investigated. A recent review that explored the relationship between all types of internet use (general use, internet addiction, social media, self-harm websites, forums, etc) and SHB/suicidal behavior found that young people with internet addiction, high levels of internet use, and a tendency to view websites with self-harm or suicidal content were at higher risk of engaging in SHB/suicidal behavior.32 This study did not use a specific definition for SHB or suicidal behavior.32
Continue to: Membership in certain youth...
Membership in certain youth subcultures (eg, emo or goth) has been evaluated as potential risk factors for depression and deliberate self-harm. Bowes et al33 found that for each unit increase in goth affiliation (not at all, not very much, somewhat, more than somewhat, very much), youth were 1.52 times more likely to engage in SHB; these researchers also reported a dose-response association between goth identification and future SHB. This study asked participants if they have ever tried to harm or hurt themselves in any manner, but did not distinguish between individuals who had harmed themselves with and without suicidal intent.33
Personality traits such as impulsiveness and loneliness have been linked to NSSI among adolescents.34,35 A recent study found that adolescents who met the proposed DSM-5 diagnostic criteria for NSSI scored higher on the Barratt Impulsiveness Scale, specifically in measures of:
- motor impulsiveness (ie, acting without thinking)
- attentional impulsiveness (ie, making decisions quickly)
- impulsiveness due to lack of planning (ie, failure to plan for the future).34
This study also found that adolescents who identified as being lonely based on scores on the Brazilian Loneliness Scale were at a higher risk for NSSI.34
A recent systematic review (32 studies) and meta-analysis (9 studies) found that school absenteeism was associated with a risk of self-harm (pooled aOR 1.37, P = .01) and suicidal ideation (pooled aOR 1.20, P = .03).36 This study suggested that school absenteeism, an important marker of social exclusion, was associated with both SHB and suicidal ideation in young people.36 It defined SHB as any act of self-injury or self-poisoning, regardless of intent.36
Finally, family-related factors have been associated with an increased risk of NSSI. One study of 11,814 children age 9 and 10 revealed that high family conflict (OR 1.09; 95% CI, 1.05 to 1.14) and low parental monitoring (OR 0.95; 95% CI, 0.93 to 0.98) were associated with NSSI.37 A smaller, community-based study found that adolescents with NSSI reported significantly less maternal support and warmth than nonclinical controls, but a cause-and-effect relationship has not yet been determined.38 Parental history alone may influence adolescents’ risk of NSSI. A study that included nearly 76,000 youth found that adolescents with perceived parental alcohol problems had higher odds of self-injury, suicidal ideation, and suicide attempts.39 Adolescents exposed to maternal or paternal adversities were also at a higher risk of self-harm (hazard ratio 1.5 to 5.4 among males, 1.7 to 3.9 among females).40
Continue to: NSSI risk factors for adults
NSSI risk factors for adults
Although data regarding the prevalence of NSSI in adults are lacking, available studies report a 12-month prevalence of 0.9%2 and a lifetime prevalence of 5.5% to 5.9%.43 There is a significant overlap in risk factors for NSSI in adolescent and adult populations, but there are also many important differences. The static and dynamic risk factors for NSSI in adults are described in Table 3.44-66 Table 444-66 summarizes the studies of NSSI in adults that we reviewed.

Static risk factors
Research findings regarding the prevalence of NSSI based on gender are varied. For years, it has been believed that women are more likely to engage in NSSI than men. Recent meta-analyses that have examined this relationship closely found that the gender difference is larger for clinical samples compared to community samples and more pronounced in younger individuals.11

As is the case with adolescents, there may be ethnic variations in rates of self-harm and NSSI among adults. A 2013 study by Chesin et al44 found that Asian and White young adults experience higher rates of NSSI than their Hispanic and Black counterparts. Evidence suggests that relative rates of self-harm for older South Asian adults are lower than in older White adults.15
Compared to heterosexual or cisgender individuals, members of sexual and gender minorities have a higher past-year and lifetime prevalence of NSSI.45 One study found that the weighted effect size between sexual orientation and NSSI had an OR of 3 (95% CI, 2.46 to 3.66), indicating a medium-to-large effect.46 Bisexual and transgender individuals appear to be at the highest risk for NSSI when compared to members of other sexual and gender minority groups.45 One review that included mostly cross-sectional studies found that individuals identifying as bisexual had up to 6 times the odds of engaging in NSSI when compared to those of other sexual orientations.47
Incarceration is a risk factor for NSSI. The rates of NSSI in criminal justice settings are higher (up to 61%) than in the general adult population (approximately 4%).48 Recent research found that NSSI serves similar functions in correctional and non-correctional settings, primarily to regulate emotions.48 However, there is also evidence of higher rates of NSSI being motivated by an attempt to influence the environment (ie, engaging in NSSI in order to be transferred to another prison unit) compared to NSSI in community settings.48
Continue to: Though less robust than data...
Though less robust than data published regarding adolescents, the role of biological processes in adults engaging in NSSI has also been studied. A 2021 study by Störkel et al49 found that levels of salivary beta-endorphins were significantly lower in adults immediately before engaging in NSSI compared to after NSSI. Furthermore, adults who engage in NSSI have lower levels of met-enkephalin (P < .01), an opioid growth factor, compared to adults who have never engaged in NSSI.22
Dynamic risk factors
Individuals who engage in NSSI often report substance use, but there is little data on whether substance use is an independent risk factor for NSSI. Although limited, recent evidence suggests illicit substance use in both adolescents41 and adults50 increases risk for NSSI. Richardson et al50 found that the use of barbiturates, opiates, and sedatives significantly increased the frequency of NSSI, whereas use of marijuana, phencyclidine, and medications used to treat anxiety significantly increased the severity of NSSI. A smaller study conducted in South Africa found that individuals who engage in substance use and NSSI were more likely to be male (P < .001).51
Eating disorders and NSSI are highly comorbid.52 The lifetime prevalence of NSSI among individuals with eating disorders ranges from 20.6%to 37.1%.52,53 Results are inconsistent regarding which eating disorders (if any) are greater risk factors for NSSI. One study found that the prevalence of NSSI in patients with bulimia nervosa was 32.7% (95% CI, 26.9% to 39.1%) vs 21.8% in patients with anorexia nervosa (95% CI, 18.5% to 25.6%).54 Another study found that individuals with binge eating/purging–type eating disorders reported engaging in NSSI more frequently than those with other types of eating disorders.55 Among patients with eating disorders who reported NSSI, risk factors included younger age of onset, more negative self-evaluation, more impulsive behavior, concomitant substance use, history of suicide attempts, childhood abuse, and peer aggression.53,55 Body image dissatisfaction and self-criticism, even in individuals not formally diagnosed with an eating disorder, are small but significant predictors of NSSI.56,57
Mood disorders have also been linked to NSSI.58,59 Anxiety disorders (including generalized anxiety disorder, social phobia, panic disorder, and agoraphobia) as well as anxiety-related disorders such as obsessive-compulsive disorder have been significantly associated with NSSI (P < .001), but this relationship decreased in strength when mood instability was removed as a confounder.58 Among patients with anxiety and anxiety-related disorders, panic disorder and posttraumatic stress disorder (PTSD) have shown the strongest association with NSSI, with pooled aORs of 2.67 and 2.06, respectively.59
Recent studies have examined the association of other mental health disorders and symptoms with NSSI, including psychosis60 and dissociative symptoms.61 One study found that paranoia, thought control, and auditory hallucinations were significantly associated with NSSI60; however, after controlling for concomitant BPD, only paranoia was significantly associated with NSSI.60 Individuals diagnosed with dissociative disorders were more likely than patients without such disorders to endorse NSSI and suicide attempts.61
Continue to: Emotional dysregulation...
Emotional dysregulation (EDR)—defined as difficulty understanding, recognizing, and managing one’s emotions—has been researched extensively in relation to NSSI.62 A recent review that included studies of both adolescents and adults reported a significant association between EDR and NSSI, with an OR of 2.40 (95% CI, 2.01 to 2.86).62 A larger effect size was observed between EDR and lifetime NSSI (OR 3.21; 95% CI, 2.63 to 3.91) compared to past-year NSSI (OR 2.32; 95% CI, 1.84 to 2.92).62 Patient age, sex, and sample type (clinical vs community) were not significant moderators of strength between the reported associations.62
Studies examining intimate partner violence (IPV) and NSSI have found that young adults who engage in IPV (both as victims and as perpetrators) are more likely to report NSSI.63-65 Researchers have proposed that anxiety over abandonment may explain this relationship.64 A recent study found that individuals with bidirectional IPV (ie, both victimization and perpetration) engaged in NSSI at a higher prevalence than those engaging in unidirectional IPV or no IPV.65 This suggests that relationship violence in general (rather than just being a victim of IPV) may be a risk factor for NSSI.65
Finally, studies suggest that adolescents and adults who have sleep problems (insomnia, short sleep duration, long sleep onset latency, waking after sleep onset, and poor quality sleep) are more likely to report self-harm or NSSI than those without sleep problems.42,66 In adults, this relationship is partially mediated by depressive symptoms, EDR, and PTSD.66 In adolescents, depressive symptoms are a mediator for this relationship.42
Bottom Line
Nonsuicidal self-injury (NSSI) is a significant health concern due to its association with suicide attempts. Although there are similarities in NSSI risk factors between adolescents and adults, there are also important differences. Understanding these differences is necessary to develop appropriate treatment plans.
Related Resources
- American Foundation for Suicide Prevention. https://afsp.org/
- Cipriano A, Cella S, Cotrufo P. Nonsuicidal self-injury: a systematic review. Front Psych. 2017;8:1946. doi:10.3389/ fpsyg.2017.01946
- Gold LH, Frierson RL, eds. Textbook of Suicide Risk Assessment and Management. 3rd ed. American Psychiatric Association Publishing; 2020.
Nonsuicidal self-injury (NSSI) is the direct and deliberate destruction of body tissue without intent to die.1 Common forms of NSSI include cutting, burning, scraping/scratching skin, biting, hitting, and interfering with wound healing.2 Functional theories suggest that NSSI temporarily alleviates overwhelming negative emotions and can produce feelings of relief, resulting in a reinforcing effect.3
NSSI has been shown to be a risk factor for future suicide attempts.4 A 2018 study found that NSSI is associated with an increased risk of subsequent suicidal ideation (odds ratio [OR] 2.8), suicide plan (OR 3.0), and suicide attempt (OR 5.5).5 NSSI is also associated with individuals who had suicidal ideation and formed a suicide plan, and individuals who had a suicide plan and attempted suicide (ORs 1.7 to 2.1).5 Another study found that 70% of adolescents who engage in NSSI have attempted suicide during their lifetime, and 55% have multiple attempts.6
Given the overlap between suicide attempts and NSSI, performing a thorough suicide risk assessment (which is beyond the scope of this article) is crucial. This article describes the static and dynamic risk factors for NSSI in adolescents and adults, which can help us perform a suicide risk assessment and allow us to formulate an appropriate treatment plan that includes safety-based interventions.

NSSI risk factors for adolescents
From developing sexual identity and undergoing puberty to achieving increased independence from their parents and developing a sense of autonomy, adolescents undergo many biological, psychological, and social changes before reaching adulthood.7 Data suggest that NSSI often begins in adolescence, with a typical onset at age 13 or 14.3 Community studies show that one-third to one-half of adolescents in the United States have engaged in NSSI.8,9 Previously, NSSI during adolescence was associated with 3 major diagnostic categories: eating disorders, developmental disabilities, and borderline personality disorder (BPD).10 However, recent data suggest that NSSI is also common outside of these categories. Here we describe static and dynamic risk factors for NSSI in adolescents (Table 111-42). Table 211-42 summarizes the studies of NSSI in adolescents that we reviewed.

Static risk factors
Female adolescents and adults engage in NSSI at higher rates than males. The difference is larger in clinical populations compared to the general population.11
A large portion of research about NSSI has been conducted in studies in which the majority of participants were White.12 Most studies report a higher prevalence of NSSI among non-Hispanic White youth,13 but some suggest other ethnic groups may also experience high rates of self-harm and NSSI.13-15 Several studies have demonstrated high rates of self-harm among South Asian adult females compared with White adult females, but this difference may be less pronounced in adolescents.14 One study in the United Kingdom found that White females age 10 to 14 had higher rates of self-harm compared to South Asian females,14 while another found that risk and rates of self-harm in young South Asian people varied by city and country of origin.15 Young Black females15 and young Black males13 also may be at an increased risk of self-harm. One review found that Black females were more likely to self-harm than Asian or White groups.15
Several studies suggest that sexual minority adolescents (SMA) (eg, lesbian, gay, bisexual, transgender, queer) are at greater risk for NSSI than heterosexual adolescents.16 SMA have been shown to engage in a significantly greater frequency of NSSI and more types of NSSI than heterosexual adolescents.16 Furthermore, on the Inventory of Statements about Self-Injury, SMA self-reported using NSSI for intrapersonal functions (eg, for affect regulation, antisuicide, self-punishment) significantly greater than their heterosexual peers; however, there were no significant differences between the 2 groups on interpersonal functions (eg, autonomy, interpersonal boundaries, peer bonding, sensation-seeking).16
Continue to: Transgender and gender nonconfirming...
Transgender and gender nonconfirming (GNC) youth are at a particularly high risk for NSSI; 30% to 45.5% of transgender adolescents report self-injury.17 Factors shown to distinguish transgender/GNC youth who engage in NSSI from those who do not include having a mental health problem, depression, running away from home, substance use, lower self-esteem/greater self-criticism, experiencing transphobia victimization, and having more interpersonal problems.18,19 Among transgender/GNC youth, those whose biological sex is female are more likely to report NSSI than those whose biological sex is male (ie, transgendered adolescent males are more likely to report NSSI than transgendered adolescent females).18,19
Most forms of childhood maltreatment have been associated with NSSI. In a recently published review, Liu et al20 found that childhood maltreatment (including sexual abuse, physical abuse, emotional abuse, and physical neglect) was associated with an increased risk for NSSI. However, conflicting evidence suggests that when confounders are removed, only childhood emotional abuse was directly associated with NSSI.21 Current evidence is modest for childhood emotional neglect as a risk factor for NSSI.20
Increasing research is investigating the biological processes that may be implicated in NSSI. Some studies suggest that endogenous opioids,22 monoamine neurotransmitters,22 and the hypothalamic-pituitary-adrenal (HPA) axis23 may play a role in NSSI. Compared to healthy controls, adolescents engaging in NSSI have been shown to have lower pain intensity (P = .036), higher pain thresholds (P = .040), and lower beta-endorphins (endogenous opioid hormones involved in mediating stress and pain) (P = .002).24 There may be alterations in the HPA axis among adolescents who engage in NSSI, more specifically stronger cortisol awakening responses.23 Both functional and standard MRI have been used to study the neurobiology of NSSI. One study demonstrated differences in functional connectivity between brain areas linked to neuroregulation of emotions in adolescents who engage in NSSI,25 while another found volume reduction in the insula of these adolescents, which suggests a possible neurobiological reason for impulsivity and the increased risk of suicidal behavior.26
Dynamic risk factors
Research has repeatedly shown bullying is a risk factor for NSSI.27 One study found that younger children who were victimized reported significantly more NSSI than older children.28 New data suggest that perpetrators of bullying are also at risk for deliberate self-harm behavior (SHB), which this study defined as a behavior that is intended to cause self-harm but without suicidal intent and having a nonfatal outcome.29 Victims of cyberbullying also are at a greater risk for self-harm, suicidal behaviors, and suicide attempt.30 To a lesser extent, cyberbullying perpetrators are at greater risk for suicidal behaviors and suicidal ideation.30 Bullying is a risk factor for NSSI not only in adolescence, but also in adulthood. Lereya et al31 found that victims of bullying in childhood and early adolescence were more likely to have mental health problems (including anxiety and depression) and more likely to engage in SHB—which this study defined as hurting oneself on purpose in any way—as adults.
The effects of internet use on adolescents’ mental health also has been investigated. A recent review that explored the relationship between all types of internet use (general use, internet addiction, social media, self-harm websites, forums, etc) and SHB/suicidal behavior found that young people with internet addiction, high levels of internet use, and a tendency to view websites with self-harm or suicidal content were at higher risk of engaging in SHB/suicidal behavior.32 This study did not use a specific definition for SHB or suicidal behavior.32
Continue to: Membership in certain youth...
Membership in certain youth subcultures (eg, emo or goth) has been evaluated as potential risk factors for depression and deliberate self-harm. Bowes et al33 found that for each unit increase in goth affiliation (not at all, not very much, somewhat, more than somewhat, very much), youth were 1.52 times more likely to engage in SHB; these researchers also reported a dose-response association between goth identification and future SHB. This study asked participants if they have ever tried to harm or hurt themselves in any manner, but did not distinguish between individuals who had harmed themselves with and without suicidal intent.33
Personality traits such as impulsiveness and loneliness have been linked to NSSI among adolescents.34,35 A recent study found that adolescents who met the proposed DSM-5 diagnostic criteria for NSSI scored higher on the Barratt Impulsiveness Scale, specifically in measures of:
- motor impulsiveness (ie, acting without thinking)
- attentional impulsiveness (ie, making decisions quickly)
- impulsiveness due to lack of planning (ie, failure to plan for the future).34
This study also found that adolescents who identified as being lonely based on scores on the Brazilian Loneliness Scale were at a higher risk for NSSI.34
A recent systematic review (32 studies) and meta-analysis (9 studies) found that school absenteeism was associated with a risk of self-harm (pooled aOR 1.37, P = .01) and suicidal ideation (pooled aOR 1.20, P = .03).36 This study suggested that school absenteeism, an important marker of social exclusion, was associated with both SHB and suicidal ideation in young people.36 It defined SHB as any act of self-injury or self-poisoning, regardless of intent.36
Finally, family-related factors have been associated with an increased risk of NSSI. One study of 11,814 children age 9 and 10 revealed that high family conflict (OR 1.09; 95% CI, 1.05 to 1.14) and low parental monitoring (OR 0.95; 95% CI, 0.93 to 0.98) were associated with NSSI.37 A smaller, community-based study found that adolescents with NSSI reported significantly less maternal support and warmth than nonclinical controls, but a cause-and-effect relationship has not yet been determined.38 Parental history alone may influence adolescents’ risk of NSSI. A study that included nearly 76,000 youth found that adolescents with perceived parental alcohol problems had higher odds of self-injury, suicidal ideation, and suicide attempts.39 Adolescents exposed to maternal or paternal adversities were also at a higher risk of self-harm (hazard ratio 1.5 to 5.4 among males, 1.7 to 3.9 among females).40
Continue to: NSSI risk factors for adults
NSSI risk factors for adults
Although data regarding the prevalence of NSSI in adults are lacking, available studies report a 12-month prevalence of 0.9%2 and a lifetime prevalence of 5.5% to 5.9%.43 There is a significant overlap in risk factors for NSSI in adolescent and adult populations, but there are also many important differences. The static and dynamic risk factors for NSSI in adults are described in Table 3.44-66 Table 444-66 summarizes the studies of NSSI in adults that we reviewed.

Static risk factors
Research findings regarding the prevalence of NSSI based on gender are varied. For years, it has been believed that women are more likely to engage in NSSI than men. Recent meta-analyses that have examined this relationship closely found that the gender difference is larger for clinical samples compared to community samples and more pronounced in younger individuals.11

As is the case with adolescents, there may be ethnic variations in rates of self-harm and NSSI among adults. A 2013 study by Chesin et al44 found that Asian and White young adults experience higher rates of NSSI than their Hispanic and Black counterparts. Evidence suggests that relative rates of self-harm for older South Asian adults are lower than in older White adults.15
Compared to heterosexual or cisgender individuals, members of sexual and gender minorities have a higher past-year and lifetime prevalence of NSSI.45 One study found that the weighted effect size between sexual orientation and NSSI had an OR of 3 (95% CI, 2.46 to 3.66), indicating a medium-to-large effect.46 Bisexual and transgender individuals appear to be at the highest risk for NSSI when compared to members of other sexual and gender minority groups.45 One review that included mostly cross-sectional studies found that individuals identifying as bisexual had up to 6 times the odds of engaging in NSSI when compared to those of other sexual orientations.47
Incarceration is a risk factor for NSSI. The rates of NSSI in criminal justice settings are higher (up to 61%) than in the general adult population (approximately 4%).48 Recent research found that NSSI serves similar functions in correctional and non-correctional settings, primarily to regulate emotions.48 However, there is also evidence of higher rates of NSSI being motivated by an attempt to influence the environment (ie, engaging in NSSI in order to be transferred to another prison unit) compared to NSSI in community settings.48
Continue to: Though less robust than data...
Though less robust than data published regarding adolescents, the role of biological processes in adults engaging in NSSI has also been studied. A 2021 study by Störkel et al49 found that levels of salivary beta-endorphins were significantly lower in adults immediately before engaging in NSSI compared to after NSSI. Furthermore, adults who engage in NSSI have lower levels of met-enkephalin (P < .01), an opioid growth factor, compared to adults who have never engaged in NSSI.22
Dynamic risk factors
Individuals who engage in NSSI often report substance use, but there is little data on whether substance use is an independent risk factor for NSSI. Although limited, recent evidence suggests illicit substance use in both adolescents41 and adults50 increases risk for NSSI. Richardson et al50 found that the use of barbiturates, opiates, and sedatives significantly increased the frequency of NSSI, whereas use of marijuana, phencyclidine, and medications used to treat anxiety significantly increased the severity of NSSI. A smaller study conducted in South Africa found that individuals who engage in substance use and NSSI were more likely to be male (P < .001).51
Eating disorders and NSSI are highly comorbid.52 The lifetime prevalence of NSSI among individuals with eating disorders ranges from 20.6%to 37.1%.52,53 Results are inconsistent regarding which eating disorders (if any) are greater risk factors for NSSI. One study found that the prevalence of NSSI in patients with bulimia nervosa was 32.7% (95% CI, 26.9% to 39.1%) vs 21.8% in patients with anorexia nervosa (95% CI, 18.5% to 25.6%).54 Another study found that individuals with binge eating/purging–type eating disorders reported engaging in NSSI more frequently than those with other types of eating disorders.55 Among patients with eating disorders who reported NSSI, risk factors included younger age of onset, more negative self-evaluation, more impulsive behavior, concomitant substance use, history of suicide attempts, childhood abuse, and peer aggression.53,55 Body image dissatisfaction and self-criticism, even in individuals not formally diagnosed with an eating disorder, are small but significant predictors of NSSI.56,57
Mood disorders have also been linked to NSSI.58,59 Anxiety disorders (including generalized anxiety disorder, social phobia, panic disorder, and agoraphobia) as well as anxiety-related disorders such as obsessive-compulsive disorder have been significantly associated with NSSI (P < .001), but this relationship decreased in strength when mood instability was removed as a confounder.58 Among patients with anxiety and anxiety-related disorders, panic disorder and posttraumatic stress disorder (PTSD) have shown the strongest association with NSSI, with pooled aORs of 2.67 and 2.06, respectively.59
Recent studies have examined the association of other mental health disorders and symptoms with NSSI, including psychosis60 and dissociative symptoms.61 One study found that paranoia, thought control, and auditory hallucinations were significantly associated with NSSI60; however, after controlling for concomitant BPD, only paranoia was significantly associated with NSSI.60 Individuals diagnosed with dissociative disorders were more likely than patients without such disorders to endorse NSSI and suicide attempts.61
Continue to: Emotional dysregulation...
Emotional dysregulation (EDR)—defined as difficulty understanding, recognizing, and managing one’s emotions—has been researched extensively in relation to NSSI.62 A recent review that included studies of both adolescents and adults reported a significant association between EDR and NSSI, with an OR of 2.40 (95% CI, 2.01 to 2.86).62 A larger effect size was observed between EDR and lifetime NSSI (OR 3.21; 95% CI, 2.63 to 3.91) compared to past-year NSSI (OR 2.32; 95% CI, 1.84 to 2.92).62 Patient age, sex, and sample type (clinical vs community) were not significant moderators of strength between the reported associations.62
Studies examining intimate partner violence (IPV) and NSSI have found that young adults who engage in IPV (both as victims and as perpetrators) are more likely to report NSSI.63-65 Researchers have proposed that anxiety over abandonment may explain this relationship.64 A recent study found that individuals with bidirectional IPV (ie, both victimization and perpetration) engaged in NSSI at a higher prevalence than those engaging in unidirectional IPV or no IPV.65 This suggests that relationship violence in general (rather than just being a victim of IPV) may be a risk factor for NSSI.65
Finally, studies suggest that adolescents and adults who have sleep problems (insomnia, short sleep duration, long sleep onset latency, waking after sleep onset, and poor quality sleep) are more likely to report self-harm or NSSI than those without sleep problems.42,66 In adults, this relationship is partially mediated by depressive symptoms, EDR, and PTSD.66 In adolescents, depressive symptoms are a mediator for this relationship.42
Bottom Line
Nonsuicidal self-injury (NSSI) is a significant health concern due to its association with suicide attempts. Although there are similarities in NSSI risk factors between adolescents and adults, there are also important differences. Understanding these differences is necessary to develop appropriate treatment plans.
Related Resources
- American Foundation for Suicide Prevention. https://afsp.org/
- Cipriano A, Cella S, Cotrufo P. Nonsuicidal self-injury: a systematic review. Front Psych. 2017;8:1946. doi:10.3389/ fpsyg.2017.01946
- Gold LH, Frierson RL, eds. Textbook of Suicide Risk Assessment and Management. 3rd ed. American Psychiatric Association Publishing; 2020.
1. Nock MK. Self-injury. Annu Rev Clin Psychol. 2010;6:339-363.
2. Klonsky ED. Non-suicidal self-injury in United States adults: prevalence, sociodemographics, topography and functions. Psychol Med. 2011;41(9):1981-1986.
3. Klonsky ED. Nonsuicidal self-injury: what we know, and what we need to know. Can J Psychiatry. 2014;59(11):565-568.
4. Wilkinson P, Kelvin R, Roberts C, et al. Clinical and psychosocial predictors of suicide attempts and nonsuicidal self-injury in the Adolescent Depression Antidepressants and Psychotherapy Trial (ADAPT). Am J Psychiatry. 2011;168(5):495-501.
5. Kiekens G, Hasking P, Boyes M, et al. The associations between non-suicidal self-injury and first onset suicidal thoughts and behaviors. J Affect Disord. 2018;239:171-179.
6. Nock MK, Joiner TE, Gordon KH, et al. Non-suicidal self-injury among adolescents: diagnostic correlates and relation to suicide attempts. Psychiatry Res. 2006;144(1):65-72.
7. Christie D, Viner R. Adolescent development. BMJ. 2005;330(7486):301-304.
8. Yates TM, Tracy AJ, Luthar SS. Nonsuicidal self-injury among “privileged” youths: longitudinal and cross-sectional approaches to developmental process. J Consult Clin Psychol. 2008;76(1):52-62.
9. Lloyd-Richardson EE, Perrine N, Dierker L, et al. Characteristics and functions of non-suicidal self-injury in a community sample of adolescents. Psychol Med. 2007;37(8):1183-1192.
10. Peterson J, Freedenthal S, Sheldon C, et al. Nonsuicidal self injury in adolescents. Psychiatry(Edgmont). 2008;5(11):20-26.
11. Bresin K, Schoenleber M. Gender differences in the prevalence of nonsuicidal self-injury: a meta-analysis. Clin Psychol Rev. 2015;38:55-64.
12. Gholamrezaei M, Stefano JD, Heath NL. Nonsuicidal self-injury across cultures and ethnic and racial minorities: a review. Int J Psychol. 2015;52(4):316-326.
13. Rojas-Velasquez DA, Pluhar EI, Burns PA, et al. Nonsuicidal self-injury among African American and Hispanic adolescents and young adults: a systematic review. Prev Sci. 2021;22:367-377.
14. Bhui K, McKenzie K, Rasul F. Rates, risk factors & methods of self harm among minority ethnic groups in the UK: a systematic review. BMC Public Health. 2007;7:336.
15. Cooper J, Murphy E, Webb R, et al. Ethnic differences in self-harm, rates, characteristics and service provision: three-city cohort study. Br J Psychiatry. 2010;197(3):212-218.
16. Peters JR, Mereish EH, Krek MA, et al. Sexual orientation differences in non-suicidal self-injury, suicidality, and psychosocial factors among an inpatient psychiatric sample of adolescents. Psychiatry Res. 2020;284:112664.
17. Connolly MD, Zervos MJ, Barone 2nd CJ, et al. The mental health of transgender youth: advances in understanding. J Adolesc Health. 2016;59(5):489-495.
18. Taliaferro LA, McMorris BJ, Rider GN, et al. Risk and protective factors for self-harm in a population-based sample of transgender youth. Archives Suicide Res. 2019;23(2):203-221.
19. Arcelus J, Claes L, Witcomb GL, et al. Risk factors for non-suicidal self-injury among trans youth. J Sex Med. 2016;13(3):402-412.
20. Liu RT, Scopelliti KM, Pittman SK, et al. Childhood maltreatment and non-suicidal self-injury: a systematic review and meta-analysis. Lancet Psychiatry. 2018;5(1):51-64.
21. Thomassin K, Shaffer A, Madden A, et al. Specificity of childhood maltreatment and emotion deficit in nonsuicidal self-injury in an inpatient sample of youth. Psychiatry Res. 2016;244:103-108.
22. Stanley B, Sher L, Wilson S, et al. Non-suicidal self-injurious behavior, endogenous opioids and monoamine neurotransmitters. J Affect Disord. 2010;124(1-2):134-140.
23. Reichl C, Heyer A, Brunner R, et al. Hypothalamic-pituitary-adrenal axis, childhood adversity and adolescent nonsuicidal self-injury. Psychoneuroendocrinology. 2016;74:203-211.
24. van der Venne P, Balint A, Drews E, et al. Pain sensitivity and plasma beta-endorphin in adolescent non-suicidal self-injury. J Affect Disord. 2021;278:199-209.
25. Osuch E, Ford K, Wrath A, et al. Functional MRI of pain application in youth who engaged in repetitive non-suicidal self-injury vs. psychiatric controls. Psychiatry Res. 2014;223(2):104-112.
26. Ando A, Reichl C, Scheu F, et al. Regional grey matter volume reduction in adolescents engaging in non-suicidal self-injury. Psychiatry Res Neuroimaging. 2018;280:48-55.
27. Karanikola MNK, Lyberg A, Holm A-L, et al. The association between deliberate self-harm and school bullying victimization and the mediating effect of depressive symptoms and self-stigma: a systematic review. BioMed Res Int. 2018;4745791. doi: 10.1155/2018/4745791
28. van Geel M, Goemans A, Vedder P. A meta-analysis on the relation between peer victimization and adolescent non-suicidal self-injury. Psychiatry Res. 2015;230(2):364-368.
29. Heerde JA, Hemphill SA. Are bullying perpetration and victimization associated with adolescent deliberate self-harm? A meta-analysis. Arch Suicide Res. 2019;23(3):353-381.
30. John A, Glendenning AC, Marchant A, et al. Self-harm, suicidal behaviours, and cyberbullying in children and young people: systematic review. J Med Internet Res. 2018;20(4):e129. doi: 10.2196/jmir.9044
31. Lereya ST, Copeland WE, Costello EJ, et al. Adult mental health consequences of peer bullying and maltreatment in childhood: two cohorts in two countries. Lancet Psychiatry. 2015;2(6):524-531.
32. Marchant A, Hawton K, Stewart A, et al. A systematic review of the relationship between internet use, self-harm and suicidal behaviour in young people: the good, the bad and the unknown. PLoS One. 2017;12(8):e0181722. doi: 10.1371/journal.pone.0181722
33. Bowes L, Carnegie R, Pearson R, et al. Risk of depression and self-harm in teenagers identifying with goth subculture: a longitudinal cohort study. Lancet Psychiatry. 2015;2(9):793-800.
34. Costa RPO, Peixoto ALRP, Lucas CCA, et al. Profile of non-suicidal self-injury in adolescents: interface with impulsiveness and loneliness. J Pediatr (Rio J). 2021;97(2):184-190.
35. McHugh CM, Lee RSC, Hermens DF, et al. Impulsivity in the self-harm and suicidal behavior of young people: a systematic review and meta-analysis. J Psychiatr Res. 2019;116:51-60.
36. Epstein S, Roberts E, Sedgwick R, et al. School absenteeism as a risk factor for self-harm and suicidal ideation in children and adolescents: a systematic review and meta-analysis. Eur Child Adolesc Psychiatry. 2020;29(9):1175-1194.
37. DeVille DC, Whalen D, Breslin FJ, et al. Prevalence and family-related factors associated with suicidal ideation, suicide attempts, and self-injury in children aged 9 to 10 years. JAMA Netw Open. 2020;3(2):e1920956. doi: 10.1001/jamanetworkopen.2019.20956
38. Tschan T, Schmid M, In-Albon T. Parenting behavior in families of female adolescents with nonsuicidal self-injury in comparison to a clinical and a nonclinical control group. Child Adolesc Psychiatry Ment Health. 2015;9:17.
39. Pisinger V, Hawton K, Tolstrup JS. Self-injury and suicide behavior among young people with perceived parental alcohol problems in Denmark: a school-based survey. Eur Child Adolesc Psychiatry. 2018;27(2):201-208.
40. Pitkänen J, Remes H, Aaltonen M, et al. Experience of maternal and paternal adversities in childhood as determinants of self-harm in adolescence and young adulthood. J Epidemiol Community Health. 2019;73(11):1040-1046.
41. Monto MA, McRee N, Deryck FS. Nonsuicidal self-injury among a representative sample of US adolescents, 2015. Am J Public Health. 2018;108(8):1042-1048.
42. Hysing M, Sivertsen B, Stormark KM, et al. Sleep problems and self-harm in adolescence. Br J Psychiatry. 2015;207(4):306-312.
43. Swannell SV, Martin GE, Page A, et al. Prevalence of nonsuicidal self-injury in nonclinical samples: systematic review, meta-analysis and meta-regression. Suicide Life Threat Behav. 2014;44(3):273-303.
44. Chesin M, Moster A, Jeglic E. Non-suicidal self-injury among ethnically and racially diverse emerging adults: do factors unique to the minority experience matter? Current Psychology. 2013;32:318-328.
45. Liu RT, Sheehan AE, Walsh RFL, et al. Prevalence and correlates of non-suicidal self-injury among lesbian, gay, bisexual, and transgender individuals: a systematic review and meta-analysis. Clin Psychol Rev. 2019;74:101-783. doi:10.1016/j.cpr.2019.101783
46. Batejan KL, Jarvi SM, Swenson LP. Sexual orientation and non-suicidal self-injury: a meta-analytic review. Arch Suicide Res. 2015;19(2):131-150.
47. Dunlop BJ, Hartley S, Oladokun O, et al. Bisexuality and non-suicidal self-injury (NSSI): a narrative synthesis of associated variables and a meta-analysis of risk. J Affect Disord. 2020;276:1159-1172.
48. Dixon-Gordon K, Harrison N, Roesch R. Non-suicidal self-injury within offender populations: a systematic review. Int J Forensic Ment Health. 2012;11(1):33-50.
49. Störkel LM, Karabatsiakis A, Hepp K, et al. Salivary beta-endorphin in nonsuicidal self-injury: an ambulatory assessment study. Neuropsychopharmacology. 2021;46(7):1357-1363.
50. Richardson E, DePue MK, Therriault DJ, et al. The influence of substance use on engagement in non-suicidal self-injury (NSI) in adults. Subst Use Misuse. 2020;55(1):89-94.
51. Breet E, Bantjes J, Lewis I. Chronic substance use and self-harm in a primary health care setting. Afr J Prim Health Care Fam Med. 2018;10(1):e1-e9. doi: 10.4102/phcfm.v10i1.1544
52. Pérez S, Marco JH, Cañabate M. Non-suicidal self-injury in patients with eating disorders: prevalence, forms, functions, and body image correlates. Compr Psychiatry. 2018;84:32-38.
53. Islam MA, Steiger H, Jimenez-Murcia S, et al. Non-suicidal self-injury in different eating disorder types: relevance of personality traits and gender. Eur Eat Disord Rev. 2015;23(6):553-560.
54. Cucchi A, Ryan D, Konstantakopoulos G, et al. Lifetime prevalence of non-suicidal self-injury in patients with eating disorders: a systematic review and meta-analysis. Psychol Med. 2016;46(7):1345-1358.
55. Vieira AI, Machado BC, Machado PPP, et al. Putative risk factors for non-suicidal self-injury in eating disorders. Eur Eat Disord Rev. 2017;25(6):544-550.
56. Black EB, Garratt M, Beccaria G, et al. Body image as a predictor of nonsuicidal self-injury in women: a longitudinal study. Compr Psychiatry. 2019;88:83-89.
57. Zelkowitz RL, Cole DA. Self-criticism as a transdiagnostic process in nonsuicidal self-injury and disordered eating: systematic review and meta-analysis. Suicide Life Threat Behav. 2019;49(1):310-327.
58. Peters EM, Bowen R, Balbuena L. Mood instability contributes to impulsivity, non-suicidal self-injury, and binge eating/purging in people with anxiety disorders. Psychol Psychother. 2019;92(3):422-438.
59. Bentley KH, Cassiello-Robbins CF, Vittorio L, et al. The association between nonsuicidal self-injury and the emotional disorders: a meta-analytic review. Clin Psychol Rev. 2015;37:72-88.
60. Koyanagi A, Stickley A, Haro JM. Psychotic-like experiences and nonsuicidal self-injury in England: results from a national survey [corrected]. PLoS One. 2015;10(12):e0145533. doi: 10.1371/journal.pone.0145533
61. Calati R, Bensassi I, Courtet P. The link between dissociation and both suicide attempts and non-suicidal self-injury: meta-analyses. Psychiatry Res. 2017;251:103-114.
62. Wolff JC, Thompson E, Thomas SA, et al. Emotion dysregulation and non-suicidal self-injury: a systematic review and meta-analysis. Eur Psychiatry. 2019;59:25-36.
63. Vaughn MG, Salas-Wright CP, DeLisi M, et al. Deliberate self-harm and the nexus of violence, victimization, and mental health problems in the United States. Psychiatry Res. 2015;225(3):588-595.
64. Levesque C, Lafontaine M-F, Bureau J-F, et al. The influence of romantic attachment and intimate partner violence on nonsuicidal self-injury in young adults. J Youth Adolesc. 2010;39(5):474-483.
65. Carranza AB, Wallis CRD, Jonnson MR, et al. Nonsuicidal self-injury and intimate partner violence: directionality of violence and motives for self-injury. J Interpers Violence. 2020;886260520922372. doi: 10.1177/0886260520922372
66. Khazaie H, Zakiei A, McCall WV, et al. Relationship between sleep problems and self-injury: a systematic review. Behav Sleep Med. 2020;1-16. doi: 10.1080/15402002.2020.1822360
1. Nock MK. Self-injury. Annu Rev Clin Psychol. 2010;6:339-363.
2. Klonsky ED. Non-suicidal self-injury in United States adults: prevalence, sociodemographics, topography and functions. Psychol Med. 2011;41(9):1981-1986.
3. Klonsky ED. Nonsuicidal self-injury: what we know, and what we need to know. Can J Psychiatry. 2014;59(11):565-568.
4. Wilkinson P, Kelvin R, Roberts C, et al. Clinical and psychosocial predictors of suicide attempts and nonsuicidal self-injury in the Adolescent Depression Antidepressants and Psychotherapy Trial (ADAPT). Am J Psychiatry. 2011;168(5):495-501.
5. Kiekens G, Hasking P, Boyes M, et al. The associations between non-suicidal self-injury and first onset suicidal thoughts and behaviors. J Affect Disord. 2018;239:171-179.
6. Nock MK, Joiner TE, Gordon KH, et al. Non-suicidal self-injury among adolescents: diagnostic correlates and relation to suicide attempts. Psychiatry Res. 2006;144(1):65-72.
7. Christie D, Viner R. Adolescent development. BMJ. 2005;330(7486):301-304.
8. Yates TM, Tracy AJ, Luthar SS. Nonsuicidal self-injury among “privileged” youths: longitudinal and cross-sectional approaches to developmental process. J Consult Clin Psychol. 2008;76(1):52-62.
9. Lloyd-Richardson EE, Perrine N, Dierker L, et al. Characteristics and functions of non-suicidal self-injury in a community sample of adolescents. Psychol Med. 2007;37(8):1183-1192.
10. Peterson J, Freedenthal S, Sheldon C, et al. Nonsuicidal self injury in adolescents. Psychiatry(Edgmont). 2008;5(11):20-26.
11. Bresin K, Schoenleber M. Gender differences in the prevalence of nonsuicidal self-injury: a meta-analysis. Clin Psychol Rev. 2015;38:55-64.
12. Gholamrezaei M, Stefano JD, Heath NL. Nonsuicidal self-injury across cultures and ethnic and racial minorities: a review. Int J Psychol. 2015;52(4):316-326.
13. Rojas-Velasquez DA, Pluhar EI, Burns PA, et al. Nonsuicidal self-injury among African American and Hispanic adolescents and young adults: a systematic review. Prev Sci. 2021;22:367-377.
14. Bhui K, McKenzie K, Rasul F. Rates, risk factors & methods of self harm among minority ethnic groups in the UK: a systematic review. BMC Public Health. 2007;7:336.
15. Cooper J, Murphy E, Webb R, et al. Ethnic differences in self-harm, rates, characteristics and service provision: three-city cohort study. Br J Psychiatry. 2010;197(3):212-218.
16. Peters JR, Mereish EH, Krek MA, et al. Sexual orientation differences in non-suicidal self-injury, suicidality, and psychosocial factors among an inpatient psychiatric sample of adolescents. Psychiatry Res. 2020;284:112664.
17. Connolly MD, Zervos MJ, Barone 2nd CJ, et al. The mental health of transgender youth: advances in understanding. J Adolesc Health. 2016;59(5):489-495.
18. Taliaferro LA, McMorris BJ, Rider GN, et al. Risk and protective factors for self-harm in a population-based sample of transgender youth. Archives Suicide Res. 2019;23(2):203-221.
19. Arcelus J, Claes L, Witcomb GL, et al. Risk factors for non-suicidal self-injury among trans youth. J Sex Med. 2016;13(3):402-412.
20. Liu RT, Scopelliti KM, Pittman SK, et al. Childhood maltreatment and non-suicidal self-injury: a systematic review and meta-analysis. Lancet Psychiatry. 2018;5(1):51-64.
21. Thomassin K, Shaffer A, Madden A, et al. Specificity of childhood maltreatment and emotion deficit in nonsuicidal self-injury in an inpatient sample of youth. Psychiatry Res. 2016;244:103-108.
22. Stanley B, Sher L, Wilson S, et al. Non-suicidal self-injurious behavior, endogenous opioids and monoamine neurotransmitters. J Affect Disord. 2010;124(1-2):134-140.
23. Reichl C, Heyer A, Brunner R, et al. Hypothalamic-pituitary-adrenal axis, childhood adversity and adolescent nonsuicidal self-injury. Psychoneuroendocrinology. 2016;74:203-211.
24. van der Venne P, Balint A, Drews E, et al. Pain sensitivity and plasma beta-endorphin in adolescent non-suicidal self-injury. J Affect Disord. 2021;278:199-209.
25. Osuch E, Ford K, Wrath A, et al. Functional MRI of pain application in youth who engaged in repetitive non-suicidal self-injury vs. psychiatric controls. Psychiatry Res. 2014;223(2):104-112.
26. Ando A, Reichl C, Scheu F, et al. Regional grey matter volume reduction in adolescents engaging in non-suicidal self-injury. Psychiatry Res Neuroimaging. 2018;280:48-55.
27. Karanikola MNK, Lyberg A, Holm A-L, et al. The association between deliberate self-harm and school bullying victimization and the mediating effect of depressive symptoms and self-stigma: a systematic review. BioMed Res Int. 2018;4745791. doi: 10.1155/2018/4745791
28. van Geel M, Goemans A, Vedder P. A meta-analysis on the relation between peer victimization and adolescent non-suicidal self-injury. Psychiatry Res. 2015;230(2):364-368.
29. Heerde JA, Hemphill SA. Are bullying perpetration and victimization associated with adolescent deliberate self-harm? A meta-analysis. Arch Suicide Res. 2019;23(3):353-381.
30. John A, Glendenning AC, Marchant A, et al. Self-harm, suicidal behaviours, and cyberbullying in children and young people: systematic review. J Med Internet Res. 2018;20(4):e129. doi: 10.2196/jmir.9044
31. Lereya ST, Copeland WE, Costello EJ, et al. Adult mental health consequences of peer bullying and maltreatment in childhood: two cohorts in two countries. Lancet Psychiatry. 2015;2(6):524-531.
32. Marchant A, Hawton K, Stewart A, et al. A systematic review of the relationship between internet use, self-harm and suicidal behaviour in young people: the good, the bad and the unknown. PLoS One. 2017;12(8):e0181722. doi: 10.1371/journal.pone.0181722
33. Bowes L, Carnegie R, Pearson R, et al. Risk of depression and self-harm in teenagers identifying with goth subculture: a longitudinal cohort study. Lancet Psychiatry. 2015;2(9):793-800.
34. Costa RPO, Peixoto ALRP, Lucas CCA, et al. Profile of non-suicidal self-injury in adolescents: interface with impulsiveness and loneliness. J Pediatr (Rio J). 2021;97(2):184-190.
35. McHugh CM, Lee RSC, Hermens DF, et al. Impulsivity in the self-harm and suicidal behavior of young people: a systematic review and meta-analysis. J Psychiatr Res. 2019;116:51-60.
36. Epstein S, Roberts E, Sedgwick R, et al. School absenteeism as a risk factor for self-harm and suicidal ideation in children and adolescents: a systematic review and meta-analysis. Eur Child Adolesc Psychiatry. 2020;29(9):1175-1194.
37. DeVille DC, Whalen D, Breslin FJ, et al. Prevalence and family-related factors associated with suicidal ideation, suicide attempts, and self-injury in children aged 9 to 10 years. JAMA Netw Open. 2020;3(2):e1920956. doi: 10.1001/jamanetworkopen.2019.20956
38. Tschan T, Schmid M, In-Albon T. Parenting behavior in families of female adolescents with nonsuicidal self-injury in comparison to a clinical and a nonclinical control group. Child Adolesc Psychiatry Ment Health. 2015;9:17.
39. Pisinger V, Hawton K, Tolstrup JS. Self-injury and suicide behavior among young people with perceived parental alcohol problems in Denmark: a school-based survey. Eur Child Adolesc Psychiatry. 2018;27(2):201-208.
40. Pitkänen J, Remes H, Aaltonen M, et al. Experience of maternal and paternal adversities in childhood as determinants of self-harm in adolescence and young adulthood. J Epidemiol Community Health. 2019;73(11):1040-1046.
41. Monto MA, McRee N, Deryck FS. Nonsuicidal self-injury among a representative sample of US adolescents, 2015. Am J Public Health. 2018;108(8):1042-1048.
42. Hysing M, Sivertsen B, Stormark KM, et al. Sleep problems and self-harm in adolescence. Br J Psychiatry. 2015;207(4):306-312.
43. Swannell SV, Martin GE, Page A, et al. Prevalence of nonsuicidal self-injury in nonclinical samples: systematic review, meta-analysis and meta-regression. Suicide Life Threat Behav. 2014;44(3):273-303.
44. Chesin M, Moster A, Jeglic E. Non-suicidal self-injury among ethnically and racially diverse emerging adults: do factors unique to the minority experience matter? Current Psychology. 2013;32:318-328.
45. Liu RT, Sheehan AE, Walsh RFL, et al. Prevalence and correlates of non-suicidal self-injury among lesbian, gay, bisexual, and transgender individuals: a systematic review and meta-analysis. Clin Psychol Rev. 2019;74:101-783. doi:10.1016/j.cpr.2019.101783
46. Batejan KL, Jarvi SM, Swenson LP. Sexual orientation and non-suicidal self-injury: a meta-analytic review. Arch Suicide Res. 2015;19(2):131-150.
47. Dunlop BJ, Hartley S, Oladokun O, et al. Bisexuality and non-suicidal self-injury (NSSI): a narrative synthesis of associated variables and a meta-analysis of risk. J Affect Disord. 2020;276:1159-1172.
48. Dixon-Gordon K, Harrison N, Roesch R. Non-suicidal self-injury within offender populations: a systematic review. Int J Forensic Ment Health. 2012;11(1):33-50.
49. Störkel LM, Karabatsiakis A, Hepp K, et al. Salivary beta-endorphin in nonsuicidal self-injury: an ambulatory assessment study. Neuropsychopharmacology. 2021;46(7):1357-1363.
50. Richardson E, DePue MK, Therriault DJ, et al. The influence of substance use on engagement in non-suicidal self-injury (NSI) in adults. Subst Use Misuse. 2020;55(1):89-94.
51. Breet E, Bantjes J, Lewis I. Chronic substance use and self-harm in a primary health care setting. Afr J Prim Health Care Fam Med. 2018;10(1):e1-e9. doi: 10.4102/phcfm.v10i1.1544
52. Pérez S, Marco JH, Cañabate M. Non-suicidal self-injury in patients with eating disorders: prevalence, forms, functions, and body image correlates. Compr Psychiatry. 2018;84:32-38.
53. Islam MA, Steiger H, Jimenez-Murcia S, et al. Non-suicidal self-injury in different eating disorder types: relevance of personality traits and gender. Eur Eat Disord Rev. 2015;23(6):553-560.
54. Cucchi A, Ryan D, Konstantakopoulos G, et al. Lifetime prevalence of non-suicidal self-injury in patients with eating disorders: a systematic review and meta-analysis. Psychol Med. 2016;46(7):1345-1358.
55. Vieira AI, Machado BC, Machado PPP, et al. Putative risk factors for non-suicidal self-injury in eating disorders. Eur Eat Disord Rev. 2017;25(6):544-550.
56. Black EB, Garratt M, Beccaria G, et al. Body image as a predictor of nonsuicidal self-injury in women: a longitudinal study. Compr Psychiatry. 2019;88:83-89.
57. Zelkowitz RL, Cole DA. Self-criticism as a transdiagnostic process in nonsuicidal self-injury and disordered eating: systematic review and meta-analysis. Suicide Life Threat Behav. 2019;49(1):310-327.
58. Peters EM, Bowen R, Balbuena L. Mood instability contributes to impulsivity, non-suicidal self-injury, and binge eating/purging in people with anxiety disorders. Psychol Psychother. 2019;92(3):422-438.
59. Bentley KH, Cassiello-Robbins CF, Vittorio L, et al. The association between nonsuicidal self-injury and the emotional disorders: a meta-analytic review. Clin Psychol Rev. 2015;37:72-88.
60. Koyanagi A, Stickley A, Haro JM. Psychotic-like experiences and nonsuicidal self-injury in England: results from a national survey [corrected]. PLoS One. 2015;10(12):e0145533. doi: 10.1371/journal.pone.0145533
61. Calati R, Bensassi I, Courtet P. The link between dissociation and both suicide attempts and non-suicidal self-injury: meta-analyses. Psychiatry Res. 2017;251:103-114.
62. Wolff JC, Thompson E, Thomas SA, et al. Emotion dysregulation and non-suicidal self-injury: a systematic review and meta-analysis. Eur Psychiatry. 2019;59:25-36.
63. Vaughn MG, Salas-Wright CP, DeLisi M, et al. Deliberate self-harm and the nexus of violence, victimization, and mental health problems in the United States. Psychiatry Res. 2015;225(3):588-595.
64. Levesque C, Lafontaine M-F, Bureau J-F, et al. The influence of romantic attachment and intimate partner violence on nonsuicidal self-injury in young adults. J Youth Adolesc. 2010;39(5):474-483.
65. Carranza AB, Wallis CRD, Jonnson MR, et al. Nonsuicidal self-injury and intimate partner violence: directionality of violence and motives for self-injury. J Interpers Violence. 2020;886260520922372. doi: 10.1177/0886260520922372
66. Khazaie H, Zakiei A, McCall WV, et al. Relationship between sleep problems and self-injury: a systematic review. Behav Sleep Med. 2020;1-16. doi: 10.1080/15402002.2020.1822360