User login
Cardiology News is an independent news source that provides cardiologists with timely and relevant news and commentary about clinical developments and the impact of health care policy on cardiology and the cardiologist's practice. Cardiology News Digital Network is the online destination and multimedia properties of Cardiology News, the independent news publication for cardiologists. Cardiology news is the leading source of news and commentary about clinical developments in cardiology as well as health care policy and regulations that affect the cardiologist's practice. Cardiology News Digital Network is owned by Frontline Medical Communications.
Working Hard or Work Addiction — Have You Crossed the Line?
When child psychiatrist Javeed Sukhera, MD, PhD, was a few years into his career, he found himself doing it all. “I was in a leadership role academically at the medical school, I had a leadership role at the hospital, and I was seeing as many patients as I could. I could work all day every day.”
“It still wouldn’t have been enough,” he said.
Whenever there was a shift available, Dr. Sukhera would take it. His job was stressful, but as a new physician with a young family, he saw this obsession with work as necessary. “I began to cope with the stress from work by doing extra work and feeling like I needed to be everywhere. It was like I became a hamster on a spinning wheel. I was just running, running, running.”
Things shifted for Dr. Sukhera when he realized that while he was emotionally available for the children who were his patients, at home, his own children weren’t getting the best of him. “There was a specific moment when I thought my son was afraid of me,” he said. “I just stopped and realized that there was something happening that I needed to break. I needed to make a change.”
Dr. Sukhera, now chair of psychiatry at the Institute of Living and chief of the Department of Psychiatry at Hartford Hospital, Hartford, Connecticut, believes what he experienced was a steep fall into work addiction.
What Does Work Addiction Look Like for Doctors?
Behavioral addictions are fairly new in the addiction space. When gambling disorder, the first and only behavioral addiction in the Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition, was added in 2013, it was seen as a “breakthrough addiction,” said Mark D. Griffiths, PhD, a leading behavioral addiction researcher and a distinguished professor at Nottingham Trent University.
Because there is not enough evidence yet to classify work addiction as a formal diagnosis, there is no clear consensus on how to define it. To further complicate things, the terms “workaholism” and “work addiction” can be used interchangeably, and some experts say the two are not the same, though they can overlap.
That said, a 2018 review of literature from several countries found that work addiction “fits very well into recently postulated criteria for conceptualization of a behavioral addiction.
“If you accept that gambling can be genuinely addictive, then there’s no reason to think that something like work, exercise, or video game playing couldn’t be an addiction as well,” said Dr. Griffiths.
“The neurobiology of addiction is that we get drawn to something that gives us a dopamine hit,” Dr. Sukhera added. “But to do that all day, every day, has consequences. It drains our emotional reserves, and it can greatly impact our relationships.”
On top of that, work addiction has been linked with poor sleep, poor cardiovascular health, high blood pressure, burnout, the development of autoimmune disorders, and other health issues.
Physicians are particularly susceptible. Doctors, after all, are expected to work long hours and put their patients’ needs first, even at the expense of their own health and well-being.
“Workaholism is not just socially acceptable in medicine,” said Dr. Sukhera. “It’s baked into the system and built into the structures. The healthcare system has largely functioned on the emotional labor of health workers, whose tendency to show up and work harder can, at times, in certain organizations, be exploited.”
Dr. Griffiths agreed that with the limited amount of data available, work addiction does appear to exist at higher rates in medicine than in other fields. As early as the 1970s, medical literature describes work as a “socially acceptable” addiction among doctors. A 2014 study published in Occupational Medicine reported that of 445 physicians who took part in the research, nearly half exhibited some level of work addiction with 13% “highly work addicted.”
Of course, working hard or even meeting unreasonable demands from work is not the same as work addiction, as Dr. Griffiths clarified in a 2023 editorial in BMJ Quality & Safety. The difference, as with other behavioral addictions, is when people obsess about work and use it to cope with stress. It can be easier to stay distracted and busy to gain a sense of control rather than learning to deal with complex emotions.
A 2021 study that Dr. Sukhera conducted with resident physicians found that working harder was one of the main ways they dealt with stress during the COVID-19 pandemic. “This idea that we deal with the stress of being burnt out by doing more and more of what burns us out is fairly ubiquitous at all stages of medical professionals’ careers,” he said.
Financial incentives also can fuel work addiction, said Dr. Sukhera. In residency, there are some safeguards around overwork and duty hours. When you become an attending, those limits no longer exist. As a young physician, Dr. Sukhera had student debt to pay off and a family to support. When he found opportunities to earn more by working more, his answer was always “yes.”
Pressure to produce medical research also can pose issues. Some physicians can become addicted to publishing studies, fearing that they might lose their professional status or position if they stop. It’s a cycle that can force a doctor to not only work long hours doing their job but also practically take on a second one.
How Physicians Can Recognize Work Addiction in Themselves
Work addiction can look and feel different for every person, said Malissa Clark, PhD, associate professor at the University of Georgia and author of the recent book Never Not Working: Why the Always-On Culture Is Bad for Business—and How to Fix It.
Dr. Clark noted that people who are highly engaged in their work tend to be driven by intrinsic motivation: “You work because you love it.” With work addiction, “you work because you feel like you ought to be working all the time.”
Of course, it’s not always so cut and dried; you can experience both forms of motivation and not necessarily become addicted to work. But if you are solely driven by the feeling that you ought to be working all the time, that can be a red flag.
Dr. Griffiths said that while many people may have problematic work habits or work too much, true work addicts must meet six criteria that apply to all addictions:
1. Salience: Work is the single most important thing in your life, to the point of neglecting everything else. Even if you’re on vacation, your mind might be flooded with work thoughts.
2. Mood modification: You use work to modify your mood, either to get a “high” or to cope with stress.
3. Tolerance: Over time, you’ve gone from working 8 or 10 hours a day to 12 hours a day, to a point where you’re working all the time.
4. Withdrawal: On a physiological level, you will have symptoms such as anxiety, nausea, or headaches when unable to work.
5. Conflict: You feel conflicted with yourself (you know you’re working too much) or with others (partners, friends, and children) about work, but you can’t stop.
6. Relapse: If you manage to cut down your hours but can’t resist overworking 1 day, you wind up right back where you were.
When It’s Time to Address Work Addiction
The lack of a formal diagnosis for work addiction makes getting treatment difficult. But there are ways to seek help. Unlike the drug and alcohol literature, abstinence is not the goal. “The therapeutic goal is getting a behavior under control and looking for the triggers of why you’re compulsively working,” said Dr. Griffiths.
Practice self-compassion
Dr. Sukhera eventually realized that his work addiction stemmed from the fear of being somehow excluded or unworthy. He actively corrected much of this through self-compassion and self-kindness, which helped him set boundaries. “Self-compassion is the root of everything,” he said. “Reminding ourselves that we’re doing our best is an important ingredient in breaking the cycle.”
Slowly expose yourself to relaxation
Many workaholics find rest very difficult. “When I conducted interviews with people [who considered themselves workaholics], a very common thing I heard was, ‘I have a very hard time being idle,’ ” said Dr. Clark. If rest feels hard, Dr. Sukhera suggests practicing relaxation for 2 minutes to start. Even small periods of downtime can challenge the belief that you must be constantly productive.
Reframe your to-do list
For work addicts, to-do lists can seem like they must be finished, which prolongs work hours. Instead, use to-do lists to help prioritize what is urgent, identify what can wait, and delegate out tasks to others, Dr. Clark recommends.
Pick up a mastery experience
Research from professor Sabine Sonnentag, Dr. rer. nat., at the University of Mannheim, Mannheim, Germany, suggests that mastery experiences — leisure activities that require thought and focus like learning a new language or taking a woodworking class — can help you actively disengage from work.
Try cognitive behavioral therapy
Widely used for other forms of addiction, cognitive behavioral therapy centers around recognizing emotions, challenging thought patterns, and changing behaviors. However, Dr. Clark admits the research on its impact on work addiction, in particular, is “pretty nascent.”
Shift your mindset
It seems logical to think that detaching from your feelings will allow you to “do more,” but experts say that idea is both untrue and dangerous. “The safest hospitals are the hospitals where people are attuned to their humanness,” said Dr. Sukhera. “It’s normal to overwork in medicine, and if you’re challenging a norm, you really have to be thoughtful about how you frame that for yourself.”
Most importantly: Seek support
Today, there is increased awareness about work addiction and more resources for physicians who are struggling, including programs such as Workaholics Anonymous or Physicians Anonymous and workplace wellness initiatives. But try not to overwhelm yourself with choosing whom to talk to or what specific resource to utilize, Dr. Sukhera advised. “Just talk to someone about it. You don’t have to carry this on your own.”
A version of this article appeared on Medscape.com.
When child psychiatrist Javeed Sukhera, MD, PhD, was a few years into his career, he found himself doing it all. “I was in a leadership role academically at the medical school, I had a leadership role at the hospital, and I was seeing as many patients as I could. I could work all day every day.”
“It still wouldn’t have been enough,” he said.
Whenever there was a shift available, Dr. Sukhera would take it. His job was stressful, but as a new physician with a young family, he saw this obsession with work as necessary. “I began to cope with the stress from work by doing extra work and feeling like I needed to be everywhere. It was like I became a hamster on a spinning wheel. I was just running, running, running.”
Things shifted for Dr. Sukhera when he realized that while he was emotionally available for the children who were his patients, at home, his own children weren’t getting the best of him. “There was a specific moment when I thought my son was afraid of me,” he said. “I just stopped and realized that there was something happening that I needed to break. I needed to make a change.”
Dr. Sukhera, now chair of psychiatry at the Institute of Living and chief of the Department of Psychiatry at Hartford Hospital, Hartford, Connecticut, believes what he experienced was a steep fall into work addiction.
What Does Work Addiction Look Like for Doctors?
Behavioral addictions are fairly new in the addiction space. When gambling disorder, the first and only behavioral addiction in the Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition, was added in 2013, it was seen as a “breakthrough addiction,” said Mark D. Griffiths, PhD, a leading behavioral addiction researcher and a distinguished professor at Nottingham Trent University.
Because there is not enough evidence yet to classify work addiction as a formal diagnosis, there is no clear consensus on how to define it. To further complicate things, the terms “workaholism” and “work addiction” can be used interchangeably, and some experts say the two are not the same, though they can overlap.
That said, a 2018 review of literature from several countries found that work addiction “fits very well into recently postulated criteria for conceptualization of a behavioral addiction.
“If you accept that gambling can be genuinely addictive, then there’s no reason to think that something like work, exercise, or video game playing couldn’t be an addiction as well,” said Dr. Griffiths.
“The neurobiology of addiction is that we get drawn to something that gives us a dopamine hit,” Dr. Sukhera added. “But to do that all day, every day, has consequences. It drains our emotional reserves, and it can greatly impact our relationships.”
On top of that, work addiction has been linked with poor sleep, poor cardiovascular health, high blood pressure, burnout, the development of autoimmune disorders, and other health issues.
Physicians are particularly susceptible. Doctors, after all, are expected to work long hours and put their patients’ needs first, even at the expense of their own health and well-being.
“Workaholism is not just socially acceptable in medicine,” said Dr. Sukhera. “It’s baked into the system and built into the structures. The healthcare system has largely functioned on the emotional labor of health workers, whose tendency to show up and work harder can, at times, in certain organizations, be exploited.”
Dr. Griffiths agreed that with the limited amount of data available, work addiction does appear to exist at higher rates in medicine than in other fields. As early as the 1970s, medical literature describes work as a “socially acceptable” addiction among doctors. A 2014 study published in Occupational Medicine reported that of 445 physicians who took part in the research, nearly half exhibited some level of work addiction with 13% “highly work addicted.”
Of course, working hard or even meeting unreasonable demands from work is not the same as work addiction, as Dr. Griffiths clarified in a 2023 editorial in BMJ Quality & Safety. The difference, as with other behavioral addictions, is when people obsess about work and use it to cope with stress. It can be easier to stay distracted and busy to gain a sense of control rather than learning to deal with complex emotions.
A 2021 study that Dr. Sukhera conducted with resident physicians found that working harder was one of the main ways they dealt with stress during the COVID-19 pandemic. “This idea that we deal with the stress of being burnt out by doing more and more of what burns us out is fairly ubiquitous at all stages of medical professionals’ careers,” he said.
Financial incentives also can fuel work addiction, said Dr. Sukhera. In residency, there are some safeguards around overwork and duty hours. When you become an attending, those limits no longer exist. As a young physician, Dr. Sukhera had student debt to pay off and a family to support. When he found opportunities to earn more by working more, his answer was always “yes.”
Pressure to produce medical research also can pose issues. Some physicians can become addicted to publishing studies, fearing that they might lose their professional status or position if they stop. It’s a cycle that can force a doctor to not only work long hours doing their job but also practically take on a second one.
How Physicians Can Recognize Work Addiction in Themselves
Work addiction can look and feel different for every person, said Malissa Clark, PhD, associate professor at the University of Georgia and author of the recent book Never Not Working: Why the Always-On Culture Is Bad for Business—and How to Fix It.
Dr. Clark noted that people who are highly engaged in their work tend to be driven by intrinsic motivation: “You work because you love it.” With work addiction, “you work because you feel like you ought to be working all the time.”
Of course, it’s not always so cut and dried; you can experience both forms of motivation and not necessarily become addicted to work. But if you are solely driven by the feeling that you ought to be working all the time, that can be a red flag.
Dr. Griffiths said that while many people may have problematic work habits or work too much, true work addicts must meet six criteria that apply to all addictions:
1. Salience: Work is the single most important thing in your life, to the point of neglecting everything else. Even if you’re on vacation, your mind might be flooded with work thoughts.
2. Mood modification: You use work to modify your mood, either to get a “high” or to cope with stress.
3. Tolerance: Over time, you’ve gone from working 8 or 10 hours a day to 12 hours a day, to a point where you’re working all the time.
4. Withdrawal: On a physiological level, you will have symptoms such as anxiety, nausea, or headaches when unable to work.
5. Conflict: You feel conflicted with yourself (you know you’re working too much) or with others (partners, friends, and children) about work, but you can’t stop.
6. Relapse: If you manage to cut down your hours but can’t resist overworking 1 day, you wind up right back where you were.
When It’s Time to Address Work Addiction
The lack of a formal diagnosis for work addiction makes getting treatment difficult. But there are ways to seek help. Unlike the drug and alcohol literature, abstinence is not the goal. “The therapeutic goal is getting a behavior under control and looking for the triggers of why you’re compulsively working,” said Dr. Griffiths.
Practice self-compassion
Dr. Sukhera eventually realized that his work addiction stemmed from the fear of being somehow excluded or unworthy. He actively corrected much of this through self-compassion and self-kindness, which helped him set boundaries. “Self-compassion is the root of everything,” he said. “Reminding ourselves that we’re doing our best is an important ingredient in breaking the cycle.”
Slowly expose yourself to relaxation
Many workaholics find rest very difficult. “When I conducted interviews with people [who considered themselves workaholics], a very common thing I heard was, ‘I have a very hard time being idle,’ ” said Dr. Clark. If rest feels hard, Dr. Sukhera suggests practicing relaxation for 2 minutes to start. Even small periods of downtime can challenge the belief that you must be constantly productive.
Reframe your to-do list
For work addicts, to-do lists can seem like they must be finished, which prolongs work hours. Instead, use to-do lists to help prioritize what is urgent, identify what can wait, and delegate out tasks to others, Dr. Clark recommends.
Pick up a mastery experience
Research from professor Sabine Sonnentag, Dr. rer. nat., at the University of Mannheim, Mannheim, Germany, suggests that mastery experiences — leisure activities that require thought and focus like learning a new language or taking a woodworking class — can help you actively disengage from work.
Try cognitive behavioral therapy
Widely used for other forms of addiction, cognitive behavioral therapy centers around recognizing emotions, challenging thought patterns, and changing behaviors. However, Dr. Clark admits the research on its impact on work addiction, in particular, is “pretty nascent.”
Shift your mindset
It seems logical to think that detaching from your feelings will allow you to “do more,” but experts say that idea is both untrue and dangerous. “The safest hospitals are the hospitals where people are attuned to their humanness,” said Dr. Sukhera. “It’s normal to overwork in medicine, and if you’re challenging a norm, you really have to be thoughtful about how you frame that for yourself.”
Most importantly: Seek support
Today, there is increased awareness about work addiction and more resources for physicians who are struggling, including programs such as Workaholics Anonymous or Physicians Anonymous and workplace wellness initiatives. But try not to overwhelm yourself with choosing whom to talk to or what specific resource to utilize, Dr. Sukhera advised. “Just talk to someone about it. You don’t have to carry this on your own.”
A version of this article appeared on Medscape.com.
When child psychiatrist Javeed Sukhera, MD, PhD, was a few years into his career, he found himself doing it all. “I was in a leadership role academically at the medical school, I had a leadership role at the hospital, and I was seeing as many patients as I could. I could work all day every day.”
“It still wouldn’t have been enough,” he said.
Whenever there was a shift available, Dr. Sukhera would take it. His job was stressful, but as a new physician with a young family, he saw this obsession with work as necessary. “I began to cope with the stress from work by doing extra work and feeling like I needed to be everywhere. It was like I became a hamster on a spinning wheel. I was just running, running, running.”
Things shifted for Dr. Sukhera when he realized that while he was emotionally available for the children who were his patients, at home, his own children weren’t getting the best of him. “There was a specific moment when I thought my son was afraid of me,” he said. “I just stopped and realized that there was something happening that I needed to break. I needed to make a change.”
Dr. Sukhera, now chair of psychiatry at the Institute of Living and chief of the Department of Psychiatry at Hartford Hospital, Hartford, Connecticut, believes what he experienced was a steep fall into work addiction.
What Does Work Addiction Look Like for Doctors?
Behavioral addictions are fairly new in the addiction space. When gambling disorder, the first and only behavioral addiction in the Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition, was added in 2013, it was seen as a “breakthrough addiction,” said Mark D. Griffiths, PhD, a leading behavioral addiction researcher and a distinguished professor at Nottingham Trent University.
Because there is not enough evidence yet to classify work addiction as a formal diagnosis, there is no clear consensus on how to define it. To further complicate things, the terms “workaholism” and “work addiction” can be used interchangeably, and some experts say the two are not the same, though they can overlap.
That said, a 2018 review of literature from several countries found that work addiction “fits very well into recently postulated criteria for conceptualization of a behavioral addiction.
“If you accept that gambling can be genuinely addictive, then there’s no reason to think that something like work, exercise, or video game playing couldn’t be an addiction as well,” said Dr. Griffiths.
“The neurobiology of addiction is that we get drawn to something that gives us a dopamine hit,” Dr. Sukhera added. “But to do that all day, every day, has consequences. It drains our emotional reserves, and it can greatly impact our relationships.”
On top of that, work addiction has been linked with poor sleep, poor cardiovascular health, high blood pressure, burnout, the development of autoimmune disorders, and other health issues.
Physicians are particularly susceptible. Doctors, after all, are expected to work long hours and put their patients’ needs first, even at the expense of their own health and well-being.
“Workaholism is not just socially acceptable in medicine,” said Dr. Sukhera. “It’s baked into the system and built into the structures. The healthcare system has largely functioned on the emotional labor of health workers, whose tendency to show up and work harder can, at times, in certain organizations, be exploited.”
Dr. Griffiths agreed that with the limited amount of data available, work addiction does appear to exist at higher rates in medicine than in other fields. As early as the 1970s, medical literature describes work as a “socially acceptable” addiction among doctors. A 2014 study published in Occupational Medicine reported that of 445 physicians who took part in the research, nearly half exhibited some level of work addiction with 13% “highly work addicted.”
Of course, working hard or even meeting unreasonable demands from work is not the same as work addiction, as Dr. Griffiths clarified in a 2023 editorial in BMJ Quality & Safety. The difference, as with other behavioral addictions, is when people obsess about work and use it to cope with stress. It can be easier to stay distracted and busy to gain a sense of control rather than learning to deal with complex emotions.
A 2021 study that Dr. Sukhera conducted with resident physicians found that working harder was one of the main ways they dealt with stress during the COVID-19 pandemic. “This idea that we deal with the stress of being burnt out by doing more and more of what burns us out is fairly ubiquitous at all stages of medical professionals’ careers,” he said.
Financial incentives also can fuel work addiction, said Dr. Sukhera. In residency, there are some safeguards around overwork and duty hours. When you become an attending, those limits no longer exist. As a young physician, Dr. Sukhera had student debt to pay off and a family to support. When he found opportunities to earn more by working more, his answer was always “yes.”
Pressure to produce medical research also can pose issues. Some physicians can become addicted to publishing studies, fearing that they might lose their professional status or position if they stop. It’s a cycle that can force a doctor to not only work long hours doing their job but also practically take on a second one.
How Physicians Can Recognize Work Addiction in Themselves
Work addiction can look and feel different for every person, said Malissa Clark, PhD, associate professor at the University of Georgia and author of the recent book Never Not Working: Why the Always-On Culture Is Bad for Business—and How to Fix It.
Dr. Clark noted that people who are highly engaged in their work tend to be driven by intrinsic motivation: “You work because you love it.” With work addiction, “you work because you feel like you ought to be working all the time.”
Of course, it’s not always so cut and dried; you can experience both forms of motivation and not necessarily become addicted to work. But if you are solely driven by the feeling that you ought to be working all the time, that can be a red flag.
Dr. Griffiths said that while many people may have problematic work habits or work too much, true work addicts must meet six criteria that apply to all addictions:
1. Salience: Work is the single most important thing in your life, to the point of neglecting everything else. Even if you’re on vacation, your mind might be flooded with work thoughts.
2. Mood modification: You use work to modify your mood, either to get a “high” or to cope with stress.
3. Tolerance: Over time, you’ve gone from working 8 or 10 hours a day to 12 hours a day, to a point where you’re working all the time.
4. Withdrawal: On a physiological level, you will have symptoms such as anxiety, nausea, or headaches when unable to work.
5. Conflict: You feel conflicted with yourself (you know you’re working too much) or with others (partners, friends, and children) about work, but you can’t stop.
6. Relapse: If you manage to cut down your hours but can’t resist overworking 1 day, you wind up right back where you were.
When It’s Time to Address Work Addiction
The lack of a formal diagnosis for work addiction makes getting treatment difficult. But there are ways to seek help. Unlike the drug and alcohol literature, abstinence is not the goal. “The therapeutic goal is getting a behavior under control and looking for the triggers of why you’re compulsively working,” said Dr. Griffiths.
Practice self-compassion
Dr. Sukhera eventually realized that his work addiction stemmed from the fear of being somehow excluded or unworthy. He actively corrected much of this through self-compassion and self-kindness, which helped him set boundaries. “Self-compassion is the root of everything,” he said. “Reminding ourselves that we’re doing our best is an important ingredient in breaking the cycle.”
Slowly expose yourself to relaxation
Many workaholics find rest very difficult. “When I conducted interviews with people [who considered themselves workaholics], a very common thing I heard was, ‘I have a very hard time being idle,’ ” said Dr. Clark. If rest feels hard, Dr. Sukhera suggests practicing relaxation for 2 minutes to start. Even small periods of downtime can challenge the belief that you must be constantly productive.
Reframe your to-do list
For work addicts, to-do lists can seem like they must be finished, which prolongs work hours. Instead, use to-do lists to help prioritize what is urgent, identify what can wait, and delegate out tasks to others, Dr. Clark recommends.
Pick up a mastery experience
Research from professor Sabine Sonnentag, Dr. rer. nat., at the University of Mannheim, Mannheim, Germany, suggests that mastery experiences — leisure activities that require thought and focus like learning a new language or taking a woodworking class — can help you actively disengage from work.
Try cognitive behavioral therapy
Widely used for other forms of addiction, cognitive behavioral therapy centers around recognizing emotions, challenging thought patterns, and changing behaviors. However, Dr. Clark admits the research on its impact on work addiction, in particular, is “pretty nascent.”
Shift your mindset
It seems logical to think that detaching from your feelings will allow you to “do more,” but experts say that idea is both untrue and dangerous. “The safest hospitals are the hospitals where people are attuned to their humanness,” said Dr. Sukhera. “It’s normal to overwork in medicine, and if you’re challenging a norm, you really have to be thoughtful about how you frame that for yourself.”
Most importantly: Seek support
Today, there is increased awareness about work addiction and more resources for physicians who are struggling, including programs such as Workaholics Anonymous or Physicians Anonymous and workplace wellness initiatives. But try not to overwhelm yourself with choosing whom to talk to or what specific resource to utilize, Dr. Sukhera advised. “Just talk to someone about it. You don’t have to carry this on your own.”
A version of this article appeared on Medscape.com.
Oregon Physician Assistants Get Name Change
On April 4, Oregon’s Governor Tina Kotek signed a bill into law that officially changed the title of “physician assistants” to “physician associates” in the state.
In the Medscape Physician Assistant Career Satisfaction Report 2023, a diverse range of opinions on the title switch was reflected. Only 40% of PAs favored the name change at the time, 45% neither opposed nor favored it, and 15% opposed the name change, reflecting the complexity of the issue.
According to the AAPA, the change came about to better reflect the work PAs do in not just “assisting” physicians but in working independently with patients. Some also felt that the word “assistant” implies dependence. However, despite associate’s more accurate reflection of the job, PAs mostly remain split on whether they want the new moniker.
Many say that the name change will be confusing for the public and their patients, while others say that physician assistant was already not well understood, as patients often thought of the profession as a doctor’s helper or an assistant, like a medical assistant.
Yet many long-time PAs say that they prefer the title they’ve always had and that explaining to patients the new associate title will be equally confusing. Some mentioned patients may think they’re a business associate of the physician.
Oregon PAs won’t immediately switch to the new name. The new law takes effect on June 6, 2024. The Oregon Medical Board will establish regulations and guidance before PAs adopt the new name in their practices.
The law only changes the name of PAs in Oregon, not in other states. In fact, prematurely using the title of physician associate could subject a PA to regulatory challenges or disciplinary actions.
A version of this article appeared on Medscape.com.
On April 4, Oregon’s Governor Tina Kotek signed a bill into law that officially changed the title of “physician assistants” to “physician associates” in the state.
In the Medscape Physician Assistant Career Satisfaction Report 2023, a diverse range of opinions on the title switch was reflected. Only 40% of PAs favored the name change at the time, 45% neither opposed nor favored it, and 15% opposed the name change, reflecting the complexity of the issue.
According to the AAPA, the change came about to better reflect the work PAs do in not just “assisting” physicians but in working independently with patients. Some also felt that the word “assistant” implies dependence. However, despite associate’s more accurate reflection of the job, PAs mostly remain split on whether they want the new moniker.
Many say that the name change will be confusing for the public and their patients, while others say that physician assistant was already not well understood, as patients often thought of the profession as a doctor’s helper or an assistant, like a medical assistant.
Yet many long-time PAs say that they prefer the title they’ve always had and that explaining to patients the new associate title will be equally confusing. Some mentioned patients may think they’re a business associate of the physician.
Oregon PAs won’t immediately switch to the new name. The new law takes effect on June 6, 2024. The Oregon Medical Board will establish regulations and guidance before PAs adopt the new name in their practices.
The law only changes the name of PAs in Oregon, not in other states. In fact, prematurely using the title of physician associate could subject a PA to regulatory challenges or disciplinary actions.
A version of this article appeared on Medscape.com.
On April 4, Oregon’s Governor Tina Kotek signed a bill into law that officially changed the title of “physician assistants” to “physician associates” in the state.
In the Medscape Physician Assistant Career Satisfaction Report 2023, a diverse range of opinions on the title switch was reflected. Only 40% of PAs favored the name change at the time, 45% neither opposed nor favored it, and 15% opposed the name change, reflecting the complexity of the issue.
According to the AAPA, the change came about to better reflect the work PAs do in not just “assisting” physicians but in working independently with patients. Some also felt that the word “assistant” implies dependence. However, despite associate’s more accurate reflection of the job, PAs mostly remain split on whether they want the new moniker.
Many say that the name change will be confusing for the public and their patients, while others say that physician assistant was already not well understood, as patients often thought of the profession as a doctor’s helper or an assistant, like a medical assistant.
Yet many long-time PAs say that they prefer the title they’ve always had and that explaining to patients the new associate title will be equally confusing. Some mentioned patients may think they’re a business associate of the physician.
Oregon PAs won’t immediately switch to the new name. The new law takes effect on June 6, 2024. The Oregon Medical Board will establish regulations and guidance before PAs adopt the new name in their practices.
The law only changes the name of PAs in Oregon, not in other states. In fact, prematurely using the title of physician associate could subject a PA to regulatory challenges or disciplinary actions.
A version of this article appeared on Medscape.com.
The History of Aspirin in Heart Disease Prevention
As the pendulum has swung against recommending aspirin for the primary prevention of heart attacks and strokes, clinicians should focus on other ways to help patients avoid cardiovascular events.
A landmark study published in 1988 in The New England Journal of Medicine reported an astonishing 44% drop in the number of heart attacks among US male physicians aged 40-84 years who took aspirin.
Aspirin subsequently became a daily habit for millions of Americans. In 2017, nearly a quarter of Americans over age 40 who did not have cardiovascular disease (CVD) took the drug, and over 20% of those were doing so without a physician’s recommendation.
But in 2018, three studies (ASCEND, ARRIVE, and ASPREE) showed a stunning reversal in the purported benefit, according to John Wong, MD, vice-chair of the US Preventive Services Task Force (USPSTF).
The calculus for taking aspirin appeared to have changed dramatically: The drug decreased the risk for myocardial infarction by only 11% among study subjects, while its potential harms were much more pronounced.
According to Dr. Wong, who is also a professor of medicine and a primary care physician at Tufts University School of Medicine in Boston, Massachusetts, patients taking low-dose aspirin had a 58% increase in their risk for gastrointestinal bleeding compared with those not on aspirin, as well as a 31% increased risk for intracranial bleeding.
who haven’t had a heart attack or stroke.”
Fewer Americans smoke cigarettes, more realize the benefits of a healthy diet and physical activity, and the medical community better recognizes and treats hypertension. New classes of medications such as statins for high cholesterol are also moving the needle.
But a newer class of drugs may provide a safer replacement for aspirin, according to Muhammad Maqsood, MD, a cardiology fellow at DeBakey Heart and Vascular Center at Methodist Hospital in Houston, Texas. P2Y purinoceptor 12 (P2Y12) inhibitors are effective in lowering the risk for heart attack and stroke in patients with acute coronary syndrome or those undergoing elective percutaneous coronary interventions.
“They have shown a better bleeding profile, especially clopidogrel compared to aspirin,” Dr. Maqsood said.
However, the findings come from trials of patients who already had CVD, so results cannot yet be extrapolated to primary prevention. Dr. Maqsood said the gap highlights the need for clinical trials that evaluate P2Y12 inhibitors for primary prevention, but no such study is registered on clinicaltrials.gov.
Benefits Persist for Some Patients
The new evidence led the USPSTF to publish new guidelines in 2022, downgrading the recommendation for low-dose aspirin use for primary prevention. Previously, the organization stated that clinicians “should” initiate daily low-dose aspirin in adults aged 50-59 years and “consider” its use in adults aged 60-69 years whose 10-year risk for CVD was higher than 10%.
The updated guidelines stated that the decision to initiate low-dose aspirin in adults aged 40-59 years with a greater than 10% risk for CVD “should be an individual one,” based on professional judgment and individual patient preferences. The USPSTF also recommended against the use of aspirin in anyone over the age of 60.
Meanwhile, the American College of Cardiology and American Heart Association also dialed down previously strong recommendations on low-dose aspirin to a more nuanced recommendation stating, “low-dose aspirin might be considered for primary prevention of ASCVD among select adults 40-70 years of age.”
With a varying age limit for recommending aspirin, clinicians may take into consideration several variables.
“Is there a magic age? I don’t think there is,” said Douglas Lloyd-Jones, the former president of the American Heart Association and current chair of the Department of Preventive Medicine and a practicing cardiologist at Northwestern University Feinberg School of Medicine in Chicago, Illinois.
For a patient over age 60 who is at a high risk for adverse cardiovascular outcomes, is unable to quit smoking, and is not likely to experience problematic bleeding, a clinician might recommend aspirin, Dr. Lloyd-Jones said. He said he sometimes also assesses coronary artery calcium to guide his clinical decisions: If elevated (an Agatston score above 100), he might recommend low-dose aspirin.
Dr. Lloyd-Jones also reiterated that patients should continue taking low-dose aspirin if they have already experienced a heart attack, stroke, episode of atrial fibrillation, or required a vascular stent.
Unless a patient with established CVD has intractable bleeding, “the aspirin is really for life,” Dr. Lloyd-Jones said. Patients who have a stent or who are at high risk for recurrence of stroke are more likely to experience thrombosis, and aspirin can decrease the risk.
“In our cardiology community, we don’t just strictly use the age of 70; the decision is always individualized,” Dr. Maqsood said.
Dr. Wong said primary care providers should focus on the USPSTF’s other recommendations that address CVD (Table), such as smoking cessation and screening for hypertension.
“I think our challenge is that we have so many of those A and B recommendations,” Dr. Wong said. “And I think part of the challenge for us is working with the patient to find out what’s most important to them.”
Discussing heart attacks and strokes often will strike a chord with patients because someone they know has been affected.
Dr. Maqsood emphasized the importance of behavioral interventions, such as helping patients decrease their body mass index and control their hyperlipidemia.
“The behavioral interventions are those which are the most cost-effective without any side effects,” he said.
His other piece of advice is to inquire with younger patients about a family history of heart attacks. Familial hypercholesteremia is unlikely to be controlled by diet and exercise and will need medical therapy.
Dr. Lloyd-Jones described the discussions he has with patients about preventing heart attacks as “the most important conversations we can have: Remember that cardiovascular disease is still the leading cause of death and disability in the world and in the United States.”
Dr. Wong, Dr. Lloyd-Jones, and Dr. Maqsood reported no relevant financial relationships.
A version of this article appeared on Medscape.com.
As the pendulum has swung against recommending aspirin for the primary prevention of heart attacks and strokes, clinicians should focus on other ways to help patients avoid cardiovascular events.
A landmark study published in 1988 in The New England Journal of Medicine reported an astonishing 44% drop in the number of heart attacks among US male physicians aged 40-84 years who took aspirin.
Aspirin subsequently became a daily habit for millions of Americans. In 2017, nearly a quarter of Americans over age 40 who did not have cardiovascular disease (CVD) took the drug, and over 20% of those were doing so without a physician’s recommendation.
But in 2018, three studies (ASCEND, ARRIVE, and ASPREE) showed a stunning reversal in the purported benefit, according to John Wong, MD, vice-chair of the US Preventive Services Task Force (USPSTF).
The calculus for taking aspirin appeared to have changed dramatically: The drug decreased the risk for myocardial infarction by only 11% among study subjects, while its potential harms were much more pronounced.
According to Dr. Wong, who is also a professor of medicine and a primary care physician at Tufts University School of Medicine in Boston, Massachusetts, patients taking low-dose aspirin had a 58% increase in their risk for gastrointestinal bleeding compared with those not on aspirin, as well as a 31% increased risk for intracranial bleeding.
who haven’t had a heart attack or stroke.”
Fewer Americans smoke cigarettes, more realize the benefits of a healthy diet and physical activity, and the medical community better recognizes and treats hypertension. New classes of medications such as statins for high cholesterol are also moving the needle.
But a newer class of drugs may provide a safer replacement for aspirin, according to Muhammad Maqsood, MD, a cardiology fellow at DeBakey Heart and Vascular Center at Methodist Hospital in Houston, Texas. P2Y purinoceptor 12 (P2Y12) inhibitors are effective in lowering the risk for heart attack and stroke in patients with acute coronary syndrome or those undergoing elective percutaneous coronary interventions.
“They have shown a better bleeding profile, especially clopidogrel compared to aspirin,” Dr. Maqsood said.
However, the findings come from trials of patients who already had CVD, so results cannot yet be extrapolated to primary prevention. Dr. Maqsood said the gap highlights the need for clinical trials that evaluate P2Y12 inhibitors for primary prevention, but no such study is registered on clinicaltrials.gov.
Benefits Persist for Some Patients
The new evidence led the USPSTF to publish new guidelines in 2022, downgrading the recommendation for low-dose aspirin use for primary prevention. Previously, the organization stated that clinicians “should” initiate daily low-dose aspirin in adults aged 50-59 years and “consider” its use in adults aged 60-69 years whose 10-year risk for CVD was higher than 10%.
The updated guidelines stated that the decision to initiate low-dose aspirin in adults aged 40-59 years with a greater than 10% risk for CVD “should be an individual one,” based on professional judgment and individual patient preferences. The USPSTF also recommended against the use of aspirin in anyone over the age of 60.
Meanwhile, the American College of Cardiology and American Heart Association also dialed down previously strong recommendations on low-dose aspirin to a more nuanced recommendation stating, “low-dose aspirin might be considered for primary prevention of ASCVD among select adults 40-70 years of age.”
With a varying age limit for recommending aspirin, clinicians may take into consideration several variables.
“Is there a magic age? I don’t think there is,” said Douglas Lloyd-Jones, the former president of the American Heart Association and current chair of the Department of Preventive Medicine and a practicing cardiologist at Northwestern University Feinberg School of Medicine in Chicago, Illinois.
For a patient over age 60 who is at a high risk for adverse cardiovascular outcomes, is unable to quit smoking, and is not likely to experience problematic bleeding, a clinician might recommend aspirin, Dr. Lloyd-Jones said. He said he sometimes also assesses coronary artery calcium to guide his clinical decisions: If elevated (an Agatston score above 100), he might recommend low-dose aspirin.
Dr. Lloyd-Jones also reiterated that patients should continue taking low-dose aspirin if they have already experienced a heart attack, stroke, episode of atrial fibrillation, or required a vascular stent.
Unless a patient with established CVD has intractable bleeding, “the aspirin is really for life,” Dr. Lloyd-Jones said. Patients who have a stent or who are at high risk for recurrence of stroke are more likely to experience thrombosis, and aspirin can decrease the risk.
“In our cardiology community, we don’t just strictly use the age of 70; the decision is always individualized,” Dr. Maqsood said.
Dr. Wong said primary care providers should focus on the USPSTF’s other recommendations that address CVD (Table), such as smoking cessation and screening for hypertension.
“I think our challenge is that we have so many of those A and B recommendations,” Dr. Wong said. “And I think part of the challenge for us is working with the patient to find out what’s most important to them.”
Discussing heart attacks and strokes often will strike a chord with patients because someone they know has been affected.
Dr. Maqsood emphasized the importance of behavioral interventions, such as helping patients decrease their body mass index and control their hyperlipidemia.
“The behavioral interventions are those which are the most cost-effective without any side effects,” he said.
His other piece of advice is to inquire with younger patients about a family history of heart attacks. Familial hypercholesteremia is unlikely to be controlled by diet and exercise and will need medical therapy.
Dr. Lloyd-Jones described the discussions he has with patients about preventing heart attacks as “the most important conversations we can have: Remember that cardiovascular disease is still the leading cause of death and disability in the world and in the United States.”
Dr. Wong, Dr. Lloyd-Jones, and Dr. Maqsood reported no relevant financial relationships.
A version of this article appeared on Medscape.com.
As the pendulum has swung against recommending aspirin for the primary prevention of heart attacks and strokes, clinicians should focus on other ways to help patients avoid cardiovascular events.
A landmark study published in 1988 in The New England Journal of Medicine reported an astonishing 44% drop in the number of heart attacks among US male physicians aged 40-84 years who took aspirin.
Aspirin subsequently became a daily habit for millions of Americans. In 2017, nearly a quarter of Americans over age 40 who did not have cardiovascular disease (CVD) took the drug, and over 20% of those were doing so without a physician’s recommendation.
But in 2018, three studies (ASCEND, ARRIVE, and ASPREE) showed a stunning reversal in the purported benefit, according to John Wong, MD, vice-chair of the US Preventive Services Task Force (USPSTF).
The calculus for taking aspirin appeared to have changed dramatically: The drug decreased the risk for myocardial infarction by only 11% among study subjects, while its potential harms were much more pronounced.
According to Dr. Wong, who is also a professor of medicine and a primary care physician at Tufts University School of Medicine in Boston, Massachusetts, patients taking low-dose aspirin had a 58% increase in their risk for gastrointestinal bleeding compared with those not on aspirin, as well as a 31% increased risk for intracranial bleeding.
who haven’t had a heart attack or stroke.”
Fewer Americans smoke cigarettes, more realize the benefits of a healthy diet and physical activity, and the medical community better recognizes and treats hypertension. New classes of medications such as statins for high cholesterol are also moving the needle.
But a newer class of drugs may provide a safer replacement for aspirin, according to Muhammad Maqsood, MD, a cardiology fellow at DeBakey Heart and Vascular Center at Methodist Hospital in Houston, Texas. P2Y purinoceptor 12 (P2Y12) inhibitors are effective in lowering the risk for heart attack and stroke in patients with acute coronary syndrome or those undergoing elective percutaneous coronary interventions.
“They have shown a better bleeding profile, especially clopidogrel compared to aspirin,” Dr. Maqsood said.
However, the findings come from trials of patients who already had CVD, so results cannot yet be extrapolated to primary prevention. Dr. Maqsood said the gap highlights the need for clinical trials that evaluate P2Y12 inhibitors for primary prevention, but no such study is registered on clinicaltrials.gov.
Benefits Persist for Some Patients
The new evidence led the USPSTF to publish new guidelines in 2022, downgrading the recommendation for low-dose aspirin use for primary prevention. Previously, the organization stated that clinicians “should” initiate daily low-dose aspirin in adults aged 50-59 years and “consider” its use in adults aged 60-69 years whose 10-year risk for CVD was higher than 10%.
The updated guidelines stated that the decision to initiate low-dose aspirin in adults aged 40-59 years with a greater than 10% risk for CVD “should be an individual one,” based on professional judgment and individual patient preferences. The USPSTF also recommended against the use of aspirin in anyone over the age of 60.
Meanwhile, the American College of Cardiology and American Heart Association also dialed down previously strong recommendations on low-dose aspirin to a more nuanced recommendation stating, “low-dose aspirin might be considered for primary prevention of ASCVD among select adults 40-70 years of age.”
With a varying age limit for recommending aspirin, clinicians may take into consideration several variables.
“Is there a magic age? I don’t think there is,” said Douglas Lloyd-Jones, the former president of the American Heart Association and current chair of the Department of Preventive Medicine and a practicing cardiologist at Northwestern University Feinberg School of Medicine in Chicago, Illinois.
For a patient over age 60 who is at a high risk for adverse cardiovascular outcomes, is unable to quit smoking, and is not likely to experience problematic bleeding, a clinician might recommend aspirin, Dr. Lloyd-Jones said. He said he sometimes also assesses coronary artery calcium to guide his clinical decisions: If elevated (an Agatston score above 100), he might recommend low-dose aspirin.
Dr. Lloyd-Jones also reiterated that patients should continue taking low-dose aspirin if they have already experienced a heart attack, stroke, episode of atrial fibrillation, or required a vascular stent.
Unless a patient with established CVD has intractable bleeding, “the aspirin is really for life,” Dr. Lloyd-Jones said. Patients who have a stent or who are at high risk for recurrence of stroke are more likely to experience thrombosis, and aspirin can decrease the risk.
“In our cardiology community, we don’t just strictly use the age of 70; the decision is always individualized,” Dr. Maqsood said.
Dr. Wong said primary care providers should focus on the USPSTF’s other recommendations that address CVD (Table), such as smoking cessation and screening for hypertension.
“I think our challenge is that we have so many of those A and B recommendations,” Dr. Wong said. “And I think part of the challenge for us is working with the patient to find out what’s most important to them.”
Discussing heart attacks and strokes often will strike a chord with patients because someone they know has been affected.
Dr. Maqsood emphasized the importance of behavioral interventions, such as helping patients decrease their body mass index and control their hyperlipidemia.
“The behavioral interventions are those which are the most cost-effective without any side effects,” he said.
His other piece of advice is to inquire with younger patients about a family history of heart attacks. Familial hypercholesteremia is unlikely to be controlled by diet and exercise and will need medical therapy.
Dr. Lloyd-Jones described the discussions he has with patients about preventing heart attacks as “the most important conversations we can have: Remember that cardiovascular disease is still the leading cause of death and disability in the world and in the United States.”
Dr. Wong, Dr. Lloyd-Jones, and Dr. Maqsood reported no relevant financial relationships.
A version of this article appeared on Medscape.com.
Evening May Be the Best Time for Exercise
TOPLINE:
Moderate to vigorous aerobic physical activity performed in the evening is associated with the lowest risk for mortality, cardiovascular disease (CVD), and microvascular disease (MVD) in adults with obesity, including those with type 2 diabetes (T2D).
METHODOLOGY:
- Bouts of moderate to vigorous aerobic physical activity are widely recognized to improve cardiometabolic risk factors, but whether morning, afternoon, or evening timing may lead to greater improvements is unclear.
- Researchers analyzed UK Biobank data of 29,836 participants with obesity (body mass index, › 30; mean age, 62.2 years; 53.2% women), including 2995 also diagnosed with T2D, all enrolled in 2006-2010.
- Aerobic activity was defined as bouts lasting ≥ 3 minutes, and the intensity of activity was classified as light, moderate, or vigorous using accelerometer data collected from participants.
- Participants were stratified into the morning (6 a.m. to < 12 p.m.), afternoon (12 p.m. to < 6 p.m.), and evening (6 p.m. to < 12 a.m.) groups based on when > 50% of their total moderate to vigorous activity occurred, and those with no aerobic bouts were considered the reference group.
- The association between the timing of aerobic physical activity and risk for all-cause mortality, CVD (defined as circulatory, such as hypertension), and MVD (neuropathy, nephropathy, or retinopathy) was evaluated over a median follow-up of 7.9 years.
TAKEAWAY:
- Mortality risk was lower in the afternoon (HR, 0.60; 95% CI, 0.51-0.71) and morning (HR, 0.67; 95% CI, 0.56-0.79) activity groups than in the reference group, but this association was weaker than that observed in the evening activity group.
- The evening moderate to vigorous activity group had a lower risk for CVD (HR, 0.64; 95% CI, 0.54-0.75) and MVD (HR, 0.76; 95% CI, 0.63-0.92) than the reference group.
- Among participants with obesity and T2D, moderate to vigorous physical activity in the evening was associated with a lower risk for mortality, CVD, and MVD.
IN PRACTICE:
The authors wrote, “The results of this study emphasize that beyond the total volume of MVPA [moderate to vigorous physical activity], its timing, particularly in the evening, was consistently associated with the lowest risk of mortality relative to other timing windows.”
SOURCE:
The study, led by Angelo Sabag, PhD, Charles Perkins Centre, University of Sydney, Australia, was published online in Diabetes Care.
LIMITATIONS:
Because this was an observational study, the possibility of reverse causation from prodromal disease and unaccounted confounding factors could not have been ruled out. There was a lag of a median of 5.5 years between the UK Biobank baseline, when covariate measurements were taken, and the accelerometry study. Moreover, the response rate of the UK Biobank was low.
DISCLOSURES:
The study was funded by an Australian National Health and Medical Research Council Investigator Grant and the National Heart Foundation of Australia Postdoctoral Fellowship. The authors reported no conflicts of interest.
A version of this article appeared on Medscape.com.
TOPLINE:
Moderate to vigorous aerobic physical activity performed in the evening is associated with the lowest risk for mortality, cardiovascular disease (CVD), and microvascular disease (MVD) in adults with obesity, including those with type 2 diabetes (T2D).
METHODOLOGY:
- Bouts of moderate to vigorous aerobic physical activity are widely recognized to improve cardiometabolic risk factors, but whether morning, afternoon, or evening timing may lead to greater improvements is unclear.
- Researchers analyzed UK Biobank data of 29,836 participants with obesity (body mass index, › 30; mean age, 62.2 years; 53.2% women), including 2995 also diagnosed with T2D, all enrolled in 2006-2010.
- Aerobic activity was defined as bouts lasting ≥ 3 minutes, and the intensity of activity was classified as light, moderate, or vigorous using accelerometer data collected from participants.
- Participants were stratified into the morning (6 a.m. to < 12 p.m.), afternoon (12 p.m. to < 6 p.m.), and evening (6 p.m. to < 12 a.m.) groups based on when > 50% of their total moderate to vigorous activity occurred, and those with no aerobic bouts were considered the reference group.
- The association between the timing of aerobic physical activity and risk for all-cause mortality, CVD (defined as circulatory, such as hypertension), and MVD (neuropathy, nephropathy, or retinopathy) was evaluated over a median follow-up of 7.9 years.
TAKEAWAY:
- Mortality risk was lower in the afternoon (HR, 0.60; 95% CI, 0.51-0.71) and morning (HR, 0.67; 95% CI, 0.56-0.79) activity groups than in the reference group, but this association was weaker than that observed in the evening activity group.
- The evening moderate to vigorous activity group had a lower risk for CVD (HR, 0.64; 95% CI, 0.54-0.75) and MVD (HR, 0.76; 95% CI, 0.63-0.92) than the reference group.
- Among participants with obesity and T2D, moderate to vigorous physical activity in the evening was associated with a lower risk for mortality, CVD, and MVD.
IN PRACTICE:
The authors wrote, “The results of this study emphasize that beyond the total volume of MVPA [moderate to vigorous physical activity], its timing, particularly in the evening, was consistently associated with the lowest risk of mortality relative to other timing windows.”
SOURCE:
The study, led by Angelo Sabag, PhD, Charles Perkins Centre, University of Sydney, Australia, was published online in Diabetes Care.
LIMITATIONS:
Because this was an observational study, the possibility of reverse causation from prodromal disease and unaccounted confounding factors could not have been ruled out. There was a lag of a median of 5.5 years between the UK Biobank baseline, when covariate measurements were taken, and the accelerometry study. Moreover, the response rate of the UK Biobank was low.
DISCLOSURES:
The study was funded by an Australian National Health and Medical Research Council Investigator Grant and the National Heart Foundation of Australia Postdoctoral Fellowship. The authors reported no conflicts of interest.
A version of this article appeared on Medscape.com.
TOPLINE:
Moderate to vigorous aerobic physical activity performed in the evening is associated with the lowest risk for mortality, cardiovascular disease (CVD), and microvascular disease (MVD) in adults with obesity, including those with type 2 diabetes (T2D).
METHODOLOGY:
- Bouts of moderate to vigorous aerobic physical activity are widely recognized to improve cardiometabolic risk factors, but whether morning, afternoon, or evening timing may lead to greater improvements is unclear.
- Researchers analyzed UK Biobank data of 29,836 participants with obesity (body mass index, › 30; mean age, 62.2 years; 53.2% women), including 2995 also diagnosed with T2D, all enrolled in 2006-2010.
- Aerobic activity was defined as bouts lasting ≥ 3 minutes, and the intensity of activity was classified as light, moderate, or vigorous using accelerometer data collected from participants.
- Participants were stratified into the morning (6 a.m. to < 12 p.m.), afternoon (12 p.m. to < 6 p.m.), and evening (6 p.m. to < 12 a.m.) groups based on when > 50% of their total moderate to vigorous activity occurred, and those with no aerobic bouts were considered the reference group.
- The association between the timing of aerobic physical activity and risk for all-cause mortality, CVD (defined as circulatory, such as hypertension), and MVD (neuropathy, nephropathy, or retinopathy) was evaluated over a median follow-up of 7.9 years.
TAKEAWAY:
- Mortality risk was lower in the afternoon (HR, 0.60; 95% CI, 0.51-0.71) and morning (HR, 0.67; 95% CI, 0.56-0.79) activity groups than in the reference group, but this association was weaker than that observed in the evening activity group.
- The evening moderate to vigorous activity group had a lower risk for CVD (HR, 0.64; 95% CI, 0.54-0.75) and MVD (HR, 0.76; 95% CI, 0.63-0.92) than the reference group.
- Among participants with obesity and T2D, moderate to vigorous physical activity in the evening was associated with a lower risk for mortality, CVD, and MVD.
IN PRACTICE:
The authors wrote, “The results of this study emphasize that beyond the total volume of MVPA [moderate to vigorous physical activity], its timing, particularly in the evening, was consistently associated with the lowest risk of mortality relative to other timing windows.”
SOURCE:
The study, led by Angelo Sabag, PhD, Charles Perkins Centre, University of Sydney, Australia, was published online in Diabetes Care.
LIMITATIONS:
Because this was an observational study, the possibility of reverse causation from prodromal disease and unaccounted confounding factors could not have been ruled out. There was a lag of a median of 5.5 years between the UK Biobank baseline, when covariate measurements were taken, and the accelerometry study. Moreover, the response rate of the UK Biobank was low.
DISCLOSURES:
The study was funded by an Australian National Health and Medical Research Council Investigator Grant and the National Heart Foundation of Australia Postdoctoral Fellowship. The authors reported no conflicts of interest.
A version of this article appeared on Medscape.com.
How These Young MDs Impressed the Hell Out of Their Bosses
Safe to say that anyone undertaking the physician journey does so with intense motivation and book smarts. Still, it can be incredibly hard to stand out. Everyone’s a go-getter, but what’s the X factor?
Here’s what they said ...
Lesson #1: Never Be Scared to Ask
Brien Barnewolt, MD, chairman and chief of the Department of Emergency Medicine at Tufts Medical Center, was very much surprised when a resident named Scott G. Weiner did something unexpected: Go after a job in the fall of his junior year residency instead of following the typical senior year trajectory.
“It’s very unusual for a trainee to apply for a job virtually a year ahead of schedule. But he knew what he wanted,” said Dr. Barnewolt. “I’d never had anybody come to me in that same scenario, and I’ve been doing this a long time.”
Under normal circumstances it would’ve been easy for Dr. Barnewolt to say no. But the unexpected request made him and his colleagues take a closer look, and they were impressed with Dr. Weiner’s skills. That, paired with his ambition and demeanor, compelled them to offer him an early job. But there’s more.
As the next year approached, Dr. Weiner explained he had an opportunity to work in emergency medicine in Tuscany and asked if he could take a 1-year delayed start for the position he applied a year early for.
The department held his position, and upon his return, Dr. Weiner made a lasting impact at Tufts before eventually moving on. “He outgrew us, which is nice to see,” Dr. Barnewolt said. (Dr. Weiner is currently McGraw Distinguished Chair in Emergency Medicine at Brigham and Women’s Hospital and associate professor at Harvard Medical School.)
Bottom line: Why did Dr. Barnewolt and his colleagues do so much to accommodate a young candidate? Yes, Dr. Weiner was talented, but he was also up-front about his ambitions from the get-go. Dr. Barnewolt said that kind of initiative can only be looked at positively.
“My advice would be, if you see an opportunity or a potential place where you might want to work, put out those feelers, start those conversations,” he said. “It’s not too early, especially in certain specialties, where the job market is very tight. Then, when circumstances change, be open about it and have that conversation. The worst that somebody can say is no, so it never hurts to be honest and open about where you want to go and what you want to be.”
Lesson #2: Chase Your Passion ‘Relentlessly’
Vance G. Fowler, MD, MHS, an infectious disease specialist at Duke University School of Medicine, runs a laboratory that researches methicillin-resistant Staphylococcus aureus (MRSA). Over the years, he’s mentored many doctors but understands the ambitions of young trainees don’t always align with the little free time that they have. “Many of them drop away when you give them a [side] project,” he said.
So when Tori Kinamon asked him to work on an MRSA project — in her first year — he gave her one that focused on researching vertebral osteomyelitis, a bone infection that can coincide with S aureus. What Dr. Fowler didn’t know: Kinamon (now MD) had been a competitive gymnast at Brown and battled her own life-threatening infection with MRSA.
“To my absolute astonishment, not only did she stick to it, but she was able to compile a presentation on the science and gave an oral presentation within a year of walking in the door,” said Dr. Fowler.
She went on to lead an initiative between the National Institutes of Health and US Food and Drug Administration to create endpoints for clinical drug trials, all of which occurred before starting her residency, which she’s about to embark upon.
Dr. Kinamon’s a good example, he said, of what happens when you add genuine passion to book smarts. Those who do always stand out because you can’t fake that. “Find your passion, and then chase it down relentlessly,” he said. “Once you’ve found your passion, things get easy because it stops being work and it starts being something else.”
If you haven’t identified a focus area, Dr. Fowler said to “be agnostic and observant. Keep your eyes open and your options open because you may surprise yourself. It may turn out that you end up liking something a whole lot more than you thought you did.”
Lesson #3: When You Say You’ve Always Wanted to Do Something, Do Something
As the chief of pulmonary and critical care medicine at the Northwestern Medicine Canning Thoracic Institute, Scott Budinger, MD, often hears lip service from doctors who want to put their skills to use in their local communities. One of his students actually did it.
Justin Fiala, MD, a pulmonary, critical care, and sleep specialist at Northwestern Medicine, joined Northwestern as a pulmonary fellow with a big interest in addressing health equity issues.
Dr. Fiala began volunteering with CommunityHealth during his fellowship and saw that many patients of the free Chicago-area clinic needed help with sleep disorders. He launched the organization’s first sleep clinic and its Patient-Centered Apnea Protocols Initiative.
“He developed a plan with some of the partners of the sleep apnea equipment to do home sleep testing for these patients that’s free of cost,” said Dr. Budinger.
Dr. Fiala goes in on Saturdays and runs a free clinic conducting sleep studies for patients and outfits them with devices that they need to improve their conditions, said Dr. Budinger.
“And these patients are the severest of the severe patients,” he said. “These are people that have severe sleep apnea that are driving around the roads, oftentimes don’t have insurance because they’re also precluded from having auto insurance. So, this is really something that not just benefits these patients but benefits our whole community.”
The fact that Dr. Fiala followed through on something that all doctors aspire to do — and in the middle of a very busy training program — is something that Dr. Budinger said makes him stand out in a big way.
“If you talk to any of our trainees or young faculty, everybody’s interested in addressing the issue of health disparities,” said Dr. Budinger. “Justin looked at that and said, ‘Well, you know, I’m not interested in talking about it. What can I do about this problem? And how can I actually get boots on the ground and help?’ That requires a big activation energy that many people don’t have.”
Lesson #4: Be a People-Person and a Patient-Person
When hiring employees at American Family Care in Portland, Oregon, Andrew Miller, MD, director of provider training, is always on the lookout for young MDs with emotional intelligence and a good bedside manner. He has been recently blown away, however, by a young physician’s assistant named Joseph Van Bindsbergen, PA-C, who was described as “all-around wonderful” during his reference check.
“Having less than 6 months of experience out of school, he is our highest ranked provider, whether it’s a nurse practitioner, PA, or doctor, in terms of patient satisfaction,” said Dr. Miller. The young PA has an “unprecedented perfect score” on his NPS rating.
Why? Patients said they’ve never felt as heard as they felt with Van Bindsbergen.
“That’s the thing I think that the up-and-coming providers should be focusing on is making your patients feel heard,” explained Dr. Miller. Van Bindsbergen is great at building rapport with a patient, whether they are 6 or 96. “He doesn’t just ask about sore throat symptoms. He asks, ‘what is the impact on your life of the sore throat? How does it affect your family or your work? What do you think this could be besides just strep? What are your concerns?’ ”
Dr. Miller said the magic of Van Bindsbergen is that he has an innate ability to look at patients “not just as a diagnosis but as a person, which they love.”
Lesson #5: Remember to Make That Difference With Each Patient
Doctors are used to swooping in and seeing a patient, ordering further testing if needed, and then moving on to the next patient. But one young intern at the start of his medical career broke this mold by giving a very anxious patient some much-needed support.
“There was a resident who was working overnight, and this poor young woman came in who had a new diagnosis of an advanced illness and a lot of anxiety around her condition, the newness of it, and the impact this is going to have on her family and her life,” said Elizabeth Horn Prsic, MD, assistant professor at Yale School of Medicine and firm chief for medical oncology and the director of Adult Inpatient Palliative Care.
Dr. Prsic found out the next morning that this trainee accompanied the patient to the MRI and held her hand as much as he was allowed to throughout the entire experience. “I was like, ‘wait you went down with her to radiology?’ And he’s like, ‘Yes, I was there the whole time,’ ” she recalled.
This gesture not only helped the patient feel calmer after receiving a potentially life-altering diagnosis but also helped ensure the test results were as clear as possible.
“If the study is not done well and a patient is moving or uncomfortable, it has to be stopped early or paused,” said Dr. Prsic. “Then the study is not very useful. In situations like these, medical decisions may be made based on imperfect data. The fact that we had this full complete good quality scan helped us get the care that she needed in a much timelier manner to help her and to move along the care that she that was medically appropriate for her.”
Dr. Prsic got emotional reflecting on the experience. Working at Yale, she saw a ton of intelligent doctors come through the ranks. But this gesture, she said, should serve as a reminder that “you don’t need to be the smartest person in the room to just be there for a patient. It was pure empathic presence and human connection. It gave me hope in the next generation of physicians.”
A version of this article appeared on Medscape.com.
Safe to say that anyone undertaking the physician journey does so with intense motivation and book smarts. Still, it can be incredibly hard to stand out. Everyone’s a go-getter, but what’s the X factor?
Here’s what they said ...
Lesson #1: Never Be Scared to Ask
Brien Barnewolt, MD, chairman and chief of the Department of Emergency Medicine at Tufts Medical Center, was very much surprised when a resident named Scott G. Weiner did something unexpected: Go after a job in the fall of his junior year residency instead of following the typical senior year trajectory.
“It’s very unusual for a trainee to apply for a job virtually a year ahead of schedule. But he knew what he wanted,” said Dr. Barnewolt. “I’d never had anybody come to me in that same scenario, and I’ve been doing this a long time.”
Under normal circumstances it would’ve been easy for Dr. Barnewolt to say no. But the unexpected request made him and his colleagues take a closer look, and they were impressed with Dr. Weiner’s skills. That, paired with his ambition and demeanor, compelled them to offer him an early job. But there’s more.
As the next year approached, Dr. Weiner explained he had an opportunity to work in emergency medicine in Tuscany and asked if he could take a 1-year delayed start for the position he applied a year early for.
The department held his position, and upon his return, Dr. Weiner made a lasting impact at Tufts before eventually moving on. “He outgrew us, which is nice to see,” Dr. Barnewolt said. (Dr. Weiner is currently McGraw Distinguished Chair in Emergency Medicine at Brigham and Women’s Hospital and associate professor at Harvard Medical School.)
Bottom line: Why did Dr. Barnewolt and his colleagues do so much to accommodate a young candidate? Yes, Dr. Weiner was talented, but he was also up-front about his ambitions from the get-go. Dr. Barnewolt said that kind of initiative can only be looked at positively.
“My advice would be, if you see an opportunity or a potential place where you might want to work, put out those feelers, start those conversations,” he said. “It’s not too early, especially in certain specialties, where the job market is very tight. Then, when circumstances change, be open about it and have that conversation. The worst that somebody can say is no, so it never hurts to be honest and open about where you want to go and what you want to be.”
Lesson #2: Chase Your Passion ‘Relentlessly’
Vance G. Fowler, MD, MHS, an infectious disease specialist at Duke University School of Medicine, runs a laboratory that researches methicillin-resistant Staphylococcus aureus (MRSA). Over the years, he’s mentored many doctors but understands the ambitions of young trainees don’t always align with the little free time that they have. “Many of them drop away when you give them a [side] project,” he said.
So when Tori Kinamon asked him to work on an MRSA project — in her first year — he gave her one that focused on researching vertebral osteomyelitis, a bone infection that can coincide with S aureus. What Dr. Fowler didn’t know: Kinamon (now MD) had been a competitive gymnast at Brown and battled her own life-threatening infection with MRSA.
“To my absolute astonishment, not only did she stick to it, but she was able to compile a presentation on the science and gave an oral presentation within a year of walking in the door,” said Dr. Fowler.
She went on to lead an initiative between the National Institutes of Health and US Food and Drug Administration to create endpoints for clinical drug trials, all of which occurred before starting her residency, which she’s about to embark upon.
Dr. Kinamon’s a good example, he said, of what happens when you add genuine passion to book smarts. Those who do always stand out because you can’t fake that. “Find your passion, and then chase it down relentlessly,” he said. “Once you’ve found your passion, things get easy because it stops being work and it starts being something else.”
If you haven’t identified a focus area, Dr. Fowler said to “be agnostic and observant. Keep your eyes open and your options open because you may surprise yourself. It may turn out that you end up liking something a whole lot more than you thought you did.”
Lesson #3: When You Say You’ve Always Wanted to Do Something, Do Something
As the chief of pulmonary and critical care medicine at the Northwestern Medicine Canning Thoracic Institute, Scott Budinger, MD, often hears lip service from doctors who want to put their skills to use in their local communities. One of his students actually did it.
Justin Fiala, MD, a pulmonary, critical care, and sleep specialist at Northwestern Medicine, joined Northwestern as a pulmonary fellow with a big interest in addressing health equity issues.
Dr. Fiala began volunteering with CommunityHealth during his fellowship and saw that many patients of the free Chicago-area clinic needed help with sleep disorders. He launched the organization’s first sleep clinic and its Patient-Centered Apnea Protocols Initiative.
“He developed a plan with some of the partners of the sleep apnea equipment to do home sleep testing for these patients that’s free of cost,” said Dr. Budinger.
Dr. Fiala goes in on Saturdays and runs a free clinic conducting sleep studies for patients and outfits them with devices that they need to improve their conditions, said Dr. Budinger.
“And these patients are the severest of the severe patients,” he said. “These are people that have severe sleep apnea that are driving around the roads, oftentimes don’t have insurance because they’re also precluded from having auto insurance. So, this is really something that not just benefits these patients but benefits our whole community.”
The fact that Dr. Fiala followed through on something that all doctors aspire to do — and in the middle of a very busy training program — is something that Dr. Budinger said makes him stand out in a big way.
“If you talk to any of our trainees or young faculty, everybody’s interested in addressing the issue of health disparities,” said Dr. Budinger. “Justin looked at that and said, ‘Well, you know, I’m not interested in talking about it. What can I do about this problem? And how can I actually get boots on the ground and help?’ That requires a big activation energy that many people don’t have.”
Lesson #4: Be a People-Person and a Patient-Person
When hiring employees at American Family Care in Portland, Oregon, Andrew Miller, MD, director of provider training, is always on the lookout for young MDs with emotional intelligence and a good bedside manner. He has been recently blown away, however, by a young physician’s assistant named Joseph Van Bindsbergen, PA-C, who was described as “all-around wonderful” during his reference check.
“Having less than 6 months of experience out of school, he is our highest ranked provider, whether it’s a nurse practitioner, PA, or doctor, in terms of patient satisfaction,” said Dr. Miller. The young PA has an “unprecedented perfect score” on his NPS rating.
Why? Patients said they’ve never felt as heard as they felt with Van Bindsbergen.
“That’s the thing I think that the up-and-coming providers should be focusing on is making your patients feel heard,” explained Dr. Miller. Van Bindsbergen is great at building rapport with a patient, whether they are 6 or 96. “He doesn’t just ask about sore throat symptoms. He asks, ‘what is the impact on your life of the sore throat? How does it affect your family or your work? What do you think this could be besides just strep? What are your concerns?’ ”
Dr. Miller said the magic of Van Bindsbergen is that he has an innate ability to look at patients “not just as a diagnosis but as a person, which they love.”
Lesson #5: Remember to Make That Difference With Each Patient
Doctors are used to swooping in and seeing a patient, ordering further testing if needed, and then moving on to the next patient. But one young intern at the start of his medical career broke this mold by giving a very anxious patient some much-needed support.
“There was a resident who was working overnight, and this poor young woman came in who had a new diagnosis of an advanced illness and a lot of anxiety around her condition, the newness of it, and the impact this is going to have on her family and her life,” said Elizabeth Horn Prsic, MD, assistant professor at Yale School of Medicine and firm chief for medical oncology and the director of Adult Inpatient Palliative Care.
Dr. Prsic found out the next morning that this trainee accompanied the patient to the MRI and held her hand as much as he was allowed to throughout the entire experience. “I was like, ‘wait you went down with her to radiology?’ And he’s like, ‘Yes, I was there the whole time,’ ” she recalled.
This gesture not only helped the patient feel calmer after receiving a potentially life-altering diagnosis but also helped ensure the test results were as clear as possible.
“If the study is not done well and a patient is moving or uncomfortable, it has to be stopped early or paused,” said Dr. Prsic. “Then the study is not very useful. In situations like these, medical decisions may be made based on imperfect data. The fact that we had this full complete good quality scan helped us get the care that she needed in a much timelier manner to help her and to move along the care that she that was medically appropriate for her.”
Dr. Prsic got emotional reflecting on the experience. Working at Yale, she saw a ton of intelligent doctors come through the ranks. But this gesture, she said, should serve as a reminder that “you don’t need to be the smartest person in the room to just be there for a patient. It was pure empathic presence and human connection. It gave me hope in the next generation of physicians.”
A version of this article appeared on Medscape.com.
Safe to say that anyone undertaking the physician journey does so with intense motivation and book smarts. Still, it can be incredibly hard to stand out. Everyone’s a go-getter, but what’s the X factor?
Here’s what they said ...
Lesson #1: Never Be Scared to Ask
Brien Barnewolt, MD, chairman and chief of the Department of Emergency Medicine at Tufts Medical Center, was very much surprised when a resident named Scott G. Weiner did something unexpected: Go after a job in the fall of his junior year residency instead of following the typical senior year trajectory.
“It’s very unusual for a trainee to apply for a job virtually a year ahead of schedule. But he knew what he wanted,” said Dr. Barnewolt. “I’d never had anybody come to me in that same scenario, and I’ve been doing this a long time.”
Under normal circumstances it would’ve been easy for Dr. Barnewolt to say no. But the unexpected request made him and his colleagues take a closer look, and they were impressed with Dr. Weiner’s skills. That, paired with his ambition and demeanor, compelled them to offer him an early job. But there’s more.
As the next year approached, Dr. Weiner explained he had an opportunity to work in emergency medicine in Tuscany and asked if he could take a 1-year delayed start for the position he applied a year early for.
The department held his position, and upon his return, Dr. Weiner made a lasting impact at Tufts before eventually moving on. “He outgrew us, which is nice to see,” Dr. Barnewolt said. (Dr. Weiner is currently McGraw Distinguished Chair in Emergency Medicine at Brigham and Women’s Hospital and associate professor at Harvard Medical School.)
Bottom line: Why did Dr. Barnewolt and his colleagues do so much to accommodate a young candidate? Yes, Dr. Weiner was talented, but he was also up-front about his ambitions from the get-go. Dr. Barnewolt said that kind of initiative can only be looked at positively.
“My advice would be, if you see an opportunity or a potential place where you might want to work, put out those feelers, start those conversations,” he said. “It’s not too early, especially in certain specialties, where the job market is very tight. Then, when circumstances change, be open about it and have that conversation. The worst that somebody can say is no, so it never hurts to be honest and open about where you want to go and what you want to be.”
Lesson #2: Chase Your Passion ‘Relentlessly’
Vance G. Fowler, MD, MHS, an infectious disease specialist at Duke University School of Medicine, runs a laboratory that researches methicillin-resistant Staphylococcus aureus (MRSA). Over the years, he’s mentored many doctors but understands the ambitions of young trainees don’t always align with the little free time that they have. “Many of them drop away when you give them a [side] project,” he said.
So when Tori Kinamon asked him to work on an MRSA project — in her first year — he gave her one that focused on researching vertebral osteomyelitis, a bone infection that can coincide with S aureus. What Dr. Fowler didn’t know: Kinamon (now MD) had been a competitive gymnast at Brown and battled her own life-threatening infection with MRSA.
“To my absolute astonishment, not only did she stick to it, but she was able to compile a presentation on the science and gave an oral presentation within a year of walking in the door,” said Dr. Fowler.
She went on to lead an initiative between the National Institutes of Health and US Food and Drug Administration to create endpoints for clinical drug trials, all of which occurred before starting her residency, which she’s about to embark upon.
Dr. Kinamon’s a good example, he said, of what happens when you add genuine passion to book smarts. Those who do always stand out because you can’t fake that. “Find your passion, and then chase it down relentlessly,” he said. “Once you’ve found your passion, things get easy because it stops being work and it starts being something else.”
If you haven’t identified a focus area, Dr. Fowler said to “be agnostic and observant. Keep your eyes open and your options open because you may surprise yourself. It may turn out that you end up liking something a whole lot more than you thought you did.”
Lesson #3: When You Say You’ve Always Wanted to Do Something, Do Something
As the chief of pulmonary and critical care medicine at the Northwestern Medicine Canning Thoracic Institute, Scott Budinger, MD, often hears lip service from doctors who want to put their skills to use in their local communities. One of his students actually did it.
Justin Fiala, MD, a pulmonary, critical care, and sleep specialist at Northwestern Medicine, joined Northwestern as a pulmonary fellow with a big interest in addressing health equity issues.
Dr. Fiala began volunteering with CommunityHealth during his fellowship and saw that many patients of the free Chicago-area clinic needed help with sleep disorders. He launched the organization’s first sleep clinic and its Patient-Centered Apnea Protocols Initiative.
“He developed a plan with some of the partners of the sleep apnea equipment to do home sleep testing for these patients that’s free of cost,” said Dr. Budinger.
Dr. Fiala goes in on Saturdays and runs a free clinic conducting sleep studies for patients and outfits them with devices that they need to improve their conditions, said Dr. Budinger.
“And these patients are the severest of the severe patients,” he said. “These are people that have severe sleep apnea that are driving around the roads, oftentimes don’t have insurance because they’re also precluded from having auto insurance. So, this is really something that not just benefits these patients but benefits our whole community.”
The fact that Dr. Fiala followed through on something that all doctors aspire to do — and in the middle of a very busy training program — is something that Dr. Budinger said makes him stand out in a big way.
“If you talk to any of our trainees or young faculty, everybody’s interested in addressing the issue of health disparities,” said Dr. Budinger. “Justin looked at that and said, ‘Well, you know, I’m not interested in talking about it. What can I do about this problem? And how can I actually get boots on the ground and help?’ That requires a big activation energy that many people don’t have.”
Lesson #4: Be a People-Person and a Patient-Person
When hiring employees at American Family Care in Portland, Oregon, Andrew Miller, MD, director of provider training, is always on the lookout for young MDs with emotional intelligence and a good bedside manner. He has been recently blown away, however, by a young physician’s assistant named Joseph Van Bindsbergen, PA-C, who was described as “all-around wonderful” during his reference check.
“Having less than 6 months of experience out of school, he is our highest ranked provider, whether it’s a nurse practitioner, PA, or doctor, in terms of patient satisfaction,” said Dr. Miller. The young PA has an “unprecedented perfect score” on his NPS rating.
Why? Patients said they’ve never felt as heard as they felt with Van Bindsbergen.
“That’s the thing I think that the up-and-coming providers should be focusing on is making your patients feel heard,” explained Dr. Miller. Van Bindsbergen is great at building rapport with a patient, whether they are 6 or 96. “He doesn’t just ask about sore throat symptoms. He asks, ‘what is the impact on your life of the sore throat? How does it affect your family or your work? What do you think this could be besides just strep? What are your concerns?’ ”
Dr. Miller said the magic of Van Bindsbergen is that he has an innate ability to look at patients “not just as a diagnosis but as a person, which they love.”
Lesson #5: Remember to Make That Difference With Each Patient
Doctors are used to swooping in and seeing a patient, ordering further testing if needed, and then moving on to the next patient. But one young intern at the start of his medical career broke this mold by giving a very anxious patient some much-needed support.
“There was a resident who was working overnight, and this poor young woman came in who had a new diagnosis of an advanced illness and a lot of anxiety around her condition, the newness of it, and the impact this is going to have on her family and her life,” said Elizabeth Horn Prsic, MD, assistant professor at Yale School of Medicine and firm chief for medical oncology and the director of Adult Inpatient Palliative Care.
Dr. Prsic found out the next morning that this trainee accompanied the patient to the MRI and held her hand as much as he was allowed to throughout the entire experience. “I was like, ‘wait you went down with her to radiology?’ And he’s like, ‘Yes, I was there the whole time,’ ” she recalled.
This gesture not only helped the patient feel calmer after receiving a potentially life-altering diagnosis but also helped ensure the test results were as clear as possible.
“If the study is not done well and a patient is moving or uncomfortable, it has to be stopped early or paused,” said Dr. Prsic. “Then the study is not very useful. In situations like these, medical decisions may be made based on imperfect data. The fact that we had this full complete good quality scan helped us get the care that she needed in a much timelier manner to help her and to move along the care that she that was medically appropriate for her.”
Dr. Prsic got emotional reflecting on the experience. Working at Yale, she saw a ton of intelligent doctors come through the ranks. But this gesture, she said, should serve as a reminder that “you don’t need to be the smartest person in the room to just be there for a patient. It was pure empathic presence and human connection. It gave me hope in the next generation of physicians.”
A version of this article appeared on Medscape.com.
Teleneurology for Suspected Stroke Speeds Treatment
, new research showed.
“This preliminary evidence supports adopting teleneurology prenotification as a best practice within health systems that have telestroke capabilities,” said study investigator Mark McDonald, MD, a neurologist at TeleSpecialists, Fort Myers, Florida.
The findings were presented at the 2024 annual meeting of the American Academy of Neurology.
Best Practices
The impact of emergency medical services prenotification, which refers to paramedics alerting receiving hospital emergency departments (EDs) of a suspected stroke on the way for appropriate preparations to be made, is well-defined, said Dr. McDonald.
“What we’re proposing as a best practice is not only should the ED or ED provider be aware, but there needs to be a system in place for standardizing communication to the neurology team so they’re aware, too.”
Prenotification allows a neurologist to “get on the screen to begin coordinating with the ED team to adequately prepare for the possibility of thrombolytic treatment,” he added.
Currently, teleneurology prenotification, he said, is variable and its benefits unclear.
Dr. McDonald said “his organization, TeleSpecialists, maintains a large detailed medical records database for emergency-related, teleneurology, and other cases. For stroke, it recommends 15 best practices” for facilities including prenotification of teleneurology.
Other best practices include evaluating and administering thrombolysis in the CT imaging suite, a preassembled stroke kit that includes antihypertensives and thrombolytic agents, ensuring a weigh bed is available to determine the exact dose of thrombolysis treatment, and implementing “mock” stroke alerts, said Dr. McDonald.
From the database, researchers extracted acute telestroke consultations seen in the ED in 103 facilities in 15 states. Facilities that did not adhere to the 14 best practices other than teleneurologist prenotification were excluded from the analysis.
Of 9290 patients included in the study, 731 were treated with thrombolysis at prenotification facilities (median age, 69 years; median National Institutes of Health Stroke Score [NIHSS], 8) and 31 were treated at facilities without prenotification (median age, 63 years; median NIHSS score, 4). The thrombolytic treatment rate was 8.5% at prenotification facilities versus 4.8% at facilities without prenotification — a difference that was statistically significant.
Prenotification facilities had a significantly shorter median door-to-needle (DTN) time than those without such a process at 35 versus 43 minutes. In addition, there was a statistically significant difference in the percentage of patients with times less than 60 minutes at approximately 88% at prenotification facilities versus about 68% at the facilities without prenotification.
Case-Level Analysis
However, just because a facility adheres to teleneurology prenotification as a whole, doesn’t mean it occurs in every case. Researchers explored the impact of teleneurology prenotification at the case level rather than the facility level.
“That gave us a bit more insight into the real impact because it’s not just being at a facility with the best practice; it’s actually working case by case to see whether it happened or not and that’s where we get the most compelling findings,” said Dr. McDonald.
Of 761 treatment cases, there was prenotification to the neurology team in 401 cases. In 360 cases, prenotification did not occur.
The median DTN time was 29 minutes in the group with actual prenotification vs 41.5 minutes in the group without actual prenotification, a difference that was statistically significant, Dr. McDonald said.
As for treatment within 30 minutes of arrival, 50.4% of patients in the teleneurology prenotification group versus 18.9% in the no prenotification group — a statistically significant difference.
DTN time of less than 30 minutes is increasingly used as a target. “Being treated within this time frame improves outcomes and reduces length of hospital stay,” said Dr. McDonald.
The prenotification group also had a statistically significant higher percentage of treatment within 60 minutes of hospital arrival (93.5% vs 80%).
These new findings should help convince health and telestroke systems that teleneurology prenotification is worth implementing. “We want to achieve consensus on this as a best practice,” said Dr. McDonald.
Prenotification, he added, “coordinates the process and eliminates unnecessary and time-consuming steps.”
Dr. McDonald plans to prospectively study prenotification by collecting data on a facility before and after implementing a prenotification process.
Compelling Evidence
Commenting on the research, David L. Tirschwell, MD, Harborview Medical Center, Department of Neurology, Seattle, who cochaired the AAN session featuring the research, said the study provides compelling evidence that teleneurologist prenotification improves DTN time.
“Prenotifications are often standard of care in many healthcare settings and should likely be considered a best practice. When possible, extending such prenotification to a teleconsultant would make sense, and these preliminary data support that approach.”
However, more details are needed “to consider whether the intervention is possibly generalizable to other telestroke practices across the United States,” said Dr. Tirschwell.
Dr. McDonald reported receiving personal compensation for serving as a consultant for Syntrillo Inc. and has stock in Syntrillo Inc. Dr. Tirschwell reported no relevant conflicts of interest.
A version of this article appeared on Medscape.com.
, new research showed.
“This preliminary evidence supports adopting teleneurology prenotification as a best practice within health systems that have telestroke capabilities,” said study investigator Mark McDonald, MD, a neurologist at TeleSpecialists, Fort Myers, Florida.
The findings were presented at the 2024 annual meeting of the American Academy of Neurology.
Best Practices
The impact of emergency medical services prenotification, which refers to paramedics alerting receiving hospital emergency departments (EDs) of a suspected stroke on the way for appropriate preparations to be made, is well-defined, said Dr. McDonald.
“What we’re proposing as a best practice is not only should the ED or ED provider be aware, but there needs to be a system in place for standardizing communication to the neurology team so they’re aware, too.”
Prenotification allows a neurologist to “get on the screen to begin coordinating with the ED team to adequately prepare for the possibility of thrombolytic treatment,” he added.
Currently, teleneurology prenotification, he said, is variable and its benefits unclear.
Dr. McDonald said “his organization, TeleSpecialists, maintains a large detailed medical records database for emergency-related, teleneurology, and other cases. For stroke, it recommends 15 best practices” for facilities including prenotification of teleneurology.
Other best practices include evaluating and administering thrombolysis in the CT imaging suite, a preassembled stroke kit that includes antihypertensives and thrombolytic agents, ensuring a weigh bed is available to determine the exact dose of thrombolysis treatment, and implementing “mock” stroke alerts, said Dr. McDonald.
From the database, researchers extracted acute telestroke consultations seen in the ED in 103 facilities in 15 states. Facilities that did not adhere to the 14 best practices other than teleneurologist prenotification were excluded from the analysis.
Of 9290 patients included in the study, 731 were treated with thrombolysis at prenotification facilities (median age, 69 years; median National Institutes of Health Stroke Score [NIHSS], 8) and 31 were treated at facilities without prenotification (median age, 63 years; median NIHSS score, 4). The thrombolytic treatment rate was 8.5% at prenotification facilities versus 4.8% at facilities without prenotification — a difference that was statistically significant.
Prenotification facilities had a significantly shorter median door-to-needle (DTN) time than those without such a process at 35 versus 43 minutes. In addition, there was a statistically significant difference in the percentage of patients with times less than 60 minutes at approximately 88% at prenotification facilities versus about 68% at the facilities without prenotification.
Case-Level Analysis
However, just because a facility adheres to teleneurology prenotification as a whole, doesn’t mean it occurs in every case. Researchers explored the impact of teleneurology prenotification at the case level rather than the facility level.
“That gave us a bit more insight into the real impact because it’s not just being at a facility with the best practice; it’s actually working case by case to see whether it happened or not and that’s where we get the most compelling findings,” said Dr. McDonald.
Of 761 treatment cases, there was prenotification to the neurology team in 401 cases. In 360 cases, prenotification did not occur.
The median DTN time was 29 minutes in the group with actual prenotification vs 41.5 minutes in the group without actual prenotification, a difference that was statistically significant, Dr. McDonald said.
As for treatment within 30 minutes of arrival, 50.4% of patients in the teleneurology prenotification group versus 18.9% in the no prenotification group — a statistically significant difference.
DTN time of less than 30 minutes is increasingly used as a target. “Being treated within this time frame improves outcomes and reduces length of hospital stay,” said Dr. McDonald.
The prenotification group also had a statistically significant higher percentage of treatment within 60 minutes of hospital arrival (93.5% vs 80%).
These new findings should help convince health and telestroke systems that teleneurology prenotification is worth implementing. “We want to achieve consensus on this as a best practice,” said Dr. McDonald.
Prenotification, he added, “coordinates the process and eliminates unnecessary and time-consuming steps.”
Dr. McDonald plans to prospectively study prenotification by collecting data on a facility before and after implementing a prenotification process.
Compelling Evidence
Commenting on the research, David L. Tirschwell, MD, Harborview Medical Center, Department of Neurology, Seattle, who cochaired the AAN session featuring the research, said the study provides compelling evidence that teleneurologist prenotification improves DTN time.
“Prenotifications are often standard of care in many healthcare settings and should likely be considered a best practice. When possible, extending such prenotification to a teleconsultant would make sense, and these preliminary data support that approach.”
However, more details are needed “to consider whether the intervention is possibly generalizable to other telestroke practices across the United States,” said Dr. Tirschwell.
Dr. McDonald reported receiving personal compensation for serving as a consultant for Syntrillo Inc. and has stock in Syntrillo Inc. Dr. Tirschwell reported no relevant conflicts of interest.
A version of this article appeared on Medscape.com.
, new research showed.
“This preliminary evidence supports adopting teleneurology prenotification as a best practice within health systems that have telestroke capabilities,” said study investigator Mark McDonald, MD, a neurologist at TeleSpecialists, Fort Myers, Florida.
The findings were presented at the 2024 annual meeting of the American Academy of Neurology.
Best Practices
The impact of emergency medical services prenotification, which refers to paramedics alerting receiving hospital emergency departments (EDs) of a suspected stroke on the way for appropriate preparations to be made, is well-defined, said Dr. McDonald.
“What we’re proposing as a best practice is not only should the ED or ED provider be aware, but there needs to be a system in place for standardizing communication to the neurology team so they’re aware, too.”
Prenotification allows a neurologist to “get on the screen to begin coordinating with the ED team to adequately prepare for the possibility of thrombolytic treatment,” he added.
Currently, teleneurology prenotification, he said, is variable and its benefits unclear.
Dr. McDonald said “his organization, TeleSpecialists, maintains a large detailed medical records database for emergency-related, teleneurology, and other cases. For stroke, it recommends 15 best practices” for facilities including prenotification of teleneurology.
Other best practices include evaluating and administering thrombolysis in the CT imaging suite, a preassembled stroke kit that includes antihypertensives and thrombolytic agents, ensuring a weigh bed is available to determine the exact dose of thrombolysis treatment, and implementing “mock” stroke alerts, said Dr. McDonald.
From the database, researchers extracted acute telestroke consultations seen in the ED in 103 facilities in 15 states. Facilities that did not adhere to the 14 best practices other than teleneurologist prenotification were excluded from the analysis.
Of 9290 patients included in the study, 731 were treated with thrombolysis at prenotification facilities (median age, 69 years; median National Institutes of Health Stroke Score [NIHSS], 8) and 31 were treated at facilities without prenotification (median age, 63 years; median NIHSS score, 4). The thrombolytic treatment rate was 8.5% at prenotification facilities versus 4.8% at facilities without prenotification — a difference that was statistically significant.
Prenotification facilities had a significantly shorter median door-to-needle (DTN) time than those without such a process at 35 versus 43 minutes. In addition, there was a statistically significant difference in the percentage of patients with times less than 60 minutes at approximately 88% at prenotification facilities versus about 68% at the facilities without prenotification.
Case-Level Analysis
However, just because a facility adheres to teleneurology prenotification as a whole, doesn’t mean it occurs in every case. Researchers explored the impact of teleneurology prenotification at the case level rather than the facility level.
“That gave us a bit more insight into the real impact because it’s not just being at a facility with the best practice; it’s actually working case by case to see whether it happened or not and that’s where we get the most compelling findings,” said Dr. McDonald.
Of 761 treatment cases, there was prenotification to the neurology team in 401 cases. In 360 cases, prenotification did not occur.
The median DTN time was 29 minutes in the group with actual prenotification vs 41.5 minutes in the group without actual prenotification, a difference that was statistically significant, Dr. McDonald said.
As for treatment within 30 minutes of arrival, 50.4% of patients in the teleneurology prenotification group versus 18.9% in the no prenotification group — a statistically significant difference.
DTN time of less than 30 minutes is increasingly used as a target. “Being treated within this time frame improves outcomes and reduces length of hospital stay,” said Dr. McDonald.
The prenotification group also had a statistically significant higher percentage of treatment within 60 minutes of hospital arrival (93.5% vs 80%).
These new findings should help convince health and telestroke systems that teleneurology prenotification is worth implementing. “We want to achieve consensus on this as a best practice,” said Dr. McDonald.
Prenotification, he added, “coordinates the process and eliminates unnecessary and time-consuming steps.”
Dr. McDonald plans to prospectively study prenotification by collecting data on a facility before and after implementing a prenotification process.
Compelling Evidence
Commenting on the research, David L. Tirschwell, MD, Harborview Medical Center, Department of Neurology, Seattle, who cochaired the AAN session featuring the research, said the study provides compelling evidence that teleneurologist prenotification improves DTN time.
“Prenotifications are often standard of care in many healthcare settings and should likely be considered a best practice. When possible, extending such prenotification to a teleconsultant would make sense, and these preliminary data support that approach.”
However, more details are needed “to consider whether the intervention is possibly generalizable to other telestroke practices across the United States,” said Dr. Tirschwell.
Dr. McDonald reported receiving personal compensation for serving as a consultant for Syntrillo Inc. and has stock in Syntrillo Inc. Dr. Tirschwell reported no relevant conflicts of interest.
A version of this article appeared on Medscape.com.
FROM AAN 2024
Which Emergencies Are Genuine Emergencies?
WIESBADEN, GERMANY — Crowded waiting rooms, long wait times, irritable patients, and aggression toward nursing staff and doctors are increasingly the reality in German emergency rooms. Clearly, emergencies belong in the emergency room. However, “In about half of all patients in the emergency room, there is no urgent medical emergency,” Norbert Schütz, MD, director of geriatrics and rheumatology at Helios Dr. Horst Schmidt Hospital in Wiesbaden, Germany, said at a press conference for the 130th Annual Meeting of the German Society of Internal Medicine (DGIM).
“In our daily medical practice, we repeatedly experience people either accessing our emergency departments and ambulances too quickly or lingering at home for too long when they have severe symptoms,” said Dr. Schütz, who organized the Patient Day during the Internist Congress.
DGIM Educates Patients
What is an emergency? “I think the public is quite well informed about conditions associated with loss of consciousness, severe pain, chest pain, or paralysis: Think stroke or heart attack. This is undoubtedly a success of recent years. The difficulty arises with everything in between. For instance, should I go to the hospital with severe headaches?” asked Dr. Schütz.
When is a patient a case for the emergency room, the physician on-call service, or the general practitioner? At the Patient Day in Wiesbaden, DGIM aims to educate and train interested parties with a dedicated lecture. The focus is on recognizing an emergency, specifically emergencies in children and mental illnesses.
“Our Patient Day aims to contribute to making the right decisions. We want to inform, answer questions, and alleviate fears,” said Dr. Schütz. Interested parties can refresh their emergency knowledge, tour ambulances, and have the equipment explained. The public also has the opportunity to learn about resuscitation techniques theoretically and practically.
“Should, for whatever reason, the general practitioner not be reachable, the physician on-call service can be reached,” said Dr. Schütz. It may happen, however, that neither the general practitioner nor the on-call physician is immediately available.
What Are Emergencies?
In cases of severe health impairment, urgency is required, and a severe emergency should be assumed in the following cases:
- Chest pain
- Circulatory disorder
- Disorders of consciousness
- Breathing difficulties
- Sudden weakness or numbness/paralysis
- Severe bleeding
- Allergic shock
“In such cases, the emergency departments of the hospitals are available around the clock, and if necessary, an emergency doctor should be present during transportation to the hospital,” said Dr. Schütz.
Classifying emergencies is challenging, especially with children. “Children often find it difficult to clearly categorize or describe symptoms,” said Dr. Schütz. A situation is critical if, for example, the child’s breathing or consciousness is impaired.
Mental emergencies pose a particular challenge for patients and relatives because the patient and relatives are often overwhelmed by the situation. If there are suicidal thoughts, the patient should present him- or herself immediately to an emergency room.
“Patients who come to the emergency room because they cannot get appointments with their general practitioner or specialist, for whatever reason, are no emergency. We also see this in the emergency room from time to time,” said Dr. Schütz. Emergency rooms are not intended for this purpose. “And generally, these are not emergencies.”
Four of 10 Cases
The number of patients in emergency rooms has steadily increased in recent years. Statistically, only 4 out of 10 cases are genuine emergencies, as detailed surveys of patients in the emergency rooms of northern German hospitals have shown.
In the PiNo Nord cross-sectional study, Martin Scherer, MD, of University Hospital Hamburg-Eppendorf in Hamburg, Germany, and his team examined the reasons why patients visit the emergency room. They interviewed 1175 patients in five hospitals and documented the medical diagnoses. Patients classified as “immediately” or “very urgently” in need of treatment were excluded.
The surveyed patients were on average 41.8 years old, 52.9% were men, and 54.7% of the patients indicated a low urgency of treatment. About 41% of the patients visited the emergency room on their own initiative, 17% stated they were referred or entrusted by their general practitioner, and 8% were referred by a specialist in the emergency room.
The strongest predictors for low subjective treatment urgency were musculoskeletal trauma (odds ratio [OR], 2.18), skin afflictions (OR, 2.15), and the unavailability of an open general practitioner’s office (OR, 1.70).
According to Dr. Scherer and his colleagues, the reasons for visiting an emergency room are diverse and can be based on the perceived structural conditions and individual patient preferences in addition to the urgency of the health problem.
This story was translated from the Medscape German edition using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
WIESBADEN, GERMANY — Crowded waiting rooms, long wait times, irritable patients, and aggression toward nursing staff and doctors are increasingly the reality in German emergency rooms. Clearly, emergencies belong in the emergency room. However, “In about half of all patients in the emergency room, there is no urgent medical emergency,” Norbert Schütz, MD, director of geriatrics and rheumatology at Helios Dr. Horst Schmidt Hospital in Wiesbaden, Germany, said at a press conference for the 130th Annual Meeting of the German Society of Internal Medicine (DGIM).
“In our daily medical practice, we repeatedly experience people either accessing our emergency departments and ambulances too quickly or lingering at home for too long when they have severe symptoms,” said Dr. Schütz, who organized the Patient Day during the Internist Congress.
DGIM Educates Patients
What is an emergency? “I think the public is quite well informed about conditions associated with loss of consciousness, severe pain, chest pain, or paralysis: Think stroke or heart attack. This is undoubtedly a success of recent years. The difficulty arises with everything in between. For instance, should I go to the hospital with severe headaches?” asked Dr. Schütz.
When is a patient a case for the emergency room, the physician on-call service, or the general practitioner? At the Patient Day in Wiesbaden, DGIM aims to educate and train interested parties with a dedicated lecture. The focus is on recognizing an emergency, specifically emergencies in children and mental illnesses.
“Our Patient Day aims to contribute to making the right decisions. We want to inform, answer questions, and alleviate fears,” said Dr. Schütz. Interested parties can refresh their emergency knowledge, tour ambulances, and have the equipment explained. The public also has the opportunity to learn about resuscitation techniques theoretically and practically.
“Should, for whatever reason, the general practitioner not be reachable, the physician on-call service can be reached,” said Dr. Schütz. It may happen, however, that neither the general practitioner nor the on-call physician is immediately available.
What Are Emergencies?
In cases of severe health impairment, urgency is required, and a severe emergency should be assumed in the following cases:
- Chest pain
- Circulatory disorder
- Disorders of consciousness
- Breathing difficulties
- Sudden weakness or numbness/paralysis
- Severe bleeding
- Allergic shock
“In such cases, the emergency departments of the hospitals are available around the clock, and if necessary, an emergency doctor should be present during transportation to the hospital,” said Dr. Schütz.
Classifying emergencies is challenging, especially with children. “Children often find it difficult to clearly categorize or describe symptoms,” said Dr. Schütz. A situation is critical if, for example, the child’s breathing or consciousness is impaired.
Mental emergencies pose a particular challenge for patients and relatives because the patient and relatives are often overwhelmed by the situation. If there are suicidal thoughts, the patient should present him- or herself immediately to an emergency room.
“Patients who come to the emergency room because they cannot get appointments with their general practitioner or specialist, for whatever reason, are no emergency. We also see this in the emergency room from time to time,” said Dr. Schütz. Emergency rooms are not intended for this purpose. “And generally, these are not emergencies.”
Four of 10 Cases
The number of patients in emergency rooms has steadily increased in recent years. Statistically, only 4 out of 10 cases are genuine emergencies, as detailed surveys of patients in the emergency rooms of northern German hospitals have shown.
In the PiNo Nord cross-sectional study, Martin Scherer, MD, of University Hospital Hamburg-Eppendorf in Hamburg, Germany, and his team examined the reasons why patients visit the emergency room. They interviewed 1175 patients in five hospitals and documented the medical diagnoses. Patients classified as “immediately” or “very urgently” in need of treatment were excluded.
The surveyed patients were on average 41.8 years old, 52.9% were men, and 54.7% of the patients indicated a low urgency of treatment. About 41% of the patients visited the emergency room on their own initiative, 17% stated they were referred or entrusted by their general practitioner, and 8% were referred by a specialist in the emergency room.
The strongest predictors for low subjective treatment urgency were musculoskeletal trauma (odds ratio [OR], 2.18), skin afflictions (OR, 2.15), and the unavailability of an open general practitioner’s office (OR, 1.70).
According to Dr. Scherer and his colleagues, the reasons for visiting an emergency room are diverse and can be based on the perceived structural conditions and individual patient preferences in addition to the urgency of the health problem.
This story was translated from the Medscape German edition using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
WIESBADEN, GERMANY — Crowded waiting rooms, long wait times, irritable patients, and aggression toward nursing staff and doctors are increasingly the reality in German emergency rooms. Clearly, emergencies belong in the emergency room. However, “In about half of all patients in the emergency room, there is no urgent medical emergency,” Norbert Schütz, MD, director of geriatrics and rheumatology at Helios Dr. Horst Schmidt Hospital in Wiesbaden, Germany, said at a press conference for the 130th Annual Meeting of the German Society of Internal Medicine (DGIM).
“In our daily medical practice, we repeatedly experience people either accessing our emergency departments and ambulances too quickly or lingering at home for too long when they have severe symptoms,” said Dr. Schütz, who organized the Patient Day during the Internist Congress.
DGIM Educates Patients
What is an emergency? “I think the public is quite well informed about conditions associated with loss of consciousness, severe pain, chest pain, or paralysis: Think stroke or heart attack. This is undoubtedly a success of recent years. The difficulty arises with everything in between. For instance, should I go to the hospital with severe headaches?” asked Dr. Schütz.
When is a patient a case for the emergency room, the physician on-call service, or the general practitioner? At the Patient Day in Wiesbaden, DGIM aims to educate and train interested parties with a dedicated lecture. The focus is on recognizing an emergency, specifically emergencies in children and mental illnesses.
“Our Patient Day aims to contribute to making the right decisions. We want to inform, answer questions, and alleviate fears,” said Dr. Schütz. Interested parties can refresh their emergency knowledge, tour ambulances, and have the equipment explained. The public also has the opportunity to learn about resuscitation techniques theoretically and practically.
“Should, for whatever reason, the general practitioner not be reachable, the physician on-call service can be reached,” said Dr. Schütz. It may happen, however, that neither the general practitioner nor the on-call physician is immediately available.
What Are Emergencies?
In cases of severe health impairment, urgency is required, and a severe emergency should be assumed in the following cases:
- Chest pain
- Circulatory disorder
- Disorders of consciousness
- Breathing difficulties
- Sudden weakness or numbness/paralysis
- Severe bleeding
- Allergic shock
“In such cases, the emergency departments of the hospitals are available around the clock, and if necessary, an emergency doctor should be present during transportation to the hospital,” said Dr. Schütz.
Classifying emergencies is challenging, especially with children. “Children often find it difficult to clearly categorize or describe symptoms,” said Dr. Schütz. A situation is critical if, for example, the child’s breathing or consciousness is impaired.
Mental emergencies pose a particular challenge for patients and relatives because the patient and relatives are often overwhelmed by the situation. If there are suicidal thoughts, the patient should present him- or herself immediately to an emergency room.
“Patients who come to the emergency room because they cannot get appointments with their general practitioner or specialist, for whatever reason, are no emergency. We also see this in the emergency room from time to time,” said Dr. Schütz. Emergency rooms are not intended for this purpose. “And generally, these are not emergencies.”
Four of 10 Cases
The number of patients in emergency rooms has steadily increased in recent years. Statistically, only 4 out of 10 cases are genuine emergencies, as detailed surveys of patients in the emergency rooms of northern German hospitals have shown.
In the PiNo Nord cross-sectional study, Martin Scherer, MD, of University Hospital Hamburg-Eppendorf in Hamburg, Germany, and his team examined the reasons why patients visit the emergency room. They interviewed 1175 patients in five hospitals and documented the medical diagnoses. Patients classified as “immediately” or “very urgently” in need of treatment were excluded.
The surveyed patients were on average 41.8 years old, 52.9% were men, and 54.7% of the patients indicated a low urgency of treatment. About 41% of the patients visited the emergency room on their own initiative, 17% stated they were referred or entrusted by their general practitioner, and 8% were referred by a specialist in the emergency room.
The strongest predictors for low subjective treatment urgency were musculoskeletal trauma (odds ratio [OR], 2.18), skin afflictions (OR, 2.15), and the unavailability of an open general practitioner’s office (OR, 1.70).
According to Dr. Scherer and his colleagues, the reasons for visiting an emergency room are diverse and can be based on the perceived structural conditions and individual patient preferences in addition to the urgency of the health problem.
This story was translated from the Medscape German edition using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
Federal Trade Commission Bans Noncompete Agreements, Urges More Protections for Healthcare Workers
But business groups have vowed to challenge the decision in court.
The proposed final rule passed on a 3-2 vote, with the dissenting commissioners disputing the FTC’s authority to broadly ban noncompetes.
Tensions around noncompetes have been building for years. In 2021, President Biden issued an executive order supporting measures to improve economic competition, in which he urged the FTC to consider its rulemaking authority to address noncompete clauses that unfairly limit workers’ mobility. In January 2023, per that directive, the agency proposed ending the restrictive covenants.
While the FTC estimates that the final rule will reduce healthcare costs by up to $194 billion over the next decade and increase worker earnings by $300 million annually, the ruling faces legal hurdles.
US Chamber of Commerce president and CEO Suzanne P. Clark said in a statement that the move is a “blatant power grab” that will undermine competitive business practices, adding that the Chamber will sue to block the measure.
The FTC received more than 26,000 comments on noncompetes during the public feedback period, with about 25,000 supporting the measure, said Benjamin Cady, JD, an FTC attorney.
Mr. Cady called the feedback “compelling,” citing instances of workers who were forced to commute long distances, uproot their families, or risk expensive litigation for wanting to pursue job opportunities.
For example, a comment from a physician working in Appalachia highlights the potential real-life implications of the agreements. “With hospital systems merging, providers with aggressive noncompetes must abandon the community that they serve if they [choose] to leave their employer. Healthcare providers feel trapped in their current employment situation, leading to significant burnout that can shorten their [career] longevity.”
Commissioner Alvaro Bedoya said physicians have had their lives upended by cumbersome noncompetes, often having to move out of state to practice. “A pandemic killed a million people in this country, and there are doctors who cannot work because of a noncompete,” he said.
It’s unclear whether physicians and others who work for nonprofit healthcare groups or hospitals will be covered by the new ban. FTC Commissioner Rebecca Slaughter acknowledged that the agency’s jurisdictional limitations mean that employees of “certain nonprofit organizations” may not benefit from the rule.
“We want to be transparent about the limitation and recognize there are workers, especially healthcare workers, who are bound by anticompetitive and unfair noncompete clauses, that our rule will struggle to reach,” she said. To cover nonprofit healthcare employees, Ms. Slaughter urged Congress to pass legislation banning noncompetes, such as the Workforce Mobility Act of 2021 and the Freedom to Compete Act of 2023.
The FTC final rule will take effect 120 days after it is published in the federal register, and new noncompete agreements will be banned as of this date. However, existing contracts for senior executives will remain in effect because these individuals are less likely to experience “acute harm” due to their ability to negotiate accordingly, said Mr. Cady.
States, AMA Take Aim at Noncompetes
Before the federal ban, several states had already passed legislation limiting the reach of noncompetes. According to a recent article in the Journal of the American College of Cardiology, 12 states prohibit noncompete clauses for physicians: Alabama, California, Colorado, Delaware, Massachusetts, Montana, New Hampshire, New Mexico, North Dakota, Oklahoma, Rhode Island, and South Dakota.
The remaining states allow noncompetes in some form, often excluding them for employees earning below a certain threshold. For example, in Oregon, noncompete agreements may apply to employees earning more than $113,241. Most states have provisions to adjust the threshold annually. The District of Columbia permits 2-year noncompetes for “medical specialists” earning over $250,000 annually.
Indiana employers can no longer enter into noncompete agreements with primary care providers. Other specialties may be subject to the clauses, except when the physician terminates the contract for cause or when an employer terminates the contract without cause.
Rachel Marcus, MD, a cardiologist in Washington, DC, found out how limiting her employment contract’s noncompete clause was when she wanted to leave a former position. Due to the restrictions, she told this news organization that she couldn’t work locally for a competitor for 2 years. The closest location she could seek employment without violating the agreement was Baltimore, approximately 40 miles away.
Dr. Marcus ultimately moved to another position within the same organization because of the company’s reputation for being “aggressive” in their enforcement actions.
Although the American Medical Association (AMA) does not support a total ban, its House of Delegates adopted policies last year to support the prohibition of noncompete contracts for physicians employed by for-profit or nonprofit hospitals, hospital systems, or staffing companies.
Challenges Await
The American Hospital Association, which opposed the proposed rule, called it “bad policy.” The decision “will likely be short-lived, with courts almost certain to stop it before it can do damage to hospitals’ ability to care for their patients and communities,” the association said in a statement.
To ease the transition to the new rule, the FTC also released a model language for employers to use when discussing the changes with their employees. “All employers need to do to comply with the rule is to stop enforcing existing noncompetes with workers other than senior executives and provide notice to such workers,” he said.
Dr. Marcus hopes the ban improves doctors’ lives. “Your employer is going to have to treat you better because they know that you can easily go across town to a place that has a higher salary, and your patient can go with you.”
A version of this article appeared on Medscape.com.
But business groups have vowed to challenge the decision in court.
The proposed final rule passed on a 3-2 vote, with the dissenting commissioners disputing the FTC’s authority to broadly ban noncompetes.
Tensions around noncompetes have been building for years. In 2021, President Biden issued an executive order supporting measures to improve economic competition, in which he urged the FTC to consider its rulemaking authority to address noncompete clauses that unfairly limit workers’ mobility. In January 2023, per that directive, the agency proposed ending the restrictive covenants.
While the FTC estimates that the final rule will reduce healthcare costs by up to $194 billion over the next decade and increase worker earnings by $300 million annually, the ruling faces legal hurdles.
US Chamber of Commerce president and CEO Suzanne P. Clark said in a statement that the move is a “blatant power grab” that will undermine competitive business practices, adding that the Chamber will sue to block the measure.
The FTC received more than 26,000 comments on noncompetes during the public feedback period, with about 25,000 supporting the measure, said Benjamin Cady, JD, an FTC attorney.
Mr. Cady called the feedback “compelling,” citing instances of workers who were forced to commute long distances, uproot their families, or risk expensive litigation for wanting to pursue job opportunities.
For example, a comment from a physician working in Appalachia highlights the potential real-life implications of the agreements. “With hospital systems merging, providers with aggressive noncompetes must abandon the community that they serve if they [choose] to leave their employer. Healthcare providers feel trapped in their current employment situation, leading to significant burnout that can shorten their [career] longevity.”
Commissioner Alvaro Bedoya said physicians have had their lives upended by cumbersome noncompetes, often having to move out of state to practice. “A pandemic killed a million people in this country, and there are doctors who cannot work because of a noncompete,” he said.
It’s unclear whether physicians and others who work for nonprofit healthcare groups or hospitals will be covered by the new ban. FTC Commissioner Rebecca Slaughter acknowledged that the agency’s jurisdictional limitations mean that employees of “certain nonprofit organizations” may not benefit from the rule.
“We want to be transparent about the limitation and recognize there are workers, especially healthcare workers, who are bound by anticompetitive and unfair noncompete clauses, that our rule will struggle to reach,” she said. To cover nonprofit healthcare employees, Ms. Slaughter urged Congress to pass legislation banning noncompetes, such as the Workforce Mobility Act of 2021 and the Freedom to Compete Act of 2023.
The FTC final rule will take effect 120 days after it is published in the federal register, and new noncompete agreements will be banned as of this date. However, existing contracts for senior executives will remain in effect because these individuals are less likely to experience “acute harm” due to their ability to negotiate accordingly, said Mr. Cady.
States, AMA Take Aim at Noncompetes
Before the federal ban, several states had already passed legislation limiting the reach of noncompetes. According to a recent article in the Journal of the American College of Cardiology, 12 states prohibit noncompete clauses for physicians: Alabama, California, Colorado, Delaware, Massachusetts, Montana, New Hampshire, New Mexico, North Dakota, Oklahoma, Rhode Island, and South Dakota.
The remaining states allow noncompetes in some form, often excluding them for employees earning below a certain threshold. For example, in Oregon, noncompete agreements may apply to employees earning more than $113,241. Most states have provisions to adjust the threshold annually. The District of Columbia permits 2-year noncompetes for “medical specialists” earning over $250,000 annually.
Indiana employers can no longer enter into noncompete agreements with primary care providers. Other specialties may be subject to the clauses, except when the physician terminates the contract for cause or when an employer terminates the contract without cause.
Rachel Marcus, MD, a cardiologist in Washington, DC, found out how limiting her employment contract’s noncompete clause was when she wanted to leave a former position. Due to the restrictions, she told this news organization that she couldn’t work locally for a competitor for 2 years. The closest location she could seek employment without violating the agreement was Baltimore, approximately 40 miles away.
Dr. Marcus ultimately moved to another position within the same organization because of the company’s reputation for being “aggressive” in their enforcement actions.
Although the American Medical Association (AMA) does not support a total ban, its House of Delegates adopted policies last year to support the prohibition of noncompete contracts for physicians employed by for-profit or nonprofit hospitals, hospital systems, or staffing companies.
Challenges Await
The American Hospital Association, which opposed the proposed rule, called it “bad policy.” The decision “will likely be short-lived, with courts almost certain to stop it before it can do damage to hospitals’ ability to care for their patients and communities,” the association said in a statement.
To ease the transition to the new rule, the FTC also released a model language for employers to use when discussing the changes with their employees. “All employers need to do to comply with the rule is to stop enforcing existing noncompetes with workers other than senior executives and provide notice to such workers,” he said.
Dr. Marcus hopes the ban improves doctors’ lives. “Your employer is going to have to treat you better because they know that you can easily go across town to a place that has a higher salary, and your patient can go with you.”
A version of this article appeared on Medscape.com.
But business groups have vowed to challenge the decision in court.
The proposed final rule passed on a 3-2 vote, with the dissenting commissioners disputing the FTC’s authority to broadly ban noncompetes.
Tensions around noncompetes have been building for years. In 2021, President Biden issued an executive order supporting measures to improve economic competition, in which he urged the FTC to consider its rulemaking authority to address noncompete clauses that unfairly limit workers’ mobility. In January 2023, per that directive, the agency proposed ending the restrictive covenants.
While the FTC estimates that the final rule will reduce healthcare costs by up to $194 billion over the next decade and increase worker earnings by $300 million annually, the ruling faces legal hurdles.
US Chamber of Commerce president and CEO Suzanne P. Clark said in a statement that the move is a “blatant power grab” that will undermine competitive business practices, adding that the Chamber will sue to block the measure.
The FTC received more than 26,000 comments on noncompetes during the public feedback period, with about 25,000 supporting the measure, said Benjamin Cady, JD, an FTC attorney.
Mr. Cady called the feedback “compelling,” citing instances of workers who were forced to commute long distances, uproot their families, or risk expensive litigation for wanting to pursue job opportunities.
For example, a comment from a physician working in Appalachia highlights the potential real-life implications of the agreements. “With hospital systems merging, providers with aggressive noncompetes must abandon the community that they serve if they [choose] to leave their employer. Healthcare providers feel trapped in their current employment situation, leading to significant burnout that can shorten their [career] longevity.”
Commissioner Alvaro Bedoya said physicians have had their lives upended by cumbersome noncompetes, often having to move out of state to practice. “A pandemic killed a million people in this country, and there are doctors who cannot work because of a noncompete,” he said.
It’s unclear whether physicians and others who work for nonprofit healthcare groups or hospitals will be covered by the new ban. FTC Commissioner Rebecca Slaughter acknowledged that the agency’s jurisdictional limitations mean that employees of “certain nonprofit organizations” may not benefit from the rule.
“We want to be transparent about the limitation and recognize there are workers, especially healthcare workers, who are bound by anticompetitive and unfair noncompete clauses, that our rule will struggle to reach,” she said. To cover nonprofit healthcare employees, Ms. Slaughter urged Congress to pass legislation banning noncompetes, such as the Workforce Mobility Act of 2021 and the Freedom to Compete Act of 2023.
The FTC final rule will take effect 120 days after it is published in the federal register, and new noncompete agreements will be banned as of this date. However, existing contracts for senior executives will remain in effect because these individuals are less likely to experience “acute harm” due to their ability to negotiate accordingly, said Mr. Cady.
States, AMA Take Aim at Noncompetes
Before the federal ban, several states had already passed legislation limiting the reach of noncompetes. According to a recent article in the Journal of the American College of Cardiology, 12 states prohibit noncompete clauses for physicians: Alabama, California, Colorado, Delaware, Massachusetts, Montana, New Hampshire, New Mexico, North Dakota, Oklahoma, Rhode Island, and South Dakota.
The remaining states allow noncompetes in some form, often excluding them for employees earning below a certain threshold. For example, in Oregon, noncompete agreements may apply to employees earning more than $113,241. Most states have provisions to adjust the threshold annually. The District of Columbia permits 2-year noncompetes for “medical specialists” earning over $250,000 annually.
Indiana employers can no longer enter into noncompete agreements with primary care providers. Other specialties may be subject to the clauses, except when the physician terminates the contract for cause or when an employer terminates the contract without cause.
Rachel Marcus, MD, a cardiologist in Washington, DC, found out how limiting her employment contract’s noncompete clause was when she wanted to leave a former position. Due to the restrictions, she told this news organization that she couldn’t work locally for a competitor for 2 years. The closest location she could seek employment without violating the agreement was Baltimore, approximately 40 miles away.
Dr. Marcus ultimately moved to another position within the same organization because of the company’s reputation for being “aggressive” in their enforcement actions.
Although the American Medical Association (AMA) does not support a total ban, its House of Delegates adopted policies last year to support the prohibition of noncompete contracts for physicians employed by for-profit or nonprofit hospitals, hospital systems, or staffing companies.
Challenges Await
The American Hospital Association, which opposed the proposed rule, called it “bad policy.” The decision “will likely be short-lived, with courts almost certain to stop it before it can do damage to hospitals’ ability to care for their patients and communities,” the association said in a statement.
To ease the transition to the new rule, the FTC also released a model language for employers to use when discussing the changes with their employees. “All employers need to do to comply with the rule is to stop enforcing existing noncompetes with workers other than senior executives and provide notice to such workers,” he said.
Dr. Marcus hopes the ban improves doctors’ lives. “Your employer is going to have to treat you better because they know that you can easily go across town to a place that has a higher salary, and your patient can go with you.”
A version of this article appeared on Medscape.com.
Are Women Better Doctors Than Men?
This transcript has been edited for clarity.
It’s a battle of the sexes today as we dive into a paper that makes you say, “Wow, what an interesting study” and also “Boy, am I glad I didn’t do that study.” That’s because studies like this are always somewhat fraught; they say something about medicine but also something about society — and that makes this a bit precarious. But that’s never stopped us before. So, let’s go ahead and try to answer the question: Do women make better doctors than men?
On the surface, this question seems nearly impossible to answer. It’s too broad; what does it mean to be a “better” doctor? At first blush it seems that there are just too many variables to control for here: the type of doctor, the type of patient, the clinical scenario, and so on.
But this study, “Comparison of hospital mortality and readmission rates by physician and patient sex,” which appears in Annals of Internal Medicine, uses a fairly ingenious method to cut through all the bias by leveraging two simple facts: First, hospital medicine is largely conducted by hospitalists these days; second, due to the shift-based nature of hospitalist work, the hospitalist you get when you are admitted to the hospital is pretty much random.
In other words, if you are admitted to the hospital for an acute illness and get a hospitalist as your attending, you have no control over whether it is a man or a woman. Is this a randomized trial? No, but it’s not bad.
Researchers used Medicare claims data to identify adults over age 65 who had nonelective hospital admissions throughout the United States. The claims revealed the sex of the patient and the name of the attending physician. By linking to a medical provider database, they could determine the sex of the provider.
The goal was to look at outcomes across four dyads:
- Male patient – male doctor
- Male patient – female doctor
- Female patient – male doctor
- Female patient – female doctor
The primary outcome was 30-day mortality.
I told you that focusing on hospitalists produces some pseudorandomization, but let’s look at the data to be sure. Just under a million patients were treated by approximately 50,000 physicians, 30% of whom were female. And, though female patients and male patients differed, they did not differ with respect to the sex of their hospitalist. So, by physician sex, patients were similar in mean age, race, ethnicity, household income, eligibility for Medicaid, and comorbid conditions. The authors even created a “predicted mortality” score which was similar across the groups as well.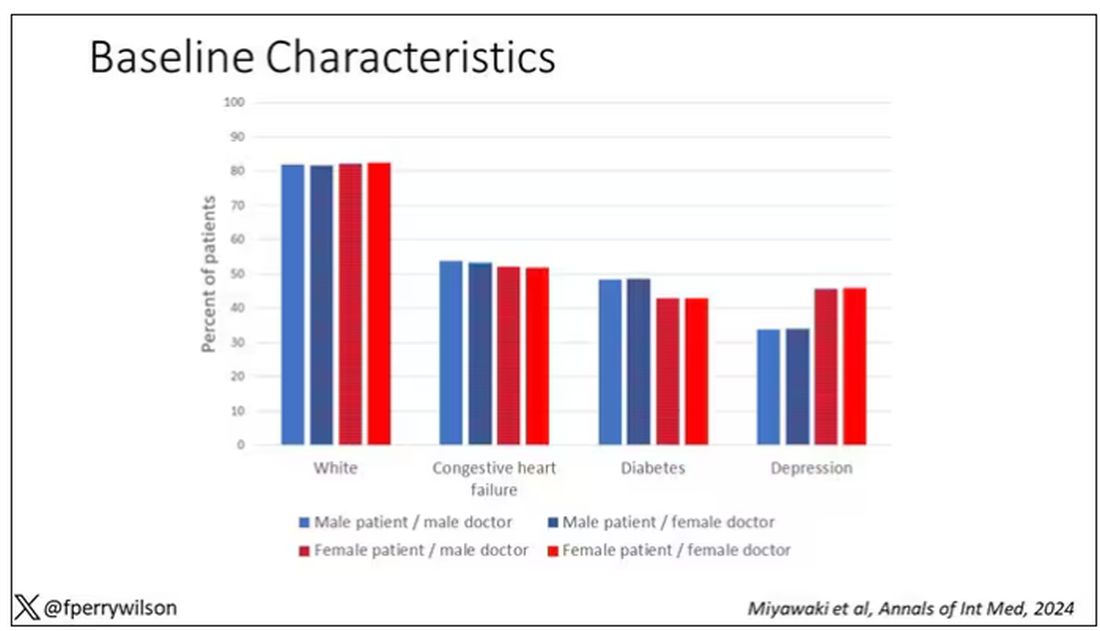
Now, the female physicians were a bit different from the male physicians. The female hospitalists were slightly more likely to have an osteopathic degree, had slightly fewer admissions per year, and were a bit younger.
So, we have broadly similar patients regardless of who their hospitalist was, but hospitalists differ by factors other than their sex. Fine.
I’ve graphed the results here. 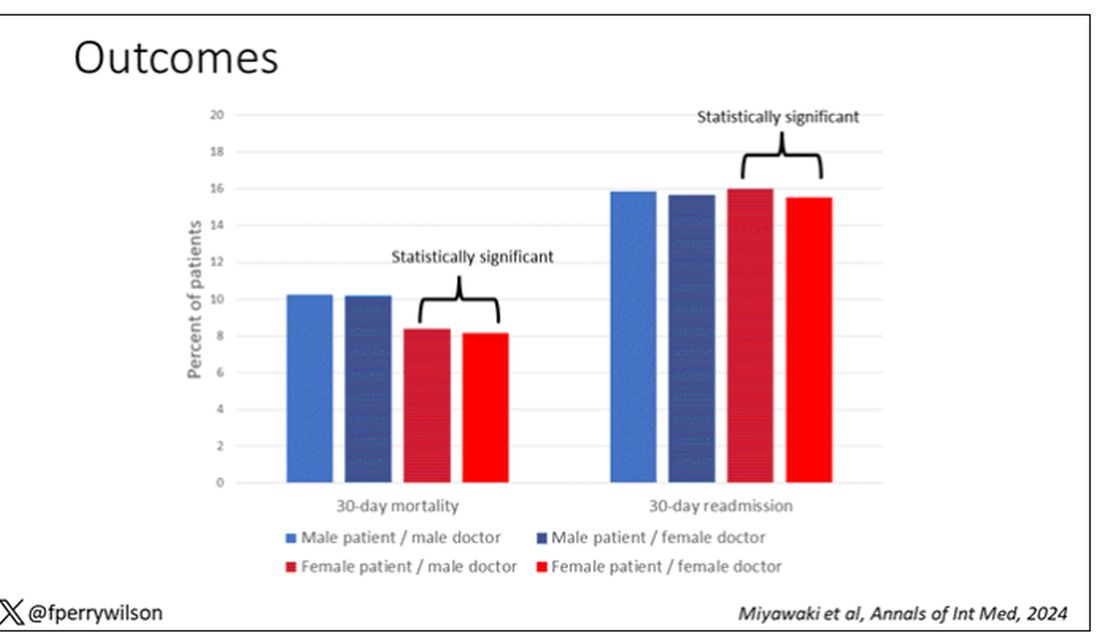
This is a relatively small effect, to be sure, but if you multiply it across the millions of hospitalist admissions per year, you can start to put up some real numbers.
So, what is going on here? I see four broad buckets of possibilities.
Let’s start with the obvious explanation: Women, on average, are better doctors than men. I am married to a woman doctor, and based on my personal experience, this explanation is undoubtedly true. But why would that be?
The authors cite data that suggest that female physicians are less likely than male physicians to dismiss patient concerns — and in particular, the concerns of female patients — perhaps leading to fewer missed diagnoses. But this is impossible to measure with administrative data, so this study can no more tell us whether these female hospitalists are more attentive than their male counterparts than it can suggest that the benefit is mediated by the shorter average height of female physicians. Perhaps the key is being closer to the patient?
The second possibility here is that this has nothing to do with the sex of the physician at all; it has to do with those other things that associate with the sex of the physician. We know, for example, that the female physicians saw fewer patients per year than the male physicians, but the study authors adjusted for this in the statistical models. Still, other unmeasured factors (confounders) could be present. By the way, confounders wouldn’t necessarily change the primary finding — you are better off being cared for by female physicians. It’s just not because they are female; it’s a convenient marker for some other quality, such as age.
The third possibility is that the study represents a phenomenon called collider bias. The idea here is that physicians only get into the study if they are hospitalists, and the quality of physicians who choose to become a hospitalist may differ by sex. When deciding on a specialty, a talented resident considering certain lifestyle issues may find hospital medicine particularly attractive — and that draw toward a more lifestyle-friendly specialty may differ by sex, as some prior studies have shown. If true, the pool of women hospitalists may be better than their male counterparts because male physicians of that caliber don’t become hospitalists.
Okay, don’t write in. I’m just trying to cite examples of how to think about collider bias. I can’t prove that this is the case, and in fact the authors do a sensitivity analysis of all physicians, not just hospitalists, and show the same thing. So this is probably not true, but epidemiology is fun, right?
And the fourth possibility: This is nothing but statistical noise. The effect size is incredibly small and just on the border of statistical significance. Especially when you’re working with very large datasets like this, you’ve got to be really careful about overinterpreting statistically significant findings that are nevertheless of small magnitude.
Regardless, it’s an interesting study, one that made me think and, of course, worry a bit about how I would present it. Forgive me if I’ve been indelicate in handling the complex issues of sex, gender, and society here. But I’m not sure what you expect; after all, I’m only a male doctor.
Dr. Wilson is associate professor of medicine and public health and director of the Clinical and Translational Research Accelerator at Yale University, New Haven, Conn. He has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
This transcript has been edited for clarity.
It’s a battle of the sexes today as we dive into a paper that makes you say, “Wow, what an interesting study” and also “Boy, am I glad I didn’t do that study.” That’s because studies like this are always somewhat fraught; they say something about medicine but also something about society — and that makes this a bit precarious. But that’s never stopped us before. So, let’s go ahead and try to answer the question: Do women make better doctors than men?
On the surface, this question seems nearly impossible to answer. It’s too broad; what does it mean to be a “better” doctor? At first blush it seems that there are just too many variables to control for here: the type of doctor, the type of patient, the clinical scenario, and so on.
But this study, “Comparison of hospital mortality and readmission rates by physician and patient sex,” which appears in Annals of Internal Medicine, uses a fairly ingenious method to cut through all the bias by leveraging two simple facts: First, hospital medicine is largely conducted by hospitalists these days; second, due to the shift-based nature of hospitalist work, the hospitalist you get when you are admitted to the hospital is pretty much random.
In other words, if you are admitted to the hospital for an acute illness and get a hospitalist as your attending, you have no control over whether it is a man or a woman. Is this a randomized trial? No, but it’s not bad.
Researchers used Medicare claims data to identify adults over age 65 who had nonelective hospital admissions throughout the United States. The claims revealed the sex of the patient and the name of the attending physician. By linking to a medical provider database, they could determine the sex of the provider.
The goal was to look at outcomes across four dyads:
- Male patient – male doctor
- Male patient – female doctor
- Female patient – male doctor
- Female patient – female doctor
The primary outcome was 30-day mortality.
I told you that focusing on hospitalists produces some pseudorandomization, but let’s look at the data to be sure. Just under a million patients were treated by approximately 50,000 physicians, 30% of whom were female. And, though female patients and male patients differed, they did not differ with respect to the sex of their hospitalist. So, by physician sex, patients were similar in mean age, race, ethnicity, household income, eligibility for Medicaid, and comorbid conditions. The authors even created a “predicted mortality” score which was similar across the groups as well.
Now, the female physicians were a bit different from the male physicians. The female hospitalists were slightly more likely to have an osteopathic degree, had slightly fewer admissions per year, and were a bit younger.
So, we have broadly similar patients regardless of who their hospitalist was, but hospitalists differ by factors other than their sex. Fine.
I’ve graphed the results here. 
This is a relatively small effect, to be sure, but if you multiply it across the millions of hospitalist admissions per year, you can start to put up some real numbers.
So, what is going on here? I see four broad buckets of possibilities.
Let’s start with the obvious explanation: Women, on average, are better doctors than men. I am married to a woman doctor, and based on my personal experience, this explanation is undoubtedly true. But why would that be?
The authors cite data that suggest that female physicians are less likely than male physicians to dismiss patient concerns — and in particular, the concerns of female patients — perhaps leading to fewer missed diagnoses. But this is impossible to measure with administrative data, so this study can no more tell us whether these female hospitalists are more attentive than their male counterparts than it can suggest that the benefit is mediated by the shorter average height of female physicians. Perhaps the key is being closer to the patient?
The second possibility here is that this has nothing to do with the sex of the physician at all; it has to do with those other things that associate with the sex of the physician. We know, for example, that the female physicians saw fewer patients per year than the male physicians, but the study authors adjusted for this in the statistical models. Still, other unmeasured factors (confounders) could be present. By the way, confounders wouldn’t necessarily change the primary finding — you are better off being cared for by female physicians. It’s just not because they are female; it’s a convenient marker for some other quality, such as age.
The third possibility is that the study represents a phenomenon called collider bias. The idea here is that physicians only get into the study if they are hospitalists, and the quality of physicians who choose to become a hospitalist may differ by sex. When deciding on a specialty, a talented resident considering certain lifestyle issues may find hospital medicine particularly attractive — and that draw toward a more lifestyle-friendly specialty may differ by sex, as some prior studies have shown. If true, the pool of women hospitalists may be better than their male counterparts because male physicians of that caliber don’t become hospitalists.
Okay, don’t write in. I’m just trying to cite examples of how to think about collider bias. I can’t prove that this is the case, and in fact the authors do a sensitivity analysis of all physicians, not just hospitalists, and show the same thing. So this is probably not true, but epidemiology is fun, right?
And the fourth possibility: This is nothing but statistical noise. The effect size is incredibly small and just on the border of statistical significance. Especially when you’re working with very large datasets like this, you’ve got to be really careful about overinterpreting statistically significant findings that are nevertheless of small magnitude.
Regardless, it’s an interesting study, one that made me think and, of course, worry a bit about how I would present it. Forgive me if I’ve been indelicate in handling the complex issues of sex, gender, and society here. But I’m not sure what you expect; after all, I’m only a male doctor.
Dr. Wilson is associate professor of medicine and public health and director of the Clinical and Translational Research Accelerator at Yale University, New Haven, Conn. He has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
This transcript has been edited for clarity.
It’s a battle of the sexes today as we dive into a paper that makes you say, “Wow, what an interesting study” and also “Boy, am I glad I didn’t do that study.” That’s because studies like this are always somewhat fraught; they say something about medicine but also something about society — and that makes this a bit precarious. But that’s never stopped us before. So, let’s go ahead and try to answer the question: Do women make better doctors than men?
On the surface, this question seems nearly impossible to answer. It’s too broad; what does it mean to be a “better” doctor? At first blush it seems that there are just too many variables to control for here: the type of doctor, the type of patient, the clinical scenario, and so on.
But this study, “Comparison of hospital mortality and readmission rates by physician and patient sex,” which appears in Annals of Internal Medicine, uses a fairly ingenious method to cut through all the bias by leveraging two simple facts: First, hospital medicine is largely conducted by hospitalists these days; second, due to the shift-based nature of hospitalist work, the hospitalist you get when you are admitted to the hospital is pretty much random.
In other words, if you are admitted to the hospital for an acute illness and get a hospitalist as your attending, you have no control over whether it is a man or a woman. Is this a randomized trial? No, but it’s not bad.
Researchers used Medicare claims data to identify adults over age 65 who had nonelective hospital admissions throughout the United States. The claims revealed the sex of the patient and the name of the attending physician. By linking to a medical provider database, they could determine the sex of the provider.
The goal was to look at outcomes across four dyads:
- Male patient – male doctor
- Male patient – female doctor
- Female patient – male doctor
- Female patient – female doctor
The primary outcome was 30-day mortality.
I told you that focusing on hospitalists produces some pseudorandomization, but let’s look at the data to be sure. Just under a million patients were treated by approximately 50,000 physicians, 30% of whom were female. And, though female patients and male patients differed, they did not differ with respect to the sex of their hospitalist. So, by physician sex, patients were similar in mean age, race, ethnicity, household income, eligibility for Medicaid, and comorbid conditions. The authors even created a “predicted mortality” score which was similar across the groups as well.
Now, the female physicians were a bit different from the male physicians. The female hospitalists were slightly more likely to have an osteopathic degree, had slightly fewer admissions per year, and were a bit younger.
So, we have broadly similar patients regardless of who their hospitalist was, but hospitalists differ by factors other than their sex. Fine.
I’ve graphed the results here. 
This is a relatively small effect, to be sure, but if you multiply it across the millions of hospitalist admissions per year, you can start to put up some real numbers.
So, what is going on here? I see four broad buckets of possibilities.
Let’s start with the obvious explanation: Women, on average, are better doctors than men. I am married to a woman doctor, and based on my personal experience, this explanation is undoubtedly true. But why would that be?
The authors cite data that suggest that female physicians are less likely than male physicians to dismiss patient concerns — and in particular, the concerns of female patients — perhaps leading to fewer missed diagnoses. But this is impossible to measure with administrative data, so this study can no more tell us whether these female hospitalists are more attentive than their male counterparts than it can suggest that the benefit is mediated by the shorter average height of female physicians. Perhaps the key is being closer to the patient?
The second possibility here is that this has nothing to do with the sex of the physician at all; it has to do with those other things that associate with the sex of the physician. We know, for example, that the female physicians saw fewer patients per year than the male physicians, but the study authors adjusted for this in the statistical models. Still, other unmeasured factors (confounders) could be present. By the way, confounders wouldn’t necessarily change the primary finding — you are better off being cared for by female physicians. It’s just not because they are female; it’s a convenient marker for some other quality, such as age.
The third possibility is that the study represents a phenomenon called collider bias. The idea here is that physicians only get into the study if they are hospitalists, and the quality of physicians who choose to become a hospitalist may differ by sex. When deciding on a specialty, a talented resident considering certain lifestyle issues may find hospital medicine particularly attractive — and that draw toward a more lifestyle-friendly specialty may differ by sex, as some prior studies have shown. If true, the pool of women hospitalists may be better than their male counterparts because male physicians of that caliber don’t become hospitalists.
Okay, don’t write in. I’m just trying to cite examples of how to think about collider bias. I can’t prove that this is the case, and in fact the authors do a sensitivity analysis of all physicians, not just hospitalists, and show the same thing. So this is probably not true, but epidemiology is fun, right?
And the fourth possibility: This is nothing but statistical noise. The effect size is incredibly small and just on the border of statistical significance. Especially when you’re working with very large datasets like this, you’ve got to be really careful about overinterpreting statistically significant findings that are nevertheless of small magnitude.
Regardless, it’s an interesting study, one that made me think and, of course, worry a bit about how I would present it. Forgive me if I’ve been indelicate in handling the complex issues of sex, gender, and society here. But I’m not sure what you expect; after all, I’m only a male doctor.
Dr. Wilson is associate professor of medicine and public health and director of the Clinical and Translational Research Accelerator at Yale University, New Haven, Conn. He has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
Adding Life to Your Patients’ Years
Caring for older adults was one of the most rewarding parts of my years practicing as a clinical cardiologist. I appreciated their wisdom, humor, and, very often, their respect and appreciation for physicians. It was always upsetting to see them suffer a mild fall or episode of atrial fibrillation and recognize that it could have major health ramifications.
That is not just a question for geriatric care. With fewer than two practicing geriatricians for every 10,000 older individuals, it is obvious that geriatricians cannot shoulder this responsibility alone. Almost all primary care physicians and subspecialists should prepare to care for older individuals and help them age healthfully.
Susan Friedman, MD, a board-certified geriatrics and lifestyle medicine clinician at the University of Rochester School of Medicine and Dentistry, Rochester, New York, reviewed the literature on the connection between lifestyle and healthy aging and concluded that the integration of lifestyle medicine into medical care for older adults is key to compressing morbidity. The pillars of lifestyle medicine — optimal nutrition, physical activity, stress management, restorative sleep, positive social connections, and avoidance of risky substances — both individually or as a sum are associated with less chronic disease, improved engagement in life, better physical and cognitive function, less frailty, and less sarcopenia. Framing discussions with patients around the six pillars of lifestyle medicine can be an effective strategy.
Optimal Nutrition
For a variety of reasons, older adults, especially those living alone, often lose the desire to prepare a nourishing meal. Older adults require different protein intake than younger patients to offset age-related sarcopenia, but helping them select healthy sources of protein is imperative. Both adequate protein consumption and eating patterns high in vegetables, legumes, fruit, and nuts and low in saturated fat, red meat, and processed meat can lower the risk of developing frailty.
Asking a patient to share a 24-hour food recall, and based upon that, resourcing nutritional guidance, a lifestyle medicine program or specialist, and insurance or community resources for food-as-medicine services, is a good first step.
Physical Activity
Increasing general physical activity can be a tough ask for many older adults, and joint pain is a common reason they demur. Messaging around targeted exercises to mitigate falls, improve muscle strength, and reduce joint pain may be more appealing. Contemporary research demonstrates that exercise, particularly open-skill exercise that requires quick decisions (such as table tennis) can be powerful. Maintaining cognition, mood enhancement, and independence may also be motivating messages.
The first step is curiosity: What does your patient like to do? Referral to a physical therapist or an exercise specialist to provide stepwise guidance along with resourcing community opportunities can then follow.
Restorative Sleep
“I’m old. I don’t need as much sleep.” We’ve probably all heard older patients say this. But the National Sleep Foundation’s report on sleep health and aging indicates that the need to sleep does not decrease with age. The ability to sleep, however, may decline. Assessing and treating disordered sleep is another example of how each lifestyle medicine pillar, such as nutrition and physical activity, is multidimensional and interacts to support the functional integrity of older patients. It’s hard to feel motivated to go for a walk if you lack adequate sleep.
Stress Management
Exploring stress with patients can be very revealing. Do they experience stress that energizes and has a positive effect? How much of their day is spent in negatively impactful distress? Chronic stress has been shown to affect immune function in older individuals. Start conversations with your older patients to normalize the importance of stress as a health measure.
Positive Social Connections
Loneliness puts individuals at higher risk for heart disease, stroke, and dementia and even increases the risk for premature death by up to 60%. Yet, clinicians and patients rarely discuss social connections during medical appointments. Tools such as the UCLA Loneliness Scale exist for health practitioners to assess and identify patients at risk for loneliness, as do resources to integrate social care into the delivery of healthcare.
Avoidance of Risky Substances
Alcohol assessments are not just for younger patients. One study found that 5.6 million adults ages 65 or older engaged in binge drinking in the past month. Because of body changes, the negative effects of alcohol may be greater on older adults, including interactions between alcohol and commonly prescribed medications.
Conducting a lifestyle assessment is an important way to engage with older patients and allows clinicians to identify opportunities to improve health behaviors, understand obstacles, and support patients to make lifestyle changes. It may uncover ways to remove some of the pill and treatment burdens that older adults often experience. The American College of Lifestyle Medicine (ACLM) offers clinical practice resources to support clinicians as well as “Lifestyle Medicine and Food as Medicine Essentials,” a 5.5-hour complimentary CE/CME course on food and lifestyle medicine that introduces clinicians to the therapeutic use of lifestyle medicine. ACLM also offers members interest groups focused on geriatrics, fitness, and mental health, which may be beneficial to clinicians treating older adults.
By engaging with older patients on their lifestyle behaviors, we can ensure that we are doing all we can to help them live longer — and live better.
Dr. Collings is director of lifestyle medicine, Silicon Valley Medical Development, and past president, American College of Lifestyle Medicine, Mountain View, California. She has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
Caring for older adults was one of the most rewarding parts of my years practicing as a clinical cardiologist. I appreciated their wisdom, humor, and, very often, their respect and appreciation for physicians. It was always upsetting to see them suffer a mild fall or episode of atrial fibrillation and recognize that it could have major health ramifications.
That is not just a question for geriatric care. With fewer than two practicing geriatricians for every 10,000 older individuals, it is obvious that geriatricians cannot shoulder this responsibility alone. Almost all primary care physicians and subspecialists should prepare to care for older individuals and help them age healthfully.
Susan Friedman, MD, a board-certified geriatrics and lifestyle medicine clinician at the University of Rochester School of Medicine and Dentistry, Rochester, New York, reviewed the literature on the connection between lifestyle and healthy aging and concluded that the integration of lifestyle medicine into medical care for older adults is key to compressing morbidity. The pillars of lifestyle medicine — optimal nutrition, physical activity, stress management, restorative sleep, positive social connections, and avoidance of risky substances — both individually or as a sum are associated with less chronic disease, improved engagement in life, better physical and cognitive function, less frailty, and less sarcopenia. Framing discussions with patients around the six pillars of lifestyle medicine can be an effective strategy.
Optimal Nutrition
For a variety of reasons, older adults, especially those living alone, often lose the desire to prepare a nourishing meal. Older adults require different protein intake than younger patients to offset age-related sarcopenia, but helping them select healthy sources of protein is imperative. Both adequate protein consumption and eating patterns high in vegetables, legumes, fruit, and nuts and low in saturated fat, red meat, and processed meat can lower the risk of developing frailty.
Asking a patient to share a 24-hour food recall, and based upon that, resourcing nutritional guidance, a lifestyle medicine program or specialist, and insurance or community resources for food-as-medicine services, is a good first step.
Physical Activity
Increasing general physical activity can be a tough ask for many older adults, and joint pain is a common reason they demur. Messaging around targeted exercises to mitigate falls, improve muscle strength, and reduce joint pain may be more appealing. Contemporary research demonstrates that exercise, particularly open-skill exercise that requires quick decisions (such as table tennis) can be powerful. Maintaining cognition, mood enhancement, and independence may also be motivating messages.
The first step is curiosity: What does your patient like to do? Referral to a physical therapist or an exercise specialist to provide stepwise guidance along with resourcing community opportunities can then follow.
Restorative Sleep
“I’m old. I don’t need as much sleep.” We’ve probably all heard older patients say this. But the National Sleep Foundation’s report on sleep health and aging indicates that the need to sleep does not decrease with age. The ability to sleep, however, may decline. Assessing and treating disordered sleep is another example of how each lifestyle medicine pillar, such as nutrition and physical activity, is multidimensional and interacts to support the functional integrity of older patients. It’s hard to feel motivated to go for a walk if you lack adequate sleep.
Stress Management
Exploring stress with patients can be very revealing. Do they experience stress that energizes and has a positive effect? How much of their day is spent in negatively impactful distress? Chronic stress has been shown to affect immune function in older individuals. Start conversations with your older patients to normalize the importance of stress as a health measure.
Positive Social Connections
Loneliness puts individuals at higher risk for heart disease, stroke, and dementia and even increases the risk for premature death by up to 60%. Yet, clinicians and patients rarely discuss social connections during medical appointments. Tools such as the UCLA Loneliness Scale exist for health practitioners to assess and identify patients at risk for loneliness, as do resources to integrate social care into the delivery of healthcare.
Avoidance of Risky Substances
Alcohol assessments are not just for younger patients. One study found that 5.6 million adults ages 65 or older engaged in binge drinking in the past month. Because of body changes, the negative effects of alcohol may be greater on older adults, including interactions between alcohol and commonly prescribed medications.
Conducting a lifestyle assessment is an important way to engage with older patients and allows clinicians to identify opportunities to improve health behaviors, understand obstacles, and support patients to make lifestyle changes. It may uncover ways to remove some of the pill and treatment burdens that older adults often experience. The American College of Lifestyle Medicine (ACLM) offers clinical practice resources to support clinicians as well as “Lifestyle Medicine and Food as Medicine Essentials,” a 5.5-hour complimentary CE/CME course on food and lifestyle medicine that introduces clinicians to the therapeutic use of lifestyle medicine. ACLM also offers members interest groups focused on geriatrics, fitness, and mental health, which may be beneficial to clinicians treating older adults.
By engaging with older patients on their lifestyle behaviors, we can ensure that we are doing all we can to help them live longer — and live better.
Dr. Collings is director of lifestyle medicine, Silicon Valley Medical Development, and past president, American College of Lifestyle Medicine, Mountain View, California. She has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
Caring for older adults was one of the most rewarding parts of my years practicing as a clinical cardiologist. I appreciated their wisdom, humor, and, very often, their respect and appreciation for physicians. It was always upsetting to see them suffer a mild fall or episode of atrial fibrillation and recognize that it could have major health ramifications.
That is not just a question for geriatric care. With fewer than two practicing geriatricians for every 10,000 older individuals, it is obvious that geriatricians cannot shoulder this responsibility alone. Almost all primary care physicians and subspecialists should prepare to care for older individuals and help them age healthfully.
Susan Friedman, MD, a board-certified geriatrics and lifestyle medicine clinician at the University of Rochester School of Medicine and Dentistry, Rochester, New York, reviewed the literature on the connection between lifestyle and healthy aging and concluded that the integration of lifestyle medicine into medical care for older adults is key to compressing morbidity. The pillars of lifestyle medicine — optimal nutrition, physical activity, stress management, restorative sleep, positive social connections, and avoidance of risky substances — both individually or as a sum are associated with less chronic disease, improved engagement in life, better physical and cognitive function, less frailty, and less sarcopenia. Framing discussions with patients around the six pillars of lifestyle medicine can be an effective strategy.
Optimal Nutrition
For a variety of reasons, older adults, especially those living alone, often lose the desire to prepare a nourishing meal. Older adults require different protein intake than younger patients to offset age-related sarcopenia, but helping them select healthy sources of protein is imperative. Both adequate protein consumption and eating patterns high in vegetables, legumes, fruit, and nuts and low in saturated fat, red meat, and processed meat can lower the risk of developing frailty.
Asking a patient to share a 24-hour food recall, and based upon that, resourcing nutritional guidance, a lifestyle medicine program or specialist, and insurance or community resources for food-as-medicine services, is a good first step.
Physical Activity
Increasing general physical activity can be a tough ask for many older adults, and joint pain is a common reason they demur. Messaging around targeted exercises to mitigate falls, improve muscle strength, and reduce joint pain may be more appealing. Contemporary research demonstrates that exercise, particularly open-skill exercise that requires quick decisions (such as table tennis) can be powerful. Maintaining cognition, mood enhancement, and independence may also be motivating messages.
The first step is curiosity: What does your patient like to do? Referral to a physical therapist or an exercise specialist to provide stepwise guidance along with resourcing community opportunities can then follow.
Restorative Sleep
“I’m old. I don’t need as much sleep.” We’ve probably all heard older patients say this. But the National Sleep Foundation’s report on sleep health and aging indicates that the need to sleep does not decrease with age. The ability to sleep, however, may decline. Assessing and treating disordered sleep is another example of how each lifestyle medicine pillar, such as nutrition and physical activity, is multidimensional and interacts to support the functional integrity of older patients. It’s hard to feel motivated to go for a walk if you lack adequate sleep.
Stress Management
Exploring stress with patients can be very revealing. Do they experience stress that energizes and has a positive effect? How much of their day is spent in negatively impactful distress? Chronic stress has been shown to affect immune function in older individuals. Start conversations with your older patients to normalize the importance of stress as a health measure.
Positive Social Connections
Loneliness puts individuals at higher risk for heart disease, stroke, and dementia and even increases the risk for premature death by up to 60%. Yet, clinicians and patients rarely discuss social connections during medical appointments. Tools such as the UCLA Loneliness Scale exist for health practitioners to assess and identify patients at risk for loneliness, as do resources to integrate social care into the delivery of healthcare.
Avoidance of Risky Substances
Alcohol assessments are not just for younger patients. One study found that 5.6 million adults ages 65 or older engaged in binge drinking in the past month. Because of body changes, the negative effects of alcohol may be greater on older adults, including interactions between alcohol and commonly prescribed medications.
Conducting a lifestyle assessment is an important way to engage with older patients and allows clinicians to identify opportunities to improve health behaviors, understand obstacles, and support patients to make lifestyle changes. It may uncover ways to remove some of the pill and treatment burdens that older adults often experience. The American College of Lifestyle Medicine (ACLM) offers clinical practice resources to support clinicians as well as “Lifestyle Medicine and Food as Medicine Essentials,” a 5.5-hour complimentary CE/CME course on food and lifestyle medicine that introduces clinicians to the therapeutic use of lifestyle medicine. ACLM also offers members interest groups focused on geriatrics, fitness, and mental health, which may be beneficial to clinicians treating older adults.
By engaging with older patients on their lifestyle behaviors, we can ensure that we are doing all we can to help them live longer — and live better.
Dr. Collings is director of lifestyle medicine, Silicon Valley Medical Development, and past president, American College of Lifestyle Medicine, Mountain View, California. She has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.