User login
ID Practitioner is an independent news source that provides infectious disease specialists with timely and relevant news and commentary about clinical developments and the impact of health care policy on the infectious disease specialist’s practice. Specialty focus topics include antimicrobial resistance, emerging infections, global ID, hepatitis, HIV, hospital-acquired infections, immunizations and vaccines, influenza, mycoses, pediatric infections, and STIs. Infectious Diseases News is owned by Frontline Medical Communications.
sofosbuvir
ritonavir with dasabuvir
discount
support path
program
ritonavir
greedy
ledipasvir
assistance
viekira pak
vpak
advocacy
needy
protest
abbvie
paritaprevir
ombitasvir
direct-acting antivirals
dasabuvir
gilead
fake-ovir
support
v pak
oasis
harvoni
section[contains(@class, 'footer-nav-section-wrapper')]
div[contains(@class, 'pane-pub-article-idp')]
div[contains(@class, 'pane-medstat-latest-articles-articles-section')]
div[contains(@class, 'pane-pub-home-idp')]
div[contains(@class, 'pane-pub-topic-idp')]
Children and COVID: New cases climb slowly but steadily
The current sustained increase in COVID-19 has brought the total number of cases in children to over 13 million since the start of the pandemic, according to the American Academy of Pediatrics and the Children’s Hospital Association.
, when cases dropped to their lowest point since last summer. The cumulative number of cases in children is 13,052,988, which accounts for 19.0% of all cases reported in the United States, the AAP and CHA said in their weekly COVID-19 report.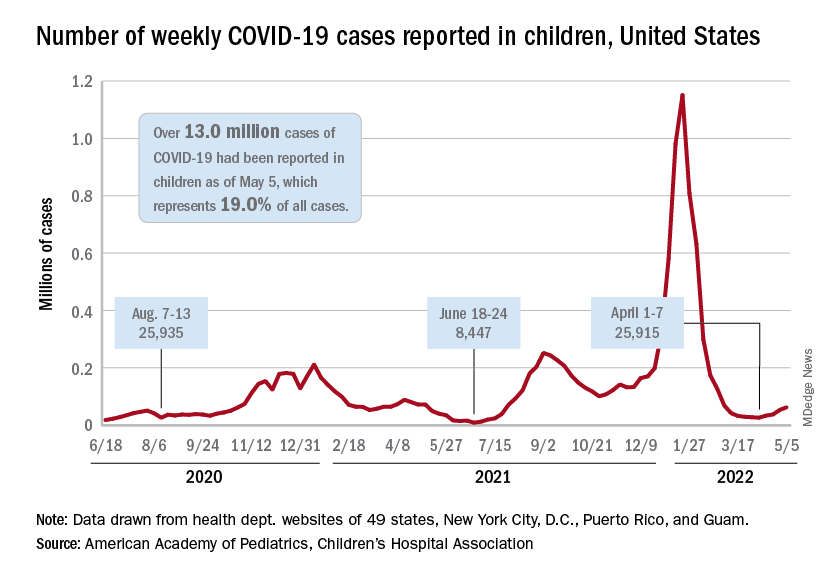
Other measures of incidence show the same steady rise. The rate of new admissions of children aged 0-17 with confirmed COVID-19, which had dipped as low as 0.13 per 100,000 population on April 11, was up to 0.19 per 100,000 on May 6, and the 7-day average for total admissions was 136 per day for May 1-7, compared with 118 for the last week of April, according to the Centers for Disease Control and Prevention.
At the state level, new admission rates for May 6 show wide variation, even regionally. Rhode Island came in with a 0.00 per 100,000 on that day, while Vermont recorded 0.88 admissions per 100,000, the highest of any state and lower only than the District of Columbia’s 1.23 per 100,000. Connecticut (0.45) and Massachusetts (0.33) also were in the highest group (see map), while Maine was in the lowest, CDC data show.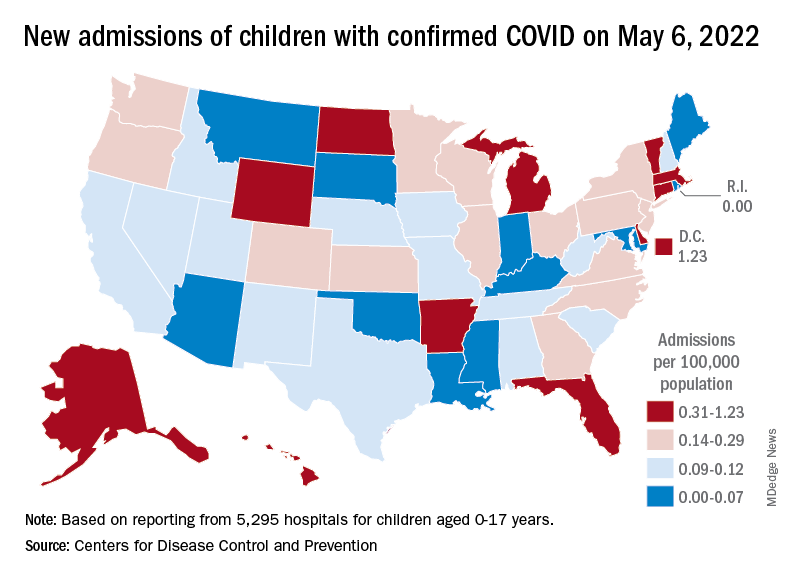
Nationally, emergency department visits also have been rising over the last month or so. Children aged 0-11 years, who were down to a 7-day average of 0.5% of ED visits with diagnosed COVID-19 in early April, saw that number rise to 1.4% on May 5. Children aged 12-15 years went from a rate of 0.3% in late March to the current 1.2%, as did 16- to 17-year-olds, the CDC said on its COVID Data Tracker.
The vaccination effort, meanwhile, continues to lose steam, at least among children who are currently eligible. Initial vaccinations in those aged 5-11 slipped to their lowest-ever 1-week total, 47,000 for April 28 to May 4, while children aged 16-17 continued a long-term slide that has the weekly count down to just 29,000, the AAP said in its weekly vaccination report.
Here’s how those latest recipients changed the populations of vaccinated children in the last week: 35.4% of all 5- to 11-year-olds had received at least one dose as of May 4, compared with 35.3% on April 27, with increases from 67.4% to 67.5% for 12- to 15-year-olds and 72.7% to 72.8% among those aged 16-17, the CDC reported.
The current sustained increase in COVID-19 has brought the total number of cases in children to over 13 million since the start of the pandemic, according to the American Academy of Pediatrics and the Children’s Hospital Association.
, when cases dropped to their lowest point since last summer. The cumulative number of cases in children is 13,052,988, which accounts for 19.0% of all cases reported in the United States, the AAP and CHA said in their weekly COVID-19 report.
Other measures of incidence show the same steady rise. The rate of new admissions of children aged 0-17 with confirmed COVID-19, which had dipped as low as 0.13 per 100,000 population on April 11, was up to 0.19 per 100,000 on May 6, and the 7-day average for total admissions was 136 per day for May 1-7, compared with 118 for the last week of April, according to the Centers for Disease Control and Prevention.
At the state level, new admission rates for May 6 show wide variation, even regionally. Rhode Island came in with a 0.00 per 100,000 on that day, while Vermont recorded 0.88 admissions per 100,000, the highest of any state and lower only than the District of Columbia’s 1.23 per 100,000. Connecticut (0.45) and Massachusetts (0.33) also were in the highest group (see map), while Maine was in the lowest, CDC data show.
Nationally, emergency department visits also have been rising over the last month or so. Children aged 0-11 years, who were down to a 7-day average of 0.5% of ED visits with diagnosed COVID-19 in early April, saw that number rise to 1.4% on May 5. Children aged 12-15 years went from a rate of 0.3% in late March to the current 1.2%, as did 16- to 17-year-olds, the CDC said on its COVID Data Tracker.
The vaccination effort, meanwhile, continues to lose steam, at least among children who are currently eligible. Initial vaccinations in those aged 5-11 slipped to their lowest-ever 1-week total, 47,000 for April 28 to May 4, while children aged 16-17 continued a long-term slide that has the weekly count down to just 29,000, the AAP said in its weekly vaccination report.
Here’s how those latest recipients changed the populations of vaccinated children in the last week: 35.4% of all 5- to 11-year-olds had received at least one dose as of May 4, compared with 35.3% on April 27, with increases from 67.4% to 67.5% for 12- to 15-year-olds and 72.7% to 72.8% among those aged 16-17, the CDC reported.
The current sustained increase in COVID-19 has brought the total number of cases in children to over 13 million since the start of the pandemic, according to the American Academy of Pediatrics and the Children’s Hospital Association.
, when cases dropped to their lowest point since last summer. The cumulative number of cases in children is 13,052,988, which accounts for 19.0% of all cases reported in the United States, the AAP and CHA said in their weekly COVID-19 report.
Other measures of incidence show the same steady rise. The rate of new admissions of children aged 0-17 with confirmed COVID-19, which had dipped as low as 0.13 per 100,000 population on April 11, was up to 0.19 per 100,000 on May 6, and the 7-day average for total admissions was 136 per day for May 1-7, compared with 118 for the last week of April, according to the Centers for Disease Control and Prevention.
At the state level, new admission rates for May 6 show wide variation, even regionally. Rhode Island came in with a 0.00 per 100,000 on that day, while Vermont recorded 0.88 admissions per 100,000, the highest of any state and lower only than the District of Columbia’s 1.23 per 100,000. Connecticut (0.45) and Massachusetts (0.33) also were in the highest group (see map), while Maine was in the lowest, CDC data show.
Nationally, emergency department visits also have been rising over the last month or so. Children aged 0-11 years, who were down to a 7-day average of 0.5% of ED visits with diagnosed COVID-19 in early April, saw that number rise to 1.4% on May 5. Children aged 12-15 years went from a rate of 0.3% in late March to the current 1.2%, as did 16- to 17-year-olds, the CDC said on its COVID Data Tracker.
The vaccination effort, meanwhile, continues to lose steam, at least among children who are currently eligible. Initial vaccinations in those aged 5-11 slipped to their lowest-ever 1-week total, 47,000 for April 28 to May 4, while children aged 16-17 continued a long-term slide that has the weekly count down to just 29,000, the AAP said in its weekly vaccination report.
Here’s how those latest recipients changed the populations of vaccinated children in the last week: 35.4% of all 5- to 11-year-olds had received at least one dose as of May 4, compared with 35.3% on April 27, with increases from 67.4% to 67.5% for 12- to 15-year-olds and 72.7% to 72.8% among those aged 16-17, the CDC reported.
My choice? Unvaccinated pose outsize risk to vaccinated
according to a mathematical modeling study.
The study, which simulated patterns of infection among vaccinated and unvaccinated populations, showed that, as the populations mixed less, attack rates decreased among vaccinated people (from 15% to 10%) and increased among unvaccinated people (from 62% to 79%). The unvaccinated increasingly became the source of infection, however.
“When the vaccinated and unvaccinated mix, indirect protection is conferred upon the unvaccinated by the buffering effect of vaccinated individuals, and by contrast, risk in the vaccinated goes up,” lead author David Fisman, MD, professor of epidemiology at the University of Toronto, told this news organization.
As the groups mix less and less, the size of the epidemic increases among the unvaccinated and decreases among the vaccinated. “But the impact of the unvaccinated on risk in the vaccinated is disproportionate to the numbers of contacts between the two groups,” said Dr. Fisman.
The study was published online in the Canadian Medical Association Journal.
Relative contributions to risk
The researchers used a model of a respiratory viral disease “similar to SARS-CoV-2 infection with Delta variant.” They included reproduction values to capture the dynamics of the Omicron variant, which was emerging at the time. In the study, vaccines ranged in effectiveness from 40% to 80%. The study incorporated various levels of mixing between a partially vaccinated and an unvaccinated population. The mixing ranged from random mixing to like-with-like mixing (“assortativity”). There were three possible “compartments” of people in the model: those considered susceptible to infection, those considered infected and infectious, and those considered immune because of recovery.
The model showed that, as mixing between the vaccinated and the unvaccinated populations increased, case numbers rose, “with cases in the unvaccinated subpopulation accounting for a substantial proportion of infections.” However, as mixing between the populations decreased, the final attack rate decreased among vaccinated people, but the relative “contribution of risk to vaccinated people caused by infection acquired from contact with unvaccinated people ... increased.”
When the vaccination rate was increased in the model, case numbers among the vaccinated declined “as expected, owing to indirect protective effects,” the researchers noted. But this also “further increased the relative contribution to risk in vaccinated people by those who were unvaccinated.”
Self-regarding risk?
The findings show that “choices made by people who forgo vaccination contribute disproportionately to risk among those who do get vaccinated,” the researchers wrote. “Although risk associated with avoiding vaccination during a virulent pandemic accrues chiefly to those who are unvaccinated, the choice of some individuals to refuse vaccination is likely to affect the health and safety of vaccinated people in a manner disproportionate to the fraction of unvaccinated people in the population.”
The fact that like-with-like mixing cannot mitigate the risk to vaccinated people “undermines the assertion that vaccine choice is best left to the individual and supports strong public actions aimed at enhancing vaccine uptake and limiting access to public spaces for unvaccinated people,” they wrote.
Mandates and passports
“Our model provides support for vaccine mandates and passports during epidemics, such that vaccination is required for people to take part in nonessential activities,” said Dr. Fisman. The choice to not be vaccinated against COVID-19 should not be considered “self-regarding,” he added. “Risk is self-regarding when it only impacts the person engaging in the activity. Something like smoking cigarettes (alone, without others around) creates a lot of risk over time, but if nobody is breathing your secondhand smoke, you’re only creating risk for yourself. By contrast, we regulate, in Ontario, your right to smoke in public indoor spaces such as restaurants, because once other people are around, the risk isn’t self-regarding anymore. You’re creating risk for others.”
The authors also noted that the risks created by the unvaccinated extend beyond those of infection by “creating a risk that those around them may not be able to obtain the care they need.” They recommended that considerations of equity and justice for people who do choose to be vaccinated, as well as those who choose not to be, need to be included in formulating vaccination policy.
Illuminating the discussion
Asked to comment on the study, Matthew Oughton, MD, assistant professor of medicine at McGill University, Montreal, said: “It is easy to dismiss a mathematical model as a series of assumptions that leads to an implausible conclusion. ... However, they can serve to illustrate and, to an extent, quantify the results of complex interactions, and this study does just that.” Dr. Oughton was not involved in the research.
During the past 2 years, the scientific press and the general press have often discussed the individual and collective effects of disease-prevention methods, including nonpharmaceutical interventions. “Models like this can help illuminate those discussions by highlighting important consequences of preventive measures,” said Dr. Oughton, who also works in the division of infectious diseases at the Jewish General Hospital, Montreal.
It’s worth noting that the authors modeled vaccine effectiveness against all infection, “rather than the generally greater and more durable effects we have seen for vaccines in prevention of severe infection,” said Dr. Oughton. He added that the authors did not include the effect of vaccination in reducing forward transmission. “Inclusion of this effect would presumably have reduced overall infectious burden in mixed populations and increased the difference between groups at lower levels of mixing between populations.”
The research was supported by a grant from the Canadian Institutes of Health Research. Dr. Fisman has served on advisory boards related to influenza and SARS-CoV-2 vaccines for Seqirus, Pfizer, AstraZeneca, and Sanofi-Pasteur Vaccines and has served as a legal expert on issues related to COVID-19 epidemiology for the Elementary Teachers Federation of Ontario and the Registered Nurses Association of Ontario. Dr. Oughton disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
according to a mathematical modeling study.
The study, which simulated patterns of infection among vaccinated and unvaccinated populations, showed that, as the populations mixed less, attack rates decreased among vaccinated people (from 15% to 10%) and increased among unvaccinated people (from 62% to 79%). The unvaccinated increasingly became the source of infection, however.
“When the vaccinated and unvaccinated mix, indirect protection is conferred upon the unvaccinated by the buffering effect of vaccinated individuals, and by contrast, risk in the vaccinated goes up,” lead author David Fisman, MD, professor of epidemiology at the University of Toronto, told this news organization.
As the groups mix less and less, the size of the epidemic increases among the unvaccinated and decreases among the vaccinated. “But the impact of the unvaccinated on risk in the vaccinated is disproportionate to the numbers of contacts between the two groups,” said Dr. Fisman.
The study was published online in the Canadian Medical Association Journal.
Relative contributions to risk
The researchers used a model of a respiratory viral disease “similar to SARS-CoV-2 infection with Delta variant.” They included reproduction values to capture the dynamics of the Omicron variant, which was emerging at the time. In the study, vaccines ranged in effectiveness from 40% to 80%. The study incorporated various levels of mixing between a partially vaccinated and an unvaccinated population. The mixing ranged from random mixing to like-with-like mixing (“assortativity”). There were three possible “compartments” of people in the model: those considered susceptible to infection, those considered infected and infectious, and those considered immune because of recovery.
The model showed that, as mixing between the vaccinated and the unvaccinated populations increased, case numbers rose, “with cases in the unvaccinated subpopulation accounting for a substantial proportion of infections.” However, as mixing between the populations decreased, the final attack rate decreased among vaccinated people, but the relative “contribution of risk to vaccinated people caused by infection acquired from contact with unvaccinated people ... increased.”
When the vaccination rate was increased in the model, case numbers among the vaccinated declined “as expected, owing to indirect protective effects,” the researchers noted. But this also “further increased the relative contribution to risk in vaccinated people by those who were unvaccinated.”
Self-regarding risk?
The findings show that “choices made by people who forgo vaccination contribute disproportionately to risk among those who do get vaccinated,” the researchers wrote. “Although risk associated with avoiding vaccination during a virulent pandemic accrues chiefly to those who are unvaccinated, the choice of some individuals to refuse vaccination is likely to affect the health and safety of vaccinated people in a manner disproportionate to the fraction of unvaccinated people in the population.”
The fact that like-with-like mixing cannot mitigate the risk to vaccinated people “undermines the assertion that vaccine choice is best left to the individual and supports strong public actions aimed at enhancing vaccine uptake and limiting access to public spaces for unvaccinated people,” they wrote.
Mandates and passports
“Our model provides support for vaccine mandates and passports during epidemics, such that vaccination is required for people to take part in nonessential activities,” said Dr. Fisman. The choice to not be vaccinated against COVID-19 should not be considered “self-regarding,” he added. “Risk is self-regarding when it only impacts the person engaging in the activity. Something like smoking cigarettes (alone, without others around) creates a lot of risk over time, but if nobody is breathing your secondhand smoke, you’re only creating risk for yourself. By contrast, we regulate, in Ontario, your right to smoke in public indoor spaces such as restaurants, because once other people are around, the risk isn’t self-regarding anymore. You’re creating risk for others.”
The authors also noted that the risks created by the unvaccinated extend beyond those of infection by “creating a risk that those around them may not be able to obtain the care they need.” They recommended that considerations of equity and justice for people who do choose to be vaccinated, as well as those who choose not to be, need to be included in formulating vaccination policy.
Illuminating the discussion
Asked to comment on the study, Matthew Oughton, MD, assistant professor of medicine at McGill University, Montreal, said: “It is easy to dismiss a mathematical model as a series of assumptions that leads to an implausible conclusion. ... However, they can serve to illustrate and, to an extent, quantify the results of complex interactions, and this study does just that.” Dr. Oughton was not involved in the research.
During the past 2 years, the scientific press and the general press have often discussed the individual and collective effects of disease-prevention methods, including nonpharmaceutical interventions. “Models like this can help illuminate those discussions by highlighting important consequences of preventive measures,” said Dr. Oughton, who also works in the division of infectious diseases at the Jewish General Hospital, Montreal.
It’s worth noting that the authors modeled vaccine effectiveness against all infection, “rather than the generally greater and more durable effects we have seen for vaccines in prevention of severe infection,” said Dr. Oughton. He added that the authors did not include the effect of vaccination in reducing forward transmission. “Inclusion of this effect would presumably have reduced overall infectious burden in mixed populations and increased the difference between groups at lower levels of mixing between populations.”
The research was supported by a grant from the Canadian Institutes of Health Research. Dr. Fisman has served on advisory boards related to influenza and SARS-CoV-2 vaccines for Seqirus, Pfizer, AstraZeneca, and Sanofi-Pasteur Vaccines and has served as a legal expert on issues related to COVID-19 epidemiology for the Elementary Teachers Federation of Ontario and the Registered Nurses Association of Ontario. Dr. Oughton disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
according to a mathematical modeling study.
The study, which simulated patterns of infection among vaccinated and unvaccinated populations, showed that, as the populations mixed less, attack rates decreased among vaccinated people (from 15% to 10%) and increased among unvaccinated people (from 62% to 79%). The unvaccinated increasingly became the source of infection, however.
“When the vaccinated and unvaccinated mix, indirect protection is conferred upon the unvaccinated by the buffering effect of vaccinated individuals, and by contrast, risk in the vaccinated goes up,” lead author David Fisman, MD, professor of epidemiology at the University of Toronto, told this news organization.
As the groups mix less and less, the size of the epidemic increases among the unvaccinated and decreases among the vaccinated. “But the impact of the unvaccinated on risk in the vaccinated is disproportionate to the numbers of contacts between the two groups,” said Dr. Fisman.
The study was published online in the Canadian Medical Association Journal.
Relative contributions to risk
The researchers used a model of a respiratory viral disease “similar to SARS-CoV-2 infection with Delta variant.” They included reproduction values to capture the dynamics of the Omicron variant, which was emerging at the time. In the study, vaccines ranged in effectiveness from 40% to 80%. The study incorporated various levels of mixing between a partially vaccinated and an unvaccinated population. The mixing ranged from random mixing to like-with-like mixing (“assortativity”). There were three possible “compartments” of people in the model: those considered susceptible to infection, those considered infected and infectious, and those considered immune because of recovery.
The model showed that, as mixing between the vaccinated and the unvaccinated populations increased, case numbers rose, “with cases in the unvaccinated subpopulation accounting for a substantial proportion of infections.” However, as mixing between the populations decreased, the final attack rate decreased among vaccinated people, but the relative “contribution of risk to vaccinated people caused by infection acquired from contact with unvaccinated people ... increased.”
When the vaccination rate was increased in the model, case numbers among the vaccinated declined “as expected, owing to indirect protective effects,” the researchers noted. But this also “further increased the relative contribution to risk in vaccinated people by those who were unvaccinated.”
Self-regarding risk?
The findings show that “choices made by people who forgo vaccination contribute disproportionately to risk among those who do get vaccinated,” the researchers wrote. “Although risk associated with avoiding vaccination during a virulent pandemic accrues chiefly to those who are unvaccinated, the choice of some individuals to refuse vaccination is likely to affect the health and safety of vaccinated people in a manner disproportionate to the fraction of unvaccinated people in the population.”
The fact that like-with-like mixing cannot mitigate the risk to vaccinated people “undermines the assertion that vaccine choice is best left to the individual and supports strong public actions aimed at enhancing vaccine uptake and limiting access to public spaces for unvaccinated people,” they wrote.
Mandates and passports
“Our model provides support for vaccine mandates and passports during epidemics, such that vaccination is required for people to take part in nonessential activities,” said Dr. Fisman. The choice to not be vaccinated against COVID-19 should not be considered “self-regarding,” he added. “Risk is self-regarding when it only impacts the person engaging in the activity. Something like smoking cigarettes (alone, without others around) creates a lot of risk over time, but if nobody is breathing your secondhand smoke, you’re only creating risk for yourself. By contrast, we regulate, in Ontario, your right to smoke in public indoor spaces such as restaurants, because once other people are around, the risk isn’t self-regarding anymore. You’re creating risk for others.”
The authors also noted that the risks created by the unvaccinated extend beyond those of infection by “creating a risk that those around them may not be able to obtain the care they need.” They recommended that considerations of equity and justice for people who do choose to be vaccinated, as well as those who choose not to be, need to be included in formulating vaccination policy.
Illuminating the discussion
Asked to comment on the study, Matthew Oughton, MD, assistant professor of medicine at McGill University, Montreal, said: “It is easy to dismiss a mathematical model as a series of assumptions that leads to an implausible conclusion. ... However, they can serve to illustrate and, to an extent, quantify the results of complex interactions, and this study does just that.” Dr. Oughton was not involved in the research.
During the past 2 years, the scientific press and the general press have often discussed the individual and collective effects of disease-prevention methods, including nonpharmaceutical interventions. “Models like this can help illuminate those discussions by highlighting important consequences of preventive measures,” said Dr. Oughton, who also works in the division of infectious diseases at the Jewish General Hospital, Montreal.
It’s worth noting that the authors modeled vaccine effectiveness against all infection, “rather than the generally greater and more durable effects we have seen for vaccines in prevention of severe infection,” said Dr. Oughton. He added that the authors did not include the effect of vaccination in reducing forward transmission. “Inclusion of this effect would presumably have reduced overall infectious burden in mixed populations and increased the difference between groups at lower levels of mixing between populations.”
The research was supported by a grant from the Canadian Institutes of Health Research. Dr. Fisman has served on advisory boards related to influenza and SARS-CoV-2 vaccines for Seqirus, Pfizer, AstraZeneca, and Sanofi-Pasteur Vaccines and has served as a legal expert on issues related to COVID-19 epidemiology for the Elementary Teachers Federation of Ontario and the Registered Nurses Association of Ontario. Dr. Oughton disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM THE CANADIAN MEDICAL ASSOCIATION JOURNAL
COVID fallout: ‘Alarming’ dip in routine vax for pregnant women
The percentage of low-income pregnant mothers who received influenza and Tdap vaccinations fell sharply during the COVID-19 pandemic, especially in Black and Hispanic patients, a new study finds.
The percentage of patients who received the influenza vaccines at two Medicaid clinics in Houston dropped from 78% before the pandemic to 61% during it (adjusted odds ratio, 0.38; 95% CI, 0.26-0.53; P < .01), researchers reported at the annual clinical and scientific meeting of the American College of Obstetricians and Gynecologists. The percentage receiving the Tdap vaccine dipped from 85% to 76% (aOR, 0.56; 95% CI, 0.40-0.79; P < .01).
New York–Presbyterian/Weill Cornell Medical Center pediatrician Sallie Permar, MD, PhD, who’s familiar with the study findings, called them “alarming” and said in an interview that they should be “a call to action for providers.”
“Continuing the status quo in our routine preventative health care and clinic operations means that we are losing ground in reduction and elimination of vaccine-preventable diseases,” Dr. Permar said in an interview.
According to corresponding author Bani Ratan, MD, an ob.gyn. with the Baylor College of Medicine, Houston, there’s been little if any previous research into routine, non-COVID vaccination in pregnant women during the pandemic.
For the study, researchers retrospectively analyzed the records of 939 pregnant women who entered prenatal care before 20 weeks (462 from May–November 2019, and 477 from May–November 2020) and delivered at full term.
Among ethnic groups, non-Hispanic Blacks saw the largest decline in influenza vaccines. Among them, the percentage who got them fell from 64% (73/114) to 35% (35/101; aOR, 0.30; 95% CI, 0.17-0.52; P < .01). Only Hispanics had a statistically significant decline in Tdap vaccination (OR, 0.52, 95% CI, 0.34-0.80; P < .01, percentages not provided).
Another study presented at ACOG examined vaccination rates during the pandemic and found that Tdap vaccination rates dipped among pregnant women in a Philadelphia-area health care system.
Possible causes for the decline in routine vaccination include hesitancy linked to the COVID-19 vaccines and fewer office visits because of telemedicine, said Dr. Batan in an interview.
Dr. Permar blamed the role of vaccine misinformation during the pandemic and the mistrust caused by the exclusion of pregnant women from early vaccine trials. She added that “challenges in health care staffing and issues of health care provider burnout that worsened during the pandemic likely contributed to a fraying of the focus on preventive health maintenance simply due to bandwidth of health professionals.”
In a separate study presented at ACOG, researchers at the State University of New York, Syracuse, reported on a survey of 157 pregnant women of whom just 38.2% were vaccinated against COVID-19. Among the unvaccinated, who were more likely to have less education, 66% reported that lack of data about vaccination was their primary concern.
No funding or disclosures are reported by study authors. Dr. Permar reported consulting for Merck, Moderna, GlaxoSmithKline, Pfizer, Dynavax, and Hookipa on cytomegalovirus vaccine programs.
*This story was updated on 5/11/2022.
The percentage of low-income pregnant mothers who received influenza and Tdap vaccinations fell sharply during the COVID-19 pandemic, especially in Black and Hispanic patients, a new study finds.
The percentage of patients who received the influenza vaccines at two Medicaid clinics in Houston dropped from 78% before the pandemic to 61% during it (adjusted odds ratio, 0.38; 95% CI, 0.26-0.53; P < .01), researchers reported at the annual clinical and scientific meeting of the American College of Obstetricians and Gynecologists. The percentage receiving the Tdap vaccine dipped from 85% to 76% (aOR, 0.56; 95% CI, 0.40-0.79; P < .01).
New York–Presbyterian/Weill Cornell Medical Center pediatrician Sallie Permar, MD, PhD, who’s familiar with the study findings, called them “alarming” and said in an interview that they should be “a call to action for providers.”
“Continuing the status quo in our routine preventative health care and clinic operations means that we are losing ground in reduction and elimination of vaccine-preventable diseases,” Dr. Permar said in an interview.
According to corresponding author Bani Ratan, MD, an ob.gyn. with the Baylor College of Medicine, Houston, there’s been little if any previous research into routine, non-COVID vaccination in pregnant women during the pandemic.
For the study, researchers retrospectively analyzed the records of 939 pregnant women who entered prenatal care before 20 weeks (462 from May–November 2019, and 477 from May–November 2020) and delivered at full term.
Among ethnic groups, non-Hispanic Blacks saw the largest decline in influenza vaccines. Among them, the percentage who got them fell from 64% (73/114) to 35% (35/101; aOR, 0.30; 95% CI, 0.17-0.52; P < .01). Only Hispanics had a statistically significant decline in Tdap vaccination (OR, 0.52, 95% CI, 0.34-0.80; P < .01, percentages not provided).
Another study presented at ACOG examined vaccination rates during the pandemic and found that Tdap vaccination rates dipped among pregnant women in a Philadelphia-area health care system.
Possible causes for the decline in routine vaccination include hesitancy linked to the COVID-19 vaccines and fewer office visits because of telemedicine, said Dr. Batan in an interview.
Dr. Permar blamed the role of vaccine misinformation during the pandemic and the mistrust caused by the exclusion of pregnant women from early vaccine trials. She added that “challenges in health care staffing and issues of health care provider burnout that worsened during the pandemic likely contributed to a fraying of the focus on preventive health maintenance simply due to bandwidth of health professionals.”
In a separate study presented at ACOG, researchers at the State University of New York, Syracuse, reported on a survey of 157 pregnant women of whom just 38.2% were vaccinated against COVID-19. Among the unvaccinated, who were more likely to have less education, 66% reported that lack of data about vaccination was their primary concern.
No funding or disclosures are reported by study authors. Dr. Permar reported consulting for Merck, Moderna, GlaxoSmithKline, Pfizer, Dynavax, and Hookipa on cytomegalovirus vaccine programs.
*This story was updated on 5/11/2022.
The percentage of low-income pregnant mothers who received influenza and Tdap vaccinations fell sharply during the COVID-19 pandemic, especially in Black and Hispanic patients, a new study finds.
The percentage of patients who received the influenza vaccines at two Medicaid clinics in Houston dropped from 78% before the pandemic to 61% during it (adjusted odds ratio, 0.38; 95% CI, 0.26-0.53; P < .01), researchers reported at the annual clinical and scientific meeting of the American College of Obstetricians and Gynecologists. The percentage receiving the Tdap vaccine dipped from 85% to 76% (aOR, 0.56; 95% CI, 0.40-0.79; P < .01).
New York–Presbyterian/Weill Cornell Medical Center pediatrician Sallie Permar, MD, PhD, who’s familiar with the study findings, called them “alarming” and said in an interview that they should be “a call to action for providers.”
“Continuing the status quo in our routine preventative health care and clinic operations means that we are losing ground in reduction and elimination of vaccine-preventable diseases,” Dr. Permar said in an interview.
According to corresponding author Bani Ratan, MD, an ob.gyn. with the Baylor College of Medicine, Houston, there’s been little if any previous research into routine, non-COVID vaccination in pregnant women during the pandemic.
For the study, researchers retrospectively analyzed the records of 939 pregnant women who entered prenatal care before 20 weeks (462 from May–November 2019, and 477 from May–November 2020) and delivered at full term.
Among ethnic groups, non-Hispanic Blacks saw the largest decline in influenza vaccines. Among them, the percentage who got them fell from 64% (73/114) to 35% (35/101; aOR, 0.30; 95% CI, 0.17-0.52; P < .01). Only Hispanics had a statistically significant decline in Tdap vaccination (OR, 0.52, 95% CI, 0.34-0.80; P < .01, percentages not provided).
Another study presented at ACOG examined vaccination rates during the pandemic and found that Tdap vaccination rates dipped among pregnant women in a Philadelphia-area health care system.
Possible causes for the decline in routine vaccination include hesitancy linked to the COVID-19 vaccines and fewer office visits because of telemedicine, said Dr. Batan in an interview.
Dr. Permar blamed the role of vaccine misinformation during the pandemic and the mistrust caused by the exclusion of pregnant women from early vaccine trials. She added that “challenges in health care staffing and issues of health care provider burnout that worsened during the pandemic likely contributed to a fraying of the focus on preventive health maintenance simply due to bandwidth of health professionals.”
In a separate study presented at ACOG, researchers at the State University of New York, Syracuse, reported on a survey of 157 pregnant women of whom just 38.2% were vaccinated against COVID-19. Among the unvaccinated, who were more likely to have less education, 66% reported that lack of data about vaccination was their primary concern.
No funding or disclosures are reported by study authors. Dr. Permar reported consulting for Merck, Moderna, GlaxoSmithKline, Pfizer, Dynavax, and Hookipa on cytomegalovirus vaccine programs.
*This story was updated on 5/11/2022.
FROM ACOG 2022
CDC predicts a rise in COVID-19 hospitalizations and deaths in coming weeks
, according to a national forecast used by the Centers for Disease Control and Prevention.
The national model also predicts that about 5,000 deaths will occur over the next two weeks, with Ohio, New Jersey, and New York projected to see the largest totals of daily deaths in upcoming weeks.
The numbers follow several weeks of steady increases in infections across the country. More than 67,000 new cases are being reported daily, according to the data tracker from The New York Times, marking a 59% increase in the past two weeks.
In the Northeast, infection rates have risen by nearly 65%. In the New York and New Jersey region, infection rates are up about 55% in the past two weeks.
Hospitalizations have already begun to climb as well, with about 19,000 COVID-19 patients hospitalized nationwide and 1,725 in intensive care, according to the latest data from the Department of Health and Human Services. In the last week, hospital admissions have jumped by 20%, and emergency department visits are up by 18%.
The CDC forecast shows that 42 states and territories will see increases in hospital admissions during the next two weeks. Florida, Minnesota, New York, and Wisconsin will see some of the largest increases.
On average, more than 2,200 COVID-19 patients are entering the hospital each day, which has increased about 20% in the last week, according to ABC News. This also marks the highest number of COVID-19 patients needing hospital care since mid-March.
Public health officials have cited several factors for the increase in cases, such as states lifting mask mandates and other safety restrictions, ABC News reported. Highly contagious Omicron subvariants, such as BA.2 and BA.2.12.1, continue to spread in the United States and escape immunity from previous infections.
The BA.2 subvariant accounts for 62% of new national cases, according to the latest CDC data. The BA.2.12.1 subvariant makes up about 36% of new cases across the United States but 62% in the New York area.
A version of this article first appeared on WebMD.com.
, according to a national forecast used by the Centers for Disease Control and Prevention.
The national model also predicts that about 5,000 deaths will occur over the next two weeks, with Ohio, New Jersey, and New York projected to see the largest totals of daily deaths in upcoming weeks.
The numbers follow several weeks of steady increases in infections across the country. More than 67,000 new cases are being reported daily, according to the data tracker from The New York Times, marking a 59% increase in the past two weeks.
In the Northeast, infection rates have risen by nearly 65%. In the New York and New Jersey region, infection rates are up about 55% in the past two weeks.
Hospitalizations have already begun to climb as well, with about 19,000 COVID-19 patients hospitalized nationwide and 1,725 in intensive care, according to the latest data from the Department of Health and Human Services. In the last week, hospital admissions have jumped by 20%, and emergency department visits are up by 18%.
The CDC forecast shows that 42 states and territories will see increases in hospital admissions during the next two weeks. Florida, Minnesota, New York, and Wisconsin will see some of the largest increases.
On average, more than 2,200 COVID-19 patients are entering the hospital each day, which has increased about 20% in the last week, according to ABC News. This also marks the highest number of COVID-19 patients needing hospital care since mid-March.
Public health officials have cited several factors for the increase in cases, such as states lifting mask mandates and other safety restrictions, ABC News reported. Highly contagious Omicron subvariants, such as BA.2 and BA.2.12.1, continue to spread in the United States and escape immunity from previous infections.
The BA.2 subvariant accounts for 62% of new national cases, according to the latest CDC data. The BA.2.12.1 subvariant makes up about 36% of new cases across the United States but 62% in the New York area.
A version of this article first appeared on WebMD.com.
, according to a national forecast used by the Centers for Disease Control and Prevention.
The national model also predicts that about 5,000 deaths will occur over the next two weeks, with Ohio, New Jersey, and New York projected to see the largest totals of daily deaths in upcoming weeks.
The numbers follow several weeks of steady increases in infections across the country. More than 67,000 new cases are being reported daily, according to the data tracker from The New York Times, marking a 59% increase in the past two weeks.
In the Northeast, infection rates have risen by nearly 65%. In the New York and New Jersey region, infection rates are up about 55% in the past two weeks.
Hospitalizations have already begun to climb as well, with about 19,000 COVID-19 patients hospitalized nationwide and 1,725 in intensive care, according to the latest data from the Department of Health and Human Services. In the last week, hospital admissions have jumped by 20%, and emergency department visits are up by 18%.
The CDC forecast shows that 42 states and territories will see increases in hospital admissions during the next two weeks. Florida, Minnesota, New York, and Wisconsin will see some of the largest increases.
On average, more than 2,200 COVID-19 patients are entering the hospital each day, which has increased about 20% in the last week, according to ABC News. This also marks the highest number of COVID-19 patients needing hospital care since mid-March.
Public health officials have cited several factors for the increase in cases, such as states lifting mask mandates and other safety restrictions, ABC News reported. Highly contagious Omicron subvariants, such as BA.2 and BA.2.12.1, continue to spread in the United States and escape immunity from previous infections.
The BA.2 subvariant accounts for 62% of new national cases, according to the latest CDC data. The BA.2.12.1 subvariant makes up about 36% of new cases across the United States but 62% in the New York area.
A version of this article first appeared on WebMD.com.
More selective antibiotic shows promise for C. diff. infection
An investigational, novel oral antibiotic with greater selectivity than vancomycin, metronidazole, and even fidaxomicin may offer improved protection of healthy gut bacteria during the treatment of Clostridium difficile infection (CDI), according to ongoing research.
“CDI treatment has historically been dominated by metronidazole and vancomycin,” said Katherine Johnson, DO, from the Western Infectious Disease Consultants, P.C., Denver. However, these broad-spectrum drugs negatively affect healthy bacteria in the gut and increase the risk of CDI recurrence.
This is also a problem for drugs in the CDI antibiotic pipeline: Many candidate drugs have failed because of their broad-spectrum activity, she added during a session at the Peggy Lillis Foundation 2022 National C. diff. Advocacy Summit.
“An ideal CDI therapy would be a very narrow-spectrum antibiotic that has a minimal effect on normal gut bacteria,” she said.
Dr. Johnson is currently working on a phase 2 clinical trial that is evaluating the novel antibiotic, dubbed CRS3123, for the treatment of primary CDI and first-recurrence CDI. The investigational agent targets and inhibits a form of the methionyl tRNA synthetase enzyme, which is strictly required for protein biosynthesis in C. diff. and is therefore an ideal target for treatment of primary and recurrent CDI.
In her session, Dr. Johnson reported that CRS3123 inhibits the damaging toxins produced by C. diff., potentially resulting in more rapid symptom resolution. Additionally, owing to its novel mode of action, no strains are currently resistant to CRS3123.
She presented findings from an animal study that showed that CRS3123 was superior to vancomycin in terms of prolonging survival. She also presented findings from phase 1 clinical trials that showed that most adverse events (AEs) associated with CRS3123 were mild. No serious AEs were reported.
A ‘huge infectious burden’
If successful in further research, CRS3123 could offer significant value to patients with C. diff., especially those with recurrent infection, given the sometimes extreme clinical, quality-of-life, and economic burdens associated with CDI.
“CDI is a huge infectious burden to the U.S. health care system and globally has been listed by the Centers for Disease Control and Prevention as an urgent threat,” Byron Vaughn, MD, from the University of Minnesota, told this news organization.
“Despite a number of antibiotic stewardship and infection control and prevention efforts, we haven’t seen much of a change in the incidence of CDI,” he said. He said that the risk of recurrence can be as high as 30%.
While oral vancomycin is effective for treating C. diff., Dr. Vaughn noted that the antibiotic lacks selectivity and destroys healthy gut bacteria, resulting in substantial dysbiosis. “Dysbiosis is really the key to getting recurrent C. diff.,” he explained, “because if you have healthy gut bacteria, you will inherently resist CDI.”
Dr. Vaughn stated that his center is in the startup phase for being a site for a clinical trial of CRS3123. The hope is that CRS3123, because its spectrum is narrower than that of fidaxomicin and vancomycin, doesn’t induce intestinal dysbiosis. “It really just treats the C. diff. and leaves every other bug alone so that your gut bacteria can recover while the C. diff. is being treated,” he said. “And then when you stop CRS3123, you have healthy gut bacteria already present to prevent recurrence.”
If this is confirmed in large-scale trials, there could be a “dramatic decrease” in the rates of recurrent C. diff., said Dr. Vaughn.
Aside from the potential clinical impact, the economic implications of a novel selective antibiotic that preserves healthy gut bacteria could be significant, he added. “Depending on exactly what population you’re looking at, probably about a third of the cost of C. diff. is actually attributable to recurrence. That’s a huge economic burden that could be improved.”
Dr. Johnson is an employee of Crestone, which is developing CRS3123. Dr. Vaughn reports no relevant financial relationships.
A version of this article first appeared on Medscape.com.
An investigational, novel oral antibiotic with greater selectivity than vancomycin, metronidazole, and even fidaxomicin may offer improved protection of healthy gut bacteria during the treatment of Clostridium difficile infection (CDI), according to ongoing research.
“CDI treatment has historically been dominated by metronidazole and vancomycin,” said Katherine Johnson, DO, from the Western Infectious Disease Consultants, P.C., Denver. However, these broad-spectrum drugs negatively affect healthy bacteria in the gut and increase the risk of CDI recurrence.
This is also a problem for drugs in the CDI antibiotic pipeline: Many candidate drugs have failed because of their broad-spectrum activity, she added during a session at the Peggy Lillis Foundation 2022 National C. diff. Advocacy Summit.
“An ideal CDI therapy would be a very narrow-spectrum antibiotic that has a minimal effect on normal gut bacteria,” she said.
Dr. Johnson is currently working on a phase 2 clinical trial that is evaluating the novel antibiotic, dubbed CRS3123, for the treatment of primary CDI and first-recurrence CDI. The investigational agent targets and inhibits a form of the methionyl tRNA synthetase enzyme, which is strictly required for protein biosynthesis in C. diff. and is therefore an ideal target for treatment of primary and recurrent CDI.
In her session, Dr. Johnson reported that CRS3123 inhibits the damaging toxins produced by C. diff., potentially resulting in more rapid symptom resolution. Additionally, owing to its novel mode of action, no strains are currently resistant to CRS3123.
She presented findings from an animal study that showed that CRS3123 was superior to vancomycin in terms of prolonging survival. She also presented findings from phase 1 clinical trials that showed that most adverse events (AEs) associated with CRS3123 were mild. No serious AEs were reported.
A ‘huge infectious burden’
If successful in further research, CRS3123 could offer significant value to patients with C. diff., especially those with recurrent infection, given the sometimes extreme clinical, quality-of-life, and economic burdens associated with CDI.
“CDI is a huge infectious burden to the U.S. health care system and globally has been listed by the Centers for Disease Control and Prevention as an urgent threat,” Byron Vaughn, MD, from the University of Minnesota, told this news organization.
“Despite a number of antibiotic stewardship and infection control and prevention efforts, we haven’t seen much of a change in the incidence of CDI,” he said. He said that the risk of recurrence can be as high as 30%.
While oral vancomycin is effective for treating C. diff., Dr. Vaughn noted that the antibiotic lacks selectivity and destroys healthy gut bacteria, resulting in substantial dysbiosis. “Dysbiosis is really the key to getting recurrent C. diff.,” he explained, “because if you have healthy gut bacteria, you will inherently resist CDI.”
Dr. Vaughn stated that his center is in the startup phase for being a site for a clinical trial of CRS3123. The hope is that CRS3123, because its spectrum is narrower than that of fidaxomicin and vancomycin, doesn’t induce intestinal dysbiosis. “It really just treats the C. diff. and leaves every other bug alone so that your gut bacteria can recover while the C. diff. is being treated,” he said. “And then when you stop CRS3123, you have healthy gut bacteria already present to prevent recurrence.”
If this is confirmed in large-scale trials, there could be a “dramatic decrease” in the rates of recurrent C. diff., said Dr. Vaughn.
Aside from the potential clinical impact, the economic implications of a novel selective antibiotic that preserves healthy gut bacteria could be significant, he added. “Depending on exactly what population you’re looking at, probably about a third of the cost of C. diff. is actually attributable to recurrence. That’s a huge economic burden that could be improved.”
Dr. Johnson is an employee of Crestone, which is developing CRS3123. Dr. Vaughn reports no relevant financial relationships.
A version of this article first appeared on Medscape.com.
An investigational, novel oral antibiotic with greater selectivity than vancomycin, metronidazole, and even fidaxomicin may offer improved protection of healthy gut bacteria during the treatment of Clostridium difficile infection (CDI), according to ongoing research.
“CDI treatment has historically been dominated by metronidazole and vancomycin,” said Katherine Johnson, DO, from the Western Infectious Disease Consultants, P.C., Denver. However, these broad-spectrum drugs negatively affect healthy bacteria in the gut and increase the risk of CDI recurrence.
This is also a problem for drugs in the CDI antibiotic pipeline: Many candidate drugs have failed because of their broad-spectrum activity, she added during a session at the Peggy Lillis Foundation 2022 National C. diff. Advocacy Summit.
“An ideal CDI therapy would be a very narrow-spectrum antibiotic that has a minimal effect on normal gut bacteria,” she said.
Dr. Johnson is currently working on a phase 2 clinical trial that is evaluating the novel antibiotic, dubbed CRS3123, for the treatment of primary CDI and first-recurrence CDI. The investigational agent targets and inhibits a form of the methionyl tRNA synthetase enzyme, which is strictly required for protein biosynthesis in C. diff. and is therefore an ideal target for treatment of primary and recurrent CDI.
In her session, Dr. Johnson reported that CRS3123 inhibits the damaging toxins produced by C. diff., potentially resulting in more rapid symptom resolution. Additionally, owing to its novel mode of action, no strains are currently resistant to CRS3123.
She presented findings from an animal study that showed that CRS3123 was superior to vancomycin in terms of prolonging survival. She also presented findings from phase 1 clinical trials that showed that most adverse events (AEs) associated with CRS3123 were mild. No serious AEs were reported.
A ‘huge infectious burden’
If successful in further research, CRS3123 could offer significant value to patients with C. diff., especially those with recurrent infection, given the sometimes extreme clinical, quality-of-life, and economic burdens associated with CDI.
“CDI is a huge infectious burden to the U.S. health care system and globally has been listed by the Centers for Disease Control and Prevention as an urgent threat,” Byron Vaughn, MD, from the University of Minnesota, told this news organization.
“Despite a number of antibiotic stewardship and infection control and prevention efforts, we haven’t seen much of a change in the incidence of CDI,” he said. He said that the risk of recurrence can be as high as 30%.
While oral vancomycin is effective for treating C. diff., Dr. Vaughn noted that the antibiotic lacks selectivity and destroys healthy gut bacteria, resulting in substantial dysbiosis. “Dysbiosis is really the key to getting recurrent C. diff.,” he explained, “because if you have healthy gut bacteria, you will inherently resist CDI.”
Dr. Vaughn stated that his center is in the startup phase for being a site for a clinical trial of CRS3123. The hope is that CRS3123, because its spectrum is narrower than that of fidaxomicin and vancomycin, doesn’t induce intestinal dysbiosis. “It really just treats the C. diff. and leaves every other bug alone so that your gut bacteria can recover while the C. diff. is being treated,” he said. “And then when you stop CRS3123, you have healthy gut bacteria already present to prevent recurrence.”
If this is confirmed in large-scale trials, there could be a “dramatic decrease” in the rates of recurrent C. diff., said Dr. Vaughn.
Aside from the potential clinical impact, the economic implications of a novel selective antibiotic that preserves healthy gut bacteria could be significant, he added. “Depending on exactly what population you’re looking at, probably about a third of the cost of C. diff. is actually attributable to recurrence. That’s a huge economic burden that could be improved.”
Dr. Johnson is an employee of Crestone, which is developing CRS3123. Dr. Vaughn reports no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The importance of toxin testing in C. difficile infection: Understanding the results
Clostridioides difficile infection is often confirmed through toxin testing, yet toxin tests alone may not be sufficient for diagnosing and guiding treatment decisions for patients with CDI.
“The presence of a toxigenic strain does not always equal disease,” said David Lyerly, PhD, during a session on C. difficile toxin testing at the Peggy Lillis Foundation 2022 National C. diff Advocacy Summit.
Dr. Lyerly, the chief science officer at Techlab, explained that exotoxins A and B are produced by specific strains of C. difficile and are involved in infections, but some patients who test positive for these toxins by polymerase chain reaction or other tests do not have CDI or they are not appropriate candidates for CDI treatment.
Several studies conducted during the past decade, however, support the importance of toxin detection. Some research has suggested that toxin-positive patients tend to have more clinically severe disease than those who test negative, he noted.
Although its use is limited when it is used alone, toxin testing is needed to confirm a CDI diagnosis and to ensure antibiotic stewardship, Dr. Lyerly said.
He suggested that, in addition to toxin testing, there is a need for molecular measures and other improved diagnostics to identify candidates most likely to benefit from CDI treatment.
“Because we generally detect toxin genes instead of toxin proteins, you can identify persons colonized with toxigenic C. difficile who do not actually have CDI,” Kevin W. Garey, PharmD, from the University of Houston, said in an interview.
Dr. Garey added that a person could likewise have low levels of toxins that aren’t detected by toxin tests but could still have CDI.
“Given this, better diagnostics that incorporate active toxin production and your body’s response to those toxins are needed,” he said, especially since C. difficile toxins are responsible for disease sequelae, including gastroenteritis, colonic perforation, sepsis, and death.
Toxin testing a ‘controversial area’
“C. difficile toxin testing has been a controversial area for almost a decade or more,” Shruti K. Gohil, MD, from University of California, Irvine, Health Epidemiology and Infection Prevention, said in an interview.
Dr. Gohil noted that toxin testing is a better test for clinical C. difficile colitis but by itself can miss C. difficile. “So, we are in this conundrum nationally,” she said.
“Many facilities will use a double- or triple-test strategy to make sure that you have a true C. difficile case mandating the use of antibiotics,” she explained. “The reason we test specifically with the enzyme immunoassay or toxin test is to know whether or not you have real C. difficile that’s actively producing the toxin for colitis.”
A patient with C. difficile who has been treated and is in recovery may still test positive on a C. difficile toxin test, added Dr. Gohil. “It would be great if we had a test that could really judge an active, clinical C. difficile infection. This [test] would help in identifying the right patients who need treatment and would also be able to tell if a patient has been cleared of C. difficile.”
Dr. Lyerly is an employee of Techlab. Dr. Garey and Dr. Gohil reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Clostridioides difficile infection is often confirmed through toxin testing, yet toxin tests alone may not be sufficient for diagnosing and guiding treatment decisions for patients with CDI.
“The presence of a toxigenic strain does not always equal disease,” said David Lyerly, PhD, during a session on C. difficile toxin testing at the Peggy Lillis Foundation 2022 National C. diff Advocacy Summit.
Dr. Lyerly, the chief science officer at Techlab, explained that exotoxins A and B are produced by specific strains of C. difficile and are involved in infections, but some patients who test positive for these toxins by polymerase chain reaction or other tests do not have CDI or they are not appropriate candidates for CDI treatment.
Several studies conducted during the past decade, however, support the importance of toxin detection. Some research has suggested that toxin-positive patients tend to have more clinically severe disease than those who test negative, he noted.
Although its use is limited when it is used alone, toxin testing is needed to confirm a CDI diagnosis and to ensure antibiotic stewardship, Dr. Lyerly said.
He suggested that, in addition to toxin testing, there is a need for molecular measures and other improved diagnostics to identify candidates most likely to benefit from CDI treatment.
“Because we generally detect toxin genes instead of toxin proteins, you can identify persons colonized with toxigenic C. difficile who do not actually have CDI,” Kevin W. Garey, PharmD, from the University of Houston, said in an interview.
Dr. Garey added that a person could likewise have low levels of toxins that aren’t detected by toxin tests but could still have CDI.
“Given this, better diagnostics that incorporate active toxin production and your body’s response to those toxins are needed,” he said, especially since C. difficile toxins are responsible for disease sequelae, including gastroenteritis, colonic perforation, sepsis, and death.
Toxin testing a ‘controversial area’
“C. difficile toxin testing has been a controversial area for almost a decade or more,” Shruti K. Gohil, MD, from University of California, Irvine, Health Epidemiology and Infection Prevention, said in an interview.
Dr. Gohil noted that toxin testing is a better test for clinical C. difficile colitis but by itself can miss C. difficile. “So, we are in this conundrum nationally,” she said.
“Many facilities will use a double- or triple-test strategy to make sure that you have a true C. difficile case mandating the use of antibiotics,” she explained. “The reason we test specifically with the enzyme immunoassay or toxin test is to know whether or not you have real C. difficile that’s actively producing the toxin for colitis.”
A patient with C. difficile who has been treated and is in recovery may still test positive on a C. difficile toxin test, added Dr. Gohil. “It would be great if we had a test that could really judge an active, clinical C. difficile infection. This [test] would help in identifying the right patients who need treatment and would also be able to tell if a patient has been cleared of C. difficile.”
Dr. Lyerly is an employee of Techlab. Dr. Garey and Dr. Gohil reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Clostridioides difficile infection is often confirmed through toxin testing, yet toxin tests alone may not be sufficient for diagnosing and guiding treatment decisions for patients with CDI.
“The presence of a toxigenic strain does not always equal disease,” said David Lyerly, PhD, during a session on C. difficile toxin testing at the Peggy Lillis Foundation 2022 National C. diff Advocacy Summit.
Dr. Lyerly, the chief science officer at Techlab, explained that exotoxins A and B are produced by specific strains of C. difficile and are involved in infections, but some patients who test positive for these toxins by polymerase chain reaction or other tests do not have CDI or they are not appropriate candidates for CDI treatment.
Several studies conducted during the past decade, however, support the importance of toxin detection. Some research has suggested that toxin-positive patients tend to have more clinically severe disease than those who test negative, he noted.
Although its use is limited when it is used alone, toxin testing is needed to confirm a CDI diagnosis and to ensure antibiotic stewardship, Dr. Lyerly said.
He suggested that, in addition to toxin testing, there is a need for molecular measures and other improved diagnostics to identify candidates most likely to benefit from CDI treatment.
“Because we generally detect toxin genes instead of toxin proteins, you can identify persons colonized with toxigenic C. difficile who do not actually have CDI,” Kevin W. Garey, PharmD, from the University of Houston, said in an interview.
Dr. Garey added that a person could likewise have low levels of toxins that aren’t detected by toxin tests but could still have CDI.
“Given this, better diagnostics that incorporate active toxin production and your body’s response to those toxins are needed,” he said, especially since C. difficile toxins are responsible for disease sequelae, including gastroenteritis, colonic perforation, sepsis, and death.
Toxin testing a ‘controversial area’
“C. difficile toxin testing has been a controversial area for almost a decade or more,” Shruti K. Gohil, MD, from University of California, Irvine, Health Epidemiology and Infection Prevention, said in an interview.
Dr. Gohil noted that toxin testing is a better test for clinical C. difficile colitis but by itself can miss C. difficile. “So, we are in this conundrum nationally,” she said.
“Many facilities will use a double- or triple-test strategy to make sure that you have a true C. difficile case mandating the use of antibiotics,” she explained. “The reason we test specifically with the enzyme immunoassay or toxin test is to know whether or not you have real C. difficile that’s actively producing the toxin for colitis.”
A patient with C. difficile who has been treated and is in recovery may still test positive on a C. difficile toxin test, added Dr. Gohil. “It would be great if we had a test that could really judge an active, clinical C. difficile infection. This [test] would help in identifying the right patients who need treatment and would also be able to tell if a patient has been cleared of C. difficile.”
Dr. Lyerly is an employee of Techlab. Dr. Garey and Dr. Gohil reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Alarming global rise in pediatric hepatitis: Expert Q&A
This spring, global health advisories have been issued regarding an alarming – and as-yet unexplained – uptick of hepatitis in children. Currently, over 200 cases have been reported worldwide, a relatively small amount that nonetheless belies a considerable toll, including several deaths and the need for liver transplantation in a number of patients. The long-term implications are not yet known. Global health officials are working hard to determine a cause, with many focusing on the underlying cases of adenovirus that several patients have presented with.
To understand more, this news organization reached out to frequent contributor William F. Balistreri, MD, a specialist in pediatric gastroenterology and hepatology at Cincinnati Children’s Hospital Medical Center, where to date they have treated at least six cases of hepatitis in otherwise healthy young children, with one requiring a liver transplant. Dr. Balistreri discussed how the outbreak has developed to date, his advice to hepatologists and pediatricians, and where we stand now in this fast-evolving crisis.
Tracing the outbreak in the United States
How has this outbreak played out thus far in the United States, and what have we learned from that?
Sporadic reports of cases in multiple states are appearing. On April 21, 2022, a health alert was issued by the Centers for Disease Control and Prevention, recommending testing for adenovirus in children with acute hepatitis of an unknown etiology.
Baker and colleagues recently described five children with severe hepatitis and adenovirus viremia who were admitted to a children’s hospital in Birmingham, Ala., between October and November 2021. In collaboration with local and state officials, the CDC reviewed clinical records in order to identify patients with hepatitis and concomitant adenovirus infection, confirmed by polymerase chain reaction (PCR).
By February 2022, a total of nine children were identified. There was no epidemiologic linkage among these nine patients; all were well and immunocompetent. The prodromal features were somewhat similar: upper respiratory infection, vomiting, diarrhea, and jaundice. All children had markedly elevated aminotransferase levels and variably elevated total bilirubin levels. Extensive workup for other causes of acute liver injury (for example, other viruses, toxins/drugs, metabolic and autoimmune diseases) was unrevealing.
Specifically, none had documented SARS-CoV-2 infection. However, in all nine children, adenovirus was detected in whole blood samples. In the six children who underwent liver biopsy, there was nonspecific hepatitis, without inclusions or immunohistochemical detection of viral agents, including adenovirus. In three patients, the liver injury progressed, and despite the administration of antiviral agents, two underwent liver transplantation.
Baker and colleagues also suggested that measurement of adenovirus titers in whole blood (rather than plasma) may be more sensitive.
The CDC has recommended monitoring and surveillance in order to more fully understand the nature of the illness.
European and global cases
What has been the experience with this in Europe and elsewhere globally?
In mid-to-late 2021, several cases of acute hepatitis of unknown nature in children were identified in Europe. Public health officials in the United Kingdom investigated the high number of cases seen in children from England, Scotland, and Wales. They noted approximately 60 cases in England, mostly in children aged 2-5 years.
Marsh and colleagues reported a cluster of cases of severe hepatitis of unknown origin in Scotland affecting children aged 3-5 years. In Scotland, admitted cases were routinely tested for SARS-CoV-2. Of the 13 cases, five had a recent positive test. They discussed the possibility of increased severity of disease following infection with Omicron BA.2 (the dominant SARS-CoV-2 virus circulating in Scotland at that time) or infection by an uncharacterized SARS-CoV-2 variant. None of the children had been vaccinated for SARS-CoV-2.
On April 15, 2022, the World Health Organization Disease Outbreak News published a report of acute hepatitis of unknown etiology occurring in Great Britain and Northern Ireland. By April 21, 2022, 169 cases of acute hepatitis of unknown origin in children younger than 16 years had been reported from 11 countries in the WHO European region and 1 country in the WHO region of the Americas. Approximately 10% required a liver transplantation and at least one death was reported.
What has been established about the possible connection to the SARS-CoV-2 virus, particularly as it relates to coinfection with adenovirus?
In that WHO report of 169 cases, adenovirus was detected in 74 and SARS-CoV-2 in 20. Of note, 19 cases had a SARS-CoV-2 and adenovirus coinfection.
The report’s authors emphasized that, “while adenovirus is a possible hypothesis, investigations are ongoing for the causative agent.” The authors questioned whether this represents a continuing increase in cases of hepatitis or reflects an increased awareness.
The stated priority of the WHO is to determine the cause and to further refine control and prevention actions.
Given the worldwide nature of this outbreak, have connections between any of the cases been made yet?
Not to my knowledge.
What clinicians need to know
What makes this outbreak of hepatitis cases particularly concerning to the health care community, in comparison to other childhood diseases that occur globally? Is it because the cause is unknown or is it for other reasons?
It may be a collective heightened concern following the emergence of COVID.
Whether it represents a new form of acute hepatitis, a continuing increase in cases of hepatitis, or an increased awareness because of the well-publicized alerts remains to be determined. We certainly saw “viral-induced hepatitis” in the past.
Young patients may first be brought to pediatricians. What, if anything, should pediatricians be on the lookout for? Do they need a heightened index of suspicion or are the cases too rare at this point?
An awareness of the “outbreak” may allow the clinician to extend the typical workup of a child presenting with an undefined, presumably viral illness.
In the cases reported, the prodromal and/or presenting symptoms were respiratory and gastrointestinal in nature. They include nausea, vomiting, diarrhea, and abdominal pain.
Specifically, if jaundice and/or scleral icterus is noted, then hepatitis should be suspected.
Should pediatricians consider early referral to a pediatric gastroenterologist or hepatologist?
Yes, because there is the potential for finding a treatable cause (for example, autoimmune hepatitis or a specific metabolic disease) in a patient presenting in this fashion.
In addition, the potential for progression to acute liver failure (with coagulopathy and encephalopathy), albeit rare, exists.
What do hepatologists need to be doing when presented with suspected cases?
The typical clinical picture holds and the workup is standard. The one new key, given the recent data, is to test for adenovirus, using whole blood versus plasma, as the former may be more sensitive.
In addition, it is prudent to check for SARS-CoV-2 by PCR.
What are the major questions that remain and that you’d like to see elucidated going forward?
There are many. Is this a new disease? A new variant of adenovirus? A synergy or susceptibility related to SARS-CoV-2? Is it related to a variant of SARS-CoV-2? Is it triggering an adverse immune response? Are there other epigenetic factors involved? And finally, is this an increase, or is it related to a collective heightened concern following the pandemic?
Dr. Balistreri is the Dorothy M.M. Kersten Professor of Pediatrics, director emeritus of the Pediatric Liver Care Center, medical director emeritus of liver transplantation, and professor at the University of Cincinnati; he is also with the department of pediatrics at Cincinnati Children’s Hospital Medical Center.
A version of this article first appeared on Medscape.com.
This spring, global health advisories have been issued regarding an alarming – and as-yet unexplained – uptick of hepatitis in children. Currently, over 200 cases have been reported worldwide, a relatively small amount that nonetheless belies a considerable toll, including several deaths and the need for liver transplantation in a number of patients. The long-term implications are not yet known. Global health officials are working hard to determine a cause, with many focusing on the underlying cases of adenovirus that several patients have presented with.
To understand more, this news organization reached out to frequent contributor William F. Balistreri, MD, a specialist in pediatric gastroenterology and hepatology at Cincinnati Children’s Hospital Medical Center, where to date they have treated at least six cases of hepatitis in otherwise healthy young children, with one requiring a liver transplant. Dr. Balistreri discussed how the outbreak has developed to date, his advice to hepatologists and pediatricians, and where we stand now in this fast-evolving crisis.
Tracing the outbreak in the United States
How has this outbreak played out thus far in the United States, and what have we learned from that?
Sporadic reports of cases in multiple states are appearing. On April 21, 2022, a health alert was issued by the Centers for Disease Control and Prevention, recommending testing for adenovirus in children with acute hepatitis of an unknown etiology.
Baker and colleagues recently described five children with severe hepatitis and adenovirus viremia who were admitted to a children’s hospital in Birmingham, Ala., between October and November 2021. In collaboration with local and state officials, the CDC reviewed clinical records in order to identify patients with hepatitis and concomitant adenovirus infection, confirmed by polymerase chain reaction (PCR).
By February 2022, a total of nine children were identified. There was no epidemiologic linkage among these nine patients; all were well and immunocompetent. The prodromal features were somewhat similar: upper respiratory infection, vomiting, diarrhea, and jaundice. All children had markedly elevated aminotransferase levels and variably elevated total bilirubin levels. Extensive workup for other causes of acute liver injury (for example, other viruses, toxins/drugs, metabolic and autoimmune diseases) was unrevealing.
Specifically, none had documented SARS-CoV-2 infection. However, in all nine children, adenovirus was detected in whole blood samples. In the six children who underwent liver biopsy, there was nonspecific hepatitis, without inclusions or immunohistochemical detection of viral agents, including adenovirus. In three patients, the liver injury progressed, and despite the administration of antiviral agents, two underwent liver transplantation.
Baker and colleagues also suggested that measurement of adenovirus titers in whole blood (rather than plasma) may be more sensitive.
The CDC has recommended monitoring and surveillance in order to more fully understand the nature of the illness.
European and global cases
What has been the experience with this in Europe and elsewhere globally?
In mid-to-late 2021, several cases of acute hepatitis of unknown nature in children were identified in Europe. Public health officials in the United Kingdom investigated the high number of cases seen in children from England, Scotland, and Wales. They noted approximately 60 cases in England, mostly in children aged 2-5 years.
Marsh and colleagues reported a cluster of cases of severe hepatitis of unknown origin in Scotland affecting children aged 3-5 years. In Scotland, admitted cases were routinely tested for SARS-CoV-2. Of the 13 cases, five had a recent positive test. They discussed the possibility of increased severity of disease following infection with Omicron BA.2 (the dominant SARS-CoV-2 virus circulating in Scotland at that time) or infection by an uncharacterized SARS-CoV-2 variant. None of the children had been vaccinated for SARS-CoV-2.
On April 15, 2022, the World Health Organization Disease Outbreak News published a report of acute hepatitis of unknown etiology occurring in Great Britain and Northern Ireland. By April 21, 2022, 169 cases of acute hepatitis of unknown origin in children younger than 16 years had been reported from 11 countries in the WHO European region and 1 country in the WHO region of the Americas. Approximately 10% required a liver transplantation and at least one death was reported.
What has been established about the possible connection to the SARS-CoV-2 virus, particularly as it relates to coinfection with adenovirus?
In that WHO report of 169 cases, adenovirus was detected in 74 and SARS-CoV-2 in 20. Of note, 19 cases had a SARS-CoV-2 and adenovirus coinfection.
The report’s authors emphasized that, “while adenovirus is a possible hypothesis, investigations are ongoing for the causative agent.” The authors questioned whether this represents a continuing increase in cases of hepatitis or reflects an increased awareness.
The stated priority of the WHO is to determine the cause and to further refine control and prevention actions.
Given the worldwide nature of this outbreak, have connections between any of the cases been made yet?
Not to my knowledge.
What clinicians need to know
What makes this outbreak of hepatitis cases particularly concerning to the health care community, in comparison to other childhood diseases that occur globally? Is it because the cause is unknown or is it for other reasons?
It may be a collective heightened concern following the emergence of COVID.
Whether it represents a new form of acute hepatitis, a continuing increase in cases of hepatitis, or an increased awareness because of the well-publicized alerts remains to be determined. We certainly saw “viral-induced hepatitis” in the past.
Young patients may first be brought to pediatricians. What, if anything, should pediatricians be on the lookout for? Do they need a heightened index of suspicion or are the cases too rare at this point?
An awareness of the “outbreak” may allow the clinician to extend the typical workup of a child presenting with an undefined, presumably viral illness.
In the cases reported, the prodromal and/or presenting symptoms were respiratory and gastrointestinal in nature. They include nausea, vomiting, diarrhea, and abdominal pain.
Specifically, if jaundice and/or scleral icterus is noted, then hepatitis should be suspected.
Should pediatricians consider early referral to a pediatric gastroenterologist or hepatologist?
Yes, because there is the potential for finding a treatable cause (for example, autoimmune hepatitis or a specific metabolic disease) in a patient presenting in this fashion.
In addition, the potential for progression to acute liver failure (with coagulopathy and encephalopathy), albeit rare, exists.
What do hepatologists need to be doing when presented with suspected cases?
The typical clinical picture holds and the workup is standard. The one new key, given the recent data, is to test for adenovirus, using whole blood versus plasma, as the former may be more sensitive.
In addition, it is prudent to check for SARS-CoV-2 by PCR.
What are the major questions that remain and that you’d like to see elucidated going forward?
There are many. Is this a new disease? A new variant of adenovirus? A synergy or susceptibility related to SARS-CoV-2? Is it related to a variant of SARS-CoV-2? Is it triggering an adverse immune response? Are there other epigenetic factors involved? And finally, is this an increase, or is it related to a collective heightened concern following the pandemic?
Dr. Balistreri is the Dorothy M.M. Kersten Professor of Pediatrics, director emeritus of the Pediatric Liver Care Center, medical director emeritus of liver transplantation, and professor at the University of Cincinnati; he is also with the department of pediatrics at Cincinnati Children’s Hospital Medical Center.
A version of this article first appeared on Medscape.com.
This spring, global health advisories have been issued regarding an alarming – and as-yet unexplained – uptick of hepatitis in children. Currently, over 200 cases have been reported worldwide, a relatively small amount that nonetheless belies a considerable toll, including several deaths and the need for liver transplantation in a number of patients. The long-term implications are not yet known. Global health officials are working hard to determine a cause, with many focusing on the underlying cases of adenovirus that several patients have presented with.
To understand more, this news organization reached out to frequent contributor William F. Balistreri, MD, a specialist in pediatric gastroenterology and hepatology at Cincinnati Children’s Hospital Medical Center, where to date they have treated at least six cases of hepatitis in otherwise healthy young children, with one requiring a liver transplant. Dr. Balistreri discussed how the outbreak has developed to date, his advice to hepatologists and pediatricians, and where we stand now in this fast-evolving crisis.
Tracing the outbreak in the United States
How has this outbreak played out thus far in the United States, and what have we learned from that?
Sporadic reports of cases in multiple states are appearing. On April 21, 2022, a health alert was issued by the Centers for Disease Control and Prevention, recommending testing for adenovirus in children with acute hepatitis of an unknown etiology.
Baker and colleagues recently described five children with severe hepatitis and adenovirus viremia who were admitted to a children’s hospital in Birmingham, Ala., between October and November 2021. In collaboration with local and state officials, the CDC reviewed clinical records in order to identify patients with hepatitis and concomitant adenovirus infection, confirmed by polymerase chain reaction (PCR).
By February 2022, a total of nine children were identified. There was no epidemiologic linkage among these nine patients; all were well and immunocompetent. The prodromal features were somewhat similar: upper respiratory infection, vomiting, diarrhea, and jaundice. All children had markedly elevated aminotransferase levels and variably elevated total bilirubin levels. Extensive workup for other causes of acute liver injury (for example, other viruses, toxins/drugs, metabolic and autoimmune diseases) was unrevealing.
Specifically, none had documented SARS-CoV-2 infection. However, in all nine children, adenovirus was detected in whole blood samples. In the six children who underwent liver biopsy, there was nonspecific hepatitis, without inclusions or immunohistochemical detection of viral agents, including adenovirus. In three patients, the liver injury progressed, and despite the administration of antiviral agents, two underwent liver transplantation.
Baker and colleagues also suggested that measurement of adenovirus titers in whole blood (rather than plasma) may be more sensitive.
The CDC has recommended monitoring and surveillance in order to more fully understand the nature of the illness.
European and global cases
What has been the experience with this in Europe and elsewhere globally?
In mid-to-late 2021, several cases of acute hepatitis of unknown nature in children were identified in Europe. Public health officials in the United Kingdom investigated the high number of cases seen in children from England, Scotland, and Wales. They noted approximately 60 cases in England, mostly in children aged 2-5 years.
Marsh and colleagues reported a cluster of cases of severe hepatitis of unknown origin in Scotland affecting children aged 3-5 years. In Scotland, admitted cases were routinely tested for SARS-CoV-2. Of the 13 cases, five had a recent positive test. They discussed the possibility of increased severity of disease following infection with Omicron BA.2 (the dominant SARS-CoV-2 virus circulating in Scotland at that time) or infection by an uncharacterized SARS-CoV-2 variant. None of the children had been vaccinated for SARS-CoV-2.
On April 15, 2022, the World Health Organization Disease Outbreak News published a report of acute hepatitis of unknown etiology occurring in Great Britain and Northern Ireland. By April 21, 2022, 169 cases of acute hepatitis of unknown origin in children younger than 16 years had been reported from 11 countries in the WHO European region and 1 country in the WHO region of the Americas. Approximately 10% required a liver transplantation and at least one death was reported.
What has been established about the possible connection to the SARS-CoV-2 virus, particularly as it relates to coinfection with adenovirus?
In that WHO report of 169 cases, adenovirus was detected in 74 and SARS-CoV-2 in 20. Of note, 19 cases had a SARS-CoV-2 and adenovirus coinfection.
The report’s authors emphasized that, “while adenovirus is a possible hypothesis, investigations are ongoing for the causative agent.” The authors questioned whether this represents a continuing increase in cases of hepatitis or reflects an increased awareness.
The stated priority of the WHO is to determine the cause and to further refine control and prevention actions.
Given the worldwide nature of this outbreak, have connections between any of the cases been made yet?
Not to my knowledge.
What clinicians need to know
What makes this outbreak of hepatitis cases particularly concerning to the health care community, in comparison to other childhood diseases that occur globally? Is it because the cause is unknown or is it for other reasons?
It may be a collective heightened concern following the emergence of COVID.
Whether it represents a new form of acute hepatitis, a continuing increase in cases of hepatitis, or an increased awareness because of the well-publicized alerts remains to be determined. We certainly saw “viral-induced hepatitis” in the past.
Young patients may first be brought to pediatricians. What, if anything, should pediatricians be on the lookout for? Do they need a heightened index of suspicion or are the cases too rare at this point?
An awareness of the “outbreak” may allow the clinician to extend the typical workup of a child presenting with an undefined, presumably viral illness.
In the cases reported, the prodromal and/or presenting symptoms were respiratory and gastrointestinal in nature. They include nausea, vomiting, diarrhea, and abdominal pain.
Specifically, if jaundice and/or scleral icterus is noted, then hepatitis should be suspected.
Should pediatricians consider early referral to a pediatric gastroenterologist or hepatologist?
Yes, because there is the potential for finding a treatable cause (for example, autoimmune hepatitis or a specific metabolic disease) in a patient presenting in this fashion.
In addition, the potential for progression to acute liver failure (with coagulopathy and encephalopathy), albeit rare, exists.
What do hepatologists need to be doing when presented with suspected cases?
The typical clinical picture holds and the workup is standard. The one new key, given the recent data, is to test for adenovirus, using whole blood versus plasma, as the former may be more sensitive.
In addition, it is prudent to check for SARS-CoV-2 by PCR.
What are the major questions that remain and that you’d like to see elucidated going forward?
There are many. Is this a new disease? A new variant of adenovirus? A synergy or susceptibility related to SARS-CoV-2? Is it related to a variant of SARS-CoV-2? Is it triggering an adverse immune response? Are there other epigenetic factors involved? And finally, is this an increase, or is it related to a collective heightened concern following the pandemic?
Dr. Balistreri is the Dorothy M.M. Kersten Professor of Pediatrics, director emeritus of the Pediatric Liver Care Center, medical director emeritus of liver transplantation, and professor at the University of Cincinnati; he is also with the department of pediatrics at Cincinnati Children’s Hospital Medical Center.
A version of this article first appeared on Medscape.com.
Flu vaccine linked to lower risk for stroke: INTERSTROKE
in a large new case-control study.
“While influenza vaccination is a cost-effective method to prevent influenza, it is also an effective way to reduce the burden of stroke,” said study author Christopher Schwarzbach, MD, of Ludwigshafen (Germany) Hospital.
“Our results therefore encourage the wider use of influenza vaccination,” he concluded.
Dr. Schwarzbach presented these data from the INTERSTROKE study at the 2022 European Stroke Organisation Conference.
He explained that acute inflammatory disease is thought to increase the risk for cerebrovascular events, and the seasonality of influenza-like illness appears to be associated with the seasonality of cardiovascular and cerebrovascular events. Previous observational studies have also shown a link between influenza vaccination and a reduced risk for stroke.
The current INTERSTROKE study was a large international case-control study conducted between 2007 and 2015 that involved 13,447 cases (patients within 5 days of their first stroke) and a similar number of age- and gender-matched people from 32 countries across the world.
All cases and control subjects were systematically asked whether they had acute febrile illness in the previous 4 weeks and whether they had received an influenza vaccination within the previous year.
Conditional logistical regression was used to quantify the results, with adjustment for 13 different possible confounding factors, including hypertension, activity, smoking, cardiovascular risk factors, and socioeconomic factors.
Results showed that having had an acute febrile illness in the previous 4 weeks was more commonly reported in the patients with an acute ischemic stroke (8.7%) than in control patients (5.6%). After adjustment for confounding factors, this gives an adjusted risk ratio of 1.18, which was of borderline statistical significance (95% confidence limits, 1.01-1.39), Dr. Schwarzbach reported.
The association between recent febrile illness and acute ischemic stroke was stronger when compared with community control subjects (adjusted odds ratio, 2.0), but it was absent when compared with hospital control subjects.
The association was also only apparent in Australia, China, North America, and Western Europe; it was not seen in other parts of the world.
There was no association between acute febrile illness and acute cerebral hemorrhage.
Flu vaccine linked to halving of stroke risk
Having received a flu vaccine in the previous year was strongly associated with a lower risk for any type of stroke (aOR, 0.53), ischemic stroke (aOR, 0.57), and hemorrhagic stroke (aOR, 0.34).
Dr. Schwarzbach noted that these results were also consistent in an extended statistical model that included variables that might reflect a willingness to be vaccinated and when compared with both community and hospital-based control subjects.
The strength of the association between influenza vaccination and reduced risk for stroke was similar when compared with either community or hospital control subjects, and was only moderately stronger during than outside the influenza season.
The association was also seen in all regions of the world apart from Africa and South Asia, Dr. Schwarzbach reported, but he noted that vaccination rates in these two regions were extremely low.
The researchers also found that the magnitude of the associations between flu vaccination and lower risk for stroke were stronger in individuals who had multiple annual vaccinations, with an odds ratio of 0.54 in those who had received a vaccine every year for the previous 5 years, and of 0.79 in those who had received one to four vaccinations in the previous 5 years.
Mechanism: Immune stimulation?
Discussing possible mechanisms behind these results, Dr. Schwarzbach noted that the finding that the association with influenza vaccination and reduced stroke risk was independent of seasonality was surprising. “We had expected the protective effect of vaccination to be bigger during the influenza season, but this wasn’t the case.”
He suggested that one explanation might be the inclusion of regions of the world where this seasonality doesn’t exist.
But he pointed out that the finding of a stronger association between flu vaccination and lower stroke risk in those who had received more vaccinations has given rise to another theory: that it is the stimulation of the immune system rather than the protection of infection against influenza that is the key factor.
In an interview with Dr. Schwarzbach, Guillaume Turk, MD, professor of neurology at GHU Paris, pointed out that causal inferences are always difficult in case-control studies and in clinical epidemiology in general.
“What makes you think that this association between influenza vaccination and decreased risk is causal rather than due to unmeasured confounders? For example, patients who received vaccination may have received more medical attention and may have been more aware of the risk factors for stroke,” he asked.
Dr. Schwarzbach replied: “Yes, this is the issue of healthy user bias, which is always a problem in this type of research and is hard to address.”
“What we tried to do here is to adjust for variables that might influence the willingness of people to get vaccinated,” he added. “These were mainly socioeconomic factors. But, of course, this is something that we can’t rule out.”
Dr. Schwarzbach noted that, for more reliable information on this association, prospective studies are needed.
‘A plausible effect’
Discussing the study after the presentation, William Whiteley, BM, PhD, a clinical epidemiologist at the University of Edinburgh and a consultant neurologist in NHS Lothian, said vaccination was a potentially important way to reduce stroke.
“In this study, there was a plausible effect on reducing stroke incidence from vaccination against influenza, and also a plausible increase in the risk of stroke from having a recent febrile illness, which we have seen in other studies,” he commented.
Dr. Whiteley noted that this observation was particularly relevant now because of the COVID-19 pandemic.
“We’ve all been worried about the risk of heart attack and stroke after COVID, where we’ve seen quite early high risks, and we are also optimistic about the effect of vaccination on reducing those incidences. We’ve seen data from the U.K. that there may be around a 20% reduction in risk of stroke from vaccination. So, it’s all quite plausible, but at the moment it’s all based on observational evidence and we really need some randomized evidence,” he said.
“Vaccination and infections have all sorts of odd confounders,” he added. “People who get vaccines tend to be more healthy than those who don’t get vaccines, so you can start to see quite implausible effects of vaccination on overall mortality, which probably aren’t real, and you probably can’t get rid of that totally with statistical methods.”
Alastair Webb, MD, University of Oxford (England), asked how reliable the current findings were, given that the occurrence of febrile illnesses and receipt of vaccines were all self-reported, and although there was an association for ischemic stroke and febrile illness, this seemed to go in the opposite direction for hemorrhagic stroke. He also noted that the 50% reduction in stroke risk with vaccination in this study seemed “quite a large magnitude of effect.”
Dr. Whiteley replied: “Yes, it is large, but it is promising.” He cited a previous meta-analysis of randomized studies that showed a roughly 25%-35% reduction in vascular events after flu vaccination, but noted that there was a lot of heterogeneity between studies.
“I’m not sure we’re going to see much more randomized evidence, but I think we can probably all agree that having a vaccine against flu or COVID is a good thing for all of us,” Dr. Whiteley concluded.
The INTERSTROKE study was funded by the Canadian Institutes of Health Research, Heart and Stroke Foundation of Canada, Canadian Stroke Network, Health Research Board Ireland, Swedish Research Council, Swedish Heart and Lung Foundation, The Health & Medical Care Committee of the Regional Executive Board, Region Vastra Gotaland (Sweden), AstraZeneca, Boehringer Ingelheim, Pfizer, MSD, Chest, Heart and Stroke Scotland, and The Stroke Association, with support from The UK Stroke Research Network. The authors reported no relevant disclosures.
A version of this article first appeared on Medscape.com.
in a large new case-control study.
“While influenza vaccination is a cost-effective method to prevent influenza, it is also an effective way to reduce the burden of stroke,” said study author Christopher Schwarzbach, MD, of Ludwigshafen (Germany) Hospital.
“Our results therefore encourage the wider use of influenza vaccination,” he concluded.
Dr. Schwarzbach presented these data from the INTERSTROKE study at the 2022 European Stroke Organisation Conference.
He explained that acute inflammatory disease is thought to increase the risk for cerebrovascular events, and the seasonality of influenza-like illness appears to be associated with the seasonality of cardiovascular and cerebrovascular events. Previous observational studies have also shown a link between influenza vaccination and a reduced risk for stroke.
The current INTERSTROKE study was a large international case-control study conducted between 2007 and 2015 that involved 13,447 cases (patients within 5 days of their first stroke) and a similar number of age- and gender-matched people from 32 countries across the world.
All cases and control subjects were systematically asked whether they had acute febrile illness in the previous 4 weeks and whether they had received an influenza vaccination within the previous year.
Conditional logistical regression was used to quantify the results, with adjustment for 13 different possible confounding factors, including hypertension, activity, smoking, cardiovascular risk factors, and socioeconomic factors.
Results showed that having had an acute febrile illness in the previous 4 weeks was more commonly reported in the patients with an acute ischemic stroke (8.7%) than in control patients (5.6%). After adjustment for confounding factors, this gives an adjusted risk ratio of 1.18, which was of borderline statistical significance (95% confidence limits, 1.01-1.39), Dr. Schwarzbach reported.
The association between recent febrile illness and acute ischemic stroke was stronger when compared with community control subjects (adjusted odds ratio, 2.0), but it was absent when compared with hospital control subjects.
The association was also only apparent in Australia, China, North America, and Western Europe; it was not seen in other parts of the world.
There was no association between acute febrile illness and acute cerebral hemorrhage.
Flu vaccine linked to halving of stroke risk
Having received a flu vaccine in the previous year was strongly associated with a lower risk for any type of stroke (aOR, 0.53), ischemic stroke (aOR, 0.57), and hemorrhagic stroke (aOR, 0.34).
Dr. Schwarzbach noted that these results were also consistent in an extended statistical model that included variables that might reflect a willingness to be vaccinated and when compared with both community and hospital-based control subjects.
The strength of the association between influenza vaccination and reduced risk for stroke was similar when compared with either community or hospital control subjects, and was only moderately stronger during than outside the influenza season.
The association was also seen in all regions of the world apart from Africa and South Asia, Dr. Schwarzbach reported, but he noted that vaccination rates in these two regions were extremely low.
The researchers also found that the magnitude of the associations between flu vaccination and lower risk for stroke were stronger in individuals who had multiple annual vaccinations, with an odds ratio of 0.54 in those who had received a vaccine every year for the previous 5 years, and of 0.79 in those who had received one to four vaccinations in the previous 5 years.
Mechanism: Immune stimulation?
Discussing possible mechanisms behind these results, Dr. Schwarzbach noted that the finding that the association with influenza vaccination and reduced stroke risk was independent of seasonality was surprising. “We had expected the protective effect of vaccination to be bigger during the influenza season, but this wasn’t the case.”
He suggested that one explanation might be the inclusion of regions of the world where this seasonality doesn’t exist.
But he pointed out that the finding of a stronger association between flu vaccination and lower stroke risk in those who had received more vaccinations has given rise to another theory: that it is the stimulation of the immune system rather than the protection of infection against influenza that is the key factor.
In an interview with Dr. Schwarzbach, Guillaume Turk, MD, professor of neurology at GHU Paris, pointed out that causal inferences are always difficult in case-control studies and in clinical epidemiology in general.
“What makes you think that this association between influenza vaccination and decreased risk is causal rather than due to unmeasured confounders? For example, patients who received vaccination may have received more medical attention and may have been more aware of the risk factors for stroke,” he asked.
Dr. Schwarzbach replied: “Yes, this is the issue of healthy user bias, which is always a problem in this type of research and is hard to address.”
“What we tried to do here is to adjust for variables that might influence the willingness of people to get vaccinated,” he added. “These were mainly socioeconomic factors. But, of course, this is something that we can’t rule out.”
Dr. Schwarzbach noted that, for more reliable information on this association, prospective studies are needed.
‘A plausible effect’
Discussing the study after the presentation, William Whiteley, BM, PhD, a clinical epidemiologist at the University of Edinburgh and a consultant neurologist in NHS Lothian, said vaccination was a potentially important way to reduce stroke.
“In this study, there was a plausible effect on reducing stroke incidence from vaccination against influenza, and also a plausible increase in the risk of stroke from having a recent febrile illness, which we have seen in other studies,” he commented.
Dr. Whiteley noted that this observation was particularly relevant now because of the COVID-19 pandemic.
“We’ve all been worried about the risk of heart attack and stroke after COVID, where we’ve seen quite early high risks, and we are also optimistic about the effect of vaccination on reducing those incidences. We’ve seen data from the U.K. that there may be around a 20% reduction in risk of stroke from vaccination. So, it’s all quite plausible, but at the moment it’s all based on observational evidence and we really need some randomized evidence,” he said.
“Vaccination and infections have all sorts of odd confounders,” he added. “People who get vaccines tend to be more healthy than those who don’t get vaccines, so you can start to see quite implausible effects of vaccination on overall mortality, which probably aren’t real, and you probably can’t get rid of that totally with statistical methods.”
Alastair Webb, MD, University of Oxford (England), asked how reliable the current findings were, given that the occurrence of febrile illnesses and receipt of vaccines were all self-reported, and although there was an association for ischemic stroke and febrile illness, this seemed to go in the opposite direction for hemorrhagic stroke. He also noted that the 50% reduction in stroke risk with vaccination in this study seemed “quite a large magnitude of effect.”
Dr. Whiteley replied: “Yes, it is large, but it is promising.” He cited a previous meta-analysis of randomized studies that showed a roughly 25%-35% reduction in vascular events after flu vaccination, but noted that there was a lot of heterogeneity between studies.
“I’m not sure we’re going to see much more randomized evidence, but I think we can probably all agree that having a vaccine against flu or COVID is a good thing for all of us,” Dr. Whiteley concluded.
The INTERSTROKE study was funded by the Canadian Institutes of Health Research, Heart and Stroke Foundation of Canada, Canadian Stroke Network, Health Research Board Ireland, Swedish Research Council, Swedish Heart and Lung Foundation, The Health & Medical Care Committee of the Regional Executive Board, Region Vastra Gotaland (Sweden), AstraZeneca, Boehringer Ingelheim, Pfizer, MSD, Chest, Heart and Stroke Scotland, and The Stroke Association, with support from The UK Stroke Research Network. The authors reported no relevant disclosures.
A version of this article first appeared on Medscape.com.
in a large new case-control study.
“While influenza vaccination is a cost-effective method to prevent influenza, it is also an effective way to reduce the burden of stroke,” said study author Christopher Schwarzbach, MD, of Ludwigshafen (Germany) Hospital.
“Our results therefore encourage the wider use of influenza vaccination,” he concluded.
Dr. Schwarzbach presented these data from the INTERSTROKE study at the 2022 European Stroke Organisation Conference.
He explained that acute inflammatory disease is thought to increase the risk for cerebrovascular events, and the seasonality of influenza-like illness appears to be associated with the seasonality of cardiovascular and cerebrovascular events. Previous observational studies have also shown a link between influenza vaccination and a reduced risk for stroke.
The current INTERSTROKE study was a large international case-control study conducted between 2007 and 2015 that involved 13,447 cases (patients within 5 days of their first stroke) and a similar number of age- and gender-matched people from 32 countries across the world.
All cases and control subjects were systematically asked whether they had acute febrile illness in the previous 4 weeks and whether they had received an influenza vaccination within the previous year.
Conditional logistical regression was used to quantify the results, with adjustment for 13 different possible confounding factors, including hypertension, activity, smoking, cardiovascular risk factors, and socioeconomic factors.
Results showed that having had an acute febrile illness in the previous 4 weeks was more commonly reported in the patients with an acute ischemic stroke (8.7%) than in control patients (5.6%). After adjustment for confounding factors, this gives an adjusted risk ratio of 1.18, which was of borderline statistical significance (95% confidence limits, 1.01-1.39), Dr. Schwarzbach reported.
The association between recent febrile illness and acute ischemic stroke was stronger when compared with community control subjects (adjusted odds ratio, 2.0), but it was absent when compared with hospital control subjects.
The association was also only apparent in Australia, China, North America, and Western Europe; it was not seen in other parts of the world.
There was no association between acute febrile illness and acute cerebral hemorrhage.
Flu vaccine linked to halving of stroke risk
Having received a flu vaccine in the previous year was strongly associated with a lower risk for any type of stroke (aOR, 0.53), ischemic stroke (aOR, 0.57), and hemorrhagic stroke (aOR, 0.34).
Dr. Schwarzbach noted that these results were also consistent in an extended statistical model that included variables that might reflect a willingness to be vaccinated and when compared with both community and hospital-based control subjects.
The strength of the association between influenza vaccination and reduced risk for stroke was similar when compared with either community or hospital control subjects, and was only moderately stronger during than outside the influenza season.
The association was also seen in all regions of the world apart from Africa and South Asia, Dr. Schwarzbach reported, but he noted that vaccination rates in these two regions were extremely low.
The researchers also found that the magnitude of the associations between flu vaccination and lower risk for stroke were stronger in individuals who had multiple annual vaccinations, with an odds ratio of 0.54 in those who had received a vaccine every year for the previous 5 years, and of 0.79 in those who had received one to four vaccinations in the previous 5 years.
Mechanism: Immune stimulation?
Discussing possible mechanisms behind these results, Dr. Schwarzbach noted that the finding that the association with influenza vaccination and reduced stroke risk was independent of seasonality was surprising. “We had expected the protective effect of vaccination to be bigger during the influenza season, but this wasn’t the case.”
He suggested that one explanation might be the inclusion of regions of the world where this seasonality doesn’t exist.
But he pointed out that the finding of a stronger association between flu vaccination and lower stroke risk in those who had received more vaccinations has given rise to another theory: that it is the stimulation of the immune system rather than the protection of infection against influenza that is the key factor.
In an interview with Dr. Schwarzbach, Guillaume Turk, MD, professor of neurology at GHU Paris, pointed out that causal inferences are always difficult in case-control studies and in clinical epidemiology in general.
“What makes you think that this association between influenza vaccination and decreased risk is causal rather than due to unmeasured confounders? For example, patients who received vaccination may have received more medical attention and may have been more aware of the risk factors for stroke,” he asked.
Dr. Schwarzbach replied: “Yes, this is the issue of healthy user bias, which is always a problem in this type of research and is hard to address.”
“What we tried to do here is to adjust for variables that might influence the willingness of people to get vaccinated,” he added. “These were mainly socioeconomic factors. But, of course, this is something that we can’t rule out.”
Dr. Schwarzbach noted that, for more reliable information on this association, prospective studies are needed.
‘A plausible effect’
Discussing the study after the presentation, William Whiteley, BM, PhD, a clinical epidemiologist at the University of Edinburgh and a consultant neurologist in NHS Lothian, said vaccination was a potentially important way to reduce stroke.
“In this study, there was a plausible effect on reducing stroke incidence from vaccination against influenza, and also a plausible increase in the risk of stroke from having a recent febrile illness, which we have seen in other studies,” he commented.
Dr. Whiteley noted that this observation was particularly relevant now because of the COVID-19 pandemic.
“We’ve all been worried about the risk of heart attack and stroke after COVID, where we’ve seen quite early high risks, and we are also optimistic about the effect of vaccination on reducing those incidences. We’ve seen data from the U.K. that there may be around a 20% reduction in risk of stroke from vaccination. So, it’s all quite plausible, but at the moment it’s all based on observational evidence and we really need some randomized evidence,” he said.
“Vaccination and infections have all sorts of odd confounders,” he added. “People who get vaccines tend to be more healthy than those who don’t get vaccines, so you can start to see quite implausible effects of vaccination on overall mortality, which probably aren’t real, and you probably can’t get rid of that totally with statistical methods.”
Alastair Webb, MD, University of Oxford (England), asked how reliable the current findings were, given that the occurrence of febrile illnesses and receipt of vaccines were all self-reported, and although there was an association for ischemic stroke and febrile illness, this seemed to go in the opposite direction for hemorrhagic stroke. He also noted that the 50% reduction in stroke risk with vaccination in this study seemed “quite a large magnitude of effect.”
Dr. Whiteley replied: “Yes, it is large, but it is promising.” He cited a previous meta-analysis of randomized studies that showed a roughly 25%-35% reduction in vascular events after flu vaccination, but noted that there was a lot of heterogeneity between studies.
“I’m not sure we’re going to see much more randomized evidence, but I think we can probably all agree that having a vaccine against flu or COVID is a good thing for all of us,” Dr. Whiteley concluded.
The INTERSTROKE study was funded by the Canadian Institutes of Health Research, Heart and Stroke Foundation of Canada, Canadian Stroke Network, Health Research Board Ireland, Swedish Research Council, Swedish Heart and Lung Foundation, The Health & Medical Care Committee of the Regional Executive Board, Region Vastra Gotaland (Sweden), AstraZeneca, Boehringer Ingelheim, Pfizer, MSD, Chest, Heart and Stroke Scotland, and The Stroke Association, with support from The UK Stroke Research Network. The authors reported no relevant disclosures.
A version of this article first appeared on Medscape.com.
Innocent doc sued after 'secret' medical expert says claim has merit
When the hospital’s trauma team could not get an IV inserted into an accident victim, they called Illinois emergency physician William Sullivan, DO, JD, for help. Dr. Sullivan, who is based in the Chicago suburb of Frankfort, inserted a central line into the patient’s leg on his first attempt – a task that took about 20 minutes.
A year later, Dr. Sullivan was shocked and angry to learn he was being sued by the trauma patient’s family. Inserting the line was his only interaction with the woman, and he had no role in her care management, he said. Yet, the suit claimed he was negligent for failing to diagnose the patient with internal bleeding and for not performing surgery.
“The lawsuit put a lot of stress on our family,” Dr. Sullivan recalled. “At the time my wife was pregnant. I was in law school, and I was also working full time in the ER to support our family. I remember my wife crying on the couch after reading the complaint and asking how the plaintiff’s attorney could get away with making the allegations he made.”
Dr. Sullivan soon learned that 15 medical providers in the patient’s medical record were named as defendants. This included the director of the radiology department, whose name was on a radiology report as “director” but who was actually out of the country when the incident occurred.
Despite some of the accusations being impossible, a medical expert had claimed there was a “meritorious claim” against every health professional named in the suit. Illinois is among the 28 states that require plaintiffs’ attorneys to file an affidavit of merit for medical malpractice claims to move forward.
Dr. Sullivan wondered who would endorse such outlandish accusations, but the expert’s identity was a mystery. According to Illinois law, About one-third of states with merit requirements permit anonymous experts, according to research and attorneys familiar with the issue.
Because the expert’s identity remains hidden, physicians have no way of knowing whether they were qualified to render an opinion, Dr. Sullivan said. The loopholes can drag out frivolous claims and waste significant time and expense, say legal experts. Frequently, it takes a year or more before innocent physicians are dismissed from unfounded lawsuits by the court or dropped when plaintiffs can’t support the claim.
“It’s hugely frustrating,” said Bruce Montoya, JD, a Colorado medical liability defense attorney. “You have an expert who is not disclosed. Further down the road, when experts are being deposed, the plaintiff does not have to reveal whether any of those testifying experts is the same one who certified the case. You never get to determine whether they, in fact, had a certificate reviewer who was legitimate.”
The laws have led to a recent outcry among physicians and fueled a revised resolution by the American College of Emergency Physicians (ACEP) denouncing anonymous affidavits of merit. (The revision has not yet been published online.)
“The minute experts are identified, they can be vetted,” said Rade B. Vukmir, MD, JD, chair of ACEP’s Medical Legal Committee. “There are reasons that you want to clarify the qualification and veracity of the witness. [Anonymous affidavits of merit] don’t allow that, and there’s something inherently wrong with that.”
Because the identities of consulting experts are unknown, it’s hard to know how many are unqualified. Expert witnesses who testify during trials, on the other hand, have long come under scrutiny for questionable qualifications. Some have come under fire for allegedly lying under oath about their experience, misrepresenting their credentials, and falsely representing their knowledge.
“Considering the known problem of potentially unethical expert witness testimony at trial, there’s is the potential likelihood that experts in anonymous affidavits of merit may sometimes lack the qualifications to give opinions,” said Dr. Vukmir, an emergency care physician in Pittsburgh.
Attorneys: Hidden experts increase costs, waste time
In Colorado, Mr. Montoya has seen firsthand how anonymous experts can prolong questionable claims and burden defendants.
Like Illinois, Colorado does not require attorneys to identify the medical experts used to fulfill its certificate of review statute. The expert consulted must have expertise in the same area of the alleged negligence, but does not have to practice in the same specialty, and the statute allows one expert to certify a lawsuit against multiple doctors.
In a recent case, Mr. Montoya represented a Denver neurosurgeon who was sued along with multiple other health care professionals. From the outset, Mr. Montoya argued the claim had no merit against the neurosurgeon, but the plaintiff’s attorney refused to dismiss the physician. Mr. Montoya asked whether the expert consulted for the certificate of merit was a neurosurgeon, but the attorney declined to disclose that information, he said.
The case progressed and Mr. Montoya eventually asked the judge to review the certificate of merit. By law, a judge can confidentially review the certificate of merit and decide whether it aligns with the state statute, but without disclosing the expert’s identity to the defense. The judge ruled the certificate appeared to conform with state law, and the case continued.
A year later, as both sides were getting ready to disclose their experts who would testify, Mr. Montoya again argued the neurosurgeon should be dropped from the suit. This time, he warned if the claim continued against the neurosurgeon, the defense would be filing a motion for summary judgment and pursuing attorney fees and costs. Colorado law allows for such fees if the filing or pursuit of an action is frivolous.
“Boom, my client was dismissed,” Mr. Montoya said. “This is a year later, after multiple conferences among the attorneys, multiple pleadings filed, expert witnesses retained to review the care, discovery exchanged, and records obtained. If we had [a stronger] certificate of review statute, it would have been a different ballgame. It’s never going to get a year down the road.”
In New York, physician defendants have experienced similar woes. The state’s law requires plaintiffs’ attorneys to certify that they consulted with a physician prior to filing the claim, and that they believe based on that discussion, there’s a reasonable basis for the claim to move forward. Attorneys are not required to disclose the expert’s identity.
The law also allows “an out,” explained Morris Auster, JD, senior vice president and chief legislative counsel for the Medical Society of the State of New York. If the attorney made three separate attempts to obtain a consultation, and all three experts would not agree to the consultation, the lawsuit can be filed anyway, he said.
“From our standpoint, it’s important to have an affidavit of merit requirement; it’s better than not having it,” Mr. Auster said. “But its effectiveness in providing control over the filing of lawsuits in New York has never been as strong as it could’ve been.”
Mr. Auster notes that New York has some of the highest liability costs in the country in addition to doctors paying some of the steepest medical liability insurance premiums.
“This really affects a lot of physicians and it’s driving physicians into employment arrangements, so they don’t have to deal with it on their own,” he said. “We support a number of measures to address these significantly high costs, and stronger certificate of merit requirements would certainly be one of those advocacy goals.”
Why are anonymous experts allowed?
Certificates of merit that shield the identity of consultants encourage a greater pool of physicians willing to review cases, said J. Matthew Dudley, JD, president of the Illinois Trial Lawyers Association. When the requirements first went into effect in Illinois, there was significant animosity among physicians toward doctors who testified in medical malpractice cases for patients, Mr. Dudley explained.
“Sometimes they would be ostracized from their professional societies, or it would hurt a referral relationship.” he said. “Over time, that animosity has lessened, but there was a concern that if the identity of physicians in certificates of merit weren’t protected, then doctors would not look at cases for patients.”
This would result in additional barriers for patients and their attorneys in pursuing their legal rights, Mr. Dudley said. He said Illinois’ certificate of merit statute is successful in fulfilling its intended purpose, and he has not seen any statistical evidence to suggest otherwise.
“It has proven effective at decreasing filings in medical malpractice and effectively screening medical malpractice cases,” he said. “Certificates of merit help to decrease filings by firms that aren’t that experienced in dealing with those kinds of cases.”
Kentucky is another state that does not require attorneys to identity the experts consulted for certificates of merit. Malpractice defense attorney Andrew DeSimone, JD, who practices in Kentucky, said this isn’t a problem since attorneys eventually must disclose the expert witnesses who will testify at trial.
“Knowing the name behind the certificate of merit is not that pertinent,” Mr. DeSimone said. “Physicians and their attorneys will ultimately have the chance to question and evaluate the expert witnesses used at trial. The certificate of merit is designed to weed out totally frivolous cases that do not have expert support. It’s not designed to be a trial on the merits.”
The belief that plaintiffs’ attorneys frequently bring weak cases and use unqualified experts to certify claims is not realistic or logical, added Sean Domnick, JD, a Florida medical malpractice attorney and vice president for the American Association for Justice. Medical malpractice cases are extremely challenging for plaintiffs – and they’re expensive, Mr. Domnick said.
“We can’t afford to take bad cases,” he said. “For me to take on a medical malpractice case, it’s not unusual for me to spend well over $100,000. Remember, if we lose, I don’t get that money back and I don’t get paid. Why in the world would a plaintiff take on that type of a burden for a case they didn’t believe in? The logic escapes me.”
In Florida, where Mr. Domnick practices, plaintiffs’ attorneys must send their certificates of merit to the defense with the expert identified. Domnick believes the requirement is a hindrance.
“It creates a delay that is unnecessary in a system that is already designed to wear our clients down,” he said. “It’s just another component that makes it harder on them.”
Hidden experts may insulate plaintiffs’ attorneys from liability
Dr. Sullivan, the Illinois emergency physician, was ultimately dismissed from the multiparty lawsuit, but not for roughly 18 months. After the dismissal, he fought back. He sued the plaintiff’s law firm for malicious prosecution, negligence in hiring, and relying on the opinion of an expert who was unqualified to render an opinion against an emergency physician.
The law firm, however, argued that it was immune from liability because it reasonably relied on the expert’s opinion as required by Illinois law. A trial court agreed with the plaintiffs’ firm. The judge denied Dr. Sullivan’s request to identify the expert, ruling there was no finding that the affidavit was untrue or made without reasonable cause. Dr. Sullivan appealed, and the appellate court upheld the trial’s court decision.
“As happened with my case, law firms can use the affidavit as a defense against countersuits or motions for sanctions,” Dr. Sullivan said. “Although the certificate of merit is intended to prevent attorneys from filing frivolous cases, it can also have the opposite effect of helping to insulate plaintiff attorneys from liability for filing a frivolous lawsuit.”
In Colorado, complaints about the state’s certificate of merit statute have gone before the Colorado Supreme Court. In one case, a lower court ruled that a certificate of merit was deficient because the consultants were not chiropractors. In another case, a nurse defendant argued the claim’s certificate of review was insufficient because the consulting expert was a physician.
In both instances, Colorado judges held the state’s statute does not require consultants to be in the same profession or the same specialty as the health professional defendant.
In New York, meanwhile, Mr. Auster said several bills to strengthen the state’s certificate of merit requirements have failed in recent years.
“It’s hard to say whether it will improve anytime soon,” he said. “The trial lawyers are a very powerful advocacy force in the state, and they tend to oppose even the slightest of changes in civil liability. [In addition], some of these issues have been put on a lower tier because of trying to manage the pandemic.”
Ultimately, Dr. Sullivan said that courts and legislatures need to strongly consider the ethics of allowing anonymous experts to provide testimony against defendant physicians.
“I also think we need to consider how the notion of a secret expert comports with a defendant physician’s due process,” he said. “If an expert’s opinion is appropriate, why would there be a need to shroud one’s identity in a veil of secrecy?”
A version of this article first appeared on Medscape.com.
When the hospital’s trauma team could not get an IV inserted into an accident victim, they called Illinois emergency physician William Sullivan, DO, JD, for help. Dr. Sullivan, who is based in the Chicago suburb of Frankfort, inserted a central line into the patient’s leg on his first attempt – a task that took about 20 minutes.
A year later, Dr. Sullivan was shocked and angry to learn he was being sued by the trauma patient’s family. Inserting the line was his only interaction with the woman, and he had no role in her care management, he said. Yet, the suit claimed he was negligent for failing to diagnose the patient with internal bleeding and for not performing surgery.
“The lawsuit put a lot of stress on our family,” Dr. Sullivan recalled. “At the time my wife was pregnant. I was in law school, and I was also working full time in the ER to support our family. I remember my wife crying on the couch after reading the complaint and asking how the plaintiff’s attorney could get away with making the allegations he made.”
Dr. Sullivan soon learned that 15 medical providers in the patient’s medical record were named as defendants. This included the director of the radiology department, whose name was on a radiology report as “director” but who was actually out of the country when the incident occurred.
Despite some of the accusations being impossible, a medical expert had claimed there was a “meritorious claim” against every health professional named in the suit. Illinois is among the 28 states that require plaintiffs’ attorneys to file an affidavit of merit for medical malpractice claims to move forward.
Dr. Sullivan wondered who would endorse such outlandish accusations, but the expert’s identity was a mystery. According to Illinois law, About one-third of states with merit requirements permit anonymous experts, according to research and attorneys familiar with the issue.
Because the expert’s identity remains hidden, physicians have no way of knowing whether they were qualified to render an opinion, Dr. Sullivan said. The loopholes can drag out frivolous claims and waste significant time and expense, say legal experts. Frequently, it takes a year or more before innocent physicians are dismissed from unfounded lawsuits by the court or dropped when plaintiffs can’t support the claim.
“It’s hugely frustrating,” said Bruce Montoya, JD, a Colorado medical liability defense attorney. “You have an expert who is not disclosed. Further down the road, when experts are being deposed, the plaintiff does not have to reveal whether any of those testifying experts is the same one who certified the case. You never get to determine whether they, in fact, had a certificate reviewer who was legitimate.”
The laws have led to a recent outcry among physicians and fueled a revised resolution by the American College of Emergency Physicians (ACEP) denouncing anonymous affidavits of merit. (The revision has not yet been published online.)
“The minute experts are identified, they can be vetted,” said Rade B. Vukmir, MD, JD, chair of ACEP’s Medical Legal Committee. “There are reasons that you want to clarify the qualification and veracity of the witness. [Anonymous affidavits of merit] don’t allow that, and there’s something inherently wrong with that.”
Because the identities of consulting experts are unknown, it’s hard to know how many are unqualified. Expert witnesses who testify during trials, on the other hand, have long come under scrutiny for questionable qualifications. Some have come under fire for allegedly lying under oath about their experience, misrepresenting their credentials, and falsely representing their knowledge.
“Considering the known problem of potentially unethical expert witness testimony at trial, there’s is the potential likelihood that experts in anonymous affidavits of merit may sometimes lack the qualifications to give opinions,” said Dr. Vukmir, an emergency care physician in Pittsburgh.
Attorneys: Hidden experts increase costs, waste time
In Colorado, Mr. Montoya has seen firsthand how anonymous experts can prolong questionable claims and burden defendants.
Like Illinois, Colorado does not require attorneys to identify the medical experts used to fulfill its certificate of review statute. The expert consulted must have expertise in the same area of the alleged negligence, but does not have to practice in the same specialty, and the statute allows one expert to certify a lawsuit against multiple doctors.
In a recent case, Mr. Montoya represented a Denver neurosurgeon who was sued along with multiple other health care professionals. From the outset, Mr. Montoya argued the claim had no merit against the neurosurgeon, but the plaintiff’s attorney refused to dismiss the physician. Mr. Montoya asked whether the expert consulted for the certificate of merit was a neurosurgeon, but the attorney declined to disclose that information, he said.
The case progressed and Mr. Montoya eventually asked the judge to review the certificate of merit. By law, a judge can confidentially review the certificate of merit and decide whether it aligns with the state statute, but without disclosing the expert’s identity to the defense. The judge ruled the certificate appeared to conform with state law, and the case continued.
A year later, as both sides were getting ready to disclose their experts who would testify, Mr. Montoya again argued the neurosurgeon should be dropped from the suit. This time, he warned if the claim continued against the neurosurgeon, the defense would be filing a motion for summary judgment and pursuing attorney fees and costs. Colorado law allows for such fees if the filing or pursuit of an action is frivolous.
“Boom, my client was dismissed,” Mr. Montoya said. “This is a year later, after multiple conferences among the attorneys, multiple pleadings filed, expert witnesses retained to review the care, discovery exchanged, and records obtained. If we had [a stronger] certificate of review statute, it would have been a different ballgame. It’s never going to get a year down the road.”
In New York, physician defendants have experienced similar woes. The state’s law requires plaintiffs’ attorneys to certify that they consulted with a physician prior to filing the claim, and that they believe based on that discussion, there’s a reasonable basis for the claim to move forward. Attorneys are not required to disclose the expert’s identity.
The law also allows “an out,” explained Morris Auster, JD, senior vice president and chief legislative counsel for the Medical Society of the State of New York. If the attorney made three separate attempts to obtain a consultation, and all three experts would not agree to the consultation, the lawsuit can be filed anyway, he said.
“From our standpoint, it’s important to have an affidavit of merit requirement; it’s better than not having it,” Mr. Auster said. “But its effectiveness in providing control over the filing of lawsuits in New York has never been as strong as it could’ve been.”
Mr. Auster notes that New York has some of the highest liability costs in the country in addition to doctors paying some of the steepest medical liability insurance premiums.
“This really affects a lot of physicians and it’s driving physicians into employment arrangements, so they don’t have to deal with it on their own,” he said. “We support a number of measures to address these significantly high costs, and stronger certificate of merit requirements would certainly be one of those advocacy goals.”
Why are anonymous experts allowed?
Certificates of merit that shield the identity of consultants encourage a greater pool of physicians willing to review cases, said J. Matthew Dudley, JD, president of the Illinois Trial Lawyers Association. When the requirements first went into effect in Illinois, there was significant animosity among physicians toward doctors who testified in medical malpractice cases for patients, Mr. Dudley explained.
“Sometimes they would be ostracized from their professional societies, or it would hurt a referral relationship.” he said. “Over time, that animosity has lessened, but there was a concern that if the identity of physicians in certificates of merit weren’t protected, then doctors would not look at cases for patients.”
This would result in additional barriers for patients and their attorneys in pursuing their legal rights, Mr. Dudley said. He said Illinois’ certificate of merit statute is successful in fulfilling its intended purpose, and he has not seen any statistical evidence to suggest otherwise.
“It has proven effective at decreasing filings in medical malpractice and effectively screening medical malpractice cases,” he said. “Certificates of merit help to decrease filings by firms that aren’t that experienced in dealing with those kinds of cases.”
Kentucky is another state that does not require attorneys to identity the experts consulted for certificates of merit. Malpractice defense attorney Andrew DeSimone, JD, who practices in Kentucky, said this isn’t a problem since attorneys eventually must disclose the expert witnesses who will testify at trial.
“Knowing the name behind the certificate of merit is not that pertinent,” Mr. DeSimone said. “Physicians and their attorneys will ultimately have the chance to question and evaluate the expert witnesses used at trial. The certificate of merit is designed to weed out totally frivolous cases that do not have expert support. It’s not designed to be a trial on the merits.”
The belief that plaintiffs’ attorneys frequently bring weak cases and use unqualified experts to certify claims is not realistic or logical, added Sean Domnick, JD, a Florida medical malpractice attorney and vice president for the American Association for Justice. Medical malpractice cases are extremely challenging for plaintiffs – and they’re expensive, Mr. Domnick said.
“We can’t afford to take bad cases,” he said. “For me to take on a medical malpractice case, it’s not unusual for me to spend well over $100,000. Remember, if we lose, I don’t get that money back and I don’t get paid. Why in the world would a plaintiff take on that type of a burden for a case they didn’t believe in? The logic escapes me.”
In Florida, where Mr. Domnick practices, plaintiffs’ attorneys must send their certificates of merit to the defense with the expert identified. Domnick believes the requirement is a hindrance.
“It creates a delay that is unnecessary in a system that is already designed to wear our clients down,” he said. “It’s just another component that makes it harder on them.”
Hidden experts may insulate plaintiffs’ attorneys from liability
Dr. Sullivan, the Illinois emergency physician, was ultimately dismissed from the multiparty lawsuit, but not for roughly 18 months. After the dismissal, he fought back. He sued the plaintiff’s law firm for malicious prosecution, negligence in hiring, and relying on the opinion of an expert who was unqualified to render an opinion against an emergency physician.
The law firm, however, argued that it was immune from liability because it reasonably relied on the expert’s opinion as required by Illinois law. A trial court agreed with the plaintiffs’ firm. The judge denied Dr. Sullivan’s request to identify the expert, ruling there was no finding that the affidavit was untrue or made without reasonable cause. Dr. Sullivan appealed, and the appellate court upheld the trial’s court decision.
“As happened with my case, law firms can use the affidavit as a defense against countersuits or motions for sanctions,” Dr. Sullivan said. “Although the certificate of merit is intended to prevent attorneys from filing frivolous cases, it can also have the opposite effect of helping to insulate plaintiff attorneys from liability for filing a frivolous lawsuit.”
In Colorado, complaints about the state’s certificate of merit statute have gone before the Colorado Supreme Court. In one case, a lower court ruled that a certificate of merit was deficient because the consultants were not chiropractors. In another case, a nurse defendant argued the claim’s certificate of review was insufficient because the consulting expert was a physician.
In both instances, Colorado judges held the state’s statute does not require consultants to be in the same profession or the same specialty as the health professional defendant.
In New York, meanwhile, Mr. Auster said several bills to strengthen the state’s certificate of merit requirements have failed in recent years.
“It’s hard to say whether it will improve anytime soon,” he said. “The trial lawyers are a very powerful advocacy force in the state, and they tend to oppose even the slightest of changes in civil liability. [In addition], some of these issues have been put on a lower tier because of trying to manage the pandemic.”
Ultimately, Dr. Sullivan said that courts and legislatures need to strongly consider the ethics of allowing anonymous experts to provide testimony against defendant physicians.
“I also think we need to consider how the notion of a secret expert comports with a defendant physician’s due process,” he said. “If an expert’s opinion is appropriate, why would there be a need to shroud one’s identity in a veil of secrecy?”
A version of this article first appeared on Medscape.com.
When the hospital’s trauma team could not get an IV inserted into an accident victim, they called Illinois emergency physician William Sullivan, DO, JD, for help. Dr. Sullivan, who is based in the Chicago suburb of Frankfort, inserted a central line into the patient’s leg on his first attempt – a task that took about 20 minutes.
A year later, Dr. Sullivan was shocked and angry to learn he was being sued by the trauma patient’s family. Inserting the line was his only interaction with the woman, and he had no role in her care management, he said. Yet, the suit claimed he was negligent for failing to diagnose the patient with internal bleeding and for not performing surgery.
“The lawsuit put a lot of stress on our family,” Dr. Sullivan recalled. “At the time my wife was pregnant. I was in law school, and I was also working full time in the ER to support our family. I remember my wife crying on the couch after reading the complaint and asking how the plaintiff’s attorney could get away with making the allegations he made.”
Dr. Sullivan soon learned that 15 medical providers in the patient’s medical record were named as defendants. This included the director of the radiology department, whose name was on a radiology report as “director” but who was actually out of the country when the incident occurred.
Despite some of the accusations being impossible, a medical expert had claimed there was a “meritorious claim” against every health professional named in the suit. Illinois is among the 28 states that require plaintiffs’ attorneys to file an affidavit of merit for medical malpractice claims to move forward.
Dr. Sullivan wondered who would endorse such outlandish accusations, but the expert’s identity was a mystery. According to Illinois law, About one-third of states with merit requirements permit anonymous experts, according to research and attorneys familiar with the issue.
Because the expert’s identity remains hidden, physicians have no way of knowing whether they were qualified to render an opinion, Dr. Sullivan said. The loopholes can drag out frivolous claims and waste significant time and expense, say legal experts. Frequently, it takes a year or more before innocent physicians are dismissed from unfounded lawsuits by the court or dropped when plaintiffs can’t support the claim.
“It’s hugely frustrating,” said Bruce Montoya, JD, a Colorado medical liability defense attorney. “You have an expert who is not disclosed. Further down the road, when experts are being deposed, the plaintiff does not have to reveal whether any of those testifying experts is the same one who certified the case. You never get to determine whether they, in fact, had a certificate reviewer who was legitimate.”
The laws have led to a recent outcry among physicians and fueled a revised resolution by the American College of Emergency Physicians (ACEP) denouncing anonymous affidavits of merit. (The revision has not yet been published online.)
“The minute experts are identified, they can be vetted,” said Rade B. Vukmir, MD, JD, chair of ACEP’s Medical Legal Committee. “There are reasons that you want to clarify the qualification and veracity of the witness. [Anonymous affidavits of merit] don’t allow that, and there’s something inherently wrong with that.”
Because the identities of consulting experts are unknown, it’s hard to know how many are unqualified. Expert witnesses who testify during trials, on the other hand, have long come under scrutiny for questionable qualifications. Some have come under fire for allegedly lying under oath about their experience, misrepresenting their credentials, and falsely representing their knowledge.
“Considering the known problem of potentially unethical expert witness testimony at trial, there’s is the potential likelihood that experts in anonymous affidavits of merit may sometimes lack the qualifications to give opinions,” said Dr. Vukmir, an emergency care physician in Pittsburgh.
Attorneys: Hidden experts increase costs, waste time
In Colorado, Mr. Montoya has seen firsthand how anonymous experts can prolong questionable claims and burden defendants.
Like Illinois, Colorado does not require attorneys to identify the medical experts used to fulfill its certificate of review statute. The expert consulted must have expertise in the same area of the alleged negligence, but does not have to practice in the same specialty, and the statute allows one expert to certify a lawsuit against multiple doctors.
In a recent case, Mr. Montoya represented a Denver neurosurgeon who was sued along with multiple other health care professionals. From the outset, Mr. Montoya argued the claim had no merit against the neurosurgeon, but the plaintiff’s attorney refused to dismiss the physician. Mr. Montoya asked whether the expert consulted for the certificate of merit was a neurosurgeon, but the attorney declined to disclose that information, he said.
The case progressed and Mr. Montoya eventually asked the judge to review the certificate of merit. By law, a judge can confidentially review the certificate of merit and decide whether it aligns with the state statute, but without disclosing the expert’s identity to the defense. The judge ruled the certificate appeared to conform with state law, and the case continued.
A year later, as both sides were getting ready to disclose their experts who would testify, Mr. Montoya again argued the neurosurgeon should be dropped from the suit. This time, he warned if the claim continued against the neurosurgeon, the defense would be filing a motion for summary judgment and pursuing attorney fees and costs. Colorado law allows for such fees if the filing or pursuit of an action is frivolous.
“Boom, my client was dismissed,” Mr. Montoya said. “This is a year later, after multiple conferences among the attorneys, multiple pleadings filed, expert witnesses retained to review the care, discovery exchanged, and records obtained. If we had [a stronger] certificate of review statute, it would have been a different ballgame. It’s never going to get a year down the road.”
In New York, physician defendants have experienced similar woes. The state’s law requires plaintiffs’ attorneys to certify that they consulted with a physician prior to filing the claim, and that they believe based on that discussion, there’s a reasonable basis for the claim to move forward. Attorneys are not required to disclose the expert’s identity.
The law also allows “an out,” explained Morris Auster, JD, senior vice president and chief legislative counsel for the Medical Society of the State of New York. If the attorney made three separate attempts to obtain a consultation, and all three experts would not agree to the consultation, the lawsuit can be filed anyway, he said.
“From our standpoint, it’s important to have an affidavit of merit requirement; it’s better than not having it,” Mr. Auster said. “But its effectiveness in providing control over the filing of lawsuits in New York has never been as strong as it could’ve been.”
Mr. Auster notes that New York has some of the highest liability costs in the country in addition to doctors paying some of the steepest medical liability insurance premiums.
“This really affects a lot of physicians and it’s driving physicians into employment arrangements, so they don’t have to deal with it on their own,” he said. “We support a number of measures to address these significantly high costs, and stronger certificate of merit requirements would certainly be one of those advocacy goals.”
Why are anonymous experts allowed?
Certificates of merit that shield the identity of consultants encourage a greater pool of physicians willing to review cases, said J. Matthew Dudley, JD, president of the Illinois Trial Lawyers Association. When the requirements first went into effect in Illinois, there was significant animosity among physicians toward doctors who testified in medical malpractice cases for patients, Mr. Dudley explained.
“Sometimes they would be ostracized from their professional societies, or it would hurt a referral relationship.” he said. “Over time, that animosity has lessened, but there was a concern that if the identity of physicians in certificates of merit weren’t protected, then doctors would not look at cases for patients.”
This would result in additional barriers for patients and their attorneys in pursuing their legal rights, Mr. Dudley said. He said Illinois’ certificate of merit statute is successful in fulfilling its intended purpose, and he has not seen any statistical evidence to suggest otherwise.
“It has proven effective at decreasing filings in medical malpractice and effectively screening medical malpractice cases,” he said. “Certificates of merit help to decrease filings by firms that aren’t that experienced in dealing with those kinds of cases.”
Kentucky is another state that does not require attorneys to identity the experts consulted for certificates of merit. Malpractice defense attorney Andrew DeSimone, JD, who practices in Kentucky, said this isn’t a problem since attorneys eventually must disclose the expert witnesses who will testify at trial.
“Knowing the name behind the certificate of merit is not that pertinent,” Mr. DeSimone said. “Physicians and their attorneys will ultimately have the chance to question and evaluate the expert witnesses used at trial. The certificate of merit is designed to weed out totally frivolous cases that do not have expert support. It’s not designed to be a trial on the merits.”
The belief that plaintiffs’ attorneys frequently bring weak cases and use unqualified experts to certify claims is not realistic or logical, added Sean Domnick, JD, a Florida medical malpractice attorney and vice president for the American Association for Justice. Medical malpractice cases are extremely challenging for plaintiffs – and they’re expensive, Mr. Domnick said.
“We can’t afford to take bad cases,” he said. “For me to take on a medical malpractice case, it’s not unusual for me to spend well over $100,000. Remember, if we lose, I don’t get that money back and I don’t get paid. Why in the world would a plaintiff take on that type of a burden for a case they didn’t believe in? The logic escapes me.”
In Florida, where Mr. Domnick practices, plaintiffs’ attorneys must send their certificates of merit to the defense with the expert identified. Domnick believes the requirement is a hindrance.
“It creates a delay that is unnecessary in a system that is already designed to wear our clients down,” he said. “It’s just another component that makes it harder on them.”
Hidden experts may insulate plaintiffs’ attorneys from liability
Dr. Sullivan, the Illinois emergency physician, was ultimately dismissed from the multiparty lawsuit, but not for roughly 18 months. After the dismissal, he fought back. He sued the plaintiff’s law firm for malicious prosecution, negligence in hiring, and relying on the opinion of an expert who was unqualified to render an opinion against an emergency physician.
The law firm, however, argued that it was immune from liability because it reasonably relied on the expert’s opinion as required by Illinois law. A trial court agreed with the plaintiffs’ firm. The judge denied Dr. Sullivan’s request to identify the expert, ruling there was no finding that the affidavit was untrue or made without reasonable cause. Dr. Sullivan appealed, and the appellate court upheld the trial’s court decision.
“As happened with my case, law firms can use the affidavit as a defense against countersuits or motions for sanctions,” Dr. Sullivan said. “Although the certificate of merit is intended to prevent attorneys from filing frivolous cases, it can also have the opposite effect of helping to insulate plaintiff attorneys from liability for filing a frivolous lawsuit.”
In Colorado, complaints about the state’s certificate of merit statute have gone before the Colorado Supreme Court. In one case, a lower court ruled that a certificate of merit was deficient because the consultants were not chiropractors. In another case, a nurse defendant argued the claim’s certificate of review was insufficient because the consulting expert was a physician.
In both instances, Colorado judges held the state’s statute does not require consultants to be in the same profession or the same specialty as the health professional defendant.
In New York, meanwhile, Mr. Auster said several bills to strengthen the state’s certificate of merit requirements have failed in recent years.
“It’s hard to say whether it will improve anytime soon,” he said. “The trial lawyers are a very powerful advocacy force in the state, and they tend to oppose even the slightest of changes in civil liability. [In addition], some of these issues have been put on a lower tier because of trying to manage the pandemic.”
Ultimately, Dr. Sullivan said that courts and legislatures need to strongly consider the ethics of allowing anonymous experts to provide testimony against defendant physicians.
“I also think we need to consider how the notion of a secret expert comports with a defendant physician’s due process,” he said. “If an expert’s opinion is appropriate, why would there be a need to shroud one’s identity in a veil of secrecy?”
A version of this article first appeared on Medscape.com.
COVID booster mounts ‘brisk’ response in patients with cancer
New data shed light on the durability of antibody responses to SARS-CoV-2 vaccines and the impact of booster doses for patients with cancer undergoing systemic therapy or who have received a stem cell transplant (SCT).
In a cross-sectional study of 453 such patients, anti–SARS-CoV-2 spike protein receptor binding domain (anti-RBD) antibodies peaked 1 month after the second dose of an mRNA vaccine and remained stable over the next 6 months.
Notably, compared with the primary vaccine course, patients experienced a 20-fold increase in anti-RBD antibodies after the third vaccine dose, “indicative of a brisk anamnestic response from memory B cells,” Qamar Khan, MD, a medical oncologist at the University of Kansas Medical Center, Kansas City, and colleagues report.
The study appeared online in JAMA Oncology.
Given the risk of poor outcomes among patients with cancer and recipients of SCTs who get COVID, Dr. Khan and colleagues wanted to understand the durability of the antibody response to COVID vaccines in this population.
Among the 453 patients enrolled in the study, 70% had solid tumors and 30% had hematologic malignancies. Just over 40% were receiving chemotherapy, 16% were receiving immunotherapy, 14% were receiving a targeted oral agent, 5% were receiving chemoimmunotherapy, and 25% had received an SCT.
Regarding vaccine type, 61% received the Pfizer-BioNTech mRNA vaccine, 36% received the Moderna mRNA vaccine, and 4% got the Janssen/Johnson & Johnson vaccine. The mean age of the cohort was 60.4 years; 56% were women.
Prior to vaccination, the geometric mean titer (GMT) of anti-RBD antibodies for all patients was 1.7; it increased to 18.65 2 weeks after the first dose.
At 1 month after the second mRNA dose (or 2 months after the Johnson & Johnson vaccine), GMTs of anti-RBD antibodies reached 470.38 and then decreased to 425.8 at 3 months after the second dose (or 4 months after the Johnson & Johnson vaccine). Patients who were male, older than 65 years, and who had been diagnosed with a hematologic malignant tumor were more likely to have lower anti-RBD GMT 3 months after the second vaccine dose.
GMTs subsequently increased to 447.23 6 months after the second dose (7 months for Johnson & Johnson).
One month after the third dose, GMTs of anti-RBD antibodies rose to 9,224.85 – more than 20 times the previous GMT value.
According to the investigators, roughly 80% of these patients remained above the threshold of an anti-RBD level of 100 U/mL or higher at 6 months.
“While still an arbitrary cutoff, an anti-RBD level of 100 U/mL or higher has been associated with protection and has been used to evaluate the effectiveness of a third dose of an mRNA vaccine in a randomized clinical trial of patients who received a solid organ transplant,” Dr. Khan and colleagues write.
“Although more data are needed to confirm this level as protective, if established, anti-RBD can potentially be used to prioritize additional vaccine doses, especially in regions of the world with limited vaccine resources,” the authors conclude.
The study was supported in part by the University of Kansas Cancer Center and the Investigator Initiated Steering Committee, by a grant from the National Institute of General Medical Sciences, and a University of Kansas Cancer Center Support Grant from the National Cancer Institute. Dr. Khan reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
New data shed light on the durability of antibody responses to SARS-CoV-2 vaccines and the impact of booster doses for patients with cancer undergoing systemic therapy or who have received a stem cell transplant (SCT).
In a cross-sectional study of 453 such patients, anti–SARS-CoV-2 spike protein receptor binding domain (anti-RBD) antibodies peaked 1 month after the second dose of an mRNA vaccine and remained stable over the next 6 months.
Notably, compared with the primary vaccine course, patients experienced a 20-fold increase in anti-RBD antibodies after the third vaccine dose, “indicative of a brisk anamnestic response from memory B cells,” Qamar Khan, MD, a medical oncologist at the University of Kansas Medical Center, Kansas City, and colleagues report.
The study appeared online in JAMA Oncology.
Given the risk of poor outcomes among patients with cancer and recipients of SCTs who get COVID, Dr. Khan and colleagues wanted to understand the durability of the antibody response to COVID vaccines in this population.
Among the 453 patients enrolled in the study, 70% had solid tumors and 30% had hematologic malignancies. Just over 40% were receiving chemotherapy, 16% were receiving immunotherapy, 14% were receiving a targeted oral agent, 5% were receiving chemoimmunotherapy, and 25% had received an SCT.
Regarding vaccine type, 61% received the Pfizer-BioNTech mRNA vaccine, 36% received the Moderna mRNA vaccine, and 4% got the Janssen/Johnson & Johnson vaccine. The mean age of the cohort was 60.4 years; 56% were women.
Prior to vaccination, the geometric mean titer (GMT) of anti-RBD antibodies for all patients was 1.7; it increased to 18.65 2 weeks after the first dose.
At 1 month after the second mRNA dose (or 2 months after the Johnson & Johnson vaccine), GMTs of anti-RBD antibodies reached 470.38 and then decreased to 425.8 at 3 months after the second dose (or 4 months after the Johnson & Johnson vaccine). Patients who were male, older than 65 years, and who had been diagnosed with a hematologic malignant tumor were more likely to have lower anti-RBD GMT 3 months after the second vaccine dose.
GMTs subsequently increased to 447.23 6 months after the second dose (7 months for Johnson & Johnson).
One month after the third dose, GMTs of anti-RBD antibodies rose to 9,224.85 – more than 20 times the previous GMT value.
According to the investigators, roughly 80% of these patients remained above the threshold of an anti-RBD level of 100 U/mL or higher at 6 months.
“While still an arbitrary cutoff, an anti-RBD level of 100 U/mL or higher has been associated with protection and has been used to evaluate the effectiveness of a third dose of an mRNA vaccine in a randomized clinical trial of patients who received a solid organ transplant,” Dr. Khan and colleagues write.
“Although more data are needed to confirm this level as protective, if established, anti-RBD can potentially be used to prioritize additional vaccine doses, especially in regions of the world with limited vaccine resources,” the authors conclude.
The study was supported in part by the University of Kansas Cancer Center and the Investigator Initiated Steering Committee, by a grant from the National Institute of General Medical Sciences, and a University of Kansas Cancer Center Support Grant from the National Cancer Institute. Dr. Khan reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
New data shed light on the durability of antibody responses to SARS-CoV-2 vaccines and the impact of booster doses for patients with cancer undergoing systemic therapy or who have received a stem cell transplant (SCT).
In a cross-sectional study of 453 such patients, anti–SARS-CoV-2 spike protein receptor binding domain (anti-RBD) antibodies peaked 1 month after the second dose of an mRNA vaccine and remained stable over the next 6 months.
Notably, compared with the primary vaccine course, patients experienced a 20-fold increase in anti-RBD antibodies after the third vaccine dose, “indicative of a brisk anamnestic response from memory B cells,” Qamar Khan, MD, a medical oncologist at the University of Kansas Medical Center, Kansas City, and colleagues report.
The study appeared online in JAMA Oncology.
Given the risk of poor outcomes among patients with cancer and recipients of SCTs who get COVID, Dr. Khan and colleagues wanted to understand the durability of the antibody response to COVID vaccines in this population.
Among the 453 patients enrolled in the study, 70% had solid tumors and 30% had hematologic malignancies. Just over 40% were receiving chemotherapy, 16% were receiving immunotherapy, 14% were receiving a targeted oral agent, 5% were receiving chemoimmunotherapy, and 25% had received an SCT.
Regarding vaccine type, 61% received the Pfizer-BioNTech mRNA vaccine, 36% received the Moderna mRNA vaccine, and 4% got the Janssen/Johnson & Johnson vaccine. The mean age of the cohort was 60.4 years; 56% were women.
Prior to vaccination, the geometric mean titer (GMT) of anti-RBD antibodies for all patients was 1.7; it increased to 18.65 2 weeks after the first dose.
At 1 month after the second mRNA dose (or 2 months after the Johnson & Johnson vaccine), GMTs of anti-RBD antibodies reached 470.38 and then decreased to 425.8 at 3 months after the second dose (or 4 months after the Johnson & Johnson vaccine). Patients who were male, older than 65 years, and who had been diagnosed with a hematologic malignant tumor were more likely to have lower anti-RBD GMT 3 months after the second vaccine dose.
GMTs subsequently increased to 447.23 6 months after the second dose (7 months for Johnson & Johnson).
One month after the third dose, GMTs of anti-RBD antibodies rose to 9,224.85 – more than 20 times the previous GMT value.
According to the investigators, roughly 80% of these patients remained above the threshold of an anti-RBD level of 100 U/mL or higher at 6 months.
“While still an arbitrary cutoff, an anti-RBD level of 100 U/mL or higher has been associated with protection and has been used to evaluate the effectiveness of a third dose of an mRNA vaccine in a randomized clinical trial of patients who received a solid organ transplant,” Dr. Khan and colleagues write.
“Although more data are needed to confirm this level as protective, if established, anti-RBD can potentially be used to prioritize additional vaccine doses, especially in regions of the world with limited vaccine resources,” the authors conclude.
The study was supported in part by the University of Kansas Cancer Center and the Investigator Initiated Steering Committee, by a grant from the National Institute of General Medical Sciences, and a University of Kansas Cancer Center Support Grant from the National Cancer Institute. Dr. Khan reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM JAMA ONCOLOGY