User login
ID Practitioner is an independent news source that provides infectious disease specialists with timely and relevant news and commentary about clinical developments and the impact of health care policy on the infectious disease specialist’s practice. Specialty focus topics include antimicrobial resistance, emerging infections, global ID, hepatitis, HIV, hospital-acquired infections, immunizations and vaccines, influenza, mycoses, pediatric infections, and STIs. Infectious Diseases News is owned by Frontline Medical Communications.
sofosbuvir
ritonavir with dasabuvir
discount
support path
program
ritonavir
greedy
ledipasvir
assistance
viekira pak
vpak
advocacy
needy
protest
abbvie
paritaprevir
ombitasvir
direct-acting antivirals
dasabuvir
gilead
fake-ovir
support
v pak
oasis
harvoni
section[contains(@class, 'footer-nav-section-wrapper')]
div[contains(@class, 'pane-pub-article-idp')]
div[contains(@class, 'pane-medstat-latest-articles-articles-section')]
div[contains(@class, 'pane-pub-home-idp')]
div[contains(@class, 'pane-pub-topic-idp')]
COVID-19 vaccines: Safe for immunocompromised patients?
Coronavirus vaccines have become a reality, as they are now being approved and authorized for use in a growing number of countries including the United States. The U.S. Food and Drug Administration has just issued emergency authorization for the use of the COVID-19 vaccine produced by Pfizer and BioNTech. Close behind is the vaccine developed by Moderna, which has also applied to the FDA for emergency authorization.
The efficacy of a two-dose administration of the vaccine has been pegged at 95.0%, and the FDA has said that the 95% credible interval for the vaccine efficacy was 90.3%-97.6%. But as with many initial clinical trials, whether for drugs or vaccines, not all populations were represented in the trial cohort, including individuals who are immunocompromised. At the current time, it is largely unknown how safe or effective the vaccine may be in this large population, many of whom are at high risk for serious COVID-19 complications.
At a special session held during the recent annual meeting of the American Society of Hematology, Anthony Fauci, MD, the nation’s leading infectious disease expert, said that individuals with compromised immune systems, whether because of chemotherapy or a bone marrow transplant, should plan to be vaccinated when the opportunity arises.
In response to a question from ASH President Stephanie J. Lee, MD, of the Fred Hutchinson Cancer Center, Seattle, Dr. Fauci emphasized that, despite being excluded from clinical trials, this population should get vaccinated. “I think we should recommend that they get vaccinated,” he said. “I mean, it is clear that, if you are on immunosuppressive agents, history tells us that you’re not going to have as robust a response as if you had an intact immune system that was not being compromised. But some degree of immunity is better than no degree of immunity.”
That does seem to be the consensus among experts who spoke in interviews: that as long as these are not live attenuated vaccines, they hold no specific risk to an immunocompromised patient, other than any factors specific to the individual that could be a contraindication.
“Patients, family members, friends, and work contacts should be encouraged to receive the vaccine,” said William Stohl, MD, PhD, chief of the division of rheumatology at the University of Southern California, Los Angeles. “Clinicians should advise patients to obtain the vaccine sooner rather than later.”
Kevin C. Wang, MD, PhD, of the department of dermatology at Stanford (Calif.) University, agreed. “I am 100% with Dr. Fauci. Everyone should get the vaccine, even if it may not be as effective,” he said. “I would treat it exactly like the flu vaccines that we recommend folks get every year.”
Dr. Wang noted that he couldn’t think of any contraindications unless the immunosuppressed patients have a history of severe allergic reactions to prior vaccinations. “But I would even say patients with history of cancer, upon recommendation of their oncologists, are likely to be suitable candidates for the vaccine,” he added. “I would say clinicians should approach counseling the same way they counsel patients for the flu vaccine, and as far as I know, there are no concerns for systemic drugs commonly used in dermatology patients.”
However, guidance has not yet been issued from either the FDA or the Centers for Disease Control and Prevention regarding the use of the vaccine in immunocompromised individuals. Given the lack of data, the FDA has said that “it will be something that providers will need to consider on an individual basis,” and that individuals should consult with physicians to weigh the potential benefits and potential risks.
The CDC’s Advisory Committee on Immunization Practices has said that clinicians need more guidance on whether to use the vaccine in pregnant or breastfeeding women, the immunocompromised, or those who have a history of allergies. The CDC itself has not yet released its formal guidance on vaccine use.
COVID-19 vaccines
Vaccines typically require years of research and testing before reaching the clinic, but this year researchers embarked on a global effort to develop safe and effective coronavirus vaccines in record time. Both the Pfizer/BioNTech and Moderna vaccines have only a few months of phase 3 clinical trial data, so much remains unknown about them, including their duration of effect and any long-term safety signals. In addition to excluding immunocompromised individuals, the clinical trials did not include children or pregnant women, so data are lacking for several population subgroups.
But these will not be the only vaccines available, as the pipeline is already becoming crowded. U.S. clinical trial data from a vaccine jointly being developed by Oxford-AstraZeneca, could potentially be ready, along with a request for FDA emergency use authorization, by late January 2021.
In addition, China and Russia have released vaccines, and there are currently 61 vaccines being investigated in clinical trials and at least 85 preclinical products under active investigation.
The vaccine candidates are using both conventional and novel mechanisms of action to elicit an immune response in patients. Conventional methods include attenuated inactivated (killed) virus and recombinant viral protein vaccines to develop immunity. Novel approaches include replication-deficient, adenovirus vector-based vaccines that contain the viral protein, and mRNA-based vaccines, such as the Pfizer and Moderna vaccines, that encode for a SARS-CoV-2 spike protein.
“The special vaccine concern for immunocompromised individuals is introduction of a live virus,” Dr. Stohl said. “Neither the Moderna nor Pfizer vaccines are live viruses, so there should be no special contraindication for such individuals.”
Live vaccine should be avoided in immunocompromised patients, and currently, live SARS-CoV-2 vaccines are only being developed in India and Turkey.
It is not unusual for vaccine trials to begin with cohorts that exclude participants with various health conditions, including those who are immunocompromised. These groups are generally then evaluated in phase 4 trials, or postmarketing surveillance. While the precise number of immunosuppressed adults in the United States is not known, the numbers are believed to be rising because of increased life expectancy among immunosuppressed adults as a result of advances in treatment and new and wider indications for therapies that can affect the immune system.
According to data from the 2013 National Health Interview Survey, an estimated 2.7% of U.S. adults are immunosuppressed. This population covers a broad array of health conditions and medical specialties; people living with inflammatory or autoimmune conditions, such as inflammatory rheumatic diseases (rheumatoid arthritis, axial spondyloarthritis, lupus); inflammatory bowel disease (Crohn’s disease and ulcerative colitis); psoriasis; multiple sclerosis; organ transplant recipients; patients undergoing chemotherapy; and life-long immunosuppression attributable to HIV infection.
As the vaccines begin to roll out and become available, how should clinicians advise their patients, in the absence of any clinical trial data?
Risk vs. benefit
Gilaad Kaplan, MD, MPH, a gastroenterologist and professor of medicine at the University of Calgary (Alta.), noted that the inflammatory bowel disease (IBD) community has dealt with tremendous anxiety during the pandemic because many are immunocompromised because of the medications they use to treat their disease.
“For example, many patients with IBD are on biologics like anti-TNF [tumor necrosis factor] therapies, which are also used in other immune-mediated inflammatory diseases such as rheumatoid arthritis,” he said. “Understandably, individuals with IBD on immunosuppressive medications are concerned about the risk of severe complications due to COVID-19.”
The entire IBD community, along with the world, celebrated the announcement that multiple vaccines are protective against SARS-CoV-2, he noted. “Vaccines offer the potential to reduce the spread of COVID-19, allowing society to revert back to normalcy,” Dr. Kaplan said. “Moreover, for vulnerable populations, including those who are immunocompromised, vaccines offer the potential to directly protect them from the morbidity and mortality associated with COVID-19.”
That said, even though the news of vaccines are extremely promising, some cautions must be raised regarding their use in immunocompromised populations, such as persons with IBD. “The current trials, to my knowledge, did not include immunocompromised individuals and thus, we can only extrapolate from what we know from other trials of different vaccines,” he explained. “We know from prior vaccines studies that the immune response following vaccination is less robust in those who are immunocompromised as compared to a healthy control population.”
Dr. Kaplan also pointed to recent reports of allergic reactions that have been reported in healthy individuals. “We don’t know whether side effects, like allergic reactions, may be different in unstudied populations,” he said. “Thus, the medical and scientific community should prioritize clinical studies of safety and effectiveness of COVID-19 vaccines in immunocompromised populations.”
So, what does this mean for an individual with an immune-mediated inflammatory disease like Crohn’s disease or ulcerative colitis who is immunocompromised? Dr. Kaplan explained that it is a balance between the potential harm of being infected with COVID-19 and the uncertainty of receiving a vaccine in an understudied population. For those who are highly susceptible to dying from COVID-19, such as an older adult with IBD, or someone who faces high exposure, such as a health care worker, the potential protection of the vaccine greatly outweighs the uncertainty.
“However, for individuals who are at otherwise lower risk – for example, young and able to work from home – then waiting a few extra months for postmarketing surveillance studies in immunocompromised populations may be a reasonable approach, as long as these individuals are taking great care to avoid infection,” he said.
No waiting needed
Joel M. Gelfand, MD, MSCE, professor of dermatology and epidemiology at the University of Pennsylvania, Philadelphia, feels that the newly approved vaccine should be safe for most of his patients.
“Patients with psoriatic disease should get the mRNA-based COVID-19 vaccine as soon as possible based on eligibility as determined by the CDC and local public health officials,” he said. “It is not a live vaccine, and therefore patients on biologics or other immune-modulating or immune-suppressing treatment can receive it.”
However, the impact of psoriasis treatment on immune response to the mRNA-based vaccines is not known. Dr. Gelfand noted that, extrapolating from the vaccine literature, there is some evidence that methotrexate reduces response to the influenza vaccine. “However, the clinical significance of this finding is not clear,” he said. “Since the mRNA vaccine needs to be taken twice, a few weeks apart, I do not recommend interrupting or delaying treatment for psoriatic disease while undergoing vaccination for COVID-19.”
Given the reports of allergic reactions, he added that it is advisable for patients with a history of life-threatening allergic reactions such as anaphylaxis or who have been advised to carry an epinephrine autoinjector, to talk with their health care provider to determine if COVID-19 vaccination is medically appropriate.
The National Psoriasis Foundation has issued guidance on COVID-19, explained Steven R. Feldman, MD, PhD, professor of dermatology, pathology, and social sciences & health policy at Wake Forest University, Winston-Salem, N.C., who is also a member of the committee that is working on those guidelines and keeping them up to date. “We are in the process of updating the guidelines with information on COVID vaccines,” he said.
He agreed that there are no contraindications for psoriasis patients to receive the vaccine, regardless of whether they are on immunosuppressive treatment, even though definitive data are lacking. “Fortunately, there’s a lot of good data coming out of Italy that patients with psoriasis on biologics do not appear to be at increased risk of getting COVID or of having worse outcomes from COVID,” he said.
Patients are going to ask about the vaccines, and when counseling them, clinicians should discuss the available data, the residual uncertainty, and patients’ concerns should be considered, Dr. Feldman explained. “There may be some concern that steroids and cyclosporine would reduce the effectiveness of vaccines, but there is no concern that any of the drugs would cause increased risk from nonlive vaccines.”
He added that there is evidence that “patients on biologics who receive nonlive vaccines do develop antibody responses and are immunized.”
Boosting efficacy
Even prior to making their announcement, the American College of Rheumatology had said that they would endorse the vaccine for all patients, explained rheumatologist Brett Smith, DO, from Blount Memorial Physicians Group and East Tennessee Children’s Hospital, Alcoa. “The vaccine is safe for all patients, but the problem may be that it’s not as effective,” he said. “But we don’t know that because it hasn’t been tested.”
With other vaccines, biologic medicines are held for 2 weeks before and afterwards, to get the best response. “But some patients don’t want to stop the medication,” Dr. Smith said. “They are afraid that their symptoms will return.”
As for counseling patients as to whether they should receive this vaccine, he explained that he typically doesn’t try to sway patients one way or another until they are really high risk. “When I counsel, it really depends on the individual situation. And for this vaccine, we have to be open to the fact that many people have already made up their mind.”
There are a lot of questions regarding the vaccine. One is the short time frame of development. “Vaccines typically take 6-10 years to come on the market, and this one is now available after a 3-month study,” Dr. Smith said. “Some have already decided that it’s too new for them.”
The process is also new, and patients need to understand that it doesn’t contain an active virus and “you can’t catch coronavirus from it.”
Dr. Smith also explained that, because the vaccine may be less effective in a person using biologic therapies, there is currently no information available on repeat vaccination. “These are all unanswered questions,” he said. “If the antibodies wane in a short time, can we be revaccinated and in what time frame? We just don’t know that yet.”
Marcelo Bonomi, MD, a medical oncologist from The Ohio State University Comprehensive Cancer Center, Columbus, explained that one way to ensure a more optimal response to the vaccine would be to wait until the patient has finished chemotherapy.* “The vaccine can be offered at that time, and in the meantime, they can take other steps to avoid infection,” he said. “If they are very immunosuppressed, it isn’t worth trying to give the vaccine.”
Cancer patients should be encouraged to stay as healthy as possible, and to wear masks and social distance. “It’s a comprehensive approach. Eat healthy, avoid alcohol and tobacco, and exercise. [These things] will help boost the immune system,” Dr. Bonomi said. “Family members should be encouraged to get vaccinated, which will help them avoid infection and exposing the patient.”
Jim Boonyaratanakornkit, MD, PhD, an infectious disease specialist who cares for cancer patients at the Fred Hutchinson Cancer Research Center, agreed. “Giving a vaccine right after a transplant is a futile endeavor,” he said. “We need to wait 6 months to have an immune response.”
He pointed out there may be a continuing higher number of cases, with high levels peaking in Washington in February and March. “Close friends and family should be vaccinated if possible,” he said, “which will help interrupt transmission.”
The vaccines are using new platforms that are totally different, and there is no clear data as to how long the antibodies will persist. “We know that they last for at least 4 months,” said Dr. Boonyaratanakornkit. “We don’t know what level of antibody will protect them from COVID-19 infection. Current studies are being conducted, but we don’t have that information for anyone yet.”
*Correction, 1/7/21: An earlier version of this article misattributed quotes from Dr. Marcelo Bonomi.
Coronavirus vaccines have become a reality, as they are now being approved and authorized for use in a growing number of countries including the United States. The U.S. Food and Drug Administration has just issued emergency authorization for the use of the COVID-19 vaccine produced by Pfizer and BioNTech. Close behind is the vaccine developed by Moderna, which has also applied to the FDA for emergency authorization.
The efficacy of a two-dose administration of the vaccine has been pegged at 95.0%, and the FDA has said that the 95% credible interval for the vaccine efficacy was 90.3%-97.6%. But as with many initial clinical trials, whether for drugs or vaccines, not all populations were represented in the trial cohort, including individuals who are immunocompromised. At the current time, it is largely unknown how safe or effective the vaccine may be in this large population, many of whom are at high risk for serious COVID-19 complications.
At a special session held during the recent annual meeting of the American Society of Hematology, Anthony Fauci, MD, the nation’s leading infectious disease expert, said that individuals with compromised immune systems, whether because of chemotherapy or a bone marrow transplant, should plan to be vaccinated when the opportunity arises.
In response to a question from ASH President Stephanie J. Lee, MD, of the Fred Hutchinson Cancer Center, Seattle, Dr. Fauci emphasized that, despite being excluded from clinical trials, this population should get vaccinated. “I think we should recommend that they get vaccinated,” he said. “I mean, it is clear that, if you are on immunosuppressive agents, history tells us that you’re not going to have as robust a response as if you had an intact immune system that was not being compromised. But some degree of immunity is better than no degree of immunity.”
That does seem to be the consensus among experts who spoke in interviews: that as long as these are not live attenuated vaccines, they hold no specific risk to an immunocompromised patient, other than any factors specific to the individual that could be a contraindication.
“Patients, family members, friends, and work contacts should be encouraged to receive the vaccine,” said William Stohl, MD, PhD, chief of the division of rheumatology at the University of Southern California, Los Angeles. “Clinicians should advise patients to obtain the vaccine sooner rather than later.”
Kevin C. Wang, MD, PhD, of the department of dermatology at Stanford (Calif.) University, agreed. “I am 100% with Dr. Fauci. Everyone should get the vaccine, even if it may not be as effective,” he said. “I would treat it exactly like the flu vaccines that we recommend folks get every year.”
Dr. Wang noted that he couldn’t think of any contraindications unless the immunosuppressed patients have a history of severe allergic reactions to prior vaccinations. “But I would even say patients with history of cancer, upon recommendation of their oncologists, are likely to be suitable candidates for the vaccine,” he added. “I would say clinicians should approach counseling the same way they counsel patients for the flu vaccine, and as far as I know, there are no concerns for systemic drugs commonly used in dermatology patients.”
However, guidance has not yet been issued from either the FDA or the Centers for Disease Control and Prevention regarding the use of the vaccine in immunocompromised individuals. Given the lack of data, the FDA has said that “it will be something that providers will need to consider on an individual basis,” and that individuals should consult with physicians to weigh the potential benefits and potential risks.
The CDC’s Advisory Committee on Immunization Practices has said that clinicians need more guidance on whether to use the vaccine in pregnant or breastfeeding women, the immunocompromised, or those who have a history of allergies. The CDC itself has not yet released its formal guidance on vaccine use.
COVID-19 vaccines
Vaccines typically require years of research and testing before reaching the clinic, but this year researchers embarked on a global effort to develop safe and effective coronavirus vaccines in record time. Both the Pfizer/BioNTech and Moderna vaccines have only a few months of phase 3 clinical trial data, so much remains unknown about them, including their duration of effect and any long-term safety signals. In addition to excluding immunocompromised individuals, the clinical trials did not include children or pregnant women, so data are lacking for several population subgroups.
But these will not be the only vaccines available, as the pipeline is already becoming crowded. U.S. clinical trial data from a vaccine jointly being developed by Oxford-AstraZeneca, could potentially be ready, along with a request for FDA emergency use authorization, by late January 2021.
In addition, China and Russia have released vaccines, and there are currently 61 vaccines being investigated in clinical trials and at least 85 preclinical products under active investigation.
The vaccine candidates are using both conventional and novel mechanisms of action to elicit an immune response in patients. Conventional methods include attenuated inactivated (killed) virus and recombinant viral protein vaccines to develop immunity. Novel approaches include replication-deficient, adenovirus vector-based vaccines that contain the viral protein, and mRNA-based vaccines, such as the Pfizer and Moderna vaccines, that encode for a SARS-CoV-2 spike protein.
“The special vaccine concern for immunocompromised individuals is introduction of a live virus,” Dr. Stohl said. “Neither the Moderna nor Pfizer vaccines are live viruses, so there should be no special contraindication for such individuals.”
Live vaccine should be avoided in immunocompromised patients, and currently, live SARS-CoV-2 vaccines are only being developed in India and Turkey.
It is not unusual for vaccine trials to begin with cohorts that exclude participants with various health conditions, including those who are immunocompromised. These groups are generally then evaluated in phase 4 trials, or postmarketing surveillance. While the precise number of immunosuppressed adults in the United States is not known, the numbers are believed to be rising because of increased life expectancy among immunosuppressed adults as a result of advances in treatment and new and wider indications for therapies that can affect the immune system.
According to data from the 2013 National Health Interview Survey, an estimated 2.7% of U.S. adults are immunosuppressed. This population covers a broad array of health conditions and medical specialties; people living with inflammatory or autoimmune conditions, such as inflammatory rheumatic diseases (rheumatoid arthritis, axial spondyloarthritis, lupus); inflammatory bowel disease (Crohn’s disease and ulcerative colitis); psoriasis; multiple sclerosis; organ transplant recipients; patients undergoing chemotherapy; and life-long immunosuppression attributable to HIV infection.
As the vaccines begin to roll out and become available, how should clinicians advise their patients, in the absence of any clinical trial data?
Risk vs. benefit
Gilaad Kaplan, MD, MPH, a gastroenterologist and professor of medicine at the University of Calgary (Alta.), noted that the inflammatory bowel disease (IBD) community has dealt with tremendous anxiety during the pandemic because many are immunocompromised because of the medications they use to treat their disease.
“For example, many patients with IBD are on biologics like anti-TNF [tumor necrosis factor] therapies, which are also used in other immune-mediated inflammatory diseases such as rheumatoid arthritis,” he said. “Understandably, individuals with IBD on immunosuppressive medications are concerned about the risk of severe complications due to COVID-19.”
The entire IBD community, along with the world, celebrated the announcement that multiple vaccines are protective against SARS-CoV-2, he noted. “Vaccines offer the potential to reduce the spread of COVID-19, allowing society to revert back to normalcy,” Dr. Kaplan said. “Moreover, for vulnerable populations, including those who are immunocompromised, vaccines offer the potential to directly protect them from the morbidity and mortality associated with COVID-19.”
That said, even though the news of vaccines are extremely promising, some cautions must be raised regarding their use in immunocompromised populations, such as persons with IBD. “The current trials, to my knowledge, did not include immunocompromised individuals and thus, we can only extrapolate from what we know from other trials of different vaccines,” he explained. “We know from prior vaccines studies that the immune response following vaccination is less robust in those who are immunocompromised as compared to a healthy control population.”
Dr. Kaplan also pointed to recent reports of allergic reactions that have been reported in healthy individuals. “We don’t know whether side effects, like allergic reactions, may be different in unstudied populations,” he said. “Thus, the medical and scientific community should prioritize clinical studies of safety and effectiveness of COVID-19 vaccines in immunocompromised populations.”
So, what does this mean for an individual with an immune-mediated inflammatory disease like Crohn’s disease or ulcerative colitis who is immunocompromised? Dr. Kaplan explained that it is a balance between the potential harm of being infected with COVID-19 and the uncertainty of receiving a vaccine in an understudied population. For those who are highly susceptible to dying from COVID-19, such as an older adult with IBD, or someone who faces high exposure, such as a health care worker, the potential protection of the vaccine greatly outweighs the uncertainty.
“However, for individuals who are at otherwise lower risk – for example, young and able to work from home – then waiting a few extra months for postmarketing surveillance studies in immunocompromised populations may be a reasonable approach, as long as these individuals are taking great care to avoid infection,” he said.
No waiting needed
Joel M. Gelfand, MD, MSCE, professor of dermatology and epidemiology at the University of Pennsylvania, Philadelphia, feels that the newly approved vaccine should be safe for most of his patients.
“Patients with psoriatic disease should get the mRNA-based COVID-19 vaccine as soon as possible based on eligibility as determined by the CDC and local public health officials,” he said. “It is not a live vaccine, and therefore patients on biologics or other immune-modulating or immune-suppressing treatment can receive it.”
However, the impact of psoriasis treatment on immune response to the mRNA-based vaccines is not known. Dr. Gelfand noted that, extrapolating from the vaccine literature, there is some evidence that methotrexate reduces response to the influenza vaccine. “However, the clinical significance of this finding is not clear,” he said. “Since the mRNA vaccine needs to be taken twice, a few weeks apart, I do not recommend interrupting or delaying treatment for psoriatic disease while undergoing vaccination for COVID-19.”
Given the reports of allergic reactions, he added that it is advisable for patients with a history of life-threatening allergic reactions such as anaphylaxis or who have been advised to carry an epinephrine autoinjector, to talk with their health care provider to determine if COVID-19 vaccination is medically appropriate.
The National Psoriasis Foundation has issued guidance on COVID-19, explained Steven R. Feldman, MD, PhD, professor of dermatology, pathology, and social sciences & health policy at Wake Forest University, Winston-Salem, N.C., who is also a member of the committee that is working on those guidelines and keeping them up to date. “We are in the process of updating the guidelines with information on COVID vaccines,” he said.
He agreed that there are no contraindications for psoriasis patients to receive the vaccine, regardless of whether they are on immunosuppressive treatment, even though definitive data are lacking. “Fortunately, there’s a lot of good data coming out of Italy that patients with psoriasis on biologics do not appear to be at increased risk of getting COVID or of having worse outcomes from COVID,” he said.
Patients are going to ask about the vaccines, and when counseling them, clinicians should discuss the available data, the residual uncertainty, and patients’ concerns should be considered, Dr. Feldman explained. “There may be some concern that steroids and cyclosporine would reduce the effectiveness of vaccines, but there is no concern that any of the drugs would cause increased risk from nonlive vaccines.”
He added that there is evidence that “patients on biologics who receive nonlive vaccines do develop antibody responses and are immunized.”
Boosting efficacy
Even prior to making their announcement, the American College of Rheumatology had said that they would endorse the vaccine for all patients, explained rheumatologist Brett Smith, DO, from Blount Memorial Physicians Group and East Tennessee Children’s Hospital, Alcoa. “The vaccine is safe for all patients, but the problem may be that it’s not as effective,” he said. “But we don’t know that because it hasn’t been tested.”
With other vaccines, biologic medicines are held for 2 weeks before and afterwards, to get the best response. “But some patients don’t want to stop the medication,” Dr. Smith said. “They are afraid that their symptoms will return.”
As for counseling patients as to whether they should receive this vaccine, he explained that he typically doesn’t try to sway patients one way or another until they are really high risk. “When I counsel, it really depends on the individual situation. And for this vaccine, we have to be open to the fact that many people have already made up their mind.”
There are a lot of questions regarding the vaccine. One is the short time frame of development. “Vaccines typically take 6-10 years to come on the market, and this one is now available after a 3-month study,” Dr. Smith said. “Some have already decided that it’s too new for them.”
The process is also new, and patients need to understand that it doesn’t contain an active virus and “you can’t catch coronavirus from it.”
Dr. Smith also explained that, because the vaccine may be less effective in a person using biologic therapies, there is currently no information available on repeat vaccination. “These are all unanswered questions,” he said. “If the antibodies wane in a short time, can we be revaccinated and in what time frame? We just don’t know that yet.”
Marcelo Bonomi, MD, a medical oncologist from The Ohio State University Comprehensive Cancer Center, Columbus, explained that one way to ensure a more optimal response to the vaccine would be to wait until the patient has finished chemotherapy.* “The vaccine can be offered at that time, and in the meantime, they can take other steps to avoid infection,” he said. “If they are very immunosuppressed, it isn’t worth trying to give the vaccine.”
Cancer patients should be encouraged to stay as healthy as possible, and to wear masks and social distance. “It’s a comprehensive approach. Eat healthy, avoid alcohol and tobacco, and exercise. [These things] will help boost the immune system,” Dr. Bonomi said. “Family members should be encouraged to get vaccinated, which will help them avoid infection and exposing the patient.”
Jim Boonyaratanakornkit, MD, PhD, an infectious disease specialist who cares for cancer patients at the Fred Hutchinson Cancer Research Center, agreed. “Giving a vaccine right after a transplant is a futile endeavor,” he said. “We need to wait 6 months to have an immune response.”
He pointed out there may be a continuing higher number of cases, with high levels peaking in Washington in February and March. “Close friends and family should be vaccinated if possible,” he said, “which will help interrupt transmission.”
The vaccines are using new platforms that are totally different, and there is no clear data as to how long the antibodies will persist. “We know that they last for at least 4 months,” said Dr. Boonyaratanakornkit. “We don’t know what level of antibody will protect them from COVID-19 infection. Current studies are being conducted, but we don’t have that information for anyone yet.”
*Correction, 1/7/21: An earlier version of this article misattributed quotes from Dr. Marcelo Bonomi.
Coronavirus vaccines have become a reality, as they are now being approved and authorized for use in a growing number of countries including the United States. The U.S. Food and Drug Administration has just issued emergency authorization for the use of the COVID-19 vaccine produced by Pfizer and BioNTech. Close behind is the vaccine developed by Moderna, which has also applied to the FDA for emergency authorization.
The efficacy of a two-dose administration of the vaccine has been pegged at 95.0%, and the FDA has said that the 95% credible interval for the vaccine efficacy was 90.3%-97.6%. But as with many initial clinical trials, whether for drugs or vaccines, not all populations were represented in the trial cohort, including individuals who are immunocompromised. At the current time, it is largely unknown how safe or effective the vaccine may be in this large population, many of whom are at high risk for serious COVID-19 complications.
At a special session held during the recent annual meeting of the American Society of Hematology, Anthony Fauci, MD, the nation’s leading infectious disease expert, said that individuals with compromised immune systems, whether because of chemotherapy or a bone marrow transplant, should plan to be vaccinated when the opportunity arises.
In response to a question from ASH President Stephanie J. Lee, MD, of the Fred Hutchinson Cancer Center, Seattle, Dr. Fauci emphasized that, despite being excluded from clinical trials, this population should get vaccinated. “I think we should recommend that they get vaccinated,” he said. “I mean, it is clear that, if you are on immunosuppressive agents, history tells us that you’re not going to have as robust a response as if you had an intact immune system that was not being compromised. But some degree of immunity is better than no degree of immunity.”
That does seem to be the consensus among experts who spoke in interviews: that as long as these are not live attenuated vaccines, they hold no specific risk to an immunocompromised patient, other than any factors specific to the individual that could be a contraindication.
“Patients, family members, friends, and work contacts should be encouraged to receive the vaccine,” said William Stohl, MD, PhD, chief of the division of rheumatology at the University of Southern California, Los Angeles. “Clinicians should advise patients to obtain the vaccine sooner rather than later.”
Kevin C. Wang, MD, PhD, of the department of dermatology at Stanford (Calif.) University, agreed. “I am 100% with Dr. Fauci. Everyone should get the vaccine, even if it may not be as effective,” he said. “I would treat it exactly like the flu vaccines that we recommend folks get every year.”
Dr. Wang noted that he couldn’t think of any contraindications unless the immunosuppressed patients have a history of severe allergic reactions to prior vaccinations. “But I would even say patients with history of cancer, upon recommendation of their oncologists, are likely to be suitable candidates for the vaccine,” he added. “I would say clinicians should approach counseling the same way they counsel patients for the flu vaccine, and as far as I know, there are no concerns for systemic drugs commonly used in dermatology patients.”
However, guidance has not yet been issued from either the FDA or the Centers for Disease Control and Prevention regarding the use of the vaccine in immunocompromised individuals. Given the lack of data, the FDA has said that “it will be something that providers will need to consider on an individual basis,” and that individuals should consult with physicians to weigh the potential benefits and potential risks.
The CDC’s Advisory Committee on Immunization Practices has said that clinicians need more guidance on whether to use the vaccine in pregnant or breastfeeding women, the immunocompromised, or those who have a history of allergies. The CDC itself has not yet released its formal guidance on vaccine use.
COVID-19 vaccines
Vaccines typically require years of research and testing before reaching the clinic, but this year researchers embarked on a global effort to develop safe and effective coronavirus vaccines in record time. Both the Pfizer/BioNTech and Moderna vaccines have only a few months of phase 3 clinical trial data, so much remains unknown about them, including their duration of effect and any long-term safety signals. In addition to excluding immunocompromised individuals, the clinical trials did not include children or pregnant women, so data are lacking for several population subgroups.
But these will not be the only vaccines available, as the pipeline is already becoming crowded. U.S. clinical trial data from a vaccine jointly being developed by Oxford-AstraZeneca, could potentially be ready, along with a request for FDA emergency use authorization, by late January 2021.
In addition, China and Russia have released vaccines, and there are currently 61 vaccines being investigated in clinical trials and at least 85 preclinical products under active investigation.
The vaccine candidates are using both conventional and novel mechanisms of action to elicit an immune response in patients. Conventional methods include attenuated inactivated (killed) virus and recombinant viral protein vaccines to develop immunity. Novel approaches include replication-deficient, adenovirus vector-based vaccines that contain the viral protein, and mRNA-based vaccines, such as the Pfizer and Moderna vaccines, that encode for a SARS-CoV-2 spike protein.
“The special vaccine concern for immunocompromised individuals is introduction of a live virus,” Dr. Stohl said. “Neither the Moderna nor Pfizer vaccines are live viruses, so there should be no special contraindication for such individuals.”
Live vaccine should be avoided in immunocompromised patients, and currently, live SARS-CoV-2 vaccines are only being developed in India and Turkey.
It is not unusual for vaccine trials to begin with cohorts that exclude participants with various health conditions, including those who are immunocompromised. These groups are generally then evaluated in phase 4 trials, or postmarketing surveillance. While the precise number of immunosuppressed adults in the United States is not known, the numbers are believed to be rising because of increased life expectancy among immunosuppressed adults as a result of advances in treatment and new and wider indications for therapies that can affect the immune system.
According to data from the 2013 National Health Interview Survey, an estimated 2.7% of U.S. adults are immunosuppressed. This population covers a broad array of health conditions and medical specialties; people living with inflammatory or autoimmune conditions, such as inflammatory rheumatic diseases (rheumatoid arthritis, axial spondyloarthritis, lupus); inflammatory bowel disease (Crohn’s disease and ulcerative colitis); psoriasis; multiple sclerosis; organ transplant recipients; patients undergoing chemotherapy; and life-long immunosuppression attributable to HIV infection.
As the vaccines begin to roll out and become available, how should clinicians advise their patients, in the absence of any clinical trial data?
Risk vs. benefit
Gilaad Kaplan, MD, MPH, a gastroenterologist and professor of medicine at the University of Calgary (Alta.), noted that the inflammatory bowel disease (IBD) community has dealt with tremendous anxiety during the pandemic because many are immunocompromised because of the medications they use to treat their disease.
“For example, many patients with IBD are on biologics like anti-TNF [tumor necrosis factor] therapies, which are also used in other immune-mediated inflammatory diseases such as rheumatoid arthritis,” he said. “Understandably, individuals with IBD on immunosuppressive medications are concerned about the risk of severe complications due to COVID-19.”
The entire IBD community, along with the world, celebrated the announcement that multiple vaccines are protective against SARS-CoV-2, he noted. “Vaccines offer the potential to reduce the spread of COVID-19, allowing society to revert back to normalcy,” Dr. Kaplan said. “Moreover, for vulnerable populations, including those who are immunocompromised, vaccines offer the potential to directly protect them from the morbidity and mortality associated with COVID-19.”
That said, even though the news of vaccines are extremely promising, some cautions must be raised regarding their use in immunocompromised populations, such as persons with IBD. “The current trials, to my knowledge, did not include immunocompromised individuals and thus, we can only extrapolate from what we know from other trials of different vaccines,” he explained. “We know from prior vaccines studies that the immune response following vaccination is less robust in those who are immunocompromised as compared to a healthy control population.”
Dr. Kaplan also pointed to recent reports of allergic reactions that have been reported in healthy individuals. “We don’t know whether side effects, like allergic reactions, may be different in unstudied populations,” he said. “Thus, the medical and scientific community should prioritize clinical studies of safety and effectiveness of COVID-19 vaccines in immunocompromised populations.”
So, what does this mean for an individual with an immune-mediated inflammatory disease like Crohn’s disease or ulcerative colitis who is immunocompromised? Dr. Kaplan explained that it is a balance between the potential harm of being infected with COVID-19 and the uncertainty of receiving a vaccine in an understudied population. For those who are highly susceptible to dying from COVID-19, such as an older adult with IBD, or someone who faces high exposure, such as a health care worker, the potential protection of the vaccine greatly outweighs the uncertainty.
“However, for individuals who are at otherwise lower risk – for example, young and able to work from home – then waiting a few extra months for postmarketing surveillance studies in immunocompromised populations may be a reasonable approach, as long as these individuals are taking great care to avoid infection,” he said.
No waiting needed
Joel M. Gelfand, MD, MSCE, professor of dermatology and epidemiology at the University of Pennsylvania, Philadelphia, feels that the newly approved vaccine should be safe for most of his patients.
“Patients with psoriatic disease should get the mRNA-based COVID-19 vaccine as soon as possible based on eligibility as determined by the CDC and local public health officials,” he said. “It is not a live vaccine, and therefore patients on biologics or other immune-modulating or immune-suppressing treatment can receive it.”
However, the impact of psoriasis treatment on immune response to the mRNA-based vaccines is not known. Dr. Gelfand noted that, extrapolating from the vaccine literature, there is some evidence that methotrexate reduces response to the influenza vaccine. “However, the clinical significance of this finding is not clear,” he said. “Since the mRNA vaccine needs to be taken twice, a few weeks apart, I do not recommend interrupting or delaying treatment for psoriatic disease while undergoing vaccination for COVID-19.”
Given the reports of allergic reactions, he added that it is advisable for patients with a history of life-threatening allergic reactions such as anaphylaxis or who have been advised to carry an epinephrine autoinjector, to talk with their health care provider to determine if COVID-19 vaccination is medically appropriate.
The National Psoriasis Foundation has issued guidance on COVID-19, explained Steven R. Feldman, MD, PhD, professor of dermatology, pathology, and social sciences & health policy at Wake Forest University, Winston-Salem, N.C., who is also a member of the committee that is working on those guidelines and keeping them up to date. “We are in the process of updating the guidelines with information on COVID vaccines,” he said.
He agreed that there are no contraindications for psoriasis patients to receive the vaccine, regardless of whether they are on immunosuppressive treatment, even though definitive data are lacking. “Fortunately, there’s a lot of good data coming out of Italy that patients with psoriasis on biologics do not appear to be at increased risk of getting COVID or of having worse outcomes from COVID,” he said.
Patients are going to ask about the vaccines, and when counseling them, clinicians should discuss the available data, the residual uncertainty, and patients’ concerns should be considered, Dr. Feldman explained. “There may be some concern that steroids and cyclosporine would reduce the effectiveness of vaccines, but there is no concern that any of the drugs would cause increased risk from nonlive vaccines.”
He added that there is evidence that “patients on biologics who receive nonlive vaccines do develop antibody responses and are immunized.”
Boosting efficacy
Even prior to making their announcement, the American College of Rheumatology had said that they would endorse the vaccine for all patients, explained rheumatologist Brett Smith, DO, from Blount Memorial Physicians Group and East Tennessee Children’s Hospital, Alcoa. “The vaccine is safe for all patients, but the problem may be that it’s not as effective,” he said. “But we don’t know that because it hasn’t been tested.”
With other vaccines, biologic medicines are held for 2 weeks before and afterwards, to get the best response. “But some patients don’t want to stop the medication,” Dr. Smith said. “They are afraid that their symptoms will return.”
As for counseling patients as to whether they should receive this vaccine, he explained that he typically doesn’t try to sway patients one way or another until they are really high risk. “When I counsel, it really depends on the individual situation. And for this vaccine, we have to be open to the fact that many people have already made up their mind.”
There are a lot of questions regarding the vaccine. One is the short time frame of development. “Vaccines typically take 6-10 years to come on the market, and this one is now available after a 3-month study,” Dr. Smith said. “Some have already decided that it’s too new for them.”
The process is also new, and patients need to understand that it doesn’t contain an active virus and “you can’t catch coronavirus from it.”
Dr. Smith also explained that, because the vaccine may be less effective in a person using biologic therapies, there is currently no information available on repeat vaccination. “These are all unanswered questions,” he said. “If the antibodies wane in a short time, can we be revaccinated and in what time frame? We just don’t know that yet.”
Marcelo Bonomi, MD, a medical oncologist from The Ohio State University Comprehensive Cancer Center, Columbus, explained that one way to ensure a more optimal response to the vaccine would be to wait until the patient has finished chemotherapy.* “The vaccine can be offered at that time, and in the meantime, they can take other steps to avoid infection,” he said. “If they are very immunosuppressed, it isn’t worth trying to give the vaccine.”
Cancer patients should be encouraged to stay as healthy as possible, and to wear masks and social distance. “It’s a comprehensive approach. Eat healthy, avoid alcohol and tobacco, and exercise. [These things] will help boost the immune system,” Dr. Bonomi said. “Family members should be encouraged to get vaccinated, which will help them avoid infection and exposing the patient.”
Jim Boonyaratanakornkit, MD, PhD, an infectious disease specialist who cares for cancer patients at the Fred Hutchinson Cancer Research Center, agreed. “Giving a vaccine right after a transplant is a futile endeavor,” he said. “We need to wait 6 months to have an immune response.”
He pointed out there may be a continuing higher number of cases, with high levels peaking in Washington in February and March. “Close friends and family should be vaccinated if possible,” he said, “which will help interrupt transmission.”
The vaccines are using new platforms that are totally different, and there is no clear data as to how long the antibodies will persist. “We know that they last for at least 4 months,” said Dr. Boonyaratanakornkit. “We don’t know what level of antibody will protect them from COVID-19 infection. Current studies are being conducted, but we don’t have that information for anyone yet.”
*Correction, 1/7/21: An earlier version of this article misattributed quotes from Dr. Marcelo Bonomi.
Parents favored virtual learning over in-person school attendance
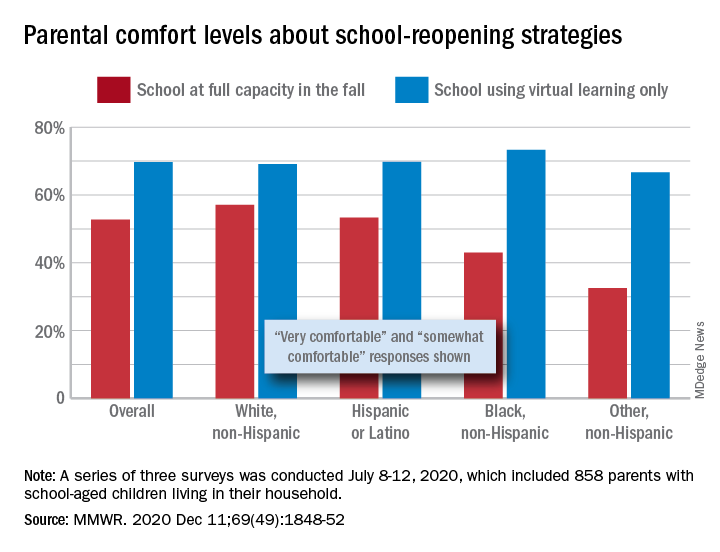
Parents of school-aged children were generally more comfortable with full-time virtual learning in schools in the fall of 2020, compared with full-capacity in-person attendance, according to a survey conducted in July.
Those of racial/ethnic minorities, however, “were less likely to feel that schools should reopen for all students and were more concerned about” several aspects of in-person instruction than were White parents, Leah K. Gilbert, MD, and associates at the Centers for Disease Control and Prevention’s COVID-19 Response Team said in the Morbidity and Mortality Weekly Report.
A slim majority, just under 53% of the 858 parents surveyed, said that they were very or somewhat comfortable with their children returning to schools that were reopening at full capacity, while almost 70% said they were very/somewhat comfortable with schools going exclusively with virtual learning, the investigators reported.
The question about full-capacity attendance in particular showed considerable variation by race and ethnicity, with 57% of White parents saying they were very/somewhat comfortable, versus 53% of Hispanic or Latino parents, 43% of Black parents, and 32.5% of parents of other races/ethnicities (American Indian/Alaska Native, Asian, or multiracial).
Comfort levels were closer regarding virtual learning: Parents of other races/ethnicities were lowest at 67% and Black parents were highest at 73%. When asked about schools reopening at 50% capacity and 50% virtual learning, Black parents were again lowest at 58% with strong or moderate comfort and White parents were highest at 68%, Dr. Gilbert and associates said.
“Although the majority of parent respondents had concerns about both school reopening for in-person instruction and virtual learning, the perceived risk for SARS-CoV-2 infection and poor health outcomes might account for the differences in parental attitudes and concerns by race and ethnicity,” they wrote.
SOURCE: Gilbert LK et al. MMWR. 2020 Dec 11;69(49):1848-52.
Parents of school-aged children were generally more comfortable with full-time virtual learning in schools in the fall of 2020, compared with full-capacity in-person attendance, according to a survey conducted in July.

Those of racial/ethnic minorities, however, “were less likely to feel that schools should reopen for all students and were more concerned about” several aspects of in-person instruction than were White parents, Leah K. Gilbert, MD, and associates at the Centers for Disease Control and Prevention’s COVID-19 Response Team said in the Morbidity and Mortality Weekly Report.
A slim majority, just under 53% of the 858 parents surveyed, said that they were very or somewhat comfortable with their children returning to schools that were reopening at full capacity, while almost 70% said they were very/somewhat comfortable with schools going exclusively with virtual learning, the investigators reported.
The question about full-capacity attendance in particular showed considerable variation by race and ethnicity, with 57% of White parents saying they were very/somewhat comfortable, versus 53% of Hispanic or Latino parents, 43% of Black parents, and 32.5% of parents of other races/ethnicities (American Indian/Alaska Native, Asian, or multiracial).
Comfort levels were closer regarding virtual learning: Parents of other races/ethnicities were lowest at 67% and Black parents were highest at 73%. When asked about schools reopening at 50% capacity and 50% virtual learning, Black parents were again lowest at 58% with strong or moderate comfort and White parents were highest at 68%, Dr. Gilbert and associates said.
“Although the majority of parent respondents had concerns about both school reopening for in-person instruction and virtual learning, the perceived risk for SARS-CoV-2 infection and poor health outcomes might account for the differences in parental attitudes and concerns by race and ethnicity,” they wrote.
SOURCE: Gilbert LK et al. MMWR. 2020 Dec 11;69(49):1848-52.
Parents of school-aged children were generally more comfortable with full-time virtual learning in schools in the fall of 2020, compared with full-capacity in-person attendance, according to a survey conducted in July.

Those of racial/ethnic minorities, however, “were less likely to feel that schools should reopen for all students and were more concerned about” several aspects of in-person instruction than were White parents, Leah K. Gilbert, MD, and associates at the Centers for Disease Control and Prevention’s COVID-19 Response Team said in the Morbidity and Mortality Weekly Report.
A slim majority, just under 53% of the 858 parents surveyed, said that they were very or somewhat comfortable with their children returning to schools that were reopening at full capacity, while almost 70% said they were very/somewhat comfortable with schools going exclusively with virtual learning, the investigators reported.
The question about full-capacity attendance in particular showed considerable variation by race and ethnicity, with 57% of White parents saying they were very/somewhat comfortable, versus 53% of Hispanic or Latino parents, 43% of Black parents, and 32.5% of parents of other races/ethnicities (American Indian/Alaska Native, Asian, or multiracial).
Comfort levels were closer regarding virtual learning: Parents of other races/ethnicities were lowest at 67% and Black parents were highest at 73%. When asked about schools reopening at 50% capacity and 50% virtual learning, Black parents were again lowest at 58% with strong or moderate comfort and White parents were highest at 68%, Dr. Gilbert and associates said.
“Although the majority of parent respondents had concerns about both school reopening for in-person instruction and virtual learning, the perceived risk for SARS-CoV-2 infection and poor health outcomes might account for the differences in parental attitudes and concerns by race and ethnicity,” they wrote.
SOURCE: Gilbert LK et al. MMWR. 2020 Dec 11;69(49):1848-52.
FROM MMWR
FDA clears first OTC rapid at-home COVID diagnostic test
The Food and Drug Administration has issued an emergency-use authorization (EUA) for the first COVID-19 diagnostic test that can be completed at home without a prescription.
Authorization of the Ellume COVID-19 Home Test is “a major milestone in diagnostic testing for COVID-19,” FDA Commissioner Stephen M. Hahn, MD, said in a news release.
“By authorizing a test for over-the-counter use, the FDA allows it to be sold in places like drug stores, where a patient can buy it, swab their nose, run the test, and find out their results in as little as 20 minutes,” said Dr. Hahn.
The Ellume COVID-19 Home Test is a rapid antigen test that detects fragments of the SARS-CoV-2 virus from a nasal swab sample taken from anyone aged 2 years and older, including those not showing any symptoms.
In testing, the Ellume COVID-19 Home Test correctly identified 96% of positive samples and 100% of negative samples in individuals with symptoms.
In people without symptoms, the test correctly identified 91% of positive samples and 96% of negative samples, the FDA said.
The test includes a sterile nasal swab, a dropper, processing fluid, and a Bluetooth-connected analyzer for use with an app on the user’s smartphone. The sample is analyzed and results are automatically transmitted to the user’s smartphone.
“The Ellume COVID-19 home test’s core technology combines ultra-sensitive optics, electronics, and proprietary software to leverage best-in-class digital immunoassay technology with next-generation multi-quantum dot fluorescence technology,” the company said in a news release.
The mobile app requires individuals to input their ZIP code and date of birth, with optional fields including name and email address. The app automatically reports the results as appropriate to public health authorities to monitor disease prevalence.
Ellume expects to produce more than 3 million tests in January 2021. The company said the test will cost around $30.
FDA authorization of this first fully at-home nonprescription COVID-19 diagnostic test follows last month’s EUA for the first prescription COVID-19 test for home use, as reported this news organization.
Since the start of the pandemic, the FDA has authorized more than 225 diagnostic tests for COVID-19, including more than 25 tests that allow for home collection of samples, which are then sent to a lab for testing.
“As we continue to authorize additional tests for home use, we are helping expand Americans’ access to testing, reducing the burden on laboratories and test supplies, and giving Americans more testing options from the comfort and safety of their own homes,” Dr. Hahn said.
“This test, like other antigen tests, is less sensitive and less specific than typical molecular tests run in a lab,” said Jeffrey Shuren, MD, JD, director of FDA’s Center for Devices and Radiological Health, in the release. “However, the fact that it can be used completely at home and return results quickly means that it can play an important role in response to the pandemic.”
As with other antigen tests, a small percentage of positive and negative results from the Ellume test may be false. In patients without symptoms, positive results should be treated as presumptively positive until confirmed by another test as soon as possible, the FDA advised.
This is especially true if there are fewer infections in a particular community, as false-positive results can be more common when antigen tests are used in populations where there is a low prevalence of COVID-19, the agency said.
Because all tests can give false-negative and false-positive results, individuals with positive results should self-isolate and seek additional care from their health care provider.
Individuals who test negative and have symptoms of COVID-19 should follow up with their health care provider, as negative results don’t preclude an individual from SARS-CoV-2 infection.
A version of this article first appeared on Medscape.com.
The Food and Drug Administration has issued an emergency-use authorization (EUA) for the first COVID-19 diagnostic test that can be completed at home without a prescription.
Authorization of the Ellume COVID-19 Home Test is “a major milestone in diagnostic testing for COVID-19,” FDA Commissioner Stephen M. Hahn, MD, said in a news release.
“By authorizing a test for over-the-counter use, the FDA allows it to be sold in places like drug stores, where a patient can buy it, swab their nose, run the test, and find out their results in as little as 20 minutes,” said Dr. Hahn.
The Ellume COVID-19 Home Test is a rapid antigen test that detects fragments of the SARS-CoV-2 virus from a nasal swab sample taken from anyone aged 2 years and older, including those not showing any symptoms.
In testing, the Ellume COVID-19 Home Test correctly identified 96% of positive samples and 100% of negative samples in individuals with symptoms.
In people without symptoms, the test correctly identified 91% of positive samples and 96% of negative samples, the FDA said.
The test includes a sterile nasal swab, a dropper, processing fluid, and a Bluetooth-connected analyzer for use with an app on the user’s smartphone. The sample is analyzed and results are automatically transmitted to the user’s smartphone.
“The Ellume COVID-19 home test’s core technology combines ultra-sensitive optics, electronics, and proprietary software to leverage best-in-class digital immunoassay technology with next-generation multi-quantum dot fluorescence technology,” the company said in a news release.
The mobile app requires individuals to input their ZIP code and date of birth, with optional fields including name and email address. The app automatically reports the results as appropriate to public health authorities to monitor disease prevalence.
Ellume expects to produce more than 3 million tests in January 2021. The company said the test will cost around $30.
FDA authorization of this first fully at-home nonprescription COVID-19 diagnostic test follows last month’s EUA for the first prescription COVID-19 test for home use, as reported this news organization.
Since the start of the pandemic, the FDA has authorized more than 225 diagnostic tests for COVID-19, including more than 25 tests that allow for home collection of samples, which are then sent to a lab for testing.
“As we continue to authorize additional tests for home use, we are helping expand Americans’ access to testing, reducing the burden on laboratories and test supplies, and giving Americans more testing options from the comfort and safety of their own homes,” Dr. Hahn said.
“This test, like other antigen tests, is less sensitive and less specific than typical molecular tests run in a lab,” said Jeffrey Shuren, MD, JD, director of FDA’s Center for Devices and Radiological Health, in the release. “However, the fact that it can be used completely at home and return results quickly means that it can play an important role in response to the pandemic.”
As with other antigen tests, a small percentage of positive and negative results from the Ellume test may be false. In patients without symptoms, positive results should be treated as presumptively positive until confirmed by another test as soon as possible, the FDA advised.
This is especially true if there are fewer infections in a particular community, as false-positive results can be more common when antigen tests are used in populations where there is a low prevalence of COVID-19, the agency said.
Because all tests can give false-negative and false-positive results, individuals with positive results should self-isolate and seek additional care from their health care provider.
Individuals who test negative and have symptoms of COVID-19 should follow up with their health care provider, as negative results don’t preclude an individual from SARS-CoV-2 infection.
A version of this article first appeared on Medscape.com.
The Food and Drug Administration has issued an emergency-use authorization (EUA) for the first COVID-19 diagnostic test that can be completed at home without a prescription.
Authorization of the Ellume COVID-19 Home Test is “a major milestone in diagnostic testing for COVID-19,” FDA Commissioner Stephen M. Hahn, MD, said in a news release.
“By authorizing a test for over-the-counter use, the FDA allows it to be sold in places like drug stores, where a patient can buy it, swab their nose, run the test, and find out their results in as little as 20 minutes,” said Dr. Hahn.
The Ellume COVID-19 Home Test is a rapid antigen test that detects fragments of the SARS-CoV-2 virus from a nasal swab sample taken from anyone aged 2 years and older, including those not showing any symptoms.
In testing, the Ellume COVID-19 Home Test correctly identified 96% of positive samples and 100% of negative samples in individuals with symptoms.
In people without symptoms, the test correctly identified 91% of positive samples and 96% of negative samples, the FDA said.
The test includes a sterile nasal swab, a dropper, processing fluid, and a Bluetooth-connected analyzer for use with an app on the user’s smartphone. The sample is analyzed and results are automatically transmitted to the user’s smartphone.
“The Ellume COVID-19 home test’s core technology combines ultra-sensitive optics, electronics, and proprietary software to leverage best-in-class digital immunoassay technology with next-generation multi-quantum dot fluorescence technology,” the company said in a news release.
The mobile app requires individuals to input their ZIP code and date of birth, with optional fields including name and email address. The app automatically reports the results as appropriate to public health authorities to monitor disease prevalence.
Ellume expects to produce more than 3 million tests in January 2021. The company said the test will cost around $30.
FDA authorization of this first fully at-home nonprescription COVID-19 diagnostic test follows last month’s EUA for the first prescription COVID-19 test for home use, as reported this news organization.
Since the start of the pandemic, the FDA has authorized more than 225 diagnostic tests for COVID-19, including more than 25 tests that allow for home collection of samples, which are then sent to a lab for testing.
“As we continue to authorize additional tests for home use, we are helping expand Americans’ access to testing, reducing the burden on laboratories and test supplies, and giving Americans more testing options from the comfort and safety of their own homes,” Dr. Hahn said.
“This test, like other antigen tests, is less sensitive and less specific than typical molecular tests run in a lab,” said Jeffrey Shuren, MD, JD, director of FDA’s Center for Devices and Radiological Health, in the release. “However, the fact that it can be used completely at home and return results quickly means that it can play an important role in response to the pandemic.”
As with other antigen tests, a small percentage of positive and negative results from the Ellume test may be false. In patients without symptoms, positive results should be treated as presumptively positive until confirmed by another test as soon as possible, the FDA advised.
This is especially true if there are fewer infections in a particular community, as false-positive results can be more common when antigen tests are used in populations where there is a low prevalence of COVID-19, the agency said.
Because all tests can give false-negative and false-positive results, individuals with positive results should self-isolate and seek additional care from their health care provider.
Individuals who test negative and have symptoms of COVID-19 should follow up with their health care provider, as negative results don’t preclude an individual from SARS-CoV-2 infection.
A version of this article first appeared on Medscape.com.
Nobel laureates on their hunt for the cure
Researchers have worked at record-breaking speed to not only identify and characterize the novel coronavirus, but also to develop potential vaccines; it is a race that another trio of scientists, awarded the 2020 Nobel Prize in Medicine, know well after their own decades-long marathon to crack the code of hepatitis C.
The RNA virus vexed researchers like Harvey J. Alter, MD; Michael Houghton, PhD; and Charles M. Rice, PhD, for years. Unlike today’s parallel sequencing and polymerase chain reaction, screening at the time was tedious and painstaking. But they were all in for some big highs, some dead-ends, and a little inspiration by way of author Lewis Carroll.
“Our undertaking was a success,” Dr. Alter said in an interview, “because everyone got on board.” Investigators, the Food and Drug Administration, and blood banks all worked together. It was a national effort, he added.
The Laureates will share the $1 million prize that recognizes their achievement. This is the second time that scientists who have devoted their time to the pursuit of viral hepatitis have been honored.
Two main types of infectious hepatitis were identified in the 1940s. The first, hepatitis A, is transmitted by polluted water or food and generally has little long-term effect on the patient. The second, transmitted through blood and other bodily fluids, is a much more serious threat.
In the 1960s, Baruch Blumberg, MD, ascertained that blood-borne hepatitis was caused by an insidious virus – hepatitis B – which silently causes liver complications in otherwise healthy people years after infection. Dr. Blumberg’s discovery led to the development of diagnostic tests and an effective vaccine; he was awarded the Nobel Prize in Physiology or Medicine in 1976.
At that time, Dr. Alter was working at the National Institutes of Health in Bethesda, Md., studying the occurrence of hepatitis in patients who had received blood transfusions. Globally, blood-borne hepatitis was causing more than a million deaths each year.
When screening began in 1969 to prevent people with hepatitis A or hepatitis B from donating blood, many recipients were spared from developing liver inflammation after transfusion. However, some people were still getting sick and researchers began to suspect that something dubbed “non-A, non-B” was lurking in donated blood.
As scientists were tracking it down in the micro world, public health officials and others tried to block the presumed virus in the macro world. In the United States, this meant changing blood-donation practices. When it became clear that as many as one in five transfusion recipients developed hepatitis, probably as the result of a virus, the blood donation system switched from being a paid enterprise to a volunteer activity.
It was anticipated that this would eliminate one source of the virus – users of street drugs – and it did; rates of transmission after transfusion dropped by half. By the 1980s, researchers had figured out that donors carrying the mysterious pathogen often had elevated levels of antibodies to the hepatitis B virus or elevated levels of ALT. Blood banks then began testing prospective donors and cut the incidence of non-A, non-B hepatitis by another half.
When Dr. Alter and his team got to work studying chimpanzees, they were able to confirm that the non-A, non-B agent was transmittable by blood transfusion. But the next step proved insurmountable. “People knew what the virus would look like if we found it, but we couldn’t find it,” Dr. Alter said.
“Then Chiron came along and cloned it,” he recounts.
The tiny start-up company was housed in a few rented rooms in an abandoned plant in Emeryville, Calif. This is where Nobel Laureate Dr. Houghton and coworkers Qui-Lim Choo, PhD, and George Kuo, PhD, spent 7 years chasing the mystery pathogen. When every traditional method to pin it down failed, the team tried some nontraditional approaches.
They found success when they painstakingly cloned genomic fragments from the blood of an infected chimpanzee and then screened this library using serum isolated from affected patients. The hope was that antibodies in the serum samples would stick to the viral genetic material and create a signpost indicating its presence.
This “fishing expedition” was disappointing at first, said Dr. Choo, now vice president of research at Nansha Biologics Limited in Hong Kong. “We didn’t catch any fish.”
He likened the pursuit, which took place under growing pressure from upper management, to that in “The Imitation Game,” the film that depicted Alan Turing’s effort to decrypt German intelligence messages for the British government during World War II.
The effort wasn’t working out and the team was down. So to keep spirits up, Dr. Kuo shared a snippet of the Lewis Carroll poem “The Hunting of the Snark.” It just happened to encapsulate the frustrations of their pursuit while also encouraging perseverance: “For the Snark’s a peculiar creature, that won’t / Be caught in a commonplace way. / Do all that you know, and try all that you don’t: / Not a chance must be wasted today!”
Those words motivated the team to keep going, to try different approaches and, ultimately, to find the hepatitis C virus, said Dr. Kuo, now retired.
During the tedious process of screening millions of clones, Dr. Choo spotted a single likely area on one of the plates that his “gut feeling” told him would contain the viral material. And it did. The pathogen – RNA packed in a lipid coat – was a member of the Flaviviridae family.
In 1989, the team reported that they’d identified a new flavivirus associated with posttransfusion hepatitis, and published their findings in Science. Later that year, an antibody test for it was described in Science by the team, which included Dr. Alter.
The success was a “fantastic feeling,” mused Dr. Houghton, now a virologist at the University of Alberta, Edmonton. “I’ve often said that, from 1989 to 1991, I was high just on the discovery.” After that, “researchers and the pharmaceutical industry did a great job coming up with really effective antivirals.”
“Mike, with his team, took 7 years to get this tiny little piece of viral genome, which reacted with convalescent patient serum, and used that to build up the sequence. That’s where I come into it,” said Dr. Rice, professor of virology at Rockefeller University, New York, and the third scientist in the Nobel Prize–winning trio.
The next step was to confirm that this was causing the hepatitis in patients who received blood transfusions. Over the course of several years, Dr. Rice and colleagues at the Washington University, St. Louis, engineered a version of the virus that retained its replicating capacity and injected it into chimpanzees. The animals developed hepatitis, confirming that this flavivirus was indeed the cause. More than 8 years after the virus was identified, scientists had proof of its infectiousness.
“We finally knew the structure of the viral genome,” said Dr. Rice. “We showed that the sequence was sufficient to initiate infection and cause disease.”
Those findings, published in 1997 in Science, were the key final step in the search for a molecular target for therapies and, maybe someday, a vaccine.
For the first time in history, hepatitis C can now be cured, raising hopes that the virus will be eradicated from the world population. But obstacles remain as the World Health Organization works toward its goal of eliminating hepatitis C–related disease by 2030.
Rates have actually risen in the United States as the opioid epidemic has taken hold because the virus is common in people who use street drugs and are at increased risk for behaviors that lead to transmission.
The prevalence of chronic hepatitis C now hovers around 1%. In 2019, it affected an estimated 3.8 million Americans, and more than 39,000 people died of hepatitis C–related causes. Of all deaths from liver cancer in 2019, 39% were attributable to hepatitis C.
This “silent killer” can take years to develop, which dampens the sense of immediacy to help, said Jean-Michel Piedagnel, director of the Drugs for Neglected Diseases nonprofit initiative.
People infected with hepatitis C are often part of marginalized groups. There isn’t typically a common demographic factor to unite them in advocacy.
And even though treatment can cost as little as $200 in countries where generic drugs are available, money can still be an issue, Mr. Piedagnel pointed out.
The COVID-19 response shows how quickly health systems can evolve in a crisis and adapt. “If there is political will, means can be found, said Cary James, CEO at the World Hepatitis Alliance.
The incredible speed of vaccine development for SARS-CoV-2 has impressed the Nobel Laureates, who have yet to see a vaccine for hepatitis C.
Dr. Houghton said he hopes this momentum will perpetuate new emergency filings for hepatitis C vaccines.
If even a tiny fraction of the money that’s gone into COVID-19 were used, Dr. Rice pointed out, “we’d have a hep C vaccine by now.”
A version of this article first appeared on Medscape.com
Researchers have worked at record-breaking speed to not only identify and characterize the novel coronavirus, but also to develop potential vaccines; it is a race that another trio of scientists, awarded the 2020 Nobel Prize in Medicine, know well after their own decades-long marathon to crack the code of hepatitis C.
The RNA virus vexed researchers like Harvey J. Alter, MD; Michael Houghton, PhD; and Charles M. Rice, PhD, for years. Unlike today’s parallel sequencing and polymerase chain reaction, screening at the time was tedious and painstaking. But they were all in for some big highs, some dead-ends, and a little inspiration by way of author Lewis Carroll.
“Our undertaking was a success,” Dr. Alter said in an interview, “because everyone got on board.” Investigators, the Food and Drug Administration, and blood banks all worked together. It was a national effort, he added.
The Laureates will share the $1 million prize that recognizes their achievement. This is the second time that scientists who have devoted their time to the pursuit of viral hepatitis have been honored.
Two main types of infectious hepatitis were identified in the 1940s. The first, hepatitis A, is transmitted by polluted water or food and generally has little long-term effect on the patient. The second, transmitted through blood and other bodily fluids, is a much more serious threat.
In the 1960s, Baruch Blumberg, MD, ascertained that blood-borne hepatitis was caused by an insidious virus – hepatitis B – which silently causes liver complications in otherwise healthy people years after infection. Dr. Blumberg’s discovery led to the development of diagnostic tests and an effective vaccine; he was awarded the Nobel Prize in Physiology or Medicine in 1976.
At that time, Dr. Alter was working at the National Institutes of Health in Bethesda, Md., studying the occurrence of hepatitis in patients who had received blood transfusions. Globally, blood-borne hepatitis was causing more than a million deaths each year.
When screening began in 1969 to prevent people with hepatitis A or hepatitis B from donating blood, many recipients were spared from developing liver inflammation after transfusion. However, some people were still getting sick and researchers began to suspect that something dubbed “non-A, non-B” was lurking in donated blood.
As scientists were tracking it down in the micro world, public health officials and others tried to block the presumed virus in the macro world. In the United States, this meant changing blood-donation practices. When it became clear that as many as one in five transfusion recipients developed hepatitis, probably as the result of a virus, the blood donation system switched from being a paid enterprise to a volunteer activity.
It was anticipated that this would eliminate one source of the virus – users of street drugs – and it did; rates of transmission after transfusion dropped by half. By the 1980s, researchers had figured out that donors carrying the mysterious pathogen often had elevated levels of antibodies to the hepatitis B virus or elevated levels of ALT. Blood banks then began testing prospective donors and cut the incidence of non-A, non-B hepatitis by another half.
When Dr. Alter and his team got to work studying chimpanzees, they were able to confirm that the non-A, non-B agent was transmittable by blood transfusion. But the next step proved insurmountable. “People knew what the virus would look like if we found it, but we couldn’t find it,” Dr. Alter said.
“Then Chiron came along and cloned it,” he recounts.
The tiny start-up company was housed in a few rented rooms in an abandoned plant in Emeryville, Calif. This is where Nobel Laureate Dr. Houghton and coworkers Qui-Lim Choo, PhD, and George Kuo, PhD, spent 7 years chasing the mystery pathogen. When every traditional method to pin it down failed, the team tried some nontraditional approaches.
They found success when they painstakingly cloned genomic fragments from the blood of an infected chimpanzee and then screened this library using serum isolated from affected patients. The hope was that antibodies in the serum samples would stick to the viral genetic material and create a signpost indicating its presence.
This “fishing expedition” was disappointing at first, said Dr. Choo, now vice president of research at Nansha Biologics Limited in Hong Kong. “We didn’t catch any fish.”
He likened the pursuit, which took place under growing pressure from upper management, to that in “The Imitation Game,” the film that depicted Alan Turing’s effort to decrypt German intelligence messages for the British government during World War II.
The effort wasn’t working out and the team was down. So to keep spirits up, Dr. Kuo shared a snippet of the Lewis Carroll poem “The Hunting of the Snark.” It just happened to encapsulate the frustrations of their pursuit while also encouraging perseverance: “For the Snark’s a peculiar creature, that won’t / Be caught in a commonplace way. / Do all that you know, and try all that you don’t: / Not a chance must be wasted today!”
Those words motivated the team to keep going, to try different approaches and, ultimately, to find the hepatitis C virus, said Dr. Kuo, now retired.
During the tedious process of screening millions of clones, Dr. Choo spotted a single likely area on one of the plates that his “gut feeling” told him would contain the viral material. And it did. The pathogen – RNA packed in a lipid coat – was a member of the Flaviviridae family.
In 1989, the team reported that they’d identified a new flavivirus associated with posttransfusion hepatitis, and published their findings in Science. Later that year, an antibody test for it was described in Science by the team, which included Dr. Alter.
The success was a “fantastic feeling,” mused Dr. Houghton, now a virologist at the University of Alberta, Edmonton. “I’ve often said that, from 1989 to 1991, I was high just on the discovery.” After that, “researchers and the pharmaceutical industry did a great job coming up with really effective antivirals.”
“Mike, with his team, took 7 years to get this tiny little piece of viral genome, which reacted with convalescent patient serum, and used that to build up the sequence. That’s where I come into it,” said Dr. Rice, professor of virology at Rockefeller University, New York, and the third scientist in the Nobel Prize–winning trio.
The next step was to confirm that this was causing the hepatitis in patients who received blood transfusions. Over the course of several years, Dr. Rice and colleagues at the Washington University, St. Louis, engineered a version of the virus that retained its replicating capacity and injected it into chimpanzees. The animals developed hepatitis, confirming that this flavivirus was indeed the cause. More than 8 years after the virus was identified, scientists had proof of its infectiousness.
“We finally knew the structure of the viral genome,” said Dr. Rice. “We showed that the sequence was sufficient to initiate infection and cause disease.”
Those findings, published in 1997 in Science, were the key final step in the search for a molecular target for therapies and, maybe someday, a vaccine.
For the first time in history, hepatitis C can now be cured, raising hopes that the virus will be eradicated from the world population. But obstacles remain as the World Health Organization works toward its goal of eliminating hepatitis C–related disease by 2030.
Rates have actually risen in the United States as the opioid epidemic has taken hold because the virus is common in people who use street drugs and are at increased risk for behaviors that lead to transmission.
The prevalence of chronic hepatitis C now hovers around 1%. In 2019, it affected an estimated 3.8 million Americans, and more than 39,000 people died of hepatitis C–related causes. Of all deaths from liver cancer in 2019, 39% were attributable to hepatitis C.
This “silent killer” can take years to develop, which dampens the sense of immediacy to help, said Jean-Michel Piedagnel, director of the Drugs for Neglected Diseases nonprofit initiative.
People infected with hepatitis C are often part of marginalized groups. There isn’t typically a common demographic factor to unite them in advocacy.
And even though treatment can cost as little as $200 in countries where generic drugs are available, money can still be an issue, Mr. Piedagnel pointed out.
The COVID-19 response shows how quickly health systems can evolve in a crisis and adapt. “If there is political will, means can be found, said Cary James, CEO at the World Hepatitis Alliance.
The incredible speed of vaccine development for SARS-CoV-2 has impressed the Nobel Laureates, who have yet to see a vaccine for hepatitis C.
Dr. Houghton said he hopes this momentum will perpetuate new emergency filings for hepatitis C vaccines.
If even a tiny fraction of the money that’s gone into COVID-19 were used, Dr. Rice pointed out, “we’d have a hep C vaccine by now.”
A version of this article first appeared on Medscape.com
Researchers have worked at record-breaking speed to not only identify and characterize the novel coronavirus, but also to develop potential vaccines; it is a race that another trio of scientists, awarded the 2020 Nobel Prize in Medicine, know well after their own decades-long marathon to crack the code of hepatitis C.
The RNA virus vexed researchers like Harvey J. Alter, MD; Michael Houghton, PhD; and Charles M. Rice, PhD, for years. Unlike today’s parallel sequencing and polymerase chain reaction, screening at the time was tedious and painstaking. But they were all in for some big highs, some dead-ends, and a little inspiration by way of author Lewis Carroll.
“Our undertaking was a success,” Dr. Alter said in an interview, “because everyone got on board.” Investigators, the Food and Drug Administration, and blood banks all worked together. It was a national effort, he added.
The Laureates will share the $1 million prize that recognizes their achievement. This is the second time that scientists who have devoted their time to the pursuit of viral hepatitis have been honored.
Two main types of infectious hepatitis were identified in the 1940s. The first, hepatitis A, is transmitted by polluted water or food and generally has little long-term effect on the patient. The second, transmitted through blood and other bodily fluids, is a much more serious threat.
In the 1960s, Baruch Blumberg, MD, ascertained that blood-borne hepatitis was caused by an insidious virus – hepatitis B – which silently causes liver complications in otherwise healthy people years after infection. Dr. Blumberg’s discovery led to the development of diagnostic tests and an effective vaccine; he was awarded the Nobel Prize in Physiology or Medicine in 1976.
At that time, Dr. Alter was working at the National Institutes of Health in Bethesda, Md., studying the occurrence of hepatitis in patients who had received blood transfusions. Globally, blood-borne hepatitis was causing more than a million deaths each year.
When screening began in 1969 to prevent people with hepatitis A or hepatitis B from donating blood, many recipients were spared from developing liver inflammation after transfusion. However, some people were still getting sick and researchers began to suspect that something dubbed “non-A, non-B” was lurking in donated blood.
As scientists were tracking it down in the micro world, public health officials and others tried to block the presumed virus in the macro world. In the United States, this meant changing blood-donation practices. When it became clear that as many as one in five transfusion recipients developed hepatitis, probably as the result of a virus, the blood donation system switched from being a paid enterprise to a volunteer activity.
It was anticipated that this would eliminate one source of the virus – users of street drugs – and it did; rates of transmission after transfusion dropped by half. By the 1980s, researchers had figured out that donors carrying the mysterious pathogen often had elevated levels of antibodies to the hepatitis B virus or elevated levels of ALT. Blood banks then began testing prospective donors and cut the incidence of non-A, non-B hepatitis by another half.
When Dr. Alter and his team got to work studying chimpanzees, they were able to confirm that the non-A, non-B agent was transmittable by blood transfusion. But the next step proved insurmountable. “People knew what the virus would look like if we found it, but we couldn’t find it,” Dr. Alter said.
“Then Chiron came along and cloned it,” he recounts.
The tiny start-up company was housed in a few rented rooms in an abandoned plant in Emeryville, Calif. This is where Nobel Laureate Dr. Houghton and coworkers Qui-Lim Choo, PhD, and George Kuo, PhD, spent 7 years chasing the mystery pathogen. When every traditional method to pin it down failed, the team tried some nontraditional approaches.
They found success when they painstakingly cloned genomic fragments from the blood of an infected chimpanzee and then screened this library using serum isolated from affected patients. The hope was that antibodies in the serum samples would stick to the viral genetic material and create a signpost indicating its presence.
This “fishing expedition” was disappointing at first, said Dr. Choo, now vice president of research at Nansha Biologics Limited in Hong Kong. “We didn’t catch any fish.”
He likened the pursuit, which took place under growing pressure from upper management, to that in “The Imitation Game,” the film that depicted Alan Turing’s effort to decrypt German intelligence messages for the British government during World War II.
The effort wasn’t working out and the team was down. So to keep spirits up, Dr. Kuo shared a snippet of the Lewis Carroll poem “The Hunting of the Snark.” It just happened to encapsulate the frustrations of their pursuit while also encouraging perseverance: “For the Snark’s a peculiar creature, that won’t / Be caught in a commonplace way. / Do all that you know, and try all that you don’t: / Not a chance must be wasted today!”
Those words motivated the team to keep going, to try different approaches and, ultimately, to find the hepatitis C virus, said Dr. Kuo, now retired.
During the tedious process of screening millions of clones, Dr. Choo spotted a single likely area on one of the plates that his “gut feeling” told him would contain the viral material. And it did. The pathogen – RNA packed in a lipid coat – was a member of the Flaviviridae family.
In 1989, the team reported that they’d identified a new flavivirus associated with posttransfusion hepatitis, and published their findings in Science. Later that year, an antibody test for it was described in Science by the team, which included Dr. Alter.
The success was a “fantastic feeling,” mused Dr. Houghton, now a virologist at the University of Alberta, Edmonton. “I’ve often said that, from 1989 to 1991, I was high just on the discovery.” After that, “researchers and the pharmaceutical industry did a great job coming up with really effective antivirals.”
“Mike, with his team, took 7 years to get this tiny little piece of viral genome, which reacted with convalescent patient serum, and used that to build up the sequence. That’s where I come into it,” said Dr. Rice, professor of virology at Rockefeller University, New York, and the third scientist in the Nobel Prize–winning trio.
The next step was to confirm that this was causing the hepatitis in patients who received blood transfusions. Over the course of several years, Dr. Rice and colleagues at the Washington University, St. Louis, engineered a version of the virus that retained its replicating capacity and injected it into chimpanzees. The animals developed hepatitis, confirming that this flavivirus was indeed the cause. More than 8 years after the virus was identified, scientists had proof of its infectiousness.
“We finally knew the structure of the viral genome,” said Dr. Rice. “We showed that the sequence was sufficient to initiate infection and cause disease.”
Those findings, published in 1997 in Science, were the key final step in the search for a molecular target for therapies and, maybe someday, a vaccine.
For the first time in history, hepatitis C can now be cured, raising hopes that the virus will be eradicated from the world population. But obstacles remain as the World Health Organization works toward its goal of eliminating hepatitis C–related disease by 2030.
Rates have actually risen in the United States as the opioid epidemic has taken hold because the virus is common in people who use street drugs and are at increased risk for behaviors that lead to transmission.
The prevalence of chronic hepatitis C now hovers around 1%. In 2019, it affected an estimated 3.8 million Americans, and more than 39,000 people died of hepatitis C–related causes. Of all deaths from liver cancer in 2019, 39% were attributable to hepatitis C.
This “silent killer” can take years to develop, which dampens the sense of immediacy to help, said Jean-Michel Piedagnel, director of the Drugs for Neglected Diseases nonprofit initiative.
People infected with hepatitis C are often part of marginalized groups. There isn’t typically a common demographic factor to unite them in advocacy.
And even though treatment can cost as little as $200 in countries where generic drugs are available, money can still be an issue, Mr. Piedagnel pointed out.
The COVID-19 response shows how quickly health systems can evolve in a crisis and adapt. “If there is political will, means can be found, said Cary James, CEO at the World Hepatitis Alliance.
The incredible speed of vaccine development for SARS-CoV-2 has impressed the Nobel Laureates, who have yet to see a vaccine for hepatitis C.
Dr. Houghton said he hopes this momentum will perpetuate new emergency filings for hepatitis C vaccines.
If even a tiny fraction of the money that’s gone into COVID-19 were used, Dr. Rice pointed out, “we’d have a hep C vaccine by now.”
A version of this article first appeared on Medscape.com
Six big changes coming for office-visit coding
Betsy Nicoletti, MS, a nationally recognized coding expert, will take your coding questions via email and provide guidance on how to code properly to maximize reimbursement. Have a question about coding? Send it to [email protected].
thanks to the American Medical Association.
The first major changes to the definitions for E/M services will be in effect as of Jan. 1, 2021, with all payers expected to adopt these new guidelines. In particular, the AMA has revised the definitions for E/M codes 99202-99215 in the Current Procedural Terminology (CPT) 2021 codebook. The existing guidelines were developed in 1995 and 1997 and remain in effect for all other E/M services determined by history, exam, and medical decision-making (MDM).
What do the new changes mean to you? In 2021, for new and established office and other outpatient services reported with codes 99202-99215, a clinician may select the code on the basis of time or MDM.
There are three elements in MDM, and two of three are required. These elements are the number and complexity of problems addressed, amount and/or complexity of data to be reviewed and analyzed, and risk of complications and/or morbidity or mortality of patient management.
Make sure you familiarize yourself with these six big changes. It may take a bit of time to integrate these new processes into your daily routine, but wrapping your head around them as soon as possible can help boost your bottom line:
1. History and exam don’t count toward level of service
Physicians, advanced practice registered nurses, and physician assistants won’t use history or exam to select what level of code to bill for office visits 99202-99215, as they did in the past. They need only document a medically appropriate history and exam. The history may be obtained by staff members and reviewed by the billing practitioner.
While specific history and exam requirements disappear for office visit codes, they remain for all other types of visits, selected on the basis of history, exam, and MDM, such as hospital services, nursing facility services, and home and domiciliary care. So, say goodbye to “all other systems reviewed and negative” in office notes, but keep it handy for those other E/M codes.
2. All time spent caring for the patient on a particular day counts
This includes all time spent on the day of service, including preparing to see the patient, seeing the patient, phone calls or other work done after the visit (if not billed with a care management or other CPT code), and documenting in the medical record. The AMA developed new guidelines for using time for office and other outpatient services. For codes 99202-99215, count all of the face-to-face and non–face-to-face time spent by the billing clinician on the day of the visit. Counseling does not need to be more than 50% of the total time.
Do not include any staff time or time spent on any days before or after the visit. This allows clinicians to capture the work when a significant amount of it takes place before or after the visit with the patient, and to bill for it on the day of the visit.
According to the 2021 CPT codebook, physician or other qualified health care professional time includes the following activities:
- Preparing to see the patient (e.g., review of tests).
- Obtaining and/or reviewing separately obtained history.
- Performing a medically appropriate examination and/or evaluation.
- Counseling and educating the patient/family/caregiver.
- Ordering medications, tests, or procedures.
- Referring and communicating with other health care professionals (when not separately reported).
- Documenting clinical information in the electronic or other health record.
- Independently interpreting results (not separately reported) and communicating results to the patient/family/caregiver.
- Care coordination (not separately reported).
3. Soon to be gone: ‘new to the examiner’ and ‘workup planned’
The current guidelines don’t differentiate between a new problem to the clinician or an established problem to the clinician. So it doesn’t matter whether you’re hearing about a particular problem for the first time or the fifth time. The new office and outpatient services guidelines define problems only as they relate to the patient. For example, when selecting a level of service, a chronic problem with a mild exacerbation is the same level whether it’s the primary care physician seeing the patient for the 10th time to help manage her diabetes or the endocrinologist seeing the patient for the first time.
In the current guidelines (1995 and 1997), additional weight is given in selecting the level of MDM for a problem that’s new to the examiner with a workup planned, yet when the diagnostic test couldn’t be completed at the visit. This concept is gone from element of number and complexity of new problems. Ordering diagnostic tests is part of the second element, the amount and/or complexity of data to be reviewed.
4. Different guidelines if you need a history from a parent or other source
The new guidelines recognize the additional work required by the clinician when the patient is unable to give a history or when the practitioner doesn’t find the history to be reliable.
For example, in the case of a baby or child who is unable to give a history, the parent counts as an “independent historian,” according to the new guidelines. Likewise, for a patient with dementia, the caregiver counts as a historian. Note, however, that the criteria is not met simply because the patient is accompanied by another person. The additional weight in selecting the level of service is based on the patient being unable to give a reliable history.
Bottom line: In cases where patients are unable to communicate clearly, physicians or other providers should document the necessity of getting a complete history and who provided it.
5. A new spin on social determinants of health (SDoH)
In the risk of morbidity and/or mortality element, conditions described as “social determinants of health” are considered moderate complexity. SDoH are social and environmental factors that affect a patient’s health and medical outcomes. These include homelessness, inability to afford medications, food insecurity, and occupational exposure to risk factors. These circumstances are reported with codes in categories Z55-Z65.
In the past, physicians often documented this information in their office notes but rarely added a diagnosis code that described the patient’s situation. The ICD-10-CM code set includes codes that describe these factors. Using them allows the practice to track patients who have increased needs, and it communicates to payers the complexity of caring for these patients.
6. Risks related to surgery are defined
The current guidelines assign different levels of risk to minor and major surgery. They also include differentiation for “minor surgery with no identified risk factors,” “minor surgery with identified risk factors,” “elective major surgery with no identified risk factors,” and “elective major surgery with identified risk factors.” The old guidelines didn’t state whether the risk factors pertained to the patient – such as smoking, heart disease, or high body mass index – or to the procedure itself.
The new guidelines specifically say that it’s both. In the risk column, “decision regarding minor surgery with identified patient or procedure risk factors” and “decision regarding elective major surgery without patient or procedure risk factors” are both considered moderate. “Decision regarding elective major surgery with identified patient or procedure risk factors” and “decision regarding emergency major surgery” are in the high complexity column for risk.
Keep in mind that two of three elements are required: the number and complexity of problems, amount of data, and morbidity/mortality risk. Risk of morbidity/mortality alone doesn’t count as the basis for selecting the code. Of course, when surgeons see this, they ask, “What major procedures don’t have identified risk factors?”
Note, too, that these new CPT guidelines do not define the terms “minor” and “major” surgery. For payment reasons related to the postop period, the Centers for Medicare & Medicaid Services defines minor surgery as a procedure with 0-10 global days and a major surgery as a procedure with 90 global days. However, there are many procedures with 0 global days (endoscopy, cardiac catheterization) that are not minor procedures. Hopefully, the AMA will clarify this in 2021.
What’s the take-away for clinicians?
There are sure to be shifts in coding patterns based on these new guidelines. Some specialties will find that not being able to select a service based on history and exam alone will lower the level of service for which they can bill. Some practices, on the other hand, will be able to code for more high-level visits, without the need for a complete review of systems or a comprehensive exam.
The biggest challenge will be for practices that provide services both in the hospital and in the office, because they’ll have to use both sets of guidelines, depending on which type of service they’re performing.
For more details on what’s coming your way beginning on New Year’s Day, you may want to read the 16-page AMA document .
A version of this article first appeared on Medscape.com.
Betsy Nicoletti, MS, a nationally recognized coding expert, will take your coding questions via email and provide guidance on how to code properly to maximize reimbursement. Have a question about coding? Send it to [email protected].
thanks to the American Medical Association.
The first major changes to the definitions for E/M services will be in effect as of Jan. 1, 2021, with all payers expected to adopt these new guidelines. In particular, the AMA has revised the definitions for E/M codes 99202-99215 in the Current Procedural Terminology (CPT) 2021 codebook. The existing guidelines were developed in 1995 and 1997 and remain in effect for all other E/M services determined by history, exam, and medical decision-making (MDM).
What do the new changes mean to you? In 2021, for new and established office and other outpatient services reported with codes 99202-99215, a clinician may select the code on the basis of time or MDM.
There are three elements in MDM, and two of three are required. These elements are the number and complexity of problems addressed, amount and/or complexity of data to be reviewed and analyzed, and risk of complications and/or morbidity or mortality of patient management.
Make sure you familiarize yourself with these six big changes. It may take a bit of time to integrate these new processes into your daily routine, but wrapping your head around them as soon as possible can help boost your bottom line:
1. History and exam don’t count toward level of service
Physicians, advanced practice registered nurses, and physician assistants won’t use history or exam to select what level of code to bill for office visits 99202-99215, as they did in the past. They need only document a medically appropriate history and exam. The history may be obtained by staff members and reviewed by the billing practitioner.
While specific history and exam requirements disappear for office visit codes, they remain for all other types of visits, selected on the basis of history, exam, and MDM, such as hospital services, nursing facility services, and home and domiciliary care. So, say goodbye to “all other systems reviewed and negative” in office notes, but keep it handy for those other E/M codes.
2. All time spent caring for the patient on a particular day counts
This includes all time spent on the day of service, including preparing to see the patient, seeing the patient, phone calls or other work done after the visit (if not billed with a care management or other CPT code), and documenting in the medical record. The AMA developed new guidelines for using time for office and other outpatient services. For codes 99202-99215, count all of the face-to-face and non–face-to-face time spent by the billing clinician on the day of the visit. Counseling does not need to be more than 50% of the total time.
Do not include any staff time or time spent on any days before or after the visit. This allows clinicians to capture the work when a significant amount of it takes place before or after the visit with the patient, and to bill for it on the day of the visit.
According to the 2021 CPT codebook, physician or other qualified health care professional time includes the following activities:
- Preparing to see the patient (e.g., review of tests).
- Obtaining and/or reviewing separately obtained history.
- Performing a medically appropriate examination and/or evaluation.
- Counseling and educating the patient/family/caregiver.
- Ordering medications, tests, or procedures.
- Referring and communicating with other health care professionals (when not separately reported).
- Documenting clinical information in the electronic or other health record.
- Independently interpreting results (not separately reported) and communicating results to the patient/family/caregiver.
- Care coordination (not separately reported).

3. Soon to be gone: ‘new to the examiner’ and ‘workup planned’
The current guidelines don’t differentiate between a new problem to the clinician or an established problem to the clinician. So it doesn’t matter whether you’re hearing about a particular problem for the first time or the fifth time. The new office and outpatient services guidelines define problems only as they relate to the patient. For example, when selecting a level of service, a chronic problem with a mild exacerbation is the same level whether it’s the primary care physician seeing the patient for the 10th time to help manage her diabetes or the endocrinologist seeing the patient for the first time.
In the current guidelines (1995 and 1997), additional weight is given in selecting the level of MDM for a problem that’s new to the examiner with a workup planned, yet when the diagnostic test couldn’t be completed at the visit. This concept is gone from element of number and complexity of new problems. Ordering diagnostic tests is part of the second element, the amount and/or complexity of data to be reviewed.
4. Different guidelines if you need a history from a parent or other source
The new guidelines recognize the additional work required by the clinician when the patient is unable to give a history or when the practitioner doesn’t find the history to be reliable.
For example, in the case of a baby or child who is unable to give a history, the parent counts as an “independent historian,” according to the new guidelines. Likewise, for a patient with dementia, the caregiver counts as a historian. Note, however, that the criteria is not met simply because the patient is accompanied by another person. The additional weight in selecting the level of service is based on the patient being unable to give a reliable history.
Bottom line: In cases where patients are unable to communicate clearly, physicians or other providers should document the necessity of getting a complete history and who provided it.
5. A new spin on social determinants of health (SDoH)
In the risk of morbidity and/or mortality element, conditions described as “social determinants of health” are considered moderate complexity. SDoH are social and environmental factors that affect a patient’s health and medical outcomes. These include homelessness, inability to afford medications, food insecurity, and occupational exposure to risk factors. These circumstances are reported with codes in categories Z55-Z65.
In the past, physicians often documented this information in their office notes but rarely added a diagnosis code that described the patient’s situation. The ICD-10-CM code set includes codes that describe these factors. Using them allows the practice to track patients who have increased needs, and it communicates to payers the complexity of caring for these patients.
6. Risks related to surgery are defined
The current guidelines assign different levels of risk to minor and major surgery. They also include differentiation for “minor surgery with no identified risk factors,” “minor surgery with identified risk factors,” “elective major surgery with no identified risk factors,” and “elective major surgery with identified risk factors.” The old guidelines didn’t state whether the risk factors pertained to the patient – such as smoking, heart disease, or high body mass index – or to the procedure itself.
The new guidelines specifically say that it’s both. In the risk column, “decision regarding minor surgery with identified patient or procedure risk factors” and “decision regarding elective major surgery without patient or procedure risk factors” are both considered moderate. “Decision regarding elective major surgery with identified patient or procedure risk factors” and “decision regarding emergency major surgery” are in the high complexity column for risk.
Keep in mind that two of three elements are required: the number and complexity of problems, amount of data, and morbidity/mortality risk. Risk of morbidity/mortality alone doesn’t count as the basis for selecting the code. Of course, when surgeons see this, they ask, “What major procedures don’t have identified risk factors?”
Note, too, that these new CPT guidelines do not define the terms “minor” and “major” surgery. For payment reasons related to the postop period, the Centers for Medicare & Medicaid Services defines minor surgery as a procedure with 0-10 global days and a major surgery as a procedure with 90 global days. However, there are many procedures with 0 global days (endoscopy, cardiac catheterization) that are not minor procedures. Hopefully, the AMA will clarify this in 2021.
What’s the take-away for clinicians?
There are sure to be shifts in coding patterns based on these new guidelines. Some specialties will find that not being able to select a service based on history and exam alone will lower the level of service for which they can bill. Some practices, on the other hand, will be able to code for more high-level visits, without the need for a complete review of systems or a comprehensive exam.
The biggest challenge will be for practices that provide services both in the hospital and in the office, because they’ll have to use both sets of guidelines, depending on which type of service they’re performing.
For more details on what’s coming your way beginning on New Year’s Day, you may want to read the 16-page AMA document .
A version of this article first appeared on Medscape.com.
Betsy Nicoletti, MS, a nationally recognized coding expert, will take your coding questions via email and provide guidance on how to code properly to maximize reimbursement. Have a question about coding? Send it to [email protected].
thanks to the American Medical Association.
The first major changes to the definitions for E/M services will be in effect as of Jan. 1, 2021, with all payers expected to adopt these new guidelines. In particular, the AMA has revised the definitions for E/M codes 99202-99215 in the Current Procedural Terminology (CPT) 2021 codebook. The existing guidelines were developed in 1995 and 1997 and remain in effect for all other E/M services determined by history, exam, and medical decision-making (MDM).
What do the new changes mean to you? In 2021, for new and established office and other outpatient services reported with codes 99202-99215, a clinician may select the code on the basis of time or MDM.
There are three elements in MDM, and two of three are required. These elements are the number and complexity of problems addressed, amount and/or complexity of data to be reviewed and analyzed, and risk of complications and/or morbidity or mortality of patient management.
Make sure you familiarize yourself with these six big changes. It may take a bit of time to integrate these new processes into your daily routine, but wrapping your head around them as soon as possible can help boost your bottom line:
1. History and exam don’t count toward level of service
Physicians, advanced practice registered nurses, and physician assistants won’t use history or exam to select what level of code to bill for office visits 99202-99215, as they did in the past. They need only document a medically appropriate history and exam. The history may be obtained by staff members and reviewed by the billing practitioner.
While specific history and exam requirements disappear for office visit codes, they remain for all other types of visits, selected on the basis of history, exam, and MDM, such as hospital services, nursing facility services, and home and domiciliary care. So, say goodbye to “all other systems reviewed and negative” in office notes, but keep it handy for those other E/M codes.
2. All time spent caring for the patient on a particular day counts
This includes all time spent on the day of service, including preparing to see the patient, seeing the patient, phone calls or other work done after the visit (if not billed with a care management or other CPT code), and documenting in the medical record. The AMA developed new guidelines for using time for office and other outpatient services. For codes 99202-99215, count all of the face-to-face and non–face-to-face time spent by the billing clinician on the day of the visit. Counseling does not need to be more than 50% of the total time.
Do not include any staff time or time spent on any days before or after the visit. This allows clinicians to capture the work when a significant amount of it takes place before or after the visit with the patient, and to bill for it on the day of the visit.
According to the 2021 CPT codebook, physician or other qualified health care professional time includes the following activities:
- Preparing to see the patient (e.g., review of tests).
- Obtaining and/or reviewing separately obtained history.
- Performing a medically appropriate examination and/or evaluation.
- Counseling and educating the patient/family/caregiver.
- Ordering medications, tests, or procedures.
- Referring and communicating with other health care professionals (when not separately reported).
- Documenting clinical information in the electronic or other health record.
- Independently interpreting results (not separately reported) and communicating results to the patient/family/caregiver.
- Care coordination (not separately reported).

3. Soon to be gone: ‘new to the examiner’ and ‘workup planned’
The current guidelines don’t differentiate between a new problem to the clinician or an established problem to the clinician. So it doesn’t matter whether you’re hearing about a particular problem for the first time or the fifth time. The new office and outpatient services guidelines define problems only as they relate to the patient. For example, when selecting a level of service, a chronic problem with a mild exacerbation is the same level whether it’s the primary care physician seeing the patient for the 10th time to help manage her diabetes or the endocrinologist seeing the patient for the first time.
In the current guidelines (1995 and 1997), additional weight is given in selecting the level of MDM for a problem that’s new to the examiner with a workup planned, yet when the diagnostic test couldn’t be completed at the visit. This concept is gone from element of number and complexity of new problems. Ordering diagnostic tests is part of the second element, the amount and/or complexity of data to be reviewed.
4. Different guidelines if you need a history from a parent or other source
The new guidelines recognize the additional work required by the clinician when the patient is unable to give a history or when the practitioner doesn’t find the history to be reliable.
For example, in the case of a baby or child who is unable to give a history, the parent counts as an “independent historian,” according to the new guidelines. Likewise, for a patient with dementia, the caregiver counts as a historian. Note, however, that the criteria is not met simply because the patient is accompanied by another person. The additional weight in selecting the level of service is based on the patient being unable to give a reliable history.
Bottom line: In cases where patients are unable to communicate clearly, physicians or other providers should document the necessity of getting a complete history and who provided it.
5. A new spin on social determinants of health (SDoH)
In the risk of morbidity and/or mortality element, conditions described as “social determinants of health” are considered moderate complexity. SDoH are social and environmental factors that affect a patient’s health and medical outcomes. These include homelessness, inability to afford medications, food insecurity, and occupational exposure to risk factors. These circumstances are reported with codes in categories Z55-Z65.
In the past, physicians often documented this information in their office notes but rarely added a diagnosis code that described the patient’s situation. The ICD-10-CM code set includes codes that describe these factors. Using them allows the practice to track patients who have increased needs, and it communicates to payers the complexity of caring for these patients.
6. Risks related to surgery are defined
The current guidelines assign different levels of risk to minor and major surgery. They also include differentiation for “minor surgery with no identified risk factors,” “minor surgery with identified risk factors,” “elective major surgery with no identified risk factors,” and “elective major surgery with identified risk factors.” The old guidelines didn’t state whether the risk factors pertained to the patient – such as smoking, heart disease, or high body mass index – or to the procedure itself.
The new guidelines specifically say that it’s both. In the risk column, “decision regarding minor surgery with identified patient or procedure risk factors” and “decision regarding elective major surgery without patient or procedure risk factors” are both considered moderate. “Decision regarding elective major surgery with identified patient or procedure risk factors” and “decision regarding emergency major surgery” are in the high complexity column for risk.
Keep in mind that two of three elements are required: the number and complexity of problems, amount of data, and morbidity/mortality risk. Risk of morbidity/mortality alone doesn’t count as the basis for selecting the code. Of course, when surgeons see this, they ask, “What major procedures don’t have identified risk factors?”
Note, too, that these new CPT guidelines do not define the terms “minor” and “major” surgery. For payment reasons related to the postop period, the Centers for Medicare & Medicaid Services defines minor surgery as a procedure with 0-10 global days and a major surgery as a procedure with 90 global days. However, there are many procedures with 0 global days (endoscopy, cardiac catheterization) that are not minor procedures. Hopefully, the AMA will clarify this in 2021.
What’s the take-away for clinicians?
There are sure to be shifts in coding patterns based on these new guidelines. Some specialties will find that not being able to select a service based on history and exam alone will lower the level of service for which they can bill. Some practices, on the other hand, will be able to code for more high-level visits, without the need for a complete review of systems or a comprehensive exam.
The biggest challenge will be for practices that provide services both in the hospital and in the office, because they’ll have to use both sets of guidelines, depending on which type of service they’re performing.
For more details on what’s coming your way beginning on New Year’s Day, you may want to read the 16-page AMA document .
A version of this article first appeared on Medscape.com.
COVID-related harm to HCWs must be tracked more rigorously: NAS panel
A panel of scientific experts is urging the nation to do more to track morbidity and mortality among health care workers (HCWs), given the large and disproportionate number who have been infected with or died from SARS-CoV-2.
The National Academies of Sciences, Engineering, and Medicine’s Standing Committee on Emerging Infectious Diseases and 21st Century Health Threats issued a 10-page “rapid expert consultation” on what is known about deaths and mental health problems among HCWs associated with the COVID-19 pandemic and how to protect workers.
“The absence of a uniform national framework and inconsistent requirements across states for collecting, recording, and reporting HCW mortality and morbidity data associated with COVID-19 impairs anyone’s ability to make comparisons, do combined analyses, or draw conclusions about the scale of the problem,” says the panel in the report.
Mental health, in particular, needs to be examined, it says. Although the data are still limited, the prevalence of burnout and suicide “points to a serious concern,” according to the report.
“As with mortality due to COVID-19, there are currently no national systems nor reporting standards for morbidity measures related to the pandemic, such as mental health status, provider well-being, and other psychological effects on HCWs,” the report says.
A more robust national system that collected data on circumstances and interventions that may raise or lower risk, as well as on where the infection occurred, “would support the adoption of effective mitigation strategies,” says the report. It would also facilitate epidemiologic studies on risk factors, such as face-to-face contact with COVID-19 patients and the availability and use of personal protective equipment (PPE). Studies could also examine the impact of institutional requirements for masking.
Studies have consistently shown that universal mask wearing and access to appropriate PPE support the physical safety and mental health of HCWs, says the report.
Track scale of crisis
The committee cited many gaps in the current system. The Occupational Safety and Health Administration, for instance, doesn’t count deaths from occupationally acquired infection. Many states don’t report COVID-19 deaths by profession. The Centers for Disease Control and Prevention (CDC) relies on case report forms from local health departments for all COVID-19 cases, which typically are lacking in specifics, such as occupation or job setting, says the committee’s report.
As of Nov. 3, the CDC had reported 786 deaths among HCWs that were attributable to COVID-19 – a far lower number than other sources have reported.
The committee notes that much could be done immediately. A National Academy of Medicine panel on clinician well-being and resilience in August recommended that the CDC establish a national epidemiologic tracking program to measure HCWs’ well-being, assess the acute and long-term effects of COVID-19 on those workers, and report on the outcomes of interventions.
Such a program “is needed to comprehensively acknowledge the scale of the COVID-19 crisis and protect the health care workforce that is already stretched to the breaking point in many locations,” the committee says in its report.
A version of this article originally appeared on Medscape.com.
A panel of scientific experts is urging the nation to do more to track morbidity and mortality among health care workers (HCWs), given the large and disproportionate number who have been infected with or died from SARS-CoV-2.
The National Academies of Sciences, Engineering, and Medicine’s Standing Committee on Emerging Infectious Diseases and 21st Century Health Threats issued a 10-page “rapid expert consultation” on what is known about deaths and mental health problems among HCWs associated with the COVID-19 pandemic and how to protect workers.
“The absence of a uniform national framework and inconsistent requirements across states for collecting, recording, and reporting HCW mortality and morbidity data associated with COVID-19 impairs anyone’s ability to make comparisons, do combined analyses, or draw conclusions about the scale of the problem,” says the panel in the report.
Mental health, in particular, needs to be examined, it says. Although the data are still limited, the prevalence of burnout and suicide “points to a serious concern,” according to the report.
“As with mortality due to COVID-19, there are currently no national systems nor reporting standards for morbidity measures related to the pandemic, such as mental health status, provider well-being, and other psychological effects on HCWs,” the report says.
A more robust national system that collected data on circumstances and interventions that may raise or lower risk, as well as on where the infection occurred, “would support the adoption of effective mitigation strategies,” says the report. It would also facilitate epidemiologic studies on risk factors, such as face-to-face contact with COVID-19 patients and the availability and use of personal protective equipment (PPE). Studies could also examine the impact of institutional requirements for masking.
Studies have consistently shown that universal mask wearing and access to appropriate PPE support the physical safety and mental health of HCWs, says the report.
Track scale of crisis
The committee cited many gaps in the current system. The Occupational Safety and Health Administration, for instance, doesn’t count deaths from occupationally acquired infection. Many states don’t report COVID-19 deaths by profession. The Centers for Disease Control and Prevention (CDC) relies on case report forms from local health departments for all COVID-19 cases, which typically are lacking in specifics, such as occupation or job setting, says the committee’s report.
As of Nov. 3, the CDC had reported 786 deaths among HCWs that were attributable to COVID-19 – a far lower number than other sources have reported.
The committee notes that much could be done immediately. A National Academy of Medicine panel on clinician well-being and resilience in August recommended that the CDC establish a national epidemiologic tracking program to measure HCWs’ well-being, assess the acute and long-term effects of COVID-19 on those workers, and report on the outcomes of interventions.
Such a program “is needed to comprehensively acknowledge the scale of the COVID-19 crisis and protect the health care workforce that is already stretched to the breaking point in many locations,” the committee says in its report.
A version of this article originally appeared on Medscape.com.
A panel of scientific experts is urging the nation to do more to track morbidity and mortality among health care workers (HCWs), given the large and disproportionate number who have been infected with or died from SARS-CoV-2.
The National Academies of Sciences, Engineering, and Medicine’s Standing Committee on Emerging Infectious Diseases and 21st Century Health Threats issued a 10-page “rapid expert consultation” on what is known about deaths and mental health problems among HCWs associated with the COVID-19 pandemic and how to protect workers.
“The absence of a uniform national framework and inconsistent requirements across states for collecting, recording, and reporting HCW mortality and morbidity data associated with COVID-19 impairs anyone’s ability to make comparisons, do combined analyses, or draw conclusions about the scale of the problem,” says the panel in the report.
Mental health, in particular, needs to be examined, it says. Although the data are still limited, the prevalence of burnout and suicide “points to a serious concern,” according to the report.
“As with mortality due to COVID-19, there are currently no national systems nor reporting standards for morbidity measures related to the pandemic, such as mental health status, provider well-being, and other psychological effects on HCWs,” the report says.
A more robust national system that collected data on circumstances and interventions that may raise or lower risk, as well as on where the infection occurred, “would support the adoption of effective mitigation strategies,” says the report. It would also facilitate epidemiologic studies on risk factors, such as face-to-face contact with COVID-19 patients and the availability and use of personal protective equipment (PPE). Studies could also examine the impact of institutional requirements for masking.
Studies have consistently shown that universal mask wearing and access to appropriate PPE support the physical safety and mental health of HCWs, says the report.
Track scale of crisis
The committee cited many gaps in the current system. The Occupational Safety and Health Administration, for instance, doesn’t count deaths from occupationally acquired infection. Many states don’t report COVID-19 deaths by profession. The Centers for Disease Control and Prevention (CDC) relies on case report forms from local health departments for all COVID-19 cases, which typically are lacking in specifics, such as occupation or job setting, says the committee’s report.
As of Nov. 3, the CDC had reported 786 deaths among HCWs that were attributable to COVID-19 – a far lower number than other sources have reported.
The committee notes that much could be done immediately. A National Academy of Medicine panel on clinician well-being and resilience in August recommended that the CDC establish a national epidemiologic tracking program to measure HCWs’ well-being, assess the acute and long-term effects of COVID-19 on those workers, and report on the outcomes of interventions.
Such a program “is needed to comprehensively acknowledge the scale of the COVID-19 crisis and protect the health care workforce that is already stretched to the breaking point in many locations,” the committee says in its report.
A version of this article originally appeared on Medscape.com.
Coronavirus has infected over 2% of U.S. children
After last week’s ever-so-slightly positive news, the COVID-19 numbers in children have gone back to their old ways.
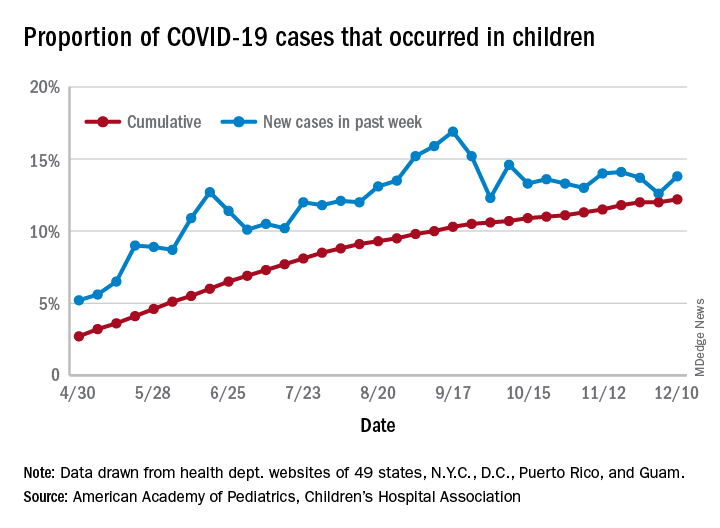
For the week ending Dec. 10, there were 178,823 new COVID-19 cases reported in U.S. children, the highest weekly total yet during the pandemic. The number of new cases had dropped the week before after setting a new high of almost 154,000 during the last full week of November, according to a report from the American Academy of Pediatrics and the Children’s Hospital Association.
A new weekly high has been seen in 9 of the last 10 weeks, during which time the weekly total of child cases has gone from just over 40,000 (week ending Oct. 8) to almost 179,000, the two organizations said.
and that 2.1% of all children (2,179 per 100,000) in the United States have been infected with the coronavirus, the AAP and CHA said in their weekly report, which includes health department data from 49 states (New York does not report age distribution), the District of Columbia, New York City, Puerto Rico, and Guam.
The cumulative proportion of 12.2% has been exceeded in 27 states, as well as Puerto Rico and Guam, with the highest coming in Wyoming (21.3%), South Carolina (18.1%), and Tennessee (18.1%) and the lowest in Florida (6.7%, but the state uses an age range of 0-14 years) and New Jersey (7.6%), the AAP/CHA data show.
In a separate statement, AAP president Sally Goza, MD, welcomed the approval of the Pfizer-BioNTech COVID-19 vaccine but noted that the “virus is at unprecedented levels in nearly every community in the U.S., and in many areas, our health care system is terribly overburdened. The vaccine will not solve this overnight. I urge everyone to continue to practice social distancing, and wear masks or cloth face coverings, and get a flu shot, so we can protect the people we care about.”
Dr. Goza continued: “We applaud Pfizer-BioNTech for including children ages 12 through 17 in their clinical trials and we look forward to learning more about the data from children aged 12-15. We also want to acknowledge the discussion during the committee meeting on including 16- to 17-year-olds in the EUA [emergency-use authorization]. We believe that discussion underscores the need to keep expanding these trials to the pediatric population so we can collect robust data on this age group.”
[email protected]
After last week’s ever-so-slightly positive news, the COVID-19 numbers in children have gone back to their old ways.

For the week ending Dec. 10, there were 178,823 new COVID-19 cases reported in U.S. children, the highest weekly total yet during the pandemic. The number of new cases had dropped the week before after setting a new high of almost 154,000 during the last full week of November, according to a report from the American Academy of Pediatrics and the Children’s Hospital Association.
A new weekly high has been seen in 9 of the last 10 weeks, during which time the weekly total of child cases has gone from just over 40,000 (week ending Oct. 8) to almost 179,000, the two organizations said.
and that 2.1% of all children (2,179 per 100,000) in the United States have been infected with the coronavirus, the AAP and CHA said in their weekly report, which includes health department data from 49 states (New York does not report age distribution), the District of Columbia, New York City, Puerto Rico, and Guam.
The cumulative proportion of 12.2% has been exceeded in 27 states, as well as Puerto Rico and Guam, with the highest coming in Wyoming (21.3%), South Carolina (18.1%), and Tennessee (18.1%) and the lowest in Florida (6.7%, but the state uses an age range of 0-14 years) and New Jersey (7.6%), the AAP/CHA data show.
In a separate statement, AAP president Sally Goza, MD, welcomed the approval of the Pfizer-BioNTech COVID-19 vaccine but noted that the “virus is at unprecedented levels in nearly every community in the U.S., and in many areas, our health care system is terribly overburdened. The vaccine will not solve this overnight. I urge everyone to continue to practice social distancing, and wear masks or cloth face coverings, and get a flu shot, so we can protect the people we care about.”
Dr. Goza continued: “We applaud Pfizer-BioNTech for including children ages 12 through 17 in their clinical trials and we look forward to learning more about the data from children aged 12-15. We also want to acknowledge the discussion during the committee meeting on including 16- to 17-year-olds in the EUA [emergency-use authorization]. We believe that discussion underscores the need to keep expanding these trials to the pediatric population so we can collect robust data on this age group.”
[email protected]
After last week’s ever-so-slightly positive news, the COVID-19 numbers in children have gone back to their old ways.

For the week ending Dec. 10, there were 178,823 new COVID-19 cases reported in U.S. children, the highest weekly total yet during the pandemic. The number of new cases had dropped the week before after setting a new high of almost 154,000 during the last full week of November, according to a report from the American Academy of Pediatrics and the Children’s Hospital Association.
A new weekly high has been seen in 9 of the last 10 weeks, during which time the weekly total of child cases has gone from just over 40,000 (week ending Oct. 8) to almost 179,000, the two organizations said.
and that 2.1% of all children (2,179 per 100,000) in the United States have been infected with the coronavirus, the AAP and CHA said in their weekly report, which includes health department data from 49 states (New York does not report age distribution), the District of Columbia, New York City, Puerto Rico, and Guam.
The cumulative proportion of 12.2% has been exceeded in 27 states, as well as Puerto Rico and Guam, with the highest coming in Wyoming (21.3%), South Carolina (18.1%), and Tennessee (18.1%) and the lowest in Florida (6.7%, but the state uses an age range of 0-14 years) and New Jersey (7.6%), the AAP/CHA data show.
In a separate statement, AAP president Sally Goza, MD, welcomed the approval of the Pfizer-BioNTech COVID-19 vaccine but noted that the “virus is at unprecedented levels in nearly every community in the U.S., and in many areas, our health care system is terribly overburdened. The vaccine will not solve this overnight. I urge everyone to continue to practice social distancing, and wear masks or cloth face coverings, and get a flu shot, so we can protect the people we care about.”
Dr. Goza continued: “We applaud Pfizer-BioNTech for including children ages 12 through 17 in their clinical trials and we look forward to learning more about the data from children aged 12-15. We also want to acknowledge the discussion during the committee meeting on including 16- to 17-year-olds in the EUA [emergency-use authorization]. We believe that discussion underscores the need to keep expanding these trials to the pediatric population so we can collect robust data on this age group.”
[email protected]
Proposed HIPAA overhaul to ease access to patient health info
The Department of Health & Human Services is proposing an overhaul of HIPAA that will make it easier to access patients’ personal health information, including the health records of patients with mental illness. The proposal would also do away with the requirement that all patients sign a notice of privacy practices.
The changes are contained in a 357-page proposed rule, which was unveiled by federal officials Dec. 10. Roger Severino, director of HHS’ Office for Civil Rights, said in a briefing that the sweeping proposal would empower patients, reduce the administrative burden for health care providers, and pave the way to better-coordinated care.
HHS estimated that the rule could save $3.2 billion over 5 years, but it’s not clear how much of that would accrue to clinical practices.
The most obvious cost-saving aspect for medical and dental practices is the proposal that practitioners would no longer have to provide and collect signed notifications of privacy practices.
“This has been a tremendous waste of time and effort and has caused massive confusion,” said Mr. Severino. He said some patients thought they were waiving privacy rights and that, in some cases, physicians refused to administer care unless patients signed the notices. “That was never the intent.”
Requiring that patients sign the form and that practices keep copies for 6 years is an “unnecessary burden,” said Mr. Severino. “We’ve lost whole forests from this regulation.”
Under the new proposal, health care providers would merely have to let patients know where to find their privacy policies.
Sharing mental health info
The rule would also ease the standard for sharing information about a patient who is in a mental health crisis, such as an exacerbation of a serious mental illness or a crisis related to a substance use disorder, including an overdose.
Currently, clinicians can choose to disclose protected health information – to a family member, a caregiver, a law enforcement official, a doctor, or an insurer – if they believe that doing so is advisable in their “professional judgment.” The rule proposes to ease that to a “good faith” belief that a disclosure would be in the best interest of the patient. In both instances, the patient can still object and block the disclosure.
As an example, HHS said that, in the case of a young adult who had experienced an overdose of opioids, a licensed health care professional could make the determination to “disclose relevant information to a parent who is involved in the patient’s treatment and who the young adult would expect, based on their relationship, to participate in or be involved with the patient’s recovery from the overdose.”
HHS is also proposing to let clinicians disclose information in cases in which an individual might be a threat to himself or others, provided the harm is “serious and reasonably foreseeable.”
Currently, information can only be disclosed if it appears there is a “serious and imminent” threat to health or safety. If an individual experienced suicidal ideation, for instance, a health care professional could notify family that the individual is at risk.
Fast, no-cost access
The rule also aims to make it easier for patients to get access to their own health care information quickly – within 15 days of a request – instead of the 30 days currently allowed, and sometimes at no cost.
The 30-day time frame is “a relic of a pre-Internet age that should be dispensed with,” said Mr. Severino.
Patients can also request that a treating physician get his or her records from a clinician who had previously treated the individual. The request would be fulfilled within 15 days, although extensions might be possible.
“That takes away the burden of coordination from the patient and puts it on those parties that are responsible for the actual provision of care and that are better positioned to do that coordination,” Mr. Severino said.
Health care professionals will also have to share with patients a fee schedule for records requests. However, if records are shared through a patient portal with view, download, and transmit capabilities, the provider can’t charge the patient for the time it took to upload the information into the system.
“We do not believe a patient’s personal medical record should be profit centers for providers,” Mr. Severino said.
Patients will be allowed to take photos with a smartphone of personal health information – such as an x-ray or sonogram – while receiving care.
The rule is open for public comment until mid-February. After that, it will become final in 180 days. The agency said it would not begin enforcement until 240 days after the final rule was published.
A version of this article originally appeared on Medscape.com.
The Department of Health & Human Services is proposing an overhaul of HIPAA that will make it easier to access patients’ personal health information, including the health records of patients with mental illness. The proposal would also do away with the requirement that all patients sign a notice of privacy practices.
The changes are contained in a 357-page proposed rule, which was unveiled by federal officials Dec. 10. Roger Severino, director of HHS’ Office for Civil Rights, said in a briefing that the sweeping proposal would empower patients, reduce the administrative burden for health care providers, and pave the way to better-coordinated care.
HHS estimated that the rule could save $3.2 billion over 5 years, but it’s not clear how much of that would accrue to clinical practices.
The most obvious cost-saving aspect for medical and dental practices is the proposal that practitioners would no longer have to provide and collect signed notifications of privacy practices.
“This has been a tremendous waste of time and effort and has caused massive confusion,” said Mr. Severino. He said some patients thought they were waiving privacy rights and that, in some cases, physicians refused to administer care unless patients signed the notices. “That was never the intent.”
Requiring that patients sign the form and that practices keep copies for 6 years is an “unnecessary burden,” said Mr. Severino. “We’ve lost whole forests from this regulation.”
Under the new proposal, health care providers would merely have to let patients know where to find their privacy policies.
Sharing mental health info
The rule would also ease the standard for sharing information about a patient who is in a mental health crisis, such as an exacerbation of a serious mental illness or a crisis related to a substance use disorder, including an overdose.
Currently, clinicians can choose to disclose protected health information – to a family member, a caregiver, a law enforcement official, a doctor, or an insurer – if they believe that doing so is advisable in their “professional judgment.” The rule proposes to ease that to a “good faith” belief that a disclosure would be in the best interest of the patient. In both instances, the patient can still object and block the disclosure.
As an example, HHS said that, in the case of a young adult who had experienced an overdose of opioids, a licensed health care professional could make the determination to “disclose relevant information to a parent who is involved in the patient’s treatment and who the young adult would expect, based on their relationship, to participate in or be involved with the patient’s recovery from the overdose.”
HHS is also proposing to let clinicians disclose information in cases in which an individual might be a threat to himself or others, provided the harm is “serious and reasonably foreseeable.”
Currently, information can only be disclosed if it appears there is a “serious and imminent” threat to health or safety. If an individual experienced suicidal ideation, for instance, a health care professional could notify family that the individual is at risk.
Fast, no-cost access
The rule also aims to make it easier for patients to get access to their own health care information quickly – within 15 days of a request – instead of the 30 days currently allowed, and sometimes at no cost.
The 30-day time frame is “a relic of a pre-Internet age that should be dispensed with,” said Mr. Severino.
Patients can also request that a treating physician get his or her records from a clinician who had previously treated the individual. The request would be fulfilled within 15 days, although extensions might be possible.
“That takes away the burden of coordination from the patient and puts it on those parties that are responsible for the actual provision of care and that are better positioned to do that coordination,” Mr. Severino said.
Health care professionals will also have to share with patients a fee schedule for records requests. However, if records are shared through a patient portal with view, download, and transmit capabilities, the provider can’t charge the patient for the time it took to upload the information into the system.
“We do not believe a patient’s personal medical record should be profit centers for providers,” Mr. Severino said.
Patients will be allowed to take photos with a smartphone of personal health information – such as an x-ray or sonogram – while receiving care.
The rule is open for public comment until mid-February. After that, it will become final in 180 days. The agency said it would not begin enforcement until 240 days after the final rule was published.
A version of this article originally appeared on Medscape.com.
The Department of Health & Human Services is proposing an overhaul of HIPAA that will make it easier to access patients’ personal health information, including the health records of patients with mental illness. The proposal would also do away with the requirement that all patients sign a notice of privacy practices.
The changes are contained in a 357-page proposed rule, which was unveiled by federal officials Dec. 10. Roger Severino, director of HHS’ Office for Civil Rights, said in a briefing that the sweeping proposal would empower patients, reduce the administrative burden for health care providers, and pave the way to better-coordinated care.
HHS estimated that the rule could save $3.2 billion over 5 years, but it’s not clear how much of that would accrue to clinical practices.
The most obvious cost-saving aspect for medical and dental practices is the proposal that practitioners would no longer have to provide and collect signed notifications of privacy practices.
“This has been a tremendous waste of time and effort and has caused massive confusion,” said Mr. Severino. He said some patients thought they were waiving privacy rights and that, in some cases, physicians refused to administer care unless patients signed the notices. “That was never the intent.”
Requiring that patients sign the form and that practices keep copies for 6 years is an “unnecessary burden,” said Mr. Severino. “We’ve lost whole forests from this regulation.”
Under the new proposal, health care providers would merely have to let patients know where to find their privacy policies.
Sharing mental health info
The rule would also ease the standard for sharing information about a patient who is in a mental health crisis, such as an exacerbation of a serious mental illness or a crisis related to a substance use disorder, including an overdose.
Currently, clinicians can choose to disclose protected health information – to a family member, a caregiver, a law enforcement official, a doctor, or an insurer – if they believe that doing so is advisable in their “professional judgment.” The rule proposes to ease that to a “good faith” belief that a disclosure would be in the best interest of the patient. In both instances, the patient can still object and block the disclosure.
As an example, HHS said that, in the case of a young adult who had experienced an overdose of opioids, a licensed health care professional could make the determination to “disclose relevant information to a parent who is involved in the patient’s treatment and who the young adult would expect, based on their relationship, to participate in or be involved with the patient’s recovery from the overdose.”
HHS is also proposing to let clinicians disclose information in cases in which an individual might be a threat to himself or others, provided the harm is “serious and reasonably foreseeable.”
Currently, information can only be disclosed if it appears there is a “serious and imminent” threat to health or safety. If an individual experienced suicidal ideation, for instance, a health care professional could notify family that the individual is at risk.
Fast, no-cost access
The rule also aims to make it easier for patients to get access to their own health care information quickly – within 15 days of a request – instead of the 30 days currently allowed, and sometimes at no cost.
The 30-day time frame is “a relic of a pre-Internet age that should be dispensed with,” said Mr. Severino.
Patients can also request that a treating physician get his or her records from a clinician who had previously treated the individual. The request would be fulfilled within 15 days, although extensions might be possible.
“That takes away the burden of coordination from the patient and puts it on those parties that are responsible for the actual provision of care and that are better positioned to do that coordination,” Mr. Severino said.
Health care professionals will also have to share with patients a fee schedule for records requests. However, if records are shared through a patient portal with view, download, and transmit capabilities, the provider can’t charge the patient for the time it took to upload the information into the system.
“We do not believe a patient’s personal medical record should be profit centers for providers,” Mr. Severino said.
Patients will be allowed to take photos with a smartphone of personal health information – such as an x-ray or sonogram – while receiving care.
The rule is open for public comment until mid-February. After that, it will become final in 180 days. The agency said it would not begin enforcement until 240 days after the final rule was published.
A version of this article originally appeared on Medscape.com.
Medicare payments could get tougher for docs
More than 40 value-based payment models – from direct contracting to bundled payments – have been introduced into the Medicare program in the past 10 years, with the goal of improving care while lowering costs. Hopes were high that they would be successful.
Physicians could suffer a huge blow to their income.
Many of the value-based care models simply did not work as expected, said Seema Verma, head of the Centers for Medicare & Medicaid Services, at a recent HLTH Conference. “They are not producing the types of savings the taxpayers deserve,” Ms. Verma said.
The Medicare Payment Advisory Commission (MedPac) concluded that, while dozens of payment models were tested, most failed to generate net savings for Medicare. Even the most successful of the models produced only modest savings. MedPac elaborated: “The track record raises the question of whether changes to particular models or CMMI’s [Center for Medicare & Medicaid Innovation’s] broader strategies might be warranted.”
What will happen now, as government officials admit that their value-based programs haven’t worked? The value-based programs could become more stringent. Here’s what physicians will have to contend with.
More risk. Experts agree that risk – financial risk – will be a component of future programs. Two-sided risk is likely to be the norm. This means that both parties – the provider and the insurer – are at financial risk for the patients covered by the program.
For example, a plan with 50,000 beneficiary patients would estimate the cost of caring for those patients on the basis of multiple variables. If the actual cost is lower than anticipated, both parties share in the savings. However, both share in the loss if the cost of caring for their patient population exceeds expectations.
This may compel physicians to enhance efficiency and potentially limit the services provided to patients. Typically, however, the strategy is to make efforts to prevent services like ED visits and admissions by focusing on health maintenance.
In contrast to most current value-based models, which feature little to no downside risk for physicians, double-sided risk means physicians could lose money. The loss may incorporate a cap – 5%, for example – but programs may differ. Experts concur that double-sided risk will be a hallmark of future programs.
Better data. The majority of health care services are rendered via fee-for-service: Patients receive services and physicians are paid, yet little or no information about outcomes is exchanged between insurers and physicians.
Penny Noyes, president of Health Business Navigators and contract negotiator for physicians, is not a fan of the current crop of value-based programs and feels that data transparency is positive. Sound metrics can lead to improvement, she said, adding: “It’s not money that drives physicians to make decisions; it’s what’s in the best interest of their patients and their patients’ long-term care.”
Value-based programs can work but only if applicable data are developed and given to physicians so that they can better understand their current performance and how to improve.
Mandated participation. Participation in value-based programs has been voluntary, but that may have skewed the results, which were better than what typical practice would have shown. Acknowledging this may lead CMS to call for mandated participation as a component of future programs. Physicians may be brought into programs, if only to determine whether the models really work. To date, participation in the programs has been voluntary, but that may change in the future.
Innovation. The private insurance market may end up as a key player. Over the past 6 months, health insurers have either consolidated partnerships with telemedicine companies to provide no-cost care to beneficiaries or have launched their own initiatives.
Others are focused on bringing together patients and providers operating outside of the traditional health care system, such as Aetna’s merger with CVS which now offers retail-based acute care (MinuteClinic) and chronic care (HealthHUB). Still other payers are gambling with physician practice ownership, as in the case of United Healthcare’s OptumHealth, which now boasts around 50,000 physicians throughout the country.
New practice models are emerging in private practices as well. Physicians are embracing remote care, proactively managing care transitions, and seeking out more methods to keep patients healthy and at home.
Not much was expected from value-based plans
Many are not surprised that the value-based models did not produce impressive results. Ms. Noyes doubted that positive outcomes will be achieved for physicians in comparison with what could have been attained under fee-for-service arrangements with lower administrative costs.
While the Affordable Care Act attempted to encourage alternative reimbursement, it limits the maximum medical loss ratio (MLR) a payer could achieve. For many plans, that maximum was 85%. Simply put, at least $0.85 of each premium collected had to be paid in claims; the remaining $0.15 went to margin, claims, and other administrative costs. A payer with an 82% MLR then would have to rebate the 3% difference to enrollees.
But that’s not what occurred, according to Ms. Noyes. Because value-based payments to providers are considered a claims expense, an MLR ratio of 82% allowed the payer to distribute the 3% difference to providers as value-based payments. Ms. Noyes said: “That may sound good for the provider, but the result was essentially a freeze on the provider’s fee-for-service reimbursement with the prospect of getting value-based payments like ‘shared savings.’
“When the providers tried to increase their base fee-for-service rates just to match inflation, payers often advised that any future raises had to be earned through value-based programs,” Ms. Noyes added. The value-based formulas confuse providers because payments are often made for periods as far back as 18 months, and providers do not have data systems to reconcile their payer report cards retrospectively. The result is that providers tended to accept whatever amount the payer distributed.
Executives at Lumeris, a company that helps health systems participate successfully in value-based care, see potential in a newer approach to alternative payments, such as CMS’ Direct Contracting initiative. This voluntary payment model offers options tailored to several types of organizations that aim to reduce costs while preserving or enhancing the quality of care for Medicare fee-for-service beneficiaries.
Jeff Smith, chief commercial officer for population health at Lumeris, explained that the Direct Contracting initiative can provide physicians with a more attractive option than prior value-based models because it adjusts for the complexity and fragility of patients with complex and chronic conditions. By allowing providers to participate in the savings generated, the initiative stands in stark contrast to what Mr. Smith described as the “shared savings to nothingness” experienced by providers in earlier-stage alternative payment models.
Physicians engaged with value-based programs like Direct Contracting are investing in nurses to aid with initiatives regarding health promotion and transitions of care. When a patient is discharged, for example, the nurse contacts the patient to discuss medications, schedule follow-up appointments, and so forth – tasks typically left to the patient (or caregiver) to navigate in the traditional system.
The initiative recognizes the importance of managing high-risk patients, those whom physicians identify as having an extraordinary number of ED visits and admissions. These patients, as well as so-called “rising-risk” patients, are targeted by nurses who proactively communicate with patients (and caregivers) to address patient’s needs, including social determinants of health.
Physicians who have a large load of patients in value-based programs are hiring social workers, pharmacists, and behavioral health experts to help. Of course, these personnel are costly, but that’s what the value-based programs aim to reimburse.
Still, the road ahead to value based is rocky and may not gain momentum for some time. Johns Hopkins University’s Doug Hough, PhD, an economist, recounts a government research study that sought to assess the university’s health system participation in a value-based payment program. While there were positive impacts on the program’s target population, Hough and his team discovered that the returns achieved by the optional model didn’t justify the health system’s financial support for it. The increasingly indebted health system ultimately decided to drop the optional program.
Dr. Hough indicated that the health system – Johns Hopkins Medicine – likely would have continued its support for the program had the government at least allowed it to break even. Although the payment program under study was a 3-year project, the bigger challenge, declared Dr. Hough, is that “we can’t turn an aircraft carrier that quickly.”
“Three years won’t show whether value-based care is really working,” Dr. Hough said.
Robert Zipper, MD, a hospitalist and senior policy advisor for Sound Physicians, a company that works to improve outcomes in acute care, agreed with Dr. Hough that performance tends to improve with time. Yet, Dr. Zipper doesn’t see much change in the near term, because “after all, there is nothing to replace them [the programs].”
The problem gets even stickier for private payers because patients may be on an insurance panel for as little as a year or 2. Thanks to this rapid churn of beneficiaries, even the best-designed value-based program will have little time to prove its worth.
Dr. Zipper is among the many who don’t expect significant changes in the near term, asserting that “President Biden will want to get a few policy wins first, and health care is not the easiest place to start.”
But it’s likely that payers and others will want to see more emphasis on value-based programs despite these programs’ possible value to patients, physicians, and health systems alike.
A version of this article originally appeared on Medscape.com.
More than 40 value-based payment models – from direct contracting to bundled payments – have been introduced into the Medicare program in the past 10 years, with the goal of improving care while lowering costs. Hopes were high that they would be successful.
Physicians could suffer a huge blow to their income.
Many of the value-based care models simply did not work as expected, said Seema Verma, head of the Centers for Medicare & Medicaid Services, at a recent HLTH Conference. “They are not producing the types of savings the taxpayers deserve,” Ms. Verma said.
The Medicare Payment Advisory Commission (MedPac) concluded that, while dozens of payment models were tested, most failed to generate net savings for Medicare. Even the most successful of the models produced only modest savings. MedPac elaborated: “The track record raises the question of whether changes to particular models or CMMI’s [Center for Medicare & Medicaid Innovation’s] broader strategies might be warranted.”
What will happen now, as government officials admit that their value-based programs haven’t worked? The value-based programs could become more stringent. Here’s what physicians will have to contend with.
More risk. Experts agree that risk – financial risk – will be a component of future programs. Two-sided risk is likely to be the norm. This means that both parties – the provider and the insurer – are at financial risk for the patients covered by the program.
For example, a plan with 50,000 beneficiary patients would estimate the cost of caring for those patients on the basis of multiple variables. If the actual cost is lower than anticipated, both parties share in the savings. However, both share in the loss if the cost of caring for their patient population exceeds expectations.
This may compel physicians to enhance efficiency and potentially limit the services provided to patients. Typically, however, the strategy is to make efforts to prevent services like ED visits and admissions by focusing on health maintenance.
In contrast to most current value-based models, which feature little to no downside risk for physicians, double-sided risk means physicians could lose money. The loss may incorporate a cap – 5%, for example – but programs may differ. Experts concur that double-sided risk will be a hallmark of future programs.
Better data. The majority of health care services are rendered via fee-for-service: Patients receive services and physicians are paid, yet little or no information about outcomes is exchanged between insurers and physicians.
Penny Noyes, president of Health Business Navigators and contract negotiator for physicians, is not a fan of the current crop of value-based programs and feels that data transparency is positive. Sound metrics can lead to improvement, she said, adding: “It’s not money that drives physicians to make decisions; it’s what’s in the best interest of their patients and their patients’ long-term care.”
Value-based programs can work but only if applicable data are developed and given to physicians so that they can better understand their current performance and how to improve.
Mandated participation. Participation in value-based programs has been voluntary, but that may have skewed the results, which were better than what typical practice would have shown. Acknowledging this may lead CMS to call for mandated participation as a component of future programs. Physicians may be brought into programs, if only to determine whether the models really work. To date, participation in the programs has been voluntary, but that may change in the future.
Innovation. The private insurance market may end up as a key player. Over the past 6 months, health insurers have either consolidated partnerships with telemedicine companies to provide no-cost care to beneficiaries or have launched their own initiatives.
Others are focused on bringing together patients and providers operating outside of the traditional health care system, such as Aetna’s merger with CVS which now offers retail-based acute care (MinuteClinic) and chronic care (HealthHUB). Still other payers are gambling with physician practice ownership, as in the case of United Healthcare’s OptumHealth, which now boasts around 50,000 physicians throughout the country.
New practice models are emerging in private practices as well. Physicians are embracing remote care, proactively managing care transitions, and seeking out more methods to keep patients healthy and at home.
Not much was expected from value-based plans
Many are not surprised that the value-based models did not produce impressive results. Ms. Noyes doubted that positive outcomes will be achieved for physicians in comparison with what could have been attained under fee-for-service arrangements with lower administrative costs.
While the Affordable Care Act attempted to encourage alternative reimbursement, it limits the maximum medical loss ratio (MLR) a payer could achieve. For many plans, that maximum was 85%. Simply put, at least $0.85 of each premium collected had to be paid in claims; the remaining $0.15 went to margin, claims, and other administrative costs. A payer with an 82% MLR then would have to rebate the 3% difference to enrollees.
But that’s not what occurred, according to Ms. Noyes. Because value-based payments to providers are considered a claims expense, an MLR ratio of 82% allowed the payer to distribute the 3% difference to providers as value-based payments. Ms. Noyes said: “That may sound good for the provider, but the result was essentially a freeze on the provider’s fee-for-service reimbursement with the prospect of getting value-based payments like ‘shared savings.’
“When the providers tried to increase their base fee-for-service rates just to match inflation, payers often advised that any future raises had to be earned through value-based programs,” Ms. Noyes added. The value-based formulas confuse providers because payments are often made for periods as far back as 18 months, and providers do not have data systems to reconcile their payer report cards retrospectively. The result is that providers tended to accept whatever amount the payer distributed.
Executives at Lumeris, a company that helps health systems participate successfully in value-based care, see potential in a newer approach to alternative payments, such as CMS’ Direct Contracting initiative. This voluntary payment model offers options tailored to several types of organizations that aim to reduce costs while preserving or enhancing the quality of care for Medicare fee-for-service beneficiaries.
Jeff Smith, chief commercial officer for population health at Lumeris, explained that the Direct Contracting initiative can provide physicians with a more attractive option than prior value-based models because it adjusts for the complexity and fragility of patients with complex and chronic conditions. By allowing providers to participate in the savings generated, the initiative stands in stark contrast to what Mr. Smith described as the “shared savings to nothingness” experienced by providers in earlier-stage alternative payment models.
Physicians engaged with value-based programs like Direct Contracting are investing in nurses to aid with initiatives regarding health promotion and transitions of care. When a patient is discharged, for example, the nurse contacts the patient to discuss medications, schedule follow-up appointments, and so forth – tasks typically left to the patient (or caregiver) to navigate in the traditional system.
The initiative recognizes the importance of managing high-risk patients, those whom physicians identify as having an extraordinary number of ED visits and admissions. These patients, as well as so-called “rising-risk” patients, are targeted by nurses who proactively communicate with patients (and caregivers) to address patient’s needs, including social determinants of health.
Physicians who have a large load of patients in value-based programs are hiring social workers, pharmacists, and behavioral health experts to help. Of course, these personnel are costly, but that’s what the value-based programs aim to reimburse.
Still, the road ahead to value based is rocky and may not gain momentum for some time. Johns Hopkins University’s Doug Hough, PhD, an economist, recounts a government research study that sought to assess the university’s health system participation in a value-based payment program. While there were positive impacts on the program’s target population, Hough and his team discovered that the returns achieved by the optional model didn’t justify the health system’s financial support for it. The increasingly indebted health system ultimately decided to drop the optional program.
Dr. Hough indicated that the health system – Johns Hopkins Medicine – likely would have continued its support for the program had the government at least allowed it to break even. Although the payment program under study was a 3-year project, the bigger challenge, declared Dr. Hough, is that “we can’t turn an aircraft carrier that quickly.”
“Three years won’t show whether value-based care is really working,” Dr. Hough said.
Robert Zipper, MD, a hospitalist and senior policy advisor for Sound Physicians, a company that works to improve outcomes in acute care, agreed with Dr. Hough that performance tends to improve with time. Yet, Dr. Zipper doesn’t see much change in the near term, because “after all, there is nothing to replace them [the programs].”
The problem gets even stickier for private payers because patients may be on an insurance panel for as little as a year or 2. Thanks to this rapid churn of beneficiaries, even the best-designed value-based program will have little time to prove its worth.
Dr. Zipper is among the many who don’t expect significant changes in the near term, asserting that “President Biden will want to get a few policy wins first, and health care is not the easiest place to start.”
But it’s likely that payers and others will want to see more emphasis on value-based programs despite these programs’ possible value to patients, physicians, and health systems alike.
A version of this article originally appeared on Medscape.com.
More than 40 value-based payment models – from direct contracting to bundled payments – have been introduced into the Medicare program in the past 10 years, with the goal of improving care while lowering costs. Hopes were high that they would be successful.
Physicians could suffer a huge blow to their income.
Many of the value-based care models simply did not work as expected, said Seema Verma, head of the Centers for Medicare & Medicaid Services, at a recent HLTH Conference. “They are not producing the types of savings the taxpayers deserve,” Ms. Verma said.
The Medicare Payment Advisory Commission (MedPac) concluded that, while dozens of payment models were tested, most failed to generate net savings for Medicare. Even the most successful of the models produced only modest savings. MedPac elaborated: “The track record raises the question of whether changes to particular models or CMMI’s [Center for Medicare & Medicaid Innovation’s] broader strategies might be warranted.”
What will happen now, as government officials admit that their value-based programs haven’t worked? The value-based programs could become more stringent. Here’s what physicians will have to contend with.
More risk. Experts agree that risk – financial risk – will be a component of future programs. Two-sided risk is likely to be the norm. This means that both parties – the provider and the insurer – are at financial risk for the patients covered by the program.
For example, a plan with 50,000 beneficiary patients would estimate the cost of caring for those patients on the basis of multiple variables. If the actual cost is lower than anticipated, both parties share in the savings. However, both share in the loss if the cost of caring for their patient population exceeds expectations.
This may compel physicians to enhance efficiency and potentially limit the services provided to patients. Typically, however, the strategy is to make efforts to prevent services like ED visits and admissions by focusing on health maintenance.
In contrast to most current value-based models, which feature little to no downside risk for physicians, double-sided risk means physicians could lose money. The loss may incorporate a cap – 5%, for example – but programs may differ. Experts concur that double-sided risk will be a hallmark of future programs.
Better data. The majority of health care services are rendered via fee-for-service: Patients receive services and physicians are paid, yet little or no information about outcomes is exchanged between insurers and physicians.
Penny Noyes, president of Health Business Navigators and contract negotiator for physicians, is not a fan of the current crop of value-based programs and feels that data transparency is positive. Sound metrics can lead to improvement, she said, adding: “It’s not money that drives physicians to make decisions; it’s what’s in the best interest of their patients and their patients’ long-term care.”
Value-based programs can work but only if applicable data are developed and given to physicians so that they can better understand their current performance and how to improve.
Mandated participation. Participation in value-based programs has been voluntary, but that may have skewed the results, which were better than what typical practice would have shown. Acknowledging this may lead CMS to call for mandated participation as a component of future programs. Physicians may be brought into programs, if only to determine whether the models really work. To date, participation in the programs has been voluntary, but that may change in the future.
Innovation. The private insurance market may end up as a key player. Over the past 6 months, health insurers have either consolidated partnerships with telemedicine companies to provide no-cost care to beneficiaries or have launched their own initiatives.
Others are focused on bringing together patients and providers operating outside of the traditional health care system, such as Aetna’s merger with CVS which now offers retail-based acute care (MinuteClinic) and chronic care (HealthHUB). Still other payers are gambling with physician practice ownership, as in the case of United Healthcare’s OptumHealth, which now boasts around 50,000 physicians throughout the country.
New practice models are emerging in private practices as well. Physicians are embracing remote care, proactively managing care transitions, and seeking out more methods to keep patients healthy and at home.
Not much was expected from value-based plans
Many are not surprised that the value-based models did not produce impressive results. Ms. Noyes doubted that positive outcomes will be achieved for physicians in comparison with what could have been attained under fee-for-service arrangements with lower administrative costs.
While the Affordable Care Act attempted to encourage alternative reimbursement, it limits the maximum medical loss ratio (MLR) a payer could achieve. For many plans, that maximum was 85%. Simply put, at least $0.85 of each premium collected had to be paid in claims; the remaining $0.15 went to margin, claims, and other administrative costs. A payer with an 82% MLR then would have to rebate the 3% difference to enrollees.
But that’s not what occurred, according to Ms. Noyes. Because value-based payments to providers are considered a claims expense, an MLR ratio of 82% allowed the payer to distribute the 3% difference to providers as value-based payments. Ms. Noyes said: “That may sound good for the provider, but the result was essentially a freeze on the provider’s fee-for-service reimbursement with the prospect of getting value-based payments like ‘shared savings.’
“When the providers tried to increase their base fee-for-service rates just to match inflation, payers often advised that any future raises had to be earned through value-based programs,” Ms. Noyes added. The value-based formulas confuse providers because payments are often made for periods as far back as 18 months, and providers do not have data systems to reconcile their payer report cards retrospectively. The result is that providers tended to accept whatever amount the payer distributed.
Executives at Lumeris, a company that helps health systems participate successfully in value-based care, see potential in a newer approach to alternative payments, such as CMS’ Direct Contracting initiative. This voluntary payment model offers options tailored to several types of organizations that aim to reduce costs while preserving or enhancing the quality of care for Medicare fee-for-service beneficiaries.
Jeff Smith, chief commercial officer for population health at Lumeris, explained that the Direct Contracting initiative can provide physicians with a more attractive option than prior value-based models because it adjusts for the complexity and fragility of patients with complex and chronic conditions. By allowing providers to participate in the savings generated, the initiative stands in stark contrast to what Mr. Smith described as the “shared savings to nothingness” experienced by providers in earlier-stage alternative payment models.
Physicians engaged with value-based programs like Direct Contracting are investing in nurses to aid with initiatives regarding health promotion and transitions of care. When a patient is discharged, for example, the nurse contacts the patient to discuss medications, schedule follow-up appointments, and so forth – tasks typically left to the patient (or caregiver) to navigate in the traditional system.
The initiative recognizes the importance of managing high-risk patients, those whom physicians identify as having an extraordinary number of ED visits and admissions. These patients, as well as so-called “rising-risk” patients, are targeted by nurses who proactively communicate with patients (and caregivers) to address patient’s needs, including social determinants of health.
Physicians who have a large load of patients in value-based programs are hiring social workers, pharmacists, and behavioral health experts to help. Of course, these personnel are costly, but that’s what the value-based programs aim to reimburse.
Still, the road ahead to value based is rocky and may not gain momentum for some time. Johns Hopkins University’s Doug Hough, PhD, an economist, recounts a government research study that sought to assess the university’s health system participation in a value-based payment program. While there were positive impacts on the program’s target population, Hough and his team discovered that the returns achieved by the optional model didn’t justify the health system’s financial support for it. The increasingly indebted health system ultimately decided to drop the optional program.
Dr. Hough indicated that the health system – Johns Hopkins Medicine – likely would have continued its support for the program had the government at least allowed it to break even. Although the payment program under study was a 3-year project, the bigger challenge, declared Dr. Hough, is that “we can’t turn an aircraft carrier that quickly.”
“Three years won’t show whether value-based care is really working,” Dr. Hough said.
Robert Zipper, MD, a hospitalist and senior policy advisor for Sound Physicians, a company that works to improve outcomes in acute care, agreed with Dr. Hough that performance tends to improve with time. Yet, Dr. Zipper doesn’t see much change in the near term, because “after all, there is nothing to replace them [the programs].”
The problem gets even stickier for private payers because patients may be on an insurance panel for as little as a year or 2. Thanks to this rapid churn of beneficiaries, even the best-designed value-based program will have little time to prove its worth.
Dr. Zipper is among the many who don’t expect significant changes in the near term, asserting that “President Biden will want to get a few policy wins first, and health care is not the easiest place to start.”
But it’s likely that payers and others will want to see more emphasis on value-based programs despite these programs’ possible value to patients, physicians, and health systems alike.
A version of this article originally appeared on Medscape.com.
FDA gives guidance on allergy, pregnancy concerns for Pfizer COVID vaccine
stating that it is safe for people with any history of allergies, but not for those who might have a known history of severe allergic reaction to any component of the vaccine.
The warning is included in the FDA’s information sheet for health care providers, but questions are arising as to whether the vaccine – which was authorized for emergency use by the FDA on Friday – should not be given to anyone with a history of allergies.
Sara Oliver, MD, an epidemic intelligence service officer with the Centers for Disease Control and Prevention reported at a Dec. 11 meeting of the agency’s Advisory Committee on Immunization Practices that two U.K. health care workers with a history of significant allergic reactions had a reaction to the Pfizer vaccine. A third health care worker with no history of allergies developed tachycardia, Dr. Oliver said.
“I want to reassure the public that although there were these few reactions in Great Britain, these were not seen in the larger clinical trial datasets,” said Peter Marks, MD, PhD, director of the Center for Biologics Evaluation and Research at the FDA, during a press briefing on Dec. 12.
The Pfizer vaccine “is one that we’re comfortable giving to patients who have had other allergic reactions besides those other than severe allergic reactions to a vaccine or one of its components,” he said.
Dr. Marks suggested that individuals let their physicians know about any history of allergic reactions. He also noted that the federal government will be supplying vaccine administration sites, at least initially, with epinephrine, diphenhydramine, hydrocortisone, and other medications needed to manage allergic reactions.
The FDA is going to monitor side effects such as allergic reactions very closely, “but I think we still need to learn more and that’s why we’re going to be taking precautions. We may have to modify things as we move forward,” said Dr. Marks.
Dr. Oliver said that on Dec. 12 the CDC convened an external panel with experience in vaccine safety, immunology, and allergies “to collate expert knowledge regarding possible cases,” and that the FDA is getting more data from U.K. regulatory authorities.
Pregnancy concerns
Agency officials had little to say, however, about the safety or efficacy of the vaccine for pregnant or breastfeeding women.
The FDA’s information to health care professionals noted that “available data on Pfizer-BioNTech COVID-19 vaccine administered to pregnant women are insufficient to inform vaccine-associated risks in pregnancy.”
Additionally, the agency stated, “data are not available to assess the effects of Pfizer-BioNTech COVID-19 vaccine on the breastfed infant or on milk production/excretion.”
Dr. Marks said that, for pregnant women and people who are immunocompromised, “it will be something that providers will need to consider on an individual basis.” He suggested that individuals consult with physicians to weigh the potential benefits and potential risks.
“Certainly, COVID-19 in a pregnant woman is not a good thing,” Dr. Marks said.
An individual might decide to go ahead with vaccination. “But that’s not something we’re recommending, that’s something we’re leaving up to the individual,” he said.
A version of this article originally appeared on Medscape.com.
stating that it is safe for people with any history of allergies, but not for those who might have a known history of severe allergic reaction to any component of the vaccine.
The warning is included in the FDA’s information sheet for health care providers, but questions are arising as to whether the vaccine – which was authorized for emergency use by the FDA on Friday – should not be given to anyone with a history of allergies.
Sara Oliver, MD, an epidemic intelligence service officer with the Centers for Disease Control and Prevention reported at a Dec. 11 meeting of the agency’s Advisory Committee on Immunization Practices that two U.K. health care workers with a history of significant allergic reactions had a reaction to the Pfizer vaccine. A third health care worker with no history of allergies developed tachycardia, Dr. Oliver said.
“I want to reassure the public that although there were these few reactions in Great Britain, these were not seen in the larger clinical trial datasets,” said Peter Marks, MD, PhD, director of the Center for Biologics Evaluation and Research at the FDA, during a press briefing on Dec. 12.
The Pfizer vaccine “is one that we’re comfortable giving to patients who have had other allergic reactions besides those other than severe allergic reactions to a vaccine or one of its components,” he said.
Dr. Marks suggested that individuals let their physicians know about any history of allergic reactions. He also noted that the federal government will be supplying vaccine administration sites, at least initially, with epinephrine, diphenhydramine, hydrocortisone, and other medications needed to manage allergic reactions.
The FDA is going to monitor side effects such as allergic reactions very closely, “but I think we still need to learn more and that’s why we’re going to be taking precautions. We may have to modify things as we move forward,” said Dr. Marks.
Dr. Oliver said that on Dec. 12 the CDC convened an external panel with experience in vaccine safety, immunology, and allergies “to collate expert knowledge regarding possible cases,” and that the FDA is getting more data from U.K. regulatory authorities.
Pregnancy concerns
Agency officials had little to say, however, about the safety or efficacy of the vaccine for pregnant or breastfeeding women.
The FDA’s information to health care professionals noted that “available data on Pfizer-BioNTech COVID-19 vaccine administered to pregnant women are insufficient to inform vaccine-associated risks in pregnancy.”
Additionally, the agency stated, “data are not available to assess the effects of Pfizer-BioNTech COVID-19 vaccine on the breastfed infant or on milk production/excretion.”
Dr. Marks said that, for pregnant women and people who are immunocompromised, “it will be something that providers will need to consider on an individual basis.” He suggested that individuals consult with physicians to weigh the potential benefits and potential risks.
“Certainly, COVID-19 in a pregnant woman is not a good thing,” Dr. Marks said.
An individual might decide to go ahead with vaccination. “But that’s not something we’re recommending, that’s something we’re leaving up to the individual,” he said.
A version of this article originally appeared on Medscape.com.
stating that it is safe for people with any history of allergies, but not for those who might have a known history of severe allergic reaction to any component of the vaccine.
The warning is included in the FDA’s information sheet for health care providers, but questions are arising as to whether the vaccine – which was authorized for emergency use by the FDA on Friday – should not be given to anyone with a history of allergies.
Sara Oliver, MD, an epidemic intelligence service officer with the Centers for Disease Control and Prevention reported at a Dec. 11 meeting of the agency’s Advisory Committee on Immunization Practices that two U.K. health care workers with a history of significant allergic reactions had a reaction to the Pfizer vaccine. A third health care worker with no history of allergies developed tachycardia, Dr. Oliver said.
“I want to reassure the public that although there were these few reactions in Great Britain, these were not seen in the larger clinical trial datasets,” said Peter Marks, MD, PhD, director of the Center for Biologics Evaluation and Research at the FDA, during a press briefing on Dec. 12.
The Pfizer vaccine “is one that we’re comfortable giving to patients who have had other allergic reactions besides those other than severe allergic reactions to a vaccine or one of its components,” he said.
Dr. Marks suggested that individuals let their physicians know about any history of allergic reactions. He also noted that the federal government will be supplying vaccine administration sites, at least initially, with epinephrine, diphenhydramine, hydrocortisone, and other medications needed to manage allergic reactions.
The FDA is going to monitor side effects such as allergic reactions very closely, “but I think we still need to learn more and that’s why we’re going to be taking precautions. We may have to modify things as we move forward,” said Dr. Marks.
Dr. Oliver said that on Dec. 12 the CDC convened an external panel with experience in vaccine safety, immunology, and allergies “to collate expert knowledge regarding possible cases,” and that the FDA is getting more data from U.K. regulatory authorities.
Pregnancy concerns
Agency officials had little to say, however, about the safety or efficacy of the vaccine for pregnant or breastfeeding women.
The FDA’s information to health care professionals noted that “available data on Pfizer-BioNTech COVID-19 vaccine administered to pregnant women are insufficient to inform vaccine-associated risks in pregnancy.”
Additionally, the agency stated, “data are not available to assess the effects of Pfizer-BioNTech COVID-19 vaccine on the breastfed infant or on milk production/excretion.”
Dr. Marks said that, for pregnant women and people who are immunocompromised, “it will be something that providers will need to consider on an individual basis.” He suggested that individuals consult with physicians to weigh the potential benefits and potential risks.
“Certainly, COVID-19 in a pregnant woman is not a good thing,” Dr. Marks said.
An individual might decide to go ahead with vaccination. “But that’s not something we’re recommending, that’s something we’re leaving up to the individual,” he said.
A version of this article originally appeared on Medscape.com.