User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
nav[contains(@class, 'nav-ce-stack nav-ce-stack__large-screen')]
header[@id='header']
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'main-prefix')]
footer[@id='footer']
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
div[contains(@class, 'view-medstat-quiz-listing-panes')]
Meghan Markle interview resurfaces suicidality screening debate
An eye-opening moment during March 7’s Meghan Markle/Prince Harry interview with Oprah Winfrey was Markle’s admission that, before the royal couple moved from the U.K., she was suicidal and had nowhere to turn for help.
For health care practitioners, this has resurfaced the debate over universal suicidality screening and discussion about what should happen when patients screen positive.
The American Psychiatric Association reports suicide is the 10th leading cause of death in the United States, but the second leading cause of death in people age 10-34 years old.
The latest data from the Centers for Disease Control and Prevention show that, in 2019, suicide rates dropped for the first time in 14 years. However, it is widely expected that, in the face of the COVID-19 pandemic and its associated isolation, loneliness, and stress, the next round of data will show a surge in suicide deaths.
, chair of the APA’s Committee on Women’s Mental Health.
“I can see it in my office. People who didn’t necessarily complain about anxiety and depression before or who had been stable for many years are decompensating now,” Dr. De Faria said in an interview.
Although other parts of the interview may have been controversial, said Dr. De Faria, Markle’s disclosure has opened up a much-needed discussion.
“I’m all for people talking about mental health, and I commend [Markle] for sharing her struggle and putting it out there,” she added.
In a perfect world, she noted, there would be universal suicide screening by all medical professionals.
However, Dr. De Faria, associate clinical professor of psychiatry, University of Florida, Gainesville, acknowledged that from a resource standpoint this is not a pragmatic solution.
Primary care physicians are often the frontline defense for suicide prevention, noted Mona Masood, DO, a Philadelphia-area psychiatrist and founder and chief organizer of the Physician Support Line, a free mental health hotline exclusively for doctors staffed by volunteer psychiatrists.
“I believe our general practitioner colleagues, our family medicine colleagues are the ones who are going to be seeing the majority of mental health concerns or illnesses because of the stigma that is often there from seeing a psychiatrist,” Dr. Masood said in an interview.
Dr. De Faria noted that the Patient Health Questionnaire-2 (PHQ-2) for mental health offers a simple screen that includes two key questions. It asks: Over the last 2 weeks, how often have you been bothered by the following problems?
- Little interest or pleasure in doing things.
- Feeling down, depressed, or hopeless.
However, both Dr. De Faria and Dr. Masood emphasized that individualized follow-up questions and follow-up care are equally important.
Unlike Dr. De Faria, who prefers universal screening but understands the challenges of implementing it, Dr. Masood favors targeted screening.
“For physicians, the whole point of what we do is to save lives. To talk to somebody about the complete opposite and to ask, ‘Are you planning on ending your life?’ is very jarring. But for the patient, that may be their only outlet. A primary care provider may be the only professional that they talk to about their mental health,” said Dr. Masood.
Patients can easily say “no” to suicidal ideation questions from a general screen, but targeted, probing questions let patients know that they’re being heard and seen beyond their physical examination, she added.
She also suggested that clinicians ask open-ended questions of those patients who are struggling.
Dr. Masood noted that having a plan in place before screening a patient is especially key.
“I’d argue that one of the subconscious reasons why so many doctors do not ask the question [about suicidality] is because if you ask it, you have to be ready for the answer and to know what you’d do,” she said.
All primary care physicians should have “mental health professionals as resources in your back pocket” in order to have a referral ready to give to patients in need, she said.
“Outside of your clinic time, have a rapport with your local psychiatrist or therapist and know where to send someone who is suicidal,” Dr. Masood said. “Know what is in your local area so you’ll already know how to implement your plan.”
Dr. Masood also recommended:
- Informing staff about protocols for patients with suicidal thoughts who need to go to the hospital for evaluation.
- Creating a safe space in the clinic/office, such as an unused exam room, where patients can wait for next steps.
- Having staff inform a patient’s emergency contact about the situation.
- Trying for a “warm handoff,” where the emergency contact takes the patient to the nearest crisis center and having staff call ahead to let them know to expect the patient.
- If the patient has no emergency contact, following your state’s involuntary crisis response protocol, which involves calling 911 for emergency services.
In addition, the APA’s suicide prevention page includes a long list of helpful resources for patients, families, and physicians.
A version of this article first appeared on Medscape.com.
An eye-opening moment during March 7’s Meghan Markle/Prince Harry interview with Oprah Winfrey was Markle’s admission that, before the royal couple moved from the U.K., she was suicidal and had nowhere to turn for help.
For health care practitioners, this has resurfaced the debate over universal suicidality screening and discussion about what should happen when patients screen positive.
The American Psychiatric Association reports suicide is the 10th leading cause of death in the United States, but the second leading cause of death in people age 10-34 years old.
The latest data from the Centers for Disease Control and Prevention show that, in 2019, suicide rates dropped for the first time in 14 years. However, it is widely expected that, in the face of the COVID-19 pandemic and its associated isolation, loneliness, and stress, the next round of data will show a surge in suicide deaths.
, chair of the APA’s Committee on Women’s Mental Health.
“I can see it in my office. People who didn’t necessarily complain about anxiety and depression before or who had been stable for many years are decompensating now,” Dr. De Faria said in an interview.
Although other parts of the interview may have been controversial, said Dr. De Faria, Markle’s disclosure has opened up a much-needed discussion.
“I’m all for people talking about mental health, and I commend [Markle] for sharing her struggle and putting it out there,” she added.
In a perfect world, she noted, there would be universal suicide screening by all medical professionals.
However, Dr. De Faria, associate clinical professor of psychiatry, University of Florida, Gainesville, acknowledged that from a resource standpoint this is not a pragmatic solution.
Primary care physicians are often the frontline defense for suicide prevention, noted Mona Masood, DO, a Philadelphia-area psychiatrist and founder and chief organizer of the Physician Support Line, a free mental health hotline exclusively for doctors staffed by volunteer psychiatrists.
“I believe our general practitioner colleagues, our family medicine colleagues are the ones who are going to be seeing the majority of mental health concerns or illnesses because of the stigma that is often there from seeing a psychiatrist,” Dr. Masood said in an interview.
Dr. De Faria noted that the Patient Health Questionnaire-2 (PHQ-2) for mental health offers a simple screen that includes two key questions. It asks: Over the last 2 weeks, how often have you been bothered by the following problems?
- Little interest or pleasure in doing things.
- Feeling down, depressed, or hopeless.
However, both Dr. De Faria and Dr. Masood emphasized that individualized follow-up questions and follow-up care are equally important.
Unlike Dr. De Faria, who prefers universal screening but understands the challenges of implementing it, Dr. Masood favors targeted screening.
“For physicians, the whole point of what we do is to save lives. To talk to somebody about the complete opposite and to ask, ‘Are you planning on ending your life?’ is very jarring. But for the patient, that may be their only outlet. A primary care provider may be the only professional that they talk to about their mental health,” said Dr. Masood.
Patients can easily say “no” to suicidal ideation questions from a general screen, but targeted, probing questions let patients know that they’re being heard and seen beyond their physical examination, she added.
She also suggested that clinicians ask open-ended questions of those patients who are struggling.
Dr. Masood noted that having a plan in place before screening a patient is especially key.
“I’d argue that one of the subconscious reasons why so many doctors do not ask the question [about suicidality] is because if you ask it, you have to be ready for the answer and to know what you’d do,” she said.
All primary care physicians should have “mental health professionals as resources in your back pocket” in order to have a referral ready to give to patients in need, she said.
“Outside of your clinic time, have a rapport with your local psychiatrist or therapist and know where to send someone who is suicidal,” Dr. Masood said. “Know what is in your local area so you’ll already know how to implement your plan.”
Dr. Masood also recommended:
- Informing staff about protocols for patients with suicidal thoughts who need to go to the hospital for evaluation.
- Creating a safe space in the clinic/office, such as an unused exam room, where patients can wait for next steps.
- Having staff inform a patient’s emergency contact about the situation.
- Trying for a “warm handoff,” where the emergency contact takes the patient to the nearest crisis center and having staff call ahead to let them know to expect the patient.
- If the patient has no emergency contact, following your state’s involuntary crisis response protocol, which involves calling 911 for emergency services.
In addition, the APA’s suicide prevention page includes a long list of helpful resources for patients, families, and physicians.
A version of this article first appeared on Medscape.com.
An eye-opening moment during March 7’s Meghan Markle/Prince Harry interview with Oprah Winfrey was Markle’s admission that, before the royal couple moved from the U.K., she was suicidal and had nowhere to turn for help.
For health care practitioners, this has resurfaced the debate over universal suicidality screening and discussion about what should happen when patients screen positive.
The American Psychiatric Association reports suicide is the 10th leading cause of death in the United States, but the second leading cause of death in people age 10-34 years old.
The latest data from the Centers for Disease Control and Prevention show that, in 2019, suicide rates dropped for the first time in 14 years. However, it is widely expected that, in the face of the COVID-19 pandemic and its associated isolation, loneliness, and stress, the next round of data will show a surge in suicide deaths.
, chair of the APA’s Committee on Women’s Mental Health.
“I can see it in my office. People who didn’t necessarily complain about anxiety and depression before or who had been stable for many years are decompensating now,” Dr. De Faria said in an interview.
Although other parts of the interview may have been controversial, said Dr. De Faria, Markle’s disclosure has opened up a much-needed discussion.
“I’m all for people talking about mental health, and I commend [Markle] for sharing her struggle and putting it out there,” she added.
In a perfect world, she noted, there would be universal suicide screening by all medical professionals.
However, Dr. De Faria, associate clinical professor of psychiatry, University of Florida, Gainesville, acknowledged that from a resource standpoint this is not a pragmatic solution.
Primary care physicians are often the frontline defense for suicide prevention, noted Mona Masood, DO, a Philadelphia-area psychiatrist and founder and chief organizer of the Physician Support Line, a free mental health hotline exclusively for doctors staffed by volunteer psychiatrists.
“I believe our general practitioner colleagues, our family medicine colleagues are the ones who are going to be seeing the majority of mental health concerns or illnesses because of the stigma that is often there from seeing a psychiatrist,” Dr. Masood said in an interview.
Dr. De Faria noted that the Patient Health Questionnaire-2 (PHQ-2) for mental health offers a simple screen that includes two key questions. It asks: Over the last 2 weeks, how often have you been bothered by the following problems?
- Little interest or pleasure in doing things.
- Feeling down, depressed, or hopeless.
However, both Dr. De Faria and Dr. Masood emphasized that individualized follow-up questions and follow-up care are equally important.
Unlike Dr. De Faria, who prefers universal screening but understands the challenges of implementing it, Dr. Masood favors targeted screening.
“For physicians, the whole point of what we do is to save lives. To talk to somebody about the complete opposite and to ask, ‘Are you planning on ending your life?’ is very jarring. But for the patient, that may be their only outlet. A primary care provider may be the only professional that they talk to about their mental health,” said Dr. Masood.
Patients can easily say “no” to suicidal ideation questions from a general screen, but targeted, probing questions let patients know that they’re being heard and seen beyond their physical examination, she added.
She also suggested that clinicians ask open-ended questions of those patients who are struggling.
Dr. Masood noted that having a plan in place before screening a patient is especially key.
“I’d argue that one of the subconscious reasons why so many doctors do not ask the question [about suicidality] is because if you ask it, you have to be ready for the answer and to know what you’d do,” she said.
All primary care physicians should have “mental health professionals as resources in your back pocket” in order to have a referral ready to give to patients in need, she said.
“Outside of your clinic time, have a rapport with your local psychiatrist or therapist and know where to send someone who is suicidal,” Dr. Masood said. “Know what is in your local area so you’ll already know how to implement your plan.”
Dr. Masood also recommended:
- Informing staff about protocols for patients with suicidal thoughts who need to go to the hospital for evaluation.
- Creating a safe space in the clinic/office, such as an unused exam room, where patients can wait for next steps.
- Having staff inform a patient’s emergency contact about the situation.
- Trying for a “warm handoff,” where the emergency contact takes the patient to the nearest crisis center and having staff call ahead to let them know to expect the patient.
- If the patient has no emergency contact, following your state’s involuntary crisis response protocol, which involves calling 911 for emergency services.
In addition, the APA’s suicide prevention page includes a long list of helpful resources for patients, families, and physicians.
A version of this article first appeared on Medscape.com.
Meghan Markle disclosures can be used to raise awareness, reduce stigma
Suicidal thoughts require compassion and action
When I sat down to watch Oprah Winfrey’s interview with Meghan Markle on Sunday night, I didn’t know what to expect. As a psychiatrist dedicated to reducing the loss of life through suicide, I homed in on the aspect of the interview in which she discussed the depth of her suffering.
Meghan Markle is one of many celebrities to speak about their experience with suicidal crisis. Those disclosures provide opportunities to increase the public’s understanding of mental health and to deepen compassion for what others may be going through. They challenge our culturally normed assumptions: false beliefs – such as the idea that beauty, wealth, and success protect people from mental health struggles. We would do well to trust that the dichotomy between external appearances and internal experiences is always at play, and we never know what someone is going through. Human beings are both enormously resilient and vulnerable.
Suicide, while complex, is a health issue. Therefore, it is important that we all do our part in approaching mental health and suicide risk in a similar manner to other health issues.
We all have a dynamic and continuous interplay going on between life circumstances and our internal biological, genetic, and psychological makeup. The more honest and the more we learn about our own vulnerabilities and strengths, the more proactive we can be in protecting and optimizing our mental health. All individuals should be able to receive support and access to care to have their mental health needs addressed.
It’s important to note that distress leads many people to instinctually withdraw, just at a time when receiving support is even more important. In addition, cultures that traditionally emphasize self-sufficiency or stoicism may unintentionally create additional barriers to reaching out for help. Therefore, many people who experience suicidal thoughts do not disclose them to anyone. If someone does mention they are struggling, you can thank them for opening up and let them know you want to support them, and that you are there to help them find the help they need.
This is what we should tell our patients: If someone you know is struggling and might be thinking about suicide, assume you are the only one who will reach out. Having an honest conversation with the person will let them know that you care. Take the person seriously and speak up if you’re concerned about them. Ask directly if they are thinking about suicide. Help the person by connecting them with a mental health professional. If you aren’t sure if their safety is imminently at risk, connect with crisis resources and services immediately. Realize that most people who are suicidal are ambivalent about taking their life and really just want relief to their pain: Part of them wants to live and part of them wants to die. by helping them secure firearms, medications, and any other potentially lethal means.
Suicidal thoughts cut across all demographic groups, occupations, and socioeconomic strata. By understanding that life presents intense challenges for all of us and that suicidal thoughts are an indication of suffering warranting professional evaluation and intervention, we can approach the next person who discloses suicidal thoughts with compassion and action. To learn more, go to afsp.org.
Dr. Moutier is chief medical officer of the American Foundation for Suicide Prevention. She reported no disclosures.
Suicidal thoughts require compassion and action
Suicidal thoughts require compassion and action
When I sat down to watch Oprah Winfrey’s interview with Meghan Markle on Sunday night, I didn’t know what to expect. As a psychiatrist dedicated to reducing the loss of life through suicide, I homed in on the aspect of the interview in which she discussed the depth of her suffering.
Meghan Markle is one of many celebrities to speak about their experience with suicidal crisis. Those disclosures provide opportunities to increase the public’s understanding of mental health and to deepen compassion for what others may be going through. They challenge our culturally normed assumptions: false beliefs – such as the idea that beauty, wealth, and success protect people from mental health struggles. We would do well to trust that the dichotomy between external appearances and internal experiences is always at play, and we never know what someone is going through. Human beings are both enormously resilient and vulnerable.
Suicide, while complex, is a health issue. Therefore, it is important that we all do our part in approaching mental health and suicide risk in a similar manner to other health issues.
We all have a dynamic and continuous interplay going on between life circumstances and our internal biological, genetic, and psychological makeup. The more honest and the more we learn about our own vulnerabilities and strengths, the more proactive we can be in protecting and optimizing our mental health. All individuals should be able to receive support and access to care to have their mental health needs addressed.
It’s important to note that distress leads many people to instinctually withdraw, just at a time when receiving support is even more important. In addition, cultures that traditionally emphasize self-sufficiency or stoicism may unintentionally create additional barriers to reaching out for help. Therefore, many people who experience suicidal thoughts do not disclose them to anyone. If someone does mention they are struggling, you can thank them for opening up and let them know you want to support them, and that you are there to help them find the help they need.
This is what we should tell our patients: If someone you know is struggling and might be thinking about suicide, assume you are the only one who will reach out. Having an honest conversation with the person will let them know that you care. Take the person seriously and speak up if you’re concerned about them. Ask directly if they are thinking about suicide. Help the person by connecting them with a mental health professional. If you aren’t sure if their safety is imminently at risk, connect with crisis resources and services immediately. Realize that most people who are suicidal are ambivalent about taking their life and really just want relief to their pain: Part of them wants to live and part of them wants to die. by helping them secure firearms, medications, and any other potentially lethal means.
Suicidal thoughts cut across all demographic groups, occupations, and socioeconomic strata. By understanding that life presents intense challenges for all of us and that suicidal thoughts are an indication of suffering warranting professional evaluation and intervention, we can approach the next person who discloses suicidal thoughts with compassion and action. To learn more, go to afsp.org.
Dr. Moutier is chief medical officer of the American Foundation for Suicide Prevention. She reported no disclosures.
When I sat down to watch Oprah Winfrey’s interview with Meghan Markle on Sunday night, I didn’t know what to expect. As a psychiatrist dedicated to reducing the loss of life through suicide, I homed in on the aspect of the interview in which she discussed the depth of her suffering.
Meghan Markle is one of many celebrities to speak about their experience with suicidal crisis. Those disclosures provide opportunities to increase the public’s understanding of mental health and to deepen compassion for what others may be going through. They challenge our culturally normed assumptions: false beliefs – such as the idea that beauty, wealth, and success protect people from mental health struggles. We would do well to trust that the dichotomy between external appearances and internal experiences is always at play, and we never know what someone is going through. Human beings are both enormously resilient and vulnerable.
Suicide, while complex, is a health issue. Therefore, it is important that we all do our part in approaching mental health and suicide risk in a similar manner to other health issues.
We all have a dynamic and continuous interplay going on between life circumstances and our internal biological, genetic, and psychological makeup. The more honest and the more we learn about our own vulnerabilities and strengths, the more proactive we can be in protecting and optimizing our mental health. All individuals should be able to receive support and access to care to have their mental health needs addressed.
It’s important to note that distress leads many people to instinctually withdraw, just at a time when receiving support is even more important. In addition, cultures that traditionally emphasize self-sufficiency or stoicism may unintentionally create additional barriers to reaching out for help. Therefore, many people who experience suicidal thoughts do not disclose them to anyone. If someone does mention they are struggling, you can thank them for opening up and let them know you want to support them, and that you are there to help them find the help they need.
This is what we should tell our patients: If someone you know is struggling and might be thinking about suicide, assume you are the only one who will reach out. Having an honest conversation with the person will let them know that you care. Take the person seriously and speak up if you’re concerned about them. Ask directly if they are thinking about suicide. Help the person by connecting them with a mental health professional. If you aren’t sure if their safety is imminently at risk, connect with crisis resources and services immediately. Realize that most people who are suicidal are ambivalent about taking their life and really just want relief to their pain: Part of them wants to live and part of them wants to die. by helping them secure firearms, medications, and any other potentially lethal means.
Suicidal thoughts cut across all demographic groups, occupations, and socioeconomic strata. By understanding that life presents intense challenges for all of us and that suicidal thoughts are an indication of suffering warranting professional evaluation and intervention, we can approach the next person who discloses suicidal thoughts with compassion and action. To learn more, go to afsp.org.
Dr. Moutier is chief medical officer of the American Foundation for Suicide Prevention. She reported no disclosures.
Traumatic brain injury tied to long-term sleep problems
Veterans who have suffered a traumatic brain injury (TBI) are significantly more likely to develop insomnia and other sleep problems years later compared to their counterparts who have not suffered a brain injury, a new study shows.
Results of a large longitudinal study show that those with TBI were about 40% more likely to develop insomnia, sleep apnea, excessive daytime sleepiness, or another sleep disorder in later years, after adjusting for demographics and medical and psychiatric conditions.
Interestingly, the association with sleep disorders was strongest among those with mild TBI versus a more severe brain injury.
The study showed that the risk for sleep disorders increased up to 14 years after a brain injury, an indicator that “clinicians should really pay attention to sleep disorders in TBI patients both in the short term and the long term,” study investigator Yue Leng, MD, PhD, assistant professor, department of psychiatry and behavioral sciences, University of California, San Francisco, told this news organization.
The study was published online March 3 in Neurology.
First long-term look
TBI is common among veterans, who may have sleep complaints or psychiatric symptoms, but previous studies into the consequences of TBI have examined the short- vs. long-term impact, said Dr. Leng.
To examine the longitudinal association between TBI and sleep disorders, the investigators examined data on 98,709 Veterans Health Administration patients diagnosed with TBI and an age-matched group of the same number of veterans who had not received such a diagnosis. The mean age of the participants was 49 years at baseline, and 11.7% were women. Of the TBI cases, 49.6% were mild.
Researchers used an exposure survey and diagnostic codes to establish TBI and its severity.
Patients with TBI were more likely to be male and were much more likely to have a psychiatric condition, such as a mood disorder (22.4% vs. 9.3%), anxiety (10.5% vs. 4.4%), posttraumatic stress disorder (19.5% vs. 4.4%), or substance abuse (11.4% vs. 5.2%). They were also more likely to smoke or use tobacco (13.5% vs. 8.7%).
Researchers assessed a number of sleep disorders, including insomnia, hypersomnia disorders, narcolepsy, sleep-related breathing disorders, and sleep-related movement disorders.
During a follow-up period that averaged 5 years but ranged up to 14 years, 23.4% of veterans with TBI and 15.8% of those without TBI developed a sleep disorder.
After adjusting for age, sex, race, education, and income, those who had suffered a TBI were 50% more likely to develop any sleep disorder, compared with those who had not had a TBI (hazard ratio, 1.50; 95% confidence interval, 1.47-1.53.)
After controlling for medical conditions that included diabetes, hypertension, myocardial infarction, and cerebrovascular disease, as well as psychiatric disorders such as mood disorders, anxiety, PTSD, substance use disorder, and tobacco use, the HR for developing a sleep disorder was 1.41 (95% CI, 1.37-1.44).
The association with TBI was stronger for some sleep disorders. Adjusted HRs were 1.50 (95% CI, 1.45-1.55) for insomnia, 1.50 (95% CI, 1.39-1.61) for hypersomnia, 1.33 (95% CI, 1.16-1.52) for sleep-related movement disorders, and 1.28 (95% CI, 1.24-1.32) for sleep apnea.
It’s unclear what causes postinjury sleep problems, but it could be that TBI induces structural brain damage, or it could affect melatonin secretion or wake-promoting neurons.
Damage to arousal-promoting neurons could help explain the reason the link between TBI and sleep disorders was strongest for insomnia and hypersomnia, although the exact mechanism is unclear, said Dr. Leng.
Greater risk with mild TBI
Overall, the association was stronger for mild TBI than for moderate to severe TBI. This, said Dr. Leng, might be because of differences in the brain injury mechanism.
Mild TBI often involves repetitive concussive or subconcussive injuries, such as sports injuries or blast injury among active-duty military personnel. This type of injury is more likely to cause diffuse axonal injury and inflammation, whereas moderate or severe TBI is often attributable to a direct blow with more focal but severe damage, explained Dr. Leng.
She noted that veterans with mild TBI were more likely to have a psychiatric condition, but because the study controlled for such conditions, this doesn’t fully explain the stronger association between mild TBI and sleep disorders.
Further studies are needed to sort out the exact mechanisms, she said.
The association between TBI and risk for sleep disorders was reduced somewhat but was still moderate in an analysis that excluded patients who developed a sleep disorder within 2 years of a brain injury.
This analysis, said Dr. Leng, helped ensure that the sleep disorder developed after the brain injury.
The researchers could not examine the trajectory of sleep problems, so it’s not clear whether sleep problems worsen or get better over time, said Dr. Leng.
Because PTSD also leads to sleep problems, the researchers thought that having both PTSD and TBI might increase the risk for sleep problems. “But actually we found the association was pretty similar in those with, and without, PTSD, so that was kind of contrary to our hypothesis,” she said.
The new results underline the need for more screening for sleep disorders among patients with TBI, both in the short term and the long term, said Dr. Leng. “Clinicians should ask TBI patients about their sleep, and they should follow that up,” she said.
She added that long-term sleep disorders can affect a patient’s health and can lead to psychiatric problems and neurodegenerative diseases.
Depending on the type of sleep disorder, there are a number of possible treatments. For example, for patients with sleep apnea, continuous positive airway pressure treatment may be considered.
‘Outstanding’ research
Commenting for this news organization, Frank Conidi, MD, director, Florida Center for Headache and Sports Neurology; CEO, Brainsport, Team Neurologist, the Florida Panthers of the National Hockey League; and past president, Florida Society of Neurology, said the study is “by far” the largest to investigate the correlation between sleep disorders and head trauma.
The design and outcome measures “were well thought out,” and the researchers “did an outstanding job in sorting through and analyzing the data,” said Dr. Conidi.
He added that he was particularly impressed with how the researchers addressed PTSD, which is highly prevalent among veterans with head trauma and is known to affect sleep.
The new results “solidify what those of us who see individuals with TBI have observed over the years: that there is a higher incidence of all types of sleep disorders” in individuals with a TBI, said Dr. Conidi.
However, he questioned the study’s use of guidelines to classify the various types of head trauma. These guidelines, he said, “are based on loss of consciousness, which we have started to move away from when classifying TBI.”
In addition, Dr. Conidi said he “would have loved to have seen” some correlation with neuroimaging studies, such as those used to assess subdural hematoma, epidural hematoma, subarachnoid hemorrhage, and diffuse axonal injury, but that this “could be an impetus for future studies.”
In “a perfect world,” all patients with a TBI would undergo a polysomnography study in a sleep laboratory, but insurance companies now rarely cover such studies and have attempted to have clinicians shift to home sleep studies, said Dr. Conidi. “These are marginal at best for screening for sleep disorders,” he noted.
At his centers, every TBI patient is screened for sleep disorders and, whenever possible, undergoes formal evaluation in the sleep lab, he added.
The study was supported by the U.S. Army Medical Research and Material Command and the U.S. Department of Veterans Affairs. Dr. Leng and Dr. Conidi have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Veterans who have suffered a traumatic brain injury (TBI) are significantly more likely to develop insomnia and other sleep problems years later compared to their counterparts who have not suffered a brain injury, a new study shows.
Results of a large longitudinal study show that those with TBI were about 40% more likely to develop insomnia, sleep apnea, excessive daytime sleepiness, or another sleep disorder in later years, after adjusting for demographics and medical and psychiatric conditions.
Interestingly, the association with sleep disorders was strongest among those with mild TBI versus a more severe brain injury.
The study showed that the risk for sleep disorders increased up to 14 years after a brain injury, an indicator that “clinicians should really pay attention to sleep disorders in TBI patients both in the short term and the long term,” study investigator Yue Leng, MD, PhD, assistant professor, department of psychiatry and behavioral sciences, University of California, San Francisco, told this news organization.
The study was published online March 3 in Neurology.
First long-term look
TBI is common among veterans, who may have sleep complaints or psychiatric symptoms, but previous studies into the consequences of TBI have examined the short- vs. long-term impact, said Dr. Leng.
To examine the longitudinal association between TBI and sleep disorders, the investigators examined data on 98,709 Veterans Health Administration patients diagnosed with TBI and an age-matched group of the same number of veterans who had not received such a diagnosis. The mean age of the participants was 49 years at baseline, and 11.7% were women. Of the TBI cases, 49.6% were mild.
Researchers used an exposure survey and diagnostic codes to establish TBI and its severity.
Patients with TBI were more likely to be male and were much more likely to have a psychiatric condition, such as a mood disorder (22.4% vs. 9.3%), anxiety (10.5% vs. 4.4%), posttraumatic stress disorder (19.5% vs. 4.4%), or substance abuse (11.4% vs. 5.2%). They were also more likely to smoke or use tobacco (13.5% vs. 8.7%).
Researchers assessed a number of sleep disorders, including insomnia, hypersomnia disorders, narcolepsy, sleep-related breathing disorders, and sleep-related movement disorders.
During a follow-up period that averaged 5 years but ranged up to 14 years, 23.4% of veterans with TBI and 15.8% of those without TBI developed a sleep disorder.
After adjusting for age, sex, race, education, and income, those who had suffered a TBI were 50% more likely to develop any sleep disorder, compared with those who had not had a TBI (hazard ratio, 1.50; 95% confidence interval, 1.47-1.53.)
After controlling for medical conditions that included diabetes, hypertension, myocardial infarction, and cerebrovascular disease, as well as psychiatric disorders such as mood disorders, anxiety, PTSD, substance use disorder, and tobacco use, the HR for developing a sleep disorder was 1.41 (95% CI, 1.37-1.44).
The association with TBI was stronger for some sleep disorders. Adjusted HRs were 1.50 (95% CI, 1.45-1.55) for insomnia, 1.50 (95% CI, 1.39-1.61) for hypersomnia, 1.33 (95% CI, 1.16-1.52) for sleep-related movement disorders, and 1.28 (95% CI, 1.24-1.32) for sleep apnea.
It’s unclear what causes postinjury sleep problems, but it could be that TBI induces structural brain damage, or it could affect melatonin secretion or wake-promoting neurons.
Damage to arousal-promoting neurons could help explain the reason the link between TBI and sleep disorders was strongest for insomnia and hypersomnia, although the exact mechanism is unclear, said Dr. Leng.
Greater risk with mild TBI
Overall, the association was stronger for mild TBI than for moderate to severe TBI. This, said Dr. Leng, might be because of differences in the brain injury mechanism.
Mild TBI often involves repetitive concussive or subconcussive injuries, such as sports injuries or blast injury among active-duty military personnel. This type of injury is more likely to cause diffuse axonal injury and inflammation, whereas moderate or severe TBI is often attributable to a direct blow with more focal but severe damage, explained Dr. Leng.
She noted that veterans with mild TBI were more likely to have a psychiatric condition, but because the study controlled for such conditions, this doesn’t fully explain the stronger association between mild TBI and sleep disorders.
Further studies are needed to sort out the exact mechanisms, she said.
The association between TBI and risk for sleep disorders was reduced somewhat but was still moderate in an analysis that excluded patients who developed a sleep disorder within 2 years of a brain injury.
This analysis, said Dr. Leng, helped ensure that the sleep disorder developed after the brain injury.
The researchers could not examine the trajectory of sleep problems, so it’s not clear whether sleep problems worsen or get better over time, said Dr. Leng.
Because PTSD also leads to sleep problems, the researchers thought that having both PTSD and TBI might increase the risk for sleep problems. “But actually we found the association was pretty similar in those with, and without, PTSD, so that was kind of contrary to our hypothesis,” she said.
The new results underline the need for more screening for sleep disorders among patients with TBI, both in the short term and the long term, said Dr. Leng. “Clinicians should ask TBI patients about their sleep, and they should follow that up,” she said.
She added that long-term sleep disorders can affect a patient’s health and can lead to psychiatric problems and neurodegenerative diseases.
Depending on the type of sleep disorder, there are a number of possible treatments. For example, for patients with sleep apnea, continuous positive airway pressure treatment may be considered.
‘Outstanding’ research
Commenting for this news organization, Frank Conidi, MD, director, Florida Center for Headache and Sports Neurology; CEO, Brainsport, Team Neurologist, the Florida Panthers of the National Hockey League; and past president, Florida Society of Neurology, said the study is “by far” the largest to investigate the correlation between sleep disorders and head trauma.
The design and outcome measures “were well thought out,” and the researchers “did an outstanding job in sorting through and analyzing the data,” said Dr. Conidi.
He added that he was particularly impressed with how the researchers addressed PTSD, which is highly prevalent among veterans with head trauma and is known to affect sleep.
The new results “solidify what those of us who see individuals with TBI have observed over the years: that there is a higher incidence of all types of sleep disorders” in individuals with a TBI, said Dr. Conidi.
However, he questioned the study’s use of guidelines to classify the various types of head trauma. These guidelines, he said, “are based on loss of consciousness, which we have started to move away from when classifying TBI.”
In addition, Dr. Conidi said he “would have loved to have seen” some correlation with neuroimaging studies, such as those used to assess subdural hematoma, epidural hematoma, subarachnoid hemorrhage, and diffuse axonal injury, but that this “could be an impetus for future studies.”
In “a perfect world,” all patients with a TBI would undergo a polysomnography study in a sleep laboratory, but insurance companies now rarely cover such studies and have attempted to have clinicians shift to home sleep studies, said Dr. Conidi. “These are marginal at best for screening for sleep disorders,” he noted.
At his centers, every TBI patient is screened for sleep disorders and, whenever possible, undergoes formal evaluation in the sleep lab, he added.
The study was supported by the U.S. Army Medical Research and Material Command and the U.S. Department of Veterans Affairs. Dr. Leng and Dr. Conidi have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Veterans who have suffered a traumatic brain injury (TBI) are significantly more likely to develop insomnia and other sleep problems years later compared to their counterparts who have not suffered a brain injury, a new study shows.
Results of a large longitudinal study show that those with TBI were about 40% more likely to develop insomnia, sleep apnea, excessive daytime sleepiness, or another sleep disorder in later years, after adjusting for demographics and medical and psychiatric conditions.
Interestingly, the association with sleep disorders was strongest among those with mild TBI versus a more severe brain injury.
The study showed that the risk for sleep disorders increased up to 14 years after a brain injury, an indicator that “clinicians should really pay attention to sleep disorders in TBI patients both in the short term and the long term,” study investigator Yue Leng, MD, PhD, assistant professor, department of psychiatry and behavioral sciences, University of California, San Francisco, told this news organization.
The study was published online March 3 in Neurology.
First long-term look
TBI is common among veterans, who may have sleep complaints or psychiatric symptoms, but previous studies into the consequences of TBI have examined the short- vs. long-term impact, said Dr. Leng.
To examine the longitudinal association between TBI and sleep disorders, the investigators examined data on 98,709 Veterans Health Administration patients diagnosed with TBI and an age-matched group of the same number of veterans who had not received such a diagnosis. The mean age of the participants was 49 years at baseline, and 11.7% were women. Of the TBI cases, 49.6% were mild.
Researchers used an exposure survey and diagnostic codes to establish TBI and its severity.
Patients with TBI were more likely to be male and were much more likely to have a psychiatric condition, such as a mood disorder (22.4% vs. 9.3%), anxiety (10.5% vs. 4.4%), posttraumatic stress disorder (19.5% vs. 4.4%), or substance abuse (11.4% vs. 5.2%). They were also more likely to smoke or use tobacco (13.5% vs. 8.7%).
Researchers assessed a number of sleep disorders, including insomnia, hypersomnia disorders, narcolepsy, sleep-related breathing disorders, and sleep-related movement disorders.
During a follow-up period that averaged 5 years but ranged up to 14 years, 23.4% of veterans with TBI and 15.8% of those without TBI developed a sleep disorder.
After adjusting for age, sex, race, education, and income, those who had suffered a TBI were 50% more likely to develop any sleep disorder, compared with those who had not had a TBI (hazard ratio, 1.50; 95% confidence interval, 1.47-1.53.)
After controlling for medical conditions that included diabetes, hypertension, myocardial infarction, and cerebrovascular disease, as well as psychiatric disorders such as mood disorders, anxiety, PTSD, substance use disorder, and tobacco use, the HR for developing a sleep disorder was 1.41 (95% CI, 1.37-1.44).
The association with TBI was stronger for some sleep disorders. Adjusted HRs were 1.50 (95% CI, 1.45-1.55) for insomnia, 1.50 (95% CI, 1.39-1.61) for hypersomnia, 1.33 (95% CI, 1.16-1.52) for sleep-related movement disorders, and 1.28 (95% CI, 1.24-1.32) for sleep apnea.
It’s unclear what causes postinjury sleep problems, but it could be that TBI induces structural brain damage, or it could affect melatonin secretion or wake-promoting neurons.
Damage to arousal-promoting neurons could help explain the reason the link between TBI and sleep disorders was strongest for insomnia and hypersomnia, although the exact mechanism is unclear, said Dr. Leng.
Greater risk with mild TBI
Overall, the association was stronger for mild TBI than for moderate to severe TBI. This, said Dr. Leng, might be because of differences in the brain injury mechanism.
Mild TBI often involves repetitive concussive or subconcussive injuries, such as sports injuries or blast injury among active-duty military personnel. This type of injury is more likely to cause diffuse axonal injury and inflammation, whereas moderate or severe TBI is often attributable to a direct blow with more focal but severe damage, explained Dr. Leng.
She noted that veterans with mild TBI were more likely to have a psychiatric condition, but because the study controlled for such conditions, this doesn’t fully explain the stronger association between mild TBI and sleep disorders.
Further studies are needed to sort out the exact mechanisms, she said.
The association between TBI and risk for sleep disorders was reduced somewhat but was still moderate in an analysis that excluded patients who developed a sleep disorder within 2 years of a brain injury.
This analysis, said Dr. Leng, helped ensure that the sleep disorder developed after the brain injury.
The researchers could not examine the trajectory of sleep problems, so it’s not clear whether sleep problems worsen or get better over time, said Dr. Leng.
Because PTSD also leads to sleep problems, the researchers thought that having both PTSD and TBI might increase the risk for sleep problems. “But actually we found the association was pretty similar in those with, and without, PTSD, so that was kind of contrary to our hypothesis,” she said.
The new results underline the need for more screening for sleep disorders among patients with TBI, both in the short term and the long term, said Dr. Leng. “Clinicians should ask TBI patients about their sleep, and they should follow that up,” she said.
She added that long-term sleep disorders can affect a patient’s health and can lead to psychiatric problems and neurodegenerative diseases.
Depending on the type of sleep disorder, there are a number of possible treatments. For example, for patients with sleep apnea, continuous positive airway pressure treatment may be considered.
‘Outstanding’ research
Commenting for this news organization, Frank Conidi, MD, director, Florida Center for Headache and Sports Neurology; CEO, Brainsport, Team Neurologist, the Florida Panthers of the National Hockey League; and past president, Florida Society of Neurology, said the study is “by far” the largest to investigate the correlation between sleep disorders and head trauma.
The design and outcome measures “were well thought out,” and the researchers “did an outstanding job in sorting through and analyzing the data,” said Dr. Conidi.
He added that he was particularly impressed with how the researchers addressed PTSD, which is highly prevalent among veterans with head trauma and is known to affect sleep.
The new results “solidify what those of us who see individuals with TBI have observed over the years: that there is a higher incidence of all types of sleep disorders” in individuals with a TBI, said Dr. Conidi.
However, he questioned the study’s use of guidelines to classify the various types of head trauma. These guidelines, he said, “are based on loss of consciousness, which we have started to move away from when classifying TBI.”
In addition, Dr. Conidi said he “would have loved to have seen” some correlation with neuroimaging studies, such as those used to assess subdural hematoma, epidural hematoma, subarachnoid hemorrhage, and diffuse axonal injury, but that this “could be an impetus for future studies.”
In “a perfect world,” all patients with a TBI would undergo a polysomnography study in a sleep laboratory, but insurance companies now rarely cover such studies and have attempted to have clinicians shift to home sleep studies, said Dr. Conidi. “These are marginal at best for screening for sleep disorders,” he noted.
At his centers, every TBI patient is screened for sleep disorders and, whenever possible, undergoes formal evaluation in the sleep lab, he added.
The study was supported by the U.S. Army Medical Research and Material Command and the U.S. Department of Veterans Affairs. Dr. Leng and Dr. Conidi have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
JAMA editor resigns over controversial podcast
JAMA editor in chief Howard Bauchner, MD, apologized to JAMA staff and stakeholders and asked for and received Dr. Livingston’s resignation, according to a statement from AMA CEO James Madara.
More than 2,000 people have signed a petition on Change.org calling for an investigation at JAMA over the podcast, called “Structural Racism for Doctors: What Is It?”
It appears they are now getting their wish. Dr. Bauchner announced that the journal’s oversight committee is investigating how the podcast and a tweet promoting the episode were developed, reviewed, and ultimately posted.
“This investigation and report of its findings will be thorough and completed rapidly,” Dr. Bauchner said.
Dr. Livingston, the host of the podcast, has been heavily criticized across social media. During the podcast, Dr. Livingston, who is White, said: “Structural racism is an unfortunate term. Personally, I think taking racism out of the conversation will help. Many of us are offended by the concept that we are racist.”
The audio of the podcast has been deleted from JAMA’s website. In its place is audio of a statement from Dr. Bauchner. In his statement, which he released last week, he said the comments in the podcast, which also featured Mitch Katz, MD, were “inaccurate, offensive, hurtful, and inconsistent with the standards of JAMA.”
Dr. Katz is an editor at JAMA Internal Medicine and CEO of NYC Health + Hospitals in New York.
Also deleted was a JAMA tweet promoting the podcast episode. The tweet said: “No physician is racist, so how can there be structural racism in health care? An explanation of the idea by doctors for doctors in this user-friendly podcast.”
The incident was met with anger and confusion in the medical community.
Herbert C. Smitherman, MD, vice dean of diversity and community affairs at Wayne State University, Detroit, noted after hearing the podcast that it was a symptom of a much larger problem.
“At its core, this podcast had racist tendencies. Those attitudes are why you don’t have as many articles by Black and Brown people in JAMA,” he said. “People’s attitudes, whether conscious or unconscious, are what drive the policies and practices which create the structural racism.”
Dr. Katz responded to the backlash last week with the following statement: “Systemic racism exists in our country. The disparate effects of the pandemic have made this painfully clear in New York City and across the country.
“As clinicians, we must understand how these structures and policies have a direct impact on the health outcomes of the patients and communities we serve. It is woefully naive to say that no physician is a racist just because the Civil Rights Act of 1964 forbade it, or that we should avoid the term ‘systematic racism’ because it makes people uncomfortable. We must and can do better.”
JAMA, an independent arm of the AMA, is taking other steps to address concerns. Its executive publisher, Thomas Easley, held an employee town hall this week, and said JAMA acknowledges that “structural racism is real, pernicious, and pervasive in health care.” The journal is also starting an “end-to-end review” of all editorial processes across all JAMA publications. Finally, the journal will also create a new associate editor’s position who will provide “insight and counsel” on racism and structural racism in health care.
A version of this article first appeared on WebMD.com .
JAMA editor in chief Howard Bauchner, MD, apologized to JAMA staff and stakeholders and asked for and received Dr. Livingston’s resignation, according to a statement from AMA CEO James Madara.
More than 2,000 people have signed a petition on Change.org calling for an investigation at JAMA over the podcast, called “Structural Racism for Doctors: What Is It?”
It appears they are now getting their wish. Dr. Bauchner announced that the journal’s oversight committee is investigating how the podcast and a tweet promoting the episode were developed, reviewed, and ultimately posted.
“This investigation and report of its findings will be thorough and completed rapidly,” Dr. Bauchner said.
Dr. Livingston, the host of the podcast, has been heavily criticized across social media. During the podcast, Dr. Livingston, who is White, said: “Structural racism is an unfortunate term. Personally, I think taking racism out of the conversation will help. Many of us are offended by the concept that we are racist.”
The audio of the podcast has been deleted from JAMA’s website. In its place is audio of a statement from Dr. Bauchner. In his statement, which he released last week, he said the comments in the podcast, which also featured Mitch Katz, MD, were “inaccurate, offensive, hurtful, and inconsistent with the standards of JAMA.”
Dr. Katz is an editor at JAMA Internal Medicine and CEO of NYC Health + Hospitals in New York.
Also deleted was a JAMA tweet promoting the podcast episode. The tweet said: “No physician is racist, so how can there be structural racism in health care? An explanation of the idea by doctors for doctors in this user-friendly podcast.”
The incident was met with anger and confusion in the medical community.
Herbert C. Smitherman, MD, vice dean of diversity and community affairs at Wayne State University, Detroit, noted after hearing the podcast that it was a symptom of a much larger problem.
“At its core, this podcast had racist tendencies. Those attitudes are why you don’t have as many articles by Black and Brown people in JAMA,” he said. “People’s attitudes, whether conscious or unconscious, are what drive the policies and practices which create the structural racism.”
Dr. Katz responded to the backlash last week with the following statement: “Systemic racism exists in our country. The disparate effects of the pandemic have made this painfully clear in New York City and across the country.
“As clinicians, we must understand how these structures and policies have a direct impact on the health outcomes of the patients and communities we serve. It is woefully naive to say that no physician is a racist just because the Civil Rights Act of 1964 forbade it, or that we should avoid the term ‘systematic racism’ because it makes people uncomfortable. We must and can do better.”
JAMA, an independent arm of the AMA, is taking other steps to address concerns. Its executive publisher, Thomas Easley, held an employee town hall this week, and said JAMA acknowledges that “structural racism is real, pernicious, and pervasive in health care.” The journal is also starting an “end-to-end review” of all editorial processes across all JAMA publications. Finally, the journal will also create a new associate editor’s position who will provide “insight and counsel” on racism and structural racism in health care.
A version of this article first appeared on WebMD.com .
JAMA editor in chief Howard Bauchner, MD, apologized to JAMA staff and stakeholders and asked for and received Dr. Livingston’s resignation, according to a statement from AMA CEO James Madara.
More than 2,000 people have signed a petition on Change.org calling for an investigation at JAMA over the podcast, called “Structural Racism for Doctors: What Is It?”
It appears they are now getting their wish. Dr. Bauchner announced that the journal’s oversight committee is investigating how the podcast and a tweet promoting the episode were developed, reviewed, and ultimately posted.
“This investigation and report of its findings will be thorough and completed rapidly,” Dr. Bauchner said.
Dr. Livingston, the host of the podcast, has been heavily criticized across social media. During the podcast, Dr. Livingston, who is White, said: “Structural racism is an unfortunate term. Personally, I think taking racism out of the conversation will help. Many of us are offended by the concept that we are racist.”
The audio of the podcast has been deleted from JAMA’s website. In its place is audio of a statement from Dr. Bauchner. In his statement, which he released last week, he said the comments in the podcast, which also featured Mitch Katz, MD, were “inaccurate, offensive, hurtful, and inconsistent with the standards of JAMA.”
Dr. Katz is an editor at JAMA Internal Medicine and CEO of NYC Health + Hospitals in New York.
Also deleted was a JAMA tweet promoting the podcast episode. The tweet said: “No physician is racist, so how can there be structural racism in health care? An explanation of the idea by doctors for doctors in this user-friendly podcast.”
The incident was met with anger and confusion in the medical community.
Herbert C. Smitherman, MD, vice dean of diversity and community affairs at Wayne State University, Detroit, noted after hearing the podcast that it was a symptom of a much larger problem.
“At its core, this podcast had racist tendencies. Those attitudes are why you don’t have as many articles by Black and Brown people in JAMA,” he said. “People’s attitudes, whether conscious or unconscious, are what drive the policies and practices which create the structural racism.”
Dr. Katz responded to the backlash last week with the following statement: “Systemic racism exists in our country. The disparate effects of the pandemic have made this painfully clear in New York City and across the country.
“As clinicians, we must understand how these structures and policies have a direct impact on the health outcomes of the patients and communities we serve. It is woefully naive to say that no physician is a racist just because the Civil Rights Act of 1964 forbade it, or that we should avoid the term ‘systematic racism’ because it makes people uncomfortable. We must and can do better.”
JAMA, an independent arm of the AMA, is taking other steps to address concerns. Its executive publisher, Thomas Easley, held an employee town hall this week, and said JAMA acknowledges that “structural racism is real, pernicious, and pervasive in health care.” The journal is also starting an “end-to-end review” of all editorial processes across all JAMA publications. Finally, the journal will also create a new associate editor’s position who will provide “insight and counsel” on racism and structural racism in health care.
A version of this article first appeared on WebMD.com .
Smartphone ‘addiction’ tied to poor sleep in young adults
Smartphone “addiction” may explain poor sleep quality in a significant proportion of young adults, new research suggests.
Investigators found that almost 40% of adults aged 18-30 years who self-reported excessive smartphone use also reported poor sleep.
“Our study provides further support to the growing body of evidence that smartphone addiction has a deleterious impact on sleep,” wrote the researchers.
The study was published online March 2 in Frontiers of Psychiatry.
Not a clinical diagnosis
Smartphone addiction is not formally recognized as a clinical diagnosis, but it’s an “active” area of research, lead investigator Ben Carter, PhD, King’s College London, noted in the report.
In a cross-sectional survey, 1,043 college students (aged 18-30 years, 73% women) completed the 10-question validated Smartphone Addiction Scale Short Version (SAS-SV) and the adapted Pittsburgh Sleep Quality Score Index.
On the SAS-SV, 406 students (38.9%) reported “addiction” to their smartphones. This estimated prevalence is consistent with other reported studies in young adult populations globally, which is in the range of 30%-45%, the researchers noted.
Overall, 61.6% of participants surveyed reported poor sleep; among those who reported smartphone addiction, 68.7% had poor sleep quality, vs. 57.1% of those who did not report smartphone addiction.
In multivariable analysis that adjusted for a variety of relevant factors, among those for whom there was evidence of smartphone addiction, the odds of poor sleep were increased by 41% (adjusted odds ratio [aOR] = 1.41; 95% confidence interval, 1.06-1.87, P = .018).
The findings also suggest that a greater amount of time spent using the phone and greater use late at night can raise the risk for smartphone addiction.
“Should smartphone addiction become firmly established as a focus of clinical concern, those using their phones after midnight or using their phones for four or more hours per day are likely to be at high risk, and should guide administration of the SAS-SV,” the researchers wrote.
Caveats, cautions, and concerns
Reached for comment, Paul Weigle, MD, psychiatrist with Hartford HealthCare and Hartford (Conn.) Hospital, and member of the American Academy of Child and Adolescent Psychiatry, said the finding of a relationship between addictive smartphone usage and poor sleep quality is not surprising.
“Great increases in adolescent screen media habits in recent decades have seen a concurrent increase in rates of insomnia among this population,” he said in an interview.
Dr. Weigle also noted that young people who use the phone excessively often do so in bed, “which decreases sleep onset by disrupting conditioning (the tendency for our bodies to relate bed with sleep) and by increasing physiological arousal, which makes it more difficult to fall asleep. The blue light from smartphones used at night disrupts our body’s natural circadian rhythms, confusing our brains regarding whether it is night or day, and further worsens sleep.”
Dr. Weigle said in an interview that some of his patients come to him seeking sleep medications, although the best treatment is to perform a “smartphone-ectomy” every evening.
Teenage patients will “beg, borrow, or steal” to be allowed to keep their phones by the bed with the promise not to use them overnight. Three-quarters of the time, when the parents are able to charge the phone in another room, “the sleep problem resolves,” Dr. Weigle said.
One caveat, he said, is that it’s “somewhat unclear whether this is best classified as an addiction or simply a seriously problematic habit. Either way, this type of habit causes a great deal of distress and dysfunction in the lives of those it affects, so it is important to understand,” he said.
In a statement, Bob Patton, PhD, lecturer in clinical psychology, University of Surrey, Guildford, England, noted that this is a cross-sectional study “and as such cannot lead to any firm conclusions about phone usage as the cause of reduced sleep quality.
“It does, however, provide some compelling evidence,” Dr. Patton said, “that the nature of smartphone usage and its related consequences are important considerations in addressing the emerging phenomenon of ‘smartphone addiction.’ ”
Also weighing in, Andrew Przybylski, PhD, director of research, Oxford (England) Internet Institute, University of Oxford, said by any global health body and is not a psychiatric disorder.
“The study is a correlational analysis of a sample of participants recruited on university campuses and therefore only reflects the experiences of those who had the purpose of the study explained to them. It can say nothing about behaviors in the general population,” Dr. Przybylski said in a statement.
“Readers should be cautious of making any firm conclusions about the impact of smartphone use in the general population, or the idea that they’re addictive in any objective sense, on the basis of this work,” he added. The study had no specific funding. Dr. Carter, Dr. Weigle, Dr. Patton, and Dr. Przybylski have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Smartphone “addiction” may explain poor sleep quality in a significant proportion of young adults, new research suggests.
Investigators found that almost 40% of adults aged 18-30 years who self-reported excessive smartphone use also reported poor sleep.
“Our study provides further support to the growing body of evidence that smartphone addiction has a deleterious impact on sleep,” wrote the researchers.
The study was published online March 2 in Frontiers of Psychiatry.
Not a clinical diagnosis
Smartphone addiction is not formally recognized as a clinical diagnosis, but it’s an “active” area of research, lead investigator Ben Carter, PhD, King’s College London, noted in the report.
In a cross-sectional survey, 1,043 college students (aged 18-30 years, 73% women) completed the 10-question validated Smartphone Addiction Scale Short Version (SAS-SV) and the adapted Pittsburgh Sleep Quality Score Index.
On the SAS-SV, 406 students (38.9%) reported “addiction” to their smartphones. This estimated prevalence is consistent with other reported studies in young adult populations globally, which is in the range of 30%-45%, the researchers noted.
Overall, 61.6% of participants surveyed reported poor sleep; among those who reported smartphone addiction, 68.7% had poor sleep quality, vs. 57.1% of those who did not report smartphone addiction.
In multivariable analysis that adjusted for a variety of relevant factors, among those for whom there was evidence of smartphone addiction, the odds of poor sleep were increased by 41% (adjusted odds ratio [aOR] = 1.41; 95% confidence interval, 1.06-1.87, P = .018).
The findings also suggest that a greater amount of time spent using the phone and greater use late at night can raise the risk for smartphone addiction.
“Should smartphone addiction become firmly established as a focus of clinical concern, those using their phones after midnight or using their phones for four or more hours per day are likely to be at high risk, and should guide administration of the SAS-SV,” the researchers wrote.
Caveats, cautions, and concerns
Reached for comment, Paul Weigle, MD, psychiatrist with Hartford HealthCare and Hartford (Conn.) Hospital, and member of the American Academy of Child and Adolescent Psychiatry, said the finding of a relationship between addictive smartphone usage and poor sleep quality is not surprising.
“Great increases in adolescent screen media habits in recent decades have seen a concurrent increase in rates of insomnia among this population,” he said in an interview.
Dr. Weigle also noted that young people who use the phone excessively often do so in bed, “which decreases sleep onset by disrupting conditioning (the tendency for our bodies to relate bed with sleep) and by increasing physiological arousal, which makes it more difficult to fall asleep. The blue light from smartphones used at night disrupts our body’s natural circadian rhythms, confusing our brains regarding whether it is night or day, and further worsens sleep.”
Dr. Weigle said in an interview that some of his patients come to him seeking sleep medications, although the best treatment is to perform a “smartphone-ectomy” every evening.
Teenage patients will “beg, borrow, or steal” to be allowed to keep their phones by the bed with the promise not to use them overnight. Three-quarters of the time, when the parents are able to charge the phone in another room, “the sleep problem resolves,” Dr. Weigle said.
One caveat, he said, is that it’s “somewhat unclear whether this is best classified as an addiction or simply a seriously problematic habit. Either way, this type of habit causes a great deal of distress and dysfunction in the lives of those it affects, so it is important to understand,” he said.
In a statement, Bob Patton, PhD, lecturer in clinical psychology, University of Surrey, Guildford, England, noted that this is a cross-sectional study “and as such cannot lead to any firm conclusions about phone usage as the cause of reduced sleep quality.
“It does, however, provide some compelling evidence,” Dr. Patton said, “that the nature of smartphone usage and its related consequences are important considerations in addressing the emerging phenomenon of ‘smartphone addiction.’ ”
Also weighing in, Andrew Przybylski, PhD, director of research, Oxford (England) Internet Institute, University of Oxford, said by any global health body and is not a psychiatric disorder.
“The study is a correlational analysis of a sample of participants recruited on university campuses and therefore only reflects the experiences of those who had the purpose of the study explained to them. It can say nothing about behaviors in the general population,” Dr. Przybylski said in a statement.
“Readers should be cautious of making any firm conclusions about the impact of smartphone use in the general population, or the idea that they’re addictive in any objective sense, on the basis of this work,” he added. The study had no specific funding. Dr. Carter, Dr. Weigle, Dr. Patton, and Dr. Przybylski have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Smartphone “addiction” may explain poor sleep quality in a significant proportion of young adults, new research suggests.
Investigators found that almost 40% of adults aged 18-30 years who self-reported excessive smartphone use also reported poor sleep.
“Our study provides further support to the growing body of evidence that smartphone addiction has a deleterious impact on sleep,” wrote the researchers.
The study was published online March 2 in Frontiers of Psychiatry.
Not a clinical diagnosis
Smartphone addiction is not formally recognized as a clinical diagnosis, but it’s an “active” area of research, lead investigator Ben Carter, PhD, King’s College London, noted in the report.
In a cross-sectional survey, 1,043 college students (aged 18-30 years, 73% women) completed the 10-question validated Smartphone Addiction Scale Short Version (SAS-SV) and the adapted Pittsburgh Sleep Quality Score Index.
On the SAS-SV, 406 students (38.9%) reported “addiction” to their smartphones. This estimated prevalence is consistent with other reported studies in young adult populations globally, which is in the range of 30%-45%, the researchers noted.
Overall, 61.6% of participants surveyed reported poor sleep; among those who reported smartphone addiction, 68.7% had poor sleep quality, vs. 57.1% of those who did not report smartphone addiction.
In multivariable analysis that adjusted for a variety of relevant factors, among those for whom there was evidence of smartphone addiction, the odds of poor sleep were increased by 41% (adjusted odds ratio [aOR] = 1.41; 95% confidence interval, 1.06-1.87, P = .018).
The findings also suggest that a greater amount of time spent using the phone and greater use late at night can raise the risk for smartphone addiction.
“Should smartphone addiction become firmly established as a focus of clinical concern, those using their phones after midnight or using their phones for four or more hours per day are likely to be at high risk, and should guide administration of the SAS-SV,” the researchers wrote.
Caveats, cautions, and concerns
Reached for comment, Paul Weigle, MD, psychiatrist with Hartford HealthCare and Hartford (Conn.) Hospital, and member of the American Academy of Child and Adolescent Psychiatry, said the finding of a relationship between addictive smartphone usage and poor sleep quality is not surprising.
“Great increases in adolescent screen media habits in recent decades have seen a concurrent increase in rates of insomnia among this population,” he said in an interview.
Dr. Weigle also noted that young people who use the phone excessively often do so in bed, “which decreases sleep onset by disrupting conditioning (the tendency for our bodies to relate bed with sleep) and by increasing physiological arousal, which makes it more difficult to fall asleep. The blue light from smartphones used at night disrupts our body’s natural circadian rhythms, confusing our brains regarding whether it is night or day, and further worsens sleep.”
Dr. Weigle said in an interview that some of his patients come to him seeking sleep medications, although the best treatment is to perform a “smartphone-ectomy” every evening.
Teenage patients will “beg, borrow, or steal” to be allowed to keep their phones by the bed with the promise not to use them overnight. Three-quarters of the time, when the parents are able to charge the phone in another room, “the sleep problem resolves,” Dr. Weigle said.
One caveat, he said, is that it’s “somewhat unclear whether this is best classified as an addiction or simply a seriously problematic habit. Either way, this type of habit causes a great deal of distress and dysfunction in the lives of those it affects, so it is important to understand,” he said.
In a statement, Bob Patton, PhD, lecturer in clinical psychology, University of Surrey, Guildford, England, noted that this is a cross-sectional study “and as such cannot lead to any firm conclusions about phone usage as the cause of reduced sleep quality.
“It does, however, provide some compelling evidence,” Dr. Patton said, “that the nature of smartphone usage and its related consequences are important considerations in addressing the emerging phenomenon of ‘smartphone addiction.’ ”
Also weighing in, Andrew Przybylski, PhD, director of research, Oxford (England) Internet Institute, University of Oxford, said by any global health body and is not a psychiatric disorder.
“The study is a correlational analysis of a sample of participants recruited on university campuses and therefore only reflects the experiences of those who had the purpose of the study explained to them. It can say nothing about behaviors in the general population,” Dr. Przybylski said in a statement.
“Readers should be cautious of making any firm conclusions about the impact of smartphone use in the general population, or the idea that they’re addictive in any objective sense, on the basis of this work,” he added. The study had no specific funding. Dr. Carter, Dr. Weigle, Dr. Patton, and Dr. Przybylski have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
‘Stunning’ report shows eating disorders are vastly underestimated
A “stunning” new analysis of global data on eating disorders show that they are far more prevalent and disabling than previously reported.
Investigators found the Global Burden of Diseases, Injuries, and Risk Factors Study (GBD) 2019 underestimated the prevalence of eating disorders by nearly 42 million cases, meaning these disorders are four times more common than previously reported.
“Our work highlights that eating disorders are far more prevalent and disabling than previously quantified,” lead author Damian F. Santomauro, PhD, University of Queensland and Center for Mental Health Research, Brisbane, Australia, said in an interview.
The study was published online March 3 in The Lancet Psychiatry.
Policy implications
The GBD Study 2019 reports the prevalence and burden of anorexia nervosa and bulimia nervosa under the umbrella of “eating disorders.”
However, binge-eating disorder (BED) and other specified feeding or eating disorder (OSFED) are more common, the investigators noted.
By excluding BED and OSFED, 41.9 million cases of eating disorders were not represented in the study.
The researchers calculate that the GBD 2019 overlooked 17.3 million people with BED and 24.6 million people with OSFED.
, bringing the total eating disorder DALYs to 6.6 million in 2019, they reported.
“When disorders are left out of the GBD, there is a risk that policymakers and service planners will interpret that these diseases are not prevalent or disabling and therefore not important to address,” said Dr. Santomauro.
“Our results show that the formal inclusion of binge-eating disorder and OSFED in GBD is both feasible and important and will lead to better representation of eating disorder burden globally.
“In turn, this will enhance recognition of the burden experienced by people living with these disorders and hopefully motivate increased investment in research, prevention, and treatment in future,” he added.
Landmark article, clarion call for action
In an accompanying commentary, Jennifer J. Thomas, PhD, and Kendra R. Becker, PhD, with the Eating Disorders Clinical and Research Program, Massachusetts General Hospital, Boston, said that this “stunning” analysis highlights that eating disorders are four times more common than previously thought.
This “landmark” analysis also demonstrates that BED and OSFED are especially common with increasing age. It highlights the burden of eating disorders in men, “shattering the inaccurate but entrenched stereotype that eating disorders affect only thin, young, White women,” Dr. Thomas and Dr. Becker pointed out.
This article, they wrote, is a “clarion call” for BED and OSFED to be included in future versions of the GBD Study.
Going a step further, Dr. Thomas and Dr. Becker said the GBD Study should also include estimates of the prevalence of avoidant/restrictive food intake disorder, rumination disorder, and pica and that the investigators should obtain direct measures of the disability associated with all feeding and eating disorders included in the DSM-5.
“If they do, the reported global burden will be even greater, underscoring the clear need for increased funding to study, prevent, and treat these debilitating illnesses,” they concluded.
The study was funded by Queensland Health, the Australian National Health and Medical Research Council, and the Bill & Melinda Gates Foundation. The authors have disclosed no relevant financial relationships. Disclosures for the editorialists are listed with the original article.
A version of this article first appeared on Medscape.com.
A “stunning” new analysis of global data on eating disorders show that they are far more prevalent and disabling than previously reported.
Investigators found the Global Burden of Diseases, Injuries, and Risk Factors Study (GBD) 2019 underestimated the prevalence of eating disorders by nearly 42 million cases, meaning these disorders are four times more common than previously reported.
“Our work highlights that eating disorders are far more prevalent and disabling than previously quantified,” lead author Damian F. Santomauro, PhD, University of Queensland and Center for Mental Health Research, Brisbane, Australia, said in an interview.
The study was published online March 3 in The Lancet Psychiatry.
Policy implications
The GBD Study 2019 reports the prevalence and burden of anorexia nervosa and bulimia nervosa under the umbrella of “eating disorders.”
However, binge-eating disorder (BED) and other specified feeding or eating disorder (OSFED) are more common, the investigators noted.
By excluding BED and OSFED, 41.9 million cases of eating disorders were not represented in the study.
The researchers calculate that the GBD 2019 overlooked 17.3 million people with BED and 24.6 million people with OSFED.
, bringing the total eating disorder DALYs to 6.6 million in 2019, they reported.
“When disorders are left out of the GBD, there is a risk that policymakers and service planners will interpret that these diseases are not prevalent or disabling and therefore not important to address,” said Dr. Santomauro.
“Our results show that the formal inclusion of binge-eating disorder and OSFED in GBD is both feasible and important and will lead to better representation of eating disorder burden globally.
“In turn, this will enhance recognition of the burden experienced by people living with these disorders and hopefully motivate increased investment in research, prevention, and treatment in future,” he added.
Landmark article, clarion call for action
In an accompanying commentary, Jennifer J. Thomas, PhD, and Kendra R. Becker, PhD, with the Eating Disorders Clinical and Research Program, Massachusetts General Hospital, Boston, said that this “stunning” analysis highlights that eating disorders are four times more common than previously thought.
This “landmark” analysis also demonstrates that BED and OSFED are especially common with increasing age. It highlights the burden of eating disorders in men, “shattering the inaccurate but entrenched stereotype that eating disorders affect only thin, young, White women,” Dr. Thomas and Dr. Becker pointed out.
This article, they wrote, is a “clarion call” for BED and OSFED to be included in future versions of the GBD Study.
Going a step further, Dr. Thomas and Dr. Becker said the GBD Study should also include estimates of the prevalence of avoidant/restrictive food intake disorder, rumination disorder, and pica and that the investigators should obtain direct measures of the disability associated with all feeding and eating disorders included in the DSM-5.
“If they do, the reported global burden will be even greater, underscoring the clear need for increased funding to study, prevent, and treat these debilitating illnesses,” they concluded.
The study was funded by Queensland Health, the Australian National Health and Medical Research Council, and the Bill & Melinda Gates Foundation. The authors have disclosed no relevant financial relationships. Disclosures for the editorialists are listed with the original article.
A version of this article first appeared on Medscape.com.
A “stunning” new analysis of global data on eating disorders show that they are far more prevalent and disabling than previously reported.
Investigators found the Global Burden of Diseases, Injuries, and Risk Factors Study (GBD) 2019 underestimated the prevalence of eating disorders by nearly 42 million cases, meaning these disorders are four times more common than previously reported.
“Our work highlights that eating disorders are far more prevalent and disabling than previously quantified,” lead author Damian F. Santomauro, PhD, University of Queensland and Center for Mental Health Research, Brisbane, Australia, said in an interview.
The study was published online March 3 in The Lancet Psychiatry.
Policy implications
The GBD Study 2019 reports the prevalence and burden of anorexia nervosa and bulimia nervosa under the umbrella of “eating disorders.”
However, binge-eating disorder (BED) and other specified feeding or eating disorder (OSFED) are more common, the investigators noted.
By excluding BED and OSFED, 41.9 million cases of eating disorders were not represented in the study.
The researchers calculate that the GBD 2019 overlooked 17.3 million people with BED and 24.6 million people with OSFED.
, bringing the total eating disorder DALYs to 6.6 million in 2019, they reported.
“When disorders are left out of the GBD, there is a risk that policymakers and service planners will interpret that these diseases are not prevalent or disabling and therefore not important to address,” said Dr. Santomauro.
“Our results show that the formal inclusion of binge-eating disorder and OSFED in GBD is both feasible and important and will lead to better representation of eating disorder burden globally.
“In turn, this will enhance recognition of the burden experienced by people living with these disorders and hopefully motivate increased investment in research, prevention, and treatment in future,” he added.
Landmark article, clarion call for action
In an accompanying commentary, Jennifer J. Thomas, PhD, and Kendra R. Becker, PhD, with the Eating Disorders Clinical and Research Program, Massachusetts General Hospital, Boston, said that this “stunning” analysis highlights that eating disorders are four times more common than previously thought.
This “landmark” analysis also demonstrates that BED and OSFED are especially common with increasing age. It highlights the burden of eating disorders in men, “shattering the inaccurate but entrenched stereotype that eating disorders affect only thin, young, White women,” Dr. Thomas and Dr. Becker pointed out.
This article, they wrote, is a “clarion call” for BED and OSFED to be included in future versions of the GBD Study.
Going a step further, Dr. Thomas and Dr. Becker said the GBD Study should also include estimates of the prevalence of avoidant/restrictive food intake disorder, rumination disorder, and pica and that the investigators should obtain direct measures of the disability associated with all feeding and eating disorders included in the DSM-5.
“If they do, the reported global burden will be even greater, underscoring the clear need for increased funding to study, prevent, and treat these debilitating illnesses,” they concluded.
The study was funded by Queensland Health, the Australian National Health and Medical Research Council, and the Bill & Melinda Gates Foundation. The authors have disclosed no relevant financial relationships. Disclosures for the editorialists are listed with the original article.
A version of this article first appeared on Medscape.com.
Delay surgery by 7 weeks after COVID-19 diagnosis, study shows
Seven weeks appears to be the ideal amount of time to delay surgery, when possible, after someone tests positive for COVID-19, researchers in the United Kingdom report.
Risk for death was about 3.5 to 4 times higher in the first 6 weeks after surgery among more than 3,000 people with a preoperative COVID-19 diagnosis compared with patients without COVID-19. After 7 weeks, the 30-day mortality rate dropped to a baseline level.
The study was published online March 9 in Anaesthesia.
Surgery should be further delayed for people who remain symptomatic at 7 weeks post diagnosis, lead author Dmitri Nepogodiev, MBChB, said in an interview.
“In this group we recommend waiting until COVID-19 symptoms resolve, if possible. However, our study did not capture specific data on long COVID … so we are unable to make specific recommendations for this group,” said Dr. Nepogodiev, research fellow at the NIHR Global Health Research Unit on Global Surgery at the University of Birmingham (England).
“This should be an area for future research,” he added.
The international, multicenter, prospective cohort study is notable for its sheer size – more than 15,000 investigators reported outcomes for 140,231 surgical patients from 1,674 hospitals across 116 countries. In total, 2.2% of these patients tested positive for SARS-CoV-2 prior to surgery.
Surgery of any type performed in October 2020 was assessed. A greater proportion of patients with a preoperative COVID-19 diagnosis had emergency surgery, 44%, compared with 30% of people who never had a COVID-19 diagnosis.
Most patients were asymptomatic at the time of surgery, either because they never experienced COVID-19 symptoms or their symptoms resolved. The 30-day mortality rate was the primary outcome.
Death rates among surgical patients with preoperative COVID-19 diagnosis
Comparing the timing of surgery after COVID-19 diagnosis vs. 30-day mortality yielded the following results:
- 0 to 2 weeks – 9.1% mortality.
- 3 to 4 weeks – 6.9%.
- 5 to 6 weeks – 5.5%.
- 7 weeks or longer – 2.0%..
For comparison, the 30-day mortality rate for surgical patients without a preoperative COVID-19 diagnosis was 1.4%. A COVID-19 diagnosis more than 7 weeks before surgery did not make a significant difference on outcomes.
The ‘why’ remains unknown
The reasons for the association between a COVID-19 diagnosis and higher postoperative death rates remain unknown. However, Dr. Nepogodiev speculated that it could be related to “some degree of lung injury, even if patients are initially asymptomatic.”
Intubation and mechanical ventilation during surgery could exacerbate the existing lung injury, he said, thereby leading to more severe COVID-19.
In fact, Dr. Nepogodiev and colleagues found that postoperative pulmonary complications followed a pattern similar to the findings on death. They reported higher rates of pneumonia, acute respiratory distress syndrome, and unexpected reventilation in the first 6 weeks following a COVID-19 diagnosis. Again, at 7 weeks and beyond, the rates returned to be relatively the same as those for people who never had COVID-19.
“Waiting for 7 or more weeks may allow time for the initial COVID-19 injury to resolve,” Dr. Nepogodiev said.
‘An important study’
“This is an important study of postoperative mortality among patients recovered from COVID-19,” Adrian Diaz, MD, MPH, said in an interview when asked to comment.
The large cohort and numerous practice settings are among the strengths of the research, said Dr. Diaz, of the University of Michigan Institute for Healthcare Policy and Innovation in Ann Arbor. He was lead author of a June 2020 review article on elective surgery in the time of COVID-19, published in The American Journal of Surgery.
“As with nearly all studies of this nature, results must be interpreted on a case-by-case basis for individual patients. However, this study does add important information for patients and providers in helping them have an informed discussion on the timing of surgery,” said Dr. Diaz, a fellow in the Center for Healthcare Outcomes and Policy and a resident in general surgery at the Ohio State University, Columbus.
Dr. Nepogodiev and colleagues included both urgent and elective surgeries in the study. Dr. Diaz said this was a potential limitation because emergency operations “should never be delayed, by definition.” Lack of indications for the surgeries and information on cause of death were additional limitations.
Future research should evaluate any benefit in delaying surgery longer than 7 or more weeks, Dr. Diaz added, perhaps looking specifically at 10, 12, or 14 weeks, or considering outcomes as a continuous variable. This would help health care providers “garner more insight into risk and benefits of delaying surgery beyond 7 weeks.”
Dr. Nepogodiev and Dr. Diaz disclosed no relevant financial relationships. The study had multiple funding sources, including the National Institute for Health Research Global Health Research Unit, the Association of Upper Gastrointestinal Surgeons, the British Association of Surgical Oncology, and Medtronic.
A version of this article first appeared on Medscape.com.
Seven weeks appears to be the ideal amount of time to delay surgery, when possible, after someone tests positive for COVID-19, researchers in the United Kingdom report.
Risk for death was about 3.5 to 4 times higher in the first 6 weeks after surgery among more than 3,000 people with a preoperative COVID-19 diagnosis compared with patients without COVID-19. After 7 weeks, the 30-day mortality rate dropped to a baseline level.
The study was published online March 9 in Anaesthesia.
Surgery should be further delayed for people who remain symptomatic at 7 weeks post diagnosis, lead author Dmitri Nepogodiev, MBChB, said in an interview.
“In this group we recommend waiting until COVID-19 symptoms resolve, if possible. However, our study did not capture specific data on long COVID … so we are unable to make specific recommendations for this group,” said Dr. Nepogodiev, research fellow at the NIHR Global Health Research Unit on Global Surgery at the University of Birmingham (England).
“This should be an area for future research,” he added.
The international, multicenter, prospective cohort study is notable for its sheer size – more than 15,000 investigators reported outcomes for 140,231 surgical patients from 1,674 hospitals across 116 countries. In total, 2.2% of these patients tested positive for SARS-CoV-2 prior to surgery.
Surgery of any type performed in October 2020 was assessed. A greater proportion of patients with a preoperative COVID-19 diagnosis had emergency surgery, 44%, compared with 30% of people who never had a COVID-19 diagnosis.
Most patients were asymptomatic at the time of surgery, either because they never experienced COVID-19 symptoms or their symptoms resolved. The 30-day mortality rate was the primary outcome.
Death rates among surgical patients with preoperative COVID-19 diagnosis
Comparing the timing of surgery after COVID-19 diagnosis vs. 30-day mortality yielded the following results:
- 0 to 2 weeks – 9.1% mortality.
- 3 to 4 weeks – 6.9%.
- 5 to 6 weeks – 5.5%.
- 7 weeks or longer – 2.0%..
For comparison, the 30-day mortality rate for surgical patients without a preoperative COVID-19 diagnosis was 1.4%. A COVID-19 diagnosis more than 7 weeks before surgery did not make a significant difference on outcomes.
The ‘why’ remains unknown
The reasons for the association between a COVID-19 diagnosis and higher postoperative death rates remain unknown. However, Dr. Nepogodiev speculated that it could be related to “some degree of lung injury, even if patients are initially asymptomatic.”
Intubation and mechanical ventilation during surgery could exacerbate the existing lung injury, he said, thereby leading to more severe COVID-19.
In fact, Dr. Nepogodiev and colleagues found that postoperative pulmonary complications followed a pattern similar to the findings on death. They reported higher rates of pneumonia, acute respiratory distress syndrome, and unexpected reventilation in the first 6 weeks following a COVID-19 diagnosis. Again, at 7 weeks and beyond, the rates returned to be relatively the same as those for people who never had COVID-19.
“Waiting for 7 or more weeks may allow time for the initial COVID-19 injury to resolve,” Dr. Nepogodiev said.
‘An important study’
“This is an important study of postoperative mortality among patients recovered from COVID-19,” Adrian Diaz, MD, MPH, said in an interview when asked to comment.
The large cohort and numerous practice settings are among the strengths of the research, said Dr. Diaz, of the University of Michigan Institute for Healthcare Policy and Innovation in Ann Arbor. He was lead author of a June 2020 review article on elective surgery in the time of COVID-19, published in The American Journal of Surgery.
“As with nearly all studies of this nature, results must be interpreted on a case-by-case basis for individual patients. However, this study does add important information for patients and providers in helping them have an informed discussion on the timing of surgery,” said Dr. Diaz, a fellow in the Center for Healthcare Outcomes and Policy and a resident in general surgery at the Ohio State University, Columbus.
Dr. Nepogodiev and colleagues included both urgent and elective surgeries in the study. Dr. Diaz said this was a potential limitation because emergency operations “should never be delayed, by definition.” Lack of indications for the surgeries and information on cause of death were additional limitations.
Future research should evaluate any benefit in delaying surgery longer than 7 or more weeks, Dr. Diaz added, perhaps looking specifically at 10, 12, or 14 weeks, or considering outcomes as a continuous variable. This would help health care providers “garner more insight into risk and benefits of delaying surgery beyond 7 weeks.”
Dr. Nepogodiev and Dr. Diaz disclosed no relevant financial relationships. The study had multiple funding sources, including the National Institute for Health Research Global Health Research Unit, the Association of Upper Gastrointestinal Surgeons, the British Association of Surgical Oncology, and Medtronic.
A version of this article first appeared on Medscape.com.
Seven weeks appears to be the ideal amount of time to delay surgery, when possible, after someone tests positive for COVID-19, researchers in the United Kingdom report.
Risk for death was about 3.5 to 4 times higher in the first 6 weeks after surgery among more than 3,000 people with a preoperative COVID-19 diagnosis compared with patients without COVID-19. After 7 weeks, the 30-day mortality rate dropped to a baseline level.
The study was published online March 9 in Anaesthesia.
Surgery should be further delayed for people who remain symptomatic at 7 weeks post diagnosis, lead author Dmitri Nepogodiev, MBChB, said in an interview.
“In this group we recommend waiting until COVID-19 symptoms resolve, if possible. However, our study did not capture specific data on long COVID … so we are unable to make specific recommendations for this group,” said Dr. Nepogodiev, research fellow at the NIHR Global Health Research Unit on Global Surgery at the University of Birmingham (England).
“This should be an area for future research,” he added.
The international, multicenter, prospective cohort study is notable for its sheer size – more than 15,000 investigators reported outcomes for 140,231 surgical patients from 1,674 hospitals across 116 countries. In total, 2.2% of these patients tested positive for SARS-CoV-2 prior to surgery.
Surgery of any type performed in October 2020 was assessed. A greater proportion of patients with a preoperative COVID-19 diagnosis had emergency surgery, 44%, compared with 30% of people who never had a COVID-19 diagnosis.
Most patients were asymptomatic at the time of surgery, either because they never experienced COVID-19 symptoms or their symptoms resolved. The 30-day mortality rate was the primary outcome.
Death rates among surgical patients with preoperative COVID-19 diagnosis
Comparing the timing of surgery after COVID-19 diagnosis vs. 30-day mortality yielded the following results:
- 0 to 2 weeks – 9.1% mortality.
- 3 to 4 weeks – 6.9%.
- 5 to 6 weeks – 5.5%.
- 7 weeks or longer – 2.0%..
For comparison, the 30-day mortality rate for surgical patients without a preoperative COVID-19 diagnosis was 1.4%. A COVID-19 diagnosis more than 7 weeks before surgery did not make a significant difference on outcomes.
The ‘why’ remains unknown
The reasons for the association between a COVID-19 diagnosis and higher postoperative death rates remain unknown. However, Dr. Nepogodiev speculated that it could be related to “some degree of lung injury, even if patients are initially asymptomatic.”
Intubation and mechanical ventilation during surgery could exacerbate the existing lung injury, he said, thereby leading to more severe COVID-19.
In fact, Dr. Nepogodiev and colleagues found that postoperative pulmonary complications followed a pattern similar to the findings on death. They reported higher rates of pneumonia, acute respiratory distress syndrome, and unexpected reventilation in the first 6 weeks following a COVID-19 diagnosis. Again, at 7 weeks and beyond, the rates returned to be relatively the same as those for people who never had COVID-19.
“Waiting for 7 or more weeks may allow time for the initial COVID-19 injury to resolve,” Dr. Nepogodiev said.
‘An important study’
“This is an important study of postoperative mortality among patients recovered from COVID-19,” Adrian Diaz, MD, MPH, said in an interview when asked to comment.
The large cohort and numerous practice settings are among the strengths of the research, said Dr. Diaz, of the University of Michigan Institute for Healthcare Policy and Innovation in Ann Arbor. He was lead author of a June 2020 review article on elective surgery in the time of COVID-19, published in The American Journal of Surgery.
“As with nearly all studies of this nature, results must be interpreted on a case-by-case basis for individual patients. However, this study does add important information for patients and providers in helping them have an informed discussion on the timing of surgery,” said Dr. Diaz, a fellow in the Center for Healthcare Outcomes and Policy and a resident in general surgery at the Ohio State University, Columbus.
Dr. Nepogodiev and colleagues included both urgent and elective surgeries in the study. Dr. Diaz said this was a potential limitation because emergency operations “should never be delayed, by definition.” Lack of indications for the surgeries and information on cause of death were additional limitations.
Future research should evaluate any benefit in delaying surgery longer than 7 or more weeks, Dr. Diaz added, perhaps looking specifically at 10, 12, or 14 weeks, or considering outcomes as a continuous variable. This would help health care providers “garner more insight into risk and benefits of delaying surgery beyond 7 weeks.”
Dr. Nepogodiev and Dr. Diaz disclosed no relevant financial relationships. The study had multiple funding sources, including the National Institute for Health Research Global Health Research Unit, the Association of Upper Gastrointestinal Surgeons, the British Association of Surgical Oncology, and Medtronic.
A version of this article first appeared on Medscape.com.
Benefits of bremelanotide to women with HSDD questioned in analysis paper
Dr. Spielmans, professor of psychology at Metropolitan State University in Saint Paul, Minn., examined data from the FDA application for bremelanotide, clinicaltrials.gov entries for two phase 3 trials of the drug, and a 2019 article published in Obstetrics & Gynecology that described results from the 24-week trials.
In Dr. Speilman’s analysis, which was published online March 7 in the Journal of Sex Research, he notes that 42.1% of trial participants who received bremelanotide did not complete the trial, compared with 20.48% of participants who received placebo.
Of those who completed the study, 87.22% who received placebo wanted to continue treatment in an open-label extension, compared with 69.97% who received bremelanotide, he wrote.
Women “should be aware of the small degree of bremelanotide’s efficacy, that the protocol-specified outcomes of bremelanotide are mostly unknown, and that participants would rather take a placebo than bremelanotide,” Dr. Spielmans said.
Anita H. Clayton, MD, an author of the Obstetrics & Gynecology paper addressed in Dr. Spielmans’ analysis, says the Journal of Sex Research article does not provide new information and is a disservice to women because it questions accurate scientific data.
Measuring outcomes in HSDD is an evolving field, Dr. Clayton, a psychiatrist at the University of Virginia in Charlottesville, said in an interview. Initial FDA guidance relied on satisfying sexual events as an outcome measure, but this measure was derived from erectile dysfunction studies and is not necessarily adequate for assessing HSDD, she said. The FDA and drug developers agreed to use the desire subscale of the Female Sexual Function Index (FSFI-D) as a coprimary outcome measure instead, she noted.
Dr. Spielmans’ critique of Obstetrics & Gynecology paper
The article published in Obstetrics & Gynecology reporting bremelanotide trial results was noteworthy, although the various issues involved can be seen in reports about other drug trials, Dr. Spielmans said in an interview.
“It is well-established that journal articles reporting clinical trial data overstate benefits and understate harms,” he continued. In this case, “the very incomplete data reporting, reliance on many post-hoc measures of questionable validity, hiding the concerning number of dropouts due to adverse events, and putting a positive spin on efficacy and tolerability is both remarkable and highly problematic,” Dr. Spielmans said.
Dr. Clayton’s reaction
Data about dropout rates due to adverse events have been reported and presented at national meetings, she said in an interview. In addition, a questionnaire found that bremelanotide was superior to placebo in terms of patients feeling that the treatment had provided clinically meaningful benefit, Dr. Clayton said.
The available information enables patients to make informed treatment decisions, Dr. Clayton continued. “There is really this sexist attitude of women needing protection from their own decisions,” she said.
Diagnosing and treating HSDD
Eight of 11 efficacy outcomes in the clinicaltrials.gov study protocols for bremelanotide were not reported in the Obstetrics & Gynecology article in a way that was consistent with the protocols, Dr. Spielmans said. Changing a coprimary outcome to the key secondary outcome “occurred over a year after the trials had begun,” and the authors of the journal article “did not mention that this change occurred,” Dr. Spielmans wrote.
For the coprimary outcome measures of mean change on FSFI-D and Female Sexual Distress Scale–Desire/Arousal/Orgasm #13, “bremelanotide offers modest benefits over placebo,” Dr. Spielmans reported.
In addition to outlining his concerns about transparency in the reporting of trial data and raising questions about the outcome measures used in the Obstetrics & Gynecology article, Dr. Spielmans wrote that the diagnosis of HSDD is problematic.
“The lack of specifying symptom duration, questionable validity for the lack of sexual fantasies as a diagnostic criterion, difficulty in disentangling individual sexual problems from relational problems, and the failure to consider cultural influence (including the pressure on women to satisfy the sexual desires of their male partners) in the experience of sexuality all render HSDD as a problematic entity,” Dr. Spielmans wrote.
The fifth edition of the Diagnostic and Statistical Manual of Mental Disorders replaced HSDD and female sexual arousal disorder with the combined condition female sexual interest/arousal disorder. HSDD is in the 11th edition of the International Classification of Diseases and can be applied to men or women, Dr. Spielmans said.
FDA acknowledged HSDD as an unmet medical need
Dr. Clayton pointed out that HSDD was described decades ago and the FDA acknowledged it as an unmet medical need, and she expressed dissatisfaction with the fact the hypoactive sexual desire disorder appears with quotation marks around it in the title of Dr. Spielmans’ article. This way of presenting HSDD indicates that “the author has no concept of sexual health or sexual dysfunction,” Dr. Clayton said. “Basically this is sort of a dramatic tool, I think, to act like this is not a real disorder,” she added.
Carl Spana, PhD, CEO and president of Palatin Technologies, the developer of bremelanotide, defined the article in the Journal of Sex Research as a “retrospective meta-analysis, and not a re-analysis of the data.
“As a meta-analysis, it is open to various interpretations and reflects the author’s interpretations, which appear to have clear biases,” Dr. Spana said in an interview. “We believe several of this author’s interpretations are contrary to the FDA’s positive assessment that led to Vyleesi’s approval as a safe and effective treatment for women suffering from hypoactive sexual desire disorder.”
The author is unaware of the validation that was conducted at the direction of the FDA to establish clinically meaningful cutoffs for patient-reported outcomes and to establish metrics that define clinical benefit, Dr. Spana said
“Vyleesi was approved by the FDA after a thorough analysis of data from two well-controlled phase 3 clinical studies and multiple clinical and preclinical safety studies,” he said. “The analyses in the New Drug Application were prespecified and conducted according to a statistical analysis plan that the sponsor and FDA agreed to prior to database lock.”
Dr. Spielmans disclosed holdings in Vanguard Healthcare, a mutual fund that invests in pharmaceutical firms. Dr. Clayton has received financial support from Palatin and AMAG Pharmaceuticals, the companies that developed bremelanotide, in previous years.
Dr. Spielmans, professor of psychology at Metropolitan State University in Saint Paul, Minn., examined data from the FDA application for bremelanotide, clinicaltrials.gov entries for two phase 3 trials of the drug, and a 2019 article published in Obstetrics & Gynecology that described results from the 24-week trials.
In Dr. Speilman’s analysis, which was published online March 7 in the Journal of Sex Research, he notes that 42.1% of trial participants who received bremelanotide did not complete the trial, compared with 20.48% of participants who received placebo.
Of those who completed the study, 87.22% who received placebo wanted to continue treatment in an open-label extension, compared with 69.97% who received bremelanotide, he wrote.
Women “should be aware of the small degree of bremelanotide’s efficacy, that the protocol-specified outcomes of bremelanotide are mostly unknown, and that participants would rather take a placebo than bremelanotide,” Dr. Spielmans said.
Anita H. Clayton, MD, an author of the Obstetrics & Gynecology paper addressed in Dr. Spielmans’ analysis, says the Journal of Sex Research article does not provide new information and is a disservice to women because it questions accurate scientific data.
Measuring outcomes in HSDD is an evolving field, Dr. Clayton, a psychiatrist at the University of Virginia in Charlottesville, said in an interview. Initial FDA guidance relied on satisfying sexual events as an outcome measure, but this measure was derived from erectile dysfunction studies and is not necessarily adequate for assessing HSDD, she said. The FDA and drug developers agreed to use the desire subscale of the Female Sexual Function Index (FSFI-D) as a coprimary outcome measure instead, she noted.
Dr. Spielmans’ critique of Obstetrics & Gynecology paper
The article published in Obstetrics & Gynecology reporting bremelanotide trial results was noteworthy, although the various issues involved can be seen in reports about other drug trials, Dr. Spielmans said in an interview.
“It is well-established that journal articles reporting clinical trial data overstate benefits and understate harms,” he continued. In this case, “the very incomplete data reporting, reliance on many post-hoc measures of questionable validity, hiding the concerning number of dropouts due to adverse events, and putting a positive spin on efficacy and tolerability is both remarkable and highly problematic,” Dr. Spielmans said.
Dr. Clayton’s reaction
Data about dropout rates due to adverse events have been reported and presented at national meetings, she said in an interview. In addition, a questionnaire found that bremelanotide was superior to placebo in terms of patients feeling that the treatment had provided clinically meaningful benefit, Dr. Clayton said.
The available information enables patients to make informed treatment decisions, Dr. Clayton continued. “There is really this sexist attitude of women needing protection from their own decisions,” she said.
Diagnosing and treating HSDD
Eight of 11 efficacy outcomes in the clinicaltrials.gov study protocols for bremelanotide were not reported in the Obstetrics & Gynecology article in a way that was consistent with the protocols, Dr. Spielmans said. Changing a coprimary outcome to the key secondary outcome “occurred over a year after the trials had begun,” and the authors of the journal article “did not mention that this change occurred,” Dr. Spielmans wrote.
For the coprimary outcome measures of mean change on FSFI-D and Female Sexual Distress Scale–Desire/Arousal/Orgasm #13, “bremelanotide offers modest benefits over placebo,” Dr. Spielmans reported.
In addition to outlining his concerns about transparency in the reporting of trial data and raising questions about the outcome measures used in the Obstetrics & Gynecology article, Dr. Spielmans wrote that the diagnosis of HSDD is problematic.
“The lack of specifying symptom duration, questionable validity for the lack of sexual fantasies as a diagnostic criterion, difficulty in disentangling individual sexual problems from relational problems, and the failure to consider cultural influence (including the pressure on women to satisfy the sexual desires of their male partners) in the experience of sexuality all render HSDD as a problematic entity,” Dr. Spielmans wrote.
The fifth edition of the Diagnostic and Statistical Manual of Mental Disorders replaced HSDD and female sexual arousal disorder with the combined condition female sexual interest/arousal disorder. HSDD is in the 11th edition of the International Classification of Diseases and can be applied to men or women, Dr. Spielmans said.
FDA acknowledged HSDD as an unmet medical need
Dr. Clayton pointed out that HSDD was described decades ago and the FDA acknowledged it as an unmet medical need, and she expressed dissatisfaction with the fact the hypoactive sexual desire disorder appears with quotation marks around it in the title of Dr. Spielmans’ article. This way of presenting HSDD indicates that “the author has no concept of sexual health or sexual dysfunction,” Dr. Clayton said. “Basically this is sort of a dramatic tool, I think, to act like this is not a real disorder,” she added.
Carl Spana, PhD, CEO and president of Palatin Technologies, the developer of bremelanotide, defined the article in the Journal of Sex Research as a “retrospective meta-analysis, and not a re-analysis of the data.
“As a meta-analysis, it is open to various interpretations and reflects the author’s interpretations, which appear to have clear biases,” Dr. Spana said in an interview. “We believe several of this author’s interpretations are contrary to the FDA’s positive assessment that led to Vyleesi’s approval as a safe and effective treatment for women suffering from hypoactive sexual desire disorder.”
The author is unaware of the validation that was conducted at the direction of the FDA to establish clinically meaningful cutoffs for patient-reported outcomes and to establish metrics that define clinical benefit, Dr. Spana said
“Vyleesi was approved by the FDA after a thorough analysis of data from two well-controlled phase 3 clinical studies and multiple clinical and preclinical safety studies,” he said. “The analyses in the New Drug Application were prespecified and conducted according to a statistical analysis plan that the sponsor and FDA agreed to prior to database lock.”
Dr. Spielmans disclosed holdings in Vanguard Healthcare, a mutual fund that invests in pharmaceutical firms. Dr. Clayton has received financial support from Palatin and AMAG Pharmaceuticals, the companies that developed bremelanotide, in previous years.
Dr. Spielmans, professor of psychology at Metropolitan State University in Saint Paul, Minn., examined data from the FDA application for bremelanotide, clinicaltrials.gov entries for two phase 3 trials of the drug, and a 2019 article published in Obstetrics & Gynecology that described results from the 24-week trials.
In Dr. Speilman’s analysis, which was published online March 7 in the Journal of Sex Research, he notes that 42.1% of trial participants who received bremelanotide did not complete the trial, compared with 20.48% of participants who received placebo.
Of those who completed the study, 87.22% who received placebo wanted to continue treatment in an open-label extension, compared with 69.97% who received bremelanotide, he wrote.
Women “should be aware of the small degree of bremelanotide’s efficacy, that the protocol-specified outcomes of bremelanotide are mostly unknown, and that participants would rather take a placebo than bremelanotide,” Dr. Spielmans said.
Anita H. Clayton, MD, an author of the Obstetrics & Gynecology paper addressed in Dr. Spielmans’ analysis, says the Journal of Sex Research article does not provide new information and is a disservice to women because it questions accurate scientific data.
Measuring outcomes in HSDD is an evolving field, Dr. Clayton, a psychiatrist at the University of Virginia in Charlottesville, said in an interview. Initial FDA guidance relied on satisfying sexual events as an outcome measure, but this measure was derived from erectile dysfunction studies and is not necessarily adequate for assessing HSDD, she said. The FDA and drug developers agreed to use the desire subscale of the Female Sexual Function Index (FSFI-D) as a coprimary outcome measure instead, she noted.
Dr. Spielmans’ critique of Obstetrics & Gynecology paper
The article published in Obstetrics & Gynecology reporting bremelanotide trial results was noteworthy, although the various issues involved can be seen in reports about other drug trials, Dr. Spielmans said in an interview.
“It is well-established that journal articles reporting clinical trial data overstate benefits and understate harms,” he continued. In this case, “the very incomplete data reporting, reliance on many post-hoc measures of questionable validity, hiding the concerning number of dropouts due to adverse events, and putting a positive spin on efficacy and tolerability is both remarkable and highly problematic,” Dr. Spielmans said.
Dr. Clayton’s reaction
Data about dropout rates due to adverse events have been reported and presented at national meetings, she said in an interview. In addition, a questionnaire found that bremelanotide was superior to placebo in terms of patients feeling that the treatment had provided clinically meaningful benefit, Dr. Clayton said.
The available information enables patients to make informed treatment decisions, Dr. Clayton continued. “There is really this sexist attitude of women needing protection from their own decisions,” she said.
Diagnosing and treating HSDD
Eight of 11 efficacy outcomes in the clinicaltrials.gov study protocols for bremelanotide were not reported in the Obstetrics & Gynecology article in a way that was consistent with the protocols, Dr. Spielmans said. Changing a coprimary outcome to the key secondary outcome “occurred over a year after the trials had begun,” and the authors of the journal article “did not mention that this change occurred,” Dr. Spielmans wrote.
For the coprimary outcome measures of mean change on FSFI-D and Female Sexual Distress Scale–Desire/Arousal/Orgasm #13, “bremelanotide offers modest benefits over placebo,” Dr. Spielmans reported.
In addition to outlining his concerns about transparency in the reporting of trial data and raising questions about the outcome measures used in the Obstetrics & Gynecology article, Dr. Spielmans wrote that the diagnosis of HSDD is problematic.
“The lack of specifying symptom duration, questionable validity for the lack of sexual fantasies as a diagnostic criterion, difficulty in disentangling individual sexual problems from relational problems, and the failure to consider cultural influence (including the pressure on women to satisfy the sexual desires of their male partners) in the experience of sexuality all render HSDD as a problematic entity,” Dr. Spielmans wrote.
The fifth edition of the Diagnostic and Statistical Manual of Mental Disorders replaced HSDD and female sexual arousal disorder with the combined condition female sexual interest/arousal disorder. HSDD is in the 11th edition of the International Classification of Diseases and can be applied to men or women, Dr. Spielmans said.
FDA acknowledged HSDD as an unmet medical need
Dr. Clayton pointed out that HSDD was described decades ago and the FDA acknowledged it as an unmet medical need, and she expressed dissatisfaction with the fact the hypoactive sexual desire disorder appears with quotation marks around it in the title of Dr. Spielmans’ article. This way of presenting HSDD indicates that “the author has no concept of sexual health or sexual dysfunction,” Dr. Clayton said. “Basically this is sort of a dramatic tool, I think, to act like this is not a real disorder,” she added.
Carl Spana, PhD, CEO and president of Palatin Technologies, the developer of bremelanotide, defined the article in the Journal of Sex Research as a “retrospective meta-analysis, and not a re-analysis of the data.
“As a meta-analysis, it is open to various interpretations and reflects the author’s interpretations, which appear to have clear biases,” Dr. Spana said in an interview. “We believe several of this author’s interpretations are contrary to the FDA’s positive assessment that led to Vyleesi’s approval as a safe and effective treatment for women suffering from hypoactive sexual desire disorder.”
The author is unaware of the validation that was conducted at the direction of the FDA to establish clinically meaningful cutoffs for patient-reported outcomes and to establish metrics that define clinical benefit, Dr. Spana said
“Vyleesi was approved by the FDA after a thorough analysis of data from two well-controlled phase 3 clinical studies and multiple clinical and preclinical safety studies,” he said. “The analyses in the New Drug Application were prespecified and conducted according to a statistical analysis plan that the sponsor and FDA agreed to prior to database lock.”
Dr. Spielmans disclosed holdings in Vanguard Healthcare, a mutual fund that invests in pharmaceutical firms. Dr. Clayton has received financial support from Palatin and AMAG Pharmaceuticals, the companies that developed bremelanotide, in previous years.
FROM THE JOURNAL OF SEX RESEARCH
Novel therapeutic target for depression identified
An antiseizure medication appears to reduce anhedonia in patients with depression via a novel mechanism that may offer a new therapeutic target for the disorder, new research suggests.
Results of a small, randomized trial show those who received ezogabine (Potiga) experienced a significant reduction in key measures of depression and anhedonia versus placebo.
Participants in the treatment group also showed a trend toward increased response to reward anticipation on functional MRI (fMRI), compared with those treated with placebo, although the effect did not reach statistical significance.
“Our study was the first randomized, placebo-controlled trial to show that a drug affecting this kind of ion channel in the brain can improve depression and anhedonia in patients,” senior investigator James Murrough, MD, PhD, associate professor of psychiatry and neuroscience at the Icahn School of Medicine at Mount Sinai, New York, said in a press release.
“Targeting this channel represents a completely different mechanism of action than any currently available antidepressant treatment,” said Dr. Murrough, who is also director of the Depression and Anxiety Center for Discovery and Treatment at Mount Sinai.
The study was published online March 3 in the American Journal of Psychiatry.
Need for a novel target
“One of the main issues in treating depression is that many of our current antidepressants have similar mechanisms of action,” Dr. Murrough said in an interview. “Once a patient hasn’t responded to currently available agents, it’s hard think of new medications to fill that need.”
This need for a novel target motivated Dr. Murrough and associates to research the KCNQ2/3 potassium channel, which has not been previously studied as a therapeutic target for depression.
The KCNQ2/3 channel controls brain cell excitability and function by controlling the flow of the electrical charge across the cell membrane in the form of potassium ions, Dr. Murrough explained.
Previous research using a chronic social defeat model of depression in mice showed changes in the KCNQ2/3 channel. “This was key to determining whether a mouse showed depressive behavior in the context of stress, or whether the mice were resistant or resilient to stress,” he said.
Mice resistant to stress showed increased markers in brain regions associated with reward, while the less resilient mice showed excessive excitability and dysfunction. Dysfunction in the brain’s reward system leads to anhedonia, a “core feature” of depressive disorders.
This inspired Dr. Murrough’s group to identify ezogabine, a drug that acts on this channel.
“ for addressing depressive symptoms,” Dr. Murrough said
Nonsignificant trend
The researchers studied 45 adults diagnosed with depression who exhibited significant anhedonia and at least moderate illness severity.
Participants were randomly assigned to receive either ezogabine (n = 21, mean age 44 years at enrollment, 28.3% male) or placebo (n = 24, mean age 39 years at enrollment, 50% male). At baseline and following treatment, participants completed the incentive flanker test under fMRI conditions to model brain activity during anticipation of a reward. In addition, clinical measures of depression and anhedonia were assessed at weekly visits.
The study groups did not differ significantly in performance accuracy during the fMRI task at baseline or following treatment. The table below summarizes the percentage of errors in each group, with standard deviation.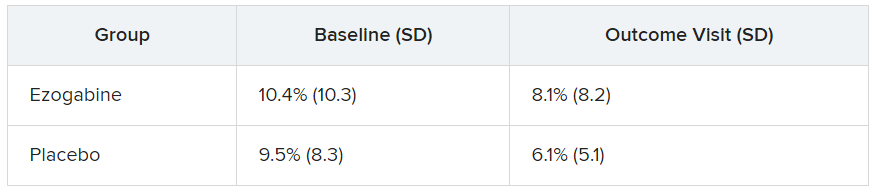
Participants in the ezogabine group showed a numerical increase in ventral striatum activation in response to reward anticipation, compared with participants in the placebo group, but this trend was not considered significant.
Heterogeneous condition
In contrast, there were notable improvements from baseline to the final outcome visit in clinical measures of depression and anhedonia in the ezogabine group, compared with the placebo group. Mean (SD) differences in depression scores, based on the MADRS (Montgomery-Åsberg Depression Rating Scale) from baseline to endpoint as follows: mean difference, –7.9 (3.0); effect size, 0.76; response rate, 61.9% (ezogabine) and 37.5% (placebo); remission rate: 38.1% (ezogabine) and 20.8% (placebo)
Compared with placebo, there were also large improvements in hedonic capacity, as measured by the Snaith-Hamilton Pleasure Scale and the anticipatory subscale of the Temporal Experience of Pleasure Scale (t, –4.1; df, 212; P < .001 and t, 3.4; df, 213; P < .001, respectively).
Compared with placebo, ezogabine was associated with “significant benefit” in global illness severity and improvement (Clinical Global Impression–Severity: t, –2.2; df, 214; P = .026 and CGI-Improvement: t, –2.9; d, 214; P = .004, respectively).
Ezogabine was well tolerated. Dizziness and headache were the most common adverse events.
Depression is a “heterogeneous condition” with a single diagnosis encompassing a “large, multifaceted” array of symptoms, Dr. Murrough noted. A growing body of research is focusing on specific components as potential treatment targets. “Our study looked specifically at patients with a diagnosis of depression but high scores on the anhedonia scale and we found that boosting the function of the KCNQ2/3 channel may have a beneficial antidepressant effect by improving anhedonia.”
Potential gain
In a comment, Alan Schatzberg, MD, professor in the department of psychiatry and behavioral sciences at Stanford (Calif.) University, said that “anytime there’s a new treatment with a new mechanism of action for a given condition, there’s a potential gain for the field.”
Dr. Schatzberg, who was not involved with the study, said that ezogabine, with its “potentially new mechanism of action, seems to have an effect and reasonable safety and could be important for patients who may not respond to traditional medications. It might also be important for all sorts of patients, depending on findings of later trials.”
Dr. Murrough said that ezogabine in still in “early stages” of research. “We hope that future studies will look at other agents that would also affect this channel,” he added.
This research was supported by the National Institute of Mental Health. Additional funding was provided by the Friedman Brain Institute at the Icahn School of Medicine at Mount Sinai and the Ehrenkranz Laboratory for Human Resilience, a component of the Depression and Anxiety Center for Discovery and Treatment at the Icahn School of Medicine at Mount Sinai. Dr. Murrough is an inventor of a pending patent application for the use of ezogabine and other KCNQ channel openers to treat depression and related disorders. Dr. Schatzberg disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
An antiseizure medication appears to reduce anhedonia in patients with depression via a novel mechanism that may offer a new therapeutic target for the disorder, new research suggests.
Results of a small, randomized trial show those who received ezogabine (Potiga) experienced a significant reduction in key measures of depression and anhedonia versus placebo.
Participants in the treatment group also showed a trend toward increased response to reward anticipation on functional MRI (fMRI), compared with those treated with placebo, although the effect did not reach statistical significance.
“Our study was the first randomized, placebo-controlled trial to show that a drug affecting this kind of ion channel in the brain can improve depression and anhedonia in patients,” senior investigator James Murrough, MD, PhD, associate professor of psychiatry and neuroscience at the Icahn School of Medicine at Mount Sinai, New York, said in a press release.
“Targeting this channel represents a completely different mechanism of action than any currently available antidepressant treatment,” said Dr. Murrough, who is also director of the Depression and Anxiety Center for Discovery and Treatment at Mount Sinai.
The study was published online March 3 in the American Journal of Psychiatry.
Need for a novel target
“One of the main issues in treating depression is that many of our current antidepressants have similar mechanisms of action,” Dr. Murrough said in an interview. “Once a patient hasn’t responded to currently available agents, it’s hard think of new medications to fill that need.”
This need for a novel target motivated Dr. Murrough and associates to research the KCNQ2/3 potassium channel, which has not been previously studied as a therapeutic target for depression.
The KCNQ2/3 channel controls brain cell excitability and function by controlling the flow of the electrical charge across the cell membrane in the form of potassium ions, Dr. Murrough explained.
Previous research using a chronic social defeat model of depression in mice showed changes in the KCNQ2/3 channel. “This was key to determining whether a mouse showed depressive behavior in the context of stress, or whether the mice were resistant or resilient to stress,” he said.
Mice resistant to stress showed increased markers in brain regions associated with reward, while the less resilient mice showed excessive excitability and dysfunction. Dysfunction in the brain’s reward system leads to anhedonia, a “core feature” of depressive disorders.
This inspired Dr. Murrough’s group to identify ezogabine, a drug that acts on this channel.
“ for addressing depressive symptoms,” Dr. Murrough said
Nonsignificant trend
The researchers studied 45 adults diagnosed with depression who exhibited significant anhedonia and at least moderate illness severity.
Participants were randomly assigned to receive either ezogabine (n = 21, mean age 44 years at enrollment, 28.3% male) or placebo (n = 24, mean age 39 years at enrollment, 50% male). At baseline and following treatment, participants completed the incentive flanker test under fMRI conditions to model brain activity during anticipation of a reward. In addition, clinical measures of depression and anhedonia were assessed at weekly visits.
The study groups did not differ significantly in performance accuracy during the fMRI task at baseline or following treatment. The table below summarizes the percentage of errors in each group, with standard deviation.
Participants in the ezogabine group showed a numerical increase in ventral striatum activation in response to reward anticipation, compared with participants in the placebo group, but this trend was not considered significant.
Heterogeneous condition
In contrast, there were notable improvements from baseline to the final outcome visit in clinical measures of depression and anhedonia in the ezogabine group, compared with the placebo group. Mean (SD) differences in depression scores, based on the MADRS (Montgomery-Åsberg Depression Rating Scale) from baseline to endpoint as follows: mean difference, –7.9 (3.0); effect size, 0.76; response rate, 61.9% (ezogabine) and 37.5% (placebo); remission rate: 38.1% (ezogabine) and 20.8% (placebo)
Compared with placebo, there were also large improvements in hedonic capacity, as measured by the Snaith-Hamilton Pleasure Scale and the anticipatory subscale of the Temporal Experience of Pleasure Scale (t, –4.1; df, 212; P < .001 and t, 3.4; df, 213; P < .001, respectively).
Compared with placebo, ezogabine was associated with “significant benefit” in global illness severity and improvement (Clinical Global Impression–Severity: t, –2.2; df, 214; P = .026 and CGI-Improvement: t, –2.9; d, 214; P = .004, respectively).
Ezogabine was well tolerated. Dizziness and headache were the most common adverse events.
Depression is a “heterogeneous condition” with a single diagnosis encompassing a “large, multifaceted” array of symptoms, Dr. Murrough noted. A growing body of research is focusing on specific components as potential treatment targets. “Our study looked specifically at patients with a diagnosis of depression but high scores on the anhedonia scale and we found that boosting the function of the KCNQ2/3 channel may have a beneficial antidepressant effect by improving anhedonia.”
Potential gain
In a comment, Alan Schatzberg, MD, professor in the department of psychiatry and behavioral sciences at Stanford (Calif.) University, said that “anytime there’s a new treatment with a new mechanism of action for a given condition, there’s a potential gain for the field.”
Dr. Schatzberg, who was not involved with the study, said that ezogabine, with its “potentially new mechanism of action, seems to have an effect and reasonable safety and could be important for patients who may not respond to traditional medications. It might also be important for all sorts of patients, depending on findings of later trials.”
Dr. Murrough said that ezogabine in still in “early stages” of research. “We hope that future studies will look at other agents that would also affect this channel,” he added.
This research was supported by the National Institute of Mental Health. Additional funding was provided by the Friedman Brain Institute at the Icahn School of Medicine at Mount Sinai and the Ehrenkranz Laboratory for Human Resilience, a component of the Depression and Anxiety Center for Discovery and Treatment at the Icahn School of Medicine at Mount Sinai. Dr. Murrough is an inventor of a pending patent application for the use of ezogabine and other KCNQ channel openers to treat depression and related disorders. Dr. Schatzberg disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
An antiseizure medication appears to reduce anhedonia in patients with depression via a novel mechanism that may offer a new therapeutic target for the disorder, new research suggests.
Results of a small, randomized trial show those who received ezogabine (Potiga) experienced a significant reduction in key measures of depression and anhedonia versus placebo.
Participants in the treatment group also showed a trend toward increased response to reward anticipation on functional MRI (fMRI), compared with those treated with placebo, although the effect did not reach statistical significance.
“Our study was the first randomized, placebo-controlled trial to show that a drug affecting this kind of ion channel in the brain can improve depression and anhedonia in patients,” senior investigator James Murrough, MD, PhD, associate professor of psychiatry and neuroscience at the Icahn School of Medicine at Mount Sinai, New York, said in a press release.
“Targeting this channel represents a completely different mechanism of action than any currently available antidepressant treatment,” said Dr. Murrough, who is also director of the Depression and Anxiety Center for Discovery and Treatment at Mount Sinai.
The study was published online March 3 in the American Journal of Psychiatry.
Need for a novel target
“One of the main issues in treating depression is that many of our current antidepressants have similar mechanisms of action,” Dr. Murrough said in an interview. “Once a patient hasn’t responded to currently available agents, it’s hard think of new medications to fill that need.”
This need for a novel target motivated Dr. Murrough and associates to research the KCNQ2/3 potassium channel, which has not been previously studied as a therapeutic target for depression.
The KCNQ2/3 channel controls brain cell excitability and function by controlling the flow of the electrical charge across the cell membrane in the form of potassium ions, Dr. Murrough explained.
Previous research using a chronic social defeat model of depression in mice showed changes in the KCNQ2/3 channel. “This was key to determining whether a mouse showed depressive behavior in the context of stress, or whether the mice were resistant or resilient to stress,” he said.
Mice resistant to stress showed increased markers in brain regions associated with reward, while the less resilient mice showed excessive excitability and dysfunction. Dysfunction in the brain’s reward system leads to anhedonia, a “core feature” of depressive disorders.
This inspired Dr. Murrough’s group to identify ezogabine, a drug that acts on this channel.
“ for addressing depressive symptoms,” Dr. Murrough said
Nonsignificant trend
The researchers studied 45 adults diagnosed with depression who exhibited significant anhedonia and at least moderate illness severity.
Participants were randomly assigned to receive either ezogabine (n = 21, mean age 44 years at enrollment, 28.3% male) or placebo (n = 24, mean age 39 years at enrollment, 50% male). At baseline and following treatment, participants completed the incentive flanker test under fMRI conditions to model brain activity during anticipation of a reward. In addition, clinical measures of depression and anhedonia were assessed at weekly visits.
The study groups did not differ significantly in performance accuracy during the fMRI task at baseline or following treatment. The table below summarizes the percentage of errors in each group, with standard deviation.
Participants in the ezogabine group showed a numerical increase in ventral striatum activation in response to reward anticipation, compared with participants in the placebo group, but this trend was not considered significant.
Heterogeneous condition
In contrast, there were notable improvements from baseline to the final outcome visit in clinical measures of depression and anhedonia in the ezogabine group, compared with the placebo group. Mean (SD) differences in depression scores, based on the MADRS (Montgomery-Åsberg Depression Rating Scale) from baseline to endpoint as follows: mean difference, –7.9 (3.0); effect size, 0.76; response rate, 61.9% (ezogabine) and 37.5% (placebo); remission rate: 38.1% (ezogabine) and 20.8% (placebo)
Compared with placebo, there were also large improvements in hedonic capacity, as measured by the Snaith-Hamilton Pleasure Scale and the anticipatory subscale of the Temporal Experience of Pleasure Scale (t, –4.1; df, 212; P < .001 and t, 3.4; df, 213; P < .001, respectively).
Compared with placebo, ezogabine was associated with “significant benefit” in global illness severity and improvement (Clinical Global Impression–Severity: t, –2.2; df, 214; P = .026 and CGI-Improvement: t, –2.9; d, 214; P = .004, respectively).
Ezogabine was well tolerated. Dizziness and headache were the most common adverse events.
Depression is a “heterogeneous condition” with a single diagnosis encompassing a “large, multifaceted” array of symptoms, Dr. Murrough noted. A growing body of research is focusing on specific components as potential treatment targets. “Our study looked specifically at patients with a diagnosis of depression but high scores on the anhedonia scale and we found that boosting the function of the KCNQ2/3 channel may have a beneficial antidepressant effect by improving anhedonia.”
Potential gain
In a comment, Alan Schatzberg, MD, professor in the department of psychiatry and behavioral sciences at Stanford (Calif.) University, said that “anytime there’s a new treatment with a new mechanism of action for a given condition, there’s a potential gain for the field.”
Dr. Schatzberg, who was not involved with the study, said that ezogabine, with its “potentially new mechanism of action, seems to have an effect and reasonable safety and could be important for patients who may not respond to traditional medications. It might also be important for all sorts of patients, depending on findings of later trials.”
Dr. Murrough said that ezogabine in still in “early stages” of research. “We hope that future studies will look at other agents that would also affect this channel,” he added.
This research was supported by the National Institute of Mental Health. Additional funding was provided by the Friedman Brain Institute at the Icahn School of Medicine at Mount Sinai and the Ehrenkranz Laboratory for Human Resilience, a component of the Depression and Anxiety Center for Discovery and Treatment at the Icahn School of Medicine at Mount Sinai. Dr. Murrough is an inventor of a pending patent application for the use of ezogabine and other KCNQ channel openers to treat depression and related disorders. Dr. Schatzberg disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.