User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
Powered by CHEST Physician, Clinician Reviews, MDedge Family Medicine, Internal Medicine News, and The Journal of Clinical Outcomes Management.
Acid-suppressant medications in infants with bronchiolitis raises later allergy risk
Infants who are hospitalized for severe bronchiolitis and receive acid-suppressant medications may be at risk of developing allergic disease by age 3 years, according to recent research released as an abstract for the American Academy of Allergy, Asthma & Immunology (AAAAI) Annual Meeting.
The AAAAI canceled their annual meeting and provided abstracts and access to presenters for press coverage
“Among children with a history of severe bronchiolitis during infancy, exposure to acid-suppressant medications during infancy further increases the risk of developing recurrent wheeze by age 3 years,” Lacey B. Robinson, MD, of the division of rheumatology, allergy, and immunology in the department of medicine at Massachusetts General Hospital in Boston, said in an interview.
Bronchiolitis is a risk factor in infants for developing conditions such as recurrent wheeze and childhood asthma in early childhood. Acid-suppressant medications like proton pump inhibitors (PPIs) and histamine-2 receptor antagonists (H2RAs) may further increase the risk of allergic disease. One study by Mitre et al. published in JAMA Pediatrics showed use of acid-suppressant medications in infants up to 6 months raised the risk of allergic disease (JAMA Pediatr. 2018;172[6]:e180315). Some studies suggest between 30% and 50% of infants diagnosed with bronchiolitis requiring hospitalization will develop asthma by age 5 years (J Allergy Clin Immunol Pract. 2017 Jan - Feb;5[1]:92-6).
“Children with severe bronchiolitis during infancy are at a high risk of developing recurrent wheeze and subsequent asthma. There is limited evidence to suggest that exposure to acid suppressant medications [such as proton pump inhibitors and histamine-2 receptor antagonists] prenatally and during early childhood increases the risk of childhood asthma,” Dr. Robinson said. “It is not known if exposure to acid suppressant medications during infancy further increases the risk of developing recurrent wheeze among high-risk children, such as in those with a history of severe bronchiolitis during infancy.”
Dr. Robinson and colleagues performed a multicenter, prospective cohort study of 921 infants who were hospitalized for severe bronchiolitis between 2011 and 2014. The investigators reviewed the medical records of the infants for acid suppressant medication use, as well as parent report of acid suppressant medication use, during an infant’s first 12 months. Overall, 879 children were analyzed after excluding for patients who developed recurrent wheeze prior to receiving acid suppressant medications, as well as patients with incomplete data. The investigators used the National Institutes of Health Guidelines for the Diagnosis and Management of Asthma (EPR-3) to define recurrent wheeze. A Cox-proportional hazard model was used to analyze the time to event, which was stratified by age and adjusted for confounders.
Infants with a history of severe bronchiolitis were at greater risk of developing recurrent wheeze by age 3 years after being exposed to acid-suppressant medications, compared with infants who were not exposed, Dr. Robinson said. Of the 879 infants in the final analysis, 159 (18%) received acid-suppressant medications, and 68 of 159 patients (43%) went on to develop recurrent wheeze, compared with 206 of 720 infants (29%) who were not exposed (unadjusted hazard ratio, 1.63; 95% confidence interval, 1.24-2.14).
After adjustment for confounders such as gender, race and ethnicity; gestational age; delivery type; severity of bronchiolitis; respiratory syncytial virus (RSV) infection status; maternal atopy; use of acid-suppressant medications during pregnancy; median household income; and insurance status, the association between recurrent wheeze and acid-suppressant medication use during infancy remained (adjusted HR, 1.54; 95% CI, 1.15-2.07).
“More research is needed on this important topic including studies in other populations,” such as in healthy children, Dr. Robinson said. “We encourage future research on this important and understudied topic, including further research on the potential underlying mechanisms of this association.”
Dr. Robinson reported no relevant financial disclosures.
SOURCE: Robinson L. AAAAI 2020, Abstract L1
Infants who are hospitalized for severe bronchiolitis and receive acid-suppressant medications may be at risk of developing allergic disease by age 3 years, according to recent research released as an abstract for the American Academy of Allergy, Asthma & Immunology (AAAAI) Annual Meeting.
The AAAAI canceled their annual meeting and provided abstracts and access to presenters for press coverage
“Among children with a history of severe bronchiolitis during infancy, exposure to acid-suppressant medications during infancy further increases the risk of developing recurrent wheeze by age 3 years,” Lacey B. Robinson, MD, of the division of rheumatology, allergy, and immunology in the department of medicine at Massachusetts General Hospital in Boston, said in an interview.
Bronchiolitis is a risk factor in infants for developing conditions such as recurrent wheeze and childhood asthma in early childhood. Acid-suppressant medications like proton pump inhibitors (PPIs) and histamine-2 receptor antagonists (H2RAs) may further increase the risk of allergic disease. One study by Mitre et al. published in JAMA Pediatrics showed use of acid-suppressant medications in infants up to 6 months raised the risk of allergic disease (JAMA Pediatr. 2018;172[6]:e180315). Some studies suggest between 30% and 50% of infants diagnosed with bronchiolitis requiring hospitalization will develop asthma by age 5 years (J Allergy Clin Immunol Pract. 2017 Jan - Feb;5[1]:92-6).
“Children with severe bronchiolitis during infancy are at a high risk of developing recurrent wheeze and subsequent asthma. There is limited evidence to suggest that exposure to acid suppressant medications [such as proton pump inhibitors and histamine-2 receptor antagonists] prenatally and during early childhood increases the risk of childhood asthma,” Dr. Robinson said. “It is not known if exposure to acid suppressant medications during infancy further increases the risk of developing recurrent wheeze among high-risk children, such as in those with a history of severe bronchiolitis during infancy.”
Dr. Robinson and colleagues performed a multicenter, prospective cohort study of 921 infants who were hospitalized for severe bronchiolitis between 2011 and 2014. The investigators reviewed the medical records of the infants for acid suppressant medication use, as well as parent report of acid suppressant medication use, during an infant’s first 12 months. Overall, 879 children were analyzed after excluding for patients who developed recurrent wheeze prior to receiving acid suppressant medications, as well as patients with incomplete data. The investigators used the National Institutes of Health Guidelines for the Diagnosis and Management of Asthma (EPR-3) to define recurrent wheeze. A Cox-proportional hazard model was used to analyze the time to event, which was stratified by age and adjusted for confounders.
Infants with a history of severe bronchiolitis were at greater risk of developing recurrent wheeze by age 3 years after being exposed to acid-suppressant medications, compared with infants who were not exposed, Dr. Robinson said. Of the 879 infants in the final analysis, 159 (18%) received acid-suppressant medications, and 68 of 159 patients (43%) went on to develop recurrent wheeze, compared with 206 of 720 infants (29%) who were not exposed (unadjusted hazard ratio, 1.63; 95% confidence interval, 1.24-2.14).
After adjustment for confounders such as gender, race and ethnicity; gestational age; delivery type; severity of bronchiolitis; respiratory syncytial virus (RSV) infection status; maternal atopy; use of acid-suppressant medications during pregnancy; median household income; and insurance status, the association between recurrent wheeze and acid-suppressant medication use during infancy remained (adjusted HR, 1.54; 95% CI, 1.15-2.07).
“More research is needed on this important topic including studies in other populations,” such as in healthy children, Dr. Robinson said. “We encourage future research on this important and understudied topic, including further research on the potential underlying mechanisms of this association.”
Dr. Robinson reported no relevant financial disclosures.
SOURCE: Robinson L. AAAAI 2020, Abstract L1
Infants who are hospitalized for severe bronchiolitis and receive acid-suppressant medications may be at risk of developing allergic disease by age 3 years, according to recent research released as an abstract for the American Academy of Allergy, Asthma & Immunology (AAAAI) Annual Meeting.
The AAAAI canceled their annual meeting and provided abstracts and access to presenters for press coverage
“Among children with a history of severe bronchiolitis during infancy, exposure to acid-suppressant medications during infancy further increases the risk of developing recurrent wheeze by age 3 years,” Lacey B. Robinson, MD, of the division of rheumatology, allergy, and immunology in the department of medicine at Massachusetts General Hospital in Boston, said in an interview.
Bronchiolitis is a risk factor in infants for developing conditions such as recurrent wheeze and childhood asthma in early childhood. Acid-suppressant medications like proton pump inhibitors (PPIs) and histamine-2 receptor antagonists (H2RAs) may further increase the risk of allergic disease. One study by Mitre et al. published in JAMA Pediatrics showed use of acid-suppressant medications in infants up to 6 months raised the risk of allergic disease (JAMA Pediatr. 2018;172[6]:e180315). Some studies suggest between 30% and 50% of infants diagnosed with bronchiolitis requiring hospitalization will develop asthma by age 5 years (J Allergy Clin Immunol Pract. 2017 Jan - Feb;5[1]:92-6).
“Children with severe bronchiolitis during infancy are at a high risk of developing recurrent wheeze and subsequent asthma. There is limited evidence to suggest that exposure to acid suppressant medications [such as proton pump inhibitors and histamine-2 receptor antagonists] prenatally and during early childhood increases the risk of childhood asthma,” Dr. Robinson said. “It is not known if exposure to acid suppressant medications during infancy further increases the risk of developing recurrent wheeze among high-risk children, such as in those with a history of severe bronchiolitis during infancy.”
Dr. Robinson and colleagues performed a multicenter, prospective cohort study of 921 infants who were hospitalized for severe bronchiolitis between 2011 and 2014. The investigators reviewed the medical records of the infants for acid suppressant medication use, as well as parent report of acid suppressant medication use, during an infant’s first 12 months. Overall, 879 children were analyzed after excluding for patients who developed recurrent wheeze prior to receiving acid suppressant medications, as well as patients with incomplete data. The investigators used the National Institutes of Health Guidelines for the Diagnosis and Management of Asthma (EPR-3) to define recurrent wheeze. A Cox-proportional hazard model was used to analyze the time to event, which was stratified by age and adjusted for confounders.
Infants with a history of severe bronchiolitis were at greater risk of developing recurrent wheeze by age 3 years after being exposed to acid-suppressant medications, compared with infants who were not exposed, Dr. Robinson said. Of the 879 infants in the final analysis, 159 (18%) received acid-suppressant medications, and 68 of 159 patients (43%) went on to develop recurrent wheeze, compared with 206 of 720 infants (29%) who were not exposed (unadjusted hazard ratio, 1.63; 95% confidence interval, 1.24-2.14).
After adjustment for confounders such as gender, race and ethnicity; gestational age; delivery type; severity of bronchiolitis; respiratory syncytial virus (RSV) infection status; maternal atopy; use of acid-suppressant medications during pregnancy; median household income; and insurance status, the association between recurrent wheeze and acid-suppressant medication use during infancy remained (adjusted HR, 1.54; 95% CI, 1.15-2.07).
“More research is needed on this important topic including studies in other populations,” such as in healthy children, Dr. Robinson said. “We encourage future research on this important and understudied topic, including further research on the potential underlying mechanisms of this association.”
Dr. Robinson reported no relevant financial disclosures.
SOURCE: Robinson L. AAAAI 2020, Abstract L1
REPORTING FROM AAAAI 2020
Physician couples draft wills, face tough questions amid COVID-19
Not long ago, weekends for Cornelia Griggs, MD, meant making trips to the grocery store, chasing after two active toddlers, and eating brunch with her husband after a busy work week. But life has changed dramatically for the family since the spread of COVID-19. On a recent weekend, Dr. Griggs and her husband, Robert Goldstone, MD, spent their days off drafting a will.
“We’re both doctors, and we know that health care workers have an increased risk of contracting COVID,” said Dr. Griggs, a pediatric surgery fellow at Columbia University Irving Medical Center in New York. “It felt like the responsible thing to do: Have a will in place to make sure our wishes are clear about who would manage our property and assets, and who would take care of our kids – God forbid.”
Outlining their final wishes is among many difficult decisions the doctors, both 36, have been forced to make in recent weeks. Dr. Goldstone, a general surgeon at Massachusetts General Hospital in Boston, is no longer returning to New York during his time off, said Dr. Griggs, who has had known COVID-19 exposures. The couple’s children, aged 4 and almost 2, are temporarily living with their grandparents in Connecticut to decrease their exposure risk.
“I felt like it was safer for all of them to be there while I was going back and forth from the hospital,” Dr. Griggs said. “My husband is in Boston. The kids are in Connecticut and I’m in New York. That inherently is hard because our whole family is split up. I don’t know when it will be safe for me to see them again.”
Health professional couples across the country are facing similar challenges as they navigate the risk of contracting COVID-19 at work, while trying to protect their families at home. From childcare dilemmas to quarantine quandaries to end-of-life considerations, partners who work in health care are confronting tough questions as the pandemic continues.
The biggest challenge is the uncertainty, says Angela Weyand, MD, an Ann Arbor, Mich.–based pediatric hematologist/oncologist who shares two young daughters with husband Ted Claflin, MD, a physical medicine and rehabilitation physician. Dr. Weyand said she and her husband are primarily working remotely now, but she knows that one or both could be deployed to the hospital to help care for patients, if the need arises. Nearby Detroit has been labeled a coronavirus “hot spot” by the U.S. Surgeon General.
“Right now, I think our biggest fear is spreading coronavirus to those we love, especially those in higher risk groups,” she said. “At the same time, we are also concerned about our own health and our future ability to be there for our children, a fear that, thankfully, neither one of us has ever had to face before. We are trying to take things one day at a time, acknowledging all that we have to be grateful for, and also learning to accept that many things right now are outside of our control.”
Dr. Weyand, 38, and her husband, 40, finalized their wills in March.
“We have been working on them for quite some time, but before now, there has never been any urgency,” Dr. Weyand said. “Hearing about the high rate of infection in health care workers and the increasing number of deaths in young healthy people made us realize that this should be a priority.”
Dallas internist Bethany Agusala, MD, 36, and her husband, Kartik Agusala, MD, 41, a cardiologist, recently spent time engaged in the same activity. The couple, who work for the University of Texas Southwestern Medical Center, have two children, aged 2 and 4.
“The chances are hopefully small that something bad would happen to either one of us, but it just seemed like a good time to get [a will] in place,” Dr. Bethany Agusala said in an interview. “It’s never an easy thing to think about.
Pediatric surgeon Chethan Sathya, MD, 34, and his wife, 31, a physician assistant, have vastly altered their home routine to prevent the risk of exposure to their 16-month-old daughter. Dr. Sathya works for the Northwell Health System in New York, which has hundreds of hospitalized patients with COVID-19, Dr. Sathya said in an interview. He did not want to disclose his wife's name or institution, but said she works in a COVID-19 unit at a New York hospital.
When his wife returns home, she removes all of her clothes and places them in a bag, showers, and then isolates herself in the bedroom. Dr. Sathya brings his wife meals and then remains in a different room with their baby.
“It’s only been a few days,” he said. “We’re going to decide: Does she just stay in one room at all times or when she doesn’t work for a few days then after 1 day, can she come out? Should she get a hotel room elsewhere? These are the considerations.”
They employ an older nanny whom they also worry about, and with whom they try to limit contact, said Dr. Sathya, who practices at Cohen Children’s Medical Center. In a matter of weeks, Dr. Sathya anticipates he will be called upon to assist in some form with the COVID crisis.
“We haven’t figured that out. I’m not sure what we’ll do,” he said. “There is no perfect solution. You have to adapt. It’s very difficult to do so when you’re living in a condo in New York.”
For Dr. Griggs, life is much quieter at home without her husband and two “laughing, wiggly,” toddlers. Weekends are now defined by resting, video calls with her family, and exercising, when it’s safe, said Dr. Griggs, who recently penned a New York Times opinion piece about the pandemic and is also active on social media regarding personal protective equipment. She calls her husband her “rock” who never fails to put a smile on her face when they chat from across the miles. Her advice for other health care couples is to take it “one day at a time.”
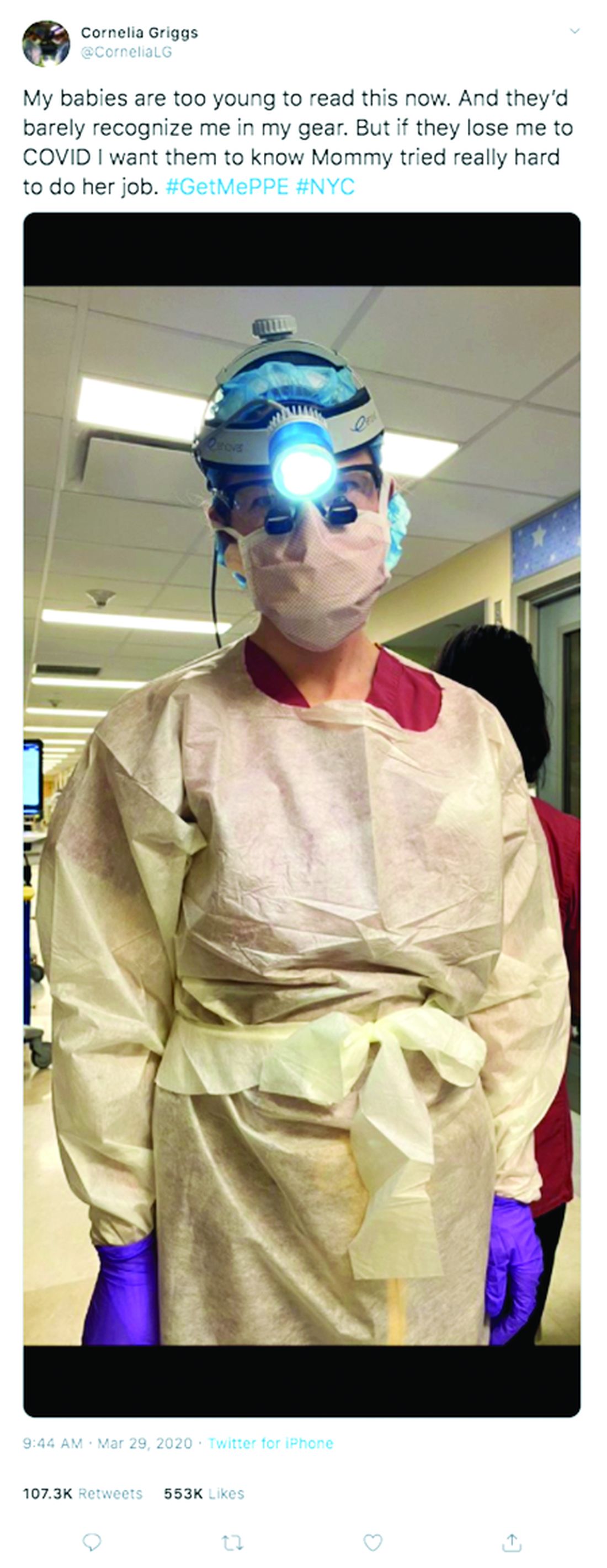
“Don’t try to make plans weeks in advance or let your mind go to a dark place,” she said. “It’s so easy to feel overwhelmed. The only way to get through this is to focus on surviving each day.”
Editor's Note, 3/31/20: Due to incorrect information provided, the hospital where Dr. Sathya's wife works was misidentified. We have removed the name of that hospital. The story does not include his wife's employer, because Dr. Sathya did not have permission to disclose her workplace and she wishes to remain anonymous.
Not long ago, weekends for Cornelia Griggs, MD, meant making trips to the grocery store, chasing after two active toddlers, and eating brunch with her husband after a busy work week. But life has changed dramatically for the family since the spread of COVID-19. On a recent weekend, Dr. Griggs and her husband, Robert Goldstone, MD, spent their days off drafting a will.
“We’re both doctors, and we know that health care workers have an increased risk of contracting COVID,” said Dr. Griggs, a pediatric surgery fellow at Columbia University Irving Medical Center in New York. “It felt like the responsible thing to do: Have a will in place to make sure our wishes are clear about who would manage our property and assets, and who would take care of our kids – God forbid.”
Outlining their final wishes is among many difficult decisions the doctors, both 36, have been forced to make in recent weeks. Dr. Goldstone, a general surgeon at Massachusetts General Hospital in Boston, is no longer returning to New York during his time off, said Dr. Griggs, who has had known COVID-19 exposures. The couple’s children, aged 4 and almost 2, are temporarily living with their grandparents in Connecticut to decrease their exposure risk.
“I felt like it was safer for all of them to be there while I was going back and forth from the hospital,” Dr. Griggs said. “My husband is in Boston. The kids are in Connecticut and I’m in New York. That inherently is hard because our whole family is split up. I don’t know when it will be safe for me to see them again.”
Health professional couples across the country are facing similar challenges as they navigate the risk of contracting COVID-19 at work, while trying to protect their families at home. From childcare dilemmas to quarantine quandaries to end-of-life considerations, partners who work in health care are confronting tough questions as the pandemic continues.
The biggest challenge is the uncertainty, says Angela Weyand, MD, an Ann Arbor, Mich.–based pediatric hematologist/oncologist who shares two young daughters with husband Ted Claflin, MD, a physical medicine and rehabilitation physician. Dr. Weyand said she and her husband are primarily working remotely now, but she knows that one or both could be deployed to the hospital to help care for patients, if the need arises. Nearby Detroit has been labeled a coronavirus “hot spot” by the U.S. Surgeon General.
“Right now, I think our biggest fear is spreading coronavirus to those we love, especially those in higher risk groups,” she said. “At the same time, we are also concerned about our own health and our future ability to be there for our children, a fear that, thankfully, neither one of us has ever had to face before. We are trying to take things one day at a time, acknowledging all that we have to be grateful for, and also learning to accept that many things right now are outside of our control.”
Dr. Weyand, 38, and her husband, 40, finalized their wills in March.
“We have been working on them for quite some time, but before now, there has never been any urgency,” Dr. Weyand said. “Hearing about the high rate of infection in health care workers and the increasing number of deaths in young healthy people made us realize that this should be a priority.”
Dallas internist Bethany Agusala, MD, 36, and her husband, Kartik Agusala, MD, 41, a cardiologist, recently spent time engaged in the same activity. The couple, who work for the University of Texas Southwestern Medical Center, have two children, aged 2 and 4.
“The chances are hopefully small that something bad would happen to either one of us, but it just seemed like a good time to get [a will] in place,” Dr. Bethany Agusala said in an interview. “It’s never an easy thing to think about.
Pediatric surgeon Chethan Sathya, MD, 34, and his wife, 31, a physician assistant, have vastly altered their home routine to prevent the risk of exposure to their 16-month-old daughter. Dr. Sathya works for the Northwell Health System in New York, which has hundreds of hospitalized patients with COVID-19, Dr. Sathya said in an interview. He did not want to disclose his wife's name or institution, but said she works in a COVID-19 unit at a New York hospital.
When his wife returns home, she removes all of her clothes and places them in a bag, showers, and then isolates herself in the bedroom. Dr. Sathya brings his wife meals and then remains in a different room with their baby.
“It’s only been a few days,” he said. “We’re going to decide: Does she just stay in one room at all times or when she doesn’t work for a few days then after 1 day, can she come out? Should she get a hotel room elsewhere? These are the considerations.”
They employ an older nanny whom they also worry about, and with whom they try to limit contact, said Dr. Sathya, who practices at Cohen Children’s Medical Center. In a matter of weeks, Dr. Sathya anticipates he will be called upon to assist in some form with the COVID crisis.
“We haven’t figured that out. I’m not sure what we’ll do,” he said. “There is no perfect solution. You have to adapt. It’s very difficult to do so when you’re living in a condo in New York.”
For Dr. Griggs, life is much quieter at home without her husband and two “laughing, wiggly,” toddlers. Weekends are now defined by resting, video calls with her family, and exercising, when it’s safe, said Dr. Griggs, who recently penned a New York Times opinion piece about the pandemic and is also active on social media regarding personal protective equipment. She calls her husband her “rock” who never fails to put a smile on her face when they chat from across the miles. Her advice for other health care couples is to take it “one day at a time.”

“Don’t try to make plans weeks in advance or let your mind go to a dark place,” she said. “It’s so easy to feel overwhelmed. The only way to get through this is to focus on surviving each day.”
Editor's Note, 3/31/20: Due to incorrect information provided, the hospital where Dr. Sathya's wife works was misidentified. We have removed the name of that hospital. The story does not include his wife's employer, because Dr. Sathya did not have permission to disclose her workplace and she wishes to remain anonymous.
Not long ago, weekends for Cornelia Griggs, MD, meant making trips to the grocery store, chasing after two active toddlers, and eating brunch with her husband after a busy work week. But life has changed dramatically for the family since the spread of COVID-19. On a recent weekend, Dr. Griggs and her husband, Robert Goldstone, MD, spent their days off drafting a will.
“We’re both doctors, and we know that health care workers have an increased risk of contracting COVID,” said Dr. Griggs, a pediatric surgery fellow at Columbia University Irving Medical Center in New York. “It felt like the responsible thing to do: Have a will in place to make sure our wishes are clear about who would manage our property and assets, and who would take care of our kids – God forbid.”
Outlining their final wishes is among many difficult decisions the doctors, both 36, have been forced to make in recent weeks. Dr. Goldstone, a general surgeon at Massachusetts General Hospital in Boston, is no longer returning to New York during his time off, said Dr. Griggs, who has had known COVID-19 exposures. The couple’s children, aged 4 and almost 2, are temporarily living with their grandparents in Connecticut to decrease their exposure risk.
“I felt like it was safer for all of them to be there while I was going back and forth from the hospital,” Dr. Griggs said. “My husband is in Boston. The kids are in Connecticut and I’m in New York. That inherently is hard because our whole family is split up. I don’t know when it will be safe for me to see them again.”
Health professional couples across the country are facing similar challenges as they navigate the risk of contracting COVID-19 at work, while trying to protect their families at home. From childcare dilemmas to quarantine quandaries to end-of-life considerations, partners who work in health care are confronting tough questions as the pandemic continues.
The biggest challenge is the uncertainty, says Angela Weyand, MD, an Ann Arbor, Mich.–based pediatric hematologist/oncologist who shares two young daughters with husband Ted Claflin, MD, a physical medicine and rehabilitation physician. Dr. Weyand said she and her husband are primarily working remotely now, but she knows that one or both could be deployed to the hospital to help care for patients, if the need arises. Nearby Detroit has been labeled a coronavirus “hot spot” by the U.S. Surgeon General.
“Right now, I think our biggest fear is spreading coronavirus to those we love, especially those in higher risk groups,” she said. “At the same time, we are also concerned about our own health and our future ability to be there for our children, a fear that, thankfully, neither one of us has ever had to face before. We are trying to take things one day at a time, acknowledging all that we have to be grateful for, and also learning to accept that many things right now are outside of our control.”
Dr. Weyand, 38, and her husband, 40, finalized their wills in March.
“We have been working on them for quite some time, but before now, there has never been any urgency,” Dr. Weyand said. “Hearing about the high rate of infection in health care workers and the increasing number of deaths in young healthy people made us realize that this should be a priority.”
Dallas internist Bethany Agusala, MD, 36, and her husband, Kartik Agusala, MD, 41, a cardiologist, recently spent time engaged in the same activity. The couple, who work for the University of Texas Southwestern Medical Center, have two children, aged 2 and 4.
“The chances are hopefully small that something bad would happen to either one of us, but it just seemed like a good time to get [a will] in place,” Dr. Bethany Agusala said in an interview. “It’s never an easy thing to think about.
Pediatric surgeon Chethan Sathya, MD, 34, and his wife, 31, a physician assistant, have vastly altered their home routine to prevent the risk of exposure to their 16-month-old daughter. Dr. Sathya works for the Northwell Health System in New York, which has hundreds of hospitalized patients with COVID-19, Dr. Sathya said in an interview. He did not want to disclose his wife's name or institution, but said she works in a COVID-19 unit at a New York hospital.
When his wife returns home, she removes all of her clothes and places them in a bag, showers, and then isolates herself in the bedroom. Dr. Sathya brings his wife meals and then remains in a different room with their baby.
“It’s only been a few days,” he said. “We’re going to decide: Does she just stay in one room at all times or when she doesn’t work for a few days then after 1 day, can she come out? Should she get a hotel room elsewhere? These are the considerations.”
They employ an older nanny whom they also worry about, and with whom they try to limit contact, said Dr. Sathya, who practices at Cohen Children’s Medical Center. In a matter of weeks, Dr. Sathya anticipates he will be called upon to assist in some form with the COVID crisis.
“We haven’t figured that out. I’m not sure what we’ll do,” he said. “There is no perfect solution. You have to adapt. It’s very difficult to do so when you’re living in a condo in New York.”
For Dr. Griggs, life is much quieter at home without her husband and two “laughing, wiggly,” toddlers. Weekends are now defined by resting, video calls with her family, and exercising, when it’s safe, said Dr. Griggs, who recently penned a New York Times opinion piece about the pandemic and is also active on social media regarding personal protective equipment. She calls her husband her “rock” who never fails to put a smile on her face when they chat from across the miles. Her advice for other health care couples is to take it “one day at a time.”

“Don’t try to make plans weeks in advance or let your mind go to a dark place,” she said. “It’s so easy to feel overwhelmed. The only way to get through this is to focus on surviving each day.”
Editor's Note, 3/31/20: Due to incorrect information provided, the hospital where Dr. Sathya's wife works was misidentified. We have removed the name of that hospital. The story does not include his wife's employer, because Dr. Sathya did not have permission to disclose her workplace and she wishes to remain anonymous.
Flu activity measures continue COVID-19–related divergence
The 2019-2020 flu paradox continues in the United States: Fewer respiratory samples are testing positive for influenza, but more people are seeking care for respiratory symptoms because of COVID-19, according to the Centers for Disease Control and Prevention.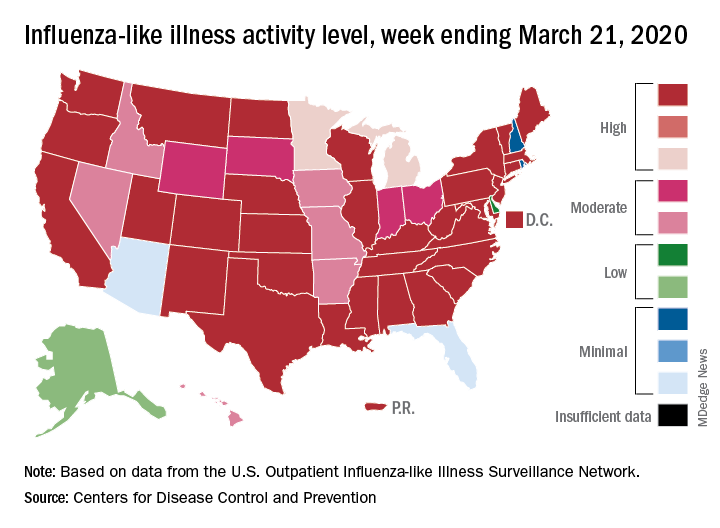
compared with 14.9% the week before, but outpatient visits for influenza-like illness (ILI) rose from 5.6% of all visits to 6.2% for third week of March, the CDC’s influenza division reported.
The CDC defines ILI as “fever (temperature of 100°F [37.8°C] or greater) and a cough and/or a sore throat without a known cause other than influenza.” The outpatient ILI visit rate needs to get below the national baseline of 2.4% for the CDC to call the end of the 2019-2020 flu season.
This week’s map shows that fewer states are at the highest level of ILI activity on the CDC’s 1-10 scale: 33 states plus Puerto Rico for the week ending March 21, compared with 35 and Puerto Rico the previous week. The number of states at level 10 had risen the two previous weeks, CDC data show.
“Influenza severity indicators remain moderate to low overall, but hospitalization rates differ by age group, with high rates among children and young adults,” the influenza division said.
Overall mortality also has not been high, but 155 children have died from the flu so far in 2019-2020, which is more than any season since the 2009 pandemic, the CDC noted.
The 2019-2020 flu paradox continues in the United States: Fewer respiratory samples are testing positive for influenza, but more people are seeking care for respiratory symptoms because of COVID-19, according to the Centers for Disease Control and Prevention.
compared with 14.9% the week before, but outpatient visits for influenza-like illness (ILI) rose from 5.6% of all visits to 6.2% for third week of March, the CDC’s influenza division reported.
The CDC defines ILI as “fever (temperature of 100°F [37.8°C] or greater) and a cough and/or a sore throat without a known cause other than influenza.” The outpatient ILI visit rate needs to get below the national baseline of 2.4% for the CDC to call the end of the 2019-2020 flu season.
This week’s map shows that fewer states are at the highest level of ILI activity on the CDC’s 1-10 scale: 33 states plus Puerto Rico for the week ending March 21, compared with 35 and Puerto Rico the previous week. The number of states at level 10 had risen the two previous weeks, CDC data show.
“Influenza severity indicators remain moderate to low overall, but hospitalization rates differ by age group, with high rates among children and young adults,” the influenza division said.
Overall mortality also has not been high, but 155 children have died from the flu so far in 2019-2020, which is more than any season since the 2009 pandemic, the CDC noted.
The 2019-2020 flu paradox continues in the United States: Fewer respiratory samples are testing positive for influenza, but more people are seeking care for respiratory symptoms because of COVID-19, according to the Centers for Disease Control and Prevention.
compared with 14.9% the week before, but outpatient visits for influenza-like illness (ILI) rose from 5.6% of all visits to 6.2% for third week of March, the CDC’s influenza division reported.
The CDC defines ILI as “fever (temperature of 100°F [37.8°C] or greater) and a cough and/or a sore throat without a known cause other than influenza.” The outpatient ILI visit rate needs to get below the national baseline of 2.4% for the CDC to call the end of the 2019-2020 flu season.
This week’s map shows that fewer states are at the highest level of ILI activity on the CDC’s 1-10 scale: 33 states plus Puerto Rico for the week ending March 21, compared with 35 and Puerto Rico the previous week. The number of states at level 10 had risen the two previous weeks, CDC data show.
“Influenza severity indicators remain moderate to low overall, but hospitalization rates differ by age group, with high rates among children and young adults,” the influenza division said.
Overall mortality also has not been high, but 155 children have died from the flu so far in 2019-2020, which is more than any season since the 2009 pandemic, the CDC noted.
Before the COVID-19 surge hits your facility, take steps to boost capacity
, according to a physician leader and a health workforce expert.
Polly Pittman, PhD, is hearing a lot of concern among health care workers that it’s difficult to find definitive and accurate information about how best to protect themselves and their families, she said during a webinar by the Alliance for Health Policy titled Health System Capacity: Protecting Frontline Health Workers. “The knowledge base is evolving very quickly,” said Dr. Pittman, Fitzhugh Mullan Professor of Health Workforce Equity at the Milken Institute School of Public Health, George Washington University, Washington.
Stephen Parodi, MD, agreed that effective communication is job one in the health care workplace during the crisis. “I can’t stress enough ... that communications are paramount and you can’t overcommunicate,” said Dr. Parodi, executive vice president of external affairs, communications, and brand at the Permanente Federation and associate executive director of the Permanente Medical Group, Vallejo, Calif.
“We’re in a situation of confusion and improvisation right now,” regarding protection of health care workers, said Dr. Pittman. The potential exists for “a downward spiral where you have the lack of training, the shortages in terms of protective gear, weakening of guidelines, and confusion regarding guidelines at federal level, creating a potential cascade” that may result in “moral distress and fatigue. ... That’s not occurring now, but that’s the danger” unless the personal protective equipment (PPE) situation is adequately addressed very soon, she said.
Dr. Pittman also pointed out the concerns that many of the 18 million U.S. health care workers have for their families should they themselves fall ill or transmit coronavirus to family members. “The danger exists of a mass exodus. People don’t have to show up at work, and they won’t show up at work if they don’t feel supported and safe.”
Dr. Parodi said that the Permanente organization is on a better footing than many workplaces. “We actually had an early experience because of the work that we did to support the Diamond Princess cruise ship evacuees from Yokahama in February.” That ship was quarantined upon arrival in Yokahama on Feb. 3 because a passenger had a confirmed test for SARS-CoV-2 infection, and a quarter of the 428 Americans on board subsequently tested positive. Most of them were evacuated to California or Texas. “That actually gave us the experience for providing care within the hospital setting – and also for containment strategies,” he said.
“We quickly understood that we needed to move to a mitigation strategy,” said Dr. Parodi. Use of PPE has been “tailored for how the virus is spread.” In the absence of the risk of aerosol transmission from certain procedures, health care workers use gowns, gloves, surgical masks, and goggles.
Because of anticipated “supply chain shortfalls,” Dr. Parodi said that his organization implemented Centers for Disease Control and Prevention guidelines for reuse and extended use of N95 respirators early on. “Even if you’re not in a locale that’s been hit, you need to be on wartime footing for preserving PPE.”
Telehealth, said Dr. Parodi, has been implemented “in a huge way” throughout the Permanente system. “We have reduced primary care visits by 90% in the past week, and also subspecialty visits by 50%. … A large amount of the workforce can work from home. We turned off elective surgeries more than a week ago to reduce the number of patients who are requiring intensive care.” Making these changes means the organization is more prepared now for a surge they expect in the coming weeks.
Dr. Pittman voiced an opinion widely shared by those who are implementing large-scale telehealth efforts “We’re going to learn a lot. Many of the traditional doctor-patient visits can be done by telemedicine in the future.”
Knowledge about local trends in infection rates is key to preparedness. “We’ve ramped up testing, to understand what’s happening in the community,” said Dr. Parodi, noting that test turnaround time is currently running 8-24 hours. Tightening up this window can free up resources when an admitted patient’s test is negative.
Still, some national projections forecast a need for hospital beds at two to three times current capacity – or even more, said Dr. Parodi.
He noted that Permanente is “working hand in glove with state authorities throughout the country.” Efforts include establishing alternative sites for assessment and testing, as well as opening up closed hospitals and working with the National Guard and the Department of Defense to prepare mobile hospital units that can be deployed in areas with peak infection rates. “Having all of those options available to us is critically important,” he said.
To mitigate potential provider shortages, Dr. Pittman said, “All members of the care team could potentially do more” than their current licenses allow. Expanding the scope of practice for pharmacists, clinical laboratory staff, licensed practical nurses, and medical assistants can help with efficient care delivery.
Other measures include expedited licensing for near-graduates and nonpracticing foreign medical graduates, as well as relicensing for retired health care personnel and those who are not currently working directly with patients, she said.
Getting these things done “requires leadership on behalf of the licensing bodies,” as well as coordination with state regulatory authorities, Dr. Pittman pointed out.
Dr. Parodi called for state and federal governments to implement emergency declarations that suspend some existing health codes to achieve repurposing of staff. Getting these measures in place now will allow facilities “to be able to provide that in-time training now before the surge occurs. ... We are actively developing plans knowing that there’s going to be a need for more critical care.”
The game plan at Permanente, he said, is to repurpose critical care physicians to provide consultations to multiple hospitalists who are providing the bulk of frontline care. At the same time, they plan to repurpose other specialists to backfill the hospitalists, and to repurpose family medicine physicians to supplement staff in emergency departments and other frontline intake areas.
All the organizational measures being taken won’t be in vain if they increase preparedness for the long battle ahead, he said. “We need to double down on the work. ... We need to continue social distancing, and we’ve got to ramp up testing. Until we do that we have to hold the line on basic public health measures.”
Dr. Parodi is employed by Permanente. The panelists reported no disclosures relevant to the presentation, which was sponsored by the Alliance for Health Policy, the Commonwealth Fund, and the National Institute for Health Care Management Foundation.
, according to a physician leader and a health workforce expert.
Polly Pittman, PhD, is hearing a lot of concern among health care workers that it’s difficult to find definitive and accurate information about how best to protect themselves and their families, she said during a webinar by the Alliance for Health Policy titled Health System Capacity: Protecting Frontline Health Workers. “The knowledge base is evolving very quickly,” said Dr. Pittman, Fitzhugh Mullan Professor of Health Workforce Equity at the Milken Institute School of Public Health, George Washington University, Washington.
Stephen Parodi, MD, agreed that effective communication is job one in the health care workplace during the crisis. “I can’t stress enough ... that communications are paramount and you can’t overcommunicate,” said Dr. Parodi, executive vice president of external affairs, communications, and brand at the Permanente Federation and associate executive director of the Permanente Medical Group, Vallejo, Calif.
“We’re in a situation of confusion and improvisation right now,” regarding protection of health care workers, said Dr. Pittman. The potential exists for “a downward spiral where you have the lack of training, the shortages in terms of protective gear, weakening of guidelines, and confusion regarding guidelines at federal level, creating a potential cascade” that may result in “moral distress and fatigue. ... That’s not occurring now, but that’s the danger” unless the personal protective equipment (PPE) situation is adequately addressed very soon, she said.
Dr. Pittman also pointed out the concerns that many of the 18 million U.S. health care workers have for their families should they themselves fall ill or transmit coronavirus to family members. “The danger exists of a mass exodus. People don’t have to show up at work, and they won’t show up at work if they don’t feel supported and safe.”
Dr. Parodi said that the Permanente organization is on a better footing than many workplaces. “We actually had an early experience because of the work that we did to support the Diamond Princess cruise ship evacuees from Yokahama in February.” That ship was quarantined upon arrival in Yokahama on Feb. 3 because a passenger had a confirmed test for SARS-CoV-2 infection, and a quarter of the 428 Americans on board subsequently tested positive. Most of them were evacuated to California or Texas. “That actually gave us the experience for providing care within the hospital setting – and also for containment strategies,” he said.
“We quickly understood that we needed to move to a mitigation strategy,” said Dr. Parodi. Use of PPE has been “tailored for how the virus is spread.” In the absence of the risk of aerosol transmission from certain procedures, health care workers use gowns, gloves, surgical masks, and goggles.
Because of anticipated “supply chain shortfalls,” Dr. Parodi said that his organization implemented Centers for Disease Control and Prevention guidelines for reuse and extended use of N95 respirators early on. “Even if you’re not in a locale that’s been hit, you need to be on wartime footing for preserving PPE.”
Telehealth, said Dr. Parodi, has been implemented “in a huge way” throughout the Permanente system. “We have reduced primary care visits by 90% in the past week, and also subspecialty visits by 50%. … A large amount of the workforce can work from home. We turned off elective surgeries more than a week ago to reduce the number of patients who are requiring intensive care.” Making these changes means the organization is more prepared now for a surge they expect in the coming weeks.
Dr. Pittman voiced an opinion widely shared by those who are implementing large-scale telehealth efforts “We’re going to learn a lot. Many of the traditional doctor-patient visits can be done by telemedicine in the future.”
Knowledge about local trends in infection rates is key to preparedness. “We’ve ramped up testing, to understand what’s happening in the community,” said Dr. Parodi, noting that test turnaround time is currently running 8-24 hours. Tightening up this window can free up resources when an admitted patient’s test is negative.
Still, some national projections forecast a need for hospital beds at two to three times current capacity – or even more, said Dr. Parodi.
He noted that Permanente is “working hand in glove with state authorities throughout the country.” Efforts include establishing alternative sites for assessment and testing, as well as opening up closed hospitals and working with the National Guard and the Department of Defense to prepare mobile hospital units that can be deployed in areas with peak infection rates. “Having all of those options available to us is critically important,” he said.
To mitigate potential provider shortages, Dr. Pittman said, “All members of the care team could potentially do more” than their current licenses allow. Expanding the scope of practice for pharmacists, clinical laboratory staff, licensed practical nurses, and medical assistants can help with efficient care delivery.
Other measures include expedited licensing for near-graduates and nonpracticing foreign medical graduates, as well as relicensing for retired health care personnel and those who are not currently working directly with patients, she said.
Getting these things done “requires leadership on behalf of the licensing bodies,” as well as coordination with state regulatory authorities, Dr. Pittman pointed out.
Dr. Parodi called for state and federal governments to implement emergency declarations that suspend some existing health codes to achieve repurposing of staff. Getting these measures in place now will allow facilities “to be able to provide that in-time training now before the surge occurs. ... We are actively developing plans knowing that there’s going to be a need for more critical care.”
The game plan at Permanente, he said, is to repurpose critical care physicians to provide consultations to multiple hospitalists who are providing the bulk of frontline care. At the same time, they plan to repurpose other specialists to backfill the hospitalists, and to repurpose family medicine physicians to supplement staff in emergency departments and other frontline intake areas.
All the organizational measures being taken won’t be in vain if they increase preparedness for the long battle ahead, he said. “We need to double down on the work. ... We need to continue social distancing, and we’ve got to ramp up testing. Until we do that we have to hold the line on basic public health measures.”
Dr. Parodi is employed by Permanente. The panelists reported no disclosures relevant to the presentation, which was sponsored by the Alliance for Health Policy, the Commonwealth Fund, and the National Institute for Health Care Management Foundation.
, according to a physician leader and a health workforce expert.
Polly Pittman, PhD, is hearing a lot of concern among health care workers that it’s difficult to find definitive and accurate information about how best to protect themselves and their families, she said during a webinar by the Alliance for Health Policy titled Health System Capacity: Protecting Frontline Health Workers. “The knowledge base is evolving very quickly,” said Dr. Pittman, Fitzhugh Mullan Professor of Health Workforce Equity at the Milken Institute School of Public Health, George Washington University, Washington.
Stephen Parodi, MD, agreed that effective communication is job one in the health care workplace during the crisis. “I can’t stress enough ... that communications are paramount and you can’t overcommunicate,” said Dr. Parodi, executive vice president of external affairs, communications, and brand at the Permanente Federation and associate executive director of the Permanente Medical Group, Vallejo, Calif.
“We’re in a situation of confusion and improvisation right now,” regarding protection of health care workers, said Dr. Pittman. The potential exists for “a downward spiral where you have the lack of training, the shortages in terms of protective gear, weakening of guidelines, and confusion regarding guidelines at federal level, creating a potential cascade” that may result in “moral distress and fatigue. ... That’s not occurring now, but that’s the danger” unless the personal protective equipment (PPE) situation is adequately addressed very soon, she said.
Dr. Pittman also pointed out the concerns that many of the 18 million U.S. health care workers have for their families should they themselves fall ill or transmit coronavirus to family members. “The danger exists of a mass exodus. People don’t have to show up at work, and they won’t show up at work if they don’t feel supported and safe.”
Dr. Parodi said that the Permanente organization is on a better footing than many workplaces. “We actually had an early experience because of the work that we did to support the Diamond Princess cruise ship evacuees from Yokahama in February.” That ship was quarantined upon arrival in Yokahama on Feb. 3 because a passenger had a confirmed test for SARS-CoV-2 infection, and a quarter of the 428 Americans on board subsequently tested positive. Most of them were evacuated to California or Texas. “That actually gave us the experience for providing care within the hospital setting – and also for containment strategies,” he said.
“We quickly understood that we needed to move to a mitigation strategy,” said Dr. Parodi. Use of PPE has been “tailored for how the virus is spread.” In the absence of the risk of aerosol transmission from certain procedures, health care workers use gowns, gloves, surgical masks, and goggles.
Because of anticipated “supply chain shortfalls,” Dr. Parodi said that his organization implemented Centers for Disease Control and Prevention guidelines for reuse and extended use of N95 respirators early on. “Even if you’re not in a locale that’s been hit, you need to be on wartime footing for preserving PPE.”
Telehealth, said Dr. Parodi, has been implemented “in a huge way” throughout the Permanente system. “We have reduced primary care visits by 90% in the past week, and also subspecialty visits by 50%. … A large amount of the workforce can work from home. We turned off elective surgeries more than a week ago to reduce the number of patients who are requiring intensive care.” Making these changes means the organization is more prepared now for a surge they expect in the coming weeks.
Dr. Pittman voiced an opinion widely shared by those who are implementing large-scale telehealth efforts “We’re going to learn a lot. Many of the traditional doctor-patient visits can be done by telemedicine in the future.”
Knowledge about local trends in infection rates is key to preparedness. “We’ve ramped up testing, to understand what’s happening in the community,” said Dr. Parodi, noting that test turnaround time is currently running 8-24 hours. Tightening up this window can free up resources when an admitted patient’s test is negative.
Still, some national projections forecast a need for hospital beds at two to three times current capacity – or even more, said Dr. Parodi.
He noted that Permanente is “working hand in glove with state authorities throughout the country.” Efforts include establishing alternative sites for assessment and testing, as well as opening up closed hospitals and working with the National Guard and the Department of Defense to prepare mobile hospital units that can be deployed in areas with peak infection rates. “Having all of those options available to us is critically important,” he said.
To mitigate potential provider shortages, Dr. Pittman said, “All members of the care team could potentially do more” than their current licenses allow. Expanding the scope of practice for pharmacists, clinical laboratory staff, licensed practical nurses, and medical assistants can help with efficient care delivery.
Other measures include expedited licensing for near-graduates and nonpracticing foreign medical graduates, as well as relicensing for retired health care personnel and those who are not currently working directly with patients, she said.
Getting these things done “requires leadership on behalf of the licensing bodies,” as well as coordination with state regulatory authorities, Dr. Pittman pointed out.
Dr. Parodi called for state and federal governments to implement emergency declarations that suspend some existing health codes to achieve repurposing of staff. Getting these measures in place now will allow facilities “to be able to provide that in-time training now before the surge occurs. ... We are actively developing plans knowing that there’s going to be a need for more critical care.”
The game plan at Permanente, he said, is to repurpose critical care physicians to provide consultations to multiple hospitalists who are providing the bulk of frontline care. At the same time, they plan to repurpose other specialists to backfill the hospitalists, and to repurpose family medicine physicians to supplement staff in emergency departments and other frontline intake areas.
All the organizational measures being taken won’t be in vain if they increase preparedness for the long battle ahead, he said. “We need to double down on the work. ... We need to continue social distancing, and we’ve got to ramp up testing. Until we do that we have to hold the line on basic public health measures.”
Dr. Parodi is employed by Permanente. The panelists reported no disclosures relevant to the presentation, which was sponsored by the Alliance for Health Policy, the Commonwealth Fund, and the National Institute for Health Care Management Foundation.
REPORTING FROM AN ALLIANCE FOR HEALTH POLICY WEBINAR
FDA okays emergency use of convalescent plasma for seriously ill COVID-19 patients
As the proportion of patients infected with COVID-19 continues to rise in the United States, the Food and Drug Administration is facilitating access to COVID-19 convalescent plasma for use in patients with serious or immediately life-threatening COVID-19 infections.
While clinical trials are underway to evaluate the safety and efficacy of administering convalescent plasma to patients with COVID-19, the FDA is granting clinicians permission for use of investigational convalescent plasma under single-patient emergency Investigational New Drug Applications (INDs), since no known cure exists and a vaccine is more than 1 year away from becoming available.
This allows the use of an investigational drug for the treatment of an individual patient by a licensed physician upon FDA authorization. This does not include the use of COVID-19 convalescent plasma for the prevention of infection, according to a statement issued by the agency on March 24.
“It is possible that convalescent plasma that contains antibodies to SARS-CoV-2 (the virus that causes COVID-19) might be effective against the infection,” the FDA statement reads. “Use of convalescent plasma has been studied in outbreaks of other respiratory infections, including the 2009-2010 H1N1 influenza virus pandemic, 2003 SARS-CoV-1 epidemic, and the 2012 MERS-CoV epidemic. Although promising, convalescent plasma has not been shown to be effective in every disease studied.”
“I think the FDA got caught initially a little flat-footed when it came to the development of COVID-19 tests, but they’re quickly catching up,” Peter J. Pitts, who was the FDA’s associate commissioner from 2002 to 2004, said in an interview. “I think that the attitude now is, ‘If it’s safe, let’s create a pathway to see how these things work in the real world.’ I think that’s going to be as true for treatments to lessen the symptoms and shorten the duration of the disease, as well as convalescent plasma as a potential alternative to a yet-to-be-developed vaccine.”
At the University of Washington School of Medicine, Seattle, Terry B. Gernsheimer, MD, and her colleagues are recruiting recovered COVID-19 patients to donate plasma for seriously ill patients affected with the virus. “The thought of using convalescent plasma makes total sense, because it’s immediately available, and it’s something that we can try to give people,” said Dr. Gernsheimer, a hematologist who is professor of medicine at the medical school. “It’s been used in China, and reports should be coming out shortly about their experience with this.”
In a case series that appeared in JAMA on March 27 (doi: 10.1001/jama.2020.4783), Chinese researchers led by Chenguang Shen, PhD, reported findings from five critically ill COVID-19 patients with acute respiratory distress syndrome who received a transfusion with convalescent plasma at Shenzhen Third People’s Hospital 10 and 22 days after hospital admission. The patients ranged in age from 36 to 73 years, three were men, and all were receiving mechanical ventilation at the time of treatment.
Dr. Shen and colleagues reported that viral loads decreased and became negative within 12 days following the transfusion. Three of the patients were discharged from the hospital after a length of stay that ranged from 51 to 55 days, and two remain in stable condition at 37 days after the transfusion. The researchers pointed out that all patients received antiviral agents, including interferon and lopinavir/ritonavir, during and following convalescent plasma treatment, “which also may have contributed to the viral clearance observed.”
Under the FDA policy on emergency IND use, COVID-19 convalescent plasma must only be collected from recovered individuals if they are eligible to donate blood, required testing must be performed, and the donation must be found suitable.
Potential donors “are going to be screened the way all blood donors are screened,” Dr. Gernsheimer said. “It’s not going to be any less safe than any unit of plasma that’s on the shelf that comes from our volunteer donors. There are always transfusion reactions that we have to worry about, [and] there are potentially unknown pathogens that we don’t yet know about that we are not yet testing for. It’s the regular risk we see with any unit of plasma.”
She added that COVID-19 survivors appear to start increasing their titer of the antibody around day 28. “We’ll be looking for recovered individuals who have had a documented infection, and whose symptoms started about 28 days before we collect,” she said.
The FDA advises clinicians to address several considerations for donor eligibility, including prior diagnosis of COVID-19 documented by a laboratory test; complete resolution of symptoms at least 14 days prior to donation; female donors negative for HLA antibodies or male donors, and negative results for COVID-19 either from one or more nasopharyngeal swab specimens or by a molecular diagnostic test from blood. [A partial list of available tests can be accessed on the FDA website.] The agency also advises that donors have defined SARS-CoV-2–neutralizing antibody titers, if testing can be conducted (optimally greater than 1:320).
Patients eligible to receive COVID-19 convalescent plasma must have a severe or immediately life-threatening infection with laboratory-confirmed COVID-19. The agency defines severe disease as dyspnea, respiratory frequency of 30 per minute or greater, blood oxygen saturation of 93% or less, partial pressure of arterial oxygen to fraction of inspired oxygen ratio of less than 300, and/or lung infiltrates of greater than 50% within 24-48 hours. Life-threatening disease is defined as respiratory failure, septic shock, and/or multiple organ dysfunction or failure. Patients must provide informed consent.
The potential risks of receiving COVID-19 convalescent plasma remain unknown, according to Dr. Gernsheimer. “What some people have thought about is, could there be such an inflammatory response with the virus that we would initially see these patients get worse?” she said. “My understanding is that has not occurred in China yet, but we don’t have all those data. But we always worry if we have something that’s going to cause inflammation around an infection, for example, that could initially make it more difficult to breathe if it’s a lung infection. So far, my understanding is that has not been seen.”
For COVID-19 convalescent plasma authorization requests that require a response within 4-8 hours, requesting clinicians may complete form 3296 and submit it by email to [email protected].
For COVID-19 convalescent plasma authorization requests that require a response in less than 4 hours, or if the clinician is unable to complete and submit form 3926 because of extenuating circumstances, verbal authorization can be sought by calling the FDA’s Office of Emergency Operations at 1-866-300-4374.
The FDA is working with the National Institutes of Health, the Centers for Disease Control and Prevention, and other government partners to develop protocols for use by multiple investigators in order to coordinate the collection and use of COVID-19 convalescent plasma.
“It’s crucial that data be captured for every patient so that we really understand what safety and effectiveness looks like on as close to a real-world level as we can, as quickly as we can,” said Mr. Pitts, who is president and cofounder of the Center for Medicine in the Public Interest, and who also does consulting work for the FDA. “I understand that health care professionals are overworked and overburdened right now. I applaud them for their heroic work. But that doesn’t mean that we can shirk off collecting the data. When I was at the FDA, I helped address the SARS epidemic. The agency attitude at that point was, ‘Let’s get things that just might work through the process, as long as the cure isn’t going to be worse than the disease.’ I think that’s the attitude that’s leading the charge today.”
As the proportion of patients infected with COVID-19 continues to rise in the United States, the Food and Drug Administration is facilitating access to COVID-19 convalescent plasma for use in patients with serious or immediately life-threatening COVID-19 infections.
While clinical trials are underway to evaluate the safety and efficacy of administering convalescent plasma to patients with COVID-19, the FDA is granting clinicians permission for use of investigational convalescent plasma under single-patient emergency Investigational New Drug Applications (INDs), since no known cure exists and a vaccine is more than 1 year away from becoming available.
This allows the use of an investigational drug for the treatment of an individual patient by a licensed physician upon FDA authorization. This does not include the use of COVID-19 convalescent plasma for the prevention of infection, according to a statement issued by the agency on March 24.
“It is possible that convalescent plasma that contains antibodies to SARS-CoV-2 (the virus that causes COVID-19) might be effective against the infection,” the FDA statement reads. “Use of convalescent plasma has been studied in outbreaks of other respiratory infections, including the 2009-2010 H1N1 influenza virus pandemic, 2003 SARS-CoV-1 epidemic, and the 2012 MERS-CoV epidemic. Although promising, convalescent plasma has not been shown to be effective in every disease studied.”
“I think the FDA got caught initially a little flat-footed when it came to the development of COVID-19 tests, but they’re quickly catching up,” Peter J. Pitts, who was the FDA’s associate commissioner from 2002 to 2004, said in an interview. “I think that the attitude now is, ‘If it’s safe, let’s create a pathway to see how these things work in the real world.’ I think that’s going to be as true for treatments to lessen the symptoms and shorten the duration of the disease, as well as convalescent plasma as a potential alternative to a yet-to-be-developed vaccine.”
At the University of Washington School of Medicine, Seattle, Terry B. Gernsheimer, MD, and her colleagues are recruiting recovered COVID-19 patients to donate plasma for seriously ill patients affected with the virus. “The thought of using convalescent plasma makes total sense, because it’s immediately available, and it’s something that we can try to give people,” said Dr. Gernsheimer, a hematologist who is professor of medicine at the medical school. “It’s been used in China, and reports should be coming out shortly about their experience with this.”
In a case series that appeared in JAMA on March 27 (doi: 10.1001/jama.2020.4783), Chinese researchers led by Chenguang Shen, PhD, reported findings from five critically ill COVID-19 patients with acute respiratory distress syndrome who received a transfusion with convalescent plasma at Shenzhen Third People’s Hospital 10 and 22 days after hospital admission. The patients ranged in age from 36 to 73 years, three were men, and all were receiving mechanical ventilation at the time of treatment.
Dr. Shen and colleagues reported that viral loads decreased and became negative within 12 days following the transfusion. Three of the patients were discharged from the hospital after a length of stay that ranged from 51 to 55 days, and two remain in stable condition at 37 days after the transfusion. The researchers pointed out that all patients received antiviral agents, including interferon and lopinavir/ritonavir, during and following convalescent plasma treatment, “which also may have contributed to the viral clearance observed.”
Under the FDA policy on emergency IND use, COVID-19 convalescent plasma must only be collected from recovered individuals if they are eligible to donate blood, required testing must be performed, and the donation must be found suitable.
Potential donors “are going to be screened the way all blood donors are screened,” Dr. Gernsheimer said. “It’s not going to be any less safe than any unit of plasma that’s on the shelf that comes from our volunteer donors. There are always transfusion reactions that we have to worry about, [and] there are potentially unknown pathogens that we don’t yet know about that we are not yet testing for. It’s the regular risk we see with any unit of plasma.”
She added that COVID-19 survivors appear to start increasing their titer of the antibody around day 28. “We’ll be looking for recovered individuals who have had a documented infection, and whose symptoms started about 28 days before we collect,” she said.
The FDA advises clinicians to address several considerations for donor eligibility, including prior diagnosis of COVID-19 documented by a laboratory test; complete resolution of symptoms at least 14 days prior to donation; female donors negative for HLA antibodies or male donors, and negative results for COVID-19 either from one or more nasopharyngeal swab specimens or by a molecular diagnostic test from blood. [A partial list of available tests can be accessed on the FDA website.] The agency also advises that donors have defined SARS-CoV-2–neutralizing antibody titers, if testing can be conducted (optimally greater than 1:320).
Patients eligible to receive COVID-19 convalescent plasma must have a severe or immediately life-threatening infection with laboratory-confirmed COVID-19. The agency defines severe disease as dyspnea, respiratory frequency of 30 per minute or greater, blood oxygen saturation of 93% or less, partial pressure of arterial oxygen to fraction of inspired oxygen ratio of less than 300, and/or lung infiltrates of greater than 50% within 24-48 hours. Life-threatening disease is defined as respiratory failure, septic shock, and/or multiple organ dysfunction or failure. Patients must provide informed consent.
The potential risks of receiving COVID-19 convalescent plasma remain unknown, according to Dr. Gernsheimer. “What some people have thought about is, could there be such an inflammatory response with the virus that we would initially see these patients get worse?” she said. “My understanding is that has not occurred in China yet, but we don’t have all those data. But we always worry if we have something that’s going to cause inflammation around an infection, for example, that could initially make it more difficult to breathe if it’s a lung infection. So far, my understanding is that has not been seen.”
For COVID-19 convalescent plasma authorization requests that require a response within 4-8 hours, requesting clinicians may complete form 3296 and submit it by email to [email protected].
For COVID-19 convalescent plasma authorization requests that require a response in less than 4 hours, or if the clinician is unable to complete and submit form 3926 because of extenuating circumstances, verbal authorization can be sought by calling the FDA’s Office of Emergency Operations at 1-866-300-4374.
The FDA is working with the National Institutes of Health, the Centers for Disease Control and Prevention, and other government partners to develop protocols for use by multiple investigators in order to coordinate the collection and use of COVID-19 convalescent plasma.
“It’s crucial that data be captured for every patient so that we really understand what safety and effectiveness looks like on as close to a real-world level as we can, as quickly as we can,” said Mr. Pitts, who is president and cofounder of the Center for Medicine in the Public Interest, and who also does consulting work for the FDA. “I understand that health care professionals are overworked and overburdened right now. I applaud them for their heroic work. But that doesn’t mean that we can shirk off collecting the data. When I was at the FDA, I helped address the SARS epidemic. The agency attitude at that point was, ‘Let’s get things that just might work through the process, as long as the cure isn’t going to be worse than the disease.’ I think that’s the attitude that’s leading the charge today.”
As the proportion of patients infected with COVID-19 continues to rise in the United States, the Food and Drug Administration is facilitating access to COVID-19 convalescent plasma for use in patients with serious or immediately life-threatening COVID-19 infections.
While clinical trials are underway to evaluate the safety and efficacy of administering convalescent plasma to patients with COVID-19, the FDA is granting clinicians permission for use of investigational convalescent plasma under single-patient emergency Investigational New Drug Applications (INDs), since no known cure exists and a vaccine is more than 1 year away from becoming available.
This allows the use of an investigational drug for the treatment of an individual patient by a licensed physician upon FDA authorization. This does not include the use of COVID-19 convalescent plasma for the prevention of infection, according to a statement issued by the agency on March 24.
“It is possible that convalescent plasma that contains antibodies to SARS-CoV-2 (the virus that causes COVID-19) might be effective against the infection,” the FDA statement reads. “Use of convalescent plasma has been studied in outbreaks of other respiratory infections, including the 2009-2010 H1N1 influenza virus pandemic, 2003 SARS-CoV-1 epidemic, and the 2012 MERS-CoV epidemic. Although promising, convalescent plasma has not been shown to be effective in every disease studied.”
“I think the FDA got caught initially a little flat-footed when it came to the development of COVID-19 tests, but they’re quickly catching up,” Peter J. Pitts, who was the FDA’s associate commissioner from 2002 to 2004, said in an interview. “I think that the attitude now is, ‘If it’s safe, let’s create a pathway to see how these things work in the real world.’ I think that’s going to be as true for treatments to lessen the symptoms and shorten the duration of the disease, as well as convalescent plasma as a potential alternative to a yet-to-be-developed vaccine.”
At the University of Washington School of Medicine, Seattle, Terry B. Gernsheimer, MD, and her colleagues are recruiting recovered COVID-19 patients to donate plasma for seriously ill patients affected with the virus. “The thought of using convalescent plasma makes total sense, because it’s immediately available, and it’s something that we can try to give people,” said Dr. Gernsheimer, a hematologist who is professor of medicine at the medical school. “It’s been used in China, and reports should be coming out shortly about their experience with this.”
In a case series that appeared in JAMA on March 27 (doi: 10.1001/jama.2020.4783), Chinese researchers led by Chenguang Shen, PhD, reported findings from five critically ill COVID-19 patients with acute respiratory distress syndrome who received a transfusion with convalescent plasma at Shenzhen Third People’s Hospital 10 and 22 days after hospital admission. The patients ranged in age from 36 to 73 years, three were men, and all were receiving mechanical ventilation at the time of treatment.
Dr. Shen and colleagues reported that viral loads decreased and became negative within 12 days following the transfusion. Three of the patients were discharged from the hospital after a length of stay that ranged from 51 to 55 days, and two remain in stable condition at 37 days after the transfusion. The researchers pointed out that all patients received antiviral agents, including interferon and lopinavir/ritonavir, during and following convalescent plasma treatment, “which also may have contributed to the viral clearance observed.”
Under the FDA policy on emergency IND use, COVID-19 convalescent plasma must only be collected from recovered individuals if they are eligible to donate blood, required testing must be performed, and the donation must be found suitable.
Potential donors “are going to be screened the way all blood donors are screened,” Dr. Gernsheimer said. “It’s not going to be any less safe than any unit of plasma that’s on the shelf that comes from our volunteer donors. There are always transfusion reactions that we have to worry about, [and] there are potentially unknown pathogens that we don’t yet know about that we are not yet testing for. It’s the regular risk we see with any unit of plasma.”
She added that COVID-19 survivors appear to start increasing their titer of the antibody around day 28. “We’ll be looking for recovered individuals who have had a documented infection, and whose symptoms started about 28 days before we collect,” she said.
The FDA advises clinicians to address several considerations for donor eligibility, including prior diagnosis of COVID-19 documented by a laboratory test; complete resolution of symptoms at least 14 days prior to donation; female donors negative for HLA antibodies or male donors, and negative results for COVID-19 either from one or more nasopharyngeal swab specimens or by a molecular diagnostic test from blood. [A partial list of available tests can be accessed on the FDA website.] The agency also advises that donors have defined SARS-CoV-2–neutralizing antibody titers, if testing can be conducted (optimally greater than 1:320).
Patients eligible to receive COVID-19 convalescent plasma must have a severe or immediately life-threatening infection with laboratory-confirmed COVID-19. The agency defines severe disease as dyspnea, respiratory frequency of 30 per minute or greater, blood oxygen saturation of 93% or less, partial pressure of arterial oxygen to fraction of inspired oxygen ratio of less than 300, and/or lung infiltrates of greater than 50% within 24-48 hours. Life-threatening disease is defined as respiratory failure, septic shock, and/or multiple organ dysfunction or failure. Patients must provide informed consent.
The potential risks of receiving COVID-19 convalescent plasma remain unknown, according to Dr. Gernsheimer. “What some people have thought about is, could there be such an inflammatory response with the virus that we would initially see these patients get worse?” she said. “My understanding is that has not occurred in China yet, but we don’t have all those data. But we always worry if we have something that’s going to cause inflammation around an infection, for example, that could initially make it more difficult to breathe if it’s a lung infection. So far, my understanding is that has not been seen.”
For COVID-19 convalescent plasma authorization requests that require a response within 4-8 hours, requesting clinicians may complete form 3296 and submit it by email to [email protected].
For COVID-19 convalescent plasma authorization requests that require a response in less than 4 hours, or if the clinician is unable to complete and submit form 3926 because of extenuating circumstances, verbal authorization can be sought by calling the FDA’s Office of Emergency Operations at 1-866-300-4374.
The FDA is working with the National Institutes of Health, the Centers for Disease Control and Prevention, and other government partners to develop protocols for use by multiple investigators in order to coordinate the collection and use of COVID-19 convalescent plasma.
“It’s crucial that data be captured for every patient so that we really understand what safety and effectiveness looks like on as close to a real-world level as we can, as quickly as we can,” said Mr. Pitts, who is president and cofounder of the Center for Medicine in the Public Interest, and who also does consulting work for the FDA. “I understand that health care professionals are overworked and overburdened right now. I applaud them for their heroic work. But that doesn’t mean that we can shirk off collecting the data. When I was at the FDA, I helped address the SARS epidemic. The agency attitude at that point was, ‘Let’s get things that just might work through the process, as long as the cure isn’t going to be worse than the disease.’ I think that’s the attitude that’s leading the charge today.”
Reports suggest possible in utero transmission of novel coronavirus 2019
Reports of three neonates with elevated IgM antibody concentrations whose mothers had COVID-19 in two articles raise questions about whether the infants may have been infected with the virus in utero.
The data, while provocative, “are not conclusive and do not prove in utero transmission” of the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), editorialists cautioned.
“The suggestion of in utero transmission rests on IgM detection in these 3 neonates, and IgM is a challenging way to diagnose many congenital infections,” David W. Kimberlin, MD, and Sergio Stagno, MD, of the division of pediatric infectious diseases at University of Alabama at Birmingham, wrote in their editorial. “IgM antibodies are too large to cross the placenta and so detection in a newborn reasonably could be assumed to reflect fetal production following in utero infection. However, most congenital infections are not diagnosed based on IgM detection because IgM assays can be prone to false-positive and false-negative results, along with cross-reactivity and testing challenges.”
None of the three infants had a positive reverse transcriptase–polymerase chain reaction (RT-PCR) test result, “so there is not virologic evidence for congenital infection in these cases to support the serologic suggestion of in utero transmission,” the editorialists noted.
Examining the possibility of vertical transmission
A prior case series of nine pregnant women found no transmission of the virus from mother to child, but the question of in utero transmission is not settled, said Lan Dong, MD, of the department of obstetrics and gynecology at Renmin Hospital of Wuhan University in China and colleagues. In their research letter, the investigators described a newborn with elevated IgM antibodies to novel coronavirus 2019 born to a mother with COVID-19. The infant was delivered by cesarean section February 22, 2020, at Renmin Hospital in a negative-pressure isolation room.
“The mother wore an N95 mask and did not hold the infant,” the researchers said. “The neonate had no symptoms and was immediately quarantined in the neonatal intensive care unit. At 2 hours of age, the SARS-CoV-2 IgG level was 140.32 AU/mL and the IgM level was 45.83 AU/mL.” Although the infant may have been infected at delivery, IgM antibodies usually take days to appear, Dr. Dong and colleagues wrote. “The infant’s repeatedly negative RT-PCR test results on nasopharyngeal swabs are difficult to explain, although these tests are not always positive with infection. ... Additional examination of maternal and newborn samples should be done to confirm this preliminary observation.”
A review of infants’ serologic characteristics
Hui Zeng, MD, of the department of laboratory medicine at Zhongnan Hospital of Wuhan University in China and colleagues retrospectively reviewed clinical records and laboratory results for six pregnant women with COVID-19, according to a study in JAMA. The women had mild clinical manifestations and were admitted to Zhongnan Hospital between February 16 and March 6. “All had cesarean deliveries in their third trimester in negative pressure isolation rooms,” the investigators said. “All mothers wore masks, and all medical staff wore protective suits and double masks. The infants were isolated from their mothers immediately after delivery.”
Two of the infants had elevated IgG and IgM concentrations. IgM “is not usually transferred from mother to fetus because of its larger macromolecular structure. ... Whether the placentas of women in this study were damaged and abnormal is unknown,” Dr. Zeng and colleagues said. “Alternatively, IgM could have been produced by the infant if the virus crossed the placenta.”
“Although these 2 studies deserve careful evaluation, more definitive evidence is needed” before physicians can “counsel pregnant women that their fetuses are at risk from congenital infection with SARS-CoV-2,” Dr. Kimberlin and Dr. Stagno concluded.
Dr. Dong and associates had no conflicts of interest. Their work was supported by the National Key Research and Development Project and others. Dr. Zeng and colleagues had no relevant financial disclosures. Their study was supported by grants from the National Natural Science Foundation of China and Zhongnan Hospital. Dr. Kimberlin and Dr. Stagno had no conflicts of interest.
SOURCE: Dong L et al. JAMA. 2020 Mar 26. doi: 10.1001/jama.2020.4621; Zeng H et al. JAMA. 2020 Mar 26. doi: 10.1001/jama.2020.4861.
Reports of three neonates with elevated IgM antibody concentrations whose mothers had COVID-19 in two articles raise questions about whether the infants may have been infected with the virus in utero.
The data, while provocative, “are not conclusive and do not prove in utero transmission” of the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), editorialists cautioned.
“The suggestion of in utero transmission rests on IgM detection in these 3 neonates, and IgM is a challenging way to diagnose many congenital infections,” David W. Kimberlin, MD, and Sergio Stagno, MD, of the division of pediatric infectious diseases at University of Alabama at Birmingham, wrote in their editorial. “IgM antibodies are too large to cross the placenta and so detection in a newborn reasonably could be assumed to reflect fetal production following in utero infection. However, most congenital infections are not diagnosed based on IgM detection because IgM assays can be prone to false-positive and false-negative results, along with cross-reactivity and testing challenges.”
None of the three infants had a positive reverse transcriptase–polymerase chain reaction (RT-PCR) test result, “so there is not virologic evidence for congenital infection in these cases to support the serologic suggestion of in utero transmission,” the editorialists noted.
Examining the possibility of vertical transmission
A prior case series of nine pregnant women found no transmission of the virus from mother to child, but the question of in utero transmission is not settled, said Lan Dong, MD, of the department of obstetrics and gynecology at Renmin Hospital of Wuhan University in China and colleagues. In their research letter, the investigators described a newborn with elevated IgM antibodies to novel coronavirus 2019 born to a mother with COVID-19. The infant was delivered by cesarean section February 22, 2020, at Renmin Hospital in a negative-pressure isolation room.
“The mother wore an N95 mask and did not hold the infant,” the researchers said. “The neonate had no symptoms and was immediately quarantined in the neonatal intensive care unit. At 2 hours of age, the SARS-CoV-2 IgG level was 140.32 AU/mL and the IgM level was 45.83 AU/mL.” Although the infant may have been infected at delivery, IgM antibodies usually take days to appear, Dr. Dong and colleagues wrote. “The infant’s repeatedly negative RT-PCR test results on nasopharyngeal swabs are difficult to explain, although these tests are not always positive with infection. ... Additional examination of maternal and newborn samples should be done to confirm this preliminary observation.”
A review of infants’ serologic characteristics
Hui Zeng, MD, of the department of laboratory medicine at Zhongnan Hospital of Wuhan University in China and colleagues retrospectively reviewed clinical records and laboratory results for six pregnant women with COVID-19, according to a study in JAMA. The women had mild clinical manifestations and were admitted to Zhongnan Hospital between February 16 and March 6. “All had cesarean deliveries in their third trimester in negative pressure isolation rooms,” the investigators said. “All mothers wore masks, and all medical staff wore protective suits and double masks. The infants were isolated from their mothers immediately after delivery.”
Two of the infants had elevated IgG and IgM concentrations. IgM “is not usually transferred from mother to fetus because of its larger macromolecular structure. ... Whether the placentas of women in this study were damaged and abnormal is unknown,” Dr. Zeng and colleagues said. “Alternatively, IgM could have been produced by the infant if the virus crossed the placenta.”
“Although these 2 studies deserve careful evaluation, more definitive evidence is needed” before physicians can “counsel pregnant women that their fetuses are at risk from congenital infection with SARS-CoV-2,” Dr. Kimberlin and Dr. Stagno concluded.
Dr. Dong and associates had no conflicts of interest. Their work was supported by the National Key Research and Development Project and others. Dr. Zeng and colleagues had no relevant financial disclosures. Their study was supported by grants from the National Natural Science Foundation of China and Zhongnan Hospital. Dr. Kimberlin and Dr. Stagno had no conflicts of interest.
SOURCE: Dong L et al. JAMA. 2020 Mar 26. doi: 10.1001/jama.2020.4621; Zeng H et al. JAMA. 2020 Mar 26. doi: 10.1001/jama.2020.4861.
Reports of three neonates with elevated IgM antibody concentrations whose mothers had COVID-19 in two articles raise questions about whether the infants may have been infected with the virus in utero.
The data, while provocative, “are not conclusive and do not prove in utero transmission” of the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), editorialists cautioned.
“The suggestion of in utero transmission rests on IgM detection in these 3 neonates, and IgM is a challenging way to diagnose many congenital infections,” David W. Kimberlin, MD, and Sergio Stagno, MD, of the division of pediatric infectious diseases at University of Alabama at Birmingham, wrote in their editorial. “IgM antibodies are too large to cross the placenta and so detection in a newborn reasonably could be assumed to reflect fetal production following in utero infection. However, most congenital infections are not diagnosed based on IgM detection because IgM assays can be prone to false-positive and false-negative results, along with cross-reactivity and testing challenges.”
None of the three infants had a positive reverse transcriptase–polymerase chain reaction (RT-PCR) test result, “so there is not virologic evidence for congenital infection in these cases to support the serologic suggestion of in utero transmission,” the editorialists noted.
Examining the possibility of vertical transmission
A prior case series of nine pregnant women found no transmission of the virus from mother to child, but the question of in utero transmission is not settled, said Lan Dong, MD, of the department of obstetrics and gynecology at Renmin Hospital of Wuhan University in China and colleagues. In their research letter, the investigators described a newborn with elevated IgM antibodies to novel coronavirus 2019 born to a mother with COVID-19. The infant was delivered by cesarean section February 22, 2020, at Renmin Hospital in a negative-pressure isolation room.
“The mother wore an N95 mask and did not hold the infant,” the researchers said. “The neonate had no symptoms and was immediately quarantined in the neonatal intensive care unit. At 2 hours of age, the SARS-CoV-2 IgG level was 140.32 AU/mL and the IgM level was 45.83 AU/mL.” Although the infant may have been infected at delivery, IgM antibodies usually take days to appear, Dr. Dong and colleagues wrote. “The infant’s repeatedly negative RT-PCR test results on nasopharyngeal swabs are difficult to explain, although these tests are not always positive with infection. ... Additional examination of maternal and newborn samples should be done to confirm this preliminary observation.”
A review of infants’ serologic characteristics
Hui Zeng, MD, of the department of laboratory medicine at Zhongnan Hospital of Wuhan University in China and colleagues retrospectively reviewed clinical records and laboratory results for six pregnant women with COVID-19, according to a study in JAMA. The women had mild clinical manifestations and were admitted to Zhongnan Hospital between February 16 and March 6. “All had cesarean deliveries in their third trimester in negative pressure isolation rooms,” the investigators said. “All mothers wore masks, and all medical staff wore protective suits and double masks. The infants were isolated from their mothers immediately after delivery.”
Two of the infants had elevated IgG and IgM concentrations. IgM “is not usually transferred from mother to fetus because of its larger macromolecular structure. ... Whether the placentas of women in this study were damaged and abnormal is unknown,” Dr. Zeng and colleagues said. “Alternatively, IgM could have been produced by the infant if the virus crossed the placenta.”
“Although these 2 studies deserve careful evaluation, more definitive evidence is needed” before physicians can “counsel pregnant women that their fetuses are at risk from congenital infection with SARS-CoV-2,” Dr. Kimberlin and Dr. Stagno concluded.
Dr. Dong and associates had no conflicts of interest. Their work was supported by the National Key Research and Development Project and others. Dr. Zeng and colleagues had no relevant financial disclosures. Their study was supported by grants from the National Natural Science Foundation of China and Zhongnan Hospital. Dr. Kimberlin and Dr. Stagno had no conflicts of interest.
SOURCE: Dong L et al. JAMA. 2020 Mar 26. doi: 10.1001/jama.2020.4621; Zeng H et al. JAMA. 2020 Mar 26. doi: 10.1001/jama.2020.4861.
FROM JAMA
Despite strict controls, some infants born to mothers with COVID-19 appear infected
Despite implementation of strict infection control and prevention procedures in a hospital in Wuhan, China, according to Lingkong Zeng, MD, of the department of neonatology at Wuhan Children’s Hospital, and associates.
Thirty-three neonates born to mothers with COVID-19 were included in the study, published as a research letter in JAMA Pediatrics. Of this group, three neonates (9%) were confirmed to be infected with the novel coronavirus 2019 at 2 and 4 days of life through nasopharyngeal and anal swabs.
Of the three infected neonates, two were born at 40 weeks’ gestation and the third was born at 31 weeks. The two full-term infants had mild symptoms such as lethargy and fever and were negative for the virus at 6 days of life. The preterm infant had somewhat worse symptoms, but the investigators acknowledged that “the most seriously ill neonate may have been symptomatic from prematurity, asphyxia, and sepsis, rather than [the novel coronavirus 2019] infection.” They added that outcomes for all three neonates were favorable, consistent with past research.
“Because strict infection control and prevention procedures were implemented during the delivery, it is likely that the sources of [novel coronavirus 2019] in the neonates’ upper respiratory tracts or anuses were maternal in origin,” Dr. Zeng and associates surmised.
While previous studies have shown no evidence of COVID-19 transmission between mothers and neonates, and all samples, including amniotic fluid, cord blood, and breast milk, were negative for the novel coronavirus 2019, “vertical maternal-fetal transmission cannot be ruled out in the current cohort. Therefore, it is crucial to screen pregnant women and implement strict infection control measures, quarantine of infected mothers, and close monitoring of neonates at risk of COVID-19,” the investigators concluded.
The study authors reported that they had no conflicts of interest.
SOURCE: Zeng L et al. JAMA Pediatrics. 2020 Mar 26. doi: 10.1001/jamapediatrics.2020.0878.
Despite implementation of strict infection control and prevention procedures in a hospital in Wuhan, China, according to Lingkong Zeng, MD, of the department of neonatology at Wuhan Children’s Hospital, and associates.
Thirty-three neonates born to mothers with COVID-19 were included in the study, published as a research letter in JAMA Pediatrics. Of this group, three neonates (9%) were confirmed to be infected with the novel coronavirus 2019 at 2 and 4 days of life through nasopharyngeal and anal swabs.
Of the three infected neonates, two were born at 40 weeks’ gestation and the third was born at 31 weeks. The two full-term infants had mild symptoms such as lethargy and fever and were negative for the virus at 6 days of life. The preterm infant had somewhat worse symptoms, but the investigators acknowledged that “the most seriously ill neonate may have been symptomatic from prematurity, asphyxia, and sepsis, rather than [the novel coronavirus 2019] infection.” They added that outcomes for all three neonates were favorable, consistent with past research.
“Because strict infection control and prevention procedures were implemented during the delivery, it is likely that the sources of [novel coronavirus 2019] in the neonates’ upper respiratory tracts or anuses were maternal in origin,” Dr. Zeng and associates surmised.
While previous studies have shown no evidence of COVID-19 transmission between mothers and neonates, and all samples, including amniotic fluid, cord blood, and breast milk, were negative for the novel coronavirus 2019, “vertical maternal-fetal transmission cannot be ruled out in the current cohort. Therefore, it is crucial to screen pregnant women and implement strict infection control measures, quarantine of infected mothers, and close monitoring of neonates at risk of COVID-19,” the investigators concluded.
The study authors reported that they had no conflicts of interest.
SOURCE: Zeng L et al. JAMA Pediatrics. 2020 Mar 26. doi: 10.1001/jamapediatrics.2020.0878.
Despite implementation of strict infection control and prevention procedures in a hospital in Wuhan, China, according to Lingkong Zeng, MD, of the department of neonatology at Wuhan Children’s Hospital, and associates.
Thirty-three neonates born to mothers with COVID-19 were included in the study, published as a research letter in JAMA Pediatrics. Of this group, three neonates (9%) were confirmed to be infected with the novel coronavirus 2019 at 2 and 4 days of life through nasopharyngeal and anal swabs.
Of the three infected neonates, two were born at 40 weeks’ gestation and the third was born at 31 weeks. The two full-term infants had mild symptoms such as lethargy and fever and were negative for the virus at 6 days of life. The preterm infant had somewhat worse symptoms, but the investigators acknowledged that “the most seriously ill neonate may have been symptomatic from prematurity, asphyxia, and sepsis, rather than [the novel coronavirus 2019] infection.” They added that outcomes for all three neonates were favorable, consistent with past research.
“Because strict infection control and prevention procedures were implemented during the delivery, it is likely that the sources of [novel coronavirus 2019] in the neonates’ upper respiratory tracts or anuses were maternal in origin,” Dr. Zeng and associates surmised.
While previous studies have shown no evidence of COVID-19 transmission between mothers and neonates, and all samples, including amniotic fluid, cord blood, and breast milk, were negative for the novel coronavirus 2019, “vertical maternal-fetal transmission cannot be ruled out in the current cohort. Therefore, it is crucial to screen pregnant women and implement strict infection control measures, quarantine of infected mothers, and close monitoring of neonates at risk of COVID-19,” the investigators concluded.
The study authors reported that they had no conflicts of interest.
SOURCE: Zeng L et al. JAMA Pediatrics. 2020 Mar 26. doi: 10.1001/jamapediatrics.2020.0878.
FROM JAMA PEDIATRICS
Wuhan data link COVID-19 with myocardial damage
The first data on myocardial injury linked with COVID-19 disease during the start of the pandemic in Wuhan, China serves as a “wake up call” for clinicians and the general public on what the United States and other Western countries can expect as the SARS-CoV-2 virus spreads and case numbers mount: a potentially “daunting” toll of deaths as an infection with a tendency to be most severe in patients with underlying cardiovascular disease hits populations that include large numbers of such patients.
“A consistent picture emerges” from two reports on a total of 603 COVID-19 patients treated at two academic hospitals in Wuhan, which described “remarkably similar characteristics of patients who develop myocardial injury” associated with their infection. “Patients who develop myocardial injury with COVID-19 have clinical evidence of higher acuity, with a higher incidence of acute respiratory distress syndrome and more frequent need for assisted ventilation than those without myocardial injury, and the patients who are more prone to have myocardial injury are “older patients with preexisting cardiovascular complications and diabetes,” Robert O. Bonow, MD, and coauthors wrote in an editorial published online (JAMA Cardiol. 2020 Mar 27. doi: 10.1001/jamacardio.2020.1105).
These new findings have special relevance to the United States and other Western countries because of their substantial numbers of elderly patients with cardiovascular diseases, said Dr. Bonow, professor of medicine at Northwestern University, Chicago, and coauthors.
One of the two reports cited in the editorial reviewed 416 patients hospitalized at Renmin Hospital in Wuhan during the period of Jan. 20 to Feb. 10, 2020, with confirmed COVID-19 disease, and found that 20% of the cohort had evidence of cardiac injury, defined as blood levels of the high-sensitivity troponin I cardiac biomarker above the 99th-percentile upper reference limit, regardless of new abnormalities in electrocardiography and echocardiography.
The analysis also showed that patients with myocardial injury had a significantly higher in-hospital mortality rate, 51%, compared with a 5% mortality rate among patients without myocardial injury, and among patients with myocardial injury those with elevated high-sensitivity troponin I had an even higher mortality rate (JAMA Cardiol. 2020 Mar 25. doi: 10.1001/jamacardio.2020.0950).
A second review of 187 confirmed COVID-19 cases at Seventh Hospital in Wuhan during the period of Jan. 23 to Feb. 23, 2020, showed similar findings, with a 28% prevalence of myocardial injury at admission based on an elevated level of plasma troponin T (TnT), and 35% had cardiovascular disease (CVD) including hypertension, coronary heart disease, and cardiomyopathy. Elevated TnT levels and CVD at entry each linked with substantially increased mortality. The incidence of death among patients with elevated TnT and no underlying CVD was 38% compared with 8% among patients without elevated TnT or underlying CVD. Among patients admitted with underlying CVD those who also had an elevated TnT had a 69% death rate during hospitalization compared with a 13% rate in those without TnT elevation (JAMA Cardiol. 2020 Mar 27. doi: 10.1001/jamacardio.2020.1017).
Dr. Bonow and coauthors noted that patients with chronic coronary artery disease have a heightened risk for developing acute coronary syndrome during acute infection, potentially resulting from a severe increase in myocardial demand during infection, or severe systemic inflammatory stress that could result in atherosclerotic plaque instability and rupture as well as vascular and myocardial inflammation.
In addition, patients with heart failure are prone to hemodynamic instability during severe infection. “Thus it is anticipated that patients with underlying cardiovascular diseases, which are more prevalent in older adults, would be susceptible to higher risks of adverse outcomes and death during the severe and aggressive inflammatory responses to COVID-19 than individuals who are younger and healthier,” they wrote.
They also cited the potential for acute or fulminant myocarditis as well as new-onset heart failure caused by the SARS-CoV-2 virus that causes COVID-19 disease based on experience with the related Middle East respiratory syndrome coronavirus. Another concerning observation is that the SARS-CoV-2 virus binds to the angiotensin-converting enzyme 2 protein on cell surfaces as its main entry receptor, “raising the possibility of direct viral infection of vascular endothelium and myocardium,” a process that itself could produce myocardial injury and myocarditis.
These new findings from COVID-19 patients in Wuhan represent early data from what has become a global pandemic, and raise questions about generalizability, but for the time being a key message from these early cases is that prevention of SARS-CoV-2 infection is paramount. “Until we know more, the populations described in these primary data reports should be most observant of strict hand hygiene, social distancing, and, where available, COVID-19 testing,” the authors said.
Dr. Bonow and coauthors had no disclosures.
The first data on myocardial injury linked with COVID-19 disease during the start of the pandemic in Wuhan, China serves as a “wake up call” for clinicians and the general public on what the United States and other Western countries can expect as the SARS-CoV-2 virus spreads and case numbers mount: a potentially “daunting” toll of deaths as an infection with a tendency to be most severe in patients with underlying cardiovascular disease hits populations that include large numbers of such patients.
“A consistent picture emerges” from two reports on a total of 603 COVID-19 patients treated at two academic hospitals in Wuhan, which described “remarkably similar characteristics of patients who develop myocardial injury” associated with their infection. “Patients who develop myocardial injury with COVID-19 have clinical evidence of higher acuity, with a higher incidence of acute respiratory distress syndrome and more frequent need for assisted ventilation than those without myocardial injury, and the patients who are more prone to have myocardial injury are “older patients with preexisting cardiovascular complications and diabetes,” Robert O. Bonow, MD, and coauthors wrote in an editorial published online (JAMA Cardiol. 2020 Mar 27. doi: 10.1001/jamacardio.2020.1105).
These new findings have special relevance to the United States and other Western countries because of their substantial numbers of elderly patients with cardiovascular diseases, said Dr. Bonow, professor of medicine at Northwestern University, Chicago, and coauthors.
One of the two reports cited in the editorial reviewed 416 patients hospitalized at Renmin Hospital in Wuhan during the period of Jan. 20 to Feb. 10, 2020, with confirmed COVID-19 disease, and found that 20% of the cohort had evidence of cardiac injury, defined as blood levels of the high-sensitivity troponin I cardiac biomarker above the 99th-percentile upper reference limit, regardless of new abnormalities in electrocardiography and echocardiography.
The analysis also showed that patients with myocardial injury had a significantly higher in-hospital mortality rate, 51%, compared with a 5% mortality rate among patients without myocardial injury, and among patients with myocardial injury those with elevated high-sensitivity troponin I had an even higher mortality rate (JAMA Cardiol. 2020 Mar 25. doi: 10.1001/jamacardio.2020.0950).
A second review of 187 confirmed COVID-19 cases at Seventh Hospital in Wuhan during the period of Jan. 23 to Feb. 23, 2020, showed similar findings, with a 28% prevalence of myocardial injury at admission based on an elevated level of plasma troponin T (TnT), and 35% had cardiovascular disease (CVD) including hypertension, coronary heart disease, and cardiomyopathy. Elevated TnT levels and CVD at entry each linked with substantially increased mortality. The incidence of death among patients with elevated TnT and no underlying CVD was 38% compared with 8% among patients without elevated TnT or underlying CVD. Among patients admitted with underlying CVD those who also had an elevated TnT had a 69% death rate during hospitalization compared with a 13% rate in those without TnT elevation (JAMA Cardiol. 2020 Mar 27. doi: 10.1001/jamacardio.2020.1017).
Dr. Bonow and coauthors noted that patients with chronic coronary artery disease have a heightened risk for developing acute coronary syndrome during acute infection, potentially resulting from a severe increase in myocardial demand during infection, or severe systemic inflammatory stress that could result in atherosclerotic plaque instability and rupture as well as vascular and myocardial inflammation.
In addition, patients with heart failure are prone to hemodynamic instability during severe infection. “Thus it is anticipated that patients with underlying cardiovascular diseases, which are more prevalent in older adults, would be susceptible to higher risks of adverse outcomes and death during the severe and aggressive inflammatory responses to COVID-19 than individuals who are younger and healthier,” they wrote.
They also cited the potential for acute or fulminant myocarditis as well as new-onset heart failure caused by the SARS-CoV-2 virus that causes COVID-19 disease based on experience with the related Middle East respiratory syndrome coronavirus. Another concerning observation is that the SARS-CoV-2 virus binds to the angiotensin-converting enzyme 2 protein on cell surfaces as its main entry receptor, “raising the possibility of direct viral infection of vascular endothelium and myocardium,” a process that itself could produce myocardial injury and myocarditis.
These new findings from COVID-19 patients in Wuhan represent early data from what has become a global pandemic, and raise questions about generalizability, but for the time being a key message from these early cases is that prevention of SARS-CoV-2 infection is paramount. “Until we know more, the populations described in these primary data reports should be most observant of strict hand hygiene, social distancing, and, where available, COVID-19 testing,” the authors said.
Dr. Bonow and coauthors had no disclosures.
The first data on myocardial injury linked with COVID-19 disease during the start of the pandemic in Wuhan, China serves as a “wake up call” for clinicians and the general public on what the United States and other Western countries can expect as the SARS-CoV-2 virus spreads and case numbers mount: a potentially “daunting” toll of deaths as an infection with a tendency to be most severe in patients with underlying cardiovascular disease hits populations that include large numbers of such patients.
“A consistent picture emerges” from two reports on a total of 603 COVID-19 patients treated at two academic hospitals in Wuhan, which described “remarkably similar characteristics of patients who develop myocardial injury” associated with their infection. “Patients who develop myocardial injury with COVID-19 have clinical evidence of higher acuity, with a higher incidence of acute respiratory distress syndrome and more frequent need for assisted ventilation than those without myocardial injury, and the patients who are more prone to have myocardial injury are “older patients with preexisting cardiovascular complications and diabetes,” Robert O. Bonow, MD, and coauthors wrote in an editorial published online (JAMA Cardiol. 2020 Mar 27. doi: 10.1001/jamacardio.2020.1105).
These new findings have special relevance to the United States and other Western countries because of their substantial numbers of elderly patients with cardiovascular diseases, said Dr. Bonow, professor of medicine at Northwestern University, Chicago, and coauthors.
One of the two reports cited in the editorial reviewed 416 patients hospitalized at Renmin Hospital in Wuhan during the period of Jan. 20 to Feb. 10, 2020, with confirmed COVID-19 disease, and found that 20% of the cohort had evidence of cardiac injury, defined as blood levels of the high-sensitivity troponin I cardiac biomarker above the 99th-percentile upper reference limit, regardless of new abnormalities in electrocardiography and echocardiography.
The analysis also showed that patients with myocardial injury had a significantly higher in-hospital mortality rate, 51%, compared with a 5% mortality rate among patients without myocardial injury, and among patients with myocardial injury those with elevated high-sensitivity troponin I had an even higher mortality rate (JAMA Cardiol. 2020 Mar 25. doi: 10.1001/jamacardio.2020.0950).
A second review of 187 confirmed COVID-19 cases at Seventh Hospital in Wuhan during the period of Jan. 23 to Feb. 23, 2020, showed similar findings, with a 28% prevalence of myocardial injury at admission based on an elevated level of plasma troponin T (TnT), and 35% had cardiovascular disease (CVD) including hypertension, coronary heart disease, and cardiomyopathy. Elevated TnT levels and CVD at entry each linked with substantially increased mortality. The incidence of death among patients with elevated TnT and no underlying CVD was 38% compared with 8% among patients without elevated TnT or underlying CVD. Among patients admitted with underlying CVD those who also had an elevated TnT had a 69% death rate during hospitalization compared with a 13% rate in those without TnT elevation (JAMA Cardiol. 2020 Mar 27. doi: 10.1001/jamacardio.2020.1017).
Dr. Bonow and coauthors noted that patients with chronic coronary artery disease have a heightened risk for developing acute coronary syndrome during acute infection, potentially resulting from a severe increase in myocardial demand during infection, or severe systemic inflammatory stress that could result in atherosclerotic plaque instability and rupture as well as vascular and myocardial inflammation.
In addition, patients with heart failure are prone to hemodynamic instability during severe infection. “Thus it is anticipated that patients with underlying cardiovascular diseases, which are more prevalent in older adults, would be susceptible to higher risks of adverse outcomes and death during the severe and aggressive inflammatory responses to COVID-19 than individuals who are younger and healthier,” they wrote.
They also cited the potential for acute or fulminant myocarditis as well as new-onset heart failure caused by the SARS-CoV-2 virus that causes COVID-19 disease based on experience with the related Middle East respiratory syndrome coronavirus. Another concerning observation is that the SARS-CoV-2 virus binds to the angiotensin-converting enzyme 2 protein on cell surfaces as its main entry receptor, “raising the possibility of direct viral infection of vascular endothelium and myocardium,” a process that itself could produce myocardial injury and myocarditis.
These new findings from COVID-19 patients in Wuhan represent early data from what has become a global pandemic, and raise questions about generalizability, but for the time being a key message from these early cases is that prevention of SARS-CoV-2 infection is paramount. “Until we know more, the populations described in these primary data reports should be most observant of strict hand hygiene, social distancing, and, where available, COVID-19 testing,” the authors said.
Dr. Bonow and coauthors had no disclosures.
FROM JAMA CARDIOLOGY
Rheumatologists seek to reassure amid hydroxychloroquine shortage
Physicians and pharmacists are reporting shortages of hydroxychloroquine and chloroquine following President Trump’s promotion of the medications as potential COVID-19 treatments, leaving patients with rheumatic diseases wondering how it will impact their access.
The American Medical Association, the American Pharmacists Association, and the American Society of Health-System Pharmacists, issued a joint statement that strongly opposed prophylactic prescribing of these medications for COVID-19 or stockpiling them in anticipation of use for COVID-19. The concerns over shortages have also prompted the American College of Rheumatology, American Academy of Dermatology, Arthritis Foundation, and Lupus Foundation of America to send a joint statement to the Trump administration and the nation’s governors highlighting critical hydroxychloroquine access issues and asking policymakers to work together with health care providers and patient communities to ensure continued availability of these drugs.
Now
In a Q and A interview, NYU Langone Health rheumatology division director and Lupus Center director Jill P. Buyon, MD, and associate professor of rheumatology, Peter M. Izmirly, MD, noted that, while shortages have been reported across the United States because of large increases in off-label prescribing, many of the drugs’ manufacturers have committed to donating millions of doses and/or stepping up production to meet demand.
Later in this article, Michael H. Pillinger, MD, a rheumatologist and professor of medicine, biochemistry, and molecular pharmacology at NYU Langone Health, New York, answered questions about a new multicenter study called COLCORONA getting underway to test the anti-inflammatory drug colchicine. The answers in this Q&A have been edited for length and clarity.
Questions about hydroxychloroquine shortage
Q: What is the current situation with hydroxychloroquine in your practice?
A: We have been getting calls from our patients asking about getting refills for hydroxychloroquine. Our group has been calling local pharmacies asking about the availability of hydroxychloroquine, and we are compiling a list of pharmacies in New York with current availabilities to share with patients. We are somewhat limited by our electronic health record system, Epic, which can only send a prescription to one pharmacy, so that has placed some limitations on knowing where it is available. Some pharmacies have not had hydroxychloroquine available, while others have. We have also been encouraging patients to check online and look for mail-order possibilities for 90-day supplies.
Nearly all prescriptions are for generic hydroxychloroquine. Branded hydroxychloroquine (Plaquenil) is much more expensive, and we can run into obstacles with getting it approved by insurers, too.
Q: What are you telling patients who seek to refill their prescription or call with concerns? Is it feasible for patients to stop hydroxychloroquine or cut their dosage if necessary?
A: If someone’s been on hydroxychloroquine and has benefited from its use there’s no reason to come off it at this time, and given the possibility that it may have an effect on COVID-19, that is all the better. But we want to reassure patients that they can get the drug and that it is not difficult to manufacture.
Given the significantly higher risk of disease flare that was first described in lupus patients who discontinued hydroxychloroquine in the Canadian Hydroxychloroquine Study Group’s 1991 randomized, controlled trial, it is not advisable for patients to stop the drug.
Some patients do split their dosage day-to-day if they are taking less than 400 mg daily, such that someone taking 300 mg daily may take two 200-mg tablets one day and just one 200-mg tablet the next day, and so on. To avoid eye toxicity that can occur after years of taking the drug, hydroxychloroquine is generally prescribed based on weight at 5 mg/kg.
The drug also stays in the body for quite a while [often up to 3 months and even longer], so that is helpful for patients to know.
Given the current situation and the possibility of its effectiveness against COVID-19, it is ironic that we are actually trying to recruit older lupus patients who have had long-term stable disease while on hydroxychloroquine to a trial of stopping the drug to reduce the risk of developing the side effect of retinopathy. We want to see if patients can safely withdraw hydroxychloroquine without flaring, so we hope to not run into enrollment difficulties based on the current situation with COVID-19.
Q: How do you view the balance between having enough hydroxychloroquine for patients with lupus or other rheumatic diseases and its use in COVID-19 patients?
A: We want to reassure patients that hydroxychloroquine will be available, and there is no reason to hoard the drug or to worry excessively about being unable to obtain it. Efforts to increase production by Mylan, Teva, Sanofi, Novartis, and other manufacturers of hydroxychloroquine should really help out.
Q: Are there pharmacy restrictions on prescription amounts?
A: This is not universal at this time, but some institutions are cutting back and offering only 1-month supplies.
Colchicine COVID-19 trial underway
Dr. Pillinger, of NYU Langone Health, explored the COLCORONA study of colchicine as a treatment for people infected with COVID-19 and the worry that shortage concerns may arise for it, too.
Q: What is the general availability of colchicine and its susceptibility to shortage?
A: There are two major manufacturers of colchicine in the United States, Takeda and Hikma, who together manufacture the majority of the drug.
The greatest use of colchicine in the United States is for gout, which affects approximately 4 million Americans, but the drug is not used chronically, so a much smaller number of patients are using colchicine at any one time. Colchicine is also used for other inflammatory conditions, primarily calcium pyrophosphate crystal disease and familial Mediterranean fever (FMF is rare in the United States). Cardiologists also regularly prescribe colchicine in pericarditis for short-term use. Physicians may use it off label for other purposes, too.
Overall, the number of patients using colchicine is much larger than that for the use of hydroxychloroquine, for example, suggesting that the immediate risk of shortage could be lower. However, if individuals started using it off label, or prescribing inappropriately for the COVID-19 indication, the supply would rapidly run short.
Q: What other points are there to consider regarding the use of colchicine to treat COVID-19?
A: There is no evidence – zero – that colchicine has any benefit for COVID-19, not even case reports. There is some rationale that it might be beneficial, but that is exactly why the COLCORONA trial would be logical to try.
The COLCORONA trial is exactly the kind of trial that would be needed for assessing colchicine, and it is big enough and happening quickly enough to get an answer. But if people start to use colchicine off label, we may never know the truth.
While colchicine can be used safely in most people, it can be very problematic and requires an experienced doctor’s supervision. Overdoses can be fatal, and colchicine interacts with many drugs, all of which require dose adjustment and some of which must be stopped in order to use colchicine – it isn’t candy. Some of the other drugs being looked at for COVID-19 in fact may interact with colchicine.
Colchicine must also be dose adjusted for kidney disease, and, in some of the COVID-19 patients, kidney function changes rapidly. So again, its use would require expert supervision even if there were evidence for its utility.
The side effects of colchicine, if mis-dosed, can be very unpleasant, including nausea, vomiting, and diarrhea. Even at the apparent right dose, some people will get these side effects, so colchicine has to be something that works to make the risk/benefit ratio worth it.
Some preparations of colchicine are made combined with probenecid, a gout drug. This is even more problematic because probenecid can raise the level of drugs excreted by the kidney and could affect other treatments.
So in sum, what may be a good idea in theory can turn out to be a disastrous idea in practice, and here we have nothing but theory. This is not an agent to use randomly; the studies will be rushed out quickly and hopefully will give us the knowledge to know what to do.
Dr. Izmirly and Dr. Buyon said they have research grants with the National Institutes of Health to study hydroxychloroquine in patients with lupus and in anti–SSA/Ro-positive pregnant women with a previous child with congenital heart block. Dr. Pillinger reports that he has an investigator-initiated grant from Hikma to study colchicine in osteoarthritis.
This article was reformatted on 3/30/2020 for clarity.
Physicians and pharmacists are reporting shortages of hydroxychloroquine and chloroquine following President Trump’s promotion of the medications as potential COVID-19 treatments, leaving patients with rheumatic diseases wondering how it will impact their access.
The American Medical Association, the American Pharmacists Association, and the American Society of Health-System Pharmacists, issued a joint statement that strongly opposed prophylactic prescribing of these medications for COVID-19 or stockpiling them in anticipation of use for COVID-19. The concerns over shortages have also prompted the American College of Rheumatology, American Academy of Dermatology, Arthritis Foundation, and Lupus Foundation of America to send a joint statement to the Trump administration and the nation’s governors highlighting critical hydroxychloroquine access issues and asking policymakers to work together with health care providers and patient communities to ensure continued availability of these drugs.
Now
In a Q and A interview, NYU Langone Health rheumatology division director and Lupus Center director Jill P. Buyon, MD, and associate professor of rheumatology, Peter M. Izmirly, MD, noted that, while shortages have been reported across the United States because of large increases in off-label prescribing, many of the drugs’ manufacturers have committed to donating millions of doses and/or stepping up production to meet demand.
Later in this article, Michael H. Pillinger, MD, a rheumatologist and professor of medicine, biochemistry, and molecular pharmacology at NYU Langone Health, New York, answered questions about a new multicenter study called COLCORONA getting underway to test the anti-inflammatory drug colchicine. The answers in this Q&A have been edited for length and clarity.
Questions about hydroxychloroquine shortage
Q: What is the current situation with hydroxychloroquine in your practice?
A: We have been getting calls from our patients asking about getting refills for hydroxychloroquine. Our group has been calling local pharmacies asking about the availability of hydroxychloroquine, and we are compiling a list of pharmacies in New York with current availabilities to share with patients. We are somewhat limited by our electronic health record system, Epic, which can only send a prescription to one pharmacy, so that has placed some limitations on knowing where it is available. Some pharmacies have not had hydroxychloroquine available, while others have. We have also been encouraging patients to check online and look for mail-order possibilities for 90-day supplies.
Nearly all prescriptions are for generic hydroxychloroquine. Branded hydroxychloroquine (Plaquenil) is much more expensive, and we can run into obstacles with getting it approved by insurers, too.
Q: What are you telling patients who seek to refill their prescription or call with concerns? Is it feasible for patients to stop hydroxychloroquine or cut their dosage if necessary?
A: If someone’s been on hydroxychloroquine and has benefited from its use there’s no reason to come off it at this time, and given the possibility that it may have an effect on COVID-19, that is all the better. But we want to reassure patients that they can get the drug and that it is not difficult to manufacture.
Given the significantly higher risk of disease flare that was first described in lupus patients who discontinued hydroxychloroquine in the Canadian Hydroxychloroquine Study Group’s 1991 randomized, controlled trial, it is not advisable for patients to stop the drug.
Some patients do split their dosage day-to-day if they are taking less than 400 mg daily, such that someone taking 300 mg daily may take two 200-mg tablets one day and just one 200-mg tablet the next day, and so on. To avoid eye toxicity that can occur after years of taking the drug, hydroxychloroquine is generally prescribed based on weight at 5 mg/kg.
The drug also stays in the body for quite a while [often up to 3 months and even longer], so that is helpful for patients to know.
Given the current situation and the possibility of its effectiveness against COVID-19, it is ironic that we are actually trying to recruit older lupus patients who have had long-term stable disease while on hydroxychloroquine to a trial of stopping the drug to reduce the risk of developing the side effect of retinopathy. We want to see if patients can safely withdraw hydroxychloroquine without flaring, so we hope to not run into enrollment difficulties based on the current situation with COVID-19.
Q: How do you view the balance between having enough hydroxychloroquine for patients with lupus or other rheumatic diseases and its use in COVID-19 patients?
A: We want to reassure patients that hydroxychloroquine will be available, and there is no reason to hoard the drug or to worry excessively about being unable to obtain it. Efforts to increase production by Mylan, Teva, Sanofi, Novartis, and other manufacturers of hydroxychloroquine should really help out.
Q: Are there pharmacy restrictions on prescription amounts?
A: This is not universal at this time, but some institutions are cutting back and offering only 1-month supplies.
Colchicine COVID-19 trial underway
Dr. Pillinger, of NYU Langone Health, explored the COLCORONA study of colchicine as a treatment for people infected with COVID-19 and the worry that shortage concerns may arise for it, too.
Q: What is the general availability of colchicine and its susceptibility to shortage?
A: There are two major manufacturers of colchicine in the United States, Takeda and Hikma, who together manufacture the majority of the drug.
The greatest use of colchicine in the United States is for gout, which affects approximately 4 million Americans, but the drug is not used chronically, so a much smaller number of patients are using colchicine at any one time. Colchicine is also used for other inflammatory conditions, primarily calcium pyrophosphate crystal disease and familial Mediterranean fever (FMF is rare in the United States). Cardiologists also regularly prescribe colchicine in pericarditis for short-term use. Physicians may use it off label for other purposes, too.
Overall, the number of patients using colchicine is much larger than that for the use of hydroxychloroquine, for example, suggesting that the immediate risk of shortage could be lower. However, if individuals started using it off label, or prescribing inappropriately for the COVID-19 indication, the supply would rapidly run short.
Q: What other points are there to consider regarding the use of colchicine to treat COVID-19?
A: There is no evidence – zero – that colchicine has any benefit for COVID-19, not even case reports. There is some rationale that it might be beneficial, but that is exactly why the COLCORONA trial would be logical to try.
The COLCORONA trial is exactly the kind of trial that would be needed for assessing colchicine, and it is big enough and happening quickly enough to get an answer. But if people start to use colchicine off label, we may never know the truth.
While colchicine can be used safely in most people, it can be very problematic and requires an experienced doctor’s supervision. Overdoses can be fatal, and colchicine interacts with many drugs, all of which require dose adjustment and some of which must be stopped in order to use colchicine – it isn’t candy. Some of the other drugs being looked at for COVID-19 in fact may interact with colchicine.
Colchicine must also be dose adjusted for kidney disease, and, in some of the COVID-19 patients, kidney function changes rapidly. So again, its use would require expert supervision even if there were evidence for its utility.
The side effects of colchicine, if mis-dosed, can be very unpleasant, including nausea, vomiting, and diarrhea. Even at the apparent right dose, some people will get these side effects, so colchicine has to be something that works to make the risk/benefit ratio worth it.
Some preparations of colchicine are made combined with probenecid, a gout drug. This is even more problematic because probenecid can raise the level of drugs excreted by the kidney and could affect other treatments.
So in sum, what may be a good idea in theory can turn out to be a disastrous idea in practice, and here we have nothing but theory. This is not an agent to use randomly; the studies will be rushed out quickly and hopefully will give us the knowledge to know what to do.
Dr. Izmirly and Dr. Buyon said they have research grants with the National Institutes of Health to study hydroxychloroquine in patients with lupus and in anti–SSA/Ro-positive pregnant women with a previous child with congenital heart block. Dr. Pillinger reports that he has an investigator-initiated grant from Hikma to study colchicine in osteoarthritis.
This article was reformatted on 3/30/2020 for clarity.
Physicians and pharmacists are reporting shortages of hydroxychloroquine and chloroquine following President Trump’s promotion of the medications as potential COVID-19 treatments, leaving patients with rheumatic diseases wondering how it will impact their access.
The American Medical Association, the American Pharmacists Association, and the American Society of Health-System Pharmacists, issued a joint statement that strongly opposed prophylactic prescribing of these medications for COVID-19 or stockpiling them in anticipation of use for COVID-19. The concerns over shortages have also prompted the American College of Rheumatology, American Academy of Dermatology, Arthritis Foundation, and Lupus Foundation of America to send a joint statement to the Trump administration and the nation’s governors highlighting critical hydroxychloroquine access issues and asking policymakers to work together with health care providers and patient communities to ensure continued availability of these drugs.
Now
In a Q and A interview, NYU Langone Health rheumatology division director and Lupus Center director Jill P. Buyon, MD, and associate professor of rheumatology, Peter M. Izmirly, MD, noted that, while shortages have been reported across the United States because of large increases in off-label prescribing, many of the drugs’ manufacturers have committed to donating millions of doses and/or stepping up production to meet demand.
Later in this article, Michael H. Pillinger, MD, a rheumatologist and professor of medicine, biochemistry, and molecular pharmacology at NYU Langone Health, New York, answered questions about a new multicenter study called COLCORONA getting underway to test the anti-inflammatory drug colchicine. The answers in this Q&A have been edited for length and clarity.
Questions about hydroxychloroquine shortage
Q: What is the current situation with hydroxychloroquine in your practice?
A: We have been getting calls from our patients asking about getting refills for hydroxychloroquine. Our group has been calling local pharmacies asking about the availability of hydroxychloroquine, and we are compiling a list of pharmacies in New York with current availabilities to share with patients. We are somewhat limited by our electronic health record system, Epic, which can only send a prescription to one pharmacy, so that has placed some limitations on knowing where it is available. Some pharmacies have not had hydroxychloroquine available, while others have. We have also been encouraging patients to check online and look for mail-order possibilities for 90-day supplies.
Nearly all prescriptions are for generic hydroxychloroquine. Branded hydroxychloroquine (Plaquenil) is much more expensive, and we can run into obstacles with getting it approved by insurers, too.
Q: What are you telling patients who seek to refill their prescription or call with concerns? Is it feasible for patients to stop hydroxychloroquine or cut their dosage if necessary?
A: If someone’s been on hydroxychloroquine and has benefited from its use there’s no reason to come off it at this time, and given the possibility that it may have an effect on COVID-19, that is all the better. But we want to reassure patients that they can get the drug and that it is not difficult to manufacture.
Given the significantly higher risk of disease flare that was first described in lupus patients who discontinued hydroxychloroquine in the Canadian Hydroxychloroquine Study Group’s 1991 randomized, controlled trial, it is not advisable for patients to stop the drug.
Some patients do split their dosage day-to-day if they are taking less than 400 mg daily, such that someone taking 300 mg daily may take two 200-mg tablets one day and just one 200-mg tablet the next day, and so on. To avoid eye toxicity that can occur after years of taking the drug, hydroxychloroquine is generally prescribed based on weight at 5 mg/kg.
The drug also stays in the body for quite a while [often up to 3 months and even longer], so that is helpful for patients to know.
Given the current situation and the possibility of its effectiveness against COVID-19, it is ironic that we are actually trying to recruit older lupus patients who have had long-term stable disease while on hydroxychloroquine to a trial of stopping the drug to reduce the risk of developing the side effect of retinopathy. We want to see if patients can safely withdraw hydroxychloroquine without flaring, so we hope to not run into enrollment difficulties based on the current situation with COVID-19.
Q: How do you view the balance between having enough hydroxychloroquine for patients with lupus or other rheumatic diseases and its use in COVID-19 patients?
A: We want to reassure patients that hydroxychloroquine will be available, and there is no reason to hoard the drug or to worry excessively about being unable to obtain it. Efforts to increase production by Mylan, Teva, Sanofi, Novartis, and other manufacturers of hydroxychloroquine should really help out.
Q: Are there pharmacy restrictions on prescription amounts?
A: This is not universal at this time, but some institutions are cutting back and offering only 1-month supplies.
Colchicine COVID-19 trial underway
Dr. Pillinger, of NYU Langone Health, explored the COLCORONA study of colchicine as a treatment for people infected with COVID-19 and the worry that shortage concerns may arise for it, too.
Q: What is the general availability of colchicine and its susceptibility to shortage?
A: There are two major manufacturers of colchicine in the United States, Takeda and Hikma, who together manufacture the majority of the drug.
The greatest use of colchicine in the United States is for gout, which affects approximately 4 million Americans, but the drug is not used chronically, so a much smaller number of patients are using colchicine at any one time. Colchicine is also used for other inflammatory conditions, primarily calcium pyrophosphate crystal disease and familial Mediterranean fever (FMF is rare in the United States). Cardiologists also regularly prescribe colchicine in pericarditis for short-term use. Physicians may use it off label for other purposes, too.
Overall, the number of patients using colchicine is much larger than that for the use of hydroxychloroquine, for example, suggesting that the immediate risk of shortage could be lower. However, if individuals started using it off label, or prescribing inappropriately for the COVID-19 indication, the supply would rapidly run short.
Q: What other points are there to consider regarding the use of colchicine to treat COVID-19?
A: There is no evidence – zero – that colchicine has any benefit for COVID-19, not even case reports. There is some rationale that it might be beneficial, but that is exactly why the COLCORONA trial would be logical to try.
The COLCORONA trial is exactly the kind of trial that would be needed for assessing colchicine, and it is big enough and happening quickly enough to get an answer. But if people start to use colchicine off label, we may never know the truth.
While colchicine can be used safely in most people, it can be very problematic and requires an experienced doctor’s supervision. Overdoses can be fatal, and colchicine interacts with many drugs, all of which require dose adjustment and some of which must be stopped in order to use colchicine – it isn’t candy. Some of the other drugs being looked at for COVID-19 in fact may interact with colchicine.
Colchicine must also be dose adjusted for kidney disease, and, in some of the COVID-19 patients, kidney function changes rapidly. So again, its use would require expert supervision even if there were evidence for its utility.
The side effects of colchicine, if mis-dosed, can be very unpleasant, including nausea, vomiting, and diarrhea. Even at the apparent right dose, some people will get these side effects, so colchicine has to be something that works to make the risk/benefit ratio worth it.
Some preparations of colchicine are made combined with probenecid, a gout drug. This is even more problematic because probenecid can raise the level of drugs excreted by the kidney and could affect other treatments.
So in sum, what may be a good idea in theory can turn out to be a disastrous idea in practice, and here we have nothing but theory. This is not an agent to use randomly; the studies will be rushed out quickly and hopefully will give us the knowledge to know what to do.
Dr. Izmirly and Dr. Buyon said they have research grants with the National Institutes of Health to study hydroxychloroquine in patients with lupus and in anti–SSA/Ro-positive pregnant women with a previous child with congenital heart block. Dr. Pillinger reports that he has an investigator-initiated grant from Hikma to study colchicine in osteoarthritis.
This article was reformatted on 3/30/2020 for clarity.
Physicians pessimistic despite increased COVID-19 test kits
according to a survey.
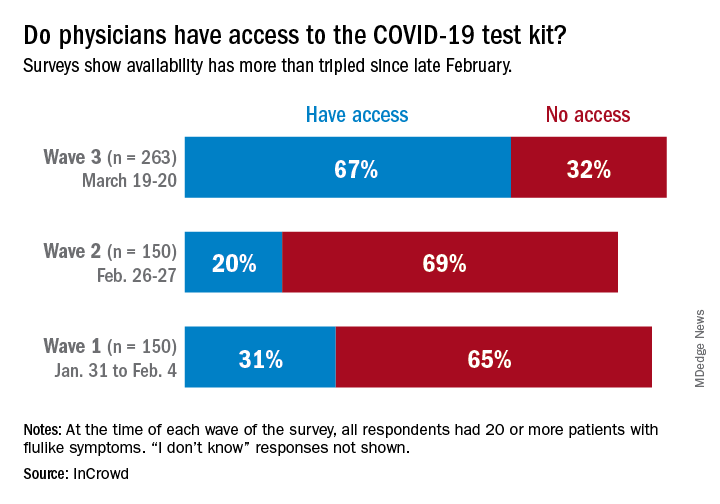
One positive finding from the physicians who participated in this survey March 19-20 was that the availability of COVID-19 test kits has more than doubled since late February.
Reported access to test kits went from 31% in the first wave of a series of surveys (Jan. 31–Feb. 4), down to 20% in the second (Feb. 26-27), and then jumped to 67% by the third wave (March 19-20), InCrowd reported March 26.
Views on several other COVID-related topics were negative among the majority of responding physicians – all of whom had or were currently treating 20 or more patients with flu-like symptoms at the time of the survey.
“Their frustrations and concerns about their ability to protect themselves while meeting upcoming patient care levels has increased significantly in the last 3 months,” Daniel S. Fitzgerald, CEO and president of InCrowd, said in a written statement.
In the third wave, 78% of respondents were “concerned for the safety of loved ones due to my exposure as a physician to COVID-19” and only 16% believed that their facility was “staffed adequately to treat the influx of patients anticipated in the next 30 days,” InCrowd said.
One primary care physician from California elaborated on the issue of safety equipment: “First, [the CDC] said we need N95 masks and other masks would not protect us. As those are running out then they said just use regular surgical masks. Now they are saying use bandannas and scarves! It’s like they don’t care about the safety of the people who will be treating the ill! We don’t want to bring it home to our families!”
“Overall, morale appears low, with few optimistic about the efficacy of public-private collaboration (21%), their own safety given current PPE [personal protective equipment] supply (13%), and the U.S.’s ability to ‘flatten the curve’ (12%),” InCrowd noted in the report.
The first two waves each had 150 respondents, but the number increased to 263 for wave 3, with similar proportions – about 50% emergency medicine or critical care specialists, 25% pediatricians, and 25% primary care physicians – in all three.
according to a survey.

One positive finding from the physicians who participated in this survey March 19-20 was that the availability of COVID-19 test kits has more than doubled since late February.
Reported access to test kits went from 31% in the first wave of a series of surveys (Jan. 31–Feb. 4), down to 20% in the second (Feb. 26-27), and then jumped to 67% by the third wave (March 19-20), InCrowd reported March 26.
Views on several other COVID-related topics were negative among the majority of responding physicians – all of whom had or were currently treating 20 or more patients with flu-like symptoms at the time of the survey.
“Their frustrations and concerns about their ability to protect themselves while meeting upcoming patient care levels has increased significantly in the last 3 months,” Daniel S. Fitzgerald, CEO and president of InCrowd, said in a written statement.
In the third wave, 78% of respondents were “concerned for the safety of loved ones due to my exposure as a physician to COVID-19” and only 16% believed that their facility was “staffed adequately to treat the influx of patients anticipated in the next 30 days,” InCrowd said.
One primary care physician from California elaborated on the issue of safety equipment: “First, [the CDC] said we need N95 masks and other masks would not protect us. As those are running out then they said just use regular surgical masks. Now they are saying use bandannas and scarves! It’s like they don’t care about the safety of the people who will be treating the ill! We don’t want to bring it home to our families!”
“Overall, morale appears low, with few optimistic about the efficacy of public-private collaboration (21%), their own safety given current PPE [personal protective equipment] supply (13%), and the U.S.’s ability to ‘flatten the curve’ (12%),” InCrowd noted in the report.
The first two waves each had 150 respondents, but the number increased to 263 for wave 3, with similar proportions – about 50% emergency medicine or critical care specialists, 25% pediatricians, and 25% primary care physicians – in all three.
according to a survey.

One positive finding from the physicians who participated in this survey March 19-20 was that the availability of COVID-19 test kits has more than doubled since late February.
Reported access to test kits went from 31% in the first wave of a series of surveys (Jan. 31–Feb. 4), down to 20% in the second (Feb. 26-27), and then jumped to 67% by the third wave (March 19-20), InCrowd reported March 26.
Views on several other COVID-related topics were negative among the majority of responding physicians – all of whom had or were currently treating 20 or more patients with flu-like symptoms at the time of the survey.
“Their frustrations and concerns about their ability to protect themselves while meeting upcoming patient care levels has increased significantly in the last 3 months,” Daniel S. Fitzgerald, CEO and president of InCrowd, said in a written statement.
In the third wave, 78% of respondents were “concerned for the safety of loved ones due to my exposure as a physician to COVID-19” and only 16% believed that their facility was “staffed adequately to treat the influx of patients anticipated in the next 30 days,” InCrowd said.
One primary care physician from California elaborated on the issue of safety equipment: “First, [the CDC] said we need N95 masks and other masks would not protect us. As those are running out then they said just use regular surgical masks. Now they are saying use bandannas and scarves! It’s like they don’t care about the safety of the people who will be treating the ill! We don’t want to bring it home to our families!”
“Overall, morale appears low, with few optimistic about the efficacy of public-private collaboration (21%), their own safety given current PPE [personal protective equipment] supply (13%), and the U.S.’s ability to ‘flatten the curve’ (12%),” InCrowd noted in the report.
The first two waves each had 150 respondents, but the number increased to 263 for wave 3, with similar proportions – about 50% emergency medicine or critical care specialists, 25% pediatricians, and 25% primary care physicians – in all three.