User login
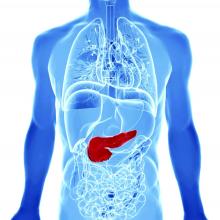
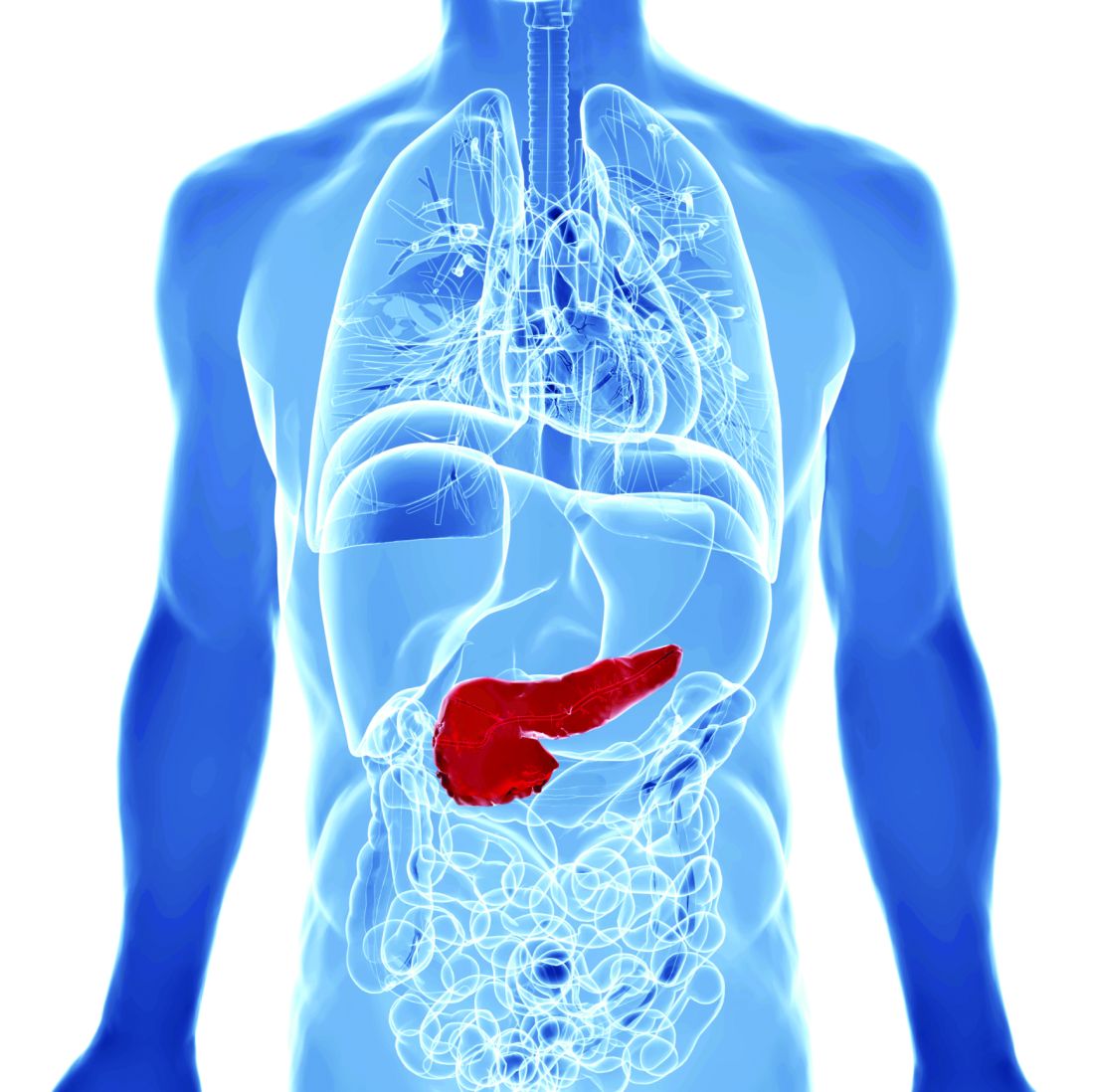
AGA Clinical Practice Update: Diagnosis of rumination syndrome
Additionally, promote diaphragmatic breathing to help manage the condition, advised authors of an expert review of clinical practice updates for rumination syndrome published in Clinical Gastroenterology and Hepatology.
“Patients, not unsurprisingly, typically use the word ‘vomiting’ to describe rumination events, and many patients are misdiagnosed as having refractory vomiting, gastroesophageal reflux disease, or gastroparesis,” Magnus Halland, MD, of the Mayo Clinic in Rochester, Minn., and colleagues wrote in the review. “A long delay in receiving a diagnosis is common and can lead to unnecessary testing, reduced quality of life, and even invasive procedures such as surgery or feeding tubes.”
Rumination syndrome differs from vomiting, the authors noted, because the retrograde flow of ingested gastric content does not have an acidic taste and may in fact taste like food or drink recently ingested. Rumination can occur without any preceding events, after a reflux episode or by the swallowing of air that causes gastric straining but typically happens within 1 hour to 2 hours after a meal. Patients can experience weight loss, dental erosions and caries, heartburn, nausea, bloating, diarrhea, abdominal pain, abdominal discomfort, and belching, among other symptoms, in the presence of rumination syndrome, the authors said.
Dr. Halland and his colleagues provided seven best practice recommendations for rumination syndrome in their updates, which include:
- Patients who show symptoms of consistent postprandial regurgitation, often misdiagnosed with refractory gastroesophageal reflux or vomiting, should be considered for rumination syndrome.
- Patients who have dysphagia, nausea, nocturnal regurgitation, or gastric symptoms outside of meals are less likely to have rumination syndrome, but those symptoms do not exclude the condition.
- Rome IV criteria are advised to diagnose rumination syndrome after medical work-up, which includes “persistent or recurrent regurgitation of recently ingested food into the mouth with subsequent spitting or remastication and swallowing” not preceded by retching where patients fulfill these symptom criteria for 3 months with a minimum of 6 months of symptoms before diagnosis.
- Patients should receive first-line therapy for rumination syndrome consisting of diaphragmatic breathing with or without biofeedback.
- Patients should be referred to a speech therapist, gastroenterologist, psychologist, or other knowledgeable health practitioners to learn effective diaphragmatic breathing.
- Current limitations in the diagnosis of rumination syndrome include need for expertise and lack of standardized protocols, but “testing for rumination syndrome with postprandial high-resolution esophageal impedance manometry can be used to support the diagnosis.”
- Bacloflen (10 mg) taken three times daily is a “reasonable next step” for patients who do not respond to treatment.
The authors acknowledged that many questions, such as the pathophysiology and initiating factors of rumination syndrome, are unknown and noted future studies are needed to address epidemiology, develop validated tools for measuring symptoms, and study diaphragmatic breathing’s effect on reducing symptoms of rumination syndrome as well as the condition’s impact on quality of life.
“Indeed, the basic question of how subconsciously one can learn to regurgitate still needs to be answered,” Dr. Halland and his colleagues wrote.
The authors report no relevant conflicts of interest.
SOURCE: Halland M et al. Clin Gastroenterol Hepatol. 2018 Jun 11. doi: 10.1016/j.cgh.2018.05.049.
Additionally, promote diaphragmatic breathing to help manage the condition, advised authors of an expert review of clinical practice updates for rumination syndrome published in Clinical Gastroenterology and Hepatology.
“Patients, not unsurprisingly, typically use the word ‘vomiting’ to describe rumination events, and many patients are misdiagnosed as having refractory vomiting, gastroesophageal reflux disease, or gastroparesis,” Magnus Halland, MD, of the Mayo Clinic in Rochester, Minn., and colleagues wrote in the review. “A long delay in receiving a diagnosis is common and can lead to unnecessary testing, reduced quality of life, and even invasive procedures such as surgery or feeding tubes.”
Rumination syndrome differs from vomiting, the authors noted, because the retrograde flow of ingested gastric content does not have an acidic taste and may in fact taste like food or drink recently ingested. Rumination can occur without any preceding events, after a reflux episode or by the swallowing of air that causes gastric straining but typically happens within 1 hour to 2 hours after a meal. Patients can experience weight loss, dental erosions and caries, heartburn, nausea, bloating, diarrhea, abdominal pain, abdominal discomfort, and belching, among other symptoms, in the presence of rumination syndrome, the authors said.
Dr. Halland and his colleagues provided seven best practice recommendations for rumination syndrome in their updates, which include:
- Patients who show symptoms of consistent postprandial regurgitation, often misdiagnosed with refractory gastroesophageal reflux or vomiting, should be considered for rumination syndrome.
- Patients who have dysphagia, nausea, nocturnal regurgitation, or gastric symptoms outside of meals are less likely to have rumination syndrome, but those symptoms do not exclude the condition.
- Rome IV criteria are advised to diagnose rumination syndrome after medical work-up, which includes “persistent or recurrent regurgitation of recently ingested food into the mouth with subsequent spitting or remastication and swallowing” not preceded by retching where patients fulfill these symptom criteria for 3 months with a minimum of 6 months of symptoms before diagnosis.
- Patients should receive first-line therapy for rumination syndrome consisting of diaphragmatic breathing with or without biofeedback.
- Patients should be referred to a speech therapist, gastroenterologist, psychologist, or other knowledgeable health practitioners to learn effective diaphragmatic breathing.
- Current limitations in the diagnosis of rumination syndrome include need for expertise and lack of standardized protocols, but “testing for rumination syndrome with postprandial high-resolution esophageal impedance manometry can be used to support the diagnosis.”
- Bacloflen (10 mg) taken three times daily is a “reasonable next step” for patients who do not respond to treatment.
The authors acknowledged that many questions, such as the pathophysiology and initiating factors of rumination syndrome, are unknown and noted future studies are needed to address epidemiology, develop validated tools for measuring symptoms, and study diaphragmatic breathing’s effect on reducing symptoms of rumination syndrome as well as the condition’s impact on quality of life.
“Indeed, the basic question of how subconsciously one can learn to regurgitate still needs to be answered,” Dr. Halland and his colleagues wrote.
The authors report no relevant conflicts of interest.
SOURCE: Halland M et al. Clin Gastroenterol Hepatol. 2018 Jun 11. doi: 10.1016/j.cgh.2018.05.049.
Additionally, promote diaphragmatic breathing to help manage the condition, advised authors of an expert review of clinical practice updates for rumination syndrome published in Clinical Gastroenterology and Hepatology.
“Patients, not unsurprisingly, typically use the word ‘vomiting’ to describe rumination events, and many patients are misdiagnosed as having refractory vomiting, gastroesophageal reflux disease, or gastroparesis,” Magnus Halland, MD, of the Mayo Clinic in Rochester, Minn., and colleagues wrote in the review. “A long delay in receiving a diagnosis is common and can lead to unnecessary testing, reduced quality of life, and even invasive procedures such as surgery or feeding tubes.”
Rumination syndrome differs from vomiting, the authors noted, because the retrograde flow of ingested gastric content does not have an acidic taste and may in fact taste like food or drink recently ingested. Rumination can occur without any preceding events, after a reflux episode or by the swallowing of air that causes gastric straining but typically happens within 1 hour to 2 hours after a meal. Patients can experience weight loss, dental erosions and caries, heartburn, nausea, bloating, diarrhea, abdominal pain, abdominal discomfort, and belching, among other symptoms, in the presence of rumination syndrome, the authors said.
Dr. Halland and his colleagues provided seven best practice recommendations for rumination syndrome in their updates, which include:
- Patients who show symptoms of consistent postprandial regurgitation, often misdiagnosed with refractory gastroesophageal reflux or vomiting, should be considered for rumination syndrome.
- Patients who have dysphagia, nausea, nocturnal regurgitation, or gastric symptoms outside of meals are less likely to have rumination syndrome, but those symptoms do not exclude the condition.
- Rome IV criteria are advised to diagnose rumination syndrome after medical work-up, which includes “persistent or recurrent regurgitation of recently ingested food into the mouth with subsequent spitting or remastication and swallowing” not preceded by retching where patients fulfill these symptom criteria for 3 months with a minimum of 6 months of symptoms before diagnosis.
- Patients should receive first-line therapy for rumination syndrome consisting of diaphragmatic breathing with or without biofeedback.
- Patients should be referred to a speech therapist, gastroenterologist, psychologist, or other knowledgeable health practitioners to learn effective diaphragmatic breathing.
- Current limitations in the diagnosis of rumination syndrome include need for expertise and lack of standardized protocols, but “testing for rumination syndrome with postprandial high-resolution esophageal impedance manometry can be used to support the diagnosis.”
- Bacloflen (10 mg) taken three times daily is a “reasonable next step” for patients who do not respond to treatment.
The authors acknowledged that many questions, such as the pathophysiology and initiating factors of rumination syndrome, are unknown and noted future studies are needed to address epidemiology, develop validated tools for measuring symptoms, and study diaphragmatic breathing’s effect on reducing symptoms of rumination syndrome as well as the condition’s impact on quality of life.
“Indeed, the basic question of how subconsciously one can learn to regurgitate still needs to be answered,” Dr. Halland and his colleagues wrote.
The authors report no relevant conflicts of interest.
SOURCE: Halland M et al. Clin Gastroenterol Hepatol. 2018 Jun 11. doi: 10.1016/j.cgh.2018.05.049.
FROM CLINICAL GASTROENTEROLOGY AND HEPATOLOGY
Commentary: Composite risk, not age, is key for timing first colorectal cancer screening
The American Cancer Society’s recent recommendation to lower the age of first screening for colorectal cancer to 45 years does not reflect clear knowledge of risks versus benefits, experts wrote in a recent commentary.
“In the big picture, [the question of whether to start screening at 45 versus 50 years] seems relatively unimportant compared with using individual patient risk for advanced neoplasia in practical, feasible models” that are geared toward adherence, efficiency, and cost-efficacy, wrote Thomas F. Imperiale, MD, of Indiana University, Indianapolis, and his associates. The commentary is in the October issue of Clinical Gastroenterology and Hepatology.
Tailoring age of first screening on an individual level, based on other risk factors and patient preferences, might improve uptake and benefit-risk ratios, balance, they argued.
Rates of colorectal cancer in persons under age 50 years rose by about 22% between 2000 and 2013. However, estimates for the most recent birth cohorts have wide confidence intervals, “indicating imprecision and uncertainty that this trend will continue,” the experts wrote. Furthermore, the absolute risk of colorectal cancer among individuals younger than 50 years has risen only slightly, from 5.9 cases per 100,000 population to 7.2 cases per 100,000 population. “[This] small increase in incidence may represent a true increase or could be due to increased use of colonoscopy in general, and specifically, for diagnosis or high-risk screening of first-degree relatives of persons with colorectal cancer,” the experts wrote.
Implementing the new recommendation could detect earlier-stage (curable) colorectal cancer “in a youthful and productive age group that may be sandwiched between raising children and caring for aging parents,” they continued. Earlier detection could reduce mortality and reduce the costs of treating a disease that often exceeds $100,000 per person annually.
However, the recommendation was based on a modeling study that assumed 100% adherence. In reality, uptake among 45- to 49-year-olds might be 15%-20%, and “who actually shows up for screening could make or break this recommendation,” the experts said. If younger individuals who underwent screening tended to have few risk factors for colorectal cancer, then the new recommendation would lead to many false positives and unnecessary colonoscopies, with the associated fallout of emotional harm and wasted health care resources, they added.
Population-level studies have identified age as the strongest predictor of colorectal cancer, but age “does not perform as well” at patient level, the experts said. They emphasized the role of other risk factors, such as male sex, having a first-degree relative with colorectal cancer, high body mass index, metabolic syndrome, cigarette smoking, diet, adherence to screening, and use of aspirin, nonsteroidal anti-inflammatory drugs, and hormone therapy. “The goal for providers and health systems is to determine whether and how to change screening practice and policy, and how to incorporate this new recommendation into practice, a necessarily complex process that requires knowing patient risk, patient preferences, and the long-term balance of benefits and burdens,” they concluded (Clin Gastroenterol Hepatol. 2018 Aug 13. doi: 10.1016/j.cgh.2018.08.023).
Dr. Imperiale and coauthor Charles J. Kahi, MD, MS, had no disclosures. Coauthor Douglas K. Rex, MD, disclosed ties to Aries Pharmaceutical, Cosmo Pharmaceuticals, Boston Scientific, Sebela, Medtronic, EndoAid Ltd, Olympus, Paion, Braintree, and Medivators. He also chairs the U.S. Multi-Society Task Force on Colorectal Cancer.
* This story was updated on 10/16/2018.
The American Cancer Society’s recent recommendation to lower the age of first screening for colorectal cancer to 45 years does not reflect clear knowledge of risks versus benefits, experts wrote in a recent commentary.
“In the big picture, [the question of whether to start screening at 45 versus 50 years] seems relatively unimportant compared with using individual patient risk for advanced neoplasia in practical, feasible models” that are geared toward adherence, efficiency, and cost-efficacy, wrote Thomas F. Imperiale, MD, of Indiana University, Indianapolis, and his associates. The commentary is in the October issue of Clinical Gastroenterology and Hepatology.
Tailoring age of first screening on an individual level, based on other risk factors and patient preferences, might improve uptake and benefit-risk ratios, balance, they argued.
Rates of colorectal cancer in persons under age 50 years rose by about 22% between 2000 and 2013. However, estimates for the most recent birth cohorts have wide confidence intervals, “indicating imprecision and uncertainty that this trend will continue,” the experts wrote. Furthermore, the absolute risk of colorectal cancer among individuals younger than 50 years has risen only slightly, from 5.9 cases per 100,000 population to 7.2 cases per 100,000 population. “[This] small increase in incidence may represent a true increase or could be due to increased use of colonoscopy in general, and specifically, for diagnosis or high-risk screening of first-degree relatives of persons with colorectal cancer,” the experts wrote.
Implementing the new recommendation could detect earlier-stage (curable) colorectal cancer “in a youthful and productive age group that may be sandwiched between raising children and caring for aging parents,” they continued. Earlier detection could reduce mortality and reduce the costs of treating a disease that often exceeds $100,000 per person annually.
However, the recommendation was based on a modeling study that assumed 100% adherence. In reality, uptake among 45- to 49-year-olds might be 15%-20%, and “who actually shows up for screening could make or break this recommendation,” the experts said. If younger individuals who underwent screening tended to have few risk factors for colorectal cancer, then the new recommendation would lead to many false positives and unnecessary colonoscopies, with the associated fallout of emotional harm and wasted health care resources, they added.
Population-level studies have identified age as the strongest predictor of colorectal cancer, but age “does not perform as well” at patient level, the experts said. They emphasized the role of other risk factors, such as male sex, having a first-degree relative with colorectal cancer, high body mass index, metabolic syndrome, cigarette smoking, diet, adherence to screening, and use of aspirin, nonsteroidal anti-inflammatory drugs, and hormone therapy. “The goal for providers and health systems is to determine whether and how to change screening practice and policy, and how to incorporate this new recommendation into practice, a necessarily complex process that requires knowing patient risk, patient preferences, and the long-term balance of benefits and burdens,” they concluded (Clin Gastroenterol Hepatol. 2018 Aug 13. doi: 10.1016/j.cgh.2018.08.023).
Dr. Imperiale and coauthor Charles J. Kahi, MD, MS, had no disclosures. Coauthor Douglas K. Rex, MD, disclosed ties to Aries Pharmaceutical, Cosmo Pharmaceuticals, Boston Scientific, Sebela, Medtronic, EndoAid Ltd, Olympus, Paion, Braintree, and Medivators. He also chairs the U.S. Multi-Society Task Force on Colorectal Cancer.
* This story was updated on 10/16/2018.
The American Cancer Society’s recent recommendation to lower the age of first screening for colorectal cancer to 45 years does not reflect clear knowledge of risks versus benefits, experts wrote in a recent commentary.
“In the big picture, [the question of whether to start screening at 45 versus 50 years] seems relatively unimportant compared with using individual patient risk for advanced neoplasia in practical, feasible models” that are geared toward adherence, efficiency, and cost-efficacy, wrote Thomas F. Imperiale, MD, of Indiana University, Indianapolis, and his associates. The commentary is in the October issue of Clinical Gastroenterology and Hepatology.
Tailoring age of first screening on an individual level, based on other risk factors and patient preferences, might improve uptake and benefit-risk ratios, balance, they argued.
Rates of colorectal cancer in persons under age 50 years rose by about 22% between 2000 and 2013. However, estimates for the most recent birth cohorts have wide confidence intervals, “indicating imprecision and uncertainty that this trend will continue,” the experts wrote. Furthermore, the absolute risk of colorectal cancer among individuals younger than 50 years has risen only slightly, from 5.9 cases per 100,000 population to 7.2 cases per 100,000 population. “[This] small increase in incidence may represent a true increase or could be due to increased use of colonoscopy in general, and specifically, for diagnosis or high-risk screening of first-degree relatives of persons with colorectal cancer,” the experts wrote.
Implementing the new recommendation could detect earlier-stage (curable) colorectal cancer “in a youthful and productive age group that may be sandwiched between raising children and caring for aging parents,” they continued. Earlier detection could reduce mortality and reduce the costs of treating a disease that often exceeds $100,000 per person annually.
However, the recommendation was based on a modeling study that assumed 100% adherence. In reality, uptake among 45- to 49-year-olds might be 15%-20%, and “who actually shows up for screening could make or break this recommendation,” the experts said. If younger individuals who underwent screening tended to have few risk factors for colorectal cancer, then the new recommendation would lead to many false positives and unnecessary colonoscopies, with the associated fallout of emotional harm and wasted health care resources, they added.
Population-level studies have identified age as the strongest predictor of colorectal cancer, but age “does not perform as well” at patient level, the experts said. They emphasized the role of other risk factors, such as male sex, having a first-degree relative with colorectal cancer, high body mass index, metabolic syndrome, cigarette smoking, diet, adherence to screening, and use of aspirin, nonsteroidal anti-inflammatory drugs, and hormone therapy. “The goal for providers and health systems is to determine whether and how to change screening practice and policy, and how to incorporate this new recommendation into practice, a necessarily complex process that requires knowing patient risk, patient preferences, and the long-term balance of benefits and burdens,” they concluded (Clin Gastroenterol Hepatol. 2018 Aug 13. doi: 10.1016/j.cgh.2018.08.023).
Dr. Imperiale and coauthor Charles J. Kahi, MD, MS, had no disclosures. Coauthor Douglas K. Rex, MD, disclosed ties to Aries Pharmaceutical, Cosmo Pharmaceuticals, Boston Scientific, Sebela, Medtronic, EndoAid Ltd, Olympus, Paion, Braintree, and Medivators. He also chairs the U.S. Multi-Society Task Force on Colorectal Cancer.
* This story was updated on 10/16/2018.
FROM CLINICAL GASTROENTEROLOGY AND HEPATOLOGY
Proinflammatory diet linked to colorectal cancer testing positive for Fusobacterium nucleatum
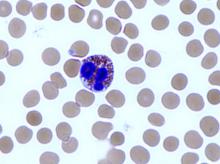
Diets promoting colonic inflammation were associated with a greater risk of colorectal carcinomas containing Fusobacterium nucleatum bacteria, according to a report in the October issue of Clinical Gastroenterology and Hepatology.
Courtesy American Gastroenterological Association
Proinflammatory diets were not linked to heightened risk for colon cancers without these bacteria, reported Li Liu, MD, PhD, of Dana-Farber Cancer Institute and Harvard Medical School, Boston. “These findings indicate that diet-induced intestinal inflammation alters the gut microbiome to contribute to colorectal carcinogenesis,” they wrote. “Nutritional interventions might be used in precision medicine and cancer prevention.”
Intestinal inflammation, a risk factor for colorectal cancer, is associated with high levels of circulating interleukin 6, C-reactive protein, and tumor necrosis factor–receptor superfamily member 1B. Colonic inflammation impairs the mucosal barrier and alters immune cell responses, which affects the composition of colonic microbiota. Among these, F. nucleatum is known to potentiate colorectal tumors and is associated with proximal tumor location, other tumor features, and cancer progression and chemoresistance.
For the study, the investigators examined self-reported data from more than 124,000 individuals followed for 28 years as part of the Nurses’ Health Study and the Health Professionals Follow-Up Study. They calculated average dietary patterns based on the empiric dietary inflammatory pattern (EDIP) score, which sums weighted intake scores for 18 foods (such as red and processed meat, coffee, tea, and leafy green or dark yellow vegetables) that are known to affect plasma levels of interleukin 6, C-reactive protein, tumor necrosis factor–receptor superfamily member 1B, and tumor necrosis factor alpha–receptor 2. A higher EDIP score denotes a more inflammatory diet.
During the 28-year follow-up period, 951 individuals developed colorectal carcinomas that were tested with a polymerase chain reaction assay for F. nucleatum DNA. A total of 115 tumors tested positive for F. nucleatum. After the researchers controlled for potential confounders, individuals whose EDIP scores were in the highest tertile were significantly more likely to develop F. nucleatum–positive colorectal cancer than were those who scored in the lowest tertile (adjusted hazard ratio, 1.63; 95% confidence interval, 1.03 to 2.58; P = .03). This differential association “appeared to be stronger in proximal colon cancer than in distal colon and rectal cancer,” the researchers said.
More than 90% of individuals in this study were non-Hispanic white, the researchers noted. Tumor tissue was not available from all cases of colorectal cancer and a fairly small number of cases tested positive for tumor F. nucleatum. Nonetheless, the findings suggest that an inflammatory diet could help amplify gut microbiota involved in tumorigenesis, they said. Pending confirmatory studies, they recommended an anti-inflammatory diet with high intake of green leafy vegetables, dark yellow vegetables, coffee, and tea, and with low intake of red meat, processed meat, refined grain, and sugary beverages. They also recommended studying whether F. nucleatum tumor or stool tests could help personalize dietary interventions.
Funders included the National Institutes of Health, Dana Farber Harvard Cancer, Project P. Fund for Colorectal Cancer Research, Friends of the Dana-Farber Cancer Institute, Bennett Family Fund, the Entertainment Industry Foundation, and American Association for Cancer Research, National Natural Science Foundation of China, Chinese Scholarship Council, Huazhong University of Science and Technology, and others. Dr. Liu had no disclosures. One coinvestigator disclosed ties to Genentech/Roche, Lilly, Sanofi, Bayer, and several other biomedical companies.
SOURCE: Liu L et al. Clin Gastroenterol Hepatol. 2018 Apr 24. doi: 10.1016/j.cgh.2018.04.030.
The underlying reasons colorectal cancer (CRC) develops are unknown, but they likely include a complex interaction between genetics and environmental exposures. Recent studies have highlighted important links between diet, the intestinal microbiota, and CRC development and progression.
Liu et al. used the Nurses’ Health Study and Health Professionals Follow-Up Study cohorts to extend our understanding of the relationship between diet, the intestinal microbiota, and CRC. They utilized validated food frequency questionnaires obtained every 4 years and formalin-fixed paraffin embedded CRC tissue samples collected from 951 individuals. They calculated an empiric dietary inflammatory pattern (EDIP) score, which correlates components of the diet with plasma inflammatory markers. After adjusting for confounders, they found high EDIP scores were significantly associated with Fusobacterium nucleatum–positive CRC, but not with F. nucleatum–negative CRC. In addition, they demonstrated this association was stronger for proximal compared with distal CRC. Their findings suggest an inflammatory diet may interact with the intestinal microbiota to promote the development of CRC and they provide a preliminary recommendation to minimize intake of potentially harmful foods (i.e., red meat, processed meat, refined grains, etc.). Despite the intriguing results, the authors do recognize limitations including the small number of cases with F. nucleatum present (n = 115) and the homogeneous cohort (90% non-Hispanic whites), which may limit generalizability.
As clinicians, we should continue strongly advocating for CRC screening and, based on these findings, may consider dietary recommendations to reduce intake of potentially harmful foods. Further research will be needed to confirm these findings in additional cohorts and to clarify the molecular interactions between dietary components, intestinal microbiota, and development of CRC.
Rajesh R. Shah, MD, is assistant professor of gastroenterology, department of internal medicine, Baylor College of Medicine, Houston. He has no conflicts of interest.
The underlying reasons colorectal cancer (CRC) develops are unknown, but they likely include a complex interaction between genetics and environmental exposures. Recent studies have highlighted important links between diet, the intestinal microbiota, and CRC development and progression.
Liu et al. used the Nurses’ Health Study and Health Professionals Follow-Up Study cohorts to extend our understanding of the relationship between diet, the intestinal microbiota, and CRC. They utilized validated food frequency questionnaires obtained every 4 years and formalin-fixed paraffin embedded CRC tissue samples collected from 951 individuals. They calculated an empiric dietary inflammatory pattern (EDIP) score, which correlates components of the diet with plasma inflammatory markers. After adjusting for confounders, they found high EDIP scores were significantly associated with Fusobacterium nucleatum–positive CRC, but not with F. nucleatum–negative CRC. In addition, they demonstrated this association was stronger for proximal compared with distal CRC. Their findings suggest an inflammatory diet may interact with the intestinal microbiota to promote the development of CRC and they provide a preliminary recommendation to minimize intake of potentially harmful foods (i.e., red meat, processed meat, refined grains, etc.). Despite the intriguing results, the authors do recognize limitations including the small number of cases with F. nucleatum present (n = 115) and the homogeneous cohort (90% non-Hispanic whites), which may limit generalizability.
As clinicians, we should continue strongly advocating for CRC screening and, based on these findings, may consider dietary recommendations to reduce intake of potentially harmful foods. Further research will be needed to confirm these findings in additional cohorts and to clarify the molecular interactions between dietary components, intestinal microbiota, and development of CRC.
Rajesh R. Shah, MD, is assistant professor of gastroenterology, department of internal medicine, Baylor College of Medicine, Houston. He has no conflicts of interest.
The underlying reasons colorectal cancer (CRC) develops are unknown, but they likely include a complex interaction between genetics and environmental exposures. Recent studies have highlighted important links between diet, the intestinal microbiota, and CRC development and progression.
Liu et al. used the Nurses’ Health Study and Health Professionals Follow-Up Study cohorts to extend our understanding of the relationship between diet, the intestinal microbiota, and CRC. They utilized validated food frequency questionnaires obtained every 4 years and formalin-fixed paraffin embedded CRC tissue samples collected from 951 individuals. They calculated an empiric dietary inflammatory pattern (EDIP) score, which correlates components of the diet with plasma inflammatory markers. After adjusting for confounders, they found high EDIP scores were significantly associated with Fusobacterium nucleatum–positive CRC, but not with F. nucleatum–negative CRC. In addition, they demonstrated this association was stronger for proximal compared with distal CRC. Their findings suggest an inflammatory diet may interact with the intestinal microbiota to promote the development of CRC and they provide a preliminary recommendation to minimize intake of potentially harmful foods (i.e., red meat, processed meat, refined grains, etc.). Despite the intriguing results, the authors do recognize limitations including the small number of cases with F. nucleatum present (n = 115) and the homogeneous cohort (90% non-Hispanic whites), which may limit generalizability.
As clinicians, we should continue strongly advocating for CRC screening and, based on these findings, may consider dietary recommendations to reduce intake of potentially harmful foods. Further research will be needed to confirm these findings in additional cohorts and to clarify the molecular interactions between dietary components, intestinal microbiota, and development of CRC.
Rajesh R. Shah, MD, is assistant professor of gastroenterology, department of internal medicine, Baylor College of Medicine, Houston. He has no conflicts of interest.
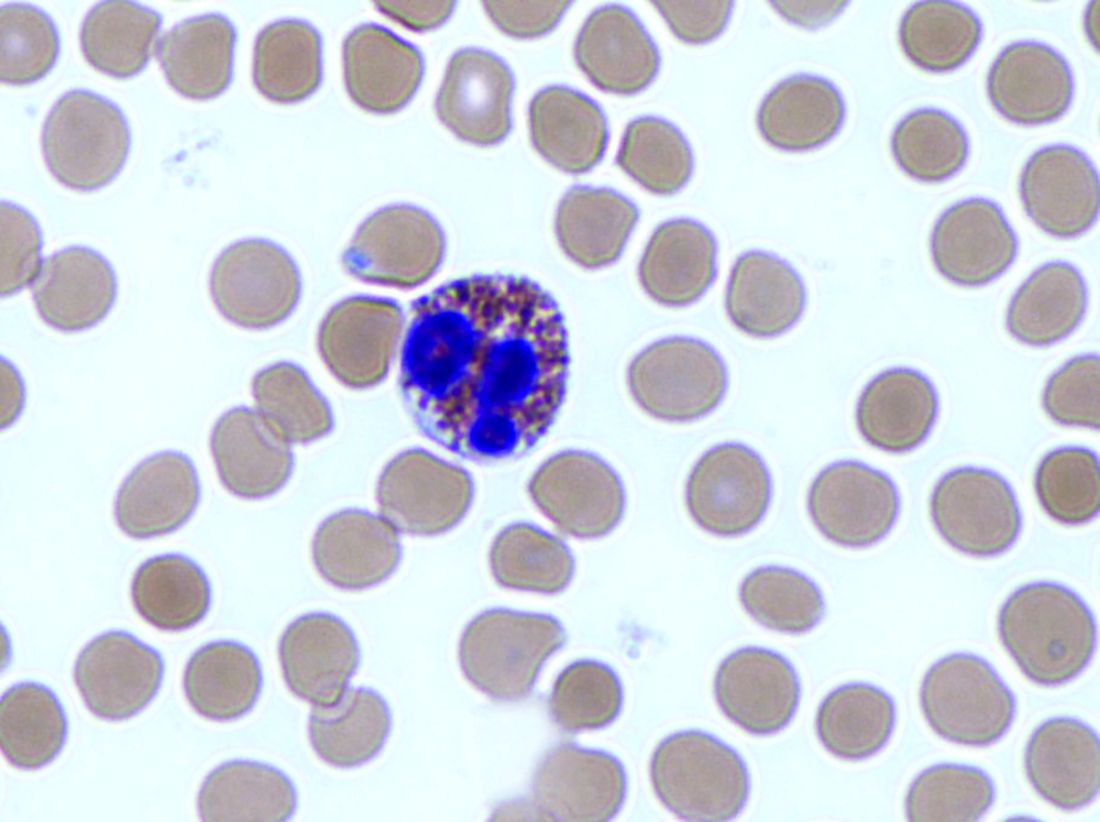
Diets promoting colonic inflammation were associated with a greater risk of colorectal carcinomas containing Fusobacterium nucleatum bacteria, according to a report in the October issue of Clinical Gastroenterology and Hepatology.
Courtesy American Gastroenterological Association
Proinflammatory diets were not linked to heightened risk for colon cancers without these bacteria, reported Li Liu, MD, PhD, of Dana-Farber Cancer Institute and Harvard Medical School, Boston. “These findings indicate that diet-induced intestinal inflammation alters the gut microbiome to contribute to colorectal carcinogenesis,” they wrote. “Nutritional interventions might be used in precision medicine and cancer prevention.”
Intestinal inflammation, a risk factor for colorectal cancer, is associated with high levels of circulating interleukin 6, C-reactive protein, and tumor necrosis factor–receptor superfamily member 1B. Colonic inflammation impairs the mucosal barrier and alters immune cell responses, which affects the composition of colonic microbiota. Among these, F. nucleatum is known to potentiate colorectal tumors and is associated with proximal tumor location, other tumor features, and cancer progression and chemoresistance.
For the study, the investigators examined self-reported data from more than 124,000 individuals followed for 28 years as part of the Nurses’ Health Study and the Health Professionals Follow-Up Study. They calculated average dietary patterns based on the empiric dietary inflammatory pattern (EDIP) score, which sums weighted intake scores for 18 foods (such as red and processed meat, coffee, tea, and leafy green or dark yellow vegetables) that are known to affect plasma levels of interleukin 6, C-reactive protein, tumor necrosis factor–receptor superfamily member 1B, and tumor necrosis factor alpha–receptor 2. A higher EDIP score denotes a more inflammatory diet.
During the 28-year follow-up period, 951 individuals developed colorectal carcinomas that were tested with a polymerase chain reaction assay for F. nucleatum DNA. A total of 115 tumors tested positive for F. nucleatum. After the researchers controlled for potential confounders, individuals whose EDIP scores were in the highest tertile were significantly more likely to develop F. nucleatum–positive colorectal cancer than were those who scored in the lowest tertile (adjusted hazard ratio, 1.63; 95% confidence interval, 1.03 to 2.58; P = .03). This differential association “appeared to be stronger in proximal colon cancer than in distal colon and rectal cancer,” the researchers said.
More than 90% of individuals in this study were non-Hispanic white, the researchers noted. Tumor tissue was not available from all cases of colorectal cancer and a fairly small number of cases tested positive for tumor F. nucleatum. Nonetheless, the findings suggest that an inflammatory diet could help amplify gut microbiota involved in tumorigenesis, they said. Pending confirmatory studies, they recommended an anti-inflammatory diet with high intake of green leafy vegetables, dark yellow vegetables, coffee, and tea, and with low intake of red meat, processed meat, refined grain, and sugary beverages. They also recommended studying whether F. nucleatum tumor or stool tests could help personalize dietary interventions.
Funders included the National Institutes of Health, Dana Farber Harvard Cancer, Project P. Fund for Colorectal Cancer Research, Friends of the Dana-Farber Cancer Institute, Bennett Family Fund, the Entertainment Industry Foundation, and American Association for Cancer Research, National Natural Science Foundation of China, Chinese Scholarship Council, Huazhong University of Science and Technology, and others. Dr. Liu had no disclosures. One coinvestigator disclosed ties to Genentech/Roche, Lilly, Sanofi, Bayer, and several other biomedical companies.
SOURCE: Liu L et al. Clin Gastroenterol Hepatol. 2018 Apr 24. doi: 10.1016/j.cgh.2018.04.030.
Diets promoting colonic inflammation were associated with a greater risk of colorectal carcinomas containing Fusobacterium nucleatum bacteria, according to a report in the October issue of Clinical Gastroenterology and Hepatology.
Courtesy American Gastroenterological Association
Proinflammatory diets were not linked to heightened risk for colon cancers without these bacteria, reported Li Liu, MD, PhD, of Dana-Farber Cancer Institute and Harvard Medical School, Boston. “These findings indicate that diet-induced intestinal inflammation alters the gut microbiome to contribute to colorectal carcinogenesis,” they wrote. “Nutritional interventions might be used in precision medicine and cancer prevention.”
Intestinal inflammation, a risk factor for colorectal cancer, is associated with high levels of circulating interleukin 6, C-reactive protein, and tumor necrosis factor–receptor superfamily member 1B. Colonic inflammation impairs the mucosal barrier and alters immune cell responses, which affects the composition of colonic microbiota. Among these, F. nucleatum is known to potentiate colorectal tumors and is associated with proximal tumor location, other tumor features, and cancer progression and chemoresistance.
For the study, the investigators examined self-reported data from more than 124,000 individuals followed for 28 years as part of the Nurses’ Health Study and the Health Professionals Follow-Up Study. They calculated average dietary patterns based on the empiric dietary inflammatory pattern (EDIP) score, which sums weighted intake scores for 18 foods (such as red and processed meat, coffee, tea, and leafy green or dark yellow vegetables) that are known to affect plasma levels of interleukin 6, C-reactive protein, tumor necrosis factor–receptor superfamily member 1B, and tumor necrosis factor alpha–receptor 2. A higher EDIP score denotes a more inflammatory diet.
During the 28-year follow-up period, 951 individuals developed colorectal carcinomas that were tested with a polymerase chain reaction assay for F. nucleatum DNA. A total of 115 tumors tested positive for F. nucleatum. After the researchers controlled for potential confounders, individuals whose EDIP scores were in the highest tertile were significantly more likely to develop F. nucleatum–positive colorectal cancer than were those who scored in the lowest tertile (adjusted hazard ratio, 1.63; 95% confidence interval, 1.03 to 2.58; P = .03). This differential association “appeared to be stronger in proximal colon cancer than in distal colon and rectal cancer,” the researchers said.
More than 90% of individuals in this study were non-Hispanic white, the researchers noted. Tumor tissue was not available from all cases of colorectal cancer and a fairly small number of cases tested positive for tumor F. nucleatum. Nonetheless, the findings suggest that an inflammatory diet could help amplify gut microbiota involved in tumorigenesis, they said. Pending confirmatory studies, they recommended an anti-inflammatory diet with high intake of green leafy vegetables, dark yellow vegetables, coffee, and tea, and with low intake of red meat, processed meat, refined grain, and sugary beverages. They also recommended studying whether F. nucleatum tumor or stool tests could help personalize dietary interventions.
Funders included the National Institutes of Health, Dana Farber Harvard Cancer, Project P. Fund for Colorectal Cancer Research, Friends of the Dana-Farber Cancer Institute, Bennett Family Fund, the Entertainment Industry Foundation, and American Association for Cancer Research, National Natural Science Foundation of China, Chinese Scholarship Council, Huazhong University of Science and Technology, and others. Dr. Liu had no disclosures. One coinvestigator disclosed ties to Genentech/Roche, Lilly, Sanofi, Bayer, and several other biomedical companies.
SOURCE: Liu L et al. Clin Gastroenterol Hepatol. 2018 Apr 24. doi: 10.1016/j.cgh.2018.04.030.
FROM CLINICAL GASTROENTEROLOGY AND HEPATOLOGY
Key clinical point: A proinflammatory diet was associated with a significantly increased risk for colorectal cancer testing positive for Fusobacterium nucleatum.
Major finding: Dietary scores in the highest inflammatory tertile correlated with significantly increased risk (HR, 1.63; P = .03).
Study details: Longitudinal study of self-reported dietary patterns and cancers among 124,433 individuals with 28 years of follow-up.
Disclosures: Funders included the National Institutes of Health, Dana Farber Harvard Cancer, Project P. Fund for Colorectal Cancer Research, Friends of the Dana-Farber Cancer Institute, Bennett Family Fund, the Entertainment Industry Foundation, and American Association for Cancer Research, National Natural Science Foundation of China, Chinese Scholarship Council, Huazhong University of Science and Technology, and others. Dr. Liu had no disclosures. One coinvestigator disclosed ties to Genentech/Roche, Lilly, Sanofi, Bayer, and several other biomedical companies.
Source: Liu L et al. Clin Gastroenterol Hepatol. 2018 Apr 24. doi: 10.1016/j.cgh.2018.04.030.
Cirrhosis study finds no link between screening, liver cancer mortality
In a case-control study of patients with cirrhosis, screening for hepatocellular carcinoma up to 4 years prior to diagnosis was not associated with lower mortality.
Similar proportions of cases and controls underwent screening with abdominal ultrasonography, serum alpha-fetoprotein (AFP) testing, or both, reported Andrew M. Moon, MD, MPH, of the University of North Carolina at Chapel Hill, and his associates. “There was also no difference in receipt of these screening tests within 1, 2, or 3 years prior to the index date,” they wrote. The report was published in Gastroenterology. The findings “[suggest] that either these screening tests or the currently available treatments [for liver cancer], or both, are suboptimal and need to be improved.”
Because cirrhosis significantly increases the risk of hepatocellular carcinoma, the American Association for the Study of Liver Diseases, the European Association for the Study of the Liver, and the Asian Pacific Association for the Study of the Liver recommend screening cirrhotic patients every 6 months with abdominal ultrasonography with or without concomitant serum AFP. But nonliver societies have not endorsed this approach, citing a lack of high-quality data. One problem is that studies have compared patients whose liver cancer was diagnosed by screening with those diagnosed after they became symptomatic, which creates a lead-time bias that inherently favors screening, Dr. Moon and his associates noted.
To help fill the evidence gap, they identified 238 patients from the Veterans Affairs health care system who had died of hepatocellular carcinoma between 2013 and 2015 and who had been diagnosed with cirrhosis at least 4 years beforehand. They compared these cases with an equal number of patients with cirrhosis who had been in VA care for a similar amount of time and had not died of hepatocellular carcinoma. Cases and controls were matched by etiology of cirrhosis, year that cirrhosis was diagnosed, race, age, sex, Model for End-Stage Liver Disease score, and VA medical center. The researchers identified screening tests by reviewing blinded medical charts.
There were no significant differences in the proportions of cases and controls who underwent screening ultrasonography (52.9% versus 54.2%, respectively), screening serum AFP (74.8% versus 73.5%), either test (81.1% versus 79.4%), or both tests (46.6% versus 48.3%) within 4 years of the index date or the matched control. The result was similar after potential confounders were controlled for and when examining shorter time frames of 1, 2, and 3 years.
It was unlikely that these results reflect delayed diagnosis of liver cancer or a lack of treatment within the VA system, the experts wrote. A total of 51.3% of cases were diagnosed with Milan criteria, which exceeds the proportion in the national Surveillance, Epidemiology, and End Results registry, they noted. None of the fatal cases underwent liver transplantation, but 66.8% received other treatments for liver cancer.
Funders included the National Institutes of Health and the Veterans Affairs Clinical Science Research & Development. The investigators reported having no conflicts of interest.
SOURCE: Moon AM et al. Gastroenterology. 2018 Jul 5. doi: 10.1053/j.gastro.2018.06.079.
In a case-control study of patients with cirrhosis, screening for hepatocellular carcinoma up to 4 years prior to diagnosis was not associated with lower mortality.
Similar proportions of cases and controls underwent screening with abdominal ultrasonography, serum alpha-fetoprotein (AFP) testing, or both, reported Andrew M. Moon, MD, MPH, of the University of North Carolina at Chapel Hill, and his associates. “There was also no difference in receipt of these screening tests within 1, 2, or 3 years prior to the index date,” they wrote. The report was published in Gastroenterology. The findings “[suggest] that either these screening tests or the currently available treatments [for liver cancer], or both, are suboptimal and need to be improved.”
Because cirrhosis significantly increases the risk of hepatocellular carcinoma, the American Association for the Study of Liver Diseases, the European Association for the Study of the Liver, and the Asian Pacific Association for the Study of the Liver recommend screening cirrhotic patients every 6 months with abdominal ultrasonography with or without concomitant serum AFP. But nonliver societies have not endorsed this approach, citing a lack of high-quality data. One problem is that studies have compared patients whose liver cancer was diagnosed by screening with those diagnosed after they became symptomatic, which creates a lead-time bias that inherently favors screening, Dr. Moon and his associates noted.
To help fill the evidence gap, they identified 238 patients from the Veterans Affairs health care system who had died of hepatocellular carcinoma between 2013 and 2015 and who had been diagnosed with cirrhosis at least 4 years beforehand. They compared these cases with an equal number of patients with cirrhosis who had been in VA care for a similar amount of time and had not died of hepatocellular carcinoma. Cases and controls were matched by etiology of cirrhosis, year that cirrhosis was diagnosed, race, age, sex, Model for End-Stage Liver Disease score, and VA medical center. The researchers identified screening tests by reviewing blinded medical charts.
There were no significant differences in the proportions of cases and controls who underwent screening ultrasonography (52.9% versus 54.2%, respectively), screening serum AFP (74.8% versus 73.5%), either test (81.1% versus 79.4%), or both tests (46.6% versus 48.3%) within 4 years of the index date or the matched control. The result was similar after potential confounders were controlled for and when examining shorter time frames of 1, 2, and 3 years.
It was unlikely that these results reflect delayed diagnosis of liver cancer or a lack of treatment within the VA system, the experts wrote. A total of 51.3% of cases were diagnosed with Milan criteria, which exceeds the proportion in the national Surveillance, Epidemiology, and End Results registry, they noted. None of the fatal cases underwent liver transplantation, but 66.8% received other treatments for liver cancer.
Funders included the National Institutes of Health and the Veterans Affairs Clinical Science Research & Development. The investigators reported having no conflicts of interest.
SOURCE: Moon AM et al. Gastroenterology. 2018 Jul 5. doi: 10.1053/j.gastro.2018.06.079.
In a case-control study of patients with cirrhosis, screening for hepatocellular carcinoma up to 4 years prior to diagnosis was not associated with lower mortality.
Similar proportions of cases and controls underwent screening with abdominal ultrasonography, serum alpha-fetoprotein (AFP) testing, or both, reported Andrew M. Moon, MD, MPH, of the University of North Carolina at Chapel Hill, and his associates. “There was also no difference in receipt of these screening tests within 1, 2, or 3 years prior to the index date,” they wrote. The report was published in Gastroenterology. The findings “[suggest] that either these screening tests or the currently available treatments [for liver cancer], or both, are suboptimal and need to be improved.”
Because cirrhosis significantly increases the risk of hepatocellular carcinoma, the American Association for the Study of Liver Diseases, the European Association for the Study of the Liver, and the Asian Pacific Association for the Study of the Liver recommend screening cirrhotic patients every 6 months with abdominal ultrasonography with or without concomitant serum AFP. But nonliver societies have not endorsed this approach, citing a lack of high-quality data. One problem is that studies have compared patients whose liver cancer was diagnosed by screening with those diagnosed after they became symptomatic, which creates a lead-time bias that inherently favors screening, Dr. Moon and his associates noted.
To help fill the evidence gap, they identified 238 patients from the Veterans Affairs health care system who had died of hepatocellular carcinoma between 2013 and 2015 and who had been diagnosed with cirrhosis at least 4 years beforehand. They compared these cases with an equal number of patients with cirrhosis who had been in VA care for a similar amount of time and had not died of hepatocellular carcinoma. Cases and controls were matched by etiology of cirrhosis, year that cirrhosis was diagnosed, race, age, sex, Model for End-Stage Liver Disease score, and VA medical center. The researchers identified screening tests by reviewing blinded medical charts.
There were no significant differences in the proportions of cases and controls who underwent screening ultrasonography (52.9% versus 54.2%, respectively), screening serum AFP (74.8% versus 73.5%), either test (81.1% versus 79.4%), or both tests (46.6% versus 48.3%) within 4 years of the index date or the matched control. The result was similar after potential confounders were controlled for and when examining shorter time frames of 1, 2, and 3 years.
It was unlikely that these results reflect delayed diagnosis of liver cancer or a lack of treatment within the VA system, the experts wrote. A total of 51.3% of cases were diagnosed with Milan criteria, which exceeds the proportion in the national Surveillance, Epidemiology, and End Results registry, they noted. None of the fatal cases underwent liver transplantation, but 66.8% received other treatments for liver cancer.
Funders included the National Institutes of Health and the Veterans Affairs Clinical Science Research & Development. The investigators reported having no conflicts of interest.
SOURCE: Moon AM et al. Gastroenterology. 2018 Jul 5. doi: 10.1053/j.gastro.2018.06.079.
FROM GASTROENTEROLOGY
Key clinical point: Among patients with cirrhosis, screening for hepatocellular carcinoma was not associated with reductions in liver cancer mortality.
Major finding: Similar proportions of cases and controls were screened by abdominal ultrasonography, serum alpha-fetoprotein, or both up to 4 years before the index date and even after researchers controlled for relevant confounders.
Study details: A matched case-control study of 476 patients from the Veterans Affairs health care system.
Disclosures: Funders included the National Institutes of Health and the Veterans Affairs Clinical Science Research & Development. The investigators reported no conflicts of interest.
Source: Moon AM et al. Gastroenterology. 2018 Jul 5. doi: 10.1053/j.gastro.2018.06.079.
Experts update diagnostic guidelines for eosinophilic esophagitis
The diagnosis of eosinophilic esophagitis no longer needs to include a trial of proton pump inhibitor (PPI) therapy, according to an updated international consensus statement published in the October issue of Gastroenterology.
“An initial rationale for the PPI trial was to distinguish eosinophilic esophagitis from gastroesophageal reflux disease, but it is now known that these conditions have a complex relationship and are not necessarily mutually exclusive,” wrote Evan S. Dellon, MD, of the University of North Carolina at Chapel Hill, and his associates. According to current evidence, “PPIs are better classified as a treatment for esophageal eosinophilia that may be due to eosinophilic esophagitis than as a diagnostic criterion,” they said.
Diagnostic guidelines for eosinophilic esophagitis were published first in 2007 and were updated in 2011. The guideline authors recommended either pH monitoring or an 8-week trial of high-dose PPI therapy to rule out inflammation from gastroesophageal reflux disease (GERD).
But subsequent publications described patients with symptomatic esophageal eosinophilia who responded to PPIs and lacked classic GERD symptoms. Guidelines called this condition “PPI-responsive esophageal eosinophilia” and considered it a separate entity from GERD.
However, an “evolving body of research” shows that eosinophilic esophagitis can overlap with GERD, Dr. Dellon and his associates wrote. Furthermore, each of these conditions can trigger the other. Eosinophilic esophagitis can decrease esophageal compliance, leading to secondary reflux, while gastroesophageal reflux can erode the esophageal epithelium, triggering antigen exposure and eosinophilia.
Therefore, Dr. Dellon and his associates recommended defining eosinophilic esophagitis as signs and symptoms of esophageal dysfunction and an esophageal biopsy showing at least 15 eosinophils per high-power field, or approximately 60 eosinophils per millimeter, with infiltration limited to the esophagus. They stressed the importance of esophageal biopsy even if endoscopy shows normal mucosa. “As per prior guidelines, multiple biopsies from two or more esophageal levels, targeting areas of apparent inflammation, are recommended to increase the diagnostic yield,” they added. “Gastric and duodenal biopsies should be obtained as clinically indicated by symptoms, endoscopic findings in the stomach or duodenum, or high index of suspicion for a mucosal process.”
Physicians should increase their suspicion of eosinophilic esophagitis if patients have other types of atopy or endoscopic findings of “rings, furrows, exudates, edema, stricture, narrowing, and crepe-paper mucosa,” they added. In addition to GERD, they recommended looking carefully for other conditions that can trigger esophageal eosinophilia, such as pemphigus, drug hypersensitivity reactions, achalasia, and Crohn’s disease with esophageal involvement.
To create the guideline, Dr. Dellon and his associates searched PubMed for studies of all designs and sizes published from 1966 through December 2016. Teams of experts on specific topics then reviewed and discussed relevant literature. In May 2017, 43 reviewers met for 8 hours to present and discuss conclusions. There was 100% agreement to remove the PPI trial from the diagnostic criteria, the experts noted.
The authors disclosed financial support from the International Gastrointestinal Eosinophilic Diseases Researchers (TIGERS), The David and Denise Bunning Family, and the Rare Disease Clinical Research Network. Dr. Dellon disclosed consulting relationships and receiving research funding from Adare, Celgene/Receptos, Regeneron, and Shire among others. The majority of his coauthors also disclosed relationships with numerous medical companies.
SOURCE: Dellon ES et al. Gastroenterology. 2018 Jul 12. doi: 10.1053/j.gastro.2018.07.009.
Studies in the 1980s linked the presence of esophageal mucosal eosinophils with increased acid exposure on pH monitoring. For the next 2 decades, clinicians viewed eosinophils on esophageal biopsies as diagnostic for GERD such that the initial description of EoE by Attwood in 1993 distinguished EoE from GERD by the presence of esophageal eosinophilia in the absence of either reflux esophagitis or abnormal acid exposure on pH testing. Consequently, the initial diagnostic criteria for EoE in 2007 included a lack of response to PPI and/or normal pH testing to establish the diagnosis of EoE. Reflecting growing uncertainty regarding the ability of PPI therapy to differentiate acid-induced from allergic inflammatory mechanisms, an updated consensus in 2011 introduced the terminology “PPI responsive esophageal eosinophilia (PPIREE)” to describe an increasingly recognized subset of patients with suspected EoE that resolved with PPI. Now, supported by scientific evidence accumulated over the past decade, AGREE has taken a step back by removing the PPI trial from the diagnosis of EoE, thereby abandoning the PPIREE terminology. This step simplifies the diagnosis of EoE and acknowledges that a histologic response to PPI does not “rule in” GERD or “rule out” EoE. It is important to emphasize that the updated criteria still advocate careful consideration of secondary causes of esophageal eosinophilia prior to the diagnosis of EoE.
Ramifications of the updated diagnostic criteria include the opportunities for clinicians to consider use of topical corticosteroids and diet therapies, rather than mandate an up-front PPI trial, in patients with EoE. On a practical level, based on their effectiveness, safety, and ease of administration, PPIs remain positioned as a favorable initial intervention for EoE. Conceptually, however, the paradigm shift highlights the ability of research to improve our understanding of disease pathogenesis and thereby impact clinical management.
Ikuo Hirano, MD, AGAF, is in the division of gastroenterology, Northwestern University, Chicago. He has received grant support from the NIH Consortium of Eosinophilic Gastrointestinal Disease Researchers (CEGIR, U54 AI117804), which is part of the Rare Disease Clinical Research Network. He has received research funding and consulting fees from Celgene, Regeneron, Shire, and others.
Studies in the 1980s linked the presence of esophageal mucosal eosinophils with increased acid exposure on pH monitoring. For the next 2 decades, clinicians viewed eosinophils on esophageal biopsies as diagnostic for GERD such that the initial description of EoE by Attwood in 1993 distinguished EoE from GERD by the presence of esophageal eosinophilia in the absence of either reflux esophagitis or abnormal acid exposure on pH testing. Consequently, the initial diagnostic criteria for EoE in 2007 included a lack of response to PPI and/or normal pH testing to establish the diagnosis of EoE. Reflecting growing uncertainty regarding the ability of PPI therapy to differentiate acid-induced from allergic inflammatory mechanisms, an updated consensus in 2011 introduced the terminology “PPI responsive esophageal eosinophilia (PPIREE)” to describe an increasingly recognized subset of patients with suspected EoE that resolved with PPI. Now, supported by scientific evidence accumulated over the past decade, AGREE has taken a step back by removing the PPI trial from the diagnosis of EoE, thereby abandoning the PPIREE terminology. This step simplifies the diagnosis of EoE and acknowledges that a histologic response to PPI does not “rule in” GERD or “rule out” EoE. It is important to emphasize that the updated criteria still advocate careful consideration of secondary causes of esophageal eosinophilia prior to the diagnosis of EoE.
Ramifications of the updated diagnostic criteria include the opportunities for clinicians to consider use of topical corticosteroids and diet therapies, rather than mandate an up-front PPI trial, in patients with EoE. On a practical level, based on their effectiveness, safety, and ease of administration, PPIs remain positioned as a favorable initial intervention for EoE. Conceptually, however, the paradigm shift highlights the ability of research to improve our understanding of disease pathogenesis and thereby impact clinical management.
Ikuo Hirano, MD, AGAF, is in the division of gastroenterology, Northwestern University, Chicago. He has received grant support from the NIH Consortium of Eosinophilic Gastrointestinal Disease Researchers (CEGIR, U54 AI117804), which is part of the Rare Disease Clinical Research Network. He has received research funding and consulting fees from Celgene, Regeneron, Shire, and others.
Studies in the 1980s linked the presence of esophageal mucosal eosinophils with increased acid exposure on pH monitoring. For the next 2 decades, clinicians viewed eosinophils on esophageal biopsies as diagnostic for GERD such that the initial description of EoE by Attwood in 1993 distinguished EoE from GERD by the presence of esophageal eosinophilia in the absence of either reflux esophagitis or abnormal acid exposure on pH testing. Consequently, the initial diagnostic criteria for EoE in 2007 included a lack of response to PPI and/or normal pH testing to establish the diagnosis of EoE. Reflecting growing uncertainty regarding the ability of PPI therapy to differentiate acid-induced from allergic inflammatory mechanisms, an updated consensus in 2011 introduced the terminology “PPI responsive esophageal eosinophilia (PPIREE)” to describe an increasingly recognized subset of patients with suspected EoE that resolved with PPI. Now, supported by scientific evidence accumulated over the past decade, AGREE has taken a step back by removing the PPI trial from the diagnosis of EoE, thereby abandoning the PPIREE terminology. This step simplifies the diagnosis of EoE and acknowledges that a histologic response to PPI does not “rule in” GERD or “rule out” EoE. It is important to emphasize that the updated criteria still advocate careful consideration of secondary causes of esophageal eosinophilia prior to the diagnosis of EoE.
Ramifications of the updated diagnostic criteria include the opportunities for clinicians to consider use of topical corticosteroids and diet therapies, rather than mandate an up-front PPI trial, in patients with EoE. On a practical level, based on their effectiveness, safety, and ease of administration, PPIs remain positioned as a favorable initial intervention for EoE. Conceptually, however, the paradigm shift highlights the ability of research to improve our understanding of disease pathogenesis and thereby impact clinical management.
Ikuo Hirano, MD, AGAF, is in the division of gastroenterology, Northwestern University, Chicago. He has received grant support from the NIH Consortium of Eosinophilic Gastrointestinal Disease Researchers (CEGIR, U54 AI117804), which is part of the Rare Disease Clinical Research Network. He has received research funding and consulting fees from Celgene, Regeneron, Shire, and others.
The diagnosis of eosinophilic esophagitis no longer needs to include a trial of proton pump inhibitor (PPI) therapy, according to an updated international consensus statement published in the October issue of Gastroenterology.
“An initial rationale for the PPI trial was to distinguish eosinophilic esophagitis from gastroesophageal reflux disease, but it is now known that these conditions have a complex relationship and are not necessarily mutually exclusive,” wrote Evan S. Dellon, MD, of the University of North Carolina at Chapel Hill, and his associates. According to current evidence, “PPIs are better classified as a treatment for esophageal eosinophilia that may be due to eosinophilic esophagitis than as a diagnostic criterion,” they said.
Diagnostic guidelines for eosinophilic esophagitis were published first in 2007 and were updated in 2011. The guideline authors recommended either pH monitoring or an 8-week trial of high-dose PPI therapy to rule out inflammation from gastroesophageal reflux disease (GERD).
But subsequent publications described patients with symptomatic esophageal eosinophilia who responded to PPIs and lacked classic GERD symptoms. Guidelines called this condition “PPI-responsive esophageal eosinophilia” and considered it a separate entity from GERD.
However, an “evolving body of research” shows that eosinophilic esophagitis can overlap with GERD, Dr. Dellon and his associates wrote. Furthermore, each of these conditions can trigger the other. Eosinophilic esophagitis can decrease esophageal compliance, leading to secondary reflux, while gastroesophageal reflux can erode the esophageal epithelium, triggering antigen exposure and eosinophilia.
Therefore, Dr. Dellon and his associates recommended defining eosinophilic esophagitis as signs and symptoms of esophageal dysfunction and an esophageal biopsy showing at least 15 eosinophils per high-power field, or approximately 60 eosinophils per millimeter, with infiltration limited to the esophagus. They stressed the importance of esophageal biopsy even if endoscopy shows normal mucosa. “As per prior guidelines, multiple biopsies from two or more esophageal levels, targeting areas of apparent inflammation, are recommended to increase the diagnostic yield,” they added. “Gastric and duodenal biopsies should be obtained as clinically indicated by symptoms, endoscopic findings in the stomach or duodenum, or high index of suspicion for a mucosal process.”
Physicians should increase their suspicion of eosinophilic esophagitis if patients have other types of atopy or endoscopic findings of “rings, furrows, exudates, edema, stricture, narrowing, and crepe-paper mucosa,” they added. In addition to GERD, they recommended looking carefully for other conditions that can trigger esophageal eosinophilia, such as pemphigus, drug hypersensitivity reactions, achalasia, and Crohn’s disease with esophageal involvement.
To create the guideline, Dr. Dellon and his associates searched PubMed for studies of all designs and sizes published from 1966 through December 2016. Teams of experts on specific topics then reviewed and discussed relevant literature. In May 2017, 43 reviewers met for 8 hours to present and discuss conclusions. There was 100% agreement to remove the PPI trial from the diagnostic criteria, the experts noted.
The authors disclosed financial support from the International Gastrointestinal Eosinophilic Diseases Researchers (TIGERS), The David and Denise Bunning Family, and the Rare Disease Clinical Research Network. Dr. Dellon disclosed consulting relationships and receiving research funding from Adare, Celgene/Receptos, Regeneron, and Shire among others. The majority of his coauthors also disclosed relationships with numerous medical companies.
SOURCE: Dellon ES et al. Gastroenterology. 2018 Jul 12. doi: 10.1053/j.gastro.2018.07.009.
The diagnosis of eosinophilic esophagitis no longer needs to include a trial of proton pump inhibitor (PPI) therapy, according to an updated international consensus statement published in the October issue of Gastroenterology.
“An initial rationale for the PPI trial was to distinguish eosinophilic esophagitis from gastroesophageal reflux disease, but it is now known that these conditions have a complex relationship and are not necessarily mutually exclusive,” wrote Evan S. Dellon, MD, of the University of North Carolina at Chapel Hill, and his associates. According to current evidence, “PPIs are better classified as a treatment for esophageal eosinophilia that may be due to eosinophilic esophagitis than as a diagnostic criterion,” they said.
Diagnostic guidelines for eosinophilic esophagitis were published first in 2007 and were updated in 2011. The guideline authors recommended either pH monitoring or an 8-week trial of high-dose PPI therapy to rule out inflammation from gastroesophageal reflux disease (GERD).
But subsequent publications described patients with symptomatic esophageal eosinophilia who responded to PPIs and lacked classic GERD symptoms. Guidelines called this condition “PPI-responsive esophageal eosinophilia” and considered it a separate entity from GERD.
However, an “evolving body of research” shows that eosinophilic esophagitis can overlap with GERD, Dr. Dellon and his associates wrote. Furthermore, each of these conditions can trigger the other. Eosinophilic esophagitis can decrease esophageal compliance, leading to secondary reflux, while gastroesophageal reflux can erode the esophageal epithelium, triggering antigen exposure and eosinophilia.
Therefore, Dr. Dellon and his associates recommended defining eosinophilic esophagitis as signs and symptoms of esophageal dysfunction and an esophageal biopsy showing at least 15 eosinophils per high-power field, or approximately 60 eosinophils per millimeter, with infiltration limited to the esophagus. They stressed the importance of esophageal biopsy even if endoscopy shows normal mucosa. “As per prior guidelines, multiple biopsies from two or more esophageal levels, targeting areas of apparent inflammation, are recommended to increase the diagnostic yield,” they added. “Gastric and duodenal biopsies should be obtained as clinically indicated by symptoms, endoscopic findings in the stomach or duodenum, or high index of suspicion for a mucosal process.”
Physicians should increase their suspicion of eosinophilic esophagitis if patients have other types of atopy or endoscopic findings of “rings, furrows, exudates, edema, stricture, narrowing, and crepe-paper mucosa,” they added. In addition to GERD, they recommended looking carefully for other conditions that can trigger esophageal eosinophilia, such as pemphigus, drug hypersensitivity reactions, achalasia, and Crohn’s disease with esophageal involvement.
To create the guideline, Dr. Dellon and his associates searched PubMed for studies of all designs and sizes published from 1966 through December 2016. Teams of experts on specific topics then reviewed and discussed relevant literature. In May 2017, 43 reviewers met for 8 hours to present and discuss conclusions. There was 100% agreement to remove the PPI trial from the diagnostic criteria, the experts noted.
The authors disclosed financial support from the International Gastrointestinal Eosinophilic Diseases Researchers (TIGERS), The David and Denise Bunning Family, and the Rare Disease Clinical Research Network. Dr. Dellon disclosed consulting relationships and receiving research funding from Adare, Celgene/Receptos, Regeneron, and Shire among others. The majority of his coauthors also disclosed relationships with numerous medical companies.
SOURCE: Dellon ES et al. Gastroenterology. 2018 Jul 12. doi: 10.1053/j.gastro.2018.07.009.
FROM GASTROENTEROLOGY
Key clinical point: The diagnosis of eosinophilic esophagitis no longer needs to include a trial of proton pump inhibitor therapy.
Major finding: Eosinophilic esophagitis and gastroesophageal reflux disease are not mutually exclusive.
Study details: Review by an international consensus panel of studies published between 1966 and 2016.
Disclosures: The authors disclosed financial support from the International Gastrointestinal Eosinophilic Diseases Researchers (TIGERS), The David and Denise Bunning Family, the Rare Disease Clinical Research Network. Dr. Dellon disclosed consulting relationships with Adare, Allakos, Alivio, Banner, Celgene/Receptos, Enumeral, GSK, Regeneron, and Shire. He also reported receiving research funding from Adare, Celgene/Receptos, Miraca, Meritage, Nutricia, Regeneron, and Shire and educational grants from Banner and Holoclara. The majority of his coauthors disclosed relationships with numerous medical companies.
Source: Dellon ES et al. Gastroenterology. 2018 Jul 12. doi: 10.1053/j.gastro.2018.07.009.
AGA Guideline: Treatment of opioid-induced constipation
For patients with suspected opioid-induced constipation, start by taking a careful history of defecation and dietary patterns, stool consistency, incomplete evacuation, and “alarm symptoms,” such as bloody stools or weight loss, state new guidelines from the American Gastroenterological Association in Gastroenterology.
Clinicians also should rule out other causes of constipation, such as pelvic outlet dysfunction, mechanical obstruction, metabolic abnormalities, and comorbidities or concurrent medications, wrote Seth D. Crockett, MD, MPH, of the University of North Carolina at Chapel Hill, together with his associates. The guideline was published online Sept. 1.
Opioid therapy can lead to a range of gastrointestinal symptoms, such as constipation, gastroesophageal reflux, nausea and vomiting, bloating, and abdominal pain. Among these, constipation is by far the most common and debilitating, the guideline notes. In past studies, 40%-80% of patients who received opioids developed opioid-induced constipation (OIC), a more severe presentation that involves a combination of reduced stool frequency in addition to other symptoms, such as harder stools, new or worsening straining during defecation, and a sense of incomplete rectal evacuation.
Treating OIC should start with lifestyle interventions, such as drinking more fluids, toileting as soon as possible when feeling the urge to defecate, and adding regular moderate exercise whenever tolerable, the guideline advises. For patients on oral or parenteral therapy, consider switching to an equianalgesic dose of a less-constipating opioid, such as transdermal fentanyl or oxycodone-naloxone combination therapy.
Many patients with OIC require interventions beyond lifestyle changes or opioid switching. For these patients, the guideline advises starting with conventional laxative therapies based on their safety, low cost, and “established efficacy” in the OIC setting. Options include stool softeners (docusate sodium), osmotic laxatives (polyethylene glycol, magnesium hydroxide, magnesium citrate, and lactulose), lubricants (mineral oil), and stimulant laxatives (bisacodyl, sodium picosulfate, and senna). “Of note, there is little evidence that routine use of stimulant laxatives is harmful to the colon, despite widespread concern to the contrary,” the guideline states. Although randomized, controlled trials have not evaluated particular laxative combinations or titrations for OIC, the best evidence supports stimulant and osmotic laxative therapy, the authors note.
Before deeming any case of OIC laxative refractory, ensure that a patient receives an adequate trial of at least two classes of laxatives administered on a regular schedule, not just “as needed,” the guideline specifies. For example, a patient might receive a 2-week trial of a daily osmotic laxative plus a stimulant laxative two to three times weekly. The guideline authors suggest restricting the use of enemas to rescue therapy. They also note that consuming more fiber tends not to help patients with OIC because fiber does not affect colonic motility.
For truly laxative-refractory OIC, the guidelines recommend escalating treatment to peripherally acting mu-opioid receptor antagonists (PAMORAs). These drugs restore the function of the enteric nervous system by blocking mu-opioid receptors in the gut. Among the PAMORAs, the guideline strongly recommends the use of naldemedine or naloxegol over no treatment, based on robust data from randomized, double-blind, placebo-controlled trials. In the phase 3 COMPOSE 1, 2, and 3 trials, about 52% of patients who received naldemedine achieved at least three spontaneous bowel movements per week, compared with 35% of patients who received placebo. Additionally, in a 52-week safety and efficacy study (COMPOSE 3), naldemedine was associated with one more spontaneous bowel movement per week versus placebo and with a low absolute increase in adverse events.
The guideline bases its strong recommendation for naloxegol on moderate-quality data from three studies, including two phase 3, double-blind, randomized, placebo-controlled trials. Although at least five randomized, controlled trials have evaluated methylnaltrexone, the evidence was low quality and therefore the guideline only conditionally recommends prescribing this PAMORA over no treatment.
The guideline also makes no recommendation on the use of the intestinal secretagogue lubiprostone or the 5HT agonist prucalopride. Studies of lubiprostone were limited by possible reporting bias and showed no clear treatment benefit, the authors state. They describe a similar evidence gap for prucalopride, noting that at least one trial ended early without publication of the findings. They recommend further studying lubiprostone as well as prucalopride and other highly selective 5-HT4 agonists for treating OIC. Head-to-head trials would help guide treatment choice for patients with laxative-refractory OIC, they add. “Cost-effectiveness studies are also lacking in this field, which could inform prescribing strategy, particularly for newer, more expensive agents.”
For patients with suspected opioid-induced constipation, start by taking a careful history of defecation and dietary patterns, stool consistency, incomplete evacuation, and “alarm symptoms,” such as bloody stools or weight loss, state new guidelines from the American Gastroenterological Association in Gastroenterology.
Clinicians also should rule out other causes of constipation, such as pelvic outlet dysfunction, mechanical obstruction, metabolic abnormalities, and comorbidities or concurrent medications, wrote Seth D. Crockett, MD, MPH, of the University of North Carolina at Chapel Hill, together with his associates. The guideline was published online Sept. 1.
Opioid therapy can lead to a range of gastrointestinal symptoms, such as constipation, gastroesophageal reflux, nausea and vomiting, bloating, and abdominal pain. Among these, constipation is by far the most common and debilitating, the guideline notes. In past studies, 40%-80% of patients who received opioids developed opioid-induced constipation (OIC), a more severe presentation that involves a combination of reduced stool frequency in addition to other symptoms, such as harder stools, new or worsening straining during defecation, and a sense of incomplete rectal evacuation.
Treating OIC should start with lifestyle interventions, such as drinking more fluids, toileting as soon as possible when feeling the urge to defecate, and adding regular moderate exercise whenever tolerable, the guideline advises. For patients on oral or parenteral therapy, consider switching to an equianalgesic dose of a less-constipating opioid, such as transdermal fentanyl or oxycodone-naloxone combination therapy.
Many patients with OIC require interventions beyond lifestyle changes or opioid switching. For these patients, the guideline advises starting with conventional laxative therapies based on their safety, low cost, and “established efficacy” in the OIC setting. Options include stool softeners (docusate sodium), osmotic laxatives (polyethylene glycol, magnesium hydroxide, magnesium citrate, and lactulose), lubricants (mineral oil), and stimulant laxatives (bisacodyl, sodium picosulfate, and senna). “Of note, there is little evidence that routine use of stimulant laxatives is harmful to the colon, despite widespread concern to the contrary,” the guideline states. Although randomized, controlled trials have not evaluated particular laxative combinations or titrations for OIC, the best evidence supports stimulant and osmotic laxative therapy, the authors note.
Before deeming any case of OIC laxative refractory, ensure that a patient receives an adequate trial of at least two classes of laxatives administered on a regular schedule, not just “as needed,” the guideline specifies. For example, a patient might receive a 2-week trial of a daily osmotic laxative plus a stimulant laxative two to three times weekly. The guideline authors suggest restricting the use of enemas to rescue therapy. They also note that consuming more fiber tends not to help patients with OIC because fiber does not affect colonic motility.
For truly laxative-refractory OIC, the guidelines recommend escalating treatment to peripherally acting mu-opioid receptor antagonists (PAMORAs). These drugs restore the function of the enteric nervous system by blocking mu-opioid receptors in the gut. Among the PAMORAs, the guideline strongly recommends the use of naldemedine or naloxegol over no treatment, based on robust data from randomized, double-blind, placebo-controlled trials. In the phase 3 COMPOSE 1, 2, and 3 trials, about 52% of patients who received naldemedine achieved at least three spontaneous bowel movements per week, compared with 35% of patients who received placebo. Additionally, in a 52-week safety and efficacy study (COMPOSE 3), naldemedine was associated with one more spontaneous bowel movement per week versus placebo and with a low absolute increase in adverse events.
The guideline bases its strong recommendation for naloxegol on moderate-quality data from three studies, including two phase 3, double-blind, randomized, placebo-controlled trials. Although at least five randomized, controlled trials have evaluated methylnaltrexone, the evidence was low quality and therefore the guideline only conditionally recommends prescribing this PAMORA over no treatment.
The guideline also makes no recommendation on the use of the intestinal secretagogue lubiprostone or the 5HT agonist prucalopride. Studies of lubiprostone were limited by possible reporting bias and showed no clear treatment benefit, the authors state. They describe a similar evidence gap for prucalopride, noting that at least one trial ended early without publication of the findings. They recommend further studying lubiprostone as well as prucalopride and other highly selective 5-HT4 agonists for treating OIC. Head-to-head trials would help guide treatment choice for patients with laxative-refractory OIC, they add. “Cost-effectiveness studies are also lacking in this field, which could inform prescribing strategy, particularly for newer, more expensive agents.”
For patients with suspected opioid-induced constipation, start by taking a careful history of defecation and dietary patterns, stool consistency, incomplete evacuation, and “alarm symptoms,” such as bloody stools or weight loss, state new guidelines from the American Gastroenterological Association in Gastroenterology.
Clinicians also should rule out other causes of constipation, such as pelvic outlet dysfunction, mechanical obstruction, metabolic abnormalities, and comorbidities or concurrent medications, wrote Seth D. Crockett, MD, MPH, of the University of North Carolina at Chapel Hill, together with his associates. The guideline was published online Sept. 1.
Opioid therapy can lead to a range of gastrointestinal symptoms, such as constipation, gastroesophageal reflux, nausea and vomiting, bloating, and abdominal pain. Among these, constipation is by far the most common and debilitating, the guideline notes. In past studies, 40%-80% of patients who received opioids developed opioid-induced constipation (OIC), a more severe presentation that involves a combination of reduced stool frequency in addition to other symptoms, such as harder stools, new or worsening straining during defecation, and a sense of incomplete rectal evacuation.
Treating OIC should start with lifestyle interventions, such as drinking more fluids, toileting as soon as possible when feeling the urge to defecate, and adding regular moderate exercise whenever tolerable, the guideline advises. For patients on oral or parenteral therapy, consider switching to an equianalgesic dose of a less-constipating opioid, such as transdermal fentanyl or oxycodone-naloxone combination therapy.
Many patients with OIC require interventions beyond lifestyle changes or opioid switching. For these patients, the guideline advises starting with conventional laxative therapies based on their safety, low cost, and “established efficacy” in the OIC setting. Options include stool softeners (docusate sodium), osmotic laxatives (polyethylene glycol, magnesium hydroxide, magnesium citrate, and lactulose), lubricants (mineral oil), and stimulant laxatives (bisacodyl, sodium picosulfate, and senna). “Of note, there is little evidence that routine use of stimulant laxatives is harmful to the colon, despite widespread concern to the contrary,” the guideline states. Although randomized, controlled trials have not evaluated particular laxative combinations or titrations for OIC, the best evidence supports stimulant and osmotic laxative therapy, the authors note.
Before deeming any case of OIC laxative refractory, ensure that a patient receives an adequate trial of at least two classes of laxatives administered on a regular schedule, not just “as needed,” the guideline specifies. For example, a patient might receive a 2-week trial of a daily osmotic laxative plus a stimulant laxative two to three times weekly. The guideline authors suggest restricting the use of enemas to rescue therapy. They also note that consuming more fiber tends not to help patients with OIC because fiber does not affect colonic motility.
For truly laxative-refractory OIC, the guidelines recommend escalating treatment to peripherally acting mu-opioid receptor antagonists (PAMORAs). These drugs restore the function of the enteric nervous system by blocking mu-opioid receptors in the gut. Among the PAMORAs, the guideline strongly recommends the use of naldemedine or naloxegol over no treatment, based on robust data from randomized, double-blind, placebo-controlled trials. In the phase 3 COMPOSE 1, 2, and 3 trials, about 52% of patients who received naldemedine achieved at least three spontaneous bowel movements per week, compared with 35% of patients who received placebo. Additionally, in a 52-week safety and efficacy study (COMPOSE 3), naldemedine was associated with one more spontaneous bowel movement per week versus placebo and with a low absolute increase in adverse events.
The guideline bases its strong recommendation for naloxegol on moderate-quality data from three studies, including two phase 3, double-blind, randomized, placebo-controlled trials. Although at least five randomized, controlled trials have evaluated methylnaltrexone, the evidence was low quality and therefore the guideline only conditionally recommends prescribing this PAMORA over no treatment.
The guideline also makes no recommendation on the use of the intestinal secretagogue lubiprostone or the 5HT agonist prucalopride. Studies of lubiprostone were limited by possible reporting bias and showed no clear treatment benefit, the authors state. They describe a similar evidence gap for prucalopride, noting that at least one trial ended early without publication of the findings. They recommend further studying lubiprostone as well as prucalopride and other highly selective 5-HT4 agonists for treating OIC. Head-to-head trials would help guide treatment choice for patients with laxative-refractory OIC, they add. “Cost-effectiveness studies are also lacking in this field, which could inform prescribing strategy, particularly for newer, more expensive agents.”
FROM GASTROENTEROLOGY
Nerve growth factor therapy speeds gastric ulcer healing
a recent study found.
Compared with young individuals, elderly people have significantly lower levels of NGF in gastric endothelial cells (GECs), a finding that is associated with impaired angiogenesis and delayed gastric ulcer healing.
“Our previous studies have shown that the gastric mucosa of aging individuals ... has increased susceptibility to injury and delayed healing owing to impaired angiogenesis, but the mechanisms are not fully elucidated,” wrote Amrita Ahluwalia, PhD, of Medical and Research Services at the Veterans Affairs Long Beach (Calif.) Healthcare System and her coauthors.
Mapping the drivers of angiogenesis in the gastric mucosa could lead to treatment options for elderly patients with injured or ulcerated gastric tissue. In prior trials (with rats), “treatment with VEGF [vascular endothelial growth factor] only partly reversed impaired angiogenesis in aging [GECs], indicating an essential role for other factor(s) in addition to VEGF,” the investigators wrote in the September issue of Cellular and Molecular Gastroenterology and Hepatology. They looked to NGF as another possible factor because recent studies had shown it could improve angiogenesis in the brain.
The present study measured NGF expression in rats and humans of varying ages, with NGF treatment and gene therapy performed in rats (in vitro and in vivo).
In vitro angiogenesis was 4.1-fold lower in GECs from aging rats (24 months of age) than it was in GECs from young rats (3 months of age; P less than .001). NGF protein and NGF mRNA levels were also significantly lower in aging GECs than they were with young GECs (NGF protein, 3.0-fold lower; NGF mRNA, 4.2-fold lower; P less than .001).
Treatment of aging rat GECs with exogenous NGF increased angiogenesis by 1.5-fold (P less than .001). Pretreatment with a PI3 kinase inhibitor or an mTOR inhibitor abolished this improvement, suggesting that the PI3 kinase/Akt and mTOR pathways are involved.
When NGF gene therapy was performed in aging GECs, NGF levels rose to the level of that in young GECs, with an accompanying restoration of angiogenesis (threefold increase; P less than .001). Proliferation of aging GECs also increased with gene therapy (P less than .001).
In vivo studies revealed that NGF expression and cell proliferation in aging rat gastric mucosa were lower than in younger rats. Of note, older rats treated with local NGF protein showed increased gastric mucosa angiogenesis and faster ulcer healing, compared with phosphate-buffered saline treatment.
Similar age-related NGF declines were found in humans. When gastric mucosa biopsies were collected from younger individuals (younger than 40 years old; n = 10) and compared with samples from an older population (at least 70 years old; n = 10), the investigators found that NGF expression was 5.5-fold lower in the older people (P less than .001).
“This clearly indicates human relevance of our experimental findings and also can explain impaired angiogenesis and delayed healing of injured gastric mucosa in aging individuals,” the investigators wrote.
“Aging gastropathy and its consequences are clinically critical issues,” the investigators noted, “especially because the aging U.S. population is growing rapidly and it is estimated that, by the year 2030, approximately 70 million Americans will be older than 65 years of age.” Gastric ulcers become more common with age, and individuals 70 years or older have an eightfold increased risk of associated complications, compared with people under 50 years.
The investigators noted that multiple growth factors likely play a role in stimulation of angiogenesis, including NGF, VEGF, epidermal growth factor, and basic fibroblast growth factor. “Further studies are necessary to investigate the role of other growth factors and cytokines in angiogenesis, [gastric ulcer] healing, and their impairment in aging,” the investigators concluded.
The study was funded by the Department of Veterans Affairs Biomedical Laboratory Research and Development Service. The authors declared no conflicts of interest.
SOURCE: Ahluwalia A et al. CMGH. 2018 May 17. doi: 10.1016/j.jcmgh.2018.05.003
Although the incidence of gastric ulcers has been declining in the general population, hospitalization and mortality linked to gastric ulcers remains high in the elderly population. One of the major risk factors for gastric ulceration is the use of NSAIDs. It is estimated that 40% of individuals aged 65 years and older fill at least one prescription for an NSAID each year. Given that the elderly population (those aged 65 years and older) is anticipated to more than double by the year 2050, reaching 84 million, understanding the pathogenesis of gastric ulceration is increasingly relevant.
This study described a new role for nerve growth factor (NGF) in promoting angiogenesis during gastric ulcer repair. The authors observed that aged rats exhibited low NGF levels in gastric endothelial cells that corresponded with impaired ulcer healing of the gastric mucosa following injury. Local NGF treatment to aged rats significantly increased angiogenesis and gastric regeneration. Consistent with their in vivo rat model, analysis of human gastric biopsy specimens showed that individuals more than 70 years of age had decreased expression of NGF in gastric endothelial cells, compared with individuals younger than 40 years.
Ahluwalia and colleagues are the first to demonstrate the role of NGF in aging gastropathy, and their work highlights a key mechanism of angiogenesis during gastric repair that may inform future therapeutic strategies.
Amy Christine Engevik, PhD, is a postdoctoral fellow in the division of surgical sciences at Vanderbilt University Medical Center, Nashville, Tenn. She has no conflicts of interest.
Although the incidence of gastric ulcers has been declining in the general population, hospitalization and mortality linked to gastric ulcers remains high in the elderly population. One of the major risk factors for gastric ulceration is the use of NSAIDs. It is estimated that 40% of individuals aged 65 years and older fill at least one prescription for an NSAID each year. Given that the elderly population (those aged 65 years and older) is anticipated to more than double by the year 2050, reaching 84 million, understanding the pathogenesis of gastric ulceration is increasingly relevant.
This study described a new role for nerve growth factor (NGF) in promoting angiogenesis during gastric ulcer repair. The authors observed that aged rats exhibited low NGF levels in gastric endothelial cells that corresponded with impaired ulcer healing of the gastric mucosa following injury. Local NGF treatment to aged rats significantly increased angiogenesis and gastric regeneration. Consistent with their in vivo rat model, analysis of human gastric biopsy specimens showed that individuals more than 70 years of age had decreased expression of NGF in gastric endothelial cells, compared with individuals younger than 40 years.
Ahluwalia and colleagues are the first to demonstrate the role of NGF in aging gastropathy, and their work highlights a key mechanism of angiogenesis during gastric repair that may inform future therapeutic strategies.
Amy Christine Engevik, PhD, is a postdoctoral fellow in the division of surgical sciences at Vanderbilt University Medical Center, Nashville, Tenn. She has no conflicts of interest.
Although the incidence of gastric ulcers has been declining in the general population, hospitalization and mortality linked to gastric ulcers remains high in the elderly population. One of the major risk factors for gastric ulceration is the use of NSAIDs. It is estimated that 40% of individuals aged 65 years and older fill at least one prescription for an NSAID each year. Given that the elderly population (those aged 65 years and older) is anticipated to more than double by the year 2050, reaching 84 million, understanding the pathogenesis of gastric ulceration is increasingly relevant.
This study described a new role for nerve growth factor (NGF) in promoting angiogenesis during gastric ulcer repair. The authors observed that aged rats exhibited low NGF levels in gastric endothelial cells that corresponded with impaired ulcer healing of the gastric mucosa following injury. Local NGF treatment to aged rats significantly increased angiogenesis and gastric regeneration. Consistent with their in vivo rat model, analysis of human gastric biopsy specimens showed that individuals more than 70 years of age had decreased expression of NGF in gastric endothelial cells, compared with individuals younger than 40 years.
Ahluwalia and colleagues are the first to demonstrate the role of NGF in aging gastropathy, and their work highlights a key mechanism of angiogenesis during gastric repair that may inform future therapeutic strategies.
Amy Christine Engevik, PhD, is a postdoctoral fellow in the division of surgical sciences at Vanderbilt University Medical Center, Nashville, Tenn. She has no conflicts of interest.
a recent study found.
Compared with young individuals, elderly people have significantly lower levels of NGF in gastric endothelial cells (GECs), a finding that is associated with impaired angiogenesis and delayed gastric ulcer healing.
“Our previous studies have shown that the gastric mucosa of aging individuals ... has increased susceptibility to injury and delayed healing owing to impaired angiogenesis, but the mechanisms are not fully elucidated,” wrote Amrita Ahluwalia, PhD, of Medical and Research Services at the Veterans Affairs Long Beach (Calif.) Healthcare System and her coauthors.
Mapping the drivers of angiogenesis in the gastric mucosa could lead to treatment options for elderly patients with injured or ulcerated gastric tissue. In prior trials (with rats), “treatment with VEGF [vascular endothelial growth factor] only partly reversed impaired angiogenesis in aging [GECs], indicating an essential role for other factor(s) in addition to VEGF,” the investigators wrote in the September issue of Cellular and Molecular Gastroenterology and Hepatology. They looked to NGF as another possible factor because recent studies had shown it could improve angiogenesis in the brain.
The present study measured NGF expression in rats and humans of varying ages, with NGF treatment and gene therapy performed in rats (in vitro and in vivo).
In vitro angiogenesis was 4.1-fold lower in GECs from aging rats (24 months of age) than it was in GECs from young rats (3 months of age; P less than .001). NGF protein and NGF mRNA levels were also significantly lower in aging GECs than they were with young GECs (NGF protein, 3.0-fold lower; NGF mRNA, 4.2-fold lower; P less than .001).
Treatment of aging rat GECs with exogenous NGF increased angiogenesis by 1.5-fold (P less than .001). Pretreatment with a PI3 kinase inhibitor or an mTOR inhibitor abolished this improvement, suggesting that the PI3 kinase/Akt and mTOR pathways are involved.
When NGF gene therapy was performed in aging GECs, NGF levels rose to the level of that in young GECs, with an accompanying restoration of angiogenesis (threefold increase; P less than .001). Proliferation of aging GECs also increased with gene therapy (P less than .001).
In vivo studies revealed that NGF expression and cell proliferation in aging rat gastric mucosa were lower than in younger rats. Of note, older rats treated with local NGF protein showed increased gastric mucosa angiogenesis and faster ulcer healing, compared with phosphate-buffered saline treatment.
Similar age-related NGF declines were found in humans. When gastric mucosa biopsies were collected from younger individuals (younger than 40 years old; n = 10) and compared with samples from an older population (at least 70 years old; n = 10), the investigators found that NGF expression was 5.5-fold lower in the older people (P less than .001).
“This clearly indicates human relevance of our experimental findings and also can explain impaired angiogenesis and delayed healing of injured gastric mucosa in aging individuals,” the investigators wrote.
“Aging gastropathy and its consequences are clinically critical issues,” the investigators noted, “especially because the aging U.S. population is growing rapidly and it is estimated that, by the year 2030, approximately 70 million Americans will be older than 65 years of age.” Gastric ulcers become more common with age, and individuals 70 years or older have an eightfold increased risk of associated complications, compared with people under 50 years.
The investigators noted that multiple growth factors likely play a role in stimulation of angiogenesis, including NGF, VEGF, epidermal growth factor, and basic fibroblast growth factor. “Further studies are necessary to investigate the role of other growth factors and cytokines in angiogenesis, [gastric ulcer] healing, and their impairment in aging,” the investigators concluded.
The study was funded by the Department of Veterans Affairs Biomedical Laboratory Research and Development Service. The authors declared no conflicts of interest.
SOURCE: Ahluwalia A et al. CMGH. 2018 May 17. doi: 10.1016/j.jcmgh.2018.05.003
a recent study found.
Compared with young individuals, elderly people have significantly lower levels of NGF in gastric endothelial cells (GECs), a finding that is associated with impaired angiogenesis and delayed gastric ulcer healing.
“Our previous studies have shown that the gastric mucosa of aging individuals ... has increased susceptibility to injury and delayed healing owing to impaired angiogenesis, but the mechanisms are not fully elucidated,” wrote Amrita Ahluwalia, PhD, of Medical and Research Services at the Veterans Affairs Long Beach (Calif.) Healthcare System and her coauthors.
Mapping the drivers of angiogenesis in the gastric mucosa could lead to treatment options for elderly patients with injured or ulcerated gastric tissue. In prior trials (with rats), “treatment with VEGF [vascular endothelial growth factor] only partly reversed impaired angiogenesis in aging [GECs], indicating an essential role for other factor(s) in addition to VEGF,” the investigators wrote in the September issue of Cellular and Molecular Gastroenterology and Hepatology. They looked to NGF as another possible factor because recent studies had shown it could improve angiogenesis in the brain.
The present study measured NGF expression in rats and humans of varying ages, with NGF treatment and gene therapy performed in rats (in vitro and in vivo).
In vitro angiogenesis was 4.1-fold lower in GECs from aging rats (24 months of age) than it was in GECs from young rats (3 months of age; P less than .001). NGF protein and NGF mRNA levels were also significantly lower in aging GECs than they were with young GECs (NGF protein, 3.0-fold lower; NGF mRNA, 4.2-fold lower; P less than .001).
Treatment of aging rat GECs with exogenous NGF increased angiogenesis by 1.5-fold (P less than .001). Pretreatment with a PI3 kinase inhibitor or an mTOR inhibitor abolished this improvement, suggesting that the PI3 kinase/Akt and mTOR pathways are involved.
When NGF gene therapy was performed in aging GECs, NGF levels rose to the level of that in young GECs, with an accompanying restoration of angiogenesis (threefold increase; P less than .001). Proliferation of aging GECs also increased with gene therapy (P less than .001).
In vivo studies revealed that NGF expression and cell proliferation in aging rat gastric mucosa were lower than in younger rats. Of note, older rats treated with local NGF protein showed increased gastric mucosa angiogenesis and faster ulcer healing, compared with phosphate-buffered saline treatment.
Similar age-related NGF declines were found in humans. When gastric mucosa biopsies were collected from younger individuals (younger than 40 years old; n = 10) and compared with samples from an older population (at least 70 years old; n = 10), the investigators found that NGF expression was 5.5-fold lower in the older people (P less than .001).
“This clearly indicates human relevance of our experimental findings and also can explain impaired angiogenesis and delayed healing of injured gastric mucosa in aging individuals,” the investigators wrote.
“Aging gastropathy and its consequences are clinically critical issues,” the investigators noted, “especially because the aging U.S. population is growing rapidly and it is estimated that, by the year 2030, approximately 70 million Americans will be older than 65 years of age.” Gastric ulcers become more common with age, and individuals 70 years or older have an eightfold increased risk of associated complications, compared with people under 50 years.
The investigators noted that multiple growth factors likely play a role in stimulation of angiogenesis, including NGF, VEGF, epidermal growth factor, and basic fibroblast growth factor. “Further studies are necessary to investigate the role of other growth factors and cytokines in angiogenesis, [gastric ulcer] healing, and their impairment in aging,” the investigators concluded.
The study was funded by the Department of Veterans Affairs Biomedical Laboratory Research and Development Service. The authors declared no conflicts of interest.
SOURCE: Ahluwalia A et al. CMGH. 2018 May 17. doi: 10.1016/j.jcmgh.2018.05.003
FROM CELLULAR AND MOLECULAR GASTROENTEROLOGY AND HEPATOLOGY
Key clinical point: Nerve growth factor (NGF) therapy in aging rats improves angiogenesis and speeds gastric ulcer (GU) healing, suggesting possible applications in human medicine.
Major finding: Compared with young individuals, elderly people have 5.5-fold lower NGF expression in their gastric mucosa.
Study details: A prospective study involving rats and humans, with NGF therapy performed in rats.
Disclosures: The study was funded by the Department of Veterans Affairs Biomedical Laboratory Research and Development Service. The authors declared no conflicts of interest.
Source: Ahluwalia et al. CMGH. 2018 May 17. doi: 10.1016/j.jcmgh.2018.05.003
Study examines POEM learning curve
Technical failures or adverse events complicated 4% of peroral endoscopic myotomies (POEMs) in a large single-center retrospective study.
Individual predictors of this composite negative outcome included case number, full-thickness myotomy, and procedure time, Zuqiang Liu, PhD, and his associates at Fudan University, Shanghai, China, wrote in the September issue of Clinical Gastroenterology and Hepatology.
After controlling for these risk factors, the composite rate of adverse events and technical failures dropped gradually after an endoscopist had performed his or her first 100 cases, according to the researchers. “Technical proficiency, demonstrated by plateauing of the procedure time, could be achieved after 70 cases,” they wrote. “The volume of cases required to manage challenging situations and prevent adverse events was thus higher than that needed for simple technical proficiency.” The experience of the training surgeon helped trainees gain technical proficiency faster, they added.
Peroral endoscopic myotomy (POEM) is minimally invasive and effectively treats spastic esophageal motility disorders. However, it is also a challenging procedure, and little is known about its learning curve. For the study, the researchers retrospectively reviewed technical failures and adverse events among 1,346 POEMs performed for achalasia at a single hospital in China between August 2010 and July 2015. They also assessed procedure time and a secondary composite outcome consisting of technical failure, adverse events, and clinical failure (further symptoms) for the first 192 cases performed by the original training surgeon.
There were 10 technical failures and 44 adverse events affecting a total of 54 patients (4%). Case number (P = .010), full-thickness myotomy (P = .002), and procedure time (P = .001) independently predicted this primary composite outcome. Adjusted cumulative sum analysis showed that the rate of this composite outcome decreased gradually after a surgeon had performed his or her first 100 cases. “The procedure time was high during the first few cases and decreased after endoscopists performed 70 cases,” indicating technical proficiency, the investigators wrote. The rate of the secondary composite outcome also fell gradually after the primary surgeon had performed between 90 and 100 cases.
For the first 192 cases performed by the lead surgeon, postprocedural follow-up time was typically 59 months, with a range of 3-71 months. Clinical failures occurred in 20 cases (10%). Rates of clinical failure were 6% at 1 year, 8% at 2 years, and 10% at 3 years.
This is the first study and the largest POEM database so far to assess the learning curve for this procedure by evaluating adverse events and clinical and technical failure, said the researchers. Previous studies consisted of small cases, usually of less than 100 patients each, they added. Such studies would inherently be biased because the smaller the caseload, the longer it might take for the learning curves of surgeons to plateau, they added.
Funders included the National Natural Science Foundation of China, the Major Project of Shanghai Municipal Science and Technology Committee, the Chen Guang Program of Shanghai Municipal Education Commission, and the Outstanding Young Doctor Training Project of Shanghai Municipal Commission of Health and Family Planning. The investigators reported having no relevant conflicts of interest.
SOURCE: Zuqiang L et al. Clin Gastroenterol Hepatol. 2017 Dec 5. doi: 10.1016/j.cgh.2017.11.048.
Determining competency in endoscopic procedures has been a vexing challenge since the introduction of flexible endoscopy. Traditionally, procedure volume has been used as a surrogate for technical competence. However, each endoscopist has their own learning curve. Furthermore, that curve is influenced by both the endoscopist and the characteristics of each patient. Thus, relying on procedure volume or length of time are likely inadequate markers of the true learning process. It has become more important to rely on more sophisticated measurements of competence, as illustrated in this study by Liu et al.
By using a large database of patients undergoing POEM, the authors applied risk-adjusted cumulative sum and moving averages (CUSUM) analysis to develop individual learning curves of six training endoscopists. The primary outcomes used to develop the curve were technical failure and adverse effects (likely the two outcomes patients are most concerned about). The analysis was adjusted for case complexity as well, reflecting that not all training episodes are the same. The results reveal that, although trainee endoscopists were able to perform POEM “quickly” by 70 cases, they did not achieve the more important primary outcomes of technical success and low adverse events until at least 100 procedures. This is akin to the difference between getting to the cecum quickly and having a high adenoma detection rate in colonoscopy.
Moving forward, using sophisticated measurement of individual endoscopists’ learning curves will allow maximal effectiveness of routine procedures such as colonoscopy.
Kal Patel, MD, is associate professor of medicine, Baylor College of Medicine, Houston. He has no conflicts of interest.
Determining competency in endoscopic procedures has been a vexing challenge since the introduction of flexible endoscopy. Traditionally, procedure volume has been used as a surrogate for technical competence. However, each endoscopist has their own learning curve. Furthermore, that curve is influenced by both the endoscopist and the characteristics of each patient. Thus, relying on procedure volume or length of time are likely inadequate markers of the true learning process. It has become more important to rely on more sophisticated measurements of competence, as illustrated in this study by Liu et al.
By using a large database of patients undergoing POEM, the authors applied risk-adjusted cumulative sum and moving averages (CUSUM) analysis to develop individual learning curves of six training endoscopists. The primary outcomes used to develop the curve were technical failure and adverse effects (likely the two outcomes patients are most concerned about). The analysis was adjusted for case complexity as well, reflecting that not all training episodes are the same. The results reveal that, although trainee endoscopists were able to perform POEM “quickly” by 70 cases, they did not achieve the more important primary outcomes of technical success and low adverse events until at least 100 procedures. This is akin to the difference between getting to the cecum quickly and having a high adenoma detection rate in colonoscopy.
Moving forward, using sophisticated measurement of individual endoscopists’ learning curves will allow maximal effectiveness of routine procedures such as colonoscopy.
Kal Patel, MD, is associate professor of medicine, Baylor College of Medicine, Houston. He has no conflicts of interest.
Determining competency in endoscopic procedures has been a vexing challenge since the introduction of flexible endoscopy. Traditionally, procedure volume has been used as a surrogate for technical competence. However, each endoscopist has their own learning curve. Furthermore, that curve is influenced by both the endoscopist and the characteristics of each patient. Thus, relying on procedure volume or length of time are likely inadequate markers of the true learning process. It has become more important to rely on more sophisticated measurements of competence, as illustrated in this study by Liu et al.
By using a large database of patients undergoing POEM, the authors applied risk-adjusted cumulative sum and moving averages (CUSUM) analysis to develop individual learning curves of six training endoscopists. The primary outcomes used to develop the curve were technical failure and adverse effects (likely the two outcomes patients are most concerned about). The analysis was adjusted for case complexity as well, reflecting that not all training episodes are the same. The results reveal that, although trainee endoscopists were able to perform POEM “quickly” by 70 cases, they did not achieve the more important primary outcomes of technical success and low adverse events until at least 100 procedures. This is akin to the difference between getting to the cecum quickly and having a high adenoma detection rate in colonoscopy.
Moving forward, using sophisticated measurement of individual endoscopists’ learning curves will allow maximal effectiveness of routine procedures such as colonoscopy.
Kal Patel, MD, is associate professor of medicine, Baylor College of Medicine, Houston. He has no conflicts of interest.
Technical failures or adverse events complicated 4% of peroral endoscopic myotomies (POEMs) in a large single-center retrospective study.
Individual predictors of this composite negative outcome included case number, full-thickness myotomy, and procedure time, Zuqiang Liu, PhD, and his associates at Fudan University, Shanghai, China, wrote in the September issue of Clinical Gastroenterology and Hepatology.
After controlling for these risk factors, the composite rate of adverse events and technical failures dropped gradually after an endoscopist had performed his or her first 100 cases, according to the researchers. “Technical proficiency, demonstrated by plateauing of the procedure time, could be achieved after 70 cases,” they wrote. “The volume of cases required to manage challenging situations and prevent adverse events was thus higher than that needed for simple technical proficiency.” The experience of the training surgeon helped trainees gain technical proficiency faster, they added.
Peroral endoscopic myotomy (POEM) is minimally invasive and effectively treats spastic esophageal motility disorders. However, it is also a challenging procedure, and little is known about its learning curve. For the study, the researchers retrospectively reviewed technical failures and adverse events among 1,346 POEMs performed for achalasia at a single hospital in China between August 2010 and July 2015. They also assessed procedure time and a secondary composite outcome consisting of technical failure, adverse events, and clinical failure (further symptoms) for the first 192 cases performed by the original training surgeon.
There were 10 technical failures and 44 adverse events affecting a total of 54 patients (4%). Case number (P = .010), full-thickness myotomy (P = .002), and procedure time (P = .001) independently predicted this primary composite outcome. Adjusted cumulative sum analysis showed that the rate of this composite outcome decreased gradually after a surgeon had performed his or her first 100 cases. “The procedure time was high during the first few cases and decreased after endoscopists performed 70 cases,” indicating technical proficiency, the investigators wrote. The rate of the secondary composite outcome also fell gradually after the primary surgeon had performed between 90 and 100 cases.
For the first 192 cases performed by the lead surgeon, postprocedural follow-up time was typically 59 months, with a range of 3-71 months. Clinical failures occurred in 20 cases (10%). Rates of clinical failure were 6% at 1 year, 8% at 2 years, and 10% at 3 years.
This is the first study and the largest POEM database so far to assess the learning curve for this procedure by evaluating adverse events and clinical and technical failure, said the researchers. Previous studies consisted of small cases, usually of less than 100 patients each, they added. Such studies would inherently be biased because the smaller the caseload, the longer it might take for the learning curves of surgeons to plateau, they added.
Funders included the National Natural Science Foundation of China, the Major Project of Shanghai Municipal Science and Technology Committee, the Chen Guang Program of Shanghai Municipal Education Commission, and the Outstanding Young Doctor Training Project of Shanghai Municipal Commission of Health and Family Planning. The investigators reported having no relevant conflicts of interest.
SOURCE: Zuqiang L et al. Clin Gastroenterol Hepatol. 2017 Dec 5. doi: 10.1016/j.cgh.2017.11.048.
Technical failures or adverse events complicated 4% of peroral endoscopic myotomies (POEMs) in a large single-center retrospective study.
Individual predictors of this composite negative outcome included case number, full-thickness myotomy, and procedure time, Zuqiang Liu, PhD, and his associates at Fudan University, Shanghai, China, wrote in the September issue of Clinical Gastroenterology and Hepatology.
After controlling for these risk factors, the composite rate of adverse events and technical failures dropped gradually after an endoscopist had performed his or her first 100 cases, according to the researchers. “Technical proficiency, demonstrated by plateauing of the procedure time, could be achieved after 70 cases,” they wrote. “The volume of cases required to manage challenging situations and prevent adverse events was thus higher than that needed for simple technical proficiency.” The experience of the training surgeon helped trainees gain technical proficiency faster, they added.
Peroral endoscopic myotomy (POEM) is minimally invasive and effectively treats spastic esophageal motility disorders. However, it is also a challenging procedure, and little is known about its learning curve. For the study, the researchers retrospectively reviewed technical failures and adverse events among 1,346 POEMs performed for achalasia at a single hospital in China between August 2010 and July 2015. They also assessed procedure time and a secondary composite outcome consisting of technical failure, adverse events, and clinical failure (further symptoms) for the first 192 cases performed by the original training surgeon.
There were 10 technical failures and 44 adverse events affecting a total of 54 patients (4%). Case number (P = .010), full-thickness myotomy (P = .002), and procedure time (P = .001) independently predicted this primary composite outcome. Adjusted cumulative sum analysis showed that the rate of this composite outcome decreased gradually after a surgeon had performed his or her first 100 cases. “The procedure time was high during the first few cases and decreased after endoscopists performed 70 cases,” indicating technical proficiency, the investigators wrote. The rate of the secondary composite outcome also fell gradually after the primary surgeon had performed between 90 and 100 cases.
For the first 192 cases performed by the lead surgeon, postprocedural follow-up time was typically 59 months, with a range of 3-71 months. Clinical failures occurred in 20 cases (10%). Rates of clinical failure were 6% at 1 year, 8% at 2 years, and 10% at 3 years.
This is the first study and the largest POEM database so far to assess the learning curve for this procedure by evaluating adverse events and clinical and technical failure, said the researchers. Previous studies consisted of small cases, usually of less than 100 patients each, they added. Such studies would inherently be biased because the smaller the caseload, the longer it might take for the learning curves of surgeons to plateau, they added.
Funders included the National Natural Science Foundation of China, the Major Project of Shanghai Municipal Science and Technology Committee, the Chen Guang Program of Shanghai Municipal Education Commission, and the Outstanding Young Doctor Training Project of Shanghai Municipal Commission of Health and Family Planning. The investigators reported having no relevant conflicts of interest.
SOURCE: Zuqiang L et al. Clin Gastroenterol Hepatol. 2017 Dec 5. doi: 10.1016/j.cgh.2017.11.048.
FROM CLINICAL GASTROENTEROLOGY AND HEPATOLOGY
Key clinical point: One hundred cases was the typical learning curve for peroral endoscopic myotomy (POEM).
Major finding: The composite rate of technical failure or adverse events was 4%. An adjusted analysis showed that the rate of this negative composite outcome fell gradually after an endoscopist had performed his or her first 100 cases.
Study details: Single-center retrospective study of 1,346 patients undergoing POEM.
Disclosures: Funders included the National Natural Science Foundation of China, the Major Project of Shanghai Municipal Science and Technology Committee, the Chen Guang Program of Shanghai Municipal Education Commission, and the Outstanding Young Doctor Training Project of Shanghai Municipal Commission of Health and Family Planning. The investigators reported having no relevant conflicts of interest..
Source: Zuqiang L et al. Clin Gastroenterol Hepatol. 2017 Dec 5. doi: 10.1016/j.cgh.2017.11.048.
Even modest alcohol use may worsen NAFLD
Patients with nonalcoholic fatty liver disease who consumed modest quantities of alcohol had significantly less improvement in steatosis and significantly lower odds of resolution of nonalcoholic steatohepatitis, compared with nondrinkers, according to the results of a longitudinal cohort study published in the Clinical Gastroenterology and Hepatology.
Modest drinkers also had significantly less improvement in their AST levels, compared with nondrinkers, said Veeral Ajmera, MD, of the University of California, San Diego, and his associates. “Importantly, our results suggest that cessation of alcohol use may mitigate these changes,” they wrote. Clinicians should consider the spectrum of nonalcoholic fatty liver disease (NAFLD), and especially nonalcoholic steatohepatitis (NASH), when making recommendations about alcohol use. “More advanced NAFLD severity may warrant counseling against [even] modest alcohol use.”
More than one in three adults in the United States has NAFLD and about two-thirds drink alcohol, almost always in moderation, the researchers noted. Modest alcohol use has been linked to decreased cardiovascular risk, which is particularly relevant because patients with NAFLD tend to have risk factors for cardiovascular disease. Results from at least two cross-sectional studies also suggest modest drinkers with NAFLD have less severe histology, including less NASH and fibrosis. However, modest drinkers tend to be more physically active, with lower body mass indices, higher physical activity levels, and less obesity, which are potential confounders. To better understand the effects of modest alcohol consumption on NAFLD, the researchers studied adults with NAFLD who participated in studies conducted by the multicenter NASH Clinical Research Network.
The 285 participants were typically aged in their late 40s, female, white, and obese, with an average body mass index of 34.7 kg/m2. In all, 168 participants (59%) reported consuming up to two drinks per day, while 41% abstained from alcohol use. During an average of 47 months between biopsies (standard deviation, 26 months), nondrinkers averaged a 0.49 reduction in steatosis grade, significantly more than that of modest drinkers (reduction, 0.30; P = .04). Nondrinkers also had a greater decrease in mean AST level (7 U/L), compared with drinkers (2 U/L; P = .04).
A total of 64% of patients were classified as having definite NASH, 19% had NAFLD without NASH, and 17% had borderline NASH. At baseline, 23% of patients did not have fibrosis, 32% had stage 1 fibrosis, 21% had stage 2, 21% had stage 3, and 3% had stage 4. Modest drinkers were more likely to be white and were less likely to be diagnosed with definitive NASH at baseline. After controlling for these potential confounders, modest drinkers had significantly lower odds of NASH resolution, compared with nondrinkers (adjusted odds ratio, 0.32; 95% confidence interval, 0.11-0.92; P = .04).
“[The] presence of NASH has consistently been shown to predict increased risk for fibrosis progression, and therefore, our finding of less NASH resolution among consistent modest drinkers is clinically relevant,” the investigators wrote. “Although we were unable to assess the association between modest alcohol consumption and cardiovascular risk, we did not see any significant changes in measured metabolic risk factors with known associations with cardiovascular disease including low-density lipoprotein and high-density lipoprotein cholesterol and insulin resistance.”
Funders of the study included the National Institute of Diabetes and Digestive and Kidney Diseases, the National Center for Advancing Translational Sciences, the Advanced/Transplant Hepatology Fellowship, the American Association for the Study of Liver Diseases Foundation, and the Intramural Research Program of the National Institutes of Health.
SOURCE: Ajmera V et al. Clin Gastroenterol Hepatol. 2018 Mar 14. doi: 10.1016/j.cgh.2018.01.026.
Patients with nonalcoholic fatty liver disease who consumed modest quantities of alcohol had significantly less improvement in steatosis and significantly lower odds of resolution of nonalcoholic steatohepatitis, compared with nondrinkers, according to the results of a longitudinal cohort study published in the Clinical Gastroenterology and Hepatology.
Modest drinkers also had significantly less improvement in their AST levels, compared with nondrinkers, said Veeral Ajmera, MD, of the University of California, San Diego, and his associates. “Importantly, our results suggest that cessation of alcohol use may mitigate these changes,” they wrote. Clinicians should consider the spectrum of nonalcoholic fatty liver disease (NAFLD), and especially nonalcoholic steatohepatitis (NASH), when making recommendations about alcohol use. “More advanced NAFLD severity may warrant counseling against [even] modest alcohol use.”
More than one in three adults in the United States has NAFLD and about two-thirds drink alcohol, almost always in moderation, the researchers noted. Modest alcohol use has been linked to decreased cardiovascular risk, which is particularly relevant because patients with NAFLD tend to have risk factors for cardiovascular disease. Results from at least two cross-sectional studies also suggest modest drinkers with NAFLD have less severe histology, including less NASH and fibrosis. However, modest drinkers tend to be more physically active, with lower body mass indices, higher physical activity levels, and less obesity, which are potential confounders. To better understand the effects of modest alcohol consumption on NAFLD, the researchers studied adults with NAFLD who participated in studies conducted by the multicenter NASH Clinical Research Network.
The 285 participants were typically aged in their late 40s, female, white, and obese, with an average body mass index of 34.7 kg/m2. In all, 168 participants (59%) reported consuming up to two drinks per day, while 41% abstained from alcohol use. During an average of 47 months between biopsies (standard deviation, 26 months), nondrinkers averaged a 0.49 reduction in steatosis grade, significantly more than that of modest drinkers (reduction, 0.30; P = .04). Nondrinkers also had a greater decrease in mean AST level (7 U/L), compared with drinkers (2 U/L; P = .04).
A total of 64% of patients were classified as having definite NASH, 19% had NAFLD without NASH, and 17% had borderline NASH. At baseline, 23% of patients did not have fibrosis, 32% had stage 1 fibrosis, 21% had stage 2, 21% had stage 3, and 3% had stage 4. Modest drinkers were more likely to be white and were less likely to be diagnosed with definitive NASH at baseline. After controlling for these potential confounders, modest drinkers had significantly lower odds of NASH resolution, compared with nondrinkers (adjusted odds ratio, 0.32; 95% confidence interval, 0.11-0.92; P = .04).
“[The] presence of NASH has consistently been shown to predict increased risk for fibrosis progression, and therefore, our finding of less NASH resolution among consistent modest drinkers is clinically relevant,” the investigators wrote. “Although we were unable to assess the association between modest alcohol consumption and cardiovascular risk, we did not see any significant changes in measured metabolic risk factors with known associations with cardiovascular disease including low-density lipoprotein and high-density lipoprotein cholesterol and insulin resistance.”
Funders of the study included the National Institute of Diabetes and Digestive and Kidney Diseases, the National Center for Advancing Translational Sciences, the Advanced/Transplant Hepatology Fellowship, the American Association for the Study of Liver Diseases Foundation, and the Intramural Research Program of the National Institutes of Health.
SOURCE: Ajmera V et al. Clin Gastroenterol Hepatol. 2018 Mar 14. doi: 10.1016/j.cgh.2018.01.026.
Patients with nonalcoholic fatty liver disease who consumed modest quantities of alcohol had significantly less improvement in steatosis and significantly lower odds of resolution of nonalcoholic steatohepatitis, compared with nondrinkers, according to the results of a longitudinal cohort study published in the Clinical Gastroenterology and Hepatology.
Modest drinkers also had significantly less improvement in their AST levels, compared with nondrinkers, said Veeral Ajmera, MD, of the University of California, San Diego, and his associates. “Importantly, our results suggest that cessation of alcohol use may mitigate these changes,” they wrote. Clinicians should consider the spectrum of nonalcoholic fatty liver disease (NAFLD), and especially nonalcoholic steatohepatitis (NASH), when making recommendations about alcohol use. “More advanced NAFLD severity may warrant counseling against [even] modest alcohol use.”
More than one in three adults in the United States has NAFLD and about two-thirds drink alcohol, almost always in moderation, the researchers noted. Modest alcohol use has been linked to decreased cardiovascular risk, which is particularly relevant because patients with NAFLD tend to have risk factors for cardiovascular disease. Results from at least two cross-sectional studies also suggest modest drinkers with NAFLD have less severe histology, including less NASH and fibrosis. However, modest drinkers tend to be more physically active, with lower body mass indices, higher physical activity levels, and less obesity, which are potential confounders. To better understand the effects of modest alcohol consumption on NAFLD, the researchers studied adults with NAFLD who participated in studies conducted by the multicenter NASH Clinical Research Network.
The 285 participants were typically aged in their late 40s, female, white, and obese, with an average body mass index of 34.7 kg/m2. In all, 168 participants (59%) reported consuming up to two drinks per day, while 41% abstained from alcohol use. During an average of 47 months between biopsies (standard deviation, 26 months), nondrinkers averaged a 0.49 reduction in steatosis grade, significantly more than that of modest drinkers (reduction, 0.30; P = .04). Nondrinkers also had a greater decrease in mean AST level (7 U/L), compared with drinkers (2 U/L; P = .04).
A total of 64% of patients were classified as having definite NASH, 19% had NAFLD without NASH, and 17% had borderline NASH. At baseline, 23% of patients did not have fibrosis, 32% had stage 1 fibrosis, 21% had stage 2, 21% had stage 3, and 3% had stage 4. Modest drinkers were more likely to be white and were less likely to be diagnosed with definitive NASH at baseline. After controlling for these potential confounders, modest drinkers had significantly lower odds of NASH resolution, compared with nondrinkers (adjusted odds ratio, 0.32; 95% confidence interval, 0.11-0.92; P = .04).
“[The] presence of NASH has consistently been shown to predict increased risk for fibrosis progression, and therefore, our finding of less NASH resolution among consistent modest drinkers is clinically relevant,” the investigators wrote. “Although we were unable to assess the association between modest alcohol consumption and cardiovascular risk, we did not see any significant changes in measured metabolic risk factors with known associations with cardiovascular disease including low-density lipoprotein and high-density lipoprotein cholesterol and insulin resistance.”
Funders of the study included the National Institute of Diabetes and Digestive and Kidney Diseases, the National Center for Advancing Translational Sciences, the Advanced/Transplant Hepatology Fellowship, the American Association for the Study of Liver Diseases Foundation, and the Intramural Research Program of the National Institutes of Health.
SOURCE: Ajmera V et al. Clin Gastroenterol Hepatol. 2018 Mar 14. doi: 10.1016/j.cgh.2018.01.026.
FROM CLINICAL GASTROENTEROLOGY AND HEPATOLOGY
Key clinical point: Consider counseling patients with more advanced nonalcoholic fatty liver disease to avoid alcohol.
Major finding: Compared with nondrinkers, patients who reported modest alcohol use had significantly less improvement in steatosis and significantly lower odds of resolution of nonalcoholic steatohepatitis (P = .04 for both comparisons).
Study details: A longitudinal cohort study of 285 adults with nonalcoholic fatty liver disease with paired biopsy specimens obtained an average of 4 years apart.
Disclosures: Funders of the study included the National Institute of Diabetes and Digestive and Kidney Diseases, the National Center for Advancing Translational Sciences, the Advanced/Transplant Hepatology Fellowship, the American Association for the Study of Liver Diseases Foundation, and the Intramural Research Program of the National Institutes of Health.
Source: Ajmera V et al. Clin Gastroenterol Hepatol. 2018 Mar 14. doi: 10.1016/j.cgh.2018.01.026.
Pancreatic surveillance identified resectable cancers
Long-term pancreatic surveillance of high-risk patients identified cancers while they were still resectable, and 85% of such patients remained alive 3 years after diagnosis, researchers reported.
“Among individuals undergoing pancreatic surveillance, specific detectable lesions with worrisome features predicted neoplastic progression. The short-term outcomes of patients with screening-detected PDACs [pancreatic ductal adenocarcinomas] improved,” wrote Marcia Irene Canto, MD, MHS, of the Johns Hopkins University, Baltimore, together with her associates in the September issue of Gastroenterology.
The lifetime risk of PDAC is about 1.5%, the researchers noted. Consequently, the U.S. Preventive Services Task Force does not recommend pancreatic surveillance at a population level. However, pancreatic screening is being evaluated for individuals who are at significantly elevated risk of PDAC, including those with at least two first-degree relatives with PDAC or who have germline mutations in BRCA1, BRCA2, PALB2, PRSS1 (hereditary pancreatitis), CDKN2A, MLH1, MSH2, MSH6, PMS2 (Lynch syndrome), or STK11 (Peutz-Jeghers syndrome).
For the study, Dr. Canto and her associates analyzed data from 354 such high-risk individuals enrolled in the CAPS (Cancer of the Pancreas Screening) cohort studies, which were conducted at tertiary care academic centers during 1998-2014. All participants underwent endoscopic ultrasound at baseline, followed by surveillance with endoscopic ultrasound, magnetic resonance imaging, computed tomography, or some combination of these modalities. Patients who developed pancreatic cancer or high-grade dysplasia were offered surgery.
In all, 68 patients (19%) developed pancreatic lesions with worrisome features, such as solid masses, multiple cysts, mural nodules, thickened or enhancing walls, cysts exceeding 3 cm in size or that grew more than 4 mm annually, a greater than 5-mm dilation of the main pancreatic duct, or an abrupt change in duct caliber. The lesions developed over a median of 13.1 months (interquartile range, 0.2-52 months).
A total of 7% of patients had neoplastic progression, including 14 cases of PDAC and 10 cases of high-grade dysplasia. Median times from baseline to detection of PDAC were 4.8 years overall (IQR, 1.6-6.9 years), 1.7 years (IQR, 0.5-4.4 years) among patients aged at least 60 years at baseline, and 5.2 years among younger patients (IQR, 0.4-8 years). Patients developed PDAC at a median of 67 years old.
Among 10 PDACs detected by surveillance, 9 were resectable. Three patients subsequently died of PDAC, while one patient died of complications of gastric cancer surgery. However, 85% of patients survived for at least 3 years after surgical resection of PDAC. The remaining four cases of PDAC were detected outside surveillance or after patients stopped surveillance.
The 10 cases of high-grade dysplasia consisted of intraductal papillary mucinous neoplasm with high-grade dysplasia or high-grade pancreatic intraepithelial neoplasia. Patients whose PDAC or high-grade dysplasia was detected by surveillance survived a median of 5.3 years, while patients whose surveillance was late or stopped and who subsequently developed neoplasia survived a median of only 1.4 years after diagnosis (P less than .0001).
Funders included the Pancreatic Cancer Action Network, Lustgarten Foundation for Pancreatic Cancer Research, the John and Peter Hooven Memorial Endowment, Hugh and Rachel Victor, and ChiRhoClin. Dr. Canto had no disclosures. Three coinvestigators disclosed royalties for licensing of PALB2 as a pancreatic cancer susceptibility gene.
SOURCE: Canto MI et al. Gastroenterology. 2018 May 24. doi: 10.1053/j.gastro.2018.05.035
Long-term pancreatic surveillance of high-risk patients identified cancers while they were still resectable, and 85% of such patients remained alive 3 years after diagnosis, researchers reported.
“Among individuals undergoing pancreatic surveillance, specific detectable lesions with worrisome features predicted neoplastic progression. The short-term outcomes of patients with screening-detected PDACs [pancreatic ductal adenocarcinomas] improved,” wrote Marcia Irene Canto, MD, MHS, of the Johns Hopkins University, Baltimore, together with her associates in the September issue of Gastroenterology.
The lifetime risk of PDAC is about 1.5%, the researchers noted. Consequently, the U.S. Preventive Services Task Force does not recommend pancreatic surveillance at a population level. However, pancreatic screening is being evaluated for individuals who are at significantly elevated risk of PDAC, including those with at least two first-degree relatives with PDAC or who have germline mutations in BRCA1, BRCA2, PALB2, PRSS1 (hereditary pancreatitis), CDKN2A, MLH1, MSH2, MSH6, PMS2 (Lynch syndrome), or STK11 (Peutz-Jeghers syndrome).
For the study, Dr. Canto and her associates analyzed data from 354 such high-risk individuals enrolled in the CAPS (Cancer of the Pancreas Screening) cohort studies, which were conducted at tertiary care academic centers during 1998-2014. All participants underwent endoscopic ultrasound at baseline, followed by surveillance with endoscopic ultrasound, magnetic resonance imaging, computed tomography, or some combination of these modalities. Patients who developed pancreatic cancer or high-grade dysplasia were offered surgery.
In all, 68 patients (19%) developed pancreatic lesions with worrisome features, such as solid masses, multiple cysts, mural nodules, thickened or enhancing walls, cysts exceeding 3 cm in size or that grew more than 4 mm annually, a greater than 5-mm dilation of the main pancreatic duct, or an abrupt change in duct caliber. The lesions developed over a median of 13.1 months (interquartile range, 0.2-52 months).
A total of 7% of patients had neoplastic progression, including 14 cases of PDAC and 10 cases of high-grade dysplasia. Median times from baseline to detection of PDAC were 4.8 years overall (IQR, 1.6-6.9 years), 1.7 years (IQR, 0.5-4.4 years) among patients aged at least 60 years at baseline, and 5.2 years among younger patients (IQR, 0.4-8 years). Patients developed PDAC at a median of 67 years old.
Among 10 PDACs detected by surveillance, 9 were resectable. Three patients subsequently died of PDAC, while one patient died of complications of gastric cancer surgery. However, 85% of patients survived for at least 3 years after surgical resection of PDAC. The remaining four cases of PDAC were detected outside surveillance or after patients stopped surveillance.
The 10 cases of high-grade dysplasia consisted of intraductal papillary mucinous neoplasm with high-grade dysplasia or high-grade pancreatic intraepithelial neoplasia. Patients whose PDAC or high-grade dysplasia was detected by surveillance survived a median of 5.3 years, while patients whose surveillance was late or stopped and who subsequently developed neoplasia survived a median of only 1.4 years after diagnosis (P less than .0001).
Funders included the Pancreatic Cancer Action Network, Lustgarten Foundation for Pancreatic Cancer Research, the John and Peter Hooven Memorial Endowment, Hugh and Rachel Victor, and ChiRhoClin. Dr. Canto had no disclosures. Three coinvestigators disclosed royalties for licensing of PALB2 as a pancreatic cancer susceptibility gene.
SOURCE: Canto MI et al. Gastroenterology. 2018 May 24. doi: 10.1053/j.gastro.2018.05.035
Long-term pancreatic surveillance of high-risk patients identified cancers while they were still resectable, and 85% of such patients remained alive 3 years after diagnosis, researchers reported.
“Among individuals undergoing pancreatic surveillance, specific detectable lesions with worrisome features predicted neoplastic progression. The short-term outcomes of patients with screening-detected PDACs [pancreatic ductal adenocarcinomas] improved,” wrote Marcia Irene Canto, MD, MHS, of the Johns Hopkins University, Baltimore, together with her associates in the September issue of Gastroenterology.
The lifetime risk of PDAC is about 1.5%, the researchers noted. Consequently, the U.S. Preventive Services Task Force does not recommend pancreatic surveillance at a population level. However, pancreatic screening is being evaluated for individuals who are at significantly elevated risk of PDAC, including those with at least two first-degree relatives with PDAC or who have germline mutations in BRCA1, BRCA2, PALB2, PRSS1 (hereditary pancreatitis), CDKN2A, MLH1, MSH2, MSH6, PMS2 (Lynch syndrome), or STK11 (Peutz-Jeghers syndrome).
For the study, Dr. Canto and her associates analyzed data from 354 such high-risk individuals enrolled in the CAPS (Cancer of the Pancreas Screening) cohort studies, which were conducted at tertiary care academic centers during 1998-2014. All participants underwent endoscopic ultrasound at baseline, followed by surveillance with endoscopic ultrasound, magnetic resonance imaging, computed tomography, or some combination of these modalities. Patients who developed pancreatic cancer or high-grade dysplasia were offered surgery.
In all, 68 patients (19%) developed pancreatic lesions with worrisome features, such as solid masses, multiple cysts, mural nodules, thickened or enhancing walls, cysts exceeding 3 cm in size or that grew more than 4 mm annually, a greater than 5-mm dilation of the main pancreatic duct, or an abrupt change in duct caliber. The lesions developed over a median of 13.1 months (interquartile range, 0.2-52 months).
A total of 7% of patients had neoplastic progression, including 14 cases of PDAC and 10 cases of high-grade dysplasia. Median times from baseline to detection of PDAC were 4.8 years overall (IQR, 1.6-6.9 years), 1.7 years (IQR, 0.5-4.4 years) among patients aged at least 60 years at baseline, and 5.2 years among younger patients (IQR, 0.4-8 years). Patients developed PDAC at a median of 67 years old.
Among 10 PDACs detected by surveillance, 9 were resectable. Three patients subsequently died of PDAC, while one patient died of complications of gastric cancer surgery. However, 85% of patients survived for at least 3 years after surgical resection of PDAC. The remaining four cases of PDAC were detected outside surveillance or after patients stopped surveillance.
The 10 cases of high-grade dysplasia consisted of intraductal papillary mucinous neoplasm with high-grade dysplasia or high-grade pancreatic intraepithelial neoplasia. Patients whose PDAC or high-grade dysplasia was detected by surveillance survived a median of 5.3 years, while patients whose surveillance was late or stopped and who subsequently developed neoplasia survived a median of only 1.4 years after diagnosis (P less than .0001).
Funders included the Pancreatic Cancer Action Network, Lustgarten Foundation for Pancreatic Cancer Research, the John and Peter Hooven Memorial Endowment, Hugh and Rachel Victor, and ChiRhoClin. Dr. Canto had no disclosures. Three coinvestigators disclosed royalties for licensing of PALB2 as a pancreatic cancer susceptibility gene.
SOURCE: Canto MI et al. Gastroenterology. 2018 May 24. doi: 10.1053/j.gastro.2018.05.035
FROM GASTROENTEROLOGY
Key clinical point: Pancreatic surveillance of high-risk individuals identified neoplasias when they were still resectable.
Major finding: Nine of ten tumors detected by surveillance were resectable, and 85% of patients remained alive 3 years after surgery, versus 25% of patients who were diagnosed after stopping or delaying surveillance (P less than .001).
Study details: Prospective cohort study of 354 high-risk individuals with 16 years of follow-up.
Disclosures: Funders included the Pancreatic Cancer Action Network, Lustgarten Foundation for Pancreatic Cancer Research, the John and Peter Hooven Memorial Endowment, Hugh and Rachel Victor, and ChiRhoClin. Dr. Canto had no disclosures. Three coinvestigators disclosed royalties for licensing of PALB2 as a pancreatic cancer susceptibility gene.
Source: Canto MI et al. Gastroenterology. 2018 May 24. doi: 10.1053/j.gastro.2018.05.035.