User login
When War Follows Combat Veterans Home
› Ask, “Have you or a loved one ever served in the military?” as a way to uncover service-related concerns. C
› Conduct a thorough neurological evaluation with suspected mild traumatic brain injury, including vestibular, vision, postural, and neuro-cognitive assessments. C
› Use the Post-Traumatic Checklist–Military to assess individuals with possible post-traumatic stress disorder. C
Strength of recommendation (SOR)
A Good-quality patient-oriented evidence
B Inconsistent or limited-quality patient-oriented evidence
C Consensus, usual practice, opinion, disease-oriented evidence, case series
CASE A 37-year-old white woman presents for an employment physical. Your nurse reports that she also has a complaint of headaches, that she scored an 8 on the Alcohol Use Disorders identification Test-consumption (AUDiT-c), and that the result on her patient health Questionnaire (phQ-2) suggests a depressive disorder. You ask the patient whether she has served in the military and discover that, in the last 4 years, she served 2 year-long tours in Afghanistan with her Army reserve unit, returning home 6 months ago. Since her return, she has lost her job due to chronic tardiness (sleeping through her alarm, she says) and admits she has “started drinking again.” Her visit with you this day is only to undergo the physical exam required by her new employer. What are your next steps with this patient? What resources can you use to help her?
As long as human beings have engaged in combat, there have often been extraordinarily damaging psychiatric1 injuries among those who survive. Combat survivability today is 84% to 90%, the highest in the history of armed conflict,2,3 thanks to improvements in personal protective gear, vehicle armor, rapid casualty evacuation, and surgical resuscitation and stabilization that is “far forward” on the battlefield. These survivors are subsequently at high risk for a host of other medical conditions, which commonly include traumatic brain injury (TBI), post-traumatic stress disorder (PTSD), depression, suicide, and substance abuse.4-8
Family physicians—both civilian and uniformed—may be the first to encounter these individuals. Of the more than 2.4 million US service members who have been deployed to Afghanistan or Iraq in support of Operation Enduring Freedom (OEF) or Operation Iraqi Freedom (OIF), nearly 60% are no longer on active duty.
Among this group, only half receive care from the US Department of Veterans Affairs (VA).9 Despite a concerted effort on the part of the Department of Defense (DoD) and the VA to develop and distribute effective, evidenced-based treatment protocols for veterans with combat-related conditions, major gaps remain in the care provided to combat veterans.10
This article seeks to help fill that gap by providing the information you need to recognize and treat common combat-related illness, as well as resources to help improve the quality of life for veterans and their families (TABLE 1).
Initial roadblocks to care
One of the biggest challenges in treating veterans with behavioral health issues is the fact that only 23% to 40% of those with mental illness seek care.11 Among the reasons veterans have offered for avoiding behavioral health care are a fear of the stigma associated with mental illness, concern that treatment will negatively affect their career, lack of comfort with mental health professionals, and the perception that mental health treatment is a “last resort.”12 Unfortunately, efforts by the DoD leadership to overcome these inherent biases have been largely unsuccessful13 and much work is still required to see that service members get the care they need.
Due to low rates of self-reporting, effective screening is essential. With this in mind, the DoD has implemented the deployment health assessment program (DHAP), which requires service members to be screened for common conditions within 60 days of deployment, within 30 days of returning, and again at 90 to 180 days after their return.
While the long-term effects of this program are yet to be determined, results to date are promising. Since the DHAP was implemented, there has been a significant decrease in occupationally impairing mental health problems and suicidal ideation requiring medical evacuation from a combat theater.14
FPs should begin with a simple question. Many of the 20+ million veterans living in the United States will not be wearing a uniform when they enter your office. Simply asking all of your patients, “Have you or a loved one ever served in the military?” may help you discover service-related questions or concerns.15,16 Underscoring the importance of such screening is the recent decision by the American Academy of Family Physicians to partner with First Lady Michelle Obama and Dr. Jill Biden in a new campaign called “Joining Forces,” which aims to support veterans and their families.16
Mild traumatic brain injury: Common—though overlooked
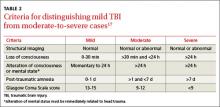
A TBI is any temporary or permanent neurologic dysfunction after a blow to the head.10,17 TBI is classified based on severity and mechanism (direct blow to the head or exposure to blast waves). Mild TBI (mTBI) is commonly referred to as a concussion and usually is not associated with loss of consciousness or altered mental status. Brain imaging results are also normal with mTBI. Severe TBI, on the other hand, is associated with prolonged loss of consciousness, altered mental status, and abnormal brain imaging results (TABLE 2).17
A unique obstacle to accurate evaluation in the field. It is important to emphasize that mTBI is a clinical diagnosis, and its detection requires honest patient communication. This can be problematic with motivated soldiers who are anxious to continue the mission and fear that any admission of symptoms might delay a return to their unit. As with a concussed athlete eager to return to the field of play, the clinical diagnosis of mTBI requires a high index of clinical suspicion and constant vigilance by the health care provider. Despite being the most common combat- related injury, mTBI is often overlooked due to the absence of obvious physical injuries.4 Recent data suggest that 28% to 60% of ser- vice members evacuated from combat have a TBI. Most of these injuries (77%) are mTBI.18-20 Improved personal protective equipment (including Kevlar helmets and body armor) and the high number of blast-related injuries are likely responsible for the high incidence of mTBI among OEF/OIF veterans.8,21,22 The prevalence of mTBI among service members not evacuated is estimated to be 20% to 30%.20
Symptoms can persist. Most patients with mTBI completely recover within 30 days of the injury. Unfortunately, 10% to 15% of mTBI patients develop chronic problems lasting months to years.4 Residual symptoms most commonly include headache, irritability, depression, sleep disturbance, impaired reasoning, memory problems, and difficulty concentrating. These symptoms are not unique to mTBI and overlap with comorbid combat diagnoses like PTSD, depression, and sleep deprivation.10 The following tools can help physicians determine whether mTBI is present.
Checking for possible mtBi. In the field, patients with possible mTBI can be screened rapidly using the Military Acute Concussion Evaluation (MACE, found at www.dvbic.org), a modification of the validated and widely used Sideline Assessment of Concussion (SAC) tool. More challenging is evaluating potential mTBI patients who present weeks or months after a traumatic event, for which there are no simple confirmatory tests. In this event, conduct a thorough neurological evaluation that includes vestibular, vision, postural, and neurocognitive assessments. For patients with persistent symptoms or possible anatomic brain abnormalities, magnetic resonance imaging (MRI) is the imaging modality of choice. Patients with complications or a questionable diagnosis are best managed in consultation with a neurologist.
Initial treatment of mtBi is symptom-based. When practical, try nonpharmacologic interventions first (TABLE 3).10 In particular, have the patient avoid further high-risk exposures that could lead to second impact syndrome (an issue increasingly recognized in contact sports). Also critical are physical and cognitive rest and the restoration of sleep until the patient is completely asymptomatic.
If the patient exhibits irritability and depression, selective serotonin reuptake inhibitors (SSRIs) are first-line treatment. Avoid narcotics and sedative-hypnotic sleep medications if treating comorbidities such as pain and sleep deprivation. The VA/DoD guideline on managing concussion and mTBI provides additional detailed, evidence-based treatment recommendations.17
Reliving the horror again and again: PTSD
PTSD is a persistent and, at times, debilitating clinical syndrome that develops after exposure to a psychologically traumatic event. It’s the second most common illness among OEF/OIF combat veterans, with an estimated prevalence of 3% to 20%, a finding consistent with prior wars.6,23-25 In the case of combat veterans, the inciting event usually involves an actual or perceived risk of death or serious injury. The individual’s response to the event involves intense fear, helplessness, or horror. The traumatic event is persistently re-experienced through intrusive and disturbing recollections or dreams that cause intense psychological distress. This, in turn, leads to a state of persistent sympathetic arousal. As symptoms are often triggered by specific cues, individuals with PTSD actively seek to avoid thoughts, situations, or stimuli associated with the event.23,26
Symptoms commonly associated with PTSD include difficulty falling or staying asleep, recurrent nightmares, hypervigilance, and an exaggerated startle response. Individuals with PTSD also have a poorer sense of well-being, a higher rate of work absenteeism, and significantly more somatic complaints than age-matched peers.27 For symptoms to be attributable to PTSD, their onset must follow a recent inciting event and must also cause clinically significant distress or impairment in social, occupational, or other areas of daily living. Common comorbid illnesses include mTBI, depression, and substance abuse. As with mTBI, the presence of multiple comorbidities in patients with PTSD can complicate evaluation, diagnosis, and treatment.
Diagnosis. PTSD is subdivided into acute (symptoms lasting more than one month but less than 3 months after the traumatic event) and chronic (symptoms lasting longer than 3 months after the traumatic event).28 The distinction of acute or chronic does not affect treatment, but it is useful information for the patient to have regarding prognosis and eventual outcome. Like mTBI, PTSD is a clinical diagnosis made only after a thorough, structured diagnostic interview. The use of a validated, self-administered checklist, such as the Post-Traumatic Checklist-Military (PCL-M), allows for an efficient review of a patient’s symptoms and a reliable way to track treatment progress (http://www.ptsd.va.gov/professional/ pages/assessments/ptsd-checklist.asp).
Treatment Options. Effective evidence-based treatments for PTSD are cognitive behavioral therapy, eye movement desensitization and reprocessing (EMDR), and pharmacotherapy. SSRIs and serotonin- norepinephrine reuptake inhibitors (SNRIs) have the strongest evidence for pharmacologic benefit in the treatment of PTSD.28,29 Other helpful medications are prazosin for nightmares and trazodone for sleep. Family physicians can use these medications as part of a patient-centered collaboration with the rest of the integrated care team, to offer the best chance for treatment success.10,28,30
Depression: Vets are reluctant to self-report
Combat experience is a significant risk factor for major depression. Estimates of the lifetime prevalence of depression in the general US population vary from 9% to 25% in women and 5% to 12% in men. By contrast, the prevalence of depression in OIF/OEF veterans ranges from 2% to 37%.24,31,32
Screening can yield false negatives. Many combat veterans are reluctant to self-report behavioral conditions, including depression. Screening, therefore, is important to identify potential depression and allow for intervention. Validated screening tools for depression include the PHQ-2 and PHQ-9, which are easy to use in the office setting. (See http://www.cqaimh.org/pdf/ tool_phq2.pdf [PHQ-2] and http://www. integration.samhsa.gov/images/res/ PHQ%20-%20Questions.pdf [PHQ-9]). Importantly, some veterans will have a negative depression screen on return from deployment, and then test positive 6 to 12 months later.24 Explanations for the early false-negative results include the excitement of being home and patients intentionally answering questions inaccurately to avoid excessive screening at their home base.11
Treatment is most effective with a combination approach. As with most cases of depression, combining psychotherapy and psychopharmacology appears to be most effective for treating depression related to combat experience.33,34 While SSRIs and SNRIs are typical first-line pharmacologic agents, combat veterans often have comorbid mTBI, PTSD, or substance abuse issues that may influence the initial choice of therapy35 (TABLE 3).10
Suicide is on the rise in the military
Historically, the incidence of suicide has been 25% lower in military personnel than in civilian peers.36 However, between 2005 and 2009, the incidence of suicide in the Marine Corps and Army almost doubled.37 While the exact reasons remain unknown, it is likely due to prolonged and repeated deployments to a combat environment.12 While the incidence of suicide has been particularly high in the Army (22 per 100,000 active-duty and reserve personnel per year), all services have been affected. In fact, since 2009, the number of suicides among active duty service members exceeds those killed in action.37
Consider all veterans to be at risk for suicide, and screen accordingly. An effective screening tool is the Columbia-Suicide Severity Rating Scale (C-SSRS), which is able to predict those most at risk for an impending suicide attempt.34 Service members identified as high risk for suicide require unhindered access to care. The VA has worked to improve access to care and provide evidence-based point-of-care treatment strategies.38 Available resources can be found in TABLE 1.
Unfortunately, even with effective screening and treatment, not all suicides can be prevented. Studies have demonstrated that approximately 65% of service members who commit suicide had no known history of communicating their suicidal intent. Sadly, 25% of service members who committed suicide had seen a mental health provider within the previous 30 days.39
Alcohol abuse is common; opioids present a unique risk
Excessive use of alcohol and recreational and prescription drugs is common among OEF/ OIF veterans, especially those with comorbid mental health disorders. Retrospective cross-sectional studies show that 11% to 20% of OEF/OIF veterans met DSM-IV-TR diagnostic criteria for substance use disorders.40-42 At highest risk are single enlisted men under the age of 24 in the Army or Marine Corps who serve in a combat-specific capacity. Interestingly, the prevalence of substance use disorders among OEF/OIF veterans closely mirrors that reported in epidemiologic studies of Vietnam veterans (11%-14%).41 This similarity, combined with the 39% lifetime prevalence of substance use disorders among Vietnam veterans, may foreshadow a similar lifetime prevalence of substance use disorders among OEF/OIF veterans.41
Most-abused substances. Alcohol is the most commonly abused substance among OEF/OIF veterans (10%-20%).40,41,43-45 Other abused substances include opioids (prescribed or illicitly obtained), synthetic marijuana (“Spice” and “K2”), and “bath salts” (synthetic stimulants) (W.M. Sauve, MD, personal communication, August 27, 2012).
OEF/OIF veterans seem to be at particular risk for developing problems related to opioid use. A 2012 retrospective cohort study showed that veterans with non–cancer- related pain diagnoses treated with opioid analgesics had an increased risk for adverse clinical outcomes compared with those not treated with opioid analgesics (9.5% vs 4.1%; relative risk [RR]=2.33; 95% confidence interval [CI], 2.20-2.46). These outcomes included traumatic accidents, overdoses, self-inflicted injuries, and injuries related to violence. This study also demonstrated that, compared with veterans without mental illness, veterans with mental illness (particularly PTSD) and non–cancer-related pain were significantly more likely to receive opioids to treat their pain and had a higher risk of adverse clinical outcomes, including overdose.46,47
Recreational use of synthetic marijuana and “bath salts” has increased in recent years. These substances are commonly labeled “not for human consumption,” which allows them to remain outside US Food and Drug Administration (FDA) regulation and be sold legally in the United States. Efforts to prohibit the sale or possession of these drugs, including the Federal Synthetic Bath Salt Ban in 2012, have fallen short, often due to creative product ”re-engineering.”33 Synthetic marijuana and stimulants are inexpensive, readily available, and perceived by users to be safe. Health care providers are often unaware that their patients are using these products. Adverse health outcomes associated with the use of these synthetic drugs include memory loss, depression, and psychosis.
These alcohol and drug screens can help
One efficient screening tool to identify veterans at risk for alcohol abuse is the AUDIT-C, developed by the World Health Organization. This brief 3-question test identifies past-year hazardous drinking and alcohol abuse or dependence with >79% sensitivity and >56% specificity in male veterans, and >66% sensitivity and >87% specificity in female veterans. These numbers are similar to those provided by the full 10-question AUDIT.48,49 The Drug Abuse Screen Test-10 (DAST-10) provides a similar screening instrument for other substances. Condensed from the original DAST-28 instrument, the DAST-10 identifies high-risk substance abuse with 74% to 94% sensitivity and 68% to 88% specificity.3
Screen for comorbidities. When you see veterans with a diagnosis of substance abuse, also evaluate for comorbid disease. Most veterans with substance use disorders (82%-93%) have at least one other mental health diagnosis (a 45% greater risk than that of civilians with substance abuse disorders),50 most commonly PTSD, depression, anxiety, and adjustment disorders.41,44,45 A number of hypotheses exist to explain the association between substance use disorders and other mental health diagnoses (“dual diagnoses”). The prevailing theory, in both veteran and civilian populations, is that substance abuse is an attempt to self-treat mental illness. Other evidence suggests that substance abuse promotes the development of mental illness, either by leading to a higher risk for traumatic experiences (increasing the chance of developing PTSD) or through a direct biochemical mechanism. Finally, con- current substance use disorder and mental illness may be due to an undefined genetic or biological vulnerability.38,44 This complicated relationship between substance abuse and behavioral health reinforces the need for screening, early diagnosis, and a comprehensive, multidisciplinary approach to treatment.
Treatment options. Office-based treatment options for narcotic and alcohol abuse and dependency are available to family physicians. Methadone has been used since the 1950s to treat opioid addiction and remains one of the mainstays of outpatient treatment.47,51 Originally, methadone was restricted to detoxification and maintenance treatment in narcotic addiction treatment programs approved by the FDA. In 1976, this restriction was lifted, and all physicians registered with the Drug Enforcement Agency (DEA) were permitted to prescribe methadone for analgesia.
In 2002, the FDA approved buprenorphine monotherapy and the combination product buprenorphine/naloxone for the treatment of opioid addiction. The prescribing of buprenorphine products requires physicians to undergo extra training, declare to the DEA their intent to prescribe buprenorphine, and obtain a special DEA identification number.52,53 Physicians interested in finding out more about buprenorphine treatment and prescribing requirements can go to the Substance Abuse and Mental Health Services Administration (SAMHSA) Web page at http://samhsa.gov.
Naltrexone is an opioid receptor agonist that is used primarily to treat alcohol dependency, and is thought to work by reducing the craving for alcohol. Multiple studies have proven the efficacy of naltrexone in an outpatient setting when used alone or in combination with psychotherapy.54,55 If you are uncomfortable or unfamiliar with the use or prescribing of these medications, referral to a substance abuse clinic specializing in dual-diagnosis treatment (TABLE 1) may offer optimal outcomes for patients with substance abuse disorders and other mental illness.
Cognitive behavioral therapy—including coping skills training, relapse prevention, contingency management, and behavioral couples’ therapy—and 12-step treatment programs are evidence-based options for the treatment of substance abuse disorders. Behavioral counseling interventions in the primary care setting (typically lasting 5-15 minutes) result in decreases in alcohol consumption, heavy drinking episodes, drinking above recommended amounts, and the number of days spent in the hospital, but have not been demonstrated to affect mortality, alcohol-related liver problems, outpatient visits, legal problems, or quality of life.56 Resources can be found at www.niaaa.nih.gov. For patients with dual diagnoses, it is not yet known whether sequential therapy (in which substance abuse is treated first, followed by treatment of the comorbid mental illness) or concurrent therapy results in better outcomes.57
CASE Your patient’s history of recent combat service, acknowledgement of employment and behavioral difficulties, and initial screening results lead you to diagnose alcoholism and depression. Additionally, she denies any suicidal ideation, but admits to experimenting with synthetic marijuana. After some discussion, she agrees to see your clinic’s social worker, and you start her on an SSri with scheduled follow-up.
CORRESPONDENCE
Shawn Kane, MD, USASoc, Attn: Surgeon (AomD), 2929 Desert Storm Drive, Ft. Bragg, NC 28310, or PO Box 3639 Pinehurst, NC 28374; [email protected]
1. Wessely S. Risk, psychiatry and the military. Br J Psychiatry. 2005;186:459-466.
2. Gawande A. Casualties of war—military care for the wounded from Iraq and Afghanistan. N Engl J Med. 2004;351:2471-2475.
3. Kotwal RS, Montgomery HR, Kotwal BM, et al. Eliminating pre- ventable death on the battlefield. Arch Surg. 2011;146:1350-1358.
4. Belanger HG, Uomoto JM, Vanderploeg RD. The Veterans Health Administration’s (VHA’s) Polytrauma System of Care for mild traumatic brain injury: costs, benefits, and controversies. J Head Trauma Rehabil. 2009;24:4-13.
5. Galarneau MR, Woodruff SI, Dye JL, et al. Traumatic brain in- jury during Operation Iraqi Freedom: findings from the United States Navy-Marine Corps Combat Trauma Registry. J Neurosurg. 2008;108:950-957.
6. Hermann BA, Shiner B, Friedman MJ. Epidemiology and preven- tion of combat-related post-traumatic stress in OEF/OIF/OND service members. Mil Med. 2012;177:1-6.
7. Uomoto JM. Best practices in veteran traumatic brain injury care. J Head Trauma Rehabil. 2012;27:241-243.
8. Warden D. Military TBI during the Iraq and Afghanistan wars. J Head Trauma Rehabil. 2006;21:398-402.
9. Taylor BC, Hagel EM, Carlson KF, et al. Prevalence and costs of co-occurring traumatic brain injury with and without psychiatric disturbance and pain among Afghanistan and Iraq War Veteran V.A. users. Med Care. 2012;50:342-346.
10. Quinlan JD, Guaron MR, Deschere BR, et al. Care of the returning veteran. Am Fam Physician. 2010;82:43-49.
11. Hoge CW, Castro CA, Messer SC, et al. Combat duty in Iraq and Afghanistan, mental health problems, and barriers to care. N Engl J Med. 2004;351:13-22.
12. Hoge CW, Castro CA. Preventing suicides in US service mem- bers and veterans: concerns after a decade of war. JAMA. 2012;308:671-672.
13. Jaffe G. New name for PTSD could mean less stigma. The Washington Post. May 5, 2012. Available at: http://articles. washingtonpost.com/2012-05-05/world/35454931_1_ptsd-post- traumatic-stress-psychiatrists. Accessed June 19, 2013.
14. Warner CH, Appenzeller GN, Parker JR, et al. Effectiveness of mental health screening and coordination of in-theater care prior to deployment to Iraq: a cohort study. Am J Psychiatry. 2011;168:378-385.
15. United States Census Bureau. Sex by age by veteran sta- tus for civilian population 18 years and over. 2010 American community survey 1-year estimates. Available at: https:// d3gqux9sl0z33u.cloudfront.net/AA/AT/gambillingonjustice- com/downloads/206273/ACS_10_1YR_B21001A.pdf. Accessed June 19, 2013.
16. American Academy of Family Physicians. Joining forces. Avail- able at: http://www.aafp.org/online/en/home/membership/ initiatives/joiningforces.html. Accessed June 19, 2013.
17. Department of Veterans Affairs and Department of Defense. Clinical Practice Guideline for Management of Concussion/Mild Traumatic Brain Injury. April 2009. Available at: http://www. healthquality.va.gov/mtbi/concussion_mtbi_full_1_0.pdf. Accessed June 19, 2013.
18. Lew HL, Poole JH, Alvarez S, et al. Soldiers with occult traumatic brain injury. Am J Phys Med Rehabil. 2005;84:393-398.
19. Marshall KR, Holland SL, Meyer KS, et al. Mild traumatic brain injury screening, diagnosis, and treatment. Mil Med. 2012;177:67- 75.
20. Terrio H, Brenner LA, Ivins BJ, et al. Traumatic brain injury screening: preliminary findings in a US Army Brigade Combat Team. J Head Trauma Rehabil. 2009;24:14-23.
21. Mossadegh S, Tai N, Midwinter M, et al. Improvised explosive de- vice related pelvi-perineal trauma: anatomic injuries and surgical management. J Trauma Acute Care Surg. 2012;73:S24-S31.
22. Okie S. Traumatic brain injury in the war zone. N Engl J Med. 2005;352:2043-2047.
23. Espinoza JM. Posttraumatic stress disorder and the perceived consequences of seeking therapy among US Army special forces operators exposed to combat. J Psychol Issues Organ Culture. 2010;1:6-28.
24. Grieger TA, Cozza SJ, Ursano RJ, et al. Posttraumatic stress dis- order and depression in battle-injured soldiers. Am J Psychiatry. 2006;163:1777-1783.
25. Hoge CW, Auchterlonie JL, Milliken CS. Mental health problems, use of mental health services, and attrition from military service after returning from deployment to Iraq or Afghanistan. JAMA. 2006;295:1023-1032.
26. Adler AB, Wright KM, Bliese PD, et al. A2 diagnostic criterion for combat-related posttraumatic stress disorder. J Trauma Stress. 2008;21:301-308.
27. Hoge CW, Terhakopian A, Castro CA, et al. Association of post- traumatic stress disorder with somatic symptoms, health care vis- its, and absenteeism among Iraq war veterans. Am J Psychiatry. 2007;164:150-153.
28. Department of Veterans Affairs and Department of Defense. Clin- ical Practice Guideline for Management of Post-Traumatic Stress. October 2010. Available at: http://www.healthquality.va.gov/ ptsd/cpg_PTSD-FULL-201011612.pdf. Accessed June 19, 2013.
29. Alexander W. Pharmacotherapy for post-traumatic stress disor- der in combat veterans: focus on antidepressants and atypical antipsychotic agents. P T. 2012;37:32-38.
30. Wisco BE, Marx BP, Keane TM. Screening, diagnosis, and treat- ment of post-traumatic stress disorder. Mil Med. 2012;177:7-13.
31. Gadermann AM, Engel CC, Naifeh JA, et al. Prevalence of DSM-IV major depression among U.S. military personnel: meta-analysis and simulation. Mil Med. 2012;177:47-59.
32. Seal KH, Shi Y, Cohen G, et al. Association of mental health dis- orders with prescription opioids and high-risk opioid use in US veterans of Iraq and Afghanistan. JAMA. 2012;307:940-947.
33. Perrone M. Many drugs remain legal after ‘bath salts’ ban. Boston. com. July 25, 2012. Available at: http://articles.boston.com/2012- 07-25/lifestyle/32850962_1_bath-salts-mdpv-synthetic-drugs. Accessed June 19, 2013.
34. Posner K, Brown GK, Stanley B, et al. The Columbia-Suicide Se- verity Rating Scale: initial validity and internal consistency find- ings from three multisite studies with adolescents and adults. Am J Psychiatry. 2011;168:1266-1277.
35. Greenberg J, Tesfazion AA, Robinson CS. Screening, diagnosis, and treatment of depression. Mil Med. 2012;177:60-66.
36. Eaton KM, Messer SC, Garvey Wilson AL, et al. Strengthening the validity of population-based suicide rate comparisons: an il- lustration using U.S. military and civilian data. Suicide Life Threat Behav. 2006;36:182-191.
37. Miller M, Azrael D, Barber C, et al. A call to link data to answer pressing questions about suicide risk among veterans. Am J Pub Health. 2012;102(suppl 1):S20-S22.
38. Department of Veterans Affairs. Report of the Blue Ribbon Work Group on suicide prevention in the veteran population. June 2008. Available at: http://www.mentalhealth.va.gov/suicide_ prevention/Blue_Ribbon_Report-FINAL_June-30-08.pdf. Accessed July 18, 2013.
39. Kinn JT, Luxton DD, Reger MA, et al. Department of Defense sui- cide event report: calendar year 2010 annual report. September 2011. Available at: http://t2health.org/sites/default/files/dodser/ DoDSER_2010_Annual_Report.pdf. Accessed June 19, 2013.
40. Fontana A, Rosenheck R. Treatment-seeking veterans of Iraq and Afghanistan: comparison with veterans of previous wars. J Nerv Ment Dis. 2008;196:513-521.
41. Seal KH, Cohen G, Waldrop A, et al. Substance use disorders in Iraq and Afghanistan veterans in VA healthcare, 2001-2010: implications for screening, diagnosis and treatment. Drug Alcohol Depend. 2011;116:93-101.
42. Mirza RA, Eick-Cost A, Otto JL. The risk of mental health disor- ders among U.S. military personnel infected with human immu- nodeficiency virus, active component, U.S. Armed Forces, 2000- 2011. MSMR. 2012;19:10-13.
43. Bohnert AS, Ilgen MA, Bossarte RM, et al. Veteran status and alco- hol use in men in the United States. Mil Med. 2012;177:198-203.
44. Erbes CR, Kaler ME, Schult T, et al. Mental health diagnosis and occupational functioning in National Guard/Reserve veterans re- turning from Iraq. J Rehabil Res Dev. 2011;48:1159-1170.
45. Stecker T, Fortney J, Owen R, et al. Co-occurring medical, psychi- atric, and alcohol-related disorders among veterans returning from Iraq and Afghanistan. Psychosomatics. 2010;51:503-507.
46. Seal KH, Shi Y, Cohen G, et al. Association of mental health dis- orders with prescription opioids and high-risk opioid use in US veterans of Iraq and Afghanistan. JAMA. 2012;307:940-947.
47. Praveen KT, Law F, O’Shea J, et al. Opioid dependence. Am Fam Physician. 2012;86:565-566.
48. Bradley KA, Bush KR, Epler AJ, et al. Two brief alcohol-screening tests from the Alcohol Use Disorders Identification Test (AUDIT): validation in a female Veterans Affairs patient population. Arch Intern Med. 2003;163:821-829.
49. Bush K, Kivlahan DR, McDonell MB, et al. The AUDIT alcohol consumption questions (AUDIT-C): an effective brief screening test for problem drinking. Ambulatory Care Quality Improvement Project (ACQUIP). Alcohol Use Disorders Identification Test. Arch Intern Med. 1998;158:1789-1795.
50. Farrell M, Howes S, Taylor C, et al. Substance misuse and psychi- atric comorbidity: an overview of the OPCS National Psychiatric Morbidity Survey. Addict Behav. 1998;23:909-918.
51. Toombs JD, Kral LA. Methadone treatment for pain states. Am Fam Physician. 2005;71:1353-1358.
52. Center for Substance Abuse Treatment. Clinical Guidelines for the Use of Buprenorphine in the Treatment of Opioid Addiction. Treatment Improvement Protocol (TIP) series 40. DHHS pub- lication (SMA) 04-3939. Rockville, MD: Substance Abuse and Mental Health Services Administration; 2004. Available at: http:// buprenorphine.samhsa.gov/Bup_Guidelines.pdf. Accessed June 19, 2013.
53. U.S.DepartmentofHealthandHumanServices,SubstanceAbuse and Mental Health Services Administration Web site. About buprenorphine therapy. Available at: http://buprenorphine. samhsa.gov/about.html. Accessed June 19, 2013.
54. Volpicelli JR, Alterman AI, Hayashida M, et al. Naltrexone in the treatment of alcohol dependence. Arch Gen Psychiatry. 1992;49:876-880.
55. O’Brien CP, Volpicelli LA, Volpicelli JR. Naltrexone in the treat- ment of alcoholism: a clinical review. Alcohol. 1996;13:35-39.
56. Jonas DE, Garbutt JC, Amick HR, et al. Behavioral counseling after screening for alcohol misuse in primary care: a systematic review and meta-analysis for the U.S. Preventive Services Task Force. Ann Intern Med. 2012;157:645-654.
57. van Dam D, Vedel E, Ehring T, et al. Psychological treatments for concurrent posttraumatic stress disorder and substance use dis- order: a systematic review. Clin Psychol Rev. 2012;32:202-214.
› Ask, “Have you or a loved one ever served in the military?” as a way to uncover service-related concerns. C
› Conduct a thorough neurological evaluation with suspected mild traumatic brain injury, including vestibular, vision, postural, and neuro-cognitive assessments. C
› Use the Post-Traumatic Checklist–Military to assess individuals with possible post-traumatic stress disorder. C
Strength of recommendation (SOR)
A Good-quality patient-oriented evidence
B Inconsistent or limited-quality patient-oriented evidence
C Consensus, usual practice, opinion, disease-oriented evidence, case series
CASE A 37-year-old white woman presents for an employment physical. Your nurse reports that she also has a complaint of headaches, that she scored an 8 on the Alcohol Use Disorders identification Test-consumption (AUDiT-c), and that the result on her patient health Questionnaire (phQ-2) suggests a depressive disorder. You ask the patient whether she has served in the military and discover that, in the last 4 years, she served 2 year-long tours in Afghanistan with her Army reserve unit, returning home 6 months ago. Since her return, she has lost her job due to chronic tardiness (sleeping through her alarm, she says) and admits she has “started drinking again.” Her visit with you this day is only to undergo the physical exam required by her new employer. What are your next steps with this patient? What resources can you use to help her?
As long as human beings have engaged in combat, there have often been extraordinarily damaging psychiatric1 injuries among those who survive. Combat survivability today is 84% to 90%, the highest in the history of armed conflict,2,3 thanks to improvements in personal protective gear, vehicle armor, rapid casualty evacuation, and surgical resuscitation and stabilization that is “far forward” on the battlefield. These survivors are subsequently at high risk for a host of other medical conditions, which commonly include traumatic brain injury (TBI), post-traumatic stress disorder (PTSD), depression, suicide, and substance abuse.4-8
Family physicians—both civilian and uniformed—may be the first to encounter these individuals. Of the more than 2.4 million US service members who have been deployed to Afghanistan or Iraq in support of Operation Enduring Freedom (OEF) or Operation Iraqi Freedom (OIF), nearly 60% are no longer on active duty.
Among this group, only half receive care from the US Department of Veterans Affairs (VA).9 Despite a concerted effort on the part of the Department of Defense (DoD) and the VA to develop and distribute effective, evidenced-based treatment protocols for veterans with combat-related conditions, major gaps remain in the care provided to combat veterans.10
This article seeks to help fill that gap by providing the information you need to recognize and treat common combat-related illness, as well as resources to help improve the quality of life for veterans and their families (TABLE 1).
Initial roadblocks to care
One of the biggest challenges in treating veterans with behavioral health issues is the fact that only 23% to 40% of those with mental illness seek care.11 Among the reasons veterans have offered for avoiding behavioral health care are a fear of the stigma associated with mental illness, concern that treatment will negatively affect their career, lack of comfort with mental health professionals, and the perception that mental health treatment is a “last resort.”12 Unfortunately, efforts by the DoD leadership to overcome these inherent biases have been largely unsuccessful13 and much work is still required to see that service members get the care they need.
Due to low rates of self-reporting, effective screening is essential. With this in mind, the DoD has implemented the deployment health assessment program (DHAP), which requires service members to be screened for common conditions within 60 days of deployment, within 30 days of returning, and again at 90 to 180 days after their return.
While the long-term effects of this program are yet to be determined, results to date are promising. Since the DHAP was implemented, there has been a significant decrease in occupationally impairing mental health problems and suicidal ideation requiring medical evacuation from a combat theater.14
FPs should begin with a simple question. Many of the 20+ million veterans living in the United States will not be wearing a uniform when they enter your office. Simply asking all of your patients, “Have you or a loved one ever served in the military?” may help you discover service-related questions or concerns.15,16 Underscoring the importance of such screening is the recent decision by the American Academy of Family Physicians to partner with First Lady Michelle Obama and Dr. Jill Biden in a new campaign called “Joining Forces,” which aims to support veterans and their families.16
Mild traumatic brain injury: Common—though overlooked
A TBI is any temporary or permanent neurologic dysfunction after a blow to the head.10,17 TBI is classified based on severity and mechanism (direct blow to the head or exposure to blast waves). Mild TBI (mTBI) is commonly referred to as a concussion and usually is not associated with loss of consciousness or altered mental status. Brain imaging results are also normal with mTBI. Severe TBI, on the other hand, is associated with prolonged loss of consciousness, altered mental status, and abnormal brain imaging results (TABLE 2).17
A unique obstacle to accurate evaluation in the field. It is important to emphasize that mTBI is a clinical diagnosis, and its detection requires honest patient communication. This can be problematic with motivated soldiers who are anxious to continue the mission and fear that any admission of symptoms might delay a return to their unit. As with a concussed athlete eager to return to the field of play, the clinical diagnosis of mTBI requires a high index of clinical suspicion and constant vigilance by the health care provider. Despite being the most common combat- related injury, mTBI is often overlooked due to the absence of obvious physical injuries.4 Recent data suggest that 28% to 60% of ser- vice members evacuated from combat have a TBI. Most of these injuries (77%) are mTBI.18-20 Improved personal protective equipment (including Kevlar helmets and body armor) and the high number of blast-related injuries are likely responsible for the high incidence of mTBI among OEF/OIF veterans.8,21,22 The prevalence of mTBI among service members not evacuated is estimated to be 20% to 30%.20
Symptoms can persist. Most patients with mTBI completely recover within 30 days of the injury. Unfortunately, 10% to 15% of mTBI patients develop chronic problems lasting months to years.4 Residual symptoms most commonly include headache, irritability, depression, sleep disturbance, impaired reasoning, memory problems, and difficulty concentrating. These symptoms are not unique to mTBI and overlap with comorbid combat diagnoses like PTSD, depression, and sleep deprivation.10 The following tools can help physicians determine whether mTBI is present.
Checking for possible mtBi. In the field, patients with possible mTBI can be screened rapidly using the Military Acute Concussion Evaluation (MACE, found at www.dvbic.org), a modification of the validated and widely used Sideline Assessment of Concussion (SAC) tool. More challenging is evaluating potential mTBI patients who present weeks or months after a traumatic event, for which there are no simple confirmatory tests. In this event, conduct a thorough neurological evaluation that includes vestibular, vision, postural, and neurocognitive assessments. For patients with persistent symptoms or possible anatomic brain abnormalities, magnetic resonance imaging (MRI) is the imaging modality of choice. Patients with complications or a questionable diagnosis are best managed in consultation with a neurologist.
Initial treatment of mtBi is symptom-based. When practical, try nonpharmacologic interventions first (TABLE 3).10 In particular, have the patient avoid further high-risk exposures that could lead to second impact syndrome (an issue increasingly recognized in contact sports). Also critical are physical and cognitive rest and the restoration of sleep until the patient is completely asymptomatic.
If the patient exhibits irritability and depression, selective serotonin reuptake inhibitors (SSRIs) are first-line treatment. Avoid narcotics and sedative-hypnotic sleep medications if treating comorbidities such as pain and sleep deprivation. The VA/DoD guideline on managing concussion and mTBI provides additional detailed, evidence-based treatment recommendations.17
Reliving the horror again and again: PTSD
PTSD is a persistent and, at times, debilitating clinical syndrome that develops after exposure to a psychologically traumatic event. It’s the second most common illness among OEF/OIF combat veterans, with an estimated prevalence of 3% to 20%, a finding consistent with prior wars.6,23-25 In the case of combat veterans, the inciting event usually involves an actual or perceived risk of death or serious injury. The individual’s response to the event involves intense fear, helplessness, or horror. The traumatic event is persistently re-experienced through intrusive and disturbing recollections or dreams that cause intense psychological distress. This, in turn, leads to a state of persistent sympathetic arousal. As symptoms are often triggered by specific cues, individuals with PTSD actively seek to avoid thoughts, situations, or stimuli associated with the event.23,26
Symptoms commonly associated with PTSD include difficulty falling or staying asleep, recurrent nightmares, hypervigilance, and an exaggerated startle response. Individuals with PTSD also have a poorer sense of well-being, a higher rate of work absenteeism, and significantly more somatic complaints than age-matched peers.27 For symptoms to be attributable to PTSD, their onset must follow a recent inciting event and must also cause clinically significant distress or impairment in social, occupational, or other areas of daily living. Common comorbid illnesses include mTBI, depression, and substance abuse. As with mTBI, the presence of multiple comorbidities in patients with PTSD can complicate evaluation, diagnosis, and treatment.
Diagnosis. PTSD is subdivided into acute (symptoms lasting more than one month but less than 3 months after the traumatic event) and chronic (symptoms lasting longer than 3 months after the traumatic event).28 The distinction of acute or chronic does not affect treatment, but it is useful information for the patient to have regarding prognosis and eventual outcome. Like mTBI, PTSD is a clinical diagnosis made only after a thorough, structured diagnostic interview. The use of a validated, self-administered checklist, such as the Post-Traumatic Checklist-Military (PCL-M), allows for an efficient review of a patient’s symptoms and a reliable way to track treatment progress (http://www.ptsd.va.gov/professional/ pages/assessments/ptsd-checklist.asp).
Treatment Options. Effective evidence-based treatments for PTSD are cognitive behavioral therapy, eye movement desensitization and reprocessing (EMDR), and pharmacotherapy. SSRIs and serotonin- norepinephrine reuptake inhibitors (SNRIs) have the strongest evidence for pharmacologic benefit in the treatment of PTSD.28,29 Other helpful medications are prazosin for nightmares and trazodone for sleep. Family physicians can use these medications as part of a patient-centered collaboration with the rest of the integrated care team, to offer the best chance for treatment success.10,28,30
Depression: Vets are reluctant to self-report
Combat experience is a significant risk factor for major depression. Estimates of the lifetime prevalence of depression in the general US population vary from 9% to 25% in women and 5% to 12% in men. By contrast, the prevalence of depression in OIF/OEF veterans ranges from 2% to 37%.24,31,32
Screening can yield false negatives. Many combat veterans are reluctant to self-report behavioral conditions, including depression. Screening, therefore, is important to identify potential depression and allow for intervention. Validated screening tools for depression include the PHQ-2 and PHQ-9, which are easy to use in the office setting. (See http://www.cqaimh.org/pdf/ tool_phq2.pdf [PHQ-2] and http://www. integration.samhsa.gov/images/res/ PHQ%20-%20Questions.pdf [PHQ-9]). Importantly, some veterans will have a negative depression screen on return from deployment, and then test positive 6 to 12 months later.24 Explanations for the early false-negative results include the excitement of being home and patients intentionally answering questions inaccurately to avoid excessive screening at their home base.11
Treatment is most effective with a combination approach. As with most cases of depression, combining psychotherapy and psychopharmacology appears to be most effective for treating depression related to combat experience.33,34 While SSRIs and SNRIs are typical first-line pharmacologic agents, combat veterans often have comorbid mTBI, PTSD, or substance abuse issues that may influence the initial choice of therapy35 (TABLE 3).10
Suicide is on the rise in the military
Historically, the incidence of suicide has been 25% lower in military personnel than in civilian peers.36 However, between 2005 and 2009, the incidence of suicide in the Marine Corps and Army almost doubled.37 While the exact reasons remain unknown, it is likely due to prolonged and repeated deployments to a combat environment.12 While the incidence of suicide has been particularly high in the Army (22 per 100,000 active-duty and reserve personnel per year), all services have been affected. In fact, since 2009, the number of suicides among active duty service members exceeds those killed in action.37
Consider all veterans to be at risk for suicide, and screen accordingly. An effective screening tool is the Columbia-Suicide Severity Rating Scale (C-SSRS), which is able to predict those most at risk for an impending suicide attempt.34 Service members identified as high risk for suicide require unhindered access to care. The VA has worked to improve access to care and provide evidence-based point-of-care treatment strategies.38 Available resources can be found in TABLE 1.
Unfortunately, even with effective screening and treatment, not all suicides can be prevented. Studies have demonstrated that approximately 65% of service members who commit suicide had no known history of communicating their suicidal intent. Sadly, 25% of service members who committed suicide had seen a mental health provider within the previous 30 days.39
Alcohol abuse is common; opioids present a unique risk
Excessive use of alcohol and recreational and prescription drugs is common among OEF/ OIF veterans, especially those with comorbid mental health disorders. Retrospective cross-sectional studies show that 11% to 20% of OEF/OIF veterans met DSM-IV-TR diagnostic criteria for substance use disorders.40-42 At highest risk are single enlisted men under the age of 24 in the Army or Marine Corps who serve in a combat-specific capacity. Interestingly, the prevalence of substance use disorders among OEF/OIF veterans closely mirrors that reported in epidemiologic studies of Vietnam veterans (11%-14%).41 This similarity, combined with the 39% lifetime prevalence of substance use disorders among Vietnam veterans, may foreshadow a similar lifetime prevalence of substance use disorders among OEF/OIF veterans.41
Most-abused substances. Alcohol is the most commonly abused substance among OEF/OIF veterans (10%-20%).40,41,43-45 Other abused substances include opioids (prescribed or illicitly obtained), synthetic marijuana (“Spice” and “K2”), and “bath salts” (synthetic stimulants) (W.M. Sauve, MD, personal communication, August 27, 2012).
OEF/OIF veterans seem to be at particular risk for developing problems related to opioid use. A 2012 retrospective cohort study showed that veterans with non–cancer- related pain diagnoses treated with opioid analgesics had an increased risk for adverse clinical outcomes compared with those not treated with opioid analgesics (9.5% vs 4.1%; relative risk [RR]=2.33; 95% confidence interval [CI], 2.20-2.46). These outcomes included traumatic accidents, overdoses, self-inflicted injuries, and injuries related to violence. This study also demonstrated that, compared with veterans without mental illness, veterans with mental illness (particularly PTSD) and non–cancer-related pain were significantly more likely to receive opioids to treat their pain and had a higher risk of adverse clinical outcomes, including overdose.46,47
Recreational use of synthetic marijuana and “bath salts” has increased in recent years. These substances are commonly labeled “not for human consumption,” which allows them to remain outside US Food and Drug Administration (FDA) regulation and be sold legally in the United States. Efforts to prohibit the sale or possession of these drugs, including the Federal Synthetic Bath Salt Ban in 2012, have fallen short, often due to creative product ”re-engineering.”33 Synthetic marijuana and stimulants are inexpensive, readily available, and perceived by users to be safe. Health care providers are often unaware that their patients are using these products. Adverse health outcomes associated with the use of these synthetic drugs include memory loss, depression, and psychosis.
These alcohol and drug screens can help
One efficient screening tool to identify veterans at risk for alcohol abuse is the AUDIT-C, developed by the World Health Organization. This brief 3-question test identifies past-year hazardous drinking and alcohol abuse or dependence with >79% sensitivity and >56% specificity in male veterans, and >66% sensitivity and >87% specificity in female veterans. These numbers are similar to those provided by the full 10-question AUDIT.48,49 The Drug Abuse Screen Test-10 (DAST-10) provides a similar screening instrument for other substances. Condensed from the original DAST-28 instrument, the DAST-10 identifies high-risk substance abuse with 74% to 94% sensitivity and 68% to 88% specificity.3
Screen for comorbidities. When you see veterans with a diagnosis of substance abuse, also evaluate for comorbid disease. Most veterans with substance use disorders (82%-93%) have at least one other mental health diagnosis (a 45% greater risk than that of civilians with substance abuse disorders),50 most commonly PTSD, depression, anxiety, and adjustment disorders.41,44,45 A number of hypotheses exist to explain the association between substance use disorders and other mental health diagnoses (“dual diagnoses”). The prevailing theory, in both veteran and civilian populations, is that substance abuse is an attempt to self-treat mental illness. Other evidence suggests that substance abuse promotes the development of mental illness, either by leading to a higher risk for traumatic experiences (increasing the chance of developing PTSD) or through a direct biochemical mechanism. Finally, con- current substance use disorder and mental illness may be due to an undefined genetic or biological vulnerability.38,44 This complicated relationship between substance abuse and behavioral health reinforces the need for screening, early diagnosis, and a comprehensive, multidisciplinary approach to treatment.
Treatment options. Office-based treatment options for narcotic and alcohol abuse and dependency are available to family physicians. Methadone has been used since the 1950s to treat opioid addiction and remains one of the mainstays of outpatient treatment.47,51 Originally, methadone was restricted to detoxification and maintenance treatment in narcotic addiction treatment programs approved by the FDA. In 1976, this restriction was lifted, and all physicians registered with the Drug Enforcement Agency (DEA) were permitted to prescribe methadone for analgesia.
In 2002, the FDA approved buprenorphine monotherapy and the combination product buprenorphine/naloxone for the treatment of opioid addiction. The prescribing of buprenorphine products requires physicians to undergo extra training, declare to the DEA their intent to prescribe buprenorphine, and obtain a special DEA identification number.52,53 Physicians interested in finding out more about buprenorphine treatment and prescribing requirements can go to the Substance Abuse and Mental Health Services Administration (SAMHSA) Web page at http://samhsa.gov.
Naltrexone is an opioid receptor agonist that is used primarily to treat alcohol dependency, and is thought to work by reducing the craving for alcohol. Multiple studies have proven the efficacy of naltrexone in an outpatient setting when used alone or in combination with psychotherapy.54,55 If you are uncomfortable or unfamiliar with the use or prescribing of these medications, referral to a substance abuse clinic specializing in dual-diagnosis treatment (TABLE 1) may offer optimal outcomes for patients with substance abuse disorders and other mental illness.
Cognitive behavioral therapy—including coping skills training, relapse prevention, contingency management, and behavioral couples’ therapy—and 12-step treatment programs are evidence-based options for the treatment of substance abuse disorders. Behavioral counseling interventions in the primary care setting (typically lasting 5-15 minutes) result in decreases in alcohol consumption, heavy drinking episodes, drinking above recommended amounts, and the number of days spent in the hospital, but have not been demonstrated to affect mortality, alcohol-related liver problems, outpatient visits, legal problems, or quality of life.56 Resources can be found at www.niaaa.nih.gov. For patients with dual diagnoses, it is not yet known whether sequential therapy (in which substance abuse is treated first, followed by treatment of the comorbid mental illness) or concurrent therapy results in better outcomes.57
CASE Your patient’s history of recent combat service, acknowledgement of employment and behavioral difficulties, and initial screening results lead you to diagnose alcoholism and depression. Additionally, she denies any suicidal ideation, but admits to experimenting with synthetic marijuana. After some discussion, she agrees to see your clinic’s social worker, and you start her on an SSri with scheduled follow-up.
CORRESPONDENCE
Shawn Kane, MD, USASoc, Attn: Surgeon (AomD), 2929 Desert Storm Drive, Ft. Bragg, NC 28310, or PO Box 3639 Pinehurst, NC 28374; [email protected]
› Ask, “Have you or a loved one ever served in the military?” as a way to uncover service-related concerns. C
› Conduct a thorough neurological evaluation with suspected mild traumatic brain injury, including vestibular, vision, postural, and neuro-cognitive assessments. C
› Use the Post-Traumatic Checklist–Military to assess individuals with possible post-traumatic stress disorder. C
Strength of recommendation (SOR)
A Good-quality patient-oriented evidence
B Inconsistent or limited-quality patient-oriented evidence
C Consensus, usual practice, opinion, disease-oriented evidence, case series
CASE A 37-year-old white woman presents for an employment physical. Your nurse reports that she also has a complaint of headaches, that she scored an 8 on the Alcohol Use Disorders identification Test-consumption (AUDiT-c), and that the result on her patient health Questionnaire (phQ-2) suggests a depressive disorder. You ask the patient whether she has served in the military and discover that, in the last 4 years, she served 2 year-long tours in Afghanistan with her Army reserve unit, returning home 6 months ago. Since her return, she has lost her job due to chronic tardiness (sleeping through her alarm, she says) and admits she has “started drinking again.” Her visit with you this day is only to undergo the physical exam required by her new employer. What are your next steps with this patient? What resources can you use to help her?
As long as human beings have engaged in combat, there have often been extraordinarily damaging psychiatric1 injuries among those who survive. Combat survivability today is 84% to 90%, the highest in the history of armed conflict,2,3 thanks to improvements in personal protective gear, vehicle armor, rapid casualty evacuation, and surgical resuscitation and stabilization that is “far forward” on the battlefield. These survivors are subsequently at high risk for a host of other medical conditions, which commonly include traumatic brain injury (TBI), post-traumatic stress disorder (PTSD), depression, suicide, and substance abuse.4-8
Family physicians—both civilian and uniformed—may be the first to encounter these individuals. Of the more than 2.4 million US service members who have been deployed to Afghanistan or Iraq in support of Operation Enduring Freedom (OEF) or Operation Iraqi Freedom (OIF), nearly 60% are no longer on active duty.
Among this group, only half receive care from the US Department of Veterans Affairs (VA).9 Despite a concerted effort on the part of the Department of Defense (DoD) and the VA to develop and distribute effective, evidenced-based treatment protocols for veterans with combat-related conditions, major gaps remain in the care provided to combat veterans.10
This article seeks to help fill that gap by providing the information you need to recognize and treat common combat-related illness, as well as resources to help improve the quality of life for veterans and their families (TABLE 1).
Initial roadblocks to care
One of the biggest challenges in treating veterans with behavioral health issues is the fact that only 23% to 40% of those with mental illness seek care.11 Among the reasons veterans have offered for avoiding behavioral health care are a fear of the stigma associated with mental illness, concern that treatment will negatively affect their career, lack of comfort with mental health professionals, and the perception that mental health treatment is a “last resort.”12 Unfortunately, efforts by the DoD leadership to overcome these inherent biases have been largely unsuccessful13 and much work is still required to see that service members get the care they need.
Due to low rates of self-reporting, effective screening is essential. With this in mind, the DoD has implemented the deployment health assessment program (DHAP), which requires service members to be screened for common conditions within 60 days of deployment, within 30 days of returning, and again at 90 to 180 days after their return.
While the long-term effects of this program are yet to be determined, results to date are promising. Since the DHAP was implemented, there has been a significant decrease in occupationally impairing mental health problems and suicidal ideation requiring medical evacuation from a combat theater.14
FPs should begin with a simple question. Many of the 20+ million veterans living in the United States will not be wearing a uniform when they enter your office. Simply asking all of your patients, “Have you or a loved one ever served in the military?” may help you discover service-related questions or concerns.15,16 Underscoring the importance of such screening is the recent decision by the American Academy of Family Physicians to partner with First Lady Michelle Obama and Dr. Jill Biden in a new campaign called “Joining Forces,” which aims to support veterans and their families.16
Mild traumatic brain injury: Common—though overlooked
A TBI is any temporary or permanent neurologic dysfunction after a blow to the head.10,17 TBI is classified based on severity and mechanism (direct blow to the head or exposure to blast waves). Mild TBI (mTBI) is commonly referred to as a concussion and usually is not associated with loss of consciousness or altered mental status. Brain imaging results are also normal with mTBI. Severe TBI, on the other hand, is associated with prolonged loss of consciousness, altered mental status, and abnormal brain imaging results (TABLE 2).17
A unique obstacle to accurate evaluation in the field. It is important to emphasize that mTBI is a clinical diagnosis, and its detection requires honest patient communication. This can be problematic with motivated soldiers who are anxious to continue the mission and fear that any admission of symptoms might delay a return to their unit. As with a concussed athlete eager to return to the field of play, the clinical diagnosis of mTBI requires a high index of clinical suspicion and constant vigilance by the health care provider. Despite being the most common combat- related injury, mTBI is often overlooked due to the absence of obvious physical injuries.4 Recent data suggest that 28% to 60% of ser- vice members evacuated from combat have a TBI. Most of these injuries (77%) are mTBI.18-20 Improved personal protective equipment (including Kevlar helmets and body armor) and the high number of blast-related injuries are likely responsible for the high incidence of mTBI among OEF/OIF veterans.8,21,22 The prevalence of mTBI among service members not evacuated is estimated to be 20% to 30%.20
Symptoms can persist. Most patients with mTBI completely recover within 30 days of the injury. Unfortunately, 10% to 15% of mTBI patients develop chronic problems lasting months to years.4 Residual symptoms most commonly include headache, irritability, depression, sleep disturbance, impaired reasoning, memory problems, and difficulty concentrating. These symptoms are not unique to mTBI and overlap with comorbid combat diagnoses like PTSD, depression, and sleep deprivation.10 The following tools can help physicians determine whether mTBI is present.
Checking for possible mtBi. In the field, patients with possible mTBI can be screened rapidly using the Military Acute Concussion Evaluation (MACE, found at www.dvbic.org), a modification of the validated and widely used Sideline Assessment of Concussion (SAC) tool. More challenging is evaluating potential mTBI patients who present weeks or months after a traumatic event, for which there are no simple confirmatory tests. In this event, conduct a thorough neurological evaluation that includes vestibular, vision, postural, and neurocognitive assessments. For patients with persistent symptoms or possible anatomic brain abnormalities, magnetic resonance imaging (MRI) is the imaging modality of choice. Patients with complications or a questionable diagnosis are best managed in consultation with a neurologist.
Initial treatment of mtBi is symptom-based. When practical, try nonpharmacologic interventions first (TABLE 3).10 In particular, have the patient avoid further high-risk exposures that could lead to second impact syndrome (an issue increasingly recognized in contact sports). Also critical are physical and cognitive rest and the restoration of sleep until the patient is completely asymptomatic.
If the patient exhibits irritability and depression, selective serotonin reuptake inhibitors (SSRIs) are first-line treatment. Avoid narcotics and sedative-hypnotic sleep medications if treating comorbidities such as pain and sleep deprivation. The VA/DoD guideline on managing concussion and mTBI provides additional detailed, evidence-based treatment recommendations.17
Reliving the horror again and again: PTSD
PTSD is a persistent and, at times, debilitating clinical syndrome that develops after exposure to a psychologically traumatic event. It’s the second most common illness among OEF/OIF combat veterans, with an estimated prevalence of 3% to 20%, a finding consistent with prior wars.6,23-25 In the case of combat veterans, the inciting event usually involves an actual or perceived risk of death or serious injury. The individual’s response to the event involves intense fear, helplessness, or horror. The traumatic event is persistently re-experienced through intrusive and disturbing recollections or dreams that cause intense psychological distress. This, in turn, leads to a state of persistent sympathetic arousal. As symptoms are often triggered by specific cues, individuals with PTSD actively seek to avoid thoughts, situations, or stimuli associated with the event.23,26
Symptoms commonly associated with PTSD include difficulty falling or staying asleep, recurrent nightmares, hypervigilance, and an exaggerated startle response. Individuals with PTSD also have a poorer sense of well-being, a higher rate of work absenteeism, and significantly more somatic complaints than age-matched peers.27 For symptoms to be attributable to PTSD, their onset must follow a recent inciting event and must also cause clinically significant distress or impairment in social, occupational, or other areas of daily living. Common comorbid illnesses include mTBI, depression, and substance abuse. As with mTBI, the presence of multiple comorbidities in patients with PTSD can complicate evaluation, diagnosis, and treatment.
Diagnosis. PTSD is subdivided into acute (symptoms lasting more than one month but less than 3 months after the traumatic event) and chronic (symptoms lasting longer than 3 months after the traumatic event).28 The distinction of acute or chronic does not affect treatment, but it is useful information for the patient to have regarding prognosis and eventual outcome. Like mTBI, PTSD is a clinical diagnosis made only after a thorough, structured diagnostic interview. The use of a validated, self-administered checklist, such as the Post-Traumatic Checklist-Military (PCL-M), allows for an efficient review of a patient’s symptoms and a reliable way to track treatment progress (http://www.ptsd.va.gov/professional/ pages/assessments/ptsd-checklist.asp).
Treatment Options. Effective evidence-based treatments for PTSD are cognitive behavioral therapy, eye movement desensitization and reprocessing (EMDR), and pharmacotherapy. SSRIs and serotonin- norepinephrine reuptake inhibitors (SNRIs) have the strongest evidence for pharmacologic benefit in the treatment of PTSD.28,29 Other helpful medications are prazosin for nightmares and trazodone for sleep. Family physicians can use these medications as part of a patient-centered collaboration with the rest of the integrated care team, to offer the best chance for treatment success.10,28,30
Depression: Vets are reluctant to self-report
Combat experience is a significant risk factor for major depression. Estimates of the lifetime prevalence of depression in the general US population vary from 9% to 25% in women and 5% to 12% in men. By contrast, the prevalence of depression in OIF/OEF veterans ranges from 2% to 37%.24,31,32
Screening can yield false negatives. Many combat veterans are reluctant to self-report behavioral conditions, including depression. Screening, therefore, is important to identify potential depression and allow for intervention. Validated screening tools for depression include the PHQ-2 and PHQ-9, which are easy to use in the office setting. (See http://www.cqaimh.org/pdf/ tool_phq2.pdf [PHQ-2] and http://www. integration.samhsa.gov/images/res/ PHQ%20-%20Questions.pdf [PHQ-9]). Importantly, some veterans will have a negative depression screen on return from deployment, and then test positive 6 to 12 months later.24 Explanations for the early false-negative results include the excitement of being home and patients intentionally answering questions inaccurately to avoid excessive screening at their home base.11
Treatment is most effective with a combination approach. As with most cases of depression, combining psychotherapy and psychopharmacology appears to be most effective for treating depression related to combat experience.33,34 While SSRIs and SNRIs are typical first-line pharmacologic agents, combat veterans often have comorbid mTBI, PTSD, or substance abuse issues that may influence the initial choice of therapy35 (TABLE 3).10
Suicide is on the rise in the military
Historically, the incidence of suicide has been 25% lower in military personnel than in civilian peers.36 However, between 2005 and 2009, the incidence of suicide in the Marine Corps and Army almost doubled.37 While the exact reasons remain unknown, it is likely due to prolonged and repeated deployments to a combat environment.12 While the incidence of suicide has been particularly high in the Army (22 per 100,000 active-duty and reserve personnel per year), all services have been affected. In fact, since 2009, the number of suicides among active duty service members exceeds those killed in action.37
Consider all veterans to be at risk for suicide, and screen accordingly. An effective screening tool is the Columbia-Suicide Severity Rating Scale (C-SSRS), which is able to predict those most at risk for an impending suicide attempt.34 Service members identified as high risk for suicide require unhindered access to care. The VA has worked to improve access to care and provide evidence-based point-of-care treatment strategies.38 Available resources can be found in TABLE 1.
Unfortunately, even with effective screening and treatment, not all suicides can be prevented. Studies have demonstrated that approximately 65% of service members who commit suicide had no known history of communicating their suicidal intent. Sadly, 25% of service members who committed suicide had seen a mental health provider within the previous 30 days.39
Alcohol abuse is common; opioids present a unique risk
Excessive use of alcohol and recreational and prescription drugs is common among OEF/ OIF veterans, especially those with comorbid mental health disorders. Retrospective cross-sectional studies show that 11% to 20% of OEF/OIF veterans met DSM-IV-TR diagnostic criteria for substance use disorders.40-42 At highest risk are single enlisted men under the age of 24 in the Army or Marine Corps who serve in a combat-specific capacity. Interestingly, the prevalence of substance use disorders among OEF/OIF veterans closely mirrors that reported in epidemiologic studies of Vietnam veterans (11%-14%).41 This similarity, combined with the 39% lifetime prevalence of substance use disorders among Vietnam veterans, may foreshadow a similar lifetime prevalence of substance use disorders among OEF/OIF veterans.41
Most-abused substances. Alcohol is the most commonly abused substance among OEF/OIF veterans (10%-20%).40,41,43-45 Other abused substances include opioids (prescribed or illicitly obtained), synthetic marijuana (“Spice” and “K2”), and “bath salts” (synthetic stimulants) (W.M. Sauve, MD, personal communication, August 27, 2012).
OEF/OIF veterans seem to be at particular risk for developing problems related to opioid use. A 2012 retrospective cohort study showed that veterans with non–cancer- related pain diagnoses treated with opioid analgesics had an increased risk for adverse clinical outcomes compared with those not treated with opioid analgesics (9.5% vs 4.1%; relative risk [RR]=2.33; 95% confidence interval [CI], 2.20-2.46). These outcomes included traumatic accidents, overdoses, self-inflicted injuries, and injuries related to violence. This study also demonstrated that, compared with veterans without mental illness, veterans with mental illness (particularly PTSD) and non–cancer-related pain were significantly more likely to receive opioids to treat their pain and had a higher risk of adverse clinical outcomes, including overdose.46,47
Recreational use of synthetic marijuana and “bath salts” has increased in recent years. These substances are commonly labeled “not for human consumption,” which allows them to remain outside US Food and Drug Administration (FDA) regulation and be sold legally in the United States. Efforts to prohibit the sale or possession of these drugs, including the Federal Synthetic Bath Salt Ban in 2012, have fallen short, often due to creative product ”re-engineering.”33 Synthetic marijuana and stimulants are inexpensive, readily available, and perceived by users to be safe. Health care providers are often unaware that their patients are using these products. Adverse health outcomes associated with the use of these synthetic drugs include memory loss, depression, and psychosis.
These alcohol and drug screens can help
One efficient screening tool to identify veterans at risk for alcohol abuse is the AUDIT-C, developed by the World Health Organization. This brief 3-question test identifies past-year hazardous drinking and alcohol abuse or dependence with >79% sensitivity and >56% specificity in male veterans, and >66% sensitivity and >87% specificity in female veterans. These numbers are similar to those provided by the full 10-question AUDIT.48,49 The Drug Abuse Screen Test-10 (DAST-10) provides a similar screening instrument for other substances. Condensed from the original DAST-28 instrument, the DAST-10 identifies high-risk substance abuse with 74% to 94% sensitivity and 68% to 88% specificity.3
Screen for comorbidities. When you see veterans with a diagnosis of substance abuse, also evaluate for comorbid disease. Most veterans with substance use disorders (82%-93%) have at least one other mental health diagnosis (a 45% greater risk than that of civilians with substance abuse disorders),50 most commonly PTSD, depression, anxiety, and adjustment disorders.41,44,45 A number of hypotheses exist to explain the association between substance use disorders and other mental health diagnoses (“dual diagnoses”). The prevailing theory, in both veteran and civilian populations, is that substance abuse is an attempt to self-treat mental illness. Other evidence suggests that substance abuse promotes the development of mental illness, either by leading to a higher risk for traumatic experiences (increasing the chance of developing PTSD) or through a direct biochemical mechanism. Finally, con- current substance use disorder and mental illness may be due to an undefined genetic or biological vulnerability.38,44 This complicated relationship between substance abuse and behavioral health reinforces the need for screening, early diagnosis, and a comprehensive, multidisciplinary approach to treatment.
Treatment options. Office-based treatment options for narcotic and alcohol abuse and dependency are available to family physicians. Methadone has been used since the 1950s to treat opioid addiction and remains one of the mainstays of outpatient treatment.47,51 Originally, methadone was restricted to detoxification and maintenance treatment in narcotic addiction treatment programs approved by the FDA. In 1976, this restriction was lifted, and all physicians registered with the Drug Enforcement Agency (DEA) were permitted to prescribe methadone for analgesia.
In 2002, the FDA approved buprenorphine monotherapy and the combination product buprenorphine/naloxone for the treatment of opioid addiction. The prescribing of buprenorphine products requires physicians to undergo extra training, declare to the DEA their intent to prescribe buprenorphine, and obtain a special DEA identification number.52,53 Physicians interested in finding out more about buprenorphine treatment and prescribing requirements can go to the Substance Abuse and Mental Health Services Administration (SAMHSA) Web page at http://samhsa.gov.
Naltrexone is an opioid receptor agonist that is used primarily to treat alcohol dependency, and is thought to work by reducing the craving for alcohol. Multiple studies have proven the efficacy of naltrexone in an outpatient setting when used alone or in combination with psychotherapy.54,55 If you are uncomfortable or unfamiliar with the use or prescribing of these medications, referral to a substance abuse clinic specializing in dual-diagnosis treatment (TABLE 1) may offer optimal outcomes for patients with substance abuse disorders and other mental illness.
Cognitive behavioral therapy—including coping skills training, relapse prevention, contingency management, and behavioral couples’ therapy—and 12-step treatment programs are evidence-based options for the treatment of substance abuse disorders. Behavioral counseling interventions in the primary care setting (typically lasting 5-15 minutes) result in decreases in alcohol consumption, heavy drinking episodes, drinking above recommended amounts, and the number of days spent in the hospital, but have not been demonstrated to affect mortality, alcohol-related liver problems, outpatient visits, legal problems, or quality of life.56 Resources can be found at www.niaaa.nih.gov. For patients with dual diagnoses, it is not yet known whether sequential therapy (in which substance abuse is treated first, followed by treatment of the comorbid mental illness) or concurrent therapy results in better outcomes.57
CASE Your patient’s history of recent combat service, acknowledgement of employment and behavioral difficulties, and initial screening results lead you to diagnose alcoholism and depression. Additionally, she denies any suicidal ideation, but admits to experimenting with synthetic marijuana. After some discussion, she agrees to see your clinic’s social worker, and you start her on an SSri with scheduled follow-up.
CORRESPONDENCE
Shawn Kane, MD, USASoc, Attn: Surgeon (AomD), 2929 Desert Storm Drive, Ft. Bragg, NC 28310, or PO Box 3639 Pinehurst, NC 28374; [email protected]
1. Wessely S. Risk, psychiatry and the military. Br J Psychiatry. 2005;186:459-466.
2. Gawande A. Casualties of war—military care for the wounded from Iraq and Afghanistan. N Engl J Med. 2004;351:2471-2475.
3. Kotwal RS, Montgomery HR, Kotwal BM, et al. Eliminating pre- ventable death on the battlefield. Arch Surg. 2011;146:1350-1358.
4. Belanger HG, Uomoto JM, Vanderploeg RD. The Veterans Health Administration’s (VHA’s) Polytrauma System of Care for mild traumatic brain injury: costs, benefits, and controversies. J Head Trauma Rehabil. 2009;24:4-13.
5. Galarneau MR, Woodruff SI, Dye JL, et al. Traumatic brain in- jury during Operation Iraqi Freedom: findings from the United States Navy-Marine Corps Combat Trauma Registry. J Neurosurg. 2008;108:950-957.
6. Hermann BA, Shiner B, Friedman MJ. Epidemiology and preven- tion of combat-related post-traumatic stress in OEF/OIF/OND service members. Mil Med. 2012;177:1-6.
7. Uomoto JM. Best practices in veteran traumatic brain injury care. J Head Trauma Rehabil. 2012;27:241-243.
8. Warden D. Military TBI during the Iraq and Afghanistan wars. J Head Trauma Rehabil. 2006;21:398-402.
9. Taylor BC, Hagel EM, Carlson KF, et al. Prevalence and costs of co-occurring traumatic brain injury with and without psychiatric disturbance and pain among Afghanistan and Iraq War Veteran V.A. users. Med Care. 2012;50:342-346.
10. Quinlan JD, Guaron MR, Deschere BR, et al. Care of the returning veteran. Am Fam Physician. 2010;82:43-49.
11. Hoge CW, Castro CA, Messer SC, et al. Combat duty in Iraq and Afghanistan, mental health problems, and barriers to care. N Engl J Med. 2004;351:13-22.
12. Hoge CW, Castro CA. Preventing suicides in US service mem- bers and veterans: concerns after a decade of war. JAMA. 2012;308:671-672.
13. Jaffe G. New name for PTSD could mean less stigma. The Washington Post. May 5, 2012. Available at: http://articles. washingtonpost.com/2012-05-05/world/35454931_1_ptsd-post- traumatic-stress-psychiatrists. Accessed June 19, 2013.
14. Warner CH, Appenzeller GN, Parker JR, et al. Effectiveness of mental health screening and coordination of in-theater care prior to deployment to Iraq: a cohort study. Am J Psychiatry. 2011;168:378-385.
15. United States Census Bureau. Sex by age by veteran sta- tus for civilian population 18 years and over. 2010 American community survey 1-year estimates. Available at: https:// d3gqux9sl0z33u.cloudfront.net/AA/AT/gambillingonjustice- com/downloads/206273/ACS_10_1YR_B21001A.pdf. Accessed June 19, 2013.
16. American Academy of Family Physicians. Joining forces. Avail- able at: http://www.aafp.org/online/en/home/membership/ initiatives/joiningforces.html. Accessed June 19, 2013.
17. Department of Veterans Affairs and Department of Defense. Clinical Practice Guideline for Management of Concussion/Mild Traumatic Brain Injury. April 2009. Available at: http://www. healthquality.va.gov/mtbi/concussion_mtbi_full_1_0.pdf. Accessed June 19, 2013.
18. Lew HL, Poole JH, Alvarez S, et al. Soldiers with occult traumatic brain injury. Am J Phys Med Rehabil. 2005;84:393-398.
19. Marshall KR, Holland SL, Meyer KS, et al. Mild traumatic brain injury screening, diagnosis, and treatment. Mil Med. 2012;177:67- 75.
20. Terrio H, Brenner LA, Ivins BJ, et al. Traumatic brain injury screening: preliminary findings in a US Army Brigade Combat Team. J Head Trauma Rehabil. 2009;24:14-23.
21. Mossadegh S, Tai N, Midwinter M, et al. Improvised explosive de- vice related pelvi-perineal trauma: anatomic injuries and surgical management. J Trauma Acute Care Surg. 2012;73:S24-S31.
22. Okie S. Traumatic brain injury in the war zone. N Engl J Med. 2005;352:2043-2047.
23. Espinoza JM. Posttraumatic stress disorder and the perceived consequences of seeking therapy among US Army special forces operators exposed to combat. J Psychol Issues Organ Culture. 2010;1:6-28.
24. Grieger TA, Cozza SJ, Ursano RJ, et al. Posttraumatic stress dis- order and depression in battle-injured soldiers. Am J Psychiatry. 2006;163:1777-1783.
25. Hoge CW, Auchterlonie JL, Milliken CS. Mental health problems, use of mental health services, and attrition from military service after returning from deployment to Iraq or Afghanistan. JAMA. 2006;295:1023-1032.
26. Adler AB, Wright KM, Bliese PD, et al. A2 diagnostic criterion for combat-related posttraumatic stress disorder. J Trauma Stress. 2008;21:301-308.
27. Hoge CW, Terhakopian A, Castro CA, et al. Association of post- traumatic stress disorder with somatic symptoms, health care vis- its, and absenteeism among Iraq war veterans. Am J Psychiatry. 2007;164:150-153.
28. Department of Veterans Affairs and Department of Defense. Clin- ical Practice Guideline for Management of Post-Traumatic Stress. October 2010. Available at: http://www.healthquality.va.gov/ ptsd/cpg_PTSD-FULL-201011612.pdf. Accessed June 19, 2013.
29. Alexander W. Pharmacotherapy for post-traumatic stress disor- der in combat veterans: focus on antidepressants and atypical antipsychotic agents. P T. 2012;37:32-38.
30. Wisco BE, Marx BP, Keane TM. Screening, diagnosis, and treat- ment of post-traumatic stress disorder. Mil Med. 2012;177:7-13.
31. Gadermann AM, Engel CC, Naifeh JA, et al. Prevalence of DSM-IV major depression among U.S. military personnel: meta-analysis and simulation. Mil Med. 2012;177:47-59.
32. Seal KH, Shi Y, Cohen G, et al. Association of mental health dis- orders with prescription opioids and high-risk opioid use in US veterans of Iraq and Afghanistan. JAMA. 2012;307:940-947.
33. Perrone M. Many drugs remain legal after ‘bath salts’ ban. Boston. com. July 25, 2012. Available at: http://articles.boston.com/2012- 07-25/lifestyle/32850962_1_bath-salts-mdpv-synthetic-drugs. Accessed June 19, 2013.
34. Posner K, Brown GK, Stanley B, et al. The Columbia-Suicide Se- verity Rating Scale: initial validity and internal consistency find- ings from three multisite studies with adolescents and adults. Am J Psychiatry. 2011;168:1266-1277.
35. Greenberg J, Tesfazion AA, Robinson CS. Screening, diagnosis, and treatment of depression. Mil Med. 2012;177:60-66.
36. Eaton KM, Messer SC, Garvey Wilson AL, et al. Strengthening the validity of population-based suicide rate comparisons: an il- lustration using U.S. military and civilian data. Suicide Life Threat Behav. 2006;36:182-191.
37. Miller M, Azrael D, Barber C, et al. A call to link data to answer pressing questions about suicide risk among veterans. Am J Pub Health. 2012;102(suppl 1):S20-S22.
38. Department of Veterans Affairs. Report of the Blue Ribbon Work Group on suicide prevention in the veteran population. June 2008. Available at: http://www.mentalhealth.va.gov/suicide_ prevention/Blue_Ribbon_Report-FINAL_June-30-08.pdf. Accessed July 18, 2013.
39. Kinn JT, Luxton DD, Reger MA, et al. Department of Defense sui- cide event report: calendar year 2010 annual report. September 2011. Available at: http://t2health.org/sites/default/files/dodser/ DoDSER_2010_Annual_Report.pdf. Accessed June 19, 2013.
40. Fontana A, Rosenheck R. Treatment-seeking veterans of Iraq and Afghanistan: comparison with veterans of previous wars. J Nerv Ment Dis. 2008;196:513-521.
41. Seal KH, Cohen G, Waldrop A, et al. Substance use disorders in Iraq and Afghanistan veterans in VA healthcare, 2001-2010: implications for screening, diagnosis and treatment. Drug Alcohol Depend. 2011;116:93-101.
42. Mirza RA, Eick-Cost A, Otto JL. The risk of mental health disor- ders among U.S. military personnel infected with human immu- nodeficiency virus, active component, U.S. Armed Forces, 2000- 2011. MSMR. 2012;19:10-13.
43. Bohnert AS, Ilgen MA, Bossarte RM, et al. Veteran status and alco- hol use in men in the United States. Mil Med. 2012;177:198-203.
44. Erbes CR, Kaler ME, Schult T, et al. Mental health diagnosis and occupational functioning in National Guard/Reserve veterans re- turning from Iraq. J Rehabil Res Dev. 2011;48:1159-1170.
45. Stecker T, Fortney J, Owen R, et al. Co-occurring medical, psychi- atric, and alcohol-related disorders among veterans returning from Iraq and Afghanistan. Psychosomatics. 2010;51:503-507.
46. Seal KH, Shi Y, Cohen G, et al. Association of mental health dis- orders with prescription opioids and high-risk opioid use in US veterans of Iraq and Afghanistan. JAMA. 2012;307:940-947.
47. Praveen KT, Law F, O’Shea J, et al. Opioid dependence. Am Fam Physician. 2012;86:565-566.
48. Bradley KA, Bush KR, Epler AJ, et al. Two brief alcohol-screening tests from the Alcohol Use Disorders Identification Test (AUDIT): validation in a female Veterans Affairs patient population. Arch Intern Med. 2003;163:821-829.
49. Bush K, Kivlahan DR, McDonell MB, et al. The AUDIT alcohol consumption questions (AUDIT-C): an effective brief screening test for problem drinking. Ambulatory Care Quality Improvement Project (ACQUIP). Alcohol Use Disorders Identification Test. Arch Intern Med. 1998;158:1789-1795.
50. Farrell M, Howes S, Taylor C, et al. Substance misuse and psychi- atric comorbidity: an overview of the OPCS National Psychiatric Morbidity Survey. Addict Behav. 1998;23:909-918.
51. Toombs JD, Kral LA. Methadone treatment for pain states. Am Fam Physician. 2005;71:1353-1358.
52. Center for Substance Abuse Treatment. Clinical Guidelines for the Use of Buprenorphine in the Treatment of Opioid Addiction. Treatment Improvement Protocol (TIP) series 40. DHHS pub- lication (SMA) 04-3939. Rockville, MD: Substance Abuse and Mental Health Services Administration; 2004. Available at: http:// buprenorphine.samhsa.gov/Bup_Guidelines.pdf. Accessed June 19, 2013.
53. U.S.DepartmentofHealthandHumanServices,SubstanceAbuse and Mental Health Services Administration Web site. About buprenorphine therapy. Available at: http://buprenorphine. samhsa.gov/about.html. Accessed June 19, 2013.
54. Volpicelli JR, Alterman AI, Hayashida M, et al. Naltrexone in the treatment of alcohol dependence. Arch Gen Psychiatry. 1992;49:876-880.
55. O’Brien CP, Volpicelli LA, Volpicelli JR. Naltrexone in the treat- ment of alcoholism: a clinical review. Alcohol. 1996;13:35-39.
56. Jonas DE, Garbutt JC, Amick HR, et al. Behavioral counseling after screening for alcohol misuse in primary care: a systematic review and meta-analysis for the U.S. Preventive Services Task Force. Ann Intern Med. 2012;157:645-654.
57. van Dam D, Vedel E, Ehring T, et al. Psychological treatments for concurrent posttraumatic stress disorder and substance use dis- order: a systematic review. Clin Psychol Rev. 2012;32:202-214.
1. Wessely S. Risk, psychiatry and the military. Br J Psychiatry. 2005;186:459-466.
2. Gawande A. Casualties of war—military care for the wounded from Iraq and Afghanistan. N Engl J Med. 2004;351:2471-2475.
3. Kotwal RS, Montgomery HR, Kotwal BM, et al. Eliminating pre- ventable death on the battlefield. Arch Surg. 2011;146:1350-1358.
4. Belanger HG, Uomoto JM, Vanderploeg RD. The Veterans Health Administration’s (VHA’s) Polytrauma System of Care for mild traumatic brain injury: costs, benefits, and controversies. J Head Trauma Rehabil. 2009;24:4-13.
5. Galarneau MR, Woodruff SI, Dye JL, et al. Traumatic brain in- jury during Operation Iraqi Freedom: findings from the United States Navy-Marine Corps Combat Trauma Registry. J Neurosurg. 2008;108:950-957.
6. Hermann BA, Shiner B, Friedman MJ. Epidemiology and preven- tion of combat-related post-traumatic stress in OEF/OIF/OND service members. Mil Med. 2012;177:1-6.
7. Uomoto JM. Best practices in veteran traumatic brain injury care. J Head Trauma Rehabil. 2012;27:241-243.
8. Warden D. Military TBI during the Iraq and Afghanistan wars. J Head Trauma Rehabil. 2006;21:398-402.
9. Taylor BC, Hagel EM, Carlson KF, et al. Prevalence and costs of co-occurring traumatic brain injury with and without psychiatric disturbance and pain among Afghanistan and Iraq War Veteran V.A. users. Med Care. 2012;50:342-346.
10. Quinlan JD, Guaron MR, Deschere BR, et al. Care of the returning veteran. Am Fam Physician. 2010;82:43-49.
11. Hoge CW, Castro CA, Messer SC, et al. Combat duty in Iraq and Afghanistan, mental health problems, and barriers to care. N Engl J Med. 2004;351:13-22.
12. Hoge CW, Castro CA. Preventing suicides in US service mem- bers and veterans: concerns after a decade of war. JAMA. 2012;308:671-672.
13. Jaffe G. New name for PTSD could mean less stigma. The Washington Post. May 5, 2012. Available at: http://articles. washingtonpost.com/2012-05-05/world/35454931_1_ptsd-post- traumatic-stress-psychiatrists. Accessed June 19, 2013.
14. Warner CH, Appenzeller GN, Parker JR, et al. Effectiveness of mental health screening and coordination of in-theater care prior to deployment to Iraq: a cohort study. Am J Psychiatry. 2011;168:378-385.
15. United States Census Bureau. Sex by age by veteran sta- tus for civilian population 18 years and over. 2010 American community survey 1-year estimates. Available at: https:// d3gqux9sl0z33u.cloudfront.net/AA/AT/gambillingonjustice- com/downloads/206273/ACS_10_1YR_B21001A.pdf. Accessed June 19, 2013.
16. American Academy of Family Physicians. Joining forces. Avail- able at: http://www.aafp.org/online/en/home/membership/ initiatives/joiningforces.html. Accessed June 19, 2013.
17. Department of Veterans Affairs and Department of Defense. Clinical Practice Guideline for Management of Concussion/Mild Traumatic Brain Injury. April 2009. Available at: http://www. healthquality.va.gov/mtbi/concussion_mtbi_full_1_0.pdf. Accessed June 19, 2013.
18. Lew HL, Poole JH, Alvarez S, et al. Soldiers with occult traumatic brain injury. Am J Phys Med Rehabil. 2005;84:393-398.
19. Marshall KR, Holland SL, Meyer KS, et al. Mild traumatic brain injury screening, diagnosis, and treatment. Mil Med. 2012;177:67- 75.
20. Terrio H, Brenner LA, Ivins BJ, et al. Traumatic brain injury screening: preliminary findings in a US Army Brigade Combat Team. J Head Trauma Rehabil. 2009;24:14-23.
21. Mossadegh S, Tai N, Midwinter M, et al. Improvised explosive de- vice related pelvi-perineal trauma: anatomic injuries and surgical management. J Trauma Acute Care Surg. 2012;73:S24-S31.
22. Okie S. Traumatic brain injury in the war zone. N Engl J Med. 2005;352:2043-2047.
23. Espinoza JM. Posttraumatic stress disorder and the perceived consequences of seeking therapy among US Army special forces operators exposed to combat. J Psychol Issues Organ Culture. 2010;1:6-28.
24. Grieger TA, Cozza SJ, Ursano RJ, et al. Posttraumatic stress dis- order and depression in battle-injured soldiers. Am J Psychiatry. 2006;163:1777-1783.
25. Hoge CW, Auchterlonie JL, Milliken CS. Mental health problems, use of mental health services, and attrition from military service after returning from deployment to Iraq or Afghanistan. JAMA. 2006;295:1023-1032.
26. Adler AB, Wright KM, Bliese PD, et al. A2 diagnostic criterion for combat-related posttraumatic stress disorder. J Trauma Stress. 2008;21:301-308.
27. Hoge CW, Terhakopian A, Castro CA, et al. Association of post- traumatic stress disorder with somatic symptoms, health care vis- its, and absenteeism among Iraq war veterans. Am J Psychiatry. 2007;164:150-153.
28. Department of Veterans Affairs and Department of Defense. Clin- ical Practice Guideline for Management of Post-Traumatic Stress. October 2010. Available at: http://www.healthquality.va.gov/ ptsd/cpg_PTSD-FULL-201011612.pdf. Accessed June 19, 2013.
29. Alexander W. Pharmacotherapy for post-traumatic stress disor- der in combat veterans: focus on antidepressants and atypical antipsychotic agents. P T. 2012;37:32-38.
30. Wisco BE, Marx BP, Keane TM. Screening, diagnosis, and treat- ment of post-traumatic stress disorder. Mil Med. 2012;177:7-13.
31. Gadermann AM, Engel CC, Naifeh JA, et al. Prevalence of DSM-IV major depression among U.S. military personnel: meta-analysis and simulation. Mil Med. 2012;177:47-59.
32. Seal KH, Shi Y, Cohen G, et al. Association of mental health dis- orders with prescription opioids and high-risk opioid use in US veterans of Iraq and Afghanistan. JAMA. 2012;307:940-947.
33. Perrone M. Many drugs remain legal after ‘bath salts’ ban. Boston. com. July 25, 2012. Available at: http://articles.boston.com/2012- 07-25/lifestyle/32850962_1_bath-salts-mdpv-synthetic-drugs. Accessed June 19, 2013.
34. Posner K, Brown GK, Stanley B, et al. The Columbia-Suicide Se- verity Rating Scale: initial validity and internal consistency find- ings from three multisite studies with adolescents and adults. Am J Psychiatry. 2011;168:1266-1277.
35. Greenberg J, Tesfazion AA, Robinson CS. Screening, diagnosis, and treatment of depression. Mil Med. 2012;177:60-66.
36. Eaton KM, Messer SC, Garvey Wilson AL, et al. Strengthening the validity of population-based suicide rate comparisons: an il- lustration using U.S. military and civilian data. Suicide Life Threat Behav. 2006;36:182-191.
37. Miller M, Azrael D, Barber C, et al. A call to link data to answer pressing questions about suicide risk among veterans. Am J Pub Health. 2012;102(suppl 1):S20-S22.
38. Department of Veterans Affairs. Report of the Blue Ribbon Work Group on suicide prevention in the veteran population. June 2008. Available at: http://www.mentalhealth.va.gov/suicide_ prevention/Blue_Ribbon_Report-FINAL_June-30-08.pdf. Accessed July 18, 2013.
39. Kinn JT, Luxton DD, Reger MA, et al. Department of Defense sui- cide event report: calendar year 2010 annual report. September 2011. Available at: http://t2health.org/sites/default/files/dodser/ DoDSER_2010_Annual_Report.pdf. Accessed June 19, 2013.
40. Fontana A, Rosenheck R. Treatment-seeking veterans of Iraq and Afghanistan: comparison with veterans of previous wars. J Nerv Ment Dis. 2008;196:513-521.
41. Seal KH, Cohen G, Waldrop A, et al. Substance use disorders in Iraq and Afghanistan veterans in VA healthcare, 2001-2010: implications for screening, diagnosis and treatment. Drug Alcohol Depend. 2011;116:93-101.
42. Mirza RA, Eick-Cost A, Otto JL. The risk of mental health disor- ders among U.S. military personnel infected with human immu- nodeficiency virus, active component, U.S. Armed Forces, 2000- 2011. MSMR. 2012;19:10-13.
43. Bohnert AS, Ilgen MA, Bossarte RM, et al. Veteran status and alco- hol use in men in the United States. Mil Med. 2012;177:198-203.
44. Erbes CR, Kaler ME, Schult T, et al. Mental health diagnosis and occupational functioning in National Guard/Reserve veterans re- turning from Iraq. J Rehabil Res Dev. 2011;48:1159-1170.
45. Stecker T, Fortney J, Owen R, et al. Co-occurring medical, psychi- atric, and alcohol-related disorders among veterans returning from Iraq and Afghanistan. Psychosomatics. 2010;51:503-507.
46. Seal KH, Shi Y, Cohen G, et al. Association of mental health dis- orders with prescription opioids and high-risk opioid use in US veterans of Iraq and Afghanistan. JAMA. 2012;307:940-947.
47. Praveen KT, Law F, O’Shea J, et al. Opioid dependence. Am Fam Physician. 2012;86:565-566.
48. Bradley KA, Bush KR, Epler AJ, et al. Two brief alcohol-screening tests from the Alcohol Use Disorders Identification Test (AUDIT): validation in a female Veterans Affairs patient population. Arch Intern Med. 2003;163:821-829.
49. Bush K, Kivlahan DR, McDonell MB, et al. The AUDIT alcohol consumption questions (AUDIT-C): an effective brief screening test for problem drinking. Ambulatory Care Quality Improvement Project (ACQUIP). Alcohol Use Disorders Identification Test. Arch Intern Med. 1998;158:1789-1795.
50. Farrell M, Howes S, Taylor C, et al. Substance misuse and psychi- atric comorbidity: an overview of the OPCS National Psychiatric Morbidity Survey. Addict Behav. 1998;23:909-918.
51. Toombs JD, Kral LA. Methadone treatment for pain states. Am Fam Physician. 2005;71:1353-1358.
52. Center for Substance Abuse Treatment. Clinical Guidelines for the Use of Buprenorphine in the Treatment of Opioid Addiction. Treatment Improvement Protocol (TIP) series 40. DHHS pub- lication (SMA) 04-3939. Rockville, MD: Substance Abuse and Mental Health Services Administration; 2004. Available at: http:// buprenorphine.samhsa.gov/Bup_Guidelines.pdf. Accessed June 19, 2013.
53. U.S.DepartmentofHealthandHumanServices,SubstanceAbuse and Mental Health Services Administration Web site. About buprenorphine therapy. Available at: http://buprenorphine. samhsa.gov/about.html. Accessed June 19, 2013.
54. Volpicelli JR, Alterman AI, Hayashida M, et al. Naltrexone in the treatment of alcohol dependence. Arch Gen Psychiatry. 1992;49:876-880.
55. O’Brien CP, Volpicelli LA, Volpicelli JR. Naltrexone in the treat- ment of alcoholism: a clinical review. Alcohol. 1996;13:35-39.
56. Jonas DE, Garbutt JC, Amick HR, et al. Behavioral counseling after screening for alcohol misuse in primary care: a systematic review and meta-analysis for the U.S. Preventive Services Task Force. Ann Intern Med. 2012;157:645-654.
57. van Dam D, Vedel E, Ehring T, et al. Psychological treatments for concurrent posttraumatic stress disorder and substance use dis- order: a systematic review. Clin Psychol Rev. 2012;32:202-214.
Weight-loss program for veterans cut diabetes risk
CHICAGO – A low-cost lifestyle intervention program designed for real-world application in obese and overweight patients achieved sustained weight loss and a reduced risk of developing diabetes in a large 3-year observational study.
The MOVE! program (Managing Overweight and/or Obesity in Veterans Everywhere) was launched in 2005 in 130 hospitals and clinics in the Department of Veterans Affairs (VA) system, the nation’s largest integrated health care system. MOVE! is based upon principles proven effective in the National Institutes of Health’s landmark Diabetes Prevention Program (DPP), an intensive diet and exercise program that achieved a sustained 34% reduction in the incidence of diabetes at 10 years.
The problem, according to Sandra L. Jackson, is that the DPP and other successful research projects use patients who volunteer to participate and thus may be particularly highly motivated.
"We know little about the results that can be achieved in real-world health care settings, where participants are patients and their health care providers recommend a change in lifestyle," she noted in presenting the MOVE! results at the annual scientific sessions of the American Diabetes Association.
That was the impetus for her study of 3-year outcomes nationally in MOVE! The results were so impressive that MOVE! was named one of the five studies selected for an encore presentation at the ADA President’s Oral Session out of the more than 2,000 studies presented at the conference.
The MOVE! program consists of 8-12 weekly group sessions focused primarily on physical activity and nutrition. More than 400,000 veterans have participated in MOVE! since 2005. They signed up for one of two reasons: either they were obese, or they were overweight with a weight-related chronic health condition, such as osteoarthritis, coronary artery disease, diabetes, or sleep apnea. At their first MOVE! session, 38% of participants were known to have diabetes.
Ms. Jackson, a PhD candidate at Emory University, Atlanta, reported on the 135,686 MOVE! participants with 3-year follow-up data, comparing their outcomes with roughly 1.5 million VA patients who were MOVE! eligible but chose not to participate.
A total of 8.7% of participants were classified as intense and sustained in their involvement with the program based upon their having attended at least eight sessions within a 6-month period, with at least 129 days between the first and last session. Everyone else was categorized as "less involved."
Among the overall group of nearly 136,000 patients, mean body mass index dropped over the course of 3 years of follow-up from 36.3 kg/m2 to 35.8 kg/m2, representing a 1.3% loss in body weight. The intense and sustained participants lost an average of 2.5% of their initial body weight, compared with a 1% loss in the less-involved subjects.
The active participants typically experienced virtually all of their weight loss during the first 6 months, then maintained their new body weight for the next 2.5 years.
Overall, three-quarters of the intense and sustained participants lost any weight or maintained their baseline body weight over 3 years. In contrast, two-thirds of the less-active participants did so.
Diabetes risk moves down
In a multivariate analysis adjusted for baseline BMI, age, sex, and the use of medications that affect body weight, patients who lost any weight or remained weight stable over 3 years were 16% less likely to develop new-onset diabetes than those who gained weight.
The intense and sustained MOVE! participants were significantly more likely to experience a clinically meaningful weight loss of 5%. A total of 28% of them did so, compared with 16% of the less-active participants and 11% of MOVE!-eligible nonparticipants.
MOVE! enrollees with diabetes at baseline were more likely to become intense and sustained participants than those without baseline diabetes, by a margin of 9.6% to 7.8%. Overall, patients with diabetes also lost more weight: a mean of 1.7% body weight at 3 years, compared with a 0.9% drop in nondiabetic participants.
Among the 66,933 MOVE! participants without diabetes at baseline, the 3-year incidence of diabetes was 18.7%. A progressive relationship existed between weight change and diabetes incidence. At the extremes, participants who lost at least 10% of their initial body weight had a 3-year incidence of diabetes of 15%, while those with a 10% or greater weight gain had a 22% incidence of diabetes.
In a multivariate analysis, the intense and sustained participants in MOVE! had a 33% reduction in incident diabetes over 3 years, compared with the roughly 1.5 million VA patients who were MOVE! eligible but didn’t participate.
Ms. Jackson noted that a major limitation of the MOVE! program is that less than 10% of participants are actively involved. Those are the ones who reap the greatest benefits in terms of weight loss and reduced risk of diabetes.
"We need to learn how to encourage participation," she observed.
MOVE! to more health plans?
Ms. Jackson and her coinvestigators see MOVE! as well suited for adoption by other large national health care organizations.
MOVE! differs from the DPP in several key ways. It’s shorter, with 8-12 weekly group sessions largely devoted to nutrition and physical activity, compared with 16 sessions in DPP. The MOVE! classes can be taken in any order, while the DPP program requires sessions to be done in a specific sequence. MOVE! sessions can be run by exercise physiologists, nutritionists, diabetes educators, and other professionals; DPP uses a single coach.
Also, MOVE! is less structured than the DPP in that MOVE! emphasizes individualized, patient-determined goal setting developed through motivational interviewing techniques, while the DPP features fixed, generic goals.
For example, whereas the DPP set a target of 150 minutes of moderate exercise per week, MOVE! is more inclusive. It is open to veterans for whom that exercise goal may not be achievable.
Another important difference: Eligibility for MOVE! is based upon body weight, and many participants already have diabetes. In contrast, DPP participants had to be prediabetic, Ms. Jackson noted.
Future MOVE! analyses will explore the program’s impact upon participants’ health and resource utilization.
The originality of the MOVE! program is that it allows people with or without diabetes to participate, explained session chair Dr. Elbert S. Huang of the department of medicine at the University of Chicago. In contrast, most of the classic diabetes prevention studies had very narrow entry criteria. How is it possible, he asked, for such a wide range of patients in a given class to stay on the same page in terms of goal setting?
"As a practical matter," Ms. Jackson explained, "it’s much easier for the VA system to allow all comers who are obese or overweight with a weight-related health condition to participate. The goals are individualized. The program uses the principles of motivational interviewing to ask veterans, ‘How do you want to change your life?’ "
The Department of Veterans Affairs supported the study. Ms. Jackson reported having no conflicts of interest.
CHICAGO – A low-cost lifestyle intervention program designed for real-world application in obese and overweight patients achieved sustained weight loss and a reduced risk of developing diabetes in a large 3-year observational study.
The MOVE! program (Managing Overweight and/or Obesity in Veterans Everywhere) was launched in 2005 in 130 hospitals and clinics in the Department of Veterans Affairs (VA) system, the nation’s largest integrated health care system. MOVE! is based upon principles proven effective in the National Institutes of Health’s landmark Diabetes Prevention Program (DPP), an intensive diet and exercise program that achieved a sustained 34% reduction in the incidence of diabetes at 10 years.
The problem, according to Sandra L. Jackson, is that the DPP and other successful research projects use patients who volunteer to participate and thus may be particularly highly motivated.
"We know little about the results that can be achieved in real-world health care settings, where participants are patients and their health care providers recommend a change in lifestyle," she noted in presenting the MOVE! results at the annual scientific sessions of the American Diabetes Association.
That was the impetus for her study of 3-year outcomes nationally in MOVE! The results were so impressive that MOVE! was named one of the five studies selected for an encore presentation at the ADA President’s Oral Session out of the more than 2,000 studies presented at the conference.
The MOVE! program consists of 8-12 weekly group sessions focused primarily on physical activity and nutrition. More than 400,000 veterans have participated in MOVE! since 2005. They signed up for one of two reasons: either they were obese, or they were overweight with a weight-related chronic health condition, such as osteoarthritis, coronary artery disease, diabetes, or sleep apnea. At their first MOVE! session, 38% of participants were known to have diabetes.
Ms. Jackson, a PhD candidate at Emory University, Atlanta, reported on the 135,686 MOVE! participants with 3-year follow-up data, comparing their outcomes with roughly 1.5 million VA patients who were MOVE! eligible but chose not to participate.
A total of 8.7% of participants were classified as intense and sustained in their involvement with the program based upon their having attended at least eight sessions within a 6-month period, with at least 129 days between the first and last session. Everyone else was categorized as "less involved."
Among the overall group of nearly 136,000 patients, mean body mass index dropped over the course of 3 years of follow-up from 36.3 kg/m2 to 35.8 kg/m2, representing a 1.3% loss in body weight. The intense and sustained participants lost an average of 2.5% of their initial body weight, compared with a 1% loss in the less-involved subjects.
The active participants typically experienced virtually all of their weight loss during the first 6 months, then maintained their new body weight for the next 2.5 years.
Overall, three-quarters of the intense and sustained participants lost any weight or maintained their baseline body weight over 3 years. In contrast, two-thirds of the less-active participants did so.
Diabetes risk moves down
In a multivariate analysis adjusted for baseline BMI, age, sex, and the use of medications that affect body weight, patients who lost any weight or remained weight stable over 3 years were 16% less likely to develop new-onset diabetes than those who gained weight.
The intense and sustained MOVE! participants were significantly more likely to experience a clinically meaningful weight loss of 5%. A total of 28% of them did so, compared with 16% of the less-active participants and 11% of MOVE!-eligible nonparticipants.
MOVE! enrollees with diabetes at baseline were more likely to become intense and sustained participants than those without baseline diabetes, by a margin of 9.6% to 7.8%. Overall, patients with diabetes also lost more weight: a mean of 1.7% body weight at 3 years, compared with a 0.9% drop in nondiabetic participants.
Among the 66,933 MOVE! participants without diabetes at baseline, the 3-year incidence of diabetes was 18.7%. A progressive relationship existed between weight change and diabetes incidence. At the extremes, participants who lost at least 10% of their initial body weight had a 3-year incidence of diabetes of 15%, while those with a 10% or greater weight gain had a 22% incidence of diabetes.
In a multivariate analysis, the intense and sustained participants in MOVE! had a 33% reduction in incident diabetes over 3 years, compared with the roughly 1.5 million VA patients who were MOVE! eligible but didn’t participate.
Ms. Jackson noted that a major limitation of the MOVE! program is that less than 10% of participants are actively involved. Those are the ones who reap the greatest benefits in terms of weight loss and reduced risk of diabetes.
"We need to learn how to encourage participation," she observed.
MOVE! to more health plans?
Ms. Jackson and her coinvestigators see MOVE! as well suited for adoption by other large national health care organizations.
MOVE! differs from the DPP in several key ways. It’s shorter, with 8-12 weekly group sessions largely devoted to nutrition and physical activity, compared with 16 sessions in DPP. The MOVE! classes can be taken in any order, while the DPP program requires sessions to be done in a specific sequence. MOVE! sessions can be run by exercise physiologists, nutritionists, diabetes educators, and other professionals; DPP uses a single coach.
Also, MOVE! is less structured than the DPP in that MOVE! emphasizes individualized, patient-determined goal setting developed through motivational interviewing techniques, while the DPP features fixed, generic goals.
For example, whereas the DPP set a target of 150 minutes of moderate exercise per week, MOVE! is more inclusive. It is open to veterans for whom that exercise goal may not be achievable.
Another important difference: Eligibility for MOVE! is based upon body weight, and many participants already have diabetes. In contrast, DPP participants had to be prediabetic, Ms. Jackson noted.
Future MOVE! analyses will explore the program’s impact upon participants’ health and resource utilization.
The originality of the MOVE! program is that it allows people with or without diabetes to participate, explained session chair Dr. Elbert S. Huang of the department of medicine at the University of Chicago. In contrast, most of the classic diabetes prevention studies had very narrow entry criteria. How is it possible, he asked, for such a wide range of patients in a given class to stay on the same page in terms of goal setting?
"As a practical matter," Ms. Jackson explained, "it’s much easier for the VA system to allow all comers who are obese or overweight with a weight-related health condition to participate. The goals are individualized. The program uses the principles of motivational interviewing to ask veterans, ‘How do you want to change your life?’ "
The Department of Veterans Affairs supported the study. Ms. Jackson reported having no conflicts of interest.
CHICAGO – A low-cost lifestyle intervention program designed for real-world application in obese and overweight patients achieved sustained weight loss and a reduced risk of developing diabetes in a large 3-year observational study.
The MOVE! program (Managing Overweight and/or Obesity in Veterans Everywhere) was launched in 2005 in 130 hospitals and clinics in the Department of Veterans Affairs (VA) system, the nation’s largest integrated health care system. MOVE! is based upon principles proven effective in the National Institutes of Health’s landmark Diabetes Prevention Program (DPP), an intensive diet and exercise program that achieved a sustained 34% reduction in the incidence of diabetes at 10 years.
The problem, according to Sandra L. Jackson, is that the DPP and other successful research projects use patients who volunteer to participate and thus may be particularly highly motivated.
"We know little about the results that can be achieved in real-world health care settings, where participants are patients and their health care providers recommend a change in lifestyle," she noted in presenting the MOVE! results at the annual scientific sessions of the American Diabetes Association.
That was the impetus for her study of 3-year outcomes nationally in MOVE! The results were so impressive that MOVE! was named one of the five studies selected for an encore presentation at the ADA President’s Oral Session out of the more than 2,000 studies presented at the conference.
The MOVE! program consists of 8-12 weekly group sessions focused primarily on physical activity and nutrition. More than 400,000 veterans have participated in MOVE! since 2005. They signed up for one of two reasons: either they were obese, or they were overweight with a weight-related chronic health condition, such as osteoarthritis, coronary artery disease, diabetes, or sleep apnea. At their first MOVE! session, 38% of participants were known to have diabetes.
Ms. Jackson, a PhD candidate at Emory University, Atlanta, reported on the 135,686 MOVE! participants with 3-year follow-up data, comparing their outcomes with roughly 1.5 million VA patients who were MOVE! eligible but chose not to participate.
A total of 8.7% of participants were classified as intense and sustained in their involvement with the program based upon their having attended at least eight sessions within a 6-month period, with at least 129 days between the first and last session. Everyone else was categorized as "less involved."
Among the overall group of nearly 136,000 patients, mean body mass index dropped over the course of 3 years of follow-up from 36.3 kg/m2 to 35.8 kg/m2, representing a 1.3% loss in body weight. The intense and sustained participants lost an average of 2.5% of their initial body weight, compared with a 1% loss in the less-involved subjects.
The active participants typically experienced virtually all of their weight loss during the first 6 months, then maintained their new body weight for the next 2.5 years.
Overall, three-quarters of the intense and sustained participants lost any weight or maintained their baseline body weight over 3 years. In contrast, two-thirds of the less-active participants did so.
Diabetes risk moves down
In a multivariate analysis adjusted for baseline BMI, age, sex, and the use of medications that affect body weight, patients who lost any weight or remained weight stable over 3 years were 16% less likely to develop new-onset diabetes than those who gained weight.
The intense and sustained MOVE! participants were significantly more likely to experience a clinically meaningful weight loss of 5%. A total of 28% of them did so, compared with 16% of the less-active participants and 11% of MOVE!-eligible nonparticipants.
MOVE! enrollees with diabetes at baseline were more likely to become intense and sustained participants than those without baseline diabetes, by a margin of 9.6% to 7.8%. Overall, patients with diabetes also lost more weight: a mean of 1.7% body weight at 3 years, compared with a 0.9% drop in nondiabetic participants.
Among the 66,933 MOVE! participants without diabetes at baseline, the 3-year incidence of diabetes was 18.7%. A progressive relationship existed between weight change and diabetes incidence. At the extremes, participants who lost at least 10% of their initial body weight had a 3-year incidence of diabetes of 15%, while those with a 10% or greater weight gain had a 22% incidence of diabetes.
In a multivariate analysis, the intense and sustained participants in MOVE! had a 33% reduction in incident diabetes over 3 years, compared with the roughly 1.5 million VA patients who were MOVE! eligible but didn’t participate.
Ms. Jackson noted that a major limitation of the MOVE! program is that less than 10% of participants are actively involved. Those are the ones who reap the greatest benefits in terms of weight loss and reduced risk of diabetes.
"We need to learn how to encourage participation," she observed.
MOVE! to more health plans?
Ms. Jackson and her coinvestigators see MOVE! as well suited for adoption by other large national health care organizations.
MOVE! differs from the DPP in several key ways. It’s shorter, with 8-12 weekly group sessions largely devoted to nutrition and physical activity, compared with 16 sessions in DPP. The MOVE! classes can be taken in any order, while the DPP program requires sessions to be done in a specific sequence. MOVE! sessions can be run by exercise physiologists, nutritionists, diabetes educators, and other professionals; DPP uses a single coach.
Also, MOVE! is less structured than the DPP in that MOVE! emphasizes individualized, patient-determined goal setting developed through motivational interviewing techniques, while the DPP features fixed, generic goals.
For example, whereas the DPP set a target of 150 minutes of moderate exercise per week, MOVE! is more inclusive. It is open to veterans for whom that exercise goal may not be achievable.
Another important difference: Eligibility for MOVE! is based upon body weight, and many participants already have diabetes. In contrast, DPP participants had to be prediabetic, Ms. Jackson noted.
Future MOVE! analyses will explore the program’s impact upon participants’ health and resource utilization.
The originality of the MOVE! program is that it allows people with or without diabetes to participate, explained session chair Dr. Elbert S. Huang of the department of medicine at the University of Chicago. In contrast, most of the classic diabetes prevention studies had very narrow entry criteria. How is it possible, he asked, for such a wide range of patients in a given class to stay on the same page in terms of goal setting?
"As a practical matter," Ms. Jackson explained, "it’s much easier for the VA system to allow all comers who are obese or overweight with a weight-related health condition to participate. The goals are individualized. The program uses the principles of motivational interviewing to ask veterans, ‘How do you want to change your life?’ "
The Department of Veterans Affairs supported the study. Ms. Jackson reported having no conflicts of interest.
AT THE ADA ANNUAL SCIENTIFIC SESSIONS
Overtreatment common in high-risk diabetes patients
Earn 0.25 hours AMA PRA Category 1 credit: Read this article, and click the link at the end to take the post-test.
CHICAGO – Glycemic overtreatment of high-risk diabetic patients is rampant within the Veterans Affairs health care system, according to a national study.
Moreover, because many of these diabetic veterans who are at high risk for serious hypoglycemia are also Medicare eligible, it’s quite likely that overtreatment is a common problem in the Medicare population as well.
"I think these findings are directly relevant to Medicare," Dr. Leonard M. Pogach said in presenting the VA study results at the annual scientific sessions of the American Diabetes Association.
Current performance measures do not assess potential overtreatment of high-risk diabetic patients in either the VA or Medicare populations. But as a result of the VA study findings, joint federal efforts are underway to address this shortcoming, according to Dr. Pogach, who is national program director for endocrinology and diabetes at the Veterans Health Administration and professor of medicine at the New Jersey Medical School, Newark.
He cited as a major impetus for the VA study an eye-opening 2011 report by investigators at the Centers for Disease Control and Prevention that identified insulin and sulfonylurea drugs as the combined number-two cause of emergency hospitalizations for adverse drug events in the United States, second only to warfarin (N. Engl. J. Med. 2011;365:2002-12).
As part of the Choosing Wisely campaign, the American Geriatrics Society recommends that medications other than metformin not be routinely used to lower hemoglobin A1c below 7.5% in patients older than 65 years. The group further recommends an HbA1c target of 8%-9% for those with serious comorbid conditions.
Similarly, the ADA now recommends an HbA1c target of 7.5%-8.0% or slightly more in patients at increased risk for serious hypoglycemia or with reduced life expectancy, rather than its former universal goal of less than 7.0%.
For purposes of the VA study, Dr. Pogach and coinvestigators defined "high-risk" diabetes patients as those receiving insulin and/or sulfonylurea therapy and who are at least 70 years of age, have renal impairment as reflected in a serum creatinine level greater than 1.7 mg/dL, or have been diagnosed with cognitive impairment or dementia.
During the study year of 2009, a total of 285,476 of 652,738 VA patients, or 44%,with diabetes on insulin and/or sulfonylurea therapy qualified as high risk, based upon that definition. They received treatment in 139 VA facilities in 21 regions nationally.
Overall, 48% of these high-risk diabetes patients were likely being overtreated, as reflected in an HbA1c below 7.0%. Moreover, one-quarter of the high-risk group had an HbA1c below 6.5%, and 10% had an HbA1c of less than 6.0%, Dr. Pogach reported.
He added that these results likely underestimate the true extent of the glycemic overtreatment problem, because he and his coinvestigators defined "high risk" quite conservatively – based simply upon advanced age, renal dysfunction, and cognitive impairment.
Had they included other reasonable criteria – specifically, diminished life expectancy; stroke and other major neurologic disorders; cardiovascular disease; major depression; alcohol and/or drug abuse; and advanced diabetic complications – the proportion of the 652,738 diabetic VA patients on insulin and/or sulfonylurea therapy who would have qualified as being at high risk for serious hypoglycemia would have climbed from 44% all the way up to 71%.
The variation in overtreatment rates from region to region within the VA system was fairly tight. The range was much greater among facilities within a given regional district, where overtreatment rates varied from a low of 37% at a select few facilities to as high as 63%.
"We were able to identify several facilities with statistically remarkably lower rates of overtreatment than in the larger regions they lie in," Dr. Pogach noted. "Those are the sites where we might want to do site visits and qualitative studies to try to figure out what made them different. What is it about their culture or their patients? What happened in those places that we might subsequently want to replicate?"
Of note, the VA never adopted the one-size-fits-all goal of an HbA1c below 7.0% that the ADA recommended until recently. For more than a decade, VA clinical practice guidelines have included stratified glycemic targets based on comorbidities and life expectancy.
One audience member took issue with Dr. Pogach’s use of the word "overtreatment."
"Overtreatment is really a somewhat pejorative term," he argued. "I think you really have to have data showing that we are harming these people, not in terms of the surrogate outcome of hypoglycemia, but in real negative consequences."
Dr. Pogach was quick to rebut.
"I think the published results of the ACCORD and ADVANCE studies demonstrate that there’s a very strong association between cardiovascular morbidity and mortality and self-reported hypoglycemia, with adjusted odds ratios of about two and one-half," he explained. "I don’t think those data will ever be beat. We’re never going to have a randomized trial. So, I think the association with adverse outcomes is very clear, although we don’t know if hypoglycemia is the proximate cause or not."
The Department of Veterans Affairs supported the study. Dr. Pogach reported having no financial conflicts of interest.
To earn 0.25 hours AMA PRA Category 1 credit after reading this article, take the post-test here.
Earn 0.25 hours AMA PRA Category 1 credit: Read this article, and click the link at the end to take the post-test.
CHICAGO – Glycemic overtreatment of high-risk diabetic patients is rampant within the Veterans Affairs health care system, according to a national study.
Moreover, because many of these diabetic veterans who are at high risk for serious hypoglycemia are also Medicare eligible, it’s quite likely that overtreatment is a common problem in the Medicare population as well.
"I think these findings are directly relevant to Medicare," Dr. Leonard M. Pogach said in presenting the VA study results at the annual scientific sessions of the American Diabetes Association.
Current performance measures do not assess potential overtreatment of high-risk diabetic patients in either the VA or Medicare populations. But as a result of the VA study findings, joint federal efforts are underway to address this shortcoming, according to Dr. Pogach, who is national program director for endocrinology and diabetes at the Veterans Health Administration and professor of medicine at the New Jersey Medical School, Newark.
He cited as a major impetus for the VA study an eye-opening 2011 report by investigators at the Centers for Disease Control and Prevention that identified insulin and sulfonylurea drugs as the combined number-two cause of emergency hospitalizations for adverse drug events in the United States, second only to warfarin (N. Engl. J. Med. 2011;365:2002-12).
As part of the Choosing Wisely campaign, the American Geriatrics Society recommends that medications other than metformin not be routinely used to lower hemoglobin A1c below 7.5% in patients older than 65 years. The group further recommends an HbA1c target of 8%-9% for those with serious comorbid conditions.
Similarly, the ADA now recommends an HbA1c target of 7.5%-8.0% or slightly more in patients at increased risk for serious hypoglycemia or with reduced life expectancy, rather than its former universal goal of less than 7.0%.
For purposes of the VA study, Dr. Pogach and coinvestigators defined "high-risk" diabetes patients as those receiving insulin and/or sulfonylurea therapy and who are at least 70 years of age, have renal impairment as reflected in a serum creatinine level greater than 1.7 mg/dL, or have been diagnosed with cognitive impairment or dementia.
During the study year of 2009, a total of 285,476 of 652,738 VA patients, or 44%,with diabetes on insulin and/or sulfonylurea therapy qualified as high risk, based upon that definition. They received treatment in 139 VA facilities in 21 regions nationally.
Overall, 48% of these high-risk diabetes patients were likely being overtreated, as reflected in an HbA1c below 7.0%. Moreover, one-quarter of the high-risk group had an HbA1c below 6.5%, and 10% had an HbA1c of less than 6.0%, Dr. Pogach reported.
He added that these results likely underestimate the true extent of the glycemic overtreatment problem, because he and his coinvestigators defined "high risk" quite conservatively – based simply upon advanced age, renal dysfunction, and cognitive impairment.
Had they included other reasonable criteria – specifically, diminished life expectancy; stroke and other major neurologic disorders; cardiovascular disease; major depression; alcohol and/or drug abuse; and advanced diabetic complications – the proportion of the 652,738 diabetic VA patients on insulin and/or sulfonylurea therapy who would have qualified as being at high risk for serious hypoglycemia would have climbed from 44% all the way up to 71%.
The variation in overtreatment rates from region to region within the VA system was fairly tight. The range was much greater among facilities within a given regional district, where overtreatment rates varied from a low of 37% at a select few facilities to as high as 63%.
"We were able to identify several facilities with statistically remarkably lower rates of overtreatment than in the larger regions they lie in," Dr. Pogach noted. "Those are the sites where we might want to do site visits and qualitative studies to try to figure out what made them different. What is it about their culture or their patients? What happened in those places that we might subsequently want to replicate?"
Of note, the VA never adopted the one-size-fits-all goal of an HbA1c below 7.0% that the ADA recommended until recently. For more than a decade, VA clinical practice guidelines have included stratified glycemic targets based on comorbidities and life expectancy.
One audience member took issue with Dr. Pogach’s use of the word "overtreatment."
"Overtreatment is really a somewhat pejorative term," he argued. "I think you really have to have data showing that we are harming these people, not in terms of the surrogate outcome of hypoglycemia, but in real negative consequences."
Dr. Pogach was quick to rebut.
"I think the published results of the ACCORD and ADVANCE studies demonstrate that there’s a very strong association between cardiovascular morbidity and mortality and self-reported hypoglycemia, with adjusted odds ratios of about two and one-half," he explained. "I don’t think those data will ever be beat. We’re never going to have a randomized trial. So, I think the association with adverse outcomes is very clear, although we don’t know if hypoglycemia is the proximate cause or not."
The Department of Veterans Affairs supported the study. Dr. Pogach reported having no financial conflicts of interest.
To earn 0.25 hours AMA PRA Category 1 credit after reading this article, take the post-test here.
Earn 0.25 hours AMA PRA Category 1 credit: Read this article, and click the link at the end to take the post-test.
CHICAGO – Glycemic overtreatment of high-risk diabetic patients is rampant within the Veterans Affairs health care system, according to a national study.
Moreover, because many of these diabetic veterans who are at high risk for serious hypoglycemia are also Medicare eligible, it’s quite likely that overtreatment is a common problem in the Medicare population as well.
"I think these findings are directly relevant to Medicare," Dr. Leonard M. Pogach said in presenting the VA study results at the annual scientific sessions of the American Diabetes Association.
Current performance measures do not assess potential overtreatment of high-risk diabetic patients in either the VA or Medicare populations. But as a result of the VA study findings, joint federal efforts are underway to address this shortcoming, according to Dr. Pogach, who is national program director for endocrinology and diabetes at the Veterans Health Administration and professor of medicine at the New Jersey Medical School, Newark.
He cited as a major impetus for the VA study an eye-opening 2011 report by investigators at the Centers for Disease Control and Prevention that identified insulin and sulfonylurea drugs as the combined number-two cause of emergency hospitalizations for adverse drug events in the United States, second only to warfarin (N. Engl. J. Med. 2011;365:2002-12).
As part of the Choosing Wisely campaign, the American Geriatrics Society recommends that medications other than metformin not be routinely used to lower hemoglobin A1c below 7.5% in patients older than 65 years. The group further recommends an HbA1c target of 8%-9% for those with serious comorbid conditions.
Similarly, the ADA now recommends an HbA1c target of 7.5%-8.0% or slightly more in patients at increased risk for serious hypoglycemia or with reduced life expectancy, rather than its former universal goal of less than 7.0%.
For purposes of the VA study, Dr. Pogach and coinvestigators defined "high-risk" diabetes patients as those receiving insulin and/or sulfonylurea therapy and who are at least 70 years of age, have renal impairment as reflected in a serum creatinine level greater than 1.7 mg/dL, or have been diagnosed with cognitive impairment or dementia.
During the study year of 2009, a total of 285,476 of 652,738 VA patients, or 44%,with diabetes on insulin and/or sulfonylurea therapy qualified as high risk, based upon that definition. They received treatment in 139 VA facilities in 21 regions nationally.
Overall, 48% of these high-risk diabetes patients were likely being overtreated, as reflected in an HbA1c below 7.0%. Moreover, one-quarter of the high-risk group had an HbA1c below 6.5%, and 10% had an HbA1c of less than 6.0%, Dr. Pogach reported.
He added that these results likely underestimate the true extent of the glycemic overtreatment problem, because he and his coinvestigators defined "high risk" quite conservatively – based simply upon advanced age, renal dysfunction, and cognitive impairment.
Had they included other reasonable criteria – specifically, diminished life expectancy; stroke and other major neurologic disorders; cardiovascular disease; major depression; alcohol and/or drug abuse; and advanced diabetic complications – the proportion of the 652,738 diabetic VA patients on insulin and/or sulfonylurea therapy who would have qualified as being at high risk for serious hypoglycemia would have climbed from 44% all the way up to 71%.
The variation in overtreatment rates from region to region within the VA system was fairly tight. The range was much greater among facilities within a given regional district, where overtreatment rates varied from a low of 37% at a select few facilities to as high as 63%.
"We were able to identify several facilities with statistically remarkably lower rates of overtreatment than in the larger regions they lie in," Dr. Pogach noted. "Those are the sites where we might want to do site visits and qualitative studies to try to figure out what made them different. What is it about their culture or their patients? What happened in those places that we might subsequently want to replicate?"
Of note, the VA never adopted the one-size-fits-all goal of an HbA1c below 7.0% that the ADA recommended until recently. For more than a decade, VA clinical practice guidelines have included stratified glycemic targets based on comorbidities and life expectancy.
One audience member took issue with Dr. Pogach’s use of the word "overtreatment."
"Overtreatment is really a somewhat pejorative term," he argued. "I think you really have to have data showing that we are harming these people, not in terms of the surrogate outcome of hypoglycemia, but in real negative consequences."
Dr. Pogach was quick to rebut.
"I think the published results of the ACCORD and ADVANCE studies demonstrate that there’s a very strong association between cardiovascular morbidity and mortality and self-reported hypoglycemia, with adjusted odds ratios of about two and one-half," he explained. "I don’t think those data will ever be beat. We’re never going to have a randomized trial. So, I think the association with adverse outcomes is very clear, although we don’t know if hypoglycemia is the proximate cause or not."
The Department of Veterans Affairs supported the study. Dr. Pogach reported having no financial conflicts of interest.
To earn 0.25 hours AMA PRA Category 1 credit after reading this article, take the post-test here.
AT THE ADA ANNUAL SCIENTIFIC SESSIONS
Imperforate hymen in your adolescent patient: Don’t miss the diagnosis
Many gynecologists encounter imperforate hymen, a congenital vaginal anomaly, in general practice. As such, it is important to have a basic understanding of the condition and to be aware of appropriate screening, evaluation, and management. This knowledge will allow you to differentiate imperforate hymen from more complex anomalies—preventing significant morbidity that could result from performing the wrong surgical procedure on this condition—and to provide optimal surgical management.
How often and why does it occur?
Imperforate hymen occurs in approximately 1/1000 newborn girls. It is the most common obstructive anomaly of the female reproductive tract.1,2
The hymen consists of fibrous connective tissue attached to the vaginal wall. In the perinatal period, the hymen serves to separate the vaginal lumen from the urogenital sinus (UGS); this is usually perforated during embryonic life by canalization of the most caudal portion of the vaginal plate at the UGS. This establishes a connection between the lumen of the vaginal canal and the vaginal vestibule.3 Failure of the hymen to perforate completely in the perinatal period can result in varying anomalies, including imperforate (FIGURE 1), microperforate, cribiform, or septated hymen.
Figure 1. Imperforate hymen
How does it present?
Its presentation is variable and frequently asymptomatic in infants and children.4 As a result, the diagnosis is often delayed until puberty.3
In infancy. Newborns typically will present with a hymenal bulge from hydrocolpos or mucocolpos, which result from maternal estrogen secretion on the newborn’s vaginal epithelium.5 This is usually asymptomatic and self limited.
Rarely, large hydrocolpos/mucocolpos may become symptomatic and can lead to urinary obstruction, or they can present as an abdominal mass or intestinal obstruction.4
In adolescence. The majority of adolescents will present with cyclic or persistent pelvic pain and primary amenorrhea. If significant hematometra is present, an abdominal mass also may be palpated. In extreme cases, the patient may present with mass effect symptoms, including back pain, pain with defecation, constipation, nausea and vomiting, urinary retention, or hydronephrosis.6 Retrograde passage of blood into the fallopian tubes can cause hematosalpinx, which can lead to endometriosis and adhesion formation. Blood also may pass freely into the peritoneal cavity, forming hemoperitoneum.3
Related article: Your age-based guide to comprehensive well-woman care
Robert L. Barbieri, MD (October 2012)
Imperforate hymen, vaginal septum, or distal vaginal atresia?
When in doubt, refer. Imperforate hymen can be confused with distal vaginal atresia or low transverse vaginal septum. Often, the patient may present with similar signs and symptoms in all 3 cases. Accurately differentiating imperforate hymen from the former two more complex congenital anomalies prior to surgery is of utmost importance because management is very different, and performing the wrong procedure can result in serious morbidity. As such, it is important to appropriately define the anatomy and refer the complex cases to a specialist comfortable and skilled in managing congenital anomalies, usually a pediatric and adolescent gynecologist or reproductive endocrinologist.
Imperforate hymen
Examination of the external genitalia reveals a perineal bulge secondary to hematocolpos.7 This finding, coupled with a rectal examination and pelvic ultrasonography is usually sufficient to make the diagnosis.6,8 However, magnetic resonance imaging (MRI) of the pelvis should be obtained in cases where the diagnosis is uncertain or the physical exam is more consistent with vaginal septum or agenesis.
Transverse vaginal septum
A reverse septum results from failure of the müllerian duct derivatives and UGS to fuse or canalize. This can occur in the lower, middle, or upper portion of the vagina, and septa may be thick or thin.6 Low transverse septa are more easily confused with imperforate hymen. Examination usually reveals a normal hymen with a short vagina posteriorly. In cases of extreme hematocolpos, vaginal septa also may present with a perineal bulge but, again, this will be posterior to a normal hymen.
Distal vaginal atresia
This condition occurs during embryonic development when the UGS fails to contribute to the lower portion of the vagina (FIGURE 2).5 In cases of distal vaginal atresia there is a lack of vaginal orifice, or only a vaginal dimple may be present.5,6 Rectovaginal examination will reveal a palpable mass if the upper vagina is distended with blood.6
Figure 2. Lower vaginal atresia
MRI is vital to firm diagnosis
In addition to pelvic ultrasonography, pelvic MRI is necessary to delineate the anatomy with both vaginal septum (FIGURE 3) and lower vaginal atresia (FIGURE 4), as preoperative evaluation of location and thickness of a vaginal septum as well as measurement of the total length of agenesis is imperative.6-8 Misdiagnosis of the vaginal septa or atresia as an imperforate hymen can lead to significant scarring and stenosis and can make corrective surgical procedures difficult or suboptimal.
| Figure 3. MRI of transverse vaginal septum |
Figure 4. MRI of lower vaginal atresia
Surgical management: hymenectomy
Imperforate hymen is managed surgically with hymenectomy. Repair is generally reserved for the newborn period or, ideally, in adolescence, as at puberty the presence of estrogen aids in surgical repair and healing.5 Simple aspiration of hematocolpos/ mucocolpos can lead to ascending infection, and pyocolpos and should be avoided.6
The goal of hymenectomy is to:
-
open the hymeneal membrane to allow egress of fluid and menstrual flow
-
allow for tampon use
The procedure is relatively straightforward and usually is performed under general anesthesia, although regional anesthesia also is an option.
Steps to the varying hymenectomy incisions
Cruciate incision
1. Incise the hymen at the 2-, 4-, 8-, and 10-o’clock positions into four quadrants.
2. Excise the quadrants along the lateral wall of the vagina.
Elliptical incision
1. Make a circumferential incision with the Bovie electrocautery, incising the hymenal membrane close to the hymenal ring.
U-incision
1. Similar to the elliptical incision, use the Bovie electrocautery to incise the tissue close to the hymenal ring posterior and laterally in a “u” shape.
2. Make a horizontal incision superiorly to remove the extra tissue.
Vertical incision
This incision has been described in cases where there is an attempt to spare the hymen for religious or cultural preference.
1. Make a midline vertical hymenotomy less than 1 cm. Drain the borders of the hymen.
2. Apply suture obliquely to form a circular opening.
References
1. Dominguez C, Rock J, Horowitz I. Surgical conditions of the vagina and urethra. In: TeLinde’s Operative Gynecology. 10th ed. Philadelphia, PA: Lippincott Williams and Wilkins; 1997.
2. Basaran M, Usal D, Aydemir C. Hymen sparing surgery for imperforate hymen: case reports and review of literature. J Pediatr Adolesc Gynecol. 2009;22(4):e61–e64.
Tips to a successful procedure
Ensure adequate suctioning. Before starting the procedure, insert a Foley catheter to completely drain the bladder and delineate the urethra. Making an initial incision into the hymen usually results in the expulsion of the old blood and mucus, which can be very thick and viscous; therefore, it is important to have adequate suction tubing.
Prevent scarring. After evacuating the old blood and mucus, excise the hymeneal membrane with a cruciate incision as is traditionally described. Alternatively, some experts use an elliptical incision or u-incision. (See “Steps to the varying hymenectomy incisions”.) Prevent excision of the hymenal tissue too close to the vaginal mucosa, as this can lead to scarring and stenosis and dyspareunia.3
Suturing the mucosal margins is likely unnecessary in adolescent patients. After excision of the hymenal tissue, one option is to suture the mucosal margins of the hymenal ring in an interrupted fashion with a fine, delayed-absorbable suture. Alternatively, at our institution, where we employ the u-incision (FIGURE 5), we assure hemostasis of the mucosal margins and do not suture the margins. Suturing the margins is believed to prevent adherence of the edges; however, in the pubertal girl, adherence is unlikely secondary to estrogen exposure.
Figure 5. Surgical correction with u-incision
Avoid infection; do not irrigate. We do not recommend that you irrigate the vagina and perform unnecessary uterine manipulation, as this may introduce bacteria into the dilated cervix and uterus.3,8
Septate/microperforate/cribiform hymen
These other hymeneal anomalies also may require surgical correction if they become clinically significant. Patients may present with difficulty inserting or removing a tampon, insertional dyspareunia, or incomplete drainage of menstrual blood.6
Imaging is usually not indicated to diagnose these hymenal anomalies, as physical examination will reveal a patent vaginal tract. A moistened Q-tip can be placed into the orifice or behind the septate hymen for confirmation (FIGURE 6).
Surgical correction of a microperforate or cribiform hymen is performed using the same principles as imperforate hymen.
Surgical correction of a septate hymen involves tying and suturing or clamping with a hemostat the upper and lower edges, with the excess hymenal tissue between the sutures then excised.8
Figure 6. Septate hymen
Postop care and follow up
Postoperative analgesia with lidocaine jelly or ice packs is usually sufficient for pain management. Reinforce proper hygienic care measures. At 2- to 3-week follow up, assess the patient for healing and evaluate the size of the hymenal orifice.
Key takeaways
-Differentiating imperforate hymen from low transverse vaginal septum or distal vaginal agenesis prior to surgery is of utmost importance because management is very different, and performing the wrong procedure can result in serious morbidity.
-With imperforate hymen, examination of the external genitalia reveals a perineal bulge secondary to hematocolpos.
-Pelvic MRI is essential to delineate the anatomy with both vaginal septum and agenesis, for preoperative evaluation of location and thickness of septum as well as measurement of total length of agenesis.
-Hymenectomy is relatively straightforward and may be performed using a cruciate, elliptical, or u-incision.
-Care should be taken to prevent excision of hymeneal tissue too close to the vaginal mucosa, as this can lead to scarring and stenosis, and later lead to dyspareunia.
Many gynecologists encounter imperforate hymen, a congenital vaginal anomaly, in general practice. As such, it is important to have a basic understanding of the condition and to be aware of appropriate screening, evaluation, and management. This knowledge will allow you to differentiate imperforate hymen from more complex anomalies—preventing significant morbidity that could result from performing the wrong surgical procedure on this condition—and to provide optimal surgical management.
How often and why does it occur?
Imperforate hymen occurs in approximately 1/1000 newborn girls. It is the most common obstructive anomaly of the female reproductive tract.1,2
The hymen consists of fibrous connective tissue attached to the vaginal wall. In the perinatal period, the hymen serves to separate the vaginal lumen from the urogenital sinus (UGS); this is usually perforated during embryonic life by canalization of the most caudal portion of the vaginal plate at the UGS. This establishes a connection between the lumen of the vaginal canal and the vaginal vestibule.3 Failure of the hymen to perforate completely in the perinatal period can result in varying anomalies, including imperforate (FIGURE 1), microperforate, cribiform, or septated hymen.
Figure 1. Imperforate hymen
How does it present?
Its presentation is variable and frequently asymptomatic in infants and children.4 As a result, the diagnosis is often delayed until puberty.3
In infancy. Newborns typically will present with a hymenal bulge from hydrocolpos or mucocolpos, which result from maternal estrogen secretion on the newborn’s vaginal epithelium.5 This is usually asymptomatic and self limited.
Rarely, large hydrocolpos/mucocolpos may become symptomatic and can lead to urinary obstruction, or they can present as an abdominal mass or intestinal obstruction.4
In adolescence. The majority of adolescents will present with cyclic or persistent pelvic pain and primary amenorrhea. If significant hematometra is present, an abdominal mass also may be palpated. In extreme cases, the patient may present with mass effect symptoms, including back pain, pain with defecation, constipation, nausea and vomiting, urinary retention, or hydronephrosis.6 Retrograde passage of blood into the fallopian tubes can cause hematosalpinx, which can lead to endometriosis and adhesion formation. Blood also may pass freely into the peritoneal cavity, forming hemoperitoneum.3
Related article: Your age-based guide to comprehensive well-woman care
Robert L. Barbieri, MD (October 2012)
Imperforate hymen, vaginal septum, or distal vaginal atresia?
When in doubt, refer. Imperforate hymen can be confused with distal vaginal atresia or low transverse vaginal septum. Often, the patient may present with similar signs and symptoms in all 3 cases. Accurately differentiating imperforate hymen from the former two more complex congenital anomalies prior to surgery is of utmost importance because management is very different, and performing the wrong procedure can result in serious morbidity. As such, it is important to appropriately define the anatomy and refer the complex cases to a specialist comfortable and skilled in managing congenital anomalies, usually a pediatric and adolescent gynecologist or reproductive endocrinologist.
Imperforate hymen
Examination of the external genitalia reveals a perineal bulge secondary to hematocolpos.7 This finding, coupled with a rectal examination and pelvic ultrasonography is usually sufficient to make the diagnosis.6,8 However, magnetic resonance imaging (MRI) of the pelvis should be obtained in cases where the diagnosis is uncertain or the physical exam is more consistent with vaginal septum or agenesis.
Transverse vaginal septum
A reverse septum results from failure of the müllerian duct derivatives and UGS to fuse or canalize. This can occur in the lower, middle, or upper portion of the vagina, and septa may be thick or thin.6 Low transverse septa are more easily confused with imperforate hymen. Examination usually reveals a normal hymen with a short vagina posteriorly. In cases of extreme hematocolpos, vaginal septa also may present with a perineal bulge but, again, this will be posterior to a normal hymen.
Distal vaginal atresia
This condition occurs during embryonic development when the UGS fails to contribute to the lower portion of the vagina (FIGURE 2).5 In cases of distal vaginal atresia there is a lack of vaginal orifice, or only a vaginal dimple may be present.5,6 Rectovaginal examination will reveal a palpable mass if the upper vagina is distended with blood.6
Figure 2. Lower vaginal atresia
MRI is vital to firm diagnosis
In addition to pelvic ultrasonography, pelvic MRI is necessary to delineate the anatomy with both vaginal septum (FIGURE 3) and lower vaginal atresia (FIGURE 4), as preoperative evaluation of location and thickness of a vaginal septum as well as measurement of the total length of agenesis is imperative.6-8 Misdiagnosis of the vaginal septa or atresia as an imperforate hymen can lead to significant scarring and stenosis and can make corrective surgical procedures difficult or suboptimal.
| Figure 3. MRI of transverse vaginal septum |
Figure 4. MRI of lower vaginal atresia
Surgical management: hymenectomy
Imperforate hymen is managed surgically with hymenectomy. Repair is generally reserved for the newborn period or, ideally, in adolescence, as at puberty the presence of estrogen aids in surgical repair and healing.5 Simple aspiration of hematocolpos/ mucocolpos can lead to ascending infection, and pyocolpos and should be avoided.6
The goal of hymenectomy is to:
-
open the hymeneal membrane to allow egress of fluid and menstrual flow
-
allow for tampon use
The procedure is relatively straightforward and usually is performed under general anesthesia, although regional anesthesia also is an option.
Steps to the varying hymenectomy incisions
Cruciate incision
1. Incise the hymen at the 2-, 4-, 8-, and 10-o’clock positions into four quadrants.
2. Excise the quadrants along the lateral wall of the vagina.
Elliptical incision
1. Make a circumferential incision with the Bovie electrocautery, incising the hymenal membrane close to the hymenal ring.
U-incision
1. Similar to the elliptical incision, use the Bovie electrocautery to incise the tissue close to the hymenal ring posterior and laterally in a “u” shape.
2. Make a horizontal incision superiorly to remove the extra tissue.
Vertical incision
This incision has been described in cases where there is an attempt to spare the hymen for religious or cultural preference.
1. Make a midline vertical hymenotomy less than 1 cm. Drain the borders of the hymen.
2. Apply suture obliquely to form a circular opening.
References
1. Dominguez C, Rock J, Horowitz I. Surgical conditions of the vagina and urethra. In: TeLinde’s Operative Gynecology. 10th ed. Philadelphia, PA: Lippincott Williams and Wilkins; 1997.
2. Basaran M, Usal D, Aydemir C. Hymen sparing surgery for imperforate hymen: case reports and review of literature. J Pediatr Adolesc Gynecol. 2009;22(4):e61–e64.
Tips to a successful procedure
Ensure adequate suctioning. Before starting the procedure, insert a Foley catheter to completely drain the bladder and delineate the urethra. Making an initial incision into the hymen usually results in the expulsion of the old blood and mucus, which can be very thick and viscous; therefore, it is important to have adequate suction tubing.
Prevent scarring. After evacuating the old blood and mucus, excise the hymeneal membrane with a cruciate incision as is traditionally described. Alternatively, some experts use an elliptical incision or u-incision. (See “Steps to the varying hymenectomy incisions”.) Prevent excision of the hymenal tissue too close to the vaginal mucosa, as this can lead to scarring and stenosis and dyspareunia.3
Suturing the mucosal margins is likely unnecessary in adolescent patients. After excision of the hymenal tissue, one option is to suture the mucosal margins of the hymenal ring in an interrupted fashion with a fine, delayed-absorbable suture. Alternatively, at our institution, where we employ the u-incision (FIGURE 5), we assure hemostasis of the mucosal margins and do not suture the margins. Suturing the margins is believed to prevent adherence of the edges; however, in the pubertal girl, adherence is unlikely secondary to estrogen exposure.
Figure 5. Surgical correction with u-incision
Avoid infection; do not irrigate. We do not recommend that you irrigate the vagina and perform unnecessary uterine manipulation, as this may introduce bacteria into the dilated cervix and uterus.3,8
Septate/microperforate/cribiform hymen
These other hymeneal anomalies also may require surgical correction if they become clinically significant. Patients may present with difficulty inserting or removing a tampon, insertional dyspareunia, or incomplete drainage of menstrual blood.6
Imaging is usually not indicated to diagnose these hymenal anomalies, as physical examination will reveal a patent vaginal tract. A moistened Q-tip can be placed into the orifice or behind the septate hymen for confirmation (FIGURE 6).
Surgical correction of a microperforate or cribiform hymen is performed using the same principles as imperforate hymen.
Surgical correction of a septate hymen involves tying and suturing or clamping with a hemostat the upper and lower edges, with the excess hymenal tissue between the sutures then excised.8
Figure 6. Septate hymen
Postop care and follow up
Postoperative analgesia with lidocaine jelly or ice packs is usually sufficient for pain management. Reinforce proper hygienic care measures. At 2- to 3-week follow up, assess the patient for healing and evaluate the size of the hymenal orifice.
Key takeaways
-Differentiating imperforate hymen from low transverse vaginal septum or distal vaginal agenesis prior to surgery is of utmost importance because management is very different, and performing the wrong procedure can result in serious morbidity.
-With imperforate hymen, examination of the external genitalia reveals a perineal bulge secondary to hematocolpos.
-Pelvic MRI is essential to delineate the anatomy with both vaginal septum and agenesis, for preoperative evaluation of location and thickness of septum as well as measurement of total length of agenesis.
-Hymenectomy is relatively straightforward and may be performed using a cruciate, elliptical, or u-incision.
-Care should be taken to prevent excision of hymeneal tissue too close to the vaginal mucosa, as this can lead to scarring and stenosis, and later lead to dyspareunia.
Many gynecologists encounter imperforate hymen, a congenital vaginal anomaly, in general practice. As such, it is important to have a basic understanding of the condition and to be aware of appropriate screening, evaluation, and management. This knowledge will allow you to differentiate imperforate hymen from more complex anomalies—preventing significant morbidity that could result from performing the wrong surgical procedure on this condition—and to provide optimal surgical management.
How often and why does it occur?
Imperforate hymen occurs in approximately 1/1000 newborn girls. It is the most common obstructive anomaly of the female reproductive tract.1,2
The hymen consists of fibrous connective tissue attached to the vaginal wall. In the perinatal period, the hymen serves to separate the vaginal lumen from the urogenital sinus (UGS); this is usually perforated during embryonic life by canalization of the most caudal portion of the vaginal plate at the UGS. This establishes a connection between the lumen of the vaginal canal and the vaginal vestibule.3 Failure of the hymen to perforate completely in the perinatal period can result in varying anomalies, including imperforate (FIGURE 1), microperforate, cribiform, or septated hymen.
Figure 1. Imperforate hymen
How does it present?
Its presentation is variable and frequently asymptomatic in infants and children.4 As a result, the diagnosis is often delayed until puberty.3
In infancy. Newborns typically will present with a hymenal bulge from hydrocolpos or mucocolpos, which result from maternal estrogen secretion on the newborn’s vaginal epithelium.5 This is usually asymptomatic and self limited.
Rarely, large hydrocolpos/mucocolpos may become symptomatic and can lead to urinary obstruction, or they can present as an abdominal mass or intestinal obstruction.4
In adolescence. The majority of adolescents will present with cyclic or persistent pelvic pain and primary amenorrhea. If significant hematometra is present, an abdominal mass also may be palpated. In extreme cases, the patient may present with mass effect symptoms, including back pain, pain with defecation, constipation, nausea and vomiting, urinary retention, or hydronephrosis.6 Retrograde passage of blood into the fallopian tubes can cause hematosalpinx, which can lead to endometriosis and adhesion formation. Blood also may pass freely into the peritoneal cavity, forming hemoperitoneum.3
Related article: Your age-based guide to comprehensive well-woman care
Robert L. Barbieri, MD (October 2012)
Imperforate hymen, vaginal septum, or distal vaginal atresia?
When in doubt, refer. Imperforate hymen can be confused with distal vaginal atresia or low transverse vaginal septum. Often, the patient may present with similar signs and symptoms in all 3 cases. Accurately differentiating imperforate hymen from the former two more complex congenital anomalies prior to surgery is of utmost importance because management is very different, and performing the wrong procedure can result in serious morbidity. As such, it is important to appropriately define the anatomy and refer the complex cases to a specialist comfortable and skilled in managing congenital anomalies, usually a pediatric and adolescent gynecologist or reproductive endocrinologist.
Imperforate hymen
Examination of the external genitalia reveals a perineal bulge secondary to hematocolpos.7 This finding, coupled with a rectal examination and pelvic ultrasonography is usually sufficient to make the diagnosis.6,8 However, magnetic resonance imaging (MRI) of the pelvis should be obtained in cases where the diagnosis is uncertain or the physical exam is more consistent with vaginal septum or agenesis.
Transverse vaginal septum
A reverse septum results from failure of the müllerian duct derivatives and UGS to fuse or canalize. This can occur in the lower, middle, or upper portion of the vagina, and septa may be thick or thin.6 Low transverse septa are more easily confused with imperforate hymen. Examination usually reveals a normal hymen with a short vagina posteriorly. In cases of extreme hematocolpos, vaginal septa also may present with a perineal bulge but, again, this will be posterior to a normal hymen.
Distal vaginal atresia
This condition occurs during embryonic development when the UGS fails to contribute to the lower portion of the vagina (FIGURE 2).5 In cases of distal vaginal atresia there is a lack of vaginal orifice, or only a vaginal dimple may be present.5,6 Rectovaginal examination will reveal a palpable mass if the upper vagina is distended with blood.6
Figure 2. Lower vaginal atresia
MRI is vital to firm diagnosis
In addition to pelvic ultrasonography, pelvic MRI is necessary to delineate the anatomy with both vaginal septum (FIGURE 3) and lower vaginal atresia (FIGURE 4), as preoperative evaluation of location and thickness of a vaginal septum as well as measurement of the total length of agenesis is imperative.6-8 Misdiagnosis of the vaginal septa or atresia as an imperforate hymen can lead to significant scarring and stenosis and can make corrective surgical procedures difficult or suboptimal.
| Figure 3. MRI of transverse vaginal septum |
Figure 4. MRI of lower vaginal atresia
Surgical management: hymenectomy
Imperforate hymen is managed surgically with hymenectomy. Repair is generally reserved for the newborn period or, ideally, in adolescence, as at puberty the presence of estrogen aids in surgical repair and healing.5 Simple aspiration of hematocolpos/ mucocolpos can lead to ascending infection, and pyocolpos and should be avoided.6
The goal of hymenectomy is to:
-
open the hymeneal membrane to allow egress of fluid and menstrual flow
-
allow for tampon use
The procedure is relatively straightforward and usually is performed under general anesthesia, although regional anesthesia also is an option.
Steps to the varying hymenectomy incisions
Cruciate incision
1. Incise the hymen at the 2-, 4-, 8-, and 10-o’clock positions into four quadrants.
2. Excise the quadrants along the lateral wall of the vagina.
Elliptical incision
1. Make a circumferential incision with the Bovie electrocautery, incising the hymenal membrane close to the hymenal ring.
U-incision
1. Similar to the elliptical incision, use the Bovie electrocautery to incise the tissue close to the hymenal ring posterior and laterally in a “u” shape.
2. Make a horizontal incision superiorly to remove the extra tissue.
Vertical incision
This incision has been described in cases where there is an attempt to spare the hymen for religious or cultural preference.
1. Make a midline vertical hymenotomy less than 1 cm. Drain the borders of the hymen.
2. Apply suture obliquely to form a circular opening.
References
1. Dominguez C, Rock J, Horowitz I. Surgical conditions of the vagina and urethra. In: TeLinde’s Operative Gynecology. 10th ed. Philadelphia, PA: Lippincott Williams and Wilkins; 1997.
2. Basaran M, Usal D, Aydemir C. Hymen sparing surgery for imperforate hymen: case reports and review of literature. J Pediatr Adolesc Gynecol. 2009;22(4):e61–e64.
Tips to a successful procedure
Ensure adequate suctioning. Before starting the procedure, insert a Foley catheter to completely drain the bladder and delineate the urethra. Making an initial incision into the hymen usually results in the expulsion of the old blood and mucus, which can be very thick and viscous; therefore, it is important to have adequate suction tubing.
Prevent scarring. After evacuating the old blood and mucus, excise the hymeneal membrane with a cruciate incision as is traditionally described. Alternatively, some experts use an elliptical incision or u-incision. (See “Steps to the varying hymenectomy incisions”.) Prevent excision of the hymenal tissue too close to the vaginal mucosa, as this can lead to scarring and stenosis and dyspareunia.3
Suturing the mucosal margins is likely unnecessary in adolescent patients. After excision of the hymenal tissue, one option is to suture the mucosal margins of the hymenal ring in an interrupted fashion with a fine, delayed-absorbable suture. Alternatively, at our institution, where we employ the u-incision (FIGURE 5), we assure hemostasis of the mucosal margins and do not suture the margins. Suturing the margins is believed to prevent adherence of the edges; however, in the pubertal girl, adherence is unlikely secondary to estrogen exposure.
Figure 5. Surgical correction with u-incision
Avoid infection; do not irrigate. We do not recommend that you irrigate the vagina and perform unnecessary uterine manipulation, as this may introduce bacteria into the dilated cervix and uterus.3,8
Septate/microperforate/cribiform hymen
These other hymeneal anomalies also may require surgical correction if they become clinically significant. Patients may present with difficulty inserting or removing a tampon, insertional dyspareunia, or incomplete drainage of menstrual blood.6
Imaging is usually not indicated to diagnose these hymenal anomalies, as physical examination will reveal a patent vaginal tract. A moistened Q-tip can be placed into the orifice or behind the septate hymen for confirmation (FIGURE 6).
Surgical correction of a microperforate or cribiform hymen is performed using the same principles as imperforate hymen.
Surgical correction of a septate hymen involves tying and suturing or clamping with a hemostat the upper and lower edges, with the excess hymenal tissue between the sutures then excised.8
Figure 6. Septate hymen
Postop care and follow up
Postoperative analgesia with lidocaine jelly or ice packs is usually sufficient for pain management. Reinforce proper hygienic care measures. At 2- to 3-week follow up, assess the patient for healing and evaluate the size of the hymenal orifice.
Key takeaways
-Differentiating imperforate hymen from low transverse vaginal septum or distal vaginal agenesis prior to surgery is of utmost importance because management is very different, and performing the wrong procedure can result in serious morbidity.
-With imperforate hymen, examination of the external genitalia reveals a perineal bulge secondary to hematocolpos.
-Pelvic MRI is essential to delineate the anatomy with both vaginal septum and agenesis, for preoperative evaluation of location and thickness of septum as well as measurement of total length of agenesis.
-Hymenectomy is relatively straightforward and may be performed using a cruciate, elliptical, or u-incision.
-Care should be taken to prevent excision of hymeneal tissue too close to the vaginal mucosa, as this can lead to scarring and stenosis, and later lead to dyspareunia.
Transtibial Anterior Cruciate Ligament Reconstruction
Surgeons perform an estimated 150,000 anterior cruciate ligament reconstructions (ACLRs) each year in the United States.1 Most surgeons who perform ACLRs do so infrequently; American Board of Orthopaedic Surgery data suggest that about 90% of ACL surgical procedures are performed by surgeons who do fewer than 10 ACLRs annually.2 Multiple studies have listed technical aspects as the most common reason for ACLR failure.3-6 For this reason, it is important that surgeons focus on the technical aspects of the procedure to improve outcomes.
[Introductory paragraph provided in lieu of abstract.]
Surgeons perform an estimated 150,000 anterior cruciate ligament reconstructions (ACLRs) each year in the United States.1 Most surgeons who perform ACLRs do so infrequently; American Board of Orthopaedic Surgery data suggest that about 90% of ACL surgical procedures are performed by surgeons who do fewer than 10 ACLRs annually.2 Multiple studies have listed technical aspects as the most common reason for ACLR failure.3-6 For this reason, it is important that surgeons focus on the technical aspects of the procedure to improve outcomes.
[Introductory paragraph provided in lieu of abstract.]
Surgeons perform an estimated 150,000 anterior cruciate ligament reconstructions (ACLRs) each year in the United States.1 Most surgeons who perform ACLRs do so infrequently; American Board of Orthopaedic Surgery data suggest that about 90% of ACL surgical procedures are performed by surgeons who do fewer than 10 ACLRs annually.2 Multiple studies have listed technical aspects as the most common reason for ACLR failure.3-6 For this reason, it is important that surgeons focus on the technical aspects of the procedure to improve outcomes.
[Introductory paragraph provided in lieu of abstract.]
Excessive visceral fat linked to increased risk of CVD, cancer
Earn 0.25 hours AMA PRA Category 1 credit: Read this article, and click the link at the end to take the post-test.
Excessive visceral fat was associated with incident cardiovascular disease and cancer after adjustment for clinical risk factors and general adiposity, results from a study of Framingham Heart Study participants showed.
The findings "support the growing appreciation of a pathogenic role of ectopic fat," researchers led by Dr. Kathryn A. Britton of the division of cardiovascular medicine at Brigham and Women’s Hospital, Boston, reported. The study was published online July 10 in the Journal of the American College of Cardiology. "Given the worldwide obesity epidemic, identification of high-risk individuals is important as it allows targeting of preventive and therapeutic measures. Furthermore, markers of risk may provide insight into the biology linking body fat distribution and outcomes."
Since few studies have examined prospective outcomes in people with ectopic fat, the researchers set out to examine the association of directly-imaged fat measurements with incident CVD, cancer, and all-cause mortality in 3,086 participants from the Framingham Heart Study. All of the patients underwent multidetector computerized tomography with an 8-slice scanner in an effort to identify and measure areas of visceral adipose tissue, pericardial adipose tissue, and periaortic adipose tissue. During a median follow-up of 5 years, the study participants were assessed for heart disease, cancer, and death risk after adjustment for standard risk factors.
The mean age of the 3,086 patients was 50 years; 51% were men. At the end of the follow-up period, there were 90 cardiovascular events, 141 cancer cases, and 71 deaths. After multivariable adjustment using Cox proportional hazards regression models, the researchers found that each standard deviation increase in visceral adipose tissue was associated with cardiovascular disease (HR 1.44; P =.01) and cancer (HR 1.43; P = .005). None of the fat depots were associated with all-cause mortality.
"Numerous experimental studies support a potential link between visceral adipose tissue and biological pathways important in the pathogenesis of multiple disease outcomes," Dr. Britton and her colleagues wrote. "Adipokines, biologically active molecules secreted from adipose tissue, are key components of these pathways and include inflammatory cytokines, angiogenic factors, lipid metabolites, and extracellular matrix components. Adipokine secretion appears to differ between specific fat depots with visceral adipose tissue demonstrating greater expression of proinflammatory and proangiogenic genes, compared with subcutaneous adipose tissue."
The researchers acknowledged certain limitations of the study including the fact that the study sample was predominately white and that weight change data were not available on the participants during the follow-up period.
The study was supported by the National Heart, Lung and Blood Institute’s Framingham Heart Study. Dr. Britton was supported by a Research Career Development Award from the NHLBI.
To earn 0.25 hours AMA PRA Category 1 credit after reading this article, take the post-test here.
Earn 0.25 hours AMA PRA Category 1 credit: Read this article, and click the link at the end to take the post-test.
Excessive visceral fat was associated with incident cardiovascular disease and cancer after adjustment for clinical risk factors and general adiposity, results from a study of Framingham Heart Study participants showed.
The findings "support the growing appreciation of a pathogenic role of ectopic fat," researchers led by Dr. Kathryn A. Britton of the division of cardiovascular medicine at Brigham and Women’s Hospital, Boston, reported. The study was published online July 10 in the Journal of the American College of Cardiology. "Given the worldwide obesity epidemic, identification of high-risk individuals is important as it allows targeting of preventive and therapeutic measures. Furthermore, markers of risk may provide insight into the biology linking body fat distribution and outcomes."
Since few studies have examined prospective outcomes in people with ectopic fat, the researchers set out to examine the association of directly-imaged fat measurements with incident CVD, cancer, and all-cause mortality in 3,086 participants from the Framingham Heart Study. All of the patients underwent multidetector computerized tomography with an 8-slice scanner in an effort to identify and measure areas of visceral adipose tissue, pericardial adipose tissue, and periaortic adipose tissue. During a median follow-up of 5 years, the study participants were assessed for heart disease, cancer, and death risk after adjustment for standard risk factors.
The mean age of the 3,086 patients was 50 years; 51% were men. At the end of the follow-up period, there were 90 cardiovascular events, 141 cancer cases, and 71 deaths. After multivariable adjustment using Cox proportional hazards regression models, the researchers found that each standard deviation increase in visceral adipose tissue was associated with cardiovascular disease (HR 1.44; P =.01) and cancer (HR 1.43; P = .005). None of the fat depots were associated with all-cause mortality.
"Numerous experimental studies support a potential link between visceral adipose tissue and biological pathways important in the pathogenesis of multiple disease outcomes," Dr. Britton and her colleagues wrote. "Adipokines, biologically active molecules secreted from adipose tissue, are key components of these pathways and include inflammatory cytokines, angiogenic factors, lipid metabolites, and extracellular matrix components. Adipokine secretion appears to differ between specific fat depots with visceral adipose tissue demonstrating greater expression of proinflammatory and proangiogenic genes, compared with subcutaneous adipose tissue."
The researchers acknowledged certain limitations of the study including the fact that the study sample was predominately white and that weight change data were not available on the participants during the follow-up period.
The study was supported by the National Heart, Lung and Blood Institute’s Framingham Heart Study. Dr. Britton was supported by a Research Career Development Award from the NHLBI.
To earn 0.25 hours AMA PRA Category 1 credit after reading this article, take the post-test here.
Earn 0.25 hours AMA PRA Category 1 credit: Read this article, and click the link at the end to take the post-test.
Excessive visceral fat was associated with incident cardiovascular disease and cancer after adjustment for clinical risk factors and general adiposity, results from a study of Framingham Heart Study participants showed.
The findings "support the growing appreciation of a pathogenic role of ectopic fat," researchers led by Dr. Kathryn A. Britton of the division of cardiovascular medicine at Brigham and Women’s Hospital, Boston, reported. The study was published online July 10 in the Journal of the American College of Cardiology. "Given the worldwide obesity epidemic, identification of high-risk individuals is important as it allows targeting of preventive and therapeutic measures. Furthermore, markers of risk may provide insight into the biology linking body fat distribution and outcomes."
Since few studies have examined prospective outcomes in people with ectopic fat, the researchers set out to examine the association of directly-imaged fat measurements with incident CVD, cancer, and all-cause mortality in 3,086 participants from the Framingham Heart Study. All of the patients underwent multidetector computerized tomography with an 8-slice scanner in an effort to identify and measure areas of visceral adipose tissue, pericardial adipose tissue, and periaortic adipose tissue. During a median follow-up of 5 years, the study participants were assessed for heart disease, cancer, and death risk after adjustment for standard risk factors.
The mean age of the 3,086 patients was 50 years; 51% were men. At the end of the follow-up period, there were 90 cardiovascular events, 141 cancer cases, and 71 deaths. After multivariable adjustment using Cox proportional hazards regression models, the researchers found that each standard deviation increase in visceral adipose tissue was associated with cardiovascular disease (HR 1.44; P =.01) and cancer (HR 1.43; P = .005). None of the fat depots were associated with all-cause mortality.
"Numerous experimental studies support a potential link between visceral adipose tissue and biological pathways important in the pathogenesis of multiple disease outcomes," Dr. Britton and her colleagues wrote. "Adipokines, biologically active molecules secreted from adipose tissue, are key components of these pathways and include inflammatory cytokines, angiogenic factors, lipid metabolites, and extracellular matrix components. Adipokine secretion appears to differ between specific fat depots with visceral adipose tissue demonstrating greater expression of proinflammatory and proangiogenic genes, compared with subcutaneous adipose tissue."
The researchers acknowledged certain limitations of the study including the fact that the study sample was predominately white and that weight change data were not available on the participants during the follow-up period.
The study was supported by the National Heart, Lung and Blood Institute’s Framingham Heart Study. Dr. Britton was supported by a Research Career Development Award from the NHLBI.
To earn 0.25 hours AMA PRA Category 1 credit after reading this article, take the post-test here.
FROM THE JOURNAL OF THE AMERICAN COLLEGE OF CARDIOLOGY
UPDATE ON INFECTIOUS DISEASE
The past year has seen the publication of four studies with immediate relevance for clinicians:
- a retrospective, population-based cohort study that explores whether women who have chorioamnionitis in one pregnancy are at risk for the same type of infection in a subsequent pregnancy
- another retrospective cohort study that assesses the clinical utility of testing for gonorrhea and chlamydia before inserting an intrauterine device (IUD)
- an elegant primate experiment that highlights the value of azithromycin in subjects with chorioamnionitis
- a multicenter, randomized, nonblinded trial in seriously ill patients to determine whether daily bathing with chlorhexidine-impregnated washcloths can reduce the acquisition of multidrug-resistant organisms and the incidence of hospital-acquired bloodstream infection.
CHORIOAMNIONITIS IN ONE PREGNANCY IS LIKELY TO RECUR IN THE NEXT GENERATION
Cohen-Cline HN, Kahn TR, Hutter CM. A population-based study of the risk of repeat clinical chorioamnionitis in Washington State, 1989–2008. Am J Obstet Gynecol. 2012;207(6):473.e1–e7.
This retrospective, population-based cohort study (Level II evidence) is one of the few to examine the risk of recurrence for chorioamnionitis, and the findings are intriguing. Women who were infected during their first delivery were 3.43 times more likely to become infected in their second delivery than women who did not have chorioamnionitis in their first pregnancy (95% confidence interval [CI], 2.67–4.42; P <.001). This association persisted even after adjustment for potential confounders, such as age, ethnicity, presence of premature rupture of membranes (PROM), and internal fetal monitoring.
Chorioamnionitis is a common affliction
This infection complicates approximately 5% of term deliveries and a significantly higher percentage of preterm deliveries. The principal causative organisms are group B streptococci (GBS), Escherichia coli and other aerobic Gram-negative bacilli, both Gram-positive and Gram-negative anaerobes, and genital mycoplasmas.
The main risk factors for chorioamnionitis are:
- prematurity
- prolonged labor
- prolonged rupture of membranes
- multiple internal examinations
- internal fetal monitoring
- low socioeconomic status
- preexisting genital tract infection (eg, bacterial vaginosis, GBS colonization).
Infants delivered to infected mothers are at increased risk for sepsis, pneumonia, and meningitis. Severely infected infants, particularly those who are premature, are also at increased risk for cerebral palsy.
Details of the study
This investigation focused on women in Washington State who had a first pregnancy from 1989 through 2008 and then had at least one additional birth during the study period.
Participants included 6,219 women who had chorioamnionitis in their first pregnancy and 25,294 women who did not. Using logistic regression, Cohen-Cline and colleagues estimated the odds ratio for chorioamnionitis in the second delivery, taking into account the following potential confounders:
- maternal age
- ethnicity
- presence of PROM
- use of internal monitoring
- smoking.
As I stated above, women who had chorioamnionitis in their first pregnancy were 3.43 times as likely to have it again in their second pregnancy.
What this EVIDENCE means for practice
When a patient has a history of chorioamnionitis, we should do everything possible to reduce her risk for recurrent infection. For example, we should screen her for lower genital tract infections that predispose to chorioamnionitis:
-gonorrhea
-chlamydia
-bacterial vaginosis
-GBS.
If the patient has any of the first three infections, treat her immediately with the appropriate antibiotics. If she is colonized with GBS, administer one of the intrapartum antibiotic regimens recommended by the Centers for Disease Control and Prevention (CDC).
If the patient has a history of preterm PROM or spontaneous preterm delivery, initiate prophylaxis with progesterone and assess her cervical length periodically to determine whether cerclage is indicated.
During labor, make every effort to minimize the duration of ruptured membranes, the length of invasive monitoring, and the number of internal vaginal examinations.
At the earliest sign of intra-amniotic infection, treat the patient with broad-spectrum antibiotics, usually ampicillin plus gentamicin.
In low-risk populations, universal screening for sexually transmitted infections is probably unnecessary before IUD insertion
Sufrin CB, Postlethwaite D, Armstrong MA, et al. Neisseria gonorrhea and Chlamydia trachomatis screening at intrauterine device insertion and pelvic inflammatory disease. Obstet Gynecol. 2012;120(6):1314–1321.
This retrospective cohort study (Level II evidence) focused on women who had an IUD inserted in a managed-care practice at Kaiser Permanente of Northern California during a 5-year period. Sufrin and colleagues compared the incidence of pelvic inflammatory disease (PID) within 90 days after insertion among women who were, and were not, screened for gonorrhea and chlamydia.
Among 57,728 IUD insertions, 47% involved women who were unscreened within 1 year of the procedure. Among women who were screened, 19% were tested on the day of IUD insertion.
The overall risk of PID in the study cohort was very low—0.54% (95% CI, 0.48–0.60). Investigators were unable to identify any significant difference in the risk of PID between women who had no screening versus those who were screened. Among women who were screened, same-day screening was equivalent to prescreening.
Investigators concluded that the most reasonable protocol is to screen on the basis of risk factors on the same day as IUD insertion. If the patient has obvious evidence of endocervicitis (ie, mucopurulent discharge), IUD insertion should be delayed. Otherwise, if the patient has risk factors for infection, screening should be followed by IUD insertion.
If the screen is positive, the patient should be treated in accordance with the latest CDC recommendations, and the IUD can be left in place.
Sufrin and colleagues concluded that adherence to this protocol would be associated with a very low, and clinically acceptable, risk of PID.
STI screening need not be an obstacle to IUD use
The IUD is an excellent method of contraception, and it is suitable for most patients. It is particularly useful for women who have difficulty remembering to take a pill each day or to use a barrier method of contraception at each episode of coitus.
Obstacles to more widespread use of the IUD include:
- high initial cost
- misconceptions on the part of the patient about the mechanism of action and adverse effects of the device
- cumbersome protocols that require multiple physician visits for counseling and sexually transmitted infection (STI) testing before the device is inserted.
What this EVIDENCE means for practice
This study provides reassurance that, at least in a relatively affluent managed-care population, universal testing for STIs is probably not necessary. When testing is indicated, it can be performed on the same day that the IUD is inserted, minimizing the number of office visits.
What is less clear is whether the same protocol can be applied to a population with a significantly higher prevalence of STIs. In such a population, universal screening for gonorrhea and chlamydia may be more prudent. However, screening still can be performed on the same day as IUD insertion.
In a primate model of intra-amniotic infection with Ureaplasma, maternal azithromycin prolonged gestation
Grigsby PL, Novy MJ, Sadowsky DW, et al. Maternal azithromycin therapy for Ureaplasma intraamniotic infection delays preterm delivery and reduces fetal lung injury in a primate model. Am J Obstet Gynecol. 2012;207(6):475.e1–e14.
Grigsby and colleagues assessed the efficacy of azithromycin—with and without anti-inflammatory agents—in delaying preterm birth and minimizing fetal lung injury in a primate model. They found that azithromycin significantly prolonged gestation.
Details of the study
The study involved 16 chronically instrumented rhesus monkeys who received intra-amniotic inoculation with Ureaplasma parvum (107 colony-forming units/mL) and were then observed. When contractions began, as they invariably did, six monkeys received no treatment, five received intravenous (IV) azithromycin (12.5 mg/kg every 12 hours) for 10 days, and five received azithromycin plus dexamethasone and indomethacin.
Key outcome measures were the intra-amniotic concentration of proinflammatory mediators, the frequency of positive amniotic fluid cultures for U parvum, and the extent of histologic fetal lung injury.
In treated animals, the mean (SD) inoculation-to-delivery interval was 20.9 (1.4) days, compared with 13.7 (2.5) days in untreated monkeys (P <.05).
In addition, there was a twofold to threefold increase in the percentage of undelivered animals at 18 to 20 days after inoculation in the treatment group, compared with the no-treatment group. Treatment also significantly decreased the Ureaplasma colony count in the amniotic fluid, effectively eliminating the organism within 4 days.
In both treatment groups, the amniotic fluid concentration of proinflammatory mediators decreased significantly, compared with the untreated group. Treatment also significantly reduced the magnitude of deleterious histologic changes in the fetal lungs.
Somewhat surprisingly, dexamethasone and indomethacin did not enhance the treatment effect of azithromycin. Moreover, despite prolongation of pregnancy, all animals in the treatment group still delivered prematurely.
Why treatment should target genital mycoplasmas
Chorioamnionitis is an importance cause of preterm labor and preterm delivery. The principal pathogens are part of the normal vaginal flora: aerobic Gram-negative bacilli, aerobic Gram-positive cocci, anaerobes, and genital mycoplasmas.
Most treatment regimens for chorioamnionitis (eg, ampicillin plus gentamicin) do not specifically target the genital mycoplasmas. However, the most commonly recommended prophylactic antibiotic regimens for patients with preterm PROM include agents with specific action against mycoplasmas, namely erythromycin and azithromycin.
In this clinical setting, antibiotic prophylaxis prolongs the latency period and decreases the frequency of both maternal and fetal/neonatal infection.
This elegant basic science investigation sheds new light on the importance of the genital mycoplasmas in the pathogenesis of preterm labor and helps to explain why drugs like erythromycin and azithromycin may be so valuable in prolonging the latent period and reducing the frequency of infection and injury in the baby.
What this EVIDENCE means for practice
Because IV azithromycin rapidly achieved inhibitory concentrations in amniotic fluid and maintained these concentrations over 10 days of treatment, it significantly reduced the concentration of Ureaplasma in the amniotic fluid as well as the risk of histologic injury to the fetal lung.
Accordingly, I recommend that azithromycin remain a key component of the prophylactic regimen for patients with preterm PROM. It also may be advisable to add azithromycin to the usual combination of ampicillin plus gentamicin for empiric treatment of chorioamnionitis.
Daily bathing with chlorhexidine cloths can protect hospitalized patients from serious infection
Climo MW, Yokoe DS, Warren DK, et al. Effect of daily chlorhexidine bathing on hospital-acquired infection. N Engl J Med. 2013;368(6):533–542.
This multicenter, randomized, nonblinded trial of 7,727 seriously ill patients sought to determine whether daily bathing with chlorhexidine-impregnated washcloths can decrease the acquisition of multidrug-resistant organisms and the incidence of hospital-acquired bloodstream infection.
Each day, patients in eight ICUs and one bone-marrow transplant unit bathed themselves, or were bathed by nursing staff, with 2% chlorhexidine-impregnated cloths or non–antimicrobial washcloths. All body surfaces except the face were cleansed. After 6 months, each unit changed to the other method of bathing.
Investigators focused on two outcomes:
- the prevalence of colonization of the nares with methicillin-resistant Staphylococcus aureus (MRSA) or colonization of the perirectal area with vancomycin-resistant enterococci (VRE)
- the frequency of hospital-acquired bloodstream infection (bacterial or fungal) detected more than 48 hours after admission to the unit.
The overall rate of MRSA or VRE acquisition was reduced by 23% when patients were bathed with chlorhexidine (5.10 versus 6.60 cases per 100 patient-days; P = .03). The overall rate of hospital-acquired bloodstream infection was reduced by 28% during the intervention period (4.78 vs 6.60 cases per 1,000 patient-days; P = .006).
In particular, the rate of central-catheter–associated bloodstream infection was 53% lower during the intervention (1.55 vs 3.30 cases per 1,000 catheter-days; P = .004).
The intervention had the greatest impact on infections caused by Gram-positive and fungal organisms.
The protective effect of chlorhexidine bathing was greatest among patients who had the longest length of stay in the unit.
Chlorhexidine did not cause an increased frequency of skin reactions. Moreover, use of the antiseptic washes did not cause the emergence of MRSA or VRA isolates with high-level resistance.
This study is of great interest in light of a recent report that demonstrated that preoperative preparation of the skin with chlorhexidine was more effective than preparation with povidone-iodine in reducing the risk of surgical-site infections after major operative procedures.1 Not only is chlorhexidine highly active against the usual bacteria that colonize the skin of hospitalized patients, it also has residual antibacterial activity that further decreases the colonization of the patient’s skin by microbes.
What this EVIDENCE means for practice
This study has two clear implications for ObGyns. First, chlorhexidine washes should be used by all patients who are scheduled for surgery, particularly those undergoing procedures that carry a relatively high risk of postoperative wound infection, such as total abdominal hysterectomy, radical hysterectomy, and cesarean delivery. In morbidly obese patients, particular attention should be directed to the skin beneath the abdominal panniculus.
Second, when we have seriously ill obstetric or gynecologic patients, especially those with long-term indwelling catheters who require prolonged hospitalization, we should order daily bathing (excluding the face) with chlorhexidine.
Reference
1. Darouiche RO, Wall MJ Jr, Itani KM, et al. Chlorhexidine-alcohol versus povidone-iodine for surgical-site antisepsis. N Engl J Med. 2010;362(1):18–26.
The past year has seen the publication of four studies with immediate relevance for clinicians:
- a retrospective, population-based cohort study that explores whether women who have chorioamnionitis in one pregnancy are at risk for the same type of infection in a subsequent pregnancy
- another retrospective cohort study that assesses the clinical utility of testing for gonorrhea and chlamydia before inserting an intrauterine device (IUD)
- an elegant primate experiment that highlights the value of azithromycin in subjects with chorioamnionitis
- a multicenter, randomized, nonblinded trial in seriously ill patients to determine whether daily bathing with chlorhexidine-impregnated washcloths can reduce the acquisition of multidrug-resistant organisms and the incidence of hospital-acquired bloodstream infection.
CHORIOAMNIONITIS IN ONE PREGNANCY IS LIKELY TO RECUR IN THE NEXT GENERATION
Cohen-Cline HN, Kahn TR, Hutter CM. A population-based study of the risk of repeat clinical chorioamnionitis in Washington State, 1989–2008. Am J Obstet Gynecol. 2012;207(6):473.e1–e7.
This retrospective, population-based cohort study (Level II evidence) is one of the few to examine the risk of recurrence for chorioamnionitis, and the findings are intriguing. Women who were infected during their first delivery were 3.43 times more likely to become infected in their second delivery than women who did not have chorioamnionitis in their first pregnancy (95% confidence interval [CI], 2.67–4.42; P <.001). This association persisted even after adjustment for potential confounders, such as age, ethnicity, presence of premature rupture of membranes (PROM), and internal fetal monitoring.
Chorioamnionitis is a common affliction
This infection complicates approximately 5% of term deliveries and a significantly higher percentage of preterm deliveries. The principal causative organisms are group B streptococci (GBS), Escherichia coli and other aerobic Gram-negative bacilli, both Gram-positive and Gram-negative anaerobes, and genital mycoplasmas.
The main risk factors for chorioamnionitis are:
- prematurity
- prolonged labor
- prolonged rupture of membranes
- multiple internal examinations
- internal fetal monitoring
- low socioeconomic status
- preexisting genital tract infection (eg, bacterial vaginosis, GBS colonization).
Infants delivered to infected mothers are at increased risk for sepsis, pneumonia, and meningitis. Severely infected infants, particularly those who are premature, are also at increased risk for cerebral palsy.
Details of the study
This investigation focused on women in Washington State who had a first pregnancy from 1989 through 2008 and then had at least one additional birth during the study period.
Participants included 6,219 women who had chorioamnionitis in their first pregnancy and 25,294 women who did not. Using logistic regression, Cohen-Cline and colleagues estimated the odds ratio for chorioamnionitis in the second delivery, taking into account the following potential confounders:
- maternal age
- ethnicity
- presence of PROM
- use of internal monitoring
- smoking.
As I stated above, women who had chorioamnionitis in their first pregnancy were 3.43 times as likely to have it again in their second pregnancy.
What this EVIDENCE means for practice
When a patient has a history of chorioamnionitis, we should do everything possible to reduce her risk for recurrent infection. For example, we should screen her for lower genital tract infections that predispose to chorioamnionitis:
-gonorrhea
-chlamydia
-bacterial vaginosis
-GBS.
If the patient has any of the first three infections, treat her immediately with the appropriate antibiotics. If she is colonized with GBS, administer one of the intrapartum antibiotic regimens recommended by the Centers for Disease Control and Prevention (CDC).
If the patient has a history of preterm PROM or spontaneous preterm delivery, initiate prophylaxis with progesterone and assess her cervical length periodically to determine whether cerclage is indicated.
During labor, make every effort to minimize the duration of ruptured membranes, the length of invasive monitoring, and the number of internal vaginal examinations.
At the earliest sign of intra-amniotic infection, treat the patient with broad-spectrum antibiotics, usually ampicillin plus gentamicin.
In low-risk populations, universal screening for sexually transmitted infections is probably unnecessary before IUD insertion
Sufrin CB, Postlethwaite D, Armstrong MA, et al. Neisseria gonorrhea and Chlamydia trachomatis screening at intrauterine device insertion and pelvic inflammatory disease. Obstet Gynecol. 2012;120(6):1314–1321.
This retrospective cohort study (Level II evidence) focused on women who had an IUD inserted in a managed-care practice at Kaiser Permanente of Northern California during a 5-year period. Sufrin and colleagues compared the incidence of pelvic inflammatory disease (PID) within 90 days after insertion among women who were, and were not, screened for gonorrhea and chlamydia.
Among 57,728 IUD insertions, 47% involved women who were unscreened within 1 year of the procedure. Among women who were screened, 19% were tested on the day of IUD insertion.
The overall risk of PID in the study cohort was very low—0.54% (95% CI, 0.48–0.60). Investigators were unable to identify any significant difference in the risk of PID between women who had no screening versus those who were screened. Among women who were screened, same-day screening was equivalent to prescreening.
Investigators concluded that the most reasonable protocol is to screen on the basis of risk factors on the same day as IUD insertion. If the patient has obvious evidence of endocervicitis (ie, mucopurulent discharge), IUD insertion should be delayed. Otherwise, if the patient has risk factors for infection, screening should be followed by IUD insertion.
If the screen is positive, the patient should be treated in accordance with the latest CDC recommendations, and the IUD can be left in place.
Sufrin and colleagues concluded that adherence to this protocol would be associated with a very low, and clinically acceptable, risk of PID.
STI screening need not be an obstacle to IUD use
The IUD is an excellent method of contraception, and it is suitable for most patients. It is particularly useful for women who have difficulty remembering to take a pill each day or to use a barrier method of contraception at each episode of coitus.
Obstacles to more widespread use of the IUD include:
- high initial cost
- misconceptions on the part of the patient about the mechanism of action and adverse effects of the device
- cumbersome protocols that require multiple physician visits for counseling and sexually transmitted infection (STI) testing before the device is inserted.
What this EVIDENCE means for practice
This study provides reassurance that, at least in a relatively affluent managed-care population, universal testing for STIs is probably not necessary. When testing is indicated, it can be performed on the same day that the IUD is inserted, minimizing the number of office visits.
What is less clear is whether the same protocol can be applied to a population with a significantly higher prevalence of STIs. In such a population, universal screening for gonorrhea and chlamydia may be more prudent. However, screening still can be performed on the same day as IUD insertion.
In a primate model of intra-amniotic infection with Ureaplasma, maternal azithromycin prolonged gestation
Grigsby PL, Novy MJ, Sadowsky DW, et al. Maternal azithromycin therapy for Ureaplasma intraamniotic infection delays preterm delivery and reduces fetal lung injury in a primate model. Am J Obstet Gynecol. 2012;207(6):475.e1–e14.
Grigsby and colleagues assessed the efficacy of azithromycin—with and without anti-inflammatory agents—in delaying preterm birth and minimizing fetal lung injury in a primate model. They found that azithromycin significantly prolonged gestation.
Details of the study
The study involved 16 chronically instrumented rhesus monkeys who received intra-amniotic inoculation with Ureaplasma parvum (107 colony-forming units/mL) and were then observed. When contractions began, as they invariably did, six monkeys received no treatment, five received intravenous (IV) azithromycin (12.5 mg/kg every 12 hours) for 10 days, and five received azithromycin plus dexamethasone and indomethacin.
Key outcome measures were the intra-amniotic concentration of proinflammatory mediators, the frequency of positive amniotic fluid cultures for U parvum, and the extent of histologic fetal lung injury.
In treated animals, the mean (SD) inoculation-to-delivery interval was 20.9 (1.4) days, compared with 13.7 (2.5) days in untreated monkeys (P <.05).
In addition, there was a twofold to threefold increase in the percentage of undelivered animals at 18 to 20 days after inoculation in the treatment group, compared with the no-treatment group. Treatment also significantly decreased the Ureaplasma colony count in the amniotic fluid, effectively eliminating the organism within 4 days.
In both treatment groups, the amniotic fluid concentration of proinflammatory mediators decreased significantly, compared with the untreated group. Treatment also significantly reduced the magnitude of deleterious histologic changes in the fetal lungs.
Somewhat surprisingly, dexamethasone and indomethacin did not enhance the treatment effect of azithromycin. Moreover, despite prolongation of pregnancy, all animals in the treatment group still delivered prematurely.
Why treatment should target genital mycoplasmas
Chorioamnionitis is an importance cause of preterm labor and preterm delivery. The principal pathogens are part of the normal vaginal flora: aerobic Gram-negative bacilli, aerobic Gram-positive cocci, anaerobes, and genital mycoplasmas.
Most treatment regimens for chorioamnionitis (eg, ampicillin plus gentamicin) do not specifically target the genital mycoplasmas. However, the most commonly recommended prophylactic antibiotic regimens for patients with preterm PROM include agents with specific action against mycoplasmas, namely erythromycin and azithromycin.
In this clinical setting, antibiotic prophylaxis prolongs the latency period and decreases the frequency of both maternal and fetal/neonatal infection.
This elegant basic science investigation sheds new light on the importance of the genital mycoplasmas in the pathogenesis of preterm labor and helps to explain why drugs like erythromycin and azithromycin may be so valuable in prolonging the latent period and reducing the frequency of infection and injury in the baby.
What this EVIDENCE means for practice
Because IV azithromycin rapidly achieved inhibitory concentrations in amniotic fluid and maintained these concentrations over 10 days of treatment, it significantly reduced the concentration of Ureaplasma in the amniotic fluid as well as the risk of histologic injury to the fetal lung.
Accordingly, I recommend that azithromycin remain a key component of the prophylactic regimen for patients with preterm PROM. It also may be advisable to add azithromycin to the usual combination of ampicillin plus gentamicin for empiric treatment of chorioamnionitis.
Daily bathing with chlorhexidine cloths can protect hospitalized patients from serious infection
Climo MW, Yokoe DS, Warren DK, et al. Effect of daily chlorhexidine bathing on hospital-acquired infection. N Engl J Med. 2013;368(6):533–542.
This multicenter, randomized, nonblinded trial of 7,727 seriously ill patients sought to determine whether daily bathing with chlorhexidine-impregnated washcloths can decrease the acquisition of multidrug-resistant organisms and the incidence of hospital-acquired bloodstream infection.
Each day, patients in eight ICUs and one bone-marrow transplant unit bathed themselves, or were bathed by nursing staff, with 2% chlorhexidine-impregnated cloths or non–antimicrobial washcloths. All body surfaces except the face were cleansed. After 6 months, each unit changed to the other method of bathing.
Investigators focused on two outcomes:
- the prevalence of colonization of the nares with methicillin-resistant Staphylococcus aureus (MRSA) or colonization of the perirectal area with vancomycin-resistant enterococci (VRE)
- the frequency of hospital-acquired bloodstream infection (bacterial or fungal) detected more than 48 hours after admission to the unit.
The overall rate of MRSA or VRE acquisition was reduced by 23% when patients were bathed with chlorhexidine (5.10 versus 6.60 cases per 100 patient-days; P = .03). The overall rate of hospital-acquired bloodstream infection was reduced by 28% during the intervention period (4.78 vs 6.60 cases per 1,000 patient-days; P = .006).
In particular, the rate of central-catheter–associated bloodstream infection was 53% lower during the intervention (1.55 vs 3.30 cases per 1,000 catheter-days; P = .004).
The intervention had the greatest impact on infections caused by Gram-positive and fungal organisms.
The protective effect of chlorhexidine bathing was greatest among patients who had the longest length of stay in the unit.
Chlorhexidine did not cause an increased frequency of skin reactions. Moreover, use of the antiseptic washes did not cause the emergence of MRSA or VRA isolates with high-level resistance.
This study is of great interest in light of a recent report that demonstrated that preoperative preparation of the skin with chlorhexidine was more effective than preparation with povidone-iodine in reducing the risk of surgical-site infections after major operative procedures.1 Not only is chlorhexidine highly active against the usual bacteria that colonize the skin of hospitalized patients, it also has residual antibacterial activity that further decreases the colonization of the patient’s skin by microbes.
What this EVIDENCE means for practice
This study has two clear implications for ObGyns. First, chlorhexidine washes should be used by all patients who are scheduled for surgery, particularly those undergoing procedures that carry a relatively high risk of postoperative wound infection, such as total abdominal hysterectomy, radical hysterectomy, and cesarean delivery. In morbidly obese patients, particular attention should be directed to the skin beneath the abdominal panniculus.
Second, when we have seriously ill obstetric or gynecologic patients, especially those with long-term indwelling catheters who require prolonged hospitalization, we should order daily bathing (excluding the face) with chlorhexidine.
The past year has seen the publication of four studies with immediate relevance for clinicians:
- a retrospective, population-based cohort study that explores whether women who have chorioamnionitis in one pregnancy are at risk for the same type of infection in a subsequent pregnancy
- another retrospective cohort study that assesses the clinical utility of testing for gonorrhea and chlamydia before inserting an intrauterine device (IUD)
- an elegant primate experiment that highlights the value of azithromycin in subjects with chorioamnionitis
- a multicenter, randomized, nonblinded trial in seriously ill patients to determine whether daily bathing with chlorhexidine-impregnated washcloths can reduce the acquisition of multidrug-resistant organisms and the incidence of hospital-acquired bloodstream infection.
CHORIOAMNIONITIS IN ONE PREGNANCY IS LIKELY TO RECUR IN THE NEXT GENERATION
Cohen-Cline HN, Kahn TR, Hutter CM. A population-based study of the risk of repeat clinical chorioamnionitis in Washington State, 1989–2008. Am J Obstet Gynecol. 2012;207(6):473.e1–e7.
This retrospective, population-based cohort study (Level II evidence) is one of the few to examine the risk of recurrence for chorioamnionitis, and the findings are intriguing. Women who were infected during their first delivery were 3.43 times more likely to become infected in their second delivery than women who did not have chorioamnionitis in their first pregnancy (95% confidence interval [CI], 2.67–4.42; P <.001). This association persisted even after adjustment for potential confounders, such as age, ethnicity, presence of premature rupture of membranes (PROM), and internal fetal monitoring.
Chorioamnionitis is a common affliction
This infection complicates approximately 5% of term deliveries and a significantly higher percentage of preterm deliveries. The principal causative organisms are group B streptococci (GBS), Escherichia coli and other aerobic Gram-negative bacilli, both Gram-positive and Gram-negative anaerobes, and genital mycoplasmas.
The main risk factors for chorioamnionitis are:
- prematurity
- prolonged labor
- prolonged rupture of membranes
- multiple internal examinations
- internal fetal monitoring
- low socioeconomic status
- preexisting genital tract infection (eg, bacterial vaginosis, GBS colonization).
Infants delivered to infected mothers are at increased risk for sepsis, pneumonia, and meningitis. Severely infected infants, particularly those who are premature, are also at increased risk for cerebral palsy.
Details of the study
This investigation focused on women in Washington State who had a first pregnancy from 1989 through 2008 and then had at least one additional birth during the study period.
Participants included 6,219 women who had chorioamnionitis in their first pregnancy and 25,294 women who did not. Using logistic regression, Cohen-Cline and colleagues estimated the odds ratio for chorioamnionitis in the second delivery, taking into account the following potential confounders:
- maternal age
- ethnicity
- presence of PROM
- use of internal monitoring
- smoking.
As I stated above, women who had chorioamnionitis in their first pregnancy were 3.43 times as likely to have it again in their second pregnancy.
What this EVIDENCE means for practice
When a patient has a history of chorioamnionitis, we should do everything possible to reduce her risk for recurrent infection. For example, we should screen her for lower genital tract infections that predispose to chorioamnionitis:
-gonorrhea
-chlamydia
-bacterial vaginosis
-GBS.
If the patient has any of the first three infections, treat her immediately with the appropriate antibiotics. If she is colonized with GBS, administer one of the intrapartum antibiotic regimens recommended by the Centers for Disease Control and Prevention (CDC).
If the patient has a history of preterm PROM or spontaneous preterm delivery, initiate prophylaxis with progesterone and assess her cervical length periodically to determine whether cerclage is indicated.
During labor, make every effort to minimize the duration of ruptured membranes, the length of invasive monitoring, and the number of internal vaginal examinations.
At the earliest sign of intra-amniotic infection, treat the patient with broad-spectrum antibiotics, usually ampicillin plus gentamicin.
In low-risk populations, universal screening for sexually transmitted infections is probably unnecessary before IUD insertion
Sufrin CB, Postlethwaite D, Armstrong MA, et al. Neisseria gonorrhea and Chlamydia trachomatis screening at intrauterine device insertion and pelvic inflammatory disease. Obstet Gynecol. 2012;120(6):1314–1321.
This retrospective cohort study (Level II evidence) focused on women who had an IUD inserted in a managed-care practice at Kaiser Permanente of Northern California during a 5-year period. Sufrin and colleagues compared the incidence of pelvic inflammatory disease (PID) within 90 days after insertion among women who were, and were not, screened for gonorrhea and chlamydia.
Among 57,728 IUD insertions, 47% involved women who were unscreened within 1 year of the procedure. Among women who were screened, 19% were tested on the day of IUD insertion.
The overall risk of PID in the study cohort was very low—0.54% (95% CI, 0.48–0.60). Investigators were unable to identify any significant difference in the risk of PID between women who had no screening versus those who were screened. Among women who were screened, same-day screening was equivalent to prescreening.
Investigators concluded that the most reasonable protocol is to screen on the basis of risk factors on the same day as IUD insertion. If the patient has obvious evidence of endocervicitis (ie, mucopurulent discharge), IUD insertion should be delayed. Otherwise, if the patient has risk factors for infection, screening should be followed by IUD insertion.
If the screen is positive, the patient should be treated in accordance with the latest CDC recommendations, and the IUD can be left in place.
Sufrin and colleagues concluded that adherence to this protocol would be associated with a very low, and clinically acceptable, risk of PID.
STI screening need not be an obstacle to IUD use
The IUD is an excellent method of contraception, and it is suitable for most patients. It is particularly useful for women who have difficulty remembering to take a pill each day or to use a barrier method of contraception at each episode of coitus.
Obstacles to more widespread use of the IUD include:
- high initial cost
- misconceptions on the part of the patient about the mechanism of action and adverse effects of the device
- cumbersome protocols that require multiple physician visits for counseling and sexually transmitted infection (STI) testing before the device is inserted.
What this EVIDENCE means for practice
This study provides reassurance that, at least in a relatively affluent managed-care population, universal testing for STIs is probably not necessary. When testing is indicated, it can be performed on the same day that the IUD is inserted, minimizing the number of office visits.
What is less clear is whether the same protocol can be applied to a population with a significantly higher prevalence of STIs. In such a population, universal screening for gonorrhea and chlamydia may be more prudent. However, screening still can be performed on the same day as IUD insertion.
In a primate model of intra-amniotic infection with Ureaplasma, maternal azithromycin prolonged gestation
Grigsby PL, Novy MJ, Sadowsky DW, et al. Maternal azithromycin therapy for Ureaplasma intraamniotic infection delays preterm delivery and reduces fetal lung injury in a primate model. Am J Obstet Gynecol. 2012;207(6):475.e1–e14.
Grigsby and colleagues assessed the efficacy of azithromycin—with and without anti-inflammatory agents—in delaying preterm birth and minimizing fetal lung injury in a primate model. They found that azithromycin significantly prolonged gestation.
Details of the study
The study involved 16 chronically instrumented rhesus monkeys who received intra-amniotic inoculation with Ureaplasma parvum (107 colony-forming units/mL) and were then observed. When contractions began, as they invariably did, six monkeys received no treatment, five received intravenous (IV) azithromycin (12.5 mg/kg every 12 hours) for 10 days, and five received azithromycin plus dexamethasone and indomethacin.
Key outcome measures were the intra-amniotic concentration of proinflammatory mediators, the frequency of positive amniotic fluid cultures for U parvum, and the extent of histologic fetal lung injury.
In treated animals, the mean (SD) inoculation-to-delivery interval was 20.9 (1.4) days, compared with 13.7 (2.5) days in untreated monkeys (P <.05).
In addition, there was a twofold to threefold increase in the percentage of undelivered animals at 18 to 20 days after inoculation in the treatment group, compared with the no-treatment group. Treatment also significantly decreased the Ureaplasma colony count in the amniotic fluid, effectively eliminating the organism within 4 days.
In both treatment groups, the amniotic fluid concentration of proinflammatory mediators decreased significantly, compared with the untreated group. Treatment also significantly reduced the magnitude of deleterious histologic changes in the fetal lungs.
Somewhat surprisingly, dexamethasone and indomethacin did not enhance the treatment effect of azithromycin. Moreover, despite prolongation of pregnancy, all animals in the treatment group still delivered prematurely.
Why treatment should target genital mycoplasmas
Chorioamnionitis is an importance cause of preterm labor and preterm delivery. The principal pathogens are part of the normal vaginal flora: aerobic Gram-negative bacilli, aerobic Gram-positive cocci, anaerobes, and genital mycoplasmas.
Most treatment regimens for chorioamnionitis (eg, ampicillin plus gentamicin) do not specifically target the genital mycoplasmas. However, the most commonly recommended prophylactic antibiotic regimens for patients with preterm PROM include agents with specific action against mycoplasmas, namely erythromycin and azithromycin.
In this clinical setting, antibiotic prophylaxis prolongs the latency period and decreases the frequency of both maternal and fetal/neonatal infection.
This elegant basic science investigation sheds new light on the importance of the genital mycoplasmas in the pathogenesis of preterm labor and helps to explain why drugs like erythromycin and azithromycin may be so valuable in prolonging the latent period and reducing the frequency of infection and injury in the baby.
What this EVIDENCE means for practice
Because IV azithromycin rapidly achieved inhibitory concentrations in amniotic fluid and maintained these concentrations over 10 days of treatment, it significantly reduced the concentration of Ureaplasma in the amniotic fluid as well as the risk of histologic injury to the fetal lung.
Accordingly, I recommend that azithromycin remain a key component of the prophylactic regimen for patients with preterm PROM. It also may be advisable to add azithromycin to the usual combination of ampicillin plus gentamicin for empiric treatment of chorioamnionitis.
Daily bathing with chlorhexidine cloths can protect hospitalized patients from serious infection
Climo MW, Yokoe DS, Warren DK, et al. Effect of daily chlorhexidine bathing on hospital-acquired infection. N Engl J Med. 2013;368(6):533–542.
This multicenter, randomized, nonblinded trial of 7,727 seriously ill patients sought to determine whether daily bathing with chlorhexidine-impregnated washcloths can decrease the acquisition of multidrug-resistant organisms and the incidence of hospital-acquired bloodstream infection.
Each day, patients in eight ICUs and one bone-marrow transplant unit bathed themselves, or were bathed by nursing staff, with 2% chlorhexidine-impregnated cloths or non–antimicrobial washcloths. All body surfaces except the face were cleansed. After 6 months, each unit changed to the other method of bathing.
Investigators focused on two outcomes:
- the prevalence of colonization of the nares with methicillin-resistant Staphylococcus aureus (MRSA) or colonization of the perirectal area with vancomycin-resistant enterococci (VRE)
- the frequency of hospital-acquired bloodstream infection (bacterial or fungal) detected more than 48 hours after admission to the unit.
The overall rate of MRSA or VRE acquisition was reduced by 23% when patients were bathed with chlorhexidine (5.10 versus 6.60 cases per 100 patient-days; P = .03). The overall rate of hospital-acquired bloodstream infection was reduced by 28% during the intervention period (4.78 vs 6.60 cases per 1,000 patient-days; P = .006).
In particular, the rate of central-catheter–associated bloodstream infection was 53% lower during the intervention (1.55 vs 3.30 cases per 1,000 catheter-days; P = .004).
The intervention had the greatest impact on infections caused by Gram-positive and fungal organisms.
The protective effect of chlorhexidine bathing was greatest among patients who had the longest length of stay in the unit.
Chlorhexidine did not cause an increased frequency of skin reactions. Moreover, use of the antiseptic washes did not cause the emergence of MRSA or VRA isolates with high-level resistance.
This study is of great interest in light of a recent report that demonstrated that preoperative preparation of the skin with chlorhexidine was more effective than preparation with povidone-iodine in reducing the risk of surgical-site infections after major operative procedures.1 Not only is chlorhexidine highly active against the usual bacteria that colonize the skin of hospitalized patients, it also has residual antibacterial activity that further decreases the colonization of the patient’s skin by microbes.
What this EVIDENCE means for practice
This study has two clear implications for ObGyns. First, chlorhexidine washes should be used by all patients who are scheduled for surgery, particularly those undergoing procedures that carry a relatively high risk of postoperative wound infection, such as total abdominal hysterectomy, radical hysterectomy, and cesarean delivery. In morbidly obese patients, particular attention should be directed to the skin beneath the abdominal panniculus.
Second, when we have seriously ill obstetric or gynecologic patients, especially those with long-term indwelling catheters who require prolonged hospitalization, we should order daily bathing (excluding the face) with chlorhexidine.
Reference
1. Darouiche RO, Wall MJ Jr, Itani KM, et al. Chlorhexidine-alcohol versus povidone-iodine for surgical-site antisepsis. N Engl J Med. 2010;362(1):18–26.
Reference
1. Darouiche RO, Wall MJ Jr, Itani KM, et al. Chlorhexidine-alcohol versus povidone-iodine for surgical-site antisepsis. N Engl J Med. 2010;362(1):18–26.
How to manage emergencies associated with tocolysis for preterm labor
CASE 1: Preterm labor with cervical changes
Ms. M, a 42-year-old woman pregnant with her second child, begins having contractions at 30 weeks’ gestation. Examination reveals that her cervix is dilated 2 cm and effaced 50%. She is given subcutaneous terbutaline to suppress her contractions. Thirty minutes later, she complains of shortness of breath and chest pain. An electrocardiogram reveals depression of the ST segment, and a chest radiograph shows mild pulmonary edema.
How should her symptoms be managed?
Preterm labor precedes delivery in about 50% of preterm births. Approximately 33% of women who have preterm labor will experience spontaneous resolution, and more than 50% of women who have preterm labor will deliver at term. Although the use of tocolytic therapy has proved to be effective at temporarily suppressing uterine activity, it has not been shown to delay delivery for more than a few hours or days.1
The American College of Obstetricians and Gynecologists (ACOG) recommends the use of tocolytics only when a delay in labor for approximately 48 hours would improve outcome. Therefore, tocolytic therapy should be reserved for the following circumstances:
- to stop the progress of labor long enough to administer antenatal corticosteroid therapy
- to prolong pregnancy when there is an underlying self-limiting condition that can cause labor, such as pyelonephritis
- to provide time for safe transport to a facility with a higher level of neonatal care.2
Tocolytics are generally not indicated before the fetus is viable, although we lack data from randomized, controlled trials to support a specific recommendation. The approach is clearer when the fetus is near the upper limits of viability. Most studies suggest that 34 weeks’ gestation is the threshold at which the perinatal morbidity and mortality associated with delivery are too low to justify the cost and potential complications of tocolysis.3
Women who experience preterm labor without cervical changes generally should not be treated with tocolytics.2 Contraindications to tocolytic therapy include:
- lethal fetal anomaly
- nonreassuring fetal status
- maternal disease
- maternal hemorrhage with hemodynamic instability.
Beta-adrenergic agonists carry many risks
These agents have been studied in several randomized, controlled trials. Although ritodrine was approved as tocolytic therapy by the US Food and Drug Administration (FDA), it has since been removed from the US market. Terbutaline is still available but lacks FDA approval as a tocolytic.
Maternal side effects associated with beta-adrenergic agonists are thought to arise from stimulation of the beta-1 and beta-2 adrenergic receptors. Stimulation of the former increases maternal heart rate and stroke volume, whereas stimulation of the beta-2 adrenergic receptors causes the relaxation of smooth muscle, including the muscles of the myometrium, blood vessels, and bronchial tree. The resulting symptoms may include maternal tachycardia, cardiac arrhythmias, palpitations, and metabolic aberrations (including hyperglycemia, hypokalemia, and hypotension). Common symptoms associated with the administration of a beta-adrenergic agonist include tremor, shortness of breath, and chest discomfort.4 Although pulmonary edema and myocardial ischemia are uncommon, they can occur even when there is no history of underlying maternal disease.
Terbutaline has been linked to maternal deaths
Sixteen maternal deaths were reported following initial marketing of terbutaline in 1976 until 2009. Three of the 16 cases involved outpatient use of terbutaline administered by a subcutaneous pump, and nine cases involved use of oral terbutaline alone or in addition to subcutaneous or IV terbutaline. In addition, 12 cases of serious maternal cardiovascular events were reported in association with terbutaline. These events included cardiac arrhythmias, myocardial infarction, pulmonary edema, hypertension, and tachycardia.
Because of these events, the FDA issued a black box warning for terbutaline that prohibits its use in the treatment of preterm labor for longer than 48 to 72 hours in the inpatient or outpatient setting because of the potential for serious maternal heart problems and death.5 Oral terbutaline should be avoided entirely in the prevention and treatment of preterm labor. However, the use of terbutaline for the management of acute tachysystole with an abnormal fetal heart-rate (FHR) pattern remains a reasonable course of treatment.6
Fetal tachycardia is the most common side effect of beta-adrenergic receptor agonists. For this reason, use of these drugs is not recommended when changes in FHR may be the first sign of fetal compromise, such as in patients with hemorrhage or infection. Neonatal hypoglycemia may also occur if maternal hyperglycemia is not controlled.7
Case 1 Resolved
Terbutaline is discontinued, and the patient’s pulmonary edema is treated with a single dose of furosemide. Electrolyte abnormalities resolve with discontinuation of medication. The patient stabilizes. Once her cardiorespiratory status improves, her contractions lessen and the cervix remains unchanged. She requires no further tocolysis and is discharged home. She presents again at 38 weeks in spontaneous labor.
CASE 2: Preterm labor treated with indomethacin
Ms. J, age 23, is 26 weeks’ pregnant with her first child. When she experienced preterm labor at 24.5 weeks’ gestation, she was given indomethacin. Now, ultrasonographic imaging reveals decreased amniotic fluid volume.
How should she be managed?
Indomethacin is a cyclooxygenase (COX) inhibitor. These drugs reduce prostaglandin production through the general inhibition of cyclooxygenase or by a specific receptor.8 Indomethacin is the most commonly used tocolytic in this class. It is a nonspecific COX inhibitor, as opposed to a COX-2 inhibitor. The latter has been associated with serious adverse outcomes in the nonobstetric population. COX-2 inhibitors now carry a black box warning or are no longer available.
Maternal contraindications for COX inhibitors include asthma, bleeding disorders, and significant renal dysfunction.
Although maternal side effects with COX inhibitors are usually mild, fetal side effects may be serious enough to cause perinatal morbidity or death.9
How indomethacin can lead to oligohydramnios
Maternal administration of indomethacin or ibuprofen can reduce fetal urine output and decrease the volume of amniotic fluid. In most cases, oligohydramnios occurs when indomethacin or ibuprofen has been given for more than 72 hours. For this reason, long-term use of a COX inhibitor should be accompanied by frequent monitoring of amniotic fluid volume by ultrasonography.
The most serious fetal complication associated with prolonged indomethacin administration (longer than 72 hours) is premature constriction of the ductus arteriosus. Ductal constriction appears to be contingent on gestational age. It has been described as early as 24 weeks’ gestation but is most common after 31 or 32 weeks. Therefore, indomethacin is not recommended for use after 32 weeks’ gestation.10
CASE 2 Resolved
The indomethacin is discontinued as soon as the decreased amniotic fluid is noted. The fluid volume returns to normal over the next 3 to 5 days. Because of the early gestational age, nifedipine is given to suppress contractions, and the patient has no further complications.
CASE 3: Preterm labor and magnesium intoxication
Ms. K experiences contractions and rapid cervical change at 32 weeks’ gestation. She is given magnesium for the preterm labor and fetal neuroprophylaxis, with nifedipine, a calcium-channel blocker, added as second-line tocolysis. Approximately 8 hours later, she reports difficulty breathing and moving.
How should her obstetrician proceed?
Calcium-channel blockers such as nifedipine are used for acute and maintenance tocolysis. This class of drugs is often selected for its relative ease of use and safety, as it has few maternal and fetal side effects. However, concomitant use of a calcium-channel blocker and magnesium sulfate can sometimes lead to neuromuscular blockade and significant respiratory depression, even necessitating mechanical ventilation.9 Treatment of these effects includes IV administration of 10% calcium gluconate (5–10 mEq), which usually reverses respiratory depression and heart block caused by magnesium intoxication. In extreme cases, peritoneal dialysis or hemodialysis may be required.
CASE 3 Resolved
The patient is given 10% calcium gluconate in the dosage described above, and she stabilizes. However, her contractions continue and she delivers at 32 weeks’ gestation. The infant does well in the NICU.
CASE 4: Preterm labor in a woman with kidney dysfunction
Ms. F, age 40, presents at 30 weeks’ gestation with regular contractions and cervical dilation of more than 3 cm. She also reports a history of kidney disease.
What steps are recommended prior to the initiation of magnesium therapy?
Magnesium sulfate has been used for more than 40 years to treat preterm labor and is still considered a first-line therapy in many centers. Although maternal side effects usually are mild, an adverse event may occur if the patient is not monitored carefully. An absence of deep-tendon reflexes should alert the clinician that magnesium levels need to be measured. Reflexes usually are lost at a serum level of 10 mEq/L or higher. When the magnesium level exceeds 13 mEq/L, cardiac arrest is a risk. IV calcium should be administered immediately in such patients.
Magnesium should be used with caution in patients with myocardial compromise. Because magnesium is eliminated by the kidneys, women with impaired renal function may experience magnesium toxicity at normal doses. If a patient has a creatinine level above 1 mg/dL, consider alternative treatment for her preterm labor. If magnesium is given, the normal loading dose (4–6 g) is appropriate, but the maintenance dose should be reduced.11
Fetal effects of magnesium sulfate
Recent studies indicate that predelivery magnesium may offer fetal neuroprotection. The minimum duration of administration for such neuroprotection is unknown but is less than 24 hours.8
Although magnesium can alter FHR patterns slightly, these changes are not clinically significant. Magnesium can also cause mild neonatal suppression at the time of delivery, but its effects quickly resolve with appropriate neonatal resuscitation. Long-term (>5 days) therapy is not recommended.
In May 2013, the FDA issued a warning about the risk of neonatal complications with long-term maternal magnesium administration. These complications include osteopenia, low calcium, and bone fracture. The pregnancy category for magnesium sulfate will be changed from “A” to “D” because of these teratogenic effects.12
CASE 4 Resolved
Because magnesium is mainly cleared by renal excretion, the clinician administers the medication with caution in this patient with reduced renal function. The clinician administers the same 4- to 6-g bolus that would be given a patient with normal kidney function, but the maintenance dose is reduced to 1 g. Magnesium levels are obtained every 12 hours or when clinically indicated.
Bottom line: Be ready to act
The short-term use of tocolytic therapy usually is not associated with maternal or fetal complications. After initial administration, maintenance tocolytic therapy probably does not prolong gestation.
Given the potential for harm without additional fetal benefit associated with extended therapy, I recommend that clinicians follow current clinical guidelines from ACOG for use of tocolytic agents. In the process, be vigilant for complications and be ready to act appropriately. Keep maternal and fetal conditions in mind when selecting a tocolytic agent.
9. US Food and Drug Administration. Terbutaline: Label Change—Warnings Against Use for Treatment of Preterm Labor. Published February 17, 2011. http://www.fda.gov/Safety/MedWatch/SafetyInformation/SafetyalertsforHumanMedicalProducts/ucm243843.htm. Accessed June 17, 2013.
12. US Food and Drug Administration. Magnesium Sulfate: Drug Safety Communication—Recommendation against Prolonged Use in Preterm Labor. Published May 30, 2013. http://www.fda.gov/Safety/MedWatch/SafetyInformation/SafetyAlertsforHumanMedicalProducts/ucm354603.htm. Accessed June 17, 2013.
CASE 1: Preterm labor with cervical changes
Ms. M, a 42-year-old woman pregnant with her second child, begins having contractions at 30 weeks’ gestation. Examination reveals that her cervix is dilated 2 cm and effaced 50%. She is given subcutaneous terbutaline to suppress her contractions. Thirty minutes later, she complains of shortness of breath and chest pain. An electrocardiogram reveals depression of the ST segment, and a chest radiograph shows mild pulmonary edema.
How should her symptoms be managed?
Preterm labor precedes delivery in about 50% of preterm births. Approximately 33% of women who have preterm labor will experience spontaneous resolution, and more than 50% of women who have preterm labor will deliver at term. Although the use of tocolytic therapy has proved to be effective at temporarily suppressing uterine activity, it has not been shown to delay delivery for more than a few hours or days.1
The American College of Obstetricians and Gynecologists (ACOG) recommends the use of tocolytics only when a delay in labor for approximately 48 hours would improve outcome. Therefore, tocolytic therapy should be reserved for the following circumstances:
- to stop the progress of labor long enough to administer antenatal corticosteroid therapy
- to prolong pregnancy when there is an underlying self-limiting condition that can cause labor, such as pyelonephritis
- to provide time for safe transport to a facility with a higher level of neonatal care.2
Tocolytics are generally not indicated before the fetus is viable, although we lack data from randomized, controlled trials to support a specific recommendation. The approach is clearer when the fetus is near the upper limits of viability. Most studies suggest that 34 weeks’ gestation is the threshold at which the perinatal morbidity and mortality associated with delivery are too low to justify the cost and potential complications of tocolysis.3
Women who experience preterm labor without cervical changes generally should not be treated with tocolytics.2 Contraindications to tocolytic therapy include:
- lethal fetal anomaly
- nonreassuring fetal status
- maternal disease
- maternal hemorrhage with hemodynamic instability.
Beta-adrenergic agonists carry many risks
These agents have been studied in several randomized, controlled trials. Although ritodrine was approved as tocolytic therapy by the US Food and Drug Administration (FDA), it has since been removed from the US market. Terbutaline is still available but lacks FDA approval as a tocolytic.
Maternal side effects associated with beta-adrenergic agonists are thought to arise from stimulation of the beta-1 and beta-2 adrenergic receptors. Stimulation of the former increases maternal heart rate and stroke volume, whereas stimulation of the beta-2 adrenergic receptors causes the relaxation of smooth muscle, including the muscles of the myometrium, blood vessels, and bronchial tree. The resulting symptoms may include maternal tachycardia, cardiac arrhythmias, palpitations, and metabolic aberrations (including hyperglycemia, hypokalemia, and hypotension). Common symptoms associated with the administration of a beta-adrenergic agonist include tremor, shortness of breath, and chest discomfort.4 Although pulmonary edema and myocardial ischemia are uncommon, they can occur even when there is no history of underlying maternal disease.
Terbutaline has been linked to maternal deaths
Sixteen maternal deaths were reported following initial marketing of terbutaline in 1976 until 2009. Three of the 16 cases involved outpatient use of terbutaline administered by a subcutaneous pump, and nine cases involved use of oral terbutaline alone or in addition to subcutaneous or IV terbutaline. In addition, 12 cases of serious maternal cardiovascular events were reported in association with terbutaline. These events included cardiac arrhythmias, myocardial infarction, pulmonary edema, hypertension, and tachycardia.
Because of these events, the FDA issued a black box warning for terbutaline that prohibits its use in the treatment of preterm labor for longer than 48 to 72 hours in the inpatient or outpatient setting because of the potential for serious maternal heart problems and death.5 Oral terbutaline should be avoided entirely in the prevention and treatment of preterm labor. However, the use of terbutaline for the management of acute tachysystole with an abnormal fetal heart-rate (FHR) pattern remains a reasonable course of treatment.6
Fetal tachycardia is the most common side effect of beta-adrenergic receptor agonists. For this reason, use of these drugs is not recommended when changes in FHR may be the first sign of fetal compromise, such as in patients with hemorrhage or infection. Neonatal hypoglycemia may also occur if maternal hyperglycemia is not controlled.7
Case 1 Resolved
Terbutaline is discontinued, and the patient’s pulmonary edema is treated with a single dose of furosemide. Electrolyte abnormalities resolve with discontinuation of medication. The patient stabilizes. Once her cardiorespiratory status improves, her contractions lessen and the cervix remains unchanged. She requires no further tocolysis and is discharged home. She presents again at 38 weeks in spontaneous labor.
CASE 2: Preterm labor treated with indomethacin
Ms. J, age 23, is 26 weeks’ pregnant with her first child. When she experienced preterm labor at 24.5 weeks’ gestation, she was given indomethacin. Now, ultrasonographic imaging reveals decreased amniotic fluid volume.
How should she be managed?
Indomethacin is a cyclooxygenase (COX) inhibitor. These drugs reduce prostaglandin production through the general inhibition of cyclooxygenase or by a specific receptor.8 Indomethacin is the most commonly used tocolytic in this class. It is a nonspecific COX inhibitor, as opposed to a COX-2 inhibitor. The latter has been associated with serious adverse outcomes in the nonobstetric population. COX-2 inhibitors now carry a black box warning or are no longer available.
Maternal contraindications for COX inhibitors include asthma, bleeding disorders, and significant renal dysfunction.
Although maternal side effects with COX inhibitors are usually mild, fetal side effects may be serious enough to cause perinatal morbidity or death.9
How indomethacin can lead to oligohydramnios
Maternal administration of indomethacin or ibuprofen can reduce fetal urine output and decrease the volume of amniotic fluid. In most cases, oligohydramnios occurs when indomethacin or ibuprofen has been given for more than 72 hours. For this reason, long-term use of a COX inhibitor should be accompanied by frequent monitoring of amniotic fluid volume by ultrasonography.
The most serious fetal complication associated with prolonged indomethacin administration (longer than 72 hours) is premature constriction of the ductus arteriosus. Ductal constriction appears to be contingent on gestational age. It has been described as early as 24 weeks’ gestation but is most common after 31 or 32 weeks. Therefore, indomethacin is not recommended for use after 32 weeks’ gestation.10
CASE 2 Resolved
The indomethacin is discontinued as soon as the decreased amniotic fluid is noted. The fluid volume returns to normal over the next 3 to 5 days. Because of the early gestational age, nifedipine is given to suppress contractions, and the patient has no further complications.
CASE 3: Preterm labor and magnesium intoxication
Ms. K experiences contractions and rapid cervical change at 32 weeks’ gestation. She is given magnesium for the preterm labor and fetal neuroprophylaxis, with nifedipine, a calcium-channel blocker, added as second-line tocolysis. Approximately 8 hours later, she reports difficulty breathing and moving.
How should her obstetrician proceed?
Calcium-channel blockers such as nifedipine are used for acute and maintenance tocolysis. This class of drugs is often selected for its relative ease of use and safety, as it has few maternal and fetal side effects. However, concomitant use of a calcium-channel blocker and magnesium sulfate can sometimes lead to neuromuscular blockade and significant respiratory depression, even necessitating mechanical ventilation.9 Treatment of these effects includes IV administration of 10% calcium gluconate (5–10 mEq), which usually reverses respiratory depression and heart block caused by magnesium intoxication. In extreme cases, peritoneal dialysis or hemodialysis may be required.
CASE 3 Resolved
The patient is given 10% calcium gluconate in the dosage described above, and she stabilizes. However, her contractions continue and she delivers at 32 weeks’ gestation. The infant does well in the NICU.
CASE 4: Preterm labor in a woman with kidney dysfunction
Ms. F, age 40, presents at 30 weeks’ gestation with regular contractions and cervical dilation of more than 3 cm. She also reports a history of kidney disease.
What steps are recommended prior to the initiation of magnesium therapy?
Magnesium sulfate has been used for more than 40 years to treat preterm labor and is still considered a first-line therapy in many centers. Although maternal side effects usually are mild, an adverse event may occur if the patient is not monitored carefully. An absence of deep-tendon reflexes should alert the clinician that magnesium levels need to be measured. Reflexes usually are lost at a serum level of 10 mEq/L or higher. When the magnesium level exceeds 13 mEq/L, cardiac arrest is a risk. IV calcium should be administered immediately in such patients.
Magnesium should be used with caution in patients with myocardial compromise. Because magnesium is eliminated by the kidneys, women with impaired renal function may experience magnesium toxicity at normal doses. If a patient has a creatinine level above 1 mg/dL, consider alternative treatment for her preterm labor. If magnesium is given, the normal loading dose (4–6 g) is appropriate, but the maintenance dose should be reduced.11
Fetal effects of magnesium sulfate
Recent studies indicate that predelivery magnesium may offer fetal neuroprotection. The minimum duration of administration for such neuroprotection is unknown but is less than 24 hours.8
Although magnesium can alter FHR patterns slightly, these changes are not clinically significant. Magnesium can also cause mild neonatal suppression at the time of delivery, but its effects quickly resolve with appropriate neonatal resuscitation. Long-term (>5 days) therapy is not recommended.
In May 2013, the FDA issued a warning about the risk of neonatal complications with long-term maternal magnesium administration. These complications include osteopenia, low calcium, and bone fracture. The pregnancy category for magnesium sulfate will be changed from “A” to “D” because of these teratogenic effects.12
CASE 4 Resolved
Because magnesium is mainly cleared by renal excretion, the clinician administers the medication with caution in this patient with reduced renal function. The clinician administers the same 4- to 6-g bolus that would be given a patient with normal kidney function, but the maintenance dose is reduced to 1 g. Magnesium levels are obtained every 12 hours or when clinically indicated.
Bottom line: Be ready to act
The short-term use of tocolytic therapy usually is not associated with maternal or fetal complications. After initial administration, maintenance tocolytic therapy probably does not prolong gestation.
Given the potential for harm without additional fetal benefit associated with extended therapy, I recommend that clinicians follow current clinical guidelines from ACOG for use of tocolytic agents. In the process, be vigilant for complications and be ready to act appropriately. Keep maternal and fetal conditions in mind when selecting a tocolytic agent.
CASE 1: Preterm labor with cervical changes
Ms. M, a 42-year-old woman pregnant with her second child, begins having contractions at 30 weeks’ gestation. Examination reveals that her cervix is dilated 2 cm and effaced 50%. She is given subcutaneous terbutaline to suppress her contractions. Thirty minutes later, she complains of shortness of breath and chest pain. An electrocardiogram reveals depression of the ST segment, and a chest radiograph shows mild pulmonary edema.
How should her symptoms be managed?
Preterm labor precedes delivery in about 50% of preterm births. Approximately 33% of women who have preterm labor will experience spontaneous resolution, and more than 50% of women who have preterm labor will deliver at term. Although the use of tocolytic therapy has proved to be effective at temporarily suppressing uterine activity, it has not been shown to delay delivery for more than a few hours or days.1
The American College of Obstetricians and Gynecologists (ACOG) recommends the use of tocolytics only when a delay in labor for approximately 48 hours would improve outcome. Therefore, tocolytic therapy should be reserved for the following circumstances:
- to stop the progress of labor long enough to administer antenatal corticosteroid therapy
- to prolong pregnancy when there is an underlying self-limiting condition that can cause labor, such as pyelonephritis
- to provide time for safe transport to a facility with a higher level of neonatal care.2
Tocolytics are generally not indicated before the fetus is viable, although we lack data from randomized, controlled trials to support a specific recommendation. The approach is clearer when the fetus is near the upper limits of viability. Most studies suggest that 34 weeks’ gestation is the threshold at which the perinatal morbidity and mortality associated with delivery are too low to justify the cost and potential complications of tocolysis.3
Women who experience preterm labor without cervical changes generally should not be treated with tocolytics.2 Contraindications to tocolytic therapy include:
- lethal fetal anomaly
- nonreassuring fetal status
- maternal disease
- maternal hemorrhage with hemodynamic instability.
Beta-adrenergic agonists carry many risks
These agents have been studied in several randomized, controlled trials. Although ritodrine was approved as tocolytic therapy by the US Food and Drug Administration (FDA), it has since been removed from the US market. Terbutaline is still available but lacks FDA approval as a tocolytic.
Maternal side effects associated with beta-adrenergic agonists are thought to arise from stimulation of the beta-1 and beta-2 adrenergic receptors. Stimulation of the former increases maternal heart rate and stroke volume, whereas stimulation of the beta-2 adrenergic receptors causes the relaxation of smooth muscle, including the muscles of the myometrium, blood vessels, and bronchial tree. The resulting symptoms may include maternal tachycardia, cardiac arrhythmias, palpitations, and metabolic aberrations (including hyperglycemia, hypokalemia, and hypotension). Common symptoms associated with the administration of a beta-adrenergic agonist include tremor, shortness of breath, and chest discomfort.4 Although pulmonary edema and myocardial ischemia are uncommon, they can occur even when there is no history of underlying maternal disease.
Terbutaline has been linked to maternal deaths
Sixteen maternal deaths were reported following initial marketing of terbutaline in 1976 until 2009. Three of the 16 cases involved outpatient use of terbutaline administered by a subcutaneous pump, and nine cases involved use of oral terbutaline alone or in addition to subcutaneous or IV terbutaline. In addition, 12 cases of serious maternal cardiovascular events were reported in association with terbutaline. These events included cardiac arrhythmias, myocardial infarction, pulmonary edema, hypertension, and tachycardia.
Because of these events, the FDA issued a black box warning for terbutaline that prohibits its use in the treatment of preterm labor for longer than 48 to 72 hours in the inpatient or outpatient setting because of the potential for serious maternal heart problems and death.5 Oral terbutaline should be avoided entirely in the prevention and treatment of preterm labor. However, the use of terbutaline for the management of acute tachysystole with an abnormal fetal heart-rate (FHR) pattern remains a reasonable course of treatment.6
Fetal tachycardia is the most common side effect of beta-adrenergic receptor agonists. For this reason, use of these drugs is not recommended when changes in FHR may be the first sign of fetal compromise, such as in patients with hemorrhage or infection. Neonatal hypoglycemia may also occur if maternal hyperglycemia is not controlled.7
Case 1 Resolved
Terbutaline is discontinued, and the patient’s pulmonary edema is treated with a single dose of furosemide. Electrolyte abnormalities resolve with discontinuation of medication. The patient stabilizes. Once her cardiorespiratory status improves, her contractions lessen and the cervix remains unchanged. She requires no further tocolysis and is discharged home. She presents again at 38 weeks in spontaneous labor.
CASE 2: Preterm labor treated with indomethacin
Ms. J, age 23, is 26 weeks’ pregnant with her first child. When she experienced preterm labor at 24.5 weeks’ gestation, she was given indomethacin. Now, ultrasonographic imaging reveals decreased amniotic fluid volume.
How should she be managed?
Indomethacin is a cyclooxygenase (COX) inhibitor. These drugs reduce prostaglandin production through the general inhibition of cyclooxygenase or by a specific receptor.8 Indomethacin is the most commonly used tocolytic in this class. It is a nonspecific COX inhibitor, as opposed to a COX-2 inhibitor. The latter has been associated with serious adverse outcomes in the nonobstetric population. COX-2 inhibitors now carry a black box warning or are no longer available.
Maternal contraindications for COX inhibitors include asthma, bleeding disorders, and significant renal dysfunction.
Although maternal side effects with COX inhibitors are usually mild, fetal side effects may be serious enough to cause perinatal morbidity or death.9
How indomethacin can lead to oligohydramnios
Maternal administration of indomethacin or ibuprofen can reduce fetal urine output and decrease the volume of amniotic fluid. In most cases, oligohydramnios occurs when indomethacin or ibuprofen has been given for more than 72 hours. For this reason, long-term use of a COX inhibitor should be accompanied by frequent monitoring of amniotic fluid volume by ultrasonography.
The most serious fetal complication associated with prolonged indomethacin administration (longer than 72 hours) is premature constriction of the ductus arteriosus. Ductal constriction appears to be contingent on gestational age. It has been described as early as 24 weeks’ gestation but is most common after 31 or 32 weeks. Therefore, indomethacin is not recommended for use after 32 weeks’ gestation.10
CASE 2 Resolved
The indomethacin is discontinued as soon as the decreased amniotic fluid is noted. The fluid volume returns to normal over the next 3 to 5 days. Because of the early gestational age, nifedipine is given to suppress contractions, and the patient has no further complications.
CASE 3: Preterm labor and magnesium intoxication
Ms. K experiences contractions and rapid cervical change at 32 weeks’ gestation. She is given magnesium for the preterm labor and fetal neuroprophylaxis, with nifedipine, a calcium-channel blocker, added as second-line tocolysis. Approximately 8 hours later, she reports difficulty breathing and moving.
How should her obstetrician proceed?
Calcium-channel blockers such as nifedipine are used for acute and maintenance tocolysis. This class of drugs is often selected for its relative ease of use and safety, as it has few maternal and fetal side effects. However, concomitant use of a calcium-channel blocker and magnesium sulfate can sometimes lead to neuromuscular blockade and significant respiratory depression, even necessitating mechanical ventilation.9 Treatment of these effects includes IV administration of 10% calcium gluconate (5–10 mEq), which usually reverses respiratory depression and heart block caused by magnesium intoxication. In extreme cases, peritoneal dialysis or hemodialysis may be required.
CASE 3 Resolved
The patient is given 10% calcium gluconate in the dosage described above, and she stabilizes. However, her contractions continue and she delivers at 32 weeks’ gestation. The infant does well in the NICU.
CASE 4: Preterm labor in a woman with kidney dysfunction
Ms. F, age 40, presents at 30 weeks’ gestation with regular contractions and cervical dilation of more than 3 cm. She also reports a history of kidney disease.
What steps are recommended prior to the initiation of magnesium therapy?
Magnesium sulfate has been used for more than 40 years to treat preterm labor and is still considered a first-line therapy in many centers. Although maternal side effects usually are mild, an adverse event may occur if the patient is not monitored carefully. An absence of deep-tendon reflexes should alert the clinician that magnesium levels need to be measured. Reflexes usually are lost at a serum level of 10 mEq/L or higher. When the magnesium level exceeds 13 mEq/L, cardiac arrest is a risk. IV calcium should be administered immediately in such patients.
Magnesium should be used with caution in patients with myocardial compromise. Because magnesium is eliminated by the kidneys, women with impaired renal function may experience magnesium toxicity at normal doses. If a patient has a creatinine level above 1 mg/dL, consider alternative treatment for her preterm labor. If magnesium is given, the normal loading dose (4–6 g) is appropriate, but the maintenance dose should be reduced.11
Fetal effects of magnesium sulfate
Recent studies indicate that predelivery magnesium may offer fetal neuroprotection. The minimum duration of administration for such neuroprotection is unknown but is less than 24 hours.8
Although magnesium can alter FHR patterns slightly, these changes are not clinically significant. Magnesium can also cause mild neonatal suppression at the time of delivery, but its effects quickly resolve with appropriate neonatal resuscitation. Long-term (>5 days) therapy is not recommended.
In May 2013, the FDA issued a warning about the risk of neonatal complications with long-term maternal magnesium administration. These complications include osteopenia, low calcium, and bone fracture. The pregnancy category for magnesium sulfate will be changed from “A” to “D” because of these teratogenic effects.12
CASE 4 Resolved
Because magnesium is mainly cleared by renal excretion, the clinician administers the medication with caution in this patient with reduced renal function. The clinician administers the same 4- to 6-g bolus that would be given a patient with normal kidney function, but the maintenance dose is reduced to 1 g. Magnesium levels are obtained every 12 hours or when clinically indicated.
Bottom line: Be ready to act
The short-term use of tocolytic therapy usually is not associated with maternal or fetal complications. After initial administration, maintenance tocolytic therapy probably does not prolong gestation.
Given the potential for harm without additional fetal benefit associated with extended therapy, I recommend that clinicians follow current clinical guidelines from ACOG for use of tocolytic agents. In the process, be vigilant for complications and be ready to act appropriately. Keep maternal and fetal conditions in mind when selecting a tocolytic agent.
9. US Food and Drug Administration. Terbutaline: Label Change—Warnings Against Use for Treatment of Preterm Labor. Published February 17, 2011. http://www.fda.gov/Safety/MedWatch/SafetyInformation/SafetyalertsforHumanMedicalProducts/ucm243843.htm. Accessed June 17, 2013.
12. US Food and Drug Administration. Magnesium Sulfate: Drug Safety Communication—Recommendation against Prolonged Use in Preterm Labor. Published May 30, 2013. http://www.fda.gov/Safety/MedWatch/SafetyInformation/SafetyAlertsforHumanMedicalProducts/ucm354603.htm. Accessed June 17, 2013.
9. US Food and Drug Administration. Terbutaline: Label Change—Warnings Against Use for Treatment of Preterm Labor. Published February 17, 2011. http://www.fda.gov/Safety/MedWatch/SafetyInformation/SafetyalertsforHumanMedicalProducts/ucm243843.htm. Accessed June 17, 2013.
12. US Food and Drug Administration. Magnesium Sulfate: Drug Safety Communication—Recommendation against Prolonged Use in Preterm Labor. Published May 30, 2013. http://www.fda.gov/Safety/MedWatch/SafetyInformation/SafetyAlertsforHumanMedicalProducts/ucm354603.htm. Accessed June 17, 2013.
Beta-adrenergic agonists carry many risks
Fetal effects of magnesium sulfate
Girl, 13, With a Bump on Her Leg
A girl, age 13 years, 4 months, presented to her primary care provider’s office for a well visit. Among her concerns, she mentioned a “bump” she had had on her right leg “for the past six months, maybe longer.” The area felt irritated when touched or when the patient “ran too much.” She had seen no change in the bump since she first noticed it. The patient knew of no trauma or other preceding factors. She denied any fever or warmth, redness, or ecchymosis to the area.
Medical history was unremarkable except for familial short stature and myopia. The patient was the fifth of eight children born to nonconsanguinous parents. She denied any surgical history or hospitalizations and was premenarcheal. She was up to date on all age-appropriate vaccines, with her meningococcal vaccine administered at that visit.
The patient’s blood pressure was 99/58 mm Hg with an apical pulse rate of 82 beats/min. Her growth parameters were following her curve. Her height was 55” (0.3 percentile); weight, 81 lb (7.5 percentile); and BMI, 18.8 (48.6 percentile).
The physical exam was normal with the exception of the musculoskeletal exam. Examination of the lower extremities revealed a palpable, 4 cm x 5 cm lesion at the right distal medial thigh just proximal to the knee. The lesion could not be visualized but on palpation was tender and firm. There was some question as to whether the lesion itself or inflamed soft tissue overlying the lesion was mobile. No overlying warmth, induration, erythema, or ecchymosis was noted.
Passive and active range of motion was intact at the hip and knee. No lesions to the upper extremities were evident, and no scoliosis was seen.
Blood work was done to rule out certain diagnoses. Results from a complete blood count with differential, lactate dehydrogenase (LDH), parathyroid hormone, lipid profiles, thyroid function, and a comprehensive metabolic profile were unremarkable. A low level of vitamin D 25-OH was detected: 21.7 ng/mL (normal range, 32 to 100 ng/mL).
Distal femur x-rays with posteroanterior, lateral, and oblique views were ordered. The imaging revealed a 3 cm x 3 cm lesion projecting from the “distal, somewhat medial” femur, which was diagnosed as a benign femoral osteochondroma. Significant inflammation to the surrounding soft-tissue structures was observed. A questionable old fracture of the osteochondroma was noted. The remaining bony structures and joints appeared normal.
An ultrasound of the lesion was also ordered to investigate soft-tissue swelling. This revealed a hypoechoic collar around the distal end of the osteochondroma, which could represent a fluid collection, hematoma from trauma, or bursitis. The soft tissues were deemed normal.
Because of the extent of inflammation, the radiologist recommended MRI without contrast to rule out bursitis or trauma to the osteochondroma.
DISCUSSION
Osteochondromas, which may be present in up to 3% of the general population, are the most common benign bone tumors.1-3 An osteochondroma is a cartilage-capped bony projection that arises on the external surface of the bone; it contains a marrow cavity that is continuous with the underlying bone.2,4 The majority of osteochondromas are solitary, accounting for perhaps 85% to 90% of all such lesions, and they are typically nonhereditary; the remaining 10% to 15% of osteochondromas are hereditary multiple osteochondromas or exostoses1,2 (see “Definition of Multiple Exostoses Syndrome”2,5,6,7).

Most lesions are painless and slow growing, and they usually occur in children and adolescents.2 They typically stop growing at skeletal maturity with the closure of the growth plates.3,8,9 There is no predilection for males or females in single lesions.2
Solitary osteochondromas typically appear in the lower extremities and at long tubular bone metaphyses,1-3,10 especially on the femur, humerus, tibia, spine, and hip. Any part of the skeleton can be affected, but 30% of lesions occur on the femur and 40% at either the proximal metaphysis of the tibia or the distal metaphysis of the femur.2,11
Most osteochondromas are asymptomatic and are found incidentally.1,3 However, some patients present with local pain as a result of irritation to adjacent structures, limitation of joint motion, growth disturbance, or fracture of the pedicle.3,4,9,11,12 A very small proportion of patients (no more than 1%) with solitary osteochondromas experience malignant transformation.2,3,6,7 No particular blood work is recommended for patients with solitary osteochondromas.2
Differential Diagnosis
In addition to osteochondromas, several other lesions should be considered in the patient with musculoskeletal lesions (see Table 15,6,13-19).
Cartilaginous tumors. Chondrosarcomas are malignant cartilaginous tumors.20-22 They commonly affect long bones, including the humerus and femur, and some flat bones, such as the pelvic bones.13,22 They are most commonly seen in adults, and have no predisposition by gender.13
Chondrosarcomas can be primary (ie, arising de novo) or secondary (developing on preexisting benign cartilaginous neoplasms, including osteochondromas). The majority of chondrosarcomas are slow growing, and they rarely metastasize. It is difficult to differentiate between a benign lesion (such as an osteochondroma) and a chondrosarcoma by either histology or radiology. However, reliable predictors for malignancy include size exceeding 5 cm and location in the axial skeleton.20
Bone tumors.Osteosarcomas are the most common malignant bone tumors in children and adolescents, with 400 to 560 US patients in this age-group diagnosed each year.14-16 Osteosarcomas are uncommon in children younger than 10; their incidence peaks during the early teenage years (median peak age, 16), then declines rapidly among older patients. They are more common in males than females.15
Osteosarcomas commonly develop during periods of rapid bone turnover, such as the adolescent growth spurt. Common sites include the distal femur, proximal humerus, and proximal tibia,15,16 particularly near the knee.13 Usually, osteosarcomas present with nonspecific symptoms, including strain-related pain of several months’ duration, which may disrupt sleep.16 Laboratory findings in affected patients may include elevations in LDH, alkaline phosphatase, and/or ESR.15,23
Physical exam reveals a visible or palpable mass in the affected area, along with decreased joint motion; localized warmth or erythema may also be present. Late signs of osteosarcoma include weight loss, general malaise, and fever. First-line imaging for the patient with a suspected osteosarcoma is x-ray, which will show ill-defined borders, osteoblastic and/or osteolytic features, and an associated soft tissue mass. Advanced imaging, such as MRI, is warranted.16
Ewing sarcoma, the second most common bone tumor in children and adolescents, is an aggressive form of childhood cancer.14,18 Approximately 25% of all Ewing sarcomas arise in soft tissues rather than bones.18 They are more common in whites than in other ethnic groups and have a slight male predominance.13,18 The median age at diagnosis is 15.13 The most common presenting symptoms are tumor related, such as pain or a noticeable mass. While x-rays are usually ordered first, MRI is preferred.18
Soft tissue tumors and masses.Rhabdomyosarcomas are malignancies that account for more than half of the soft tissue sarcomas in children and adolescents. Less than one-fifth of cases occur in the extremities, and most occur in children younger than 10. These lesions have a slight male predominance and are more common in whites than in other patients.14,17,24
Approximately 6% of childhood soft tissue tumors are adipose tissue tumors, which may be benign (eg, lipomas) or malignant (eg, liposarcomas). Lipomas in children account for nearly 4% of all soft tissue tumors and can be classified as superficial (which are often diagnosed clinically) or deep (frequently requiring imaging).25
Lymphomaaccounts for 7% of cancers in US children and adolescents and more than 25% of newly diagnosed cancers in patients between 15 and 19, making it the most common malignancy in adolescents and the third most common in children.26,27 Non-Hodgkin lymphoma is the fourth leading type of malignancy in US adolescents.27 Rarely, lymphomas present with primary event soft tissue involvement.28
Myositis ossificans (MO) is a rare benign disorder involving formation of heterotrophic bone in skeletal muscles and soft tissues.29 Though possible in patients of any age, MO is most commonly seen in adolescents and young adults. Often the result of soft tissue injury (in which case it is referred to as myositis ossificans circumscripta or traumatic), MO develops in areas that are exposed to trauma, such as the anterior thighs or arms. Lesions can be diagnosed via plain x-ray or CT, although MRI and ultrasound can also be useful evaluation tools.17,29,30
Because MO circumscripta typically presents as a painful soft tissue mass, it can be mistaken for a soft tissue sarcoma or an osteosarcoma; radiologic evaluation is required to make the proper diagnosis. Less common forms of MO are myositis ossificans progressiva and myositis ossificans without a history of trauma.29
Ollier diseaseis a rare, nonfamilial disorder characterized by multiple enchondromas (or enchondromatoses), which are distributed asymmetrically with areas of dysplastic cartilage. Enchondromas are benign cartilage tumors that frequently affect long tubular bones along the metaphyses in proximity to the growth plate. The enchondromas result in significant growth abnormalities. About one in 1 million people are diagnosed yearly.5,19 (Similarly, Maffucci syndrome is represented by multiple enchondromas in association with hemangiomas.5)
Ollier disease typically manifests during childhood5 with bone swelling, local pain, and palpable bony masses, which are often associated with bone deformities.19 Patients generally present with an asymmetric shortening of one extremity and the appearance of palpable bony masses on their fingers or toes, which may or may not be associated with pathologic fractures.5,19 In 20% to 50% of patients with Ollier disease, enchondromas are at risk for malignant transformation into chondrosarcomas.5
Vascular malformations. Certain abnormalities of vascular development cause birthmarks and abnormalities of varying degree in underlying tissues.31 They are usually present at birth and grow proportionally to the child’s growth.25,31 However, they can also be seen in later childhood and adolescence.
Radiologic Investigation
Plain radiography of the affected area is the first-line radiologic study to be performed.13 While most osteochondromas can be diagnosed by plain x-ray, cross-sectional imaging via CT or MRI is recommended in lesions with certain characteristics, such as a broad stalk or location in the axial skeleton. Because MRI involves no radiation exposure, it is a particularly good diagnostic tool for children.32
Ultrasound is a good imaging method for evaluating for complications of osteochondromas, including bursa formation or vascular compromise.32
Treatment and Management
Although usually asymptomatic, osteochondroma can trigger some significant symptoms. Osteochondromas are at risk for fracture and can cause body deformities, mechanical joint problems, weakness of the affected limb, numbness, vascular compression, aneurysm, arterial thrombosis, venous thrombosis, pain, acute ischemia, and nerve compression. Clinical signs of malignant transformation include pain, swelling, and increased lesion size.2
Surgical excision is recommended but should be delayed until after the patient has reached skeletal maturity in order to decrease the risk for recurrence.33
Patient Education and Follow-up
In addition to explaining appropriate pain management (eg, NSAIDs), it is especially important for the pediatric NP or PA to encourage the patient with a solitary osteochondroma to follow up with the pediatric orthopedic surgeon. Reasons include the need to monitor growth of the lesion (which is likely to continue in a patient who has not yet reached skeletal maturity) and assess for associated functional or joint problems. Patients should also be advised to seek the specialist’s attention if such problems develop or if pain increases.
Generally, the pediatric clinician should be sufficiently informed to answer questions about this condition from the patient or family. Any follow-up laboratory work recommended by the specialist can also be performed by the pediatric NP or PA.
OUTCOME FOR THE CASE PATIENT
MRI without contrast, as recommended by the radiologist to rule out a bursa or trauma to the osteochondroma, was considered an important part of the follow-up plan. As the patient had not yet reached skeletal maturity, she was referred to a pediatric orthopedic surgeon for possible excision of the lesion, due to its size and the pain associated with running or other exertion.
CONCLUSION
Solitary osteochondromas are the most common benign bone tumors. Although they are generally asymptomatic, pain and other symptoms can arise as a result of irritation to the adjacent structures. In this case, the patient’s chief complaint was an irritating “bump” that she had had on her right leg for at least six months.
Generally, follow-up monitoring of the osteochondroma and orthopedic follow-up care are warranted, at least until the patient reaches skeletal maturity. At that point, surgical excision of the lesion is recommended.
REFERENCES
1. Florez B, Mönckeberg J, Castillo G, Beguiristain J. Solitary osteochondroma long-term follow-up. J Pediatr Orthop B. 2008;17:91-94.
2. Kitsoulis P, Galani V, Stefanaki K, et al. Osteochondromas: review of the clinical, radiological and pathological features. In Vivo. 2008;22:633-646.
3. Ramos-Pascua LR, Sánchez-Herráez S, Alonso-Barrio JA, Alonso-León A. Solitary proximal end of femur osteochondroma: an indication and result of the en bloc resection without hip luxation [in Spanish]. Rev Esp Cir Ortop Traumatol. 2012;56:24-31.
4. Payne WT, Merrell G. Benign bony and soft tissue tumors of the hand. J Hand Surg. 2010;35:1901-1910.
5. Pannier S, Legeai-Mallet L. Hereditary multiple exostoses and enchondromatosis. Best Pract Res Clin Rheumatol. 2008;22:45-54.
6. Bovée JV. Multiple osteochondromas. Orphanet J Rare Dis. 2008;3(3).
7. Staals EL, Bacchini P, Mercuri M, Bertoni F. Dedifferentiated chondrosarcomas arising in preexisting osteochondromas. J Bone Joint Surg Am. 2007;89:987-993.
8. Singh R, Jain M, Siwach R, et al. Large para-articular osteochondroma of the knee joint: a case report. Acta Orthop Traumatol Turc. 2012;46:139-143.
9. Lee JY, Lee S, Joo KB, et al. Intraarticular osteochondroma of shoulder: a case report. Clin Imaging. 2013;37:379-381.
10. Kim Y-C, Ahn JH, Lee JW. Osteochondroma of the distal tibia complicated by a tibialis posterior tendon tear. J Foot Ankle Surg. 2012;51: 660-663.
11. Allagui M, Amara K, Aloui I, et al. Historical giant near-circumferential osteochondroma of the proximal humerus. J Shoulder Elbow Surg. 2010;19:e12-e15.
12. Li M, Luettringhaus T, Walker KR, Cole PA. Operative treatment of femoral neck osteochondroma through a digastric approach in a pediatric patient: a case report and review of the literature. J Pediatr Orthop B. 2012;21:230-234.
13. Hogendoorn PC, Athanasou N, Bielack S, et al; ESMO/EUROBONET Working Group. Bone sarcomas: ESMO clinical practice guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2010;21 suppl 5:v204-v213.
14. Arndt CAS, Rose PS, Folpe AL, Laack NN. Common musculoskeletal tumors of childhood and adolescence. Mayo Clin Proc. 2012;87:475-487.
15. Ta HT, Dass CR, Choong PF, Dunstan DE. Osteosarcoma treatment: state of the art. Cancer Metastasis Rev. 2009;28:247-263.
16. Messerschmitt PJ, Garcia RM, Abdul-Karim FW, et al. Osteosarcoma. J Am Acad Orthop Surg. 2009;17:515-527.
17. Laffan EE, Ngan B-Y, Navarro OM. Pediatric soft-tissue tumors and pseudotumors: MR imaging features with pathologic correlation: Part 2. Tumors of fibroblastic/myofibroblastic, so-called fibrohistiocytic, muscular, lymphomatous, neurogenic, hair matrix, and uncertain origin. Radiographics. 2009;29:e36.
18. Balamuth NJ, Womer RB. Ewing’s sarcoma. Lancet Oncol. 2010;11(2):184.
19. D’Angelo L, Massimi L, Narducci A, Di Rocco C. Ollier disease. Childs Nerv Syst. 2009;25:647-653.
20. Gelderblom H, Hogendoorn PC, Dijkstra SD, et al. The clinical approach towards chondrosarcoma. Oncologist. 2008;13:320-329.
21. Nosratzehi T, Pakfetrat A. Chondrosarcoma. Zahedan J Res Med Sci. 2013;15:64-64.
22. Prado FO, Nishimoto IN, Perez DE, et al. Head and neck chondrosarcoma: analysis of 16 cases. Br J Oral Maxillofacial Surg. 2009;47:555-557.
23. Kim HJ, Chalmers PN, Morris CD. Pediatric osteogenic sarcoma. Curr Opin Pediatr. 2010;22:61-66.
24. Sultan I, Qaddoumi I, Yaser S, et al. Comparing adult and pediatric rhabdomyosarcoma in the surveillance, epidemiology and end results program, 1973 to 2005: an analysis of 2,600 patients. J Clin Oncol. 2009;27:3391-3397.
25. Navarro OM, Laffan EE, Ngan B-Y. Pediatric soft-tissue tumors and pseudo-tumors: MR imaging features with pathologic correlation: Part 1. Imaging approach, pseudotumors, vascular lesions, and adipocytic tumors. Radiographics. 2009;29:887-906.
26. Gross TG, Termuhlen AM. Pediatric non-Hodgkin lymphoma. Curr Hematol Malig Rep. 2008;3:167-173.
27. Hochberg J, Waxman IM, Kelly KM, et al. Adolescent non-Hodgkin lymphoma and Hodgkin lymphoma: state of the science. Br J Haematol. 2009;144:24-40.
28. Derenzini E, Casadei B, Pellegrini C, et al. Non-Hodgkin lymphomas presenting as soft tissue masses: A single center experience and meta-analysis of the published series. Clin Lymphoma Myeloma Leuk. 2012 Dec 12. [Epub ahead of print]
29. Micheli A, Trapani S, Brizzi I, et al. Myositis ossificans circumscripta: a paediatric case and review of the literature. Eur J Pediatr. 2009;168:523-529.
30. McKenzie G, Raby N, Ritchie D. Non-neoplastic soft-tissue masses. Br J Radiol. 2009;82:775-785.
31. Buckmiller LM, Richter GT, Suen JY. Diagnosis and management of hemangiomas and vascular malformations of the head and neck. Oral Dis. 2010;16:405-418.
32. Khanna G, Bennett DL. Pediatric bone lesions: beyond the plain radiographic evaluation. Semin Roentgenol. 2012;47:90-99.
33. Rijal L, Nepal P, Baral S, et al. Solitary diaphyseal exostosis of femur, how common is it? Eur J Orthop Surg Traumatol. 2011;21:363-365.
A girl, age 13 years, 4 months, presented to her primary care provider’s office for a well visit. Among her concerns, she mentioned a “bump” she had had on her right leg “for the past six months, maybe longer.” The area felt irritated when touched or when the patient “ran too much.” She had seen no change in the bump since she first noticed it. The patient knew of no trauma or other preceding factors. She denied any fever or warmth, redness, or ecchymosis to the area.
Medical history was unremarkable except for familial short stature and myopia. The patient was the fifth of eight children born to nonconsanguinous parents. She denied any surgical history or hospitalizations and was premenarcheal. She was up to date on all age-appropriate vaccines, with her meningococcal vaccine administered at that visit.
The patient’s blood pressure was 99/58 mm Hg with an apical pulse rate of 82 beats/min. Her growth parameters were following her curve. Her height was 55” (0.3 percentile); weight, 81 lb (7.5 percentile); and BMI, 18.8 (48.6 percentile).
The physical exam was normal with the exception of the musculoskeletal exam. Examination of the lower extremities revealed a palpable, 4 cm x 5 cm lesion at the right distal medial thigh just proximal to the knee. The lesion could not be visualized but on palpation was tender and firm. There was some question as to whether the lesion itself or inflamed soft tissue overlying the lesion was mobile. No overlying warmth, induration, erythema, or ecchymosis was noted.
Passive and active range of motion was intact at the hip and knee. No lesions to the upper extremities were evident, and no scoliosis was seen.
Blood work was done to rule out certain diagnoses. Results from a complete blood count with differential, lactate dehydrogenase (LDH), parathyroid hormone, lipid profiles, thyroid function, and a comprehensive metabolic profile were unremarkable. A low level of vitamin D 25-OH was detected: 21.7 ng/mL (normal range, 32 to 100 ng/mL).
Distal femur x-rays with posteroanterior, lateral, and oblique views were ordered. The imaging revealed a 3 cm x 3 cm lesion projecting from the “distal, somewhat medial” femur, which was diagnosed as a benign femoral osteochondroma. Significant inflammation to the surrounding soft-tissue structures was observed. A questionable old fracture of the osteochondroma was noted. The remaining bony structures and joints appeared normal.
An ultrasound of the lesion was also ordered to investigate soft-tissue swelling. This revealed a hypoechoic collar around the distal end of the osteochondroma, which could represent a fluid collection, hematoma from trauma, or bursitis. The soft tissues were deemed normal.
Because of the extent of inflammation, the radiologist recommended MRI without contrast to rule out bursitis or trauma to the osteochondroma.
DISCUSSION
Osteochondromas, which may be present in up to 3% of the general population, are the most common benign bone tumors.1-3 An osteochondroma is a cartilage-capped bony projection that arises on the external surface of the bone; it contains a marrow cavity that is continuous with the underlying bone.2,4 The majority of osteochondromas are solitary, accounting for perhaps 85% to 90% of all such lesions, and they are typically nonhereditary; the remaining 10% to 15% of osteochondromas are hereditary multiple osteochondromas or exostoses1,2 (see “Definition of Multiple Exostoses Syndrome”2,5,6,7).

Most lesions are painless and slow growing, and they usually occur in children and adolescents.2 They typically stop growing at skeletal maturity with the closure of the growth plates.3,8,9 There is no predilection for males or females in single lesions.2
Solitary osteochondromas typically appear in the lower extremities and at long tubular bone metaphyses,1-3,10 especially on the femur, humerus, tibia, spine, and hip. Any part of the skeleton can be affected, but 30% of lesions occur on the femur and 40% at either the proximal metaphysis of the tibia or the distal metaphysis of the femur.2,11
Most osteochondromas are asymptomatic and are found incidentally.1,3 However, some patients present with local pain as a result of irritation to adjacent structures, limitation of joint motion, growth disturbance, or fracture of the pedicle.3,4,9,11,12 A very small proportion of patients (no more than 1%) with solitary osteochondromas experience malignant transformation.2,3,6,7 No particular blood work is recommended for patients with solitary osteochondromas.2
Differential Diagnosis
In addition to osteochondromas, several other lesions should be considered in the patient with musculoskeletal lesions (see Table 15,6,13-19).

Cartilaginous tumors. Chondrosarcomas are malignant cartilaginous tumors.20-22 They commonly affect long bones, including the humerus and femur, and some flat bones, such as the pelvic bones.13,22 They are most commonly seen in adults, and have no predisposition by gender.13
Chondrosarcomas can be primary (ie, arising de novo) or secondary (developing on preexisting benign cartilaginous neoplasms, including osteochondromas). The majority of chondrosarcomas are slow growing, and they rarely metastasize. It is difficult to differentiate between a benign lesion (such as an osteochondroma) and a chondrosarcoma by either histology or radiology. However, reliable predictors for malignancy include size exceeding 5 cm and location in the axial skeleton.20
Bone tumors.Osteosarcomas are the most common malignant bone tumors in children and adolescents, with 400 to 560 US patients in this age-group diagnosed each year.14-16 Osteosarcomas are uncommon in children younger than 10; their incidence peaks during the early teenage years (median peak age, 16), then declines rapidly among older patients. They are more common in males than females.15
Osteosarcomas commonly develop during periods of rapid bone turnover, such as the adolescent growth spurt. Common sites include the distal femur, proximal humerus, and proximal tibia,15,16 particularly near the knee.13 Usually, osteosarcomas present with nonspecific symptoms, including strain-related pain of several months’ duration, which may disrupt sleep.16 Laboratory findings in affected patients may include elevations in LDH, alkaline phosphatase, and/or ESR.15,23
Physical exam reveals a visible or palpable mass in the affected area, along with decreased joint motion; localized warmth or erythema may also be present. Late signs of osteosarcoma include weight loss, general malaise, and fever. First-line imaging for the patient with a suspected osteosarcoma is x-ray, which will show ill-defined borders, osteoblastic and/or osteolytic features, and an associated soft tissue mass. Advanced imaging, such as MRI, is warranted.16
Ewing sarcoma, the second most common bone tumor in children and adolescents, is an aggressive form of childhood cancer.14,18 Approximately 25% of all Ewing sarcomas arise in soft tissues rather than bones.18 They are more common in whites than in other ethnic groups and have a slight male predominance.13,18 The median age at diagnosis is 15.13 The most common presenting symptoms are tumor related, such as pain or a noticeable mass. While x-rays are usually ordered first, MRI is preferred.18
Soft tissue tumors and masses.Rhabdomyosarcomas are malignancies that account for more than half of the soft tissue sarcomas in children and adolescents. Less than one-fifth of cases occur in the extremities, and most occur in children younger than 10. These lesions have a slight male predominance and are more common in whites than in other patients.14,17,24
Approximately 6% of childhood soft tissue tumors are adipose tissue tumors, which may be benign (eg, lipomas) or malignant (eg, liposarcomas). Lipomas in children account for nearly 4% of all soft tissue tumors and can be classified as superficial (which are often diagnosed clinically) or deep (frequently requiring imaging).25
Lymphomaaccounts for 7% of cancers in US children and adolescents and more than 25% of newly diagnosed cancers in patients between 15 and 19, making it the most common malignancy in adolescents and the third most common in children.26,27 Non-Hodgkin lymphoma is the fourth leading type of malignancy in US adolescents.27 Rarely, lymphomas present with primary event soft tissue involvement.28
Myositis ossificans (MO) is a rare benign disorder involving formation of heterotrophic bone in skeletal muscles and soft tissues.29 Though possible in patients of any age, MO is most commonly seen in adolescents and young adults. Often the result of soft tissue injury (in which case it is referred to as myositis ossificans circumscripta or traumatic), MO develops in areas that are exposed to trauma, such as the anterior thighs or arms. Lesions can be diagnosed via plain x-ray or CT, although MRI and ultrasound can also be useful evaluation tools.17,29,30
Because MO circumscripta typically presents as a painful soft tissue mass, it can be mistaken for a soft tissue sarcoma or an osteosarcoma; radiologic evaluation is required to make the proper diagnosis. Less common forms of MO are myositis ossificans progressiva and myositis ossificans without a history of trauma.29
Ollier diseaseis a rare, nonfamilial disorder characterized by multiple enchondromas (or enchondromatoses), which are distributed asymmetrically with areas of dysplastic cartilage. Enchondromas are benign cartilage tumors that frequently affect long tubular bones along the metaphyses in proximity to the growth plate. The enchondromas result in significant growth abnormalities. About one in 1 million people are diagnosed yearly.5,19 (Similarly, Maffucci syndrome is represented by multiple enchondromas in association with hemangiomas.5)
Ollier disease typically manifests during childhood5 with bone swelling, local pain, and palpable bony masses, which are often associated with bone deformities.19 Patients generally present with an asymmetric shortening of one extremity and the appearance of palpable bony masses on their fingers or toes, which may or may not be associated with pathologic fractures.5,19 In 20% to 50% of patients with Ollier disease, enchondromas are at risk for malignant transformation into chondrosarcomas.5
Vascular malformations. Certain abnormalities of vascular development cause birthmarks and abnormalities of varying degree in underlying tissues.31 They are usually present at birth and grow proportionally to the child’s growth.25,31 However, they can also be seen in later childhood and adolescence.
Radiologic Investigation
Plain radiography of the affected area is the first-line radiologic study to be performed.13 While most osteochondromas can be diagnosed by plain x-ray, cross-sectional imaging via CT or MRI is recommended in lesions with certain characteristics, such as a broad stalk or location in the axial skeleton. Because MRI involves no radiation exposure, it is a particularly good diagnostic tool for children.32
Ultrasound is a good imaging method for evaluating for complications of osteochondromas, including bursa formation or vascular compromise.32
Treatment and Management
Although usually asymptomatic, osteochondroma can trigger some significant symptoms. Osteochondromas are at risk for fracture and can cause body deformities, mechanical joint problems, weakness of the affected limb, numbness, vascular compression, aneurysm, arterial thrombosis, venous thrombosis, pain, acute ischemia, and nerve compression. Clinical signs of malignant transformation include pain, swelling, and increased lesion size.2
Surgical excision is recommended but should be delayed until after the patient has reached skeletal maturity in order to decrease the risk for recurrence.33
Patient Education and Follow-up
In addition to explaining appropriate pain management (eg, NSAIDs), it is especially important for the pediatric NP or PA to encourage the patient with a solitary osteochondroma to follow up with the pediatric orthopedic surgeon. Reasons include the need to monitor growth of the lesion (which is likely to continue in a patient who has not yet reached skeletal maturity) and assess for associated functional or joint problems. Patients should also be advised to seek the specialist’s attention if such problems develop or if pain increases.
Generally, the pediatric clinician should be sufficiently informed to answer questions about this condition from the patient or family. Any follow-up laboratory work recommended by the specialist can also be performed by the pediatric NP or PA.
OUTCOME FOR THE CASE PATIENT
MRI without contrast, as recommended by the radiologist to rule out a bursa or trauma to the osteochondroma, was considered an important part of the follow-up plan. As the patient had not yet reached skeletal maturity, she was referred to a pediatric orthopedic surgeon for possible excision of the lesion, due to its size and the pain associated with running or other exertion.
CONCLUSION
Solitary osteochondromas are the most common benign bone tumors. Although they are generally asymptomatic, pain and other symptoms can arise as a result of irritation to the adjacent structures. In this case, the patient’s chief complaint was an irritating “bump” that she had had on her right leg for at least six months.
Generally, follow-up monitoring of the osteochondroma and orthopedic follow-up care are warranted, at least until the patient reaches skeletal maturity. At that point, surgical excision of the lesion is recommended.
REFERENCES
1. Florez B, Mönckeberg J, Castillo G, Beguiristain J. Solitary osteochondroma long-term follow-up. J Pediatr Orthop B. 2008;17:91-94.
2. Kitsoulis P, Galani V, Stefanaki K, et al. Osteochondromas: review of the clinical, radiological and pathological features. In Vivo. 2008;22:633-646.
3. Ramos-Pascua LR, Sánchez-Herráez S, Alonso-Barrio JA, Alonso-León A. Solitary proximal end of femur osteochondroma: an indication and result of the en bloc resection without hip luxation [in Spanish]. Rev Esp Cir Ortop Traumatol. 2012;56:24-31.
4. Payne WT, Merrell G. Benign bony and soft tissue tumors of the hand. J Hand Surg. 2010;35:1901-1910.
5. Pannier S, Legeai-Mallet L. Hereditary multiple exostoses and enchondromatosis. Best Pract Res Clin Rheumatol. 2008;22:45-54.
6. Bovée JV. Multiple osteochondromas. Orphanet J Rare Dis. 2008;3(3).
7. Staals EL, Bacchini P, Mercuri M, Bertoni F. Dedifferentiated chondrosarcomas arising in preexisting osteochondromas. J Bone Joint Surg Am. 2007;89:987-993.
8. Singh R, Jain M, Siwach R, et al. Large para-articular osteochondroma of the knee joint: a case report. Acta Orthop Traumatol Turc. 2012;46:139-143.
9. Lee JY, Lee S, Joo KB, et al. Intraarticular osteochondroma of shoulder: a case report. Clin Imaging. 2013;37:379-381.
10. Kim Y-C, Ahn JH, Lee JW. Osteochondroma of the distal tibia complicated by a tibialis posterior tendon tear. J Foot Ankle Surg. 2012;51: 660-663.
11. Allagui M, Amara K, Aloui I, et al. Historical giant near-circumferential osteochondroma of the proximal humerus. J Shoulder Elbow Surg. 2010;19:e12-e15.
12. Li M, Luettringhaus T, Walker KR, Cole PA. Operative treatment of femoral neck osteochondroma through a digastric approach in a pediatric patient: a case report and review of the literature. J Pediatr Orthop B. 2012;21:230-234.
13. Hogendoorn PC, Athanasou N, Bielack S, et al; ESMO/EUROBONET Working Group. Bone sarcomas: ESMO clinical practice guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2010;21 suppl 5:v204-v213.
14. Arndt CAS, Rose PS, Folpe AL, Laack NN. Common musculoskeletal tumors of childhood and adolescence. Mayo Clin Proc. 2012;87:475-487.
15. Ta HT, Dass CR, Choong PF, Dunstan DE. Osteosarcoma treatment: state of the art. Cancer Metastasis Rev. 2009;28:247-263.
16. Messerschmitt PJ, Garcia RM, Abdul-Karim FW, et al. Osteosarcoma. J Am Acad Orthop Surg. 2009;17:515-527.
17. Laffan EE, Ngan B-Y, Navarro OM. Pediatric soft-tissue tumors and pseudotumors: MR imaging features with pathologic correlation: Part 2. Tumors of fibroblastic/myofibroblastic, so-called fibrohistiocytic, muscular, lymphomatous, neurogenic, hair matrix, and uncertain origin. Radiographics. 2009;29:e36.
18. Balamuth NJ, Womer RB. Ewing’s sarcoma. Lancet Oncol. 2010;11(2):184.
19. D’Angelo L, Massimi L, Narducci A, Di Rocco C. Ollier disease. Childs Nerv Syst. 2009;25:647-653.
20. Gelderblom H, Hogendoorn PC, Dijkstra SD, et al. The clinical approach towards chondrosarcoma. Oncologist. 2008;13:320-329.
21. Nosratzehi T, Pakfetrat A. Chondrosarcoma. Zahedan J Res Med Sci. 2013;15:64-64.
22. Prado FO, Nishimoto IN, Perez DE, et al. Head and neck chondrosarcoma: analysis of 16 cases. Br J Oral Maxillofacial Surg. 2009;47:555-557.
23. Kim HJ, Chalmers PN, Morris CD. Pediatric osteogenic sarcoma. Curr Opin Pediatr. 2010;22:61-66.
24. Sultan I, Qaddoumi I, Yaser S, et al. Comparing adult and pediatric rhabdomyosarcoma in the surveillance, epidemiology and end results program, 1973 to 2005: an analysis of 2,600 patients. J Clin Oncol. 2009;27:3391-3397.
25. Navarro OM, Laffan EE, Ngan B-Y. Pediatric soft-tissue tumors and pseudo-tumors: MR imaging features with pathologic correlation: Part 1. Imaging approach, pseudotumors, vascular lesions, and adipocytic tumors. Radiographics. 2009;29:887-906.
26. Gross TG, Termuhlen AM. Pediatric non-Hodgkin lymphoma. Curr Hematol Malig Rep. 2008;3:167-173.
27. Hochberg J, Waxman IM, Kelly KM, et al. Adolescent non-Hodgkin lymphoma and Hodgkin lymphoma: state of the science. Br J Haematol. 2009;144:24-40.
28. Derenzini E, Casadei B, Pellegrini C, et al. Non-Hodgkin lymphomas presenting as soft tissue masses: A single center experience and meta-analysis of the published series. Clin Lymphoma Myeloma Leuk. 2012 Dec 12. [Epub ahead of print]
29. Micheli A, Trapani S, Brizzi I, et al. Myositis ossificans circumscripta: a paediatric case and review of the literature. Eur J Pediatr. 2009;168:523-529.
30. McKenzie G, Raby N, Ritchie D. Non-neoplastic soft-tissue masses. Br J Radiol. 2009;82:775-785.
31. Buckmiller LM, Richter GT, Suen JY. Diagnosis and management of hemangiomas and vascular malformations of the head and neck. Oral Dis. 2010;16:405-418.
32. Khanna G, Bennett DL. Pediatric bone lesions: beyond the plain radiographic evaluation. Semin Roentgenol. 2012;47:90-99.
33. Rijal L, Nepal P, Baral S, et al. Solitary diaphyseal exostosis of femur, how common is it? Eur J Orthop Surg Traumatol. 2011;21:363-365.
A girl, age 13 years, 4 months, presented to her primary care provider’s office for a well visit. Among her concerns, she mentioned a “bump” she had had on her right leg “for the past six months, maybe longer.” The area felt irritated when touched or when the patient “ran too much.” She had seen no change in the bump since she first noticed it. The patient knew of no trauma or other preceding factors. She denied any fever or warmth, redness, or ecchymosis to the area.
Medical history was unremarkable except for familial short stature and myopia. The patient was the fifth of eight children born to nonconsanguinous parents. She denied any surgical history or hospitalizations and was premenarcheal. She was up to date on all age-appropriate vaccines, with her meningococcal vaccine administered at that visit.
The patient’s blood pressure was 99/58 mm Hg with an apical pulse rate of 82 beats/min. Her growth parameters were following her curve. Her height was 55” (0.3 percentile); weight, 81 lb (7.5 percentile); and BMI, 18.8 (48.6 percentile).
The physical exam was normal with the exception of the musculoskeletal exam. Examination of the lower extremities revealed a palpable, 4 cm x 5 cm lesion at the right distal medial thigh just proximal to the knee. The lesion could not be visualized but on palpation was tender and firm. There was some question as to whether the lesion itself or inflamed soft tissue overlying the lesion was mobile. No overlying warmth, induration, erythema, or ecchymosis was noted.
Passive and active range of motion was intact at the hip and knee. No lesions to the upper extremities were evident, and no scoliosis was seen.
Blood work was done to rule out certain diagnoses. Results from a complete blood count with differential, lactate dehydrogenase (LDH), parathyroid hormone, lipid profiles, thyroid function, and a comprehensive metabolic profile were unremarkable. A low level of vitamin D 25-OH was detected: 21.7 ng/mL (normal range, 32 to 100 ng/mL).
Distal femur x-rays with posteroanterior, lateral, and oblique views were ordered. The imaging revealed a 3 cm x 3 cm lesion projecting from the “distal, somewhat medial” femur, which was diagnosed as a benign femoral osteochondroma. Significant inflammation to the surrounding soft-tissue structures was observed. A questionable old fracture of the osteochondroma was noted. The remaining bony structures and joints appeared normal.
An ultrasound of the lesion was also ordered to investigate soft-tissue swelling. This revealed a hypoechoic collar around the distal end of the osteochondroma, which could represent a fluid collection, hematoma from trauma, or bursitis. The soft tissues were deemed normal.
Because of the extent of inflammation, the radiologist recommended MRI without contrast to rule out bursitis or trauma to the osteochondroma.
DISCUSSION
Osteochondromas, which may be present in up to 3% of the general population, are the most common benign bone tumors.1-3 An osteochondroma is a cartilage-capped bony projection that arises on the external surface of the bone; it contains a marrow cavity that is continuous with the underlying bone.2,4 The majority of osteochondromas are solitary, accounting for perhaps 85% to 90% of all such lesions, and they are typically nonhereditary; the remaining 10% to 15% of osteochondromas are hereditary multiple osteochondromas or exostoses1,2 (see “Definition of Multiple Exostoses Syndrome”2,5,6,7).

Most lesions are painless and slow growing, and they usually occur in children and adolescents.2 They typically stop growing at skeletal maturity with the closure of the growth plates.3,8,9 There is no predilection for males or females in single lesions.2
Solitary osteochondromas typically appear in the lower extremities and at long tubular bone metaphyses,1-3,10 especially on the femur, humerus, tibia, spine, and hip. Any part of the skeleton can be affected, but 30% of lesions occur on the femur and 40% at either the proximal metaphysis of the tibia or the distal metaphysis of the femur.2,11
Most osteochondromas are asymptomatic and are found incidentally.1,3 However, some patients present with local pain as a result of irritation to adjacent structures, limitation of joint motion, growth disturbance, or fracture of the pedicle.3,4,9,11,12 A very small proportion of patients (no more than 1%) with solitary osteochondromas experience malignant transformation.2,3,6,7 No particular blood work is recommended for patients with solitary osteochondromas.2
Differential Diagnosis
In addition to osteochondromas, several other lesions should be considered in the patient with musculoskeletal lesions (see Table 15,6,13-19).

Cartilaginous tumors. Chondrosarcomas are malignant cartilaginous tumors.20-22 They commonly affect long bones, including the humerus and femur, and some flat bones, such as the pelvic bones.13,22 They are most commonly seen in adults, and have no predisposition by gender.13
Chondrosarcomas can be primary (ie, arising de novo) or secondary (developing on preexisting benign cartilaginous neoplasms, including osteochondromas). The majority of chondrosarcomas are slow growing, and they rarely metastasize. It is difficult to differentiate between a benign lesion (such as an osteochondroma) and a chondrosarcoma by either histology or radiology. However, reliable predictors for malignancy include size exceeding 5 cm and location in the axial skeleton.20
Bone tumors.Osteosarcomas are the most common malignant bone tumors in children and adolescents, with 400 to 560 US patients in this age-group diagnosed each year.14-16 Osteosarcomas are uncommon in children younger than 10; their incidence peaks during the early teenage years (median peak age, 16), then declines rapidly among older patients. They are more common in males than females.15
Osteosarcomas commonly develop during periods of rapid bone turnover, such as the adolescent growth spurt. Common sites include the distal femur, proximal humerus, and proximal tibia,15,16 particularly near the knee.13 Usually, osteosarcomas present with nonspecific symptoms, including strain-related pain of several months’ duration, which may disrupt sleep.16 Laboratory findings in affected patients may include elevations in LDH, alkaline phosphatase, and/or ESR.15,23
Physical exam reveals a visible or palpable mass in the affected area, along with decreased joint motion; localized warmth or erythema may also be present. Late signs of osteosarcoma include weight loss, general malaise, and fever. First-line imaging for the patient with a suspected osteosarcoma is x-ray, which will show ill-defined borders, osteoblastic and/or osteolytic features, and an associated soft tissue mass. Advanced imaging, such as MRI, is warranted.16
Ewing sarcoma, the second most common bone tumor in children and adolescents, is an aggressive form of childhood cancer.14,18 Approximately 25% of all Ewing sarcomas arise in soft tissues rather than bones.18 They are more common in whites than in other ethnic groups and have a slight male predominance.13,18 The median age at diagnosis is 15.13 The most common presenting symptoms are tumor related, such as pain or a noticeable mass. While x-rays are usually ordered first, MRI is preferred.18
Soft tissue tumors and masses.Rhabdomyosarcomas are malignancies that account for more than half of the soft tissue sarcomas in children and adolescents. Less than one-fifth of cases occur in the extremities, and most occur in children younger than 10. These lesions have a slight male predominance and are more common in whites than in other patients.14,17,24
Approximately 6% of childhood soft tissue tumors are adipose tissue tumors, which may be benign (eg, lipomas) or malignant (eg, liposarcomas). Lipomas in children account for nearly 4% of all soft tissue tumors and can be classified as superficial (which are often diagnosed clinically) or deep (frequently requiring imaging).25
Lymphomaaccounts for 7% of cancers in US children and adolescents and more than 25% of newly diagnosed cancers in patients between 15 and 19, making it the most common malignancy in adolescents and the third most common in children.26,27 Non-Hodgkin lymphoma is the fourth leading type of malignancy in US adolescents.27 Rarely, lymphomas present with primary event soft tissue involvement.28
Myositis ossificans (MO) is a rare benign disorder involving formation of heterotrophic bone in skeletal muscles and soft tissues.29 Though possible in patients of any age, MO is most commonly seen in adolescents and young adults. Often the result of soft tissue injury (in which case it is referred to as myositis ossificans circumscripta or traumatic), MO develops in areas that are exposed to trauma, such as the anterior thighs or arms. Lesions can be diagnosed via plain x-ray or CT, although MRI and ultrasound can also be useful evaluation tools.17,29,30
Because MO circumscripta typically presents as a painful soft tissue mass, it can be mistaken for a soft tissue sarcoma or an osteosarcoma; radiologic evaluation is required to make the proper diagnosis. Less common forms of MO are myositis ossificans progressiva and myositis ossificans without a history of trauma.29
Ollier diseaseis a rare, nonfamilial disorder characterized by multiple enchondromas (or enchondromatoses), which are distributed asymmetrically with areas of dysplastic cartilage. Enchondromas are benign cartilage tumors that frequently affect long tubular bones along the metaphyses in proximity to the growth plate. The enchondromas result in significant growth abnormalities. About one in 1 million people are diagnosed yearly.5,19 (Similarly, Maffucci syndrome is represented by multiple enchondromas in association with hemangiomas.5)
Ollier disease typically manifests during childhood5 with bone swelling, local pain, and palpable bony masses, which are often associated with bone deformities.19 Patients generally present with an asymmetric shortening of one extremity and the appearance of palpable bony masses on their fingers or toes, which may or may not be associated with pathologic fractures.5,19 In 20% to 50% of patients with Ollier disease, enchondromas are at risk for malignant transformation into chondrosarcomas.5
Vascular malformations. Certain abnormalities of vascular development cause birthmarks and abnormalities of varying degree in underlying tissues.31 They are usually present at birth and grow proportionally to the child’s growth.25,31 However, they can also be seen in later childhood and adolescence.
Radiologic Investigation
Plain radiography of the affected area is the first-line radiologic study to be performed.13 While most osteochondromas can be diagnosed by plain x-ray, cross-sectional imaging via CT or MRI is recommended in lesions with certain characteristics, such as a broad stalk or location in the axial skeleton. Because MRI involves no radiation exposure, it is a particularly good diagnostic tool for children.32
Ultrasound is a good imaging method for evaluating for complications of osteochondromas, including bursa formation or vascular compromise.32
Treatment and Management
Although usually asymptomatic, osteochondroma can trigger some significant symptoms. Osteochondromas are at risk for fracture and can cause body deformities, mechanical joint problems, weakness of the affected limb, numbness, vascular compression, aneurysm, arterial thrombosis, venous thrombosis, pain, acute ischemia, and nerve compression. Clinical signs of malignant transformation include pain, swelling, and increased lesion size.2
Surgical excision is recommended but should be delayed until after the patient has reached skeletal maturity in order to decrease the risk for recurrence.33
Patient Education and Follow-up
In addition to explaining appropriate pain management (eg, NSAIDs), it is especially important for the pediatric NP or PA to encourage the patient with a solitary osteochondroma to follow up with the pediatric orthopedic surgeon. Reasons include the need to monitor growth of the lesion (which is likely to continue in a patient who has not yet reached skeletal maturity) and assess for associated functional or joint problems. Patients should also be advised to seek the specialist’s attention if such problems develop or if pain increases.
Generally, the pediatric clinician should be sufficiently informed to answer questions about this condition from the patient or family. Any follow-up laboratory work recommended by the specialist can also be performed by the pediatric NP or PA.
OUTCOME FOR THE CASE PATIENT
MRI without contrast, as recommended by the radiologist to rule out a bursa or trauma to the osteochondroma, was considered an important part of the follow-up plan. As the patient had not yet reached skeletal maturity, she was referred to a pediatric orthopedic surgeon for possible excision of the lesion, due to its size and the pain associated with running or other exertion.
CONCLUSION
Solitary osteochondromas are the most common benign bone tumors. Although they are generally asymptomatic, pain and other symptoms can arise as a result of irritation to the adjacent structures. In this case, the patient’s chief complaint was an irritating “bump” that she had had on her right leg for at least six months.
Generally, follow-up monitoring of the osteochondroma and orthopedic follow-up care are warranted, at least until the patient reaches skeletal maturity. At that point, surgical excision of the lesion is recommended.
REFERENCES
1. Florez B, Mönckeberg J, Castillo G, Beguiristain J. Solitary osteochondroma long-term follow-up. J Pediatr Orthop B. 2008;17:91-94.
2. Kitsoulis P, Galani V, Stefanaki K, et al. Osteochondromas: review of the clinical, radiological and pathological features. In Vivo. 2008;22:633-646.
3. Ramos-Pascua LR, Sánchez-Herráez S, Alonso-Barrio JA, Alonso-León A. Solitary proximal end of femur osteochondroma: an indication and result of the en bloc resection without hip luxation [in Spanish]. Rev Esp Cir Ortop Traumatol. 2012;56:24-31.
4. Payne WT, Merrell G. Benign bony and soft tissue tumors of the hand. J Hand Surg. 2010;35:1901-1910.
5. Pannier S, Legeai-Mallet L. Hereditary multiple exostoses and enchondromatosis. Best Pract Res Clin Rheumatol. 2008;22:45-54.
6. Bovée JV. Multiple osteochondromas. Orphanet J Rare Dis. 2008;3(3).
7. Staals EL, Bacchini P, Mercuri M, Bertoni F. Dedifferentiated chondrosarcomas arising in preexisting osteochondromas. J Bone Joint Surg Am. 2007;89:987-993.
8. Singh R, Jain M, Siwach R, et al. Large para-articular osteochondroma of the knee joint: a case report. Acta Orthop Traumatol Turc. 2012;46:139-143.
9. Lee JY, Lee S, Joo KB, et al. Intraarticular osteochondroma of shoulder: a case report. Clin Imaging. 2013;37:379-381.
10. Kim Y-C, Ahn JH, Lee JW. Osteochondroma of the distal tibia complicated by a tibialis posterior tendon tear. J Foot Ankle Surg. 2012;51: 660-663.
11. Allagui M, Amara K, Aloui I, et al. Historical giant near-circumferential osteochondroma of the proximal humerus. J Shoulder Elbow Surg. 2010;19:e12-e15.
12. Li M, Luettringhaus T, Walker KR, Cole PA. Operative treatment of femoral neck osteochondroma through a digastric approach in a pediatric patient: a case report and review of the literature. J Pediatr Orthop B. 2012;21:230-234.
13. Hogendoorn PC, Athanasou N, Bielack S, et al; ESMO/EUROBONET Working Group. Bone sarcomas: ESMO clinical practice guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2010;21 suppl 5:v204-v213.
14. Arndt CAS, Rose PS, Folpe AL, Laack NN. Common musculoskeletal tumors of childhood and adolescence. Mayo Clin Proc. 2012;87:475-487.
15. Ta HT, Dass CR, Choong PF, Dunstan DE. Osteosarcoma treatment: state of the art. Cancer Metastasis Rev. 2009;28:247-263.
16. Messerschmitt PJ, Garcia RM, Abdul-Karim FW, et al. Osteosarcoma. J Am Acad Orthop Surg. 2009;17:515-527.
17. Laffan EE, Ngan B-Y, Navarro OM. Pediatric soft-tissue tumors and pseudotumors: MR imaging features with pathologic correlation: Part 2. Tumors of fibroblastic/myofibroblastic, so-called fibrohistiocytic, muscular, lymphomatous, neurogenic, hair matrix, and uncertain origin. Radiographics. 2009;29:e36.
18. Balamuth NJ, Womer RB. Ewing’s sarcoma. Lancet Oncol. 2010;11(2):184.
19. D’Angelo L, Massimi L, Narducci A, Di Rocco C. Ollier disease. Childs Nerv Syst. 2009;25:647-653.
20. Gelderblom H, Hogendoorn PC, Dijkstra SD, et al. The clinical approach towards chondrosarcoma. Oncologist. 2008;13:320-329.
21. Nosratzehi T, Pakfetrat A. Chondrosarcoma. Zahedan J Res Med Sci. 2013;15:64-64.
22. Prado FO, Nishimoto IN, Perez DE, et al. Head and neck chondrosarcoma: analysis of 16 cases. Br J Oral Maxillofacial Surg. 2009;47:555-557.
23. Kim HJ, Chalmers PN, Morris CD. Pediatric osteogenic sarcoma. Curr Opin Pediatr. 2010;22:61-66.
24. Sultan I, Qaddoumi I, Yaser S, et al. Comparing adult and pediatric rhabdomyosarcoma in the surveillance, epidemiology and end results program, 1973 to 2005: an analysis of 2,600 patients. J Clin Oncol. 2009;27:3391-3397.
25. Navarro OM, Laffan EE, Ngan B-Y. Pediatric soft-tissue tumors and pseudo-tumors: MR imaging features with pathologic correlation: Part 1. Imaging approach, pseudotumors, vascular lesions, and adipocytic tumors. Radiographics. 2009;29:887-906.
26. Gross TG, Termuhlen AM. Pediatric non-Hodgkin lymphoma. Curr Hematol Malig Rep. 2008;3:167-173.
27. Hochberg J, Waxman IM, Kelly KM, et al. Adolescent non-Hodgkin lymphoma and Hodgkin lymphoma: state of the science. Br J Haematol. 2009;144:24-40.
28. Derenzini E, Casadei B, Pellegrini C, et al. Non-Hodgkin lymphomas presenting as soft tissue masses: A single center experience and meta-analysis of the published series. Clin Lymphoma Myeloma Leuk. 2012 Dec 12. [Epub ahead of print]
29. Micheli A, Trapani S, Brizzi I, et al. Myositis ossificans circumscripta: a paediatric case and review of the literature. Eur J Pediatr. 2009;168:523-529.
30. McKenzie G, Raby N, Ritchie D. Non-neoplastic soft-tissue masses. Br J Radiol. 2009;82:775-785.
31. Buckmiller LM, Richter GT, Suen JY. Diagnosis and management of hemangiomas and vascular malformations of the head and neck. Oral Dis. 2010;16:405-418.
32. Khanna G, Bennett DL. Pediatric bone lesions: beyond the plain radiographic evaluation. Semin Roentgenol. 2012;47:90-99.
33. Rijal L, Nepal P, Baral S, et al. Solitary diaphyseal exostosis of femur, how common is it? Eur J Orthop Surg Traumatol. 2011;21:363-365.
Nonmedically indicated early term delivery: Are your patients requesting it before 39 weeks?
There’s a serious push to end the practice of elective early term delivery once and for all. Not only has the American College of Obstetricians and Gynecologists (ACOG) teamed up with the March of Dimes to curtail nonmedically indicated deliveries between 37 and 39 weeks of gestation, but in April 2013, ACOG published a Committee Opinion on the issue, stating, in part:
| Although there are specific indications for delivery before 39 weeks of gestation, a nonmedically indicated early term delivery is not appropriate…There are greater reported rates of morbidity and mortality among neonates and infants delivered during the early term period, compared with those delivered at 39 weeks and 40 weeks of gestation. The differences between 37 weeks of gestation and 39 weeks of gestation are consistent, larger, and statistically significant across multiple studies.1 |
According to ACOG, medically justified indications for early term delivery include:
-
complications of hypertension, including preeclampsia, eclampsia, and gestational hypertension
-
history of myomectomy or classical cesarean delivery
-
multiple gestation
-
fetal growth restriction
-
congenital malformation
-
placenta previa, placenta accreta, or placental abruption
-
oligohydramnios
-
poorly controlled pregestational or gestational diabetes, or pregestational diabetes in combination with vascular disease
-
chorioamnionitis
-
premature rupture of membranes
-
alloimmunization of pregnancy with suspected or known effects on the fetus.1
Among the nonmedically justified indications for early term delivery are:
-
maternal intolerance to late pregnancy
-
previous complication of labor
-
history of shoulder dystocia
-
suspected fetal macrosomia
-
history of rapid labor
-
mother lives far from hospital.
Some physicians may consider a positive test for fetal lung maturity an indication for early term delivery as well, but ACOG very clearly states that this practice is unjustified.
“The rate of respiratory morbidity remains higher among neonates delivered during both the late-preterm and early term periods when compared with neonates delivered at 39 weeks of gestation,” the ACOG Committee Opinion states. “However, because nonrespiratory morbidity also is increased, documentation of fetal pulmonary maturity does not justify early nonindicated delivery.”1
ACOG also points out that, “at least one state Medicaid agency has stopped reimbursement for nonindicated deliveries before 39 weeks of gestation.”1
COMPLICATIONS ASSOCIATED WITH EARLY TERM DELIVERY
Increased likelihood of admission to the neonatal intensive care unit (NICU). Among infants delivered by nonmedically indicated cesarean, 17.8% of infants delivered at 37 to 38 weeks and 8% of those delivered at 38 to 39 weeks required NICU admission for an average of 4.5 days, compared with 4.6% of infants delivered at 39 weeks or beyond.2
Respiratory distress. Infants born at 37 weeks’ gestation have three times the risk of respiratory distress syndrome of infants born at 38 weeks, and infants born at 38 weeks have 7.5 times the rate of respiratory distress syndrome of infants born at 39 to 41 weeks.3 In addition, infants born at 37 to 38 weeks’ gestation have a significantly elevated risk of transient tachypnea of the newborn (TTN) and persistent pulmonary hypertension.3
When the infant is delivered by cesarean, the risk of respiratory morbidity is heightened further because cesarean delivery is an independent risk factor for such morbidity.3
In a cohort of consecutive women undergoing elective repeat cesarean delivery, Tita and colleagues found increased rates of adverse respiratory outcomes, need for mechanical ventilation, newborn sepsis, hypoglycemia, NICU admission, and hospitalization. These outcomes were increased by a factor of 1.8 to 4.2 for births at 37 weeks and by a factor of 1.3 to 2.1 for births at 38 weeks, compared with delivery at 39 weeks’ gestation.4
Cerebral palsy. In a Norwegian birth cohort of 1,682,441 singleton term births (no congenital anomalies) followed for a minimum of 4 and a maximum of 20 years, the rate of cerebral palsy was 2.3 times higher at 37 weeks and 1.5 times higher at 38 weeks than it was at 39 to 41 weeks of gestation.5
Neonatal mortality. The relative risk of neonatal mortality among infants born at 37 weeks’ gestation, compared with those born at 39 weeks, is 2.3, and it is 1.4 among infants born at 38 weeks. ACOG notes, “these increased mortality rates need to be balanced against the ongoing risk of stillbirth from week to week in the early term pregnancy.”1
Other moribidities. ACOG also lists pneumonia, hypoglycemia, and a 5-minute Apgar score of less than 7 as potential morbidities associated with early term delivery.1
When patients ask for early term delivery
Although most clinicians are aware of the risks of nonmedically indicated early term delivery, many patients aren’t, and a significant number of patients request it.
In an effort to gauge the extent of patient requests for early term delivery, we polled the members of the OBG Management Virtual Board of Editors. More than 90% of respondents reported that their patients still request elective early term delivery. How often these requests are made varies from “rarely” to “daily,” with most respondents reporting requests once or twice per month.
The most common reason given for such a request: “They are just tired of being pregnant,” one VBE member reported.
Family logistics is another frequent justification.
“Our practice provides obstetric services to a large military population as well as a large geographic area,” said E. William McGrath Jr., MD, of Fernandina Beach, Florida. “Military deployment of a spouse and large travel distances are common reasons for induction requests prior to 39 weeks.”
HOW TO MANAGE PATIENT REQUESTS FOR ELECTIVE EARLY TERM DELIVERY
“We are careful to empathize with rather than criticize the patient and her family for the early delivery request,” Dr. McGrath explained. “Our providers cite ACOG guidelines, but we also mention the statements and policies of the March of Dimes, which disallows elective deliveries prior to 39 weeks. The March of Dimes has greater name recognition among the general public than ACOG does. We attempt to make the patient feel good about her request for early delivery, regardless of the reason—and help her feel even better about her decision to withdraw the request once she learns about the potential complications.”
“I tell all my patients that unless there is a maternal or fetal indication or a strong psychosocial indication, I will not induce them,” reported Sabina K. Cherian, MD, of Houston. “It is usually the multiparous patients who have had previous deliveries at earlier gestational ages who request these early inductions.”
“I tell patients that their due date is arbitrary and not an exact date in which we can guarantee that everything is ok,” said Brian Bernick, MD, of Boca Raton, Florida. Accordingly, “I advise them that their baby is not fully developed until at least 39 weeks. An early, unindicated induction puts both the baby and mother at risk. Lastly, I remind them that a healthy baby and mom are worth the wait.”
“I counsel my patients that even normal pregnancies with infants born at 37 to 38 weeks have a higher rate of complications, compared with those born at 39 weeks gestation, and that an earlier induction may also be more likely to lead to cesarean if the cervix is not yet favorable,” said Devin Namaky, MD, of Cincinnati, Ohio.
One simple response to a patient’s request for early term delivery?
It isn’t possible.
Increasing numbers of hospitals are establishing firm policies against elective early term delivery.
“Our hospital has a hard stop,” said Michael Kirwin, MD, of Freehold, New Jersey. “That makes it easy for me to tell the patient, ‘No.’”
We want to hear from you! Tell us what you think.
There’s a serious push to end the practice of elective early term delivery once and for all. Not only has the American College of Obstetricians and Gynecologists (ACOG) teamed up with the March of Dimes to curtail nonmedically indicated deliveries between 37 and 39 weeks of gestation, but in April 2013, ACOG published a Committee Opinion on the issue, stating, in part:
| Although there are specific indications for delivery before 39 weeks of gestation, a nonmedically indicated early term delivery is not appropriate…There are greater reported rates of morbidity and mortality among neonates and infants delivered during the early term period, compared with those delivered at 39 weeks and 40 weeks of gestation. The differences between 37 weeks of gestation and 39 weeks of gestation are consistent, larger, and statistically significant across multiple studies.1 |
According to ACOG, medically justified indications for early term delivery include:
-
complications of hypertension, including preeclampsia, eclampsia, and gestational hypertension
-
history of myomectomy or classical cesarean delivery
-
multiple gestation
-
fetal growth restriction
-
congenital malformation
-
placenta previa, placenta accreta, or placental abruption
-
oligohydramnios
-
poorly controlled pregestational or gestational diabetes, or pregestational diabetes in combination with vascular disease
-
chorioamnionitis
-
premature rupture of membranes
-
alloimmunization of pregnancy with suspected or known effects on the fetus.1
Among the nonmedically justified indications for early term delivery are:
-
maternal intolerance to late pregnancy
-
previous complication of labor
-
history of shoulder dystocia
-
suspected fetal macrosomia
-
history of rapid labor
-
mother lives far from hospital.
Some physicians may consider a positive test for fetal lung maturity an indication for early term delivery as well, but ACOG very clearly states that this practice is unjustified.
“The rate of respiratory morbidity remains higher among neonates delivered during both the late-preterm and early term periods when compared with neonates delivered at 39 weeks of gestation,” the ACOG Committee Opinion states. “However, because nonrespiratory morbidity also is increased, documentation of fetal pulmonary maturity does not justify early nonindicated delivery.”1
ACOG also points out that, “at least one state Medicaid agency has stopped reimbursement for nonindicated deliveries before 39 weeks of gestation.”1
COMPLICATIONS ASSOCIATED WITH EARLY TERM DELIVERY
Increased likelihood of admission to the neonatal intensive care unit (NICU). Among infants delivered by nonmedically indicated cesarean, 17.8% of infants delivered at 37 to 38 weeks and 8% of those delivered at 38 to 39 weeks required NICU admission for an average of 4.5 days, compared with 4.6% of infants delivered at 39 weeks or beyond.2
Respiratory distress. Infants born at 37 weeks’ gestation have three times the risk of respiratory distress syndrome of infants born at 38 weeks, and infants born at 38 weeks have 7.5 times the rate of respiratory distress syndrome of infants born at 39 to 41 weeks.3 In addition, infants born at 37 to 38 weeks’ gestation have a significantly elevated risk of transient tachypnea of the newborn (TTN) and persistent pulmonary hypertension.3
When the infant is delivered by cesarean, the risk of respiratory morbidity is heightened further because cesarean delivery is an independent risk factor for such morbidity.3
In a cohort of consecutive women undergoing elective repeat cesarean delivery, Tita and colleagues found increased rates of adverse respiratory outcomes, need for mechanical ventilation, newborn sepsis, hypoglycemia, NICU admission, and hospitalization. These outcomes were increased by a factor of 1.8 to 4.2 for births at 37 weeks and by a factor of 1.3 to 2.1 for births at 38 weeks, compared with delivery at 39 weeks’ gestation.4
Cerebral palsy. In a Norwegian birth cohort of 1,682,441 singleton term births (no congenital anomalies) followed for a minimum of 4 and a maximum of 20 years, the rate of cerebral palsy was 2.3 times higher at 37 weeks and 1.5 times higher at 38 weeks than it was at 39 to 41 weeks of gestation.5
Neonatal mortality. The relative risk of neonatal mortality among infants born at 37 weeks’ gestation, compared with those born at 39 weeks, is 2.3, and it is 1.4 among infants born at 38 weeks. ACOG notes, “these increased mortality rates need to be balanced against the ongoing risk of stillbirth from week to week in the early term pregnancy.”1
Other moribidities. ACOG also lists pneumonia, hypoglycemia, and a 5-minute Apgar score of less than 7 as potential morbidities associated with early term delivery.1
When patients ask for early term delivery
Although most clinicians are aware of the risks of nonmedically indicated early term delivery, many patients aren’t, and a significant number of patients request it.
In an effort to gauge the extent of patient requests for early term delivery, we polled the members of the OBG Management Virtual Board of Editors. More than 90% of respondents reported that their patients still request elective early term delivery. How often these requests are made varies from “rarely” to “daily,” with most respondents reporting requests once or twice per month.
The most common reason given for such a request: “They are just tired of being pregnant,” one VBE member reported.
Family logistics is another frequent justification.
“Our practice provides obstetric services to a large military population as well as a large geographic area,” said E. William McGrath Jr., MD, of Fernandina Beach, Florida. “Military deployment of a spouse and large travel distances are common reasons for induction requests prior to 39 weeks.”
HOW TO MANAGE PATIENT REQUESTS FOR ELECTIVE EARLY TERM DELIVERY
“We are careful to empathize with rather than criticize the patient and her family for the early delivery request,” Dr. McGrath explained. “Our providers cite ACOG guidelines, but we also mention the statements and policies of the March of Dimes, which disallows elective deliveries prior to 39 weeks. The March of Dimes has greater name recognition among the general public than ACOG does. We attempt to make the patient feel good about her request for early delivery, regardless of the reason—and help her feel even better about her decision to withdraw the request once she learns about the potential complications.”
“I tell all my patients that unless there is a maternal or fetal indication or a strong psychosocial indication, I will not induce them,” reported Sabina K. Cherian, MD, of Houston. “It is usually the multiparous patients who have had previous deliveries at earlier gestational ages who request these early inductions.”
“I tell patients that their due date is arbitrary and not an exact date in which we can guarantee that everything is ok,” said Brian Bernick, MD, of Boca Raton, Florida. Accordingly, “I advise them that their baby is not fully developed until at least 39 weeks. An early, unindicated induction puts both the baby and mother at risk. Lastly, I remind them that a healthy baby and mom are worth the wait.”
“I counsel my patients that even normal pregnancies with infants born at 37 to 38 weeks have a higher rate of complications, compared with those born at 39 weeks gestation, and that an earlier induction may also be more likely to lead to cesarean if the cervix is not yet favorable,” said Devin Namaky, MD, of Cincinnati, Ohio.
One simple response to a patient’s request for early term delivery?
It isn’t possible.
Increasing numbers of hospitals are establishing firm policies against elective early term delivery.
“Our hospital has a hard stop,” said Michael Kirwin, MD, of Freehold, New Jersey. “That makes it easy for me to tell the patient, ‘No.’”
We want to hear from you! Tell us what you think.
There’s a serious push to end the practice of elective early term delivery once and for all. Not only has the American College of Obstetricians and Gynecologists (ACOG) teamed up with the March of Dimes to curtail nonmedically indicated deliveries between 37 and 39 weeks of gestation, but in April 2013, ACOG published a Committee Opinion on the issue, stating, in part:
| Although there are specific indications for delivery before 39 weeks of gestation, a nonmedically indicated early term delivery is not appropriate…There are greater reported rates of morbidity and mortality among neonates and infants delivered during the early term period, compared with those delivered at 39 weeks and 40 weeks of gestation. The differences between 37 weeks of gestation and 39 weeks of gestation are consistent, larger, and statistically significant across multiple studies.1 |
According to ACOG, medically justified indications for early term delivery include:
-
complications of hypertension, including preeclampsia, eclampsia, and gestational hypertension
-
history of myomectomy or classical cesarean delivery
-
multiple gestation
-
fetal growth restriction
-
congenital malformation
-
placenta previa, placenta accreta, or placental abruption
-
oligohydramnios
-
poorly controlled pregestational or gestational diabetes, or pregestational diabetes in combination with vascular disease
-
chorioamnionitis
-
premature rupture of membranes
-
alloimmunization of pregnancy with suspected or known effects on the fetus.1
Among the nonmedically justified indications for early term delivery are:
-
maternal intolerance to late pregnancy
-
previous complication of labor
-
history of shoulder dystocia
-
suspected fetal macrosomia
-
history of rapid labor
-
mother lives far from hospital.
Some physicians may consider a positive test for fetal lung maturity an indication for early term delivery as well, but ACOG very clearly states that this practice is unjustified.
“The rate of respiratory morbidity remains higher among neonates delivered during both the late-preterm and early term periods when compared with neonates delivered at 39 weeks of gestation,” the ACOG Committee Opinion states. “However, because nonrespiratory morbidity also is increased, documentation of fetal pulmonary maturity does not justify early nonindicated delivery.”1
ACOG also points out that, “at least one state Medicaid agency has stopped reimbursement for nonindicated deliveries before 39 weeks of gestation.”1
COMPLICATIONS ASSOCIATED WITH EARLY TERM DELIVERY
Increased likelihood of admission to the neonatal intensive care unit (NICU). Among infants delivered by nonmedically indicated cesarean, 17.8% of infants delivered at 37 to 38 weeks and 8% of those delivered at 38 to 39 weeks required NICU admission for an average of 4.5 days, compared with 4.6% of infants delivered at 39 weeks or beyond.2
Respiratory distress. Infants born at 37 weeks’ gestation have three times the risk of respiratory distress syndrome of infants born at 38 weeks, and infants born at 38 weeks have 7.5 times the rate of respiratory distress syndrome of infants born at 39 to 41 weeks.3 In addition, infants born at 37 to 38 weeks’ gestation have a significantly elevated risk of transient tachypnea of the newborn (TTN) and persistent pulmonary hypertension.3
When the infant is delivered by cesarean, the risk of respiratory morbidity is heightened further because cesarean delivery is an independent risk factor for such morbidity.3
In a cohort of consecutive women undergoing elective repeat cesarean delivery, Tita and colleagues found increased rates of adverse respiratory outcomes, need for mechanical ventilation, newborn sepsis, hypoglycemia, NICU admission, and hospitalization. These outcomes were increased by a factor of 1.8 to 4.2 for births at 37 weeks and by a factor of 1.3 to 2.1 for births at 38 weeks, compared with delivery at 39 weeks’ gestation.4
Cerebral palsy. In a Norwegian birth cohort of 1,682,441 singleton term births (no congenital anomalies) followed for a minimum of 4 and a maximum of 20 years, the rate of cerebral palsy was 2.3 times higher at 37 weeks and 1.5 times higher at 38 weeks than it was at 39 to 41 weeks of gestation.5
Neonatal mortality. The relative risk of neonatal mortality among infants born at 37 weeks’ gestation, compared with those born at 39 weeks, is 2.3, and it is 1.4 among infants born at 38 weeks. ACOG notes, “these increased mortality rates need to be balanced against the ongoing risk of stillbirth from week to week in the early term pregnancy.”1
Other moribidities. ACOG also lists pneumonia, hypoglycemia, and a 5-minute Apgar score of less than 7 as potential morbidities associated with early term delivery.1
When patients ask for early term delivery
Although most clinicians are aware of the risks of nonmedically indicated early term delivery, many patients aren’t, and a significant number of patients request it.
In an effort to gauge the extent of patient requests for early term delivery, we polled the members of the OBG Management Virtual Board of Editors. More than 90% of respondents reported that their patients still request elective early term delivery. How often these requests are made varies from “rarely” to “daily,” with most respondents reporting requests once or twice per month.
The most common reason given for such a request: “They are just tired of being pregnant,” one VBE member reported.
Family logistics is another frequent justification.
“Our practice provides obstetric services to a large military population as well as a large geographic area,” said E. William McGrath Jr., MD, of Fernandina Beach, Florida. “Military deployment of a spouse and large travel distances are common reasons for induction requests prior to 39 weeks.”
HOW TO MANAGE PATIENT REQUESTS FOR ELECTIVE EARLY TERM DELIVERY
“We are careful to empathize with rather than criticize the patient and her family for the early delivery request,” Dr. McGrath explained. “Our providers cite ACOG guidelines, but we also mention the statements and policies of the March of Dimes, which disallows elective deliveries prior to 39 weeks. The March of Dimes has greater name recognition among the general public than ACOG does. We attempt to make the patient feel good about her request for early delivery, regardless of the reason—and help her feel even better about her decision to withdraw the request once she learns about the potential complications.”
“I tell all my patients that unless there is a maternal or fetal indication or a strong psychosocial indication, I will not induce them,” reported Sabina K. Cherian, MD, of Houston. “It is usually the multiparous patients who have had previous deliveries at earlier gestational ages who request these early inductions.”
“I tell patients that their due date is arbitrary and not an exact date in which we can guarantee that everything is ok,” said Brian Bernick, MD, of Boca Raton, Florida. Accordingly, “I advise them that their baby is not fully developed until at least 39 weeks. An early, unindicated induction puts both the baby and mother at risk. Lastly, I remind them that a healthy baby and mom are worth the wait.”
“I counsel my patients that even normal pregnancies with infants born at 37 to 38 weeks have a higher rate of complications, compared with those born at 39 weeks gestation, and that an earlier induction may also be more likely to lead to cesarean if the cervix is not yet favorable,” said Devin Namaky, MD, of Cincinnati, Ohio.
One simple response to a patient’s request for early term delivery?
It isn’t possible.
Increasing numbers of hospitals are establishing firm policies against elective early term delivery.
“Our hospital has a hard stop,” said Michael Kirwin, MD, of Freehold, New Jersey. “That makes it easy for me to tell the patient, ‘No.’”
We want to hear from you! Tell us what you think.