User login
An ObGyn’s guide to aromatase inhibitors as adjuvant therapy for breast CA
Think breast cancer survivors are unlikely to show up in your practice? You should think again.
At last official count, 2.5 million women in the United States had a history of breast cancer.1 Most of them are now free of malignancy, but others are still grappling with the disease in some form or fashion.2 All need continuing health care.
Roughly two thirds of women who have breast cancer have disease that is hormone-receptor–positive.2,3 Recently updated guidelines from the American Society of Clinical Oncology (ASCO) recommend that adjuvant therapy for postmenopausal women who have hormone-positive breast cancer include an aromatase inhibitor (AI) (see a summary of these guidelines on page 36). That makes it likely that a good number of breast cancer survivors who visit your practice are taking one of these medications: anastrozole (Arimidex), letrozole (Femara), or exemestane (Aromasin). These drugs are antiestrogens, given to postmenopausal women to reduce the likelihood of disease recurrence and progression.
The antiestrogenic properties of these drugs are what make them lifesavers. But the same qualities can create a range of health issues, from increased risk of osteoporosis and fracture to vasomotor and joint symptoms. And although ObGyns are not the physicians who prescribe these drugs, you may be the provider one of these women consults about their side effects and related issues.
To find out the latest on the management of women who are taking one of these agents, we inserted ourselves into the busy schedule of Andrew M. Kaunitz, MD, who agreed to address some fundamental—and some not so basic—questions about the drugs. In this extended Q&A, Dr. Kaunitz touches on mechanism of action, benefits versus risks, common side effects, compliance with therapy, and the ill effects of early discontinuation.
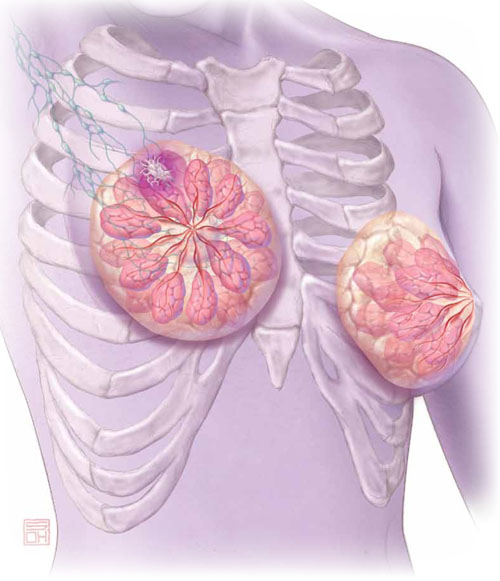
Aromatase inhibitors are better than tamoxifen at reducing the risk of breast cancer recurrence in postmenopausal women who have hormone-receptor–positive disease. But, they increase the risk of osteoporosis and fracture and often cause arthralgias and other complaints that ObGyn practitioners may be called upon to manage.
OBG Management: What is the overall aim of adjuvant endocrine therapy in the setting of breast cancer?
Dr. Kaunitz: Endocrine therapy—specifically, use of an AI—prevents the stimulation of breast cancer cells by endogenous estrogen. In other words, aromatase inhibitors suppress the growth of cancer cells that have estrogen receptors. These drugs also inhibit aromatase near any breast tumor and reduce estrogen levels in breast tissue.
OBG Management: What is the mechanism of action of adjuvant endocrine therapy?
Dr. Kaunitz: In postmenopausal women, androgens are converted to estrogens via the aromatase enzyme, which is present in adipose tissue and other sites. By blocking this enzyme, AIs reduce endogenous estrogen levels by as much as 95%.4
OBG Management: How does that differ from the mechanism of action of tamoxifen, another drug used in breast cancer patients?
Dr. Kaunitz: Tamoxifen is a selective estrogen receptor modulator (SERM). It blocks estrogen in breast tissue selectively, by competitively binding to estrogen receptors.5 However, tamoxifen has estrogenic effects in the uterus, bone, and liver, as well as other tissues.
The efficacy of AIs in preventing breast cancer recurrence in the first 2 years after breast cancer surgery is higher than that of tamoxifen. And unlike tamoxifen, the AIs do not increase the risk of venous thromboembolism or cause endometrial disease.
OBG Management: What effects do aromatase inhibitors have in premenopausal women?
Dr. Kaunitz: These agents are not recommended for use in premenopausal women because, in that population, the lion’s share of estrogen production takes place in the ovary rather than in adipose tissue and muscle. If you were to administer an AI to a premenopausal woman, the reduced hypothalamic and pituitary estrogen feedback could lead to ovarian stimulation—which could increase ovarian steroid production.
OBG Management: What about women who become amenorrheic as a result of chemotherapy or other cancer treatment? Do most oncologists assume that they are postmenopausal and prescribe an aromatase inhibitor?
Dr. Kaunitz: Clinicians should not assume that chemotherapy-induced amenorrhea signals permanent cessation of ovarian function. It is common for ovarian function to return in this setting. Accordingly, follicle-stimulating hormone (FSH) and estradiol levels should be assessed before an AI is considered as adjuvant therapy. Some investigators have suggested that the use of an AI in women who have chemotherapy-induced amenorrhea may actually increase the likelihood that ovarian function will return.6
OBG Management: Do all AIs produce the same effects?
Dr. Kaunitz: The AIs used in women with breast cancer are third-generation drugs. These AIs are classified as steroidal (type 1; exemestane) or nonsteroidal (type 2; anastrozole, letrozole). Exemestane, a steroid derived from androstenedione, inhibits the aromatase enzyme irreversibly. The nonsteroidal AIs are reversible.
Although all three AIs have numerous similarities, there are other distinctions between them in pharmacokinetics, mechanism of action, and toxicity—so they are not completely interchangeable.7 However, from our perspective as ObGyns caring for breast cancer survivors, we can assume that all three AIs will have similar effects on skeletal health and produce similar side effects in postmenopausal women.
- The American Society of Clinical Oncology recommends that adjuvant therapy for postmenopausal women who have hormone-positive breast cancer include an aromatase inhibitor (AI).
- AIs are not recommended for use in premenopausal women.
- By blocking the aromatase enzyme, AIs reduce endogenous estrogen levels by as much as 95%.4
- AIs are more effective than tamoxifen at preventing recurrence in the first 2 years after breast cancer surgery. Postmenopausal women taking an AI have a longer disease-free survival and time to recurrence than do women taking tamoxifen. They also have a lower incidence of contralateral breast cancer.
- The most prominent side effects of AI therapy include arthralgias and hot flushes, while the most serious health impact appears to be a decrease in bone mineral density (BMD). However, endometrial cancer, vaginal bleeding and discharge, cerebrovascular events, venous thromboembolic events, and hot flushes all are less common among women taking an AI than among those taking tamoxifen.
- The FDA strongly discourages the use of estrogen therapy—systemic or local—in women who are taking an AI. Accordingly, bisphospho-nate therapy is recommended as first-line treatment of low bone mineral density. Vaginal lubricants and moisturizers are the mainstay strategy for symptomatic genital atrophy. And gabapentin, selective serotonin reuptake inhibitors, and serotonin norepinephrine reuptake inhibitors are the mainstay of therapy for vasomotor flushes.
- Roughly 50% of women who are prescribed adjuvant endocrine therapy with tamoxifen or an AI discontinue the drug early.32 Early discontinuation is associated with an increase in mortality.33
How much do we know about these drugs?
OBG Management: How long does a woman typically take an AI?
Dr. Kaunitz: At present, in women treated for early-stage, hormone-positive breast cancer, the optimal duration of treatment is unknown. Most oncologists prescribe an AI for 5 years, the length of treatment in a prominent trial of the drugs.8
OBG Management: Is that duration likely to increase as more data come in?
Dr. Kaunitz: The optimal duration of adjuvant AI therapy will be determined by the findings of long-term clinical trials. The National Surgical Adjuvant Breast and Bowel Project B-42 trial may provide new insights into optimal duration of AI treatment after initial tamoxifen therapy.9
OBG Management: How thoroughly have AIs been studied in regard to their use in breast cancer survivors and women who have early-stage disease? How would you characterize the quantity and quality of data that we have so far?
Dr. Kaunitz: AIs have been extensively studied. The most important clinical trials of AIs in this setting, including the Anastrozole, Tamoxifen Alone or in Combination (ATAC) trial (over 6,000 participants, median follow-up of 100 months) and the Breast International Group (BIG) trial (almost 5,000 participants, median follow-up of 76 months) have been detailed in the recent ASCO report.10 These two large landmark trials, in particular, formed the basis for ASCO’s recommendations to routinely incorporate AIs into the therapy of postmenopausal women who have hormone-receptor–positive breast cancer.
OBG Management: In treating breast cancer, what other applications are AIs used for?
Dr. Kaunitz: AIs appear to be slightly more effective than tamoxifen in treating postmenopausal women who have metastatic breast cancer.11
They are approved as first-line therapy for breast cancer in:
- postmenopausal women who have hormone-receptor–positive disease
- postmenopausal women who have locally advanced disease when the hormone receptor is unknown
- postmenopausal women who have metastatic disease.
In addition, they are approved as second-line treatment of advanced breast cancer in postmenopausal women who have disease progression following tamoxifen therapy.12
How effective is AI therapy?
OBG Management: What do we know about the efficacy of these drugs?
Dr. Kaunitz: Most of the studies that have explored efficacy compare an AI with tamoxifen rather than with placebo. In the ATAC trial, after a median follow-up of 33 months, women who were taking anastrozole for early-stage breast cancer had longer disease-free survival and time to recurrence and a lower incidence of contralateral breast cancer than did women taking tamoxifen.8
After 4 years of follow-up in the ATAC trial, women taking anastrozole continued to have more favorable disease-free survival (86.9% vs 84.5% for anastrozole and tamoxifen, respectively; hazard ratio [HR], 0.86; 95% confidence interval [CI], 0.76–0.99; P =.03).13 They also had a more favorable time to recurrence than did women taking tamoxifen (HR, 0.83; 95% CI, 0.71–0.96; P =.015). And women taking anastrozole had a lower incidence of contralateral breast cancer, as well, although this different did not achieve statistical significance (HR, 0.62; 95% CI, 0.38–1.02; P =.062).13
In the BIG study, women taking letrozole had a 5-year disease-free survival estimate of 84.0%, compared with 81.4% for women taking tamoxifen.14 In addition, women taking letrozole were significantly less likely than those taking tamoxifen to experience an event that ended a period of disease-free survival (HR, 0.81; 95% CI, 0.70–0.93; P =.003), especially the event of distant recurrence (HR, 0.73; 95% CI, 0.60–0.88; P =.001).14
And a phase-3 study of exemestane versus tamoxifen in women who had metastatic breast cancer found that the AI produced a superior response rate (46% vs 31% for exemestane and tamoxifen, respectively; odds ratio [OR], 1.85; 95% CI, 1.21–2.82; P =.005). In addition, median progression-free survival was greater with exemestane (9.9 months; 95% CI, 8.7–11.8 months) than with tamoxifen (5.8 months; 95% CI, 5.3–8.1 months). However, there was no difference between arms in progression-free survival or overall survival.
Postmenopausal women who have hormone-receptor–positive breast cancer should consider taking an aromatase inhibitor (AI) to lengthen disease-free survival and lower the risk of recurrence. That’s one of the recommendations in updated guidelines issued earlier this year by the American Society of Clinical Oncology (ASCO). The guidelines suggest a duration of AI therapy of 5 years. In the event that a woman discontinues AI therapy before 5 years are up, she should consider using tamoxifen to bring the total duration of treatment to 5 years.
Other recommendations in the guidelines include:
- Women who have taken tamoxifen for 5 years stand to benefit from switching to an AI for as long as 5 additional years.
- When advising a woman about adjuvant therapy with an AI, clinicians should consider the potential adverse effects, which include osteoporosis, fracture, and arthralgias.
- The third-generation AIs on the market today have not been found to have clinically important differences between them. A woman who cannot tolerate a particular AI should consider switching to a different AI.
- Switching from an AI to tamoxifen (or vice versa) may be an appropriate option for patients who cannot tolerate a drug’s adverse effects. In the event of a switch to tamoxifen, the clinician should counsel the patient about its adverse effects, which include venous thromboembolism and endometrial polyps, hyperplasia, and cancer.
The full guidelines can be accessed at http://jco.ascopubs.org/content/early/2010/07/12/JCO.2009.26.3756.full.pdf.
—Andrew M. Kaunitz, MD
How well tolerated are AIs?
OBG Management: What adverse effects are associated with AIs?
Dr. Kaunitz: Although AIs, overall, are safe medications, their use is associated with a number of adverse events. The most prominent side effects include arthralgias and hot flushes, while the most serious health impact appears to be a decrease in bone mineral density (BMD).
However, the drugs are generally perceived as being easier to tolerate than tamoxifen. That’s because endometrial cancer, vaginal bleeding and discharge, cerebrovascular events, venous thromboembolic events, and hot flushes all are less common among women taking an AI than among those taking tamoxifen.8,13
For overweight women, who face an elevated baseline risk of thromboembolism, the availability of AIs represents a major advantage over tamoxifen. Similarly, AIs offer advantages over tamoxifen for women who have an intact uterus. In addition, postmenopausal women who are taking a selective serotonin reuptake inhibitor (SSRI) such as paroxetine should take an AI rather than tamoxifen, because the concomitant use of SSRIs attenuates the efficacy of tamoxifen.15
What can be done about the most prominent risks?
OBG Management: Let’s focus on what’s probably the best-known adverse effect of AIs—the heightened risk of osteoporosis and fracture. How significant is this effect?
Dr. Kaunitz: Because use of an AI is associated with a profound reduction in endogenous estrogen levels, it also decreases BMD and can lead to osteoporotic fractures. All major phase-3 trials of adjuvant use of AIs in women who have early breast cancer found an increased risk of fracture, with no significant differences between AIs.16
Fortunately, bisphosphonate therapy (oral or intravenous) has been found to reduce bone loss associated with AI therapy.17,18
Assessing baseline BMD is important as women initiate AI therapy. Although no consensus exists regarding follow-up BMD assessment in the setting of AI use, an interval of 2 years is prudent, with the follow-up study preferably performed at the same imaging center and by the same technician as the first. If baseline osteoporosis is observed at the lumbar spine or hip, bisphosphonate therapy is appropriate. If a woman taking an AI has low bone mass (osteopenia) but not osteoporosis, bisphosphonate therapy should be considered if any of the following risk factors are present:
- advanced age
- history of fracture
- glucocorticoid therapy
- parental history of hip fracture
- low body weight
- current smoking status
- excess alcohol consumption
- rheumatoid arthritis
- known risk factors for secondary osteoporosis.19
In breast cancer survivors initiating or continuing AI therapy, it is also appropriate to check a serum vitamin D level and ensure that intake of this nutrient is adequate.
Bisphosphonates may offer oncologic benefits, as well; preliminary evidence suggests that the drugs may prevent recurrence of the cancer and prolong survival.20
OBG Management: What can an ObGyn offer to a woman who complains of significant AI-related arthralgia?
Dr. Kaunitz: Bone and joint symptoms, including aches, pain, and stiffness that is bilateral and not associated with other evidence of rheumatologic disorders, are among the most common side effects of AI therapy. On the plus side of the equation, these symptoms are more likely to be mild to moderate than severe. On the negative side, no specific treatment has been found to be effective in relieving these symptoms, which usually resolve within 2 months or so after discontinuing AI therapy.10
OBG Management: Do AIs have a negative impact on cardiovascular health?
Dr. Kaunitz: Unlike tamoxifen, AIs do not increase the risk of thromboembolic disease. Although the use of an AI may modestly increase the risk of ischemic cardiovascular disease (and lipid changes), compared with tamoxifen, AIs do not appear to increase cardiovascular risk compared with placebo.21,22
OBG Management: Do the antiestrogenic effects of AIs have a significant impact on vaginal health and sexual desire?
Dr. Kaunitz: A review of published reports did not find that the use of AIs has a predictable impact on vaginal dryness or sexual desire.10 However, symptomatic genital atrophy is common in postmenopausal breast cancer survivors, whether or not they use adjuvant therapy.
Although the FDA considers the use of any estrogen (systemic or vaginal) following a diagnosis of breast cancer to be contraindicated, some breast cancer survivors who have symptomatic genital atrophy express an interest in the use of vaginal estrogen. Use of 25-μg estradiol tablets (Vagifem) is associated with a short-term increase in serum estradiol levels.23 This finding has reinforced caution among medical oncologists about the safety of vaginal estrogen in breast cancer survivors. (The 25-μg tablets are no longer marketed.) The lowest dosage of vaginal estrogen available for the treatment of genital atrophy is found in 10-μg estradiol tablets (Vagifem) and the estradiol (2-mg) 3-month vaginal ring (Estring). Nonetheless, in the absence of data, oncologists will likely continue to be concerned that even the lowest dosage of vaginal estrogen could attenuate the favorable impact of AIs on breast cancer. Accordingly, use of vaginal lubricants and moisturizers are the mainstay strategy for symptomatic genital atrophy.
OBG Management: What about the ubiquitous hot flush? Vasomotor symptoms may be more common in women who take tamoxifen, but women on AIs are also bothered by flushes. What are the alternatives to estrogen therapy?
Dr. Kaunitz: Both nonprescription and prescription alternatives are available. Nonprescription options include soy extract and red clover isoflavones, black cohosh, and Chinese herbs. However, none of these over-the-counter approaches has been found to be more effective than placebo in the treatment of menopausal hot flushes.24-26
As for prescription nonhormonal options, ObGyns should recognize that all such treatments are off-label and that none attain the efficacy of hormone therapy in the treatment of vasomotor symptoms. The best-studied and most effective medications include gabapentin, SSRIs (especially paroxetine), and serotonin-norepinephrine reuptake inhibitors (venlafaxine and desvenlafaxine).24,27
OBG Management: Is there any evidence that AIs impair cognitive function in postmenopausal women?
Dr. Kaunitz: Because estrogen is important for cognition, one might anticipate that the profound reduction in background estrogen associated with AI use would impair cognition. Fortunately, the evidence to date is reassuring. Substudies of the BIG trial and the Tamoxifen and Exemestane Adjuvant Multinational Trial indicate that, compared with tamoxifen (which is associated with declines in cognitive function in postmenopausal women), letrozole and exemestane do not diminish cognitive function.28,29
OBG Management: Overall, what is the typical impact of an AI on a woman’s quality of life?
Dr. Kaunitz: Most women do very well on an AI, finding it easier to tolerate than tamoxifen, as we have discussed. However, a significant minority of women is seriously bothered by the adverse effects, with arthralgias usually leading the pack of complaints.30,31
OBG Management: Do some women discontinue adjuvant endocrine therapy because of adverse effects?
Dr. Kaunitz: Regrettably, the answer is “Yes.” A recent study from Kaiser Permanente of northern California found that roughly 50% of women who are prescribed adjuvant endocrine therapy with tamoxifen or an AI discontinue the drug early.32
OBG Management: What can an ObGyn do to encourage compliance with and completion of AI therapy?
Dr. Kaunitz: First, it is critical that patients understand that AIs are lifesaving drugs. As a recent paper points out, early discontinuation or noncompliance with AI therapy is associated with higher mortality.33
Clinicians should also help breast cancer patients understand what common side effects to anticipate with these medications.
Finally, clinicians who understand the financial toll a breast cancer diagnosis and treatment can take are better positioned to help women overcome challenges that may interfere with long-term compliance with AI therapy.
OBG Management: Do you expect the use of AIs in breast cancer survivors to become more commonplace?
Dr. Kaunitz: Given how common breast cancer is, and given the new ASCO guidelines and the extensive literature upon which they are based, ObGyns will be seeing more women using AIs. Although we are not the physicians who prescribe AIs, we need to remain up to date on their benefits and side effects. This important class of drugs is positioned to improve outcomes for postmenopausal women with breast cancer.
- Do certain SSRIs reduce the benefits of tamoxifen in breast cancer survivors?
Examining the Evidence
Andrew M. Kaunitz, MD - Does the clinical breast exam boost the sensitivity of mammography?
Examining the Evidence
Jennifer Griffin, MD, and Mark Pearlman, MD - A guide to lotions and potions for treating vaginal atrophy
Danielle D. Marshall, MD, and Cheryl Iglesia, MD
We want to hear from you! Tell us what you think.
1. Horner MJ, Ries LAG, Krapcho M, et al. eds. SEER Cancer Statistics Review, 1975–2006. Bethesda, Md: National Cancer Institute; 2009.http://seer.cancer.gov/csr/1975_2006. Accessed August 18, 2010.
2. American Cancer Society. Breast cancer facts and figures, 2009–2010. Atlanta, Ga: American Cancer Society; 2010. http://www.cancer.org/Research/CancerFactsFigures/BreastCancerFactsFigures/breast-cancer-facts—figures-2009-2010. Accessed August 18, 2010.
3. Li CI, Daling JR, Malone KE. Incidence of invasive breast cancer by hormone receptor status from 1992 to 1998. J Clin Oncol. 2003;21(1):28-34.
4. Miller WR. Aromatase inhibitors: mechanism of action and role in the treatment of breast cancer. Semin Oncol. 2003;30(4 suppl 14):3-11.
5. Peng J, Sengupta S, Jordan VC. Potential of selective estrogen receptor modulators as treatments and preventives of breast cancer. Anticancer Agents Med Chem. 2009;9(5):481-499.
6. Smith IE, Dowsett M, Yap YS, et al. Adjuvant aromatase inhibitors for early breast cancer after chemotherapy-induced amenorrhea: caution and suggested guidelines. J Clin Oncol. 2006;24(16):2444-2447.
7. Nabholtz JM, Mouret-Reynier MA, Durando X, et al. Comparative review of anastrozole, letrozole and exemestane in the management of early breast cancer. Expert Opin Pharmacother. 2009;10(9):1435-1447.
8. Baum M, Budzar AU, Cuzick J, et al. ATAC Trialists’ Group. Anastrozole alone or in combination with tamoxifen versus tamoxifen alone for adjuvant treatment of postmenopausal women with early breast cancer: first results of the ATAC randomized trial. Lancet. 2002;359(9324):2131-2139.
9. Letrozole in treating postmenopausal women who have received hormone therapy for hormone receptor-positive breast cancer. National Cancer Institute Web site.http://www.cancer.gov/clinicaltrials/NSABP-B-42. Published August 16, 2010. Accessed August 18, 2010.
10. Burstein HJ, Prestrud AA, Seidenfeld J, et al. American Society of Clinical Oncology clinical practice guideline: update on adjuvant endocrine therapy for women with hormone-receptor–positive breast cancer. J Clin Oncol. 2010;28(23):3784-3796.
11. Mauri D, Pavlidis N, Polyzos NP, Ioanidis JP. Survival with aromatase inhibitors and inactivators versus standard hormonal therapy in advanced breast cancer: meta-analysis. J Natl Cancer Inst. 2006;98(18):1285-1291.
12. Arimidex [package insert]. AstraZeneca; 2009.
13. Baum M, Buzdar A, Cuzick J, et al. Anastrozole alone or in combination with tamoxifen versus tamoxifen alone for adjuvant treatment of postmenopausal women with early-stage breast cancer: results of the ATAC (Arimidex, Taxoxifen Alone or in Combination) trial efficacy and safety update analyses. Cancer. 2003;98(9):1802-1810.
14. Thurlimann B, Keshaviah A, Coates AS, et al. Breast International Group (BIG) 1-98 Collaborative Group. A comparison of letrozole and tamoxifen in postmenopausal women with early breast cancer. N Engl J Med. 2005;353(26):2747-2757.
15. Kelly CM, Juurlink DM, Gomes T, et al. Selective serotonin reuptake inhibitors and breast cancer mortality in women receiving tamoxifen: a population-based cohort study. 2010;340:c693.-doi: 10.1136/bmj.c693.
16. Geisler J, Lonning PE. Impact of aromatase inhibitors on bone health in breast cancer patients. J Steroid Biochem Mol Biol. 2010;118(4–5):294-299.
17. Van Poznak C, Hannon RA, Mackey JR, et al. Prevention of aromatase inhibitor-induced bone loss using risedronate: the SABRE trial. J Clin Oncol. 2010;28(6):967-975.
18. Brufsky AM, Bosserman LD, Caradonna RR, et al. Zoledronic acid effectively prevents aromatase-inhibitor associated bone loss in postmenopausal women with early breast cancer receiving adjuvant letrozole. Z-FAST study 36-month follow-up results. Clin Breast Cancer. 2009;9(2):77-85.
19. Kanis JA, Borgstrom F, De Laet C, et al. Assessment of fracture risk. Osteoporosis Int. 2005;16(6):581-589.
20. Reeder JG, Brufsky AM. The role of bisphosphonates in the adjuvant setting for breast cancer. Oncology. 2010;24(6):462-467,475.
21. Nabholtz JM. Long-term safety of aromatase inhibitors in the treatment of breast cancer. Ther Clin Risk Manag. 2008;4(1):189-204.
22. Cuppone F, Bria E, Verma S, et al. Do adjuvant aromatase inhibitors increase the cardiovascular risk in postmenopausal women with early breast cancer? Meta-analysis of randomized trials. Cancer. 2008;112(2):260-267.
23. Kendall A, Dowsett M, Folkerd E, Smith I. Caution: Vaginal estradiol appears to be contraindicated in postmenopausal women on adjuvant aromatase inhibitors. Ann Oncol. 2006;17(4):584-587.
24. Nelson HD. Menopause. Lancet. 2008;371(9614):760-770.
25. Geller SE, Shulman LP, van Breemen RB, et al. Safety and efficacy of black cohosh and red clover for the management of vasomotor symptoms: a randomized controlled trial. Menopause. 2009;16(6):1156-1166.
26. Kaunitz AM. Effective herbal treatment of vasomotor symptoms—are we any closer? Menopause. 2009;16(3):428-429.
27. Archer DF, Dupont CM, Constantine GD, Pickar JH, Olivier S. Study 319 Investigators. Desvenlafaxine for the treatment of vasomotor symptoms associated with menopause: a double-blind, randomized, placebo-controlled trial of efficacy and safety. Am J Obstet Gynecol. 2009;200(3):238.e1-e10.
28. Phillips KA, Ribi K, Sun Z, et al. Cognitive function in postmenopausal women receiving adjuvant letrozole or tamoxifen for breast cancer in the BIG 1-98 randomized trial [published online ahead of print April 10, 2010]. Breast. doi:10.1016/j.breast.2010.03.025.
29. Schilder CM, Seynaeve C, Beex LV, et al. Effects of tamoxifen and exemestane on cognitive functioning of postmenopausal patients with breast cancer: results from the neuropsychological side study of the Tamoxifen and Exemestane Adjuvant Multinational Trial. J Clin Oncol. 2010;28(8):1294-1300.
30. Din OS, Dodwell D, Wakefield RJ, Coleman RE. Aromatase inhibitor-induced arthralgia in early breast cancer: what do we know and how can we find out more? Breast Cancer Res Treat. 2010;120(3):525-538.
31. Burstein HJ. Aromatase inhibitor-associated arthralgia syndrome. Breast. 2007;16(3):223-234.
32. Hershman DL, Kushi LH, Shao T, et al. Early discontinuation and nonadherence to adjuvant hormonal therapy in a cohort of 8,769 early-stage breast cancer patients [published online ahead of print June 28, 2010]. J Clin Oncol. doi: 10.1200/JCO.2009.25.9655.
33. Hershman DL, Shao T, Kushi LH, et al. Early discontinuation and nonadherence to adjuvant hormonal therapy are associated with increased mortality in women with breast cancer [published online ahead of print August 28, 2010]. Breast Cancer Res Treat. doi: 10.1007/ s10549-010-1132-4.
Think breast cancer survivors are unlikely to show up in your practice? You should think again.
At last official count, 2.5 million women in the United States had a history of breast cancer.1 Most of them are now free of malignancy, but others are still grappling with the disease in some form or fashion.2 All need continuing health care.
Roughly two thirds of women who have breast cancer have disease that is hormone-receptor–positive.2,3 Recently updated guidelines from the American Society of Clinical Oncology (ASCO) recommend that adjuvant therapy for postmenopausal women who have hormone-positive breast cancer include an aromatase inhibitor (AI) (see a summary of these guidelines on page 36). That makes it likely that a good number of breast cancer survivors who visit your practice are taking one of these medications: anastrozole (Arimidex), letrozole (Femara), or exemestane (Aromasin). These drugs are antiestrogens, given to postmenopausal women to reduce the likelihood of disease recurrence and progression.
The antiestrogenic properties of these drugs are what make them lifesavers. But the same qualities can create a range of health issues, from increased risk of osteoporosis and fracture to vasomotor and joint symptoms. And although ObGyns are not the physicians who prescribe these drugs, you may be the provider one of these women consults about their side effects and related issues.
To find out the latest on the management of women who are taking one of these agents, we inserted ourselves into the busy schedule of Andrew M. Kaunitz, MD, who agreed to address some fundamental—and some not so basic—questions about the drugs. In this extended Q&A, Dr. Kaunitz touches on mechanism of action, benefits versus risks, common side effects, compliance with therapy, and the ill effects of early discontinuation.

Aromatase inhibitors are better than tamoxifen at reducing the risk of breast cancer recurrence in postmenopausal women who have hormone-receptor–positive disease. But, they increase the risk of osteoporosis and fracture and often cause arthralgias and other complaints that ObGyn practitioners may be called upon to manage.
OBG Management: What is the overall aim of adjuvant endocrine therapy in the setting of breast cancer?
Dr. Kaunitz: Endocrine therapy—specifically, use of an AI—prevents the stimulation of breast cancer cells by endogenous estrogen. In other words, aromatase inhibitors suppress the growth of cancer cells that have estrogen receptors. These drugs also inhibit aromatase near any breast tumor and reduce estrogen levels in breast tissue.
OBG Management: What is the mechanism of action of adjuvant endocrine therapy?
Dr. Kaunitz: In postmenopausal women, androgens are converted to estrogens via the aromatase enzyme, which is present in adipose tissue and other sites. By blocking this enzyme, AIs reduce endogenous estrogen levels by as much as 95%.4
OBG Management: How does that differ from the mechanism of action of tamoxifen, another drug used in breast cancer patients?
Dr. Kaunitz: Tamoxifen is a selective estrogen receptor modulator (SERM). It blocks estrogen in breast tissue selectively, by competitively binding to estrogen receptors.5 However, tamoxifen has estrogenic effects in the uterus, bone, and liver, as well as other tissues.
The efficacy of AIs in preventing breast cancer recurrence in the first 2 years after breast cancer surgery is higher than that of tamoxifen. And unlike tamoxifen, the AIs do not increase the risk of venous thromboembolism or cause endometrial disease.
OBG Management: What effects do aromatase inhibitors have in premenopausal women?
Dr. Kaunitz: These agents are not recommended for use in premenopausal women because, in that population, the lion’s share of estrogen production takes place in the ovary rather than in adipose tissue and muscle. If you were to administer an AI to a premenopausal woman, the reduced hypothalamic and pituitary estrogen feedback could lead to ovarian stimulation—which could increase ovarian steroid production.
OBG Management: What about women who become amenorrheic as a result of chemotherapy or other cancer treatment? Do most oncologists assume that they are postmenopausal and prescribe an aromatase inhibitor?
Dr. Kaunitz: Clinicians should not assume that chemotherapy-induced amenorrhea signals permanent cessation of ovarian function. It is common for ovarian function to return in this setting. Accordingly, follicle-stimulating hormone (FSH) and estradiol levels should be assessed before an AI is considered as adjuvant therapy. Some investigators have suggested that the use of an AI in women who have chemotherapy-induced amenorrhea may actually increase the likelihood that ovarian function will return.6
OBG Management: Do all AIs produce the same effects?
Dr. Kaunitz: The AIs used in women with breast cancer are third-generation drugs. These AIs are classified as steroidal (type 1; exemestane) or nonsteroidal (type 2; anastrozole, letrozole). Exemestane, a steroid derived from androstenedione, inhibits the aromatase enzyme irreversibly. The nonsteroidal AIs are reversible.
Although all three AIs have numerous similarities, there are other distinctions between them in pharmacokinetics, mechanism of action, and toxicity—so they are not completely interchangeable.7 However, from our perspective as ObGyns caring for breast cancer survivors, we can assume that all three AIs will have similar effects on skeletal health and produce similar side effects in postmenopausal women.
- The American Society of Clinical Oncology recommends that adjuvant therapy for postmenopausal women who have hormone-positive breast cancer include an aromatase inhibitor (AI).
- AIs are not recommended for use in premenopausal women.
- By blocking the aromatase enzyme, AIs reduce endogenous estrogen levels by as much as 95%.4
- AIs are more effective than tamoxifen at preventing recurrence in the first 2 years after breast cancer surgery. Postmenopausal women taking an AI have a longer disease-free survival and time to recurrence than do women taking tamoxifen. They also have a lower incidence of contralateral breast cancer.
- The most prominent side effects of AI therapy include arthralgias and hot flushes, while the most serious health impact appears to be a decrease in bone mineral density (BMD). However, endometrial cancer, vaginal bleeding and discharge, cerebrovascular events, venous thromboembolic events, and hot flushes all are less common among women taking an AI than among those taking tamoxifen.
- The FDA strongly discourages the use of estrogen therapy—systemic or local—in women who are taking an AI. Accordingly, bisphospho-nate therapy is recommended as first-line treatment of low bone mineral density. Vaginal lubricants and moisturizers are the mainstay strategy for symptomatic genital atrophy. And gabapentin, selective serotonin reuptake inhibitors, and serotonin norepinephrine reuptake inhibitors are the mainstay of therapy for vasomotor flushes.
- Roughly 50% of women who are prescribed adjuvant endocrine therapy with tamoxifen or an AI discontinue the drug early.32 Early discontinuation is associated with an increase in mortality.33
How much do we know about these drugs?
OBG Management: How long does a woman typically take an AI?
Dr. Kaunitz: At present, in women treated for early-stage, hormone-positive breast cancer, the optimal duration of treatment is unknown. Most oncologists prescribe an AI for 5 years, the length of treatment in a prominent trial of the drugs.8
OBG Management: Is that duration likely to increase as more data come in?
Dr. Kaunitz: The optimal duration of adjuvant AI therapy will be determined by the findings of long-term clinical trials. The National Surgical Adjuvant Breast and Bowel Project B-42 trial may provide new insights into optimal duration of AI treatment after initial tamoxifen therapy.9
OBG Management: How thoroughly have AIs been studied in regard to their use in breast cancer survivors and women who have early-stage disease? How would you characterize the quantity and quality of data that we have so far?
Dr. Kaunitz: AIs have been extensively studied. The most important clinical trials of AIs in this setting, including the Anastrozole, Tamoxifen Alone or in Combination (ATAC) trial (over 6,000 participants, median follow-up of 100 months) and the Breast International Group (BIG) trial (almost 5,000 participants, median follow-up of 76 months) have been detailed in the recent ASCO report.10 These two large landmark trials, in particular, formed the basis for ASCO’s recommendations to routinely incorporate AIs into the therapy of postmenopausal women who have hormone-receptor–positive breast cancer.
OBG Management: In treating breast cancer, what other applications are AIs used for?
Dr. Kaunitz: AIs appear to be slightly more effective than tamoxifen in treating postmenopausal women who have metastatic breast cancer.11
They are approved as first-line therapy for breast cancer in:
- postmenopausal women who have hormone-receptor–positive disease
- postmenopausal women who have locally advanced disease when the hormone receptor is unknown
- postmenopausal women who have metastatic disease.
In addition, they are approved as second-line treatment of advanced breast cancer in postmenopausal women who have disease progression following tamoxifen therapy.12
How effective is AI therapy?
OBG Management: What do we know about the efficacy of these drugs?
Dr. Kaunitz: Most of the studies that have explored efficacy compare an AI with tamoxifen rather than with placebo. In the ATAC trial, after a median follow-up of 33 months, women who were taking anastrozole for early-stage breast cancer had longer disease-free survival and time to recurrence and a lower incidence of contralateral breast cancer than did women taking tamoxifen.8
After 4 years of follow-up in the ATAC trial, women taking anastrozole continued to have more favorable disease-free survival (86.9% vs 84.5% for anastrozole and tamoxifen, respectively; hazard ratio [HR], 0.86; 95% confidence interval [CI], 0.76–0.99; P =.03).13 They also had a more favorable time to recurrence than did women taking tamoxifen (HR, 0.83; 95% CI, 0.71–0.96; P =.015). And women taking anastrozole had a lower incidence of contralateral breast cancer, as well, although this different did not achieve statistical significance (HR, 0.62; 95% CI, 0.38–1.02; P =.062).13
In the BIG study, women taking letrozole had a 5-year disease-free survival estimate of 84.0%, compared with 81.4% for women taking tamoxifen.14 In addition, women taking letrozole were significantly less likely than those taking tamoxifen to experience an event that ended a period of disease-free survival (HR, 0.81; 95% CI, 0.70–0.93; P =.003), especially the event of distant recurrence (HR, 0.73; 95% CI, 0.60–0.88; P =.001).14
And a phase-3 study of exemestane versus tamoxifen in women who had metastatic breast cancer found that the AI produced a superior response rate (46% vs 31% for exemestane and tamoxifen, respectively; odds ratio [OR], 1.85; 95% CI, 1.21–2.82; P =.005). In addition, median progression-free survival was greater with exemestane (9.9 months; 95% CI, 8.7–11.8 months) than with tamoxifen (5.8 months; 95% CI, 5.3–8.1 months). However, there was no difference between arms in progression-free survival or overall survival.
Postmenopausal women who have hormone-receptor–positive breast cancer should consider taking an aromatase inhibitor (AI) to lengthen disease-free survival and lower the risk of recurrence. That’s one of the recommendations in updated guidelines issued earlier this year by the American Society of Clinical Oncology (ASCO). The guidelines suggest a duration of AI therapy of 5 years. In the event that a woman discontinues AI therapy before 5 years are up, she should consider using tamoxifen to bring the total duration of treatment to 5 years.
Other recommendations in the guidelines include:
- Women who have taken tamoxifen for 5 years stand to benefit from switching to an AI for as long as 5 additional years.
- When advising a woman about adjuvant therapy with an AI, clinicians should consider the potential adverse effects, which include osteoporosis, fracture, and arthralgias.
- The third-generation AIs on the market today have not been found to have clinically important differences between them. A woman who cannot tolerate a particular AI should consider switching to a different AI.
- Switching from an AI to tamoxifen (or vice versa) may be an appropriate option for patients who cannot tolerate a drug’s adverse effects. In the event of a switch to tamoxifen, the clinician should counsel the patient about its adverse effects, which include venous thromboembolism and endometrial polyps, hyperplasia, and cancer.
The full guidelines can be accessed at http://jco.ascopubs.org/content/early/2010/07/12/JCO.2009.26.3756.full.pdf.
—Andrew M. Kaunitz, MD
How well tolerated are AIs?
OBG Management: What adverse effects are associated with AIs?
Dr. Kaunitz: Although AIs, overall, are safe medications, their use is associated with a number of adverse events. The most prominent side effects include arthralgias and hot flushes, while the most serious health impact appears to be a decrease in bone mineral density (BMD).
However, the drugs are generally perceived as being easier to tolerate than tamoxifen. That’s because endometrial cancer, vaginal bleeding and discharge, cerebrovascular events, venous thromboembolic events, and hot flushes all are less common among women taking an AI than among those taking tamoxifen.8,13
For overweight women, who face an elevated baseline risk of thromboembolism, the availability of AIs represents a major advantage over tamoxifen. Similarly, AIs offer advantages over tamoxifen for women who have an intact uterus. In addition, postmenopausal women who are taking a selective serotonin reuptake inhibitor (SSRI) such as paroxetine should take an AI rather than tamoxifen, because the concomitant use of SSRIs attenuates the efficacy of tamoxifen.15
What can be done about the most prominent risks?
OBG Management: Let’s focus on what’s probably the best-known adverse effect of AIs—the heightened risk of osteoporosis and fracture. How significant is this effect?
Dr. Kaunitz: Because use of an AI is associated with a profound reduction in endogenous estrogen levels, it also decreases BMD and can lead to osteoporotic fractures. All major phase-3 trials of adjuvant use of AIs in women who have early breast cancer found an increased risk of fracture, with no significant differences between AIs.16
Fortunately, bisphosphonate therapy (oral or intravenous) has been found to reduce bone loss associated with AI therapy.17,18
Assessing baseline BMD is important as women initiate AI therapy. Although no consensus exists regarding follow-up BMD assessment in the setting of AI use, an interval of 2 years is prudent, with the follow-up study preferably performed at the same imaging center and by the same technician as the first. If baseline osteoporosis is observed at the lumbar spine or hip, bisphosphonate therapy is appropriate. If a woman taking an AI has low bone mass (osteopenia) but not osteoporosis, bisphosphonate therapy should be considered if any of the following risk factors are present:
- advanced age
- history of fracture
- glucocorticoid therapy
- parental history of hip fracture
- low body weight
- current smoking status
- excess alcohol consumption
- rheumatoid arthritis
- known risk factors for secondary osteoporosis.19
In breast cancer survivors initiating or continuing AI therapy, it is also appropriate to check a serum vitamin D level and ensure that intake of this nutrient is adequate.
Bisphosphonates may offer oncologic benefits, as well; preliminary evidence suggests that the drugs may prevent recurrence of the cancer and prolong survival.20
OBG Management: What can an ObGyn offer to a woman who complains of significant AI-related arthralgia?
Dr. Kaunitz: Bone and joint symptoms, including aches, pain, and stiffness that is bilateral and not associated with other evidence of rheumatologic disorders, are among the most common side effects of AI therapy. On the plus side of the equation, these symptoms are more likely to be mild to moderate than severe. On the negative side, no specific treatment has been found to be effective in relieving these symptoms, which usually resolve within 2 months or so after discontinuing AI therapy.10
OBG Management: Do AIs have a negative impact on cardiovascular health?
Dr. Kaunitz: Unlike tamoxifen, AIs do not increase the risk of thromboembolic disease. Although the use of an AI may modestly increase the risk of ischemic cardiovascular disease (and lipid changes), compared with tamoxifen, AIs do not appear to increase cardiovascular risk compared with placebo.21,22
OBG Management: Do the antiestrogenic effects of AIs have a significant impact on vaginal health and sexual desire?
Dr. Kaunitz: A review of published reports did not find that the use of AIs has a predictable impact on vaginal dryness or sexual desire.10 However, symptomatic genital atrophy is common in postmenopausal breast cancer survivors, whether or not they use adjuvant therapy.
Although the FDA considers the use of any estrogen (systemic or vaginal) following a diagnosis of breast cancer to be contraindicated, some breast cancer survivors who have symptomatic genital atrophy express an interest in the use of vaginal estrogen. Use of 25-μg estradiol tablets (Vagifem) is associated with a short-term increase in serum estradiol levels.23 This finding has reinforced caution among medical oncologists about the safety of vaginal estrogen in breast cancer survivors. (The 25-μg tablets are no longer marketed.) The lowest dosage of vaginal estrogen available for the treatment of genital atrophy is found in 10-μg estradiol tablets (Vagifem) and the estradiol (2-mg) 3-month vaginal ring (Estring). Nonetheless, in the absence of data, oncologists will likely continue to be concerned that even the lowest dosage of vaginal estrogen could attenuate the favorable impact of AIs on breast cancer. Accordingly, use of vaginal lubricants and moisturizers are the mainstay strategy for symptomatic genital atrophy.
OBG Management: What about the ubiquitous hot flush? Vasomotor symptoms may be more common in women who take tamoxifen, but women on AIs are also bothered by flushes. What are the alternatives to estrogen therapy?
Dr. Kaunitz: Both nonprescription and prescription alternatives are available. Nonprescription options include soy extract and red clover isoflavones, black cohosh, and Chinese herbs. However, none of these over-the-counter approaches has been found to be more effective than placebo in the treatment of menopausal hot flushes.24-26
As for prescription nonhormonal options, ObGyns should recognize that all such treatments are off-label and that none attain the efficacy of hormone therapy in the treatment of vasomotor symptoms. The best-studied and most effective medications include gabapentin, SSRIs (especially paroxetine), and serotonin-norepinephrine reuptake inhibitors (venlafaxine and desvenlafaxine).24,27
OBG Management: Is there any evidence that AIs impair cognitive function in postmenopausal women?
Dr. Kaunitz: Because estrogen is important for cognition, one might anticipate that the profound reduction in background estrogen associated with AI use would impair cognition. Fortunately, the evidence to date is reassuring. Substudies of the BIG trial and the Tamoxifen and Exemestane Adjuvant Multinational Trial indicate that, compared with tamoxifen (which is associated with declines in cognitive function in postmenopausal women), letrozole and exemestane do not diminish cognitive function.28,29
OBG Management: Overall, what is the typical impact of an AI on a woman’s quality of life?
Dr. Kaunitz: Most women do very well on an AI, finding it easier to tolerate than tamoxifen, as we have discussed. However, a significant minority of women is seriously bothered by the adverse effects, with arthralgias usually leading the pack of complaints.30,31
OBG Management: Do some women discontinue adjuvant endocrine therapy because of adverse effects?
Dr. Kaunitz: Regrettably, the answer is “Yes.” A recent study from Kaiser Permanente of northern California found that roughly 50% of women who are prescribed adjuvant endocrine therapy with tamoxifen or an AI discontinue the drug early.32
OBG Management: What can an ObGyn do to encourage compliance with and completion of AI therapy?
Dr. Kaunitz: First, it is critical that patients understand that AIs are lifesaving drugs. As a recent paper points out, early discontinuation or noncompliance with AI therapy is associated with higher mortality.33
Clinicians should also help breast cancer patients understand what common side effects to anticipate with these medications.
Finally, clinicians who understand the financial toll a breast cancer diagnosis and treatment can take are better positioned to help women overcome challenges that may interfere with long-term compliance with AI therapy.
OBG Management: Do you expect the use of AIs in breast cancer survivors to become more commonplace?
Dr. Kaunitz: Given how common breast cancer is, and given the new ASCO guidelines and the extensive literature upon which they are based, ObGyns will be seeing more women using AIs. Although we are not the physicians who prescribe AIs, we need to remain up to date on their benefits and side effects. This important class of drugs is positioned to improve outcomes for postmenopausal women with breast cancer.
- Do certain SSRIs reduce the benefits of tamoxifen in breast cancer survivors?
Examining the Evidence
Andrew M. Kaunitz, MD - Does the clinical breast exam boost the sensitivity of mammography?
Examining the Evidence
Jennifer Griffin, MD, and Mark Pearlman, MD - A guide to lotions and potions for treating vaginal atrophy
Danielle D. Marshall, MD, and Cheryl Iglesia, MD
We want to hear from you! Tell us what you think.
Think breast cancer survivors are unlikely to show up in your practice? You should think again.
At last official count, 2.5 million women in the United States had a history of breast cancer.1 Most of them are now free of malignancy, but others are still grappling with the disease in some form or fashion.2 All need continuing health care.
Roughly two thirds of women who have breast cancer have disease that is hormone-receptor–positive.2,3 Recently updated guidelines from the American Society of Clinical Oncology (ASCO) recommend that adjuvant therapy for postmenopausal women who have hormone-positive breast cancer include an aromatase inhibitor (AI) (see a summary of these guidelines on page 36). That makes it likely that a good number of breast cancer survivors who visit your practice are taking one of these medications: anastrozole (Arimidex), letrozole (Femara), or exemestane (Aromasin). These drugs are antiestrogens, given to postmenopausal women to reduce the likelihood of disease recurrence and progression.
The antiestrogenic properties of these drugs are what make them lifesavers. But the same qualities can create a range of health issues, from increased risk of osteoporosis and fracture to vasomotor and joint symptoms. And although ObGyns are not the physicians who prescribe these drugs, you may be the provider one of these women consults about their side effects and related issues.
To find out the latest on the management of women who are taking one of these agents, we inserted ourselves into the busy schedule of Andrew M. Kaunitz, MD, who agreed to address some fundamental—and some not so basic—questions about the drugs. In this extended Q&A, Dr. Kaunitz touches on mechanism of action, benefits versus risks, common side effects, compliance with therapy, and the ill effects of early discontinuation.

Aromatase inhibitors are better than tamoxifen at reducing the risk of breast cancer recurrence in postmenopausal women who have hormone-receptor–positive disease. But, they increase the risk of osteoporosis and fracture and often cause arthralgias and other complaints that ObGyn practitioners may be called upon to manage.
OBG Management: What is the overall aim of adjuvant endocrine therapy in the setting of breast cancer?
Dr. Kaunitz: Endocrine therapy—specifically, use of an AI—prevents the stimulation of breast cancer cells by endogenous estrogen. In other words, aromatase inhibitors suppress the growth of cancer cells that have estrogen receptors. These drugs also inhibit aromatase near any breast tumor and reduce estrogen levels in breast tissue.
OBG Management: What is the mechanism of action of adjuvant endocrine therapy?
Dr. Kaunitz: In postmenopausal women, androgens are converted to estrogens via the aromatase enzyme, which is present in adipose tissue and other sites. By blocking this enzyme, AIs reduce endogenous estrogen levels by as much as 95%.4
OBG Management: How does that differ from the mechanism of action of tamoxifen, another drug used in breast cancer patients?
Dr. Kaunitz: Tamoxifen is a selective estrogen receptor modulator (SERM). It blocks estrogen in breast tissue selectively, by competitively binding to estrogen receptors.5 However, tamoxifen has estrogenic effects in the uterus, bone, and liver, as well as other tissues.
The efficacy of AIs in preventing breast cancer recurrence in the first 2 years after breast cancer surgery is higher than that of tamoxifen. And unlike tamoxifen, the AIs do not increase the risk of venous thromboembolism or cause endometrial disease.
OBG Management: What effects do aromatase inhibitors have in premenopausal women?
Dr. Kaunitz: These agents are not recommended for use in premenopausal women because, in that population, the lion’s share of estrogen production takes place in the ovary rather than in adipose tissue and muscle. If you were to administer an AI to a premenopausal woman, the reduced hypothalamic and pituitary estrogen feedback could lead to ovarian stimulation—which could increase ovarian steroid production.
OBG Management: What about women who become amenorrheic as a result of chemotherapy or other cancer treatment? Do most oncologists assume that they are postmenopausal and prescribe an aromatase inhibitor?
Dr. Kaunitz: Clinicians should not assume that chemotherapy-induced amenorrhea signals permanent cessation of ovarian function. It is common for ovarian function to return in this setting. Accordingly, follicle-stimulating hormone (FSH) and estradiol levels should be assessed before an AI is considered as adjuvant therapy. Some investigators have suggested that the use of an AI in women who have chemotherapy-induced amenorrhea may actually increase the likelihood that ovarian function will return.6
OBG Management: Do all AIs produce the same effects?
Dr. Kaunitz: The AIs used in women with breast cancer are third-generation drugs. These AIs are classified as steroidal (type 1; exemestane) or nonsteroidal (type 2; anastrozole, letrozole). Exemestane, a steroid derived from androstenedione, inhibits the aromatase enzyme irreversibly. The nonsteroidal AIs are reversible.
Although all three AIs have numerous similarities, there are other distinctions between them in pharmacokinetics, mechanism of action, and toxicity—so they are not completely interchangeable.7 However, from our perspective as ObGyns caring for breast cancer survivors, we can assume that all three AIs will have similar effects on skeletal health and produce similar side effects in postmenopausal women.
- The American Society of Clinical Oncology recommends that adjuvant therapy for postmenopausal women who have hormone-positive breast cancer include an aromatase inhibitor (AI).
- AIs are not recommended for use in premenopausal women.
- By blocking the aromatase enzyme, AIs reduce endogenous estrogen levels by as much as 95%.4
- AIs are more effective than tamoxifen at preventing recurrence in the first 2 years after breast cancer surgery. Postmenopausal women taking an AI have a longer disease-free survival and time to recurrence than do women taking tamoxifen. They also have a lower incidence of contralateral breast cancer.
- The most prominent side effects of AI therapy include arthralgias and hot flushes, while the most serious health impact appears to be a decrease in bone mineral density (BMD). However, endometrial cancer, vaginal bleeding and discharge, cerebrovascular events, venous thromboembolic events, and hot flushes all are less common among women taking an AI than among those taking tamoxifen.
- The FDA strongly discourages the use of estrogen therapy—systemic or local—in women who are taking an AI. Accordingly, bisphospho-nate therapy is recommended as first-line treatment of low bone mineral density. Vaginal lubricants and moisturizers are the mainstay strategy for symptomatic genital atrophy. And gabapentin, selective serotonin reuptake inhibitors, and serotonin norepinephrine reuptake inhibitors are the mainstay of therapy for vasomotor flushes.
- Roughly 50% of women who are prescribed adjuvant endocrine therapy with tamoxifen or an AI discontinue the drug early.32 Early discontinuation is associated with an increase in mortality.33
How much do we know about these drugs?
OBG Management: How long does a woman typically take an AI?
Dr. Kaunitz: At present, in women treated for early-stage, hormone-positive breast cancer, the optimal duration of treatment is unknown. Most oncologists prescribe an AI for 5 years, the length of treatment in a prominent trial of the drugs.8
OBG Management: Is that duration likely to increase as more data come in?
Dr. Kaunitz: The optimal duration of adjuvant AI therapy will be determined by the findings of long-term clinical trials. The National Surgical Adjuvant Breast and Bowel Project B-42 trial may provide new insights into optimal duration of AI treatment after initial tamoxifen therapy.9
OBG Management: How thoroughly have AIs been studied in regard to their use in breast cancer survivors and women who have early-stage disease? How would you characterize the quantity and quality of data that we have so far?
Dr. Kaunitz: AIs have been extensively studied. The most important clinical trials of AIs in this setting, including the Anastrozole, Tamoxifen Alone or in Combination (ATAC) trial (over 6,000 participants, median follow-up of 100 months) and the Breast International Group (BIG) trial (almost 5,000 participants, median follow-up of 76 months) have been detailed in the recent ASCO report.10 These two large landmark trials, in particular, formed the basis for ASCO’s recommendations to routinely incorporate AIs into the therapy of postmenopausal women who have hormone-receptor–positive breast cancer.
OBG Management: In treating breast cancer, what other applications are AIs used for?
Dr. Kaunitz: AIs appear to be slightly more effective than tamoxifen in treating postmenopausal women who have metastatic breast cancer.11
They are approved as first-line therapy for breast cancer in:
- postmenopausal women who have hormone-receptor–positive disease
- postmenopausal women who have locally advanced disease when the hormone receptor is unknown
- postmenopausal women who have metastatic disease.
In addition, they are approved as second-line treatment of advanced breast cancer in postmenopausal women who have disease progression following tamoxifen therapy.12
How effective is AI therapy?
OBG Management: What do we know about the efficacy of these drugs?
Dr. Kaunitz: Most of the studies that have explored efficacy compare an AI with tamoxifen rather than with placebo. In the ATAC trial, after a median follow-up of 33 months, women who were taking anastrozole for early-stage breast cancer had longer disease-free survival and time to recurrence and a lower incidence of contralateral breast cancer than did women taking tamoxifen.8
After 4 years of follow-up in the ATAC trial, women taking anastrozole continued to have more favorable disease-free survival (86.9% vs 84.5% for anastrozole and tamoxifen, respectively; hazard ratio [HR], 0.86; 95% confidence interval [CI], 0.76–0.99; P =.03).13 They also had a more favorable time to recurrence than did women taking tamoxifen (HR, 0.83; 95% CI, 0.71–0.96; P =.015). And women taking anastrozole had a lower incidence of contralateral breast cancer, as well, although this different did not achieve statistical significance (HR, 0.62; 95% CI, 0.38–1.02; P =.062).13
In the BIG study, women taking letrozole had a 5-year disease-free survival estimate of 84.0%, compared with 81.4% for women taking tamoxifen.14 In addition, women taking letrozole were significantly less likely than those taking tamoxifen to experience an event that ended a period of disease-free survival (HR, 0.81; 95% CI, 0.70–0.93; P =.003), especially the event of distant recurrence (HR, 0.73; 95% CI, 0.60–0.88; P =.001).14
And a phase-3 study of exemestane versus tamoxifen in women who had metastatic breast cancer found that the AI produced a superior response rate (46% vs 31% for exemestane and tamoxifen, respectively; odds ratio [OR], 1.85; 95% CI, 1.21–2.82; P =.005). In addition, median progression-free survival was greater with exemestane (9.9 months; 95% CI, 8.7–11.8 months) than with tamoxifen (5.8 months; 95% CI, 5.3–8.1 months). However, there was no difference between arms in progression-free survival or overall survival.
Postmenopausal women who have hormone-receptor–positive breast cancer should consider taking an aromatase inhibitor (AI) to lengthen disease-free survival and lower the risk of recurrence. That’s one of the recommendations in updated guidelines issued earlier this year by the American Society of Clinical Oncology (ASCO). The guidelines suggest a duration of AI therapy of 5 years. In the event that a woman discontinues AI therapy before 5 years are up, she should consider using tamoxifen to bring the total duration of treatment to 5 years.
Other recommendations in the guidelines include:
- Women who have taken tamoxifen for 5 years stand to benefit from switching to an AI for as long as 5 additional years.
- When advising a woman about adjuvant therapy with an AI, clinicians should consider the potential adverse effects, which include osteoporosis, fracture, and arthralgias.
- The third-generation AIs on the market today have not been found to have clinically important differences between them. A woman who cannot tolerate a particular AI should consider switching to a different AI.
- Switching from an AI to tamoxifen (or vice versa) may be an appropriate option for patients who cannot tolerate a drug’s adverse effects. In the event of a switch to tamoxifen, the clinician should counsel the patient about its adverse effects, which include venous thromboembolism and endometrial polyps, hyperplasia, and cancer.
The full guidelines can be accessed at http://jco.ascopubs.org/content/early/2010/07/12/JCO.2009.26.3756.full.pdf.
—Andrew M. Kaunitz, MD
How well tolerated are AIs?
OBG Management: What adverse effects are associated with AIs?
Dr. Kaunitz: Although AIs, overall, are safe medications, their use is associated with a number of adverse events. The most prominent side effects include arthralgias and hot flushes, while the most serious health impact appears to be a decrease in bone mineral density (BMD).
However, the drugs are generally perceived as being easier to tolerate than tamoxifen. That’s because endometrial cancer, vaginal bleeding and discharge, cerebrovascular events, venous thromboembolic events, and hot flushes all are less common among women taking an AI than among those taking tamoxifen.8,13
For overweight women, who face an elevated baseline risk of thromboembolism, the availability of AIs represents a major advantage over tamoxifen. Similarly, AIs offer advantages over tamoxifen for women who have an intact uterus. In addition, postmenopausal women who are taking a selective serotonin reuptake inhibitor (SSRI) such as paroxetine should take an AI rather than tamoxifen, because the concomitant use of SSRIs attenuates the efficacy of tamoxifen.15
What can be done about the most prominent risks?
OBG Management: Let’s focus on what’s probably the best-known adverse effect of AIs—the heightened risk of osteoporosis and fracture. How significant is this effect?
Dr. Kaunitz: Because use of an AI is associated with a profound reduction in endogenous estrogen levels, it also decreases BMD and can lead to osteoporotic fractures. All major phase-3 trials of adjuvant use of AIs in women who have early breast cancer found an increased risk of fracture, with no significant differences between AIs.16
Fortunately, bisphosphonate therapy (oral or intravenous) has been found to reduce bone loss associated with AI therapy.17,18
Assessing baseline BMD is important as women initiate AI therapy. Although no consensus exists regarding follow-up BMD assessment in the setting of AI use, an interval of 2 years is prudent, with the follow-up study preferably performed at the same imaging center and by the same technician as the first. If baseline osteoporosis is observed at the lumbar spine or hip, bisphosphonate therapy is appropriate. If a woman taking an AI has low bone mass (osteopenia) but not osteoporosis, bisphosphonate therapy should be considered if any of the following risk factors are present:
- advanced age
- history of fracture
- glucocorticoid therapy
- parental history of hip fracture
- low body weight
- current smoking status
- excess alcohol consumption
- rheumatoid arthritis
- known risk factors for secondary osteoporosis.19
In breast cancer survivors initiating or continuing AI therapy, it is also appropriate to check a serum vitamin D level and ensure that intake of this nutrient is adequate.
Bisphosphonates may offer oncologic benefits, as well; preliminary evidence suggests that the drugs may prevent recurrence of the cancer and prolong survival.20
OBG Management: What can an ObGyn offer to a woman who complains of significant AI-related arthralgia?
Dr. Kaunitz: Bone and joint symptoms, including aches, pain, and stiffness that is bilateral and not associated with other evidence of rheumatologic disorders, are among the most common side effects of AI therapy. On the plus side of the equation, these symptoms are more likely to be mild to moderate than severe. On the negative side, no specific treatment has been found to be effective in relieving these symptoms, which usually resolve within 2 months or so after discontinuing AI therapy.10
OBG Management: Do AIs have a negative impact on cardiovascular health?
Dr. Kaunitz: Unlike tamoxifen, AIs do not increase the risk of thromboembolic disease. Although the use of an AI may modestly increase the risk of ischemic cardiovascular disease (and lipid changes), compared with tamoxifen, AIs do not appear to increase cardiovascular risk compared with placebo.21,22
OBG Management: Do the antiestrogenic effects of AIs have a significant impact on vaginal health and sexual desire?
Dr. Kaunitz: A review of published reports did not find that the use of AIs has a predictable impact on vaginal dryness or sexual desire.10 However, symptomatic genital atrophy is common in postmenopausal breast cancer survivors, whether or not they use adjuvant therapy.
Although the FDA considers the use of any estrogen (systemic or vaginal) following a diagnosis of breast cancer to be contraindicated, some breast cancer survivors who have symptomatic genital atrophy express an interest in the use of vaginal estrogen. Use of 25-μg estradiol tablets (Vagifem) is associated with a short-term increase in serum estradiol levels.23 This finding has reinforced caution among medical oncologists about the safety of vaginal estrogen in breast cancer survivors. (The 25-μg tablets are no longer marketed.) The lowest dosage of vaginal estrogen available for the treatment of genital atrophy is found in 10-μg estradiol tablets (Vagifem) and the estradiol (2-mg) 3-month vaginal ring (Estring). Nonetheless, in the absence of data, oncologists will likely continue to be concerned that even the lowest dosage of vaginal estrogen could attenuate the favorable impact of AIs on breast cancer. Accordingly, use of vaginal lubricants and moisturizers are the mainstay strategy for symptomatic genital atrophy.
OBG Management: What about the ubiquitous hot flush? Vasomotor symptoms may be more common in women who take tamoxifen, but women on AIs are also bothered by flushes. What are the alternatives to estrogen therapy?
Dr. Kaunitz: Both nonprescription and prescription alternatives are available. Nonprescription options include soy extract and red clover isoflavones, black cohosh, and Chinese herbs. However, none of these over-the-counter approaches has been found to be more effective than placebo in the treatment of menopausal hot flushes.24-26
As for prescription nonhormonal options, ObGyns should recognize that all such treatments are off-label and that none attain the efficacy of hormone therapy in the treatment of vasomotor symptoms. The best-studied and most effective medications include gabapentin, SSRIs (especially paroxetine), and serotonin-norepinephrine reuptake inhibitors (venlafaxine and desvenlafaxine).24,27
OBG Management: Is there any evidence that AIs impair cognitive function in postmenopausal women?
Dr. Kaunitz: Because estrogen is important for cognition, one might anticipate that the profound reduction in background estrogen associated with AI use would impair cognition. Fortunately, the evidence to date is reassuring. Substudies of the BIG trial and the Tamoxifen and Exemestane Adjuvant Multinational Trial indicate that, compared with tamoxifen (which is associated with declines in cognitive function in postmenopausal women), letrozole and exemestane do not diminish cognitive function.28,29
OBG Management: Overall, what is the typical impact of an AI on a woman’s quality of life?
Dr. Kaunitz: Most women do very well on an AI, finding it easier to tolerate than tamoxifen, as we have discussed. However, a significant minority of women is seriously bothered by the adverse effects, with arthralgias usually leading the pack of complaints.30,31
OBG Management: Do some women discontinue adjuvant endocrine therapy because of adverse effects?
Dr. Kaunitz: Regrettably, the answer is “Yes.” A recent study from Kaiser Permanente of northern California found that roughly 50% of women who are prescribed adjuvant endocrine therapy with tamoxifen or an AI discontinue the drug early.32
OBG Management: What can an ObGyn do to encourage compliance with and completion of AI therapy?
Dr. Kaunitz: First, it is critical that patients understand that AIs are lifesaving drugs. As a recent paper points out, early discontinuation or noncompliance with AI therapy is associated with higher mortality.33
Clinicians should also help breast cancer patients understand what common side effects to anticipate with these medications.
Finally, clinicians who understand the financial toll a breast cancer diagnosis and treatment can take are better positioned to help women overcome challenges that may interfere with long-term compliance with AI therapy.
OBG Management: Do you expect the use of AIs in breast cancer survivors to become more commonplace?
Dr. Kaunitz: Given how common breast cancer is, and given the new ASCO guidelines and the extensive literature upon which they are based, ObGyns will be seeing more women using AIs. Although we are not the physicians who prescribe AIs, we need to remain up to date on their benefits and side effects. This important class of drugs is positioned to improve outcomes for postmenopausal women with breast cancer.
- Do certain SSRIs reduce the benefits of tamoxifen in breast cancer survivors?
Examining the Evidence
Andrew M. Kaunitz, MD - Does the clinical breast exam boost the sensitivity of mammography?
Examining the Evidence
Jennifer Griffin, MD, and Mark Pearlman, MD - A guide to lotions and potions for treating vaginal atrophy
Danielle D. Marshall, MD, and Cheryl Iglesia, MD
We want to hear from you! Tell us what you think.
1. Horner MJ, Ries LAG, Krapcho M, et al. eds. SEER Cancer Statistics Review, 1975–2006. Bethesda, Md: National Cancer Institute; 2009.http://seer.cancer.gov/csr/1975_2006. Accessed August 18, 2010.
2. American Cancer Society. Breast cancer facts and figures, 2009–2010. Atlanta, Ga: American Cancer Society; 2010. http://www.cancer.org/Research/CancerFactsFigures/BreastCancerFactsFigures/breast-cancer-facts—figures-2009-2010. Accessed August 18, 2010.
3. Li CI, Daling JR, Malone KE. Incidence of invasive breast cancer by hormone receptor status from 1992 to 1998. J Clin Oncol. 2003;21(1):28-34.
4. Miller WR. Aromatase inhibitors: mechanism of action and role in the treatment of breast cancer. Semin Oncol. 2003;30(4 suppl 14):3-11.
5. Peng J, Sengupta S, Jordan VC. Potential of selective estrogen receptor modulators as treatments and preventives of breast cancer. Anticancer Agents Med Chem. 2009;9(5):481-499.
6. Smith IE, Dowsett M, Yap YS, et al. Adjuvant aromatase inhibitors for early breast cancer after chemotherapy-induced amenorrhea: caution and suggested guidelines. J Clin Oncol. 2006;24(16):2444-2447.
7. Nabholtz JM, Mouret-Reynier MA, Durando X, et al. Comparative review of anastrozole, letrozole and exemestane in the management of early breast cancer. Expert Opin Pharmacother. 2009;10(9):1435-1447.
8. Baum M, Budzar AU, Cuzick J, et al. ATAC Trialists’ Group. Anastrozole alone or in combination with tamoxifen versus tamoxifen alone for adjuvant treatment of postmenopausal women with early breast cancer: first results of the ATAC randomized trial. Lancet. 2002;359(9324):2131-2139.
9. Letrozole in treating postmenopausal women who have received hormone therapy for hormone receptor-positive breast cancer. National Cancer Institute Web site.http://www.cancer.gov/clinicaltrials/NSABP-B-42. Published August 16, 2010. Accessed August 18, 2010.
10. Burstein HJ, Prestrud AA, Seidenfeld J, et al. American Society of Clinical Oncology clinical practice guideline: update on adjuvant endocrine therapy for women with hormone-receptor–positive breast cancer. J Clin Oncol. 2010;28(23):3784-3796.
11. Mauri D, Pavlidis N, Polyzos NP, Ioanidis JP. Survival with aromatase inhibitors and inactivators versus standard hormonal therapy in advanced breast cancer: meta-analysis. J Natl Cancer Inst. 2006;98(18):1285-1291.
12. Arimidex [package insert]. AstraZeneca; 2009.
13. Baum M, Buzdar A, Cuzick J, et al. Anastrozole alone or in combination with tamoxifen versus tamoxifen alone for adjuvant treatment of postmenopausal women with early-stage breast cancer: results of the ATAC (Arimidex, Taxoxifen Alone or in Combination) trial efficacy and safety update analyses. Cancer. 2003;98(9):1802-1810.
14. Thurlimann B, Keshaviah A, Coates AS, et al. Breast International Group (BIG) 1-98 Collaborative Group. A comparison of letrozole and tamoxifen in postmenopausal women with early breast cancer. N Engl J Med. 2005;353(26):2747-2757.
15. Kelly CM, Juurlink DM, Gomes T, et al. Selective serotonin reuptake inhibitors and breast cancer mortality in women receiving tamoxifen: a population-based cohort study. 2010;340:c693.-doi: 10.1136/bmj.c693.
16. Geisler J, Lonning PE. Impact of aromatase inhibitors on bone health in breast cancer patients. J Steroid Biochem Mol Biol. 2010;118(4–5):294-299.
17. Van Poznak C, Hannon RA, Mackey JR, et al. Prevention of aromatase inhibitor-induced bone loss using risedronate: the SABRE trial. J Clin Oncol. 2010;28(6):967-975.
18. Brufsky AM, Bosserman LD, Caradonna RR, et al. Zoledronic acid effectively prevents aromatase-inhibitor associated bone loss in postmenopausal women with early breast cancer receiving adjuvant letrozole. Z-FAST study 36-month follow-up results. Clin Breast Cancer. 2009;9(2):77-85.
19. Kanis JA, Borgstrom F, De Laet C, et al. Assessment of fracture risk. Osteoporosis Int. 2005;16(6):581-589.
20. Reeder JG, Brufsky AM. The role of bisphosphonates in the adjuvant setting for breast cancer. Oncology. 2010;24(6):462-467,475.
21. Nabholtz JM. Long-term safety of aromatase inhibitors in the treatment of breast cancer. Ther Clin Risk Manag. 2008;4(1):189-204.
22. Cuppone F, Bria E, Verma S, et al. Do adjuvant aromatase inhibitors increase the cardiovascular risk in postmenopausal women with early breast cancer? Meta-analysis of randomized trials. Cancer. 2008;112(2):260-267.
23. Kendall A, Dowsett M, Folkerd E, Smith I. Caution: Vaginal estradiol appears to be contraindicated in postmenopausal women on adjuvant aromatase inhibitors. Ann Oncol. 2006;17(4):584-587.
24. Nelson HD. Menopause. Lancet. 2008;371(9614):760-770.
25. Geller SE, Shulman LP, van Breemen RB, et al. Safety and efficacy of black cohosh and red clover for the management of vasomotor symptoms: a randomized controlled trial. Menopause. 2009;16(6):1156-1166.
26. Kaunitz AM. Effective herbal treatment of vasomotor symptoms—are we any closer? Menopause. 2009;16(3):428-429.
27. Archer DF, Dupont CM, Constantine GD, Pickar JH, Olivier S. Study 319 Investigators. Desvenlafaxine for the treatment of vasomotor symptoms associated with menopause: a double-blind, randomized, placebo-controlled trial of efficacy and safety. Am J Obstet Gynecol. 2009;200(3):238.e1-e10.
28. Phillips KA, Ribi K, Sun Z, et al. Cognitive function in postmenopausal women receiving adjuvant letrozole or tamoxifen for breast cancer in the BIG 1-98 randomized trial [published online ahead of print April 10, 2010]. Breast. doi:10.1016/j.breast.2010.03.025.
29. Schilder CM, Seynaeve C, Beex LV, et al. Effects of tamoxifen and exemestane on cognitive functioning of postmenopausal patients with breast cancer: results from the neuropsychological side study of the Tamoxifen and Exemestane Adjuvant Multinational Trial. J Clin Oncol. 2010;28(8):1294-1300.
30. Din OS, Dodwell D, Wakefield RJ, Coleman RE. Aromatase inhibitor-induced arthralgia in early breast cancer: what do we know and how can we find out more? Breast Cancer Res Treat. 2010;120(3):525-538.
31. Burstein HJ. Aromatase inhibitor-associated arthralgia syndrome. Breast. 2007;16(3):223-234.
32. Hershman DL, Kushi LH, Shao T, et al. Early discontinuation and nonadherence to adjuvant hormonal therapy in a cohort of 8,769 early-stage breast cancer patients [published online ahead of print June 28, 2010]. J Clin Oncol. doi: 10.1200/JCO.2009.25.9655.
33. Hershman DL, Shao T, Kushi LH, et al. Early discontinuation and nonadherence to adjuvant hormonal therapy are associated with increased mortality in women with breast cancer [published online ahead of print August 28, 2010]. Breast Cancer Res Treat. doi: 10.1007/ s10549-010-1132-4.
1. Horner MJ, Ries LAG, Krapcho M, et al. eds. SEER Cancer Statistics Review, 1975–2006. Bethesda, Md: National Cancer Institute; 2009.http://seer.cancer.gov/csr/1975_2006. Accessed August 18, 2010.
2. American Cancer Society. Breast cancer facts and figures, 2009–2010. Atlanta, Ga: American Cancer Society; 2010. http://www.cancer.org/Research/CancerFactsFigures/BreastCancerFactsFigures/breast-cancer-facts—figures-2009-2010. Accessed August 18, 2010.
3. Li CI, Daling JR, Malone KE. Incidence of invasive breast cancer by hormone receptor status from 1992 to 1998. J Clin Oncol. 2003;21(1):28-34.
4. Miller WR. Aromatase inhibitors: mechanism of action and role in the treatment of breast cancer. Semin Oncol. 2003;30(4 suppl 14):3-11.
5. Peng J, Sengupta S, Jordan VC. Potential of selective estrogen receptor modulators as treatments and preventives of breast cancer. Anticancer Agents Med Chem. 2009;9(5):481-499.
6. Smith IE, Dowsett M, Yap YS, et al. Adjuvant aromatase inhibitors for early breast cancer after chemotherapy-induced amenorrhea: caution and suggested guidelines. J Clin Oncol. 2006;24(16):2444-2447.
7. Nabholtz JM, Mouret-Reynier MA, Durando X, et al. Comparative review of anastrozole, letrozole and exemestane in the management of early breast cancer. Expert Opin Pharmacother. 2009;10(9):1435-1447.
8. Baum M, Budzar AU, Cuzick J, et al. ATAC Trialists’ Group. Anastrozole alone or in combination with tamoxifen versus tamoxifen alone for adjuvant treatment of postmenopausal women with early breast cancer: first results of the ATAC randomized trial. Lancet. 2002;359(9324):2131-2139.
9. Letrozole in treating postmenopausal women who have received hormone therapy for hormone receptor-positive breast cancer. National Cancer Institute Web site.http://www.cancer.gov/clinicaltrials/NSABP-B-42. Published August 16, 2010. Accessed August 18, 2010.
10. Burstein HJ, Prestrud AA, Seidenfeld J, et al. American Society of Clinical Oncology clinical practice guideline: update on adjuvant endocrine therapy for women with hormone-receptor–positive breast cancer. J Clin Oncol. 2010;28(23):3784-3796.
11. Mauri D, Pavlidis N, Polyzos NP, Ioanidis JP. Survival with aromatase inhibitors and inactivators versus standard hormonal therapy in advanced breast cancer: meta-analysis. J Natl Cancer Inst. 2006;98(18):1285-1291.
12. Arimidex [package insert]. AstraZeneca; 2009.
13. Baum M, Buzdar A, Cuzick J, et al. Anastrozole alone or in combination with tamoxifen versus tamoxifen alone for adjuvant treatment of postmenopausal women with early-stage breast cancer: results of the ATAC (Arimidex, Taxoxifen Alone or in Combination) trial efficacy and safety update analyses. Cancer. 2003;98(9):1802-1810.
14. Thurlimann B, Keshaviah A, Coates AS, et al. Breast International Group (BIG) 1-98 Collaborative Group. A comparison of letrozole and tamoxifen in postmenopausal women with early breast cancer. N Engl J Med. 2005;353(26):2747-2757.
15. Kelly CM, Juurlink DM, Gomes T, et al. Selective serotonin reuptake inhibitors and breast cancer mortality in women receiving tamoxifen: a population-based cohort study. 2010;340:c693.-doi: 10.1136/bmj.c693.
16. Geisler J, Lonning PE. Impact of aromatase inhibitors on bone health in breast cancer patients. J Steroid Biochem Mol Biol. 2010;118(4–5):294-299.
17. Van Poznak C, Hannon RA, Mackey JR, et al. Prevention of aromatase inhibitor-induced bone loss using risedronate: the SABRE trial. J Clin Oncol. 2010;28(6):967-975.
18. Brufsky AM, Bosserman LD, Caradonna RR, et al. Zoledronic acid effectively prevents aromatase-inhibitor associated bone loss in postmenopausal women with early breast cancer receiving adjuvant letrozole. Z-FAST study 36-month follow-up results. Clin Breast Cancer. 2009;9(2):77-85.
19. Kanis JA, Borgstrom F, De Laet C, et al. Assessment of fracture risk. Osteoporosis Int. 2005;16(6):581-589.
20. Reeder JG, Brufsky AM. The role of bisphosphonates in the adjuvant setting for breast cancer. Oncology. 2010;24(6):462-467,475.
21. Nabholtz JM. Long-term safety of aromatase inhibitors in the treatment of breast cancer. Ther Clin Risk Manag. 2008;4(1):189-204.
22. Cuppone F, Bria E, Verma S, et al. Do adjuvant aromatase inhibitors increase the cardiovascular risk in postmenopausal women with early breast cancer? Meta-analysis of randomized trials. Cancer. 2008;112(2):260-267.
23. Kendall A, Dowsett M, Folkerd E, Smith I. Caution: Vaginal estradiol appears to be contraindicated in postmenopausal women on adjuvant aromatase inhibitors. Ann Oncol. 2006;17(4):584-587.
24. Nelson HD. Menopause. Lancet. 2008;371(9614):760-770.
25. Geller SE, Shulman LP, van Breemen RB, et al. Safety and efficacy of black cohosh and red clover for the management of vasomotor symptoms: a randomized controlled trial. Menopause. 2009;16(6):1156-1166.
26. Kaunitz AM. Effective herbal treatment of vasomotor symptoms—are we any closer? Menopause. 2009;16(3):428-429.
27. Archer DF, Dupont CM, Constantine GD, Pickar JH, Olivier S. Study 319 Investigators. Desvenlafaxine for the treatment of vasomotor symptoms associated with menopause: a double-blind, randomized, placebo-controlled trial of efficacy and safety. Am J Obstet Gynecol. 2009;200(3):238.e1-e10.
28. Phillips KA, Ribi K, Sun Z, et al. Cognitive function in postmenopausal women receiving adjuvant letrozole or tamoxifen for breast cancer in the BIG 1-98 randomized trial [published online ahead of print April 10, 2010]. Breast. doi:10.1016/j.breast.2010.03.025.
29. Schilder CM, Seynaeve C, Beex LV, et al. Effects of tamoxifen and exemestane on cognitive functioning of postmenopausal patients with breast cancer: results from the neuropsychological side study of the Tamoxifen and Exemestane Adjuvant Multinational Trial. J Clin Oncol. 2010;28(8):1294-1300.
30. Din OS, Dodwell D, Wakefield RJ, Coleman RE. Aromatase inhibitor-induced arthralgia in early breast cancer: what do we know and how can we find out more? Breast Cancer Res Treat. 2010;120(3):525-538.
31. Burstein HJ. Aromatase inhibitor-associated arthralgia syndrome. Breast. 2007;16(3):223-234.
32. Hershman DL, Kushi LH, Shao T, et al. Early discontinuation and nonadherence to adjuvant hormonal therapy in a cohort of 8,769 early-stage breast cancer patients [published online ahead of print June 28, 2010]. J Clin Oncol. doi: 10.1200/JCO.2009.25.9655.
33. Hershman DL, Shao T, Kushi LH, et al. Early discontinuation and nonadherence to adjuvant hormonal therapy are associated with increased mortality in women with breast cancer [published online ahead of print August 28, 2010]. Breast Cancer Res Treat. doi: 10.1007/ s10549-010-1132-4.
Breast cancer prevention and Tx: An evidence-based guide
• Offer screening magnetic resonance imaging (MRI) to patients with a known BRCA 1 or 2 mutation, a strong family history of breast cancer, or a lifetime risk of breast cancer >20% to 25%. B
• For early-stage breast cancer, lumpectomy and sentinel node mapping with excision is the preferred method for staging. A
• Monitor patients receiving tamoxifen for signs and symptoms of venous thromboembolism, cataracts, and uterine malignancy, and patients on aromatase inhibitors for the development of osteoporosis. A
Strength of recommendation (SOR)
A Good-quality patient-oriented evidence
B Inconsistent or limited-quality patient-oriented evidence
C Consensus, usual practice, opinion, disease-oriented evidence, case series
Late last year, the US Preventive Services Task Force (USPSTF) sparked a nationwide controversy when it announced that it was recommending against routine screening mammography for women younger than age 50.1 Indeed, that’s a recommendation that many other organizations, including the American Cancer Society (ACS),2 the American College of Obstetricians and Gynecologists (ACOG),3 and the National Comprehensive Cancer Network (NCCN),4 disagree with. But the age at which women should begin routine mammography isn’t the only controversial question. Experts disagree on the benefits of breast self-examination, the optimal frequency of clinical breast exams, and the use of digital mammography—among other issues. This evidence-based review can help you cut through the confusion.
CASE Carrie, a 39-year-old woman who has never been pregnant, comes in for an annual Pap smear and gynecologic exam. She has a negative past medical history, but a positive family history for breast cancer—both her mother and 1 of her sisters had the disease. How would you assess Carrie’s risk of breast cancer, and what preventive measures would you recommend?
Use this predictive model to pinpoint your patient’s risk
When making decisions regarding primary prevention of and screening for breast cancer, an accurate assessment of risk is critical. Many predictive models have been developed with that in mind. The most widely studied, the Gail model, incorporates a number of important risk factors (TABLE 1), including age; race; family history; reproductive factors such as age of menarche, menopause, and first childbirth; and previous history of breast biopsy and atypical findings, to calculate a woman’s 5-year risk.5
A risk calculator (the Breast Cancer Risk Assessment Tool) based on the Gail model is available on the National Cancer Institute’s Web site, at http://www.cancer.gov/bcrisktool. Generally, a score ≥1.66%,5 which indicates that a patient has at least a 1.66% chance of developing breast cancer over the next 5 years, is considered high risk.6,7
CASE Carrie’s 2 first-degree relatives affected by breast cancer and her nulliparous status place her at increased risk. Further questioning reveals a particularly strong family history, as both relatives were diagnosed before the age of 50 (her mom at 45 years of age and her sister, at 39). Carrie’s 5-year risk is 1.8%.
TABLE 1
Risk factors for breast cancer5,29
|
| *African American and Caucasian women are at higher risk compared with Asian, Hispanic, and Native American women. |
| †1 drink/day results in minimal increase in risk; 2-5 drinks/day result in 1.5 increased risk compared with nondrinkers. |
| HRT, hormone replacement therapy; LCIS, lobular carcinoma in situ. |
All women can benefit from these preventive measures
As primary care physicians, we have a responsibility to stress lifestyle modification as the mainstay of breast cancer prevention. Whether or not a woman is at high risk, advise her that maintaining a normal weight, exercising vigorously, limiting alcohol consumption, and breastfeeding are evidence-based methods of primary prevention. Diets low in fat and high in fiber may be associated with a lower risk of invasive breast cancer, but there is no conclusive evidence to support specific dietary interventions to reduce the risk.8-11 Nor has a link between active or passive smoking, antioxidants, or fruit and vegetable intake been firmly established.12
There is a clear association between prolonged estrogen exposure and breast cancer, however. Many reproductive factors, such as early menarche, late menopause, later age at time of first full-term pregnancy, and nulliparity, increase a woman’s exposure to endogenous estrogen—and her risk of developing breast cancer.12,13
Exposure to exogenous estrogen is also linked to the development of breast cancer. In 2002, the Women’s Health Initiative (WHI) was stopped early after a report was released stating that the risks of hormone replacement therapy (HRT)—a higher incidence of cardiovascular events, stroke, and venous thromboembolism, as well as breast cancer—outweighed the benefits.14 Subsequent analyses have found a relationship between the declining incidence of breast cancer and the marked decrease in HRT use prompted by the WHI report. While causality has not been firmly established, multiple studies strongly suggest it.15,16
The association between oral contraceptives (OCs) and breast cancer is more controversial. Some studies have found an increased breast cancer risk among OC users, but both the relative risk and absolute risk were found to be very small and to dissipate 10 years after stopping OC use. More recent studies with newer formulations containing lower doses of estrogen have failed to show an increased risk.8
Breast cancer screening: The parameters have changed
Various organizations have published guidelines for breast cancer screening (TABLE 2), and all are somewhat different. Here’s what you need to know.
Breast self-examination (BSE), which women were previously advised to perform monthly, has not been shown to improve mortality in any age group, and is no longer routinely recommended.17 While both the USPSTF and the Canadian Task Force on Preventive Health Care recommend against teaching women BSE, the ACS, ACOG, and NCCN encourage self-examination—particularly among women older than 40 years.1-4,17,18
Clinical breast examination has an average sensitivity of 50% and detects approximately 5% of mammographically occult cancers.19 It is still not clear whether clinical breast exams save lives, however—a finding that is reflected in the USPSTF’s “I” (insufficient evidence to assess the benefits and harms) recommendation.1 Other consensus guidelines still recommend clinical breast examination, albeit at varying frequencies.
Screening mammography decreases mortality rates by anywhere from 28% to 65%, depending on the statistical model used.20 The benefit is greatest in women between the ages of 50 and 69 years, however, and most groups agree that mammography every 1 to 2 years is advisable for this age group. (There is limited data on the value of mammography for women 70 years of age and older, and no consensus on the age at which to stop screening.1,21) But because the mortality benefit from screening mammography is lower for women aged 40 to 49, guidelines for this age group are more controversial.
Mammography’s sensitivity is affected by a variety of factors, including age and menopausal status, prior breast surgery or radiation, breast density, and the experience of the radiologist. Women in their 40s have denser breast tissue than older women, making mammography less sensitive for this age group. Because of that, and because the overall incidence of breast cancer is lower for women younger than 50, some argue that screening mammography for women between the ages of 40 and 49 years leads to unacceptably high false-positive rates (9.8% annually22) and that the harm associated with mammography may outweigh the benefit. Others counter that tumors in younger women tend to be more aggressive and faster growing, making early detection even more critical than for older women.
What should you advise women in this age group? You might point out that the USPSTF recommends against routine screening, but indicates that the decision to begin (or defer) routine mammography before age 50 should be individualized, based on the needs and values of each patient.1
Digital mammography. A recent study of more than 43,000 women demonstrated that digital mammography is more accurate than film—but only for certain groups: These include women <50 years of age, women with dense breasts, and pre- and perimenopausal women. 23 Because it is still not clear whether the increased accuracy will translate into a mortality benefit, more research is needed before digital mammography is widely adopted. The USPSTF maintains that there is insufficient evidence to assess the benefits and harms of using either digital mammography or magnetic resonance imaging (MRI) rather than film mammography to screen for breast cancer.1
MRI. In 2007, the ACS published guidelines on the use of MRI as an adjunct to mammography for breast cancer screening in high-risk women.24 According to ACS guidelines, screening MRI should be offered to patients with a known BRCA 1 or 2 mutation (5%-10% of all breast cancers are associated with a mutation in the BRCA 1 or BRCA 2 gene, which is transmitted in an autosomal dominant pattern6). It also should be offered to those with a strong family history, or a lifetime risk of developing breast cancer that is >20% to 25%. And finally, MRI should be offered to women who had chest wall radiation when they were between the ages of 10 and 30 years—another significant risk factor for breast cancer—and those with other genetic syndromes that increase their lifetime risk of breast cancer.24
Evidence is insufficient for or against MRI screening for women with a personal history of breast cancer, atypical hyperplasia, or lobular carcinoma in situ, however, and neither breast ultrasound (which is generally used diagnostically, not for screening purposes) nor MRI has been shown to be helpful as a screening tool in women with <15% lifetime risk of developing breast cancer.24,25
TABLE 2
Guidelines for breast cancer screening for women with average risk
| Organization | Age (years) | Breast self-exam | Clinical breast exam | Mammography |
|---|---|---|---|---|
| American Cancer Society2 | 20-40 | Optional | Every 3 y | NA |
| >40 | Encourages | Annually | Annually | |
| American College of Obstetricians and Gynecologists3 | 40-49 | Encourages | Annually | 1-2 y |
| 50-69 | Encourages | Annually | Annually | |
| Canadian Task force on Preventive Health Care18 | 40-49 | Recommends against teaching | Insufficient evidence | Insufficient evidence |
| 50-69 | Recommends against teaching | 1-2 y | 1-2 y | |
| National Comprehensive Cancer Network4 | 20-40 | Encourages | 1-3 y | NA |
| >40 | Encourages | Annually | Annually | |
| US Preventive Services Task Force1 | 40-49 | Recommends against teaching | Insufficient evidence | Not routinely recommended |
| 50-74 | Recommends against teaching | Insufficient evidence | Every 2 y | |
| NA, not addressed. | ||||
When to consider chemoprevention
For women like Carrie, who are at high risk of developing breast cancer, selective estrogen receptor modulator (SERM) therapy and surgical interventions may be options to consider. The Breast Cancer Prevention Trial demonstrated the efficacy of tamoxifen as a preventive agent. This landmark trial showed that for high-risk women older than 35, 5 years of tamoxifen therapy can reduce the incidence of invasive breast cancer by nearly 50%.26
Women with the BRCA 1 or 2 mutation—all of whom should be offered genetic counseling—were included in the study. Tamoxifen reduced the incidence of breast cancer in BRCA 2 carriers by 62%, the researchers found, but did not reduce risk in carriers of the BRCA 1 gene. This is likely due to the high prevalence of estrogen receptor-negative breast cancers among BRCA 1 carriers.26
More recently, the Study of Tamoxifen and Raloxifene (STAR) trial compared the efficacy of tamoxifen and raloxifene, a second-generation SERM, in high-risk postmenopausal women ages 35 and older. The drugs were found to be equally effective in reducing the risk of invasive breast cancer, but raloxifene had a better side effect profile, with a lower incidence of thromboembolism and cataracts. 27
What the guidelines call for. In 2003, the USPSTF recommended that clinicians discuss chemoprevention with women at high risk for breast cancer and low risk for adverse effects of SERMs.28
The most recent update to the NCCN breast cancer risk reduction guidelines recommends that clinicians offer tamoxifen to premenopausal women with a 5-year projected breast cancer risk ≥1.7% and offer tamoxifen or raloxifene to high-risk postmenopausal women.29 It is worth noting, however, that SERMs can have significant adverse effects, including venous thromboembolism, stroke, cataracts, uterine malignancy, and hot flashes, while lifestyle modifications and the avoidance of HRT have few, if any, negative effects.
CASE After consultation with a genetic counselor, Carrie underwent testing for both the BRCA 1 and BRCA 2 mutations. She tested negative for both. She declined chemoprevention and prophylactic surgery, opting for enhanced screening with yearly mammography and MRI and lifestyle modification instead.
When a mass is found
For women ages 30 or older with palpable masses or solid masses ≥2 cm found on imaging, core needle biopsy is recommended.30,31 Biopsy is indicated for women younger than 30 as well, if the mass is >2 cm or imaging is suspicious. In general, a needle biopsy read as benign is considered adequate for diagnostic purposes only if the lesion appeared benign on imaging.
For lesions shown to be cystic on imaging, recommendations for follow-up or additional testing are based on the characteristics of the cyst. For simple cysts, 2- to 4-month follow up for stability, followed by routine screening, is adequate.21 Additional evaluation of complex cysts is indicated, including aspiration for complicated cysts and biopsy for complex cysts. After aspiration, surgical excision of bloody aspirates or persistent masses is recommended.30,31
Staging using the TNM system
The TNM (tumor, node, metastases) classification system is used for the staging of breast cancer:
- T refers to the tumor type, size, and extent of local involvement
- N describes regional lymph node involvement
- M refers to distant metastases.
The TNM classifications are also grouped by stage (I through IV).,
Lumpectomy and sentinel node mapping with excision is the preferred method for staging of early-stage breast cancer without palpable lymphadenopathy—provided that the surgical team has documented experience with sentinel node biopsy.32 Sentinel node biopsy is preferred because of its safety, low (<10%) false negative rate, and decreased morbidity compared with full axillary dissection, although dissection is recommended for patients with more advanced cancer or a positive sentinel node.32 The comparative effects of sentinel node biopsy vs axillary node dissection on tumor recurrence and patient survival are not known.33
Testing for tumor markers such as estrogen and progesterone receptors and human epidermal growth factor receptor 2 (HER2) expression status in biopsy-proven breast cancer is now the standard of care. Seventy percent of breast cancers are estrogen receptor-positive, with increasing frequency associated with older age.34 Estrogen/progesterone receptor positivity is associated with a more favorable outcome, and multiple hormonal therapies can be aimed at these receptors.34 While HER2 overexpression—which occurs in 15% to 30% of newly diagnosed breast cancers35—is associated with more aggressive tumors, women with this type of tumor cell can benefit from trastuzumab, an anti-HER2 drug.36
Key factors that affect prognosis
Important factors affecting prognosis and treatment of localized breast cancer are tumor size, age and menopausal status, tumor expression of hormone receptors and/ or the HER2 protein, as well as the status of the draining axillary nodes. Factors that predict a greater chance of recurrence include the spread of disease to axillary nodes, larger tumor size, invasive histology, inflammatory pathology, lack of estrogen/progesterone receptors, and age <50 years or premenopausal status.
Treatment options include surgical resection, radiation, and systemic adjuvant therapy in the form of chemotherapy, endocrine therapy, or anti-HER2 monoclonal antibodies.37 (For more on treatment, see “Surgery, radiation, and systemic therapy: Making the most of what’s in our arsenal” at jfponline.com.)
Don’t overlook quality-of-life issues
Follow-up of breast cancer patients should go beyond treatment and work-up for recurrence and metastatic disease to focus on health and lifestyle issues, such as stress reduction, mood, smoking cessation, diet and exercise, treatment of hot flashes, sexual dysfunction, and bone health. A recent study found both reduced recurrence and increased survival in women receiving psychological interventions to improve quality-of-life measures after an 11-year follow-up.38
Refer women to targeted Web sites such as the National Breast Cancer Awareness Month organization (http://www.nbcam.org/), the National Breast Cancer Foundation (http://community.nationalbreastcancer.org/), and the Susan G. Komen Breast Cancer Foundation (http://ww5.komen.org/). Offer treatment for bothersome symptoms. Hot flashes and depression, for example, often related to endocrine therapy, can be treated with selective serotonin reuptake inhibitors (SSRIs). That said, some SSRIs decrease the active metabolite of tamoxifen by inhibiting CYP2D6 enzyme and must, therefore, be used with caution. However, venlafaxine and citalopram are less likely to alter tamoxifen metabolism than other SSRIs.39
CASE When Carrie was 47, she had an abnormal MRI of the left breast. Core needle biopsy and pathology of the lesion revealed an estrogen and progesterone receptor-positive tumor that was negative for HER2 overexpression. She underwent lumpectomy, which revealed a 1.5 cm tumor, followed by a negative sentinel node biopsy, and was diagnosed with stage I (T1N0M0) breast cancer. Carrie had radiation after surgery; she did not require chemotherapy, but was told to take tamoxifen for 5 years. This adjuvant endocrine therapy led to hot flashes and depression, both of which were successfully treated with venlafaxine. Carrie is currently cancer-free and participates in a breast cancer survivor program that includes regular visits with her primary physician and her oncologist.
CORRESPONDENCE Denise Sur, MD, 1920 Colorado Avenue, Santa Monica, CA 90404; [email protected]
Surgery, radiation, and systemic therapy: Making the most of what’s in our arsenal
Breast cancer surgery has changed dramatically over the years. Multiple studies have shown that breast-conserving therapy (lumpectomy followed by radiation) for carefully selected women is comparable to mastectomy for local recurrence and survival. While there has been much interest in determining whether a subset of patients could forego radiation after lumpectomy, a meta-analysis by the Early Breast Cancer Trialists Collaborative Group demonstrated that radiation after lumpectomy provides an absolute local recurrence risk reduction of 19%, and a 5.4% absolute reduction in 15-year breast cancer mortality rates compared with lumpectomy without radiation.1 Thus, radiation after lumpectomy remains the standard of care for all women undergoing breast-conserving therapy, regardless of tumor characteristics.
In certain women with a high risk of recurrence (≥4 positive nodes), radiation is also recommended after mastectomy. Women undergoing mastectomy have numerous options for immediate or delayed breast reconstruction. Consultation with a multidisciplinary team, including a plastic surgeon, prior to any surgical intervention is advised.2
Multiple systemic chemotherapy regimens have been shown to be beneficial in carefully selected patients with breast cancer. Systematic reviews have demonstrated that an anthracycline-based regimen can decrease annual breast cancer mortality by 38% in women <50 years old and by 20% in women ages 50 to 69 years.1 in more recent randomized controlled trials, the addition of taxanes to anthracycline-based regimens has produced promising results.3
Numerous hormonal therapies benefit women with estrogen or progesterone receptor-positive breast cancer. Tamoxifen blocks the activity of estrogen on receptors located in breast cancer tissue, for example; aromatase inhibitors block the conversion of androgens to estrogen; and gonadotropin-releasing hormone (GnRH) analogs such as leuprolide and goserelin suppress ovarian production of estrogen.
For postmenopausal women, options include an aromatase inhibitor alone or tamoxifen followed by an aromatase inhibitor.
In premenopausal women, aromatase inhibitors are not very effective, as decreasing peripheral estrogen stimulates the ovaries to produce more estrogen. Thus, for these patients, adjuvant endocrine therapy consists of tamoxifen, with ovarian ablation (via surgery or radiation) or ovarian suppression with a GnRH analog. If the patient goes through menopause as a result of this therapy, she may benefit from aromatase inhibitors at that time.4,5
Women with breast cancer that overexpresses the HER2 gene benefit from adjuvant treatment with trastuzumab, an anti-HER2 antibody.6 While current guidelines advise treatment for 1 year, multiple studies are evaluating dosing schedules and optimal duration of treatment. for now, patients should be monitored for signs of cardiotoxicity at baseline and every 3 months thereafter until completion of therapy.4
References
1. Early Breast Caner Trialists’ Collaborative Group (EBCTCG). Effects of chemotherapy and hormonal therapy for early breast cancer on recurrence and 15-year survival: an overview of the randomised trials. Lancet. 2005;265:1687-1717.
2. Codeiro P. Breast reconstruction after surgery for breast cancer. N Engl J Med. 2008;359:1590-1601.
3. DeLaurentiis M, Cancello G, D’Agostino D, et al. Taxane-based combinations as adjuvant chemotherapy of early breast cancer: a meta-analysis of randomized trials. J Clin Oncol. 2008;26:44-53.
4. National Comprehensive Cancer Network. Breast cancer risk reduction clinical practice guidelines in oncology. J Natl Compr Canc Netw. 2003;1:280-296.
5. Baum M, Budzar AU, Cuzick J, et al. Anastrozole alone or in combination with tamoxifen versus tamoxifen alone for adjuvant treatment of postmenopausal women with early breast cancer: first results of the ATAC randomised trial. Lancet. 2002;359:2131-2139.
6. Piccart-Gebhart MJ, Procter M, Leyland-Jones B, et al. Herceptin adjuvant (HERA) Trial Study Team. Trastuzumab after adjuvant chemotherapy in HER2-positive breast cancer. N Engl J Med. 2005;353:1659-1672.
1. US Preventive Services Task Force Screening for breast cancer. Available at: http://www.uspreventiveservicestaskforce.org/uspstf/uspsbrca.htm. Accessed September 2, 2010.
2. American Cancer Society. American Cancer Society guidelines for the early detection of cancer. Available at: http://www.cancer.org/Healthy/FindCancerEarly/CancerScreeningGuidelines/american-cancer-society-guidelines-for-the-early-detection-of-cancer. Accessed September 2, 2010.
3. American College of Obstetricians and Gynecologists. Response of the American College of Obstetricians and Gynecologists to the new breast cancer screening recommendations from the US Preventive Services Task Force. Available at: http://www.acog.org/from_home/misc/uspstfresponse.cfm. Accessed September 2, 2010.
4. National Comprehensive Cancer Network. Breast cancer screening and diagnosis. Clinical Practice Guidelines in Oncology-V.1.2010. Fort Washington, Pa: National Comprehensive Cancer Network: November 3, 2009. Available at: http://www.nccn.org/professionals/physician_gls/f_guidelines.asp. Accessed September 17 , 2010.
5. National Cancer Institute. Breast cancer risk assessment tool. Available at: http://www.cancer.gov/bcrisktool/. Accessed September 2, 2010
6. Robson M, Offit K. Clinical practice. Management of an inherited predisposition to breast cancer. N Engl J Med. 2007;357:154-162.
7. Fisher B, Costantino JP, Wickerham DL, et al. Tamoxifen for prevention of breast cancer: report of the National Surgical Adjuvant Breast and Bowel Project P-1 Study. J Natl Cancer Inst. 1998;90:1371-1388.
8. Cummings SR, Tice JA, Bauer S, et al. Prevention of breast cancer in postmenopausal women: approaches to estimating and reducing risk. J Natl Cancer Inst. 2009;18:101,-384-398.
9. Prentice RL, Caan B, Chlebowski RT, et al. Low-fat dietary pattern and risk of invasive breast cancer: the Women’s Health Initiative Randomized Controlled Dietary Modification Trial. JAMA. 2006;295:629-642.
10. Pierce JP, Natarajan L, Caan BJ, et al. Influence of a diet very high in vegetables, fruit, and fiber and low in fat on prognosis following treatment for breast cancer: the Women’s Healthy Eating and Living (WHEL) randomized trial. JAMA. 2007;298:289-298.
11. Chlebowski RT, Blackburn GL, Thomson CA, et al. Dietary fat reduction and breast cancer outcome: interim efficacy results from the Women’s Intervention Nutrition Study (WINS). J Natl Cancer Inst. 2006;98:1767-1776.
12. PDQ Cancer Information Summary. Breast Cancer Prevention (PRQ) – Health Professional. Date last modified April 30, 2009. Available at: http://www.cancer.gov/cancertopics/pdq/prevention/breast/healthprofessional. Accessed May 12, 2009.
13. Hulka BS, Moorman PG. Breast cancer: hormones and other risk factors. Maturitas. 2001;38:103-113.
14. Rossouw JE, Anderson GL, Prentice RL, et al. Risks and benefits of estrogen plus progestin in healthy postmenopausal women: principal results From the Women’s Health Initiative randomized controlled trial. JAMA. 2002;288:321-333.
15. Ravdin PM, Cronin KA, Howlader N, et al. The decrease in breast-cancer incidence in 2003 in the United States. N Engl J Med. 2007;356:1670-1674.
16. Chlebowski RT, Kuller LH, Prentice RL, et al. WHI Investigators. Breast cancer after use of estrogen plus progestin in postmenopausal women. N Engl J Med. 2009;360:573-587.
17. Kosters JP, Gotzsche PC. Regular self-examination or clinical examination for early detection of breast cancer. Cochrane Database Syst Rev. 2003;(2):CD003373.-
18. Canadian Task Force on the Periodic Health Examination Ottawa, Canada: Health Canada; 1994:788-795 (reaffirmed by the Canadian Task Force on the Periodic Health Examination 1999, 2001).Available at: http://www.ctfphc.org/index/html. Accessed August 12, 2009.
19. Green BB, Taplin SH. Breast cancer screening controversies. J Am Board Fam Pract. 2003;16:233-241.
20. Berry DA, Cronin KA, Plevritis SK, et al. Effect of screening and adjuvant therapy on mortality from breast cancer. N Engl J Med. 2005;353:1784-1792.
21. Tice JA, Kerlikowske K. Screening and prevention of breast cancer in primary care. Prim Care. 2009;36:533-558.
22. US Preventive Services Task Force. Screening for breast cancer. Systematic evidence review update for the US Preventive Services Task Force. Available at: http://www.ncbi.nlm.nih.gov/bookshelf/br.fcgi?book=es74. Accessed September 2, 2010.
23. Pisano ED, Gatsonis C, Hendrick E, et al. Digital Mammographic Imaging Screening Trial (DMIST) Investigators Group. Diagnostic performance of digital versus film mammography for breast-cancer screening. N Engl J Med. 2005;353:1773-1783.
24. Saslow D, Boetes C, Burke W, et al. American Cancer Society Breast Cancer Advisory Group. American Cancer Society guidelines for breast screening with MRI as an adjunct to mammography. CA Cancer J Clin. 2007;57:75-89.
25. Berg W, Blume J, Cormack J, et al. Combined screening with ultrasound and mammography vs mammography alone in women at elevated risk of breast cancer. JAMA. 2008;299:2151-2163.
26. King M, Wieand S, Hale K, et al. Tamoxifen and breast cancer incidence among women with inherited mutations in BRCA1 and BRCA2: National Surgical Adjuvant Breast and Bowel Project (NSABP-P1) Breast Cancer Prevention Trial. JAMA. 2001;286:2251-2256.
27. Vogel VG, Costantino JP, Wickerham DL, et al. Effects of tamoxifen vs raloxifene on the risk of developing invasive breast cancer and other disease outcomes: the NSABP Study of Tamoxifen and Raloxifene (STAR) P-2 trial. JAMA. 2006;295:2727-2741.
28. Berg AO. US Preventive Services Task Force. Chemoprevention of breast cancer: recommendations and rationale. Am J Nurs. 2003;103:107-113.
29. National Comprehensive Cancer Network. Breast cancer risk reduction. Clinical Practice Guidelines in Oncology-V.2.2010. Fort Washington, Pa: National Comprehensive Cancer Network; August 7, 2010. Available at: http://www.nccn.org/professionals/physician_gls/f_guidelines.asp. Accessed September 17, 2010.
30. Sickles EA, Filly RA, Cllen PW. Benign breast lesions: ultrasound detection and diagnosis. Radiology. 1984;151:467.-
31. Beavers TB, Anderson BO, Bonaccio E, et al. NCCN clinical practice guidelines in oncology: breast cancer screening and diagnosis. J Natl Compr Canc Netw. 2009;7:1060-1096.
32. Mansel RE, Fallowfield L, Kissin M, et al. Randomized multicenter trial of sentinel node biopsy versus standard axillary treatment in operable breast cancer: the ALMANAC trial. J Natl Canc Inst. 2006;98:599-609
33. Lyman GH, Guiliano AE, Somerfield MR, et al. American Society of Clinical Oncology guideline recommendations for sentinel lymph node biopsy in early- stage breast cancer. J Clin Oncol. 2005;23:7703-7720.
34. Turner N, Jones A. Management of breast cancer-Part II. BMJ. 2008;337:a540.-
35. Harris L, Fritsche H, Mennel R, et al:. American Society of Clinical Oncology 2007 update of recommendations for the use of tumor markers in breast cancer J Clin Oncol. 2007;25:5287-5312.
36. Piccart-Gebhart MJ, Procter M, Leyland-Jones B, et al. Herceptin adjuvant (HERA) Trial Study Team. Trastuzumab after adjuvant chemotherapy in HER2-positive breast cancer. N Engl J Med. 2005;353:1659-1672.
37. PDQ Cancer Information Summary Breast cancer treatment– health professional. Available at: http://www.cancer.gov/cancertopics/pdq/treatment/breast/HealthProfessional. Accessed May 12, 2009.
38. Anderson BL, Yang HC, Farrar WB, et al. Psychologic intervention improves survival for breast cancer patients: a randomized clinical trial. Cancer. 2008;113:3450-3458.
39. Jin Y, Desta Z, Stearns V, et al. CYP2D6 genotype, antidepressant use, and tamoxifen metabolism during adjuvant breast cancer treatment. J Natl Cancer Inst. 2005;97:30-39.
• Offer screening magnetic resonance imaging (MRI) to patients with a known BRCA 1 or 2 mutation, a strong family history of breast cancer, or a lifetime risk of breast cancer >20% to 25%. B
• For early-stage breast cancer, lumpectomy and sentinel node mapping with excision is the preferred method for staging. A
• Monitor patients receiving tamoxifen for signs and symptoms of venous thromboembolism, cataracts, and uterine malignancy, and patients on aromatase inhibitors for the development of osteoporosis. A
Strength of recommendation (SOR)
A Good-quality patient-oriented evidence
B Inconsistent or limited-quality patient-oriented evidence
C Consensus, usual practice, opinion, disease-oriented evidence, case series
Late last year, the US Preventive Services Task Force (USPSTF) sparked a nationwide controversy when it announced that it was recommending against routine screening mammography for women younger than age 50.1 Indeed, that’s a recommendation that many other organizations, including the American Cancer Society (ACS),2 the American College of Obstetricians and Gynecologists (ACOG),3 and the National Comprehensive Cancer Network (NCCN),4 disagree with. But the age at which women should begin routine mammography isn’t the only controversial question. Experts disagree on the benefits of breast self-examination, the optimal frequency of clinical breast exams, and the use of digital mammography—among other issues. This evidence-based review can help you cut through the confusion.
CASE Carrie, a 39-year-old woman who has never been pregnant, comes in for an annual Pap smear and gynecologic exam. She has a negative past medical history, but a positive family history for breast cancer—both her mother and 1 of her sisters had the disease. How would you assess Carrie’s risk of breast cancer, and what preventive measures would you recommend?
Use this predictive model to pinpoint your patient’s risk
When making decisions regarding primary prevention of and screening for breast cancer, an accurate assessment of risk is critical. Many predictive models have been developed with that in mind. The most widely studied, the Gail model, incorporates a number of important risk factors (TABLE 1), including age; race; family history; reproductive factors such as age of menarche, menopause, and first childbirth; and previous history of breast biopsy and atypical findings, to calculate a woman’s 5-year risk.5
A risk calculator (the Breast Cancer Risk Assessment Tool) based on the Gail model is available on the National Cancer Institute’s Web site, at http://www.cancer.gov/bcrisktool. Generally, a score ≥1.66%,5 which indicates that a patient has at least a 1.66% chance of developing breast cancer over the next 5 years, is considered high risk.6,7
CASE Carrie’s 2 first-degree relatives affected by breast cancer and her nulliparous status place her at increased risk. Further questioning reveals a particularly strong family history, as both relatives were diagnosed before the age of 50 (her mom at 45 years of age and her sister, at 39). Carrie’s 5-year risk is 1.8%.
TABLE 1
Risk factors for breast cancer5,29
|
| *African American and Caucasian women are at higher risk compared with Asian, Hispanic, and Native American women. |
| †1 drink/day results in minimal increase in risk; 2-5 drinks/day result in 1.5 increased risk compared with nondrinkers. |
| HRT, hormone replacement therapy; LCIS, lobular carcinoma in situ. |
All women can benefit from these preventive measures
As primary care physicians, we have a responsibility to stress lifestyle modification as the mainstay of breast cancer prevention. Whether or not a woman is at high risk, advise her that maintaining a normal weight, exercising vigorously, limiting alcohol consumption, and breastfeeding are evidence-based methods of primary prevention. Diets low in fat and high in fiber may be associated with a lower risk of invasive breast cancer, but there is no conclusive evidence to support specific dietary interventions to reduce the risk.8-11 Nor has a link between active or passive smoking, antioxidants, or fruit and vegetable intake been firmly established.12
There is a clear association between prolonged estrogen exposure and breast cancer, however. Many reproductive factors, such as early menarche, late menopause, later age at time of first full-term pregnancy, and nulliparity, increase a woman’s exposure to endogenous estrogen—and her risk of developing breast cancer.12,13
Exposure to exogenous estrogen is also linked to the development of breast cancer. In 2002, the Women’s Health Initiative (WHI) was stopped early after a report was released stating that the risks of hormone replacement therapy (HRT)—a higher incidence of cardiovascular events, stroke, and venous thromboembolism, as well as breast cancer—outweighed the benefits.14 Subsequent analyses have found a relationship between the declining incidence of breast cancer and the marked decrease in HRT use prompted by the WHI report. While causality has not been firmly established, multiple studies strongly suggest it.15,16
The association between oral contraceptives (OCs) and breast cancer is more controversial. Some studies have found an increased breast cancer risk among OC users, but both the relative risk and absolute risk were found to be very small and to dissipate 10 years after stopping OC use. More recent studies with newer formulations containing lower doses of estrogen have failed to show an increased risk.8
Breast cancer screening: The parameters have changed
Various organizations have published guidelines for breast cancer screening (TABLE 2), and all are somewhat different. Here’s what you need to know.
Breast self-examination (BSE), which women were previously advised to perform monthly, has not been shown to improve mortality in any age group, and is no longer routinely recommended.17 While both the USPSTF and the Canadian Task Force on Preventive Health Care recommend against teaching women BSE, the ACS, ACOG, and NCCN encourage self-examination—particularly among women older than 40 years.1-4,17,18
Clinical breast examination has an average sensitivity of 50% and detects approximately 5% of mammographically occult cancers.19 It is still not clear whether clinical breast exams save lives, however—a finding that is reflected in the USPSTF’s “I” (insufficient evidence to assess the benefits and harms) recommendation.1 Other consensus guidelines still recommend clinical breast examination, albeit at varying frequencies.
Screening mammography decreases mortality rates by anywhere from 28% to 65%, depending on the statistical model used.20 The benefit is greatest in women between the ages of 50 and 69 years, however, and most groups agree that mammography every 1 to 2 years is advisable for this age group. (There is limited data on the value of mammography for women 70 years of age and older, and no consensus on the age at which to stop screening.1,21) But because the mortality benefit from screening mammography is lower for women aged 40 to 49, guidelines for this age group are more controversial.
Mammography’s sensitivity is affected by a variety of factors, including age and menopausal status, prior breast surgery or radiation, breast density, and the experience of the radiologist. Women in their 40s have denser breast tissue than older women, making mammography less sensitive for this age group. Because of that, and because the overall incidence of breast cancer is lower for women younger than 50, some argue that screening mammography for women between the ages of 40 and 49 years leads to unacceptably high false-positive rates (9.8% annually22) and that the harm associated with mammography may outweigh the benefit. Others counter that tumors in younger women tend to be more aggressive and faster growing, making early detection even more critical than for older women.
What should you advise women in this age group? You might point out that the USPSTF recommends against routine screening, but indicates that the decision to begin (or defer) routine mammography before age 50 should be individualized, based on the needs and values of each patient.1
Digital mammography. A recent study of more than 43,000 women demonstrated that digital mammography is more accurate than film—but only for certain groups: These include women <50 years of age, women with dense breasts, and pre- and perimenopausal women. 23 Because it is still not clear whether the increased accuracy will translate into a mortality benefit, more research is needed before digital mammography is widely adopted. The USPSTF maintains that there is insufficient evidence to assess the benefits and harms of using either digital mammography or magnetic resonance imaging (MRI) rather than film mammography to screen for breast cancer.1
MRI. In 2007, the ACS published guidelines on the use of MRI as an adjunct to mammography for breast cancer screening in high-risk women.24 According to ACS guidelines, screening MRI should be offered to patients with a known BRCA 1 or 2 mutation (5%-10% of all breast cancers are associated with a mutation in the BRCA 1 or BRCA 2 gene, which is transmitted in an autosomal dominant pattern6). It also should be offered to those with a strong family history, or a lifetime risk of developing breast cancer that is >20% to 25%. And finally, MRI should be offered to women who had chest wall radiation when they were between the ages of 10 and 30 years—another significant risk factor for breast cancer—and those with other genetic syndromes that increase their lifetime risk of breast cancer.24
Evidence is insufficient for or against MRI screening for women with a personal history of breast cancer, atypical hyperplasia, or lobular carcinoma in situ, however, and neither breast ultrasound (which is generally used diagnostically, not for screening purposes) nor MRI has been shown to be helpful as a screening tool in women with <15% lifetime risk of developing breast cancer.24,25
TABLE 2
Guidelines for breast cancer screening for women with average risk
| Organization | Age (years) | Breast self-exam | Clinical breast exam | Mammography |
|---|---|---|---|---|
| American Cancer Society2 | 20-40 | Optional | Every 3 y | NA |
| >40 | Encourages | Annually | Annually | |
| American College of Obstetricians and Gynecologists3 | 40-49 | Encourages | Annually | 1-2 y |
| 50-69 | Encourages | Annually | Annually | |
| Canadian Task force on Preventive Health Care18 | 40-49 | Recommends against teaching | Insufficient evidence | Insufficient evidence |
| 50-69 | Recommends against teaching | 1-2 y | 1-2 y | |
| National Comprehensive Cancer Network4 | 20-40 | Encourages | 1-3 y | NA |
| >40 | Encourages | Annually | Annually | |
| US Preventive Services Task Force1 | 40-49 | Recommends against teaching | Insufficient evidence | Not routinely recommended |
| 50-74 | Recommends against teaching | Insufficient evidence | Every 2 y | |
| NA, not addressed. | ||||
When to consider chemoprevention
For women like Carrie, who are at high risk of developing breast cancer, selective estrogen receptor modulator (SERM) therapy and surgical interventions may be options to consider. The Breast Cancer Prevention Trial demonstrated the efficacy of tamoxifen as a preventive agent. This landmark trial showed that for high-risk women older than 35, 5 years of tamoxifen therapy can reduce the incidence of invasive breast cancer by nearly 50%.26
Women with the BRCA 1 or 2 mutation—all of whom should be offered genetic counseling—were included in the study. Tamoxifen reduced the incidence of breast cancer in BRCA 2 carriers by 62%, the researchers found, but did not reduce risk in carriers of the BRCA 1 gene. This is likely due to the high prevalence of estrogen receptor-negative breast cancers among BRCA 1 carriers.26
More recently, the Study of Tamoxifen and Raloxifene (STAR) trial compared the efficacy of tamoxifen and raloxifene, a second-generation SERM, in high-risk postmenopausal women ages 35 and older. The drugs were found to be equally effective in reducing the risk of invasive breast cancer, but raloxifene had a better side effect profile, with a lower incidence of thromboembolism and cataracts. 27
What the guidelines call for. In 2003, the USPSTF recommended that clinicians discuss chemoprevention with women at high risk for breast cancer and low risk for adverse effects of SERMs.28
The most recent update to the NCCN breast cancer risk reduction guidelines recommends that clinicians offer tamoxifen to premenopausal women with a 5-year projected breast cancer risk ≥1.7% and offer tamoxifen or raloxifene to high-risk postmenopausal women.29 It is worth noting, however, that SERMs can have significant adverse effects, including venous thromboembolism, stroke, cataracts, uterine malignancy, and hot flashes, while lifestyle modifications and the avoidance of HRT have few, if any, negative effects.
CASE After consultation with a genetic counselor, Carrie underwent testing for both the BRCA 1 and BRCA 2 mutations. She tested negative for both. She declined chemoprevention and prophylactic surgery, opting for enhanced screening with yearly mammography and MRI and lifestyle modification instead.
When a mass is found
For women ages 30 or older with palpable masses or solid masses ≥2 cm found on imaging, core needle biopsy is recommended.30,31 Biopsy is indicated for women younger than 30 as well, if the mass is >2 cm or imaging is suspicious. In general, a needle biopsy read as benign is considered adequate for diagnostic purposes only if the lesion appeared benign on imaging.
For lesions shown to be cystic on imaging, recommendations for follow-up or additional testing are based on the characteristics of the cyst. For simple cysts, 2- to 4-month follow up for stability, followed by routine screening, is adequate.21 Additional evaluation of complex cysts is indicated, including aspiration for complicated cysts and biopsy for complex cysts. After aspiration, surgical excision of bloody aspirates or persistent masses is recommended.30,31
Staging using the TNM system
The TNM (tumor, node, metastases) classification system is used for the staging of breast cancer:
- T refers to the tumor type, size, and extent of local involvement
- N describes regional lymph node involvement
- M refers to distant metastases.
The TNM classifications are also grouped by stage (I through IV).,
Lumpectomy and sentinel node mapping with excision is the preferred method for staging of early-stage breast cancer without palpable lymphadenopathy—provided that the surgical team has documented experience with sentinel node biopsy.32 Sentinel node biopsy is preferred because of its safety, low (<10%) false negative rate, and decreased morbidity compared with full axillary dissection, although dissection is recommended for patients with more advanced cancer or a positive sentinel node.32 The comparative effects of sentinel node biopsy vs axillary node dissection on tumor recurrence and patient survival are not known.33
Testing for tumor markers such as estrogen and progesterone receptors and human epidermal growth factor receptor 2 (HER2) expression status in biopsy-proven breast cancer is now the standard of care. Seventy percent of breast cancers are estrogen receptor-positive, with increasing frequency associated with older age.34 Estrogen/progesterone receptor positivity is associated with a more favorable outcome, and multiple hormonal therapies can be aimed at these receptors.34 While HER2 overexpression—which occurs in 15% to 30% of newly diagnosed breast cancers35—is associated with more aggressive tumors, women with this type of tumor cell can benefit from trastuzumab, an anti-HER2 drug.36
Key factors that affect prognosis
Important factors affecting prognosis and treatment of localized breast cancer are tumor size, age and menopausal status, tumor expression of hormone receptors and/ or the HER2 protein, as well as the status of the draining axillary nodes. Factors that predict a greater chance of recurrence include the spread of disease to axillary nodes, larger tumor size, invasive histology, inflammatory pathology, lack of estrogen/progesterone receptors, and age <50 years or premenopausal status.
Treatment options include surgical resection, radiation, and systemic adjuvant therapy in the form of chemotherapy, endocrine therapy, or anti-HER2 monoclonal antibodies.37 (For more on treatment, see “Surgery, radiation, and systemic therapy: Making the most of what’s in our arsenal” at jfponline.com.)
Don’t overlook quality-of-life issues
Follow-up of breast cancer patients should go beyond treatment and work-up for recurrence and metastatic disease to focus on health and lifestyle issues, such as stress reduction, mood, smoking cessation, diet and exercise, treatment of hot flashes, sexual dysfunction, and bone health. A recent study found both reduced recurrence and increased survival in women receiving psychological interventions to improve quality-of-life measures after an 11-year follow-up.38
Refer women to targeted Web sites such as the National Breast Cancer Awareness Month organization (http://www.nbcam.org/), the National Breast Cancer Foundation (http://community.nationalbreastcancer.org/), and the Susan G. Komen Breast Cancer Foundation (http://ww5.komen.org/). Offer treatment for bothersome symptoms. Hot flashes and depression, for example, often related to endocrine therapy, can be treated with selective serotonin reuptake inhibitors (SSRIs). That said, some SSRIs decrease the active metabolite of tamoxifen by inhibiting CYP2D6 enzyme and must, therefore, be used with caution. However, venlafaxine and citalopram are less likely to alter tamoxifen metabolism than other SSRIs.39
CASE When Carrie was 47, she had an abnormal MRI of the left breast. Core needle biopsy and pathology of the lesion revealed an estrogen and progesterone receptor-positive tumor that was negative for HER2 overexpression. She underwent lumpectomy, which revealed a 1.5 cm tumor, followed by a negative sentinel node biopsy, and was diagnosed with stage I (T1N0M0) breast cancer. Carrie had radiation after surgery; she did not require chemotherapy, but was told to take tamoxifen for 5 years. This adjuvant endocrine therapy led to hot flashes and depression, both of which were successfully treated with venlafaxine. Carrie is currently cancer-free and participates in a breast cancer survivor program that includes regular visits with her primary physician and her oncologist.
CORRESPONDENCE Denise Sur, MD, 1920 Colorado Avenue, Santa Monica, CA 90404; [email protected]
Surgery, radiation, and systemic therapy: Making the most of what’s in our arsenal
Breast cancer surgery has changed dramatically over the years. Multiple studies have shown that breast-conserving therapy (lumpectomy followed by radiation) for carefully selected women is comparable to mastectomy for local recurrence and survival. While there has been much interest in determining whether a subset of patients could forego radiation after lumpectomy, a meta-analysis by the Early Breast Cancer Trialists Collaborative Group demonstrated that radiation after lumpectomy provides an absolute local recurrence risk reduction of 19%, and a 5.4% absolute reduction in 15-year breast cancer mortality rates compared with lumpectomy without radiation.1 Thus, radiation after lumpectomy remains the standard of care for all women undergoing breast-conserving therapy, regardless of tumor characteristics.
In certain women with a high risk of recurrence (≥4 positive nodes), radiation is also recommended after mastectomy. Women undergoing mastectomy have numerous options for immediate or delayed breast reconstruction. Consultation with a multidisciplinary team, including a plastic surgeon, prior to any surgical intervention is advised.2
Multiple systemic chemotherapy regimens have been shown to be beneficial in carefully selected patients with breast cancer. Systematic reviews have demonstrated that an anthracycline-based regimen can decrease annual breast cancer mortality by 38% in women <50 years old and by 20% in women ages 50 to 69 years.1 in more recent randomized controlled trials, the addition of taxanes to anthracycline-based regimens has produced promising results.3
Numerous hormonal therapies benefit women with estrogen or progesterone receptor-positive breast cancer. Tamoxifen blocks the activity of estrogen on receptors located in breast cancer tissue, for example; aromatase inhibitors block the conversion of androgens to estrogen; and gonadotropin-releasing hormone (GnRH) analogs such as leuprolide and goserelin suppress ovarian production of estrogen.
For postmenopausal women, options include an aromatase inhibitor alone or tamoxifen followed by an aromatase inhibitor.
In premenopausal women, aromatase inhibitors are not very effective, as decreasing peripheral estrogen stimulates the ovaries to produce more estrogen. Thus, for these patients, adjuvant endocrine therapy consists of tamoxifen, with ovarian ablation (via surgery or radiation) or ovarian suppression with a GnRH analog. If the patient goes through menopause as a result of this therapy, she may benefit from aromatase inhibitors at that time.4,5
Women with breast cancer that overexpresses the HER2 gene benefit from adjuvant treatment with trastuzumab, an anti-HER2 antibody.6 While current guidelines advise treatment for 1 year, multiple studies are evaluating dosing schedules and optimal duration of treatment. for now, patients should be monitored for signs of cardiotoxicity at baseline and every 3 months thereafter until completion of therapy.4
References
1. Early Breast Caner Trialists’ Collaborative Group (EBCTCG). Effects of chemotherapy and hormonal therapy for early breast cancer on recurrence and 15-year survival: an overview of the randomised trials. Lancet. 2005;265:1687-1717.
2. Codeiro P. Breast reconstruction after surgery for breast cancer. N Engl J Med. 2008;359:1590-1601.
3. DeLaurentiis M, Cancello G, D’Agostino D, et al. Taxane-based combinations as adjuvant chemotherapy of early breast cancer: a meta-analysis of randomized trials. J Clin Oncol. 2008;26:44-53.
4. National Comprehensive Cancer Network. Breast cancer risk reduction clinical practice guidelines in oncology. J Natl Compr Canc Netw. 2003;1:280-296.
5. Baum M, Budzar AU, Cuzick J, et al. Anastrozole alone or in combination with tamoxifen versus tamoxifen alone for adjuvant treatment of postmenopausal women with early breast cancer: first results of the ATAC randomised trial. Lancet. 2002;359:2131-2139.
6. Piccart-Gebhart MJ, Procter M, Leyland-Jones B, et al. Herceptin adjuvant (HERA) Trial Study Team. Trastuzumab after adjuvant chemotherapy in HER2-positive breast cancer. N Engl J Med. 2005;353:1659-1672.
• Offer screening magnetic resonance imaging (MRI) to patients with a known BRCA 1 or 2 mutation, a strong family history of breast cancer, or a lifetime risk of breast cancer >20% to 25%. B
• For early-stage breast cancer, lumpectomy and sentinel node mapping with excision is the preferred method for staging. A
• Monitor patients receiving tamoxifen for signs and symptoms of venous thromboembolism, cataracts, and uterine malignancy, and patients on aromatase inhibitors for the development of osteoporosis. A
Strength of recommendation (SOR)
A Good-quality patient-oriented evidence
B Inconsistent or limited-quality patient-oriented evidence
C Consensus, usual practice, opinion, disease-oriented evidence, case series
Late last year, the US Preventive Services Task Force (USPSTF) sparked a nationwide controversy when it announced that it was recommending against routine screening mammography for women younger than age 50.1 Indeed, that’s a recommendation that many other organizations, including the American Cancer Society (ACS),2 the American College of Obstetricians and Gynecologists (ACOG),3 and the National Comprehensive Cancer Network (NCCN),4 disagree with. But the age at which women should begin routine mammography isn’t the only controversial question. Experts disagree on the benefits of breast self-examination, the optimal frequency of clinical breast exams, and the use of digital mammography—among other issues. This evidence-based review can help you cut through the confusion.
CASE Carrie, a 39-year-old woman who has never been pregnant, comes in for an annual Pap smear and gynecologic exam. She has a negative past medical history, but a positive family history for breast cancer—both her mother and 1 of her sisters had the disease. How would you assess Carrie’s risk of breast cancer, and what preventive measures would you recommend?
Use this predictive model to pinpoint your patient’s risk
When making decisions regarding primary prevention of and screening for breast cancer, an accurate assessment of risk is critical. Many predictive models have been developed with that in mind. The most widely studied, the Gail model, incorporates a number of important risk factors (TABLE 1), including age; race; family history; reproductive factors such as age of menarche, menopause, and first childbirth; and previous history of breast biopsy and atypical findings, to calculate a woman’s 5-year risk.5
A risk calculator (the Breast Cancer Risk Assessment Tool) based on the Gail model is available on the National Cancer Institute’s Web site, at http://www.cancer.gov/bcrisktool. Generally, a score ≥1.66%,5 which indicates that a patient has at least a 1.66% chance of developing breast cancer over the next 5 years, is considered high risk.6,7
CASE Carrie’s 2 first-degree relatives affected by breast cancer and her nulliparous status place her at increased risk. Further questioning reveals a particularly strong family history, as both relatives were diagnosed before the age of 50 (her mom at 45 years of age and her sister, at 39). Carrie’s 5-year risk is 1.8%.
TABLE 1
Risk factors for breast cancer5,29
|
| *African American and Caucasian women are at higher risk compared with Asian, Hispanic, and Native American women. |
| †1 drink/day results in minimal increase in risk; 2-5 drinks/day result in 1.5 increased risk compared with nondrinkers. |
| HRT, hormone replacement therapy; LCIS, lobular carcinoma in situ. |
All women can benefit from these preventive measures
As primary care physicians, we have a responsibility to stress lifestyle modification as the mainstay of breast cancer prevention. Whether or not a woman is at high risk, advise her that maintaining a normal weight, exercising vigorously, limiting alcohol consumption, and breastfeeding are evidence-based methods of primary prevention. Diets low in fat and high in fiber may be associated with a lower risk of invasive breast cancer, but there is no conclusive evidence to support specific dietary interventions to reduce the risk.8-11 Nor has a link between active or passive smoking, antioxidants, or fruit and vegetable intake been firmly established.12
There is a clear association between prolonged estrogen exposure and breast cancer, however. Many reproductive factors, such as early menarche, late menopause, later age at time of first full-term pregnancy, and nulliparity, increase a woman’s exposure to endogenous estrogen—and her risk of developing breast cancer.12,13
Exposure to exogenous estrogen is also linked to the development of breast cancer. In 2002, the Women’s Health Initiative (WHI) was stopped early after a report was released stating that the risks of hormone replacement therapy (HRT)—a higher incidence of cardiovascular events, stroke, and venous thromboembolism, as well as breast cancer—outweighed the benefits.14 Subsequent analyses have found a relationship between the declining incidence of breast cancer and the marked decrease in HRT use prompted by the WHI report. While causality has not been firmly established, multiple studies strongly suggest it.15,16
The association between oral contraceptives (OCs) and breast cancer is more controversial. Some studies have found an increased breast cancer risk among OC users, but both the relative risk and absolute risk were found to be very small and to dissipate 10 years after stopping OC use. More recent studies with newer formulations containing lower doses of estrogen have failed to show an increased risk.8
Breast cancer screening: The parameters have changed
Various organizations have published guidelines for breast cancer screening (TABLE 2), and all are somewhat different. Here’s what you need to know.
Breast self-examination (BSE), which women were previously advised to perform monthly, has not been shown to improve mortality in any age group, and is no longer routinely recommended.17 While both the USPSTF and the Canadian Task Force on Preventive Health Care recommend against teaching women BSE, the ACS, ACOG, and NCCN encourage self-examination—particularly among women older than 40 years.1-4,17,18
Clinical breast examination has an average sensitivity of 50% and detects approximately 5% of mammographically occult cancers.19 It is still not clear whether clinical breast exams save lives, however—a finding that is reflected in the USPSTF’s “I” (insufficient evidence to assess the benefits and harms) recommendation.1 Other consensus guidelines still recommend clinical breast examination, albeit at varying frequencies.
Screening mammography decreases mortality rates by anywhere from 28% to 65%, depending on the statistical model used.20 The benefit is greatest in women between the ages of 50 and 69 years, however, and most groups agree that mammography every 1 to 2 years is advisable for this age group. (There is limited data on the value of mammography for women 70 years of age and older, and no consensus on the age at which to stop screening.1,21) But because the mortality benefit from screening mammography is lower for women aged 40 to 49, guidelines for this age group are more controversial.
Mammography’s sensitivity is affected by a variety of factors, including age and menopausal status, prior breast surgery or radiation, breast density, and the experience of the radiologist. Women in their 40s have denser breast tissue than older women, making mammography less sensitive for this age group. Because of that, and because the overall incidence of breast cancer is lower for women younger than 50, some argue that screening mammography for women between the ages of 40 and 49 years leads to unacceptably high false-positive rates (9.8% annually22) and that the harm associated with mammography may outweigh the benefit. Others counter that tumors in younger women tend to be more aggressive and faster growing, making early detection even more critical than for older women.
What should you advise women in this age group? You might point out that the USPSTF recommends against routine screening, but indicates that the decision to begin (or defer) routine mammography before age 50 should be individualized, based on the needs and values of each patient.1
Digital mammography. A recent study of more than 43,000 women demonstrated that digital mammography is more accurate than film—but only for certain groups: These include women <50 years of age, women with dense breasts, and pre- and perimenopausal women. 23 Because it is still not clear whether the increased accuracy will translate into a mortality benefit, more research is needed before digital mammography is widely adopted. The USPSTF maintains that there is insufficient evidence to assess the benefits and harms of using either digital mammography or magnetic resonance imaging (MRI) rather than film mammography to screen for breast cancer.1
MRI. In 2007, the ACS published guidelines on the use of MRI as an adjunct to mammography for breast cancer screening in high-risk women.24 According to ACS guidelines, screening MRI should be offered to patients with a known BRCA 1 or 2 mutation (5%-10% of all breast cancers are associated with a mutation in the BRCA 1 or BRCA 2 gene, which is transmitted in an autosomal dominant pattern6). It also should be offered to those with a strong family history, or a lifetime risk of developing breast cancer that is >20% to 25%. And finally, MRI should be offered to women who had chest wall radiation when they were between the ages of 10 and 30 years—another significant risk factor for breast cancer—and those with other genetic syndromes that increase their lifetime risk of breast cancer.24
Evidence is insufficient for or against MRI screening for women with a personal history of breast cancer, atypical hyperplasia, or lobular carcinoma in situ, however, and neither breast ultrasound (which is generally used diagnostically, not for screening purposes) nor MRI has been shown to be helpful as a screening tool in women with <15% lifetime risk of developing breast cancer.24,25
TABLE 2
Guidelines for breast cancer screening for women with average risk
| Organization | Age (years) | Breast self-exam | Clinical breast exam | Mammography |
|---|---|---|---|---|
| American Cancer Society2 | 20-40 | Optional | Every 3 y | NA |
| >40 | Encourages | Annually | Annually | |
| American College of Obstetricians and Gynecologists3 | 40-49 | Encourages | Annually | 1-2 y |
| 50-69 | Encourages | Annually | Annually | |
| Canadian Task force on Preventive Health Care18 | 40-49 | Recommends against teaching | Insufficient evidence | Insufficient evidence |
| 50-69 | Recommends against teaching | 1-2 y | 1-2 y | |
| National Comprehensive Cancer Network4 | 20-40 | Encourages | 1-3 y | NA |
| >40 | Encourages | Annually | Annually | |
| US Preventive Services Task Force1 | 40-49 | Recommends against teaching | Insufficient evidence | Not routinely recommended |
| 50-74 | Recommends against teaching | Insufficient evidence | Every 2 y | |
| NA, not addressed. | ||||
When to consider chemoprevention
For women like Carrie, who are at high risk of developing breast cancer, selective estrogen receptor modulator (SERM) therapy and surgical interventions may be options to consider. The Breast Cancer Prevention Trial demonstrated the efficacy of tamoxifen as a preventive agent. This landmark trial showed that for high-risk women older than 35, 5 years of tamoxifen therapy can reduce the incidence of invasive breast cancer by nearly 50%.26
Women with the BRCA 1 or 2 mutation—all of whom should be offered genetic counseling—were included in the study. Tamoxifen reduced the incidence of breast cancer in BRCA 2 carriers by 62%, the researchers found, but did not reduce risk in carriers of the BRCA 1 gene. This is likely due to the high prevalence of estrogen receptor-negative breast cancers among BRCA 1 carriers.26
More recently, the Study of Tamoxifen and Raloxifene (STAR) trial compared the efficacy of tamoxifen and raloxifene, a second-generation SERM, in high-risk postmenopausal women ages 35 and older. The drugs were found to be equally effective in reducing the risk of invasive breast cancer, but raloxifene had a better side effect profile, with a lower incidence of thromboembolism and cataracts. 27
What the guidelines call for. In 2003, the USPSTF recommended that clinicians discuss chemoprevention with women at high risk for breast cancer and low risk for adverse effects of SERMs.28
The most recent update to the NCCN breast cancer risk reduction guidelines recommends that clinicians offer tamoxifen to premenopausal women with a 5-year projected breast cancer risk ≥1.7% and offer tamoxifen or raloxifene to high-risk postmenopausal women.29 It is worth noting, however, that SERMs can have significant adverse effects, including venous thromboembolism, stroke, cataracts, uterine malignancy, and hot flashes, while lifestyle modifications and the avoidance of HRT have few, if any, negative effects.
CASE After consultation with a genetic counselor, Carrie underwent testing for both the BRCA 1 and BRCA 2 mutations. She tested negative for both. She declined chemoprevention and prophylactic surgery, opting for enhanced screening with yearly mammography and MRI and lifestyle modification instead.
When a mass is found
For women ages 30 or older with palpable masses or solid masses ≥2 cm found on imaging, core needle biopsy is recommended.30,31 Biopsy is indicated for women younger than 30 as well, if the mass is >2 cm or imaging is suspicious. In general, a needle biopsy read as benign is considered adequate for diagnostic purposes only if the lesion appeared benign on imaging.
For lesions shown to be cystic on imaging, recommendations for follow-up or additional testing are based on the characteristics of the cyst. For simple cysts, 2- to 4-month follow up for stability, followed by routine screening, is adequate.21 Additional evaluation of complex cysts is indicated, including aspiration for complicated cysts and biopsy for complex cysts. After aspiration, surgical excision of bloody aspirates or persistent masses is recommended.30,31
Staging using the TNM system
The TNM (tumor, node, metastases) classification system is used for the staging of breast cancer:
- T refers to the tumor type, size, and extent of local involvement
- N describes regional lymph node involvement
- M refers to distant metastases.
The TNM classifications are also grouped by stage (I through IV).,
Lumpectomy and sentinel node mapping with excision is the preferred method for staging of early-stage breast cancer without palpable lymphadenopathy—provided that the surgical team has documented experience with sentinel node biopsy.32 Sentinel node biopsy is preferred because of its safety, low (<10%) false negative rate, and decreased morbidity compared with full axillary dissection, although dissection is recommended for patients with more advanced cancer or a positive sentinel node.32 The comparative effects of sentinel node biopsy vs axillary node dissection on tumor recurrence and patient survival are not known.33
Testing for tumor markers such as estrogen and progesterone receptors and human epidermal growth factor receptor 2 (HER2) expression status in biopsy-proven breast cancer is now the standard of care. Seventy percent of breast cancers are estrogen receptor-positive, with increasing frequency associated with older age.34 Estrogen/progesterone receptor positivity is associated with a more favorable outcome, and multiple hormonal therapies can be aimed at these receptors.34 While HER2 overexpression—which occurs in 15% to 30% of newly diagnosed breast cancers35—is associated with more aggressive tumors, women with this type of tumor cell can benefit from trastuzumab, an anti-HER2 drug.36
Key factors that affect prognosis
Important factors affecting prognosis and treatment of localized breast cancer are tumor size, age and menopausal status, tumor expression of hormone receptors and/ or the HER2 protein, as well as the status of the draining axillary nodes. Factors that predict a greater chance of recurrence include the spread of disease to axillary nodes, larger tumor size, invasive histology, inflammatory pathology, lack of estrogen/progesterone receptors, and age <50 years or premenopausal status.
Treatment options include surgical resection, radiation, and systemic adjuvant therapy in the form of chemotherapy, endocrine therapy, or anti-HER2 monoclonal antibodies.37 (For more on treatment, see “Surgery, radiation, and systemic therapy: Making the most of what’s in our arsenal” at jfponline.com.)
Don’t overlook quality-of-life issues
Follow-up of breast cancer patients should go beyond treatment and work-up for recurrence and metastatic disease to focus on health and lifestyle issues, such as stress reduction, mood, smoking cessation, diet and exercise, treatment of hot flashes, sexual dysfunction, and bone health. A recent study found both reduced recurrence and increased survival in women receiving psychological interventions to improve quality-of-life measures after an 11-year follow-up.38
Refer women to targeted Web sites such as the National Breast Cancer Awareness Month organization (http://www.nbcam.org/), the National Breast Cancer Foundation (http://community.nationalbreastcancer.org/), and the Susan G. Komen Breast Cancer Foundation (http://ww5.komen.org/). Offer treatment for bothersome symptoms. Hot flashes and depression, for example, often related to endocrine therapy, can be treated with selective serotonin reuptake inhibitors (SSRIs). That said, some SSRIs decrease the active metabolite of tamoxifen by inhibiting CYP2D6 enzyme and must, therefore, be used with caution. However, venlafaxine and citalopram are less likely to alter tamoxifen metabolism than other SSRIs.39
CASE When Carrie was 47, she had an abnormal MRI of the left breast. Core needle biopsy and pathology of the lesion revealed an estrogen and progesterone receptor-positive tumor that was negative for HER2 overexpression. She underwent lumpectomy, which revealed a 1.5 cm tumor, followed by a negative sentinel node biopsy, and was diagnosed with stage I (T1N0M0) breast cancer. Carrie had radiation after surgery; she did not require chemotherapy, but was told to take tamoxifen for 5 years. This adjuvant endocrine therapy led to hot flashes and depression, both of which were successfully treated with venlafaxine. Carrie is currently cancer-free and participates in a breast cancer survivor program that includes regular visits with her primary physician and her oncologist.
CORRESPONDENCE Denise Sur, MD, 1920 Colorado Avenue, Santa Monica, CA 90404; [email protected]
Surgery, radiation, and systemic therapy: Making the most of what’s in our arsenal
Breast cancer surgery has changed dramatically over the years. Multiple studies have shown that breast-conserving therapy (lumpectomy followed by radiation) for carefully selected women is comparable to mastectomy for local recurrence and survival. While there has been much interest in determining whether a subset of patients could forego radiation after lumpectomy, a meta-analysis by the Early Breast Cancer Trialists Collaborative Group demonstrated that radiation after lumpectomy provides an absolute local recurrence risk reduction of 19%, and a 5.4% absolute reduction in 15-year breast cancer mortality rates compared with lumpectomy without radiation.1 Thus, radiation after lumpectomy remains the standard of care for all women undergoing breast-conserving therapy, regardless of tumor characteristics.
In certain women with a high risk of recurrence (≥4 positive nodes), radiation is also recommended after mastectomy. Women undergoing mastectomy have numerous options for immediate or delayed breast reconstruction. Consultation with a multidisciplinary team, including a plastic surgeon, prior to any surgical intervention is advised.2
Multiple systemic chemotherapy regimens have been shown to be beneficial in carefully selected patients with breast cancer. Systematic reviews have demonstrated that an anthracycline-based regimen can decrease annual breast cancer mortality by 38% in women <50 years old and by 20% in women ages 50 to 69 years.1 in more recent randomized controlled trials, the addition of taxanes to anthracycline-based regimens has produced promising results.3
Numerous hormonal therapies benefit women with estrogen or progesterone receptor-positive breast cancer. Tamoxifen blocks the activity of estrogen on receptors located in breast cancer tissue, for example; aromatase inhibitors block the conversion of androgens to estrogen; and gonadotropin-releasing hormone (GnRH) analogs such as leuprolide and goserelin suppress ovarian production of estrogen.
For postmenopausal women, options include an aromatase inhibitor alone or tamoxifen followed by an aromatase inhibitor.
In premenopausal women, aromatase inhibitors are not very effective, as decreasing peripheral estrogen stimulates the ovaries to produce more estrogen. Thus, for these patients, adjuvant endocrine therapy consists of tamoxifen, with ovarian ablation (via surgery or radiation) or ovarian suppression with a GnRH analog. If the patient goes through menopause as a result of this therapy, she may benefit from aromatase inhibitors at that time.4,5
Women with breast cancer that overexpresses the HER2 gene benefit from adjuvant treatment with trastuzumab, an anti-HER2 antibody.6 While current guidelines advise treatment for 1 year, multiple studies are evaluating dosing schedules and optimal duration of treatment. for now, patients should be monitored for signs of cardiotoxicity at baseline and every 3 months thereafter until completion of therapy.4
References
1. Early Breast Caner Trialists’ Collaborative Group (EBCTCG). Effects of chemotherapy and hormonal therapy for early breast cancer on recurrence and 15-year survival: an overview of the randomised trials. Lancet. 2005;265:1687-1717.
2. Codeiro P. Breast reconstruction after surgery for breast cancer. N Engl J Med. 2008;359:1590-1601.
3. DeLaurentiis M, Cancello G, D’Agostino D, et al. Taxane-based combinations as adjuvant chemotherapy of early breast cancer: a meta-analysis of randomized trials. J Clin Oncol. 2008;26:44-53.
4. National Comprehensive Cancer Network. Breast cancer risk reduction clinical practice guidelines in oncology. J Natl Compr Canc Netw. 2003;1:280-296.
5. Baum M, Budzar AU, Cuzick J, et al. Anastrozole alone or in combination with tamoxifen versus tamoxifen alone for adjuvant treatment of postmenopausal women with early breast cancer: first results of the ATAC randomised trial. Lancet. 2002;359:2131-2139.
6. Piccart-Gebhart MJ, Procter M, Leyland-Jones B, et al. Herceptin adjuvant (HERA) Trial Study Team. Trastuzumab after adjuvant chemotherapy in HER2-positive breast cancer. N Engl J Med. 2005;353:1659-1672.
1. US Preventive Services Task Force Screening for breast cancer. Available at: http://www.uspreventiveservicestaskforce.org/uspstf/uspsbrca.htm. Accessed September 2, 2010.
2. American Cancer Society. American Cancer Society guidelines for the early detection of cancer. Available at: http://www.cancer.org/Healthy/FindCancerEarly/CancerScreeningGuidelines/american-cancer-society-guidelines-for-the-early-detection-of-cancer. Accessed September 2, 2010.
3. American College of Obstetricians and Gynecologists. Response of the American College of Obstetricians and Gynecologists to the new breast cancer screening recommendations from the US Preventive Services Task Force. Available at: http://www.acog.org/from_home/misc/uspstfresponse.cfm. Accessed September 2, 2010.
4. National Comprehensive Cancer Network. Breast cancer screening and diagnosis. Clinical Practice Guidelines in Oncology-V.1.2010. Fort Washington, Pa: National Comprehensive Cancer Network: November 3, 2009. Available at: http://www.nccn.org/professionals/physician_gls/f_guidelines.asp. Accessed September 17 , 2010.
5. National Cancer Institute. Breast cancer risk assessment tool. Available at: http://www.cancer.gov/bcrisktool/. Accessed September 2, 2010
6. Robson M, Offit K. Clinical practice. Management of an inherited predisposition to breast cancer. N Engl J Med. 2007;357:154-162.
7. Fisher B, Costantino JP, Wickerham DL, et al. Tamoxifen for prevention of breast cancer: report of the National Surgical Adjuvant Breast and Bowel Project P-1 Study. J Natl Cancer Inst. 1998;90:1371-1388.
8. Cummings SR, Tice JA, Bauer S, et al. Prevention of breast cancer in postmenopausal women: approaches to estimating and reducing risk. J Natl Cancer Inst. 2009;18:101,-384-398.
9. Prentice RL, Caan B, Chlebowski RT, et al. Low-fat dietary pattern and risk of invasive breast cancer: the Women’s Health Initiative Randomized Controlled Dietary Modification Trial. JAMA. 2006;295:629-642.
10. Pierce JP, Natarajan L, Caan BJ, et al. Influence of a diet very high in vegetables, fruit, and fiber and low in fat on prognosis following treatment for breast cancer: the Women’s Healthy Eating and Living (WHEL) randomized trial. JAMA. 2007;298:289-298.
11. Chlebowski RT, Blackburn GL, Thomson CA, et al. Dietary fat reduction and breast cancer outcome: interim efficacy results from the Women’s Intervention Nutrition Study (WINS). J Natl Cancer Inst. 2006;98:1767-1776.
12. PDQ Cancer Information Summary. Breast Cancer Prevention (PRQ) – Health Professional. Date last modified April 30, 2009. Available at: http://www.cancer.gov/cancertopics/pdq/prevention/breast/healthprofessional. Accessed May 12, 2009.
13. Hulka BS, Moorman PG. Breast cancer: hormones and other risk factors. Maturitas. 2001;38:103-113.
14. Rossouw JE, Anderson GL, Prentice RL, et al. Risks and benefits of estrogen plus progestin in healthy postmenopausal women: principal results From the Women’s Health Initiative randomized controlled trial. JAMA. 2002;288:321-333.
15. Ravdin PM, Cronin KA, Howlader N, et al. The decrease in breast-cancer incidence in 2003 in the United States. N Engl J Med. 2007;356:1670-1674.
16. Chlebowski RT, Kuller LH, Prentice RL, et al. WHI Investigators. Breast cancer after use of estrogen plus progestin in postmenopausal women. N Engl J Med. 2009;360:573-587.
17. Kosters JP, Gotzsche PC. Regular self-examination or clinical examination for early detection of breast cancer. Cochrane Database Syst Rev. 2003;(2):CD003373.-
18. Canadian Task Force on the Periodic Health Examination Ottawa, Canada: Health Canada; 1994:788-795 (reaffirmed by the Canadian Task Force on the Periodic Health Examination 1999, 2001).Available at: http://www.ctfphc.org/index/html. Accessed August 12, 2009.
19. Green BB, Taplin SH. Breast cancer screening controversies. J Am Board Fam Pract. 2003;16:233-241.
20. Berry DA, Cronin KA, Plevritis SK, et al. Effect of screening and adjuvant therapy on mortality from breast cancer. N Engl J Med. 2005;353:1784-1792.
21. Tice JA, Kerlikowske K. Screening and prevention of breast cancer in primary care. Prim Care. 2009;36:533-558.
22. US Preventive Services Task Force. Screening for breast cancer. Systematic evidence review update for the US Preventive Services Task Force. Available at: http://www.ncbi.nlm.nih.gov/bookshelf/br.fcgi?book=es74. Accessed September 2, 2010.
23. Pisano ED, Gatsonis C, Hendrick E, et al. Digital Mammographic Imaging Screening Trial (DMIST) Investigators Group. Diagnostic performance of digital versus film mammography for breast-cancer screening. N Engl J Med. 2005;353:1773-1783.
24. Saslow D, Boetes C, Burke W, et al. American Cancer Society Breast Cancer Advisory Group. American Cancer Society guidelines for breast screening with MRI as an adjunct to mammography. CA Cancer J Clin. 2007;57:75-89.
25. Berg W, Blume J, Cormack J, et al. Combined screening with ultrasound and mammography vs mammography alone in women at elevated risk of breast cancer. JAMA. 2008;299:2151-2163.
26. King M, Wieand S, Hale K, et al. Tamoxifen and breast cancer incidence among women with inherited mutations in BRCA1 and BRCA2: National Surgical Adjuvant Breast and Bowel Project (NSABP-P1) Breast Cancer Prevention Trial. JAMA. 2001;286:2251-2256.
27. Vogel VG, Costantino JP, Wickerham DL, et al. Effects of tamoxifen vs raloxifene on the risk of developing invasive breast cancer and other disease outcomes: the NSABP Study of Tamoxifen and Raloxifene (STAR) P-2 trial. JAMA. 2006;295:2727-2741.
28. Berg AO. US Preventive Services Task Force. Chemoprevention of breast cancer: recommendations and rationale. Am J Nurs. 2003;103:107-113.
29. National Comprehensive Cancer Network. Breast cancer risk reduction. Clinical Practice Guidelines in Oncology-V.2.2010. Fort Washington, Pa: National Comprehensive Cancer Network; August 7, 2010. Available at: http://www.nccn.org/professionals/physician_gls/f_guidelines.asp. Accessed September 17, 2010.
30. Sickles EA, Filly RA, Cllen PW. Benign breast lesions: ultrasound detection and diagnosis. Radiology. 1984;151:467.-
31. Beavers TB, Anderson BO, Bonaccio E, et al. NCCN clinical practice guidelines in oncology: breast cancer screening and diagnosis. J Natl Compr Canc Netw. 2009;7:1060-1096.
32. Mansel RE, Fallowfield L, Kissin M, et al. Randomized multicenter trial of sentinel node biopsy versus standard axillary treatment in operable breast cancer: the ALMANAC trial. J Natl Canc Inst. 2006;98:599-609
33. Lyman GH, Guiliano AE, Somerfield MR, et al. American Society of Clinical Oncology guideline recommendations for sentinel lymph node biopsy in early- stage breast cancer. J Clin Oncol. 2005;23:7703-7720.
34. Turner N, Jones A. Management of breast cancer-Part II. BMJ. 2008;337:a540.-
35. Harris L, Fritsche H, Mennel R, et al:. American Society of Clinical Oncology 2007 update of recommendations for the use of tumor markers in breast cancer J Clin Oncol. 2007;25:5287-5312.
36. Piccart-Gebhart MJ, Procter M, Leyland-Jones B, et al. Herceptin adjuvant (HERA) Trial Study Team. Trastuzumab after adjuvant chemotherapy in HER2-positive breast cancer. N Engl J Med. 2005;353:1659-1672.
37. PDQ Cancer Information Summary Breast cancer treatment– health professional. Available at: http://www.cancer.gov/cancertopics/pdq/treatment/breast/HealthProfessional. Accessed May 12, 2009.
38. Anderson BL, Yang HC, Farrar WB, et al. Psychologic intervention improves survival for breast cancer patients: a randomized clinical trial. Cancer. 2008;113:3450-3458.
39. Jin Y, Desta Z, Stearns V, et al. CYP2D6 genotype, antidepressant use, and tamoxifen metabolism during adjuvant breast cancer treatment. J Natl Cancer Inst. 2005;97:30-39.
1. US Preventive Services Task Force Screening for breast cancer. Available at: http://www.uspreventiveservicestaskforce.org/uspstf/uspsbrca.htm. Accessed September 2, 2010.
2. American Cancer Society. American Cancer Society guidelines for the early detection of cancer. Available at: http://www.cancer.org/Healthy/FindCancerEarly/CancerScreeningGuidelines/american-cancer-society-guidelines-for-the-early-detection-of-cancer. Accessed September 2, 2010.
3. American College of Obstetricians and Gynecologists. Response of the American College of Obstetricians and Gynecologists to the new breast cancer screening recommendations from the US Preventive Services Task Force. Available at: http://www.acog.org/from_home/misc/uspstfresponse.cfm. Accessed September 2, 2010.
4. National Comprehensive Cancer Network. Breast cancer screening and diagnosis. Clinical Practice Guidelines in Oncology-V.1.2010. Fort Washington, Pa: National Comprehensive Cancer Network: November 3, 2009. Available at: http://www.nccn.org/professionals/physician_gls/f_guidelines.asp. Accessed September 17 , 2010.
5. National Cancer Institute. Breast cancer risk assessment tool. Available at: http://www.cancer.gov/bcrisktool/. Accessed September 2, 2010
6. Robson M, Offit K. Clinical practice. Management of an inherited predisposition to breast cancer. N Engl J Med. 2007;357:154-162.
7. Fisher B, Costantino JP, Wickerham DL, et al. Tamoxifen for prevention of breast cancer: report of the National Surgical Adjuvant Breast and Bowel Project P-1 Study. J Natl Cancer Inst. 1998;90:1371-1388.
8. Cummings SR, Tice JA, Bauer S, et al. Prevention of breast cancer in postmenopausal women: approaches to estimating and reducing risk. J Natl Cancer Inst. 2009;18:101,-384-398.
9. Prentice RL, Caan B, Chlebowski RT, et al. Low-fat dietary pattern and risk of invasive breast cancer: the Women’s Health Initiative Randomized Controlled Dietary Modification Trial. JAMA. 2006;295:629-642.
10. Pierce JP, Natarajan L, Caan BJ, et al. Influence of a diet very high in vegetables, fruit, and fiber and low in fat on prognosis following treatment for breast cancer: the Women’s Healthy Eating and Living (WHEL) randomized trial. JAMA. 2007;298:289-298.
11. Chlebowski RT, Blackburn GL, Thomson CA, et al. Dietary fat reduction and breast cancer outcome: interim efficacy results from the Women’s Intervention Nutrition Study (WINS). J Natl Cancer Inst. 2006;98:1767-1776.
12. PDQ Cancer Information Summary. Breast Cancer Prevention (PRQ) – Health Professional. Date last modified April 30, 2009. Available at: http://www.cancer.gov/cancertopics/pdq/prevention/breast/healthprofessional. Accessed May 12, 2009.
13. Hulka BS, Moorman PG. Breast cancer: hormones and other risk factors. Maturitas. 2001;38:103-113.
14. Rossouw JE, Anderson GL, Prentice RL, et al. Risks and benefits of estrogen plus progestin in healthy postmenopausal women: principal results From the Women’s Health Initiative randomized controlled trial. JAMA. 2002;288:321-333.
15. Ravdin PM, Cronin KA, Howlader N, et al. The decrease in breast-cancer incidence in 2003 in the United States. N Engl J Med. 2007;356:1670-1674.
16. Chlebowski RT, Kuller LH, Prentice RL, et al. WHI Investigators. Breast cancer after use of estrogen plus progestin in postmenopausal women. N Engl J Med. 2009;360:573-587.
17. Kosters JP, Gotzsche PC. Regular self-examination or clinical examination for early detection of breast cancer. Cochrane Database Syst Rev. 2003;(2):CD003373.-
18. Canadian Task Force on the Periodic Health Examination Ottawa, Canada: Health Canada; 1994:788-795 (reaffirmed by the Canadian Task Force on the Periodic Health Examination 1999, 2001).Available at: http://www.ctfphc.org/index/html. Accessed August 12, 2009.
19. Green BB, Taplin SH. Breast cancer screening controversies. J Am Board Fam Pract. 2003;16:233-241.
20. Berry DA, Cronin KA, Plevritis SK, et al. Effect of screening and adjuvant therapy on mortality from breast cancer. N Engl J Med. 2005;353:1784-1792.
21. Tice JA, Kerlikowske K. Screening and prevention of breast cancer in primary care. Prim Care. 2009;36:533-558.
22. US Preventive Services Task Force. Screening for breast cancer. Systematic evidence review update for the US Preventive Services Task Force. Available at: http://www.ncbi.nlm.nih.gov/bookshelf/br.fcgi?book=es74. Accessed September 2, 2010.
23. Pisano ED, Gatsonis C, Hendrick E, et al. Digital Mammographic Imaging Screening Trial (DMIST) Investigators Group. Diagnostic performance of digital versus film mammography for breast-cancer screening. N Engl J Med. 2005;353:1773-1783.
24. Saslow D, Boetes C, Burke W, et al. American Cancer Society Breast Cancer Advisory Group. American Cancer Society guidelines for breast screening with MRI as an adjunct to mammography. CA Cancer J Clin. 2007;57:75-89.
25. Berg W, Blume J, Cormack J, et al. Combined screening with ultrasound and mammography vs mammography alone in women at elevated risk of breast cancer. JAMA. 2008;299:2151-2163.
26. King M, Wieand S, Hale K, et al. Tamoxifen and breast cancer incidence among women with inherited mutations in BRCA1 and BRCA2: National Surgical Adjuvant Breast and Bowel Project (NSABP-P1) Breast Cancer Prevention Trial. JAMA. 2001;286:2251-2256.
27. Vogel VG, Costantino JP, Wickerham DL, et al. Effects of tamoxifen vs raloxifene on the risk of developing invasive breast cancer and other disease outcomes: the NSABP Study of Tamoxifen and Raloxifene (STAR) P-2 trial. JAMA. 2006;295:2727-2741.
28. Berg AO. US Preventive Services Task Force. Chemoprevention of breast cancer: recommendations and rationale. Am J Nurs. 2003;103:107-113.
29. National Comprehensive Cancer Network. Breast cancer risk reduction. Clinical Practice Guidelines in Oncology-V.2.2010. Fort Washington, Pa: National Comprehensive Cancer Network; August 7, 2010. Available at: http://www.nccn.org/professionals/physician_gls/f_guidelines.asp. Accessed September 17, 2010.
30. Sickles EA, Filly RA, Cllen PW. Benign breast lesions: ultrasound detection and diagnosis. Radiology. 1984;151:467.-
31. Beavers TB, Anderson BO, Bonaccio E, et al. NCCN clinical practice guidelines in oncology: breast cancer screening and diagnosis. J Natl Compr Canc Netw. 2009;7:1060-1096.
32. Mansel RE, Fallowfield L, Kissin M, et al. Randomized multicenter trial of sentinel node biopsy versus standard axillary treatment in operable breast cancer: the ALMANAC trial. J Natl Canc Inst. 2006;98:599-609
33. Lyman GH, Guiliano AE, Somerfield MR, et al. American Society of Clinical Oncology guideline recommendations for sentinel lymph node biopsy in early- stage breast cancer. J Clin Oncol. 2005;23:7703-7720.
34. Turner N, Jones A. Management of breast cancer-Part II. BMJ. 2008;337:a540.-
35. Harris L, Fritsche H, Mennel R, et al:. American Society of Clinical Oncology 2007 update of recommendations for the use of tumor markers in breast cancer J Clin Oncol. 2007;25:5287-5312.
36. Piccart-Gebhart MJ, Procter M, Leyland-Jones B, et al. Herceptin adjuvant (HERA) Trial Study Team. Trastuzumab after adjuvant chemotherapy in HER2-positive breast cancer. N Engl J Med. 2005;353:1659-1672.
37. PDQ Cancer Information Summary Breast cancer treatment– health professional. Available at: http://www.cancer.gov/cancertopics/pdq/treatment/breast/HealthProfessional. Accessed May 12, 2009.
38. Anderson BL, Yang HC, Farrar WB, et al. Psychologic intervention improves survival for breast cancer patients: a randomized clinical trial. Cancer. 2008;113:3450-3458.
39. Jin Y, Desta Z, Stearns V, et al. CYP2D6 genotype, antidepressant use, and tamoxifen metabolism during adjuvant breast cancer treatment. J Natl Cancer Inst. 2005;97:30-39.
Transition From Venipuncture to Point-of-Care International Normalized Ratio Testing in a VA Anticoagulation Clinic
Posttraumatic Stress Disorder and Benzodiazepines: A Myth Agreed Upon
More Knowledge, Same Outcomes
Tattoo Removal by Q-Switched Laser
Between seven and 20 million people in the United States, including adolescents as young as 12, are estimated to have at least one tattoo.1-3 Perhaps half later regret the decision to acquire a tattoo—for reasons ranging from an acute inflammatory reaction to the perception that having a tattoo might interfere with opportunities for professional advancement.4 The rising incidence of tattooing may be accompanied by increasing numbers of persons seeking to have decorative tattoos removed. Health care providers need to be aware of the modalities available, along with the risks and benefits of laser tattoo removal.
Tattoo types vary according to etiology, pigment, depth, and purpose. Cosmetic tattoos (“permanent makeup”) often serve to enhance physical features or mask scars; traumatic tattoos result from an injury in which foreign material is embedded in the skin. This article will focus on decorative tattoos and the clinical options for tattooed patients who regret these permanent markings and desire their removal.5
Decorative tattoos can be applied professionally or by amateurs, with pigment initially remaining in the superficial dermis; after several years, the pigment may migrate into deeper layers of the skin.6 Amateur tattoos are composed of ink or carbon; these pigments are usually less dense than those used by professionals, often making amateur tattoos easier to remove (ie, about five sessions of laser therapy for 90% clearance vs six to 10; see figures below).1,7
Professional tattoos are composed of organic pigments that vary in particle size but are applied at a uniform depth of needle penetration.5 The deposited pigment particles reside mainly in dermal fibroblasts and macrophages, although smaller collections of particles can be found within the interstitial space.
Tattoo Removal Techniques
Older techniques of tattoo removal, including surgical excision, salabrasion, dermabrasion, cryosurgery, and chemical peels, have largely been relinquished. Not only did these methods fail to yield desirable results, but they were associated with adverse effects, including hypopigmentation and scarring.5
Although continuous-wave lasers can also cause scarring, quality-switched (Q-switched) lasers have produced more favorable outcomes. The specific color and absorptive characteristics of each tattoo ink will help determine the ideal laser type to be used. In rare cases, patients may be able to contact the responsible artist and inquire about the inks used; information about the absorption spectrum of each pigment could facilitate the treatment plan. Even with this information, however, removal of intricate, colorful tattoos can be a challenge, since several different lasers, used in stages, may be needed.1,8
The patient’s skin type, too, may affect the response to treatment. Q-switched lasers are effective for all skin types, but complete pigment removal may be more difficult in lighter-skinned patients1 (ie, Fitzgerald skin types I and II). Similarly, in older tattoos, complete removal may not be possible, since some dermal pigment may have penetrated too deeply to be reached.5
Selective Photothermolysis
Laser removal of tattoos is accomplished by selective photothermolysis, a process that was first described by Anderson and Parrish in 1983.9,10 Photothermolysis targets specific microscopic sites on the skin, with effectiveness depending on the absorption spectrum of each pigment.4 The epidermis, dermis, and skin appendages are only minimally damaged in the process.6
Laser therapy modifies the optic properties of the tattoo pigments to be removed. The pigments absorb short laser pulses, which produce a high-intensity light in the pigments that is converted into heat.9 Shock waves shatter the pigment particles, achieving the selective death of the pigment-containing cells. The chemical composition of the pigment is also altered.
Lastly, the cell debris is phagocytized and transported to regional lymph nodes. Although they are hardly visible, some residual, scattered particles remain in the dermis. Only superficial pigment fragments are entirely eliminated during epidermal desquamation as repair is occurring, a development called transepidermal elimination.9
To produce the most effective treatment results, the laser wavelength must be absorbed by the ink, the heat should be confined to the target, and adequate energy must be delivered.1
Clinician–Patient Communication
Since the cosmetic outcome of laser therapy depends on both the laser wavelength and the absorption spectrum of each pigment, it is important for health care providers to understand the optimal wavelengths for each pigment type. They should be prepared to address with their patients the issues of incompatible lasers and resistance of certain pigments to treatment.11
Additionally, although Q-switched lasers are considered the gold standard for tattoo removal, realistic expectations should be established. Patients interested in treatment must be informed at the outset that complete clearance is not guaranteed and that the number of treatments and end result depend on factors that vary from patient to patient.4 Ten to 15 sessions, spaced six to eight weeks apart, may be required to achieve a desirable or even acceptable result,9 and the entire process could take a year or longer. The total cost can reach thousands of dollars.12
Before initiating laser therapy (or referring the patient for it), the primary care clinician should collect a history regarding the tattoo’s age and etiology, as well as the patient’s tanning habits, in order to recommend the best treatment. As stated earlier, professional tattoos generally require more treatments than amateur tattoos. Distally located tattoos are the most difficult to remove.1,13
Q-Switched Laser Types
The most common Q-switched lasers are:
• Q-switched ruby laser
• Q-switched Nd:YAG (neodymium:yttrium aluminum garnet) laser
• Q-switched alexandrite laser (see table1,4,6,14).
The choice of laser type is based on several factors, including the presumed absorption spectrum of the target, the desired depth of penetration, the size of the target particle, and the laser’s wavelength and pulse duration.11
Black and India inks absorb broadly across the spectrum. In the case of blue, yellow, or orange pigment, the optimal wavelength for pigment absorption is in an adjacent color. Green pigment absorption spectra vary due to the pigment’s multiple components. The spectra of white, yellow, and “flesh-colored” pigments do not have absorption peaks at the wavelengths of currently used Q-switched lasers; this explains their resistance to removal.11
Use of the Q-switched ruby laser (QSR) is indicated for the removal of black, blue-black, and dark blue pigments. Mixed results have been reported for removal of green and medium blue pigments, and poor results for red, orange, and pale blue. Six treatments at three-week intervals have been reported to yield clearance of 75% or greater in only about 25% of professional, dark-colored tattoos.1,15
Since this laser’s wavelength (694 nm) is absorbed by melanin, its use may result in transient hypopigmentation, depigmentation, and textural changes.1
The Q-switched Nd-YAG lasers (with wavelengths of 532 nm or 1064 nm) have a large spot size, concentrated energy densities, high repetition rates, and greater beam diameter, allowing for rapid, effective treatment of closely clustered and deep tattoos.6 Five treatments of red or orange tattoos may achieve 75% clearance in about 60% of patients.1
The 1064-nm Nd:YAG laser, which has the deepest penetration and carries the least risk for hypopigmentation,1 is indicated for black and dark blue pigments. It is considered the ideal choice for tattoo removal in dark-skinned patients,14,16 since its longer wavelength represents a lower affinity for melanin.4 The 532-nm Nd:YAG laser is effective for removal of red, yellow, and orange pigments.6 Ten or more treatments may be required for 75% clearance of a professional tattoo.1
Some adverse effects of Nd:YAG laser use include whitening of the skin, with occasional mild pinpoint bleeding. Use of the 532-nm model is associated with purpura, resulting from hemoglobin absorption; this may last from one week to 10 days. The 1064-nm Nd:YAG laser is the least effective for removing bright-colored pigments.1,17
The Q-switched alexandrite laser is generally used to remove black, blue, and green pigments. Typically, four to 10 sessions are required at intervals of one to two months. Transient hypopigmentation, typically lasting three to four months, occurs in about half of patients, and textural changes have been reported in about 12%.6,8
The 510-nm, pulsed-dye Q-switched alexandrite laser is reportedly effective in removing red pigment.4
Complications, Adverse Effects, and Their Management
Although Q-switched lasers appear quite effective in tattoo removal, their use is not without adverse effects.
Hypopigmentation
The most common chronic adverse effect of laser treatment is hypopigmentation. The risk is high in dark-skinned patients undergoing treatment with the QSR or alexandrite lasers4 and increases in any patient according to the number of treatment sessions. Hypopigmentation occurs in more than 38% of patients treated with QSR lasers and typically lasts for two to six months.6
In a 2004 study, Gundogan et al18 attempted repigmentation with an excimer laser (Nd:YAG/potassium titanyl phosphate–Nd:YAG) in a patient with hypopigmentation following laser tattoo removal. Repigmentation required 40 treatment sessions over 15 months—not a cost-effective option.6 A better solution might be to minimize the risk for hypopigmentation by use of picosecond lasers (see “Better Options on the Horizon?”, below).8
Hyperpigmentation
Hyperpigmentation can occur as a result of melanocytes’ increased melanin production in response to laser-generated heat. This effect is usually temporary, but recovery time varies.4 The risk of hyperpigmentation depends largely on skin type, with darker-skinned patients (ie, Fitzgerald type III or IV skin) at higher risk.6,19 Patients at risk for hyperpigmentation should avoid sun exposure before and after laser treatments; UVA/UVB sun blocks are essential if sun exposure cannot be avoided.1
Hyperpigmentation can also be treated with hydroquinone or fractional photothermolysis.6
Paradoxical Darkening of the Tattoo
Paradoxical darkening occurs when the chemical composition of the ink is changed by laser treatment—for example, from rust-colored ferric oxide to jet black ferrous oxide. Similarly, titanium dioxide contained in white ink that is used to brighten other colors can be reduced to titanium oxide or blue Ti3+ in response to laser therapy.1 Darkening is often difficult to correct, requiring the use of several lasers, including Q-switched or ablative (eg, ultrapulse CO2, pulsed erbium:YAG) lasers.9
In order to avoid darkening, a spot test is recommended. The patient should return to the studio, if possible, and have a sample of the pigment to be removed tattooed in the axillary region. After a month, a laser test spot can be performed. If laser treatment fails, the test spot can be removed by surgical excision, and laser treatment abandoned.9
Blistering
Blistering can occur as a result of overaggressive laser treatment or inadvertent absorption of laser energy due to the specific pigment. Blisters may be avoided by using a tissue-cooling system, such as a contact chill tip or cryogen spray.4 To avoid adverse effects such as wheals, punctate bleeding, blisters, and crusts, a minimum of four weeks between sessions should be maintained. Topical antiseptics can be used to prevent infection.6
Allergic Reactions
Tattoos containing metal salts—mercury (red), cadmium (yellow), chrome (green), or cobalt (blue)—may be subject to a local allergic or photoallergic skin reaction.6 A preexisting local allergic reaction may be exacerbated by laser treatment, resulting in urticaria or a systemic allergic reaction. The tattoo should be treated with corticosteroids and an allergist consulted.1 Some providers recommend avoiding laser therapy altogether.
Red is the pigment most often associated with allergic reactions, resulting in nodular, scaly, pruritic areas.12 Removal of areas of red pigment with the 532-nm Nd:YAG laser can help prevent complications.9 Photoallergic reactions most commonly involve cadmium. Affected patients typically report a history of pruritus in the tattoo and raised skin after UV exposure.
Allergic reactions can also be treated with topical or intralesional corticosteroids.12
Scarring
Cobblestone texture is a sign of early scarring, usually appearing within two weeks of treatment. The risk for scarring is highest on the chest, outer upper arm, and ankle.1
The risk is especially great in laser treatment of areas that have been retattooed (ie, a second tattoo applied to cover an older tattoo) because of the high density of pigment and increased laser resistance.9 Patients should be asked about the possible presence of a cover-up tattoo, since this may not be detectable on casual inspection.
In a study of Chinese patients who underwent laser removal of professional blue-black tattoos,20 prophylactic use of a gel containing onion extract, heparin, and allantoin had no effect on pigment clearance, but it reduced the rate of scarring, compared with controls. Additional studies are needed to evaluate the gel’s effectiveness in patients with other skin types and with tattoos containing pigments of various colors.20
Topical steroids are sometimes helpful for scarring.1 More pronounced scarring resulting from laser tattoo removal can be treated with the erbium:YAG laser or pulsed CO2 laser, as well as fractional photothermolysis.6
Cutaneous Lymphoma
Two types of red azo dyes have been shown to generate toxic or carcinogenic decomposition products (eg, nitroaniline) under in vitro conditions; whether this occurs in vivo is unknown. Concern has been expressed that laser stimulation of lymphocytes or dendritic cells could lead to cutaneous lymphoma.6
Resistance
Certain pigments are resistant to laser treatment, and multicolored tattoos are difficult to treat because of the limited number of laser wavelengths.11 If a tattoo’s nonresponsive area exceeds 10% of its total area, laser treatment should be abandoned for financial reasons.9 A smaller resistant area, however, may be treated with ablative lasers (ultrapulse CO2, pulsed erbium:YAG ). This requires numerous sessions, one to three months apart. Aggressive measures, such as attempting to remove all of the pigment in one session, should be avoided, since heavy scarring can occur.9
Better Options on the Horizon?
Computer simulations have confirmed that laser tattoo removal is photoacoustic and that shorter pulses delivering the same amount of laser energy as longer pulses may be more efficient. According to Ho et al,21 the optimal pulse length is approximately 10 to 100 picoseconds. Thus, picosecond lasers (such as the 795-nm titanium:sapphire laser), which have been shown to be effective in removing traumatic tattoos, are being investigated for application in decorative tattoos.8,22,23 It is hoped that these lasers, with action that increases phagocytosis or transepidermal elimination, will achieve higher rates of clearance with fewer treatments, less collateral damage, and improved cosmetic outcomes. Currently, only prototypes of this laser are available for removal of decorative tattoos.1,24
Topical imiquimod 5% cream and tretinoin have been studied in conjunction with laser therapy to remove tattoos. In one animal study in which these agents were applied shortly after tattooing, pigments faded significantly, but inflammation and fibrosis occurred.25 In subsequent small studies in humans, imiquimod cream used in conjunction with Q-switched laser treatment yielded only slight improvements, compared with placebo-enhanced laser treatments.26,27 Larger studies of imiquimod and similar agents may be warranted.5
New tattoo pigments, with documented absorption characteristics within the treatable range of current Q-switched lasers, are in development. One permanent ink made of D&C and USP-grade ingredients, currently available only in black, is reported by the manufacturer to be more easily removed than conventional inks by laser therapy.28 Last year in the United States, only 13 tattoo studios used this novel tattooing pigment.
Tattoo inks in the United States are neither regulated nor approved by the FDA, and manufacturers are not required to monitor the composition of their pigments.11,29 Additionally, not all states require artists to report infections or other complications associated with a healing tattoo.
Conclusion
Primary care providers must be aware of the benefits and shortcomings of currently available laser treatments for removal of decorative tattoos. With an understanding of the numerous factors that influence the cosmetic outcome of these treatments, clinicians can help patients set realistic goals and avoid complications.
Collaborative efforts among clinicians, researchers, and laser manufacturers should lead to improvements in laser tattoo removal outcomes.
1. Mariwalla K, Dover JS. The use of lasers for decorative tattoo removal. Skin Therapy Lett. 2006;11(5):8-11.
2. Braverman PK. Body art: piercing, tattooing, and scarification. Adolesc Med Clin. 2006; 17(3):505-519.
3. Roberts TA, Ryan SA. Tattooing and high-risk behavior in adolescents. Pediatrics. 2002; 110(6):1058-1063.
4. Burris K, Kim K. Tattoo removal. Clin Dermatol. 2007;25(4):388-392.
5. Sweeney SM. Tattoos: a review of tattoo practices and potential treatment options for removal. Curr Opin Pediatr. 2006;18(4):391-395.
6. Pfirrmann G, Karsai S, Roos S, et al. Tattoo removal: state of the art. J Dtsch Dermatol Ges. 2007;5(10):889-897.
7. Reid WH, Miller ID, Murphy MJ, et al. Q-switched ruby laser treatment of tattoos: a 9-year experience. Br J Plastic Surg. 2001;43(6): 663-669.
8. Kuperman-Meade M, Levine VJ, Ashinoff R. Laser removal of tattoos. Am J Clin Dermatol. 2001;2(1):21-25.
9. Adatto MA. Laser tattoo removal: benefits and caveats. Med Laser Appl. 2004;19:175-185.
10. Anderson RR, Parrish JA. Selective photothermolysis: precise microsurgery by selective absorption of pulsed radiation. Science. 1983; 220(4596):524–527.
11. Beute TC, Miller CH, Timko AL, Ross EV. In vitro spectral analysis of tattoo pigments. Dermatol Surg. 2008;34(4):508-515.
12. Bernstein EF. Laser treatment of tattoos. Clin Dermatol. 2006;24(1):43-55.
13. Prinz BM, Vavricka SR, Graf P, et al. Efficacy of laser treatment of tattoos and using lasers emitting wavelengths of 532nm, 755nm and 1064nm. Br J Dermatol. 2004;150(2):245-251.
14. Talakoub L, Wesley NO. Differences in perceptions of beauty and cosmetic procedures performed in ethnic patients. Semin Cutan Med Surg. 2009;28(2):115-129.
15. Taylor CR, Gange WR, Dover JS, et al. Treatment of tattoos by Q-switched ruby laser. Arch Dermatol. 1990;126(7):893-899.
16. Jones A, Roddey P, Orengo I, Rosen T. The Q-switched Nd:YAG laser effectively treats tattoos in darkly pigmented skin. Dermatol Surg. 1996;22(12):999-1001.
17. Kilmer SL, Lee MS, Grevelink JM, et al. The Q-switched Nd:YAG laser effectively treats tattoos: a controlled, dose-response study. Arch Dermatol. 1993;129(8):971-978.
18. Gundogan C, Greve B, Hausser I, Raulin C. Repigmentation of persistent laser-induced hypopigmentation after tattoo ablation with the excimer laser [in German]. Hautarzt. 2004;55(6):549-552.
19. Rauscher GE, Maneckshana BT, Schwartz RA, Mekles TJ. Preoperative evaluation and management. http://emedicine.medscape.com/article/1127055-overview. Accessed August 18, 2010.
20. Ho WS, Ying SY, Chan PC, Chan HH. Use of onion extract, heparin, allantoin gel in prevention of scarring in Chinese patients having laser removal of tattoos: a prospective randomized controlled trial. Dermatol Surg. 2006; 32(7):891-896.
21. Ho DD, London R, Zimmerman GB, Young DA. Laser-tattoo removal: a study of the mechanism and the optimal treatment strategy via computer simulations. Lasers Surg Med. 2002;30(5):389-397.
22. Gorouhi F, Davari P, Kashani MN, Firooz A. Treatment of traumatic tattoo with the Q-switched Nd:YAG laser. J Cosmet Laser Ther. 2007;9(4):253-255.
23. Herd RM, Alora MB, Smoller B, et al. A clinical and histologic prospective controlled comparative study of the picosecond titanium:sapphire (795 nm) laser versus the Q-switched alexandrite (752 nm) laser for removing tattoo pigment. J Am Acad Dermatol. 1999;40(4):603-606.
24. Ross V, Naseef G, Lin G, et al. Comparison of responses of tattoos to picosecond and nanosecond Q-switched Neodymium:YAG lasers. Arch Dermatol. 1998;134(2):167-171.
25. Solis RR, Diven DG, Colome-Grimmer, et al. Experimental nonsurgical tattoo removal in a guinea pig model with topical imiquimod and tretinoin. Dermatol Surg. 2002;28(1):83-86.
26. Ricotti CA, Colaco SM, Shamma HN, et al. Laser-assisted tattoo removal with topical 5% imiquimod cream. Dermatol Surg. 2007;33(9): 1082-1091.
27. Elsaie ML, Nouri K, Vejjabhinanta V, et al. Topical imiquimod in conjunction with Nd:YAG laser for tattoo removal. Lasers Med Sci. 2009; 4(6):871-875.
28. Freedom 2™, Inc. InfinitInk™. www.infinitink.com/intro/intro.html. Accessed August 18, 2010.
29. Mayo Foundation for Medical Education and Research. Tattoos: understand risks and precautions (2010). www.mayoclinic.com/health/tattoos-and-piercings/MC00020. Accessed August 18, 2010.
Between seven and 20 million people in the United States, including adolescents as young as 12, are estimated to have at least one tattoo.1-3 Perhaps half later regret the decision to acquire a tattoo—for reasons ranging from an acute inflammatory reaction to the perception that having a tattoo might interfere with opportunities for professional advancement.4 The rising incidence of tattooing may be accompanied by increasing numbers of persons seeking to have decorative tattoos removed. Health care providers need to be aware of the modalities available, along with the risks and benefits of laser tattoo removal.
Tattoo types vary according to etiology, pigment, depth, and purpose. Cosmetic tattoos (“permanent makeup”) often serve to enhance physical features or mask scars; traumatic tattoos result from an injury in which foreign material is embedded in the skin. This article will focus on decorative tattoos and the clinical options for tattooed patients who regret these permanent markings and desire their removal.5
Decorative tattoos can be applied professionally or by amateurs, with pigment initially remaining in the superficial dermis; after several years, the pigment may migrate into deeper layers of the skin.6 Amateur tattoos are composed of ink or carbon; these pigments are usually less dense than those used by professionals, often making amateur tattoos easier to remove (ie, about five sessions of laser therapy for 90% clearance vs six to 10; see figures below).1,7
Professional tattoos are composed of organic pigments that vary in particle size but are applied at a uniform depth of needle penetration.5 The deposited pigment particles reside mainly in dermal fibroblasts and macrophages, although smaller collections of particles can be found within the interstitial space.
Tattoo Removal Techniques
Older techniques of tattoo removal, including surgical excision, salabrasion, dermabrasion, cryosurgery, and chemical peels, have largely been relinquished. Not only did these methods fail to yield desirable results, but they were associated with adverse effects, including hypopigmentation and scarring.5
Although continuous-wave lasers can also cause scarring, quality-switched (Q-switched) lasers have produced more favorable outcomes. The specific color and absorptive characteristics of each tattoo ink will help determine the ideal laser type to be used. In rare cases, patients may be able to contact the responsible artist and inquire about the inks used; information about the absorption spectrum of each pigment could facilitate the treatment plan. Even with this information, however, removal of intricate, colorful tattoos can be a challenge, since several different lasers, used in stages, may be needed.1,8
The patient’s skin type, too, may affect the response to treatment. Q-switched lasers are effective for all skin types, but complete pigment removal may be more difficult in lighter-skinned patients1 (ie, Fitzgerald skin types I and II). Similarly, in older tattoos, complete removal may not be possible, since some dermal pigment may have penetrated too deeply to be reached.5
Selective Photothermolysis
Laser removal of tattoos is accomplished by selective photothermolysis, a process that was first described by Anderson and Parrish in 1983.9,10 Photothermolysis targets specific microscopic sites on the skin, with effectiveness depending on the absorption spectrum of each pigment.4 The epidermis, dermis, and skin appendages are only minimally damaged in the process.6
Laser therapy modifies the optic properties of the tattoo pigments to be removed. The pigments absorb short laser pulses, which produce a high-intensity light in the pigments that is converted into heat.9 Shock waves shatter the pigment particles, achieving the selective death of the pigment-containing cells. The chemical composition of the pigment is also altered.
Lastly, the cell debris is phagocytized and transported to regional lymph nodes. Although they are hardly visible, some residual, scattered particles remain in the dermis. Only superficial pigment fragments are entirely eliminated during epidermal desquamation as repair is occurring, a development called transepidermal elimination.9
To produce the most effective treatment results, the laser wavelength must be absorbed by the ink, the heat should be confined to the target, and adequate energy must be delivered.1
Clinician–Patient Communication
Since the cosmetic outcome of laser therapy depends on both the laser wavelength and the absorption spectrum of each pigment, it is important for health care providers to understand the optimal wavelengths for each pigment type. They should be prepared to address with their patients the issues of incompatible lasers and resistance of certain pigments to treatment.11
Additionally, although Q-switched lasers are considered the gold standard for tattoo removal, realistic expectations should be established. Patients interested in treatment must be informed at the outset that complete clearance is not guaranteed and that the number of treatments and end result depend on factors that vary from patient to patient.4 Ten to 15 sessions, spaced six to eight weeks apart, may be required to achieve a desirable or even acceptable result,9 and the entire process could take a year or longer. The total cost can reach thousands of dollars.12
Before initiating laser therapy (or referring the patient for it), the primary care clinician should collect a history regarding the tattoo’s age and etiology, as well as the patient’s tanning habits, in order to recommend the best treatment. As stated earlier, professional tattoos generally require more treatments than amateur tattoos. Distally located tattoos are the most difficult to remove.1,13
Q-Switched Laser Types
The most common Q-switched lasers are:
• Q-switched ruby laser
• Q-switched Nd:YAG (neodymium:yttrium aluminum garnet) laser
• Q-switched alexandrite laser (see table1,4,6,14).
The choice of laser type is based on several factors, including the presumed absorption spectrum of the target, the desired depth of penetration, the size of the target particle, and the laser’s wavelength and pulse duration.11
Black and India inks absorb broadly across the spectrum. In the case of blue, yellow, or orange pigment, the optimal wavelength for pigment absorption is in an adjacent color. Green pigment absorption spectra vary due to the pigment’s multiple components. The spectra of white, yellow, and “flesh-colored” pigments do not have absorption peaks at the wavelengths of currently used Q-switched lasers; this explains their resistance to removal.11
Use of the Q-switched ruby laser (QSR) is indicated for the removal of black, blue-black, and dark blue pigments. Mixed results have been reported for removal of green and medium blue pigments, and poor results for red, orange, and pale blue. Six treatments at three-week intervals have been reported to yield clearance of 75% or greater in only about 25% of professional, dark-colored tattoos.1,15
Since this laser’s wavelength (694 nm) is absorbed by melanin, its use may result in transient hypopigmentation, depigmentation, and textural changes.1
The Q-switched Nd-YAG lasers (with wavelengths of 532 nm or 1064 nm) have a large spot size, concentrated energy densities, high repetition rates, and greater beam diameter, allowing for rapid, effective treatment of closely clustered and deep tattoos.6 Five treatments of red or orange tattoos may achieve 75% clearance in about 60% of patients.1
The 1064-nm Nd:YAG laser, which has the deepest penetration and carries the least risk for hypopigmentation,1 is indicated for black and dark blue pigments. It is considered the ideal choice for tattoo removal in dark-skinned patients,14,16 since its longer wavelength represents a lower affinity for melanin.4 The 532-nm Nd:YAG laser is effective for removal of red, yellow, and orange pigments.6 Ten or more treatments may be required for 75% clearance of a professional tattoo.1
Some adverse effects of Nd:YAG laser use include whitening of the skin, with occasional mild pinpoint bleeding. Use of the 532-nm model is associated with purpura, resulting from hemoglobin absorption; this may last from one week to 10 days. The 1064-nm Nd:YAG laser is the least effective for removing bright-colored pigments.1,17
The Q-switched alexandrite laser is generally used to remove black, blue, and green pigments. Typically, four to 10 sessions are required at intervals of one to two months. Transient hypopigmentation, typically lasting three to four months, occurs in about half of patients, and textural changes have been reported in about 12%.6,8
The 510-nm, pulsed-dye Q-switched alexandrite laser is reportedly effective in removing red pigment.4
Complications, Adverse Effects, and Their Management
Although Q-switched lasers appear quite effective in tattoo removal, their use is not without adverse effects.
Hypopigmentation
The most common chronic adverse effect of laser treatment is hypopigmentation. The risk is high in dark-skinned patients undergoing treatment with the QSR or alexandrite lasers4 and increases in any patient according to the number of treatment sessions. Hypopigmentation occurs in more than 38% of patients treated with QSR lasers and typically lasts for two to six months.6
In a 2004 study, Gundogan et al18 attempted repigmentation with an excimer laser (Nd:YAG/potassium titanyl phosphate–Nd:YAG) in a patient with hypopigmentation following laser tattoo removal. Repigmentation required 40 treatment sessions over 15 months—not a cost-effective option.6 A better solution might be to minimize the risk for hypopigmentation by use of picosecond lasers (see “Better Options on the Horizon?”, below).8
Hyperpigmentation
Hyperpigmentation can occur as a result of melanocytes’ increased melanin production in response to laser-generated heat. This effect is usually temporary, but recovery time varies.4 The risk of hyperpigmentation depends largely on skin type, with darker-skinned patients (ie, Fitzgerald type III or IV skin) at higher risk.6,19 Patients at risk for hyperpigmentation should avoid sun exposure before and after laser treatments; UVA/UVB sun blocks are essential if sun exposure cannot be avoided.1
Hyperpigmentation can also be treated with hydroquinone or fractional photothermolysis.6
Paradoxical Darkening of the Tattoo
Paradoxical darkening occurs when the chemical composition of the ink is changed by laser treatment—for example, from rust-colored ferric oxide to jet black ferrous oxide. Similarly, titanium dioxide contained in white ink that is used to brighten other colors can be reduced to titanium oxide or blue Ti3+ in response to laser therapy.1 Darkening is often difficult to correct, requiring the use of several lasers, including Q-switched or ablative (eg, ultrapulse CO2, pulsed erbium:YAG) lasers.9
In order to avoid darkening, a spot test is recommended. The patient should return to the studio, if possible, and have a sample of the pigment to be removed tattooed in the axillary region. After a month, a laser test spot can be performed. If laser treatment fails, the test spot can be removed by surgical excision, and laser treatment abandoned.9
Blistering
Blistering can occur as a result of overaggressive laser treatment or inadvertent absorption of laser energy due to the specific pigment. Blisters may be avoided by using a tissue-cooling system, such as a contact chill tip or cryogen spray.4 To avoid adverse effects such as wheals, punctate bleeding, blisters, and crusts, a minimum of four weeks between sessions should be maintained. Topical antiseptics can be used to prevent infection.6
Allergic Reactions
Tattoos containing metal salts—mercury (red), cadmium (yellow), chrome (green), or cobalt (blue)—may be subject to a local allergic or photoallergic skin reaction.6 A preexisting local allergic reaction may be exacerbated by laser treatment, resulting in urticaria or a systemic allergic reaction. The tattoo should be treated with corticosteroids and an allergist consulted.1 Some providers recommend avoiding laser therapy altogether.
Red is the pigment most often associated with allergic reactions, resulting in nodular, scaly, pruritic areas.12 Removal of areas of red pigment with the 532-nm Nd:YAG laser can help prevent complications.9 Photoallergic reactions most commonly involve cadmium. Affected patients typically report a history of pruritus in the tattoo and raised skin after UV exposure.
Allergic reactions can also be treated with topical or intralesional corticosteroids.12
Scarring
Cobblestone texture is a sign of early scarring, usually appearing within two weeks of treatment. The risk for scarring is highest on the chest, outer upper arm, and ankle.1
The risk is especially great in laser treatment of areas that have been retattooed (ie, a second tattoo applied to cover an older tattoo) because of the high density of pigment and increased laser resistance.9 Patients should be asked about the possible presence of a cover-up tattoo, since this may not be detectable on casual inspection.
In a study of Chinese patients who underwent laser removal of professional blue-black tattoos,20 prophylactic use of a gel containing onion extract, heparin, and allantoin had no effect on pigment clearance, but it reduced the rate of scarring, compared with controls. Additional studies are needed to evaluate the gel’s effectiveness in patients with other skin types and with tattoos containing pigments of various colors.20
Topical steroids are sometimes helpful for scarring.1 More pronounced scarring resulting from laser tattoo removal can be treated with the erbium:YAG laser or pulsed CO2 laser, as well as fractional photothermolysis.6
Cutaneous Lymphoma
Two types of red azo dyes have been shown to generate toxic or carcinogenic decomposition products (eg, nitroaniline) under in vitro conditions; whether this occurs in vivo is unknown. Concern has been expressed that laser stimulation of lymphocytes or dendritic cells could lead to cutaneous lymphoma.6
Resistance
Certain pigments are resistant to laser treatment, and multicolored tattoos are difficult to treat because of the limited number of laser wavelengths.11 If a tattoo’s nonresponsive area exceeds 10% of its total area, laser treatment should be abandoned for financial reasons.9 A smaller resistant area, however, may be treated with ablative lasers (ultrapulse CO2, pulsed erbium:YAG ). This requires numerous sessions, one to three months apart. Aggressive measures, such as attempting to remove all of the pigment in one session, should be avoided, since heavy scarring can occur.9
Better Options on the Horizon?
Computer simulations have confirmed that laser tattoo removal is photoacoustic and that shorter pulses delivering the same amount of laser energy as longer pulses may be more efficient. According to Ho et al,21 the optimal pulse length is approximately 10 to 100 picoseconds. Thus, picosecond lasers (such as the 795-nm titanium:sapphire laser), which have been shown to be effective in removing traumatic tattoos, are being investigated for application in decorative tattoos.8,22,23 It is hoped that these lasers, with action that increases phagocytosis or transepidermal elimination, will achieve higher rates of clearance with fewer treatments, less collateral damage, and improved cosmetic outcomes. Currently, only prototypes of this laser are available for removal of decorative tattoos.1,24
Topical imiquimod 5% cream and tretinoin have been studied in conjunction with laser therapy to remove tattoos. In one animal study in which these agents were applied shortly after tattooing, pigments faded significantly, but inflammation and fibrosis occurred.25 In subsequent small studies in humans, imiquimod cream used in conjunction with Q-switched laser treatment yielded only slight improvements, compared with placebo-enhanced laser treatments.26,27 Larger studies of imiquimod and similar agents may be warranted.5
New tattoo pigments, with documented absorption characteristics within the treatable range of current Q-switched lasers, are in development. One permanent ink made of D&C and USP-grade ingredients, currently available only in black, is reported by the manufacturer to be more easily removed than conventional inks by laser therapy.28 Last year in the United States, only 13 tattoo studios used this novel tattooing pigment.
Tattoo inks in the United States are neither regulated nor approved by the FDA, and manufacturers are not required to monitor the composition of their pigments.11,29 Additionally, not all states require artists to report infections or other complications associated with a healing tattoo.
Conclusion
Primary care providers must be aware of the benefits and shortcomings of currently available laser treatments for removal of decorative tattoos. With an understanding of the numerous factors that influence the cosmetic outcome of these treatments, clinicians can help patients set realistic goals and avoid complications.
Collaborative efforts among clinicians, researchers, and laser manufacturers should lead to improvements in laser tattoo removal outcomes.
Between seven and 20 million people in the United States, including adolescents as young as 12, are estimated to have at least one tattoo.1-3 Perhaps half later regret the decision to acquire a tattoo—for reasons ranging from an acute inflammatory reaction to the perception that having a tattoo might interfere with opportunities for professional advancement.4 The rising incidence of tattooing may be accompanied by increasing numbers of persons seeking to have decorative tattoos removed. Health care providers need to be aware of the modalities available, along with the risks and benefits of laser tattoo removal.
Tattoo types vary according to etiology, pigment, depth, and purpose. Cosmetic tattoos (“permanent makeup”) often serve to enhance physical features or mask scars; traumatic tattoos result from an injury in which foreign material is embedded in the skin. This article will focus on decorative tattoos and the clinical options for tattooed patients who regret these permanent markings and desire their removal.5
Decorative tattoos can be applied professionally or by amateurs, with pigment initially remaining in the superficial dermis; after several years, the pigment may migrate into deeper layers of the skin.6 Amateur tattoos are composed of ink or carbon; these pigments are usually less dense than those used by professionals, often making amateur tattoos easier to remove (ie, about five sessions of laser therapy for 90% clearance vs six to 10; see figures below).1,7
Professional tattoos are composed of organic pigments that vary in particle size but are applied at a uniform depth of needle penetration.5 The deposited pigment particles reside mainly in dermal fibroblasts and macrophages, although smaller collections of particles can be found within the interstitial space.
Tattoo Removal Techniques
Older techniques of tattoo removal, including surgical excision, salabrasion, dermabrasion, cryosurgery, and chemical peels, have largely been relinquished. Not only did these methods fail to yield desirable results, but they were associated with adverse effects, including hypopigmentation and scarring.5
Although continuous-wave lasers can also cause scarring, quality-switched (Q-switched) lasers have produced more favorable outcomes. The specific color and absorptive characteristics of each tattoo ink will help determine the ideal laser type to be used. In rare cases, patients may be able to contact the responsible artist and inquire about the inks used; information about the absorption spectrum of each pigment could facilitate the treatment plan. Even with this information, however, removal of intricate, colorful tattoos can be a challenge, since several different lasers, used in stages, may be needed.1,8
The patient’s skin type, too, may affect the response to treatment. Q-switched lasers are effective for all skin types, but complete pigment removal may be more difficult in lighter-skinned patients1 (ie, Fitzgerald skin types I and II). Similarly, in older tattoos, complete removal may not be possible, since some dermal pigment may have penetrated too deeply to be reached.5
Selective Photothermolysis
Laser removal of tattoos is accomplished by selective photothermolysis, a process that was first described by Anderson and Parrish in 1983.9,10 Photothermolysis targets specific microscopic sites on the skin, with effectiveness depending on the absorption spectrum of each pigment.4 The epidermis, dermis, and skin appendages are only minimally damaged in the process.6
Laser therapy modifies the optic properties of the tattoo pigments to be removed. The pigments absorb short laser pulses, which produce a high-intensity light in the pigments that is converted into heat.9 Shock waves shatter the pigment particles, achieving the selective death of the pigment-containing cells. The chemical composition of the pigment is also altered.
Lastly, the cell debris is phagocytized and transported to regional lymph nodes. Although they are hardly visible, some residual, scattered particles remain in the dermis. Only superficial pigment fragments are entirely eliminated during epidermal desquamation as repair is occurring, a development called transepidermal elimination.9
To produce the most effective treatment results, the laser wavelength must be absorbed by the ink, the heat should be confined to the target, and adequate energy must be delivered.1
Clinician–Patient Communication
Since the cosmetic outcome of laser therapy depends on both the laser wavelength and the absorption spectrum of each pigment, it is important for health care providers to understand the optimal wavelengths for each pigment type. They should be prepared to address with their patients the issues of incompatible lasers and resistance of certain pigments to treatment.11
Additionally, although Q-switched lasers are considered the gold standard for tattoo removal, realistic expectations should be established. Patients interested in treatment must be informed at the outset that complete clearance is not guaranteed and that the number of treatments and end result depend on factors that vary from patient to patient.4 Ten to 15 sessions, spaced six to eight weeks apart, may be required to achieve a desirable or even acceptable result,9 and the entire process could take a year or longer. The total cost can reach thousands of dollars.12
Before initiating laser therapy (or referring the patient for it), the primary care clinician should collect a history regarding the tattoo’s age and etiology, as well as the patient’s tanning habits, in order to recommend the best treatment. As stated earlier, professional tattoos generally require more treatments than amateur tattoos. Distally located tattoos are the most difficult to remove.1,13
Q-Switched Laser Types
The most common Q-switched lasers are:
• Q-switched ruby laser
• Q-switched Nd:YAG (neodymium:yttrium aluminum garnet) laser
• Q-switched alexandrite laser (see table1,4,6,14).
The choice of laser type is based on several factors, including the presumed absorption spectrum of the target, the desired depth of penetration, the size of the target particle, and the laser’s wavelength and pulse duration.11
Black and India inks absorb broadly across the spectrum. In the case of blue, yellow, or orange pigment, the optimal wavelength for pigment absorption is in an adjacent color. Green pigment absorption spectra vary due to the pigment’s multiple components. The spectra of white, yellow, and “flesh-colored” pigments do not have absorption peaks at the wavelengths of currently used Q-switched lasers; this explains their resistance to removal.11
Use of the Q-switched ruby laser (QSR) is indicated for the removal of black, blue-black, and dark blue pigments. Mixed results have been reported for removal of green and medium blue pigments, and poor results for red, orange, and pale blue. Six treatments at three-week intervals have been reported to yield clearance of 75% or greater in only about 25% of professional, dark-colored tattoos.1,15
Since this laser’s wavelength (694 nm) is absorbed by melanin, its use may result in transient hypopigmentation, depigmentation, and textural changes.1
The Q-switched Nd-YAG lasers (with wavelengths of 532 nm or 1064 nm) have a large spot size, concentrated energy densities, high repetition rates, and greater beam diameter, allowing for rapid, effective treatment of closely clustered and deep tattoos.6 Five treatments of red or orange tattoos may achieve 75% clearance in about 60% of patients.1
The 1064-nm Nd:YAG laser, which has the deepest penetration and carries the least risk for hypopigmentation,1 is indicated for black and dark blue pigments. It is considered the ideal choice for tattoo removal in dark-skinned patients,14,16 since its longer wavelength represents a lower affinity for melanin.4 The 532-nm Nd:YAG laser is effective for removal of red, yellow, and orange pigments.6 Ten or more treatments may be required for 75% clearance of a professional tattoo.1
Some adverse effects of Nd:YAG laser use include whitening of the skin, with occasional mild pinpoint bleeding. Use of the 532-nm model is associated with purpura, resulting from hemoglobin absorption; this may last from one week to 10 days. The 1064-nm Nd:YAG laser is the least effective for removing bright-colored pigments.1,17
The Q-switched alexandrite laser is generally used to remove black, blue, and green pigments. Typically, four to 10 sessions are required at intervals of one to two months. Transient hypopigmentation, typically lasting three to four months, occurs in about half of patients, and textural changes have been reported in about 12%.6,8
The 510-nm, pulsed-dye Q-switched alexandrite laser is reportedly effective in removing red pigment.4
Complications, Adverse Effects, and Their Management
Although Q-switched lasers appear quite effective in tattoo removal, their use is not without adverse effects.
Hypopigmentation
The most common chronic adverse effect of laser treatment is hypopigmentation. The risk is high in dark-skinned patients undergoing treatment with the QSR or alexandrite lasers4 and increases in any patient according to the number of treatment sessions. Hypopigmentation occurs in more than 38% of patients treated with QSR lasers and typically lasts for two to six months.6
In a 2004 study, Gundogan et al18 attempted repigmentation with an excimer laser (Nd:YAG/potassium titanyl phosphate–Nd:YAG) in a patient with hypopigmentation following laser tattoo removal. Repigmentation required 40 treatment sessions over 15 months—not a cost-effective option.6 A better solution might be to minimize the risk for hypopigmentation by use of picosecond lasers (see “Better Options on the Horizon?”, below).8
Hyperpigmentation
Hyperpigmentation can occur as a result of melanocytes’ increased melanin production in response to laser-generated heat. This effect is usually temporary, but recovery time varies.4 The risk of hyperpigmentation depends largely on skin type, with darker-skinned patients (ie, Fitzgerald type III or IV skin) at higher risk.6,19 Patients at risk for hyperpigmentation should avoid sun exposure before and after laser treatments; UVA/UVB sun blocks are essential if sun exposure cannot be avoided.1
Hyperpigmentation can also be treated with hydroquinone or fractional photothermolysis.6
Paradoxical Darkening of the Tattoo
Paradoxical darkening occurs when the chemical composition of the ink is changed by laser treatment—for example, from rust-colored ferric oxide to jet black ferrous oxide. Similarly, titanium dioxide contained in white ink that is used to brighten other colors can be reduced to titanium oxide or blue Ti3+ in response to laser therapy.1 Darkening is often difficult to correct, requiring the use of several lasers, including Q-switched or ablative (eg, ultrapulse CO2, pulsed erbium:YAG) lasers.9
In order to avoid darkening, a spot test is recommended. The patient should return to the studio, if possible, and have a sample of the pigment to be removed tattooed in the axillary region. After a month, a laser test spot can be performed. If laser treatment fails, the test spot can be removed by surgical excision, and laser treatment abandoned.9
Blistering
Blistering can occur as a result of overaggressive laser treatment or inadvertent absorption of laser energy due to the specific pigment. Blisters may be avoided by using a tissue-cooling system, such as a contact chill tip or cryogen spray.4 To avoid adverse effects such as wheals, punctate bleeding, blisters, and crusts, a minimum of four weeks between sessions should be maintained. Topical antiseptics can be used to prevent infection.6
Allergic Reactions
Tattoos containing metal salts—mercury (red), cadmium (yellow), chrome (green), or cobalt (blue)—may be subject to a local allergic or photoallergic skin reaction.6 A preexisting local allergic reaction may be exacerbated by laser treatment, resulting in urticaria or a systemic allergic reaction. The tattoo should be treated with corticosteroids and an allergist consulted.1 Some providers recommend avoiding laser therapy altogether.
Red is the pigment most often associated with allergic reactions, resulting in nodular, scaly, pruritic areas.12 Removal of areas of red pigment with the 532-nm Nd:YAG laser can help prevent complications.9 Photoallergic reactions most commonly involve cadmium. Affected patients typically report a history of pruritus in the tattoo and raised skin after UV exposure.
Allergic reactions can also be treated with topical or intralesional corticosteroids.12
Scarring
Cobblestone texture is a sign of early scarring, usually appearing within two weeks of treatment. The risk for scarring is highest on the chest, outer upper arm, and ankle.1
The risk is especially great in laser treatment of areas that have been retattooed (ie, a second tattoo applied to cover an older tattoo) because of the high density of pigment and increased laser resistance.9 Patients should be asked about the possible presence of a cover-up tattoo, since this may not be detectable on casual inspection.
In a study of Chinese patients who underwent laser removal of professional blue-black tattoos,20 prophylactic use of a gel containing onion extract, heparin, and allantoin had no effect on pigment clearance, but it reduced the rate of scarring, compared with controls. Additional studies are needed to evaluate the gel’s effectiveness in patients with other skin types and with tattoos containing pigments of various colors.20
Topical steroids are sometimes helpful for scarring.1 More pronounced scarring resulting from laser tattoo removal can be treated with the erbium:YAG laser or pulsed CO2 laser, as well as fractional photothermolysis.6
Cutaneous Lymphoma
Two types of red azo dyes have been shown to generate toxic or carcinogenic decomposition products (eg, nitroaniline) under in vitro conditions; whether this occurs in vivo is unknown. Concern has been expressed that laser stimulation of lymphocytes or dendritic cells could lead to cutaneous lymphoma.6
Resistance
Certain pigments are resistant to laser treatment, and multicolored tattoos are difficult to treat because of the limited number of laser wavelengths.11 If a tattoo’s nonresponsive area exceeds 10% of its total area, laser treatment should be abandoned for financial reasons.9 A smaller resistant area, however, may be treated with ablative lasers (ultrapulse CO2, pulsed erbium:YAG ). This requires numerous sessions, one to three months apart. Aggressive measures, such as attempting to remove all of the pigment in one session, should be avoided, since heavy scarring can occur.9
Better Options on the Horizon?
Computer simulations have confirmed that laser tattoo removal is photoacoustic and that shorter pulses delivering the same amount of laser energy as longer pulses may be more efficient. According to Ho et al,21 the optimal pulse length is approximately 10 to 100 picoseconds. Thus, picosecond lasers (such as the 795-nm titanium:sapphire laser), which have been shown to be effective in removing traumatic tattoos, are being investigated for application in decorative tattoos.8,22,23 It is hoped that these lasers, with action that increases phagocytosis or transepidermal elimination, will achieve higher rates of clearance with fewer treatments, less collateral damage, and improved cosmetic outcomes. Currently, only prototypes of this laser are available for removal of decorative tattoos.1,24
Topical imiquimod 5% cream and tretinoin have been studied in conjunction with laser therapy to remove tattoos. In one animal study in which these agents were applied shortly after tattooing, pigments faded significantly, but inflammation and fibrosis occurred.25 In subsequent small studies in humans, imiquimod cream used in conjunction with Q-switched laser treatment yielded only slight improvements, compared with placebo-enhanced laser treatments.26,27 Larger studies of imiquimod and similar agents may be warranted.5
New tattoo pigments, with documented absorption characteristics within the treatable range of current Q-switched lasers, are in development. One permanent ink made of D&C and USP-grade ingredients, currently available only in black, is reported by the manufacturer to be more easily removed than conventional inks by laser therapy.28 Last year in the United States, only 13 tattoo studios used this novel tattooing pigment.
Tattoo inks in the United States are neither regulated nor approved by the FDA, and manufacturers are not required to monitor the composition of their pigments.11,29 Additionally, not all states require artists to report infections or other complications associated with a healing tattoo.
Conclusion
Primary care providers must be aware of the benefits and shortcomings of currently available laser treatments for removal of decorative tattoos. With an understanding of the numerous factors that influence the cosmetic outcome of these treatments, clinicians can help patients set realistic goals and avoid complications.
Collaborative efforts among clinicians, researchers, and laser manufacturers should lead to improvements in laser tattoo removal outcomes.
1. Mariwalla K, Dover JS. The use of lasers for decorative tattoo removal. Skin Therapy Lett. 2006;11(5):8-11.
2. Braverman PK. Body art: piercing, tattooing, and scarification. Adolesc Med Clin. 2006; 17(3):505-519.
3. Roberts TA, Ryan SA. Tattooing and high-risk behavior in adolescents. Pediatrics. 2002; 110(6):1058-1063.
4. Burris K, Kim K. Tattoo removal. Clin Dermatol. 2007;25(4):388-392.
5. Sweeney SM. Tattoos: a review of tattoo practices and potential treatment options for removal. Curr Opin Pediatr. 2006;18(4):391-395.
6. Pfirrmann G, Karsai S, Roos S, et al. Tattoo removal: state of the art. J Dtsch Dermatol Ges. 2007;5(10):889-897.
7. Reid WH, Miller ID, Murphy MJ, et al. Q-switched ruby laser treatment of tattoos: a 9-year experience. Br J Plastic Surg. 2001;43(6): 663-669.
8. Kuperman-Meade M, Levine VJ, Ashinoff R. Laser removal of tattoos. Am J Clin Dermatol. 2001;2(1):21-25.
9. Adatto MA. Laser tattoo removal: benefits and caveats. Med Laser Appl. 2004;19:175-185.
10. Anderson RR, Parrish JA. Selective photothermolysis: precise microsurgery by selective absorption of pulsed radiation. Science. 1983; 220(4596):524–527.
11. Beute TC, Miller CH, Timko AL, Ross EV. In vitro spectral analysis of tattoo pigments. Dermatol Surg. 2008;34(4):508-515.
12. Bernstein EF. Laser treatment of tattoos. Clin Dermatol. 2006;24(1):43-55.
13. Prinz BM, Vavricka SR, Graf P, et al. Efficacy of laser treatment of tattoos and using lasers emitting wavelengths of 532nm, 755nm and 1064nm. Br J Dermatol. 2004;150(2):245-251.
14. Talakoub L, Wesley NO. Differences in perceptions of beauty and cosmetic procedures performed in ethnic patients. Semin Cutan Med Surg. 2009;28(2):115-129.
15. Taylor CR, Gange WR, Dover JS, et al. Treatment of tattoos by Q-switched ruby laser. Arch Dermatol. 1990;126(7):893-899.
16. Jones A, Roddey P, Orengo I, Rosen T. The Q-switched Nd:YAG laser effectively treats tattoos in darkly pigmented skin. Dermatol Surg. 1996;22(12):999-1001.
17. Kilmer SL, Lee MS, Grevelink JM, et al. The Q-switched Nd:YAG laser effectively treats tattoos: a controlled, dose-response study. Arch Dermatol. 1993;129(8):971-978.
18. Gundogan C, Greve B, Hausser I, Raulin C. Repigmentation of persistent laser-induced hypopigmentation after tattoo ablation with the excimer laser [in German]. Hautarzt. 2004;55(6):549-552.
19. Rauscher GE, Maneckshana BT, Schwartz RA, Mekles TJ. Preoperative evaluation and management. http://emedicine.medscape.com/article/1127055-overview. Accessed August 18, 2010.
20. Ho WS, Ying SY, Chan PC, Chan HH. Use of onion extract, heparin, allantoin gel in prevention of scarring in Chinese patients having laser removal of tattoos: a prospective randomized controlled trial. Dermatol Surg. 2006; 32(7):891-896.
21. Ho DD, London R, Zimmerman GB, Young DA. Laser-tattoo removal: a study of the mechanism and the optimal treatment strategy via computer simulations. Lasers Surg Med. 2002;30(5):389-397.
22. Gorouhi F, Davari P, Kashani MN, Firooz A. Treatment of traumatic tattoo with the Q-switched Nd:YAG laser. J Cosmet Laser Ther. 2007;9(4):253-255.
23. Herd RM, Alora MB, Smoller B, et al. A clinical and histologic prospective controlled comparative study of the picosecond titanium:sapphire (795 nm) laser versus the Q-switched alexandrite (752 nm) laser for removing tattoo pigment. J Am Acad Dermatol. 1999;40(4):603-606.
24. Ross V, Naseef G, Lin G, et al. Comparison of responses of tattoos to picosecond and nanosecond Q-switched Neodymium:YAG lasers. Arch Dermatol. 1998;134(2):167-171.
25. Solis RR, Diven DG, Colome-Grimmer, et al. Experimental nonsurgical tattoo removal in a guinea pig model with topical imiquimod and tretinoin. Dermatol Surg. 2002;28(1):83-86.
26. Ricotti CA, Colaco SM, Shamma HN, et al. Laser-assisted tattoo removal with topical 5% imiquimod cream. Dermatol Surg. 2007;33(9): 1082-1091.
27. Elsaie ML, Nouri K, Vejjabhinanta V, et al. Topical imiquimod in conjunction with Nd:YAG laser for tattoo removal. Lasers Med Sci. 2009; 4(6):871-875.
28. Freedom 2™, Inc. InfinitInk™. www.infinitink.com/intro/intro.html. Accessed August 18, 2010.
29. Mayo Foundation for Medical Education and Research. Tattoos: understand risks and precautions (2010). www.mayoclinic.com/health/tattoos-and-piercings/MC00020. Accessed August 18, 2010.
1. Mariwalla K, Dover JS. The use of lasers for decorative tattoo removal. Skin Therapy Lett. 2006;11(5):8-11.
2. Braverman PK. Body art: piercing, tattooing, and scarification. Adolesc Med Clin. 2006; 17(3):505-519.
3. Roberts TA, Ryan SA. Tattooing and high-risk behavior in adolescents. Pediatrics. 2002; 110(6):1058-1063.
4. Burris K, Kim K. Tattoo removal. Clin Dermatol. 2007;25(4):388-392.
5. Sweeney SM. Tattoos: a review of tattoo practices and potential treatment options for removal. Curr Opin Pediatr. 2006;18(4):391-395.
6. Pfirrmann G, Karsai S, Roos S, et al. Tattoo removal: state of the art. J Dtsch Dermatol Ges. 2007;5(10):889-897.
7. Reid WH, Miller ID, Murphy MJ, et al. Q-switched ruby laser treatment of tattoos: a 9-year experience. Br J Plastic Surg. 2001;43(6): 663-669.
8. Kuperman-Meade M, Levine VJ, Ashinoff R. Laser removal of tattoos. Am J Clin Dermatol. 2001;2(1):21-25.
9. Adatto MA. Laser tattoo removal: benefits and caveats. Med Laser Appl. 2004;19:175-185.
10. Anderson RR, Parrish JA. Selective photothermolysis: precise microsurgery by selective absorption of pulsed radiation. Science. 1983; 220(4596):524–527.
11. Beute TC, Miller CH, Timko AL, Ross EV. In vitro spectral analysis of tattoo pigments. Dermatol Surg. 2008;34(4):508-515.
12. Bernstein EF. Laser treatment of tattoos. Clin Dermatol. 2006;24(1):43-55.
13. Prinz BM, Vavricka SR, Graf P, et al. Efficacy of laser treatment of tattoos and using lasers emitting wavelengths of 532nm, 755nm and 1064nm. Br J Dermatol. 2004;150(2):245-251.
14. Talakoub L, Wesley NO. Differences in perceptions of beauty and cosmetic procedures performed in ethnic patients. Semin Cutan Med Surg. 2009;28(2):115-129.
15. Taylor CR, Gange WR, Dover JS, et al. Treatment of tattoos by Q-switched ruby laser. Arch Dermatol. 1990;126(7):893-899.
16. Jones A, Roddey P, Orengo I, Rosen T. The Q-switched Nd:YAG laser effectively treats tattoos in darkly pigmented skin. Dermatol Surg. 1996;22(12):999-1001.
17. Kilmer SL, Lee MS, Grevelink JM, et al. The Q-switched Nd:YAG laser effectively treats tattoos: a controlled, dose-response study. Arch Dermatol. 1993;129(8):971-978.
18. Gundogan C, Greve B, Hausser I, Raulin C. Repigmentation of persistent laser-induced hypopigmentation after tattoo ablation with the excimer laser [in German]. Hautarzt. 2004;55(6):549-552.
19. Rauscher GE, Maneckshana BT, Schwartz RA, Mekles TJ. Preoperative evaluation and management. http://emedicine.medscape.com/article/1127055-overview. Accessed August 18, 2010.
20. Ho WS, Ying SY, Chan PC, Chan HH. Use of onion extract, heparin, allantoin gel in prevention of scarring in Chinese patients having laser removal of tattoos: a prospective randomized controlled trial. Dermatol Surg. 2006; 32(7):891-896.
21. Ho DD, London R, Zimmerman GB, Young DA. Laser-tattoo removal: a study of the mechanism and the optimal treatment strategy via computer simulations. Lasers Surg Med. 2002;30(5):389-397.
22. Gorouhi F, Davari P, Kashani MN, Firooz A. Treatment of traumatic tattoo with the Q-switched Nd:YAG laser. J Cosmet Laser Ther. 2007;9(4):253-255.
23. Herd RM, Alora MB, Smoller B, et al. A clinical and histologic prospective controlled comparative study of the picosecond titanium:sapphire (795 nm) laser versus the Q-switched alexandrite (752 nm) laser for removing tattoo pigment. J Am Acad Dermatol. 1999;40(4):603-606.
24. Ross V, Naseef G, Lin G, et al. Comparison of responses of tattoos to picosecond and nanosecond Q-switched Neodymium:YAG lasers. Arch Dermatol. 1998;134(2):167-171.
25. Solis RR, Diven DG, Colome-Grimmer, et al. Experimental nonsurgical tattoo removal in a guinea pig model with topical imiquimod and tretinoin. Dermatol Surg. 2002;28(1):83-86.
26. Ricotti CA, Colaco SM, Shamma HN, et al. Laser-assisted tattoo removal with topical 5% imiquimod cream. Dermatol Surg. 2007;33(9): 1082-1091.
27. Elsaie ML, Nouri K, Vejjabhinanta V, et al. Topical imiquimod in conjunction with Nd:YAG laser for tattoo removal. Lasers Med Sci. 2009; 4(6):871-875.
28. Freedom 2™, Inc. InfinitInk™. www.infinitink.com/intro/intro.html. Accessed August 18, 2010.
29. Mayo Foundation for Medical Education and Research. Tattoos: understand risks and precautions (2010). www.mayoclinic.com/health/tattoos-and-piercings/MC00020. Accessed August 18, 2010.
Dental Emergencies
Management of Elderly Patients with Hip Fractures and Cardiac Rhythm Devices
UPDATE: SEXUAL DYSFUNCTION
Since sildenafil (Viagra) was approved by the US Food and Drug Administration to treat erectile dysfunction, women have been calling for research and development of treatments for female sexual dysfunction.
Despite considerable research documenting improvement in sexual responsiveness, genital sensation, and overall well-being among women who were given testosterone after undergoing bilateral oophorectomy, there remains only one testosterone formulation for women. A combination of synthetic estrogen and methyl testosterone (Estratest; Abbott) is indicated for management of moderate to severe vasomotor symptoms associated with menopause in patients who do not respond to estrogens alone.
In the testing stage from BioSante is LibiGel, a transdermal testosterone product. Acrux is developing Luramist, a daily testosterone spray. Proctor & Gamble’s efforts to gain approval of a testosterone-containing transdermal patch (Intrinsa) for treatment of low libido were unsuccessful, largely because of concern about potential increases in the risks of coronary artery disease and breast cancer. Pivotal trial data did not demonstrate enhanced risk, but the numbers were too small and the timeframe too short (a maximum follow-up of 2 years) to establish an effect, so the FDA asked for long-term studies. In 2006, European regulators approved Intrinsa to treat low sexual desire in surgically menopausal women.
Then there’s flibanserin, which also failed to win approval from an FDA advisory committee after numerous concerns were raised about its safety and efficacy in premenopausal women.
The lack of approved drugs leaves gynecologists and women’s health providers with little to offer our patients who are distressed by sexual dysfunction.
In this Update, I discuss:
- the complexity of female sexual function
- what derailed flibanserin
- recent findings that suggest dehydroepiandrosterone (DHEA) may be beneficial
- recommendations for clinical practice.
As understanding of female sexual dysfunction evolves, so do its labels
The fourth edition of the Diagnostic and Statistical Manual of Mental Disorders (DSM-IV) divides female sexual dysfunction into four categories:
- hypoactive sexual desire disorder (HSDD)—a persistent or recurrent deficiency or absence of sexual fantasies and desire for sexual activity
- female sexual arousal disorder—a persistent or recurrent inability to achieve or maintain adequate vaginal lubrication or vulvar swelling (i.e., sexual excitement)
- female orgasmic disorder—persistent or recurrent delay in or absence of orgasm following a normal sexual excitement phase
- dyspareunia—persistent or recurrent genital pain that is associated with sexual intercourse.
These categories were revised in 2003 by an international consensus committee sponsored by the American Urological Association Foundation; arousal disorder has been subdivided into:
- combined arousal disorder—absent feelings of sexual arousal from any type of stimulation, as well as absent or impaired genital sexual arousal (vulvar swelling and vaginal lubrication)
- subjective arousal disorder—absent feelings of sexual excitement and pleasure from any type of stimulation in the presence of genital sexual arousal (vulvar swelling and vaginal lubrication)
- genital arousal disorder—subjective sexual excitement from nongenital sexual stimuli with reduced sensation from genital touching and an absence of genital sexual arousal from any type of sexual stimulation.
These updated definitions will be incorporated into DSM-V, to be published in 2013.
Also likely to change in DSM-V: HSDD and female sexual arousal disorder may be subsumed into a new category, “sexual interest/arousal disorder in women”.1
The female response to sexual stimuli is complex
The complexity of sexual arousal disorders in women complicates research into the pathophysiology and potential pharmacologic treatment of these conditions. Conflicting evidence for any benefit of the phosphodiesterase type-5 (PDE5) inhibitors, such as sildenafil, in the treatment of sexual dysfunction in women likely arises from a lack of precision in defining the conditions in which and patients for whom these interventions are appropriate.
Functional magnetic resonance imaging (MRI) studies of men and women reveal differences in areas of brain activity related to sexual arousal. The neurophysiology of sexual desire and response is complex, involving multiple neurotransmitters, peptides, and hormones as well as multiple structural regions within the brain. Dopamine, norepinephrine, melanocortin, oxytocin, and serotonin (at some of its receptors) promote sexual activity, whereas prolactin, gamma amino butyric acid (GABA), and serotonin (at most of its receptors) are inhibitory.
In animal studies, both an increase in dopamine and a change in social environment can trigger increased sexual behavior. In women, a dopaminergic drug such as buproprion may increase arousability and pleasure—but so can a new partner.
All these bits of the “big picture” continue to complicate research in female sexual function.
It is imperative that we begin to understand the nuances of our patients’ sexual problems if we are to offer effective suggestions for treatment and management. Objectively determined genital arousal disorder very likely derives from neurovascular causes and is likely to respond to PDE5 inhibitors, but subjective arousal disorder with normal vulvar and vaginal engorgement and lubrication is not likely to respond to these agents.
This is the state of our basic science knowledge in 2010. What’s out there and on the horizon for us to offer our patients?
Flibanserin gets an unequivocal thumbs down
Phase-3 Trial 511.71. A twenty-four week, randomized, double-blind, placebo-controlled, safety and efficacy trial of flibanserin 50 milligrams every evening and flibanserin 100 mg every evening in women with hypoactive sexual desire disorder in North America. NCT00360529.
Phase-3 Trial 511.75. Best tolerability: 50 mg twice daily versus 100 mg in the evening versus 25 mg twice daily versus placebo in younger women in North America. NCT00360555.
Flibanserin is a 5HT 1A agonist, 2A antagonist, and weak dopamine agonist. It was originally studied as a treatment for major depressive disorder. In phase-2 trials, it was ineffective for management of depression but superior to placebo and an active comparator in improving sex drive (based on validated questionnaires). These results formed the basis for studying flibanserin as a treatment for HSDD. More than 5,000 women have been involved in phase-2 and phase-3 trials in the United States, Canada, and Europe.
Following FDA guidance for sponsors developing treatments for HSDD, drug maker Boehringer Ingelheim defined the primary endpoints for the pivotal trials as an increase in the number of sexually satisfying events (SSEs) and sexual desire, as measured by a daily diary. Sexual events included:
- genital touching by the partner
- masturbation
- oral sex
- intercourse
- orgasm.
Sexual desire was rated daily by the participants using an eDiary.
In North American phase-3 trials, 2,462 premenopausal women with acquired HSDD in stable, monogamous, functionally heterosexual, communicative relationships for at least 1 year were enrolled. Comorbid arousal and orgasmic disorders were allowed if they were secondary to decreased desire. Mean age of the participants was 35 to 36 years, and they were predominantly white, highly educated women in long-term relationships.
Two important exclusions worth noting:
- women who had depression, breast or other cancers (except skin cancer), or any major medical condition
- women who were taking any of the medications on a five-page list of excluded drugs (due to metabolism with the enzyme cytochrome P3A4).
Once they were screened, women completed a 4-week baseline assessment of sexual activity and desire, followed by a 24-week study period. They were randomized to receive 50 mg or 100 mg of flibanserin or placebo daily. Improvements in sexual function, compared with the 4-week baseline, had to be both statistically and clinically significant for the studies to be successful.
Flibanserin’s effects were clinically unimpressive
At a daily dosage of 100 mg, flibanserin was associated with a significant increase in the number of SSEs, compared with placebo. However, the co-primary endpoint of an increase in desire, as assessed by the eDiary, was not achieved in the active treatment group. Women who took flibanserin had a response rate of 30% to 40%, compared with 15% to 30% for women who took placebo. Although this difference was significant, it was clinically unimpressive, with fewer than 50% of participants experiencing significant improvement (TABLE 1).
TABLE 1
Flibanserin increased the mean number of sexually satisfying events—but improvement was modest
| Phase-3 trial | Measure | Placebo | Flibanserin | P value |
|---|---|---|---|---|
| 511.71 | Baseline | 2.7 | 3.0 | |
| 24 weeks | 3.5 | 4.6 | .005 | |
| 511.75 | Baseline | 2.7 | 2.6 | |
| 24 weeks | 2.6 | 4.4 | .024 |
Frequency of side effects is troubling
Among women who took the active drug, 34.6% discontinued the medication because of side effects, compared with 6.8% of women who took placebo. Most common side effects (and their incidence) were:
- nausea (12%)
- dizziness (11%)
- fatigue (11%)
- daytime somnolence (9.5%)
- anxiety (2%).
In the healthy study population, no major safety issues were associated with flibanserin. However, concomitant use of alcohol, a selective serotonin reuptake inhibitor (SSRI) or serotonin-norepinephrine reuptake inhibitor (SNRI), or a triptan was associated with a marked increase in side effects.
In addition, women who were predisposed to depression or suicidal ideation were more likely to develop suicidal tendencies while taking flibanserin, compared with placebo.
Given the long list of prohibited medications (CYP3A4 promoters or inhibitors), many of which are in widespread use, and given the lack of pharmacodynamic assessment by the sponsor of circulating levels of active drug if used with any of these medications, FDA reviewers and advisory panel members grew concerned about the potential for major side effects if flibanserin were to be released for commercial use.
Ultimately, the FDA advisory panel voted unanimously to withhold approval of flibanserin for treatment of HSDD in premenopausal women but encouraged the company to continue studies in postmenopausal women. Recruitment is under way for NCT00996372.
Attendees who spoke during the open public hearing on flibanserin, held June 8 in Gaithersburg, Md, had mixed opinions of the drug’s utility in premenopausal women with hypoactive sexual desire disorder (HSDD). Some believe approval of a “female Viagra” is long overdue. Others, not so much.
“In 1920, women were given the right to vote,” said Sue Goldstein, clinical trial coordinator at San Diego Sexual Medicine in San Diego, Calif. “What we’re asking now is that women be empowered again, that they be given the right to choose to be treated with an FDA-approved product for HSDD.” She added: “Once again, we are the forgotten gender.”
Leonore Tiefer, PhD, sees things differently.
“The simple but appealing notion that a new brain drug can help you with [hypoactive sexual desire disorder] because, well, desire is in the brain, has been peddled for the past year as if it were a fancy pair of shoes,” said Tiefer, clinical associate professor of psychiatry at New York University School of Medicine. “Flibanserin is not a choice when it’s promoted by bad science and half-truths and when self-diagnosis checklists are passed off as medical care.” Her last comment was a reference to sexbrainbody.com, a Web site sponsored by Boehringer Ingelheim that offers, among other resources, a “sexual satisfaction checklist.”
Many other attendees had a personal interest in the issue or represented advocacy groups, and their responses were just as mixed.
Michelle King Robson, founder and CEO of Empow-Her, a women’s health media company, favored approval of flibanserin.
“Women are struggling to find solutions to their sexual dysfunction,” she said.
Liz Canner, director of the feature documentary film Orgasm, Inc., which takes as its subject “the strange science of female pleasure,” accused Boehringer Ingelheim of “disease mongering” (and held a viewing of her film in a room down the hall).
One of the last to speak was Amy Allina, program director of the National Women’s Health Network, who asserted that flibanserin offers “little benefit for real women in the real world.”
There may one day be an effective agent for HSDD, she said.
“This drug is not it,” she added.—Janelle Yates, Senior Editor
Intravaginal DHEA improves postmenopausal sexual function
Labrie F, Archer D, Bouchard C, et al. Effect of intravaginal dehydroepiandrosterone (Prasterone) on libido and sexual dysfunction in postmenopausal women. Menopause. 2009;16(5):923–931.
DHEA has been studied as a treatment for female sexual dysfunction in postmenopausal women, in whom it acts as a precursor for both estrogen and androgen synthesis. In this study by Labrie and colleagues, all aspects of female sexual function—desire, arousal, orgasm, and pain—improved significantly with intravaginal DHEA (TABLE 2).
This phase-3, multicenter, placebo-controlled, randomized clinical trial of 216 participants—50 in each arm—randomized women to placebo or 3.25 mg, 6.5 mg, or 13 mg of DHEA daily. Median age of participants was 58.
The study began with a 4-week baseline screening phase, followed by 12 weeks of placebo or active treatment. Women were enrolled if they were postmenopausal and experienced vaginal dryness or vulvar or vaginal irritation or pain. Most women had moderate to severe symptoms.
Topical or systemic hormone therapy was prohibited, and women who had preexisting cancer (except skin cancer) or endometrial hyperplasia were excluded. The primary endpoint was improvement in the four domains of sexual dysfunction.
Although women were not initially selected for this trial based on measures of personal distress related to their sexual dysfunction (marked distress or interpersonal difficulty is required by DSM-IV for a diagnosis), approximately 50% indicated a desire for improvement on the intake questionnaire. Women were not excluded from this study if they were taking other medications known to affect sexual function, with the exception of systemic or topical hormonal treatment.
The robust results in this study were achieved without increasing circulating levels of estrogen, testosterone, or DHEA beyond the normal postmenopausal range.
TABLE 2
Women using intravaginal DHEA experienced improvement in all four domains of female sexual function
| Domain | Improvement after 12 weeks of 1% DHEA | P value |
|---|---|---|
| Desire | 49% | <.0001 |
| Arousal | 68% | .0004 |
| Orgasm | 75% | <.0001 |
| Dryness (pain) | 57% | .0001 |
What can we offer to our patients?
Female sexual dysfunction is more difficult to categorize and certainly more difficult to measure scientifically than male sexual dysfunction. The distinction between desire, arousal, and pain disorders in women is easily blurred. Certainly, the ability to declare success in clinical trials is straightforward and unequivocal for men. Not so for women.
Female sexual dysfunction that causes distress for our patients is not uncommon. It is a source of frustration for our patients and for us as providers. For now, in the absence of a “little pink pill,” we can offer:
- techniques to eliminate pain such as topical estrogen for atrophy and physical therapy and biofeedback for secondary vaginismus
- adjustment of medications that may thwart sexual desire, arousal, or orgasm, such as SSRIs and antihypertensive regimens
- counseling and psychotherapy to help focus the relationship back to intimacy and sexuality. Remember that just enrolling in the clinical trials I described and paying attention to sexuality increased measures of female sexual function by as much as 30%
- encouragement about a healthy lifestyle, such as regular exercise, which increases blood flow to the genitalia—as does discontinuation of smoking. Sildenafil may have a role in managing SSRI-induced or vascular disease–related genital arousal disorder.2
Stay tuned
Despite recent disappointments in pharmacotherapy, our awareness about and knowledge of female sexual dysfunction continues to grow. Safe and effective treatments for HSDD and the other conditions affecting women’s sexual function are in the pipeline.
We want to hear from you! Tell us what you think.
1. American Psychiatric Association. DSM-5 development. Sexual and gender identity disorders. http://www.dsm5.org/ProposedRevisions/Pages/SexualandGenderIdentity
Disorders.aspx. Accessed July 27, 2010.
2. Caruso S, Rugolo S, Agnello C, Intelisano G, Di Mari L, Cianci A. Sildenafil improves sexual functioning in premenopausal women with type 1 diabetes who are affected by sexual arousal disorder: a double-blind, crossover, placebo-controlled pilot study.
Fertil Steril. 2006;85(5):1496-1501.
Since sildenafil (Viagra) was approved by the US Food and Drug Administration to treat erectile dysfunction, women have been calling for research and development of treatments for female sexual dysfunction.
Despite considerable research documenting improvement in sexual responsiveness, genital sensation, and overall well-being among women who were given testosterone after undergoing bilateral oophorectomy, there remains only one testosterone formulation for women. A combination of synthetic estrogen and methyl testosterone (Estratest; Abbott) is indicated for management of moderate to severe vasomotor symptoms associated with menopause in patients who do not respond to estrogens alone.
In the testing stage from BioSante is LibiGel, a transdermal testosterone product. Acrux is developing Luramist, a daily testosterone spray. Proctor & Gamble’s efforts to gain approval of a testosterone-containing transdermal patch (Intrinsa) for treatment of low libido were unsuccessful, largely because of concern about potential increases in the risks of coronary artery disease and breast cancer. Pivotal trial data did not demonstrate enhanced risk, but the numbers were too small and the timeframe too short (a maximum follow-up of 2 years) to establish an effect, so the FDA asked for long-term studies. In 2006, European regulators approved Intrinsa to treat low sexual desire in surgically menopausal women.
Then there’s flibanserin, which also failed to win approval from an FDA advisory committee after numerous concerns were raised about its safety and efficacy in premenopausal women.
The lack of approved drugs leaves gynecologists and women’s health providers with little to offer our patients who are distressed by sexual dysfunction.
In this Update, I discuss:
- the complexity of female sexual function
- what derailed flibanserin
- recent findings that suggest dehydroepiandrosterone (DHEA) may be beneficial
- recommendations for clinical practice.
As understanding of female sexual dysfunction evolves, so do its labels
The fourth edition of the Diagnostic and Statistical Manual of Mental Disorders (DSM-IV) divides female sexual dysfunction into four categories:
- hypoactive sexual desire disorder (HSDD)—a persistent or recurrent deficiency or absence of sexual fantasies and desire for sexual activity
- female sexual arousal disorder—a persistent or recurrent inability to achieve or maintain adequate vaginal lubrication or vulvar swelling (i.e., sexual excitement)
- female orgasmic disorder—persistent or recurrent delay in or absence of orgasm following a normal sexual excitement phase
- dyspareunia—persistent or recurrent genital pain that is associated with sexual intercourse.
These categories were revised in 2003 by an international consensus committee sponsored by the American Urological Association Foundation; arousal disorder has been subdivided into:
- combined arousal disorder—absent feelings of sexual arousal from any type of stimulation, as well as absent or impaired genital sexual arousal (vulvar swelling and vaginal lubrication)
- subjective arousal disorder—absent feelings of sexual excitement and pleasure from any type of stimulation in the presence of genital sexual arousal (vulvar swelling and vaginal lubrication)
- genital arousal disorder—subjective sexual excitement from nongenital sexual stimuli with reduced sensation from genital touching and an absence of genital sexual arousal from any type of sexual stimulation.
These updated definitions will be incorporated into DSM-V, to be published in 2013.
Also likely to change in DSM-V: HSDD and female sexual arousal disorder may be subsumed into a new category, “sexual interest/arousal disorder in women”.1
The female response to sexual stimuli is complex
The complexity of sexual arousal disorders in women complicates research into the pathophysiology and potential pharmacologic treatment of these conditions. Conflicting evidence for any benefit of the phosphodiesterase type-5 (PDE5) inhibitors, such as sildenafil, in the treatment of sexual dysfunction in women likely arises from a lack of precision in defining the conditions in which and patients for whom these interventions are appropriate.
Functional magnetic resonance imaging (MRI) studies of men and women reveal differences in areas of brain activity related to sexual arousal. The neurophysiology of sexual desire and response is complex, involving multiple neurotransmitters, peptides, and hormones as well as multiple structural regions within the brain. Dopamine, norepinephrine, melanocortin, oxytocin, and serotonin (at some of its receptors) promote sexual activity, whereas prolactin, gamma amino butyric acid (GABA), and serotonin (at most of its receptors) are inhibitory.
In animal studies, both an increase in dopamine and a change in social environment can trigger increased sexual behavior. In women, a dopaminergic drug such as buproprion may increase arousability and pleasure—but so can a new partner.
All these bits of the “big picture” continue to complicate research in female sexual function.
It is imperative that we begin to understand the nuances of our patients’ sexual problems if we are to offer effective suggestions for treatment and management. Objectively determined genital arousal disorder very likely derives from neurovascular causes and is likely to respond to PDE5 inhibitors, but subjective arousal disorder with normal vulvar and vaginal engorgement and lubrication is not likely to respond to these agents.
This is the state of our basic science knowledge in 2010. What’s out there and on the horizon for us to offer our patients?
Flibanserin gets an unequivocal thumbs down
Phase-3 Trial 511.71. A twenty-four week, randomized, double-blind, placebo-controlled, safety and efficacy trial of flibanserin 50 milligrams every evening and flibanserin 100 mg every evening in women with hypoactive sexual desire disorder in North America. NCT00360529.
Phase-3 Trial 511.75. Best tolerability: 50 mg twice daily versus 100 mg in the evening versus 25 mg twice daily versus placebo in younger women in North America. NCT00360555.
Flibanserin is a 5HT 1A agonist, 2A antagonist, and weak dopamine agonist. It was originally studied as a treatment for major depressive disorder. In phase-2 trials, it was ineffective for management of depression but superior to placebo and an active comparator in improving sex drive (based on validated questionnaires). These results formed the basis for studying flibanserin as a treatment for HSDD. More than 5,000 women have been involved in phase-2 and phase-3 trials in the United States, Canada, and Europe.
Following FDA guidance for sponsors developing treatments for HSDD, drug maker Boehringer Ingelheim defined the primary endpoints for the pivotal trials as an increase in the number of sexually satisfying events (SSEs) and sexual desire, as measured by a daily diary. Sexual events included:
- genital touching by the partner
- masturbation
- oral sex
- intercourse
- orgasm.
Sexual desire was rated daily by the participants using an eDiary.
In North American phase-3 trials, 2,462 premenopausal women with acquired HSDD in stable, monogamous, functionally heterosexual, communicative relationships for at least 1 year were enrolled. Comorbid arousal and orgasmic disorders were allowed if they were secondary to decreased desire. Mean age of the participants was 35 to 36 years, and they were predominantly white, highly educated women in long-term relationships.
Two important exclusions worth noting:
- women who had depression, breast or other cancers (except skin cancer), or any major medical condition
- women who were taking any of the medications on a five-page list of excluded drugs (due to metabolism with the enzyme cytochrome P3A4).
Once they were screened, women completed a 4-week baseline assessment of sexual activity and desire, followed by a 24-week study period. They were randomized to receive 50 mg or 100 mg of flibanserin or placebo daily. Improvements in sexual function, compared with the 4-week baseline, had to be both statistically and clinically significant for the studies to be successful.
Flibanserin’s effects were clinically unimpressive
At a daily dosage of 100 mg, flibanserin was associated with a significant increase in the number of SSEs, compared with placebo. However, the co-primary endpoint of an increase in desire, as assessed by the eDiary, was not achieved in the active treatment group. Women who took flibanserin had a response rate of 30% to 40%, compared with 15% to 30% for women who took placebo. Although this difference was significant, it was clinically unimpressive, with fewer than 50% of participants experiencing significant improvement (TABLE 1).
TABLE 1
Flibanserin increased the mean number of sexually satisfying events—but improvement was modest
| Phase-3 trial | Measure | Placebo | Flibanserin | P value |
|---|---|---|---|---|
| 511.71 | Baseline | 2.7 | 3.0 | |
| 24 weeks | 3.5 | 4.6 | .005 | |
| 511.75 | Baseline | 2.7 | 2.6 | |
| 24 weeks | 2.6 | 4.4 | .024 |
Frequency of side effects is troubling
Among women who took the active drug, 34.6% discontinued the medication because of side effects, compared with 6.8% of women who took placebo. Most common side effects (and their incidence) were:
- nausea (12%)
- dizziness (11%)
- fatigue (11%)
- daytime somnolence (9.5%)
- anxiety (2%).
In the healthy study population, no major safety issues were associated with flibanserin. However, concomitant use of alcohol, a selective serotonin reuptake inhibitor (SSRI) or serotonin-norepinephrine reuptake inhibitor (SNRI), or a triptan was associated with a marked increase in side effects.
In addition, women who were predisposed to depression or suicidal ideation were more likely to develop suicidal tendencies while taking flibanserin, compared with placebo.
Given the long list of prohibited medications (CYP3A4 promoters or inhibitors), many of which are in widespread use, and given the lack of pharmacodynamic assessment by the sponsor of circulating levels of active drug if used with any of these medications, FDA reviewers and advisory panel members grew concerned about the potential for major side effects if flibanserin were to be released for commercial use.
Ultimately, the FDA advisory panel voted unanimously to withhold approval of flibanserin for treatment of HSDD in premenopausal women but encouraged the company to continue studies in postmenopausal women. Recruitment is under way for NCT00996372.
Attendees who spoke during the open public hearing on flibanserin, held June 8 in Gaithersburg, Md, had mixed opinions of the drug’s utility in premenopausal women with hypoactive sexual desire disorder (HSDD). Some believe approval of a “female Viagra” is long overdue. Others, not so much.
“In 1920, women were given the right to vote,” said Sue Goldstein, clinical trial coordinator at San Diego Sexual Medicine in San Diego, Calif. “What we’re asking now is that women be empowered again, that they be given the right to choose to be treated with an FDA-approved product for HSDD.” She added: “Once again, we are the forgotten gender.”
Leonore Tiefer, PhD, sees things differently.
“The simple but appealing notion that a new brain drug can help you with [hypoactive sexual desire disorder] because, well, desire is in the brain, has been peddled for the past year as if it were a fancy pair of shoes,” said Tiefer, clinical associate professor of psychiatry at New York University School of Medicine. “Flibanserin is not a choice when it’s promoted by bad science and half-truths and when self-diagnosis checklists are passed off as medical care.” Her last comment was a reference to sexbrainbody.com, a Web site sponsored by Boehringer Ingelheim that offers, among other resources, a “sexual satisfaction checklist.”
Many other attendees had a personal interest in the issue or represented advocacy groups, and their responses were just as mixed.
Michelle King Robson, founder and CEO of Empow-Her, a women’s health media company, favored approval of flibanserin.
“Women are struggling to find solutions to their sexual dysfunction,” she said.
Liz Canner, director of the feature documentary film Orgasm, Inc., which takes as its subject “the strange science of female pleasure,” accused Boehringer Ingelheim of “disease mongering” (and held a viewing of her film in a room down the hall).
One of the last to speak was Amy Allina, program director of the National Women’s Health Network, who asserted that flibanserin offers “little benefit for real women in the real world.”
There may one day be an effective agent for HSDD, she said.
“This drug is not it,” she added.—Janelle Yates, Senior Editor
Intravaginal DHEA improves postmenopausal sexual function
Labrie F, Archer D, Bouchard C, et al. Effect of intravaginal dehydroepiandrosterone (Prasterone) on libido and sexual dysfunction in postmenopausal women. Menopause. 2009;16(5):923–931.
DHEA has been studied as a treatment for female sexual dysfunction in postmenopausal women, in whom it acts as a precursor for both estrogen and androgen synthesis. In this study by Labrie and colleagues, all aspects of female sexual function—desire, arousal, orgasm, and pain—improved significantly with intravaginal DHEA (TABLE 2).
This phase-3, multicenter, placebo-controlled, randomized clinical trial of 216 participants—50 in each arm—randomized women to placebo or 3.25 mg, 6.5 mg, or 13 mg of DHEA daily. Median age of participants was 58.
The study began with a 4-week baseline screening phase, followed by 12 weeks of placebo or active treatment. Women were enrolled if they were postmenopausal and experienced vaginal dryness or vulvar or vaginal irritation or pain. Most women had moderate to severe symptoms.
Topical or systemic hormone therapy was prohibited, and women who had preexisting cancer (except skin cancer) or endometrial hyperplasia were excluded. The primary endpoint was improvement in the four domains of sexual dysfunction.
Although women were not initially selected for this trial based on measures of personal distress related to their sexual dysfunction (marked distress or interpersonal difficulty is required by DSM-IV for a diagnosis), approximately 50% indicated a desire for improvement on the intake questionnaire. Women were not excluded from this study if they were taking other medications known to affect sexual function, with the exception of systemic or topical hormonal treatment.
The robust results in this study were achieved without increasing circulating levels of estrogen, testosterone, or DHEA beyond the normal postmenopausal range.
TABLE 2
Women using intravaginal DHEA experienced improvement in all four domains of female sexual function
| Domain | Improvement after 12 weeks of 1% DHEA | P value |
|---|---|---|
| Desire | 49% | <.0001 |
| Arousal | 68% | .0004 |
| Orgasm | 75% | <.0001 |
| Dryness (pain) | 57% | .0001 |
What can we offer to our patients?
Female sexual dysfunction is more difficult to categorize and certainly more difficult to measure scientifically than male sexual dysfunction. The distinction between desire, arousal, and pain disorders in women is easily blurred. Certainly, the ability to declare success in clinical trials is straightforward and unequivocal for men. Not so for women.
Female sexual dysfunction that causes distress for our patients is not uncommon. It is a source of frustration for our patients and for us as providers. For now, in the absence of a “little pink pill,” we can offer:
- techniques to eliminate pain such as topical estrogen for atrophy and physical therapy and biofeedback for secondary vaginismus
- adjustment of medications that may thwart sexual desire, arousal, or orgasm, such as SSRIs and antihypertensive regimens
- counseling and psychotherapy to help focus the relationship back to intimacy and sexuality. Remember that just enrolling in the clinical trials I described and paying attention to sexuality increased measures of female sexual function by as much as 30%
- encouragement about a healthy lifestyle, such as regular exercise, which increases blood flow to the genitalia—as does discontinuation of smoking. Sildenafil may have a role in managing SSRI-induced or vascular disease–related genital arousal disorder.2
Stay tuned
Despite recent disappointments in pharmacotherapy, our awareness about and knowledge of female sexual dysfunction continues to grow. Safe and effective treatments for HSDD and the other conditions affecting women’s sexual function are in the pipeline.
We want to hear from you! Tell us what you think.
Since sildenafil (Viagra) was approved by the US Food and Drug Administration to treat erectile dysfunction, women have been calling for research and development of treatments for female sexual dysfunction.
Despite considerable research documenting improvement in sexual responsiveness, genital sensation, and overall well-being among women who were given testosterone after undergoing bilateral oophorectomy, there remains only one testosterone formulation for women. A combination of synthetic estrogen and methyl testosterone (Estratest; Abbott) is indicated for management of moderate to severe vasomotor symptoms associated with menopause in patients who do not respond to estrogens alone.
In the testing stage from BioSante is LibiGel, a transdermal testosterone product. Acrux is developing Luramist, a daily testosterone spray. Proctor & Gamble’s efforts to gain approval of a testosterone-containing transdermal patch (Intrinsa) for treatment of low libido were unsuccessful, largely because of concern about potential increases in the risks of coronary artery disease and breast cancer. Pivotal trial data did not demonstrate enhanced risk, but the numbers were too small and the timeframe too short (a maximum follow-up of 2 years) to establish an effect, so the FDA asked for long-term studies. In 2006, European regulators approved Intrinsa to treat low sexual desire in surgically menopausal women.
Then there’s flibanserin, which also failed to win approval from an FDA advisory committee after numerous concerns were raised about its safety and efficacy in premenopausal women.
The lack of approved drugs leaves gynecologists and women’s health providers with little to offer our patients who are distressed by sexual dysfunction.
In this Update, I discuss:
- the complexity of female sexual function
- what derailed flibanserin
- recent findings that suggest dehydroepiandrosterone (DHEA) may be beneficial
- recommendations for clinical practice.
As understanding of female sexual dysfunction evolves, so do its labels
The fourth edition of the Diagnostic and Statistical Manual of Mental Disorders (DSM-IV) divides female sexual dysfunction into four categories:
- hypoactive sexual desire disorder (HSDD)—a persistent or recurrent deficiency or absence of sexual fantasies and desire for sexual activity
- female sexual arousal disorder—a persistent or recurrent inability to achieve or maintain adequate vaginal lubrication or vulvar swelling (i.e., sexual excitement)
- female orgasmic disorder—persistent or recurrent delay in or absence of orgasm following a normal sexual excitement phase
- dyspareunia—persistent or recurrent genital pain that is associated with sexual intercourse.
These categories were revised in 2003 by an international consensus committee sponsored by the American Urological Association Foundation; arousal disorder has been subdivided into:
- combined arousal disorder—absent feelings of sexual arousal from any type of stimulation, as well as absent or impaired genital sexual arousal (vulvar swelling and vaginal lubrication)
- subjective arousal disorder—absent feelings of sexual excitement and pleasure from any type of stimulation in the presence of genital sexual arousal (vulvar swelling and vaginal lubrication)
- genital arousal disorder—subjective sexual excitement from nongenital sexual stimuli with reduced sensation from genital touching and an absence of genital sexual arousal from any type of sexual stimulation.
These updated definitions will be incorporated into DSM-V, to be published in 2013.
Also likely to change in DSM-V: HSDD and female sexual arousal disorder may be subsumed into a new category, “sexual interest/arousal disorder in women”.1
The female response to sexual stimuli is complex
The complexity of sexual arousal disorders in women complicates research into the pathophysiology and potential pharmacologic treatment of these conditions. Conflicting evidence for any benefit of the phosphodiesterase type-5 (PDE5) inhibitors, such as sildenafil, in the treatment of sexual dysfunction in women likely arises from a lack of precision in defining the conditions in which and patients for whom these interventions are appropriate.
Functional magnetic resonance imaging (MRI) studies of men and women reveal differences in areas of brain activity related to sexual arousal. The neurophysiology of sexual desire and response is complex, involving multiple neurotransmitters, peptides, and hormones as well as multiple structural regions within the brain. Dopamine, norepinephrine, melanocortin, oxytocin, and serotonin (at some of its receptors) promote sexual activity, whereas prolactin, gamma amino butyric acid (GABA), and serotonin (at most of its receptors) are inhibitory.
In animal studies, both an increase in dopamine and a change in social environment can trigger increased sexual behavior. In women, a dopaminergic drug such as buproprion may increase arousability and pleasure—but so can a new partner.
All these bits of the “big picture” continue to complicate research in female sexual function.
It is imperative that we begin to understand the nuances of our patients’ sexual problems if we are to offer effective suggestions for treatment and management. Objectively determined genital arousal disorder very likely derives from neurovascular causes and is likely to respond to PDE5 inhibitors, but subjective arousal disorder with normal vulvar and vaginal engorgement and lubrication is not likely to respond to these agents.
This is the state of our basic science knowledge in 2010. What’s out there and on the horizon for us to offer our patients?
Flibanserin gets an unequivocal thumbs down
Phase-3 Trial 511.71. A twenty-four week, randomized, double-blind, placebo-controlled, safety and efficacy trial of flibanserin 50 milligrams every evening and flibanserin 100 mg every evening in women with hypoactive sexual desire disorder in North America. NCT00360529.
Phase-3 Trial 511.75. Best tolerability: 50 mg twice daily versus 100 mg in the evening versus 25 mg twice daily versus placebo in younger women in North America. NCT00360555.
Flibanserin is a 5HT 1A agonist, 2A antagonist, and weak dopamine agonist. It was originally studied as a treatment for major depressive disorder. In phase-2 trials, it was ineffective for management of depression but superior to placebo and an active comparator in improving sex drive (based on validated questionnaires). These results formed the basis for studying flibanserin as a treatment for HSDD. More than 5,000 women have been involved in phase-2 and phase-3 trials in the United States, Canada, and Europe.
Following FDA guidance for sponsors developing treatments for HSDD, drug maker Boehringer Ingelheim defined the primary endpoints for the pivotal trials as an increase in the number of sexually satisfying events (SSEs) and sexual desire, as measured by a daily diary. Sexual events included:
- genital touching by the partner
- masturbation
- oral sex
- intercourse
- orgasm.
Sexual desire was rated daily by the participants using an eDiary.
In North American phase-3 trials, 2,462 premenopausal women with acquired HSDD in stable, monogamous, functionally heterosexual, communicative relationships for at least 1 year were enrolled. Comorbid arousal and orgasmic disorders were allowed if they were secondary to decreased desire. Mean age of the participants was 35 to 36 years, and they were predominantly white, highly educated women in long-term relationships.
Two important exclusions worth noting:
- women who had depression, breast or other cancers (except skin cancer), or any major medical condition
- women who were taking any of the medications on a five-page list of excluded drugs (due to metabolism with the enzyme cytochrome P3A4).
Once they were screened, women completed a 4-week baseline assessment of sexual activity and desire, followed by a 24-week study period. They were randomized to receive 50 mg or 100 mg of flibanserin or placebo daily. Improvements in sexual function, compared with the 4-week baseline, had to be both statistically and clinically significant for the studies to be successful.
Flibanserin’s effects were clinically unimpressive
At a daily dosage of 100 mg, flibanserin was associated with a significant increase in the number of SSEs, compared with placebo. However, the co-primary endpoint of an increase in desire, as assessed by the eDiary, was not achieved in the active treatment group. Women who took flibanserin had a response rate of 30% to 40%, compared with 15% to 30% for women who took placebo. Although this difference was significant, it was clinically unimpressive, with fewer than 50% of participants experiencing significant improvement (TABLE 1).
TABLE 1
Flibanserin increased the mean number of sexually satisfying events—but improvement was modest
| Phase-3 trial | Measure | Placebo | Flibanserin | P value |
|---|---|---|---|---|
| 511.71 | Baseline | 2.7 | 3.0 | |
| 24 weeks | 3.5 | 4.6 | .005 | |
| 511.75 | Baseline | 2.7 | 2.6 | |
| 24 weeks | 2.6 | 4.4 | .024 |
Frequency of side effects is troubling
Among women who took the active drug, 34.6% discontinued the medication because of side effects, compared with 6.8% of women who took placebo. Most common side effects (and their incidence) were:
- nausea (12%)
- dizziness (11%)
- fatigue (11%)
- daytime somnolence (9.5%)
- anxiety (2%).
In the healthy study population, no major safety issues were associated with flibanserin. However, concomitant use of alcohol, a selective serotonin reuptake inhibitor (SSRI) or serotonin-norepinephrine reuptake inhibitor (SNRI), or a triptan was associated with a marked increase in side effects.
In addition, women who were predisposed to depression or suicidal ideation were more likely to develop suicidal tendencies while taking flibanserin, compared with placebo.
Given the long list of prohibited medications (CYP3A4 promoters or inhibitors), many of which are in widespread use, and given the lack of pharmacodynamic assessment by the sponsor of circulating levels of active drug if used with any of these medications, FDA reviewers and advisory panel members grew concerned about the potential for major side effects if flibanserin were to be released for commercial use.
Ultimately, the FDA advisory panel voted unanimously to withhold approval of flibanserin for treatment of HSDD in premenopausal women but encouraged the company to continue studies in postmenopausal women. Recruitment is under way for NCT00996372.
Attendees who spoke during the open public hearing on flibanserin, held June 8 in Gaithersburg, Md, had mixed opinions of the drug’s utility in premenopausal women with hypoactive sexual desire disorder (HSDD). Some believe approval of a “female Viagra” is long overdue. Others, not so much.
“In 1920, women were given the right to vote,” said Sue Goldstein, clinical trial coordinator at San Diego Sexual Medicine in San Diego, Calif. “What we’re asking now is that women be empowered again, that they be given the right to choose to be treated with an FDA-approved product for HSDD.” She added: “Once again, we are the forgotten gender.”
Leonore Tiefer, PhD, sees things differently.
“The simple but appealing notion that a new brain drug can help you with [hypoactive sexual desire disorder] because, well, desire is in the brain, has been peddled for the past year as if it were a fancy pair of shoes,” said Tiefer, clinical associate professor of psychiatry at New York University School of Medicine. “Flibanserin is not a choice when it’s promoted by bad science and half-truths and when self-diagnosis checklists are passed off as medical care.” Her last comment was a reference to sexbrainbody.com, a Web site sponsored by Boehringer Ingelheim that offers, among other resources, a “sexual satisfaction checklist.”
Many other attendees had a personal interest in the issue or represented advocacy groups, and their responses were just as mixed.
Michelle King Robson, founder and CEO of Empow-Her, a women’s health media company, favored approval of flibanserin.
“Women are struggling to find solutions to their sexual dysfunction,” she said.
Liz Canner, director of the feature documentary film Orgasm, Inc., which takes as its subject “the strange science of female pleasure,” accused Boehringer Ingelheim of “disease mongering” (and held a viewing of her film in a room down the hall).
One of the last to speak was Amy Allina, program director of the National Women’s Health Network, who asserted that flibanserin offers “little benefit for real women in the real world.”
There may one day be an effective agent for HSDD, she said.
“This drug is not it,” she added.—Janelle Yates, Senior Editor
Intravaginal DHEA improves postmenopausal sexual function
Labrie F, Archer D, Bouchard C, et al. Effect of intravaginal dehydroepiandrosterone (Prasterone) on libido and sexual dysfunction in postmenopausal women. Menopause. 2009;16(5):923–931.
DHEA has been studied as a treatment for female sexual dysfunction in postmenopausal women, in whom it acts as a precursor for both estrogen and androgen synthesis. In this study by Labrie and colleagues, all aspects of female sexual function—desire, arousal, orgasm, and pain—improved significantly with intravaginal DHEA (TABLE 2).
This phase-3, multicenter, placebo-controlled, randomized clinical trial of 216 participants—50 in each arm—randomized women to placebo or 3.25 mg, 6.5 mg, or 13 mg of DHEA daily. Median age of participants was 58.
The study began with a 4-week baseline screening phase, followed by 12 weeks of placebo or active treatment. Women were enrolled if they were postmenopausal and experienced vaginal dryness or vulvar or vaginal irritation or pain. Most women had moderate to severe symptoms.
Topical or systemic hormone therapy was prohibited, and women who had preexisting cancer (except skin cancer) or endometrial hyperplasia were excluded. The primary endpoint was improvement in the four domains of sexual dysfunction.
Although women were not initially selected for this trial based on measures of personal distress related to their sexual dysfunction (marked distress or interpersonal difficulty is required by DSM-IV for a diagnosis), approximately 50% indicated a desire for improvement on the intake questionnaire. Women were not excluded from this study if they were taking other medications known to affect sexual function, with the exception of systemic or topical hormonal treatment.
The robust results in this study were achieved without increasing circulating levels of estrogen, testosterone, or DHEA beyond the normal postmenopausal range.
TABLE 2
Women using intravaginal DHEA experienced improvement in all four domains of female sexual function
| Domain | Improvement after 12 weeks of 1% DHEA | P value |
|---|---|---|
| Desire | 49% | <.0001 |
| Arousal | 68% | .0004 |
| Orgasm | 75% | <.0001 |
| Dryness (pain) | 57% | .0001 |
What can we offer to our patients?
Female sexual dysfunction is more difficult to categorize and certainly more difficult to measure scientifically than male sexual dysfunction. The distinction between desire, arousal, and pain disorders in women is easily blurred. Certainly, the ability to declare success in clinical trials is straightforward and unequivocal for men. Not so for women.
Female sexual dysfunction that causes distress for our patients is not uncommon. It is a source of frustration for our patients and for us as providers. For now, in the absence of a “little pink pill,” we can offer:
- techniques to eliminate pain such as topical estrogen for atrophy and physical therapy and biofeedback for secondary vaginismus
- adjustment of medications that may thwart sexual desire, arousal, or orgasm, such as SSRIs and antihypertensive regimens
- counseling and psychotherapy to help focus the relationship back to intimacy and sexuality. Remember that just enrolling in the clinical trials I described and paying attention to sexuality increased measures of female sexual function by as much as 30%
- encouragement about a healthy lifestyle, such as regular exercise, which increases blood flow to the genitalia—as does discontinuation of smoking. Sildenafil may have a role in managing SSRI-induced or vascular disease–related genital arousal disorder.2
Stay tuned
Despite recent disappointments in pharmacotherapy, our awareness about and knowledge of female sexual dysfunction continues to grow. Safe and effective treatments for HSDD and the other conditions affecting women’s sexual function are in the pipeline.
We want to hear from you! Tell us what you think.
1. American Psychiatric Association. DSM-5 development. Sexual and gender identity disorders. http://www.dsm5.org/ProposedRevisions/Pages/SexualandGenderIdentity
Disorders.aspx. Accessed July 27, 2010.
2. Caruso S, Rugolo S, Agnello C, Intelisano G, Di Mari L, Cianci A. Sildenafil improves sexual functioning in premenopausal women with type 1 diabetes who are affected by sexual arousal disorder: a double-blind, crossover, placebo-controlled pilot study.
Fertil Steril. 2006;85(5):1496-1501.
1. American Psychiatric Association. DSM-5 development. Sexual and gender identity disorders. http://www.dsm5.org/ProposedRevisions/Pages/SexualandGenderIdentity
Disorders.aspx. Accessed July 27, 2010.
2. Caruso S, Rugolo S, Agnello C, Intelisano G, Di Mari L, Cianci A. Sildenafil improves sexual functioning in premenopausal women with type 1 diabetes who are affected by sexual arousal disorder: a double-blind, crossover, placebo-controlled pilot study.
Fertil Steril. 2006;85(5):1496-1501.
Skilled US imaging of the adnexal mass: Starting point
No doubt about it: Scanning the adnexae is the most challenging task in gynecologic ultrasonography (US). There are many reasons for the difficulty, but probably none more important than the fact that you are expected to reach a conclusion about what you see—or at least narrow the differential diagnosis.
Some ultrasound laboratories try to hedge their bets, sending the referring physician a report that is nothing more than an exhaustive differential diagnosis, similar to what we see in textbooks. Such a list is useless to a referring clinician, who has probably already considered most of the possibilities and involved the lab to help narrow them down. Labs that send such reports are usually trying to protect themselves from litigation—typically involving cases in which ovarian cancer was missed—or attempting to accomplish a “self-referral” by encouraging further imaging.1
The referring physician is not perfect, either. In our practice, we often receive reports like the following terse description:
A complex cyst was seen in the adnexa. Ovarian malignancy cannot be ruled out.
That’s it. No description of the actual sonographic characteristics. No Doppler velocity flow studies. Yet, the few remarks include a mention of malignancy, and the provider often suggests that “additional imaging such as CT and MRI should be considered.”
When we scrutinize the sonographic images upon which these reports are based, we often discover a corpus luteum, cystic teratoma, benign cystadenoma, endometrioma, or, even, a simple cyst.
The need for competency is compelling
Now that gynecologic US has matured as a field in its own right, the referring physician should expect much more from a laboratory’s pelvic scan than a long recitation of potential diagnoses. And the lab should expect more basic information from the referring provider.
That is the primary reason for this four-part series—to help you identify some of the most prevalent adnexal masses, so that you can exclude cases that are no cause for concern, such as a corpus luteum, and refer patients who really do need additional imaging and expertise, providing as much information in the process as you can.
In Part 1 of the series, we introduce you to basic concepts, recommend equipment, and step you through numerous fundamental scans. Part 2 will focus on nonneoplastic ovarian masses, Part 3 on ovarian neoplasms, and Part 4 on tubal entities such as ectopic pregnancy and torsion.
As much as possible, we educate you by providing actual scans that represent real cases, pointing out the elements that should grab your attention. After all, a picture paints a thousand words.
Ultrasound reveals the polycystic nature of a patient’s ovary. The hilus is prominently hyperechoic.
A few fundamental practices enhance consistency and thoroughness
Before we shift our focus to scanning techniques and interpretation of images, we’d like to offer several basic pointers.
Establish, and document, the hormonal milieu. One of the most important requirements of US imaging, particularly during the reproductive years, is determining and documenting the date of the patient’s last menstrual period (LMP). The reason? Physiologic and pathologic processes involving the reproductive organs are driven by the menstrual cycle—or by therapeutic (or pathologic) hormonal stimulation. We mark each scan with the date of the LMP. If the patient is on hormone therapy, we also mark the scan “HT.” We make these marks on the screen in a way that prevents their erasure every time the picture is frozen and unfrozen. This makes it possible for us to look at the scan days, weeks, or even years later and know what day of the cycle it represents. Every finding must be judged in light of the patient’s hormonal status.
Use a transvaginal transducer. It provides a high-resolution view of any pathology. If need be, it can be combined with a trans-abdominal transducer to afford a more deeply penetrating, panoramic view of the pelvis. We use a variety of transducers to achieve depth, color, power Doppler, and three- dimensional (3D) US.
Take a history and examine the patient. Before scanning your own patient, take a short history and perform a bimanual, palpatory pelvic exam. You may need to examine her again after the scan to verify a sonographic finding.
It is doubly important to take a history if you are scanning a referred patient. Omitting this element is no excuse for overlooking a disease or pathology.
A bimanual, palpatory pelvic exam may also be recommended for some referred patients.
A transvaginal scan is not always possible. There are a number of reasons why the transvaginal approach may not be advisable for some patients, including virginal status, atrophic postmenopausal vagina, agenesis of the vagina, and transverse vaginal septae. In such cases, the best alternative is a transrectal scan, which makes it possible to image the pelvic organs from almost exactly the same vantage point as transvaginal US.2 With proper explanation (particularly with virginal patients), the initial reluctance and apprehension can usually be assuaged.
Don’t trust the referral slip. We recommend that you read, but do not overly trust, the referral slip. It often offers little useful information.
Helpful scanning techniques
Consider applying these maneuvers:
- place your non-scanning hand on the patient’s abdomen to help mobilize the pelvic contents as the transvaginal probe slides across the organs
- use the probe as an “eye” while your palpating finger touches the cervix, uterus, ovaries, and any adnexal mass. Observe the mobility of these structures in relation to each other and the pelvic wall. This technique yields what is often referred to as the “sliding organs” sign. It is possible to identify pelvic adhesions (if the structures do not slide freely) or rule them out (if they do)
- pinpoint the origin of any pain the patient may have by touching the ovary, cervix, and any adnexal mass. This technique is important in cases of ectopic pregnancy, adnexal torsion, or inflammatory disease of the pelvis or adnexae.
Start with a basic scan of key structures
On the way “in” toward the adnexae, take the time to look at the bladder and urethra (FIGURE 1). Some common pathologies of the bladder are diverticulae; calculi; and a thick and vascular bladder wall suggestive of cancer or endometrioma. Ask the patient whether she has experienced any hematuria if any of these pathologies are detected.
FIGURE 1 Imaging the bladder
(A, B) The bladder (bl), urethra (u), vagina (v), and rectum (r) appear in their proper relation in this sagittal view. The posterior angle of the bladder is also apparent (arrow closing an angle of about 110°). (C) Excessive thickness of the bladder wall suggests that this patient has cystitis. (D) Coronal view of the bladder and urethra (solid arrows).
Also take a look at the cervix, searching for Nabothian cysts, endocervical polyps, extreme vascularization (a possible indicator of cervical cancer), and prolapsing submucous myomas (FIGURE 2).
FIGURE 2 Uncommon pathology
A submucous myoma prolapses into the cervical canal in a 13-week intrauterine pregnancy. (A) Grayscale sagittal image and (B) outline view of the same image. (C,D) Color and power Doppler images show the blood supply to the myoma from the uterine cavity.
While you are looking, attempt to scan both kidneys and Morrison’s pouch. Large adnexal masses or fibroids of the uterus may put pressure on the ureter, causing various degrees of hydronephrosis.
Sometimes, when the right kidney is correctly imaged below the liver, you may detect fluid in the space between them (called Morrison’s space). This information has clear value that may aid in diagnosing the main pathology (i.e., ruptured tubal pregnancy, ascites, etc.).
Imaging of the ovaries
The best way to scan the ovaries is to use a high-frequency (4–9 MHz) transvaginal probe. In general, as the frequency of the probe increases, so does resolution of the image—but the ability to penetrate tissue diminishes. For this reason, for abdominal imaging, a 3-MHz probe is often used. For a transvaginal scan, in which the probe can be placed near an ovary, a 5-MHz probe is common. And for a scan of, say, the parathyroid gland, a 12-MHz probe is utilized.
During the reproductive years, the ovaries can be localized by their sonographic markers—the follicles (FIGURE 3A). The ovaries usually lie near the large hypogastric blood vessels (FIGURE 3B). During the secretory phase of the cycle, look for the corpus luteum, switching on the color or power Doppler mode to help locate it (FIGURES 3C, 3D).
The ovaries usually can be distinguished by their relative anechoic sono-texture in juxtaposition to the surrounding, constantly peristalsing small bowel. This strategy is the only help for spotting the ovaries in menopause, when they lose their follicles.
The size of the ovaries may be an important indicator of pathology. During the reproductive years, mean size is 8 mL (standard deviation [SD], 2–3 mL; range, 5–15 mL). Post-menopausal ovaries are small, with a mean size of 3.6 mL (SD, 1.4 mL; range, 1–14 mL).
FIGURE 3 How to spot the ovaries
(A) Anechoic follicles are markers of the ovary during the reproductive years. (B) The ovaries in relation to the hypogastric vessels. (C) Gray-scale image of the corpus luteum and the same image in (D) color Doppler.
A word about terminology: Don’t call follicles “cysts”
During a normal menstrual cycle, one or more follicles mature, reaching about 2 to 2.5 cm in diameter around mid-cycle. Do not call these follicles “cysts” or “follicular cysts.” They are follicles. Calling them cysts, or even including the word cyst in their description, suggests to many gynecology and radiology providers—and to patients themselves—the idea of pathology.1
An exception to that rule: An ovary that is larger than 12 to 14 mL and has a hyperechoic hilus and more than 12 small (4–5 mm), peripherally pushed follicles is usually called “polycystic” (FIGURE 4).3 However, not every ovary that fulfills these sonographic criteria is indeed polycystic. At times normal ovaries may contain multiple follicles without any of the clinical or laboratory indications of a polycystic ovary. In these cases, the ovary may be of normal size and may lack a hyperechoic hilus with rich hilar vascularity. We term such ovaries “multicystic” in their appearance.

FIGURE 4 The polycystic ovary
(A) Gray-scale image of a polycystic ovary. The typical hyperechoic hilus is evident (H). (B) Gross pathologic section of a polycystic ovary. (C) 3D orthogonal planes of a large ovary with a multitude of small follicles pushed peripherally by a voluminous hyperechoic hilus. (D) 3D inversion rendering of the same ovary.
We employ 3D inversion rendering to better see and count the number of follicles (FIGURE 4D).
An ovary can have a polycystic appearance in the following clinical situations:
- hyperthyroid state (36% of affected women)
- hyperprolactinemia (50%)
- hypothalamic hypogonadism (24%).
It also can appear polycystic for no apparent reason.
Stay tuned!
Next month, we continue our focus on adnexal imaging by describing (and showing) nonneoplastic ovarian masses.
We want to hear from you! Tell us what you think.
1. Timor-Tritsch IE, Goldstein SR. The complexity of a complex mass and the simplicity of a simple cyst. J Ultrasound Med. 2005;24(3):255-258.
2. Timor-Tritsch IE, Monteagudo A, Rebarber A, Goldstein SR, Tsymbal T. Transrectal scanning an alternative when transvaginal scanning is not feasible. Ultrasound Obstet Gynecol. 2003;21(5):443-479.
3. Abdel Gadir A, Khatim MS, Mowafi RS, Alnaser HM, Muharib NS, Shaw RW. Implications of ultrasonically diagnosed polycystic ovaries. II. Studies of dynamic and pulsatile hormonal patterns. Human Reprod. 1992;7(4):458-461.
No doubt about it: Scanning the adnexae is the most challenging task in gynecologic ultrasonography (US). There are many reasons for the difficulty, but probably none more important than the fact that you are expected to reach a conclusion about what you see—or at least narrow the differential diagnosis.
Some ultrasound laboratories try to hedge their bets, sending the referring physician a report that is nothing more than an exhaustive differential diagnosis, similar to what we see in textbooks. Such a list is useless to a referring clinician, who has probably already considered most of the possibilities and involved the lab to help narrow them down. Labs that send such reports are usually trying to protect themselves from litigation—typically involving cases in which ovarian cancer was missed—or attempting to accomplish a “self-referral” by encouraging further imaging.1
The referring physician is not perfect, either. In our practice, we often receive reports like the following terse description:
A complex cyst was seen in the adnexa. Ovarian malignancy cannot be ruled out.
That’s it. No description of the actual sonographic characteristics. No Doppler velocity flow studies. Yet, the few remarks include a mention of malignancy, and the provider often suggests that “additional imaging such as CT and MRI should be considered.”
When we scrutinize the sonographic images upon which these reports are based, we often discover a corpus luteum, cystic teratoma, benign cystadenoma, endometrioma, or, even, a simple cyst.
The need for competency is compelling
Now that gynecologic US has matured as a field in its own right, the referring physician should expect much more from a laboratory’s pelvic scan than a long recitation of potential diagnoses. And the lab should expect more basic information from the referring provider.
That is the primary reason for this four-part series—to help you identify some of the most prevalent adnexal masses, so that you can exclude cases that are no cause for concern, such as a corpus luteum, and refer patients who really do need additional imaging and expertise, providing as much information in the process as you can.
In Part 1 of the series, we introduce you to basic concepts, recommend equipment, and step you through numerous fundamental scans. Part 2 will focus on nonneoplastic ovarian masses, Part 3 on ovarian neoplasms, and Part 4 on tubal entities such as ectopic pregnancy and torsion.
As much as possible, we educate you by providing actual scans that represent real cases, pointing out the elements that should grab your attention. After all, a picture paints a thousand words.

Ultrasound reveals the polycystic nature of a patient’s ovary. The hilus is prominently hyperechoic.
A few fundamental practices enhance consistency and thoroughness
Before we shift our focus to scanning techniques and interpretation of images, we’d like to offer several basic pointers.
Establish, and document, the hormonal milieu. One of the most important requirements of US imaging, particularly during the reproductive years, is determining and documenting the date of the patient’s last menstrual period (LMP). The reason? Physiologic and pathologic processes involving the reproductive organs are driven by the menstrual cycle—or by therapeutic (or pathologic) hormonal stimulation. We mark each scan with the date of the LMP. If the patient is on hormone therapy, we also mark the scan “HT.” We make these marks on the screen in a way that prevents their erasure every time the picture is frozen and unfrozen. This makes it possible for us to look at the scan days, weeks, or even years later and know what day of the cycle it represents. Every finding must be judged in light of the patient’s hormonal status.
Use a transvaginal transducer. It provides a high-resolution view of any pathology. If need be, it can be combined with a trans-abdominal transducer to afford a more deeply penetrating, panoramic view of the pelvis. We use a variety of transducers to achieve depth, color, power Doppler, and three- dimensional (3D) US.
Take a history and examine the patient. Before scanning your own patient, take a short history and perform a bimanual, palpatory pelvic exam. You may need to examine her again after the scan to verify a sonographic finding.
It is doubly important to take a history if you are scanning a referred patient. Omitting this element is no excuse for overlooking a disease or pathology.
A bimanual, palpatory pelvic exam may also be recommended for some referred patients.
A transvaginal scan is not always possible. There are a number of reasons why the transvaginal approach may not be advisable for some patients, including virginal status, atrophic postmenopausal vagina, agenesis of the vagina, and transverse vaginal septae. In such cases, the best alternative is a transrectal scan, which makes it possible to image the pelvic organs from almost exactly the same vantage point as transvaginal US.2 With proper explanation (particularly with virginal patients), the initial reluctance and apprehension can usually be assuaged.
Don’t trust the referral slip. We recommend that you read, but do not overly trust, the referral slip. It often offers little useful information.
Helpful scanning techniques
Consider applying these maneuvers:
- place your non-scanning hand on the patient’s abdomen to help mobilize the pelvic contents as the transvaginal probe slides across the organs
- use the probe as an “eye” while your palpating finger touches the cervix, uterus, ovaries, and any adnexal mass. Observe the mobility of these structures in relation to each other and the pelvic wall. This technique yields what is often referred to as the “sliding organs” sign. It is possible to identify pelvic adhesions (if the structures do not slide freely) or rule them out (if they do)
- pinpoint the origin of any pain the patient may have by touching the ovary, cervix, and any adnexal mass. This technique is important in cases of ectopic pregnancy, adnexal torsion, or inflammatory disease of the pelvis or adnexae.
Start with a basic scan of key structures
On the way “in” toward the adnexae, take the time to look at the bladder and urethra (FIGURE 1). Some common pathologies of the bladder are diverticulae; calculi; and a thick and vascular bladder wall suggestive of cancer or endometrioma. Ask the patient whether she has experienced any hematuria if any of these pathologies are detected.

FIGURE 1 Imaging the bladder
(A, B) The bladder (bl), urethra (u), vagina (v), and rectum (r) appear in their proper relation in this sagittal view. The posterior angle of the bladder is also apparent (arrow closing an angle of about 110°). (C) Excessive thickness of the bladder wall suggests that this patient has cystitis. (D) Coronal view of the bladder and urethra (solid arrows).
Also take a look at the cervix, searching for Nabothian cysts, endocervical polyps, extreme vascularization (a possible indicator of cervical cancer), and prolapsing submucous myomas (FIGURE 2).

FIGURE 2 Uncommon pathology
A submucous myoma prolapses into the cervical canal in a 13-week intrauterine pregnancy. (A) Grayscale sagittal image and (B) outline view of the same image. (C,D) Color and power Doppler images show the blood supply to the myoma from the uterine cavity.
While you are looking, attempt to scan both kidneys and Morrison’s pouch. Large adnexal masses or fibroids of the uterus may put pressure on the ureter, causing various degrees of hydronephrosis.
Sometimes, when the right kidney is correctly imaged below the liver, you may detect fluid in the space between them (called Morrison’s space). This information has clear value that may aid in diagnosing the main pathology (i.e., ruptured tubal pregnancy, ascites, etc.).
Imaging of the ovaries
The best way to scan the ovaries is to use a high-frequency (4–9 MHz) transvaginal probe. In general, as the frequency of the probe increases, so does resolution of the image—but the ability to penetrate tissue diminishes. For this reason, for abdominal imaging, a 3-MHz probe is often used. For a transvaginal scan, in which the probe can be placed near an ovary, a 5-MHz probe is common. And for a scan of, say, the parathyroid gland, a 12-MHz probe is utilized.
During the reproductive years, the ovaries can be localized by their sonographic markers—the follicles (FIGURE 3A). The ovaries usually lie near the large hypogastric blood vessels (FIGURE 3B). During the secretory phase of the cycle, look for the corpus luteum, switching on the color or power Doppler mode to help locate it (FIGURES 3C, 3D).
The ovaries usually can be distinguished by their relative anechoic sono-texture in juxtaposition to the surrounding, constantly peristalsing small bowel. This strategy is the only help for spotting the ovaries in menopause, when they lose their follicles.
The size of the ovaries may be an important indicator of pathology. During the reproductive years, mean size is 8 mL (standard deviation [SD], 2–3 mL; range, 5–15 mL). Post-menopausal ovaries are small, with a mean size of 3.6 mL (SD, 1.4 mL; range, 1–14 mL).

FIGURE 3 How to spot the ovaries
(A) Anechoic follicles are markers of the ovary during the reproductive years. (B) The ovaries in relation to the hypogastric vessels. (C) Gray-scale image of the corpus luteum and the same image in (D) color Doppler.
A word about terminology: Don’t call follicles “cysts”
During a normal menstrual cycle, one or more follicles mature, reaching about 2 to 2.5 cm in diameter around mid-cycle. Do not call these follicles “cysts” or “follicular cysts.” They are follicles. Calling them cysts, or even including the word cyst in their description, suggests to many gynecology and radiology providers—and to patients themselves—the idea of pathology.1
An exception to that rule: An ovary that is larger than 12 to 14 mL and has a hyperechoic hilus and more than 12 small (4–5 mm), peripherally pushed follicles is usually called “polycystic” (FIGURE 4).3 However, not every ovary that fulfills these sonographic criteria is indeed polycystic. At times normal ovaries may contain multiple follicles without any of the clinical or laboratory indications of a polycystic ovary. In these cases, the ovary may be of normal size and may lack a hyperechoic hilus with rich hilar vascularity. We term such ovaries “multicystic” in their appearance.

FIGURE 4 The polycystic ovary
(A) Gray-scale image of a polycystic ovary. The typical hyperechoic hilus is evident (H). (B) Gross pathologic section of a polycystic ovary. (C) 3D orthogonal planes of a large ovary with a multitude of small follicles pushed peripherally by a voluminous hyperechoic hilus. (D) 3D inversion rendering of the same ovary.
We employ 3D inversion rendering to better see and count the number of follicles (FIGURE 4D).
An ovary can have a polycystic appearance in the following clinical situations:
- hyperthyroid state (36% of affected women)
- hyperprolactinemia (50%)
- hypothalamic hypogonadism (24%).
It also can appear polycystic for no apparent reason.
Stay tuned!
Next month, we continue our focus on adnexal imaging by describing (and showing) nonneoplastic ovarian masses.
We want to hear from you! Tell us what you think.
No doubt about it: Scanning the adnexae is the most challenging task in gynecologic ultrasonography (US). There are many reasons for the difficulty, but probably none more important than the fact that you are expected to reach a conclusion about what you see—or at least narrow the differential diagnosis.
Some ultrasound laboratories try to hedge their bets, sending the referring physician a report that is nothing more than an exhaustive differential diagnosis, similar to what we see in textbooks. Such a list is useless to a referring clinician, who has probably already considered most of the possibilities and involved the lab to help narrow them down. Labs that send such reports are usually trying to protect themselves from litigation—typically involving cases in which ovarian cancer was missed—or attempting to accomplish a “self-referral” by encouraging further imaging.1
The referring physician is not perfect, either. In our practice, we often receive reports like the following terse description:
A complex cyst was seen in the adnexa. Ovarian malignancy cannot be ruled out.
That’s it. No description of the actual sonographic characteristics. No Doppler velocity flow studies. Yet, the few remarks include a mention of malignancy, and the provider often suggests that “additional imaging such as CT and MRI should be considered.”
When we scrutinize the sonographic images upon which these reports are based, we often discover a corpus luteum, cystic teratoma, benign cystadenoma, endometrioma, or, even, a simple cyst.
The need for competency is compelling
Now that gynecologic US has matured as a field in its own right, the referring physician should expect much more from a laboratory’s pelvic scan than a long recitation of potential diagnoses. And the lab should expect more basic information from the referring provider.
That is the primary reason for this four-part series—to help you identify some of the most prevalent adnexal masses, so that you can exclude cases that are no cause for concern, such as a corpus luteum, and refer patients who really do need additional imaging and expertise, providing as much information in the process as you can.
In Part 1 of the series, we introduce you to basic concepts, recommend equipment, and step you through numerous fundamental scans. Part 2 will focus on nonneoplastic ovarian masses, Part 3 on ovarian neoplasms, and Part 4 on tubal entities such as ectopic pregnancy and torsion.
As much as possible, we educate you by providing actual scans that represent real cases, pointing out the elements that should grab your attention. After all, a picture paints a thousand words.

Ultrasound reveals the polycystic nature of a patient’s ovary. The hilus is prominently hyperechoic.
A few fundamental practices enhance consistency and thoroughness
Before we shift our focus to scanning techniques and interpretation of images, we’d like to offer several basic pointers.
Establish, and document, the hormonal milieu. One of the most important requirements of US imaging, particularly during the reproductive years, is determining and documenting the date of the patient’s last menstrual period (LMP). The reason? Physiologic and pathologic processes involving the reproductive organs are driven by the menstrual cycle—or by therapeutic (or pathologic) hormonal stimulation. We mark each scan with the date of the LMP. If the patient is on hormone therapy, we also mark the scan “HT.” We make these marks on the screen in a way that prevents their erasure every time the picture is frozen and unfrozen. This makes it possible for us to look at the scan days, weeks, or even years later and know what day of the cycle it represents. Every finding must be judged in light of the patient’s hormonal status.
Use a transvaginal transducer. It provides a high-resolution view of any pathology. If need be, it can be combined with a trans-abdominal transducer to afford a more deeply penetrating, panoramic view of the pelvis. We use a variety of transducers to achieve depth, color, power Doppler, and three- dimensional (3D) US.
Take a history and examine the patient. Before scanning your own patient, take a short history and perform a bimanual, palpatory pelvic exam. You may need to examine her again after the scan to verify a sonographic finding.
It is doubly important to take a history if you are scanning a referred patient. Omitting this element is no excuse for overlooking a disease or pathology.
A bimanual, palpatory pelvic exam may also be recommended for some referred patients.
A transvaginal scan is not always possible. There are a number of reasons why the transvaginal approach may not be advisable for some patients, including virginal status, atrophic postmenopausal vagina, agenesis of the vagina, and transverse vaginal septae. In such cases, the best alternative is a transrectal scan, which makes it possible to image the pelvic organs from almost exactly the same vantage point as transvaginal US.2 With proper explanation (particularly with virginal patients), the initial reluctance and apprehension can usually be assuaged.
Don’t trust the referral slip. We recommend that you read, but do not overly trust, the referral slip. It often offers little useful information.
Helpful scanning techniques
Consider applying these maneuvers:
- place your non-scanning hand on the patient’s abdomen to help mobilize the pelvic contents as the transvaginal probe slides across the organs
- use the probe as an “eye” while your palpating finger touches the cervix, uterus, ovaries, and any adnexal mass. Observe the mobility of these structures in relation to each other and the pelvic wall. This technique yields what is often referred to as the “sliding organs” sign. It is possible to identify pelvic adhesions (if the structures do not slide freely) or rule them out (if they do)
- pinpoint the origin of any pain the patient may have by touching the ovary, cervix, and any adnexal mass. This technique is important in cases of ectopic pregnancy, adnexal torsion, or inflammatory disease of the pelvis or adnexae.
Start with a basic scan of key structures
On the way “in” toward the adnexae, take the time to look at the bladder and urethra (FIGURE 1). Some common pathologies of the bladder are diverticulae; calculi; and a thick and vascular bladder wall suggestive of cancer or endometrioma. Ask the patient whether she has experienced any hematuria if any of these pathologies are detected.

FIGURE 1 Imaging the bladder
(A, B) The bladder (bl), urethra (u), vagina (v), and rectum (r) appear in their proper relation in this sagittal view. The posterior angle of the bladder is also apparent (arrow closing an angle of about 110°). (C) Excessive thickness of the bladder wall suggests that this patient has cystitis. (D) Coronal view of the bladder and urethra (solid arrows).
Also take a look at the cervix, searching for Nabothian cysts, endocervical polyps, extreme vascularization (a possible indicator of cervical cancer), and prolapsing submucous myomas (FIGURE 2).

FIGURE 2 Uncommon pathology
A submucous myoma prolapses into the cervical canal in a 13-week intrauterine pregnancy. (A) Grayscale sagittal image and (B) outline view of the same image. (C,D) Color and power Doppler images show the blood supply to the myoma from the uterine cavity.
While you are looking, attempt to scan both kidneys and Morrison’s pouch. Large adnexal masses or fibroids of the uterus may put pressure on the ureter, causing various degrees of hydronephrosis.
Sometimes, when the right kidney is correctly imaged below the liver, you may detect fluid in the space between them (called Morrison’s space). This information has clear value that may aid in diagnosing the main pathology (i.e., ruptured tubal pregnancy, ascites, etc.).
Imaging of the ovaries
The best way to scan the ovaries is to use a high-frequency (4–9 MHz) transvaginal probe. In general, as the frequency of the probe increases, so does resolution of the image—but the ability to penetrate tissue diminishes. For this reason, for abdominal imaging, a 3-MHz probe is often used. For a transvaginal scan, in which the probe can be placed near an ovary, a 5-MHz probe is common. And for a scan of, say, the parathyroid gland, a 12-MHz probe is utilized.
During the reproductive years, the ovaries can be localized by their sonographic markers—the follicles (FIGURE 3A). The ovaries usually lie near the large hypogastric blood vessels (FIGURE 3B). During the secretory phase of the cycle, look for the corpus luteum, switching on the color or power Doppler mode to help locate it (FIGURES 3C, 3D).
The ovaries usually can be distinguished by their relative anechoic sono-texture in juxtaposition to the surrounding, constantly peristalsing small bowel. This strategy is the only help for spotting the ovaries in menopause, when they lose their follicles.
The size of the ovaries may be an important indicator of pathology. During the reproductive years, mean size is 8 mL (standard deviation [SD], 2–3 mL; range, 5–15 mL). Post-menopausal ovaries are small, with a mean size of 3.6 mL (SD, 1.4 mL; range, 1–14 mL).

FIGURE 3 How to spot the ovaries
(A) Anechoic follicles are markers of the ovary during the reproductive years. (B) The ovaries in relation to the hypogastric vessels. (C) Gray-scale image of the corpus luteum and the same image in (D) color Doppler.
A word about terminology: Don’t call follicles “cysts”
During a normal menstrual cycle, one or more follicles mature, reaching about 2 to 2.5 cm in diameter around mid-cycle. Do not call these follicles “cysts” or “follicular cysts.” They are follicles. Calling them cysts, or even including the word cyst in their description, suggests to many gynecology and radiology providers—and to patients themselves—the idea of pathology.1
An exception to that rule: An ovary that is larger than 12 to 14 mL and has a hyperechoic hilus and more than 12 small (4–5 mm), peripherally pushed follicles is usually called “polycystic” (FIGURE 4).3 However, not every ovary that fulfills these sonographic criteria is indeed polycystic. At times normal ovaries may contain multiple follicles without any of the clinical or laboratory indications of a polycystic ovary. In these cases, the ovary may be of normal size and may lack a hyperechoic hilus with rich hilar vascularity. We term such ovaries “multicystic” in their appearance.

FIGURE 4 The polycystic ovary
(A) Gray-scale image of a polycystic ovary. The typical hyperechoic hilus is evident (H). (B) Gross pathologic section of a polycystic ovary. (C) 3D orthogonal planes of a large ovary with a multitude of small follicles pushed peripherally by a voluminous hyperechoic hilus. (D) 3D inversion rendering of the same ovary.
We employ 3D inversion rendering to better see and count the number of follicles (FIGURE 4D).
An ovary can have a polycystic appearance in the following clinical situations:
- hyperthyroid state (36% of affected women)
- hyperprolactinemia (50%)
- hypothalamic hypogonadism (24%).
It also can appear polycystic for no apparent reason.
Stay tuned!
Next month, we continue our focus on adnexal imaging by describing (and showing) nonneoplastic ovarian masses.
We want to hear from you! Tell us what you think.
1. Timor-Tritsch IE, Goldstein SR. The complexity of a complex mass and the simplicity of a simple cyst. J Ultrasound Med. 2005;24(3):255-258.
2. Timor-Tritsch IE, Monteagudo A, Rebarber A, Goldstein SR, Tsymbal T. Transrectal scanning an alternative when transvaginal scanning is not feasible. Ultrasound Obstet Gynecol. 2003;21(5):443-479.
3. Abdel Gadir A, Khatim MS, Mowafi RS, Alnaser HM, Muharib NS, Shaw RW. Implications of ultrasonically diagnosed polycystic ovaries. II. Studies of dynamic and pulsatile hormonal patterns. Human Reprod. 1992;7(4):458-461.
1. Timor-Tritsch IE, Goldstein SR. The complexity of a complex mass and the simplicity of a simple cyst. J Ultrasound Med. 2005;24(3):255-258.
2. Timor-Tritsch IE, Monteagudo A, Rebarber A, Goldstein SR, Tsymbal T. Transrectal scanning an alternative when transvaginal scanning is not feasible. Ultrasound Obstet Gynecol. 2003;21(5):443-479.
3. Abdel Gadir A, Khatim MS, Mowafi RS, Alnaser HM, Muharib NS, Shaw RW. Implications of ultrasonically diagnosed polycystic ovaries. II. Studies of dynamic and pulsatile hormonal patterns. Human Reprod. 1992;7(4):458-461.