User login
UPDATE: CONTRACEPTION
Adolescents, nulliparous women, and the IUD
The professional journals scattered on your desk every month always seem to have a review article, or a study, on “long-acting reversible contraception” (LARC)—and you’re not certain why. More and more, your younger patients are asking about intrauterine devices and contraceptive implants, but you’re unsure about the most up-to-date information on the safety of these methods in adolescents. Nulliparous women are inquiring about contraception with, for one, the levonorgestrel-releasing intrauterine system (LNG-IUS; Mirena), but the Mirena package insert tells you that they are not candidates for the method.
Does this sound familiar? How do you sort through all data and advice on IUD use in adolescents and nulliparas?
Fortunately for clinicians, a great deal of research in the last few years has focused on these topics. Recommendations and reviews have been published, and public health agencies have developed easily accessible guidelines for reference.
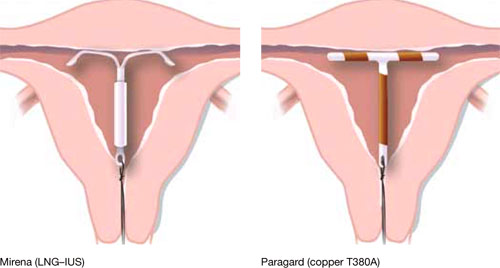
Our goal in this year’s installment of the Update on Contraception is to familiarize you with the evidence and ease any confusion, even misgivings, you might have about the two intrauterine devices available in the United States, the copper T380A (Paragard*) and Mirena* (FIGURE), especially in regard to their use in adolescents and nulliparous women.
*For ease of discussion only, we call these two systems by their brand names throughout.
FIGURE Two intrauterine devices for long-term contraception
There is real need for long-acting reversible contraception
The public health perspective
Consider these statistics about adult and adolescent women:
- Approximately 3 million pregnancies annually in the United States are unintended
- In almost half of those pregnancies, the woman undergoes an abortion
- Approximately one half of all US women have an unintended pregnancy by 45 years of age
- Given current statistics, nearly one third of all women will have chosen abortion by 45 years of age
- 80% of pregnancies in adolescents are unintended; 45% end in abortion
- 54% of women who have an abortion used a contraceptive method during the month they became pregnant—generally, a condom or an oral contraceptive.1,2
Patients’ perspective
More and more, adolescent women and nulliparous women request an IUD. Indeed, recent studies show that these populations are interested in the long-term protection that an IUD offers, and are likely to be more compliant with the method. Recent data from the National Survey of Family Growth showed an increase in IUD use in the United States: 5.5% of women who use birth control use an intrauterine device, and 14% of women who choose an IUD are adolescents.
A review of IUD use in adolescents found that, across six cohort studies and seven case reports, the continuation rate with an IUD after 1 year ranged from 48% to 88%—similar to, or better than, what is seen with oral contraceptives (OCs).3 Furthermore, two recent studies3,4 showed that:
- young nulliparous and parous women exhibit a positive attitude toward IUD use once they have been counseled on the risks and benefits of the device
- they desire effective long-term contraception
- more than 50% of the women who were surveyed thought positively about IUDs after being educated about them.
Intrauterine contraceptive use in the United States is very low compared to the rates in other developed countries—as noted, the rate here is 5.5%, for both types of IUD. Contrast that rate with what is documented in other nations: France, 20%; China, 34%; and Norway, 24%, for example. Across the developed world, IUD use is at 7.6%; in developing nations, the rate is even higher: 14.5 %.5,6
Women in the United States use the most effective forms of birth control at lower rates than the rest of the developed world; conversely, they choose permanent sterilization at a higher rate. When American women were asked, they expressed a desire for longer-acting contraception that is easy to use. Yet, they fail to take advantage of the options—often, because they lack information about them or have received erroneous education.2
Indeed, when providers of contraceptive services have been surveyed about barriers to IUD use, they point to women’s misconceptions about the devices and express their own concerns about the incidence of pelvic inflammatory disease (PID) and infertility; difficulty of insertion; and expulsion.
Findings of a survey of clinicians. In a 2008 poll of 816 health care providers (including 399 physicians and 402 advanced practice clinicians), 40% did not offer intrauterine contraception to any patients who sought contraception.7 Most (55%) providers considered less than one quarter of their patients to be a candidate for intrauterine contraception.
Furthermore, fewer than one half of providers considered nulliparous, immediate postpartum or post-abortion, or teenage patients to be a candidate for intrauterine contraception. They also thought that women who had a history of ectopic pregnancy or pelvic inflammatory disease, or who were HIV-positive, were not candidates for intrauterine contraception—despite recommendations by the Centers for Disease Control and Prevention and the World Health Organization to use intrauterine contraception in those populations (TABLE).8,9
TABLE
Medical eligibility criteria for using an IUD (in selected conditions)*
| Condition | Paragard | Mirena |
|---|---|---|
| Age | ||
| • Menarche to age 20 | 2 | 2 |
| • ≥Age 20 | 1 | 1 |
| Parity | ||
| • Nulliparous | 2 | 2 |
| • Parous | 1 | 1 |
| Postpartum (breastfeeding or not breastfeeding, including post-cesarean section) | ||
| • <10 min after placental delivery | 1 | 2 |
| • 10 min after placental delivery to 4 weeks | 2 | 2 |
| • ≥4 weeks | 1 | 1 |
| Postabortion | ||
| • First trimester | 1 | 1 |
| • Second trimester | 2 | 2 |
| Past ectopic pregnancy | 1 | 1 |
| Past pelvic inflammatory disease (PID) (assuming no current risk factors for sexually transmitted infection) | ||
| • With subsequent pregnancy | 1 | 1 |
| • Without subsequent pregnancy | 2 | 2 |
| Continuation with current PID, infection with Chlamydia trachomatis or Neisseria gonorrhoeae | 2 | 2 |
| HIV-infected | 2 | 2 |
| Obesity | 1 | 1 |
| Venous thromboembolic disease | ||
| • History of deep-venous thromboembolism (DVT) or pulmonary embolism (PE) | 1 | 2 |
| • High risk for DVT or Pe | 1 | 2 |
| • Acute DVT or Pe | 2 | 2 |
| Key 1 There is no restriction on the use of the contraceptive method for this condition 2 The advantages of using the contraceptive method generally outweigh its theoretical or proven risks in this condition 3 The theoretical or proven risks of the contraceptive method generally outweigh its benefits in this condition * Adapted from: centers for Disease control and Prevention. US Medical eligibility criteria for contraceptive Use, 2010. MMWR early release 2010;59:52-7. for other recommendations, see: centers for Disease control and Prevention.8 | ||
IUDs are safe
Do you relate to what respondents said in the survey just discussed? Do you have concerns about intrauterine contraception in adolescents or nulliparas, especially about:
- perforation at the time an IUD is placed
- risk of expulsion
- side effects
- risk of PID
- risk of infertility?
Let’s examine each of these concerns against the backdrop of clinical guidelines issued recently by the Society of Family Planning and the group’s analysis of the medical literature on which those guidelines are based.10
Perforation at placement. No studies have examined the rate of perforation during IUD placement in nulliparas or adolescents alone; we do know that the overall (i.e., for all women) risk of perforation when an IUD is inserted has been reported as zero to 1.3%. General studies of perforation include a very small number of nulliparas; results are difficult to generalize to a larger population. At least two ongoing large, multicenter trials include a large number of nulliparas; one of them includes adolescents.
Expulsion. The rate of IUD expulsion in parous women has varied across studies and types of IUDs. In a recent retrospective cohort study, nulliparous and parous women were compared for complications with both copper and levonorgestrel-releasing IUDs. Rates of expulsion for copper IUDs were 0 to 1.2% a year. Rates of expulsion for the levonorgestrel-releasing IUDs were 0 to 0.2% a year. Nulliparous women did not have more complications than parous women.11
A review of studies examining the expulsion rate with the copper IUD found a slightly higher rate in nulliparas, but the copper IUD that had been used in 19 of 20 of those studies was not Paragard, the only copper IUD available in the United States. In the one study included in the review that looked at Paragard, there was one expulsion in the nulliparous group and none in the parous group.12
Side effects. In a review of copper-based IUDs, removals for pain and bleeding were slightly higher in nulliparas. Again, the majority of these studies reviewed did not use Paragard. In the one study that did examine Paragard, there were no removals for bleeding or pain in nulliparas or multiparas.10
No studies have compared nulliparous and parous women in regard to side effects associated with Mirena.
Pelvic inflammatory disease. Misgivings that providers have about the IUD often hearken back to the Dalkon Shield, which had a multifilament string that allowed bacteria to climb from the vagina into the uterus, with damaging consequences. Current IUDs have a monofilament string; they do not increase the user’s risk of pelvic infection.
Through recent research on antibiotic prophylaxis for IUD insertion, we have learned that the risk of PID in this setting is not as great as once thought. Antibiotic prophylaxis is unnecessary for IUD insertion because cases of PID after IUD insertion occur infrequently, with or without an antibiotic. A randomized clinical trial of 1,833 patients treated with azithromycin or placebo before IUD insertion demonstrated this low risk of PID: Only one patient in each group was given a diagnosis of salpingitis during the 90-day period after insertion.13
In addition, a recent study found that subjects could be screened for gonorrhea and chlamydial infection when an IUD was being placed and treated after insertion if either of those tests was positive—without increasing their risk of PID.14
Mirena may, in fact, have a protective effect against infection. When the device was compared to a copper IUD (Nova-T; not available in the United States) in a randomized, comparative, multicenter trial, subjects in whom Mirena was inserted had a cumulative gross rate of PID of 0.5 at 36 months; Nova-T users had a rate of 2.0.15
As we well know, women who require protection from sexually transmitted infection (STI) need to have their partner use a condom. But condoms are not, comparatively, a very good method of contraception; for a woman who is at risk of STI and pregnancy, we need to consider what method she will use in addition to a condom to protect against pregnancy. Is she better off using a condom and an OC, or a condom and an IUD? The answer may well be that, because an IUD does not increase the risk of STI or PID and is more effective at preventing pregnancy than an OC, she would be better off using a condom plus IUD when it comes to protecting herself against STI and pregnancy.
Infertility. The risk of infertility has been linked to the risk of PID, which, simply, has been shown to be unfounded with an IUD.
In 2001, a cohort study focused on three groups of patients: women seeking treatment for primary infertility with diagnosed tubal occlusion; women seeking treatment for primary infertility without tubal occlusion; and primigravida pregnant women.16 In all three groups, the same percentage reported prior copper IUD use—suggesting no increased risk of either tubal or nontubal infertility among IUD users. This finding is in concordance with other studies that examined the risk of infertility among parous IUD users.17
Clinical guidelines from the Society of family planning
Based on the evidence reviewed by the Society of Family Planning (SFP) on the use of intrauterine contraception in nulliparous women, SFP offers recommendations.10
Level-A evidence is that:
- Mirena and Paragard are effective and safe for nulliparous women
- compared with other methods, IUDs have a comparable or higher continuation-of-use rate in nulliparous women
- IUDs do not increase the risk of pelvic infection or infertility. Mirena probably reduces users’ risk of infection.
Level-B evidence is that:
- because of the expulsion rate and bleeding profile, Mirena might be better tolerated than Paragard in nulliparas
- insertion of an IUD may be more challenging in nulliparous women; given the benefits, however, clinicians should not be discouraged from considering them as a first-line contraceptive choice in this population.
Level-C evidence is that:
- adolescent women should be considered a candidate for an IUD.
Are adolescents more likely to discontinue use of an IUD than they are known to discontinue OCs and injectable contraceptives?
According to ACOG’s most recent Committee Opinion on IUDs in adolescents,18 the rate of IUD discontinuation might be slightly higher because of side effects, but this problem might be alleviated by counseling patients about the rate of amenorrhea with Mirena and providing adequate education about the side effects seen with both IUDs.
The authors of ACOG’s Committee Opinion also recommend that clinicians be familiar with their state’s consent laws regarding adolescents and contraception.
The conclusion of the Committee?
The IUD is a highly effective method of contraception that is underused in the United States. Because adolescents contribute disproportionately to the epidemic of unintended pregnancy in this country, top-tier methods of contraception, including IUDs and implants, should be considered as first-line choices for both nulliparous and parous adolescents.
How do I put the IUD into practice for these populations?
Here are tips about placing an IUD in nulliparous or adolescent women, gleaned from practice. Consider discussing placement techniques with clinicians and using their experiences as a way of expanding your repertoire when dealing with a difficult insertion.
A small body of literature on misoprostol and ibuprofen, including two recent randomized controlled trials,19,20 has failed to show that pain associated with insertion is relieved using either treatment. Below, we offer several recommendations on this point.
Counsel the patient extensively about what to expect with an IUD. Namely:
- how the IUD is inserted, with attention to female anatomy
- the most common side effects, especially bleeding
- cramping and pain with insertion
- spotting after insertion
- the need to use back-up contraception
- the need to use a condom to prevent STI.
Have various items available, as needed, during insertion. This includes, but isn’t limited to:
- various-sized specula
- cervical dilators
- an examination table adjustable for height and position
- an assistant to reassure and comfort the patient and to assist you.
We’re out from under a dark cloud
It’s been a long road for US clinicians, coming back from the damage done by the Dalkon Shield to their interest in inserting IUDs in nulliparous and adolescent women. But we are gradually seeing a change in both physicians’ and patients’ opinions about using intrauterine devices for these populations.
Demand is growing in the United States for long-acting reversible contraception; we need to challenge our reservations and provide the care that our patients are requesting. The opinions and advice of our supporting professional organizations, based on the recent literature, point to the appropriateness of embracing IUDs for nulliparous and adolescent women.
We urge you: Heed the call.
We want to hear from you! Tell us what you think.
1. Guttmacher Institute. Facts on induced abortion in the United States. In Brief. http://www.guttmacher.org/pubs/
fb_induced_abortion.pdf. Published May 2010.
2. Stanwood NL, Bradley KA. Young pregnant women’s knowledge of modern intrauterine devices. Obstet Gynecol. 2006;108(6):1417-1422.
3. Deans EI, Grimes DA. Intrauterine devices for adolescents: a systematic review. Contraception. 2009;79(6):418-423.
4. Whitaker AK, Johnson LM, Harwood B, Chiappetta L, Creinin MD, Gold MA. Adolescent and young adult women’s knowledge of and attitudes toward the intrauterine device. Contraception. 2008;78(3):211-217.
5. United Nations Department of Economic and Social Affairs, Population Division. World contraceptive use 2005. http://www.un.org/esa/population/publications/contraceptive2005/2005_World_
Contraceptive_files/WallChart_WCU2005.pdf. Accessed May 25, 2010.
6. Sonfield A. For the Guttmacher Institute. Popularity disparity: attitudes about the IUD in Europe and the United States. Guttmacher Policy Review. 2007;10(4):http://www.guttmacher.org/pubs/gpr/10/4/gpr100419.pdf. Accessed May 21, 2010.
7. Harper CC, Blum M, de Bocanegra HT, et al. Challenges in translating evidence into practice: the provision of intrauterine contraception. Obstet Gynecol. 2008;111(6):1359-1369.
8. Centers for Disease Control and Prevention. U.S. medical eligibility criteria for contraceptive use, 2010. MMWR. 2010;59(Early Release; RR04):1-6.
9. World Health Organization, Department of Reproductive Health and Research Medical eligibility criteria for contraceptive use. Geneva: WHO; 2009.
10. Lyus R, Lohr P, Prager S. for Board of the Society of Family Planning. Use of the Mirena LNG-IUS and Paragard CuT380 devices in nulliparous women. Contraception. 2010;81(5):367-371.
11. Veldhuis HM, Vos AG, Lagro-Janssen AL. Complications of the intrauterine device in nulliparous and parous women. Eur J Gen Pract. 2004;10(3):82-87.
12. Hubacher D. Copper intrauterine device use by nulliparous women: review of side effects. Contraception. 2007;75(suppl 6):S8-S11.
13. Walsh T, Grimes DA, Frezieres R, et al. for IUD Study Group. Randomised controlled trial of prophylactic antibiotics before insertion of intrauterine devices. Lancet. 1998;351(9108):1005-1008.
14. Goodman S, Hendlish SK, Benedict C, Reeves MF, Pera-Floyd M, Foster-Rosales A. Increasing intrauterine contraception use by reducing barriers to post-abortal and interval insertion. Contraception. 2008;78(2):136-142.
15. Toivonen J, Luukkainen T, Allonen H. Protective effect of intrauterine release of levonorgestrel on pelvic infection: three years’ comparative experience of levonorgestrel- and copper-releasing intrauterine devices. Obstet Gynecol. 1991;77(2):261-264.
16. Hubacher D, Lara-Ricalde R, Taylor DJ, Guerra-Infante F, Guzmán-Rodrigues R. Use of copper intrauterine devices and the risk of tubal infertility among nulligravid women. N Engl J Med. 2001;345(8):561-567.
17. Hov GG, Skjeldestad FE, Hilstad T. Use of IUD and subsequent fertility—follow-up after participation in a randomized clinical trial. Contraception. 2007;75(2):88-92.
18. ACOG Committee Opinion No. 392, December 2007. Intrauterine device and adolescents. Obstet Gynecol. 2007;110(6):1493-1495.
19. Sääv I, Aronsson A, Marions L, et al. Cervical priming with sublingual misoprostol prior to insertion of an intrauterine device in nulliparous women: a randomized controlled trial. Hum Reprod. 2007;22(10):2647-2652.
20. Hubacher D, Reyes V, Lillo S, Zepeda A, Chen PL, Croxatto H. Pain from copper intrauterine device insertion: randomized trial of prophylactic ibuprofen. Am J Obstet Gynecol. 2006;195(5):1272-1277.
Adolescents, nulliparous women, and the IUD
The professional journals scattered on your desk every month always seem to have a review article, or a study, on “long-acting reversible contraception” (LARC)—and you’re not certain why. More and more, your younger patients are asking about intrauterine devices and contraceptive implants, but you’re unsure about the most up-to-date information on the safety of these methods in adolescents. Nulliparous women are inquiring about contraception with, for one, the levonorgestrel-releasing intrauterine system (LNG-IUS; Mirena), but the Mirena package insert tells you that they are not candidates for the method.
Does this sound familiar? How do you sort through all data and advice on IUD use in adolescents and nulliparas?
Fortunately for clinicians, a great deal of research in the last few years has focused on these topics. Recommendations and reviews have been published, and public health agencies have developed easily accessible guidelines for reference.
Our goal in this year’s installment of the Update on Contraception is to familiarize you with the evidence and ease any confusion, even misgivings, you might have about the two intrauterine devices available in the United States, the copper T380A (Paragard*) and Mirena* (FIGURE), especially in regard to their use in adolescents and nulliparous women.
*For ease of discussion only, we call these two systems by their brand names throughout.
FIGURE Two intrauterine devices for long-term contraception

There is real need for long-acting reversible contraception
The public health perspective
Consider these statistics about adult and adolescent women:
- Approximately 3 million pregnancies annually in the United States are unintended
- In almost half of those pregnancies, the woman undergoes an abortion
- Approximately one half of all US women have an unintended pregnancy by 45 years of age
- Given current statistics, nearly one third of all women will have chosen abortion by 45 years of age
- 80% of pregnancies in adolescents are unintended; 45% end in abortion
- 54% of women who have an abortion used a contraceptive method during the month they became pregnant—generally, a condom or an oral contraceptive.1,2
Patients’ perspective
More and more, adolescent women and nulliparous women request an IUD. Indeed, recent studies show that these populations are interested in the long-term protection that an IUD offers, and are likely to be more compliant with the method. Recent data from the National Survey of Family Growth showed an increase in IUD use in the United States: 5.5% of women who use birth control use an intrauterine device, and 14% of women who choose an IUD are adolescents.
A review of IUD use in adolescents found that, across six cohort studies and seven case reports, the continuation rate with an IUD after 1 year ranged from 48% to 88%—similar to, or better than, what is seen with oral contraceptives (OCs).3 Furthermore, two recent studies3,4 showed that:
- young nulliparous and parous women exhibit a positive attitude toward IUD use once they have been counseled on the risks and benefits of the device
- they desire effective long-term contraception
- more than 50% of the women who were surveyed thought positively about IUDs after being educated about them.
Intrauterine contraceptive use in the United States is very low compared to the rates in other developed countries—as noted, the rate here is 5.5%, for both types of IUD. Contrast that rate with what is documented in other nations: France, 20%; China, 34%; and Norway, 24%, for example. Across the developed world, IUD use is at 7.6%; in developing nations, the rate is even higher: 14.5 %.5,6
Women in the United States use the most effective forms of birth control at lower rates than the rest of the developed world; conversely, they choose permanent sterilization at a higher rate. When American women were asked, they expressed a desire for longer-acting contraception that is easy to use. Yet, they fail to take advantage of the options—often, because they lack information about them or have received erroneous education.2
Indeed, when providers of contraceptive services have been surveyed about barriers to IUD use, they point to women’s misconceptions about the devices and express their own concerns about the incidence of pelvic inflammatory disease (PID) and infertility; difficulty of insertion; and expulsion.
Findings of a survey of clinicians. In a 2008 poll of 816 health care providers (including 399 physicians and 402 advanced practice clinicians), 40% did not offer intrauterine contraception to any patients who sought contraception.7 Most (55%) providers considered less than one quarter of their patients to be a candidate for intrauterine contraception.
Furthermore, fewer than one half of providers considered nulliparous, immediate postpartum or post-abortion, or teenage patients to be a candidate for intrauterine contraception. They also thought that women who had a history of ectopic pregnancy or pelvic inflammatory disease, or who were HIV-positive, were not candidates for intrauterine contraception—despite recommendations by the Centers for Disease Control and Prevention and the World Health Organization to use intrauterine contraception in those populations (TABLE).8,9
TABLE
Medical eligibility criteria for using an IUD (in selected conditions)*
| Condition | Paragard | Mirena |
|---|---|---|
| Age | ||
| • Menarche to age 20 | 2 | 2 |
| • ≥Age 20 | 1 | 1 |
| Parity | ||
| • Nulliparous | 2 | 2 |
| • Parous | 1 | 1 |
| Postpartum (breastfeeding or not breastfeeding, including post-cesarean section) | ||
| • <10 min after placental delivery | 1 | 2 |
| • 10 min after placental delivery to 4 weeks | 2 | 2 |
| • ≥4 weeks | 1 | 1 |
| Postabortion | ||
| • First trimester | 1 | 1 |
| • Second trimester | 2 | 2 |
| Past ectopic pregnancy | 1 | 1 |
| Past pelvic inflammatory disease (PID) (assuming no current risk factors for sexually transmitted infection) | ||
| • With subsequent pregnancy | 1 | 1 |
| • Without subsequent pregnancy | 2 | 2 |
| Continuation with current PID, infection with Chlamydia trachomatis or Neisseria gonorrhoeae | 2 | 2 |
| HIV-infected | 2 | 2 |
| Obesity | 1 | 1 |
| Venous thromboembolic disease | ||
| • History of deep-venous thromboembolism (DVT) or pulmonary embolism (PE) | 1 | 2 |
| • High risk for DVT or Pe | 1 | 2 |
| • Acute DVT or Pe | 2 | 2 |
| Key 1 There is no restriction on the use of the contraceptive method for this condition 2 The advantages of using the contraceptive method generally outweigh its theoretical or proven risks in this condition 3 The theoretical or proven risks of the contraceptive method generally outweigh its benefits in this condition * Adapted from: centers for Disease control and Prevention. US Medical eligibility criteria for contraceptive Use, 2010. MMWR early release 2010;59:52-7. for other recommendations, see: centers for Disease control and Prevention.8 | ||
IUDs are safe
Do you relate to what respondents said in the survey just discussed? Do you have concerns about intrauterine contraception in adolescents or nulliparas, especially about:
- perforation at the time an IUD is placed
- risk of expulsion
- side effects
- risk of PID
- risk of infertility?
Let’s examine each of these concerns against the backdrop of clinical guidelines issued recently by the Society of Family Planning and the group’s analysis of the medical literature on which those guidelines are based.10
Perforation at placement. No studies have examined the rate of perforation during IUD placement in nulliparas or adolescents alone; we do know that the overall (i.e., for all women) risk of perforation when an IUD is inserted has been reported as zero to 1.3%. General studies of perforation include a very small number of nulliparas; results are difficult to generalize to a larger population. At least two ongoing large, multicenter trials include a large number of nulliparas; one of them includes adolescents.
Expulsion. The rate of IUD expulsion in parous women has varied across studies and types of IUDs. In a recent retrospective cohort study, nulliparous and parous women were compared for complications with both copper and levonorgestrel-releasing IUDs. Rates of expulsion for copper IUDs were 0 to 1.2% a year. Rates of expulsion for the levonorgestrel-releasing IUDs were 0 to 0.2% a year. Nulliparous women did not have more complications than parous women.11
A review of studies examining the expulsion rate with the copper IUD found a slightly higher rate in nulliparas, but the copper IUD that had been used in 19 of 20 of those studies was not Paragard, the only copper IUD available in the United States. In the one study included in the review that looked at Paragard, there was one expulsion in the nulliparous group and none in the parous group.12
Side effects. In a review of copper-based IUDs, removals for pain and bleeding were slightly higher in nulliparas. Again, the majority of these studies reviewed did not use Paragard. In the one study that did examine Paragard, there were no removals for bleeding or pain in nulliparas or multiparas.10
No studies have compared nulliparous and parous women in regard to side effects associated with Mirena.
Pelvic inflammatory disease. Misgivings that providers have about the IUD often hearken back to the Dalkon Shield, which had a multifilament string that allowed bacteria to climb from the vagina into the uterus, with damaging consequences. Current IUDs have a monofilament string; they do not increase the user’s risk of pelvic infection.
Through recent research on antibiotic prophylaxis for IUD insertion, we have learned that the risk of PID in this setting is not as great as once thought. Antibiotic prophylaxis is unnecessary for IUD insertion because cases of PID after IUD insertion occur infrequently, with or without an antibiotic. A randomized clinical trial of 1,833 patients treated with azithromycin or placebo before IUD insertion demonstrated this low risk of PID: Only one patient in each group was given a diagnosis of salpingitis during the 90-day period after insertion.13
In addition, a recent study found that subjects could be screened for gonorrhea and chlamydial infection when an IUD was being placed and treated after insertion if either of those tests was positive—without increasing their risk of PID.14
Mirena may, in fact, have a protective effect against infection. When the device was compared to a copper IUD (Nova-T; not available in the United States) in a randomized, comparative, multicenter trial, subjects in whom Mirena was inserted had a cumulative gross rate of PID of 0.5 at 36 months; Nova-T users had a rate of 2.0.15
As we well know, women who require protection from sexually transmitted infection (STI) need to have their partner use a condom. But condoms are not, comparatively, a very good method of contraception; for a woman who is at risk of STI and pregnancy, we need to consider what method she will use in addition to a condom to protect against pregnancy. Is she better off using a condom and an OC, or a condom and an IUD? The answer may well be that, because an IUD does not increase the risk of STI or PID and is more effective at preventing pregnancy than an OC, she would be better off using a condom plus IUD when it comes to protecting herself against STI and pregnancy.
Infertility. The risk of infertility has been linked to the risk of PID, which, simply, has been shown to be unfounded with an IUD.
In 2001, a cohort study focused on three groups of patients: women seeking treatment for primary infertility with diagnosed tubal occlusion; women seeking treatment for primary infertility without tubal occlusion; and primigravida pregnant women.16 In all three groups, the same percentage reported prior copper IUD use—suggesting no increased risk of either tubal or nontubal infertility among IUD users. This finding is in concordance with other studies that examined the risk of infertility among parous IUD users.17
Clinical guidelines from the Society of family planning
Based on the evidence reviewed by the Society of Family Planning (SFP) on the use of intrauterine contraception in nulliparous women, SFP offers recommendations.10
Level-A evidence is that:
- Mirena and Paragard are effective and safe for nulliparous women
- compared with other methods, IUDs have a comparable or higher continuation-of-use rate in nulliparous women
- IUDs do not increase the risk of pelvic infection or infertility. Mirena probably reduces users’ risk of infection.
Level-B evidence is that:
- because of the expulsion rate and bleeding profile, Mirena might be better tolerated than Paragard in nulliparas
- insertion of an IUD may be more challenging in nulliparous women; given the benefits, however, clinicians should not be discouraged from considering them as a first-line contraceptive choice in this population.
Level-C evidence is that:
- adolescent women should be considered a candidate for an IUD.
Are adolescents more likely to discontinue use of an IUD than they are known to discontinue OCs and injectable contraceptives?
According to ACOG’s most recent Committee Opinion on IUDs in adolescents,18 the rate of IUD discontinuation might be slightly higher because of side effects, but this problem might be alleviated by counseling patients about the rate of amenorrhea with Mirena and providing adequate education about the side effects seen with both IUDs.
The authors of ACOG’s Committee Opinion also recommend that clinicians be familiar with their state’s consent laws regarding adolescents and contraception.
The conclusion of the Committee?
The IUD is a highly effective method of contraception that is underused in the United States. Because adolescents contribute disproportionately to the epidemic of unintended pregnancy in this country, top-tier methods of contraception, including IUDs and implants, should be considered as first-line choices for both nulliparous and parous adolescents.
How do I put the IUD into practice for these populations?
Here are tips about placing an IUD in nulliparous or adolescent women, gleaned from practice. Consider discussing placement techniques with clinicians and using their experiences as a way of expanding your repertoire when dealing with a difficult insertion.
A small body of literature on misoprostol and ibuprofen, including two recent randomized controlled trials,19,20 has failed to show that pain associated with insertion is relieved using either treatment. Below, we offer several recommendations on this point.
Counsel the patient extensively about what to expect with an IUD. Namely:
- how the IUD is inserted, with attention to female anatomy
- the most common side effects, especially bleeding
- cramping and pain with insertion
- spotting after insertion
- the need to use back-up contraception
- the need to use a condom to prevent STI.
Have various items available, as needed, during insertion. This includes, but isn’t limited to:
- various-sized specula
- cervical dilators
- an examination table adjustable for height and position
- an assistant to reassure and comfort the patient and to assist you.
We’re out from under a dark cloud
It’s been a long road for US clinicians, coming back from the damage done by the Dalkon Shield to their interest in inserting IUDs in nulliparous and adolescent women. But we are gradually seeing a change in both physicians’ and patients’ opinions about using intrauterine devices for these populations.
Demand is growing in the United States for long-acting reversible contraception; we need to challenge our reservations and provide the care that our patients are requesting. The opinions and advice of our supporting professional organizations, based on the recent literature, point to the appropriateness of embracing IUDs for nulliparous and adolescent women.
We urge you: Heed the call.
We want to hear from you! Tell us what you think.
Adolescents, nulliparous women, and the IUD
The professional journals scattered on your desk every month always seem to have a review article, or a study, on “long-acting reversible contraception” (LARC)—and you’re not certain why. More and more, your younger patients are asking about intrauterine devices and contraceptive implants, but you’re unsure about the most up-to-date information on the safety of these methods in adolescents. Nulliparous women are inquiring about contraception with, for one, the levonorgestrel-releasing intrauterine system (LNG-IUS; Mirena), but the Mirena package insert tells you that they are not candidates for the method.
Does this sound familiar? How do you sort through all data and advice on IUD use in adolescents and nulliparas?
Fortunately for clinicians, a great deal of research in the last few years has focused on these topics. Recommendations and reviews have been published, and public health agencies have developed easily accessible guidelines for reference.
Our goal in this year’s installment of the Update on Contraception is to familiarize you with the evidence and ease any confusion, even misgivings, you might have about the two intrauterine devices available in the United States, the copper T380A (Paragard*) and Mirena* (FIGURE), especially in regard to their use in adolescents and nulliparous women.
*For ease of discussion only, we call these two systems by their brand names throughout.
FIGURE Two intrauterine devices for long-term contraception

There is real need for long-acting reversible contraception
The public health perspective
Consider these statistics about adult and adolescent women:
- Approximately 3 million pregnancies annually in the United States are unintended
- In almost half of those pregnancies, the woman undergoes an abortion
- Approximately one half of all US women have an unintended pregnancy by 45 years of age
- Given current statistics, nearly one third of all women will have chosen abortion by 45 years of age
- 80% of pregnancies in adolescents are unintended; 45% end in abortion
- 54% of women who have an abortion used a contraceptive method during the month they became pregnant—generally, a condom or an oral contraceptive.1,2
Patients’ perspective
More and more, adolescent women and nulliparous women request an IUD. Indeed, recent studies show that these populations are interested in the long-term protection that an IUD offers, and are likely to be more compliant with the method. Recent data from the National Survey of Family Growth showed an increase in IUD use in the United States: 5.5% of women who use birth control use an intrauterine device, and 14% of women who choose an IUD are adolescents.
A review of IUD use in adolescents found that, across six cohort studies and seven case reports, the continuation rate with an IUD after 1 year ranged from 48% to 88%—similar to, or better than, what is seen with oral contraceptives (OCs).3 Furthermore, two recent studies3,4 showed that:
- young nulliparous and parous women exhibit a positive attitude toward IUD use once they have been counseled on the risks and benefits of the device
- they desire effective long-term contraception
- more than 50% of the women who were surveyed thought positively about IUDs after being educated about them.
Intrauterine contraceptive use in the United States is very low compared to the rates in other developed countries—as noted, the rate here is 5.5%, for both types of IUD. Contrast that rate with what is documented in other nations: France, 20%; China, 34%; and Norway, 24%, for example. Across the developed world, IUD use is at 7.6%; in developing nations, the rate is even higher: 14.5 %.5,6
Women in the United States use the most effective forms of birth control at lower rates than the rest of the developed world; conversely, they choose permanent sterilization at a higher rate. When American women were asked, they expressed a desire for longer-acting contraception that is easy to use. Yet, they fail to take advantage of the options—often, because they lack information about them or have received erroneous education.2
Indeed, when providers of contraceptive services have been surveyed about barriers to IUD use, they point to women’s misconceptions about the devices and express their own concerns about the incidence of pelvic inflammatory disease (PID) and infertility; difficulty of insertion; and expulsion.
Findings of a survey of clinicians. In a 2008 poll of 816 health care providers (including 399 physicians and 402 advanced practice clinicians), 40% did not offer intrauterine contraception to any patients who sought contraception.7 Most (55%) providers considered less than one quarter of their patients to be a candidate for intrauterine contraception.
Furthermore, fewer than one half of providers considered nulliparous, immediate postpartum or post-abortion, or teenage patients to be a candidate for intrauterine contraception. They also thought that women who had a history of ectopic pregnancy or pelvic inflammatory disease, or who were HIV-positive, were not candidates for intrauterine contraception—despite recommendations by the Centers for Disease Control and Prevention and the World Health Organization to use intrauterine contraception in those populations (TABLE).8,9
TABLE
Medical eligibility criteria for using an IUD (in selected conditions)*
| Condition | Paragard | Mirena |
|---|---|---|
| Age | ||
| • Menarche to age 20 | 2 | 2 |
| • ≥Age 20 | 1 | 1 |
| Parity | ||
| • Nulliparous | 2 | 2 |
| • Parous | 1 | 1 |
| Postpartum (breastfeeding or not breastfeeding, including post-cesarean section) | ||
| • <10 min after placental delivery | 1 | 2 |
| • 10 min after placental delivery to 4 weeks | 2 | 2 |
| • ≥4 weeks | 1 | 1 |
| Postabortion | ||
| • First trimester | 1 | 1 |
| • Second trimester | 2 | 2 |
| Past ectopic pregnancy | 1 | 1 |
| Past pelvic inflammatory disease (PID) (assuming no current risk factors for sexually transmitted infection) | ||
| • With subsequent pregnancy | 1 | 1 |
| • Without subsequent pregnancy | 2 | 2 |
| Continuation with current PID, infection with Chlamydia trachomatis or Neisseria gonorrhoeae | 2 | 2 |
| HIV-infected | 2 | 2 |
| Obesity | 1 | 1 |
| Venous thromboembolic disease | ||
| • History of deep-venous thromboembolism (DVT) or pulmonary embolism (PE) | 1 | 2 |
| • High risk for DVT or Pe | 1 | 2 |
| • Acute DVT or Pe | 2 | 2 |
| Key 1 There is no restriction on the use of the contraceptive method for this condition 2 The advantages of using the contraceptive method generally outweigh its theoretical or proven risks in this condition 3 The theoretical or proven risks of the contraceptive method generally outweigh its benefits in this condition * Adapted from: centers for Disease control and Prevention. US Medical eligibility criteria for contraceptive Use, 2010. MMWR early release 2010;59:52-7. for other recommendations, see: centers for Disease control and Prevention.8 | ||
IUDs are safe
Do you relate to what respondents said in the survey just discussed? Do you have concerns about intrauterine contraception in adolescents or nulliparas, especially about:
- perforation at the time an IUD is placed
- risk of expulsion
- side effects
- risk of PID
- risk of infertility?
Let’s examine each of these concerns against the backdrop of clinical guidelines issued recently by the Society of Family Planning and the group’s analysis of the medical literature on which those guidelines are based.10
Perforation at placement. No studies have examined the rate of perforation during IUD placement in nulliparas or adolescents alone; we do know that the overall (i.e., for all women) risk of perforation when an IUD is inserted has been reported as zero to 1.3%. General studies of perforation include a very small number of nulliparas; results are difficult to generalize to a larger population. At least two ongoing large, multicenter trials include a large number of nulliparas; one of them includes adolescents.
Expulsion. The rate of IUD expulsion in parous women has varied across studies and types of IUDs. In a recent retrospective cohort study, nulliparous and parous women were compared for complications with both copper and levonorgestrel-releasing IUDs. Rates of expulsion for copper IUDs were 0 to 1.2% a year. Rates of expulsion for the levonorgestrel-releasing IUDs were 0 to 0.2% a year. Nulliparous women did not have more complications than parous women.11
A review of studies examining the expulsion rate with the copper IUD found a slightly higher rate in nulliparas, but the copper IUD that had been used in 19 of 20 of those studies was not Paragard, the only copper IUD available in the United States. In the one study included in the review that looked at Paragard, there was one expulsion in the nulliparous group and none in the parous group.12
Side effects. In a review of copper-based IUDs, removals for pain and bleeding were slightly higher in nulliparas. Again, the majority of these studies reviewed did not use Paragard. In the one study that did examine Paragard, there were no removals for bleeding or pain in nulliparas or multiparas.10
No studies have compared nulliparous and parous women in regard to side effects associated with Mirena.
Pelvic inflammatory disease. Misgivings that providers have about the IUD often hearken back to the Dalkon Shield, which had a multifilament string that allowed bacteria to climb from the vagina into the uterus, with damaging consequences. Current IUDs have a monofilament string; they do not increase the user’s risk of pelvic infection.
Through recent research on antibiotic prophylaxis for IUD insertion, we have learned that the risk of PID in this setting is not as great as once thought. Antibiotic prophylaxis is unnecessary for IUD insertion because cases of PID after IUD insertion occur infrequently, with or without an antibiotic. A randomized clinical trial of 1,833 patients treated with azithromycin or placebo before IUD insertion demonstrated this low risk of PID: Only one patient in each group was given a diagnosis of salpingitis during the 90-day period after insertion.13
In addition, a recent study found that subjects could be screened for gonorrhea and chlamydial infection when an IUD was being placed and treated after insertion if either of those tests was positive—without increasing their risk of PID.14
Mirena may, in fact, have a protective effect against infection. When the device was compared to a copper IUD (Nova-T; not available in the United States) in a randomized, comparative, multicenter trial, subjects in whom Mirena was inserted had a cumulative gross rate of PID of 0.5 at 36 months; Nova-T users had a rate of 2.0.15
As we well know, women who require protection from sexually transmitted infection (STI) need to have their partner use a condom. But condoms are not, comparatively, a very good method of contraception; for a woman who is at risk of STI and pregnancy, we need to consider what method she will use in addition to a condom to protect against pregnancy. Is she better off using a condom and an OC, or a condom and an IUD? The answer may well be that, because an IUD does not increase the risk of STI or PID and is more effective at preventing pregnancy than an OC, she would be better off using a condom plus IUD when it comes to protecting herself against STI and pregnancy.
Infertility. The risk of infertility has been linked to the risk of PID, which, simply, has been shown to be unfounded with an IUD.
In 2001, a cohort study focused on three groups of patients: women seeking treatment for primary infertility with diagnosed tubal occlusion; women seeking treatment for primary infertility without tubal occlusion; and primigravida pregnant women.16 In all three groups, the same percentage reported prior copper IUD use—suggesting no increased risk of either tubal or nontubal infertility among IUD users. This finding is in concordance with other studies that examined the risk of infertility among parous IUD users.17
Clinical guidelines from the Society of family planning
Based on the evidence reviewed by the Society of Family Planning (SFP) on the use of intrauterine contraception in nulliparous women, SFP offers recommendations.10
Level-A evidence is that:
- Mirena and Paragard are effective and safe for nulliparous women
- compared with other methods, IUDs have a comparable or higher continuation-of-use rate in nulliparous women
- IUDs do not increase the risk of pelvic infection or infertility. Mirena probably reduces users’ risk of infection.
Level-B evidence is that:
- because of the expulsion rate and bleeding profile, Mirena might be better tolerated than Paragard in nulliparas
- insertion of an IUD may be more challenging in nulliparous women; given the benefits, however, clinicians should not be discouraged from considering them as a first-line contraceptive choice in this population.
Level-C evidence is that:
- adolescent women should be considered a candidate for an IUD.
Are adolescents more likely to discontinue use of an IUD than they are known to discontinue OCs and injectable contraceptives?
According to ACOG’s most recent Committee Opinion on IUDs in adolescents,18 the rate of IUD discontinuation might be slightly higher because of side effects, but this problem might be alleviated by counseling patients about the rate of amenorrhea with Mirena and providing adequate education about the side effects seen with both IUDs.
The authors of ACOG’s Committee Opinion also recommend that clinicians be familiar with their state’s consent laws regarding adolescents and contraception.
The conclusion of the Committee?
The IUD is a highly effective method of contraception that is underused in the United States. Because adolescents contribute disproportionately to the epidemic of unintended pregnancy in this country, top-tier methods of contraception, including IUDs and implants, should be considered as first-line choices for both nulliparous and parous adolescents.
How do I put the IUD into practice for these populations?
Here are tips about placing an IUD in nulliparous or adolescent women, gleaned from practice. Consider discussing placement techniques with clinicians and using their experiences as a way of expanding your repertoire when dealing with a difficult insertion.
A small body of literature on misoprostol and ibuprofen, including two recent randomized controlled trials,19,20 has failed to show that pain associated with insertion is relieved using either treatment. Below, we offer several recommendations on this point.
Counsel the patient extensively about what to expect with an IUD. Namely:
- how the IUD is inserted, with attention to female anatomy
- the most common side effects, especially bleeding
- cramping and pain with insertion
- spotting after insertion
- the need to use back-up contraception
- the need to use a condom to prevent STI.
Have various items available, as needed, during insertion. This includes, but isn’t limited to:
- various-sized specula
- cervical dilators
- an examination table adjustable for height and position
- an assistant to reassure and comfort the patient and to assist you.
We’re out from under a dark cloud
It’s been a long road for US clinicians, coming back from the damage done by the Dalkon Shield to their interest in inserting IUDs in nulliparous and adolescent women. But we are gradually seeing a change in both physicians’ and patients’ opinions about using intrauterine devices for these populations.
Demand is growing in the United States for long-acting reversible contraception; we need to challenge our reservations and provide the care that our patients are requesting. The opinions and advice of our supporting professional organizations, based on the recent literature, point to the appropriateness of embracing IUDs for nulliparous and adolescent women.
We urge you: Heed the call.
We want to hear from you! Tell us what you think.
1. Guttmacher Institute. Facts on induced abortion in the United States. In Brief. http://www.guttmacher.org/pubs/
fb_induced_abortion.pdf. Published May 2010.
2. Stanwood NL, Bradley KA. Young pregnant women’s knowledge of modern intrauterine devices. Obstet Gynecol. 2006;108(6):1417-1422.
3. Deans EI, Grimes DA. Intrauterine devices for adolescents: a systematic review. Contraception. 2009;79(6):418-423.
4. Whitaker AK, Johnson LM, Harwood B, Chiappetta L, Creinin MD, Gold MA. Adolescent and young adult women’s knowledge of and attitudes toward the intrauterine device. Contraception. 2008;78(3):211-217.
5. United Nations Department of Economic and Social Affairs, Population Division. World contraceptive use 2005. http://www.un.org/esa/population/publications/contraceptive2005/2005_World_
Contraceptive_files/WallChart_WCU2005.pdf. Accessed May 25, 2010.
6. Sonfield A. For the Guttmacher Institute. Popularity disparity: attitudes about the IUD in Europe and the United States. Guttmacher Policy Review. 2007;10(4):http://www.guttmacher.org/pubs/gpr/10/4/gpr100419.pdf. Accessed May 21, 2010.
7. Harper CC, Blum M, de Bocanegra HT, et al. Challenges in translating evidence into practice: the provision of intrauterine contraception. Obstet Gynecol. 2008;111(6):1359-1369.
8. Centers for Disease Control and Prevention. U.S. medical eligibility criteria for contraceptive use, 2010. MMWR. 2010;59(Early Release; RR04):1-6.
9. World Health Organization, Department of Reproductive Health and Research Medical eligibility criteria for contraceptive use. Geneva: WHO; 2009.
10. Lyus R, Lohr P, Prager S. for Board of the Society of Family Planning. Use of the Mirena LNG-IUS and Paragard CuT380 devices in nulliparous women. Contraception. 2010;81(5):367-371.
11. Veldhuis HM, Vos AG, Lagro-Janssen AL. Complications of the intrauterine device in nulliparous and parous women. Eur J Gen Pract. 2004;10(3):82-87.
12. Hubacher D. Copper intrauterine device use by nulliparous women: review of side effects. Contraception. 2007;75(suppl 6):S8-S11.
13. Walsh T, Grimes DA, Frezieres R, et al. for IUD Study Group. Randomised controlled trial of prophylactic antibiotics before insertion of intrauterine devices. Lancet. 1998;351(9108):1005-1008.
14. Goodman S, Hendlish SK, Benedict C, Reeves MF, Pera-Floyd M, Foster-Rosales A. Increasing intrauterine contraception use by reducing barriers to post-abortal and interval insertion. Contraception. 2008;78(2):136-142.
15. Toivonen J, Luukkainen T, Allonen H. Protective effect of intrauterine release of levonorgestrel on pelvic infection: three years’ comparative experience of levonorgestrel- and copper-releasing intrauterine devices. Obstet Gynecol. 1991;77(2):261-264.
16. Hubacher D, Lara-Ricalde R, Taylor DJ, Guerra-Infante F, Guzmán-Rodrigues R. Use of copper intrauterine devices and the risk of tubal infertility among nulligravid women. N Engl J Med. 2001;345(8):561-567.
17. Hov GG, Skjeldestad FE, Hilstad T. Use of IUD and subsequent fertility—follow-up after participation in a randomized clinical trial. Contraception. 2007;75(2):88-92.
18. ACOG Committee Opinion No. 392, December 2007. Intrauterine device and adolescents. Obstet Gynecol. 2007;110(6):1493-1495.
19. Sääv I, Aronsson A, Marions L, et al. Cervical priming with sublingual misoprostol prior to insertion of an intrauterine device in nulliparous women: a randomized controlled trial. Hum Reprod. 2007;22(10):2647-2652.
20. Hubacher D, Reyes V, Lillo S, Zepeda A, Chen PL, Croxatto H. Pain from copper intrauterine device insertion: randomized trial of prophylactic ibuprofen. Am J Obstet Gynecol. 2006;195(5):1272-1277.
1. Guttmacher Institute. Facts on induced abortion in the United States. In Brief. http://www.guttmacher.org/pubs/
fb_induced_abortion.pdf. Published May 2010.
2. Stanwood NL, Bradley KA. Young pregnant women’s knowledge of modern intrauterine devices. Obstet Gynecol. 2006;108(6):1417-1422.
3. Deans EI, Grimes DA. Intrauterine devices for adolescents: a systematic review. Contraception. 2009;79(6):418-423.
4. Whitaker AK, Johnson LM, Harwood B, Chiappetta L, Creinin MD, Gold MA. Adolescent and young adult women’s knowledge of and attitudes toward the intrauterine device. Contraception. 2008;78(3):211-217.
5. United Nations Department of Economic and Social Affairs, Population Division. World contraceptive use 2005. http://www.un.org/esa/population/publications/contraceptive2005/2005_World_
Contraceptive_files/WallChart_WCU2005.pdf. Accessed May 25, 2010.
6. Sonfield A. For the Guttmacher Institute. Popularity disparity: attitudes about the IUD in Europe and the United States. Guttmacher Policy Review. 2007;10(4):http://www.guttmacher.org/pubs/gpr/10/4/gpr100419.pdf. Accessed May 21, 2010.
7. Harper CC, Blum M, de Bocanegra HT, et al. Challenges in translating evidence into practice: the provision of intrauterine contraception. Obstet Gynecol. 2008;111(6):1359-1369.
8. Centers for Disease Control and Prevention. U.S. medical eligibility criteria for contraceptive use, 2010. MMWR. 2010;59(Early Release; RR04):1-6.
9. World Health Organization, Department of Reproductive Health and Research Medical eligibility criteria for contraceptive use. Geneva: WHO; 2009.
10. Lyus R, Lohr P, Prager S. for Board of the Society of Family Planning. Use of the Mirena LNG-IUS and Paragard CuT380 devices in nulliparous women. Contraception. 2010;81(5):367-371.
11. Veldhuis HM, Vos AG, Lagro-Janssen AL. Complications of the intrauterine device in nulliparous and parous women. Eur J Gen Pract. 2004;10(3):82-87.
12. Hubacher D. Copper intrauterine device use by nulliparous women: review of side effects. Contraception. 2007;75(suppl 6):S8-S11.
13. Walsh T, Grimes DA, Frezieres R, et al. for IUD Study Group. Randomised controlled trial of prophylactic antibiotics before insertion of intrauterine devices. Lancet. 1998;351(9108):1005-1008.
14. Goodman S, Hendlish SK, Benedict C, Reeves MF, Pera-Floyd M, Foster-Rosales A. Increasing intrauterine contraception use by reducing barriers to post-abortal and interval insertion. Contraception. 2008;78(2):136-142.
15. Toivonen J, Luukkainen T, Allonen H. Protective effect of intrauterine release of levonorgestrel on pelvic infection: three years’ comparative experience of levonorgestrel- and copper-releasing intrauterine devices. Obstet Gynecol. 1991;77(2):261-264.
16. Hubacher D, Lara-Ricalde R, Taylor DJ, Guerra-Infante F, Guzmán-Rodrigues R. Use of copper intrauterine devices and the risk of tubal infertility among nulligravid women. N Engl J Med. 2001;345(8):561-567.
17. Hov GG, Skjeldestad FE, Hilstad T. Use of IUD and subsequent fertility—follow-up after participation in a randomized clinical trial. Contraception. 2007;75(2):88-92.
18. ACOG Committee Opinion No. 392, December 2007. Intrauterine device and adolescents. Obstet Gynecol. 2007;110(6):1493-1495.
19. Sääv I, Aronsson A, Marions L, et al. Cervical priming with sublingual misoprostol prior to insertion of an intrauterine device in nulliparous women: a randomized controlled trial. Hum Reprod. 2007;22(10):2647-2652.
20. Hubacher D, Reyes V, Lillo S, Zepeda A, Chen PL, Croxatto H. Pain from copper intrauterine device insertion: randomized trial of prophylactic ibuprofen. Am J Obstet Gynecol. 2006;195(5):1272-1277.
2 HPV vaccines, 7 questions that you need answered
Not long ago (in medical years), we were still trying to discover the cause of cervical cancer. Today, not only do we know that cause to be persistent human papillomavirus (HPV) infection, but we have two vaccines at our disposal to prevent the primary oncogenic strains of the virus.
We’ve come a long way.
The availability of two vaccines raises questions, however. What kind of data do we have on the bivalent (Cervarix, GlaxoSmithKline) and quadrivalent (Gardasil, Merck) vaccines so far? Is one of them clearly superior to the other? If not, what population is each vaccine best suited for—and how do we counsel patients about their options?
To address these and other questions, OBG Management Contributing Editor Neal M. Lonky, MD, MPH, assembled a panel of physicians who have expertise in cervical disease detection and prevention and asked them to sift the data that have accumulated thus far. In the discussion that follows, they touch on long-term efficacy, the likely impact of the vaccines on cervical cancer screening, and other aspects of disease prevention in the era of HPV vaccination.

Juan C. Felix, MD
Professor of Clinical Pathology and Obstetrics and Gynecology; Director of Cytopathology fellowship; and Chief of Gynecologic Pathology at the Keck School of Medicine, University of Southern California; and Chief of Cytopathology at Los Angeles County and University of Southern California Medical Center in Los Angeles.
Dr. Felix reports that he is a speaker for Merck and GlaxoSmithKline.

Diane M. Harper, MD, MS, MPH
Director of the Gynecologic Cancer Prevention Research Group and Professor of Obstetrics and Gynecology, Community and family Medicine, and Informatics and Personalized Medicine at the University of Missouri–Kansas City School of Medicine.
Dr. Harper reports that she has served as a speaker and advisor for Merck and GlaxoSmithKline, and that the institutions at which she conducted HPV vaccination trials have received funding from Merck and GlaxoSmithKline.

Warner K. Huh, MD
Associate Professor in the Department of Obstetrics and Gynecology, and Associate Scientist at the Comprehensive Cancer Center at the University of Alabama– Birmingham.
Dr. Huh reports that he receives grant or research support from and is a speaker and consultant to Merck and GlaxoSmithKline.

Karen K. Smith-McCune, MD, PhD
John Kerner Endowed Chair of Gynecologic Oncology, Director of the Dysplasia Clinic, and Professor of Obstetrics, Gynecology, and Reproductive Sciences at the University of California–San francisco.
Dr. Smith-McCune reports she has performed unpaid consulting for OncoHealth Inc. and is planning to join its Scientific Advisory Board.
1. How were the vaccines developed?
Neal M. Lonky, MD, MPH: What should clinicians know about the development, function, and mechanism of action of the two HPV vaccines?
Warner K. Huh, MD: The bivalent and quadrivalent vaccines are both excellent products, and their respective Phase-3 trials demonstrate that they provide impressive protection against HPV, particularly among women who test negative (by polymerase chain reaction) for the specific HPV types contained within the vaccines.1-3
Cervarix protects against HPV types 16 and 18, whereas Gardasil is effective against HPV types 6, 11, 16, and 18.
Dr. Lonky: Do the vaccines function similarly?
Diane M. Harper, MD, MS, MPH: Yes. Both stimulate an immediate antibody response in the woman who is not infected with the relevant virus and are effective in preventing cervical intraepithelial neoplasia grade 2 and higher (CIN 2+), as well as persistent infection, caused by vaccine-related and cross-protected HPV types. The quality of the antibody response is best for HPV 16 for both vaccines. The quality of the antibody response for HPV 6, 11, and 18 for Gardasil is much poorer than its response for HPV 16. Cervarix induces an equally high and sustained antibody response for HPV 18 as for HPV 16.
Juan C. Felix, MD: Both vaccines are based on the same virus-like particles (VLP). The functionality of the vaccines is, therefore, mainly dependent on the dosage of VLP and the adjuvant used. Gardasil uses a proprietary aluminum sulfate adjuvant, whereas Cervarix uses aluminum hydroxide and monophosphoryl lipid A.
Karen K. Smith-McCune, MD, PhD: Both adjuvants have an extensive track record of safety and efficacy in other vaccines. Because they have different structures, however, they may have varying effects on many components of the immune response elicited by the L1 antigens.
Dr. Harper: Both adjuvants contain aluminum, which has so far proved to be safe despite the newly established association between high aluminum intake and Alzheimer’s disease.
Dr. Lonky: Were there any notable challenges in developing the vaccines?
Dr. Harper: It was difficult to formulate the appropriate dosages of VLP in Gardasil. Higher dosages of HPV 11 and 16 were needed to prevent cross-inhibition by HPV 6 and 18. As a result, the antigenic protein component of Gardasil that is necessary to effect an immunologic antibody response is high, at 120 μg. In Cervarix, the antigenic VLP load is 20 μg each for HPV 16 and 18.
Dr. Lonky: What is the significance of the different VLP loads?
Dr. Harper: Side effects, such as autoimmune neurologic demyelination, albeit rare, have been associated with a higher antigenic protein load. Multiple reports of autoimmune demyelinating diseases—including paralysis, blindness, and death—have been published by neurologists in regard to Gardasil.4,5 Others have shown that young girls are more at risk than young boys for these neurologic side effects.6
Dr. Felix: Some data suggest that the two vaccine formulations interact differently with the human immune system. In a head-to-head trial funded by GlaxoSmithKline, Cervarix produced higher total and neutralizing antibody titers than Gardasil did.7
Although higher immunogenicity is generally thought to be beneficial, the ultimate determinant of a vaccine’s success is its efficacy—and duration of that efficacy—in clinical trials and follow-up of vaccinated populations. So far, Cervarix has demonstrated efficacy through 8.4 years in its follow-up cohort.8 Similarly, Gardasil has proved to be effective after 5 years of follow-up, with no incident cases of cervical cancer reported in the vaccinated arm.9
2. Does either vaccine offer “extra” immunity?
Dr. Lonky: What is the potential for overlapping immunity to other high-risk viral types with these vaccines?
Dr. Harper: It is quite clear from pivotal trials of both vaccines that Gardasil produces efficacy of 46% against persistent infection caused by HPV 31. Data from the pivotal Phase-3 trial of Gardasil also show that it offers no protection against persistent infection with HPV 45, an important cause of adenocarcinoma.10
In contrast, Cervarix demonstrates substantial efficacy against both persistent infection and CIN 2+ disease caused by HPV 31, 33, and 45.3
These findings mean that Cervarix is 91% effective against HPV types that cause adenocarcinoma and 83% effective overall against squamous cell carcinoma. Compare that with Gardasil, which is 78% effective overall against HPV types that cause adenocarcinoma and 73% effective against HPV types that cause squamous cell carcinoma.
The immune titers tell a supportive story. After vaccination with Gardasil, the antibody titer immediately declines for HPV 6, 11, and 18, reaching the baseline for natural infection within 18 months.7 HPV 18 shows continued, significant loss of seropositivity over time, and antibody titers for HPV 6 and 11 also decline. In the monovalent HPV 16 pre-Gardasil experimental vaccine, 14% of women no longer had measurable titers to HPV 16 after 8.5 years.11
After vaccination with Cervarix, antibody titers for HPV 16 and 18 remain more than seven times and more than four times higher, respectively, than natural infection titers for 8.4 years, with no loss of measurable antibody titer for either type. The antibody titers for HPV 31, 33, and 45 remain substantially higher than natural infection titers for at least 6.4 years. These titers correlate with the vaccine’s very high efficacy against CIN 2+ lesions caused by HPV 16, 18, 31, 33, and 45.
In other words, Cervarix generates an immune response (and efficacy) that indicates robust protection against five of the most common oncogenic HPV types, providing maximal protection against nearly 85% of all cervical cancers. Gardasil protects against 74% of all cervical cancers overall.12 This makes Cervarix the superior cervical cancer vaccine.
Gardasil is the superior vaccine against genital warts, although the duration of its protection is uncertain.
Dr. Huh: I’d just like to point out that there are no head-to-head trials comparing the vaccines in terms of efficacy. Antibody titers are higher with Cervarix than with Gardasil, as you noted, and it may be that, over time, the higher titers are more durable with Cervarix. However, we have yet to fully correlate clinical efficacy with antibody titers. In other words, immunogenicity does not equal clinical efficacy.
Dr. Felix: The data for Gardasil are particularly interesting because there have been no incident HPV-18 lesions detected despite the absence of detectable HPV-18 antibody titers in more than 20% of vaccinated women as soon as 2 years after immunization.9 These data strongly suggest that it is not antibody titer alone that grants protection against HPV-induced lesions of the cervix.
Dr. Harper: This speaks to the difficulty of running a trial to ensure both enough participants and a sufficient attack rate of HPV 18 to cause new lesions to be detected in vaccinated women. In the relevant trial, there were only 112 vaccinated women—not nearly enough women to overcome the very low attack rate of HPV 18 in the trial population—and they were followed for 5 years.9 We cannot be sure that the lack of incident HPV-18 lesions in the vaccinated women is the result of efficacy.
Dr. Felix: As for overlapping immunity to HPV types not included in the vaccines, it has been described for both Cervarix and Gardasil. In the case of Cervarix, the manufacturer demonstrated unexpectedly high rates of protection against all CIN 2+ and CIN 3+ lesions—70% and 87%, respectively. These rates were too high to be explained by protection against types 16, 18, 31, and 45 alone. It is possible, therefore, that Cervarix may protect against other high-risk HPV types.13
Gardasil has proved to be effective against HPV types 31, 33, 52, and others.10 When total protection against CIN 2+ and CIN 3+ lesions is examined from Phase-3 trials of the vaccine, however, the rates are only 42% and 43%, respectively. These data are difficult to interpret because HPV 16 and 18 together are thought to account for 70% of CIN 3. Some reassurance can be gained from the fact that the number of incident cases of CIN 2+ and CIN 3+ caused by HPV 16 and 18 in the vaccinated group in the Gardasil trial was identical to the number seen in the Cervarix trial.3,10 The reason for the discrepancy in total number of cases of CIN 2+ and CIN 3+ between the two trials—and, therefore, between the two vaccines—cannot be explained by cross-protection alone and is probably attributable to differences in study populations. The Gardasil trial had a higher baseline prevalence of HPV 16 and 18 (9% and 4%, respectively) than the Cervarix trial did (5% and 2%, respectively), a fact that may be explained by the different demographics of their respective populations.2,14
Ultimately, it is hazardous to compare trials, particularly when they are conducted in significantly different populations. On this issue, I concur with the World Health Organization (WHO), which recommended that such comparisons be avoided in the determination of which type of HPV vaccine to recommend.15
Dr. Huh: I agree that it would be inappropriate to make cross-trial comparisons, given differences in the way the trials were designed and conducted. To draw conclusions about clinical efficacy of these two excellent vaccines, based on a comparison of their trials, is completely unscientific. Only a true head-to-head study that has efficacy as its endpoint can tell us which vaccine is superior—and such a trial would require thousands (if not tens of thousands) of subjects and a considerable amount of time to complete. In my opinion, such a study would be counterproductive to our goal of vaccination.
Dr. Harper: I disagree. The whole purpose of this roundtable is to compare vaccines. It is not “unscientific” to compare the trials.
Dr. Huh: On the contrary—it is completely inappropriate to directly compare the Phase-3 clinical trials from Merck and GlaxoSmithKline. One can speculate about the differences between them, but any clinical trialist knows that a direct, scientific comparison cannot be made. Only a real head-to-head study powered for efficacy can do this.
- Both the bivalent and quadrivalent vaccines appear to be excellent products. Besides protecting against the main oncogenic strains of human papillomavirus (HPV) (types 16 and 18 for both vaccines, and the genital-wart-associated strains 6 and 11 for the quadrivalent vaccine), both Cervarix and Gardasil offer some degree of cross-protection against additional HPV strains.
- Vaccination of the sexually naïve patient with either vaccine provides significant protection against cervical intraepithelial neoplasia 2 (CIN 2) or worse.
- HPV vaccination is expected to reduce the rate of abnormal Pap tests and the need for common excisional treatments for cervical dysplasia in vaccinated women. It will do the same in the population as a whole if rates of vaccination are sufficient to provide “herd” immunity.
3. Is one vaccine more effective than the other?
Dr. Lonky: How do the vaccines compare in terms of efficacy?
Dr. Smith-McCune: In discussing efficacy, I think we should focus on CIN 3 because it is the immediate surrogate for cancer, whereas CIN 2 lesions can be transient in younger women. I think it is also important to focus on outcomes regardless of the HPV types associated with the lesions. This approach is more clinically relevant, as we don’t perform HPV typing of lesions in clinical practice. Nor do we manage lesions differently depending on the HPV type in the lesion.
That said, it is difficult to compare efficacy of the vaccines for several reasons, a few of which we have already discussed. For example, the bivalent and quadrivalent vaccines were studied in separate randomized trials. Although the study populations were similar, they were not identical. Women in both trials were relatively sexually naïve, but the cutoff for number of lifetime sexual partners was different (5 for Gardasil versus 7 for Cervarix). In trials of Gardasil, women who had a history of abnormal cytology or genital warts were excluded. In trials of Cervarix, women who had a history of colposcopy were excluded. In Gardasil trials, approximately 3% of women were from the Asian Pacific, versus 34% in the Cervarix trials, and so on.3,16
The trials also had different protocols for referral to colposcopy, which would affect disease detection. And the length of follow-up differs between trials.3,9
Dr. Lonky: Can we draw any conclusions about efficacy?
Dr. Smith-McCune: Yes. The trials defined outcomes in several populations of participants. In addition to the overall population (called the “intention-to-treat population” in the Gardasil trials and the “total vaccinated cohort” in the Cervarix trials), the trials defined a subpopulation of women naïve to oncogenic HPV types to gain information about the likely impact of vaccinating girls before the onset of sexual activity. The definitions of these “naïve” populations were slightly different, mainly in the number of HPV types tested, so again, some caution needs to be exercised in making comparisons.
End-of-trial data in the naïve population show a 43% reduction in CIN 3 lesions for Gardasil and 87% for Cervarix (for CIN 3 or worse). By inference, we can tell the sexually naïve patient that vaccination with either vaccine will provide significant protection against CIN 3 lesions, likely to result in significant protection against cervical cancer over time.
We can gather some estimates of efficacy in sexually non-naïve women by looking at results from all trial participants. Gardasil reduced overall CIN 3 lesions by 16% overall; Cervarix reduced CIN 3 or worse by 33%. When counseling an individual patient, if she has had a similarly low number of lifetime sexual partners (e.g., the median number in the Gardasil trials was 2), these results provide an estimate of her likely protection against CIN 3 with vaccination.
Common excisional treatments for cervical dysplasia are known to be associated with adverse perinatal outcomes.17 The ability to reduce the need for these treatments is an important outcome of vaccination. In the HPV-naïve populations, vaccination reduced definitive cervical therapies or excisions by 42% (Gardasil) and 69% (Cervarix). These figures are useful in counseling virginal patients about the long-term benefits of vaccination.
For sexually active patients 26 years and younger, HPV vaccination significantly reduced definitive cervical therapy or excisions by 23% (Gardasil) and 25% (Cervarix). Again, these figures are most applicable for counseling patients who have had relatively few lifetime sexual partners. So the exact extent of protection is likely to vary by the patient’s total number of lifetime sexual partners.
I expect that we will see more data on the effects of vaccination stratified by the number of lifetime sexual partners, because that information would be very useful in counseling individual sexually active women.
Dr. Harper: Both vaccines reduce the rate of abnormal Pap tests by 10% regardless of HPV type in that population of women.9,18
4. Are the two vaccines safe?
Dr. Lonky: What about safety of the vaccines? What do we know?
Dr. Felix: The safety profiles seen in clinical trials of both vaccines are very similar and consist almost entirely of nonserious adverse events.2,9 In the United States, a greater number of Gardasil doses has been administered, owing to its earlier development. As of January 1, 2010, more than 28 million doses had been distributed, and numerous major events had been recorded in the Vaccine Adverse Event Reporting System (VAERS). Of 15,829 adverse events reported, only 8% were considered serious by the CDC. CDC investigation, by expert panels, of all serious adverse events found no evidence linking Gardasil to any of them, including Guillain-Barré syndrome, blood clots, and death.19
Dr. Huh: A few other points to consider:
- The reporting rate for Gardasil is triple that for all other vaccines combined
- Because VAERS is a passive reporting system, under-reporting is distinctly possible
- Post-licensure safety surveillance is still underway
- Both products have pregnancy registries.
Dr. Harper: The current postmarketing commitment between Merck and the FDA is to recognize a rate of serious adverse events that exceeds 2 cases in every 10,000 women in a cohort of 44,000 women who have received all three doses of Gardasil. Although autoimmune neurologic sequelae have occurred after Gardasil administration, regulatory authorities are not required to evaluate these reactions, such as Guillain-Barré syndrome, because the frequency is lower than the agreed-upon threshold. Nevertheless, adverse events could be life threatening to some girls.
Any risk of death—even if it is lower than the agreed-upon threshold—should be presented to women as a possible risk of vaccination with Gardasil. In the United States, the same women could choose a lifetime of Pap screening and be afforded the same protection against cervical cancer as they would get from vaccination.
5. Is quadrivalent better than bivalent?
Dr. Lonky: Why would a clinician choose a bivalent vaccine when the quadrivalent vaccine protects not only against carcinogenic types 16 and 18, but also against HPV-associated genital warts?
Dr. Harper: A smart clinician would ask the patient what she values. The physician is obligated to present the evidence and let her choose!
Dr. Lonky: What does the evidence suggest?
Dr. Felix: A clear recommendation between Cervarix and Gardasil is very difficult to make at this time, for the reasons already stated. Both vaccines provide 98% protection against HPV 16 and 18 for the prevention of CIN 2+ lesions.3,9 As we have discussed, both vaccines also provide protection against high-risk strains of HPV other than types 16 and 18.
In the Cervarix trial, there was an overall reduction of all CIN 2+ lesions that was higher as a percentage of total lesions than the reduction seen in the Gardasil trial.3,9 However, unlike Cervarix, Gardasil significantly reduced the rate of vaginal intraepithelial neoplasia (VaIN) and vulvar intraepithelial neoplasia (VIN).9
Dr. Harper: GlaxoSmithKline is analyzing its data on vulvar and vaginal protection, and it is likely that Cervarix will demonstrate some efficacy in this regard, too. But the economic burden of noncervical cancers is estimated to be only 8% of the economic burden of all HPV-related diseases.20 The prevention of cervical cancer is the dominant clinical and economic force for vaccination.
Dr. Felix: Clearly, the protection against genital warts demonstrated in the Gardasil trial will not be realized with Cervarix, as it does not offer immunization or cross-protection against HPV 6 or 11. Gardasil’s protection against HPV 6 and 11 prompted FDA approval of the vaccine for boys and men.21
According to the WHO, when counseling girls and women about the HPV vaccine, the clinician should weigh the possible value of a deep reduction in total CIN 2+ lesions provided by Cervarix against the reduction in VaIN, VIN, and genital warts provided by Gardasil.15 Boys and men will see clinically proven benefits only from Gardasil for the prevention of external genital warts. Other benefits are strictly theoretical.22,23
Dr. Harper: Vaccine protection must last at least 15 years to reduce the rate of cervical cancer. Otherwise, the development of cervical cancer will only be postponed, if boosters are not implemented.
It is now widely recognized that Cervarix induces high antibody titers, offering 100% efficacy even after 8.4 years, making it very likely that the protection it provides will continue for at least 15 years. It is also widely acknowledged by immunologists that Gardasil-induced titers for HPV 6, 11, and 18 are much shorter-lived, so protection is likely to wane 5 to 10 years after vaccination.
That means that Gardasil provides excellent protection against one cancer-causing type of HPV. In addition, it protects against genital warts caused by HPV types 6 and 11 for at least 5 years. In comparison, Cervarix protects against five cancer-causing types of HPV, thereby preventing about 90% of cervical cancers, and is likely to remain effective for at least 15 years.
There are 10 times as many women who have an abnormal Pap test as there are women who have genital warts, so one would think that Cervarix would be the vaccine of choice in preventing the life-threatening disease of cervical cancer.
Dr. Smith-McCune: I would agree that the choice should be discussed with patients—and with parents. If the objective is primarily to protect against cervical cancer precursors, then the bivalent vaccine may be the better choice, with the caveat that we can’t really compare the results from the vaccine trials for reasons discussed earlier, and there are no data from a randomized head-to-head trial comparing the two vaccines. If the decision involves a desire to reduce genital warts or vulvar and vaginal dysplasia, then the quadrivalent vaccine would be the better choice.
6. What impact do the vaccines have on screening?
Dr. Lonky: Do the vaccines have varying effects on our need to screen for, triage, and treat cervical cancer precursors?
Dr. Harper: The Pap smear has reduced the rate of cervical cancer in the United States by 75%—that rate is now at an all-time low of 8 cases for every 100,000 women. But the Pap smear is not perfect; there is a 30% false-negative rate among women who develop cervical cancer, and a large false-positive rate that involves referral to colposcopy for minimally abnormal cytology reports. And when CIN 2+ disease is detected, treatment is not without risk. Surgery increases the risk of reproductive morbidity in future pregnancies. Having protection against this outcome could be tremendously valuable for some women.
Compare the HPV vaccine, which has probable benefit but also the potential for serious adverse events, including demyelinating diseases that cause blindness, paralysis, and death in a small number of recipients.
If women were to choose to be vaccinated with Gardasil and forgo further Pap screening, the rate of cervical cancer in the United States would rise from 8 to 14 cases for every 100,000 women. If they were to choose Cervarix instead, with no further Pap screening, the rate would rise from 8 to 9.5 cases for every 100,000 women.
If women were to choose both HPV vaccination and continued Pap screening, the rate of cervical cancer still would not decline from its current level of 8 cases for every 100,000 women. Instead, the benefit would be that fewer women have abnormal Pap tests, and fewer women would need to be treated for CIN 2+ disease.
Women and physicians must understand these facts. A woman who chooses to be vaccinated may gain individual protection, but the overall rate of cervical cancer will not be affected.
Dr. Huh: Regardless of the HPV vaccine selected, we need to seriously rethink how we screen women in the United States. One could easily argue that the combination of the vaccine and continued screening is too expensive. It might be wise to consider lengthening the screening interval—and, perhaps, further delaying initial screening to 25 years of age—to make cervical cancer prevention with both modalities more cost-effective.24 The most important thing to recognize is that women still need to be screened, even if they have been vaccinated.
As more women are vaccinated, we expect to see a decline in the prevalence of CIN 2+ and CIN 3+ lesions, and this will ultimately weaken the positive predictive value of cytology. Perhaps it is time to consider the HPV test as a primary screen, with triage to cytology in women who test HPV-positive.25
Dr. Smith-McCune: As we accumulate data over time about the effects of vaccination on the rates of CIN 3 and cancer, modeling will be helpful in determining the best screening algorithm for women who have been vaccinated against HPV.
It is important to remember that approximately 50% of women who are given a diagnosis of cervical cancer in the United States have never been screened. It is vital that we continue to reach out to the under-screened population and focus vaccination efforts on populations of girls who are likely to have limited access to care in the future.
7. Can we vaccinate every woman?
Dr. Lonky: Is universal vaccination of women achievable for either vaccine?
Dr. Felix: Universal vaccination against HPV would be achievable only via school mandates. Without them, vaccination will not approach the 80% threshold needed to produce herd immunity.
Despite the clear benefit of such mandates to the general population—particularly the medically under-served—the issue has become a political football. As a result, school mandates will probably never be realized.
Dr. Harper: I don’t believe it is ethical to mandate vaccination of all girls and women. It is a choice that women and parents, in conversation with their physicians and daughters, must make when considering how to be protected against cervical cancer. Herd immunity is a moot point because we are only vaccinating girls (50% of the population) and can never reach the theoretical 70% threshold for herd immunity to be apparent.
Dr. Lonky: Is the availability of two vaccines a boon or a hindrance?
Dr. Smith-McCune: I think it is always a good thing to have choices in medicine.
Dr. Huh: I see the availability of two vaccines as a boon. That availability means that two companies are now putting forth consistent educational messages about the importance of vaccination and, I hope, stimulating competition that will reduce the overall cost of the vaccine series. Having two vaccines can only promote awareness, access, and greater appreciation of the considerable protection these two vaccines provide.
Despite solid evidence that the quadrivalent (Gardasil) HPV vaccine and the bivalent (Cervarix) HPV vaccine protect against cervical cancer, only about one fifth of the female population between 11 and 26 years of age has received the full series of Gardasil since it won FDA approval in 2006. Barriers to vaccination are not financial alone, as the vaccination rate is similarly low among women who have health insurance.
Why isn’t the vaccination rate higher? I see eight barriers to full implementation:
- Economic disparities. Each vaccine costs roughly $400 (national average) for the full series of injections. Although women who do not get Pap screening are most likely to benefit from the vaccines, they usually cannot afford them. federal childhood immunization programs cover teens and young women until 18 years of age in most states, and until 21 years in a few. that leaves most women who seek vaccination from gynecologists without coverage.
- Fear. Pain at the injection site, syncope, and a slightly elevated incidence of thromboembolism are the adverse events most commonly associated with HPV vaccination in the literature. In the life cycle of a vaccine, reports of sudden death or neurologic injury (Guillain-Barré syndrome) occur in the early years, but are reported at a rate lower than 2 cases in every 10,000 women. Nevertheless, such events may create fear about undergoing immunization.
- Long latency period. Because the outcome of cancer prevention won’t become apparent for 20 to 40 years following vaccination, the need for immunization may seem less than urgent.
- Cultural and religious beliefs. Because carcinogenic HPV strains are sexually transmitted, some families may associate vaccination with the promotion of sexual activity. Even in states that mandate vaccination, the courts have upheld a parent’s right to refuse vaccination on these grounds.
- The premarket push. Aggressive promotion of vaccination by both manufacturers and a push by advocates for legislation to mandate the vaccine prior to completion of Phase-3 trials and gathering of robust safety data may have diminished trust in the vaccine and reduced its acceptance.
- Lack of legislation. In states that do not consider HPV vaccination to be a necessary public health intervention, the lack of mandates and funding reduce the vaccination rate. In addition, some legislators have been more active advocates of vaccination than others.
- Reduced involvement of the obGyn. ObGyns don’t routinely vaccinate patients; pediatricians do. Young women are slipping through the cracks because the conventional ObGyn practice does not have a vaccination program that ensures payment, reimbursement, and completion of the vaccine series. Many ObGyn practices are reluctant to institute such a program because the profit margin is small, there are associated risks, and the time required to counsel the patient and for follow-up is extensive.
- Failure to complete the series. Some women do not complete the full vaccine series, owing to cost or side effects, or both. Solid evidence that a single dose could be as protective as the full series would be compelling. A single-dose vaccine would also be less expensive.
The principal danger of a low vaccination rate is the loss of insurance coverage for immunization against HPV. On one hand, payers may begin to ask whether coverage is justified when so few girls and women are vaccinated, leaving the payer with two burdens: the expense of vaccination and the expense of conventional screening programs and treatment, although the costs of treatment would be reduced with vaccination. On the other hand, Gardasil’s protection against genital warts may provide incentive for payers to cover or discount the vaccine because of the reduction in the need to diagnose, triage, and treat condyloma.
Ultimately, HPV vaccination may become another optional intervention that is paid for by the individual, despite evidence in girls and women that cervical cancer can be prevented.
—NEAL M. LONKY, MD, MPH
We want to hear from you! Tell us what you think.
1. The FUTURE II Study Group. Quadrivalent vaccine against human papillomavirus to prevent high-grade cervical lesions. N Engl J Med. 2007;356(19):1915-1927.
2. Paavonen J, Jenkins D, Bosch FX, Naud P, Salmerón J, Wheeler CM, et al. HPV PATRICIA study group. Efficacy of a prophylactic adjuvanted bivalent L1 virus-like-particle vaccine against infection with human papillomavirus types 16 and 18 in young women: an interim analysis of a phase III double-blind, randomised controlled trial. Lancet. 2007;369(9580):2161-2170.
3. Paavonen J, Naud P, Salmeron J, et al. For the HPV PATRICIA Study Group. Efficacy of human papillomavirus (HPV)-16/18 AS04-adjuvanted vaccine against cervical infection and precancer caused by oncogenic HPV types (PATRICIA): final analysis of a double-blind, randomised study in young women. Lancet. 2009;374(9686):301-314.
4. DiMario FJ, Hajjar M, Ciesielski T. A 16-year-old girl with bilateral visual loss and left hemiparesis following an immunization against human papillomavirus. J Child Neurology. 2010;25(3):321-327.
5. Sutton I, Lahoria R, Tan I, Clouston P, Barnett M. CNS demyelination and quadrivalent HPV vaccination. Multiple Sclerosis. 2009;15(1):116-119.
6. Klein SL, Jedlicka A, Pekosz A. The Xs and Y of immune responses to viral vaccines. Lancet Infect Dis. 2010;10(5):338-349.
7. Einstein MH, Baron M, Levin MJ, et al. Comparison of the immunogenicity and safety of Cervarix and Gardasil human papillomavirus (HPV) cervical cancer vaccines in healthy women aged 18-45 years. Hum Vaccin. 2009;5(10):705-719.
8. GlaxoSmithKline Vaccine HPV-007 Study Group. Sustained efficacy and immunogenicity of the HPV-16/18 AS04-adjuvanted vaccine: analysis of a randomised placebo-controlled trial up to 8.4 years. Presented at: 28th Annual Meeting of the European Society for Paediatric Infectious Diseases (ESPID); May 4-8, 2010; Nice, France.
9. Muñoz N, Kjaer SK, Sigurdsson K, et al. Impact of human papillomavirus (HPV)-6/11/16/18 vaccine on all HPV-associated genital diseases in young women. J Natl Cancer Inst. 2010;102(5):325-339.
10. Brown DR, Kjaer SK, Sigurdsson K, et al. The impact of quadrivalent human papillomavirus (HPV; types 6, 11, 16, and 18) L1 virus-like particle vaccine on infection and disease due to oncogenic nonvaccine HPV types in generally HPV-naïve women aged 16-26 years. J Infect Dis. 2009;199(7):926-935.
11. Rowhani-Rahbar A, Mao C, Hughes JP, et al. Longer term efficacy of a prophylactic monovalent human papillomavirus type 16 vaccine. Vaccine. 2009;27(41):5612-5619.
12. Harper DM, Williams KB. Prophylactic HPV vaccines: current knowledge of impact on gynecologic premalignancies [published online ahead of print July 3, 2010]. Discovery Medicine. 2010;10(50).http://www.discoverymedicine.com/Diane-M-Harper/2010/07/03/prophylactichpv-vaccines-current-knowledge-of-impact-ongynecologic-premalignancies. Accessed July 13, 2010.
13. Cervarix [human papillomavirus bivalent (types 16 and 18) vaccine recombinant]. Food and Drug Administration. Initial US approval: 2009:1–12.
14. Garland SM, Hernandez-Avila M, Wheeler CM, et al. Quadrivalent vaccine against human papillomavirus to prevent anogenital diseases. N Engl J Med. 2007;356(19):1928-1943.
15. World Health Organization. Human papillomavirus vaccines: WHO position paper. Weekly Epidemiological Record. 2009;84(15):118-131.http://www.who.int/wer/2009/wer8415/en/index.html. Published April 10, 2009. Accessed July 13, 2010.
16. Ault KA. For Future II Study Group. Effect of prophylactic human papillomavirus L1 virus-like-particle vaccine on the risk of cervical intraepithelial neoplasia grade 2, grade 3, and adenocarcinoma in situ: a combined analysis of four randomised clinical trials. Lancet. 2007;369(9576):1861-1868.
17. Arbyn M, Kyrgiou M, Simoens C, et al. Perinatal mortality and other severe adverse pregnancy outcomes associated with treatment of cervical intraepithelial neoplasia: meta-analysis. BMJ. 2008;337:a1284.-Doi: 10.1136/bmj.a1284.
18. Paavonen J. For the HPV PATRICIA Study Group. Efficacy of HPV 16/18 ASO4-adjuvanted vaccine against abnormal cytology, colposcopy referrals, and cervical procedures. Abstract SS 4–1. Data presented at Cervical Cancer Prevention, EuroGin 2010. European Research Organisation on Genital Infection and Neoplasia (EUROGIN); February 17-20, 2010; Monte Carlo, Monaco.http://www.eurogin.com/2010/programoverview.html. Accessed July 13, 2010.
19. Centers for Disease Control and Prevention. Reports of health concerns following HPV vaccination. 2010;1-3.http://www.cdc.gov/vaccinesafety/Vaccines/HPV/gardasil.html. Published June 21, 2010. Accessed July 13, 2010.
20. Myers ER. The economic impact of HPV vaccines: not just cervical cancer. Am J Obstet Gynecol. 2008;198(5):487-488.
21. Gardasil [human papillomavirus quadrivalent (types 6, 11, 16, and 18) vaccine, recombinant]. Food and Drug Administration; 2009. Initial Approval 2006;1-26.
22. Olsson SE, Kjaer SK, Sigurdsson K, et al. Evaluation of quadrivalent HPV 6/11/16/18 vaccine efficacy against cervical and anogenital disease in subjects with serological evidence of prior vaccine type HPV infection. Hum Vaccin. 2009;5(10).
23. Pirotta M, Ung L, Stein A, et al. The psychosocial burden of human papillomavirus related disease and screening interventions. Sex Transm Infect. 2009;85(7):508-513.
24. Kim JJ, Goldie SJ. Health and economic implications of HPV vaccination in the United States. N Engl J Med. 2008;359(8):821-832.
25. Cox JT. Is the HPV test effective as the primary screen for cervical cancer? Examining the Evidence. OBG Management. 2010;22(7):10-11.
Not long ago (in medical years), we were still trying to discover the cause of cervical cancer. Today, not only do we know that cause to be persistent human papillomavirus (HPV) infection, but we have two vaccines at our disposal to prevent the primary oncogenic strains of the virus.
We’ve come a long way.
The availability of two vaccines raises questions, however. What kind of data do we have on the bivalent (Cervarix, GlaxoSmithKline) and quadrivalent (Gardasil, Merck) vaccines so far? Is one of them clearly superior to the other? If not, what population is each vaccine best suited for—and how do we counsel patients about their options?
To address these and other questions, OBG Management Contributing Editor Neal M. Lonky, MD, MPH, assembled a panel of physicians who have expertise in cervical disease detection and prevention and asked them to sift the data that have accumulated thus far. In the discussion that follows, they touch on long-term efficacy, the likely impact of the vaccines on cervical cancer screening, and other aspects of disease prevention in the era of HPV vaccination.

Juan C. Felix, MD
Professor of Clinical Pathology and Obstetrics and Gynecology; Director of Cytopathology fellowship; and Chief of Gynecologic Pathology at the Keck School of Medicine, University of Southern California; and Chief of Cytopathology at Los Angeles County and University of Southern California Medical Center in Los Angeles.
Dr. Felix reports that he is a speaker for Merck and GlaxoSmithKline.

Diane M. Harper, MD, MS, MPH
Director of the Gynecologic Cancer Prevention Research Group and Professor of Obstetrics and Gynecology, Community and family Medicine, and Informatics and Personalized Medicine at the University of Missouri–Kansas City School of Medicine.
Dr. Harper reports that she has served as a speaker and advisor for Merck and GlaxoSmithKline, and that the institutions at which she conducted HPV vaccination trials have received funding from Merck and GlaxoSmithKline.

Warner K. Huh, MD
Associate Professor in the Department of Obstetrics and Gynecology, and Associate Scientist at the Comprehensive Cancer Center at the University of Alabama– Birmingham.
Dr. Huh reports that he receives grant or research support from and is a speaker and consultant to Merck and GlaxoSmithKline.

Karen K. Smith-McCune, MD, PhD
John Kerner Endowed Chair of Gynecologic Oncology, Director of the Dysplasia Clinic, and Professor of Obstetrics, Gynecology, and Reproductive Sciences at the University of California–San francisco.
Dr. Smith-McCune reports she has performed unpaid consulting for OncoHealth Inc. and is planning to join its Scientific Advisory Board.
1. How were the vaccines developed?
Neal M. Lonky, MD, MPH: What should clinicians know about the development, function, and mechanism of action of the two HPV vaccines?
Warner K. Huh, MD: The bivalent and quadrivalent vaccines are both excellent products, and their respective Phase-3 trials demonstrate that they provide impressive protection against HPV, particularly among women who test negative (by polymerase chain reaction) for the specific HPV types contained within the vaccines.1-3
Cervarix protects against HPV types 16 and 18, whereas Gardasil is effective against HPV types 6, 11, 16, and 18.
Dr. Lonky: Do the vaccines function similarly?
Diane M. Harper, MD, MS, MPH: Yes. Both stimulate an immediate antibody response in the woman who is not infected with the relevant virus and are effective in preventing cervical intraepithelial neoplasia grade 2 and higher (CIN 2+), as well as persistent infection, caused by vaccine-related and cross-protected HPV types. The quality of the antibody response is best for HPV 16 for both vaccines. The quality of the antibody response for HPV 6, 11, and 18 for Gardasil is much poorer than its response for HPV 16. Cervarix induces an equally high and sustained antibody response for HPV 18 as for HPV 16.
Juan C. Felix, MD: Both vaccines are based on the same virus-like particles (VLP). The functionality of the vaccines is, therefore, mainly dependent on the dosage of VLP and the adjuvant used. Gardasil uses a proprietary aluminum sulfate adjuvant, whereas Cervarix uses aluminum hydroxide and monophosphoryl lipid A.
Karen K. Smith-McCune, MD, PhD: Both adjuvants have an extensive track record of safety and efficacy in other vaccines. Because they have different structures, however, they may have varying effects on many components of the immune response elicited by the L1 antigens.
Dr. Harper: Both adjuvants contain aluminum, which has so far proved to be safe despite the newly established association between high aluminum intake and Alzheimer’s disease.
Dr. Lonky: Were there any notable challenges in developing the vaccines?
Dr. Harper: It was difficult to formulate the appropriate dosages of VLP in Gardasil. Higher dosages of HPV 11 and 16 were needed to prevent cross-inhibition by HPV 6 and 18. As a result, the antigenic protein component of Gardasil that is necessary to effect an immunologic antibody response is high, at 120 μg. In Cervarix, the antigenic VLP load is 20 μg each for HPV 16 and 18.
Dr. Lonky: What is the significance of the different VLP loads?
Dr. Harper: Side effects, such as autoimmune neurologic demyelination, albeit rare, have been associated with a higher antigenic protein load. Multiple reports of autoimmune demyelinating diseases—including paralysis, blindness, and death—have been published by neurologists in regard to Gardasil.4,5 Others have shown that young girls are more at risk than young boys for these neurologic side effects.6
Dr. Felix: Some data suggest that the two vaccine formulations interact differently with the human immune system. In a head-to-head trial funded by GlaxoSmithKline, Cervarix produced higher total and neutralizing antibody titers than Gardasil did.7
Although higher immunogenicity is generally thought to be beneficial, the ultimate determinant of a vaccine’s success is its efficacy—and duration of that efficacy—in clinical trials and follow-up of vaccinated populations. So far, Cervarix has demonstrated efficacy through 8.4 years in its follow-up cohort.8 Similarly, Gardasil has proved to be effective after 5 years of follow-up, with no incident cases of cervical cancer reported in the vaccinated arm.9

2. Does either vaccine offer “extra” immunity?
Dr. Lonky: What is the potential for overlapping immunity to other high-risk viral types with these vaccines?
Dr. Harper: It is quite clear from pivotal trials of both vaccines that Gardasil produces efficacy of 46% against persistent infection caused by HPV 31. Data from the pivotal Phase-3 trial of Gardasil also show that it offers no protection against persistent infection with HPV 45, an important cause of adenocarcinoma.10
In contrast, Cervarix demonstrates substantial efficacy against both persistent infection and CIN 2+ disease caused by HPV 31, 33, and 45.3
These findings mean that Cervarix is 91% effective against HPV types that cause adenocarcinoma and 83% effective overall against squamous cell carcinoma. Compare that with Gardasil, which is 78% effective overall against HPV types that cause adenocarcinoma and 73% effective against HPV types that cause squamous cell carcinoma.
The immune titers tell a supportive story. After vaccination with Gardasil, the antibody titer immediately declines for HPV 6, 11, and 18, reaching the baseline for natural infection within 18 months.7 HPV 18 shows continued, significant loss of seropositivity over time, and antibody titers for HPV 6 and 11 also decline. In the monovalent HPV 16 pre-Gardasil experimental vaccine, 14% of women no longer had measurable titers to HPV 16 after 8.5 years.11
After vaccination with Cervarix, antibody titers for HPV 16 and 18 remain more than seven times and more than four times higher, respectively, than natural infection titers for 8.4 years, with no loss of measurable antibody titer for either type. The antibody titers for HPV 31, 33, and 45 remain substantially higher than natural infection titers for at least 6.4 years. These titers correlate with the vaccine’s very high efficacy against CIN 2+ lesions caused by HPV 16, 18, 31, 33, and 45.
In other words, Cervarix generates an immune response (and efficacy) that indicates robust protection against five of the most common oncogenic HPV types, providing maximal protection against nearly 85% of all cervical cancers. Gardasil protects against 74% of all cervical cancers overall.12 This makes Cervarix the superior cervical cancer vaccine.
Gardasil is the superior vaccine against genital warts, although the duration of its protection is uncertain.
Dr. Huh: I’d just like to point out that there are no head-to-head trials comparing the vaccines in terms of efficacy. Antibody titers are higher with Cervarix than with Gardasil, as you noted, and it may be that, over time, the higher titers are more durable with Cervarix. However, we have yet to fully correlate clinical efficacy with antibody titers. In other words, immunogenicity does not equal clinical efficacy.
Dr. Felix: The data for Gardasil are particularly interesting because there have been no incident HPV-18 lesions detected despite the absence of detectable HPV-18 antibody titers in more than 20% of vaccinated women as soon as 2 years after immunization.9 These data strongly suggest that it is not antibody titer alone that grants protection against HPV-induced lesions of the cervix.
Dr. Harper: This speaks to the difficulty of running a trial to ensure both enough participants and a sufficient attack rate of HPV 18 to cause new lesions to be detected in vaccinated women. In the relevant trial, there were only 112 vaccinated women—not nearly enough women to overcome the very low attack rate of HPV 18 in the trial population—and they were followed for 5 years.9 We cannot be sure that the lack of incident HPV-18 lesions in the vaccinated women is the result of efficacy.
Dr. Felix: As for overlapping immunity to HPV types not included in the vaccines, it has been described for both Cervarix and Gardasil. In the case of Cervarix, the manufacturer demonstrated unexpectedly high rates of protection against all CIN 2+ and CIN 3+ lesions—70% and 87%, respectively. These rates were too high to be explained by protection against types 16, 18, 31, and 45 alone. It is possible, therefore, that Cervarix may protect against other high-risk HPV types.13
Gardasil has proved to be effective against HPV types 31, 33, 52, and others.10 When total protection against CIN 2+ and CIN 3+ lesions is examined from Phase-3 trials of the vaccine, however, the rates are only 42% and 43%, respectively. These data are difficult to interpret because HPV 16 and 18 together are thought to account for 70% of CIN 3. Some reassurance can be gained from the fact that the number of incident cases of CIN 2+ and CIN 3+ caused by HPV 16 and 18 in the vaccinated group in the Gardasil trial was identical to the number seen in the Cervarix trial.3,10 The reason for the discrepancy in total number of cases of CIN 2+ and CIN 3+ between the two trials—and, therefore, between the two vaccines—cannot be explained by cross-protection alone and is probably attributable to differences in study populations. The Gardasil trial had a higher baseline prevalence of HPV 16 and 18 (9% and 4%, respectively) than the Cervarix trial did (5% and 2%, respectively), a fact that may be explained by the different demographics of their respective populations.2,14
Ultimately, it is hazardous to compare trials, particularly when they are conducted in significantly different populations. On this issue, I concur with the World Health Organization (WHO), which recommended that such comparisons be avoided in the determination of which type of HPV vaccine to recommend.15
Dr. Huh: I agree that it would be inappropriate to make cross-trial comparisons, given differences in the way the trials were designed and conducted. To draw conclusions about clinical efficacy of these two excellent vaccines, based on a comparison of their trials, is completely unscientific. Only a true head-to-head study that has efficacy as its endpoint can tell us which vaccine is superior—and such a trial would require thousands (if not tens of thousands) of subjects and a considerable amount of time to complete. In my opinion, such a study would be counterproductive to our goal of vaccination.
Dr. Harper: I disagree. The whole purpose of this roundtable is to compare vaccines. It is not “unscientific” to compare the trials.
Dr. Huh: On the contrary—it is completely inappropriate to directly compare the Phase-3 clinical trials from Merck and GlaxoSmithKline. One can speculate about the differences between them, but any clinical trialist knows that a direct, scientific comparison cannot be made. Only a real head-to-head study powered for efficacy can do this.
- Both the bivalent and quadrivalent vaccines appear to be excellent products. Besides protecting against the main oncogenic strains of human papillomavirus (HPV) (types 16 and 18 for both vaccines, and the genital-wart-associated strains 6 and 11 for the quadrivalent vaccine), both Cervarix and Gardasil offer some degree of cross-protection against additional HPV strains.
- Vaccination of the sexually naïve patient with either vaccine provides significant protection against cervical intraepithelial neoplasia 2 (CIN 2) or worse.
- HPV vaccination is expected to reduce the rate of abnormal Pap tests and the need for common excisional treatments for cervical dysplasia in vaccinated women. It will do the same in the population as a whole if rates of vaccination are sufficient to provide “herd” immunity.
3. Is one vaccine more effective than the other?
Dr. Lonky: How do the vaccines compare in terms of efficacy?
Dr. Smith-McCune: In discussing efficacy, I think we should focus on CIN 3 because it is the immediate surrogate for cancer, whereas CIN 2 lesions can be transient in younger women. I think it is also important to focus on outcomes regardless of the HPV types associated with the lesions. This approach is more clinically relevant, as we don’t perform HPV typing of lesions in clinical practice. Nor do we manage lesions differently depending on the HPV type in the lesion.
That said, it is difficult to compare efficacy of the vaccines for several reasons, a few of which we have already discussed. For example, the bivalent and quadrivalent vaccines were studied in separate randomized trials. Although the study populations were similar, they were not identical. Women in both trials were relatively sexually naïve, but the cutoff for number of lifetime sexual partners was different (5 for Gardasil versus 7 for Cervarix). In trials of Gardasil, women who had a history of abnormal cytology or genital warts were excluded. In trials of Cervarix, women who had a history of colposcopy were excluded. In Gardasil trials, approximately 3% of women were from the Asian Pacific, versus 34% in the Cervarix trials, and so on.3,16
The trials also had different protocols for referral to colposcopy, which would affect disease detection. And the length of follow-up differs between trials.3,9
Dr. Lonky: Can we draw any conclusions about efficacy?
Dr. Smith-McCune: Yes. The trials defined outcomes in several populations of participants. In addition to the overall population (called the “intention-to-treat population” in the Gardasil trials and the “total vaccinated cohort” in the Cervarix trials), the trials defined a subpopulation of women naïve to oncogenic HPV types to gain information about the likely impact of vaccinating girls before the onset of sexual activity. The definitions of these “naïve” populations were slightly different, mainly in the number of HPV types tested, so again, some caution needs to be exercised in making comparisons.
End-of-trial data in the naïve population show a 43% reduction in CIN 3 lesions for Gardasil and 87% for Cervarix (for CIN 3 or worse). By inference, we can tell the sexually naïve patient that vaccination with either vaccine will provide significant protection against CIN 3 lesions, likely to result in significant protection against cervical cancer over time.
We can gather some estimates of efficacy in sexually non-naïve women by looking at results from all trial participants. Gardasil reduced overall CIN 3 lesions by 16% overall; Cervarix reduced CIN 3 or worse by 33%. When counseling an individual patient, if she has had a similarly low number of lifetime sexual partners (e.g., the median number in the Gardasil trials was 2), these results provide an estimate of her likely protection against CIN 3 with vaccination.
Common excisional treatments for cervical dysplasia are known to be associated with adverse perinatal outcomes.17 The ability to reduce the need for these treatments is an important outcome of vaccination. In the HPV-naïve populations, vaccination reduced definitive cervical therapies or excisions by 42% (Gardasil) and 69% (Cervarix). These figures are useful in counseling virginal patients about the long-term benefits of vaccination.
For sexually active patients 26 years and younger, HPV vaccination significantly reduced definitive cervical therapy or excisions by 23% (Gardasil) and 25% (Cervarix). Again, these figures are most applicable for counseling patients who have had relatively few lifetime sexual partners. So the exact extent of protection is likely to vary by the patient’s total number of lifetime sexual partners.
I expect that we will see more data on the effects of vaccination stratified by the number of lifetime sexual partners, because that information would be very useful in counseling individual sexually active women.
Dr. Harper: Both vaccines reduce the rate of abnormal Pap tests by 10% regardless of HPV type in that population of women.9,18
4. Are the two vaccines safe?
Dr. Lonky: What about safety of the vaccines? What do we know?
Dr. Felix: The safety profiles seen in clinical trials of both vaccines are very similar and consist almost entirely of nonserious adverse events.2,9 In the United States, a greater number of Gardasil doses has been administered, owing to its earlier development. As of January 1, 2010, more than 28 million doses had been distributed, and numerous major events had been recorded in the Vaccine Adverse Event Reporting System (VAERS). Of 15,829 adverse events reported, only 8% were considered serious by the CDC. CDC investigation, by expert panels, of all serious adverse events found no evidence linking Gardasil to any of them, including Guillain-Barré syndrome, blood clots, and death.19
Dr. Huh: A few other points to consider:
- The reporting rate for Gardasil is triple that for all other vaccines combined
- Because VAERS is a passive reporting system, under-reporting is distinctly possible
- Post-licensure safety surveillance is still underway
- Both products have pregnancy registries.
Dr. Harper: The current postmarketing commitment between Merck and the FDA is to recognize a rate of serious adverse events that exceeds 2 cases in every 10,000 women in a cohort of 44,000 women who have received all three doses of Gardasil. Although autoimmune neurologic sequelae have occurred after Gardasil administration, regulatory authorities are not required to evaluate these reactions, such as Guillain-Barré syndrome, because the frequency is lower than the agreed-upon threshold. Nevertheless, adverse events could be life threatening to some girls.
Any risk of death—even if it is lower than the agreed-upon threshold—should be presented to women as a possible risk of vaccination with Gardasil. In the United States, the same women could choose a lifetime of Pap screening and be afforded the same protection against cervical cancer as they would get from vaccination.
5. Is quadrivalent better than bivalent?
Dr. Lonky: Why would a clinician choose a bivalent vaccine when the quadrivalent vaccine protects not only against carcinogenic types 16 and 18, but also against HPV-associated genital warts?
Dr. Harper: A smart clinician would ask the patient what she values. The physician is obligated to present the evidence and let her choose!
Dr. Lonky: What does the evidence suggest?
Dr. Felix: A clear recommendation between Cervarix and Gardasil is very difficult to make at this time, for the reasons already stated. Both vaccines provide 98% protection against HPV 16 and 18 for the prevention of CIN 2+ lesions.3,9 As we have discussed, both vaccines also provide protection against high-risk strains of HPV other than types 16 and 18.
In the Cervarix trial, there was an overall reduction of all CIN 2+ lesions that was higher as a percentage of total lesions than the reduction seen in the Gardasil trial.3,9 However, unlike Cervarix, Gardasil significantly reduced the rate of vaginal intraepithelial neoplasia (VaIN) and vulvar intraepithelial neoplasia (VIN).9
Dr. Harper: GlaxoSmithKline is analyzing its data on vulvar and vaginal protection, and it is likely that Cervarix will demonstrate some efficacy in this regard, too. But the economic burden of noncervical cancers is estimated to be only 8% of the economic burden of all HPV-related diseases.20 The prevention of cervical cancer is the dominant clinical and economic force for vaccination.
Dr. Felix: Clearly, the protection against genital warts demonstrated in the Gardasil trial will not be realized with Cervarix, as it does not offer immunization or cross-protection against HPV 6 or 11. Gardasil’s protection against HPV 6 and 11 prompted FDA approval of the vaccine for boys and men.21
According to the WHO, when counseling girls and women about the HPV vaccine, the clinician should weigh the possible value of a deep reduction in total CIN 2+ lesions provided by Cervarix against the reduction in VaIN, VIN, and genital warts provided by Gardasil.15 Boys and men will see clinically proven benefits only from Gardasil for the prevention of external genital warts. Other benefits are strictly theoretical.22,23
Dr. Harper: Vaccine protection must last at least 15 years to reduce the rate of cervical cancer. Otherwise, the development of cervical cancer will only be postponed, if boosters are not implemented.
It is now widely recognized that Cervarix induces high antibody titers, offering 100% efficacy even after 8.4 years, making it very likely that the protection it provides will continue for at least 15 years. It is also widely acknowledged by immunologists that Gardasil-induced titers for HPV 6, 11, and 18 are much shorter-lived, so protection is likely to wane 5 to 10 years after vaccination.
That means that Gardasil provides excellent protection against one cancer-causing type of HPV. In addition, it protects against genital warts caused by HPV types 6 and 11 for at least 5 years. In comparison, Cervarix protects against five cancer-causing types of HPV, thereby preventing about 90% of cervical cancers, and is likely to remain effective for at least 15 years.
There are 10 times as many women who have an abnormal Pap test as there are women who have genital warts, so one would think that Cervarix would be the vaccine of choice in preventing the life-threatening disease of cervical cancer.
Dr. Smith-McCune: I would agree that the choice should be discussed with patients—and with parents. If the objective is primarily to protect against cervical cancer precursors, then the bivalent vaccine may be the better choice, with the caveat that we can’t really compare the results from the vaccine trials for reasons discussed earlier, and there are no data from a randomized head-to-head trial comparing the two vaccines. If the decision involves a desire to reduce genital warts or vulvar and vaginal dysplasia, then the quadrivalent vaccine would be the better choice.
6. What impact do the vaccines have on screening?
Dr. Lonky: Do the vaccines have varying effects on our need to screen for, triage, and treat cervical cancer precursors?
Dr. Harper: The Pap smear has reduced the rate of cervical cancer in the United States by 75%—that rate is now at an all-time low of 8 cases for every 100,000 women. But the Pap smear is not perfect; there is a 30% false-negative rate among women who develop cervical cancer, and a large false-positive rate that involves referral to colposcopy for minimally abnormal cytology reports. And when CIN 2+ disease is detected, treatment is not without risk. Surgery increases the risk of reproductive morbidity in future pregnancies. Having protection against this outcome could be tremendously valuable for some women.
Compare the HPV vaccine, which has probable benefit but also the potential for serious adverse events, including demyelinating diseases that cause blindness, paralysis, and death in a small number of recipients.
If women were to choose to be vaccinated with Gardasil and forgo further Pap screening, the rate of cervical cancer in the United States would rise from 8 to 14 cases for every 100,000 women. If they were to choose Cervarix instead, with no further Pap screening, the rate would rise from 8 to 9.5 cases for every 100,000 women.
If women were to choose both HPV vaccination and continued Pap screening, the rate of cervical cancer still would not decline from its current level of 8 cases for every 100,000 women. Instead, the benefit would be that fewer women have abnormal Pap tests, and fewer women would need to be treated for CIN 2+ disease.
Women and physicians must understand these facts. A woman who chooses to be vaccinated may gain individual protection, but the overall rate of cervical cancer will not be affected.
Dr. Huh: Regardless of the HPV vaccine selected, we need to seriously rethink how we screen women in the United States. One could easily argue that the combination of the vaccine and continued screening is too expensive. It might be wise to consider lengthening the screening interval—and, perhaps, further delaying initial screening to 25 years of age—to make cervical cancer prevention with both modalities more cost-effective.24 The most important thing to recognize is that women still need to be screened, even if they have been vaccinated.
As more women are vaccinated, we expect to see a decline in the prevalence of CIN 2+ and CIN 3+ lesions, and this will ultimately weaken the positive predictive value of cytology. Perhaps it is time to consider the HPV test as a primary screen, with triage to cytology in women who test HPV-positive.25
Dr. Smith-McCune: As we accumulate data over time about the effects of vaccination on the rates of CIN 3 and cancer, modeling will be helpful in determining the best screening algorithm for women who have been vaccinated against HPV.
It is important to remember that approximately 50% of women who are given a diagnosis of cervical cancer in the United States have never been screened. It is vital that we continue to reach out to the under-screened population and focus vaccination efforts on populations of girls who are likely to have limited access to care in the future.
7. Can we vaccinate every woman?
Dr. Lonky: Is universal vaccination of women achievable for either vaccine?
Dr. Felix: Universal vaccination against HPV would be achievable only via school mandates. Without them, vaccination will not approach the 80% threshold needed to produce herd immunity.
Despite the clear benefit of such mandates to the general population—particularly the medically under-served—the issue has become a political football. As a result, school mandates will probably never be realized.
Dr. Harper: I don’t believe it is ethical to mandate vaccination of all girls and women. It is a choice that women and parents, in conversation with their physicians and daughters, must make when considering how to be protected against cervical cancer. Herd immunity is a moot point because we are only vaccinating girls (50% of the population) and can never reach the theoretical 70% threshold for herd immunity to be apparent.
Dr. Lonky: Is the availability of two vaccines a boon or a hindrance?
Dr. Smith-McCune: I think it is always a good thing to have choices in medicine.
Dr. Huh: I see the availability of two vaccines as a boon. That availability means that two companies are now putting forth consistent educational messages about the importance of vaccination and, I hope, stimulating competition that will reduce the overall cost of the vaccine series. Having two vaccines can only promote awareness, access, and greater appreciation of the considerable protection these two vaccines provide.
Despite solid evidence that the quadrivalent (Gardasil) HPV vaccine and the bivalent (Cervarix) HPV vaccine protect against cervical cancer, only about one fifth of the female population between 11 and 26 years of age has received the full series of Gardasil since it won FDA approval in 2006. Barriers to vaccination are not financial alone, as the vaccination rate is similarly low among women who have health insurance.
Why isn’t the vaccination rate higher? I see eight barriers to full implementation:
- Economic disparities. Each vaccine costs roughly $400 (national average) for the full series of injections. Although women who do not get Pap screening are most likely to benefit from the vaccines, they usually cannot afford them. federal childhood immunization programs cover teens and young women until 18 years of age in most states, and until 21 years in a few. that leaves most women who seek vaccination from gynecologists without coverage.
- Fear. Pain at the injection site, syncope, and a slightly elevated incidence of thromboembolism are the adverse events most commonly associated with HPV vaccination in the literature. In the life cycle of a vaccine, reports of sudden death or neurologic injury (Guillain-Barré syndrome) occur in the early years, but are reported at a rate lower than 2 cases in every 10,000 women. Nevertheless, such events may create fear about undergoing immunization.
- Long latency period. Because the outcome of cancer prevention won’t become apparent for 20 to 40 years following vaccination, the need for immunization may seem less than urgent.
- Cultural and religious beliefs. Because carcinogenic HPV strains are sexually transmitted, some families may associate vaccination with the promotion of sexual activity. Even in states that mandate vaccination, the courts have upheld a parent’s right to refuse vaccination on these grounds.
- The premarket push. Aggressive promotion of vaccination by both manufacturers and a push by advocates for legislation to mandate the vaccine prior to completion of Phase-3 trials and gathering of robust safety data may have diminished trust in the vaccine and reduced its acceptance.
- Lack of legislation. In states that do not consider HPV vaccination to be a necessary public health intervention, the lack of mandates and funding reduce the vaccination rate. In addition, some legislators have been more active advocates of vaccination than others.
- Reduced involvement of the obGyn. ObGyns don’t routinely vaccinate patients; pediatricians do. Young women are slipping through the cracks because the conventional ObGyn practice does not have a vaccination program that ensures payment, reimbursement, and completion of the vaccine series. Many ObGyn practices are reluctant to institute such a program because the profit margin is small, there are associated risks, and the time required to counsel the patient and for follow-up is extensive.
- Failure to complete the series. Some women do not complete the full vaccine series, owing to cost or side effects, or both. Solid evidence that a single dose could be as protective as the full series would be compelling. A single-dose vaccine would also be less expensive.
The principal danger of a low vaccination rate is the loss of insurance coverage for immunization against HPV. On one hand, payers may begin to ask whether coverage is justified when so few girls and women are vaccinated, leaving the payer with two burdens: the expense of vaccination and the expense of conventional screening programs and treatment, although the costs of treatment would be reduced with vaccination. On the other hand, Gardasil’s protection against genital warts may provide incentive for payers to cover or discount the vaccine because of the reduction in the need to diagnose, triage, and treat condyloma.
Ultimately, HPV vaccination may become another optional intervention that is paid for by the individual, despite evidence in girls and women that cervical cancer can be prevented.
—NEAL M. LONKY, MD, MPH
We want to hear from you! Tell us what you think.
Not long ago (in medical years), we were still trying to discover the cause of cervical cancer. Today, not only do we know that cause to be persistent human papillomavirus (HPV) infection, but we have two vaccines at our disposal to prevent the primary oncogenic strains of the virus.
We’ve come a long way.
The availability of two vaccines raises questions, however. What kind of data do we have on the bivalent (Cervarix, GlaxoSmithKline) and quadrivalent (Gardasil, Merck) vaccines so far? Is one of them clearly superior to the other? If not, what population is each vaccine best suited for—and how do we counsel patients about their options?
To address these and other questions, OBG Management Contributing Editor Neal M. Lonky, MD, MPH, assembled a panel of physicians who have expertise in cervical disease detection and prevention and asked them to sift the data that have accumulated thus far. In the discussion that follows, they touch on long-term efficacy, the likely impact of the vaccines on cervical cancer screening, and other aspects of disease prevention in the era of HPV vaccination.

Juan C. Felix, MD
Professor of Clinical Pathology and Obstetrics and Gynecology; Director of Cytopathology fellowship; and Chief of Gynecologic Pathology at the Keck School of Medicine, University of Southern California; and Chief of Cytopathology at Los Angeles County and University of Southern California Medical Center in Los Angeles.
Dr. Felix reports that he is a speaker for Merck and GlaxoSmithKline.

Diane M. Harper, MD, MS, MPH
Director of the Gynecologic Cancer Prevention Research Group and Professor of Obstetrics and Gynecology, Community and family Medicine, and Informatics and Personalized Medicine at the University of Missouri–Kansas City School of Medicine.
Dr. Harper reports that she has served as a speaker and advisor for Merck and GlaxoSmithKline, and that the institutions at which she conducted HPV vaccination trials have received funding from Merck and GlaxoSmithKline.

Warner K. Huh, MD
Associate Professor in the Department of Obstetrics and Gynecology, and Associate Scientist at the Comprehensive Cancer Center at the University of Alabama– Birmingham.
Dr. Huh reports that he receives grant or research support from and is a speaker and consultant to Merck and GlaxoSmithKline.

Karen K. Smith-McCune, MD, PhD
John Kerner Endowed Chair of Gynecologic Oncology, Director of the Dysplasia Clinic, and Professor of Obstetrics, Gynecology, and Reproductive Sciences at the University of California–San francisco.
Dr. Smith-McCune reports she has performed unpaid consulting for OncoHealth Inc. and is planning to join its Scientific Advisory Board.
1. How were the vaccines developed?
Neal M. Lonky, MD, MPH: What should clinicians know about the development, function, and mechanism of action of the two HPV vaccines?
Warner K. Huh, MD: The bivalent and quadrivalent vaccines are both excellent products, and their respective Phase-3 trials demonstrate that they provide impressive protection against HPV, particularly among women who test negative (by polymerase chain reaction) for the specific HPV types contained within the vaccines.1-3
Cervarix protects against HPV types 16 and 18, whereas Gardasil is effective against HPV types 6, 11, 16, and 18.
Dr. Lonky: Do the vaccines function similarly?
Diane M. Harper, MD, MS, MPH: Yes. Both stimulate an immediate antibody response in the woman who is not infected with the relevant virus and are effective in preventing cervical intraepithelial neoplasia grade 2 and higher (CIN 2+), as well as persistent infection, caused by vaccine-related and cross-protected HPV types. The quality of the antibody response is best for HPV 16 for both vaccines. The quality of the antibody response for HPV 6, 11, and 18 for Gardasil is much poorer than its response for HPV 16. Cervarix induces an equally high and sustained antibody response for HPV 18 as for HPV 16.
Juan C. Felix, MD: Both vaccines are based on the same virus-like particles (VLP). The functionality of the vaccines is, therefore, mainly dependent on the dosage of VLP and the adjuvant used. Gardasil uses a proprietary aluminum sulfate adjuvant, whereas Cervarix uses aluminum hydroxide and monophosphoryl lipid A.
Karen K. Smith-McCune, MD, PhD: Both adjuvants have an extensive track record of safety and efficacy in other vaccines. Because they have different structures, however, they may have varying effects on many components of the immune response elicited by the L1 antigens.
Dr. Harper: Both adjuvants contain aluminum, which has so far proved to be safe despite the newly established association between high aluminum intake and Alzheimer’s disease.
Dr. Lonky: Were there any notable challenges in developing the vaccines?
Dr. Harper: It was difficult to formulate the appropriate dosages of VLP in Gardasil. Higher dosages of HPV 11 and 16 were needed to prevent cross-inhibition by HPV 6 and 18. As a result, the antigenic protein component of Gardasil that is necessary to effect an immunologic antibody response is high, at 120 μg. In Cervarix, the antigenic VLP load is 20 μg each for HPV 16 and 18.
Dr. Lonky: What is the significance of the different VLP loads?
Dr. Harper: Side effects, such as autoimmune neurologic demyelination, albeit rare, have been associated with a higher antigenic protein load. Multiple reports of autoimmune demyelinating diseases—including paralysis, blindness, and death—have been published by neurologists in regard to Gardasil.4,5 Others have shown that young girls are more at risk than young boys for these neurologic side effects.6
Dr. Felix: Some data suggest that the two vaccine formulations interact differently with the human immune system. In a head-to-head trial funded by GlaxoSmithKline, Cervarix produced higher total and neutralizing antibody titers than Gardasil did.7
Although higher immunogenicity is generally thought to be beneficial, the ultimate determinant of a vaccine’s success is its efficacy—and duration of that efficacy—in clinical trials and follow-up of vaccinated populations. So far, Cervarix has demonstrated efficacy through 8.4 years in its follow-up cohort.8 Similarly, Gardasil has proved to be effective after 5 years of follow-up, with no incident cases of cervical cancer reported in the vaccinated arm.9

2. Does either vaccine offer “extra” immunity?
Dr. Lonky: What is the potential for overlapping immunity to other high-risk viral types with these vaccines?
Dr. Harper: It is quite clear from pivotal trials of both vaccines that Gardasil produces efficacy of 46% against persistent infection caused by HPV 31. Data from the pivotal Phase-3 trial of Gardasil also show that it offers no protection against persistent infection with HPV 45, an important cause of adenocarcinoma.10
In contrast, Cervarix demonstrates substantial efficacy against both persistent infection and CIN 2+ disease caused by HPV 31, 33, and 45.3
These findings mean that Cervarix is 91% effective against HPV types that cause adenocarcinoma and 83% effective overall against squamous cell carcinoma. Compare that with Gardasil, which is 78% effective overall against HPV types that cause adenocarcinoma and 73% effective against HPV types that cause squamous cell carcinoma.
The immune titers tell a supportive story. After vaccination with Gardasil, the antibody titer immediately declines for HPV 6, 11, and 18, reaching the baseline for natural infection within 18 months.7 HPV 18 shows continued, significant loss of seropositivity over time, and antibody titers for HPV 6 and 11 also decline. In the monovalent HPV 16 pre-Gardasil experimental vaccine, 14% of women no longer had measurable titers to HPV 16 after 8.5 years.11
After vaccination with Cervarix, antibody titers for HPV 16 and 18 remain more than seven times and more than four times higher, respectively, than natural infection titers for 8.4 years, with no loss of measurable antibody titer for either type. The antibody titers for HPV 31, 33, and 45 remain substantially higher than natural infection titers for at least 6.4 years. These titers correlate with the vaccine’s very high efficacy against CIN 2+ lesions caused by HPV 16, 18, 31, 33, and 45.
In other words, Cervarix generates an immune response (and efficacy) that indicates robust protection against five of the most common oncogenic HPV types, providing maximal protection against nearly 85% of all cervical cancers. Gardasil protects against 74% of all cervical cancers overall.12 This makes Cervarix the superior cervical cancer vaccine.
Gardasil is the superior vaccine against genital warts, although the duration of its protection is uncertain.
Dr. Huh: I’d just like to point out that there are no head-to-head trials comparing the vaccines in terms of efficacy. Antibody titers are higher with Cervarix than with Gardasil, as you noted, and it may be that, over time, the higher titers are more durable with Cervarix. However, we have yet to fully correlate clinical efficacy with antibody titers. In other words, immunogenicity does not equal clinical efficacy.
Dr. Felix: The data for Gardasil are particularly interesting because there have been no incident HPV-18 lesions detected despite the absence of detectable HPV-18 antibody titers in more than 20% of vaccinated women as soon as 2 years after immunization.9 These data strongly suggest that it is not antibody titer alone that grants protection against HPV-induced lesions of the cervix.
Dr. Harper: This speaks to the difficulty of running a trial to ensure both enough participants and a sufficient attack rate of HPV 18 to cause new lesions to be detected in vaccinated women. In the relevant trial, there were only 112 vaccinated women—not nearly enough women to overcome the very low attack rate of HPV 18 in the trial population—and they were followed for 5 years.9 We cannot be sure that the lack of incident HPV-18 lesions in the vaccinated women is the result of efficacy.
Dr. Felix: As for overlapping immunity to HPV types not included in the vaccines, it has been described for both Cervarix and Gardasil. In the case of Cervarix, the manufacturer demonstrated unexpectedly high rates of protection against all CIN 2+ and CIN 3+ lesions—70% and 87%, respectively. These rates were too high to be explained by protection against types 16, 18, 31, and 45 alone. It is possible, therefore, that Cervarix may protect against other high-risk HPV types.13
Gardasil has proved to be effective against HPV types 31, 33, 52, and others.10 When total protection against CIN 2+ and CIN 3+ lesions is examined from Phase-3 trials of the vaccine, however, the rates are only 42% and 43%, respectively. These data are difficult to interpret because HPV 16 and 18 together are thought to account for 70% of CIN 3. Some reassurance can be gained from the fact that the number of incident cases of CIN 2+ and CIN 3+ caused by HPV 16 and 18 in the vaccinated group in the Gardasil trial was identical to the number seen in the Cervarix trial.3,10 The reason for the discrepancy in total number of cases of CIN 2+ and CIN 3+ between the two trials—and, therefore, between the two vaccines—cannot be explained by cross-protection alone and is probably attributable to differences in study populations. The Gardasil trial had a higher baseline prevalence of HPV 16 and 18 (9% and 4%, respectively) than the Cervarix trial did (5% and 2%, respectively), a fact that may be explained by the different demographics of their respective populations.2,14
Ultimately, it is hazardous to compare trials, particularly when they are conducted in significantly different populations. On this issue, I concur with the World Health Organization (WHO), which recommended that such comparisons be avoided in the determination of which type of HPV vaccine to recommend.15
Dr. Huh: I agree that it would be inappropriate to make cross-trial comparisons, given differences in the way the trials were designed and conducted. To draw conclusions about clinical efficacy of these two excellent vaccines, based on a comparison of their trials, is completely unscientific. Only a true head-to-head study that has efficacy as its endpoint can tell us which vaccine is superior—and such a trial would require thousands (if not tens of thousands) of subjects and a considerable amount of time to complete. In my opinion, such a study would be counterproductive to our goal of vaccination.
Dr. Harper: I disagree. The whole purpose of this roundtable is to compare vaccines. It is not “unscientific” to compare the trials.
Dr. Huh: On the contrary—it is completely inappropriate to directly compare the Phase-3 clinical trials from Merck and GlaxoSmithKline. One can speculate about the differences between them, but any clinical trialist knows that a direct, scientific comparison cannot be made. Only a real head-to-head study powered for efficacy can do this.
- Both the bivalent and quadrivalent vaccines appear to be excellent products. Besides protecting against the main oncogenic strains of human papillomavirus (HPV) (types 16 and 18 for both vaccines, and the genital-wart-associated strains 6 and 11 for the quadrivalent vaccine), both Cervarix and Gardasil offer some degree of cross-protection against additional HPV strains.
- Vaccination of the sexually naïve patient with either vaccine provides significant protection against cervical intraepithelial neoplasia 2 (CIN 2) or worse.
- HPV vaccination is expected to reduce the rate of abnormal Pap tests and the need for common excisional treatments for cervical dysplasia in vaccinated women. It will do the same in the population as a whole if rates of vaccination are sufficient to provide “herd” immunity.
3. Is one vaccine more effective than the other?
Dr. Lonky: How do the vaccines compare in terms of efficacy?
Dr. Smith-McCune: In discussing efficacy, I think we should focus on CIN 3 because it is the immediate surrogate for cancer, whereas CIN 2 lesions can be transient in younger women. I think it is also important to focus on outcomes regardless of the HPV types associated with the lesions. This approach is more clinically relevant, as we don’t perform HPV typing of lesions in clinical practice. Nor do we manage lesions differently depending on the HPV type in the lesion.
That said, it is difficult to compare efficacy of the vaccines for several reasons, a few of which we have already discussed. For example, the bivalent and quadrivalent vaccines were studied in separate randomized trials. Although the study populations were similar, they were not identical. Women in both trials were relatively sexually naïve, but the cutoff for number of lifetime sexual partners was different (5 for Gardasil versus 7 for Cervarix). In trials of Gardasil, women who had a history of abnormal cytology or genital warts were excluded. In trials of Cervarix, women who had a history of colposcopy were excluded. In Gardasil trials, approximately 3% of women were from the Asian Pacific, versus 34% in the Cervarix trials, and so on.3,16
The trials also had different protocols for referral to colposcopy, which would affect disease detection. And the length of follow-up differs between trials.3,9
Dr. Lonky: Can we draw any conclusions about efficacy?
Dr. Smith-McCune: Yes. The trials defined outcomes in several populations of participants. In addition to the overall population (called the “intention-to-treat population” in the Gardasil trials and the “total vaccinated cohort” in the Cervarix trials), the trials defined a subpopulation of women naïve to oncogenic HPV types to gain information about the likely impact of vaccinating girls before the onset of sexual activity. The definitions of these “naïve” populations were slightly different, mainly in the number of HPV types tested, so again, some caution needs to be exercised in making comparisons.
End-of-trial data in the naïve population show a 43% reduction in CIN 3 lesions for Gardasil and 87% for Cervarix (for CIN 3 or worse). By inference, we can tell the sexually naïve patient that vaccination with either vaccine will provide significant protection against CIN 3 lesions, likely to result in significant protection against cervical cancer over time.
We can gather some estimates of efficacy in sexually non-naïve women by looking at results from all trial participants. Gardasil reduced overall CIN 3 lesions by 16% overall; Cervarix reduced CIN 3 or worse by 33%. When counseling an individual patient, if she has had a similarly low number of lifetime sexual partners (e.g., the median number in the Gardasil trials was 2), these results provide an estimate of her likely protection against CIN 3 with vaccination.
Common excisional treatments for cervical dysplasia are known to be associated with adverse perinatal outcomes.17 The ability to reduce the need for these treatments is an important outcome of vaccination. In the HPV-naïve populations, vaccination reduced definitive cervical therapies or excisions by 42% (Gardasil) and 69% (Cervarix). These figures are useful in counseling virginal patients about the long-term benefits of vaccination.
For sexually active patients 26 years and younger, HPV vaccination significantly reduced definitive cervical therapy or excisions by 23% (Gardasil) and 25% (Cervarix). Again, these figures are most applicable for counseling patients who have had relatively few lifetime sexual partners. So the exact extent of protection is likely to vary by the patient’s total number of lifetime sexual partners.
I expect that we will see more data on the effects of vaccination stratified by the number of lifetime sexual partners, because that information would be very useful in counseling individual sexually active women.
Dr. Harper: Both vaccines reduce the rate of abnormal Pap tests by 10% regardless of HPV type in that population of women.9,18
4. Are the two vaccines safe?
Dr. Lonky: What about safety of the vaccines? What do we know?
Dr. Felix: The safety profiles seen in clinical trials of both vaccines are very similar and consist almost entirely of nonserious adverse events.2,9 In the United States, a greater number of Gardasil doses has been administered, owing to its earlier development. As of January 1, 2010, more than 28 million doses had been distributed, and numerous major events had been recorded in the Vaccine Adverse Event Reporting System (VAERS). Of 15,829 adverse events reported, only 8% were considered serious by the CDC. CDC investigation, by expert panels, of all serious adverse events found no evidence linking Gardasil to any of them, including Guillain-Barré syndrome, blood clots, and death.19
Dr. Huh: A few other points to consider:
- The reporting rate for Gardasil is triple that for all other vaccines combined
- Because VAERS is a passive reporting system, under-reporting is distinctly possible
- Post-licensure safety surveillance is still underway
- Both products have pregnancy registries.
Dr. Harper: The current postmarketing commitment between Merck and the FDA is to recognize a rate of serious adverse events that exceeds 2 cases in every 10,000 women in a cohort of 44,000 women who have received all three doses of Gardasil. Although autoimmune neurologic sequelae have occurred after Gardasil administration, regulatory authorities are not required to evaluate these reactions, such as Guillain-Barré syndrome, because the frequency is lower than the agreed-upon threshold. Nevertheless, adverse events could be life threatening to some girls.
Any risk of death—even if it is lower than the agreed-upon threshold—should be presented to women as a possible risk of vaccination with Gardasil. In the United States, the same women could choose a lifetime of Pap screening and be afforded the same protection against cervical cancer as they would get from vaccination.
5. Is quadrivalent better than bivalent?
Dr. Lonky: Why would a clinician choose a bivalent vaccine when the quadrivalent vaccine protects not only against carcinogenic types 16 and 18, but also against HPV-associated genital warts?
Dr. Harper: A smart clinician would ask the patient what she values. The physician is obligated to present the evidence and let her choose!
Dr. Lonky: What does the evidence suggest?
Dr. Felix: A clear recommendation between Cervarix and Gardasil is very difficult to make at this time, for the reasons already stated. Both vaccines provide 98% protection against HPV 16 and 18 for the prevention of CIN 2+ lesions.3,9 As we have discussed, both vaccines also provide protection against high-risk strains of HPV other than types 16 and 18.
In the Cervarix trial, there was an overall reduction of all CIN 2+ lesions that was higher as a percentage of total lesions than the reduction seen in the Gardasil trial.3,9 However, unlike Cervarix, Gardasil significantly reduced the rate of vaginal intraepithelial neoplasia (VaIN) and vulvar intraepithelial neoplasia (VIN).9
Dr. Harper: GlaxoSmithKline is analyzing its data on vulvar and vaginal protection, and it is likely that Cervarix will demonstrate some efficacy in this regard, too. But the economic burden of noncervical cancers is estimated to be only 8% of the economic burden of all HPV-related diseases.20 The prevention of cervical cancer is the dominant clinical and economic force for vaccination.
Dr. Felix: Clearly, the protection against genital warts demonstrated in the Gardasil trial will not be realized with Cervarix, as it does not offer immunization or cross-protection against HPV 6 or 11. Gardasil’s protection against HPV 6 and 11 prompted FDA approval of the vaccine for boys and men.21
According to the WHO, when counseling girls and women about the HPV vaccine, the clinician should weigh the possible value of a deep reduction in total CIN 2+ lesions provided by Cervarix against the reduction in VaIN, VIN, and genital warts provided by Gardasil.15 Boys and men will see clinically proven benefits only from Gardasil for the prevention of external genital warts. Other benefits are strictly theoretical.22,23
Dr. Harper: Vaccine protection must last at least 15 years to reduce the rate of cervical cancer. Otherwise, the development of cervical cancer will only be postponed, if boosters are not implemented.
It is now widely recognized that Cervarix induces high antibody titers, offering 100% efficacy even after 8.4 years, making it very likely that the protection it provides will continue for at least 15 years. It is also widely acknowledged by immunologists that Gardasil-induced titers for HPV 6, 11, and 18 are much shorter-lived, so protection is likely to wane 5 to 10 years after vaccination.
That means that Gardasil provides excellent protection against one cancer-causing type of HPV. In addition, it protects against genital warts caused by HPV types 6 and 11 for at least 5 years. In comparison, Cervarix protects against five cancer-causing types of HPV, thereby preventing about 90% of cervical cancers, and is likely to remain effective for at least 15 years.
There are 10 times as many women who have an abnormal Pap test as there are women who have genital warts, so one would think that Cervarix would be the vaccine of choice in preventing the life-threatening disease of cervical cancer.
Dr. Smith-McCune: I would agree that the choice should be discussed with patients—and with parents. If the objective is primarily to protect against cervical cancer precursors, then the bivalent vaccine may be the better choice, with the caveat that we can’t really compare the results from the vaccine trials for reasons discussed earlier, and there are no data from a randomized head-to-head trial comparing the two vaccines. If the decision involves a desire to reduce genital warts or vulvar and vaginal dysplasia, then the quadrivalent vaccine would be the better choice.
6. What impact do the vaccines have on screening?
Dr. Lonky: Do the vaccines have varying effects on our need to screen for, triage, and treat cervical cancer precursors?
Dr. Harper: The Pap smear has reduced the rate of cervical cancer in the United States by 75%—that rate is now at an all-time low of 8 cases for every 100,000 women. But the Pap smear is not perfect; there is a 30% false-negative rate among women who develop cervical cancer, and a large false-positive rate that involves referral to colposcopy for minimally abnormal cytology reports. And when CIN 2+ disease is detected, treatment is not without risk. Surgery increases the risk of reproductive morbidity in future pregnancies. Having protection against this outcome could be tremendously valuable for some women.
Compare the HPV vaccine, which has probable benefit but also the potential for serious adverse events, including demyelinating diseases that cause blindness, paralysis, and death in a small number of recipients.
If women were to choose to be vaccinated with Gardasil and forgo further Pap screening, the rate of cervical cancer in the United States would rise from 8 to 14 cases for every 100,000 women. If they were to choose Cervarix instead, with no further Pap screening, the rate would rise from 8 to 9.5 cases for every 100,000 women.
If women were to choose both HPV vaccination and continued Pap screening, the rate of cervical cancer still would not decline from its current level of 8 cases for every 100,000 women. Instead, the benefit would be that fewer women have abnormal Pap tests, and fewer women would need to be treated for CIN 2+ disease.
Women and physicians must understand these facts. A woman who chooses to be vaccinated may gain individual protection, but the overall rate of cervical cancer will not be affected.
Dr. Huh: Regardless of the HPV vaccine selected, we need to seriously rethink how we screen women in the United States. One could easily argue that the combination of the vaccine and continued screening is too expensive. It might be wise to consider lengthening the screening interval—and, perhaps, further delaying initial screening to 25 years of age—to make cervical cancer prevention with both modalities more cost-effective.24 The most important thing to recognize is that women still need to be screened, even if they have been vaccinated.
As more women are vaccinated, we expect to see a decline in the prevalence of CIN 2+ and CIN 3+ lesions, and this will ultimately weaken the positive predictive value of cytology. Perhaps it is time to consider the HPV test as a primary screen, with triage to cytology in women who test HPV-positive.25
Dr. Smith-McCune: As we accumulate data over time about the effects of vaccination on the rates of CIN 3 and cancer, modeling will be helpful in determining the best screening algorithm for women who have been vaccinated against HPV.
It is important to remember that approximately 50% of women who are given a diagnosis of cervical cancer in the United States have never been screened. It is vital that we continue to reach out to the under-screened population and focus vaccination efforts on populations of girls who are likely to have limited access to care in the future.
7. Can we vaccinate every woman?
Dr. Lonky: Is universal vaccination of women achievable for either vaccine?
Dr. Felix: Universal vaccination against HPV would be achievable only via school mandates. Without them, vaccination will not approach the 80% threshold needed to produce herd immunity.
Despite the clear benefit of such mandates to the general population—particularly the medically under-served—the issue has become a political football. As a result, school mandates will probably never be realized.
Dr. Harper: I don’t believe it is ethical to mandate vaccination of all girls and women. It is a choice that women and parents, in conversation with their physicians and daughters, must make when considering how to be protected against cervical cancer. Herd immunity is a moot point because we are only vaccinating girls (50% of the population) and can never reach the theoretical 70% threshold for herd immunity to be apparent.
Dr. Lonky: Is the availability of two vaccines a boon or a hindrance?
Dr. Smith-McCune: I think it is always a good thing to have choices in medicine.
Dr. Huh: I see the availability of two vaccines as a boon. That availability means that two companies are now putting forth consistent educational messages about the importance of vaccination and, I hope, stimulating competition that will reduce the overall cost of the vaccine series. Having two vaccines can only promote awareness, access, and greater appreciation of the considerable protection these two vaccines provide.
Despite solid evidence that the quadrivalent (Gardasil) HPV vaccine and the bivalent (Cervarix) HPV vaccine protect against cervical cancer, only about one fifth of the female population between 11 and 26 years of age has received the full series of Gardasil since it won FDA approval in 2006. Barriers to vaccination are not financial alone, as the vaccination rate is similarly low among women who have health insurance.
Why isn’t the vaccination rate higher? I see eight barriers to full implementation:
- Economic disparities. Each vaccine costs roughly $400 (national average) for the full series of injections. Although women who do not get Pap screening are most likely to benefit from the vaccines, they usually cannot afford them. federal childhood immunization programs cover teens and young women until 18 years of age in most states, and until 21 years in a few. that leaves most women who seek vaccination from gynecologists without coverage.
- Fear. Pain at the injection site, syncope, and a slightly elevated incidence of thromboembolism are the adverse events most commonly associated with HPV vaccination in the literature. In the life cycle of a vaccine, reports of sudden death or neurologic injury (Guillain-Barré syndrome) occur in the early years, but are reported at a rate lower than 2 cases in every 10,000 women. Nevertheless, such events may create fear about undergoing immunization.
- Long latency period. Because the outcome of cancer prevention won’t become apparent for 20 to 40 years following vaccination, the need for immunization may seem less than urgent.
- Cultural and religious beliefs. Because carcinogenic HPV strains are sexually transmitted, some families may associate vaccination with the promotion of sexual activity. Even in states that mandate vaccination, the courts have upheld a parent’s right to refuse vaccination on these grounds.
- The premarket push. Aggressive promotion of vaccination by both manufacturers and a push by advocates for legislation to mandate the vaccine prior to completion of Phase-3 trials and gathering of robust safety data may have diminished trust in the vaccine and reduced its acceptance.
- Lack of legislation. In states that do not consider HPV vaccination to be a necessary public health intervention, the lack of mandates and funding reduce the vaccination rate. In addition, some legislators have been more active advocates of vaccination than others.
- Reduced involvement of the obGyn. ObGyns don’t routinely vaccinate patients; pediatricians do. Young women are slipping through the cracks because the conventional ObGyn practice does not have a vaccination program that ensures payment, reimbursement, and completion of the vaccine series. Many ObGyn practices are reluctant to institute such a program because the profit margin is small, there are associated risks, and the time required to counsel the patient and for follow-up is extensive.
- Failure to complete the series. Some women do not complete the full vaccine series, owing to cost or side effects, or both. Solid evidence that a single dose could be as protective as the full series would be compelling. A single-dose vaccine would also be less expensive.
The principal danger of a low vaccination rate is the loss of insurance coverage for immunization against HPV. On one hand, payers may begin to ask whether coverage is justified when so few girls and women are vaccinated, leaving the payer with two burdens: the expense of vaccination and the expense of conventional screening programs and treatment, although the costs of treatment would be reduced with vaccination. On the other hand, Gardasil’s protection against genital warts may provide incentive for payers to cover or discount the vaccine because of the reduction in the need to diagnose, triage, and treat condyloma.
Ultimately, HPV vaccination may become another optional intervention that is paid for by the individual, despite evidence in girls and women that cervical cancer can be prevented.
—NEAL M. LONKY, MD, MPH
We want to hear from you! Tell us what you think.
1. The FUTURE II Study Group. Quadrivalent vaccine against human papillomavirus to prevent high-grade cervical lesions. N Engl J Med. 2007;356(19):1915-1927.
2. Paavonen J, Jenkins D, Bosch FX, Naud P, Salmerón J, Wheeler CM, et al. HPV PATRICIA study group. Efficacy of a prophylactic adjuvanted bivalent L1 virus-like-particle vaccine against infection with human papillomavirus types 16 and 18 in young women: an interim analysis of a phase III double-blind, randomised controlled trial. Lancet. 2007;369(9580):2161-2170.
3. Paavonen J, Naud P, Salmeron J, et al. For the HPV PATRICIA Study Group. Efficacy of human papillomavirus (HPV)-16/18 AS04-adjuvanted vaccine against cervical infection and precancer caused by oncogenic HPV types (PATRICIA): final analysis of a double-blind, randomised study in young women. Lancet. 2009;374(9686):301-314.
4. DiMario FJ, Hajjar M, Ciesielski T. A 16-year-old girl with bilateral visual loss and left hemiparesis following an immunization against human papillomavirus. J Child Neurology. 2010;25(3):321-327.
5. Sutton I, Lahoria R, Tan I, Clouston P, Barnett M. CNS demyelination and quadrivalent HPV vaccination. Multiple Sclerosis. 2009;15(1):116-119.
6. Klein SL, Jedlicka A, Pekosz A. The Xs and Y of immune responses to viral vaccines. Lancet Infect Dis. 2010;10(5):338-349.
7. Einstein MH, Baron M, Levin MJ, et al. Comparison of the immunogenicity and safety of Cervarix and Gardasil human papillomavirus (HPV) cervical cancer vaccines in healthy women aged 18-45 years. Hum Vaccin. 2009;5(10):705-719.
8. GlaxoSmithKline Vaccine HPV-007 Study Group. Sustained efficacy and immunogenicity of the HPV-16/18 AS04-adjuvanted vaccine: analysis of a randomised placebo-controlled trial up to 8.4 years. Presented at: 28th Annual Meeting of the European Society for Paediatric Infectious Diseases (ESPID); May 4-8, 2010; Nice, France.
9. Muñoz N, Kjaer SK, Sigurdsson K, et al. Impact of human papillomavirus (HPV)-6/11/16/18 vaccine on all HPV-associated genital diseases in young women. J Natl Cancer Inst. 2010;102(5):325-339.
10. Brown DR, Kjaer SK, Sigurdsson K, et al. The impact of quadrivalent human papillomavirus (HPV; types 6, 11, 16, and 18) L1 virus-like particle vaccine on infection and disease due to oncogenic nonvaccine HPV types in generally HPV-naïve women aged 16-26 years. J Infect Dis. 2009;199(7):926-935.
11. Rowhani-Rahbar A, Mao C, Hughes JP, et al. Longer term efficacy of a prophylactic monovalent human papillomavirus type 16 vaccine. Vaccine. 2009;27(41):5612-5619.
12. Harper DM, Williams KB. Prophylactic HPV vaccines: current knowledge of impact on gynecologic premalignancies [published online ahead of print July 3, 2010]. Discovery Medicine. 2010;10(50).http://www.discoverymedicine.com/Diane-M-Harper/2010/07/03/prophylactichpv-vaccines-current-knowledge-of-impact-ongynecologic-premalignancies. Accessed July 13, 2010.
13. Cervarix [human papillomavirus bivalent (types 16 and 18) vaccine recombinant]. Food and Drug Administration. Initial US approval: 2009:1–12.
14. Garland SM, Hernandez-Avila M, Wheeler CM, et al. Quadrivalent vaccine against human papillomavirus to prevent anogenital diseases. N Engl J Med. 2007;356(19):1928-1943.
15. World Health Organization. Human papillomavirus vaccines: WHO position paper. Weekly Epidemiological Record. 2009;84(15):118-131.http://www.who.int/wer/2009/wer8415/en/index.html. Published April 10, 2009. Accessed July 13, 2010.
16. Ault KA. For Future II Study Group. Effect of prophylactic human papillomavirus L1 virus-like-particle vaccine on the risk of cervical intraepithelial neoplasia grade 2, grade 3, and adenocarcinoma in situ: a combined analysis of four randomised clinical trials. Lancet. 2007;369(9576):1861-1868.
17. Arbyn M, Kyrgiou M, Simoens C, et al. Perinatal mortality and other severe adverse pregnancy outcomes associated with treatment of cervical intraepithelial neoplasia: meta-analysis. BMJ. 2008;337:a1284.-Doi: 10.1136/bmj.a1284.
18. Paavonen J. For the HPV PATRICIA Study Group. Efficacy of HPV 16/18 ASO4-adjuvanted vaccine against abnormal cytology, colposcopy referrals, and cervical procedures. Abstract SS 4–1. Data presented at Cervical Cancer Prevention, EuroGin 2010. European Research Organisation on Genital Infection and Neoplasia (EUROGIN); February 17-20, 2010; Monte Carlo, Monaco.http://www.eurogin.com/2010/programoverview.html. Accessed July 13, 2010.
19. Centers for Disease Control and Prevention. Reports of health concerns following HPV vaccination. 2010;1-3.http://www.cdc.gov/vaccinesafety/Vaccines/HPV/gardasil.html. Published June 21, 2010. Accessed July 13, 2010.
20. Myers ER. The economic impact of HPV vaccines: not just cervical cancer. Am J Obstet Gynecol. 2008;198(5):487-488.
21. Gardasil [human papillomavirus quadrivalent (types 6, 11, 16, and 18) vaccine, recombinant]. Food and Drug Administration; 2009. Initial Approval 2006;1-26.
22. Olsson SE, Kjaer SK, Sigurdsson K, et al. Evaluation of quadrivalent HPV 6/11/16/18 vaccine efficacy against cervical and anogenital disease in subjects with serological evidence of prior vaccine type HPV infection. Hum Vaccin. 2009;5(10).
23. Pirotta M, Ung L, Stein A, et al. The psychosocial burden of human papillomavirus related disease and screening interventions. Sex Transm Infect. 2009;85(7):508-513.
24. Kim JJ, Goldie SJ. Health and economic implications of HPV vaccination in the United States. N Engl J Med. 2008;359(8):821-832.
25. Cox JT. Is the HPV test effective as the primary screen for cervical cancer? Examining the Evidence. OBG Management. 2010;22(7):10-11.
1. The FUTURE II Study Group. Quadrivalent vaccine against human papillomavirus to prevent high-grade cervical lesions. N Engl J Med. 2007;356(19):1915-1927.
2. Paavonen J, Jenkins D, Bosch FX, Naud P, Salmerón J, Wheeler CM, et al. HPV PATRICIA study group. Efficacy of a prophylactic adjuvanted bivalent L1 virus-like-particle vaccine against infection with human papillomavirus types 16 and 18 in young women: an interim analysis of a phase III double-blind, randomised controlled trial. Lancet. 2007;369(9580):2161-2170.
3. Paavonen J, Naud P, Salmeron J, et al. For the HPV PATRICIA Study Group. Efficacy of human papillomavirus (HPV)-16/18 AS04-adjuvanted vaccine against cervical infection and precancer caused by oncogenic HPV types (PATRICIA): final analysis of a double-blind, randomised study in young women. Lancet. 2009;374(9686):301-314.
4. DiMario FJ, Hajjar M, Ciesielski T. A 16-year-old girl with bilateral visual loss and left hemiparesis following an immunization against human papillomavirus. J Child Neurology. 2010;25(3):321-327.
5. Sutton I, Lahoria R, Tan I, Clouston P, Barnett M. CNS demyelination and quadrivalent HPV vaccination. Multiple Sclerosis. 2009;15(1):116-119.
6. Klein SL, Jedlicka A, Pekosz A. The Xs and Y of immune responses to viral vaccines. Lancet Infect Dis. 2010;10(5):338-349.
7. Einstein MH, Baron M, Levin MJ, et al. Comparison of the immunogenicity and safety of Cervarix and Gardasil human papillomavirus (HPV) cervical cancer vaccines in healthy women aged 18-45 years. Hum Vaccin. 2009;5(10):705-719.
8. GlaxoSmithKline Vaccine HPV-007 Study Group. Sustained efficacy and immunogenicity of the HPV-16/18 AS04-adjuvanted vaccine: analysis of a randomised placebo-controlled trial up to 8.4 years. Presented at: 28th Annual Meeting of the European Society for Paediatric Infectious Diseases (ESPID); May 4-8, 2010; Nice, France.
9. Muñoz N, Kjaer SK, Sigurdsson K, et al. Impact of human papillomavirus (HPV)-6/11/16/18 vaccine on all HPV-associated genital diseases in young women. J Natl Cancer Inst. 2010;102(5):325-339.
10. Brown DR, Kjaer SK, Sigurdsson K, et al. The impact of quadrivalent human papillomavirus (HPV; types 6, 11, 16, and 18) L1 virus-like particle vaccine on infection and disease due to oncogenic nonvaccine HPV types in generally HPV-naïve women aged 16-26 years. J Infect Dis. 2009;199(7):926-935.
11. Rowhani-Rahbar A, Mao C, Hughes JP, et al. Longer term efficacy of a prophylactic monovalent human papillomavirus type 16 vaccine. Vaccine. 2009;27(41):5612-5619.
12. Harper DM, Williams KB. Prophylactic HPV vaccines: current knowledge of impact on gynecologic premalignancies [published online ahead of print July 3, 2010]. Discovery Medicine. 2010;10(50).http://www.discoverymedicine.com/Diane-M-Harper/2010/07/03/prophylactichpv-vaccines-current-knowledge-of-impact-ongynecologic-premalignancies. Accessed July 13, 2010.
13. Cervarix [human papillomavirus bivalent (types 16 and 18) vaccine recombinant]. Food and Drug Administration. Initial US approval: 2009:1–12.
14. Garland SM, Hernandez-Avila M, Wheeler CM, et al. Quadrivalent vaccine against human papillomavirus to prevent anogenital diseases. N Engl J Med. 2007;356(19):1928-1943.
15. World Health Organization. Human papillomavirus vaccines: WHO position paper. Weekly Epidemiological Record. 2009;84(15):118-131.http://www.who.int/wer/2009/wer8415/en/index.html. Published April 10, 2009. Accessed July 13, 2010.
16. Ault KA. For Future II Study Group. Effect of prophylactic human papillomavirus L1 virus-like-particle vaccine on the risk of cervical intraepithelial neoplasia grade 2, grade 3, and adenocarcinoma in situ: a combined analysis of four randomised clinical trials. Lancet. 2007;369(9576):1861-1868.
17. Arbyn M, Kyrgiou M, Simoens C, et al. Perinatal mortality and other severe adverse pregnancy outcomes associated with treatment of cervical intraepithelial neoplasia: meta-analysis. BMJ. 2008;337:a1284.-Doi: 10.1136/bmj.a1284.
18. Paavonen J. For the HPV PATRICIA Study Group. Efficacy of HPV 16/18 ASO4-adjuvanted vaccine against abnormal cytology, colposcopy referrals, and cervical procedures. Abstract SS 4–1. Data presented at Cervical Cancer Prevention, EuroGin 2010. European Research Organisation on Genital Infection and Neoplasia (EUROGIN); February 17-20, 2010; Monte Carlo, Monaco.http://www.eurogin.com/2010/programoverview.html. Accessed July 13, 2010.
19. Centers for Disease Control and Prevention. Reports of health concerns following HPV vaccination. 2010;1-3.http://www.cdc.gov/vaccinesafety/Vaccines/HPV/gardasil.html. Published June 21, 2010. Accessed July 13, 2010.
20. Myers ER. The economic impact of HPV vaccines: not just cervical cancer. Am J Obstet Gynecol. 2008;198(5):487-488.
21. Gardasil [human papillomavirus quadrivalent (types 6, 11, 16, and 18) vaccine, recombinant]. Food and Drug Administration; 2009. Initial Approval 2006;1-26.
22. Olsson SE, Kjaer SK, Sigurdsson K, et al. Evaluation of quadrivalent HPV 6/11/16/18 vaccine efficacy against cervical and anogenital disease in subjects with serological evidence of prior vaccine type HPV infection. Hum Vaccin. 2009;5(10).
23. Pirotta M, Ung L, Stein A, et al. The psychosocial burden of human papillomavirus related disease and screening interventions. Sex Transm Infect. 2009;85(7):508-513.
24. Kim JJ, Goldie SJ. Health and economic implications of HPV vaccination in the United States. N Engl J Med. 2008;359(8):821-832.
25. Cox JT. Is the HPV test effective as the primary screen for cervical cancer? Examining the Evidence. OBG Management. 2010;22(7):10-11.
Military Sexual Trauma Services Offered in the VHA
Vitamin K Supplementation in a Patient Taking Warfarin
Pathological Gambling in Combat Veterans
Fanning Away Dyspnea
Too Much Screening for Chlamydia?
Grand Rounds: Woman, 30, Survives Near-Exsanguination
While waiting to cross a street, a 30-year-old woman was suddenly struck by an oncoming vehicle, which crushed her legs against a parked automobile. She sustained a life-threatening traumatic injury and nearly exsanguinated at the scene. Nearby pedestrians assisted her, including a man who applied his belt to the woman’s left thigh to prevent complete exsanguination following the crush. She was emergently transported to an adult regional trauma center and admitted to the ICU.
The patient was given multiple transfusions of packed red blood cells, platelets, and frozen plasma in attempts to restore hemostasis. She underwent emergent surgery for a complete washout, debridement, and compartment fasciotomy on the right leg. The left leg required an above-knee amputation. Following surgery, full-thickness and split-thickness wounds were present on both extremities.
Before the accident, the woman had a history of hypertension controlled with a single antihypertensive. She was obese, with a BMI of 31.9. She had no surgical history. She denied excessive alcohol consumption, illicit drug use, or smoking. She was unaware of having any food or drug allergies.
The woman was married and had a 6-month-old baby. Until her accident, she was employed full-time as an investment accountant. She expressed contentment regarding her home, family, work, and busy lifestyle.
Once the patient’s condition was stabilized and hemostasis achieved in the trauma ICU, the bilateral lower-extremity wounds were managed by application of foam dressings via negative-pressure therapy. The dressings were changed on the patient’s lower-extremity wounds three times per week for about three weeks. When the wounds’ depth decreased and granulation was achieved, split-thickness skin grafts (STSGs) harvested from the right anterior thigh were applied to the open wounds (see Figure 1) in the operating room.
Following application of the STSGs and hemostasis of the patient’s donor site, silver silicone foam dressings were applied directly over the right lower-extremity graft and the donor site in the operating room. The dressings remained in place for four days (see Figure 2). A nonadherent, petrolatum-based contact layer was then applied to the left lower-extremity amputation graft site, followed by a negative-pressure foam dressing.
The negative-pressure pump was programmed for 75 mm Hg continuous therapy for four days. The silver silicone foam and negative-pressure foam dressings were removed from the respective graft sites on the fourth postpostoperative day. The grafts were viable and intact (see Figures 3 and 4). The silver silicone foam was reapplied to the lower-extremity STSGs and donor site and changed every four days.
When a few pinpoint dehisced areas were noted on the grafts, a silver-coated absorbent antimicrobial dressing was applied. A nonadherent, petrolatum-based contact layer, followed by wide-mesh stretch gauze, was secured as an exterior dressing over the graft sites. Both lower-extremity dressings were layered with elastic wraps to prevent edema. The dressings were changed daily for two weeks.
On postoperative Day 4, the silver silicone foam was removed from the donor site. A nonadherent contact layer of bismuth tribromophenate petrolatum, followed by the silver silicone foam, was selected for placement over the donor site. Gauze and an elastic wrap were secured as an exterior dressing and removed three days later.
The donor site dressing was reduced to a layer of bismuth tribromophenate petrolatum and left open to air. As the edges of the nonadherent contact layer dried, they were trimmed with scissors (see Figure 5). A moisturizing cocoa butter–based lotion was applied daily to the exposed areas of the donor site.
During the patient’s third postoperative week at the trauma center, as she underwent a continuum of aggressive rehabilitation and wound care, the donor and STSG sites were pronounced healed (see Figures 6, 7, and 8). The donor site was left open to air, with daily use of cocoa butter lotion. Maintenance care of the graft sites included daily application of cocoa butter lotion, stretch gauze, and elastic wraps. The patient was discharged from the rehabilitation unit to home, where she awaited a prosthetic fitting.
Throughout the patient’s hospitalization and rehabilitation, surgical, medical, pain, and nutrition management were monitored on a continuum, as were laboratory values. Her vital signs remained within reasonable limits. The patient remained infection free and experienced neither medical nor surgical complications during the course of her hospital stay.
DISCUSSION
Traumatic injuries often result in bodily deformities, amputations, and death. They represent the leading cause of death among people in the US younger than 45.1,2
Compartment syndrome develops when increased pressure within a bodily cavity minimizes capillary perfusion, resulting in decreased tissue viability.3 Edema and hemorrhage are also precipitating factors for this condition.3 When it goes unrelieved, compromised circulation can lead to muscle devitalization. Amputation of the affected appendage may be necessary unless circulation is restored.
Surgical fasciotomy can help alleviate pressure within the musculofascial compartment to improve circulation.4 Typically, such a procedure leaves large open wounds—a challenge for the clinician who cares for the affected patient. Once the wound is stabilized and the tissue becomes viable with granulation, application of an STSG can be considered.5,6
STSGs provide effective closure for open wounds. The grafting procedure entails removing, processing, and placing a portion of skin, both epidermal and partial dermal layers, on an open wound. Successful grafting requires adequate circulation, but excess bacteria can impede graft viability.7 Graft sites must be kept clean and moist without edema.5 Immediate application of negative-pressure therapy directly on an STSG has been shown to result in an outstanding graft “take,” as compared with use of traditional dressings.5,6,8
Nutritional status must be optimal for a successful graft take, with adequate intake of protein, calories, fluids, vitamins, and minerals.9,10
Treatment
Since days of old when traumatic wounds were treated with goat dung and honey, an array of methods and products has been developed, including numerous agents for cutaneous injuries alone.4 Occlusive, semiocclusive, or bacteriostatic topical ointments, foam, silver, or a combination of products can be used to manage and heal surgical wounds, grafts (including STSGs), and donor sites.11,12
Currently, negative pressure is also used in STSGs to accelerate healing.7,11 When applied to a wound, negative-pressure therapy enhances granulation, removes excess exudate, and creates a moist environment for healing.5,13
Silver-coated absorbent antimicrobial dressings appear to reduce bacteria on the surface of the wound surfaces and postoperative surgical sites without inducing bacterial resistance or adversely affecting healthy tissue.12,14-16 Silver silicone foam reduces bacterial colonization on wound surfaces and absorbs exudate into the foam dressing.17,18 Wounds with devitalized tissue and excessive drainage are at risk for infection, inflammation, and chronic duration.19
Nutrition
It has been reported that about half of all persons admitted to US hospitals are malnourished, with increased risk for morbidity and mortality.20 After experiencing hemorrhage, even well-nourished patients require additional protein and iron for successful recovery.10 Obese patients appear more susceptible to infection and surgical wound dehiscence than are their thinner counterparts, but further research is needed to study the impact of wound development and healing in this population.14
Tissue regeneration is known to require the amino acids arginine and glutamine for the construction of protein; additional research is also needed to support the theory that supplemental glutamine promotes wound healing.9,21 Surgical patients and patients with wounds benefit from protein-enhanced diets; zinc and vitamins A and C can also help improve wound healing and clinical outcomes.22 Monitoring protein and prealbumin levels is helpful in evaluating nutritional status, allowing the clinician to modify the medical nutrition plan and optimize the patient’s health and wellness.22
Rehabilitation
Surgical amputations of the lower extremities impair balance and mobility, necessitating extensive physical therapy and rehabilitation for affected patients.23,24 Aggressive rehabilitation typically is exhausting for patients. It is important to initiate a supportive team approach (including physical and occupational therapists) soon after surgery, continuing beyond the acute hospitalization into rehabilitation. Individualized, patient-centered goals are targeted and amended as necessary. Physical and occupational therapy increase in intensity and duration to optimize the patient’s functionality. Prosthetic fitting takes place after edema diminishes and the limb is fully healed.24
Patient Outcome
An obese young woman who sustained traumatic lower-extremity injuries and amputation experienced an optimal clinical outcome after 54 days of management. Exceptional surgical and medical strategies were initiated in the adult regional trauma center’s ICU and concluded at the adjoining rehabilitation center.
Strategic selection of products and interventions—negative pressure, silver silicone foam, silver-coated absorbent antimicrobial dressings, nonadherent contact layers, stretch gauze, and elastic wraps—and constant monitoring, including that of the patient’s nutritional status, resulted in expedient resolution of her traumatic wounds, STSGs, and donor site.
CONCLUSION
Despite revolutionary advances and life-sustaining measures in the surgical, medical, and wound care arena, traumatic events remain potentially debilitating and life-threatening for young adults. Triage of the trauma patient for appropriate medical care and collaborative management involving a team of trauma specialists and clinicians of all disciplines can now provide life-sustaining opportunities for these patients.
1. WISQARS™ (Web-based Injury Statistics Query and Reporting System). Leading Causes of Death Reports, 1999–2007. webappa.cdc.gov/sasweb/ncipc/leadcaus10.html. June 18, 2010.
2. Sasser SM, Hunt RC, Sullivent EE, et al; CDC. Guidelines for field triage of injured patients: recommendations of the National Expert Panel on Field Triage. MMWR Morb Mortal Wkly Rep. 2009;58(RR01): 1-35.
3. Feliciano DV. The management of extremity compartment syndrome. In: Cameron JL, ed. Current Surgical Therapy. 9th ed. Philadelphia, PA: Elsevier; 2008:1032-1036.
4. Kaufmann CR. Initial assessment and management. In: Feliciano DV, Mattox KL, Moore EE, eds. Trauma. 6th ed. New York: McGraw-Hill; 2008:169-184.
5. Mendez-Eastman S. Guidelines for using negative pressure wound therapy. Adv Skin Wound Care. 2001;14(6):314-322.
6. Snyder RJ, Doyle H, Delbridge T. Applying split-thickness skin grafts: a step-by-step clinical guide and nursing implications. Ostomy Wound Manage. 2001;47(11):20-26.
7. Sood R. Achauer and Sood’s Burn Surgery, Reconstruction and Rehabilitation. Philadelphia: WB Saunders. 2006.
8. Hanasono MM, Skoracki RJ. Securing skin grafts to microvascular free flaps using the vacuum-assisted closure (VAC) device. Ann Plast Surg. 2007;58(5):573-576.
9. Dorner B, Posthauer ME, Thomas D; National Pressure Ulcer Advisory Panel. The role of nutrition in pressure ulcer prevention and treatment (2009). www.npuap.org/Nutrition%20White%20Paper%20Website%20Version.pdf. Accessed June 18, 2010.
10. Frankenfield D. Energy expenditure and protein requirements after traumatic injury. Nutr Clin Pract. 2006;21(5):430-437.
11. Greenhalgh D. Topical antimicrobial agents for burn wounds. Clin Plast Surg. 2009;36(4):597-606.
12. Castellano JJ, Shafii SM, Ko F, et al. Comparative evaluation of silver-containing antimicrobial dressings and drugs. Int Wound J. 2007;4(2):114-122.
13. Baharestani MM. Negative pressure wound therapy in the adjunctive management of necrotizing fasciitis: examining clinical outcomes. Ostomy Wound Manage. 2008;54(4):44-50.
14. Childress BB, Berceli SA, Nelson PR, et al. Impact of an absorbent silver-eluting dressing system on lower extremity revascularization wound complications. Ann Vasc Surg. 2007;21(5):598-602.
15. Sibbald RG, Contreras-Ruiz J, Coutts P, et al. Bacteriology, inflammation, and healing: a study of nanocrystalline silver dressings in chronic venous leg ulcers. Adv Skin Wound Care. 2007;20(10):549-558.
16. Brett DW. A discussion of silver as an antimicrobial agent: alleviating the confusion. Ostomy Wound Manage. 2006;52(1):34-41.
17. Barrett S. Mepilex Ag: an antimicrobial, absorbent foam dressing with Safetac technology. Br J Nurs. 2009;18(20):S28, S30-S36.
18. Barrows C. Enhancing patient outcomes—reducing the bottom line: the use of antimicrobial soft silicone foam dressing in home health. Home Healthc Nurse. 2009;27(5):279-284.
19. National Pressure Ulcer Advisory Panel and European Pressure Ulcer Advisory Panel. Prevention and treatment of pressure ulcers: clinical practice guideline. Washington, DC: National Pressure Ulcer Advisory Panel; 2009.
20. Naber TH, Schermer T, de Bree A, et al. Prevalence of malnutrition in nonsurgical hospitalized patients and its association with disease complications. Am J Clin Nutr. 1997;66(5):1232-1239.
21. Ziegler TR, Benfell K, Smith RJ, et al. Safety and metabolic effects of L-glutamine administration in humans. JPEN J Parenter Enteral Nutr. 1990;14(4 suppl):137S-146S.
22. Skin conditions, pressure ulcers, and vitamin deficiencies. In: Escott-Stump S. Nutrition and Diagnosis–Related Care. 6th ed. Baltimore: Lippincott Williams & Wilkins. 2007:108-117.
23. van Velzen JM, van Bennekom CA, Polomski W, et al. Physical capacity and walking ability after lower limb amputation: a systematic review. Clin Rehabil. 2006;20(11):999-1016.
24. Ehlers CF. Integumentary disease and disorders/wound management. In: Malone DJ, Lindsay KLB, eds. Physical Therapy in Acute Care: A Clinician’s Guide. Thorofare, NJ: Slack Inc US; 2006:585-616.
While waiting to cross a street, a 30-year-old woman was suddenly struck by an oncoming vehicle, which crushed her legs against a parked automobile. She sustained a life-threatening traumatic injury and nearly exsanguinated at the scene. Nearby pedestrians assisted her, including a man who applied his belt to the woman’s left thigh to prevent complete exsanguination following the crush. She was emergently transported to an adult regional trauma center and admitted to the ICU.
The patient was given multiple transfusions of packed red blood cells, platelets, and frozen plasma in attempts to restore hemostasis. She underwent emergent surgery for a complete washout, debridement, and compartment fasciotomy on the right leg. The left leg required an above-knee amputation. Following surgery, full-thickness and split-thickness wounds were present on both extremities.
Before the accident, the woman had a history of hypertension controlled with a single antihypertensive. She was obese, with a BMI of 31.9. She had no surgical history. She denied excessive alcohol consumption, illicit drug use, or smoking. She was unaware of having any food or drug allergies.
The woman was married and had a 6-month-old baby. Until her accident, she was employed full-time as an investment accountant. She expressed contentment regarding her home, family, work, and busy lifestyle.
Once the patient’s condition was stabilized and hemostasis achieved in the trauma ICU, the bilateral lower-extremity wounds were managed by application of foam dressings via negative-pressure therapy. The dressings were changed on the patient’s lower-extremity wounds three times per week for about three weeks. When the wounds’ depth decreased and granulation was achieved, split-thickness skin grafts (STSGs) harvested from the right anterior thigh were applied to the open wounds (see Figure 1) in the operating room.
Following application of the STSGs and hemostasis of the patient’s donor site, silver silicone foam dressings were applied directly over the right lower-extremity graft and the donor site in the operating room. The dressings remained in place for four days (see Figure 2). A nonadherent, petrolatum-based contact layer was then applied to the left lower-extremity amputation graft site, followed by a negative-pressure foam dressing.
The negative-pressure pump was programmed for 75 mm Hg continuous therapy for four days. The silver silicone foam and negative-pressure foam dressings were removed from the respective graft sites on the fourth postpostoperative day. The grafts were viable and intact (see Figures 3 and 4). The silver silicone foam was reapplied to the lower-extremity STSGs and donor site and changed every four days.
When a few pinpoint dehisced areas were noted on the grafts, a silver-coated absorbent antimicrobial dressing was applied. A nonadherent, petrolatum-based contact layer, followed by wide-mesh stretch gauze, was secured as an exterior dressing over the graft sites. Both lower-extremity dressings were layered with elastic wraps to prevent edema. The dressings were changed daily for two weeks.
On postoperative Day 4, the silver silicone foam was removed from the donor site. A nonadherent contact layer of bismuth tribromophenate petrolatum, followed by the silver silicone foam, was selected for placement over the donor site. Gauze and an elastic wrap were secured as an exterior dressing and removed three days later.
The donor site dressing was reduced to a layer of bismuth tribromophenate petrolatum and left open to air. As the edges of the nonadherent contact layer dried, they were trimmed with scissors (see Figure 5). A moisturizing cocoa butter–based lotion was applied daily to the exposed areas of the donor site.
During the patient’s third postoperative week at the trauma center, as she underwent a continuum of aggressive rehabilitation and wound care, the donor and STSG sites were pronounced healed (see Figures 6, 7, and 8). The donor site was left open to air, with daily use of cocoa butter lotion. Maintenance care of the graft sites included daily application of cocoa butter lotion, stretch gauze, and elastic wraps. The patient was discharged from the rehabilitation unit to home, where she awaited a prosthetic fitting.
Throughout the patient’s hospitalization and rehabilitation, surgical, medical, pain, and nutrition management were monitored on a continuum, as were laboratory values. Her vital signs remained within reasonable limits. The patient remained infection free and experienced neither medical nor surgical complications during the course of her hospital stay.
DISCUSSION
Traumatic injuries often result in bodily deformities, amputations, and death. They represent the leading cause of death among people in the US younger than 45.1,2
Compartment syndrome develops when increased pressure within a bodily cavity minimizes capillary perfusion, resulting in decreased tissue viability.3 Edema and hemorrhage are also precipitating factors for this condition.3 When it goes unrelieved, compromised circulation can lead to muscle devitalization. Amputation of the affected appendage may be necessary unless circulation is restored.
Surgical fasciotomy can help alleviate pressure within the musculofascial compartment to improve circulation.4 Typically, such a procedure leaves large open wounds—a challenge for the clinician who cares for the affected patient. Once the wound is stabilized and the tissue becomes viable with granulation, application of an STSG can be considered.5,6
STSGs provide effective closure for open wounds. The grafting procedure entails removing, processing, and placing a portion of skin, both epidermal and partial dermal layers, on an open wound. Successful grafting requires adequate circulation, but excess bacteria can impede graft viability.7 Graft sites must be kept clean and moist without edema.5 Immediate application of negative-pressure therapy directly on an STSG has been shown to result in an outstanding graft “take,” as compared with use of traditional dressings.5,6,8
Nutritional status must be optimal for a successful graft take, with adequate intake of protein, calories, fluids, vitamins, and minerals.9,10
Treatment
Since days of old when traumatic wounds were treated with goat dung and honey, an array of methods and products has been developed, including numerous agents for cutaneous injuries alone.4 Occlusive, semiocclusive, or bacteriostatic topical ointments, foam, silver, or a combination of products can be used to manage and heal surgical wounds, grafts (including STSGs), and donor sites.11,12
Currently, negative pressure is also used in STSGs to accelerate healing.7,11 When applied to a wound, negative-pressure therapy enhances granulation, removes excess exudate, and creates a moist environment for healing.5,13
Silver-coated absorbent antimicrobial dressings appear to reduce bacteria on the surface of the wound surfaces and postoperative surgical sites without inducing bacterial resistance or adversely affecting healthy tissue.12,14-16 Silver silicone foam reduces bacterial colonization on wound surfaces and absorbs exudate into the foam dressing.17,18 Wounds with devitalized tissue and excessive drainage are at risk for infection, inflammation, and chronic duration.19
Nutrition
It has been reported that about half of all persons admitted to US hospitals are malnourished, with increased risk for morbidity and mortality.20 After experiencing hemorrhage, even well-nourished patients require additional protein and iron for successful recovery.10 Obese patients appear more susceptible to infection and surgical wound dehiscence than are their thinner counterparts, but further research is needed to study the impact of wound development and healing in this population.14
Tissue regeneration is known to require the amino acids arginine and glutamine for the construction of protein; additional research is also needed to support the theory that supplemental glutamine promotes wound healing.9,21 Surgical patients and patients with wounds benefit from protein-enhanced diets; zinc and vitamins A and C can also help improve wound healing and clinical outcomes.22 Monitoring protein and prealbumin levels is helpful in evaluating nutritional status, allowing the clinician to modify the medical nutrition plan and optimize the patient’s health and wellness.22
Rehabilitation
Surgical amputations of the lower extremities impair balance and mobility, necessitating extensive physical therapy and rehabilitation for affected patients.23,24 Aggressive rehabilitation typically is exhausting for patients. It is important to initiate a supportive team approach (including physical and occupational therapists) soon after surgery, continuing beyond the acute hospitalization into rehabilitation. Individualized, patient-centered goals are targeted and amended as necessary. Physical and occupational therapy increase in intensity and duration to optimize the patient’s functionality. Prosthetic fitting takes place after edema diminishes and the limb is fully healed.24
Patient Outcome
An obese young woman who sustained traumatic lower-extremity injuries and amputation experienced an optimal clinical outcome after 54 days of management. Exceptional surgical and medical strategies were initiated in the adult regional trauma center’s ICU and concluded at the adjoining rehabilitation center.
Strategic selection of products and interventions—negative pressure, silver silicone foam, silver-coated absorbent antimicrobial dressings, nonadherent contact layers, stretch gauze, and elastic wraps—and constant monitoring, including that of the patient’s nutritional status, resulted in expedient resolution of her traumatic wounds, STSGs, and donor site.
CONCLUSION
Despite revolutionary advances and life-sustaining measures in the surgical, medical, and wound care arena, traumatic events remain potentially debilitating and life-threatening for young adults. Triage of the trauma patient for appropriate medical care and collaborative management involving a team of trauma specialists and clinicians of all disciplines can now provide life-sustaining opportunities for these patients.
While waiting to cross a street, a 30-year-old woman was suddenly struck by an oncoming vehicle, which crushed her legs against a parked automobile. She sustained a life-threatening traumatic injury and nearly exsanguinated at the scene. Nearby pedestrians assisted her, including a man who applied his belt to the woman’s left thigh to prevent complete exsanguination following the crush. She was emergently transported to an adult regional trauma center and admitted to the ICU.
The patient was given multiple transfusions of packed red blood cells, platelets, and frozen plasma in attempts to restore hemostasis. She underwent emergent surgery for a complete washout, debridement, and compartment fasciotomy on the right leg. The left leg required an above-knee amputation. Following surgery, full-thickness and split-thickness wounds were present on both extremities.
Before the accident, the woman had a history of hypertension controlled with a single antihypertensive. She was obese, with a BMI of 31.9. She had no surgical history. She denied excessive alcohol consumption, illicit drug use, or smoking. She was unaware of having any food or drug allergies.
The woman was married and had a 6-month-old baby. Until her accident, she was employed full-time as an investment accountant. She expressed contentment regarding her home, family, work, and busy lifestyle.
Once the patient’s condition was stabilized and hemostasis achieved in the trauma ICU, the bilateral lower-extremity wounds were managed by application of foam dressings via negative-pressure therapy. The dressings were changed on the patient’s lower-extremity wounds three times per week for about three weeks. When the wounds’ depth decreased and granulation was achieved, split-thickness skin grafts (STSGs) harvested from the right anterior thigh were applied to the open wounds (see Figure 1) in the operating room.
Following application of the STSGs and hemostasis of the patient’s donor site, silver silicone foam dressings were applied directly over the right lower-extremity graft and the donor site in the operating room. The dressings remained in place for four days (see Figure 2). A nonadherent, petrolatum-based contact layer was then applied to the left lower-extremity amputation graft site, followed by a negative-pressure foam dressing.
The negative-pressure pump was programmed for 75 mm Hg continuous therapy for four days. The silver silicone foam and negative-pressure foam dressings were removed from the respective graft sites on the fourth postpostoperative day. The grafts were viable and intact (see Figures 3 and 4). The silver silicone foam was reapplied to the lower-extremity STSGs and donor site and changed every four days.
When a few pinpoint dehisced areas were noted on the grafts, a silver-coated absorbent antimicrobial dressing was applied. A nonadherent, petrolatum-based contact layer, followed by wide-mesh stretch gauze, was secured as an exterior dressing over the graft sites. Both lower-extremity dressings were layered with elastic wraps to prevent edema. The dressings were changed daily for two weeks.
On postoperative Day 4, the silver silicone foam was removed from the donor site. A nonadherent contact layer of bismuth tribromophenate petrolatum, followed by the silver silicone foam, was selected for placement over the donor site. Gauze and an elastic wrap were secured as an exterior dressing and removed three days later.
The donor site dressing was reduced to a layer of bismuth tribromophenate petrolatum and left open to air. As the edges of the nonadherent contact layer dried, they were trimmed with scissors (see Figure 5). A moisturizing cocoa butter–based lotion was applied daily to the exposed areas of the donor site.
During the patient’s third postoperative week at the trauma center, as she underwent a continuum of aggressive rehabilitation and wound care, the donor and STSG sites were pronounced healed (see Figures 6, 7, and 8). The donor site was left open to air, with daily use of cocoa butter lotion. Maintenance care of the graft sites included daily application of cocoa butter lotion, stretch gauze, and elastic wraps. The patient was discharged from the rehabilitation unit to home, where she awaited a prosthetic fitting.
Throughout the patient’s hospitalization and rehabilitation, surgical, medical, pain, and nutrition management were monitored on a continuum, as were laboratory values. Her vital signs remained within reasonable limits. The patient remained infection free and experienced neither medical nor surgical complications during the course of her hospital stay.
DISCUSSION
Traumatic injuries often result in bodily deformities, amputations, and death. They represent the leading cause of death among people in the US younger than 45.1,2
Compartment syndrome develops when increased pressure within a bodily cavity minimizes capillary perfusion, resulting in decreased tissue viability.3 Edema and hemorrhage are also precipitating factors for this condition.3 When it goes unrelieved, compromised circulation can lead to muscle devitalization. Amputation of the affected appendage may be necessary unless circulation is restored.
Surgical fasciotomy can help alleviate pressure within the musculofascial compartment to improve circulation.4 Typically, such a procedure leaves large open wounds—a challenge for the clinician who cares for the affected patient. Once the wound is stabilized and the tissue becomes viable with granulation, application of an STSG can be considered.5,6
STSGs provide effective closure for open wounds. The grafting procedure entails removing, processing, and placing a portion of skin, both epidermal and partial dermal layers, on an open wound. Successful grafting requires adequate circulation, but excess bacteria can impede graft viability.7 Graft sites must be kept clean and moist without edema.5 Immediate application of negative-pressure therapy directly on an STSG has been shown to result in an outstanding graft “take,” as compared with use of traditional dressings.5,6,8
Nutritional status must be optimal for a successful graft take, with adequate intake of protein, calories, fluids, vitamins, and minerals.9,10
Treatment
Since days of old when traumatic wounds were treated with goat dung and honey, an array of methods and products has been developed, including numerous agents for cutaneous injuries alone.4 Occlusive, semiocclusive, or bacteriostatic topical ointments, foam, silver, or a combination of products can be used to manage and heal surgical wounds, grafts (including STSGs), and donor sites.11,12
Currently, negative pressure is also used in STSGs to accelerate healing.7,11 When applied to a wound, negative-pressure therapy enhances granulation, removes excess exudate, and creates a moist environment for healing.5,13
Silver-coated absorbent antimicrobial dressings appear to reduce bacteria on the surface of the wound surfaces and postoperative surgical sites without inducing bacterial resistance or adversely affecting healthy tissue.12,14-16 Silver silicone foam reduces bacterial colonization on wound surfaces and absorbs exudate into the foam dressing.17,18 Wounds with devitalized tissue and excessive drainage are at risk for infection, inflammation, and chronic duration.19
Nutrition
It has been reported that about half of all persons admitted to US hospitals are malnourished, with increased risk for morbidity and mortality.20 After experiencing hemorrhage, even well-nourished patients require additional protein and iron for successful recovery.10 Obese patients appear more susceptible to infection and surgical wound dehiscence than are their thinner counterparts, but further research is needed to study the impact of wound development and healing in this population.14
Tissue regeneration is known to require the amino acids arginine and glutamine for the construction of protein; additional research is also needed to support the theory that supplemental glutamine promotes wound healing.9,21 Surgical patients and patients with wounds benefit from protein-enhanced diets; zinc and vitamins A and C can also help improve wound healing and clinical outcomes.22 Monitoring protein and prealbumin levels is helpful in evaluating nutritional status, allowing the clinician to modify the medical nutrition plan and optimize the patient’s health and wellness.22
Rehabilitation
Surgical amputations of the lower extremities impair balance and mobility, necessitating extensive physical therapy and rehabilitation for affected patients.23,24 Aggressive rehabilitation typically is exhausting for patients. It is important to initiate a supportive team approach (including physical and occupational therapists) soon after surgery, continuing beyond the acute hospitalization into rehabilitation. Individualized, patient-centered goals are targeted and amended as necessary. Physical and occupational therapy increase in intensity and duration to optimize the patient’s functionality. Prosthetic fitting takes place after edema diminishes and the limb is fully healed.24
Patient Outcome
An obese young woman who sustained traumatic lower-extremity injuries and amputation experienced an optimal clinical outcome after 54 days of management. Exceptional surgical and medical strategies were initiated in the adult regional trauma center’s ICU and concluded at the adjoining rehabilitation center.
Strategic selection of products and interventions—negative pressure, silver silicone foam, silver-coated absorbent antimicrobial dressings, nonadherent contact layers, stretch gauze, and elastic wraps—and constant monitoring, including that of the patient’s nutritional status, resulted in expedient resolution of her traumatic wounds, STSGs, and donor site.
CONCLUSION
Despite revolutionary advances and life-sustaining measures in the surgical, medical, and wound care arena, traumatic events remain potentially debilitating and life-threatening for young adults. Triage of the trauma patient for appropriate medical care and collaborative management involving a team of trauma specialists and clinicians of all disciplines can now provide life-sustaining opportunities for these patients.
1. WISQARS™ (Web-based Injury Statistics Query and Reporting System). Leading Causes of Death Reports, 1999–2007. webappa.cdc.gov/sasweb/ncipc/leadcaus10.html. June 18, 2010.
2. Sasser SM, Hunt RC, Sullivent EE, et al; CDC. Guidelines for field triage of injured patients: recommendations of the National Expert Panel on Field Triage. MMWR Morb Mortal Wkly Rep. 2009;58(RR01): 1-35.
3. Feliciano DV. The management of extremity compartment syndrome. In: Cameron JL, ed. Current Surgical Therapy. 9th ed. Philadelphia, PA: Elsevier; 2008:1032-1036.
4. Kaufmann CR. Initial assessment and management. In: Feliciano DV, Mattox KL, Moore EE, eds. Trauma. 6th ed. New York: McGraw-Hill; 2008:169-184.
5. Mendez-Eastman S. Guidelines for using negative pressure wound therapy. Adv Skin Wound Care. 2001;14(6):314-322.
6. Snyder RJ, Doyle H, Delbridge T. Applying split-thickness skin grafts: a step-by-step clinical guide and nursing implications. Ostomy Wound Manage. 2001;47(11):20-26.
7. Sood R. Achauer and Sood’s Burn Surgery, Reconstruction and Rehabilitation. Philadelphia: WB Saunders. 2006.
8. Hanasono MM, Skoracki RJ. Securing skin grafts to microvascular free flaps using the vacuum-assisted closure (VAC) device. Ann Plast Surg. 2007;58(5):573-576.
9. Dorner B, Posthauer ME, Thomas D; National Pressure Ulcer Advisory Panel. The role of nutrition in pressure ulcer prevention and treatment (2009). www.npuap.org/Nutrition%20White%20Paper%20Website%20Version.pdf. Accessed June 18, 2010.
10. Frankenfield D. Energy expenditure and protein requirements after traumatic injury. Nutr Clin Pract. 2006;21(5):430-437.
11. Greenhalgh D. Topical antimicrobial agents for burn wounds. Clin Plast Surg. 2009;36(4):597-606.
12. Castellano JJ, Shafii SM, Ko F, et al. Comparative evaluation of silver-containing antimicrobial dressings and drugs. Int Wound J. 2007;4(2):114-122.
13. Baharestani MM. Negative pressure wound therapy in the adjunctive management of necrotizing fasciitis: examining clinical outcomes. Ostomy Wound Manage. 2008;54(4):44-50.
14. Childress BB, Berceli SA, Nelson PR, et al. Impact of an absorbent silver-eluting dressing system on lower extremity revascularization wound complications. Ann Vasc Surg. 2007;21(5):598-602.
15. Sibbald RG, Contreras-Ruiz J, Coutts P, et al. Bacteriology, inflammation, and healing: a study of nanocrystalline silver dressings in chronic venous leg ulcers. Adv Skin Wound Care. 2007;20(10):549-558.
16. Brett DW. A discussion of silver as an antimicrobial agent: alleviating the confusion. Ostomy Wound Manage. 2006;52(1):34-41.
17. Barrett S. Mepilex Ag: an antimicrobial, absorbent foam dressing with Safetac technology. Br J Nurs. 2009;18(20):S28, S30-S36.
18. Barrows C. Enhancing patient outcomes—reducing the bottom line: the use of antimicrobial soft silicone foam dressing in home health. Home Healthc Nurse. 2009;27(5):279-284.
19. National Pressure Ulcer Advisory Panel and European Pressure Ulcer Advisory Panel. Prevention and treatment of pressure ulcers: clinical practice guideline. Washington, DC: National Pressure Ulcer Advisory Panel; 2009.
20. Naber TH, Schermer T, de Bree A, et al. Prevalence of malnutrition in nonsurgical hospitalized patients and its association with disease complications. Am J Clin Nutr. 1997;66(5):1232-1239.
21. Ziegler TR, Benfell K, Smith RJ, et al. Safety and metabolic effects of L-glutamine administration in humans. JPEN J Parenter Enteral Nutr. 1990;14(4 suppl):137S-146S.
22. Skin conditions, pressure ulcers, and vitamin deficiencies. In: Escott-Stump S. Nutrition and Diagnosis–Related Care. 6th ed. Baltimore: Lippincott Williams & Wilkins. 2007:108-117.
23. van Velzen JM, van Bennekom CA, Polomski W, et al. Physical capacity and walking ability after lower limb amputation: a systematic review. Clin Rehabil. 2006;20(11):999-1016.
24. Ehlers CF. Integumentary disease and disorders/wound management. In: Malone DJ, Lindsay KLB, eds. Physical Therapy in Acute Care: A Clinician’s Guide. Thorofare, NJ: Slack Inc US; 2006:585-616.
1. WISQARS™ (Web-based Injury Statistics Query and Reporting System). Leading Causes of Death Reports, 1999–2007. webappa.cdc.gov/sasweb/ncipc/leadcaus10.html. June 18, 2010.
2. Sasser SM, Hunt RC, Sullivent EE, et al; CDC. Guidelines for field triage of injured patients: recommendations of the National Expert Panel on Field Triage. MMWR Morb Mortal Wkly Rep. 2009;58(RR01): 1-35.
3. Feliciano DV. The management of extremity compartment syndrome. In: Cameron JL, ed. Current Surgical Therapy. 9th ed. Philadelphia, PA: Elsevier; 2008:1032-1036.
4. Kaufmann CR. Initial assessment and management. In: Feliciano DV, Mattox KL, Moore EE, eds. Trauma. 6th ed. New York: McGraw-Hill; 2008:169-184.
5. Mendez-Eastman S. Guidelines for using negative pressure wound therapy. Adv Skin Wound Care. 2001;14(6):314-322.
6. Snyder RJ, Doyle H, Delbridge T. Applying split-thickness skin grafts: a step-by-step clinical guide and nursing implications. Ostomy Wound Manage. 2001;47(11):20-26.
7. Sood R. Achauer and Sood’s Burn Surgery, Reconstruction and Rehabilitation. Philadelphia: WB Saunders. 2006.
8. Hanasono MM, Skoracki RJ. Securing skin grafts to microvascular free flaps using the vacuum-assisted closure (VAC) device. Ann Plast Surg. 2007;58(5):573-576.
9. Dorner B, Posthauer ME, Thomas D; National Pressure Ulcer Advisory Panel. The role of nutrition in pressure ulcer prevention and treatment (2009). www.npuap.org/Nutrition%20White%20Paper%20Website%20Version.pdf. Accessed June 18, 2010.
10. Frankenfield D. Energy expenditure and protein requirements after traumatic injury. Nutr Clin Pract. 2006;21(5):430-437.
11. Greenhalgh D. Topical antimicrobial agents for burn wounds. Clin Plast Surg. 2009;36(4):597-606.
12. Castellano JJ, Shafii SM, Ko F, et al. Comparative evaluation of silver-containing antimicrobial dressings and drugs. Int Wound J. 2007;4(2):114-122.
13. Baharestani MM. Negative pressure wound therapy in the adjunctive management of necrotizing fasciitis: examining clinical outcomes. Ostomy Wound Manage. 2008;54(4):44-50.
14. Childress BB, Berceli SA, Nelson PR, et al. Impact of an absorbent silver-eluting dressing system on lower extremity revascularization wound complications. Ann Vasc Surg. 2007;21(5):598-602.
15. Sibbald RG, Contreras-Ruiz J, Coutts P, et al. Bacteriology, inflammation, and healing: a study of nanocrystalline silver dressings in chronic venous leg ulcers. Adv Skin Wound Care. 2007;20(10):549-558.
16. Brett DW. A discussion of silver as an antimicrobial agent: alleviating the confusion. Ostomy Wound Manage. 2006;52(1):34-41.
17. Barrett S. Mepilex Ag: an antimicrobial, absorbent foam dressing with Safetac technology. Br J Nurs. 2009;18(20):S28, S30-S36.
18. Barrows C. Enhancing patient outcomes—reducing the bottom line: the use of antimicrobial soft silicone foam dressing in home health. Home Healthc Nurse. 2009;27(5):279-284.
19. National Pressure Ulcer Advisory Panel and European Pressure Ulcer Advisory Panel. Prevention and treatment of pressure ulcers: clinical practice guideline. Washington, DC: National Pressure Ulcer Advisory Panel; 2009.
20. Naber TH, Schermer T, de Bree A, et al. Prevalence of malnutrition in nonsurgical hospitalized patients and its association with disease complications. Am J Clin Nutr. 1997;66(5):1232-1239.
21. Ziegler TR, Benfell K, Smith RJ, et al. Safety and metabolic effects of L-glutamine administration in humans. JPEN J Parenter Enteral Nutr. 1990;14(4 suppl):137S-146S.
22. Skin conditions, pressure ulcers, and vitamin deficiencies. In: Escott-Stump S. Nutrition and Diagnosis–Related Care. 6th ed. Baltimore: Lippincott Williams & Wilkins. 2007:108-117.
23. van Velzen JM, van Bennekom CA, Polomski W, et al. Physical capacity and walking ability after lower limb amputation: a systematic review. Clin Rehabil. 2006;20(11):999-1016.
24. Ehlers CF. Integumentary disease and disorders/wound management. In: Malone DJ, Lindsay KLB, eds. Physical Therapy in Acute Care: A Clinician’s Guide. Thorofare, NJ: Slack Inc US; 2006:585-616.