User login
• Breast self-exam: Helpful or not? • HPV vaccine promises cost-effective benefits Ovarian cancer: • HRT increases risk • Consolidation therapy extends disease-free interval
Gynecologic oncology is a dynamic subspecialty—one with several important recent developments. For example, closer scrutiny of breast self-examination has changed its status from “required” to optional, and greater understanding of cervical carcinoma and its causes is leading to a vaccine for human papillomavirus (HPV) 16/18 in the near future—an achievement likely to be highly cost-effective.
On the ovarian cancer front, data from the Women’s Health Initiative suggest an increase in cancer rates in women who take hormone replacement therapy (HRT). In fact, ovarian cancer remains our greatest challenge, since 70% of patients are not diagnosed until they reach advanced stages III and IV, when 5-year survival ranges from 5% to 40%. Fortunately, recent findings suggest consolidation therapy may help extend survival and disease-free intervals.
This update focuses on important studies in each of these areas, highlighting significant progress in understanding, preventing, identifying, and treating gynecologic malignancies.
Breast self-examination: Helpful or not?
Hackshaw AK, Paul EA. Breast self-examination and death from breast cancer: a meta-analysis. Br J Cancer. 2003; 88:1047–1053.
With 1 in 6 women likely to develop breast cancer, the drive to identify patients with early disease continues apace, especially since survival frequently depends upon it. While mammography remains the gold standard for breast cancer screening, the role of breast self-examination (BSE) is controversial. In May 2003, the American Cancer Society revised its guidelines, changing BSE from a routine to an optional practice. The meta-analysis by Hackshaw and Paul evaluated the effect of BSE on the death rate from breast cancer, reviewing 20 observational and 3 clinical trials on BSE and death or, alternatively, BSE and advanced breast cancer (a surrogate for poor prognosis and increased death rate). They evaluated BSE in women who:
- Practice BSE routinely
- Found tumors during BSE
- Are trained in BSE
However, these results are likely caused by bias and confounding. For example, many patients who practiced BSE were younger and of higher socioeconomic status than women who did not. A confounding variable in the observational studies was the presence of slow-growing tumors.
BSE did not lower death rate. No study found a lower death rate in women who detected their breast cancer during BSE (RR, 0.9; 95% CI, 0.72–1.12). Nor did the death rate diminish among women who were trained to perform BSE (RR, 1.01; 95% CI 0.92–1.12), although BSE did appear to prompt many women to seek medical advice. Unfortunately, many studies evaluating the effects of BSE also include mammography, which makes it difficult to isolate effects due solely to BSE.
Clinical implications. Although this meta-analysis supports the American Cancer Society recommendation not to require breast self-examination as a cancer screening tool, I have managed a number of patients who presented with breast masses identified through BSE. Many of these women were low-risk and younger than 30 years. For that reason, I continue to instruct and encourage patients to perform BSE, since it is easily taught and carried out.
I believe BSE helps empower women to take control of their health.
Related Reading
- American Cancer Society News Today. Role of breast self-examination changes in guidelines. Available at: www.cancer.org/docroot/NWS/NWS_1.asp Accessed June 16, 2004.
- Vahabi M. Breast cancer screening methods: a review of the evidence. Health Care Women Intern. 2003;24:773–793.
Adding HPV vaccine to cervical cancer screening would be cost-effective
Goldie SJ, Kohli M, Grima D, et al. Projected clinical benefits and cost-effectiveness of a human papillomavirus 16/18 virus. J Natl Cancer Inst. 2004;96:604–615.
Because of the link between cervical cancer or dysplasia and HPV infection, HPV vaccination in childhood or adolescence would virtually eradicate cervical cancer. It also would be cost-effective, compared with the cost of screening, evaluating, and treating dysplasia and cervical cancer.
In 2002, Koutsky et al1 reported a randomized, prospective trial of an HPV-16 vaccine, in which they found vaccination to be 100% effective (95% confidence interval [CI], 90–100; P.001 with an incidence of persistent hpv-16 infection per woman-years in vaccinated women versus among controls. at the time its publication this trial was largest ever conducted hpv vaccine enrolled placebo group and>
Following the Koutsky trial, Goldie and colleagues reported on a computer-based model to evaluate the efficacy and cost of vaccinating patients with an HPV 16/18 vaccine. Using the Markov model, they simulated HPV infection and carcinogenesis. However, rather than evaluate the cost-effectiveness of the vaccine as a single modality, they added it to current cytologic testing, evaluating screening intervals of 1 to 5 years with conventional and liquid-based smears.
The most cost-effective screening strategy was vaccination at age 12 and cytologic screening every 3 years beginning at age 25. Using this approach, the lifetime risk of cervical cancer would be reduced by 94% compared with no screening—a spectacular improvement.
HPV vaccines will become available in the next several years, at which time we will need to determine their appropriate use in industrialized nations, such as the United States, where about 3,900 cervical cancer deaths will occur this year, as well as in developing countries. Worldwide, roughly 500,000 women die of the disease each year. In developing countries, the vaccine would decrease the incidence of dysplasia by more than 50%, even if used as a single modality.
Clinical implications. Although most dysplasias and carcinomas are related to HPV 16/18, it is imperative that we continue to screen to rule out infection with other high-risk subtypes. Eventually, a polyvalent vaccine may enable us to vaccinate for all known high-risk subtypes.
REFERENCE
1. Koutsky LA, Ault KA, Wheeler CM, et al. for the Proof of Principle Study Investigators. A controlled trial of a human papillomavirus type 16 vaccine. N Engl J Med. 2002;347:1645-1651.
HRT increases risk of ovarian cancer
Anderson GL, Judd HL, Kaunitz AM, et al. Effects of estrogen plus progestin on gynecologic cancers and associated diagnostic procedures: the Women’s Health Initiative randomized trial. JAMA. 2003;290:1739–1748.
The Women’s Health Initiative has produced the single largest randomized, prospective trial comparing estrogen, continuous estrogen-progesterone, and placebo. Recent findings from this population include an increased risk of breast cancer, heart disease, dementia, and vascular thrombosis with HRT use.
Now Anderson and colleagues have reported on the association between gynecologic cancers and HRT—specifically, the estrogen-progestin combination. In the randomized, double-blind study involving 16,608 postmenopausal women, participants were given 0.625 mg of conjugated equine estrogens and 2.5 mg of medroxyprogesterone acetate (n = 8,506) or placebo (n = 8,102), and the main outcome measure was invasive cancer of the ovary or endometrium. After an average follow-up of 5.6 years, Anderson et al found 32 cases of invasive ovarian cancer, 58 cases of endometrial cancer, 1 case of non-endometrial uterine cancer, 13 cases of cervical cancer, and 7 cases of other gynecologic cancers.
Compared with controls, women taking HRT experienced a significantly increased incidence of ovarian cancer, with a hazard ratio of 1.58 (95% CI, 0.77–3.24). For endometrial cancer, the hazard ratio was 0.81 (95% CI, 0.48–1.36). The groups did not differ significantly in regard to the other cancers.
More endometrial biopsies. Another important finding from this study is the greater need for endometrial biopsies among women taking HRT (33% versus 6%; P.001>
Other trials also have reported an increased risk of ovarian cancer with HRT use, as well as the decreased risk of endometrial cancer.
Because of the greater risk associated with HRT, indications for it have changed. Now most HRT users are young and take the therapy to relieve vasomotor symptoms. These women should be counseled about the risks outlined in the Women’s Health Initiative, as well as the importance of endometrial biopsies to evaluate any abnormal bleeding.
Clinical implications. In the next 5 years, alternative therapies such as selective estrogen receptor modulators are likely to replace HRT. Until then, I will continue to prescribe HRT, but only in symptomatic women for a period of less than 5 years.
Related Reading
- Hempling RE, Wong C, Piver MS, et al. Hormone replacement therapy as a risk factor for epithelial ovarian cancer: results of a case-control study. Obstet Gynecol. 1997;89:1012–1016.
- Kaufman DW, Kelly JP, Welch WR, et al. Noncontraceptive estrogen use and epithelial ovarian cancer. Am J Epidemiol. 1989;130:1142–1151.
- Purdie DM, Bain CJ, Siskind V, et al. Hormone replacement therapy and risk of epithelial ovarian cancer. Br J Cancer. 1999;81:559–563.
- Risch HA. Estrogen replacement therapy and risk of epithelial ovarian cancer. Gynecol Oncol. 1996;63:254–257.
- Rodriguez C, Calle EE, Coates RJ, et al. Estrogen replacement therapy and fatal ovarian cancer. Am J Epidemiol. 1995;141:828–835.
- Rodriguez C, Patel AV, Calle EE, et al. Estrogen replacement therapy and ovarian cancer mortality in a large prospective study of US women. JAMA. 2001;285:1460–1465.
Consolidation therapy extends disease-free interval
Markman M, Liu PY, Wilczynski S, et al. Phase III randomized trial of 12 versus 3 months of maintenance paclitaxel in patients with advanced ovarian cancer after complete response to platinum and paclitaxel-based chemotherapy. Southwest Oncology Group and Gynecologic Oncology Group Trial. J Clin Oncol. 2003;21:2460–2465.
Standard therapy for ovarian cancer consists of 6 courses of a platinum (cisplatin or carboplatin) and taxane (paclitaxel or docitaxel) regimen. Following such therapy, second-look laparotomy or laparoscopy once was widely performed. However, that strategy has not been shown to increase survival: 50% of second looks for suspected ovarian cancer are pathologically positive, while an additional 25% are pathologically positive within 3 years.
For this reason, other ways of extending the disease-free interval and improving survival are under investigation. The study by Markman and colleagues focuses on consolidation therapy, which is treatment administered after a complete pathologic or clinical response. Whole abdominal radiation, intraperitoneal radioactive phosphorus (32P), and chemotherapy have been evaluated.
In this study, the Gynecologic Oncology Group and Southwest Oncology Group compared 3 versus 12 months of maintenance paclitaxel in patients who had a complete response to platinum/paclitaxel chemotherapy. Two arms were established: one giving paclitaxel 175 mg/m2 over 3 hours every 28 days for 3 cycles, and another extending this regimen to 12 cycles.
The median progression-free survival times were 21 and 28 months in the 3- and 12-month arms, respectively. P values for the adjusted Cox model analysis and unadjusted log-rank test were .0023 and .0035, respectively, with the 12-month arm having superior results. The Cox model-adjusted 3-cycle versus 12-cycle progression hazard ratio was estimated to be 2.31 (99% CI, 1.08–4.94). Because the protocol recommended early termination of the trial at a cutoff P value of .005, the trial was discontinued and all women were given the opportunity to receive 12 courses of paclitaxel.
Although these results are statistically significant, the 5-year survival and disease-free intervals are not available. Therefore, the role of consolidation chemotherapy with agents such as paclitaxel—which is not without side effects—needs further investigation before it can become the standard of care.
Clinical implications. I present the option of consolidation chemotherapy to all patients, encouraging them to participate in clinical trials of the therapy. If a trial is not available, I give the patient the option of receiving 12 cycles of paclitaxel off protocol or continued observation. I also discuss current data, including pros and cons, with the patient prior to initiating consolidation therapy.
Related Reading
- Sorbe B, Swedish-Norwegian Ovarian Cancer Study Group. Consolidation treatment of advanced (FIGO stage III) ovarian carcinoma in complete surgical remission after induction chemotherapy: a randomized, controlled, clinical trial comparing whole abdominal radiotherapy, chemotherapy, and no further treatment. Int J Gynecol Cancer. 2003;13:278-286.
- Varia MA, Stehman FB, Bundy BN, et al. Intraperitoneal radioactive phosphorus (P32) versus observation after negative second-look laparotomy for stage III ovarian carcinoma: a randomized trial of the Gynecologic Oncology Group. J Clin Oncol. 2003;21:2849–2855.
Gynecologic oncology is a dynamic subspecialty—one with several important recent developments. For example, closer scrutiny of breast self-examination has changed its status from “required” to optional, and greater understanding of cervical carcinoma and its causes is leading to a vaccine for human papillomavirus (HPV) 16/18 in the near future—an achievement likely to be highly cost-effective.
On the ovarian cancer front, data from the Women’s Health Initiative suggest an increase in cancer rates in women who take hormone replacement therapy (HRT). In fact, ovarian cancer remains our greatest challenge, since 70% of patients are not diagnosed until they reach advanced stages III and IV, when 5-year survival ranges from 5% to 40%. Fortunately, recent findings suggest consolidation therapy may help extend survival and disease-free intervals.
This update focuses on important studies in each of these areas, highlighting significant progress in understanding, preventing, identifying, and treating gynecologic malignancies.
Breast self-examination: Helpful or not?
Hackshaw AK, Paul EA. Breast self-examination and death from breast cancer: a meta-analysis. Br J Cancer. 2003; 88:1047–1053.
With 1 in 6 women likely to develop breast cancer, the drive to identify patients with early disease continues apace, especially since survival frequently depends upon it. While mammography remains the gold standard for breast cancer screening, the role of breast self-examination (BSE) is controversial. In May 2003, the American Cancer Society revised its guidelines, changing BSE from a routine to an optional practice. The meta-analysis by Hackshaw and Paul evaluated the effect of BSE on the death rate from breast cancer, reviewing 20 observational and 3 clinical trials on BSE and death or, alternatively, BSE and advanced breast cancer (a surrogate for poor prognosis and increased death rate). They evaluated BSE in women who:
- Practice BSE routinely
- Found tumors during BSE
- Are trained in BSE
However, these results are likely caused by bias and confounding. For example, many patients who practiced BSE were younger and of higher socioeconomic status than women who did not. A confounding variable in the observational studies was the presence of slow-growing tumors.
BSE did not lower death rate. No study found a lower death rate in women who detected their breast cancer during BSE (RR, 0.9; 95% CI, 0.72–1.12). Nor did the death rate diminish among women who were trained to perform BSE (RR, 1.01; 95% CI 0.92–1.12), although BSE did appear to prompt many women to seek medical advice. Unfortunately, many studies evaluating the effects of BSE also include mammography, which makes it difficult to isolate effects due solely to BSE.
Clinical implications. Although this meta-analysis supports the American Cancer Society recommendation not to require breast self-examination as a cancer screening tool, I have managed a number of patients who presented with breast masses identified through BSE. Many of these women were low-risk and younger than 30 years. For that reason, I continue to instruct and encourage patients to perform BSE, since it is easily taught and carried out.
I believe BSE helps empower women to take control of their health.
Related Reading
- American Cancer Society News Today. Role of breast self-examination changes in guidelines. Available at: www.cancer.org/docroot/NWS/NWS_1.asp Accessed June 16, 2004.
- Vahabi M. Breast cancer screening methods: a review of the evidence. Health Care Women Intern. 2003;24:773–793.
Adding HPV vaccine to cervical cancer screening would be cost-effective
Goldie SJ, Kohli M, Grima D, et al. Projected clinical benefits and cost-effectiveness of a human papillomavirus 16/18 virus. J Natl Cancer Inst. 2004;96:604–615.
Because of the link between cervical cancer or dysplasia and HPV infection, HPV vaccination in childhood or adolescence would virtually eradicate cervical cancer. It also would be cost-effective, compared with the cost of screening, evaluating, and treating dysplasia and cervical cancer.
In 2002, Koutsky et al1 reported a randomized, prospective trial of an HPV-16 vaccine, in which they found vaccination to be 100% effective (95% confidence interval [CI], 90–100; P.001 with an incidence of persistent hpv-16 infection per woman-years in vaccinated women versus among controls. at the time its publication this trial was largest ever conducted hpv vaccine enrolled placebo group and>
Following the Koutsky trial, Goldie and colleagues reported on a computer-based model to evaluate the efficacy and cost of vaccinating patients with an HPV 16/18 vaccine. Using the Markov model, they simulated HPV infection and carcinogenesis. However, rather than evaluate the cost-effectiveness of the vaccine as a single modality, they added it to current cytologic testing, evaluating screening intervals of 1 to 5 years with conventional and liquid-based smears.
The most cost-effective screening strategy was vaccination at age 12 and cytologic screening every 3 years beginning at age 25. Using this approach, the lifetime risk of cervical cancer would be reduced by 94% compared with no screening—a spectacular improvement.
HPV vaccines will become available in the next several years, at which time we will need to determine their appropriate use in industrialized nations, such as the United States, where about 3,900 cervical cancer deaths will occur this year, as well as in developing countries. Worldwide, roughly 500,000 women die of the disease each year. In developing countries, the vaccine would decrease the incidence of dysplasia by more than 50%, even if used as a single modality.
Clinical implications. Although most dysplasias and carcinomas are related to HPV 16/18, it is imperative that we continue to screen to rule out infection with other high-risk subtypes. Eventually, a polyvalent vaccine may enable us to vaccinate for all known high-risk subtypes.
REFERENCE
1. Koutsky LA, Ault KA, Wheeler CM, et al. for the Proof of Principle Study Investigators. A controlled trial of a human papillomavirus type 16 vaccine. N Engl J Med. 2002;347:1645-1651.
HRT increases risk of ovarian cancer
Anderson GL, Judd HL, Kaunitz AM, et al. Effects of estrogen plus progestin on gynecologic cancers and associated diagnostic procedures: the Women’s Health Initiative randomized trial. JAMA. 2003;290:1739–1748.
The Women’s Health Initiative has produced the single largest randomized, prospective trial comparing estrogen, continuous estrogen-progesterone, and placebo. Recent findings from this population include an increased risk of breast cancer, heart disease, dementia, and vascular thrombosis with HRT use.
Now Anderson and colleagues have reported on the association between gynecologic cancers and HRT—specifically, the estrogen-progestin combination. In the randomized, double-blind study involving 16,608 postmenopausal women, participants were given 0.625 mg of conjugated equine estrogens and 2.5 mg of medroxyprogesterone acetate (n = 8,506) or placebo (n = 8,102), and the main outcome measure was invasive cancer of the ovary or endometrium. After an average follow-up of 5.6 years, Anderson et al found 32 cases of invasive ovarian cancer, 58 cases of endometrial cancer, 1 case of non-endometrial uterine cancer, 13 cases of cervical cancer, and 7 cases of other gynecologic cancers.
Compared with controls, women taking HRT experienced a significantly increased incidence of ovarian cancer, with a hazard ratio of 1.58 (95% CI, 0.77–3.24). For endometrial cancer, the hazard ratio was 0.81 (95% CI, 0.48–1.36). The groups did not differ significantly in regard to the other cancers.
More endometrial biopsies. Another important finding from this study is the greater need for endometrial biopsies among women taking HRT (33% versus 6%; P.001>
Other trials also have reported an increased risk of ovarian cancer with HRT use, as well as the decreased risk of endometrial cancer.
Because of the greater risk associated with HRT, indications for it have changed. Now most HRT users are young and take the therapy to relieve vasomotor symptoms. These women should be counseled about the risks outlined in the Women’s Health Initiative, as well as the importance of endometrial biopsies to evaluate any abnormal bleeding.
Clinical implications. In the next 5 years, alternative therapies such as selective estrogen receptor modulators are likely to replace HRT. Until then, I will continue to prescribe HRT, but only in symptomatic women for a period of less than 5 years.
Related Reading
- Hempling RE, Wong C, Piver MS, et al. Hormone replacement therapy as a risk factor for epithelial ovarian cancer: results of a case-control study. Obstet Gynecol. 1997;89:1012–1016.
- Kaufman DW, Kelly JP, Welch WR, et al. Noncontraceptive estrogen use and epithelial ovarian cancer. Am J Epidemiol. 1989;130:1142–1151.
- Purdie DM, Bain CJ, Siskind V, et al. Hormone replacement therapy and risk of epithelial ovarian cancer. Br J Cancer. 1999;81:559–563.
- Risch HA. Estrogen replacement therapy and risk of epithelial ovarian cancer. Gynecol Oncol. 1996;63:254–257.
- Rodriguez C, Calle EE, Coates RJ, et al. Estrogen replacement therapy and fatal ovarian cancer. Am J Epidemiol. 1995;141:828–835.
- Rodriguez C, Patel AV, Calle EE, et al. Estrogen replacement therapy and ovarian cancer mortality in a large prospective study of US women. JAMA. 2001;285:1460–1465.
Consolidation therapy extends disease-free interval
Markman M, Liu PY, Wilczynski S, et al. Phase III randomized trial of 12 versus 3 months of maintenance paclitaxel in patients with advanced ovarian cancer after complete response to platinum and paclitaxel-based chemotherapy. Southwest Oncology Group and Gynecologic Oncology Group Trial. J Clin Oncol. 2003;21:2460–2465.
Standard therapy for ovarian cancer consists of 6 courses of a platinum (cisplatin or carboplatin) and taxane (paclitaxel or docitaxel) regimen. Following such therapy, second-look laparotomy or laparoscopy once was widely performed. However, that strategy has not been shown to increase survival: 50% of second looks for suspected ovarian cancer are pathologically positive, while an additional 25% are pathologically positive within 3 years.
For this reason, other ways of extending the disease-free interval and improving survival are under investigation. The study by Markman and colleagues focuses on consolidation therapy, which is treatment administered after a complete pathologic or clinical response. Whole abdominal radiation, intraperitoneal radioactive phosphorus (32P), and chemotherapy have been evaluated.
In this study, the Gynecologic Oncology Group and Southwest Oncology Group compared 3 versus 12 months of maintenance paclitaxel in patients who had a complete response to platinum/paclitaxel chemotherapy. Two arms were established: one giving paclitaxel 175 mg/m2 over 3 hours every 28 days for 3 cycles, and another extending this regimen to 12 cycles.
The median progression-free survival times were 21 and 28 months in the 3- and 12-month arms, respectively. P values for the adjusted Cox model analysis and unadjusted log-rank test were .0023 and .0035, respectively, with the 12-month arm having superior results. The Cox model-adjusted 3-cycle versus 12-cycle progression hazard ratio was estimated to be 2.31 (99% CI, 1.08–4.94). Because the protocol recommended early termination of the trial at a cutoff P value of .005, the trial was discontinued and all women were given the opportunity to receive 12 courses of paclitaxel.
Although these results are statistically significant, the 5-year survival and disease-free intervals are not available. Therefore, the role of consolidation chemotherapy with agents such as paclitaxel—which is not without side effects—needs further investigation before it can become the standard of care.
Clinical implications. I present the option of consolidation chemotherapy to all patients, encouraging them to participate in clinical trials of the therapy. If a trial is not available, I give the patient the option of receiving 12 cycles of paclitaxel off protocol or continued observation. I also discuss current data, including pros and cons, with the patient prior to initiating consolidation therapy.
Related Reading
- Sorbe B, Swedish-Norwegian Ovarian Cancer Study Group. Consolidation treatment of advanced (FIGO stage III) ovarian carcinoma in complete surgical remission after induction chemotherapy: a randomized, controlled, clinical trial comparing whole abdominal radiotherapy, chemotherapy, and no further treatment. Int J Gynecol Cancer. 2003;13:278-286.
- Varia MA, Stehman FB, Bundy BN, et al. Intraperitoneal radioactive phosphorus (P32) versus observation after negative second-look laparotomy for stage III ovarian carcinoma: a randomized trial of the Gynecologic Oncology Group. J Clin Oncol. 2003;21:2849–2855.
Gynecologic oncology is a dynamic subspecialty—one with several important recent developments. For example, closer scrutiny of breast self-examination has changed its status from “required” to optional, and greater understanding of cervical carcinoma and its causes is leading to a vaccine for human papillomavirus (HPV) 16/18 in the near future—an achievement likely to be highly cost-effective.
On the ovarian cancer front, data from the Women’s Health Initiative suggest an increase in cancer rates in women who take hormone replacement therapy (HRT). In fact, ovarian cancer remains our greatest challenge, since 70% of patients are not diagnosed until they reach advanced stages III and IV, when 5-year survival ranges from 5% to 40%. Fortunately, recent findings suggest consolidation therapy may help extend survival and disease-free intervals.
This update focuses on important studies in each of these areas, highlighting significant progress in understanding, preventing, identifying, and treating gynecologic malignancies.
Breast self-examination: Helpful or not?
Hackshaw AK, Paul EA. Breast self-examination and death from breast cancer: a meta-analysis. Br J Cancer. 2003; 88:1047–1053.
With 1 in 6 women likely to develop breast cancer, the drive to identify patients with early disease continues apace, especially since survival frequently depends upon it. While mammography remains the gold standard for breast cancer screening, the role of breast self-examination (BSE) is controversial. In May 2003, the American Cancer Society revised its guidelines, changing BSE from a routine to an optional practice. The meta-analysis by Hackshaw and Paul evaluated the effect of BSE on the death rate from breast cancer, reviewing 20 observational and 3 clinical trials on BSE and death or, alternatively, BSE and advanced breast cancer (a surrogate for poor prognosis and increased death rate). They evaluated BSE in women who:
- Practice BSE routinely
- Found tumors during BSE
- Are trained in BSE
However, these results are likely caused by bias and confounding. For example, many patients who practiced BSE were younger and of higher socioeconomic status than women who did not. A confounding variable in the observational studies was the presence of slow-growing tumors.
BSE did not lower death rate. No study found a lower death rate in women who detected their breast cancer during BSE (RR, 0.9; 95% CI, 0.72–1.12). Nor did the death rate diminish among women who were trained to perform BSE (RR, 1.01; 95% CI 0.92–1.12), although BSE did appear to prompt many women to seek medical advice. Unfortunately, many studies evaluating the effects of BSE also include mammography, which makes it difficult to isolate effects due solely to BSE.
Clinical implications. Although this meta-analysis supports the American Cancer Society recommendation not to require breast self-examination as a cancer screening tool, I have managed a number of patients who presented with breast masses identified through BSE. Many of these women were low-risk and younger than 30 years. For that reason, I continue to instruct and encourage patients to perform BSE, since it is easily taught and carried out.
I believe BSE helps empower women to take control of their health.
Related Reading
- American Cancer Society News Today. Role of breast self-examination changes in guidelines. Available at: www.cancer.org/docroot/NWS/NWS_1.asp Accessed June 16, 2004.
- Vahabi M. Breast cancer screening methods: a review of the evidence. Health Care Women Intern. 2003;24:773–793.
Adding HPV vaccine to cervical cancer screening would be cost-effective
Goldie SJ, Kohli M, Grima D, et al. Projected clinical benefits and cost-effectiveness of a human papillomavirus 16/18 virus. J Natl Cancer Inst. 2004;96:604–615.
Because of the link between cervical cancer or dysplasia and HPV infection, HPV vaccination in childhood or adolescence would virtually eradicate cervical cancer. It also would be cost-effective, compared with the cost of screening, evaluating, and treating dysplasia and cervical cancer.
In 2002, Koutsky et al1 reported a randomized, prospective trial of an HPV-16 vaccine, in which they found vaccination to be 100% effective (95% confidence interval [CI], 90–100; P.001 with an incidence of persistent hpv-16 infection per woman-years in vaccinated women versus among controls. at the time its publication this trial was largest ever conducted hpv vaccine enrolled placebo group and>
Following the Koutsky trial, Goldie and colleagues reported on a computer-based model to evaluate the efficacy and cost of vaccinating patients with an HPV 16/18 vaccine. Using the Markov model, they simulated HPV infection and carcinogenesis. However, rather than evaluate the cost-effectiveness of the vaccine as a single modality, they added it to current cytologic testing, evaluating screening intervals of 1 to 5 years with conventional and liquid-based smears.
The most cost-effective screening strategy was vaccination at age 12 and cytologic screening every 3 years beginning at age 25. Using this approach, the lifetime risk of cervical cancer would be reduced by 94% compared with no screening—a spectacular improvement.
HPV vaccines will become available in the next several years, at which time we will need to determine their appropriate use in industrialized nations, such as the United States, where about 3,900 cervical cancer deaths will occur this year, as well as in developing countries. Worldwide, roughly 500,000 women die of the disease each year. In developing countries, the vaccine would decrease the incidence of dysplasia by more than 50%, even if used as a single modality.
Clinical implications. Although most dysplasias and carcinomas are related to HPV 16/18, it is imperative that we continue to screen to rule out infection with other high-risk subtypes. Eventually, a polyvalent vaccine may enable us to vaccinate for all known high-risk subtypes.
REFERENCE
1. Koutsky LA, Ault KA, Wheeler CM, et al. for the Proof of Principle Study Investigators. A controlled trial of a human papillomavirus type 16 vaccine. N Engl J Med. 2002;347:1645-1651.
HRT increases risk of ovarian cancer
Anderson GL, Judd HL, Kaunitz AM, et al. Effects of estrogen plus progestin on gynecologic cancers and associated diagnostic procedures: the Women’s Health Initiative randomized trial. JAMA. 2003;290:1739–1748.
The Women’s Health Initiative has produced the single largest randomized, prospective trial comparing estrogen, continuous estrogen-progesterone, and placebo. Recent findings from this population include an increased risk of breast cancer, heart disease, dementia, and vascular thrombosis with HRT use.
Now Anderson and colleagues have reported on the association between gynecologic cancers and HRT—specifically, the estrogen-progestin combination. In the randomized, double-blind study involving 16,608 postmenopausal women, participants were given 0.625 mg of conjugated equine estrogens and 2.5 mg of medroxyprogesterone acetate (n = 8,506) or placebo (n = 8,102), and the main outcome measure was invasive cancer of the ovary or endometrium. After an average follow-up of 5.6 years, Anderson et al found 32 cases of invasive ovarian cancer, 58 cases of endometrial cancer, 1 case of non-endometrial uterine cancer, 13 cases of cervical cancer, and 7 cases of other gynecologic cancers.
Compared with controls, women taking HRT experienced a significantly increased incidence of ovarian cancer, with a hazard ratio of 1.58 (95% CI, 0.77–3.24). For endometrial cancer, the hazard ratio was 0.81 (95% CI, 0.48–1.36). The groups did not differ significantly in regard to the other cancers.
More endometrial biopsies. Another important finding from this study is the greater need for endometrial biopsies among women taking HRT (33% versus 6%; P.001>
Other trials also have reported an increased risk of ovarian cancer with HRT use, as well as the decreased risk of endometrial cancer.
Because of the greater risk associated with HRT, indications for it have changed. Now most HRT users are young and take the therapy to relieve vasomotor symptoms. These women should be counseled about the risks outlined in the Women’s Health Initiative, as well as the importance of endometrial biopsies to evaluate any abnormal bleeding.
Clinical implications. In the next 5 years, alternative therapies such as selective estrogen receptor modulators are likely to replace HRT. Until then, I will continue to prescribe HRT, but only in symptomatic women for a period of less than 5 years.
Related Reading
- Hempling RE, Wong C, Piver MS, et al. Hormone replacement therapy as a risk factor for epithelial ovarian cancer: results of a case-control study. Obstet Gynecol. 1997;89:1012–1016.
- Kaufman DW, Kelly JP, Welch WR, et al. Noncontraceptive estrogen use and epithelial ovarian cancer. Am J Epidemiol. 1989;130:1142–1151.
- Purdie DM, Bain CJ, Siskind V, et al. Hormone replacement therapy and risk of epithelial ovarian cancer. Br J Cancer. 1999;81:559–563.
- Risch HA. Estrogen replacement therapy and risk of epithelial ovarian cancer. Gynecol Oncol. 1996;63:254–257.
- Rodriguez C, Calle EE, Coates RJ, et al. Estrogen replacement therapy and fatal ovarian cancer. Am J Epidemiol. 1995;141:828–835.
- Rodriguez C, Patel AV, Calle EE, et al. Estrogen replacement therapy and ovarian cancer mortality in a large prospective study of US women. JAMA. 2001;285:1460–1465.
Consolidation therapy extends disease-free interval
Markman M, Liu PY, Wilczynski S, et al. Phase III randomized trial of 12 versus 3 months of maintenance paclitaxel in patients with advanced ovarian cancer after complete response to platinum and paclitaxel-based chemotherapy. Southwest Oncology Group and Gynecologic Oncology Group Trial. J Clin Oncol. 2003;21:2460–2465.
Standard therapy for ovarian cancer consists of 6 courses of a platinum (cisplatin or carboplatin) and taxane (paclitaxel or docitaxel) regimen. Following such therapy, second-look laparotomy or laparoscopy once was widely performed. However, that strategy has not been shown to increase survival: 50% of second looks for suspected ovarian cancer are pathologically positive, while an additional 25% are pathologically positive within 3 years.
For this reason, other ways of extending the disease-free interval and improving survival are under investigation. The study by Markman and colleagues focuses on consolidation therapy, which is treatment administered after a complete pathologic or clinical response. Whole abdominal radiation, intraperitoneal radioactive phosphorus (32P), and chemotherapy have been evaluated.
In this study, the Gynecologic Oncology Group and Southwest Oncology Group compared 3 versus 12 months of maintenance paclitaxel in patients who had a complete response to platinum/paclitaxel chemotherapy. Two arms were established: one giving paclitaxel 175 mg/m2 over 3 hours every 28 days for 3 cycles, and another extending this regimen to 12 cycles.
The median progression-free survival times were 21 and 28 months in the 3- and 12-month arms, respectively. P values for the adjusted Cox model analysis and unadjusted log-rank test were .0023 and .0035, respectively, with the 12-month arm having superior results. The Cox model-adjusted 3-cycle versus 12-cycle progression hazard ratio was estimated to be 2.31 (99% CI, 1.08–4.94). Because the protocol recommended early termination of the trial at a cutoff P value of .005, the trial was discontinued and all women were given the opportunity to receive 12 courses of paclitaxel.
Although these results are statistically significant, the 5-year survival and disease-free intervals are not available. Therefore, the role of consolidation chemotherapy with agents such as paclitaxel—which is not without side effects—needs further investigation before it can become the standard of care.
Clinical implications. I present the option of consolidation chemotherapy to all patients, encouraging them to participate in clinical trials of the therapy. If a trial is not available, I give the patient the option of receiving 12 cycles of paclitaxel off protocol or continued observation. I also discuss current data, including pros and cons, with the patient prior to initiating consolidation therapy.
Related Reading
- Sorbe B, Swedish-Norwegian Ovarian Cancer Study Group. Consolidation treatment of advanced (FIGO stage III) ovarian carcinoma in complete surgical remission after induction chemotherapy: a randomized, controlled, clinical trial comparing whole abdominal radiotherapy, chemotherapy, and no further treatment. Int J Gynecol Cancer. 2003;13:278-286.
- Varia MA, Stehman FB, Bundy BN, et al. Intraperitoneal radioactive phosphorus (P32) versus observation after negative second-look laparotomy for stage III ovarian carcinoma: a randomized trial of the Gynecologic Oncology Group. J Clin Oncol. 2003;21:2849–2855.
Thromboembolic disease: The case for routine prophylaxis
- Prophylaxis must start before surgery for maximal benefit, since at least 50% of postoperative thromboembolic disease begins intraoperatively.
- A consensus conference found that prophylaxis reduced fatal pulmonary emboli by 75% in 7,000 gynecologic surgery patients.
- Low-dose unfractionated heparin and low molecular weight heparin appear similarly effective in reducing thromboembolic disease in perioperative patients, but it is unclear which form has fewer bleeding complications.
The case is strong for routine prophylaxis against venous thrombosis and pulmonary embolism. The primary reasons: efficacy, ease of use, and safety.
This article reviews the evidence on routine prophylaxis, pros and cons of mechanical and drug therapies (including a comparison of 2 heparins), patient risk factors, and cost-effectiveness. A table (page 32) lists patient characteristics for low, moderate, high, and very high levels of risk, with corresponding appropriate preventive measures.
Routine prophylaxis is a wiser strategy than postevent treatment or surveillance because:
- Thromboembolism is a stealthy adversary, particularly pulmonary embolism. Between 10% and 20% of patients with pulmonary emboli die, often within 30 minutes of the sentinel complaint.
- Even when pulmonary embolus is documented during autopsy, as many as 80% of patients have no antecedent clinical evidence of deep venous thrombosis (DVT)—and no chance of life-saving therapy.2
- Surveillance is the least desirable preventive option due to lack of sensitivity of noninvasive tests for thromboembolic disease.
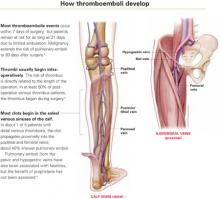
Scope of thromboembolic disease
Thromboembolism is the leading potentially preventable cause of hospital fatality.1
In postoperative gynecologic surgery patients, pulmonary embolism occurs in 0.1% to 5% of cases, depending on risk.2 Venous thrombosis occurs on average in 15% of postoperative gynecologic surgery patients, with a range of 5% to 29%, depending on patient risk factors and the surgical procedures performed.3 Almost 50% of gynecologic patients undergoing surgery for cancer develop a lower-extremity venous thrombosis if left untreated.4
Overall, thromboembolic disease is linked with some 500,000 hospital admissions every year. Pulmonary embolism, the most serious complication, causes 60,000 to 200,000 deaths annually.
High risk of recurrence. Postthrombotic syndrome is characterized by pain, swelling, and leg ulceration. At least 50% of patients successfully treated for proximal venous thrombosis (thigh) and about 33% treated for distal venous thrombosis (calf) develop postthrombotic syndrome.1 The risk for recurrent thrombosis is high.
Risk factors are listed in TABLE 1. The risk of fatal pulmonary embolism is directly related to age; persons over 60 are at greatest risk. When the patient is obese—particularly when she exceeds 120% of ideal body weight—the risk is even greater due to venous stasis.
TABLE 1
Risk factors for thromboembolism
| Age |
| Estrogen use |
| Extended pelvic surgery |
| Hypercoagulable states |
| Immobility |
| Indwelling central venous catheter(s) |
| Lower-extremity paralysis |
| Malignancy |
| Medical illnesses |
| Chronic pulmonary disease |
| Congestive heart failure |
| Diabetes mellitus |
| Inflammatory bowel disease |
| Nephrotic syndrome |
| Obesity |
| Pregnancy |
| Prior thromboembolic disease |
| Radiation therapy |
| Trauma |
| Varicose veins |
Common hypercoagulable states
Activated protein C resistance, the most common thrombophilia, occurs in 3% to 7% of Caucasians (TABLE 2).7 It is usually associated with factor V Leiden mutation.
Prothrombin gene mutation G20210A, the next most common thrombophilia, occurs in 2% of Caucasians.7
Antiphospholipid antibody syndromes are acquired thrombophilias that may be associated with arterial and venous thrombosis, thrombocytopenia, and complications of pregnancy.8
Hyperhomocystinemia may be congenital or acquired. It is associated with venous thromboembolism and early atherosclerosis with arterial thrombosis.
Thrombosis is more likely with multiple predisposing factors. Most patients with one of the thrombophilia syndromes do not have sentinel thrombotic events unless they are further challenged by environmental risks such as oral contraceptives (OCs), pregnancy, prolonged immobilization, or surgery.9
Risk is markedly enhanced if multiple predisposing factors are present. For example, when factor V Leiden mutation and hyperhomocystinemia coexist, the risk for thrombosis increases 10 to 20 times over that of patients with neither abnormality.10 This combined risk far exceeds the sum of risks for each abnormality alone.
TABLE 2
Common hypercoagulable conditions
| Factor V Leiden mutation |
| Prothrombin gene mutation G20210A |
| Antiphospholipid antibody |
| Lupus anticoagulant |
| Anticardiolipin |
| Protein C deficiency |
| Protein S deficiency |
| Antithrombin III deficiency (Heparin cofactor II) |
| Plasminogen deficiency |
| Hyperhomocystinemia |
| Myeloproliferative disorders |
| Polycythemia |
| Primary thrombocytosis |
When to test for thrombophilias
Although women with thrombophilias are at high risk for thromboembolic disease, it is unclear whether identifying the thrombophilia is more beneficial than universal prophylaxis. If a patient has a personal or family history of thromboembolic disease—especially a patient with Caucasian ancestry—testing is probably warranted. While documentation of the thrombophilia may not help with perioperative management, it may be useful for long-term care.
Testing for factor V Leiden mutation is recommended due to its prevalence. If the results are negative in a patient at risk, test for prothrombin gene mutation G20210A, as well as deficiencies in the naturally occurring inhibitors protein C, protein S, and antithrombin III.
Assess antiphospholipid antibodies in women who have experienced recurrent fetal loss or early pregnancy-induced hypertension.
Continue OCs or hormone therapy
OCs and menopausal hormone replacement therapy produce measurable prothrombotic changes in the clotting system that appear to be directly related to the estrogen content. In theory, discontinuing the OC or hormone replacement therapy preoperatively would allow these changes to return to baseline and help prevent thromboembolic disease.
Although the risk of thromboembolic disease is 0.96% if patients are current users of OCs and 0.5% if they are not, studies have failed to confirm a clinical benefit of discontinuation.11 Further, the patient does not return to baseline for 4 to 6 weeks after ceasing therapy.
The potential risk of thromboembolic events also should be weighed against the risk of conception prior to surgery. We usually do not recommend discontinuation of OCs and hormone therapy before surgery, but give prophylaxis based on risk assessment.
Venous stasis, vessel-wall trauma, and increased blood coagulability—the major contributors to perioperative DVT, known as Virchow’s triad—were identified more than 125 years ago.
Venous stasis. Intraoperatively, venous blood return from the lower extremities is reduced to less than half its normal rate,34 secondary to muscle relaxation during anesthesia, which causes venous dilation and reduced blood-flow velocity. Packing the abdominal contents may further impede blood return from the legs.
Resultant venous stasis causes platelet adhesion to the vein wall, followed by release of a thromboplastin-like substance that may trigger thrombus formation.
Blood flow increases in the immediate postoperative period with return of muscle tone, but remains significantly diminished for 21 days because of immobilization—specifically, lack of the usual pumping action of the leg muscles.
Vessel-wall trauma. Veins are highly likely to be damaged as vessels are skeletonized during major pelvic surgery, especially when malignancy is involved. Tissue injury activates the coagulation cascade by exposing blood to tissue thromboplastin (extrinsic path) and subendothelial collagen in the vessel wall, which activates factor XII (intrinsic path). Both pathways lead to conversion of factor X to its active form, factor Xa. Acting in concert with factor V, calcium, and phospholipids from platelet factor III, factor Xa catalyzes the conversion of prothrombin to thrombin. Thrombin regulates the conversion of fibrinogen to fibrin, the basic building block of a thrombus.
Increased blood coagulability. Clotting factors XI, IX, and VII increase following surgery, as do circulating platelets and platelet aggregation. This enhances coagulability, which persists from 72 to 96 hours after surgery but is usually balanced by the fibrinolytic system. Fibrinolysis is mediated primarily by plasmin, which digests fibrin and fibrinogen and activates factors V and VIII. If the fibrinolytic system is overwhelmed, the clotting system is unimpeded and thrombus formation may accelerate.
Pregnancy increases the risk of thrombosis, in part due to the progressive increase in resistance to activated protein C in the second and third trimesters. Risk is increased eightfold in women with inherited deficiency in any of the naturally occurring anticoagulants—antithrombin III, protein C, or protein S—compared with those with no deficiency.12
Preventive strategies
Two approaches to thromboembolic prophylaxis have been proposed, with prevention of fatal postoperative pulmonary emboli as a clear endpoint:
- Stratify a targeted group into levels of risk; then treat those at higher risk. Unfortunately, efforts to define risk have met with only partial success, due to limited availability of noninvasive screening, screening logistics, and expense. Further, specificity and positive predictive value of screening asymptomatic patients is low.
- Use prophylaxis in all patients in the targeted group, regardless of risk (TABLES 3 AND 4). This strategy seems effective. For example, in 2001, the Sixth American College of Chest Physicians Consensus Conference—the most recent of the consensus conferences—evaluated the risks of pulmonary embolus in 7,000 gynecologic surgery patients enrolled in prospective studies. Routine prophylaxis reduced fatal pulmonary emboli by 75%.13
The difficulty of defining patients at highest risk makes the concept of universal prophylaxis for a targeted group an attractive option unless a specific contraindication is identified.
TABLE 3
Risk stratification and prophylactic regimens
| LEVEL OF RISK | PATIENT CHARACTERISTICS | RECOMMENDED REGIMEN |
|---|---|---|
| Low | Less than 40 years of age | No specific recommendation for therapy |
| Undergoing uncomplicated minor surgery | Adequate hydration | |
| Requires less than 30 minutes of anesthesia | Aggressive early ambulation | |
| No additional risk factors | ||
| Moderate | Undergoing minor surgery with additional risk factors | Graduated compression stockings or SPCDs or |
| 40 to 60 years of age, undergoing minor surgery, and no additional risk factors | LDUH 5,000 U every 12 hours or | |
| Less than 40 years of age, undergoing major surgery, and no additional risk factors | LMWH (20 mg) or 2,500 U antifactor Xa once daily | |
| High | More than 60 years of age, undergoing minor surgery or with additional risk factors | LDUH 5,000 U every 8–12 hours or LMWH (40 mg) or 5,000 U antifactor Xa once daily |
| More than 40 years of age undergoing major surgery or with additional risk factors | SPCDs may be added | |
| Very high | More than 60 years of age, undergoing major surgery, with other risk factors such as cancer, prior venous thromboembolism, molecular hypercoagulable state, major trauma, spinal cord injury, or lower-extremity paralysis | Graduated compression stockings or SPCDs and LDUH 5,000 U every 8 hours or |
| Other risk factors such as cancer, prior venous thromboembolism, molecular hypercoagulable state, major trauma, spinal cord injury, or lower-extremity paralysis | LMWH (40 mg) or 5,000 U antifactor Xa once daily | |
| LDUH = low-dose unfractionated heparin; LMWH = low molecular weight heparin; SPCD = sequential pneumatic compression device | ||
| Note: Because perioperative prophylaxis is still evolving, these suggestions should not be considered inviolable. | ||
| Source: Modified from Geerts WH, et al.13 | ||
TABLE 4
Incidence of thromboembolic events based on level of risk
| SITE OF THROMBOSIS | RISK LEVEL (%) | |||
|---|---|---|---|---|
| Low | Moderate | High | Very high | |
| Calf veins (distal) | 2 | 10–35 | 30–60 | 63.5 |
| Iliofemoral veins (proximal) | 0.4 | 2–8 | 5–10 | 10–20 |
| Pulmonary emboli | 0.2 | 1–2 | 2–4 | 4–10 |
| Fatal pulmonary emboli | 0.002 | 0.1–0.5 | 0.4–1 | 0.2–5 |
| Source: Modified from Geerts WH, et al.13 | ||||
Drug and mechanical therapies
Strategies for preventing thromboembolic disease attempt to mitigate the impact of venous stasis, endothelial injury, and hypercoagulable states. Traditionally, early ambulation, adequate hydration, and elevation of the lower extremities have been used, because they are simple and inexpensive interventions.
More recently, pharmacologic and mechanical therapies have proven effective in reducing the incidence of DVT and fatal pulmonary emboli. Prophylactic measures commonly used today are:
- graduated compression stockings
- external pneumatic leg or foot compression devices
- low-dose unfractionated heparin
- low molecular weight heparin
MECHANICAL THERAPIESGraduated compression stockings
Compression stockings are one of the earliest methods of preventing perioperative thrombosis. Compression is greatest at the toe and gradually diminishes toward the thigh. When Belcaro14 studied the risk of recurrent venous thrombosis in nonsurgical hospitalized patients, thrombosis recurred in 40% of patients with no therapy, but only 9.4% of patients wearing graduated compression stockings (GCS). Adding oral antiplatelet therapy lowered the risk to 2%. However, GCS were not superior to any other method of preventing recurrent DVT.
As for surgical patients, a single study15 demonstrated protection against DVT when compared with no GCS in elective gynecologic surgery patients.
Today, GCS are usually a perioperative adjunct to other preventive methods, to provide added protection.
Proper fitting by trained personnel is vital; otherwise, a tourniquet effect may cause venous stasis and reduce benefit.
Sequential pneumatic compression devices
Like GCS, these devices decrease the caliber of veins by simple compression. They also increase blood flow velocity and stimulate the endogenous fibrinolytic system.
Enhanced fibrinolytic activity due to intermittent compression occurs even if the device is used on only 1 lower extremity, or on an upper extremity. Patient and nursing-staff compliance may affect efficacy. Sequential pneumatic compression devices (SPCDs) may be thought inconvenient, impeding nursing functions. Some patients may find the repetitive inflation-deflation cycles annoying.
These devices must be activated prior to surgery and continued for a minimum of 24 hours. Some studies suggest that SPCDs be used for 5 days in high-risk situations.16
Calf- and thigh-length devices have similar effects. Rare complications include peroneal nerve injury and compartment syndrome.
What the data show. In an analysis of 4 trials comparing SPCDs with no therapy, thromboembolic disease occurred in only 2% of patients who wore SPCDs but in 20% of those who did not.17 Compared with low-dose unfractionated heparin, no difference in the rate of DVT was seen.18 However, risk of transfusion and retroperitoneal drainage volume increased in the heparin group.
Contraindications include active or suspected DVT, congestive heart failure, known pulmonary embolus, and leg injuries.19
Combined therapy may be best for high-risk women. A study20 of women who developed thromboemboli despite appropriate treatment with SPCDs found the women more likely to be older than 60 years and/or to have cancer or history of thromboembolic disease or hypertension. This high-risk group may benefit from combined therapy.
Foot compression devices resemble booties and mimic the plantar compression that occurs during walking. They increase blood-flow velocity, stimulate the endogenous fibrinolytic system, and have the same indications as SPCDs. However, they are only moderately effective in reducing venous thrombosis.21 Potential drawbacks are that the devices must be removed when the patient ambulates and replaced when she returns to bed.
DRUG THERAPIESLow-dose unfractionated heparin
Heparin alters the molecular configuration of antithrombin III, making it 1,000 to 4,000 times more potent as an inhibitor of thrombin formation, which in turn limits conversion of fibrinogen to fibrin. This prolongs the activated partial thromboplastin time (aPTT) commonly used to monitor patients receiving full anticoagulation therapy.
A naturally occurring mucopolysaccharide with a molecular weight ranging from 3,000 to 30,000 daltons, heparin is richly concentrated in mast cells. Its anticoagulant properties relate primarily to interaction with antithrombin III (also known as heparin cofactor II), factor IXa, factor Xa, factor XIa, factor XIIa, and platelet aggregation.22
Heparin also inhibits the effects of factor Xa on the coagulation cascade and limits platelet aggregation.23
Half-life is 1 hour for intravenous heparin and about 3 hours for subcutaneous heparin.
Use in pregnancy. Low-dose unfractionated heparin (LDUH) does not cross the placenta and is safe to use during pregnancy.
What the data show. Randomized clinical trials conducted prior to 1988 showed venous thrombosis decreased by 70% and pulmonary embolus by 50% in patients treated with LDUH, compared with those receiving no therapy.24
Dosing options. LDUH typically is given as a 5,000-U dose 2 hours before surgery. The single preoperative dose seems to be as effective as multiple preoperative doses.25 Postoperative therapy is instituted 8 to 12 hours after surgery; heparin is given every 8 to 12 hours until the patient is fully ambulatory.
Patients having gynecologic surgery for benign conditions benefit from the twice-daily regimen, and those undergoing gynecologic oncology surgery or other high-risk procedures seem to benefit from thrice-daily dosing.26
These regimens also significantly reduced DVT and fatal pulmonary emboli in general surgery patients.27 The LDUH regimen is now used to judge the efficacy of other prophylactic measures.
Low molecular weight heparin
This form of heparin acts primarily by inhibiting factor Xa, which is higher in the coagulation cascade than antithrombin. Thus, low molecular weight heparin (LMWH) is more efficient than unfractionated heparin.
LMWH has a molecular weight of 3,000 to 6,000 daltons and is produced by concentrating the low molecular component of heparin. Because the molecular configuration of antithrombin III is not altered by LMWH, thrombin conversion is minimally inhibited and aPTT is not appreciably affected.
Half-life is approximately 4 hours, by any route of administration. The longer half-life provides a longer dosing interval. Bioavailability is more consistent than that of LDUH, approaching 90% to 95%. The excellent bioavailability allows dosing to be based on lean body mass. Less heparin-induced thrombocytopenia is another plus.
Use in pregnancy. LMWH does not cross the placenta and is safe to use during pregnancy.
What the data show. No randomized trials have compared LMWH therapy with no therapy, with efficacy assessed by venography or fibrinogen uptake. An uncontrolled series28 of 2,030 patients did show that LMWH reduced the incidence of thromboembolic disease.
A randomized controlled trial29 comparing 2,500 U and 5,000 U daily of dalteparin (an LMWH) found greater efficacy in the higher-dose group (6.6% versus 12.7% incidence of DVT), but bleeding complications also were higher (4.7% versus 2.7%). In a subgroup of patients with malignancy, the high-dose therapy remained superior in preventing DVT, and the bleeding risk was equal.
Dosing options. Once a day dosing is normally adequate for prophylaxis; twice-daily dosing is needed for therapy. Enoxaparin is an LMWH that is readily available in the United States and commonly prescribed when use of LMWH is desired. For prophylaxis, it can be given in daily doses of 20 mg, 30 mg, 40 mg, or 60 mg. None of these doses has proven superior to the others. The typical regimen for moderate risk is 20 mg per day; for high risk, 40 mg per day. Enoxaparin appears to convey the same degree of protection as 5,000 units of LDUH every 8 hours.
2 heparins compared
Randomized controlled trials comparing LDUH and LMWH have found them to be similarly effective. In addition, within the LMWH class, all compounds appear to have similar benefits. However, it remains unclear which form of heparin is associated with fewer bleeding complications.
In theory, LMWH would be associated with an increase in these complications due to its longer half-life and increased bioavailability.30 However, studies have not consistently identified excess bleeding in any group.31 Fortunately, excess postoperative bleeding requiring transfusion is uncommon.
Complications of heparin
Heparin-induced thrombocytopenia is a recognized complication, seen in as many as 20% of LDUH patients. The diagnosis is made when the platelet count falls below the lower limits of normal or when the platelet count falls by 50% but remains in the normal range. Two forms of heparin-induced thrombocytopenia have been described.
• Type 1 thrombocytopenia is initially mild, rarely dropping below 100,000 platelets per milliliter. Platelet count monitoring is important, but therapy usually can continue, since the platelet count returns to normal even with continued use. Type 1 thrombocytopenia appears to directly result from platelet activation and is not immune-mediated.
• Type 2 thrombocytopenia occurs 7 to 14 days after starting therapy. Platelet counts frequently drop to 20,000 per milliliter. Type 2 is an immune response caused by antibodies to the heparin-platelet factor 4 complex. Patients are at risk for venous and arterial thrombosis, but often the diagnosis is made only after a complicating thrombotic event.
The risk of type 2 thrombocytopenia appears to be related to the molecular weight of the compound being administered, its dose, and the duration of therapy. Therefore, unfractionated heparin—if given in full anticoagulation doses and beyond 14 days—seems to have the greatest potential for this complication.
Paradoxically, these patients also are at risk for severe hemorrhage. Because mortality and morbidity are high, immediate withdrawal of all forms of heparin—including LMWH—is mandatory.32 Catastrophic hemorrhage or thrombosis may be the first sign. Often a fall in platelet concentration precedes serious complications. Thus, platelet concentration should be monitored at least every 2 days.
Hematoma with conduction anesthesia. Use of major conduction anesthesia in patients who also need LMWH, LDUH, or oral anticoagulants is controversial. LMWH may pose a risk of hematoma if initiated preoperatively, intraoperatively, or within 3 hours of surgery in patients who have a continuous epidural. Hematoma formation seems to occur immediately following catheter withdrawal.
The risk of hematoma appears to be lower with single-dose spinal or single-dose epidural anesthesia. While the incidence of epidural or spinal hematoma is not known, some cases have been associated with long-term neurologic sequelae, including permanent paralysis.
Responding to this concern, the US Food and Drug Administration issued a 1997 advisory noting the risk of spinal hematoma in patients receiving enoxaparin plus conduction anesthesia or lumbar spinal puncture. Most anesthesiologists significantly restrict the use of conduction anesthesia in patients requiring heparin prophylaxis.33
Cost-effectiveness
The cost of perioperative prophylaxis has been compared with the cost of immediate therapy for thromboembolic disease and long-term therapy for postthrombotic syndrome. Across the board, universal prophylaxis with pharmacotherapy or mechanical devices in patients undergoing abdominal surgery is less expensive than no prophylaxis, based on a reduced incidence of DVT. Pneumatic compression seems to be more cost-effective than pharmacotherapy.34
Because LDUH is substantially less expensive than LMWH in the United States, it has a better cost profile when pharmacotherapy is warranted. In Europe, where the cost of heparin compounds is not an issue, LMWH has a slight advantage.
The authors report no financial relationships relevant to this article.
1. Dismuke SE, Wagner EH. Pulmonary embolism as a cause of death. The changing mortality in hospitalized patients. JAMA. 1986;255:2039-2042.
2. Farquharson DI, Orr JW, Jr. Prophylaxis against thromboembolism in gynecologic patients. J Reprod Med. 1984;29:845-862.
3. Bergqvist D. Prolonged prophylaxis against postoperative venous thromboembolism. Haemostasis. 1996;26(suppl 4):379-387.
4. Clarke-Pearson DL, DeLong ER, Synan IS, Coleman RE, Creasman WT. Variables associated with postoperative deep venous thrombosis: a prospective study of 411 gynecology patients and creation of a prognostic model. Obstet Gynecol. 1987;69:146-150.
5. Kemble JVH. Incidence of deep vein thrombosis. Br J Hosp Med. 1971;6:721-726.
6. Spritzer CE, Evans AC, Kay HH. Magnetic resonance imaging of deep venous thrombosis in pregnant women with lower extremity edema. Obstet Gynecol. 1995;85:603-607.
7. Florell SR, Rodgers GM. Inherited thrombotic disorders: an update. Am J Hematol. 1997;54:53-60.
8. Shapiro GA. Antiphospholipid syndrome in obstetrics and gynecology. Semin Thromb Hemost. 1994;20:64-70.
9. De Stafano V, Leone G, Mastrangelo S, et al. Clinical manifestations and management of inherited thrombophilia: retrospective analysis and follow-up after diagnosis in 238 patients with congenital deficiency of antithrombin III, protein C, protein S. S Throm Haemost. 1994;72:352-358.
10. Ridker PM, Hennekens CH, Selhub J, et al. Interrelation of hyperhomocyst(e)inemia, factor V Leiden, and risk of future venous thromboembolism. Circulation. 1997;95:1777-1782.
11. Vessey M, Jewell D, Smith A, Yeates D, McPherson K. Chronic inflammatory bowel disease, cigarette smoking, and use of oral contraceptives: findings in a large cohort study of women of childbearing age. Br Med J (Clin Res Ed). 1986;292:1101-1103.
12. Walker MC, Garner PR, Keely EJ, et al. Changes in activated protein C resistance during normal pregnancy. Am J Obstet Gynecol. 1997;177:162-169.
13. Geerts WH, Heit JA, Clagett GP, et al. Prevention of venous thromboembolism. Chest. 2001;119(suppl 1):132S-175S.
14. Belcaro G, Laurora G, Cesarone MR, et al. Prophylaxis of recurrent deep venous thrombosis. A randomized, prospective study using indobufen and graduated elastic compression stockings. Angiology. 1993;44:695-699.
15. Clarke-Pearson DL, Synan IS, Dodge R, et al. A randomized trial of low-dose heparin and intermittent pneumatic calf compression for the prevention of deep venous thrombosis after gynecologic oncology surgery. Am J Obstet Gynecol. 1993;168:1146-1153.
16. Handoll HH, Farrar MJ, McBirnie J, et al. Prophylaxis using heparin, low molecular weight heparin and physical methods against deep vein thrombosis and pulmonary embolism in hip fracture. The Cochrane Library, Issue I. 1998. Evidence-based medicine: 146.
17. Clarke-Pearson DL, Synan IS, Hinshaw WM, et al. Prevention of postoperative venous thromboembolism by external pneumatic calf compression in patients with gynecologic malignancy. Obstet Gynecol. 1984;63(1):92-98.
18. Lachman EA, Rouk JL, et al. Complications associated with intermittent pneumatic compression. Arch Phys Med Rehab. 1992;73:482-485.
19. Clarke-Pearson DL, Dodge RK, Synan I, et al. Venous thromboembolism prophylaxis: patients at high risk to fail intermittent pneumatic compression. Obstet Gynecol. 101;2003:157-163.
20. Wilson NV, Das SK, Kakkar VV, et al. Thrombo-embolic prophylaxis in total knee replacement. Evaluation of the A-V Impulse System. J Bone Joint Surg Br. 1992;74:50-52.
21. Rosenberg RD, Damus PS. The purification and mechanism of action of human antithrombin-heparin cofactor. J Biol Chem. 1973;248:6490-6505.
22. Scurr JH, Coleridge-Smith PD, Hasty JH. Regimen for improved effectiveness of intermittent pneumatic compression in deep venous thrombosis prophylaxis. Surgery. 1987;102:816-820.
23. Collins R, Scrimgeour A, Yusuf S, et al. Reduction in fatal pulmonary embolism and venous thrombosis by perioperative administration of subcutaneous heparin. Overview of results of randomized trials in general, orthopedic, and urologic surgery. N Engl J Med. 1988;318:1162-1173.
24. Hull RD, Pineo GF, Stein PD, et al. Timing of initial administration of low-molecular-weight heparin prophylaxis against deep vein thrombosis in patients following elective hip arthroplasty: a systematic review. Arch Intern Med. 2001;161:1952-1960.
25. Clarke-Pearson DL, DeLong ER, Synan IS, et al. A controlled trial of two low dose heparin regimens for the prevention of postoperative deep vein thrombosis. Obstet Gynecol. 1990;75:684-689.
26. Kakkar VV, Murray WJ. Efficacy and safety of low-molecular-weight heparin (CY216) in preventing postoperative venous thrombo-embolism: a co-operative study. Br J Surg. 1985;72:786-791.
27. Ward B, Pradhan S. Comparison of low molecular weight heparin (Fragmin) with sodium heparin for prophylaxis against postoperative thrombosis in women undergoing major gynaecological surgery. Aust N Z J Obstet Gynaecol. 1998;38:91-92.
28. Bergqvist D, Burmark US, Flordal PA, et al. Low molecular weight heparin started before surgery as prophylaxis against deep vein thrombosis: 2500 versus 5000 XaI units in 2070 patients. Br J Surg. 1995;82:496-501.
29. Bratt G, Tornebohm E, Widlund L, et al. Low molecular weight heparin (KABI 2165, Fragmin): pharmacokinetics after intravenous and subcutaneous administration in human volunteers. Thromb Res. 1986;42:613-620.
30. Kakkar VV, Boedki O, Boneau B, et al. Efficacy and safety profile of a low-molecular weight heparin and standard unfractionated heparin for prophylaxis of postoperative venous thromboembolism: European multicenter trial. World J Surg. 1997;21:2-8.
31. Warkentin TE. Heparin-induced thrombocytopenia: pathogenesis and management. Br J Haematol. 2003;121:535-555.
32. Wysowski DK, Talarico L, Bacsanyi J, et al. Spinal and epidural hematoma and low-molecular-weight heparin. N Engl J Med. 1998;338:1774-1775.
33. Maxwell GL, Myers ER, Clarke-Pearson DL. Cost-effectiveness of deep venous thrombosis prophylaxis in gynecologic oncology surgery. Obstet Gynecol. 2000;95:206-214.
34. Doran FSA. Prevention of deep vein thrombosis. Br J Hosp Med. 1971;6:773-779.
- Prophylaxis must start before surgery for maximal benefit, since at least 50% of postoperative thromboembolic disease begins intraoperatively.
- A consensus conference found that prophylaxis reduced fatal pulmonary emboli by 75% in 7,000 gynecologic surgery patients.
- Low-dose unfractionated heparin and low molecular weight heparin appear similarly effective in reducing thromboembolic disease in perioperative patients, but it is unclear which form has fewer bleeding complications.
The case is strong for routine prophylaxis against venous thrombosis and pulmonary embolism. The primary reasons: efficacy, ease of use, and safety.
This article reviews the evidence on routine prophylaxis, pros and cons of mechanical and drug therapies (including a comparison of 2 heparins), patient risk factors, and cost-effectiveness. A table (page 32) lists patient characteristics for low, moderate, high, and very high levels of risk, with corresponding appropriate preventive measures.
Routine prophylaxis is a wiser strategy than postevent treatment or surveillance because:
- Thromboembolism is a stealthy adversary, particularly pulmonary embolism. Between 10% and 20% of patients with pulmonary emboli die, often within 30 minutes of the sentinel complaint.
- Even when pulmonary embolus is documented during autopsy, as many as 80% of patients have no antecedent clinical evidence of deep venous thrombosis (DVT)—and no chance of life-saving therapy.2
- Surveillance is the least desirable preventive option due to lack of sensitivity of noninvasive tests for thromboembolic disease.
Scope of thromboembolic disease
Thromboembolism is the leading potentially preventable cause of hospital fatality.1
In postoperative gynecologic surgery patients, pulmonary embolism occurs in 0.1% to 5% of cases, depending on risk.2 Venous thrombosis occurs on average in 15% of postoperative gynecologic surgery patients, with a range of 5% to 29%, depending on patient risk factors and the surgical procedures performed.3 Almost 50% of gynecologic patients undergoing surgery for cancer develop a lower-extremity venous thrombosis if left untreated.4
Overall, thromboembolic disease is linked with some 500,000 hospital admissions every year. Pulmonary embolism, the most serious complication, causes 60,000 to 200,000 deaths annually.
High risk of recurrence. Postthrombotic syndrome is characterized by pain, swelling, and leg ulceration. At least 50% of patients successfully treated for proximal venous thrombosis (thigh) and about 33% treated for distal venous thrombosis (calf) develop postthrombotic syndrome.1 The risk for recurrent thrombosis is high.
Risk factors are listed in TABLE 1. The risk of fatal pulmonary embolism is directly related to age; persons over 60 are at greatest risk. When the patient is obese—particularly when she exceeds 120% of ideal body weight—the risk is even greater due to venous stasis.
TABLE 1
Risk factors for thromboembolism
| Age |
| Estrogen use |
| Extended pelvic surgery |
| Hypercoagulable states |
| Immobility |
| Indwelling central venous catheter(s) |
| Lower-extremity paralysis |
| Malignancy |
| Medical illnesses |
| Chronic pulmonary disease |
| Congestive heart failure |
| Diabetes mellitus |
| Inflammatory bowel disease |
| Nephrotic syndrome |
| Obesity |
| Pregnancy |
| Prior thromboembolic disease |
| Radiation therapy |
| Trauma |
| Varicose veins |
Common hypercoagulable states
Activated protein C resistance, the most common thrombophilia, occurs in 3% to 7% of Caucasians (TABLE 2).7 It is usually associated with factor V Leiden mutation.
Prothrombin gene mutation G20210A, the next most common thrombophilia, occurs in 2% of Caucasians.7
Antiphospholipid antibody syndromes are acquired thrombophilias that may be associated with arterial and venous thrombosis, thrombocytopenia, and complications of pregnancy.8
Hyperhomocystinemia may be congenital or acquired. It is associated with venous thromboembolism and early atherosclerosis with arterial thrombosis.
Thrombosis is more likely with multiple predisposing factors. Most patients with one of the thrombophilia syndromes do not have sentinel thrombotic events unless they are further challenged by environmental risks such as oral contraceptives (OCs), pregnancy, prolonged immobilization, or surgery.9
Risk is markedly enhanced if multiple predisposing factors are present. For example, when factor V Leiden mutation and hyperhomocystinemia coexist, the risk for thrombosis increases 10 to 20 times over that of patients with neither abnormality.10 This combined risk far exceeds the sum of risks for each abnormality alone.
TABLE 2
Common hypercoagulable conditions
| Factor V Leiden mutation |
| Prothrombin gene mutation G20210A |
| Antiphospholipid antibody |
| Lupus anticoagulant |
| Anticardiolipin |
| Protein C deficiency |
| Protein S deficiency |
| Antithrombin III deficiency (Heparin cofactor II) |
| Plasminogen deficiency |
| Hyperhomocystinemia |
| Myeloproliferative disorders |
| Polycythemia |
| Primary thrombocytosis |
When to test for thrombophilias
Although women with thrombophilias are at high risk for thromboembolic disease, it is unclear whether identifying the thrombophilia is more beneficial than universal prophylaxis. If a patient has a personal or family history of thromboembolic disease—especially a patient with Caucasian ancestry—testing is probably warranted. While documentation of the thrombophilia may not help with perioperative management, it may be useful for long-term care.
Testing for factor V Leiden mutation is recommended due to its prevalence. If the results are negative in a patient at risk, test for prothrombin gene mutation G20210A, as well as deficiencies in the naturally occurring inhibitors protein C, protein S, and antithrombin III.
Assess antiphospholipid antibodies in women who have experienced recurrent fetal loss or early pregnancy-induced hypertension.
Continue OCs or hormone therapy
OCs and menopausal hormone replacement therapy produce measurable prothrombotic changes in the clotting system that appear to be directly related to the estrogen content. In theory, discontinuing the OC or hormone replacement therapy preoperatively would allow these changes to return to baseline and help prevent thromboembolic disease.
Although the risk of thromboembolic disease is 0.96% if patients are current users of OCs and 0.5% if they are not, studies have failed to confirm a clinical benefit of discontinuation.11 Further, the patient does not return to baseline for 4 to 6 weeks after ceasing therapy.
The potential risk of thromboembolic events also should be weighed against the risk of conception prior to surgery. We usually do not recommend discontinuation of OCs and hormone therapy before surgery, but give prophylaxis based on risk assessment.
Venous stasis, vessel-wall trauma, and increased blood coagulability—the major contributors to perioperative DVT, known as Virchow’s triad—were identified more than 125 years ago.
Venous stasis. Intraoperatively, venous blood return from the lower extremities is reduced to less than half its normal rate,34 secondary to muscle relaxation during anesthesia, which causes venous dilation and reduced blood-flow velocity. Packing the abdominal contents may further impede blood return from the legs.
Resultant venous stasis causes platelet adhesion to the vein wall, followed by release of a thromboplastin-like substance that may trigger thrombus formation.
Blood flow increases in the immediate postoperative period with return of muscle tone, but remains significantly diminished for 21 days because of immobilization—specifically, lack of the usual pumping action of the leg muscles.
Vessel-wall trauma. Veins are highly likely to be damaged as vessels are skeletonized during major pelvic surgery, especially when malignancy is involved. Tissue injury activates the coagulation cascade by exposing blood to tissue thromboplastin (extrinsic path) and subendothelial collagen in the vessel wall, which activates factor XII (intrinsic path). Both pathways lead to conversion of factor X to its active form, factor Xa. Acting in concert with factor V, calcium, and phospholipids from platelet factor III, factor Xa catalyzes the conversion of prothrombin to thrombin. Thrombin regulates the conversion of fibrinogen to fibrin, the basic building block of a thrombus.
Increased blood coagulability. Clotting factors XI, IX, and VII increase following surgery, as do circulating platelets and platelet aggregation. This enhances coagulability, which persists from 72 to 96 hours after surgery but is usually balanced by the fibrinolytic system. Fibrinolysis is mediated primarily by plasmin, which digests fibrin and fibrinogen and activates factors V and VIII. If the fibrinolytic system is overwhelmed, the clotting system is unimpeded and thrombus formation may accelerate.
Pregnancy increases the risk of thrombosis, in part due to the progressive increase in resistance to activated protein C in the second and third trimesters. Risk is increased eightfold in women with inherited deficiency in any of the naturally occurring anticoagulants—antithrombin III, protein C, or protein S—compared with those with no deficiency.12
Preventive strategies
Two approaches to thromboembolic prophylaxis have been proposed, with prevention of fatal postoperative pulmonary emboli as a clear endpoint:
- Stratify a targeted group into levels of risk; then treat those at higher risk. Unfortunately, efforts to define risk have met with only partial success, due to limited availability of noninvasive screening, screening logistics, and expense. Further, specificity and positive predictive value of screening asymptomatic patients is low.
- Use prophylaxis in all patients in the targeted group, regardless of risk (TABLES 3 AND 4). This strategy seems effective. For example, in 2001, the Sixth American College of Chest Physicians Consensus Conference—the most recent of the consensus conferences—evaluated the risks of pulmonary embolus in 7,000 gynecologic surgery patients enrolled in prospective studies. Routine prophylaxis reduced fatal pulmonary emboli by 75%.13
The difficulty of defining patients at highest risk makes the concept of universal prophylaxis for a targeted group an attractive option unless a specific contraindication is identified.
TABLE 3
Risk stratification and prophylactic regimens
| LEVEL OF RISK | PATIENT CHARACTERISTICS | RECOMMENDED REGIMEN |
|---|---|---|
| Low | Less than 40 years of age | No specific recommendation for therapy |
| Undergoing uncomplicated minor surgery | Adequate hydration | |
| Requires less than 30 minutes of anesthesia | Aggressive early ambulation | |
| No additional risk factors | ||
| Moderate | Undergoing minor surgery with additional risk factors | Graduated compression stockings or SPCDs or |
| 40 to 60 years of age, undergoing minor surgery, and no additional risk factors | LDUH 5,000 U every 12 hours or | |
| Less than 40 years of age, undergoing major surgery, and no additional risk factors | LMWH (20 mg) or 2,500 U antifactor Xa once daily | |
| High | More than 60 years of age, undergoing minor surgery or with additional risk factors | LDUH 5,000 U every 8–12 hours or LMWH (40 mg) or 5,000 U antifactor Xa once daily |
| More than 40 years of age undergoing major surgery or with additional risk factors | SPCDs may be added | |
| Very high | More than 60 years of age, undergoing major surgery, with other risk factors such as cancer, prior venous thromboembolism, molecular hypercoagulable state, major trauma, spinal cord injury, or lower-extremity paralysis | Graduated compression stockings or SPCDs and LDUH 5,000 U every 8 hours or |
| Other risk factors such as cancer, prior venous thromboembolism, molecular hypercoagulable state, major trauma, spinal cord injury, or lower-extremity paralysis | LMWH (40 mg) or 5,000 U antifactor Xa once daily | |
| LDUH = low-dose unfractionated heparin; LMWH = low molecular weight heparin; SPCD = sequential pneumatic compression device | ||
| Note: Because perioperative prophylaxis is still evolving, these suggestions should not be considered inviolable. | ||
| Source: Modified from Geerts WH, et al.13 | ||
TABLE 4
Incidence of thromboembolic events based on level of risk
| SITE OF THROMBOSIS | RISK LEVEL (%) | |||
|---|---|---|---|---|
| Low | Moderate | High | Very high | |
| Calf veins (distal) | 2 | 10–35 | 30–60 | 63.5 |
| Iliofemoral veins (proximal) | 0.4 | 2–8 | 5–10 | 10–20 |
| Pulmonary emboli | 0.2 | 1–2 | 2–4 | 4–10 |
| Fatal pulmonary emboli | 0.002 | 0.1–0.5 | 0.4–1 | 0.2–5 |
| Source: Modified from Geerts WH, et al.13 | ||||
Drug and mechanical therapies
Strategies for preventing thromboembolic disease attempt to mitigate the impact of venous stasis, endothelial injury, and hypercoagulable states. Traditionally, early ambulation, adequate hydration, and elevation of the lower extremities have been used, because they are simple and inexpensive interventions.
More recently, pharmacologic and mechanical therapies have proven effective in reducing the incidence of DVT and fatal pulmonary emboli. Prophylactic measures commonly used today are:
- graduated compression stockings
- external pneumatic leg or foot compression devices
- low-dose unfractionated heparin
- low molecular weight heparin
MECHANICAL THERAPIESGraduated compression stockings
Compression stockings are one of the earliest methods of preventing perioperative thrombosis. Compression is greatest at the toe and gradually diminishes toward the thigh. When Belcaro14 studied the risk of recurrent venous thrombosis in nonsurgical hospitalized patients, thrombosis recurred in 40% of patients with no therapy, but only 9.4% of patients wearing graduated compression stockings (GCS). Adding oral antiplatelet therapy lowered the risk to 2%. However, GCS were not superior to any other method of preventing recurrent DVT.
As for surgical patients, a single study15 demonstrated protection against DVT when compared with no GCS in elective gynecologic surgery patients.
Today, GCS are usually a perioperative adjunct to other preventive methods, to provide added protection.
Proper fitting by trained personnel is vital; otherwise, a tourniquet effect may cause venous stasis and reduce benefit.
Sequential pneumatic compression devices
Like GCS, these devices decrease the caliber of veins by simple compression. They also increase blood flow velocity and stimulate the endogenous fibrinolytic system.
Enhanced fibrinolytic activity due to intermittent compression occurs even if the device is used on only 1 lower extremity, or on an upper extremity. Patient and nursing-staff compliance may affect efficacy. Sequential pneumatic compression devices (SPCDs) may be thought inconvenient, impeding nursing functions. Some patients may find the repetitive inflation-deflation cycles annoying.
These devices must be activated prior to surgery and continued for a minimum of 24 hours. Some studies suggest that SPCDs be used for 5 days in high-risk situations.16
Calf- and thigh-length devices have similar effects. Rare complications include peroneal nerve injury and compartment syndrome.
What the data show. In an analysis of 4 trials comparing SPCDs with no therapy, thromboembolic disease occurred in only 2% of patients who wore SPCDs but in 20% of those who did not.17 Compared with low-dose unfractionated heparin, no difference in the rate of DVT was seen.18 However, risk of transfusion and retroperitoneal drainage volume increased in the heparin group.
Contraindications include active or suspected DVT, congestive heart failure, known pulmonary embolus, and leg injuries.19
Combined therapy may be best for high-risk women. A study20 of women who developed thromboemboli despite appropriate treatment with SPCDs found the women more likely to be older than 60 years and/or to have cancer or history of thromboembolic disease or hypertension. This high-risk group may benefit from combined therapy.
Foot compression devices resemble booties and mimic the plantar compression that occurs during walking. They increase blood-flow velocity, stimulate the endogenous fibrinolytic system, and have the same indications as SPCDs. However, they are only moderately effective in reducing venous thrombosis.21 Potential drawbacks are that the devices must be removed when the patient ambulates and replaced when she returns to bed.
DRUG THERAPIESLow-dose unfractionated heparin
Heparin alters the molecular configuration of antithrombin III, making it 1,000 to 4,000 times more potent as an inhibitor of thrombin formation, which in turn limits conversion of fibrinogen to fibrin. This prolongs the activated partial thromboplastin time (aPTT) commonly used to monitor patients receiving full anticoagulation therapy.
A naturally occurring mucopolysaccharide with a molecular weight ranging from 3,000 to 30,000 daltons, heparin is richly concentrated in mast cells. Its anticoagulant properties relate primarily to interaction with antithrombin III (also known as heparin cofactor II), factor IXa, factor Xa, factor XIa, factor XIIa, and platelet aggregation.22
Heparin also inhibits the effects of factor Xa on the coagulation cascade and limits platelet aggregation.23
Half-life is 1 hour for intravenous heparin and about 3 hours for subcutaneous heparin.
Use in pregnancy. Low-dose unfractionated heparin (LDUH) does not cross the placenta and is safe to use during pregnancy.
What the data show. Randomized clinical trials conducted prior to 1988 showed venous thrombosis decreased by 70% and pulmonary embolus by 50% in patients treated with LDUH, compared with those receiving no therapy.24
Dosing options. LDUH typically is given as a 5,000-U dose 2 hours before surgery. The single preoperative dose seems to be as effective as multiple preoperative doses.25 Postoperative therapy is instituted 8 to 12 hours after surgery; heparin is given every 8 to 12 hours until the patient is fully ambulatory.
Patients having gynecologic surgery for benign conditions benefit from the twice-daily regimen, and those undergoing gynecologic oncology surgery or other high-risk procedures seem to benefit from thrice-daily dosing.26
These regimens also significantly reduced DVT and fatal pulmonary emboli in general surgery patients.27 The LDUH regimen is now used to judge the efficacy of other prophylactic measures.
Low molecular weight heparin
This form of heparin acts primarily by inhibiting factor Xa, which is higher in the coagulation cascade than antithrombin. Thus, low molecular weight heparin (LMWH) is more efficient than unfractionated heparin.
LMWH has a molecular weight of 3,000 to 6,000 daltons and is produced by concentrating the low molecular component of heparin. Because the molecular configuration of antithrombin III is not altered by LMWH, thrombin conversion is minimally inhibited and aPTT is not appreciably affected.
Half-life is approximately 4 hours, by any route of administration. The longer half-life provides a longer dosing interval. Bioavailability is more consistent than that of LDUH, approaching 90% to 95%. The excellent bioavailability allows dosing to be based on lean body mass. Less heparin-induced thrombocytopenia is another plus.
Use in pregnancy. LMWH does not cross the placenta and is safe to use during pregnancy.
What the data show. No randomized trials have compared LMWH therapy with no therapy, with efficacy assessed by venography or fibrinogen uptake. An uncontrolled series28 of 2,030 patients did show that LMWH reduced the incidence of thromboembolic disease.
A randomized controlled trial29 comparing 2,500 U and 5,000 U daily of dalteparin (an LMWH) found greater efficacy in the higher-dose group (6.6% versus 12.7% incidence of DVT), but bleeding complications also were higher (4.7% versus 2.7%). In a subgroup of patients with malignancy, the high-dose therapy remained superior in preventing DVT, and the bleeding risk was equal.
Dosing options. Once a day dosing is normally adequate for prophylaxis; twice-daily dosing is needed for therapy. Enoxaparin is an LMWH that is readily available in the United States and commonly prescribed when use of LMWH is desired. For prophylaxis, it can be given in daily doses of 20 mg, 30 mg, 40 mg, or 60 mg. None of these doses has proven superior to the others. The typical regimen for moderate risk is 20 mg per day; for high risk, 40 mg per day. Enoxaparin appears to convey the same degree of protection as 5,000 units of LDUH every 8 hours.
2 heparins compared
Randomized controlled trials comparing LDUH and LMWH have found them to be similarly effective. In addition, within the LMWH class, all compounds appear to have similar benefits. However, it remains unclear which form of heparin is associated with fewer bleeding complications.
In theory, LMWH would be associated with an increase in these complications due to its longer half-life and increased bioavailability.30 However, studies have not consistently identified excess bleeding in any group.31 Fortunately, excess postoperative bleeding requiring transfusion is uncommon.
Complications of heparin
Heparin-induced thrombocytopenia is a recognized complication, seen in as many as 20% of LDUH patients. The diagnosis is made when the platelet count falls below the lower limits of normal or when the platelet count falls by 50% but remains in the normal range. Two forms of heparin-induced thrombocytopenia have been described.
• Type 1 thrombocytopenia is initially mild, rarely dropping below 100,000 platelets per milliliter. Platelet count monitoring is important, but therapy usually can continue, since the platelet count returns to normal even with continued use. Type 1 thrombocytopenia appears to directly result from platelet activation and is not immune-mediated.
• Type 2 thrombocytopenia occurs 7 to 14 days after starting therapy. Platelet counts frequently drop to 20,000 per milliliter. Type 2 is an immune response caused by antibodies to the heparin-platelet factor 4 complex. Patients are at risk for venous and arterial thrombosis, but often the diagnosis is made only after a complicating thrombotic event.
The risk of type 2 thrombocytopenia appears to be related to the molecular weight of the compound being administered, its dose, and the duration of therapy. Therefore, unfractionated heparin—if given in full anticoagulation doses and beyond 14 days—seems to have the greatest potential for this complication.
Paradoxically, these patients also are at risk for severe hemorrhage. Because mortality and morbidity are high, immediate withdrawal of all forms of heparin—including LMWH—is mandatory.32 Catastrophic hemorrhage or thrombosis may be the first sign. Often a fall in platelet concentration precedes serious complications. Thus, platelet concentration should be monitored at least every 2 days.
Hematoma with conduction anesthesia. Use of major conduction anesthesia in patients who also need LMWH, LDUH, or oral anticoagulants is controversial. LMWH may pose a risk of hematoma if initiated preoperatively, intraoperatively, or within 3 hours of surgery in patients who have a continuous epidural. Hematoma formation seems to occur immediately following catheter withdrawal.
The risk of hematoma appears to be lower with single-dose spinal or single-dose epidural anesthesia. While the incidence of epidural or spinal hematoma is not known, some cases have been associated with long-term neurologic sequelae, including permanent paralysis.
Responding to this concern, the US Food and Drug Administration issued a 1997 advisory noting the risk of spinal hematoma in patients receiving enoxaparin plus conduction anesthesia or lumbar spinal puncture. Most anesthesiologists significantly restrict the use of conduction anesthesia in patients requiring heparin prophylaxis.33
Cost-effectiveness
The cost of perioperative prophylaxis has been compared with the cost of immediate therapy for thromboembolic disease and long-term therapy for postthrombotic syndrome. Across the board, universal prophylaxis with pharmacotherapy or mechanical devices in patients undergoing abdominal surgery is less expensive than no prophylaxis, based on a reduced incidence of DVT. Pneumatic compression seems to be more cost-effective than pharmacotherapy.34
Because LDUH is substantially less expensive than LMWH in the United States, it has a better cost profile when pharmacotherapy is warranted. In Europe, where the cost of heparin compounds is not an issue, LMWH has a slight advantage.
The authors report no financial relationships relevant to this article.
- Prophylaxis must start before surgery for maximal benefit, since at least 50% of postoperative thromboembolic disease begins intraoperatively.
- A consensus conference found that prophylaxis reduced fatal pulmonary emboli by 75% in 7,000 gynecologic surgery patients.
- Low-dose unfractionated heparin and low molecular weight heparin appear similarly effective in reducing thromboembolic disease in perioperative patients, but it is unclear which form has fewer bleeding complications.
The case is strong for routine prophylaxis against venous thrombosis and pulmonary embolism. The primary reasons: efficacy, ease of use, and safety.
This article reviews the evidence on routine prophylaxis, pros and cons of mechanical and drug therapies (including a comparison of 2 heparins), patient risk factors, and cost-effectiveness. A table (page 32) lists patient characteristics for low, moderate, high, and very high levels of risk, with corresponding appropriate preventive measures.
Routine prophylaxis is a wiser strategy than postevent treatment or surveillance because:
- Thromboembolism is a stealthy adversary, particularly pulmonary embolism. Between 10% and 20% of patients with pulmonary emboli die, often within 30 minutes of the sentinel complaint.
- Even when pulmonary embolus is documented during autopsy, as many as 80% of patients have no antecedent clinical evidence of deep venous thrombosis (DVT)—and no chance of life-saving therapy.2
- Surveillance is the least desirable preventive option due to lack of sensitivity of noninvasive tests for thromboembolic disease.
Scope of thromboembolic disease
Thromboembolism is the leading potentially preventable cause of hospital fatality.1
In postoperative gynecologic surgery patients, pulmonary embolism occurs in 0.1% to 5% of cases, depending on risk.2 Venous thrombosis occurs on average in 15% of postoperative gynecologic surgery patients, with a range of 5% to 29%, depending on patient risk factors and the surgical procedures performed.3 Almost 50% of gynecologic patients undergoing surgery for cancer develop a lower-extremity venous thrombosis if left untreated.4
Overall, thromboembolic disease is linked with some 500,000 hospital admissions every year. Pulmonary embolism, the most serious complication, causes 60,000 to 200,000 deaths annually.
High risk of recurrence. Postthrombotic syndrome is characterized by pain, swelling, and leg ulceration. At least 50% of patients successfully treated for proximal venous thrombosis (thigh) and about 33% treated for distal venous thrombosis (calf) develop postthrombotic syndrome.1 The risk for recurrent thrombosis is high.
Risk factors are listed in TABLE 1. The risk of fatal pulmonary embolism is directly related to age; persons over 60 are at greatest risk. When the patient is obese—particularly when she exceeds 120% of ideal body weight—the risk is even greater due to venous stasis.
TABLE 1
Risk factors for thromboembolism
| Age |
| Estrogen use |
| Extended pelvic surgery |
| Hypercoagulable states |
| Immobility |
| Indwelling central venous catheter(s) |
| Lower-extremity paralysis |
| Malignancy |
| Medical illnesses |
| Chronic pulmonary disease |
| Congestive heart failure |
| Diabetes mellitus |
| Inflammatory bowel disease |
| Nephrotic syndrome |
| Obesity |
| Pregnancy |
| Prior thromboembolic disease |
| Radiation therapy |
| Trauma |
| Varicose veins |
Common hypercoagulable states
Activated protein C resistance, the most common thrombophilia, occurs in 3% to 7% of Caucasians (TABLE 2).7 It is usually associated with factor V Leiden mutation.
Prothrombin gene mutation G20210A, the next most common thrombophilia, occurs in 2% of Caucasians.7
Antiphospholipid antibody syndromes are acquired thrombophilias that may be associated with arterial and venous thrombosis, thrombocytopenia, and complications of pregnancy.8
Hyperhomocystinemia may be congenital or acquired. It is associated with venous thromboembolism and early atherosclerosis with arterial thrombosis.
Thrombosis is more likely with multiple predisposing factors. Most patients with one of the thrombophilia syndromes do not have sentinel thrombotic events unless they are further challenged by environmental risks such as oral contraceptives (OCs), pregnancy, prolonged immobilization, or surgery.9
Risk is markedly enhanced if multiple predisposing factors are present. For example, when factor V Leiden mutation and hyperhomocystinemia coexist, the risk for thrombosis increases 10 to 20 times over that of patients with neither abnormality.10 This combined risk far exceeds the sum of risks for each abnormality alone.
TABLE 2
Common hypercoagulable conditions
| Factor V Leiden mutation |
| Prothrombin gene mutation G20210A |
| Antiphospholipid antibody |
| Lupus anticoagulant |
| Anticardiolipin |
| Protein C deficiency |
| Protein S deficiency |
| Antithrombin III deficiency (Heparin cofactor II) |
| Plasminogen deficiency |
| Hyperhomocystinemia |
| Myeloproliferative disorders |
| Polycythemia |
| Primary thrombocytosis |
When to test for thrombophilias
Although women with thrombophilias are at high risk for thromboembolic disease, it is unclear whether identifying the thrombophilia is more beneficial than universal prophylaxis. If a patient has a personal or family history of thromboembolic disease—especially a patient with Caucasian ancestry—testing is probably warranted. While documentation of the thrombophilia may not help with perioperative management, it may be useful for long-term care.
Testing for factor V Leiden mutation is recommended due to its prevalence. If the results are negative in a patient at risk, test for prothrombin gene mutation G20210A, as well as deficiencies in the naturally occurring inhibitors protein C, protein S, and antithrombin III.
Assess antiphospholipid antibodies in women who have experienced recurrent fetal loss or early pregnancy-induced hypertension.
Continue OCs or hormone therapy
OCs and menopausal hormone replacement therapy produce measurable prothrombotic changes in the clotting system that appear to be directly related to the estrogen content. In theory, discontinuing the OC or hormone replacement therapy preoperatively would allow these changes to return to baseline and help prevent thromboembolic disease.
Although the risk of thromboembolic disease is 0.96% if patients are current users of OCs and 0.5% if they are not, studies have failed to confirm a clinical benefit of discontinuation.11 Further, the patient does not return to baseline for 4 to 6 weeks after ceasing therapy.
The potential risk of thromboembolic events also should be weighed against the risk of conception prior to surgery. We usually do not recommend discontinuation of OCs and hormone therapy before surgery, but give prophylaxis based on risk assessment.
Venous stasis, vessel-wall trauma, and increased blood coagulability—the major contributors to perioperative DVT, known as Virchow’s triad—were identified more than 125 years ago.
Venous stasis. Intraoperatively, venous blood return from the lower extremities is reduced to less than half its normal rate,34 secondary to muscle relaxation during anesthesia, which causes venous dilation and reduced blood-flow velocity. Packing the abdominal contents may further impede blood return from the legs.
Resultant venous stasis causes platelet adhesion to the vein wall, followed by release of a thromboplastin-like substance that may trigger thrombus formation.
Blood flow increases in the immediate postoperative period with return of muscle tone, but remains significantly diminished for 21 days because of immobilization—specifically, lack of the usual pumping action of the leg muscles.
Vessel-wall trauma. Veins are highly likely to be damaged as vessels are skeletonized during major pelvic surgery, especially when malignancy is involved. Tissue injury activates the coagulation cascade by exposing blood to tissue thromboplastin (extrinsic path) and subendothelial collagen in the vessel wall, which activates factor XII (intrinsic path). Both pathways lead to conversion of factor X to its active form, factor Xa. Acting in concert with factor V, calcium, and phospholipids from platelet factor III, factor Xa catalyzes the conversion of prothrombin to thrombin. Thrombin regulates the conversion of fibrinogen to fibrin, the basic building block of a thrombus.
Increased blood coagulability. Clotting factors XI, IX, and VII increase following surgery, as do circulating platelets and platelet aggregation. This enhances coagulability, which persists from 72 to 96 hours after surgery but is usually balanced by the fibrinolytic system. Fibrinolysis is mediated primarily by plasmin, which digests fibrin and fibrinogen and activates factors V and VIII. If the fibrinolytic system is overwhelmed, the clotting system is unimpeded and thrombus formation may accelerate.
Pregnancy increases the risk of thrombosis, in part due to the progressive increase in resistance to activated protein C in the second and third trimesters. Risk is increased eightfold in women with inherited deficiency in any of the naturally occurring anticoagulants—antithrombin III, protein C, or protein S—compared with those with no deficiency.12
Preventive strategies
Two approaches to thromboembolic prophylaxis have been proposed, with prevention of fatal postoperative pulmonary emboli as a clear endpoint:
- Stratify a targeted group into levels of risk; then treat those at higher risk. Unfortunately, efforts to define risk have met with only partial success, due to limited availability of noninvasive screening, screening logistics, and expense. Further, specificity and positive predictive value of screening asymptomatic patients is low.
- Use prophylaxis in all patients in the targeted group, regardless of risk (TABLES 3 AND 4). This strategy seems effective. For example, in 2001, the Sixth American College of Chest Physicians Consensus Conference—the most recent of the consensus conferences—evaluated the risks of pulmonary embolus in 7,000 gynecologic surgery patients enrolled in prospective studies. Routine prophylaxis reduced fatal pulmonary emboli by 75%.13
The difficulty of defining patients at highest risk makes the concept of universal prophylaxis for a targeted group an attractive option unless a specific contraindication is identified.
TABLE 3
Risk stratification and prophylactic regimens
| LEVEL OF RISK | PATIENT CHARACTERISTICS | RECOMMENDED REGIMEN |
|---|---|---|
| Low | Less than 40 years of age | No specific recommendation for therapy |
| Undergoing uncomplicated minor surgery | Adequate hydration | |
| Requires less than 30 minutes of anesthesia | Aggressive early ambulation | |
| No additional risk factors | ||
| Moderate | Undergoing minor surgery with additional risk factors | Graduated compression stockings or SPCDs or |
| 40 to 60 years of age, undergoing minor surgery, and no additional risk factors | LDUH 5,000 U every 12 hours or | |
| Less than 40 years of age, undergoing major surgery, and no additional risk factors | LMWH (20 mg) or 2,500 U antifactor Xa once daily | |
| High | More than 60 years of age, undergoing minor surgery or with additional risk factors | LDUH 5,000 U every 8–12 hours or LMWH (40 mg) or 5,000 U antifactor Xa once daily |
| More than 40 years of age undergoing major surgery or with additional risk factors | SPCDs may be added | |
| Very high | More than 60 years of age, undergoing major surgery, with other risk factors such as cancer, prior venous thromboembolism, molecular hypercoagulable state, major trauma, spinal cord injury, or lower-extremity paralysis | Graduated compression stockings or SPCDs and LDUH 5,000 U every 8 hours or |
| Other risk factors such as cancer, prior venous thromboembolism, molecular hypercoagulable state, major trauma, spinal cord injury, or lower-extremity paralysis | LMWH (40 mg) or 5,000 U antifactor Xa once daily | |
| LDUH = low-dose unfractionated heparin; LMWH = low molecular weight heparin; SPCD = sequential pneumatic compression device | ||
| Note: Because perioperative prophylaxis is still evolving, these suggestions should not be considered inviolable. | ||
| Source: Modified from Geerts WH, et al.13 | ||
TABLE 4
Incidence of thromboembolic events based on level of risk
| SITE OF THROMBOSIS | RISK LEVEL (%) | |||
|---|---|---|---|---|
| Low | Moderate | High | Very high | |
| Calf veins (distal) | 2 | 10–35 | 30–60 | 63.5 |
| Iliofemoral veins (proximal) | 0.4 | 2–8 | 5–10 | 10–20 |
| Pulmonary emboli | 0.2 | 1–2 | 2–4 | 4–10 |
| Fatal pulmonary emboli | 0.002 | 0.1–0.5 | 0.4–1 | 0.2–5 |
| Source: Modified from Geerts WH, et al.13 | ||||
Drug and mechanical therapies
Strategies for preventing thromboembolic disease attempt to mitigate the impact of venous stasis, endothelial injury, and hypercoagulable states. Traditionally, early ambulation, adequate hydration, and elevation of the lower extremities have been used, because they are simple and inexpensive interventions.
More recently, pharmacologic and mechanical therapies have proven effective in reducing the incidence of DVT and fatal pulmonary emboli. Prophylactic measures commonly used today are:
- graduated compression stockings
- external pneumatic leg or foot compression devices
- low-dose unfractionated heparin
- low molecular weight heparin
MECHANICAL THERAPIESGraduated compression stockings
Compression stockings are one of the earliest methods of preventing perioperative thrombosis. Compression is greatest at the toe and gradually diminishes toward the thigh. When Belcaro14 studied the risk of recurrent venous thrombosis in nonsurgical hospitalized patients, thrombosis recurred in 40% of patients with no therapy, but only 9.4% of patients wearing graduated compression stockings (GCS). Adding oral antiplatelet therapy lowered the risk to 2%. However, GCS were not superior to any other method of preventing recurrent DVT.
As for surgical patients, a single study15 demonstrated protection against DVT when compared with no GCS in elective gynecologic surgery patients.
Today, GCS are usually a perioperative adjunct to other preventive methods, to provide added protection.
Proper fitting by trained personnel is vital; otherwise, a tourniquet effect may cause venous stasis and reduce benefit.
Sequential pneumatic compression devices
Like GCS, these devices decrease the caliber of veins by simple compression. They also increase blood flow velocity and stimulate the endogenous fibrinolytic system.
Enhanced fibrinolytic activity due to intermittent compression occurs even if the device is used on only 1 lower extremity, or on an upper extremity. Patient and nursing-staff compliance may affect efficacy. Sequential pneumatic compression devices (SPCDs) may be thought inconvenient, impeding nursing functions. Some patients may find the repetitive inflation-deflation cycles annoying.
These devices must be activated prior to surgery and continued for a minimum of 24 hours. Some studies suggest that SPCDs be used for 5 days in high-risk situations.16
Calf- and thigh-length devices have similar effects. Rare complications include peroneal nerve injury and compartment syndrome.
What the data show. In an analysis of 4 trials comparing SPCDs with no therapy, thromboembolic disease occurred in only 2% of patients who wore SPCDs but in 20% of those who did not.17 Compared with low-dose unfractionated heparin, no difference in the rate of DVT was seen.18 However, risk of transfusion and retroperitoneal drainage volume increased in the heparin group.
Contraindications include active or suspected DVT, congestive heart failure, known pulmonary embolus, and leg injuries.19
Combined therapy may be best for high-risk women. A study20 of women who developed thromboemboli despite appropriate treatment with SPCDs found the women more likely to be older than 60 years and/or to have cancer or history of thromboembolic disease or hypertension. This high-risk group may benefit from combined therapy.
Foot compression devices resemble booties and mimic the plantar compression that occurs during walking. They increase blood-flow velocity, stimulate the endogenous fibrinolytic system, and have the same indications as SPCDs. However, they are only moderately effective in reducing venous thrombosis.21 Potential drawbacks are that the devices must be removed when the patient ambulates and replaced when she returns to bed.
DRUG THERAPIESLow-dose unfractionated heparin
Heparin alters the molecular configuration of antithrombin III, making it 1,000 to 4,000 times more potent as an inhibitor of thrombin formation, which in turn limits conversion of fibrinogen to fibrin. This prolongs the activated partial thromboplastin time (aPTT) commonly used to monitor patients receiving full anticoagulation therapy.
A naturally occurring mucopolysaccharide with a molecular weight ranging from 3,000 to 30,000 daltons, heparin is richly concentrated in mast cells. Its anticoagulant properties relate primarily to interaction with antithrombin III (also known as heparin cofactor II), factor IXa, factor Xa, factor XIa, factor XIIa, and platelet aggregation.22
Heparin also inhibits the effects of factor Xa on the coagulation cascade and limits platelet aggregation.23
Half-life is 1 hour for intravenous heparin and about 3 hours for subcutaneous heparin.
Use in pregnancy. Low-dose unfractionated heparin (LDUH) does not cross the placenta and is safe to use during pregnancy.
What the data show. Randomized clinical trials conducted prior to 1988 showed venous thrombosis decreased by 70% and pulmonary embolus by 50% in patients treated with LDUH, compared with those receiving no therapy.24
Dosing options. LDUH typically is given as a 5,000-U dose 2 hours before surgery. The single preoperative dose seems to be as effective as multiple preoperative doses.25 Postoperative therapy is instituted 8 to 12 hours after surgery; heparin is given every 8 to 12 hours until the patient is fully ambulatory.
Patients having gynecologic surgery for benign conditions benefit from the twice-daily regimen, and those undergoing gynecologic oncology surgery or other high-risk procedures seem to benefit from thrice-daily dosing.26
These regimens also significantly reduced DVT and fatal pulmonary emboli in general surgery patients.27 The LDUH regimen is now used to judge the efficacy of other prophylactic measures.
Low molecular weight heparin
This form of heparin acts primarily by inhibiting factor Xa, which is higher in the coagulation cascade than antithrombin. Thus, low molecular weight heparin (LMWH) is more efficient than unfractionated heparin.
LMWH has a molecular weight of 3,000 to 6,000 daltons and is produced by concentrating the low molecular component of heparin. Because the molecular configuration of antithrombin III is not altered by LMWH, thrombin conversion is minimally inhibited and aPTT is not appreciably affected.
Half-life is approximately 4 hours, by any route of administration. The longer half-life provides a longer dosing interval. Bioavailability is more consistent than that of LDUH, approaching 90% to 95%. The excellent bioavailability allows dosing to be based on lean body mass. Less heparin-induced thrombocytopenia is another plus.
Use in pregnancy. LMWH does not cross the placenta and is safe to use during pregnancy.
What the data show. No randomized trials have compared LMWH therapy with no therapy, with efficacy assessed by venography or fibrinogen uptake. An uncontrolled series28 of 2,030 patients did show that LMWH reduced the incidence of thromboembolic disease.
A randomized controlled trial29 comparing 2,500 U and 5,000 U daily of dalteparin (an LMWH) found greater efficacy in the higher-dose group (6.6% versus 12.7% incidence of DVT), but bleeding complications also were higher (4.7% versus 2.7%). In a subgroup of patients with malignancy, the high-dose therapy remained superior in preventing DVT, and the bleeding risk was equal.
Dosing options. Once a day dosing is normally adequate for prophylaxis; twice-daily dosing is needed for therapy. Enoxaparin is an LMWH that is readily available in the United States and commonly prescribed when use of LMWH is desired. For prophylaxis, it can be given in daily doses of 20 mg, 30 mg, 40 mg, or 60 mg. None of these doses has proven superior to the others. The typical regimen for moderate risk is 20 mg per day; for high risk, 40 mg per day. Enoxaparin appears to convey the same degree of protection as 5,000 units of LDUH every 8 hours.
2 heparins compared
Randomized controlled trials comparing LDUH and LMWH have found them to be similarly effective. In addition, within the LMWH class, all compounds appear to have similar benefits. However, it remains unclear which form of heparin is associated with fewer bleeding complications.
In theory, LMWH would be associated with an increase in these complications due to its longer half-life and increased bioavailability.30 However, studies have not consistently identified excess bleeding in any group.31 Fortunately, excess postoperative bleeding requiring transfusion is uncommon.
Complications of heparin
Heparin-induced thrombocytopenia is a recognized complication, seen in as many as 20% of LDUH patients. The diagnosis is made when the platelet count falls below the lower limits of normal or when the platelet count falls by 50% but remains in the normal range. Two forms of heparin-induced thrombocytopenia have been described.
• Type 1 thrombocytopenia is initially mild, rarely dropping below 100,000 platelets per milliliter. Platelet count monitoring is important, but therapy usually can continue, since the platelet count returns to normal even with continued use. Type 1 thrombocytopenia appears to directly result from platelet activation and is not immune-mediated.
• Type 2 thrombocytopenia occurs 7 to 14 days after starting therapy. Platelet counts frequently drop to 20,000 per milliliter. Type 2 is an immune response caused by antibodies to the heparin-platelet factor 4 complex. Patients are at risk for venous and arterial thrombosis, but often the diagnosis is made only after a complicating thrombotic event.
The risk of type 2 thrombocytopenia appears to be related to the molecular weight of the compound being administered, its dose, and the duration of therapy. Therefore, unfractionated heparin—if given in full anticoagulation doses and beyond 14 days—seems to have the greatest potential for this complication.
Paradoxically, these patients also are at risk for severe hemorrhage. Because mortality and morbidity are high, immediate withdrawal of all forms of heparin—including LMWH—is mandatory.32 Catastrophic hemorrhage or thrombosis may be the first sign. Often a fall in platelet concentration precedes serious complications. Thus, platelet concentration should be monitored at least every 2 days.
Hematoma with conduction anesthesia. Use of major conduction anesthesia in patients who also need LMWH, LDUH, or oral anticoagulants is controversial. LMWH may pose a risk of hematoma if initiated preoperatively, intraoperatively, or within 3 hours of surgery in patients who have a continuous epidural. Hematoma formation seems to occur immediately following catheter withdrawal.
The risk of hematoma appears to be lower with single-dose spinal or single-dose epidural anesthesia. While the incidence of epidural or spinal hematoma is not known, some cases have been associated with long-term neurologic sequelae, including permanent paralysis.
Responding to this concern, the US Food and Drug Administration issued a 1997 advisory noting the risk of spinal hematoma in patients receiving enoxaparin plus conduction anesthesia or lumbar spinal puncture. Most anesthesiologists significantly restrict the use of conduction anesthesia in patients requiring heparin prophylaxis.33
Cost-effectiveness
The cost of perioperative prophylaxis has been compared with the cost of immediate therapy for thromboembolic disease and long-term therapy for postthrombotic syndrome. Across the board, universal prophylaxis with pharmacotherapy or mechanical devices in patients undergoing abdominal surgery is less expensive than no prophylaxis, based on a reduced incidence of DVT. Pneumatic compression seems to be more cost-effective than pharmacotherapy.34
Because LDUH is substantially less expensive than LMWH in the United States, it has a better cost profile when pharmacotherapy is warranted. In Europe, where the cost of heparin compounds is not an issue, LMWH has a slight advantage.
The authors report no financial relationships relevant to this article.
1. Dismuke SE, Wagner EH. Pulmonary embolism as a cause of death. The changing mortality in hospitalized patients. JAMA. 1986;255:2039-2042.
2. Farquharson DI, Orr JW, Jr. Prophylaxis against thromboembolism in gynecologic patients. J Reprod Med. 1984;29:845-862.
3. Bergqvist D. Prolonged prophylaxis against postoperative venous thromboembolism. Haemostasis. 1996;26(suppl 4):379-387.
4. Clarke-Pearson DL, DeLong ER, Synan IS, Coleman RE, Creasman WT. Variables associated with postoperative deep venous thrombosis: a prospective study of 411 gynecology patients and creation of a prognostic model. Obstet Gynecol. 1987;69:146-150.
5. Kemble JVH. Incidence of deep vein thrombosis. Br J Hosp Med. 1971;6:721-726.
6. Spritzer CE, Evans AC, Kay HH. Magnetic resonance imaging of deep venous thrombosis in pregnant women with lower extremity edema. Obstet Gynecol. 1995;85:603-607.
7. Florell SR, Rodgers GM. Inherited thrombotic disorders: an update. Am J Hematol. 1997;54:53-60.
8. Shapiro GA. Antiphospholipid syndrome in obstetrics and gynecology. Semin Thromb Hemost. 1994;20:64-70.
9. De Stafano V, Leone G, Mastrangelo S, et al. Clinical manifestations and management of inherited thrombophilia: retrospective analysis and follow-up after diagnosis in 238 patients with congenital deficiency of antithrombin III, protein C, protein S. S Throm Haemost. 1994;72:352-358.
10. Ridker PM, Hennekens CH, Selhub J, et al. Interrelation of hyperhomocyst(e)inemia, factor V Leiden, and risk of future venous thromboembolism. Circulation. 1997;95:1777-1782.
11. Vessey M, Jewell D, Smith A, Yeates D, McPherson K. Chronic inflammatory bowel disease, cigarette smoking, and use of oral contraceptives: findings in a large cohort study of women of childbearing age. Br Med J (Clin Res Ed). 1986;292:1101-1103.
12. Walker MC, Garner PR, Keely EJ, et al. Changes in activated protein C resistance during normal pregnancy. Am J Obstet Gynecol. 1997;177:162-169.
13. Geerts WH, Heit JA, Clagett GP, et al. Prevention of venous thromboembolism. Chest. 2001;119(suppl 1):132S-175S.
14. Belcaro G, Laurora G, Cesarone MR, et al. Prophylaxis of recurrent deep venous thrombosis. A randomized, prospective study using indobufen and graduated elastic compression stockings. Angiology. 1993;44:695-699.
15. Clarke-Pearson DL, Synan IS, Dodge R, et al. A randomized trial of low-dose heparin and intermittent pneumatic calf compression for the prevention of deep venous thrombosis after gynecologic oncology surgery. Am J Obstet Gynecol. 1993;168:1146-1153.
16. Handoll HH, Farrar MJ, McBirnie J, et al. Prophylaxis using heparin, low molecular weight heparin and physical methods against deep vein thrombosis and pulmonary embolism in hip fracture. The Cochrane Library, Issue I. 1998. Evidence-based medicine: 146.
17. Clarke-Pearson DL, Synan IS, Hinshaw WM, et al. Prevention of postoperative venous thromboembolism by external pneumatic calf compression in patients with gynecologic malignancy. Obstet Gynecol. 1984;63(1):92-98.
18. Lachman EA, Rouk JL, et al. Complications associated with intermittent pneumatic compression. Arch Phys Med Rehab. 1992;73:482-485.
19. Clarke-Pearson DL, Dodge RK, Synan I, et al. Venous thromboembolism prophylaxis: patients at high risk to fail intermittent pneumatic compression. Obstet Gynecol. 101;2003:157-163.
20. Wilson NV, Das SK, Kakkar VV, et al. Thrombo-embolic prophylaxis in total knee replacement. Evaluation of the A-V Impulse System. J Bone Joint Surg Br. 1992;74:50-52.
21. Rosenberg RD, Damus PS. The purification and mechanism of action of human antithrombin-heparin cofactor. J Biol Chem. 1973;248:6490-6505.
22. Scurr JH, Coleridge-Smith PD, Hasty JH. Regimen for improved effectiveness of intermittent pneumatic compression in deep venous thrombosis prophylaxis. Surgery. 1987;102:816-820.
23. Collins R, Scrimgeour A, Yusuf S, et al. Reduction in fatal pulmonary embolism and venous thrombosis by perioperative administration of subcutaneous heparin. Overview of results of randomized trials in general, orthopedic, and urologic surgery. N Engl J Med. 1988;318:1162-1173.
24. Hull RD, Pineo GF, Stein PD, et al. Timing of initial administration of low-molecular-weight heparin prophylaxis against deep vein thrombosis in patients following elective hip arthroplasty: a systematic review. Arch Intern Med. 2001;161:1952-1960.
25. Clarke-Pearson DL, DeLong ER, Synan IS, et al. A controlled trial of two low dose heparin regimens for the prevention of postoperative deep vein thrombosis. Obstet Gynecol. 1990;75:684-689.
26. Kakkar VV, Murray WJ. Efficacy and safety of low-molecular-weight heparin (CY216) in preventing postoperative venous thrombo-embolism: a co-operative study. Br J Surg. 1985;72:786-791.
27. Ward B, Pradhan S. Comparison of low molecular weight heparin (Fragmin) with sodium heparin for prophylaxis against postoperative thrombosis in women undergoing major gynaecological surgery. Aust N Z J Obstet Gynaecol. 1998;38:91-92.
28. Bergqvist D, Burmark US, Flordal PA, et al. Low molecular weight heparin started before surgery as prophylaxis against deep vein thrombosis: 2500 versus 5000 XaI units in 2070 patients. Br J Surg. 1995;82:496-501.
29. Bratt G, Tornebohm E, Widlund L, et al. Low molecular weight heparin (KABI 2165, Fragmin): pharmacokinetics after intravenous and subcutaneous administration in human volunteers. Thromb Res. 1986;42:613-620.
30. Kakkar VV, Boedki O, Boneau B, et al. Efficacy and safety profile of a low-molecular weight heparin and standard unfractionated heparin for prophylaxis of postoperative venous thromboembolism: European multicenter trial. World J Surg. 1997;21:2-8.
31. Warkentin TE. Heparin-induced thrombocytopenia: pathogenesis and management. Br J Haematol. 2003;121:535-555.
32. Wysowski DK, Talarico L, Bacsanyi J, et al. Spinal and epidural hematoma and low-molecular-weight heparin. N Engl J Med. 1998;338:1774-1775.
33. Maxwell GL, Myers ER, Clarke-Pearson DL. Cost-effectiveness of deep venous thrombosis prophylaxis in gynecologic oncology surgery. Obstet Gynecol. 2000;95:206-214.
34. Doran FSA. Prevention of deep vein thrombosis. Br J Hosp Med. 1971;6:773-779.
1. Dismuke SE, Wagner EH. Pulmonary embolism as a cause of death. The changing mortality in hospitalized patients. JAMA. 1986;255:2039-2042.
2. Farquharson DI, Orr JW, Jr. Prophylaxis against thromboembolism in gynecologic patients. J Reprod Med. 1984;29:845-862.
3. Bergqvist D. Prolonged prophylaxis against postoperative venous thromboembolism. Haemostasis. 1996;26(suppl 4):379-387.
4. Clarke-Pearson DL, DeLong ER, Synan IS, Coleman RE, Creasman WT. Variables associated with postoperative deep venous thrombosis: a prospective study of 411 gynecology patients and creation of a prognostic model. Obstet Gynecol. 1987;69:146-150.
5. Kemble JVH. Incidence of deep vein thrombosis. Br J Hosp Med. 1971;6:721-726.
6. Spritzer CE, Evans AC, Kay HH. Magnetic resonance imaging of deep venous thrombosis in pregnant women with lower extremity edema. Obstet Gynecol. 1995;85:603-607.
7. Florell SR, Rodgers GM. Inherited thrombotic disorders: an update. Am J Hematol. 1997;54:53-60.
8. Shapiro GA. Antiphospholipid syndrome in obstetrics and gynecology. Semin Thromb Hemost. 1994;20:64-70.
9. De Stafano V, Leone G, Mastrangelo S, et al. Clinical manifestations and management of inherited thrombophilia: retrospective analysis and follow-up after diagnosis in 238 patients with congenital deficiency of antithrombin III, protein C, protein S. S Throm Haemost. 1994;72:352-358.
10. Ridker PM, Hennekens CH, Selhub J, et al. Interrelation of hyperhomocyst(e)inemia, factor V Leiden, and risk of future venous thromboembolism. Circulation. 1997;95:1777-1782.
11. Vessey M, Jewell D, Smith A, Yeates D, McPherson K. Chronic inflammatory bowel disease, cigarette smoking, and use of oral contraceptives: findings in a large cohort study of women of childbearing age. Br Med J (Clin Res Ed). 1986;292:1101-1103.
12. Walker MC, Garner PR, Keely EJ, et al. Changes in activated protein C resistance during normal pregnancy. Am J Obstet Gynecol. 1997;177:162-169.
13. Geerts WH, Heit JA, Clagett GP, et al. Prevention of venous thromboembolism. Chest. 2001;119(suppl 1):132S-175S.
14. Belcaro G, Laurora G, Cesarone MR, et al. Prophylaxis of recurrent deep venous thrombosis. A randomized, prospective study using indobufen and graduated elastic compression stockings. Angiology. 1993;44:695-699.
15. Clarke-Pearson DL, Synan IS, Dodge R, et al. A randomized trial of low-dose heparin and intermittent pneumatic calf compression for the prevention of deep venous thrombosis after gynecologic oncology surgery. Am J Obstet Gynecol. 1993;168:1146-1153.
16. Handoll HH, Farrar MJ, McBirnie J, et al. Prophylaxis using heparin, low molecular weight heparin and physical methods against deep vein thrombosis and pulmonary embolism in hip fracture. The Cochrane Library, Issue I. 1998. Evidence-based medicine: 146.
17. Clarke-Pearson DL, Synan IS, Hinshaw WM, et al. Prevention of postoperative venous thromboembolism by external pneumatic calf compression in patients with gynecologic malignancy. Obstet Gynecol. 1984;63(1):92-98.
18. Lachman EA, Rouk JL, et al. Complications associated with intermittent pneumatic compression. Arch Phys Med Rehab. 1992;73:482-485.
19. Clarke-Pearson DL, Dodge RK, Synan I, et al. Venous thromboembolism prophylaxis: patients at high risk to fail intermittent pneumatic compression. Obstet Gynecol. 101;2003:157-163.
20. Wilson NV, Das SK, Kakkar VV, et al. Thrombo-embolic prophylaxis in total knee replacement. Evaluation of the A-V Impulse System. J Bone Joint Surg Br. 1992;74:50-52.
21. Rosenberg RD, Damus PS. The purification and mechanism of action of human antithrombin-heparin cofactor. J Biol Chem. 1973;248:6490-6505.
22. Scurr JH, Coleridge-Smith PD, Hasty JH. Regimen for improved effectiveness of intermittent pneumatic compression in deep venous thrombosis prophylaxis. Surgery. 1987;102:816-820.
23. Collins R, Scrimgeour A, Yusuf S, et al. Reduction in fatal pulmonary embolism and venous thrombosis by perioperative administration of subcutaneous heparin. Overview of results of randomized trials in general, orthopedic, and urologic surgery. N Engl J Med. 1988;318:1162-1173.
24. Hull RD, Pineo GF, Stein PD, et al. Timing of initial administration of low-molecular-weight heparin prophylaxis against deep vein thrombosis in patients following elective hip arthroplasty: a systematic review. Arch Intern Med. 2001;161:1952-1960.
25. Clarke-Pearson DL, DeLong ER, Synan IS, et al. A controlled trial of two low dose heparin regimens for the prevention of postoperative deep vein thrombosis. Obstet Gynecol. 1990;75:684-689.
26. Kakkar VV, Murray WJ. Efficacy and safety of low-molecular-weight heparin (CY216) in preventing postoperative venous thrombo-embolism: a co-operative study. Br J Surg. 1985;72:786-791.
27. Ward B, Pradhan S. Comparison of low molecular weight heparin (Fragmin) with sodium heparin for prophylaxis against postoperative thrombosis in women undergoing major gynaecological surgery. Aust N Z J Obstet Gynaecol. 1998;38:91-92.
28. Bergqvist D, Burmark US, Flordal PA, et al. Low molecular weight heparin started before surgery as prophylaxis against deep vein thrombosis: 2500 versus 5000 XaI units in 2070 patients. Br J Surg. 1995;82:496-501.
29. Bratt G, Tornebohm E, Widlund L, et al. Low molecular weight heparin (KABI 2165, Fragmin): pharmacokinetics after intravenous and subcutaneous administration in human volunteers. Thromb Res. 1986;42:613-620.
30. Kakkar VV, Boedki O, Boneau B, et al. Efficacy and safety profile of a low-molecular weight heparin and standard unfractionated heparin for prophylaxis of postoperative venous thromboembolism: European multicenter trial. World J Surg. 1997;21:2-8.
31. Warkentin TE. Heparin-induced thrombocytopenia: pathogenesis and management. Br J Haematol. 2003;121:535-555.
32. Wysowski DK, Talarico L, Bacsanyi J, et al. Spinal and epidural hematoma and low-molecular-weight heparin. N Engl J Med. 1998;338:1774-1775.
33. Maxwell GL, Myers ER, Clarke-Pearson DL. Cost-effectiveness of deep venous thrombosis prophylaxis in gynecologic oncology surgery. Obstet Gynecol. 2000;95:206-214.
34. Doran FSA. Prevention of deep vein thrombosis. Br J Hosp Med. 1971;6:773-779.
A Model of Coordinated Services
Creating a Hospital-Wide Network to Prevent Falls
The Missed Pulmonary Embolism
Recent trials spotlight herpes, BV, and labor-related neutrophilia
Over the past few years, research in obstetric and gynecologic infectious diseases has led to improvements in diagnosis and management. Notable advances have been made in preventing group B streptococcus, sexually transmitted diseases, and postoperative pelvic infection.
Despite these successes and the development of broad-spectrum antibiotics, infection remains a serious cause of morbidity and mortality.
This update focuses on 3 studies from the past year:
- A randomized, controlled trial of famciclovir to suppress shedding of recurrent anogenital herpes simplex in women with and without symptoms,
- A randomized, controlled trial of metronidazole versus placebo in gravidas with asymptomatic bacterial vaginosis (BV), and
- The first non-observational investigation that explains the effects of labor on maternal neutrophil phenotype.
Using famciclovir to suppress shedding of recurrent anogenital herpes
Sacks SL. Famciclovir suppression of asymptomatic and symptomatic recurrent anogenital herpes simplex virus shedding in women: a randomized, double-blind, doubledummy, placebo-controlled, parallel-group, single-center trial. J Infect Dis. 2004;189:1341–1347.
- LEVEL I EVIDENCE: RANDOMIZED, CONTROLLED TRIAL
In this study, 169 women with frequently recurring, culture-proven, genital herpes simplex infection were randomized to famciclovir 125 mg, 250 mg, or placebo. Patients kept a daily diary of any symptoms, and each morning performed self-sampling from as high in the vagina as possible, with a second sample taken from external genitalia. This involved swabbing the mons pubis, clitoral hood, labia minora, labia majora, perineum, and the perianal area. Data were analyzed with appropriate statistical tests.
A significant reduction in asymptomatic viral shedding occurred with famciclovir treatment, compared with placebo (P.0001 while reduction of viral shedding in symptomatic women was dose-dependent: for mg versus>P.0001 versus for placebo>P.0001>
This study is notable because viral shedding was reduced in both symptomatic and asymptomatic women. It also opens the door for studies in pregnant women known to have herpes, with the aim of reducing the risk of perinatal transmission.
Does metronidazole eliminate BV in asymptomatic gravidas?
Klebanoff MA, Hauth JC, MacPherson CA, et al. Time course of the regression of asymptomatic bacterial vaginosis in pregnancy with and without treatment. Am J Obstet Gynecol. 2004;190:363–370.
- LEVEL II-1 EVIDENCE: NONRANDOMIZED, CONTROLLED TRIAL
Resolution of BV was defined as a Nugent’s score of less than 7, and restoration of the normal vaginal microflora was defined as a score of less than 4. Overall, 72% and 55% of women in the metronidazole group had scores of less than 7 and less than 4, respectively, at follow-up. In the placebo group, these percentages were 21% and 11%, respectively.
Like most studies of BV, this one is flawed. The reason: Rather than conduct quantitative bacteriology, investigators used the Gram stain to interpret the microbiology of an ecosystem and then related it to a significant medical outcome: preterm birth. They attempted to define BV as an infectious disease and applied principles of infectious disease treatment to it. Most investigators define BV as an alteration in the endogenous microflora, specifically a flora dominated by obligate anaerobic bacteria. However, they treat it as an infection and administer an antibiotic to correct the alteration in the vaginal ecosystem.
Questions raised by this study include: How is asymptomatic BV defined? Is it truly asymptomatic, or have patients simply adapted to their condition? Do these patients have abnormal discharge in color, quantity, and odor?
In addition, the lack of definitive bacteriology in this study necessitates the following assumptions:
- All BV is similar; therefore, similar responses to antimicrobial therapy should be expected.
- A score below 7 but above 4 represents intermediate flora. This microflora is a precursor to BV or predisposes the patient to BV infection.
- A score below 4 represents Lactobacillus-dominant flora.
Lactobacillus. Moreover, it is necessary to determine the species of Lactobacillus present to confirm that it is the correct species to reestablish normal microflora.
Investigators did demonstrate a fact seen in most treatment studies: Metronidazole is not a particularly good agent for treating BV and restoring a patient’s vaginal ecology to Lactobacillus-dominant flora. However, they failed to demonstrate that treatment of asymptomatic BV reduces the risk of preterm delivery in a general obstetric population, or to establish a causal relationship between BV and preterm labor. This did not prevent them from asserting the idea that BV is a risk factor for preterm labor.
Investigators clouded their findings further by concluding that two 2-g doses of metronidazole administered 48 hours apart were effective in 72% of cases (defined as the elimination of BV) and 55% effective in restoring Gram stain scores to the normal range, noting that this effect lasted 2 to 10 weeks or longer. In reality, this is neither elimination of BV nor restoration of a normal range of microflora, but simply an interpretation of the change in Gram stain characteristics. The results do not explain what is happening microbiologically; nor do they clarify the host response to these changing conditions. Intensive quantitative bacteriology is needed, along with research into the microbial ecopathophysiology and host response to specific bacteria, in patients with and without healthy vaginal flora.
It is important to understand vaginal microflora because, when it is altered, it can impact negatively on the patient’s health. Therefore, studies that elucidate the vaginal ecology and the relationships between various bacteria further our understanding of the microbial pathophysiology leading to infection. This, in turn, leads to development of preventive measures, thus reducing the risk for adverse outcomes in both the obstetric and gynecologic patient—especially those undergoing operative procedures.
Is labor inherently protective against infection?
Molloy EJ, O’Neill AJ, Grantham JJ, et al. Labor induces a maternal inflammatory response syndrome. Am J Obstet Gynecol. 2004;190:448–455.
- LEVEL II-3 EVIDENCE: MULTIPLE OBSERVATIONS WITH OR WITHOUT INTERVENTION; UNCONTROLLED STUDIES
Although the number of participants was relatively small, this study was well considered and constructed, investigating several immunologic responses in 5 groups of patients:
- 15 nonpregnant healthy women and 17 healthy men,
- 15 healthy women in labor at term before delivery,
- 9 women with normal term pregnancies before elective cesarean with no labor,
- 9 women before emergency cesarean section after partial labor, and
- 9 women at term before emergency cesarean section without labor.
Interestingly, researchers also observed an increase of CD11b, an adhesion molecule and marker for neutrophil activation. Increased CD11b results in greater neutrophil activity.
Investigators concluded that the rise in neutrophils and diminished apoptosis in laboring patients may be an immunologic response to potential infection associated with labor, and that this phenomenon appears to be an inherent host reaction to prevent infection. It is known that bacteria from the genital tract of laboring women ascend into the uterus and amniotic fluid and colonize the decidua, amniotic membranes, and amniotic fluid. Women subsequently delivered by cesarean section are at a greater risk of infection than women delivered vaginally.
This is an extremely important finding and the first non-observational study to explain the rise in white blood cell counts during labor. It begins to explain the differences in white blood cell counts between laboring and nonlaboring patients and should spawn further investigations of a significant problem: postpartum endometritis.
Dr. Faro reports no financial relationships relevant to this article.
Over the past few years, research in obstetric and gynecologic infectious diseases has led to improvements in diagnosis and management. Notable advances have been made in preventing group B streptococcus, sexually transmitted diseases, and postoperative pelvic infection.
Despite these successes and the development of broad-spectrum antibiotics, infection remains a serious cause of morbidity and mortality.
This update focuses on 3 studies from the past year:
- A randomized, controlled trial of famciclovir to suppress shedding of recurrent anogenital herpes simplex in women with and without symptoms,
- A randomized, controlled trial of metronidazole versus placebo in gravidas with asymptomatic bacterial vaginosis (BV), and
- The first non-observational investigation that explains the effects of labor on maternal neutrophil phenotype.
Using famciclovir to suppress shedding of recurrent anogenital herpes
Sacks SL. Famciclovir suppression of asymptomatic and symptomatic recurrent anogenital herpes simplex virus shedding in women: a randomized, double-blind, doubledummy, placebo-controlled, parallel-group, single-center trial. J Infect Dis. 2004;189:1341–1347.
- LEVEL I EVIDENCE: RANDOMIZED, CONTROLLED TRIAL
In this study, 169 women with frequently recurring, culture-proven, genital herpes simplex infection were randomized to famciclovir 125 mg, 250 mg, or placebo. Patients kept a daily diary of any symptoms, and each morning performed self-sampling from as high in the vagina as possible, with a second sample taken from external genitalia. This involved swabbing the mons pubis, clitoral hood, labia minora, labia majora, perineum, and the perianal area. Data were analyzed with appropriate statistical tests.
A significant reduction in asymptomatic viral shedding occurred with famciclovir treatment, compared with placebo (P.0001 while reduction of viral shedding in symptomatic women was dose-dependent: for mg versus>P.0001 versus for placebo>P.0001>
This study is notable because viral shedding was reduced in both symptomatic and asymptomatic women. It also opens the door for studies in pregnant women known to have herpes, with the aim of reducing the risk of perinatal transmission.
Does metronidazole eliminate BV in asymptomatic gravidas?
Klebanoff MA, Hauth JC, MacPherson CA, et al. Time course of the regression of asymptomatic bacterial vaginosis in pregnancy with and without treatment. Am J Obstet Gynecol. 2004;190:363–370.
- LEVEL II-1 EVIDENCE: NONRANDOMIZED, CONTROLLED TRIAL
Resolution of BV was defined as a Nugent’s score of less than 7, and restoration of the normal vaginal microflora was defined as a score of less than 4. Overall, 72% and 55% of women in the metronidazole group had scores of less than 7 and less than 4, respectively, at follow-up. In the placebo group, these percentages were 21% and 11%, respectively.
Like most studies of BV, this one is flawed. The reason: Rather than conduct quantitative bacteriology, investigators used the Gram stain to interpret the microbiology of an ecosystem and then related it to a significant medical outcome: preterm birth. They attempted to define BV as an infectious disease and applied principles of infectious disease treatment to it. Most investigators define BV as an alteration in the endogenous microflora, specifically a flora dominated by obligate anaerobic bacteria. However, they treat it as an infection and administer an antibiotic to correct the alteration in the vaginal ecosystem.
Questions raised by this study include: How is asymptomatic BV defined? Is it truly asymptomatic, or have patients simply adapted to their condition? Do these patients have abnormal discharge in color, quantity, and odor?
In addition, the lack of definitive bacteriology in this study necessitates the following assumptions:
- All BV is similar; therefore, similar responses to antimicrobial therapy should be expected.
- A score below 7 but above 4 represents intermediate flora. This microflora is a precursor to BV or predisposes the patient to BV infection.
- A score below 4 represents Lactobacillus-dominant flora.
Lactobacillus. Moreover, it is necessary to determine the species of Lactobacillus present to confirm that it is the correct species to reestablish normal microflora.
Investigators did demonstrate a fact seen in most treatment studies: Metronidazole is not a particularly good agent for treating BV and restoring a patient’s vaginal ecology to Lactobacillus-dominant flora. However, they failed to demonstrate that treatment of asymptomatic BV reduces the risk of preterm delivery in a general obstetric population, or to establish a causal relationship between BV and preterm labor. This did not prevent them from asserting the idea that BV is a risk factor for preterm labor.
Investigators clouded their findings further by concluding that two 2-g doses of metronidazole administered 48 hours apart were effective in 72% of cases (defined as the elimination of BV) and 55% effective in restoring Gram stain scores to the normal range, noting that this effect lasted 2 to 10 weeks or longer. In reality, this is neither elimination of BV nor restoration of a normal range of microflora, but simply an interpretation of the change in Gram stain characteristics. The results do not explain what is happening microbiologically; nor do they clarify the host response to these changing conditions. Intensive quantitative bacteriology is needed, along with research into the microbial ecopathophysiology and host response to specific bacteria, in patients with and without healthy vaginal flora.
It is important to understand vaginal microflora because, when it is altered, it can impact negatively on the patient’s health. Therefore, studies that elucidate the vaginal ecology and the relationships between various bacteria further our understanding of the microbial pathophysiology leading to infection. This, in turn, leads to development of preventive measures, thus reducing the risk for adverse outcomes in both the obstetric and gynecologic patient—especially those undergoing operative procedures.
Is labor inherently protective against infection?
Molloy EJ, O’Neill AJ, Grantham JJ, et al. Labor induces a maternal inflammatory response syndrome. Am J Obstet Gynecol. 2004;190:448–455.
- LEVEL II-3 EVIDENCE: MULTIPLE OBSERVATIONS WITH OR WITHOUT INTERVENTION; UNCONTROLLED STUDIES
Although the number of participants was relatively small, this study was well considered and constructed, investigating several immunologic responses in 5 groups of patients:
- 15 nonpregnant healthy women and 17 healthy men,
- 15 healthy women in labor at term before delivery,
- 9 women with normal term pregnancies before elective cesarean with no labor,
- 9 women before emergency cesarean section after partial labor, and
- 9 women at term before emergency cesarean section without labor.
Interestingly, researchers also observed an increase of CD11b, an adhesion molecule and marker for neutrophil activation. Increased CD11b results in greater neutrophil activity.
Investigators concluded that the rise in neutrophils and diminished apoptosis in laboring patients may be an immunologic response to potential infection associated with labor, and that this phenomenon appears to be an inherent host reaction to prevent infection. It is known that bacteria from the genital tract of laboring women ascend into the uterus and amniotic fluid and colonize the decidua, amniotic membranes, and amniotic fluid. Women subsequently delivered by cesarean section are at a greater risk of infection than women delivered vaginally.
This is an extremely important finding and the first non-observational study to explain the rise in white blood cell counts during labor. It begins to explain the differences in white blood cell counts between laboring and nonlaboring patients and should spawn further investigations of a significant problem: postpartum endometritis.
Dr. Faro reports no financial relationships relevant to this article.
Over the past few years, research in obstetric and gynecologic infectious diseases has led to improvements in diagnosis and management. Notable advances have been made in preventing group B streptococcus, sexually transmitted diseases, and postoperative pelvic infection.
Despite these successes and the development of broad-spectrum antibiotics, infection remains a serious cause of morbidity and mortality.
This update focuses on 3 studies from the past year:
- A randomized, controlled trial of famciclovir to suppress shedding of recurrent anogenital herpes simplex in women with and without symptoms,
- A randomized, controlled trial of metronidazole versus placebo in gravidas with asymptomatic bacterial vaginosis (BV), and
- The first non-observational investigation that explains the effects of labor on maternal neutrophil phenotype.
Using famciclovir to suppress shedding of recurrent anogenital herpes
Sacks SL. Famciclovir suppression of asymptomatic and symptomatic recurrent anogenital herpes simplex virus shedding in women: a randomized, double-blind, doubledummy, placebo-controlled, parallel-group, single-center trial. J Infect Dis. 2004;189:1341–1347.
- LEVEL I EVIDENCE: RANDOMIZED, CONTROLLED TRIAL
In this study, 169 women with frequently recurring, culture-proven, genital herpes simplex infection were randomized to famciclovir 125 mg, 250 mg, or placebo. Patients kept a daily diary of any symptoms, and each morning performed self-sampling from as high in the vagina as possible, with a second sample taken from external genitalia. This involved swabbing the mons pubis, clitoral hood, labia minora, labia majora, perineum, and the perianal area. Data were analyzed with appropriate statistical tests.
A significant reduction in asymptomatic viral shedding occurred with famciclovir treatment, compared with placebo (P.0001 while reduction of viral shedding in symptomatic women was dose-dependent: for mg versus>P.0001 versus for placebo>P.0001>
This study is notable because viral shedding was reduced in both symptomatic and asymptomatic women. It also opens the door for studies in pregnant women known to have herpes, with the aim of reducing the risk of perinatal transmission.
Does metronidazole eliminate BV in asymptomatic gravidas?
Klebanoff MA, Hauth JC, MacPherson CA, et al. Time course of the regression of asymptomatic bacterial vaginosis in pregnancy with and without treatment. Am J Obstet Gynecol. 2004;190:363–370.
- LEVEL II-1 EVIDENCE: NONRANDOMIZED, CONTROLLED TRIAL
Resolution of BV was defined as a Nugent’s score of less than 7, and restoration of the normal vaginal microflora was defined as a score of less than 4. Overall, 72% and 55% of women in the metronidazole group had scores of less than 7 and less than 4, respectively, at follow-up. In the placebo group, these percentages were 21% and 11%, respectively.
Like most studies of BV, this one is flawed. The reason: Rather than conduct quantitative bacteriology, investigators used the Gram stain to interpret the microbiology of an ecosystem and then related it to a significant medical outcome: preterm birth. They attempted to define BV as an infectious disease and applied principles of infectious disease treatment to it. Most investigators define BV as an alteration in the endogenous microflora, specifically a flora dominated by obligate anaerobic bacteria. However, they treat it as an infection and administer an antibiotic to correct the alteration in the vaginal ecosystem.
Questions raised by this study include: How is asymptomatic BV defined? Is it truly asymptomatic, or have patients simply adapted to their condition? Do these patients have abnormal discharge in color, quantity, and odor?
In addition, the lack of definitive bacteriology in this study necessitates the following assumptions:
- All BV is similar; therefore, similar responses to antimicrobial therapy should be expected.
- A score below 7 but above 4 represents intermediate flora. This microflora is a precursor to BV or predisposes the patient to BV infection.
- A score below 4 represents Lactobacillus-dominant flora.
Lactobacillus. Moreover, it is necessary to determine the species of Lactobacillus present to confirm that it is the correct species to reestablish normal microflora.
Investigators did demonstrate a fact seen in most treatment studies: Metronidazole is not a particularly good agent for treating BV and restoring a patient’s vaginal ecology to Lactobacillus-dominant flora. However, they failed to demonstrate that treatment of asymptomatic BV reduces the risk of preterm delivery in a general obstetric population, or to establish a causal relationship between BV and preterm labor. This did not prevent them from asserting the idea that BV is a risk factor for preterm labor.
Investigators clouded their findings further by concluding that two 2-g doses of metronidazole administered 48 hours apart were effective in 72% of cases (defined as the elimination of BV) and 55% effective in restoring Gram stain scores to the normal range, noting that this effect lasted 2 to 10 weeks or longer. In reality, this is neither elimination of BV nor restoration of a normal range of microflora, but simply an interpretation of the change in Gram stain characteristics. The results do not explain what is happening microbiologically; nor do they clarify the host response to these changing conditions. Intensive quantitative bacteriology is needed, along with research into the microbial ecopathophysiology and host response to specific bacteria, in patients with and without healthy vaginal flora.
It is important to understand vaginal microflora because, when it is altered, it can impact negatively on the patient’s health. Therefore, studies that elucidate the vaginal ecology and the relationships between various bacteria further our understanding of the microbial pathophysiology leading to infection. This, in turn, leads to development of preventive measures, thus reducing the risk for adverse outcomes in both the obstetric and gynecologic patient—especially those undergoing operative procedures.
Is labor inherently protective against infection?
Molloy EJ, O’Neill AJ, Grantham JJ, et al. Labor induces a maternal inflammatory response syndrome. Am J Obstet Gynecol. 2004;190:448–455.
- LEVEL II-3 EVIDENCE: MULTIPLE OBSERVATIONS WITH OR WITHOUT INTERVENTION; UNCONTROLLED STUDIES
Although the number of participants was relatively small, this study was well considered and constructed, investigating several immunologic responses in 5 groups of patients:
- 15 nonpregnant healthy women and 17 healthy men,
- 15 healthy women in labor at term before delivery,
- 9 women with normal term pregnancies before elective cesarean with no labor,
- 9 women before emergency cesarean section after partial labor, and
- 9 women at term before emergency cesarean section without labor.
Interestingly, researchers also observed an increase of CD11b, an adhesion molecule and marker for neutrophil activation. Increased CD11b results in greater neutrophil activity.
Investigators concluded that the rise in neutrophils and diminished apoptosis in laboring patients may be an immunologic response to potential infection associated with labor, and that this phenomenon appears to be an inherent host reaction to prevent infection. It is known that bacteria from the genital tract of laboring women ascend into the uterus and amniotic fluid and colonize the decidua, amniotic membranes, and amniotic fluid. Women subsequently delivered by cesarean section are at a greater risk of infection than women delivered vaginally.
This is an extremely important finding and the first non-observational study to explain the rise in white blood cell counts during labor. It begins to explain the differences in white blood cell counts between laboring and nonlaboring patients and should spawn further investigations of a significant problem: postpartum endometritis.
Dr. Faro reports no financial relationships relevant to this article.
Vaginal intraepithelial neoplasia: Risky and underrecognized
- Most women diagnosed with vaginal intraepithelial neoplasia (VAIN) have a history of cervical intraepithelial neoplasia.
- Compelling clinical and laboratory data indicate a causal relationship between human papillomavirus and VAIN.
- Like its cervical counterpart, VAIN 3 is thought to have substantial potential to progress to invasive cancer.
- Diagnosis includes careful gross and colposcopic inspection of the entire vagina (with mapping of involved areas), representative colposcopically directed biopsies, and careful palpation of the vaginal walls, especially the vaginal cuff scar.
- Important factors to consider when selecting appropriate treatment for women with VAIN include prior hysterectomy, prior radiation therapy, age, whether she is sexually active, comorbidities, vaginal anatomy, and prior treatments.
We can easily identify vulvar intraepithelial neoplasia (VIN): The patient complains of itching and has a visible lesion. We find cervical intraepithelial neoplasia (CIN) by investigating an abnormal Pap test. But what about vaginal intraepithelial neoplasia (VAIN)? It does not itch and is invisible to the naked eye. A Pap test sometimes catches it, although this test is used mainly to screen for CIN, not VAIN.
VAIN just does not grab our attention. It is uncommon, and invasive vaginal cancer is rare. But before you slip this article into the “obscure disease” file, consider the following:
- VAIN is difficult to diagnose, but some women are at increased risk.
- It is difficult to manage, but understanding the treatment options is important to success.
- The potential for VAIN to evolve into invasive cancer is probably substantial.
- Treatment of invasive vaginal cancer has a high rate of complications and is often unsuccessful.
This article describes the epidemiology, natural history, diagnosis, and treatment of VAIN, focusing primarily on management.
A range of risk factors
As with many uncommon diseases that are difficult to diagnose, good data on the incidence of VAIN are not available. Women diagnosed with VAIN can be in their late teens or senior citizens; mean age is about 50 years.1-5 Race is not mentioned in most studies.
CIN. Although concomitant or subsequent VAIN is very unusual in the approximately 600,000 women identified with CIN each year in the United States, most patients diagnosed with VAIN have a history of CIN. The small number who still have a cervix and are diagnosed with VAIN have a high incidence of concomitant CIN.1 Of women who have had a hysterectomy for CIN, only 1% to 5% are subsequently diagnosed with VAIN.6,7
Since most women who develop vaginal intraepithelial neoplasia have a prior or current history of cervical neoplasia, the “field effect” also renders them at risk for vulvar neoplasia.
HPV. Compelling data indicate a causal relationship between human papillomavirus (HPV) and CIN; the same is true for VAIN.8
Tobacco use. A history of tobacco use is frequent among women diagnosed with VAIN.1
Pelvic radiotherapy is commonly reported in case series.1,3-5 Generally, malignancy is potentially radiation-related if it develops at least 5 to 10 years after treatment.
We do not know whether radiotherapy induces neoplastic transformation in the vagina, but women with a history of radiation warrant long-term follow-up, as they seem to be at increased risk and diagnosis may be difficult.4,5
Chronically immunocompromised women are at particular risk for multifocal lower genital tract neoplasia.9,10
Natural history
The limited data available do suggest that vaginal intraepithelial neoplasia is a premalignant condition.11,12 Unfortunately, little is known about the relationship between severity of the intraepithelial neoplastic process and degree of risk. Natural history studies of VAIN 3 are even more limited than those of CIN 3.
Like its cervical counterpart, VAIN 3 is thought to have substantial potential for progression to invasive cancer.1-4,10-13
EVALUATIONDiagnosis entails inspection, palpation, and directed biopsies
VAIN is most commonly diagnosed after investigation of an abnormal Pap test taken from the vaginal cuff of a woman who has undergone a prior hysterectomy for cervical neoplasia. Occasionally, the disease is identified during colposcopy as extension of a cervical lesion. In either case, VAIN usually involves the upper third of the vagina.1-5,11 A minority of patients will be found to have diffuse multifocal lesions along the vaginal walls.
Adequate diagnosis mandates:
- careful gross and colposcopic inspection of the entire vagina (with mapping of involved areas),
- representative colposcopically directed biopsies,
- careful palpation of the vaginal walls, especially the vaginal cuff scar, and
- in some cases, excision of the vaginal cuff scar.
VAIN is often readily visualized with a colposcope, and the appearance may be more prominent than that of a comparable cervical lesion (FIGURE 1). The lesions are sometimes hyperkeratotic and grossly visible. However, colposcopy of the vagina is more difficult than that of the cervix due to vaginal folding, a larger surface area, and vaginal cuff irregularities.
Vaginal atrophy also creates diagnostic difficulties related to colposcopic assessment, and to overreading of vaginal cytology (eg, when a lesion is interpreted as high-grade dysplasia or suspicious). Reassessment after 4 to 6 weeks of estrogen therapy helps resolve these issues.
FIGURE 1 Colposcopic view of VAIN 3
Note the hyperkeratotic, prominent appearance of the VAIN lesions.
TREATMENT
Because women with VAIN are a heterogeneous group, treatment must be individualized. Important factors to consider when selecting appropriate treatment include prior hysterectomy, prior radiation therapy, age, whether the woman is sexually active, comorbidities, vaginal anatomy, and prior treatments.
Prior hysterectomy
Most women diagnosed with VAIN have previously undergone hysterectomy, usually because of cervical neoplasia.2-5,10 In these women, VAIN is generally confined to the vaginal apex (FIGURE 2).
Watch for occult neoplasia. Buried epithelium within the vaginal cuff scar may harbor occult neoplasia. Patients with VAIN at the vaginal apex5,10,11,13 and a vaginal cuff from a hysterectomy for cervical neoplasia are analogous to women with CIN and an unsatisfactory colposcopy.
Excision is usual management. Most of these women are appropriately managed with excision of the involved vaginal apex, including the cuff scar.1,3-5,11,14 Problematic vaginal shortening is uncommon following this procedure. The status of the resection margin is predictive of the likelihood of recurrence.3
With more extensive vaginal epithelial involvement, consider cuff excision in selected cases to eliminate the potential for occult disease. The remainder of the involved vagina can be treated by other means outlined below.
FIGURE 2 VAIN 3 at vaginal apex
VAIN usually is confined to the apex in women hysterectomized for cervical neoplasia.
When CIN extends onto the vaginal fornix
This is an uncommon scenario, although its underrecognition may explain why some VAIN is diagnosed shortly after hysterectomy for CIN.
Management is simpler than for disease involving the posthysterectomy apex because there is no scarring, distortion, or buried epithelium, and traction on the cervix generally provides good exposure.
Laser vaporization is an option. For many such patients, this is one of the few good indications remaining for laser vaporization.5,15,16
If hysterectomy is indicated, remove the affected portion of the upper vagina along with the cervix.5,17
Multifocal or diffuse pattern: Typical of immunocompromise
Multifocal or diffuse manifestation is likewise uncommon, often involving low-grade neoplasia and condylomatous changes.5,18 This is the pattern typically seen in chronically immunocompromised women.5,10
The natural history of VAIN—especially the malignant potential—is less well understood for women with this disease pattern. When managing such patients, keep in mind the potential for occult neoplasia in the vaginal cuff scar. However, broaden the focus of treatment to encompass the entire vagina. The various management options are described below:
- If the patient has cervical intraepithelial neoplasia 2 or 3, treat with laser, cryotherapy, or large loop excision of the transformation zone at least 2 weeks prior to beginning 5-fluorouracil (5-FU).
- Treat vulvar, anal, or urethral lesions with a laser or local excision prior to 5-FU.
- Treat vulvovaginitis prior to 5-FU.
- Have the patient administer 1/2 vaginal applicator of 5% 5-FU (Efudex; 2.5 g) deep in the vagina weekly at bedtime for 10 weeks. Instruct her to place a tampon or cotton balls into the outer vagina, unless the distal vagina is involved. Also have her apply petroleum jelly to all vulvar tissues, around the urethra, and to the anus. In the morning, she should remove the tampon, wash the vulva with soap and water, and dry carefully. This should be followed by another application of petroleum jelly (sparingly) to vulvar tissues. A mini napkin may help prevent staining of garments.
- When 5-FU treatment ends, have the patient administer 1/3 applicator of vaginal estrogen cream nightly for 2 weeks beginning 3 weeks prior to reevaluation.
- Evaluate with colposcopy and cytology 8 weeks after completing 5-FU therapy.
Observation only is particularly suited for women who have low-grade disease or who are severely debilitated and chronically immunocompromised.
5-fluorouracil (5-FU) cream is a very good treatment when VAIN involves the upper half to two thirds of the vagina.2,9 However, the cream does not reach buried epithelium in the vaginal cuff scar and probably does not consistently treat the lower vagina when applied with the standard vaginal applicator. Applying the cream directly to the lower vagina may be more effective when that region is involved (see “6-step vaginal fluorouracil therapy for intraepithelial neoplasia,” above).
Laser vaporization is well accepted and commonly used.2,9,15,19,20 Advantages are versatility in treatment of multifocal disease without sacrificing vaginal epithelium, and low likelihood of complications. Disadvantages include inability to treat buried vaginal cuff epithelium; technical difficulties in applying the laser to a folding and often distorted (cuff) surface within a confined space; and the expensive equipment, technical support, and surgical expertise required.1,4,5,11,15,16,20
Planned combined treatment using laser vaporization followed by 5-FU cream has been reported efficacious in the treatment of diffuse vaginal condylomata.21 Selected patients with VAIN also may benefit,22 such as those with plaque-like disease (where a thick layer of keratin can reduce penetration of the 5-FU cream), diffuse/multifocal disease where laser vaporization is likely to be incomplete, or vaginal anatomy that makes it difficult to accomplish complete laser vaporization.
Vaginectomy is definitive management for selected patients with extensive VAIN. The operation is done transperineally, although hysterovaginectomy may require a combined approach.23 Leaving the distal third of the vagina intact (when disease distribution allows) makes the operation easier and may help avert iatrogenic urinary incontinence. Follow-up examination—and treatment, if necessary—of the remaining short vaginal stump is fairly easy.
Removal of the vagina is technically demanding in some women. A Schuchardt incision is useful in such instances.
The other obvious disadvantage of vaginectomy is loss of coital function, although placement of a skin graft is an option.
Brachytherapy is another option for treating extensive VAIN in highly selected patients.24,25
A cylindrical apparatus placed in the vagina delivers radiotherapy to the vaginal epithelium; the likelihood of significant morbidity is low. This method is most applicable to poor surgical candidates with extensive VAIN. Disadvantages include fibrosis of the vagina, limited data on efficacy (and particular concern about inadequate dosing to buried or distorted vaginal cuff epithelium), and potential difficulties with follow-up and treatment of recurrence.3,5
“Chemosurgery,” specifically 5-FU cream followed by surgical removal of the then-partially-detached VAIN, followed by additional 5-FU cream, has been used effectively at 1 center.10 Other reported-but-less-investigated methods include cryotherapy, electrocautery, loop electrosurgical excision, and cavitational ultrasonic surgical aspiration (CUSA).3-5
VAIN in a radiated vagina
Most women who develop VAIN in this scenario received radiation therapy many years earlier for carcinoma of the cervix.5,11
The most common sites are the upper third to upper half of the remaining vagina, where radiation changes are prominent.
Diagnosis often is problematic due to:
- difficulties with interpreting cytologic preparations in such patients;
- radiation changes in the vagina (pale and fibrotic with telangiectasis), which largely obscure colposcopic findings;
- the difficulty and potential hazards of biopsy of a thin, fibrotic upper vagina; and
- obliterative coaptation of the upper third to half of the vagina.
Significantly abnormal cytology in the absence of a colposcopically identified lesion (or palpable abnormality) is of particular concern. In such patients, view the abnormal cytologic interpretation with caution and consider further initial evaluation, including outside review of the cytology slides, treatment of the vagina with estrogen, and repeat cytologic and colposcopic evaluation.
When there is clear cytologic evidence of a severe abnormality, consider the possibility of occult neoplasia within the coapted upper vagina and/or cervix.
Management of VAIN in a radiated vagina.
Take into account the anatomic distortion of the upper vagina, the thin and fibrotic nature of the epithelium, and the potential for fistula formation with excisional procedures or other treatments that produce injury beneath the surface of the epithelium. Among the options:
- 5-FU cream or laser vaporization. VAIN that is completely visualized within the remaining vagina is probably best treated with one of these modalities, provided the physician is experienced in managing such patients.5,9
- Excision of the upper vagina,5 including the coapted portion (and sometimes the residual cervix with or without the uterus) is appropriate for highly selected cases, but only after careful consideration of:
- the likelihood of finding significant neoplasia,
- the anatomic feasibility (it is desirable to perform the procedure transvaginally), and
- the overall risk versus benefit. Such procedures should be performed only by an experienced physician.
The chronically immunocompromised
These patients often have undergone organ transplants or are human immunodeficiency virus (HIV)-positive. Diffuse HPV infection of the lower genital tract is pervasive in these women,5,10,26 and diffuse/multifocal lower genital tract intraepithelial neoplasia is often present as well.
These women may be at increased risk for progression to invasive cancer.
Eradication may not be possible. Attempts to eliminate intraepithelial disease are usually unsuccessful.
Management. Many of these women are severely debilitated, with other, more significant medical problems and a short life expectancy. Vigilance is required, as other lower genital tract (and anal) sites are frequently involved. Since eradication of diffuse/multifocal intraepithelial disease is not a realistic goal, treatment followed by chronic suppressive therapy (such as a low intermittent dose of 5-FU) is reasonable.9,10,22
Another approach is close observation, including frequent examinations, with prompt intervention when invasive disease is suspected.
FOLLOW-UP
After treatment for VAIN, follow-up is similar to that for a comparable cervical lesion. Once the vagina has healed, see the patient every 3 to 6 months for 2 years and annually thereafter. In addition to obtaining vaginal cytology, carefully inspect and palpate the vagina (including the vaginal cuff scar).
Effects of 5-FU. Following a course of 5-FU cream, vaginal mucosal ulcerations may persist for several weeks. Occasionally, these may lead to partial coaptations.27 In addition, subsequent islands of columnar epithelium have been described.28 Keep these factors in mind during the follow-up of women who have been treated with transvaginal 5-FU cream.
Long-term annual follow-up is reasonable, since these women are probably at increased risk for developing a second primary vaginal lesion and/or lower anogenital tract neoplasia at other sites.
When extra vigilance is warranted. Some patients merit closer follow-up, such as the chronically immunosuppressed women described; also, women who have been treated for vaginal apical disease without resection of the vaginal cuff scar, previously radiated patients, and women whose VAIN was treated with brachytherapy.
Dr. Hoffman reports no financial relationships relevant to this article.
1. Dodge JA, Eltabbakh GH, Mount SL, Walker RP, Morgan A. Clinical features and risk of recurrence among patients with vaginal intraepithelial neoplasia. Gynecol Oncol. 2001;83:363-369.
2. Petrilli ES, Townsend DE, Morrow CP, Nakao CY. Vaginal intraepithelial neoplasia: biologic aspects and treatment with topical 5-fluorouracil and the carbon dioxide laser. Am J Obstet Gynecol. 1980;138:321-328.
3. Benedet JL, Sanders BH. Carcinoma in situ of the vagina. Am J Obstet Gynecol. 1984;148:695-700.
4. Lenehan PM, Meffe F, Lickrish GM. Vaginal intraepithelial neoplasia: biologic aspects and management. Obstet Gynecol. 1986;68:333-337.
5. Audet-Lapointe P, Body G, Vauclair R, Drouin P, Ayoub J. Vaginal intraepithelial neoplasia. Gynecol Oncol. 1990;36:232-239.
6. Gallup DG, Morley GW. Carcinoma in situ of the vagina—a study and review. Obstet Gynecol. 1975;46:334-440.
7. Woodruff JD. Treatment of recurrent carcinoma in situ in the lower genital canal. Clin Obstet Gynecol. 1965;8:757-770.
8. McCance DJ, Clarkson PK, Dyson JL, Walker PG, Singer A. Human papillomavirus types 6 and 16 in multifocal intraepithelial neoplasias of the female lower genital tract. Br J Obstet Gynaecol. 1985;92:1093-1100.
9. Krebs HB. Treatment of vaginal intraepithelial neoplasia with laser and topical 5-fluorouracil. Obstet Gynecol. 1989;73:657-660.
10. Sillman FH, Fruchter RG, Chen YS, Camilien L, Sedlis A, McTigue E. Vaginal intraepithelial neoplasia: risk factors for persistence, recurrence, and invasion and its management. Am J Obstet Gynecol. 1997;176:93-99.
11. Woodman CBJ, Jordan JA, Wade-Evans T. The management of vaginal intraepithelial neoplasia after hysterectomy. Br J Obstet Gynaecol. 1984;91:103-105.
12. Aho M, Vesterinen E, Meyer B, Purola E, Paavonen J. Natural history of vaginal intraepithelial neoplasia. Cancer. 1991;68:195-197.
13. Hoffman MS, Roberts WS, LaPolla JP, Sterghos S, Jr, Cavanagh D. Neoplasia in vaginal cuff epithelial inclusion cysts after hysterectomy. J Reprod Med. 1989;34:412-416.
14. Hoffman MS, DeCesare SL, Roberts WS, Fiorica JV, Finan MA, Cavanagh D. Upper vaginectomy for in situ and occult, superficially invasive carcinoma of the vagina. Am J Obstet Gynecol. 1992;166:30-33.
15. Curtin JP, Twiggs LB, Julian TM. Treatment of vaginal intraepithelial neoplasia with the CO2laser. J Reprod Med. 1985;30:942-944.
16. Hoffman MS, Roberts WS, LaPolla JP, Fiorica JV, Cavanagh D. Laser vaporization of grade 3 vaginal intraepithelial neoplasia. Am J Obstet Gynecol. 1991;165:1342-1344.
17. Hoffman MS, Spellacy WN. The Difficult Vaginal Hysterectomy. New York, NY: Springer-Verlag; 1995;117-119.
18. Reid R. Human papillomaviral infection. The key to rational triage of cervical neoplasia. Obstet Gynecol Clin North Am. 1987;14:407-429.
19. Townsend DE, Levine RU, Crum CP, Richart RM. Treatment of vaginal carcinoma in situ with the carbon dioxide laser. Am J Obstet Gynecol. 1982;143:565-568.
20. Sherman AI. Laser therapy for vaginal intraepithelial neoplasia after hysterectomy. J Reprod Med. 1990;35—941-944.
21. Krebs HB. Combination of laser plus 5-fluorouracil for the treatment of extensive genital condylomata acuminata. Lasers Surg Med. 1988;8:135-138.
22. Krebs HB. Prophylactic topical 5-fluorouracil following treatment of human papillomavirus-associated lesions of the vulva and vagina. Obstet Gynecol. 1986;68:837-841.
23. Hoffman MS, Bomalaski JJ, Lockhart JL, Greenwald DP. Total removal of the normal non-prolapsed vagina. Technique, results and anatomic observations. J Pelvic Surg. 2002;8:89-94.
24. MacLeod C, Fowler A, Dalrymple C, Atkinson K, Elliott P, Carter J. High-dose-rate brachytherapy in the management of high-grade intraepithelial neoplasia of the vagina. Gynecol Oncol. 1997;65:74-77.
25. Teruya Y, Sakumoto K, Moromizato H, et al. High dose-rate intracavitary brachytherapy for carcinoma in situ of the vagina occurring after hysterectomy: a rational prescription of radiation dose. Am J Obstet Gynecol. 2002;187:360-364.
26. Sillman FH, Sedlis A. Anogenital HPV/neoplasia in immunosuppressed women: an update. Dermatol Clin North Am. 1991;9:353-369.
27. Krebs HB, Helmkamp BF. Chronic ulcerations following topical therapy with 5-fluorouracil for vaginal human papillomavirus-associated lesions. Obstet Gynecol. 1991;78:205-212.
28. Dungar CF, Wilkinson EJ. Vaginal columnar cell metaplasia: an acquired adenosis associated with topical 5-FU therapy. J Reprod Med. 1995;40:361-366.
- Most women diagnosed with vaginal intraepithelial neoplasia (VAIN) have a history of cervical intraepithelial neoplasia.
- Compelling clinical and laboratory data indicate a causal relationship between human papillomavirus and VAIN.
- Like its cervical counterpart, VAIN 3 is thought to have substantial potential to progress to invasive cancer.
- Diagnosis includes careful gross and colposcopic inspection of the entire vagina (with mapping of involved areas), representative colposcopically directed biopsies, and careful palpation of the vaginal walls, especially the vaginal cuff scar.
- Important factors to consider when selecting appropriate treatment for women with VAIN include prior hysterectomy, prior radiation therapy, age, whether she is sexually active, comorbidities, vaginal anatomy, and prior treatments.
We can easily identify vulvar intraepithelial neoplasia (VIN): The patient complains of itching and has a visible lesion. We find cervical intraepithelial neoplasia (CIN) by investigating an abnormal Pap test. But what about vaginal intraepithelial neoplasia (VAIN)? It does not itch and is invisible to the naked eye. A Pap test sometimes catches it, although this test is used mainly to screen for CIN, not VAIN.
VAIN just does not grab our attention. It is uncommon, and invasive vaginal cancer is rare. But before you slip this article into the “obscure disease” file, consider the following:
- VAIN is difficult to diagnose, but some women are at increased risk.
- It is difficult to manage, but understanding the treatment options is important to success.
- The potential for VAIN to evolve into invasive cancer is probably substantial.
- Treatment of invasive vaginal cancer has a high rate of complications and is often unsuccessful.
This article describes the epidemiology, natural history, diagnosis, and treatment of VAIN, focusing primarily on management.
A range of risk factors
As with many uncommon diseases that are difficult to diagnose, good data on the incidence of VAIN are not available. Women diagnosed with VAIN can be in their late teens or senior citizens; mean age is about 50 years.1-5 Race is not mentioned in most studies.
CIN. Although concomitant or subsequent VAIN is very unusual in the approximately 600,000 women identified with CIN each year in the United States, most patients diagnosed with VAIN have a history of CIN. The small number who still have a cervix and are diagnosed with VAIN have a high incidence of concomitant CIN.1 Of women who have had a hysterectomy for CIN, only 1% to 5% are subsequently diagnosed with VAIN.6,7
Since most women who develop vaginal intraepithelial neoplasia have a prior or current history of cervical neoplasia, the “field effect” also renders them at risk for vulvar neoplasia.
HPV. Compelling data indicate a causal relationship between human papillomavirus (HPV) and CIN; the same is true for VAIN.8
Tobacco use. A history of tobacco use is frequent among women diagnosed with VAIN.1
Pelvic radiotherapy is commonly reported in case series.1,3-5 Generally, malignancy is potentially radiation-related if it develops at least 5 to 10 years after treatment.
We do not know whether radiotherapy induces neoplastic transformation in the vagina, but women with a history of radiation warrant long-term follow-up, as they seem to be at increased risk and diagnosis may be difficult.4,5
Chronically immunocompromised women are at particular risk for multifocal lower genital tract neoplasia.9,10
Natural history
The limited data available do suggest that vaginal intraepithelial neoplasia is a premalignant condition.11,12 Unfortunately, little is known about the relationship between severity of the intraepithelial neoplastic process and degree of risk. Natural history studies of VAIN 3 are even more limited than those of CIN 3.
Like its cervical counterpart, VAIN 3 is thought to have substantial potential for progression to invasive cancer.1-4,10-13
EVALUATIONDiagnosis entails inspection, palpation, and directed biopsies
VAIN is most commonly diagnosed after investigation of an abnormal Pap test taken from the vaginal cuff of a woman who has undergone a prior hysterectomy for cervical neoplasia. Occasionally, the disease is identified during colposcopy as extension of a cervical lesion. In either case, VAIN usually involves the upper third of the vagina.1-5,11 A minority of patients will be found to have diffuse multifocal lesions along the vaginal walls.
Adequate diagnosis mandates:
- careful gross and colposcopic inspection of the entire vagina (with mapping of involved areas),
- representative colposcopically directed biopsies,
- careful palpation of the vaginal walls, especially the vaginal cuff scar, and
- in some cases, excision of the vaginal cuff scar.
VAIN is often readily visualized with a colposcope, and the appearance may be more prominent than that of a comparable cervical lesion (FIGURE 1). The lesions are sometimes hyperkeratotic and grossly visible. However, colposcopy of the vagina is more difficult than that of the cervix due to vaginal folding, a larger surface area, and vaginal cuff irregularities.
Vaginal atrophy also creates diagnostic difficulties related to colposcopic assessment, and to overreading of vaginal cytology (eg, when a lesion is interpreted as high-grade dysplasia or suspicious). Reassessment after 4 to 6 weeks of estrogen therapy helps resolve these issues.
FIGURE 1 Colposcopic view of VAIN 3
Note the hyperkeratotic, prominent appearance of the VAIN lesions.
TREATMENT
Because women with VAIN are a heterogeneous group, treatment must be individualized. Important factors to consider when selecting appropriate treatment include prior hysterectomy, prior radiation therapy, age, whether the woman is sexually active, comorbidities, vaginal anatomy, and prior treatments.
Prior hysterectomy
Most women diagnosed with VAIN have previously undergone hysterectomy, usually because of cervical neoplasia.2-5,10 In these women, VAIN is generally confined to the vaginal apex (FIGURE 2).
Watch for occult neoplasia. Buried epithelium within the vaginal cuff scar may harbor occult neoplasia. Patients with VAIN at the vaginal apex5,10,11,13 and a vaginal cuff from a hysterectomy for cervical neoplasia are analogous to women with CIN and an unsatisfactory colposcopy.
Excision is usual management. Most of these women are appropriately managed with excision of the involved vaginal apex, including the cuff scar.1,3-5,11,14 Problematic vaginal shortening is uncommon following this procedure. The status of the resection margin is predictive of the likelihood of recurrence.3
With more extensive vaginal epithelial involvement, consider cuff excision in selected cases to eliminate the potential for occult disease. The remainder of the involved vagina can be treated by other means outlined below.
FIGURE 2 VAIN 3 at vaginal apex
VAIN usually is confined to the apex in women hysterectomized for cervical neoplasia.
When CIN extends onto the vaginal fornix
This is an uncommon scenario, although its underrecognition may explain why some VAIN is diagnosed shortly after hysterectomy for CIN.
Management is simpler than for disease involving the posthysterectomy apex because there is no scarring, distortion, or buried epithelium, and traction on the cervix generally provides good exposure.
Laser vaporization is an option. For many such patients, this is one of the few good indications remaining for laser vaporization.5,15,16
If hysterectomy is indicated, remove the affected portion of the upper vagina along with the cervix.5,17
Multifocal or diffuse pattern: Typical of immunocompromise
Multifocal or diffuse manifestation is likewise uncommon, often involving low-grade neoplasia and condylomatous changes.5,18 This is the pattern typically seen in chronically immunocompromised women.5,10
The natural history of VAIN—especially the malignant potential—is less well understood for women with this disease pattern. When managing such patients, keep in mind the potential for occult neoplasia in the vaginal cuff scar. However, broaden the focus of treatment to encompass the entire vagina. The various management options are described below:
- If the patient has cervical intraepithelial neoplasia 2 or 3, treat with laser, cryotherapy, or large loop excision of the transformation zone at least 2 weeks prior to beginning 5-fluorouracil (5-FU).
- Treat vulvar, anal, or urethral lesions with a laser or local excision prior to 5-FU.
- Treat vulvovaginitis prior to 5-FU.
- Have the patient administer 1/2 vaginal applicator of 5% 5-FU (Efudex; 2.5 g) deep in the vagina weekly at bedtime for 10 weeks. Instruct her to place a tampon or cotton balls into the outer vagina, unless the distal vagina is involved. Also have her apply petroleum jelly to all vulvar tissues, around the urethra, and to the anus. In the morning, she should remove the tampon, wash the vulva with soap and water, and dry carefully. This should be followed by another application of petroleum jelly (sparingly) to vulvar tissues. A mini napkin may help prevent staining of garments.
- When 5-FU treatment ends, have the patient administer 1/3 applicator of vaginal estrogen cream nightly for 2 weeks beginning 3 weeks prior to reevaluation.
- Evaluate with colposcopy and cytology 8 weeks after completing 5-FU therapy.
Observation only is particularly suited for women who have low-grade disease or who are severely debilitated and chronically immunocompromised.
5-fluorouracil (5-FU) cream is a very good treatment when VAIN involves the upper half to two thirds of the vagina.2,9 However, the cream does not reach buried epithelium in the vaginal cuff scar and probably does not consistently treat the lower vagina when applied with the standard vaginal applicator. Applying the cream directly to the lower vagina may be more effective when that region is involved (see “6-step vaginal fluorouracil therapy for intraepithelial neoplasia,” above).
Laser vaporization is well accepted and commonly used.2,9,15,19,20 Advantages are versatility in treatment of multifocal disease without sacrificing vaginal epithelium, and low likelihood of complications. Disadvantages include inability to treat buried vaginal cuff epithelium; technical difficulties in applying the laser to a folding and often distorted (cuff) surface within a confined space; and the expensive equipment, technical support, and surgical expertise required.1,4,5,11,15,16,20
Planned combined treatment using laser vaporization followed by 5-FU cream has been reported efficacious in the treatment of diffuse vaginal condylomata.21 Selected patients with VAIN also may benefit,22 such as those with plaque-like disease (where a thick layer of keratin can reduce penetration of the 5-FU cream), diffuse/multifocal disease where laser vaporization is likely to be incomplete, or vaginal anatomy that makes it difficult to accomplish complete laser vaporization.
Vaginectomy is definitive management for selected patients with extensive VAIN. The operation is done transperineally, although hysterovaginectomy may require a combined approach.23 Leaving the distal third of the vagina intact (when disease distribution allows) makes the operation easier and may help avert iatrogenic urinary incontinence. Follow-up examination—and treatment, if necessary—of the remaining short vaginal stump is fairly easy.
Removal of the vagina is technically demanding in some women. A Schuchardt incision is useful in such instances.
The other obvious disadvantage of vaginectomy is loss of coital function, although placement of a skin graft is an option.
Brachytherapy is another option for treating extensive VAIN in highly selected patients.24,25
A cylindrical apparatus placed in the vagina delivers radiotherapy to the vaginal epithelium; the likelihood of significant morbidity is low. This method is most applicable to poor surgical candidates with extensive VAIN. Disadvantages include fibrosis of the vagina, limited data on efficacy (and particular concern about inadequate dosing to buried or distorted vaginal cuff epithelium), and potential difficulties with follow-up and treatment of recurrence.3,5
“Chemosurgery,” specifically 5-FU cream followed by surgical removal of the then-partially-detached VAIN, followed by additional 5-FU cream, has been used effectively at 1 center.10 Other reported-but-less-investigated methods include cryotherapy, electrocautery, loop electrosurgical excision, and cavitational ultrasonic surgical aspiration (CUSA).3-5
VAIN in a radiated vagina
Most women who develop VAIN in this scenario received radiation therapy many years earlier for carcinoma of the cervix.5,11
The most common sites are the upper third to upper half of the remaining vagina, where radiation changes are prominent.
Diagnosis often is problematic due to:
- difficulties with interpreting cytologic preparations in such patients;
- radiation changes in the vagina (pale and fibrotic with telangiectasis), which largely obscure colposcopic findings;
- the difficulty and potential hazards of biopsy of a thin, fibrotic upper vagina; and
- obliterative coaptation of the upper third to half of the vagina.
Significantly abnormal cytology in the absence of a colposcopically identified lesion (or palpable abnormality) is of particular concern. In such patients, view the abnormal cytologic interpretation with caution and consider further initial evaluation, including outside review of the cytology slides, treatment of the vagina with estrogen, and repeat cytologic and colposcopic evaluation.
When there is clear cytologic evidence of a severe abnormality, consider the possibility of occult neoplasia within the coapted upper vagina and/or cervix.
Management of VAIN in a radiated vagina.
Take into account the anatomic distortion of the upper vagina, the thin and fibrotic nature of the epithelium, and the potential for fistula formation with excisional procedures or other treatments that produce injury beneath the surface of the epithelium. Among the options:
- 5-FU cream or laser vaporization. VAIN that is completely visualized within the remaining vagina is probably best treated with one of these modalities, provided the physician is experienced in managing such patients.5,9
- Excision of the upper vagina,5 including the coapted portion (and sometimes the residual cervix with or without the uterus) is appropriate for highly selected cases, but only after careful consideration of:
- the likelihood of finding significant neoplasia,
- the anatomic feasibility (it is desirable to perform the procedure transvaginally), and
- the overall risk versus benefit. Such procedures should be performed only by an experienced physician.
The chronically immunocompromised
These patients often have undergone organ transplants or are human immunodeficiency virus (HIV)-positive. Diffuse HPV infection of the lower genital tract is pervasive in these women,5,10,26 and diffuse/multifocal lower genital tract intraepithelial neoplasia is often present as well.
These women may be at increased risk for progression to invasive cancer.
Eradication may not be possible. Attempts to eliminate intraepithelial disease are usually unsuccessful.
Management. Many of these women are severely debilitated, with other, more significant medical problems and a short life expectancy. Vigilance is required, as other lower genital tract (and anal) sites are frequently involved. Since eradication of diffuse/multifocal intraepithelial disease is not a realistic goal, treatment followed by chronic suppressive therapy (such as a low intermittent dose of 5-FU) is reasonable.9,10,22
Another approach is close observation, including frequent examinations, with prompt intervention when invasive disease is suspected.
FOLLOW-UP
After treatment for VAIN, follow-up is similar to that for a comparable cervical lesion. Once the vagina has healed, see the patient every 3 to 6 months for 2 years and annually thereafter. In addition to obtaining vaginal cytology, carefully inspect and palpate the vagina (including the vaginal cuff scar).
Effects of 5-FU. Following a course of 5-FU cream, vaginal mucosal ulcerations may persist for several weeks. Occasionally, these may lead to partial coaptations.27 In addition, subsequent islands of columnar epithelium have been described.28 Keep these factors in mind during the follow-up of women who have been treated with transvaginal 5-FU cream.
Long-term annual follow-up is reasonable, since these women are probably at increased risk for developing a second primary vaginal lesion and/or lower anogenital tract neoplasia at other sites.
When extra vigilance is warranted. Some patients merit closer follow-up, such as the chronically immunosuppressed women described; also, women who have been treated for vaginal apical disease without resection of the vaginal cuff scar, previously radiated patients, and women whose VAIN was treated with brachytherapy.
Dr. Hoffman reports no financial relationships relevant to this article.
- Most women diagnosed with vaginal intraepithelial neoplasia (VAIN) have a history of cervical intraepithelial neoplasia.
- Compelling clinical and laboratory data indicate a causal relationship between human papillomavirus and VAIN.
- Like its cervical counterpart, VAIN 3 is thought to have substantial potential to progress to invasive cancer.
- Diagnosis includes careful gross and colposcopic inspection of the entire vagina (with mapping of involved areas), representative colposcopically directed biopsies, and careful palpation of the vaginal walls, especially the vaginal cuff scar.
- Important factors to consider when selecting appropriate treatment for women with VAIN include prior hysterectomy, prior radiation therapy, age, whether she is sexually active, comorbidities, vaginal anatomy, and prior treatments.
We can easily identify vulvar intraepithelial neoplasia (VIN): The patient complains of itching and has a visible lesion. We find cervical intraepithelial neoplasia (CIN) by investigating an abnormal Pap test. But what about vaginal intraepithelial neoplasia (VAIN)? It does not itch and is invisible to the naked eye. A Pap test sometimes catches it, although this test is used mainly to screen for CIN, not VAIN.
VAIN just does not grab our attention. It is uncommon, and invasive vaginal cancer is rare. But before you slip this article into the “obscure disease” file, consider the following:
- VAIN is difficult to diagnose, but some women are at increased risk.
- It is difficult to manage, but understanding the treatment options is important to success.
- The potential for VAIN to evolve into invasive cancer is probably substantial.
- Treatment of invasive vaginal cancer has a high rate of complications and is often unsuccessful.
This article describes the epidemiology, natural history, diagnosis, and treatment of VAIN, focusing primarily on management.
A range of risk factors
As with many uncommon diseases that are difficult to diagnose, good data on the incidence of VAIN are not available. Women diagnosed with VAIN can be in their late teens or senior citizens; mean age is about 50 years.1-5 Race is not mentioned in most studies.
CIN. Although concomitant or subsequent VAIN is very unusual in the approximately 600,000 women identified with CIN each year in the United States, most patients diagnosed with VAIN have a history of CIN. The small number who still have a cervix and are diagnosed with VAIN have a high incidence of concomitant CIN.1 Of women who have had a hysterectomy for CIN, only 1% to 5% are subsequently diagnosed with VAIN.6,7
Since most women who develop vaginal intraepithelial neoplasia have a prior or current history of cervical neoplasia, the “field effect” also renders them at risk for vulvar neoplasia.
HPV. Compelling data indicate a causal relationship between human papillomavirus (HPV) and CIN; the same is true for VAIN.8
Tobacco use. A history of tobacco use is frequent among women diagnosed with VAIN.1
Pelvic radiotherapy is commonly reported in case series.1,3-5 Generally, malignancy is potentially radiation-related if it develops at least 5 to 10 years after treatment.
We do not know whether radiotherapy induces neoplastic transformation in the vagina, but women with a history of radiation warrant long-term follow-up, as they seem to be at increased risk and diagnosis may be difficult.4,5
Chronically immunocompromised women are at particular risk for multifocal lower genital tract neoplasia.9,10
Natural history
The limited data available do suggest that vaginal intraepithelial neoplasia is a premalignant condition.11,12 Unfortunately, little is known about the relationship between severity of the intraepithelial neoplastic process and degree of risk. Natural history studies of VAIN 3 are even more limited than those of CIN 3.
Like its cervical counterpart, VAIN 3 is thought to have substantial potential for progression to invasive cancer.1-4,10-13
EVALUATIONDiagnosis entails inspection, palpation, and directed biopsies
VAIN is most commonly diagnosed after investigation of an abnormal Pap test taken from the vaginal cuff of a woman who has undergone a prior hysterectomy for cervical neoplasia. Occasionally, the disease is identified during colposcopy as extension of a cervical lesion. In either case, VAIN usually involves the upper third of the vagina.1-5,11 A minority of patients will be found to have diffuse multifocal lesions along the vaginal walls.
Adequate diagnosis mandates:
- careful gross and colposcopic inspection of the entire vagina (with mapping of involved areas),
- representative colposcopically directed biopsies,
- careful palpation of the vaginal walls, especially the vaginal cuff scar, and
- in some cases, excision of the vaginal cuff scar.
VAIN is often readily visualized with a colposcope, and the appearance may be more prominent than that of a comparable cervical lesion (FIGURE 1). The lesions are sometimes hyperkeratotic and grossly visible. However, colposcopy of the vagina is more difficult than that of the cervix due to vaginal folding, a larger surface area, and vaginal cuff irregularities.
Vaginal atrophy also creates diagnostic difficulties related to colposcopic assessment, and to overreading of vaginal cytology (eg, when a lesion is interpreted as high-grade dysplasia or suspicious). Reassessment after 4 to 6 weeks of estrogen therapy helps resolve these issues.
FIGURE 1 Colposcopic view of VAIN 3
Note the hyperkeratotic, prominent appearance of the VAIN lesions.
TREATMENT
Because women with VAIN are a heterogeneous group, treatment must be individualized. Important factors to consider when selecting appropriate treatment include prior hysterectomy, prior radiation therapy, age, whether the woman is sexually active, comorbidities, vaginal anatomy, and prior treatments.
Prior hysterectomy
Most women diagnosed with VAIN have previously undergone hysterectomy, usually because of cervical neoplasia.2-5,10 In these women, VAIN is generally confined to the vaginal apex (FIGURE 2).
Watch for occult neoplasia. Buried epithelium within the vaginal cuff scar may harbor occult neoplasia. Patients with VAIN at the vaginal apex5,10,11,13 and a vaginal cuff from a hysterectomy for cervical neoplasia are analogous to women with CIN and an unsatisfactory colposcopy.
Excision is usual management. Most of these women are appropriately managed with excision of the involved vaginal apex, including the cuff scar.1,3-5,11,14 Problematic vaginal shortening is uncommon following this procedure. The status of the resection margin is predictive of the likelihood of recurrence.3
With more extensive vaginal epithelial involvement, consider cuff excision in selected cases to eliminate the potential for occult disease. The remainder of the involved vagina can be treated by other means outlined below.
FIGURE 2 VAIN 3 at vaginal apex
VAIN usually is confined to the apex in women hysterectomized for cervical neoplasia.
When CIN extends onto the vaginal fornix
This is an uncommon scenario, although its underrecognition may explain why some VAIN is diagnosed shortly after hysterectomy for CIN.
Management is simpler than for disease involving the posthysterectomy apex because there is no scarring, distortion, or buried epithelium, and traction on the cervix generally provides good exposure.
Laser vaporization is an option. For many such patients, this is one of the few good indications remaining for laser vaporization.5,15,16
If hysterectomy is indicated, remove the affected portion of the upper vagina along with the cervix.5,17
Multifocal or diffuse pattern: Typical of immunocompromise
Multifocal or diffuse manifestation is likewise uncommon, often involving low-grade neoplasia and condylomatous changes.5,18 This is the pattern typically seen in chronically immunocompromised women.5,10
The natural history of VAIN—especially the malignant potential—is less well understood for women with this disease pattern. When managing such patients, keep in mind the potential for occult neoplasia in the vaginal cuff scar. However, broaden the focus of treatment to encompass the entire vagina. The various management options are described below:
- If the patient has cervical intraepithelial neoplasia 2 or 3, treat with laser, cryotherapy, or large loop excision of the transformation zone at least 2 weeks prior to beginning 5-fluorouracil (5-FU).
- Treat vulvar, anal, or urethral lesions with a laser or local excision prior to 5-FU.
- Treat vulvovaginitis prior to 5-FU.
- Have the patient administer 1/2 vaginal applicator of 5% 5-FU (Efudex; 2.5 g) deep in the vagina weekly at bedtime for 10 weeks. Instruct her to place a tampon or cotton balls into the outer vagina, unless the distal vagina is involved. Also have her apply petroleum jelly to all vulvar tissues, around the urethra, and to the anus. In the morning, she should remove the tampon, wash the vulva with soap and water, and dry carefully. This should be followed by another application of petroleum jelly (sparingly) to vulvar tissues. A mini napkin may help prevent staining of garments.
- When 5-FU treatment ends, have the patient administer 1/3 applicator of vaginal estrogen cream nightly for 2 weeks beginning 3 weeks prior to reevaluation.
- Evaluate with colposcopy and cytology 8 weeks after completing 5-FU therapy.
Observation only is particularly suited for women who have low-grade disease or who are severely debilitated and chronically immunocompromised.
5-fluorouracil (5-FU) cream is a very good treatment when VAIN involves the upper half to two thirds of the vagina.2,9 However, the cream does not reach buried epithelium in the vaginal cuff scar and probably does not consistently treat the lower vagina when applied with the standard vaginal applicator. Applying the cream directly to the lower vagina may be more effective when that region is involved (see “6-step vaginal fluorouracil therapy for intraepithelial neoplasia,” above).
Laser vaporization is well accepted and commonly used.2,9,15,19,20 Advantages are versatility in treatment of multifocal disease without sacrificing vaginal epithelium, and low likelihood of complications. Disadvantages include inability to treat buried vaginal cuff epithelium; technical difficulties in applying the laser to a folding and often distorted (cuff) surface within a confined space; and the expensive equipment, technical support, and surgical expertise required.1,4,5,11,15,16,20
Planned combined treatment using laser vaporization followed by 5-FU cream has been reported efficacious in the treatment of diffuse vaginal condylomata.21 Selected patients with VAIN also may benefit,22 such as those with plaque-like disease (where a thick layer of keratin can reduce penetration of the 5-FU cream), diffuse/multifocal disease where laser vaporization is likely to be incomplete, or vaginal anatomy that makes it difficult to accomplish complete laser vaporization.
Vaginectomy is definitive management for selected patients with extensive VAIN. The operation is done transperineally, although hysterovaginectomy may require a combined approach.23 Leaving the distal third of the vagina intact (when disease distribution allows) makes the operation easier and may help avert iatrogenic urinary incontinence. Follow-up examination—and treatment, if necessary—of the remaining short vaginal stump is fairly easy.
Removal of the vagina is technically demanding in some women. A Schuchardt incision is useful in such instances.
The other obvious disadvantage of vaginectomy is loss of coital function, although placement of a skin graft is an option.
Brachytherapy is another option for treating extensive VAIN in highly selected patients.24,25
A cylindrical apparatus placed in the vagina delivers radiotherapy to the vaginal epithelium; the likelihood of significant morbidity is low. This method is most applicable to poor surgical candidates with extensive VAIN. Disadvantages include fibrosis of the vagina, limited data on efficacy (and particular concern about inadequate dosing to buried or distorted vaginal cuff epithelium), and potential difficulties with follow-up and treatment of recurrence.3,5
“Chemosurgery,” specifically 5-FU cream followed by surgical removal of the then-partially-detached VAIN, followed by additional 5-FU cream, has been used effectively at 1 center.10 Other reported-but-less-investigated methods include cryotherapy, electrocautery, loop electrosurgical excision, and cavitational ultrasonic surgical aspiration (CUSA).3-5
VAIN in a radiated vagina
Most women who develop VAIN in this scenario received radiation therapy many years earlier for carcinoma of the cervix.5,11
The most common sites are the upper third to upper half of the remaining vagina, where radiation changes are prominent.
Diagnosis often is problematic due to:
- difficulties with interpreting cytologic preparations in such patients;
- radiation changes in the vagina (pale and fibrotic with telangiectasis), which largely obscure colposcopic findings;
- the difficulty and potential hazards of biopsy of a thin, fibrotic upper vagina; and
- obliterative coaptation of the upper third to half of the vagina.
Significantly abnormal cytology in the absence of a colposcopically identified lesion (or palpable abnormality) is of particular concern. In such patients, view the abnormal cytologic interpretation with caution and consider further initial evaluation, including outside review of the cytology slides, treatment of the vagina with estrogen, and repeat cytologic and colposcopic evaluation.
When there is clear cytologic evidence of a severe abnormality, consider the possibility of occult neoplasia within the coapted upper vagina and/or cervix.
Management of VAIN in a radiated vagina.
Take into account the anatomic distortion of the upper vagina, the thin and fibrotic nature of the epithelium, and the potential for fistula formation with excisional procedures or other treatments that produce injury beneath the surface of the epithelium. Among the options:
- 5-FU cream or laser vaporization. VAIN that is completely visualized within the remaining vagina is probably best treated with one of these modalities, provided the physician is experienced in managing such patients.5,9
- Excision of the upper vagina,5 including the coapted portion (and sometimes the residual cervix with or without the uterus) is appropriate for highly selected cases, but only after careful consideration of:
- the likelihood of finding significant neoplasia,
- the anatomic feasibility (it is desirable to perform the procedure transvaginally), and
- the overall risk versus benefit. Such procedures should be performed only by an experienced physician.
The chronically immunocompromised
These patients often have undergone organ transplants or are human immunodeficiency virus (HIV)-positive. Diffuse HPV infection of the lower genital tract is pervasive in these women,5,10,26 and diffuse/multifocal lower genital tract intraepithelial neoplasia is often present as well.
These women may be at increased risk for progression to invasive cancer.
Eradication may not be possible. Attempts to eliminate intraepithelial disease are usually unsuccessful.
Management. Many of these women are severely debilitated, with other, more significant medical problems and a short life expectancy. Vigilance is required, as other lower genital tract (and anal) sites are frequently involved. Since eradication of diffuse/multifocal intraepithelial disease is not a realistic goal, treatment followed by chronic suppressive therapy (such as a low intermittent dose of 5-FU) is reasonable.9,10,22
Another approach is close observation, including frequent examinations, with prompt intervention when invasive disease is suspected.
FOLLOW-UP
After treatment for VAIN, follow-up is similar to that for a comparable cervical lesion. Once the vagina has healed, see the patient every 3 to 6 months for 2 years and annually thereafter. In addition to obtaining vaginal cytology, carefully inspect and palpate the vagina (including the vaginal cuff scar).
Effects of 5-FU. Following a course of 5-FU cream, vaginal mucosal ulcerations may persist for several weeks. Occasionally, these may lead to partial coaptations.27 In addition, subsequent islands of columnar epithelium have been described.28 Keep these factors in mind during the follow-up of women who have been treated with transvaginal 5-FU cream.
Long-term annual follow-up is reasonable, since these women are probably at increased risk for developing a second primary vaginal lesion and/or lower anogenital tract neoplasia at other sites.
When extra vigilance is warranted. Some patients merit closer follow-up, such as the chronically immunosuppressed women described; also, women who have been treated for vaginal apical disease without resection of the vaginal cuff scar, previously radiated patients, and women whose VAIN was treated with brachytherapy.
Dr. Hoffman reports no financial relationships relevant to this article.
1. Dodge JA, Eltabbakh GH, Mount SL, Walker RP, Morgan A. Clinical features and risk of recurrence among patients with vaginal intraepithelial neoplasia. Gynecol Oncol. 2001;83:363-369.
2. Petrilli ES, Townsend DE, Morrow CP, Nakao CY. Vaginal intraepithelial neoplasia: biologic aspects and treatment with topical 5-fluorouracil and the carbon dioxide laser. Am J Obstet Gynecol. 1980;138:321-328.
3. Benedet JL, Sanders BH. Carcinoma in situ of the vagina. Am J Obstet Gynecol. 1984;148:695-700.
4. Lenehan PM, Meffe F, Lickrish GM. Vaginal intraepithelial neoplasia: biologic aspects and management. Obstet Gynecol. 1986;68:333-337.
5. Audet-Lapointe P, Body G, Vauclair R, Drouin P, Ayoub J. Vaginal intraepithelial neoplasia. Gynecol Oncol. 1990;36:232-239.
6. Gallup DG, Morley GW. Carcinoma in situ of the vagina—a study and review. Obstet Gynecol. 1975;46:334-440.
7. Woodruff JD. Treatment of recurrent carcinoma in situ in the lower genital canal. Clin Obstet Gynecol. 1965;8:757-770.
8. McCance DJ, Clarkson PK, Dyson JL, Walker PG, Singer A. Human papillomavirus types 6 and 16 in multifocal intraepithelial neoplasias of the female lower genital tract. Br J Obstet Gynaecol. 1985;92:1093-1100.
9. Krebs HB. Treatment of vaginal intraepithelial neoplasia with laser and topical 5-fluorouracil. Obstet Gynecol. 1989;73:657-660.
10. Sillman FH, Fruchter RG, Chen YS, Camilien L, Sedlis A, McTigue E. Vaginal intraepithelial neoplasia: risk factors for persistence, recurrence, and invasion and its management. Am J Obstet Gynecol. 1997;176:93-99.
11. Woodman CBJ, Jordan JA, Wade-Evans T. The management of vaginal intraepithelial neoplasia after hysterectomy. Br J Obstet Gynaecol. 1984;91:103-105.
12. Aho M, Vesterinen E, Meyer B, Purola E, Paavonen J. Natural history of vaginal intraepithelial neoplasia. Cancer. 1991;68:195-197.
13. Hoffman MS, Roberts WS, LaPolla JP, Sterghos S, Jr, Cavanagh D. Neoplasia in vaginal cuff epithelial inclusion cysts after hysterectomy. J Reprod Med. 1989;34:412-416.
14. Hoffman MS, DeCesare SL, Roberts WS, Fiorica JV, Finan MA, Cavanagh D. Upper vaginectomy for in situ and occult, superficially invasive carcinoma of the vagina. Am J Obstet Gynecol. 1992;166:30-33.
15. Curtin JP, Twiggs LB, Julian TM. Treatment of vaginal intraepithelial neoplasia with the CO2laser. J Reprod Med. 1985;30:942-944.
16. Hoffman MS, Roberts WS, LaPolla JP, Fiorica JV, Cavanagh D. Laser vaporization of grade 3 vaginal intraepithelial neoplasia. Am J Obstet Gynecol. 1991;165:1342-1344.
17. Hoffman MS, Spellacy WN. The Difficult Vaginal Hysterectomy. New York, NY: Springer-Verlag; 1995;117-119.
18. Reid R. Human papillomaviral infection. The key to rational triage of cervical neoplasia. Obstet Gynecol Clin North Am. 1987;14:407-429.
19. Townsend DE, Levine RU, Crum CP, Richart RM. Treatment of vaginal carcinoma in situ with the carbon dioxide laser. Am J Obstet Gynecol. 1982;143:565-568.
20. Sherman AI. Laser therapy for vaginal intraepithelial neoplasia after hysterectomy. J Reprod Med. 1990;35—941-944.
21. Krebs HB. Combination of laser plus 5-fluorouracil for the treatment of extensive genital condylomata acuminata. Lasers Surg Med. 1988;8:135-138.
22. Krebs HB. Prophylactic topical 5-fluorouracil following treatment of human papillomavirus-associated lesions of the vulva and vagina. Obstet Gynecol. 1986;68:837-841.
23. Hoffman MS, Bomalaski JJ, Lockhart JL, Greenwald DP. Total removal of the normal non-prolapsed vagina. Technique, results and anatomic observations. J Pelvic Surg. 2002;8:89-94.
24. MacLeod C, Fowler A, Dalrymple C, Atkinson K, Elliott P, Carter J. High-dose-rate brachytherapy in the management of high-grade intraepithelial neoplasia of the vagina. Gynecol Oncol. 1997;65:74-77.
25. Teruya Y, Sakumoto K, Moromizato H, et al. High dose-rate intracavitary brachytherapy for carcinoma in situ of the vagina occurring after hysterectomy: a rational prescription of radiation dose. Am J Obstet Gynecol. 2002;187:360-364.
26. Sillman FH, Sedlis A. Anogenital HPV/neoplasia in immunosuppressed women: an update. Dermatol Clin North Am. 1991;9:353-369.
27. Krebs HB, Helmkamp BF. Chronic ulcerations following topical therapy with 5-fluorouracil for vaginal human papillomavirus-associated lesions. Obstet Gynecol. 1991;78:205-212.
28. Dungar CF, Wilkinson EJ. Vaginal columnar cell metaplasia: an acquired adenosis associated with topical 5-FU therapy. J Reprod Med. 1995;40:361-366.
1. Dodge JA, Eltabbakh GH, Mount SL, Walker RP, Morgan A. Clinical features and risk of recurrence among patients with vaginal intraepithelial neoplasia. Gynecol Oncol. 2001;83:363-369.
2. Petrilli ES, Townsend DE, Morrow CP, Nakao CY. Vaginal intraepithelial neoplasia: biologic aspects and treatment with topical 5-fluorouracil and the carbon dioxide laser. Am J Obstet Gynecol. 1980;138:321-328.
3. Benedet JL, Sanders BH. Carcinoma in situ of the vagina. Am J Obstet Gynecol. 1984;148:695-700.
4. Lenehan PM, Meffe F, Lickrish GM. Vaginal intraepithelial neoplasia: biologic aspects and management. Obstet Gynecol. 1986;68:333-337.
5. Audet-Lapointe P, Body G, Vauclair R, Drouin P, Ayoub J. Vaginal intraepithelial neoplasia. Gynecol Oncol. 1990;36:232-239.
6. Gallup DG, Morley GW. Carcinoma in situ of the vagina—a study and review. Obstet Gynecol. 1975;46:334-440.
7. Woodruff JD. Treatment of recurrent carcinoma in situ in the lower genital canal. Clin Obstet Gynecol. 1965;8:757-770.
8. McCance DJ, Clarkson PK, Dyson JL, Walker PG, Singer A. Human papillomavirus types 6 and 16 in multifocal intraepithelial neoplasias of the female lower genital tract. Br J Obstet Gynaecol. 1985;92:1093-1100.
9. Krebs HB. Treatment of vaginal intraepithelial neoplasia with laser and topical 5-fluorouracil. Obstet Gynecol. 1989;73:657-660.
10. Sillman FH, Fruchter RG, Chen YS, Camilien L, Sedlis A, McTigue E. Vaginal intraepithelial neoplasia: risk factors for persistence, recurrence, and invasion and its management. Am J Obstet Gynecol. 1997;176:93-99.
11. Woodman CBJ, Jordan JA, Wade-Evans T. The management of vaginal intraepithelial neoplasia after hysterectomy. Br J Obstet Gynaecol. 1984;91:103-105.
12. Aho M, Vesterinen E, Meyer B, Purola E, Paavonen J. Natural history of vaginal intraepithelial neoplasia. Cancer. 1991;68:195-197.
13. Hoffman MS, Roberts WS, LaPolla JP, Sterghos S, Jr, Cavanagh D. Neoplasia in vaginal cuff epithelial inclusion cysts after hysterectomy. J Reprod Med. 1989;34:412-416.
14. Hoffman MS, DeCesare SL, Roberts WS, Fiorica JV, Finan MA, Cavanagh D. Upper vaginectomy for in situ and occult, superficially invasive carcinoma of the vagina. Am J Obstet Gynecol. 1992;166:30-33.
15. Curtin JP, Twiggs LB, Julian TM. Treatment of vaginal intraepithelial neoplasia with the CO2laser. J Reprod Med. 1985;30:942-944.
16. Hoffman MS, Roberts WS, LaPolla JP, Fiorica JV, Cavanagh D. Laser vaporization of grade 3 vaginal intraepithelial neoplasia. Am J Obstet Gynecol. 1991;165:1342-1344.
17. Hoffman MS, Spellacy WN. The Difficult Vaginal Hysterectomy. New York, NY: Springer-Verlag; 1995;117-119.
18. Reid R. Human papillomaviral infection. The key to rational triage of cervical neoplasia. Obstet Gynecol Clin North Am. 1987;14:407-429.
19. Townsend DE, Levine RU, Crum CP, Richart RM. Treatment of vaginal carcinoma in situ with the carbon dioxide laser. Am J Obstet Gynecol. 1982;143:565-568.
20. Sherman AI. Laser therapy for vaginal intraepithelial neoplasia after hysterectomy. J Reprod Med. 1990;35—941-944.
21. Krebs HB. Combination of laser plus 5-fluorouracil for the treatment of extensive genital condylomata acuminata. Lasers Surg Med. 1988;8:135-138.
22. Krebs HB. Prophylactic topical 5-fluorouracil following treatment of human papillomavirus-associated lesions of the vulva and vagina. Obstet Gynecol. 1986;68:837-841.
23. Hoffman MS, Bomalaski JJ, Lockhart JL, Greenwald DP. Total removal of the normal non-prolapsed vagina. Technique, results and anatomic observations. J Pelvic Surg. 2002;8:89-94.
24. MacLeod C, Fowler A, Dalrymple C, Atkinson K, Elliott P, Carter J. High-dose-rate brachytherapy in the management of high-grade intraepithelial neoplasia of the vagina. Gynecol Oncol. 1997;65:74-77.
25. Teruya Y, Sakumoto K, Moromizato H, et al. High dose-rate intracavitary brachytherapy for carcinoma in situ of the vagina occurring after hysterectomy: a rational prescription of radiation dose. Am J Obstet Gynecol. 2002;187:360-364.
26. Sillman FH, Sedlis A. Anogenital HPV/neoplasia in immunosuppressed women: an update. Dermatol Clin North Am. 1991;9:353-369.
27. Krebs HB, Helmkamp BF. Chronic ulcerations following topical therapy with 5-fluorouracil for vaginal human papillomavirus-associated lesions. Obstet Gynecol. 1991;78:205-212.
28. Dungar CF, Wilkinson EJ. Vaginal columnar cell metaplasia: an acquired adenosis associated with topical 5-FU therapy. J Reprod Med. 1995;40:361-366.
Fecal incontinence: Current strategies for a debilitating disease
- Endoanal ultrasound is superior to other diagnostic methods, with an accuracy of over 90% in detecting sphincter injuries, compared to 75% for manometry, 75% for electromyography, and 50% for clinical assessment.
- Pelvic floor rehabilitation with biofeedback/electrical stimulation is the mainstay nonsurgical treatment. Some centers report success rates as high as 70%.
- Consider surgery only for distinct anatomic defects, and only when nonsurgical interventions have not succeeded.
Thanks to improvements in our understanding of the causes of uncontrolled defecation and an increasing array of diagnostic and treatment options, physicians can now manage this condition more effectively.
Fecal incontinence is a relevant clinical issue for Ob/Gyns, and requires compassion and emotional support on the part of the physician and health-care team. This debilitating problem is underreported, but is thought to affect 2.2% of the general population. Women are 8 times more likely to have fecal incontinence than men, in age-controlled groups.1
Although sphincter damage during vaginal delivery—even in deliveries over an intact perineum2,3—may be clinically silent in most women, these women are at risk of developing overt fecal incontinence with subsequent deliveries, and with aging and its decompensating factors (FIGURE 1).
A directed examination, optimal diagnostic testing, and a step-by-step treatment algorithm are the basis of effective management.
FIGURE 1 Common pathway leading to fecal incontinence
How ‘silent’ sphincter damage can lead to incontinence
Obstetric disruption of the anal sphincter structure or function is a common cause of fecal incontinence, although it is usually multifactorial, involving abnormalities of 1 or more continence mechanisms (FIGURE 2).
Weakening of the sphincter mechanism after vaginal delivery is attributed to both direct trauma to the muscle and indirect trauma to the pudendal nerve (unilateral or bilateral). Anatomic injury usually occurs in the anterior portion of the sphincter—a site amenable to direct repair, with good results.
Of the various investigational diagnostic modalities available, endoanal ultrasound has been superior, though not perfect. In 1 series, endoanal sonography detected anal sphincter tears in 35% of primiparous and 40% of multiparous women; of these, 13% and 23%, respectively, had symptoms of fecal incontinence or urgency.4
The pudendal nerve is susceptible to traction injury from childbirth and chronic straining. In a prospective study of the effect of vaginal deliveries on the pelvic floor, 80% of primigravidas sustained reversible pudendal nerve damage—which, in some women, persisted for up to 5 years.5
Obstetric risk factors include:
- infant weighing more than 4,000 g,
- use of forceps,
- prolonged second stage of labor,
- midline episiotomy,
- occipitoposterior position, and
- failed detection or suboptimal repair of anal sphincterinjuryat the time of vaginaldelivery.
Onset of menopause and subsequent pelvic floor weakness and perineal descent aggravates this occult neuropathy.
Minor incontinence is also common in patients with advanced age, internal sphincter defects, prolapsing hemorrhoids, rectal prolapse, and fecal impaction (the rectum chronically distends with feces, continuously inducing internal anal sphincter relaxation and resulting in leakage of stool).
Evaluation: Directed history, 4-point physical exam
A directed history helps indicate which diagnostic tests are most relevant. Establish severity and frequency of fecal incontinence, presence of fecal urgency, pad use, type of incontinence (solid, liquid, or gas), and presence of associated urinary incontinence and prolapse.
Obstetric history should identify:
- parity,
- duration of the second stage of labor,
- occipitoposterior presentation,
- use of forceps,
- extent of vaginal lacerations, and
- weight of the infant.
Dietary habits (especially fiber intake) and chronic laxative use should be noted.
Also note any history of abdominopelvic surgery, radiation, back injury, and neurologic disorders.
4-point physical exam
- Inspect the perineum/perianal area for evidence of dermatitis, infection, fistula, hemorrhoids, and deformities of the anus. Ask the patient to bear down as you look for evidence of pelvic floor weakness and rectal prolapse.
- Assess the anal wink (cutaneoanal contractile reflex) by stroking the skin surrounding the anus. Absence of a reflexive contraction of the external anal sphincter suggests nerve damage or spinal arc interruption.
- Perform a vaginal exam to identify pelvic prolapse and exclude rectovaginal fistula. At this time assess pelvic muscle strength and teach the patient proper pelvic floor exercises.
- Conduct a digital rectal examination to assess the length and orientation of the analcanal, bulk of the perianal tissues, and tone of the anal sphincters, as well as to rule out a mass or impaction. Repeat digital examination with the patient bearing down to assess the squeeze pressures, movement of the puborectalis muscle, and extent of pelvic floor descent.
These findings are predominantly subjective and depend on operator experience. In general, they correlate poorly with objective measurements of sphincter pressures.6
If a rectovaginal fistula is suspected, try retrograde filling of the rectum with 200 cc of dye-stained fluid (using a 50-cc catheter tip syringe), then observe vaginal extravasation.
Diagnostic strategy: Routine endoanal ultrasound, manometry
Our strategy is to routinely proceed with endoanal ultrasound and anal manometry to assess the anal sphincters. We may then recommend sigmoidoscopy or anoscopy to rule out mucosal lesions.
It is important to bear in mind that continence depends on multiple physiologic mechanisms and, therefore, no single diagnostic test yields positive results in all patients.
Endoanal ultrasound has significantly changed the evaluation of fecal incontinence over the last decade. In women with fecal incontinence in whom obstetric injury is suspected, 90% have sonographic evidence of injury to 1 or both sphincters.
These ultrasound images show 2 discrete rings of tissue: The inner hypoechoic ring represents the internal anal sphincter and the outer hyperechoic ring represents the external anal sphincter (FIGURE 3). Disrupted continuity of these rings is consistent with structural damage to the sphincter.
Endoanal ultrasound is regarded as superior to other diagnostic methods, with an accuracy of over 90% in detecting sphincter injuries, compared to 75% for manometry, 75% for electromyography (EMG), and 50% for clinical assessment.7,8 It is the simplest and least-invasive test to assess the structural integrity of the anal sphincters.
It can also serve as a screening tool to detect occult sphincter injury following vaginal delivery, thus identifying women at high risk for future incontinence. Women with transient stool or gas incontinence following vaginal delivery may be candidates for screening endoanal ultrasound for further evaluation.
- Where endoanal ultrasound is unavailable, transperineal ultrasound may be an alternative option.
Anorectal manometry can detect functional weakness of sphincters that are anatomically intact by measuring sensation and squeeze pressures. Patients with intact sphincters but abnormal anal manometry findings may benefit from physical therapy and biofeedback aimed at increasing muscular function by stimulating the neuromuscular feedback loop.
During manometry, the physician assesses maximum resting and squeeze pressures of the anal canal, the rectoanal inhibitory reflex, rectal sensation, and rectal compliance using either water-perfused catheters, solid-state microtransducers, or air-or water-filled balloons. Normal values vary widely among institutions and are poorly standardized.
Decreased resting pressure suggests isolated internal anal sphincter injury; decreased squeeze pressure suggests external anal sphincter injury. Of all parameters, maximum squeeze pressure has the greatest sensitivity and specificity at a cut-off of 60 mm Hg in women.9
Rectal sensory testing also is useful in identifying patients who may benefit from biofeedback therapy, and can detect afferent nerve injury as a contributing cause of incontinence. Biofeedback is unlikely to be useful in patients with a poor or absent sensation. In addition, for patients already involved in biofeedback, the test can assess rectal response to training and indicate rectal irritability or poor compliance.
Rectal sensation is assessed by inflating a balloon in the rectum and recording the smallest volume of rectal distention for first detectable sensation (rectal sensory threshold), sensory urgency, and pain (maximum tolerable volume).
Ancillary tests are of limited benefit as screening tools, but may have utility in special circumstances.
- Electromyography of the external anal sphincter and the pelvic floor muscles—traditionally performed using needle electrodes or surface electrodes—is helpful in delineating areas of sphincter injury by mapping the sphincter. However, much of this information is now obtained accurately by endoanal ultrasound, which has replaced needle EMG for most clinicians. Surface EMG is still useful for evaluating sphincter function and in biofeedback training.
- Defecography involves imaging the rectum after filling it with contrast material, and then observing the process, rate, and completeness of rectal evacuation using fluoroscopy. Its usefulness in the evaluation of fecal incontinence is limited to cases of rectal prolapse and for the diagnosis of rectocele and enterocele.
- Investigational: Pudendal nerve testing. This test uses an electrode to measure pudendal nerve conduction time, known as pudendal nerve terminal motor latency (PNTML)—thus allowing further investigation for nerve injury.
- Normal PNTML value is 2.2 milliseconds.
- A value between 2.2 and 2.6 milliseconds indicates probable nerve damage.
- A value of 2.6 milliseconds or greater confirms nerve damage.
Clinical use of PNTML is controversial; it helps diagnose nerve injury, but is currently reserved for investigational purposes.10
FIGURE 3 Endoanal ultrasound
Disruption of continuity of the inner hypoechoic ring (IS, internal sphincter) and the outer hyperechoic ring (ES, external sphincter) on endoanal ultrasound is consistent with structural damage to the sphincter. This image shows normal anatomy.
Mainstays: Nonsurgical treatments
The general principles of treatment are to identify the underlying cause of incontinence and direct therapy based on these findings. Since much of the disability in these patients is psychological, a supportive medical team is essential. Patient educational books, advice on the use of continent aides, and perianal skin care also are crucial.
The physician and patient must formulate a treatment plan based on the severity of the incontinence and degree of social impairment. In general, a trial of nonsurgical therapy is recommended before proceeding to surgery. A standardized treatment algorithm (FIGURE 4) should be adapted to each patient.
The aim of nonsurgical management is:
- to increase anorectal sensation, and
- to increase the strength of the external sphincter and pelvic floor.
First: Diet and medical therapy. For patients with fecal incontinence, nonsurgical intervention remains the first line of treatment given the high rate of underlying neuromuscular damage.
The aims of medical therapy are to decrease stool frequency and improve stool consistency. The patient’s diet should include adequate fiber and fluid intake to promote regular, soft, bulky stools. High-fiber foods including whole-wheat grains, fresh vegetables, and beans should be advised. Bulking agents like methylcellulose (eg, Citrucel), or psyllium (eg, Metamucil) can be helpful.
Stool frequency can be reduced with antidiarrheal drugs like loperamide (2-4 mg 2 or 3 times daily) or diphenoxalate (eg, Lomotil). Loperamide is usually preferred, since it also increases internal anal sphincter tone. Antiflatulants are also recommended.
Amitryptiline is useful in some women with idiopathic fecal incontinence.11,12
Biofeedback therapy. Pelvic floor rehabilitation with biofeedback and/or electrical stimulation—which focuses on the common neuromuscular etiology of fecal incontinence—is the mainstay of nonsurgical management. Some centers report a success rate as high as 70% with this therapy.13
Biofeedback is a painless, minimally invasive method of retraining pelvic floor muscles with the help of sensors placed in either the vagina or the rectum. This technique improves rectal sensation and muscular tone. It is especially useful in incontinence patients with anatomically intact anal sphincters.
Biofeedback typically involves anal manometry as well as electromyographic sensors. It is generally conducted in an outpatient treatment program that includes regular evaluation of sensation and motor tone. However, home-based units are available.
Investigational: Neuromodulation. Although approved by the US Food and Drug Administration for treatment of urge incontinence and nonobstructive voiding dysfunction, neuromodulation is still investigational for treatment of fecal incontinence—but results are encouraging.14
In this technique, electrodes attached to a portable stimulator intermittently apply low-level electrical impulses to the anal canal via the pelvic nerve supply. Newer methods involve implanting electrodes percutaneously in the S3 or S4 foramina under fluoroscopic guidance, then stimulating the sacral nerves.
Neuromodulation aims to increase maximum squeeze pressure and decrease rectal sensitivity to distention. In early trials, significant improvement of incontinence and urgency symptoms was noted.15,16 Larger and longer-duration trials are needed to elucidate long-term effectiveness.
When to consider surgery
Operative therapy should be considered only for cases in which nonsurgical interventions have failed or in which distinct anatomic defects are present.
Note that most surgical candidates still require physical therapy with biofeedback before or after the procedure.
Multiple surgical procedures have been described; some are technically challenging and should be performed only by skilled surgeons after careful preoperative counseling.
Anterior overlapping external sphincteroplasty is done in patients with obstetric or iatrogenic disruption of the external anal sphincter. In this procedure, the surgeon divides the scar and reapproximates viable sphincter muscle to produce a ring of functional muscle. Few good data exist comparing this approach with end-to-end repairs; anterior sphincteroplasty is our preferred method.
- Controversy exists over the need to plicate the internal anal sphincter. Studies have shown that more extensive repairs offer no additional advantage with regard to out-comes.17,18 Most pelvic reconstructive surgeons prefer to perform colporrhaphy and perineorrhaphy at the time of sphincter repair, since defects in the posterior compartment often coexist in these patients.
The success rate of sphincteroplasty ranges from 40% to 75% in appropriately selected patients.19,20
Patients with satisfactory results demonstrate an increase in squeeze pressure postoperatively. Failure to elicit this response correlated accurately with a persistent sphincter defect.21
Total pelvic floor repair may help patients with neurogenic or idiopathic fecal incontinence. The principle of this procedure is to restore the anorectal angle, lengthen the anal canal, and create a circumferential buttress around the anorectum.
In this rather extensive surgery, the surgeon plicates the puborectalis, ischiococcygeus, and iliococcygeus muscles to the rectum; the levator muscles and the external anal sphincter are plicated anteriorly. Success rates vary from 14% to 55%.22,23
Skeletal muscle transposition surgery, with creation of a neosphincter from the gracilis muscle, is performed in patients with pelvic floor muscle weakness so profound that the above techniques offer no improvement. It should be viewed as a salvage operation.
The gracilis muscle is detached from its insertion, wrapped around the anus, and reattached to the contralateral ischial tuberosity. The success of this surgery may be improved by electrically stimulating the transposed muscle with an implanted stimulator.
This procedure is associated with significant postoperative complications; many patients require multiple reoperations. Thus, this intervention is useful only in patients with refractory incontinence.
Artificial anal sphincter procedure involves placing a cuff around the anal canal and a pressure-regulating balloon and pump in the labia majora. The cuff remains inflated, thereby occluding the anal canal until the patient experiences a desire to defecate, at which time it is deflated.
Results are encouraging, with initial success rates from 60% to 75%.24,25 However, local infection can be problematic.
Less common procedures include anal encirclement (Thiersch procedure), perianal fat injection, antegrade continence enema, and fecal diversion.
A word on prevention
During childbirth. Strategies that may reduce pelvic floor trauma:
- Favor mediolateral over midline episiotomy,
- avoid a prolonged second stage of labor and forceps delivery, and
- repair anal sphincter lacerations with an overlapping technique.
Also, discuss regular pelvic floor exercises, constipation prevention, weight loss, and smoking cessation with all obstetric patients—especially those with fecal incontinence risk factors.
Debate continues about elective cesarean section in women with occult sphincter injury on postpartum endoanal ultrasound, transient symptoms of fecal incontinence with prior delivery, or overt gas incontinence.26 This practice may have clinical utility, but data are insufficient to recommend it routinely.
The authors report no financial relationships relevant to this article.
1. Furner S, Cautley E, Norton N, Nelson R. Community-based prevalence of anal incontinence. JAMA. 1995;274:559.-
2. Sultan AH, Kamm MA, Hudson CN, et al. Anal-sphincter disruption during vaginal delivery. N Engl J Med. 1993;329:1905.-
3. Abramowitz L, Sobhani I, Ganansia R, et al. Are sphincter defects the cause of anal incontinence after vaginal delivery? Results of a prospective study. Dis Colon Rectum. May 2000;43:590-596; discussion:596–598.
4. Fynes M, Donnelly VS, O’Connell PR, O’Herlihy C. Cesarean delivery and anal sphincter injury. Obstet Gynecol. 1998;92:496-500.
5. Haadem K, Dahlstrom JA, Lingman G. Anal sphincter function after delivery: a prospective study in women with sphincter rupture and controls. Eur J Obstet Gynecol Reprod Biol. 1990;35:7.-
6. Coller JA. Clinical application of anorectal manometry. Gastroenterol Clin North Am. 1987;16:17.-
7. Sultan AH, Kamm MA, Talbot IC, et al. Anal endosonography for identifying external sphincter defects confirmed histologically. Br J Surg. 1994;81:463.-
8. Tjandra JJ, Milsom JW, Schroeder T, Fazio VW. Endoluminal ultrasound is preferable to electromyography in mapping anal sphincteric defects. Dis Colon Rectum. 1993;36:689-692.
9. Felt-Bersma RJF, Klinkenberg-Knol EC, Meuwissen SGM. Anorectal function investigations in incontinent and continent patients. Dis Colon Rectum. 1990;33:479.-
10. Barnett JL, Hasler WL, Camilleri M. American Gastroenterological Association medical position statement on anorectal testing techniques. American Gastroenterological Association. Gastroenterology. 1999;116:732.-
11. Santoro GA, Eitan BZ, Pryde A, Bartolo DC. Open study of low-dose amitriptyline in the treatment of patients with idiopathic fecal incontinence. Dis Colon Rectum. 2000;43:1676.-
12. Read M, Read NW, Barber DC, Duthie HL. Effects of loperamide on anal sphincter function in patients complaining of chronic diarrhea with fecal incontinence and urgency. Dig Dis Sci. 1982;27:807.-
13. Ryn AK, Morren GL, Hallbook O, Sjodahl R. Long-term results of electromyo-graphic biofeedback training for fecal incontinence. Dis Colon Rectum. 2000;43:1262-1266
14. Matzel KE, Stadelmaier U, Hohenfellner M, Gall FP. Electrical stimulation of sacral spinal nerves for treatment of faecal incontinence. Lancet. 1995;346:1124.-
15. Matzel KE, Stadelmaier U, Hohenfellner M, Hohenberger W. Chronic sacral spinal nerve stimulation for fecal incontinence: long-term results with foramen and cuff electrodes. Dis Colon Rectum. 2001;44:59.-
16. Rosen HR, Urbarz C, Holzer B, et al. Sacral nerve stimulation as a treatment for fecal incontinence. Gastroenterology. 2001;121:536.-
17. Briel JW, de Boer LM, Hop WC, Schouten WR. Clinical outcome of anterior overlapping external anal sphincter repair with internal anal sphincter imbrication. Dis Colon Rectum. 1998;41:209-214.
18. Laurberg S, Swash M, Henry MM. Effect of postanal repair on progress of neurogenic damage to the pelvic floor. Br J Surg. 1990;77:519-522.
19. Kamm MA. Faecal incontinence. BMJ. 1998;316:528.-
20. Malouf AJ, Norton CS, Engel AF, et al. Long-term results of overlapping anterior anal-sphincter repair for obstetric trauma. Lancet. 2000;355:260.-
21. Barnett JL, Hasler WL, Camilleri M. American Gastroenterological Association medical position statement on anorectal testing techniques. American Gastroenterological Association. Gastroenterology. 1999;116:732.-
22. Orrom WJ, Miller R, Cornes H, et al. Comparison of anterior sphincteroplasty and postanal repair in the treatment of idiopathic fecal incontinence. Dis Colon Rectum. 1991;34:305-310.
23. Schiller, Lawrence R. Fecal incontinence. In: Feldman M, et al, eds. Sleisenger & Fordtran’s Gastrointestinal and Liver Disease. Philadelphia, Pa: WB Saunders Co; 1997.
24. Giamundo P, Welber A, Weiss EG, et al. The Procon incontinence device: a new nonsurgical approach to preventing episodes of fecal incontinence. Am J Gastroenterol. 2002;97:232.-
25. Wong WD, Congliosi SM, Spencer MP, et al. The safety and efficacy of the artificial bowel sphincter for fecal incontinence: results from a multicenter cohort study. Dis Colon Rectum. 2002;45:1139.-
26. McKenna DS, Ester JB, Fischer JR. Elective cesarean delivery for women with a previous anal sphincter rupture. Am J Obstet Gynecol. 2003;189:1251.-
- Endoanal ultrasound is superior to other diagnostic methods, with an accuracy of over 90% in detecting sphincter injuries, compared to 75% for manometry, 75% for electromyography, and 50% for clinical assessment.
- Pelvic floor rehabilitation with biofeedback/electrical stimulation is the mainstay nonsurgical treatment. Some centers report success rates as high as 70%.
- Consider surgery only for distinct anatomic defects, and only when nonsurgical interventions have not succeeded.
Thanks to improvements in our understanding of the causes of uncontrolled defecation and an increasing array of diagnostic and treatment options, physicians can now manage this condition more effectively.
Fecal incontinence is a relevant clinical issue for Ob/Gyns, and requires compassion and emotional support on the part of the physician and health-care team. This debilitating problem is underreported, but is thought to affect 2.2% of the general population. Women are 8 times more likely to have fecal incontinence than men, in age-controlled groups.1
Although sphincter damage during vaginal delivery—even in deliveries over an intact perineum2,3—may be clinically silent in most women, these women are at risk of developing overt fecal incontinence with subsequent deliveries, and with aging and its decompensating factors (FIGURE 1).
A directed examination, optimal diagnostic testing, and a step-by-step treatment algorithm are the basis of effective management.
FIGURE 1 Common pathway leading to fecal incontinence
How ‘silent’ sphincter damage can lead to incontinence
Obstetric disruption of the anal sphincter structure or function is a common cause of fecal incontinence, although it is usually multifactorial, involving abnormalities of 1 or more continence mechanisms (FIGURE 2).
Weakening of the sphincter mechanism after vaginal delivery is attributed to both direct trauma to the muscle and indirect trauma to the pudendal nerve (unilateral or bilateral). Anatomic injury usually occurs in the anterior portion of the sphincter—a site amenable to direct repair, with good results.
Of the various investigational diagnostic modalities available, endoanal ultrasound has been superior, though not perfect. In 1 series, endoanal sonography detected anal sphincter tears in 35% of primiparous and 40% of multiparous women; of these, 13% and 23%, respectively, had symptoms of fecal incontinence or urgency.4
The pudendal nerve is susceptible to traction injury from childbirth and chronic straining. In a prospective study of the effect of vaginal deliveries on the pelvic floor, 80% of primigravidas sustained reversible pudendal nerve damage—which, in some women, persisted for up to 5 years.5
Obstetric risk factors include:
- infant weighing more than 4,000 g,
- use of forceps,
- prolonged second stage of labor,
- midline episiotomy,
- occipitoposterior position, and
- failed detection or suboptimal repair of anal sphincterinjuryat the time of vaginaldelivery.
Onset of menopause and subsequent pelvic floor weakness and perineal descent aggravates this occult neuropathy.
Minor incontinence is also common in patients with advanced age, internal sphincter defects, prolapsing hemorrhoids, rectal prolapse, and fecal impaction (the rectum chronically distends with feces, continuously inducing internal anal sphincter relaxation and resulting in leakage of stool).
Evaluation: Directed history, 4-point physical exam
A directed history helps indicate which diagnostic tests are most relevant. Establish severity and frequency of fecal incontinence, presence of fecal urgency, pad use, type of incontinence (solid, liquid, or gas), and presence of associated urinary incontinence and prolapse.
Obstetric history should identify:
- parity,
- duration of the second stage of labor,
- occipitoposterior presentation,
- use of forceps,
- extent of vaginal lacerations, and
- weight of the infant.
Dietary habits (especially fiber intake) and chronic laxative use should be noted.
Also note any history of abdominopelvic surgery, radiation, back injury, and neurologic disorders.
4-point physical exam
- Inspect the perineum/perianal area for evidence of dermatitis, infection, fistula, hemorrhoids, and deformities of the anus. Ask the patient to bear down as you look for evidence of pelvic floor weakness and rectal prolapse.
- Assess the anal wink (cutaneoanal contractile reflex) by stroking the skin surrounding the anus. Absence of a reflexive contraction of the external anal sphincter suggests nerve damage or spinal arc interruption.
- Perform a vaginal exam to identify pelvic prolapse and exclude rectovaginal fistula. At this time assess pelvic muscle strength and teach the patient proper pelvic floor exercises.
- Conduct a digital rectal examination to assess the length and orientation of the analcanal, bulk of the perianal tissues, and tone of the anal sphincters, as well as to rule out a mass or impaction. Repeat digital examination with the patient bearing down to assess the squeeze pressures, movement of the puborectalis muscle, and extent of pelvic floor descent.
These findings are predominantly subjective and depend on operator experience. In general, they correlate poorly with objective measurements of sphincter pressures.6
If a rectovaginal fistula is suspected, try retrograde filling of the rectum with 200 cc of dye-stained fluid (using a 50-cc catheter tip syringe), then observe vaginal extravasation.
Diagnostic strategy: Routine endoanal ultrasound, manometry
Our strategy is to routinely proceed with endoanal ultrasound and anal manometry to assess the anal sphincters. We may then recommend sigmoidoscopy or anoscopy to rule out mucosal lesions.
It is important to bear in mind that continence depends on multiple physiologic mechanisms and, therefore, no single diagnostic test yields positive results in all patients.
Endoanal ultrasound has significantly changed the evaluation of fecal incontinence over the last decade. In women with fecal incontinence in whom obstetric injury is suspected, 90% have sonographic evidence of injury to 1 or both sphincters.
These ultrasound images show 2 discrete rings of tissue: The inner hypoechoic ring represents the internal anal sphincter and the outer hyperechoic ring represents the external anal sphincter (FIGURE 3). Disrupted continuity of these rings is consistent with structural damage to the sphincter.
Endoanal ultrasound is regarded as superior to other diagnostic methods, with an accuracy of over 90% in detecting sphincter injuries, compared to 75% for manometry, 75% for electromyography (EMG), and 50% for clinical assessment.7,8 It is the simplest and least-invasive test to assess the structural integrity of the anal sphincters.
It can also serve as a screening tool to detect occult sphincter injury following vaginal delivery, thus identifying women at high risk for future incontinence. Women with transient stool or gas incontinence following vaginal delivery may be candidates for screening endoanal ultrasound for further evaluation.
- Where endoanal ultrasound is unavailable, transperineal ultrasound may be an alternative option.
Anorectal manometry can detect functional weakness of sphincters that are anatomically intact by measuring sensation and squeeze pressures. Patients with intact sphincters but abnormal anal manometry findings may benefit from physical therapy and biofeedback aimed at increasing muscular function by stimulating the neuromuscular feedback loop.
During manometry, the physician assesses maximum resting and squeeze pressures of the anal canal, the rectoanal inhibitory reflex, rectal sensation, and rectal compliance using either water-perfused catheters, solid-state microtransducers, or air-or water-filled balloons. Normal values vary widely among institutions and are poorly standardized.
Decreased resting pressure suggests isolated internal anal sphincter injury; decreased squeeze pressure suggests external anal sphincter injury. Of all parameters, maximum squeeze pressure has the greatest sensitivity and specificity at a cut-off of 60 mm Hg in women.9
Rectal sensory testing also is useful in identifying patients who may benefit from biofeedback therapy, and can detect afferent nerve injury as a contributing cause of incontinence. Biofeedback is unlikely to be useful in patients with a poor or absent sensation. In addition, for patients already involved in biofeedback, the test can assess rectal response to training and indicate rectal irritability or poor compliance.
Rectal sensation is assessed by inflating a balloon in the rectum and recording the smallest volume of rectal distention for first detectable sensation (rectal sensory threshold), sensory urgency, and pain (maximum tolerable volume).
Ancillary tests are of limited benefit as screening tools, but may have utility in special circumstances.
- Electromyography of the external anal sphincter and the pelvic floor muscles—traditionally performed using needle electrodes or surface electrodes—is helpful in delineating areas of sphincter injury by mapping the sphincter. However, much of this information is now obtained accurately by endoanal ultrasound, which has replaced needle EMG for most clinicians. Surface EMG is still useful for evaluating sphincter function and in biofeedback training.
- Defecography involves imaging the rectum after filling it with contrast material, and then observing the process, rate, and completeness of rectal evacuation using fluoroscopy. Its usefulness in the evaluation of fecal incontinence is limited to cases of rectal prolapse and for the diagnosis of rectocele and enterocele.
- Investigational: Pudendal nerve testing. This test uses an electrode to measure pudendal nerve conduction time, known as pudendal nerve terminal motor latency (PNTML)—thus allowing further investigation for nerve injury.
- Normal PNTML value is 2.2 milliseconds.
- A value between 2.2 and 2.6 milliseconds indicates probable nerve damage.
- A value of 2.6 milliseconds or greater confirms nerve damage.
Clinical use of PNTML is controversial; it helps diagnose nerve injury, but is currently reserved for investigational purposes.10
FIGURE 3 Endoanal ultrasound
Disruption of continuity of the inner hypoechoic ring (IS, internal sphincter) and the outer hyperechoic ring (ES, external sphincter) on endoanal ultrasound is consistent with structural damage to the sphincter. This image shows normal anatomy.
Mainstays: Nonsurgical treatments
The general principles of treatment are to identify the underlying cause of incontinence and direct therapy based on these findings. Since much of the disability in these patients is psychological, a supportive medical team is essential. Patient educational books, advice on the use of continent aides, and perianal skin care also are crucial.
The physician and patient must formulate a treatment plan based on the severity of the incontinence and degree of social impairment. In general, a trial of nonsurgical therapy is recommended before proceeding to surgery. A standardized treatment algorithm (FIGURE 4) should be adapted to each patient.
The aim of nonsurgical management is:
- to increase anorectal sensation, and
- to increase the strength of the external sphincter and pelvic floor.
First: Diet and medical therapy. For patients with fecal incontinence, nonsurgical intervention remains the first line of treatment given the high rate of underlying neuromuscular damage.
The aims of medical therapy are to decrease stool frequency and improve stool consistency. The patient’s diet should include adequate fiber and fluid intake to promote regular, soft, bulky stools. High-fiber foods including whole-wheat grains, fresh vegetables, and beans should be advised. Bulking agents like methylcellulose (eg, Citrucel), or psyllium (eg, Metamucil) can be helpful.
Stool frequency can be reduced with antidiarrheal drugs like loperamide (2-4 mg 2 or 3 times daily) or diphenoxalate (eg, Lomotil). Loperamide is usually preferred, since it also increases internal anal sphincter tone. Antiflatulants are also recommended.
Amitryptiline is useful in some women with idiopathic fecal incontinence.11,12
Biofeedback therapy. Pelvic floor rehabilitation with biofeedback and/or electrical stimulation—which focuses on the common neuromuscular etiology of fecal incontinence—is the mainstay of nonsurgical management. Some centers report a success rate as high as 70% with this therapy.13
Biofeedback is a painless, minimally invasive method of retraining pelvic floor muscles with the help of sensors placed in either the vagina or the rectum. This technique improves rectal sensation and muscular tone. It is especially useful in incontinence patients with anatomically intact anal sphincters.
Biofeedback typically involves anal manometry as well as electromyographic sensors. It is generally conducted in an outpatient treatment program that includes regular evaluation of sensation and motor tone. However, home-based units are available.
Investigational: Neuromodulation. Although approved by the US Food and Drug Administration for treatment of urge incontinence and nonobstructive voiding dysfunction, neuromodulation is still investigational for treatment of fecal incontinence—but results are encouraging.14
In this technique, electrodes attached to a portable stimulator intermittently apply low-level electrical impulses to the anal canal via the pelvic nerve supply. Newer methods involve implanting electrodes percutaneously in the S3 or S4 foramina under fluoroscopic guidance, then stimulating the sacral nerves.
Neuromodulation aims to increase maximum squeeze pressure and decrease rectal sensitivity to distention. In early trials, significant improvement of incontinence and urgency symptoms was noted.15,16 Larger and longer-duration trials are needed to elucidate long-term effectiveness.
When to consider surgery
Operative therapy should be considered only for cases in which nonsurgical interventions have failed or in which distinct anatomic defects are present.
Note that most surgical candidates still require physical therapy with biofeedback before or after the procedure.
Multiple surgical procedures have been described; some are technically challenging and should be performed only by skilled surgeons after careful preoperative counseling.
Anterior overlapping external sphincteroplasty is done in patients with obstetric or iatrogenic disruption of the external anal sphincter. In this procedure, the surgeon divides the scar and reapproximates viable sphincter muscle to produce a ring of functional muscle. Few good data exist comparing this approach with end-to-end repairs; anterior sphincteroplasty is our preferred method.
- Controversy exists over the need to plicate the internal anal sphincter. Studies have shown that more extensive repairs offer no additional advantage with regard to out-comes.17,18 Most pelvic reconstructive surgeons prefer to perform colporrhaphy and perineorrhaphy at the time of sphincter repair, since defects in the posterior compartment often coexist in these patients.
The success rate of sphincteroplasty ranges from 40% to 75% in appropriately selected patients.19,20
Patients with satisfactory results demonstrate an increase in squeeze pressure postoperatively. Failure to elicit this response correlated accurately with a persistent sphincter defect.21
Total pelvic floor repair may help patients with neurogenic or idiopathic fecal incontinence. The principle of this procedure is to restore the anorectal angle, lengthen the anal canal, and create a circumferential buttress around the anorectum.
In this rather extensive surgery, the surgeon plicates the puborectalis, ischiococcygeus, and iliococcygeus muscles to the rectum; the levator muscles and the external anal sphincter are plicated anteriorly. Success rates vary from 14% to 55%.22,23
Skeletal muscle transposition surgery, with creation of a neosphincter from the gracilis muscle, is performed in patients with pelvic floor muscle weakness so profound that the above techniques offer no improvement. It should be viewed as a salvage operation.
The gracilis muscle is detached from its insertion, wrapped around the anus, and reattached to the contralateral ischial tuberosity. The success of this surgery may be improved by electrically stimulating the transposed muscle with an implanted stimulator.
This procedure is associated with significant postoperative complications; many patients require multiple reoperations. Thus, this intervention is useful only in patients with refractory incontinence.
Artificial anal sphincter procedure involves placing a cuff around the anal canal and a pressure-regulating balloon and pump in the labia majora. The cuff remains inflated, thereby occluding the anal canal until the patient experiences a desire to defecate, at which time it is deflated.
Results are encouraging, with initial success rates from 60% to 75%.24,25 However, local infection can be problematic.
Less common procedures include anal encirclement (Thiersch procedure), perianal fat injection, antegrade continence enema, and fecal diversion.
A word on prevention
During childbirth. Strategies that may reduce pelvic floor trauma:
- Favor mediolateral over midline episiotomy,
- avoid a prolonged second stage of labor and forceps delivery, and
- repair anal sphincter lacerations with an overlapping technique.
Also, discuss regular pelvic floor exercises, constipation prevention, weight loss, and smoking cessation with all obstetric patients—especially those with fecal incontinence risk factors.
Debate continues about elective cesarean section in women with occult sphincter injury on postpartum endoanal ultrasound, transient symptoms of fecal incontinence with prior delivery, or overt gas incontinence.26 This practice may have clinical utility, but data are insufficient to recommend it routinely.
The authors report no financial relationships relevant to this article.
- Endoanal ultrasound is superior to other diagnostic methods, with an accuracy of over 90% in detecting sphincter injuries, compared to 75% for manometry, 75% for electromyography, and 50% for clinical assessment.
- Pelvic floor rehabilitation with biofeedback/electrical stimulation is the mainstay nonsurgical treatment. Some centers report success rates as high as 70%.
- Consider surgery only for distinct anatomic defects, and only when nonsurgical interventions have not succeeded.
Thanks to improvements in our understanding of the causes of uncontrolled defecation and an increasing array of diagnostic and treatment options, physicians can now manage this condition more effectively.
Fecal incontinence is a relevant clinical issue for Ob/Gyns, and requires compassion and emotional support on the part of the physician and health-care team. This debilitating problem is underreported, but is thought to affect 2.2% of the general population. Women are 8 times more likely to have fecal incontinence than men, in age-controlled groups.1
Although sphincter damage during vaginal delivery—even in deliveries over an intact perineum2,3—may be clinically silent in most women, these women are at risk of developing overt fecal incontinence with subsequent deliveries, and with aging and its decompensating factors (FIGURE 1).
A directed examination, optimal diagnostic testing, and a step-by-step treatment algorithm are the basis of effective management.
FIGURE 1 Common pathway leading to fecal incontinence
How ‘silent’ sphincter damage can lead to incontinence
Obstetric disruption of the anal sphincter structure or function is a common cause of fecal incontinence, although it is usually multifactorial, involving abnormalities of 1 or more continence mechanisms (FIGURE 2).
Weakening of the sphincter mechanism after vaginal delivery is attributed to both direct trauma to the muscle and indirect trauma to the pudendal nerve (unilateral or bilateral). Anatomic injury usually occurs in the anterior portion of the sphincter—a site amenable to direct repair, with good results.
Of the various investigational diagnostic modalities available, endoanal ultrasound has been superior, though not perfect. In 1 series, endoanal sonography detected anal sphincter tears in 35% of primiparous and 40% of multiparous women; of these, 13% and 23%, respectively, had symptoms of fecal incontinence or urgency.4
The pudendal nerve is susceptible to traction injury from childbirth and chronic straining. In a prospective study of the effect of vaginal deliveries on the pelvic floor, 80% of primigravidas sustained reversible pudendal nerve damage—which, in some women, persisted for up to 5 years.5
Obstetric risk factors include:
- infant weighing more than 4,000 g,
- use of forceps,
- prolonged second stage of labor,
- midline episiotomy,
- occipitoposterior position, and
- failed detection or suboptimal repair of anal sphincterinjuryat the time of vaginaldelivery.
Onset of menopause and subsequent pelvic floor weakness and perineal descent aggravates this occult neuropathy.
Minor incontinence is also common in patients with advanced age, internal sphincter defects, prolapsing hemorrhoids, rectal prolapse, and fecal impaction (the rectum chronically distends with feces, continuously inducing internal anal sphincter relaxation and resulting in leakage of stool).
Evaluation: Directed history, 4-point physical exam
A directed history helps indicate which diagnostic tests are most relevant. Establish severity and frequency of fecal incontinence, presence of fecal urgency, pad use, type of incontinence (solid, liquid, or gas), and presence of associated urinary incontinence and prolapse.
Obstetric history should identify:
- parity,
- duration of the second stage of labor,
- occipitoposterior presentation,
- use of forceps,
- extent of vaginal lacerations, and
- weight of the infant.
Dietary habits (especially fiber intake) and chronic laxative use should be noted.
Also note any history of abdominopelvic surgery, radiation, back injury, and neurologic disorders.
4-point physical exam
- Inspect the perineum/perianal area for evidence of dermatitis, infection, fistula, hemorrhoids, and deformities of the anus. Ask the patient to bear down as you look for evidence of pelvic floor weakness and rectal prolapse.
- Assess the anal wink (cutaneoanal contractile reflex) by stroking the skin surrounding the anus. Absence of a reflexive contraction of the external anal sphincter suggests nerve damage or spinal arc interruption.
- Perform a vaginal exam to identify pelvic prolapse and exclude rectovaginal fistula. At this time assess pelvic muscle strength and teach the patient proper pelvic floor exercises.
- Conduct a digital rectal examination to assess the length and orientation of the analcanal, bulk of the perianal tissues, and tone of the anal sphincters, as well as to rule out a mass or impaction. Repeat digital examination with the patient bearing down to assess the squeeze pressures, movement of the puborectalis muscle, and extent of pelvic floor descent.
These findings are predominantly subjective and depend on operator experience. In general, they correlate poorly with objective measurements of sphincter pressures.6
If a rectovaginal fistula is suspected, try retrograde filling of the rectum with 200 cc of dye-stained fluid (using a 50-cc catheter tip syringe), then observe vaginal extravasation.
Diagnostic strategy: Routine endoanal ultrasound, manometry
Our strategy is to routinely proceed with endoanal ultrasound and anal manometry to assess the anal sphincters. We may then recommend sigmoidoscopy or anoscopy to rule out mucosal lesions.
It is important to bear in mind that continence depends on multiple physiologic mechanisms and, therefore, no single diagnostic test yields positive results in all patients.
Endoanal ultrasound has significantly changed the evaluation of fecal incontinence over the last decade. In women with fecal incontinence in whom obstetric injury is suspected, 90% have sonographic evidence of injury to 1 or both sphincters.
These ultrasound images show 2 discrete rings of tissue: The inner hypoechoic ring represents the internal anal sphincter and the outer hyperechoic ring represents the external anal sphincter (FIGURE 3). Disrupted continuity of these rings is consistent with structural damage to the sphincter.
Endoanal ultrasound is regarded as superior to other diagnostic methods, with an accuracy of over 90% in detecting sphincter injuries, compared to 75% for manometry, 75% for electromyography (EMG), and 50% for clinical assessment.7,8 It is the simplest and least-invasive test to assess the structural integrity of the anal sphincters.
It can also serve as a screening tool to detect occult sphincter injury following vaginal delivery, thus identifying women at high risk for future incontinence. Women with transient stool or gas incontinence following vaginal delivery may be candidates for screening endoanal ultrasound for further evaluation.
- Where endoanal ultrasound is unavailable, transperineal ultrasound may be an alternative option.
Anorectal manometry can detect functional weakness of sphincters that are anatomically intact by measuring sensation and squeeze pressures. Patients with intact sphincters but abnormal anal manometry findings may benefit from physical therapy and biofeedback aimed at increasing muscular function by stimulating the neuromuscular feedback loop.
During manometry, the physician assesses maximum resting and squeeze pressures of the anal canal, the rectoanal inhibitory reflex, rectal sensation, and rectal compliance using either water-perfused catheters, solid-state microtransducers, or air-or water-filled balloons. Normal values vary widely among institutions and are poorly standardized.
Decreased resting pressure suggests isolated internal anal sphincter injury; decreased squeeze pressure suggests external anal sphincter injury. Of all parameters, maximum squeeze pressure has the greatest sensitivity and specificity at a cut-off of 60 mm Hg in women.9
Rectal sensory testing also is useful in identifying patients who may benefit from biofeedback therapy, and can detect afferent nerve injury as a contributing cause of incontinence. Biofeedback is unlikely to be useful in patients with a poor or absent sensation. In addition, for patients already involved in biofeedback, the test can assess rectal response to training and indicate rectal irritability or poor compliance.
Rectal sensation is assessed by inflating a balloon in the rectum and recording the smallest volume of rectal distention for first detectable sensation (rectal sensory threshold), sensory urgency, and pain (maximum tolerable volume).
Ancillary tests are of limited benefit as screening tools, but may have utility in special circumstances.
- Electromyography of the external anal sphincter and the pelvic floor muscles—traditionally performed using needle electrodes or surface electrodes—is helpful in delineating areas of sphincter injury by mapping the sphincter. However, much of this information is now obtained accurately by endoanal ultrasound, which has replaced needle EMG for most clinicians. Surface EMG is still useful for evaluating sphincter function and in biofeedback training.
- Defecography involves imaging the rectum after filling it with contrast material, and then observing the process, rate, and completeness of rectal evacuation using fluoroscopy. Its usefulness in the evaluation of fecal incontinence is limited to cases of rectal prolapse and for the diagnosis of rectocele and enterocele.
- Investigational: Pudendal nerve testing. This test uses an electrode to measure pudendal nerve conduction time, known as pudendal nerve terminal motor latency (PNTML)—thus allowing further investigation for nerve injury.
- Normal PNTML value is 2.2 milliseconds.
- A value between 2.2 and 2.6 milliseconds indicates probable nerve damage.
- A value of 2.6 milliseconds or greater confirms nerve damage.
Clinical use of PNTML is controversial; it helps diagnose nerve injury, but is currently reserved for investigational purposes.10
FIGURE 3 Endoanal ultrasound
Disruption of continuity of the inner hypoechoic ring (IS, internal sphincter) and the outer hyperechoic ring (ES, external sphincter) on endoanal ultrasound is consistent with structural damage to the sphincter. This image shows normal anatomy.
Mainstays: Nonsurgical treatments
The general principles of treatment are to identify the underlying cause of incontinence and direct therapy based on these findings. Since much of the disability in these patients is psychological, a supportive medical team is essential. Patient educational books, advice on the use of continent aides, and perianal skin care also are crucial.
The physician and patient must formulate a treatment plan based on the severity of the incontinence and degree of social impairment. In general, a trial of nonsurgical therapy is recommended before proceeding to surgery. A standardized treatment algorithm (FIGURE 4) should be adapted to each patient.
The aim of nonsurgical management is:
- to increase anorectal sensation, and
- to increase the strength of the external sphincter and pelvic floor.
First: Diet and medical therapy. For patients with fecal incontinence, nonsurgical intervention remains the first line of treatment given the high rate of underlying neuromuscular damage.
The aims of medical therapy are to decrease stool frequency and improve stool consistency. The patient’s diet should include adequate fiber and fluid intake to promote regular, soft, bulky stools. High-fiber foods including whole-wheat grains, fresh vegetables, and beans should be advised. Bulking agents like methylcellulose (eg, Citrucel), or psyllium (eg, Metamucil) can be helpful.
Stool frequency can be reduced with antidiarrheal drugs like loperamide (2-4 mg 2 or 3 times daily) or diphenoxalate (eg, Lomotil). Loperamide is usually preferred, since it also increases internal anal sphincter tone. Antiflatulants are also recommended.
Amitryptiline is useful in some women with idiopathic fecal incontinence.11,12
Biofeedback therapy. Pelvic floor rehabilitation with biofeedback and/or electrical stimulation—which focuses on the common neuromuscular etiology of fecal incontinence—is the mainstay of nonsurgical management. Some centers report a success rate as high as 70% with this therapy.13
Biofeedback is a painless, minimally invasive method of retraining pelvic floor muscles with the help of sensors placed in either the vagina or the rectum. This technique improves rectal sensation and muscular tone. It is especially useful in incontinence patients with anatomically intact anal sphincters.
Biofeedback typically involves anal manometry as well as electromyographic sensors. It is generally conducted in an outpatient treatment program that includes regular evaluation of sensation and motor tone. However, home-based units are available.
Investigational: Neuromodulation. Although approved by the US Food and Drug Administration for treatment of urge incontinence and nonobstructive voiding dysfunction, neuromodulation is still investigational for treatment of fecal incontinence—but results are encouraging.14
In this technique, electrodes attached to a portable stimulator intermittently apply low-level electrical impulses to the anal canal via the pelvic nerve supply. Newer methods involve implanting electrodes percutaneously in the S3 or S4 foramina under fluoroscopic guidance, then stimulating the sacral nerves.
Neuromodulation aims to increase maximum squeeze pressure and decrease rectal sensitivity to distention. In early trials, significant improvement of incontinence and urgency symptoms was noted.15,16 Larger and longer-duration trials are needed to elucidate long-term effectiveness.
When to consider surgery
Operative therapy should be considered only for cases in which nonsurgical interventions have failed or in which distinct anatomic defects are present.
Note that most surgical candidates still require physical therapy with biofeedback before or after the procedure.
Multiple surgical procedures have been described; some are technically challenging and should be performed only by skilled surgeons after careful preoperative counseling.
Anterior overlapping external sphincteroplasty is done in patients with obstetric or iatrogenic disruption of the external anal sphincter. In this procedure, the surgeon divides the scar and reapproximates viable sphincter muscle to produce a ring of functional muscle. Few good data exist comparing this approach with end-to-end repairs; anterior sphincteroplasty is our preferred method.
- Controversy exists over the need to plicate the internal anal sphincter. Studies have shown that more extensive repairs offer no additional advantage with regard to out-comes.17,18 Most pelvic reconstructive surgeons prefer to perform colporrhaphy and perineorrhaphy at the time of sphincter repair, since defects in the posterior compartment often coexist in these patients.
The success rate of sphincteroplasty ranges from 40% to 75% in appropriately selected patients.19,20
Patients with satisfactory results demonstrate an increase in squeeze pressure postoperatively. Failure to elicit this response correlated accurately with a persistent sphincter defect.21
Total pelvic floor repair may help patients with neurogenic or idiopathic fecal incontinence. The principle of this procedure is to restore the anorectal angle, lengthen the anal canal, and create a circumferential buttress around the anorectum.
In this rather extensive surgery, the surgeon plicates the puborectalis, ischiococcygeus, and iliococcygeus muscles to the rectum; the levator muscles and the external anal sphincter are plicated anteriorly. Success rates vary from 14% to 55%.22,23
Skeletal muscle transposition surgery, with creation of a neosphincter from the gracilis muscle, is performed in patients with pelvic floor muscle weakness so profound that the above techniques offer no improvement. It should be viewed as a salvage operation.
The gracilis muscle is detached from its insertion, wrapped around the anus, and reattached to the contralateral ischial tuberosity. The success of this surgery may be improved by electrically stimulating the transposed muscle with an implanted stimulator.
This procedure is associated with significant postoperative complications; many patients require multiple reoperations. Thus, this intervention is useful only in patients with refractory incontinence.
Artificial anal sphincter procedure involves placing a cuff around the anal canal and a pressure-regulating balloon and pump in the labia majora. The cuff remains inflated, thereby occluding the anal canal until the patient experiences a desire to defecate, at which time it is deflated.
Results are encouraging, with initial success rates from 60% to 75%.24,25 However, local infection can be problematic.
Less common procedures include anal encirclement (Thiersch procedure), perianal fat injection, antegrade continence enema, and fecal diversion.
A word on prevention
During childbirth. Strategies that may reduce pelvic floor trauma:
- Favor mediolateral over midline episiotomy,
- avoid a prolonged second stage of labor and forceps delivery, and
- repair anal sphincter lacerations with an overlapping technique.
Also, discuss regular pelvic floor exercises, constipation prevention, weight loss, and smoking cessation with all obstetric patients—especially those with fecal incontinence risk factors.
Debate continues about elective cesarean section in women with occult sphincter injury on postpartum endoanal ultrasound, transient symptoms of fecal incontinence with prior delivery, or overt gas incontinence.26 This practice may have clinical utility, but data are insufficient to recommend it routinely.
The authors report no financial relationships relevant to this article.
1. Furner S, Cautley E, Norton N, Nelson R. Community-based prevalence of anal incontinence. JAMA. 1995;274:559.-
2. Sultan AH, Kamm MA, Hudson CN, et al. Anal-sphincter disruption during vaginal delivery. N Engl J Med. 1993;329:1905.-
3. Abramowitz L, Sobhani I, Ganansia R, et al. Are sphincter defects the cause of anal incontinence after vaginal delivery? Results of a prospective study. Dis Colon Rectum. May 2000;43:590-596; discussion:596–598.
4. Fynes M, Donnelly VS, O’Connell PR, O’Herlihy C. Cesarean delivery and anal sphincter injury. Obstet Gynecol. 1998;92:496-500.
5. Haadem K, Dahlstrom JA, Lingman G. Anal sphincter function after delivery: a prospective study in women with sphincter rupture and controls. Eur J Obstet Gynecol Reprod Biol. 1990;35:7.-
6. Coller JA. Clinical application of anorectal manometry. Gastroenterol Clin North Am. 1987;16:17.-
7. Sultan AH, Kamm MA, Talbot IC, et al. Anal endosonography for identifying external sphincter defects confirmed histologically. Br J Surg. 1994;81:463.-
8. Tjandra JJ, Milsom JW, Schroeder T, Fazio VW. Endoluminal ultrasound is preferable to electromyography in mapping anal sphincteric defects. Dis Colon Rectum. 1993;36:689-692.
9. Felt-Bersma RJF, Klinkenberg-Knol EC, Meuwissen SGM. Anorectal function investigations in incontinent and continent patients. Dis Colon Rectum. 1990;33:479.-
10. Barnett JL, Hasler WL, Camilleri M. American Gastroenterological Association medical position statement on anorectal testing techniques. American Gastroenterological Association. Gastroenterology. 1999;116:732.-
11. Santoro GA, Eitan BZ, Pryde A, Bartolo DC. Open study of low-dose amitriptyline in the treatment of patients with idiopathic fecal incontinence. Dis Colon Rectum. 2000;43:1676.-
12. Read M, Read NW, Barber DC, Duthie HL. Effects of loperamide on anal sphincter function in patients complaining of chronic diarrhea with fecal incontinence and urgency. Dig Dis Sci. 1982;27:807.-
13. Ryn AK, Morren GL, Hallbook O, Sjodahl R. Long-term results of electromyo-graphic biofeedback training for fecal incontinence. Dis Colon Rectum. 2000;43:1262-1266
14. Matzel KE, Stadelmaier U, Hohenfellner M, Gall FP. Electrical stimulation of sacral spinal nerves for treatment of faecal incontinence. Lancet. 1995;346:1124.-
15. Matzel KE, Stadelmaier U, Hohenfellner M, Hohenberger W. Chronic sacral spinal nerve stimulation for fecal incontinence: long-term results with foramen and cuff electrodes. Dis Colon Rectum. 2001;44:59.-
16. Rosen HR, Urbarz C, Holzer B, et al. Sacral nerve stimulation as a treatment for fecal incontinence. Gastroenterology. 2001;121:536.-
17. Briel JW, de Boer LM, Hop WC, Schouten WR. Clinical outcome of anterior overlapping external anal sphincter repair with internal anal sphincter imbrication. Dis Colon Rectum. 1998;41:209-214.
18. Laurberg S, Swash M, Henry MM. Effect of postanal repair on progress of neurogenic damage to the pelvic floor. Br J Surg. 1990;77:519-522.
19. Kamm MA. Faecal incontinence. BMJ. 1998;316:528.-
20. Malouf AJ, Norton CS, Engel AF, et al. Long-term results of overlapping anterior anal-sphincter repair for obstetric trauma. Lancet. 2000;355:260.-
21. Barnett JL, Hasler WL, Camilleri M. American Gastroenterological Association medical position statement on anorectal testing techniques. American Gastroenterological Association. Gastroenterology. 1999;116:732.-
22. Orrom WJ, Miller R, Cornes H, et al. Comparison of anterior sphincteroplasty and postanal repair in the treatment of idiopathic fecal incontinence. Dis Colon Rectum. 1991;34:305-310.
23. Schiller, Lawrence R. Fecal incontinence. In: Feldman M, et al, eds. Sleisenger & Fordtran’s Gastrointestinal and Liver Disease. Philadelphia, Pa: WB Saunders Co; 1997.
24. Giamundo P, Welber A, Weiss EG, et al. The Procon incontinence device: a new nonsurgical approach to preventing episodes of fecal incontinence. Am J Gastroenterol. 2002;97:232.-
25. Wong WD, Congliosi SM, Spencer MP, et al. The safety and efficacy of the artificial bowel sphincter for fecal incontinence: results from a multicenter cohort study. Dis Colon Rectum. 2002;45:1139.-
26. McKenna DS, Ester JB, Fischer JR. Elective cesarean delivery for women with a previous anal sphincter rupture. Am J Obstet Gynecol. 2003;189:1251.-
1. Furner S, Cautley E, Norton N, Nelson R. Community-based prevalence of anal incontinence. JAMA. 1995;274:559.-
2. Sultan AH, Kamm MA, Hudson CN, et al. Anal-sphincter disruption during vaginal delivery. N Engl J Med. 1993;329:1905.-
3. Abramowitz L, Sobhani I, Ganansia R, et al. Are sphincter defects the cause of anal incontinence after vaginal delivery? Results of a prospective study. Dis Colon Rectum. May 2000;43:590-596; discussion:596–598.
4. Fynes M, Donnelly VS, O’Connell PR, O’Herlihy C. Cesarean delivery and anal sphincter injury. Obstet Gynecol. 1998;92:496-500.
5. Haadem K, Dahlstrom JA, Lingman G. Anal sphincter function after delivery: a prospective study in women with sphincter rupture and controls. Eur J Obstet Gynecol Reprod Biol. 1990;35:7.-
6. Coller JA. Clinical application of anorectal manometry. Gastroenterol Clin North Am. 1987;16:17.-
7. Sultan AH, Kamm MA, Talbot IC, et al. Anal endosonography for identifying external sphincter defects confirmed histologically. Br J Surg. 1994;81:463.-
8. Tjandra JJ, Milsom JW, Schroeder T, Fazio VW. Endoluminal ultrasound is preferable to electromyography in mapping anal sphincteric defects. Dis Colon Rectum. 1993;36:689-692.
9. Felt-Bersma RJF, Klinkenberg-Knol EC, Meuwissen SGM. Anorectal function investigations in incontinent and continent patients. Dis Colon Rectum. 1990;33:479.-
10. Barnett JL, Hasler WL, Camilleri M. American Gastroenterological Association medical position statement on anorectal testing techniques. American Gastroenterological Association. Gastroenterology. 1999;116:732.-
11. Santoro GA, Eitan BZ, Pryde A, Bartolo DC. Open study of low-dose amitriptyline in the treatment of patients with idiopathic fecal incontinence. Dis Colon Rectum. 2000;43:1676.-
12. Read M, Read NW, Barber DC, Duthie HL. Effects of loperamide on anal sphincter function in patients complaining of chronic diarrhea with fecal incontinence and urgency. Dig Dis Sci. 1982;27:807.-
13. Ryn AK, Morren GL, Hallbook O, Sjodahl R. Long-term results of electromyo-graphic biofeedback training for fecal incontinence. Dis Colon Rectum. 2000;43:1262-1266
14. Matzel KE, Stadelmaier U, Hohenfellner M, Gall FP. Electrical stimulation of sacral spinal nerves for treatment of faecal incontinence. Lancet. 1995;346:1124.-
15. Matzel KE, Stadelmaier U, Hohenfellner M, Hohenberger W. Chronic sacral spinal nerve stimulation for fecal incontinence: long-term results with foramen and cuff electrodes. Dis Colon Rectum. 2001;44:59.-
16. Rosen HR, Urbarz C, Holzer B, et al. Sacral nerve stimulation as a treatment for fecal incontinence. Gastroenterology. 2001;121:536.-
17. Briel JW, de Boer LM, Hop WC, Schouten WR. Clinical outcome of anterior overlapping external anal sphincter repair with internal anal sphincter imbrication. Dis Colon Rectum. 1998;41:209-214.
18. Laurberg S, Swash M, Henry MM. Effect of postanal repair on progress of neurogenic damage to the pelvic floor. Br J Surg. 1990;77:519-522.
19. Kamm MA. Faecal incontinence. BMJ. 1998;316:528.-
20. Malouf AJ, Norton CS, Engel AF, et al. Long-term results of overlapping anterior anal-sphincter repair for obstetric trauma. Lancet. 2000;355:260.-
21. Barnett JL, Hasler WL, Camilleri M. American Gastroenterological Association medical position statement on anorectal testing techniques. American Gastroenterological Association. Gastroenterology. 1999;116:732.-
22. Orrom WJ, Miller R, Cornes H, et al. Comparison of anterior sphincteroplasty and postanal repair in the treatment of idiopathic fecal incontinence. Dis Colon Rectum. 1991;34:305-310.
23. Schiller, Lawrence R. Fecal incontinence. In: Feldman M, et al, eds. Sleisenger & Fordtran’s Gastrointestinal and Liver Disease. Philadelphia, Pa: WB Saunders Co; 1997.
24. Giamundo P, Welber A, Weiss EG, et al. The Procon incontinence device: a new nonsurgical approach to preventing episodes of fecal incontinence. Am J Gastroenterol. 2002;97:232.-
25. Wong WD, Congliosi SM, Spencer MP, et al. The safety and efficacy of the artificial bowel sphincter for fecal incontinence: results from a multicenter cohort study. Dis Colon Rectum. 2002;45:1139.-
26. McKenna DS, Ester JB, Fischer JR. Elective cesarean delivery for women with a previous anal sphincter rupture. Am J Obstet Gynecol. 2003;189:1251.-
Fetal growth restriction
- Antepartum recognition of fetal growth restriction is essential for proper surveillance and management.
- Because growth-restricted fetuses are at risk for adverse outcomes in utero, fetal surveillance is vital for timely recognition and intervention.
- Once growth restriction is identified, management should be individualized to ensure optimal gestational development and safe delivery.
Not only is fetal growth restriction (FGR) associated with perinatal mortality and morbidity, but it may be linked to adverse consequences in adulthood.1 Its many causes involve diverse pathological processes; thus, it should not be considered a single disease. Many aspects remain unclear, a substantial number of affected infants are not identified before birth, and effective antepartum prevention and treatment remain elusive.
Fortunately, managing FGR has hopeful aspects. Meaningful recent advances elucidate its etiologic and pathophysiologic mechanisms and help clarify diagnosis and management.
This article offers an up-to-date, evidence-based approach and includes guidelines on 3 keys to success: antepartum recognition, fetal surveillance, and antepartum and intrapartum management. These guidelines are not intended as a strict protocol, since the clinical course of FGR is highly variable, but as the starting point for individualized care.
An amorphous entity: FGR terminology
Fetal growth restriction implies failure to realize genetically determined growth potential. Terms include fetal growth restriction, intrauterine growth restriction, and small for gestational age (SGA). The pejorative term growth retardation is obsolete.
Traditionally, FGR refers to prenatally identified fetal growth deficiency, whereas SGA refers to suboptimal birth weight for the gestational age. However, some small fetuses are merely constitutionally small, not growth-restricted. Conversely, not all growth-restricted fetuses are small in size or weight for gestational age. Yet defining these groups is difficult, as the tools are imprecise and controversial.
This article uses these definitions:
Fetal growth restriction identified in the antepartum period refers to a fetus with sonographically measured fetal dimensions, particularly abdominal circumference or estimated weight, below an age-specific threshold, typically the 10th percentile.
Fetal growth restriction identified at birth is birth weight below the 10th percentile for gestational age, or SGA. Unfortunately, this definition may fail to identify some fetuses that are truly growth-restricted. Alternative, more sensitive definitions, such as the Ponderal index or birth weight ratio, are used primarily in research.
Consequences of FGR
Perinatal outcomes. Perinatal morbidities include prematurity, oligohydramnios, nonreassuring fetal heart rate patterns with a higher incidence of cesarean delivery, birth asphyxia, low Apgar score, neonatal hypoglycemia, hypocalcemia, polycythemia, hyperbilirubinemia, hypothermia, apnea, seizure disorders, and infection.
Fetal and neonatal mortality is significantly increased. Perinatal mortality is influenced by many factors, including severity of growth restriction, timing of onset, gestational age, and cause of growth restriction. The lower the birth-weight percentile for gestational age, the higher the mortality rate.
Effects in infancy. Although many SGA infants “catch up” growth in infancy, the pattern varies. Height and weight catch-up growth of preterm FGR infants lags behind that of preterm infants that are appropriate for gestational age (AGA) at birth.2 Those with early-onset or severe growth deficit continue to lag behind in postnatal growth. Although recent studies indicate that rapid postnatal growth in SGA infants may lead to increased risk of chronic diseases, including type 2 diabetes, others have found tangible short-term benefits of less frequent morbidity and mortality in infancy.3
FGR has been linked to a spectrum of neurodevelopmental risks, including subtle behavioral abnormalities, immature sleep patterns, decreased visual fixation, decreased general activity, altered early mother-infant interaction, altered motor skills, and hyperactivity.4,5 Infants born SGA at 32 to 42 weeks were 4 to 6 times more likely to have cerebral palsy, yet those whose birth weight was above the 97th percentile also had increased risk. It remains uncertain whether deviant growth is the cause or a consequence of this disability.6
Long-term effects. Lifetime sequelae of early nutritional deprivation have been demonstrated in animals.7 Moreover, epidemiological evidence suggesting an association between SGA at birth or infancy and increased risk of abnormal blood lipid values, diabetes, hypertension, and ischemic heart disease in adult life led Barker and associates to propose the fetal origins hypothesis.1
1. Antepartum recognitionDetermine gestational age
A reliable estimate of gestational age is central to identification of fetal growth compromise in utero or at birth. In pregnancies at risk for fetal growth restriction, gestational age should be established early, preferably in the first trimester.
The method of determining gestational age influences the observed frequency of FGR and SGA births. Estimates are more accurate when based on early ultrasound biometry than on menstrual history. The latter, if well documented, regular, and ovulatory, may be reliable if it differs by no more than 1 week from the sonographic gestational age. Otherwise, early-pregnancy, ultrasoundbased age is more accurate.
Screening for FGR
Screening can be done clinically or by special investigation. The following methods are used, some of which remain experimental:
- assessment of historical clinical risks
- clinical evaluation of fetal and uterine size
- ultrasound fetal biometry
- umbilical arterial and uterine arterial
Doppler ultrasound
Clinical risk assessment. Evaluate all gravidas for risk factors (TABLE 1). If a woman is determined to be at heightened risk, take appropriate steps to diagnose FGR as early as possible.
Clinical evaluation of fetal and uterine size. Clinical assessment of fetal growth includes estimating fetal size by traditional obstetrical manual examination and by measuring the uterine fundal height.
- Abdominal palpation is inadequate to identify the fetus at increased risk of growth restriction, missing 74% of cases.8
- Serial measurements of the uterine fundal height, however, may be more reliable.9 This method consists of measuring fundal height from the symphysis pubis using a nonstretchable tape measure and assessing the results against a gestational age-specific nomogram. Limitations include significant interobserver differences and varying effects of maternal weight, parity, and fetal sex. A randomized trial of the method did not demonstrate any benefits, and a subsequent Cochrane review of the same study considered the evidence insufficient for any recommendations.10,11
Nevertheless, clinical assessment of uterine and fetal size is an essential, inexpensive component of prenatal care and a simple screening tool for identifying mothers who would benefit from further, more definitive sonographic investigation.
Routine ultrasound biometry. The potential benefits of accurately determined gestational age, and recognition of fetal malformations and multiple gestation, via earlyor mid-pregnancy ultrasound are well recognized and justify widespread use.
Routine scanning increases detection of SGA infants.12 A population-based cohort study13 involving over 16,000 singleton pregnancies found that fetuses smaller than expected at mid-second trimester ultrasound (discrepancy exceeding 14 days) were at increased risk for adverse outcomes, including perinatal mortality and SGA.
Unfortunately, these findings have not led to improved outcomes. A high false-positive rate remains a major concern. A prospective observational study14 found that routine ultrasound did not identify most cases of FGR, but resulted in a fivefold increase in iatrogenic premature delivery and significantly increased neonatal intensive care admissions.
- The Routine Antenatal Diagnostic Imaging with Ultrasound (RADIUS) trial15 randomized 15,000 low-risk gravidas to routine ultrasound imaging (at 15 to 22 weeks and again at 31 to 35 weeks) or to ultrasound only when indicated. The groups had similar rates of adverse perinatal outcome, distribution of birth weights, and preterm delivery. The trial’s weaknesses include selection criteria for low risk that excluded most pregnancies, inappropriate perinatal-outcome parameters, and suboptimal ultrasonographer expertise.
- A recent Cochrane review16 of 7 trials involving more than 25,000 women failed to demonstrate any improvements in perinatal mortality and morbidity with routine ultrasound, or any difference in antenatal, obstetric, and neonatal interventions.
Estimated fetal weight may differ from actual weight by as much as 20%.
We also lack evidence regarding longterm outcomes such as neurodevelopment.
Umbilical arterial Doppler. A meta-analysis17 of 4 randomized trials in unselected or low-risk pregnancies with a total population of 11,375 women found no effect of screening Doppler umbilical artery velocimetry on perinatal death, stillbirth, antenatal hospitalization, obstetric outcome, or perinatal morbidity.
A subsequent meta-analysis18 of 5 trials of routine Doppler ultrasound in unselected and low-risk pregnancies with a total population of more than 14,000 women also found no benefit for mother or infant.
We lack evidence on long-term outcomes.
Uterine arterial Doppler screening. Increased pulsatility of the uterine arterial Doppler waveform, persistence of the notch, and a significant difference between right and left uterine arteries have been associated with FGR, pregnancy-induced hypertension, and adverse perinatal outcome. A review19 of 15 studies of routine uterine Doppler in unselected populations showed considerable heterogeneity, but indicated that increased impedance in the uterine arteries identifies about 20% of those who develop FGR, with a positive likelihood ratio of 3.5.
We lack exclusive randomized trials of routine uterine Doppler sonography in unselected and low-risk pregnancies. However, 2 studies done in conjunction with umbilical arterial Doppler found no impact on outcome, and a recent Cochrane review18 found insufficient evidence to support routine uterine Doppler for FGR screening.
TABLE 1
Risk factors
| MATERNAL FACTORS |
| Medical disease |
| Preeclampsia/hypertension |
| Renal disease |
| Antiphospholipid antibody syndrome |
| Inherited thrombophilia |
| Diabetes with vasculopathy |
| Cyanotic heart disease |
| Asthma |
| Hemoglobinopathy |
| Phenylketonuria |
| Life circumstance |
| Severe malnutrition |
| Smoking |
| Substance abuse (eg, alcohol, heroine, cocaine) |
| PLACENTAL FACTORS |
| Confined placental mosaic |
| Placenta previa |
| Abruptio placentae |
| Infarction |
| Circumvallate placenta |
| Placenta accreta |
| Hemangioma |
| FETAL FACTORS |
| Multiple gestation |
| Prematurity |
| Unexplained elevated alpha-fetoprotein |
| Infection (eg, rubella, cytomegalovirus, herpes, malaria, toxoplasmosis) |
| Malformations (eg, gastroschisis, omphalocele, diaphragmatic hernia, congenital heart defect) |
| Genetic disorders (eg, trisomy 13, 18, and 21; triploidy; some cases of Turner’s syndrome) |
Identifying FGR in utero
Antepartum diagnosis is based on sonographic measurement of various fetal dimensions.
Abdominal circumference and estimated fetal weight. A review20 of 60 studies found that abdominal circumference (AC) and sonographically estimated fetal weight (EFW) were the best predictors of birth weight below the 10th percentile in high-risk pregnancies. AC below the 10th percentile had sensitivities ranging from 72.9% to 94.5%, false-positive rates of 16.2% to 49.4%, and a common odds ratio of 18.4. An EFW below the 10th percentile had a common odds ratio of 39.1, which was the highest among all the biometric measurements. Its sensitivity ranged from 33.3% to 89.2%, and false-positive rates ranged from 9.1% to 46.3%.
The proportionality of fetal dimensions, such as the head/abdominal ratio, was not a good predictor, although it is routinely generated in ultrasound biometry.
Ultrasound estimation of fetal weight is based on a combination of 2 or 3 biometric measurements of the fetus, including the biparietal diameter (BPD) or the head circumference (HC), AC, and femur length (FL). Several formulae yield varying estimations of weight. Thus, a fetus identified as growth-restricted by 1 formula may not be so diagnosed by another. For this reason, it is prudent to be consistent in their use. EFW generated from measurements of the head (BPD or HC), AC, and FL is most reliable.21
The EFW also is expressed as the percentile for the gestational age.
Limitations of ultrasound estimation.
The optimal process of translating dimensional measurements into fetal body mass for both health and disease remains unknown. This leads to inaccurate assumptions and erroneous weight projections.
Inaccuracies also result from variations in measurement. In addition, the estimation is less accurate in the lower and upper ranges of fetal weight distribution and in the presence of oligohydramnios. As a result, estimated weight may differ from actual weight by as much as 20%, with greater margins of error at the lower and upper extremes.
Guidelines for screening and diagnosis
Assess all pregnancies for risk factors and determine the gestational age in early pregnancy, especially in women at higher risk of FGR. If the patient has substantial clinical risks or there is suspicion of growth restriction, fetal ultrasound biometry is recommended. Diagnosis of FGR is based on fetal sonographic measurements, especially AC; estimated fetal weight derived from BPD or HC, AC and FL; and longitudinal progression of fetal growth.
Current evidence suggests the use of a 10th percentile diagnostic threshold for these measurements. Follow the biometric parameters longitudinally, repeating the measurement every 2 to 4 weeks. More frequent examination is unreliable.
2. Fetal surveillanceAntepartum monitoring
Fetuses identified as growth-restricted are at risk for adverse outcomes in utero. Thus, antepartum monitoring is indicated for timely recognition and intervention.
Tests commonly used include the nonstress test (NST), biophysical profile (BPP), sonographic estimation of amniotic fluid volume, and Doppler velocimetry of the umbilical artery or other vessels of interest.
With progressive fetal compromise, compensatory mechanisms may be insufficient to maintain fetal homeostasis. The sequence of loss of compensatory mechanisms is reflected in surveillance tests. Thus, changes in umbil-ical arterial Doppler indices usually precede nonreassuring NST or BPP.
Nonstress test: The standard of care. The NST is probably the most common fetal surveillance test in high-risk pregnancies, including those complicated by FGR. However, it is personnel- and time-intensive, and interpretation often is subjective.
A nonreactive NST is associated with adverse perinatal outcome, although it more commonly is related to the fetal sleep cycle.
In predicting a potentially preventable fetal death, the NST has a false-negative rate of 2 to 3 per 1,000, a negative predictive value of 99.8%, and a false-positive rate of 80%.
Evidence of its efficacy is lacking. Four randomized trials in the early 1980s, which involved more than 1,500 women, found no improvement in outcomes. (All 4 trials lacked sufficient power.)
- When to test. Despite these limitations, the NST has become an integral part of obstetri-cal practice and remains a standard of care for antepartum fetal surveillance in high-risk pregnancies. It should be used as a surveillance tool for the growth-restricted fetus. The test initially is performed weekly.
Depending on severity of growth restriction, testing may increase to twice weekly or even daily.
Amniotic fluid volume monitoring is warranted. Oligohydramnios—highly prevalent in FGR—is associated with adverse perina-tal outcomes.
Although the sonographically determined amniotic fluid index (AFI) is the most frequently used modality to evaluate amniotic fluid volume, its accuracy has been questioned. Nevertheless, a meta-analysis22 of 18 studies involving more than 10,000 patients showed that antepartum AFI equal to or below 5 cm was associated with an increased risk of cesarean section for fetal distress and an Apgar score below 7 at 5 minutes.
Even marginal AFI values (5–10 cm) have been associated with doubling of the risk of adverse perinatal outcome.23
- When to test. We lack randomized trials of amniotic fluid volume assessment in high-risk pregnancies, but observational studies suggest that weekly determination of AFI is helpful in sonographically identified growth-restricted fetuses.
A low or marginal AFI should be followed by more frequent examinations. Even a marginal AFI may be followed by twice-weekly surveillance.
Biophysical profile effectively predicts adverse perinatal outcome, with a false-negative rate of 0.8 per 1,000, negative predictive value of 99.9%, and a false-positive rate of 40% to 50%.24 A low BPP is associated with fetal hypoxia and acidosis, and a declining BPP reflects progressive worsening of the fetal condition.
In FGR, a significant association exists between abnormal Doppler indices and fetal acid-base compromise.
Although equal weight was originally accorded all the parameters of the profile, experience suggests that the independent risk of oligohydramnios warrants immediate reassessment of the management plan.
Like the nonstress test, the BPP has a low false-negative rate and a high false-positive rate. Four randomized trials of the test have been conducted, involving about 2,800 patients, with no confirmation of its effectiveness. A Cochrane systematic review25 concluded that this cumulative sample size is insufficient to draw definitive conclusions.
The test remains a standard, however, and is recommended to confirm fetal wellbeing or as part of a comprehensive test panel in significantly high-risk pregnancies.
- When to test. Initially perform the test once a week, increasing to twice weekly when the severity of growth restriction warrants.
Daily testing may be indicated in complications such as severe growth restriction and pregestational diabetes. In early preterm pregnancies (less than 32 weeks) with nonreassuring NST or umbilical arterial Doppler, BPP can help guide optimal management.
Umbilical arterial Doppler ultrasound is a powerful predictor of adverse perinatal outcomes in high-risk pregnancies.
Doppler indices include the pulsatility index, systolic/diastolic ratio, diastolic average ratio, and the resistance index. Of these, the last demonstrated the greatest ability to predict abnormal perinatal outcomes.
A prospective, blinded study26 in highrisk pregnancies demonstrated significant diagnostic efficacy with a sensitivity of 79%, specificity of 93%, positive predictive value of 83%, negative predictive value of 91%, and kappa index of 73%. The last value is consistent with a good to excellent diagnostic test.
A significant association also exists between abnormal Doppler indices and fetal acid-base compromise in FGR. The absence of enddiastolic flow (FIGURE) is associated with markedly adverse perinatal outcome, particularly a high perinatal mortality rate, malformations, and aneuploidy. Nicolaides and coworkers27 found that, when the end-diastolic velocity was absent, 67% to 80% of the fetuses were hypoxic and 45% were acidotic. Reversal of end-diastolic flow is associated with an even worse prognosis. The adverse consequences of absent or reverse end-diastolic flow are listed in TABLE 2.
In contrast to the NST and BPP, the effectiveness of fetal surveillance with umbilical arterial Doppler ultrasound in improving perinatal outcome in high-risk pregnancies has been confirmed by extensive randomized trials and their meta-analyses.28,29 These studies provide compelling evidence that, when used in well-defined high-risk pregnancies, especially those involving FGR or preeclampsia, umbilical arterial Doppler sonography reduces perinatal death, cesarean delivery for fetal distress, elective deliveries, and antenatal admissions.
Doppler sonography of the fetal cerebral and venous circulation also helps identify progressive fetal compromise in high-risk pregnancies, including those with FGR. When the fetal condition is deteriorating, cerebral arteries dilate to compensate, and pulsations appear in the umbilical vein with reversal of flow in the ductus venous.
These signs of grave fetal status indicate immediate delivery.
FIGURE 1 Duplex pulsed Doppler
Sonogram showing absent end-diastolic flow in the umbilical artery of a growth-restricted fetus.TABLE 2
Perinatal outcomes for absent and reversed end-diastolic velocity in the umbilical artery
| PERINATAL OUTCOME | MEAN | RANGE |
|---|---|---|
| Mortality | 45% | 17–100% |
| Gestational age | 31.6 weeks | 29–33 weeks |
| Birth weight | 1,056 g | 910–1,481 g |
| SGA | 68% | 53–100% |
| Cesarean section for fetal distress | 73% | 24–100% |
| Apgar score <7 at 5 minutes | 26% | 7–69% |
| Admission to NICU | 84% | 77–97% |
| Congenital anomalies | 10% | 0–24% |
| Aneuploidy | 6.4% | 0–18% |
| SGA = small for gestational age; NICU = neonatal intensive care unit | ||
| Reprinted with permission from: Maulik D, ed. Doppler Ultrasound in Obstetrics and Gynecology. | ||
| New York: Springer-Verlag; 1997:364, Table 21.2. | ||
Assessing the fetus for malformation and aneuploidy
Because of the association between FGR and fetal malformations and aneuploidy, the fetus should be assessed for these complications, especially when growth restriction is severe, develops early, and is not associated with hypertensive disorders, oligohydramnios, or abnormal umbilical arterial Doppler indices.
Most etiologies are either not amenable to therapy or fetal growth is not improved by treatments that benefit the mother.
The current standard practice is aneuploidy screening in early pregnancy and fetal anatomical scanning at midgestation. If significant risks of aneuploidy are present, a fetal karyotype is recommended, along with appropriate counseling.
3. Individualized managementAntepartum treatment
The question of bed rest. At present, there are no grounds for recommending strict bed rest in FGR cases. A Cochrane review30 showed no improvements with hospitalized bed rest compared with ambulation. Prolonged rest increases risk of thromboembolism and can be costly and inconvenient.
Nutritional remedies. Of the options that have been tested, which include high protein supplementation, nutrient supplementation such as beef liver extract, and balanced energy/protein supplementation, only the last (with protein content comprising less than 25% of total energy content) has led to a significant reduction in SGA births.31-33
Other approaches. We lack reliable evidence that other interventions such as oxygen administration, abdominal decompression, and pharmacological agents, including calcium channel blockers, beta mimetics, and magnesium, are beneficial or effective in improving or preventing FGR.
A meta-analysis34 of 13 trials involving more than 13,000 women showed that early aspirin treatment reduced the risk of FGR but failed to improve outcome. A more recent meta-analysis35 of 38 trials of aspirin in highrisk pregnancies found no reduction in the incidence of FGR or perinatal death, although a reduction in risk of preterm births was noted.
Etiologic management
Most etiologic conditions are either not amenable to therapy or fetal growth is not improved by treatments that benefit the mother. An example is maternal hypertensive disease, in which the indicated treatment has no beneficial effect on fetal growth.
Treatment of poor lifestyle habits may be helpful. If the mother smokes, vigorous smoking-cessation education and counseling is urged. Also address alcohol consumption and other substance abuse, and offer remedial measures.
The diagnosis of fetal viral and parasitic infections is important for prognostication and neonatal management. Although few perinatal infections are treatable in utero, maternal therapy may prevent certain infections from spreading to the fetus. Examples include toxoplasmosis and malaria.
If lethal malformations or lethal aneuploidy are identified prenatally, avoid fetal surveillance and unnecessary intervention, which may expose the mother to unnecessary and unjustifiable risks.
Fetal surveillance strategy
If fetal biometry indicates fetal weight below the 10th percentile, begin fetal surveillance. The current standard, umbilical arterial Doppler sonography, is the primary test in sonographically documented growth restriction. Also assess amniotic fluid volume—as part of the BPP or independently. AFI is the most commonly used BPP tool.
In the United States, the nonstress test traditionally is the primary monitoring modality, with the BPP as backup, although the BPP—which includes the NST—has been recommended as the primary test.
Normal Doppler findings. When the Doppler index remains within normal limits or does not progressively rise, weekly testing should suffice, with the NST or BPP as backup or in conjunction with Doppler.
If fetal and maternal conditions remain reassuring, allow the pregnancy to continue to maturity and assess the patient for delivery. Postdate pregnancy is not advised in the presence of sonographically confirmed growth compromise.
A high or increasing Doppler index warrants more intensive fetal surveillance consisting of weekly umbilical arterial Doppler and once- or twice-weekly NST and BPP until fetal maturity.
If these tests indicate fetal compromise, or absent end-diastolic velocity develops, the likelihood of poor perinatal outcome is increased and an urgent clinical response is indicated. Hospitalize the patient and individualize management, depending on gestational age and fetal status.
Optimal timing of delivery
The optimal timing of delivery in a preterm pregnancy with FGR is unclear. A recent multicenter randomized controlled trial, the Growth Restriction Intervention Trial (GRIT),36 compared 2 strategies: delivery within 48 hours with steroid administration or delivery delayed as long as fetal status permits. The population consisted of high-risk gravidas between 24 and 36 weeks’ gestation. More than 90% of the women had pregnancies complicated by FGR. No significant differences were noted between the 2 groups in stillbirth rates.
Fortunately, thanks to recent advances in perinatal care, a management strategy can be recommended:
At or near term (34 weeks or beyond), the absence of end-diastolic flow in the umbilical artery should prompt consideration of immediate delivery.
Other ominous findings that prompt such consideration include:
- Cessation of fetal growth on successive ultrasound examinations
- Progression of umbilical arterial absent enddiastolic flow to reversed end-diastolic flow
- Nonreassuring heart rate patterns including nonreactive NST, poor fetal heart rate baseline variability, and persistent variable or late decelerations
- Oligohydramnios
- BPP score of 4 or below
Less than 34 weeks. When absent enddiastolic flow develops in a preterm pregnancy with a significant risk of fetal lung immaturity, seek further assurance of fetal well-being via daily surveillance with umbilical arterial Doppler sonography, nonstress test, and biophysical profile. Administer betamethasone to enhance fetal lung maturity. Delivery is indicated regardless of maturity when a single test or combination indicates imminent fetal danger and the fetal risk from a hostile intrauterine environment is judged to be greater than that from pulmonary immaturity.
Intrapartum management
Labor and delivery of growth-restricted infants often is associated with a higher risk of asphyxia. The frequency of nonreassuring fetal heart rate patterns is increased. In addition, because of the greater prevalence of oligohydramnios, cord compression is frequent and associated with variable decelerations. There also is an increased risk of neonatal morbidities, as discussed above. Because of these risks, the growth-restricted fetus should be delivered in a facility that can offer appropriate inhouse support services such as neonatology and anesthesia.
If the patient is allowed to labor, use continuous electronic fetal heart rate monitoring with vigilance and perform the appropriate secondary tests, such as fetal scalp sampling or scalp stimulation. The ability of a growth-restricted fetus to tolerate labor is likely very limited.
Any confirmed signs of fetal compromise indicate cesarean as the optimal mode of delivery.
The authors report no financial relationships relevant to this article.
1. Barker DJP, ed. Fetal and Infant Origins of Adult Disease. London, UK: BMJ Publishing; 1992.
2. Strauss RS, Dietz WH. Effects of intrauterine growth retardation in premature infants on early childhood growth. J Pediatr. 1997;13:95-102.
3. Victora CG, Barros FC, Horta BL, Martorell R. Short-term benefits of catch-up growth for small-for-gestational-age infants. Int J Epidemiol. 2001;30:1325-1330.
4. Manning FA, Morrison I, Harman CR, Lange IR, Menticoglou S. Fetal assessment based on the fetal biophysical profile scoring: experience in 19,221 referred high-risk pregnancies. II. An analysis of false-negative fetal deaths. Am J Obstet Gynecol. 1987;157:880-884.
5. Low JA, Handley-Derry MH, Burke SO, et al. Association of intrauterine fetal growth retardation and learning deficits at age 9 to 11 years. Am J Obstet Gynecol. 1992;167:1499-1505.
6. Jarvis S, Glinianaia SV, Torrioli MG, et al. Cerebral palsy and intrauterine growth in single births: European collaborative study. Lancet. 2003;362:1106-1111.
7. Lucas A. Programming by early nutrition: an experimental approach. J Nutr. 1998;128(suppl 2):401-406S.
8. Hepburn M, Rosenberg K. An audit of the detection and management of small-for-gestational age babies. Br J Obstet Gynaecol. 1986;93:212-216.
9. Belizan JM, Villar J, Nardin JC, Malamud J, Sainz de Vicuna L. Diagnosis of intrauterine growth retardation by a simple clinical method: measurement of uterine height. Am J Obstet Gynecol. 1978;131:643-646.
10. Lindhard A, Nielsen PV, Mouritsen LA, Zachariassen A, Sorensen HU, Roseno H. The implications of introducing the symphyseal-fundal height measurement. A prospective randomized controlled trial. Br J Obstet Gynaecol. 1990;97:675-680.
11. Neilson JP. Symphysis-fundal height measurement in pregnancy. Cochrane Database Syst Rev. 2000;(2):CD000944.-
12. Hughey MJ. Routine ultrasound for detection and management of the small for-gestational-age fetus. Obstet Gynecol. 1984;64:101-107.
13. Nakling J, Backe B. Adverse obstetric outcome in fetuses that are smaller than expected at second trimester routine ultrasound examination. Acta Obstet Gynecol Scand. 2002;81:846-851.
14. Jahn A, Razum O, Berle P. Routine screening for intrauterine growth retardation in Germany: low sensitivity and questionable benefit for diagnosed cases. Acta Obstet Gynecol Scand. 2002;81:846-851.
15. Ewigman BG, Crane JP, Frigoletto FD, LeFevre ML, Bain RP, McNellis D. Effect of prenatal ultrasound screening on perinatal outcome. RADIUS Study Group. N Engl J Med. 1993; 16;329:821-827.
16. Bricker L, Neilson JP. Routine ultrasound in late pregnancy (after 24 weeks gestation). Cochrane Database Syst Rev. 2000;(2):CD001451.-
17. Goffinet F, Paris Llado J, Nisand I, Breart G. Umbilical artery Doppler velocimetry in unselected and low risk pregnancies: a review of randomized controlled trials. Br J Obstet Gynaecol. 1997;104:425-430.
18. Bricker L, Neilson JP. Routine Doppler ultrasound in pregnancy. Cochrane Database Syst Rev. 2000;(2):CD001450.-
19. Papageorghiou AT, Yu CK, Cicero S, Bower S, Nicolaides KH. Second-trimester uterine artery Doppler screening in unselected populations: a review. J Matern Fetal Neonatal Med. 2002;12:78-88.
20. Chang TC, Robson SC, Boys RJ, Spencer JA. Prediction of the small for gestational age infant: which ultrasonic measurement is best? Obstet Gynecol. 1992;80:1030-1038.
21. Hadlock FP, Harrist RB, Sharman RS, Deter RL, Park SK. Estimation of fetal weight with the use of head, body, and femur measurements—a prospective study. Am J Obstet Gynecol. 1985;151:333-337.
22. Chauhan SP, Sanderson M, Hendrix NW, Magann EF, Devoe LD. Perinatal outcome and amniotic fluid index in the antepartum and intrapartum periods: a meta-analysis. Am J Obstet Gynecol. 1999;18:1473-1478.
23. Banks EH, Miller DA. Perinatal risks associated with borderline amniotic fluid index. Am J Obstet Gynecol. 1999;180:1461-1463.
24. Manning FA, Morrison I, Harman CR, Lange IR, Menticoglou S. Fetal assessment based on fetal biophysical profile scoring: experience in 19,221 referred high-risk pregnancies. II. An analysis of false-negative fetal deaths. Am J Obstet Gynecol. 1987;157:880-884.
25. Alfirevic Z, Neilson JP. Biophysical profile for fetal assessment in high risk pregnancies. Cochrane Database Syst Rev. 2003;(1).:The Cochrane Collaboration.
26. Maulik D, Yarlagadda P, Youngblood JP, et al. The diagnostic efficacy of the UA systolic/diastolic ratio as a screening tool a prospective blinded study. Am J Obstet Gynecol. 1990;162:1518-1523.
27. Nicolaides KH, Bilardo CM, Soothill PW, et al. Absence of end diastolic frequencies in umbilical artery: a sign of fetal hypoxia and acidosis. B Med J. 1988;297:1026-1027.
28. Alfirevic Z, Neilson JP. Doppler ultrasonography in high-risk pregnancies: systematic review with meta-analysis. Am J Obstet Gynecol. 1995;172:1379-1387.
29. Westergaard HB, Langhoff-Roos J, Lingman G, Marsal K, Kreiner S. A critical appraisal of the use of umbilical artery Doppler ultrasound in high-risk pregnancies: use of meta-analyses in evidence-based obstetrics. Ultrasound Obstet Gynecol. 2001;17:464-465.
30. Gulmezoglu AM, Hofmeyr GJ. Bed rest in hospital for suspected impaired fetal growth. Cochrane Database Syst Rev. 2002;(4).-Oxford Update Software.
31. Kramer MS. High protein supplementation in pregnancy. Cochrane Database Syst Rev. 2002;(4).-Oxford Update Software.
32. Kramer MS. Balanced protein/energy supplementation in pregnancy. Cochrane Database Syst Rev. 2002;(4).-Oxford Update Software.
33. Gulmezoglu AM, Hofmeyr GJ. Maternal nutrient supplementation for suspected impaired fetal growth. Cochrane Database Syst Rev. 2002;(4).-Oxford Update Software.
34. Leitich H, Egarter C, Husslein P, Kaider A, Schemper M. A meta-analysis of low dose aspirin for the prevention of intrauterine growth retardation. Br J Obstet Gynaecol. 1997;104:450-459.
35. Kozer E, Costei AM, Boskovic R, Nulman I, Nikfar S, Koren G. Effects of aspirin consumption during pregnancy on pregnancy outcomes: meta-analysis. Birth Defects Res Part B Dev Reprod Toxicol. 2003;68:70-84.
36. The GRIT Study Group. A randomized trial of timed delivery for the compromised preterm fetus: short term outcomes and Bayesian interpretation. BJOG. 2003;110:27-32.
- Antepartum recognition of fetal growth restriction is essential for proper surveillance and management.
- Because growth-restricted fetuses are at risk for adverse outcomes in utero, fetal surveillance is vital for timely recognition and intervention.
- Once growth restriction is identified, management should be individualized to ensure optimal gestational development and safe delivery.
Not only is fetal growth restriction (FGR) associated with perinatal mortality and morbidity, but it may be linked to adverse consequences in adulthood.1 Its many causes involve diverse pathological processes; thus, it should not be considered a single disease. Many aspects remain unclear, a substantial number of affected infants are not identified before birth, and effective antepartum prevention and treatment remain elusive.
Fortunately, managing FGR has hopeful aspects. Meaningful recent advances elucidate its etiologic and pathophysiologic mechanisms and help clarify diagnosis and management.
This article offers an up-to-date, evidence-based approach and includes guidelines on 3 keys to success: antepartum recognition, fetal surveillance, and antepartum and intrapartum management. These guidelines are not intended as a strict protocol, since the clinical course of FGR is highly variable, but as the starting point for individualized care.
An amorphous entity: FGR terminology
Fetal growth restriction implies failure to realize genetically determined growth potential. Terms include fetal growth restriction, intrauterine growth restriction, and small for gestational age (SGA). The pejorative term growth retardation is obsolete.
Traditionally, FGR refers to prenatally identified fetal growth deficiency, whereas SGA refers to suboptimal birth weight for the gestational age. However, some small fetuses are merely constitutionally small, not growth-restricted. Conversely, not all growth-restricted fetuses are small in size or weight for gestational age. Yet defining these groups is difficult, as the tools are imprecise and controversial.
This article uses these definitions:
Fetal growth restriction identified in the antepartum period refers to a fetus with sonographically measured fetal dimensions, particularly abdominal circumference or estimated weight, below an age-specific threshold, typically the 10th percentile.
Fetal growth restriction identified at birth is birth weight below the 10th percentile for gestational age, or SGA. Unfortunately, this definition may fail to identify some fetuses that are truly growth-restricted. Alternative, more sensitive definitions, such as the Ponderal index or birth weight ratio, are used primarily in research.
Consequences of FGR
Perinatal outcomes. Perinatal morbidities include prematurity, oligohydramnios, nonreassuring fetal heart rate patterns with a higher incidence of cesarean delivery, birth asphyxia, low Apgar score, neonatal hypoglycemia, hypocalcemia, polycythemia, hyperbilirubinemia, hypothermia, apnea, seizure disorders, and infection.
Fetal and neonatal mortality is significantly increased. Perinatal mortality is influenced by many factors, including severity of growth restriction, timing of onset, gestational age, and cause of growth restriction. The lower the birth-weight percentile for gestational age, the higher the mortality rate.
Effects in infancy. Although many SGA infants “catch up” growth in infancy, the pattern varies. Height and weight catch-up growth of preterm FGR infants lags behind that of preterm infants that are appropriate for gestational age (AGA) at birth.2 Those with early-onset or severe growth deficit continue to lag behind in postnatal growth. Although recent studies indicate that rapid postnatal growth in SGA infants may lead to increased risk of chronic diseases, including type 2 diabetes, others have found tangible short-term benefits of less frequent morbidity and mortality in infancy.3
FGR has been linked to a spectrum of neurodevelopmental risks, including subtle behavioral abnormalities, immature sleep patterns, decreased visual fixation, decreased general activity, altered early mother-infant interaction, altered motor skills, and hyperactivity.4,5 Infants born SGA at 32 to 42 weeks were 4 to 6 times more likely to have cerebral palsy, yet those whose birth weight was above the 97th percentile also had increased risk. It remains uncertain whether deviant growth is the cause or a consequence of this disability.6
Long-term effects. Lifetime sequelae of early nutritional deprivation have been demonstrated in animals.7 Moreover, epidemiological evidence suggesting an association between SGA at birth or infancy and increased risk of abnormal blood lipid values, diabetes, hypertension, and ischemic heart disease in adult life led Barker and associates to propose the fetal origins hypothesis.1
1. Antepartum recognitionDetermine gestational age
A reliable estimate of gestational age is central to identification of fetal growth compromise in utero or at birth. In pregnancies at risk for fetal growth restriction, gestational age should be established early, preferably in the first trimester.
The method of determining gestational age influences the observed frequency of FGR and SGA births. Estimates are more accurate when based on early ultrasound biometry than on menstrual history. The latter, if well documented, regular, and ovulatory, may be reliable if it differs by no more than 1 week from the sonographic gestational age. Otherwise, early-pregnancy, ultrasoundbased age is more accurate.
Screening for FGR
Screening can be done clinically or by special investigation. The following methods are used, some of which remain experimental:
- assessment of historical clinical risks
- clinical evaluation of fetal and uterine size
- ultrasound fetal biometry
- umbilical arterial and uterine arterial
Doppler ultrasound
Clinical risk assessment. Evaluate all gravidas for risk factors (TABLE 1). If a woman is determined to be at heightened risk, take appropriate steps to diagnose FGR as early as possible.
Clinical evaluation of fetal and uterine size. Clinical assessment of fetal growth includes estimating fetal size by traditional obstetrical manual examination and by measuring the uterine fundal height.
- Abdominal palpation is inadequate to identify the fetus at increased risk of growth restriction, missing 74% of cases.8
- Serial measurements of the uterine fundal height, however, may be more reliable.9 This method consists of measuring fundal height from the symphysis pubis using a nonstretchable tape measure and assessing the results against a gestational age-specific nomogram. Limitations include significant interobserver differences and varying effects of maternal weight, parity, and fetal sex. A randomized trial of the method did not demonstrate any benefits, and a subsequent Cochrane review of the same study considered the evidence insufficient for any recommendations.10,11
Nevertheless, clinical assessment of uterine and fetal size is an essential, inexpensive component of prenatal care and a simple screening tool for identifying mothers who would benefit from further, more definitive sonographic investigation.
Routine ultrasound biometry. The potential benefits of accurately determined gestational age, and recognition of fetal malformations and multiple gestation, via earlyor mid-pregnancy ultrasound are well recognized and justify widespread use.
Routine scanning increases detection of SGA infants.12 A population-based cohort study13 involving over 16,000 singleton pregnancies found that fetuses smaller than expected at mid-second trimester ultrasound (discrepancy exceeding 14 days) were at increased risk for adverse outcomes, including perinatal mortality and SGA.
Unfortunately, these findings have not led to improved outcomes. A high false-positive rate remains a major concern. A prospective observational study14 found that routine ultrasound did not identify most cases of FGR, but resulted in a fivefold increase in iatrogenic premature delivery and significantly increased neonatal intensive care admissions.
- The Routine Antenatal Diagnostic Imaging with Ultrasound (RADIUS) trial15 randomized 15,000 low-risk gravidas to routine ultrasound imaging (at 15 to 22 weeks and again at 31 to 35 weeks) or to ultrasound only when indicated. The groups had similar rates of adverse perinatal outcome, distribution of birth weights, and preterm delivery. The trial’s weaknesses include selection criteria for low risk that excluded most pregnancies, inappropriate perinatal-outcome parameters, and suboptimal ultrasonographer expertise.
- A recent Cochrane review16 of 7 trials involving more than 25,000 women failed to demonstrate any improvements in perinatal mortality and morbidity with routine ultrasound, or any difference in antenatal, obstetric, and neonatal interventions.
Estimated fetal weight may differ from actual weight by as much as 20%.
We also lack evidence regarding longterm outcomes such as neurodevelopment.
Umbilical arterial Doppler. A meta-analysis17 of 4 randomized trials in unselected or low-risk pregnancies with a total population of 11,375 women found no effect of screening Doppler umbilical artery velocimetry on perinatal death, stillbirth, antenatal hospitalization, obstetric outcome, or perinatal morbidity.
A subsequent meta-analysis18 of 5 trials of routine Doppler ultrasound in unselected and low-risk pregnancies with a total population of more than 14,000 women also found no benefit for mother or infant.
We lack evidence on long-term outcomes.
Uterine arterial Doppler screening. Increased pulsatility of the uterine arterial Doppler waveform, persistence of the notch, and a significant difference between right and left uterine arteries have been associated with FGR, pregnancy-induced hypertension, and adverse perinatal outcome. A review19 of 15 studies of routine uterine Doppler in unselected populations showed considerable heterogeneity, but indicated that increased impedance in the uterine arteries identifies about 20% of those who develop FGR, with a positive likelihood ratio of 3.5.
We lack exclusive randomized trials of routine uterine Doppler sonography in unselected and low-risk pregnancies. However, 2 studies done in conjunction with umbilical arterial Doppler found no impact on outcome, and a recent Cochrane review18 found insufficient evidence to support routine uterine Doppler for FGR screening.
TABLE 1
Risk factors
| MATERNAL FACTORS |
| Medical disease |
| Preeclampsia/hypertension |
| Renal disease |
| Antiphospholipid antibody syndrome |
| Inherited thrombophilia |
| Diabetes with vasculopathy |
| Cyanotic heart disease |
| Asthma |
| Hemoglobinopathy |
| Phenylketonuria |
| Life circumstance |
| Severe malnutrition |
| Smoking |
| Substance abuse (eg, alcohol, heroine, cocaine) |
| PLACENTAL FACTORS |
| Confined placental mosaic |
| Placenta previa |
| Abruptio placentae |
| Infarction |
| Circumvallate placenta |
| Placenta accreta |
| Hemangioma |
| FETAL FACTORS |
| Multiple gestation |
| Prematurity |
| Unexplained elevated alpha-fetoprotein |
| Infection (eg, rubella, cytomegalovirus, herpes, malaria, toxoplasmosis) |
| Malformations (eg, gastroschisis, omphalocele, diaphragmatic hernia, congenital heart defect) |
| Genetic disorders (eg, trisomy 13, 18, and 21; triploidy; some cases of Turner’s syndrome) |
Identifying FGR in utero
Antepartum diagnosis is based on sonographic measurement of various fetal dimensions.
Abdominal circumference and estimated fetal weight. A review20 of 60 studies found that abdominal circumference (AC) and sonographically estimated fetal weight (EFW) were the best predictors of birth weight below the 10th percentile in high-risk pregnancies. AC below the 10th percentile had sensitivities ranging from 72.9% to 94.5%, false-positive rates of 16.2% to 49.4%, and a common odds ratio of 18.4. An EFW below the 10th percentile had a common odds ratio of 39.1, which was the highest among all the biometric measurements. Its sensitivity ranged from 33.3% to 89.2%, and false-positive rates ranged from 9.1% to 46.3%.
The proportionality of fetal dimensions, such as the head/abdominal ratio, was not a good predictor, although it is routinely generated in ultrasound biometry.
Ultrasound estimation of fetal weight is based on a combination of 2 or 3 biometric measurements of the fetus, including the biparietal diameter (BPD) or the head circumference (HC), AC, and femur length (FL). Several formulae yield varying estimations of weight. Thus, a fetus identified as growth-restricted by 1 formula may not be so diagnosed by another. For this reason, it is prudent to be consistent in their use. EFW generated from measurements of the head (BPD or HC), AC, and FL is most reliable.21
The EFW also is expressed as the percentile for the gestational age.
Limitations of ultrasound estimation.
The optimal process of translating dimensional measurements into fetal body mass for both health and disease remains unknown. This leads to inaccurate assumptions and erroneous weight projections.
Inaccuracies also result from variations in measurement. In addition, the estimation is less accurate in the lower and upper ranges of fetal weight distribution and in the presence of oligohydramnios. As a result, estimated weight may differ from actual weight by as much as 20%, with greater margins of error at the lower and upper extremes.
Guidelines for screening and diagnosis
Assess all pregnancies for risk factors and determine the gestational age in early pregnancy, especially in women at higher risk of FGR. If the patient has substantial clinical risks or there is suspicion of growth restriction, fetal ultrasound biometry is recommended. Diagnosis of FGR is based on fetal sonographic measurements, especially AC; estimated fetal weight derived from BPD or HC, AC and FL; and longitudinal progression of fetal growth.
Current evidence suggests the use of a 10th percentile diagnostic threshold for these measurements. Follow the biometric parameters longitudinally, repeating the measurement every 2 to 4 weeks. More frequent examination is unreliable.
2. Fetal surveillanceAntepartum monitoring
Fetuses identified as growth-restricted are at risk for adverse outcomes in utero. Thus, antepartum monitoring is indicated for timely recognition and intervention.
Tests commonly used include the nonstress test (NST), biophysical profile (BPP), sonographic estimation of amniotic fluid volume, and Doppler velocimetry of the umbilical artery or other vessels of interest.
With progressive fetal compromise, compensatory mechanisms may be insufficient to maintain fetal homeostasis. The sequence of loss of compensatory mechanisms is reflected in surveillance tests. Thus, changes in umbil-ical arterial Doppler indices usually precede nonreassuring NST or BPP.
Nonstress test: The standard of care. The NST is probably the most common fetal surveillance test in high-risk pregnancies, including those complicated by FGR. However, it is personnel- and time-intensive, and interpretation often is subjective.
A nonreactive NST is associated with adverse perinatal outcome, although it more commonly is related to the fetal sleep cycle.
In predicting a potentially preventable fetal death, the NST has a false-negative rate of 2 to 3 per 1,000, a negative predictive value of 99.8%, and a false-positive rate of 80%.
Evidence of its efficacy is lacking. Four randomized trials in the early 1980s, which involved more than 1,500 women, found no improvement in outcomes. (All 4 trials lacked sufficient power.)
- When to test. Despite these limitations, the NST has become an integral part of obstetri-cal practice and remains a standard of care for antepartum fetal surveillance in high-risk pregnancies. It should be used as a surveillance tool for the growth-restricted fetus. The test initially is performed weekly.
Depending on severity of growth restriction, testing may increase to twice weekly or even daily.
Amniotic fluid volume monitoring is warranted. Oligohydramnios—highly prevalent in FGR—is associated with adverse perina-tal outcomes.
Although the sonographically determined amniotic fluid index (AFI) is the most frequently used modality to evaluate amniotic fluid volume, its accuracy has been questioned. Nevertheless, a meta-analysis22 of 18 studies involving more than 10,000 patients showed that antepartum AFI equal to or below 5 cm was associated with an increased risk of cesarean section for fetal distress and an Apgar score below 7 at 5 minutes.
Even marginal AFI values (5–10 cm) have been associated with doubling of the risk of adverse perinatal outcome.23
- When to test. We lack randomized trials of amniotic fluid volume assessment in high-risk pregnancies, but observational studies suggest that weekly determination of AFI is helpful in sonographically identified growth-restricted fetuses.
A low or marginal AFI should be followed by more frequent examinations. Even a marginal AFI may be followed by twice-weekly surveillance.
Biophysical profile effectively predicts adverse perinatal outcome, with a false-negative rate of 0.8 per 1,000, negative predictive value of 99.9%, and a false-positive rate of 40% to 50%.24 A low BPP is associated with fetal hypoxia and acidosis, and a declining BPP reflects progressive worsening of the fetal condition.
In FGR, a significant association exists between abnormal Doppler indices and fetal acid-base compromise.
Although equal weight was originally accorded all the parameters of the profile, experience suggests that the independent risk of oligohydramnios warrants immediate reassessment of the management plan.
Like the nonstress test, the BPP has a low false-negative rate and a high false-positive rate. Four randomized trials of the test have been conducted, involving about 2,800 patients, with no confirmation of its effectiveness. A Cochrane systematic review25 concluded that this cumulative sample size is insufficient to draw definitive conclusions.
The test remains a standard, however, and is recommended to confirm fetal wellbeing or as part of a comprehensive test panel in significantly high-risk pregnancies.
- When to test. Initially perform the test once a week, increasing to twice weekly when the severity of growth restriction warrants.
Daily testing may be indicated in complications such as severe growth restriction and pregestational diabetes. In early preterm pregnancies (less than 32 weeks) with nonreassuring NST or umbilical arterial Doppler, BPP can help guide optimal management.
Umbilical arterial Doppler ultrasound is a powerful predictor of adverse perinatal outcomes in high-risk pregnancies.
Doppler indices include the pulsatility index, systolic/diastolic ratio, diastolic average ratio, and the resistance index. Of these, the last demonstrated the greatest ability to predict abnormal perinatal outcomes.
A prospective, blinded study26 in highrisk pregnancies demonstrated significant diagnostic efficacy with a sensitivity of 79%, specificity of 93%, positive predictive value of 83%, negative predictive value of 91%, and kappa index of 73%. The last value is consistent with a good to excellent diagnostic test.
A significant association also exists between abnormal Doppler indices and fetal acid-base compromise in FGR. The absence of enddiastolic flow (FIGURE) is associated with markedly adverse perinatal outcome, particularly a high perinatal mortality rate, malformations, and aneuploidy. Nicolaides and coworkers27 found that, when the end-diastolic velocity was absent, 67% to 80% of the fetuses were hypoxic and 45% were acidotic. Reversal of end-diastolic flow is associated with an even worse prognosis. The adverse consequences of absent or reverse end-diastolic flow are listed in TABLE 2.
In contrast to the NST and BPP, the effectiveness of fetal surveillance with umbilical arterial Doppler ultrasound in improving perinatal outcome in high-risk pregnancies has been confirmed by extensive randomized trials and their meta-analyses.28,29 These studies provide compelling evidence that, when used in well-defined high-risk pregnancies, especially those involving FGR or preeclampsia, umbilical arterial Doppler sonography reduces perinatal death, cesarean delivery for fetal distress, elective deliveries, and antenatal admissions.
Doppler sonography of the fetal cerebral and venous circulation also helps identify progressive fetal compromise in high-risk pregnancies, including those with FGR. When the fetal condition is deteriorating, cerebral arteries dilate to compensate, and pulsations appear in the umbilical vein with reversal of flow in the ductus venous.
These signs of grave fetal status indicate immediate delivery.
FIGURE 1 Duplex pulsed Doppler
Sonogram showing absent end-diastolic flow in the umbilical artery of a growth-restricted fetus.TABLE 2
Perinatal outcomes for absent and reversed end-diastolic velocity in the umbilical artery
| PERINATAL OUTCOME | MEAN | RANGE |
|---|---|---|
| Mortality | 45% | 17–100% |
| Gestational age | 31.6 weeks | 29–33 weeks |
| Birth weight | 1,056 g | 910–1,481 g |
| SGA | 68% | 53–100% |
| Cesarean section for fetal distress | 73% | 24–100% |
| Apgar score <7 at 5 minutes | 26% | 7–69% |
| Admission to NICU | 84% | 77–97% |
| Congenital anomalies | 10% | 0–24% |
| Aneuploidy | 6.4% | 0–18% |
| SGA = small for gestational age; NICU = neonatal intensive care unit | ||
| Reprinted with permission from: Maulik D, ed. Doppler Ultrasound in Obstetrics and Gynecology. | ||
| New York: Springer-Verlag; 1997:364, Table 21.2. | ||
Assessing the fetus for malformation and aneuploidy
Because of the association between FGR and fetal malformations and aneuploidy, the fetus should be assessed for these complications, especially when growth restriction is severe, develops early, and is not associated with hypertensive disorders, oligohydramnios, or abnormal umbilical arterial Doppler indices.
Most etiologies are either not amenable to therapy or fetal growth is not improved by treatments that benefit the mother.
The current standard practice is aneuploidy screening in early pregnancy and fetal anatomical scanning at midgestation. If significant risks of aneuploidy are present, a fetal karyotype is recommended, along with appropriate counseling.
3. Individualized managementAntepartum treatment
The question of bed rest. At present, there are no grounds for recommending strict bed rest in FGR cases. A Cochrane review30 showed no improvements with hospitalized bed rest compared with ambulation. Prolonged rest increases risk of thromboembolism and can be costly and inconvenient.
Nutritional remedies. Of the options that have been tested, which include high protein supplementation, nutrient supplementation such as beef liver extract, and balanced energy/protein supplementation, only the last (with protein content comprising less than 25% of total energy content) has led to a significant reduction in SGA births.31-33
Other approaches. We lack reliable evidence that other interventions such as oxygen administration, abdominal decompression, and pharmacological agents, including calcium channel blockers, beta mimetics, and magnesium, are beneficial or effective in improving or preventing FGR.
A meta-analysis34 of 13 trials involving more than 13,000 women showed that early aspirin treatment reduced the risk of FGR but failed to improve outcome. A more recent meta-analysis35 of 38 trials of aspirin in highrisk pregnancies found no reduction in the incidence of FGR or perinatal death, although a reduction in risk of preterm births was noted.
Etiologic management
Most etiologic conditions are either not amenable to therapy or fetal growth is not improved by treatments that benefit the mother. An example is maternal hypertensive disease, in which the indicated treatment has no beneficial effect on fetal growth.
Treatment of poor lifestyle habits may be helpful. If the mother smokes, vigorous smoking-cessation education and counseling is urged. Also address alcohol consumption and other substance abuse, and offer remedial measures.
The diagnosis of fetal viral and parasitic infections is important for prognostication and neonatal management. Although few perinatal infections are treatable in utero, maternal therapy may prevent certain infections from spreading to the fetus. Examples include toxoplasmosis and malaria.
If lethal malformations or lethal aneuploidy are identified prenatally, avoid fetal surveillance and unnecessary intervention, which may expose the mother to unnecessary and unjustifiable risks.
Fetal surveillance strategy
If fetal biometry indicates fetal weight below the 10th percentile, begin fetal surveillance. The current standard, umbilical arterial Doppler sonography, is the primary test in sonographically documented growth restriction. Also assess amniotic fluid volume—as part of the BPP or independently. AFI is the most commonly used BPP tool.
In the United States, the nonstress test traditionally is the primary monitoring modality, with the BPP as backup, although the BPP—which includes the NST—has been recommended as the primary test.
Normal Doppler findings. When the Doppler index remains within normal limits or does not progressively rise, weekly testing should suffice, with the NST or BPP as backup or in conjunction with Doppler.
If fetal and maternal conditions remain reassuring, allow the pregnancy to continue to maturity and assess the patient for delivery. Postdate pregnancy is not advised in the presence of sonographically confirmed growth compromise.
A high or increasing Doppler index warrants more intensive fetal surveillance consisting of weekly umbilical arterial Doppler and once- or twice-weekly NST and BPP until fetal maturity.
If these tests indicate fetal compromise, or absent end-diastolic velocity develops, the likelihood of poor perinatal outcome is increased and an urgent clinical response is indicated. Hospitalize the patient and individualize management, depending on gestational age and fetal status.
Optimal timing of delivery
The optimal timing of delivery in a preterm pregnancy with FGR is unclear. A recent multicenter randomized controlled trial, the Growth Restriction Intervention Trial (GRIT),36 compared 2 strategies: delivery within 48 hours with steroid administration or delivery delayed as long as fetal status permits. The population consisted of high-risk gravidas between 24 and 36 weeks’ gestation. More than 90% of the women had pregnancies complicated by FGR. No significant differences were noted between the 2 groups in stillbirth rates.
Fortunately, thanks to recent advances in perinatal care, a management strategy can be recommended:
At or near term (34 weeks or beyond), the absence of end-diastolic flow in the umbilical artery should prompt consideration of immediate delivery.
Other ominous findings that prompt such consideration include:
- Cessation of fetal growth on successive ultrasound examinations
- Progression of umbilical arterial absent enddiastolic flow to reversed end-diastolic flow
- Nonreassuring heart rate patterns including nonreactive NST, poor fetal heart rate baseline variability, and persistent variable or late decelerations
- Oligohydramnios
- BPP score of 4 or below
Less than 34 weeks. When absent enddiastolic flow develops in a preterm pregnancy with a significant risk of fetal lung immaturity, seek further assurance of fetal well-being via daily surveillance with umbilical arterial Doppler sonography, nonstress test, and biophysical profile. Administer betamethasone to enhance fetal lung maturity. Delivery is indicated regardless of maturity when a single test or combination indicates imminent fetal danger and the fetal risk from a hostile intrauterine environment is judged to be greater than that from pulmonary immaturity.
Intrapartum management
Labor and delivery of growth-restricted infants often is associated with a higher risk of asphyxia. The frequency of nonreassuring fetal heart rate patterns is increased. In addition, because of the greater prevalence of oligohydramnios, cord compression is frequent and associated with variable decelerations. There also is an increased risk of neonatal morbidities, as discussed above. Because of these risks, the growth-restricted fetus should be delivered in a facility that can offer appropriate inhouse support services such as neonatology and anesthesia.
If the patient is allowed to labor, use continuous electronic fetal heart rate monitoring with vigilance and perform the appropriate secondary tests, such as fetal scalp sampling or scalp stimulation. The ability of a growth-restricted fetus to tolerate labor is likely very limited.
Any confirmed signs of fetal compromise indicate cesarean as the optimal mode of delivery.
The authors report no financial relationships relevant to this article.
- Antepartum recognition of fetal growth restriction is essential for proper surveillance and management.
- Because growth-restricted fetuses are at risk for adverse outcomes in utero, fetal surveillance is vital for timely recognition and intervention.
- Once growth restriction is identified, management should be individualized to ensure optimal gestational development and safe delivery.
Not only is fetal growth restriction (FGR) associated with perinatal mortality and morbidity, but it may be linked to adverse consequences in adulthood.1 Its many causes involve diverse pathological processes; thus, it should not be considered a single disease. Many aspects remain unclear, a substantial number of affected infants are not identified before birth, and effective antepartum prevention and treatment remain elusive.
Fortunately, managing FGR has hopeful aspects. Meaningful recent advances elucidate its etiologic and pathophysiologic mechanisms and help clarify diagnosis and management.
This article offers an up-to-date, evidence-based approach and includes guidelines on 3 keys to success: antepartum recognition, fetal surveillance, and antepartum and intrapartum management. These guidelines are not intended as a strict protocol, since the clinical course of FGR is highly variable, but as the starting point for individualized care.
An amorphous entity: FGR terminology
Fetal growth restriction implies failure to realize genetically determined growth potential. Terms include fetal growth restriction, intrauterine growth restriction, and small for gestational age (SGA). The pejorative term growth retardation is obsolete.
Traditionally, FGR refers to prenatally identified fetal growth deficiency, whereas SGA refers to suboptimal birth weight for the gestational age. However, some small fetuses are merely constitutionally small, not growth-restricted. Conversely, not all growth-restricted fetuses are small in size or weight for gestational age. Yet defining these groups is difficult, as the tools are imprecise and controversial.
This article uses these definitions:
Fetal growth restriction identified in the antepartum period refers to a fetus with sonographically measured fetal dimensions, particularly abdominal circumference or estimated weight, below an age-specific threshold, typically the 10th percentile.
Fetal growth restriction identified at birth is birth weight below the 10th percentile for gestational age, or SGA. Unfortunately, this definition may fail to identify some fetuses that are truly growth-restricted. Alternative, more sensitive definitions, such as the Ponderal index or birth weight ratio, are used primarily in research.
Consequences of FGR
Perinatal outcomes. Perinatal morbidities include prematurity, oligohydramnios, nonreassuring fetal heart rate patterns with a higher incidence of cesarean delivery, birth asphyxia, low Apgar score, neonatal hypoglycemia, hypocalcemia, polycythemia, hyperbilirubinemia, hypothermia, apnea, seizure disorders, and infection.
Fetal and neonatal mortality is significantly increased. Perinatal mortality is influenced by many factors, including severity of growth restriction, timing of onset, gestational age, and cause of growth restriction. The lower the birth-weight percentile for gestational age, the higher the mortality rate.
Effects in infancy. Although many SGA infants “catch up” growth in infancy, the pattern varies. Height and weight catch-up growth of preterm FGR infants lags behind that of preterm infants that are appropriate for gestational age (AGA) at birth.2 Those with early-onset or severe growth deficit continue to lag behind in postnatal growth. Although recent studies indicate that rapid postnatal growth in SGA infants may lead to increased risk of chronic diseases, including type 2 diabetes, others have found tangible short-term benefits of less frequent morbidity and mortality in infancy.3
FGR has been linked to a spectrum of neurodevelopmental risks, including subtle behavioral abnormalities, immature sleep patterns, decreased visual fixation, decreased general activity, altered early mother-infant interaction, altered motor skills, and hyperactivity.4,5 Infants born SGA at 32 to 42 weeks were 4 to 6 times more likely to have cerebral palsy, yet those whose birth weight was above the 97th percentile also had increased risk. It remains uncertain whether deviant growth is the cause or a consequence of this disability.6
Long-term effects. Lifetime sequelae of early nutritional deprivation have been demonstrated in animals.7 Moreover, epidemiological evidence suggesting an association between SGA at birth or infancy and increased risk of abnormal blood lipid values, diabetes, hypertension, and ischemic heart disease in adult life led Barker and associates to propose the fetal origins hypothesis.1
1. Antepartum recognitionDetermine gestational age
A reliable estimate of gestational age is central to identification of fetal growth compromise in utero or at birth. In pregnancies at risk for fetal growth restriction, gestational age should be established early, preferably in the first trimester.
The method of determining gestational age influences the observed frequency of FGR and SGA births. Estimates are more accurate when based on early ultrasound biometry than on menstrual history. The latter, if well documented, regular, and ovulatory, may be reliable if it differs by no more than 1 week from the sonographic gestational age. Otherwise, early-pregnancy, ultrasoundbased age is more accurate.
Screening for FGR
Screening can be done clinically or by special investigation. The following methods are used, some of which remain experimental:
- assessment of historical clinical risks
- clinical evaluation of fetal and uterine size
- ultrasound fetal biometry
- umbilical arterial and uterine arterial
Doppler ultrasound
Clinical risk assessment. Evaluate all gravidas for risk factors (TABLE 1). If a woman is determined to be at heightened risk, take appropriate steps to diagnose FGR as early as possible.
Clinical evaluation of fetal and uterine size. Clinical assessment of fetal growth includes estimating fetal size by traditional obstetrical manual examination and by measuring the uterine fundal height.
- Abdominal palpation is inadequate to identify the fetus at increased risk of growth restriction, missing 74% of cases.8
- Serial measurements of the uterine fundal height, however, may be more reliable.9 This method consists of measuring fundal height from the symphysis pubis using a nonstretchable tape measure and assessing the results against a gestational age-specific nomogram. Limitations include significant interobserver differences and varying effects of maternal weight, parity, and fetal sex. A randomized trial of the method did not demonstrate any benefits, and a subsequent Cochrane review of the same study considered the evidence insufficient for any recommendations.10,11
Nevertheless, clinical assessment of uterine and fetal size is an essential, inexpensive component of prenatal care and a simple screening tool for identifying mothers who would benefit from further, more definitive sonographic investigation.
Routine ultrasound biometry. The potential benefits of accurately determined gestational age, and recognition of fetal malformations and multiple gestation, via earlyor mid-pregnancy ultrasound are well recognized and justify widespread use.
Routine scanning increases detection of SGA infants.12 A population-based cohort study13 involving over 16,000 singleton pregnancies found that fetuses smaller than expected at mid-second trimester ultrasound (discrepancy exceeding 14 days) were at increased risk for adverse outcomes, including perinatal mortality and SGA.
Unfortunately, these findings have not led to improved outcomes. A high false-positive rate remains a major concern. A prospective observational study14 found that routine ultrasound did not identify most cases of FGR, but resulted in a fivefold increase in iatrogenic premature delivery and significantly increased neonatal intensive care admissions.
- The Routine Antenatal Diagnostic Imaging with Ultrasound (RADIUS) trial15 randomized 15,000 low-risk gravidas to routine ultrasound imaging (at 15 to 22 weeks and again at 31 to 35 weeks) or to ultrasound only when indicated. The groups had similar rates of adverse perinatal outcome, distribution of birth weights, and preterm delivery. The trial’s weaknesses include selection criteria for low risk that excluded most pregnancies, inappropriate perinatal-outcome parameters, and suboptimal ultrasonographer expertise.
- A recent Cochrane review16 of 7 trials involving more than 25,000 women failed to demonstrate any improvements in perinatal mortality and morbidity with routine ultrasound, or any difference in antenatal, obstetric, and neonatal interventions.
Estimated fetal weight may differ from actual weight by as much as 20%.
We also lack evidence regarding longterm outcomes such as neurodevelopment.
Umbilical arterial Doppler. A meta-analysis17 of 4 randomized trials in unselected or low-risk pregnancies with a total population of 11,375 women found no effect of screening Doppler umbilical artery velocimetry on perinatal death, stillbirth, antenatal hospitalization, obstetric outcome, or perinatal morbidity.
A subsequent meta-analysis18 of 5 trials of routine Doppler ultrasound in unselected and low-risk pregnancies with a total population of more than 14,000 women also found no benefit for mother or infant.
We lack evidence on long-term outcomes.
Uterine arterial Doppler screening. Increased pulsatility of the uterine arterial Doppler waveform, persistence of the notch, and a significant difference between right and left uterine arteries have been associated with FGR, pregnancy-induced hypertension, and adverse perinatal outcome. A review19 of 15 studies of routine uterine Doppler in unselected populations showed considerable heterogeneity, but indicated that increased impedance in the uterine arteries identifies about 20% of those who develop FGR, with a positive likelihood ratio of 3.5.
We lack exclusive randomized trials of routine uterine Doppler sonography in unselected and low-risk pregnancies. However, 2 studies done in conjunction with umbilical arterial Doppler found no impact on outcome, and a recent Cochrane review18 found insufficient evidence to support routine uterine Doppler for FGR screening.
TABLE 1
Risk factors
| MATERNAL FACTORS |
| Medical disease |
| Preeclampsia/hypertension |
| Renal disease |
| Antiphospholipid antibody syndrome |
| Inherited thrombophilia |
| Diabetes with vasculopathy |
| Cyanotic heart disease |
| Asthma |
| Hemoglobinopathy |
| Phenylketonuria |
| Life circumstance |
| Severe malnutrition |
| Smoking |
| Substance abuse (eg, alcohol, heroine, cocaine) |
| PLACENTAL FACTORS |
| Confined placental mosaic |
| Placenta previa |
| Abruptio placentae |
| Infarction |
| Circumvallate placenta |
| Placenta accreta |
| Hemangioma |
| FETAL FACTORS |
| Multiple gestation |
| Prematurity |
| Unexplained elevated alpha-fetoprotein |
| Infection (eg, rubella, cytomegalovirus, herpes, malaria, toxoplasmosis) |
| Malformations (eg, gastroschisis, omphalocele, diaphragmatic hernia, congenital heart defect) |
| Genetic disorders (eg, trisomy 13, 18, and 21; triploidy; some cases of Turner’s syndrome) |
Identifying FGR in utero
Antepartum diagnosis is based on sonographic measurement of various fetal dimensions.
Abdominal circumference and estimated fetal weight. A review20 of 60 studies found that abdominal circumference (AC) and sonographically estimated fetal weight (EFW) were the best predictors of birth weight below the 10th percentile in high-risk pregnancies. AC below the 10th percentile had sensitivities ranging from 72.9% to 94.5%, false-positive rates of 16.2% to 49.4%, and a common odds ratio of 18.4. An EFW below the 10th percentile had a common odds ratio of 39.1, which was the highest among all the biometric measurements. Its sensitivity ranged from 33.3% to 89.2%, and false-positive rates ranged from 9.1% to 46.3%.
The proportionality of fetal dimensions, such as the head/abdominal ratio, was not a good predictor, although it is routinely generated in ultrasound biometry.
Ultrasound estimation of fetal weight is based on a combination of 2 or 3 biometric measurements of the fetus, including the biparietal diameter (BPD) or the head circumference (HC), AC, and femur length (FL). Several formulae yield varying estimations of weight. Thus, a fetus identified as growth-restricted by 1 formula may not be so diagnosed by another. For this reason, it is prudent to be consistent in their use. EFW generated from measurements of the head (BPD or HC), AC, and FL is most reliable.21
The EFW also is expressed as the percentile for the gestational age.
Limitations of ultrasound estimation.
The optimal process of translating dimensional measurements into fetal body mass for both health and disease remains unknown. This leads to inaccurate assumptions and erroneous weight projections.
Inaccuracies also result from variations in measurement. In addition, the estimation is less accurate in the lower and upper ranges of fetal weight distribution and in the presence of oligohydramnios. As a result, estimated weight may differ from actual weight by as much as 20%, with greater margins of error at the lower and upper extremes.
Guidelines for screening and diagnosis
Assess all pregnancies for risk factors and determine the gestational age in early pregnancy, especially in women at higher risk of FGR. If the patient has substantial clinical risks or there is suspicion of growth restriction, fetal ultrasound biometry is recommended. Diagnosis of FGR is based on fetal sonographic measurements, especially AC; estimated fetal weight derived from BPD or HC, AC and FL; and longitudinal progression of fetal growth.
Current evidence suggests the use of a 10th percentile diagnostic threshold for these measurements. Follow the biometric parameters longitudinally, repeating the measurement every 2 to 4 weeks. More frequent examination is unreliable.
2. Fetal surveillanceAntepartum monitoring
Fetuses identified as growth-restricted are at risk for adverse outcomes in utero. Thus, antepartum monitoring is indicated for timely recognition and intervention.
Tests commonly used include the nonstress test (NST), biophysical profile (BPP), sonographic estimation of amniotic fluid volume, and Doppler velocimetry of the umbilical artery or other vessels of interest.
With progressive fetal compromise, compensatory mechanisms may be insufficient to maintain fetal homeostasis. The sequence of loss of compensatory mechanisms is reflected in surveillance tests. Thus, changes in umbil-ical arterial Doppler indices usually precede nonreassuring NST or BPP.
Nonstress test: The standard of care. The NST is probably the most common fetal surveillance test in high-risk pregnancies, including those complicated by FGR. However, it is personnel- and time-intensive, and interpretation often is subjective.
A nonreactive NST is associated with adverse perinatal outcome, although it more commonly is related to the fetal sleep cycle.
In predicting a potentially preventable fetal death, the NST has a false-negative rate of 2 to 3 per 1,000, a negative predictive value of 99.8%, and a false-positive rate of 80%.
Evidence of its efficacy is lacking. Four randomized trials in the early 1980s, which involved more than 1,500 women, found no improvement in outcomes. (All 4 trials lacked sufficient power.)
- When to test. Despite these limitations, the NST has become an integral part of obstetri-cal practice and remains a standard of care for antepartum fetal surveillance in high-risk pregnancies. It should be used as a surveillance tool for the growth-restricted fetus. The test initially is performed weekly.
Depending on severity of growth restriction, testing may increase to twice weekly or even daily.
Amniotic fluid volume monitoring is warranted. Oligohydramnios—highly prevalent in FGR—is associated with adverse perina-tal outcomes.
Although the sonographically determined amniotic fluid index (AFI) is the most frequently used modality to evaluate amniotic fluid volume, its accuracy has been questioned. Nevertheless, a meta-analysis22 of 18 studies involving more than 10,000 patients showed that antepartum AFI equal to or below 5 cm was associated with an increased risk of cesarean section for fetal distress and an Apgar score below 7 at 5 minutes.
Even marginal AFI values (5–10 cm) have been associated with doubling of the risk of adverse perinatal outcome.23
- When to test. We lack randomized trials of amniotic fluid volume assessment in high-risk pregnancies, but observational studies suggest that weekly determination of AFI is helpful in sonographically identified growth-restricted fetuses.
A low or marginal AFI should be followed by more frequent examinations. Even a marginal AFI may be followed by twice-weekly surveillance.
Biophysical profile effectively predicts adverse perinatal outcome, with a false-negative rate of 0.8 per 1,000, negative predictive value of 99.9%, and a false-positive rate of 40% to 50%.24 A low BPP is associated with fetal hypoxia and acidosis, and a declining BPP reflects progressive worsening of the fetal condition.
In FGR, a significant association exists between abnormal Doppler indices and fetal acid-base compromise.
Although equal weight was originally accorded all the parameters of the profile, experience suggests that the independent risk of oligohydramnios warrants immediate reassessment of the management plan.
Like the nonstress test, the BPP has a low false-negative rate and a high false-positive rate. Four randomized trials of the test have been conducted, involving about 2,800 patients, with no confirmation of its effectiveness. A Cochrane systematic review25 concluded that this cumulative sample size is insufficient to draw definitive conclusions.
The test remains a standard, however, and is recommended to confirm fetal wellbeing or as part of a comprehensive test panel in significantly high-risk pregnancies.
- When to test. Initially perform the test once a week, increasing to twice weekly when the severity of growth restriction warrants.
Daily testing may be indicated in complications such as severe growth restriction and pregestational diabetes. In early preterm pregnancies (less than 32 weeks) with nonreassuring NST or umbilical arterial Doppler, BPP can help guide optimal management.
Umbilical arterial Doppler ultrasound is a powerful predictor of adverse perinatal outcomes in high-risk pregnancies.
Doppler indices include the pulsatility index, systolic/diastolic ratio, diastolic average ratio, and the resistance index. Of these, the last demonstrated the greatest ability to predict abnormal perinatal outcomes.
A prospective, blinded study26 in highrisk pregnancies demonstrated significant diagnostic efficacy with a sensitivity of 79%, specificity of 93%, positive predictive value of 83%, negative predictive value of 91%, and kappa index of 73%. The last value is consistent with a good to excellent diagnostic test.
A significant association also exists between abnormal Doppler indices and fetal acid-base compromise in FGR. The absence of enddiastolic flow (FIGURE) is associated with markedly adverse perinatal outcome, particularly a high perinatal mortality rate, malformations, and aneuploidy. Nicolaides and coworkers27 found that, when the end-diastolic velocity was absent, 67% to 80% of the fetuses were hypoxic and 45% were acidotic. Reversal of end-diastolic flow is associated with an even worse prognosis. The adverse consequences of absent or reverse end-diastolic flow are listed in TABLE 2.
In contrast to the NST and BPP, the effectiveness of fetal surveillance with umbilical arterial Doppler ultrasound in improving perinatal outcome in high-risk pregnancies has been confirmed by extensive randomized trials and their meta-analyses.28,29 These studies provide compelling evidence that, when used in well-defined high-risk pregnancies, especially those involving FGR or preeclampsia, umbilical arterial Doppler sonography reduces perinatal death, cesarean delivery for fetal distress, elective deliveries, and antenatal admissions.
Doppler sonography of the fetal cerebral and venous circulation also helps identify progressive fetal compromise in high-risk pregnancies, including those with FGR. When the fetal condition is deteriorating, cerebral arteries dilate to compensate, and pulsations appear in the umbilical vein with reversal of flow in the ductus venous.
These signs of grave fetal status indicate immediate delivery.
FIGURE 1 Duplex pulsed Doppler
Sonogram showing absent end-diastolic flow in the umbilical artery of a growth-restricted fetus.TABLE 2
Perinatal outcomes for absent and reversed end-diastolic velocity in the umbilical artery
| PERINATAL OUTCOME | MEAN | RANGE |
|---|---|---|
| Mortality | 45% | 17–100% |
| Gestational age | 31.6 weeks | 29–33 weeks |
| Birth weight | 1,056 g | 910–1,481 g |
| SGA | 68% | 53–100% |
| Cesarean section for fetal distress | 73% | 24–100% |
| Apgar score <7 at 5 minutes | 26% | 7–69% |
| Admission to NICU | 84% | 77–97% |
| Congenital anomalies | 10% | 0–24% |
| Aneuploidy | 6.4% | 0–18% |
| SGA = small for gestational age; NICU = neonatal intensive care unit | ||
| Reprinted with permission from: Maulik D, ed. Doppler Ultrasound in Obstetrics and Gynecology. | ||
| New York: Springer-Verlag; 1997:364, Table 21.2. | ||
Assessing the fetus for malformation and aneuploidy
Because of the association between FGR and fetal malformations and aneuploidy, the fetus should be assessed for these complications, especially when growth restriction is severe, develops early, and is not associated with hypertensive disorders, oligohydramnios, or abnormal umbilical arterial Doppler indices.
Most etiologies are either not amenable to therapy or fetal growth is not improved by treatments that benefit the mother.
The current standard practice is aneuploidy screening in early pregnancy and fetal anatomical scanning at midgestation. If significant risks of aneuploidy are present, a fetal karyotype is recommended, along with appropriate counseling.
3. Individualized managementAntepartum treatment
The question of bed rest. At present, there are no grounds for recommending strict bed rest in FGR cases. A Cochrane review30 showed no improvements with hospitalized bed rest compared with ambulation. Prolonged rest increases risk of thromboembolism and can be costly and inconvenient.
Nutritional remedies. Of the options that have been tested, which include high protein supplementation, nutrient supplementation such as beef liver extract, and balanced energy/protein supplementation, only the last (with protein content comprising less than 25% of total energy content) has led to a significant reduction in SGA births.31-33
Other approaches. We lack reliable evidence that other interventions such as oxygen administration, abdominal decompression, and pharmacological agents, including calcium channel blockers, beta mimetics, and magnesium, are beneficial or effective in improving or preventing FGR.
A meta-analysis34 of 13 trials involving more than 13,000 women showed that early aspirin treatment reduced the risk of FGR but failed to improve outcome. A more recent meta-analysis35 of 38 trials of aspirin in highrisk pregnancies found no reduction in the incidence of FGR or perinatal death, although a reduction in risk of preterm births was noted.
Etiologic management
Most etiologic conditions are either not amenable to therapy or fetal growth is not improved by treatments that benefit the mother. An example is maternal hypertensive disease, in which the indicated treatment has no beneficial effect on fetal growth.
Treatment of poor lifestyle habits may be helpful. If the mother smokes, vigorous smoking-cessation education and counseling is urged. Also address alcohol consumption and other substance abuse, and offer remedial measures.
The diagnosis of fetal viral and parasitic infections is important for prognostication and neonatal management. Although few perinatal infections are treatable in utero, maternal therapy may prevent certain infections from spreading to the fetus. Examples include toxoplasmosis and malaria.
If lethal malformations or lethal aneuploidy are identified prenatally, avoid fetal surveillance and unnecessary intervention, which may expose the mother to unnecessary and unjustifiable risks.
Fetal surveillance strategy
If fetal biometry indicates fetal weight below the 10th percentile, begin fetal surveillance. The current standard, umbilical arterial Doppler sonography, is the primary test in sonographically documented growth restriction. Also assess amniotic fluid volume—as part of the BPP or independently. AFI is the most commonly used BPP tool.
In the United States, the nonstress test traditionally is the primary monitoring modality, with the BPP as backup, although the BPP—which includes the NST—has been recommended as the primary test.
Normal Doppler findings. When the Doppler index remains within normal limits or does not progressively rise, weekly testing should suffice, with the NST or BPP as backup or in conjunction with Doppler.
If fetal and maternal conditions remain reassuring, allow the pregnancy to continue to maturity and assess the patient for delivery. Postdate pregnancy is not advised in the presence of sonographically confirmed growth compromise.
A high or increasing Doppler index warrants more intensive fetal surveillance consisting of weekly umbilical arterial Doppler and once- or twice-weekly NST and BPP until fetal maturity.
If these tests indicate fetal compromise, or absent end-diastolic velocity develops, the likelihood of poor perinatal outcome is increased and an urgent clinical response is indicated. Hospitalize the patient and individualize management, depending on gestational age and fetal status.
Optimal timing of delivery
The optimal timing of delivery in a preterm pregnancy with FGR is unclear. A recent multicenter randomized controlled trial, the Growth Restriction Intervention Trial (GRIT),36 compared 2 strategies: delivery within 48 hours with steroid administration or delivery delayed as long as fetal status permits. The population consisted of high-risk gravidas between 24 and 36 weeks’ gestation. More than 90% of the women had pregnancies complicated by FGR. No significant differences were noted between the 2 groups in stillbirth rates.
Fortunately, thanks to recent advances in perinatal care, a management strategy can be recommended:
At or near term (34 weeks or beyond), the absence of end-diastolic flow in the umbilical artery should prompt consideration of immediate delivery.
Other ominous findings that prompt such consideration include:
- Cessation of fetal growth on successive ultrasound examinations
- Progression of umbilical arterial absent enddiastolic flow to reversed end-diastolic flow
- Nonreassuring heart rate patterns including nonreactive NST, poor fetal heart rate baseline variability, and persistent variable or late decelerations
- Oligohydramnios
- BPP score of 4 or below
Less than 34 weeks. When absent enddiastolic flow develops in a preterm pregnancy with a significant risk of fetal lung immaturity, seek further assurance of fetal well-being via daily surveillance with umbilical arterial Doppler sonography, nonstress test, and biophysical profile. Administer betamethasone to enhance fetal lung maturity. Delivery is indicated regardless of maturity when a single test or combination indicates imminent fetal danger and the fetal risk from a hostile intrauterine environment is judged to be greater than that from pulmonary immaturity.
Intrapartum management
Labor and delivery of growth-restricted infants often is associated with a higher risk of asphyxia. The frequency of nonreassuring fetal heart rate patterns is increased. In addition, because of the greater prevalence of oligohydramnios, cord compression is frequent and associated with variable decelerations. There also is an increased risk of neonatal morbidities, as discussed above. Because of these risks, the growth-restricted fetus should be delivered in a facility that can offer appropriate inhouse support services such as neonatology and anesthesia.
If the patient is allowed to labor, use continuous electronic fetal heart rate monitoring with vigilance and perform the appropriate secondary tests, such as fetal scalp sampling or scalp stimulation. The ability of a growth-restricted fetus to tolerate labor is likely very limited.
Any confirmed signs of fetal compromise indicate cesarean as the optimal mode of delivery.
The authors report no financial relationships relevant to this article.
1. Barker DJP, ed. Fetal and Infant Origins of Adult Disease. London, UK: BMJ Publishing; 1992.
2. Strauss RS, Dietz WH. Effects of intrauterine growth retardation in premature infants on early childhood growth. J Pediatr. 1997;13:95-102.
3. Victora CG, Barros FC, Horta BL, Martorell R. Short-term benefits of catch-up growth for small-for-gestational-age infants. Int J Epidemiol. 2001;30:1325-1330.
4. Manning FA, Morrison I, Harman CR, Lange IR, Menticoglou S. Fetal assessment based on the fetal biophysical profile scoring: experience in 19,221 referred high-risk pregnancies. II. An analysis of false-negative fetal deaths. Am J Obstet Gynecol. 1987;157:880-884.
5. Low JA, Handley-Derry MH, Burke SO, et al. Association of intrauterine fetal growth retardation and learning deficits at age 9 to 11 years. Am J Obstet Gynecol. 1992;167:1499-1505.
6. Jarvis S, Glinianaia SV, Torrioli MG, et al. Cerebral palsy and intrauterine growth in single births: European collaborative study. Lancet. 2003;362:1106-1111.
7. Lucas A. Programming by early nutrition: an experimental approach. J Nutr. 1998;128(suppl 2):401-406S.
8. Hepburn M, Rosenberg K. An audit of the detection and management of small-for-gestational age babies. Br J Obstet Gynaecol. 1986;93:212-216.
9. Belizan JM, Villar J, Nardin JC, Malamud J, Sainz de Vicuna L. Diagnosis of intrauterine growth retardation by a simple clinical method: measurement of uterine height. Am J Obstet Gynecol. 1978;131:643-646.
10. Lindhard A, Nielsen PV, Mouritsen LA, Zachariassen A, Sorensen HU, Roseno H. The implications of introducing the symphyseal-fundal height measurement. A prospective randomized controlled trial. Br J Obstet Gynaecol. 1990;97:675-680.
11. Neilson JP. Symphysis-fundal height measurement in pregnancy. Cochrane Database Syst Rev. 2000;(2):CD000944.-
12. Hughey MJ. Routine ultrasound for detection and management of the small for-gestational-age fetus. Obstet Gynecol. 1984;64:101-107.
13. Nakling J, Backe B. Adverse obstetric outcome in fetuses that are smaller than expected at second trimester routine ultrasound examination. Acta Obstet Gynecol Scand. 2002;81:846-851.
14. Jahn A, Razum O, Berle P. Routine screening for intrauterine growth retardation in Germany: low sensitivity and questionable benefit for diagnosed cases. Acta Obstet Gynecol Scand. 2002;81:846-851.
15. Ewigman BG, Crane JP, Frigoletto FD, LeFevre ML, Bain RP, McNellis D. Effect of prenatal ultrasound screening on perinatal outcome. RADIUS Study Group. N Engl J Med. 1993; 16;329:821-827.
16. Bricker L, Neilson JP. Routine ultrasound in late pregnancy (after 24 weeks gestation). Cochrane Database Syst Rev. 2000;(2):CD001451.-
17. Goffinet F, Paris Llado J, Nisand I, Breart G. Umbilical artery Doppler velocimetry in unselected and low risk pregnancies: a review of randomized controlled trials. Br J Obstet Gynaecol. 1997;104:425-430.
18. Bricker L, Neilson JP. Routine Doppler ultrasound in pregnancy. Cochrane Database Syst Rev. 2000;(2):CD001450.-
19. Papageorghiou AT, Yu CK, Cicero S, Bower S, Nicolaides KH. Second-trimester uterine artery Doppler screening in unselected populations: a review. J Matern Fetal Neonatal Med. 2002;12:78-88.
20. Chang TC, Robson SC, Boys RJ, Spencer JA. Prediction of the small for gestational age infant: which ultrasonic measurement is best? Obstet Gynecol. 1992;80:1030-1038.
21. Hadlock FP, Harrist RB, Sharman RS, Deter RL, Park SK. Estimation of fetal weight with the use of head, body, and femur measurements—a prospective study. Am J Obstet Gynecol. 1985;151:333-337.
22. Chauhan SP, Sanderson M, Hendrix NW, Magann EF, Devoe LD. Perinatal outcome and amniotic fluid index in the antepartum and intrapartum periods: a meta-analysis. Am J Obstet Gynecol. 1999;18:1473-1478.
23. Banks EH, Miller DA. Perinatal risks associated with borderline amniotic fluid index. Am J Obstet Gynecol. 1999;180:1461-1463.
24. Manning FA, Morrison I, Harman CR, Lange IR, Menticoglou S. Fetal assessment based on fetal biophysical profile scoring: experience in 19,221 referred high-risk pregnancies. II. An analysis of false-negative fetal deaths. Am J Obstet Gynecol. 1987;157:880-884.
25. Alfirevic Z, Neilson JP. Biophysical profile for fetal assessment in high risk pregnancies. Cochrane Database Syst Rev. 2003;(1).:The Cochrane Collaboration.
26. Maulik D, Yarlagadda P, Youngblood JP, et al. The diagnostic efficacy of the UA systolic/diastolic ratio as a screening tool a prospective blinded study. Am J Obstet Gynecol. 1990;162:1518-1523.
27. Nicolaides KH, Bilardo CM, Soothill PW, et al. Absence of end diastolic frequencies in umbilical artery: a sign of fetal hypoxia and acidosis. B Med J. 1988;297:1026-1027.
28. Alfirevic Z, Neilson JP. Doppler ultrasonography in high-risk pregnancies: systematic review with meta-analysis. Am J Obstet Gynecol. 1995;172:1379-1387.
29. Westergaard HB, Langhoff-Roos J, Lingman G, Marsal K, Kreiner S. A critical appraisal of the use of umbilical artery Doppler ultrasound in high-risk pregnancies: use of meta-analyses in evidence-based obstetrics. Ultrasound Obstet Gynecol. 2001;17:464-465.
30. Gulmezoglu AM, Hofmeyr GJ. Bed rest in hospital for suspected impaired fetal growth. Cochrane Database Syst Rev. 2002;(4).-Oxford Update Software.
31. Kramer MS. High protein supplementation in pregnancy. Cochrane Database Syst Rev. 2002;(4).-Oxford Update Software.
32. Kramer MS. Balanced protein/energy supplementation in pregnancy. Cochrane Database Syst Rev. 2002;(4).-Oxford Update Software.
33. Gulmezoglu AM, Hofmeyr GJ. Maternal nutrient supplementation for suspected impaired fetal growth. Cochrane Database Syst Rev. 2002;(4).-Oxford Update Software.
34. Leitich H, Egarter C, Husslein P, Kaider A, Schemper M. A meta-analysis of low dose aspirin for the prevention of intrauterine growth retardation. Br J Obstet Gynaecol. 1997;104:450-459.
35. Kozer E, Costei AM, Boskovic R, Nulman I, Nikfar S, Koren G. Effects of aspirin consumption during pregnancy on pregnancy outcomes: meta-analysis. Birth Defects Res Part B Dev Reprod Toxicol. 2003;68:70-84.
36. The GRIT Study Group. A randomized trial of timed delivery for the compromised preterm fetus: short term outcomes and Bayesian interpretation. BJOG. 2003;110:27-32.
1. Barker DJP, ed. Fetal and Infant Origins of Adult Disease. London, UK: BMJ Publishing; 1992.
2. Strauss RS, Dietz WH. Effects of intrauterine growth retardation in premature infants on early childhood growth. J Pediatr. 1997;13:95-102.
3. Victora CG, Barros FC, Horta BL, Martorell R. Short-term benefits of catch-up growth for small-for-gestational-age infants. Int J Epidemiol. 2001;30:1325-1330.
4. Manning FA, Morrison I, Harman CR, Lange IR, Menticoglou S. Fetal assessment based on the fetal biophysical profile scoring: experience in 19,221 referred high-risk pregnancies. II. An analysis of false-negative fetal deaths. Am J Obstet Gynecol. 1987;157:880-884.
5. Low JA, Handley-Derry MH, Burke SO, et al. Association of intrauterine fetal growth retardation and learning deficits at age 9 to 11 years. Am J Obstet Gynecol. 1992;167:1499-1505.
6. Jarvis S, Glinianaia SV, Torrioli MG, et al. Cerebral palsy and intrauterine growth in single births: European collaborative study. Lancet. 2003;362:1106-1111.
7. Lucas A. Programming by early nutrition: an experimental approach. J Nutr. 1998;128(suppl 2):401-406S.
8. Hepburn M, Rosenberg K. An audit of the detection and management of small-for-gestational age babies. Br J Obstet Gynaecol. 1986;93:212-216.
9. Belizan JM, Villar J, Nardin JC, Malamud J, Sainz de Vicuna L. Diagnosis of intrauterine growth retardation by a simple clinical method: measurement of uterine height. Am J Obstet Gynecol. 1978;131:643-646.
10. Lindhard A, Nielsen PV, Mouritsen LA, Zachariassen A, Sorensen HU, Roseno H. The implications of introducing the symphyseal-fundal height measurement. A prospective randomized controlled trial. Br J Obstet Gynaecol. 1990;97:675-680.
11. Neilson JP. Symphysis-fundal height measurement in pregnancy. Cochrane Database Syst Rev. 2000;(2):CD000944.-
12. Hughey MJ. Routine ultrasound for detection and management of the small for-gestational-age fetus. Obstet Gynecol. 1984;64:101-107.
13. Nakling J, Backe B. Adverse obstetric outcome in fetuses that are smaller than expected at second trimester routine ultrasound examination. Acta Obstet Gynecol Scand. 2002;81:846-851.
14. Jahn A, Razum O, Berle P. Routine screening for intrauterine growth retardation in Germany: low sensitivity and questionable benefit for diagnosed cases. Acta Obstet Gynecol Scand. 2002;81:846-851.
15. Ewigman BG, Crane JP, Frigoletto FD, LeFevre ML, Bain RP, McNellis D. Effect of prenatal ultrasound screening on perinatal outcome. RADIUS Study Group. N Engl J Med. 1993; 16;329:821-827.
16. Bricker L, Neilson JP. Routine ultrasound in late pregnancy (after 24 weeks gestation). Cochrane Database Syst Rev. 2000;(2):CD001451.-
17. Goffinet F, Paris Llado J, Nisand I, Breart G. Umbilical artery Doppler velocimetry in unselected and low risk pregnancies: a review of randomized controlled trials. Br J Obstet Gynaecol. 1997;104:425-430.
18. Bricker L, Neilson JP. Routine Doppler ultrasound in pregnancy. Cochrane Database Syst Rev. 2000;(2):CD001450.-
19. Papageorghiou AT, Yu CK, Cicero S, Bower S, Nicolaides KH. Second-trimester uterine artery Doppler screening in unselected populations: a review. J Matern Fetal Neonatal Med. 2002;12:78-88.
20. Chang TC, Robson SC, Boys RJ, Spencer JA. Prediction of the small for gestational age infant: which ultrasonic measurement is best? Obstet Gynecol. 1992;80:1030-1038.
21. Hadlock FP, Harrist RB, Sharman RS, Deter RL, Park SK. Estimation of fetal weight with the use of head, body, and femur measurements—a prospective study. Am J Obstet Gynecol. 1985;151:333-337.
22. Chauhan SP, Sanderson M, Hendrix NW, Magann EF, Devoe LD. Perinatal outcome and amniotic fluid index in the antepartum and intrapartum periods: a meta-analysis. Am J Obstet Gynecol. 1999;18:1473-1478.
23. Banks EH, Miller DA. Perinatal risks associated with borderline amniotic fluid index. Am J Obstet Gynecol. 1999;180:1461-1463.
24. Manning FA, Morrison I, Harman CR, Lange IR, Menticoglou S. Fetal assessment based on fetal biophysical profile scoring: experience in 19,221 referred high-risk pregnancies. II. An analysis of false-negative fetal deaths. Am J Obstet Gynecol. 1987;157:880-884.
25. Alfirevic Z, Neilson JP. Biophysical profile for fetal assessment in high risk pregnancies. Cochrane Database Syst Rev. 2003;(1).:The Cochrane Collaboration.
26. Maulik D, Yarlagadda P, Youngblood JP, et al. The diagnostic efficacy of the UA systolic/diastolic ratio as a screening tool a prospective blinded study. Am J Obstet Gynecol. 1990;162:1518-1523.
27. Nicolaides KH, Bilardo CM, Soothill PW, et al. Absence of end diastolic frequencies in umbilical artery: a sign of fetal hypoxia and acidosis. B Med J. 1988;297:1026-1027.
28. Alfirevic Z, Neilson JP. Doppler ultrasonography in high-risk pregnancies: systematic review with meta-analysis. Am J Obstet Gynecol. 1995;172:1379-1387.
29. Westergaard HB, Langhoff-Roos J, Lingman G, Marsal K, Kreiner S. A critical appraisal of the use of umbilical artery Doppler ultrasound in high-risk pregnancies: use of meta-analyses in evidence-based obstetrics. Ultrasound Obstet Gynecol. 2001;17:464-465.
30. Gulmezoglu AM, Hofmeyr GJ. Bed rest in hospital for suspected impaired fetal growth. Cochrane Database Syst Rev. 2002;(4).-Oxford Update Software.
31. Kramer MS. High protein supplementation in pregnancy. Cochrane Database Syst Rev. 2002;(4).-Oxford Update Software.
32. Kramer MS. Balanced protein/energy supplementation in pregnancy. Cochrane Database Syst Rev. 2002;(4).-Oxford Update Software.
33. Gulmezoglu AM, Hofmeyr GJ. Maternal nutrient supplementation for suspected impaired fetal growth. Cochrane Database Syst Rev. 2002;(4).-Oxford Update Software.
34. Leitich H, Egarter C, Husslein P, Kaider A, Schemper M. A meta-analysis of low dose aspirin for the prevention of intrauterine growth retardation. Br J Obstet Gynaecol. 1997;104:450-459.
35. Kozer E, Costei AM, Boskovic R, Nulman I, Nikfar S, Koren G. Effects of aspirin consumption during pregnancy on pregnancy outcomes: meta-analysis. Birth Defects Res Part B Dev Reprod Toxicol. 2003;68:70-84.
36. The GRIT Study Group. A randomized trial of timed delivery for the compromised preterm fetus: short term outcomes and Bayesian interpretation. BJOG. 2003;110:27-32.