User login
Upper Extremity Injuries in Soccer
ABSTRACT
Upper limb injuries in soccer represent only a marginal portion of injuries, however this is mainly true for outfield players. Goalkeepers are reported to have up to 5 times more upper extremity injuries, many of them requiring substantial time-loss for treatment and rehabilitation. The most common upper extremity injury locations are the shoulder/clavicle followed by the hand/finger/thumb, elbow, wrist, forearm, and upper arm. The mechanism of injury, presentation, physical examination, and imaging features all play a significant role in reaching the correct diagnosis. Taking to consideration the position the player plays and his demands will also enable tailoring the optimal treatment plan that allows timely and safe return to play. This article discusses common upper extremity injuries observed in soccer players, focusing on proper diagnosis and optimal management.
Continue to: Upper limb injuries in association with soccer...
Upper limb injuries in association with soccer have been reported to represent only 3% of all time-loss injuries in professional soccer players1. However, they are considered an increasing problem in recent years2-4 and have been reported in high proportions in children under the age of 15 years.5 Some of the reasons for the increase in upper extremity injuries may be explained by modern soccer tactics that have been characterized by high speed, pressing, and marking.2 Furthermore, upper extremity injuries may still be underestimated in soccer, mainly because outfield players are sometimes able to train and play even when they suffer from an upper extremity injury.
Unsurprisingly, upper extremity injuries are reported to be up to 5 times more common in goalkeepers than in outfield players,1,2 reaching a high rate of up to 18% of all injuries among professional goalkeepers. The usage of upper extremities to stop the ball and repeated reaching to the ball and landing on the ground with changing upper extremity positions are some of the contributors to the increased upper extremity injury risk in goalkeepers.
Following 57 male professional European soccer teams from 16 countries between the years 2001 and 2011, Ekstrand and colleagues1 showed that 90% of upper extremity injuries are traumatic, and only 10% are related to overuse. They also reported that the most common upper extremity injury location is the shoulder/clavicle (56%), followed by the hand/finger/thumb (24%), elbow (10%), wrist (5%), forearm (4%), and upper arm (1%). Specifically, the 6 most common injuries are acromioclavicular joint (ACJ) sprain (13%), shoulder dislocation (12%), hand metacarpal fracture (8%), shoulder rotator cuff tendinopathy (6%), hand phalanx fracture (6%), and shoulder ACJ dislocation (5%).
This article will discuss common upper extremity injuries observed in soccer players, focusing on proper diagnosis and optimal management.
Continue to: THE SHOULDER...
THE SHOULDER
The majority of upper extremity injuries in professional soccer players are shoulder injuries.1,2,4 Almost a third of these injuries (28%) are considered severe, preventing participation in training and matches for 28 days or more.6Ekstrand and colleagues1 reported that shoulder dislocation represents the most severe upper extremity injury with a mean of 41 days of absence from soccer. When considering the position of the player, they further demonstrated that absence from full training and matches is twice as long for goalkeepers as for outfield players, which reflects the importance of shoulder function for goalkeepers.
In terms of the mechanism of shoulder instability injuries in soccer players, more than half (56%) of these injuries occur with a high-energy mechanism in the recognized position of combined humeral abduction and external rotation against a force of external rotation and horizontal extension.3 However, almost a quarter (24%) occur with a mechanism of varied upper extremity position and low-energy trauma, and 20% of injuries are either a low energy injury with little or no contact or gradual onset. These unique characteristics of shoulder instability injuries in soccer players should be accounted for during training and may imply that current training programs are suboptimal for the prevention of upper extremity injuries and shoulder injuries. Ejnisman and colleagues2 reported on the development of a Fédération Internationale de Football Association (FIFA) 11+ shoulder injury prevention program for soccer goalkeepers as one of the ways to promote training programs that address the risk of shoulder injuries.
Reporting on the management of severe shoulder injuries requiring surgery in 25 professional soccer players in England, between 2007 and 2011, Hart and Funk3 found that the majority of subjects (88%) reported a dislocation as a feature of their presentation. Twenty-one (84%) subjects were diagnosed with labral injuries, of which 7 had an associated Hill-Sachs lesion. Two (8%) subjects were diagnosed with rotator cuff tears requiring repair, and 2 (8%) subjects had a combination of rotator cuff and labral injury repair. All patients underwent arthroscopic repair, except for 5 who had a Latarjet coracoid transfer. Post-surgery, all players were able to return to unrestricted participation in soccer at a mean of 11.4 weeks, with no significant difference between goalkeepers and outfield players and no recurrences at a mean of 91 weeks’ follow-up.
Up to one-third of shoulder instability injuries in soccer players are reported to be recurrences,1,3 which emphasizes the need to carefully assess soccer players before clearing them to return to play. These data raise the controversy over the treatment of first time shoulder dislocators and may support early surgical intervention.7-9 In terms of the preferred surgical intervention in these cases, Balg and Boileau10 suggested a simple scoring system based on factors derived from a preoperative questionnaire, physical examination, and anteroposterior radiographs to help distinguish between patients who will benefit from an arthroscopic anterior stabilization using suture anchors and those who will require a bony procedure (open or arthroscopic). Cerciello and colleagues11 reported excellent results for bony stabilization (modified Latarjet) in a population of 26 soccer players (28 shoulders) affected by chronic anterior instability. Only 1 player did not return to soccer, and 18 players (20 shoulders, 71%) returned to the same level. One re-dislocation was noted in a goalkeeper 74 months after surgery.
An injury to the ACJ has been previously reported to be the most prevalent type of shoulder injury in contact sports.12In soccer, injury to the ACJ is responsible for 18% of upper extremity injuries, and the majority (72%) are sprains.1Interestingly, but unsurprisingly, implications of such an injury differ significantly between goalkeepers and outfield players with up to 3 times longer required absence periods for goalkeepers vs outfield players sustaining the same injury.
ACJ injury is commonly the result of a direct fall on the shoulder with the arm adducted or extended. Six grades of ACJ injuries have been described and distinguished by the injured anatomical structure (acromioclavicular ligaments and coracoclavicular ligaments) and the direction and magnitude of clavicular dislocation.13,14 Presentation will usually include anterior shoulder pain, a noticeable swelling or change in morphology of the lateral end of the clavicle (mainly in dislocation types), and sharp pain provoked by palpation of the ACJ. Radiographic imaging will confirm the diagnosis and help with identifying the specific grade/type of injury.
Decision making and management of acute ACJ injury should be based on the type/grade of injury. Nonoperative treatment is recommended for types I and II, and most athletes have a successful outcome with a full return to play.12Types IV, V, and VI are treated early with operative intervention, mostly due to the morbidity associated with prolonged dislocation of the joint and subsequent soft tissue damage.12 Treatment of type III injury remains controversial. Pereira-Graterol and colleagues15 reported the effectiveness of clavicular hook plate (DePuy Synthes) in the surgical stabilization of grade III ACJ dislocation in 11 professional soccer players. At a mean follow-up of 4 years, they showed excellent functional results with full shoulder range of motion at 5 weeks and latest return to soccer at 6 months. The hook plate was removed after 16 weeks in 10 patients in whom no apparent complication was observed.
Continue to: THE ELBOW...
THE ELBOW
Ekstrand and colleagues1 reported that 10% of all upper extremity injuries in professional soccer players are elbow injuries, of which only 19% are considered severe injuries that require more than 28 days of absence from playing soccer. The most common elbow injuries in their cohort were elbow medial collateral ligament (MCL) sprain and olecranon bursitis.
Elbow MCL is the primary constraint of the elbow joint to valgus stress, and MCL sprain occurs when the elbow is subjected to a valgus, or laterally directed force, which distracts the medial side of the elbow, exceeding the tensile properties of the MCL.16 A thorough physical examination that includes valgus stress tests through the arc of elbow flexion and extension to elicit a possible subjective feeling of apprehension, instability, or localized pain is essential for optimal evaluation and treatment.16,17 Imaging studies (X-ray and stress X-rays, dynamic ultrasound, computed tomography [CT], magnetic resonance imaging [MRI], and MR arthrography) have a role in further establishing the diagnosis and identifying possible additional associated injuries.16 The treatment plan should be specifically tailored to the individual athlete, depending on his demands and the degree of MCL injury. In soccer, which is a non-throwing shoulder sport, nonoperative treatment should be the preferred initial treatment in most cases. Ekstrand and colleagues1 showed that this injury requires a mean of 4 days of absence from soccer for outfield players and a mean of 21 days of absence from soccer for goalkeepers, thereby indicating more severe sprains and cautious return to soccer in goalkeepers. Athletes who fail nonoperative treatment are candidates for MCL reconstruction.16
The olecranon bursa is a synovium-lined sac that facilitates gliding between the olecranon and overlying skin. Olecranon bursitis is characterized by accumulation of fluid in the bursa with or without inflammation. The fluid can be serous, sanguineous, or purulent depending on the etiology.18 In soccer, traumatic etiology is common, but infection secondary to cuts or scratches of the skin around the elbow or previous therapeutic injections around the elbow should always be ruled out. Local pain, swelling, warmth, and redness are usually the presenting symptoms. Aseptic olecranon bursitis may be managed non-surgically with ‘‘benign neglect’’ and avoidance of pressure to the area, non-steroidal anti-inflammatory drugs, needle aspiration, corticosteroid injection, compression dressings, and/or padded splinting; whereas acute septic bursitis requires needle aspiration for diagnosis, appropriate oral or intravenous antibiotics directed toward the offending organism, and, when clinically indicated, surgical evacuation/excision of the bursa.18 When treating this condition with cortisone injection, possible complications, such as skin atrophy, secondary infection, and chronic local pain, have been reported and should be considered.19
Severe elbow injuries in professional athletes in general,20-22 and soccer players specifically, are elbow subluxations/dislocations and elbow fracture. The mechanism of injury is usually contact injury with an opponent player or a fall on the palm with the arm extended. Posterolateral is the most common type of elbow dislocation. Elbow dislocations are further classified into simple (no associated fractures) and complex (associated with fractures) categories.22 Simple dislocations are usually treated with early mobilization after closed reduction; it is associated with a low risk for re-dislocation and with generally good results. The complex type of elbow fracture dislocation is more difficult to treat, has higher complication and re-dislocation rates, and requires operative treatment in most cases compared with simple dislocation.22 The “terrible triad” of the elbow (posterior elbow dislocation, radial head fracture, and coronoid fracture) represents a specific complex elbow dislocation scenario that is difficult to manage because of conflicting aims of ensuring elbow stability while maintaining early range of motion.22
Isolated fracture around the elbow should be treated based on known principles of fracture management: mechanism of injury, fracture patterns, fracture displacement, intra-articular involvement, soft tissue condition, and associated injuries.
Continue to: THE WRIST...
THE WRIST
Ekstrand and colleagues1 reported that 5% of all upper extremity injuries in their cohort of professional soccer players are wrist injuries, of which, only 2% are considered severe injuries that require >28 days of absence from playing soccer. The more common wrist injuries in soccer, which is considered a high-impact sport, are fractures (distal radius, scaphoid, capitate), and less reported injuries are dislocations (lunate, perilunate) and ligamentous injuries or tears (scapholunate ligament).23
Distal radius fractures in high-impact sports, like soccer, usually occur as a result of a fall on an out-stretched hand and will usually be more comminuted, displaced, and intra-articular compared with low-impact sports.23 All these aforementioned characteristics usually indicate surgical management of open reduction and internal fixation, which will allow for rapid start of rehabilitation and return to play.
Scaphoid fracture is the most common carpal bone fracture and presents unique challenges in terms of diagnosis and optimal treatment24 in professional athletes. A typical injury scenario would be a player falling on an outstretched hand and sustaining a scaphoid fracture during a match or training session. The player may acknowledge some wrist pain but will often continue to play with minimal or no limitation. As wrist pain and swelling become more evident after the match/training session, the player will seek medical evaluation.24 A complete wrist and upper extremity examination should be performed in addition to a specific assessment, which includes palpation of the distal scaphoid pole at the distal wrist flexion crease, palpation of the scaphoid waist through the wrist snuff box, and palpation dorsally just distal to the Lister tubercle at the scapholunate joint. Any wrist injury that results in decreased range of motion, snuff box swelling, and scaphoid tenderness should be further evaluated with imaging. Plain radiographs with special scaphoid views are the initial preferred imaging studies, but occult fracture will require an additional study such as a bone scan, CT, or MRI. Several studies have validated the benefit of MRI and the fact that it may outweigh the costs associated with lost productivity from unnecessary cast immobilization, especially in elite athletes.23-25Casting the patient with a nondisplaced scaphoid waist fracture has been the traditional treatment; however, stiffness, weakness, and deconditioning that can occur with long-term casting required for scaphoid fractures are significant impairments for the professional athlete and usually end the player’s season. Surgical treatment, which was traditionally indicated for displaced or proximal pole fractures, is currently also considered for non-displaced scaphoid waist fractures in professional athletes. This treatment allows for a rapid return to the rehabilitation of the extremity and possible early return to professional sport. In view of the known complications (eg, malunion, nonunion, and avascular necrosis), return to soccer can be considered when imaging confirms advanced healing, which some consider as at least 50% of bone fracture bridging on CT scan, no pain, excellent motion, and at least 80% of normal grip strength.24 Outfield players can return to play with a protective cast or brace until full healing is observed on imaging.
Continue to: THE HAND/FINGERS/THUMB...
THE HAND/FINGERS/THUMB
Almost a quarter of upper extremity injuries in professional soccer players were reported to involve the hand, fingers, and thumb. A quarter of them were classified as severe injuries requiring >28 days of absence from playing soccer.1Specifically, hand metacarpal and phalanx fractures are the most common reported injuries in sports in general,26 and in soccer,1 and account for 14% of all upper extremity injuries1 in professional soccer players. Goalkeepers require a functional hand to play, whereas an outfielder can play with protection on the injured area; thus, the period of absence from soccer in these injuries is significantly different between goalkeepers and outfielders with more than 4 times longer absence from soccer for a goalkeeper compared with an outfielder. The fifth ray has been shown to be the most frequently fractured ray in the hand in soccer with 51.7% of all hand fractures reported.26 The common mechanism is a full hit on the hand, and a direct hit from the ball is another possible mechanism in goalkeepers.
In general, the diagnosis of hand injuries requires evaluation of the mechanism of injury and injury symptoms, proper and comprehensive physical examination of the whole extremity, and prompt imaging. In most cases, plain radiographs in several projections will suffice for the diagnosis of obvious fractures, but CT scan is an additional modality that allows for improved appreciation of occult or complex and comminuted fracture patterns. MRI or ultrasound can be used additionally whenever associated soft tissue injury is suspected. Optimal management of the hand is based on the specific characteristics of the fractures, which include location, direction of the fracture line, presence of comminution, displacement, articular involvement, and associated soft tissue injury. Nondisplaced extra-articular fractures often can be treated with buddy taping or splinting, whereas intra-articular fractures often require surgical treatment. Displaced fractures of the hand have a tendency to angulate volarly because of attachments of the interosseous muscles. Marginal fractures or avulsion fractures involving the metacarpals or phalanges can be sentinels of serious associated soft tissue injuries.27
Phalangeal fractures can potentially affect the function of the entire hand; therefore, no malrotation is acceptable for phalangeal fractures because they can lead to overlap and malalignment of the digit. Displaced or malrotated fractures should be reduced either by closed or open techniques. Acceptable reduction is <6 mm of shortening, <15° of angulation, and no rotational deformity.27,28 Nondisplaced phalangeal fractures can be treated nonoperatively with buddy taping and splinting with good results.27 Interphalangeal (IP) dislocations can be reduced on the sidelines and then taped or splinted. Any injury with a force significant enough to cause joint dislocation indicates further evaluation for associated fractures and ligamentous injury or tear. The proximal interphalangeal (PIP) joint is the most common IP joint dislocation and is usually a dorsal dislocation. Reduction is often achieved by traction and flexion of the middle phalanx,27 followed by splinting of the finger with the PIP in 30° of flexion or an extension block splint.29 Successful reduction with no associated intra-articular fractures involving more than a third of the joint can be further managed nonoperatively with the splint, allowing 2 to 4 weeks for the volar plate, joint capsule, and collateral ligaments to heal. Additional 2 to 4 weeks of splinting with buddy taping to the adjacent finger is usually recommended.29
The “Mallet finger” injury can be observed in goalkeepers and is caused by a flexion force on the tip of the finger while the distal interphalangeal (DIP) joint is extended. This force results in tearing of the extensor tendon or an avulsion fracture at the tendinous attachment on the dorsal lip of the distal phalangeal base. The classic mechanism of injury is an extended finger struck on the tip by a ball. Physical examination will indicate loss of DIP joint active extension, and the joint rests in an abnormally flexed position. Treatment typically consists of splinting the DIP joint in extension for 6 to 8 weeks. Operative treatment is reserved for severe injuries or fractures involving greater than one-third of the articular surface of the DIP joint or with failed nonoperative treatment.27
Metacarpal fractures can be subdivided into distal, metacarpal neck, metacarpal shaft, and metacarpal base fractures. Metacarpal shaft fractures raise a specific concern regarding rotation, because even a small degree of rotation can create a substantial degree of deformity at the fingertip. This concern must be addressed during evaluation of the player. Fractures of the metacarpal base most commonly involve the fourth and fifth metacarpals and are often reduced easily but have a tendency to re-subluxate, which may indicate operative treatment. Most fractures of the metacarpals are low energy and result in simple fracture patterns that can be treated nonoperatively. Open reduction is reserved for high-energy trauma, fractures with excessive angulation, or multiple fractures.27
Continue to: An important subgroup of metacarpal injuries...
An important subgroup of metacarpal injuries involves the base of the thumb. These injuries result from an axial load applied to the thumb. The most common injury is the “Bennett fracture,” which is an intra-articular fracture or dislocation involving the base of the first metacarpal. Bennett fractures are unstable fractures; unless properly recognized and treated, this intra-articular fracture subluxation may result in an unstable arthritic first carpometacarpal joint. These fractures are most commonly treated with closed or open reduction combined with internal fixation.27 “Rolando fractures” are similar in location and etiology but are comminuted and usually require operative treatment.27, 29
Another common hand injury found in soccer goalkeepers and involving the base of the thumb is disruption of the ulnar collateral ligament (UCL) of the first metacarpophalangeal (MCP) joint as a result of an acute radial or valgus stress on the thumb. Known as “gamekeeper’s thumb” or “skier’s thumb,” this injury can occur in the form of an avulsion fracture, an isolated ligament tear, or combined fracture and ligament rupture. On examination, swelling and tenderness over the thumb UCL are observed. A MCP joint stress test should be performed by gently applying a radially directed force to the thumb while stabilizing the metacarpal bone at both 0° and 30° at the MCP joint. Increased laxity, a soft or nonexistent end point, and gaping of the joint, as compared with the contralateral side, will indicate this injury.29 Radiographs may show a small avulsion fracture fragment at the ulnar aspect of the base of the first metacarpal and at the attachment of the UCL. A Stener lesion is an abnormality that occurs when the thumb adductor muscle aponeurosis interposes between the 2 ends of the ruptured UCL, preventing UCL healing by immobilization alone. Ultrasound and MRI are additional imaging modalities that can assist with the diagnosis of a Stener lesion. The presence of a Stener lesion is a prime indication for surgical intervention. A nondisplaced fracture or isolated ligament injury with no evidence of a Stener lesion can be treated nonoperatively with splinting of the thumb and may lead to healing and restoration of stability. However, in professional players, surgical repair is often times preferred.27
CONCLUSION
Upper extremity injuries are less common injuries among soccer players, but their prevalence is on the rise in recent years. Modern playing tactics and the increase in participation in soccer in younger age groups may be 2 contributing factors to this rise. Given the characteristics of their unique playing role and specific demands, the risk for upper extremity injuries among goalkeepers is significantly higher than that in outfielders and will usually result in a long absence period from soccer before they return to play. A thorough understanding of the mechanism of injury, players’ complaints and presentation, osseous and soft tissue involvement based on a systematic physical examination, imaging features, and treatment options is important for the optimal care of the players. Prompt and accurate diagnosis and appropriate management are essential for improved outcomes and timely return to play.
1. Ekstrand J, Hagglund M, Tornqvist H, et al. Upper extremity injuries in male elite football players. Knee Surg Sports Traumatol Arthrosc. 2013;21(7):1626-1632. doi:10.1007/s00167-012-2164-6.
2. Ejnisman B, Barbosa G, Andreoli CV, et al. Shoulder injuries in soccer goalkeepers: Review and development of a FIFA 11+ shoulder injury prevention program. Open Access J Sports Med. 2016;7:75-80. doi:10.2147/OAJSM.S97917.
3. Hart D, Funk L. Serious shoulder injuries in professional soccer: Return to participation after surgery. Knee Surg Sports Traumatol Arthrosc. 2015;23(7):2123-2129. doi:10.1007/s00167-013-2796-1.
4. Longo UG, Loppini M, Berton A, Martinelli N, Maffulli N, Denaro V. Shoulder injuries in soccer players. Clin Cases Miner Bone Metab. 2012;9(3):138-141.
5. Faude O, Rossler R, Junge A. Football injuries in children and adolescent players: Are there clues for prevention? Sports Med. 2013;43(9):819-837. doi:10.1007/s40279-013-0061-x.
6. Ekstrand J, Hagglund M, Walden M. Injury incidence and injury patterns in professional football: The UEFA injury study. Br J Sports Med. 2011;45(7):553-558. doi:10.1136/bjsm.2009.060582.
7. Boone JL, Arciero RA. First-time anterior shoulder dislocations: Has the standard changed? Br J Sports Med. 2010;44(5):355-360. doi:10.1136/bjsm.2009.062596.
8. Handoll HH, Almaiyah MA, Rangan A. Surgical versus non-surgical treatment for acute anterior shoulder dislocation. Cochrane Database Syst Rev. 2004;(1):CD004325.
9. Kirkley A, Werstine R, Ratjek A, Griffin S. Prospective randomized clinical trial comparing the effectiveness of immediate arthroscopic stabilization versus immobilization and rehabilitation in first traumatic anterior dislocations of the shoulder: Long-term evaluation. Arthroscopy. 2005;21(1):55-63.
10. Balg F, Boileau P. The instability severity index score. A simple pre-operative score to select patients for arthroscopic or open shoulder stabilisation. J Bone Joint Surg Br. 2007;89(11):1470-1477.
11. Cerciello S, Edwards TB, Walch G. Chronic anterior glenohumeral instability in soccer players: Results for a series of 28 shoulders treated with the latarjet procedure. J Orthop Traumatol. 2012;13(4):197-202. doi:10.1007/s10195-012-0201-3.
12. Bishop JY, Kaeding C. Treatment of the acute traumatic acromioclavicular separation. Sports Med Arthrosc Rev. 2006;14(4):237-245. doi:10.1097/01.jsa.0000212330.32969.6e.
13. de Putter CE, van Beeck EF, Burdorf A, et al. Increase in upper extremity fractures in young male soccer players in the netherlands, 1998-2009. Scand J Med Sci Sports. 2015;25(4):462-466. doi:10.1111/sms.12287.
14. Rockwood CJ, Williams G, Young D. Disorders of the acromioclavicular joint. In: Rockwood CJ, Matsen FA III, eds. The Shoulder. 2nd ed. Philadelphia: WB Saunders; 1998:483-553.
15. Pereira-Graterol E, Alvarez-Diaz P, Seijas R, Ares O, Cusco X, Cugat R. Treatment and evolution of grade III acromioclavicular dislocations in soccer players. Knee Surg Sports Traumatol Arthrosc. 2013;21(7):1633-1635. doi:10.1007/s00167-012-2186-0.
16. Rahman RK, Levine WN, Ahmad CS. Elbow medial collateral ligament injuries. Curr Rev Musculoskelet Med. 2008;1(3-4):197-204. doi:10.1007/s12178-008-9026-3.
17. Redler LH, Watling JP, Ahmad CS. Physical examination of the throwing athlete's elbow. Am J Orthop. 2015;44(1):13-18.
18. Sayegh ET, Strauch RJ. Treatment of olecranon bursitis: A systematic review. Arch Orthop Trauma Surg. 2014;134(11):1517-1536. doi:10.1007/s00402-014-2088-3.
19. Weinstein PS, Canoso JJ, Wohlgethan JR. Long-term follow-up of corticosteroid injection for traumatic olecranon bursitis. Ann Rheum Dis. 1984;43(1):44-46.
20. Carlisle JC, Goldfarb CA, Mall N, Powell JW, Matava MJ. Upper extremity injuries in the national football league: Part II: Elbow, forearm, and wrist injuries. Am J Sports Med. 2008;36(10):1945-1952. doi:10.1177/0363546508318198.
21. Dizdarevic I, Low S, Currie DW, Comstock RD, Hammoud S, Atanda A Jr. Epidemiology of elbow dislocations in high school athletes. Am J Sports Med. 2016;44(1):202-208. doi:10.1177/0363546515610527.
22. Saati AZ, McKee MD. Fracture-dislocation of the elbow: Diagnosis, treatment, and prognosis. Hand Clin. 2004;20(4):405-414.
23. Bancroft LW. Wrist injuries: A comparison between high- and low-impact sports. Radiol Clin North Am. 2013;51(2):299-311. doi:10.1016/j.rcl.2012.09.017.
24. Belsky MR, Leibman MI, Ruchelsman DE. Scaphoid fracture in the elite athlete. Hand Clin. 2012;28(3):78, vii. doi:10.1016/j.hcl.2012.05.005.
25. Mallee W, Doornberg JN, Ring D, van Dijk CN, Maas M, Goslings JC. Comparison of CT and MRI for diagnosis of suspected scaphoid fractures. J Bone Joint Surg Am. 2011;93(1):20-28. doi:10.2106/JBJS.I.01523.
26. Aitken S, Court-Brown CM. The epidemiology of sports-related fractures of the hand. Injury. 2008;39(12):1377-1383. doi:10.1016/j.injury.2008.04.012.
27. Peterson JJ, Bancroft LW. Injuries of the fingers and thumb in the athlete. Clin Sports Med. 2006;25(3):viii.
28. Walsh JJ 4th. Fractures of the hand and carpal navicular bone in athletes. South Med J. 2004;97(8):762-765.
29. Hong E. Hand injuries in sports medicine. Prim Care. 2005;32(1):91-103.
ABSTRACT
Upper limb injuries in soccer represent only a marginal portion of injuries, however this is mainly true for outfield players. Goalkeepers are reported to have up to 5 times more upper extremity injuries, many of them requiring substantial time-loss for treatment and rehabilitation. The most common upper extremity injury locations are the shoulder/clavicle followed by the hand/finger/thumb, elbow, wrist, forearm, and upper arm. The mechanism of injury, presentation, physical examination, and imaging features all play a significant role in reaching the correct diagnosis. Taking to consideration the position the player plays and his demands will also enable tailoring the optimal treatment plan that allows timely and safe return to play. This article discusses common upper extremity injuries observed in soccer players, focusing on proper diagnosis and optimal management.
Continue to: Upper limb injuries in association with soccer...
Upper limb injuries in association with soccer have been reported to represent only 3% of all time-loss injuries in professional soccer players1. However, they are considered an increasing problem in recent years2-4 and have been reported in high proportions in children under the age of 15 years.5 Some of the reasons for the increase in upper extremity injuries may be explained by modern soccer tactics that have been characterized by high speed, pressing, and marking.2 Furthermore, upper extremity injuries may still be underestimated in soccer, mainly because outfield players are sometimes able to train and play even when they suffer from an upper extremity injury.
Unsurprisingly, upper extremity injuries are reported to be up to 5 times more common in goalkeepers than in outfield players,1,2 reaching a high rate of up to 18% of all injuries among professional goalkeepers. The usage of upper extremities to stop the ball and repeated reaching to the ball and landing on the ground with changing upper extremity positions are some of the contributors to the increased upper extremity injury risk in goalkeepers.
Following 57 male professional European soccer teams from 16 countries between the years 2001 and 2011, Ekstrand and colleagues1 showed that 90% of upper extremity injuries are traumatic, and only 10% are related to overuse. They also reported that the most common upper extremity injury location is the shoulder/clavicle (56%), followed by the hand/finger/thumb (24%), elbow (10%), wrist (5%), forearm (4%), and upper arm (1%). Specifically, the 6 most common injuries are acromioclavicular joint (ACJ) sprain (13%), shoulder dislocation (12%), hand metacarpal fracture (8%), shoulder rotator cuff tendinopathy (6%), hand phalanx fracture (6%), and shoulder ACJ dislocation (5%).
This article will discuss common upper extremity injuries observed in soccer players, focusing on proper diagnosis and optimal management.
Continue to: THE SHOULDER...
THE SHOULDER
The majority of upper extremity injuries in professional soccer players are shoulder injuries.1,2,4 Almost a third of these injuries (28%) are considered severe, preventing participation in training and matches for 28 days or more.6Ekstrand and colleagues1 reported that shoulder dislocation represents the most severe upper extremity injury with a mean of 41 days of absence from soccer. When considering the position of the player, they further demonstrated that absence from full training and matches is twice as long for goalkeepers as for outfield players, which reflects the importance of shoulder function for goalkeepers.
In terms of the mechanism of shoulder instability injuries in soccer players, more than half (56%) of these injuries occur with a high-energy mechanism in the recognized position of combined humeral abduction and external rotation against a force of external rotation and horizontal extension.3 However, almost a quarter (24%) occur with a mechanism of varied upper extremity position and low-energy trauma, and 20% of injuries are either a low energy injury with little or no contact or gradual onset. These unique characteristics of shoulder instability injuries in soccer players should be accounted for during training and may imply that current training programs are suboptimal for the prevention of upper extremity injuries and shoulder injuries. Ejnisman and colleagues2 reported on the development of a Fédération Internationale de Football Association (FIFA) 11+ shoulder injury prevention program for soccer goalkeepers as one of the ways to promote training programs that address the risk of shoulder injuries.
Reporting on the management of severe shoulder injuries requiring surgery in 25 professional soccer players in England, between 2007 and 2011, Hart and Funk3 found that the majority of subjects (88%) reported a dislocation as a feature of their presentation. Twenty-one (84%) subjects were diagnosed with labral injuries, of which 7 had an associated Hill-Sachs lesion. Two (8%) subjects were diagnosed with rotator cuff tears requiring repair, and 2 (8%) subjects had a combination of rotator cuff and labral injury repair. All patients underwent arthroscopic repair, except for 5 who had a Latarjet coracoid transfer. Post-surgery, all players were able to return to unrestricted participation in soccer at a mean of 11.4 weeks, with no significant difference between goalkeepers and outfield players and no recurrences at a mean of 91 weeks’ follow-up.
Up to one-third of shoulder instability injuries in soccer players are reported to be recurrences,1,3 which emphasizes the need to carefully assess soccer players before clearing them to return to play. These data raise the controversy over the treatment of first time shoulder dislocators and may support early surgical intervention.7-9 In terms of the preferred surgical intervention in these cases, Balg and Boileau10 suggested a simple scoring system based on factors derived from a preoperative questionnaire, physical examination, and anteroposterior radiographs to help distinguish between patients who will benefit from an arthroscopic anterior stabilization using suture anchors and those who will require a bony procedure (open or arthroscopic). Cerciello and colleagues11 reported excellent results for bony stabilization (modified Latarjet) in a population of 26 soccer players (28 shoulders) affected by chronic anterior instability. Only 1 player did not return to soccer, and 18 players (20 shoulders, 71%) returned to the same level. One re-dislocation was noted in a goalkeeper 74 months after surgery.
An injury to the ACJ has been previously reported to be the most prevalent type of shoulder injury in contact sports.12In soccer, injury to the ACJ is responsible for 18% of upper extremity injuries, and the majority (72%) are sprains.1Interestingly, but unsurprisingly, implications of such an injury differ significantly between goalkeepers and outfield players with up to 3 times longer required absence periods for goalkeepers vs outfield players sustaining the same injury.
ACJ injury is commonly the result of a direct fall on the shoulder with the arm adducted or extended. Six grades of ACJ injuries have been described and distinguished by the injured anatomical structure (acromioclavicular ligaments and coracoclavicular ligaments) and the direction and magnitude of clavicular dislocation.13,14 Presentation will usually include anterior shoulder pain, a noticeable swelling or change in morphology of the lateral end of the clavicle (mainly in dislocation types), and sharp pain provoked by palpation of the ACJ. Radiographic imaging will confirm the diagnosis and help with identifying the specific grade/type of injury.
Decision making and management of acute ACJ injury should be based on the type/grade of injury. Nonoperative treatment is recommended for types I and II, and most athletes have a successful outcome with a full return to play.12Types IV, V, and VI are treated early with operative intervention, mostly due to the morbidity associated with prolonged dislocation of the joint and subsequent soft tissue damage.12 Treatment of type III injury remains controversial. Pereira-Graterol and colleagues15 reported the effectiveness of clavicular hook plate (DePuy Synthes) in the surgical stabilization of grade III ACJ dislocation in 11 professional soccer players. At a mean follow-up of 4 years, they showed excellent functional results with full shoulder range of motion at 5 weeks and latest return to soccer at 6 months. The hook plate was removed after 16 weeks in 10 patients in whom no apparent complication was observed.
Continue to: THE ELBOW...
THE ELBOW
Ekstrand and colleagues1 reported that 10% of all upper extremity injuries in professional soccer players are elbow injuries, of which only 19% are considered severe injuries that require more than 28 days of absence from playing soccer. The most common elbow injuries in their cohort were elbow medial collateral ligament (MCL) sprain and olecranon bursitis.
Elbow MCL is the primary constraint of the elbow joint to valgus stress, and MCL sprain occurs when the elbow is subjected to a valgus, or laterally directed force, which distracts the medial side of the elbow, exceeding the tensile properties of the MCL.16 A thorough physical examination that includes valgus stress tests through the arc of elbow flexion and extension to elicit a possible subjective feeling of apprehension, instability, or localized pain is essential for optimal evaluation and treatment.16,17 Imaging studies (X-ray and stress X-rays, dynamic ultrasound, computed tomography [CT], magnetic resonance imaging [MRI], and MR arthrography) have a role in further establishing the diagnosis and identifying possible additional associated injuries.16 The treatment plan should be specifically tailored to the individual athlete, depending on his demands and the degree of MCL injury. In soccer, which is a non-throwing shoulder sport, nonoperative treatment should be the preferred initial treatment in most cases. Ekstrand and colleagues1 showed that this injury requires a mean of 4 days of absence from soccer for outfield players and a mean of 21 days of absence from soccer for goalkeepers, thereby indicating more severe sprains and cautious return to soccer in goalkeepers. Athletes who fail nonoperative treatment are candidates for MCL reconstruction.16
The olecranon bursa is a synovium-lined sac that facilitates gliding between the olecranon and overlying skin. Olecranon bursitis is characterized by accumulation of fluid in the bursa with or without inflammation. The fluid can be serous, sanguineous, or purulent depending on the etiology.18 In soccer, traumatic etiology is common, but infection secondary to cuts or scratches of the skin around the elbow or previous therapeutic injections around the elbow should always be ruled out. Local pain, swelling, warmth, and redness are usually the presenting symptoms. Aseptic olecranon bursitis may be managed non-surgically with ‘‘benign neglect’’ and avoidance of pressure to the area, non-steroidal anti-inflammatory drugs, needle aspiration, corticosteroid injection, compression dressings, and/or padded splinting; whereas acute septic bursitis requires needle aspiration for diagnosis, appropriate oral or intravenous antibiotics directed toward the offending organism, and, when clinically indicated, surgical evacuation/excision of the bursa.18 When treating this condition with cortisone injection, possible complications, such as skin atrophy, secondary infection, and chronic local pain, have been reported and should be considered.19
Severe elbow injuries in professional athletes in general,20-22 and soccer players specifically, are elbow subluxations/dislocations and elbow fracture. The mechanism of injury is usually contact injury with an opponent player or a fall on the palm with the arm extended. Posterolateral is the most common type of elbow dislocation. Elbow dislocations are further classified into simple (no associated fractures) and complex (associated with fractures) categories.22 Simple dislocations are usually treated with early mobilization after closed reduction; it is associated with a low risk for re-dislocation and with generally good results. The complex type of elbow fracture dislocation is more difficult to treat, has higher complication and re-dislocation rates, and requires operative treatment in most cases compared with simple dislocation.22 The “terrible triad” of the elbow (posterior elbow dislocation, radial head fracture, and coronoid fracture) represents a specific complex elbow dislocation scenario that is difficult to manage because of conflicting aims of ensuring elbow stability while maintaining early range of motion.22
Isolated fracture around the elbow should be treated based on known principles of fracture management: mechanism of injury, fracture patterns, fracture displacement, intra-articular involvement, soft tissue condition, and associated injuries.
Continue to: THE WRIST...
THE WRIST
Ekstrand and colleagues1 reported that 5% of all upper extremity injuries in their cohort of professional soccer players are wrist injuries, of which, only 2% are considered severe injuries that require >28 days of absence from playing soccer. The more common wrist injuries in soccer, which is considered a high-impact sport, are fractures (distal radius, scaphoid, capitate), and less reported injuries are dislocations (lunate, perilunate) and ligamentous injuries or tears (scapholunate ligament).23
Distal radius fractures in high-impact sports, like soccer, usually occur as a result of a fall on an out-stretched hand and will usually be more comminuted, displaced, and intra-articular compared with low-impact sports.23 All these aforementioned characteristics usually indicate surgical management of open reduction and internal fixation, which will allow for rapid start of rehabilitation and return to play.
Scaphoid fracture is the most common carpal bone fracture and presents unique challenges in terms of diagnosis and optimal treatment24 in professional athletes. A typical injury scenario would be a player falling on an outstretched hand and sustaining a scaphoid fracture during a match or training session. The player may acknowledge some wrist pain but will often continue to play with minimal or no limitation. As wrist pain and swelling become more evident after the match/training session, the player will seek medical evaluation.24 A complete wrist and upper extremity examination should be performed in addition to a specific assessment, which includes palpation of the distal scaphoid pole at the distal wrist flexion crease, palpation of the scaphoid waist through the wrist snuff box, and palpation dorsally just distal to the Lister tubercle at the scapholunate joint. Any wrist injury that results in decreased range of motion, snuff box swelling, and scaphoid tenderness should be further evaluated with imaging. Plain radiographs with special scaphoid views are the initial preferred imaging studies, but occult fracture will require an additional study such as a bone scan, CT, or MRI. Several studies have validated the benefit of MRI and the fact that it may outweigh the costs associated with lost productivity from unnecessary cast immobilization, especially in elite athletes.23-25Casting the patient with a nondisplaced scaphoid waist fracture has been the traditional treatment; however, stiffness, weakness, and deconditioning that can occur with long-term casting required for scaphoid fractures are significant impairments for the professional athlete and usually end the player’s season. Surgical treatment, which was traditionally indicated for displaced or proximal pole fractures, is currently also considered for non-displaced scaphoid waist fractures in professional athletes. This treatment allows for a rapid return to the rehabilitation of the extremity and possible early return to professional sport. In view of the known complications (eg, malunion, nonunion, and avascular necrosis), return to soccer can be considered when imaging confirms advanced healing, which some consider as at least 50% of bone fracture bridging on CT scan, no pain, excellent motion, and at least 80% of normal grip strength.24 Outfield players can return to play with a protective cast or brace until full healing is observed on imaging.
Continue to: THE HAND/FINGERS/THUMB...
THE HAND/FINGERS/THUMB
Almost a quarter of upper extremity injuries in professional soccer players were reported to involve the hand, fingers, and thumb. A quarter of them were classified as severe injuries requiring >28 days of absence from playing soccer.1Specifically, hand metacarpal and phalanx fractures are the most common reported injuries in sports in general,26 and in soccer,1 and account for 14% of all upper extremity injuries1 in professional soccer players. Goalkeepers require a functional hand to play, whereas an outfielder can play with protection on the injured area; thus, the period of absence from soccer in these injuries is significantly different between goalkeepers and outfielders with more than 4 times longer absence from soccer for a goalkeeper compared with an outfielder. The fifth ray has been shown to be the most frequently fractured ray in the hand in soccer with 51.7% of all hand fractures reported.26 The common mechanism is a full hit on the hand, and a direct hit from the ball is another possible mechanism in goalkeepers.
In general, the diagnosis of hand injuries requires evaluation of the mechanism of injury and injury symptoms, proper and comprehensive physical examination of the whole extremity, and prompt imaging. In most cases, plain radiographs in several projections will suffice for the diagnosis of obvious fractures, but CT scan is an additional modality that allows for improved appreciation of occult or complex and comminuted fracture patterns. MRI or ultrasound can be used additionally whenever associated soft tissue injury is suspected. Optimal management of the hand is based on the specific characteristics of the fractures, which include location, direction of the fracture line, presence of comminution, displacement, articular involvement, and associated soft tissue injury. Nondisplaced extra-articular fractures often can be treated with buddy taping or splinting, whereas intra-articular fractures often require surgical treatment. Displaced fractures of the hand have a tendency to angulate volarly because of attachments of the interosseous muscles. Marginal fractures or avulsion fractures involving the metacarpals or phalanges can be sentinels of serious associated soft tissue injuries.27
Phalangeal fractures can potentially affect the function of the entire hand; therefore, no malrotation is acceptable for phalangeal fractures because they can lead to overlap and malalignment of the digit. Displaced or malrotated fractures should be reduced either by closed or open techniques. Acceptable reduction is <6 mm of shortening, <15° of angulation, and no rotational deformity.27,28 Nondisplaced phalangeal fractures can be treated nonoperatively with buddy taping and splinting with good results.27 Interphalangeal (IP) dislocations can be reduced on the sidelines and then taped or splinted. Any injury with a force significant enough to cause joint dislocation indicates further evaluation for associated fractures and ligamentous injury or tear. The proximal interphalangeal (PIP) joint is the most common IP joint dislocation and is usually a dorsal dislocation. Reduction is often achieved by traction and flexion of the middle phalanx,27 followed by splinting of the finger with the PIP in 30° of flexion or an extension block splint.29 Successful reduction with no associated intra-articular fractures involving more than a third of the joint can be further managed nonoperatively with the splint, allowing 2 to 4 weeks for the volar plate, joint capsule, and collateral ligaments to heal. Additional 2 to 4 weeks of splinting with buddy taping to the adjacent finger is usually recommended.29
The “Mallet finger” injury can be observed in goalkeepers and is caused by a flexion force on the tip of the finger while the distal interphalangeal (DIP) joint is extended. This force results in tearing of the extensor tendon or an avulsion fracture at the tendinous attachment on the dorsal lip of the distal phalangeal base. The classic mechanism of injury is an extended finger struck on the tip by a ball. Physical examination will indicate loss of DIP joint active extension, and the joint rests in an abnormally flexed position. Treatment typically consists of splinting the DIP joint in extension for 6 to 8 weeks. Operative treatment is reserved for severe injuries or fractures involving greater than one-third of the articular surface of the DIP joint or with failed nonoperative treatment.27
Metacarpal fractures can be subdivided into distal, metacarpal neck, metacarpal shaft, and metacarpal base fractures. Metacarpal shaft fractures raise a specific concern regarding rotation, because even a small degree of rotation can create a substantial degree of deformity at the fingertip. This concern must be addressed during evaluation of the player. Fractures of the metacarpal base most commonly involve the fourth and fifth metacarpals and are often reduced easily but have a tendency to re-subluxate, which may indicate operative treatment. Most fractures of the metacarpals are low energy and result in simple fracture patterns that can be treated nonoperatively. Open reduction is reserved for high-energy trauma, fractures with excessive angulation, or multiple fractures.27
Continue to: An important subgroup of metacarpal injuries...
An important subgroup of metacarpal injuries involves the base of the thumb. These injuries result from an axial load applied to the thumb. The most common injury is the “Bennett fracture,” which is an intra-articular fracture or dislocation involving the base of the first metacarpal. Bennett fractures are unstable fractures; unless properly recognized and treated, this intra-articular fracture subluxation may result in an unstable arthritic first carpometacarpal joint. These fractures are most commonly treated with closed or open reduction combined with internal fixation.27 “Rolando fractures” are similar in location and etiology but are comminuted and usually require operative treatment.27, 29
Another common hand injury found in soccer goalkeepers and involving the base of the thumb is disruption of the ulnar collateral ligament (UCL) of the first metacarpophalangeal (MCP) joint as a result of an acute radial or valgus stress on the thumb. Known as “gamekeeper’s thumb” or “skier’s thumb,” this injury can occur in the form of an avulsion fracture, an isolated ligament tear, or combined fracture and ligament rupture. On examination, swelling and tenderness over the thumb UCL are observed. A MCP joint stress test should be performed by gently applying a radially directed force to the thumb while stabilizing the metacarpal bone at both 0° and 30° at the MCP joint. Increased laxity, a soft or nonexistent end point, and gaping of the joint, as compared with the contralateral side, will indicate this injury.29 Radiographs may show a small avulsion fracture fragment at the ulnar aspect of the base of the first metacarpal and at the attachment of the UCL. A Stener lesion is an abnormality that occurs when the thumb adductor muscle aponeurosis interposes between the 2 ends of the ruptured UCL, preventing UCL healing by immobilization alone. Ultrasound and MRI are additional imaging modalities that can assist with the diagnosis of a Stener lesion. The presence of a Stener lesion is a prime indication for surgical intervention. A nondisplaced fracture or isolated ligament injury with no evidence of a Stener lesion can be treated nonoperatively with splinting of the thumb and may lead to healing and restoration of stability. However, in professional players, surgical repair is often times preferred.27
CONCLUSION
Upper extremity injuries are less common injuries among soccer players, but their prevalence is on the rise in recent years. Modern playing tactics and the increase in participation in soccer in younger age groups may be 2 contributing factors to this rise. Given the characteristics of their unique playing role and specific demands, the risk for upper extremity injuries among goalkeepers is significantly higher than that in outfielders and will usually result in a long absence period from soccer before they return to play. A thorough understanding of the mechanism of injury, players’ complaints and presentation, osseous and soft tissue involvement based on a systematic physical examination, imaging features, and treatment options is important for the optimal care of the players. Prompt and accurate diagnosis and appropriate management are essential for improved outcomes and timely return to play.
ABSTRACT
Upper limb injuries in soccer represent only a marginal portion of injuries, however this is mainly true for outfield players. Goalkeepers are reported to have up to 5 times more upper extremity injuries, many of them requiring substantial time-loss for treatment and rehabilitation. The most common upper extremity injury locations are the shoulder/clavicle followed by the hand/finger/thumb, elbow, wrist, forearm, and upper arm. The mechanism of injury, presentation, physical examination, and imaging features all play a significant role in reaching the correct diagnosis. Taking to consideration the position the player plays and his demands will also enable tailoring the optimal treatment plan that allows timely and safe return to play. This article discusses common upper extremity injuries observed in soccer players, focusing on proper diagnosis and optimal management.
Continue to: Upper limb injuries in association with soccer...
Upper limb injuries in association with soccer have been reported to represent only 3% of all time-loss injuries in professional soccer players1. However, they are considered an increasing problem in recent years2-4 and have been reported in high proportions in children under the age of 15 years.5 Some of the reasons for the increase in upper extremity injuries may be explained by modern soccer tactics that have been characterized by high speed, pressing, and marking.2 Furthermore, upper extremity injuries may still be underestimated in soccer, mainly because outfield players are sometimes able to train and play even when they suffer from an upper extremity injury.
Unsurprisingly, upper extremity injuries are reported to be up to 5 times more common in goalkeepers than in outfield players,1,2 reaching a high rate of up to 18% of all injuries among professional goalkeepers. The usage of upper extremities to stop the ball and repeated reaching to the ball and landing on the ground with changing upper extremity positions are some of the contributors to the increased upper extremity injury risk in goalkeepers.
Following 57 male professional European soccer teams from 16 countries between the years 2001 and 2011, Ekstrand and colleagues1 showed that 90% of upper extremity injuries are traumatic, and only 10% are related to overuse. They also reported that the most common upper extremity injury location is the shoulder/clavicle (56%), followed by the hand/finger/thumb (24%), elbow (10%), wrist (5%), forearm (4%), and upper arm (1%). Specifically, the 6 most common injuries are acromioclavicular joint (ACJ) sprain (13%), shoulder dislocation (12%), hand metacarpal fracture (8%), shoulder rotator cuff tendinopathy (6%), hand phalanx fracture (6%), and shoulder ACJ dislocation (5%).
This article will discuss common upper extremity injuries observed in soccer players, focusing on proper diagnosis and optimal management.
Continue to: THE SHOULDER...
THE SHOULDER
The majority of upper extremity injuries in professional soccer players are shoulder injuries.1,2,4 Almost a third of these injuries (28%) are considered severe, preventing participation in training and matches for 28 days or more.6Ekstrand and colleagues1 reported that shoulder dislocation represents the most severe upper extremity injury with a mean of 41 days of absence from soccer. When considering the position of the player, they further demonstrated that absence from full training and matches is twice as long for goalkeepers as for outfield players, which reflects the importance of shoulder function for goalkeepers.
In terms of the mechanism of shoulder instability injuries in soccer players, more than half (56%) of these injuries occur with a high-energy mechanism in the recognized position of combined humeral abduction and external rotation against a force of external rotation and horizontal extension.3 However, almost a quarter (24%) occur with a mechanism of varied upper extremity position and low-energy trauma, and 20% of injuries are either a low energy injury with little or no contact or gradual onset. These unique characteristics of shoulder instability injuries in soccer players should be accounted for during training and may imply that current training programs are suboptimal for the prevention of upper extremity injuries and shoulder injuries. Ejnisman and colleagues2 reported on the development of a Fédération Internationale de Football Association (FIFA) 11+ shoulder injury prevention program for soccer goalkeepers as one of the ways to promote training programs that address the risk of shoulder injuries.
Reporting on the management of severe shoulder injuries requiring surgery in 25 professional soccer players in England, between 2007 and 2011, Hart and Funk3 found that the majority of subjects (88%) reported a dislocation as a feature of their presentation. Twenty-one (84%) subjects were diagnosed with labral injuries, of which 7 had an associated Hill-Sachs lesion. Two (8%) subjects were diagnosed with rotator cuff tears requiring repair, and 2 (8%) subjects had a combination of rotator cuff and labral injury repair. All patients underwent arthroscopic repair, except for 5 who had a Latarjet coracoid transfer. Post-surgery, all players were able to return to unrestricted participation in soccer at a mean of 11.4 weeks, with no significant difference between goalkeepers and outfield players and no recurrences at a mean of 91 weeks’ follow-up.
Up to one-third of shoulder instability injuries in soccer players are reported to be recurrences,1,3 which emphasizes the need to carefully assess soccer players before clearing them to return to play. These data raise the controversy over the treatment of first time shoulder dislocators and may support early surgical intervention.7-9 In terms of the preferred surgical intervention in these cases, Balg and Boileau10 suggested a simple scoring system based on factors derived from a preoperative questionnaire, physical examination, and anteroposterior radiographs to help distinguish between patients who will benefit from an arthroscopic anterior stabilization using suture anchors and those who will require a bony procedure (open or arthroscopic). Cerciello and colleagues11 reported excellent results for bony stabilization (modified Latarjet) in a population of 26 soccer players (28 shoulders) affected by chronic anterior instability. Only 1 player did not return to soccer, and 18 players (20 shoulders, 71%) returned to the same level. One re-dislocation was noted in a goalkeeper 74 months after surgery.
An injury to the ACJ has been previously reported to be the most prevalent type of shoulder injury in contact sports.12In soccer, injury to the ACJ is responsible for 18% of upper extremity injuries, and the majority (72%) are sprains.1Interestingly, but unsurprisingly, implications of such an injury differ significantly between goalkeepers and outfield players with up to 3 times longer required absence periods for goalkeepers vs outfield players sustaining the same injury.
ACJ injury is commonly the result of a direct fall on the shoulder with the arm adducted or extended. Six grades of ACJ injuries have been described and distinguished by the injured anatomical structure (acromioclavicular ligaments and coracoclavicular ligaments) and the direction and magnitude of clavicular dislocation.13,14 Presentation will usually include anterior shoulder pain, a noticeable swelling or change in morphology of the lateral end of the clavicle (mainly in dislocation types), and sharp pain provoked by palpation of the ACJ. Radiographic imaging will confirm the diagnosis and help with identifying the specific grade/type of injury.
Decision making and management of acute ACJ injury should be based on the type/grade of injury. Nonoperative treatment is recommended for types I and II, and most athletes have a successful outcome with a full return to play.12Types IV, V, and VI are treated early with operative intervention, mostly due to the morbidity associated with prolonged dislocation of the joint and subsequent soft tissue damage.12 Treatment of type III injury remains controversial. Pereira-Graterol and colleagues15 reported the effectiveness of clavicular hook plate (DePuy Synthes) in the surgical stabilization of grade III ACJ dislocation in 11 professional soccer players. At a mean follow-up of 4 years, they showed excellent functional results with full shoulder range of motion at 5 weeks and latest return to soccer at 6 months. The hook plate was removed after 16 weeks in 10 patients in whom no apparent complication was observed.
Continue to: THE ELBOW...
THE ELBOW
Ekstrand and colleagues1 reported that 10% of all upper extremity injuries in professional soccer players are elbow injuries, of which only 19% are considered severe injuries that require more than 28 days of absence from playing soccer. The most common elbow injuries in their cohort were elbow medial collateral ligament (MCL) sprain and olecranon bursitis.
Elbow MCL is the primary constraint of the elbow joint to valgus stress, and MCL sprain occurs when the elbow is subjected to a valgus, or laterally directed force, which distracts the medial side of the elbow, exceeding the tensile properties of the MCL.16 A thorough physical examination that includes valgus stress tests through the arc of elbow flexion and extension to elicit a possible subjective feeling of apprehension, instability, or localized pain is essential for optimal evaluation and treatment.16,17 Imaging studies (X-ray and stress X-rays, dynamic ultrasound, computed tomography [CT], magnetic resonance imaging [MRI], and MR arthrography) have a role in further establishing the diagnosis and identifying possible additional associated injuries.16 The treatment plan should be specifically tailored to the individual athlete, depending on his demands and the degree of MCL injury. In soccer, which is a non-throwing shoulder sport, nonoperative treatment should be the preferred initial treatment in most cases. Ekstrand and colleagues1 showed that this injury requires a mean of 4 days of absence from soccer for outfield players and a mean of 21 days of absence from soccer for goalkeepers, thereby indicating more severe sprains and cautious return to soccer in goalkeepers. Athletes who fail nonoperative treatment are candidates for MCL reconstruction.16
The olecranon bursa is a synovium-lined sac that facilitates gliding between the olecranon and overlying skin. Olecranon bursitis is characterized by accumulation of fluid in the bursa with or without inflammation. The fluid can be serous, sanguineous, or purulent depending on the etiology.18 In soccer, traumatic etiology is common, but infection secondary to cuts or scratches of the skin around the elbow or previous therapeutic injections around the elbow should always be ruled out. Local pain, swelling, warmth, and redness are usually the presenting symptoms. Aseptic olecranon bursitis may be managed non-surgically with ‘‘benign neglect’’ and avoidance of pressure to the area, non-steroidal anti-inflammatory drugs, needle aspiration, corticosteroid injection, compression dressings, and/or padded splinting; whereas acute septic bursitis requires needle aspiration for diagnosis, appropriate oral or intravenous antibiotics directed toward the offending organism, and, when clinically indicated, surgical evacuation/excision of the bursa.18 When treating this condition with cortisone injection, possible complications, such as skin atrophy, secondary infection, and chronic local pain, have been reported and should be considered.19
Severe elbow injuries in professional athletes in general,20-22 and soccer players specifically, are elbow subluxations/dislocations and elbow fracture. The mechanism of injury is usually contact injury with an opponent player or a fall on the palm with the arm extended. Posterolateral is the most common type of elbow dislocation. Elbow dislocations are further classified into simple (no associated fractures) and complex (associated with fractures) categories.22 Simple dislocations are usually treated with early mobilization after closed reduction; it is associated with a low risk for re-dislocation and with generally good results. The complex type of elbow fracture dislocation is more difficult to treat, has higher complication and re-dislocation rates, and requires operative treatment in most cases compared with simple dislocation.22 The “terrible triad” of the elbow (posterior elbow dislocation, radial head fracture, and coronoid fracture) represents a specific complex elbow dislocation scenario that is difficult to manage because of conflicting aims of ensuring elbow stability while maintaining early range of motion.22
Isolated fracture around the elbow should be treated based on known principles of fracture management: mechanism of injury, fracture patterns, fracture displacement, intra-articular involvement, soft tissue condition, and associated injuries.
Continue to: THE WRIST...
THE WRIST
Ekstrand and colleagues1 reported that 5% of all upper extremity injuries in their cohort of professional soccer players are wrist injuries, of which, only 2% are considered severe injuries that require >28 days of absence from playing soccer. The more common wrist injuries in soccer, which is considered a high-impact sport, are fractures (distal radius, scaphoid, capitate), and less reported injuries are dislocations (lunate, perilunate) and ligamentous injuries or tears (scapholunate ligament).23
Distal radius fractures in high-impact sports, like soccer, usually occur as a result of a fall on an out-stretched hand and will usually be more comminuted, displaced, and intra-articular compared with low-impact sports.23 All these aforementioned characteristics usually indicate surgical management of open reduction and internal fixation, which will allow for rapid start of rehabilitation and return to play.
Scaphoid fracture is the most common carpal bone fracture and presents unique challenges in terms of diagnosis and optimal treatment24 in professional athletes. A typical injury scenario would be a player falling on an outstretched hand and sustaining a scaphoid fracture during a match or training session. The player may acknowledge some wrist pain but will often continue to play with minimal or no limitation. As wrist pain and swelling become more evident after the match/training session, the player will seek medical evaluation.24 A complete wrist and upper extremity examination should be performed in addition to a specific assessment, which includes palpation of the distal scaphoid pole at the distal wrist flexion crease, palpation of the scaphoid waist through the wrist snuff box, and palpation dorsally just distal to the Lister tubercle at the scapholunate joint. Any wrist injury that results in decreased range of motion, snuff box swelling, and scaphoid tenderness should be further evaluated with imaging. Plain radiographs with special scaphoid views are the initial preferred imaging studies, but occult fracture will require an additional study such as a bone scan, CT, or MRI. Several studies have validated the benefit of MRI and the fact that it may outweigh the costs associated with lost productivity from unnecessary cast immobilization, especially in elite athletes.23-25Casting the patient with a nondisplaced scaphoid waist fracture has been the traditional treatment; however, stiffness, weakness, and deconditioning that can occur with long-term casting required for scaphoid fractures are significant impairments for the professional athlete and usually end the player’s season. Surgical treatment, which was traditionally indicated for displaced or proximal pole fractures, is currently also considered for non-displaced scaphoid waist fractures in professional athletes. This treatment allows for a rapid return to the rehabilitation of the extremity and possible early return to professional sport. In view of the known complications (eg, malunion, nonunion, and avascular necrosis), return to soccer can be considered when imaging confirms advanced healing, which some consider as at least 50% of bone fracture bridging on CT scan, no pain, excellent motion, and at least 80% of normal grip strength.24 Outfield players can return to play with a protective cast or brace until full healing is observed on imaging.
Continue to: THE HAND/FINGERS/THUMB...
THE HAND/FINGERS/THUMB
Almost a quarter of upper extremity injuries in professional soccer players were reported to involve the hand, fingers, and thumb. A quarter of them were classified as severe injuries requiring >28 days of absence from playing soccer.1Specifically, hand metacarpal and phalanx fractures are the most common reported injuries in sports in general,26 and in soccer,1 and account for 14% of all upper extremity injuries1 in professional soccer players. Goalkeepers require a functional hand to play, whereas an outfielder can play with protection on the injured area; thus, the period of absence from soccer in these injuries is significantly different between goalkeepers and outfielders with more than 4 times longer absence from soccer for a goalkeeper compared with an outfielder. The fifth ray has been shown to be the most frequently fractured ray in the hand in soccer with 51.7% of all hand fractures reported.26 The common mechanism is a full hit on the hand, and a direct hit from the ball is another possible mechanism in goalkeepers.
In general, the diagnosis of hand injuries requires evaluation of the mechanism of injury and injury symptoms, proper and comprehensive physical examination of the whole extremity, and prompt imaging. In most cases, plain radiographs in several projections will suffice for the diagnosis of obvious fractures, but CT scan is an additional modality that allows for improved appreciation of occult or complex and comminuted fracture patterns. MRI or ultrasound can be used additionally whenever associated soft tissue injury is suspected. Optimal management of the hand is based on the specific characteristics of the fractures, which include location, direction of the fracture line, presence of comminution, displacement, articular involvement, and associated soft tissue injury. Nondisplaced extra-articular fractures often can be treated with buddy taping or splinting, whereas intra-articular fractures often require surgical treatment. Displaced fractures of the hand have a tendency to angulate volarly because of attachments of the interosseous muscles. Marginal fractures or avulsion fractures involving the metacarpals or phalanges can be sentinels of serious associated soft tissue injuries.27
Phalangeal fractures can potentially affect the function of the entire hand; therefore, no malrotation is acceptable for phalangeal fractures because they can lead to overlap and malalignment of the digit. Displaced or malrotated fractures should be reduced either by closed or open techniques. Acceptable reduction is <6 mm of shortening, <15° of angulation, and no rotational deformity.27,28 Nondisplaced phalangeal fractures can be treated nonoperatively with buddy taping and splinting with good results.27 Interphalangeal (IP) dislocations can be reduced on the sidelines and then taped or splinted. Any injury with a force significant enough to cause joint dislocation indicates further evaluation for associated fractures and ligamentous injury or tear. The proximal interphalangeal (PIP) joint is the most common IP joint dislocation and is usually a dorsal dislocation. Reduction is often achieved by traction and flexion of the middle phalanx,27 followed by splinting of the finger with the PIP in 30° of flexion or an extension block splint.29 Successful reduction with no associated intra-articular fractures involving more than a third of the joint can be further managed nonoperatively with the splint, allowing 2 to 4 weeks for the volar plate, joint capsule, and collateral ligaments to heal. Additional 2 to 4 weeks of splinting with buddy taping to the adjacent finger is usually recommended.29
The “Mallet finger” injury can be observed in goalkeepers and is caused by a flexion force on the tip of the finger while the distal interphalangeal (DIP) joint is extended. This force results in tearing of the extensor tendon or an avulsion fracture at the tendinous attachment on the dorsal lip of the distal phalangeal base. The classic mechanism of injury is an extended finger struck on the tip by a ball. Physical examination will indicate loss of DIP joint active extension, and the joint rests in an abnormally flexed position. Treatment typically consists of splinting the DIP joint in extension for 6 to 8 weeks. Operative treatment is reserved for severe injuries or fractures involving greater than one-third of the articular surface of the DIP joint or with failed nonoperative treatment.27
Metacarpal fractures can be subdivided into distal, metacarpal neck, metacarpal shaft, and metacarpal base fractures. Metacarpal shaft fractures raise a specific concern regarding rotation, because even a small degree of rotation can create a substantial degree of deformity at the fingertip. This concern must be addressed during evaluation of the player. Fractures of the metacarpal base most commonly involve the fourth and fifth metacarpals and are often reduced easily but have a tendency to re-subluxate, which may indicate operative treatment. Most fractures of the metacarpals are low energy and result in simple fracture patterns that can be treated nonoperatively. Open reduction is reserved for high-energy trauma, fractures with excessive angulation, or multiple fractures.27
Continue to: An important subgroup of metacarpal injuries...
An important subgroup of metacarpal injuries involves the base of the thumb. These injuries result from an axial load applied to the thumb. The most common injury is the “Bennett fracture,” which is an intra-articular fracture or dislocation involving the base of the first metacarpal. Bennett fractures are unstable fractures; unless properly recognized and treated, this intra-articular fracture subluxation may result in an unstable arthritic first carpometacarpal joint. These fractures are most commonly treated with closed or open reduction combined with internal fixation.27 “Rolando fractures” are similar in location and etiology but are comminuted and usually require operative treatment.27, 29
Another common hand injury found in soccer goalkeepers and involving the base of the thumb is disruption of the ulnar collateral ligament (UCL) of the first metacarpophalangeal (MCP) joint as a result of an acute radial or valgus stress on the thumb. Known as “gamekeeper’s thumb” or “skier’s thumb,” this injury can occur in the form of an avulsion fracture, an isolated ligament tear, or combined fracture and ligament rupture. On examination, swelling and tenderness over the thumb UCL are observed. A MCP joint stress test should be performed by gently applying a radially directed force to the thumb while stabilizing the metacarpal bone at both 0° and 30° at the MCP joint. Increased laxity, a soft or nonexistent end point, and gaping of the joint, as compared with the contralateral side, will indicate this injury.29 Radiographs may show a small avulsion fracture fragment at the ulnar aspect of the base of the first metacarpal and at the attachment of the UCL. A Stener lesion is an abnormality that occurs when the thumb adductor muscle aponeurosis interposes between the 2 ends of the ruptured UCL, preventing UCL healing by immobilization alone. Ultrasound and MRI are additional imaging modalities that can assist with the diagnosis of a Stener lesion. The presence of a Stener lesion is a prime indication for surgical intervention. A nondisplaced fracture or isolated ligament injury with no evidence of a Stener lesion can be treated nonoperatively with splinting of the thumb and may lead to healing and restoration of stability. However, in professional players, surgical repair is often times preferred.27
CONCLUSION
Upper extremity injuries are less common injuries among soccer players, but their prevalence is on the rise in recent years. Modern playing tactics and the increase in participation in soccer in younger age groups may be 2 contributing factors to this rise. Given the characteristics of their unique playing role and specific demands, the risk for upper extremity injuries among goalkeepers is significantly higher than that in outfielders and will usually result in a long absence period from soccer before they return to play. A thorough understanding of the mechanism of injury, players’ complaints and presentation, osseous and soft tissue involvement based on a systematic physical examination, imaging features, and treatment options is important for the optimal care of the players. Prompt and accurate diagnosis and appropriate management are essential for improved outcomes and timely return to play.
1. Ekstrand J, Hagglund M, Tornqvist H, et al. Upper extremity injuries in male elite football players. Knee Surg Sports Traumatol Arthrosc. 2013;21(7):1626-1632. doi:10.1007/s00167-012-2164-6.
2. Ejnisman B, Barbosa G, Andreoli CV, et al. Shoulder injuries in soccer goalkeepers: Review and development of a FIFA 11+ shoulder injury prevention program. Open Access J Sports Med. 2016;7:75-80. doi:10.2147/OAJSM.S97917.
3. Hart D, Funk L. Serious shoulder injuries in professional soccer: Return to participation after surgery. Knee Surg Sports Traumatol Arthrosc. 2015;23(7):2123-2129. doi:10.1007/s00167-013-2796-1.
4. Longo UG, Loppini M, Berton A, Martinelli N, Maffulli N, Denaro V. Shoulder injuries in soccer players. Clin Cases Miner Bone Metab. 2012;9(3):138-141.
5. Faude O, Rossler R, Junge A. Football injuries in children and adolescent players: Are there clues for prevention? Sports Med. 2013;43(9):819-837. doi:10.1007/s40279-013-0061-x.
6. Ekstrand J, Hagglund M, Walden M. Injury incidence and injury patterns in professional football: The UEFA injury study. Br J Sports Med. 2011;45(7):553-558. doi:10.1136/bjsm.2009.060582.
7. Boone JL, Arciero RA. First-time anterior shoulder dislocations: Has the standard changed? Br J Sports Med. 2010;44(5):355-360. doi:10.1136/bjsm.2009.062596.
8. Handoll HH, Almaiyah MA, Rangan A. Surgical versus non-surgical treatment for acute anterior shoulder dislocation. Cochrane Database Syst Rev. 2004;(1):CD004325.
9. Kirkley A, Werstine R, Ratjek A, Griffin S. Prospective randomized clinical trial comparing the effectiveness of immediate arthroscopic stabilization versus immobilization and rehabilitation in first traumatic anterior dislocations of the shoulder: Long-term evaluation. Arthroscopy. 2005;21(1):55-63.
10. Balg F, Boileau P. The instability severity index score. A simple pre-operative score to select patients for arthroscopic or open shoulder stabilisation. J Bone Joint Surg Br. 2007;89(11):1470-1477.
11. Cerciello S, Edwards TB, Walch G. Chronic anterior glenohumeral instability in soccer players: Results for a series of 28 shoulders treated with the latarjet procedure. J Orthop Traumatol. 2012;13(4):197-202. doi:10.1007/s10195-012-0201-3.
12. Bishop JY, Kaeding C. Treatment of the acute traumatic acromioclavicular separation. Sports Med Arthrosc Rev. 2006;14(4):237-245. doi:10.1097/01.jsa.0000212330.32969.6e.
13. de Putter CE, van Beeck EF, Burdorf A, et al. Increase in upper extremity fractures in young male soccer players in the netherlands, 1998-2009. Scand J Med Sci Sports. 2015;25(4):462-466. doi:10.1111/sms.12287.
14. Rockwood CJ, Williams G, Young D. Disorders of the acromioclavicular joint. In: Rockwood CJ, Matsen FA III, eds. The Shoulder. 2nd ed. Philadelphia: WB Saunders; 1998:483-553.
15. Pereira-Graterol E, Alvarez-Diaz P, Seijas R, Ares O, Cusco X, Cugat R. Treatment and evolution of grade III acromioclavicular dislocations in soccer players. Knee Surg Sports Traumatol Arthrosc. 2013;21(7):1633-1635. doi:10.1007/s00167-012-2186-0.
16. Rahman RK, Levine WN, Ahmad CS. Elbow medial collateral ligament injuries. Curr Rev Musculoskelet Med. 2008;1(3-4):197-204. doi:10.1007/s12178-008-9026-3.
17. Redler LH, Watling JP, Ahmad CS. Physical examination of the throwing athlete's elbow. Am J Orthop. 2015;44(1):13-18.
18. Sayegh ET, Strauch RJ. Treatment of olecranon bursitis: A systematic review. Arch Orthop Trauma Surg. 2014;134(11):1517-1536. doi:10.1007/s00402-014-2088-3.
19. Weinstein PS, Canoso JJ, Wohlgethan JR. Long-term follow-up of corticosteroid injection for traumatic olecranon bursitis. Ann Rheum Dis. 1984;43(1):44-46.
20. Carlisle JC, Goldfarb CA, Mall N, Powell JW, Matava MJ. Upper extremity injuries in the national football league: Part II: Elbow, forearm, and wrist injuries. Am J Sports Med. 2008;36(10):1945-1952. doi:10.1177/0363546508318198.
21. Dizdarevic I, Low S, Currie DW, Comstock RD, Hammoud S, Atanda A Jr. Epidemiology of elbow dislocations in high school athletes. Am J Sports Med. 2016;44(1):202-208. doi:10.1177/0363546515610527.
22. Saati AZ, McKee MD. Fracture-dislocation of the elbow: Diagnosis, treatment, and prognosis. Hand Clin. 2004;20(4):405-414.
23. Bancroft LW. Wrist injuries: A comparison between high- and low-impact sports. Radiol Clin North Am. 2013;51(2):299-311. doi:10.1016/j.rcl.2012.09.017.
24. Belsky MR, Leibman MI, Ruchelsman DE. Scaphoid fracture in the elite athlete. Hand Clin. 2012;28(3):78, vii. doi:10.1016/j.hcl.2012.05.005.
25. Mallee W, Doornberg JN, Ring D, van Dijk CN, Maas M, Goslings JC. Comparison of CT and MRI for diagnosis of suspected scaphoid fractures. J Bone Joint Surg Am. 2011;93(1):20-28. doi:10.2106/JBJS.I.01523.
26. Aitken S, Court-Brown CM. The epidemiology of sports-related fractures of the hand. Injury. 2008;39(12):1377-1383. doi:10.1016/j.injury.2008.04.012.
27. Peterson JJ, Bancroft LW. Injuries of the fingers and thumb in the athlete. Clin Sports Med. 2006;25(3):viii.
28. Walsh JJ 4th. Fractures of the hand and carpal navicular bone in athletes. South Med J. 2004;97(8):762-765.
29. Hong E. Hand injuries in sports medicine. Prim Care. 2005;32(1):91-103.
1. Ekstrand J, Hagglund M, Tornqvist H, et al. Upper extremity injuries in male elite football players. Knee Surg Sports Traumatol Arthrosc. 2013;21(7):1626-1632. doi:10.1007/s00167-012-2164-6.
2. Ejnisman B, Barbosa G, Andreoli CV, et al. Shoulder injuries in soccer goalkeepers: Review and development of a FIFA 11+ shoulder injury prevention program. Open Access J Sports Med. 2016;7:75-80. doi:10.2147/OAJSM.S97917.
3. Hart D, Funk L. Serious shoulder injuries in professional soccer: Return to participation after surgery. Knee Surg Sports Traumatol Arthrosc. 2015;23(7):2123-2129. doi:10.1007/s00167-013-2796-1.
4. Longo UG, Loppini M, Berton A, Martinelli N, Maffulli N, Denaro V. Shoulder injuries in soccer players. Clin Cases Miner Bone Metab. 2012;9(3):138-141.
5. Faude O, Rossler R, Junge A. Football injuries in children and adolescent players: Are there clues for prevention? Sports Med. 2013;43(9):819-837. doi:10.1007/s40279-013-0061-x.
6. Ekstrand J, Hagglund M, Walden M. Injury incidence and injury patterns in professional football: The UEFA injury study. Br J Sports Med. 2011;45(7):553-558. doi:10.1136/bjsm.2009.060582.
7. Boone JL, Arciero RA. First-time anterior shoulder dislocations: Has the standard changed? Br J Sports Med. 2010;44(5):355-360. doi:10.1136/bjsm.2009.062596.
8. Handoll HH, Almaiyah MA, Rangan A. Surgical versus non-surgical treatment for acute anterior shoulder dislocation. Cochrane Database Syst Rev. 2004;(1):CD004325.
9. Kirkley A, Werstine R, Ratjek A, Griffin S. Prospective randomized clinical trial comparing the effectiveness of immediate arthroscopic stabilization versus immobilization and rehabilitation in first traumatic anterior dislocations of the shoulder: Long-term evaluation. Arthroscopy. 2005;21(1):55-63.
10. Balg F, Boileau P. The instability severity index score. A simple pre-operative score to select patients for arthroscopic or open shoulder stabilisation. J Bone Joint Surg Br. 2007;89(11):1470-1477.
11. Cerciello S, Edwards TB, Walch G. Chronic anterior glenohumeral instability in soccer players: Results for a series of 28 shoulders treated with the latarjet procedure. J Orthop Traumatol. 2012;13(4):197-202. doi:10.1007/s10195-012-0201-3.
12. Bishop JY, Kaeding C. Treatment of the acute traumatic acromioclavicular separation. Sports Med Arthrosc Rev. 2006;14(4):237-245. doi:10.1097/01.jsa.0000212330.32969.6e.
13. de Putter CE, van Beeck EF, Burdorf A, et al. Increase in upper extremity fractures in young male soccer players in the netherlands, 1998-2009. Scand J Med Sci Sports. 2015;25(4):462-466. doi:10.1111/sms.12287.
14. Rockwood CJ, Williams G, Young D. Disorders of the acromioclavicular joint. In: Rockwood CJ, Matsen FA III, eds. The Shoulder. 2nd ed. Philadelphia: WB Saunders; 1998:483-553.
15. Pereira-Graterol E, Alvarez-Diaz P, Seijas R, Ares O, Cusco X, Cugat R. Treatment and evolution of grade III acromioclavicular dislocations in soccer players. Knee Surg Sports Traumatol Arthrosc. 2013;21(7):1633-1635. doi:10.1007/s00167-012-2186-0.
16. Rahman RK, Levine WN, Ahmad CS. Elbow medial collateral ligament injuries. Curr Rev Musculoskelet Med. 2008;1(3-4):197-204. doi:10.1007/s12178-008-9026-3.
17. Redler LH, Watling JP, Ahmad CS. Physical examination of the throwing athlete's elbow. Am J Orthop. 2015;44(1):13-18.
18. Sayegh ET, Strauch RJ. Treatment of olecranon bursitis: A systematic review. Arch Orthop Trauma Surg. 2014;134(11):1517-1536. doi:10.1007/s00402-014-2088-3.
19. Weinstein PS, Canoso JJ, Wohlgethan JR. Long-term follow-up of corticosteroid injection for traumatic olecranon bursitis. Ann Rheum Dis. 1984;43(1):44-46.
20. Carlisle JC, Goldfarb CA, Mall N, Powell JW, Matava MJ. Upper extremity injuries in the national football league: Part II: Elbow, forearm, and wrist injuries. Am J Sports Med. 2008;36(10):1945-1952. doi:10.1177/0363546508318198.
21. Dizdarevic I, Low S, Currie DW, Comstock RD, Hammoud S, Atanda A Jr. Epidemiology of elbow dislocations in high school athletes. Am J Sports Med. 2016;44(1):202-208. doi:10.1177/0363546515610527.
22. Saati AZ, McKee MD. Fracture-dislocation of the elbow: Diagnosis, treatment, and prognosis. Hand Clin. 2004;20(4):405-414.
23. Bancroft LW. Wrist injuries: A comparison between high- and low-impact sports. Radiol Clin North Am. 2013;51(2):299-311. doi:10.1016/j.rcl.2012.09.017.
24. Belsky MR, Leibman MI, Ruchelsman DE. Scaphoid fracture in the elite athlete. Hand Clin. 2012;28(3):78, vii. doi:10.1016/j.hcl.2012.05.005.
25. Mallee W, Doornberg JN, Ring D, van Dijk CN, Maas M, Goslings JC. Comparison of CT and MRI for diagnosis of suspected scaphoid fractures. J Bone Joint Surg Am. 2011;93(1):20-28. doi:10.2106/JBJS.I.01523.
26. Aitken S, Court-Brown CM. The epidemiology of sports-related fractures of the hand. Injury. 2008;39(12):1377-1383. doi:10.1016/j.injury.2008.04.012.
27. Peterson JJ, Bancroft LW. Injuries of the fingers and thumb in the athlete. Clin Sports Med. 2006;25(3):viii.
28. Walsh JJ 4th. Fractures of the hand and carpal navicular bone in athletes. South Med J. 2004;97(8):762-765.
29. Hong E. Hand injuries in sports medicine. Prim Care. 2005;32(1):91-103.
TAKE-HOME POINTS
- Upper extremity injuries in soccer are not common, however they can reach up to 18% of all injuries in professional goalkeepers.
- Common injury locations in the upper extremity in soccer are the shoulder/clavicle, hand/finger/thumb, the elbow, and the wrist and most of these injuries are traumatic injuries.
- Mechanism of injury, players’ complaints and presentation, physical examination, and imaging features are all important for a proper evaluation and optimal management.
- Position of play is an important consideration in the management of upper extremity injuries in soccer. Outfield players may be able to return to play before a complete resolution of their injury, with protective accessories.
- Prompt and accurate diagnosis and appropriate management are essential for improved outcomes and timely return to play.
Schizophrenia: Recognition and Management in the ED
In this review, the author describes the symptoms and proper diagnosis of schizophrenia, focusing on the appropriate management and treatment options for patients in the ED setting.
Schizophrenia has a heterogeneous presentation including both positive and negative symptoms. It is a relatively common disorder, affecting 0.7% of the world’s population.1,2 Schizophrenia is defined by abnormalities in two or more of five domains:3
- Delusions,
- Hallucinations,
- Disorganized thinking/speech (eg, frequent derailment or incoherence),
- Grossly disorganized or abnormal motor behavior (including catatonia), or
- Negative symptoms (ie, diminished emotional expression or avolition).
Schizophrenia is defined as having two or more of the above domains. Each must present for a significant portion of time during a 1-month period (or less if successfully treated). At least one of these must be delusions, hallucinations, or disorganized speech. This must cause dysfunction in a major area, such as work, interpersonal relations, or self-care. Signs of the disturbance persist for at least 6 months. If mood symptoms exist, they may only be present for a minority of the total duration of symptoms. Symptoms are not attributable to the physiological effects of a substance (eg, a drug of abuse, a medication) or another medical condition.3
If symptoms are present for less than 1 month, then the diagnosis is brief psychotic disorder. If symptoms are present between 1 month and 6 months, then the diagnosis is schizophreniform disorder. If a patient has only delusions but does not hallucinate; possesses disorganized speech, grossly disorganized, or catatonic behavior; and negative symptoms, then the diagnosis is delusional disorder.
The exact pathophysiology of schizophrenia is not known at this time. However, it is theorized to be a mix of biological and environmental factors leading to dysfunction of the neurotransmitters dopamine and glutamate, as well as structural changes to the brain.2
Methods
There is an abundance of research on schizophrenia in the psychiatry literature; however, much of this deals with chronic management in patients with established diagnoses, thus this may be hard to translate into the realities of emergency practice. At the same time, although schizophrenia is discussed in the emergency medicine literature, most ED-based research is performed in patients with undifferentiated agitation and not specifically on one diagnostic subset. Therefore, a narrative review was utilized in order to focus in on studies that are relevant to emergency practice.
A narrative literature search was performed utilizing PubMed, the Cochrane database, and American Psychiatric Association Practice Alerts for the past 3 years; bibliographies of major psychiatric textbooks; and reviews and clinical policies in the National Guidelines Clearinghouse.
Epidemiology
The lifetime prevalence of schizophrenia is approximately 0.3% to 0.7%.2 In addition, schizophrenia is a frequently deadly disease as one meta-analysis found lifetime suicide to be 4.9 %, which is far higher than the average risk in the United States,4 which is approximately 0.5%. From 1992 to 2000 there has been a 15% increase in ED visits for psychiatric problems.5 Patients with schizophrenia have been found to be significantly more likely to be “high utilizers” of ED services in comparison to those with other psychiatric diagnoses.6
The increase in psychiatric presentations to EDs is coupled with a lack of psychiatrists in many areas of the United States. The American Hospital Association has reported that 40% of American hospitals have difficulty maintaining adequate psychiatric coverage to meet patient demand in the ED, forcing many emergency medicine physicians to act as a primary psychiatrist.7
Differential Diagnosis
The characteristic symptoms of schizophrenia can be present in many other illnesses; therefore, the emergency clinician must be able to distinguish schizophrenia from other illnesses presenting with a psychotic component. Furthermore, schizophrenia is a diagnosis of exclusion as the diagnosis can only be made after all medical etiologies of the symptoms have been excluded.
Items on the differential include delirium, a substance induced psychosis,8 which can be caused by both substance intoxication and substance withdrawal; as well as psychosis caused by another medical condition.8 It should be stressed that a prior diagnosis of schizophrenia does not rule out a medical etiology of a patient’s current psychotic episode; therefore, a thorough history and examination is fundamental, even in patients with a known history of psychiatric illness. Moreover, many patients with schizophrenia present with a medical chief complaint, despite being symptomatic regarding their schizophrenia. In the latter case, providers must be vigilant regarding any medical comorbidities, particularly when patients are actively psychotic.
Finally, within the psychiatric disorders there are multiple disorders other than schizophrenia that may have psychotic features, including depression with psychotic features, bipolar disorder with psychotic features, and the other schizophrenia spectrum disorders; including brief psychotic disorder, schizophreniform disorder, schizoaffective disorder, and delusional disorder.
Emergency Department Evaluation
The process of evaluation of psychiatric patients who present to the ED should be directed towards a determination as to whether hospitalization is warranted, treatment of underlying medical condition is needed, or psychiatric care is indicated. New onset of psychiatric illness will commonly call for extensive evaluation, whereas patients with chronic schizophrenia may not need testing but may need psychiatric hospitalization.9 Additionally, emergency clinicians are frequently requested to preform “medical clearance” by their psychiatrist colleagues, before a patient can be transferred to a psychiatry department or psychiatric hospital. However, this is a poor term and a better nomenclature would be “assess for medical stability.”9 Emergency clinicians are, in essence, being asked whether the patient is medically stable enough for transfer to a unit or hospital where there is little or no medical support. Prior to transfer to a psychiatric unit, a patient’s non-psychiatric medical conditions should be stable enough for outpatient management.
Interacting with Patients Suffering from Psychosis
Although providers should not pretend to interact in the patient’s reality, they should express understanding in what the patient thinks or feels and kindly direct them back to the current situation.10 It is important for providers in the acute setting to be non-judgmental about reasons for relapse (in particular non-adherence to medication or substance misuse).10 Challenging the patient, while at times useful, is best done once the patient is stabilized and in a controlled environment.
History
The history should focus on age of onset; prior history of psychiatric symptoms; history of medical illness and use or abuse of illicit substances, as well as medications; the stability of psychotic symptoms; and if hallucinations are present, whether they are auditory or visual in nature.10 Table 1 highlights the differences between a patient presenting with a psychiatric complaint and the psychotic symptoms secondary to a medical illness. Table 2 provides salient differences between the examination of a patient with psychosis due to a psychiatric illness or another medical condition. Additionally, Table 3 lists various features, which differentiates schizophrenia from other psychiatric illnesses.
Additionally, patients with schizophrenia can become just as physically ill as anyone else; however, they may have difficulty expressing their specific physical complaints. Furthermore, patients with schizophrenia have a pronounced incidence of early death compared to the general population.11 Therefore, performing a simple physical examination and paying attention to vital signs can rule out or detect a great deal of organic pathology.12Once a diagnosis of schizophrenia is made, the next priority is to determine whether the patient is a threat to themselves or others and whether this threat is imminent or not. Statements of suicidal or homicidal ideation, intent, and/or plan are the basic methods of screening for danger. However, physicians also need to screen for command auditory hallucinations, prior history of suicidal behavior, and violence. Finally, even in a patient with previously diagnosed schizophrenia, screening must also be done for substance intoxication and withdrawal.
Diagnostic Studies
The indications for diagnostic studies are influenced by whether the presentation is experiencing first break psychoses vs a presentation in a patient with known schizophrenia. In the former case there is a greater need for laboratory and other diagnostic tests. In the latter case minimal testing will likely be necessary. Patients with new onset psychiatric symptoms generally need extensive laboratory and radiographic evaluations, including a computed tomography scan of the head.9
For patients who have an established schizophrenia, extensive evidence supports that there is no need for testing and indeed more often results in false positives,11,13and selective testing based on findings in the history and physical examination is the correct strategy.11
Complicating the decision to obtain laboratory tests is the fact that many psychiatric facilities rely on referring hospitals to perform baseline testing, as they do not have the resources to perform the tests or to manage any identified abnormalities. Therefore, some tests are needed prior to transfer. In addition, at times, baseline laboratory tests are needed prior to starting antipsychotics.14
Additionally, some tests are unlikely to affect immediate disposition or treatment but may be useful to psychiatrists for long-term treatment planning.15 A common disagreement between psychiatrists and emergency providers is the urine toxicology screen or a blood alcohol level. Urine toxicology or a blood alcohol level does not usually affect initial treatment and disposition in the ED.11,16 However, it may be invalid if collected subsequent to a transfer from the ED, and can determine whether a patient requires specific substance use disorder treatment or not.16-18 Therefore, many psychiatric providers will request a urine toxicology screening or a blood alcohol level before accepting a transfer. Please note, urine toxicology screens only identify a small number of toxic ingestions. Therefore, a negative screen does not eliminate drug abuse or overdose.
Finally, in the case that a diagnostic examination is being done to satisfy a requirement for an institution to accept a patient or to assist in long-term treatment planning, disposition should not be delayed in order to wait for the result to return.11
Treatment
Unfortunately, no level I or II studies have been found that examine the indications for treatment of the psychiatric patient in the ED.9 Additionally, in the ED, there is an ill-defined difference between the use of psychotropic medications as an intervention after patient assessment and plan of care, and the use of these medications to control behavior without an assessment and treatment plan.9 Therefore, many treatment recommendations have been made based on studies in other settings.
The first line treatment for a patient with schizophrenia is monotherapy with an antipsychotic agent other than clozapine.19-21 (Clozapine is not used as a first line agent due to its high side effect burden and the testing necessary before starting.) This is supported by multiple randomized controlled trials and is the standard of care.22-24 The response rates in treatment with antipsychotics in studies specifically designed to examine treatment of first-episode schizophrenia are high, ranging from 46% to 96%.23 Therefore, the emergency medicine provider can confidently reassure patients and families that antipsychotic medications are effective in treating schizophrenia. Loading doses of antipsychotics should not be used.19
Although there are only small and inconsistent differences between different antipsychotics, other than clozapine, with respect to efficacy, there are large differences in adverse effect profiles.25-30Therefore, the choice of antipsychotic medication is generally made based on previous response to individual antipsychotic medications and relative side effects.31
If extrapyramidal symptoms, including tardive dyskinesia, are of particular concern to a patient, then second generation or low potency first generation antipsychotics should be used.25,32 If a patient complains of previous problems with sedation, then haloperidol or aripiprazole should be preferred.25 Haloperidol, aripiprazole, or amisulpride should be considered for patients who are particularly concerned about weight gain, or who may be at the greatest risk of weight gain.25
If there is no response to medication after 4 weeks,32 despite dose optimization, a change in antipsychotic should be considered.33,34 Where there is partial response, the patient should be re-assessed after 8 weeks unless there are significant adverse effects.14,19,20,34 A combination of different antipsychotic medications should not be used, except during transitional periods when patients are being switched from one antipsychotic to another, or when used for clozapine augmentation.14After an acute episode has passed, providers can consider offering depot/long-acting injectable antipsychotic medication to people with schizophrenia who would prefer such treatment; as long acting injectable antipsychotics have been shown to reduce medication non-adherence.35,36 Long-acting depot antipsychotics should not be used for acute episodes because it may take 3 to 6 months for the medications to reach a stable state.14Once a patient achieves a remission of their symptoms, patients are recommended to stay on their antipsychotic medication for at least 2 years since their last acute exacerbation.14,34 For maintenance therapy,the antipsychotic dose should be reduced gradually to the lowest possible effective dose, which should not be lower than half of the effective dose during the acute phase.37
Clozapine should be offered to people with schizophrenia whose illness has not responded adequately to treatment with adequate doses of at least two different antipsychotic drugs.38-41There is no indication to using dual antipsychotic agents on patients with schizophrenia prior to starting a clozapine trial.42,43
Non-Pharmacological Intervention
There is strong evidence that people diagnosed with schizophrenia benefit from psychosocial treatments in addition to pharmacological treatment. Multiple randomized controlled studies, as well as national guidelines, have demonstrated cognitive behavioral therapy and family therapy to be effective for the treatment of schizophrenia.44-51 Particular focuses of therapy include communication skills, problem solving, psychoeducation, and assisting with family conflict,18,19 which are feasible in an emergency setting.52
Recent studies have shown promise using a “crisis intervention” approach as an alternative option to the hospital or emergency medical services systems. Treatment usually involves a combination of medication as well as counseling (practical help with living skills and support for close family members). After the crisis has been stabilized, sufferers are carefully introduced to other models of care more suited for the chronic phases of psychiatric illnesses.53 This is particularly important as psychosocial instability, such as changes in the psychosocial environment (ie, a primary caretaker going on vacation, or a patient being distanced from their family or support environment) have been associated with relapse.19
Acute Agitation
The vast majority of patients suffering from mental illness, including schizophrenia, are not violent and only a small proportion of the violence in our society can be attributed to persons who are mentally ill.54 Furthermore, people with psychiatric disabilities are far more likely to be victims than perpetrators of violent crime.55 However, schizophrenia and related disorders are associated with substantially increased rates of violence and violent offences compared to those who did not, independent of substance use.56,57 Furthermore, the majority of acute hospital assaults occur in the ED.58
Two emergency medicine evidence-based guidelines state a benzodiazepine (lorazepam or midazolam) or a conventional antipsychotic (droperidol or haloperidol) should be used as monotherapy for the treatment of acute undifferentiated agitation in the ED.12,59 However, in cases of patients with a diagnosis or suspicion of schizophrenia, an antipsychotic should be used in order to start a “disease modifying agent” earlier in the course of treatment.20,59,60Where possible, the same antipsychotic should be used as monotherapy for treatment of both acute agitation and standing antipsychotic medication.13The choice of medication for the treatment of acute agitation should be based on patient preference, past experience of antipsychotic treatment, the adverse effect profile, and concurrent medical history.19,58 This is supported by a case controlled study, which showed patients’ subjective experiences in the acute phase of treatment affected their long-term adherence to medication.61
Haloperidol is the most commonly used medication for agitation in patients with schizophrenia.59,62 When haloperidol is used to treat acute violence, it is almost always used in combination with a benzodiazepine unless medically compromised.62,63 Intramuscular olanzapine has been shown to be effective in managing acute aggression or agitation in patients with schizophrenia, especially where it is necessary to avoid some of the older treatments. Olanzapine causes fewer movement disorders than halopridol.64 Chlorpromazine is available in both per os and intramuscular formulations. However, chlorpromazine is associated with more side effects than other antipsychotics, including a higher incidence of prolonged QT and Torsades.65 However, where choices are limited, chlorpromazine may be the only treatment available for acute agitation. If used, the close monitoring of blood pressure is indicated.65 Inhaled loxapine received US Food and Drug Administration approval for treatment of acute agitation in 2012.66 This is a useful option for an agitated patient who is calm enough to receive an inhaler, but is unable to take medication per os.
Restrictive intervention (restraints or seclusion) should only be used if de-escalation and other preventive strategies, including medication as needed, have failed and there is potential for harm to the patient or other people if no action is taken.58
Telepsychiatry Consultation
Telepsychiatry is an approach, where by EDs that do not have access to psychiatrists can see patients in the ED itself, by forming a contract with either psychiatrists or companies to allow a psychiatrist at another location to perform an examination on a patient via video connection and advise the emergency room providers on treatment and intervention. There is very little data in the peer-reviewed literature on the use of telepsychiatry. The literature is limited to case reports, program descriptions, and randomized studies for reliability.67,68 One randomized controlled trial showed that a telepsychiatry evaluation had a reliable diagnosis and disposition when compared to a face-to-face assessment.69 One case control showed reduction in time-to-consult done in the ED, length of stay, and door-to-consult stay, once a telepsychiatry program was initiated.70 A large case control study showed that telepsychiatry recipients were more likely to receive 30-day follow-up, 90-day follow-up, and total inpatient length of stay, totaling both initial admission and subsequent admissions.71 Therefore, it is reasonable for EDs to take advantage of telepsychiatry services if no immediate psychiatrist coverage is available.
Disposition
The primary disposition question regarding a patient presenting with a psychiatric chief complaint to the ED, including schizophrenia, is whether they require a psychiatric admission or not. If a patient requires hospitalization but refuses, the emergency clinician must decide if the patient meets criteria for involuntarily hospitalization. If a patient does not require an inpatient admission or would benefit from an inpatient admission but does not meet criteria for civil commitment, then the next major question is what type of outpatient treatment is sufficient for this particular patient (Figure).
For a patient with schizophrenia to require inpatient psychiatric admission he must be either a danger to himself or others or has failed outpatient treatment. In addition, the patient must currently have undergone a clinical evaluation of any suspected medical illness, and any medical problems the patient is experiencing must be sufficiently stable to allow safe transport and treatment at a psychiatric hospital unit.9 If a patient’s medical condition is not sufficiently stable for either transport or hospitalization at a psychiatric hospital but the patient still requires inpatient psychiatric treatment, the patient should be hospitalized on a standard medical ward and receive psychiatric care on that unit.
Patients with mental illness pose an additional challenge, in that their illness often time interferes with their judgment about whether they require treatment. Because of this possible lack of capacity and the potential danger of a patient with mental illness to self or others, most jurisdictions have a legal code that allows for involuntary hospitalization of the mentally ill. However, which patients qualify for involuntary hospitalizations vary by jurisdiction. Case law criteria for emergencies range from a risk of “imminent” harm to self or others to a deterioration in the patient’s mental condition if treatment is halted.72 Providers are recommended to familiarize themselves with laws regarding inpatient civil commitment in their particular jurisdiction.
When a patient is being discharged from the ED, either because they do not require a psychiatric inpatient admission or because they don’t meet criteria for involuntary commitment, there are various outpatient dispositions available. Any patient with a diagnosis of schizophrenia should have outpatient follow-up with a mental health provider upon discharge from the ED.
For patients who have a failed community treatment in the past, who do not have a significant support network, or whose disease is too severe for a regular outpatient clinic to handle would be better suited for a disposition to a transitional program over a standard outpatient appointment; such as partial hospitalization programs, intensive outpatient programs, and community mental health treatment teams (CMHT). Assertive community treatment (ACT) teams are a form of CMHT comprised of several disciplines, including nurses, occupational therapists, psychiatrists, psychologists, and social workers.73 A large prospective cohort study found homeless patients to have made gains in multiple spheres of mental health while enrolled in ACT programs, and these benefits were retained after discharge from the ACT program.74
Conclusion
Schizophrenia is characterized primarily by delusions, hallucinations, and disorganized thinking. Emergency medicine providers are frequently at the front lines of diagnosing, initiating treatment, and managing relapses for these individuals. As schizophrenia is an exclusion, the primary task of an emergency physician is to rule out all “organic”, for lack of a better term, causes of the psychosis or disordered thinking before making a definitive diagnosis of schizophrenia. As many EDs do not have sufficient psychiatry support and many patients with schizophrenia are using the ED as their location for primary mental health, many emergency physicians are finding themselves acting as the main providers for these individuals. The primary treatment for patients with schizophrenia remains monotherapy with antipsychotic medication. Although the highest-level disposition for a patient with schizophrenia remains the inpatient psychiatry department, there are many transitional dispositions that can bridge the gap between a discharge to standard community care and an inpatient admission.
1. MacDonald AW, Schulz SC: What we know: findings that every theory of schizophrenia should explain. Schizophr Bull. 2009;35(3):493-508. doi:10.1093/schbul/sbp017.
2. McGrath J, Saha S, Chant D, Welham J. Schizophrenia: a concise overview of incidence, prevalence, and mortality. Epidemiol Rev. 2008;30:67-76. doi:10.1093/epirev/mxn001.
3. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders: DSM–5. 5th ed. Washington, DC: American Psychiatric Association; 2013.
4. Palmer BA, Pankratz VS, Bostwick JM. The lifetime risk of suicide in schizophrenia: a reexamination. Arch Gen Psychiatry. 2005;62(3):247-253.
5. American College of Emergency Physicians. Psychiatric and substance abuse survey 2008. http://newsroom.acep.org/download/ACEP%2BPsychiatric%2Band%2BSubstance%2BAbuse%2BSurvey%2B-%2BApril%2B2008.pdf. Accessed October 7, 2018.
6. Pasic J, Russo J, Roy-Byrne P. High utilizers of psychiatric emergency services. Psychiatr Serv. 2005;56(6):678-684.
7. American Hospital Association. AHA annual survery database. http://www.ahadata.com/aha-annual-survey-database-asdb/. Accessed October 7, 2018.
8. Testa A, Giannuzzi R, Daini S, Bernardini L, Petrongolo L, Gentiloni Silveri N. Psychiatric emergencies (part III): psychiatric symptoms resulting from organic diseases. Eur Rev Med Pharmacol Sci. 2013;17 Suppl 1:86-99.
9. Zun LS. Evidence-based evaluation of psychiatric patient. J Emerg Med. 2005;28(3):277-283.
10. Bryan CA, Mistovich JJ, Krost WS, Limmer DD. In two minds? EMS care of the schizophrenic patient. Recognizing the signs and symptoms of schizophrenia is key to prehospital care. EMS Mag. 2009;38(12):63-71.
11. Lukens TW, Wolf SJ, Edlow JA, et al; American College of Emergency Physicians Clinical Policies Subcommittee (Writing Committee) on Critical Issues in the Diagnosis and Management of the Adult Psychiatric Patient in the Emergency Department. Clinical policy: critical issues in the diagnosis and management of the adult psychiatric patient in the emergency department. Ann Emerg Med. 2006;47(1):79-99.
12. Crump C, Winkleby MA, Sundquist K, Sundquist J. Comorbidities and mortality in persons with schizophrenia: a Swedish national cohort study. Am J Psychiatry. 2013;170(3):324-33. doi:10.1176/appi.ajp.2012.12050599.
13. Kroll DS, Smallwood J, Chang G. Drug screens for psychiatric patients in the emergency department: evaluation and recommendations. Psychosomatics. 2013;54(1):60-66. doi:10.1016/j.psym.2012.08.007.
14. Verma S, Chan LL, Chee KS, et al; MOH Clinical Practice Guidelines Workgroup on Schizophrenia. Ministry of Health clinical practice guidelines: schizophrenia. Singapore Med J. 2011;52(7):521-525.
15. Fochtman LJ. Psychiatric perspectives on medical screening of psychiatric patients. Acad Emerg Med. 2002;9(9):963-964.
16. Tijdink JK, van den Heuvel J, Vasbinder EC, can de Ven PM, Honig A. Does on-site urine toxicology screening have an added diagnostic value in psychiatric referrals in an emergency setting? Gen Hosp Psychiatry. 2011;33(6):626-6
17. Janiak BD, Atteberry S. Medical clearance of the psychiatric patient in the emergency department. J Emerg Med. 2012;43(5):866-870. doi:10.1016/j.jemermed.2009.10.026.
18. Lehman AF, Lieberman JA, Dixon LB, et al; American Psychiatric Association; Steering Committee on Practice Guidelines. Practice guideline for the treatment of patients with schizophrenia, second edition. Am J Psychiatry. 2004;161(2 Suppl):1-56.
19. National Institute for Health and Care Excellence. Psychosis and schizophrenia in adults: prevention and management. https://www.nice.org.uk/guidance/cg178. Published February 2014. Accessed September 25, 2018.
20. Scottish Intercollegiate Guidelines Network. SIGN 131: Management of schizophrenia. https://www.sign.ac.uk/assets/sign131.pdf. Published March 2013. Accessed September 25, 2018. 15.
21. Tucci V, Siever K, Matorin A, Moukaddam N. Down the rabbit hole: emergency department medical clearance of patients with psychiatric or behavioral emergencies. Emerg Med Clin North Am. 2015;33(4):721-737. doi:10.1016/j.emc.2015.07.002.
22. Tandon R, Belmaker RH, Gattaz WF, et al; Section of Pharmacopsychiatry, World Psychiatric Association. World Psychiatric Association Pharmacopsychiatry Section statement on comparative effectiveness of antipsychotics in the treatment of schizophrenia. Schizophr Res. 2008;100(1-3):20-38. doi:10.1016/j.schres.2007.11.033.
23. Leucht S, Arbter D, Engel RR, Kissling W, Davis JM. How effective are second-generation antipsychotic drugs? A meta-analysis of placebo-controlled trials. Mol Psychiatry. 2009;14(4):429-447. doi:10.1038/sj.mp.4002136.
24. Robinson DG, Woerner MG, Delman HM, Kane JM. Pharmacological treatments for first-episode schizophrenia. Schizophr Bull. 2005;31(3):705-722.
25. Leucht S, Corves C, Arbter D, Engel RR, Li C, Davis JM. Second-generation versus first-generation antipsychotic drugs for schizophrenia: a meta-analysis. Lancet. 2009;373(9657):31-41. doi:10.1016/S0140-6736(08)61764-X.
26. Komossa K, Rummel-Kluge C, Hunger H, et al. Olanzapine versus other atypical antipsychotics for schizophrenia. Cochrane Database Syst Rev. 2010;3:CD006654. doi:10.1002/14651858.CD006654.pub2.
27. Komossa K, Rummel-Kluge C, Hunger H, et al. Amisulpride versus other atypical antipsychotics for schizophrenia. Cochrane Database Syst Rev. 2010;1:CD006624. doi:10.1002/14651858.CD006624.pub2.
28. Komossa K, Rummel-Kluge C, Hunger H, et al. Ziprasidone versus other atypical antipsychotics for schizophrenia. Cochrane Database Syst Rev. 2009;4:CD006627. doi:10.1002/14651858.CD006627.pub2.
29. Komossa K, Rummel-Kluge C, Schmid F, et al. Aripiprazole versus other atypical antipsychotics for schizophrenia. Cochrane Database Syst Rev. 2009;4:CD006569. doi:10.1002/14651858.CD006569.pub3.
30. Komossa K, Rummel-Kluge C, Schmid F, et al. Quetiapine versus other atypical antipsychotics for schizophrenia. Cochrane Database Syst Rev. 2010;1:CD006625. doi:10.1002/14651858.CD006625.pub2.
31. Crossley NA, Constante M, McGuire P, Power P. Efficacy of atypical v. typical antipsychotics in the treatment of early psychosis: meta-analysis. Br J Psychiatry. 2010;196(6):434-439. doi:10.1192/bjp.bp.109.066217.
32. Correll CU, Schenk EM. Tardive dyskinesia and new antipsychotics. Curr Opin Psychiatry. 2008;21(2):151-156. doi:10.1097/YCO.0b013e3282f53132.

33. Emsley R, Rabinowitz J, Medori R. Time course for antipsychotic treatment response in first-episode schizophrenia. Am J Psychiatry. 2006;163(4):743-745.
34. Barnes TR; Schizophrenia Consensus Group of British Association for Psychopharmacology. Evidence-based guidelines for the pharmacological treatment of schizophrenia: recommendations from the British Association for Psychopharmacology. J Psychopharmacol. 2011;25(5):567-620. doi:10.1177/0269881110391123.
35. MacEwan JP, Kamat SA, Duffy RA, et al. Hospital readmission rates among patients with schizophrenia treated with long-acting injectables or oral antipsychotics. Psychiatr Serv. 2016;67(11):1183-1188.
36. Patel MX, Taylor M, David AS. Antipsychotic long-acting injections: mind the gap. Br J Psychiatry. 2009;52:S1-S4. doi:10.1192/bjp.195.52.s1.
37. Uchida H, Suzuki T, Takeuchi H, Arenovich T, Mamo DC. Low dose vs standard dose of antipsychotics for relapse prevention in schizophrenia: meta-analysis. Schizophr Bull. 2011;37(4):788-799. doi:10.1093/schbul/sbp149.
38. Swartz MS, Stroup TS, McEvoy JP, et al. What CATIE found: results from the schizophrenia trial. Psychiatr Serv. 2008;59(5):500-506. doi:10.1176/ps.2008.59.5.500.
39. Jones PB, Barnes TR, Davies L, et al. Randomized controlled trial of the effect on Quality of Life of second- vs first-generation antipsychotic drugs in schizophrenia: Cost Utility of the Latest Antipsychotic Drugs in Schizophrenia Study (CUtLASS 1). Arch Gen Psychiatry. 2006;63(10):1079-1087.
40. Chakos M, Lieberman J, Hoffman E, Bradford D, Sheitman B. Effectiveness of second-generation antipsychotics in patients with treatment-resistant schizophrenia: a review and meta-analysis of randomized trials. Am J Psychiatry. 2001;158(4):518-526.
41. McEvoy JP, Lieberman JA, Stroup TS, et al; CATIE Investigators. Effectiveness of clozapine versus olanzapine, quetiapine, and risperidone in patients with chronic schizophrenia who did not respond to prior atypical antipsychotic treatment. Am J Psychiatry. 2006;163(4):600-610.
43. Stahl SM, Grady MM. A critical review of atypical antipsychotic utilization: comparing monotherapy with polypharmacy and augmentation. Curr Med Chem. 2004;11(3):313-327.
44. Turkington D, Sensky T, Scott J, et al. A randomized controlled trial of cognitive-behavior therapy for persistent symptoms in schizophrenia: a five-year follow-up. Schizophr Res. 2008;98(1-3):1-7.
45. Malik N, Kingdon D, Pelton J, Mehta R, Turkington D. Effectiveness of brief cognitive-behavioral therapy for schizophrenia delivered by mental health nurses: relapse and recovery at 24 months. J Clin Psychiatry. 2009;70(2):201-207.
46. Haddock G, Barrowclough C, Shaw JJ, Dunn G, Novaco RW, Tarrier N. Cognitive-behavioural therapy v. social activity therapy for people with psychosis and a history of violence: randomised controlled trial. Br J Psychiatry. 2009;194(2):152-157. doi:10.1192/bjp.bp.107.039859.
47. Lysaker PH, Davis LW, Bryson GJ, Bell MD. Effects of cognitive behavioral therapy on work outcomes in vocational rehabilitation for participants with schizophrenia spectrum disorders. Schizophr Res. 2009;107(2-3):186-191. doi:10.1016/j.schres.2008.10.018.
48. Grant PM, Huh GA, Perivoliotis D, Stolar NM, Beck AT. Randomized trial to evaluate the efficacy of cognitive therapy for low-functioning patients with schizophrenia. Arch Gen Psychiatry. 2012;69(2):121-127. doi:10.1001/archgenpsychiatry.2011.129.
49. Girón M, Fernández-Yañez A, Mañá-Alvarenga S, Molina-Habas A, Nolasco A, Gómez-Beneyto M. Efficacy and effectiveness of individual family intervention on social and clinical functioning and family burden in severe schizophrenia: a 2-year randomized controlled study. Psychol Med. 2010;40(1):73-84. doi:10.1017/S0033291709006126.
50. Bressi C, Manenti S, Frongia P, Porcellana M, Invernizzi G. Systemic family therapy in schizophrenia: a randomized clinical trial of effectiveness. Psychother Psychosom. 2008;77(1):43-49.
51. Chien WT, Lee IY. The schizophrenia care management program for family caregivers of Chinese patients with schizophrenia. Psychiatr Serv. 2010;61(3):317-320. doi:10.1176/ps.2010.61.3.317.
52. Sneed J, Balestri M, Belfi BJ. The use of dialectical behavior therapy strategies in the psychiatric emergency room. Psychotherapy Theory, Research & Practice. 2003;40(4):265-277.
53. Murphy SM, Irving CB, Adams CE, Wagar M. Crisis intervention for people with severe mental illnesses. Cochrane Database Syst Rev. 2015;12:CD001087. doi:10.1002/14651858.CD001087.pub5.
54. Mulvey EP. Assessing the evidence of a link between mental illness and violence. Hosp Community Psychiatry. 1994;45(7):663-668.
55. Hiroeh U, Appleby L, Mortensen PB, Dunn G. Death by homicide, suicide, and other unnatural causes in people with mental illness: a population-based study. Lancet. 2001;358(9299):2110-2112.
56. Van Dorn R, Volavka J, Johnson N. Mental disorder and violence: is there a relationship beyond substance use? Soc Psychiatry Psychiatr Epidemiol. 2012;47(3):487-503. doi:10.1007/s00127-011-0356-x.
57. Fazel S, Wolf A, Palm C, Lichtenstein P. Violent crime, suicide, and premature mortality in patients with schizophrenia and related disorders: a 38-year total population study in Sweden. Lancet Psychiatry. 2014;1(1):44-54.
58. National Institute for Health and Care Excellence. Violence and aggression: short-term management in mental health, health and community settings. https://www.nice.org.uk/guidance/ng10/chapter/About-this-guideline. Published May 2015. Accessed September 24, 2018.
59. Wilson MP, Pepper D, Currier GW, Holloman GH Jr, Feifel D. The psychopharmacology of agitation: consensus statement of the american association for emergency psychiatry project Beta psychopharmacology workgroup. West J Emerg Med. 2012;13(1):26-34. doi:10.5811/westjem.2011.9.6866.
63. Leucht S, Heres S, Kissling W, Davis JM. Evidence based pharmacotherapy of schizophrenia. Int J Neuropsychopharmacol. 2011;14(2):269-284. doi:10.1017/S1461145710001380.
64. Belgamwar RB, Fenton M. Olanzapine IM or velotab for acutely disturbed/agitated people with suspected serious mental illnesses. Cochrane Database Syst Rev. 2005;2:CD003729.

65. Ahmed U, Jones H, Adams CE. Chlorpromazine for psychosis induced aggression or agitation. Cochrane Database Syst Rev. 2010;4:CD007445. doi:10.1002/14651858.CD007445.pub2.
66. US Food and Drug Administration. Drug approval package. https://www.accessdata.fda.gov/drugsatfda_docs/nda/2012/022549_adasuve_toc.cfm. Accessed September 28, 2018.
67. Salmoiraghi A, Hussain S. A systematic review of the use of telepsychiatry in acute settings. J Psychiatr Pract. 2015;21(5):389-393. doi:10.1097/PRA.0000000000000103.
68. Shore JH, Hilty DM, Yellowlees P. Emergency management guidelines for telepsychiatry. Gen Hosp Psychiatry. 2007;29(3):199-206.
69. Seidel RW, Kilqus MD. Agreement between telepsychiatry assessment and face-to-face assessment for emergency department psychiatry patients. J Telemed Telecare. 2014;20(2):59-62. doi:10.1177/1357633X13519902.
70. DeVido J, Glezer A, Branagan L, Lau A, Bourgeois JA. Telepsychiatry for inpatient consultations at a separate campus of an academic medical center. Telemed J E Health. 2016;22(7):572-576. doi:10.1089/tmj.2015.0125.
71. Narasimhan M, Druss BG, Hockenberry JM, et al. Impact of a telepsychiatry program at emergency departments statewide on the quality, utilization, and costs of mental health services. Psychiatr Serv. 2015;66(11):1167-1172. doi:10.1176/appi.ps.201400122.
72. Simon RI, Shuman DW. Clinical issues in psychiatry and the law. In: Hales RE, Yudofsky SC, Roberts LW, eds. The American Psychiatric Publishing Textbook of Psychiatry. 6th ed. Washington, DC: American Psychiatric Publishing; 2014:175-204.
73. Malone D, Newron-Howes G, Simmonds S, Marriot S, Tyrer P. Community mental health teams (CMHTs) for people with severe mental illnesses and disordered personality. Cochrane Database Syst Rev. 2007;3:CD000270.
74. Rosenheck RA, Dennis D. Time-limited assertive community treatment for homeless persons with severe mental illness. Arch Gen Psychiatry. 2001;58(11):1073-1080.
75. Testa A, Giannuzzi R, Sollazzo F, Petrongolo L, Bernardini L, Daini S. Psychiatric emergencies (part I): psychiatric disorders causing organic symptoms. Eur Rev Med Pharmacol Sci. 2013;17 Suppl 1:55-64.
In this review, the author describes the symptoms and proper diagnosis of schizophrenia, focusing on the appropriate management and treatment options for patients in the ED setting.
In this review, the author describes the symptoms and proper diagnosis of schizophrenia, focusing on the appropriate management and treatment options for patients in the ED setting.
Schizophrenia has a heterogeneous presentation including both positive and negative symptoms. It is a relatively common disorder, affecting 0.7% of the world’s population.1,2 Schizophrenia is defined by abnormalities in two or more of five domains:3
- Delusions,
- Hallucinations,
- Disorganized thinking/speech (eg, frequent derailment or incoherence),
- Grossly disorganized or abnormal motor behavior (including catatonia), or
- Negative symptoms (ie, diminished emotional expression or avolition).
Schizophrenia is defined as having two or more of the above domains. Each must present for a significant portion of time during a 1-month period (or less if successfully treated). At least one of these must be delusions, hallucinations, or disorganized speech. This must cause dysfunction in a major area, such as work, interpersonal relations, or self-care. Signs of the disturbance persist for at least 6 months. If mood symptoms exist, they may only be present for a minority of the total duration of symptoms. Symptoms are not attributable to the physiological effects of a substance (eg, a drug of abuse, a medication) or another medical condition.3
If symptoms are present for less than 1 month, then the diagnosis is brief psychotic disorder. If symptoms are present between 1 month and 6 months, then the diagnosis is schizophreniform disorder. If a patient has only delusions but does not hallucinate; possesses disorganized speech, grossly disorganized, or catatonic behavior; and negative symptoms, then the diagnosis is delusional disorder.
The exact pathophysiology of schizophrenia is not known at this time. However, it is theorized to be a mix of biological and environmental factors leading to dysfunction of the neurotransmitters dopamine and glutamate, as well as structural changes to the brain.2
Methods
There is an abundance of research on schizophrenia in the psychiatry literature; however, much of this deals with chronic management in patients with established diagnoses, thus this may be hard to translate into the realities of emergency practice. At the same time, although schizophrenia is discussed in the emergency medicine literature, most ED-based research is performed in patients with undifferentiated agitation and not specifically on one diagnostic subset. Therefore, a narrative review was utilized in order to focus in on studies that are relevant to emergency practice.
A narrative literature search was performed utilizing PubMed, the Cochrane database, and American Psychiatric Association Practice Alerts for the past 3 years; bibliographies of major psychiatric textbooks; and reviews and clinical policies in the National Guidelines Clearinghouse.
Epidemiology
The lifetime prevalence of schizophrenia is approximately 0.3% to 0.7%.2 In addition, schizophrenia is a frequently deadly disease as one meta-analysis found lifetime suicide to be 4.9 %, which is far higher than the average risk in the United States,4 which is approximately 0.5%. From 1992 to 2000 there has been a 15% increase in ED visits for psychiatric problems.5 Patients with schizophrenia have been found to be significantly more likely to be “high utilizers” of ED services in comparison to those with other psychiatric diagnoses.6
The increase in psychiatric presentations to EDs is coupled with a lack of psychiatrists in many areas of the United States. The American Hospital Association has reported that 40% of American hospitals have difficulty maintaining adequate psychiatric coverage to meet patient demand in the ED, forcing many emergency medicine physicians to act as a primary psychiatrist.7
Differential Diagnosis
The characteristic symptoms of schizophrenia can be present in many other illnesses; therefore, the emergency clinician must be able to distinguish schizophrenia from other illnesses presenting with a psychotic component. Furthermore, schizophrenia is a diagnosis of exclusion as the diagnosis can only be made after all medical etiologies of the symptoms have been excluded.
Items on the differential include delirium, a substance induced psychosis,8 which can be caused by both substance intoxication and substance withdrawal; as well as psychosis caused by another medical condition.8 It should be stressed that a prior diagnosis of schizophrenia does not rule out a medical etiology of a patient’s current psychotic episode; therefore, a thorough history and examination is fundamental, even in patients with a known history of psychiatric illness. Moreover, many patients with schizophrenia present with a medical chief complaint, despite being symptomatic regarding their schizophrenia. In the latter case, providers must be vigilant regarding any medical comorbidities, particularly when patients are actively psychotic.
Finally, within the psychiatric disorders there are multiple disorders other than schizophrenia that may have psychotic features, including depression with psychotic features, bipolar disorder with psychotic features, and the other schizophrenia spectrum disorders; including brief psychotic disorder, schizophreniform disorder, schizoaffective disorder, and delusional disorder.
Emergency Department Evaluation
The process of evaluation of psychiatric patients who present to the ED should be directed towards a determination as to whether hospitalization is warranted, treatment of underlying medical condition is needed, or psychiatric care is indicated. New onset of psychiatric illness will commonly call for extensive evaluation, whereas patients with chronic schizophrenia may not need testing but may need psychiatric hospitalization.9 Additionally, emergency clinicians are frequently requested to preform “medical clearance” by their psychiatrist colleagues, before a patient can be transferred to a psychiatry department or psychiatric hospital. However, this is a poor term and a better nomenclature would be “assess for medical stability.”9 Emergency clinicians are, in essence, being asked whether the patient is medically stable enough for transfer to a unit or hospital where there is little or no medical support. Prior to transfer to a psychiatric unit, a patient’s non-psychiatric medical conditions should be stable enough for outpatient management.
Interacting with Patients Suffering from Psychosis
Although providers should not pretend to interact in the patient’s reality, they should express understanding in what the patient thinks or feels and kindly direct them back to the current situation.10 It is important for providers in the acute setting to be non-judgmental about reasons for relapse (in particular non-adherence to medication or substance misuse).10 Challenging the patient, while at times useful, is best done once the patient is stabilized and in a controlled environment.
History
The history should focus on age of onset; prior history of psychiatric symptoms; history of medical illness and use or abuse of illicit substances, as well as medications; the stability of psychotic symptoms; and if hallucinations are present, whether they are auditory or visual in nature.10 Table 1 highlights the differences between a patient presenting with a psychiatric complaint and the psychotic symptoms secondary to a medical illness. Table 2 provides salient differences between the examination of a patient with psychosis due to a psychiatric illness or another medical condition. Additionally, Table 3 lists various features, which differentiates schizophrenia from other psychiatric illnesses.
Additionally, patients with schizophrenia can become just as physically ill as anyone else; however, they may have difficulty expressing their specific physical complaints. Furthermore, patients with schizophrenia have a pronounced incidence of early death compared to the general population.11 Therefore, performing a simple physical examination and paying attention to vital signs can rule out or detect a great deal of organic pathology.12Once a diagnosis of schizophrenia is made, the next priority is to determine whether the patient is a threat to themselves or others and whether this threat is imminent or not. Statements of suicidal or homicidal ideation, intent, and/or plan are the basic methods of screening for danger. However, physicians also need to screen for command auditory hallucinations, prior history of suicidal behavior, and violence. Finally, even in a patient with previously diagnosed schizophrenia, screening must also be done for substance intoxication and withdrawal.
Diagnostic Studies
The indications for diagnostic studies are influenced by whether the presentation is experiencing first break psychoses vs a presentation in a patient with known schizophrenia. In the former case there is a greater need for laboratory and other diagnostic tests. In the latter case minimal testing will likely be necessary. Patients with new onset psychiatric symptoms generally need extensive laboratory and radiographic evaluations, including a computed tomography scan of the head.9
For patients who have an established schizophrenia, extensive evidence supports that there is no need for testing and indeed more often results in false positives,11,13and selective testing based on findings in the history and physical examination is the correct strategy.11
Complicating the decision to obtain laboratory tests is the fact that many psychiatric facilities rely on referring hospitals to perform baseline testing, as they do not have the resources to perform the tests or to manage any identified abnormalities. Therefore, some tests are needed prior to transfer. In addition, at times, baseline laboratory tests are needed prior to starting antipsychotics.14
Additionally, some tests are unlikely to affect immediate disposition or treatment but may be useful to psychiatrists for long-term treatment planning.15 A common disagreement between psychiatrists and emergency providers is the urine toxicology screen or a blood alcohol level. Urine toxicology or a blood alcohol level does not usually affect initial treatment and disposition in the ED.11,16 However, it may be invalid if collected subsequent to a transfer from the ED, and can determine whether a patient requires specific substance use disorder treatment or not.16-18 Therefore, many psychiatric providers will request a urine toxicology screening or a blood alcohol level before accepting a transfer. Please note, urine toxicology screens only identify a small number of toxic ingestions. Therefore, a negative screen does not eliminate drug abuse or overdose.
Finally, in the case that a diagnostic examination is being done to satisfy a requirement for an institution to accept a patient or to assist in long-term treatment planning, disposition should not be delayed in order to wait for the result to return.11
Treatment
Unfortunately, no level I or II studies have been found that examine the indications for treatment of the psychiatric patient in the ED.9 Additionally, in the ED, there is an ill-defined difference between the use of psychotropic medications as an intervention after patient assessment and plan of care, and the use of these medications to control behavior without an assessment and treatment plan.9 Therefore, many treatment recommendations have been made based on studies in other settings.
The first line treatment for a patient with schizophrenia is monotherapy with an antipsychotic agent other than clozapine.19-21 (Clozapine is not used as a first line agent due to its high side effect burden and the testing necessary before starting.) This is supported by multiple randomized controlled trials and is the standard of care.22-24 The response rates in treatment with antipsychotics in studies specifically designed to examine treatment of first-episode schizophrenia are high, ranging from 46% to 96%.23 Therefore, the emergency medicine provider can confidently reassure patients and families that antipsychotic medications are effective in treating schizophrenia. Loading doses of antipsychotics should not be used.19
Although there are only small and inconsistent differences between different antipsychotics, other than clozapine, with respect to efficacy, there are large differences in adverse effect profiles.25-30Therefore, the choice of antipsychotic medication is generally made based on previous response to individual antipsychotic medications and relative side effects.31
If extrapyramidal symptoms, including tardive dyskinesia, are of particular concern to a patient, then second generation or low potency first generation antipsychotics should be used.25,32 If a patient complains of previous problems with sedation, then haloperidol or aripiprazole should be preferred.25 Haloperidol, aripiprazole, or amisulpride should be considered for patients who are particularly concerned about weight gain, or who may be at the greatest risk of weight gain.25
If there is no response to medication after 4 weeks,32 despite dose optimization, a change in antipsychotic should be considered.33,34 Where there is partial response, the patient should be re-assessed after 8 weeks unless there are significant adverse effects.14,19,20,34 A combination of different antipsychotic medications should not be used, except during transitional periods when patients are being switched from one antipsychotic to another, or when used for clozapine augmentation.14After an acute episode has passed, providers can consider offering depot/long-acting injectable antipsychotic medication to people with schizophrenia who would prefer such treatment; as long acting injectable antipsychotics have been shown to reduce medication non-adherence.35,36 Long-acting depot antipsychotics should not be used for acute episodes because it may take 3 to 6 months for the medications to reach a stable state.14Once a patient achieves a remission of their symptoms, patients are recommended to stay on their antipsychotic medication for at least 2 years since their last acute exacerbation.14,34 For maintenance therapy,the antipsychotic dose should be reduced gradually to the lowest possible effective dose, which should not be lower than half of the effective dose during the acute phase.37
Clozapine should be offered to people with schizophrenia whose illness has not responded adequately to treatment with adequate doses of at least two different antipsychotic drugs.38-41There is no indication to using dual antipsychotic agents on patients with schizophrenia prior to starting a clozapine trial.42,43
Non-Pharmacological Intervention
There is strong evidence that people diagnosed with schizophrenia benefit from psychosocial treatments in addition to pharmacological treatment. Multiple randomized controlled studies, as well as national guidelines, have demonstrated cognitive behavioral therapy and family therapy to be effective for the treatment of schizophrenia.44-51 Particular focuses of therapy include communication skills, problem solving, psychoeducation, and assisting with family conflict,18,19 which are feasible in an emergency setting.52
Recent studies have shown promise using a “crisis intervention” approach as an alternative option to the hospital or emergency medical services systems. Treatment usually involves a combination of medication as well as counseling (practical help with living skills and support for close family members). After the crisis has been stabilized, sufferers are carefully introduced to other models of care more suited for the chronic phases of psychiatric illnesses.53 This is particularly important as psychosocial instability, such as changes in the psychosocial environment (ie, a primary caretaker going on vacation, or a patient being distanced from their family or support environment) have been associated with relapse.19
Acute Agitation
The vast majority of patients suffering from mental illness, including schizophrenia, are not violent and only a small proportion of the violence in our society can be attributed to persons who are mentally ill.54 Furthermore, people with psychiatric disabilities are far more likely to be victims than perpetrators of violent crime.55 However, schizophrenia and related disorders are associated with substantially increased rates of violence and violent offences compared to those who did not, independent of substance use.56,57 Furthermore, the majority of acute hospital assaults occur in the ED.58
Two emergency medicine evidence-based guidelines state a benzodiazepine (lorazepam or midazolam) or a conventional antipsychotic (droperidol or haloperidol) should be used as monotherapy for the treatment of acute undifferentiated agitation in the ED.12,59 However, in cases of patients with a diagnosis or suspicion of schizophrenia, an antipsychotic should be used in order to start a “disease modifying agent” earlier in the course of treatment.20,59,60Where possible, the same antipsychotic should be used as monotherapy for treatment of both acute agitation and standing antipsychotic medication.13The choice of medication for the treatment of acute agitation should be based on patient preference, past experience of antipsychotic treatment, the adverse effect profile, and concurrent medical history.19,58 This is supported by a case controlled study, which showed patients’ subjective experiences in the acute phase of treatment affected their long-term adherence to medication.61
Haloperidol is the most commonly used medication for agitation in patients with schizophrenia.59,62 When haloperidol is used to treat acute violence, it is almost always used in combination with a benzodiazepine unless medically compromised.62,63 Intramuscular olanzapine has been shown to be effective in managing acute aggression or agitation in patients with schizophrenia, especially where it is necessary to avoid some of the older treatments. Olanzapine causes fewer movement disorders than halopridol.64 Chlorpromazine is available in both per os and intramuscular formulations. However, chlorpromazine is associated with more side effects than other antipsychotics, including a higher incidence of prolonged QT and Torsades.65 However, where choices are limited, chlorpromazine may be the only treatment available for acute agitation. If used, the close monitoring of blood pressure is indicated.65 Inhaled loxapine received US Food and Drug Administration approval for treatment of acute agitation in 2012.66 This is a useful option for an agitated patient who is calm enough to receive an inhaler, but is unable to take medication per os.
Restrictive intervention (restraints or seclusion) should only be used if de-escalation and other preventive strategies, including medication as needed, have failed and there is potential for harm to the patient or other people if no action is taken.58
Telepsychiatry Consultation
Telepsychiatry is an approach, where by EDs that do not have access to psychiatrists can see patients in the ED itself, by forming a contract with either psychiatrists or companies to allow a psychiatrist at another location to perform an examination on a patient via video connection and advise the emergency room providers on treatment and intervention. There is very little data in the peer-reviewed literature on the use of telepsychiatry. The literature is limited to case reports, program descriptions, and randomized studies for reliability.67,68 One randomized controlled trial showed that a telepsychiatry evaluation had a reliable diagnosis and disposition when compared to a face-to-face assessment.69 One case control showed reduction in time-to-consult done in the ED, length of stay, and door-to-consult stay, once a telepsychiatry program was initiated.70 A large case control study showed that telepsychiatry recipients were more likely to receive 30-day follow-up, 90-day follow-up, and total inpatient length of stay, totaling both initial admission and subsequent admissions.71 Therefore, it is reasonable for EDs to take advantage of telepsychiatry services if no immediate psychiatrist coverage is available.
Disposition
The primary disposition question regarding a patient presenting with a psychiatric chief complaint to the ED, including schizophrenia, is whether they require a psychiatric admission or not. If a patient requires hospitalization but refuses, the emergency clinician must decide if the patient meets criteria for involuntarily hospitalization. If a patient does not require an inpatient admission or would benefit from an inpatient admission but does not meet criteria for civil commitment, then the next major question is what type of outpatient treatment is sufficient for this particular patient (Figure).
For a patient with schizophrenia to require inpatient psychiatric admission he must be either a danger to himself or others or has failed outpatient treatment. In addition, the patient must currently have undergone a clinical evaluation of any suspected medical illness, and any medical problems the patient is experiencing must be sufficiently stable to allow safe transport and treatment at a psychiatric hospital unit.9 If a patient’s medical condition is not sufficiently stable for either transport or hospitalization at a psychiatric hospital but the patient still requires inpatient psychiatric treatment, the patient should be hospitalized on a standard medical ward and receive psychiatric care on that unit.
Patients with mental illness pose an additional challenge, in that their illness often time interferes with their judgment about whether they require treatment. Because of this possible lack of capacity and the potential danger of a patient with mental illness to self or others, most jurisdictions have a legal code that allows for involuntary hospitalization of the mentally ill. However, which patients qualify for involuntary hospitalizations vary by jurisdiction. Case law criteria for emergencies range from a risk of “imminent” harm to self or others to a deterioration in the patient’s mental condition if treatment is halted.72 Providers are recommended to familiarize themselves with laws regarding inpatient civil commitment in their particular jurisdiction.
When a patient is being discharged from the ED, either because they do not require a psychiatric inpatient admission or because they don’t meet criteria for involuntary commitment, there are various outpatient dispositions available. Any patient with a diagnosis of schizophrenia should have outpatient follow-up with a mental health provider upon discharge from the ED.
For patients who have a failed community treatment in the past, who do not have a significant support network, or whose disease is too severe for a regular outpatient clinic to handle would be better suited for a disposition to a transitional program over a standard outpatient appointment; such as partial hospitalization programs, intensive outpatient programs, and community mental health treatment teams (CMHT). Assertive community treatment (ACT) teams are a form of CMHT comprised of several disciplines, including nurses, occupational therapists, psychiatrists, psychologists, and social workers.73 A large prospective cohort study found homeless patients to have made gains in multiple spheres of mental health while enrolled in ACT programs, and these benefits were retained after discharge from the ACT program.74
Conclusion
Schizophrenia is characterized primarily by delusions, hallucinations, and disorganized thinking. Emergency medicine providers are frequently at the front lines of diagnosing, initiating treatment, and managing relapses for these individuals. As schizophrenia is an exclusion, the primary task of an emergency physician is to rule out all “organic”, for lack of a better term, causes of the psychosis or disordered thinking before making a definitive diagnosis of schizophrenia. As many EDs do not have sufficient psychiatry support and many patients with schizophrenia are using the ED as their location for primary mental health, many emergency physicians are finding themselves acting as the main providers for these individuals. The primary treatment for patients with schizophrenia remains monotherapy with antipsychotic medication. Although the highest-level disposition for a patient with schizophrenia remains the inpatient psychiatry department, there are many transitional dispositions that can bridge the gap between a discharge to standard community care and an inpatient admission.
Schizophrenia has a heterogeneous presentation including both positive and negative symptoms. It is a relatively common disorder, affecting 0.7% of the world’s population.1,2 Schizophrenia is defined by abnormalities in two or more of five domains:3
- Delusions,
- Hallucinations,
- Disorganized thinking/speech (eg, frequent derailment or incoherence),
- Grossly disorganized or abnormal motor behavior (including catatonia), or
- Negative symptoms (ie, diminished emotional expression or avolition).
Schizophrenia is defined as having two or more of the above domains. Each must present for a significant portion of time during a 1-month period (or less if successfully treated). At least one of these must be delusions, hallucinations, or disorganized speech. This must cause dysfunction in a major area, such as work, interpersonal relations, or self-care. Signs of the disturbance persist for at least 6 months. If mood symptoms exist, they may only be present for a minority of the total duration of symptoms. Symptoms are not attributable to the physiological effects of a substance (eg, a drug of abuse, a medication) or another medical condition.3
If symptoms are present for less than 1 month, then the diagnosis is brief psychotic disorder. If symptoms are present between 1 month and 6 months, then the diagnosis is schizophreniform disorder. If a patient has only delusions but does not hallucinate; possesses disorganized speech, grossly disorganized, or catatonic behavior; and negative symptoms, then the diagnosis is delusional disorder.
The exact pathophysiology of schizophrenia is not known at this time. However, it is theorized to be a mix of biological and environmental factors leading to dysfunction of the neurotransmitters dopamine and glutamate, as well as structural changes to the brain.2
Methods
There is an abundance of research on schizophrenia in the psychiatry literature; however, much of this deals with chronic management in patients with established diagnoses, thus this may be hard to translate into the realities of emergency practice. At the same time, although schizophrenia is discussed in the emergency medicine literature, most ED-based research is performed in patients with undifferentiated agitation and not specifically on one diagnostic subset. Therefore, a narrative review was utilized in order to focus in on studies that are relevant to emergency practice.
A narrative literature search was performed utilizing PubMed, the Cochrane database, and American Psychiatric Association Practice Alerts for the past 3 years; bibliographies of major psychiatric textbooks; and reviews and clinical policies in the National Guidelines Clearinghouse.
Epidemiology
The lifetime prevalence of schizophrenia is approximately 0.3% to 0.7%.2 In addition, schizophrenia is a frequently deadly disease as one meta-analysis found lifetime suicide to be 4.9 %, which is far higher than the average risk in the United States,4 which is approximately 0.5%. From 1992 to 2000 there has been a 15% increase in ED visits for psychiatric problems.5 Patients with schizophrenia have been found to be significantly more likely to be “high utilizers” of ED services in comparison to those with other psychiatric diagnoses.6
The increase in psychiatric presentations to EDs is coupled with a lack of psychiatrists in many areas of the United States. The American Hospital Association has reported that 40% of American hospitals have difficulty maintaining adequate psychiatric coverage to meet patient demand in the ED, forcing many emergency medicine physicians to act as a primary psychiatrist.7
Differential Diagnosis
The characteristic symptoms of schizophrenia can be present in many other illnesses; therefore, the emergency clinician must be able to distinguish schizophrenia from other illnesses presenting with a psychotic component. Furthermore, schizophrenia is a diagnosis of exclusion as the diagnosis can only be made after all medical etiologies of the symptoms have been excluded.
Items on the differential include delirium, a substance induced psychosis,8 which can be caused by both substance intoxication and substance withdrawal; as well as psychosis caused by another medical condition.8 It should be stressed that a prior diagnosis of schizophrenia does not rule out a medical etiology of a patient’s current psychotic episode; therefore, a thorough history and examination is fundamental, even in patients with a known history of psychiatric illness. Moreover, many patients with schizophrenia present with a medical chief complaint, despite being symptomatic regarding their schizophrenia. In the latter case, providers must be vigilant regarding any medical comorbidities, particularly when patients are actively psychotic.
Finally, within the psychiatric disorders there are multiple disorders other than schizophrenia that may have psychotic features, including depression with psychotic features, bipolar disorder with psychotic features, and the other schizophrenia spectrum disorders; including brief psychotic disorder, schizophreniform disorder, schizoaffective disorder, and delusional disorder.
Emergency Department Evaluation
The process of evaluation of psychiatric patients who present to the ED should be directed towards a determination as to whether hospitalization is warranted, treatment of underlying medical condition is needed, or psychiatric care is indicated. New onset of psychiatric illness will commonly call for extensive evaluation, whereas patients with chronic schizophrenia may not need testing but may need psychiatric hospitalization.9 Additionally, emergency clinicians are frequently requested to preform “medical clearance” by their psychiatrist colleagues, before a patient can be transferred to a psychiatry department or psychiatric hospital. However, this is a poor term and a better nomenclature would be “assess for medical stability.”9 Emergency clinicians are, in essence, being asked whether the patient is medically stable enough for transfer to a unit or hospital where there is little or no medical support. Prior to transfer to a psychiatric unit, a patient’s non-psychiatric medical conditions should be stable enough for outpatient management.
Interacting with Patients Suffering from Psychosis
Although providers should not pretend to interact in the patient’s reality, they should express understanding in what the patient thinks or feels and kindly direct them back to the current situation.10 It is important for providers in the acute setting to be non-judgmental about reasons for relapse (in particular non-adherence to medication or substance misuse).10 Challenging the patient, while at times useful, is best done once the patient is stabilized and in a controlled environment.
History
The history should focus on age of onset; prior history of psychiatric symptoms; history of medical illness and use or abuse of illicit substances, as well as medications; the stability of psychotic symptoms; and if hallucinations are present, whether they are auditory or visual in nature.10 Table 1 highlights the differences between a patient presenting with a psychiatric complaint and the psychotic symptoms secondary to a medical illness. Table 2 provides salient differences between the examination of a patient with psychosis due to a psychiatric illness or another medical condition. Additionally, Table 3 lists various features, which differentiates schizophrenia from other psychiatric illnesses.
Additionally, patients with schizophrenia can become just as physically ill as anyone else; however, they may have difficulty expressing their specific physical complaints. Furthermore, patients with schizophrenia have a pronounced incidence of early death compared to the general population.11 Therefore, performing a simple physical examination and paying attention to vital signs can rule out or detect a great deal of organic pathology.12Once a diagnosis of schizophrenia is made, the next priority is to determine whether the patient is a threat to themselves or others and whether this threat is imminent or not. Statements of suicidal or homicidal ideation, intent, and/or plan are the basic methods of screening for danger. However, physicians also need to screen for command auditory hallucinations, prior history of suicidal behavior, and violence. Finally, even in a patient with previously diagnosed schizophrenia, screening must also be done for substance intoxication and withdrawal.
Diagnostic Studies
The indications for diagnostic studies are influenced by whether the presentation is experiencing first break psychoses vs a presentation in a patient with known schizophrenia. In the former case there is a greater need for laboratory and other diagnostic tests. In the latter case minimal testing will likely be necessary. Patients with new onset psychiatric symptoms generally need extensive laboratory and radiographic evaluations, including a computed tomography scan of the head.9
For patients who have an established schizophrenia, extensive evidence supports that there is no need for testing and indeed more often results in false positives,11,13and selective testing based on findings in the history and physical examination is the correct strategy.11
Complicating the decision to obtain laboratory tests is the fact that many psychiatric facilities rely on referring hospitals to perform baseline testing, as they do not have the resources to perform the tests or to manage any identified abnormalities. Therefore, some tests are needed prior to transfer. In addition, at times, baseline laboratory tests are needed prior to starting antipsychotics.14
Additionally, some tests are unlikely to affect immediate disposition or treatment but may be useful to psychiatrists for long-term treatment planning.15 A common disagreement between psychiatrists and emergency providers is the urine toxicology screen or a blood alcohol level. Urine toxicology or a blood alcohol level does not usually affect initial treatment and disposition in the ED.11,16 However, it may be invalid if collected subsequent to a transfer from the ED, and can determine whether a patient requires specific substance use disorder treatment or not.16-18 Therefore, many psychiatric providers will request a urine toxicology screening or a blood alcohol level before accepting a transfer. Please note, urine toxicology screens only identify a small number of toxic ingestions. Therefore, a negative screen does not eliminate drug abuse or overdose.
Finally, in the case that a diagnostic examination is being done to satisfy a requirement for an institution to accept a patient or to assist in long-term treatment planning, disposition should not be delayed in order to wait for the result to return.11
Treatment
Unfortunately, no level I or II studies have been found that examine the indications for treatment of the psychiatric patient in the ED.9 Additionally, in the ED, there is an ill-defined difference between the use of psychotropic medications as an intervention after patient assessment and plan of care, and the use of these medications to control behavior without an assessment and treatment plan.9 Therefore, many treatment recommendations have been made based on studies in other settings.
The first line treatment for a patient with schizophrenia is monotherapy with an antipsychotic agent other than clozapine.19-21 (Clozapine is not used as a first line agent due to its high side effect burden and the testing necessary before starting.) This is supported by multiple randomized controlled trials and is the standard of care.22-24 The response rates in treatment with antipsychotics in studies specifically designed to examine treatment of first-episode schizophrenia are high, ranging from 46% to 96%.23 Therefore, the emergency medicine provider can confidently reassure patients and families that antipsychotic medications are effective in treating schizophrenia. Loading doses of antipsychotics should not be used.19
Although there are only small and inconsistent differences between different antipsychotics, other than clozapine, with respect to efficacy, there are large differences in adverse effect profiles.25-30Therefore, the choice of antipsychotic medication is generally made based on previous response to individual antipsychotic medications and relative side effects.31
If extrapyramidal symptoms, including tardive dyskinesia, are of particular concern to a patient, then second generation or low potency first generation antipsychotics should be used.25,32 If a patient complains of previous problems with sedation, then haloperidol or aripiprazole should be preferred.25 Haloperidol, aripiprazole, or amisulpride should be considered for patients who are particularly concerned about weight gain, or who may be at the greatest risk of weight gain.25
If there is no response to medication after 4 weeks,32 despite dose optimization, a change in antipsychotic should be considered.33,34 Where there is partial response, the patient should be re-assessed after 8 weeks unless there are significant adverse effects.14,19,20,34 A combination of different antipsychotic medications should not be used, except during transitional periods when patients are being switched from one antipsychotic to another, or when used for clozapine augmentation.14After an acute episode has passed, providers can consider offering depot/long-acting injectable antipsychotic medication to people with schizophrenia who would prefer such treatment; as long acting injectable antipsychotics have been shown to reduce medication non-adherence.35,36 Long-acting depot antipsychotics should not be used for acute episodes because it may take 3 to 6 months for the medications to reach a stable state.14Once a patient achieves a remission of their symptoms, patients are recommended to stay on their antipsychotic medication for at least 2 years since their last acute exacerbation.14,34 For maintenance therapy,the antipsychotic dose should be reduced gradually to the lowest possible effective dose, which should not be lower than half of the effective dose during the acute phase.37
Clozapine should be offered to people with schizophrenia whose illness has not responded adequately to treatment with adequate doses of at least two different antipsychotic drugs.38-41There is no indication to using dual antipsychotic agents on patients with schizophrenia prior to starting a clozapine trial.42,43
Non-Pharmacological Intervention
There is strong evidence that people diagnosed with schizophrenia benefit from psychosocial treatments in addition to pharmacological treatment. Multiple randomized controlled studies, as well as national guidelines, have demonstrated cognitive behavioral therapy and family therapy to be effective for the treatment of schizophrenia.44-51 Particular focuses of therapy include communication skills, problem solving, psychoeducation, and assisting with family conflict,18,19 which are feasible in an emergency setting.52
Recent studies have shown promise using a “crisis intervention” approach as an alternative option to the hospital or emergency medical services systems. Treatment usually involves a combination of medication as well as counseling (practical help with living skills and support for close family members). After the crisis has been stabilized, sufferers are carefully introduced to other models of care more suited for the chronic phases of psychiatric illnesses.53 This is particularly important as psychosocial instability, such as changes in the psychosocial environment (ie, a primary caretaker going on vacation, or a patient being distanced from their family or support environment) have been associated with relapse.19
Acute Agitation
The vast majority of patients suffering from mental illness, including schizophrenia, are not violent and only a small proportion of the violence in our society can be attributed to persons who are mentally ill.54 Furthermore, people with psychiatric disabilities are far more likely to be victims than perpetrators of violent crime.55 However, schizophrenia and related disorders are associated with substantially increased rates of violence and violent offences compared to those who did not, independent of substance use.56,57 Furthermore, the majority of acute hospital assaults occur in the ED.58
Two emergency medicine evidence-based guidelines state a benzodiazepine (lorazepam or midazolam) or a conventional antipsychotic (droperidol or haloperidol) should be used as monotherapy for the treatment of acute undifferentiated agitation in the ED.12,59 However, in cases of patients with a diagnosis or suspicion of schizophrenia, an antipsychotic should be used in order to start a “disease modifying agent” earlier in the course of treatment.20,59,60Where possible, the same antipsychotic should be used as monotherapy for treatment of both acute agitation and standing antipsychotic medication.13The choice of medication for the treatment of acute agitation should be based on patient preference, past experience of antipsychotic treatment, the adverse effect profile, and concurrent medical history.19,58 This is supported by a case controlled study, which showed patients’ subjective experiences in the acute phase of treatment affected their long-term adherence to medication.61
Haloperidol is the most commonly used medication for agitation in patients with schizophrenia.59,62 When haloperidol is used to treat acute violence, it is almost always used in combination with a benzodiazepine unless medically compromised.62,63 Intramuscular olanzapine has been shown to be effective in managing acute aggression or agitation in patients with schizophrenia, especially where it is necessary to avoid some of the older treatments. Olanzapine causes fewer movement disorders than halopridol.64 Chlorpromazine is available in both per os and intramuscular formulations. However, chlorpromazine is associated with more side effects than other antipsychotics, including a higher incidence of prolonged QT and Torsades.65 However, where choices are limited, chlorpromazine may be the only treatment available for acute agitation. If used, the close monitoring of blood pressure is indicated.65 Inhaled loxapine received US Food and Drug Administration approval for treatment of acute agitation in 2012.66 This is a useful option for an agitated patient who is calm enough to receive an inhaler, but is unable to take medication per os.
Restrictive intervention (restraints or seclusion) should only be used if de-escalation and other preventive strategies, including medication as needed, have failed and there is potential for harm to the patient or other people if no action is taken.58
Telepsychiatry Consultation
Telepsychiatry is an approach, where by EDs that do not have access to psychiatrists can see patients in the ED itself, by forming a contract with either psychiatrists or companies to allow a psychiatrist at another location to perform an examination on a patient via video connection and advise the emergency room providers on treatment and intervention. There is very little data in the peer-reviewed literature on the use of telepsychiatry. The literature is limited to case reports, program descriptions, and randomized studies for reliability.67,68 One randomized controlled trial showed that a telepsychiatry evaluation had a reliable diagnosis and disposition when compared to a face-to-face assessment.69 One case control showed reduction in time-to-consult done in the ED, length of stay, and door-to-consult stay, once a telepsychiatry program was initiated.70 A large case control study showed that telepsychiatry recipients were more likely to receive 30-day follow-up, 90-day follow-up, and total inpatient length of stay, totaling both initial admission and subsequent admissions.71 Therefore, it is reasonable for EDs to take advantage of telepsychiatry services if no immediate psychiatrist coverage is available.
Disposition
The primary disposition question regarding a patient presenting with a psychiatric chief complaint to the ED, including schizophrenia, is whether they require a psychiatric admission or not. If a patient requires hospitalization but refuses, the emergency clinician must decide if the patient meets criteria for involuntarily hospitalization. If a patient does not require an inpatient admission or would benefit from an inpatient admission but does not meet criteria for civil commitment, then the next major question is what type of outpatient treatment is sufficient for this particular patient (Figure).
For a patient with schizophrenia to require inpatient psychiatric admission he must be either a danger to himself or others or has failed outpatient treatment. In addition, the patient must currently have undergone a clinical evaluation of any suspected medical illness, and any medical problems the patient is experiencing must be sufficiently stable to allow safe transport and treatment at a psychiatric hospital unit.9 If a patient’s medical condition is not sufficiently stable for either transport or hospitalization at a psychiatric hospital but the patient still requires inpatient psychiatric treatment, the patient should be hospitalized on a standard medical ward and receive psychiatric care on that unit.
Patients with mental illness pose an additional challenge, in that their illness often time interferes with their judgment about whether they require treatment. Because of this possible lack of capacity and the potential danger of a patient with mental illness to self or others, most jurisdictions have a legal code that allows for involuntary hospitalization of the mentally ill. However, which patients qualify for involuntary hospitalizations vary by jurisdiction. Case law criteria for emergencies range from a risk of “imminent” harm to self or others to a deterioration in the patient’s mental condition if treatment is halted.72 Providers are recommended to familiarize themselves with laws regarding inpatient civil commitment in their particular jurisdiction.
When a patient is being discharged from the ED, either because they do not require a psychiatric inpatient admission or because they don’t meet criteria for involuntary commitment, there are various outpatient dispositions available. Any patient with a diagnosis of schizophrenia should have outpatient follow-up with a mental health provider upon discharge from the ED.
For patients who have a failed community treatment in the past, who do not have a significant support network, or whose disease is too severe for a regular outpatient clinic to handle would be better suited for a disposition to a transitional program over a standard outpatient appointment; such as partial hospitalization programs, intensive outpatient programs, and community mental health treatment teams (CMHT). Assertive community treatment (ACT) teams are a form of CMHT comprised of several disciplines, including nurses, occupational therapists, psychiatrists, psychologists, and social workers.73 A large prospective cohort study found homeless patients to have made gains in multiple spheres of mental health while enrolled in ACT programs, and these benefits were retained after discharge from the ACT program.74
Conclusion
Schizophrenia is characterized primarily by delusions, hallucinations, and disorganized thinking. Emergency medicine providers are frequently at the front lines of diagnosing, initiating treatment, and managing relapses for these individuals. As schizophrenia is an exclusion, the primary task of an emergency physician is to rule out all “organic”, for lack of a better term, causes of the psychosis or disordered thinking before making a definitive diagnosis of schizophrenia. As many EDs do not have sufficient psychiatry support and many patients with schizophrenia are using the ED as their location for primary mental health, many emergency physicians are finding themselves acting as the main providers for these individuals. The primary treatment for patients with schizophrenia remains monotherapy with antipsychotic medication. Although the highest-level disposition for a patient with schizophrenia remains the inpatient psychiatry department, there are many transitional dispositions that can bridge the gap between a discharge to standard community care and an inpatient admission.
1. MacDonald AW, Schulz SC: What we know: findings that every theory of schizophrenia should explain. Schizophr Bull. 2009;35(3):493-508. doi:10.1093/schbul/sbp017.
2. McGrath J, Saha S, Chant D, Welham J. Schizophrenia: a concise overview of incidence, prevalence, and mortality. Epidemiol Rev. 2008;30:67-76. doi:10.1093/epirev/mxn001.
3. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders: DSM–5. 5th ed. Washington, DC: American Psychiatric Association; 2013.
4. Palmer BA, Pankratz VS, Bostwick JM. The lifetime risk of suicide in schizophrenia: a reexamination. Arch Gen Psychiatry. 2005;62(3):247-253.
5. American College of Emergency Physicians. Psychiatric and substance abuse survey 2008. http://newsroom.acep.org/download/ACEP%2BPsychiatric%2Band%2BSubstance%2BAbuse%2BSurvey%2B-%2BApril%2B2008.pdf. Accessed October 7, 2018.
6. Pasic J, Russo J, Roy-Byrne P. High utilizers of psychiatric emergency services. Psychiatr Serv. 2005;56(6):678-684.
7. American Hospital Association. AHA annual survery database. http://www.ahadata.com/aha-annual-survey-database-asdb/. Accessed October 7, 2018.
8. Testa A, Giannuzzi R, Daini S, Bernardini L, Petrongolo L, Gentiloni Silveri N. Psychiatric emergencies (part III): psychiatric symptoms resulting from organic diseases. Eur Rev Med Pharmacol Sci. 2013;17 Suppl 1:86-99.
9. Zun LS. Evidence-based evaluation of psychiatric patient. J Emerg Med. 2005;28(3):277-283.
10. Bryan CA, Mistovich JJ, Krost WS, Limmer DD. In two minds? EMS care of the schizophrenic patient. Recognizing the signs and symptoms of schizophrenia is key to prehospital care. EMS Mag. 2009;38(12):63-71.
11. Lukens TW, Wolf SJ, Edlow JA, et al; American College of Emergency Physicians Clinical Policies Subcommittee (Writing Committee) on Critical Issues in the Diagnosis and Management of the Adult Psychiatric Patient in the Emergency Department. Clinical policy: critical issues in the diagnosis and management of the adult psychiatric patient in the emergency department. Ann Emerg Med. 2006;47(1):79-99.
12. Crump C, Winkleby MA, Sundquist K, Sundquist J. Comorbidities and mortality in persons with schizophrenia: a Swedish national cohort study. Am J Psychiatry. 2013;170(3):324-33. doi:10.1176/appi.ajp.2012.12050599.
13. Kroll DS, Smallwood J, Chang G. Drug screens for psychiatric patients in the emergency department: evaluation and recommendations. Psychosomatics. 2013;54(1):60-66. doi:10.1016/j.psym.2012.08.007.
14. Verma S, Chan LL, Chee KS, et al; MOH Clinical Practice Guidelines Workgroup on Schizophrenia. Ministry of Health clinical practice guidelines: schizophrenia. Singapore Med J. 2011;52(7):521-525.
15. Fochtman LJ. Psychiatric perspectives on medical screening of psychiatric patients. Acad Emerg Med. 2002;9(9):963-964.
16. Tijdink JK, van den Heuvel J, Vasbinder EC, can de Ven PM, Honig A. Does on-site urine toxicology screening have an added diagnostic value in psychiatric referrals in an emergency setting? Gen Hosp Psychiatry. 2011;33(6):626-6
17. Janiak BD, Atteberry S. Medical clearance of the psychiatric patient in the emergency department. J Emerg Med. 2012;43(5):866-870. doi:10.1016/j.jemermed.2009.10.026.
18. Lehman AF, Lieberman JA, Dixon LB, et al; American Psychiatric Association; Steering Committee on Practice Guidelines. Practice guideline for the treatment of patients with schizophrenia, second edition. Am J Psychiatry. 2004;161(2 Suppl):1-56.
19. National Institute for Health and Care Excellence. Psychosis and schizophrenia in adults: prevention and management. https://www.nice.org.uk/guidance/cg178. Published February 2014. Accessed September 25, 2018.
20. Scottish Intercollegiate Guidelines Network. SIGN 131: Management of schizophrenia. https://www.sign.ac.uk/assets/sign131.pdf. Published March 2013. Accessed September 25, 2018. 15.
21. Tucci V, Siever K, Matorin A, Moukaddam N. Down the rabbit hole: emergency department medical clearance of patients with psychiatric or behavioral emergencies. Emerg Med Clin North Am. 2015;33(4):721-737. doi:10.1016/j.emc.2015.07.002.
22. Tandon R, Belmaker RH, Gattaz WF, et al; Section of Pharmacopsychiatry, World Psychiatric Association. World Psychiatric Association Pharmacopsychiatry Section statement on comparative effectiveness of antipsychotics in the treatment of schizophrenia. Schizophr Res. 2008;100(1-3):20-38. doi:10.1016/j.schres.2007.11.033.
23. Leucht S, Arbter D, Engel RR, Kissling W, Davis JM. How effective are second-generation antipsychotic drugs? A meta-analysis of placebo-controlled trials. Mol Psychiatry. 2009;14(4):429-447. doi:10.1038/sj.mp.4002136.
24. Robinson DG, Woerner MG, Delman HM, Kane JM. Pharmacological treatments for first-episode schizophrenia. Schizophr Bull. 2005;31(3):705-722.
25. Leucht S, Corves C, Arbter D, Engel RR, Li C, Davis JM. Second-generation versus first-generation antipsychotic drugs for schizophrenia: a meta-analysis. Lancet. 2009;373(9657):31-41. doi:10.1016/S0140-6736(08)61764-X.
26. Komossa K, Rummel-Kluge C, Hunger H, et al. Olanzapine versus other atypical antipsychotics for schizophrenia. Cochrane Database Syst Rev. 2010;3:CD006654. doi:10.1002/14651858.CD006654.pub2.
27. Komossa K, Rummel-Kluge C, Hunger H, et al. Amisulpride versus other atypical antipsychotics for schizophrenia. Cochrane Database Syst Rev. 2010;1:CD006624. doi:10.1002/14651858.CD006624.pub2.
28. Komossa K, Rummel-Kluge C, Hunger H, et al. Ziprasidone versus other atypical antipsychotics for schizophrenia. Cochrane Database Syst Rev. 2009;4:CD006627. doi:10.1002/14651858.CD006627.pub2.
29. Komossa K, Rummel-Kluge C, Schmid F, et al. Aripiprazole versus other atypical antipsychotics for schizophrenia. Cochrane Database Syst Rev. 2009;4:CD006569. doi:10.1002/14651858.CD006569.pub3.
30. Komossa K, Rummel-Kluge C, Schmid F, et al. Quetiapine versus other atypical antipsychotics for schizophrenia. Cochrane Database Syst Rev. 2010;1:CD006625. doi:10.1002/14651858.CD006625.pub2.
31. Crossley NA, Constante M, McGuire P, Power P. Efficacy of atypical v. typical antipsychotics in the treatment of early psychosis: meta-analysis. Br J Psychiatry. 2010;196(6):434-439. doi:10.1192/bjp.bp.109.066217.
32. Correll CU, Schenk EM. Tardive dyskinesia and new antipsychotics. Curr Opin Psychiatry. 2008;21(2):151-156. doi:10.1097/YCO.0b013e3282f53132.

33. Emsley R, Rabinowitz J, Medori R. Time course for antipsychotic treatment response in first-episode schizophrenia. Am J Psychiatry. 2006;163(4):743-745.
34. Barnes TR; Schizophrenia Consensus Group of British Association for Psychopharmacology. Evidence-based guidelines for the pharmacological treatment of schizophrenia: recommendations from the British Association for Psychopharmacology. J Psychopharmacol. 2011;25(5):567-620. doi:10.1177/0269881110391123.
35. MacEwan JP, Kamat SA, Duffy RA, et al. Hospital readmission rates among patients with schizophrenia treated with long-acting injectables or oral antipsychotics. Psychiatr Serv. 2016;67(11):1183-1188.
36. Patel MX, Taylor M, David AS. Antipsychotic long-acting injections: mind the gap. Br J Psychiatry. 2009;52:S1-S4. doi:10.1192/bjp.195.52.s1.
37. Uchida H, Suzuki T, Takeuchi H, Arenovich T, Mamo DC. Low dose vs standard dose of antipsychotics for relapse prevention in schizophrenia: meta-analysis. Schizophr Bull. 2011;37(4):788-799. doi:10.1093/schbul/sbp149.
38. Swartz MS, Stroup TS, McEvoy JP, et al. What CATIE found: results from the schizophrenia trial. Psychiatr Serv. 2008;59(5):500-506. doi:10.1176/ps.2008.59.5.500.
39. Jones PB, Barnes TR, Davies L, et al. Randomized controlled trial of the effect on Quality of Life of second- vs first-generation antipsychotic drugs in schizophrenia: Cost Utility of the Latest Antipsychotic Drugs in Schizophrenia Study (CUtLASS 1). Arch Gen Psychiatry. 2006;63(10):1079-1087.
40. Chakos M, Lieberman J, Hoffman E, Bradford D, Sheitman B. Effectiveness of second-generation antipsychotics in patients with treatment-resistant schizophrenia: a review and meta-analysis of randomized trials. Am J Psychiatry. 2001;158(4):518-526.
41. McEvoy JP, Lieberman JA, Stroup TS, et al; CATIE Investigators. Effectiveness of clozapine versus olanzapine, quetiapine, and risperidone in patients with chronic schizophrenia who did not respond to prior atypical antipsychotic treatment. Am J Psychiatry. 2006;163(4):600-610.
43. Stahl SM, Grady MM. A critical review of atypical antipsychotic utilization: comparing monotherapy with polypharmacy and augmentation. Curr Med Chem. 2004;11(3):313-327.
44. Turkington D, Sensky T, Scott J, et al. A randomized controlled trial of cognitive-behavior therapy for persistent symptoms in schizophrenia: a five-year follow-up. Schizophr Res. 2008;98(1-3):1-7.
45. Malik N, Kingdon D, Pelton J, Mehta R, Turkington D. Effectiveness of brief cognitive-behavioral therapy for schizophrenia delivered by mental health nurses: relapse and recovery at 24 months. J Clin Psychiatry. 2009;70(2):201-207.
46. Haddock G, Barrowclough C, Shaw JJ, Dunn G, Novaco RW, Tarrier N. Cognitive-behavioural therapy v. social activity therapy for people with psychosis and a history of violence: randomised controlled trial. Br J Psychiatry. 2009;194(2):152-157. doi:10.1192/bjp.bp.107.039859.
47. Lysaker PH, Davis LW, Bryson GJ, Bell MD. Effects of cognitive behavioral therapy on work outcomes in vocational rehabilitation for participants with schizophrenia spectrum disorders. Schizophr Res. 2009;107(2-3):186-191. doi:10.1016/j.schres.2008.10.018.
48. Grant PM, Huh GA, Perivoliotis D, Stolar NM, Beck AT. Randomized trial to evaluate the efficacy of cognitive therapy for low-functioning patients with schizophrenia. Arch Gen Psychiatry. 2012;69(2):121-127. doi:10.1001/archgenpsychiatry.2011.129.
49. Girón M, Fernández-Yañez A, Mañá-Alvarenga S, Molina-Habas A, Nolasco A, Gómez-Beneyto M. Efficacy and effectiveness of individual family intervention on social and clinical functioning and family burden in severe schizophrenia: a 2-year randomized controlled study. Psychol Med. 2010;40(1):73-84. doi:10.1017/S0033291709006126.
50. Bressi C, Manenti S, Frongia P, Porcellana M, Invernizzi G. Systemic family therapy in schizophrenia: a randomized clinical trial of effectiveness. Psychother Psychosom. 2008;77(1):43-49.
51. Chien WT, Lee IY. The schizophrenia care management program for family caregivers of Chinese patients with schizophrenia. Psychiatr Serv. 2010;61(3):317-320. doi:10.1176/ps.2010.61.3.317.
52. Sneed J, Balestri M, Belfi BJ. The use of dialectical behavior therapy strategies in the psychiatric emergency room. Psychotherapy Theory, Research & Practice. 2003;40(4):265-277.
53. Murphy SM, Irving CB, Adams CE, Wagar M. Crisis intervention for people with severe mental illnesses. Cochrane Database Syst Rev. 2015;12:CD001087. doi:10.1002/14651858.CD001087.pub5.
54. Mulvey EP. Assessing the evidence of a link between mental illness and violence. Hosp Community Psychiatry. 1994;45(7):663-668.
55. Hiroeh U, Appleby L, Mortensen PB, Dunn G. Death by homicide, suicide, and other unnatural causes in people with mental illness: a population-based study. Lancet. 2001;358(9299):2110-2112.
56. Van Dorn R, Volavka J, Johnson N. Mental disorder and violence: is there a relationship beyond substance use? Soc Psychiatry Psychiatr Epidemiol. 2012;47(3):487-503. doi:10.1007/s00127-011-0356-x.
57. Fazel S, Wolf A, Palm C, Lichtenstein P. Violent crime, suicide, and premature mortality in patients with schizophrenia and related disorders: a 38-year total population study in Sweden. Lancet Psychiatry. 2014;1(1):44-54.
58. National Institute for Health and Care Excellence. Violence and aggression: short-term management in mental health, health and community settings. https://www.nice.org.uk/guidance/ng10/chapter/About-this-guideline. Published May 2015. Accessed September 24, 2018.
59. Wilson MP, Pepper D, Currier GW, Holloman GH Jr, Feifel D. The psychopharmacology of agitation: consensus statement of the american association for emergency psychiatry project Beta psychopharmacology workgroup. West J Emerg Med. 2012;13(1):26-34. doi:10.5811/westjem.2011.9.6866.
63. Leucht S, Heres S, Kissling W, Davis JM. Evidence based pharmacotherapy of schizophrenia. Int J Neuropsychopharmacol. 2011;14(2):269-284. doi:10.1017/S1461145710001380.
64. Belgamwar RB, Fenton M. Olanzapine IM or velotab for acutely disturbed/agitated people with suspected serious mental illnesses. Cochrane Database Syst Rev. 2005;2:CD003729.

65. Ahmed U, Jones H, Adams CE. Chlorpromazine for psychosis induced aggression or agitation. Cochrane Database Syst Rev. 2010;4:CD007445. doi:10.1002/14651858.CD007445.pub2.
66. US Food and Drug Administration. Drug approval package. https://www.accessdata.fda.gov/drugsatfda_docs/nda/2012/022549_adasuve_toc.cfm. Accessed September 28, 2018.
67. Salmoiraghi A, Hussain S. A systematic review of the use of telepsychiatry in acute settings. J Psychiatr Pract. 2015;21(5):389-393. doi:10.1097/PRA.0000000000000103.
68. Shore JH, Hilty DM, Yellowlees P. Emergency management guidelines for telepsychiatry. Gen Hosp Psychiatry. 2007;29(3):199-206.
69. Seidel RW, Kilqus MD. Agreement between telepsychiatry assessment and face-to-face assessment for emergency department psychiatry patients. J Telemed Telecare. 2014;20(2):59-62. doi:10.1177/1357633X13519902.
70. DeVido J, Glezer A, Branagan L, Lau A, Bourgeois JA. Telepsychiatry for inpatient consultations at a separate campus of an academic medical center. Telemed J E Health. 2016;22(7):572-576. doi:10.1089/tmj.2015.0125.
71. Narasimhan M, Druss BG, Hockenberry JM, et al. Impact of a telepsychiatry program at emergency departments statewide on the quality, utilization, and costs of mental health services. Psychiatr Serv. 2015;66(11):1167-1172. doi:10.1176/appi.ps.201400122.
72. Simon RI, Shuman DW. Clinical issues in psychiatry and the law. In: Hales RE, Yudofsky SC, Roberts LW, eds. The American Psychiatric Publishing Textbook of Psychiatry. 6th ed. Washington, DC: American Psychiatric Publishing; 2014:175-204.
73. Malone D, Newron-Howes G, Simmonds S, Marriot S, Tyrer P. Community mental health teams (CMHTs) for people with severe mental illnesses and disordered personality. Cochrane Database Syst Rev. 2007;3:CD000270.
74. Rosenheck RA, Dennis D. Time-limited assertive community treatment for homeless persons with severe mental illness. Arch Gen Psychiatry. 2001;58(11):1073-1080.
75. Testa A, Giannuzzi R, Sollazzo F, Petrongolo L, Bernardini L, Daini S. Psychiatric emergencies (part I): psychiatric disorders causing organic symptoms. Eur Rev Med Pharmacol Sci. 2013;17 Suppl 1:55-64.
1. MacDonald AW, Schulz SC: What we know: findings that every theory of schizophrenia should explain. Schizophr Bull. 2009;35(3):493-508. doi:10.1093/schbul/sbp017.
2. McGrath J, Saha S, Chant D, Welham J. Schizophrenia: a concise overview of incidence, prevalence, and mortality. Epidemiol Rev. 2008;30:67-76. doi:10.1093/epirev/mxn001.
3. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders: DSM–5. 5th ed. Washington, DC: American Psychiatric Association; 2013.
4. Palmer BA, Pankratz VS, Bostwick JM. The lifetime risk of suicide in schizophrenia: a reexamination. Arch Gen Psychiatry. 2005;62(3):247-253.
5. American College of Emergency Physicians. Psychiatric and substance abuse survey 2008. http://newsroom.acep.org/download/ACEP%2BPsychiatric%2Band%2BSubstance%2BAbuse%2BSurvey%2B-%2BApril%2B2008.pdf. Accessed October 7, 2018.
6. Pasic J, Russo J, Roy-Byrne P. High utilizers of psychiatric emergency services. Psychiatr Serv. 2005;56(6):678-684.
7. American Hospital Association. AHA annual survery database. http://www.ahadata.com/aha-annual-survey-database-asdb/. Accessed October 7, 2018.
8. Testa A, Giannuzzi R, Daini S, Bernardini L, Petrongolo L, Gentiloni Silveri N. Psychiatric emergencies (part III): psychiatric symptoms resulting from organic diseases. Eur Rev Med Pharmacol Sci. 2013;17 Suppl 1:86-99.
9. Zun LS. Evidence-based evaluation of psychiatric patient. J Emerg Med. 2005;28(3):277-283.
10. Bryan CA, Mistovich JJ, Krost WS, Limmer DD. In two minds? EMS care of the schizophrenic patient. Recognizing the signs and symptoms of schizophrenia is key to prehospital care. EMS Mag. 2009;38(12):63-71.
11. Lukens TW, Wolf SJ, Edlow JA, et al; American College of Emergency Physicians Clinical Policies Subcommittee (Writing Committee) on Critical Issues in the Diagnosis and Management of the Adult Psychiatric Patient in the Emergency Department. Clinical policy: critical issues in the diagnosis and management of the adult psychiatric patient in the emergency department. Ann Emerg Med. 2006;47(1):79-99.
12. Crump C, Winkleby MA, Sundquist K, Sundquist J. Comorbidities and mortality in persons with schizophrenia: a Swedish national cohort study. Am J Psychiatry. 2013;170(3):324-33. doi:10.1176/appi.ajp.2012.12050599.
13. Kroll DS, Smallwood J, Chang G. Drug screens for psychiatric patients in the emergency department: evaluation and recommendations. Psychosomatics. 2013;54(1):60-66. doi:10.1016/j.psym.2012.08.007.
14. Verma S, Chan LL, Chee KS, et al; MOH Clinical Practice Guidelines Workgroup on Schizophrenia. Ministry of Health clinical practice guidelines: schizophrenia. Singapore Med J. 2011;52(7):521-525.
15. Fochtman LJ. Psychiatric perspectives on medical screening of psychiatric patients. Acad Emerg Med. 2002;9(9):963-964.
16. Tijdink JK, van den Heuvel J, Vasbinder EC, can de Ven PM, Honig A. Does on-site urine toxicology screening have an added diagnostic value in psychiatric referrals in an emergency setting? Gen Hosp Psychiatry. 2011;33(6):626-6
17. Janiak BD, Atteberry S. Medical clearance of the psychiatric patient in the emergency department. J Emerg Med. 2012;43(5):866-870. doi:10.1016/j.jemermed.2009.10.026.
18. Lehman AF, Lieberman JA, Dixon LB, et al; American Psychiatric Association; Steering Committee on Practice Guidelines. Practice guideline for the treatment of patients with schizophrenia, second edition. Am J Psychiatry. 2004;161(2 Suppl):1-56.
19. National Institute for Health and Care Excellence. Psychosis and schizophrenia in adults: prevention and management. https://www.nice.org.uk/guidance/cg178. Published February 2014. Accessed September 25, 2018.
20. Scottish Intercollegiate Guidelines Network. SIGN 131: Management of schizophrenia. https://www.sign.ac.uk/assets/sign131.pdf. Published March 2013. Accessed September 25, 2018. 15.
21. Tucci V, Siever K, Matorin A, Moukaddam N. Down the rabbit hole: emergency department medical clearance of patients with psychiatric or behavioral emergencies. Emerg Med Clin North Am. 2015;33(4):721-737. doi:10.1016/j.emc.2015.07.002.
22. Tandon R, Belmaker RH, Gattaz WF, et al; Section of Pharmacopsychiatry, World Psychiatric Association. World Psychiatric Association Pharmacopsychiatry Section statement on comparative effectiveness of antipsychotics in the treatment of schizophrenia. Schizophr Res. 2008;100(1-3):20-38. doi:10.1016/j.schres.2007.11.033.
23. Leucht S, Arbter D, Engel RR, Kissling W, Davis JM. How effective are second-generation antipsychotic drugs? A meta-analysis of placebo-controlled trials. Mol Psychiatry. 2009;14(4):429-447. doi:10.1038/sj.mp.4002136.
24. Robinson DG, Woerner MG, Delman HM, Kane JM. Pharmacological treatments for first-episode schizophrenia. Schizophr Bull. 2005;31(3):705-722.
25. Leucht S, Corves C, Arbter D, Engel RR, Li C, Davis JM. Second-generation versus first-generation antipsychotic drugs for schizophrenia: a meta-analysis. Lancet. 2009;373(9657):31-41. doi:10.1016/S0140-6736(08)61764-X.
26. Komossa K, Rummel-Kluge C, Hunger H, et al. Olanzapine versus other atypical antipsychotics for schizophrenia. Cochrane Database Syst Rev. 2010;3:CD006654. doi:10.1002/14651858.CD006654.pub2.
27. Komossa K, Rummel-Kluge C, Hunger H, et al. Amisulpride versus other atypical antipsychotics for schizophrenia. Cochrane Database Syst Rev. 2010;1:CD006624. doi:10.1002/14651858.CD006624.pub2.
28. Komossa K, Rummel-Kluge C, Hunger H, et al. Ziprasidone versus other atypical antipsychotics for schizophrenia. Cochrane Database Syst Rev. 2009;4:CD006627. doi:10.1002/14651858.CD006627.pub2.
29. Komossa K, Rummel-Kluge C, Schmid F, et al. Aripiprazole versus other atypical antipsychotics for schizophrenia. Cochrane Database Syst Rev. 2009;4:CD006569. doi:10.1002/14651858.CD006569.pub3.
30. Komossa K, Rummel-Kluge C, Schmid F, et al. Quetiapine versus other atypical antipsychotics for schizophrenia. Cochrane Database Syst Rev. 2010;1:CD006625. doi:10.1002/14651858.CD006625.pub2.
31. Crossley NA, Constante M, McGuire P, Power P. Efficacy of atypical v. typical antipsychotics in the treatment of early psychosis: meta-analysis. Br J Psychiatry. 2010;196(6):434-439. doi:10.1192/bjp.bp.109.066217.
32. Correll CU, Schenk EM. Tardive dyskinesia and new antipsychotics. Curr Opin Psychiatry. 2008;21(2):151-156. doi:10.1097/YCO.0b013e3282f53132.

33. Emsley R, Rabinowitz J, Medori R. Time course for antipsychotic treatment response in first-episode schizophrenia. Am J Psychiatry. 2006;163(4):743-745.
34. Barnes TR; Schizophrenia Consensus Group of British Association for Psychopharmacology. Evidence-based guidelines for the pharmacological treatment of schizophrenia: recommendations from the British Association for Psychopharmacology. J Psychopharmacol. 2011;25(5):567-620. doi:10.1177/0269881110391123.
35. MacEwan JP, Kamat SA, Duffy RA, et al. Hospital readmission rates among patients with schizophrenia treated with long-acting injectables or oral antipsychotics. Psychiatr Serv. 2016;67(11):1183-1188.
36. Patel MX, Taylor M, David AS. Antipsychotic long-acting injections: mind the gap. Br J Psychiatry. 2009;52:S1-S4. doi:10.1192/bjp.195.52.s1.
37. Uchida H, Suzuki T, Takeuchi H, Arenovich T, Mamo DC. Low dose vs standard dose of antipsychotics for relapse prevention in schizophrenia: meta-analysis. Schizophr Bull. 2011;37(4):788-799. doi:10.1093/schbul/sbp149.
38. Swartz MS, Stroup TS, McEvoy JP, et al. What CATIE found: results from the schizophrenia trial. Psychiatr Serv. 2008;59(5):500-506. doi:10.1176/ps.2008.59.5.500.
39. Jones PB, Barnes TR, Davies L, et al. Randomized controlled trial of the effect on Quality of Life of second- vs first-generation antipsychotic drugs in schizophrenia: Cost Utility of the Latest Antipsychotic Drugs in Schizophrenia Study (CUtLASS 1). Arch Gen Psychiatry. 2006;63(10):1079-1087.
40. Chakos M, Lieberman J, Hoffman E, Bradford D, Sheitman B. Effectiveness of second-generation antipsychotics in patients with treatment-resistant schizophrenia: a review and meta-analysis of randomized trials. Am J Psychiatry. 2001;158(4):518-526.
41. McEvoy JP, Lieberman JA, Stroup TS, et al; CATIE Investigators. Effectiveness of clozapine versus olanzapine, quetiapine, and risperidone in patients with chronic schizophrenia who did not respond to prior atypical antipsychotic treatment. Am J Psychiatry. 2006;163(4):600-610.
43. Stahl SM, Grady MM. A critical review of atypical antipsychotic utilization: comparing monotherapy with polypharmacy and augmentation. Curr Med Chem. 2004;11(3):313-327.
44. Turkington D, Sensky T, Scott J, et al. A randomized controlled trial of cognitive-behavior therapy for persistent symptoms in schizophrenia: a five-year follow-up. Schizophr Res. 2008;98(1-3):1-7.
45. Malik N, Kingdon D, Pelton J, Mehta R, Turkington D. Effectiveness of brief cognitive-behavioral therapy for schizophrenia delivered by mental health nurses: relapse and recovery at 24 months. J Clin Psychiatry. 2009;70(2):201-207.
46. Haddock G, Barrowclough C, Shaw JJ, Dunn G, Novaco RW, Tarrier N. Cognitive-behavioural therapy v. social activity therapy for people with psychosis and a history of violence: randomised controlled trial. Br J Psychiatry. 2009;194(2):152-157. doi:10.1192/bjp.bp.107.039859.
47. Lysaker PH, Davis LW, Bryson GJ, Bell MD. Effects of cognitive behavioral therapy on work outcomes in vocational rehabilitation for participants with schizophrenia spectrum disorders. Schizophr Res. 2009;107(2-3):186-191. doi:10.1016/j.schres.2008.10.018.
48. Grant PM, Huh GA, Perivoliotis D, Stolar NM, Beck AT. Randomized trial to evaluate the efficacy of cognitive therapy for low-functioning patients with schizophrenia. Arch Gen Psychiatry. 2012;69(2):121-127. doi:10.1001/archgenpsychiatry.2011.129.
49. Girón M, Fernández-Yañez A, Mañá-Alvarenga S, Molina-Habas A, Nolasco A, Gómez-Beneyto M. Efficacy and effectiveness of individual family intervention on social and clinical functioning and family burden in severe schizophrenia: a 2-year randomized controlled study. Psychol Med. 2010;40(1):73-84. doi:10.1017/S0033291709006126.
50. Bressi C, Manenti S, Frongia P, Porcellana M, Invernizzi G. Systemic family therapy in schizophrenia: a randomized clinical trial of effectiveness. Psychother Psychosom. 2008;77(1):43-49.
51. Chien WT, Lee IY. The schizophrenia care management program for family caregivers of Chinese patients with schizophrenia. Psychiatr Serv. 2010;61(3):317-320. doi:10.1176/ps.2010.61.3.317.
52. Sneed J, Balestri M, Belfi BJ. The use of dialectical behavior therapy strategies in the psychiatric emergency room. Psychotherapy Theory, Research & Practice. 2003;40(4):265-277.
53. Murphy SM, Irving CB, Adams CE, Wagar M. Crisis intervention for people with severe mental illnesses. Cochrane Database Syst Rev. 2015;12:CD001087. doi:10.1002/14651858.CD001087.pub5.
54. Mulvey EP. Assessing the evidence of a link between mental illness and violence. Hosp Community Psychiatry. 1994;45(7):663-668.
55. Hiroeh U, Appleby L, Mortensen PB, Dunn G. Death by homicide, suicide, and other unnatural causes in people with mental illness: a population-based study. Lancet. 2001;358(9299):2110-2112.
56. Van Dorn R, Volavka J, Johnson N. Mental disorder and violence: is there a relationship beyond substance use? Soc Psychiatry Psychiatr Epidemiol. 2012;47(3):487-503. doi:10.1007/s00127-011-0356-x.
57. Fazel S, Wolf A, Palm C, Lichtenstein P. Violent crime, suicide, and premature mortality in patients with schizophrenia and related disorders: a 38-year total population study in Sweden. Lancet Psychiatry. 2014;1(1):44-54.
58. National Institute for Health and Care Excellence. Violence and aggression: short-term management in mental health, health and community settings. https://www.nice.org.uk/guidance/ng10/chapter/About-this-guideline. Published May 2015. Accessed September 24, 2018.
59. Wilson MP, Pepper D, Currier GW, Holloman GH Jr, Feifel D. The psychopharmacology of agitation: consensus statement of the american association for emergency psychiatry project Beta psychopharmacology workgroup. West J Emerg Med. 2012;13(1):26-34. doi:10.5811/westjem.2011.9.6866.
63. Leucht S, Heres S, Kissling W, Davis JM. Evidence based pharmacotherapy of schizophrenia. Int J Neuropsychopharmacol. 2011;14(2):269-284. doi:10.1017/S1461145710001380.
64. Belgamwar RB, Fenton M. Olanzapine IM or velotab for acutely disturbed/agitated people with suspected serious mental illnesses. Cochrane Database Syst Rev. 2005;2:CD003729.

65. Ahmed U, Jones H, Adams CE. Chlorpromazine for psychosis induced aggression or agitation. Cochrane Database Syst Rev. 2010;4:CD007445. doi:10.1002/14651858.CD007445.pub2.
66. US Food and Drug Administration. Drug approval package. https://www.accessdata.fda.gov/drugsatfda_docs/nda/2012/022549_adasuve_toc.cfm. Accessed September 28, 2018.
67. Salmoiraghi A, Hussain S. A systematic review of the use of telepsychiatry in acute settings. J Psychiatr Pract. 2015;21(5):389-393. doi:10.1097/PRA.0000000000000103.
68. Shore JH, Hilty DM, Yellowlees P. Emergency management guidelines for telepsychiatry. Gen Hosp Psychiatry. 2007;29(3):199-206.
69. Seidel RW, Kilqus MD. Agreement between telepsychiatry assessment and face-to-face assessment for emergency department psychiatry patients. J Telemed Telecare. 2014;20(2):59-62. doi:10.1177/1357633X13519902.
70. DeVido J, Glezer A, Branagan L, Lau A, Bourgeois JA. Telepsychiatry for inpatient consultations at a separate campus of an academic medical center. Telemed J E Health. 2016;22(7):572-576. doi:10.1089/tmj.2015.0125.
71. Narasimhan M, Druss BG, Hockenberry JM, et al. Impact of a telepsychiatry program at emergency departments statewide on the quality, utilization, and costs of mental health services. Psychiatr Serv. 2015;66(11):1167-1172. doi:10.1176/appi.ps.201400122.
72. Simon RI, Shuman DW. Clinical issues in psychiatry and the law. In: Hales RE, Yudofsky SC, Roberts LW, eds. The American Psychiatric Publishing Textbook of Psychiatry. 6th ed. Washington, DC: American Psychiatric Publishing; 2014:175-204.
73. Malone D, Newron-Howes G, Simmonds S, Marriot S, Tyrer P. Community mental health teams (CMHTs) for people with severe mental illnesses and disordered personality. Cochrane Database Syst Rev. 2007;3:CD000270.
74. Rosenheck RA, Dennis D. Time-limited assertive community treatment for homeless persons with severe mental illness. Arch Gen Psychiatry. 2001;58(11):1073-1080.
75. Testa A, Giannuzzi R, Sollazzo F, Petrongolo L, Bernardini L, Daini S. Psychiatric emergencies (part I): psychiatric disorders causing organic symptoms. Eur Rev Med Pharmacol Sci. 2013;17 Suppl 1:55-64.
The Three H’s: Head, Heart, and Heat Considerations in Soccer
ABSTRACT
Soccer requires significant physical conditioning and endurance, as well as the physicality required for contact play. In order to keep athletes safe, it is important that coaches, medical staff, and the players themselves are educated on the most common dangers to their health that they may encounter on a soccer pitch. This article aims to review the current literature and recommendations on concussion, cardiovascular considerations, and heat-related illness as they relate to competitive soccer, with a goal of educating all those who help to keep athletes healthy and competing to their full potential.
Continue to: Soccer is one of the most popular sports...
Soccer is one of the most popular sports in the modern world and requires significant physical conditioning and endurance, as well as the physicality required for contact play. This article covers the topics of concussion, cardiovascular considerations, and heat-related illness as they relate to competitive soccer players. We provide a review of the prevention, recognition, and management required to keep athletes safe on the soccer pitch, both in practice and in competitive play.
HEAD
With an estimated 1.6 to 3.8 million sports-related mild traumatic brain injuries (ie, concussions) occurring annually in the United States,1 there has been an appropriate increase in the focus on prevention and treatment of these injuries. For more than a decade the spotlight has been on concussions that occur in American football, but other sports have also had to examine the prevalence of concussions in their sport. This is certainly true for soccer.
There has been a steady increase in soccer participation in the United States. From 1973 to 2014 there was a 4-fold increase in high school boys and a 35-fold increase in high school girls playing soccer.2 Currently, there are more than 3.7 million youth who play on teams under the supervision of the US Soccer Federation, the sport’s national governing body.3 With the growth of the sport, there has also been an intensified focus on injury prevention in soccer players, including concussive brain injuries.
A recent study examined injury rates in high school soccer players and noted that concussion is the second most common injury (17.9%), after ligament sprains (29.7%).4 The overall injury rate was 2.06 per 1000 athletic exposures (AEs [defined as participation in practice or game play]), but higher in games (4.42 per 1000 AEs) than during practices (1.05 per 1000 AEs). The overall concussion rate was 0.36 per 1000 AEs.4
Most concussions (54.8%) resulted in missing play between 1 to 3 weeks, but a sizeable portion of the athletes (14.9%) were out of play for more than 3 weeks. Additionally, 10.7% of all medical disqualifications were due to concussive injuries. Khodaee and colleagues4 found no statistically significant difference in concussion rates between male and female soccer players over the 9-year period of time that they examined, however previous studies have found higher rates in female athletes.5
In soccer, as in other sports, there is a concern about both the adequate recognition of concussions during practice and play and the underreporting of concussions by athletes. The US Soccer Federation has taken a proactive stance on addressing concussion in youth soccer by developing the “Recognize to Recover” program.6 Recognize to Recover is the US Soccer Federation’s “comprehensive player health and safety program aimed at promoting safe play and reducing injuries in soccer players of all ages.” The website provides an educational video geared toward players, along with links to concussion assessment tools, the US Soccer Federation Concussion Protocol, and US Soccer Federation-Centers for Disease Control fact sheets for athletes, parents, and coaches.6
Continue to: A challenge for all sports...
A challenge for all sports is allowing adequate evaluation of a suspected concussion by properly trained healthcare professionals. The 2017 Berlin Concussion in Sport Group position paper stated, “when a concussion is suspected, the athlete should be removed from the sporting environment and a multimodal assessment should be conducted in standardized fashion (eg, Sport Concussion Assessment Tool- 5th edition). Sporting bodies should allow adequate time to conduct this evaluation.”7 However, the International Federation of Football Association (FIFA) rules limit substitutions to 3 over the course of the game, which can make a thorough evaluation of players difficult as trainers and coaches are under increased pressure to quickly determine whether to use one of their valuable substitutions. Fortunately, the National Collegiate Athletic Association (NCAA) soccer has mitigated this issue by allowing unlimited substitutions during matches, and high school teams generally follow similar rules.
One of the goals of any safety education program is not only to raise the awareness of the signs and symptoms of concussion by all those involved in the sport, but also to increase the number of athletes who self-report their symptoms and decrease those who hide any possible concussions. A study found that a majority (58.6%) of middle school soccer players continued to play while experiencing concussion symptoms.5 However, in a very recent (not yet published) study, 92% of US Soccer Federation players reported that they did seek out a medical evaluation for their concussion.8 This is certainly a positive sign and further research needs to clarify what methods of education or training will maintain this level of self-reporting in soccer players.
There has also been an increased focus on understanding the mechanism of injury of concussions. In soccer, concussions can occur from player-to-player contact, contact with the player surface, contact with playing apparatus (eg, goal posts) and non-contact mechanisms. While there has been a focus on concussions from heading the ball, player-to-player contact is the most common cause of concussions. A 2017 study of 7- to 12-year-old soccer players in 4 European countries found that about 1 out of 10 concussions were caused by heading the ball.9 Comstock and colleageues10 found slightly higher numbers, roughly 25% to 30% (depending on gender), but 70% to 78% (again depending on gender) of those were caused by player-to-player contact rather than contact with the ball.
To date there has not been any meta-analytic review evaluating the cognitive and physical symptoms associated with heading in soccer. A recent review paper stated that the “current evidence seems insufficient to support a ban of heading in children’s football (soccer).”10 However, in December 2015 the US Soccer Federation included age-specific heading limitations. Players ages 10 years and under “shall not engage in heading, either in practices or in games” and players age 11 years and 12 years should have “limited heading in practice; maximum of 30 minutes of heading training per week, with no more than 15-20 headers per player, per week.”11 US Soccer Federation officials acknowledged the limitations in the current science regarding heading in young soccer players but chose to err on the side of caution until further empirical evidence regarding the risks associated with repetitive heading is available.
The US Soccer Federation is also exploring other ways to reduce the incidence rate of concussions, including ensuring that the age-appropriate sized ball is used in practice and play, possible rule changes, evaluation of different playing surfaces, and equipment usage. To date, there is no strong evidence to support the use of mouth guards or helmets to reduce concussions in soccer. Additionally, the current data about the value of head impact sensors in soccer has not supported its widespread use.
Continue to: Finally, the issue of the prevalence...
Finally, the issue of the prevalence of chronic traumatic encephalopathy (CTE) in soccer players is beyond the scope of this article. The expert opinion from the 2017 US Soccer Federation, Major League Soccer (MLS), and National Women’s Soccer League (NWSL) conference concluded, “At present, no data exist that support that soccer participation is a risk factor for the development of neurodegenerative disease. Similarly, at this time, consistent with evidence discussed in the Berlin Concussion in Sport Group (CISG) Consensus Conference, our review suggests no causal relationship has been demonstrated between soccer and CTE pathology.”12
The more we know about concussions, both in general and those sustained during soccer play, the better we are able to diagnose and manage these injuries in our athletes. An important step is creating evidence-based protocols that evolve as our knowledge of concussions does as well. In April 2017, the US Soccer Federation, MLS, and the NWSL held a joint summit entitled, “Head Injury in Soccer: From Science to the Field” to address the current evidence-based science of concussions in soccer.8 An article discussing the findings of this meeting is forthcoming and will undoubtedly guide further development of concussion protocols for soccer players of all ages.
HEART
The physiologic demands of soccer place considerable stress on the cardiovascular system. Participation in training and competition is characterized by a combination of aerobic and anaerobic physiology with the typical athlete covering approximately 10 km over the course of the 90-minute match. The primary role of the heart and blood vessels is to supply the exercising skeletal muscle with oxygen and energy substrate and to clear the byproducts of metabolism. Among healthy athletes without cardiovascular disease, these processes are typically well tolerated and may be associated with beneficial cardiovascular adaptations over time. However, competitive soccer players are not completely immune to cardiovascular disease. Athletes across the age and competition spectrum may develop symptoms suggestive of underlying cardiovascular disease during play including exertional chest pain, inappropriate shortness of breath, palpitations, and syncope. These athletes require timely clinical evaluation. In extremely rare but high visibility cases, competitive soccer players may succumb to cardiac arrest on the pitch, underscoring the need for comprehensive emergency action plans (EAPs). We provide the practicing clinician with an overview of cardiovascular issues relevant to the competitive soccer athlete.
CARDIOVASCULAR ADAPTATIONS TO SPORT
The pressure (ie, repetitive surges in systemic blood pressure) and volume (ie, sustained increases in high cardiac output) challenges inherent in soccer participation place stress on the cardiovascular system. Healthy athletes across the age spectrum typically tolerate the hemodynamic stressors of participation without issues. Athletes that engage in training and competition over months to years often develop beneficial adaptations of the cardiovascular system that enhance on-field performance and contribute to optimal long-term health. Detailed discussion of how the heart and blood vessels respond to exercise training is beyond the scope of this article, but the interested reader is referred to several prior publications.13,14 In brief, the heart of the healthy soccer athlete demonstrates the balanced mild chamber dilation and wall thickening characteristic of left ventricular eccentric remodeling. This form of exercise-induced cardiac remodeling facilitates maintenance of high stroke volume during exercise with minimal increases in cardiac work. In parallel, routine aerobic exercise training confers favorable changes in the systemic arterial system, which leads to reductions in age-associated ventricular stiffening and maintenance of healthy low blood pressure. It must be emphasized that the healthy heart muscle dilation and thickening that develop in response to sports participation, regardless of age, ethnicity, or gender, are relatively mild and should not be confused with common forms of heart muscle disease that may be seen in athletes at risk for adverse outcomes. In some situations, consultation with an extreme sports cardiologist may be required to differentiate exercise-induced remodeling from over heart muscle pathology.15
Continue to: THE SYMPTOMATIC ATHLETE...
THE SYMPTOMATIC ATHLETE
Any athlete presenting with symptoms suggestive of underlying cardiovascular disease should be withheld from training and competition until a comprehensive clinical evaluation has been completed. Common manifestations of underlying heart disease that occur in soccer players include exertional chest pain/pressure/tightness, shortness of breath that is out of proportion to workload, palpitations or the perception of irregular cardiac activity, and syncope. Chest discomfort, inappropriate shortness of breath, and palpitations that occur during training or competition should be managed with immediate removal from the playing field and prompt medical assessment. In many cases, thorough evaluation will involve collaboration between sports medicine and sports cardiology providers. Evaluations must be individualized on a case-by-case basis as tailored to the athlete’s presenting chief complaint and prior medical history. Most of these assessments will include a detailed medical history and physical examination, a 12-lead electrocardiogram (ECG), provocative exercise testing in a controlled environment, noninvasive cardiac imaging, and in some cases ambulatory rhythm monitoring. Uniformly, the athlete should be withheld from further training and competition until high-risk cardiovascular disease has been excluded. We refer the interested reader to a comprehensive discussion of symptom-based assessment of the athlete with suspected cardiovascular disease.16
Syncope (sudden and abrupt loss of consciousness with spontaneous neurologic recovery) is common among trained athletes. The vast majority of syncope is caused by “neurocardiogenic” mechanisms and carries a benign prognosis. Benign neurally-mediated syncope most often occurs outside of training and competition among athletes with heightened vagal tone and a predisposed susceptibility to triggers including pain, anxiety, emotional stimulation, and sudden postural change. Athletes who experience neurally-mediated syncope outside of training and competition routinely report a pre-event prodrome or aura that permits them to lower themselves to the ground, thereby avoiding injury. A distinct, but similarly benign and common, form of neurally-mediated fainting is post-exertional syncope. Here, fainting occurs within seconds of abrupt termination of exercise due to a rapid reduction cardiac preload and corollary cerebral blood supply. When either form of neurally-mediated syncope is suggested by a comprehensive medical history, normal physical examination, and a normal 12- lead ECG, further evaluation is unnecessary. However, the athlete and their coaching staff should be educated about avoidance tactics including hydration, dietary sodium supplementation, and avoidance of abrupt exercise termination as neutrally-mediated syncope tends to be recurrent without such measures. Fainting episodes that occur during training or competition that are not clearly post-exertional should be considered a medical emergency and should prompt comprehensive evaluation by a qualified cardiovascular specialist. Working closely with team physicians and athletic training staff, it is the responsibility of sports cardiologists to exclude potentially life-threatening forms of electrical, muscular, coronary, and valvular heart disease. Ideally, this evaluation should be conducted rapidly to avoid unnecessary delays in return to play.
CARDIAC ARREST AND SUDDEN DEATH
Numerous high-visibility cases of cardiac arrest on the soccer pitch have alerted the sporting community to the potential for these rare and potentially tragic events. Definitive incidence statistics defining the risk of cardiac arrest among soccer players are lacking. Data from the NCAA database suggest a sudden death incidence rate among collegiate male soccer athletes of approximately 1:24,000 athlete years.8 Similar data documenting incidence among female athletes and among those at lower (ie, youth level and high school) and higher (ie, professional) levels of play are unavailable. Underlying cardiovascular disease in the forms of heart muscle abnormalities (ie, genetic and acquired cardiomyopathy), coronary artery abnormalities (ie, genetic coronary anomalies and atherosclerotic coronary disease), valvular heart problems (ie, congenitally malformed aortic valves), and primary disturbances of the cardiac electrical system (ie, Wolf-Parkinson-White syndrome, long QT syndrome, etc.) explain a substantial percentage of on-pitch cardiac arrest. However, it is increasingly recognized that a significant minority of sudden cardiac deaths among athletes occur in the absence of attributable cardiovascular abnormality.17 Such cases, often referred to as “sudden unexplained death,” present unique challenges in the context of pre-participation screening, as they are undetectable and thus unpredictable.
Reduction of cardiac arrest and sudden death may best be accomplished through a combination of focused pre-participation screening and the development and implementation of a comprehensive EAP. Pre-participation involves the performance of a battery of tests prior to training and competition that are geared toward the detection of occult high-risk cardiovascular disease. Recommendations regarding pre-participation screening vary both across and within countries. Current US recommendations call for a focused medical history and physical examination prior to training and competition with consideration of the addition of a 12-lead ECG on a local level based on expertise and available resources.18 Conversely, current European guidelines including those endorsed by FIFA, suggest routine inclusion of a 12-lead ECG and in some cases, a transthoracic ECG.19 It must be emphasized that no screening approach has been confirmed to reduce the incidence of sudden death, and the decision to extend screening beyond the medical history and physical examination to include a 12-lead ECG or echocardiogram may come at the cost of increased false positive testing. In practice, decisions about how and when to screen are ideally made at a local level after consideration of medical and financial resources.20
Continue to: Even the most comprehensive approach...
Even the most comprehensive approach to screening and evaluation of symptomatic athletes will not completely eliminate on-pitch cardiac arrest. Thus, all stakeholders that engage in the oversight of organized soccer must be committed to the development and implementation of an EAP.21 Key components of an effective EAP include the training of coaching staff, athletic trainers, and players in basic cardiopulmonary resuscitation, access to and training in the use of automated external defibrillators, and a triage/transport protocol that ensures timely access to advanced cardiac life support. Much like screening, emergency action planning involving these key core components must be developed and tailored locally. In the era of contemporary organized athletics, the absence of an EAP at any level of competition, from youth to professional leagues, is unacceptable. Effective EAPs must be developed, documented, and rehearsed at regular intervals. For the health and safety of competitive soccer players, as well as coaching staff and spectators, these steps are of critical importance.
HEAT
Heat-related illnesses can be serious and, at times, even life threatening. It is important for athletic staff and athletes to be well versed in the prevention, signs and symptoms, and treatment of heat-related illnesses in order to prevent serious and lasting injury. We aim to educate physicians about the prevention, recognition, and management of heat-related injury, and stress the importance of similarly educating athletes and coaching staff.
Exertional heat illnesses most often occur at temperatures >86°F, however they can occur at any temperature with heavy exertion.22 Signs and symptoms can be nonspecific early on, including weakness, fatigue, headache, nausea, and dizziness. Later signs can include imbalance, altered mentation, confusion, and behavior that is out of character such as irritability or aggression.23 It is easy to see how the later signs can be confused for concussion in the right context. We cover the recognition and treatment of two common and serious heat-related illnesses: heat exhaustion and exertional heat stroke (EHS).
HEAT EXHAUSTION
Heat exhaustion occurs when an athlete cannot continue to exercise due to weakness and fatigue. While the exact mechanism is not well understood, it has been established that the combined effect of heat and dehydration have been proven to decrease exercise capacity and performance to a greater degree than either alone. The heat created by the body during exercise is 15 to 20 times greater than when at rest, and can increase core body temperature by 1°C every 5 minutes if no heat is lost, such as through sweating.24 Additionally, when fluid deficits reach >3% to 5% of total body water, sweat production and skin blood flow decline, blunting the ability for the body to cool itself and causing progressive elevation of core body temperature if the athlete continues exerting him or herself. When fluid deficits reach 6% to 10%, cardiac output, sweat, and muscle blood flow decrease, likely leading to the symptoms seen with heat exhaustion: weakness, profound fatigue, and occasionally confusion and disorientation. Athletes with suspected heat exhaustion should be moved to a cooler area, laid down with legs elevated, and orally rehydrated. If they do not improve with oral rehydration, they may require intravenous fluids. The diagnosis of heat exhaustion hinges on a rectal temperature of <104°F; if >104°F the athlete should be presumed to have heat stroke, which will be addressed in the following paragraphs. Players can be cleared to return to play in mild cases within 24 to 48 hours with gradual increases in exercise intensity.24
EXERTIONAL HEAT STROKE
EHS occurs when the body can no longer regulate the core body temperature and it rises to upwards of 104°F. In EHS, elevated core body temperature is associated with evidence of end organ dysfunction. The most easily identified on the playing field is likely central nervous system dysfunction, including irritability, confusion, irrational behavior, lethargy, dizziness, confusion, and even loss of consciousness. Temperature should be measured with rectal temperature only, as other methods of measurement have been shown to be consistently inaccurate.22 Heat stroke can be confused with exertional hyponatremia, heat exhaustion, or concussion, especially when core body temperature cannot be determined. However, EHS should always be the presumed cause of altered mentation when no rectal temperature is available because rapid cooling is critical to minimizing lasting effects. Morbidity and mortality are directly related to the length of time required to cool the athlete under 40°C (104°F).24 Cooling should be completed on site prior to transport to a medical facility and is best achieved with submersion in an ice bath (ie, a kiddie pool or soaking tub full of ice and water).22,25 If an ice bath is not available, ice bags should be applied to the neck axilla and groin and exchanged for fresh bags every 2 to 3 minutes.22 Ice bags have been shown to be inferior to whole body cooling, only cooling the athlete .04°C to .08°C/min compared to .15°C to .24°C/min with the ice bath.24 All other tests should be delayed until cooling is achieved, unless they can be completed while cooling the athlete. The athlete can be removed from the ice bath once rectal temperatures reach <101°F to 102°F.23 If the athlete returns to baseline after cooling, transportation to a medical facility may not be necessary. However, they should refrain from physical activity and heat exposure for at least 7 days and should be evaluated by a physician at that time. If all labs are normal and the athlete is asymptomatic, they can start progressive return to play under the direction of an athletic trainer or a sports medicine physician.23
Continue to: HEAT-RELATED ILLNESS...
HEAT-RELATED ILLNESS
It is impossible to predict exactly which athletes will be most at risk for heat-related illness, so it is important to have a high degree of suspicion when environmental conditions are right. Athletes with recent illness, fever, or lack of sleep are at higher risk. Additional intrinsic risk factors include low fitness level, obesity, and inadequate hydration. Athletes who are highly competitive or motivated can be more likely to push through the early signs of illness or be reluctant to report symptoms.23 Those with a history of exertional heat illness are more at risk for developing it again in the future.23
The extrinsic risk factors for the development of heat-related illness are much easier to identify and modify in order to keep athletes safe. High temperature and high humidity conditions, heavy sun exposure, and exposure to similar conditions the preceding day put athletes at risk for exertional heat illness. Risks are even greater when the exercise is prolonged or intense with few breaks and access to hydration is limited.23 Therefore, prevention of exertional heat illness is centered on these external risk factors.
Each team should have a heat policy as part of their EAP aimed at prevention and early recognition of heat-related illness. This policy should be shared with all athletes and coaches. The plan should be centered on acclimatization, activity modification, and early recognition and management as previously discussed. The US Soccer Federation “Recognize to Recover” Heat Guidelines suggest a 3-step process for appropriate activity modification:22
1. Find the wet bulb globe temperature, either using a wet bulb globe thermometer or the temperature and humidity (Figure 1).
2. Find your regional weather category on the map (Figure 2).
3. Find your alert level and work to rest ratio recommendations (Figure 3).
Scheduled hydration breaks should be given as listed in Figure 3. Breaks of 4 minutes should be given for each 30 minutes of continuous practice or play. In a regulation 90-minute match, a hydration break should be given at 30 and 75 minutes (with half time at 45 minutes) at minimum. Athletes should be educated about where hydration can be accessed, and given unlimited access to hydration even outside of planned breaks.22
Acclimatization to conditions is another integral part of preventing heat-related illness. It allows the body time to adapt to exercising in heat gradually, with a measured progression of exertion over the course of 10 to 14 days. The “Recognize to Recover” Heat Guidelines also provide guidance on acclimatization, and specifics can be found on the website.1 Generally speaking, the warmest part of the day, usually between 11 AM and 4 PM, should be avoided for all training sessions, and length of practice and exertion should be gradually increased over 2 weeks.22
In summary, appropriate acclimatization, hydration, activity modification, and education of athletes and staff are essential for the prevention of heat-related illness. Athletes and staff should understand the signs and symptoms of heat-related illness so that it can be recognized early and treated appropriately. If an athlete is altered in the heat and rectal temperature is >104°F or rectal temperature cannot be obtained, rapid cooling using an ice bath or ice bags is essential to prevent the morbidity and mortality associated with EHS. Above all, teams should have an explicit plan that includes protocols for acclimatization, activity modification, and all necessary equipment to prevent and treat heat-related illnesses should they occur, and ultimately keep athletes safe and healthy.
1. Langlois JA, Rutland-Brown W, Wald MM. The epidemiology and impact of traumatic brain Injury: a brief overview. J Head Trauma Rehabil. 2006;21(5):375-378.
2. The National Federation of State High School Associations. 2013-14 high school athletics participation survey. http://www.nfhs.org/ParticipationStatics/PDF/2013-14_Participation_Survey_PDF.pdf. Accessed August 6, 2018.
3. Youth Council. US Soccer Federation Web site. https://www.ussoccer.com/about/affiliates/youth-council. Accessed July 31, 2018.
4. Khodaee M, Currie DW, Asif IM, Comstock RD. Nine-year study of US high school soccer injuries: data from a national sports injury surveillance programme. Br J Sports Med. 2017;51(3):185-193. doi:10.1136/bjsports-2015-095946.
5. Schallmo MS, Weiner JA, Hsu WK. Sport and sex-specific trends in the epidemiology of concussions sustained by high school athletes. J Bone Joint Surg Am. 2017;99(15):1314-1320. doi:10.2106/JBJS.16.01573.
6. US Soccer Federation. U.S. Soccer’s comprehensive player health and safety program. Recognize to Recover Web site. http://www.recognizetorecover.org/#us-soccers-comprehensive-player-health-and-safety-program. Accessed July 31, 2018.
7. McCrory P, Meeuwisse W, Dvořák J, et al. Consensus statement on concussion in sport-the 5th international conference on concussion in sport held in Berlin, October 2016. Br J Sports Med. 2017;51(11):838-847. doi:10.1136/bjsports-2017-097699.
8. Harmon KG, Asif IM, Klossner D, Drezner JA. Incidence of sudden cardiac death in National Collegiate Athletic Association athletes. Circulation. 2011;123(15):1594-1600. doi:10.1161/CIRCULATIONAHA.110.004622.
9. Faude O, Rössler R, Junge A, et al. Head injuries in children’s football-results from two prospective cohort studies in four European countries. Scand J Med Sci Sports. 2017;27(12):1986-1992. doi:10.1111/sms.12839.
10. Comstock RD, Currie DW, Pierpoint LA, Grubenhoff JA, Fields SK. An evidence-based discussion of heading the ball and concussions in high school soccer. JAMA Pediatr. 2015;169(9):830-837. doi:10.1001/jamapediatrics.2015.1062.
11. Tarnutzer AA. Should heading be forbidden in children’s football? Sci Med Football. 2018;2(1):75-79.
12. US Soccer Federation. US Soccer, NWSL and MLS to host “head injury in soccer; science to field”. https://www.ussoccer.com/stories/2017/04/18/17/35/20170418-news-us-soccer-nwsl-mls-host-head-injury-in-soccer-science-to-field. Published April 18, 2017. Accessed August 6, 2018.
13. Weiner RB, Baggish AL. Exercise-induced cardiac remodeling. Prog Cardiovasc Dis. 2012;54(5):380-386. doi:10.1016/j.pcad.2012.01.006.
14. Baggish AL, Wood MJ. Athlete's heart and cardiovascular care of the athlete: scientific and clinical update. Circulation. 2011;123(23):2723-2735. doi:10.1161/CIRCULATIONAHA.110.981571.
15. Kim JH, Baggish AL. Differentiating exercise-induced cardiac adaptations from cardiac pathology: the "Grey Zone" of clinical uncertainty. Can J Cardiol. 2016;32(4):429-437. doi:10.1016/j.cjca.2015.11.025.
16. Baggish AL, Battle RW, Beckerman JG, et al; ACC’s Sports and Exercise Council Leadership Group. Sports cardiology: core curriculum for providing cardiovascular care to competitive athletes and highly active people. J Am Coll Cardiol. 2017;70(15):1902-1918. doi:10.1016/j.jacc.2017.08.055.
17. Harmon KG, Asif IM, Maleszewski JJ, et al. Incidence, cause, and comparative frequency of sudden cardiac death in National Collegiate Athletic Association athletes: a decade in review. Circulation. 2015;132(1):10-19. doi:10.1161/CIRCULATIONAHA.115.015431.
18. Maron BJ, Levine BD, Washington RL, Baggish AL, Kovacs RJ, Maron MS. Eligibility and disqualification recommendations for competitive athletes with cardiovascular abnormalities: task force 2: preparticipation screening for cardiovascular disease in competitive athletes: a scientific statement from the American Heart Association and American College of Cardiology. J Am Coll Cardiol. 2015;66(21):2356-2361. doi:10.1016/j.jacc.2015.09.034.
19. Corrado D, Pelliccia A, Bjørnstad HH, et al. Cardiovascular pre-participation screening of young competitive athletes for prevention of sudden death: proposal for a common European protocol. Consensus Statement of the Study Group of Sport Cardiology of the Working Group of Cardiac Rehabilitation and Exercise Physiology and the Working Group of Myocardial and Pericardial Diseases of the European Society of Cardiology. Eur Heart J. 2005;26(5):516-524.
20. Baggish AL, Kovacs RJ. Preparticipation cardiovascular screening: clinical partnership is the only certainty. Br J Sports Med. 2017;51(3):150-151. doi:10.1136/bjsports-2016-096954.
21. Hainline B, Drezner J, Baggish A, et al. Interassociation consensus statement on cardiovascular care of college student-athletes. J Athl Train. 2016;51(4):344-357. doi:10.4085/j.jacc.2016.03.527.
22. US Soccer Federation. Environmental conditions. Recognize to Recover Web site. http://www.recognizetorecover.org/environmental/#environmental-conditions. Accessed April 15, 2018.
23. Korey Stringer Institute. Emergency conditions: heat illnesses. University of Connecticut Web site. https://ksi.uconn.edu/. Accessed April 15, 2018.
24. American College of Sports Medicine, Armstrong LE, Casa DJ, et al. American College of Sports Medicine position stand. Exertional heat illness during training and competition. Med Sci Sports Exerc. 2007;39(3):556-572.
25. Belval LN, Casa DJ, Adams WM, et al. Consensus statement- prehospital care of exertional heat stroke. Prehosp Emerg Care. 2018;22(3):392-397. doi:10.1080/10903127.2017.1392666.
ABSTRACT
Soccer requires significant physical conditioning and endurance, as well as the physicality required for contact play. In order to keep athletes safe, it is important that coaches, medical staff, and the players themselves are educated on the most common dangers to their health that they may encounter on a soccer pitch. This article aims to review the current literature and recommendations on concussion, cardiovascular considerations, and heat-related illness as they relate to competitive soccer, with a goal of educating all those who help to keep athletes healthy and competing to their full potential.
Continue to: Soccer is one of the most popular sports...
Soccer is one of the most popular sports in the modern world and requires significant physical conditioning and endurance, as well as the physicality required for contact play. This article covers the topics of concussion, cardiovascular considerations, and heat-related illness as they relate to competitive soccer players. We provide a review of the prevention, recognition, and management required to keep athletes safe on the soccer pitch, both in practice and in competitive play.
HEAD
With an estimated 1.6 to 3.8 million sports-related mild traumatic brain injuries (ie, concussions) occurring annually in the United States,1 there has been an appropriate increase in the focus on prevention and treatment of these injuries. For more than a decade the spotlight has been on concussions that occur in American football, but other sports have also had to examine the prevalence of concussions in their sport. This is certainly true for soccer.
There has been a steady increase in soccer participation in the United States. From 1973 to 2014 there was a 4-fold increase in high school boys and a 35-fold increase in high school girls playing soccer.2 Currently, there are more than 3.7 million youth who play on teams under the supervision of the US Soccer Federation, the sport’s national governing body.3 With the growth of the sport, there has also been an intensified focus on injury prevention in soccer players, including concussive brain injuries.
A recent study examined injury rates in high school soccer players and noted that concussion is the second most common injury (17.9%), after ligament sprains (29.7%).4 The overall injury rate was 2.06 per 1000 athletic exposures (AEs [defined as participation in practice or game play]), but higher in games (4.42 per 1000 AEs) than during practices (1.05 per 1000 AEs). The overall concussion rate was 0.36 per 1000 AEs.4
Most concussions (54.8%) resulted in missing play between 1 to 3 weeks, but a sizeable portion of the athletes (14.9%) were out of play for more than 3 weeks. Additionally, 10.7% of all medical disqualifications were due to concussive injuries. Khodaee and colleagues4 found no statistically significant difference in concussion rates between male and female soccer players over the 9-year period of time that they examined, however previous studies have found higher rates in female athletes.5
In soccer, as in other sports, there is a concern about both the adequate recognition of concussions during practice and play and the underreporting of concussions by athletes. The US Soccer Federation has taken a proactive stance on addressing concussion in youth soccer by developing the “Recognize to Recover” program.6 Recognize to Recover is the US Soccer Federation’s “comprehensive player health and safety program aimed at promoting safe play and reducing injuries in soccer players of all ages.” The website provides an educational video geared toward players, along with links to concussion assessment tools, the US Soccer Federation Concussion Protocol, and US Soccer Federation-Centers for Disease Control fact sheets for athletes, parents, and coaches.6
Continue to: A challenge for all sports...
A challenge for all sports is allowing adequate evaluation of a suspected concussion by properly trained healthcare professionals. The 2017 Berlin Concussion in Sport Group position paper stated, “when a concussion is suspected, the athlete should be removed from the sporting environment and a multimodal assessment should be conducted in standardized fashion (eg, Sport Concussion Assessment Tool- 5th edition). Sporting bodies should allow adequate time to conduct this evaluation.”7 However, the International Federation of Football Association (FIFA) rules limit substitutions to 3 over the course of the game, which can make a thorough evaluation of players difficult as trainers and coaches are under increased pressure to quickly determine whether to use one of their valuable substitutions. Fortunately, the National Collegiate Athletic Association (NCAA) soccer has mitigated this issue by allowing unlimited substitutions during matches, and high school teams generally follow similar rules.
One of the goals of any safety education program is not only to raise the awareness of the signs and symptoms of concussion by all those involved in the sport, but also to increase the number of athletes who self-report their symptoms and decrease those who hide any possible concussions. A study found that a majority (58.6%) of middle school soccer players continued to play while experiencing concussion symptoms.5 However, in a very recent (not yet published) study, 92% of US Soccer Federation players reported that they did seek out a medical evaluation for their concussion.8 This is certainly a positive sign and further research needs to clarify what methods of education or training will maintain this level of self-reporting in soccer players.
There has also been an increased focus on understanding the mechanism of injury of concussions. In soccer, concussions can occur from player-to-player contact, contact with the player surface, contact with playing apparatus (eg, goal posts) and non-contact mechanisms. While there has been a focus on concussions from heading the ball, player-to-player contact is the most common cause of concussions. A 2017 study of 7- to 12-year-old soccer players in 4 European countries found that about 1 out of 10 concussions were caused by heading the ball.9 Comstock and colleageues10 found slightly higher numbers, roughly 25% to 30% (depending on gender), but 70% to 78% (again depending on gender) of those were caused by player-to-player contact rather than contact with the ball.
To date there has not been any meta-analytic review evaluating the cognitive and physical symptoms associated with heading in soccer. A recent review paper stated that the “current evidence seems insufficient to support a ban of heading in children’s football (soccer).”10 However, in December 2015 the US Soccer Federation included age-specific heading limitations. Players ages 10 years and under “shall not engage in heading, either in practices or in games” and players age 11 years and 12 years should have “limited heading in practice; maximum of 30 minutes of heading training per week, with no more than 15-20 headers per player, per week.”11 US Soccer Federation officials acknowledged the limitations in the current science regarding heading in young soccer players but chose to err on the side of caution until further empirical evidence regarding the risks associated with repetitive heading is available.
The US Soccer Federation is also exploring other ways to reduce the incidence rate of concussions, including ensuring that the age-appropriate sized ball is used in practice and play, possible rule changes, evaluation of different playing surfaces, and equipment usage. To date, there is no strong evidence to support the use of mouth guards or helmets to reduce concussions in soccer. Additionally, the current data about the value of head impact sensors in soccer has not supported its widespread use.
Continue to: Finally, the issue of the prevalence...
Finally, the issue of the prevalence of chronic traumatic encephalopathy (CTE) in soccer players is beyond the scope of this article. The expert opinion from the 2017 US Soccer Federation, Major League Soccer (MLS), and National Women’s Soccer League (NWSL) conference concluded, “At present, no data exist that support that soccer participation is a risk factor for the development of neurodegenerative disease. Similarly, at this time, consistent with evidence discussed in the Berlin Concussion in Sport Group (CISG) Consensus Conference, our review suggests no causal relationship has been demonstrated between soccer and CTE pathology.”12
The more we know about concussions, both in general and those sustained during soccer play, the better we are able to diagnose and manage these injuries in our athletes. An important step is creating evidence-based protocols that evolve as our knowledge of concussions does as well. In April 2017, the US Soccer Federation, MLS, and the NWSL held a joint summit entitled, “Head Injury in Soccer: From Science to the Field” to address the current evidence-based science of concussions in soccer.8 An article discussing the findings of this meeting is forthcoming and will undoubtedly guide further development of concussion protocols for soccer players of all ages.
HEART
The physiologic demands of soccer place considerable stress on the cardiovascular system. Participation in training and competition is characterized by a combination of aerobic and anaerobic physiology with the typical athlete covering approximately 10 km over the course of the 90-minute match. The primary role of the heart and blood vessels is to supply the exercising skeletal muscle with oxygen and energy substrate and to clear the byproducts of metabolism. Among healthy athletes without cardiovascular disease, these processes are typically well tolerated and may be associated with beneficial cardiovascular adaptations over time. However, competitive soccer players are not completely immune to cardiovascular disease. Athletes across the age and competition spectrum may develop symptoms suggestive of underlying cardiovascular disease during play including exertional chest pain, inappropriate shortness of breath, palpitations, and syncope. These athletes require timely clinical evaluation. In extremely rare but high visibility cases, competitive soccer players may succumb to cardiac arrest on the pitch, underscoring the need for comprehensive emergency action plans (EAPs). We provide the practicing clinician with an overview of cardiovascular issues relevant to the competitive soccer athlete.
CARDIOVASCULAR ADAPTATIONS TO SPORT
The pressure (ie, repetitive surges in systemic blood pressure) and volume (ie, sustained increases in high cardiac output) challenges inherent in soccer participation place stress on the cardiovascular system. Healthy athletes across the age spectrum typically tolerate the hemodynamic stressors of participation without issues. Athletes that engage in training and competition over months to years often develop beneficial adaptations of the cardiovascular system that enhance on-field performance and contribute to optimal long-term health. Detailed discussion of how the heart and blood vessels respond to exercise training is beyond the scope of this article, but the interested reader is referred to several prior publications.13,14 In brief, the heart of the healthy soccer athlete demonstrates the balanced mild chamber dilation and wall thickening characteristic of left ventricular eccentric remodeling. This form of exercise-induced cardiac remodeling facilitates maintenance of high stroke volume during exercise with minimal increases in cardiac work. In parallel, routine aerobic exercise training confers favorable changes in the systemic arterial system, which leads to reductions in age-associated ventricular stiffening and maintenance of healthy low blood pressure. It must be emphasized that the healthy heart muscle dilation and thickening that develop in response to sports participation, regardless of age, ethnicity, or gender, are relatively mild and should not be confused with common forms of heart muscle disease that may be seen in athletes at risk for adverse outcomes. In some situations, consultation with an extreme sports cardiologist may be required to differentiate exercise-induced remodeling from over heart muscle pathology.15
Continue to: THE SYMPTOMATIC ATHLETE...
THE SYMPTOMATIC ATHLETE
Any athlete presenting with symptoms suggestive of underlying cardiovascular disease should be withheld from training and competition until a comprehensive clinical evaluation has been completed. Common manifestations of underlying heart disease that occur in soccer players include exertional chest pain/pressure/tightness, shortness of breath that is out of proportion to workload, palpitations or the perception of irregular cardiac activity, and syncope. Chest discomfort, inappropriate shortness of breath, and palpitations that occur during training or competition should be managed with immediate removal from the playing field and prompt medical assessment. In many cases, thorough evaluation will involve collaboration between sports medicine and sports cardiology providers. Evaluations must be individualized on a case-by-case basis as tailored to the athlete’s presenting chief complaint and prior medical history. Most of these assessments will include a detailed medical history and physical examination, a 12-lead electrocardiogram (ECG), provocative exercise testing in a controlled environment, noninvasive cardiac imaging, and in some cases ambulatory rhythm monitoring. Uniformly, the athlete should be withheld from further training and competition until high-risk cardiovascular disease has been excluded. We refer the interested reader to a comprehensive discussion of symptom-based assessment of the athlete with suspected cardiovascular disease.16
Syncope (sudden and abrupt loss of consciousness with spontaneous neurologic recovery) is common among trained athletes. The vast majority of syncope is caused by “neurocardiogenic” mechanisms and carries a benign prognosis. Benign neurally-mediated syncope most often occurs outside of training and competition among athletes with heightened vagal tone and a predisposed susceptibility to triggers including pain, anxiety, emotional stimulation, and sudden postural change. Athletes who experience neurally-mediated syncope outside of training and competition routinely report a pre-event prodrome or aura that permits them to lower themselves to the ground, thereby avoiding injury. A distinct, but similarly benign and common, form of neurally-mediated fainting is post-exertional syncope. Here, fainting occurs within seconds of abrupt termination of exercise due to a rapid reduction cardiac preload and corollary cerebral blood supply. When either form of neurally-mediated syncope is suggested by a comprehensive medical history, normal physical examination, and a normal 12- lead ECG, further evaluation is unnecessary. However, the athlete and their coaching staff should be educated about avoidance tactics including hydration, dietary sodium supplementation, and avoidance of abrupt exercise termination as neutrally-mediated syncope tends to be recurrent without such measures. Fainting episodes that occur during training or competition that are not clearly post-exertional should be considered a medical emergency and should prompt comprehensive evaluation by a qualified cardiovascular specialist. Working closely with team physicians and athletic training staff, it is the responsibility of sports cardiologists to exclude potentially life-threatening forms of electrical, muscular, coronary, and valvular heart disease. Ideally, this evaluation should be conducted rapidly to avoid unnecessary delays in return to play.
CARDIAC ARREST AND SUDDEN DEATH
Numerous high-visibility cases of cardiac arrest on the soccer pitch have alerted the sporting community to the potential for these rare and potentially tragic events. Definitive incidence statistics defining the risk of cardiac arrest among soccer players are lacking. Data from the NCAA database suggest a sudden death incidence rate among collegiate male soccer athletes of approximately 1:24,000 athlete years.8 Similar data documenting incidence among female athletes and among those at lower (ie, youth level and high school) and higher (ie, professional) levels of play are unavailable. Underlying cardiovascular disease in the forms of heart muscle abnormalities (ie, genetic and acquired cardiomyopathy), coronary artery abnormalities (ie, genetic coronary anomalies and atherosclerotic coronary disease), valvular heart problems (ie, congenitally malformed aortic valves), and primary disturbances of the cardiac electrical system (ie, Wolf-Parkinson-White syndrome, long QT syndrome, etc.) explain a substantial percentage of on-pitch cardiac arrest. However, it is increasingly recognized that a significant minority of sudden cardiac deaths among athletes occur in the absence of attributable cardiovascular abnormality.17 Such cases, often referred to as “sudden unexplained death,” present unique challenges in the context of pre-participation screening, as they are undetectable and thus unpredictable.
Reduction of cardiac arrest and sudden death may best be accomplished through a combination of focused pre-participation screening and the development and implementation of a comprehensive EAP. Pre-participation involves the performance of a battery of tests prior to training and competition that are geared toward the detection of occult high-risk cardiovascular disease. Recommendations regarding pre-participation screening vary both across and within countries. Current US recommendations call for a focused medical history and physical examination prior to training and competition with consideration of the addition of a 12-lead ECG on a local level based on expertise and available resources.18 Conversely, current European guidelines including those endorsed by FIFA, suggest routine inclusion of a 12-lead ECG and in some cases, a transthoracic ECG.19 It must be emphasized that no screening approach has been confirmed to reduce the incidence of sudden death, and the decision to extend screening beyond the medical history and physical examination to include a 12-lead ECG or echocardiogram may come at the cost of increased false positive testing. In practice, decisions about how and when to screen are ideally made at a local level after consideration of medical and financial resources.20
Continue to: Even the most comprehensive approach...
Even the most comprehensive approach to screening and evaluation of symptomatic athletes will not completely eliminate on-pitch cardiac arrest. Thus, all stakeholders that engage in the oversight of organized soccer must be committed to the development and implementation of an EAP.21 Key components of an effective EAP include the training of coaching staff, athletic trainers, and players in basic cardiopulmonary resuscitation, access to and training in the use of automated external defibrillators, and a triage/transport protocol that ensures timely access to advanced cardiac life support. Much like screening, emergency action planning involving these key core components must be developed and tailored locally. In the era of contemporary organized athletics, the absence of an EAP at any level of competition, from youth to professional leagues, is unacceptable. Effective EAPs must be developed, documented, and rehearsed at regular intervals. For the health and safety of competitive soccer players, as well as coaching staff and spectators, these steps are of critical importance.
HEAT
Heat-related illnesses can be serious and, at times, even life threatening. It is important for athletic staff and athletes to be well versed in the prevention, signs and symptoms, and treatment of heat-related illnesses in order to prevent serious and lasting injury. We aim to educate physicians about the prevention, recognition, and management of heat-related injury, and stress the importance of similarly educating athletes and coaching staff.
Exertional heat illnesses most often occur at temperatures >86°F, however they can occur at any temperature with heavy exertion.22 Signs and symptoms can be nonspecific early on, including weakness, fatigue, headache, nausea, and dizziness. Later signs can include imbalance, altered mentation, confusion, and behavior that is out of character such as irritability or aggression.23 It is easy to see how the later signs can be confused for concussion in the right context. We cover the recognition and treatment of two common and serious heat-related illnesses: heat exhaustion and exertional heat stroke (EHS).
HEAT EXHAUSTION
Heat exhaustion occurs when an athlete cannot continue to exercise due to weakness and fatigue. While the exact mechanism is not well understood, it has been established that the combined effect of heat and dehydration have been proven to decrease exercise capacity and performance to a greater degree than either alone. The heat created by the body during exercise is 15 to 20 times greater than when at rest, and can increase core body temperature by 1°C every 5 minutes if no heat is lost, such as through sweating.24 Additionally, when fluid deficits reach >3% to 5% of total body water, sweat production and skin blood flow decline, blunting the ability for the body to cool itself and causing progressive elevation of core body temperature if the athlete continues exerting him or herself. When fluid deficits reach 6% to 10%, cardiac output, sweat, and muscle blood flow decrease, likely leading to the symptoms seen with heat exhaustion: weakness, profound fatigue, and occasionally confusion and disorientation. Athletes with suspected heat exhaustion should be moved to a cooler area, laid down with legs elevated, and orally rehydrated. If they do not improve with oral rehydration, they may require intravenous fluids. The diagnosis of heat exhaustion hinges on a rectal temperature of <104°F; if >104°F the athlete should be presumed to have heat stroke, which will be addressed in the following paragraphs. Players can be cleared to return to play in mild cases within 24 to 48 hours with gradual increases in exercise intensity.24
EXERTIONAL HEAT STROKE
EHS occurs when the body can no longer regulate the core body temperature and it rises to upwards of 104°F. In EHS, elevated core body temperature is associated with evidence of end organ dysfunction. The most easily identified on the playing field is likely central nervous system dysfunction, including irritability, confusion, irrational behavior, lethargy, dizziness, confusion, and even loss of consciousness. Temperature should be measured with rectal temperature only, as other methods of measurement have been shown to be consistently inaccurate.22 Heat stroke can be confused with exertional hyponatremia, heat exhaustion, or concussion, especially when core body temperature cannot be determined. However, EHS should always be the presumed cause of altered mentation when no rectal temperature is available because rapid cooling is critical to minimizing lasting effects. Morbidity and mortality are directly related to the length of time required to cool the athlete under 40°C (104°F).24 Cooling should be completed on site prior to transport to a medical facility and is best achieved with submersion in an ice bath (ie, a kiddie pool or soaking tub full of ice and water).22,25 If an ice bath is not available, ice bags should be applied to the neck axilla and groin and exchanged for fresh bags every 2 to 3 minutes.22 Ice bags have been shown to be inferior to whole body cooling, only cooling the athlete .04°C to .08°C/min compared to .15°C to .24°C/min with the ice bath.24 All other tests should be delayed until cooling is achieved, unless they can be completed while cooling the athlete. The athlete can be removed from the ice bath once rectal temperatures reach <101°F to 102°F.23 If the athlete returns to baseline after cooling, transportation to a medical facility may not be necessary. However, they should refrain from physical activity and heat exposure for at least 7 days and should be evaluated by a physician at that time. If all labs are normal and the athlete is asymptomatic, they can start progressive return to play under the direction of an athletic trainer or a sports medicine physician.23
Continue to: HEAT-RELATED ILLNESS...
HEAT-RELATED ILLNESS
It is impossible to predict exactly which athletes will be most at risk for heat-related illness, so it is important to have a high degree of suspicion when environmental conditions are right. Athletes with recent illness, fever, or lack of sleep are at higher risk. Additional intrinsic risk factors include low fitness level, obesity, and inadequate hydration. Athletes who are highly competitive or motivated can be more likely to push through the early signs of illness or be reluctant to report symptoms.23 Those with a history of exertional heat illness are more at risk for developing it again in the future.23
The extrinsic risk factors for the development of heat-related illness are much easier to identify and modify in order to keep athletes safe. High temperature and high humidity conditions, heavy sun exposure, and exposure to similar conditions the preceding day put athletes at risk for exertional heat illness. Risks are even greater when the exercise is prolonged or intense with few breaks and access to hydration is limited.23 Therefore, prevention of exertional heat illness is centered on these external risk factors.
Each team should have a heat policy as part of their EAP aimed at prevention and early recognition of heat-related illness. This policy should be shared with all athletes and coaches. The plan should be centered on acclimatization, activity modification, and early recognition and management as previously discussed. The US Soccer Federation “Recognize to Recover” Heat Guidelines suggest a 3-step process for appropriate activity modification:22
1. Find the wet bulb globe temperature, either using a wet bulb globe thermometer or the temperature and humidity (Figure 1).
2. Find your regional weather category on the map (Figure 2).
3. Find your alert level and work to rest ratio recommendations (Figure 3).
Scheduled hydration breaks should be given as listed in Figure 3. Breaks of 4 minutes should be given for each 30 minutes of continuous practice or play. In a regulation 90-minute match, a hydration break should be given at 30 and 75 minutes (with half time at 45 minutes) at minimum. Athletes should be educated about where hydration can be accessed, and given unlimited access to hydration even outside of planned breaks.22
Acclimatization to conditions is another integral part of preventing heat-related illness. It allows the body time to adapt to exercising in heat gradually, with a measured progression of exertion over the course of 10 to 14 days. The “Recognize to Recover” Heat Guidelines also provide guidance on acclimatization, and specifics can be found on the website.1 Generally speaking, the warmest part of the day, usually between 11 AM and 4 PM, should be avoided for all training sessions, and length of practice and exertion should be gradually increased over 2 weeks.22
In summary, appropriate acclimatization, hydration, activity modification, and education of athletes and staff are essential for the prevention of heat-related illness. Athletes and staff should understand the signs and symptoms of heat-related illness so that it can be recognized early and treated appropriately. If an athlete is altered in the heat and rectal temperature is >104°F or rectal temperature cannot be obtained, rapid cooling using an ice bath or ice bags is essential to prevent the morbidity and mortality associated with EHS. Above all, teams should have an explicit plan that includes protocols for acclimatization, activity modification, and all necessary equipment to prevent and treat heat-related illnesses should they occur, and ultimately keep athletes safe and healthy.
ABSTRACT
Soccer requires significant physical conditioning and endurance, as well as the physicality required for contact play. In order to keep athletes safe, it is important that coaches, medical staff, and the players themselves are educated on the most common dangers to their health that they may encounter on a soccer pitch. This article aims to review the current literature and recommendations on concussion, cardiovascular considerations, and heat-related illness as they relate to competitive soccer, with a goal of educating all those who help to keep athletes healthy and competing to their full potential.
Continue to: Soccer is one of the most popular sports...
Soccer is one of the most popular sports in the modern world and requires significant physical conditioning and endurance, as well as the physicality required for contact play. This article covers the topics of concussion, cardiovascular considerations, and heat-related illness as they relate to competitive soccer players. We provide a review of the prevention, recognition, and management required to keep athletes safe on the soccer pitch, both in practice and in competitive play.
HEAD
With an estimated 1.6 to 3.8 million sports-related mild traumatic brain injuries (ie, concussions) occurring annually in the United States,1 there has been an appropriate increase in the focus on prevention and treatment of these injuries. For more than a decade the spotlight has been on concussions that occur in American football, but other sports have also had to examine the prevalence of concussions in their sport. This is certainly true for soccer.
There has been a steady increase in soccer participation in the United States. From 1973 to 2014 there was a 4-fold increase in high school boys and a 35-fold increase in high school girls playing soccer.2 Currently, there are more than 3.7 million youth who play on teams under the supervision of the US Soccer Federation, the sport’s national governing body.3 With the growth of the sport, there has also been an intensified focus on injury prevention in soccer players, including concussive brain injuries.
A recent study examined injury rates in high school soccer players and noted that concussion is the second most common injury (17.9%), after ligament sprains (29.7%).4 The overall injury rate was 2.06 per 1000 athletic exposures (AEs [defined as participation in practice or game play]), but higher in games (4.42 per 1000 AEs) than during practices (1.05 per 1000 AEs). The overall concussion rate was 0.36 per 1000 AEs.4
Most concussions (54.8%) resulted in missing play between 1 to 3 weeks, but a sizeable portion of the athletes (14.9%) were out of play for more than 3 weeks. Additionally, 10.7% of all medical disqualifications were due to concussive injuries. Khodaee and colleagues4 found no statistically significant difference in concussion rates between male and female soccer players over the 9-year period of time that they examined, however previous studies have found higher rates in female athletes.5
In soccer, as in other sports, there is a concern about both the adequate recognition of concussions during practice and play and the underreporting of concussions by athletes. The US Soccer Federation has taken a proactive stance on addressing concussion in youth soccer by developing the “Recognize to Recover” program.6 Recognize to Recover is the US Soccer Federation’s “comprehensive player health and safety program aimed at promoting safe play and reducing injuries in soccer players of all ages.” The website provides an educational video geared toward players, along with links to concussion assessment tools, the US Soccer Federation Concussion Protocol, and US Soccer Federation-Centers for Disease Control fact sheets for athletes, parents, and coaches.6
Continue to: A challenge for all sports...
A challenge for all sports is allowing adequate evaluation of a suspected concussion by properly trained healthcare professionals. The 2017 Berlin Concussion in Sport Group position paper stated, “when a concussion is suspected, the athlete should be removed from the sporting environment and a multimodal assessment should be conducted in standardized fashion (eg, Sport Concussion Assessment Tool- 5th edition). Sporting bodies should allow adequate time to conduct this evaluation.”7 However, the International Federation of Football Association (FIFA) rules limit substitutions to 3 over the course of the game, which can make a thorough evaluation of players difficult as trainers and coaches are under increased pressure to quickly determine whether to use one of their valuable substitutions. Fortunately, the National Collegiate Athletic Association (NCAA) soccer has mitigated this issue by allowing unlimited substitutions during matches, and high school teams generally follow similar rules.
One of the goals of any safety education program is not only to raise the awareness of the signs and symptoms of concussion by all those involved in the sport, but also to increase the number of athletes who self-report their symptoms and decrease those who hide any possible concussions. A study found that a majority (58.6%) of middle school soccer players continued to play while experiencing concussion symptoms.5 However, in a very recent (not yet published) study, 92% of US Soccer Federation players reported that they did seek out a medical evaluation for their concussion.8 This is certainly a positive sign and further research needs to clarify what methods of education or training will maintain this level of self-reporting in soccer players.
There has also been an increased focus on understanding the mechanism of injury of concussions. In soccer, concussions can occur from player-to-player contact, contact with the player surface, contact with playing apparatus (eg, goal posts) and non-contact mechanisms. While there has been a focus on concussions from heading the ball, player-to-player contact is the most common cause of concussions. A 2017 study of 7- to 12-year-old soccer players in 4 European countries found that about 1 out of 10 concussions were caused by heading the ball.9 Comstock and colleageues10 found slightly higher numbers, roughly 25% to 30% (depending on gender), but 70% to 78% (again depending on gender) of those were caused by player-to-player contact rather than contact with the ball.
To date there has not been any meta-analytic review evaluating the cognitive and physical symptoms associated with heading in soccer. A recent review paper stated that the “current evidence seems insufficient to support a ban of heading in children’s football (soccer).”10 However, in December 2015 the US Soccer Federation included age-specific heading limitations. Players ages 10 years and under “shall not engage in heading, either in practices or in games” and players age 11 years and 12 years should have “limited heading in practice; maximum of 30 minutes of heading training per week, with no more than 15-20 headers per player, per week.”11 US Soccer Federation officials acknowledged the limitations in the current science regarding heading in young soccer players but chose to err on the side of caution until further empirical evidence regarding the risks associated with repetitive heading is available.
The US Soccer Federation is also exploring other ways to reduce the incidence rate of concussions, including ensuring that the age-appropriate sized ball is used in practice and play, possible rule changes, evaluation of different playing surfaces, and equipment usage. To date, there is no strong evidence to support the use of mouth guards or helmets to reduce concussions in soccer. Additionally, the current data about the value of head impact sensors in soccer has not supported its widespread use.
Continue to: Finally, the issue of the prevalence...
Finally, the issue of the prevalence of chronic traumatic encephalopathy (CTE) in soccer players is beyond the scope of this article. The expert opinion from the 2017 US Soccer Federation, Major League Soccer (MLS), and National Women’s Soccer League (NWSL) conference concluded, “At present, no data exist that support that soccer participation is a risk factor for the development of neurodegenerative disease. Similarly, at this time, consistent with evidence discussed in the Berlin Concussion in Sport Group (CISG) Consensus Conference, our review suggests no causal relationship has been demonstrated between soccer and CTE pathology.”12
The more we know about concussions, both in general and those sustained during soccer play, the better we are able to diagnose and manage these injuries in our athletes. An important step is creating evidence-based protocols that evolve as our knowledge of concussions does as well. In April 2017, the US Soccer Federation, MLS, and the NWSL held a joint summit entitled, “Head Injury in Soccer: From Science to the Field” to address the current evidence-based science of concussions in soccer.8 An article discussing the findings of this meeting is forthcoming and will undoubtedly guide further development of concussion protocols for soccer players of all ages.
HEART
The physiologic demands of soccer place considerable stress on the cardiovascular system. Participation in training and competition is characterized by a combination of aerobic and anaerobic physiology with the typical athlete covering approximately 10 km over the course of the 90-minute match. The primary role of the heart and blood vessels is to supply the exercising skeletal muscle with oxygen and energy substrate and to clear the byproducts of metabolism. Among healthy athletes without cardiovascular disease, these processes are typically well tolerated and may be associated with beneficial cardiovascular adaptations over time. However, competitive soccer players are not completely immune to cardiovascular disease. Athletes across the age and competition spectrum may develop symptoms suggestive of underlying cardiovascular disease during play including exertional chest pain, inappropriate shortness of breath, palpitations, and syncope. These athletes require timely clinical evaluation. In extremely rare but high visibility cases, competitive soccer players may succumb to cardiac arrest on the pitch, underscoring the need for comprehensive emergency action plans (EAPs). We provide the practicing clinician with an overview of cardiovascular issues relevant to the competitive soccer athlete.
CARDIOVASCULAR ADAPTATIONS TO SPORT
The pressure (ie, repetitive surges in systemic blood pressure) and volume (ie, sustained increases in high cardiac output) challenges inherent in soccer participation place stress on the cardiovascular system. Healthy athletes across the age spectrum typically tolerate the hemodynamic stressors of participation without issues. Athletes that engage in training and competition over months to years often develop beneficial adaptations of the cardiovascular system that enhance on-field performance and contribute to optimal long-term health. Detailed discussion of how the heart and blood vessels respond to exercise training is beyond the scope of this article, but the interested reader is referred to several prior publications.13,14 In brief, the heart of the healthy soccer athlete demonstrates the balanced mild chamber dilation and wall thickening characteristic of left ventricular eccentric remodeling. This form of exercise-induced cardiac remodeling facilitates maintenance of high stroke volume during exercise with minimal increases in cardiac work. In parallel, routine aerobic exercise training confers favorable changes in the systemic arterial system, which leads to reductions in age-associated ventricular stiffening and maintenance of healthy low blood pressure. It must be emphasized that the healthy heart muscle dilation and thickening that develop in response to sports participation, regardless of age, ethnicity, or gender, are relatively mild and should not be confused with common forms of heart muscle disease that may be seen in athletes at risk for adverse outcomes. In some situations, consultation with an extreme sports cardiologist may be required to differentiate exercise-induced remodeling from over heart muscle pathology.15
Continue to: THE SYMPTOMATIC ATHLETE...
THE SYMPTOMATIC ATHLETE
Any athlete presenting with symptoms suggestive of underlying cardiovascular disease should be withheld from training and competition until a comprehensive clinical evaluation has been completed. Common manifestations of underlying heart disease that occur in soccer players include exertional chest pain/pressure/tightness, shortness of breath that is out of proportion to workload, palpitations or the perception of irregular cardiac activity, and syncope. Chest discomfort, inappropriate shortness of breath, and palpitations that occur during training or competition should be managed with immediate removal from the playing field and prompt medical assessment. In many cases, thorough evaluation will involve collaboration between sports medicine and sports cardiology providers. Evaluations must be individualized on a case-by-case basis as tailored to the athlete’s presenting chief complaint and prior medical history. Most of these assessments will include a detailed medical history and physical examination, a 12-lead electrocardiogram (ECG), provocative exercise testing in a controlled environment, noninvasive cardiac imaging, and in some cases ambulatory rhythm monitoring. Uniformly, the athlete should be withheld from further training and competition until high-risk cardiovascular disease has been excluded. We refer the interested reader to a comprehensive discussion of symptom-based assessment of the athlete with suspected cardiovascular disease.16
Syncope (sudden and abrupt loss of consciousness with spontaneous neurologic recovery) is common among trained athletes. The vast majority of syncope is caused by “neurocardiogenic” mechanisms and carries a benign prognosis. Benign neurally-mediated syncope most often occurs outside of training and competition among athletes with heightened vagal tone and a predisposed susceptibility to triggers including pain, anxiety, emotional stimulation, and sudden postural change. Athletes who experience neurally-mediated syncope outside of training and competition routinely report a pre-event prodrome or aura that permits them to lower themselves to the ground, thereby avoiding injury. A distinct, but similarly benign and common, form of neurally-mediated fainting is post-exertional syncope. Here, fainting occurs within seconds of abrupt termination of exercise due to a rapid reduction cardiac preload and corollary cerebral blood supply. When either form of neurally-mediated syncope is suggested by a comprehensive medical history, normal physical examination, and a normal 12- lead ECG, further evaluation is unnecessary. However, the athlete and their coaching staff should be educated about avoidance tactics including hydration, dietary sodium supplementation, and avoidance of abrupt exercise termination as neutrally-mediated syncope tends to be recurrent without such measures. Fainting episodes that occur during training or competition that are not clearly post-exertional should be considered a medical emergency and should prompt comprehensive evaluation by a qualified cardiovascular specialist. Working closely with team physicians and athletic training staff, it is the responsibility of sports cardiologists to exclude potentially life-threatening forms of electrical, muscular, coronary, and valvular heart disease. Ideally, this evaluation should be conducted rapidly to avoid unnecessary delays in return to play.
CARDIAC ARREST AND SUDDEN DEATH
Numerous high-visibility cases of cardiac arrest on the soccer pitch have alerted the sporting community to the potential for these rare and potentially tragic events. Definitive incidence statistics defining the risk of cardiac arrest among soccer players are lacking. Data from the NCAA database suggest a sudden death incidence rate among collegiate male soccer athletes of approximately 1:24,000 athlete years.8 Similar data documenting incidence among female athletes and among those at lower (ie, youth level and high school) and higher (ie, professional) levels of play are unavailable. Underlying cardiovascular disease in the forms of heart muscle abnormalities (ie, genetic and acquired cardiomyopathy), coronary artery abnormalities (ie, genetic coronary anomalies and atherosclerotic coronary disease), valvular heart problems (ie, congenitally malformed aortic valves), and primary disturbances of the cardiac electrical system (ie, Wolf-Parkinson-White syndrome, long QT syndrome, etc.) explain a substantial percentage of on-pitch cardiac arrest. However, it is increasingly recognized that a significant minority of sudden cardiac deaths among athletes occur in the absence of attributable cardiovascular abnormality.17 Such cases, often referred to as “sudden unexplained death,” present unique challenges in the context of pre-participation screening, as they are undetectable and thus unpredictable.
Reduction of cardiac arrest and sudden death may best be accomplished through a combination of focused pre-participation screening and the development and implementation of a comprehensive EAP. Pre-participation involves the performance of a battery of tests prior to training and competition that are geared toward the detection of occult high-risk cardiovascular disease. Recommendations regarding pre-participation screening vary both across and within countries. Current US recommendations call for a focused medical history and physical examination prior to training and competition with consideration of the addition of a 12-lead ECG on a local level based on expertise and available resources.18 Conversely, current European guidelines including those endorsed by FIFA, suggest routine inclusion of a 12-lead ECG and in some cases, a transthoracic ECG.19 It must be emphasized that no screening approach has been confirmed to reduce the incidence of sudden death, and the decision to extend screening beyond the medical history and physical examination to include a 12-lead ECG or echocardiogram may come at the cost of increased false positive testing. In practice, decisions about how and when to screen are ideally made at a local level after consideration of medical and financial resources.20
Continue to: Even the most comprehensive approach...
Even the most comprehensive approach to screening and evaluation of symptomatic athletes will not completely eliminate on-pitch cardiac arrest. Thus, all stakeholders that engage in the oversight of organized soccer must be committed to the development and implementation of an EAP.21 Key components of an effective EAP include the training of coaching staff, athletic trainers, and players in basic cardiopulmonary resuscitation, access to and training in the use of automated external defibrillators, and a triage/transport protocol that ensures timely access to advanced cardiac life support. Much like screening, emergency action planning involving these key core components must be developed and tailored locally. In the era of contemporary organized athletics, the absence of an EAP at any level of competition, from youth to professional leagues, is unacceptable. Effective EAPs must be developed, documented, and rehearsed at regular intervals. For the health and safety of competitive soccer players, as well as coaching staff and spectators, these steps are of critical importance.
HEAT
Heat-related illnesses can be serious and, at times, even life threatening. It is important for athletic staff and athletes to be well versed in the prevention, signs and symptoms, and treatment of heat-related illnesses in order to prevent serious and lasting injury. We aim to educate physicians about the prevention, recognition, and management of heat-related injury, and stress the importance of similarly educating athletes and coaching staff.
Exertional heat illnesses most often occur at temperatures >86°F, however they can occur at any temperature with heavy exertion.22 Signs and symptoms can be nonspecific early on, including weakness, fatigue, headache, nausea, and dizziness. Later signs can include imbalance, altered mentation, confusion, and behavior that is out of character such as irritability or aggression.23 It is easy to see how the later signs can be confused for concussion in the right context. We cover the recognition and treatment of two common and serious heat-related illnesses: heat exhaustion and exertional heat stroke (EHS).
HEAT EXHAUSTION
Heat exhaustion occurs when an athlete cannot continue to exercise due to weakness and fatigue. While the exact mechanism is not well understood, it has been established that the combined effect of heat and dehydration have been proven to decrease exercise capacity and performance to a greater degree than either alone. The heat created by the body during exercise is 15 to 20 times greater than when at rest, and can increase core body temperature by 1°C every 5 minutes if no heat is lost, such as through sweating.24 Additionally, when fluid deficits reach >3% to 5% of total body water, sweat production and skin blood flow decline, blunting the ability for the body to cool itself and causing progressive elevation of core body temperature if the athlete continues exerting him or herself. When fluid deficits reach 6% to 10%, cardiac output, sweat, and muscle blood flow decrease, likely leading to the symptoms seen with heat exhaustion: weakness, profound fatigue, and occasionally confusion and disorientation. Athletes with suspected heat exhaustion should be moved to a cooler area, laid down with legs elevated, and orally rehydrated. If they do not improve with oral rehydration, they may require intravenous fluids. The diagnosis of heat exhaustion hinges on a rectal temperature of <104°F; if >104°F the athlete should be presumed to have heat stroke, which will be addressed in the following paragraphs. Players can be cleared to return to play in mild cases within 24 to 48 hours with gradual increases in exercise intensity.24
EXERTIONAL HEAT STROKE
EHS occurs when the body can no longer regulate the core body temperature and it rises to upwards of 104°F. In EHS, elevated core body temperature is associated with evidence of end organ dysfunction. The most easily identified on the playing field is likely central nervous system dysfunction, including irritability, confusion, irrational behavior, lethargy, dizziness, confusion, and even loss of consciousness. Temperature should be measured with rectal temperature only, as other methods of measurement have been shown to be consistently inaccurate.22 Heat stroke can be confused with exertional hyponatremia, heat exhaustion, or concussion, especially when core body temperature cannot be determined. However, EHS should always be the presumed cause of altered mentation when no rectal temperature is available because rapid cooling is critical to minimizing lasting effects. Morbidity and mortality are directly related to the length of time required to cool the athlete under 40°C (104°F).24 Cooling should be completed on site prior to transport to a medical facility and is best achieved with submersion in an ice bath (ie, a kiddie pool or soaking tub full of ice and water).22,25 If an ice bath is not available, ice bags should be applied to the neck axilla and groin and exchanged for fresh bags every 2 to 3 minutes.22 Ice bags have been shown to be inferior to whole body cooling, only cooling the athlete .04°C to .08°C/min compared to .15°C to .24°C/min with the ice bath.24 All other tests should be delayed until cooling is achieved, unless they can be completed while cooling the athlete. The athlete can be removed from the ice bath once rectal temperatures reach <101°F to 102°F.23 If the athlete returns to baseline after cooling, transportation to a medical facility may not be necessary. However, they should refrain from physical activity and heat exposure for at least 7 days and should be evaluated by a physician at that time. If all labs are normal and the athlete is asymptomatic, they can start progressive return to play under the direction of an athletic trainer or a sports medicine physician.23
Continue to: HEAT-RELATED ILLNESS...
HEAT-RELATED ILLNESS
It is impossible to predict exactly which athletes will be most at risk for heat-related illness, so it is important to have a high degree of suspicion when environmental conditions are right. Athletes with recent illness, fever, or lack of sleep are at higher risk. Additional intrinsic risk factors include low fitness level, obesity, and inadequate hydration. Athletes who are highly competitive or motivated can be more likely to push through the early signs of illness or be reluctant to report symptoms.23 Those with a history of exertional heat illness are more at risk for developing it again in the future.23
The extrinsic risk factors for the development of heat-related illness are much easier to identify and modify in order to keep athletes safe. High temperature and high humidity conditions, heavy sun exposure, and exposure to similar conditions the preceding day put athletes at risk for exertional heat illness. Risks are even greater when the exercise is prolonged or intense with few breaks and access to hydration is limited.23 Therefore, prevention of exertional heat illness is centered on these external risk factors.
Each team should have a heat policy as part of their EAP aimed at prevention and early recognition of heat-related illness. This policy should be shared with all athletes and coaches. The plan should be centered on acclimatization, activity modification, and early recognition and management as previously discussed. The US Soccer Federation “Recognize to Recover” Heat Guidelines suggest a 3-step process for appropriate activity modification:22
1. Find the wet bulb globe temperature, either using a wet bulb globe thermometer or the temperature and humidity (Figure 1).
2. Find your regional weather category on the map (Figure 2).
3. Find your alert level and work to rest ratio recommendations (Figure 3).
Scheduled hydration breaks should be given as listed in Figure 3. Breaks of 4 minutes should be given for each 30 minutes of continuous practice or play. In a regulation 90-minute match, a hydration break should be given at 30 and 75 minutes (with half time at 45 minutes) at minimum. Athletes should be educated about where hydration can be accessed, and given unlimited access to hydration even outside of planned breaks.22
Acclimatization to conditions is another integral part of preventing heat-related illness. It allows the body time to adapt to exercising in heat gradually, with a measured progression of exertion over the course of 10 to 14 days. The “Recognize to Recover” Heat Guidelines also provide guidance on acclimatization, and specifics can be found on the website.1 Generally speaking, the warmest part of the day, usually between 11 AM and 4 PM, should be avoided for all training sessions, and length of practice and exertion should be gradually increased over 2 weeks.22
In summary, appropriate acclimatization, hydration, activity modification, and education of athletes and staff are essential for the prevention of heat-related illness. Athletes and staff should understand the signs and symptoms of heat-related illness so that it can be recognized early and treated appropriately. If an athlete is altered in the heat and rectal temperature is >104°F or rectal temperature cannot be obtained, rapid cooling using an ice bath or ice bags is essential to prevent the morbidity and mortality associated with EHS. Above all, teams should have an explicit plan that includes protocols for acclimatization, activity modification, and all necessary equipment to prevent and treat heat-related illnesses should they occur, and ultimately keep athletes safe and healthy.
1. Langlois JA, Rutland-Brown W, Wald MM. The epidemiology and impact of traumatic brain Injury: a brief overview. J Head Trauma Rehabil. 2006;21(5):375-378.
2. The National Federation of State High School Associations. 2013-14 high school athletics participation survey. http://www.nfhs.org/ParticipationStatics/PDF/2013-14_Participation_Survey_PDF.pdf. Accessed August 6, 2018.
3. Youth Council. US Soccer Federation Web site. https://www.ussoccer.com/about/affiliates/youth-council. Accessed July 31, 2018.
4. Khodaee M, Currie DW, Asif IM, Comstock RD. Nine-year study of US high school soccer injuries: data from a national sports injury surveillance programme. Br J Sports Med. 2017;51(3):185-193. doi:10.1136/bjsports-2015-095946.
5. Schallmo MS, Weiner JA, Hsu WK. Sport and sex-specific trends in the epidemiology of concussions sustained by high school athletes. J Bone Joint Surg Am. 2017;99(15):1314-1320. doi:10.2106/JBJS.16.01573.
6. US Soccer Federation. U.S. Soccer’s comprehensive player health and safety program. Recognize to Recover Web site. http://www.recognizetorecover.org/#us-soccers-comprehensive-player-health-and-safety-program. Accessed July 31, 2018.
7. McCrory P, Meeuwisse W, Dvořák J, et al. Consensus statement on concussion in sport-the 5th international conference on concussion in sport held in Berlin, October 2016. Br J Sports Med. 2017;51(11):838-847. doi:10.1136/bjsports-2017-097699.
8. Harmon KG, Asif IM, Klossner D, Drezner JA. Incidence of sudden cardiac death in National Collegiate Athletic Association athletes. Circulation. 2011;123(15):1594-1600. doi:10.1161/CIRCULATIONAHA.110.004622.
9. Faude O, Rössler R, Junge A, et al. Head injuries in children’s football-results from two prospective cohort studies in four European countries. Scand J Med Sci Sports. 2017;27(12):1986-1992. doi:10.1111/sms.12839.
10. Comstock RD, Currie DW, Pierpoint LA, Grubenhoff JA, Fields SK. An evidence-based discussion of heading the ball and concussions in high school soccer. JAMA Pediatr. 2015;169(9):830-837. doi:10.1001/jamapediatrics.2015.1062.
11. Tarnutzer AA. Should heading be forbidden in children’s football? Sci Med Football. 2018;2(1):75-79.
12. US Soccer Federation. US Soccer, NWSL and MLS to host “head injury in soccer; science to field”. https://www.ussoccer.com/stories/2017/04/18/17/35/20170418-news-us-soccer-nwsl-mls-host-head-injury-in-soccer-science-to-field. Published April 18, 2017. Accessed August 6, 2018.
13. Weiner RB, Baggish AL. Exercise-induced cardiac remodeling. Prog Cardiovasc Dis. 2012;54(5):380-386. doi:10.1016/j.pcad.2012.01.006.
14. Baggish AL, Wood MJ. Athlete's heart and cardiovascular care of the athlete: scientific and clinical update. Circulation. 2011;123(23):2723-2735. doi:10.1161/CIRCULATIONAHA.110.981571.
15. Kim JH, Baggish AL. Differentiating exercise-induced cardiac adaptations from cardiac pathology: the "Grey Zone" of clinical uncertainty. Can J Cardiol. 2016;32(4):429-437. doi:10.1016/j.cjca.2015.11.025.
16. Baggish AL, Battle RW, Beckerman JG, et al; ACC’s Sports and Exercise Council Leadership Group. Sports cardiology: core curriculum for providing cardiovascular care to competitive athletes and highly active people. J Am Coll Cardiol. 2017;70(15):1902-1918. doi:10.1016/j.jacc.2017.08.055.
17. Harmon KG, Asif IM, Maleszewski JJ, et al. Incidence, cause, and comparative frequency of sudden cardiac death in National Collegiate Athletic Association athletes: a decade in review. Circulation. 2015;132(1):10-19. doi:10.1161/CIRCULATIONAHA.115.015431.
18. Maron BJ, Levine BD, Washington RL, Baggish AL, Kovacs RJ, Maron MS. Eligibility and disqualification recommendations for competitive athletes with cardiovascular abnormalities: task force 2: preparticipation screening for cardiovascular disease in competitive athletes: a scientific statement from the American Heart Association and American College of Cardiology. J Am Coll Cardiol. 2015;66(21):2356-2361. doi:10.1016/j.jacc.2015.09.034.
19. Corrado D, Pelliccia A, Bjørnstad HH, et al. Cardiovascular pre-participation screening of young competitive athletes for prevention of sudden death: proposal for a common European protocol. Consensus Statement of the Study Group of Sport Cardiology of the Working Group of Cardiac Rehabilitation and Exercise Physiology and the Working Group of Myocardial and Pericardial Diseases of the European Society of Cardiology. Eur Heart J. 2005;26(5):516-524.
20. Baggish AL, Kovacs RJ. Preparticipation cardiovascular screening: clinical partnership is the only certainty. Br J Sports Med. 2017;51(3):150-151. doi:10.1136/bjsports-2016-096954.
21. Hainline B, Drezner J, Baggish A, et al. Interassociation consensus statement on cardiovascular care of college student-athletes. J Athl Train. 2016;51(4):344-357. doi:10.4085/j.jacc.2016.03.527.
22. US Soccer Federation. Environmental conditions. Recognize to Recover Web site. http://www.recognizetorecover.org/environmental/#environmental-conditions. Accessed April 15, 2018.
23. Korey Stringer Institute. Emergency conditions: heat illnesses. University of Connecticut Web site. https://ksi.uconn.edu/. Accessed April 15, 2018.
24. American College of Sports Medicine, Armstrong LE, Casa DJ, et al. American College of Sports Medicine position stand. Exertional heat illness during training and competition. Med Sci Sports Exerc. 2007;39(3):556-572.
25. Belval LN, Casa DJ, Adams WM, et al. Consensus statement- prehospital care of exertional heat stroke. Prehosp Emerg Care. 2018;22(3):392-397. doi:10.1080/10903127.2017.1392666.
1. Langlois JA, Rutland-Brown W, Wald MM. The epidemiology and impact of traumatic brain Injury: a brief overview. J Head Trauma Rehabil. 2006;21(5):375-378.
2. The National Federation of State High School Associations. 2013-14 high school athletics participation survey. http://www.nfhs.org/ParticipationStatics/PDF/2013-14_Participation_Survey_PDF.pdf. Accessed August 6, 2018.
3. Youth Council. US Soccer Federation Web site. https://www.ussoccer.com/about/affiliates/youth-council. Accessed July 31, 2018.
4. Khodaee M, Currie DW, Asif IM, Comstock RD. Nine-year study of US high school soccer injuries: data from a national sports injury surveillance programme. Br J Sports Med. 2017;51(3):185-193. doi:10.1136/bjsports-2015-095946.
5. Schallmo MS, Weiner JA, Hsu WK. Sport and sex-specific trends in the epidemiology of concussions sustained by high school athletes. J Bone Joint Surg Am. 2017;99(15):1314-1320. doi:10.2106/JBJS.16.01573.
6. US Soccer Federation. U.S. Soccer’s comprehensive player health and safety program. Recognize to Recover Web site. http://www.recognizetorecover.org/#us-soccers-comprehensive-player-health-and-safety-program. Accessed July 31, 2018.
7. McCrory P, Meeuwisse W, Dvořák J, et al. Consensus statement on concussion in sport-the 5th international conference on concussion in sport held in Berlin, October 2016. Br J Sports Med. 2017;51(11):838-847. doi:10.1136/bjsports-2017-097699.
8. Harmon KG, Asif IM, Klossner D, Drezner JA. Incidence of sudden cardiac death in National Collegiate Athletic Association athletes. Circulation. 2011;123(15):1594-1600. doi:10.1161/CIRCULATIONAHA.110.004622.
9. Faude O, Rössler R, Junge A, et al. Head injuries in children’s football-results from two prospective cohort studies in four European countries. Scand J Med Sci Sports. 2017;27(12):1986-1992. doi:10.1111/sms.12839.
10. Comstock RD, Currie DW, Pierpoint LA, Grubenhoff JA, Fields SK. An evidence-based discussion of heading the ball and concussions in high school soccer. JAMA Pediatr. 2015;169(9):830-837. doi:10.1001/jamapediatrics.2015.1062.
11. Tarnutzer AA. Should heading be forbidden in children’s football? Sci Med Football. 2018;2(1):75-79.
12. US Soccer Federation. US Soccer, NWSL and MLS to host “head injury in soccer; science to field”. https://www.ussoccer.com/stories/2017/04/18/17/35/20170418-news-us-soccer-nwsl-mls-host-head-injury-in-soccer-science-to-field. Published April 18, 2017. Accessed August 6, 2018.
13. Weiner RB, Baggish AL. Exercise-induced cardiac remodeling. Prog Cardiovasc Dis. 2012;54(5):380-386. doi:10.1016/j.pcad.2012.01.006.
14. Baggish AL, Wood MJ. Athlete's heart and cardiovascular care of the athlete: scientific and clinical update. Circulation. 2011;123(23):2723-2735. doi:10.1161/CIRCULATIONAHA.110.981571.
15. Kim JH, Baggish AL. Differentiating exercise-induced cardiac adaptations from cardiac pathology: the "Grey Zone" of clinical uncertainty. Can J Cardiol. 2016;32(4):429-437. doi:10.1016/j.cjca.2015.11.025.
16. Baggish AL, Battle RW, Beckerman JG, et al; ACC’s Sports and Exercise Council Leadership Group. Sports cardiology: core curriculum for providing cardiovascular care to competitive athletes and highly active people. J Am Coll Cardiol. 2017;70(15):1902-1918. doi:10.1016/j.jacc.2017.08.055.
17. Harmon KG, Asif IM, Maleszewski JJ, et al. Incidence, cause, and comparative frequency of sudden cardiac death in National Collegiate Athletic Association athletes: a decade in review. Circulation. 2015;132(1):10-19. doi:10.1161/CIRCULATIONAHA.115.015431.
18. Maron BJ, Levine BD, Washington RL, Baggish AL, Kovacs RJ, Maron MS. Eligibility and disqualification recommendations for competitive athletes with cardiovascular abnormalities: task force 2: preparticipation screening for cardiovascular disease in competitive athletes: a scientific statement from the American Heart Association and American College of Cardiology. J Am Coll Cardiol. 2015;66(21):2356-2361. doi:10.1016/j.jacc.2015.09.034.
19. Corrado D, Pelliccia A, Bjørnstad HH, et al. Cardiovascular pre-participation screening of young competitive athletes for prevention of sudden death: proposal for a common European protocol. Consensus Statement of the Study Group of Sport Cardiology of the Working Group of Cardiac Rehabilitation and Exercise Physiology and the Working Group of Myocardial and Pericardial Diseases of the European Society of Cardiology. Eur Heart J. 2005;26(5):516-524.
20. Baggish AL, Kovacs RJ. Preparticipation cardiovascular screening: clinical partnership is the only certainty. Br J Sports Med. 2017;51(3):150-151. doi:10.1136/bjsports-2016-096954.
21. Hainline B, Drezner J, Baggish A, et al. Interassociation consensus statement on cardiovascular care of college student-athletes. J Athl Train. 2016;51(4):344-357. doi:10.4085/j.jacc.2016.03.527.
22. US Soccer Federation. Environmental conditions. Recognize to Recover Web site. http://www.recognizetorecover.org/environmental/#environmental-conditions. Accessed April 15, 2018.
23. Korey Stringer Institute. Emergency conditions: heat illnesses. University of Connecticut Web site. https://ksi.uconn.edu/. Accessed April 15, 2018.
24. American College of Sports Medicine, Armstrong LE, Casa DJ, et al. American College of Sports Medicine position stand. Exertional heat illness during training and competition. Med Sci Sports Exerc. 2007;39(3):556-572.
25. Belval LN, Casa DJ, Adams WM, et al. Consensus statement- prehospital care of exertional heat stroke. Prehosp Emerg Care. 2018;22(3):392-397. doi:10.1080/10903127.2017.1392666.
TAKE-HOME POINTS
- Current concussion education programs such as “Recognize to Recover” aim to increase self-reporting of concussion symptoms by players, and recognition and appropriate evaluation by medical and coaching staff.
- Athletes who develop symptoms suggestive of underlying cardiovascular disease during play, including exertional chest pain, inappropriate shortness of breath, palpitations, and syncope should be withheld from play until they can be evaluated by a qualified medical professional.
- Key components of an effective EAP include the training of coaching staff, athletic trainers, and players in basic cardiopulmonary resuscitation, access to and training in the use of automated external defibrillators, and a triage/transport protocol that ensures timely access to advanced cardiac life support.
- Exertional heat stroke should always be the presumed cause of altered mentation when no rectal temperature is available because rapid cooling is critical to minimizing lasting effects.
- Prevention of exertional heat illness should center around appropriate acclimatization, access to adequate hydration and scheduled hydration breaks, and avoiding exertion all together when conditions are too dangerous.
Knee Injuries in Elite Level Soccer Players
ABSTRACT
As one of the most popular sports in the world, soccer injury rates involving the knee continue to rise. An alarming trend of knee injuries, including increased anterior cruciate ligament ruptures, underscores the need to review our current understanding of these injuries in soccer players. This article includes a critical review of the epidemiology of knee injuries in soccer, anterior cruciate ligament and other ligamentous injuries, cartilage and meniscal injury, post-traumatic osteoarthritis, as well as current prevention initiatives.
Continue to: EPIDEMIOLOGY...
EPIDEMIOLOGY
There are currently 28 players on each of the Major League Soccer (MLS) teams, and during the 2013 to 2014 academic year, the National Federation of State High School Associations (NFHS) reported that 417,419 boys and 374,564 girls played high school soccer and the National Collegiate Athletic Association (NCAA) reported that 23,602 males and 26,358 females played collegiate soccer.5 As such, knee injuries in this population are a major concern for those involved in sports medicine. Several injuries occurring during soccer involve the lower extremity, particularly the knee.1 In fact, multiple reports estimate that up to 17.6% of soccer-related injuries presenting to the emergency room involved the knee.1,6-9 The majority of these injuries are noncontact injuries, although contact injuries do still occur.10,11
Risk factors for injuries in soccer may be non-modifiable (such as age and gender) and modifiable (such as level of conditioning, force, balance, and flexibility). Inadequate lower motor coordination may result in injury in the adolescent population, and advanced age >28 years in males and >25 years in females is considered as a high-risk factor for injury.12,13 Importantly, gender and age have been reported to play a significant role as risk factors for ACL injury.6 In fact, female players have a 3 to 5 times higher risk of significant knee injury, including ACL injuries, than male players.4,14-16 Preventative programs such as the FIFA 11+ program have been set forth to augment conditioning as part of managing the modifiable risk factors.
Like American football, playing on artificial turf has been questioned as a contributor to injury compared to playing on natural grass.17,18 In recent years, newer generations of artificial turf have been developed to more closely replicate the characteristics of natural grass. Meyers19 compared the incidence, mechanisms, and severity of match-related collegiate men’s soccer injuries on artificial turf and those on natural grass and demonstrated no significant difference in knee injuries between the 2 surfaces. This finding was consistent with previous studies that reported no difference in the incidence of knee injuries on either surface in women’s collegiate and elite-level soccer.15,20,21
Continue to: ACL INJURIES...
ACL INJURIES
ACL injuries are life-changing events that can significantly affect the career of a soccer athlete. As a major stabilizer of the knee, the ACL primarily prevents anterior tibial translation with the anteromedial bundle and secondarily resists tibial rotation with the posterolateral bundle. The ligament takes origin from the posteromedial aspect of the lateral femoral condyle and inserts anterior to the tibial intercondylar eminence. Grading of ACL injuries is based on the Lachman test, which is performed between 20°and 30° of knee flexion and measures the amount of anterior tibial translation relative to the femur (A = firm endpoint, B = no endpoint; grade I: 3-5 mm, grade II (A/B): 5-10 mm, grade III (A/B): >10 mm).
ACL injury may occur via contact or noncontact mechanisms. Noncontact mechanisms of ACL injury in soccer athletes contribute to about 85% of injuries.6,22-25 Typical noncontact mechanism of injury involves a forceful valgus collapse with the knee near full extension and combined external or internal rotation of the tibia23,26 (Figure 1). This on-field scenario generally involves cutting and torsional movement, as well as landing after a jump, particularly in 1-legged stance. Similarly, a disturbance in balance caused by the opponent may incite a noncontact mechanism resulting in ACL rupture.6,27 Video analyses of professional soccer players have also demonstrated a higher risk of noncontact ACL injury within the first 9 minutes of the match, with the most common playing situation resulting in injury being pressing, followed by kicking and heading.24,25,28 Contact mechanisms resulting in ACL injury, however, are not an uncommon occurrence in soccer players with higher risk for certain positions. Brophy and colleagues29 reviewed ACL injuries in professional and collegiate soccer players and reported a higher risk of ACL injury during defending and tackling. Similarly, Faude and colleagues30 found the risk of injury to be higher in defenders and strikers than in goalkeepers and midfielders.

Female athletes participating in elite-level athletics, especially soccer, represent a high-risk group for ACL injury. In fact, these soccer athletes experience ACL injury at an incidence 3 times higher than that in male athletes.31-35 Female soccer athletes may also be at risk for reinjury to the ACL and contralateral ACL injury. Female gender, in combination with participation in soccer, thus represents a high-risk group for ACL tear in athletics. Allen and colleagues36 retrospectively reviewed 180 female patients who had undergone ACL reconstruction (ACLR) (90 soccer players and 90 non-soccer players) over a mean period of 68.8 months. In their series, soccer players sustained significantly more ACL injuries than non-soccer players, including graft failures (11% vs 1%) and contralateral ACL tears (17% vs 4%).
ACLR is the gold standard treatment for elite soccer athletes. A recent survey of MLS team orthopedic surgeons revealed several important details regarding decision-making in ACLR in this population. From a technical standpoint, the vast majority of surgeons used a single incision, arthroscopically assisted, single-bundle reconstruction (91%). Femoral tunnel drilling was almost equally split between transtibial (51%) and use of an accessory medial portal (46%). Bone-patella-tendon-bone (BPTB) autograft was the most preferred graft choice (68%), and quadriceps tendon autograft was the least preferred. The majority of surgeons preformed ACLR within 4 weeks and permitted return to sport (RTS) without restrictions at 6 to 8 months.37
Continue to: There is a scarcity of literature regarding...
There is a scarcity of literature regarding the use of soft tissue and BPTB allografts in soccer athletes. However, one study reported no difference in patient-reported outcomes and return to preinjury level of activity (including soccer) with the use of either autograft or allograft BPTB in ACLR.38 The authors’ preference was to avoid the use of allograft in elite-level soccer athletes as the reported rate of ACL re-tear was 4 to 8 times higher than that with autograft reconstruction, as shown in athletes and military personnel.39,40 BPTB autograft and hamstring autograft (semitendinosus and/or gracilis) are common graft choices for soccer athletes. Gifstad and colleagues41 compared BPTB autograft and hamstring autograft in 45,998 primary ACLRs performed in Scandinavia. Although the cohort included, but was not limited to, soccer players, the authors reported an overall risk of revision that was significantly lower in the BPTB autograft group than in the hamstring autograft group (hazard ratio, 0.63; 95% confidence interval, 0.53-0.74).41 Mohammadi and colleagues42 prospectively compared the functional outcomes of 42 competitive soccer players who underwent ACLR with BPTB autograft vs those who underwent ACLR with hamstring autograft at the time of RTS. Players who had undergone ACLR with hamstring autograft demonstrated greater quadriceps torque, as well as better performance with triple-hop, crossover-hop, and jump-landing tests; however, both groups demonstrated similar hamstring torque and performance in 2 other hop tests.42 In the authors’ opinion, there may be a concern regarding the use of hamstring autograft in elite soccer players considering that hamstring strains are extremely common in this athletic population; however, further research would be necessary to elucidate whether this is an actual or a theoretical risk. Although not yet studied in elite-level athletes, early clinical results of ACL repair with suture augmentation show promise for certain injury patterns. These include proximal femoral ACL avulsion injuries (Sherman type 1) of excellent tissue quality that have the ability to be reapproximated to the femoral origin43 (Figures 2A, 2B). In a recent series,43-45 early clinical outcomes were found to be excellent and maintained at midterm follow-up.
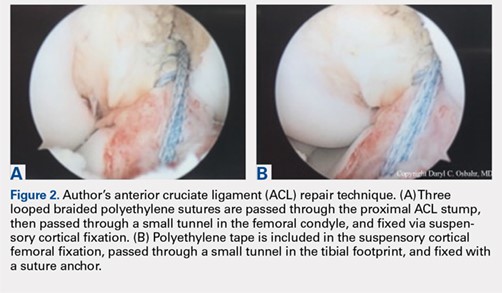
In the NCAA soccer athletes, an overall RTS rate of 85% has been reported in those undergoing ACLR, with a significantly higher rate observed in scholarship versus non-scholarship athletes.46 Howard and colleagues46 reported median time to unrestricted game play of 6.1 months, with 75% returning to the same or higher level position on the depth chart. Among their studied collegiate soccer athletes, 32% reported continued participation in soccer on some level after college (recreational, semiprofessional, or professional).46 RTS rates for MLS soccer players have also been reported to be high, ranging from 74% to 77%, most of them returning within the following season at 10 ± 2.8 months.47,48 These findings were consistent with the RTS rate of 72% reported by the Multicenter Orthopaedic Outcomes Network (MOON) group, which analyzed 100 female and male soccer players undergoing ACLR at a minimum 7-year follow-up. In this series, Brophy and colleagues29,49 reported an RTS at 12 ± 14.3 months, with 85% returning to the same or a higher level of play prior to their injury. Erickson and colleagues47 analyzed a series of 57 ACLRs performed in MLS athletes and reported no significant difference in preinjury or postoperative performance, or between cases and uninjured controls. Arundale and colleagues48 demonstrated no significantly increased risk of lower extremity injury in MLS athletes after ACLR, but the athletes had significantly shorter careers than their uninjured counterparts. Curiously, RTS rates for European professional soccer athletes have been reported to be substantially higher at 95% to 97%.50,51 Although we can only speculate the reasons for such a discrepancy, the difference in RTS rates for similar athletes highlights a need for objective criteria to determine and report RTS rates, while also providing guidelines to prevent reinjury. Such a consensus among orthopedists is not yet present in the literature.
Soccer players and adolescent age in combination have been shown to portend a 3-fold increased risk of revision surgery for ACL failure in a cohort of 16,930 patients from the Swedish National Knee Ligament Register.52 Published data regarding ACL failure and management of revision ACLR in elite-level soccer athletes are currently lacking. However, low failure rates of 3% to 10% requiring revision reconstruction have been reported.47,49 Arundale and colleagues48 reported 2 incidences of players with ACL graft failures, 1 BPTB autograft and 1 BPTB allograft, both of whom were able to return to MLS after revision ACLR. It is the authors’ preference to use ipsilateral hamstring autograft or contralateral BPTB autograft when an ACL revision reconstruction is required.
Continue to: OTHER LIGAMENTOUS INJURIES...
OTHER LIGAMENTOUS INJURIES
The majority of research efforts regarding knee injuries in this population are focused on the ACL. Correspondingly, literature regarding injury to the collateral ligaments and the posterior cruciate ligament (PCL) in soccer players is sparse. The lateral collateral ligament (LCL) and the medial collateral ligament (MCL) play important roles as primary stabilizers to varus and valgus forces, respectively. The PCL is the primary posterior stabilizer of the knee, preventing posterior translation of the tibia. Injury to these structures may result in significant time lost from soccer and risk of reinjury.53,54
The MCL is the one of the most commonly injured ligaments in sports, including soccer.53,55 The injury mechanism generally involves contact with a resulting valgus force applied to the knee.55 Grading of MCL injuries is based on the amount of medial joint gapping with applied valgus force during examination (grade I: <5 mm, grade II: 5-10 mm, grade III: >10 mm). Kramer and colleagues53 reviewed collateral ligament injuries in the adolescent population and found that MCL injuries occurred 4 times more often than LCL injuries and about 25% were grade III injuries, most commonly occurring in American football and soccer players. Soccer also touts the highest sport-specific MCL injury rate for high school and collegiate athletics, particularly for female NCAA soccer players.56 At the professional level, Lundblad and colleagues55 reported 346 MCL injuries in 27 European teams over an 11-year period, of which 70% were contact-related, and the average time-off from soccer was 28 days.
Most surgeons treat isolated MCL injuries nonoperatively, regardless of grade.57,58 This includes activity modification, use of a hinged knee brace, quadriceps strengthening, and progressive return to play. The literature currently lacks substantial data to guide MCL injury management, specifically in elite soccer athletes. In our experience, grade I injuries are managed nonoperatively and RTS is allowed at 4 to 6 weeks. Grade II injuries are also managed nonoperatively and RTS is allowed at 6 to 8 weeks. Grade III injuries are generally allowed RTS at 8 to 12 weeks and may be considered for surgery in the context of concomitant injuries (eg, posteromedial capsular injury, multiligamentous knee injuries, and meniscal injuries). In some athletes, we consider using a varus unloader brace to help maximize decreased stress on the MCL while still allowing the athlete to be fully weight-bearing. We have found it less ideal to limit weight-bearing in elite athletes, which may negatively affect overall lower extremity neuromuscular proprioception and potentially prolong a safe return to play. Some athletes may experience prolonged soreness at the MCL femoral or tibial attachment despite being able to return to play. It is important to counsel athletes about these prolonged symptoms to set expectations, as this may even occur with grade I MCL injuries. Other rare instances where surgical management may be indicated include persistent pain and instability following nonoperative treatment of grade III injuries and highly displaced tibial avulsions of the ligament resulting in poor healing.59,60
Data regarding LCL injuries in soccer are extremely sparse. In our experience, treatment and RTS rates for isolated LCL injuries are similar to those for MCL injuries. However, it is worth noting that one-quarter of LCL injuries may occur in combination with injury to other posterolateral corner structures.53
PCL injuries are more commonly associated with vehicular trauma but have also been reported to occur in sports at a rate of 33% to 40%.61,62 The mechanism of injury in athletes generally involves a fall onto the hyperflexed knee with the foot in plantarflexion or a direct blow to the anterior tibia in a flexed knee.62,63 Classification of PCL injuries is based on posterior translation of the tibia relative to the femur with the knee flexed to 90°(grade I: 1-5 mm, grade II: 6-10 mm, grade III: >10 mm). In one cohort of 62 patients with isolated PCL injuries, soccer was found to be among the top 5 causes of injury.64 A Scandinavian review of 1287 patients who underwent PCL reconstruction found soccer to be the sport with the highest number of injuries (13.1%).65 The goalkeeper was most commonly subjected to this injury.62 Krutsch and colleagues54 compared PCL injuries in new, professional soccer players to those in players at the closest amateur level of play. In their series, 90% of PCL injuries occurred during preseason in players who were at a lower level of play in the previous season. This finding suggested that a rapid increase in training and playing intensity may have been a significant risk factor for PCL injury. Substantial literature supporting nonoperative or operative management of PCL injuries in soccer athletes is currently lacking. Historically, nonoperative treatment has been the initial management for isolated PCL injuries; however, surgical intervention has become increasingly used for both isolated and combined PCL injuries.66
Continue to: CARTILAGE AND MENISCAL INJURIES...
CARTILAGE AND MENISCAL INJURIES
The prevalence of osteoarthritis (OA) in retired soccer players is high.67,68 Articular cartilage degeneration with subsequent OA occurs in up to 32% of soccer players and ultimately leads to significant disability and retirement from the sport. High physical demands and concomitant knee injuries probably predispose to the development of posttraumatic OA.69-71
Several techniques addressing cartilage débridement or restoration have been reported, with successful RTS but with variable durability.72-75 Recently, Andrade and colleagues76 performed a systematic review of 217 articular cartilage defects in soccer players that were treated using restoration techniques, including chondroplasty, microfracture, autologous chondrocyte implantation (ACI), and osteochondral autograft. Although no superior technique could be ascertained, microfracture and osteochondral autograft procedures led to the quickest return to play, and ACI techniques enhanced long-standing clinical and functional results.76 More recently, osteochondral allograft transplantation has also been described with an 84% return to some level of activity (including soccer) and 60% of athletes returning to high-level sports participation at a mean follow-up of 4.5 years77 (Figures 3A-3C). Although chondroplasty may be successful and allow for a quicker return to play in some soccer players (return to play from 6-12 weeks), the authors believe that a strong cartilage scaffold repair strategy with early weight-bearing, including osteochondral autograft and allograft procedures (return to play from 6-9 months), must also be considered in focal chondral defects to optimize both short-term and potential long-term success.
Meniscal injuries are also prevalent in the soccer population, and consistent with ACL injuries, female players are at least twice as likely to sustain a meniscal tear.78,79 Meniscal damage can occur in isolation or in association with ACL rupture. Repair techniques should be strongly considered as chondral changes in the setting of meniscal deficiency are a significant short- and long-term concern for elite athletes. However, due to intrinsically poor healing potential, partial meniscectomy is unfortunately more often performed.79,80 In either case, meniscal deficiency is recognized as a precursor to the development of OA as meniscal functionality is lost and the articular cartilage is subjected to increased biomechanical loading.81,82 Nawabi and colleagues83 analyzed RTS in 90 professional soccer players following partial meniscectomy. Median RTS was at 7 weeks for lateral meniscectomies and at 5 weeks for medial meniscectomies. RTS probability was 5.99 times greater after medial meniscectomy at all time points. Lateral meniscectomies were associated with an increased risk of postoperative adverse events, reoperation, and a significantly lower rate of return to play.83 In the case of severe meniscal deficiency, particularly post-meniscectomy, meniscal allograft transplantation (MAT) may be considered. In a series of MATs in lower division Spanish players, 12/14 (85.7%) returned to play at an average of 7.6 months.84 A more recent series of professional players reported 9/12 (75%) RTS as professionals and 2/12 (17%) as semiprofessionals at an average of 10.5 months.85 The authors’ strong preference is to perform meniscus-saving procedures whenever possible. Due to the longer recovery and return to play associated with meniscus repair than partial meniscectomy, most of the soccer players will often prefer to proceed with partial meniscectomy. Despite the ultimate treatment, it is critical that the surgeon and the soccer player have an in-depth conversation concerning the risks and benefits for each procedure and individualize treatment to the individual soccer player accordingly.
Continue to: INJURY PREVENTION...
INJURY PREVENTION
Given the breadth and the prevalence of soccer-related injuries, the FIFA11+ program was developed in 2006 as an injury prevention measure (Figure 4). The warm-up program includes 15 structured exercises emphasizing core stabilization, thigh muscle training, proprioception, dynamic stabilization, and plyometric exercises. The routine is believed to be easily executed and effective at preventing the incidence of noncontact injuries.86,87 Recently, Sadigursky and colleagues1 performed a systematic review of randomized clinical trials examining the efficacy of FIFA11+. The authors reported a reduction in injuries by 30% and a relative risk of 0.70 for lower limb injuries, highlighting the significant preventative importance of the program.1 Post-training programs may also be beneficial as it has been shown that performing FIFA11+ both before and after training reduced overall injury rates in male, amateur soccer players.88 Regardless of the prevention program, it is critical that every league, team, medical team, and athlete have a thorough injury prevention strategy to help keep players healthy and not wait until they have instead sustained a significant injury.

CONCLUSION
Knee injuries are common in soccer, with an alarming number of ACL injuries, as well as other significant pathology. Understanding the unique epidemiology, risk factors, treatment, and injury prevention strategies is critically important in helping medical professionals provide care for all levels of elite soccer players.
1. Sadigursky D, Braid JA, De Lira DNL, Machado BAB, Carneiro RJF, Colavolpe PO. The FIFA 11+ injury prevention program for soccer players: a systematic review. BMC Sports Sci Med Rehabil. 2017;9:18. doi:10.1186/s13102-017-0083-z.
2. Junge A, Dvorak J. Soccer injuries: a review on incidence and prevention. Sports Med. 2004;34(13):929-938. doi:10.2165/00007256-200434130-00004.
3. Hootman JM, Dick R, Agel J. Epidemiology of collegiate injuries for 15 sports: summary and recommendations for injury prevention initiatives. J Athl Train. 2007;42(2):311-319.
4. Agel J, Rockwood T, Klossner D. Collegiate ACL Injury rates across 15 sports: National collegiate athletic association injury surveillance system data update (2004-2005 Through 2012-2013). Clin J Sport Med. 2016;26(6):518-523. doi:10.1097/JSM.0000000000000290.
5. Kerr ZY, Pierpoint LA, Currie DW, Wasserman EB, Comstock RD. Epidemiologic comparisons of soccer-related injuries presenting to emergency departments and reported within high school and collegiate settings. Inj Epidemiol. 2017;4(1):19. doi:10.1186/s40621-017-0116-9.
6. Volpi P, Bisciotti GN, Chamari K, Cena E, Carimati G, Bragazzi NL. Risk factors of anterior cruciate ligament injury in football players: a systematic review of the literature. Muscles Ligaments Tendons J. 2016;6(4):480-485. doi:10.11138/mltj/2016.6.4.480.
7. Smith NA, Chounthirath T, Xiang H. Soccer-related injuries treated in emergency departments: 1990-2014. Pediatrics. 2016;138(4). doi:10.1542/peds.2016-0346.
8. Leininger RE, Knox CL, Comstock RD. Epidemiology of 1.6 million pediatric soccer-related injuries presenting to US emergency departments from 1990 to 2003. Am J Sports Med. 2007;35(2):288-293. doi:10.1177/0363546506294060.
9. Adams AL, Schiff MA. Childhood soccer injuries treated in U.S. emergency departments. Acad Emerg Med. 2006;13(5):571-574. doi:10.1197/j.aem.2005.12.015.
10. Woods C, Hawkins R, Hulse M, Hodson A. The football association medical research programme: an audit of injuries in professional football-analysis of preseason injuries. Br J Sports Med. 2002;36(6):436-441. doi:10.1136/bjsm.36.6.436.
11. Chomiak J, Junge A, Peterson L, Dvorak J. Severe injuries in football players. Influencing factors. Am J Sports Med. 2000;28(5 Suppl):S58-68. doi:10.1177/28.suppl_5.s-58.
12. Ostenberg A, Roos H. Injury risk factors in female European football. a prospective study of 123 players during one season. Scand J Med Sci Sports. 2000;10(5):279-285. doi:10.1034/j.1600-0838.2000.010005279.x.
13. Backous DD, Friedl KE, Smith NJ, Parr TJ, Carpine WD. Soccer injuries and their relation to physical maturity. Am J Dis Child. 1988;142(8):839-842. doi:10.1001/archpedi.1988.02150080045019.
14. Grimm NL, Jacobs JC, Kim J, Denney BS, Shea KG. Anterior cruciate ligament and knee injury prevention programs for soccer players: a systematic review and meta-analysis. Am J Sports Med. 2015;43(8):2049-2056. doi:10.1177/0363546514556737.
15. Dick R, Putukian M, Agel J, Evans TA, Marshall SW. Descriptive epidemiology of collegiate women's soccer injuries: National Collegiate Athletic Association Injury Surveillance System, 1988-1989 through 2002-2003. J Athl Train. 2007;42(2):278-285.
16. Renstrom P, Ljungqvist A, Arendt E, et al. Non-contact ACL injuries in female athletes: an International Olympic Committee current concepts statement. Br J Sports Med. 2008;42(6):394-412. doi:10.1136/bjsm.2008.048934.
17. Guskiewicz KM, Weaver NL, Padua DA, Garrett WE. Epidemiology of concussion in collegiate and high school football players. Am J Sports Med. 2000;28(5):643-650. doi:10.1177/03635465000280050401.
18. Levy IM, Skovron ML, Agel J. Living with artificial grass: a knowledge update. Part 1: Basic science. Am J Sports Med. 1990;18(4):406-412. doi:10.1177/036354659001800413.
19. Meyers MC. Incidence, Mechanisms, and severity of match-related collegiate men's soccer injuries on fieldturf and natural grass surfaces: a 6-year prospective study. Am J Sports Med. 2017;45(3):708-718. doi:10.1177/0363546516671715.
20. Ekstrand J, Hägglund M, Fuller CW. Comparison of injuries sustained on artificial turf and grass by male and female elite football players. Scand J Med Sci Sports. 2011;21(6):824-832. doi:10.1111/j.1600-0838.2010.01118.x.
21. Meyers MC. Incidence, mechanisms, and severity of match-related collegiate women's soccer injuries on FieldTurf and natural grass surfaces: a 5-year prospective study. Am J Sports Med. 2013;41(10):2409-2420. doi:10.1177/0363546513498994.
22. Dragoo JL, Braun HJ, Harris AH. The effect of playing surface on the incidence of ACL injuries in National Collegiate Athletic Association American Football. Knee. 2013;20(3):191-195. doi:10.1016/j.knee.2012.07.006.
23. Rothenberg P, Grau L, Kaplan L, Baraga MG. Knee injuries in american football: an epidemiological review. Am J Orthop. 2016;45(6):368-373.
24. Waldén M, Hägglund M, Magnusson H, Ekstrand J. Anterior cruciate ligament injury in elite football: a prospective three-cohort study. Knee Surg Sports Traumatol Arthrosc. 2011;19(1):11-19. doi:10.1007/s00167-010-1170-9.
25. Waldén M, Krosshaug T, Bjørneboe J, Andersen TE, Faul O, Hägglund M. Three distinct mechanisms predominate in non-contact anterior cruciate ligament injuries in male professional football players: a systematic video analysis of 39 cases. Br J Sports Med. 2015;49(22):1452-1460. doi:10.1136/bjsports-2014-094573.
26. Olsen OE, Myklebust G, Engebretsen L, Bahr R. Injury mechanisms for anterior cruciate ligament injuries in team handball: a systematic video analysis. Am J Sports Med. 2004;32(4):1002-1012. doi:10.1177/0363546503261724.
27. Giza E, Mithöfer K, Farrell L, Zarins B, Gill T. Injuries in women's professional soccer. Br J Sports Med. 2005;39(4):212-216; discussion 212-216. doi:10.1136/bjsm.2004.011973.
28. Grassi A, Smiley SP, Roberti di Sarsina T, et al. Mechanisms and situations of anterior cruciate ligament injuries in professional male soccer players: a YouTube-based video analysis. Eur J Orthop Surg Traumatol. 2017;27(7):967-981. doi:10.1007/s00590-017-1905-0.
29. Brophy RH, Stepan JG, Silvers HJ, Mandelbaum BR. Defending puts the anterior cruciate ligament at risk during soccer: a gender-based analysis. Sports Health. 2015;7(3):244-249. doi:10.1177/1941738114535184.
30. Faude O, Junge A, Kindermann W, Dvorak J. Risk factors for injuries in elite female soccer players. Br J Sports Med. 2006;40(9):785-790. doi:10.1136/bjsm.2006.027540.
31. Agel J, Arendt EA, Bershadsky B. Anterior cruciate ligament injury in national collegiate athletic association basketball and soccer: a 13-year review. Am J Sports Med. 2005;33(4):524-530. doi:10.1177/0363546504269937.
32. Gwinn DE, Wilckens JH, McDevitt ER, Ross G, Kao TC. The relative incidence of anterior cruciate ligament injury in men and women at the United States Naval Academy. Am J Sports Med. 2000;28(1):98-102. doi:10.1177/03635465000280012901.
33. Arendt E, Dick R. Knee injury patterns among men and women in collegiate basketball and soccer. NCAA data and review of literature. Am J Sports Med. 1995;23(6):694-701. doi:10.1177/036354659502300611.
34. Mihata LC, Beutler AI, Boden BP. Comparing the incidence of anterior cruciate ligament injury in collegiate lacrosse, soccer, and basketball players: implications for anterior cruciate ligament mechanism and prevention. Am J Sports Med. 2006;34(6):899-904. doi:10.1177/0363546505285582.
35. Prodromos CC, Han Y, Rogowski J, Joyce B, Shi K. A meta-analysis of the incidence of anterior cruciate ligament tears as a function of gender, sport, and a knee injury-reduction regimen. Arthroscopy. 2007;23(12):1320-1325.e1326. doi:10.1016/j.arthro.2007.07.003.
36. Allen MM, Pareek A, Krych AJ, et al. Are female soccer players at an increased risk of second anterior cruciate ligament injury compared with their athletic peers? Am J Sports Med. 2016;44(10):2492-2498. doi:10.1177/0363546516648439.
37. Farber J, Harris JD, Kolstad K, McCulloch PC. Treatment of anterior cruciate ligament injuries by major league soccer team physicians. Orthop J Sports Med. 2014;2(11):2325967114559892. doi:10.1177/2325967114559892.
38. Mascarenhas R, Tranovich M, Karpie JC, Irrgang JJ, Fu FH, Harner CD. Patellar tendon anterior cruciate ligament reconstruction in the high-demand patient: evaluation of autograft versus allograft reconstruction. Arthroscopy. 2010;26(9 Suppl):S58-66. doi:10.1016/j.arthro.2010.01.004.
39. Kaeding CC, Aros B, Pedroza A, et al. Allograft versus autograft anterior cruciate ligament reconstruction: predictors of failure from a MOON prospective longitudinal cohort. Sports Health. 2011;3(1):73-81. doi:10.1177/1941738110386185.
40. Pallis M, Svoboda SJ, Cameron KL, Owens BD. Survival comparison of allograft and autograft anterior cruciate ligament reconstruction at the United States Military Academy. Am J Sports Med. 2012;40(6):1242-1246. doi:10.1177/0363546512443945.
41. Gifstad T, Foss OA, Engebretsen L, et al. Lower risk of revision with patellar tendon autografts compared with hamstring autografts: a registry study based on 45,998 primary ACL reconstructions in Scandinavia. Am J Sports Med. 2014;42(10):2319-2328. doi:10.1177/0363546514548164.
42. Mohammadi F, Salavati M, Akhbari B, Mazaheri M, Mohsen Mir S, Etemadi Y. Comparison of functional outcome measures after ACL reconstruction in competitive soccer players: a randomized trial. J Bone Joint Surg Am. 2013;95(14):1271-1277. doi:10.2106/JBJS.L.00724.
43. van der List JP, DiFelice GS. Arthroscopic primary anterior cruciate ligament repair with suture augmentation. Arthrosc Tech. 2017;6(5):e1529-e1534. doi:10.1016/j.eats.2017.06.009.
44. Murray MM, Flutie BM, Kalish LA, et al. The bridge-enhanced anterior cruciate ligament repair (BEAR) procedure: an early feasibility cohort study. Orthop J Sports Med. 2016;4(11):2325967116672176. doi:10.1177/2325967116672176.
45. DiFelice GS, van der List JP. Clinical outcomes of arthroscopic primary repair of proximal anterior cruciate ligament tears are maintained at mid-term follow-up. Arthroscopy. 2018;34(4):1085-1093. doi:10.1016/j.arthro.2017.10.028.
46. Howard JS, Lembach ML, Metzler AV, Johnson DL. Rates and determinants of return to play after anterior cruciate ligament reconstruction in national collegiate athletic association division I soccer athletes: a study of the southeastern conference. Am J Sports Med. 2016;44(2):433-439. doi:10.1177/0363546515614315.
47. Erickson BJ, Harris JD, Cvetanovich GL, et al. Performance and return to sport after anterior cruciate ligament reconstruction in male major league soccer players. Orthop J Sports Med. 2013;1(2):2325967113497189. doi:10.1177/2325967113497189.
48. Arundale AJH, Silvers-Granelli HJ, Snyder-Mackler L. Career length and injury incidence after anterior cruciate ligament reconstruction in major league soccer players. Orthop J Sports Med. 2018;6(1):2325967117750825. doi:10.1177/2325967117750825.
49. Brophy RH, Schmitz L, Wright RW, et al. Return to play and future ACL injury risk after ACL reconstruction in soccer athletes from the Multicenter Orthopaedic Outcomes Network (MOON) group. Am J Sports Med. 2012;40(11):2517-2522. doi:10.1177/0363546512459476.
50. Zaffagnini S, Grassi A, Marcheggiani Muccioli GM, et al. Return to sport after anterior cruciate ligament reconstruction in professional soccer players. Knee. 2014;21(3):731-735. doi:10.1016/j.knee.2014.02.005.
51. Waldén M, Hägglund M, Magnusson H, Ekstrand J. ACL injuries in men's professional football: a 15-year prospective study on time trends and return-to-play rates reveals only 65% of players still play at the top level 3 years after ACL rupture. Br J Sports Med. 2016;50(12):744-750. doi:10.1136/bjsports-2015-095952.
52. Andernord D, Desai N, Björnsson H, Ylander M, Karlsson J, Samuelsson K. Patient predictors of early revision surgery after anterior cruciate ligament reconstruction: a cohort study of 16,930 patients with 2-year follow-up. Am J Sports Med. 2015;43(1):121-127. doi:10.1177/0363546514552788.
53. Kramer DE, Miller PE, Berrahou IK, Yen YM, Heyworth BE. Collateral ligament knee injuries in pediatric and adolescent athletes. J Pediatr Orthop. 2017. doi:10.1097/BPO.0000000000001112.
54. Krutsch W, Zeman F, Zellner J, Pfeifer C, Nerlich M, Angele P. Increase in ACL and PCL injuries after implementation of a new professional football league. Knee Surg Sports Traumatol Arthrosc. 2016;24(7):2271-2279. doi:10.1007/s00167-014-3357-y.
55. Lundblad M, Waldén M, Magnusson H, Karlsson J, Ekstrand J. The UEFA injury study: 11-year data concerning 346 MCL injuries and time to return to play. Br J Sports Med. 2013;47(12):759-762. doi:10.1136/bjsports-2013-092305.
56. Stanley LE, Kerr ZY, Dompier TP, Padua DA. Sex differences in the incidence of anterior cruciate ligament, medial collateral ligament, and meniscal injuries in collegiate and high school sports: 2009-2010 Through 2013-2014. Am J Sports Med. 2016;44(6):1565-1572. doi:10.1177/0363546516630927.
57. Lind M, Jakobsen BW, Lund B, Hansen MS, Abdallah O, Christiansen SE. Anatomical reconstruction of the medial collateral ligament and posteromedial corner of the knee in patients with chronic medial collateral ligament instability. Am J Sports Med. 2009;37(6):1116-1122. doi:10.1177/0363546509332498.
58. Wijdicks CA, Griffith CJ, Johansen S, Engebretsen L, LaPrade RF. Injuries to the medial collateral ligament and associated medial structures of the knee. J Bone Joint Surg Am. 2010;92(5):1266-1280. doi:10.2106/JBJS.I.01229.
59. Marchant MH, Tibor LM, Sekiya JK, Hardaker WT, Garrett WE, Taylor DC. Management of medial-sided knee injuries, part 1: medial collateral ligament. Am J Sports Med. 2011;39(5):1102-1113. doi:10.1177/0363546510385999.
60. Corten K, Hoser C, Fink C, Bellemans J. Case reports: a Stener-like lesion of the medial collateral ligament of the knee. Clin Orthop Relat Res. 2010;468(1):289-293. doi:10.1007/s11999-009-0992-6
61. Fanelli GC, Edson CJ. Posterior cruciate ligament injuries in trauma patients: Part II. Arthroscopy. 1995;11(5):526-529. doi:10.1016/0749-8063(95)90127-2.
62. Schulz MS, Russe K, Weiler A, Eichhorn HJ, Strobel MJ. Epidemiology of posterior cruciate ligament injuries. Arch Orthop Trauma Surg. 2003;123(4):186-191. doi:10.1007/s00402-002-0471-y.
63. Fowler PJ, Messieh SS. Isolated posterior cruciate ligament injuries in athletes. Am J Sports Med. 1987;15(6):553-557. doi:10.1177/036354658701500606.
64. Patel DV, Allen AA, Warren RF, Wickiewicz TL, Simonian PT. The nonoperative treatment of acute, isolated (partial or complete) posterior cruciate ligament-deficient knees: an intermediate-term follow-up study. HSS J. 2007;3(2):137-146. doi:10.1007/s11420-007-9058-z.
65. Owesen C, Sandven-Thrane S, Lind M, Forssblad M, Granan LP, Årøen A. Epidemiology of surgically treated posterior cruciate ligament injuries in Scandinavia. Knee Surg Sports Traumatol Arthrosc. 2017;25(8):2384-2391. doi:10.1007/s00167-015-3786-2.
66. LaPrade CM, Civitarese DM, Rasmussen MT, LaPrade RF. Emerging updates on the posterior cruciate ligament: a review of the current literature. Am J Sports Med. 2015;43(12):3077-3092. doi:10.1177/0363546515572770.
67. Anderson CL. High rate of osteoarthritis of the knee in former soccer players. Med Sci Sports Exerc. 1986;18(1):141.
68. Arliani GG, Astur DC, Yamada RK, et al. Early osteoarthritis and reduced quality of life after retirement in former professional soccer players. Clinics (Sao Paulo). 2014;69(9):589-594. doi:10.6061/clinics/2014(09)03.
69. Wong P, Hong Y. Soccer injury in the lower extremities. Br J Sports Med. 2005;39(8):473-482. doi:10.1136/bjsm.2004.015511.
70. Thelin N, Holmberg S, Thelin A. Knee injuries account for the sports-related increased risk of knee osteoarthritis. Scand J Med Sci Sports. 2006;16(5):329-333. doi:10.1111/j.1600-0838.2005.00497.x.
71. Lohmander LS, Englund PM, Dahl LL, Roos EM. The long-term consequence of anterior cruciate ligament and meniscus injuries: osteoarthritis. Am J Sports Med. 2007;35(10):1756-1769. doi:10.1177/0363546507307396.
72. Mithöfer K, Peterson L, Mandelbaum BR, Minas T. Articular cartilage repair in soccer players with autologous chondrocyte transplantation: functional outcome and return to competition. Am J Sports Med. 2005;33(11):1639-1646. doi:10.1177/0363546505275647
73. Steadman JR, Briggs KK, Rodrigo JJ, Kocher MS, Gill TJ, Rodkey WG. Outcomes of microfracture for traumatic chondral defects of the knee: average 11-year follow-up. Arthroscopy. 2003;19(5):477-484. doi:10.1053/jars.2003.50112.
74. Hangody L, Ráthonyi GK, Duska Z, Vásárhelyi G, Füles P, Módis L. Autologous osteochondral mosaicplasty. Surgical technique. J Bone Joint Surg Am. 2004;86-A Suppl 1:65-72.
75. Sherman SL, Garrity J, Bauer K, Cook J, Stannard J, Bugbee W. Fresh osteochondral allograft transplantation for the knee: current concepts. J Am Acad Orthop Surg. 2014;22(2):121-133. doi:10.5435/JAAOS-22-02-121.
76. Andrade R, Vasta S, Papalia R, et al. Prevalence of articular cartilage lesions and surgical clinical outcomes in football (soccer) players' knees: a systematic review. Arthroscopy. 2016;32(7):1466-1477. doi:10.1016/j.arthro.2016.01.055.
77. Görtz S, Williams RJ, Gersoff WK, Bugbee WD. Osteochondral and meniscal allograft transplantation in the football (soccer) player. Cartilage. 2012;3(1 Suppl):37S-42S. doi:10.1177/1947603511416974.
78. Junge A, Grimm K, Feddermann N, Dvorak J. Precompetition orthopedic assessment of international elite football players. Clin J Sport Med. 2009;19(4):326-328. doi:10.1097/JSM.0b013e3181b21b56.
79. Salzmann GM, Preiss S, Zenobi-Wong M, Harder LP, Maier D, Dvorák J. Osteoarthritis in Football. Cartilage. 2017;8(2):162-172. doi:10.1177/1947603516648186.
80. Makris EA, Hadidi P, Athanasiou KA. The knee meniscus: structure-function, pathophysiology, current repair techniques, and prospects for regeneration. Biomaterials. 2011;32(30):7411-7431. doi:10.1016/j.biomaterials.2011.06.037
81. Freutel M, Seitz AM, Ignatius A, Dürselen L. Influence of partial meniscectomy on attachment forces, superficial strain and contact mechanics in porcine knee joints. Knee Surg Sports Traumatol Arthrosc. 2015;23(1):74-82. doi:10.1007/s00167-014-2951-3.
82. Papalia R, Del Buono A, Osti L, Denaro V, Maffulli N. Meniscectomy as a risk factor for knee osteoarthritis: a systematic review. Br Med Bull. 2011;99:89-106. doi:10.1093/bmb/ldq043.
83. Nawabi DH, Cro S, Hamid IP, Williams A. Return to play after lateral meniscectomy compared with medial meniscectomy in elite professional soccer players. Am J Sports Med. 2014;42(9):2193-2198. doi:10.1177/0363546514540271.
84. Alentorn-Geli E, Vázquez RS, Díaz PA, Cuscó X, Cugat R. Arthroscopic meniscal transplants in soccer players: outcomes at 2- to 5-year follow-up. Clin J Sport Med. 2010;20(5):340-343. doi:10.1097/JSM.0b013e3181f207dc.
85. Marcacci M, Marcheggiani Muccioli GM, Grassi A, et al. Arthroscopic meniscus allograft transplantation in male professional soccer players: a 36-month follow-up study. Am J Sports Med. 2014;42(2):382-388. doi:10.1177/0363546513508763.
86. Bizzini M, Dvorak J. FIFA 11+: an effective programme to prevent football injuries in various player groups worldwide-a narrative review. Br J Sports Med. 2015;49(9):577-579. doi:10.1136/bjsports-2015-094765.
87. Junge A, Lamprecht M, Stamm H, et al. Countrywide campaign to prevent soccer injuries in Swiss amateur players. Am J Sports Med. 2011;39(1):57-63. doi:10.1177/0363546510377424.
88. Al Attar WSA, Soomro N, Pappas E, Sinclair PJ, Sanders RH. Adding a post-training FIFA 11+ exercise program to the pre-training FIFA 11+ injury prevention program reduces injury rates among male amateur soccer players: a cluster-randomised trial. J Physiother. 2017;63(4):235-242. doi:10.1016/j.jphys.2017.08.004.
ABSTRACT
As one of the most popular sports in the world, soccer injury rates involving the knee continue to rise. An alarming trend of knee injuries, including increased anterior cruciate ligament ruptures, underscores the need to review our current understanding of these injuries in soccer players. This article includes a critical review of the epidemiology of knee injuries in soccer, anterior cruciate ligament and other ligamentous injuries, cartilage and meniscal injury, post-traumatic osteoarthritis, as well as current prevention initiatives.
Continue to: EPIDEMIOLOGY...
EPIDEMIOLOGY
There are currently 28 players on each of the Major League Soccer (MLS) teams, and during the 2013 to 2014 academic year, the National Federation of State High School Associations (NFHS) reported that 417,419 boys and 374,564 girls played high school soccer and the National Collegiate Athletic Association (NCAA) reported that 23,602 males and 26,358 females played collegiate soccer.5 As such, knee injuries in this population are a major concern for those involved in sports medicine. Several injuries occurring during soccer involve the lower extremity, particularly the knee.1 In fact, multiple reports estimate that up to 17.6% of soccer-related injuries presenting to the emergency room involved the knee.1,6-9 The majority of these injuries are noncontact injuries, although contact injuries do still occur.10,11
Risk factors for injuries in soccer may be non-modifiable (such as age and gender) and modifiable (such as level of conditioning, force, balance, and flexibility). Inadequate lower motor coordination may result in injury in the adolescent population, and advanced age >28 years in males and >25 years in females is considered as a high-risk factor for injury.12,13 Importantly, gender and age have been reported to play a significant role as risk factors for ACL injury.6 In fact, female players have a 3 to 5 times higher risk of significant knee injury, including ACL injuries, than male players.4,14-16 Preventative programs such as the FIFA 11+ program have been set forth to augment conditioning as part of managing the modifiable risk factors.
Like American football, playing on artificial turf has been questioned as a contributor to injury compared to playing on natural grass.17,18 In recent years, newer generations of artificial turf have been developed to more closely replicate the characteristics of natural grass. Meyers19 compared the incidence, mechanisms, and severity of match-related collegiate men’s soccer injuries on artificial turf and those on natural grass and demonstrated no significant difference in knee injuries between the 2 surfaces. This finding was consistent with previous studies that reported no difference in the incidence of knee injuries on either surface in women’s collegiate and elite-level soccer.15,20,21
Continue to: ACL INJURIES...
ACL INJURIES
ACL injuries are life-changing events that can significantly affect the career of a soccer athlete. As a major stabilizer of the knee, the ACL primarily prevents anterior tibial translation with the anteromedial bundle and secondarily resists tibial rotation with the posterolateral bundle. The ligament takes origin from the posteromedial aspect of the lateral femoral condyle and inserts anterior to the tibial intercondylar eminence. Grading of ACL injuries is based on the Lachman test, which is performed between 20°and 30° of knee flexion and measures the amount of anterior tibial translation relative to the femur (A = firm endpoint, B = no endpoint; grade I: 3-5 mm, grade II (A/B): 5-10 mm, grade III (A/B): >10 mm).
ACL injury may occur via contact or noncontact mechanisms. Noncontact mechanisms of ACL injury in soccer athletes contribute to about 85% of injuries.6,22-25 Typical noncontact mechanism of injury involves a forceful valgus collapse with the knee near full extension and combined external or internal rotation of the tibia23,26 (Figure 1). This on-field scenario generally involves cutting and torsional movement, as well as landing after a jump, particularly in 1-legged stance. Similarly, a disturbance in balance caused by the opponent may incite a noncontact mechanism resulting in ACL rupture.6,27 Video analyses of professional soccer players have also demonstrated a higher risk of noncontact ACL injury within the first 9 minutes of the match, with the most common playing situation resulting in injury being pressing, followed by kicking and heading.24,25,28 Contact mechanisms resulting in ACL injury, however, are not an uncommon occurrence in soccer players with higher risk for certain positions. Brophy and colleagues29 reviewed ACL injuries in professional and collegiate soccer players and reported a higher risk of ACL injury during defending and tackling. Similarly, Faude and colleagues30 found the risk of injury to be higher in defenders and strikers than in goalkeepers and midfielders.

Female athletes participating in elite-level athletics, especially soccer, represent a high-risk group for ACL injury. In fact, these soccer athletes experience ACL injury at an incidence 3 times higher than that in male athletes.31-35 Female soccer athletes may also be at risk for reinjury to the ACL and contralateral ACL injury. Female gender, in combination with participation in soccer, thus represents a high-risk group for ACL tear in athletics. Allen and colleagues36 retrospectively reviewed 180 female patients who had undergone ACL reconstruction (ACLR) (90 soccer players and 90 non-soccer players) over a mean period of 68.8 months. In their series, soccer players sustained significantly more ACL injuries than non-soccer players, including graft failures (11% vs 1%) and contralateral ACL tears (17% vs 4%).
ACLR is the gold standard treatment for elite soccer athletes. A recent survey of MLS team orthopedic surgeons revealed several important details regarding decision-making in ACLR in this population. From a technical standpoint, the vast majority of surgeons used a single incision, arthroscopically assisted, single-bundle reconstruction (91%). Femoral tunnel drilling was almost equally split between transtibial (51%) and use of an accessory medial portal (46%). Bone-patella-tendon-bone (BPTB) autograft was the most preferred graft choice (68%), and quadriceps tendon autograft was the least preferred. The majority of surgeons preformed ACLR within 4 weeks and permitted return to sport (RTS) without restrictions at 6 to 8 months.37
Continue to: There is a scarcity of literature regarding...
There is a scarcity of literature regarding the use of soft tissue and BPTB allografts in soccer athletes. However, one study reported no difference in patient-reported outcomes and return to preinjury level of activity (including soccer) with the use of either autograft or allograft BPTB in ACLR.38 The authors’ preference was to avoid the use of allograft in elite-level soccer athletes as the reported rate of ACL re-tear was 4 to 8 times higher than that with autograft reconstruction, as shown in athletes and military personnel.39,40 BPTB autograft and hamstring autograft (semitendinosus and/or gracilis) are common graft choices for soccer athletes. Gifstad and colleagues41 compared BPTB autograft and hamstring autograft in 45,998 primary ACLRs performed in Scandinavia. Although the cohort included, but was not limited to, soccer players, the authors reported an overall risk of revision that was significantly lower in the BPTB autograft group than in the hamstring autograft group (hazard ratio, 0.63; 95% confidence interval, 0.53-0.74).41 Mohammadi and colleagues42 prospectively compared the functional outcomes of 42 competitive soccer players who underwent ACLR with BPTB autograft vs those who underwent ACLR with hamstring autograft at the time of RTS. Players who had undergone ACLR with hamstring autograft demonstrated greater quadriceps torque, as well as better performance with triple-hop, crossover-hop, and jump-landing tests; however, both groups demonstrated similar hamstring torque and performance in 2 other hop tests.42 In the authors’ opinion, there may be a concern regarding the use of hamstring autograft in elite soccer players considering that hamstring strains are extremely common in this athletic population; however, further research would be necessary to elucidate whether this is an actual or a theoretical risk. Although not yet studied in elite-level athletes, early clinical results of ACL repair with suture augmentation show promise for certain injury patterns. These include proximal femoral ACL avulsion injuries (Sherman type 1) of excellent tissue quality that have the ability to be reapproximated to the femoral origin43 (Figures 2A, 2B). In a recent series,43-45 early clinical outcomes were found to be excellent and maintained at midterm follow-up.

In the NCAA soccer athletes, an overall RTS rate of 85% has been reported in those undergoing ACLR, with a significantly higher rate observed in scholarship versus non-scholarship athletes.46 Howard and colleagues46 reported median time to unrestricted game play of 6.1 months, with 75% returning to the same or higher level position on the depth chart. Among their studied collegiate soccer athletes, 32% reported continued participation in soccer on some level after college (recreational, semiprofessional, or professional).46 RTS rates for MLS soccer players have also been reported to be high, ranging from 74% to 77%, most of them returning within the following season at 10 ± 2.8 months.47,48 These findings were consistent with the RTS rate of 72% reported by the Multicenter Orthopaedic Outcomes Network (MOON) group, which analyzed 100 female and male soccer players undergoing ACLR at a minimum 7-year follow-up. In this series, Brophy and colleagues29,49 reported an RTS at 12 ± 14.3 months, with 85% returning to the same or a higher level of play prior to their injury. Erickson and colleagues47 analyzed a series of 57 ACLRs performed in MLS athletes and reported no significant difference in preinjury or postoperative performance, or between cases and uninjured controls. Arundale and colleagues48 demonstrated no significantly increased risk of lower extremity injury in MLS athletes after ACLR, but the athletes had significantly shorter careers than their uninjured counterparts. Curiously, RTS rates for European professional soccer athletes have been reported to be substantially higher at 95% to 97%.50,51 Although we can only speculate the reasons for such a discrepancy, the difference in RTS rates for similar athletes highlights a need for objective criteria to determine and report RTS rates, while also providing guidelines to prevent reinjury. Such a consensus among orthopedists is not yet present in the literature.
Soccer players and adolescent age in combination have been shown to portend a 3-fold increased risk of revision surgery for ACL failure in a cohort of 16,930 patients from the Swedish National Knee Ligament Register.52 Published data regarding ACL failure and management of revision ACLR in elite-level soccer athletes are currently lacking. However, low failure rates of 3% to 10% requiring revision reconstruction have been reported.47,49 Arundale and colleagues48 reported 2 incidences of players with ACL graft failures, 1 BPTB autograft and 1 BPTB allograft, both of whom were able to return to MLS after revision ACLR. It is the authors’ preference to use ipsilateral hamstring autograft or contralateral BPTB autograft when an ACL revision reconstruction is required.
Continue to: OTHER LIGAMENTOUS INJURIES...
OTHER LIGAMENTOUS INJURIES
The majority of research efforts regarding knee injuries in this population are focused on the ACL. Correspondingly, literature regarding injury to the collateral ligaments and the posterior cruciate ligament (PCL) in soccer players is sparse. The lateral collateral ligament (LCL) and the medial collateral ligament (MCL) play important roles as primary stabilizers to varus and valgus forces, respectively. The PCL is the primary posterior stabilizer of the knee, preventing posterior translation of the tibia. Injury to these structures may result in significant time lost from soccer and risk of reinjury.53,54
The MCL is the one of the most commonly injured ligaments in sports, including soccer.53,55 The injury mechanism generally involves contact with a resulting valgus force applied to the knee.55 Grading of MCL injuries is based on the amount of medial joint gapping with applied valgus force during examination (grade I: <5 mm, grade II: 5-10 mm, grade III: >10 mm). Kramer and colleagues53 reviewed collateral ligament injuries in the adolescent population and found that MCL injuries occurred 4 times more often than LCL injuries and about 25% were grade III injuries, most commonly occurring in American football and soccer players. Soccer also touts the highest sport-specific MCL injury rate for high school and collegiate athletics, particularly for female NCAA soccer players.56 At the professional level, Lundblad and colleagues55 reported 346 MCL injuries in 27 European teams over an 11-year period, of which 70% were contact-related, and the average time-off from soccer was 28 days.
Most surgeons treat isolated MCL injuries nonoperatively, regardless of grade.57,58 This includes activity modification, use of a hinged knee brace, quadriceps strengthening, and progressive return to play. The literature currently lacks substantial data to guide MCL injury management, specifically in elite soccer athletes. In our experience, grade I injuries are managed nonoperatively and RTS is allowed at 4 to 6 weeks. Grade II injuries are also managed nonoperatively and RTS is allowed at 6 to 8 weeks. Grade III injuries are generally allowed RTS at 8 to 12 weeks and may be considered for surgery in the context of concomitant injuries (eg, posteromedial capsular injury, multiligamentous knee injuries, and meniscal injuries). In some athletes, we consider using a varus unloader brace to help maximize decreased stress on the MCL while still allowing the athlete to be fully weight-bearing. We have found it less ideal to limit weight-bearing in elite athletes, which may negatively affect overall lower extremity neuromuscular proprioception and potentially prolong a safe return to play. Some athletes may experience prolonged soreness at the MCL femoral or tibial attachment despite being able to return to play. It is important to counsel athletes about these prolonged symptoms to set expectations, as this may even occur with grade I MCL injuries. Other rare instances where surgical management may be indicated include persistent pain and instability following nonoperative treatment of grade III injuries and highly displaced tibial avulsions of the ligament resulting in poor healing.59,60
Data regarding LCL injuries in soccer are extremely sparse. In our experience, treatment and RTS rates for isolated LCL injuries are similar to those for MCL injuries. However, it is worth noting that one-quarter of LCL injuries may occur in combination with injury to other posterolateral corner structures.53
PCL injuries are more commonly associated with vehicular trauma but have also been reported to occur in sports at a rate of 33% to 40%.61,62 The mechanism of injury in athletes generally involves a fall onto the hyperflexed knee with the foot in plantarflexion or a direct blow to the anterior tibia in a flexed knee.62,63 Classification of PCL injuries is based on posterior translation of the tibia relative to the femur with the knee flexed to 90°(grade I: 1-5 mm, grade II: 6-10 mm, grade III: >10 mm). In one cohort of 62 patients with isolated PCL injuries, soccer was found to be among the top 5 causes of injury.64 A Scandinavian review of 1287 patients who underwent PCL reconstruction found soccer to be the sport with the highest number of injuries (13.1%).65 The goalkeeper was most commonly subjected to this injury.62 Krutsch and colleagues54 compared PCL injuries in new, professional soccer players to those in players at the closest amateur level of play. In their series, 90% of PCL injuries occurred during preseason in players who were at a lower level of play in the previous season. This finding suggested that a rapid increase in training and playing intensity may have been a significant risk factor for PCL injury. Substantial literature supporting nonoperative or operative management of PCL injuries in soccer athletes is currently lacking. Historically, nonoperative treatment has been the initial management for isolated PCL injuries; however, surgical intervention has become increasingly used for both isolated and combined PCL injuries.66
Continue to: CARTILAGE AND MENISCAL INJURIES...
CARTILAGE AND MENISCAL INJURIES
The prevalence of osteoarthritis (OA) in retired soccer players is high.67,68 Articular cartilage degeneration with subsequent OA occurs in up to 32% of soccer players and ultimately leads to significant disability and retirement from the sport. High physical demands and concomitant knee injuries probably predispose to the development of posttraumatic OA.69-71
Several techniques addressing cartilage débridement or restoration have been reported, with successful RTS but with variable durability.72-75 Recently, Andrade and colleagues76 performed a systematic review of 217 articular cartilage defects in soccer players that were treated using restoration techniques, including chondroplasty, microfracture, autologous chondrocyte implantation (ACI), and osteochondral autograft. Although no superior technique could be ascertained, microfracture and osteochondral autograft procedures led to the quickest return to play, and ACI techniques enhanced long-standing clinical and functional results.76 More recently, osteochondral allograft transplantation has also been described with an 84% return to some level of activity (including soccer) and 60% of athletes returning to high-level sports participation at a mean follow-up of 4.5 years77 (Figures 3A-3C). Although chondroplasty may be successful and allow for a quicker return to play in some soccer players (return to play from 6-12 weeks), the authors believe that a strong cartilage scaffold repair strategy with early weight-bearing, including osteochondral autograft and allograft procedures (return to play from 6-9 months), must also be considered in focal chondral defects to optimize both short-term and potential long-term success.

Meniscal injuries are also prevalent in the soccer population, and consistent with ACL injuries, female players are at least twice as likely to sustain a meniscal tear.78,79 Meniscal damage can occur in isolation or in association with ACL rupture. Repair techniques should be strongly considered as chondral changes in the setting of meniscal deficiency are a significant short- and long-term concern for elite athletes. However, due to intrinsically poor healing potential, partial meniscectomy is unfortunately more often performed.79,80 In either case, meniscal deficiency is recognized as a precursor to the development of OA as meniscal functionality is lost and the articular cartilage is subjected to increased biomechanical loading.81,82 Nawabi and colleagues83 analyzed RTS in 90 professional soccer players following partial meniscectomy. Median RTS was at 7 weeks for lateral meniscectomies and at 5 weeks for medial meniscectomies. RTS probability was 5.99 times greater after medial meniscectomy at all time points. Lateral meniscectomies were associated with an increased risk of postoperative adverse events, reoperation, and a significantly lower rate of return to play.83 In the case of severe meniscal deficiency, particularly post-meniscectomy, meniscal allograft transplantation (MAT) may be considered. In a series of MATs in lower division Spanish players, 12/14 (85.7%) returned to play at an average of 7.6 months.84 A more recent series of professional players reported 9/12 (75%) RTS as professionals and 2/12 (17%) as semiprofessionals at an average of 10.5 months.85 The authors’ strong preference is to perform meniscus-saving procedures whenever possible. Due to the longer recovery and return to play associated with meniscus repair than partial meniscectomy, most of the soccer players will often prefer to proceed with partial meniscectomy. Despite the ultimate treatment, it is critical that the surgeon and the soccer player have an in-depth conversation concerning the risks and benefits for each procedure and individualize treatment to the individual soccer player accordingly.
Continue to: INJURY PREVENTION...
INJURY PREVENTION
Given the breadth and the prevalence of soccer-related injuries, the FIFA11+ program was developed in 2006 as an injury prevention measure (Figure 4). The warm-up program includes 15 structured exercises emphasizing core stabilization, thigh muscle training, proprioception, dynamic stabilization, and plyometric exercises. The routine is believed to be easily executed and effective at preventing the incidence of noncontact injuries.86,87 Recently, Sadigursky and colleagues1 performed a systematic review of randomized clinical trials examining the efficacy of FIFA11+. The authors reported a reduction in injuries by 30% and a relative risk of 0.70 for lower limb injuries, highlighting the significant preventative importance of the program.1 Post-training programs may also be beneficial as it has been shown that performing FIFA11+ both before and after training reduced overall injury rates in male, amateur soccer players.88 Regardless of the prevention program, it is critical that every league, team, medical team, and athlete have a thorough injury prevention strategy to help keep players healthy and not wait until they have instead sustained a significant injury.

CONCLUSION
Knee injuries are common in soccer, with an alarming number of ACL injuries, as well as other significant pathology. Understanding the unique epidemiology, risk factors, treatment, and injury prevention strategies is critically important in helping medical professionals provide care for all levels of elite soccer players.
ABSTRACT
As one of the most popular sports in the world, soccer injury rates involving the knee continue to rise. An alarming trend of knee injuries, including increased anterior cruciate ligament ruptures, underscores the need to review our current understanding of these injuries in soccer players. This article includes a critical review of the epidemiology of knee injuries in soccer, anterior cruciate ligament and other ligamentous injuries, cartilage and meniscal injury, post-traumatic osteoarthritis, as well as current prevention initiatives.
Continue to: EPIDEMIOLOGY...
EPIDEMIOLOGY
There are currently 28 players on each of the Major League Soccer (MLS) teams, and during the 2013 to 2014 academic year, the National Federation of State High School Associations (NFHS) reported that 417,419 boys and 374,564 girls played high school soccer and the National Collegiate Athletic Association (NCAA) reported that 23,602 males and 26,358 females played collegiate soccer.5 As such, knee injuries in this population are a major concern for those involved in sports medicine. Several injuries occurring during soccer involve the lower extremity, particularly the knee.1 In fact, multiple reports estimate that up to 17.6% of soccer-related injuries presenting to the emergency room involved the knee.1,6-9 The majority of these injuries are noncontact injuries, although contact injuries do still occur.10,11
Risk factors for injuries in soccer may be non-modifiable (such as age and gender) and modifiable (such as level of conditioning, force, balance, and flexibility). Inadequate lower motor coordination may result in injury in the adolescent population, and advanced age >28 years in males and >25 years in females is considered as a high-risk factor for injury.12,13 Importantly, gender and age have been reported to play a significant role as risk factors for ACL injury.6 In fact, female players have a 3 to 5 times higher risk of significant knee injury, including ACL injuries, than male players.4,14-16 Preventative programs such as the FIFA 11+ program have been set forth to augment conditioning as part of managing the modifiable risk factors.
Like American football, playing on artificial turf has been questioned as a contributor to injury compared to playing on natural grass.17,18 In recent years, newer generations of artificial turf have been developed to more closely replicate the characteristics of natural grass. Meyers19 compared the incidence, mechanisms, and severity of match-related collegiate men’s soccer injuries on artificial turf and those on natural grass and demonstrated no significant difference in knee injuries between the 2 surfaces. This finding was consistent with previous studies that reported no difference in the incidence of knee injuries on either surface in women’s collegiate and elite-level soccer.15,20,21
Continue to: ACL INJURIES...
ACL INJURIES
ACL injuries are life-changing events that can significantly affect the career of a soccer athlete. As a major stabilizer of the knee, the ACL primarily prevents anterior tibial translation with the anteromedial bundle and secondarily resists tibial rotation with the posterolateral bundle. The ligament takes origin from the posteromedial aspect of the lateral femoral condyle and inserts anterior to the tibial intercondylar eminence. Grading of ACL injuries is based on the Lachman test, which is performed between 20°and 30° of knee flexion and measures the amount of anterior tibial translation relative to the femur (A = firm endpoint, B = no endpoint; grade I: 3-5 mm, grade II (A/B): 5-10 mm, grade III (A/B): >10 mm).
ACL injury may occur via contact or noncontact mechanisms. Noncontact mechanisms of ACL injury in soccer athletes contribute to about 85% of injuries.6,22-25 Typical noncontact mechanism of injury involves a forceful valgus collapse with the knee near full extension and combined external or internal rotation of the tibia23,26 (Figure 1). This on-field scenario generally involves cutting and torsional movement, as well as landing after a jump, particularly in 1-legged stance. Similarly, a disturbance in balance caused by the opponent may incite a noncontact mechanism resulting in ACL rupture.6,27 Video analyses of professional soccer players have also demonstrated a higher risk of noncontact ACL injury within the first 9 minutes of the match, with the most common playing situation resulting in injury being pressing, followed by kicking and heading.24,25,28 Contact mechanisms resulting in ACL injury, however, are not an uncommon occurrence in soccer players with higher risk for certain positions. Brophy and colleagues29 reviewed ACL injuries in professional and collegiate soccer players and reported a higher risk of ACL injury during defending and tackling. Similarly, Faude and colleagues30 found the risk of injury to be higher in defenders and strikers than in goalkeepers and midfielders.

Female athletes participating in elite-level athletics, especially soccer, represent a high-risk group for ACL injury. In fact, these soccer athletes experience ACL injury at an incidence 3 times higher than that in male athletes.31-35 Female soccer athletes may also be at risk for reinjury to the ACL and contralateral ACL injury. Female gender, in combination with participation in soccer, thus represents a high-risk group for ACL tear in athletics. Allen and colleagues36 retrospectively reviewed 180 female patients who had undergone ACL reconstruction (ACLR) (90 soccer players and 90 non-soccer players) over a mean period of 68.8 months. In their series, soccer players sustained significantly more ACL injuries than non-soccer players, including graft failures (11% vs 1%) and contralateral ACL tears (17% vs 4%).
ACLR is the gold standard treatment for elite soccer athletes. A recent survey of MLS team orthopedic surgeons revealed several important details regarding decision-making in ACLR in this population. From a technical standpoint, the vast majority of surgeons used a single incision, arthroscopically assisted, single-bundle reconstruction (91%). Femoral tunnel drilling was almost equally split between transtibial (51%) and use of an accessory medial portal (46%). Bone-patella-tendon-bone (BPTB) autograft was the most preferred graft choice (68%), and quadriceps tendon autograft was the least preferred. The majority of surgeons preformed ACLR within 4 weeks and permitted return to sport (RTS) without restrictions at 6 to 8 months.37
Continue to: There is a scarcity of literature regarding...
There is a scarcity of literature regarding the use of soft tissue and BPTB allografts in soccer athletes. However, one study reported no difference in patient-reported outcomes and return to preinjury level of activity (including soccer) with the use of either autograft or allograft BPTB in ACLR.38 The authors’ preference was to avoid the use of allograft in elite-level soccer athletes as the reported rate of ACL re-tear was 4 to 8 times higher than that with autograft reconstruction, as shown in athletes and military personnel.39,40 BPTB autograft and hamstring autograft (semitendinosus and/or gracilis) are common graft choices for soccer athletes. Gifstad and colleagues41 compared BPTB autograft and hamstring autograft in 45,998 primary ACLRs performed in Scandinavia. Although the cohort included, but was not limited to, soccer players, the authors reported an overall risk of revision that was significantly lower in the BPTB autograft group than in the hamstring autograft group (hazard ratio, 0.63; 95% confidence interval, 0.53-0.74).41 Mohammadi and colleagues42 prospectively compared the functional outcomes of 42 competitive soccer players who underwent ACLR with BPTB autograft vs those who underwent ACLR with hamstring autograft at the time of RTS. Players who had undergone ACLR with hamstring autograft demonstrated greater quadriceps torque, as well as better performance with triple-hop, crossover-hop, and jump-landing tests; however, both groups demonstrated similar hamstring torque and performance in 2 other hop tests.42 In the authors’ opinion, there may be a concern regarding the use of hamstring autograft in elite soccer players considering that hamstring strains are extremely common in this athletic population; however, further research would be necessary to elucidate whether this is an actual or a theoretical risk. Although not yet studied in elite-level athletes, early clinical results of ACL repair with suture augmentation show promise for certain injury patterns. These include proximal femoral ACL avulsion injuries (Sherman type 1) of excellent tissue quality that have the ability to be reapproximated to the femoral origin43 (Figures 2A, 2B). In a recent series,43-45 early clinical outcomes were found to be excellent and maintained at midterm follow-up.

In the NCAA soccer athletes, an overall RTS rate of 85% has been reported in those undergoing ACLR, with a significantly higher rate observed in scholarship versus non-scholarship athletes.46 Howard and colleagues46 reported median time to unrestricted game play of 6.1 months, with 75% returning to the same or higher level position on the depth chart. Among their studied collegiate soccer athletes, 32% reported continued participation in soccer on some level after college (recreational, semiprofessional, or professional).46 RTS rates for MLS soccer players have also been reported to be high, ranging from 74% to 77%, most of them returning within the following season at 10 ± 2.8 months.47,48 These findings were consistent with the RTS rate of 72% reported by the Multicenter Orthopaedic Outcomes Network (MOON) group, which analyzed 100 female and male soccer players undergoing ACLR at a minimum 7-year follow-up. In this series, Brophy and colleagues29,49 reported an RTS at 12 ± 14.3 months, with 85% returning to the same or a higher level of play prior to their injury. Erickson and colleagues47 analyzed a series of 57 ACLRs performed in MLS athletes and reported no significant difference in preinjury or postoperative performance, or between cases and uninjured controls. Arundale and colleagues48 demonstrated no significantly increased risk of lower extremity injury in MLS athletes after ACLR, but the athletes had significantly shorter careers than their uninjured counterparts. Curiously, RTS rates for European professional soccer athletes have been reported to be substantially higher at 95% to 97%.50,51 Although we can only speculate the reasons for such a discrepancy, the difference in RTS rates for similar athletes highlights a need for objective criteria to determine and report RTS rates, while also providing guidelines to prevent reinjury. Such a consensus among orthopedists is not yet present in the literature.
Soccer players and adolescent age in combination have been shown to portend a 3-fold increased risk of revision surgery for ACL failure in a cohort of 16,930 patients from the Swedish National Knee Ligament Register.52 Published data regarding ACL failure and management of revision ACLR in elite-level soccer athletes are currently lacking. However, low failure rates of 3% to 10% requiring revision reconstruction have been reported.47,49 Arundale and colleagues48 reported 2 incidences of players with ACL graft failures, 1 BPTB autograft and 1 BPTB allograft, both of whom were able to return to MLS after revision ACLR. It is the authors’ preference to use ipsilateral hamstring autograft or contralateral BPTB autograft when an ACL revision reconstruction is required.
Continue to: OTHER LIGAMENTOUS INJURIES...
OTHER LIGAMENTOUS INJURIES
The majority of research efforts regarding knee injuries in this population are focused on the ACL. Correspondingly, literature regarding injury to the collateral ligaments and the posterior cruciate ligament (PCL) in soccer players is sparse. The lateral collateral ligament (LCL) and the medial collateral ligament (MCL) play important roles as primary stabilizers to varus and valgus forces, respectively. The PCL is the primary posterior stabilizer of the knee, preventing posterior translation of the tibia. Injury to these structures may result in significant time lost from soccer and risk of reinjury.53,54
The MCL is the one of the most commonly injured ligaments in sports, including soccer.53,55 The injury mechanism generally involves contact with a resulting valgus force applied to the knee.55 Grading of MCL injuries is based on the amount of medial joint gapping with applied valgus force during examination (grade I: <5 mm, grade II: 5-10 mm, grade III: >10 mm). Kramer and colleagues53 reviewed collateral ligament injuries in the adolescent population and found that MCL injuries occurred 4 times more often than LCL injuries and about 25% were grade III injuries, most commonly occurring in American football and soccer players. Soccer also touts the highest sport-specific MCL injury rate for high school and collegiate athletics, particularly for female NCAA soccer players.56 At the professional level, Lundblad and colleagues55 reported 346 MCL injuries in 27 European teams over an 11-year period, of which 70% were contact-related, and the average time-off from soccer was 28 days.
Most surgeons treat isolated MCL injuries nonoperatively, regardless of grade.57,58 This includes activity modification, use of a hinged knee brace, quadriceps strengthening, and progressive return to play. The literature currently lacks substantial data to guide MCL injury management, specifically in elite soccer athletes. In our experience, grade I injuries are managed nonoperatively and RTS is allowed at 4 to 6 weeks. Grade II injuries are also managed nonoperatively and RTS is allowed at 6 to 8 weeks. Grade III injuries are generally allowed RTS at 8 to 12 weeks and may be considered for surgery in the context of concomitant injuries (eg, posteromedial capsular injury, multiligamentous knee injuries, and meniscal injuries). In some athletes, we consider using a varus unloader brace to help maximize decreased stress on the MCL while still allowing the athlete to be fully weight-bearing. We have found it less ideal to limit weight-bearing in elite athletes, which may negatively affect overall lower extremity neuromuscular proprioception and potentially prolong a safe return to play. Some athletes may experience prolonged soreness at the MCL femoral or tibial attachment despite being able to return to play. It is important to counsel athletes about these prolonged symptoms to set expectations, as this may even occur with grade I MCL injuries. Other rare instances where surgical management may be indicated include persistent pain and instability following nonoperative treatment of grade III injuries and highly displaced tibial avulsions of the ligament resulting in poor healing.59,60
Data regarding LCL injuries in soccer are extremely sparse. In our experience, treatment and RTS rates for isolated LCL injuries are similar to those for MCL injuries. However, it is worth noting that one-quarter of LCL injuries may occur in combination with injury to other posterolateral corner structures.53
PCL injuries are more commonly associated with vehicular trauma but have also been reported to occur in sports at a rate of 33% to 40%.61,62 The mechanism of injury in athletes generally involves a fall onto the hyperflexed knee with the foot in plantarflexion or a direct blow to the anterior tibia in a flexed knee.62,63 Classification of PCL injuries is based on posterior translation of the tibia relative to the femur with the knee flexed to 90°(grade I: 1-5 mm, grade II: 6-10 mm, grade III: >10 mm). In one cohort of 62 patients with isolated PCL injuries, soccer was found to be among the top 5 causes of injury.64 A Scandinavian review of 1287 patients who underwent PCL reconstruction found soccer to be the sport with the highest number of injuries (13.1%).65 The goalkeeper was most commonly subjected to this injury.62 Krutsch and colleagues54 compared PCL injuries in new, professional soccer players to those in players at the closest amateur level of play. In their series, 90% of PCL injuries occurred during preseason in players who were at a lower level of play in the previous season. This finding suggested that a rapid increase in training and playing intensity may have been a significant risk factor for PCL injury. Substantial literature supporting nonoperative or operative management of PCL injuries in soccer athletes is currently lacking. Historically, nonoperative treatment has been the initial management for isolated PCL injuries; however, surgical intervention has become increasingly used for both isolated and combined PCL injuries.66
Continue to: CARTILAGE AND MENISCAL INJURIES...
CARTILAGE AND MENISCAL INJURIES
The prevalence of osteoarthritis (OA) in retired soccer players is high.67,68 Articular cartilage degeneration with subsequent OA occurs in up to 32% of soccer players and ultimately leads to significant disability and retirement from the sport. High physical demands and concomitant knee injuries probably predispose to the development of posttraumatic OA.69-71
Several techniques addressing cartilage débridement or restoration have been reported, with successful RTS but with variable durability.72-75 Recently, Andrade and colleagues76 performed a systematic review of 217 articular cartilage defects in soccer players that were treated using restoration techniques, including chondroplasty, microfracture, autologous chondrocyte implantation (ACI), and osteochondral autograft. Although no superior technique could be ascertained, microfracture and osteochondral autograft procedures led to the quickest return to play, and ACI techniques enhanced long-standing clinical and functional results.76 More recently, osteochondral allograft transplantation has also been described with an 84% return to some level of activity (including soccer) and 60% of athletes returning to high-level sports participation at a mean follow-up of 4.5 years77 (Figures 3A-3C). Although chondroplasty may be successful and allow for a quicker return to play in some soccer players (return to play from 6-12 weeks), the authors believe that a strong cartilage scaffold repair strategy with early weight-bearing, including osteochondral autograft and allograft procedures (return to play from 6-9 months), must also be considered in focal chondral defects to optimize both short-term and potential long-term success.

Meniscal injuries are also prevalent in the soccer population, and consistent with ACL injuries, female players are at least twice as likely to sustain a meniscal tear.78,79 Meniscal damage can occur in isolation or in association with ACL rupture. Repair techniques should be strongly considered as chondral changes in the setting of meniscal deficiency are a significant short- and long-term concern for elite athletes. However, due to intrinsically poor healing potential, partial meniscectomy is unfortunately more often performed.79,80 In either case, meniscal deficiency is recognized as a precursor to the development of OA as meniscal functionality is lost and the articular cartilage is subjected to increased biomechanical loading.81,82 Nawabi and colleagues83 analyzed RTS in 90 professional soccer players following partial meniscectomy. Median RTS was at 7 weeks for lateral meniscectomies and at 5 weeks for medial meniscectomies. RTS probability was 5.99 times greater after medial meniscectomy at all time points. Lateral meniscectomies were associated with an increased risk of postoperative adverse events, reoperation, and a significantly lower rate of return to play.83 In the case of severe meniscal deficiency, particularly post-meniscectomy, meniscal allograft transplantation (MAT) may be considered. In a series of MATs in lower division Spanish players, 12/14 (85.7%) returned to play at an average of 7.6 months.84 A more recent series of professional players reported 9/12 (75%) RTS as professionals and 2/12 (17%) as semiprofessionals at an average of 10.5 months.85 The authors’ strong preference is to perform meniscus-saving procedures whenever possible. Due to the longer recovery and return to play associated with meniscus repair than partial meniscectomy, most of the soccer players will often prefer to proceed with partial meniscectomy. Despite the ultimate treatment, it is critical that the surgeon and the soccer player have an in-depth conversation concerning the risks and benefits for each procedure and individualize treatment to the individual soccer player accordingly.
Continue to: INJURY PREVENTION...
INJURY PREVENTION
Given the breadth and the prevalence of soccer-related injuries, the FIFA11+ program was developed in 2006 as an injury prevention measure (Figure 4). The warm-up program includes 15 structured exercises emphasizing core stabilization, thigh muscle training, proprioception, dynamic stabilization, and plyometric exercises. The routine is believed to be easily executed and effective at preventing the incidence of noncontact injuries.86,87 Recently, Sadigursky and colleagues1 performed a systematic review of randomized clinical trials examining the efficacy of FIFA11+. The authors reported a reduction in injuries by 30% and a relative risk of 0.70 for lower limb injuries, highlighting the significant preventative importance of the program.1 Post-training programs may also be beneficial as it has been shown that performing FIFA11+ both before and after training reduced overall injury rates in male, amateur soccer players.88 Regardless of the prevention program, it is critical that every league, team, medical team, and athlete have a thorough injury prevention strategy to help keep players healthy and not wait until they have instead sustained a significant injury.

CONCLUSION
Knee injuries are common in soccer, with an alarming number of ACL injuries, as well as other significant pathology. Understanding the unique epidemiology, risk factors, treatment, and injury prevention strategies is critically important in helping medical professionals provide care for all levels of elite soccer players.
1. Sadigursky D, Braid JA, De Lira DNL, Machado BAB, Carneiro RJF, Colavolpe PO. The FIFA 11+ injury prevention program for soccer players: a systematic review. BMC Sports Sci Med Rehabil. 2017;9:18. doi:10.1186/s13102-017-0083-z.
2. Junge A, Dvorak J. Soccer injuries: a review on incidence and prevention. Sports Med. 2004;34(13):929-938. doi:10.2165/00007256-200434130-00004.
3. Hootman JM, Dick R, Agel J. Epidemiology of collegiate injuries for 15 sports: summary and recommendations for injury prevention initiatives. J Athl Train. 2007;42(2):311-319.
4. Agel J, Rockwood T, Klossner D. Collegiate ACL Injury rates across 15 sports: National collegiate athletic association injury surveillance system data update (2004-2005 Through 2012-2013). Clin J Sport Med. 2016;26(6):518-523. doi:10.1097/JSM.0000000000000290.
5. Kerr ZY, Pierpoint LA, Currie DW, Wasserman EB, Comstock RD. Epidemiologic comparisons of soccer-related injuries presenting to emergency departments and reported within high school and collegiate settings. Inj Epidemiol. 2017;4(1):19. doi:10.1186/s40621-017-0116-9.
6. Volpi P, Bisciotti GN, Chamari K, Cena E, Carimati G, Bragazzi NL. Risk factors of anterior cruciate ligament injury in football players: a systematic review of the literature. Muscles Ligaments Tendons J. 2016;6(4):480-485. doi:10.11138/mltj/2016.6.4.480.
7. Smith NA, Chounthirath T, Xiang H. Soccer-related injuries treated in emergency departments: 1990-2014. Pediatrics. 2016;138(4). doi:10.1542/peds.2016-0346.
8. Leininger RE, Knox CL, Comstock RD. Epidemiology of 1.6 million pediatric soccer-related injuries presenting to US emergency departments from 1990 to 2003. Am J Sports Med. 2007;35(2):288-293. doi:10.1177/0363546506294060.
9. Adams AL, Schiff MA. Childhood soccer injuries treated in U.S. emergency departments. Acad Emerg Med. 2006;13(5):571-574. doi:10.1197/j.aem.2005.12.015.
10. Woods C, Hawkins R, Hulse M, Hodson A. The football association medical research programme: an audit of injuries in professional football-analysis of preseason injuries. Br J Sports Med. 2002;36(6):436-441. doi:10.1136/bjsm.36.6.436.
11. Chomiak J, Junge A, Peterson L, Dvorak J. Severe injuries in football players. Influencing factors. Am J Sports Med. 2000;28(5 Suppl):S58-68. doi:10.1177/28.suppl_5.s-58.
12. Ostenberg A, Roos H. Injury risk factors in female European football. a prospective study of 123 players during one season. Scand J Med Sci Sports. 2000;10(5):279-285. doi:10.1034/j.1600-0838.2000.010005279.x.
13. Backous DD, Friedl KE, Smith NJ, Parr TJ, Carpine WD. Soccer injuries and their relation to physical maturity. Am J Dis Child. 1988;142(8):839-842. doi:10.1001/archpedi.1988.02150080045019.
14. Grimm NL, Jacobs JC, Kim J, Denney BS, Shea KG. Anterior cruciate ligament and knee injury prevention programs for soccer players: a systematic review and meta-analysis. Am J Sports Med. 2015;43(8):2049-2056. doi:10.1177/0363546514556737.
15. Dick R, Putukian M, Agel J, Evans TA, Marshall SW. Descriptive epidemiology of collegiate women's soccer injuries: National Collegiate Athletic Association Injury Surveillance System, 1988-1989 through 2002-2003. J Athl Train. 2007;42(2):278-285.
16. Renstrom P, Ljungqvist A, Arendt E, et al. Non-contact ACL injuries in female athletes: an International Olympic Committee current concepts statement. Br J Sports Med. 2008;42(6):394-412. doi:10.1136/bjsm.2008.048934.
17. Guskiewicz KM, Weaver NL, Padua DA, Garrett WE. Epidemiology of concussion in collegiate and high school football players. Am J Sports Med. 2000;28(5):643-650. doi:10.1177/03635465000280050401.
18. Levy IM, Skovron ML, Agel J. Living with artificial grass: a knowledge update. Part 1: Basic science. Am J Sports Med. 1990;18(4):406-412. doi:10.1177/036354659001800413.
19. Meyers MC. Incidence, Mechanisms, and severity of match-related collegiate men's soccer injuries on fieldturf and natural grass surfaces: a 6-year prospective study. Am J Sports Med. 2017;45(3):708-718. doi:10.1177/0363546516671715.
20. Ekstrand J, Hägglund M, Fuller CW. Comparison of injuries sustained on artificial turf and grass by male and female elite football players. Scand J Med Sci Sports. 2011;21(6):824-832. doi:10.1111/j.1600-0838.2010.01118.x.
21. Meyers MC. Incidence, mechanisms, and severity of match-related collegiate women's soccer injuries on FieldTurf and natural grass surfaces: a 5-year prospective study. Am J Sports Med. 2013;41(10):2409-2420. doi:10.1177/0363546513498994.
22. Dragoo JL, Braun HJ, Harris AH. The effect of playing surface on the incidence of ACL injuries in National Collegiate Athletic Association American Football. Knee. 2013;20(3):191-195. doi:10.1016/j.knee.2012.07.006.
23. Rothenberg P, Grau L, Kaplan L, Baraga MG. Knee injuries in american football: an epidemiological review. Am J Orthop. 2016;45(6):368-373.
24. Waldén M, Hägglund M, Magnusson H, Ekstrand J. Anterior cruciate ligament injury in elite football: a prospective three-cohort study. Knee Surg Sports Traumatol Arthrosc. 2011;19(1):11-19. doi:10.1007/s00167-010-1170-9.
25. Waldén M, Krosshaug T, Bjørneboe J, Andersen TE, Faul O, Hägglund M. Three distinct mechanisms predominate in non-contact anterior cruciate ligament injuries in male professional football players: a systematic video analysis of 39 cases. Br J Sports Med. 2015;49(22):1452-1460. doi:10.1136/bjsports-2014-094573.
26. Olsen OE, Myklebust G, Engebretsen L, Bahr R. Injury mechanisms for anterior cruciate ligament injuries in team handball: a systematic video analysis. Am J Sports Med. 2004;32(4):1002-1012. doi:10.1177/0363546503261724.
27. Giza E, Mithöfer K, Farrell L, Zarins B, Gill T. Injuries in women's professional soccer. Br J Sports Med. 2005;39(4):212-216; discussion 212-216. doi:10.1136/bjsm.2004.011973.
28. Grassi A, Smiley SP, Roberti di Sarsina T, et al. Mechanisms and situations of anterior cruciate ligament injuries in professional male soccer players: a YouTube-based video analysis. Eur J Orthop Surg Traumatol. 2017;27(7):967-981. doi:10.1007/s00590-017-1905-0.
29. Brophy RH, Stepan JG, Silvers HJ, Mandelbaum BR. Defending puts the anterior cruciate ligament at risk during soccer: a gender-based analysis. Sports Health. 2015;7(3):244-249. doi:10.1177/1941738114535184.
30. Faude O, Junge A, Kindermann W, Dvorak J. Risk factors for injuries in elite female soccer players. Br J Sports Med. 2006;40(9):785-790. doi:10.1136/bjsm.2006.027540.
31. Agel J, Arendt EA, Bershadsky B. Anterior cruciate ligament injury in national collegiate athletic association basketball and soccer: a 13-year review. Am J Sports Med. 2005;33(4):524-530. doi:10.1177/0363546504269937.
32. Gwinn DE, Wilckens JH, McDevitt ER, Ross G, Kao TC. The relative incidence of anterior cruciate ligament injury in men and women at the United States Naval Academy. Am J Sports Med. 2000;28(1):98-102. doi:10.1177/03635465000280012901.
33. Arendt E, Dick R. Knee injury patterns among men and women in collegiate basketball and soccer. NCAA data and review of literature. Am J Sports Med. 1995;23(6):694-701. doi:10.1177/036354659502300611.
34. Mihata LC, Beutler AI, Boden BP. Comparing the incidence of anterior cruciate ligament injury in collegiate lacrosse, soccer, and basketball players: implications for anterior cruciate ligament mechanism and prevention. Am J Sports Med. 2006;34(6):899-904. doi:10.1177/0363546505285582.
35. Prodromos CC, Han Y, Rogowski J, Joyce B, Shi K. A meta-analysis of the incidence of anterior cruciate ligament tears as a function of gender, sport, and a knee injury-reduction regimen. Arthroscopy. 2007;23(12):1320-1325.e1326. doi:10.1016/j.arthro.2007.07.003.
36. Allen MM, Pareek A, Krych AJ, et al. Are female soccer players at an increased risk of second anterior cruciate ligament injury compared with their athletic peers? Am J Sports Med. 2016;44(10):2492-2498. doi:10.1177/0363546516648439.
37. Farber J, Harris JD, Kolstad K, McCulloch PC. Treatment of anterior cruciate ligament injuries by major league soccer team physicians. Orthop J Sports Med. 2014;2(11):2325967114559892. doi:10.1177/2325967114559892.
38. Mascarenhas R, Tranovich M, Karpie JC, Irrgang JJ, Fu FH, Harner CD. Patellar tendon anterior cruciate ligament reconstruction in the high-demand patient: evaluation of autograft versus allograft reconstruction. Arthroscopy. 2010;26(9 Suppl):S58-66. doi:10.1016/j.arthro.2010.01.004.
39. Kaeding CC, Aros B, Pedroza A, et al. Allograft versus autograft anterior cruciate ligament reconstruction: predictors of failure from a MOON prospective longitudinal cohort. Sports Health. 2011;3(1):73-81. doi:10.1177/1941738110386185.
40. Pallis M, Svoboda SJ, Cameron KL, Owens BD. Survival comparison of allograft and autograft anterior cruciate ligament reconstruction at the United States Military Academy. Am J Sports Med. 2012;40(6):1242-1246. doi:10.1177/0363546512443945.
41. Gifstad T, Foss OA, Engebretsen L, et al. Lower risk of revision with patellar tendon autografts compared with hamstring autografts: a registry study based on 45,998 primary ACL reconstructions in Scandinavia. Am J Sports Med. 2014;42(10):2319-2328. doi:10.1177/0363546514548164.
42. Mohammadi F, Salavati M, Akhbari B, Mazaheri M, Mohsen Mir S, Etemadi Y. Comparison of functional outcome measures after ACL reconstruction in competitive soccer players: a randomized trial. J Bone Joint Surg Am. 2013;95(14):1271-1277. doi:10.2106/JBJS.L.00724.
43. van der List JP, DiFelice GS. Arthroscopic primary anterior cruciate ligament repair with suture augmentation. Arthrosc Tech. 2017;6(5):e1529-e1534. doi:10.1016/j.eats.2017.06.009.
44. Murray MM, Flutie BM, Kalish LA, et al. The bridge-enhanced anterior cruciate ligament repair (BEAR) procedure: an early feasibility cohort study. Orthop J Sports Med. 2016;4(11):2325967116672176. doi:10.1177/2325967116672176.
45. DiFelice GS, van der List JP. Clinical outcomes of arthroscopic primary repair of proximal anterior cruciate ligament tears are maintained at mid-term follow-up. Arthroscopy. 2018;34(4):1085-1093. doi:10.1016/j.arthro.2017.10.028.
46. Howard JS, Lembach ML, Metzler AV, Johnson DL. Rates and determinants of return to play after anterior cruciate ligament reconstruction in national collegiate athletic association division I soccer athletes: a study of the southeastern conference. Am J Sports Med. 2016;44(2):433-439. doi:10.1177/0363546515614315.
47. Erickson BJ, Harris JD, Cvetanovich GL, et al. Performance and return to sport after anterior cruciate ligament reconstruction in male major league soccer players. Orthop J Sports Med. 2013;1(2):2325967113497189. doi:10.1177/2325967113497189.
48. Arundale AJH, Silvers-Granelli HJ, Snyder-Mackler L. Career length and injury incidence after anterior cruciate ligament reconstruction in major league soccer players. Orthop J Sports Med. 2018;6(1):2325967117750825. doi:10.1177/2325967117750825.
49. Brophy RH, Schmitz L, Wright RW, et al. Return to play and future ACL injury risk after ACL reconstruction in soccer athletes from the Multicenter Orthopaedic Outcomes Network (MOON) group. Am J Sports Med. 2012;40(11):2517-2522. doi:10.1177/0363546512459476.
50. Zaffagnini S, Grassi A, Marcheggiani Muccioli GM, et al. Return to sport after anterior cruciate ligament reconstruction in professional soccer players. Knee. 2014;21(3):731-735. doi:10.1016/j.knee.2014.02.005.
51. Waldén M, Hägglund M, Magnusson H, Ekstrand J. ACL injuries in men's professional football: a 15-year prospective study on time trends and return-to-play rates reveals only 65% of players still play at the top level 3 years after ACL rupture. Br J Sports Med. 2016;50(12):744-750. doi:10.1136/bjsports-2015-095952.
52. Andernord D, Desai N, Björnsson H, Ylander M, Karlsson J, Samuelsson K. Patient predictors of early revision surgery after anterior cruciate ligament reconstruction: a cohort study of 16,930 patients with 2-year follow-up. Am J Sports Med. 2015;43(1):121-127. doi:10.1177/0363546514552788.
53. Kramer DE, Miller PE, Berrahou IK, Yen YM, Heyworth BE. Collateral ligament knee injuries in pediatric and adolescent athletes. J Pediatr Orthop. 2017. doi:10.1097/BPO.0000000000001112.
54. Krutsch W, Zeman F, Zellner J, Pfeifer C, Nerlich M, Angele P. Increase in ACL and PCL injuries after implementation of a new professional football league. Knee Surg Sports Traumatol Arthrosc. 2016;24(7):2271-2279. doi:10.1007/s00167-014-3357-y.
55. Lundblad M, Waldén M, Magnusson H, Karlsson J, Ekstrand J. The UEFA injury study: 11-year data concerning 346 MCL injuries and time to return to play. Br J Sports Med. 2013;47(12):759-762. doi:10.1136/bjsports-2013-092305.
56. Stanley LE, Kerr ZY, Dompier TP, Padua DA. Sex differences in the incidence of anterior cruciate ligament, medial collateral ligament, and meniscal injuries in collegiate and high school sports: 2009-2010 Through 2013-2014. Am J Sports Med. 2016;44(6):1565-1572. doi:10.1177/0363546516630927.
57. Lind M, Jakobsen BW, Lund B, Hansen MS, Abdallah O, Christiansen SE. Anatomical reconstruction of the medial collateral ligament and posteromedial corner of the knee in patients with chronic medial collateral ligament instability. Am J Sports Med. 2009;37(6):1116-1122. doi:10.1177/0363546509332498.
58. Wijdicks CA, Griffith CJ, Johansen S, Engebretsen L, LaPrade RF. Injuries to the medial collateral ligament and associated medial structures of the knee. J Bone Joint Surg Am. 2010;92(5):1266-1280. doi:10.2106/JBJS.I.01229.
59. Marchant MH, Tibor LM, Sekiya JK, Hardaker WT, Garrett WE, Taylor DC. Management of medial-sided knee injuries, part 1: medial collateral ligament. Am J Sports Med. 2011;39(5):1102-1113. doi:10.1177/0363546510385999.
60. Corten K, Hoser C, Fink C, Bellemans J. Case reports: a Stener-like lesion of the medial collateral ligament of the knee. Clin Orthop Relat Res. 2010;468(1):289-293. doi:10.1007/s11999-009-0992-6
61. Fanelli GC, Edson CJ. Posterior cruciate ligament injuries in trauma patients: Part II. Arthroscopy. 1995;11(5):526-529. doi:10.1016/0749-8063(95)90127-2.
62. Schulz MS, Russe K, Weiler A, Eichhorn HJ, Strobel MJ. Epidemiology of posterior cruciate ligament injuries. Arch Orthop Trauma Surg. 2003;123(4):186-191. doi:10.1007/s00402-002-0471-y.
63. Fowler PJ, Messieh SS. Isolated posterior cruciate ligament injuries in athletes. Am J Sports Med. 1987;15(6):553-557. doi:10.1177/036354658701500606.
64. Patel DV, Allen AA, Warren RF, Wickiewicz TL, Simonian PT. The nonoperative treatment of acute, isolated (partial or complete) posterior cruciate ligament-deficient knees: an intermediate-term follow-up study. HSS J. 2007;3(2):137-146. doi:10.1007/s11420-007-9058-z.
65. Owesen C, Sandven-Thrane S, Lind M, Forssblad M, Granan LP, Årøen A. Epidemiology of surgically treated posterior cruciate ligament injuries in Scandinavia. Knee Surg Sports Traumatol Arthrosc. 2017;25(8):2384-2391. doi:10.1007/s00167-015-3786-2.
66. LaPrade CM, Civitarese DM, Rasmussen MT, LaPrade RF. Emerging updates on the posterior cruciate ligament: a review of the current literature. Am J Sports Med. 2015;43(12):3077-3092. doi:10.1177/0363546515572770.
67. Anderson CL. High rate of osteoarthritis of the knee in former soccer players. Med Sci Sports Exerc. 1986;18(1):141.
68. Arliani GG, Astur DC, Yamada RK, et al. Early osteoarthritis and reduced quality of life after retirement in former professional soccer players. Clinics (Sao Paulo). 2014;69(9):589-594. doi:10.6061/clinics/2014(09)03.
69. Wong P, Hong Y. Soccer injury in the lower extremities. Br J Sports Med. 2005;39(8):473-482. doi:10.1136/bjsm.2004.015511.
70. Thelin N, Holmberg S, Thelin A. Knee injuries account for the sports-related increased risk of knee osteoarthritis. Scand J Med Sci Sports. 2006;16(5):329-333. doi:10.1111/j.1600-0838.2005.00497.x.
71. Lohmander LS, Englund PM, Dahl LL, Roos EM. The long-term consequence of anterior cruciate ligament and meniscus injuries: osteoarthritis. Am J Sports Med. 2007;35(10):1756-1769. doi:10.1177/0363546507307396.
72. Mithöfer K, Peterson L, Mandelbaum BR, Minas T. Articular cartilage repair in soccer players with autologous chondrocyte transplantation: functional outcome and return to competition. Am J Sports Med. 2005;33(11):1639-1646. doi:10.1177/0363546505275647
73. Steadman JR, Briggs KK, Rodrigo JJ, Kocher MS, Gill TJ, Rodkey WG. Outcomes of microfracture for traumatic chondral defects of the knee: average 11-year follow-up. Arthroscopy. 2003;19(5):477-484. doi:10.1053/jars.2003.50112.
74. Hangody L, Ráthonyi GK, Duska Z, Vásárhelyi G, Füles P, Módis L. Autologous osteochondral mosaicplasty. Surgical technique. J Bone Joint Surg Am. 2004;86-A Suppl 1:65-72.
75. Sherman SL, Garrity J, Bauer K, Cook J, Stannard J, Bugbee W. Fresh osteochondral allograft transplantation for the knee: current concepts. J Am Acad Orthop Surg. 2014;22(2):121-133. doi:10.5435/JAAOS-22-02-121.
76. Andrade R, Vasta S, Papalia R, et al. Prevalence of articular cartilage lesions and surgical clinical outcomes in football (soccer) players' knees: a systematic review. Arthroscopy. 2016;32(7):1466-1477. doi:10.1016/j.arthro.2016.01.055.
77. Görtz S, Williams RJ, Gersoff WK, Bugbee WD. Osteochondral and meniscal allograft transplantation in the football (soccer) player. Cartilage. 2012;3(1 Suppl):37S-42S. doi:10.1177/1947603511416974.
78. Junge A, Grimm K, Feddermann N, Dvorak J. Precompetition orthopedic assessment of international elite football players. Clin J Sport Med. 2009;19(4):326-328. doi:10.1097/JSM.0b013e3181b21b56.
79. Salzmann GM, Preiss S, Zenobi-Wong M, Harder LP, Maier D, Dvorák J. Osteoarthritis in Football. Cartilage. 2017;8(2):162-172. doi:10.1177/1947603516648186.
80. Makris EA, Hadidi P, Athanasiou KA. The knee meniscus: structure-function, pathophysiology, current repair techniques, and prospects for regeneration. Biomaterials. 2011;32(30):7411-7431. doi:10.1016/j.biomaterials.2011.06.037
81. Freutel M, Seitz AM, Ignatius A, Dürselen L. Influence of partial meniscectomy on attachment forces, superficial strain and contact mechanics in porcine knee joints. Knee Surg Sports Traumatol Arthrosc. 2015;23(1):74-82. doi:10.1007/s00167-014-2951-3.
82. Papalia R, Del Buono A, Osti L, Denaro V, Maffulli N. Meniscectomy as a risk factor for knee osteoarthritis: a systematic review. Br Med Bull. 2011;99:89-106. doi:10.1093/bmb/ldq043.
83. Nawabi DH, Cro S, Hamid IP, Williams A. Return to play after lateral meniscectomy compared with medial meniscectomy in elite professional soccer players. Am J Sports Med. 2014;42(9):2193-2198. doi:10.1177/0363546514540271.
84. Alentorn-Geli E, Vázquez RS, Díaz PA, Cuscó X, Cugat R. Arthroscopic meniscal transplants in soccer players: outcomes at 2- to 5-year follow-up. Clin J Sport Med. 2010;20(5):340-343. doi:10.1097/JSM.0b013e3181f207dc.
85. Marcacci M, Marcheggiani Muccioli GM, Grassi A, et al. Arthroscopic meniscus allograft transplantation in male professional soccer players: a 36-month follow-up study. Am J Sports Med. 2014;42(2):382-388. doi:10.1177/0363546513508763.
86. Bizzini M, Dvorak J. FIFA 11+: an effective programme to prevent football injuries in various player groups worldwide-a narrative review. Br J Sports Med. 2015;49(9):577-579. doi:10.1136/bjsports-2015-094765.
87. Junge A, Lamprecht M, Stamm H, et al. Countrywide campaign to prevent soccer injuries in Swiss amateur players. Am J Sports Med. 2011;39(1):57-63. doi:10.1177/0363546510377424.
88. Al Attar WSA, Soomro N, Pappas E, Sinclair PJ, Sanders RH. Adding a post-training FIFA 11+ exercise program to the pre-training FIFA 11+ injury prevention program reduces injury rates among male amateur soccer players: a cluster-randomised trial. J Physiother. 2017;63(4):235-242. doi:10.1016/j.jphys.2017.08.004.
1. Sadigursky D, Braid JA, De Lira DNL, Machado BAB, Carneiro RJF, Colavolpe PO. The FIFA 11+ injury prevention program for soccer players: a systematic review. BMC Sports Sci Med Rehabil. 2017;9:18. doi:10.1186/s13102-017-0083-z.
2. Junge A, Dvorak J. Soccer injuries: a review on incidence and prevention. Sports Med. 2004;34(13):929-938. doi:10.2165/00007256-200434130-00004.
3. Hootman JM, Dick R, Agel J. Epidemiology of collegiate injuries for 15 sports: summary and recommendations for injury prevention initiatives. J Athl Train. 2007;42(2):311-319.
4. Agel J, Rockwood T, Klossner D. Collegiate ACL Injury rates across 15 sports: National collegiate athletic association injury surveillance system data update (2004-2005 Through 2012-2013). Clin J Sport Med. 2016;26(6):518-523. doi:10.1097/JSM.0000000000000290.
5. Kerr ZY, Pierpoint LA, Currie DW, Wasserman EB, Comstock RD. Epidemiologic comparisons of soccer-related injuries presenting to emergency departments and reported within high school and collegiate settings. Inj Epidemiol. 2017;4(1):19. doi:10.1186/s40621-017-0116-9.
6. Volpi P, Bisciotti GN, Chamari K, Cena E, Carimati G, Bragazzi NL. Risk factors of anterior cruciate ligament injury in football players: a systematic review of the literature. Muscles Ligaments Tendons J. 2016;6(4):480-485. doi:10.11138/mltj/2016.6.4.480.
7. Smith NA, Chounthirath T, Xiang H. Soccer-related injuries treated in emergency departments: 1990-2014. Pediatrics. 2016;138(4). doi:10.1542/peds.2016-0346.
8. Leininger RE, Knox CL, Comstock RD. Epidemiology of 1.6 million pediatric soccer-related injuries presenting to US emergency departments from 1990 to 2003. Am J Sports Med. 2007;35(2):288-293. doi:10.1177/0363546506294060.
9. Adams AL, Schiff MA. Childhood soccer injuries treated in U.S. emergency departments. Acad Emerg Med. 2006;13(5):571-574. doi:10.1197/j.aem.2005.12.015.
10. Woods C, Hawkins R, Hulse M, Hodson A. The football association medical research programme: an audit of injuries in professional football-analysis of preseason injuries. Br J Sports Med. 2002;36(6):436-441. doi:10.1136/bjsm.36.6.436.
11. Chomiak J, Junge A, Peterson L, Dvorak J. Severe injuries in football players. Influencing factors. Am J Sports Med. 2000;28(5 Suppl):S58-68. doi:10.1177/28.suppl_5.s-58.
12. Ostenberg A, Roos H. Injury risk factors in female European football. a prospective study of 123 players during one season. Scand J Med Sci Sports. 2000;10(5):279-285. doi:10.1034/j.1600-0838.2000.010005279.x.
13. Backous DD, Friedl KE, Smith NJ, Parr TJ, Carpine WD. Soccer injuries and their relation to physical maturity. Am J Dis Child. 1988;142(8):839-842. doi:10.1001/archpedi.1988.02150080045019.
14. Grimm NL, Jacobs JC, Kim J, Denney BS, Shea KG. Anterior cruciate ligament and knee injury prevention programs for soccer players: a systematic review and meta-analysis. Am J Sports Med. 2015;43(8):2049-2056. doi:10.1177/0363546514556737.
15. Dick R, Putukian M, Agel J, Evans TA, Marshall SW. Descriptive epidemiology of collegiate women's soccer injuries: National Collegiate Athletic Association Injury Surveillance System, 1988-1989 through 2002-2003. J Athl Train. 2007;42(2):278-285.
16. Renstrom P, Ljungqvist A, Arendt E, et al. Non-contact ACL injuries in female athletes: an International Olympic Committee current concepts statement. Br J Sports Med. 2008;42(6):394-412. doi:10.1136/bjsm.2008.048934.
17. Guskiewicz KM, Weaver NL, Padua DA, Garrett WE. Epidemiology of concussion in collegiate and high school football players. Am J Sports Med. 2000;28(5):643-650. doi:10.1177/03635465000280050401.
18. Levy IM, Skovron ML, Agel J. Living with artificial grass: a knowledge update. Part 1: Basic science. Am J Sports Med. 1990;18(4):406-412. doi:10.1177/036354659001800413.
19. Meyers MC. Incidence, Mechanisms, and severity of match-related collegiate men's soccer injuries on fieldturf and natural grass surfaces: a 6-year prospective study. Am J Sports Med. 2017;45(3):708-718. doi:10.1177/0363546516671715.
20. Ekstrand J, Hägglund M, Fuller CW. Comparison of injuries sustained on artificial turf and grass by male and female elite football players. Scand J Med Sci Sports. 2011;21(6):824-832. doi:10.1111/j.1600-0838.2010.01118.x.
21. Meyers MC. Incidence, mechanisms, and severity of match-related collegiate women's soccer injuries on FieldTurf and natural grass surfaces: a 5-year prospective study. Am J Sports Med. 2013;41(10):2409-2420. doi:10.1177/0363546513498994.
22. Dragoo JL, Braun HJ, Harris AH. The effect of playing surface on the incidence of ACL injuries in National Collegiate Athletic Association American Football. Knee. 2013;20(3):191-195. doi:10.1016/j.knee.2012.07.006.
23. Rothenberg P, Grau L, Kaplan L, Baraga MG. Knee injuries in american football: an epidemiological review. Am J Orthop. 2016;45(6):368-373.
24. Waldén M, Hägglund M, Magnusson H, Ekstrand J. Anterior cruciate ligament injury in elite football: a prospective three-cohort study. Knee Surg Sports Traumatol Arthrosc. 2011;19(1):11-19. doi:10.1007/s00167-010-1170-9.
25. Waldén M, Krosshaug T, Bjørneboe J, Andersen TE, Faul O, Hägglund M. Three distinct mechanisms predominate in non-contact anterior cruciate ligament injuries in male professional football players: a systematic video analysis of 39 cases. Br J Sports Med. 2015;49(22):1452-1460. doi:10.1136/bjsports-2014-094573.
26. Olsen OE, Myklebust G, Engebretsen L, Bahr R. Injury mechanisms for anterior cruciate ligament injuries in team handball: a systematic video analysis. Am J Sports Med. 2004;32(4):1002-1012. doi:10.1177/0363546503261724.
27. Giza E, Mithöfer K, Farrell L, Zarins B, Gill T. Injuries in women's professional soccer. Br J Sports Med. 2005;39(4):212-216; discussion 212-216. doi:10.1136/bjsm.2004.011973.
28. Grassi A, Smiley SP, Roberti di Sarsina T, et al. Mechanisms and situations of anterior cruciate ligament injuries in professional male soccer players: a YouTube-based video analysis. Eur J Orthop Surg Traumatol. 2017;27(7):967-981. doi:10.1007/s00590-017-1905-0.
29. Brophy RH, Stepan JG, Silvers HJ, Mandelbaum BR. Defending puts the anterior cruciate ligament at risk during soccer: a gender-based analysis. Sports Health. 2015;7(3):244-249. doi:10.1177/1941738114535184.
30. Faude O, Junge A, Kindermann W, Dvorak J. Risk factors for injuries in elite female soccer players. Br J Sports Med. 2006;40(9):785-790. doi:10.1136/bjsm.2006.027540.
31. Agel J, Arendt EA, Bershadsky B. Anterior cruciate ligament injury in national collegiate athletic association basketball and soccer: a 13-year review. Am J Sports Med. 2005;33(4):524-530. doi:10.1177/0363546504269937.
32. Gwinn DE, Wilckens JH, McDevitt ER, Ross G, Kao TC. The relative incidence of anterior cruciate ligament injury in men and women at the United States Naval Academy. Am J Sports Med. 2000;28(1):98-102. doi:10.1177/03635465000280012901.
33. Arendt E, Dick R. Knee injury patterns among men and women in collegiate basketball and soccer. NCAA data and review of literature. Am J Sports Med. 1995;23(6):694-701. doi:10.1177/036354659502300611.
34. Mihata LC, Beutler AI, Boden BP. Comparing the incidence of anterior cruciate ligament injury in collegiate lacrosse, soccer, and basketball players: implications for anterior cruciate ligament mechanism and prevention. Am J Sports Med. 2006;34(6):899-904. doi:10.1177/0363546505285582.
35. Prodromos CC, Han Y, Rogowski J, Joyce B, Shi K. A meta-analysis of the incidence of anterior cruciate ligament tears as a function of gender, sport, and a knee injury-reduction regimen. Arthroscopy. 2007;23(12):1320-1325.e1326. doi:10.1016/j.arthro.2007.07.003.
36. Allen MM, Pareek A, Krych AJ, et al. Are female soccer players at an increased risk of second anterior cruciate ligament injury compared with their athletic peers? Am J Sports Med. 2016;44(10):2492-2498. doi:10.1177/0363546516648439.
37. Farber J, Harris JD, Kolstad K, McCulloch PC. Treatment of anterior cruciate ligament injuries by major league soccer team physicians. Orthop J Sports Med. 2014;2(11):2325967114559892. doi:10.1177/2325967114559892.
38. Mascarenhas R, Tranovich M, Karpie JC, Irrgang JJ, Fu FH, Harner CD. Patellar tendon anterior cruciate ligament reconstruction in the high-demand patient: evaluation of autograft versus allograft reconstruction. Arthroscopy. 2010;26(9 Suppl):S58-66. doi:10.1016/j.arthro.2010.01.004.
39. Kaeding CC, Aros B, Pedroza A, et al. Allograft versus autograft anterior cruciate ligament reconstruction: predictors of failure from a MOON prospective longitudinal cohort. Sports Health. 2011;3(1):73-81. doi:10.1177/1941738110386185.
40. Pallis M, Svoboda SJ, Cameron KL, Owens BD. Survival comparison of allograft and autograft anterior cruciate ligament reconstruction at the United States Military Academy. Am J Sports Med. 2012;40(6):1242-1246. doi:10.1177/0363546512443945.
41. Gifstad T, Foss OA, Engebretsen L, et al. Lower risk of revision with patellar tendon autografts compared with hamstring autografts: a registry study based on 45,998 primary ACL reconstructions in Scandinavia. Am J Sports Med. 2014;42(10):2319-2328. doi:10.1177/0363546514548164.
42. Mohammadi F, Salavati M, Akhbari B, Mazaheri M, Mohsen Mir S, Etemadi Y. Comparison of functional outcome measures after ACL reconstruction in competitive soccer players: a randomized trial. J Bone Joint Surg Am. 2013;95(14):1271-1277. doi:10.2106/JBJS.L.00724.
43. van der List JP, DiFelice GS. Arthroscopic primary anterior cruciate ligament repair with suture augmentation. Arthrosc Tech. 2017;6(5):e1529-e1534. doi:10.1016/j.eats.2017.06.009.
44. Murray MM, Flutie BM, Kalish LA, et al. The bridge-enhanced anterior cruciate ligament repair (BEAR) procedure: an early feasibility cohort study. Orthop J Sports Med. 2016;4(11):2325967116672176. doi:10.1177/2325967116672176.
45. DiFelice GS, van der List JP. Clinical outcomes of arthroscopic primary repair of proximal anterior cruciate ligament tears are maintained at mid-term follow-up. Arthroscopy. 2018;34(4):1085-1093. doi:10.1016/j.arthro.2017.10.028.
46. Howard JS, Lembach ML, Metzler AV, Johnson DL. Rates and determinants of return to play after anterior cruciate ligament reconstruction in national collegiate athletic association division I soccer athletes: a study of the southeastern conference. Am J Sports Med. 2016;44(2):433-439. doi:10.1177/0363546515614315.
47. Erickson BJ, Harris JD, Cvetanovich GL, et al. Performance and return to sport after anterior cruciate ligament reconstruction in male major league soccer players. Orthop J Sports Med. 2013;1(2):2325967113497189. doi:10.1177/2325967113497189.
48. Arundale AJH, Silvers-Granelli HJ, Snyder-Mackler L. Career length and injury incidence after anterior cruciate ligament reconstruction in major league soccer players. Orthop J Sports Med. 2018;6(1):2325967117750825. doi:10.1177/2325967117750825.
49. Brophy RH, Schmitz L, Wright RW, et al. Return to play and future ACL injury risk after ACL reconstruction in soccer athletes from the Multicenter Orthopaedic Outcomes Network (MOON) group. Am J Sports Med. 2012;40(11):2517-2522. doi:10.1177/0363546512459476.
50. Zaffagnini S, Grassi A, Marcheggiani Muccioli GM, et al. Return to sport after anterior cruciate ligament reconstruction in professional soccer players. Knee. 2014;21(3):731-735. doi:10.1016/j.knee.2014.02.005.
51. Waldén M, Hägglund M, Magnusson H, Ekstrand J. ACL injuries in men's professional football: a 15-year prospective study on time trends and return-to-play rates reveals only 65% of players still play at the top level 3 years after ACL rupture. Br J Sports Med. 2016;50(12):744-750. doi:10.1136/bjsports-2015-095952.
52. Andernord D, Desai N, Björnsson H, Ylander M, Karlsson J, Samuelsson K. Patient predictors of early revision surgery after anterior cruciate ligament reconstruction: a cohort study of 16,930 patients with 2-year follow-up. Am J Sports Med. 2015;43(1):121-127. doi:10.1177/0363546514552788.
53. Kramer DE, Miller PE, Berrahou IK, Yen YM, Heyworth BE. Collateral ligament knee injuries in pediatric and adolescent athletes. J Pediatr Orthop. 2017. doi:10.1097/BPO.0000000000001112.
54. Krutsch W, Zeman F, Zellner J, Pfeifer C, Nerlich M, Angele P. Increase in ACL and PCL injuries after implementation of a new professional football league. Knee Surg Sports Traumatol Arthrosc. 2016;24(7):2271-2279. doi:10.1007/s00167-014-3357-y.
55. Lundblad M, Waldén M, Magnusson H, Karlsson J, Ekstrand J. The UEFA injury study: 11-year data concerning 346 MCL injuries and time to return to play. Br J Sports Med. 2013;47(12):759-762. doi:10.1136/bjsports-2013-092305.
56. Stanley LE, Kerr ZY, Dompier TP, Padua DA. Sex differences in the incidence of anterior cruciate ligament, medial collateral ligament, and meniscal injuries in collegiate and high school sports: 2009-2010 Through 2013-2014. Am J Sports Med. 2016;44(6):1565-1572. doi:10.1177/0363546516630927.
57. Lind M, Jakobsen BW, Lund B, Hansen MS, Abdallah O, Christiansen SE. Anatomical reconstruction of the medial collateral ligament and posteromedial corner of the knee in patients with chronic medial collateral ligament instability. Am J Sports Med. 2009;37(6):1116-1122. doi:10.1177/0363546509332498.
58. Wijdicks CA, Griffith CJ, Johansen S, Engebretsen L, LaPrade RF. Injuries to the medial collateral ligament and associated medial structures of the knee. J Bone Joint Surg Am. 2010;92(5):1266-1280. doi:10.2106/JBJS.I.01229.
59. Marchant MH, Tibor LM, Sekiya JK, Hardaker WT, Garrett WE, Taylor DC. Management of medial-sided knee injuries, part 1: medial collateral ligament. Am J Sports Med. 2011;39(5):1102-1113. doi:10.1177/0363546510385999.
60. Corten K, Hoser C, Fink C, Bellemans J. Case reports: a Stener-like lesion of the medial collateral ligament of the knee. Clin Orthop Relat Res. 2010;468(1):289-293. doi:10.1007/s11999-009-0992-6
61. Fanelli GC, Edson CJ. Posterior cruciate ligament injuries in trauma patients: Part II. Arthroscopy. 1995;11(5):526-529. doi:10.1016/0749-8063(95)90127-2.
62. Schulz MS, Russe K, Weiler A, Eichhorn HJ, Strobel MJ. Epidemiology of posterior cruciate ligament injuries. Arch Orthop Trauma Surg. 2003;123(4):186-191. doi:10.1007/s00402-002-0471-y.
63. Fowler PJ, Messieh SS. Isolated posterior cruciate ligament injuries in athletes. Am J Sports Med. 1987;15(6):553-557. doi:10.1177/036354658701500606.
64. Patel DV, Allen AA, Warren RF, Wickiewicz TL, Simonian PT. The nonoperative treatment of acute, isolated (partial or complete) posterior cruciate ligament-deficient knees: an intermediate-term follow-up study. HSS J. 2007;3(2):137-146. doi:10.1007/s11420-007-9058-z.
65. Owesen C, Sandven-Thrane S, Lind M, Forssblad M, Granan LP, Årøen A. Epidemiology of surgically treated posterior cruciate ligament injuries in Scandinavia. Knee Surg Sports Traumatol Arthrosc. 2017;25(8):2384-2391. doi:10.1007/s00167-015-3786-2.
66. LaPrade CM, Civitarese DM, Rasmussen MT, LaPrade RF. Emerging updates on the posterior cruciate ligament: a review of the current literature. Am J Sports Med. 2015;43(12):3077-3092. doi:10.1177/0363546515572770.
67. Anderson CL. High rate of osteoarthritis of the knee in former soccer players. Med Sci Sports Exerc. 1986;18(1):141.
68. Arliani GG, Astur DC, Yamada RK, et al. Early osteoarthritis and reduced quality of life after retirement in former professional soccer players. Clinics (Sao Paulo). 2014;69(9):589-594. doi:10.6061/clinics/2014(09)03.
69. Wong P, Hong Y. Soccer injury in the lower extremities. Br J Sports Med. 2005;39(8):473-482. doi:10.1136/bjsm.2004.015511.
70. Thelin N, Holmberg S, Thelin A. Knee injuries account for the sports-related increased risk of knee osteoarthritis. Scand J Med Sci Sports. 2006;16(5):329-333. doi:10.1111/j.1600-0838.2005.00497.x.
71. Lohmander LS, Englund PM, Dahl LL, Roos EM. The long-term consequence of anterior cruciate ligament and meniscus injuries: osteoarthritis. Am J Sports Med. 2007;35(10):1756-1769. doi:10.1177/0363546507307396.
72. Mithöfer K, Peterson L, Mandelbaum BR, Minas T. Articular cartilage repair in soccer players with autologous chondrocyte transplantation: functional outcome and return to competition. Am J Sports Med. 2005;33(11):1639-1646. doi:10.1177/0363546505275647
73. Steadman JR, Briggs KK, Rodrigo JJ, Kocher MS, Gill TJ, Rodkey WG. Outcomes of microfracture for traumatic chondral defects of the knee: average 11-year follow-up. Arthroscopy. 2003;19(5):477-484. doi:10.1053/jars.2003.50112.
74. Hangody L, Ráthonyi GK, Duska Z, Vásárhelyi G, Füles P, Módis L. Autologous osteochondral mosaicplasty. Surgical technique. J Bone Joint Surg Am. 2004;86-A Suppl 1:65-72.
75. Sherman SL, Garrity J, Bauer K, Cook J, Stannard J, Bugbee W. Fresh osteochondral allograft transplantation for the knee: current concepts. J Am Acad Orthop Surg. 2014;22(2):121-133. doi:10.5435/JAAOS-22-02-121.
76. Andrade R, Vasta S, Papalia R, et al. Prevalence of articular cartilage lesions and surgical clinical outcomes in football (soccer) players' knees: a systematic review. Arthroscopy. 2016;32(7):1466-1477. doi:10.1016/j.arthro.2016.01.055.
77. Görtz S, Williams RJ, Gersoff WK, Bugbee WD. Osteochondral and meniscal allograft transplantation in the football (soccer) player. Cartilage. 2012;3(1 Suppl):37S-42S. doi:10.1177/1947603511416974.
78. Junge A, Grimm K, Feddermann N, Dvorak J. Precompetition orthopedic assessment of international elite football players. Clin J Sport Med. 2009;19(4):326-328. doi:10.1097/JSM.0b013e3181b21b56.
79. Salzmann GM, Preiss S, Zenobi-Wong M, Harder LP, Maier D, Dvorák J. Osteoarthritis in Football. Cartilage. 2017;8(2):162-172. doi:10.1177/1947603516648186.
80. Makris EA, Hadidi P, Athanasiou KA. The knee meniscus: structure-function, pathophysiology, current repair techniques, and prospects for regeneration. Biomaterials. 2011;32(30):7411-7431. doi:10.1016/j.biomaterials.2011.06.037
81. Freutel M, Seitz AM, Ignatius A, Dürselen L. Influence of partial meniscectomy on attachment forces, superficial strain and contact mechanics in porcine knee joints. Knee Surg Sports Traumatol Arthrosc. 2015;23(1):74-82. doi:10.1007/s00167-014-2951-3.
82. Papalia R, Del Buono A, Osti L, Denaro V, Maffulli N. Meniscectomy as a risk factor for knee osteoarthritis: a systematic review. Br Med Bull. 2011;99:89-106. doi:10.1093/bmb/ldq043.
83. Nawabi DH, Cro S, Hamid IP, Williams A. Return to play after lateral meniscectomy compared with medial meniscectomy in elite professional soccer players. Am J Sports Med. 2014;42(9):2193-2198. doi:10.1177/0363546514540271.
84. Alentorn-Geli E, Vázquez RS, Díaz PA, Cuscó X, Cugat R. Arthroscopic meniscal transplants in soccer players: outcomes at 2- to 5-year follow-up. Clin J Sport Med. 2010;20(5):340-343. doi:10.1097/JSM.0b013e3181f207dc.
85. Marcacci M, Marcheggiani Muccioli GM, Grassi A, et al. Arthroscopic meniscus allograft transplantation in male professional soccer players: a 36-month follow-up study. Am J Sports Med. 2014;42(2):382-388. doi:10.1177/0363546513508763.
86. Bizzini M, Dvorak J. FIFA 11+: an effective programme to prevent football injuries in various player groups worldwide-a narrative review. Br J Sports Med. 2015;49(9):577-579. doi:10.1136/bjsports-2015-094765.
87. Junge A, Lamprecht M, Stamm H, et al. Countrywide campaign to prevent soccer injuries in Swiss amateur players. Am J Sports Med. 2011;39(1):57-63. doi:10.1177/0363546510377424.
88. Al Attar WSA, Soomro N, Pappas E, Sinclair PJ, Sanders RH. Adding a post-training FIFA 11+ exercise program to the pre-training FIFA 11+ injury prevention program reduces injury rates among male amateur soccer players: a cluster-randomised trial. J Physiother. 2017;63(4):235-242. doi:10.1016/j.jphys.2017.08.004.
TAKE-HOME POINTS
- Soccer is one of the most popular sports in the world and has a high incidence of resultant knee injuries.
- Significant, identifiable risk factors put soccer players at risk for serious knee injuries, such as ACL ruptures; age, female sex, and position played influence injury susceptibility.
- ACL injury most commonly occurs via non-contact mechanisms, and female players are at a significantly higher risk of ACL injury than male counterparts.
- The prevalence of osteoarthritis in retired soccer players is high, underscoring the need to be familiar with meniscal and cartilage repair/restoration techniques and associated outcomes.
- The FIFA11+ program reduces injury by 30%, with reported relative risk of 0.70 for lower limb injuries, highlighting the significant preventative importance of this warm-up program.
Diagnosing placenta accreta spectrum with prenatal ultrasound
Placenta accreta spectrum (PAS) describes abnormal invasion of placental tissue into or through the myometrium, comprising 3 distinct conditions: placenta accreta, placenta increta, and placenta percreta. This complication is relatively new to obstetrics, first described in 1937.1
The overall incidence of PAS has been increasing over several decades, in parallel to an increasing rate of cesarean delivery (CD), with an incidence from 1982 through 2002 of 1 in 533 pregnancies, representing a 5-fold increase since the 1980s.2 PAS is associated with significant morbidity and mortality, including fetal growth restriction, preterm delivery, placental abruption antenatally, and hemorrhage during delivery or postpartum.
Prenatal diagnosis of PAS and planned delivery at an experienced center are associated with significant reduction in maternal and fetal morbidity.3 In an era of advanced imaging modalities, prenatal detection of PAS regrettably remains variable and largely subjective: As many as 20% to 50% of cases of PAS escape prenatal diagnosis.3,4
In this article, we review the sonographic markers of PAS, including diagnostic accuracy, and propose a standardized approach to prenatal diagnosis. Throughout our discussion, we describe protocols for detection of PAS practiced at our Maternal-Fetal Medicine Program in the Department of Obstetrics and Gynecology, Eastern Virginia Medical School (also see “US evaluation of PAS risk: The authors’ recommended approach”).
Numerous risk factors
There are many risk factors for PAS, including prior uterine surgery or instrumentation, such as CD, uterine curettage, myomectomy, pelvic radiation, and endometrial ablation. Other risk factors include smoking, in vitro fertilization, advanced maternal age, multiparity, and a brief interval between prior CD and subsequent pregnancy.5 Of major significance is the increased risk of PAS in the presence of placenta previa with prior CD.6 Knowledge of clinical risk factors by the interpreting physician appears to be associated with improved detection of PAS on ultrasonography (US).4
Ultrasonographic markers of PAS
First-trimester markers
Sonographic markers of PAS in the first trimester include:
- a gestational sac implanted in the lower uterine segment or in a CD scar
- multiple hypoechoic spaces within the placenta (lacunae).7
Lower uterine-segment implantation has been defined by Ballas and colleagues as 1) a gestational sac implanted in the lower one-third of the uterus between 8 and 10 weeks’ gestation or 2) a gestational sac occupying primarily the lower uterine segment from 10 weeks’ gestation onward (FIGURE 1).8 Our experience is that it is difficult to accurately assess lower uterine-segment implantation beyond 13 weeks of gestation because the sac typically expands to fill the upper uterine cavity.
Continue to: Color Doppler US...
Color Doppler US can help differentiate lower uterine-segment implantation from a gestational sac of a failed pregnancy in the process of expulsion by demonstrating loss of circumferential blood flow in the failed pregnancy. Furthermore, applying pressure to the anterior surface of the uterus will result in downward movement of the gestational sac of a failed pregnancy.9
Not all gestational sacs that implant in the lower uterine segment lead to PAS: Subsequent normal pregnancies have been reported in this circumstance. In such cases, a normal thick myometrium is noted anterior to the gestational sac.7 A patient with lower uterine-segment implantation without evidence of anterior myometrial thinning remains at risk for third-trimester placenta previa.7
Cesarean scar pregnancy carries significant risk of PAS. In these cases, the gestational sac is typically implanted within the scar, resulting in a thin anterior myometrium and significantly increased vascularity of the placental–myometrial and bladder–uterine wall interfaces (FIGURE 2).9 Differentiating cesarean scar pregnancy from a lower uterine-segment implantation is easier to perform before the eighth week of gestation but becomes more difficult as pregnancy advances. Although it might be useful to distinguish between true cesarean scar pregnancy and lower uterine-segment implantation adjacent to or involving the scar, both carry considerable risk of PAS and excessive hemorrhage, and the approach to treating both conditions is quite similar.
Lacunae, with or without documented blood flow on color Doppler US, are the third marker of PAS in the first trimester.8 Although some retrospective series and case reports describe the finding of lacunae in the first trimester of patients with diagnosed PAS, more recent literature suggests that these spaces are seen infrequently and at a similar frequency in women with and without PAS at delivery.7
Second- and third-trimester markers
Multiple diagnostic sonographic markers of PAS have been described in the second and third trimesters.
Placental location is a significant risk factor for PAS. Placenta previa in the setting of prior CD carries the highest risk of PAS—as high as 61% in women with both placenta previa and a history of 3 CDs.10 An anterior placenta appears to be a stronger risk factor for PAS than a posterior placenta in women with prior CD; the location of the placenta should therefore be evaluated in all women in the second trimester.
Continue to: Lacunae
Lacunae. The finding of multiple hypoechoic vascular spaces within the placental parenchyma has been associated with PAS (FIGURES 3 and 4). The pathogenesis of this finding is probably related to alterations in placental tissue resulting from long-term exposure to pulsatile blood flow.11
Finberg and colleagues introduced a grading system for placental lacunae in 1992 that is still used:
- Grade 0: no lacunae seen
- Grade 1: 1 to 3 lacunae seen
- Grade 2: 4 to 6 lacunae seen
- Grade 3: multiple lacunae seen throughout the placenta.12
The sensitivity and specificity of lacunae as an independent marker for PAS have been reported to be 77% and 95%, respectively.13 Despite these findings, several studies report a range of sensitivity (73% to 100%) and negative predictive value (88% to 100%).14 Even in Finberg’s original work, 27% of cases of confirmed PAS had Grade 0 or Grade 1 placental lacunae and 11% of cases of placenta previa, without PAS, demonstrated Grade 2 lacunae.12 There is agreement, however, that, the more lacunae, the higher the risk of PAS.
Continue to: Other US markers for PAS
Other US markers of PAS
Retroplacental–myometrial interface

Loss of the normal hypoechoic (clear) retroplacental zone, also referred to as loss of the clear space between placenta and uterus, is another marker of PAS (FIGURE 5). This finding corresponds to pathologic loss of the decidua basalis as trophoblastic tissue invades directly through the myometrium.15 This sonographic finding has been reported to have a detection rate of approximately 93%, with sensitivity of 52% and specificity of 57%, for PAS; the false-positive rate, however, has been in the range of 21% or higher. This marker should not be used alone because it is angle-dependent and can be found (as an absent clear zone) in normal anterior placentas.16
The strength of this US marker is in its negative predictive value, which ranges from 96% to 100%. The presence of a hypoechoic retroplacental clear space that extends the length of the placenta makes PAS unlikely.17 Of note, the clear zone may appear falsely absent as a result of increased pressure from the US probe.
Retroplacental myometrial thickness
Retroplacental myometrial thickness is difficult to assess because the lower uterine-segment myometrium thins in normal pregnancy as term approaches. This measurement also can be influenced by direct pressure of the US probe and fullness of the maternal bladder.18 In patients who have had a CD but who do not have PAS, the median myometrial thickness of the lower uterine segment in the third trimester is 2.4 mm.19
Thinning of the myometrium in the upper uterine segment always should be of concern. Studies of this marker have reported sensitivity of US ranging from 22% to 100% and specificity from 72% to 100%.9,20 Given such variability, it is important to standardize the gestational age and sonographic approach for this marker.
Continue to: Uterovesical interface
Uterovesical interface
Studies also have reported that abnormalities of the uterovesical interface are predictive of PAS. The uterovesical interface is best evaluated in a sagittal plane containing the lower uterine segment and a partially full bladder in gray-scale and color Doppler US.15 The normal uterovesical interface appears as a smooth line, without irregularities or increased vascularity on sagittal imaging.
Abnormalities include focal interruption of the hyperechoic bladder wall, bulging of the bladder wall, and increased vascularity, such as varicosities (FIGURES 5, 6, and 7).15 These findings may be seen as early as the first trimester but are more commonly noted in the second and third trimesters.7 The authors of a recent meta-analysis concluded that irregularity of the uterovesical interface is the most specific marker for invasive placentation (99.75% confidence interval; range, 99.5% to 99.9%).13
Other US markers and modalities
Three-dimensional US. Studies have evaluated the role of 3-dimensional (3D) US for predicting PAS. Application of 3D US in vascular mode has shown promise because it allows for semiquantitative assessment of placental vasculature.22 Using 3D US to screen for PAS presents drawbacks, however: The technology is not well-standardized and requires significant operator expertise for volume acquisition and manipulation. Prospective studies are needed before 3D US can be applied routinely to screen for and diagnose PAS.
Color Doppler US. As an adjunct to gray-scale US, color Doppler US can be used for making a diagnosis of PAS. Color Doppler US helps differentiate a normal subplacental venous complex with nonpulsatile, low-velocity venous blood flow waveforms from markedly dilated peripheral subplacental vascular channels with pulsatile venous-type flow, which suggests PAS. These vascular channels are often located directly over the cervix. In addition, the observation of bridging vessels linking the placenta and bladder with high diastolic arterial blood flow also suggests invasion.21 In a meta-analysis, overall sensitivity of color Doppler US for the diagnosis of PAS was 91%, with specificity of 87%.13
The value of utilizing multiple markers
The accuracy of US diagnosis of PAS is likely improved by using more than 1 sonographic marker. Pilloni and colleagues,20 in a prospective analysis, found that 81% of cases of confirmed PAS had ≥2 markers and 51% of cases had ≥3 markers.
Several scoring systems have been proposed for making the diagnosis of PAS using combinations of sonographic markers, placental location, and clinical history.19,24,25 In 2016, Tovbin and colleagues,25 in a prospective study, evaluated a scoring system that included:
- number of previous CDs
- number of, maximum dimension of, and presence of blood flow in lacunae
- loss of uteroplacental clear zone
- placental location
- hypervascularity of the uterovesical or uteroplacental interface.
Tovbin assigned 1 or 2 points to each criterion. Each sonographic marker was found to be significantly associated with PAS when compared to a high-risk control group. A score of ≥8 was considered “at high risk” and predicted 69% of PAS cases.
Regrettably, no combination of US markers reliably predicts the depth of invasion of the placenta.26
Continue to: A standardized approach is needed
A standardized approach is needed
To decrease variability and improve the US diagnosis of PAS, it is important to define and standardize the diagnosis of each sonographic marker for PAS.4 In 2016, the European Working Group on Abnormally Invasive Placenta (EW-AIP) proposed a set of US markers that always should be reported when performing an US examination for suspected abnormal placentation (TABLE).23 Despite this effort by the EW-AIP, ambiguity remains over sonographic definitions of several PAS markers. For example, what determines a placental lacuna on US? And what constitutes an abnormal uterovesical interface? There is a need for a more objective definition of US markers of PAS and a standardized approach to the US examination in at-risk pregnancies.
The Society for Maternal-Fetal Medicine is coordinating a multi-society task force to address the need to define and standardize the US diagnosis of PAS.
Observations on other PAS diagnostic modalities
Magnetic resonance imaging
Adjunctive role. Magnetic resonance imaging (MRI) is often used as an adjunctive diagnostic modality in cases of suspected PAS. Several markers for PAS have been described on MRI, including15:
- intraplacental T2-weighted dark bands
- abnormal intraplacental vascularity
- heterogeneous intraplacental signal intensity
- focal interruption of the myometrium by the placenta
- uterine bulging.
- Assess a priori risk for the patient before initiating the US exam
- In the presence of a placenta previa, or low-lying placenta, we strongly recommend a transvaginal, in addition to transabdominal, US to further assess for the presence of placenta accreta spectrum (PAS) markers
- Until prospective studies clearly define the diagnostic accuracy of PAS sonographic markers and their performance in high-risk and low-risk pregnancies, we recommend that US findings be reported as a risk profile—that is, high, moderate, and low risk of PAS
- Be especially cautious with patients who are at substantially increased risk for PAS, such as those with placenta previa and prior multiple CDs. In this setting, a low-risk report for PAS only should be provided when none of the PAS markers are seen on transabdominal and transvaginal US examinations
- While awaiting national guidelines that 1) standardize the approach to the US examination and 2) define PAS US markers, we encourage US laboratories to develop local protocols to standardize the sonographic evaluation of the placenta and ensure uniform and complete placental assessment
Based on a recent meta-analysis, overall sensitivity of MRI for detecting PAS is 86% to 95%, with specificity of 80% to 95%. Although this is comparable to the sensitivity and specificity of US,27 studies of MRI in PAS are smaller and more prone to bias than in studies of US, because MRI typically is used only in patients at highest risk for PAS. Few studies comparing US to MRI for PAS have been performed; all are small and lack statistical power.
Complementary role. MRI can be complementary to US in cases in which the placenta is posterior or located laterally28 but, importantly, rarely changes decisions about surgical management when used in conjunction with US to assess patients for the diagnosis of PAS. (An exception might lie in the ability of MRI to assess the degree or depth of invasion of the placenta and discerning placenta percreta from placenta accreta.15)
Enhancement with contrast. Addition of gadolinium-based contrast might improve the ability of MRI to make a diagnosis of PAS, but gadolinium crosses the placenta barrier. Although fetal effects of gadolinium have not been observed, American College of Radiology guidelines recommend avoiding this contrast agent during pregnancy unless absolutely essential.29
Specific indications. MRI without contrast should be considered 1) when US is inconclusive and 2) to further evaluate a posterior placenta suspicious for invasion, to define the precise topography of extrauterine placental invasion. The additional information offered by MRI might alter surgical planning.15
Overall, based on current literature, gray-scale US appears to be an excellent tool for prenatal diagnosis of PAS in women at risk: Sensitivity has been reported in the range of 80% to 90%; specificity, 91% to 98%; positive predictive value, 65% to 93%; and negative predictive value, 98%.5,6
However, these values might overestimate the true ability of prenatal US to predict PAS. Why? Early studies that assessed the accuracy of US prediction of PAS might have been biased by inclusion of single-expert observations, high suspicion of placenta accreta, and prior knowledge of patients’ risk factors. In addition, small sample size, retrospective design, and wide variability in the definition of PAS and inclusion criteria led to inconsistency in performance and skewed sensitivity.7
In fact, when experienced providers, reviewing the same US images, were blinded to patients’ clinical history, the accuracy of US diagnosis of PAS decreased in regard to sensitivity (to 54%), specificity (88%), positive (82%) and negative (65%) predictive value, and accuracy (65%).4 Investigators also found wide inter-observer variability in the interpretation of markers of PAS.4 Furthermore, there is evidence that several PAS US markers are commonly seen in low-risk normal pregnancy.
Although studies have yielded variable findings of the precise sensitivity and positive predictive value of US in the diagnosis of PAS, there is a general agreement that US should be the primary imaging modality for this purpose, and can be used exclusively in most cases.
References
- Comstock CH, Bronsteen RA. The antenatal diagnosis of placenta accreta. BJOG. 2014;121:171-181.
- D’Antonio F, Iacovella C, Bhide A. Prenatal identification of invasive placentation using ultrasound: systematic review and metaanalysis. Ultrasound Obstet Gynecol. 2013;42:509-517.
- Comstock CH, Love JJ Jr, Bronsteen RA, et al. Sonographic detection of placenta accreta in the second and third trimesters of pregnancy. Am J Obstet Gynecol. 2004;190:1135-1140.
- Bowman ZS, Eller AG, Kennedy AM, et al. Interobserver variability of sonography for prediction of placenta accreta. J Ultrasound Med. 2014;33:2153-2158.
Biomarkers
Multiple serum biomarkers have been proposed to predict PAS in high-risk women. PAS might be associated with increased levels of first-trimester pregnancy-associated plasma protein A, second-trimester maternal serum alpha fetoprotein, and human chorionic gonadotropin, but studies of the utility of these biomarkers have yielded contradictory results.30,31 Biomarkers are of interest and have significant clinical applicability, but none of the ones identified to date have high sensitivity or specificity for predicting PAS prenatally. Research is ongoing to identify markers of PAS that have sufficient predictive power.
Share your thoughts! Send your Letter to the Editor to [email protected]. Please include your name and the city and state in which you practice.
- Irving FC, Hertig AT. A study of placenta accreta. Surg Gynec Obstet. 1937:64:178–200.
- Wu S, Kocherginsky M, Hibbard JU. Abnormal placentation: twenty-year analysis. Am J Obstet Gynecol. 2005;192:1458–1461.
- Hall T, Wax JR, Lucas FL, et al. Prenatal sonographic diagnosis of placenta accreta—impact on maternal and neonatal outcomes. J Clin Ultrasound. 2014;42:449–455.
- Bowman ZS, Eller AG, Kennedy AM, et al. Interobserver variability of sonography for prediction of placenta accreta. J Ultrasound Med. 2014;33:2153–2158.
- Silver RM. Abnormal placentation: placenta previa, vasa previa, and placenta accreta. Obstet Gynecol. 2015;126:654–668.
- Silver RM, Landon MB, Rouse DJ, et al; National Institute of Child Health and Human Development Maternal-Fetal Medicine Units Network. Maternal morbidity associated with multiple repeat cesarean deliveries. Obstet Gynecol. 2006;107:1226–1232.
- Rac MW, Moschos E, Wells CE, et al. Sonographic findings of morbidly adherent placenta in the first trimester. J Ultrasound Med. 2016;35:263–269.
- Ballas J, Pretorius D, Hull AD, et al. Identifying sonographic markers for placenta accreta in the first trimester. J Ultrasound Med. 2012;31:1835–1841.
- Comstock CH, Bronsteen RA. The antenatal diagnosis of placenta accreta. BJOG. 2014;121:171–181.
- Marshall NE, Fu R, Guise JM. Impact of multiple cesarean deliveries on maternal morbidity: a systematic review. Am J Obstet Gynecol. 2011;205:262.e1–e8.
- Baughman WC, Corteville JE, Shah RR. Placenta accreta: spectrum of US and MR imaging findings. Radiographics. 2008;28:1905–1916.
- Finberg HJ, Williams JW. Placenta accreta: prospective sonographic diagnosis in patients with placenta previa and prior cesarean section. J Ultrasound Med. 1992;11:333–343.
- D’Antonio F, Iacovella C, Bhide A. Prenatal identification of invasive placentation using ultrasound: systematic review and meta-analysis. Ultrasound Obstet Gynecol. 2013;42:509–517.
- Comstock CH, Love JJ Jr, Bronsteen RA, et al. Sonographic detection of placenta accreta in the second and third trimesters of pregnancy. Am J Obstet Gynecol. 2004;190:1135–1140.
- D’Antonio F, Palacios-Jaraquemada J, Lim PS, et al. Counseling in fetal medicine: evidence-based answers to clinical questions on morbidly adherent placenta. Ultrasound Obstet Gynecol. 2016;47:290–301.
- Hudon L, Belfort MA, Broome DR. Diagnosis and management of placenta percreta: a review. Obstet Gynecol Surv. 1998;53:509–517.
- Wong HS, Cheung YK, Zuccollo J, et al. Evaluation of sonographic diagnostic criteria for placenta accreta. J Clin Ultrasound. 2008;36:551–559.
- Jauniaux E, Collins S, Burton GJ. Placenta accreta spectrum: pathophysiology and evidence-based anatomy for prenatal ultrasound imaging. Am J Obstet Gynecol. 2018;218:75–87.
- Rac MW, Dashe JS, Wells CE, et al. Ultrasound predictors of placental invasion: the Placenta Accreta Index. Am J Obstet Gynecol. 2015;212:343.e1–e7.
- Pilloni E, Alemanno MG, Gaglioti P, et al. Accuracy of ultrasound in antenatal diagnosis of placental attachment disorders. Ultrasound Obstet Gynecol. 2016;47:302–307.
- Comstock CH. Antenatal diagnosis of placenta accreta: a review. Ultrasound Obstet Gynecol. 2005;26:89–96.
- Collins SL, Stevenson GN, Al-Khan A, et al. Three-dimensional power Doppler ultrasonography for diagnosing abnormally invasive placenta and quantifying the risk. Obstet Gynecol. 2015;126:645–653.
- Collins SL, Ashcroft A, Braun T, et al; European Working Group on Abnormally Invasive Placenta (EW-AIP). Proposal for standardized ultrasound descriptors of abnormally invasive placenta (AIP). Ultrasound Obstet Gynecol. 2016;47:271–275.
- Gilboa Y, Spira M, Mazaki-Tovi S, et al. A novel sonographic scoring system for antenatal risk assessment of obstetric complications in suspected morbidly adherent placenta. J Ultrasound Med. 2015;34:561–567.
- Tovbin J, Melcer Y, Shor S, et al. Prediction of morbidly adherent placenta using a scoring system. Ultrasound Obstet Gynecol. 2016;48:504–510.
- Jauniaux E, Collins SL, Jurkovic D, Burton GJ. Accreta placentation: a systematic review of prenatal ultrasound imaging and grading of villous invasiveness. Am J Obstet Gynecol. 2016:215:712–721.
- Familiari A, Liberati M, Lim P, et al. Diagnostic accuracy of magnetic resonance imaging in detecting the severity of abnormal invasive placenta: a systematic review and meta-analysis. Acta Obstet Gynecol Scand. 2018;97:507–520.
- Rezk MA, Shawky M. Grey-scale and colour Doppler ultrasound versus magnetic resonance imaging for the prenatal diagnosis of placenta accreta. J Matern Fetal Neonatal Med. 2016;29:218–223.
- Expert Panel on MR Safety; Kanal E, Barkovich AJ, Bell C, et al. ACR guidance document on MR safe practices: 2013. J Magn Reson Imaging. 2013;37:501–530.
- Pekar-Zlotin M, Melcer Y, Maymon R, Jauniaux E. Secondtrimester levels of fetoplacental hormones among women with placenta accreta spectrum disorders. Int J Gynaecol Obstet. 2018;140:377–378.
- Lyell DJ, Faucett AM, Baer RJ, et al. Maternal serum markers, characteristics and morbidly adherent placenta in women with previa. J Perinatol. 2015;35:570–574.
Placenta accreta spectrum (PAS) describes abnormal invasion of placental tissue into or through the myometrium, comprising 3 distinct conditions: placenta accreta, placenta increta, and placenta percreta. This complication is relatively new to obstetrics, first described in 1937.1
The overall incidence of PAS has been increasing over several decades, in parallel to an increasing rate of cesarean delivery (CD), with an incidence from 1982 through 2002 of 1 in 533 pregnancies, representing a 5-fold increase since the 1980s.2 PAS is associated with significant morbidity and mortality, including fetal growth restriction, preterm delivery, placental abruption antenatally, and hemorrhage during delivery or postpartum.
Prenatal diagnosis of PAS and planned delivery at an experienced center are associated with significant reduction in maternal and fetal morbidity.3 In an era of advanced imaging modalities, prenatal detection of PAS regrettably remains variable and largely subjective: As many as 20% to 50% of cases of PAS escape prenatal diagnosis.3,4
In this article, we review the sonographic markers of PAS, including diagnostic accuracy, and propose a standardized approach to prenatal diagnosis. Throughout our discussion, we describe protocols for detection of PAS practiced at our Maternal-Fetal Medicine Program in the Department of Obstetrics and Gynecology, Eastern Virginia Medical School (also see “US evaluation of PAS risk: The authors’ recommended approach”).
Numerous risk factors
There are many risk factors for PAS, including prior uterine surgery or instrumentation, such as CD, uterine curettage, myomectomy, pelvic radiation, and endometrial ablation. Other risk factors include smoking, in vitro fertilization, advanced maternal age, multiparity, and a brief interval between prior CD and subsequent pregnancy.5 Of major significance is the increased risk of PAS in the presence of placenta previa with prior CD.6 Knowledge of clinical risk factors by the interpreting physician appears to be associated with improved detection of PAS on ultrasonography (US).4
Ultrasonographic markers of PAS
First-trimester markers
Sonographic markers of PAS in the first trimester include:
- a gestational sac implanted in the lower uterine segment or in a CD scar
- multiple hypoechoic spaces within the placenta (lacunae).7
Lower uterine-segment implantation has been defined by Ballas and colleagues as 1) a gestational sac implanted in the lower one-third of the uterus between 8 and 10 weeks’ gestation or 2) a gestational sac occupying primarily the lower uterine segment from 10 weeks’ gestation onward (FIGURE 1).8 Our experience is that it is difficult to accurately assess lower uterine-segment implantation beyond 13 weeks of gestation because the sac typically expands to fill the upper uterine cavity.
Continue to: Color Doppler US...
Color Doppler US can help differentiate lower uterine-segment implantation from a gestational sac of a failed pregnancy in the process of expulsion by demonstrating loss of circumferential blood flow in the failed pregnancy. Furthermore, applying pressure to the anterior surface of the uterus will result in downward movement of the gestational sac of a failed pregnancy.9
Not all gestational sacs that implant in the lower uterine segment lead to PAS: Subsequent normal pregnancies have been reported in this circumstance. In such cases, a normal thick myometrium is noted anterior to the gestational sac.7 A patient with lower uterine-segment implantation without evidence of anterior myometrial thinning remains at risk for third-trimester placenta previa.7
Cesarean scar pregnancy carries significant risk of PAS. In these cases, the gestational sac is typically implanted within the scar, resulting in a thin anterior myometrium and significantly increased vascularity of the placental–myometrial and bladder–uterine wall interfaces (FIGURE 2).9 Differentiating cesarean scar pregnancy from a lower uterine-segment implantation is easier to perform before the eighth week of gestation but becomes more difficult as pregnancy advances. Although it might be useful to distinguish between true cesarean scar pregnancy and lower uterine-segment implantation adjacent to or involving the scar, both carry considerable risk of PAS and excessive hemorrhage, and the approach to treating both conditions is quite similar.
Lacunae, with or without documented blood flow on color Doppler US, are the third marker of PAS in the first trimester.8 Although some retrospective series and case reports describe the finding of lacunae in the first trimester of patients with diagnosed PAS, more recent literature suggests that these spaces are seen infrequently and at a similar frequency in women with and without PAS at delivery.7
Second- and third-trimester markers
Multiple diagnostic sonographic markers of PAS have been described in the second and third trimesters.
Placental location is a significant risk factor for PAS. Placenta previa in the setting of prior CD carries the highest risk of PAS—as high as 61% in women with both placenta previa and a history of 3 CDs.10 An anterior placenta appears to be a stronger risk factor for PAS than a posterior placenta in women with prior CD; the location of the placenta should therefore be evaluated in all women in the second trimester.
Continue to: Lacunae
Lacunae. The finding of multiple hypoechoic vascular spaces within the placental parenchyma has been associated with PAS (FIGURES 3 and 4). The pathogenesis of this finding is probably related to alterations in placental tissue resulting from long-term exposure to pulsatile blood flow.11
Finberg and colleagues introduced a grading system for placental lacunae in 1992 that is still used:
- Grade 0: no lacunae seen
- Grade 1: 1 to 3 lacunae seen
- Grade 2: 4 to 6 lacunae seen
- Grade 3: multiple lacunae seen throughout the placenta.12
The sensitivity and specificity of lacunae as an independent marker for PAS have been reported to be 77% and 95%, respectively.13 Despite these findings, several studies report a range of sensitivity (73% to 100%) and negative predictive value (88% to 100%).14 Even in Finberg’s original work, 27% of cases of confirmed PAS had Grade 0 or Grade 1 placental lacunae and 11% of cases of placenta previa, without PAS, demonstrated Grade 2 lacunae.12 There is agreement, however, that, the more lacunae, the higher the risk of PAS.
Continue to: Other US markers for PAS
Other US markers of PAS

Retroplacental–myometrial interface

Loss of the normal hypoechoic (clear) retroplacental zone, also referred to as loss of the clear space between placenta and uterus, is another marker of PAS (FIGURE 5). This finding corresponds to pathologic loss of the decidua basalis as trophoblastic tissue invades directly through the myometrium.15 This sonographic finding has been reported to have a detection rate of approximately 93%, with sensitivity of 52% and specificity of 57%, for PAS; the false-positive rate, however, has been in the range of 21% or higher. This marker should not be used alone because it is angle-dependent and can be found (as an absent clear zone) in normal anterior placentas.16
The strength of this US marker is in its negative predictive value, which ranges from 96% to 100%. The presence of a hypoechoic retroplacental clear space that extends the length of the placenta makes PAS unlikely.17 Of note, the clear zone may appear falsely absent as a result of increased pressure from the US probe.
Retroplacental myometrial thickness
Retroplacental myometrial thickness is difficult to assess because the lower uterine-segment myometrium thins in normal pregnancy as term approaches. This measurement also can be influenced by direct pressure of the US probe and fullness of the maternal bladder.18 In patients who have had a CD but who do not have PAS, the median myometrial thickness of the lower uterine segment in the third trimester is 2.4 mm.19
Thinning of the myometrium in the upper uterine segment always should be of concern. Studies of this marker have reported sensitivity of US ranging from 22% to 100% and specificity from 72% to 100%.9,20 Given such variability, it is important to standardize the gestational age and sonographic approach for this marker.
Continue to: Uterovesical interface
Uterovesical interface
Studies also have reported that abnormalities of the uterovesical interface are predictive of PAS. The uterovesical interface is best evaluated in a sagittal plane containing the lower uterine segment and a partially full bladder in gray-scale and color Doppler US.15 The normal uterovesical interface appears as a smooth line, without irregularities or increased vascularity on sagittal imaging.
Abnormalities include focal interruption of the hyperechoic bladder wall, bulging of the bladder wall, and increased vascularity, such as varicosities (FIGURES 5, 6, and 7).15 These findings may be seen as early as the first trimester but are more commonly noted in the second and third trimesters.7 The authors of a recent meta-analysis concluded that irregularity of the uterovesical interface is the most specific marker for invasive placentation (99.75% confidence interval; range, 99.5% to 99.9%).13
Other US markers and modalities
Three-dimensional US. Studies have evaluated the role of 3-dimensional (3D) US for predicting PAS. Application of 3D US in vascular mode has shown promise because it allows for semiquantitative assessment of placental vasculature.22 Using 3D US to screen for PAS presents drawbacks, however: The technology is not well-standardized and requires significant operator expertise for volume acquisition and manipulation. Prospective studies are needed before 3D US can be applied routinely to screen for and diagnose PAS.
Color Doppler US. As an adjunct to gray-scale US, color Doppler US can be used for making a diagnosis of PAS. Color Doppler US helps differentiate a normal subplacental venous complex with nonpulsatile, low-velocity venous blood flow waveforms from markedly dilated peripheral subplacental vascular channels with pulsatile venous-type flow, which suggests PAS. These vascular channels are often located directly over the cervix. In addition, the observation of bridging vessels linking the placenta and bladder with high diastolic arterial blood flow also suggests invasion.21 In a meta-analysis, overall sensitivity of color Doppler US for the diagnosis of PAS was 91%, with specificity of 87%.13
The value of utilizing multiple markers
The accuracy of US diagnosis of PAS is likely improved by using more than 1 sonographic marker. Pilloni and colleagues,20 in a prospective analysis, found that 81% of cases of confirmed PAS had ≥2 markers and 51% of cases had ≥3 markers.
Several scoring systems have been proposed for making the diagnosis of PAS using combinations of sonographic markers, placental location, and clinical history.19,24,25 In 2016, Tovbin and colleagues,25 in a prospective study, evaluated a scoring system that included:
- number of previous CDs
- number of, maximum dimension of, and presence of blood flow in lacunae
- loss of uteroplacental clear zone
- placental location
- hypervascularity of the uterovesical or uteroplacental interface.
Tovbin assigned 1 or 2 points to each criterion. Each sonographic marker was found to be significantly associated with PAS when compared to a high-risk control group. A score of ≥8 was considered “at high risk” and predicted 69% of PAS cases.
Regrettably, no combination of US markers reliably predicts the depth of invasion of the placenta.26
Continue to: A standardized approach is needed
A standardized approach is needed
To decrease variability and improve the US diagnosis of PAS, it is important to define and standardize the diagnosis of each sonographic marker for PAS.4 In 2016, the European Working Group on Abnormally Invasive Placenta (EW-AIP) proposed a set of US markers that always should be reported when performing an US examination for suspected abnormal placentation (TABLE).23 Despite this effort by the EW-AIP, ambiguity remains over sonographic definitions of several PAS markers. For example, what determines a placental lacuna on US? And what constitutes an abnormal uterovesical interface? There is a need for a more objective definition of US markers of PAS and a standardized approach to the US examination in at-risk pregnancies.
The Society for Maternal-Fetal Medicine is coordinating a multi-society task force to address the need to define and standardize the US diagnosis of PAS.
Observations on other PAS diagnostic modalities
Magnetic resonance imaging
Adjunctive role. Magnetic resonance imaging (MRI) is often used as an adjunctive diagnostic modality in cases of suspected PAS. Several markers for PAS have been described on MRI, including15:
- intraplacental T2-weighted dark bands
- abnormal intraplacental vascularity
- heterogeneous intraplacental signal intensity
- focal interruption of the myometrium by the placenta
- uterine bulging.
- Assess a priori risk for the patient before initiating the US exam
- In the presence of a placenta previa, or low-lying placenta, we strongly recommend a transvaginal, in addition to transabdominal, US to further assess for the presence of placenta accreta spectrum (PAS) markers
- Until prospective studies clearly define the diagnostic accuracy of PAS sonographic markers and their performance in high-risk and low-risk pregnancies, we recommend that US findings be reported as a risk profile—that is, high, moderate, and low risk of PAS
- Be especially cautious with patients who are at substantially increased risk for PAS, such as those with placenta previa and prior multiple CDs. In this setting, a low-risk report for PAS only should be provided when none of the PAS markers are seen on transabdominal and transvaginal US examinations
- While awaiting national guidelines that 1) standardize the approach to the US examination and 2) define PAS US markers, we encourage US laboratories to develop local protocols to standardize the sonographic evaluation of the placenta and ensure uniform and complete placental assessment
Based on a recent meta-analysis, overall sensitivity of MRI for detecting PAS is 86% to 95%, with specificity of 80% to 95%. Although this is comparable to the sensitivity and specificity of US,27 studies of MRI in PAS are smaller and more prone to bias than in studies of US, because MRI typically is used only in patients at highest risk for PAS. Few studies comparing US to MRI for PAS have been performed; all are small and lack statistical power.
Complementary role. MRI can be complementary to US in cases in which the placenta is posterior or located laterally28 but, importantly, rarely changes decisions about surgical management when used in conjunction with US to assess patients for the diagnosis of PAS. (An exception might lie in the ability of MRI to assess the degree or depth of invasion of the placenta and discerning placenta percreta from placenta accreta.15)
Enhancement with contrast. Addition of gadolinium-based contrast might improve the ability of MRI to make a diagnosis of PAS, but gadolinium crosses the placenta barrier. Although fetal effects of gadolinium have not been observed, American College of Radiology guidelines recommend avoiding this contrast agent during pregnancy unless absolutely essential.29
Specific indications. MRI without contrast should be considered 1) when US is inconclusive and 2) to further evaluate a posterior placenta suspicious for invasion, to define the precise topography of extrauterine placental invasion. The additional information offered by MRI might alter surgical planning.15
Overall, based on current literature, gray-scale US appears to be an excellent tool for prenatal diagnosis of PAS in women at risk: Sensitivity has been reported in the range of 80% to 90%; specificity, 91% to 98%; positive predictive value, 65% to 93%; and negative predictive value, 98%.5,6
However, these values might overestimate the true ability of prenatal US to predict PAS. Why? Early studies that assessed the accuracy of US prediction of PAS might have been biased by inclusion of single-expert observations, high suspicion of placenta accreta, and prior knowledge of patients’ risk factors. In addition, small sample size, retrospective design, and wide variability in the definition of PAS and inclusion criteria led to inconsistency in performance and skewed sensitivity.7
In fact, when experienced providers, reviewing the same US images, were blinded to patients’ clinical history, the accuracy of US diagnosis of PAS decreased in regard to sensitivity (to 54%), specificity (88%), positive (82%) and negative (65%) predictive value, and accuracy (65%).4 Investigators also found wide inter-observer variability in the interpretation of markers of PAS.4 Furthermore, there is evidence that several PAS US markers are commonly seen in low-risk normal pregnancy.
Although studies have yielded variable findings of the precise sensitivity and positive predictive value of US in the diagnosis of PAS, there is a general agreement that US should be the primary imaging modality for this purpose, and can be used exclusively in most cases.
References
- Comstock CH, Bronsteen RA. The antenatal diagnosis of placenta accreta. BJOG. 2014;121:171-181.
- D’Antonio F, Iacovella C, Bhide A. Prenatal identification of invasive placentation using ultrasound: systematic review and metaanalysis. Ultrasound Obstet Gynecol. 2013;42:509-517.
- Comstock CH, Love JJ Jr, Bronsteen RA, et al. Sonographic detection of placenta accreta in the second and third trimesters of pregnancy. Am J Obstet Gynecol. 2004;190:1135-1140.
- Bowman ZS, Eller AG, Kennedy AM, et al. Interobserver variability of sonography for prediction of placenta accreta. J Ultrasound Med. 2014;33:2153-2158.
Biomarkers
Multiple serum biomarkers have been proposed to predict PAS in high-risk women. PAS might be associated with increased levels of first-trimester pregnancy-associated plasma protein A, second-trimester maternal serum alpha fetoprotein, and human chorionic gonadotropin, but studies of the utility of these biomarkers have yielded contradictory results.30,31 Biomarkers are of interest and have significant clinical applicability, but none of the ones identified to date have high sensitivity or specificity for predicting PAS prenatally. Research is ongoing to identify markers of PAS that have sufficient predictive power.
Share your thoughts! Send your Letter to the Editor to [email protected]. Please include your name and the city and state in which you practice.
Placenta accreta spectrum (PAS) describes abnormal invasion of placental tissue into or through the myometrium, comprising 3 distinct conditions: placenta accreta, placenta increta, and placenta percreta. This complication is relatively new to obstetrics, first described in 1937.1
The overall incidence of PAS has been increasing over several decades, in parallel to an increasing rate of cesarean delivery (CD), with an incidence from 1982 through 2002 of 1 in 533 pregnancies, representing a 5-fold increase since the 1980s.2 PAS is associated with significant morbidity and mortality, including fetal growth restriction, preterm delivery, placental abruption antenatally, and hemorrhage during delivery or postpartum.
Prenatal diagnosis of PAS and planned delivery at an experienced center are associated with significant reduction in maternal and fetal morbidity.3 In an era of advanced imaging modalities, prenatal detection of PAS regrettably remains variable and largely subjective: As many as 20% to 50% of cases of PAS escape prenatal diagnosis.3,4
In this article, we review the sonographic markers of PAS, including diagnostic accuracy, and propose a standardized approach to prenatal diagnosis. Throughout our discussion, we describe protocols for detection of PAS practiced at our Maternal-Fetal Medicine Program in the Department of Obstetrics and Gynecology, Eastern Virginia Medical School (also see “US evaluation of PAS risk: The authors’ recommended approach”).
Numerous risk factors
There are many risk factors for PAS, including prior uterine surgery or instrumentation, such as CD, uterine curettage, myomectomy, pelvic radiation, and endometrial ablation. Other risk factors include smoking, in vitro fertilization, advanced maternal age, multiparity, and a brief interval between prior CD and subsequent pregnancy.5 Of major significance is the increased risk of PAS in the presence of placenta previa with prior CD.6 Knowledge of clinical risk factors by the interpreting physician appears to be associated with improved detection of PAS on ultrasonography (US).4
Ultrasonographic markers of PAS
First-trimester markers
Sonographic markers of PAS in the first trimester include:
- a gestational sac implanted in the lower uterine segment or in a CD scar
- multiple hypoechoic spaces within the placenta (lacunae).7
Lower uterine-segment implantation has been defined by Ballas and colleagues as 1) a gestational sac implanted in the lower one-third of the uterus between 8 and 10 weeks’ gestation or 2) a gestational sac occupying primarily the lower uterine segment from 10 weeks’ gestation onward (FIGURE 1).8 Our experience is that it is difficult to accurately assess lower uterine-segment implantation beyond 13 weeks of gestation because the sac typically expands to fill the upper uterine cavity.
Continue to: Color Doppler US...
Color Doppler US can help differentiate lower uterine-segment implantation from a gestational sac of a failed pregnancy in the process of expulsion by demonstrating loss of circumferential blood flow in the failed pregnancy. Furthermore, applying pressure to the anterior surface of the uterus will result in downward movement of the gestational sac of a failed pregnancy.9
Not all gestational sacs that implant in the lower uterine segment lead to PAS: Subsequent normal pregnancies have been reported in this circumstance. In such cases, a normal thick myometrium is noted anterior to the gestational sac.7 A patient with lower uterine-segment implantation without evidence of anterior myometrial thinning remains at risk for third-trimester placenta previa.7
Cesarean scar pregnancy carries significant risk of PAS. In these cases, the gestational sac is typically implanted within the scar, resulting in a thin anterior myometrium and significantly increased vascularity of the placental–myometrial and bladder–uterine wall interfaces (FIGURE 2).9 Differentiating cesarean scar pregnancy from a lower uterine-segment implantation is easier to perform before the eighth week of gestation but becomes more difficult as pregnancy advances. Although it might be useful to distinguish between true cesarean scar pregnancy and lower uterine-segment implantation adjacent to or involving the scar, both carry considerable risk of PAS and excessive hemorrhage, and the approach to treating both conditions is quite similar.
Lacunae, with or without documented blood flow on color Doppler US, are the third marker of PAS in the first trimester.8 Although some retrospective series and case reports describe the finding of lacunae in the first trimester of patients with diagnosed PAS, more recent literature suggests that these spaces are seen infrequently and at a similar frequency in women with and without PAS at delivery.7
Second- and third-trimester markers
Multiple diagnostic sonographic markers of PAS have been described in the second and third trimesters.
Placental location is a significant risk factor for PAS. Placenta previa in the setting of prior CD carries the highest risk of PAS—as high as 61% in women with both placenta previa and a history of 3 CDs.10 An anterior placenta appears to be a stronger risk factor for PAS than a posterior placenta in women with prior CD; the location of the placenta should therefore be evaluated in all women in the second trimester.
Continue to: Lacunae
Lacunae. The finding of multiple hypoechoic vascular spaces within the placental parenchyma has been associated with PAS (FIGURES 3 and 4). The pathogenesis of this finding is probably related to alterations in placental tissue resulting from long-term exposure to pulsatile blood flow.11
Finberg and colleagues introduced a grading system for placental lacunae in 1992 that is still used:
- Grade 0: no lacunae seen
- Grade 1: 1 to 3 lacunae seen
- Grade 2: 4 to 6 lacunae seen
- Grade 3: multiple lacunae seen throughout the placenta.12
The sensitivity and specificity of lacunae as an independent marker for PAS have been reported to be 77% and 95%, respectively.13 Despite these findings, several studies report a range of sensitivity (73% to 100%) and negative predictive value (88% to 100%).14 Even in Finberg’s original work, 27% of cases of confirmed PAS had Grade 0 or Grade 1 placental lacunae and 11% of cases of placenta previa, without PAS, demonstrated Grade 2 lacunae.12 There is agreement, however, that, the more lacunae, the higher the risk of PAS.
Continue to: Other US markers for PAS
Other US markers of PAS

Retroplacental–myometrial interface

Loss of the normal hypoechoic (clear) retroplacental zone, also referred to as loss of the clear space between placenta and uterus, is another marker of PAS (FIGURE 5). This finding corresponds to pathologic loss of the decidua basalis as trophoblastic tissue invades directly through the myometrium.15 This sonographic finding has been reported to have a detection rate of approximately 93%, with sensitivity of 52% and specificity of 57%, for PAS; the false-positive rate, however, has been in the range of 21% or higher. This marker should not be used alone because it is angle-dependent and can be found (as an absent clear zone) in normal anterior placentas.16
The strength of this US marker is in its negative predictive value, which ranges from 96% to 100%. The presence of a hypoechoic retroplacental clear space that extends the length of the placenta makes PAS unlikely.17 Of note, the clear zone may appear falsely absent as a result of increased pressure from the US probe.
Retroplacental myometrial thickness
Retroplacental myometrial thickness is difficult to assess because the lower uterine-segment myometrium thins in normal pregnancy as term approaches. This measurement also can be influenced by direct pressure of the US probe and fullness of the maternal bladder.18 In patients who have had a CD but who do not have PAS, the median myometrial thickness of the lower uterine segment in the third trimester is 2.4 mm.19
Thinning of the myometrium in the upper uterine segment always should be of concern. Studies of this marker have reported sensitivity of US ranging from 22% to 100% and specificity from 72% to 100%.9,20 Given such variability, it is important to standardize the gestational age and sonographic approach for this marker.
Continue to: Uterovesical interface
Uterovesical interface
Studies also have reported that abnormalities of the uterovesical interface are predictive of PAS. The uterovesical interface is best evaluated in a sagittal plane containing the lower uterine segment and a partially full bladder in gray-scale and color Doppler US.15 The normal uterovesical interface appears as a smooth line, without irregularities or increased vascularity on sagittal imaging.
Abnormalities include focal interruption of the hyperechoic bladder wall, bulging of the bladder wall, and increased vascularity, such as varicosities (FIGURES 5, 6, and 7).15 These findings may be seen as early as the first trimester but are more commonly noted in the second and third trimesters.7 The authors of a recent meta-analysis concluded that irregularity of the uterovesical interface is the most specific marker for invasive placentation (99.75% confidence interval; range, 99.5% to 99.9%).13
Other US markers and modalities
Three-dimensional US. Studies have evaluated the role of 3-dimensional (3D) US for predicting PAS. Application of 3D US in vascular mode has shown promise because it allows for semiquantitative assessment of placental vasculature.22 Using 3D US to screen for PAS presents drawbacks, however: The technology is not well-standardized and requires significant operator expertise for volume acquisition and manipulation. Prospective studies are needed before 3D US can be applied routinely to screen for and diagnose PAS.
Color Doppler US. As an adjunct to gray-scale US, color Doppler US can be used for making a diagnosis of PAS. Color Doppler US helps differentiate a normal subplacental venous complex with nonpulsatile, low-velocity venous blood flow waveforms from markedly dilated peripheral subplacental vascular channels with pulsatile venous-type flow, which suggests PAS. These vascular channels are often located directly over the cervix. In addition, the observation of bridging vessels linking the placenta and bladder with high diastolic arterial blood flow also suggests invasion.21 In a meta-analysis, overall sensitivity of color Doppler US for the diagnosis of PAS was 91%, with specificity of 87%.13
The value of utilizing multiple markers
The accuracy of US diagnosis of PAS is likely improved by using more than 1 sonographic marker. Pilloni and colleagues,20 in a prospective analysis, found that 81% of cases of confirmed PAS had ≥2 markers and 51% of cases had ≥3 markers.
Several scoring systems have been proposed for making the diagnosis of PAS using combinations of sonographic markers, placental location, and clinical history.19,24,25 In 2016, Tovbin and colleagues,25 in a prospective study, evaluated a scoring system that included:
- number of previous CDs
- number of, maximum dimension of, and presence of blood flow in lacunae
- loss of uteroplacental clear zone
- placental location
- hypervascularity of the uterovesical or uteroplacental interface.
Tovbin assigned 1 or 2 points to each criterion. Each sonographic marker was found to be significantly associated with PAS when compared to a high-risk control group. A score of ≥8 was considered “at high risk” and predicted 69% of PAS cases.
Regrettably, no combination of US markers reliably predicts the depth of invasion of the placenta.26
Continue to: A standardized approach is needed
A standardized approach is needed
To decrease variability and improve the US diagnosis of PAS, it is important to define and standardize the diagnosis of each sonographic marker for PAS.4 In 2016, the European Working Group on Abnormally Invasive Placenta (EW-AIP) proposed a set of US markers that always should be reported when performing an US examination for suspected abnormal placentation (TABLE).23 Despite this effort by the EW-AIP, ambiguity remains over sonographic definitions of several PAS markers. For example, what determines a placental lacuna on US? And what constitutes an abnormal uterovesical interface? There is a need for a more objective definition of US markers of PAS and a standardized approach to the US examination in at-risk pregnancies.
The Society for Maternal-Fetal Medicine is coordinating a multi-society task force to address the need to define and standardize the US diagnosis of PAS.
Observations on other PAS diagnostic modalities
Magnetic resonance imaging
Adjunctive role. Magnetic resonance imaging (MRI) is often used as an adjunctive diagnostic modality in cases of suspected PAS. Several markers for PAS have been described on MRI, including15:
- intraplacental T2-weighted dark bands
- abnormal intraplacental vascularity
- heterogeneous intraplacental signal intensity
- focal interruption of the myometrium by the placenta
- uterine bulging.
- Assess a priori risk for the patient before initiating the US exam
- In the presence of a placenta previa, or low-lying placenta, we strongly recommend a transvaginal, in addition to transabdominal, US to further assess for the presence of placenta accreta spectrum (PAS) markers
- Until prospective studies clearly define the diagnostic accuracy of PAS sonographic markers and their performance in high-risk and low-risk pregnancies, we recommend that US findings be reported as a risk profile—that is, high, moderate, and low risk of PAS
- Be especially cautious with patients who are at substantially increased risk for PAS, such as those with placenta previa and prior multiple CDs. In this setting, a low-risk report for PAS only should be provided when none of the PAS markers are seen on transabdominal and transvaginal US examinations
- While awaiting national guidelines that 1) standardize the approach to the US examination and 2) define PAS US markers, we encourage US laboratories to develop local protocols to standardize the sonographic evaluation of the placenta and ensure uniform and complete placental assessment
Based on a recent meta-analysis, overall sensitivity of MRI for detecting PAS is 86% to 95%, with specificity of 80% to 95%. Although this is comparable to the sensitivity and specificity of US,27 studies of MRI in PAS are smaller and more prone to bias than in studies of US, because MRI typically is used only in patients at highest risk for PAS. Few studies comparing US to MRI for PAS have been performed; all are small and lack statistical power.
Complementary role. MRI can be complementary to US in cases in which the placenta is posterior or located laterally28 but, importantly, rarely changes decisions about surgical management when used in conjunction with US to assess patients for the diagnosis of PAS. (An exception might lie in the ability of MRI to assess the degree or depth of invasion of the placenta and discerning placenta percreta from placenta accreta.15)
Enhancement with contrast. Addition of gadolinium-based contrast might improve the ability of MRI to make a diagnosis of PAS, but gadolinium crosses the placenta barrier. Although fetal effects of gadolinium have not been observed, American College of Radiology guidelines recommend avoiding this contrast agent during pregnancy unless absolutely essential.29
Specific indications. MRI without contrast should be considered 1) when US is inconclusive and 2) to further evaluate a posterior placenta suspicious for invasion, to define the precise topography of extrauterine placental invasion. The additional information offered by MRI might alter surgical planning.15
Overall, based on current literature, gray-scale US appears to be an excellent tool for prenatal diagnosis of PAS in women at risk: Sensitivity has been reported in the range of 80% to 90%; specificity, 91% to 98%; positive predictive value, 65% to 93%; and negative predictive value, 98%.5,6
However, these values might overestimate the true ability of prenatal US to predict PAS. Why? Early studies that assessed the accuracy of US prediction of PAS might have been biased by inclusion of single-expert observations, high suspicion of placenta accreta, and prior knowledge of patients’ risk factors. In addition, small sample size, retrospective design, and wide variability in the definition of PAS and inclusion criteria led to inconsistency in performance and skewed sensitivity.7
In fact, when experienced providers, reviewing the same US images, were blinded to patients’ clinical history, the accuracy of US diagnosis of PAS decreased in regard to sensitivity (to 54%), specificity (88%), positive (82%) and negative (65%) predictive value, and accuracy (65%).4 Investigators also found wide inter-observer variability in the interpretation of markers of PAS.4 Furthermore, there is evidence that several PAS US markers are commonly seen in low-risk normal pregnancy.
Although studies have yielded variable findings of the precise sensitivity and positive predictive value of US in the diagnosis of PAS, there is a general agreement that US should be the primary imaging modality for this purpose, and can be used exclusively in most cases.
References
- Comstock CH, Bronsteen RA. The antenatal diagnosis of placenta accreta. BJOG. 2014;121:171-181.
- D’Antonio F, Iacovella C, Bhide A. Prenatal identification of invasive placentation using ultrasound: systematic review and metaanalysis. Ultrasound Obstet Gynecol. 2013;42:509-517.
- Comstock CH, Love JJ Jr, Bronsteen RA, et al. Sonographic detection of placenta accreta in the second and third trimesters of pregnancy. Am J Obstet Gynecol. 2004;190:1135-1140.
- Bowman ZS, Eller AG, Kennedy AM, et al. Interobserver variability of sonography for prediction of placenta accreta. J Ultrasound Med. 2014;33:2153-2158.
Biomarkers
Multiple serum biomarkers have been proposed to predict PAS in high-risk women. PAS might be associated with increased levels of first-trimester pregnancy-associated plasma protein A, second-trimester maternal serum alpha fetoprotein, and human chorionic gonadotropin, but studies of the utility of these biomarkers have yielded contradictory results.30,31 Biomarkers are of interest and have significant clinical applicability, but none of the ones identified to date have high sensitivity or specificity for predicting PAS prenatally. Research is ongoing to identify markers of PAS that have sufficient predictive power.
Share your thoughts! Send your Letter to the Editor to [email protected]. Please include your name and the city and state in which you practice.
- Irving FC, Hertig AT. A study of placenta accreta. Surg Gynec Obstet. 1937:64:178–200.
- Wu S, Kocherginsky M, Hibbard JU. Abnormal placentation: twenty-year analysis. Am J Obstet Gynecol. 2005;192:1458–1461.
- Hall T, Wax JR, Lucas FL, et al. Prenatal sonographic diagnosis of placenta accreta—impact on maternal and neonatal outcomes. J Clin Ultrasound. 2014;42:449–455.
- Bowman ZS, Eller AG, Kennedy AM, et al. Interobserver variability of sonography for prediction of placenta accreta. J Ultrasound Med. 2014;33:2153–2158.
- Silver RM. Abnormal placentation: placenta previa, vasa previa, and placenta accreta. Obstet Gynecol. 2015;126:654–668.
- Silver RM, Landon MB, Rouse DJ, et al; National Institute of Child Health and Human Development Maternal-Fetal Medicine Units Network. Maternal morbidity associated with multiple repeat cesarean deliveries. Obstet Gynecol. 2006;107:1226–1232.
- Rac MW, Moschos E, Wells CE, et al. Sonographic findings of morbidly adherent placenta in the first trimester. J Ultrasound Med. 2016;35:263–269.
- Ballas J, Pretorius D, Hull AD, et al. Identifying sonographic markers for placenta accreta in the first trimester. J Ultrasound Med. 2012;31:1835–1841.
- Comstock CH, Bronsteen RA. The antenatal diagnosis of placenta accreta. BJOG. 2014;121:171–181.
- Marshall NE, Fu R, Guise JM. Impact of multiple cesarean deliveries on maternal morbidity: a systematic review. Am J Obstet Gynecol. 2011;205:262.e1–e8.
- Baughman WC, Corteville JE, Shah RR. Placenta accreta: spectrum of US and MR imaging findings. Radiographics. 2008;28:1905–1916.
- Finberg HJ, Williams JW. Placenta accreta: prospective sonographic diagnosis in patients with placenta previa and prior cesarean section. J Ultrasound Med. 1992;11:333–343.
- D’Antonio F, Iacovella C, Bhide A. Prenatal identification of invasive placentation using ultrasound: systematic review and meta-analysis. Ultrasound Obstet Gynecol. 2013;42:509–517.
- Comstock CH, Love JJ Jr, Bronsteen RA, et al. Sonographic detection of placenta accreta in the second and third trimesters of pregnancy. Am J Obstet Gynecol. 2004;190:1135–1140.
- D’Antonio F, Palacios-Jaraquemada J, Lim PS, et al. Counseling in fetal medicine: evidence-based answers to clinical questions on morbidly adherent placenta. Ultrasound Obstet Gynecol. 2016;47:290–301.
- Hudon L, Belfort MA, Broome DR. Diagnosis and management of placenta percreta: a review. Obstet Gynecol Surv. 1998;53:509–517.
- Wong HS, Cheung YK, Zuccollo J, et al. Evaluation of sonographic diagnostic criteria for placenta accreta. J Clin Ultrasound. 2008;36:551–559.
- Jauniaux E, Collins S, Burton GJ. Placenta accreta spectrum: pathophysiology and evidence-based anatomy for prenatal ultrasound imaging. Am J Obstet Gynecol. 2018;218:75–87.
- Rac MW, Dashe JS, Wells CE, et al. Ultrasound predictors of placental invasion: the Placenta Accreta Index. Am J Obstet Gynecol. 2015;212:343.e1–e7.
- Pilloni E, Alemanno MG, Gaglioti P, et al. Accuracy of ultrasound in antenatal diagnosis of placental attachment disorders. Ultrasound Obstet Gynecol. 2016;47:302–307.
- Comstock CH. Antenatal diagnosis of placenta accreta: a review. Ultrasound Obstet Gynecol. 2005;26:89–96.
- Collins SL, Stevenson GN, Al-Khan A, et al. Three-dimensional power Doppler ultrasonography for diagnosing abnormally invasive placenta and quantifying the risk. Obstet Gynecol. 2015;126:645–653.
- Collins SL, Ashcroft A, Braun T, et al; European Working Group on Abnormally Invasive Placenta (EW-AIP). Proposal for standardized ultrasound descriptors of abnormally invasive placenta (AIP). Ultrasound Obstet Gynecol. 2016;47:271–275.
- Gilboa Y, Spira M, Mazaki-Tovi S, et al. A novel sonographic scoring system for antenatal risk assessment of obstetric complications in suspected morbidly adherent placenta. J Ultrasound Med. 2015;34:561–567.
- Tovbin J, Melcer Y, Shor S, et al. Prediction of morbidly adherent placenta using a scoring system. Ultrasound Obstet Gynecol. 2016;48:504–510.
- Jauniaux E, Collins SL, Jurkovic D, Burton GJ. Accreta placentation: a systematic review of prenatal ultrasound imaging and grading of villous invasiveness. Am J Obstet Gynecol. 2016:215:712–721.
- Familiari A, Liberati M, Lim P, et al. Diagnostic accuracy of magnetic resonance imaging in detecting the severity of abnormal invasive placenta: a systematic review and meta-analysis. Acta Obstet Gynecol Scand. 2018;97:507–520.
- Rezk MA, Shawky M. Grey-scale and colour Doppler ultrasound versus magnetic resonance imaging for the prenatal diagnosis of placenta accreta. J Matern Fetal Neonatal Med. 2016;29:218–223.
- Expert Panel on MR Safety; Kanal E, Barkovich AJ, Bell C, et al. ACR guidance document on MR safe practices: 2013. J Magn Reson Imaging. 2013;37:501–530.
- Pekar-Zlotin M, Melcer Y, Maymon R, Jauniaux E. Secondtrimester levels of fetoplacental hormones among women with placenta accreta spectrum disorders. Int J Gynaecol Obstet. 2018;140:377–378.
- Lyell DJ, Faucett AM, Baer RJ, et al. Maternal serum markers, characteristics and morbidly adherent placenta in women with previa. J Perinatol. 2015;35:570–574.
- Irving FC, Hertig AT. A study of placenta accreta. Surg Gynec Obstet. 1937:64:178–200.
- Wu S, Kocherginsky M, Hibbard JU. Abnormal placentation: twenty-year analysis. Am J Obstet Gynecol. 2005;192:1458–1461.
- Hall T, Wax JR, Lucas FL, et al. Prenatal sonographic diagnosis of placenta accreta—impact on maternal and neonatal outcomes. J Clin Ultrasound. 2014;42:449–455.
- Bowman ZS, Eller AG, Kennedy AM, et al. Interobserver variability of sonography for prediction of placenta accreta. J Ultrasound Med. 2014;33:2153–2158.
- Silver RM. Abnormal placentation: placenta previa, vasa previa, and placenta accreta. Obstet Gynecol. 2015;126:654–668.
- Silver RM, Landon MB, Rouse DJ, et al; National Institute of Child Health and Human Development Maternal-Fetal Medicine Units Network. Maternal morbidity associated with multiple repeat cesarean deliveries. Obstet Gynecol. 2006;107:1226–1232.
- Rac MW, Moschos E, Wells CE, et al. Sonographic findings of morbidly adherent placenta in the first trimester. J Ultrasound Med. 2016;35:263–269.
- Ballas J, Pretorius D, Hull AD, et al. Identifying sonographic markers for placenta accreta in the first trimester. J Ultrasound Med. 2012;31:1835–1841.
- Comstock CH, Bronsteen RA. The antenatal diagnosis of placenta accreta. BJOG. 2014;121:171–181.
- Marshall NE, Fu R, Guise JM. Impact of multiple cesarean deliveries on maternal morbidity: a systematic review. Am J Obstet Gynecol. 2011;205:262.e1–e8.
- Baughman WC, Corteville JE, Shah RR. Placenta accreta: spectrum of US and MR imaging findings. Radiographics. 2008;28:1905–1916.
- Finberg HJ, Williams JW. Placenta accreta: prospective sonographic diagnosis in patients with placenta previa and prior cesarean section. J Ultrasound Med. 1992;11:333–343.
- D’Antonio F, Iacovella C, Bhide A. Prenatal identification of invasive placentation using ultrasound: systematic review and meta-analysis. Ultrasound Obstet Gynecol. 2013;42:509–517.
- Comstock CH, Love JJ Jr, Bronsteen RA, et al. Sonographic detection of placenta accreta in the second and third trimesters of pregnancy. Am J Obstet Gynecol. 2004;190:1135–1140.
- D’Antonio F, Palacios-Jaraquemada J, Lim PS, et al. Counseling in fetal medicine: evidence-based answers to clinical questions on morbidly adherent placenta. Ultrasound Obstet Gynecol. 2016;47:290–301.
- Hudon L, Belfort MA, Broome DR. Diagnosis and management of placenta percreta: a review. Obstet Gynecol Surv. 1998;53:509–517.
- Wong HS, Cheung YK, Zuccollo J, et al. Evaluation of sonographic diagnostic criteria for placenta accreta. J Clin Ultrasound. 2008;36:551–559.
- Jauniaux E, Collins S, Burton GJ. Placenta accreta spectrum: pathophysiology and evidence-based anatomy for prenatal ultrasound imaging. Am J Obstet Gynecol. 2018;218:75–87.
- Rac MW, Dashe JS, Wells CE, et al. Ultrasound predictors of placental invasion: the Placenta Accreta Index. Am J Obstet Gynecol. 2015;212:343.e1–e7.
- Pilloni E, Alemanno MG, Gaglioti P, et al. Accuracy of ultrasound in antenatal diagnosis of placental attachment disorders. Ultrasound Obstet Gynecol. 2016;47:302–307.
- Comstock CH. Antenatal diagnosis of placenta accreta: a review. Ultrasound Obstet Gynecol. 2005;26:89–96.
- Collins SL, Stevenson GN, Al-Khan A, et al. Three-dimensional power Doppler ultrasonography for diagnosing abnormally invasive placenta and quantifying the risk. Obstet Gynecol. 2015;126:645–653.
- Collins SL, Ashcroft A, Braun T, et al; European Working Group on Abnormally Invasive Placenta (EW-AIP). Proposal for standardized ultrasound descriptors of abnormally invasive placenta (AIP). Ultrasound Obstet Gynecol. 2016;47:271–275.
- Gilboa Y, Spira M, Mazaki-Tovi S, et al. A novel sonographic scoring system for antenatal risk assessment of obstetric complications in suspected morbidly adherent placenta. J Ultrasound Med. 2015;34:561–567.
- Tovbin J, Melcer Y, Shor S, et al. Prediction of morbidly adherent placenta using a scoring system. Ultrasound Obstet Gynecol. 2016;48:504–510.
- Jauniaux E, Collins SL, Jurkovic D, Burton GJ. Accreta placentation: a systematic review of prenatal ultrasound imaging and grading of villous invasiveness. Am J Obstet Gynecol. 2016:215:712–721.
- Familiari A, Liberati M, Lim P, et al. Diagnostic accuracy of magnetic resonance imaging in detecting the severity of abnormal invasive placenta: a systematic review and meta-analysis. Acta Obstet Gynecol Scand. 2018;97:507–520.
- Rezk MA, Shawky M. Grey-scale and colour Doppler ultrasound versus magnetic resonance imaging for the prenatal diagnosis of placenta accreta. J Matern Fetal Neonatal Med. 2016;29:218–223.
- Expert Panel on MR Safety; Kanal E, Barkovich AJ, Bell C, et al. ACR guidance document on MR safe practices: 2013. J Magn Reson Imaging. 2013;37:501–530.
- Pekar-Zlotin M, Melcer Y, Maymon R, Jauniaux E. Secondtrimester levels of fetoplacental hormones among women with placenta accreta spectrum disorders. Int J Gynaecol Obstet. 2018;140:377–378.
- Lyell DJ, Faucett AM, Baer RJ, et al. Maternal serum markers, characteristics and morbidly adherent placenta in women with previa. J Perinatol. 2015;35:570–574.
2018 Update on pelvic floor dysfunction
Using cystoscopy to evaluate ureteral efflux and bladder integrity following benign gynecologic surgery increases the detection rate of urinary tract injuries.1 Currently, it is standard of care to perform a cystoscopy following anti-incontinence procedures, but there is no consensus among ObGyns regarding the use of universal cystoscopy following benign gynecologic surgery.2 A number of studies, however, have suggested potential best practices for evaluating urinary tract injury during pelvic surgery for benign gynecologic conditions.
Pelvic surgeries for benign gynecologic conditions, including fibroids, menorrhagia, and pelvic organ prolapse (POP), are common. More than 500,000 hysterectomies are performed annually in the United States, and up to 11% of women will undergo at least one surgery for POP or urinary incontinence in their lifetime.3,4 During gynecologic surgery, the urinary tract is at risk, and the injury rate ranges from 0.02% to 2% for ureteral injury and from 1% to 5% for bladder injury.5,6
In a recent large randomized controlled trial, the rate of intraoperative ureteral obstruction following uterosacral ligament suspension (USLS) was 3.2%.7 Vaginal vault suspensions, as well as other vaginal cuff closure techniques, are common procedures associated with urinary tract injury.8 Additionally, ureteral injury during surgery for POP occurs in as many as 2% of anterior vaginal wall repairs.9
It is well documented that a delay in diagnosis of ureteral and/or bladder injuries is associated with increased morbidity, including the need for additional surgery to repair the injury; in addition, significant delay in identifying an injury may lead to subsequent sequela, such as renal injury and fistula formation.8
A large study in California found that 36.5% of hysterectomies performed for POP were performed by general gynecologists.10 General ObGyns performing these surgeries therefore must understand the risk of urinary tract injury during hysterectomy and reconstructive pelvic procedures so that they can appropriately identify, evaluate, and repair injuries in a timely fashion.
The best way to identify urinary tract injury at the time of gynecologic surgery is by cystoscopy, including a bladder survey and ureteral efflux evaluation. When should a cystoscopy be performed, and what is the best method for visualizing ureteral efflux? Can instituting universal cystoscopy for all gynecologic procedures make a difference in the rate of injury detection? In this Update, we summarize the data from 4 studies that help to answer these questions.
Continue to: About 30% of urinary tract injuries...
About 30% of urinary tract injuries identified prior to cystoscopy at hysterectomy (which detected 5 of 6 injuries)
Vakili B, Chesson RR, Kyle BL, et al. The incidence of urinary tract injury during hysterectomy: a prospective analysis based on universal cystoscopy. Am J Obstet Gynecol. 2005;192(5):1599–1604.
Vakili and colleagues conducted a multicenter prospective cohort study of women undergoing hysterectomy for benign indications; cystoscopy was performed in all cases. The 3 hospitals involved were all part of the Louisiana State University Health system. The investigators’ goal was to determine the rate of urinary tract injury in this patient population at the time of intraoperative cystoscopy.
Intraoperative cystoscopy beats visual evaluation
Four hundred and seventy-one women underwent hysterectomy and had intraoperative cystoscopy, including evaluation of ureteral patency with administration of intravenous (IV) indigo carmine. Patients underwent abdominal, vaginal, or laparoscopic hysterectomy, and 54 (11.4%) had concurrent POP or anti-incontinence procedures. The majority underwent an abdominal hysterectomy (59%), 31% had a vaginal hysterectomy, and 10% had a laparoscopic-assisted vaginal hysterectomy or total laparoscopic hysterectomy.
Rate of urinary tract injuries. The total urinary tract injury rate detected by cystoscopy was 4.8%. The ureteral injury rate was 1.7%, and the bladder injury rate was 3.6%. A combined ureteral and bladder injury occurred in 2 women.
Surgery for POP significantly increased the risk of ureteral injury (7.3% vs 1.2%; P = .025). All cases of ureteral injury during POP surgery occurred during USLS. There was a trend toward a higher rate of bladder injury in the group with concurrent anti-incontinence surgery (12.5% vs 3.1%; P = .049). Regarding the route of hysterectomy, the vaginal approach had the highest rate of ureteral injury; however, when prolapse cases were removed from the analysis, there were no differences between the abdominal, vaginal, and laparoscopic approaches for ureteral or bladder injuries.
Injury detection with cystoscopy. Importantly, the authors found that only 30% of injuries were identified prior to performing intraoperative cystoscopy. The majority of these were bladder injuries. In addition, despite visual confirmation of ureteral peristalsis during abdominal hysterectomy, when intraoperative cystoscopy was performed with evaluation for ureteral efflux, 5 of 6 ureteral injury cases were identified. The authors reported 1 postoperative vesicovaginal fistula and concluded that it was likely due to an unrecognized bladder injury. No other undetected injuries were identified.
Notably, no complications occurred as a result of cystoscopy.
Multiple surgical indications reflect real-world scenario
The study included physicians from 3 different hospitals and all routes of hysterectomy for multiple benign gynecologic indications as well as concomitant pelvic reconstructive procedures. While this enhances the generalizability of the study results, all study sites were located in Louisiana at hospitals with resident trainee involvement. Additionally, this study confirms previous retrospective studies that reported higher rates of injury with pelvic reconstructive procedures.
The study is limited by the inability to blind surgeons, which may have resulted in the surgeons altering their techniques and/or having a heightened awareness of the urinary tract. However, their rates of ureteral and bladder injuries were slightly higher than previously reported rates, suggesting that the procedures inherently carry risk. The study is further limited by the lack of a retrospective comparison group of hysterectomy without routine cystoscopy and a longer follow-up period that may have revealed additional missed delayed urologic injuries.
The rate of urinary tract injury, including both bladder and ureteral injuries, was more than 4% at the time of hysterectomy for benign conditions. Using intraoperative peristalsis or normal ureteral caliber could result in a false sense of security since these are not reliable signs of ureteral integrity. The majority of urinary tract injuries will not be identified without cystoscopic evaluation.
Continue to: Universal cystoscopy policy...
Universal cystoscopy policy proves protective, surgeon adherence is high
Chi AM, Curran DS, Morgan DM, Fenner DE, Swenson CW. Universal cystoscopy after benign hysterectomy: examining the effects of an institutional policy. Obstet Gynecol. 2016;127(2):369–375.
In a retrospective cohort study, Chi and colleagues evaluated urinary tract injuries at the time of hysterectomy before and after the institution of a universal cystoscopy policy. At the time of policy implementation at the University of Michigan, all faculty who performed hysterectomies attended a cystoscopy workshop. Attending physicians without prior cystoscopy training also were proctored in the operating room for 3 sessions and were required to demonstrate competency with bladder survey, visualizing ureteral efflux, and urethral assessment. Indigo carmine was used to visualize ureteral efflux.
Detection of urologic injury almost doubled with cystoscopy
A total of 2,822 hysterectomies were included in the study, with 973 in the pre–universal cystoscopy group and 1,849 in the post–universal cystoscopy group. The study period was 7 years, and data on complications were abstracted for 1 year after the completion of the study period.
The primary outcome had 3 components:
- the rate of urologic injury before and after the policy
- the cystoscopy detection rate of urologic injury
- the adherence rate to the policy.
The overall rate of bladder and ureteral injury was 2.1%; the rate of injury during pre–universal screening was 2.6%, and during post–universal screening was 1.8%. The intraoperative detection rate of injury nearly doubled, from 24% to 47%, when intraoperative cystoscopy was utilized. In addition, the percentage of delayed urologic complications decreased from 28% to 5.9% (P = .03) following implementation of the universal cystoscopy policy. With regard to surgeon adherence, cystoscopy was documented in 86.1% of the hysterectomy cases after the policy was implemented compared with 35.7% of cases before the policy.
The investigators performed a cost analysis and found that hospital costs were nearly twice as much if a delayed urologic injury was diagnosed.
Study had many strengths
This study evaluated aspects of implementing quality initiatives after proper training and proctoring of a procedure. The authors compared very large cohorts from a busy academic medical center in which surgeon adherence with routine cystoscopy was high. The majority of patient outcomes were tracked for an extended period following surgery, thereby minimizing the risk of missing delayed urologic injuries. Notably, however, there was shorter follow-up time for the post–universal cystoscopy group, which could result in underestimating the rate of delayed urologic injuries in this cohort.
Instituting a universal cystoscopy policy for hysterectomy was associated with a significant decrease in delayed postoperative urinary tract complications and an increase in the intraoperative detection rate of urologic injuries. Intraoperative detection and repair of a urinary tract injury is cost-effective compared with a delayed diagnosis.
Continue to: Cystoscopy reveals ureteral obstruction...
Cystoscopy reveals ureteral obstruction during various vaginal POP repair procedures
Gustilo-Ashby AM, Jelovsek JE, Barber MD, Yoo EH, Paraiso MF, Walters MD. The incidence of ureteral obstruction and the value of intraoperative cystoscopy during vaginal surgery for pelvic organ prolapse. Am J Obstet Gynecol. 2006;194(5):1478–1485.
To determine the rate of ureteral obstruction and ureteral injury during vaginal surgery for POP and the accuracy of using intraoperative cystoscopy to prevent upper urinary tract morbidity, Gustilo-Ashby and colleagues performed a retrospective review study of a large patient cohort.
Cystoscopy with indigo carmine is highly sensitive
The study included 700 patients who underwent vaginal surgery for anterior and/or apical POP. Patients had 1 or more of the following anterior and apical prolapse repair procedures: USLS (51%), distal McCall culdeplasty (26%), proximal McCall culdeplasty (29%), anterior colporrhaphy (82%), and colpocleisis (1.4%). Of note, distal McCall culdeplasty was defined as incorporation of the “vaginal epithelium into the uterosacral plication,” while proximal McCall culdeplasty involved plication of “the uterosacral ligaments in the midline proximal to the vaginal cuff.” All patients were given IV indigo carmine to aid in visualizing ureteral efflux.
The majority of patients had a hysterectomy (56%). When accounting for rare false-positive and negative cystoscopy results, the overall ureteral obstruction rate was 5.1% and the ureteral injury rate was 0.9%. The majority of obstructions occurred with USLS (5.9%), proximal McCall culdeplasty (4.4%), and colpocleisis (4.2%). Ureteral injuries occurred only in 6 cases: 3 USLS and 3 proximal McCall culdeplasty procedures.
Based on these findings, the authors calculated that cystoscopy at the time of vaginal surgery for anterior and/or apical prolapse has a sensitivity of 94.4% and a specificity of 99.5% for detecting ureteral obstruction. The positive predictive value of cystoscopy with the use of indigo carmine for detection of ureteral obstruction is 91.9% and the negative predictive value is 99.7%.
Impact of indigo carmine’s unavailability
This study’s strengths include its large sample size and the variety of surgical approaches used for repair of anterior vaginal wall and apical prolapse. Its retrospective design, however, is a limitation; this could result in underreporting of ureteral injuries if patients received care at another institution after surgery. Furthermore, it is unclear if cystoscopy would be as predictive of ureteral injury without the use of indigo carmine, which is no longer available at most institutions.
The utility of cystoscopy with IV indigo carmine as a screening test for ureteral obstruction is highlighted by the fact that most obstructions were relieved by intraoperative suture removal following positive cystoscopy. McCall culdeplasty procedures are commonly performed by general ObGyns at the time of vaginal hysterectomy. It is therefore important to note that rates of ureteral obstruction after proximal McCall culdeplasty were only slightly lower than those after USLS.
Continue to: Sodium fluorescein and 10% dextrose...
Sodium fluorescein and 10% dextrose provide clear visibility of ureteral jets in cystoscopy
Espaillat-Rijo L, Siff L, Alas AN, et al. Intraoperative cystoscopic evaluation of ureteral patency: a randomized controlled trial. Obstet Gynecol. 2016;128(6):1378–1383.
In a multicenter randomized controlled trial, Espaillat-Rijo and colleagues compared various methods for visualizing ureteral efflux in participants who underwent gynecologic or urogynecologic procedures in which cystoscopy was performed.
Study compared 4 media
The investigators enrolled 176 participants (174 completed the trial) and randomly assigned them to receive 1 of 4 modalities: 1) normal saline as a bladder distention medium (control), 2) 10% dextrose as a bladder distention medium, 3) 200 mg oral phenazopyridine given 30 minutes prior to cystoscopy, or 4) 50 mg IV sodium fluorescein at the start of cystoscopy. Indigo carmine was not included in this study because it has not been routinely available since 2014.
Surgeons were asked to categorize the ureteral jets as “clearly visible,” “somewhat visible,” or “not visible.”
The primary outcome was subjective visibility of the ureteral jet with each modality during cystoscopy. Secondary outcomes included surgeon satisfaction, adverse reactions to the modality used, postoperative urinary tract infection, postoperative urinary retention, and delayed diagnosis of ureteral injury.
Visibility assessment results. Overall, ureteral jets were “clearly visible” in 125 cases (71%) compared with “somewhat visible” in 45 (25.6%) and “not visible” in 4 (2.3%) cases. There was a statistically significant difference between the 4 groups. Use of sodium fluorescein and 10% dextrose resulted in significantly better visualization of ureteral jets (P < .001 and P = .004, respectively) compared with the control group. Visibility with phenazopyridine was not significantly different from that in the control group or in the 10% dextrose group (FIGURE).
Surgeon satisfaction was highest with 10% dextrose and sodium fluorescein. In 6 cases, the surgeon was not satisfied with visualization of the ureteral jets and relied on fluorescein (5 times) or 10% dextrose (1 time) to ultimately see efflux. No significant adverse events occurred; the rate of urinary tract infection was 24.1% and did not differ between groups.
Results are widely generalizable
This was a well-designed randomized multicenter trial that included both benign gynecologic and urogynecologic procedures, thus strengthening the generalizability of the study. The study was timely since proven methods for visualization of ureteral patency became limited with the withdrawal of commercially available indigo carmine, the previous gold standard.
Intravenous sodium fluorescein and 10% dextrose as bladder distention media can both safely be used to visualize ureteral efflux and result in high surgeon satisfaction. Although 10% dextrose has been associated with higher rates of postoperative urinary tract infection,11 this was not found to be the case in this study. Preoperative administration of oral phenazopyridine was no different from the control modality with regard to visibility and surgeon satisfaction.
Continue to: The cost-effectiveness consideration
The cost-effectiveness consideration
The debate around universal cystoscopy following benign gynecologic surgery is ongoing.
The studies discussed in this Update demonstrate that cystoscopy following hysterectomy for benign indications:
- is superior to visualizing ureteral peristalsis
- increases detection of urinary tract injuries, and
- decreases delayed urologic injuries.
Although these articles emphasize the importance of detecting urologic injury, the picture would not be complete without mention of cost-effectiveness. Only one study, from 2001, has evaluated the cost-effectiveness of universal cystoscopy.1 Those authors concluded that universal cystoscopy is cost-effective only when the rate of urologic injury is 1.5% to 2%, but this conclusion, admittedly, was limited by the lack of data on medicolegal settlements, outpatient expenses, and nonmedical-related economic loss from decreased productivity. Given the extensive changes that have occurred in medical practice over the last 17 years and the emphasis on quality metrics and safety, an updated analysis would be needed to make definitive conclusions about cost-effectiveness.
While this Update cannot settle the ongoing debate of universal cystoscopy in gynecology, it is important to remember that the American College of Obstetricians and Gynecologists and the American Urogynecologic Society recommend cystoscopy following all surgeries for pelvic organ prolapse and stress urinary incontinence.2
References
- Visco AG, Taber KH, Weidner AD, Barber MD, Myers ER. Cost-effectiveness of universal cystoscopy to identify ureteral injury at hysterectomy. Obstet Gynecol. 2001;97(5 pt 1):685–692.
- ACOG Committee on Practice Bulletins–Gynecology and the American Urogynecologic Society. Urinary incontinence in women. Female Pelvic Med Reconstr Surg. 2015;21(6):304–314.
Share your thoughts! Send your Letter to the Editor to [email protected]. Please include your name and the city and state in which you practice.
- Ibeanu OA, Chesson RR, Echols KT, Nieves M, Busangu F, Nolan TE. Urinary tract injury during hysterectomy based on universal cystoscopy. Obstet Gynecol. 2009;113(1):6–10.
- ACOG Committee on Practice Bulletins–Gynecology and the American Urogynecologic Society. Urinary incontinence in women. Female Pelvic Med Reconstr Surg. 2015;21(6):304–314.
- Wilcox LS, Koonin LM, Pokras R, Strauss LT, Xia Z, Peterson HB. Hysterectomy in the United States, 1988–1990. Obstet Gynecol. 1994;83(4):549–555.
- Olsen AL, Smith VJ, Bergstrom JO, Colling JC, Clark AL. Epidemiology of surgically managed pelvic organ prolapse and urinary incontinence. Obstet Gynecol. 1997;89(4):501–506.
- Mäkinen J, Johansson J, Tomás C, et al. Morbidity of 10,110 hysterectomies by type of approach. Hum Reprod. 2001;16(7):1473–1478.
- Gilmour DT, Dwyer PL, Carey MP. Lower urinary tract injury during gynecologic surgery and its detection by intraoperative cystoscopy. Obstet Gynecol. 1999;94(5 pt 2):883–889.
- Barber MD, Brubaker L, Burgio KL, et al; Eunice Kennedy Schriver National Institute of Child Health and Human Development Pelvic Floor Disorders Network. Comparison of 2 transvaginal surgical approaches and perioperative behavioral therapy for apical vaginal prolapse: the OPTIMAL randomized trial. JAMA. 2014;311(10):1023–1034.
- Brandes S, Coburn M, Armenakas N, McAninch J. Diagnosis and management of ureteric injury: an evidence-based analysis. BJU Int. 2004;94(3):277–289.
- Kwon CH, Goldberg RP, Koduri S, Sand PK. The use of intraoperative cystoscopy in major vaginal and urogynecologic surgeries. Am J Obstet Gynecol. 2002;187(6):1466–1471.
- Adams-Piper ER, Guaderrama NM, Chen Q, Whitcomb EL. Impact of surgical training on the performance of proposed quality measures for hysterectomy for pelvic organ prolapse. Am J Obstet Gynecol. 2017;216(6):588.e1–588.e5.
- Siff LN, Unger CA, Jelovsek JE, Paraiso MF, Ridgeway BM Barber MD. Assessing ureteral patency using 10% dextrose cystoscopy fluid: evaluation of urinary tract infection rates. Am J Obstet Gynecol. 2016;215(1):74.e1–74.e6.
- Espaillat-Rijo L, Siff L, Alas AN, et al. Intraoperative cystoscopic evaluation of ureteral patency: a randomized controlled trial. Obstet Gynecol. 2016;128(6):1378–1383.
Using cystoscopy to evaluate ureteral efflux and bladder integrity following benign gynecologic surgery increases the detection rate of urinary tract injuries.1 Currently, it is standard of care to perform a cystoscopy following anti-incontinence procedures, but there is no consensus among ObGyns regarding the use of universal cystoscopy following benign gynecologic surgery.2 A number of studies, however, have suggested potential best practices for evaluating urinary tract injury during pelvic surgery for benign gynecologic conditions.
Pelvic surgeries for benign gynecologic conditions, including fibroids, menorrhagia, and pelvic organ prolapse (POP), are common. More than 500,000 hysterectomies are performed annually in the United States, and up to 11% of women will undergo at least one surgery for POP or urinary incontinence in their lifetime.3,4 During gynecologic surgery, the urinary tract is at risk, and the injury rate ranges from 0.02% to 2% for ureteral injury and from 1% to 5% for bladder injury.5,6
In a recent large randomized controlled trial, the rate of intraoperative ureteral obstruction following uterosacral ligament suspension (USLS) was 3.2%.7 Vaginal vault suspensions, as well as other vaginal cuff closure techniques, are common procedures associated with urinary tract injury.8 Additionally, ureteral injury during surgery for POP occurs in as many as 2% of anterior vaginal wall repairs.9
It is well documented that a delay in diagnosis of ureteral and/or bladder injuries is associated with increased morbidity, including the need for additional surgery to repair the injury; in addition, significant delay in identifying an injury may lead to subsequent sequela, such as renal injury and fistula formation.8
A large study in California found that 36.5% of hysterectomies performed for POP were performed by general gynecologists.10 General ObGyns performing these surgeries therefore must understand the risk of urinary tract injury during hysterectomy and reconstructive pelvic procedures so that they can appropriately identify, evaluate, and repair injuries in a timely fashion.
The best way to identify urinary tract injury at the time of gynecologic surgery is by cystoscopy, including a bladder survey and ureteral efflux evaluation. When should a cystoscopy be performed, and what is the best method for visualizing ureteral efflux? Can instituting universal cystoscopy for all gynecologic procedures make a difference in the rate of injury detection? In this Update, we summarize the data from 4 studies that help to answer these questions.
Continue to: About 30% of urinary tract injuries...
About 30% of urinary tract injuries identified prior to cystoscopy at hysterectomy (which detected 5 of 6 injuries)
Vakili B, Chesson RR, Kyle BL, et al. The incidence of urinary tract injury during hysterectomy: a prospective analysis based on universal cystoscopy. Am J Obstet Gynecol. 2005;192(5):1599–1604.
Vakili and colleagues conducted a multicenter prospective cohort study of women undergoing hysterectomy for benign indications; cystoscopy was performed in all cases. The 3 hospitals involved were all part of the Louisiana State University Health system. The investigators’ goal was to determine the rate of urinary tract injury in this patient population at the time of intraoperative cystoscopy.
Intraoperative cystoscopy beats visual evaluation
Four hundred and seventy-one women underwent hysterectomy and had intraoperative cystoscopy, including evaluation of ureteral patency with administration of intravenous (IV) indigo carmine. Patients underwent abdominal, vaginal, or laparoscopic hysterectomy, and 54 (11.4%) had concurrent POP or anti-incontinence procedures. The majority underwent an abdominal hysterectomy (59%), 31% had a vaginal hysterectomy, and 10% had a laparoscopic-assisted vaginal hysterectomy or total laparoscopic hysterectomy.
Rate of urinary tract injuries. The total urinary tract injury rate detected by cystoscopy was 4.8%. The ureteral injury rate was 1.7%, and the bladder injury rate was 3.6%. A combined ureteral and bladder injury occurred in 2 women.
Surgery for POP significantly increased the risk of ureteral injury (7.3% vs 1.2%; P = .025). All cases of ureteral injury during POP surgery occurred during USLS. There was a trend toward a higher rate of bladder injury in the group with concurrent anti-incontinence surgery (12.5% vs 3.1%; P = .049). Regarding the route of hysterectomy, the vaginal approach had the highest rate of ureteral injury; however, when prolapse cases were removed from the analysis, there were no differences between the abdominal, vaginal, and laparoscopic approaches for ureteral or bladder injuries.
Injury detection with cystoscopy. Importantly, the authors found that only 30% of injuries were identified prior to performing intraoperative cystoscopy. The majority of these were bladder injuries. In addition, despite visual confirmation of ureteral peristalsis during abdominal hysterectomy, when intraoperative cystoscopy was performed with evaluation for ureteral efflux, 5 of 6 ureteral injury cases were identified. The authors reported 1 postoperative vesicovaginal fistula and concluded that it was likely due to an unrecognized bladder injury. No other undetected injuries were identified.
Notably, no complications occurred as a result of cystoscopy.
Multiple surgical indications reflect real-world scenario
The study included physicians from 3 different hospitals and all routes of hysterectomy for multiple benign gynecologic indications as well as concomitant pelvic reconstructive procedures. While this enhances the generalizability of the study results, all study sites were located in Louisiana at hospitals with resident trainee involvement. Additionally, this study confirms previous retrospective studies that reported higher rates of injury with pelvic reconstructive procedures.
The study is limited by the inability to blind surgeons, which may have resulted in the surgeons altering their techniques and/or having a heightened awareness of the urinary tract. However, their rates of ureteral and bladder injuries were slightly higher than previously reported rates, suggesting that the procedures inherently carry risk. The study is further limited by the lack of a retrospective comparison group of hysterectomy without routine cystoscopy and a longer follow-up period that may have revealed additional missed delayed urologic injuries.
The rate of urinary tract injury, including both bladder and ureteral injuries, was more than 4% at the time of hysterectomy for benign conditions. Using intraoperative peristalsis or normal ureteral caliber could result in a false sense of security since these are not reliable signs of ureteral integrity. The majority of urinary tract injuries will not be identified without cystoscopic evaluation.
Continue to: Universal cystoscopy policy...
Universal cystoscopy policy proves protective, surgeon adherence is high
Chi AM, Curran DS, Morgan DM, Fenner DE, Swenson CW. Universal cystoscopy after benign hysterectomy: examining the effects of an institutional policy. Obstet Gynecol. 2016;127(2):369–375.
In a retrospective cohort study, Chi and colleagues evaluated urinary tract injuries at the time of hysterectomy before and after the institution of a universal cystoscopy policy. At the time of policy implementation at the University of Michigan, all faculty who performed hysterectomies attended a cystoscopy workshop. Attending physicians without prior cystoscopy training also were proctored in the operating room for 3 sessions and were required to demonstrate competency with bladder survey, visualizing ureteral efflux, and urethral assessment. Indigo carmine was used to visualize ureteral efflux.
Detection of urologic injury almost doubled with cystoscopy
A total of 2,822 hysterectomies were included in the study, with 973 in the pre–universal cystoscopy group and 1,849 in the post–universal cystoscopy group. The study period was 7 years, and data on complications were abstracted for 1 year after the completion of the study period.
The primary outcome had 3 components:
- the rate of urologic injury before and after the policy
- the cystoscopy detection rate of urologic injury
- the adherence rate to the policy.
The overall rate of bladder and ureteral injury was 2.1%; the rate of injury during pre–universal screening was 2.6%, and during post–universal screening was 1.8%. The intraoperative detection rate of injury nearly doubled, from 24% to 47%, when intraoperative cystoscopy was utilized. In addition, the percentage of delayed urologic complications decreased from 28% to 5.9% (P = .03) following implementation of the universal cystoscopy policy. With regard to surgeon adherence, cystoscopy was documented in 86.1% of the hysterectomy cases after the policy was implemented compared with 35.7% of cases before the policy.
The investigators performed a cost analysis and found that hospital costs were nearly twice as much if a delayed urologic injury was diagnosed.
Study had many strengths
This study evaluated aspects of implementing quality initiatives after proper training and proctoring of a procedure. The authors compared very large cohorts from a busy academic medical center in which surgeon adherence with routine cystoscopy was high. The majority of patient outcomes were tracked for an extended period following surgery, thereby minimizing the risk of missing delayed urologic injuries. Notably, however, there was shorter follow-up time for the post–universal cystoscopy group, which could result in underestimating the rate of delayed urologic injuries in this cohort.
Instituting a universal cystoscopy policy for hysterectomy was associated with a significant decrease in delayed postoperative urinary tract complications and an increase in the intraoperative detection rate of urologic injuries. Intraoperative detection and repair of a urinary tract injury is cost-effective compared with a delayed diagnosis.
Continue to: Cystoscopy reveals ureteral obstruction...
Cystoscopy reveals ureteral obstruction during various vaginal POP repair procedures
Gustilo-Ashby AM, Jelovsek JE, Barber MD, Yoo EH, Paraiso MF, Walters MD. The incidence of ureteral obstruction and the value of intraoperative cystoscopy during vaginal surgery for pelvic organ prolapse. Am J Obstet Gynecol. 2006;194(5):1478–1485.
To determine the rate of ureteral obstruction and ureteral injury during vaginal surgery for POP and the accuracy of using intraoperative cystoscopy to prevent upper urinary tract morbidity, Gustilo-Ashby and colleagues performed a retrospective review study of a large patient cohort.
Cystoscopy with indigo carmine is highly sensitive
The study included 700 patients who underwent vaginal surgery for anterior and/or apical POP. Patients had 1 or more of the following anterior and apical prolapse repair procedures: USLS (51%), distal McCall culdeplasty (26%), proximal McCall culdeplasty (29%), anterior colporrhaphy (82%), and colpocleisis (1.4%). Of note, distal McCall culdeplasty was defined as incorporation of the “vaginal epithelium into the uterosacral plication,” while proximal McCall culdeplasty involved plication of “the uterosacral ligaments in the midline proximal to the vaginal cuff.” All patients were given IV indigo carmine to aid in visualizing ureteral efflux.
The majority of patients had a hysterectomy (56%). When accounting for rare false-positive and negative cystoscopy results, the overall ureteral obstruction rate was 5.1% and the ureteral injury rate was 0.9%. The majority of obstructions occurred with USLS (5.9%), proximal McCall culdeplasty (4.4%), and colpocleisis (4.2%). Ureteral injuries occurred only in 6 cases: 3 USLS and 3 proximal McCall culdeplasty procedures.
Based on these findings, the authors calculated that cystoscopy at the time of vaginal surgery for anterior and/or apical prolapse has a sensitivity of 94.4% and a specificity of 99.5% for detecting ureteral obstruction. The positive predictive value of cystoscopy with the use of indigo carmine for detection of ureteral obstruction is 91.9% and the negative predictive value is 99.7%.
Impact of indigo carmine’s unavailability
This study’s strengths include its large sample size and the variety of surgical approaches used for repair of anterior vaginal wall and apical prolapse. Its retrospective design, however, is a limitation; this could result in underreporting of ureteral injuries if patients received care at another institution after surgery. Furthermore, it is unclear if cystoscopy would be as predictive of ureteral injury without the use of indigo carmine, which is no longer available at most institutions.
The utility of cystoscopy with IV indigo carmine as a screening test for ureteral obstruction is highlighted by the fact that most obstructions were relieved by intraoperative suture removal following positive cystoscopy. McCall culdeplasty procedures are commonly performed by general ObGyns at the time of vaginal hysterectomy. It is therefore important to note that rates of ureteral obstruction after proximal McCall culdeplasty were only slightly lower than those after USLS.
Continue to: Sodium fluorescein and 10% dextrose...
Sodium fluorescein and 10% dextrose provide clear visibility of ureteral jets in cystoscopy
Espaillat-Rijo L, Siff L, Alas AN, et al. Intraoperative cystoscopic evaluation of ureteral patency: a randomized controlled trial. Obstet Gynecol. 2016;128(6):1378–1383.
In a multicenter randomized controlled trial, Espaillat-Rijo and colleagues compared various methods for visualizing ureteral efflux in participants who underwent gynecologic or urogynecologic procedures in which cystoscopy was performed.
Study compared 4 media
The investigators enrolled 176 participants (174 completed the trial) and randomly assigned them to receive 1 of 4 modalities: 1) normal saline as a bladder distention medium (control), 2) 10% dextrose as a bladder distention medium, 3) 200 mg oral phenazopyridine given 30 minutes prior to cystoscopy, or 4) 50 mg IV sodium fluorescein at the start of cystoscopy. Indigo carmine was not included in this study because it has not been routinely available since 2014.
Surgeons were asked to categorize the ureteral jets as “clearly visible,” “somewhat visible,” or “not visible.”
The primary outcome was subjective visibility of the ureteral jet with each modality during cystoscopy. Secondary outcomes included surgeon satisfaction, adverse reactions to the modality used, postoperative urinary tract infection, postoperative urinary retention, and delayed diagnosis of ureteral injury.
Visibility assessment results. Overall, ureteral jets were “clearly visible” in 125 cases (71%) compared with “somewhat visible” in 45 (25.6%) and “not visible” in 4 (2.3%) cases. There was a statistically significant difference between the 4 groups. Use of sodium fluorescein and 10% dextrose resulted in significantly better visualization of ureteral jets (P < .001 and P = .004, respectively) compared with the control group. Visibility with phenazopyridine was not significantly different from that in the control group or in the 10% dextrose group (FIGURE).
Surgeon satisfaction was highest with 10% dextrose and sodium fluorescein. In 6 cases, the surgeon was not satisfied with visualization of the ureteral jets and relied on fluorescein (5 times) or 10% dextrose (1 time) to ultimately see efflux. No significant adverse events occurred; the rate of urinary tract infection was 24.1% and did not differ between groups.
Results are widely generalizable
This was a well-designed randomized multicenter trial that included both benign gynecologic and urogynecologic procedures, thus strengthening the generalizability of the study. The study was timely since proven methods for visualization of ureteral patency became limited with the withdrawal of commercially available indigo carmine, the previous gold standard.
Intravenous sodium fluorescein and 10% dextrose as bladder distention media can both safely be used to visualize ureteral efflux and result in high surgeon satisfaction. Although 10% dextrose has been associated with higher rates of postoperative urinary tract infection,11 this was not found to be the case in this study. Preoperative administration of oral phenazopyridine was no different from the control modality with regard to visibility and surgeon satisfaction.
Continue to: The cost-effectiveness consideration
The cost-effectiveness consideration
The debate around universal cystoscopy following benign gynecologic surgery is ongoing.
The studies discussed in this Update demonstrate that cystoscopy following hysterectomy for benign indications:
- is superior to visualizing ureteral peristalsis
- increases detection of urinary tract injuries, and
- decreases delayed urologic injuries.
Although these articles emphasize the importance of detecting urologic injury, the picture would not be complete without mention of cost-effectiveness. Only one study, from 2001, has evaluated the cost-effectiveness of universal cystoscopy.1 Those authors concluded that universal cystoscopy is cost-effective only when the rate of urologic injury is 1.5% to 2%, but this conclusion, admittedly, was limited by the lack of data on medicolegal settlements, outpatient expenses, and nonmedical-related economic loss from decreased productivity. Given the extensive changes that have occurred in medical practice over the last 17 years and the emphasis on quality metrics and safety, an updated analysis would be needed to make definitive conclusions about cost-effectiveness.
While this Update cannot settle the ongoing debate of universal cystoscopy in gynecology, it is important to remember that the American College of Obstetricians and Gynecologists and the American Urogynecologic Society recommend cystoscopy following all surgeries for pelvic organ prolapse and stress urinary incontinence.2
References
- Visco AG, Taber KH, Weidner AD, Barber MD, Myers ER. Cost-effectiveness of universal cystoscopy to identify ureteral injury at hysterectomy. Obstet Gynecol. 2001;97(5 pt 1):685–692.
- ACOG Committee on Practice Bulletins–Gynecology and the American Urogynecologic Society. Urinary incontinence in women. Female Pelvic Med Reconstr Surg. 2015;21(6):304–314.
Share your thoughts! Send your Letter to the Editor to [email protected]. Please include your name and the city and state in which you practice.
Using cystoscopy to evaluate ureteral efflux and bladder integrity following benign gynecologic surgery increases the detection rate of urinary tract injuries.1 Currently, it is standard of care to perform a cystoscopy following anti-incontinence procedures, but there is no consensus among ObGyns regarding the use of universal cystoscopy following benign gynecologic surgery.2 A number of studies, however, have suggested potential best practices for evaluating urinary tract injury during pelvic surgery for benign gynecologic conditions.
Pelvic surgeries for benign gynecologic conditions, including fibroids, menorrhagia, and pelvic organ prolapse (POP), are common. More than 500,000 hysterectomies are performed annually in the United States, and up to 11% of women will undergo at least one surgery for POP or urinary incontinence in their lifetime.3,4 During gynecologic surgery, the urinary tract is at risk, and the injury rate ranges from 0.02% to 2% for ureteral injury and from 1% to 5% for bladder injury.5,6
In a recent large randomized controlled trial, the rate of intraoperative ureteral obstruction following uterosacral ligament suspension (USLS) was 3.2%.7 Vaginal vault suspensions, as well as other vaginal cuff closure techniques, are common procedures associated with urinary tract injury.8 Additionally, ureteral injury during surgery for POP occurs in as many as 2% of anterior vaginal wall repairs.9
It is well documented that a delay in diagnosis of ureteral and/or bladder injuries is associated with increased morbidity, including the need for additional surgery to repair the injury; in addition, significant delay in identifying an injury may lead to subsequent sequela, such as renal injury and fistula formation.8
A large study in California found that 36.5% of hysterectomies performed for POP were performed by general gynecologists.10 General ObGyns performing these surgeries therefore must understand the risk of urinary tract injury during hysterectomy and reconstructive pelvic procedures so that they can appropriately identify, evaluate, and repair injuries in a timely fashion.
The best way to identify urinary tract injury at the time of gynecologic surgery is by cystoscopy, including a bladder survey and ureteral efflux evaluation. When should a cystoscopy be performed, and what is the best method for visualizing ureteral efflux? Can instituting universal cystoscopy for all gynecologic procedures make a difference in the rate of injury detection? In this Update, we summarize the data from 4 studies that help to answer these questions.
Continue to: About 30% of urinary tract injuries...
About 30% of urinary tract injuries identified prior to cystoscopy at hysterectomy (which detected 5 of 6 injuries)
Vakili B, Chesson RR, Kyle BL, et al. The incidence of urinary tract injury during hysterectomy: a prospective analysis based on universal cystoscopy. Am J Obstet Gynecol. 2005;192(5):1599–1604.
Vakili and colleagues conducted a multicenter prospective cohort study of women undergoing hysterectomy for benign indications; cystoscopy was performed in all cases. The 3 hospitals involved were all part of the Louisiana State University Health system. The investigators’ goal was to determine the rate of urinary tract injury in this patient population at the time of intraoperative cystoscopy.
Intraoperative cystoscopy beats visual evaluation
Four hundred and seventy-one women underwent hysterectomy and had intraoperative cystoscopy, including evaluation of ureteral patency with administration of intravenous (IV) indigo carmine. Patients underwent abdominal, vaginal, or laparoscopic hysterectomy, and 54 (11.4%) had concurrent POP or anti-incontinence procedures. The majority underwent an abdominal hysterectomy (59%), 31% had a vaginal hysterectomy, and 10% had a laparoscopic-assisted vaginal hysterectomy or total laparoscopic hysterectomy.
Rate of urinary tract injuries. The total urinary tract injury rate detected by cystoscopy was 4.8%. The ureteral injury rate was 1.7%, and the bladder injury rate was 3.6%. A combined ureteral and bladder injury occurred in 2 women.
Surgery for POP significantly increased the risk of ureteral injury (7.3% vs 1.2%; P = .025). All cases of ureteral injury during POP surgery occurred during USLS. There was a trend toward a higher rate of bladder injury in the group with concurrent anti-incontinence surgery (12.5% vs 3.1%; P = .049). Regarding the route of hysterectomy, the vaginal approach had the highest rate of ureteral injury; however, when prolapse cases were removed from the analysis, there were no differences between the abdominal, vaginal, and laparoscopic approaches for ureteral or bladder injuries.
Injury detection with cystoscopy. Importantly, the authors found that only 30% of injuries were identified prior to performing intraoperative cystoscopy. The majority of these were bladder injuries. In addition, despite visual confirmation of ureteral peristalsis during abdominal hysterectomy, when intraoperative cystoscopy was performed with evaluation for ureteral efflux, 5 of 6 ureteral injury cases were identified. The authors reported 1 postoperative vesicovaginal fistula and concluded that it was likely due to an unrecognized bladder injury. No other undetected injuries were identified.
Notably, no complications occurred as a result of cystoscopy.
Multiple surgical indications reflect real-world scenario
The study included physicians from 3 different hospitals and all routes of hysterectomy for multiple benign gynecologic indications as well as concomitant pelvic reconstructive procedures. While this enhances the generalizability of the study results, all study sites were located in Louisiana at hospitals with resident trainee involvement. Additionally, this study confirms previous retrospective studies that reported higher rates of injury with pelvic reconstructive procedures.
The study is limited by the inability to blind surgeons, which may have resulted in the surgeons altering their techniques and/or having a heightened awareness of the urinary tract. However, their rates of ureteral and bladder injuries were slightly higher than previously reported rates, suggesting that the procedures inherently carry risk. The study is further limited by the lack of a retrospective comparison group of hysterectomy without routine cystoscopy and a longer follow-up period that may have revealed additional missed delayed urologic injuries.
The rate of urinary tract injury, including both bladder and ureteral injuries, was more than 4% at the time of hysterectomy for benign conditions. Using intraoperative peristalsis or normal ureteral caliber could result in a false sense of security since these are not reliable signs of ureteral integrity. The majority of urinary tract injuries will not be identified without cystoscopic evaluation.
Continue to: Universal cystoscopy policy...
Universal cystoscopy policy proves protective, surgeon adherence is high
Chi AM, Curran DS, Morgan DM, Fenner DE, Swenson CW. Universal cystoscopy after benign hysterectomy: examining the effects of an institutional policy. Obstet Gynecol. 2016;127(2):369–375.
In a retrospective cohort study, Chi and colleagues evaluated urinary tract injuries at the time of hysterectomy before and after the institution of a universal cystoscopy policy. At the time of policy implementation at the University of Michigan, all faculty who performed hysterectomies attended a cystoscopy workshop. Attending physicians without prior cystoscopy training also were proctored in the operating room for 3 sessions and were required to demonstrate competency with bladder survey, visualizing ureteral efflux, and urethral assessment. Indigo carmine was used to visualize ureteral efflux.
Detection of urologic injury almost doubled with cystoscopy
A total of 2,822 hysterectomies were included in the study, with 973 in the pre–universal cystoscopy group and 1,849 in the post–universal cystoscopy group. The study period was 7 years, and data on complications were abstracted for 1 year after the completion of the study period.
The primary outcome had 3 components:
- the rate of urologic injury before and after the policy
- the cystoscopy detection rate of urologic injury
- the adherence rate to the policy.
The overall rate of bladder and ureteral injury was 2.1%; the rate of injury during pre–universal screening was 2.6%, and during post–universal screening was 1.8%. The intraoperative detection rate of injury nearly doubled, from 24% to 47%, when intraoperative cystoscopy was utilized. In addition, the percentage of delayed urologic complications decreased from 28% to 5.9% (P = .03) following implementation of the universal cystoscopy policy. With regard to surgeon adherence, cystoscopy was documented in 86.1% of the hysterectomy cases after the policy was implemented compared with 35.7% of cases before the policy.
The investigators performed a cost analysis and found that hospital costs were nearly twice as much if a delayed urologic injury was diagnosed.
Study had many strengths
This study evaluated aspects of implementing quality initiatives after proper training and proctoring of a procedure. The authors compared very large cohorts from a busy academic medical center in which surgeon adherence with routine cystoscopy was high. The majority of patient outcomes were tracked for an extended period following surgery, thereby minimizing the risk of missing delayed urologic injuries. Notably, however, there was shorter follow-up time for the post–universal cystoscopy group, which could result in underestimating the rate of delayed urologic injuries in this cohort.
Instituting a universal cystoscopy policy for hysterectomy was associated with a significant decrease in delayed postoperative urinary tract complications and an increase in the intraoperative detection rate of urologic injuries. Intraoperative detection and repair of a urinary tract injury is cost-effective compared with a delayed diagnosis.
Continue to: Cystoscopy reveals ureteral obstruction...
Cystoscopy reveals ureteral obstruction during various vaginal POP repair procedures
Gustilo-Ashby AM, Jelovsek JE, Barber MD, Yoo EH, Paraiso MF, Walters MD. The incidence of ureteral obstruction and the value of intraoperative cystoscopy during vaginal surgery for pelvic organ prolapse. Am J Obstet Gynecol. 2006;194(5):1478–1485.
To determine the rate of ureteral obstruction and ureteral injury during vaginal surgery for POP and the accuracy of using intraoperative cystoscopy to prevent upper urinary tract morbidity, Gustilo-Ashby and colleagues performed a retrospective review study of a large patient cohort.
Cystoscopy with indigo carmine is highly sensitive
The study included 700 patients who underwent vaginal surgery for anterior and/or apical POP. Patients had 1 or more of the following anterior and apical prolapse repair procedures: USLS (51%), distal McCall culdeplasty (26%), proximal McCall culdeplasty (29%), anterior colporrhaphy (82%), and colpocleisis (1.4%). Of note, distal McCall culdeplasty was defined as incorporation of the “vaginal epithelium into the uterosacral plication,” while proximal McCall culdeplasty involved plication of “the uterosacral ligaments in the midline proximal to the vaginal cuff.” All patients were given IV indigo carmine to aid in visualizing ureteral efflux.
The majority of patients had a hysterectomy (56%). When accounting for rare false-positive and negative cystoscopy results, the overall ureteral obstruction rate was 5.1% and the ureteral injury rate was 0.9%. The majority of obstructions occurred with USLS (5.9%), proximal McCall culdeplasty (4.4%), and colpocleisis (4.2%). Ureteral injuries occurred only in 6 cases: 3 USLS and 3 proximal McCall culdeplasty procedures.
Based on these findings, the authors calculated that cystoscopy at the time of vaginal surgery for anterior and/or apical prolapse has a sensitivity of 94.4% and a specificity of 99.5% for detecting ureteral obstruction. The positive predictive value of cystoscopy with the use of indigo carmine for detection of ureteral obstruction is 91.9% and the negative predictive value is 99.7%.
Impact of indigo carmine’s unavailability
This study’s strengths include its large sample size and the variety of surgical approaches used for repair of anterior vaginal wall and apical prolapse. Its retrospective design, however, is a limitation; this could result in underreporting of ureteral injuries if patients received care at another institution after surgery. Furthermore, it is unclear if cystoscopy would be as predictive of ureteral injury without the use of indigo carmine, which is no longer available at most institutions.
The utility of cystoscopy with IV indigo carmine as a screening test for ureteral obstruction is highlighted by the fact that most obstructions were relieved by intraoperative suture removal following positive cystoscopy. McCall culdeplasty procedures are commonly performed by general ObGyns at the time of vaginal hysterectomy. It is therefore important to note that rates of ureteral obstruction after proximal McCall culdeplasty were only slightly lower than those after USLS.
Continue to: Sodium fluorescein and 10% dextrose...
Sodium fluorescein and 10% dextrose provide clear visibility of ureteral jets in cystoscopy
Espaillat-Rijo L, Siff L, Alas AN, et al. Intraoperative cystoscopic evaluation of ureteral patency: a randomized controlled trial. Obstet Gynecol. 2016;128(6):1378–1383.
In a multicenter randomized controlled trial, Espaillat-Rijo and colleagues compared various methods for visualizing ureteral efflux in participants who underwent gynecologic or urogynecologic procedures in which cystoscopy was performed.
Study compared 4 media
The investigators enrolled 176 participants (174 completed the trial) and randomly assigned them to receive 1 of 4 modalities: 1) normal saline as a bladder distention medium (control), 2) 10% dextrose as a bladder distention medium, 3) 200 mg oral phenazopyridine given 30 minutes prior to cystoscopy, or 4) 50 mg IV sodium fluorescein at the start of cystoscopy. Indigo carmine was not included in this study because it has not been routinely available since 2014.
Surgeons were asked to categorize the ureteral jets as “clearly visible,” “somewhat visible,” or “not visible.”
The primary outcome was subjective visibility of the ureteral jet with each modality during cystoscopy. Secondary outcomes included surgeon satisfaction, adverse reactions to the modality used, postoperative urinary tract infection, postoperative urinary retention, and delayed diagnosis of ureteral injury.
Visibility assessment results. Overall, ureteral jets were “clearly visible” in 125 cases (71%) compared with “somewhat visible” in 45 (25.6%) and “not visible” in 4 (2.3%) cases. There was a statistically significant difference between the 4 groups. Use of sodium fluorescein and 10% dextrose resulted in significantly better visualization of ureteral jets (P < .001 and P = .004, respectively) compared with the control group. Visibility with phenazopyridine was not significantly different from that in the control group or in the 10% dextrose group (FIGURE).
Surgeon satisfaction was highest with 10% dextrose and sodium fluorescein. In 6 cases, the surgeon was not satisfied with visualization of the ureteral jets and relied on fluorescein (5 times) or 10% dextrose (1 time) to ultimately see efflux. No significant adverse events occurred; the rate of urinary tract infection was 24.1% and did not differ between groups.
Results are widely generalizable
This was a well-designed randomized multicenter trial that included both benign gynecologic and urogynecologic procedures, thus strengthening the generalizability of the study. The study was timely since proven methods for visualization of ureteral patency became limited with the withdrawal of commercially available indigo carmine, the previous gold standard.
Intravenous sodium fluorescein and 10% dextrose as bladder distention media can both safely be used to visualize ureteral efflux and result in high surgeon satisfaction. Although 10% dextrose has been associated with higher rates of postoperative urinary tract infection,11 this was not found to be the case in this study. Preoperative administration of oral phenazopyridine was no different from the control modality with regard to visibility and surgeon satisfaction.
Continue to: The cost-effectiveness consideration
The cost-effectiveness consideration
The debate around universal cystoscopy following benign gynecologic surgery is ongoing.
The studies discussed in this Update demonstrate that cystoscopy following hysterectomy for benign indications:
- is superior to visualizing ureteral peristalsis
- increases detection of urinary tract injuries, and
- decreases delayed urologic injuries.
Although these articles emphasize the importance of detecting urologic injury, the picture would not be complete without mention of cost-effectiveness. Only one study, from 2001, has evaluated the cost-effectiveness of universal cystoscopy.1 Those authors concluded that universal cystoscopy is cost-effective only when the rate of urologic injury is 1.5% to 2%, but this conclusion, admittedly, was limited by the lack of data on medicolegal settlements, outpatient expenses, and nonmedical-related economic loss from decreased productivity. Given the extensive changes that have occurred in medical practice over the last 17 years and the emphasis on quality metrics and safety, an updated analysis would be needed to make definitive conclusions about cost-effectiveness.
While this Update cannot settle the ongoing debate of universal cystoscopy in gynecology, it is important to remember that the American College of Obstetricians and Gynecologists and the American Urogynecologic Society recommend cystoscopy following all surgeries for pelvic organ prolapse and stress urinary incontinence.2
References
- Visco AG, Taber KH, Weidner AD, Barber MD, Myers ER. Cost-effectiveness of universal cystoscopy to identify ureteral injury at hysterectomy. Obstet Gynecol. 2001;97(5 pt 1):685–692.
- ACOG Committee on Practice Bulletins–Gynecology and the American Urogynecologic Society. Urinary incontinence in women. Female Pelvic Med Reconstr Surg. 2015;21(6):304–314.
Share your thoughts! Send your Letter to the Editor to [email protected]. Please include your name and the city and state in which you practice.
- Ibeanu OA, Chesson RR, Echols KT, Nieves M, Busangu F, Nolan TE. Urinary tract injury during hysterectomy based on universal cystoscopy. Obstet Gynecol. 2009;113(1):6–10.
- ACOG Committee on Practice Bulletins–Gynecology and the American Urogynecologic Society. Urinary incontinence in women. Female Pelvic Med Reconstr Surg. 2015;21(6):304–314.
- Wilcox LS, Koonin LM, Pokras R, Strauss LT, Xia Z, Peterson HB. Hysterectomy in the United States, 1988–1990. Obstet Gynecol. 1994;83(4):549–555.
- Olsen AL, Smith VJ, Bergstrom JO, Colling JC, Clark AL. Epidemiology of surgically managed pelvic organ prolapse and urinary incontinence. Obstet Gynecol. 1997;89(4):501–506.
- Mäkinen J, Johansson J, Tomás C, et al. Morbidity of 10,110 hysterectomies by type of approach. Hum Reprod. 2001;16(7):1473–1478.
- Gilmour DT, Dwyer PL, Carey MP. Lower urinary tract injury during gynecologic surgery and its detection by intraoperative cystoscopy. Obstet Gynecol. 1999;94(5 pt 2):883–889.
- Barber MD, Brubaker L, Burgio KL, et al; Eunice Kennedy Schriver National Institute of Child Health and Human Development Pelvic Floor Disorders Network. Comparison of 2 transvaginal surgical approaches and perioperative behavioral therapy for apical vaginal prolapse: the OPTIMAL randomized trial. JAMA. 2014;311(10):1023–1034.
- Brandes S, Coburn M, Armenakas N, McAninch J. Diagnosis and management of ureteric injury: an evidence-based analysis. BJU Int. 2004;94(3):277–289.
- Kwon CH, Goldberg RP, Koduri S, Sand PK. The use of intraoperative cystoscopy in major vaginal and urogynecologic surgeries. Am J Obstet Gynecol. 2002;187(6):1466–1471.
- Adams-Piper ER, Guaderrama NM, Chen Q, Whitcomb EL. Impact of surgical training on the performance of proposed quality measures for hysterectomy for pelvic organ prolapse. Am J Obstet Gynecol. 2017;216(6):588.e1–588.e5.
- Siff LN, Unger CA, Jelovsek JE, Paraiso MF, Ridgeway BM Barber MD. Assessing ureteral patency using 10% dextrose cystoscopy fluid: evaluation of urinary tract infection rates. Am J Obstet Gynecol. 2016;215(1):74.e1–74.e6.
- Espaillat-Rijo L, Siff L, Alas AN, et al. Intraoperative cystoscopic evaluation of ureteral patency: a randomized controlled trial. Obstet Gynecol. 2016;128(6):1378–1383.
- Ibeanu OA, Chesson RR, Echols KT, Nieves M, Busangu F, Nolan TE. Urinary tract injury during hysterectomy based on universal cystoscopy. Obstet Gynecol. 2009;113(1):6–10.
- ACOG Committee on Practice Bulletins–Gynecology and the American Urogynecologic Society. Urinary incontinence in women. Female Pelvic Med Reconstr Surg. 2015;21(6):304–314.
- Wilcox LS, Koonin LM, Pokras R, Strauss LT, Xia Z, Peterson HB. Hysterectomy in the United States, 1988–1990. Obstet Gynecol. 1994;83(4):549–555.
- Olsen AL, Smith VJ, Bergstrom JO, Colling JC, Clark AL. Epidemiology of surgically managed pelvic organ prolapse and urinary incontinence. Obstet Gynecol. 1997;89(4):501–506.
- Mäkinen J, Johansson J, Tomás C, et al. Morbidity of 10,110 hysterectomies by type of approach. Hum Reprod. 2001;16(7):1473–1478.
- Gilmour DT, Dwyer PL, Carey MP. Lower urinary tract injury during gynecologic surgery and its detection by intraoperative cystoscopy. Obstet Gynecol. 1999;94(5 pt 2):883–889.
- Barber MD, Brubaker L, Burgio KL, et al; Eunice Kennedy Schriver National Institute of Child Health and Human Development Pelvic Floor Disorders Network. Comparison of 2 transvaginal surgical approaches and perioperative behavioral therapy for apical vaginal prolapse: the OPTIMAL randomized trial. JAMA. 2014;311(10):1023–1034.
- Brandes S, Coburn M, Armenakas N, McAninch J. Diagnosis and management of ureteric injury: an evidence-based analysis. BJU Int. 2004;94(3):277–289.
- Kwon CH, Goldberg RP, Koduri S, Sand PK. The use of intraoperative cystoscopy in major vaginal and urogynecologic surgeries. Am J Obstet Gynecol. 2002;187(6):1466–1471.
- Adams-Piper ER, Guaderrama NM, Chen Q, Whitcomb EL. Impact of surgical training on the performance of proposed quality measures for hysterectomy for pelvic organ prolapse. Am J Obstet Gynecol. 2017;216(6):588.e1–588.e5.
- Siff LN, Unger CA, Jelovsek JE, Paraiso MF, Ridgeway BM Barber MD. Assessing ureteral patency using 10% dextrose cystoscopy fluid: evaluation of urinary tract infection rates. Am J Obstet Gynecol. 2016;215(1):74.e1–74.e6.
- Espaillat-Rijo L, Siff L, Alas AN, et al. Intraoperative cystoscopic evaluation of ureteral patency: a randomized controlled trial. Obstet Gynecol. 2016;128(6):1378–1383.
Take these steps to improve your flu season preparedness
Last year’s influenza season was severe enough that hospitals around the United States set up special evaluation areas beyond their emergency departments, at times spilling over to tents or other temporary structures in what otherwise would be parking lots. The scale and potential severity of the annual epidemic can be difficult to convey to our patients, who sometimes say “just the flu” to refer to an illness responsible for more than 170 pediatric deaths in the United States this past year.1 The Centers for Disease Control and Prevention (CDC) recently updated its 5-year estimates of influenza-related deaths in the United States; influenza mortality ranges from about 12,000 deaths in a mild season (such as 2011-2012) to 56,000 in a more severe season (eg, 2012-2013).2
Although influenza cannot be completely prevented, the following strategies can help reduce the risk for the illness and limit its severity if contracted.
Prevention
Strategy 1: Vaccinate against influenza
While the efficacy of vaccines varies from year to year, vaccination remains the core of influenza prevention efforts. In this decade, vaccine effectiveness has ranged from 19% to 60%.3 However, models suggest that even when the vaccine is only 20% effective, vaccinating 140 million people (the average number of doses delivered annually in the United States over the past 5 seasons) prevents 21 million infections, 130,000 hospitalizations, and more than 61,000 deaths.4 In a case-control study, Flannery et al found that vaccination was 65% effective in preventing laboratory-confirmed influenza-associated death in children over 4 seasons (July 2010 through June 2014).5
Deciding who should be vaccinated is simpler than in prior years: Rather than targeting people who are at higher risk (those ages 65 and older, or those with comorbidities), the current CDC recommendation is to vaccinate nearly everyone ages 6 months or older, with limited exceptions.6,7 (See Table 18).
Formulations. Many types of influenza vaccine are approved for use in the United States; these differ in the number of strains included (3 or 4), the amount of antigen present for each strain, the presence of an adjuvant, the growth medium used for the virus, and the route of administration (see Table 29). The relative merits of each type are a matter of some debate. There is ongoing research into the comparative efficacy of vaccines comprised of egg- vs cell-based cultures, as well as studies comparing high-dose or adjuvant vaccines to standard-dose inactivated vaccines.
Previously, the CDC has recommended preferential use (or avoidance) of some vaccine types, based on their efficacy. For the 2018-2019 flu season, however, the CDC has rescinded its recommendation against vaccine containing live attenuated virus (LAIV; FluMist brand) and expresses no preference for any vaccine formulation for patients of appropriate age and health status.10 The American Academy of Pediatrics (AAP), however, is recommends that LAIV be used only if patients and their families decline injectable vaccines.11
Timing. Influenza vaccines are now distributed as early as July to some locations, raising concerns about waning immunity from early vaccination (July/August) if the influenza season does not peak until February or March.8,12,13 Currently, the CDC recommends balancing the possible benefit of delayed vaccination against the risks of missed opportunities to vaccinate, a possible early season, and logistical problems related to vaccinating the same number of people in a smaller time interval. Offering vaccination by the end of October, if possible, is recommended in order for immunity to develop by mid-November.8 Note: Children ages 6 months to 8 years will need to receive their initial vaccination in 2 half-doses administered at least 28 days apart; completing their vaccination by the end of October would require starting the process weeks earlier.
[polldaddy:10124269]
Continue to: Strategy 2
Strategy 2: Make use of chemoprophylaxis
Preventive use of antiviral medication (chemoprophylaxis) may be a useful adjunct or alternative to vaccination in certain circumstances: if the patient is at high risk for complications, has been exposed to someone with influenza, has contraindications to vaccination, or received the vaccine within the past 2 weeks. The CDC also suggests that chemoprophylaxis be considered for those with immune deficiencies or who are otherwise immunosuppressed after exposure.14 Antivirals can also be used to control outbreaks in long-term care facilities; in these cases, the recommendedregimen is daily administration for at least 2 weeks, continuing until at least 7 days after the identification of the last case.14 Oseltamivir (Tamiflu) and zanamivir (Relenza) are the recommended prophylactic agents; a related intravenous medication, peramivir (Rapivab), is recommend for treatment only (see Table 314).
Strategy 3: Prevent comorbidities and opportunistic infections
Morbidity associated with influenza often comes from secondary infection. Pneumonia is among the most common complications, so influenza season is a good time to ensure that patients are appropriately vaccinated against pneumococcus, as well. Pneumococcal conjugate vaccine (Prevnar or PCV13) is recommended for children younger than 2 years of age, to be administered in a series of 4 doses: at 2, 4, 6, and 12-15 months. Vaccination with PCV13 is also recommended for those ages 65 or older, to be followed at least one year later with pneumococcal polysaccharide vaccine (Pneumovax or PPSV23).15 Additional doses of PCV13, PPSV23, or both may be indicated, depending on health status.
Strategy 4: Encourage good hygiene
The availability of immunizations and antivirals does not replace good hygiene. Frequent handwashing reduces the transmission of respiratory viruses, including influenza.16 Few studies have evaluated the use of alcohol-based hand sanitizers, but available evidence suggests they are effective in lowering viral transmission.16
Barriers, such as masks, gloves, and gowns, are helpful for health care workers.16 Surgical masks are often considered more comfortable to wear than N95 respirators. It may therefore be welcome news that when a 2009 randomized study assessed their use by hospital-based nurses, masks were non-inferior in protecting these health care workers against influenza.17
Presenteeism, the practice of going to work while sick, should be discouraged. People at risk for influenza may wish to avoid crowds during flu season; those with symptoms should be encouraged to stay home and limit contact with others.
Continue to: Treatment
Treatment
Strategy 1: Make prompt use of antivirals
Despite available preventive measures, tens of millions of people in the United States develop influenza every year. Use of antiviral medication, begun early in the course of illness, can reduce the duration of symptoms and may reduce the risk for complications.
The neuraminidase inhibitor (NI) group of antivirals—oseltamivir, zanamivir, and peramivir—is effective against influenza types A and B and current resistance rates are low.
The adamantine family of antivirals, amantadine and rimantadine, treat type A only. Since the circulating influenza strains in the past several seasons have demonstrated resistance >99%, these medications are not currently recommended.14
NIs reduce the duration of influenza symptoms by 10% to 20%, shortening the illness by 6 to 24 hours.18,19 In otherwise healthy patients, this benefit must be balanced against the increased risk for nausea and vomiting (oseltamivir), bronchospasm and sinusitis (zanamivir), and diarrhea (peramivir). In adults, NIs reduce the risk for lower respiratory tract complications and hospitalization. A 2015 meta-analysis by Dobson et al found a relative risk for hospitalization among those prescribed oseltamivir vs placebo of 37%.18
In the past, antivirals were used only in high-risk patients, such as children younger than 2 years, adults older than 65 years, and those with chronic health conditions.14 Now, antivirals are recommended for those who are at higher risk for complications (see Table 4), those with “severe, complicated, or progressive illness,” and hospitalized patients.14
Continue to: Antiviral treatment may have some value...
Antiviral treatment may have some value for hospitalized patients when started even 5 days after symptom onset. Treatment may be extended beyond the usual recommendations (5 days for oseltamivir or zanamivir) in immunosuppressed patients or the critically ill. Additionally, recent guidelines include consideration of antiviral treatment in outpatients who are at normal risk if treatment can be started within 48 hours of symptom onset.14
The CDC currently recommends use of oseltamivir rather than other antivirals for most hospitalized patients, based on the availability of data on its use in this setting.14 Intravenous peramivir is recommended for patients who cannot tolerate or absorb oral medication; inhaled zanamivir or IV peramivir are preferred for patients with end-stage renal disease who are not undergoing dialysis (see Table 3).14
Strategy 2: Exercise caution when it comes to supportive care
There are other medications that may offer symptom relief or prevent complications, especially when antivirals are contraindicated or unavailable.
Corticosteroids are recommended as part of the treatment of community-acquired pneumonia,20 but their role in influenza is controversial. A 2016 Cochrane review21 found no randomized controlled trials on the topic. Although the balance of available data from observational studies indicated that use of corticosteroids was associated with increased mortality, the authors also noted that all the studies included in their meta-analysis were of “very low quality.” They concluded that “the use of steroids in influenza remains a clinical judgement call.”
Statins may be associated with improved outcomes in influenza and pneumonia. Studies thus far have given contradictory results,22,23 and a planned Cochrane review of the question has been withdrawn.24
Continue to: Over-the-counter medications...
Over-the-counter medications, such as aspirin, acetaminophen, and ibuprofen are often used to manage the fever and myalgia associated with influenza. Patients should be cautioned against using the same ingredient in multiple different branded medications. Acetaminophen, for example, is not limited to Tylenol-branded products. To avoid Reye’s syndrome, children and teens with febrile illness, such as influenza, should not use aspirin.
CORRESPONDENCE
Jennifer L. Hamilton, MD, PhD, Drexel Family Medicine, 10 Shurs Lane, Suite 301, Philadelphia, PA 19127; [email protected].
1. CDC. Weekly US influenza surveillance report. https://www.cdc.gov/flu/weekly/index.htm. Published June 8, 2018. Accessed August 22, 2018.
2. CDC. Estimated influenza illnesses, medical visits, hospitalizations, and deaths averted by vaccination in the United States. Published April 19, 2017. https://www.cdc.gov/flu/about/disease/2015-16.htm. Accessed Setptember 18, 2018.
3. CDC. Seasonal influenza vaccine effectiveness, 2005-2018. https://www.cdc.gov/flu/professionals/vaccination/effectiveness-studies.htm. Published February 15, 2018. Accessed August 22, 2018.
4. Sah P, Medlock J, Fitzpatrick MC, et al. Optimizing the impact of low-efficacy influenza vaccines. Proc Natl Acad Sci. 2018:201802479.
5. Flannery B, Reynolds SB, Blanton L, et al. Influenza vaccine effectiveness against pediatric deaths: 2010-2014. Pediatrics. 2017;139: e20164244.
6. Kim DK, Riley LE, Hunter P. Advisory Committee on Immunization Practices recommended immunization schedule for adults aged 19 years or older—United States, 2018. MMWR Morb Mortal Wkly Rep. 2018;67:158–160.
7. Robinson CL, Romero JR, Kempe A, et al. Advisory Committee on Immunization Practices recommended immunization schedule for children and adolescents aged 18 years or younger—United States, 2018. MMWR Morb Mortal Wkly Rep. 2018;67:156–157.
8. Grohskopf LA, Sokolow LZ, Broder KR, et al. Prevention and control of seasonal influenza with vaccines: recommendations of the Advisory Committee on Immunization Practices—United States, 2017-18 influenza season. MMWR Recomm Rep. 2017;66:1-20.
9. CDC. Influenza vaccines—United States, 2017–18 influenza season. https://www.cdc.gov/flu/protect/vaccine/vaccines.htm. Published May 16, 2018. Accessed August 22, 2018.
10. Grohskopf LA, Sokolow LZ, Fry AM, et al. Update: ACIP recommendations for the use of quadrivalent live attenuated influenza vaccine (LAIV4)—United States, 2018-19 influenza season. MMWR Morb Mortal Wkly Rep. 2018;67:643–645.
11. Jenco M. AAP: Give children IIV flu shot; use LAIV as last resort. AAP News. May 21, 2018. http://www.aappublications.org/news/2018/05/21/fluvaccine051818. Accessed August 22, 2018.
12. Glinka ER, Smith DM, Johns ST. Timing matters—influenza vaccination to HIV-infected patients. HIV Med. 2016;17:601-604.
13. Castilla J, Martínez-Baz I, Martínez-Artola V, et al. Decline in influenza vaccine effectiveness with time after vaccination, Navarre, Spain, season 2011/12. Euro Surveill. 2013;18:20388.
14. CDC. Influenza antiviral medications: summary for clinicians. https://www.cdc.gov/flu/professionals/antivirals/summary-clinicians.htm. Published May 11, 2018. Accessed August 22, 2018.
15. CDC. Pneumococcal vaccination summary: who and when to vaccinate. https://www.cdc.gov/vaccines/vpd/pneumo/hcp/who-when-to-vaccinate.html. Published February 28, 2018. Accessed August 22, 2018.
16. Jefferson T, Del Mar CB, Dooley L, et al. Physical interventions to interrupt or reduce the spread of respiratory viruses. Cochrane Database Syst Rev. 2011;(7):CD006207.
17. Loeb M, Dafoe N, Mahony J, et al. Surgical mask vs N95 respirator for preventing influenza among health care workers: a randomized trial. JAMA. 2009;302:1865-1871.
18. Dobson J, Whitley RJ, Pocock S, Monto AS. Oseltamivir treatment for influenza in adults: a meta-analysis of randomised controlled trials. Lancet. 2015;385:1729-1737.
19. Ghebrehewet S, MacPherson P, Ho A. Influenza. BMJ. 2016;355:i6258.
20. Kaysin A, Viera AJ. Community-acquired pneumonia in adults: diagnosis and management. Am Fam Physician. 2016;94:698-706.
21. Rodrigo C, Leonardi‐Bee J, Nguyen‐Van‐Tam J, et al. Corticosteroids as adjunctive therapy in the treatment of influenza. Cochrane Database Syst Rev. 2016;3:CD010406.
22. Brassard P, Wu JW, Ernst P, et al. The effect of statins on influenza-like illness morbidity and mortality. Pharmacoepidemiol Drug Saf. 2017;26:63-70.
23. Fedson DS. Treating influenza with statins and other immunomodulatory agents. Antiviral Res. 2013;99:417-435.
24. Khandaker G, Rashid H, Chow MY, et al. Statins for influenza and pneumonia. Cochrane Database Syst Rev. January 9, 2017 [withdrawn].
Last year’s influenza season was severe enough that hospitals around the United States set up special evaluation areas beyond their emergency departments, at times spilling over to tents or other temporary structures in what otherwise would be parking lots. The scale and potential severity of the annual epidemic can be difficult to convey to our patients, who sometimes say “just the flu” to refer to an illness responsible for more than 170 pediatric deaths in the United States this past year.1 The Centers for Disease Control and Prevention (CDC) recently updated its 5-year estimates of influenza-related deaths in the United States; influenza mortality ranges from about 12,000 deaths in a mild season (such as 2011-2012) to 56,000 in a more severe season (eg, 2012-2013).2
Although influenza cannot be completely prevented, the following strategies can help reduce the risk for the illness and limit its severity if contracted.
Prevention
Strategy 1: Vaccinate against influenza
While the efficacy of vaccines varies from year to year, vaccination remains the core of influenza prevention efforts. In this decade, vaccine effectiveness has ranged from 19% to 60%.3 However, models suggest that even when the vaccine is only 20% effective, vaccinating 140 million people (the average number of doses delivered annually in the United States over the past 5 seasons) prevents 21 million infections, 130,000 hospitalizations, and more than 61,000 deaths.4 In a case-control study, Flannery et al found that vaccination was 65% effective in preventing laboratory-confirmed influenza-associated death in children over 4 seasons (July 2010 through June 2014).5
Deciding who should be vaccinated is simpler than in prior years: Rather than targeting people who are at higher risk (those ages 65 and older, or those with comorbidities), the current CDC recommendation is to vaccinate nearly everyone ages 6 months or older, with limited exceptions.6,7 (See Table 18).
Formulations. Many types of influenza vaccine are approved for use in the United States; these differ in the number of strains included (3 or 4), the amount of antigen present for each strain, the presence of an adjuvant, the growth medium used for the virus, and the route of administration (see Table 29). The relative merits of each type are a matter of some debate. There is ongoing research into the comparative efficacy of vaccines comprised of egg- vs cell-based cultures, as well as studies comparing high-dose or adjuvant vaccines to standard-dose inactivated vaccines.
Previously, the CDC has recommended preferential use (or avoidance) of some vaccine types, based on their efficacy. For the 2018-2019 flu season, however, the CDC has rescinded its recommendation against vaccine containing live attenuated virus (LAIV; FluMist brand) and expresses no preference for any vaccine formulation for patients of appropriate age and health status.10 The American Academy of Pediatrics (AAP), however, is recommends that LAIV be used only if patients and their families decline injectable vaccines.11
Timing. Influenza vaccines are now distributed as early as July to some locations, raising concerns about waning immunity from early vaccination (July/August) if the influenza season does not peak until February or March.8,12,13 Currently, the CDC recommends balancing the possible benefit of delayed vaccination against the risks of missed opportunities to vaccinate, a possible early season, and logistical problems related to vaccinating the same number of people in a smaller time interval. Offering vaccination by the end of October, if possible, is recommended in order for immunity to develop by mid-November.8 Note: Children ages 6 months to 8 years will need to receive their initial vaccination in 2 half-doses administered at least 28 days apart; completing their vaccination by the end of October would require starting the process weeks earlier.
[polldaddy:10124269]
Continue to: Strategy 2
Strategy 2: Make use of chemoprophylaxis
Preventive use of antiviral medication (chemoprophylaxis) may be a useful adjunct or alternative to vaccination in certain circumstances: if the patient is at high risk for complications, has been exposed to someone with influenza, has contraindications to vaccination, or received the vaccine within the past 2 weeks. The CDC also suggests that chemoprophylaxis be considered for those with immune deficiencies or who are otherwise immunosuppressed after exposure.14 Antivirals can also be used to control outbreaks in long-term care facilities; in these cases, the recommendedregimen is daily administration for at least 2 weeks, continuing until at least 7 days after the identification of the last case.14 Oseltamivir (Tamiflu) and zanamivir (Relenza) are the recommended prophylactic agents; a related intravenous medication, peramivir (Rapivab), is recommend for treatment only (see Table 314).
Strategy 3: Prevent comorbidities and opportunistic infections
Morbidity associated with influenza often comes from secondary infection. Pneumonia is among the most common complications, so influenza season is a good time to ensure that patients are appropriately vaccinated against pneumococcus, as well. Pneumococcal conjugate vaccine (Prevnar or PCV13) is recommended for children younger than 2 years of age, to be administered in a series of 4 doses: at 2, 4, 6, and 12-15 months. Vaccination with PCV13 is also recommended for those ages 65 or older, to be followed at least one year later with pneumococcal polysaccharide vaccine (Pneumovax or PPSV23).15 Additional doses of PCV13, PPSV23, or both may be indicated, depending on health status.
Strategy 4: Encourage good hygiene
The availability of immunizations and antivirals does not replace good hygiene. Frequent handwashing reduces the transmission of respiratory viruses, including influenza.16 Few studies have evaluated the use of alcohol-based hand sanitizers, but available evidence suggests they are effective in lowering viral transmission.16
Barriers, such as masks, gloves, and gowns, are helpful for health care workers.16 Surgical masks are often considered more comfortable to wear than N95 respirators. It may therefore be welcome news that when a 2009 randomized study assessed their use by hospital-based nurses, masks were non-inferior in protecting these health care workers against influenza.17
Presenteeism, the practice of going to work while sick, should be discouraged. People at risk for influenza may wish to avoid crowds during flu season; those with symptoms should be encouraged to stay home and limit contact with others.
Continue to: Treatment
Treatment
Strategy 1: Make prompt use of antivirals
Despite available preventive measures, tens of millions of people in the United States develop influenza every year. Use of antiviral medication, begun early in the course of illness, can reduce the duration of symptoms and may reduce the risk for complications.
The neuraminidase inhibitor (NI) group of antivirals—oseltamivir, zanamivir, and peramivir—is effective against influenza types A and B and current resistance rates are low.
The adamantine family of antivirals, amantadine and rimantadine, treat type A only. Since the circulating influenza strains in the past several seasons have demonstrated resistance >99%, these medications are not currently recommended.14
NIs reduce the duration of influenza symptoms by 10% to 20%, shortening the illness by 6 to 24 hours.18,19 In otherwise healthy patients, this benefit must be balanced against the increased risk for nausea and vomiting (oseltamivir), bronchospasm and sinusitis (zanamivir), and diarrhea (peramivir). In adults, NIs reduce the risk for lower respiratory tract complications and hospitalization. A 2015 meta-analysis by Dobson et al found a relative risk for hospitalization among those prescribed oseltamivir vs placebo of 37%.18
In the past, antivirals were used only in high-risk patients, such as children younger than 2 years, adults older than 65 years, and those with chronic health conditions.14 Now, antivirals are recommended for those who are at higher risk for complications (see Table 4), those with “severe, complicated, or progressive illness,” and hospitalized patients.14
Continue to: Antiviral treatment may have some value...
Antiviral treatment may have some value for hospitalized patients when started even 5 days after symptom onset. Treatment may be extended beyond the usual recommendations (5 days for oseltamivir or zanamivir) in immunosuppressed patients or the critically ill. Additionally, recent guidelines include consideration of antiviral treatment in outpatients who are at normal risk if treatment can be started within 48 hours of symptom onset.14
The CDC currently recommends use of oseltamivir rather than other antivirals for most hospitalized patients, based on the availability of data on its use in this setting.14 Intravenous peramivir is recommended for patients who cannot tolerate or absorb oral medication; inhaled zanamivir or IV peramivir are preferred for patients with end-stage renal disease who are not undergoing dialysis (see Table 3).14
Strategy 2: Exercise caution when it comes to supportive care
There are other medications that may offer symptom relief or prevent complications, especially when antivirals are contraindicated or unavailable.
Corticosteroids are recommended as part of the treatment of community-acquired pneumonia,20 but their role in influenza is controversial. A 2016 Cochrane review21 found no randomized controlled trials on the topic. Although the balance of available data from observational studies indicated that use of corticosteroids was associated with increased mortality, the authors also noted that all the studies included in their meta-analysis were of “very low quality.” They concluded that “the use of steroids in influenza remains a clinical judgement call.”
Statins may be associated with improved outcomes in influenza and pneumonia. Studies thus far have given contradictory results,22,23 and a planned Cochrane review of the question has been withdrawn.24
Continue to: Over-the-counter medications...
Over-the-counter medications, such as aspirin, acetaminophen, and ibuprofen are often used to manage the fever and myalgia associated with influenza. Patients should be cautioned against using the same ingredient in multiple different branded medications. Acetaminophen, for example, is not limited to Tylenol-branded products. To avoid Reye’s syndrome, children and teens with febrile illness, such as influenza, should not use aspirin.
CORRESPONDENCE
Jennifer L. Hamilton, MD, PhD, Drexel Family Medicine, 10 Shurs Lane, Suite 301, Philadelphia, PA 19127; [email protected].
Last year’s influenza season was severe enough that hospitals around the United States set up special evaluation areas beyond their emergency departments, at times spilling over to tents or other temporary structures in what otherwise would be parking lots. The scale and potential severity of the annual epidemic can be difficult to convey to our patients, who sometimes say “just the flu” to refer to an illness responsible for more than 170 pediatric deaths in the United States this past year.1 The Centers for Disease Control and Prevention (CDC) recently updated its 5-year estimates of influenza-related deaths in the United States; influenza mortality ranges from about 12,000 deaths in a mild season (such as 2011-2012) to 56,000 in a more severe season (eg, 2012-2013).2
Although influenza cannot be completely prevented, the following strategies can help reduce the risk for the illness and limit its severity if contracted.
Prevention
Strategy 1: Vaccinate against influenza
While the efficacy of vaccines varies from year to year, vaccination remains the core of influenza prevention efforts. In this decade, vaccine effectiveness has ranged from 19% to 60%.3 However, models suggest that even when the vaccine is only 20% effective, vaccinating 140 million people (the average number of doses delivered annually in the United States over the past 5 seasons) prevents 21 million infections, 130,000 hospitalizations, and more than 61,000 deaths.4 In a case-control study, Flannery et al found that vaccination was 65% effective in preventing laboratory-confirmed influenza-associated death in children over 4 seasons (July 2010 through June 2014).5
Deciding who should be vaccinated is simpler than in prior years: Rather than targeting people who are at higher risk (those ages 65 and older, or those with comorbidities), the current CDC recommendation is to vaccinate nearly everyone ages 6 months or older, with limited exceptions.6,7 (See Table 18).
Formulations. Many types of influenza vaccine are approved for use in the United States; these differ in the number of strains included (3 or 4), the amount of antigen present for each strain, the presence of an adjuvant, the growth medium used for the virus, and the route of administration (see Table 29). The relative merits of each type are a matter of some debate. There is ongoing research into the comparative efficacy of vaccines comprised of egg- vs cell-based cultures, as well as studies comparing high-dose or adjuvant vaccines to standard-dose inactivated vaccines.
Previously, the CDC has recommended preferential use (or avoidance) of some vaccine types, based on their efficacy. For the 2018-2019 flu season, however, the CDC has rescinded its recommendation against vaccine containing live attenuated virus (LAIV; FluMist brand) and expresses no preference for any vaccine formulation for patients of appropriate age and health status.10 The American Academy of Pediatrics (AAP), however, is recommends that LAIV be used only if patients and their families decline injectable vaccines.11
Timing. Influenza vaccines are now distributed as early as July to some locations, raising concerns about waning immunity from early vaccination (July/August) if the influenza season does not peak until February or March.8,12,13 Currently, the CDC recommends balancing the possible benefit of delayed vaccination against the risks of missed opportunities to vaccinate, a possible early season, and logistical problems related to vaccinating the same number of people in a smaller time interval. Offering vaccination by the end of October, if possible, is recommended in order for immunity to develop by mid-November.8 Note: Children ages 6 months to 8 years will need to receive their initial vaccination in 2 half-doses administered at least 28 days apart; completing their vaccination by the end of October would require starting the process weeks earlier.
[polldaddy:10124269]
Continue to: Strategy 2
Strategy 2: Make use of chemoprophylaxis
Preventive use of antiviral medication (chemoprophylaxis) may be a useful adjunct or alternative to vaccination in certain circumstances: if the patient is at high risk for complications, has been exposed to someone with influenza, has contraindications to vaccination, or received the vaccine within the past 2 weeks. The CDC also suggests that chemoprophylaxis be considered for those with immune deficiencies or who are otherwise immunosuppressed after exposure.14 Antivirals can also be used to control outbreaks in long-term care facilities; in these cases, the recommendedregimen is daily administration for at least 2 weeks, continuing until at least 7 days after the identification of the last case.14 Oseltamivir (Tamiflu) and zanamivir (Relenza) are the recommended prophylactic agents; a related intravenous medication, peramivir (Rapivab), is recommend for treatment only (see Table 314).
Strategy 3: Prevent comorbidities and opportunistic infections
Morbidity associated with influenza often comes from secondary infection. Pneumonia is among the most common complications, so influenza season is a good time to ensure that patients are appropriately vaccinated against pneumococcus, as well. Pneumococcal conjugate vaccine (Prevnar or PCV13) is recommended for children younger than 2 years of age, to be administered in a series of 4 doses: at 2, 4, 6, and 12-15 months. Vaccination with PCV13 is also recommended for those ages 65 or older, to be followed at least one year later with pneumococcal polysaccharide vaccine (Pneumovax or PPSV23).15 Additional doses of PCV13, PPSV23, or both may be indicated, depending on health status.
Strategy 4: Encourage good hygiene
The availability of immunizations and antivirals does not replace good hygiene. Frequent handwashing reduces the transmission of respiratory viruses, including influenza.16 Few studies have evaluated the use of alcohol-based hand sanitizers, but available evidence suggests they are effective in lowering viral transmission.16
Barriers, such as masks, gloves, and gowns, are helpful for health care workers.16 Surgical masks are often considered more comfortable to wear than N95 respirators. It may therefore be welcome news that when a 2009 randomized study assessed their use by hospital-based nurses, masks were non-inferior in protecting these health care workers against influenza.17
Presenteeism, the practice of going to work while sick, should be discouraged. People at risk for influenza may wish to avoid crowds during flu season; those with symptoms should be encouraged to stay home and limit contact with others.
Continue to: Treatment
Treatment
Strategy 1: Make prompt use of antivirals
Despite available preventive measures, tens of millions of people in the United States develop influenza every year. Use of antiviral medication, begun early in the course of illness, can reduce the duration of symptoms and may reduce the risk for complications.
The neuraminidase inhibitor (NI) group of antivirals—oseltamivir, zanamivir, and peramivir—is effective against influenza types A and B and current resistance rates are low.
The adamantine family of antivirals, amantadine and rimantadine, treat type A only. Since the circulating influenza strains in the past several seasons have demonstrated resistance >99%, these medications are not currently recommended.14
NIs reduce the duration of influenza symptoms by 10% to 20%, shortening the illness by 6 to 24 hours.18,19 In otherwise healthy patients, this benefit must be balanced against the increased risk for nausea and vomiting (oseltamivir), bronchospasm and sinusitis (zanamivir), and diarrhea (peramivir). In adults, NIs reduce the risk for lower respiratory tract complications and hospitalization. A 2015 meta-analysis by Dobson et al found a relative risk for hospitalization among those prescribed oseltamivir vs placebo of 37%.18
In the past, antivirals were used only in high-risk patients, such as children younger than 2 years, adults older than 65 years, and those with chronic health conditions.14 Now, antivirals are recommended for those who are at higher risk for complications (see Table 4), those with “severe, complicated, or progressive illness,” and hospitalized patients.14
Continue to: Antiviral treatment may have some value...
Antiviral treatment may have some value for hospitalized patients when started even 5 days after symptom onset. Treatment may be extended beyond the usual recommendations (5 days for oseltamivir or zanamivir) in immunosuppressed patients or the critically ill. Additionally, recent guidelines include consideration of antiviral treatment in outpatients who are at normal risk if treatment can be started within 48 hours of symptom onset.14
The CDC currently recommends use of oseltamivir rather than other antivirals for most hospitalized patients, based on the availability of data on its use in this setting.14 Intravenous peramivir is recommended for patients who cannot tolerate or absorb oral medication; inhaled zanamivir or IV peramivir are preferred for patients with end-stage renal disease who are not undergoing dialysis (see Table 3).14
Strategy 2: Exercise caution when it comes to supportive care
There are other medications that may offer symptom relief or prevent complications, especially when antivirals are contraindicated or unavailable.
Corticosteroids are recommended as part of the treatment of community-acquired pneumonia,20 but their role in influenza is controversial. A 2016 Cochrane review21 found no randomized controlled trials on the topic. Although the balance of available data from observational studies indicated that use of corticosteroids was associated with increased mortality, the authors also noted that all the studies included in their meta-analysis were of “very low quality.” They concluded that “the use of steroids in influenza remains a clinical judgement call.”
Statins may be associated with improved outcomes in influenza and pneumonia. Studies thus far have given contradictory results,22,23 and a planned Cochrane review of the question has been withdrawn.24
Continue to: Over-the-counter medications...
Over-the-counter medications, such as aspirin, acetaminophen, and ibuprofen are often used to manage the fever and myalgia associated with influenza. Patients should be cautioned against using the same ingredient in multiple different branded medications. Acetaminophen, for example, is not limited to Tylenol-branded products. To avoid Reye’s syndrome, children and teens with febrile illness, such as influenza, should not use aspirin.
CORRESPONDENCE
Jennifer L. Hamilton, MD, PhD, Drexel Family Medicine, 10 Shurs Lane, Suite 301, Philadelphia, PA 19127; [email protected].
1. CDC. Weekly US influenza surveillance report. https://www.cdc.gov/flu/weekly/index.htm. Published June 8, 2018. Accessed August 22, 2018.
2. CDC. Estimated influenza illnesses, medical visits, hospitalizations, and deaths averted by vaccination in the United States. Published April 19, 2017. https://www.cdc.gov/flu/about/disease/2015-16.htm. Accessed Setptember 18, 2018.
3. CDC. Seasonal influenza vaccine effectiveness, 2005-2018. https://www.cdc.gov/flu/professionals/vaccination/effectiveness-studies.htm. Published February 15, 2018. Accessed August 22, 2018.
4. Sah P, Medlock J, Fitzpatrick MC, et al. Optimizing the impact of low-efficacy influenza vaccines. Proc Natl Acad Sci. 2018:201802479.
5. Flannery B, Reynolds SB, Blanton L, et al. Influenza vaccine effectiveness against pediatric deaths: 2010-2014. Pediatrics. 2017;139: e20164244.
6. Kim DK, Riley LE, Hunter P. Advisory Committee on Immunization Practices recommended immunization schedule for adults aged 19 years or older—United States, 2018. MMWR Morb Mortal Wkly Rep. 2018;67:158–160.
7. Robinson CL, Romero JR, Kempe A, et al. Advisory Committee on Immunization Practices recommended immunization schedule for children and adolescents aged 18 years or younger—United States, 2018. MMWR Morb Mortal Wkly Rep. 2018;67:156–157.
8. Grohskopf LA, Sokolow LZ, Broder KR, et al. Prevention and control of seasonal influenza with vaccines: recommendations of the Advisory Committee on Immunization Practices—United States, 2017-18 influenza season. MMWR Recomm Rep. 2017;66:1-20.
9. CDC. Influenza vaccines—United States, 2017–18 influenza season. https://www.cdc.gov/flu/protect/vaccine/vaccines.htm. Published May 16, 2018. Accessed August 22, 2018.
10. Grohskopf LA, Sokolow LZ, Fry AM, et al. Update: ACIP recommendations for the use of quadrivalent live attenuated influenza vaccine (LAIV4)—United States, 2018-19 influenza season. MMWR Morb Mortal Wkly Rep. 2018;67:643–645.
11. Jenco M. AAP: Give children IIV flu shot; use LAIV as last resort. AAP News. May 21, 2018. http://www.aappublications.org/news/2018/05/21/fluvaccine051818. Accessed August 22, 2018.
12. Glinka ER, Smith DM, Johns ST. Timing matters—influenza vaccination to HIV-infected patients. HIV Med. 2016;17:601-604.
13. Castilla J, Martínez-Baz I, Martínez-Artola V, et al. Decline in influenza vaccine effectiveness with time after vaccination, Navarre, Spain, season 2011/12. Euro Surveill. 2013;18:20388.
14. CDC. Influenza antiviral medications: summary for clinicians. https://www.cdc.gov/flu/professionals/antivirals/summary-clinicians.htm. Published May 11, 2018. Accessed August 22, 2018.
15. CDC. Pneumococcal vaccination summary: who and when to vaccinate. https://www.cdc.gov/vaccines/vpd/pneumo/hcp/who-when-to-vaccinate.html. Published February 28, 2018. Accessed August 22, 2018.
16. Jefferson T, Del Mar CB, Dooley L, et al. Physical interventions to interrupt or reduce the spread of respiratory viruses. Cochrane Database Syst Rev. 2011;(7):CD006207.
17. Loeb M, Dafoe N, Mahony J, et al. Surgical mask vs N95 respirator for preventing influenza among health care workers: a randomized trial. JAMA. 2009;302:1865-1871.
18. Dobson J, Whitley RJ, Pocock S, Monto AS. Oseltamivir treatment for influenza in adults: a meta-analysis of randomised controlled trials. Lancet. 2015;385:1729-1737.
19. Ghebrehewet S, MacPherson P, Ho A. Influenza. BMJ. 2016;355:i6258.
20. Kaysin A, Viera AJ. Community-acquired pneumonia in adults: diagnosis and management. Am Fam Physician. 2016;94:698-706.
21. Rodrigo C, Leonardi‐Bee J, Nguyen‐Van‐Tam J, et al. Corticosteroids as adjunctive therapy in the treatment of influenza. Cochrane Database Syst Rev. 2016;3:CD010406.
22. Brassard P, Wu JW, Ernst P, et al. The effect of statins on influenza-like illness morbidity and mortality. Pharmacoepidemiol Drug Saf. 2017;26:63-70.
23. Fedson DS. Treating influenza with statins and other immunomodulatory agents. Antiviral Res. 2013;99:417-435.
24. Khandaker G, Rashid H, Chow MY, et al. Statins for influenza and pneumonia. Cochrane Database Syst Rev. January 9, 2017 [withdrawn].
1. CDC. Weekly US influenza surveillance report. https://www.cdc.gov/flu/weekly/index.htm. Published June 8, 2018. Accessed August 22, 2018.
2. CDC. Estimated influenza illnesses, medical visits, hospitalizations, and deaths averted by vaccination in the United States. Published April 19, 2017. https://www.cdc.gov/flu/about/disease/2015-16.htm. Accessed Setptember 18, 2018.
3. CDC. Seasonal influenza vaccine effectiveness, 2005-2018. https://www.cdc.gov/flu/professionals/vaccination/effectiveness-studies.htm. Published February 15, 2018. Accessed August 22, 2018.
4. Sah P, Medlock J, Fitzpatrick MC, et al. Optimizing the impact of low-efficacy influenza vaccines. Proc Natl Acad Sci. 2018:201802479.
5. Flannery B, Reynolds SB, Blanton L, et al. Influenza vaccine effectiveness against pediatric deaths: 2010-2014. Pediatrics. 2017;139: e20164244.
6. Kim DK, Riley LE, Hunter P. Advisory Committee on Immunization Practices recommended immunization schedule for adults aged 19 years or older—United States, 2018. MMWR Morb Mortal Wkly Rep. 2018;67:158–160.
7. Robinson CL, Romero JR, Kempe A, et al. Advisory Committee on Immunization Practices recommended immunization schedule for children and adolescents aged 18 years or younger—United States, 2018. MMWR Morb Mortal Wkly Rep. 2018;67:156–157.
8. Grohskopf LA, Sokolow LZ, Broder KR, et al. Prevention and control of seasonal influenza with vaccines: recommendations of the Advisory Committee on Immunization Practices—United States, 2017-18 influenza season. MMWR Recomm Rep. 2017;66:1-20.
9. CDC. Influenza vaccines—United States, 2017–18 influenza season. https://www.cdc.gov/flu/protect/vaccine/vaccines.htm. Published May 16, 2018. Accessed August 22, 2018.
10. Grohskopf LA, Sokolow LZ, Fry AM, et al. Update: ACIP recommendations for the use of quadrivalent live attenuated influenza vaccine (LAIV4)—United States, 2018-19 influenza season. MMWR Morb Mortal Wkly Rep. 2018;67:643–645.
11. Jenco M. AAP: Give children IIV flu shot; use LAIV as last resort. AAP News. May 21, 2018. http://www.aappublications.org/news/2018/05/21/fluvaccine051818. Accessed August 22, 2018.
12. Glinka ER, Smith DM, Johns ST. Timing matters—influenza vaccination to HIV-infected patients. HIV Med. 2016;17:601-604.
13. Castilla J, Martínez-Baz I, Martínez-Artola V, et al. Decline in influenza vaccine effectiveness with time after vaccination, Navarre, Spain, season 2011/12. Euro Surveill. 2013;18:20388.
14. CDC. Influenza antiviral medications: summary for clinicians. https://www.cdc.gov/flu/professionals/antivirals/summary-clinicians.htm. Published May 11, 2018. Accessed August 22, 2018.
15. CDC. Pneumococcal vaccination summary: who and when to vaccinate. https://www.cdc.gov/vaccines/vpd/pneumo/hcp/who-when-to-vaccinate.html. Published February 28, 2018. Accessed August 22, 2018.
16. Jefferson T, Del Mar CB, Dooley L, et al. Physical interventions to interrupt or reduce the spread of respiratory viruses. Cochrane Database Syst Rev. 2011;(7):CD006207.
17. Loeb M, Dafoe N, Mahony J, et al. Surgical mask vs N95 respirator for preventing influenza among health care workers: a randomized trial. JAMA. 2009;302:1865-1871.
18. Dobson J, Whitley RJ, Pocock S, Monto AS. Oseltamivir treatment for influenza in adults: a meta-analysis of randomised controlled trials. Lancet. 2015;385:1729-1737.
19. Ghebrehewet S, MacPherson P, Ho A. Influenza. BMJ. 2016;355:i6258.
20. Kaysin A, Viera AJ. Community-acquired pneumonia in adults: diagnosis and management. Am Fam Physician. 2016;94:698-706.
21. Rodrigo C, Leonardi‐Bee J, Nguyen‐Van‐Tam J, et al. Corticosteroids as adjunctive therapy in the treatment of influenza. Cochrane Database Syst Rev. 2016;3:CD010406.
22. Brassard P, Wu JW, Ernst P, et al. The effect of statins on influenza-like illness morbidity and mortality. Pharmacoepidemiol Drug Saf. 2017;26:63-70.
23. Fedson DS. Treating influenza with statins and other immunomodulatory agents. Antiviral Res. 2013;99:417-435.
24. Khandaker G, Rashid H, Chow MY, et al. Statins for influenza and pneumonia. Cochrane Database Syst Rev. January 9, 2017 [withdrawn].
PRACTICE RECOMMENDATIONS
› Recommend influenza vaccination for all patients at least 6 months old unless a specific contraindication exists. A
› Recommend pneumococcal vaccination to appropriate patients to reduce the risk for a common complication of influenza. A
› Encourage hygiene-based measures to limit infection, including frequent handwashing or use of a hand sanitizer. B
› Prescribe oseltamivir to hospitalized influenza patients to limit the duration and severity of infection. B
Strength of recommendation (SOR)
A Good-quality patient-oriented evidence
B Inconsistent or limited-quality patient-oriented evidence
C Consensus, usual practice, opinion, disease-oriented evidence, case series
Small Cell Lung Cancer
INTRODUCTION
Small cell lung cancer (SCLC) is an aggressive cancer of neuroendocrine origin that accounts for approximately 15% of all lung cancer cases, with approximately 33,000 patients diagnosed annually.1 The incidence of SCLC in the United States has steadily declined over the past 30 years, presumably because of a decrease in the number of smokers and a change to low-tar filter cigarettes.2 Although the overall incidence of SCLC has been decreasing, the incidence in women is increasing and the male-to-female incidence ratio is now 1:1.3 Nearly all cases of SCLC are associated with heavy tobacco exposure, making it a heterogeneous disease with a complex genomic landscape consisting of thousands of mutations.4,5 Despite recent advances in the treatment of non-small cell lung cancer, the therapeutic options for SCLC remain limited, with a median overall survival (OS) of 9 months in patients with advanced disease.
DIAGNOSIS AND STAGING
CASE PRESENTATION
A 61-year-old man presents to the emergency department with progressive shortness of breath and cough over the past 6 weeks. He also reports a 20-lb weight loss over the same period. He is a current smoker and has been smoking 1 pack of cigarettes per day since the age of 18 years. A chest radiograph obtained in the emergency department shows a right hilar mass. Computed tomography (CT) scan confirms the presence of a 4.5-cm right hilar mass and enlarged mediastinal lymph nodes bilaterally.
• What are the next steps in diagnosis?
SCLC is characterized by rapid growth and early hematogenous metastasis. Consequently, only 25% of patients have limited-stage disease at the time of diagnosis. According to the Veterans Administration Lung Study Group (VALSG) staging system, limited-stage disease is defined as tumor that is confined to 1 hemithorax and can be encompassed within 1 radiation field. This typically includes mediastinal lymph nodes and ipsilateral supraclavicular lymph nodes. Approximately 75% of patients present with extensive-stage disease, which is defined as disease that cannot be classified as limited, including disease that extends beyond 1 hemithorax. Extensive-stage disease includes the presence of malignant pleural effusion and/or distant metastasis.6 The VALSG classification and staging system is more commonly used in clinical practice than the American Joint Committee on Cancer TNM staging system because it is less complex and directs treatment decisions, as most of the literature on SCLC classifies patients based on the VALSG system.7
Given SCLC’s propensity to metastasize quickly, none of the currently available screening methods have proven successful in early detection of SCLC. In the National Lung Cancer Screening Trial, 86% of the 125 patients who were diagnosed with SCLC while undergoing annual low-dose chest CT scans had advanced disease at diagnosis.8,9 These results highlight the fact that most cases of SCLC develop in the interval between annual screening imaging.
SCLC frequently presents with a large hilar mass that is symptomatic. Common symptoms include shortness of breath and cough. In addition, patients with SCLC usually have bulky mediastinal adenopathy at presentation. SCLC is commonly located submucosally in the bronchus, and therefore hemoptysis is not a very common symptom at the time of presentation. Patients may present with superior vena cava syndrome from local compression by the tumor. Not infrequently, SCLC is associated with paraneoplastic syndromes that arise due to ectopic secretion of hormones or antibodies by the tumor cells. The paraneoplastic syndromes can be broadly categorized as endocrine or neurologic (Table 1). The presence of a paraneoplastic syndrome is often a clue to the potential diagnosis of SCLC in the presence of a hilar mass. Additionally, some paraneoplastic syndromes, more specifically endocrine paraneoplastic syndromes, follow the pattern of disease response and relapse, and therefore can sometimes serve as an early marker of disease relapse or progression.
The common sites of metastases include brain, liver, and bone. Therefore, the staging workup should include fluorodeoxyglucose (FDG) positron emission tomography (PET)/CT scan. Contrast-enhanced CT scan of the chest and abdomen and bone scan can be obtained for staging in lieu of PET scan. Due to the physiologic FDG uptake, cerebral metastases cannot be assessed with sufficient certainty using PET-CT.10 Therefore, brain imaging with contrast-enhanced CT or magnetic resonance imaging (MRI) is also necessary. Although the incidence of metastasis to bone marrow is less than 10%, bone marrow aspiration and biopsy are warranted in patients with unexplained cytopenias, especially when the cytopenia is associated with teardrop-shaped red cells or nucleated red cells on peripheral blood smear, findings indicative of a marrow infiltrative process.7 The tissue diagnosis is established by obtaining a biopsy of the primary tumor or 1 of the metastatic sites. In localized disease, bronchoscopy (with endobronchial ultrasound, if necessary) with biopsy of the centrally located tumor and/or lymph node is required. Histologically, SCLC consists of monomorphic cells, a high nuclear-cytoplasmic ratio, and confluent necrosis. The tumor cells are positive for chromogranin, synaptophysin, and CD56 by immunohistochemistry, and very frequently are also positive for thyroid transcription factor 1.11 Although serum tumor markers, including neuron-specific enolase and progastrin-releasing peptide, are frequently elevated in patients with SCLC, these markers are of limited value in clinical practice because they lack sensitivity and specificity.12
MANAGEMENT OF LIMITED-STAGE DISEASE
CASE CONTINUED
The patient undergoes FDG PET scan, which shows the presence of a hypermetabolic right hilar mass in addition to enlarged and hypermetabolic bilateral mediastinal lymph nodes. There are no other areas of FDG avidity. Brain MRI does not show any evidence of brain metastasis. Thus, the patient is confirmed to have limited-stage SCLC.
• What is the standard of care treatment for limited-stage SCLC?
SCLC is exquisitely sensitive to both chemotherapy and radiation, especially at the time of initial presentation. The standard of care treatment of limited-stage SCLC is 4 cycles of platinum-based chemotherapy in combination with thoracic radiation started within the first 2 cycles of chemotherapy (Figure 1).
CHOICE OF CHEMOTHERAPY
Etoposide and cisplatin is the most commonly used initial combination chemotherapy regimen in limited-stage SCLC.14 This combination has largely replaced anthracycline-based regimens given its favorable efficacy and toxicity profile.15–17 Several small randomized trials have shown comparable efficacy of carboplatin and etoposide in extensive-stage SCLC.18–20 A meta-analysis of 4 randomized trials comparing cisplatin-based versus carboplatin-based regimens in 663 patients with SCLC (32% had limited-stage disease and 68% had extensive-stage disease) showed no statistically significant difference in response rate, progression-free survival (PFS), or OS between the 2 regimens.21 Therefore, in clinical practice carboplatin is frequently used instead of cisplatin in patients with extensive-stage disease. In patients with limited-stage disease, cisplatin is still the drug of choice. However, the toxicity profile of the 2 regimens is different. Cisplatin-based regimens are more commonly associated with neuropathy, nephrotoxicity, and chemotherapy-induced nausea/vomiting,18 while carboplatin-based regimens are more myelosuppressive.22 In addition, the combination of thoracic radiation with either of these regimens is associated with a higher risk of esophagitis, pneumonitis, and myelosuppression.23 The use of myeloid growth factors is not recommended in patients undergoing concurrent chemoradiation.24 Of note, intravenous etoposide is always preferred over oral etoposide, especially in the curative setting given the unreliable absorption and bioavailability of oral formulations.
THORACIC RADIOTHERAPY
Adding thoracic radiotherapy to platinum-etoposide chemotherapy improves local control and OS. Two meta-analyses of 13 trials including more than 2000 patients have shown a 25% to 30% decrease in local failure and a 5% to 7% increase in 2-year OS with chemoradiation compared to chemotherapy alone in limited-stage SCLC.25,26 Early (within the first 2 cycles) concurrent thoracic radiation is superior to delayed and/or sequential radiation in terms of local control and OS.23,27,28 The dose and fractionation of thoracic radiation in limited-stage SCLC has remained a controversial issue. The Eastern Cooperative Oncology Group/Radiation Therapy Oncology Group randomized trial compared 45 Gy of radiotherapy delivered twice daily over a period of 3 weeks to 45 Gy once daily over 5 weeks concurrently with chemotherapy. The twice daily regimen led to a 10% improvement in 5-year OS (26% versus 16%), but a higher incidence of grade 3 and 4 adverse events.13 Despite the survival advantage demonstrated by hyperfractionated radiotherapy, the results need to be interpreted with caution because the radiation doses are not biologically equivalent. In addition, the difficult logistics of patients receiving radiation twice a day has limited the routine implementation of this strategy. Subsequently, another randomized phase 3 trial (CONVERT) compared 45 Gy radiotherapy twice daily with 66 Gy radiotherapy once daily in limited-stage SCLC.29 This trial did not show any difference in OS. The patients in the twice daily arm had a higher incidence of grade 4 neutropenia. Considering the results of these trials, both strategies—45 Gy fractionated twice daily or 60 Gy fractionated once daily, delivered concurrently with chemotherapy—are acceptable in the setting of limited-stage SCLC. However, quite often a hyperfractionated regimen is not feasible for patients and many radiation oncology centers. Hopefully, the ongoing CALGB 30610 study will clarify the optimal radiation schedule for limited-stage disease.
PROPHYLACTIC CRANIAL IRRADIATION
Approximately 75% of patients with limited-stage disease experience disease recurrence, and brain is the site of recurrence in approximately half of these patients.30 Prophylactic cranial irradiation (PCI) consisting of 25 Gy radiotherapy delivered in 10 fractions has been shown to be effective in decreasing the incidence of cerebral metastases.30–32 Although individual small studies have not shown a survival benefit of PCI because of small sample size and limited power, a meta-analysis of these studies has shown a 25% decrease in the 3-year incidence of brain metastasis and 5.4% increase in 3-year OS.30 Most patients included in these studies had limited-stage disease. Therefore, PCI is the standard of care for patients with limited-stage disease who attain a partial or complete response to chemoradiation.
ROLE OF SURGERY
Surgical resection may be an acceptable choice in a very limited subset of patients with peripherally located small (< 5 cm) tumors where mediastinal lymph nodes have been confirmed to be uninvolved with complete mediastinal staging.33,34 Most of the data in this setting are derived from retrospective studies.35,36 A 5-year OS between 40% and 60% has been reported with this strategy in patients with clinical stage I disease. In general, when surgery is considered, lobectomy with mediastinal lymph node dissection followed by chemotherapy (if there is no nodal involvement) or chemoradiation (if nodal involvement) is recommended.37,38 Wedge or segmental resections are not considered to be optimal surgical options.
MANAGEMENT OF EXTENSIVE-STAGE DISEASE
CASE CONTINUED
The patient receives 4 cycles of cisplatin and etoposide along with 70 Gy radiotherapy concurrently with the first 2 cycles of chemotherapy. His post-treatment CT scans show a partial response. He undergoes PCI 6 weeks after completion of treatment. At routine follow-up 18 months later, he is doing generally well except for mildly decreased appetite and an unintentional weight loss of 5 lb. CT scans demonstrate multiple hypodense liver lesions ranging from 7 mm to 2 cm in size and a 2-cm left adrenal gland lesion highly concerning for metastasis. FDG PET scan confirms that the adrenal and liver lesions are hypermetabolic. In addition, the PET scan shows multiple FDG-avid bone lesions throughout the spine. Brain MRI is negative for brain metastasis.
• What is the standard of care for treatment of extensive-stage disease?
Chemotherapy is the mainstay of treatment for extensive-stage SCLC; the goals of treatment are prolongation of survival, prevention or alleviation of cancer-related symptoms, and improvement in quality of life. The combination of etoposide with a platinum agent (carboplatin or cisplatin) is the preferred first-line treatment option. Carboplatin is more commonly used in clinical practice in this setting because of its comparable efficacy and better tolerability compared to cisplatin (Figure 2).21 A Japanese phase 3 trial comparing cisplatin plus irinotecan with cisplatin plus etoposide in the first-line setting in extensive-stage SCLC showed improvement in median and 2-year OS with the cisplatin/irinotecan regimen; however, 2 subsequent phase 3 trials conducted in the United States comparing these 2 regimens did not show any difference in OS. In addition, the cisplatin/irinotecan regimen was more toxic than the etoposide-based regimen.39,40 Therefore, 4 to 6 cycles of platinum/etoposide remains the standard of care first-line treatment for extensive-stage SCLC in the United States. The combination yields a 60% to 70% response rate, but the majority of patients invariably experience disease progression, with a median OS of 9 to 11 months.41 Maintenance chemotherapy beyond the initial 4 to 6 cycles does not improve survival and is associated with higher cumulative toxicity.42
Multiple attempts at improving first-line chemotherapy in extensive-stage disease have failed to show any meaningful difference in OS. For example, the addition of ifosfamide, palifosfamide, cyclophosphamide, taxane, or anthracycline to platinum doublet failed to show improvement in OS and led to more toxicity.43–46 Additionally, the use of alternating or cyclic chemotherapies in an attempt to curb drug resistance has also failed to show survival benefit.47–49 The addition of the antiangiogenic agent bevacizumab to standard platinum-based doublet has not prolonged OS in SCLC and has led to an unacceptably higher rate of tracheoesophageal fistula when used in conjunction with chemoradiation in limited-stage disease.50–55 Finally, the immune checkpoint inhibitor ipilimumab in combination with platinum plus etoposide failed to improve PFS or OS compared to platinum plus etoposide alone in a recent phase 3 trial, and maintenance pembrolizumab after completion of platinum-based chemotherapy did not improve PFS.56,57
More recently, a phase 2 study of pembrolizumab in extensive-stage SCLC (KEYNOTE 158) reported an overall response rate of 35.7%, median PFS of 2.1 months, and median OS of 14.6 months in patients who tested positive for programmed death ligand-1 (PD-L1) expression (which was defined as a PD-L1 Combined Positive Score ≥ 1).58 The median duration of response has not been reached in this study, indicating that pembrolizumab may be a promising approach in patients with extensive-stage SCLC, especially for those with PD-L1–positive tumors.
Patients with extensive-stage disease who have brain metastasis at the time of diagnosis can be treated with systemic chemotherapy first if the brain metastases are asymptomatic and there is significant extracranial disease burden. In that case, whole brain radiotherapy should be given after completion of systemic therapy.
SECOND-LINE CHEMOTHERAPY
Despite being exquisitely chemosensitive, SCLC is associated with a very poor prognosis largely because of invariable disease progression following first-line therapy and lack of effective second-line treatment options that can lead to appreciable disease control. The choice of second-line treatment is predominantly determined by the time of disease relapse after first-line platinum-based therapy. If this interval is 6 months or longer, re-treatment utilizing the same platinum doublet is appropriate. However, if the interval is 6 months or less, second-line systemic therapy options should be explored. Unfortunately, the response rate tends to be less than 10% with most of the second-line therapies in platinum-resistant disease (defined as disease progression within 3 months of receiving platinum-based therapy). If disease progression occurs between 3 and 6 months after completion of platinum-based therapy, the response rate with second-line chemotherapy is in the range of 25%.59,60
A number of second-line chemotherapy options have been explored in small studies, including topotecan, irinotecan, paclitaxel, docetaxel, temozolomide, vinorelbine, oral etoposide, gemcitabine, bendamustine, and CAV (
IMMUNOTHERAPY
The role of immune checkpoint inhibitors in the treatment of SCLC is evolving, and currently there are no FDA-approved immunotherapy agents for treating SCLC. A recently conducted phase 1/2 trial (CheckMate 032) studied the anti-programmed death(PD)-1 antibody nivolumab with or without the anti-cytotoxic T-lymphocyte–associated antigen (CTLA) -4 antibody ipilimumab in patients with relapsed SCLC. The authors reported response rates of 10% with nivolumab 3 mg/kg and 21% with nivolumab 1 mg/kg plus ipilimumab 3 mg/kg.78,79 The 2-year OS was 26% with the combination and 14% with single-agent nivolumab. Only 18% of patients had PD-L1 expression of ≥ 1%, and the response rate did not correlate with PD-L1 status. The rate of grade 3 or 4 adverse events was approximately 20%, and only 10% of patients discontinued treatment because of toxicity. Based on these data, nivolumab plus ipilimumab is now included in the National Comprehensive Cancer Network guidelines as an option for patients with SCLC who experience disease relapse within 6 months of receiving platinum-based therapy;7 however, it is questionable whether routine use of this combination is justified based on currently available data. The evidence for the combination of nivolumab and ipilimumab remains limited. The efficacy and toxicity data from both randomized and nonrandomized cohorts were presented together, making it hard to interpret the results.
Another phase 1b study (KEYNOTE-028) evaluated the anti-PD-1 antibody pembrolizumab (10 mg/kg intravenously every 2 weeks) in patients with relapsed SCLC who had received 1 or more prior lines of therapy and had PD-L1 expression of ≥ 1%. This study showed a response rate of 33%, with a median duration of response of 19 months and 1-year OS of 38%.80 Although only 28% of screened patients had PD-L1 expression of ≥ 1%, these results indicated that at least a subset of SCLC patients are able to achieve durable responses with immune checkpoint inhibition. A number of clinical trials utilizing immune checkpoint inhibitors in various combinations and settings are currently underway.
ROLE OF PROPHYLACTIC CRANIAL IRRADIATION
The role of PCI in extensive-stage SCLC is not clearly defined. A randomized phase 3 trial conducted by the European Organization for Research and Treatment of Cancer (EORTC) comparing PCI with no PCI in patients with extensive-stage SCLC who had a partial or complete response to initial platinum-based chemotherapy showed a decrease in the incidence of symptomatic brain metastasis and improvement in 1-year OS with PCI.81 However, this trial did not require mandatory brain imaging prior to PCI, and thus it is unclear if some patients in the PCI group had asymptomatic brain metastasis prior to enrollment and therefore received therapeutic benefit from brain radiation. Additionally, the dose and fractionation of PCI was not standardized across patient groups.
A more recent phase 3 study conducted in Japan that compared PCI (25 Gy in 10 fractions) with no PCI reported no difference in survival between the 2 groups.82 As opposed to the EORTC study, the Japanese study did require baseline brain imaging to confirm the absence of brain metastasis prior to enrollment. In addition, the control patients underwent periodic brain MRI to allow early detection of brain metastasis. Given the emergence of the new data, the impact of PCI on survival in patients with extensive-stage SCLC is unproven, and PCI likely has a role in a highly selected small group of patients with extensive-stage SCLC. PCI is not recommended for patients with poor performance status (ECOG performance score of 3 or 4) or underlying neurocognitive disorders.34,83
The NMDA-receptor antagonist memantine can be used in patients undergoing PCI to delay the occurrence of cognitive dysfunction.61 Memantine 20 mg daily delayed time to cognitive decline and reduced the rate of decline in memory, executive function, and processing speed compared to placebo in patients receiving whole brain radiotherapy.84
ROLE OF RADIOTHERAPY
A subset of patients with extensive-stage SCLC may benefit from consolidative thoracic radiotherapy after completion of platinum-based chemotherapy. A randomized trial that enrolled patients who achieved complete or near complete response after 3 cycles of cisplatin plus etoposide compared thoracic radiotherapy in combination with continued chemotherapy versus chemotherapy alone.85 The median OS was longer with the addition of thoracic radiotherapy compared to chemotherapy alone. Another phase 3 trial did not show improvement in 1-year OS with consolidative thoracic radiotherapy, but 2-year OS and 6-month PFS were longer.86 In general, consolidative thoracic radiotherapy benefits patients who have residual thoracic disease and low-bulk extrathoracic disease that has responded to systemic therapy.87 In addition, patients who initially presented with bulky symptomatic thoracic disease should also be considered for consolidative radiation.
Similar to other solid tumors, radiotherapy should be utilized for palliative purposes in patients with painful bone metastasis, spinal cord compression, or brain metastasis. Surgery is generally not recommended for spinal cord compression given the short life expectancy of patients with extensive-stage disease. Whole brain radiotherapy is preferred over stereotactic radiosurgery because micrometastasis is frequently present even in the setting of 1 or 2 radiographically evident brain metastasis.
NOVEL THERAPIES
The very complex genetic landscape of SCLC accounts for its resistance to conventional therapy and high recurrence rate; however, at the same time this complexity can form the basis for effective targeted therapy for the disease. One of the major factors hindering the development of targeted therapies in SCLC is limited availability of tissue due to small tissue samples and the frequent presence of significant necrosis in the samples. In recent years, several different therapeutic strategies and targeted agents have been investigated for their potential role in SCLC. Several of them, including EGFR tyrosine kinase inhibitors (TKIs), BCR-ABL TKIs, mTOR inhibitors, and VEGF inhibitors, have not been shown to provide a survival advantage in this disease. Several others, including PARP inhibitors, cellular developmental pathway inhibitors, and antibody-drug conjugates, are being tested. A phase 1 study of veliparib combined with cisplatin and etoposide in patients with previously untreated extensive-stage SCLC demonstrated a complete response in 14.3%, a partial response in 57.1%, and stable disease in 28.6% of patients with an acceptable safety profile.88 So far, none of these agents are approved for use in SCLC, and the majority are in early- phase clinical trials.89
One of the emerging targets in the treatment of SCLC is delta-like protein 3 (DLL3). DLL3 is expressed on more than 80% of SCLC tumor cells and cancer stem cells. Rovalpituzumab tesirine is an antibody-drug conjugate consisting of humanized anti-DLL3 monoclonal antibody linked to SC-DR002, a DNA-crosslinking agent. A phase 1 trial of rovalpituzumab in patients with relapsed SCLC after 1 or 2 prior lines of therapy reported a response rate of 31% in patients with DLL3 expression of ≥ 50%. The median duration of response and median PFS were both 4.6 months.90 Rovalpituzumab is currently in later phases of clinical trials and has a potential to serve as an option for patients with extensive-stage disease after disease progression on platinum-based therapy.
SUMMARY
Four to 6 cycles of carboplatin and etoposide remain the standard of care first-line treatment for patients with extensive stage SCLC. The only FDA-approved second-line treatment option is topotecan. Re-treatment with the original platinum doublet is a reasonable option for patients who have disease progression 6 months or longer after completion of platinum-based therapy. The immune checkpoint inhibitors pembrolizumab and combination nivolumab and ipilimumab have shown promising results in the second-line setting and beyond. The role of PCI has become more controversial in recent years, and periodic brain MRI in lieu of PCI is now an acceptable approach.
RESPONSE ASSESSMENT/SURVEILLANCE
For patients undergoing treatment for limited-stage SCLC, response assessment with contrast-enhanced CT of the chest/abdomen should be performed after completion of 4 cycles of chemotherapy and thoracic radiation.7 The surveillance guidelines consist of history, physical exam, and imaging every 3 months during the first 2 years, every 6 months during the third year, and annually thereafter. If PCI is not performed, brain MRI or contrast-enhanced CT scan should be performed every 3 or 4 months during the first 2 years of follow up. For extensive-stage disease, response assessment should be performed after every 2 cycles of therapy. After completion of therapy, history, physical exam, and imaging should be done every 2 months during the first year, every 3 or 4 months during years 2 and 3, every 6 months during years 4 and 5, and annually thereafter. Routine use of PET scan for surveillance is not recommended. Any new pulmonary nodule should prompt evaluation for a second primary lung malignancy. Finally, smoking cessation counseling is an integral part of management of any patient with SCLC and should be included with every clinic visit.
CONCLUSION
SCLC is a heterogeneous and genetically complex disease with a very high mortality rate. The current standard of care includes concurrent chemoradiation with cisplatin and etoposide for limited-stage SCLC and the combination of platinum and etoposide for extensive SCLC. A number of novel treatment approaches, including immune checkpoint inhibitors and antibody-drug conjugates, have had promising results in early clinical trials. Given the limited treatment options and large unmet need for new treatment options, enrollment in clinical trials is strongly recommended for patients with SCLC.
1. American Cancer Society. Cancer Facts & Figures 2017. American Cancer Society website. https://www.cancer.org/content/dam/cancer-org/research/cancer-facts-and-statistics/annual-cancer-facts-and-figures/2017/cancer-facts-and-figures-2017.pdf. Published 2017. Accessed July 11, 2018.
2. Govindan R, Page N, Morgensztern D, et al. Changing epidemiology of small-cell lung cancer in the United States over the last 30 years: analysis of the surveillance, epidemiologic, and end results database. J Clin Oncol 2006;24:4539–44.
3. Howlader N, Noone AM, Krapcho M, et al. SEER Cancer Statistics Review, 1975-2014. National Cancer Institute website. https://seer.cancer.gov/csr/1975_2014/. Updated April 2, 2018. Accessed July 11, 2018.
4. Varghese AM, Zakowski MF, Yu HA, et al. Small-cell lung cancers in patients who never smoked cigarettes. J Thorac Oncol 2014;9:892–6.
5. Pleasance ED, Stephens PJ, O’Meara S, et al. A small-cell lung cancer genome with complex signatures of tobacco exposure. Nature 2010;463:184–90.
6. Green RA, Humphrey E, Close H, Patno ME. Alkylating agents in bronchogenic carcinoma. Am J Med 1969;46:516–25.
7. National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology for small cell lung cancer (Version 2.2018). www.nccn.org/professionals/physician_gls/pdf/sclc.pdf. Accessed August 12, 2018.
8. National Lung Screening Trial Research Team, Aberle DR, Adams AM, et al. Reduced lung-cancer mortality with low-dose computed tomographic screening. N Engl J Med 2011;365:395–409.
9. Aberle DR, DeMello S, Berg CD, et al. Results of the two incidence screenings in the National Lung Screening Trial. N Engl J Med 2013;369:920–31.
10. Kitajima K, Nakamoto Y, Okizuka H, et al. Accuracy of whole-body FDG-PET/CT for detecting brain metastases from non-central nervous system tumors. Ann Nucl Med 2008;22:595–602.
11. Ordonez NG. Value of thyroid transcription factor-1 immunostaining in distinguishing small cell lung carcinomas from other small cell carcinomas. Am J Surg Pathol 2000;24:1217–23.
12. Karnak D, Beder S, Kayacan O, et al. Neuron-specific enolase and lung cancer. Am J Clin Oncol 2005;28:586–90.
13. Turrisi AT 3rd, Kim K, Blum R, et al. Twice-daily compared with once-daily thoracic radiotherapy in limited small-cell lung cancer treated concurrently with cisplatin and etoposide. N Engl J Med 1999;340:265–71.
14. Evans WK, Shepherd FA, Feld R, et al. VP-16 and cisplatin as first-line therapy for small-cell lung cancer. J Clin Oncol 1985;3:1471–7.
15. Pujol JL, Carestia L, Daurés JP. Is there a case for cisplatin in the treatment of small-cell lung cancer? A meta-analysis of randomized trials of a cisplatin-containing regimen versus a regimen without this alkylating agent. Br J Cancer 2000;83:8–15.
16. Mascaux C, Paesmans M, Berghmans T, et al; European Lung Cancer Working Party (ELCWP). A systematic review of the role of etoposide and cisplatin in the chemotherapy of small cell lung cancer with methodology assessment and meta-analysis. Lung Cancer 2000;30:23–36.
17. Sundstrøm S, Bremnes RM, Kaasa S, et al; Norwegian Lung Cancer Study Group. Cisplatin and etoposide regimen is superior to cyclophosphamide, epirubicin, and vincristine regimen in small-cell lung cancer: results from a randomized phase III trial with 5 years’ follow-up. J Clin Oncol 2002;20:4665–72.
18. Hatfield LA, Huskamp HA, Lamont EB. Survival and toxicity after cisplatin plus etoposide versus carboplatin plus etoposide for extensive-stage small-cell lung cancer in elderly patients. J Oncol Pract 2016;12:666–73.
19. Okamoto H, Watanabe K, Kunikane H, et al. Randomised phase III trial of carboplatin plus etoposide vs split doses of cisplatin plus etoposide in elderly or poor-risk patients with extensive disease small-cell lung cancer: JCOG 9702. Br J Cancer 2007;97:162–9.
20. Skarlos DV, Samantas E, Kosmidis P, et al. Randomized comparison of etoposide-cisplatin vs. etoposide-carboplatin and irradiation in small-cell lung cancer. A Hellenic Co-operative Oncology Group study. Ann Oncol 1994;5:601–7.
21. Rossi A, Di Maio M, Chiodini P, et al. Carboplatin- or cisplatin-based chemotherapy in first-line treatment of small-cell lung cancer: the COCIS meta-analysis of individual patient data J Clin Oncol 2012;30:1692–8.
22. Bishop JF, Raghavan D, Stuart-Harris R, et al. Carboplatin (CBDCA, JM-8) and VP-16-213 in previously untreated patients with small-cell lung cancer. J Clin Oncol 1987;5:1574–8.
23. Takada M, Fukuoka M, Kawahara M, et al. Phase III study of concurrent versus sequential thoracic radiotherapy in combination with cisplatin and etoposide for limited-stage small-cell lung cancer: results of the Japan Clinical Oncology Group Study 9104. J Clin Oncol 2002;20:3054–60.
24. Bunn PA Jr, Crowley J, Kelly K, et al. Chemoradiotherapy with or without granulocyte-macrophage colony-stimulating factor in the treatment of limited-stage small-cell lung cancer: a prospective phase III randomized study of the Southwest Oncology Group. J Clin Oncol 1995;13:1632–41.
25. Pignon JP, Arriagada R, Ihde DC, et al. A meta-analysis of thoracic radiotherapy for small-cell lung cancer. N Engl J Med 1992;327:1618–24.
26. Warde P, Payne D. Does thoracic irradiation improve survival and local control in limited-stage small-cell carcinoma of the lung? A meta-analysis. J Clin Oncol 1992;10:890–5.
27. Murray N, Coy P, Pater JL, et al. Importance of timing for thoracic irradiation in the combined modality treatment of limited-stage small-cell lung cancer. The National Cancer Institute of Canada Clinical Trials Group. J Clin Oncol 1993;11:336–44.
28. De Ruysscher D, Lueza B, Le Péchoux C, et al. Impact of thoracic radiotherapy timing in limited-stage small-cell lung cancer: usefulness of the individual patient data meta-analysis. Ann Oncol 2016;27:1818–28.
29. Faivre-Finn C, Snee M, Ashcroft L, et al. Concurrent once-daily versus twice-daily chemoradiotherapy in patients with limited-stage small-cell lung cancer (CONVERT): an open-label, phase 3, randomised, superiority trial. Lancet Oncol 2017;18:1116–25.
30. Aupérin A, Arriagada R, Pignon JP, et al. Prophylactic cranial irradiation for patients with small-cell lung cancer in complete remission. Prophylactic Cranial Irradiation Overview Collaborative Group. N Engl J Med 1999;341:476–84.
31. Arriagada R, Le Chevalier T, Borie F, et al. Prophylactic cranial irradiation for patients with small-cell lung cancer in complete remission. J Natl Cancer Inst 1995;87:183–90.
32. Le Péchoux C, Dunant A, Senan S, et al; Prophylactic Cranial Irradiation (PCI) Collaborative Group. Standard-dose versus higher-dose prophylactic cranial irradiation (PCI) in patients with limited-stage small-cell lung cancer in complete remission after chemotherapy and thoracic radiotherapy (PCI 99-01, EORTC 22003-08004, RTOG 0212, and IFCT 99-01): a randomised clinical trial. Lancet Oncol 2009;10:467–74.
33. Schneider BJ, Saxena A, Downey RJ. Surgery for early-stage small cell lung cancer. J Natl Compr Canc Netw 2011;9:1132–9.
34. Inoue M, Nakagawa K, Fujiwara K, et al. Results of preoperative mediastinoscopy for small cell lung cancer. Ann Thorac Surg 2000;70:1620–3.
35. Lim E, Belcher E, Yap YK, et al. The role of surgery in the treatment of limited disease small cell lung cancer: time to reevaluate. J Thorac Oncol 2008;3:1267–71.
36. Inoue M, Miyoshi S, Yasumitsu T, et al. Surgical results for small cell lung cancer based on the new TNM staging system. Thoracic Surgery Study Group of Osaka University, Osaka, Japan. Ann Thorac Surg 2000;70:1615–9.
37. Yang CF, Chan DY, Speicher PJ, et al. Role of adjuvant therapy in a population-based cohort of patients with early-stage small-cell lung cancer. J Clin Oncol 2016;34:1057–64.
38. Shepherd FA, Evans WK, Feld R, et al. Adjuvant chemotherapy following surgical resection for small-cell carcinoma of the lung. J Clin Oncol 1988;6:832–8.
39. Noda K, Nishiwaki Y, Kawahara M, et al; Japan Clinical Oncology Group. Irinotecan plus cisplatin compared with etoposide plus cisplatin for extensive small-cell lung cancer. N Engl J Med 2002;346:85–91.
40. Lara PN Jr, Natale R, Crowley J, et al. Phase III trial of irinotecan/cisplatin compared with etoposide/cisplatin in extensive-stage small-cell lung cancer: clinical and pharmacogenomic results from SWOG S0124. J Clin Oncol 2009;27:2530–5.
41. Chute JP, Chen T, Feigal E, et al. Twenty years of phase III trials for patients with extensive-stage small-cell lung cancer: perceptible progress. J Clin Oncol 1999;17:1794–801.
42. Zhou H, Zeng C, Wei Y, et al. Duration of chemotherapy for small cell lung cancer: a meta-analysis. PloS One 2013;8:e73805.
43. Loehrer PJ Sr, Ansari R, Gonin R, et al. Cisplatin plus etoposide with and without ifosfamide in extensive small-cell lung cancer: a Hoosier Oncology Group study. J Clin Oncol;13:2594–9.
44. Pujol JL, Daurés JP, Riviére A, et al. Etoposide plus cisplatin with or without the combination of 4’-epidoxorubicin plus cyclophosphamide in treatment of extensive small-cell lung cancer: a French Federation of Cancer Institutes multicenter phase III randomized study. J Natl Cancer Inst 2001;93:300–8.
45. Berghmans T, Scherpereel A, Meert AP, et al; European Lung Cancer Working Party (ELCWP). A phase III randomized study comparing a chemotherapy with cisplatin and etoposide to a etoposide regimen without cisplatin for patients with extensive small-cell lung cancer. Front Oncol 2017;7:217.
46. Jalal SI, Lavin P, Lo G, et al. Carboplatin and etoposide with or without palifosfamide in untreated extensive-stage small-cell lung cancer: a Multicenter, Adaptive, Randomized Phase III Study (MATISSE). J Clin Oncol 2017;35:2619–23.
47. Fukuoka M, Furuse K, Saijo N, et al. Randomized trial of cyclophosphamide, doxorubicin, and vincristine versus cisplatin and etoposide versus alternation of these regimens in small-cell lung cancer. J Natl Cancer Inst 1991;83:855–61.
48. Roth BJ, Johnson DH, Einhorn LH, et al. Randomized study of cyclophosphamide, doxorubicin, and vincristine versus etoposide and cisplatin versus alternation of these two regimens in extensive small-cell lung cancer: a phase III trial of the Southeastern Cancer Study Group. J Clin Oncol 1992;10:282–91.
49. Miles DW, Earl HM, Souhami RL, et al. Intensive weekly chemotherapy for good-prognosis patients with small-cell lung cancer. J Clin Oncol 1991;9:280–5.
50. Petrioli R, Roviello G, Laera L, et al. Cisplatin, etoposide, and bevacizumab regimen followed by oral etoposide and bevacizumab maintenance treatment in patients with extensive-stage small cell lung cancer: a single-institution experience. Clin Lung Cancer 2015;16:e229–34.
51. Spigel DR, Greco FA, Zubkus JD, et al. Phase II trial of irinotecan, carboplatin, and bevacizumab in the treatment of patients with extensive-stage small-cell lung cancer. J Thorac Oncol 2009;4:1555–60.
52. Spigel DR, Townley PM, Waterhouse DM, et al. Randomized phase II study of bevacizumab in combination with chemotherapy in previously untreated extensive-stage small-cell lung cancer: results from the SALUTE trial. J Clin Oncol 2011;29:2215–22.
53. Horn L, Dahlberg SE, Sandler AB, et al. Phase II study of cisplatin plus etoposide and bevacizumab for previously untreated, extensive-stage small-cell lung cancer: Eastern Cooperative Oncology Group Study E3501. J Clin Oncol 2009;27:6006–11.
54. Tiseo M, Boni L, Ambrosio F, et al. Italian, multicenter, phase III, randomized study of cisplatin plus etoposide with or without bevacizumab as first-line treatment in extensive-disease small-cell lung cancer: the GOIRC-AIFA FARM6PMFJM trial. J Clin Oncol 2017;35:1281–7.
55. Pujol JL, Lavole A, Quoix E, et al. Randomized phase II-III study of bevacizumab in combination with chemotherapy in previously untreated extensive small-cell lung cancer: results from the IFCT-0802 trial. Ann Oncol 2015;26:908–14.
56. Gadgeel SM, Ventimiglia J, Kalemkerian GP, et al. Phase II study of maintenance pembrolizumab (pembro) in extensive stage small cell lung cancer (ES-SCLC) patients (pts) [abstract]. J Clin Oncol 2017;35(15_suppl):8504.
57. Reck M, Luft A, Szczesna A, et al. Phase III randomized trial of ipilimumab plus etoposide and platinum versus placebo plus etoposide and platinum in extensive-stage small-cell lung cancer. J Clin Oncol 2016;34:3740–8.
58. Chung HC, Lopez-Martin JA, Kao SC, et al. Phase 2 study of pembrolizumab in advanced small-cell lung cancer (SCLC): KEYNOTE-158 [abstract]. J Clin Oncol 2018;36(suppl):8506.
59. Owonikoko TK, Behera M, Chen Z, et al. A systematic analysis of efficacy of second-line chemotherapy in sensitive and refractory small-cell lung cancer. J Thorac Oncol 2012;7:866–72.
60. Postmus PE, Berendsen HH, van Zandwijk N, et al. Retreatment with the induction regimen in small cell lung cancer relapsing after an initial response to short term chemotherapy. Eur J Cancer Clin Oncol 1987;23:1409–11.
61. von Pawel J, Schiller JH, Shepherd FA, et al. Topotecan versus cyclophosphamide, doxorubicin, and vincristine for the treatment of recurrent small-cell lung cancer. J Clin Oncol 1999;17:658–667.
62. O’Brien ME, Ciuleanu TE, Tsekov H, et al. Phase III trial comparing supportive care alone with supportive care with oral topotecan in patients with relapsed small-cell lung cancer. J Clin Oncol 2006;24:5441–7.
63. Eckardt JR, von Pawel J, Pujol JL, et al. Phase III study of oral compared with intravenous topotecan as second-line therapy in small-cell lung cancer. J Clin Oncol 2007;25:2086–92.
64. Masuda N, Fukuoka M, Kusunoki Y, et al. CPT-11: a new derivative of camptothecin for the treatment of refractory or relapsed small-cell lung cancer. J Clin Oncol 1992;10:1225–9.
65. Smit EF, Fokkema E, Biesma B, et al. A phase II study of paclitaxel in heavily pretreated patients with small-cell lung cancer. Br J Cancer 1998;77:347–51.
66. Yamamoto N, Tsurutani J, Yoshimura N, et al. Phase II study of weekly paclitaxel for relapsed and refractory small cell lung cancer. Anticancer Res 2006;26:777–81.
67. Smyth JF, Smith IE, Sessa C, et al. Activity of docetaxel (Taxotere) in small cell lung cancer. Eur J Cancer 1994;30A:1058–60.
68. Pietanza MC, Kadota K, Huberman K, et al. Phase II trial of temozolomide with relapsed sensitive or refractory small cell lung cancer, with assessment of methylguanine-DNA methyltransferase as a potential biomarker. Clin Cancer Res 2012;18:1138–45.
69. Zauderer MG, Drilon A, Kadota K, et al. Trial of a 5-day dosing regimen of temozolomide in patients with relapsed small cell lung cancers with assessment of methylguanine-DNA methyltransferase. Lung Cancer 2014;86:237–40.
70. Jassem J, Karnicka-Mlodkowska H, van Pottelsberghe C, et al. Phase II study of vinorelbine (Navelbine) in previously treated small cell lung cancer patients. Eur J Cancer 1993;29A:1720–2.
71. Furuse K, Kuboa K, Kawahara M, et al. Phase II study of vinorelbine in heavily previously treated small cell lung cancer. Oncology 1996;53:169–72.
72. Einhorn LH, Pennington K, McClean J. Phase II trial of daily oral VP-16 in refractory small cell lung cancer. Semin Oncol 1990;17:32–5.
73. Johnson DH, Greco FA, Strupp J, et al. Prolonged administration of oral etoposide in patients with relapsed or refractory small-cell lung cancer: a phase II trial. J Clin Oncol 1990;8:1613–7.
74. Van der Lee I, Smit EF, van Putten JW, et al. Single-agent gemcitabine in patients with resistant small-cell lung cancer. Ann Oncol 2001;12:557–61.
75. Masters GA, Declerck L, Blanke C, et al. Phase II trial of gemcitabine in refractory or relapsed small-cell lung cancer. J Clin Oncol 2003;21:1550–5.
76. von Pawel J, Schiller JH, Shepherd FA, et al. Topotecan versus cyclophosphamide, doxorubicin, and vincristine for the treatment of recurrent small-cell lung cancer. J Clin Oncol 1999;17:658–67.
77. Lammers PE, Shyr Y, Li CI, et al. Phase II study of bendamustine in relapsed chemotherapy sensitive or resistant small-cell lung cancer. J Thorac Oncol 2014;9:559–62.
78. Hellmann MD, Ott PA, Zugazagoitia J, et al. Nivolumab (nivo) ± ipilimumab (ipi) in advanced small-cell lung cancer (SCLC): First report of a randomized expansion cohort from CheckMate 032 [abstract]. J Clin Oncol 2017;35(15_suppl):8503.
79. Antonia SJ, López-Martin JA, Bendell J, et al. Nivolumab alone and nivolumab plus ipilimumab in recurrent small-cell lung cancer (CheckMate 032): a multicentre, open-label, phase 1/2 trial. Lancet Oncol 2016;17:883–95.
80. Ott PA, Elez E, Hiret S, et al. Pembrolizumab in patients with extensive-stage small-cell lung cancer: results from the Phase Ib KEYNOTE-028 study. J Clin Oncol 2017;35:3823–9.
81. Slotman B, Faivre-Finn C, Kramer G, et al. Prophylactic cranial irradiation in extensive small-cell lung cancer. N Engl J Med 2007;357:664–72.
82. Takahashi T, Yamanaka T, Seto T, et al. Prophylactic cranial irradiation versus observation in patients with extensive-disease small-cell lung cancer: a multicentre, randomised, open-label, phase 3 trial. Lancet Oncol 2017;18:663–71.
83. Slotman BJ, Mauer ME, Bottomley A, et al. Prophylactic cranial irradiation in extensive disease small-cell lung cancer: short-term health-related quality of life and patient reported symptoms: results of an international Phase III randomized controlled trial by the EORTC Radiation Oncology and Lung Cancer Groups. J Clin Oncol 2009;27:78–84.
84. Brown PD, Pugh S, Laack NN, et al; Radiation Therapy Oncology Group (RTOG). Memantine for the prevention of cognitive dysfunction in patients receiving whole-brain radiotherapy: a randomized, double-blind, placebo-controlled trial. Neuro Oncol 2013;15:1429–37.
85. Jeremic B, Shibamoto Y, Nikolic N, et al. Role of radiation therapy in the combined-modality treatment of patients with extensive disease small-cell lung cancer: a randomized study. J Clin Oncol 1999;17:2092–9.
86. Slotman BJ, van Tinteren H, Praag JO, et al. Use of thoracic radiotherapy for extensive stage small-cell lung cancer: a phase 3 randomised controlled trial. Lancet 2015;385:36–42.
87. Slotman BJ, van Tinteren H, Praag JO, et al. Radiotherapy for extensive stage small-cell lung cancer - authors’ reply. Lancet 2015;385:1292–3.
88. Owonikoko TK, Dahlberg SE, Khan SA, et al. A phase 1 safety study of veliparib combined with cisplatin and etoposide in extensive stage small cell lung cancer: A trial of the ECOG-ACRIN Cancer Research Group (E2511). Lung Cancer 2015;89:66–70.
89. Mamdani H, Induru R, Jalal SI. Novel therapies in small cell lung cancer. Transl Lung Cancer Res 2015;4:533–44.
90. Rudin CM, Pietanza MC, Bauer TM, et al. Rovalpituzumab tesirine, a DLL3-targeted antibody-drug conjugate, in recurrent small-cell lung cancer: a first-in-human, first-in-class, open-label, phase 1 study. Lancet Oncol 2017;18:42–51.
INTRODUCTION
Small cell lung cancer (SCLC) is an aggressive cancer of neuroendocrine origin that accounts for approximately 15% of all lung cancer cases, with approximately 33,000 patients diagnosed annually.1 The incidence of SCLC in the United States has steadily declined over the past 30 years, presumably because of a decrease in the number of smokers and a change to low-tar filter cigarettes.2 Although the overall incidence of SCLC has been decreasing, the incidence in women is increasing and the male-to-female incidence ratio is now 1:1.3 Nearly all cases of SCLC are associated with heavy tobacco exposure, making it a heterogeneous disease with a complex genomic landscape consisting of thousands of mutations.4,5 Despite recent advances in the treatment of non-small cell lung cancer, the therapeutic options for SCLC remain limited, with a median overall survival (OS) of 9 months in patients with advanced disease.
DIAGNOSIS AND STAGING
CASE PRESENTATION
A 61-year-old man presents to the emergency department with progressive shortness of breath and cough over the past 6 weeks. He also reports a 20-lb weight loss over the same period. He is a current smoker and has been smoking 1 pack of cigarettes per day since the age of 18 years. A chest radiograph obtained in the emergency department shows a right hilar mass. Computed tomography (CT) scan confirms the presence of a 4.5-cm right hilar mass and enlarged mediastinal lymph nodes bilaterally.
• What are the next steps in diagnosis?
SCLC is characterized by rapid growth and early hematogenous metastasis. Consequently, only 25% of patients have limited-stage disease at the time of diagnosis. According to the Veterans Administration Lung Study Group (VALSG) staging system, limited-stage disease is defined as tumor that is confined to 1 hemithorax and can be encompassed within 1 radiation field. This typically includes mediastinal lymph nodes and ipsilateral supraclavicular lymph nodes. Approximately 75% of patients present with extensive-stage disease, which is defined as disease that cannot be classified as limited, including disease that extends beyond 1 hemithorax. Extensive-stage disease includes the presence of malignant pleural effusion and/or distant metastasis.6 The VALSG classification and staging system is more commonly used in clinical practice than the American Joint Committee on Cancer TNM staging system because it is less complex and directs treatment decisions, as most of the literature on SCLC classifies patients based on the VALSG system.7
Given SCLC’s propensity to metastasize quickly, none of the currently available screening methods have proven successful in early detection of SCLC. In the National Lung Cancer Screening Trial, 86% of the 125 patients who were diagnosed with SCLC while undergoing annual low-dose chest CT scans had advanced disease at diagnosis.8,9 These results highlight the fact that most cases of SCLC develop in the interval between annual screening imaging.
SCLC frequently presents with a large hilar mass that is symptomatic. Common symptoms include shortness of breath and cough. In addition, patients with SCLC usually have bulky mediastinal adenopathy at presentation. SCLC is commonly located submucosally in the bronchus, and therefore hemoptysis is not a very common symptom at the time of presentation. Patients may present with superior vena cava syndrome from local compression by the tumor. Not infrequently, SCLC is associated with paraneoplastic syndromes that arise due to ectopic secretion of hormones or antibodies by the tumor cells. The paraneoplastic syndromes can be broadly categorized as endocrine or neurologic (Table 1). The presence of a paraneoplastic syndrome is often a clue to the potential diagnosis of SCLC in the presence of a hilar mass. Additionally, some paraneoplastic syndromes, more specifically endocrine paraneoplastic syndromes, follow the pattern of disease response and relapse, and therefore can sometimes serve as an early marker of disease relapse or progression.
The common sites of metastases include brain, liver, and bone. Therefore, the staging workup should include fluorodeoxyglucose (FDG) positron emission tomography (PET)/CT scan. Contrast-enhanced CT scan of the chest and abdomen and bone scan can be obtained for staging in lieu of PET scan. Due to the physiologic FDG uptake, cerebral metastases cannot be assessed with sufficient certainty using PET-CT.10 Therefore, brain imaging with contrast-enhanced CT or magnetic resonance imaging (MRI) is also necessary. Although the incidence of metastasis to bone marrow is less than 10%, bone marrow aspiration and biopsy are warranted in patients with unexplained cytopenias, especially when the cytopenia is associated with teardrop-shaped red cells or nucleated red cells on peripheral blood smear, findings indicative of a marrow infiltrative process.7 The tissue diagnosis is established by obtaining a biopsy of the primary tumor or 1 of the metastatic sites. In localized disease, bronchoscopy (with endobronchial ultrasound, if necessary) with biopsy of the centrally located tumor and/or lymph node is required. Histologically, SCLC consists of monomorphic cells, a high nuclear-cytoplasmic ratio, and confluent necrosis. The tumor cells are positive for chromogranin, synaptophysin, and CD56 by immunohistochemistry, and very frequently are also positive for thyroid transcription factor 1.11 Although serum tumor markers, including neuron-specific enolase and progastrin-releasing peptide, are frequently elevated in patients with SCLC, these markers are of limited value in clinical practice because they lack sensitivity and specificity.12
MANAGEMENT OF LIMITED-STAGE DISEASE
CASE CONTINUED
The patient undergoes FDG PET scan, which shows the presence of a hypermetabolic right hilar mass in addition to enlarged and hypermetabolic bilateral mediastinal lymph nodes. There are no other areas of FDG avidity. Brain MRI does not show any evidence of brain metastasis. Thus, the patient is confirmed to have limited-stage SCLC.
• What is the standard of care treatment for limited-stage SCLC?
SCLC is exquisitely sensitive to both chemotherapy and radiation, especially at the time of initial presentation. The standard of care treatment of limited-stage SCLC is 4 cycles of platinum-based chemotherapy in combination with thoracic radiation started within the first 2 cycles of chemotherapy (Figure 1).
CHOICE OF CHEMOTHERAPY
Etoposide and cisplatin is the most commonly used initial combination chemotherapy regimen in limited-stage SCLC.14 This combination has largely replaced anthracycline-based regimens given its favorable efficacy and toxicity profile.15–17 Several small randomized trials have shown comparable efficacy of carboplatin and etoposide in extensive-stage SCLC.18–20 A meta-analysis of 4 randomized trials comparing cisplatin-based versus carboplatin-based regimens in 663 patients with SCLC (32% had limited-stage disease and 68% had extensive-stage disease) showed no statistically significant difference in response rate, progression-free survival (PFS), or OS between the 2 regimens.21 Therefore, in clinical practice carboplatin is frequently used instead of cisplatin in patients with extensive-stage disease. In patients with limited-stage disease, cisplatin is still the drug of choice. However, the toxicity profile of the 2 regimens is different. Cisplatin-based regimens are more commonly associated with neuropathy, nephrotoxicity, and chemotherapy-induced nausea/vomiting,18 while carboplatin-based regimens are more myelosuppressive.22 In addition, the combination of thoracic radiation with either of these regimens is associated with a higher risk of esophagitis, pneumonitis, and myelosuppression.23 The use of myeloid growth factors is not recommended in patients undergoing concurrent chemoradiation.24 Of note, intravenous etoposide is always preferred over oral etoposide, especially in the curative setting given the unreliable absorption and bioavailability of oral formulations.
THORACIC RADIOTHERAPY
Adding thoracic radiotherapy to platinum-etoposide chemotherapy improves local control and OS. Two meta-analyses of 13 trials including more than 2000 patients have shown a 25% to 30% decrease in local failure and a 5% to 7% increase in 2-year OS with chemoradiation compared to chemotherapy alone in limited-stage SCLC.25,26 Early (within the first 2 cycles) concurrent thoracic radiation is superior to delayed and/or sequential radiation in terms of local control and OS.23,27,28 The dose and fractionation of thoracic radiation in limited-stage SCLC has remained a controversial issue. The Eastern Cooperative Oncology Group/Radiation Therapy Oncology Group randomized trial compared 45 Gy of radiotherapy delivered twice daily over a period of 3 weeks to 45 Gy once daily over 5 weeks concurrently with chemotherapy. The twice daily regimen led to a 10% improvement in 5-year OS (26% versus 16%), but a higher incidence of grade 3 and 4 adverse events.13 Despite the survival advantage demonstrated by hyperfractionated radiotherapy, the results need to be interpreted with caution because the radiation doses are not biologically equivalent. In addition, the difficult logistics of patients receiving radiation twice a day has limited the routine implementation of this strategy. Subsequently, another randomized phase 3 trial (CONVERT) compared 45 Gy radiotherapy twice daily with 66 Gy radiotherapy once daily in limited-stage SCLC.29 This trial did not show any difference in OS. The patients in the twice daily arm had a higher incidence of grade 4 neutropenia. Considering the results of these trials, both strategies—45 Gy fractionated twice daily or 60 Gy fractionated once daily, delivered concurrently with chemotherapy—are acceptable in the setting of limited-stage SCLC. However, quite often a hyperfractionated regimen is not feasible for patients and many radiation oncology centers. Hopefully, the ongoing CALGB 30610 study will clarify the optimal radiation schedule for limited-stage disease.
PROPHYLACTIC CRANIAL IRRADIATION
Approximately 75% of patients with limited-stage disease experience disease recurrence, and brain is the site of recurrence in approximately half of these patients.30 Prophylactic cranial irradiation (PCI) consisting of 25 Gy radiotherapy delivered in 10 fractions has been shown to be effective in decreasing the incidence of cerebral metastases.30–32 Although individual small studies have not shown a survival benefit of PCI because of small sample size and limited power, a meta-analysis of these studies has shown a 25% decrease in the 3-year incidence of brain metastasis and 5.4% increase in 3-year OS.30 Most patients included in these studies had limited-stage disease. Therefore, PCI is the standard of care for patients with limited-stage disease who attain a partial or complete response to chemoradiation.
ROLE OF SURGERY
Surgical resection may be an acceptable choice in a very limited subset of patients with peripherally located small (< 5 cm) tumors where mediastinal lymph nodes have been confirmed to be uninvolved with complete mediastinal staging.33,34 Most of the data in this setting are derived from retrospective studies.35,36 A 5-year OS between 40% and 60% has been reported with this strategy in patients with clinical stage I disease. In general, when surgery is considered, lobectomy with mediastinal lymph node dissection followed by chemotherapy (if there is no nodal involvement) or chemoradiation (if nodal involvement) is recommended.37,38 Wedge or segmental resections are not considered to be optimal surgical options.
MANAGEMENT OF EXTENSIVE-STAGE DISEASE
CASE CONTINUED
The patient receives 4 cycles of cisplatin and etoposide along with 70 Gy radiotherapy concurrently with the first 2 cycles of chemotherapy. His post-treatment CT scans show a partial response. He undergoes PCI 6 weeks after completion of treatment. At routine follow-up 18 months later, he is doing generally well except for mildly decreased appetite and an unintentional weight loss of 5 lb. CT scans demonstrate multiple hypodense liver lesions ranging from 7 mm to 2 cm in size and a 2-cm left adrenal gland lesion highly concerning for metastasis. FDG PET scan confirms that the adrenal and liver lesions are hypermetabolic. In addition, the PET scan shows multiple FDG-avid bone lesions throughout the spine. Brain MRI is negative for brain metastasis.
• What is the standard of care for treatment of extensive-stage disease?
Chemotherapy is the mainstay of treatment for extensive-stage SCLC; the goals of treatment are prolongation of survival, prevention or alleviation of cancer-related symptoms, and improvement in quality of life. The combination of etoposide with a platinum agent (carboplatin or cisplatin) is the preferred first-line treatment option. Carboplatin is more commonly used in clinical practice in this setting because of its comparable efficacy and better tolerability compared to cisplatin (Figure 2).21 A Japanese phase 3 trial comparing cisplatin plus irinotecan with cisplatin plus etoposide in the first-line setting in extensive-stage SCLC showed improvement in median and 2-year OS with the cisplatin/irinotecan regimen; however, 2 subsequent phase 3 trials conducted in the United States comparing these 2 regimens did not show any difference in OS. In addition, the cisplatin/irinotecan regimen was more toxic than the etoposide-based regimen.39,40 Therefore, 4 to 6 cycles of platinum/etoposide remains the standard of care first-line treatment for extensive-stage SCLC in the United States. The combination yields a 60% to 70% response rate, but the majority of patients invariably experience disease progression, with a median OS of 9 to 11 months.41 Maintenance chemotherapy beyond the initial 4 to 6 cycles does not improve survival and is associated with higher cumulative toxicity.42
Multiple attempts at improving first-line chemotherapy in extensive-stage disease have failed to show any meaningful difference in OS. For example, the addition of ifosfamide, palifosfamide, cyclophosphamide, taxane, or anthracycline to platinum doublet failed to show improvement in OS and led to more toxicity.43–46 Additionally, the use of alternating or cyclic chemotherapies in an attempt to curb drug resistance has also failed to show survival benefit.47–49 The addition of the antiangiogenic agent bevacizumab to standard platinum-based doublet has not prolonged OS in SCLC and has led to an unacceptably higher rate of tracheoesophageal fistula when used in conjunction with chemoradiation in limited-stage disease.50–55 Finally, the immune checkpoint inhibitor ipilimumab in combination with platinum plus etoposide failed to improve PFS or OS compared to platinum plus etoposide alone in a recent phase 3 trial, and maintenance pembrolizumab after completion of platinum-based chemotherapy did not improve PFS.56,57
More recently, a phase 2 study of pembrolizumab in extensive-stage SCLC (KEYNOTE 158) reported an overall response rate of 35.7%, median PFS of 2.1 months, and median OS of 14.6 months in patients who tested positive for programmed death ligand-1 (PD-L1) expression (which was defined as a PD-L1 Combined Positive Score ≥ 1).58 The median duration of response has not been reached in this study, indicating that pembrolizumab may be a promising approach in patients with extensive-stage SCLC, especially for those with PD-L1–positive tumors.
Patients with extensive-stage disease who have brain metastasis at the time of diagnosis can be treated with systemic chemotherapy first if the brain metastases are asymptomatic and there is significant extracranial disease burden. In that case, whole brain radiotherapy should be given after completion of systemic therapy.
SECOND-LINE CHEMOTHERAPY
Despite being exquisitely chemosensitive, SCLC is associated with a very poor prognosis largely because of invariable disease progression following first-line therapy and lack of effective second-line treatment options that can lead to appreciable disease control. The choice of second-line treatment is predominantly determined by the time of disease relapse after first-line platinum-based therapy. If this interval is 6 months or longer, re-treatment utilizing the same platinum doublet is appropriate. However, if the interval is 6 months or less, second-line systemic therapy options should be explored. Unfortunately, the response rate tends to be less than 10% with most of the second-line therapies in platinum-resistant disease (defined as disease progression within 3 months of receiving platinum-based therapy). If disease progression occurs between 3 and 6 months after completion of platinum-based therapy, the response rate with second-line chemotherapy is in the range of 25%.59,60
A number of second-line chemotherapy options have been explored in small studies, including topotecan, irinotecan, paclitaxel, docetaxel, temozolomide, vinorelbine, oral etoposide, gemcitabine, bendamustine, and CAV (
IMMUNOTHERAPY
The role of immune checkpoint inhibitors in the treatment of SCLC is evolving, and currently there are no FDA-approved immunotherapy agents for treating SCLC. A recently conducted phase 1/2 trial (CheckMate 032) studied the anti-programmed death(PD)-1 antibody nivolumab with or without the anti-cytotoxic T-lymphocyte–associated antigen (CTLA) -4 antibody ipilimumab in patients with relapsed SCLC. The authors reported response rates of 10% with nivolumab 3 mg/kg and 21% with nivolumab 1 mg/kg plus ipilimumab 3 mg/kg.78,79 The 2-year OS was 26% with the combination and 14% with single-agent nivolumab. Only 18% of patients had PD-L1 expression of ≥ 1%, and the response rate did not correlate with PD-L1 status. The rate of grade 3 or 4 adverse events was approximately 20%, and only 10% of patients discontinued treatment because of toxicity. Based on these data, nivolumab plus ipilimumab is now included in the National Comprehensive Cancer Network guidelines as an option for patients with SCLC who experience disease relapse within 6 months of receiving platinum-based therapy;7 however, it is questionable whether routine use of this combination is justified based on currently available data. The evidence for the combination of nivolumab and ipilimumab remains limited. The efficacy and toxicity data from both randomized and nonrandomized cohorts were presented together, making it hard to interpret the results.
Another phase 1b study (KEYNOTE-028) evaluated the anti-PD-1 antibody pembrolizumab (10 mg/kg intravenously every 2 weeks) in patients with relapsed SCLC who had received 1 or more prior lines of therapy and had PD-L1 expression of ≥ 1%. This study showed a response rate of 33%, with a median duration of response of 19 months and 1-year OS of 38%.80 Although only 28% of screened patients had PD-L1 expression of ≥ 1%, these results indicated that at least a subset of SCLC patients are able to achieve durable responses with immune checkpoint inhibition. A number of clinical trials utilizing immune checkpoint inhibitors in various combinations and settings are currently underway.
ROLE OF PROPHYLACTIC CRANIAL IRRADIATION
The role of PCI in extensive-stage SCLC is not clearly defined. A randomized phase 3 trial conducted by the European Organization for Research and Treatment of Cancer (EORTC) comparing PCI with no PCI in patients with extensive-stage SCLC who had a partial or complete response to initial platinum-based chemotherapy showed a decrease in the incidence of symptomatic brain metastasis and improvement in 1-year OS with PCI.81 However, this trial did not require mandatory brain imaging prior to PCI, and thus it is unclear if some patients in the PCI group had asymptomatic brain metastasis prior to enrollment and therefore received therapeutic benefit from brain radiation. Additionally, the dose and fractionation of PCI was not standardized across patient groups.
A more recent phase 3 study conducted in Japan that compared PCI (25 Gy in 10 fractions) with no PCI reported no difference in survival between the 2 groups.82 As opposed to the EORTC study, the Japanese study did require baseline brain imaging to confirm the absence of brain metastasis prior to enrollment. In addition, the control patients underwent periodic brain MRI to allow early detection of brain metastasis. Given the emergence of the new data, the impact of PCI on survival in patients with extensive-stage SCLC is unproven, and PCI likely has a role in a highly selected small group of patients with extensive-stage SCLC. PCI is not recommended for patients with poor performance status (ECOG performance score of 3 or 4) or underlying neurocognitive disorders.34,83
The NMDA-receptor antagonist memantine can be used in patients undergoing PCI to delay the occurrence of cognitive dysfunction.61 Memantine 20 mg daily delayed time to cognitive decline and reduced the rate of decline in memory, executive function, and processing speed compared to placebo in patients receiving whole brain radiotherapy.84
ROLE OF RADIOTHERAPY
A subset of patients with extensive-stage SCLC may benefit from consolidative thoracic radiotherapy after completion of platinum-based chemotherapy. A randomized trial that enrolled patients who achieved complete or near complete response after 3 cycles of cisplatin plus etoposide compared thoracic radiotherapy in combination with continued chemotherapy versus chemotherapy alone.85 The median OS was longer with the addition of thoracic radiotherapy compared to chemotherapy alone. Another phase 3 trial did not show improvement in 1-year OS with consolidative thoracic radiotherapy, but 2-year OS and 6-month PFS were longer.86 In general, consolidative thoracic radiotherapy benefits patients who have residual thoracic disease and low-bulk extrathoracic disease that has responded to systemic therapy.87 In addition, patients who initially presented with bulky symptomatic thoracic disease should also be considered for consolidative radiation.
Similar to other solid tumors, radiotherapy should be utilized for palliative purposes in patients with painful bone metastasis, spinal cord compression, or brain metastasis. Surgery is generally not recommended for spinal cord compression given the short life expectancy of patients with extensive-stage disease. Whole brain radiotherapy is preferred over stereotactic radiosurgery because micrometastasis is frequently present even in the setting of 1 or 2 radiographically evident brain metastasis.
NOVEL THERAPIES
The very complex genetic landscape of SCLC accounts for its resistance to conventional therapy and high recurrence rate; however, at the same time this complexity can form the basis for effective targeted therapy for the disease. One of the major factors hindering the development of targeted therapies in SCLC is limited availability of tissue due to small tissue samples and the frequent presence of significant necrosis in the samples. In recent years, several different therapeutic strategies and targeted agents have been investigated for their potential role in SCLC. Several of them, including EGFR tyrosine kinase inhibitors (TKIs), BCR-ABL TKIs, mTOR inhibitors, and VEGF inhibitors, have not been shown to provide a survival advantage in this disease. Several others, including PARP inhibitors, cellular developmental pathway inhibitors, and antibody-drug conjugates, are being tested. A phase 1 study of veliparib combined with cisplatin and etoposide in patients with previously untreated extensive-stage SCLC demonstrated a complete response in 14.3%, a partial response in 57.1%, and stable disease in 28.6% of patients with an acceptable safety profile.88 So far, none of these agents are approved for use in SCLC, and the majority are in early- phase clinical trials.89
One of the emerging targets in the treatment of SCLC is delta-like protein 3 (DLL3). DLL3 is expressed on more than 80% of SCLC tumor cells and cancer stem cells. Rovalpituzumab tesirine is an antibody-drug conjugate consisting of humanized anti-DLL3 monoclonal antibody linked to SC-DR002, a DNA-crosslinking agent. A phase 1 trial of rovalpituzumab in patients with relapsed SCLC after 1 or 2 prior lines of therapy reported a response rate of 31% in patients with DLL3 expression of ≥ 50%. The median duration of response and median PFS were both 4.6 months.90 Rovalpituzumab is currently in later phases of clinical trials and has a potential to serve as an option for patients with extensive-stage disease after disease progression on platinum-based therapy.
SUMMARY
Four to 6 cycles of carboplatin and etoposide remain the standard of care first-line treatment for patients with extensive stage SCLC. The only FDA-approved second-line treatment option is topotecan. Re-treatment with the original platinum doublet is a reasonable option for patients who have disease progression 6 months or longer after completion of platinum-based therapy. The immune checkpoint inhibitors pembrolizumab and combination nivolumab and ipilimumab have shown promising results in the second-line setting and beyond. The role of PCI has become more controversial in recent years, and periodic brain MRI in lieu of PCI is now an acceptable approach.
RESPONSE ASSESSMENT/SURVEILLANCE
For patients undergoing treatment for limited-stage SCLC, response assessment with contrast-enhanced CT of the chest/abdomen should be performed after completion of 4 cycles of chemotherapy and thoracic radiation.7 The surveillance guidelines consist of history, physical exam, and imaging every 3 months during the first 2 years, every 6 months during the third year, and annually thereafter. If PCI is not performed, brain MRI or contrast-enhanced CT scan should be performed every 3 or 4 months during the first 2 years of follow up. For extensive-stage disease, response assessment should be performed after every 2 cycles of therapy. After completion of therapy, history, physical exam, and imaging should be done every 2 months during the first year, every 3 or 4 months during years 2 and 3, every 6 months during years 4 and 5, and annually thereafter. Routine use of PET scan for surveillance is not recommended. Any new pulmonary nodule should prompt evaluation for a second primary lung malignancy. Finally, smoking cessation counseling is an integral part of management of any patient with SCLC and should be included with every clinic visit.
CONCLUSION
SCLC is a heterogeneous and genetically complex disease with a very high mortality rate. The current standard of care includes concurrent chemoradiation with cisplatin and etoposide for limited-stage SCLC and the combination of platinum and etoposide for extensive SCLC. A number of novel treatment approaches, including immune checkpoint inhibitors and antibody-drug conjugates, have had promising results in early clinical trials. Given the limited treatment options and large unmet need for new treatment options, enrollment in clinical trials is strongly recommended for patients with SCLC.
INTRODUCTION
Small cell lung cancer (SCLC) is an aggressive cancer of neuroendocrine origin that accounts for approximately 15% of all lung cancer cases, with approximately 33,000 patients diagnosed annually.1 The incidence of SCLC in the United States has steadily declined over the past 30 years, presumably because of a decrease in the number of smokers and a change to low-tar filter cigarettes.2 Although the overall incidence of SCLC has been decreasing, the incidence in women is increasing and the male-to-female incidence ratio is now 1:1.3 Nearly all cases of SCLC are associated with heavy tobacco exposure, making it a heterogeneous disease with a complex genomic landscape consisting of thousands of mutations.4,5 Despite recent advances in the treatment of non-small cell lung cancer, the therapeutic options for SCLC remain limited, with a median overall survival (OS) of 9 months in patients with advanced disease.
DIAGNOSIS AND STAGING
CASE PRESENTATION
A 61-year-old man presents to the emergency department with progressive shortness of breath and cough over the past 6 weeks. He also reports a 20-lb weight loss over the same period. He is a current smoker and has been smoking 1 pack of cigarettes per day since the age of 18 years. A chest radiograph obtained in the emergency department shows a right hilar mass. Computed tomography (CT) scan confirms the presence of a 4.5-cm right hilar mass and enlarged mediastinal lymph nodes bilaterally.
• What are the next steps in diagnosis?
SCLC is characterized by rapid growth and early hematogenous metastasis. Consequently, only 25% of patients have limited-stage disease at the time of diagnosis. According to the Veterans Administration Lung Study Group (VALSG) staging system, limited-stage disease is defined as tumor that is confined to 1 hemithorax and can be encompassed within 1 radiation field. This typically includes mediastinal lymph nodes and ipsilateral supraclavicular lymph nodes. Approximately 75% of patients present with extensive-stage disease, which is defined as disease that cannot be classified as limited, including disease that extends beyond 1 hemithorax. Extensive-stage disease includes the presence of malignant pleural effusion and/or distant metastasis.6 The VALSG classification and staging system is more commonly used in clinical practice than the American Joint Committee on Cancer TNM staging system because it is less complex and directs treatment decisions, as most of the literature on SCLC classifies patients based on the VALSG system.7
Given SCLC’s propensity to metastasize quickly, none of the currently available screening methods have proven successful in early detection of SCLC. In the National Lung Cancer Screening Trial, 86% of the 125 patients who were diagnosed with SCLC while undergoing annual low-dose chest CT scans had advanced disease at diagnosis.8,9 These results highlight the fact that most cases of SCLC develop in the interval between annual screening imaging.
SCLC frequently presents with a large hilar mass that is symptomatic. Common symptoms include shortness of breath and cough. In addition, patients with SCLC usually have bulky mediastinal adenopathy at presentation. SCLC is commonly located submucosally in the bronchus, and therefore hemoptysis is not a very common symptom at the time of presentation. Patients may present with superior vena cava syndrome from local compression by the tumor. Not infrequently, SCLC is associated with paraneoplastic syndromes that arise due to ectopic secretion of hormones or antibodies by the tumor cells. The paraneoplastic syndromes can be broadly categorized as endocrine or neurologic (Table 1). The presence of a paraneoplastic syndrome is often a clue to the potential diagnosis of SCLC in the presence of a hilar mass. Additionally, some paraneoplastic syndromes, more specifically endocrine paraneoplastic syndromes, follow the pattern of disease response and relapse, and therefore can sometimes serve as an early marker of disease relapse or progression.
The common sites of metastases include brain, liver, and bone. Therefore, the staging workup should include fluorodeoxyglucose (FDG) positron emission tomography (PET)/CT scan. Contrast-enhanced CT scan of the chest and abdomen and bone scan can be obtained for staging in lieu of PET scan. Due to the physiologic FDG uptake, cerebral metastases cannot be assessed with sufficient certainty using PET-CT.10 Therefore, brain imaging with contrast-enhanced CT or magnetic resonance imaging (MRI) is also necessary. Although the incidence of metastasis to bone marrow is less than 10%, bone marrow aspiration and biopsy are warranted in patients with unexplained cytopenias, especially when the cytopenia is associated with teardrop-shaped red cells or nucleated red cells on peripheral blood smear, findings indicative of a marrow infiltrative process.7 The tissue diagnosis is established by obtaining a biopsy of the primary tumor or 1 of the metastatic sites. In localized disease, bronchoscopy (with endobronchial ultrasound, if necessary) with biopsy of the centrally located tumor and/or lymph node is required. Histologically, SCLC consists of monomorphic cells, a high nuclear-cytoplasmic ratio, and confluent necrosis. The tumor cells are positive for chromogranin, synaptophysin, and CD56 by immunohistochemistry, and very frequently are also positive for thyroid transcription factor 1.11 Although serum tumor markers, including neuron-specific enolase and progastrin-releasing peptide, are frequently elevated in patients with SCLC, these markers are of limited value in clinical practice because they lack sensitivity and specificity.12
MANAGEMENT OF LIMITED-STAGE DISEASE
CASE CONTINUED
The patient undergoes FDG PET scan, which shows the presence of a hypermetabolic right hilar mass in addition to enlarged and hypermetabolic bilateral mediastinal lymph nodes. There are no other areas of FDG avidity. Brain MRI does not show any evidence of brain metastasis. Thus, the patient is confirmed to have limited-stage SCLC.
• What is the standard of care treatment for limited-stage SCLC?
SCLC is exquisitely sensitive to both chemotherapy and radiation, especially at the time of initial presentation. The standard of care treatment of limited-stage SCLC is 4 cycles of platinum-based chemotherapy in combination with thoracic radiation started within the first 2 cycles of chemotherapy (Figure 1).
CHOICE OF CHEMOTHERAPY
Etoposide and cisplatin is the most commonly used initial combination chemotherapy regimen in limited-stage SCLC.14 This combination has largely replaced anthracycline-based regimens given its favorable efficacy and toxicity profile.15–17 Several small randomized trials have shown comparable efficacy of carboplatin and etoposide in extensive-stage SCLC.18–20 A meta-analysis of 4 randomized trials comparing cisplatin-based versus carboplatin-based regimens in 663 patients with SCLC (32% had limited-stage disease and 68% had extensive-stage disease) showed no statistically significant difference in response rate, progression-free survival (PFS), or OS between the 2 regimens.21 Therefore, in clinical practice carboplatin is frequently used instead of cisplatin in patients with extensive-stage disease. In patients with limited-stage disease, cisplatin is still the drug of choice. However, the toxicity profile of the 2 regimens is different. Cisplatin-based regimens are more commonly associated with neuropathy, nephrotoxicity, and chemotherapy-induced nausea/vomiting,18 while carboplatin-based regimens are more myelosuppressive.22 In addition, the combination of thoracic radiation with either of these regimens is associated with a higher risk of esophagitis, pneumonitis, and myelosuppression.23 The use of myeloid growth factors is not recommended in patients undergoing concurrent chemoradiation.24 Of note, intravenous etoposide is always preferred over oral etoposide, especially in the curative setting given the unreliable absorption and bioavailability of oral formulations.
THORACIC RADIOTHERAPY
Adding thoracic radiotherapy to platinum-etoposide chemotherapy improves local control and OS. Two meta-analyses of 13 trials including more than 2000 patients have shown a 25% to 30% decrease in local failure and a 5% to 7% increase in 2-year OS with chemoradiation compared to chemotherapy alone in limited-stage SCLC.25,26 Early (within the first 2 cycles) concurrent thoracic radiation is superior to delayed and/or sequential radiation in terms of local control and OS.23,27,28 The dose and fractionation of thoracic radiation in limited-stage SCLC has remained a controversial issue. The Eastern Cooperative Oncology Group/Radiation Therapy Oncology Group randomized trial compared 45 Gy of radiotherapy delivered twice daily over a period of 3 weeks to 45 Gy once daily over 5 weeks concurrently with chemotherapy. The twice daily regimen led to a 10% improvement in 5-year OS (26% versus 16%), but a higher incidence of grade 3 and 4 adverse events.13 Despite the survival advantage demonstrated by hyperfractionated radiotherapy, the results need to be interpreted with caution because the radiation doses are not biologically equivalent. In addition, the difficult logistics of patients receiving radiation twice a day has limited the routine implementation of this strategy. Subsequently, another randomized phase 3 trial (CONVERT) compared 45 Gy radiotherapy twice daily with 66 Gy radiotherapy once daily in limited-stage SCLC.29 This trial did not show any difference in OS. The patients in the twice daily arm had a higher incidence of grade 4 neutropenia. Considering the results of these trials, both strategies—45 Gy fractionated twice daily or 60 Gy fractionated once daily, delivered concurrently with chemotherapy—are acceptable in the setting of limited-stage SCLC. However, quite often a hyperfractionated regimen is not feasible for patients and many radiation oncology centers. Hopefully, the ongoing CALGB 30610 study will clarify the optimal radiation schedule for limited-stage disease.
PROPHYLACTIC CRANIAL IRRADIATION
Approximately 75% of patients with limited-stage disease experience disease recurrence, and brain is the site of recurrence in approximately half of these patients.30 Prophylactic cranial irradiation (PCI) consisting of 25 Gy radiotherapy delivered in 10 fractions has been shown to be effective in decreasing the incidence of cerebral metastases.30–32 Although individual small studies have not shown a survival benefit of PCI because of small sample size and limited power, a meta-analysis of these studies has shown a 25% decrease in the 3-year incidence of brain metastasis and 5.4% increase in 3-year OS.30 Most patients included in these studies had limited-stage disease. Therefore, PCI is the standard of care for patients with limited-stage disease who attain a partial or complete response to chemoradiation.
ROLE OF SURGERY
Surgical resection may be an acceptable choice in a very limited subset of patients with peripherally located small (< 5 cm) tumors where mediastinal lymph nodes have been confirmed to be uninvolved with complete mediastinal staging.33,34 Most of the data in this setting are derived from retrospective studies.35,36 A 5-year OS between 40% and 60% has been reported with this strategy in patients with clinical stage I disease. In general, when surgery is considered, lobectomy with mediastinal lymph node dissection followed by chemotherapy (if there is no nodal involvement) or chemoradiation (if nodal involvement) is recommended.37,38 Wedge or segmental resections are not considered to be optimal surgical options.
MANAGEMENT OF EXTENSIVE-STAGE DISEASE
CASE CONTINUED
The patient receives 4 cycles of cisplatin and etoposide along with 70 Gy radiotherapy concurrently with the first 2 cycles of chemotherapy. His post-treatment CT scans show a partial response. He undergoes PCI 6 weeks after completion of treatment. At routine follow-up 18 months later, he is doing generally well except for mildly decreased appetite and an unintentional weight loss of 5 lb. CT scans demonstrate multiple hypodense liver lesions ranging from 7 mm to 2 cm in size and a 2-cm left adrenal gland lesion highly concerning for metastasis. FDG PET scan confirms that the adrenal and liver lesions are hypermetabolic. In addition, the PET scan shows multiple FDG-avid bone lesions throughout the spine. Brain MRI is negative for brain metastasis.
• What is the standard of care for treatment of extensive-stage disease?
Chemotherapy is the mainstay of treatment for extensive-stage SCLC; the goals of treatment are prolongation of survival, prevention or alleviation of cancer-related symptoms, and improvement in quality of life. The combination of etoposide with a platinum agent (carboplatin or cisplatin) is the preferred first-line treatment option. Carboplatin is more commonly used in clinical practice in this setting because of its comparable efficacy and better tolerability compared to cisplatin (Figure 2).21 A Japanese phase 3 trial comparing cisplatin plus irinotecan with cisplatin plus etoposide in the first-line setting in extensive-stage SCLC showed improvement in median and 2-year OS with the cisplatin/irinotecan regimen; however, 2 subsequent phase 3 trials conducted in the United States comparing these 2 regimens did not show any difference in OS. In addition, the cisplatin/irinotecan regimen was more toxic than the etoposide-based regimen.39,40 Therefore, 4 to 6 cycles of platinum/etoposide remains the standard of care first-line treatment for extensive-stage SCLC in the United States. The combination yields a 60% to 70% response rate, but the majority of patients invariably experience disease progression, with a median OS of 9 to 11 months.41 Maintenance chemotherapy beyond the initial 4 to 6 cycles does not improve survival and is associated with higher cumulative toxicity.42
Multiple attempts at improving first-line chemotherapy in extensive-stage disease have failed to show any meaningful difference in OS. For example, the addition of ifosfamide, palifosfamide, cyclophosphamide, taxane, or anthracycline to platinum doublet failed to show improvement in OS and led to more toxicity.43–46 Additionally, the use of alternating or cyclic chemotherapies in an attempt to curb drug resistance has also failed to show survival benefit.47–49 The addition of the antiangiogenic agent bevacizumab to standard platinum-based doublet has not prolonged OS in SCLC and has led to an unacceptably higher rate of tracheoesophageal fistula when used in conjunction with chemoradiation in limited-stage disease.50–55 Finally, the immune checkpoint inhibitor ipilimumab in combination with platinum plus etoposide failed to improve PFS or OS compared to platinum plus etoposide alone in a recent phase 3 trial, and maintenance pembrolizumab after completion of platinum-based chemotherapy did not improve PFS.56,57
More recently, a phase 2 study of pembrolizumab in extensive-stage SCLC (KEYNOTE 158) reported an overall response rate of 35.7%, median PFS of 2.1 months, and median OS of 14.6 months in patients who tested positive for programmed death ligand-1 (PD-L1) expression (which was defined as a PD-L1 Combined Positive Score ≥ 1).58 The median duration of response has not been reached in this study, indicating that pembrolizumab may be a promising approach in patients with extensive-stage SCLC, especially for those with PD-L1–positive tumors.
Patients with extensive-stage disease who have brain metastasis at the time of diagnosis can be treated with systemic chemotherapy first if the brain metastases are asymptomatic and there is significant extracranial disease burden. In that case, whole brain radiotherapy should be given after completion of systemic therapy.
SECOND-LINE CHEMOTHERAPY
Despite being exquisitely chemosensitive, SCLC is associated with a very poor prognosis largely because of invariable disease progression following first-line therapy and lack of effective second-line treatment options that can lead to appreciable disease control. The choice of second-line treatment is predominantly determined by the time of disease relapse after first-line platinum-based therapy. If this interval is 6 months or longer, re-treatment utilizing the same platinum doublet is appropriate. However, if the interval is 6 months or less, second-line systemic therapy options should be explored. Unfortunately, the response rate tends to be less than 10% with most of the second-line therapies in platinum-resistant disease (defined as disease progression within 3 months of receiving platinum-based therapy). If disease progression occurs between 3 and 6 months after completion of platinum-based therapy, the response rate with second-line chemotherapy is in the range of 25%.59,60
A number of second-line chemotherapy options have been explored in small studies, including topotecan, irinotecan, paclitaxel, docetaxel, temozolomide, vinorelbine, oral etoposide, gemcitabine, bendamustine, and CAV (
IMMUNOTHERAPY
The role of immune checkpoint inhibitors in the treatment of SCLC is evolving, and currently there are no FDA-approved immunotherapy agents for treating SCLC. A recently conducted phase 1/2 trial (CheckMate 032) studied the anti-programmed death(PD)-1 antibody nivolumab with or without the anti-cytotoxic T-lymphocyte–associated antigen (CTLA) -4 antibody ipilimumab in patients with relapsed SCLC. The authors reported response rates of 10% with nivolumab 3 mg/kg and 21% with nivolumab 1 mg/kg plus ipilimumab 3 mg/kg.78,79 The 2-year OS was 26% with the combination and 14% with single-agent nivolumab. Only 18% of patients had PD-L1 expression of ≥ 1%, and the response rate did not correlate with PD-L1 status. The rate of grade 3 or 4 adverse events was approximately 20%, and only 10% of patients discontinued treatment because of toxicity. Based on these data, nivolumab plus ipilimumab is now included in the National Comprehensive Cancer Network guidelines as an option for patients with SCLC who experience disease relapse within 6 months of receiving platinum-based therapy;7 however, it is questionable whether routine use of this combination is justified based on currently available data. The evidence for the combination of nivolumab and ipilimumab remains limited. The efficacy and toxicity data from both randomized and nonrandomized cohorts were presented together, making it hard to interpret the results.
Another phase 1b study (KEYNOTE-028) evaluated the anti-PD-1 antibody pembrolizumab (10 mg/kg intravenously every 2 weeks) in patients with relapsed SCLC who had received 1 or more prior lines of therapy and had PD-L1 expression of ≥ 1%. This study showed a response rate of 33%, with a median duration of response of 19 months and 1-year OS of 38%.80 Although only 28% of screened patients had PD-L1 expression of ≥ 1%, these results indicated that at least a subset of SCLC patients are able to achieve durable responses with immune checkpoint inhibition. A number of clinical trials utilizing immune checkpoint inhibitors in various combinations and settings are currently underway.
ROLE OF PROPHYLACTIC CRANIAL IRRADIATION
The role of PCI in extensive-stage SCLC is not clearly defined. A randomized phase 3 trial conducted by the European Organization for Research and Treatment of Cancer (EORTC) comparing PCI with no PCI in patients with extensive-stage SCLC who had a partial or complete response to initial platinum-based chemotherapy showed a decrease in the incidence of symptomatic brain metastasis and improvement in 1-year OS with PCI.81 However, this trial did not require mandatory brain imaging prior to PCI, and thus it is unclear if some patients in the PCI group had asymptomatic brain metastasis prior to enrollment and therefore received therapeutic benefit from brain radiation. Additionally, the dose and fractionation of PCI was not standardized across patient groups.
A more recent phase 3 study conducted in Japan that compared PCI (25 Gy in 10 fractions) with no PCI reported no difference in survival between the 2 groups.82 As opposed to the EORTC study, the Japanese study did require baseline brain imaging to confirm the absence of brain metastasis prior to enrollment. In addition, the control patients underwent periodic brain MRI to allow early detection of brain metastasis. Given the emergence of the new data, the impact of PCI on survival in patients with extensive-stage SCLC is unproven, and PCI likely has a role in a highly selected small group of patients with extensive-stage SCLC. PCI is not recommended for patients with poor performance status (ECOG performance score of 3 or 4) or underlying neurocognitive disorders.34,83
The NMDA-receptor antagonist memantine can be used in patients undergoing PCI to delay the occurrence of cognitive dysfunction.61 Memantine 20 mg daily delayed time to cognitive decline and reduced the rate of decline in memory, executive function, and processing speed compared to placebo in patients receiving whole brain radiotherapy.84
ROLE OF RADIOTHERAPY
A subset of patients with extensive-stage SCLC may benefit from consolidative thoracic radiotherapy after completion of platinum-based chemotherapy. A randomized trial that enrolled patients who achieved complete or near complete response after 3 cycles of cisplatin plus etoposide compared thoracic radiotherapy in combination with continued chemotherapy versus chemotherapy alone.85 The median OS was longer with the addition of thoracic radiotherapy compared to chemotherapy alone. Another phase 3 trial did not show improvement in 1-year OS with consolidative thoracic radiotherapy, but 2-year OS and 6-month PFS were longer.86 In general, consolidative thoracic radiotherapy benefits patients who have residual thoracic disease and low-bulk extrathoracic disease that has responded to systemic therapy.87 In addition, patients who initially presented with bulky symptomatic thoracic disease should also be considered for consolidative radiation.
Similar to other solid tumors, radiotherapy should be utilized for palliative purposes in patients with painful bone metastasis, spinal cord compression, or brain metastasis. Surgery is generally not recommended for spinal cord compression given the short life expectancy of patients with extensive-stage disease. Whole brain radiotherapy is preferred over stereotactic radiosurgery because micrometastasis is frequently present even in the setting of 1 or 2 radiographically evident brain metastasis.
NOVEL THERAPIES
The very complex genetic landscape of SCLC accounts for its resistance to conventional therapy and high recurrence rate; however, at the same time this complexity can form the basis for effective targeted therapy for the disease. One of the major factors hindering the development of targeted therapies in SCLC is limited availability of tissue due to small tissue samples and the frequent presence of significant necrosis in the samples. In recent years, several different therapeutic strategies and targeted agents have been investigated for their potential role in SCLC. Several of them, including EGFR tyrosine kinase inhibitors (TKIs), BCR-ABL TKIs, mTOR inhibitors, and VEGF inhibitors, have not been shown to provide a survival advantage in this disease. Several others, including PARP inhibitors, cellular developmental pathway inhibitors, and antibody-drug conjugates, are being tested. A phase 1 study of veliparib combined with cisplatin and etoposide in patients with previously untreated extensive-stage SCLC demonstrated a complete response in 14.3%, a partial response in 57.1%, and stable disease in 28.6% of patients with an acceptable safety profile.88 So far, none of these agents are approved for use in SCLC, and the majority are in early- phase clinical trials.89
One of the emerging targets in the treatment of SCLC is delta-like protein 3 (DLL3). DLL3 is expressed on more than 80% of SCLC tumor cells and cancer stem cells. Rovalpituzumab tesirine is an antibody-drug conjugate consisting of humanized anti-DLL3 monoclonal antibody linked to SC-DR002, a DNA-crosslinking agent. A phase 1 trial of rovalpituzumab in patients with relapsed SCLC after 1 or 2 prior lines of therapy reported a response rate of 31% in patients with DLL3 expression of ≥ 50%. The median duration of response and median PFS were both 4.6 months.90 Rovalpituzumab is currently in later phases of clinical trials and has a potential to serve as an option for patients with extensive-stage disease after disease progression on platinum-based therapy.
SUMMARY
Four to 6 cycles of carboplatin and etoposide remain the standard of care first-line treatment for patients with extensive stage SCLC. The only FDA-approved second-line treatment option is topotecan. Re-treatment with the original platinum doublet is a reasonable option for patients who have disease progression 6 months or longer after completion of platinum-based therapy. The immune checkpoint inhibitors pembrolizumab and combination nivolumab and ipilimumab have shown promising results in the second-line setting and beyond. The role of PCI has become more controversial in recent years, and periodic brain MRI in lieu of PCI is now an acceptable approach.
RESPONSE ASSESSMENT/SURVEILLANCE
For patients undergoing treatment for limited-stage SCLC, response assessment with contrast-enhanced CT of the chest/abdomen should be performed after completion of 4 cycles of chemotherapy and thoracic radiation.7 The surveillance guidelines consist of history, physical exam, and imaging every 3 months during the first 2 years, every 6 months during the third year, and annually thereafter. If PCI is not performed, brain MRI or contrast-enhanced CT scan should be performed every 3 or 4 months during the first 2 years of follow up. For extensive-stage disease, response assessment should be performed after every 2 cycles of therapy. After completion of therapy, history, physical exam, and imaging should be done every 2 months during the first year, every 3 or 4 months during years 2 and 3, every 6 months during years 4 and 5, and annually thereafter. Routine use of PET scan for surveillance is not recommended. Any new pulmonary nodule should prompt evaluation for a second primary lung malignancy. Finally, smoking cessation counseling is an integral part of management of any patient with SCLC and should be included with every clinic visit.
CONCLUSION
SCLC is a heterogeneous and genetically complex disease with a very high mortality rate. The current standard of care includes concurrent chemoradiation with cisplatin and etoposide for limited-stage SCLC and the combination of platinum and etoposide for extensive SCLC. A number of novel treatment approaches, including immune checkpoint inhibitors and antibody-drug conjugates, have had promising results in early clinical trials. Given the limited treatment options and large unmet need for new treatment options, enrollment in clinical trials is strongly recommended for patients with SCLC.
1. American Cancer Society. Cancer Facts & Figures 2017. American Cancer Society website. https://www.cancer.org/content/dam/cancer-org/research/cancer-facts-and-statistics/annual-cancer-facts-and-figures/2017/cancer-facts-and-figures-2017.pdf. Published 2017. Accessed July 11, 2018.
2. Govindan R, Page N, Morgensztern D, et al. Changing epidemiology of small-cell lung cancer in the United States over the last 30 years: analysis of the surveillance, epidemiologic, and end results database. J Clin Oncol 2006;24:4539–44.
3. Howlader N, Noone AM, Krapcho M, et al. SEER Cancer Statistics Review, 1975-2014. National Cancer Institute website. https://seer.cancer.gov/csr/1975_2014/. Updated April 2, 2018. Accessed July 11, 2018.
4. Varghese AM, Zakowski MF, Yu HA, et al. Small-cell lung cancers in patients who never smoked cigarettes. J Thorac Oncol 2014;9:892–6.
5. Pleasance ED, Stephens PJ, O’Meara S, et al. A small-cell lung cancer genome with complex signatures of tobacco exposure. Nature 2010;463:184–90.
6. Green RA, Humphrey E, Close H, Patno ME. Alkylating agents in bronchogenic carcinoma. Am J Med 1969;46:516–25.
7. National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology for small cell lung cancer (Version 2.2018). www.nccn.org/professionals/physician_gls/pdf/sclc.pdf. Accessed August 12, 2018.
8. National Lung Screening Trial Research Team, Aberle DR, Adams AM, et al. Reduced lung-cancer mortality with low-dose computed tomographic screening. N Engl J Med 2011;365:395–409.
9. Aberle DR, DeMello S, Berg CD, et al. Results of the two incidence screenings in the National Lung Screening Trial. N Engl J Med 2013;369:920–31.
10. Kitajima K, Nakamoto Y, Okizuka H, et al. Accuracy of whole-body FDG-PET/CT for detecting brain metastases from non-central nervous system tumors. Ann Nucl Med 2008;22:595–602.
11. Ordonez NG. Value of thyroid transcription factor-1 immunostaining in distinguishing small cell lung carcinomas from other small cell carcinomas. Am J Surg Pathol 2000;24:1217–23.
12. Karnak D, Beder S, Kayacan O, et al. Neuron-specific enolase and lung cancer. Am J Clin Oncol 2005;28:586–90.
13. Turrisi AT 3rd, Kim K, Blum R, et al. Twice-daily compared with once-daily thoracic radiotherapy in limited small-cell lung cancer treated concurrently with cisplatin and etoposide. N Engl J Med 1999;340:265–71.
14. Evans WK, Shepherd FA, Feld R, et al. VP-16 and cisplatin as first-line therapy for small-cell lung cancer. J Clin Oncol 1985;3:1471–7.
15. Pujol JL, Carestia L, Daurés JP. Is there a case for cisplatin in the treatment of small-cell lung cancer? A meta-analysis of randomized trials of a cisplatin-containing regimen versus a regimen without this alkylating agent. Br J Cancer 2000;83:8–15.
16. Mascaux C, Paesmans M, Berghmans T, et al; European Lung Cancer Working Party (ELCWP). A systematic review of the role of etoposide and cisplatin in the chemotherapy of small cell lung cancer with methodology assessment and meta-analysis. Lung Cancer 2000;30:23–36.
17. Sundstrøm S, Bremnes RM, Kaasa S, et al; Norwegian Lung Cancer Study Group. Cisplatin and etoposide regimen is superior to cyclophosphamide, epirubicin, and vincristine regimen in small-cell lung cancer: results from a randomized phase III trial with 5 years’ follow-up. J Clin Oncol 2002;20:4665–72.
18. Hatfield LA, Huskamp HA, Lamont EB. Survival and toxicity after cisplatin plus etoposide versus carboplatin plus etoposide for extensive-stage small-cell lung cancer in elderly patients. J Oncol Pract 2016;12:666–73.
19. Okamoto H, Watanabe K, Kunikane H, et al. Randomised phase III trial of carboplatin plus etoposide vs split doses of cisplatin plus etoposide in elderly or poor-risk patients with extensive disease small-cell lung cancer: JCOG 9702. Br J Cancer 2007;97:162–9.
20. Skarlos DV, Samantas E, Kosmidis P, et al. Randomized comparison of etoposide-cisplatin vs. etoposide-carboplatin and irradiation in small-cell lung cancer. A Hellenic Co-operative Oncology Group study. Ann Oncol 1994;5:601–7.
21. Rossi A, Di Maio M, Chiodini P, et al. Carboplatin- or cisplatin-based chemotherapy in first-line treatment of small-cell lung cancer: the COCIS meta-analysis of individual patient data J Clin Oncol 2012;30:1692–8.
22. Bishop JF, Raghavan D, Stuart-Harris R, et al. Carboplatin (CBDCA, JM-8) and VP-16-213 in previously untreated patients with small-cell lung cancer. J Clin Oncol 1987;5:1574–8.
23. Takada M, Fukuoka M, Kawahara M, et al. Phase III study of concurrent versus sequential thoracic radiotherapy in combination with cisplatin and etoposide for limited-stage small-cell lung cancer: results of the Japan Clinical Oncology Group Study 9104. J Clin Oncol 2002;20:3054–60.
24. Bunn PA Jr, Crowley J, Kelly K, et al. Chemoradiotherapy with or without granulocyte-macrophage colony-stimulating factor in the treatment of limited-stage small-cell lung cancer: a prospective phase III randomized study of the Southwest Oncology Group. J Clin Oncol 1995;13:1632–41.
25. Pignon JP, Arriagada R, Ihde DC, et al. A meta-analysis of thoracic radiotherapy for small-cell lung cancer. N Engl J Med 1992;327:1618–24.
26. Warde P, Payne D. Does thoracic irradiation improve survival and local control in limited-stage small-cell carcinoma of the lung? A meta-analysis. J Clin Oncol 1992;10:890–5.
27. Murray N, Coy P, Pater JL, et al. Importance of timing for thoracic irradiation in the combined modality treatment of limited-stage small-cell lung cancer. The National Cancer Institute of Canada Clinical Trials Group. J Clin Oncol 1993;11:336–44.
28. De Ruysscher D, Lueza B, Le Péchoux C, et al. Impact of thoracic radiotherapy timing in limited-stage small-cell lung cancer: usefulness of the individual patient data meta-analysis. Ann Oncol 2016;27:1818–28.
29. Faivre-Finn C, Snee M, Ashcroft L, et al. Concurrent once-daily versus twice-daily chemoradiotherapy in patients with limited-stage small-cell lung cancer (CONVERT): an open-label, phase 3, randomised, superiority trial. Lancet Oncol 2017;18:1116–25.
30. Aupérin A, Arriagada R, Pignon JP, et al. Prophylactic cranial irradiation for patients with small-cell lung cancer in complete remission. Prophylactic Cranial Irradiation Overview Collaborative Group. N Engl J Med 1999;341:476–84.
31. Arriagada R, Le Chevalier T, Borie F, et al. Prophylactic cranial irradiation for patients with small-cell lung cancer in complete remission. J Natl Cancer Inst 1995;87:183–90.
32. Le Péchoux C, Dunant A, Senan S, et al; Prophylactic Cranial Irradiation (PCI) Collaborative Group. Standard-dose versus higher-dose prophylactic cranial irradiation (PCI) in patients with limited-stage small-cell lung cancer in complete remission after chemotherapy and thoracic radiotherapy (PCI 99-01, EORTC 22003-08004, RTOG 0212, and IFCT 99-01): a randomised clinical trial. Lancet Oncol 2009;10:467–74.
33. Schneider BJ, Saxena A, Downey RJ. Surgery for early-stage small cell lung cancer. J Natl Compr Canc Netw 2011;9:1132–9.
34. Inoue M, Nakagawa K, Fujiwara K, et al. Results of preoperative mediastinoscopy for small cell lung cancer. Ann Thorac Surg 2000;70:1620–3.
35. Lim E, Belcher E, Yap YK, et al. The role of surgery in the treatment of limited disease small cell lung cancer: time to reevaluate. J Thorac Oncol 2008;3:1267–71.
36. Inoue M, Miyoshi S, Yasumitsu T, et al. Surgical results for small cell lung cancer based on the new TNM staging system. Thoracic Surgery Study Group of Osaka University, Osaka, Japan. Ann Thorac Surg 2000;70:1615–9.
37. Yang CF, Chan DY, Speicher PJ, et al. Role of adjuvant therapy in a population-based cohort of patients with early-stage small-cell lung cancer. J Clin Oncol 2016;34:1057–64.
38. Shepherd FA, Evans WK, Feld R, et al. Adjuvant chemotherapy following surgical resection for small-cell carcinoma of the lung. J Clin Oncol 1988;6:832–8.
39. Noda K, Nishiwaki Y, Kawahara M, et al; Japan Clinical Oncology Group. Irinotecan plus cisplatin compared with etoposide plus cisplatin for extensive small-cell lung cancer. N Engl J Med 2002;346:85–91.
40. Lara PN Jr, Natale R, Crowley J, et al. Phase III trial of irinotecan/cisplatin compared with etoposide/cisplatin in extensive-stage small-cell lung cancer: clinical and pharmacogenomic results from SWOG S0124. J Clin Oncol 2009;27:2530–5.
41. Chute JP, Chen T, Feigal E, et al. Twenty years of phase III trials for patients with extensive-stage small-cell lung cancer: perceptible progress. J Clin Oncol 1999;17:1794–801.
42. Zhou H, Zeng C, Wei Y, et al. Duration of chemotherapy for small cell lung cancer: a meta-analysis. PloS One 2013;8:e73805.
43. Loehrer PJ Sr, Ansari R, Gonin R, et al. Cisplatin plus etoposide with and without ifosfamide in extensive small-cell lung cancer: a Hoosier Oncology Group study. J Clin Oncol;13:2594–9.
44. Pujol JL, Daurés JP, Riviére A, et al. Etoposide plus cisplatin with or without the combination of 4’-epidoxorubicin plus cyclophosphamide in treatment of extensive small-cell lung cancer: a French Federation of Cancer Institutes multicenter phase III randomized study. J Natl Cancer Inst 2001;93:300–8.
45. Berghmans T, Scherpereel A, Meert AP, et al; European Lung Cancer Working Party (ELCWP). A phase III randomized study comparing a chemotherapy with cisplatin and etoposide to a etoposide regimen without cisplatin for patients with extensive small-cell lung cancer. Front Oncol 2017;7:217.
46. Jalal SI, Lavin P, Lo G, et al. Carboplatin and etoposide with or without palifosfamide in untreated extensive-stage small-cell lung cancer: a Multicenter, Adaptive, Randomized Phase III Study (MATISSE). J Clin Oncol 2017;35:2619–23.
47. Fukuoka M, Furuse K, Saijo N, et al. Randomized trial of cyclophosphamide, doxorubicin, and vincristine versus cisplatin and etoposide versus alternation of these regimens in small-cell lung cancer. J Natl Cancer Inst 1991;83:855–61.
48. Roth BJ, Johnson DH, Einhorn LH, et al. Randomized study of cyclophosphamide, doxorubicin, and vincristine versus etoposide and cisplatin versus alternation of these two regimens in extensive small-cell lung cancer: a phase III trial of the Southeastern Cancer Study Group. J Clin Oncol 1992;10:282–91.
49. Miles DW, Earl HM, Souhami RL, et al. Intensive weekly chemotherapy for good-prognosis patients with small-cell lung cancer. J Clin Oncol 1991;9:280–5.
50. Petrioli R, Roviello G, Laera L, et al. Cisplatin, etoposide, and bevacizumab regimen followed by oral etoposide and bevacizumab maintenance treatment in patients with extensive-stage small cell lung cancer: a single-institution experience. Clin Lung Cancer 2015;16:e229–34.
51. Spigel DR, Greco FA, Zubkus JD, et al. Phase II trial of irinotecan, carboplatin, and bevacizumab in the treatment of patients with extensive-stage small-cell lung cancer. J Thorac Oncol 2009;4:1555–60.
52. Spigel DR, Townley PM, Waterhouse DM, et al. Randomized phase II study of bevacizumab in combination with chemotherapy in previously untreated extensive-stage small-cell lung cancer: results from the SALUTE trial. J Clin Oncol 2011;29:2215–22.
53. Horn L, Dahlberg SE, Sandler AB, et al. Phase II study of cisplatin plus etoposide and bevacizumab for previously untreated, extensive-stage small-cell lung cancer: Eastern Cooperative Oncology Group Study E3501. J Clin Oncol 2009;27:6006–11.
54. Tiseo M, Boni L, Ambrosio F, et al. Italian, multicenter, phase III, randomized study of cisplatin plus etoposide with or without bevacizumab as first-line treatment in extensive-disease small-cell lung cancer: the GOIRC-AIFA FARM6PMFJM trial. J Clin Oncol 2017;35:1281–7.
55. Pujol JL, Lavole A, Quoix E, et al. Randomized phase II-III study of bevacizumab in combination with chemotherapy in previously untreated extensive small-cell lung cancer: results from the IFCT-0802 trial. Ann Oncol 2015;26:908–14.
56. Gadgeel SM, Ventimiglia J, Kalemkerian GP, et al. Phase II study of maintenance pembrolizumab (pembro) in extensive stage small cell lung cancer (ES-SCLC) patients (pts) [abstract]. J Clin Oncol 2017;35(15_suppl):8504.
57. Reck M, Luft A, Szczesna A, et al. Phase III randomized trial of ipilimumab plus etoposide and platinum versus placebo plus etoposide and platinum in extensive-stage small-cell lung cancer. J Clin Oncol 2016;34:3740–8.
58. Chung HC, Lopez-Martin JA, Kao SC, et al. Phase 2 study of pembrolizumab in advanced small-cell lung cancer (SCLC): KEYNOTE-158 [abstract]. J Clin Oncol 2018;36(suppl):8506.
59. Owonikoko TK, Behera M, Chen Z, et al. A systematic analysis of efficacy of second-line chemotherapy in sensitive and refractory small-cell lung cancer. J Thorac Oncol 2012;7:866–72.
60. Postmus PE, Berendsen HH, van Zandwijk N, et al. Retreatment with the induction regimen in small cell lung cancer relapsing after an initial response to short term chemotherapy. Eur J Cancer Clin Oncol 1987;23:1409–11.
61. von Pawel J, Schiller JH, Shepherd FA, et al. Topotecan versus cyclophosphamide, doxorubicin, and vincristine for the treatment of recurrent small-cell lung cancer. J Clin Oncol 1999;17:658–667.
62. O’Brien ME, Ciuleanu TE, Tsekov H, et al. Phase III trial comparing supportive care alone with supportive care with oral topotecan in patients with relapsed small-cell lung cancer. J Clin Oncol 2006;24:5441–7.
63. Eckardt JR, von Pawel J, Pujol JL, et al. Phase III study of oral compared with intravenous topotecan as second-line therapy in small-cell lung cancer. J Clin Oncol 2007;25:2086–92.
64. Masuda N, Fukuoka M, Kusunoki Y, et al. CPT-11: a new derivative of camptothecin for the treatment of refractory or relapsed small-cell lung cancer. J Clin Oncol 1992;10:1225–9.
65. Smit EF, Fokkema E, Biesma B, et al. A phase II study of paclitaxel in heavily pretreated patients with small-cell lung cancer. Br J Cancer 1998;77:347–51.
66. Yamamoto N, Tsurutani J, Yoshimura N, et al. Phase II study of weekly paclitaxel for relapsed and refractory small cell lung cancer. Anticancer Res 2006;26:777–81.
67. Smyth JF, Smith IE, Sessa C, et al. Activity of docetaxel (Taxotere) in small cell lung cancer. Eur J Cancer 1994;30A:1058–60.
68. Pietanza MC, Kadota K, Huberman K, et al. Phase II trial of temozolomide with relapsed sensitive or refractory small cell lung cancer, with assessment of methylguanine-DNA methyltransferase as a potential biomarker. Clin Cancer Res 2012;18:1138–45.
69. Zauderer MG, Drilon A, Kadota K, et al. Trial of a 5-day dosing regimen of temozolomide in patients with relapsed small cell lung cancers with assessment of methylguanine-DNA methyltransferase. Lung Cancer 2014;86:237–40.
70. Jassem J, Karnicka-Mlodkowska H, van Pottelsberghe C, et al. Phase II study of vinorelbine (Navelbine) in previously treated small cell lung cancer patients. Eur J Cancer 1993;29A:1720–2.
71. Furuse K, Kuboa K, Kawahara M, et al. Phase II study of vinorelbine in heavily previously treated small cell lung cancer. Oncology 1996;53:169–72.
72. Einhorn LH, Pennington K, McClean J. Phase II trial of daily oral VP-16 in refractory small cell lung cancer. Semin Oncol 1990;17:32–5.
73. Johnson DH, Greco FA, Strupp J, et al. Prolonged administration of oral etoposide in patients with relapsed or refractory small-cell lung cancer: a phase II trial. J Clin Oncol 1990;8:1613–7.
74. Van der Lee I, Smit EF, van Putten JW, et al. Single-agent gemcitabine in patients with resistant small-cell lung cancer. Ann Oncol 2001;12:557–61.
75. Masters GA, Declerck L, Blanke C, et al. Phase II trial of gemcitabine in refractory or relapsed small-cell lung cancer. J Clin Oncol 2003;21:1550–5.
76. von Pawel J, Schiller JH, Shepherd FA, et al. Topotecan versus cyclophosphamide, doxorubicin, and vincristine for the treatment of recurrent small-cell lung cancer. J Clin Oncol 1999;17:658–67.
77. Lammers PE, Shyr Y, Li CI, et al. Phase II study of bendamustine in relapsed chemotherapy sensitive or resistant small-cell lung cancer. J Thorac Oncol 2014;9:559–62.
78. Hellmann MD, Ott PA, Zugazagoitia J, et al. Nivolumab (nivo) ± ipilimumab (ipi) in advanced small-cell lung cancer (SCLC): First report of a randomized expansion cohort from CheckMate 032 [abstract]. J Clin Oncol 2017;35(15_suppl):8503.
79. Antonia SJ, López-Martin JA, Bendell J, et al. Nivolumab alone and nivolumab plus ipilimumab in recurrent small-cell lung cancer (CheckMate 032): a multicentre, open-label, phase 1/2 trial. Lancet Oncol 2016;17:883–95.
80. Ott PA, Elez E, Hiret S, et al. Pembrolizumab in patients with extensive-stage small-cell lung cancer: results from the Phase Ib KEYNOTE-028 study. J Clin Oncol 2017;35:3823–9.
81. Slotman B, Faivre-Finn C, Kramer G, et al. Prophylactic cranial irradiation in extensive small-cell lung cancer. N Engl J Med 2007;357:664–72.
82. Takahashi T, Yamanaka T, Seto T, et al. Prophylactic cranial irradiation versus observation in patients with extensive-disease small-cell lung cancer: a multicentre, randomised, open-label, phase 3 trial. Lancet Oncol 2017;18:663–71.
83. Slotman BJ, Mauer ME, Bottomley A, et al. Prophylactic cranial irradiation in extensive disease small-cell lung cancer: short-term health-related quality of life and patient reported symptoms: results of an international Phase III randomized controlled trial by the EORTC Radiation Oncology and Lung Cancer Groups. J Clin Oncol 2009;27:78–84.
84. Brown PD, Pugh S, Laack NN, et al; Radiation Therapy Oncology Group (RTOG). Memantine for the prevention of cognitive dysfunction in patients receiving whole-brain radiotherapy: a randomized, double-blind, placebo-controlled trial. Neuro Oncol 2013;15:1429–37.
85. Jeremic B, Shibamoto Y, Nikolic N, et al. Role of radiation therapy in the combined-modality treatment of patients with extensive disease small-cell lung cancer: a randomized study. J Clin Oncol 1999;17:2092–9.
86. Slotman BJ, van Tinteren H, Praag JO, et al. Use of thoracic radiotherapy for extensive stage small-cell lung cancer: a phase 3 randomised controlled trial. Lancet 2015;385:36–42.
87. Slotman BJ, van Tinteren H, Praag JO, et al. Radiotherapy for extensive stage small-cell lung cancer - authors’ reply. Lancet 2015;385:1292–3.
88. Owonikoko TK, Dahlberg SE, Khan SA, et al. A phase 1 safety study of veliparib combined with cisplatin and etoposide in extensive stage small cell lung cancer: A trial of the ECOG-ACRIN Cancer Research Group (E2511). Lung Cancer 2015;89:66–70.
89. Mamdani H, Induru R, Jalal SI. Novel therapies in small cell lung cancer. Transl Lung Cancer Res 2015;4:533–44.
90. Rudin CM, Pietanza MC, Bauer TM, et al. Rovalpituzumab tesirine, a DLL3-targeted antibody-drug conjugate, in recurrent small-cell lung cancer: a first-in-human, first-in-class, open-label, phase 1 study. Lancet Oncol 2017;18:42–51.
1. American Cancer Society. Cancer Facts & Figures 2017. American Cancer Society website. https://www.cancer.org/content/dam/cancer-org/research/cancer-facts-and-statistics/annual-cancer-facts-and-figures/2017/cancer-facts-and-figures-2017.pdf. Published 2017. Accessed July 11, 2018.
2. Govindan R, Page N, Morgensztern D, et al. Changing epidemiology of small-cell lung cancer in the United States over the last 30 years: analysis of the surveillance, epidemiologic, and end results database. J Clin Oncol 2006;24:4539–44.
3. Howlader N, Noone AM, Krapcho M, et al. SEER Cancer Statistics Review, 1975-2014. National Cancer Institute website. https://seer.cancer.gov/csr/1975_2014/. Updated April 2, 2018. Accessed July 11, 2018.
4. Varghese AM, Zakowski MF, Yu HA, et al. Small-cell lung cancers in patients who never smoked cigarettes. J Thorac Oncol 2014;9:892–6.
5. Pleasance ED, Stephens PJ, O’Meara S, et al. A small-cell lung cancer genome with complex signatures of tobacco exposure. Nature 2010;463:184–90.
6. Green RA, Humphrey E, Close H, Patno ME. Alkylating agents in bronchogenic carcinoma. Am J Med 1969;46:516–25.
7. National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology for small cell lung cancer (Version 2.2018). www.nccn.org/professionals/physician_gls/pdf/sclc.pdf. Accessed August 12, 2018.
8. National Lung Screening Trial Research Team, Aberle DR, Adams AM, et al. Reduced lung-cancer mortality with low-dose computed tomographic screening. N Engl J Med 2011;365:395–409.
9. Aberle DR, DeMello S, Berg CD, et al. Results of the two incidence screenings in the National Lung Screening Trial. N Engl J Med 2013;369:920–31.
10. Kitajima K, Nakamoto Y, Okizuka H, et al. Accuracy of whole-body FDG-PET/CT for detecting brain metastases from non-central nervous system tumors. Ann Nucl Med 2008;22:595–602.
11. Ordonez NG. Value of thyroid transcription factor-1 immunostaining in distinguishing small cell lung carcinomas from other small cell carcinomas. Am J Surg Pathol 2000;24:1217–23.
12. Karnak D, Beder S, Kayacan O, et al. Neuron-specific enolase and lung cancer. Am J Clin Oncol 2005;28:586–90.
13. Turrisi AT 3rd, Kim K, Blum R, et al. Twice-daily compared with once-daily thoracic radiotherapy in limited small-cell lung cancer treated concurrently with cisplatin and etoposide. N Engl J Med 1999;340:265–71.
14. Evans WK, Shepherd FA, Feld R, et al. VP-16 and cisplatin as first-line therapy for small-cell lung cancer. J Clin Oncol 1985;3:1471–7.
15. Pujol JL, Carestia L, Daurés JP. Is there a case for cisplatin in the treatment of small-cell lung cancer? A meta-analysis of randomized trials of a cisplatin-containing regimen versus a regimen without this alkylating agent. Br J Cancer 2000;83:8–15.
16. Mascaux C, Paesmans M, Berghmans T, et al; European Lung Cancer Working Party (ELCWP). A systematic review of the role of etoposide and cisplatin in the chemotherapy of small cell lung cancer with methodology assessment and meta-analysis. Lung Cancer 2000;30:23–36.
17. Sundstrøm S, Bremnes RM, Kaasa S, et al; Norwegian Lung Cancer Study Group. Cisplatin and etoposide regimen is superior to cyclophosphamide, epirubicin, and vincristine regimen in small-cell lung cancer: results from a randomized phase III trial with 5 years’ follow-up. J Clin Oncol 2002;20:4665–72.
18. Hatfield LA, Huskamp HA, Lamont EB. Survival and toxicity after cisplatin plus etoposide versus carboplatin plus etoposide for extensive-stage small-cell lung cancer in elderly patients. J Oncol Pract 2016;12:666–73.
19. Okamoto H, Watanabe K, Kunikane H, et al. Randomised phase III trial of carboplatin plus etoposide vs split doses of cisplatin plus etoposide in elderly or poor-risk patients with extensive disease small-cell lung cancer: JCOG 9702. Br J Cancer 2007;97:162–9.
20. Skarlos DV, Samantas E, Kosmidis P, et al. Randomized comparison of etoposide-cisplatin vs. etoposide-carboplatin and irradiation in small-cell lung cancer. A Hellenic Co-operative Oncology Group study. Ann Oncol 1994;5:601–7.
21. Rossi A, Di Maio M, Chiodini P, et al. Carboplatin- or cisplatin-based chemotherapy in first-line treatment of small-cell lung cancer: the COCIS meta-analysis of individual patient data J Clin Oncol 2012;30:1692–8.
22. Bishop JF, Raghavan D, Stuart-Harris R, et al. Carboplatin (CBDCA, JM-8) and VP-16-213 in previously untreated patients with small-cell lung cancer. J Clin Oncol 1987;5:1574–8.
23. Takada M, Fukuoka M, Kawahara M, et al. Phase III study of concurrent versus sequential thoracic radiotherapy in combination with cisplatin and etoposide for limited-stage small-cell lung cancer: results of the Japan Clinical Oncology Group Study 9104. J Clin Oncol 2002;20:3054–60.
24. Bunn PA Jr, Crowley J, Kelly K, et al. Chemoradiotherapy with or without granulocyte-macrophage colony-stimulating factor in the treatment of limited-stage small-cell lung cancer: a prospective phase III randomized study of the Southwest Oncology Group. J Clin Oncol 1995;13:1632–41.
25. Pignon JP, Arriagada R, Ihde DC, et al. A meta-analysis of thoracic radiotherapy for small-cell lung cancer. N Engl J Med 1992;327:1618–24.
26. Warde P, Payne D. Does thoracic irradiation improve survival and local control in limited-stage small-cell carcinoma of the lung? A meta-analysis. J Clin Oncol 1992;10:890–5.
27. Murray N, Coy P, Pater JL, et al. Importance of timing for thoracic irradiation in the combined modality treatment of limited-stage small-cell lung cancer. The National Cancer Institute of Canada Clinical Trials Group. J Clin Oncol 1993;11:336–44.
28. De Ruysscher D, Lueza B, Le Péchoux C, et al. Impact of thoracic radiotherapy timing in limited-stage small-cell lung cancer: usefulness of the individual patient data meta-analysis. Ann Oncol 2016;27:1818–28.
29. Faivre-Finn C, Snee M, Ashcroft L, et al. Concurrent once-daily versus twice-daily chemoradiotherapy in patients with limited-stage small-cell lung cancer (CONVERT): an open-label, phase 3, randomised, superiority trial. Lancet Oncol 2017;18:1116–25.
30. Aupérin A, Arriagada R, Pignon JP, et al. Prophylactic cranial irradiation for patients with small-cell lung cancer in complete remission. Prophylactic Cranial Irradiation Overview Collaborative Group. N Engl J Med 1999;341:476–84.
31. Arriagada R, Le Chevalier T, Borie F, et al. Prophylactic cranial irradiation for patients with small-cell lung cancer in complete remission. J Natl Cancer Inst 1995;87:183–90.
32. Le Péchoux C, Dunant A, Senan S, et al; Prophylactic Cranial Irradiation (PCI) Collaborative Group. Standard-dose versus higher-dose prophylactic cranial irradiation (PCI) in patients with limited-stage small-cell lung cancer in complete remission after chemotherapy and thoracic radiotherapy (PCI 99-01, EORTC 22003-08004, RTOG 0212, and IFCT 99-01): a randomised clinical trial. Lancet Oncol 2009;10:467–74.
33. Schneider BJ, Saxena A, Downey RJ. Surgery for early-stage small cell lung cancer. J Natl Compr Canc Netw 2011;9:1132–9.
34. Inoue M, Nakagawa K, Fujiwara K, et al. Results of preoperative mediastinoscopy for small cell lung cancer. Ann Thorac Surg 2000;70:1620–3.
35. Lim E, Belcher E, Yap YK, et al. The role of surgery in the treatment of limited disease small cell lung cancer: time to reevaluate. J Thorac Oncol 2008;3:1267–71.
36. Inoue M, Miyoshi S, Yasumitsu T, et al. Surgical results for small cell lung cancer based on the new TNM staging system. Thoracic Surgery Study Group of Osaka University, Osaka, Japan. Ann Thorac Surg 2000;70:1615–9.
37. Yang CF, Chan DY, Speicher PJ, et al. Role of adjuvant therapy in a population-based cohort of patients with early-stage small-cell lung cancer. J Clin Oncol 2016;34:1057–64.
38. Shepherd FA, Evans WK, Feld R, et al. Adjuvant chemotherapy following surgical resection for small-cell carcinoma of the lung. J Clin Oncol 1988;6:832–8.
39. Noda K, Nishiwaki Y, Kawahara M, et al; Japan Clinical Oncology Group. Irinotecan plus cisplatin compared with etoposide plus cisplatin for extensive small-cell lung cancer. N Engl J Med 2002;346:85–91.
40. Lara PN Jr, Natale R, Crowley J, et al. Phase III trial of irinotecan/cisplatin compared with etoposide/cisplatin in extensive-stage small-cell lung cancer: clinical and pharmacogenomic results from SWOG S0124. J Clin Oncol 2009;27:2530–5.
41. Chute JP, Chen T, Feigal E, et al. Twenty years of phase III trials for patients with extensive-stage small-cell lung cancer: perceptible progress. J Clin Oncol 1999;17:1794–801.
42. Zhou H, Zeng C, Wei Y, et al. Duration of chemotherapy for small cell lung cancer: a meta-analysis. PloS One 2013;8:e73805.
43. Loehrer PJ Sr, Ansari R, Gonin R, et al. Cisplatin plus etoposide with and without ifosfamide in extensive small-cell lung cancer: a Hoosier Oncology Group study. J Clin Oncol;13:2594–9.
44. Pujol JL, Daurés JP, Riviére A, et al. Etoposide plus cisplatin with or without the combination of 4’-epidoxorubicin plus cyclophosphamide in treatment of extensive small-cell lung cancer: a French Federation of Cancer Institutes multicenter phase III randomized study. J Natl Cancer Inst 2001;93:300–8.
45. Berghmans T, Scherpereel A, Meert AP, et al; European Lung Cancer Working Party (ELCWP). A phase III randomized study comparing a chemotherapy with cisplatin and etoposide to a etoposide regimen without cisplatin for patients with extensive small-cell lung cancer. Front Oncol 2017;7:217.
46. Jalal SI, Lavin P, Lo G, et al. Carboplatin and etoposide with or without palifosfamide in untreated extensive-stage small-cell lung cancer: a Multicenter, Adaptive, Randomized Phase III Study (MATISSE). J Clin Oncol 2017;35:2619–23.
47. Fukuoka M, Furuse K, Saijo N, et al. Randomized trial of cyclophosphamide, doxorubicin, and vincristine versus cisplatin and etoposide versus alternation of these regimens in small-cell lung cancer. J Natl Cancer Inst 1991;83:855–61.
48. Roth BJ, Johnson DH, Einhorn LH, et al. Randomized study of cyclophosphamide, doxorubicin, and vincristine versus etoposide and cisplatin versus alternation of these two regimens in extensive small-cell lung cancer: a phase III trial of the Southeastern Cancer Study Group. J Clin Oncol 1992;10:282–91.
49. Miles DW, Earl HM, Souhami RL, et al. Intensive weekly chemotherapy for good-prognosis patients with small-cell lung cancer. J Clin Oncol 1991;9:280–5.
50. Petrioli R, Roviello G, Laera L, et al. Cisplatin, etoposide, and bevacizumab regimen followed by oral etoposide and bevacizumab maintenance treatment in patients with extensive-stage small cell lung cancer: a single-institution experience. Clin Lung Cancer 2015;16:e229–34.
51. Spigel DR, Greco FA, Zubkus JD, et al. Phase II trial of irinotecan, carboplatin, and bevacizumab in the treatment of patients with extensive-stage small-cell lung cancer. J Thorac Oncol 2009;4:1555–60.
52. Spigel DR, Townley PM, Waterhouse DM, et al. Randomized phase II study of bevacizumab in combination with chemotherapy in previously untreated extensive-stage small-cell lung cancer: results from the SALUTE trial. J Clin Oncol 2011;29:2215–22.
53. Horn L, Dahlberg SE, Sandler AB, et al. Phase II study of cisplatin plus etoposide and bevacizumab for previously untreated, extensive-stage small-cell lung cancer: Eastern Cooperative Oncology Group Study E3501. J Clin Oncol 2009;27:6006–11.
54. Tiseo M, Boni L, Ambrosio F, et al. Italian, multicenter, phase III, randomized study of cisplatin plus etoposide with or without bevacizumab as first-line treatment in extensive-disease small-cell lung cancer: the GOIRC-AIFA FARM6PMFJM trial. J Clin Oncol 2017;35:1281–7.
55. Pujol JL, Lavole A, Quoix E, et al. Randomized phase II-III study of bevacizumab in combination with chemotherapy in previously untreated extensive small-cell lung cancer: results from the IFCT-0802 trial. Ann Oncol 2015;26:908–14.
56. Gadgeel SM, Ventimiglia J, Kalemkerian GP, et al. Phase II study of maintenance pembrolizumab (pembro) in extensive stage small cell lung cancer (ES-SCLC) patients (pts) [abstract]. J Clin Oncol 2017;35(15_suppl):8504.
57. Reck M, Luft A, Szczesna A, et al. Phase III randomized trial of ipilimumab plus etoposide and platinum versus placebo plus etoposide and platinum in extensive-stage small-cell lung cancer. J Clin Oncol 2016;34:3740–8.
58. Chung HC, Lopez-Martin JA, Kao SC, et al. Phase 2 study of pembrolizumab in advanced small-cell lung cancer (SCLC): KEYNOTE-158 [abstract]. J Clin Oncol 2018;36(suppl):8506.
59. Owonikoko TK, Behera M, Chen Z, et al. A systematic analysis of efficacy of second-line chemotherapy in sensitive and refractory small-cell lung cancer. J Thorac Oncol 2012;7:866–72.
60. Postmus PE, Berendsen HH, van Zandwijk N, et al. Retreatment with the induction regimen in small cell lung cancer relapsing after an initial response to short term chemotherapy. Eur J Cancer Clin Oncol 1987;23:1409–11.
61. von Pawel J, Schiller JH, Shepherd FA, et al. Topotecan versus cyclophosphamide, doxorubicin, and vincristine for the treatment of recurrent small-cell lung cancer. J Clin Oncol 1999;17:658–667.
62. O’Brien ME, Ciuleanu TE, Tsekov H, et al. Phase III trial comparing supportive care alone with supportive care with oral topotecan in patients with relapsed small-cell lung cancer. J Clin Oncol 2006;24:5441–7.
63. Eckardt JR, von Pawel J, Pujol JL, et al. Phase III study of oral compared with intravenous topotecan as second-line therapy in small-cell lung cancer. J Clin Oncol 2007;25:2086–92.
64. Masuda N, Fukuoka M, Kusunoki Y, et al. CPT-11: a new derivative of camptothecin for the treatment of refractory or relapsed small-cell lung cancer. J Clin Oncol 1992;10:1225–9.
65. Smit EF, Fokkema E, Biesma B, et al. A phase II study of paclitaxel in heavily pretreated patients with small-cell lung cancer. Br J Cancer 1998;77:347–51.
66. Yamamoto N, Tsurutani J, Yoshimura N, et al. Phase II study of weekly paclitaxel for relapsed and refractory small cell lung cancer. Anticancer Res 2006;26:777–81.
67. Smyth JF, Smith IE, Sessa C, et al. Activity of docetaxel (Taxotere) in small cell lung cancer. Eur J Cancer 1994;30A:1058–60.
68. Pietanza MC, Kadota K, Huberman K, et al. Phase II trial of temozolomide with relapsed sensitive or refractory small cell lung cancer, with assessment of methylguanine-DNA methyltransferase as a potential biomarker. Clin Cancer Res 2012;18:1138–45.
69. Zauderer MG, Drilon A, Kadota K, et al. Trial of a 5-day dosing regimen of temozolomide in patients with relapsed small cell lung cancers with assessment of methylguanine-DNA methyltransferase. Lung Cancer 2014;86:237–40.
70. Jassem J, Karnicka-Mlodkowska H, van Pottelsberghe C, et al. Phase II study of vinorelbine (Navelbine) in previously treated small cell lung cancer patients. Eur J Cancer 1993;29A:1720–2.
71. Furuse K, Kuboa K, Kawahara M, et al. Phase II study of vinorelbine in heavily previously treated small cell lung cancer. Oncology 1996;53:169–72.
72. Einhorn LH, Pennington K, McClean J. Phase II trial of daily oral VP-16 in refractory small cell lung cancer. Semin Oncol 1990;17:32–5.
73. Johnson DH, Greco FA, Strupp J, et al. Prolonged administration of oral etoposide in patients with relapsed or refractory small-cell lung cancer: a phase II trial. J Clin Oncol 1990;8:1613–7.
74. Van der Lee I, Smit EF, van Putten JW, et al. Single-agent gemcitabine in patients with resistant small-cell lung cancer. Ann Oncol 2001;12:557–61.
75. Masters GA, Declerck L, Blanke C, et al. Phase II trial of gemcitabine in refractory or relapsed small-cell lung cancer. J Clin Oncol 2003;21:1550–5.
76. von Pawel J, Schiller JH, Shepherd FA, et al. Topotecan versus cyclophosphamide, doxorubicin, and vincristine for the treatment of recurrent small-cell lung cancer. J Clin Oncol 1999;17:658–67.
77. Lammers PE, Shyr Y, Li CI, et al. Phase II study of bendamustine in relapsed chemotherapy sensitive or resistant small-cell lung cancer. J Thorac Oncol 2014;9:559–62.
78. Hellmann MD, Ott PA, Zugazagoitia J, et al. Nivolumab (nivo) ± ipilimumab (ipi) in advanced small-cell lung cancer (SCLC): First report of a randomized expansion cohort from CheckMate 032 [abstract]. J Clin Oncol 2017;35(15_suppl):8503.
79. Antonia SJ, López-Martin JA, Bendell J, et al. Nivolumab alone and nivolumab plus ipilimumab in recurrent small-cell lung cancer (CheckMate 032): a multicentre, open-label, phase 1/2 trial. Lancet Oncol 2016;17:883–95.
80. Ott PA, Elez E, Hiret S, et al. Pembrolizumab in patients with extensive-stage small-cell lung cancer: results from the Phase Ib KEYNOTE-028 study. J Clin Oncol 2017;35:3823–9.
81. Slotman B, Faivre-Finn C, Kramer G, et al. Prophylactic cranial irradiation in extensive small-cell lung cancer. N Engl J Med 2007;357:664–72.
82. Takahashi T, Yamanaka T, Seto T, et al. Prophylactic cranial irradiation versus observation in patients with extensive-disease small-cell lung cancer: a multicentre, randomised, open-label, phase 3 trial. Lancet Oncol 2017;18:663–71.
83. Slotman BJ, Mauer ME, Bottomley A, et al. Prophylactic cranial irradiation in extensive disease small-cell lung cancer: short-term health-related quality of life and patient reported symptoms: results of an international Phase III randomized controlled trial by the EORTC Radiation Oncology and Lung Cancer Groups. J Clin Oncol 2009;27:78–84.
84. Brown PD, Pugh S, Laack NN, et al; Radiation Therapy Oncology Group (RTOG). Memantine for the prevention of cognitive dysfunction in patients receiving whole-brain radiotherapy: a randomized, double-blind, placebo-controlled trial. Neuro Oncol 2013;15:1429–37.
85. Jeremic B, Shibamoto Y, Nikolic N, et al. Role of radiation therapy in the combined-modality treatment of patients with extensive disease small-cell lung cancer: a randomized study. J Clin Oncol 1999;17:2092–9.
86. Slotman BJ, van Tinteren H, Praag JO, et al. Use of thoracic radiotherapy for extensive stage small-cell lung cancer: a phase 3 randomised controlled trial. Lancet 2015;385:36–42.
87. Slotman BJ, van Tinteren H, Praag JO, et al. Radiotherapy for extensive stage small-cell lung cancer - authors’ reply. Lancet 2015;385:1292–3.
88. Owonikoko TK, Dahlberg SE, Khan SA, et al. A phase 1 safety study of veliparib combined with cisplatin and etoposide in extensive stage small cell lung cancer: A trial of the ECOG-ACRIN Cancer Research Group (E2511). Lung Cancer 2015;89:66–70.
89. Mamdani H, Induru R, Jalal SI. Novel therapies in small cell lung cancer. Transl Lung Cancer Res 2015;4:533–44.
90. Rudin CM, Pietanza MC, Bauer TM, et al. Rovalpituzumab tesirine, a DLL3-targeted antibody-drug conjugate, in recurrent small-cell lung cancer: a first-in-human, first-in-class, open-label, phase 1 study. Lancet Oncol 2017;18:42–51.
Management of Metastatic Gastric Cancer
INTRODUCTION
According to the Surveillance, Epidemiology and End Results database, in 2017 there were 28,000 new cases of gastric cancer, accounting for 1.8% of all malignancies in the United States, and an estimated 10,960 gastric cancer–related deaths.1 Worldwide, gastric cancer is the fifth most common malignancy and the third most common cause of death from any cancer.2 The incidence of gastric cancer varies significantly by geographic region, with countries in Eastern Asia (China, Japan), Eastern Europe, and Central and South America accounting for 50% of all new cases.3 Although the incidence of gastric cancer has declined in recent years, this decrease has not been observed consistently across all nations.2 In particular, the incidence of gastric cancers arising from the cardia has been increasing, which is perhaps due to a higher prevalence of obesity in Western societies.4
In this article, we review key aspects of management of metastatic gastric cancer, including selection of first- and second-line therapy, and discuss targeted agents and upcoming clinical trials.
EPIDEMIOLOGY AND RISK FACTORS
Chronic infection with Helicobacter pylori, a gram-negative bacterium, is a strong etiological factor for the development of gastric cancer, contributing to up to 70% of cases.2 The pathogen can colonize the gastric mucosa, leading to chronic inflammation. Although most patients remain asymptomatic, 1% to 3% develop gastric cancer and another 0.1% develop mucosa-associated lymphoid tissue lymphoma.5 H. pylori infection is more commonly associated with cancer of the gastric body than with cancer of the gastroesophageal junction (GEJ). The increased burden of gastric cancer in countries in Eastern Asia, Latin America, and Eastern Europe has been correlated to the prevalence of chronic H. pylori infection in these areas.
Carcinogenesis secondary to H. pylori infection may occur via several mechanisms. First, H. pylori can release virulence factors, such as cytotoxin-associated gene A, vacuolating cytotoxin, and outer membrane proteins, into the cytosol of host cells, leading to changes in patterns of cell proliferation and apoptosis.6 These virulence factors can modulate the host immune system, attenuating it to promote dysplasia. In addition, continued recognition of these factors by the immune system leads to a persistent inflammatory response, with the release of cytokines such as interleukin (IL) -1β, IL-6, and IL-8. This leads to chronic mucosal damage, further promoting dysplasia with eventual transformation into adenocarcinoma.7 In Japan and Korea, where screening for H. pylori infection is routinely performed, there have been improvements in overall survival (OS) rates for gastric cancer, with 5-year OS rates of 70%.8 The International Agency for Research on Cancer recommends further research into population-based screening and treatment programs for patients with chronic H. pylori infection. However, despite this recommendation, optimal screening strategies are not clearly defined.9
Other risk factors for the development of gastric cancer include chronic gastroesophageal reflux disease; smoking; alcohol use; exposure to radiation; diets high in fats, salt, and smoked items and low in fruits and vegetables; obesity; and exposure to chemotherapeutic agents such as procarbazine.10 Another pathogen suspected, but not proven, to be associated with increased risk for gastric cancer is the Epstein-Barr virus, a human herpesvirus found in 80% of all gastric carcinomas with lymphoid features.11 In addition, whether the use of medications such as statins and nonsteroidal anti-inflammatory drugs confers a decreased risk of gastric cancers remains unclear.10
EVALUATION
CASE PRESENTATION
A 55-year-old Caucasian man with a history of type 2 diabetes mellitus presents to the gastrointestinal (GI) clinic with a 6-month history of dysphagia. The dysphagia is worsened with ingestion of solids, particularly towards the end of the day. He states that the food often gets “stuck in the middle of the chest.” The patient denies any nausea or emesis but notes that he has a poor appetite. He reports having worsening mid-epigastric abdominal pain that is non-radiating, dull in character, and 6/10 in intensity. He also reports a 10-lb weight loss over the past 2 months. He has no previous history of reflux, chest pain, dyspnea, or cough. Review of systems is otherwise benign. Physical exam is within normal limits.
• Which tests should be conducted when gastric cancer is suspected?
Persistent epigastric abdominal pain and weight loss are the most common early symptoms of gastric cancer. Nausea, early satiety, dysphagia, and occult GI bleeding can be other presenting signs. Patients presenting with alarm symptoms of nausea, emesis, early satiety, abdominal pain, or weight loss should be fully evaluated with upper GI endoscopy. Early diagnosis of gastric cancer is essential in obtaining a curative resection. However, at least 40% of patients present with de novo metastatic disease at the time of initial diagnosis.12 Gastric cancer spreads by direct extension through the gastric wall, with the liver, peritoneum, and regional lymph nodes being the most common sites of metastatic deposits.13 Classically, Virchow’s node, the left supraclavicular lymph node, is involved with metastatic gastric cancer. Involvement of the left axillary lymph node (Irish node) or a periumbilical nodule (Sister Mary Joseph node) may also be observed. Other, less commonly noted sites of metastatic disease include the ovaries, central nervous system, bone, lung, and soft tissues.13
Upper GI endoscopy is the best method for determining tumor location and extent and obtaining a specimen for a definitive tissue diagnosis.14 It is essential to accurately identify the location of the tumor in the stomach and relative to the GEJ. The American Joint Committee on Cancer classification defines tumors involving the GEJ with an epicenter no more than 2 cm into the proximal stomach as esophageal cancers.15 Tumors of the GEJ with their epicenter more than 2 cm into the proximal stomach are defined as gastric cancers. If metastatic disease is suspected, computed tomography (CT) scan of the chest, abdomen, and pelvis with oral and intravenous contrast can be obtained to determine the extent of disease spread. In the absence of any metastatic disease, endoscopic ultrasound (EUS) should be conducted to determine the depth of tumor invasion (T staging) and lymph node status. In the era of targeted therapy, patients with metastatic disease should undergo testing for human epidermal growth factor-2 (HER-2) expression, microsatellite instability (MSI), and programmed death ligand 1 (PD-L1) expression. Patients should be staged according to the TNM staging system.
FIRST-LINE TREATMENT OPTIONS
CASE CONTINUED
The patient undergoes esophagoduodenoscopy (EGD) and is found to have a gastric cardia mass extending into the distal esophagus. EUS also demonstrates multiple abdominal and mediastinal lymph nodes. No gastric outlet obstruction is found. Biopsy shows poorly differentiated invasive adenocarcinoma. Warthin–Starry stain is negative for H. pylori organism. The tumor cells are positive for cytokeratin (CK7), CK19, and mucin-1 gene (MUC1); focally positive for CK20; and negative for MUC2. HER2 testing results are reported as immunohistochemistry (IHC) 3+, consistent with strongly positive HER2 protein expression. Further IHC testing for mismatch repair (MMR) proteins shows intact nuclear expression of MLH1, MSH2, MSH6, and PMS2 protein, consistent with a low probability of MSI-high tumor. The tumor is found to be PD-L1 positive. Imaging reveals abnormal mass-like nodular thickening of the gastric wall, with an infiltrative opacity within the pancreatico-duodenal groove, suspicious for tumor infiltration. Multiple metastatic deposits are noted in the liver, peritoneum, and bilateral lungs. There is extensive gastrohepatic ligament and periportal lymphadenopathy and mild enlargement of the pulmonary hilar lymph nodes. These findings are consistent with stage 4 (T4bN3aM1) gastric cancer. Given these findings, staging laparoscopy is deferred.
• What are the first-line treatment options for patients with metastatic gastric cancer?
Patients with metastatic gastric cancer have a poor prognosis, and management is stratified based on performance status (Figure). In patients with good performance status, systemic chemotherapy is the mainstay of treatment. The goal of therapy is not curative, but rather treatment focuses on palliation of symptoms arising from tumor spread. Given this treatment goal, there has been considerable interest in clarifying the utility of chemotherapy as opposed to best supportive care. In a recent Cochrane review of 64 randomized control trials involving 11,698 patients, chemotherapy was found to improve OS by 6.7 months as compared to best supportive care (hazard ratio [HR] 0.3 [95% confidence interval {CI} 0.24 to 0.55]).16 Five classes of cytotoxic chemotherapeutic agents have demonstrated activity in gastric cancer. These include fluoropyrimidine (either infusional fluorouracil or capecitabine), platinum agents (cisplatin or oxaliplatin), taxanes (docetaxel or paclitaxel), anthracyclines (epirubicin), and irinotecan.13 Treatment options are further divided based on whether the patient has HER2-overexpressing or non-expressing malignancy.
HER2-NEGATIVE DISEASE
For patients with HER2-negative disease, National Comprehensive Cancer Network (NCCN) guidelines recommend using 2-drug combination regimens rather than 3 drugs, given concern for increased toxicity with 3-drug regimens.17 For patients with a performance status of 0 to 1, utilization of a 3-drug regimen is a reasonable alternative. The combination of a fluoropyrimidine with a platinum agent is considered the standard of care, with regimens such as fluorouracil, leucovorin, and oxaliplatin (FOLFOX) being commonly used.
Epirubicin-containing regimens have also been extensively studied in advanced gastric cancer. In a study of 274 previously untreated patients with GEJ cancers, the combination of epirubicin, cisplatin, and fluorouracil (ECF) was compared to fluorouracil, doxorubicin, and methotrexate (FAMTX). There was an OS benefit favoring ECF (8.9 months versus 5.7 months) at 1 year (95% CI 27% to 45%, P = 0.0009). The ECF regimen was associated with an increased risk of nausea, emesis, and alopecia, while more hematologic toxicity and infections were noted with the FAMTX regimen.18 In addition, in a phase 3 trial, Van Cutsem and colleagues examined the role of docetaxel in combination with cisplatin and fluorouracil (DCF) compared to cisplatin and fluorouracil alone. Addition of docetaxel led to improved OS and time to progression (9.2 months versus 8.6 months for cisplatin and fluorouracil alone, P = 0.02) but with an increased risk of grade 3 and 4 toxicities (69% versus 59%). These adverse events included neutropenia (82% versus 57% of cisplatin and fluorouracil patients), diarrhea (19% versus 8%), stomatitis (21% versus 27%), and fatigue (19% versus 14%).19
The landmark phase 3 REAL-2 study compared 4 chemotherapy regimens in patients with untreated advanced esophagogastric cancer. This study was conducted to determine if the efficacy of cisplatin and oxaliplatin, a third-generation platinum agent, is equivalent to that of fluorouracil and capecitabine, an oral fluoropyrimidine. In this trial, a 2 × 2 design was used to compare 4 regimens: ECF versus epirubicin, cisplatin, and capecitabine (ECX) versus epirubicin, oxaliplatin, and fluorouracil (EOF) versus epirubicin, oxaliplatin, and capecitabine (EOX). The study found EOX to be noninferior to ECF, with a trend towards improved OS compared to other combination regimens (11.2 months versus 9.9 months, HR 0.80 [95% CI 0.66 to 0.97], P = 0.02).20 Thus, the study demonstrated that an oxaliplatin and capecitabine-based regimen could replace cisplatin and fluorouracil. Given that fluorouracil administration requires long continuous infusions, the oral-based capecitabine regimen is an attractive option for patients.
Several trials have demonstrated the equivalency of oxaliplatin with cisplatin in combination regimens for the treatment of advanced gastric cancer. Oxaliplatin has the benefit of an improved toxicity profile as compared to cisplatin, with the major dose-limiting toxicity being peripheral neuropathy
Given previous evidence that DCF (docetaxel, cisplatin, fluorouracil) is superior to cisplatin and fluorouracil alone, there was interest in determining if the addition of docetaxel to a backbone of fluorouracil, oxaliplatin, and leucovorin (FLO) could elicit a higher response rate. This concept was investigated in a phase 2 trial that assigned 54 patients with metastatic gastric or GEJ adenocarcinoma to receive biweekly infusions of oxaliplatin, leucovorin, fluorouracil, and docetaxel.21 Median time to response was 1.54 months, and the overall response rate was 57.7%. Median progression-free survival (PFS) was 5.2 months, and OS was 11.1 months. The most common grade 3 or 4 toxicities included neutropenia (48%), leukopenia (27.8%), diarrhea (14.8%), and fatigue (11.1%).
Irinotecan-based regimens have also been extensively studied in the first-line treatment of metastatic gastric cancer, particularly as an alternative to platinum-based therapy, but superiority has not been established. The combination of fluorouracil, leucovorin, and irinotecan (FOLFIRI) was compared to ECX in a phase 3 trial.22 The study enrolled 416 patients with locally advanced or metastatic gastric or GEJ cancer. At a median follow up of 31 months, the time to progression was longer in the FOLFIRI arm as compared to the ECX arm (5.1 months versus 4.2 months, P = 0.008), but there was no difference in OS (9.5 months versus 9.7 months, P = 0.95), median PFS (5.3 months versus 5.8 months, P = 0.96), or response rate (39.2% versus 37.8%). However, the FOLFIRI regimen had an improved toxicity profile, with a lower overall rate of grade 3 or 4 toxicity (69% versus 84%, P < 0.001). Given these findings, the FOLFIRI regimen is an acceptable alternative to platinum-based therapy in suitable patients.22
HER2-POSITIVE DISEASE
The HER2 proto-oncogene, initially described in breast cancer, has been implicated in several malignancies, including gastric and esophageal cancer. Overexpression or amplification of HER2 can be found in up to 30% of gastric cancers.23 For these patients, adding trastuzumab to a standard regimen of platinum and fluoropyrimidine is the standard of care. The prospective phase 3 Trastuzumab for Gastric Cancer (ToGA) trial randomly assigned 594 patients with HER2-positive gastric cancer to receive either cisplatin and fluorouracil or capecitabine and cisplatin with trastuzumab (n = 294) or without (n = 290) trastuzumab every 3 weeks for a total of 6 cycles, followed by maintenance trastuzumab until disease progression was noted.24 HER2 positivity was defined as HER2 protein overexpression by IHC (cutoff of 3+) or gene amplification by fluorescence in situ hybridization (FISH); tumors with IHC 2+ patterns were followed with FISH studies to confirm positivity. The study found a higher incidence of HER2-positive tumors in patients with GEJ tumors compared to patients with distal gastric cancers (33% versus 20%).24 In this trial, the addition of trastuzumab was associated with an improvement in OS: 13.5 months in the trastuzumab cohort versus 11.1 months in those receiving chemotherapy alone (HR 0.74, P = 0.0048). There was not a significant difference in toxicities between the 2 cohorts, with nausea, emesis, and neutropenia being the most common adverse events. Rates of overall grade 3 or 4 events were similar as well (68% in each cohort). Further exploratory analysis was also conducted according to HER2 status by dividing patients into a “high-expressor” group (n = 446), defined as patients with IHC 3+ tumors or IHC 2+ and FISH positivity, and a “low-expressor” group (n = 131), which included patients with IHC 0 or 1+ tumors. Analysis of patients in the 2 subgroups demonstrated an improved OS with the addition of trastuzumab for the high-expressor cohort, with a median OS of 16 months (HR 0.65 [95% CI 0.51 to 0.83]) compared to 11.8 months in those receiving only chemotherapy.
Dual HER2 blockade has been investigated in metastatic gastric cancer. The phase 3 randomized JACOB trial assigned 780 patients to receive either trastuzumab with a cisplatin/fluoropyrimidine regimen with or without the addition of pertuzumab; the primary end point was OS.25 A non-statistically significant trend towards improvement in OS was found in the pertuzumab arm (17.5 months) as compared with the standard of care arm (14.2 months, HR 0.84, P = 0.0565). The pertuzumab/trastuzumab/chemotherapy cohort experienced a higher incidence of diarrhea (61.6% versus 35.1% in control arm). Cardiac toxicity was comparable in the 2 cohorts.
The Table provides a summary of relevant clinical trials in metastatic gastric cancer.
SECOND-LINE THERAPY
CASE CONTINUED
The patient receives capecitabine, oxaliplatin, and trastuzumab therapy for 6 cycles, followed by trastuzumab for another 3 cycles. While on therapy, he develops a painful right clavicular lesion. He undergoes magnetic resonance imaging of the right clavicle, which shows a lesion in the distal two-thirds of the right clavicle measuring 9.7 × 3.7 × 3.8 cm. The patient is started on palliative radiation to the clavicle. However, repeat CT imaging shows progressive liver metastases.
• What is the approach to second-line therapy for metastatic gastric cancer?
Improvements in our understanding of the molecular pathways that lead to tumorigenesis have contributed to the development of several targeted agents whose efficacy in gastric cancer is being investigated. The NCCN guidelines recommend that for all patients who progress on frontline therapy, second-line therapy consists of a combination of ramucirumab and paclitaxel. Other options include single-agent docetaxel, paclitaxel, irinotecan, or ramucirumab. Combination therapy using irinotecan with either docetaxel, fluorouracil, or cisplatin may also be used.
Ramucirumab, a human IgG1 monoclonal antibody that targets the vascular endothelial growth factor receptor 2 (VEGFR2), was initially approved in 2014 as monotherapy for patients who had previously progressed on first-line chemotherapy. Its approval was based on the results of the phase 3 randomized, double-blind placebo-controlled REGARD study.26 The trial randomly assigned 355 patients with advanced gastric or GEJ adenocarcinoma and disease progression after first-line platinum-containing or fluoropyrimidine-containing chemotherapy to receive best supportive care plus either ramucirumab (n = 238) or placebo (n = 117). Monotherapy with ramucirumab significantly improved median OS compared with placebo (5.2 months versus. 3.8 months; HR 0.776 [95% CI 0.6 to 0.99], P = 0.047). There was also an improvement in PFS of 2.1 months in the ramucirumab cohort, as compared to 1.3 months in the placebo cohort (P < 0.0001). Patients in the ramucirumab arm experienced a higher incidence of hypertension (16% versus 8%), but all other adverse events occurred at comparable rates. Five deaths in the ramucirumab group were thought to be secondary to the study drug, as compared to 2 deaths in the placebo group.
In the subsequent phase 3 RAINBOW trial, the addition of ramucirumab to paclitaxel was investigated, with 330 patients assigned to the combination group and 335 to the paclitaxel-only group.27 The trial again showed that combination therapy afforded patients a significant survival advantage compared to paclitaxel alone, with a median OS of 9.6 months versus 7.4 months for the monotherapy group (HR 0.807 [95% CI 0.678 to 0.962], P = 0.017). A PFS benefit of 4.4 months was observed in the combination therapy groups, as compared with 2.9 months in the monotherapy group (HR 0.635, P < 0.0001). The ramucirumab/paclitaxel group also had a higher overall response rate of 28% versus 16%. The combination cohort had an increased incidence of grade 3 or higher adverse hypertensive events (14% versus 2%) and neutropenia (41% versus 19%), while the incidence of grade 3 febrile neutropenic events was similar between the groups (3% versus 2%).
The addition of bevacizumab, another monoclonal antibody against VEGF, to standard chemotherapy regimens has been explored, but studies have failed to show a survival benefit with this agent in the first-line treatment of advanced gastric cancer. The phase 3 Avastin in Gastric Cancer (AVAGAST) trial was a multinational, randomized study where patients received either bevacizumab (n = 387) or placebo (n = 387) in addition to cisplatin and capecitabine.28 The substitution of fluorouracil for capecitabine was permitted for patients who were unable to tolerate oral medications. Cisplatin was administered for a maximum of 6 cycles, while capecitabine and bevacizumab were administered until disease progression. The study failed to show an improvement in OS, with a median OS of 12.1 months noted in the bevacizumab cohort, as compared to 10.1 months in the placebo arm (HR 0.87 [95% CI 0.73 to 1.03], P = 0.1002). However, there was a modest improvement in median PFS (6.7 months versus 5.3 months; HR 0.80 [95% CI 0.68 to 0.93], P = 0.0037) and overall response rate (46% versus 37.4%, P = 0.0315). The most commonly reported grade 3 to 5 adverse events included neutropenia (35%), anemia (10%), and loss of appetite (8%). Interestingly, in a follow-up report, higher serum levels of VEGF-A were thought to correlate with an enhanced response to bevacizumab.29 However, the routine use of biomarker analysis in selecting patients for treatment with bevacizumab in metastatic gastric cancer remains to be further clarified.
Use of other agents with anti-HER2 activity in the second-line treatment of patients who have experienced progression while on trastuzumab remains unclear. In the recent T-ACT trial, patients with disease refractory to frontline therapy with combination trastuzumab and fluoropyrimidine/platinum agents were randomly assigned to receive either weekly paclitaxel (n = 45) or weekly paclitaxel plus trastuzumab (n = 44).30 Patients in the combination cohort received an initial dose of trastuzumab 8 mg/kg followed by 6 mg/kg every 3 weeks until progression. The study did not find a difference in either PFS (3.19 months versus 3.68 months; HR 0.91 [95% CI 0.67 to 1.22], P = 0.33) or OS (9.95 months versus 10.2 months; HR 1.23 [95% CI 0.75 to 1.99], P = 0.20). The study thus failed to show a benefit to continuing trastuzumab after progression in the first-line setting.
Lapatinib in combination with paclitaxel has been compared to paclitaxel alone for the treatment of advanced HER2-positive gastric cancer in an Asian population in the phase 3 TyTAN trial.31 With a primary end point of OS, the study randomly assigned 129 patients to receive paclitaxel alone and 132 patients to receive paclitaxel with lapatinib. There was a nonsignificant trend towards improvement in OS in the combination group (11 months) as compared to the paclitaxel-only group(8.9 months, P = 0.1044), with no significant difference in median PFS (5.4 months versus 4.4 months). However, it is important to note that only 15 patients in this trial had previously been exposed to trastuzumab. Another trial, the phase 3 GATSBY study, examined the efficacy of trastuzumab emtansine in the second-line setting compared to taxanes alone and failed to show any improvement in PFS or OS.32 Given these results, no alternative anti-HER2 therapy has been proven to be efficacious for patients who are trastuzumab refractory. Therefore, including anti-HER2 therapy in the second-line treatment of HER2-positive gastric cancer is not recommended.
IMMUNOTHERAPY AND OTHER TARGETED THERAPIES
Several other targeted therapies have been studied in advanced gastric cancer, without any demonstrable survival benefit. The PI3K/AKT/mTOR pathway is known to be involved in regulation of cell growth and angiogenesis, and the mTOR inhibitor everolimus is widely used to treat other malignancies, including breast cancer. The use of everolimus in the second-line setting was studied in the phase 3 GRANITE-1 trial, where it was compared to best supportive care and failed to provide any survival benefit.33 Cetuximab, a recombinant human and mouse chimeric monoclonal antibody, and panitumumab, a recombinant human antibody against the epidermal growth factor receptor (EGFR), have also been examined in gastric and GEJ cancer patients. However, the large phase 3 EXPAND and REAL-3 trials did not show a survival benefit when these agents were added to standard chemotherapy.34,35
Overexpression of MET, a proto-oncogene and tyrosine kinase receptor, has also been implicated in gastric cancer progression. The ligand for MET is the hepatocyte growth factor (HGF), and aberrant signaling of this pathway has been shown to correlate with an aggressive gastric cancer phenotype and poorer OS by promoting tumor growth and angiogenesis. However, no MET inhibitors thus far have been found to be clinically effective. RILOMET-1 and RILOMET-2 were phase 3 trials examining the efficacy of rilotumumab, a humanized anti-HGF antibody, in combination with chemotherapy (ECX and cisplatin with capecitabine, respectively) for the frontline treatment of MET-positive GEJ and gastric cancers. Both studies were discontinued due to a higher treatment-related mortality in patients receiving rilotumumab, with a higher incidence of adverse events due to disease progression being noted.36 Similarly, onartuzumab, a monovalent monoclonal antibody against the MET receptor, was investigated in the phase 3 METGastric trial in combination with modified FOLFOX6 as first-line therapy for HER2-negative, MET-positive metastatic GEJ and gastric cancers. The study did not demonstrate any significant improvements in OS or PFS.37
There has been significant interest in incorporating immunotherapy in the treatment of early and metastatic gastric cancer. Pembrolizumab is the first programmed death receptor (PD-1) inhibitor to be approved for treatment of patients with PD-L1−positive advanced gastric cancer who had previously received 2 or more lines of chemotherapy. Although earlier studies of pembrolizumab in lung cancer utilized the tumor proportion score (TPS) to determine PD-L1 positivity, this was not found to be applicable to gastric cancer. Instead, the combined positive score (CPS) is used in gastric cancer. The CPS evaluates the number of tumor cells and immune cells (macrophages and lymphocytes) that stain positive for PD-L1 relative to all viable tumor cells. Comparatively, the TPS only examines the percentage of viable tumor cells that show complete or partial positive staining for PD-L1. A CPS score of 1 or greater identifies patients who would be suitable candidates for pembrolizumab.
The approval of pembrolizumab was based on the positive findings from the recent KEYNOTE-059 trial.38 The study included 259 patients who had previously received either fluoropyrimidine, cisplatin, or anti-HER2 therapy, with 148 patients (55%) of these patients having PD-L1−positive tumors. The PD-L1 status was determined using a pharmDx Kit, which is now approved by the US Food and Drug Administration to select patients who could benefit from pembrolizumab treatment. CPS was calculated as the number of PD-L1−staining cells divided by the total number of evaluated cells. The study included patients with microsatellite stable (MSI-S), undetermined, or deficient MMR status. The overall response rate to pembrolizumab across all patients was 11.6%, median PFS was 2 months, and the 12-month OS rate was 23.4%. In the subset of patients with MSI-H tumors, the overall response rate was 57.1%, with a complete response rate of 14.3%; in those with MSI-S tumors, the overall response rate was 9% and the complete response rate was 2.4%. Among patients with PD-L1–positive tumors, the overall response rate was 15.5% (95% CI 10.1% to 22.4). Common adverse events included fatigue, hypothyroidism, nausea, diarrhea, and arthralgia.38
CASE CONCLUSION
This patient with metastatic gastric cancer receives second-line chemotherapy with ramucirumab and paclitaxel. Follow-up imaging shows persistent liver metastases and new lung metastasis. Because the tumor is PD-L1–positive, the patient receives 4 cycles of pembrolizumab, with no significant change noted in disease burden. He notes a significant decline in functional status with increased weight loss, nausea, emesis, and fatigue. The patient opts to forego any further therapy and instead chooses to pursue supportive care only.
SUMMARY
Gastric cancer is the third most common cause of cancer death worldwide. Common risk factors for developing gastric cancer include H. pylori infection, smoking, alcohol abuse, radiation exposure, high-fat diet, and obesity. Patients presenting with alarm symptoms of nausea, emesis, early satiety, abdominal pain, or weight loss should be fully evaluated with upper GI endoscopy. If there is suspicion for metastatic disease, CT evaluation of the chest, abdomen, and pelvis with oral and intravenous contrast should be obtained. Treatment of patients with metastatic gastric cancer is guided by their performance status at presentation. For patients with good performance status, a combination of platinum and fluoropyrimidine therapy, such as FOLFOX, can be considered. Doublet chemotherapy regimens are preferred over triplet chemotherapy regimens given their better tolerability. For patients with HER2-positive disease, the addition of trastuzumab to the platinum and fluoropyrimidine backbone is the standard of care in the first line.
Several targeted agents have been studied in patients progressing on initial therapy, with ramucirumab and paclitaxel being considered the regimen of choice in the second line. No anti-HER2 therapy has been approved for patients who are refractory to trastuzumab. Pembrolizumab is approved for use in patients who are PD-L1–positive and have previously progressed on at least 2 lines of chemotherapy. Pembrolizumab is also approved for the treatment of patients with unresectable or metastatic, MSI-H or MMR-deficient gastric cancers that have progressed after prior treatment and who have no satisfactory alternative treatment options.
1. Noone AM, Cronin KA, Altekruse SF, et al. Cancer incidence and survival trends by subtype using data from the Surveillance Epidemiology and End Results Program, 1992-2013. Cancer Epidemiol Biomarkers Prev 2017;26:632–41
2. Global Burden of Disease Cancer Collaboration, Fitzmaurice C, Allen C, et al. Global, regional, and national cancer incidence, mortality, years of life lost, years lived with disability, and disability-adjusted life-years for 32 cancer groups, 1990 to 2015: a systematic analysis for the global burden of disease study. JAMA Oncol 2017;3:524–8.
3. Sitarz R, Skierucha M, Mielko J, et al. Gastric cancer: epidemiology, prevention, classification, and treatment. Cancer Manag Res 2018;10:239–48.
4. Olefson S, Moss SF. Obesity and related risk factors in gastric cardia adenocarcinoma. Gastric Cancer 2015;18:23–32.
5. Wang F, Meng W, Wang B, Qiao L. Helicobacter pylori-induced gastric inflammation and gastric cancer. Cancer Lett 2014;345:196–202.
6. Espinoza JL, Matsumoto A, Tanaka H, Matsumura I. Gastric microbiota: An emerging player in Helicobacter pylori-induced gastric malignancies. Cancer Lett 2018;414:147–52.
7. Chmiela M, Gonciarz W. Molecular mimicry in Helicobacter pylori infections. World J Gastroenterol 2017;23:3964–77.
8. Isobe Y, Nashimoto A, Akazawa K, et al. Gastric cancer treatment in Japan: 2008 annual report of the JGCA nationwide registry. Gastric Cancer 2011;14:301–16.
9. Ford A, Gurusamy KS, Delaney B, et al. Eradication therapy for peptic ulcer disease in Helicobacter pylori-positive patients. Cochrane Database Syst Rev 2004(4):CD003840.
10. Karimi P, Islami F, Anandasabapathy S, et al. Gastric cancer: descriptive epidemiology, risk factors, screening, and prevention. Cancer Epidemiol Biomarkers Prev 2014;23:700–13.
11. Van Cutsem E, Sagaert X, Topal B, et al. Gastric cancer. Lancet 2016;388:2654–64.
12. Chan BA , Sim HW, Natori A, et al. Survival outcomes for de novo versus relapsed stage IV gastric and gastroesophageal junction (GEJ) adenocarcinoma [abstract]. J Clin Oncol 2018;36(no. 4 suppl):148.
13. DeVita VT, Lawrence TS, Rosenberg SA. DeVita, Hellman, and Rosenberg’s cancer: principles & practice of oncology. 9th ed. Philadelphia: Wolters Kluwer Health/Lippincott Williams & Wilkins; 2011.
14. Siewert JR, Hölscher AH, Becker K, Gössner W. [Cardia cancer: attempt at a therapeutically relevant classification]. [Article in German.] Chirurg 1987;58:25–32.
15. Amin MB, Edge SB, Greene FL, et al, eds. AJCC cancer staging manual. 8th ed. New York: Springer; 2017.
16. Wagner AD, Syn NL, Moehler M, et al. Chemotherapy for advanced gastric cancer. Cochrane Database Syst Rev 2017;8:CD004064.
17. Qiu H, Zhou Z. [Updates and interpretation on NCCN clinical practice guidelines for gastric cancer 2017 version 5]. [Article in Chinese.] Zhonghua Wei Chang Wai Ke Za Zhi 2018;21:160–4.
18. Webb A, Cunningham D, Scarffe JH, et al. Randomized trial comparing epirubicin, cisplatin, and fluorouracil versus fluorouracil, doxorubicin, and methotrexate in advanced esophagogastric cancer. J Clin Oncol 1997;15:261–7.
19. Van Cutsem E, Moiseyenko VM, Tjulandin S, et al. Phase III study of docetaxel and cisplatin plus fluorouracil compared with cisplatin and fluorouracil as first-line therapy for advanced gastric cancer: a report of the V325 Study Group. J Clin Oncol 2006;24:4991–7.
20. Cunningham D, Okines AF, Ashley S. Capecitabine and oxaliplatin for advanced esophagogastric cancer. N Engl J Med 2010;362:858–9.
21. Al-Batran SE, Hartmann JT, Hofheinz R, et al. Biweekly fluorouracil, leucovorin, oxaliplatin, and docetaxel (FLOT) for patients with metastatic adenocarcinoma of the stomach or esophagogastric junction: a phase II trial of the Arbeitsgemeinschaft Internistische Onkologie. Ann Oncol 2008;19:1882–7.
22. Guimbaud R, Louvet C, Ries P, et al. Prospective, randomized, multicenter, phase III study of fluorouracil, leucovorin, and irinotecan versus epirubicin, cisplatin, and capecitabine in advanced gastric adenocarcinoma: a French intergroup (Federation Francophone de Cancerologie Digestive, Federation Nationale des Centres de Lutte Contre le Cancer, and Groupe Cooperateur Multidisciplinaire en Oncologie) study. J Clin Oncol 2014;32:3520–6.
23. Boku N. HER2-positive gastric cancer. Gastric Cancer 2014;17:1–12.
24. Bang YJ, Van Cutsem E, Feyereislova A, et al. Trastuzumab in combination with chemotherapy versus chemotherapy alone for treatment of HER2-positive advanced gastric or gastro-oesophageal junction cancer (ToGA): a phase 3, open-label, randomised controlled trial. Lancet 2010;376:687–97.
25. Tabernero J, Hoff PM, Shen L, et al. Pertuzumab + trastuzumab + chemotherapy for HER2-positive metastatic gastric or gastro-oesophageal junction cancer: Final analysis of a Phase III study (JACOB) [abstract]. Ann Oncol 2017;28(suppl 5):6160.
26. Fuchs CS, Tomasek J, Yong CJ, et al. Ramucirumab monotherapy for previously treated advanced gastric or gastro-oesophageal junction adenocarcinoma (REGARD): an international, randomised, multicentre, placebo-controlled, phase 3 trial. Lancet 2014;383:31–9.
27. Wilke H, Muro K, Van Cutsem E, et al. Ramucirumab plus paclitaxel versus placebo plus paclitaxel in patients with previously treated advanced gastric or gastro-oesophageal junction adenocarcinoma (RAINBOW): a double-blind, randomised phase 3 trial. Lancet Oncol 2014;15:1224–35.
28. Ohtsu A, Shah MA, Van Cutsem E, et al. Bevacizumab in combination with chemotherapy as first-line therapy in advanced gastric cancer: a randomized, double-blind, placebo-controlled phase III study. J Clin Oncol 2011;29:3968–76.
29. Van Cutsem E, de Haas S, Kang YK, et al, Bevacizumab in combination with chemotherapy as first-line therapy in advanced gastric cancer: a biomarker evaluation from the AVAGAST randomized phase III trial. J Clin Oncol 2012;30:2119–27.
30. Makiyama A, Sagara K, Kawada J, et al. A randomized phase II study of weekly paclitaxel ± trastuzumab in patients with HER2-positive advanced gastric or gastro-esophageal junction cancer refractory to trastuzumab combined with fluoropyrimidine and platinum: WJOG7112G (T-ACT) [abstract]. J Clin Oncol 2018;36(no. 15 suppl):4011.
31. Satoh T, Xu RH, Chung HC, et al. Lapatinib plus paclitaxel versus paclitaxel alone in the second-line treatment of HER2-amplified advanced gastric cancer in Asian populations: TyTAN--a randomized, phase III study. J Clin Oncol 2014;32:2039–49.
32. Thuss-Patience PC, Shah MA, Ohtsu A, et al. Trastuzumab emtansine versus taxane use for previously treated HER2-positive locally advanced or metastatic gastric or gastro-oesophageal junction adenocarcinoma (GATSBY): an international randomised, open-label, adaptive, phase 2/3 study. Lancet Oncol 2017;18:640–53.
33. Ohtsu A, Ajani JA, Bai YX, et al. Everolimus for previously treated advanced gastric cancer: results of the randomized, double-blind, phase III GRANITE-1 study. J Clin Oncol 2013;31:3935–43.
34. Lordick F, Kang YK, Chung HC, et al. Capecitabine and cisplatin with or without cetuximab for patients with previously untreated advanced gastric cancer (EXPAND): a randomised, open-label phase 3 trial. Lancet Oncol 2013;14:490–9.
35. Waddell T, Chau I, Cunningham D, et al. Epirubicin, oxaliplatin, and capecitabine with or without panitumumab for patients with previously untreated advanced oesophagogastric cancer (REAL3): a randomised, open-label phase 3 trial. Lancet Oncol 2013;14:481–9.
36. Catenacci DVT, Tebbutt NC, Davidenko I, et al. Rilotumumab plus epirubicin, cisplatin, and capecitabine as first-line therapy in advanced MET-positive gastric or gastro-oesophageal junction cancer (RILOMET-1): a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Oncol 2017;18:1467–82.
37. Shah MA, Bang YJ, Lordick F, et al. Effect of fluorouracil, leucovorin, and oxaliplatin with or without onartuzumab in HER2-negative, MET-positive gastroesophageal adenocarcinoma: the METGastric randomized clinical trial. JAMA Oncol 2017;3:620–7.
38. Fuchs CS, Doi T, Jang RW, et al. Safety and efficacy of pembrolizumab monotherapy in patients with previously treated advanced gastric and gastroesophageal junction cancer: phase 2 clinical KEYNOTE-059 trial. JAMA Oncol 2018;4(5):e180013.
INTRODUCTION
According to the Surveillance, Epidemiology and End Results database, in 2017 there were 28,000 new cases of gastric cancer, accounting for 1.8% of all malignancies in the United States, and an estimated 10,960 gastric cancer–related deaths.1 Worldwide, gastric cancer is the fifth most common malignancy and the third most common cause of death from any cancer.2 The incidence of gastric cancer varies significantly by geographic region, with countries in Eastern Asia (China, Japan), Eastern Europe, and Central and South America accounting for 50% of all new cases.3 Although the incidence of gastric cancer has declined in recent years, this decrease has not been observed consistently across all nations.2 In particular, the incidence of gastric cancers arising from the cardia has been increasing, which is perhaps due to a higher prevalence of obesity in Western societies.4
In this article, we review key aspects of management of metastatic gastric cancer, including selection of first- and second-line therapy, and discuss targeted agents and upcoming clinical trials.
EPIDEMIOLOGY AND RISK FACTORS
Chronic infection with Helicobacter pylori, a gram-negative bacterium, is a strong etiological factor for the development of gastric cancer, contributing to up to 70% of cases.2 The pathogen can colonize the gastric mucosa, leading to chronic inflammation. Although most patients remain asymptomatic, 1% to 3% develop gastric cancer and another 0.1% develop mucosa-associated lymphoid tissue lymphoma.5 H. pylori infection is more commonly associated with cancer of the gastric body than with cancer of the gastroesophageal junction (GEJ). The increased burden of gastric cancer in countries in Eastern Asia, Latin America, and Eastern Europe has been correlated to the prevalence of chronic H. pylori infection in these areas.
Carcinogenesis secondary to H. pylori infection may occur via several mechanisms. First, H. pylori can release virulence factors, such as cytotoxin-associated gene A, vacuolating cytotoxin, and outer membrane proteins, into the cytosol of host cells, leading to changes in patterns of cell proliferation and apoptosis.6 These virulence factors can modulate the host immune system, attenuating it to promote dysplasia. In addition, continued recognition of these factors by the immune system leads to a persistent inflammatory response, with the release of cytokines such as interleukin (IL) -1β, IL-6, and IL-8. This leads to chronic mucosal damage, further promoting dysplasia with eventual transformation into adenocarcinoma.7 In Japan and Korea, where screening for H. pylori infection is routinely performed, there have been improvements in overall survival (OS) rates for gastric cancer, with 5-year OS rates of 70%.8 The International Agency for Research on Cancer recommends further research into population-based screening and treatment programs for patients with chronic H. pylori infection. However, despite this recommendation, optimal screening strategies are not clearly defined.9
Other risk factors for the development of gastric cancer include chronic gastroesophageal reflux disease; smoking; alcohol use; exposure to radiation; diets high in fats, salt, and smoked items and low in fruits and vegetables; obesity; and exposure to chemotherapeutic agents such as procarbazine.10 Another pathogen suspected, but not proven, to be associated with increased risk for gastric cancer is the Epstein-Barr virus, a human herpesvirus found in 80% of all gastric carcinomas with lymphoid features.11 In addition, whether the use of medications such as statins and nonsteroidal anti-inflammatory drugs confers a decreased risk of gastric cancers remains unclear.10
EVALUATION
CASE PRESENTATION
A 55-year-old Caucasian man with a history of type 2 diabetes mellitus presents to the gastrointestinal (GI) clinic with a 6-month history of dysphagia. The dysphagia is worsened with ingestion of solids, particularly towards the end of the day. He states that the food often gets “stuck in the middle of the chest.” The patient denies any nausea or emesis but notes that he has a poor appetite. He reports having worsening mid-epigastric abdominal pain that is non-radiating, dull in character, and 6/10 in intensity. He also reports a 10-lb weight loss over the past 2 months. He has no previous history of reflux, chest pain, dyspnea, or cough. Review of systems is otherwise benign. Physical exam is within normal limits.
• Which tests should be conducted when gastric cancer is suspected?
Persistent epigastric abdominal pain and weight loss are the most common early symptoms of gastric cancer. Nausea, early satiety, dysphagia, and occult GI bleeding can be other presenting signs. Patients presenting with alarm symptoms of nausea, emesis, early satiety, abdominal pain, or weight loss should be fully evaluated with upper GI endoscopy. Early diagnosis of gastric cancer is essential in obtaining a curative resection. However, at least 40% of patients present with de novo metastatic disease at the time of initial diagnosis.12 Gastric cancer spreads by direct extension through the gastric wall, with the liver, peritoneum, and regional lymph nodes being the most common sites of metastatic deposits.13 Classically, Virchow’s node, the left supraclavicular lymph node, is involved with metastatic gastric cancer. Involvement of the left axillary lymph node (Irish node) or a periumbilical nodule (Sister Mary Joseph node) may also be observed. Other, less commonly noted sites of metastatic disease include the ovaries, central nervous system, bone, lung, and soft tissues.13
Upper GI endoscopy is the best method for determining tumor location and extent and obtaining a specimen for a definitive tissue diagnosis.14 It is essential to accurately identify the location of the tumor in the stomach and relative to the GEJ. The American Joint Committee on Cancer classification defines tumors involving the GEJ with an epicenter no more than 2 cm into the proximal stomach as esophageal cancers.15 Tumors of the GEJ with their epicenter more than 2 cm into the proximal stomach are defined as gastric cancers. If metastatic disease is suspected, computed tomography (CT) scan of the chest, abdomen, and pelvis with oral and intravenous contrast can be obtained to determine the extent of disease spread. In the absence of any metastatic disease, endoscopic ultrasound (EUS) should be conducted to determine the depth of tumor invasion (T staging) and lymph node status. In the era of targeted therapy, patients with metastatic disease should undergo testing for human epidermal growth factor-2 (HER-2) expression, microsatellite instability (MSI), and programmed death ligand 1 (PD-L1) expression. Patients should be staged according to the TNM staging system.
FIRST-LINE TREATMENT OPTIONS
CASE CONTINUED
The patient undergoes esophagoduodenoscopy (EGD) and is found to have a gastric cardia mass extending into the distal esophagus. EUS also demonstrates multiple abdominal and mediastinal lymph nodes. No gastric outlet obstruction is found. Biopsy shows poorly differentiated invasive adenocarcinoma. Warthin–Starry stain is negative for H. pylori organism. The tumor cells are positive for cytokeratin (CK7), CK19, and mucin-1 gene (MUC1); focally positive for CK20; and negative for MUC2. HER2 testing results are reported as immunohistochemistry (IHC) 3+, consistent with strongly positive HER2 protein expression. Further IHC testing for mismatch repair (MMR) proteins shows intact nuclear expression of MLH1, MSH2, MSH6, and PMS2 protein, consistent with a low probability of MSI-high tumor. The tumor is found to be PD-L1 positive. Imaging reveals abnormal mass-like nodular thickening of the gastric wall, with an infiltrative opacity within the pancreatico-duodenal groove, suspicious for tumor infiltration. Multiple metastatic deposits are noted in the liver, peritoneum, and bilateral lungs. There is extensive gastrohepatic ligament and periportal lymphadenopathy and mild enlargement of the pulmonary hilar lymph nodes. These findings are consistent with stage 4 (T4bN3aM1) gastric cancer. Given these findings, staging laparoscopy is deferred.
• What are the first-line treatment options for patients with metastatic gastric cancer?
Patients with metastatic gastric cancer have a poor prognosis, and management is stratified based on performance status (Figure). In patients with good performance status, systemic chemotherapy is the mainstay of treatment. The goal of therapy is not curative, but rather treatment focuses on palliation of symptoms arising from tumor spread. Given this treatment goal, there has been considerable interest in clarifying the utility of chemotherapy as opposed to best supportive care. In a recent Cochrane review of 64 randomized control trials involving 11,698 patients, chemotherapy was found to improve OS by 6.7 months as compared to best supportive care (hazard ratio [HR] 0.3 [95% confidence interval {CI} 0.24 to 0.55]).16 Five classes of cytotoxic chemotherapeutic agents have demonstrated activity in gastric cancer. These include fluoropyrimidine (either infusional fluorouracil or capecitabine), platinum agents (cisplatin or oxaliplatin), taxanes (docetaxel or paclitaxel), anthracyclines (epirubicin), and irinotecan.13 Treatment options are further divided based on whether the patient has HER2-overexpressing or non-expressing malignancy.
HER2-NEGATIVE DISEASE
For patients with HER2-negative disease, National Comprehensive Cancer Network (NCCN) guidelines recommend using 2-drug combination regimens rather than 3 drugs, given concern for increased toxicity with 3-drug regimens.17 For patients with a performance status of 0 to 1, utilization of a 3-drug regimen is a reasonable alternative. The combination of a fluoropyrimidine with a platinum agent is considered the standard of care, with regimens such as fluorouracil, leucovorin, and oxaliplatin (FOLFOX) being commonly used.
Epirubicin-containing regimens have also been extensively studied in advanced gastric cancer. In a study of 274 previously untreated patients with GEJ cancers, the combination of epirubicin, cisplatin, and fluorouracil (ECF) was compared to fluorouracil, doxorubicin, and methotrexate (FAMTX). There was an OS benefit favoring ECF (8.9 months versus 5.7 months) at 1 year (95% CI 27% to 45%, P = 0.0009). The ECF regimen was associated with an increased risk of nausea, emesis, and alopecia, while more hematologic toxicity and infections were noted with the FAMTX regimen.18 In addition, in a phase 3 trial, Van Cutsem and colleagues examined the role of docetaxel in combination with cisplatin and fluorouracil (DCF) compared to cisplatin and fluorouracil alone. Addition of docetaxel led to improved OS and time to progression (9.2 months versus 8.6 months for cisplatin and fluorouracil alone, P = 0.02) but with an increased risk of grade 3 and 4 toxicities (69% versus 59%). These adverse events included neutropenia (82% versus 57% of cisplatin and fluorouracil patients), diarrhea (19% versus 8%), stomatitis (21% versus 27%), and fatigue (19% versus 14%).19
The landmark phase 3 REAL-2 study compared 4 chemotherapy regimens in patients with untreated advanced esophagogastric cancer. This study was conducted to determine if the efficacy of cisplatin and oxaliplatin, a third-generation platinum agent, is equivalent to that of fluorouracil and capecitabine, an oral fluoropyrimidine. In this trial, a 2 × 2 design was used to compare 4 regimens: ECF versus epirubicin, cisplatin, and capecitabine (ECX) versus epirubicin, oxaliplatin, and fluorouracil (EOF) versus epirubicin, oxaliplatin, and capecitabine (EOX). The study found EOX to be noninferior to ECF, with a trend towards improved OS compared to other combination regimens (11.2 months versus 9.9 months, HR 0.80 [95% CI 0.66 to 0.97], P = 0.02).20 Thus, the study demonstrated that an oxaliplatin and capecitabine-based regimen could replace cisplatin and fluorouracil. Given that fluorouracil administration requires long continuous infusions, the oral-based capecitabine regimen is an attractive option for patients.
Several trials have demonstrated the equivalency of oxaliplatin with cisplatin in combination regimens for the treatment of advanced gastric cancer. Oxaliplatin has the benefit of an improved toxicity profile as compared to cisplatin, with the major dose-limiting toxicity being peripheral neuropathy
Given previous evidence that DCF (docetaxel, cisplatin, fluorouracil) is superior to cisplatin and fluorouracil alone, there was interest in determining if the addition of docetaxel to a backbone of fluorouracil, oxaliplatin, and leucovorin (FLO) could elicit a higher response rate. This concept was investigated in a phase 2 trial that assigned 54 patients with metastatic gastric or GEJ adenocarcinoma to receive biweekly infusions of oxaliplatin, leucovorin, fluorouracil, and docetaxel.21 Median time to response was 1.54 months, and the overall response rate was 57.7%. Median progression-free survival (PFS) was 5.2 months, and OS was 11.1 months. The most common grade 3 or 4 toxicities included neutropenia (48%), leukopenia (27.8%), diarrhea (14.8%), and fatigue (11.1%).
Irinotecan-based regimens have also been extensively studied in the first-line treatment of metastatic gastric cancer, particularly as an alternative to platinum-based therapy, but superiority has not been established. The combination of fluorouracil, leucovorin, and irinotecan (FOLFIRI) was compared to ECX in a phase 3 trial.22 The study enrolled 416 patients with locally advanced or metastatic gastric or GEJ cancer. At a median follow up of 31 months, the time to progression was longer in the FOLFIRI arm as compared to the ECX arm (5.1 months versus 4.2 months, P = 0.008), but there was no difference in OS (9.5 months versus 9.7 months, P = 0.95), median PFS (5.3 months versus 5.8 months, P = 0.96), or response rate (39.2% versus 37.8%). However, the FOLFIRI regimen had an improved toxicity profile, with a lower overall rate of grade 3 or 4 toxicity (69% versus 84%, P < 0.001). Given these findings, the FOLFIRI regimen is an acceptable alternative to platinum-based therapy in suitable patients.22
HER2-POSITIVE DISEASE
The HER2 proto-oncogene, initially described in breast cancer, has been implicated in several malignancies, including gastric and esophageal cancer. Overexpression or amplification of HER2 can be found in up to 30% of gastric cancers.23 For these patients, adding trastuzumab to a standard regimen of platinum and fluoropyrimidine is the standard of care. The prospective phase 3 Trastuzumab for Gastric Cancer (ToGA) trial randomly assigned 594 patients with HER2-positive gastric cancer to receive either cisplatin and fluorouracil or capecitabine and cisplatin with trastuzumab (n = 294) or without (n = 290) trastuzumab every 3 weeks for a total of 6 cycles, followed by maintenance trastuzumab until disease progression was noted.24 HER2 positivity was defined as HER2 protein overexpression by IHC (cutoff of 3+) or gene amplification by fluorescence in situ hybridization (FISH); tumors with IHC 2+ patterns were followed with FISH studies to confirm positivity. The study found a higher incidence of HER2-positive tumors in patients with GEJ tumors compared to patients with distal gastric cancers (33% versus 20%).24 In this trial, the addition of trastuzumab was associated with an improvement in OS: 13.5 months in the trastuzumab cohort versus 11.1 months in those receiving chemotherapy alone (HR 0.74, P = 0.0048). There was not a significant difference in toxicities between the 2 cohorts, with nausea, emesis, and neutropenia being the most common adverse events. Rates of overall grade 3 or 4 events were similar as well (68% in each cohort). Further exploratory analysis was also conducted according to HER2 status by dividing patients into a “high-expressor” group (n = 446), defined as patients with IHC 3+ tumors or IHC 2+ and FISH positivity, and a “low-expressor” group (n = 131), which included patients with IHC 0 or 1+ tumors. Analysis of patients in the 2 subgroups demonstrated an improved OS with the addition of trastuzumab for the high-expressor cohort, with a median OS of 16 months (HR 0.65 [95% CI 0.51 to 0.83]) compared to 11.8 months in those receiving only chemotherapy.
Dual HER2 blockade has been investigated in metastatic gastric cancer. The phase 3 randomized JACOB trial assigned 780 patients to receive either trastuzumab with a cisplatin/fluoropyrimidine regimen with or without the addition of pertuzumab; the primary end point was OS.25 A non-statistically significant trend towards improvement in OS was found in the pertuzumab arm (17.5 months) as compared with the standard of care arm (14.2 months, HR 0.84, P = 0.0565). The pertuzumab/trastuzumab/chemotherapy cohort experienced a higher incidence of diarrhea (61.6% versus 35.1% in control arm). Cardiac toxicity was comparable in the 2 cohorts.
The Table provides a summary of relevant clinical trials in metastatic gastric cancer.
SECOND-LINE THERAPY
CASE CONTINUED
The patient receives capecitabine, oxaliplatin, and trastuzumab therapy for 6 cycles, followed by trastuzumab for another 3 cycles. While on therapy, he develops a painful right clavicular lesion. He undergoes magnetic resonance imaging of the right clavicle, which shows a lesion in the distal two-thirds of the right clavicle measuring 9.7 × 3.7 × 3.8 cm. The patient is started on palliative radiation to the clavicle. However, repeat CT imaging shows progressive liver metastases.
• What is the approach to second-line therapy for metastatic gastric cancer?
Improvements in our understanding of the molecular pathways that lead to tumorigenesis have contributed to the development of several targeted agents whose efficacy in gastric cancer is being investigated. The NCCN guidelines recommend that for all patients who progress on frontline therapy, second-line therapy consists of a combination of ramucirumab and paclitaxel. Other options include single-agent docetaxel, paclitaxel, irinotecan, or ramucirumab. Combination therapy using irinotecan with either docetaxel, fluorouracil, or cisplatin may also be used.
Ramucirumab, a human IgG1 monoclonal antibody that targets the vascular endothelial growth factor receptor 2 (VEGFR2), was initially approved in 2014 as monotherapy for patients who had previously progressed on first-line chemotherapy. Its approval was based on the results of the phase 3 randomized, double-blind placebo-controlled REGARD study.26 The trial randomly assigned 355 patients with advanced gastric or GEJ adenocarcinoma and disease progression after first-line platinum-containing or fluoropyrimidine-containing chemotherapy to receive best supportive care plus either ramucirumab (n = 238) or placebo (n = 117). Monotherapy with ramucirumab significantly improved median OS compared with placebo (5.2 months versus. 3.8 months; HR 0.776 [95% CI 0.6 to 0.99], P = 0.047). There was also an improvement in PFS of 2.1 months in the ramucirumab cohort, as compared to 1.3 months in the placebo cohort (P < 0.0001). Patients in the ramucirumab arm experienced a higher incidence of hypertension (16% versus 8%), but all other adverse events occurred at comparable rates. Five deaths in the ramucirumab group were thought to be secondary to the study drug, as compared to 2 deaths in the placebo group.
In the subsequent phase 3 RAINBOW trial, the addition of ramucirumab to paclitaxel was investigated, with 330 patients assigned to the combination group and 335 to the paclitaxel-only group.27 The trial again showed that combination therapy afforded patients a significant survival advantage compared to paclitaxel alone, with a median OS of 9.6 months versus 7.4 months for the monotherapy group (HR 0.807 [95% CI 0.678 to 0.962], P = 0.017). A PFS benefit of 4.4 months was observed in the combination therapy groups, as compared with 2.9 months in the monotherapy group (HR 0.635, P < 0.0001). The ramucirumab/paclitaxel group also had a higher overall response rate of 28% versus 16%. The combination cohort had an increased incidence of grade 3 or higher adverse hypertensive events (14% versus 2%) and neutropenia (41% versus 19%), while the incidence of grade 3 febrile neutropenic events was similar between the groups (3% versus 2%).
The addition of bevacizumab, another monoclonal antibody against VEGF, to standard chemotherapy regimens has been explored, but studies have failed to show a survival benefit with this agent in the first-line treatment of advanced gastric cancer. The phase 3 Avastin in Gastric Cancer (AVAGAST) trial was a multinational, randomized study where patients received either bevacizumab (n = 387) or placebo (n = 387) in addition to cisplatin and capecitabine.28 The substitution of fluorouracil for capecitabine was permitted for patients who were unable to tolerate oral medications. Cisplatin was administered for a maximum of 6 cycles, while capecitabine and bevacizumab were administered until disease progression. The study failed to show an improvement in OS, with a median OS of 12.1 months noted in the bevacizumab cohort, as compared to 10.1 months in the placebo arm (HR 0.87 [95% CI 0.73 to 1.03], P = 0.1002). However, there was a modest improvement in median PFS (6.7 months versus 5.3 months; HR 0.80 [95% CI 0.68 to 0.93], P = 0.0037) and overall response rate (46% versus 37.4%, P = 0.0315). The most commonly reported grade 3 to 5 adverse events included neutropenia (35%), anemia (10%), and loss of appetite (8%). Interestingly, in a follow-up report, higher serum levels of VEGF-A were thought to correlate with an enhanced response to bevacizumab.29 However, the routine use of biomarker analysis in selecting patients for treatment with bevacizumab in metastatic gastric cancer remains to be further clarified.
Use of other agents with anti-HER2 activity in the second-line treatment of patients who have experienced progression while on trastuzumab remains unclear. In the recent T-ACT trial, patients with disease refractory to frontline therapy with combination trastuzumab and fluoropyrimidine/platinum agents were randomly assigned to receive either weekly paclitaxel (n = 45) or weekly paclitaxel plus trastuzumab (n = 44).30 Patients in the combination cohort received an initial dose of trastuzumab 8 mg/kg followed by 6 mg/kg every 3 weeks until progression. The study did not find a difference in either PFS (3.19 months versus 3.68 months; HR 0.91 [95% CI 0.67 to 1.22], P = 0.33) or OS (9.95 months versus 10.2 months; HR 1.23 [95% CI 0.75 to 1.99], P = 0.20). The study thus failed to show a benefit to continuing trastuzumab after progression in the first-line setting.
Lapatinib in combination with paclitaxel has been compared to paclitaxel alone for the treatment of advanced HER2-positive gastric cancer in an Asian population in the phase 3 TyTAN trial.31 With a primary end point of OS, the study randomly assigned 129 patients to receive paclitaxel alone and 132 patients to receive paclitaxel with lapatinib. There was a nonsignificant trend towards improvement in OS in the combination group (11 months) as compared to the paclitaxel-only group(8.9 months, P = 0.1044), with no significant difference in median PFS (5.4 months versus 4.4 months). However, it is important to note that only 15 patients in this trial had previously been exposed to trastuzumab. Another trial, the phase 3 GATSBY study, examined the efficacy of trastuzumab emtansine in the second-line setting compared to taxanes alone and failed to show any improvement in PFS or OS.32 Given these results, no alternative anti-HER2 therapy has been proven to be efficacious for patients who are trastuzumab refractory. Therefore, including anti-HER2 therapy in the second-line treatment of HER2-positive gastric cancer is not recommended.
IMMUNOTHERAPY AND OTHER TARGETED THERAPIES
Several other targeted therapies have been studied in advanced gastric cancer, without any demonstrable survival benefit. The PI3K/AKT/mTOR pathway is known to be involved in regulation of cell growth and angiogenesis, and the mTOR inhibitor everolimus is widely used to treat other malignancies, including breast cancer. The use of everolimus in the second-line setting was studied in the phase 3 GRANITE-1 trial, where it was compared to best supportive care and failed to provide any survival benefit.33 Cetuximab, a recombinant human and mouse chimeric monoclonal antibody, and panitumumab, a recombinant human antibody against the epidermal growth factor receptor (EGFR), have also been examined in gastric and GEJ cancer patients. However, the large phase 3 EXPAND and REAL-3 trials did not show a survival benefit when these agents were added to standard chemotherapy.34,35
Overexpression of MET, a proto-oncogene and tyrosine kinase receptor, has also been implicated in gastric cancer progression. The ligand for MET is the hepatocyte growth factor (HGF), and aberrant signaling of this pathway has been shown to correlate with an aggressive gastric cancer phenotype and poorer OS by promoting tumor growth and angiogenesis. However, no MET inhibitors thus far have been found to be clinically effective. RILOMET-1 and RILOMET-2 were phase 3 trials examining the efficacy of rilotumumab, a humanized anti-HGF antibody, in combination with chemotherapy (ECX and cisplatin with capecitabine, respectively) for the frontline treatment of MET-positive GEJ and gastric cancers. Both studies were discontinued due to a higher treatment-related mortality in patients receiving rilotumumab, with a higher incidence of adverse events due to disease progression being noted.36 Similarly, onartuzumab, a monovalent monoclonal antibody against the MET receptor, was investigated in the phase 3 METGastric trial in combination with modified FOLFOX6 as first-line therapy for HER2-negative, MET-positive metastatic GEJ and gastric cancers. The study did not demonstrate any significant improvements in OS or PFS.37
There has been significant interest in incorporating immunotherapy in the treatment of early and metastatic gastric cancer. Pembrolizumab is the first programmed death receptor (PD-1) inhibitor to be approved for treatment of patients with PD-L1−positive advanced gastric cancer who had previously received 2 or more lines of chemotherapy. Although earlier studies of pembrolizumab in lung cancer utilized the tumor proportion score (TPS) to determine PD-L1 positivity, this was not found to be applicable to gastric cancer. Instead, the combined positive score (CPS) is used in gastric cancer. The CPS evaluates the number of tumor cells and immune cells (macrophages and lymphocytes) that stain positive for PD-L1 relative to all viable tumor cells. Comparatively, the TPS only examines the percentage of viable tumor cells that show complete or partial positive staining for PD-L1. A CPS score of 1 or greater identifies patients who would be suitable candidates for pembrolizumab.
The approval of pembrolizumab was based on the positive findings from the recent KEYNOTE-059 trial.38 The study included 259 patients who had previously received either fluoropyrimidine, cisplatin, or anti-HER2 therapy, with 148 patients (55%) of these patients having PD-L1−positive tumors. The PD-L1 status was determined using a pharmDx Kit, which is now approved by the US Food and Drug Administration to select patients who could benefit from pembrolizumab treatment. CPS was calculated as the number of PD-L1−staining cells divided by the total number of evaluated cells. The study included patients with microsatellite stable (MSI-S), undetermined, or deficient MMR status. The overall response rate to pembrolizumab across all patients was 11.6%, median PFS was 2 months, and the 12-month OS rate was 23.4%. In the subset of patients with MSI-H tumors, the overall response rate was 57.1%, with a complete response rate of 14.3%; in those with MSI-S tumors, the overall response rate was 9% and the complete response rate was 2.4%. Among patients with PD-L1–positive tumors, the overall response rate was 15.5% (95% CI 10.1% to 22.4). Common adverse events included fatigue, hypothyroidism, nausea, diarrhea, and arthralgia.38
CASE CONCLUSION
This patient with metastatic gastric cancer receives second-line chemotherapy with ramucirumab and paclitaxel. Follow-up imaging shows persistent liver metastases and new lung metastasis. Because the tumor is PD-L1–positive, the patient receives 4 cycles of pembrolizumab, with no significant change noted in disease burden. He notes a significant decline in functional status with increased weight loss, nausea, emesis, and fatigue. The patient opts to forego any further therapy and instead chooses to pursue supportive care only.
SUMMARY
Gastric cancer is the third most common cause of cancer death worldwide. Common risk factors for developing gastric cancer include H. pylori infection, smoking, alcohol abuse, radiation exposure, high-fat diet, and obesity. Patients presenting with alarm symptoms of nausea, emesis, early satiety, abdominal pain, or weight loss should be fully evaluated with upper GI endoscopy. If there is suspicion for metastatic disease, CT evaluation of the chest, abdomen, and pelvis with oral and intravenous contrast should be obtained. Treatment of patients with metastatic gastric cancer is guided by their performance status at presentation. For patients with good performance status, a combination of platinum and fluoropyrimidine therapy, such as FOLFOX, can be considered. Doublet chemotherapy regimens are preferred over triplet chemotherapy regimens given their better tolerability. For patients with HER2-positive disease, the addition of trastuzumab to the platinum and fluoropyrimidine backbone is the standard of care in the first line.
Several targeted agents have been studied in patients progressing on initial therapy, with ramucirumab and paclitaxel being considered the regimen of choice in the second line. No anti-HER2 therapy has been approved for patients who are refractory to trastuzumab. Pembrolizumab is approved for use in patients who are PD-L1–positive and have previously progressed on at least 2 lines of chemotherapy. Pembrolizumab is also approved for the treatment of patients with unresectable or metastatic, MSI-H or MMR-deficient gastric cancers that have progressed after prior treatment and who have no satisfactory alternative treatment options.
INTRODUCTION
According to the Surveillance, Epidemiology and End Results database, in 2017 there were 28,000 new cases of gastric cancer, accounting for 1.8% of all malignancies in the United States, and an estimated 10,960 gastric cancer–related deaths.1 Worldwide, gastric cancer is the fifth most common malignancy and the third most common cause of death from any cancer.2 The incidence of gastric cancer varies significantly by geographic region, with countries in Eastern Asia (China, Japan), Eastern Europe, and Central and South America accounting for 50% of all new cases.3 Although the incidence of gastric cancer has declined in recent years, this decrease has not been observed consistently across all nations.2 In particular, the incidence of gastric cancers arising from the cardia has been increasing, which is perhaps due to a higher prevalence of obesity in Western societies.4
In this article, we review key aspects of management of metastatic gastric cancer, including selection of first- and second-line therapy, and discuss targeted agents and upcoming clinical trials.
EPIDEMIOLOGY AND RISK FACTORS
Chronic infection with Helicobacter pylori, a gram-negative bacterium, is a strong etiological factor for the development of gastric cancer, contributing to up to 70% of cases.2 The pathogen can colonize the gastric mucosa, leading to chronic inflammation. Although most patients remain asymptomatic, 1% to 3% develop gastric cancer and another 0.1% develop mucosa-associated lymphoid tissue lymphoma.5 H. pylori infection is more commonly associated with cancer of the gastric body than with cancer of the gastroesophageal junction (GEJ). The increased burden of gastric cancer in countries in Eastern Asia, Latin America, and Eastern Europe has been correlated to the prevalence of chronic H. pylori infection in these areas.
Carcinogenesis secondary to H. pylori infection may occur via several mechanisms. First, H. pylori can release virulence factors, such as cytotoxin-associated gene A, vacuolating cytotoxin, and outer membrane proteins, into the cytosol of host cells, leading to changes in patterns of cell proliferation and apoptosis.6 These virulence factors can modulate the host immune system, attenuating it to promote dysplasia. In addition, continued recognition of these factors by the immune system leads to a persistent inflammatory response, with the release of cytokines such as interleukin (IL) -1β, IL-6, and IL-8. This leads to chronic mucosal damage, further promoting dysplasia with eventual transformation into adenocarcinoma.7 In Japan and Korea, where screening for H. pylori infection is routinely performed, there have been improvements in overall survival (OS) rates for gastric cancer, with 5-year OS rates of 70%.8 The International Agency for Research on Cancer recommends further research into population-based screening and treatment programs for patients with chronic H. pylori infection. However, despite this recommendation, optimal screening strategies are not clearly defined.9
Other risk factors for the development of gastric cancer include chronic gastroesophageal reflux disease; smoking; alcohol use; exposure to radiation; diets high in fats, salt, and smoked items and low in fruits and vegetables; obesity; and exposure to chemotherapeutic agents such as procarbazine.10 Another pathogen suspected, but not proven, to be associated with increased risk for gastric cancer is the Epstein-Barr virus, a human herpesvirus found in 80% of all gastric carcinomas with lymphoid features.11 In addition, whether the use of medications such as statins and nonsteroidal anti-inflammatory drugs confers a decreased risk of gastric cancers remains unclear.10
EVALUATION
CASE PRESENTATION
A 55-year-old Caucasian man with a history of type 2 diabetes mellitus presents to the gastrointestinal (GI) clinic with a 6-month history of dysphagia. The dysphagia is worsened with ingestion of solids, particularly towards the end of the day. He states that the food often gets “stuck in the middle of the chest.” The patient denies any nausea or emesis but notes that he has a poor appetite. He reports having worsening mid-epigastric abdominal pain that is non-radiating, dull in character, and 6/10 in intensity. He also reports a 10-lb weight loss over the past 2 months. He has no previous history of reflux, chest pain, dyspnea, or cough. Review of systems is otherwise benign. Physical exam is within normal limits.
• Which tests should be conducted when gastric cancer is suspected?
Persistent epigastric abdominal pain and weight loss are the most common early symptoms of gastric cancer. Nausea, early satiety, dysphagia, and occult GI bleeding can be other presenting signs. Patients presenting with alarm symptoms of nausea, emesis, early satiety, abdominal pain, or weight loss should be fully evaluated with upper GI endoscopy. Early diagnosis of gastric cancer is essential in obtaining a curative resection. However, at least 40% of patients present with de novo metastatic disease at the time of initial diagnosis.12 Gastric cancer spreads by direct extension through the gastric wall, with the liver, peritoneum, and regional lymph nodes being the most common sites of metastatic deposits.13 Classically, Virchow’s node, the left supraclavicular lymph node, is involved with metastatic gastric cancer. Involvement of the left axillary lymph node (Irish node) or a periumbilical nodule (Sister Mary Joseph node) may also be observed. Other, less commonly noted sites of metastatic disease include the ovaries, central nervous system, bone, lung, and soft tissues.13
Upper GI endoscopy is the best method for determining tumor location and extent and obtaining a specimen for a definitive tissue diagnosis.14 It is essential to accurately identify the location of the tumor in the stomach and relative to the GEJ. The American Joint Committee on Cancer classification defines tumors involving the GEJ with an epicenter no more than 2 cm into the proximal stomach as esophageal cancers.15 Tumors of the GEJ with their epicenter more than 2 cm into the proximal stomach are defined as gastric cancers. If metastatic disease is suspected, computed tomography (CT) scan of the chest, abdomen, and pelvis with oral and intravenous contrast can be obtained to determine the extent of disease spread. In the absence of any metastatic disease, endoscopic ultrasound (EUS) should be conducted to determine the depth of tumor invasion (T staging) and lymph node status. In the era of targeted therapy, patients with metastatic disease should undergo testing for human epidermal growth factor-2 (HER-2) expression, microsatellite instability (MSI), and programmed death ligand 1 (PD-L1) expression. Patients should be staged according to the TNM staging system.
FIRST-LINE TREATMENT OPTIONS
CASE CONTINUED
The patient undergoes esophagoduodenoscopy (EGD) and is found to have a gastric cardia mass extending into the distal esophagus. EUS also demonstrates multiple abdominal and mediastinal lymph nodes. No gastric outlet obstruction is found. Biopsy shows poorly differentiated invasive adenocarcinoma. Warthin–Starry stain is negative for H. pylori organism. The tumor cells are positive for cytokeratin (CK7), CK19, and mucin-1 gene (MUC1); focally positive for CK20; and negative for MUC2. HER2 testing results are reported as immunohistochemistry (IHC) 3+, consistent with strongly positive HER2 protein expression. Further IHC testing for mismatch repair (MMR) proteins shows intact nuclear expression of MLH1, MSH2, MSH6, and PMS2 protein, consistent with a low probability of MSI-high tumor. The tumor is found to be PD-L1 positive. Imaging reveals abnormal mass-like nodular thickening of the gastric wall, with an infiltrative opacity within the pancreatico-duodenal groove, suspicious for tumor infiltration. Multiple metastatic deposits are noted in the liver, peritoneum, and bilateral lungs. There is extensive gastrohepatic ligament and periportal lymphadenopathy and mild enlargement of the pulmonary hilar lymph nodes. These findings are consistent with stage 4 (T4bN3aM1) gastric cancer. Given these findings, staging laparoscopy is deferred.
• What are the first-line treatment options for patients with metastatic gastric cancer?
Patients with metastatic gastric cancer have a poor prognosis, and management is stratified based on performance status (Figure). In patients with good performance status, systemic chemotherapy is the mainstay of treatment. The goal of therapy is not curative, but rather treatment focuses on palliation of symptoms arising from tumor spread. Given this treatment goal, there has been considerable interest in clarifying the utility of chemotherapy as opposed to best supportive care. In a recent Cochrane review of 64 randomized control trials involving 11,698 patients, chemotherapy was found to improve OS by 6.7 months as compared to best supportive care (hazard ratio [HR] 0.3 [95% confidence interval {CI} 0.24 to 0.55]).16 Five classes of cytotoxic chemotherapeutic agents have demonstrated activity in gastric cancer. These include fluoropyrimidine (either infusional fluorouracil or capecitabine), platinum agents (cisplatin or oxaliplatin), taxanes (docetaxel or paclitaxel), anthracyclines (epirubicin), and irinotecan.13 Treatment options are further divided based on whether the patient has HER2-overexpressing or non-expressing malignancy.
HER2-NEGATIVE DISEASE
For patients with HER2-negative disease, National Comprehensive Cancer Network (NCCN) guidelines recommend using 2-drug combination regimens rather than 3 drugs, given concern for increased toxicity with 3-drug regimens.17 For patients with a performance status of 0 to 1, utilization of a 3-drug regimen is a reasonable alternative. The combination of a fluoropyrimidine with a platinum agent is considered the standard of care, with regimens such as fluorouracil, leucovorin, and oxaliplatin (FOLFOX) being commonly used.
Epirubicin-containing regimens have also been extensively studied in advanced gastric cancer. In a study of 274 previously untreated patients with GEJ cancers, the combination of epirubicin, cisplatin, and fluorouracil (ECF) was compared to fluorouracil, doxorubicin, and methotrexate (FAMTX). There was an OS benefit favoring ECF (8.9 months versus 5.7 months) at 1 year (95% CI 27% to 45%, P = 0.0009). The ECF regimen was associated with an increased risk of nausea, emesis, and alopecia, while more hematologic toxicity and infections were noted with the FAMTX regimen.18 In addition, in a phase 3 trial, Van Cutsem and colleagues examined the role of docetaxel in combination with cisplatin and fluorouracil (DCF) compared to cisplatin and fluorouracil alone. Addition of docetaxel led to improved OS and time to progression (9.2 months versus 8.6 months for cisplatin and fluorouracil alone, P = 0.02) but with an increased risk of grade 3 and 4 toxicities (69% versus 59%). These adverse events included neutropenia (82% versus 57% of cisplatin and fluorouracil patients), diarrhea (19% versus 8%), stomatitis (21% versus 27%), and fatigue (19% versus 14%).19
The landmark phase 3 REAL-2 study compared 4 chemotherapy regimens in patients with untreated advanced esophagogastric cancer. This study was conducted to determine if the efficacy of cisplatin and oxaliplatin, a third-generation platinum agent, is equivalent to that of fluorouracil and capecitabine, an oral fluoropyrimidine. In this trial, a 2 × 2 design was used to compare 4 regimens: ECF versus epirubicin, cisplatin, and capecitabine (ECX) versus epirubicin, oxaliplatin, and fluorouracil (EOF) versus epirubicin, oxaliplatin, and capecitabine (EOX). The study found EOX to be noninferior to ECF, with a trend towards improved OS compared to other combination regimens (11.2 months versus 9.9 months, HR 0.80 [95% CI 0.66 to 0.97], P = 0.02).20 Thus, the study demonstrated that an oxaliplatin and capecitabine-based regimen could replace cisplatin and fluorouracil. Given that fluorouracil administration requires long continuous infusions, the oral-based capecitabine regimen is an attractive option for patients.
Several trials have demonstrated the equivalency of oxaliplatin with cisplatin in combination regimens for the treatment of advanced gastric cancer. Oxaliplatin has the benefit of an improved toxicity profile as compared to cisplatin, with the major dose-limiting toxicity being peripheral neuropathy
Given previous evidence that DCF (docetaxel, cisplatin, fluorouracil) is superior to cisplatin and fluorouracil alone, there was interest in determining if the addition of docetaxel to a backbone of fluorouracil, oxaliplatin, and leucovorin (FLO) could elicit a higher response rate. This concept was investigated in a phase 2 trial that assigned 54 patients with metastatic gastric or GEJ adenocarcinoma to receive biweekly infusions of oxaliplatin, leucovorin, fluorouracil, and docetaxel.21 Median time to response was 1.54 months, and the overall response rate was 57.7%. Median progression-free survival (PFS) was 5.2 months, and OS was 11.1 months. The most common grade 3 or 4 toxicities included neutropenia (48%), leukopenia (27.8%), diarrhea (14.8%), and fatigue (11.1%).
Irinotecan-based regimens have also been extensively studied in the first-line treatment of metastatic gastric cancer, particularly as an alternative to platinum-based therapy, but superiority has not been established. The combination of fluorouracil, leucovorin, and irinotecan (FOLFIRI) was compared to ECX in a phase 3 trial.22 The study enrolled 416 patients with locally advanced or metastatic gastric or GEJ cancer. At a median follow up of 31 months, the time to progression was longer in the FOLFIRI arm as compared to the ECX arm (5.1 months versus 4.2 months, P = 0.008), but there was no difference in OS (9.5 months versus 9.7 months, P = 0.95), median PFS (5.3 months versus 5.8 months, P = 0.96), or response rate (39.2% versus 37.8%). However, the FOLFIRI regimen had an improved toxicity profile, with a lower overall rate of grade 3 or 4 toxicity (69% versus 84%, P < 0.001). Given these findings, the FOLFIRI regimen is an acceptable alternative to platinum-based therapy in suitable patients.22
HER2-POSITIVE DISEASE
The HER2 proto-oncogene, initially described in breast cancer, has been implicated in several malignancies, including gastric and esophageal cancer. Overexpression or amplification of HER2 can be found in up to 30% of gastric cancers.23 For these patients, adding trastuzumab to a standard regimen of platinum and fluoropyrimidine is the standard of care. The prospective phase 3 Trastuzumab for Gastric Cancer (ToGA) trial randomly assigned 594 patients with HER2-positive gastric cancer to receive either cisplatin and fluorouracil or capecitabine and cisplatin with trastuzumab (n = 294) or without (n = 290) trastuzumab every 3 weeks for a total of 6 cycles, followed by maintenance trastuzumab until disease progression was noted.24 HER2 positivity was defined as HER2 protein overexpression by IHC (cutoff of 3+) or gene amplification by fluorescence in situ hybridization (FISH); tumors with IHC 2+ patterns were followed with FISH studies to confirm positivity. The study found a higher incidence of HER2-positive tumors in patients with GEJ tumors compared to patients with distal gastric cancers (33% versus 20%).24 In this trial, the addition of trastuzumab was associated with an improvement in OS: 13.5 months in the trastuzumab cohort versus 11.1 months in those receiving chemotherapy alone (HR 0.74, P = 0.0048). There was not a significant difference in toxicities between the 2 cohorts, with nausea, emesis, and neutropenia being the most common adverse events. Rates of overall grade 3 or 4 events were similar as well (68% in each cohort). Further exploratory analysis was also conducted according to HER2 status by dividing patients into a “high-expressor” group (n = 446), defined as patients with IHC 3+ tumors or IHC 2+ and FISH positivity, and a “low-expressor” group (n = 131), which included patients with IHC 0 or 1+ tumors. Analysis of patients in the 2 subgroups demonstrated an improved OS with the addition of trastuzumab for the high-expressor cohort, with a median OS of 16 months (HR 0.65 [95% CI 0.51 to 0.83]) compared to 11.8 months in those receiving only chemotherapy.
Dual HER2 blockade has been investigated in metastatic gastric cancer. The phase 3 randomized JACOB trial assigned 780 patients to receive either trastuzumab with a cisplatin/fluoropyrimidine regimen with or without the addition of pertuzumab; the primary end point was OS.25 A non-statistically significant trend towards improvement in OS was found in the pertuzumab arm (17.5 months) as compared with the standard of care arm (14.2 months, HR 0.84, P = 0.0565). The pertuzumab/trastuzumab/chemotherapy cohort experienced a higher incidence of diarrhea (61.6% versus 35.1% in control arm). Cardiac toxicity was comparable in the 2 cohorts.
The Table provides a summary of relevant clinical trials in metastatic gastric cancer.
SECOND-LINE THERAPY
CASE CONTINUED
The patient receives capecitabine, oxaliplatin, and trastuzumab therapy for 6 cycles, followed by trastuzumab for another 3 cycles. While on therapy, he develops a painful right clavicular lesion. He undergoes magnetic resonance imaging of the right clavicle, which shows a lesion in the distal two-thirds of the right clavicle measuring 9.7 × 3.7 × 3.8 cm. The patient is started on palliative radiation to the clavicle. However, repeat CT imaging shows progressive liver metastases.
• What is the approach to second-line therapy for metastatic gastric cancer?
Improvements in our understanding of the molecular pathways that lead to tumorigenesis have contributed to the development of several targeted agents whose efficacy in gastric cancer is being investigated. The NCCN guidelines recommend that for all patients who progress on frontline therapy, second-line therapy consists of a combination of ramucirumab and paclitaxel. Other options include single-agent docetaxel, paclitaxel, irinotecan, or ramucirumab. Combination therapy using irinotecan with either docetaxel, fluorouracil, or cisplatin may also be used.
Ramucirumab, a human IgG1 monoclonal antibody that targets the vascular endothelial growth factor receptor 2 (VEGFR2), was initially approved in 2014 as monotherapy for patients who had previously progressed on first-line chemotherapy. Its approval was based on the results of the phase 3 randomized, double-blind placebo-controlled REGARD study.26 The trial randomly assigned 355 patients with advanced gastric or GEJ adenocarcinoma and disease progression after first-line platinum-containing or fluoropyrimidine-containing chemotherapy to receive best supportive care plus either ramucirumab (n = 238) or placebo (n = 117). Monotherapy with ramucirumab significantly improved median OS compared with placebo (5.2 months versus. 3.8 months; HR 0.776 [95% CI 0.6 to 0.99], P = 0.047). There was also an improvement in PFS of 2.1 months in the ramucirumab cohort, as compared to 1.3 months in the placebo cohort (P < 0.0001). Patients in the ramucirumab arm experienced a higher incidence of hypertension (16% versus 8%), but all other adverse events occurred at comparable rates. Five deaths in the ramucirumab group were thought to be secondary to the study drug, as compared to 2 deaths in the placebo group.
In the subsequent phase 3 RAINBOW trial, the addition of ramucirumab to paclitaxel was investigated, with 330 patients assigned to the combination group and 335 to the paclitaxel-only group.27 The trial again showed that combination therapy afforded patients a significant survival advantage compared to paclitaxel alone, with a median OS of 9.6 months versus 7.4 months for the monotherapy group (HR 0.807 [95% CI 0.678 to 0.962], P = 0.017). A PFS benefit of 4.4 months was observed in the combination therapy groups, as compared with 2.9 months in the monotherapy group (HR 0.635, P < 0.0001). The ramucirumab/paclitaxel group also had a higher overall response rate of 28% versus 16%. The combination cohort had an increased incidence of grade 3 or higher adverse hypertensive events (14% versus 2%) and neutropenia (41% versus 19%), while the incidence of grade 3 febrile neutropenic events was similar between the groups (3% versus 2%).
The addition of bevacizumab, another monoclonal antibody against VEGF, to standard chemotherapy regimens has been explored, but studies have failed to show a survival benefit with this agent in the first-line treatment of advanced gastric cancer. The phase 3 Avastin in Gastric Cancer (AVAGAST) trial was a multinational, randomized study where patients received either bevacizumab (n = 387) or placebo (n = 387) in addition to cisplatin and capecitabine.28 The substitution of fluorouracil for capecitabine was permitted for patients who were unable to tolerate oral medications. Cisplatin was administered for a maximum of 6 cycles, while capecitabine and bevacizumab were administered until disease progression. The study failed to show an improvement in OS, with a median OS of 12.1 months noted in the bevacizumab cohort, as compared to 10.1 months in the placebo arm (HR 0.87 [95% CI 0.73 to 1.03], P = 0.1002). However, there was a modest improvement in median PFS (6.7 months versus 5.3 months; HR 0.80 [95% CI 0.68 to 0.93], P = 0.0037) and overall response rate (46% versus 37.4%, P = 0.0315). The most commonly reported grade 3 to 5 adverse events included neutropenia (35%), anemia (10%), and loss of appetite (8%). Interestingly, in a follow-up report, higher serum levels of VEGF-A were thought to correlate with an enhanced response to bevacizumab.29 However, the routine use of biomarker analysis in selecting patients for treatment with bevacizumab in metastatic gastric cancer remains to be further clarified.
Use of other agents with anti-HER2 activity in the second-line treatment of patients who have experienced progression while on trastuzumab remains unclear. In the recent T-ACT trial, patients with disease refractory to frontline therapy with combination trastuzumab and fluoropyrimidine/platinum agents were randomly assigned to receive either weekly paclitaxel (n = 45) or weekly paclitaxel plus trastuzumab (n = 44).30 Patients in the combination cohort received an initial dose of trastuzumab 8 mg/kg followed by 6 mg/kg every 3 weeks until progression. The study did not find a difference in either PFS (3.19 months versus 3.68 months; HR 0.91 [95% CI 0.67 to 1.22], P = 0.33) or OS (9.95 months versus 10.2 months; HR 1.23 [95% CI 0.75 to 1.99], P = 0.20). The study thus failed to show a benefit to continuing trastuzumab after progression in the first-line setting.
Lapatinib in combination with paclitaxel has been compared to paclitaxel alone for the treatment of advanced HER2-positive gastric cancer in an Asian population in the phase 3 TyTAN trial.31 With a primary end point of OS, the study randomly assigned 129 patients to receive paclitaxel alone and 132 patients to receive paclitaxel with lapatinib. There was a nonsignificant trend towards improvement in OS in the combination group (11 months) as compared to the paclitaxel-only group(8.9 months, P = 0.1044), with no significant difference in median PFS (5.4 months versus 4.4 months). However, it is important to note that only 15 patients in this trial had previously been exposed to trastuzumab. Another trial, the phase 3 GATSBY study, examined the efficacy of trastuzumab emtansine in the second-line setting compared to taxanes alone and failed to show any improvement in PFS or OS.32 Given these results, no alternative anti-HER2 therapy has been proven to be efficacious for patients who are trastuzumab refractory. Therefore, including anti-HER2 therapy in the second-line treatment of HER2-positive gastric cancer is not recommended.
IMMUNOTHERAPY AND OTHER TARGETED THERAPIES
Several other targeted therapies have been studied in advanced gastric cancer, without any demonstrable survival benefit. The PI3K/AKT/mTOR pathway is known to be involved in regulation of cell growth and angiogenesis, and the mTOR inhibitor everolimus is widely used to treat other malignancies, including breast cancer. The use of everolimus in the second-line setting was studied in the phase 3 GRANITE-1 trial, where it was compared to best supportive care and failed to provide any survival benefit.33 Cetuximab, a recombinant human and mouse chimeric monoclonal antibody, and panitumumab, a recombinant human antibody against the epidermal growth factor receptor (EGFR), have also been examined in gastric and GEJ cancer patients. However, the large phase 3 EXPAND and REAL-3 trials did not show a survival benefit when these agents were added to standard chemotherapy.34,35
Overexpression of MET, a proto-oncogene and tyrosine kinase receptor, has also been implicated in gastric cancer progression. The ligand for MET is the hepatocyte growth factor (HGF), and aberrant signaling of this pathway has been shown to correlate with an aggressive gastric cancer phenotype and poorer OS by promoting tumor growth and angiogenesis. However, no MET inhibitors thus far have been found to be clinically effective. RILOMET-1 and RILOMET-2 were phase 3 trials examining the efficacy of rilotumumab, a humanized anti-HGF antibody, in combination with chemotherapy (ECX and cisplatin with capecitabine, respectively) for the frontline treatment of MET-positive GEJ and gastric cancers. Both studies were discontinued due to a higher treatment-related mortality in patients receiving rilotumumab, with a higher incidence of adverse events due to disease progression being noted.36 Similarly, onartuzumab, a monovalent monoclonal antibody against the MET receptor, was investigated in the phase 3 METGastric trial in combination with modified FOLFOX6 as first-line therapy for HER2-negative, MET-positive metastatic GEJ and gastric cancers. The study did not demonstrate any significant improvements in OS or PFS.37
There has been significant interest in incorporating immunotherapy in the treatment of early and metastatic gastric cancer. Pembrolizumab is the first programmed death receptor (PD-1) inhibitor to be approved for treatment of patients with PD-L1−positive advanced gastric cancer who had previously received 2 or more lines of chemotherapy. Although earlier studies of pembrolizumab in lung cancer utilized the tumor proportion score (TPS) to determine PD-L1 positivity, this was not found to be applicable to gastric cancer. Instead, the combined positive score (CPS) is used in gastric cancer. The CPS evaluates the number of tumor cells and immune cells (macrophages and lymphocytes) that stain positive for PD-L1 relative to all viable tumor cells. Comparatively, the TPS only examines the percentage of viable tumor cells that show complete or partial positive staining for PD-L1. A CPS score of 1 or greater identifies patients who would be suitable candidates for pembrolizumab.
The approval of pembrolizumab was based on the positive findings from the recent KEYNOTE-059 trial.38 The study included 259 patients who had previously received either fluoropyrimidine, cisplatin, or anti-HER2 therapy, with 148 patients (55%) of these patients having PD-L1−positive tumors. The PD-L1 status was determined using a pharmDx Kit, which is now approved by the US Food and Drug Administration to select patients who could benefit from pembrolizumab treatment. CPS was calculated as the number of PD-L1−staining cells divided by the total number of evaluated cells. The study included patients with microsatellite stable (MSI-S), undetermined, or deficient MMR status. The overall response rate to pembrolizumab across all patients was 11.6%, median PFS was 2 months, and the 12-month OS rate was 23.4%. In the subset of patients with MSI-H tumors, the overall response rate was 57.1%, with a complete response rate of 14.3%; in those with MSI-S tumors, the overall response rate was 9% and the complete response rate was 2.4%. Among patients with PD-L1–positive tumors, the overall response rate was 15.5% (95% CI 10.1% to 22.4). Common adverse events included fatigue, hypothyroidism, nausea, diarrhea, and arthralgia.38
CASE CONCLUSION
This patient with metastatic gastric cancer receives second-line chemotherapy with ramucirumab and paclitaxel. Follow-up imaging shows persistent liver metastases and new lung metastasis. Because the tumor is PD-L1–positive, the patient receives 4 cycles of pembrolizumab, with no significant change noted in disease burden. He notes a significant decline in functional status with increased weight loss, nausea, emesis, and fatigue. The patient opts to forego any further therapy and instead chooses to pursue supportive care only.
SUMMARY
Gastric cancer is the third most common cause of cancer death worldwide. Common risk factors for developing gastric cancer include H. pylori infection, smoking, alcohol abuse, radiation exposure, high-fat diet, and obesity. Patients presenting with alarm symptoms of nausea, emesis, early satiety, abdominal pain, or weight loss should be fully evaluated with upper GI endoscopy. If there is suspicion for metastatic disease, CT evaluation of the chest, abdomen, and pelvis with oral and intravenous contrast should be obtained. Treatment of patients with metastatic gastric cancer is guided by their performance status at presentation. For patients with good performance status, a combination of platinum and fluoropyrimidine therapy, such as FOLFOX, can be considered. Doublet chemotherapy regimens are preferred over triplet chemotherapy regimens given their better tolerability. For patients with HER2-positive disease, the addition of trastuzumab to the platinum and fluoropyrimidine backbone is the standard of care in the first line.
Several targeted agents have been studied in patients progressing on initial therapy, with ramucirumab and paclitaxel being considered the regimen of choice in the second line. No anti-HER2 therapy has been approved for patients who are refractory to trastuzumab. Pembrolizumab is approved for use in patients who are PD-L1–positive and have previously progressed on at least 2 lines of chemotherapy. Pembrolizumab is also approved for the treatment of patients with unresectable or metastatic, MSI-H or MMR-deficient gastric cancers that have progressed after prior treatment and who have no satisfactory alternative treatment options.
1. Noone AM, Cronin KA, Altekruse SF, et al. Cancer incidence and survival trends by subtype using data from the Surveillance Epidemiology and End Results Program, 1992-2013. Cancer Epidemiol Biomarkers Prev 2017;26:632–41
2. Global Burden of Disease Cancer Collaboration, Fitzmaurice C, Allen C, et al. Global, regional, and national cancer incidence, mortality, years of life lost, years lived with disability, and disability-adjusted life-years for 32 cancer groups, 1990 to 2015: a systematic analysis for the global burden of disease study. JAMA Oncol 2017;3:524–8.
3. Sitarz R, Skierucha M, Mielko J, et al. Gastric cancer: epidemiology, prevention, classification, and treatment. Cancer Manag Res 2018;10:239–48.
4. Olefson S, Moss SF. Obesity and related risk factors in gastric cardia adenocarcinoma. Gastric Cancer 2015;18:23–32.
5. Wang F, Meng W, Wang B, Qiao L. Helicobacter pylori-induced gastric inflammation and gastric cancer. Cancer Lett 2014;345:196–202.
6. Espinoza JL, Matsumoto A, Tanaka H, Matsumura I. Gastric microbiota: An emerging player in Helicobacter pylori-induced gastric malignancies. Cancer Lett 2018;414:147–52.
7. Chmiela M, Gonciarz W. Molecular mimicry in Helicobacter pylori infections. World J Gastroenterol 2017;23:3964–77.
8. Isobe Y, Nashimoto A, Akazawa K, et al. Gastric cancer treatment in Japan: 2008 annual report of the JGCA nationwide registry. Gastric Cancer 2011;14:301–16.
9. Ford A, Gurusamy KS, Delaney B, et al. Eradication therapy for peptic ulcer disease in Helicobacter pylori-positive patients. Cochrane Database Syst Rev 2004(4):CD003840.
10. Karimi P, Islami F, Anandasabapathy S, et al. Gastric cancer: descriptive epidemiology, risk factors, screening, and prevention. Cancer Epidemiol Biomarkers Prev 2014;23:700–13.
11. Van Cutsem E, Sagaert X, Topal B, et al. Gastric cancer. Lancet 2016;388:2654–64.
12. Chan BA , Sim HW, Natori A, et al. Survival outcomes for de novo versus relapsed stage IV gastric and gastroesophageal junction (GEJ) adenocarcinoma [abstract]. J Clin Oncol 2018;36(no. 4 suppl):148.
13. DeVita VT, Lawrence TS, Rosenberg SA. DeVita, Hellman, and Rosenberg’s cancer: principles & practice of oncology. 9th ed. Philadelphia: Wolters Kluwer Health/Lippincott Williams & Wilkins; 2011.
14. Siewert JR, Hölscher AH, Becker K, Gössner W. [Cardia cancer: attempt at a therapeutically relevant classification]. [Article in German.] Chirurg 1987;58:25–32.
15. Amin MB, Edge SB, Greene FL, et al, eds. AJCC cancer staging manual. 8th ed. New York: Springer; 2017.
16. Wagner AD, Syn NL, Moehler M, et al. Chemotherapy for advanced gastric cancer. Cochrane Database Syst Rev 2017;8:CD004064.
17. Qiu H, Zhou Z. [Updates and interpretation on NCCN clinical practice guidelines for gastric cancer 2017 version 5]. [Article in Chinese.] Zhonghua Wei Chang Wai Ke Za Zhi 2018;21:160–4.
18. Webb A, Cunningham D, Scarffe JH, et al. Randomized trial comparing epirubicin, cisplatin, and fluorouracil versus fluorouracil, doxorubicin, and methotrexate in advanced esophagogastric cancer. J Clin Oncol 1997;15:261–7.
19. Van Cutsem E, Moiseyenko VM, Tjulandin S, et al. Phase III study of docetaxel and cisplatin plus fluorouracil compared with cisplatin and fluorouracil as first-line therapy for advanced gastric cancer: a report of the V325 Study Group. J Clin Oncol 2006;24:4991–7.
20. Cunningham D, Okines AF, Ashley S. Capecitabine and oxaliplatin for advanced esophagogastric cancer. N Engl J Med 2010;362:858–9.
21. Al-Batran SE, Hartmann JT, Hofheinz R, et al. Biweekly fluorouracil, leucovorin, oxaliplatin, and docetaxel (FLOT) for patients with metastatic adenocarcinoma of the stomach or esophagogastric junction: a phase II trial of the Arbeitsgemeinschaft Internistische Onkologie. Ann Oncol 2008;19:1882–7.
22. Guimbaud R, Louvet C, Ries P, et al. Prospective, randomized, multicenter, phase III study of fluorouracil, leucovorin, and irinotecan versus epirubicin, cisplatin, and capecitabine in advanced gastric adenocarcinoma: a French intergroup (Federation Francophone de Cancerologie Digestive, Federation Nationale des Centres de Lutte Contre le Cancer, and Groupe Cooperateur Multidisciplinaire en Oncologie) study. J Clin Oncol 2014;32:3520–6.
23. Boku N. HER2-positive gastric cancer. Gastric Cancer 2014;17:1–12.
24. Bang YJ, Van Cutsem E, Feyereislova A, et al. Trastuzumab in combination with chemotherapy versus chemotherapy alone for treatment of HER2-positive advanced gastric or gastro-oesophageal junction cancer (ToGA): a phase 3, open-label, randomised controlled trial. Lancet 2010;376:687–97.
25. Tabernero J, Hoff PM, Shen L, et al. Pertuzumab + trastuzumab + chemotherapy for HER2-positive metastatic gastric or gastro-oesophageal junction cancer: Final analysis of a Phase III study (JACOB) [abstract]. Ann Oncol 2017;28(suppl 5):6160.
26. Fuchs CS, Tomasek J, Yong CJ, et al. Ramucirumab monotherapy for previously treated advanced gastric or gastro-oesophageal junction adenocarcinoma (REGARD): an international, randomised, multicentre, placebo-controlled, phase 3 trial. Lancet 2014;383:31–9.
27. Wilke H, Muro K, Van Cutsem E, et al. Ramucirumab plus paclitaxel versus placebo plus paclitaxel in patients with previously treated advanced gastric or gastro-oesophageal junction adenocarcinoma (RAINBOW): a double-blind, randomised phase 3 trial. Lancet Oncol 2014;15:1224–35.
28. Ohtsu A, Shah MA, Van Cutsem E, et al. Bevacizumab in combination with chemotherapy as first-line therapy in advanced gastric cancer: a randomized, double-blind, placebo-controlled phase III study. J Clin Oncol 2011;29:3968–76.
29. Van Cutsem E, de Haas S, Kang YK, et al, Bevacizumab in combination with chemotherapy as first-line therapy in advanced gastric cancer: a biomarker evaluation from the AVAGAST randomized phase III trial. J Clin Oncol 2012;30:2119–27.
30. Makiyama A, Sagara K, Kawada J, et al. A randomized phase II study of weekly paclitaxel ± trastuzumab in patients with HER2-positive advanced gastric or gastro-esophageal junction cancer refractory to trastuzumab combined with fluoropyrimidine and platinum: WJOG7112G (T-ACT) [abstract]. J Clin Oncol 2018;36(no. 15 suppl):4011.
31. Satoh T, Xu RH, Chung HC, et al. Lapatinib plus paclitaxel versus paclitaxel alone in the second-line treatment of HER2-amplified advanced gastric cancer in Asian populations: TyTAN--a randomized, phase III study. J Clin Oncol 2014;32:2039–49.
32. Thuss-Patience PC, Shah MA, Ohtsu A, et al. Trastuzumab emtansine versus taxane use for previously treated HER2-positive locally advanced or metastatic gastric or gastro-oesophageal junction adenocarcinoma (GATSBY): an international randomised, open-label, adaptive, phase 2/3 study. Lancet Oncol 2017;18:640–53.
33. Ohtsu A, Ajani JA, Bai YX, et al. Everolimus for previously treated advanced gastric cancer: results of the randomized, double-blind, phase III GRANITE-1 study. J Clin Oncol 2013;31:3935–43.
34. Lordick F, Kang YK, Chung HC, et al. Capecitabine and cisplatin with or without cetuximab for patients with previously untreated advanced gastric cancer (EXPAND): a randomised, open-label phase 3 trial. Lancet Oncol 2013;14:490–9.
35. Waddell T, Chau I, Cunningham D, et al. Epirubicin, oxaliplatin, and capecitabine with or without panitumumab for patients with previously untreated advanced oesophagogastric cancer (REAL3): a randomised, open-label phase 3 trial. Lancet Oncol 2013;14:481–9.
36. Catenacci DVT, Tebbutt NC, Davidenko I, et al. Rilotumumab plus epirubicin, cisplatin, and capecitabine as first-line therapy in advanced MET-positive gastric or gastro-oesophageal junction cancer (RILOMET-1): a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Oncol 2017;18:1467–82.
37. Shah MA, Bang YJ, Lordick F, et al. Effect of fluorouracil, leucovorin, and oxaliplatin with or without onartuzumab in HER2-negative, MET-positive gastroesophageal adenocarcinoma: the METGastric randomized clinical trial. JAMA Oncol 2017;3:620–7.
38. Fuchs CS, Doi T, Jang RW, et al. Safety and efficacy of pembrolizumab monotherapy in patients with previously treated advanced gastric and gastroesophageal junction cancer: phase 2 clinical KEYNOTE-059 trial. JAMA Oncol 2018;4(5):e180013.
1. Noone AM, Cronin KA, Altekruse SF, et al. Cancer incidence and survival trends by subtype using data from the Surveillance Epidemiology and End Results Program, 1992-2013. Cancer Epidemiol Biomarkers Prev 2017;26:632–41
2. Global Burden of Disease Cancer Collaboration, Fitzmaurice C, Allen C, et al. Global, regional, and national cancer incidence, mortality, years of life lost, years lived with disability, and disability-adjusted life-years for 32 cancer groups, 1990 to 2015: a systematic analysis for the global burden of disease study. JAMA Oncol 2017;3:524–8.
3. Sitarz R, Skierucha M, Mielko J, et al. Gastric cancer: epidemiology, prevention, classification, and treatment. Cancer Manag Res 2018;10:239–48.
4. Olefson S, Moss SF. Obesity and related risk factors in gastric cardia adenocarcinoma. Gastric Cancer 2015;18:23–32.
5. Wang F, Meng W, Wang B, Qiao L. Helicobacter pylori-induced gastric inflammation and gastric cancer. Cancer Lett 2014;345:196–202.
6. Espinoza JL, Matsumoto A, Tanaka H, Matsumura I. Gastric microbiota: An emerging player in Helicobacter pylori-induced gastric malignancies. Cancer Lett 2018;414:147–52.
7. Chmiela M, Gonciarz W. Molecular mimicry in Helicobacter pylori infections. World J Gastroenterol 2017;23:3964–77.
8. Isobe Y, Nashimoto A, Akazawa K, et al. Gastric cancer treatment in Japan: 2008 annual report of the JGCA nationwide registry. Gastric Cancer 2011;14:301–16.
9. Ford A, Gurusamy KS, Delaney B, et al. Eradication therapy for peptic ulcer disease in Helicobacter pylori-positive patients. Cochrane Database Syst Rev 2004(4):CD003840.
10. Karimi P, Islami F, Anandasabapathy S, et al. Gastric cancer: descriptive epidemiology, risk factors, screening, and prevention. Cancer Epidemiol Biomarkers Prev 2014;23:700–13.
11. Van Cutsem E, Sagaert X, Topal B, et al. Gastric cancer. Lancet 2016;388:2654–64.
12. Chan BA , Sim HW, Natori A, et al. Survival outcomes for de novo versus relapsed stage IV gastric and gastroesophageal junction (GEJ) adenocarcinoma [abstract]. J Clin Oncol 2018;36(no. 4 suppl):148.
13. DeVita VT, Lawrence TS, Rosenberg SA. DeVita, Hellman, and Rosenberg’s cancer: principles & practice of oncology. 9th ed. Philadelphia: Wolters Kluwer Health/Lippincott Williams & Wilkins; 2011.
14. Siewert JR, Hölscher AH, Becker K, Gössner W. [Cardia cancer: attempt at a therapeutically relevant classification]. [Article in German.] Chirurg 1987;58:25–32.
15. Amin MB, Edge SB, Greene FL, et al, eds. AJCC cancer staging manual. 8th ed. New York: Springer; 2017.
16. Wagner AD, Syn NL, Moehler M, et al. Chemotherapy for advanced gastric cancer. Cochrane Database Syst Rev 2017;8:CD004064.
17. Qiu H, Zhou Z. [Updates and interpretation on NCCN clinical practice guidelines for gastric cancer 2017 version 5]. [Article in Chinese.] Zhonghua Wei Chang Wai Ke Za Zhi 2018;21:160–4.
18. Webb A, Cunningham D, Scarffe JH, et al. Randomized trial comparing epirubicin, cisplatin, and fluorouracil versus fluorouracil, doxorubicin, and methotrexate in advanced esophagogastric cancer. J Clin Oncol 1997;15:261–7.
19. Van Cutsem E, Moiseyenko VM, Tjulandin S, et al. Phase III study of docetaxel and cisplatin plus fluorouracil compared with cisplatin and fluorouracil as first-line therapy for advanced gastric cancer: a report of the V325 Study Group. J Clin Oncol 2006;24:4991–7.
20. Cunningham D, Okines AF, Ashley S. Capecitabine and oxaliplatin for advanced esophagogastric cancer. N Engl J Med 2010;362:858–9.
21. Al-Batran SE, Hartmann JT, Hofheinz R, et al. Biweekly fluorouracil, leucovorin, oxaliplatin, and docetaxel (FLOT) for patients with metastatic adenocarcinoma of the stomach or esophagogastric junction: a phase II trial of the Arbeitsgemeinschaft Internistische Onkologie. Ann Oncol 2008;19:1882–7.
22. Guimbaud R, Louvet C, Ries P, et al. Prospective, randomized, multicenter, phase III study of fluorouracil, leucovorin, and irinotecan versus epirubicin, cisplatin, and capecitabine in advanced gastric adenocarcinoma: a French intergroup (Federation Francophone de Cancerologie Digestive, Federation Nationale des Centres de Lutte Contre le Cancer, and Groupe Cooperateur Multidisciplinaire en Oncologie) study. J Clin Oncol 2014;32:3520–6.
23. Boku N. HER2-positive gastric cancer. Gastric Cancer 2014;17:1–12.
24. Bang YJ, Van Cutsem E, Feyereislova A, et al. Trastuzumab in combination with chemotherapy versus chemotherapy alone for treatment of HER2-positive advanced gastric or gastro-oesophageal junction cancer (ToGA): a phase 3, open-label, randomised controlled trial. Lancet 2010;376:687–97.
25. Tabernero J, Hoff PM, Shen L, et al. Pertuzumab + trastuzumab + chemotherapy for HER2-positive metastatic gastric or gastro-oesophageal junction cancer: Final analysis of a Phase III study (JACOB) [abstract]. Ann Oncol 2017;28(suppl 5):6160.
26. Fuchs CS, Tomasek J, Yong CJ, et al. Ramucirumab monotherapy for previously treated advanced gastric or gastro-oesophageal junction adenocarcinoma (REGARD): an international, randomised, multicentre, placebo-controlled, phase 3 trial. Lancet 2014;383:31–9.
27. Wilke H, Muro K, Van Cutsem E, et al. Ramucirumab plus paclitaxel versus placebo plus paclitaxel in patients with previously treated advanced gastric or gastro-oesophageal junction adenocarcinoma (RAINBOW): a double-blind, randomised phase 3 trial. Lancet Oncol 2014;15:1224–35.
28. Ohtsu A, Shah MA, Van Cutsem E, et al. Bevacizumab in combination with chemotherapy as first-line therapy in advanced gastric cancer: a randomized, double-blind, placebo-controlled phase III study. J Clin Oncol 2011;29:3968–76.
29. Van Cutsem E, de Haas S, Kang YK, et al, Bevacizumab in combination with chemotherapy as first-line therapy in advanced gastric cancer: a biomarker evaluation from the AVAGAST randomized phase III trial. J Clin Oncol 2012;30:2119–27.
30. Makiyama A, Sagara K, Kawada J, et al. A randomized phase II study of weekly paclitaxel ± trastuzumab in patients with HER2-positive advanced gastric or gastro-esophageal junction cancer refractory to trastuzumab combined with fluoropyrimidine and platinum: WJOG7112G (T-ACT) [abstract]. J Clin Oncol 2018;36(no. 15 suppl):4011.
31. Satoh T, Xu RH, Chung HC, et al. Lapatinib plus paclitaxel versus paclitaxel alone in the second-line treatment of HER2-amplified advanced gastric cancer in Asian populations: TyTAN--a randomized, phase III study. J Clin Oncol 2014;32:2039–49.
32. Thuss-Patience PC, Shah MA, Ohtsu A, et al. Trastuzumab emtansine versus taxane use for previously treated HER2-positive locally advanced or metastatic gastric or gastro-oesophageal junction adenocarcinoma (GATSBY): an international randomised, open-label, adaptive, phase 2/3 study. Lancet Oncol 2017;18:640–53.
33. Ohtsu A, Ajani JA, Bai YX, et al. Everolimus for previously treated advanced gastric cancer: results of the randomized, double-blind, phase III GRANITE-1 study. J Clin Oncol 2013;31:3935–43.
34. Lordick F, Kang YK, Chung HC, et al. Capecitabine and cisplatin with or without cetuximab for patients with previously untreated advanced gastric cancer (EXPAND): a randomised, open-label phase 3 trial. Lancet Oncol 2013;14:490–9.
35. Waddell T, Chau I, Cunningham D, et al. Epirubicin, oxaliplatin, and capecitabine with or without panitumumab for patients with previously untreated advanced oesophagogastric cancer (REAL3): a randomised, open-label phase 3 trial. Lancet Oncol 2013;14:481–9.
36. Catenacci DVT, Tebbutt NC, Davidenko I, et al. Rilotumumab plus epirubicin, cisplatin, and capecitabine as first-line therapy in advanced MET-positive gastric or gastro-oesophageal junction cancer (RILOMET-1): a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Oncol 2017;18:1467–82.
37. Shah MA, Bang YJ, Lordick F, et al. Effect of fluorouracil, leucovorin, and oxaliplatin with or without onartuzumab in HER2-negative, MET-positive gastroesophageal adenocarcinoma: the METGastric randomized clinical trial. JAMA Oncol 2017;3:620–7.
38. Fuchs CS, Doi T, Jang RW, et al. Safety and efficacy of pembrolizumab monotherapy in patients with previously treated advanced gastric and gastroesophageal junction cancer: phase 2 clinical KEYNOTE-059 trial. JAMA Oncol 2018;4(5):e180013.