User login
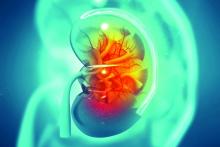
FMT cuts risk of bloodstream infections in patients with recurrent CDI
Background: After a first episode of CDI, almost 20% of patients will have a recurrence. Recurrent CDI (rCDI) is more likely to be associated with life-threatening complications including toxic megacolon, perforation, bloodstream infections, and death. Most BSIs are caused by intestinal microbes. Some evidence suggests that vancomycin therapy creates conditions that favor intestinal colonization by health care–associated pathogens. FMT aims to restore the normal composition of gut microbiota, is superior to vancomycin, and might decrease the incidence of BSI and related complications including death.
Study design: Prospective cohort study.
Setting: Fondazione Policlinico Universitario Agostino Gemelli in Rome.
Synopsis: In this study, 290 patients with rCDIs were randomized to FMT (109 patients) or antibiotic therapy (181 patients). Only patients with their first rCDIs were included. The primary outcome was the development of primary BSI after treatment of rCDI and within a 90-day follow-up period. Secondary outcomes were length of hospitalization and overall survival at 90 days.
Five patients in the FMT group and 40 in the antibiotic group (16% of total patients) developed BSIs during the 90-day follow-up. Because of baseline characteristic differences in the patients treated with FMT versus antibiotics, comparative analyses were limited to the matched cohort. Risk for BSIs was 23% lower in the FMT group (95% confidence interval, 10%-35%); the FMT group also had 14 fewer days of hospitalization (95% CI, 9-20 fewer days) and a 32% increase in overall survival (95% CI, 16%-47%), compared with the antibiotic group. Limitations of the study include its observational nature, single-center design, and differences in several baseline characteristics between the groups remaining after the match.
Bottom line: Patients with rCDI who received FMT were less likely to develop primary BSIs and related complications, including hospital length of stay and death when compared with patients who received antibiotics.
Citation: Ianiro G et al. Incidence of bloodstream infections, length of hospital stay, and survival in patients with recurrent Clostridioides difficile infection treated with fecal microbiota transplantation or antibiotics: A prospective cohort study. Ann Intern Med. 2019, Nov 5;171:695-702.
Dr. Santa is assistant professor in the division of hospital medicine, Loyola University Medical Center, Maywood, Ill.
Background: After a first episode of CDI, almost 20% of patients will have a recurrence. Recurrent CDI (rCDI) is more likely to be associated with life-threatening complications including toxic megacolon, perforation, bloodstream infections, and death. Most BSIs are caused by intestinal microbes. Some evidence suggests that vancomycin therapy creates conditions that favor intestinal colonization by health care–associated pathogens. FMT aims to restore the normal composition of gut microbiota, is superior to vancomycin, and might decrease the incidence of BSI and related complications including death.
Study design: Prospective cohort study.
Setting: Fondazione Policlinico Universitario Agostino Gemelli in Rome.
Synopsis: In this study, 290 patients with rCDIs were randomized to FMT (109 patients) or antibiotic therapy (181 patients). Only patients with their first rCDIs were included. The primary outcome was the development of primary BSI after treatment of rCDI and within a 90-day follow-up period. Secondary outcomes were length of hospitalization and overall survival at 90 days.
Five patients in the FMT group and 40 in the antibiotic group (16% of total patients) developed BSIs during the 90-day follow-up. Because of baseline characteristic differences in the patients treated with FMT versus antibiotics, comparative analyses were limited to the matched cohort. Risk for BSIs was 23% lower in the FMT group (95% confidence interval, 10%-35%); the FMT group also had 14 fewer days of hospitalization (95% CI, 9-20 fewer days) and a 32% increase in overall survival (95% CI, 16%-47%), compared with the antibiotic group. Limitations of the study include its observational nature, single-center design, and differences in several baseline characteristics between the groups remaining after the match.
Bottom line: Patients with rCDI who received FMT were less likely to develop primary BSIs and related complications, including hospital length of stay and death when compared with patients who received antibiotics.
Citation: Ianiro G et al. Incidence of bloodstream infections, length of hospital stay, and survival in patients with recurrent Clostridioides difficile infection treated with fecal microbiota transplantation or antibiotics: A prospective cohort study. Ann Intern Med. 2019, Nov 5;171:695-702.
Dr. Santa is assistant professor in the division of hospital medicine, Loyola University Medical Center, Maywood, Ill.
Background: After a first episode of CDI, almost 20% of patients will have a recurrence. Recurrent CDI (rCDI) is more likely to be associated with life-threatening complications including toxic megacolon, perforation, bloodstream infections, and death. Most BSIs are caused by intestinal microbes. Some evidence suggests that vancomycin therapy creates conditions that favor intestinal colonization by health care–associated pathogens. FMT aims to restore the normal composition of gut microbiota, is superior to vancomycin, and might decrease the incidence of BSI and related complications including death.
Study design: Prospective cohort study.
Setting: Fondazione Policlinico Universitario Agostino Gemelli in Rome.
Synopsis: In this study, 290 patients with rCDIs were randomized to FMT (109 patients) or antibiotic therapy (181 patients). Only patients with their first rCDIs were included. The primary outcome was the development of primary BSI after treatment of rCDI and within a 90-day follow-up period. Secondary outcomes were length of hospitalization and overall survival at 90 days.
Five patients in the FMT group and 40 in the antibiotic group (16% of total patients) developed BSIs during the 90-day follow-up. Because of baseline characteristic differences in the patients treated with FMT versus antibiotics, comparative analyses were limited to the matched cohort. Risk for BSIs was 23% lower in the FMT group (95% confidence interval, 10%-35%); the FMT group also had 14 fewer days of hospitalization (95% CI, 9-20 fewer days) and a 32% increase in overall survival (95% CI, 16%-47%), compared with the antibiotic group. Limitations of the study include its observational nature, single-center design, and differences in several baseline characteristics between the groups remaining after the match.
Bottom line: Patients with rCDI who received FMT were less likely to develop primary BSIs and related complications, including hospital length of stay and death when compared with patients who received antibiotics.
Citation: Ianiro G et al. Incidence of bloodstream infections, length of hospital stay, and survival in patients with recurrent Clostridioides difficile infection treated with fecal microbiota transplantation or antibiotics: A prospective cohort study. Ann Intern Med. 2019, Nov 5;171:695-702.
Dr. Santa is assistant professor in the division of hospital medicine, Loyola University Medical Center, Maywood, Ill.
CDC guidelines coming on long COVID
The Centers for Disease Control and Prevention is finalizing new guidelines to help clinicians diagnose and manage long COVID, or postacute sequelae of SARS-CoV-2 infection.
In a day-long congressional hearing on April 28, John Brooks, MD, a medical epidemiologist at the CDC’s division of HIV/AIDS prevention, testified that the guidelines were going through the clearance process at the agency, but would be forthcoming.
“They should be coming out very shortly,” Dr. Brooks said.
The guidelines, which were developed in collaboration with newly established long-COVID clinics and patient advocacy groups, will “illustrate how to diagnose and begin to pull together what we know about management,” of the complex condition, he said.
For many doctors and patients who are struggling to understand symptoms that persist for months after the initial viral infection, the guidelines can’t come soon enough.
National Institutes of Health Director Francis Collins, MD, PhD, who also testified at the hearing, estimated that as many as 3 million people could be left with chronic health problems after even mild COVID infections.
“I can’t overstate how serious this issue is for the health of our nation,” he said.
Dr. Collins said his estimate was based on studies showing that roughly 10% of people who get COVID could be affected by this and whose “long-term course is uncertain,” he said. So far, more than 32 million Americans are known to have been infected with the new coronavirus.
“We need to make sure we put our arms around them and bring answers and care to them,” said Rep. Anna Eshoo (D-Calif.), chairwoman of the Subcommittee on Health.
Jennifer Possick, MD, who directs the post-COVID recovery program at Yale New Haven (Conn.) Hospital, testified that the tidal wave of patients she and her colleagues were seeing was overwhelming.
“We are a well-resourced program at an academic medical center, but we are swamped by the need in our community. This year, we have seen more patients with post COVID-19 conditions in our clinic alone than we have new cases of asthma and COPD combined,” she said. “The magnitude of the challenge is daunting.”
Dr. Possick estimated that there are “over 60” clinics in the United States that have started to treat long-COVID patients, but said they are grassroots efforts and all very different from each other.
“Whoever had the resources, had the time, [and] was able to take the initiative and forge to the relationships because most of them are multidisciplinary, did so,” she said.
Patients testify
Several representatives shared moving personal stories of loved ones or staffers who remained ill months after a COVID diagnosis.
Rep. Ann Kuster, from New Hampshire, talked about her 34-year-old niece, a member of the U.S. Ski Team, who had COVID just over a year ago and “continues to struggle with everything, even the simplest activities of daily living” she said. “She has to choose between taking a shower or making dinner. I’m so proud of her for hanging in there.”
Long-COVID patients invited to testify by the subcommittee described months of disability that left them with soaring medical bills and no ability to work to pay them.
“I am now a poor, Black, disabled woman, living with long COVID,” said Chimere Smith, who said she had been a school teacher in Baltimore. “Saying it aloud makes it no more easy to accept.”
She said COVID had affected her ability to think clearly and caused debilitating fatigue, which prevented her from working. She said she lost her vision for almost 5 months because doctors misdiagnosed a cataract caused by long COVID as dry eye.
“If I did not have a loving family, I [would] be speaking to you today [from] my car, the only property I now own.”
Ms. Smith said that long-COVID clinics, which are mostly housed within academic medical centers, were not going to be accessible for all long-haulers, who are disproportionately women of color. She has started a clinic, based out of her church, to help other patients from her community.
“No one wants to hear that long COVID has decimated my life or the lives of other black women in less than a year,” Ms. Smith said. “We’ve just been waiting and hoping for compassionate doctors and politicians who would acknowledge us.”
A version of this article first appeared on Medscape.com.
The Centers for Disease Control and Prevention is finalizing new guidelines to help clinicians diagnose and manage long COVID, or postacute sequelae of SARS-CoV-2 infection.
In a day-long congressional hearing on April 28, John Brooks, MD, a medical epidemiologist at the CDC’s division of HIV/AIDS prevention, testified that the guidelines were going through the clearance process at the agency, but would be forthcoming.
“They should be coming out very shortly,” Dr. Brooks said.
The guidelines, which were developed in collaboration with newly established long-COVID clinics and patient advocacy groups, will “illustrate how to diagnose and begin to pull together what we know about management,” of the complex condition, he said.
For many doctors and patients who are struggling to understand symptoms that persist for months after the initial viral infection, the guidelines can’t come soon enough.
National Institutes of Health Director Francis Collins, MD, PhD, who also testified at the hearing, estimated that as many as 3 million people could be left with chronic health problems after even mild COVID infections.
“I can’t overstate how serious this issue is for the health of our nation,” he said.
Dr. Collins said his estimate was based on studies showing that roughly 10% of people who get COVID could be affected by this and whose “long-term course is uncertain,” he said. So far, more than 32 million Americans are known to have been infected with the new coronavirus.
“We need to make sure we put our arms around them and bring answers and care to them,” said Rep. Anna Eshoo (D-Calif.), chairwoman of the Subcommittee on Health.
Jennifer Possick, MD, who directs the post-COVID recovery program at Yale New Haven (Conn.) Hospital, testified that the tidal wave of patients she and her colleagues were seeing was overwhelming.
“We are a well-resourced program at an academic medical center, but we are swamped by the need in our community. This year, we have seen more patients with post COVID-19 conditions in our clinic alone than we have new cases of asthma and COPD combined,” she said. “The magnitude of the challenge is daunting.”
Dr. Possick estimated that there are “over 60” clinics in the United States that have started to treat long-COVID patients, but said they are grassroots efforts and all very different from each other.
“Whoever had the resources, had the time, [and] was able to take the initiative and forge to the relationships because most of them are multidisciplinary, did so,” she said.
Patients testify
Several representatives shared moving personal stories of loved ones or staffers who remained ill months after a COVID diagnosis.
Rep. Ann Kuster, from New Hampshire, talked about her 34-year-old niece, a member of the U.S. Ski Team, who had COVID just over a year ago and “continues to struggle with everything, even the simplest activities of daily living” she said. “She has to choose between taking a shower or making dinner. I’m so proud of her for hanging in there.”
Long-COVID patients invited to testify by the subcommittee described months of disability that left them with soaring medical bills and no ability to work to pay them.
“I am now a poor, Black, disabled woman, living with long COVID,” said Chimere Smith, who said she had been a school teacher in Baltimore. “Saying it aloud makes it no more easy to accept.”
She said COVID had affected her ability to think clearly and caused debilitating fatigue, which prevented her from working. She said she lost her vision for almost 5 months because doctors misdiagnosed a cataract caused by long COVID as dry eye.
“If I did not have a loving family, I [would] be speaking to you today [from] my car, the only property I now own.”
Ms. Smith said that long-COVID clinics, which are mostly housed within academic medical centers, were not going to be accessible for all long-haulers, who are disproportionately women of color. She has started a clinic, based out of her church, to help other patients from her community.
“No one wants to hear that long COVID has decimated my life or the lives of other black women in less than a year,” Ms. Smith said. “We’ve just been waiting and hoping for compassionate doctors and politicians who would acknowledge us.”
A version of this article first appeared on Medscape.com.
The Centers for Disease Control and Prevention is finalizing new guidelines to help clinicians diagnose and manage long COVID, or postacute sequelae of SARS-CoV-2 infection.
In a day-long congressional hearing on April 28, John Brooks, MD, a medical epidemiologist at the CDC’s division of HIV/AIDS prevention, testified that the guidelines were going through the clearance process at the agency, but would be forthcoming.
“They should be coming out very shortly,” Dr. Brooks said.
The guidelines, which were developed in collaboration with newly established long-COVID clinics and patient advocacy groups, will “illustrate how to diagnose and begin to pull together what we know about management,” of the complex condition, he said.
For many doctors and patients who are struggling to understand symptoms that persist for months after the initial viral infection, the guidelines can’t come soon enough.
National Institutes of Health Director Francis Collins, MD, PhD, who also testified at the hearing, estimated that as many as 3 million people could be left with chronic health problems after even mild COVID infections.
“I can’t overstate how serious this issue is for the health of our nation,” he said.
Dr. Collins said his estimate was based on studies showing that roughly 10% of people who get COVID could be affected by this and whose “long-term course is uncertain,” he said. So far, more than 32 million Americans are known to have been infected with the new coronavirus.
“We need to make sure we put our arms around them and bring answers and care to them,” said Rep. Anna Eshoo (D-Calif.), chairwoman of the Subcommittee on Health.
Jennifer Possick, MD, who directs the post-COVID recovery program at Yale New Haven (Conn.) Hospital, testified that the tidal wave of patients she and her colleagues were seeing was overwhelming.
“We are a well-resourced program at an academic medical center, but we are swamped by the need in our community. This year, we have seen more patients with post COVID-19 conditions in our clinic alone than we have new cases of asthma and COPD combined,” she said. “The magnitude of the challenge is daunting.”
Dr. Possick estimated that there are “over 60” clinics in the United States that have started to treat long-COVID patients, but said they are grassroots efforts and all very different from each other.
“Whoever had the resources, had the time, [and] was able to take the initiative and forge to the relationships because most of them are multidisciplinary, did so,” she said.
Patients testify
Several representatives shared moving personal stories of loved ones or staffers who remained ill months after a COVID diagnosis.
Rep. Ann Kuster, from New Hampshire, talked about her 34-year-old niece, a member of the U.S. Ski Team, who had COVID just over a year ago and “continues to struggle with everything, even the simplest activities of daily living” she said. “She has to choose between taking a shower or making dinner. I’m so proud of her for hanging in there.”
Long-COVID patients invited to testify by the subcommittee described months of disability that left them with soaring medical bills and no ability to work to pay them.
“I am now a poor, Black, disabled woman, living with long COVID,” said Chimere Smith, who said she had been a school teacher in Baltimore. “Saying it aloud makes it no more easy to accept.”
She said COVID had affected her ability to think clearly and caused debilitating fatigue, which prevented her from working. She said she lost her vision for almost 5 months because doctors misdiagnosed a cataract caused by long COVID as dry eye.
“If I did not have a loving family, I [would] be speaking to you today [from] my car, the only property I now own.”
Ms. Smith said that long-COVID clinics, which are mostly housed within academic medical centers, were not going to be accessible for all long-haulers, who are disproportionately women of color. She has started a clinic, based out of her church, to help other patients from her community.
“No one wants to hear that long COVID has decimated my life or the lives of other black women in less than a year,” Ms. Smith said. “We’ve just been waiting and hoping for compassionate doctors and politicians who would acknowledge us.”
A version of this article first appeared on Medscape.com.
Being overweight ups risk of severe COVID-19 in hospital
In a global meta-analysis of more than 7,000 patients who were hospitalized with COVID-19, individuals with overweight or obesity were more likely to need respiratory support but were not more likely to die in the hospital, compared to individuals of normal weight.
Compared to patients without diabetes, those with diabetes had higher odds of needing invasive respiratory support (with intubation) but not for needing noninvasive respiratory support or of dying in the hospital.
“Surprisingly,” among patients with diabetes, being overweight or having obesity did not further increase the odds of any of these outcomes, the researchers wrote. The finding needs to be confirmed in larger studies, they said, because the sample sizes in these subanalyses were small and the confidence intervals were large.
The study by Danielle K. Longmore, PhD, of Murdoch Children’s Research Institute (MCRI), Melbourne, and colleagues from the International BMI-COVID consortium, was published online April 15 in Diabetes Care.
This new research “adds to the known data on the associations between obesity and severe COVID-19 disease and extends these findings” to patients who are overweight and/or have diabetes, Dr. Longmore, a pediatric endocrinologist with a clinical and research interest in childhood and youth obesity, said in an interview.
Immunologist Siroon Bekkering, PhD, of Radboud University Medical Center, Nijmegen, the Netherlands, explained that never before have so much data of different types regarding obesity been combined in one large study. Dr. Bekkering is a coauthor of the article and was a principal investigator.
“Several national and international observations already showed the important role of overweight and obesity in a more severe COVID-19 course. This study adds to those observations by combining data from several countries with the possibility to look at the risk factors separately,” she said in a statement from her institution.
“Regardless of other risk factors (such as heart disease or diabetes), we now see that too high a BMI [body mass index] can actually lead to a more severe course in [coronavirus] infection,” she said.
Study implications: Data show that overweight, obesity add to risk
These latest findings highlight the urgent need to develop public health policies to address socioeconomic and psychological drivers of obesity, Dr. Longmore said.
“Although taking steps to address obesity in the short term is unlikely to have an immediate impact in the COVID-19 pandemic, it will likely reduce the disease burden in future viral pandemics and reduce risks of complications like heart disease and stroke,” she observed in a statement issued by MCRI.
Coauthor Kirsty R. Short, PhD, a research fellow at the University of Queensland, Brisbane, Australia, noted that “obesity is associated with numerous poor health outcomes, including increased risk of cardiometabolic and respiratory disease and more severe viral disease including influenza, dengue, and SARS-CoV-1.
“Given the large scale of this study,” she said, “we have conclusively shown that being overweight or obese are independent risk factors for worse outcomes in adults hospitalized with COVID-19.”
“At the moment, the World Health Organization has not had enough high-quality data to include being overweight or obese as a risk factor for severe COVID-19 disease,” added another author, David P. Burgner, PhD, a pediatric infectious diseases clinician scientist from MCRI.
“Our study should help inform decisions about which higher-risk groups should be vaccinated as a priority,” he observed.
Does being overweight up risk of worse COVID-19 outcomes?
About 13% of the world’s population are overweight, and 40% have obesity. There are wide between-country variations in these data, and about 90% of patients with type 2 diabetes are overweight or obese, the researchers noted.
The Organisation for Economic Co-operation and Development reported that the prevalence of obesity in 2016-2017 was 5.7% to 8.9% in Asia, 9.8% to 16.8% in Europe, 26.5% in South Africa, and 40.0% in the United States, they added.
Obesity is common and has emerged as an important risk factor for severe COVID-19. However, most previous studies of COVID-19 and elevated BMI were conducted in single centers and did not focus on patients with overweight.
To investigate, the researchers identified 7,244 patients (two-thirds were overweight or obese) who were hospitalized with COVID-19 in 69 hospitals (18 sites) in 11 countries from Jan. 17, 2020, to June 2, 2020.
Most patients were hospitalized with COVID-19 in the Netherlands (2,260), followed by New York City (1,682), Switzerland (920), St. Louis (805), Norway, Italy, China, South Africa, Indonesia, Denmark, Los Angeles, Austria, and Singapore.
Just over half (60%) of the individuals were male, and 52% were older than 65.
Overall, 34.8% were overweight, and 30.8% had obesity, but the average weight varied considerably between countries and sites.
Increased need for respiratory support, same mortality risk
Compared with patients with normal weight, patients who were overweight had a 44% increased risk of needing supplemental oxygen/noninvasive ventilation, and those with obesity had a 75% increased risk of this, after adjustment for age (< 65, ≥ 65), sex, hypertension, diabetes, or preexisting cardiovascular disease or respiratory conditions.
Patients who were overweight had a 22% increased risk of needing invasive (mechanical) ventilation, and those with obesity had a 73% increased risk of this, after multivariable adjustment.
Being overweight or having obesity was not associated with a significantly increased risk of dying in the hospital, however.
“In other viral respiratory infections, such as influenza, there is a similar pattern of increased requirement for ventilatory support but lower in-hospital mortality among individuals with obesity, when compared to those with normal range BMI,” Dr. Longmore noted. She said that larger studies are needed to further explore this finding regarding COVID-19.
Compared to patients without diabetes, those with diabetes had a 21% increased risk of requiring invasive ventilation, but they did not have an increased risk of needing noninvasive ventilation or of dying in the hospital.
As in previous studies, individuals who had cardiovascular and preexisting respiratory diseases were not at greater risk of needing oxygen or mechanical ventilation but were at increased risk for in-hospital death. Men had a greater risk of needing invasive mechanical ventilation, and individuals who were older than 65 had an increased risk of requiring oxygen or of dying in the hospital.
A living meta-analysis, call for more collaborators
“We consider this a ‘living meta-analysis’ and invite other centers to join us,” Dr. Longmore said. “We hope to update the analyses as more data are contributed.”
No specific project funded the study. The authors have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
In a global meta-analysis of more than 7,000 patients who were hospitalized with COVID-19, individuals with overweight or obesity were more likely to need respiratory support but were not more likely to die in the hospital, compared to individuals of normal weight.
Compared to patients without diabetes, those with diabetes had higher odds of needing invasive respiratory support (with intubation) but not for needing noninvasive respiratory support or of dying in the hospital.
“Surprisingly,” among patients with diabetes, being overweight or having obesity did not further increase the odds of any of these outcomes, the researchers wrote. The finding needs to be confirmed in larger studies, they said, because the sample sizes in these subanalyses were small and the confidence intervals were large.
The study by Danielle K. Longmore, PhD, of Murdoch Children’s Research Institute (MCRI), Melbourne, and colleagues from the International BMI-COVID consortium, was published online April 15 in Diabetes Care.
This new research “adds to the known data on the associations between obesity and severe COVID-19 disease and extends these findings” to patients who are overweight and/or have diabetes, Dr. Longmore, a pediatric endocrinologist with a clinical and research interest in childhood and youth obesity, said in an interview.
Immunologist Siroon Bekkering, PhD, of Radboud University Medical Center, Nijmegen, the Netherlands, explained that never before have so much data of different types regarding obesity been combined in one large study. Dr. Bekkering is a coauthor of the article and was a principal investigator.
“Several national and international observations already showed the important role of overweight and obesity in a more severe COVID-19 course. This study adds to those observations by combining data from several countries with the possibility to look at the risk factors separately,” she said in a statement from her institution.
“Regardless of other risk factors (such as heart disease or diabetes), we now see that too high a BMI [body mass index] can actually lead to a more severe course in [coronavirus] infection,” she said.
Study implications: Data show that overweight, obesity add to risk
These latest findings highlight the urgent need to develop public health policies to address socioeconomic and psychological drivers of obesity, Dr. Longmore said.
“Although taking steps to address obesity in the short term is unlikely to have an immediate impact in the COVID-19 pandemic, it will likely reduce the disease burden in future viral pandemics and reduce risks of complications like heart disease and stroke,” she observed in a statement issued by MCRI.
Coauthor Kirsty R. Short, PhD, a research fellow at the University of Queensland, Brisbane, Australia, noted that “obesity is associated with numerous poor health outcomes, including increased risk of cardiometabolic and respiratory disease and more severe viral disease including influenza, dengue, and SARS-CoV-1.
“Given the large scale of this study,” she said, “we have conclusively shown that being overweight or obese are independent risk factors for worse outcomes in adults hospitalized with COVID-19.”
“At the moment, the World Health Organization has not had enough high-quality data to include being overweight or obese as a risk factor for severe COVID-19 disease,” added another author, David P. Burgner, PhD, a pediatric infectious diseases clinician scientist from MCRI.
“Our study should help inform decisions about which higher-risk groups should be vaccinated as a priority,” he observed.
Does being overweight up risk of worse COVID-19 outcomes?
About 13% of the world’s population are overweight, and 40% have obesity. There are wide between-country variations in these data, and about 90% of patients with type 2 diabetes are overweight or obese, the researchers noted.
The Organisation for Economic Co-operation and Development reported that the prevalence of obesity in 2016-2017 was 5.7% to 8.9% in Asia, 9.8% to 16.8% in Europe, 26.5% in South Africa, and 40.0% in the United States, they added.
Obesity is common and has emerged as an important risk factor for severe COVID-19. However, most previous studies of COVID-19 and elevated BMI were conducted in single centers and did not focus on patients with overweight.
To investigate, the researchers identified 7,244 patients (two-thirds were overweight or obese) who were hospitalized with COVID-19 in 69 hospitals (18 sites) in 11 countries from Jan. 17, 2020, to June 2, 2020.
Most patients were hospitalized with COVID-19 in the Netherlands (2,260), followed by New York City (1,682), Switzerland (920), St. Louis (805), Norway, Italy, China, South Africa, Indonesia, Denmark, Los Angeles, Austria, and Singapore.
Just over half (60%) of the individuals were male, and 52% were older than 65.
Overall, 34.8% were overweight, and 30.8% had obesity, but the average weight varied considerably between countries and sites.
Increased need for respiratory support, same mortality risk
Compared with patients with normal weight, patients who were overweight had a 44% increased risk of needing supplemental oxygen/noninvasive ventilation, and those with obesity had a 75% increased risk of this, after adjustment for age (< 65, ≥ 65), sex, hypertension, diabetes, or preexisting cardiovascular disease or respiratory conditions.
Patients who were overweight had a 22% increased risk of needing invasive (mechanical) ventilation, and those with obesity had a 73% increased risk of this, after multivariable adjustment.
Being overweight or having obesity was not associated with a significantly increased risk of dying in the hospital, however.
“In other viral respiratory infections, such as influenza, there is a similar pattern of increased requirement for ventilatory support but lower in-hospital mortality among individuals with obesity, when compared to those with normal range BMI,” Dr. Longmore noted. She said that larger studies are needed to further explore this finding regarding COVID-19.
Compared to patients without diabetes, those with diabetes had a 21% increased risk of requiring invasive ventilation, but they did not have an increased risk of needing noninvasive ventilation or of dying in the hospital.
As in previous studies, individuals who had cardiovascular and preexisting respiratory diseases were not at greater risk of needing oxygen or mechanical ventilation but were at increased risk for in-hospital death. Men had a greater risk of needing invasive mechanical ventilation, and individuals who were older than 65 had an increased risk of requiring oxygen or of dying in the hospital.
A living meta-analysis, call for more collaborators
“We consider this a ‘living meta-analysis’ and invite other centers to join us,” Dr. Longmore said. “We hope to update the analyses as more data are contributed.”
No specific project funded the study. The authors have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
In a global meta-analysis of more than 7,000 patients who were hospitalized with COVID-19, individuals with overweight or obesity were more likely to need respiratory support but were not more likely to die in the hospital, compared to individuals of normal weight.
Compared to patients without diabetes, those with diabetes had higher odds of needing invasive respiratory support (with intubation) but not for needing noninvasive respiratory support or of dying in the hospital.
“Surprisingly,” among patients with diabetes, being overweight or having obesity did not further increase the odds of any of these outcomes, the researchers wrote. The finding needs to be confirmed in larger studies, they said, because the sample sizes in these subanalyses were small and the confidence intervals were large.
The study by Danielle K. Longmore, PhD, of Murdoch Children’s Research Institute (MCRI), Melbourne, and colleagues from the International BMI-COVID consortium, was published online April 15 in Diabetes Care.
This new research “adds to the known data on the associations between obesity and severe COVID-19 disease and extends these findings” to patients who are overweight and/or have diabetes, Dr. Longmore, a pediatric endocrinologist with a clinical and research interest in childhood and youth obesity, said in an interview.
Immunologist Siroon Bekkering, PhD, of Radboud University Medical Center, Nijmegen, the Netherlands, explained that never before have so much data of different types regarding obesity been combined in one large study. Dr. Bekkering is a coauthor of the article and was a principal investigator.
“Several national and international observations already showed the important role of overweight and obesity in a more severe COVID-19 course. This study adds to those observations by combining data from several countries with the possibility to look at the risk factors separately,” she said in a statement from her institution.
“Regardless of other risk factors (such as heart disease or diabetes), we now see that too high a BMI [body mass index] can actually lead to a more severe course in [coronavirus] infection,” she said.
Study implications: Data show that overweight, obesity add to risk
These latest findings highlight the urgent need to develop public health policies to address socioeconomic and psychological drivers of obesity, Dr. Longmore said.
“Although taking steps to address obesity in the short term is unlikely to have an immediate impact in the COVID-19 pandemic, it will likely reduce the disease burden in future viral pandemics and reduce risks of complications like heart disease and stroke,” she observed in a statement issued by MCRI.
Coauthor Kirsty R. Short, PhD, a research fellow at the University of Queensland, Brisbane, Australia, noted that “obesity is associated with numerous poor health outcomes, including increased risk of cardiometabolic and respiratory disease and more severe viral disease including influenza, dengue, and SARS-CoV-1.
“Given the large scale of this study,” she said, “we have conclusively shown that being overweight or obese are independent risk factors for worse outcomes in adults hospitalized with COVID-19.”
“At the moment, the World Health Organization has not had enough high-quality data to include being overweight or obese as a risk factor for severe COVID-19 disease,” added another author, David P. Burgner, PhD, a pediatric infectious diseases clinician scientist from MCRI.
“Our study should help inform decisions about which higher-risk groups should be vaccinated as a priority,” he observed.
Does being overweight up risk of worse COVID-19 outcomes?
About 13% of the world’s population are overweight, and 40% have obesity. There are wide between-country variations in these data, and about 90% of patients with type 2 diabetes are overweight or obese, the researchers noted.
The Organisation for Economic Co-operation and Development reported that the prevalence of obesity in 2016-2017 was 5.7% to 8.9% in Asia, 9.8% to 16.8% in Europe, 26.5% in South Africa, and 40.0% in the United States, they added.
Obesity is common and has emerged as an important risk factor for severe COVID-19. However, most previous studies of COVID-19 and elevated BMI were conducted in single centers and did not focus on patients with overweight.
To investigate, the researchers identified 7,244 patients (two-thirds were overweight or obese) who were hospitalized with COVID-19 in 69 hospitals (18 sites) in 11 countries from Jan. 17, 2020, to June 2, 2020.
Most patients were hospitalized with COVID-19 in the Netherlands (2,260), followed by New York City (1,682), Switzerland (920), St. Louis (805), Norway, Italy, China, South Africa, Indonesia, Denmark, Los Angeles, Austria, and Singapore.
Just over half (60%) of the individuals were male, and 52% were older than 65.
Overall, 34.8% were overweight, and 30.8% had obesity, but the average weight varied considerably between countries and sites.
Increased need for respiratory support, same mortality risk
Compared with patients with normal weight, patients who were overweight had a 44% increased risk of needing supplemental oxygen/noninvasive ventilation, and those with obesity had a 75% increased risk of this, after adjustment for age (< 65, ≥ 65), sex, hypertension, diabetes, or preexisting cardiovascular disease or respiratory conditions.
Patients who were overweight had a 22% increased risk of needing invasive (mechanical) ventilation, and those with obesity had a 73% increased risk of this, after multivariable adjustment.
Being overweight or having obesity was not associated with a significantly increased risk of dying in the hospital, however.
“In other viral respiratory infections, such as influenza, there is a similar pattern of increased requirement for ventilatory support but lower in-hospital mortality among individuals with obesity, when compared to those with normal range BMI,” Dr. Longmore noted. She said that larger studies are needed to further explore this finding regarding COVID-19.
Compared to patients without diabetes, those with diabetes had a 21% increased risk of requiring invasive ventilation, but they did not have an increased risk of needing noninvasive ventilation or of dying in the hospital.
As in previous studies, individuals who had cardiovascular and preexisting respiratory diseases were not at greater risk of needing oxygen or mechanical ventilation but were at increased risk for in-hospital death. Men had a greater risk of needing invasive mechanical ventilation, and individuals who were older than 65 had an increased risk of requiring oxygen or of dying in the hospital.
A living meta-analysis, call for more collaborators
“We consider this a ‘living meta-analysis’ and invite other centers to join us,” Dr. Longmore said. “We hope to update the analyses as more data are contributed.”
No specific project funded the study. The authors have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
IV drug users: The new face of candidemia
Background: Intravenous drug use is an increasingly common risk factor for candidemia as the opioid crisis worsens. This study quantifies this change and characterizes the changing epidemiology of candidemia.
Study design: A cross-sectional study.
Setting: Health departments in nine states.
Synopsis: IV drug users typically have a very distinctive phenotype among all patients with candidemia: They are younger (35 vs. 63 years), are more likely to be homeless, are not black, are smokers; they have hepatitis C, have no malignancies, have polymicrobial bacteremia, and have acquired the infection outside of the hospital. They are much less likely to die of the infection (8.6% vs 27.5%), compared with the non-IV drug users. In four states, the proportion of candidemia associated with IV drug use more than doubled, from 7% to 15% during 2014-2017, representing a possible shift in the epidemiology of candidemia.
The study did not quantify or address complications that many hospitalists see, such as endocarditis, endophthalmitis, and osteomyelitis. The study looked at only nine states, so results may not be generalizable. Nevertheless, the robust analysis suggests an alarming, increasing trend.
Bottom line: As the opioid crisis worsens, hospitalists should consider candidemia in hospitalized IV drug users and should evaluate patients with candidemia for IV drug use.
Citation: Zhang AY et al. The changing epidemiology of candidemia in the United States: Injection drug use as an increasingly common risk factor – Active surveillance in selected sites, United States, 2014-2017. Clin Infect Dis. 2019 Nov 2. doi: 10.1093/cid/ciz1061.
Dr. Raghavan is assistant professor in the division of hospital medicine, Loyola University Medical Center, Maywood, Ill.
Background: Intravenous drug use is an increasingly common risk factor for candidemia as the opioid crisis worsens. This study quantifies this change and characterizes the changing epidemiology of candidemia.
Study design: A cross-sectional study.
Setting: Health departments in nine states.
Synopsis: IV drug users typically have a very distinctive phenotype among all patients with candidemia: They are younger (35 vs. 63 years), are more likely to be homeless, are not black, are smokers; they have hepatitis C, have no malignancies, have polymicrobial bacteremia, and have acquired the infection outside of the hospital. They are much less likely to die of the infection (8.6% vs 27.5%), compared with the non-IV drug users. In four states, the proportion of candidemia associated with IV drug use more than doubled, from 7% to 15% during 2014-2017, representing a possible shift in the epidemiology of candidemia.
The study did not quantify or address complications that many hospitalists see, such as endocarditis, endophthalmitis, and osteomyelitis. The study looked at only nine states, so results may not be generalizable. Nevertheless, the robust analysis suggests an alarming, increasing trend.
Bottom line: As the opioid crisis worsens, hospitalists should consider candidemia in hospitalized IV drug users and should evaluate patients with candidemia for IV drug use.
Citation: Zhang AY et al. The changing epidemiology of candidemia in the United States: Injection drug use as an increasingly common risk factor – Active surveillance in selected sites, United States, 2014-2017. Clin Infect Dis. 2019 Nov 2. doi: 10.1093/cid/ciz1061.
Dr. Raghavan is assistant professor in the division of hospital medicine, Loyola University Medical Center, Maywood, Ill.
Background: Intravenous drug use is an increasingly common risk factor for candidemia as the opioid crisis worsens. This study quantifies this change and characterizes the changing epidemiology of candidemia.
Study design: A cross-sectional study.
Setting: Health departments in nine states.
Synopsis: IV drug users typically have a very distinctive phenotype among all patients with candidemia: They are younger (35 vs. 63 years), are more likely to be homeless, are not black, are smokers; they have hepatitis C, have no malignancies, have polymicrobial bacteremia, and have acquired the infection outside of the hospital. They are much less likely to die of the infection (8.6% vs 27.5%), compared with the non-IV drug users. In four states, the proportion of candidemia associated with IV drug use more than doubled, from 7% to 15% during 2014-2017, representing a possible shift in the epidemiology of candidemia.
The study did not quantify or address complications that many hospitalists see, such as endocarditis, endophthalmitis, and osteomyelitis. The study looked at only nine states, so results may not be generalizable. Nevertheless, the robust analysis suggests an alarming, increasing trend.
Bottom line: As the opioid crisis worsens, hospitalists should consider candidemia in hospitalized IV drug users and should evaluate patients with candidemia for IV drug use.
Citation: Zhang AY et al. The changing epidemiology of candidemia in the United States: Injection drug use as an increasingly common risk factor – Active surveillance in selected sites, United States, 2014-2017. Clin Infect Dis. 2019 Nov 2. doi: 10.1093/cid/ciz1061.
Dr. Raghavan is assistant professor in the division of hospital medicine, Loyola University Medical Center, Maywood, Ill.
Pfizer developing pill to treat COVID-19 symptoms
“If all goes well, and we implement the same speed that we are, and if regulators do the same, and they are, I hope that (it will be available) by the end of the year,” Dr. Bourla said on CNBC’s Squawk Box.
So far, the only antiviral drug authorized for use with COVID-19 is remdesivir, which is produced by Gilead Sciences and must be administered by injection in a health care setting.
An oral drug like the one Pfizer is developing could be taken at home and might keep people out of the hospital.
“Particular attention is on the oral because it provides several advantages,” Dr. Bourla said. “One of them is that you don’t need to go to the hospital to get the treatment, which is the case with all the injectables so far. You could get it at home, and that could be a game-changer.”
The drug might be effective against the emerging variants, he said. Pfizer is also working on an injectable antiviral drug.
Pfizer, with its European partner BioNTech, developed the first coronavirus vaccine authorized for use in the United States and Europe. The Pfizer pill under development would not be a vaccine to protect people from the virus but a drug to treat people who catch the virus.
The company announced in late March that it was starting clinical trials on the oral drug.
In a news release, the company said the oral drug would work by blocking protease, a critical enzyme that the virus needs to replicate. Protease inhibitors are used in medicines to treat HIV and hepatitis C.
A coronavirus vaccine that could be taken as a pill may enter clinical trials in the second quarter of 2021. The oral vaccine is being developed by Oravax Medical, a new joint venture of the Israeli-American company Oramed and the Indian company Premas Biotech. So far, all coronavirus vaccines are injectable.
A version of this article first appeared on WebMD.com.
“If all goes well, and we implement the same speed that we are, and if regulators do the same, and they are, I hope that (it will be available) by the end of the year,” Dr. Bourla said on CNBC’s Squawk Box.
So far, the only antiviral drug authorized for use with COVID-19 is remdesivir, which is produced by Gilead Sciences and must be administered by injection in a health care setting.
An oral drug like the one Pfizer is developing could be taken at home and might keep people out of the hospital.
“Particular attention is on the oral because it provides several advantages,” Dr. Bourla said. “One of them is that you don’t need to go to the hospital to get the treatment, which is the case with all the injectables so far. You could get it at home, and that could be a game-changer.”
The drug might be effective against the emerging variants, he said. Pfizer is also working on an injectable antiviral drug.
Pfizer, with its European partner BioNTech, developed the first coronavirus vaccine authorized for use in the United States and Europe. The Pfizer pill under development would not be a vaccine to protect people from the virus but a drug to treat people who catch the virus.
The company announced in late March that it was starting clinical trials on the oral drug.
In a news release, the company said the oral drug would work by blocking protease, a critical enzyme that the virus needs to replicate. Protease inhibitors are used in medicines to treat HIV and hepatitis C.
A coronavirus vaccine that could be taken as a pill may enter clinical trials in the second quarter of 2021. The oral vaccine is being developed by Oravax Medical, a new joint venture of the Israeli-American company Oramed and the Indian company Premas Biotech. So far, all coronavirus vaccines are injectable.
A version of this article first appeared on WebMD.com.
“If all goes well, and we implement the same speed that we are, and if regulators do the same, and they are, I hope that (it will be available) by the end of the year,” Dr. Bourla said on CNBC’s Squawk Box.
So far, the only antiviral drug authorized for use with COVID-19 is remdesivir, which is produced by Gilead Sciences and must be administered by injection in a health care setting.
An oral drug like the one Pfizer is developing could be taken at home and might keep people out of the hospital.
“Particular attention is on the oral because it provides several advantages,” Dr. Bourla said. “One of them is that you don’t need to go to the hospital to get the treatment, which is the case with all the injectables so far. You could get it at home, and that could be a game-changer.”
The drug might be effective against the emerging variants, he said. Pfizer is also working on an injectable antiviral drug.
Pfizer, with its European partner BioNTech, developed the first coronavirus vaccine authorized for use in the United States and Europe. The Pfizer pill under development would not be a vaccine to protect people from the virus but a drug to treat people who catch the virus.
The company announced in late March that it was starting clinical trials on the oral drug.
In a news release, the company said the oral drug would work by blocking protease, a critical enzyme that the virus needs to replicate. Protease inhibitors are used in medicines to treat HIV and hepatitis C.
A coronavirus vaccine that could be taken as a pill may enter clinical trials in the second quarter of 2021. The oral vaccine is being developed by Oravax Medical, a new joint venture of the Israeli-American company Oramed and the Indian company Premas Biotech. So far, all coronavirus vaccines are injectable.
A version of this article first appeared on WebMD.com.
Psoriasis associated with an increased risk of COVID-19 in real-world study
in patients, compared with those on topical therapy, a new study finds.
“Our study results suggest that psoriasis is an independent risk factor for COVID-19 illness,” study coauthor Jeffrey Liu, a medical student at the University of Southern California, Los Angeles, said in an interview after he presented the findings at the American Academy of Dermatology Virtual Meeting Experience. “And our findings are consistent with the hypothesis that certain systemic agents may confer a protective effect against COVID-19 illness.”
Mr. Liu and coinvestigators used a Symphony Health dataset to analyze the health records of 167,027 U.S. patients diagnosed with psoriasis and a control group of 1,002,162 patients. The participants, all at least 20 years old, had been treated for psoriasis or psoriatic arthritis from May 2019 through Jan. 1, 2020, and were tracked until Nov. 11, 2020.
The ages and races of peoples in the two groups were roughly similar. Overall, 55% were women and 75% were White, and their average age was 58 years. Type 2 diabetes was more common in the psoriasis group than the control group (23% vs. 16%), as was obesity (27% vs. 15%). Of the patients with psoriasis, 60% were on topical treatments, 19% were on oral therapies, and 22% were on biologic therapy, with only a few taking both oral and biologic therapies.
After adjustment for age and gender, patients with psoriasis were 33% more likely than the control group to develop COVID-19 (adjusted incidence rate ratio, 1.33; 95% confidence interval, 1.23-1.38; P < .0001).
In a separate analysis, the gap persisted after adjustment for demographics and comorbidities: Patients with psoriasis had a higher rate of COVID-19 infection vs. controls (adjusted odds ratio, 1.18; 95% CI, 1.13-1.23; P < .0001). Among all patients, non-White race, older age, and comorbidities were all linked to higher risk of COVID-19 (all P < .0001).
Psoriasis might make patients more vulnerable to COVID-19 because the presence of up-regulated genes in psoriatic skin “may lead to systemic hyperinflammation and sensitization of patients with psoriasis to proinflammatory cytokine storm,” Mr. Liu said. This, in turn, may trigger more severe symptomatic disease that requires medical treatment, he said.
Reduced risk, compared with topical therapies
After adjustment for age and gender, those treated with TNF-alpha inhibitors, methotrexate, and apremilast (Otezla) all had statistically lower risks of COVID-19 vs. those on topical therapy (aIRR, 0.82; 95% CI, 0.69-0.95; P < .0029 for TNF-alpha inhibitors; aIRR, 0.75; 95% CI, 0.67-0.86; P < .0001 for methotrexate; and aIRR, 0.69; 95% CI, 0.55-0.85; P < .0006 for apremilast).
Reduced risk held true for those in the separate analysis after adjustment for comorbidities and demographics (respectively, aOR, 0.87; 95% CI, 0.77-1.00; P < .0469; aOR, 0.81; 95% CI, 0.71-0.92; P < .0011; and aOR, 0.70; 95% CI, 0.57-0.87; P < .0014).
Apremilast and methotrexate may boost protection against COVID-19 by inhibiting the body’s production of cytokines, Mr. Liu said.
One message of the study is that “dermatologists should not be scared of prescribing biologics or oral therapies for psoriasis,” the study’s lead author Jashin J. Wu, MD, of the Dermatology Research and Education Foundation in Irvine, Calif., said in an interview.
However, the results on the effects of systemic therapies were not all positive. Interleukin (IL)–17 inhibitors were an outlier: After adjustment for age and gender, patients treated with this class of drugs were 36% more likely to develop COVID-19 than those on oral agents (aIRR, 1.36; 95% CI, 1.13-1.63; P < .0009).
Among patients on biologics, those taking IL-17 inhibitors had the highest risk of COVID-19, Mr. Liu said. “The risk was higher in this class regardless of reference group – general population, the topical cohort, and the oral cohort,” he said. “This may relate to the observation that this biologic class exerts more broad immunosuppressive effects on antiviral host immunity. Notably, large meta-estimates of pivotal trials have observed increased risk of respiratory tract infections for patients on IL-17 inhibitors.”
In an interview, Erica Dommasch, MD, MPH, of the department of dermatology at Beth Israel Deaconess Medical Center, Boston, cautioned that “the data from this study is very hard to interpret.”
It’s likely that some patients with psoriasis on systemic medications “may have been the most careful about limiting exposures,” she said. “Thus, it’s hard to account for behavioral changes in individuals that may have led to the decreased incidence in psoriasis in patients on systemic agents versus topical therapy alone.”
Patients with psoriasis may also be tested more often for COVID-19, and unmeasured comorbidities like chronic kidney disease may play a role too, she said. Still, she added, “it’s reassuring that the authors did not find an increased rate of COVID among psoriasis patients on systemic agents versus topicals alone.” And she agreed with Dr. Wu about the importance of treating psoriasis with therapy beyond topical treatments during the pandemic: “Providers should feel comfortable prescribing systemic medications to psoriasis patients when otherwise appropriate.”
As for the next steps, Dr. Wu said, “we will be exploring more about the prognosis of COVID-19 infection in psoriasis patients. In addition, we will be exploring the relationship of COVID-19 infection with other inflammatory skin diseases, such as atopic dermatitis.”
No study funding is reported. Dr. Wu discloses investigator, consultant, or speaker relationships with AbbVie, Almirall, Amgen, Arcutis, Aristea Therapeutics, Boehringer Ingelheim, Bristol-Myers Squibb, Dermavant, Dr. Reddy’s Laboratories, Eli Lilly, Galderma, Janssen, LEO Pharma, Mindera, Novartis, Regeneron, Sanofi Genzyme, Solius, Sun Pharmaceutical, UCB, Valeant Pharmaceuticals North America, and Zerigo Health. Mr. Liu and Dr. Dommasch have no disclosures.
in patients, compared with those on topical therapy, a new study finds.
“Our study results suggest that psoriasis is an independent risk factor for COVID-19 illness,” study coauthor Jeffrey Liu, a medical student at the University of Southern California, Los Angeles, said in an interview after he presented the findings at the American Academy of Dermatology Virtual Meeting Experience. “And our findings are consistent with the hypothesis that certain systemic agents may confer a protective effect against COVID-19 illness.”
Mr. Liu and coinvestigators used a Symphony Health dataset to analyze the health records of 167,027 U.S. patients diagnosed with psoriasis and a control group of 1,002,162 patients. The participants, all at least 20 years old, had been treated for psoriasis or psoriatic arthritis from May 2019 through Jan. 1, 2020, and were tracked until Nov. 11, 2020.
The ages and races of peoples in the two groups were roughly similar. Overall, 55% were women and 75% were White, and their average age was 58 years. Type 2 diabetes was more common in the psoriasis group than the control group (23% vs. 16%), as was obesity (27% vs. 15%). Of the patients with psoriasis, 60% were on topical treatments, 19% were on oral therapies, and 22% were on biologic therapy, with only a few taking both oral and biologic therapies.
After adjustment for age and gender, patients with psoriasis were 33% more likely than the control group to develop COVID-19 (adjusted incidence rate ratio, 1.33; 95% confidence interval, 1.23-1.38; P < .0001).
In a separate analysis, the gap persisted after adjustment for demographics and comorbidities: Patients with psoriasis had a higher rate of COVID-19 infection vs. controls (adjusted odds ratio, 1.18; 95% CI, 1.13-1.23; P < .0001). Among all patients, non-White race, older age, and comorbidities were all linked to higher risk of COVID-19 (all P < .0001).
Psoriasis might make patients more vulnerable to COVID-19 because the presence of up-regulated genes in psoriatic skin “may lead to systemic hyperinflammation and sensitization of patients with psoriasis to proinflammatory cytokine storm,” Mr. Liu said. This, in turn, may trigger more severe symptomatic disease that requires medical treatment, he said.
Reduced risk, compared with topical therapies
After adjustment for age and gender, those treated with TNF-alpha inhibitors, methotrexate, and apremilast (Otezla) all had statistically lower risks of COVID-19 vs. those on topical therapy (aIRR, 0.82; 95% CI, 0.69-0.95; P < .0029 for TNF-alpha inhibitors; aIRR, 0.75; 95% CI, 0.67-0.86; P < .0001 for methotrexate; and aIRR, 0.69; 95% CI, 0.55-0.85; P < .0006 for apremilast).
Reduced risk held true for those in the separate analysis after adjustment for comorbidities and demographics (respectively, aOR, 0.87; 95% CI, 0.77-1.00; P < .0469; aOR, 0.81; 95% CI, 0.71-0.92; P < .0011; and aOR, 0.70; 95% CI, 0.57-0.87; P < .0014).
Apremilast and methotrexate may boost protection against COVID-19 by inhibiting the body’s production of cytokines, Mr. Liu said.
One message of the study is that “dermatologists should not be scared of prescribing biologics or oral therapies for psoriasis,” the study’s lead author Jashin J. Wu, MD, of the Dermatology Research and Education Foundation in Irvine, Calif., said in an interview.
However, the results on the effects of systemic therapies were not all positive. Interleukin (IL)–17 inhibitors were an outlier: After adjustment for age and gender, patients treated with this class of drugs were 36% more likely to develop COVID-19 than those on oral agents (aIRR, 1.36; 95% CI, 1.13-1.63; P < .0009).
Among patients on biologics, those taking IL-17 inhibitors had the highest risk of COVID-19, Mr. Liu said. “The risk was higher in this class regardless of reference group – general population, the topical cohort, and the oral cohort,” he said. “This may relate to the observation that this biologic class exerts more broad immunosuppressive effects on antiviral host immunity. Notably, large meta-estimates of pivotal trials have observed increased risk of respiratory tract infections for patients on IL-17 inhibitors.”
In an interview, Erica Dommasch, MD, MPH, of the department of dermatology at Beth Israel Deaconess Medical Center, Boston, cautioned that “the data from this study is very hard to interpret.”
It’s likely that some patients with psoriasis on systemic medications “may have been the most careful about limiting exposures,” she said. “Thus, it’s hard to account for behavioral changes in individuals that may have led to the decreased incidence in psoriasis in patients on systemic agents versus topical therapy alone.”
Patients with psoriasis may also be tested more often for COVID-19, and unmeasured comorbidities like chronic kidney disease may play a role too, she said. Still, she added, “it’s reassuring that the authors did not find an increased rate of COVID among psoriasis patients on systemic agents versus topicals alone.” And she agreed with Dr. Wu about the importance of treating psoriasis with therapy beyond topical treatments during the pandemic: “Providers should feel comfortable prescribing systemic medications to psoriasis patients when otherwise appropriate.”
As for the next steps, Dr. Wu said, “we will be exploring more about the prognosis of COVID-19 infection in psoriasis patients. In addition, we will be exploring the relationship of COVID-19 infection with other inflammatory skin diseases, such as atopic dermatitis.”
No study funding is reported. Dr. Wu discloses investigator, consultant, or speaker relationships with AbbVie, Almirall, Amgen, Arcutis, Aristea Therapeutics, Boehringer Ingelheim, Bristol-Myers Squibb, Dermavant, Dr. Reddy’s Laboratories, Eli Lilly, Galderma, Janssen, LEO Pharma, Mindera, Novartis, Regeneron, Sanofi Genzyme, Solius, Sun Pharmaceutical, UCB, Valeant Pharmaceuticals North America, and Zerigo Health. Mr. Liu and Dr. Dommasch have no disclosures.
in patients, compared with those on topical therapy, a new study finds.
“Our study results suggest that psoriasis is an independent risk factor for COVID-19 illness,” study coauthor Jeffrey Liu, a medical student at the University of Southern California, Los Angeles, said in an interview after he presented the findings at the American Academy of Dermatology Virtual Meeting Experience. “And our findings are consistent with the hypothesis that certain systemic agents may confer a protective effect against COVID-19 illness.”
Mr. Liu and coinvestigators used a Symphony Health dataset to analyze the health records of 167,027 U.S. patients diagnosed with psoriasis and a control group of 1,002,162 patients. The participants, all at least 20 years old, had been treated for psoriasis or psoriatic arthritis from May 2019 through Jan. 1, 2020, and were tracked until Nov. 11, 2020.
The ages and races of peoples in the two groups were roughly similar. Overall, 55% were women and 75% were White, and their average age was 58 years. Type 2 diabetes was more common in the psoriasis group than the control group (23% vs. 16%), as was obesity (27% vs. 15%). Of the patients with psoriasis, 60% were on topical treatments, 19% were on oral therapies, and 22% were on biologic therapy, with only a few taking both oral and biologic therapies.
After adjustment for age and gender, patients with psoriasis were 33% more likely than the control group to develop COVID-19 (adjusted incidence rate ratio, 1.33; 95% confidence interval, 1.23-1.38; P < .0001).
In a separate analysis, the gap persisted after adjustment for demographics and comorbidities: Patients with psoriasis had a higher rate of COVID-19 infection vs. controls (adjusted odds ratio, 1.18; 95% CI, 1.13-1.23; P < .0001). Among all patients, non-White race, older age, and comorbidities were all linked to higher risk of COVID-19 (all P < .0001).
Psoriasis might make patients more vulnerable to COVID-19 because the presence of up-regulated genes in psoriatic skin “may lead to systemic hyperinflammation and sensitization of patients with psoriasis to proinflammatory cytokine storm,” Mr. Liu said. This, in turn, may trigger more severe symptomatic disease that requires medical treatment, he said.
Reduced risk, compared with topical therapies
After adjustment for age and gender, those treated with TNF-alpha inhibitors, methotrexate, and apremilast (Otezla) all had statistically lower risks of COVID-19 vs. those on topical therapy (aIRR, 0.82; 95% CI, 0.69-0.95; P < .0029 for TNF-alpha inhibitors; aIRR, 0.75; 95% CI, 0.67-0.86; P < .0001 for methotrexate; and aIRR, 0.69; 95% CI, 0.55-0.85; P < .0006 for apremilast).
Reduced risk held true for those in the separate analysis after adjustment for comorbidities and demographics (respectively, aOR, 0.87; 95% CI, 0.77-1.00; P < .0469; aOR, 0.81; 95% CI, 0.71-0.92; P < .0011; and aOR, 0.70; 95% CI, 0.57-0.87; P < .0014).
Apremilast and methotrexate may boost protection against COVID-19 by inhibiting the body’s production of cytokines, Mr. Liu said.
One message of the study is that “dermatologists should not be scared of prescribing biologics or oral therapies for psoriasis,” the study’s lead author Jashin J. Wu, MD, of the Dermatology Research and Education Foundation in Irvine, Calif., said in an interview.
However, the results on the effects of systemic therapies were not all positive. Interleukin (IL)–17 inhibitors were an outlier: After adjustment for age and gender, patients treated with this class of drugs were 36% more likely to develop COVID-19 than those on oral agents (aIRR, 1.36; 95% CI, 1.13-1.63; P < .0009).
Among patients on biologics, those taking IL-17 inhibitors had the highest risk of COVID-19, Mr. Liu said. “The risk was higher in this class regardless of reference group – general population, the topical cohort, and the oral cohort,” he said. “This may relate to the observation that this biologic class exerts more broad immunosuppressive effects on antiviral host immunity. Notably, large meta-estimates of pivotal trials have observed increased risk of respiratory tract infections for patients on IL-17 inhibitors.”
In an interview, Erica Dommasch, MD, MPH, of the department of dermatology at Beth Israel Deaconess Medical Center, Boston, cautioned that “the data from this study is very hard to interpret.”
It’s likely that some patients with psoriasis on systemic medications “may have been the most careful about limiting exposures,” she said. “Thus, it’s hard to account for behavioral changes in individuals that may have led to the decreased incidence in psoriasis in patients on systemic agents versus topical therapy alone.”
Patients with psoriasis may also be tested more often for COVID-19, and unmeasured comorbidities like chronic kidney disease may play a role too, she said. Still, she added, “it’s reassuring that the authors did not find an increased rate of COVID among psoriasis patients on systemic agents versus topicals alone.” And she agreed with Dr. Wu about the importance of treating psoriasis with therapy beyond topical treatments during the pandemic: “Providers should feel comfortable prescribing systemic medications to psoriasis patients when otherwise appropriate.”
As for the next steps, Dr. Wu said, “we will be exploring more about the prognosis of COVID-19 infection in psoriasis patients. In addition, we will be exploring the relationship of COVID-19 infection with other inflammatory skin diseases, such as atopic dermatitis.”
No study funding is reported. Dr. Wu discloses investigator, consultant, or speaker relationships with AbbVie, Almirall, Amgen, Arcutis, Aristea Therapeutics, Boehringer Ingelheim, Bristol-Myers Squibb, Dermavant, Dr. Reddy’s Laboratories, Eli Lilly, Galderma, Janssen, LEO Pharma, Mindera, Novartis, Regeneron, Sanofi Genzyme, Solius, Sun Pharmaceutical, UCB, Valeant Pharmaceuticals North America, and Zerigo Health. Mr. Liu and Dr. Dommasch have no disclosures.
FROM AAD VMX 2021
CDC: Vaccinated people can mostly drop masks outdoors
After hinting that new guidelines on outdoor mask-wearing were coming, the Centers for Disease Control and Prevention on April 27 officially gave a green light to fully vaccinated people gathering outside in uncrowded activities without the masks that have become so common during the COVID-19 pandemic.
It is a minor – but still significant – step toward the end of pandemic restrictions.
“Over the past year, we have spent a lot of time telling Americans what they cannot do, what they should not do,” CDC director Rochelle Walensky, MD, MPH, said at a White House press briefing. “Today, I’m going to tell you some of the things you can do if you are fully vaccinated.”
President Joe Biden affirmed the new guidelines at a press conference soon after the CDC briefing ended.
,” he said, adding “the bottom line is clear: If you’re vaccinated, you can do more things, more safely, both outdoors as well as indoors.”
President Biden emphasized the role science played in the decision, saying “The CDC is able to make this announcement because our scientists are convinced by the data that the odds of getting or giving the virus to others is very, very low if you’ve both been fully vaccinated and are out in the open air.”
President Biden also said these new guidelines should be an incentive for more people to get vaccinated. “This is another great reason to go get vaccinated now. Now,” he said.
The CDC has long advised that outdoor activities are safer than indoor activities.
“Most of transmission is happening indoors rather than outdoors. Less than 10% of documented transmissions in many studies have occurred outdoors,” said Dr. Walensky. “We also know there’s almost a 20-fold increased risk of transmission in the indoor setting, than the outdoor setting.”
Dr. Walensky said the lower risks outdoors, combined with growing vaccination coverage and falling COVID cases around the country, motivated the change.
The new guidelines come as the share of people in the United States who are vaccinated is growing. About 37% of all eligible Americans are fully vaccinated, according to the CDC. Nearly 54% have had at least one dose.
The new guidelines say unvaccinated people should continue to wear masks outdoors when gathering with others or dining at an outdoor restaurant.
And vaccinated people should continue to wear masks outdoors in crowded settings where social distancing might not always be possible, like a concert or sporting event. People are considered fully vaccinated when they are 2 weeks past their last shot
The CDC guidelines say people who live in the same house don’t need to wear masks if they’re exercising or hanging out together outdoors.
You also don’t need a mask if you’re attending a small, outdoor gathering with fully vaccinated family and friends, whether you’re vaccinated or not.
The new guidelines also say it’s OK for fully vaccinated people to take their masks off outdoors when gathering in a small group of vaccinated and unvaccinated people, but suggest that unvaccinated people should still wear a mask.
Reporter Marcia Frellick contributed to this report.
A version of this article originally appeared on WebMD.com.
After hinting that new guidelines on outdoor mask-wearing were coming, the Centers for Disease Control and Prevention on April 27 officially gave a green light to fully vaccinated people gathering outside in uncrowded activities without the masks that have become so common during the COVID-19 pandemic.
It is a minor – but still significant – step toward the end of pandemic restrictions.
“Over the past year, we have spent a lot of time telling Americans what they cannot do, what they should not do,” CDC director Rochelle Walensky, MD, MPH, said at a White House press briefing. “Today, I’m going to tell you some of the things you can do if you are fully vaccinated.”
President Joe Biden affirmed the new guidelines at a press conference soon after the CDC briefing ended.
,” he said, adding “the bottom line is clear: If you’re vaccinated, you can do more things, more safely, both outdoors as well as indoors.”
President Biden emphasized the role science played in the decision, saying “The CDC is able to make this announcement because our scientists are convinced by the data that the odds of getting or giving the virus to others is very, very low if you’ve both been fully vaccinated and are out in the open air.”
President Biden also said these new guidelines should be an incentive for more people to get vaccinated. “This is another great reason to go get vaccinated now. Now,” he said.
The CDC has long advised that outdoor activities are safer than indoor activities.
“Most of transmission is happening indoors rather than outdoors. Less than 10% of documented transmissions in many studies have occurred outdoors,” said Dr. Walensky. “We also know there’s almost a 20-fold increased risk of transmission in the indoor setting, than the outdoor setting.”
Dr. Walensky said the lower risks outdoors, combined with growing vaccination coverage and falling COVID cases around the country, motivated the change.
The new guidelines come as the share of people in the United States who are vaccinated is growing. About 37% of all eligible Americans are fully vaccinated, according to the CDC. Nearly 54% have had at least one dose.
The new guidelines say unvaccinated people should continue to wear masks outdoors when gathering with others or dining at an outdoor restaurant.
And vaccinated people should continue to wear masks outdoors in crowded settings where social distancing might not always be possible, like a concert or sporting event. People are considered fully vaccinated when they are 2 weeks past their last shot
The CDC guidelines say people who live in the same house don’t need to wear masks if they’re exercising or hanging out together outdoors.
You also don’t need a mask if you’re attending a small, outdoor gathering with fully vaccinated family and friends, whether you’re vaccinated or not.
The new guidelines also say it’s OK for fully vaccinated people to take their masks off outdoors when gathering in a small group of vaccinated and unvaccinated people, but suggest that unvaccinated people should still wear a mask.
Reporter Marcia Frellick contributed to this report.
A version of this article originally appeared on WebMD.com.
After hinting that new guidelines on outdoor mask-wearing were coming, the Centers for Disease Control and Prevention on April 27 officially gave a green light to fully vaccinated people gathering outside in uncrowded activities without the masks that have become so common during the COVID-19 pandemic.
It is a minor – but still significant – step toward the end of pandemic restrictions.
“Over the past year, we have spent a lot of time telling Americans what they cannot do, what they should not do,” CDC director Rochelle Walensky, MD, MPH, said at a White House press briefing. “Today, I’m going to tell you some of the things you can do if you are fully vaccinated.”
President Joe Biden affirmed the new guidelines at a press conference soon after the CDC briefing ended.
,” he said, adding “the bottom line is clear: If you’re vaccinated, you can do more things, more safely, both outdoors as well as indoors.”
President Biden emphasized the role science played in the decision, saying “The CDC is able to make this announcement because our scientists are convinced by the data that the odds of getting or giving the virus to others is very, very low if you’ve both been fully vaccinated and are out in the open air.”
President Biden also said these new guidelines should be an incentive for more people to get vaccinated. “This is another great reason to go get vaccinated now. Now,” he said.
The CDC has long advised that outdoor activities are safer than indoor activities.
“Most of transmission is happening indoors rather than outdoors. Less than 10% of documented transmissions in many studies have occurred outdoors,” said Dr. Walensky. “We also know there’s almost a 20-fold increased risk of transmission in the indoor setting, than the outdoor setting.”
Dr. Walensky said the lower risks outdoors, combined with growing vaccination coverage and falling COVID cases around the country, motivated the change.
The new guidelines come as the share of people in the United States who are vaccinated is growing. About 37% of all eligible Americans are fully vaccinated, according to the CDC. Nearly 54% have had at least one dose.
The new guidelines say unvaccinated people should continue to wear masks outdoors when gathering with others or dining at an outdoor restaurant.
And vaccinated people should continue to wear masks outdoors in crowded settings where social distancing might not always be possible, like a concert or sporting event. People are considered fully vaccinated when they are 2 weeks past their last shot
The CDC guidelines say people who live in the same house don’t need to wear masks if they’re exercising or hanging out together outdoors.
You also don’t need a mask if you’re attending a small, outdoor gathering with fully vaccinated family and friends, whether you’re vaccinated or not.
The new guidelines also say it’s OK for fully vaccinated people to take their masks off outdoors when gathering in a small group of vaccinated and unvaccinated people, but suggest that unvaccinated people should still wear a mask.
Reporter Marcia Frellick contributed to this report.
A version of this article originally appeared on WebMD.com.
Half of patients in hospital for COVID-19 get acute kidney injury
in two independent, European case series presented recently at the International Society of Nephrology: 2021 World Congress. Many of the cases progressed to more severe, stage 3 AKI. Factors linked with incident AKI in the two reports included use of mechanical ventilation, vasopressors, or diuretics, and elevations in inflammatory markers.
The new findings confirm several U.S. reports published during the past year. In those reports, roughly a third of patients hospitalized for COVID-19 developed AKI during their hospital stay, said Jay L. Koyner, MD, during another renal conference, the National Kidney Foundation 2021 Spring Clinical Meetings.
Experience has shown it’s bad news when hospitalized COVID-19 patients develop AKI, which can prove fatal or can lead to the development or worsening of chronic kidney disease (CKD), which in some cases rapidly progresses to end-stage disease.
COVID-19 giving nephrologists an opportunity to improve AKI care
“COVID is giving us an opportunity to do a better job of taking care of patients who develop AKI, which is something that nephrologists have not often excelled at doing,” said Dr. Koyner, professor and director of the nephrology ICU at the University of Chicago.
“Many studies will look at how we can manage COVID-19 patients better after they develop AKI, because I suspect a large number of these patients will wind up with CKD,” Dr. Koyner said during his talk.
He cited several lessons from reports of AKI that occurs in patients hospitalized for COVID-19:
- Preexisting CKD, , and severe COVID-19 appear to be risk factors for developing COVID-related AKI.
- Patients who develop AKI during acutely severe COVID-19 may have slightly worse outcomes than patients without COVID-19 who develop AKI.
- Certain genetic susceptibilities may play a role in developing COVID-19–related AKI.
- Routine follow-up of AKI is generally inadequate and is not standardized, whether AKI develops while ill with COVID-19 or in other settings.
The most encouraging AKI takeaway from COVID-19’s first year is that its incidence among patients hospitalized with COVID-19 appears to have dropped from very high rates early on, possibly because of more routine use of steroids for critically ill patients with COVID-19 and a reduction in the use of ventilators, Dr. Koyner suggested.
In-hospital diuretic treatment links with AKI
One of the World Congress of Nephrology reports involved 1,248 patients admitted with confirmed COVID-19 at two tertiary care hospitals in London during March–May 2020. The average age of the patients was 69 years, 59% were men, and 17% had CKD at admission, as determined on the basis of estimated glomerular filtration rate <60 mL/min per 1.73 m2.
During hospitalization, 487 patients (39%) developed AKI, including 175 (14%) with stage 3 AKI and 109 (9%) who required renal replacement therapy (dialysis or kidney transplant). The incidence of AKI peaked 5 weeks after COVID-19 admission, Paul Jewell and associates from King’s College Hospital, London, reported in a poster.
Multivariate analysis identified several demographic and clinical variables that were significantly linked with an increased risk of developing AKI: male sex (which boosted risk by 55%), Black race (79% higher risk), CKD at admission (triple the risk), being hypertensive on admission (73% higher risk), and being administered diuretics during hospitalization (69% higher risk).
The findings of a risk linked with diuretic use “supports the cautious use of diuretics in patients hospitalized with COVID-19, especially in the presence of background renal impairment,” the authors said.
For patients with incident AKI, the 30-day mortality rate was significantly increased; mortality was 59% higher among patients who developed stage 1 AKI and was roughly triple among patients who developed stage 2 or 3 AKI.
Second report links ventilation, vasopressors with worse AKI
A separate report from clinicians at Charité Hospital, Berlin, retrospectively analyzed 223 patients admitted with symptomatic COVID-19 to three Charité sites during March–June 2020. During hospitalization, 117 patients (52%) developed AKI, including 70 (31%) with stage 3 disease; 67 (30%) required renal replacement therapy. Half the patients with stage 3 AKI required ICU admission.
Compared with patients with less severe AKI, patients who developed stage 3 AKI were more often male, older, and had a higher body mass index.
In a multivariate model, compared with patients who developed less severe AKI, those who developed stage 3 disease also were significantly more likely to have received mechanical ventilation or vasopressor drugs and were more likely to have increased levels of leukocytes or procalcitonin, two inflammatory markers, Jan-Hendrink B. Hardenburg, MD, a Charité nephrologist, and associates reported in a poster at the meeting.
Mechanical ventilation was linked with a sixfold higher rate of stage 3 AKI, and treatment with vasopressor drugs was linked with a threefold higher rate. Elevations in procalcitonin or leukocyte levels were linked with about 60% increases in rates of stage 3 AKI. For both of these risk factors, temporal relationships were tighter; increases in both values appeared just before onset of stage 3 disease.
Dr. Joyner has been a speaker on behalf of NXStage Medical; a consultant to Astute Medical, Baxter, Mallinckrodt, Pfizer, and Sphingotec; and he has received research funding from Astute, Bioporto, NxStage, and Satellite Healthcare. Mr. Jewell and Dr. Hardenburg disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
in two independent, European case series presented recently at the International Society of Nephrology: 2021 World Congress. Many of the cases progressed to more severe, stage 3 AKI. Factors linked with incident AKI in the two reports included use of mechanical ventilation, vasopressors, or diuretics, and elevations in inflammatory markers.
The new findings confirm several U.S. reports published during the past year. In those reports, roughly a third of patients hospitalized for COVID-19 developed AKI during their hospital stay, said Jay L. Koyner, MD, during another renal conference, the National Kidney Foundation 2021 Spring Clinical Meetings.
Experience has shown it’s bad news when hospitalized COVID-19 patients develop AKI, which can prove fatal or can lead to the development or worsening of chronic kidney disease (CKD), which in some cases rapidly progresses to end-stage disease.
COVID-19 giving nephrologists an opportunity to improve AKI care
“COVID is giving us an opportunity to do a better job of taking care of patients who develop AKI, which is something that nephrologists have not often excelled at doing,” said Dr. Koyner, professor and director of the nephrology ICU at the University of Chicago.
“Many studies will look at how we can manage COVID-19 patients better after they develop AKI, because I suspect a large number of these patients will wind up with CKD,” Dr. Koyner said during his talk.
He cited several lessons from reports of AKI that occurs in patients hospitalized for COVID-19:
- Preexisting CKD, , and severe COVID-19 appear to be risk factors for developing COVID-related AKI.
- Patients who develop AKI during acutely severe COVID-19 may have slightly worse outcomes than patients without COVID-19 who develop AKI.
- Certain genetic susceptibilities may play a role in developing COVID-19–related AKI.
- Routine follow-up of AKI is generally inadequate and is not standardized, whether AKI develops while ill with COVID-19 or in other settings.
The most encouraging AKI takeaway from COVID-19’s first year is that its incidence among patients hospitalized with COVID-19 appears to have dropped from very high rates early on, possibly because of more routine use of steroids for critically ill patients with COVID-19 and a reduction in the use of ventilators, Dr. Koyner suggested.
In-hospital diuretic treatment links with AKI
One of the World Congress of Nephrology reports involved 1,248 patients admitted with confirmed COVID-19 at two tertiary care hospitals in London during March–May 2020. The average age of the patients was 69 years, 59% were men, and 17% had CKD at admission, as determined on the basis of estimated glomerular filtration rate <60 mL/min per 1.73 m2.
During hospitalization, 487 patients (39%) developed AKI, including 175 (14%) with stage 3 AKI and 109 (9%) who required renal replacement therapy (dialysis or kidney transplant). The incidence of AKI peaked 5 weeks after COVID-19 admission, Paul Jewell and associates from King’s College Hospital, London, reported in a poster.
Multivariate analysis identified several demographic and clinical variables that were significantly linked with an increased risk of developing AKI: male sex (which boosted risk by 55%), Black race (79% higher risk), CKD at admission (triple the risk), being hypertensive on admission (73% higher risk), and being administered diuretics during hospitalization (69% higher risk).
The findings of a risk linked with diuretic use “supports the cautious use of diuretics in patients hospitalized with COVID-19, especially in the presence of background renal impairment,” the authors said.
For patients with incident AKI, the 30-day mortality rate was significantly increased; mortality was 59% higher among patients who developed stage 1 AKI and was roughly triple among patients who developed stage 2 or 3 AKI.
Second report links ventilation, vasopressors with worse AKI
A separate report from clinicians at Charité Hospital, Berlin, retrospectively analyzed 223 patients admitted with symptomatic COVID-19 to three Charité sites during March–June 2020. During hospitalization, 117 patients (52%) developed AKI, including 70 (31%) with stage 3 disease; 67 (30%) required renal replacement therapy. Half the patients with stage 3 AKI required ICU admission.
Compared with patients with less severe AKI, patients who developed stage 3 AKI were more often male, older, and had a higher body mass index.
In a multivariate model, compared with patients who developed less severe AKI, those who developed stage 3 disease also were significantly more likely to have received mechanical ventilation or vasopressor drugs and were more likely to have increased levels of leukocytes or procalcitonin, two inflammatory markers, Jan-Hendrink B. Hardenburg, MD, a Charité nephrologist, and associates reported in a poster at the meeting.
Mechanical ventilation was linked with a sixfold higher rate of stage 3 AKI, and treatment with vasopressor drugs was linked with a threefold higher rate. Elevations in procalcitonin or leukocyte levels were linked with about 60% increases in rates of stage 3 AKI. For both of these risk factors, temporal relationships were tighter; increases in both values appeared just before onset of stage 3 disease.
Dr. Joyner has been a speaker on behalf of NXStage Medical; a consultant to Astute Medical, Baxter, Mallinckrodt, Pfizer, and Sphingotec; and he has received research funding from Astute, Bioporto, NxStage, and Satellite Healthcare. Mr. Jewell and Dr. Hardenburg disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
in two independent, European case series presented recently at the International Society of Nephrology: 2021 World Congress. Many of the cases progressed to more severe, stage 3 AKI. Factors linked with incident AKI in the two reports included use of mechanical ventilation, vasopressors, or diuretics, and elevations in inflammatory markers.
The new findings confirm several U.S. reports published during the past year. In those reports, roughly a third of patients hospitalized for COVID-19 developed AKI during their hospital stay, said Jay L. Koyner, MD, during another renal conference, the National Kidney Foundation 2021 Spring Clinical Meetings.
Experience has shown it’s bad news when hospitalized COVID-19 patients develop AKI, which can prove fatal or can lead to the development or worsening of chronic kidney disease (CKD), which in some cases rapidly progresses to end-stage disease.
COVID-19 giving nephrologists an opportunity to improve AKI care
“COVID is giving us an opportunity to do a better job of taking care of patients who develop AKI, which is something that nephrologists have not often excelled at doing,” said Dr. Koyner, professor and director of the nephrology ICU at the University of Chicago.
“Many studies will look at how we can manage COVID-19 patients better after they develop AKI, because I suspect a large number of these patients will wind up with CKD,” Dr. Koyner said during his talk.
He cited several lessons from reports of AKI that occurs in patients hospitalized for COVID-19:
- Preexisting CKD, , and severe COVID-19 appear to be risk factors for developing COVID-related AKI.
- Patients who develop AKI during acutely severe COVID-19 may have slightly worse outcomes than patients without COVID-19 who develop AKI.
- Certain genetic susceptibilities may play a role in developing COVID-19–related AKI.
- Routine follow-up of AKI is generally inadequate and is not standardized, whether AKI develops while ill with COVID-19 or in other settings.
The most encouraging AKI takeaway from COVID-19’s first year is that its incidence among patients hospitalized with COVID-19 appears to have dropped from very high rates early on, possibly because of more routine use of steroids for critically ill patients with COVID-19 and a reduction in the use of ventilators, Dr. Koyner suggested.
In-hospital diuretic treatment links with AKI
One of the World Congress of Nephrology reports involved 1,248 patients admitted with confirmed COVID-19 at two tertiary care hospitals in London during March–May 2020. The average age of the patients was 69 years, 59% were men, and 17% had CKD at admission, as determined on the basis of estimated glomerular filtration rate <60 mL/min per 1.73 m2.
During hospitalization, 487 patients (39%) developed AKI, including 175 (14%) with stage 3 AKI and 109 (9%) who required renal replacement therapy (dialysis or kidney transplant). The incidence of AKI peaked 5 weeks after COVID-19 admission, Paul Jewell and associates from King’s College Hospital, London, reported in a poster.
Multivariate analysis identified several demographic and clinical variables that were significantly linked with an increased risk of developing AKI: male sex (which boosted risk by 55%), Black race (79% higher risk), CKD at admission (triple the risk), being hypertensive on admission (73% higher risk), and being administered diuretics during hospitalization (69% higher risk).
The findings of a risk linked with diuretic use “supports the cautious use of diuretics in patients hospitalized with COVID-19, especially in the presence of background renal impairment,” the authors said.
For patients with incident AKI, the 30-day mortality rate was significantly increased; mortality was 59% higher among patients who developed stage 1 AKI and was roughly triple among patients who developed stage 2 or 3 AKI.
Second report links ventilation, vasopressors with worse AKI
A separate report from clinicians at Charité Hospital, Berlin, retrospectively analyzed 223 patients admitted with symptomatic COVID-19 to three Charité sites during March–June 2020. During hospitalization, 117 patients (52%) developed AKI, including 70 (31%) with stage 3 disease; 67 (30%) required renal replacement therapy. Half the patients with stage 3 AKI required ICU admission.
Compared with patients with less severe AKI, patients who developed stage 3 AKI were more often male, older, and had a higher body mass index.
In a multivariate model, compared with patients who developed less severe AKI, those who developed stage 3 disease also were significantly more likely to have received mechanical ventilation or vasopressor drugs and were more likely to have increased levels of leukocytes or procalcitonin, two inflammatory markers, Jan-Hendrink B. Hardenburg, MD, a Charité nephrologist, and associates reported in a poster at the meeting.
Mechanical ventilation was linked with a sixfold higher rate of stage 3 AKI, and treatment with vasopressor drugs was linked with a threefold higher rate. Elevations in procalcitonin or leukocyte levels were linked with about 60% increases in rates of stage 3 AKI. For both of these risk factors, temporal relationships were tighter; increases in both values appeared just before onset of stage 3 disease.
Dr. Joyner has been a speaker on behalf of NXStage Medical; a consultant to Astute Medical, Baxter, Mallinckrodt, Pfizer, and Sphingotec; and he has received research funding from Astute, Bioporto, NxStage, and Satellite Healthcare. Mr. Jewell and Dr. Hardenburg disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Infective endocarditis from IV drug use tied to hemorrhagic stroke
One consequence of the ongoing opioid epidemic in the United States may be an increase in the number of hemorrhagic strokes caused by infective endocarditis, research suggests.
Intravenous drug use (IVDU) can cause this bacterial infection of the heart. In a single-center study, infective endocarditis was associated with an increase in the risk for hemorrhagic stroke as well as an increase in health care use and costs.
“Patients who are known IV drug users who have endocarditis should be more carefully screened for symptoms of cardiovascular disease,” Shahid M. Nimjee, MD, PhD, associate professor of neurosurgery and surgical director of the Comprehensive Stroke Center at the Ohio State University Wexner Medical Center, Columbus, said in a press release.
The findings were presented at the International Stroke Conference sponsored by the American Heart Association.
In the United States, 47,000 patients are treated in the hospital for endocarditis each year. Endocarditis increases the risk for stroke, which can entail significant morbidity and mortality, the authors noted.
IVDU is a risk factor for endocarditis. In the context of the opioid epidemic, Dr. Nimjee and colleagues sought to compare the risk for stroke among patients with endocarditis from IVDU with the risk among patients with endocarditis from other causes.
They retrospectively studied patients who had undergone treatment for infective endocarditis at Wexner Medical Center between Jan. 1, 2014, and July 1, 2018. They examined patients’ concomitant intravenous drug abuse and evaluated demographics, risk factors, and associated costs.
Dramatic increase
In all, 351 patients met the study’s inclusion criteria, and 170 (48%) had a history of IVDU-associated endocarditis. The incidence of patients with IVDU-associated endocarditis increased 630% from 2014 to 2018.
The prevalence of overall intracranial hemorrhage was increased among patients with IVDU, compared with those without (25.9% vs. 13.9%; P = .005).
This increase in prevalence included increases in intraparenchymal hemorrhage (12.4% vs. 5.1%; P = .012), subarachnoid hemorrhage (17.6% vs. 4.4%; P = .0001), and cerebral microbleeds (14.1% vs. 7.2%; P = .022).
IVDU also was associated with an increase in prevalence of infectious intracranial aneurysm (10.6% vs. 1.8%; P = .0001) and brain abscess (4.7% vs. 1.1%; P = .025).
Compared with patients with endocarditis from other causes, significantly higher numbers of patients with IVDU-associated endocarditis were homeless (5.9% vs. 1.1%; P = .014), uninsured (10.0% vs. 2.8%; P = .005), and unemployed (75.9% vs. 31.7%; P = .0001).
Medical costs were more than twice as high among patients with endocarditis from IVDU than among those with endocarditis from other causes. The difference in health care costs during admission per patient was more than $100,000.
“The wider societal impact of the opioid epidemic is not well understood,” Dr. Nimjee said in the press release. “Our research suggests that the impact of the opioid epidemic is far-reaching and contributes to increased costs in the criminal justice, health care systems, and the workplace. The increased costs can be particularly substantial for stroke care.”
Nationwide data desirable
“Past publications from the U.S. have shown an increase in incidence of IVDU-related endocarditis, and the current publication emphasizes this worrying trend,” Manuel Bolognese, MD, head of the stroke center at the Lucerne (Switzerland) Cantonal Hospital, said in an interview. “The higher degree of hemorrhagic strokes and brain abscesses as further complications is alarming as well and shows that IVDU-related endocarditis is becoming a more and more relevant medical problem in the U.S., with high morbidity and mortality.”
The study period is long enough to show a clear trend of increasing incidence of IVDU-related endocarditis, Dr. Bolognese said. The study’s biggest weaknesses are its retrospective design and restriction to a single center.
“Without knowing the prevalence of drug abuse and the socioeconomical situation in Columbus, it is difficult to generalize these findings to other regions in the U.S.A. or even abroad,” he said.
Also, the abstract does not provide some essential information, said Dr. Bolognese. It would be important to know which valve was affected in each patient, which bacteria were identified, whether patients also used nonopioid drugs, and what each patient’s immune status was.
A lack of sterile material such as syringes could explain the apparent association between IVDU-associated endocarditis and low socioeconomic status, said Dr. Bolognese. Delayed presentation to medical institutions because of a lack of insurance could have led to a more complicated course.
“It would be interesting to see numbers from a broader spectrum in a nationwide registry,” said Dr. Bolognese. “It might be worth studying interventions to improve the hygienic aspects (like supply of sterile material, especially in the most vulnerable groups, like homeless people) or to provide easier access to emergency health care despite lack of insurance, which could decrease the incidence of IVDU.”
Dr. Nimjee and Dr. Bolognese disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
One consequence of the ongoing opioid epidemic in the United States may be an increase in the number of hemorrhagic strokes caused by infective endocarditis, research suggests.
Intravenous drug use (IVDU) can cause this bacterial infection of the heart. In a single-center study, infective endocarditis was associated with an increase in the risk for hemorrhagic stroke as well as an increase in health care use and costs.
“Patients who are known IV drug users who have endocarditis should be more carefully screened for symptoms of cardiovascular disease,” Shahid M. Nimjee, MD, PhD, associate professor of neurosurgery and surgical director of the Comprehensive Stroke Center at the Ohio State University Wexner Medical Center, Columbus, said in a press release.
The findings were presented at the International Stroke Conference sponsored by the American Heart Association.
In the United States, 47,000 patients are treated in the hospital for endocarditis each year. Endocarditis increases the risk for stroke, which can entail significant morbidity and mortality, the authors noted.
IVDU is a risk factor for endocarditis. In the context of the opioid epidemic, Dr. Nimjee and colleagues sought to compare the risk for stroke among patients with endocarditis from IVDU with the risk among patients with endocarditis from other causes.
They retrospectively studied patients who had undergone treatment for infective endocarditis at Wexner Medical Center between Jan. 1, 2014, and July 1, 2018. They examined patients’ concomitant intravenous drug abuse and evaluated demographics, risk factors, and associated costs.
Dramatic increase
In all, 351 patients met the study’s inclusion criteria, and 170 (48%) had a history of IVDU-associated endocarditis. The incidence of patients with IVDU-associated endocarditis increased 630% from 2014 to 2018.
The prevalence of overall intracranial hemorrhage was increased among patients with IVDU, compared with those without (25.9% vs. 13.9%; P = .005).
This increase in prevalence included increases in intraparenchymal hemorrhage (12.4% vs. 5.1%; P = .012), subarachnoid hemorrhage (17.6% vs. 4.4%; P = .0001), and cerebral microbleeds (14.1% vs. 7.2%; P = .022).
IVDU also was associated with an increase in prevalence of infectious intracranial aneurysm (10.6% vs. 1.8%; P = .0001) and brain abscess (4.7% vs. 1.1%; P = .025).
Compared with patients with endocarditis from other causes, significantly higher numbers of patients with IVDU-associated endocarditis were homeless (5.9% vs. 1.1%; P = .014), uninsured (10.0% vs. 2.8%; P = .005), and unemployed (75.9% vs. 31.7%; P = .0001).
Medical costs were more than twice as high among patients with endocarditis from IVDU than among those with endocarditis from other causes. The difference in health care costs during admission per patient was more than $100,000.
“The wider societal impact of the opioid epidemic is not well understood,” Dr. Nimjee said in the press release. “Our research suggests that the impact of the opioid epidemic is far-reaching and contributes to increased costs in the criminal justice, health care systems, and the workplace. The increased costs can be particularly substantial for stroke care.”
Nationwide data desirable
“Past publications from the U.S. have shown an increase in incidence of IVDU-related endocarditis, and the current publication emphasizes this worrying trend,” Manuel Bolognese, MD, head of the stroke center at the Lucerne (Switzerland) Cantonal Hospital, said in an interview. “The higher degree of hemorrhagic strokes and brain abscesses as further complications is alarming as well and shows that IVDU-related endocarditis is becoming a more and more relevant medical problem in the U.S., with high morbidity and mortality.”
The study period is long enough to show a clear trend of increasing incidence of IVDU-related endocarditis, Dr. Bolognese said. The study’s biggest weaknesses are its retrospective design and restriction to a single center.
“Without knowing the prevalence of drug abuse and the socioeconomical situation in Columbus, it is difficult to generalize these findings to other regions in the U.S.A. or even abroad,” he said.
Also, the abstract does not provide some essential information, said Dr. Bolognese. It would be important to know which valve was affected in each patient, which bacteria were identified, whether patients also used nonopioid drugs, and what each patient’s immune status was.
A lack of sterile material such as syringes could explain the apparent association between IVDU-associated endocarditis and low socioeconomic status, said Dr. Bolognese. Delayed presentation to medical institutions because of a lack of insurance could have led to a more complicated course.
“It would be interesting to see numbers from a broader spectrum in a nationwide registry,” said Dr. Bolognese. “It might be worth studying interventions to improve the hygienic aspects (like supply of sterile material, especially in the most vulnerable groups, like homeless people) or to provide easier access to emergency health care despite lack of insurance, which could decrease the incidence of IVDU.”
Dr. Nimjee and Dr. Bolognese disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
One consequence of the ongoing opioid epidemic in the United States may be an increase in the number of hemorrhagic strokes caused by infective endocarditis, research suggests.
Intravenous drug use (IVDU) can cause this bacterial infection of the heart. In a single-center study, infective endocarditis was associated with an increase in the risk for hemorrhagic stroke as well as an increase in health care use and costs.
“Patients who are known IV drug users who have endocarditis should be more carefully screened for symptoms of cardiovascular disease,” Shahid M. Nimjee, MD, PhD, associate professor of neurosurgery and surgical director of the Comprehensive Stroke Center at the Ohio State University Wexner Medical Center, Columbus, said in a press release.
The findings were presented at the International Stroke Conference sponsored by the American Heart Association.
In the United States, 47,000 patients are treated in the hospital for endocarditis each year. Endocarditis increases the risk for stroke, which can entail significant morbidity and mortality, the authors noted.
IVDU is a risk factor for endocarditis. In the context of the opioid epidemic, Dr. Nimjee and colleagues sought to compare the risk for stroke among patients with endocarditis from IVDU with the risk among patients with endocarditis from other causes.
They retrospectively studied patients who had undergone treatment for infective endocarditis at Wexner Medical Center between Jan. 1, 2014, and July 1, 2018. They examined patients’ concomitant intravenous drug abuse and evaluated demographics, risk factors, and associated costs.
Dramatic increase
In all, 351 patients met the study’s inclusion criteria, and 170 (48%) had a history of IVDU-associated endocarditis. The incidence of patients with IVDU-associated endocarditis increased 630% from 2014 to 2018.
The prevalence of overall intracranial hemorrhage was increased among patients with IVDU, compared with those without (25.9% vs. 13.9%; P = .005).
This increase in prevalence included increases in intraparenchymal hemorrhage (12.4% vs. 5.1%; P = .012), subarachnoid hemorrhage (17.6% vs. 4.4%; P = .0001), and cerebral microbleeds (14.1% vs. 7.2%; P = .022).
IVDU also was associated with an increase in prevalence of infectious intracranial aneurysm (10.6% vs. 1.8%; P = .0001) and brain abscess (4.7% vs. 1.1%; P = .025).
Compared with patients with endocarditis from other causes, significantly higher numbers of patients with IVDU-associated endocarditis were homeless (5.9% vs. 1.1%; P = .014), uninsured (10.0% vs. 2.8%; P = .005), and unemployed (75.9% vs. 31.7%; P = .0001).
Medical costs were more than twice as high among patients with endocarditis from IVDU than among those with endocarditis from other causes. The difference in health care costs during admission per patient was more than $100,000.
“The wider societal impact of the opioid epidemic is not well understood,” Dr. Nimjee said in the press release. “Our research suggests that the impact of the opioid epidemic is far-reaching and contributes to increased costs in the criminal justice, health care systems, and the workplace. The increased costs can be particularly substantial for stroke care.”
Nationwide data desirable
“Past publications from the U.S. have shown an increase in incidence of IVDU-related endocarditis, and the current publication emphasizes this worrying trend,” Manuel Bolognese, MD, head of the stroke center at the Lucerne (Switzerland) Cantonal Hospital, said in an interview. “The higher degree of hemorrhagic strokes and brain abscesses as further complications is alarming as well and shows that IVDU-related endocarditis is becoming a more and more relevant medical problem in the U.S., with high morbidity and mortality.”
The study period is long enough to show a clear trend of increasing incidence of IVDU-related endocarditis, Dr. Bolognese said. The study’s biggest weaknesses are its retrospective design and restriction to a single center.
“Without knowing the prevalence of drug abuse and the socioeconomical situation in Columbus, it is difficult to generalize these findings to other regions in the U.S.A. or even abroad,” he said.
Also, the abstract does not provide some essential information, said Dr. Bolognese. It would be important to know which valve was affected in each patient, which bacteria were identified, whether patients also used nonopioid drugs, and what each patient’s immune status was.
A lack of sterile material such as syringes could explain the apparent association between IVDU-associated endocarditis and low socioeconomic status, said Dr. Bolognese. Delayed presentation to medical institutions because of a lack of insurance could have led to a more complicated course.
“It would be interesting to see numbers from a broader spectrum in a nationwide registry,” said Dr. Bolognese. “It might be worth studying interventions to improve the hygienic aspects (like supply of sterile material, especially in the most vulnerable groups, like homeless people) or to provide easier access to emergency health care despite lack of insurance, which could decrease the incidence of IVDU.”
Dr. Nimjee and Dr. Bolognese disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Pediatric bronchiolitis: Less is more
A common cause of infant morbidity and hospitalization in developed countries, infant viral bronchiolitis, has long been bedeviled by treatment uncertainty beyond supportive care.
Rationales for most pharmacologic treatments continue to be debated, and clinical practice guidelines generally advise respiratory and hydration support, discouraging the use of chest radiography, albuterol, glucocorticoids, antibiotics, and epinephrine.
Despite evidence that the latter interventions are ineffective, they are still too often applied, according to two recent studies, one in Pediatrics, the other in JAMA Pediatrics.
“The pull of the therapeutic vacuum surrounding this disease has been noted in the pages of this journal for at least 50 years, with Wright and Beem writing in 1965 that ‘energies should not be frittered away by the annoyance of unnecessary or futile medications and procedures’ for the child with bronchiolitis,” said emergency physicians Matthew J. Lipshaw, MD, MS, of the Cincinnati Children’s Hospital Medical Center, and Todd A. Florin, MD, MSCE, of Ann and Robert H. Lurie Children’s Hospital of Chicago.
These remarks came in their editorial in Pediatrics wryly titled: “Don’t Just Do Something, Stand There” and published online to accompany a recent study of three network meta-analyses.
Led by Sarah A. Elliott, PhD, of the Alberta Research Centre for Health Evidence at the University of Alberta in Edmonton, this analysis amalgamated 150 randomized, controlled trials comparing a placebo or active comparator with any bronchodilator, glucocorticoid steroid, hypertonic saline solution, antibiotic, helium-oxygen therapy, or high-flow oxygen therapy. It then looked at the following outcomes in children aged 2 years and younger: hospital admission rate on day 1, hospital admission rate within 7 days, and total hospital length of stay.
Few treatments seemed more effective than nebulized placebo (0.9% saline) for short-term outcomes, the authors found. While nebulized epinephrine and nebulized hypertonic saline plus salbutamol appeared to reduce admission rates during the index ED presentation, and hypertonic saline, alone or in combination with epinephrine, seemed to reduce hospital stays, such treatment had no effect on admissions within 7 days of initial presentation. Furthermore, most benefits disappeared in higher-quality studies.
Concluding, albeit with weak evidence and low confidence, that some benefit might accrue with hypertonic saline with salbutamol to reduce admission rates on initial presentation to the ED, the authors called for well-designed studies on treatments in inpatients and outpatients.
According to Dr. Lipshaw, assistant professor of clinical pediatrics, the lack of benefit observed in superior studies limits the applicability of Dr. Elliott and colleagues’ results to immediate clinical practice. “These findings could be used, however, to target future high-quality studies toward the medications that they found might be useful,” he said in an interview.
For the present, other recent research augurs well for strategically reducing unnecessary care. In a paper published online in JAMA Pediatrics, Libby Haskell, MN, of the ED at Starship Children’s Hospital in Auckland, New Zealand, and associates reported on a cluster-randomized, controlled trial of targeted interventions.
Conducted in 2017 at 26 hospitals and with 3,727 babies in New Zealand and Australia, the study addressed drivers of non–evidence-based approaches with behavior-modifying approaches such as on-site clinical leads, stakeholder meetings, a train-the-trainer workshop, education, and audit and feedback.
The authors reported a 14.1% difference in rates of compliance during the first 24 hours of hospitalization favoring the intervention group for all five bronchiolitis guideline recommendations. The greatest change was seen in albuterol and chest radiography use, with other improvements in ED visits, inpatient consultations, and throughout hospitalization.
“These results provide clinicians and hospitals with clear implementation strategies to address unnecessary treatment of infants with bronchiolitis,” Dr. Haskell’s group wrote. Dr. Lipshaw agreed that multifaceted deimplementation packages including clinician and family education, audit and feedback, and clinical decision support have been successful. “Haskell et al. demonstrated that it is possible to successfully deimplement non–evidence-based practices for bronchiolitis with targeted inventions,” he said. “It would be wonderful to see their success replicated in the U.S.”
Why the slow adoption of guidelines?
The American Academy of Pediatrics issued bronchiolitis guidelines for babies to 23 months in 2014 and updated them in 2018. Why, then, has care in some centers been seemingly all over the map and counter to guidelines? “Both parents and clinicians are acting in what they believe to be the best interests of the child, and in the absence of high-value interventions, can feel the need to do something, even if that something is not supported by evidence,” Dr. Lipshaw said.
Furthermore, with children in obvious distress, breathing fast and with difficulty, and sometimes unable to eat or drink, “we feel like we should have some way to make them feel better quicker. Unfortunately, none of the medications we have tried seem to be useful for most children, and we are left with supportive care measures such as suctioning their noses, giving them oxygen if their oxygen is low, and giving them fluids if they are dehydrated.”
Other physicians agree that taking a less-is-more approach can be challenging and even counterintuitive. “To families, seeing their child’s doctor ‘doing less’ can be frustrating,” admitted Diana S. Lee, MD, assistant professor of pediatrics at Icahn School of Medicine at Mount Sinai, New York.
Beyond that, altering practice behavior will need more than guidelines, Dr. Lee said in an interview. “Haskell et al. showed targeted behavior-change interventions improved compliance with bronchiolitis guidelines, but such change requires motivation and resources, and the sustainability of this effect over time remains to be seen.”
At Dr. Lipshaw’s institution, treatment depends on the attending physician, “but we have an emergency department care algorithm, which does not recommend any inhaled medications or steroids in accordance with the 2014 AAP guidelines,” he said.
Similarly at Mount Sinai, practitioners strive to follow the AAP guidelines, although their implementation has not been immediate, Dr. Lee said. “This is a situation where we must make the effort to choose not to do more, given current evidence.”
But Michelle Dunn, MD, an attending physician in the division of general pediatrics at the Children’s Hospital of Philadelphia, said the American practice norm already tends more to the observance than the breach of the guidelines, noting that since 2014 quality improvement efforts have been made throughout the country. “At our institution, we have effectively reduced the use of albuterol in patients with bronchiolitis and we use evidence-based therapy as much as possible, which in the case of bronchiolitis generally involves supportive management alone,” she said in an interview.
Still, Dr. Dunn added, many patients receive unnecessary diagnostic testing and ineffective therapies, with some providers facing psychological barriers to doing less. “However, with more and more evidence to support this, hopefully, physicians will become more comfortable with this.”
To that end, Dr. Lipshaw’s editorial urges physicians to “curb the rampant use of therapies repeatedly revealed to be ineffective,” citing team engagement, clear practice guidelines, and information technology as key factors in deimplementation. In the meantime, his mantra remains: “Don’t just do something, stand there.”
The study by Dr. Elliot and colleagues was supported by the Canadian Institutes of Health Research Knowledge Synthesis grant program. One coauthor is supported by a University of Ottawa Tier I Research Chair in Pediatric Emergency Medicine. Another is supported by a Tier 1 Canada Research Chair in Knowledge Synthesis and Translation and the Stollery Science Laboratory. Dr. Lipshaw and Dr. Florin disclosed no financial relationships relevant to their commentary. Dr. Haskell and colleagues were supported, variously, by the National Health and Medical Research Council of New Zealand, the Center of Research Excellence for Pediatric Emergency Medicine, the Victorian Government’s Operational Infrastructure Support Program, Cure Kids New Zealand, the Royal Children’s Hospital Foundation, and the Starship Foundation. Dr. Lee and Dr. Dunn had no competing interests to disclose with regard to their comments.
A common cause of infant morbidity and hospitalization in developed countries, infant viral bronchiolitis, has long been bedeviled by treatment uncertainty beyond supportive care.
Rationales for most pharmacologic treatments continue to be debated, and clinical practice guidelines generally advise respiratory and hydration support, discouraging the use of chest radiography, albuterol, glucocorticoids, antibiotics, and epinephrine.
Despite evidence that the latter interventions are ineffective, they are still too often applied, according to two recent studies, one in Pediatrics, the other in JAMA Pediatrics.
“The pull of the therapeutic vacuum surrounding this disease has been noted in the pages of this journal for at least 50 years, with Wright and Beem writing in 1965 that ‘energies should not be frittered away by the annoyance of unnecessary or futile medications and procedures’ for the child with bronchiolitis,” said emergency physicians Matthew J. Lipshaw, MD, MS, of the Cincinnati Children’s Hospital Medical Center, and Todd A. Florin, MD, MSCE, of Ann and Robert H. Lurie Children’s Hospital of Chicago.
These remarks came in their editorial in Pediatrics wryly titled: “Don’t Just Do Something, Stand There” and published online to accompany a recent study of three network meta-analyses.
Led by Sarah A. Elliott, PhD, of the Alberta Research Centre for Health Evidence at the University of Alberta in Edmonton, this analysis amalgamated 150 randomized, controlled trials comparing a placebo or active comparator with any bronchodilator, glucocorticoid steroid, hypertonic saline solution, antibiotic, helium-oxygen therapy, or high-flow oxygen therapy. It then looked at the following outcomes in children aged 2 years and younger: hospital admission rate on day 1, hospital admission rate within 7 days, and total hospital length of stay.
Few treatments seemed more effective than nebulized placebo (0.9% saline) for short-term outcomes, the authors found. While nebulized epinephrine and nebulized hypertonic saline plus salbutamol appeared to reduce admission rates during the index ED presentation, and hypertonic saline, alone or in combination with epinephrine, seemed to reduce hospital stays, such treatment had no effect on admissions within 7 days of initial presentation. Furthermore, most benefits disappeared in higher-quality studies.
Concluding, albeit with weak evidence and low confidence, that some benefit might accrue with hypertonic saline with salbutamol to reduce admission rates on initial presentation to the ED, the authors called for well-designed studies on treatments in inpatients and outpatients.
According to Dr. Lipshaw, assistant professor of clinical pediatrics, the lack of benefit observed in superior studies limits the applicability of Dr. Elliott and colleagues’ results to immediate clinical practice. “These findings could be used, however, to target future high-quality studies toward the medications that they found might be useful,” he said in an interview.
For the present, other recent research augurs well for strategically reducing unnecessary care. In a paper published online in JAMA Pediatrics, Libby Haskell, MN, of the ED at Starship Children’s Hospital in Auckland, New Zealand, and associates reported on a cluster-randomized, controlled trial of targeted interventions.
Conducted in 2017 at 26 hospitals and with 3,727 babies in New Zealand and Australia, the study addressed drivers of non–evidence-based approaches with behavior-modifying approaches such as on-site clinical leads, stakeholder meetings, a train-the-trainer workshop, education, and audit and feedback.
The authors reported a 14.1% difference in rates of compliance during the first 24 hours of hospitalization favoring the intervention group for all five bronchiolitis guideline recommendations. The greatest change was seen in albuterol and chest radiography use, with other improvements in ED visits, inpatient consultations, and throughout hospitalization.
“These results provide clinicians and hospitals with clear implementation strategies to address unnecessary treatment of infants with bronchiolitis,” Dr. Haskell’s group wrote. Dr. Lipshaw agreed that multifaceted deimplementation packages including clinician and family education, audit and feedback, and clinical decision support have been successful. “Haskell et al. demonstrated that it is possible to successfully deimplement non–evidence-based practices for bronchiolitis with targeted inventions,” he said. “It would be wonderful to see their success replicated in the U.S.”
Why the slow adoption of guidelines?
The American Academy of Pediatrics issued bronchiolitis guidelines for babies to 23 months in 2014 and updated them in 2018. Why, then, has care in some centers been seemingly all over the map and counter to guidelines? “Both parents and clinicians are acting in what they believe to be the best interests of the child, and in the absence of high-value interventions, can feel the need to do something, even if that something is not supported by evidence,” Dr. Lipshaw said.
Furthermore, with children in obvious distress, breathing fast and with difficulty, and sometimes unable to eat or drink, “we feel like we should have some way to make them feel better quicker. Unfortunately, none of the medications we have tried seem to be useful for most children, and we are left with supportive care measures such as suctioning their noses, giving them oxygen if their oxygen is low, and giving them fluids if they are dehydrated.”
Other physicians agree that taking a less-is-more approach can be challenging and even counterintuitive. “To families, seeing their child’s doctor ‘doing less’ can be frustrating,” admitted Diana S. Lee, MD, assistant professor of pediatrics at Icahn School of Medicine at Mount Sinai, New York.
Beyond that, altering practice behavior will need more than guidelines, Dr. Lee said in an interview. “Haskell et al. showed targeted behavior-change interventions improved compliance with bronchiolitis guidelines, but such change requires motivation and resources, and the sustainability of this effect over time remains to be seen.”
At Dr. Lipshaw’s institution, treatment depends on the attending physician, “but we have an emergency department care algorithm, which does not recommend any inhaled medications or steroids in accordance with the 2014 AAP guidelines,” he said.
Similarly at Mount Sinai, practitioners strive to follow the AAP guidelines, although their implementation has not been immediate, Dr. Lee said. “This is a situation where we must make the effort to choose not to do more, given current evidence.”
But Michelle Dunn, MD, an attending physician in the division of general pediatrics at the Children’s Hospital of Philadelphia, said the American practice norm already tends more to the observance than the breach of the guidelines, noting that since 2014 quality improvement efforts have been made throughout the country. “At our institution, we have effectively reduced the use of albuterol in patients with bronchiolitis and we use evidence-based therapy as much as possible, which in the case of bronchiolitis generally involves supportive management alone,” she said in an interview.
Still, Dr. Dunn added, many patients receive unnecessary diagnostic testing and ineffective therapies, with some providers facing psychological barriers to doing less. “However, with more and more evidence to support this, hopefully, physicians will become more comfortable with this.”
To that end, Dr. Lipshaw’s editorial urges physicians to “curb the rampant use of therapies repeatedly revealed to be ineffective,” citing team engagement, clear practice guidelines, and information technology as key factors in deimplementation. In the meantime, his mantra remains: “Don’t just do something, stand there.”
The study by Dr. Elliot and colleagues was supported by the Canadian Institutes of Health Research Knowledge Synthesis grant program. One coauthor is supported by a University of Ottawa Tier I Research Chair in Pediatric Emergency Medicine. Another is supported by a Tier 1 Canada Research Chair in Knowledge Synthesis and Translation and the Stollery Science Laboratory. Dr. Lipshaw and Dr. Florin disclosed no financial relationships relevant to their commentary. Dr. Haskell and colleagues were supported, variously, by the National Health and Medical Research Council of New Zealand, the Center of Research Excellence for Pediatric Emergency Medicine, the Victorian Government’s Operational Infrastructure Support Program, Cure Kids New Zealand, the Royal Children’s Hospital Foundation, and the Starship Foundation. Dr. Lee and Dr. Dunn had no competing interests to disclose with regard to their comments.
A common cause of infant morbidity and hospitalization in developed countries, infant viral bronchiolitis, has long been bedeviled by treatment uncertainty beyond supportive care.
Rationales for most pharmacologic treatments continue to be debated, and clinical practice guidelines generally advise respiratory and hydration support, discouraging the use of chest radiography, albuterol, glucocorticoids, antibiotics, and epinephrine.
Despite evidence that the latter interventions are ineffective, they are still too often applied, according to two recent studies, one in Pediatrics, the other in JAMA Pediatrics.
“The pull of the therapeutic vacuum surrounding this disease has been noted in the pages of this journal for at least 50 years, with Wright and Beem writing in 1965 that ‘energies should not be frittered away by the annoyance of unnecessary or futile medications and procedures’ for the child with bronchiolitis,” said emergency physicians Matthew J. Lipshaw, MD, MS, of the Cincinnati Children’s Hospital Medical Center, and Todd A. Florin, MD, MSCE, of Ann and Robert H. Lurie Children’s Hospital of Chicago.
These remarks came in their editorial in Pediatrics wryly titled: “Don’t Just Do Something, Stand There” and published online to accompany a recent study of three network meta-analyses.
Led by Sarah A. Elliott, PhD, of the Alberta Research Centre for Health Evidence at the University of Alberta in Edmonton, this analysis amalgamated 150 randomized, controlled trials comparing a placebo or active comparator with any bronchodilator, glucocorticoid steroid, hypertonic saline solution, antibiotic, helium-oxygen therapy, or high-flow oxygen therapy. It then looked at the following outcomes in children aged 2 years and younger: hospital admission rate on day 1, hospital admission rate within 7 days, and total hospital length of stay.
Few treatments seemed more effective than nebulized placebo (0.9% saline) for short-term outcomes, the authors found. While nebulized epinephrine and nebulized hypertonic saline plus salbutamol appeared to reduce admission rates during the index ED presentation, and hypertonic saline, alone or in combination with epinephrine, seemed to reduce hospital stays, such treatment had no effect on admissions within 7 days of initial presentation. Furthermore, most benefits disappeared in higher-quality studies.
Concluding, albeit with weak evidence and low confidence, that some benefit might accrue with hypertonic saline with salbutamol to reduce admission rates on initial presentation to the ED, the authors called for well-designed studies on treatments in inpatients and outpatients.
According to Dr. Lipshaw, assistant professor of clinical pediatrics, the lack of benefit observed in superior studies limits the applicability of Dr. Elliott and colleagues’ results to immediate clinical practice. “These findings could be used, however, to target future high-quality studies toward the medications that they found might be useful,” he said in an interview.
For the present, other recent research augurs well for strategically reducing unnecessary care. In a paper published online in JAMA Pediatrics, Libby Haskell, MN, of the ED at Starship Children’s Hospital in Auckland, New Zealand, and associates reported on a cluster-randomized, controlled trial of targeted interventions.
Conducted in 2017 at 26 hospitals and with 3,727 babies in New Zealand and Australia, the study addressed drivers of non–evidence-based approaches with behavior-modifying approaches such as on-site clinical leads, stakeholder meetings, a train-the-trainer workshop, education, and audit and feedback.
The authors reported a 14.1% difference in rates of compliance during the first 24 hours of hospitalization favoring the intervention group for all five bronchiolitis guideline recommendations. The greatest change was seen in albuterol and chest radiography use, with other improvements in ED visits, inpatient consultations, and throughout hospitalization.
“These results provide clinicians and hospitals with clear implementation strategies to address unnecessary treatment of infants with bronchiolitis,” Dr. Haskell’s group wrote. Dr. Lipshaw agreed that multifaceted deimplementation packages including clinician and family education, audit and feedback, and clinical decision support have been successful. “Haskell et al. demonstrated that it is possible to successfully deimplement non–evidence-based practices for bronchiolitis with targeted inventions,” he said. “It would be wonderful to see their success replicated in the U.S.”
Why the slow adoption of guidelines?
The American Academy of Pediatrics issued bronchiolitis guidelines for babies to 23 months in 2014 and updated them in 2018. Why, then, has care in some centers been seemingly all over the map and counter to guidelines? “Both parents and clinicians are acting in what they believe to be the best interests of the child, and in the absence of high-value interventions, can feel the need to do something, even if that something is not supported by evidence,” Dr. Lipshaw said.
Furthermore, with children in obvious distress, breathing fast and with difficulty, and sometimes unable to eat or drink, “we feel like we should have some way to make them feel better quicker. Unfortunately, none of the medications we have tried seem to be useful for most children, and we are left with supportive care measures such as suctioning their noses, giving them oxygen if their oxygen is low, and giving them fluids if they are dehydrated.”
Other physicians agree that taking a less-is-more approach can be challenging and even counterintuitive. “To families, seeing their child’s doctor ‘doing less’ can be frustrating,” admitted Diana S. Lee, MD, assistant professor of pediatrics at Icahn School of Medicine at Mount Sinai, New York.
Beyond that, altering practice behavior will need more than guidelines, Dr. Lee said in an interview. “Haskell et al. showed targeted behavior-change interventions improved compliance with bronchiolitis guidelines, but such change requires motivation and resources, and the sustainability of this effect over time remains to be seen.”
At Dr. Lipshaw’s institution, treatment depends on the attending physician, “but we have an emergency department care algorithm, which does not recommend any inhaled medications or steroids in accordance with the 2014 AAP guidelines,” he said.
Similarly at Mount Sinai, practitioners strive to follow the AAP guidelines, although their implementation has not been immediate, Dr. Lee said. “This is a situation where we must make the effort to choose not to do more, given current evidence.”
But Michelle Dunn, MD, an attending physician in the division of general pediatrics at the Children’s Hospital of Philadelphia, said the American practice norm already tends more to the observance than the breach of the guidelines, noting that since 2014 quality improvement efforts have been made throughout the country. “At our institution, we have effectively reduced the use of albuterol in patients with bronchiolitis and we use evidence-based therapy as much as possible, which in the case of bronchiolitis generally involves supportive management alone,” she said in an interview.
Still, Dr. Dunn added, many patients receive unnecessary diagnostic testing and ineffective therapies, with some providers facing psychological barriers to doing less. “However, with more and more evidence to support this, hopefully, physicians will become more comfortable with this.”
To that end, Dr. Lipshaw’s editorial urges physicians to “curb the rampant use of therapies repeatedly revealed to be ineffective,” citing team engagement, clear practice guidelines, and information technology as key factors in deimplementation. In the meantime, his mantra remains: “Don’t just do something, stand there.”
The study by Dr. Elliot and colleagues was supported by the Canadian Institutes of Health Research Knowledge Synthesis grant program. One coauthor is supported by a University of Ottawa Tier I Research Chair in Pediatric Emergency Medicine. Another is supported by a Tier 1 Canada Research Chair in Knowledge Synthesis and Translation and the Stollery Science Laboratory. Dr. Lipshaw and Dr. Florin disclosed no financial relationships relevant to their commentary. Dr. Haskell and colleagues were supported, variously, by the National Health and Medical Research Council of New Zealand, the Center of Research Excellence for Pediatric Emergency Medicine, the Victorian Government’s Operational Infrastructure Support Program, Cure Kids New Zealand, the Royal Children’s Hospital Foundation, and the Starship Foundation. Dr. Lee and Dr. Dunn had no competing interests to disclose with regard to their comments.