User login
Pediatric fractures shift during pandemic
Pediatric fractures dropped by 2.5-fold during the early months of the COVID-19 pandemic, but more breaks happened at home and on bicycles, and younger kids were more affected, new research indicates.
The study of 1,745 patients also found that those with distal radius torus fractures were more likely to receive a Velcro splint during the pandemic. Experts said this key trend points toward widespread shifts to streamline treatment, which should persist after the pandemic.
“We expected to see a drop in fracture volume, but what was a bit unexpected was the proportional rise in at-home injuries, which we weren’t immediately aware of,” said senior author Apurva Shah, MD, MBA, of Children’s Hospital of Philadelphia (CHOP) and the University of Pennsylvania in Philadelphia.
“As time went on, it became more apparent that trampoline and bicycle injuries were on the rise, but at the beginning of the pandemic, we didn’t intuitively expect that,” he added.
“Whenever there’s a major shift in how the world is working, we want to understand how that impacts child safety,” Dr. Shah said in an interview. “The message to get out to parents is that it’s obviously difficult to supervise kids while working from home” during the pandemic “and that supervision obviously is not always working as well as intended.”
Joshua T. Bram, a medical student, presented the study at the virtual American Academy of Pediatrics (AAP) 2020 National Conference.
Dr. Bram, Dr. Shah, and colleagues compared patients with acute fractures who presented at CHOP between March and April 2020 with those who presented during the same months in 2018 and 2019.
Overall, the number of patients with pediatric fractures who presented to CHOP fell to an average of just under 10 per day, compared with more than 22 per day in prior years (P < .001). In addition, the age of the patients fell from an average of 9.4 years to 7.5 years (P < .001), with fewer adolescents affected in 2020.
“I think when you cancel a 14-year-old’s baseball season” because of the pandemic, “unfortunately, that lost outdoor time might be substituted with time on a screen,” he explained. “But canceling a 6-year-old’s soccer season might mean substituting that with more time outside on bikes or on a trampoline.”
As noted, because of the pandemic, a higher proportion of pediatric fractures occurred at home (57.8% vs. 32.5%; P < .001) or on bicycles (18.3% vs. 8.2%; P < .001), but there were fewer organized sports–related (7.2% vs. 26.0%; P < .001) or playground-related injuries (5.2% vs. 9.0%; P < .001).
In the study period this year, the researchers saw no increase in the amount of time between injury and presentation. However, data suggest that, in more recent months, “kids are presenting with fractures late, with sometimes great consequences,” Dr. Shah said.
“What has changed is that a lot of adults have lost their jobs, and as a consequence, a lot of children have lost their access to private insurance,” he said. “But fracture is really a major injury, and this is a reminder for pediatricians and primary care physicians to recognize that families are going through these changes and that delays in care can really be detrimental to children.”
Velcro splints more common
A potential upside to shifts seen during the pandemic, Dr. Shah said, is the finding that distal radius torus fractures were more likely to be treated with a Velcro splint than in previous years (44.2% vs. 25.9%; P = .010).
“This is hitting on something important – that sometimes it’s crisis that forces us as physicians to evolve,” he said. “This is something I think is here to stay.
“Although research had already been there suggesting a close equivalent between splints and casting, culturally, a lot of surgeons hadn’t made that shift when historically the gold standard had been casting,” Dr. Shah added. “But with the pandemic, the shift to minimize contact with the health care system to keep families safe in their COVID bubble helped [usage of] splints take off.
“I suspect – and we’ll only know when we’re on the other side of this – when physicians see good results in splints in their own patients, they’re going to adopt those strategies more permanently,” he said.
Benjamin Shore, MD, MPH, of Boston Children’s Hospital, agreed with Dr. Shah’s prediction that fracture care will be more streamlined after the pandemic. Dr. Shore, who wasn’t involved in the study, said not only are more orthopedic providers treating patients with Velcro splints and bivalve casts, but they are also monitoring patients via telehealth.
“All of these are great examples of innovation, and one of the unique parts of the pandemic is it created a lot of rapid change across healthcare because it caused us to scrutinize the ways we practice and make a change,” Dr. Shore said in an interview.
“It wasn’t a very fancy study, but it’s very important in terms of demonstrating a change in practice,” Dr. Shore said. “The research here basically validated what many of us are seeing and hopefully will help us in future pandemics – which hopefully won’t happen – to tell families what to be proactive about.”
Dr. Shah and Dr. Shore agreed that, because fewer fractures are occurring in kids during the pandemic, there is an opportunity to redeploy orthopedic providers to other clinical areas on the basis of volume and need.
Dr. Shah and Dr. Shore have disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Pediatric fractures dropped by 2.5-fold during the early months of the COVID-19 pandemic, but more breaks happened at home and on bicycles, and younger kids were more affected, new research indicates.
The study of 1,745 patients also found that those with distal radius torus fractures were more likely to receive a Velcro splint during the pandemic. Experts said this key trend points toward widespread shifts to streamline treatment, which should persist after the pandemic.
“We expected to see a drop in fracture volume, but what was a bit unexpected was the proportional rise in at-home injuries, which we weren’t immediately aware of,” said senior author Apurva Shah, MD, MBA, of Children’s Hospital of Philadelphia (CHOP) and the University of Pennsylvania in Philadelphia.
“As time went on, it became more apparent that trampoline and bicycle injuries were on the rise, but at the beginning of the pandemic, we didn’t intuitively expect that,” he added.
“Whenever there’s a major shift in how the world is working, we want to understand how that impacts child safety,” Dr. Shah said in an interview. “The message to get out to parents is that it’s obviously difficult to supervise kids while working from home” during the pandemic “and that supervision obviously is not always working as well as intended.”
Joshua T. Bram, a medical student, presented the study at the virtual American Academy of Pediatrics (AAP) 2020 National Conference.
Dr. Bram, Dr. Shah, and colleagues compared patients with acute fractures who presented at CHOP between March and April 2020 with those who presented during the same months in 2018 and 2019.
Overall, the number of patients with pediatric fractures who presented to CHOP fell to an average of just under 10 per day, compared with more than 22 per day in prior years (P < .001). In addition, the age of the patients fell from an average of 9.4 years to 7.5 years (P < .001), with fewer adolescents affected in 2020.
“I think when you cancel a 14-year-old’s baseball season” because of the pandemic, “unfortunately, that lost outdoor time might be substituted with time on a screen,” he explained. “But canceling a 6-year-old’s soccer season might mean substituting that with more time outside on bikes or on a trampoline.”
As noted, because of the pandemic, a higher proportion of pediatric fractures occurred at home (57.8% vs. 32.5%; P < .001) or on bicycles (18.3% vs. 8.2%; P < .001), but there were fewer organized sports–related (7.2% vs. 26.0%; P < .001) or playground-related injuries (5.2% vs. 9.0%; P < .001).
In the study period this year, the researchers saw no increase in the amount of time between injury and presentation. However, data suggest that, in more recent months, “kids are presenting with fractures late, with sometimes great consequences,” Dr. Shah said.
“What has changed is that a lot of adults have lost their jobs, and as a consequence, a lot of children have lost their access to private insurance,” he said. “But fracture is really a major injury, and this is a reminder for pediatricians and primary care physicians to recognize that families are going through these changes and that delays in care can really be detrimental to children.”
Velcro splints more common
A potential upside to shifts seen during the pandemic, Dr. Shah said, is the finding that distal radius torus fractures were more likely to be treated with a Velcro splint than in previous years (44.2% vs. 25.9%; P = .010).
“This is hitting on something important – that sometimes it’s crisis that forces us as physicians to evolve,” he said. “This is something I think is here to stay.
“Although research had already been there suggesting a close equivalent between splints and casting, culturally, a lot of surgeons hadn’t made that shift when historically the gold standard had been casting,” Dr. Shah added. “But with the pandemic, the shift to minimize contact with the health care system to keep families safe in their COVID bubble helped [usage of] splints take off.
“I suspect – and we’ll only know when we’re on the other side of this – when physicians see good results in splints in their own patients, they’re going to adopt those strategies more permanently,” he said.
Benjamin Shore, MD, MPH, of Boston Children’s Hospital, agreed with Dr. Shah’s prediction that fracture care will be more streamlined after the pandemic. Dr. Shore, who wasn’t involved in the study, said not only are more orthopedic providers treating patients with Velcro splints and bivalve casts, but they are also monitoring patients via telehealth.
“All of these are great examples of innovation, and one of the unique parts of the pandemic is it created a lot of rapid change across healthcare because it caused us to scrutinize the ways we practice and make a change,” Dr. Shore said in an interview.
“It wasn’t a very fancy study, but it’s very important in terms of demonstrating a change in practice,” Dr. Shore said. “The research here basically validated what many of us are seeing and hopefully will help us in future pandemics – which hopefully won’t happen – to tell families what to be proactive about.”
Dr. Shah and Dr. Shore agreed that, because fewer fractures are occurring in kids during the pandemic, there is an opportunity to redeploy orthopedic providers to other clinical areas on the basis of volume and need.
Dr. Shah and Dr. Shore have disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Pediatric fractures dropped by 2.5-fold during the early months of the COVID-19 pandemic, but more breaks happened at home and on bicycles, and younger kids were more affected, new research indicates.
The study of 1,745 patients also found that those with distal radius torus fractures were more likely to receive a Velcro splint during the pandemic. Experts said this key trend points toward widespread shifts to streamline treatment, which should persist after the pandemic.
“We expected to see a drop in fracture volume, but what was a bit unexpected was the proportional rise in at-home injuries, which we weren’t immediately aware of,” said senior author Apurva Shah, MD, MBA, of Children’s Hospital of Philadelphia (CHOP) and the University of Pennsylvania in Philadelphia.
“As time went on, it became more apparent that trampoline and bicycle injuries were on the rise, but at the beginning of the pandemic, we didn’t intuitively expect that,” he added.
“Whenever there’s a major shift in how the world is working, we want to understand how that impacts child safety,” Dr. Shah said in an interview. “The message to get out to parents is that it’s obviously difficult to supervise kids while working from home” during the pandemic “and that supervision obviously is not always working as well as intended.”
Joshua T. Bram, a medical student, presented the study at the virtual American Academy of Pediatrics (AAP) 2020 National Conference.
Dr. Bram, Dr. Shah, and colleagues compared patients with acute fractures who presented at CHOP between March and April 2020 with those who presented during the same months in 2018 and 2019.
Overall, the number of patients with pediatric fractures who presented to CHOP fell to an average of just under 10 per day, compared with more than 22 per day in prior years (P < .001). In addition, the age of the patients fell from an average of 9.4 years to 7.5 years (P < .001), with fewer adolescents affected in 2020.
“I think when you cancel a 14-year-old’s baseball season” because of the pandemic, “unfortunately, that lost outdoor time might be substituted with time on a screen,” he explained. “But canceling a 6-year-old’s soccer season might mean substituting that with more time outside on bikes or on a trampoline.”
As noted, because of the pandemic, a higher proportion of pediatric fractures occurred at home (57.8% vs. 32.5%; P < .001) or on bicycles (18.3% vs. 8.2%; P < .001), but there were fewer organized sports–related (7.2% vs. 26.0%; P < .001) or playground-related injuries (5.2% vs. 9.0%; P < .001).
In the study period this year, the researchers saw no increase in the amount of time between injury and presentation. However, data suggest that, in more recent months, “kids are presenting with fractures late, with sometimes great consequences,” Dr. Shah said.
“What has changed is that a lot of adults have lost their jobs, and as a consequence, a lot of children have lost their access to private insurance,” he said. “But fracture is really a major injury, and this is a reminder for pediatricians and primary care physicians to recognize that families are going through these changes and that delays in care can really be detrimental to children.”
Velcro splints more common
A potential upside to shifts seen during the pandemic, Dr. Shah said, is the finding that distal radius torus fractures were more likely to be treated with a Velcro splint than in previous years (44.2% vs. 25.9%; P = .010).
“This is hitting on something important – that sometimes it’s crisis that forces us as physicians to evolve,” he said. “This is something I think is here to stay.
“Although research had already been there suggesting a close equivalent between splints and casting, culturally, a lot of surgeons hadn’t made that shift when historically the gold standard had been casting,” Dr. Shah added. “But with the pandemic, the shift to minimize contact with the health care system to keep families safe in their COVID bubble helped [usage of] splints take off.
“I suspect – and we’ll only know when we’re on the other side of this – when physicians see good results in splints in their own patients, they’re going to adopt those strategies more permanently,” he said.
Benjamin Shore, MD, MPH, of Boston Children’s Hospital, agreed with Dr. Shah’s prediction that fracture care will be more streamlined after the pandemic. Dr. Shore, who wasn’t involved in the study, said not only are more orthopedic providers treating patients with Velcro splints and bivalve casts, but they are also monitoring patients via telehealth.
“All of these are great examples of innovation, and one of the unique parts of the pandemic is it created a lot of rapid change across healthcare because it caused us to scrutinize the ways we practice and make a change,” Dr. Shore said in an interview.
“It wasn’t a very fancy study, but it’s very important in terms of demonstrating a change in practice,” Dr. Shore said. “The research here basically validated what many of us are seeing and hopefully will help us in future pandemics – which hopefully won’t happen – to tell families what to be proactive about.”
Dr. Shah and Dr. Shore agreed that, because fewer fractures are occurring in kids during the pandemic, there is an opportunity to redeploy orthopedic providers to other clinical areas on the basis of volume and need.
Dr. Shah and Dr. Shore have disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
One measure of child COVID-19 may be trending downward
After increasing for several weeks, the proportion of new COVID-19 cases occurring in children has dropped for the second week in a row, according to data in a new report from the American Academy of Pediatrics and the Children’s Hospital Association.
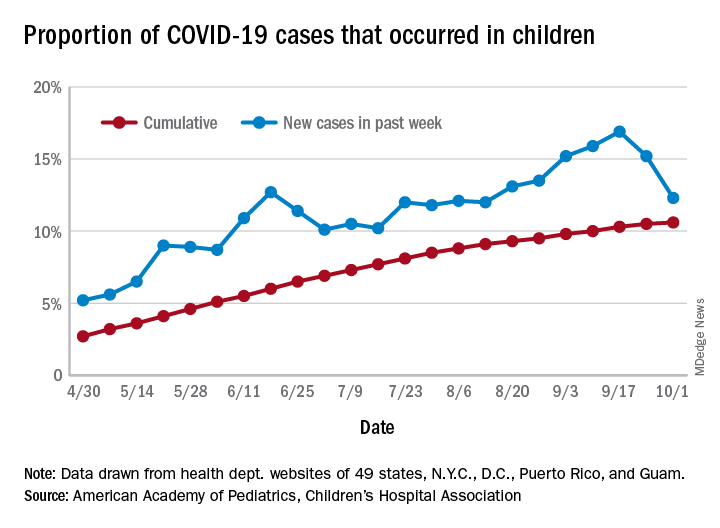
COVID-19 cases in children accounted for 12.3% of all new cases in the United States for the week ending Oct. 1, down from 15.2% the previous week. That measure had reached its highest point, 16.9%, just one week earlier (Sept. 17), the AAP and the CHA said in their weekly COVID-19 report.
based on data from the health departments of 49 states (New York does not provide ages on its website), as well as the District of Columbia, New York City, Puerto Rico, and Guam.
The child COVID-19 rate for the United States was 874 per 100,000 children as of Oct. 1, and that figure has doubled since the end of July. At the state level, the highest rates can be found in Tennessee (2,031.4 per 100,000), North Dakota (2,029.6), and South Carolina (2,002.6), with the lowest rates in Vermont (168.9), Maine (229.1), and New Hampshire (268.3), the AAP/CHA report shows.
The children of Wyoming make up the largest share, 22.4%, of any state’s COVID-19 cases, followed by North Dakota and Tennessee, both at 18.3%. New Jersey is lower than any other state at 3.9%, although New York City is a slightly lower 3.6%, the AAP and CHA said.
“The data are limited because the states differ in how they report the data, and it is unknown how many children have been infected but not tested. It is unclear how much of the increase in child cases is due to increased testing capacity,” the AAP said in an earlier statement.
After increasing for several weeks, the proportion of new COVID-19 cases occurring in children has dropped for the second week in a row, according to data in a new report from the American Academy of Pediatrics and the Children’s Hospital Association.

COVID-19 cases in children accounted for 12.3% of all new cases in the United States for the week ending Oct. 1, down from 15.2% the previous week. That measure had reached its highest point, 16.9%, just one week earlier (Sept. 17), the AAP and the CHA said in their weekly COVID-19 report.
based on data from the health departments of 49 states (New York does not provide ages on its website), as well as the District of Columbia, New York City, Puerto Rico, and Guam.
The child COVID-19 rate for the United States was 874 per 100,000 children as of Oct. 1, and that figure has doubled since the end of July. At the state level, the highest rates can be found in Tennessee (2,031.4 per 100,000), North Dakota (2,029.6), and South Carolina (2,002.6), with the lowest rates in Vermont (168.9), Maine (229.1), and New Hampshire (268.3), the AAP/CHA report shows.
The children of Wyoming make up the largest share, 22.4%, of any state’s COVID-19 cases, followed by North Dakota and Tennessee, both at 18.3%. New Jersey is lower than any other state at 3.9%, although New York City is a slightly lower 3.6%, the AAP and CHA said.
“The data are limited because the states differ in how they report the data, and it is unknown how many children have been infected but not tested. It is unclear how much of the increase in child cases is due to increased testing capacity,” the AAP said in an earlier statement.
After increasing for several weeks, the proportion of new COVID-19 cases occurring in children has dropped for the second week in a row, according to data in a new report from the American Academy of Pediatrics and the Children’s Hospital Association.

COVID-19 cases in children accounted for 12.3% of all new cases in the United States for the week ending Oct. 1, down from 15.2% the previous week. That measure had reached its highest point, 16.9%, just one week earlier (Sept. 17), the AAP and the CHA said in their weekly COVID-19 report.
based on data from the health departments of 49 states (New York does not provide ages on its website), as well as the District of Columbia, New York City, Puerto Rico, and Guam.
The child COVID-19 rate for the United States was 874 per 100,000 children as of Oct. 1, and that figure has doubled since the end of July. At the state level, the highest rates can be found in Tennessee (2,031.4 per 100,000), North Dakota (2,029.6), and South Carolina (2,002.6), with the lowest rates in Vermont (168.9), Maine (229.1), and New Hampshire (268.3), the AAP/CHA report shows.
The children of Wyoming make up the largest share, 22.4%, of any state’s COVID-19 cases, followed by North Dakota and Tennessee, both at 18.3%. New Jersey is lower than any other state at 3.9%, although New York City is a slightly lower 3.6%, the AAP and CHA said.
“The data are limited because the states differ in how they report the data, and it is unknown how many children have been infected but not tested. It is unclear how much of the increase in child cases is due to increased testing capacity,” the AAP said in an earlier statement.
CMS gives hospitals 14 weeks to start daily COVID, flu reports
The federal government is giving hospitals 14 weeks to comply with daily reporting requirements for COVID-19.
The Centers for Medicare & Medicaid Services will send letters on October 7 to all 6,200 hospitals that receive reimbursement from the two federal health programs informing them of how well they are doing now, said CMS Administrator Seema Verma on a press call.
Verma would not give an estimate on how many hospitals are currently not compliant. But Deborah Birx, MD, a member of the White House Coronavirus Task Force, said on the call that 86% of hospitals are currently reporting daily.
Federal officials on the call also announced that hospitals would have the option to begin reporting certain data on influenza starting October 19, but that it would become mandatory a few weeks later.
The reporting is important “to really ensure that we’re triangulating all data to understand where this epidemic is, how it’s moving through different populations, and ensuring that we’re meeting the needs of specific hospitals and communities,” Birx said.
The federal government began a new hospital reporting system in April but did not require hospitals to participate until it quietly issued guidance in mid-July informing facilities that they should no longer report to the Centers for Disease Control and Prevention (CDC).
The move perplexed many public health experts and epidemiologists, who expressed concern that asking hospitals to use a new data system during a pandemic could result in delays and lost information. The new HHS data collection site, HHS Protect, is being managed by a private contractor, not the CDC, which also raised alarms.
The final CMS rule issued in August went into effect immediately, without any chance for comment or revision. CMS said at the time that the pandemic was reason enough to skip over the normal bureaucratic process.
Hospitals were not pleased. But Verma claimed that since then CMS had been working with hospital organizations on enforcement.
“We’re going to do everything we can to facilitate reporting, including an enforcement timeline that will provide hospitals ample opportunity to come into compliance,” she said.
Hospitals that do not comply will get a notice every 3 weeks. Three weeks after the second notice, they’ll get weekly notices for a month, and a final termination notice at 14 weeks.
The Federation of American Hospitals (FAH), however, said their members were still not happy. “It is both inappropriate and frankly overkill for CMS to tie compliance with reporting to Medicare conditions of participation,” said FAH President and CEO Chip Kahn in a statement. He called the CMS proposal “sledgehammer enforcement,” and said that the continuing data request might weaken hospitals’ response to the pandemic because it would divert time and money away from patient care.
Rick Pollack, president and CEO of the American Hospital Association called the CMS rule an “overly heavy-handed approach that could jeopardize access to hospital care for all Americans.” He noted in a statement that barring hospitals from Medicare and Medicaid could harm beneficiaries and the effort to provide COVID care.
Pollack also noted that AHA has “observed errors in data processing and confusion about exactly what was being requested at the hospital, state, contractor, and federal level, and has worked diligently with the federal agencies to identify and correct those problems.”
The document that lays out U.S. Department of Health and Human Services (HHS) Protect reporting requirements were updated again on October 6 to add influenza data. The hospitals must report on total patients with laboratory-confirmed flu; previous day’s flu admissions; total ICU patients with lab-confirmed flu; total inpatients with either flu or COVID-19; and the previous day’s deaths for flu and COVID.
CDC Director Robert Redfield, MD, said on the press call that the new data will give the agency crucial hospital-level information and perhaps better estimates of the flu burden. Flu trends have been tracked using the CDC’s Influenza Hospitalization Surveillance Network (FluSurv-NET), which will not be replaced, Redfield said. But that network only tracks hospitalizations in 14 states and does not provide information in “nearly real-time,” he said.
Having the new data “will give us a true situational awareness of severe respiratory illness, provide local hospitalization trends, and help direct resources such as antiretrovirals to address potential increased impact of flu and COVID cocirculation,” Redfield said.
This article first appeared on Medscape.com.
The federal government is giving hospitals 14 weeks to comply with daily reporting requirements for COVID-19.
The Centers for Medicare & Medicaid Services will send letters on October 7 to all 6,200 hospitals that receive reimbursement from the two federal health programs informing them of how well they are doing now, said CMS Administrator Seema Verma on a press call.
Verma would not give an estimate on how many hospitals are currently not compliant. But Deborah Birx, MD, a member of the White House Coronavirus Task Force, said on the call that 86% of hospitals are currently reporting daily.
Federal officials on the call also announced that hospitals would have the option to begin reporting certain data on influenza starting October 19, but that it would become mandatory a few weeks later.
The reporting is important “to really ensure that we’re triangulating all data to understand where this epidemic is, how it’s moving through different populations, and ensuring that we’re meeting the needs of specific hospitals and communities,” Birx said.
The federal government began a new hospital reporting system in April but did not require hospitals to participate until it quietly issued guidance in mid-July informing facilities that they should no longer report to the Centers for Disease Control and Prevention (CDC).
The move perplexed many public health experts and epidemiologists, who expressed concern that asking hospitals to use a new data system during a pandemic could result in delays and lost information. The new HHS data collection site, HHS Protect, is being managed by a private contractor, not the CDC, which also raised alarms.
The final CMS rule issued in August went into effect immediately, without any chance for comment or revision. CMS said at the time that the pandemic was reason enough to skip over the normal bureaucratic process.
Hospitals were not pleased. But Verma claimed that since then CMS had been working with hospital organizations on enforcement.
“We’re going to do everything we can to facilitate reporting, including an enforcement timeline that will provide hospitals ample opportunity to come into compliance,” she said.
Hospitals that do not comply will get a notice every 3 weeks. Three weeks after the second notice, they’ll get weekly notices for a month, and a final termination notice at 14 weeks.
The Federation of American Hospitals (FAH), however, said their members were still not happy. “It is both inappropriate and frankly overkill for CMS to tie compliance with reporting to Medicare conditions of participation,” said FAH President and CEO Chip Kahn in a statement. He called the CMS proposal “sledgehammer enforcement,” and said that the continuing data request might weaken hospitals’ response to the pandemic because it would divert time and money away from patient care.
Rick Pollack, president and CEO of the American Hospital Association called the CMS rule an “overly heavy-handed approach that could jeopardize access to hospital care for all Americans.” He noted in a statement that barring hospitals from Medicare and Medicaid could harm beneficiaries and the effort to provide COVID care.
Pollack also noted that AHA has “observed errors in data processing and confusion about exactly what was being requested at the hospital, state, contractor, and federal level, and has worked diligently with the federal agencies to identify and correct those problems.”
The document that lays out U.S. Department of Health and Human Services (HHS) Protect reporting requirements were updated again on October 6 to add influenza data. The hospitals must report on total patients with laboratory-confirmed flu; previous day’s flu admissions; total ICU patients with lab-confirmed flu; total inpatients with either flu or COVID-19; and the previous day’s deaths for flu and COVID.
CDC Director Robert Redfield, MD, said on the press call that the new data will give the agency crucial hospital-level information and perhaps better estimates of the flu burden. Flu trends have been tracked using the CDC’s Influenza Hospitalization Surveillance Network (FluSurv-NET), which will not be replaced, Redfield said. But that network only tracks hospitalizations in 14 states and does not provide information in “nearly real-time,” he said.
Having the new data “will give us a true situational awareness of severe respiratory illness, provide local hospitalization trends, and help direct resources such as antiretrovirals to address potential increased impact of flu and COVID cocirculation,” Redfield said.
This article first appeared on Medscape.com.
The federal government is giving hospitals 14 weeks to comply with daily reporting requirements for COVID-19.
The Centers for Medicare & Medicaid Services will send letters on October 7 to all 6,200 hospitals that receive reimbursement from the two federal health programs informing them of how well they are doing now, said CMS Administrator Seema Verma on a press call.
Verma would not give an estimate on how many hospitals are currently not compliant. But Deborah Birx, MD, a member of the White House Coronavirus Task Force, said on the call that 86% of hospitals are currently reporting daily.
Federal officials on the call also announced that hospitals would have the option to begin reporting certain data on influenza starting October 19, but that it would become mandatory a few weeks later.
The reporting is important “to really ensure that we’re triangulating all data to understand where this epidemic is, how it’s moving through different populations, and ensuring that we’re meeting the needs of specific hospitals and communities,” Birx said.
The federal government began a new hospital reporting system in April but did not require hospitals to participate until it quietly issued guidance in mid-July informing facilities that they should no longer report to the Centers for Disease Control and Prevention (CDC).
The move perplexed many public health experts and epidemiologists, who expressed concern that asking hospitals to use a new data system during a pandemic could result in delays and lost information. The new HHS data collection site, HHS Protect, is being managed by a private contractor, not the CDC, which also raised alarms.
The final CMS rule issued in August went into effect immediately, without any chance for comment or revision. CMS said at the time that the pandemic was reason enough to skip over the normal bureaucratic process.
Hospitals were not pleased. But Verma claimed that since then CMS had been working with hospital organizations on enforcement.
“We’re going to do everything we can to facilitate reporting, including an enforcement timeline that will provide hospitals ample opportunity to come into compliance,” she said.
Hospitals that do not comply will get a notice every 3 weeks. Three weeks after the second notice, they’ll get weekly notices for a month, and a final termination notice at 14 weeks.
The Federation of American Hospitals (FAH), however, said their members were still not happy. “It is both inappropriate and frankly overkill for CMS to tie compliance with reporting to Medicare conditions of participation,” said FAH President and CEO Chip Kahn in a statement. He called the CMS proposal “sledgehammer enforcement,” and said that the continuing data request might weaken hospitals’ response to the pandemic because it would divert time and money away from patient care.
Rick Pollack, president and CEO of the American Hospital Association called the CMS rule an “overly heavy-handed approach that could jeopardize access to hospital care for all Americans.” He noted in a statement that barring hospitals from Medicare and Medicaid could harm beneficiaries and the effort to provide COVID care.
Pollack also noted that AHA has “observed errors in data processing and confusion about exactly what was being requested at the hospital, state, contractor, and federal level, and has worked diligently with the federal agencies to identify and correct those problems.”
The document that lays out U.S. Department of Health and Human Services (HHS) Protect reporting requirements were updated again on October 6 to add influenza data. The hospitals must report on total patients with laboratory-confirmed flu; previous day’s flu admissions; total ICU patients with lab-confirmed flu; total inpatients with either flu or COVID-19; and the previous day’s deaths for flu and COVID.
CDC Director Robert Redfield, MD, said on the press call that the new data will give the agency crucial hospital-level information and perhaps better estimates of the flu burden. Flu trends have been tracked using the CDC’s Influenza Hospitalization Surveillance Network (FluSurv-NET), which will not be replaced, Redfield said. But that network only tracks hospitalizations in 14 states and does not provide information in “nearly real-time,” he said.
Having the new data “will give us a true situational awareness of severe respiratory illness, provide local hospitalization trends, and help direct resources such as antiretrovirals to address potential increased impact of flu and COVID cocirculation,” Redfield said.
This article first appeared on Medscape.com.
Optimal sedation strategies for COVID-19 ICU patients: A work in progress
According to the best available evidence, analagosedation remains the focus for managing COVID-19 ICU patients, according to Steven B. Greenberg, MD, FCCP, FCCM.
“The choice of sedation and analgesia is important,” Dr. Greenberg, vice chair of education in the department of anesthesiology at Evanston Hospital, part of NorthShore University Health System, Chicago, said at a Society for Critical Care virtual meeting: COVID-19: What’s Next. “We know that the right choice of these two components may increase liberation from ventilators, earlier ICU discharge, and return to normal brain function and independent functional status.”
Analgesia first
Prior to the current pandemic, the approach to sedation of patients in the ICU was based on the PADIS Guidelines of 2018, which call for an assessment-driven, protocol-based stepwise approach to pain and sedation management in critically ill adults (Crit Care Med. 2018;46:e825-73). “” Dr. Greenberg said. “We know that pain management should be a priority of sedation, because pain may increase the risk of delirium, anxiety, and endocrine suppression, and may increase the risk of release of endogenous catecholamines, ischemia, and hypermetabolic states.”
Fentanyl appears to be the most common opioid analgesic used for patients in the ICU, “but fentanyl is a very lipophilic drug and has a long context-sensitive half-life,” he said. “There are components to fentanyl that allow it to become a very long-acting drug upon days and days of infusion. Another opioid used is remifentanil, which is typically short-acting because it is broken down in the blood by esterases, but may cause rigidity at higher doses. Dilaudid seems to be the least affected by organ dysfunction. In our very critically ill, prolonged mechanically ventilated COVID-19 patients, we’ve been using methadone for its NMDA [N-methyl-D-aspartate] antagonistic effect and its opioid-sparing effects.”
As for nonopioid analgesics, Dr. Greenberg said that clinicians have shied away from using NSAIDs because of their side effects. “Tramadol indirectly inhibits reuptake of norepinephrine and serotonin, and ketamine is being used a lot more because of its NMDA antagonist effect,” he said. “Lidocaine and gabapentin have also been used.”
In a recent systematic review and meta-analysis, researchers assessed 34 trials that examined adjuvant analgesic use with an opioid in critically ill patients versus an opioid alone (Crit Care Expl. 2020;2:e0157). They found that when using an adjuvant such as acetaminophen, clonidine, dexmedetomidine, gabapentin, ketamine, magnesium, nefopam, NSAIDs, pregabalin, and tramadol, there was a reduction in pain scores as well as a reduction in opioid consumption. “So, clinicians should consider using adjuvant agents to limit opioid exposure and improve pain scores in the critically ill,” Dr. Greenberg said.
ICU delirium: Risk factors, prevention
Delirium in COVID-19 patients treated in the ICU of particular concern. According to a systematic review of 33 studies, 11 risk factors for delirium in the ICU were supported by strong or moderate levels of evidence (Crit Care Med. 2015;43:40-7). These include age, dementia, hypertension, emergency surgery, trauma, APACHE score of II, need for mechanical ventilation, metabolic acidosis, delirium on prior day, coma, and dexmedetomidine use. Risk factors for ICU delirium among COVID-19 patients, however, “are far different,” Dr. Greenberg said. “Why? First and foremost, we are restricting visitation of family,” he said. “That family connection largely can be lost. Second, there are limitations of nonpharmacologic interventions. There is less mobility and physical therapy employed because of the risk of health care workers’ exposure to the virus. There’s also uncertainty about the global pandemic. Anxiety and depression come with that, as well as disruptions to spiritual and religious services.”
Strategies for preventing delirium remain the same as before the pandemic and in accord with recent clinical practice guidelines: Reduce the use of certain drugs such as benzodiazepines and narcotics, reorient the patients, treat dehydration, use hearing aids and eyeglasses in patients who have them, use ear plugs to cancel noise, mobilize patients, maintain sleep/awake cycles, and encourage sedation holidays (Crit Care Med. 2018;46[9]:e825-73).
A recent study from France found that among 58 patients with COVID-19, 65% had positive Confusion Assessment Method (CAM)–ICU findings and 69% had agitation (N Engl J Med 2020;382:2268-70). Most of the patients (86%) received midazolam, 47% received propofol, and all received sufentanil. “In the pre-COVID days, we would use midazolam as a second-line agent for many of these patients,” Dr. Greenberg said. “So, times really have changed.”
The fate of COVID-19 patients following discharge from the ICU remains a concern, continued Dr. Greenberg, clinical professor of anesthesiology at the University of Chicago. A recent journal article by Michelle Biehl, MD, and Denise Sese, MD, noted that post–intensive care syndrome (PICS) or new or worsening impairment in any physical, cognitive, or mental domain is of significant concern among COVID-19 patients following their ICU stay (Cleveland Clin J Med 2020 Aug doi: 10.3949/ccjm.87a.ccc055). The authors stated that COVID-19 patients may face a higher risk of PICS because of restricted family visitation, prolonged mechanical ventilation, exposure to higher amounts of sedatives, and limited physical therapy during hospital stay.
No ideal sedative agent
The 2018 PADIS Guidelines on the use of ICU sedation suggested strong evidence for modifiable risk factors producing delirium in the context of benzodiazepines and blood transfusion. They recommend a light level of sedation and the use of propofol or dexmedetomidine over benzodiazepines. They also recommend routine delirium testing such as using the CAM-ICU or Intensive Care Delirium Screening Checklist (ICDSC) and nonpharmacologic therapies such as reorientation, cognitive stimulation, sleep improvement, and mobilization.
Several sedation-related factors may be related to an increased risk of delirium. “The type, dose, duration, and mode of delivery are very important,” Dr. Greenberg said. “The ideal sedative agent has a rapid, predictable onset; is short-acting; has anxiolytic, amnestic, and analgesic properties; is soluble; has a high therapeutic index; and no toxicity. The ideal sedative is also easy to administrate, contains no active metabolites, has minimal actions with other drugs, is reversible, and is cost effective. The problem is, there really is no ideal sedative agent. There is inadequate knowledge about the drugs [used to treat COVID-19 in the ICU] available to us, the dosage, and importantly, the pharmacokinetics and dynamics of these medications.”
The classic types of sedation being used in the ICU, he said, include the benzodiazepines midazolam, lorazepam, and diazepam, as well as propofol. Alternatives include dexmedetomidine, clonidine, ketamine, and the neuroleptics – haloperidol, quetiapine, olanzapine, ziprasidone, and risperidone. “The advantages of benzos are that they are anxiolytics, amnestics, and they are good sedatives with minimal hemodynamic effects,” Dr. Greenberg said.
Advantages of propofol include its sedative, hypnotic, and anxiolytic properties, he said. It reduces the cerebral metabolic rate and can relieve bronchospasm. “However, small studies have found that its use may be associated with an increased risk of delirium,” he said. “It is a respiratory depressant, and it can cause hypotension and decreased contractility. It has no analgesic properties, and two of the big concerns of its use in COVID-19 are the potential for hypertriglyceridemia and propofol infusion syndrome, particularly at doses of greater than 5 mg/kg per hour for greater than 48 hours. It is being given in high doses because patients are requiring higher doses to maintain ventilator synchrony.”
Choosing the right drug
The keys to success for sedation of ICU patients are choosing the right drug at the right dose for the right duration and the right mode of delivery, and applying them to the right population. However, as noted in a recent study, the pandemic poses unique challenges to clinicians in how they care for critically ill COVID-19 patients who require sedation (Anesth Analg. 2020 Apr 22. doi: 10.1213/ANE.0000000000004887). The use of provisional work areas “has escalated because of the amount of patients we’ve had to care for over the past nine months,” Dr. Greenberg said. “We’ve used alternate providers who are not necessarily familiar with the sedation and analgesic protocols and how to use these specific medications. Drug shortages have been on the rise, so there’s a need to understand alternative agents that can be used.”
COVID-19 patients face the potential risk for an increase in drug-drug interactions and side effects due to the polypharmacy that is often required to provide adequate sedation during mechanical ventilation. He noted that these patients may have “unusually high” analgesia and sedation requirements, particularly when they’re mechanically ventilated. A hypothesis as to why patients with COVID-19 require so much sedation and analgesia is that they often have a high respiratory drive and ventilator dyssynchrony, which requires increased neuromuscular blockade. “They also have an intense inflammatory response, which may be linked to tolerance of specific opioids and other medications,” Dr. Greenberg said. “Many ventilated COVID-19 patients are of younger age and previously in good health, and therefore, have an excellent metabolism. Health care providers are concerned about self-extubation. This prompts bedside providers to administer more sedatives to prevent this unwanted complication. There may also be a reduction of drip modifications by health care workers because of the potential risk of contracting COVID-19 when going into the room multiple times and for long periods of time” (Anesth Analg. 2020;131[1]:e34-e35).
According to a sedation resource on the SCCM website, about 5% of COVID-19 patients require mechanical ventilation. “There has been a massive shortage of the usual drugs that we use,” Dr. Greenberg said. “The demand for sedatives has increased by approximately 91%, while the demand for analgesics has increased by 79%, and neuromuscular blocker demand has increased by 105%.”
A retrospective study of 24 COVID-19 patients who required ventilation in the ICU found that the median daily dose of benzodiazepines was significantly higher, compared with the median daily dose used in the OSCILLATE trial (a median of 270 mg vs. 199 mg, respectively; Anesth Analg. 2020;131[4]e198-e200. doi: 10.1213/ane.0000000000005131). In addition, their median daily dose of opioid was approximately three times higher, compared with patients in the OSCILLATE trial (a median of 775 mg vs. 289 mg). Other agents used included propofol (84%), dexmedetomidine (53%), and ketamine (11%).
“A potential strategy for COVID-19 ICU patient sedation should be analgesia first, as indicated in the 2018 PADIS guidelines,” Dr. Greenberg advised. “We should also apply nonpharmacologic measures to reduce delirium. In nonintubated patients, we should use light to moderate sedation, targeting a RASS of –2 to +1, using hydromorphone or fentanyl boluses for analgesia and midazolam boluses or dexmedetomidine for sedation,.”
For intubated patients, he continued, target a RASS of –3 to –4, or –4 to –5 in those who require neuromuscular blockade. “Use propofol first then intermittent boluses of benzodiazepines,” said Dr. Greenberg, editor-in-chief of the Anesthesia Patient Safety Foundation newsletter. “For heavy sedation, use midazolam and supplement with ketamine and other analgesics and sedatives such as barbiturates, methadone, and even inhalation anesthetics in some cases.”
For analgesia in intubated patients, use fentanyl boluses then infusion. “Patients can easily become tachyphylactic to fentanyl, and it has a long context-sensitive half time,” he said. “Hydromorphone may be least affected by organ dysfunction.”
Dr. Greenberg concluded his presentation by stating that more studies are required “to delineate the best analgesia/sedation strategies and monitoring modalities for COVID-19 ICU patients.”
In commenting on the presentation, Mangala Narasimhan, DO, FCCP, senior vice president and director of critical care services at Northwell Health, said that the recommendations regarding sedation highlight a struggle that ICU providers have been dealing with during the COVID-19 epidemic.
“There have been unique challenges with COVID-19 and intubated patients. We have seen severe ventilator dyssynchrony and prolonged duration of mechanical ventilation. I think we can all agree that these patients have extremely high metabolic rates, have required high levels of sedation, have an increased need for neuromuscular blockade, and have high levels of delirium for extended periods of time. The recommendations provided here are reasonable. Strategies to prevent delirium should be employed, pain management should be prioritized, analgesics can help reduce the need for opioids. Alternatives to sedation are useful in this patient population and are well tolerated. Drug shortages have provided additional challenges to these strategies and have required us to think about the use of alternative agents. The recommendations echo the experience we have had with large numbers of intubated COVID-19 patients.”
Dr. Greenberg disclosed that he receives a stipend from the Anesthesia Patient Safety Foundation for serving as editor-in-chief of the foundation’s newsletter.
According to the best available evidence, analagosedation remains the focus for managing COVID-19 ICU patients, according to Steven B. Greenberg, MD, FCCP, FCCM.
“The choice of sedation and analgesia is important,” Dr. Greenberg, vice chair of education in the department of anesthesiology at Evanston Hospital, part of NorthShore University Health System, Chicago, said at a Society for Critical Care virtual meeting: COVID-19: What’s Next. “We know that the right choice of these two components may increase liberation from ventilators, earlier ICU discharge, and return to normal brain function and independent functional status.”
Analgesia first
Prior to the current pandemic, the approach to sedation of patients in the ICU was based on the PADIS Guidelines of 2018, which call for an assessment-driven, protocol-based stepwise approach to pain and sedation management in critically ill adults (Crit Care Med. 2018;46:e825-73). “” Dr. Greenberg said. “We know that pain management should be a priority of sedation, because pain may increase the risk of delirium, anxiety, and endocrine suppression, and may increase the risk of release of endogenous catecholamines, ischemia, and hypermetabolic states.”
Fentanyl appears to be the most common opioid analgesic used for patients in the ICU, “but fentanyl is a very lipophilic drug and has a long context-sensitive half-life,” he said. “There are components to fentanyl that allow it to become a very long-acting drug upon days and days of infusion. Another opioid used is remifentanil, which is typically short-acting because it is broken down in the blood by esterases, but may cause rigidity at higher doses. Dilaudid seems to be the least affected by organ dysfunction. In our very critically ill, prolonged mechanically ventilated COVID-19 patients, we’ve been using methadone for its NMDA [N-methyl-D-aspartate] antagonistic effect and its opioid-sparing effects.”
As for nonopioid analgesics, Dr. Greenberg said that clinicians have shied away from using NSAIDs because of their side effects. “Tramadol indirectly inhibits reuptake of norepinephrine and serotonin, and ketamine is being used a lot more because of its NMDA antagonist effect,” he said. “Lidocaine and gabapentin have also been used.”
In a recent systematic review and meta-analysis, researchers assessed 34 trials that examined adjuvant analgesic use with an opioid in critically ill patients versus an opioid alone (Crit Care Expl. 2020;2:e0157). They found that when using an adjuvant such as acetaminophen, clonidine, dexmedetomidine, gabapentin, ketamine, magnesium, nefopam, NSAIDs, pregabalin, and tramadol, there was a reduction in pain scores as well as a reduction in opioid consumption. “So, clinicians should consider using adjuvant agents to limit opioid exposure and improve pain scores in the critically ill,” Dr. Greenberg said.
ICU delirium: Risk factors, prevention
Delirium in COVID-19 patients treated in the ICU of particular concern. According to a systematic review of 33 studies, 11 risk factors for delirium in the ICU were supported by strong or moderate levels of evidence (Crit Care Med. 2015;43:40-7). These include age, dementia, hypertension, emergency surgery, trauma, APACHE score of II, need for mechanical ventilation, metabolic acidosis, delirium on prior day, coma, and dexmedetomidine use. Risk factors for ICU delirium among COVID-19 patients, however, “are far different,” Dr. Greenberg said. “Why? First and foremost, we are restricting visitation of family,” he said. “That family connection largely can be lost. Second, there are limitations of nonpharmacologic interventions. There is less mobility and physical therapy employed because of the risk of health care workers’ exposure to the virus. There’s also uncertainty about the global pandemic. Anxiety and depression come with that, as well as disruptions to spiritual and religious services.”
Strategies for preventing delirium remain the same as before the pandemic and in accord with recent clinical practice guidelines: Reduce the use of certain drugs such as benzodiazepines and narcotics, reorient the patients, treat dehydration, use hearing aids and eyeglasses in patients who have them, use ear plugs to cancel noise, mobilize patients, maintain sleep/awake cycles, and encourage sedation holidays (Crit Care Med. 2018;46[9]:e825-73).
A recent study from France found that among 58 patients with COVID-19, 65% had positive Confusion Assessment Method (CAM)–ICU findings and 69% had agitation (N Engl J Med 2020;382:2268-70). Most of the patients (86%) received midazolam, 47% received propofol, and all received sufentanil. “In the pre-COVID days, we would use midazolam as a second-line agent for many of these patients,” Dr. Greenberg said. “So, times really have changed.”
The fate of COVID-19 patients following discharge from the ICU remains a concern, continued Dr. Greenberg, clinical professor of anesthesiology at the University of Chicago. A recent journal article by Michelle Biehl, MD, and Denise Sese, MD, noted that post–intensive care syndrome (PICS) or new or worsening impairment in any physical, cognitive, or mental domain is of significant concern among COVID-19 patients following their ICU stay (Cleveland Clin J Med 2020 Aug doi: 10.3949/ccjm.87a.ccc055). The authors stated that COVID-19 patients may face a higher risk of PICS because of restricted family visitation, prolonged mechanical ventilation, exposure to higher amounts of sedatives, and limited physical therapy during hospital stay.
No ideal sedative agent
The 2018 PADIS Guidelines on the use of ICU sedation suggested strong evidence for modifiable risk factors producing delirium in the context of benzodiazepines and blood transfusion. They recommend a light level of sedation and the use of propofol or dexmedetomidine over benzodiazepines. They also recommend routine delirium testing such as using the CAM-ICU or Intensive Care Delirium Screening Checklist (ICDSC) and nonpharmacologic therapies such as reorientation, cognitive stimulation, sleep improvement, and mobilization.
Several sedation-related factors may be related to an increased risk of delirium. “The type, dose, duration, and mode of delivery are very important,” Dr. Greenberg said. “The ideal sedative agent has a rapid, predictable onset; is short-acting; has anxiolytic, amnestic, and analgesic properties; is soluble; has a high therapeutic index; and no toxicity. The ideal sedative is also easy to administrate, contains no active metabolites, has minimal actions with other drugs, is reversible, and is cost effective. The problem is, there really is no ideal sedative agent. There is inadequate knowledge about the drugs [used to treat COVID-19 in the ICU] available to us, the dosage, and importantly, the pharmacokinetics and dynamics of these medications.”
The classic types of sedation being used in the ICU, he said, include the benzodiazepines midazolam, lorazepam, and diazepam, as well as propofol. Alternatives include dexmedetomidine, clonidine, ketamine, and the neuroleptics – haloperidol, quetiapine, olanzapine, ziprasidone, and risperidone. “The advantages of benzos are that they are anxiolytics, amnestics, and they are good sedatives with minimal hemodynamic effects,” Dr. Greenberg said.
Advantages of propofol include its sedative, hypnotic, and anxiolytic properties, he said. It reduces the cerebral metabolic rate and can relieve bronchospasm. “However, small studies have found that its use may be associated with an increased risk of delirium,” he said. “It is a respiratory depressant, and it can cause hypotension and decreased contractility. It has no analgesic properties, and two of the big concerns of its use in COVID-19 are the potential for hypertriglyceridemia and propofol infusion syndrome, particularly at doses of greater than 5 mg/kg per hour for greater than 48 hours. It is being given in high doses because patients are requiring higher doses to maintain ventilator synchrony.”
Choosing the right drug
The keys to success for sedation of ICU patients are choosing the right drug at the right dose for the right duration and the right mode of delivery, and applying them to the right population. However, as noted in a recent study, the pandemic poses unique challenges to clinicians in how they care for critically ill COVID-19 patients who require sedation (Anesth Analg. 2020 Apr 22. doi: 10.1213/ANE.0000000000004887). The use of provisional work areas “has escalated because of the amount of patients we’ve had to care for over the past nine months,” Dr. Greenberg said. “We’ve used alternate providers who are not necessarily familiar with the sedation and analgesic protocols and how to use these specific medications. Drug shortages have been on the rise, so there’s a need to understand alternative agents that can be used.”
COVID-19 patients face the potential risk for an increase in drug-drug interactions and side effects due to the polypharmacy that is often required to provide adequate sedation during mechanical ventilation. He noted that these patients may have “unusually high” analgesia and sedation requirements, particularly when they’re mechanically ventilated. A hypothesis as to why patients with COVID-19 require so much sedation and analgesia is that they often have a high respiratory drive and ventilator dyssynchrony, which requires increased neuromuscular blockade. “They also have an intense inflammatory response, which may be linked to tolerance of specific opioids and other medications,” Dr. Greenberg said. “Many ventilated COVID-19 patients are of younger age and previously in good health, and therefore, have an excellent metabolism. Health care providers are concerned about self-extubation. This prompts bedside providers to administer more sedatives to prevent this unwanted complication. There may also be a reduction of drip modifications by health care workers because of the potential risk of contracting COVID-19 when going into the room multiple times and for long periods of time” (Anesth Analg. 2020;131[1]:e34-e35).
According to a sedation resource on the SCCM website, about 5% of COVID-19 patients require mechanical ventilation. “There has been a massive shortage of the usual drugs that we use,” Dr. Greenberg said. “The demand for sedatives has increased by approximately 91%, while the demand for analgesics has increased by 79%, and neuromuscular blocker demand has increased by 105%.”
A retrospective study of 24 COVID-19 patients who required ventilation in the ICU found that the median daily dose of benzodiazepines was significantly higher, compared with the median daily dose used in the OSCILLATE trial (a median of 270 mg vs. 199 mg, respectively; Anesth Analg. 2020;131[4]e198-e200. doi: 10.1213/ane.0000000000005131). In addition, their median daily dose of opioid was approximately three times higher, compared with patients in the OSCILLATE trial (a median of 775 mg vs. 289 mg). Other agents used included propofol (84%), dexmedetomidine (53%), and ketamine (11%).
“A potential strategy for COVID-19 ICU patient sedation should be analgesia first, as indicated in the 2018 PADIS guidelines,” Dr. Greenberg advised. “We should also apply nonpharmacologic measures to reduce delirium. In nonintubated patients, we should use light to moderate sedation, targeting a RASS of –2 to +1, using hydromorphone or fentanyl boluses for analgesia and midazolam boluses or dexmedetomidine for sedation,.”
For intubated patients, he continued, target a RASS of –3 to –4, or –4 to –5 in those who require neuromuscular blockade. “Use propofol first then intermittent boluses of benzodiazepines,” said Dr. Greenberg, editor-in-chief of the Anesthesia Patient Safety Foundation newsletter. “For heavy sedation, use midazolam and supplement with ketamine and other analgesics and sedatives such as barbiturates, methadone, and even inhalation anesthetics in some cases.”
For analgesia in intubated patients, use fentanyl boluses then infusion. “Patients can easily become tachyphylactic to fentanyl, and it has a long context-sensitive half time,” he said. “Hydromorphone may be least affected by organ dysfunction.”
Dr. Greenberg concluded his presentation by stating that more studies are required “to delineate the best analgesia/sedation strategies and monitoring modalities for COVID-19 ICU patients.”
In commenting on the presentation, Mangala Narasimhan, DO, FCCP, senior vice president and director of critical care services at Northwell Health, said that the recommendations regarding sedation highlight a struggle that ICU providers have been dealing with during the COVID-19 epidemic.
“There have been unique challenges with COVID-19 and intubated patients. We have seen severe ventilator dyssynchrony and prolonged duration of mechanical ventilation. I think we can all agree that these patients have extremely high metabolic rates, have required high levels of sedation, have an increased need for neuromuscular blockade, and have high levels of delirium for extended periods of time. The recommendations provided here are reasonable. Strategies to prevent delirium should be employed, pain management should be prioritized, analgesics can help reduce the need for opioids. Alternatives to sedation are useful in this patient population and are well tolerated. Drug shortages have provided additional challenges to these strategies and have required us to think about the use of alternative agents. The recommendations echo the experience we have had with large numbers of intubated COVID-19 patients.”
Dr. Greenberg disclosed that he receives a stipend from the Anesthesia Patient Safety Foundation for serving as editor-in-chief of the foundation’s newsletter.
According to the best available evidence, analagosedation remains the focus for managing COVID-19 ICU patients, according to Steven B. Greenberg, MD, FCCP, FCCM.
“The choice of sedation and analgesia is important,” Dr. Greenberg, vice chair of education in the department of anesthesiology at Evanston Hospital, part of NorthShore University Health System, Chicago, said at a Society for Critical Care virtual meeting: COVID-19: What’s Next. “We know that the right choice of these two components may increase liberation from ventilators, earlier ICU discharge, and return to normal brain function and independent functional status.”
Analgesia first
Prior to the current pandemic, the approach to sedation of patients in the ICU was based on the PADIS Guidelines of 2018, which call for an assessment-driven, protocol-based stepwise approach to pain and sedation management in critically ill adults (Crit Care Med. 2018;46:e825-73). “” Dr. Greenberg said. “We know that pain management should be a priority of sedation, because pain may increase the risk of delirium, anxiety, and endocrine suppression, and may increase the risk of release of endogenous catecholamines, ischemia, and hypermetabolic states.”
Fentanyl appears to be the most common opioid analgesic used for patients in the ICU, “but fentanyl is a very lipophilic drug and has a long context-sensitive half-life,” he said. “There are components to fentanyl that allow it to become a very long-acting drug upon days and days of infusion. Another opioid used is remifentanil, which is typically short-acting because it is broken down in the blood by esterases, but may cause rigidity at higher doses. Dilaudid seems to be the least affected by organ dysfunction. In our very critically ill, prolonged mechanically ventilated COVID-19 patients, we’ve been using methadone for its NMDA [N-methyl-D-aspartate] antagonistic effect and its opioid-sparing effects.”
As for nonopioid analgesics, Dr. Greenberg said that clinicians have shied away from using NSAIDs because of their side effects. “Tramadol indirectly inhibits reuptake of norepinephrine and serotonin, and ketamine is being used a lot more because of its NMDA antagonist effect,” he said. “Lidocaine and gabapentin have also been used.”
In a recent systematic review and meta-analysis, researchers assessed 34 trials that examined adjuvant analgesic use with an opioid in critically ill patients versus an opioid alone (Crit Care Expl. 2020;2:e0157). They found that when using an adjuvant such as acetaminophen, clonidine, dexmedetomidine, gabapentin, ketamine, magnesium, nefopam, NSAIDs, pregabalin, and tramadol, there was a reduction in pain scores as well as a reduction in opioid consumption. “So, clinicians should consider using adjuvant agents to limit opioid exposure and improve pain scores in the critically ill,” Dr. Greenberg said.
ICU delirium: Risk factors, prevention
Delirium in COVID-19 patients treated in the ICU of particular concern. According to a systematic review of 33 studies, 11 risk factors for delirium in the ICU were supported by strong or moderate levels of evidence (Crit Care Med. 2015;43:40-7). These include age, dementia, hypertension, emergency surgery, trauma, APACHE score of II, need for mechanical ventilation, metabolic acidosis, delirium on prior day, coma, and dexmedetomidine use. Risk factors for ICU delirium among COVID-19 patients, however, “are far different,” Dr. Greenberg said. “Why? First and foremost, we are restricting visitation of family,” he said. “That family connection largely can be lost. Second, there are limitations of nonpharmacologic interventions. There is less mobility and physical therapy employed because of the risk of health care workers’ exposure to the virus. There’s also uncertainty about the global pandemic. Anxiety and depression come with that, as well as disruptions to spiritual and religious services.”
Strategies for preventing delirium remain the same as before the pandemic and in accord with recent clinical practice guidelines: Reduce the use of certain drugs such as benzodiazepines and narcotics, reorient the patients, treat dehydration, use hearing aids and eyeglasses in patients who have them, use ear plugs to cancel noise, mobilize patients, maintain sleep/awake cycles, and encourage sedation holidays (Crit Care Med. 2018;46[9]:e825-73).
A recent study from France found that among 58 patients with COVID-19, 65% had positive Confusion Assessment Method (CAM)–ICU findings and 69% had agitation (N Engl J Med 2020;382:2268-70). Most of the patients (86%) received midazolam, 47% received propofol, and all received sufentanil. “In the pre-COVID days, we would use midazolam as a second-line agent for many of these patients,” Dr. Greenberg said. “So, times really have changed.”
The fate of COVID-19 patients following discharge from the ICU remains a concern, continued Dr. Greenberg, clinical professor of anesthesiology at the University of Chicago. A recent journal article by Michelle Biehl, MD, and Denise Sese, MD, noted that post–intensive care syndrome (PICS) or new or worsening impairment in any physical, cognitive, or mental domain is of significant concern among COVID-19 patients following their ICU stay (Cleveland Clin J Med 2020 Aug doi: 10.3949/ccjm.87a.ccc055). The authors stated that COVID-19 patients may face a higher risk of PICS because of restricted family visitation, prolonged mechanical ventilation, exposure to higher amounts of sedatives, and limited physical therapy during hospital stay.
No ideal sedative agent
The 2018 PADIS Guidelines on the use of ICU sedation suggested strong evidence for modifiable risk factors producing delirium in the context of benzodiazepines and blood transfusion. They recommend a light level of sedation and the use of propofol or dexmedetomidine over benzodiazepines. They also recommend routine delirium testing such as using the CAM-ICU or Intensive Care Delirium Screening Checklist (ICDSC) and nonpharmacologic therapies such as reorientation, cognitive stimulation, sleep improvement, and mobilization.
Several sedation-related factors may be related to an increased risk of delirium. “The type, dose, duration, and mode of delivery are very important,” Dr. Greenberg said. “The ideal sedative agent has a rapid, predictable onset; is short-acting; has anxiolytic, amnestic, and analgesic properties; is soluble; has a high therapeutic index; and no toxicity. The ideal sedative is also easy to administrate, contains no active metabolites, has minimal actions with other drugs, is reversible, and is cost effective. The problem is, there really is no ideal sedative agent. There is inadequate knowledge about the drugs [used to treat COVID-19 in the ICU] available to us, the dosage, and importantly, the pharmacokinetics and dynamics of these medications.”
The classic types of sedation being used in the ICU, he said, include the benzodiazepines midazolam, lorazepam, and diazepam, as well as propofol. Alternatives include dexmedetomidine, clonidine, ketamine, and the neuroleptics – haloperidol, quetiapine, olanzapine, ziprasidone, and risperidone. “The advantages of benzos are that they are anxiolytics, amnestics, and they are good sedatives with minimal hemodynamic effects,” Dr. Greenberg said.
Advantages of propofol include its sedative, hypnotic, and anxiolytic properties, he said. It reduces the cerebral metabolic rate and can relieve bronchospasm. “However, small studies have found that its use may be associated with an increased risk of delirium,” he said. “It is a respiratory depressant, and it can cause hypotension and decreased contractility. It has no analgesic properties, and two of the big concerns of its use in COVID-19 are the potential for hypertriglyceridemia and propofol infusion syndrome, particularly at doses of greater than 5 mg/kg per hour for greater than 48 hours. It is being given in high doses because patients are requiring higher doses to maintain ventilator synchrony.”
Choosing the right drug
The keys to success for sedation of ICU patients are choosing the right drug at the right dose for the right duration and the right mode of delivery, and applying them to the right population. However, as noted in a recent study, the pandemic poses unique challenges to clinicians in how they care for critically ill COVID-19 patients who require sedation (Anesth Analg. 2020 Apr 22. doi: 10.1213/ANE.0000000000004887). The use of provisional work areas “has escalated because of the amount of patients we’ve had to care for over the past nine months,” Dr. Greenberg said. “We’ve used alternate providers who are not necessarily familiar with the sedation and analgesic protocols and how to use these specific medications. Drug shortages have been on the rise, so there’s a need to understand alternative agents that can be used.”
COVID-19 patients face the potential risk for an increase in drug-drug interactions and side effects due to the polypharmacy that is often required to provide adequate sedation during mechanical ventilation. He noted that these patients may have “unusually high” analgesia and sedation requirements, particularly when they’re mechanically ventilated. A hypothesis as to why patients with COVID-19 require so much sedation and analgesia is that they often have a high respiratory drive and ventilator dyssynchrony, which requires increased neuromuscular blockade. “They also have an intense inflammatory response, which may be linked to tolerance of specific opioids and other medications,” Dr. Greenberg said. “Many ventilated COVID-19 patients are of younger age and previously in good health, and therefore, have an excellent metabolism. Health care providers are concerned about self-extubation. This prompts bedside providers to administer more sedatives to prevent this unwanted complication. There may also be a reduction of drip modifications by health care workers because of the potential risk of contracting COVID-19 when going into the room multiple times and for long periods of time” (Anesth Analg. 2020;131[1]:e34-e35).
According to a sedation resource on the SCCM website, about 5% of COVID-19 patients require mechanical ventilation. “There has been a massive shortage of the usual drugs that we use,” Dr. Greenberg said. “The demand for sedatives has increased by approximately 91%, while the demand for analgesics has increased by 79%, and neuromuscular blocker demand has increased by 105%.”
A retrospective study of 24 COVID-19 patients who required ventilation in the ICU found that the median daily dose of benzodiazepines was significantly higher, compared with the median daily dose used in the OSCILLATE trial (a median of 270 mg vs. 199 mg, respectively; Anesth Analg. 2020;131[4]e198-e200. doi: 10.1213/ane.0000000000005131). In addition, their median daily dose of opioid was approximately three times higher, compared with patients in the OSCILLATE trial (a median of 775 mg vs. 289 mg). Other agents used included propofol (84%), dexmedetomidine (53%), and ketamine (11%).
“A potential strategy for COVID-19 ICU patient sedation should be analgesia first, as indicated in the 2018 PADIS guidelines,” Dr. Greenberg advised. “We should also apply nonpharmacologic measures to reduce delirium. In nonintubated patients, we should use light to moderate sedation, targeting a RASS of –2 to +1, using hydromorphone or fentanyl boluses for analgesia and midazolam boluses or dexmedetomidine for sedation,.”
For intubated patients, he continued, target a RASS of –3 to –4, or –4 to –5 in those who require neuromuscular blockade. “Use propofol first then intermittent boluses of benzodiazepines,” said Dr. Greenberg, editor-in-chief of the Anesthesia Patient Safety Foundation newsletter. “For heavy sedation, use midazolam and supplement with ketamine and other analgesics and sedatives such as barbiturates, methadone, and even inhalation anesthetics in some cases.”
For analgesia in intubated patients, use fentanyl boluses then infusion. “Patients can easily become tachyphylactic to fentanyl, and it has a long context-sensitive half time,” he said. “Hydromorphone may be least affected by organ dysfunction.”
Dr. Greenberg concluded his presentation by stating that more studies are required “to delineate the best analgesia/sedation strategies and monitoring modalities for COVID-19 ICU patients.”
In commenting on the presentation, Mangala Narasimhan, DO, FCCP, senior vice president and director of critical care services at Northwell Health, said that the recommendations regarding sedation highlight a struggle that ICU providers have been dealing with during the COVID-19 epidemic.
“There have been unique challenges with COVID-19 and intubated patients. We have seen severe ventilator dyssynchrony and prolonged duration of mechanical ventilation. I think we can all agree that these patients have extremely high metabolic rates, have required high levels of sedation, have an increased need for neuromuscular blockade, and have high levels of delirium for extended periods of time. The recommendations provided here are reasonable. Strategies to prevent delirium should be employed, pain management should be prioritized, analgesics can help reduce the need for opioids. Alternatives to sedation are useful in this patient population and are well tolerated. Drug shortages have provided additional challenges to these strategies and have required us to think about the use of alternative agents. The recommendations echo the experience we have had with large numbers of intubated COVID-19 patients.”
Dr. Greenberg disclosed that he receives a stipend from the Anesthesia Patient Safety Foundation for serving as editor-in-chief of the foundation’s newsletter.
FROM AN SCCM VIRTUAL MEETING
FDA posts COVID vaccine guidance amid White House pushback
while medical and trade associations called for a thorough review of any such product before approval.
The FDA took the unusual step of posting background materials much earlier than usual for its planned Oct. 22 advisory committee meeting on potential vaccines for COVID-19. The FDA also on Tuesday afternoon released a new guidance document, expanding on a previous set of recommendations the agency released in June.
In the new guidance document, FDA officials outline what will be required for even a limited clearance, known as an emergency use authorization (EUA), for a COVID-19 vaccine.
“Data from phase 3 studies should include a median follow-up duration of at least 2 months after completion of the full vaccination regimen to help provide adequate information to assess a vaccine’s benefit-risk profile,” the FDA said in the document.
FDA staff have emphasized the higher bar that drugmakers and regulators face in considering approval of a COVID-19 vaccine.
“Vaccines are complex biological products, and an EUA for a COVID-19 vaccine may allow for rapid and widespread deployment for administration of the vaccine to millions of individuals, including healthy people,” the agency staff said in the briefing documents.
The FDA’s briefing document for the Oct. 22 meeting appears to be markedly at odds with the claim Trump made in a video Monday night, in which he told the American public that “vaccines are coming momentarily.”
Trump, who is in a tightly contested presidential race against Democratic candidate Joe Biden, has repeatedly made claims of the potential arrival of COVID vaccines that are at odds with timelines offered with guarded optimism by experts in infectious diseases.
But based on these new guidelines from the FDA, it appears that the White House may now endorse the FDA’s stance, according to a Wall Street Journal report based on “people familiar with the matter.”
The publication reports that the White House, which has yet to officially comment, “endorsed the U.S. Food and Drug Administration’s plans for assessing whether a Covid-19 vaccine should be given widely, casting aside objections to requirements that would likely mean a shot won’t be cleared until after Election Day, people familiar with the matter said.”
Anthony S. Fauci, MD, director of the National Institute of Allergy and Infectious Diseases, on Monday night said during a virtual appearance at the twenty-first annual New Yorker Festival that there could be evidence as early as November or December about whether one of the vaccines now in testing will work out. He declared himself to have “cautious optimism” about potential rollout of vaccines as early as late 2020 or early 2021.
Peter Lurie, MD, MPH, who earlier served as the FDA’s associate commissioner for public health strategy and analysis, described the agency’s release of the briefing document as being a positive development.
News organizations, including the New York Times, have reported that the White House had sought to block the FDA from releasing further instructions for companies developing COVID-19 vaccines. The Associated Press on Tuesday said that a senior Trump administration official confirmed that the White House had blocked earlier FDA plans to formally publish the safety guidelines based on the 2-month data requirement, arguing that there was “no clinical or medical reason” for it.
“It is an encouraging sign that, despite opposition from the White House, the Food and Drug Administration has effectively published guidelines for emergency release of a vaccine for COVID-19 by disclosing the advice it has been providing to individual sponsors,” said Dr. Lurie, who is now executive director and president of the Center for Science in the Public Interest.
In a news release, he said the White House had sought to keep the FDA guidance under wraps “so it could maintain the public fiction that a safe and effective vaccine could be available before Election Day or even so that it could force emergency authorization of a vaccine with more limited follow-up.”
“Even the pharmaceutical industry has been clamoring for the release of these guidelines. We all want a safe and effective vaccine to end the pandemic, and we want it sooner rather than later,” Dr. Lurie said. “But we can’t afford for the Trump administration to bungle vaccine review the way they’ve bungled nearly every other aspect of its pandemic response.”
Tuesday also saw a flood of statements in support of FDA officials, including tweets from the chief executive of Pfizer, which is among the leaders in the race to develop a COVID-19 vaccine. Pfizer’s Albert Bourla, DVM, PhD, said that the FDA’s “public servants are known for their high integrity and scientific expertise and we have full faith in their ability to set appropriate standards for the approval of a COVID vaccine or treatment.”
The American Medical Association on Tuesday announced a public webinar on Wednesday where its president, Susan R. Bailey, MD, will discuss the COVID-19 vaccine review process with Peter Marks, MD, PhD, director of the Center for Biologics Evaluation and Research at the FDA. The AMA described this webinar as part of work “to restore trust in science and science-based decision-making among policymakers and the public.”
“To ensure media and the physician community are continuously informed about the federal review process for COVID-19 vaccine candidates, the AMA will host a webinar series to gain fact-based insights from the nation’s highest-ranking subject matter experts working to protect the health of the public,” the organization said in announcing the webinar.
In a statement, leaders of the Association of American Medical Colleges said that the FDA’s Vaccines and Related Biological Products Advisory Committee should evaluate any COVID-19 candidate vaccines prior to the FDA issuing an EUA.
“Full approval of a new vaccine or biologic requires demonstration of safety and effectiveness through a process that includes evaluation by the VRBPAC. Their recommendations are considered by FDA staff who ultimately have the authority to approve the new product,” said AAMC chief scientific officer Ross McKinney Jr, MD, and AAMC CEO David J. Skorton, MD, in the statement.
Thomas M. File Jr., MD, president of the Infectious Diseases Society of America, said in a statement that his association again asked the White House to “follow medical and scientific expertise in efforts to combat COVID-19.”
“It is imperative that a vaccine be approved on the basis of FDA’s quality standards and that its safety and efficacy are established before it is authorized,” Dr. File said. “A vaccine that has been approved with speed, rather than safety and efficacy, at the forefront will compound the challenges posed by this pandemic. FDA guidelines for approval that set standards the American people can trust are essential to the success of a vaccine.”
Stephen J. Ubl, chief executive of the Pharmaceutical Research and Manufacturers of America, said in a statement that his association “supports any efforts by FDA to provide clarifying guidance and we have engaged with the agency to support bringing greater transparency to the review process for COVID-19 vaccines.”
“To help address this public health crisis, our companies have also taken unprecedented steps to share vaccine clinical trial protocols and data in real time,” Mr. Ubl said. “We welcome the agency’s efforts to instill confidence in the rigorous safety of these potential vaccines.”
On Oct. 1, Michelle McMurry-Heath, MD, PhD, president and chief executive of the Biotechnology Innovation Organization, released publicly her letter urging Department of Health & Human Services Secretary Alex Azar to “publicly release all new guidance” related to a COVID-19 vaccine. Such a move would bolster public confidence in the vaccine, she said.
“We cannot allow a lack of transparency to undermine confidence in the vaccine development process. The public must have full faith in the scientific process and the rigor of FDA’s regulatory oversight if we are to end the pandemic,” she wrote in the Oct. 1 letter to Azar. “Releasing any additional guidance on granting emergency use authorization for a vaccine will go a long way in accomplishing this critical goal.”
This article first appeared on Medscape.com.
while medical and trade associations called for a thorough review of any such product before approval.
The FDA took the unusual step of posting background materials much earlier than usual for its planned Oct. 22 advisory committee meeting on potential vaccines for COVID-19. The FDA also on Tuesday afternoon released a new guidance document, expanding on a previous set of recommendations the agency released in June.
In the new guidance document, FDA officials outline what will be required for even a limited clearance, known as an emergency use authorization (EUA), for a COVID-19 vaccine.
“Data from phase 3 studies should include a median follow-up duration of at least 2 months after completion of the full vaccination regimen to help provide adequate information to assess a vaccine’s benefit-risk profile,” the FDA said in the document.
FDA staff have emphasized the higher bar that drugmakers and regulators face in considering approval of a COVID-19 vaccine.
“Vaccines are complex biological products, and an EUA for a COVID-19 vaccine may allow for rapid and widespread deployment for administration of the vaccine to millions of individuals, including healthy people,” the agency staff said in the briefing documents.
The FDA’s briefing document for the Oct. 22 meeting appears to be markedly at odds with the claim Trump made in a video Monday night, in which he told the American public that “vaccines are coming momentarily.”
Trump, who is in a tightly contested presidential race against Democratic candidate Joe Biden, has repeatedly made claims of the potential arrival of COVID vaccines that are at odds with timelines offered with guarded optimism by experts in infectious diseases.
But based on these new guidelines from the FDA, it appears that the White House may now endorse the FDA’s stance, according to a Wall Street Journal report based on “people familiar with the matter.”
The publication reports that the White House, which has yet to officially comment, “endorsed the U.S. Food and Drug Administration’s plans for assessing whether a Covid-19 vaccine should be given widely, casting aside objections to requirements that would likely mean a shot won’t be cleared until after Election Day, people familiar with the matter said.”
Anthony S. Fauci, MD, director of the National Institute of Allergy and Infectious Diseases, on Monday night said during a virtual appearance at the twenty-first annual New Yorker Festival that there could be evidence as early as November or December about whether one of the vaccines now in testing will work out. He declared himself to have “cautious optimism” about potential rollout of vaccines as early as late 2020 or early 2021.
Peter Lurie, MD, MPH, who earlier served as the FDA’s associate commissioner for public health strategy and analysis, described the agency’s release of the briefing document as being a positive development.
News organizations, including the New York Times, have reported that the White House had sought to block the FDA from releasing further instructions for companies developing COVID-19 vaccines. The Associated Press on Tuesday said that a senior Trump administration official confirmed that the White House had blocked earlier FDA plans to formally publish the safety guidelines based on the 2-month data requirement, arguing that there was “no clinical or medical reason” for it.
“It is an encouraging sign that, despite opposition from the White House, the Food and Drug Administration has effectively published guidelines for emergency release of a vaccine for COVID-19 by disclosing the advice it has been providing to individual sponsors,” said Dr. Lurie, who is now executive director and president of the Center for Science in the Public Interest.
In a news release, he said the White House had sought to keep the FDA guidance under wraps “so it could maintain the public fiction that a safe and effective vaccine could be available before Election Day or even so that it could force emergency authorization of a vaccine with more limited follow-up.”
“Even the pharmaceutical industry has been clamoring for the release of these guidelines. We all want a safe and effective vaccine to end the pandemic, and we want it sooner rather than later,” Dr. Lurie said. “But we can’t afford for the Trump administration to bungle vaccine review the way they’ve bungled nearly every other aspect of its pandemic response.”
Tuesday also saw a flood of statements in support of FDA officials, including tweets from the chief executive of Pfizer, which is among the leaders in the race to develop a COVID-19 vaccine. Pfizer’s Albert Bourla, DVM, PhD, said that the FDA’s “public servants are known for their high integrity and scientific expertise and we have full faith in their ability to set appropriate standards for the approval of a COVID vaccine or treatment.”
The American Medical Association on Tuesday announced a public webinar on Wednesday where its president, Susan R. Bailey, MD, will discuss the COVID-19 vaccine review process with Peter Marks, MD, PhD, director of the Center for Biologics Evaluation and Research at the FDA. The AMA described this webinar as part of work “to restore trust in science and science-based decision-making among policymakers and the public.”
“To ensure media and the physician community are continuously informed about the federal review process for COVID-19 vaccine candidates, the AMA will host a webinar series to gain fact-based insights from the nation’s highest-ranking subject matter experts working to protect the health of the public,” the organization said in announcing the webinar.
In a statement, leaders of the Association of American Medical Colleges said that the FDA’s Vaccines and Related Biological Products Advisory Committee should evaluate any COVID-19 candidate vaccines prior to the FDA issuing an EUA.
“Full approval of a new vaccine or biologic requires demonstration of safety and effectiveness through a process that includes evaluation by the VRBPAC. Their recommendations are considered by FDA staff who ultimately have the authority to approve the new product,” said AAMC chief scientific officer Ross McKinney Jr, MD, and AAMC CEO David J. Skorton, MD, in the statement.
Thomas M. File Jr., MD, president of the Infectious Diseases Society of America, said in a statement that his association again asked the White House to “follow medical and scientific expertise in efforts to combat COVID-19.”
“It is imperative that a vaccine be approved on the basis of FDA’s quality standards and that its safety and efficacy are established before it is authorized,” Dr. File said. “A vaccine that has been approved with speed, rather than safety and efficacy, at the forefront will compound the challenges posed by this pandemic. FDA guidelines for approval that set standards the American people can trust are essential to the success of a vaccine.”
Stephen J. Ubl, chief executive of the Pharmaceutical Research and Manufacturers of America, said in a statement that his association “supports any efforts by FDA to provide clarifying guidance and we have engaged with the agency to support bringing greater transparency to the review process for COVID-19 vaccines.”
“To help address this public health crisis, our companies have also taken unprecedented steps to share vaccine clinical trial protocols and data in real time,” Mr. Ubl said. “We welcome the agency’s efforts to instill confidence in the rigorous safety of these potential vaccines.”
On Oct. 1, Michelle McMurry-Heath, MD, PhD, president and chief executive of the Biotechnology Innovation Organization, released publicly her letter urging Department of Health & Human Services Secretary Alex Azar to “publicly release all new guidance” related to a COVID-19 vaccine. Such a move would bolster public confidence in the vaccine, she said.
“We cannot allow a lack of transparency to undermine confidence in the vaccine development process. The public must have full faith in the scientific process and the rigor of FDA’s regulatory oversight if we are to end the pandemic,” she wrote in the Oct. 1 letter to Azar. “Releasing any additional guidance on granting emergency use authorization for a vaccine will go a long way in accomplishing this critical goal.”
This article first appeared on Medscape.com.
while medical and trade associations called for a thorough review of any such product before approval.
The FDA took the unusual step of posting background materials much earlier than usual for its planned Oct. 22 advisory committee meeting on potential vaccines for COVID-19. The FDA also on Tuesday afternoon released a new guidance document, expanding on a previous set of recommendations the agency released in June.
In the new guidance document, FDA officials outline what will be required for even a limited clearance, known as an emergency use authorization (EUA), for a COVID-19 vaccine.
“Data from phase 3 studies should include a median follow-up duration of at least 2 months after completion of the full vaccination regimen to help provide adequate information to assess a vaccine’s benefit-risk profile,” the FDA said in the document.
FDA staff have emphasized the higher bar that drugmakers and regulators face in considering approval of a COVID-19 vaccine.
“Vaccines are complex biological products, and an EUA for a COVID-19 vaccine may allow for rapid and widespread deployment for administration of the vaccine to millions of individuals, including healthy people,” the agency staff said in the briefing documents.
The FDA’s briefing document for the Oct. 22 meeting appears to be markedly at odds with the claim Trump made in a video Monday night, in which he told the American public that “vaccines are coming momentarily.”
Trump, who is in a tightly contested presidential race against Democratic candidate Joe Biden, has repeatedly made claims of the potential arrival of COVID vaccines that are at odds with timelines offered with guarded optimism by experts in infectious diseases.
But based on these new guidelines from the FDA, it appears that the White House may now endorse the FDA’s stance, according to a Wall Street Journal report based on “people familiar with the matter.”
The publication reports that the White House, which has yet to officially comment, “endorsed the U.S. Food and Drug Administration’s plans for assessing whether a Covid-19 vaccine should be given widely, casting aside objections to requirements that would likely mean a shot won’t be cleared until after Election Day, people familiar with the matter said.”
Anthony S. Fauci, MD, director of the National Institute of Allergy and Infectious Diseases, on Monday night said during a virtual appearance at the twenty-first annual New Yorker Festival that there could be evidence as early as November or December about whether one of the vaccines now in testing will work out. He declared himself to have “cautious optimism” about potential rollout of vaccines as early as late 2020 or early 2021.
Peter Lurie, MD, MPH, who earlier served as the FDA’s associate commissioner for public health strategy and analysis, described the agency’s release of the briefing document as being a positive development.
News organizations, including the New York Times, have reported that the White House had sought to block the FDA from releasing further instructions for companies developing COVID-19 vaccines. The Associated Press on Tuesday said that a senior Trump administration official confirmed that the White House had blocked earlier FDA plans to formally publish the safety guidelines based on the 2-month data requirement, arguing that there was “no clinical or medical reason” for it.
“It is an encouraging sign that, despite opposition from the White House, the Food and Drug Administration has effectively published guidelines for emergency release of a vaccine for COVID-19 by disclosing the advice it has been providing to individual sponsors,” said Dr. Lurie, who is now executive director and president of the Center for Science in the Public Interest.
In a news release, he said the White House had sought to keep the FDA guidance under wraps “so it could maintain the public fiction that a safe and effective vaccine could be available before Election Day or even so that it could force emergency authorization of a vaccine with more limited follow-up.”
“Even the pharmaceutical industry has been clamoring for the release of these guidelines. We all want a safe and effective vaccine to end the pandemic, and we want it sooner rather than later,” Dr. Lurie said. “But we can’t afford for the Trump administration to bungle vaccine review the way they’ve bungled nearly every other aspect of its pandemic response.”
Tuesday also saw a flood of statements in support of FDA officials, including tweets from the chief executive of Pfizer, which is among the leaders in the race to develop a COVID-19 vaccine. Pfizer’s Albert Bourla, DVM, PhD, said that the FDA’s “public servants are known for their high integrity and scientific expertise and we have full faith in their ability to set appropriate standards for the approval of a COVID vaccine or treatment.”
The American Medical Association on Tuesday announced a public webinar on Wednesday where its president, Susan R. Bailey, MD, will discuss the COVID-19 vaccine review process with Peter Marks, MD, PhD, director of the Center for Biologics Evaluation and Research at the FDA. The AMA described this webinar as part of work “to restore trust in science and science-based decision-making among policymakers and the public.”
“To ensure media and the physician community are continuously informed about the federal review process for COVID-19 vaccine candidates, the AMA will host a webinar series to gain fact-based insights from the nation’s highest-ranking subject matter experts working to protect the health of the public,” the organization said in announcing the webinar.
In a statement, leaders of the Association of American Medical Colleges said that the FDA’s Vaccines and Related Biological Products Advisory Committee should evaluate any COVID-19 candidate vaccines prior to the FDA issuing an EUA.
“Full approval of a new vaccine or biologic requires demonstration of safety and effectiveness through a process that includes evaluation by the VRBPAC. Their recommendations are considered by FDA staff who ultimately have the authority to approve the new product,” said AAMC chief scientific officer Ross McKinney Jr, MD, and AAMC CEO David J. Skorton, MD, in the statement.
Thomas M. File Jr., MD, president of the Infectious Diseases Society of America, said in a statement that his association again asked the White House to “follow medical and scientific expertise in efforts to combat COVID-19.”
“It is imperative that a vaccine be approved on the basis of FDA’s quality standards and that its safety and efficacy are established before it is authorized,” Dr. File said. “A vaccine that has been approved with speed, rather than safety and efficacy, at the forefront will compound the challenges posed by this pandemic. FDA guidelines for approval that set standards the American people can trust are essential to the success of a vaccine.”
Stephen J. Ubl, chief executive of the Pharmaceutical Research and Manufacturers of America, said in a statement that his association “supports any efforts by FDA to provide clarifying guidance and we have engaged with the agency to support bringing greater transparency to the review process for COVID-19 vaccines.”
“To help address this public health crisis, our companies have also taken unprecedented steps to share vaccine clinical trial protocols and data in real time,” Mr. Ubl said. “We welcome the agency’s efforts to instill confidence in the rigorous safety of these potential vaccines.”
On Oct. 1, Michelle McMurry-Heath, MD, PhD, president and chief executive of the Biotechnology Innovation Organization, released publicly her letter urging Department of Health & Human Services Secretary Alex Azar to “publicly release all new guidance” related to a COVID-19 vaccine. Such a move would bolster public confidence in the vaccine, she said.
“We cannot allow a lack of transparency to undermine confidence in the vaccine development process. The public must have full faith in the scientific process and the rigor of FDA’s regulatory oversight if we are to end the pandemic,” she wrote in the Oct. 1 letter to Azar. “Releasing any additional guidance on granting emergency use authorization for a vaccine will go a long way in accomplishing this critical goal.”
This article first appeared on Medscape.com.
INR fails to predict bleeding in patients with cirrhosis
International normalized ratio (INR) does not predict periprocedural bleeding in patients with cirrhosis, according to a meta-analysis of 29 studies.
This finding should deter the common practice of delivering blood products to cirrhotic patients with an elevated INR, reported lead author Alexander J. Kovalic, MD, of Novant Forsyth Medical Center in Winston Salem, N.C., and colleagues.
“INR measurement among cirrhotic patients is important in MELD [Model for End-Stage Liver Disease] prognostication and assessment of underlying hepatic synthetic function, however the INR alone does not capture the complicated interplay of anticoagulant and procoagulant deficiencies present in cirrhotic coagulopathy,” Dr. Kovalic and colleagues wrote in Alimentary Pharmacology & Therapeutics. “Yet, the ‘correction’ of these aberrancies among peripheral coagulation tests remains common ... even in modern practice, and not uncommonly occurs in the periprocedural setting.”
According to investigators, addressing INR with blood transfusion can have a litany of negative effects. Beyond the risks faced by all patient populations, increasing blood volume in those with cirrhosis can increase portal venous pressure, thereby raising risks of portal gastropathy or variceal hemorrhage. In addition, giving plasma products to patients with cirrhotic coagulopathy may further disrupt the balance between anticoagulants and procoagulants, potentially triggering disseminated intravascular coagulation.
Dr. Kovalic and colleagues noted that the lack of correlation between peripheral coagulation tests and bleeding risk has been a longstanding subject of investigation, citing studies from as early as 1981.
To add further weight to this body of evidence, the investigators conducted a systematic review and meta-analysis involving 13,276 patients with cirrhosis who underwent various procedures between 1999 and 2019. Primary outcomes included periprocedural bleeding events and the association between preprocedural INR and periprocedural bleeding events. Secondary outcomes included mortality, quantity of blood and/or plasma products used, and relationship between preprocedural platelet count and periprocedural bleeding events.
The analysis showed that preprocedural INR was not significantly associated with periprocedural bleeding events (pooled odds ratio, 1.52; 95% confidence interval, 0.99-2.33; P = .06), a finding that held across INR threshold subgroups. Similarly, no significant difference was found between mean INR of patients who had bleeding events versus that of those who did not (pooled mean difference, 0.05; 95% CI, 0.03-0.13; P = .23).
Preprocedural platelet count was also a poor predictor of periprocedural bleeding, with a pooled odds ratio of 1.24 (95% CI, 0.55-2.77; P = .60), although the investigators noted that platelet count thresholds varied widely across studies, from 30 to 150 × 109/L. When studies were stratified by procedural bleeding risk or procedure type, subgroup effects were no longer significant. Other secondary endpoints were incalculable because of insufficient data.
“Hopefully, these findings will spark initiation of more large-scale, higher-quality studies ... to reinforce minimizing administration of fresh frozen plasma for inappropriate correction of INR, which carries a multitude of adverse effects among cirrhotic [patients],” the investigators concluded.
According to Stephen H. Caldwell, MD, of the University of Virginia in Charlottesville, “The present paper augments accumulating literature over the past 15 years that INR should be discarded as a measure of procedure-related bleeding risk.”
Dr. Caldwell pointed out that “bleeding in cirrhosis is usually related to portal hypertension not with impaired hemostasis, with the occasional exception of hyperfibrinolysis, which is very different from a prolonged INR.”
He went on to suggest that the present findings should dissuade clinicians from a practice that, for some, is reflexive rather than evidence based.
“It’s remarkable how many medical practices become entrenched based on hand-me-down teaching during our early training years, and remain so for many years beyond as we disperse into various medical and surgical fields,” Dr. Caldwell said. “These learned approaches to common problems can clearly persist for generations despite overwhelming evidence to the contrary that usually evolve slowly and well-insulated within subspecialties or sub-subspecialties, and hence take several generations of training to diffuse into the wider practice of medical care for common problems. These may become matters of expedience in decision-making, much like the old antibiotic conundrum of ‘no-think-a-cillin,’ as critics referred to over-use of broad spectrum antibiotics. And so it has been with the INR.”The investigators disclosed relationships with AbbVie, Eisai, Gilead, and others. Dr. Caldwell disclosed research support from Daiichi concerning the potential role of anticoagulation therapy in preventing cirrhosis progression.
SOURCE: Kovalic AJ et al. Aliment Pharmacol Ther. 2020 Sep 10. doi: 10.1111/apt.16078.
International normalized ratio (INR) does not predict periprocedural bleeding in patients with cirrhosis, according to a meta-analysis of 29 studies.
This finding should deter the common practice of delivering blood products to cirrhotic patients with an elevated INR, reported lead author Alexander J. Kovalic, MD, of Novant Forsyth Medical Center in Winston Salem, N.C., and colleagues.
“INR measurement among cirrhotic patients is important in MELD [Model for End-Stage Liver Disease] prognostication and assessment of underlying hepatic synthetic function, however the INR alone does not capture the complicated interplay of anticoagulant and procoagulant deficiencies present in cirrhotic coagulopathy,” Dr. Kovalic and colleagues wrote in Alimentary Pharmacology & Therapeutics. “Yet, the ‘correction’ of these aberrancies among peripheral coagulation tests remains common ... even in modern practice, and not uncommonly occurs in the periprocedural setting.”
According to investigators, addressing INR with blood transfusion can have a litany of negative effects. Beyond the risks faced by all patient populations, increasing blood volume in those with cirrhosis can increase portal venous pressure, thereby raising risks of portal gastropathy or variceal hemorrhage. In addition, giving plasma products to patients with cirrhotic coagulopathy may further disrupt the balance between anticoagulants and procoagulants, potentially triggering disseminated intravascular coagulation.
Dr. Kovalic and colleagues noted that the lack of correlation between peripheral coagulation tests and bleeding risk has been a longstanding subject of investigation, citing studies from as early as 1981.
To add further weight to this body of evidence, the investigators conducted a systematic review and meta-analysis involving 13,276 patients with cirrhosis who underwent various procedures between 1999 and 2019. Primary outcomes included periprocedural bleeding events and the association between preprocedural INR and periprocedural bleeding events. Secondary outcomes included mortality, quantity of blood and/or plasma products used, and relationship between preprocedural platelet count and periprocedural bleeding events.
The analysis showed that preprocedural INR was not significantly associated with periprocedural bleeding events (pooled odds ratio, 1.52; 95% confidence interval, 0.99-2.33; P = .06), a finding that held across INR threshold subgroups. Similarly, no significant difference was found between mean INR of patients who had bleeding events versus that of those who did not (pooled mean difference, 0.05; 95% CI, 0.03-0.13; P = .23).
Preprocedural platelet count was also a poor predictor of periprocedural bleeding, with a pooled odds ratio of 1.24 (95% CI, 0.55-2.77; P = .60), although the investigators noted that platelet count thresholds varied widely across studies, from 30 to 150 × 109/L. When studies were stratified by procedural bleeding risk or procedure type, subgroup effects were no longer significant. Other secondary endpoints were incalculable because of insufficient data.
“Hopefully, these findings will spark initiation of more large-scale, higher-quality studies ... to reinforce minimizing administration of fresh frozen plasma for inappropriate correction of INR, which carries a multitude of adverse effects among cirrhotic [patients],” the investigators concluded.
According to Stephen H. Caldwell, MD, of the University of Virginia in Charlottesville, “The present paper augments accumulating literature over the past 15 years that INR should be discarded as a measure of procedure-related bleeding risk.”
Dr. Caldwell pointed out that “bleeding in cirrhosis is usually related to portal hypertension not with impaired hemostasis, with the occasional exception of hyperfibrinolysis, which is very different from a prolonged INR.”
He went on to suggest that the present findings should dissuade clinicians from a practice that, for some, is reflexive rather than evidence based.
“It’s remarkable how many medical practices become entrenched based on hand-me-down teaching during our early training years, and remain so for many years beyond as we disperse into various medical and surgical fields,” Dr. Caldwell said. “These learned approaches to common problems can clearly persist for generations despite overwhelming evidence to the contrary that usually evolve slowly and well-insulated within subspecialties or sub-subspecialties, and hence take several generations of training to diffuse into the wider practice of medical care for common problems. These may become matters of expedience in decision-making, much like the old antibiotic conundrum of ‘no-think-a-cillin,’ as critics referred to over-use of broad spectrum antibiotics. And so it has been with the INR.”The investigators disclosed relationships with AbbVie, Eisai, Gilead, and others. Dr. Caldwell disclosed research support from Daiichi concerning the potential role of anticoagulation therapy in preventing cirrhosis progression.
SOURCE: Kovalic AJ et al. Aliment Pharmacol Ther. 2020 Sep 10. doi: 10.1111/apt.16078.
International normalized ratio (INR) does not predict periprocedural bleeding in patients with cirrhosis, according to a meta-analysis of 29 studies.
This finding should deter the common practice of delivering blood products to cirrhotic patients with an elevated INR, reported lead author Alexander J. Kovalic, MD, of Novant Forsyth Medical Center in Winston Salem, N.C., and colleagues.
“INR measurement among cirrhotic patients is important in MELD [Model for End-Stage Liver Disease] prognostication and assessment of underlying hepatic synthetic function, however the INR alone does not capture the complicated interplay of anticoagulant and procoagulant deficiencies present in cirrhotic coagulopathy,” Dr. Kovalic and colleagues wrote in Alimentary Pharmacology & Therapeutics. “Yet, the ‘correction’ of these aberrancies among peripheral coagulation tests remains common ... even in modern practice, and not uncommonly occurs in the periprocedural setting.”
According to investigators, addressing INR with blood transfusion can have a litany of negative effects. Beyond the risks faced by all patient populations, increasing blood volume in those with cirrhosis can increase portal venous pressure, thereby raising risks of portal gastropathy or variceal hemorrhage. In addition, giving plasma products to patients with cirrhotic coagulopathy may further disrupt the balance between anticoagulants and procoagulants, potentially triggering disseminated intravascular coagulation.
Dr. Kovalic and colleagues noted that the lack of correlation between peripheral coagulation tests and bleeding risk has been a longstanding subject of investigation, citing studies from as early as 1981.
To add further weight to this body of evidence, the investigators conducted a systematic review and meta-analysis involving 13,276 patients with cirrhosis who underwent various procedures between 1999 and 2019. Primary outcomes included periprocedural bleeding events and the association between preprocedural INR and periprocedural bleeding events. Secondary outcomes included mortality, quantity of blood and/or plasma products used, and relationship between preprocedural platelet count and periprocedural bleeding events.
The analysis showed that preprocedural INR was not significantly associated with periprocedural bleeding events (pooled odds ratio, 1.52; 95% confidence interval, 0.99-2.33; P = .06), a finding that held across INR threshold subgroups. Similarly, no significant difference was found between mean INR of patients who had bleeding events versus that of those who did not (pooled mean difference, 0.05; 95% CI, 0.03-0.13; P = .23).
Preprocedural platelet count was also a poor predictor of periprocedural bleeding, with a pooled odds ratio of 1.24 (95% CI, 0.55-2.77; P = .60), although the investigators noted that platelet count thresholds varied widely across studies, from 30 to 150 × 109/L. When studies were stratified by procedural bleeding risk or procedure type, subgroup effects were no longer significant. Other secondary endpoints were incalculable because of insufficient data.
“Hopefully, these findings will spark initiation of more large-scale, higher-quality studies ... to reinforce minimizing administration of fresh frozen plasma for inappropriate correction of INR, which carries a multitude of adverse effects among cirrhotic [patients],” the investigators concluded.
According to Stephen H. Caldwell, MD, of the University of Virginia in Charlottesville, “The present paper augments accumulating literature over the past 15 years that INR should be discarded as a measure of procedure-related bleeding risk.”
Dr. Caldwell pointed out that “bleeding in cirrhosis is usually related to portal hypertension not with impaired hemostasis, with the occasional exception of hyperfibrinolysis, which is very different from a prolonged INR.”
He went on to suggest that the present findings should dissuade clinicians from a practice that, for some, is reflexive rather than evidence based.
“It’s remarkable how many medical practices become entrenched based on hand-me-down teaching during our early training years, and remain so for many years beyond as we disperse into various medical and surgical fields,” Dr. Caldwell said. “These learned approaches to common problems can clearly persist for generations despite overwhelming evidence to the contrary that usually evolve slowly and well-insulated within subspecialties or sub-subspecialties, and hence take several generations of training to diffuse into the wider practice of medical care for common problems. These may become matters of expedience in decision-making, much like the old antibiotic conundrum of ‘no-think-a-cillin,’ as critics referred to over-use of broad spectrum antibiotics. And so it has been with the INR.”The investigators disclosed relationships with AbbVie, Eisai, Gilead, and others. Dr. Caldwell disclosed research support from Daiichi concerning the potential role of anticoagulation therapy in preventing cirrhosis progression.
SOURCE: Kovalic AJ et al. Aliment Pharmacol Ther. 2020 Sep 10. doi: 10.1111/apt.16078.
FROM ALIMENTARY PHARMACOLOGY & THERAPEUTICS
EMPEROR-Reduced: Empagliflozin’s HFrEF benefit holds steady on top of sacubitril/valsartan
The latest drug shown to benefit patients with heart failure with reduced ejection fraction, the SGLT2 inhibitor empagliflozin, works just as well when added on top of a second major agent used to treat these patients, the renin-angiotensin system–inhibiting combination of sacubitril/valsartan, based on a post-hoc analysis of data from the EMPEROR-Reduced trial.
“When there are two very effective treatments, it’s common for people to ask: Which should I use?’ The goal of my presentation was to emphasize that the answer is both. We shouldn’t choose between neprilysin inhibition [sacubitril inhibits the enzyme neprilysin] and SGLT2 [sodium-glucose transporter 2] inhibition; we should use both,” said Milton Packer, MD at the virtual annual meeting of the Heart Failure Society of America.
EMPEROR-Reduced had the primary goal of testing the safety and efficacy of the SGLT2 inhibitor empagliflozin (Jardiance) in patients with heart failure with reduced ejection fraction (HFrEF). The results showed that adding this drug on top of standard treatments led to a 25% relative cut in the study’s primary efficacy endpoint, compared with placebo, and had this effect regardless of whether or not patients also had type 2 diabetes (N Engl J Med. 2020 Aug 29. doi: 10.1056/NEJMoa2022190).
Among the 3,730 patients enrolled in the trial, 727 (19%) were on sacubitril/valsartan (Entresto) at entry, which gave Dr. Packer the data to perform the analysis he reported. He presented the study’s three major endpoints as well as a quality of life analysis that compared the performance of empagliflozin in patients who were on sacubitril/valsartan at baseline with the other study patients, who were either on a different type of renin-angiotensin system (RAS) blocker (roughly 70% of study patients) or on no RAS inhibition (about 10% of patients).
The results showed no statistically significant indication of an interaction, suggesting that patients with sacubitril/valsartan on board had just as good response to empagliflozin as patients who were not on this combination. The landmark PARADIGM-HF trial proved several years ago that treatment of HFrEF patients with sacubitril/valsartan led to significantly better outcomes than did treatment with another form of RAS inhibition (N Engl J Med. 2014 Sep 11;371[11]:993-1004).
For example, EMPEROR-Reduced’s primary endpoint, the combined rate of cardiovascular death or hospitalization for heart failure, fell by 36% relative to placebo in patients who received empagliflozin on top of sacubitril/valsartan, and by 23% relative to placebo among the remaining patients who received empagliflozin on top of a different type of RAS inhibitor drug or no RAS inhibition.
“Background treatment with sacubitril/valsartan did not diminish, and may have enhanced the efficacy of empagliflozin,” concluded Dr. Packer. Further analyses also showed that concurrent sacubitril/valsartan had no statistically significant impact on empagliflozin’s ability to reduce the rate of total heart failure hospitalizations, or to slow progressive loss of renal function, compared with placebo. The fourth efficacy analysis Dr. Packer presented showed that empagliflozin was also as effective for improving a quality-of-life measure in patients compared with placebo regardless of the type of RAS inhibition used. For all four outcomes, the point-estimate of empagliflozin’s benefit was higher when used along with sacubitril/valsartan.
Brian L. Claggett, PhD, a biostatistician at Brigham and Women’s Hospital and Harvard Medical School in Boston, designated discussant for the report, disagreed with Dr. Packer’s suggestion that the efficacy of empagliflozin may have been greater when administered against a background of sacubitril/valsartan. From a statistical perspective, there is no basis to suggest that patients did better when they were on both drugs, he cautioned. But Dr. Claggett acknowledged that the new analyses suggested that empagliflozin’s benefit wasn’t compromised by concurrent sacubitril/valsartan use. He also highlighted the value of more fully documenting the safety and efficacy of a new drug when used as part of “comprehensive therapy” with the established drugs that a patient may concurrently receive.
Dr. Packer also presented several measures of treatment safety that all showed similar rates of adverse effects between the empagliflozin and placebo recipients regardless of background RAS inhibition. A notable finding was that the incidence of hypokalemia was 5.9% in patients on empagliflozin and sacubitril/valsartan and 7.5% among patients on empagliflozin and a different type of RAS inhibition.
EMPEROR-Reduced was funded by Boehringer Ingelheim and Eli Lilly, the companies that market empagliflozin. Dr. Packer has received personal fees from Boehringer Ingelheim and Eli Lilly and from several other companies. Dr. Claggett has been a consultant to Amgen, AO Biome, Biogen, Corvia, Myokardia, and Novartis.
The latest drug shown to benefit patients with heart failure with reduced ejection fraction, the SGLT2 inhibitor empagliflozin, works just as well when added on top of a second major agent used to treat these patients, the renin-angiotensin system–inhibiting combination of sacubitril/valsartan, based on a post-hoc analysis of data from the EMPEROR-Reduced trial.
“When there are two very effective treatments, it’s common for people to ask: Which should I use?’ The goal of my presentation was to emphasize that the answer is both. We shouldn’t choose between neprilysin inhibition [sacubitril inhibits the enzyme neprilysin] and SGLT2 [sodium-glucose transporter 2] inhibition; we should use both,” said Milton Packer, MD at the virtual annual meeting of the Heart Failure Society of America.
EMPEROR-Reduced had the primary goal of testing the safety and efficacy of the SGLT2 inhibitor empagliflozin (Jardiance) in patients with heart failure with reduced ejection fraction (HFrEF). The results showed that adding this drug on top of standard treatments led to a 25% relative cut in the study’s primary efficacy endpoint, compared with placebo, and had this effect regardless of whether or not patients also had type 2 diabetes (N Engl J Med. 2020 Aug 29. doi: 10.1056/NEJMoa2022190).
Among the 3,730 patients enrolled in the trial, 727 (19%) were on sacubitril/valsartan (Entresto) at entry, which gave Dr. Packer the data to perform the analysis he reported. He presented the study’s three major endpoints as well as a quality of life analysis that compared the performance of empagliflozin in patients who were on sacubitril/valsartan at baseline with the other study patients, who were either on a different type of renin-angiotensin system (RAS) blocker (roughly 70% of study patients) or on no RAS inhibition (about 10% of patients).
The results showed no statistically significant indication of an interaction, suggesting that patients with sacubitril/valsartan on board had just as good response to empagliflozin as patients who were not on this combination. The landmark PARADIGM-HF trial proved several years ago that treatment of HFrEF patients with sacubitril/valsartan led to significantly better outcomes than did treatment with another form of RAS inhibition (N Engl J Med. 2014 Sep 11;371[11]:993-1004).
For example, EMPEROR-Reduced’s primary endpoint, the combined rate of cardiovascular death or hospitalization for heart failure, fell by 36% relative to placebo in patients who received empagliflozin on top of sacubitril/valsartan, and by 23% relative to placebo among the remaining patients who received empagliflozin on top of a different type of RAS inhibitor drug or no RAS inhibition.
“Background treatment with sacubitril/valsartan did not diminish, and may have enhanced the efficacy of empagliflozin,” concluded Dr. Packer. Further analyses also showed that concurrent sacubitril/valsartan had no statistically significant impact on empagliflozin’s ability to reduce the rate of total heart failure hospitalizations, or to slow progressive loss of renal function, compared with placebo. The fourth efficacy analysis Dr. Packer presented showed that empagliflozin was also as effective for improving a quality-of-life measure in patients compared with placebo regardless of the type of RAS inhibition used. For all four outcomes, the point-estimate of empagliflozin’s benefit was higher when used along with sacubitril/valsartan.
Brian L. Claggett, PhD, a biostatistician at Brigham and Women’s Hospital and Harvard Medical School in Boston, designated discussant for the report, disagreed with Dr. Packer’s suggestion that the efficacy of empagliflozin may have been greater when administered against a background of sacubitril/valsartan. From a statistical perspective, there is no basis to suggest that patients did better when they were on both drugs, he cautioned. But Dr. Claggett acknowledged that the new analyses suggested that empagliflozin’s benefit wasn’t compromised by concurrent sacubitril/valsartan use. He also highlighted the value of more fully documenting the safety and efficacy of a new drug when used as part of “comprehensive therapy” with the established drugs that a patient may concurrently receive.
Dr. Packer also presented several measures of treatment safety that all showed similar rates of adverse effects between the empagliflozin and placebo recipients regardless of background RAS inhibition. A notable finding was that the incidence of hypokalemia was 5.9% in patients on empagliflozin and sacubitril/valsartan and 7.5% among patients on empagliflozin and a different type of RAS inhibition.
EMPEROR-Reduced was funded by Boehringer Ingelheim and Eli Lilly, the companies that market empagliflozin. Dr. Packer has received personal fees from Boehringer Ingelheim and Eli Lilly and from several other companies. Dr. Claggett has been a consultant to Amgen, AO Biome, Biogen, Corvia, Myokardia, and Novartis.
The latest drug shown to benefit patients with heart failure with reduced ejection fraction, the SGLT2 inhibitor empagliflozin, works just as well when added on top of a second major agent used to treat these patients, the renin-angiotensin system–inhibiting combination of sacubitril/valsartan, based on a post-hoc analysis of data from the EMPEROR-Reduced trial.
“When there are two very effective treatments, it’s common for people to ask: Which should I use?’ The goal of my presentation was to emphasize that the answer is both. We shouldn’t choose between neprilysin inhibition [sacubitril inhibits the enzyme neprilysin] and SGLT2 [sodium-glucose transporter 2] inhibition; we should use both,” said Milton Packer, MD at the virtual annual meeting of the Heart Failure Society of America.
EMPEROR-Reduced had the primary goal of testing the safety and efficacy of the SGLT2 inhibitor empagliflozin (Jardiance) in patients with heart failure with reduced ejection fraction (HFrEF). The results showed that adding this drug on top of standard treatments led to a 25% relative cut in the study’s primary efficacy endpoint, compared with placebo, and had this effect regardless of whether or not patients also had type 2 diabetes (N Engl J Med. 2020 Aug 29. doi: 10.1056/NEJMoa2022190).
Among the 3,730 patients enrolled in the trial, 727 (19%) were on sacubitril/valsartan (Entresto) at entry, which gave Dr. Packer the data to perform the analysis he reported. He presented the study’s three major endpoints as well as a quality of life analysis that compared the performance of empagliflozin in patients who were on sacubitril/valsartan at baseline with the other study patients, who were either on a different type of renin-angiotensin system (RAS) blocker (roughly 70% of study patients) or on no RAS inhibition (about 10% of patients).
The results showed no statistically significant indication of an interaction, suggesting that patients with sacubitril/valsartan on board had just as good response to empagliflozin as patients who were not on this combination. The landmark PARADIGM-HF trial proved several years ago that treatment of HFrEF patients with sacubitril/valsartan led to significantly better outcomes than did treatment with another form of RAS inhibition (N Engl J Med. 2014 Sep 11;371[11]:993-1004).
For example, EMPEROR-Reduced’s primary endpoint, the combined rate of cardiovascular death or hospitalization for heart failure, fell by 36% relative to placebo in patients who received empagliflozin on top of sacubitril/valsartan, and by 23% relative to placebo among the remaining patients who received empagliflozin on top of a different type of RAS inhibitor drug or no RAS inhibition.
“Background treatment with sacubitril/valsartan did not diminish, and may have enhanced the efficacy of empagliflozin,” concluded Dr. Packer. Further analyses also showed that concurrent sacubitril/valsartan had no statistically significant impact on empagliflozin’s ability to reduce the rate of total heart failure hospitalizations, or to slow progressive loss of renal function, compared with placebo. The fourth efficacy analysis Dr. Packer presented showed that empagliflozin was also as effective for improving a quality-of-life measure in patients compared with placebo regardless of the type of RAS inhibition used. For all four outcomes, the point-estimate of empagliflozin’s benefit was higher when used along with sacubitril/valsartan.
Brian L. Claggett, PhD, a biostatistician at Brigham and Women’s Hospital and Harvard Medical School in Boston, designated discussant for the report, disagreed with Dr. Packer’s suggestion that the efficacy of empagliflozin may have been greater when administered against a background of sacubitril/valsartan. From a statistical perspective, there is no basis to suggest that patients did better when they were on both drugs, he cautioned. But Dr. Claggett acknowledged that the new analyses suggested that empagliflozin’s benefit wasn’t compromised by concurrent sacubitril/valsartan use. He also highlighted the value of more fully documenting the safety and efficacy of a new drug when used as part of “comprehensive therapy” with the established drugs that a patient may concurrently receive.
Dr. Packer also presented several measures of treatment safety that all showed similar rates of adverse effects between the empagliflozin and placebo recipients regardless of background RAS inhibition. A notable finding was that the incidence of hypokalemia was 5.9% in patients on empagliflozin and sacubitril/valsartan and 7.5% among patients on empagliflozin and a different type of RAS inhibition.
EMPEROR-Reduced was funded by Boehringer Ingelheim and Eli Lilly, the companies that market empagliflozin. Dr. Packer has received personal fees from Boehringer Ingelheim and Eli Lilly and from several other companies. Dr. Claggett has been a consultant to Amgen, AO Biome, Biogen, Corvia, Myokardia, and Novartis.
FROM HFSA 2020
HM20 Virtual: Key takeaways for the pediatric hospitalist
The HM20 Virtual conference in August was filled with excellent content that can be applied by all hospitalists. This article summarizes key concepts and takeaways for the pediatric hospitalist.
Racism and bias in medicine
HM20 Virtual session: Structural Racism and Bias in Hospital Medicine During Two Pandemics
Presenters: Nathan Chomilo, MD, FAAP, and Benji K. Mathews, MD, SFHM
Dr. Nathan Chomilo, of HealthPartners in Minneapolis, opened the session sharing how racial disparities were a symptom of racism. The presenters explained how structural racism has been propagated in medicine with the Hospital Survey and Construction Act of 1964 that allowed segregated hospitals, as well as the racism that exists within the “hidden curriculum.”
Dr. Chomilo discussed how personal experiences of racism can lead to worse health outcomes, including depression, obesity, and overall poor health. Dr. Mathews, also of HealthPartners, discussed how implicit biases can be addressed at the individual level, the organizational level, and simultaneously at both levels to create an antiracist culture. He presented strategies to mitigate individual biases; recognizing when biases may be triggered, checking biases at the door, connecting with others from different backgrounds as equals, and practicing antiracism by being an active bystander. Dr. Chomilo concluded the session by sharing that we can all grow by addressing racism at “our houses” (health care systems, medical schools, payer systems) with the goal to create an antiracist system.
Key takeaways
- Racial disparities are a symptom of structural racism that has been propagated in medicine for centuries.
- Addressing implicit biases at the individual level, organization level, and simultaneously at both levels can help leaders model and promote an antiracism culture.
HM20 Virtual session: When Grief and Crises Intersect: Perspectives of a Black Physician in the Time of Two Pandemics
Presenter: Kimberly Manning, MD, FACP, FAAP
Dr. Kimberly Manning, of Emory University in Atlanta, discussed the dual pandemics of COVID-19 and the racism that we are currently experiencing and tried to describe the unique perspective of Black Americans. Though it is easy to see that COVID-19 is a pandemic, racism is not always seen in this way. Dr. Manning demonstrated that, when a pandemic is defined as “that which occurs over a wide geographic area and affects a high proportion of the population,” racism is absolutely a pandemic. Black Americans have been disproportionately affected by COVID-19. Dr. Manning said we often hear that we are in unprecedented times but as far as racism is concerned, there is nothing new about this. She shared stories of personal milestones and how each of these instances, though marked by something beautiful, was also marked by something truly awful. Each time she had a reason to smile, there was something awful going on in the country that showed how racism was still present. Dr. Manning described that, though these were her stories, all Black Americans can recount the same stories, emotions, and feelings of grief.
Dr. Manning concluded by sharing how we can “Do The Work” to combat the pandemic of racism: broaden our funds of knowledge, remember that people are grieving, explore our implicit biases, be brave bystanders, and avoid performative allyship
Key takeaways
- Though the COVID19 pandemic is unprecedented, the pandemic of racism is not.
- We must “Do The Work” to combat everyday racism and to be cognizant of what our Black colleagues are going through every day.
Immigrant hospitalist challenges
HM20 Virtual session: The Immigrant Hospitalist: Navigating the Uncertain Terrain During COVID-19
Presenters: Manpreet S. Malik, MD, FHM, and Benji K. Mathews, MD, SFHM
Dr. Malik of Emory University in Atlanta, and Dr. Mathews of HealthPartners, opened this session by sharing their personal stories as immigrant physicians in the United States. Dr. Malik noted that physicians born outside the United States make up 29% of U.S. physicians, and 32% of hospitalists are international medical graduates.
The presenters revealed the structural hurdles immigrant physicians face, including lack of empowerment until achieving permanent residency status; limited leadership, administrative, and academic roles; concerns for job security and financial stability; and experiencing micro- and macroaggressions at work. The presenters shared a framework for a developmental orientation inclined toward cultural competency beginning with denial, followed by polarization, progressing to minimization, advancing to acceptance, and culminating in adaptation.
They concluded the session by stressing the importance of advocacy for immigrant physicians and encouraged colleagues to become engaged in efforts within their professional organizations.
Key takeaway
- Immigrant physicians experience structural challenges to their professional advancement because of their residency status.
Learner supervision
HM20 Virtual session: Call Me Maybe: Balancing Resident Autonomy With Sensible Supervision
Presenter: Daniel Steinberg, MD, SFHM, FACP
Dr. Steinberg, based at Mount Sinai in New York, explained that resident supervision is driven by three factors: what residents need, what residents want, and what the supervisor can provide. Although data is mixed as to whether supervision improves patient outcomes, supervision is essential for patient care and resident education. Dr. Steinberg showcased several relevant medical education studies related to supervision and focused on a key question: Do you trust the resident? The review of medical education literature discussed the meaning and development of trust, oral case presentations to determine trust, and the influence of supervisor experience.
Key takeaways
- Resident supervision is driven by what residents need, what residents want, and what the supervisor can provide.
- Trust can be determined from direct supervision, oral presentations, and remote access of electronic medical records, but is also influenced by attending experience and style.
Balancing personal and professional life
HM20 Virtual session: Being a Hospitalist and a Parent: Balancing Roles With Grace
Presenters: Heather E. Nye, MD, PhD, SFHM, and David Alfandre, MD, MSPH
Dr. Nye of the University of California, San Francisco, and Dr. Alfandre of New York University shared their challenges as hospitalists, parents, and partners before and during the COVID-19 pandemic. They described their feelings of guilt during the pandemic related to increasing their families’ infection risks, as well as the gratitude they felt for having stable jobs during the crisis. They shared solutions that worked for their families, including cooperating with their partners, expressing their scheduling needs, and negotiating to meet those needs with their employers.
Dr. Nye and Dr. Alfandre recommended staying connected with others for help, including partners, institutions, neighbors, and colleagues. They also recommended supporting, sharing, collaborating, and connecting with their partners and colleagues to maintain a balanced professional and personal life.
Key takeaway:
- Stay connected and support, collaborate, and share with your colleagues and partner at home.
Dr. Kumar is the pediatric editor of The Hospitalist. She is clinical assistant professor of pediatrics at the Cleveland Clinic Lerner College of Medicine at Case Western Reserve University and a pediatric hospitalist at Cleveland Clinic Children’s. Dr. Doraiswamy is an assistant professor of medicine and pediatrics and a med-peds hospitalist at The Ohio State University and Nationwide Children’s Hospital, both in Columbus. Dr. Tantoco is an academic med-peds hospitalist practicing in Northwestern Memorial Hospital, Chicago, and Ann & Robert H. Lurie Children’s Hospital of Chicago.
The HM20 Virtual conference in August was filled with excellent content that can be applied by all hospitalists. This article summarizes key concepts and takeaways for the pediatric hospitalist.
Racism and bias in medicine
HM20 Virtual session: Structural Racism and Bias in Hospital Medicine During Two Pandemics
Presenters: Nathan Chomilo, MD, FAAP, and Benji K. Mathews, MD, SFHM
Dr. Nathan Chomilo, of HealthPartners in Minneapolis, opened the session sharing how racial disparities were a symptom of racism. The presenters explained how structural racism has been propagated in medicine with the Hospital Survey and Construction Act of 1964 that allowed segregated hospitals, as well as the racism that exists within the “hidden curriculum.”
Dr. Chomilo discussed how personal experiences of racism can lead to worse health outcomes, including depression, obesity, and overall poor health. Dr. Mathews, also of HealthPartners, discussed how implicit biases can be addressed at the individual level, the organizational level, and simultaneously at both levels to create an antiracist culture. He presented strategies to mitigate individual biases; recognizing when biases may be triggered, checking biases at the door, connecting with others from different backgrounds as equals, and practicing antiracism by being an active bystander. Dr. Chomilo concluded the session by sharing that we can all grow by addressing racism at “our houses” (health care systems, medical schools, payer systems) with the goal to create an antiracist system.
Key takeaways
- Racial disparities are a symptom of structural racism that has been propagated in medicine for centuries.
- Addressing implicit biases at the individual level, organization level, and simultaneously at both levels can help leaders model and promote an antiracism culture.
HM20 Virtual session: When Grief and Crises Intersect: Perspectives of a Black Physician in the Time of Two Pandemics
Presenter: Kimberly Manning, MD, FACP, FAAP
Dr. Kimberly Manning, of Emory University in Atlanta, discussed the dual pandemics of COVID-19 and the racism that we are currently experiencing and tried to describe the unique perspective of Black Americans. Though it is easy to see that COVID-19 is a pandemic, racism is not always seen in this way. Dr. Manning demonstrated that, when a pandemic is defined as “that which occurs over a wide geographic area and affects a high proportion of the population,” racism is absolutely a pandemic. Black Americans have been disproportionately affected by COVID-19. Dr. Manning said we often hear that we are in unprecedented times but as far as racism is concerned, there is nothing new about this. She shared stories of personal milestones and how each of these instances, though marked by something beautiful, was also marked by something truly awful. Each time she had a reason to smile, there was something awful going on in the country that showed how racism was still present. Dr. Manning described that, though these were her stories, all Black Americans can recount the same stories, emotions, and feelings of grief.
Dr. Manning concluded by sharing how we can “Do The Work” to combat the pandemic of racism: broaden our funds of knowledge, remember that people are grieving, explore our implicit biases, be brave bystanders, and avoid performative allyship
Key takeaways
- Though the COVID19 pandemic is unprecedented, the pandemic of racism is not.
- We must “Do The Work” to combat everyday racism and to be cognizant of what our Black colleagues are going through every day.
Immigrant hospitalist challenges
HM20 Virtual session: The Immigrant Hospitalist: Navigating the Uncertain Terrain During COVID-19
Presenters: Manpreet S. Malik, MD, FHM, and Benji K. Mathews, MD, SFHM
Dr. Malik of Emory University in Atlanta, and Dr. Mathews of HealthPartners, opened this session by sharing their personal stories as immigrant physicians in the United States. Dr. Malik noted that physicians born outside the United States make up 29% of U.S. physicians, and 32% of hospitalists are international medical graduates.
The presenters revealed the structural hurdles immigrant physicians face, including lack of empowerment until achieving permanent residency status; limited leadership, administrative, and academic roles; concerns for job security and financial stability; and experiencing micro- and macroaggressions at work. The presenters shared a framework for a developmental orientation inclined toward cultural competency beginning with denial, followed by polarization, progressing to minimization, advancing to acceptance, and culminating in adaptation.
They concluded the session by stressing the importance of advocacy for immigrant physicians and encouraged colleagues to become engaged in efforts within their professional organizations.
Key takeaway
- Immigrant physicians experience structural challenges to their professional advancement because of their residency status.
Learner supervision
HM20 Virtual session: Call Me Maybe: Balancing Resident Autonomy With Sensible Supervision
Presenter: Daniel Steinberg, MD, SFHM, FACP
Dr. Steinberg, based at Mount Sinai in New York, explained that resident supervision is driven by three factors: what residents need, what residents want, and what the supervisor can provide. Although data is mixed as to whether supervision improves patient outcomes, supervision is essential for patient care and resident education. Dr. Steinberg showcased several relevant medical education studies related to supervision and focused on a key question: Do you trust the resident? The review of medical education literature discussed the meaning and development of trust, oral case presentations to determine trust, and the influence of supervisor experience.
Key takeaways
- Resident supervision is driven by what residents need, what residents want, and what the supervisor can provide.
- Trust can be determined from direct supervision, oral presentations, and remote access of electronic medical records, but is also influenced by attending experience and style.
Balancing personal and professional life
HM20 Virtual session: Being a Hospitalist and a Parent: Balancing Roles With Grace
Presenters: Heather E. Nye, MD, PhD, SFHM, and David Alfandre, MD, MSPH
Dr. Nye of the University of California, San Francisco, and Dr. Alfandre of New York University shared their challenges as hospitalists, parents, and partners before and during the COVID-19 pandemic. They described their feelings of guilt during the pandemic related to increasing their families’ infection risks, as well as the gratitude they felt for having stable jobs during the crisis. They shared solutions that worked for their families, including cooperating with their partners, expressing their scheduling needs, and negotiating to meet those needs with their employers.
Dr. Nye and Dr. Alfandre recommended staying connected with others for help, including partners, institutions, neighbors, and colleagues. They also recommended supporting, sharing, collaborating, and connecting with their partners and colleagues to maintain a balanced professional and personal life.
Key takeaway:
- Stay connected and support, collaborate, and share with your colleagues and partner at home.
Dr. Kumar is the pediatric editor of The Hospitalist. She is clinical assistant professor of pediatrics at the Cleveland Clinic Lerner College of Medicine at Case Western Reserve University and a pediatric hospitalist at Cleveland Clinic Children’s. Dr. Doraiswamy is an assistant professor of medicine and pediatrics and a med-peds hospitalist at The Ohio State University and Nationwide Children’s Hospital, both in Columbus. Dr. Tantoco is an academic med-peds hospitalist practicing in Northwestern Memorial Hospital, Chicago, and Ann & Robert H. Lurie Children’s Hospital of Chicago.
The HM20 Virtual conference in August was filled with excellent content that can be applied by all hospitalists. This article summarizes key concepts and takeaways for the pediatric hospitalist.
Racism and bias in medicine
HM20 Virtual session: Structural Racism and Bias in Hospital Medicine During Two Pandemics
Presenters: Nathan Chomilo, MD, FAAP, and Benji K. Mathews, MD, SFHM
Dr. Nathan Chomilo, of HealthPartners in Minneapolis, opened the session sharing how racial disparities were a symptom of racism. The presenters explained how structural racism has been propagated in medicine with the Hospital Survey and Construction Act of 1964 that allowed segregated hospitals, as well as the racism that exists within the “hidden curriculum.”
Dr. Chomilo discussed how personal experiences of racism can lead to worse health outcomes, including depression, obesity, and overall poor health. Dr. Mathews, also of HealthPartners, discussed how implicit biases can be addressed at the individual level, the organizational level, and simultaneously at both levels to create an antiracist culture. He presented strategies to mitigate individual biases; recognizing when biases may be triggered, checking biases at the door, connecting with others from different backgrounds as equals, and practicing antiracism by being an active bystander. Dr. Chomilo concluded the session by sharing that we can all grow by addressing racism at “our houses” (health care systems, medical schools, payer systems) with the goal to create an antiracist system.
Key takeaways
- Racial disparities are a symptom of structural racism that has been propagated in medicine for centuries.
- Addressing implicit biases at the individual level, organization level, and simultaneously at both levels can help leaders model and promote an antiracism culture.
HM20 Virtual session: When Grief and Crises Intersect: Perspectives of a Black Physician in the Time of Two Pandemics
Presenter: Kimberly Manning, MD, FACP, FAAP
Dr. Kimberly Manning, of Emory University in Atlanta, discussed the dual pandemics of COVID-19 and the racism that we are currently experiencing and tried to describe the unique perspective of Black Americans. Though it is easy to see that COVID-19 is a pandemic, racism is not always seen in this way. Dr. Manning demonstrated that, when a pandemic is defined as “that which occurs over a wide geographic area and affects a high proportion of the population,” racism is absolutely a pandemic. Black Americans have been disproportionately affected by COVID-19. Dr. Manning said we often hear that we are in unprecedented times but as far as racism is concerned, there is nothing new about this. She shared stories of personal milestones and how each of these instances, though marked by something beautiful, was also marked by something truly awful. Each time she had a reason to smile, there was something awful going on in the country that showed how racism was still present. Dr. Manning described that, though these were her stories, all Black Americans can recount the same stories, emotions, and feelings of grief.
Dr. Manning concluded by sharing how we can “Do The Work” to combat the pandemic of racism: broaden our funds of knowledge, remember that people are grieving, explore our implicit biases, be brave bystanders, and avoid performative allyship
Key takeaways
- Though the COVID19 pandemic is unprecedented, the pandemic of racism is not.
- We must “Do The Work” to combat everyday racism and to be cognizant of what our Black colleagues are going through every day.
Immigrant hospitalist challenges
HM20 Virtual session: The Immigrant Hospitalist: Navigating the Uncertain Terrain During COVID-19
Presenters: Manpreet S. Malik, MD, FHM, and Benji K. Mathews, MD, SFHM
Dr. Malik of Emory University in Atlanta, and Dr. Mathews of HealthPartners, opened this session by sharing their personal stories as immigrant physicians in the United States. Dr. Malik noted that physicians born outside the United States make up 29% of U.S. physicians, and 32% of hospitalists are international medical graduates.
The presenters revealed the structural hurdles immigrant physicians face, including lack of empowerment until achieving permanent residency status; limited leadership, administrative, and academic roles; concerns for job security and financial stability; and experiencing micro- and macroaggressions at work. The presenters shared a framework for a developmental orientation inclined toward cultural competency beginning with denial, followed by polarization, progressing to minimization, advancing to acceptance, and culminating in adaptation.
They concluded the session by stressing the importance of advocacy for immigrant physicians and encouraged colleagues to become engaged in efforts within their professional organizations.
Key takeaway
- Immigrant physicians experience structural challenges to their professional advancement because of their residency status.
Learner supervision
HM20 Virtual session: Call Me Maybe: Balancing Resident Autonomy With Sensible Supervision
Presenter: Daniel Steinberg, MD, SFHM, FACP
Dr. Steinberg, based at Mount Sinai in New York, explained that resident supervision is driven by three factors: what residents need, what residents want, and what the supervisor can provide. Although data is mixed as to whether supervision improves patient outcomes, supervision is essential for patient care and resident education. Dr. Steinberg showcased several relevant medical education studies related to supervision and focused on a key question: Do you trust the resident? The review of medical education literature discussed the meaning and development of trust, oral case presentations to determine trust, and the influence of supervisor experience.
Key takeaways
- Resident supervision is driven by what residents need, what residents want, and what the supervisor can provide.
- Trust can be determined from direct supervision, oral presentations, and remote access of electronic medical records, but is also influenced by attending experience and style.
Balancing personal and professional life
HM20 Virtual session: Being a Hospitalist and a Parent: Balancing Roles With Grace
Presenters: Heather E. Nye, MD, PhD, SFHM, and David Alfandre, MD, MSPH
Dr. Nye of the University of California, San Francisco, and Dr. Alfandre of New York University shared their challenges as hospitalists, parents, and partners before and during the COVID-19 pandemic. They described their feelings of guilt during the pandemic related to increasing their families’ infection risks, as well as the gratitude they felt for having stable jobs during the crisis. They shared solutions that worked for their families, including cooperating with their partners, expressing their scheduling needs, and negotiating to meet those needs with their employers.
Dr. Nye and Dr. Alfandre recommended staying connected with others for help, including partners, institutions, neighbors, and colleagues. They also recommended supporting, sharing, collaborating, and connecting with their partners and colleagues to maintain a balanced professional and personal life.
Key takeaway:
- Stay connected and support, collaborate, and share with your colleagues and partner at home.
Dr. Kumar is the pediatric editor of The Hospitalist. She is clinical assistant professor of pediatrics at the Cleveland Clinic Lerner College of Medicine at Case Western Reserve University and a pediatric hospitalist at Cleveland Clinic Children’s. Dr. Doraiswamy is an assistant professor of medicine and pediatrics and a med-peds hospitalist at The Ohio State University and Nationwide Children’s Hospital, both in Columbus. Dr. Tantoco is an academic med-peds hospitalist practicing in Northwestern Memorial Hospital, Chicago, and Ann & Robert H. Lurie Children’s Hospital of Chicago.
Stroke may be the first symptom of COVID-19 in younger patients
new research suggests. Investigators carried out a meta-analysis of data, including 160 patients with COVID-19 and stroke, and found that nearly half of patients under the age of 50 were asymptomatic at the time of stroke onset.
Although younger patients had the highest risk of stroke, the highest risk of death was in patients who were older, had other chronic conditions, and had more severe COVID-19–associated respiratory symptoms.
“One of the most eye-opening findings of this study is that, for patients under 50 years old, many were totally asymptomatic when they had a stroke related to COVID-19, [which] means that, for these patients, the stroke was their first symptom of the disease,” lead author Luciano Sposato, MD, MBA, associate professor and chair in stroke research at Western University, London, Ont.
The study was published online Sept. 15 in Neurology.
Anecdotal reports
“In early April of 2020, we realized that COVID-19 was a highly thrombogenic disease,” said Dr. Sposato. “Almost in parallel, I started to see anecdotal reports in social media of strokes occurring in patients with COVID-19, and there were also very few case reports.”
The investigators “thought it would be a good idea to put all the data together in one paper,” he said, and began by conducting a systematic review of 10 published studies of COVID-19 and stroke (n = 125 patients), which were then pooled with 35 unpublished cases from Canada, the United States, and Iran for a total of 160 cases.
The analysis examined in-hospital mortality rates of patients with stroke and COVID-19.
In addition, the researchers conducted a second review of 150 papers, encompassing a final cohort of 3,306 COVID-19 patients with stroke of any type and 5,322 with ischemic stroke.
“Some studies reported data for only ischemic stroke, and some reported data for all strokes considered together, which resulted in a different number of patients on each meta-analysis, with a lower number of ‘any stroke’ cases,” Dr. Sposato explained. “This review looked at the number of patients who developed a stroke during admission and included thousands of patients.”
Dr. Sposato noted that the first review was conducted on single case reports and small case series “to understand the clinical characteristics of strokes in patients with COVID-19 on an individual patient level,” since “large studies, including hundreds of thousands of patients, usually do not provide the level of detail for a descriptive analysis of the clinical characteristics of a disease.”
Cluster analyses were used to “identify specific clinical phenotypes and their relationship with death.” Patients were stratified into three age groups: <50, 50-70, and >70 years (“young,” “middle aged,” and “older,” respectively). The median age was 65 years and 43% were female.
Mortality ‘remarkably high’
The review showed that 1.8% (95% confidence interval, 0.9%-3.7%) of patients experienced a new stroke, while 1.5% (95% CI, 0.8%-2.8%) of these experienced an ischemic stroke. “These numbers are higher than historical data for other infectious diseases – for example, 0.75% in SARS-CoV-1, 0.78% in sepsis, and 0.2% in influenza,” Dr. Sposato commented.
Moreover, “this number may be an underestimate, given that many patients die without a confirmed diagnosis and that some patients did not come to the emergency department when experiencing mild symptoms during the first months of the pandemic,” he added.
Focusing on the review of 160 patients, the researchers described in-hospital mortality for strokes of all types and for ischemic strokes alone as “remarkably high” (34.4% [95% CI, 27.2%-42.4%] and 35.7% [95% CI, 27.5%-44.8%], respectively), with most deaths occurring among ischemic stroke patients.
“This high mortality rate is higher than the [roughly] 15% to 30% reported for stroke patients without COVID-19 admitted to intensive care units,” Dr. Sposato said.
High-risk phenotype
Many “young” COVID-19 patients (under age 50) who had a stroke (42.9%) had no previous risk factors or comorbidities. Moreover, in almost half of these patients (48.3%), stroke was more likely to occur before the onset of any COVID-19 respiratory symptoms.
Additionally, younger patients showed the highest frequency of elevated cardiac troponin compared with middle-aged and older patients (71.4% vs. 48.4% and 27.8%, respectively). On the other hand, mortality was 67% lower in younger versus older patients (odds ratio, 0.33; 95% CI, 0.12-0.94; P = .039).
Dr. Sposato noted that the proportion of ischemic stroke patients with large-vessel occlusion was “higher than previously reported” for patients with stroke without COVID-19 (47% compared with 29%, respectively).
“We should consider COVID-19 as a new cause or risk factor for stroke. At least, patients with stroke should probably be tested for SARS-CoV-2 infection if they are young and present with a large-vessel occlusion, even in the absence of typical COVID-19 respiratory symptoms,” he suggested.
The researchers identified a “high-risk phenotype” for death for all types of stroke considered together: older age, a higher burden of comorbidities, and severe COVID-19 respiratory symptoms. Patients with all three characteristics had the highest in-hospital mortality rate (58.6%) and a threefold risk of death, compared with the rest of the cohort (OR, 3.52; 95% CI, 1.53-8.09; P = .003).
“Several potential mechanisms can explain the increased risk of stroke among COVID-19 patients, but perhaps the most important one is increased thrombogenesis secondary to an exaggerated inflammatory response,” Dr. Sposato said.
Not just elders
Commenting on the study, Jodi Edwards, PhD, director of the Brain and Heart Nexus Research Program at the University of Ottawa Heart Institute, said the findings are “consistent with and underscore public health messaging emphasizing that COVID-19 does not only affect the elderly and those with underlying health conditions, but can have serious and even fatal consequences at any age.”
Dr. Edwards, who was not involved with the study, emphasized that “adherence to public health recommendations is critical to begin to reduce the rising incidence in younger adults.”
Dr. Sposato acknowledged that the study was small and that there “can be problems associated with a systematic review of case reports, such as publication bias, lack of completeness of data, etc, so more research is needed.”
Dr. Sposato is supported by the Kathleen & Dr. Henry Barnett Research Chair in Stroke Research at Western University, the Edward and Alma Saraydar Neurosciences Fund of the London Health Sciences Foundation, and the Opportunities Fund of the Academic Health Sciences Centre Alternative Funding Plan of the Academic Medical Organization of Southwestern Ontario. Dr. Sposato reported speaker honoraria from Boehringer Ingelheim, Pfizer, Gore, and Bayer and research/quality improvement grants from Boehringer Ingelheim and Bayer. The other authors’ disclosures are listed on the original article. Dr. Edwards has disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
new research suggests. Investigators carried out a meta-analysis of data, including 160 patients with COVID-19 and stroke, and found that nearly half of patients under the age of 50 were asymptomatic at the time of stroke onset.
Although younger patients had the highest risk of stroke, the highest risk of death was in patients who were older, had other chronic conditions, and had more severe COVID-19–associated respiratory symptoms.
“One of the most eye-opening findings of this study is that, for patients under 50 years old, many were totally asymptomatic when they had a stroke related to COVID-19, [which] means that, for these patients, the stroke was their first symptom of the disease,” lead author Luciano Sposato, MD, MBA, associate professor and chair in stroke research at Western University, London, Ont.
The study was published online Sept. 15 in Neurology.
Anecdotal reports
“In early April of 2020, we realized that COVID-19 was a highly thrombogenic disease,” said Dr. Sposato. “Almost in parallel, I started to see anecdotal reports in social media of strokes occurring in patients with COVID-19, and there were also very few case reports.”
The investigators “thought it would be a good idea to put all the data together in one paper,” he said, and began by conducting a systematic review of 10 published studies of COVID-19 and stroke (n = 125 patients), which were then pooled with 35 unpublished cases from Canada, the United States, and Iran for a total of 160 cases.
The analysis examined in-hospital mortality rates of patients with stroke and COVID-19.
In addition, the researchers conducted a second review of 150 papers, encompassing a final cohort of 3,306 COVID-19 patients with stroke of any type and 5,322 with ischemic stroke.
“Some studies reported data for only ischemic stroke, and some reported data for all strokes considered together, which resulted in a different number of patients on each meta-analysis, with a lower number of ‘any stroke’ cases,” Dr. Sposato explained. “This review looked at the number of patients who developed a stroke during admission and included thousands of patients.”
Dr. Sposato noted that the first review was conducted on single case reports and small case series “to understand the clinical characteristics of strokes in patients with COVID-19 on an individual patient level,” since “large studies, including hundreds of thousands of patients, usually do not provide the level of detail for a descriptive analysis of the clinical characteristics of a disease.”
Cluster analyses were used to “identify specific clinical phenotypes and their relationship with death.” Patients were stratified into three age groups: <50, 50-70, and >70 years (“young,” “middle aged,” and “older,” respectively). The median age was 65 years and 43% were female.
Mortality ‘remarkably high’
The review showed that 1.8% (95% confidence interval, 0.9%-3.7%) of patients experienced a new stroke, while 1.5% (95% CI, 0.8%-2.8%) of these experienced an ischemic stroke. “These numbers are higher than historical data for other infectious diseases – for example, 0.75% in SARS-CoV-1, 0.78% in sepsis, and 0.2% in influenza,” Dr. Sposato commented.
Moreover, “this number may be an underestimate, given that many patients die without a confirmed diagnosis and that some patients did not come to the emergency department when experiencing mild symptoms during the first months of the pandemic,” he added.
Focusing on the review of 160 patients, the researchers described in-hospital mortality for strokes of all types and for ischemic strokes alone as “remarkably high” (34.4% [95% CI, 27.2%-42.4%] and 35.7% [95% CI, 27.5%-44.8%], respectively), with most deaths occurring among ischemic stroke patients.
“This high mortality rate is higher than the [roughly] 15% to 30% reported for stroke patients without COVID-19 admitted to intensive care units,” Dr. Sposato said.
High-risk phenotype
Many “young” COVID-19 patients (under age 50) who had a stroke (42.9%) had no previous risk factors or comorbidities. Moreover, in almost half of these patients (48.3%), stroke was more likely to occur before the onset of any COVID-19 respiratory symptoms.
Additionally, younger patients showed the highest frequency of elevated cardiac troponin compared with middle-aged and older patients (71.4% vs. 48.4% and 27.8%, respectively). On the other hand, mortality was 67% lower in younger versus older patients (odds ratio, 0.33; 95% CI, 0.12-0.94; P = .039).
Dr. Sposato noted that the proportion of ischemic stroke patients with large-vessel occlusion was “higher than previously reported” for patients with stroke without COVID-19 (47% compared with 29%, respectively).
“We should consider COVID-19 as a new cause or risk factor for stroke. At least, patients with stroke should probably be tested for SARS-CoV-2 infection if they are young and present with a large-vessel occlusion, even in the absence of typical COVID-19 respiratory symptoms,” he suggested.
The researchers identified a “high-risk phenotype” for death for all types of stroke considered together: older age, a higher burden of comorbidities, and severe COVID-19 respiratory symptoms. Patients with all three characteristics had the highest in-hospital mortality rate (58.6%) and a threefold risk of death, compared with the rest of the cohort (OR, 3.52; 95% CI, 1.53-8.09; P = .003).
“Several potential mechanisms can explain the increased risk of stroke among COVID-19 patients, but perhaps the most important one is increased thrombogenesis secondary to an exaggerated inflammatory response,” Dr. Sposato said.
Not just elders
Commenting on the study, Jodi Edwards, PhD, director of the Brain and Heart Nexus Research Program at the University of Ottawa Heart Institute, said the findings are “consistent with and underscore public health messaging emphasizing that COVID-19 does not only affect the elderly and those with underlying health conditions, but can have serious and even fatal consequences at any age.”
Dr. Edwards, who was not involved with the study, emphasized that “adherence to public health recommendations is critical to begin to reduce the rising incidence in younger adults.”
Dr. Sposato acknowledged that the study was small and that there “can be problems associated with a systematic review of case reports, such as publication bias, lack of completeness of data, etc, so more research is needed.”
Dr. Sposato is supported by the Kathleen & Dr. Henry Barnett Research Chair in Stroke Research at Western University, the Edward and Alma Saraydar Neurosciences Fund of the London Health Sciences Foundation, and the Opportunities Fund of the Academic Health Sciences Centre Alternative Funding Plan of the Academic Medical Organization of Southwestern Ontario. Dr. Sposato reported speaker honoraria from Boehringer Ingelheim, Pfizer, Gore, and Bayer and research/quality improvement grants from Boehringer Ingelheim and Bayer. The other authors’ disclosures are listed on the original article. Dr. Edwards has disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
new research suggests. Investigators carried out a meta-analysis of data, including 160 patients with COVID-19 and stroke, and found that nearly half of patients under the age of 50 were asymptomatic at the time of stroke onset.
Although younger patients had the highest risk of stroke, the highest risk of death was in patients who were older, had other chronic conditions, and had more severe COVID-19–associated respiratory symptoms.
“One of the most eye-opening findings of this study is that, for patients under 50 years old, many were totally asymptomatic when they had a stroke related to COVID-19, [which] means that, for these patients, the stroke was their first symptom of the disease,” lead author Luciano Sposato, MD, MBA, associate professor and chair in stroke research at Western University, London, Ont.
The study was published online Sept. 15 in Neurology.
Anecdotal reports
“In early April of 2020, we realized that COVID-19 was a highly thrombogenic disease,” said Dr. Sposato. “Almost in parallel, I started to see anecdotal reports in social media of strokes occurring in patients with COVID-19, and there were also very few case reports.”
The investigators “thought it would be a good idea to put all the data together in one paper,” he said, and began by conducting a systematic review of 10 published studies of COVID-19 and stroke (n = 125 patients), which were then pooled with 35 unpublished cases from Canada, the United States, and Iran for a total of 160 cases.
The analysis examined in-hospital mortality rates of patients with stroke and COVID-19.
In addition, the researchers conducted a second review of 150 papers, encompassing a final cohort of 3,306 COVID-19 patients with stroke of any type and 5,322 with ischemic stroke.
“Some studies reported data for only ischemic stroke, and some reported data for all strokes considered together, which resulted in a different number of patients on each meta-analysis, with a lower number of ‘any stroke’ cases,” Dr. Sposato explained. “This review looked at the number of patients who developed a stroke during admission and included thousands of patients.”
Dr. Sposato noted that the first review was conducted on single case reports and small case series “to understand the clinical characteristics of strokes in patients with COVID-19 on an individual patient level,” since “large studies, including hundreds of thousands of patients, usually do not provide the level of detail for a descriptive analysis of the clinical characteristics of a disease.”
Cluster analyses were used to “identify specific clinical phenotypes and their relationship with death.” Patients were stratified into three age groups: <50, 50-70, and >70 years (“young,” “middle aged,” and “older,” respectively). The median age was 65 years and 43% were female.
Mortality ‘remarkably high’
The review showed that 1.8% (95% confidence interval, 0.9%-3.7%) of patients experienced a new stroke, while 1.5% (95% CI, 0.8%-2.8%) of these experienced an ischemic stroke. “These numbers are higher than historical data for other infectious diseases – for example, 0.75% in SARS-CoV-1, 0.78% in sepsis, and 0.2% in influenza,” Dr. Sposato commented.
Moreover, “this number may be an underestimate, given that many patients die without a confirmed diagnosis and that some patients did not come to the emergency department when experiencing mild symptoms during the first months of the pandemic,” he added.
Focusing on the review of 160 patients, the researchers described in-hospital mortality for strokes of all types and for ischemic strokes alone as “remarkably high” (34.4% [95% CI, 27.2%-42.4%] and 35.7% [95% CI, 27.5%-44.8%], respectively), with most deaths occurring among ischemic stroke patients.
“This high mortality rate is higher than the [roughly] 15% to 30% reported for stroke patients without COVID-19 admitted to intensive care units,” Dr. Sposato said.
High-risk phenotype
Many “young” COVID-19 patients (under age 50) who had a stroke (42.9%) had no previous risk factors or comorbidities. Moreover, in almost half of these patients (48.3%), stroke was more likely to occur before the onset of any COVID-19 respiratory symptoms.
Additionally, younger patients showed the highest frequency of elevated cardiac troponin compared with middle-aged and older patients (71.4% vs. 48.4% and 27.8%, respectively). On the other hand, mortality was 67% lower in younger versus older patients (odds ratio, 0.33; 95% CI, 0.12-0.94; P = .039).
Dr. Sposato noted that the proportion of ischemic stroke patients with large-vessel occlusion was “higher than previously reported” for patients with stroke without COVID-19 (47% compared with 29%, respectively).
“We should consider COVID-19 as a new cause or risk factor for stroke. At least, patients with stroke should probably be tested for SARS-CoV-2 infection if they are young and present with a large-vessel occlusion, even in the absence of typical COVID-19 respiratory symptoms,” he suggested.
The researchers identified a “high-risk phenotype” for death for all types of stroke considered together: older age, a higher burden of comorbidities, and severe COVID-19 respiratory symptoms. Patients with all three characteristics had the highest in-hospital mortality rate (58.6%) and a threefold risk of death, compared with the rest of the cohort (OR, 3.52; 95% CI, 1.53-8.09; P = .003).
“Several potential mechanisms can explain the increased risk of stroke among COVID-19 patients, but perhaps the most important one is increased thrombogenesis secondary to an exaggerated inflammatory response,” Dr. Sposato said.
Not just elders
Commenting on the study, Jodi Edwards, PhD, director of the Brain and Heart Nexus Research Program at the University of Ottawa Heart Institute, said the findings are “consistent with and underscore public health messaging emphasizing that COVID-19 does not only affect the elderly and those with underlying health conditions, but can have serious and even fatal consequences at any age.”
Dr. Edwards, who was not involved with the study, emphasized that “adherence to public health recommendations is critical to begin to reduce the rising incidence in younger adults.”
Dr. Sposato acknowledged that the study was small and that there “can be problems associated with a systematic review of case reports, such as publication bias, lack of completeness of data, etc, so more research is needed.”
Dr. Sposato is supported by the Kathleen & Dr. Henry Barnett Research Chair in Stroke Research at Western University, the Edward and Alma Saraydar Neurosciences Fund of the London Health Sciences Foundation, and the Opportunities Fund of the Academic Health Sciences Centre Alternative Funding Plan of the Academic Medical Organization of Southwestern Ontario. Dr. Sposato reported speaker honoraria from Boehringer Ingelheim, Pfizer, Gore, and Bayer and research/quality improvement grants from Boehringer Ingelheim and Bayer. The other authors’ disclosures are listed on the original article. Dr. Edwards has disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
From Neurology
CDC flips, acknowledges aerosol spread of COVID-19
The information reiterates, however, that “COVID-19 is thought to spread mainly through close contact from person to person, including between people who are physically near each other (within about 6 feet). People who are infected but do not show symptoms can also spread the virus to others.”
In a statement to the media, the CDC said, “Today’s update acknowledges the existence of some published reports showing limited, uncommon circumstances where people with COVID-19 infected others who were more than 6 feet away or shortly after the COVID-19–positive person left an area. In these instances, transmission occurred in poorly ventilated and enclosed spaces that often involved activities that caused heavier breathing, like singing or exercise. Such environments and activities may contribute to the buildup of virus-carrying particles.”
“This is HUGE and been long delayed. But glad it’s now CDC official,” tweeted Eric Feigl-Ding, MD, an epidemiologist and health economist at Harvard University, Boston on Oct. 5.
The CDC announcement follows an abrupt flip-flop on information last month surrounding the aerosol spread of the virus.
Information deleted from website last month
On September 18, the CDC had added to its existing guidance that the virus is spread “through respiratory droplets or small particles, such as those in aerosols, produced when an infected person coughs, sneezes, sings, talks, or breathes. These particles can be inhaled into the nose, mouth, airways, and lungs and cause infection.”
The CDC then deleted that guidance on Sept. 21, saying it was a draft update released in error.
A key element of the now-deleted guidance said, “this is thought to be the main way the virus spreads.”
The information updated today reverses the now-deleted guidance and says aerosol transmission is not the main way the virus spreads.
It states that people who are within 6 feet of a person with COVID-19 or have direct contact with that person have the greatest risk of infection.
The CDC reiterated in the statement to the media today, “People can protect themselves from the virus that causes COVID-19 by staying at least 6 feet away from others, wearing a mask that covers their nose and mouth, washing their hands frequently, cleaning touched surfaces often, and staying home when sick.”
Among the journals that have published evidence on aerosol spread is Clinical Infectious Diseases, which, on July 6, published the paper, “It Is Time to Address Airborne Transmission of Coronavirus Disease 2019,” which was supported by 239 scientists.
The authors wrote, “there is significant potential for inhalation exposure to viruses in microscopic respiratory droplets (microdroplets) at short to medium distances (up to several meters, or room scale).”
Aerosols and airborne transmission “are the only way to explain super-spreader events we are seeing,” said Kimberly Prather, PhD, an atmospheric chemist at the University of California at San Diego, in an interview Oct. 5 with the Washington Post.
Dr. Prather added that, once aerosolization is acknowledged, this becomes a “fixable” problem through proper ventilation.
“Wear masks at all times indoors when others are present,” Dr. Prather said. But when inside, she said, there’s no such thing as a completely safe social distance.
This article first appeared on Medscape.com.
The information reiterates, however, that “COVID-19 is thought to spread mainly through close contact from person to person, including between people who are physically near each other (within about 6 feet). People who are infected but do not show symptoms can also spread the virus to others.”
In a statement to the media, the CDC said, “Today’s update acknowledges the existence of some published reports showing limited, uncommon circumstances where people with COVID-19 infected others who were more than 6 feet away or shortly after the COVID-19–positive person left an area. In these instances, transmission occurred in poorly ventilated and enclosed spaces that often involved activities that caused heavier breathing, like singing or exercise. Such environments and activities may contribute to the buildup of virus-carrying particles.”
“This is HUGE and been long delayed. But glad it’s now CDC official,” tweeted Eric Feigl-Ding, MD, an epidemiologist and health economist at Harvard University, Boston on Oct. 5.
The CDC announcement follows an abrupt flip-flop on information last month surrounding the aerosol spread of the virus.
Information deleted from website last month
On September 18, the CDC had added to its existing guidance that the virus is spread “through respiratory droplets or small particles, such as those in aerosols, produced when an infected person coughs, sneezes, sings, talks, or breathes. These particles can be inhaled into the nose, mouth, airways, and lungs and cause infection.”
The CDC then deleted that guidance on Sept. 21, saying it was a draft update released in error.
A key element of the now-deleted guidance said, “this is thought to be the main way the virus spreads.”
The information updated today reverses the now-deleted guidance and says aerosol transmission is not the main way the virus spreads.
It states that people who are within 6 feet of a person with COVID-19 or have direct contact with that person have the greatest risk of infection.
The CDC reiterated in the statement to the media today, “People can protect themselves from the virus that causes COVID-19 by staying at least 6 feet away from others, wearing a mask that covers their nose and mouth, washing their hands frequently, cleaning touched surfaces often, and staying home when sick.”
Among the journals that have published evidence on aerosol spread is Clinical Infectious Diseases, which, on July 6, published the paper, “It Is Time to Address Airborne Transmission of Coronavirus Disease 2019,” which was supported by 239 scientists.
The authors wrote, “there is significant potential for inhalation exposure to viruses in microscopic respiratory droplets (microdroplets) at short to medium distances (up to several meters, or room scale).”
Aerosols and airborne transmission “are the only way to explain super-spreader events we are seeing,” said Kimberly Prather, PhD, an atmospheric chemist at the University of California at San Diego, in an interview Oct. 5 with the Washington Post.
Dr. Prather added that, once aerosolization is acknowledged, this becomes a “fixable” problem through proper ventilation.
“Wear masks at all times indoors when others are present,” Dr. Prather said. But when inside, she said, there’s no such thing as a completely safe social distance.
This article first appeared on Medscape.com.
The information reiterates, however, that “COVID-19 is thought to spread mainly through close contact from person to person, including between people who are physically near each other (within about 6 feet). People who are infected but do not show symptoms can also spread the virus to others.”
In a statement to the media, the CDC said, “Today’s update acknowledges the existence of some published reports showing limited, uncommon circumstances where people with COVID-19 infected others who were more than 6 feet away or shortly after the COVID-19–positive person left an area. In these instances, transmission occurred in poorly ventilated and enclosed spaces that often involved activities that caused heavier breathing, like singing or exercise. Such environments and activities may contribute to the buildup of virus-carrying particles.”
“This is HUGE and been long delayed. But glad it’s now CDC official,” tweeted Eric Feigl-Ding, MD, an epidemiologist and health economist at Harvard University, Boston on Oct. 5.
The CDC announcement follows an abrupt flip-flop on information last month surrounding the aerosol spread of the virus.
Information deleted from website last month
On September 18, the CDC had added to its existing guidance that the virus is spread “through respiratory droplets or small particles, such as those in aerosols, produced when an infected person coughs, sneezes, sings, talks, or breathes. These particles can be inhaled into the nose, mouth, airways, and lungs and cause infection.”
The CDC then deleted that guidance on Sept. 21, saying it was a draft update released in error.
A key element of the now-deleted guidance said, “this is thought to be the main way the virus spreads.”
The information updated today reverses the now-deleted guidance and says aerosol transmission is not the main way the virus spreads.
It states that people who are within 6 feet of a person with COVID-19 or have direct contact with that person have the greatest risk of infection.
The CDC reiterated in the statement to the media today, “People can protect themselves from the virus that causes COVID-19 by staying at least 6 feet away from others, wearing a mask that covers their nose and mouth, washing their hands frequently, cleaning touched surfaces often, and staying home when sick.”
Among the journals that have published evidence on aerosol spread is Clinical Infectious Diseases, which, on July 6, published the paper, “It Is Time to Address Airborne Transmission of Coronavirus Disease 2019,” which was supported by 239 scientists.
The authors wrote, “there is significant potential for inhalation exposure to viruses in microscopic respiratory droplets (microdroplets) at short to medium distances (up to several meters, or room scale).”
Aerosols and airborne transmission “are the only way to explain super-spreader events we are seeing,” said Kimberly Prather, PhD, an atmospheric chemist at the University of California at San Diego, in an interview Oct. 5 with the Washington Post.
Dr. Prather added that, once aerosolization is acknowledged, this becomes a “fixable” problem through proper ventilation.
“Wear masks at all times indoors when others are present,” Dr. Prather said. But when inside, she said, there’s no such thing as a completely safe social distance.
This article first appeared on Medscape.com.










