User login
Optimal psychiatric treatment: Target the brain and avoid the body
Pharmacotherapy for psychiatric disorders is a mixed blessing. The advent of psychotropic medications since the 1950s (antipsychotics, antidepressants, anxiolytics, mood stabilizers) has revolutionized the treatment of serious psychiatric brain disorders, allowing certain patients to be discharged to the community after a lifetime of institutionalization.
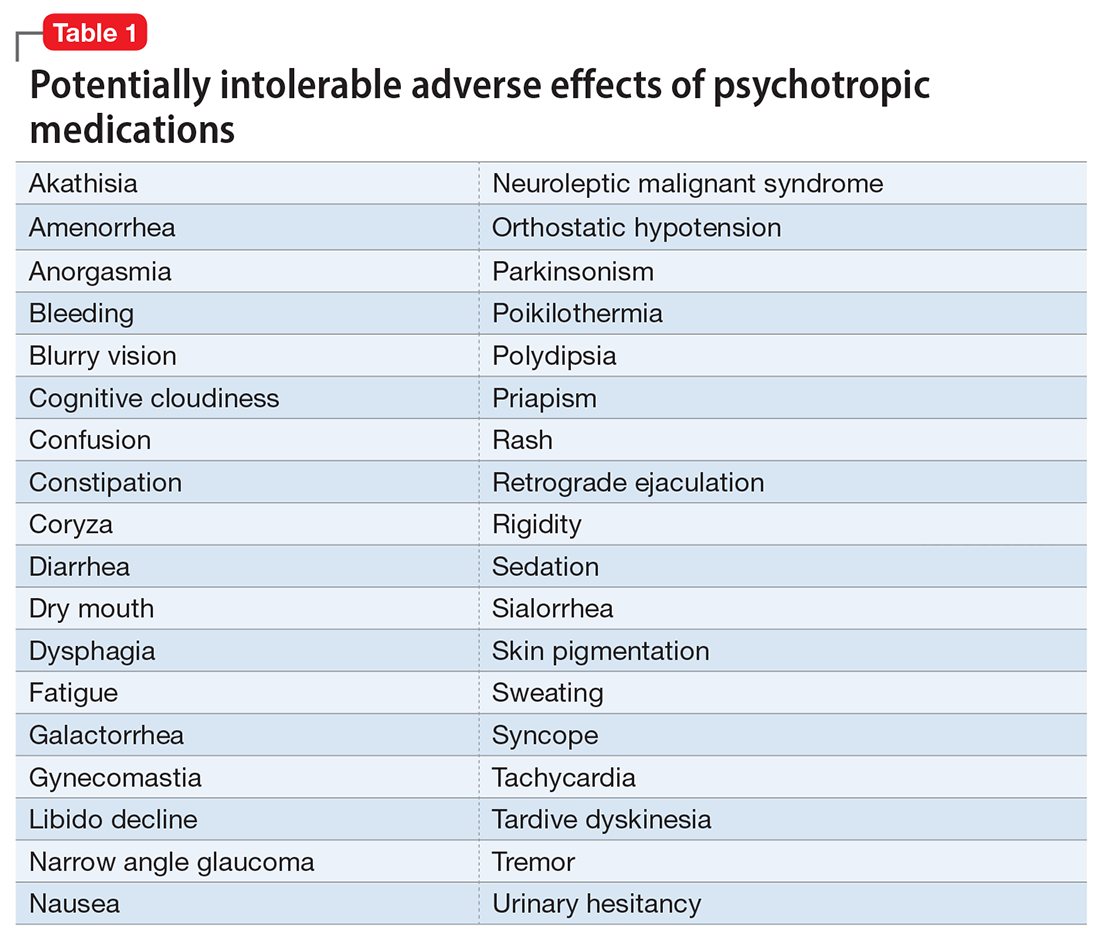
However, like all medications, psychotropic agents are often associated with various potentially intolerable symptoms (Table 1) or safety complications (Table 2) because they interact with every organ in the body besides their intended target, the brain, and its neurochemical circuitry.
Imagine if we could treat our psychiatric patients while bypassing the body and achieve response, remission, and ultimately recovery without any systemic adverse effects. Adherence would dramatically improve, our patients’ quality of life would be enhanced, and the overall effectiveness (defined as the complex package of efficacy, safety, and tolerability) would be superior to current pharmacotherapies. This is important because most psychiatric medications must be taken daily for years, even a lifetime, to avoid a relapse of the illness. Psychiatrists frequently must manage adverse effects or switch the patient to a different medication if a tolerability or safety issue emerges, which is very common in psychiatric practice. A significant part of psychopharmacologic management includes ordering various laboratory tests to monitor adverse reactions in major organs, especially the liver, kidney, and heart. Additionally, psychiatric physicians must be constantly cognizant of medications prescribed by other clinicians for comorbid medical conditions to successfully navigate the turbulent seas of pharmacokinetic interactions.
I am sure you have noticed that whenever you watch a direct-to-consumer commercial for any medication, 90% of the advertisement is a background voice listing the various tolerability and safety complications of the medication as required by the FDA. Interestingly, these ads frequently contain colorful scenery and joyful clips, which I suspect are cleverly designed to distract the audience from focusing on the list of adverse effects.
Benefits of nonpharmacologic treatments
No wonder I am a fan of psychotherapy, a well-established psychiatric treatment modality that completely avoids body tissues. It directly targets the brain without needlessly interacting with any other organ. Psychotherapy’s many benefits (improving insight, enhancing adherence, improving self-esteem, reducing risky behaviors, guiding stress management and coping skills, modifying unhealthy beliefs, and ultimately relieving symptoms such as anxiety and depression) are achieved without any somatic adverse effects! Psychotherapy has also been shown to induce neuroplasticity and reduce inflammatory biomarkers.1 Unlike FDA-approved medications, psychotherapy does not include a “package insert,” 10 to 20 pages (in small print) that mostly focus on warnings, precautions, and sundry physical adverse effects. Even the dosing of psychotherapy is left entirely up to the treating clinician!
Although I have had many gratifying results with pharmacotherapy in my practice, especially in combination with psychotherapy,2 I also have observed excellent outcomes with nonpharmacologic approaches, especially neuromodulation therapies. The best antidepressant I have ever used since my residency training days is electroconvulsive therapy (ECT). My experience is consistent with a large meta-analysis3showing a huge effect size (Cohen d = .91) in contrast to the usual effect size of .3 to .5 for standard antidepressants (except IV ketamine). A recent study showed ECT is even better than the vaunted rapid-acting ketamine,4 which is further evidence of its remarkable efficacy in depression. Neuroimaging studies report that ECT rapidly increases the volume of the hippocampus,5,6 which shrinks in size in patients with unipolar or bipolar depression.
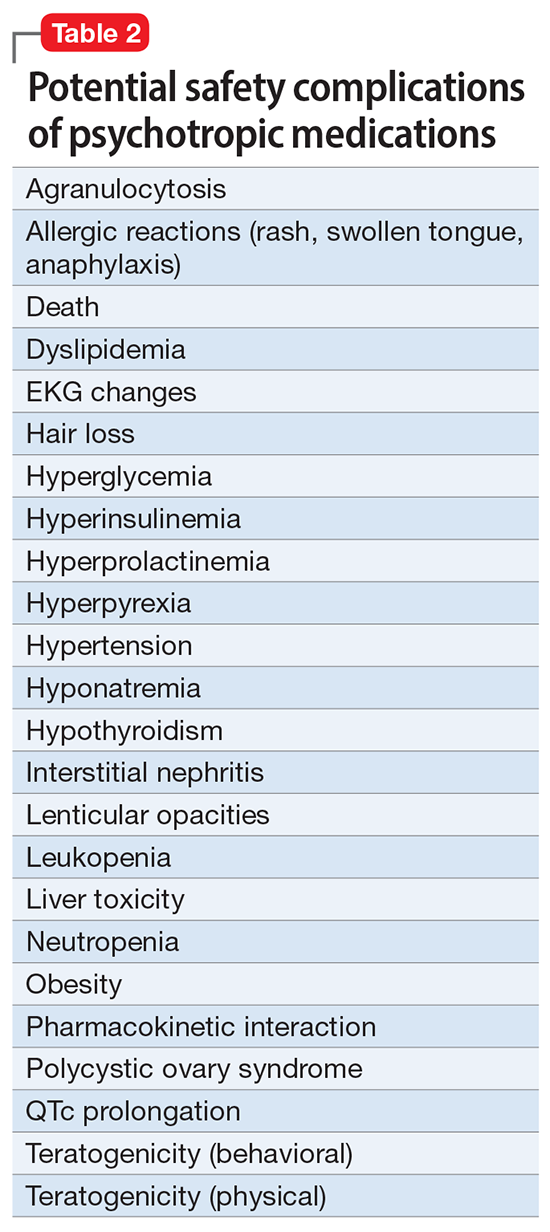
Neuromodulation may very well be the future of psychiatric therapeutics. It targets the brain and avoids the body, thus achieving efficacy with minimal systemic tolerability (ie, patient complaints) (Table 1) or safety (abnormal laboratory test results) issues (Table 2). This sounds ideal, and it is arguably an optimal approach to repairing the brain and healing the mind.
Continue to: ECT is the oldest...
ECT is the oldest neuromodulation technique (developed almost 100 years ago and significantly refined since then). Newer FDA-approved neuromodulation therapies include repetitive transcranial magnetic stimulation (rTMS), which was approved for depression in 2013, obsessive-compulsive disorder (OCD) in 2018, smoking cessation in 2020, and anxious depression in 2021.7 Vagus nerve stimulation (VNS) is used for drug-resistant epilepsy and was later approved for treatment-resistant depression,8,9 but some studies report it can be helpful for fear and anxiety in autism spectrum disorder10 and primary insomnia.11
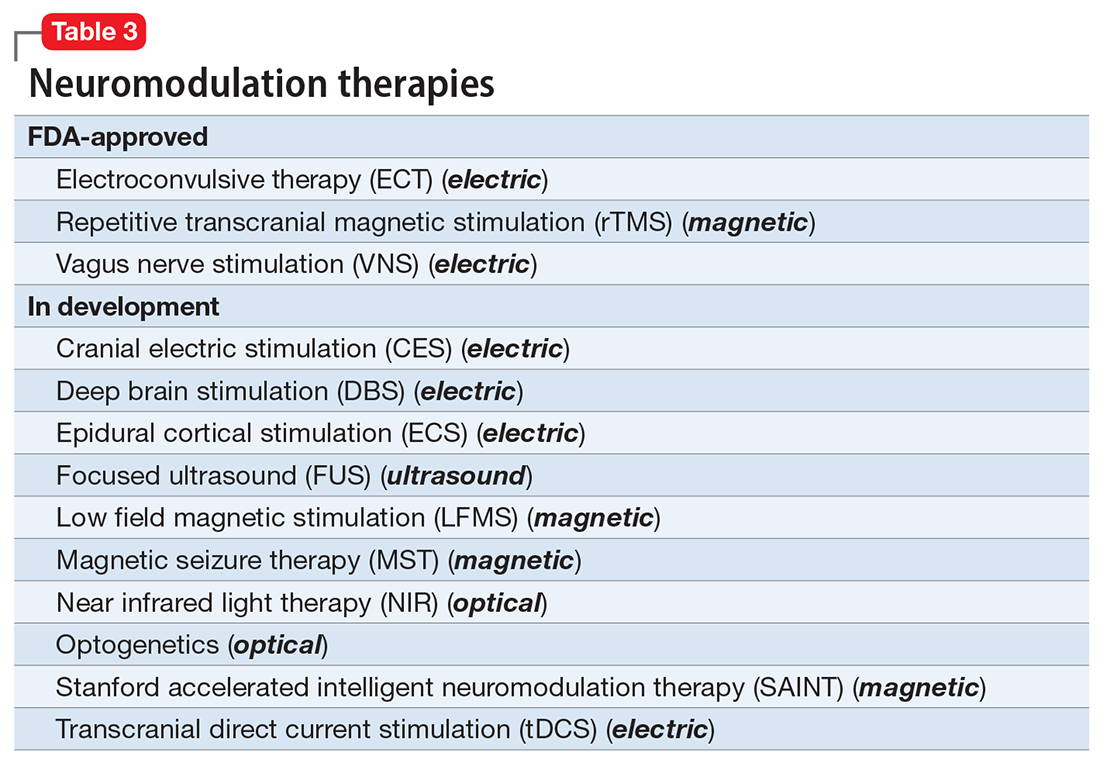
There are many other neuromodulation therapies in development12 that have not yet been FDA approved (Table 3). The most prominent of these is deep brain stimulation (DBS), which is approved for Parkinson disease and has been reported in many studies to improve treatment-resistant depression13,14 and OCD.15 Another promising neuromodulation therapy is transcranial direct current stimulation (tDCS), which has promising results in schizophrenia16 similar to ECT’s effects in treatment-resistant schizophrenia.17
A particularly exciting neuromodulation approach published by Stanford University researchers is Stanford accelerated intelligent neuromodulation therapy (SAINT),18 which uses intermittent theta-burst stimulation (iTBS) daily for 5 days, targeted at the subgenual anterior cingulate gyrus (Brodman area 25). Remarkably, efficacy was rapid, with a very high remission rate (absence of symptoms) in approximately 90% of patients with severe depression.18
The future is bright for neuromodulation therapies, and for a good reason. Why send a chemical agent to every cell and organ in the body when the brain can be targeted directly? As psychiatric neuroscience advances to a point where we can localize the abnormal neurologic circuit in a specific brain region for each psychiatric disorder, it will be possible to treat almost all psychiatric disorders without burdening patients with the intolerable symptoms or safety adverse effects of medications. Psychiatrists should modulate their perspective about the future of psychiatric treatments. And finally, I propose that psychotherapy should be reclassified as a “verbal neuromodulation” technique.
1. Nasrallah HA. Repositioning psychotherapy as a neurobiological intervention. Current Psychiatry. 2013;12(12):18-19.
2. Nasrallah HA. Bipolar disorder: clinical questions beg for answers. Current Psychiatry. 2006;5(12):11-12.
3. UK ECT Review Group. Efficacy and safety of electroconvulsive therapy in depressive disorders: a systematic review and meta-analysis. Lancet. 2003;361(9360):799-808.
4. Rhee TG, Shim SR, Forester BP, et al. Efficacy and safety of ketamine vs electroconvulsive therapy among patients with major depressive episode: a systematic review and meta-analysis. JAMA Psychiatry. 2022:e223352. doi:10.1001/jamapsychiatry.2022.3352
5. Nuninga JO, Mandl RCW, Boks MP, et al. Volume increase in the dentate gyrus after electroconvulsive therapy in depressed patients as measured with 7T. Mol Psychiatry. 2020;25(7):1559-1568.
6. Joshi SH, Espinoza RT, Pirnia T, et al. Structural plasticity of the hippocampus and amygdala induced by electroconvulsive therapy in major depression. Biol Psychiatry. 2016;79(4):282-292.
7. Rhee TG, Olfson M, Nierenberg AA, et al. 20-year trends in the pharmacologic treatment of bipolar disorder by psychiatrists in outpatient care settings. Am J Psychiatry. 2020;177(8):706-715.
8. Hilz MJ. Transcutaneous vagus nerve stimulation - a brief introduction and overview. Auton Neurosci. 2022;243:103038. doi:10.1016/j.autneu.2022.103038
9. Pigato G, Rosson S, Bresolin N, et al. Vagus nerve stimulation in treatment-resistant depression: a case series of long-term follow-up. J ECT. 2022. doi:10.1097/YCT.0000000000000869
10. Shivaswamy T, Souza RR, Engineer CT, et al. Vagus nerve stimulation as a treatment for fear and anxiety in individuals with autism spectrum disorder. J Psychiatr Brain Sci. 2022;7(4):e220007. doi:10.20900/jpbs.20220007
11. Wu Y, Song L, Wang X, et al. Transcutaneous vagus nerve stimulation could improve the effective rate on the quality of sleep in the treatment of primary insomnia: a randomized control trial. Brain Sci. 2022;12(10):1296. doi:10.3390/brainsci12101296
12. Rosa MA, Lisanby SH. Somatic treatments for mood disorders. Neuropsychopharmacology. 2012;37(1):102-116.
13. Mayberg HS, Lozano AM, Voon V, et al. Deep brain stimulation for treatment-resistant depression. Neuron. 2005;45(5):651-660.
14. Choi KS, Mayberg H. Connectomic DBS in major depression. In: Horn A, ed. Connectomic Deep Brain Stimulation. Academic Press; 2022:433-447.
15. Cruz S, Gutiérrez-Rojas L, González-Domenech P, et al. Deep brain stimulation in obsessive-compulsive disorder: results from meta-analysis. Psychiatry Res. 2022;317:114869. doi:10.1016/j.psychres.2022.114869
16. Lisoni J, Baldacci G, Nibbio G, et al. Effects of bilateral, bipolar-nonbalanced, frontal transcranial direct current stimulation (tDCS) on negative symptoms and neurocognition in a sample of patients living with schizophrenia: results of a randomized double-blind sham-controlled trial. J Psychiatr Res. 2022;155:430-442.
17. Sinclair DJ, Zhao S, Qi F, et al. Electroconvulsive therapy for treatment-resistant schizophrenia. Cochrane Database Syst Rev. 2019;3(3):CD011847. doi:10.1002/14651858.CD011847.pub2
18. Cole EJ, Stimpson KH, Bentzley BS, et al. Stanford accelerated intelligent neuromodulation therapy for treatment-resistant depression. Am J Psychiatry. 2020;177(8):716-726.
Pharmacotherapy for psychiatric disorders is a mixed blessing. The advent of psychotropic medications since the 1950s (antipsychotics, antidepressants, anxiolytics, mood stabilizers) has revolutionized the treatment of serious psychiatric brain disorders, allowing certain patients to be discharged to the community after a lifetime of institutionalization.

However, like all medications, psychotropic agents are often associated with various potentially intolerable symptoms (Table 1) or safety complications (Table 2) because they interact with every organ in the body besides their intended target, the brain, and its neurochemical circuitry.

Imagine if we could treat our psychiatric patients while bypassing the body and achieve response, remission, and ultimately recovery without any systemic adverse effects. Adherence would dramatically improve, our patients’ quality of life would be enhanced, and the overall effectiveness (defined as the complex package of efficacy, safety, and tolerability) would be superior to current pharmacotherapies. This is important because most psychiatric medications must be taken daily for years, even a lifetime, to avoid a relapse of the illness. Psychiatrists frequently must manage adverse effects or switch the patient to a different medication if a tolerability or safety issue emerges, which is very common in psychiatric practice. A significant part of psychopharmacologic management includes ordering various laboratory tests to monitor adverse reactions in major organs, especially the liver, kidney, and heart. Additionally, psychiatric physicians must be constantly cognizant of medications prescribed by other clinicians for comorbid medical conditions to successfully navigate the turbulent seas of pharmacokinetic interactions.
I am sure you have noticed that whenever you watch a direct-to-consumer commercial for any medication, 90% of the advertisement is a background voice listing the various tolerability and safety complications of the medication as required by the FDA. Interestingly, these ads frequently contain colorful scenery and joyful clips, which I suspect are cleverly designed to distract the audience from focusing on the list of adverse effects.
Benefits of nonpharmacologic treatments
No wonder I am a fan of psychotherapy, a well-established psychiatric treatment modality that completely avoids body tissues. It directly targets the brain without needlessly interacting with any other organ. Psychotherapy’s many benefits (improving insight, enhancing adherence, improving self-esteem, reducing risky behaviors, guiding stress management and coping skills, modifying unhealthy beliefs, and ultimately relieving symptoms such as anxiety and depression) are achieved without any somatic adverse effects! Psychotherapy has also been shown to induce neuroplasticity and reduce inflammatory biomarkers.1 Unlike FDA-approved medications, psychotherapy does not include a “package insert,” 10 to 20 pages (in small print) that mostly focus on warnings, precautions, and sundry physical adverse effects. Even the dosing of psychotherapy is left entirely up to the treating clinician!
Although I have had many gratifying results with pharmacotherapy in my practice, especially in combination with psychotherapy,2 I also have observed excellent outcomes with nonpharmacologic approaches, especially neuromodulation therapies. The best antidepressant I have ever used since my residency training days is electroconvulsive therapy (ECT). My experience is consistent with a large meta-analysis3showing a huge effect size (Cohen d = .91) in contrast to the usual effect size of .3 to .5 for standard antidepressants (except IV ketamine). A recent study showed ECT is even better than the vaunted rapid-acting ketamine,4 which is further evidence of its remarkable efficacy in depression. Neuroimaging studies report that ECT rapidly increases the volume of the hippocampus,5,6 which shrinks in size in patients with unipolar or bipolar depression.
Neuromodulation may very well be the future of psychiatric therapeutics. It targets the brain and avoids the body, thus achieving efficacy with minimal systemic tolerability (ie, patient complaints) (Table 1) or safety (abnormal laboratory test results) issues (Table 2). This sounds ideal, and it is arguably an optimal approach to repairing the brain and healing the mind.
Continue to: ECT is the oldest...
ECT is the oldest neuromodulation technique (developed almost 100 years ago and significantly refined since then). Newer FDA-approved neuromodulation therapies include repetitive transcranial magnetic stimulation (rTMS), which was approved for depression in 2013, obsessive-compulsive disorder (OCD) in 2018, smoking cessation in 2020, and anxious depression in 2021.7 Vagus nerve stimulation (VNS) is used for drug-resistant epilepsy and was later approved for treatment-resistant depression,8,9 but some studies report it can be helpful for fear and anxiety in autism spectrum disorder10 and primary insomnia.11
There are many other neuromodulation therapies in development12 that have not yet been FDA approved (Table 3). The most prominent of these is deep brain stimulation (DBS), which is approved for Parkinson disease and has been reported in many studies to improve treatment-resistant depression13,14 and OCD.15 Another promising neuromodulation therapy is transcranial direct current stimulation (tDCS), which has promising results in schizophrenia16 similar to ECT’s effects in treatment-resistant schizophrenia.17

A particularly exciting neuromodulation approach published by Stanford University researchers is Stanford accelerated intelligent neuromodulation therapy (SAINT),18 which uses intermittent theta-burst stimulation (iTBS) daily for 5 days, targeted at the subgenual anterior cingulate gyrus (Brodman area 25). Remarkably, efficacy was rapid, with a very high remission rate (absence of symptoms) in approximately 90% of patients with severe depression.18
The future is bright for neuromodulation therapies, and for a good reason. Why send a chemical agent to every cell and organ in the body when the brain can be targeted directly? As psychiatric neuroscience advances to a point where we can localize the abnormal neurologic circuit in a specific brain region for each psychiatric disorder, it will be possible to treat almost all psychiatric disorders without burdening patients with the intolerable symptoms or safety adverse effects of medications. Psychiatrists should modulate their perspective about the future of psychiatric treatments. And finally, I propose that psychotherapy should be reclassified as a “verbal neuromodulation” technique.
Pharmacotherapy for psychiatric disorders is a mixed blessing. The advent of psychotropic medications since the 1950s (antipsychotics, antidepressants, anxiolytics, mood stabilizers) has revolutionized the treatment of serious psychiatric brain disorders, allowing certain patients to be discharged to the community after a lifetime of institutionalization.

However, like all medications, psychotropic agents are often associated with various potentially intolerable symptoms (Table 1) or safety complications (Table 2) because they interact with every organ in the body besides their intended target, the brain, and its neurochemical circuitry.

Imagine if we could treat our psychiatric patients while bypassing the body and achieve response, remission, and ultimately recovery without any systemic adverse effects. Adherence would dramatically improve, our patients’ quality of life would be enhanced, and the overall effectiveness (defined as the complex package of efficacy, safety, and tolerability) would be superior to current pharmacotherapies. This is important because most psychiatric medications must be taken daily for years, even a lifetime, to avoid a relapse of the illness. Psychiatrists frequently must manage adverse effects or switch the patient to a different medication if a tolerability or safety issue emerges, which is very common in psychiatric practice. A significant part of psychopharmacologic management includes ordering various laboratory tests to monitor adverse reactions in major organs, especially the liver, kidney, and heart. Additionally, psychiatric physicians must be constantly cognizant of medications prescribed by other clinicians for comorbid medical conditions to successfully navigate the turbulent seas of pharmacokinetic interactions.
I am sure you have noticed that whenever you watch a direct-to-consumer commercial for any medication, 90% of the advertisement is a background voice listing the various tolerability and safety complications of the medication as required by the FDA. Interestingly, these ads frequently contain colorful scenery and joyful clips, which I suspect are cleverly designed to distract the audience from focusing on the list of adverse effects.
Benefits of nonpharmacologic treatments
No wonder I am a fan of psychotherapy, a well-established psychiatric treatment modality that completely avoids body tissues. It directly targets the brain without needlessly interacting with any other organ. Psychotherapy’s many benefits (improving insight, enhancing adherence, improving self-esteem, reducing risky behaviors, guiding stress management and coping skills, modifying unhealthy beliefs, and ultimately relieving symptoms such as anxiety and depression) are achieved without any somatic adverse effects! Psychotherapy has also been shown to induce neuroplasticity and reduce inflammatory biomarkers.1 Unlike FDA-approved medications, psychotherapy does not include a “package insert,” 10 to 20 pages (in small print) that mostly focus on warnings, precautions, and sundry physical adverse effects. Even the dosing of psychotherapy is left entirely up to the treating clinician!
Although I have had many gratifying results with pharmacotherapy in my practice, especially in combination with psychotherapy,2 I also have observed excellent outcomes with nonpharmacologic approaches, especially neuromodulation therapies. The best antidepressant I have ever used since my residency training days is electroconvulsive therapy (ECT). My experience is consistent with a large meta-analysis3showing a huge effect size (Cohen d = .91) in contrast to the usual effect size of .3 to .5 for standard antidepressants (except IV ketamine). A recent study showed ECT is even better than the vaunted rapid-acting ketamine,4 which is further evidence of its remarkable efficacy in depression. Neuroimaging studies report that ECT rapidly increases the volume of the hippocampus,5,6 which shrinks in size in patients with unipolar or bipolar depression.
Neuromodulation may very well be the future of psychiatric therapeutics. It targets the brain and avoids the body, thus achieving efficacy with minimal systemic tolerability (ie, patient complaints) (Table 1) or safety (abnormal laboratory test results) issues (Table 2). This sounds ideal, and it is arguably an optimal approach to repairing the brain and healing the mind.
Continue to: ECT is the oldest...
ECT is the oldest neuromodulation technique (developed almost 100 years ago and significantly refined since then). Newer FDA-approved neuromodulation therapies include repetitive transcranial magnetic stimulation (rTMS), which was approved for depression in 2013, obsessive-compulsive disorder (OCD) in 2018, smoking cessation in 2020, and anxious depression in 2021.7 Vagus nerve stimulation (VNS) is used for drug-resistant epilepsy and was later approved for treatment-resistant depression,8,9 but some studies report it can be helpful for fear and anxiety in autism spectrum disorder10 and primary insomnia.11
There are many other neuromodulation therapies in development12 that have not yet been FDA approved (Table 3). The most prominent of these is deep brain stimulation (DBS), which is approved for Parkinson disease and has been reported in many studies to improve treatment-resistant depression13,14 and OCD.15 Another promising neuromodulation therapy is transcranial direct current stimulation (tDCS), which has promising results in schizophrenia16 similar to ECT’s effects in treatment-resistant schizophrenia.17

A particularly exciting neuromodulation approach published by Stanford University researchers is Stanford accelerated intelligent neuromodulation therapy (SAINT),18 which uses intermittent theta-burst stimulation (iTBS) daily for 5 days, targeted at the subgenual anterior cingulate gyrus (Brodman area 25). Remarkably, efficacy was rapid, with a very high remission rate (absence of symptoms) in approximately 90% of patients with severe depression.18
The future is bright for neuromodulation therapies, and for a good reason. Why send a chemical agent to every cell and organ in the body when the brain can be targeted directly? As psychiatric neuroscience advances to a point where we can localize the abnormal neurologic circuit in a specific brain region for each psychiatric disorder, it will be possible to treat almost all psychiatric disorders without burdening patients with the intolerable symptoms or safety adverse effects of medications. Psychiatrists should modulate their perspective about the future of psychiatric treatments. And finally, I propose that psychotherapy should be reclassified as a “verbal neuromodulation” technique.
1. Nasrallah HA. Repositioning psychotherapy as a neurobiological intervention. Current Psychiatry. 2013;12(12):18-19.
2. Nasrallah HA. Bipolar disorder: clinical questions beg for answers. Current Psychiatry. 2006;5(12):11-12.
3. UK ECT Review Group. Efficacy and safety of electroconvulsive therapy in depressive disorders: a systematic review and meta-analysis. Lancet. 2003;361(9360):799-808.
4. Rhee TG, Shim SR, Forester BP, et al. Efficacy and safety of ketamine vs electroconvulsive therapy among patients with major depressive episode: a systematic review and meta-analysis. JAMA Psychiatry. 2022:e223352. doi:10.1001/jamapsychiatry.2022.3352
5. Nuninga JO, Mandl RCW, Boks MP, et al. Volume increase in the dentate gyrus after electroconvulsive therapy in depressed patients as measured with 7T. Mol Psychiatry. 2020;25(7):1559-1568.
6. Joshi SH, Espinoza RT, Pirnia T, et al. Structural plasticity of the hippocampus and amygdala induced by electroconvulsive therapy in major depression. Biol Psychiatry. 2016;79(4):282-292.
7. Rhee TG, Olfson M, Nierenberg AA, et al. 20-year trends in the pharmacologic treatment of bipolar disorder by psychiatrists in outpatient care settings. Am J Psychiatry. 2020;177(8):706-715.
8. Hilz MJ. Transcutaneous vagus nerve stimulation - a brief introduction and overview. Auton Neurosci. 2022;243:103038. doi:10.1016/j.autneu.2022.103038
9. Pigato G, Rosson S, Bresolin N, et al. Vagus nerve stimulation in treatment-resistant depression: a case series of long-term follow-up. J ECT. 2022. doi:10.1097/YCT.0000000000000869
10. Shivaswamy T, Souza RR, Engineer CT, et al. Vagus nerve stimulation as a treatment for fear and anxiety in individuals with autism spectrum disorder. J Psychiatr Brain Sci. 2022;7(4):e220007. doi:10.20900/jpbs.20220007
11. Wu Y, Song L, Wang X, et al. Transcutaneous vagus nerve stimulation could improve the effective rate on the quality of sleep in the treatment of primary insomnia: a randomized control trial. Brain Sci. 2022;12(10):1296. doi:10.3390/brainsci12101296
12. Rosa MA, Lisanby SH. Somatic treatments for mood disorders. Neuropsychopharmacology. 2012;37(1):102-116.
13. Mayberg HS, Lozano AM, Voon V, et al. Deep brain stimulation for treatment-resistant depression. Neuron. 2005;45(5):651-660.
14. Choi KS, Mayberg H. Connectomic DBS in major depression. In: Horn A, ed. Connectomic Deep Brain Stimulation. Academic Press; 2022:433-447.
15. Cruz S, Gutiérrez-Rojas L, González-Domenech P, et al. Deep brain stimulation in obsessive-compulsive disorder: results from meta-analysis. Psychiatry Res. 2022;317:114869. doi:10.1016/j.psychres.2022.114869
16. Lisoni J, Baldacci G, Nibbio G, et al. Effects of bilateral, bipolar-nonbalanced, frontal transcranial direct current stimulation (tDCS) on negative symptoms and neurocognition in a sample of patients living with schizophrenia: results of a randomized double-blind sham-controlled trial. J Psychiatr Res. 2022;155:430-442.
17. Sinclair DJ, Zhao S, Qi F, et al. Electroconvulsive therapy for treatment-resistant schizophrenia. Cochrane Database Syst Rev. 2019;3(3):CD011847. doi:10.1002/14651858.CD011847.pub2
18. Cole EJ, Stimpson KH, Bentzley BS, et al. Stanford accelerated intelligent neuromodulation therapy for treatment-resistant depression. Am J Psychiatry. 2020;177(8):716-726.
1. Nasrallah HA. Repositioning psychotherapy as a neurobiological intervention. Current Psychiatry. 2013;12(12):18-19.
2. Nasrallah HA. Bipolar disorder: clinical questions beg for answers. Current Psychiatry. 2006;5(12):11-12.
3. UK ECT Review Group. Efficacy and safety of electroconvulsive therapy in depressive disorders: a systematic review and meta-analysis. Lancet. 2003;361(9360):799-808.
4. Rhee TG, Shim SR, Forester BP, et al. Efficacy and safety of ketamine vs electroconvulsive therapy among patients with major depressive episode: a systematic review and meta-analysis. JAMA Psychiatry. 2022:e223352. doi:10.1001/jamapsychiatry.2022.3352
5. Nuninga JO, Mandl RCW, Boks MP, et al. Volume increase in the dentate gyrus after electroconvulsive therapy in depressed patients as measured with 7T. Mol Psychiatry. 2020;25(7):1559-1568.
6. Joshi SH, Espinoza RT, Pirnia T, et al. Structural plasticity of the hippocampus and amygdala induced by electroconvulsive therapy in major depression. Biol Psychiatry. 2016;79(4):282-292.
7. Rhee TG, Olfson M, Nierenberg AA, et al. 20-year trends in the pharmacologic treatment of bipolar disorder by psychiatrists in outpatient care settings. Am J Psychiatry. 2020;177(8):706-715.
8. Hilz MJ. Transcutaneous vagus nerve stimulation - a brief introduction and overview. Auton Neurosci. 2022;243:103038. doi:10.1016/j.autneu.2022.103038
9. Pigato G, Rosson S, Bresolin N, et al. Vagus nerve stimulation in treatment-resistant depression: a case series of long-term follow-up. J ECT. 2022. doi:10.1097/YCT.0000000000000869
10. Shivaswamy T, Souza RR, Engineer CT, et al. Vagus nerve stimulation as a treatment for fear and anxiety in individuals with autism spectrum disorder. J Psychiatr Brain Sci. 2022;7(4):e220007. doi:10.20900/jpbs.20220007
11. Wu Y, Song L, Wang X, et al. Transcutaneous vagus nerve stimulation could improve the effective rate on the quality of sleep in the treatment of primary insomnia: a randomized control trial. Brain Sci. 2022;12(10):1296. doi:10.3390/brainsci12101296
12. Rosa MA, Lisanby SH. Somatic treatments for mood disorders. Neuropsychopharmacology. 2012;37(1):102-116.
13. Mayberg HS, Lozano AM, Voon V, et al. Deep brain stimulation for treatment-resistant depression. Neuron. 2005;45(5):651-660.
14. Choi KS, Mayberg H. Connectomic DBS in major depression. In: Horn A, ed. Connectomic Deep Brain Stimulation. Academic Press; 2022:433-447.
15. Cruz S, Gutiérrez-Rojas L, González-Domenech P, et al. Deep brain stimulation in obsessive-compulsive disorder: results from meta-analysis. Psychiatry Res. 2022;317:114869. doi:10.1016/j.psychres.2022.114869
16. Lisoni J, Baldacci G, Nibbio G, et al. Effects of bilateral, bipolar-nonbalanced, frontal transcranial direct current stimulation (tDCS) on negative symptoms and neurocognition in a sample of patients living with schizophrenia: results of a randomized double-blind sham-controlled trial. J Psychiatr Res. 2022;155:430-442.
17. Sinclair DJ, Zhao S, Qi F, et al. Electroconvulsive therapy for treatment-resistant schizophrenia. Cochrane Database Syst Rev. 2019;3(3):CD011847. doi:10.1002/14651858.CD011847.pub2
18. Cole EJ, Stimpson KH, Bentzley BS, et al. Stanford accelerated intelligent neuromodulation therapy for treatment-resistant depression. Am J Psychiatry. 2020;177(8):716-726.
Should every scheduled cesarean birth use an Enhanced Recovery after Surgery (ERAS) pathway?

Cesarean birth is one of the most common major surgical procedures performed in developed countries1 with over 1,170,000 cesarean births in the United States in 2021.2 Many surgeons and anesthesiologists believe that Enhanced Recovery after Surgery (ERAS) pathways improve surgical outcomes.3,4 Important goals of ERAS include setting patient expectations for the surgical procedure, accelerating patient recovery to full function, and minimizing perioperative complications such as severe nausea, aspiration, surgical site infection, wound complications, and perioperative anemia. The ERAS Society in 20185-7 and the Society for Obstetric Anesthesia and Perinatology (SOAP) in 20218 proposed ERAS pathways for cesarean birth. Both societies recommended that obstetric units consider adopting an ERAS pathway compatible with local clinical resources. In addition, the American College of Obstetricians and Gynecologists (ACOG) has provided guidance for implementing ERAS pathways for gynecologic surgery.9 The consistent use of standardized protocols to improve surgical care in obstetrics should lead to a reduction in care variation and improve health equity outcomes.
The clinical interventions recommended for ERAS cesarean birth occur sequentially in the preoperative, intraoperative, and postoperative phases of care. The recommendations associated with each of these phases are reviewed below. It is important to note that each obstetric unit should use a multidisciplinary process to develop an ERAS pathway that best supports local practice given clinician preferences, patient characteristics, and resource availability.
Preoperative components of ERAS
Standardized patient education (SPE). SPE is an important component of ERAS, although evidence to support the recommendation is limited. At a minimum a written handout describing steps in the cesarean birth process, or a patient-education video should be part of patient education. The University of Michigan Medical Center has produced a 3-minute video for patients explaining ERAS cesarean birth.10 The University of Maryland Medical Center has produced a 2.5-minute video in English and Spanish, explaining ERAS cesarean birth for patients.11 Some surgeons place a telephone call to patients the evening before surgery to help orient the patient to ERAS cesarean birth.
Breastfeeding education. An important goal of obstetric care is to optimize the rate of exclusive breastfeeding at birth. Breastfeeding education, including a commitment to support the initiation of breastfeeding within 1 hour of birth, may enhance the rate of exclusive breastfeeding. There are numerous videos available for patients about breastfeeding after cesarean birth (as an example, see: https://www.youtube.com/watch?v=9iOGn85NdTg).
Limit fasting. In the past, surgical guidelines recommended fasting after midnight prior to surgery. The ERAS Society recommends that patients should be encouraged to drink clear fluids up to 2 hours before surgery and may have a light meal up to 6 hours before surgery (Part 1).
Carbohydrate loading. Surgery causes a metabolic stress that is increased by fasting. Carbohydrate loading prior to surgery reduces the magnitude of the catabolic state caused by the combination of surgery and fasting.12 SOAP and the ERAS Society recommend oral carbohydrate fluid supplementation 2 hours before surgery for nondiabetic patients. SOAP suggests 32 oz of Gatorade or 16 oz of clear apple juice as options for carbohydrate loading. For diabetic patients, the carbohydrate load can be omitted. In fasting pregnant patients at term, gastric emptying was near complete 2 hours after consumption of 400 mL of a carbohydrate drink.13 In one study, consumption of 400 mL of a carbohydrate drink 2 hours before cesarean resulted in a 7% increase in the newborn blood glucose level at 20 min after delivery.14
Minimize preoperative anemia. Approximately 50% of pregnant women are iron deficient and approximately 10% are anemic in the third trimester.15,16 Cesarean birth is associated with significant blood loss necessitating the need to optimize red blood cell mass before surgery. Measuring ferritin to identify patients with iron deficiency and aggressive iron replacement, including intravenous iron if necessary, will reduce the prevalence of anemia prior to cesarean birth.17 Another cause of anemia in pregnancy is vitamin B12 (cobalamin) deficiency. Low vitamin B12 is especially common in pregnant patients who have previously had bariatric surgery. One study reported that, of 113 pregnant patients who were, on average, 3 years from a bariatric surgery procedure, 12% had vitamin B12 circulating levels < 130 pg/mL.18 Among pregnant patients who are anemic, and do not have a hemoglobinopathy, measuring ferritin, folic acid, and vitamin B12 will help identify the cause of anemia and guide treatment.19
Optimize preoperative physical condition. Improving healthy behaviors and reducing unhealthy behaviors preoperatively may enhance patient recovery to full function. In the weeks before scheduled cesarean birth, cessation of the use of tobacco products, optimizing activity and improving diet quality, including increasing protein intake, may best prepare patients for the metabolic stress of surgery.
Continue to: Intraoperative components of ERAS...
Intraoperative components of ERAS
Reduce the risk of surgical site infection (SSI) and wound complications. Bundles that include antibiotics, chlorhexidine (or an alternative antibacterial soap) and clippers have been shown to reduce SSI.20 Routine administration of preoperative antibiotics is a consensus recommendation and there is high adherence with this recommendation in the United States. Chlorhexidine-alcohol is the preferred solution for skin preparation. Vaginal preparation with povidine-iodine or chlorhexidine may be considered.6
Surgical technique. Blunt extension of a transverse hysterotomy may reduce blood loss. Closure of the hysterotomy incision in 2 layers is recommended to reduce uterine scar dehiscence in a subsequent pregnancy. If the patient has ≥2 cm of subcutaneous tissue, this layer should be approximated with sutures. Skin closure should be with subcuticular suture.6
Optimize uterotonic administration. Routine use of uterotonics reduces the risk of blood loss, transfusion, and postoperative anemia. There is high adherence with the use of uterotonic administration after birth in the United States.6,8
Ensure normothermia. Many patients become hypothermic during a cesarean birth. Active warming of the patient with an in-line IV fluid warmer and forced air warming over the patient’s body can reduce the risk of hypothermia.8
Initiate multimodal anesthesia. Anesthesiologists often use intrathecal or epidural morphine to enhance analgesia. Ketorolac administration prior to completion of the cesarean procedure and perioperative administration of acetaminophen may reduce postoperative pain.8 The use of preoperative antiemetics will reduce intraoperative and postoperative nausea and vomiting.
Initiate VTE prophylaxis. Pneumatic compression stockings are recommended. Anticoagulation should not be routinely used for VTE prophylaxis.6
Postoperative components of ERAS
Patient education to prepare for discharge home when ready. Patient education focused on home when ready is important in preparing the patient for discharge home.7 Completion of required newborn testing, lactation education, and contraception planning plus coordination of newborn pediatric follow-up is necessary before discharge.
Support early return of bowel function. Early return of bowel function is best supported by a multimodal approach including initiation of clear fluid intake immediately following surgery, encouraging consumption of a regular diet within 27 to 4 hours8 following surgery. Gum chewing for at least 5 minutes 3 times daily accelerates return of bowel function.8 In a meta-analysis of 10 randomized studies examining the effect of gum chewing after cesarean, the investigators reported that gum chewing shortened the time to passage of flatus and defecation.21
Early ambulation.
Sequentially advanced activity, starting with sitting on the edge of the bed, sitting in a chair, and ambulation within 8 hours of surgery, is recommended to facilitate faster recovery, reduce rates of complications, and enable transition to home.8
Early removal of the urinary catheter. It is recommended that the urinary catheter be removed within 12 hours after cesarean birth.8 Early removal of the urinary catheter increases patient mobility and reduces the length of hospitalization. Early removal of the urinary catheter may be associated with postoperative urinary retention and recatheterization in a small number of patients.
Prescribe routinely scheduled acetaminophen, nonsteroidal anti-inflammatory drugs (NSAIDs) and ketorolac. A key component of ERAS cesarean birth is the standardized administration of nonopioid pain medicines, alternating doses of acetaminophen and an NSAID. ERAS cesarean birth is likely to result in a reduction in inpatient and postdischarge opioid use.22-24
VTE prophylaxis. Pneumatic compression stockings are recommended. Anticoagulation should not be routinely used for VTE prophylaxis.8
Auditing and reporting adherence with components of ERAS
In clinical practice there may be a gap between a clinician’s subjective perception of their performance and an independent audit of their clinical performance. ERAS pathways should be implemented with a commitment to performing audits and providing quantitative feedback to clinicians. Consistent use of measurement, feedback, and coaching can improve performance and reduce variation among individual clinicians. As an example, in one study of the use of a surgical safety checklist, 99% of the surgeons reported that they routinely used a surgical safety checklist, but the audit showed that the checklist was used in only 60% of cases.25 Gaps between self-reported performance and audited performance are common in clinical practice. Audits with feedback are critical to improving adherence with the components of an ERAS pathway.
Three independent systematic reviews and meta-analyses report that ERAS pathways reduce hospital length of stay without increasing the readmission rate.26-28 One meta-analysis reported that ERAS may also reduce time to first mobilization and result in earlier removal of the urinary catheter.26 ERAS pathways also may reduce postoperative complications, lower pain scores, and decrease opioid use.27 The general consensus among quality and safety experts is that reducing variation through standardization of pathways is generally associated with improved quality and enhanced safety. ERAS pathways have been widely accepted in multiple surgical fields. ERAS pathways should become the standard for performing cesarean procedures.●
1. Molina G, Weiser RG, Lipsitz SR, et al. Relationship between cesarean delivery rate and maternal and neonatal mortality. JAMA. 2015;314:2263-2270.
2. Hamilton BE, Martin JA, Osterman MJK. Births: provisional data for 2021. Vital Statistics Release; No. 20. Hyattsville, MD: National Center for Health Statistics. May 2022. https://www.cdc.gov/nchs/data/vsrr/vsrr020.pdf.
3. Berian JR, Ban KA, Liu JB, et al. Adherence to enhanced recovery protocols in NSQIP and association with colectomy outcomes. Ann Surg. 2019;486-493.
4. Ljungqvist O, Scott M, Fearon KC. Enhanced recovery after surgery: a review. JAMA Surg. 2017;152:292-298.
5. Wilson RD, Caughey AB, Wood SL, et al. Guidelines for antenatal and preoperative care in cesarean delivery: Enhanced Recovery after Surgery Society recommendations (Part 1). Am J Obstet Gynecol. 2018;219:523.e1-523.e15.
6. Caughey AB, Wood SL, Macones GA, et al Guidelines for intraoperative care in cesarean delivery: Enhanced Recovery after Surgery Society recommendations (Part 2). Am J Obstet Gynecol. 2018;219:533-544.
7. Macones GA, Caughey AB, Wood SL, et al. Guidelines for postoperative care in cesarean delivery: Enhanced Recovery after Surgery Society recommendations (Part 3). Am J Obstet Gynecol. 2019;221:247.e1-247.e9.
8. Bollag L, Lim G, Sultan P, et al. Society for Obstetric Anesthesia and Perinatology: Consensus statement and recommendations for enhanced recovery after cesarean. Anesth Analg. 2021;132:1362-1377.
9. Perioperative pathways: enhanced recovery after surgery. ACOG Committee Opinion No 750. American College of Obstetricians and Gynecologists. Obstet Gynecol. 2018;132:e120-130.
10. University of Michigan. ERAS: A patient education video. https://www.youtube.com/watch?v=CoFtgdluBc0. Accessed October 24, 2022.
11. University of Maryland. ERAS. https://www.umms.org/ummc/health-services/womens-health/ostetrics-gynecology/pregnancy-childbirth/labor-delivery/enhanced-recovery-after-cesarean. Accessed October 24, 2022.
12. Bilku DK, Dennison AR, Hall TC, et al. Role of preoperative carbohydrate loading: a systematic review. Ann R Coll Surg Engl. 2014;96:15-22.
13. Popivanov P, Irwin R, Walsh M, et al. Gastric emptying of carbohydrate drinks in term parturients before elective caesarean surgery: an observational study. Int J Obstet Anesth. 2020;41:29-34.
14. He Y, Liu C, Han Y, et al. The impact of carbohydrate-rich supplement taken two hours before caesarean delivery on maternal and neonatal perioperative outcomes- a randomized clinical trial. BMC Pregnancy Childbirth. 2021;21:682.
15. Auerbach M, Abernathy J, Juul S, et al. Prevalence of iron deficiency in first trimester, nonanemic pregnant women. J Matern Fetal Neonatal Med. 2021;34:1002-1005.
16. Mei Z, Cogswell ME, Looker AC, et al. Assessment of iron status in US pregnant women from the National Health and Nutrition Examination Survey (NHANES), 1996-2006. Am J Clin Nutr. 2011;93:1312-1320.
17. Nour N, Barbieri RL. Optimize detection and treatment of iron deficiency in pregnancy. OBG Manag. 2022;34:9-11.
18. Mead NC, Sakkatos P, Sakellaropoulos GC, et al. Pregnancy outcomes and nutritional indices after 3 types of bariatric surgery performed at a single institution. Surg Obes Relat Dis. 2014;10:1166-1173.
19. Achebe MM, Gafter-Gvili A. How I treat anemia in pregnancy: iron, cobalamin and folate. Blood. 2017;129:940-949.
20. Carter EB, Temming LA, Fowler S, et al. Evidence-based bundles and cesarean delivery surgical site infections: a systematic review and meta-analysis. Obstet Gynecol. 2017;130:735-746.
21. Wen Z, Shen M, Wu C, et al. Chewing gum for intestinal function recovery after caesarean section: a systematic review and meta-analysis. BMC Pregnancy Childbirth. 2017;17:105.
22. McCoy JA, Gutman S, Hamm RF, et al. The association between implementation of an enhanced recovery after cesarean pathway with standardized discharge prescriptions and opioid use and pain experience after cesarean delivery. Am J Perinatol. 2021;38:1341-1347.
23. Mullman L, Hilden P, Goral J, et al. Improved outcomes with an enhanced recovery approach to cesarean delivery. Obstet Gynecol. 2020;136:685-691.
24. Hedderson M, Lee D, Hunt E, et al. Enhanced recovery after surgery to change process measures and reduce opioid use after cesarean delivery: a quality improvement initiative. Obstet Gynecol. 2019;134:511-519.
25. Sendlhofer G, Lumenta DB, Leitgeb K, et al. The gap between individual perception and compliance: a quantitative follow-up study of the surgical safety checklist application. PLoS One. 2016;11:e0149212.
26. Sultan P, Sharawi N, Blake L, et al. Impact of enhanced recovery after cesarean delivery on maternal outcomes: a systematic review and meta-analysis. Anaesth Crit Care Pain Med. 2021;40:100935.
27. Meng X, Chen K, Yang C, et al. The clinical efficacy and safety of enhanced recovery after surgery for cesarean section: a systematic review and meta-analysis of randomized controlled trials and observational studies. Front Med. 2021;8:694385.
28. Corson E, Hind D, Beever D, et al. Enhanced recovery after elective caesarean: a rapid review of clinical protocols and an umbrella review of systematic reviews. BMC Pregnancy Childbirth. 2017;17:91.

Cesarean birth is one of the most common major surgical procedures performed in developed countries1 with over 1,170,000 cesarean births in the United States in 2021.2 Many surgeons and anesthesiologists believe that Enhanced Recovery after Surgery (ERAS) pathways improve surgical outcomes.3,4 Important goals of ERAS include setting patient expectations for the surgical procedure, accelerating patient recovery to full function, and minimizing perioperative complications such as severe nausea, aspiration, surgical site infection, wound complications, and perioperative anemia. The ERAS Society in 20185-7 and the Society for Obstetric Anesthesia and Perinatology (SOAP) in 20218 proposed ERAS pathways for cesarean birth. Both societies recommended that obstetric units consider adopting an ERAS pathway compatible with local clinical resources. In addition, the American College of Obstetricians and Gynecologists (ACOG) has provided guidance for implementing ERAS pathways for gynecologic surgery.9 The consistent use of standardized protocols to improve surgical care in obstetrics should lead to a reduction in care variation and improve health equity outcomes.
The clinical interventions recommended for ERAS cesarean birth occur sequentially in the preoperative, intraoperative, and postoperative phases of care. The recommendations associated with each of these phases are reviewed below. It is important to note that each obstetric unit should use a multidisciplinary process to develop an ERAS pathway that best supports local practice given clinician preferences, patient characteristics, and resource availability.
Preoperative components of ERAS
Standardized patient education (SPE). SPE is an important component of ERAS, although evidence to support the recommendation is limited. At a minimum a written handout describing steps in the cesarean birth process, or a patient-education video should be part of patient education. The University of Michigan Medical Center has produced a 3-minute video for patients explaining ERAS cesarean birth.10 The University of Maryland Medical Center has produced a 2.5-minute video in English and Spanish, explaining ERAS cesarean birth for patients.11 Some surgeons place a telephone call to patients the evening before surgery to help orient the patient to ERAS cesarean birth.
Breastfeeding education. An important goal of obstetric care is to optimize the rate of exclusive breastfeeding at birth. Breastfeeding education, including a commitment to support the initiation of breastfeeding within 1 hour of birth, may enhance the rate of exclusive breastfeeding. There are numerous videos available for patients about breastfeeding after cesarean birth (as an example, see: https://www.youtube.com/watch?v=9iOGn85NdTg).
Limit fasting. In the past, surgical guidelines recommended fasting after midnight prior to surgery. The ERAS Society recommends that patients should be encouraged to drink clear fluids up to 2 hours before surgery and may have a light meal up to 6 hours before surgery (Part 1).
Carbohydrate loading. Surgery causes a metabolic stress that is increased by fasting. Carbohydrate loading prior to surgery reduces the magnitude of the catabolic state caused by the combination of surgery and fasting.12 SOAP and the ERAS Society recommend oral carbohydrate fluid supplementation 2 hours before surgery for nondiabetic patients. SOAP suggests 32 oz of Gatorade or 16 oz of clear apple juice as options for carbohydrate loading. For diabetic patients, the carbohydrate load can be omitted. In fasting pregnant patients at term, gastric emptying was near complete 2 hours after consumption of 400 mL of a carbohydrate drink.13 In one study, consumption of 400 mL of a carbohydrate drink 2 hours before cesarean resulted in a 7% increase in the newborn blood glucose level at 20 min after delivery.14
Minimize preoperative anemia. Approximately 50% of pregnant women are iron deficient and approximately 10% are anemic in the third trimester.15,16 Cesarean birth is associated with significant blood loss necessitating the need to optimize red blood cell mass before surgery. Measuring ferritin to identify patients with iron deficiency and aggressive iron replacement, including intravenous iron if necessary, will reduce the prevalence of anemia prior to cesarean birth.17 Another cause of anemia in pregnancy is vitamin B12 (cobalamin) deficiency. Low vitamin B12 is especially common in pregnant patients who have previously had bariatric surgery. One study reported that, of 113 pregnant patients who were, on average, 3 years from a bariatric surgery procedure, 12% had vitamin B12 circulating levels < 130 pg/mL.18 Among pregnant patients who are anemic, and do not have a hemoglobinopathy, measuring ferritin, folic acid, and vitamin B12 will help identify the cause of anemia and guide treatment.19
Optimize preoperative physical condition. Improving healthy behaviors and reducing unhealthy behaviors preoperatively may enhance patient recovery to full function. In the weeks before scheduled cesarean birth, cessation of the use of tobacco products, optimizing activity and improving diet quality, including increasing protein intake, may best prepare patients for the metabolic stress of surgery.
Continue to: Intraoperative components of ERAS...
Intraoperative components of ERAS
Reduce the risk of surgical site infection (SSI) and wound complications. Bundles that include antibiotics, chlorhexidine (or an alternative antibacterial soap) and clippers have been shown to reduce SSI.20 Routine administration of preoperative antibiotics is a consensus recommendation and there is high adherence with this recommendation in the United States. Chlorhexidine-alcohol is the preferred solution for skin preparation. Vaginal preparation with povidine-iodine or chlorhexidine may be considered.6
Surgical technique. Blunt extension of a transverse hysterotomy may reduce blood loss. Closure of the hysterotomy incision in 2 layers is recommended to reduce uterine scar dehiscence in a subsequent pregnancy. If the patient has ≥2 cm of subcutaneous tissue, this layer should be approximated with sutures. Skin closure should be with subcuticular suture.6
Optimize uterotonic administration. Routine use of uterotonics reduces the risk of blood loss, transfusion, and postoperative anemia. There is high adherence with the use of uterotonic administration after birth in the United States.6,8
Ensure normothermia. Many patients become hypothermic during a cesarean birth. Active warming of the patient with an in-line IV fluid warmer and forced air warming over the patient’s body can reduce the risk of hypothermia.8
Initiate multimodal anesthesia. Anesthesiologists often use intrathecal or epidural morphine to enhance analgesia. Ketorolac administration prior to completion of the cesarean procedure and perioperative administration of acetaminophen may reduce postoperative pain.8 The use of preoperative antiemetics will reduce intraoperative and postoperative nausea and vomiting.
Initiate VTE prophylaxis. Pneumatic compression stockings are recommended. Anticoagulation should not be routinely used for VTE prophylaxis.6
Postoperative components of ERAS
Patient education to prepare for discharge home when ready. Patient education focused on home when ready is important in preparing the patient for discharge home.7 Completion of required newborn testing, lactation education, and contraception planning plus coordination of newborn pediatric follow-up is necessary before discharge.
Support early return of bowel function. Early return of bowel function is best supported by a multimodal approach including initiation of clear fluid intake immediately following surgery, encouraging consumption of a regular diet within 27 to 4 hours8 following surgery. Gum chewing for at least 5 minutes 3 times daily accelerates return of bowel function.8 In a meta-analysis of 10 randomized studies examining the effect of gum chewing after cesarean, the investigators reported that gum chewing shortened the time to passage of flatus and defecation.21
Early ambulation.
Sequentially advanced activity, starting with sitting on the edge of the bed, sitting in a chair, and ambulation within 8 hours of surgery, is recommended to facilitate faster recovery, reduce rates of complications, and enable transition to home.8
Early removal of the urinary catheter. It is recommended that the urinary catheter be removed within 12 hours after cesarean birth.8 Early removal of the urinary catheter increases patient mobility and reduces the length of hospitalization. Early removal of the urinary catheter may be associated with postoperative urinary retention and recatheterization in a small number of patients.
Prescribe routinely scheduled acetaminophen, nonsteroidal anti-inflammatory drugs (NSAIDs) and ketorolac. A key component of ERAS cesarean birth is the standardized administration of nonopioid pain medicines, alternating doses of acetaminophen and an NSAID. ERAS cesarean birth is likely to result in a reduction in inpatient and postdischarge opioid use.22-24
VTE prophylaxis. Pneumatic compression stockings are recommended. Anticoagulation should not be routinely used for VTE prophylaxis.8
Auditing and reporting adherence with components of ERAS
In clinical practice there may be a gap between a clinician’s subjective perception of their performance and an independent audit of their clinical performance. ERAS pathways should be implemented with a commitment to performing audits and providing quantitative feedback to clinicians. Consistent use of measurement, feedback, and coaching can improve performance and reduce variation among individual clinicians. As an example, in one study of the use of a surgical safety checklist, 99% of the surgeons reported that they routinely used a surgical safety checklist, but the audit showed that the checklist was used in only 60% of cases.25 Gaps between self-reported performance and audited performance are common in clinical practice. Audits with feedback are critical to improving adherence with the components of an ERAS pathway.
Three independent systematic reviews and meta-analyses report that ERAS pathways reduce hospital length of stay without increasing the readmission rate.26-28 One meta-analysis reported that ERAS may also reduce time to first mobilization and result in earlier removal of the urinary catheter.26 ERAS pathways also may reduce postoperative complications, lower pain scores, and decrease opioid use.27 The general consensus among quality and safety experts is that reducing variation through standardization of pathways is generally associated with improved quality and enhanced safety. ERAS pathways have been widely accepted in multiple surgical fields. ERAS pathways should become the standard for performing cesarean procedures.●

Cesarean birth is one of the most common major surgical procedures performed in developed countries1 with over 1,170,000 cesarean births in the United States in 2021.2 Many surgeons and anesthesiologists believe that Enhanced Recovery after Surgery (ERAS) pathways improve surgical outcomes.3,4 Important goals of ERAS include setting patient expectations for the surgical procedure, accelerating patient recovery to full function, and minimizing perioperative complications such as severe nausea, aspiration, surgical site infection, wound complications, and perioperative anemia. The ERAS Society in 20185-7 and the Society for Obstetric Anesthesia and Perinatology (SOAP) in 20218 proposed ERAS pathways for cesarean birth. Both societies recommended that obstetric units consider adopting an ERAS pathway compatible with local clinical resources. In addition, the American College of Obstetricians and Gynecologists (ACOG) has provided guidance for implementing ERAS pathways for gynecologic surgery.9 The consistent use of standardized protocols to improve surgical care in obstetrics should lead to a reduction in care variation and improve health equity outcomes.
The clinical interventions recommended for ERAS cesarean birth occur sequentially in the preoperative, intraoperative, and postoperative phases of care. The recommendations associated with each of these phases are reviewed below. It is important to note that each obstetric unit should use a multidisciplinary process to develop an ERAS pathway that best supports local practice given clinician preferences, patient characteristics, and resource availability.
Preoperative components of ERAS
Standardized patient education (SPE). SPE is an important component of ERAS, although evidence to support the recommendation is limited. At a minimum a written handout describing steps in the cesarean birth process, or a patient-education video should be part of patient education. The University of Michigan Medical Center has produced a 3-minute video for patients explaining ERAS cesarean birth.10 The University of Maryland Medical Center has produced a 2.5-minute video in English and Spanish, explaining ERAS cesarean birth for patients.11 Some surgeons place a telephone call to patients the evening before surgery to help orient the patient to ERAS cesarean birth.
Breastfeeding education. An important goal of obstetric care is to optimize the rate of exclusive breastfeeding at birth. Breastfeeding education, including a commitment to support the initiation of breastfeeding within 1 hour of birth, may enhance the rate of exclusive breastfeeding. There are numerous videos available for patients about breastfeeding after cesarean birth (as an example, see: https://www.youtube.com/watch?v=9iOGn85NdTg).
Limit fasting. In the past, surgical guidelines recommended fasting after midnight prior to surgery. The ERAS Society recommends that patients should be encouraged to drink clear fluids up to 2 hours before surgery and may have a light meal up to 6 hours before surgery (Part 1).
Carbohydrate loading. Surgery causes a metabolic stress that is increased by fasting. Carbohydrate loading prior to surgery reduces the magnitude of the catabolic state caused by the combination of surgery and fasting.12 SOAP and the ERAS Society recommend oral carbohydrate fluid supplementation 2 hours before surgery for nondiabetic patients. SOAP suggests 32 oz of Gatorade or 16 oz of clear apple juice as options for carbohydrate loading. For diabetic patients, the carbohydrate load can be omitted. In fasting pregnant patients at term, gastric emptying was near complete 2 hours after consumption of 400 mL of a carbohydrate drink.13 In one study, consumption of 400 mL of a carbohydrate drink 2 hours before cesarean resulted in a 7% increase in the newborn blood glucose level at 20 min after delivery.14
Minimize preoperative anemia. Approximately 50% of pregnant women are iron deficient and approximately 10% are anemic in the third trimester.15,16 Cesarean birth is associated with significant blood loss necessitating the need to optimize red blood cell mass before surgery. Measuring ferritin to identify patients with iron deficiency and aggressive iron replacement, including intravenous iron if necessary, will reduce the prevalence of anemia prior to cesarean birth.17 Another cause of anemia in pregnancy is vitamin B12 (cobalamin) deficiency. Low vitamin B12 is especially common in pregnant patients who have previously had bariatric surgery. One study reported that, of 113 pregnant patients who were, on average, 3 years from a bariatric surgery procedure, 12% had vitamin B12 circulating levels < 130 pg/mL.18 Among pregnant patients who are anemic, and do not have a hemoglobinopathy, measuring ferritin, folic acid, and vitamin B12 will help identify the cause of anemia and guide treatment.19
Optimize preoperative physical condition. Improving healthy behaviors and reducing unhealthy behaviors preoperatively may enhance patient recovery to full function. In the weeks before scheduled cesarean birth, cessation of the use of tobacco products, optimizing activity and improving diet quality, including increasing protein intake, may best prepare patients for the metabolic stress of surgery.
Continue to: Intraoperative components of ERAS...
Intraoperative components of ERAS
Reduce the risk of surgical site infection (SSI) and wound complications. Bundles that include antibiotics, chlorhexidine (or an alternative antibacterial soap) and clippers have been shown to reduce SSI.20 Routine administration of preoperative antibiotics is a consensus recommendation and there is high adherence with this recommendation in the United States. Chlorhexidine-alcohol is the preferred solution for skin preparation. Vaginal preparation with povidine-iodine or chlorhexidine may be considered.6
Surgical technique. Blunt extension of a transverse hysterotomy may reduce blood loss. Closure of the hysterotomy incision in 2 layers is recommended to reduce uterine scar dehiscence in a subsequent pregnancy. If the patient has ≥2 cm of subcutaneous tissue, this layer should be approximated with sutures. Skin closure should be with subcuticular suture.6
Optimize uterotonic administration. Routine use of uterotonics reduces the risk of blood loss, transfusion, and postoperative anemia. There is high adherence with the use of uterotonic administration after birth in the United States.6,8
Ensure normothermia. Many patients become hypothermic during a cesarean birth. Active warming of the patient with an in-line IV fluid warmer and forced air warming over the patient’s body can reduce the risk of hypothermia.8
Initiate multimodal anesthesia. Anesthesiologists often use intrathecal or epidural morphine to enhance analgesia. Ketorolac administration prior to completion of the cesarean procedure and perioperative administration of acetaminophen may reduce postoperative pain.8 The use of preoperative antiemetics will reduce intraoperative and postoperative nausea and vomiting.
Initiate VTE prophylaxis. Pneumatic compression stockings are recommended. Anticoagulation should not be routinely used for VTE prophylaxis.6
Postoperative components of ERAS
Patient education to prepare for discharge home when ready. Patient education focused on home when ready is important in preparing the patient for discharge home.7 Completion of required newborn testing, lactation education, and contraception planning plus coordination of newborn pediatric follow-up is necessary before discharge.
Support early return of bowel function. Early return of bowel function is best supported by a multimodal approach including initiation of clear fluid intake immediately following surgery, encouraging consumption of a regular diet within 27 to 4 hours8 following surgery. Gum chewing for at least 5 minutes 3 times daily accelerates return of bowel function.8 In a meta-analysis of 10 randomized studies examining the effect of gum chewing after cesarean, the investigators reported that gum chewing shortened the time to passage of flatus and defecation.21
Early ambulation.
Sequentially advanced activity, starting with sitting on the edge of the bed, sitting in a chair, and ambulation within 8 hours of surgery, is recommended to facilitate faster recovery, reduce rates of complications, and enable transition to home.8
Early removal of the urinary catheter. It is recommended that the urinary catheter be removed within 12 hours after cesarean birth.8 Early removal of the urinary catheter increases patient mobility and reduces the length of hospitalization. Early removal of the urinary catheter may be associated with postoperative urinary retention and recatheterization in a small number of patients.
Prescribe routinely scheduled acetaminophen, nonsteroidal anti-inflammatory drugs (NSAIDs) and ketorolac. A key component of ERAS cesarean birth is the standardized administration of nonopioid pain medicines, alternating doses of acetaminophen and an NSAID. ERAS cesarean birth is likely to result in a reduction in inpatient and postdischarge opioid use.22-24
VTE prophylaxis. Pneumatic compression stockings are recommended. Anticoagulation should not be routinely used for VTE prophylaxis.8
Auditing and reporting adherence with components of ERAS
In clinical practice there may be a gap between a clinician’s subjective perception of their performance and an independent audit of their clinical performance. ERAS pathways should be implemented with a commitment to performing audits and providing quantitative feedback to clinicians. Consistent use of measurement, feedback, and coaching can improve performance and reduce variation among individual clinicians. As an example, in one study of the use of a surgical safety checklist, 99% of the surgeons reported that they routinely used a surgical safety checklist, but the audit showed that the checklist was used in only 60% of cases.25 Gaps between self-reported performance and audited performance are common in clinical practice. Audits with feedback are critical to improving adherence with the components of an ERAS pathway.
Three independent systematic reviews and meta-analyses report that ERAS pathways reduce hospital length of stay without increasing the readmission rate.26-28 One meta-analysis reported that ERAS may also reduce time to first mobilization and result in earlier removal of the urinary catheter.26 ERAS pathways also may reduce postoperative complications, lower pain scores, and decrease opioid use.27 The general consensus among quality and safety experts is that reducing variation through standardization of pathways is generally associated with improved quality and enhanced safety. ERAS pathways have been widely accepted in multiple surgical fields. ERAS pathways should become the standard for performing cesarean procedures.●
1. Molina G, Weiser RG, Lipsitz SR, et al. Relationship between cesarean delivery rate and maternal and neonatal mortality. JAMA. 2015;314:2263-2270.
2. Hamilton BE, Martin JA, Osterman MJK. Births: provisional data for 2021. Vital Statistics Release; No. 20. Hyattsville, MD: National Center for Health Statistics. May 2022. https://www.cdc.gov/nchs/data/vsrr/vsrr020.pdf.
3. Berian JR, Ban KA, Liu JB, et al. Adherence to enhanced recovery protocols in NSQIP and association with colectomy outcomes. Ann Surg. 2019;486-493.
4. Ljungqvist O, Scott M, Fearon KC. Enhanced recovery after surgery: a review. JAMA Surg. 2017;152:292-298.
5. Wilson RD, Caughey AB, Wood SL, et al. Guidelines for antenatal and preoperative care in cesarean delivery: Enhanced Recovery after Surgery Society recommendations (Part 1). Am J Obstet Gynecol. 2018;219:523.e1-523.e15.
6. Caughey AB, Wood SL, Macones GA, et al Guidelines for intraoperative care in cesarean delivery: Enhanced Recovery after Surgery Society recommendations (Part 2). Am J Obstet Gynecol. 2018;219:533-544.
7. Macones GA, Caughey AB, Wood SL, et al. Guidelines for postoperative care in cesarean delivery: Enhanced Recovery after Surgery Society recommendations (Part 3). Am J Obstet Gynecol. 2019;221:247.e1-247.e9.
8. Bollag L, Lim G, Sultan P, et al. Society for Obstetric Anesthesia and Perinatology: Consensus statement and recommendations for enhanced recovery after cesarean. Anesth Analg. 2021;132:1362-1377.
9. Perioperative pathways: enhanced recovery after surgery. ACOG Committee Opinion No 750. American College of Obstetricians and Gynecologists. Obstet Gynecol. 2018;132:e120-130.
10. University of Michigan. ERAS: A patient education video. https://www.youtube.com/watch?v=CoFtgdluBc0. Accessed October 24, 2022.
11. University of Maryland. ERAS. https://www.umms.org/ummc/health-services/womens-health/ostetrics-gynecology/pregnancy-childbirth/labor-delivery/enhanced-recovery-after-cesarean. Accessed October 24, 2022.
12. Bilku DK, Dennison AR, Hall TC, et al. Role of preoperative carbohydrate loading: a systematic review. Ann R Coll Surg Engl. 2014;96:15-22.
13. Popivanov P, Irwin R, Walsh M, et al. Gastric emptying of carbohydrate drinks in term parturients before elective caesarean surgery: an observational study. Int J Obstet Anesth. 2020;41:29-34.
14. He Y, Liu C, Han Y, et al. The impact of carbohydrate-rich supplement taken two hours before caesarean delivery on maternal and neonatal perioperative outcomes- a randomized clinical trial. BMC Pregnancy Childbirth. 2021;21:682.
15. Auerbach M, Abernathy J, Juul S, et al. Prevalence of iron deficiency in first trimester, nonanemic pregnant women. J Matern Fetal Neonatal Med. 2021;34:1002-1005.
16. Mei Z, Cogswell ME, Looker AC, et al. Assessment of iron status in US pregnant women from the National Health and Nutrition Examination Survey (NHANES), 1996-2006. Am J Clin Nutr. 2011;93:1312-1320.
17. Nour N, Barbieri RL. Optimize detection and treatment of iron deficiency in pregnancy. OBG Manag. 2022;34:9-11.
18. Mead NC, Sakkatos P, Sakellaropoulos GC, et al. Pregnancy outcomes and nutritional indices after 3 types of bariatric surgery performed at a single institution. Surg Obes Relat Dis. 2014;10:1166-1173.
19. Achebe MM, Gafter-Gvili A. How I treat anemia in pregnancy: iron, cobalamin and folate. Blood. 2017;129:940-949.
20. Carter EB, Temming LA, Fowler S, et al. Evidence-based bundles and cesarean delivery surgical site infections: a systematic review and meta-analysis. Obstet Gynecol. 2017;130:735-746.
21. Wen Z, Shen M, Wu C, et al. Chewing gum for intestinal function recovery after caesarean section: a systematic review and meta-analysis. BMC Pregnancy Childbirth. 2017;17:105.
22. McCoy JA, Gutman S, Hamm RF, et al. The association between implementation of an enhanced recovery after cesarean pathway with standardized discharge prescriptions and opioid use and pain experience after cesarean delivery. Am J Perinatol. 2021;38:1341-1347.
23. Mullman L, Hilden P, Goral J, et al. Improved outcomes with an enhanced recovery approach to cesarean delivery. Obstet Gynecol. 2020;136:685-691.
24. Hedderson M, Lee D, Hunt E, et al. Enhanced recovery after surgery to change process measures and reduce opioid use after cesarean delivery: a quality improvement initiative. Obstet Gynecol. 2019;134:511-519.
25. Sendlhofer G, Lumenta DB, Leitgeb K, et al. The gap between individual perception and compliance: a quantitative follow-up study of the surgical safety checklist application. PLoS One. 2016;11:e0149212.
26. Sultan P, Sharawi N, Blake L, et al. Impact of enhanced recovery after cesarean delivery on maternal outcomes: a systematic review and meta-analysis. Anaesth Crit Care Pain Med. 2021;40:100935.
27. Meng X, Chen K, Yang C, et al. The clinical efficacy and safety of enhanced recovery after surgery for cesarean section: a systematic review and meta-analysis of randomized controlled trials and observational studies. Front Med. 2021;8:694385.
28. Corson E, Hind D, Beever D, et al. Enhanced recovery after elective caesarean: a rapid review of clinical protocols and an umbrella review of systematic reviews. BMC Pregnancy Childbirth. 2017;17:91.
1. Molina G, Weiser RG, Lipsitz SR, et al. Relationship between cesarean delivery rate and maternal and neonatal mortality. JAMA. 2015;314:2263-2270.
2. Hamilton BE, Martin JA, Osterman MJK. Births: provisional data for 2021. Vital Statistics Release; No. 20. Hyattsville, MD: National Center for Health Statistics. May 2022. https://www.cdc.gov/nchs/data/vsrr/vsrr020.pdf.
3. Berian JR, Ban KA, Liu JB, et al. Adherence to enhanced recovery protocols in NSQIP and association with colectomy outcomes. Ann Surg. 2019;486-493.
4. Ljungqvist O, Scott M, Fearon KC. Enhanced recovery after surgery: a review. JAMA Surg. 2017;152:292-298.
5. Wilson RD, Caughey AB, Wood SL, et al. Guidelines for antenatal and preoperative care in cesarean delivery: Enhanced Recovery after Surgery Society recommendations (Part 1). Am J Obstet Gynecol. 2018;219:523.e1-523.e15.
6. Caughey AB, Wood SL, Macones GA, et al Guidelines for intraoperative care in cesarean delivery: Enhanced Recovery after Surgery Society recommendations (Part 2). Am J Obstet Gynecol. 2018;219:533-544.
7. Macones GA, Caughey AB, Wood SL, et al. Guidelines for postoperative care in cesarean delivery: Enhanced Recovery after Surgery Society recommendations (Part 3). Am J Obstet Gynecol. 2019;221:247.e1-247.e9.
8. Bollag L, Lim G, Sultan P, et al. Society for Obstetric Anesthesia and Perinatology: Consensus statement and recommendations for enhanced recovery after cesarean. Anesth Analg. 2021;132:1362-1377.
9. Perioperative pathways: enhanced recovery after surgery. ACOG Committee Opinion No 750. American College of Obstetricians and Gynecologists. Obstet Gynecol. 2018;132:e120-130.
10. University of Michigan. ERAS: A patient education video. https://www.youtube.com/watch?v=CoFtgdluBc0. Accessed October 24, 2022.
11. University of Maryland. ERAS. https://www.umms.org/ummc/health-services/womens-health/ostetrics-gynecology/pregnancy-childbirth/labor-delivery/enhanced-recovery-after-cesarean. Accessed October 24, 2022.
12. Bilku DK, Dennison AR, Hall TC, et al. Role of preoperative carbohydrate loading: a systematic review. Ann R Coll Surg Engl. 2014;96:15-22.
13. Popivanov P, Irwin R, Walsh M, et al. Gastric emptying of carbohydrate drinks in term parturients before elective caesarean surgery: an observational study. Int J Obstet Anesth. 2020;41:29-34.
14. He Y, Liu C, Han Y, et al. The impact of carbohydrate-rich supplement taken two hours before caesarean delivery on maternal and neonatal perioperative outcomes- a randomized clinical trial. BMC Pregnancy Childbirth. 2021;21:682.
15. Auerbach M, Abernathy J, Juul S, et al. Prevalence of iron deficiency in first trimester, nonanemic pregnant women. J Matern Fetal Neonatal Med. 2021;34:1002-1005.
16. Mei Z, Cogswell ME, Looker AC, et al. Assessment of iron status in US pregnant women from the National Health and Nutrition Examination Survey (NHANES), 1996-2006. Am J Clin Nutr. 2011;93:1312-1320.
17. Nour N, Barbieri RL. Optimize detection and treatment of iron deficiency in pregnancy. OBG Manag. 2022;34:9-11.
18. Mead NC, Sakkatos P, Sakellaropoulos GC, et al. Pregnancy outcomes and nutritional indices after 3 types of bariatric surgery performed at a single institution. Surg Obes Relat Dis. 2014;10:1166-1173.
19. Achebe MM, Gafter-Gvili A. How I treat anemia in pregnancy: iron, cobalamin and folate. Blood. 2017;129:940-949.
20. Carter EB, Temming LA, Fowler S, et al. Evidence-based bundles and cesarean delivery surgical site infections: a systematic review and meta-analysis. Obstet Gynecol. 2017;130:735-746.
21. Wen Z, Shen M, Wu C, et al. Chewing gum for intestinal function recovery after caesarean section: a systematic review and meta-analysis. BMC Pregnancy Childbirth. 2017;17:105.
22. McCoy JA, Gutman S, Hamm RF, et al. The association between implementation of an enhanced recovery after cesarean pathway with standardized discharge prescriptions and opioid use and pain experience after cesarean delivery. Am J Perinatol. 2021;38:1341-1347.
23. Mullman L, Hilden P, Goral J, et al. Improved outcomes with an enhanced recovery approach to cesarean delivery. Obstet Gynecol. 2020;136:685-691.
24. Hedderson M, Lee D, Hunt E, et al. Enhanced recovery after surgery to change process measures and reduce opioid use after cesarean delivery: a quality improvement initiative. Obstet Gynecol. 2019;134:511-519.
25. Sendlhofer G, Lumenta DB, Leitgeb K, et al. The gap between individual perception and compliance: a quantitative follow-up study of the surgical safety checklist application. PLoS One. 2016;11:e0149212.
26. Sultan P, Sharawi N, Blake L, et al. Impact of enhanced recovery after cesarean delivery on maternal outcomes: a systematic review and meta-analysis. Anaesth Crit Care Pain Med. 2021;40:100935.
27. Meng X, Chen K, Yang C, et al. The clinical efficacy and safety of enhanced recovery after surgery for cesarean section: a systematic review and meta-analysis of randomized controlled trials and observational studies. Front Med. 2021;8:694385.
28. Corson E, Hind D, Beever D, et al. Enhanced recovery after elective caesarean: a rapid review of clinical protocols and an umbrella review of systematic reviews. BMC Pregnancy Childbirth. 2017;17:91.
Is evolution’s greatest triumph its worst blunder?
Of all the dazzling achievements of evolution, the most glorious by far is the emergence of the advanced human brain, especially the prefrontal cortex. Homo sapiens (the wise humans) are without doubt the most transformative development in the consequential annals of evolution. It was evolution’s spectacular “moonshot.” Ironically, it may also have been the seed of its destruction.
The unprecedented growth of the human brain over the past 7 million years (tripling in size) was a monumental tipping point in evolution that ultimately disrupted the entire orderly cascade of evolution on Planet Earth. Because of their superior intelligence, Homo sapiens have substantially “tinkered” with the foundations of evolution, such as “natural selection” and “survival of the fittest,” and may eventually change the course of evolution, or even reverse it. It should also be recognized that 20% of the human genome is Neanderthal, and the 2022 Nobel Prize in Physiology or Medicine was awarded to Svante Pääbo, the founder of the field of paleogenetics, who demonstrated genetically that Homo sapiens interbred with Homo neanderthalensis (who disappeared 30,000 years ago).
The majestic evolution of the human brain, in both size and complexity, led to monumental changes in the history of humankind compared to their primitive predecessors. Thanks to a superior cerebral cortex, humans developed traits and abilities that were nonexistent, even unimaginable, in the rest of animal kingdom, including primates and other mammals. These include thoughts; speech (hundreds of languages), spoken and written, to communicate among themselves; composed music and created numerous instruments to play it; invented mathematics, physics, and chemistry; developed agriculture to sustain and feed the masses; built homes, palaces, and pyramids, with water and sewage systems; hatched hundreds of religions and built thousands of houses of worship; built machines to transport themselves (cars, trains, ships, planes, and space shuttles); paved airports and countless miles of roads and railways; established companies, universities, hospitals, and research laboratories; built sports facilities such as stadiums for Olympic games and all its athletics; created hotels, restaurants, coffee shops, newspapers, and magazines; discovered the amazing DNA double helix and its genome with 23,000 coding genes containing instructions to build the brain and 200 other body tissues; developed surgeries and invented medications for diseases that would have killed millions every year; and established paper money to replace gold and silver coins. Humans established governments that included monarchies, dictatorships, democracies, and pseudodemocracies; stipulated constitutions, laws, and regulations to maintain various societies; and created several civilizations around the world that thrived and then faded. Over the past century, the advanced human brain elevated human existence to a higher sophistication with technologies such as electricity, phones, computers, internet, artificial intelligence, and machine learning. Using powerful rockets and space stations, humans have begun to expand their influence to the moon and planets of the solar system. Humans are very likely to continue achieving what evolution could never have done without evolving the human brain to become the most powerful force in nature.
The key ingredient of the brain that has enabled humans to achieve so much is the development of an advanced cognition, with superior functions that far exceed those of other living organisms. These include neurocognitive functions such as memory and attention, and executive functions that include planning, problem-solving, decision-making, abstract thinking, and insight. Those cognitive functions generate lofty prose, splendiferous poetry, and heavenly symphonies that inspire those who create it and others. The human brain also developed social cognition, with empathy, theory of mind, recognition of facial expressions, and courtship rituals that can trigger infatuation and love. Homo sapiens can experience a wide range of emotions in addition to love and attachment (necessary for procreation), including shame, guilt, surprise, embarrassment, disgust, and indifference, and a unique sense of right and wrong.
Perhaps the most distinctive human attribute, generated by an advanced prefrontal cortex, is a belief system that includes philosophy, politics, religion, and faith. Hundreds of different religions sprouted throughout human history (each claiming a monopoly on “the truth”), mandating rituals and behaviors, but also promoting a profound and unshakable belief in a divine “higher being” and an afterlife that mitigates the fear of death. Humans, unlike other animals, are painfully aware of mortality and the inevitability of death. Faith is an antidote for thanatophobia. Unfortunately, religious beliefs often generated severe and protracted schisms and warfare, with fatal consequences for their followers.
The anti-evolution aspect of the advanced brain
Despite remarkable talents and achievements, the unprecedented evolutionary expansion of the human brain also has a detrimental downside. The same intellectual power that led to astonishing positive accomplishments has a wicked side as well. While most animals have a predator, humans have become the “omni-predator” that preys on all living things. The balanced ecosystems of animals and plants has been dominated and disrupted by humans. Thousands of species that evolution had so ingeniously spawned became extinct because of human actions. The rainforests, jewels of nature’s plantation system, were victimized by human indifference to the deleterious effects on nature and climate. The excavation of coal and oil, exploited as necessary sources of energy for societal infrastructure, came back to haunt humans with climate consequences. In many ways, human “progress” corrupted evolution and dismantled its components. Survival of the fittest among various species was whittled down to “survival of humans” (and their domesticated animals) at the expense of all other organisms, animals, or plants.
Among Homo sapiens, momentous scientific, medical, and technological advances completely undermined the principle of survival of the fittest. Very premature infants, who would have certainly died, were kept alive. Children with disabling genetic disorders who would have perished in childhood were kept alive into the age of procreation, perpetuating the genetic mutations. The discovery of antibiotic and antiviral medications, and especially vaccines, ensured the survival of millions of humans who would have succumbed to infections. With evolution’s natural selection, humans who survived severe infections without medications would have passed on their “infection-resistant genes” to their progeny. The triumph of human medical progress can be conceptualized as a setback for the principles of evolution.
Continue to: The most malignant...
The most malignant consequence of the exceptional human brain is the evil of which it is capable. Human ingenuity led to the development of weapons of individual killing (guns), large-scale murder (machine guns), and massive destruction (nuclear weapons). And because aggression and warfare are an inherent part of human nature, the most potent predator for a human is another human. The history of humans is riddled with conflict and death on a large scale. Ironically, many wars were instigated by various religious groups around the world, who developed intense hostility towards one another.
There are other downsides to the advanced human brain. It can channel its talents and skills into unimaginably wicked and depraved behaviors, such as premeditated and well-planned murder, slavery, cults, child abuse, domestic abuse, pornography, fascism, dictatorships, and political corruption. Astonishingly, the same brain that can be loving, kind, friendly, and empathetic can suddenly become hateful, vengeful, cruel, vile, sinister, vicious, diabolical, and capable of unimaginable violence and atrocities. The advanced human brain definitely has a very dark side.
Finally, unlike other members of the animal kingdom, the human brain generates its virtual counterpart: the highly complex human mind, which is prone to various maladies, labeled as “psychiatric disorders.” No other animal species develops delusions, hallucinations, thought disorders, melancholia, mania, obsessive-compulsive disorder, generalized anxiety, panic attacks, posttraumatic stress disorder, psychopathy, narcissistic and borderline personality disorders, alcohol addiction, and drug abuse. Homo sapiens are the only species whose members decide to end their own life in large numbers. About 25% of human minds are afflicted with one or more of those psychiatric ailments.1,2 The redeeming grace of the large human brain is that it led to the development of pharmacologic and somatic treatments for most of them, including psychotherapy, which is a uniquely human treatment strategy that can mend many psychiatric disorders.
Evolution may not realize what it hath wrought when it evolved the dramatically expanded human brain, with its extraordinary cognition. This awe-inspiring “biological computer” can be creative and adaptive, with superlative survival abilities, but it can also degenerate and become nefarious, villainous, murderous, and even demonic. The human brain has essentially brought evolution to a screeching halt and may at some point end up destroying Earth and all of its Homo sapien inhabitants, who may foolishly use their weapons of mass destruction. The historic achievement of evolution has become the ultimate example of “the law of unintended consequences.”
1. Robin LN, Regier DA. Psychiatric Disorders in America: The Epidemiologic Catchment Area Study. Free Press; 1990.
2. Johns Hopkins Medicine. Mental Health Disorder Statistics. Accessed October 12, 2022. https://www.hopkinsmedicine.org/health/wellness-and-prevention/mental-health-disorder-statistics
Of all the dazzling achievements of evolution, the most glorious by far is the emergence of the advanced human brain, especially the prefrontal cortex. Homo sapiens (the wise humans) are without doubt the most transformative development in the consequential annals of evolution. It was evolution’s spectacular “moonshot.” Ironically, it may also have been the seed of its destruction.
The unprecedented growth of the human brain over the past 7 million years (tripling in size) was a monumental tipping point in evolution that ultimately disrupted the entire orderly cascade of evolution on Planet Earth. Because of their superior intelligence, Homo sapiens have substantially “tinkered” with the foundations of evolution, such as “natural selection” and “survival of the fittest,” and may eventually change the course of evolution, or even reverse it. It should also be recognized that 20% of the human genome is Neanderthal, and the 2022 Nobel Prize in Physiology or Medicine was awarded to Svante Pääbo, the founder of the field of paleogenetics, who demonstrated genetically that Homo sapiens interbred with Homo neanderthalensis (who disappeared 30,000 years ago).
The majestic evolution of the human brain, in both size and complexity, led to monumental changes in the history of humankind compared to their primitive predecessors. Thanks to a superior cerebral cortex, humans developed traits and abilities that were nonexistent, even unimaginable, in the rest of animal kingdom, including primates and other mammals. These include thoughts; speech (hundreds of languages), spoken and written, to communicate among themselves; composed music and created numerous instruments to play it; invented mathematics, physics, and chemistry; developed agriculture to sustain and feed the masses; built homes, palaces, and pyramids, with water and sewage systems; hatched hundreds of religions and built thousands of houses of worship; built machines to transport themselves (cars, trains, ships, planes, and space shuttles); paved airports and countless miles of roads and railways; established companies, universities, hospitals, and research laboratories; built sports facilities such as stadiums for Olympic games and all its athletics; created hotels, restaurants, coffee shops, newspapers, and magazines; discovered the amazing DNA double helix and its genome with 23,000 coding genes containing instructions to build the brain and 200 other body tissues; developed surgeries and invented medications for diseases that would have killed millions every year; and established paper money to replace gold and silver coins. Humans established governments that included monarchies, dictatorships, democracies, and pseudodemocracies; stipulated constitutions, laws, and regulations to maintain various societies; and created several civilizations around the world that thrived and then faded. Over the past century, the advanced human brain elevated human existence to a higher sophistication with technologies such as electricity, phones, computers, internet, artificial intelligence, and machine learning. Using powerful rockets and space stations, humans have begun to expand their influence to the moon and planets of the solar system. Humans are very likely to continue achieving what evolution could never have done without evolving the human brain to become the most powerful force in nature.
The key ingredient of the brain that has enabled humans to achieve so much is the development of an advanced cognition, with superior functions that far exceed those of other living organisms. These include neurocognitive functions such as memory and attention, and executive functions that include planning, problem-solving, decision-making, abstract thinking, and insight. Those cognitive functions generate lofty prose, splendiferous poetry, and heavenly symphonies that inspire those who create it and others. The human brain also developed social cognition, with empathy, theory of mind, recognition of facial expressions, and courtship rituals that can trigger infatuation and love. Homo sapiens can experience a wide range of emotions in addition to love and attachment (necessary for procreation), including shame, guilt, surprise, embarrassment, disgust, and indifference, and a unique sense of right and wrong.
Perhaps the most distinctive human attribute, generated by an advanced prefrontal cortex, is a belief system that includes philosophy, politics, religion, and faith. Hundreds of different religions sprouted throughout human history (each claiming a monopoly on “the truth”), mandating rituals and behaviors, but also promoting a profound and unshakable belief in a divine “higher being” and an afterlife that mitigates the fear of death. Humans, unlike other animals, are painfully aware of mortality and the inevitability of death. Faith is an antidote for thanatophobia. Unfortunately, religious beliefs often generated severe and protracted schisms and warfare, with fatal consequences for their followers.
The anti-evolution aspect of the advanced brain
Despite remarkable talents and achievements, the unprecedented evolutionary expansion of the human brain also has a detrimental downside. The same intellectual power that led to astonishing positive accomplishments has a wicked side as well. While most animals have a predator, humans have become the “omni-predator” that preys on all living things. The balanced ecosystems of animals and plants has been dominated and disrupted by humans. Thousands of species that evolution had so ingeniously spawned became extinct because of human actions. The rainforests, jewels of nature’s plantation system, were victimized by human indifference to the deleterious effects on nature and climate. The excavation of coal and oil, exploited as necessary sources of energy for societal infrastructure, came back to haunt humans with climate consequences. In many ways, human “progress” corrupted evolution and dismantled its components. Survival of the fittest among various species was whittled down to “survival of humans” (and their domesticated animals) at the expense of all other organisms, animals, or plants.
Among Homo sapiens, momentous scientific, medical, and technological advances completely undermined the principle of survival of the fittest. Very premature infants, who would have certainly died, were kept alive. Children with disabling genetic disorders who would have perished in childhood were kept alive into the age of procreation, perpetuating the genetic mutations. The discovery of antibiotic and antiviral medications, and especially vaccines, ensured the survival of millions of humans who would have succumbed to infections. With evolution’s natural selection, humans who survived severe infections without medications would have passed on their “infection-resistant genes” to their progeny. The triumph of human medical progress can be conceptualized as a setback for the principles of evolution.
Continue to: The most malignant...
The most malignant consequence of the exceptional human brain is the evil of which it is capable. Human ingenuity led to the development of weapons of individual killing (guns), large-scale murder (machine guns), and massive destruction (nuclear weapons). And because aggression and warfare are an inherent part of human nature, the most potent predator for a human is another human. The history of humans is riddled with conflict and death on a large scale. Ironically, many wars were instigated by various religious groups around the world, who developed intense hostility towards one another.
There are other downsides to the advanced human brain. It can channel its talents and skills into unimaginably wicked and depraved behaviors, such as premeditated and well-planned murder, slavery, cults, child abuse, domestic abuse, pornography, fascism, dictatorships, and political corruption. Astonishingly, the same brain that can be loving, kind, friendly, and empathetic can suddenly become hateful, vengeful, cruel, vile, sinister, vicious, diabolical, and capable of unimaginable violence and atrocities. The advanced human brain definitely has a very dark side.
Finally, unlike other members of the animal kingdom, the human brain generates its virtual counterpart: the highly complex human mind, which is prone to various maladies, labeled as “psychiatric disorders.” No other animal species develops delusions, hallucinations, thought disorders, melancholia, mania, obsessive-compulsive disorder, generalized anxiety, panic attacks, posttraumatic stress disorder, psychopathy, narcissistic and borderline personality disorders, alcohol addiction, and drug abuse. Homo sapiens are the only species whose members decide to end their own life in large numbers. About 25% of human minds are afflicted with one or more of those psychiatric ailments.1,2 The redeeming grace of the large human brain is that it led to the development of pharmacologic and somatic treatments for most of them, including psychotherapy, which is a uniquely human treatment strategy that can mend many psychiatric disorders.
Evolution may not realize what it hath wrought when it evolved the dramatically expanded human brain, with its extraordinary cognition. This awe-inspiring “biological computer” can be creative and adaptive, with superlative survival abilities, but it can also degenerate and become nefarious, villainous, murderous, and even demonic. The human brain has essentially brought evolution to a screeching halt and may at some point end up destroying Earth and all of its Homo sapien inhabitants, who may foolishly use their weapons of mass destruction. The historic achievement of evolution has become the ultimate example of “the law of unintended consequences.”
Of all the dazzling achievements of evolution, the most glorious by far is the emergence of the advanced human brain, especially the prefrontal cortex. Homo sapiens (the wise humans) are without doubt the most transformative development in the consequential annals of evolution. It was evolution’s spectacular “moonshot.” Ironically, it may also have been the seed of its destruction.
The unprecedented growth of the human brain over the past 7 million years (tripling in size) was a monumental tipping point in evolution that ultimately disrupted the entire orderly cascade of evolution on Planet Earth. Because of their superior intelligence, Homo sapiens have substantially “tinkered” with the foundations of evolution, such as “natural selection” and “survival of the fittest,” and may eventually change the course of evolution, or even reverse it. It should also be recognized that 20% of the human genome is Neanderthal, and the 2022 Nobel Prize in Physiology or Medicine was awarded to Svante Pääbo, the founder of the field of paleogenetics, who demonstrated genetically that Homo sapiens interbred with Homo neanderthalensis (who disappeared 30,000 years ago).
The majestic evolution of the human brain, in both size and complexity, led to monumental changes in the history of humankind compared to their primitive predecessors. Thanks to a superior cerebral cortex, humans developed traits and abilities that were nonexistent, even unimaginable, in the rest of animal kingdom, including primates and other mammals. These include thoughts; speech (hundreds of languages), spoken and written, to communicate among themselves; composed music and created numerous instruments to play it; invented mathematics, physics, and chemistry; developed agriculture to sustain and feed the masses; built homes, palaces, and pyramids, with water and sewage systems; hatched hundreds of religions and built thousands of houses of worship; built machines to transport themselves (cars, trains, ships, planes, and space shuttles); paved airports and countless miles of roads and railways; established companies, universities, hospitals, and research laboratories; built sports facilities such as stadiums for Olympic games and all its athletics; created hotels, restaurants, coffee shops, newspapers, and magazines; discovered the amazing DNA double helix and its genome with 23,000 coding genes containing instructions to build the brain and 200 other body tissues; developed surgeries and invented medications for diseases that would have killed millions every year; and established paper money to replace gold and silver coins. Humans established governments that included monarchies, dictatorships, democracies, and pseudodemocracies; stipulated constitutions, laws, and regulations to maintain various societies; and created several civilizations around the world that thrived and then faded. Over the past century, the advanced human brain elevated human existence to a higher sophistication with technologies such as electricity, phones, computers, internet, artificial intelligence, and machine learning. Using powerful rockets and space stations, humans have begun to expand their influence to the moon and planets of the solar system. Humans are very likely to continue achieving what evolution could never have done without evolving the human brain to become the most powerful force in nature.
The key ingredient of the brain that has enabled humans to achieve so much is the development of an advanced cognition, with superior functions that far exceed those of other living organisms. These include neurocognitive functions such as memory and attention, and executive functions that include planning, problem-solving, decision-making, abstract thinking, and insight. Those cognitive functions generate lofty prose, splendiferous poetry, and heavenly symphonies that inspire those who create it and others. The human brain also developed social cognition, with empathy, theory of mind, recognition of facial expressions, and courtship rituals that can trigger infatuation and love. Homo sapiens can experience a wide range of emotions in addition to love and attachment (necessary for procreation), including shame, guilt, surprise, embarrassment, disgust, and indifference, and a unique sense of right and wrong.
Perhaps the most distinctive human attribute, generated by an advanced prefrontal cortex, is a belief system that includes philosophy, politics, religion, and faith. Hundreds of different religions sprouted throughout human history (each claiming a monopoly on “the truth”), mandating rituals and behaviors, but also promoting a profound and unshakable belief in a divine “higher being” and an afterlife that mitigates the fear of death. Humans, unlike other animals, are painfully aware of mortality and the inevitability of death. Faith is an antidote for thanatophobia. Unfortunately, religious beliefs often generated severe and protracted schisms and warfare, with fatal consequences for their followers.
The anti-evolution aspect of the advanced brain
Despite remarkable talents and achievements, the unprecedented evolutionary expansion of the human brain also has a detrimental downside. The same intellectual power that led to astonishing positive accomplishments has a wicked side as well. While most animals have a predator, humans have become the “omni-predator” that preys on all living things. The balanced ecosystems of animals and plants has been dominated and disrupted by humans. Thousands of species that evolution had so ingeniously spawned became extinct because of human actions. The rainforests, jewels of nature’s plantation system, were victimized by human indifference to the deleterious effects on nature and climate. The excavation of coal and oil, exploited as necessary sources of energy for societal infrastructure, came back to haunt humans with climate consequences. In many ways, human “progress” corrupted evolution and dismantled its components. Survival of the fittest among various species was whittled down to “survival of humans” (and their domesticated animals) at the expense of all other organisms, animals, or plants.
Among Homo sapiens, momentous scientific, medical, and technological advances completely undermined the principle of survival of the fittest. Very premature infants, who would have certainly died, were kept alive. Children with disabling genetic disorders who would have perished in childhood were kept alive into the age of procreation, perpetuating the genetic mutations. The discovery of antibiotic and antiviral medications, and especially vaccines, ensured the survival of millions of humans who would have succumbed to infections. With evolution’s natural selection, humans who survived severe infections without medications would have passed on their “infection-resistant genes” to their progeny. The triumph of human medical progress can be conceptualized as a setback for the principles of evolution.
Continue to: The most malignant...
The most malignant consequence of the exceptional human brain is the evil of which it is capable. Human ingenuity led to the development of weapons of individual killing (guns), large-scale murder (machine guns), and massive destruction (nuclear weapons). And because aggression and warfare are an inherent part of human nature, the most potent predator for a human is another human. The history of humans is riddled with conflict and death on a large scale. Ironically, many wars were instigated by various religious groups around the world, who developed intense hostility towards one another.
There are other downsides to the advanced human brain. It can channel its talents and skills into unimaginably wicked and depraved behaviors, such as premeditated and well-planned murder, slavery, cults, child abuse, domestic abuse, pornography, fascism, dictatorships, and political corruption. Astonishingly, the same brain that can be loving, kind, friendly, and empathetic can suddenly become hateful, vengeful, cruel, vile, sinister, vicious, diabolical, and capable of unimaginable violence and atrocities. The advanced human brain definitely has a very dark side.
Finally, unlike other members of the animal kingdom, the human brain generates its virtual counterpart: the highly complex human mind, which is prone to various maladies, labeled as “psychiatric disorders.” No other animal species develops delusions, hallucinations, thought disorders, melancholia, mania, obsessive-compulsive disorder, generalized anxiety, panic attacks, posttraumatic stress disorder, psychopathy, narcissistic and borderline personality disorders, alcohol addiction, and drug abuse. Homo sapiens are the only species whose members decide to end their own life in large numbers. About 25% of human minds are afflicted with one or more of those psychiatric ailments.1,2 The redeeming grace of the large human brain is that it led to the development of pharmacologic and somatic treatments for most of them, including psychotherapy, which is a uniquely human treatment strategy that can mend many psychiatric disorders.
Evolution may not realize what it hath wrought when it evolved the dramatically expanded human brain, with its extraordinary cognition. This awe-inspiring “biological computer” can be creative and adaptive, with superlative survival abilities, but it can also degenerate and become nefarious, villainous, murderous, and even demonic. The human brain has essentially brought evolution to a screeching halt and may at some point end up destroying Earth and all of its Homo sapien inhabitants, who may foolishly use their weapons of mass destruction. The historic achievement of evolution has become the ultimate example of “the law of unintended consequences.”
1. Robin LN, Regier DA. Psychiatric Disorders in America: The Epidemiologic Catchment Area Study. Free Press; 1990.
2. Johns Hopkins Medicine. Mental Health Disorder Statistics. Accessed October 12, 2022. https://www.hopkinsmedicine.org/health/wellness-and-prevention/mental-health-disorder-statistics
1. Robin LN, Regier DA. Psychiatric Disorders in America: The Epidemiologic Catchment Area Study. Free Press; 1990.
2. Johns Hopkins Medicine. Mental Health Disorder Statistics. Accessed October 12, 2022. https://www.hopkinsmedicine.org/health/wellness-and-prevention/mental-health-disorder-statistics
Dietary sodium and potassium consumption and cardiovascular health

Hypertension is a prevalent medical problem among US women, with a higher prevalence among Black women, than among White, Hispanic, or Asian women (TABLE 1).1 Among US women aged 55 to 64 years, approximately 50% have hypertension or are taking a hypertension medicine.1 Hypertension is an important risk factor for cardiovascular disease, including stroke, coronary heart disease, heart failure, atrial fibrillation, and peripheral vascular disease.1,2 In a study of 1.3 million people, blood pressure (BP) ≥ 130/80 mm Hg was associated with an increased risk of a cardiovascular event, including myocardial infarction and stroke.2 Excessive sodium intake is an important risk factor for developing hypertension.3 In 2015–2016, 87% of US adults consumed >2,300 mg/d of sodium,4 an amount that is considered excessive.1 Less well known is the association between low potassium intake and hypertension. This editorial reviews the evidence that diets high in sodium and low in potassium contribute to the development of hypertension and cardiovascular disease.
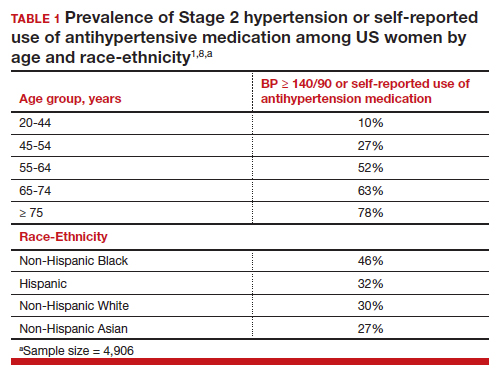
Sodium and potassium dueling cations
Many cohort studies report that diets high in sodium and low in potassium are associated with hypertension and an increased risk of cardiovascular disease. For example, in a cohort of 146,000 Chinese people, high sodium and low potassium intake was positively correlated with higher BP.5 In addition, the impact of increasing sodium intake or decreasing potassium intake was greater for people with a BMI ≥24 kg/m2, than people with a BMI <24 kg/m2. In a cohort of 11,095 US adults, high sodium and low potassium intake was associated with an increased risk of hypertension.6
In a study of 13,696 women, high potassium intake was associated with lower BP in participants with either a low or high sodium intake.7 In addition, over a 19-year follow up, higher potassium intake was associated with a lower risk of cardiovascular events.7 Comparing the highest (5,773 mg/d) vs lowest (2,783 mg/d) tertile of potassium intake, the decreased risk of a cardiovascular event was 0.89 (95% confidence interval [CI], 0.83–0.95).7
In a meta-analysis of data culled from 6 cohort studies, 10,709 adults with a mean age of 52 years, 54% of whom identified as women, were followed for a median of 8.8 years.8 Each adult contributed at least two 24-hour urine samples for measurement of sodium and potassium content. (Measurement of sodium and potassium in multiple 24-hour urine specimens from the same participant is thought to be the best way to assess sodium and potassium consumption.) The primary outcome was a cardiovascular event, including heart attack, stroke, or undergoing coronary revascularization procedures. In this study increasing consumption of sodium was associated with an increase in cardiovascular events, and increasing consumption of potassium was associated with a decrease in cardiovascular events. The hazard ratio for a cardiovascular event comparing high versus low consumption of sodium was 1.60 (95% CI, 1.19–2.14), and comparing high versus low consumption of potassium was 0.69 (95% CI, 0.51–0.91) (TABLE 2).8
Continue to: Clinical trial data on decreasing Na and/or increasing K consumption on CV outcomes...
Clinical trial data on decreasing Na and/or increasing K consumption on CV outcomes
Building on the cohort studies reporting that diets high in sodium and low in potassium are associated with hypertension and cardiovascular disease, clinical trials report that decreasing dietary sodium intake reduces BP and the risk of a cardiovascular event. For example, in a meta-analysis of 85 clinical trials studying the link between sodium and BP, the investigators concluded that there was a linear relationship between sodium intake and BP, with larger reductions in sodium intake associated with greater reductions in BP, down to a daily sodium intake of 1,000 to 1,500 mg.9 The effect of sodium reduction on BP was greatest in study participants with higher BP at baseline.
In a cluster-randomized clinical trial in China, people living in 600 villages were assigned to a control group, continuing to use sodium chloride in their food preparation or an experimental intervention, replacing sodium chloride with a substitute product containing 75% sodium chloride and 25% potassium chloride by weight.10 The inclusion criteria included people ≥60 years of age with high BP or a history of stroke. The mean duration of follow-up was 4.7 years. Half of the participants were female. A total of 73% of the participants had a history of stroke and 88% had hypertension. In this study, the rate of death was lower in the group that used the salt substitute than in the group using sodium chloride (39 vs 45 deaths per 1,000 person-years; rate ratio (RR) 0.88; 95% CI, 0.82–0.95, P<.001). The rate of major cardiovascular events (nonfatal stroke, nonfatal acute coronary syndrome or death from vascular causes) was decreased in the group that used salt substitute compared with the group using sodium chloride (49 vs 56 events per 1,000 person-years, rate ratio (RR), 0.87; 95% CI, 0.80–0.94; P<.001). Similarly, the rate of stroke was decreased in the group that used salt substitute compared with the group using sodium chloride (29 vs 34 events per 1,000 person-years; rate ratio (RR), 0.86; 95% CI, 0.77–0.96; P = .006). This study shows that by decreasing sodium intake and increasing potassium, cardiovascular outcomes are improved in people at high risk for a cardiovascular event.10 People with kidney disease or taking medications that decrease renal excretion of potassium should consult with their health care provider before using potassium chloride containing salt substitutes.
What is your daily intake of sodium and potassium?
Almost all packaged prepared foods have labels indicating the amount of sodium in one serving. Many packaged foods also report the amount of potassium in one serving. Many processed foods contain high amounts of sodium and low amounts of potassium. Processed and ultra-processed foods are a major dietary source of sodium.11 In contrast to processed foods, fresh fruits, vegetables, and milk have high quantities of potassium and low amounts of sodium. As an example, a major brand of canned chicken broth has 750 mg of sodium and 40 mg of potassium per one-half cup, a ratio of sodium to potassium of 19:1. By contrast, canned red kidney beans have 135 mg of sodium and 425 mg of potassium in one-half cup, a ratio of sodium to potassium of 1:3. Patients can better understand their daily sodium and potassium intake by reading the food labels. Calculating a sodium to potassium ratio for a food may help people better understand their salt intake and identify foods associated with positive health outcomes.
The optimal target for daily consumption of sodium and potassium is controversial (TABLE 2). The mean daily intakes of sodium and potassium in the United States are approximately 3,380 mg and 2,499 mg,respectively.12 The American College of Cardiology (ACC) recommends that an optimal diet contains <1,500 mg/d of sodium, a stringent target.1 If that target is unattainable, people should at least aim for a 1,000 mg/d-reduction in their current sodium intake.1 The World Health Organization strongly recommends that adults consume <2,000 mg/d of sodium.13 The National Academy of Science recommends adults seeking to reduce the risk of cardiovascular disease consume <2,300 mg/d of sodium.14 The top dietary sources of sodium include deli meat, pizza, burritos and tacos, soups, savory snacks (chips, crackers, popcorn), fried poultry, burgers, and eggs.15
The optimal target for daily consumption of potassium is controversial. The ACC recommends that an optimal diet contains 3,500–5,000 mg/d of potassium.1 The World Health Organization recommends that adults consume >3,510 mg/d of potassium.16 The top dietary sources of potassium include milk, fruit, vegetables, coffee, savory snacks (chips, crackers, popcorn), fruit juice, white potatoes, deli meats, burritos, and tacos.15 The foods with the greatest amount of potassium include banana, avocado, acorn squash, spinach, sweet potatoes, salmon, apricots, grapefruit, broccoli, and white beans. People with kidney disease or those who are taking medications that interfere with renal excretion of potassium should consult with their health care provider before adding more potassium to their diet.
The ACC also recommends1:
- Maintaining an optimal weight (a 1-kg reduction in weight is associated with a 1-mm Hg reduction in BP).
- Eating a healthy diet rich in fruits, vegetables, whole grains, and low-fat dairy products with reduced saturated and total fat.
- Regular aerobic physical activity 90 to 150 min/wk.
- Moderation in alcohol consumption, with men limiting consumption ≤ 2 drinks/d and women limiting consumption to ≤ 1 drink/d.
- Smoking cessation.
Most adults in the US have too much sodium and too little potassium in their daily diet. Diets high in sodium and low in potassium increase the risk of hypertension. In turn, this increases the risk of cardiovascular disease, including myocardial infarction and stroke. Many personal choices and societal factors contribute to our current imbalanced and unhealthy diet, rich in sodium and deficient in potassium. Our best approach to improve health and reduce cardiovascular disease is to guide people to modify unhealthy lifestyle behaviors.17 For patients who are ready to change, a counseling intervention using the 5 A’s (including assess risk behaviors, advise change, agree on goals/action plan, assist with treatment, and arrange follow-up) has been shown to result in improved dietary choices, increased physical activity, and reduced use of tobacco products.18 ●
Two randomized clinical trials completed in the 1990s, comparing a low-sodium and a standard diet, showed no effect of reducing sodium intake by 32% and 57% on the risk of developing preeclampsia.1,2 Based on these 2 studies, a Cochrane review concluded that during pregnancy salt consumption should remain a matter of personal preference.3 Three recent observational studies report a relationship between sodium intake and the risk of developing pregnancy-associated hypertension.
In a study of 66,651 singleton pregnancies in the Danish Birth Cohort, participants with the greatest daily sodium intake, ranging from 3,520 to 7,520 mg/d had a 54% increased risk of developing gestational hypertension (95% confidence interval [CI], 16%–104%) and a 20% increased risk of developing preeclampsia (95% CI, 1%–42%).4 Another cohort study also reported that elevated sodium chloride intake was associated with an increased risk of developing preeclampsia.5 In one study, among patients with preeclampsia, those with lower urinary sodium to potassium ratio were less likely to develop severe preeclampsia.6 In a pregnant rat model, high salt intake is associated with a severe increase in blood pressure, the development of proteinuria, and an increase in circulating plasma soluble fmslike tyrosine-kinase 1 (sFlt-1)—changes also seen in preeclampsia.7 Pregnancy associated hypertension may not be as “salt sensitive” as chronic hypertension.
Future research could explore the effect of dietary sodium and potassium intake on the risk of developing severe hypertension during pregnancy in patients with chronic hypertension.
References
1. Knuist M, Bonsel GJ, Zondervan HA, et al. Low sodium diet and pregnancy-induced hypertension, a multicenter randomised controlled trial. Brit J Obstet Gynecol. 1998;105:430-434.
2. van der Maten GD, van Raaij JMA, Visman L, et al. Low-sodium in pregnancy: effects on blood pressure and maternal nutritional status. Brit J Nutr. 1997;77:703-720.
3. Duley L, Henderson-Smart DJ, Meher S. Altered dietary salt for preventing pre-eclampsia, and its complications. Cochrane Database Syst Rev. 2005;CD005548.
4. Arvizu, M, Bjerregaard AA, Madsen MTB, et al. Sodium intake during pregnancy, but not other diet recommendations aimed at preventing cardiovascular disease, is positively related to risk of hypertensive disorders of pregnancy. J Nutr. 2020;150:159-166.
5. Birukov A, Andersen LB, Herse F, et al. Aldosterone, salt and potassium intakes as predictors of pregnancy outcome, including preeclampsia. Hypertension. 2019;74:391-398.
6. Yilmaz ZV, Akkas E, Turkmen GG, et al. Dietary sodium and potassium intake were associated with hypertension, kidney damage and adverse perinatal outcome in pregnant women with preeclampsia. Hypertension Preg. 2017;36:77-83.
7. Gillis EE, Williams JM, Garrett MR, et al. The Dahl salt-sensitive rat is a spontaneous model of superimposed preeclampsia. Am J Physiol Regul Integr Comp Physiol. 2015;309:R62-70.
- Whelton PK, Carey RM, Aronow WS, et al. ACC/ AHA/AAPA/ABC/ACPM/AGS/APHA/ASH/ ASPC/NMA/PCNA guideline for the prevention, detection, evaluation and management of high blood pressure in adults: Executive Summary: A report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. Circulation. 2018;138:e426-e483.
- Flint AC, Conell C, Ren X, et al. Effect of systolic and diastolic blood pressure on cardiovascular outcomes. N Engl J Med. 2019;381:243-251.
- Aljuraiban G, Jose AP, Gupta P, et al. Sodium intake, health implications and the role of population-level strategies. Nutr Rev. 2021;79:351-359.
- Clarke LS, Overwyk K, Bates M, et al. Temporal trends in dietary sodium intake among adults aged ≥ 19 years--United States 2003-2016. MMWR. 2021;70:1478-1482.
- Guo X, Zhang M, Li C, et al. Association between urinary sodium and potassium excretion and blood pressure among non-hypertensive adults-China, 2018-2019. China CDC Wkly. 2022;4:522-526.
- Li M, Yan S, Li X, et al. Association between blood pressure and dietary intakes of sodium and potassium among US adults using quantile regression analysis NHANES 2007-2014. J Hum Hypertens. 2020;34:346-354.
- Wouda RD, Boekholdt SM, Khaw KT, et al. Sex-specific associations between potassium intake, blood pressure and cardiovascular outcomes: the EPIC-Norfolk study. Europ Heart J. 2022, Epub July 21.
- Ma Y, He, Sun Q, et al. 24-hour urinary sodium and potassium excretion and cardiovascular risk. N Engl J Med. 2022;386:252-263.
- Filippini T, Malavolti M, Whelton PK, et al. Blood pressure effects of sodium reduction: dose-response meta-analysis of experimental studies. Circulation. 2021;143:1542-1567.
- Neal B, Wu Y, Feng X, et al. Effect of salt substitution on cardiovascular events. N Engl J Med. 2021;385:1067-1077.
- Monteiro CA, Cannon G, Moubarac JC, et al. The U.N. decade of nutrition: The NOVA food classification and the trouble with ultra-processing. Public Health Nutr. 2018;51:5-17.
- Nutrient intakes; From foods and beverages. Gender and Ag. WWEIA Data Tables. US Department of Health and Human Services, US Department of Agriculture. Web address Table 1. https://www .ars.usda.gov/ARSUserFiles/80400530/pdf /usual/Usual_Intake_gender_WWEIA_2015 _2018.pdf.
- WHO. Guideline: Sodium intake for adults and children. Geneva. World Health Organization (WHO), 2012. https://www.who.int /publications/i/item/9789241504836.
- National Academies of Sciences, Engineering and Medicine 2019. Dietary Reference Intakes for Sodium and Potassium. Washington DC: The National Academies Press. https://doi .org/10.17226/25353.
- Woodruff RC, Zhao L, Ahuja JKC, et al. Top food category contributors to sodium and potassium intake-United States 2015-2016. MMWR. 2020;69:1064-1069.
- WHO. Guideline: Potassium intake for adults and children. Geneva. World Health Organization (WHO), 2012. https://www.who.int /publications/i/item/9789241504829.
- Li Y, Pan A, Wang DD, et al. Impact of healthy lifestyle factors on life expectancies in the US population. Circulation. 2018;138:345-355.
- US Preventive Services Task Force. Behavioral counseling interventions to promote a healthy diet and physical activity for cardiovascular disease prevention in adults without cardiovascular disease risk factors. JAMA. 2022;328:367-374.

Hypertension is a prevalent medical problem among US women, with a higher prevalence among Black women, than among White, Hispanic, or Asian women (TABLE 1).1 Among US women aged 55 to 64 years, approximately 50% have hypertension or are taking a hypertension medicine.1 Hypertension is an important risk factor for cardiovascular disease, including stroke, coronary heart disease, heart failure, atrial fibrillation, and peripheral vascular disease.1,2 In a study of 1.3 million people, blood pressure (BP) ≥ 130/80 mm Hg was associated with an increased risk of a cardiovascular event, including myocardial infarction and stroke.2 Excessive sodium intake is an important risk factor for developing hypertension.3 In 2015–2016, 87% of US adults consumed >2,300 mg/d of sodium,4 an amount that is considered excessive.1 Less well known is the association between low potassium intake and hypertension. This editorial reviews the evidence that diets high in sodium and low in potassium contribute to the development of hypertension and cardiovascular disease.

Sodium and potassium dueling cations
Many cohort studies report that diets high in sodium and low in potassium are associated with hypertension and an increased risk of cardiovascular disease. For example, in a cohort of 146,000 Chinese people, high sodium and low potassium intake was positively correlated with higher BP.5 In addition, the impact of increasing sodium intake or decreasing potassium intake was greater for people with a BMI ≥24 kg/m2, than people with a BMI <24 kg/m2. In a cohort of 11,095 US adults, high sodium and low potassium intake was associated with an increased risk of hypertension.6
In a study of 13,696 women, high potassium intake was associated with lower BP in participants with either a low or high sodium intake.7 In addition, over a 19-year follow up, higher potassium intake was associated with a lower risk of cardiovascular events.7 Comparing the highest (5,773 mg/d) vs lowest (2,783 mg/d) tertile of potassium intake, the decreased risk of a cardiovascular event was 0.89 (95% confidence interval [CI], 0.83–0.95).7
In a meta-analysis of data culled from 6 cohort studies, 10,709 adults with a mean age of 52 years, 54% of whom identified as women, were followed for a median of 8.8 years.8 Each adult contributed at least two 24-hour urine samples for measurement of sodium and potassium content. (Measurement of sodium and potassium in multiple 24-hour urine specimens from the same participant is thought to be the best way to assess sodium and potassium consumption.) The primary outcome was a cardiovascular event, including heart attack, stroke, or undergoing coronary revascularization procedures. In this study increasing consumption of sodium was associated with an increase in cardiovascular events, and increasing consumption of potassium was associated with a decrease in cardiovascular events. The hazard ratio for a cardiovascular event comparing high versus low consumption of sodium was 1.60 (95% CI, 1.19–2.14), and comparing high versus low consumption of potassium was 0.69 (95% CI, 0.51–0.91) (TABLE 2).8

Continue to: Clinical trial data on decreasing Na and/or increasing K consumption on CV outcomes...
Clinical trial data on decreasing Na and/or increasing K consumption on CV outcomes
Building on the cohort studies reporting that diets high in sodium and low in potassium are associated with hypertension and cardiovascular disease, clinical trials report that decreasing dietary sodium intake reduces BP and the risk of a cardiovascular event. For example, in a meta-analysis of 85 clinical trials studying the link between sodium and BP, the investigators concluded that there was a linear relationship between sodium intake and BP, with larger reductions in sodium intake associated with greater reductions in BP, down to a daily sodium intake of 1,000 to 1,500 mg.9 The effect of sodium reduction on BP was greatest in study participants with higher BP at baseline.
In a cluster-randomized clinical trial in China, people living in 600 villages were assigned to a control group, continuing to use sodium chloride in their food preparation or an experimental intervention, replacing sodium chloride with a substitute product containing 75% sodium chloride and 25% potassium chloride by weight.10 The inclusion criteria included people ≥60 years of age with high BP or a history of stroke. The mean duration of follow-up was 4.7 years. Half of the participants were female. A total of 73% of the participants had a history of stroke and 88% had hypertension. In this study, the rate of death was lower in the group that used the salt substitute than in the group using sodium chloride (39 vs 45 deaths per 1,000 person-years; rate ratio (RR) 0.88; 95% CI, 0.82–0.95, P<.001). The rate of major cardiovascular events (nonfatal stroke, nonfatal acute coronary syndrome or death from vascular causes) was decreased in the group that used salt substitute compared with the group using sodium chloride (49 vs 56 events per 1,000 person-years, rate ratio (RR), 0.87; 95% CI, 0.80–0.94; P<.001). Similarly, the rate of stroke was decreased in the group that used salt substitute compared with the group using sodium chloride (29 vs 34 events per 1,000 person-years; rate ratio (RR), 0.86; 95% CI, 0.77–0.96; P = .006). This study shows that by decreasing sodium intake and increasing potassium, cardiovascular outcomes are improved in people at high risk for a cardiovascular event.10 People with kidney disease or taking medications that decrease renal excretion of potassium should consult with their health care provider before using potassium chloride containing salt substitutes.
What is your daily intake of sodium and potassium?
Almost all packaged prepared foods have labels indicating the amount of sodium in one serving. Many packaged foods also report the amount of potassium in one serving. Many processed foods contain high amounts of sodium and low amounts of potassium. Processed and ultra-processed foods are a major dietary source of sodium.11 In contrast to processed foods, fresh fruits, vegetables, and milk have high quantities of potassium and low amounts of sodium. As an example, a major brand of canned chicken broth has 750 mg of sodium and 40 mg of potassium per one-half cup, a ratio of sodium to potassium of 19:1. By contrast, canned red kidney beans have 135 mg of sodium and 425 mg of potassium in one-half cup, a ratio of sodium to potassium of 1:3. Patients can better understand their daily sodium and potassium intake by reading the food labels. Calculating a sodium to potassium ratio for a food may help people better understand their salt intake and identify foods associated with positive health outcomes.
The optimal target for daily consumption of sodium and potassium is controversial (TABLE 2). The mean daily intakes of sodium and potassium in the United States are approximately 3,380 mg and 2,499 mg,respectively.12 The American College of Cardiology (ACC) recommends that an optimal diet contains <1,500 mg/d of sodium, a stringent target.1 If that target is unattainable, people should at least aim for a 1,000 mg/d-reduction in their current sodium intake.1 The World Health Organization strongly recommends that adults consume <2,000 mg/d of sodium.13 The National Academy of Science recommends adults seeking to reduce the risk of cardiovascular disease consume <2,300 mg/d of sodium.14 The top dietary sources of sodium include deli meat, pizza, burritos and tacos, soups, savory snacks (chips, crackers, popcorn), fried poultry, burgers, and eggs.15
The optimal target for daily consumption of potassium is controversial. The ACC recommends that an optimal diet contains 3,500–5,000 mg/d of potassium.1 The World Health Organization recommends that adults consume >3,510 mg/d of potassium.16 The top dietary sources of potassium include milk, fruit, vegetables, coffee, savory snacks (chips, crackers, popcorn), fruit juice, white potatoes, deli meats, burritos, and tacos.15 The foods with the greatest amount of potassium include banana, avocado, acorn squash, spinach, sweet potatoes, salmon, apricots, grapefruit, broccoli, and white beans. People with kidney disease or those who are taking medications that interfere with renal excretion of potassium should consult with their health care provider before adding more potassium to their diet.
The ACC also recommends1:
- Maintaining an optimal weight (a 1-kg reduction in weight is associated with a 1-mm Hg reduction in BP).
- Eating a healthy diet rich in fruits, vegetables, whole grains, and low-fat dairy products with reduced saturated and total fat.
- Regular aerobic physical activity 90 to 150 min/wk.
- Moderation in alcohol consumption, with men limiting consumption ≤ 2 drinks/d and women limiting consumption to ≤ 1 drink/d.
- Smoking cessation.
Most adults in the US have too much sodium and too little potassium in their daily diet. Diets high in sodium and low in potassium increase the risk of hypertension. In turn, this increases the risk of cardiovascular disease, including myocardial infarction and stroke. Many personal choices and societal factors contribute to our current imbalanced and unhealthy diet, rich in sodium and deficient in potassium. Our best approach to improve health and reduce cardiovascular disease is to guide people to modify unhealthy lifestyle behaviors.17 For patients who are ready to change, a counseling intervention using the 5 A’s (including assess risk behaviors, advise change, agree on goals/action plan, assist with treatment, and arrange follow-up) has been shown to result in improved dietary choices, increased physical activity, and reduced use of tobacco products.18 ●
Two randomized clinical trials completed in the 1990s, comparing a low-sodium and a standard diet, showed no effect of reducing sodium intake by 32% and 57% on the risk of developing preeclampsia.1,2 Based on these 2 studies, a Cochrane review concluded that during pregnancy salt consumption should remain a matter of personal preference.3 Three recent observational studies report a relationship between sodium intake and the risk of developing pregnancy-associated hypertension.
In a study of 66,651 singleton pregnancies in the Danish Birth Cohort, participants with the greatest daily sodium intake, ranging from 3,520 to 7,520 mg/d had a 54% increased risk of developing gestational hypertension (95% confidence interval [CI], 16%–104%) and a 20% increased risk of developing preeclampsia (95% CI, 1%–42%).4 Another cohort study also reported that elevated sodium chloride intake was associated with an increased risk of developing preeclampsia.5 In one study, among patients with preeclampsia, those with lower urinary sodium to potassium ratio were less likely to develop severe preeclampsia.6 In a pregnant rat model, high salt intake is associated with a severe increase in blood pressure, the development of proteinuria, and an increase in circulating plasma soluble fmslike tyrosine-kinase 1 (sFlt-1)—changes also seen in preeclampsia.7 Pregnancy associated hypertension may not be as “salt sensitive” as chronic hypertension.
Future research could explore the effect of dietary sodium and potassium intake on the risk of developing severe hypertension during pregnancy in patients with chronic hypertension.
References
1. Knuist M, Bonsel GJ, Zondervan HA, et al. Low sodium diet and pregnancy-induced hypertension, a multicenter randomised controlled trial. Brit J Obstet Gynecol. 1998;105:430-434.
2. van der Maten GD, van Raaij JMA, Visman L, et al. Low-sodium in pregnancy: effects on blood pressure and maternal nutritional status. Brit J Nutr. 1997;77:703-720.
3. Duley L, Henderson-Smart DJ, Meher S. Altered dietary salt for preventing pre-eclampsia, and its complications. Cochrane Database Syst Rev. 2005;CD005548.
4. Arvizu, M, Bjerregaard AA, Madsen MTB, et al. Sodium intake during pregnancy, but not other diet recommendations aimed at preventing cardiovascular disease, is positively related to risk of hypertensive disorders of pregnancy. J Nutr. 2020;150:159-166.
5. Birukov A, Andersen LB, Herse F, et al. Aldosterone, salt and potassium intakes as predictors of pregnancy outcome, including preeclampsia. Hypertension. 2019;74:391-398.
6. Yilmaz ZV, Akkas E, Turkmen GG, et al. Dietary sodium and potassium intake were associated with hypertension, kidney damage and adverse perinatal outcome in pregnant women with preeclampsia. Hypertension Preg. 2017;36:77-83.
7. Gillis EE, Williams JM, Garrett MR, et al. The Dahl salt-sensitive rat is a spontaneous model of superimposed preeclampsia. Am J Physiol Regul Integr Comp Physiol. 2015;309:R62-70.

Hypertension is a prevalent medical problem among US women, with a higher prevalence among Black women, than among White, Hispanic, or Asian women (TABLE 1).1 Among US women aged 55 to 64 years, approximately 50% have hypertension or are taking a hypertension medicine.1 Hypertension is an important risk factor for cardiovascular disease, including stroke, coronary heart disease, heart failure, atrial fibrillation, and peripheral vascular disease.1,2 In a study of 1.3 million people, blood pressure (BP) ≥ 130/80 mm Hg was associated with an increased risk of a cardiovascular event, including myocardial infarction and stroke.2 Excessive sodium intake is an important risk factor for developing hypertension.3 In 2015–2016, 87% of US adults consumed >2,300 mg/d of sodium,4 an amount that is considered excessive.1 Less well known is the association between low potassium intake and hypertension. This editorial reviews the evidence that diets high in sodium and low in potassium contribute to the development of hypertension and cardiovascular disease.

Sodium and potassium dueling cations
Many cohort studies report that diets high in sodium and low in potassium are associated with hypertension and an increased risk of cardiovascular disease. For example, in a cohort of 146,000 Chinese people, high sodium and low potassium intake was positively correlated with higher BP.5 In addition, the impact of increasing sodium intake or decreasing potassium intake was greater for people with a BMI ≥24 kg/m2, than people with a BMI <24 kg/m2. In a cohort of 11,095 US adults, high sodium and low potassium intake was associated with an increased risk of hypertension.6
In a study of 13,696 women, high potassium intake was associated with lower BP in participants with either a low or high sodium intake.7 In addition, over a 19-year follow up, higher potassium intake was associated with a lower risk of cardiovascular events.7 Comparing the highest (5,773 mg/d) vs lowest (2,783 mg/d) tertile of potassium intake, the decreased risk of a cardiovascular event was 0.89 (95% confidence interval [CI], 0.83–0.95).7
In a meta-analysis of data culled from 6 cohort studies, 10,709 adults with a mean age of 52 years, 54% of whom identified as women, were followed for a median of 8.8 years.8 Each adult contributed at least two 24-hour urine samples for measurement of sodium and potassium content. (Measurement of sodium and potassium in multiple 24-hour urine specimens from the same participant is thought to be the best way to assess sodium and potassium consumption.) The primary outcome was a cardiovascular event, including heart attack, stroke, or undergoing coronary revascularization procedures. In this study increasing consumption of sodium was associated with an increase in cardiovascular events, and increasing consumption of potassium was associated with a decrease in cardiovascular events. The hazard ratio for a cardiovascular event comparing high versus low consumption of sodium was 1.60 (95% CI, 1.19–2.14), and comparing high versus low consumption of potassium was 0.69 (95% CI, 0.51–0.91) (TABLE 2).8

Continue to: Clinical trial data on decreasing Na and/or increasing K consumption on CV outcomes...
Clinical trial data on decreasing Na and/or increasing K consumption on CV outcomes
Building on the cohort studies reporting that diets high in sodium and low in potassium are associated with hypertension and cardiovascular disease, clinical trials report that decreasing dietary sodium intake reduces BP and the risk of a cardiovascular event. For example, in a meta-analysis of 85 clinical trials studying the link between sodium and BP, the investigators concluded that there was a linear relationship between sodium intake and BP, with larger reductions in sodium intake associated with greater reductions in BP, down to a daily sodium intake of 1,000 to 1,500 mg.9 The effect of sodium reduction on BP was greatest in study participants with higher BP at baseline.
In a cluster-randomized clinical trial in China, people living in 600 villages were assigned to a control group, continuing to use sodium chloride in their food preparation or an experimental intervention, replacing sodium chloride with a substitute product containing 75% sodium chloride and 25% potassium chloride by weight.10 The inclusion criteria included people ≥60 years of age with high BP or a history of stroke. The mean duration of follow-up was 4.7 years. Half of the participants were female. A total of 73% of the participants had a history of stroke and 88% had hypertension. In this study, the rate of death was lower in the group that used the salt substitute than in the group using sodium chloride (39 vs 45 deaths per 1,000 person-years; rate ratio (RR) 0.88; 95% CI, 0.82–0.95, P<.001). The rate of major cardiovascular events (nonfatal stroke, nonfatal acute coronary syndrome or death from vascular causes) was decreased in the group that used salt substitute compared with the group using sodium chloride (49 vs 56 events per 1,000 person-years, rate ratio (RR), 0.87; 95% CI, 0.80–0.94; P<.001). Similarly, the rate of stroke was decreased in the group that used salt substitute compared with the group using sodium chloride (29 vs 34 events per 1,000 person-years; rate ratio (RR), 0.86; 95% CI, 0.77–0.96; P = .006). This study shows that by decreasing sodium intake and increasing potassium, cardiovascular outcomes are improved in people at high risk for a cardiovascular event.10 People with kidney disease or taking medications that decrease renal excretion of potassium should consult with their health care provider before using potassium chloride containing salt substitutes.
What is your daily intake of sodium and potassium?
Almost all packaged prepared foods have labels indicating the amount of sodium in one serving. Many packaged foods also report the amount of potassium in one serving. Many processed foods contain high amounts of sodium and low amounts of potassium. Processed and ultra-processed foods are a major dietary source of sodium.11 In contrast to processed foods, fresh fruits, vegetables, and milk have high quantities of potassium and low amounts of sodium. As an example, a major brand of canned chicken broth has 750 mg of sodium and 40 mg of potassium per one-half cup, a ratio of sodium to potassium of 19:1. By contrast, canned red kidney beans have 135 mg of sodium and 425 mg of potassium in one-half cup, a ratio of sodium to potassium of 1:3. Patients can better understand their daily sodium and potassium intake by reading the food labels. Calculating a sodium to potassium ratio for a food may help people better understand their salt intake and identify foods associated with positive health outcomes.
The optimal target for daily consumption of sodium and potassium is controversial (TABLE 2). The mean daily intakes of sodium and potassium in the United States are approximately 3,380 mg and 2,499 mg,respectively.12 The American College of Cardiology (ACC) recommends that an optimal diet contains <1,500 mg/d of sodium, a stringent target.1 If that target is unattainable, people should at least aim for a 1,000 mg/d-reduction in their current sodium intake.1 The World Health Organization strongly recommends that adults consume <2,000 mg/d of sodium.13 The National Academy of Science recommends adults seeking to reduce the risk of cardiovascular disease consume <2,300 mg/d of sodium.14 The top dietary sources of sodium include deli meat, pizza, burritos and tacos, soups, savory snacks (chips, crackers, popcorn), fried poultry, burgers, and eggs.15
The optimal target for daily consumption of potassium is controversial. The ACC recommends that an optimal diet contains 3,500–5,000 mg/d of potassium.1 The World Health Organization recommends that adults consume >3,510 mg/d of potassium.16 The top dietary sources of potassium include milk, fruit, vegetables, coffee, savory snacks (chips, crackers, popcorn), fruit juice, white potatoes, deli meats, burritos, and tacos.15 The foods with the greatest amount of potassium include banana, avocado, acorn squash, spinach, sweet potatoes, salmon, apricots, grapefruit, broccoli, and white beans. People with kidney disease or those who are taking medications that interfere with renal excretion of potassium should consult with their health care provider before adding more potassium to their diet.
The ACC also recommends1:
- Maintaining an optimal weight (a 1-kg reduction in weight is associated with a 1-mm Hg reduction in BP).
- Eating a healthy diet rich in fruits, vegetables, whole grains, and low-fat dairy products with reduced saturated and total fat.
- Regular aerobic physical activity 90 to 150 min/wk.
- Moderation in alcohol consumption, with men limiting consumption ≤ 2 drinks/d and women limiting consumption to ≤ 1 drink/d.
- Smoking cessation.
Most adults in the US have too much sodium and too little potassium in their daily diet. Diets high in sodium and low in potassium increase the risk of hypertension. In turn, this increases the risk of cardiovascular disease, including myocardial infarction and stroke. Many personal choices and societal factors contribute to our current imbalanced and unhealthy diet, rich in sodium and deficient in potassium. Our best approach to improve health and reduce cardiovascular disease is to guide people to modify unhealthy lifestyle behaviors.17 For patients who are ready to change, a counseling intervention using the 5 A’s (including assess risk behaviors, advise change, agree on goals/action plan, assist with treatment, and arrange follow-up) has been shown to result in improved dietary choices, increased physical activity, and reduced use of tobacco products.18 ●
Two randomized clinical trials completed in the 1990s, comparing a low-sodium and a standard diet, showed no effect of reducing sodium intake by 32% and 57% on the risk of developing preeclampsia.1,2 Based on these 2 studies, a Cochrane review concluded that during pregnancy salt consumption should remain a matter of personal preference.3 Three recent observational studies report a relationship between sodium intake and the risk of developing pregnancy-associated hypertension.
In a study of 66,651 singleton pregnancies in the Danish Birth Cohort, participants with the greatest daily sodium intake, ranging from 3,520 to 7,520 mg/d had a 54% increased risk of developing gestational hypertension (95% confidence interval [CI], 16%–104%) and a 20% increased risk of developing preeclampsia (95% CI, 1%–42%).4 Another cohort study also reported that elevated sodium chloride intake was associated with an increased risk of developing preeclampsia.5 In one study, among patients with preeclampsia, those with lower urinary sodium to potassium ratio were less likely to develop severe preeclampsia.6 In a pregnant rat model, high salt intake is associated with a severe increase in blood pressure, the development of proteinuria, and an increase in circulating plasma soluble fmslike tyrosine-kinase 1 (sFlt-1)—changes also seen in preeclampsia.7 Pregnancy associated hypertension may not be as “salt sensitive” as chronic hypertension.
Future research could explore the effect of dietary sodium and potassium intake on the risk of developing severe hypertension during pregnancy in patients with chronic hypertension.
References
1. Knuist M, Bonsel GJ, Zondervan HA, et al. Low sodium diet and pregnancy-induced hypertension, a multicenter randomised controlled trial. Brit J Obstet Gynecol. 1998;105:430-434.
2. van der Maten GD, van Raaij JMA, Visman L, et al. Low-sodium in pregnancy: effects on blood pressure and maternal nutritional status. Brit J Nutr. 1997;77:703-720.
3. Duley L, Henderson-Smart DJ, Meher S. Altered dietary salt for preventing pre-eclampsia, and its complications. Cochrane Database Syst Rev. 2005;CD005548.
4. Arvizu, M, Bjerregaard AA, Madsen MTB, et al. Sodium intake during pregnancy, but not other diet recommendations aimed at preventing cardiovascular disease, is positively related to risk of hypertensive disorders of pregnancy. J Nutr. 2020;150:159-166.
5. Birukov A, Andersen LB, Herse F, et al. Aldosterone, salt and potassium intakes as predictors of pregnancy outcome, including preeclampsia. Hypertension. 2019;74:391-398.
6. Yilmaz ZV, Akkas E, Turkmen GG, et al. Dietary sodium and potassium intake were associated with hypertension, kidney damage and adverse perinatal outcome in pregnant women with preeclampsia. Hypertension Preg. 2017;36:77-83.
7. Gillis EE, Williams JM, Garrett MR, et al. The Dahl salt-sensitive rat is a spontaneous model of superimposed preeclampsia. Am J Physiol Regul Integr Comp Physiol. 2015;309:R62-70.
- Whelton PK, Carey RM, Aronow WS, et al. ACC/ AHA/AAPA/ABC/ACPM/AGS/APHA/ASH/ ASPC/NMA/PCNA guideline for the prevention, detection, evaluation and management of high blood pressure in adults: Executive Summary: A report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. Circulation. 2018;138:e426-e483.
- Flint AC, Conell C, Ren X, et al. Effect of systolic and diastolic blood pressure on cardiovascular outcomes. N Engl J Med. 2019;381:243-251.
- Aljuraiban G, Jose AP, Gupta P, et al. Sodium intake, health implications and the role of population-level strategies. Nutr Rev. 2021;79:351-359.
- Clarke LS, Overwyk K, Bates M, et al. Temporal trends in dietary sodium intake among adults aged ≥ 19 years--United States 2003-2016. MMWR. 2021;70:1478-1482.
- Guo X, Zhang M, Li C, et al. Association between urinary sodium and potassium excretion and blood pressure among non-hypertensive adults-China, 2018-2019. China CDC Wkly. 2022;4:522-526.
- Li M, Yan S, Li X, et al. Association between blood pressure and dietary intakes of sodium and potassium among US adults using quantile regression analysis NHANES 2007-2014. J Hum Hypertens. 2020;34:346-354.
- Wouda RD, Boekholdt SM, Khaw KT, et al. Sex-specific associations between potassium intake, blood pressure and cardiovascular outcomes: the EPIC-Norfolk study. Europ Heart J. 2022, Epub July 21.
- Ma Y, He, Sun Q, et al. 24-hour urinary sodium and potassium excretion and cardiovascular risk. N Engl J Med. 2022;386:252-263.
- Filippini T, Malavolti M, Whelton PK, et al. Blood pressure effects of sodium reduction: dose-response meta-analysis of experimental studies. Circulation. 2021;143:1542-1567.
- Neal B, Wu Y, Feng X, et al. Effect of salt substitution on cardiovascular events. N Engl J Med. 2021;385:1067-1077.
- Monteiro CA, Cannon G, Moubarac JC, et al. The U.N. decade of nutrition: The NOVA food classification and the trouble with ultra-processing. Public Health Nutr. 2018;51:5-17.
- Nutrient intakes; From foods and beverages. Gender and Ag. WWEIA Data Tables. US Department of Health and Human Services, US Department of Agriculture. Web address Table 1. https://www .ars.usda.gov/ARSUserFiles/80400530/pdf /usual/Usual_Intake_gender_WWEIA_2015 _2018.pdf.
- WHO. Guideline: Sodium intake for adults and children. Geneva. World Health Organization (WHO), 2012. https://www.who.int /publications/i/item/9789241504836.
- National Academies of Sciences, Engineering and Medicine 2019. Dietary Reference Intakes for Sodium and Potassium. Washington DC: The National Academies Press. https://doi .org/10.17226/25353.
- Woodruff RC, Zhao L, Ahuja JKC, et al. Top food category contributors to sodium and potassium intake-United States 2015-2016. MMWR. 2020;69:1064-1069.
- WHO. Guideline: Potassium intake for adults and children. Geneva. World Health Organization (WHO), 2012. https://www.who.int /publications/i/item/9789241504829.
- Li Y, Pan A, Wang DD, et al. Impact of healthy lifestyle factors on life expectancies in the US population. Circulation. 2018;138:345-355.
- US Preventive Services Task Force. Behavioral counseling interventions to promote a healthy diet and physical activity for cardiovascular disease prevention in adults without cardiovascular disease risk factors. JAMA. 2022;328:367-374.
- Whelton PK, Carey RM, Aronow WS, et al. ACC/ AHA/AAPA/ABC/ACPM/AGS/APHA/ASH/ ASPC/NMA/PCNA guideline for the prevention, detection, evaluation and management of high blood pressure in adults: Executive Summary: A report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. Circulation. 2018;138:e426-e483.
- Flint AC, Conell C, Ren X, et al. Effect of systolic and diastolic blood pressure on cardiovascular outcomes. N Engl J Med. 2019;381:243-251.
- Aljuraiban G, Jose AP, Gupta P, et al. Sodium intake, health implications and the role of population-level strategies. Nutr Rev. 2021;79:351-359.
- Clarke LS, Overwyk K, Bates M, et al. Temporal trends in dietary sodium intake among adults aged ≥ 19 years--United States 2003-2016. MMWR. 2021;70:1478-1482.
- Guo X, Zhang M, Li C, et al. Association between urinary sodium and potassium excretion and blood pressure among non-hypertensive adults-China, 2018-2019. China CDC Wkly. 2022;4:522-526.
- Li M, Yan S, Li X, et al. Association between blood pressure and dietary intakes of sodium and potassium among US adults using quantile regression analysis NHANES 2007-2014. J Hum Hypertens. 2020;34:346-354.
- Wouda RD, Boekholdt SM, Khaw KT, et al. Sex-specific associations between potassium intake, blood pressure and cardiovascular outcomes: the EPIC-Norfolk study. Europ Heart J. 2022, Epub July 21.
- Ma Y, He, Sun Q, et al. 24-hour urinary sodium and potassium excretion and cardiovascular risk. N Engl J Med. 2022;386:252-263.
- Filippini T, Malavolti M, Whelton PK, et al. Blood pressure effects of sodium reduction: dose-response meta-analysis of experimental studies. Circulation. 2021;143:1542-1567.
- Neal B, Wu Y, Feng X, et al. Effect of salt substitution on cardiovascular events. N Engl J Med. 2021;385:1067-1077.
- Monteiro CA, Cannon G, Moubarac JC, et al. The U.N. decade of nutrition: The NOVA food classification and the trouble with ultra-processing. Public Health Nutr. 2018;51:5-17.
- Nutrient intakes; From foods and beverages. Gender and Ag. WWEIA Data Tables. US Department of Health and Human Services, US Department of Agriculture. Web address Table 1. https://www .ars.usda.gov/ARSUserFiles/80400530/pdf /usual/Usual_Intake_gender_WWEIA_2015 _2018.pdf.
- WHO. Guideline: Sodium intake for adults and children. Geneva. World Health Organization (WHO), 2012. https://www.who.int /publications/i/item/9789241504836.
- National Academies of Sciences, Engineering and Medicine 2019. Dietary Reference Intakes for Sodium and Potassium. Washington DC: The National Academies Press. https://doi .org/10.17226/25353.
- Woodruff RC, Zhao L, Ahuja JKC, et al. Top food category contributors to sodium and potassium intake-United States 2015-2016. MMWR. 2020;69:1064-1069.
- WHO. Guideline: Potassium intake for adults and children. Geneva. World Health Organization (WHO), 2012. https://www.who.int /publications/i/item/9789241504829.
- Li Y, Pan A, Wang DD, et al. Impact of healthy lifestyle factors on life expectancies in the US population. Circulation. 2018;138:345-355.
- US Preventive Services Task Force. Behavioral counseling interventions to promote a healthy diet and physical activity for cardiovascular disease prevention in adults without cardiovascular disease risk factors. JAMA. 2022;328:367-374.
The accelerating societal entropy undermines mental health
According to the second law of thermodynamics, it is inevitable that entropy will continue to increase over time.1 Entropy is a measure of disorder, which can eventuate in chaos and lead to profound uncertainty, with serious psychological consequences.
The increase in entropy is usually gradual. It took hundreds of years for powerful empires and civilizations to collapse and disappear. Inanimate objects such as a house, a piece of furniture, or a piece of equipment eventually deteriorate and break down over time. Tidy offices will become messy, cluttered, and dirty unless attended to regularly. Living organisms, including humans, inevitably undergo an aging process with cellular senescence, atrophy, and loss of cerebral, muscle, and bone tissue, ending in death. Even human relationships will eventually fracture, wither, and end. The passage of time ruthlessly increases the entropy of everything in life. Even the 13-billion-year-old universe, which currently looks formidable and permanent to us, is inexorably expanding and hurtling towards a calamitous end a few billion years from now.
To slow down, halt, or reverse entropy, work and energy must be invested. A house requires regular maintenance for all its components to avoid deteriorating and becoming uninhabitable (very high entropy). Humans require massive amounts of work during fetal life, infancy, childhood, adolescence, adulthood, and throughout old age. This includes work by parents, teachers, friends, physicians, farmers, and manufacturers of food, clothing, and sundry supplies, all targeted to maintain an individual and slow the rate of entropy. But death is inevitable as the final stage of human entropy.
The brain is an entropic organ.2 Psychiatric disorders can be conceptualized as a neurobiologic consequence of a major rise in brain entropy. The chaos created by high brain entropy will lead to a disruption of basic mental functions such as thought, mood, affect, impulses, behavior, and cognition. Brain entropy increases can be due to genetics or the environment, but most often are due an interaction of both (G x E).
Societal entropy and our patients
Psychiatric patients are deeply influenced by the context in which they live (society). The entropy of contemporary society is rising at an alarming rate, which means that order is rapidly degenerating into disorder at an unprecedented pace. When the COVID-19 pandemic abruptly emerged in early 2020, it was a major public health shock that drastically changed the lives of all citizens and dramatically increased societal entropy. The pandemic led to lockdowns, fear of death, gut-wrenching uncertainty (especially for a whole year before vaccines were developed, but even after), loss of socialization and sexual intimacy, loss of employment, financial straits, and an inability to access routine medical or surgical procedures. Everyone in society developed anxiety and acute stress reaction, but those with pre-existing psychiatric disorders suffered the most with an intensification of their symptoms.
The unforeseen, sudden, and traumatically life-altering pandemic triggered various degrees of posttraumatic stress disorder across all age groups, and painful death in medically compromised individuals and older adults. Both physical and psychological entropy skyrocketed and the “order” of life as we knew it rapidly disintegrated into shambles and disorder. The abrupt traumatic jolt triggered various degrees of deleterious impacts on the brains of all who experienced it in real time. The rise in the psychobiological entropy was unprecedented across the structures of society, especially the population, its vulnerable human component.
But even as the worst of the pandemic is in our rearview mirror and life again has a semblance of normality, the rise of entropy continues to accelerate because we continue to be surrounded and engulfed by countless stressful events in contemporary society. Those nagging stresses continue to transmute order to chaos and metamorphose comforting predictability to entrenched uncertainty:
- Toxic political hyperpartisanship, with intense animus and visceral bidirectional hatred
- Racial tensions, with overt bias across groups
- Economic turmoil, with inflation and threats of recession
- Actual wars and threats of war
- Social media that spreads bad news and distorts facts
- An opioid crisis, with hundreds of thousands of deaths
- Skyrocketing crime, with a decline in policing and quick release of criminals without bail
- A ruthless and arbitrary “cancel culture” that doesn’t even spare the previously revered founders of the republic
- Cognitive dissonance of disparaging Abraham Lincoln despite his major achievement of eliminating slavery by waging a civil war
- The social and medical strife regarding access to abortion.
Continue to: I also would include...
(I also would include some “entropy pet peeves” of mine: Torn clothes as a fashion statement, transforming tattoos from an oddity to a fad, nose rings that disfigure pretty faces, and banishing neckties for men.)
Our role in this scenario
As psychiatrists, we must step up to intensify the work needed to slow down and even reverse the dangerously rising brain entropy in our patients. But that is not an easy task given the implosion of societal norms and traditional values, along with the radicalization of beliefs, with utter intolerance of others’ beliefs. We also face the challenge of maintaining a modicum of resilience and wellness in ourselves, which can be antidotes to entropy.
It’s impossible to stop the inevitability of rising entropy, both physical and psychological, but psychiatrists and other mental health professionals must invest their skills and talents now more than ever to at least slow down the pace of entropy among our patients. Otherwise, psychological chaos and disorder will be quite damaging to their lives, and worsen their outcomes.
1. Ben-Naim A. Entropy Demystified. World Scientific; 2007.
2. Carhart-Harris RL. The entropic brain - revisited. Neuropharmacology. 2018;142:167-178. doi: 10.1016/j.neuropharm.2018.03.010
According to the second law of thermodynamics, it is inevitable that entropy will continue to increase over time.1 Entropy is a measure of disorder, which can eventuate in chaos and lead to profound uncertainty, with serious psychological consequences.
The increase in entropy is usually gradual. It took hundreds of years for powerful empires and civilizations to collapse and disappear. Inanimate objects such as a house, a piece of furniture, or a piece of equipment eventually deteriorate and break down over time. Tidy offices will become messy, cluttered, and dirty unless attended to regularly. Living organisms, including humans, inevitably undergo an aging process with cellular senescence, atrophy, and loss of cerebral, muscle, and bone tissue, ending in death. Even human relationships will eventually fracture, wither, and end. The passage of time ruthlessly increases the entropy of everything in life. Even the 13-billion-year-old universe, which currently looks formidable and permanent to us, is inexorably expanding and hurtling towards a calamitous end a few billion years from now.
To slow down, halt, or reverse entropy, work and energy must be invested. A house requires regular maintenance for all its components to avoid deteriorating and becoming uninhabitable (very high entropy). Humans require massive amounts of work during fetal life, infancy, childhood, adolescence, adulthood, and throughout old age. This includes work by parents, teachers, friends, physicians, farmers, and manufacturers of food, clothing, and sundry supplies, all targeted to maintain an individual and slow the rate of entropy. But death is inevitable as the final stage of human entropy.
The brain is an entropic organ.2 Psychiatric disorders can be conceptualized as a neurobiologic consequence of a major rise in brain entropy. The chaos created by high brain entropy will lead to a disruption of basic mental functions such as thought, mood, affect, impulses, behavior, and cognition. Brain entropy increases can be due to genetics or the environment, but most often are due an interaction of both (G x E).
Societal entropy and our patients
Psychiatric patients are deeply influenced by the context in which they live (society). The entropy of contemporary society is rising at an alarming rate, which means that order is rapidly degenerating into disorder at an unprecedented pace. When the COVID-19 pandemic abruptly emerged in early 2020, it was a major public health shock that drastically changed the lives of all citizens and dramatically increased societal entropy. The pandemic led to lockdowns, fear of death, gut-wrenching uncertainty (especially for a whole year before vaccines were developed, but even after), loss of socialization and sexual intimacy, loss of employment, financial straits, and an inability to access routine medical or surgical procedures. Everyone in society developed anxiety and acute stress reaction, but those with pre-existing psychiatric disorders suffered the most with an intensification of their symptoms.
The unforeseen, sudden, and traumatically life-altering pandemic triggered various degrees of posttraumatic stress disorder across all age groups, and painful death in medically compromised individuals and older adults. Both physical and psychological entropy skyrocketed and the “order” of life as we knew it rapidly disintegrated into shambles and disorder. The abrupt traumatic jolt triggered various degrees of deleterious impacts on the brains of all who experienced it in real time. The rise in the psychobiological entropy was unprecedented across the structures of society, especially the population, its vulnerable human component.
But even as the worst of the pandemic is in our rearview mirror and life again has a semblance of normality, the rise of entropy continues to accelerate because we continue to be surrounded and engulfed by countless stressful events in contemporary society. Those nagging stresses continue to transmute order to chaos and metamorphose comforting predictability to entrenched uncertainty:
- Toxic political hyperpartisanship, with intense animus and visceral bidirectional hatred
- Racial tensions, with overt bias across groups
- Economic turmoil, with inflation and threats of recession
- Actual wars and threats of war
- Social media that spreads bad news and distorts facts
- An opioid crisis, with hundreds of thousands of deaths
- Skyrocketing crime, with a decline in policing and quick release of criminals without bail
- A ruthless and arbitrary “cancel culture” that doesn’t even spare the previously revered founders of the republic
- Cognitive dissonance of disparaging Abraham Lincoln despite his major achievement of eliminating slavery by waging a civil war
- The social and medical strife regarding access to abortion.
Continue to: I also would include...
(I also would include some “entropy pet peeves” of mine: Torn clothes as a fashion statement, transforming tattoos from an oddity to a fad, nose rings that disfigure pretty faces, and banishing neckties for men.)
Our role in this scenario
As psychiatrists, we must step up to intensify the work needed to slow down and even reverse the dangerously rising brain entropy in our patients. But that is not an easy task given the implosion of societal norms and traditional values, along with the radicalization of beliefs, with utter intolerance of others’ beliefs. We also face the challenge of maintaining a modicum of resilience and wellness in ourselves, which can be antidotes to entropy.
It’s impossible to stop the inevitability of rising entropy, both physical and psychological, but psychiatrists and other mental health professionals must invest their skills and talents now more than ever to at least slow down the pace of entropy among our patients. Otherwise, psychological chaos and disorder will be quite damaging to their lives, and worsen their outcomes.
According to the second law of thermodynamics, it is inevitable that entropy will continue to increase over time.1 Entropy is a measure of disorder, which can eventuate in chaos and lead to profound uncertainty, with serious psychological consequences.
The increase in entropy is usually gradual. It took hundreds of years for powerful empires and civilizations to collapse and disappear. Inanimate objects such as a house, a piece of furniture, or a piece of equipment eventually deteriorate and break down over time. Tidy offices will become messy, cluttered, and dirty unless attended to regularly. Living organisms, including humans, inevitably undergo an aging process with cellular senescence, atrophy, and loss of cerebral, muscle, and bone tissue, ending in death. Even human relationships will eventually fracture, wither, and end. The passage of time ruthlessly increases the entropy of everything in life. Even the 13-billion-year-old universe, which currently looks formidable and permanent to us, is inexorably expanding and hurtling towards a calamitous end a few billion years from now.
To slow down, halt, or reverse entropy, work and energy must be invested. A house requires regular maintenance for all its components to avoid deteriorating and becoming uninhabitable (very high entropy). Humans require massive amounts of work during fetal life, infancy, childhood, adolescence, adulthood, and throughout old age. This includes work by parents, teachers, friends, physicians, farmers, and manufacturers of food, clothing, and sundry supplies, all targeted to maintain an individual and slow the rate of entropy. But death is inevitable as the final stage of human entropy.
The brain is an entropic organ.2 Psychiatric disorders can be conceptualized as a neurobiologic consequence of a major rise in brain entropy. The chaos created by high brain entropy will lead to a disruption of basic mental functions such as thought, mood, affect, impulses, behavior, and cognition. Brain entropy increases can be due to genetics or the environment, but most often are due an interaction of both (G x E).
Societal entropy and our patients
Psychiatric patients are deeply influenced by the context in which they live (society). The entropy of contemporary society is rising at an alarming rate, which means that order is rapidly degenerating into disorder at an unprecedented pace. When the COVID-19 pandemic abruptly emerged in early 2020, it was a major public health shock that drastically changed the lives of all citizens and dramatically increased societal entropy. The pandemic led to lockdowns, fear of death, gut-wrenching uncertainty (especially for a whole year before vaccines were developed, but even after), loss of socialization and sexual intimacy, loss of employment, financial straits, and an inability to access routine medical or surgical procedures. Everyone in society developed anxiety and acute stress reaction, but those with pre-existing psychiatric disorders suffered the most with an intensification of their symptoms.
The unforeseen, sudden, and traumatically life-altering pandemic triggered various degrees of posttraumatic stress disorder across all age groups, and painful death in medically compromised individuals and older adults. Both physical and psychological entropy skyrocketed and the “order” of life as we knew it rapidly disintegrated into shambles and disorder. The abrupt traumatic jolt triggered various degrees of deleterious impacts on the brains of all who experienced it in real time. The rise in the psychobiological entropy was unprecedented across the structures of society, especially the population, its vulnerable human component.
But even as the worst of the pandemic is in our rearview mirror and life again has a semblance of normality, the rise of entropy continues to accelerate because we continue to be surrounded and engulfed by countless stressful events in contemporary society. Those nagging stresses continue to transmute order to chaos and metamorphose comforting predictability to entrenched uncertainty:
- Toxic political hyperpartisanship, with intense animus and visceral bidirectional hatred
- Racial tensions, with overt bias across groups
- Economic turmoil, with inflation and threats of recession
- Actual wars and threats of war
- Social media that spreads bad news and distorts facts
- An opioid crisis, with hundreds of thousands of deaths
- Skyrocketing crime, with a decline in policing and quick release of criminals without bail
- A ruthless and arbitrary “cancel culture” that doesn’t even spare the previously revered founders of the republic
- Cognitive dissonance of disparaging Abraham Lincoln despite his major achievement of eliminating slavery by waging a civil war
- The social and medical strife regarding access to abortion.
Continue to: I also would include...
(I also would include some “entropy pet peeves” of mine: Torn clothes as a fashion statement, transforming tattoos from an oddity to a fad, nose rings that disfigure pretty faces, and banishing neckties for men.)
Our role in this scenario
As psychiatrists, we must step up to intensify the work needed to slow down and even reverse the dangerously rising brain entropy in our patients. But that is not an easy task given the implosion of societal norms and traditional values, along with the radicalization of beliefs, with utter intolerance of others’ beliefs. We also face the challenge of maintaining a modicum of resilience and wellness in ourselves, which can be antidotes to entropy.
It’s impossible to stop the inevitability of rising entropy, both physical and psychological, but psychiatrists and other mental health professionals must invest their skills and talents now more than ever to at least slow down the pace of entropy among our patients. Otherwise, psychological chaos and disorder will be quite damaging to their lives, and worsen their outcomes.
1. Ben-Naim A. Entropy Demystified. World Scientific; 2007.
2. Carhart-Harris RL. The entropic brain - revisited. Neuropharmacology. 2018;142:167-178. doi: 10.1016/j.neuropharm.2018.03.010
1. Ben-Naim A. Entropy Demystified. World Scientific; 2007.
2. Carhart-Harris RL. The entropic brain - revisited. Neuropharmacology. 2018;142:167-178. doi: 10.1016/j.neuropharm.2018.03.010
An epidemic of hypertensive disorders of pregnancy
Hypertension in pregnancy is a major challenge in current obstetric practice. Based on an analysis of the National Inpatient Sample, the Centers for Disease Control and Prevention (CDC) recently reported that from 2017 to 2019 the prevalence of hypertensive disorders in pregnancy increased from 13.3% to 15.9% of hospital deliveries.1 During that same time period, the prevalence of pregnancy-associated hypertension, which includes preeclampsia, eclampsia, and gestational hypertension, increased from 10.8% to 13.0%.1 The prevalence of chronic hypertension increased from 2.0% to 2.3%.1 In 2017 and 2019, unspecified maternal hypertension was diagnosed in 0.5% and 0.6% of the sample, respectively.1
Bruno and colleagues reported a 3-fold increase in the prevalence of HDPs from 1989 to 2020, with an acceleration in the rate of increase from 2010 to 2020.2 The increase in prevalence of HDPs may be caused by an increase in the prevalence of advanced maternal age, obesity, and diabetes. Black patients are disproportionately impacted by both pregnancy-associated hypertension and chronic hypertension.1 In 2019, the prevalence of pregnancy-associated hypertension was greater among Black patients (15.6%), than White (12.1%), Hispanic (10.6%), or Asian or Pacific Islander patients (7.7%).1 Similarly, the prevalence of chronic hypertension was greater among Black patients (4.3%) than among White (2.0%), Hispanic (1.5%), or Asian or Pacific Islander patients (1.2%).1 Racial/ethnic differences in HDPs may be influenced by poverty; structural racism; or lack of access to care, diet, and obesity.3,4
HDPs are major contributors to maternal morbidity and mortality. The CDC reported that among maternal deaths occurring during the delivery hospitalization, 32% of the decedents had documented hypertension.1 HDPs are associated with an approximately 2.5-fold increased risk of a severe morbidity, a composite measure that includes blood transfusion, acute kidney injury, disseminated intravascular coagulation, sepsis, shock, and pulmonary edema.5 A history of HDPs is associated with an approximately 67% increase in the lifetime risk of cardiovascular disease, including coronary artery disease, stroke, peripheral vascular disease, and heart failure.6,7
What are the best antihypertensive medications for pregnancy?
All clinicians know that the use of angiotensin-converting-enzyme inhibitors (ACE-Is) and angiotensin-receptor-blockers (ARBs) are contraindicated in pregnancy because they cause major congenital anomalies, with an odds ratio of 1.8 (95% confidence interval [CI], 1.42-2.34), compared with no exposure.8 In addition, ACE-Is and ARBs increase the risk of stillbirth, with an odds ratio of 1.75 (95% CI, 1.21-2.53).8 No increase in congenital anomalies were detected for patients exposed to other antihypertensive medications.8 Prior to attempting conception, patients with chronic hypertension should discontinue ACE-Is and ARBs and initiate an alternative medication.
The most commonly used antihypertensive medications in pregnancy are labetalol, nifedipine, and methyldopa.9 Labetalol blocks the beta-1, beta-2, and alpha-1 adrenergic receptors.10 Nifedipine blocks calcium entry into cells through the L-type calcium channel.11 Methyldopa is a central nervous system alpha-2 adrenergic agonist.12 The dose range for these commonly used medications are labetalol 400 mg to 2,400 mg daily in divided doses every 8 to 12 hours, nifedipine extended-release 30 mg to 120 mg daily, and methyldopa 500 mg to 2 g daily in 2 to 4 divided doses. Some clinicians recommend prescribing divided doses of nifedipine extended release at doses ≥ 60 mg for patients who have bothersome adverse effects, hypotension following a single daily dose, or hypertension between single daily doses. The nifedipine extended release tablets should not be divided. If monotherapy with the maximal daily dose of labetalol does not achieve the blood pressure (BP) target, adding nifedipine as a second agent is an option.9 Similarly, if monotherapy with the maximal daily dose of nifedipine extended release does not achieve the BP target, adding labetalol as a second agent is an option.9
In a network meta-analysis of antihypertensive medications used in pregnancy, that included 61 trials and 6,923 participants, all the medications studied reduced the risk of developing severe hypertension by 30% to 70%.13 Sufficient data was available to also report that labetalol used to treat hypertension in pregnancy reduced the risk of developing proteinuria.13 Given similar efficacy among antihypertensive medications, patient comorbidities may influence the medication choice. For example, labetalol may not be the optimal medication for a patient with poorly controlled asthma due to its ability to cause bronchospasm.14,15 Methyldopa may not be the optimal medication for a patient with depression.16 Based on the available data, labetalol, nifedipine, and methyldopa are the best antihypertensive medications for pregnant patients.
Continue to: What is an optimal BP target when treating chronic hypertension in pregnancy?...
What is an optimal BP target when treating chronic hypertension in pregnancy?
When treating chronic hypertension in pregnant patients, a concern is that reducing maternal BP may decrease uteroplacental perfusion and result in fetal growth restriction. However, a recent trial reported that a BP treatment target < 140/90 mm Hg is associated with better outcomes for both mother and newborn than withholding antihypertension medications. In the trial, 2,408 women with chronic hypertension diagnosed before 20 weeks of gestation were randomly assigned to an active treatment group with prescription of antihypertension medicines to achieve a BP target of < 140/90 mm Hg; or to a control group where no antihypertension or no additional antihypertension treatment was prescribed unless BP was ≥ 160 mm Hg systolic or ≥ 105 mm Hg diastolic.9 The hypertension medications prescribed to the patients in the active treatment group were labetalol (63.2%), nifedipine (33.4%), amlodipine (1.7%), methyldopa (0.5%), hydrochlorothiazide (0.3%), metoprolol (0.2%), and missing/unknown/other (0.7%).9
If a patient in the control group developed severe hypertension, they were started on an antihypertension medicine and the BP treatment target was < 140/90 mm Hg. Compared with the control regimen, active treatment resulted in a significant decrease in the development of preeclampsia (24.4% vs 31.1%; risk ratio [RR], 0.79; 95% CI, 0.69-0.89), severe hypertension (36.1% vs 44.3%; RR, 0.82; 95% CI, 0.74-0.90), preterm birth < 37 weeks’ gestation (27.5% vs 31.4%; RR, 0.87; 95% CI, 0.77-0.99), preterm birth < 35 weeks’ gestation (12.2% vs 16.7%; odds ratio [OR], 0.69; 95% CI, 0.55-0.88), and low birth-weight (< 2,500 g) newborns (19.2% vs 23.1%; RR, 0.83; 95% CI, 0.71-0.97).9 The percentage of small for gestational age birth weight below the 10th percentile was similar in the treatment and control groups, 11.2% and 10.4%, respectively (adjusted RR, 1.04; 95% CI, 0.82-1.31).9 The number of patients who would need to be treated to prevent one primary-outcome event was 15.9 The investigators concluded that for pregnant patients with chronic hypertension, the optimal BP target is < 140/90 mm Hg.9
In pregnant patients with hypertension, BP may decrease immediately after birth. Following birth, BP tends to increase, reaching a peak 3 to 6 days postpartum.17,18 This pattern was observed in patients with and without preeclampsia in the index pregnancy. Among 136 patients without antepartum preeclampsia, the prevalence of a diastolic BP > 89 mm Hg was 5% and 15% on postpartum days 1 and 3, respectively.17 The postpartum rise in BP may be due to mobilization of water from the extravascular to the intravascular space and excretion of total body sodium that accumulated during pregnancy.19 In one study of 998 consecutive singleton cesarean births, 7.7% of the patients with no recorded elevated BP before delivery developed de novo hypertension postpartum.20 Compared with patients without antepartum or new onset postpartum hypertension, the patients who developed postpartum hypertension had a higher body mass index, were more likely to be Black and to have a history of type 2 diabetes. Compared with patients without antepartum or postpartum hypertension, the patients who developed de novo postpartum hypertension, had significantly elevated soluble fms-like tyrosine kinase-1 and significantly decreased placental growth factor, a pattern seen with preeclampsia.20 These results suggest that de novo postpartum hypertension may have molecular causes similar to preeclampsia.20
Postpartum hypertension should be treated with a medication that is thought to be safe for breastfeeding patients, including labetalol, nifedipine, or enalapril.21-23 The relative infant dose of labetalol, nifedipine, and enalapril is approximately 3.6%, ≤ 3.2%, and 1.1%, respectively.24 If the relative infant dose of a medication is < 10% it is generally considered to be compatible with breastfeeding.25
Many obstetricians have seldom prescribed enalapril, an ACE-I. The initial dose of enalapril is 5 mg or 10 mg daily. After initiation of treatment, the dose can be adjusted based on BP measurement. The maximal daily dose is 40 mg daily in one dose or two divided doses. Similar to other hypertension medicines, enalapril therapy may cause hypotension and dizziness. Enalapril should not be used by pregnant patients because it is associated with an increased risk of congenital anomalies and fetal demise.
Does a HDP increase the risk of developing chronic hypertension?
All obstetricians know that a patient with a history of a HDP is at an increased risk for developing chronic hypertension treated with a medication, but the magnitude of the risk is less well known. In a nationwide study in Denmark, the prevalence of chronic hypertension treated with medication 10 years after delivery among patients with a history of a HDP in their first pregnancy, was 14%, 21%, and 32%, if the first pregnancy occurred in the patient’s 20s, 30s, or 40s, respectively.26 The corresponding prevalence of chronic hypertension in patients without a history of a HDP was 4%, 6%, and 11%, if the first pregnancy occurred in the 20s, 30s, or 40s, respectively.26 Maternal age is an important predictor of who will develop chronic hypertension within 10 years following a pregnancy with a HDP.
In modern obstetric practice, the hypertensive disorders of pregnancy are prevalent and associated with increased maternal and newborn morbidity. Appropriate treatment of hypertension with labetalol, nifedipine, or methyldopa improves maternal and newborn health. Available evidence suggests that maintaining BP < 140/90 mm Hg during pregnancy for most patients is a practical goal with significant benefit. A significant public-health concern is that an increase in the prevalence of HDPs will eventually translate into an increase in chronic hypertension and the attendant complications of heart attack, heart failure, stroke, and renal insufficiency. Recognizing the increased prevalence of HDPs, ObGyns will need to alert patients to their long-term health risks and coordinate appropriate follow-up and treatment to optimize the future health of their patients. ●
- Ford ND, Cox S, Ko JY, et al. Hypertensive disorders in pregnancy and mortality at delivery hospitalization-United States, 2017-2019. Morb Mortal Week Report. 2022;71:585-591.
- Bruno AM, Allshouse AA, Metz TD, et al. Trends in hypertensive disorders of pregnancy in the United States from 1989 to 2020. Obstet Gynecol. 2022;140:83-86.
- Doleszar CM, McGrath JJ, Herzig AJM, et al. Perceived racial discrimination and hypertension: a comprehensive systematic review. Health Psychol. 2014;33:20-34.
- Centers for Disease Control and Prevention. A Closer Look at African American Men and High Blood Pressure Control; A Review of Psychosocial Factors and Systems-Level Interventions. Atlanta: U.S. Department of Health and Human Services; 2010.
- Boulet SL, Platner M, Joseph NT, et al. Hypertensive disorders of pregnancy, cesarean delivery and severe maternal morbidity in an urban safety-net population. Am J Epidemiol. 2020;189:1502-1511.
- Parikh NI, Gonzalez JM, Andreson CAM, et al. Adverse pregnancy outcomes and cardiovascular disease risk: unique opportunities for cardiovascular disease prevention in women: a scientific statement from the American Heart Association. Circulation. 2021;143:e902-e916.
- Okoth K, Chandan JS, Marshall T, et al. Association between the reproductive health of young women and cardiovascular disease later in life: umbrella review. BMJ. 2020;371:m3502.
- Fu J, Tomlinson G, Feig DS. Increased risk of major congenital malformations in early pregnancy uses of angiotensin-converting enzyme inhibitors or angiotensin receptor blockers: a meta-analysis. Diabetes Metab Res Rev. 2021;37:e3453.
- Tita AT, Szychowski JM, Boggess K, et al. Treatment for mild chronic hypertension during pregnancy. N Engl J Med. 2022;386:1781-1792.
- Baum T, Sybertz EJ. Pharmacology of labetalol in experimental animals. Am J Med. 1983;75:15-23.
- Khan KM, Patel JB, Schaefer TJ. StatPearls (Internet). StatPearls Publishing; 2022.
- Gupta M, Khalili. Methyldopa StatPearls (Internet). StatPearls Publishing; 2022.
- Bone JN, Sandhu A, Diablos ED, et al. Oral antihypertensives for non-severe pregnancy hypertension: systematic review, network meta-analysis and trial sequential analysis. Hypertension. 2022;79:614-628.
- Morales DR, Jackson C, Lipworth BJ, et al. Adverse respiratory effects of acute beta-blocker exposure in asthma: a systematic review and meta-analysis of randomized controlled trials. Chest. 2014;145:779-786.
- Huang KY, Tseng PT, Wu YC, et al. Do beta-adrenergic blocking agents increase asthma exacerbation? A network meta-analysis of randomized controlled trials. Sci Rep. 2021;11:452.
- Nayak AS, Nachane HB. Risk analysis of suicidal ideation and postpartum depression with antenatal alpha methyldopa use. Asian J Psychiatry. 2018;38:42-44.
- Walters BNJ, Thompson ME, Lee A, et al. Blood pressure in the puerperium. Clin Sci. 1986;71:589-594.
- Walters BNJ, Walters T. Hypertension in the puerperium. Lancet. 1987;2(8554):330.
- Magee L, von Dadelszen. Prevention and treatment of postpartum hypertension. Cochrane Database Syst Rev. 2013;CD004351.
- Goel A, Maski MR, Bajracharya S, et al. Epidemiology and mechanisms of de novo and persistent hypertension in the postpartum period. Circulation. 2015;132:1726-1733.
- Powles K, Gandhi S. Postpartum hypertension. CMAJ. 2017;189:E913.
- Tosounidou S, Gordon C. Medications in pregnancy and breastfeeding. Best Prac Res Clin Obstet Gynaecol. 2020;64:68-76.
- Anderson PO. Treating hypertension during breastfeeding. Breastfeed Med. 2018;13:95-96.
- Lexicomp web site. https://www.wolterskluwer.com/en/solutions/lexicomp.
- Ito S. Drug therapy for breast-feeding women. N Engl J Med. 2000;343:118-126.
- Behrens I, Basit S, Melbye M, et al. Risk of postpartum hypertension in women with a history of hypertensive disorders of pregnancy: nationwide cohort study. BMJ. 2017;358:j3078.

Hypertension in pregnancy is a major challenge in current obstetric practice. Based on an analysis of the National Inpatient Sample, the Centers for Disease Control and Prevention (CDC) recently reported that from 2017 to 2019 the prevalence of hypertensive disorders in pregnancy increased from 13.3% to 15.9% of hospital deliveries.1 During that same time period, the prevalence of pregnancy-associated hypertension, which includes preeclampsia, eclampsia, and gestational hypertension, increased from 10.8% to 13.0%.1 The prevalence of chronic hypertension increased from 2.0% to 2.3%.1 In 2017 and 2019, unspecified maternal hypertension was diagnosed in 0.5% and 0.6% of the sample, respectively.1
Bruno and colleagues reported a 3-fold increase in the prevalence of HDPs from 1989 to 2020, with an acceleration in the rate of increase from 2010 to 2020.2 The increase in prevalence of HDPs may be caused by an increase in the prevalence of advanced maternal age, obesity, and diabetes. Black patients are disproportionately impacted by both pregnancy-associated hypertension and chronic hypertension.1 In 2019, the prevalence of pregnancy-associated hypertension was greater among Black patients (15.6%), than White (12.1%), Hispanic (10.6%), or Asian or Pacific Islander patients (7.7%).1 Similarly, the prevalence of chronic hypertension was greater among Black patients (4.3%) than among White (2.0%), Hispanic (1.5%), or Asian or Pacific Islander patients (1.2%).1 Racial/ethnic differences in HDPs may be influenced by poverty; structural racism; or lack of access to care, diet, and obesity.3,4
HDPs are major contributors to maternal morbidity and mortality. The CDC reported that among maternal deaths occurring during the delivery hospitalization, 32% of the decedents had documented hypertension.1 HDPs are associated with an approximately 2.5-fold increased risk of a severe morbidity, a composite measure that includes blood transfusion, acute kidney injury, disseminated intravascular coagulation, sepsis, shock, and pulmonary edema.5 A history of HDPs is associated with an approximately 67% increase in the lifetime risk of cardiovascular disease, including coronary artery disease, stroke, peripheral vascular disease, and heart failure.6,7
What are the best antihypertensive medications for pregnancy?
All clinicians know that the use of angiotensin-converting-enzyme inhibitors (ACE-Is) and angiotensin-receptor-blockers (ARBs) are contraindicated in pregnancy because they cause major congenital anomalies, with an odds ratio of 1.8 (95% confidence interval [CI], 1.42-2.34), compared with no exposure.8 In addition, ACE-Is and ARBs increase the risk of stillbirth, with an odds ratio of 1.75 (95% CI, 1.21-2.53).8 No increase in congenital anomalies were detected for patients exposed to other antihypertensive medications.8 Prior to attempting conception, patients with chronic hypertension should discontinue ACE-Is and ARBs and initiate an alternative medication.
The most commonly used antihypertensive medications in pregnancy are labetalol, nifedipine, and methyldopa.9 Labetalol blocks the beta-1, beta-2, and alpha-1 adrenergic receptors.10 Nifedipine blocks calcium entry into cells through the L-type calcium channel.11 Methyldopa is a central nervous system alpha-2 adrenergic agonist.12 The dose range for these commonly used medications are labetalol 400 mg to 2,400 mg daily in divided doses every 8 to 12 hours, nifedipine extended-release 30 mg to 120 mg daily, and methyldopa 500 mg to 2 g daily in 2 to 4 divided doses. Some clinicians recommend prescribing divided doses of nifedipine extended release at doses ≥ 60 mg for patients who have bothersome adverse effects, hypotension following a single daily dose, or hypertension between single daily doses. The nifedipine extended release tablets should not be divided. If monotherapy with the maximal daily dose of labetalol does not achieve the blood pressure (BP) target, adding nifedipine as a second agent is an option.9 Similarly, if monotherapy with the maximal daily dose of nifedipine extended release does not achieve the BP target, adding labetalol as a second agent is an option.9
In a network meta-analysis of antihypertensive medications used in pregnancy, that included 61 trials and 6,923 participants, all the medications studied reduced the risk of developing severe hypertension by 30% to 70%.13 Sufficient data was available to also report that labetalol used to treat hypertension in pregnancy reduced the risk of developing proteinuria.13 Given similar efficacy among antihypertensive medications, patient comorbidities may influence the medication choice. For example, labetalol may not be the optimal medication for a patient with poorly controlled asthma due to its ability to cause bronchospasm.14,15 Methyldopa may not be the optimal medication for a patient with depression.16 Based on the available data, labetalol, nifedipine, and methyldopa are the best antihypertensive medications for pregnant patients.
Continue to: What is an optimal BP target when treating chronic hypertension in pregnancy?...
What is an optimal BP target when treating chronic hypertension in pregnancy?
When treating chronic hypertension in pregnant patients, a concern is that reducing maternal BP may decrease uteroplacental perfusion and result in fetal growth restriction. However, a recent trial reported that a BP treatment target < 140/90 mm Hg is associated with better outcomes for both mother and newborn than withholding antihypertension medications. In the trial, 2,408 women with chronic hypertension diagnosed before 20 weeks of gestation were randomly assigned to an active treatment group with prescription of antihypertension medicines to achieve a BP target of < 140/90 mm Hg; or to a control group where no antihypertension or no additional antihypertension treatment was prescribed unless BP was ≥ 160 mm Hg systolic or ≥ 105 mm Hg diastolic.9 The hypertension medications prescribed to the patients in the active treatment group were labetalol (63.2%), nifedipine (33.4%), amlodipine (1.7%), methyldopa (0.5%), hydrochlorothiazide (0.3%), metoprolol (0.2%), and missing/unknown/other (0.7%).9
If a patient in the control group developed severe hypertension, they were started on an antihypertension medicine and the BP treatment target was < 140/90 mm Hg. Compared with the control regimen, active treatment resulted in a significant decrease in the development of preeclampsia (24.4% vs 31.1%; risk ratio [RR], 0.79; 95% CI, 0.69-0.89), severe hypertension (36.1% vs 44.3%; RR, 0.82; 95% CI, 0.74-0.90), preterm birth < 37 weeks’ gestation (27.5% vs 31.4%; RR, 0.87; 95% CI, 0.77-0.99), preterm birth < 35 weeks’ gestation (12.2% vs 16.7%; odds ratio [OR], 0.69; 95% CI, 0.55-0.88), and low birth-weight (< 2,500 g) newborns (19.2% vs 23.1%; RR, 0.83; 95% CI, 0.71-0.97).9 The percentage of small for gestational age birth weight below the 10th percentile was similar in the treatment and control groups, 11.2% and 10.4%, respectively (adjusted RR, 1.04; 95% CI, 0.82-1.31).9 The number of patients who would need to be treated to prevent one primary-outcome event was 15.9 The investigators concluded that for pregnant patients with chronic hypertension, the optimal BP target is < 140/90 mm Hg.9
In pregnant patients with hypertension, BP may decrease immediately after birth. Following birth, BP tends to increase, reaching a peak 3 to 6 days postpartum.17,18 This pattern was observed in patients with and without preeclampsia in the index pregnancy. Among 136 patients without antepartum preeclampsia, the prevalence of a diastolic BP > 89 mm Hg was 5% and 15% on postpartum days 1 and 3, respectively.17 The postpartum rise in BP may be due to mobilization of water from the extravascular to the intravascular space and excretion of total body sodium that accumulated during pregnancy.19 In one study of 998 consecutive singleton cesarean births, 7.7% of the patients with no recorded elevated BP before delivery developed de novo hypertension postpartum.20 Compared with patients without antepartum or new onset postpartum hypertension, the patients who developed postpartum hypertension had a higher body mass index, were more likely to be Black and to have a history of type 2 diabetes. Compared with patients without antepartum or postpartum hypertension, the patients who developed de novo postpartum hypertension, had significantly elevated soluble fms-like tyrosine kinase-1 and significantly decreased placental growth factor, a pattern seen with preeclampsia.20 These results suggest that de novo postpartum hypertension may have molecular causes similar to preeclampsia.20
Postpartum hypertension should be treated with a medication that is thought to be safe for breastfeeding patients, including labetalol, nifedipine, or enalapril.21-23 The relative infant dose of labetalol, nifedipine, and enalapril is approximately 3.6%, ≤ 3.2%, and 1.1%, respectively.24 If the relative infant dose of a medication is < 10% it is generally considered to be compatible with breastfeeding.25
Many obstetricians have seldom prescribed enalapril, an ACE-I. The initial dose of enalapril is 5 mg or 10 mg daily. After initiation of treatment, the dose can be adjusted based on BP measurement. The maximal daily dose is 40 mg daily in one dose or two divided doses. Similar to other hypertension medicines, enalapril therapy may cause hypotension and dizziness. Enalapril should not be used by pregnant patients because it is associated with an increased risk of congenital anomalies and fetal demise.
Does a HDP increase the risk of developing chronic hypertension?
All obstetricians know that a patient with a history of a HDP is at an increased risk for developing chronic hypertension treated with a medication, but the magnitude of the risk is less well known. In a nationwide study in Denmark, the prevalence of chronic hypertension treated with medication 10 years after delivery among patients with a history of a HDP in their first pregnancy, was 14%, 21%, and 32%, if the first pregnancy occurred in the patient’s 20s, 30s, or 40s, respectively.26 The corresponding prevalence of chronic hypertension in patients without a history of a HDP was 4%, 6%, and 11%, if the first pregnancy occurred in the 20s, 30s, or 40s, respectively.26 Maternal age is an important predictor of who will develop chronic hypertension within 10 years following a pregnancy with a HDP.
In modern obstetric practice, the hypertensive disorders of pregnancy are prevalent and associated with increased maternal and newborn morbidity. Appropriate treatment of hypertension with labetalol, nifedipine, or methyldopa improves maternal and newborn health. Available evidence suggests that maintaining BP < 140/90 mm Hg during pregnancy for most patients is a practical goal with significant benefit. A significant public-health concern is that an increase in the prevalence of HDPs will eventually translate into an increase in chronic hypertension and the attendant complications of heart attack, heart failure, stroke, and renal insufficiency. Recognizing the increased prevalence of HDPs, ObGyns will need to alert patients to their long-term health risks and coordinate appropriate follow-up and treatment to optimize the future health of their patients. ●

Hypertension in pregnancy is a major challenge in current obstetric practice. Based on an analysis of the National Inpatient Sample, the Centers for Disease Control and Prevention (CDC) recently reported that from 2017 to 2019 the prevalence of hypertensive disorders in pregnancy increased from 13.3% to 15.9% of hospital deliveries.1 During that same time period, the prevalence of pregnancy-associated hypertension, which includes preeclampsia, eclampsia, and gestational hypertension, increased from 10.8% to 13.0%.1 The prevalence of chronic hypertension increased from 2.0% to 2.3%.1 In 2017 and 2019, unspecified maternal hypertension was diagnosed in 0.5% and 0.6% of the sample, respectively.1
Bruno and colleagues reported a 3-fold increase in the prevalence of HDPs from 1989 to 2020, with an acceleration in the rate of increase from 2010 to 2020.2 The increase in prevalence of HDPs may be caused by an increase in the prevalence of advanced maternal age, obesity, and diabetes. Black patients are disproportionately impacted by both pregnancy-associated hypertension and chronic hypertension.1 In 2019, the prevalence of pregnancy-associated hypertension was greater among Black patients (15.6%), than White (12.1%), Hispanic (10.6%), or Asian or Pacific Islander patients (7.7%).1 Similarly, the prevalence of chronic hypertension was greater among Black patients (4.3%) than among White (2.0%), Hispanic (1.5%), or Asian or Pacific Islander patients (1.2%).1 Racial/ethnic differences in HDPs may be influenced by poverty; structural racism; or lack of access to care, diet, and obesity.3,4
HDPs are major contributors to maternal morbidity and mortality. The CDC reported that among maternal deaths occurring during the delivery hospitalization, 32% of the decedents had documented hypertension.1 HDPs are associated with an approximately 2.5-fold increased risk of a severe morbidity, a composite measure that includes blood transfusion, acute kidney injury, disseminated intravascular coagulation, sepsis, shock, and pulmonary edema.5 A history of HDPs is associated with an approximately 67% increase in the lifetime risk of cardiovascular disease, including coronary artery disease, stroke, peripheral vascular disease, and heart failure.6,7
What are the best antihypertensive medications for pregnancy?
All clinicians know that the use of angiotensin-converting-enzyme inhibitors (ACE-Is) and angiotensin-receptor-blockers (ARBs) are contraindicated in pregnancy because they cause major congenital anomalies, with an odds ratio of 1.8 (95% confidence interval [CI], 1.42-2.34), compared with no exposure.8 In addition, ACE-Is and ARBs increase the risk of stillbirth, with an odds ratio of 1.75 (95% CI, 1.21-2.53).8 No increase in congenital anomalies were detected for patients exposed to other antihypertensive medications.8 Prior to attempting conception, patients with chronic hypertension should discontinue ACE-Is and ARBs and initiate an alternative medication.
The most commonly used antihypertensive medications in pregnancy are labetalol, nifedipine, and methyldopa.9 Labetalol blocks the beta-1, beta-2, and alpha-1 adrenergic receptors.10 Nifedipine blocks calcium entry into cells through the L-type calcium channel.11 Methyldopa is a central nervous system alpha-2 adrenergic agonist.12 The dose range for these commonly used medications are labetalol 400 mg to 2,400 mg daily in divided doses every 8 to 12 hours, nifedipine extended-release 30 mg to 120 mg daily, and methyldopa 500 mg to 2 g daily in 2 to 4 divided doses. Some clinicians recommend prescribing divided doses of nifedipine extended release at doses ≥ 60 mg for patients who have bothersome adverse effects, hypotension following a single daily dose, or hypertension between single daily doses. The nifedipine extended release tablets should not be divided. If monotherapy with the maximal daily dose of labetalol does not achieve the blood pressure (BP) target, adding nifedipine as a second agent is an option.9 Similarly, if monotherapy with the maximal daily dose of nifedipine extended release does not achieve the BP target, adding labetalol as a second agent is an option.9
In a network meta-analysis of antihypertensive medications used in pregnancy, that included 61 trials and 6,923 participants, all the medications studied reduced the risk of developing severe hypertension by 30% to 70%.13 Sufficient data was available to also report that labetalol used to treat hypertension in pregnancy reduced the risk of developing proteinuria.13 Given similar efficacy among antihypertensive medications, patient comorbidities may influence the medication choice. For example, labetalol may not be the optimal medication for a patient with poorly controlled asthma due to its ability to cause bronchospasm.14,15 Methyldopa may not be the optimal medication for a patient with depression.16 Based on the available data, labetalol, nifedipine, and methyldopa are the best antihypertensive medications for pregnant patients.
Continue to: What is an optimal BP target when treating chronic hypertension in pregnancy?...
What is an optimal BP target when treating chronic hypertension in pregnancy?
When treating chronic hypertension in pregnant patients, a concern is that reducing maternal BP may decrease uteroplacental perfusion and result in fetal growth restriction. However, a recent trial reported that a BP treatment target < 140/90 mm Hg is associated with better outcomes for both mother and newborn than withholding antihypertension medications. In the trial, 2,408 women with chronic hypertension diagnosed before 20 weeks of gestation were randomly assigned to an active treatment group with prescription of antihypertension medicines to achieve a BP target of < 140/90 mm Hg; or to a control group where no antihypertension or no additional antihypertension treatment was prescribed unless BP was ≥ 160 mm Hg systolic or ≥ 105 mm Hg diastolic.9 The hypertension medications prescribed to the patients in the active treatment group were labetalol (63.2%), nifedipine (33.4%), amlodipine (1.7%), methyldopa (0.5%), hydrochlorothiazide (0.3%), metoprolol (0.2%), and missing/unknown/other (0.7%).9
If a patient in the control group developed severe hypertension, they were started on an antihypertension medicine and the BP treatment target was < 140/90 mm Hg. Compared with the control regimen, active treatment resulted in a significant decrease in the development of preeclampsia (24.4% vs 31.1%; risk ratio [RR], 0.79; 95% CI, 0.69-0.89), severe hypertension (36.1% vs 44.3%; RR, 0.82; 95% CI, 0.74-0.90), preterm birth < 37 weeks’ gestation (27.5% vs 31.4%; RR, 0.87; 95% CI, 0.77-0.99), preterm birth < 35 weeks’ gestation (12.2% vs 16.7%; odds ratio [OR], 0.69; 95% CI, 0.55-0.88), and low birth-weight (< 2,500 g) newborns (19.2% vs 23.1%; RR, 0.83; 95% CI, 0.71-0.97).9 The percentage of small for gestational age birth weight below the 10th percentile was similar in the treatment and control groups, 11.2% and 10.4%, respectively (adjusted RR, 1.04; 95% CI, 0.82-1.31).9 The number of patients who would need to be treated to prevent one primary-outcome event was 15.9 The investigators concluded that for pregnant patients with chronic hypertension, the optimal BP target is < 140/90 mm Hg.9
In pregnant patients with hypertension, BP may decrease immediately after birth. Following birth, BP tends to increase, reaching a peak 3 to 6 days postpartum.17,18 This pattern was observed in patients with and without preeclampsia in the index pregnancy. Among 136 patients without antepartum preeclampsia, the prevalence of a diastolic BP > 89 mm Hg was 5% and 15% on postpartum days 1 and 3, respectively.17 The postpartum rise in BP may be due to mobilization of water from the extravascular to the intravascular space and excretion of total body sodium that accumulated during pregnancy.19 In one study of 998 consecutive singleton cesarean births, 7.7% of the patients with no recorded elevated BP before delivery developed de novo hypertension postpartum.20 Compared with patients without antepartum or new onset postpartum hypertension, the patients who developed postpartum hypertension had a higher body mass index, were more likely to be Black and to have a history of type 2 diabetes. Compared with patients without antepartum or postpartum hypertension, the patients who developed de novo postpartum hypertension, had significantly elevated soluble fms-like tyrosine kinase-1 and significantly decreased placental growth factor, a pattern seen with preeclampsia.20 These results suggest that de novo postpartum hypertension may have molecular causes similar to preeclampsia.20
Postpartum hypertension should be treated with a medication that is thought to be safe for breastfeeding patients, including labetalol, nifedipine, or enalapril.21-23 The relative infant dose of labetalol, nifedipine, and enalapril is approximately 3.6%, ≤ 3.2%, and 1.1%, respectively.24 If the relative infant dose of a medication is < 10% it is generally considered to be compatible with breastfeeding.25
Many obstetricians have seldom prescribed enalapril, an ACE-I. The initial dose of enalapril is 5 mg or 10 mg daily. After initiation of treatment, the dose can be adjusted based on BP measurement. The maximal daily dose is 40 mg daily in one dose or two divided doses. Similar to other hypertension medicines, enalapril therapy may cause hypotension and dizziness. Enalapril should not be used by pregnant patients because it is associated with an increased risk of congenital anomalies and fetal demise.
Does a HDP increase the risk of developing chronic hypertension?
All obstetricians know that a patient with a history of a HDP is at an increased risk for developing chronic hypertension treated with a medication, but the magnitude of the risk is less well known. In a nationwide study in Denmark, the prevalence of chronic hypertension treated with medication 10 years after delivery among patients with a history of a HDP in their first pregnancy, was 14%, 21%, and 32%, if the first pregnancy occurred in the patient’s 20s, 30s, or 40s, respectively.26 The corresponding prevalence of chronic hypertension in patients without a history of a HDP was 4%, 6%, and 11%, if the first pregnancy occurred in the 20s, 30s, or 40s, respectively.26 Maternal age is an important predictor of who will develop chronic hypertension within 10 years following a pregnancy with a HDP.
In modern obstetric practice, the hypertensive disorders of pregnancy are prevalent and associated with increased maternal and newborn morbidity. Appropriate treatment of hypertension with labetalol, nifedipine, or methyldopa improves maternal and newborn health. Available evidence suggests that maintaining BP < 140/90 mm Hg during pregnancy for most patients is a practical goal with significant benefit. A significant public-health concern is that an increase in the prevalence of HDPs will eventually translate into an increase in chronic hypertension and the attendant complications of heart attack, heart failure, stroke, and renal insufficiency. Recognizing the increased prevalence of HDPs, ObGyns will need to alert patients to their long-term health risks and coordinate appropriate follow-up and treatment to optimize the future health of their patients. ●
- Ford ND, Cox S, Ko JY, et al. Hypertensive disorders in pregnancy and mortality at delivery hospitalization-United States, 2017-2019. Morb Mortal Week Report. 2022;71:585-591.
- Bruno AM, Allshouse AA, Metz TD, et al. Trends in hypertensive disorders of pregnancy in the United States from 1989 to 2020. Obstet Gynecol. 2022;140:83-86.
- Doleszar CM, McGrath JJ, Herzig AJM, et al. Perceived racial discrimination and hypertension: a comprehensive systematic review. Health Psychol. 2014;33:20-34.
- Centers for Disease Control and Prevention. A Closer Look at African American Men and High Blood Pressure Control; A Review of Psychosocial Factors and Systems-Level Interventions. Atlanta: U.S. Department of Health and Human Services; 2010.
- Boulet SL, Platner M, Joseph NT, et al. Hypertensive disorders of pregnancy, cesarean delivery and severe maternal morbidity in an urban safety-net population. Am J Epidemiol. 2020;189:1502-1511.
- Parikh NI, Gonzalez JM, Andreson CAM, et al. Adverse pregnancy outcomes and cardiovascular disease risk: unique opportunities for cardiovascular disease prevention in women: a scientific statement from the American Heart Association. Circulation. 2021;143:e902-e916.
- Okoth K, Chandan JS, Marshall T, et al. Association between the reproductive health of young women and cardiovascular disease later in life: umbrella review. BMJ. 2020;371:m3502.
- Fu J, Tomlinson G, Feig DS. Increased risk of major congenital malformations in early pregnancy uses of angiotensin-converting enzyme inhibitors or angiotensin receptor blockers: a meta-analysis. Diabetes Metab Res Rev. 2021;37:e3453.
- Tita AT, Szychowski JM, Boggess K, et al. Treatment for mild chronic hypertension during pregnancy. N Engl J Med. 2022;386:1781-1792.
- Baum T, Sybertz EJ. Pharmacology of labetalol in experimental animals. Am J Med. 1983;75:15-23.
- Khan KM, Patel JB, Schaefer TJ. StatPearls (Internet). StatPearls Publishing; 2022.
- Gupta M, Khalili. Methyldopa StatPearls (Internet). StatPearls Publishing; 2022.
- Bone JN, Sandhu A, Diablos ED, et al. Oral antihypertensives for non-severe pregnancy hypertension: systematic review, network meta-analysis and trial sequential analysis. Hypertension. 2022;79:614-628.
- Morales DR, Jackson C, Lipworth BJ, et al. Adverse respiratory effects of acute beta-blocker exposure in asthma: a systematic review and meta-analysis of randomized controlled trials. Chest. 2014;145:779-786.
- Huang KY, Tseng PT, Wu YC, et al. Do beta-adrenergic blocking agents increase asthma exacerbation? A network meta-analysis of randomized controlled trials. Sci Rep. 2021;11:452.
- Nayak AS, Nachane HB. Risk analysis of suicidal ideation and postpartum depression with antenatal alpha methyldopa use. Asian J Psychiatry. 2018;38:42-44.
- Walters BNJ, Thompson ME, Lee A, et al. Blood pressure in the puerperium. Clin Sci. 1986;71:589-594.
- Walters BNJ, Walters T. Hypertension in the puerperium. Lancet. 1987;2(8554):330.
- Magee L, von Dadelszen. Prevention and treatment of postpartum hypertension. Cochrane Database Syst Rev. 2013;CD004351.
- Goel A, Maski MR, Bajracharya S, et al. Epidemiology and mechanisms of de novo and persistent hypertension in the postpartum period. Circulation. 2015;132:1726-1733.
- Powles K, Gandhi S. Postpartum hypertension. CMAJ. 2017;189:E913.
- Tosounidou S, Gordon C. Medications in pregnancy and breastfeeding. Best Prac Res Clin Obstet Gynaecol. 2020;64:68-76.
- Anderson PO. Treating hypertension during breastfeeding. Breastfeed Med. 2018;13:95-96.
- Lexicomp web site. https://www.wolterskluwer.com/en/solutions/lexicomp.
- Ito S. Drug therapy for breast-feeding women. N Engl J Med. 2000;343:118-126.
- Behrens I, Basit S, Melbye M, et al. Risk of postpartum hypertension in women with a history of hypertensive disorders of pregnancy: nationwide cohort study. BMJ. 2017;358:j3078.
- Ford ND, Cox S, Ko JY, et al. Hypertensive disorders in pregnancy and mortality at delivery hospitalization-United States, 2017-2019. Morb Mortal Week Report. 2022;71:585-591.
- Bruno AM, Allshouse AA, Metz TD, et al. Trends in hypertensive disorders of pregnancy in the United States from 1989 to 2020. Obstet Gynecol. 2022;140:83-86.
- Doleszar CM, McGrath JJ, Herzig AJM, et al. Perceived racial discrimination and hypertension: a comprehensive systematic review. Health Psychol. 2014;33:20-34.
- Centers for Disease Control and Prevention. A Closer Look at African American Men and High Blood Pressure Control; A Review of Psychosocial Factors and Systems-Level Interventions. Atlanta: U.S. Department of Health and Human Services; 2010.
- Boulet SL, Platner M, Joseph NT, et al. Hypertensive disorders of pregnancy, cesarean delivery and severe maternal morbidity in an urban safety-net population. Am J Epidemiol. 2020;189:1502-1511.
- Parikh NI, Gonzalez JM, Andreson CAM, et al. Adverse pregnancy outcomes and cardiovascular disease risk: unique opportunities for cardiovascular disease prevention in women: a scientific statement from the American Heart Association. Circulation. 2021;143:e902-e916.
- Okoth K, Chandan JS, Marshall T, et al. Association between the reproductive health of young women and cardiovascular disease later in life: umbrella review. BMJ. 2020;371:m3502.
- Fu J, Tomlinson G, Feig DS. Increased risk of major congenital malformations in early pregnancy uses of angiotensin-converting enzyme inhibitors or angiotensin receptor blockers: a meta-analysis. Diabetes Metab Res Rev. 2021;37:e3453.
- Tita AT, Szychowski JM, Boggess K, et al. Treatment for mild chronic hypertension during pregnancy. N Engl J Med. 2022;386:1781-1792.
- Baum T, Sybertz EJ. Pharmacology of labetalol in experimental animals. Am J Med. 1983;75:15-23.
- Khan KM, Patel JB, Schaefer TJ. StatPearls (Internet). StatPearls Publishing; 2022.
- Gupta M, Khalili. Methyldopa StatPearls (Internet). StatPearls Publishing; 2022.
- Bone JN, Sandhu A, Diablos ED, et al. Oral antihypertensives for non-severe pregnancy hypertension: systematic review, network meta-analysis and trial sequential analysis. Hypertension. 2022;79:614-628.
- Morales DR, Jackson C, Lipworth BJ, et al. Adverse respiratory effects of acute beta-blocker exposure in asthma: a systematic review and meta-analysis of randomized controlled trials. Chest. 2014;145:779-786.
- Huang KY, Tseng PT, Wu YC, et al. Do beta-adrenergic blocking agents increase asthma exacerbation? A network meta-analysis of randomized controlled trials. Sci Rep. 2021;11:452.
- Nayak AS, Nachane HB. Risk analysis of suicidal ideation and postpartum depression with antenatal alpha methyldopa use. Asian J Psychiatry. 2018;38:42-44.
- Walters BNJ, Thompson ME, Lee A, et al. Blood pressure in the puerperium. Clin Sci. 1986;71:589-594.
- Walters BNJ, Walters T. Hypertension in the puerperium. Lancet. 1987;2(8554):330.
- Magee L, von Dadelszen. Prevention and treatment of postpartum hypertension. Cochrane Database Syst Rev. 2013;CD004351.
- Goel A, Maski MR, Bajracharya S, et al. Epidemiology and mechanisms of de novo and persistent hypertension in the postpartum period. Circulation. 2015;132:1726-1733.
- Powles K, Gandhi S. Postpartum hypertension. CMAJ. 2017;189:E913.
- Tosounidou S, Gordon C. Medications in pregnancy and breastfeeding. Best Prac Res Clin Obstet Gynaecol. 2020;64:68-76.
- Anderson PO. Treating hypertension during breastfeeding. Breastfeed Med. 2018;13:95-96.
- Lexicomp web site. https://www.wolterskluwer.com/en/solutions/lexicomp.
- Ito S. Drug therapy for breast-feeding women. N Engl J Med. 2000;343:118-126.
- Behrens I, Basit S, Melbye M, et al. Risk of postpartum hypertension in women with a history of hypertensive disorders of pregnancy: nationwide cohort study. BMJ. 2017;358:j3078.