User login
‘Autoantibody Signature’ Flags MS Years Before Symptom Onset
, according to a new study.
Investigators screened blood samples from 250 individuals with MS drawn 5 years before and 1 year after symptom onset, profiled MS-related autoantibodies, and compared the sample with 250 matched controls.
A unique cluster of autoantibodies was found in 10% of people with MS, appearing up to 5 years before the onset of clinical symptoms and remaining higher 1 year after diagnosis.
“Our work demonstrates that a subset of MS patients has antibodies that react to a common protein motif, both before, during, and after diagnosis and symptom onset,” said lead investigator Colin R. Zamecnik, PhD, a postdoctoral researcher at UCSF School of Medicine, University of California, San Francisco.
Such a discovery could aid in early diagnosis, Dr. Zamecnik added. MS treatments “have gotten much better in the last 15-20 years and evidence shows early treatment can improve outcomes,” he said.
The study was published online in Nature Medicine.
Seeking Earlier Diagnosis
Previous research shows that nonspecific neurologic episodes occur more frequently in people who received an MS diagnosis later in life, pointing to the possibility of an MS prodrome, the authors noted.
These neurologic episodes may be indicative of ongoing neuroinflammatory processes in the preclinical period, they added. Studies in several other autoimmune diseases show that diagnostic autoantibodies can appear years before symptom onset. However, no such antibodies have previously been identified in MS patients.
To investigate, the researchers turned to data from a large, prospective incident MS cohort assembled during the Gulf War era in more than 10 million US military veterans.
Records of those with the earliest diagnosis (an average of 5 years before symptom onset) and 1 year after the first attack were analyzed, and matched controls were selected.
Investigators used a technique called phage display immunoprecipitation sequencing to screen human blood for antibodies. They conducted a whole-proteome autoantibody screen and serum neurofilament light (sNfL) measurements on these samples in both case patients and controls at the same time points.
Early Signs of Injury
In the preclinical serum samples, sNfL levels were higher nearer the date of diagnosis and significantly higher in post- versus pre-onset samples in people with MS. “Together, these data provide evidence that at least some people with MS exhibit early signs of neuroaxonal injury long before onset of symptoms,” the authors noted.
Analysis of the collection of peptides, described by the investigators as an “autoantibody signature,” was consistent over time and was present regardless of diagnosis.
Further analysis of the autoantibodies revealed a characteristic protein motif found in common viruses, including Epstein-Barr virus (EBV) and hepatitis C virus, among others.
The motif “shares remarkable similarity to those found on many pathogens that infect humans, including EBV, which is known to be a risk factor for development of MS,” Dr. Zamecnik said.
The researchers validated these findings by analyzing serum and cerebrospinal fluid samples from participants in ORIGINS, an MS cohort at the University of California, San Francisco, that enrolled patients at clinical onset. As with the other cohort, 10% of patients had the autoantibody signature.
The investigators added that the findings detail some of the first autoantigen-specific biomarkers found in preclinical MS.
“Taken together, our future work will focus on profiling these patients more closely over time to see how they differ from their counterparts and gives further evidence of viral-host crosstalk as a hallmark of this disease,” Dr. Zamecnik said.
Not Ready for Prime Time
Commenting on the findings, Bruce Bebo, PhD, executive vice president of research, National Multiple Sclerosis Society, said the study corroborates the “growing appreciation that MS has a prodrome.”
Such a discovery might “accelerate progress toward the possibility of treating MS ever-earlier in the course of the disease, or possibly even preventing MS from occurring in the first place,” he added.
Dr. Bebo, who was not involved in this research, noted that it was conducted at a single center, is only preliminary, and “has no immediate clinical applicability.”
Also, because this pattern was identified in only 10% of individuals with MS, “an additional hurdle is whether we can identify other patterns in greater numbers of people,” he added.
This work was supported by the Valhalla Foundation; the Weill Neurohub; the Westridge Foundation; the National Institute of Neurological Disorders and Stroke; the National Institute of Allergy and Infectious Diseases; National Multiple Sclerosis Society; the Department of Defense; the German Society of Multiple Sclerosis; the Water Cove Charitable Foundation; Tim and Laura O’Shaughnessy; the Littera Family; School of Medicine Dean’s Yearlong Fellowship, supported by residual funds from the Howard Hughes Medical Institute Medical Fellows at UCSF; the Chan Zuckerberg Biohub San Francisco; the John A. Watson Scholar Program at UCSF; the Hanna H. Gray Fellowship, Howard Hughes Medical Institute; the National Institutes of Health; and the University of California President’s Postdoctoral Fellowship Program. Dr. Zamecnik received funding toward this study from the National Multiple Sclerosis Society and the Water Cove Charitable Foundation. He declared no competing financial interests. The other authors’ disclosures are listed on the original paper. Dr. Bebo is the executive vice president of the National Multiple Sclerosis Society, which provided support for the study.
A version of this article appeared on Medscape.com.
, according to a new study.
Investigators screened blood samples from 250 individuals with MS drawn 5 years before and 1 year after symptom onset, profiled MS-related autoantibodies, and compared the sample with 250 matched controls.
A unique cluster of autoantibodies was found in 10% of people with MS, appearing up to 5 years before the onset of clinical symptoms and remaining higher 1 year after diagnosis.
“Our work demonstrates that a subset of MS patients has antibodies that react to a common protein motif, both before, during, and after diagnosis and symptom onset,” said lead investigator Colin R. Zamecnik, PhD, a postdoctoral researcher at UCSF School of Medicine, University of California, San Francisco.
Such a discovery could aid in early diagnosis, Dr. Zamecnik added. MS treatments “have gotten much better in the last 15-20 years and evidence shows early treatment can improve outcomes,” he said.
The study was published online in Nature Medicine.
Seeking Earlier Diagnosis
Previous research shows that nonspecific neurologic episodes occur more frequently in people who received an MS diagnosis later in life, pointing to the possibility of an MS prodrome, the authors noted.
These neurologic episodes may be indicative of ongoing neuroinflammatory processes in the preclinical period, they added. Studies in several other autoimmune diseases show that diagnostic autoantibodies can appear years before symptom onset. However, no such antibodies have previously been identified in MS patients.
To investigate, the researchers turned to data from a large, prospective incident MS cohort assembled during the Gulf War era in more than 10 million US military veterans.
Records of those with the earliest diagnosis (an average of 5 years before symptom onset) and 1 year after the first attack were analyzed, and matched controls were selected.
Investigators used a technique called phage display immunoprecipitation sequencing to screen human blood for antibodies. They conducted a whole-proteome autoantibody screen and serum neurofilament light (sNfL) measurements on these samples in both case patients and controls at the same time points.
Early Signs of Injury
In the preclinical serum samples, sNfL levels were higher nearer the date of diagnosis and significantly higher in post- versus pre-onset samples in people with MS. “Together, these data provide evidence that at least some people with MS exhibit early signs of neuroaxonal injury long before onset of symptoms,” the authors noted.
Analysis of the collection of peptides, described by the investigators as an “autoantibody signature,” was consistent over time and was present regardless of diagnosis.
Further analysis of the autoantibodies revealed a characteristic protein motif found in common viruses, including Epstein-Barr virus (EBV) and hepatitis C virus, among others.
The motif “shares remarkable similarity to those found on many pathogens that infect humans, including EBV, which is known to be a risk factor for development of MS,” Dr. Zamecnik said.
The researchers validated these findings by analyzing serum and cerebrospinal fluid samples from participants in ORIGINS, an MS cohort at the University of California, San Francisco, that enrolled patients at clinical onset. As with the other cohort, 10% of patients had the autoantibody signature.
The investigators added that the findings detail some of the first autoantigen-specific biomarkers found in preclinical MS.
“Taken together, our future work will focus on profiling these patients more closely over time to see how they differ from their counterparts and gives further evidence of viral-host crosstalk as a hallmark of this disease,” Dr. Zamecnik said.
Not Ready for Prime Time
Commenting on the findings, Bruce Bebo, PhD, executive vice president of research, National Multiple Sclerosis Society, said the study corroborates the “growing appreciation that MS has a prodrome.”
Such a discovery might “accelerate progress toward the possibility of treating MS ever-earlier in the course of the disease, or possibly even preventing MS from occurring in the first place,” he added.
Dr. Bebo, who was not involved in this research, noted that it was conducted at a single center, is only preliminary, and “has no immediate clinical applicability.”
Also, because this pattern was identified in only 10% of individuals with MS, “an additional hurdle is whether we can identify other patterns in greater numbers of people,” he added.
This work was supported by the Valhalla Foundation; the Weill Neurohub; the Westridge Foundation; the National Institute of Neurological Disorders and Stroke; the National Institute of Allergy and Infectious Diseases; National Multiple Sclerosis Society; the Department of Defense; the German Society of Multiple Sclerosis; the Water Cove Charitable Foundation; Tim and Laura O’Shaughnessy; the Littera Family; School of Medicine Dean’s Yearlong Fellowship, supported by residual funds from the Howard Hughes Medical Institute Medical Fellows at UCSF; the Chan Zuckerberg Biohub San Francisco; the John A. Watson Scholar Program at UCSF; the Hanna H. Gray Fellowship, Howard Hughes Medical Institute; the National Institutes of Health; and the University of California President’s Postdoctoral Fellowship Program. Dr. Zamecnik received funding toward this study from the National Multiple Sclerosis Society and the Water Cove Charitable Foundation. He declared no competing financial interests. The other authors’ disclosures are listed on the original paper. Dr. Bebo is the executive vice president of the National Multiple Sclerosis Society, which provided support for the study.
A version of this article appeared on Medscape.com.
, according to a new study.
Investigators screened blood samples from 250 individuals with MS drawn 5 years before and 1 year after symptom onset, profiled MS-related autoantibodies, and compared the sample with 250 matched controls.
A unique cluster of autoantibodies was found in 10% of people with MS, appearing up to 5 years before the onset of clinical symptoms and remaining higher 1 year after diagnosis.
“Our work demonstrates that a subset of MS patients has antibodies that react to a common protein motif, both before, during, and after diagnosis and symptom onset,” said lead investigator Colin R. Zamecnik, PhD, a postdoctoral researcher at UCSF School of Medicine, University of California, San Francisco.
Such a discovery could aid in early diagnosis, Dr. Zamecnik added. MS treatments “have gotten much better in the last 15-20 years and evidence shows early treatment can improve outcomes,” he said.
The study was published online in Nature Medicine.
Seeking Earlier Diagnosis
Previous research shows that nonspecific neurologic episodes occur more frequently in people who received an MS diagnosis later in life, pointing to the possibility of an MS prodrome, the authors noted.
These neurologic episodes may be indicative of ongoing neuroinflammatory processes in the preclinical period, they added. Studies in several other autoimmune diseases show that diagnostic autoantibodies can appear years before symptom onset. However, no such antibodies have previously been identified in MS patients.
To investigate, the researchers turned to data from a large, prospective incident MS cohort assembled during the Gulf War era in more than 10 million US military veterans.
Records of those with the earliest diagnosis (an average of 5 years before symptom onset) and 1 year after the first attack were analyzed, and matched controls were selected.
Investigators used a technique called phage display immunoprecipitation sequencing to screen human blood for antibodies. They conducted a whole-proteome autoantibody screen and serum neurofilament light (sNfL) measurements on these samples in both case patients and controls at the same time points.
Early Signs of Injury
In the preclinical serum samples, sNfL levels were higher nearer the date of diagnosis and significantly higher in post- versus pre-onset samples in people with MS. “Together, these data provide evidence that at least some people with MS exhibit early signs of neuroaxonal injury long before onset of symptoms,” the authors noted.
Analysis of the collection of peptides, described by the investigators as an “autoantibody signature,” was consistent over time and was present regardless of diagnosis.
Further analysis of the autoantibodies revealed a characteristic protein motif found in common viruses, including Epstein-Barr virus (EBV) and hepatitis C virus, among others.
The motif “shares remarkable similarity to those found on many pathogens that infect humans, including EBV, which is known to be a risk factor for development of MS,” Dr. Zamecnik said.
The researchers validated these findings by analyzing serum and cerebrospinal fluid samples from participants in ORIGINS, an MS cohort at the University of California, San Francisco, that enrolled patients at clinical onset. As with the other cohort, 10% of patients had the autoantibody signature.
The investigators added that the findings detail some of the first autoantigen-specific biomarkers found in preclinical MS.
“Taken together, our future work will focus on profiling these patients more closely over time to see how they differ from their counterparts and gives further evidence of viral-host crosstalk as a hallmark of this disease,” Dr. Zamecnik said.
Not Ready for Prime Time
Commenting on the findings, Bruce Bebo, PhD, executive vice president of research, National Multiple Sclerosis Society, said the study corroborates the “growing appreciation that MS has a prodrome.”
Such a discovery might “accelerate progress toward the possibility of treating MS ever-earlier in the course of the disease, or possibly even preventing MS from occurring in the first place,” he added.
Dr. Bebo, who was not involved in this research, noted that it was conducted at a single center, is only preliminary, and “has no immediate clinical applicability.”
Also, because this pattern was identified in only 10% of individuals with MS, “an additional hurdle is whether we can identify other patterns in greater numbers of people,” he added.
This work was supported by the Valhalla Foundation; the Weill Neurohub; the Westridge Foundation; the National Institute of Neurological Disorders and Stroke; the National Institute of Allergy and Infectious Diseases; National Multiple Sclerosis Society; the Department of Defense; the German Society of Multiple Sclerosis; the Water Cove Charitable Foundation; Tim and Laura O’Shaughnessy; the Littera Family; School of Medicine Dean’s Yearlong Fellowship, supported by residual funds from the Howard Hughes Medical Institute Medical Fellows at UCSF; the Chan Zuckerberg Biohub San Francisco; the John A. Watson Scholar Program at UCSF; the Hanna H. Gray Fellowship, Howard Hughes Medical Institute; the National Institutes of Health; and the University of California President’s Postdoctoral Fellowship Program. Dr. Zamecnik received funding toward this study from the National Multiple Sclerosis Society and the Water Cove Charitable Foundation. He declared no competing financial interests. The other authors’ disclosures are listed on the original paper. Dr. Bebo is the executive vice president of the National Multiple Sclerosis Society, which provided support for the study.
A version of this article appeared on Medscape.com.
FROM NATURE MEDICINE
Antidepressants and Dementia Risk: Reassuring Data
TOPLINE:
, new research suggests.
METHODOLOGY:
- Investigators studied 5511 individuals (58% women; mean age, 71 years) from the Rotterdam study, an ongoing prospective population-based cohort study.
- Participants were free from dementia at baseline, and incident dementia was monitored from baseline until 2018 with repeated cognitive assessments using the Mini-Mental Status Examination (MMSE) and the Geriatric Mental Schedule, as well as MRIs.
- Information on participants’ antidepressant use was extracted from pharmacy records from 1992 until baseline (2002-2008).
- During a mean follow-up of 10 years, 12% of participants developed dementia.
TAKEAWAY:
- Overall, 17% of participants had used antidepressants during the roughly 10-year period prior to baseline, and 4.1% were still using antidepressants at baseline.
- Medication use at baseline was more common in women than in men (21% vs 18%), and use increased with age: From 2.1% in participants aged between 45 and 50 years to 4.5% in those older than 80 years.
- After adjustment for confounders, there was no association between antidepressant use and dementia risk (hazard ratio [HR], 1.14; 95% CI, 0.92-1.41), accelerated cognitive decline, or atrophy of white and gray matter.
- However, tricyclic antidepressant use was associated with increased dementia risk (HR, 1.36; 95% CI, 1.01-1.83) compared with the use of selective serotonin reuptake inhibitors (HR, 1.12; 95% CI, 0.81-1.54).
IN PRACTICE:
“Although prescription of antidepressant medication in older individuals, in particular those with some cognitive impairment, may have acute symptomatic anticholinergic effects that warrant consideration in clinical practice, our results show that long-term antidepressant use does not have lasting effects on cognition or brain health in older adults without indication of cognitive impairment,” the authors wrote.
SOURCE:
Frank J. Wolters, MD, of the Department of Epidemiology and the Department of Radiology and Nuclear Medicine and Alzheimer Center, Erasmus University Medical Center, Rotterdam, the Netherlands, was the senior author on this study that was published online in Alzheimer’s and Dementia.
LIMITATIONS:
Limitations included the concern that although exclusion of participants with MMSE < 26 at baseline prevented reversed causation (ie, antidepressant use in response to depression during the prodromal phase of dementia), it may have introduced selection bias by disregarding the effects of antidepressant use prior to baseline and excluding participants with lower education.
DISCLOSURES:
This study was conducted as part of the Netherlands Consortium of Dementia Cohorts, which receives funding in the context of Deltaplan Dementie from ZonMW Memorabel and Alzheimer Nederland. Further funding was also obtained from the Stichting Erasmus Trustfonds. This study was further supported by a 2020 NARSAD Young Investigator Grant from the Brain & Behavior Research Foundation. The authors reported no conflicts of interest or relevant financial relationships.
A version of this article appeared on Medscape.com.
TOPLINE:
, new research suggests.
METHODOLOGY:
- Investigators studied 5511 individuals (58% women; mean age, 71 years) from the Rotterdam study, an ongoing prospective population-based cohort study.
- Participants were free from dementia at baseline, and incident dementia was monitored from baseline until 2018 with repeated cognitive assessments using the Mini-Mental Status Examination (MMSE) and the Geriatric Mental Schedule, as well as MRIs.
- Information on participants’ antidepressant use was extracted from pharmacy records from 1992 until baseline (2002-2008).
- During a mean follow-up of 10 years, 12% of participants developed dementia.
TAKEAWAY:
- Overall, 17% of participants had used antidepressants during the roughly 10-year period prior to baseline, and 4.1% were still using antidepressants at baseline.
- Medication use at baseline was more common in women than in men (21% vs 18%), and use increased with age: From 2.1% in participants aged between 45 and 50 years to 4.5% in those older than 80 years.
- After adjustment for confounders, there was no association between antidepressant use and dementia risk (hazard ratio [HR], 1.14; 95% CI, 0.92-1.41), accelerated cognitive decline, or atrophy of white and gray matter.
- However, tricyclic antidepressant use was associated with increased dementia risk (HR, 1.36; 95% CI, 1.01-1.83) compared with the use of selective serotonin reuptake inhibitors (HR, 1.12; 95% CI, 0.81-1.54).
IN PRACTICE:
“Although prescription of antidepressant medication in older individuals, in particular those with some cognitive impairment, may have acute symptomatic anticholinergic effects that warrant consideration in clinical practice, our results show that long-term antidepressant use does not have lasting effects on cognition or brain health in older adults without indication of cognitive impairment,” the authors wrote.
SOURCE:
Frank J. Wolters, MD, of the Department of Epidemiology and the Department of Radiology and Nuclear Medicine and Alzheimer Center, Erasmus University Medical Center, Rotterdam, the Netherlands, was the senior author on this study that was published online in Alzheimer’s and Dementia.
LIMITATIONS:
Limitations included the concern that although exclusion of participants with MMSE < 26 at baseline prevented reversed causation (ie, antidepressant use in response to depression during the prodromal phase of dementia), it may have introduced selection bias by disregarding the effects of antidepressant use prior to baseline and excluding participants with lower education.
DISCLOSURES:
This study was conducted as part of the Netherlands Consortium of Dementia Cohorts, which receives funding in the context of Deltaplan Dementie from ZonMW Memorabel and Alzheimer Nederland. Further funding was also obtained from the Stichting Erasmus Trustfonds. This study was further supported by a 2020 NARSAD Young Investigator Grant from the Brain & Behavior Research Foundation. The authors reported no conflicts of interest or relevant financial relationships.
A version of this article appeared on Medscape.com.
TOPLINE:
, new research suggests.
METHODOLOGY:
- Investigators studied 5511 individuals (58% women; mean age, 71 years) from the Rotterdam study, an ongoing prospective population-based cohort study.
- Participants were free from dementia at baseline, and incident dementia was monitored from baseline until 2018 with repeated cognitive assessments using the Mini-Mental Status Examination (MMSE) and the Geriatric Mental Schedule, as well as MRIs.
- Information on participants’ antidepressant use was extracted from pharmacy records from 1992 until baseline (2002-2008).
- During a mean follow-up of 10 years, 12% of participants developed dementia.
TAKEAWAY:
- Overall, 17% of participants had used antidepressants during the roughly 10-year period prior to baseline, and 4.1% were still using antidepressants at baseline.
- Medication use at baseline was more common in women than in men (21% vs 18%), and use increased with age: From 2.1% in participants aged between 45 and 50 years to 4.5% in those older than 80 years.
- After adjustment for confounders, there was no association between antidepressant use and dementia risk (hazard ratio [HR], 1.14; 95% CI, 0.92-1.41), accelerated cognitive decline, or atrophy of white and gray matter.
- However, tricyclic antidepressant use was associated with increased dementia risk (HR, 1.36; 95% CI, 1.01-1.83) compared with the use of selective serotonin reuptake inhibitors (HR, 1.12; 95% CI, 0.81-1.54).
IN PRACTICE:
“Although prescription of antidepressant medication in older individuals, in particular those with some cognitive impairment, may have acute symptomatic anticholinergic effects that warrant consideration in clinical practice, our results show that long-term antidepressant use does not have lasting effects on cognition or brain health in older adults without indication of cognitive impairment,” the authors wrote.
SOURCE:
Frank J. Wolters, MD, of the Department of Epidemiology and the Department of Radiology and Nuclear Medicine and Alzheimer Center, Erasmus University Medical Center, Rotterdam, the Netherlands, was the senior author on this study that was published online in Alzheimer’s and Dementia.
LIMITATIONS:
Limitations included the concern that although exclusion of participants with MMSE < 26 at baseline prevented reversed causation (ie, antidepressant use in response to depression during the prodromal phase of dementia), it may have introduced selection bias by disregarding the effects of antidepressant use prior to baseline and excluding participants with lower education.
DISCLOSURES:
This study was conducted as part of the Netherlands Consortium of Dementia Cohorts, which receives funding in the context of Deltaplan Dementie from ZonMW Memorabel and Alzheimer Nederland. Further funding was also obtained from the Stichting Erasmus Trustfonds. This study was further supported by a 2020 NARSAD Young Investigator Grant from the Brain & Behavior Research Foundation. The authors reported no conflicts of interest or relevant financial relationships.
A version of this article appeared on Medscape.com.
Mandatory DMV Reporting Tied to Dementia Underdiagnosis
, new research suggests.
Investigators found that primary care physicians (PCPs) in states with clinician reporting mandates had a 59% higher probability of underdiagnosing dementia compared with their counterparts in states that require patients to self-report or that have no reporting mandates.
“Our findings in this cross-sectional study raise concerns about potential adverse effects of mandatory clinician reporting for dementia diagnosis and underscore the need for careful consideration of the effect of such policies,” wrote the investigators, led by Soeren Mattke, MD, DSc, director of the USC Brain Health Observatory and research professor of economics at the University of Southern California, Los Angeles.
The study was published online in JAMA Network Open.
Lack of Guidance
As the US population ages, the number of older drivers is increasing, with 55.8 million drivers 65 years old or older. Approximately 7 million people in this age group have dementia — an estimate that is expected to increase to nearly 12 million by 2040.
The aging population raises a “critical policy question” about how to ensure road safety. Although the American Medical Association’s Code of Ethics outlines a physician’s obligation to identify drivers with medical impairments that impede safe driving, guidance restricting cognitively impaired drivers from driving is lacking.
In addition, evidence as to whether cognitive impairment indeed poses a threat to driving safety is mixed and has led to a lack of uniform policies with respect to reporting dementia.
Four states explicitly require clinicians to report dementia diagnoses to the DMV, which will then determine the patient’s fitness to drive, whereas 14 states require people with dementia to self-report. The remaining states have no explicit reporting requirements.
The issue of mandatory reporting is controversial, the researchers noted. On the one hand, physicians could protect patients and others by reporting potentially unsafe drivers.
On the other hand, evidence of an association with lower accident risks in patients with dementia is sparse and mandatory reporting may adversely affect physician-patient relationships. Empirical evidence for unintended consequences of reporting laws is lacking.
To examine the potential link between dementia underdiagnosis and mandatory reporting policies, the investigators analyzed the 100% data from the Medicare fee-for-service program and Medicare Advantage plans from 2017 to 2019, which included 223,036 PCPs with a panel of 25 or more Medicare patients.
The researchers examined dementia diagnosis rates in the patient panel of PCPs, rather than neurologists or gerontologists, regardless of who documented the diagnosis. Dr. Mattke said that it is possible that the diagnosis was established after referral to a specialist.
Each physician’s expected number of dementia cases was estimated using a predictive model based on patient characteristics. The researchers then compared the estimate with observed dementia diagnoses, thereby identifying clinicians who underdiagnosed dementia after sampling errors were accounted for.
‘Heavy-Handed Interference’
The researchers adjusted for several covariates potentially associated with a clinician’s probability of underdiagnosing dementia. These included sex, office location, practice specialty, racial/ethnic composition of the patient panel, and percentage of patients dually eligible for Medicare and Medicaid. The table shows PCP characteristics.
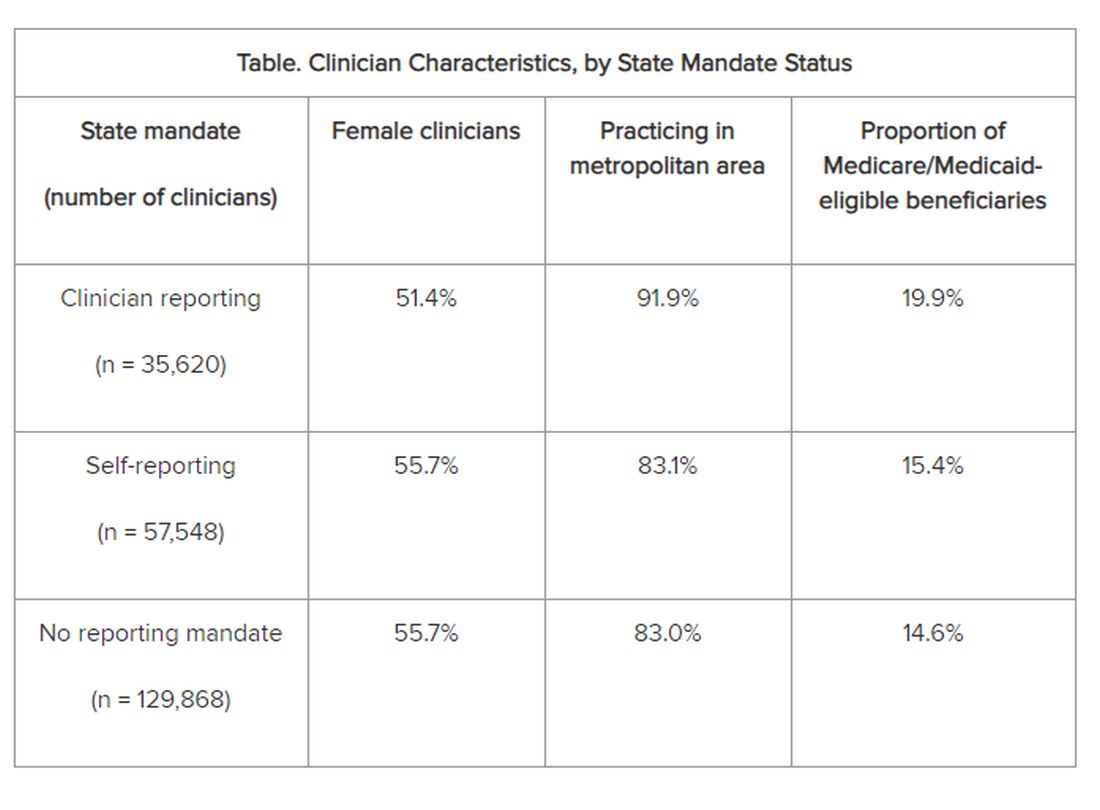
Adjusted results showed that PCPs practicing in states with clinician reporting mandates had a 12.4% (95% confidence interval [CI], 10.5%-14.2%) probability of underdiagnosing dementia versus 7.8% (95% CI, 6.9%-8.7%) in states with self-reporting and 7.7% (95% CI, 6.9%-8.4%) in states with no mandates, translating into a 4–percentage point difference (P < .001).
“Our study is the first to provide empirical evidence for the potential adverse effects of reporting policies,” the researchers noted. “Although we found that some clinicians underdiagnosed dementia regardless of state mandates, the key finding of this study reveals that primary care clinicians who practice in states with clinician reporting mandates were 59% more likely to do so…compared with those states with no reporting requirements…or driver self-reporting requirements.”
The investigators suggested that one potential explanation for underdiagnosis is patient resistance to cognitive testing. If patients were aware that the clinician was obligated by law to report their dementia diagnosis to the DMV, “they might be more inclined to conceal their symptoms or refuse further assessments, in addition to the general stigma and resistance to a formal assessment after a positive dementia screening result.”
“The findings suggest that policymakers might want to rethink those physician reporting mandates, since we also could not find conclusive evidence that they improve road safety,” Dr. Mattke said. “Maybe patients and their physicians can arrive at a sensible approach to determine driving fitness without such heavy-handed interference.”
However, he cautioned that the findings are not definitive and further study is needed before firm recommendations either for or against mandatory reporting.
In addition, the researchers noted several study limitations. One is that dementia underdiagnosis may also be associated with factors not captured in their model, including physician-patient relationships, health literacy, or language barriers.
However, Dr. Mattke noted, “ my sense is that those unobservable factors are not systematically related to state reporting policies and having omitted them would therefore not bias our results.”
Experts Weigh In
Commenting on the research, Morgan Daven, MA, the Alzheimer’s Association vice president of health systems, said that dementia is widely and significantly underdiagnosed, and not only in the states with dementia reporting mandates. Many factors may contribute to underdiagnosis, and although the study shows an association between reporting mandates and underdiagnosis, it does not demonstrate causation.
That said, Mr. Daven added, “fear and stigma related to dementia may inhibit the clinician, the patient, and their family from pursuing detection and diagnosis for dementia. As a society, we need to address dementia fear and stigma for all parties.”
He noted that useful tools include healthcare policies, workforce training, public awareness and education, and public policies to mitigate fear and stigma and their negative effects on diagnosis, care, support, and communication.
A potential study limitation is that it relied only on diagnoses by PCPs. Mr. Daven noted that the diagnosis of Alzheimer’ disease — the most common cause of dementia — is confirmation of amyloid buildup via a biomarker test, using PET or cerebrospinal fluid analysis.
“Both of these tests are extremely limited in their use and accessibility in a primary care setting. Inclusion of diagnoses by dementia specialists would provide a more complete picture,” he said.
Mr. Daven added that the Alzheimer’s Association encourages families to proactively discuss driving and other disease-related safety concerns as soon as possible. The Alzheimer’s Association Dementia and Driving webpage offers tips and strategies to discuss driving concerns with a family member.
In an accompanying editorial, Donald Redelmeier, MD, MS(HSR), and Vidhi Bhatt, BSc, both of the Department of Medicine, University of Toronto, differentiate the mandate for physicians to warn patients with dementia about traffic safety from the mandate for reporting child maltreatment, gunshot victims, or communicable diseases. They noted that mandated warnings “are not easy, can engender patient dissatisfaction, and need to be handled with tact.”
Yet, they pointed out, “breaking bad news is what practicing medicine entails.” They emphasized that, regardless of government mandates, “counseling patients for more road safety is an essential skill for clinicians in diverse states who hope to help their patients avoid becoming more traffic statistics.”
Research reported in this publication was supported by Genentech, a member of the Roche Group, and a grant from the National Institute on Aging of the National Institutes of Health. Dr. Mattke reported receiving grants from Genentech for a research contract with USC during the conduct of the study; personal fees from Eisai, Biogen, C2N, Novo Nordisk, Novartis, and Roche Genentech; and serving on the Senscio Systems board of directors, ALZpath scientific advisory board, AiCure scientific advisory board, and Boston Millennia Partners scientific advisory board outside the submitted work. The other authors’ disclosures are listed on the original paper. The editorial was supported by the Canada Research Chair in Medical Decision Sciences, the Canadian Institutes of Health Research, Kimel-Schatzky Traumatic Brain Injury Research Fund, and the Graduate Diploma Program in Health Research at the University of Toronto. The editorial authors report no other relevant financial relationships.
A version of this article appeared on Medscape.com.
, new research suggests.
Investigators found that primary care physicians (PCPs) in states with clinician reporting mandates had a 59% higher probability of underdiagnosing dementia compared with their counterparts in states that require patients to self-report or that have no reporting mandates.
“Our findings in this cross-sectional study raise concerns about potential adverse effects of mandatory clinician reporting for dementia diagnosis and underscore the need for careful consideration of the effect of such policies,” wrote the investigators, led by Soeren Mattke, MD, DSc, director of the USC Brain Health Observatory and research professor of economics at the University of Southern California, Los Angeles.
The study was published online in JAMA Network Open.
Lack of Guidance
As the US population ages, the number of older drivers is increasing, with 55.8 million drivers 65 years old or older. Approximately 7 million people in this age group have dementia — an estimate that is expected to increase to nearly 12 million by 2040.
The aging population raises a “critical policy question” about how to ensure road safety. Although the American Medical Association’s Code of Ethics outlines a physician’s obligation to identify drivers with medical impairments that impede safe driving, guidance restricting cognitively impaired drivers from driving is lacking.
In addition, evidence as to whether cognitive impairment indeed poses a threat to driving safety is mixed and has led to a lack of uniform policies with respect to reporting dementia.
Four states explicitly require clinicians to report dementia diagnoses to the DMV, which will then determine the patient’s fitness to drive, whereas 14 states require people with dementia to self-report. The remaining states have no explicit reporting requirements.
The issue of mandatory reporting is controversial, the researchers noted. On the one hand, physicians could protect patients and others by reporting potentially unsafe drivers.
On the other hand, evidence of an association with lower accident risks in patients with dementia is sparse and mandatory reporting may adversely affect physician-patient relationships. Empirical evidence for unintended consequences of reporting laws is lacking.
To examine the potential link between dementia underdiagnosis and mandatory reporting policies, the investigators analyzed the 100% data from the Medicare fee-for-service program and Medicare Advantage plans from 2017 to 2019, which included 223,036 PCPs with a panel of 25 or more Medicare patients.
The researchers examined dementia diagnosis rates in the patient panel of PCPs, rather than neurologists or gerontologists, regardless of who documented the diagnosis. Dr. Mattke said that it is possible that the diagnosis was established after referral to a specialist.
Each physician’s expected number of dementia cases was estimated using a predictive model based on patient characteristics. The researchers then compared the estimate with observed dementia diagnoses, thereby identifying clinicians who underdiagnosed dementia after sampling errors were accounted for.
‘Heavy-Handed Interference’
The researchers adjusted for several covariates potentially associated with a clinician’s probability of underdiagnosing dementia. These included sex, office location, practice specialty, racial/ethnic composition of the patient panel, and percentage of patients dually eligible for Medicare and Medicaid. The table shows PCP characteristics.

Adjusted results showed that PCPs practicing in states with clinician reporting mandates had a 12.4% (95% confidence interval [CI], 10.5%-14.2%) probability of underdiagnosing dementia versus 7.8% (95% CI, 6.9%-8.7%) in states with self-reporting and 7.7% (95% CI, 6.9%-8.4%) in states with no mandates, translating into a 4–percentage point difference (P < .001).
“Our study is the first to provide empirical evidence for the potential adverse effects of reporting policies,” the researchers noted. “Although we found that some clinicians underdiagnosed dementia regardless of state mandates, the key finding of this study reveals that primary care clinicians who practice in states with clinician reporting mandates were 59% more likely to do so…compared with those states with no reporting requirements…or driver self-reporting requirements.”
The investigators suggested that one potential explanation for underdiagnosis is patient resistance to cognitive testing. If patients were aware that the clinician was obligated by law to report their dementia diagnosis to the DMV, “they might be more inclined to conceal their symptoms or refuse further assessments, in addition to the general stigma and resistance to a formal assessment after a positive dementia screening result.”
“The findings suggest that policymakers might want to rethink those physician reporting mandates, since we also could not find conclusive evidence that they improve road safety,” Dr. Mattke said. “Maybe patients and their physicians can arrive at a sensible approach to determine driving fitness without such heavy-handed interference.”
However, he cautioned that the findings are not definitive and further study is needed before firm recommendations either for or against mandatory reporting.
In addition, the researchers noted several study limitations. One is that dementia underdiagnosis may also be associated with factors not captured in their model, including physician-patient relationships, health literacy, or language barriers.
However, Dr. Mattke noted, “ my sense is that those unobservable factors are not systematically related to state reporting policies and having omitted them would therefore not bias our results.”
Experts Weigh In
Commenting on the research, Morgan Daven, MA, the Alzheimer’s Association vice president of health systems, said that dementia is widely and significantly underdiagnosed, and not only in the states with dementia reporting mandates. Many factors may contribute to underdiagnosis, and although the study shows an association between reporting mandates and underdiagnosis, it does not demonstrate causation.
That said, Mr. Daven added, “fear and stigma related to dementia may inhibit the clinician, the patient, and their family from pursuing detection and diagnosis for dementia. As a society, we need to address dementia fear and stigma for all parties.”
He noted that useful tools include healthcare policies, workforce training, public awareness and education, and public policies to mitigate fear and stigma and their negative effects on diagnosis, care, support, and communication.
A potential study limitation is that it relied only on diagnoses by PCPs. Mr. Daven noted that the diagnosis of Alzheimer’ disease — the most common cause of dementia — is confirmation of amyloid buildup via a biomarker test, using PET or cerebrospinal fluid analysis.
“Both of these tests are extremely limited in their use and accessibility in a primary care setting. Inclusion of diagnoses by dementia specialists would provide a more complete picture,” he said.
Mr. Daven added that the Alzheimer’s Association encourages families to proactively discuss driving and other disease-related safety concerns as soon as possible. The Alzheimer’s Association Dementia and Driving webpage offers tips and strategies to discuss driving concerns with a family member.
In an accompanying editorial, Donald Redelmeier, MD, MS(HSR), and Vidhi Bhatt, BSc, both of the Department of Medicine, University of Toronto, differentiate the mandate for physicians to warn patients with dementia about traffic safety from the mandate for reporting child maltreatment, gunshot victims, or communicable diseases. They noted that mandated warnings “are not easy, can engender patient dissatisfaction, and need to be handled with tact.”
Yet, they pointed out, “breaking bad news is what practicing medicine entails.” They emphasized that, regardless of government mandates, “counseling patients for more road safety is an essential skill for clinicians in diverse states who hope to help their patients avoid becoming more traffic statistics.”
Research reported in this publication was supported by Genentech, a member of the Roche Group, and a grant from the National Institute on Aging of the National Institutes of Health. Dr. Mattke reported receiving grants from Genentech for a research contract with USC during the conduct of the study; personal fees from Eisai, Biogen, C2N, Novo Nordisk, Novartis, and Roche Genentech; and serving on the Senscio Systems board of directors, ALZpath scientific advisory board, AiCure scientific advisory board, and Boston Millennia Partners scientific advisory board outside the submitted work. The other authors’ disclosures are listed on the original paper. The editorial was supported by the Canada Research Chair in Medical Decision Sciences, the Canadian Institutes of Health Research, Kimel-Schatzky Traumatic Brain Injury Research Fund, and the Graduate Diploma Program in Health Research at the University of Toronto. The editorial authors report no other relevant financial relationships.
A version of this article appeared on Medscape.com.
, new research suggests.
Investigators found that primary care physicians (PCPs) in states with clinician reporting mandates had a 59% higher probability of underdiagnosing dementia compared with their counterparts in states that require patients to self-report or that have no reporting mandates.
“Our findings in this cross-sectional study raise concerns about potential adverse effects of mandatory clinician reporting for dementia diagnosis and underscore the need for careful consideration of the effect of such policies,” wrote the investigators, led by Soeren Mattke, MD, DSc, director of the USC Brain Health Observatory and research professor of economics at the University of Southern California, Los Angeles.
The study was published online in JAMA Network Open.
Lack of Guidance
As the US population ages, the number of older drivers is increasing, with 55.8 million drivers 65 years old or older. Approximately 7 million people in this age group have dementia — an estimate that is expected to increase to nearly 12 million by 2040.
The aging population raises a “critical policy question” about how to ensure road safety. Although the American Medical Association’s Code of Ethics outlines a physician’s obligation to identify drivers with medical impairments that impede safe driving, guidance restricting cognitively impaired drivers from driving is lacking.
In addition, evidence as to whether cognitive impairment indeed poses a threat to driving safety is mixed and has led to a lack of uniform policies with respect to reporting dementia.
Four states explicitly require clinicians to report dementia diagnoses to the DMV, which will then determine the patient’s fitness to drive, whereas 14 states require people with dementia to self-report. The remaining states have no explicit reporting requirements.
The issue of mandatory reporting is controversial, the researchers noted. On the one hand, physicians could protect patients and others by reporting potentially unsafe drivers.
On the other hand, evidence of an association with lower accident risks in patients with dementia is sparse and mandatory reporting may adversely affect physician-patient relationships. Empirical evidence for unintended consequences of reporting laws is lacking.
To examine the potential link between dementia underdiagnosis and mandatory reporting policies, the investigators analyzed the 100% data from the Medicare fee-for-service program and Medicare Advantage plans from 2017 to 2019, which included 223,036 PCPs with a panel of 25 or more Medicare patients.
The researchers examined dementia diagnosis rates in the patient panel of PCPs, rather than neurologists or gerontologists, regardless of who documented the diagnosis. Dr. Mattke said that it is possible that the diagnosis was established after referral to a specialist.
Each physician’s expected number of dementia cases was estimated using a predictive model based on patient characteristics. The researchers then compared the estimate with observed dementia diagnoses, thereby identifying clinicians who underdiagnosed dementia after sampling errors were accounted for.
‘Heavy-Handed Interference’
The researchers adjusted for several covariates potentially associated with a clinician’s probability of underdiagnosing dementia. These included sex, office location, practice specialty, racial/ethnic composition of the patient panel, and percentage of patients dually eligible for Medicare and Medicaid. The table shows PCP characteristics.

Adjusted results showed that PCPs practicing in states with clinician reporting mandates had a 12.4% (95% confidence interval [CI], 10.5%-14.2%) probability of underdiagnosing dementia versus 7.8% (95% CI, 6.9%-8.7%) in states with self-reporting and 7.7% (95% CI, 6.9%-8.4%) in states with no mandates, translating into a 4–percentage point difference (P < .001).
“Our study is the first to provide empirical evidence for the potential adverse effects of reporting policies,” the researchers noted. “Although we found that some clinicians underdiagnosed dementia regardless of state mandates, the key finding of this study reveals that primary care clinicians who practice in states with clinician reporting mandates were 59% more likely to do so…compared with those states with no reporting requirements…or driver self-reporting requirements.”
The investigators suggested that one potential explanation for underdiagnosis is patient resistance to cognitive testing. If patients were aware that the clinician was obligated by law to report their dementia diagnosis to the DMV, “they might be more inclined to conceal their symptoms or refuse further assessments, in addition to the general stigma and resistance to a formal assessment after a positive dementia screening result.”
“The findings suggest that policymakers might want to rethink those physician reporting mandates, since we also could not find conclusive evidence that they improve road safety,” Dr. Mattke said. “Maybe patients and their physicians can arrive at a sensible approach to determine driving fitness without such heavy-handed interference.”
However, he cautioned that the findings are not definitive and further study is needed before firm recommendations either for or against mandatory reporting.
In addition, the researchers noted several study limitations. One is that dementia underdiagnosis may also be associated with factors not captured in their model, including physician-patient relationships, health literacy, or language barriers.
However, Dr. Mattke noted, “ my sense is that those unobservable factors are not systematically related to state reporting policies and having omitted them would therefore not bias our results.”
Experts Weigh In
Commenting on the research, Morgan Daven, MA, the Alzheimer’s Association vice president of health systems, said that dementia is widely and significantly underdiagnosed, and not only in the states with dementia reporting mandates. Many factors may contribute to underdiagnosis, and although the study shows an association between reporting mandates and underdiagnosis, it does not demonstrate causation.
That said, Mr. Daven added, “fear and stigma related to dementia may inhibit the clinician, the patient, and their family from pursuing detection and diagnosis for dementia. As a society, we need to address dementia fear and stigma for all parties.”
He noted that useful tools include healthcare policies, workforce training, public awareness and education, and public policies to mitigate fear and stigma and their negative effects on diagnosis, care, support, and communication.
A potential study limitation is that it relied only on diagnoses by PCPs. Mr. Daven noted that the diagnosis of Alzheimer’ disease — the most common cause of dementia — is confirmation of amyloid buildup via a biomarker test, using PET or cerebrospinal fluid analysis.
“Both of these tests are extremely limited in their use and accessibility in a primary care setting. Inclusion of diagnoses by dementia specialists would provide a more complete picture,” he said.
Mr. Daven added that the Alzheimer’s Association encourages families to proactively discuss driving and other disease-related safety concerns as soon as possible. The Alzheimer’s Association Dementia and Driving webpage offers tips and strategies to discuss driving concerns with a family member.
In an accompanying editorial, Donald Redelmeier, MD, MS(HSR), and Vidhi Bhatt, BSc, both of the Department of Medicine, University of Toronto, differentiate the mandate for physicians to warn patients with dementia about traffic safety from the mandate for reporting child maltreatment, gunshot victims, or communicable diseases. They noted that mandated warnings “are not easy, can engender patient dissatisfaction, and need to be handled with tact.”
Yet, they pointed out, “breaking bad news is what practicing medicine entails.” They emphasized that, regardless of government mandates, “counseling patients for more road safety is an essential skill for clinicians in diverse states who hope to help their patients avoid becoming more traffic statistics.”
Research reported in this publication was supported by Genentech, a member of the Roche Group, and a grant from the National Institute on Aging of the National Institutes of Health. Dr. Mattke reported receiving grants from Genentech for a research contract with USC during the conduct of the study; personal fees from Eisai, Biogen, C2N, Novo Nordisk, Novartis, and Roche Genentech; and serving on the Senscio Systems board of directors, ALZpath scientific advisory board, AiCure scientific advisory board, and Boston Millennia Partners scientific advisory board outside the submitted work. The other authors’ disclosures are listed on the original paper. The editorial was supported by the Canada Research Chair in Medical Decision Sciences, the Canadian Institutes of Health Research, Kimel-Schatzky Traumatic Brain Injury Research Fund, and the Graduate Diploma Program in Health Research at the University of Toronto. The editorial authors report no other relevant financial relationships.
A version of this article appeared on Medscape.com.
From JAMA Network Open
Few Cancer Survivors Meet ACS Nutrition, Exercise Guidelines
TOPLINE:
METHODOLOGY:
- The ACS has published nutrition and exercise guidelines for cancer survivors, which include recommendations to maintain a healthy weight and diet, cut out alcohol, and participate in regular physical activities. Engaging in these behaviors is associated with longer survival among cancer survivors, but whether survivors follow these nutrition and activity recommendations has not been systematically tracked.
- Researchers evaluated data on 10,020 individuals (mean age, 64.2 years) who had completed cancer treatment. Data came from the Behavioral Risk Factor Surveillance System telephone-based survey administered in 2017, 2019, and 2021, which represents 2.7 million cancer survivors.
- The researchers estimated survivors’ adherence to guidelines across four domains: Weight, physical activity, fruit and vegetable consumption, and alcohol intake. Factors associated with adherence were also evaluated.
- Overall, 9,121 survivors (91%) completed questionnaires for all four domains.
TAKEAWAY:
Only 4% of patients (365 of 9121) followed ACS guidelines in all four categories.
When assessing adherence to each category, the researchers found that 72% of cancer survivors reported engaging in recommended levels of physical activity, 68% maintained a nonobese weight, 50% said they did not consume alcohol, and 12% said they consumed recommended quantities of fruits and vegetables.
Compared with people in the general population, cancer survivors generally engaged in fewer healthy behaviors than those who had never been diagnosed with cancer.
The authors identified certain factors associated with greater guideline adherence, including female sex, older age, Black (vs White) race, and higher education level (college graduate).
IN PRACTICE:
This study highlights a potential “gap between published guidelines regarding behavioral modifications for cancer survivors and uptake of these behaviors,” the authors wrote, adding that “it is essential for oncologists and general internists to improve widespread and systematic counseling on these guidelines to improve uptake of healthy behaviors in this vulnerable patient population.”
SOURCE:
This work, led by Carter Baughman, MD, from the Division of Internal Medicine at Beth Israel Deaconess Medical Center, Boston, Massachusetts, was published online in JAMA Oncology.
LIMITATIONS:
The authors reported several study limitations, most notably that self-reported data may introduce biases.
DISCLOSURES:
The study funding source was not reported. One author received grants from the US Highbush Blueberry Council outside the submitted work. No other disclosures were reported.
A version of this article appeared on Medscape.com.
TOPLINE:
METHODOLOGY:
- The ACS has published nutrition and exercise guidelines for cancer survivors, which include recommendations to maintain a healthy weight and diet, cut out alcohol, and participate in regular physical activities. Engaging in these behaviors is associated with longer survival among cancer survivors, but whether survivors follow these nutrition and activity recommendations has not been systematically tracked.
- Researchers evaluated data on 10,020 individuals (mean age, 64.2 years) who had completed cancer treatment. Data came from the Behavioral Risk Factor Surveillance System telephone-based survey administered in 2017, 2019, and 2021, which represents 2.7 million cancer survivors.
- The researchers estimated survivors’ adherence to guidelines across four domains: Weight, physical activity, fruit and vegetable consumption, and alcohol intake. Factors associated with adherence were also evaluated.
- Overall, 9,121 survivors (91%) completed questionnaires for all four domains.
TAKEAWAY:
Only 4% of patients (365 of 9121) followed ACS guidelines in all four categories.
When assessing adherence to each category, the researchers found that 72% of cancer survivors reported engaging in recommended levels of physical activity, 68% maintained a nonobese weight, 50% said they did not consume alcohol, and 12% said they consumed recommended quantities of fruits and vegetables.
Compared with people in the general population, cancer survivors generally engaged in fewer healthy behaviors than those who had never been diagnosed with cancer.
The authors identified certain factors associated with greater guideline adherence, including female sex, older age, Black (vs White) race, and higher education level (college graduate).
IN PRACTICE:
This study highlights a potential “gap between published guidelines regarding behavioral modifications for cancer survivors and uptake of these behaviors,” the authors wrote, adding that “it is essential for oncologists and general internists to improve widespread and systematic counseling on these guidelines to improve uptake of healthy behaviors in this vulnerable patient population.”
SOURCE:
This work, led by Carter Baughman, MD, from the Division of Internal Medicine at Beth Israel Deaconess Medical Center, Boston, Massachusetts, was published online in JAMA Oncology.
LIMITATIONS:
The authors reported several study limitations, most notably that self-reported data may introduce biases.
DISCLOSURES:
The study funding source was not reported. One author received grants from the US Highbush Blueberry Council outside the submitted work. No other disclosures were reported.
A version of this article appeared on Medscape.com.
TOPLINE:
METHODOLOGY:
- The ACS has published nutrition and exercise guidelines for cancer survivors, which include recommendations to maintain a healthy weight and diet, cut out alcohol, and participate in regular physical activities. Engaging in these behaviors is associated with longer survival among cancer survivors, but whether survivors follow these nutrition and activity recommendations has not been systematically tracked.
- Researchers evaluated data on 10,020 individuals (mean age, 64.2 years) who had completed cancer treatment. Data came from the Behavioral Risk Factor Surveillance System telephone-based survey administered in 2017, 2019, and 2021, which represents 2.7 million cancer survivors.
- The researchers estimated survivors’ adherence to guidelines across four domains: Weight, physical activity, fruit and vegetable consumption, and alcohol intake. Factors associated with adherence were also evaluated.
- Overall, 9,121 survivors (91%) completed questionnaires for all four domains.
TAKEAWAY:
Only 4% of patients (365 of 9121) followed ACS guidelines in all four categories.
When assessing adherence to each category, the researchers found that 72% of cancer survivors reported engaging in recommended levels of physical activity, 68% maintained a nonobese weight, 50% said they did not consume alcohol, and 12% said they consumed recommended quantities of fruits and vegetables.
Compared with people in the general population, cancer survivors generally engaged in fewer healthy behaviors than those who had never been diagnosed with cancer.
The authors identified certain factors associated with greater guideline adherence, including female sex, older age, Black (vs White) race, and higher education level (college graduate).
IN PRACTICE:
This study highlights a potential “gap between published guidelines regarding behavioral modifications for cancer survivors and uptake of these behaviors,” the authors wrote, adding that “it is essential for oncologists and general internists to improve widespread and systematic counseling on these guidelines to improve uptake of healthy behaviors in this vulnerable patient population.”
SOURCE:
This work, led by Carter Baughman, MD, from the Division of Internal Medicine at Beth Israel Deaconess Medical Center, Boston, Massachusetts, was published online in JAMA Oncology.
LIMITATIONS:
The authors reported several study limitations, most notably that self-reported data may introduce biases.
DISCLOSURES:
The study funding source was not reported. One author received grants from the US Highbush Blueberry Council outside the submitted work. No other disclosures were reported.
A version of this article appeared on Medscape.com.
Vaccine Safety and DMT for Highly Active Multiple Sclerosis: New Data
, new research shows.
The study, the first to examine vaccine safety and immunogenicity in highly active MS, revealed high seroprotection rates following receipt of vaccines for COVID-19 and hepatitis A and B, regardless of the duration of treatment with natalizumab.
On the basis of these findings, investigators created an algorithm that clinicians can use to map an immunization schedule in patients who might otherwise delay initiation of disease-modifying therapy until they are fully vaccinated.
“We observed seroprotection rates exceeding 90% for hepatitis A and B, and mRNA COVID-19 vaccines, and all vaccines demonstrated a favorable safety profile, with no exacerbation of disease activity detected,” said lead author René Carvajal, MD, of the Department of Neurology-Neuroimmunology, Multiple Sclerosis Centre of Catalonia (Cemcat), Hospital Universitari Vall d’Hebron, Universitat Autònoma de Barcelona, Barcelona, Spain. “This points to potential benefits for patients with highly active MS who require both immunization and high-efficacy therapies that may impact vaccine responses.”
The study was published online in JAMA Network Open.
A Controversial Issue
Today’s high-efficacy therapies for MS may increase the risk of acquiring new infections, reactivate latent pathogens, or worsen ongoing infectious conditions, and immunogenicity of vaccination can be compromised by immunosuppressive agents, particularly CD20 therapies, researchers noted.
As a result, many clinicians opt to delay initiation of such therapies until vaccination schedules are complete to avoid exposure to vaccine-preventable infections. But delaying treatment can potentially affect disease progression.
Reports of disease worsening following vaccination “have raised controversy around vaccine safety,” the authors wrote. The issue is especially relevant to those with highly active MS due to the scarcity of available data in this population.
The motivation for the study “stemmed from the complex balance clinicians face between initiating highly effective therapies promptly in patients with highly active MS and ensuring adequate protection against preventable infections through vaccination,” Dr. Carvajal said.
High Seroprotection Rate
Researchers analyzed data on 60 patients (mean age, 43 years; 44 female; mean disease duration, 17 years) participating in one of two prospectively followed cohorts: The Barcelona Clinically Isolated Syndromes Inception Cohort and the Barcelona Treatment Cohort. Data included demographic, clinical, radiologic, and biological data as well as regular clinical assessments, evaluations of the Expanded Disability Status Scale (EDSS), and MRI scans.
Patients enrolled in the current study had received at least one of these vaccines between September 2016 and February 2022: hepatitis A virus (HAV), hepatitis B virus (HBV; enhanced immunity high load or adjuvanted), or COVID-19 (BNT162b2 [Pfizer-BioNTech], mRAN-1273 [Moderna], or ChAdOx1-S [recombinant; AstraZeneca]).
The researchers conducted a retrospective, self-controlled analysis to compare the annualized relapse rate, EDSS score, and new T2 lesions counts during the 12 months before and after vaccination in patients with short- and long-term treatment duration.
They also compared John Cunningham virus serostatus between the two periods, as well as immunoglobulin G titers for each vaccine.
The global seroprotection rate was 93% (95% CI, 86%-98%). Individual vaccine rates were 92% for HAV, 93% for HBV, and 100% for COVID-19.
There was a significant reduction between the pre- and postvaccination periods in mean relapse rates (P = .004) and median number of new T2 lesions (P = .01).
There were no changes in EDSS scores before and after vaccinations and duration of natalizumab treatment had no impact on safety and immunogenicity.
‘Viable Option’
The researchers used their findings to create a proposed algorithm to inform immunization decisions in patients with highly active MS who require prompt initiation of high-efficacy disease-modifying therapy.
The algorithm is “integrated into a risk-minimization strategy tailored for patients with highly active MS, emphasizing in this case the pivotal role of natalizumab in averting treatment delays and providing adequate protection against potentially severe infections,” Dr. Carvajal said.
Participants who initiated or continued treatment with natalizumab completed their vaccination regimen without any incidents of progressive multifocal leukoencephalopathy (PML) or disease activity rebound following natalizumab discontinuation.
This suggests that using natalizumab for a brief duration might be a “viable option to contemplate,” the authors noted.
Commenting on the findings, Grace Gombolay, MD, assistant professor of pediatrics in the Division of Pediatric Neurology and director of the Pediatric Neuroimmunology and Multiple Sclerosis Clinic, Emory University, Atlanta, Georgia, said the study “demonstrates that vaccines are safe and do not trigger attacks in patients with MS on natalizumab, and that immunity — as measured by antibodies — is preserved in MS patients who receive natalizumab.”
This “contrasts with other treatments, as decreased antibody responses in COVID-19 are noted in certain treatments,” said Dr. Gombolay, who was not part of the study. “If both disease control and immunity against infection are the goals for the patient, then natalizumab is a reasonable option.”
“However, this must be balanced with other considerations,” she added, including the risk for PML and pregnancy.
This study was supported by grants from the European Committee for Treatment and Research in Multiple Sclerosis, Instituto de Salud Carlos III, and the European Union. Dr. Carvajal reported receiving grants from Vall d’Hebron Institut de Recerca and the European Committee for Treatment and Research in Multiple Sclerosis and honoraria from Roche, Novartis, BIIB-Colombia, Merck, and Sanofi outside the submitted work. Dr. Gombolay serves as media editor for Pediatric Neurology and as associate editor of the Annals of the Child Neurology Society. She is also a part-time CDC consultant for acute flaccid myelitis and received an honorarium as a speaker at the Georgia Neurological Society meeting, sponsored by Academic CME and TG Therapeutics.
A version of this article appeared on Medscape.com.
, new research shows.
The study, the first to examine vaccine safety and immunogenicity in highly active MS, revealed high seroprotection rates following receipt of vaccines for COVID-19 and hepatitis A and B, regardless of the duration of treatment with natalizumab.
On the basis of these findings, investigators created an algorithm that clinicians can use to map an immunization schedule in patients who might otherwise delay initiation of disease-modifying therapy until they are fully vaccinated.
“We observed seroprotection rates exceeding 90% for hepatitis A and B, and mRNA COVID-19 vaccines, and all vaccines demonstrated a favorable safety profile, with no exacerbation of disease activity detected,” said lead author René Carvajal, MD, of the Department of Neurology-Neuroimmunology, Multiple Sclerosis Centre of Catalonia (Cemcat), Hospital Universitari Vall d’Hebron, Universitat Autònoma de Barcelona, Barcelona, Spain. “This points to potential benefits for patients with highly active MS who require both immunization and high-efficacy therapies that may impact vaccine responses.”
The study was published online in JAMA Network Open.
A Controversial Issue
Today’s high-efficacy therapies for MS may increase the risk of acquiring new infections, reactivate latent pathogens, or worsen ongoing infectious conditions, and immunogenicity of vaccination can be compromised by immunosuppressive agents, particularly CD20 therapies, researchers noted.
As a result, many clinicians opt to delay initiation of such therapies until vaccination schedules are complete to avoid exposure to vaccine-preventable infections. But delaying treatment can potentially affect disease progression.
Reports of disease worsening following vaccination “have raised controversy around vaccine safety,” the authors wrote. The issue is especially relevant to those with highly active MS due to the scarcity of available data in this population.
The motivation for the study “stemmed from the complex balance clinicians face between initiating highly effective therapies promptly in patients with highly active MS and ensuring adequate protection against preventable infections through vaccination,” Dr. Carvajal said.
High Seroprotection Rate
Researchers analyzed data on 60 patients (mean age, 43 years; 44 female; mean disease duration, 17 years) participating in one of two prospectively followed cohorts: The Barcelona Clinically Isolated Syndromes Inception Cohort and the Barcelona Treatment Cohort. Data included demographic, clinical, radiologic, and biological data as well as regular clinical assessments, evaluations of the Expanded Disability Status Scale (EDSS), and MRI scans.
Patients enrolled in the current study had received at least one of these vaccines between September 2016 and February 2022: hepatitis A virus (HAV), hepatitis B virus (HBV; enhanced immunity high load or adjuvanted), or COVID-19 (BNT162b2 [Pfizer-BioNTech], mRAN-1273 [Moderna], or ChAdOx1-S [recombinant; AstraZeneca]).
The researchers conducted a retrospective, self-controlled analysis to compare the annualized relapse rate, EDSS score, and new T2 lesions counts during the 12 months before and after vaccination in patients with short- and long-term treatment duration.
They also compared John Cunningham virus serostatus between the two periods, as well as immunoglobulin G titers for each vaccine.
The global seroprotection rate was 93% (95% CI, 86%-98%). Individual vaccine rates were 92% for HAV, 93% for HBV, and 100% for COVID-19.
There was a significant reduction between the pre- and postvaccination periods in mean relapse rates (P = .004) and median number of new T2 lesions (P = .01).
There were no changes in EDSS scores before and after vaccinations and duration of natalizumab treatment had no impact on safety and immunogenicity.
‘Viable Option’
The researchers used their findings to create a proposed algorithm to inform immunization decisions in patients with highly active MS who require prompt initiation of high-efficacy disease-modifying therapy.
The algorithm is “integrated into a risk-minimization strategy tailored for patients with highly active MS, emphasizing in this case the pivotal role of natalizumab in averting treatment delays and providing adequate protection against potentially severe infections,” Dr. Carvajal said.
Participants who initiated or continued treatment with natalizumab completed their vaccination regimen without any incidents of progressive multifocal leukoencephalopathy (PML) or disease activity rebound following natalizumab discontinuation.
This suggests that using natalizumab for a brief duration might be a “viable option to contemplate,” the authors noted.
Commenting on the findings, Grace Gombolay, MD, assistant professor of pediatrics in the Division of Pediatric Neurology and director of the Pediatric Neuroimmunology and Multiple Sclerosis Clinic, Emory University, Atlanta, Georgia, said the study “demonstrates that vaccines are safe and do not trigger attacks in patients with MS on natalizumab, and that immunity — as measured by antibodies — is preserved in MS patients who receive natalizumab.”
This “contrasts with other treatments, as decreased antibody responses in COVID-19 are noted in certain treatments,” said Dr. Gombolay, who was not part of the study. “If both disease control and immunity against infection are the goals for the patient, then natalizumab is a reasonable option.”
“However, this must be balanced with other considerations,” she added, including the risk for PML and pregnancy.
This study was supported by grants from the European Committee for Treatment and Research in Multiple Sclerosis, Instituto de Salud Carlos III, and the European Union. Dr. Carvajal reported receiving grants from Vall d’Hebron Institut de Recerca and the European Committee for Treatment and Research in Multiple Sclerosis and honoraria from Roche, Novartis, BIIB-Colombia, Merck, and Sanofi outside the submitted work. Dr. Gombolay serves as media editor for Pediatric Neurology and as associate editor of the Annals of the Child Neurology Society. She is also a part-time CDC consultant for acute flaccid myelitis and received an honorarium as a speaker at the Georgia Neurological Society meeting, sponsored by Academic CME and TG Therapeutics.
A version of this article appeared on Medscape.com.
, new research shows.
The study, the first to examine vaccine safety and immunogenicity in highly active MS, revealed high seroprotection rates following receipt of vaccines for COVID-19 and hepatitis A and B, regardless of the duration of treatment with natalizumab.
On the basis of these findings, investigators created an algorithm that clinicians can use to map an immunization schedule in patients who might otherwise delay initiation of disease-modifying therapy until they are fully vaccinated.
“We observed seroprotection rates exceeding 90% for hepatitis A and B, and mRNA COVID-19 vaccines, and all vaccines demonstrated a favorable safety profile, with no exacerbation of disease activity detected,” said lead author René Carvajal, MD, of the Department of Neurology-Neuroimmunology, Multiple Sclerosis Centre of Catalonia (Cemcat), Hospital Universitari Vall d’Hebron, Universitat Autònoma de Barcelona, Barcelona, Spain. “This points to potential benefits for patients with highly active MS who require both immunization and high-efficacy therapies that may impact vaccine responses.”
The study was published online in JAMA Network Open.
A Controversial Issue
Today’s high-efficacy therapies for MS may increase the risk of acquiring new infections, reactivate latent pathogens, or worsen ongoing infectious conditions, and immunogenicity of vaccination can be compromised by immunosuppressive agents, particularly CD20 therapies, researchers noted.
As a result, many clinicians opt to delay initiation of such therapies until vaccination schedules are complete to avoid exposure to vaccine-preventable infections. But delaying treatment can potentially affect disease progression.
Reports of disease worsening following vaccination “have raised controversy around vaccine safety,” the authors wrote. The issue is especially relevant to those with highly active MS due to the scarcity of available data in this population.
The motivation for the study “stemmed from the complex balance clinicians face between initiating highly effective therapies promptly in patients with highly active MS and ensuring adequate protection against preventable infections through vaccination,” Dr. Carvajal said.
High Seroprotection Rate
Researchers analyzed data on 60 patients (mean age, 43 years; 44 female; mean disease duration, 17 years) participating in one of two prospectively followed cohorts: The Barcelona Clinically Isolated Syndromes Inception Cohort and the Barcelona Treatment Cohort. Data included demographic, clinical, radiologic, and biological data as well as regular clinical assessments, evaluations of the Expanded Disability Status Scale (EDSS), and MRI scans.
Patients enrolled in the current study had received at least one of these vaccines between September 2016 and February 2022: hepatitis A virus (HAV), hepatitis B virus (HBV; enhanced immunity high load or adjuvanted), or COVID-19 (BNT162b2 [Pfizer-BioNTech], mRAN-1273 [Moderna], or ChAdOx1-S [recombinant; AstraZeneca]).
The researchers conducted a retrospective, self-controlled analysis to compare the annualized relapse rate, EDSS score, and new T2 lesions counts during the 12 months before and after vaccination in patients with short- and long-term treatment duration.
They also compared John Cunningham virus serostatus between the two periods, as well as immunoglobulin G titers for each vaccine.
The global seroprotection rate was 93% (95% CI, 86%-98%). Individual vaccine rates were 92% for HAV, 93% for HBV, and 100% for COVID-19.
There was a significant reduction between the pre- and postvaccination periods in mean relapse rates (P = .004) and median number of new T2 lesions (P = .01).
There were no changes in EDSS scores before and after vaccinations and duration of natalizumab treatment had no impact on safety and immunogenicity.
‘Viable Option’
The researchers used their findings to create a proposed algorithm to inform immunization decisions in patients with highly active MS who require prompt initiation of high-efficacy disease-modifying therapy.
The algorithm is “integrated into a risk-minimization strategy tailored for patients with highly active MS, emphasizing in this case the pivotal role of natalizumab in averting treatment delays and providing adequate protection against potentially severe infections,” Dr. Carvajal said.
Participants who initiated or continued treatment with natalizumab completed their vaccination regimen without any incidents of progressive multifocal leukoencephalopathy (PML) or disease activity rebound following natalizumab discontinuation.
This suggests that using natalizumab for a brief duration might be a “viable option to contemplate,” the authors noted.
Commenting on the findings, Grace Gombolay, MD, assistant professor of pediatrics in the Division of Pediatric Neurology and director of the Pediatric Neuroimmunology and Multiple Sclerosis Clinic, Emory University, Atlanta, Georgia, said the study “demonstrates that vaccines are safe and do not trigger attacks in patients with MS on natalizumab, and that immunity — as measured by antibodies — is preserved in MS patients who receive natalizumab.”
This “contrasts with other treatments, as decreased antibody responses in COVID-19 are noted in certain treatments,” said Dr. Gombolay, who was not part of the study. “If both disease control and immunity against infection are the goals for the patient, then natalizumab is a reasonable option.”
“However, this must be balanced with other considerations,” she added, including the risk for PML and pregnancy.
This study was supported by grants from the European Committee for Treatment and Research in Multiple Sclerosis, Instituto de Salud Carlos III, and the European Union. Dr. Carvajal reported receiving grants from Vall d’Hebron Institut de Recerca and the European Committee for Treatment and Research in Multiple Sclerosis and honoraria from Roche, Novartis, BIIB-Colombia, Merck, and Sanofi outside the submitted work. Dr. Gombolay serves as media editor for Pediatric Neurology and as associate editor of the Annals of the Child Neurology Society. She is also a part-time CDC consultant for acute flaccid myelitis and received an honorarium as a speaker at the Georgia Neurological Society meeting, sponsored by Academic CME and TG Therapeutics.
A version of this article appeared on Medscape.com.
FROM JAMA OPEN NETWORK
Oncologists Voice Ethical Concerns Over AI in Cancer Care
TOPLINE:
Most respondents, for instance, said patients should not be expected to understand how AI tools work, but many also felt patients could make treatment decisions based on AI-generated recommendations. Most oncologists also felt responsible for protecting patients from biased AI, but few were confident that they could do so.
METHODOLOGY:
- The US Food and Drug Administration (FDA) has for use in various medical specialties over the past few decades, and increasingly, AI tools are being integrated into cancer care.
- However, the uptake of these tools in oncology has raised ethical questions and concerns, including challenges with AI bias, error, or misuse, as well as issues explaining how an AI model reached a result.
- In the current study, researchers asked 204 oncologists from 37 states for their views on the ethical implications of using AI for cancer care.
- Among the survey respondents, 64% were men and 63% were non-Hispanic White; 29% were from academic practices, 47% had received some education on AI use in healthcare, and 45% were familiar with clinical decision models.
- The researchers assessed respondents’ answers to various questions, including whether to provide informed consent for AI use and how oncologists would approach a scenario where the AI model and the oncologist recommended a different treatment regimen.
TAKEAWAY:
- Overall, 81% of oncologists supported having patient consent to use an AI model during treatment decisions, and 85% felt that oncologists needed to be able to explain an AI-based clinical decision model to use it in the clinic; however, only 23% felt that patients also needed to be able to explain an AI model.
- When an AI decision model recommended a different treatment regimen than the treating oncologist, the most common response (36.8%) was to present both options to the patient and let the patient decide. Oncologists from academic settings were about 2.5 times more likely than those from other settings to let the patient decide. About 34% of respondents said they would present both options but recommend the oncologist’s regimen, whereas about 22% said they would present both but recommend the AI’s regimen. A small percentage would only present the oncologist’s regimen (5%) or the AI’s regimen (about 2.5%).
- About three of four respondents (76.5%) agreed that oncologists should protect patients from biased AI tools; however, only about one of four (27.9%) felt confident they could identify biased AI models.
- Most oncologists (91%) felt that AI developers were responsible for the medico-legal problems associated with AI use; less than half (47%) said oncologists or hospitals (43%) shared this responsibility.
IN PRACTICE:
“Together, these data characterize barriers that may impede the ethical adoption of AI into cancer care. The findings suggest that the implementation of AI in oncology must include rigorous assessments of its effect on care decisions, as well as decisional responsibility when problems related to AI use arise,” the authors concluded.
SOURCE:
The study, with first author Andrew Hantel, MD, from Dana-Farber Cancer Institute, Boston, was published last month in JAMA Network Open.
LIMITATIONS:
The study had a moderate sample size and response rate, although demographics of participating oncologists appear to be nationally representative. The cross-sectional study design limited the generalizability of the findings over time as AI is integrated into cancer care.
DISCLOSURES:
The study was funded by the National Cancer Institute, the Dana-Farber McGraw/Patterson Research Fund, and the Mark Foundation Emerging Leader Award. Dr. Hantel reported receiving personal fees from AbbVie, AstraZeneca, the American Journal of Managed Care, Genentech, and GSK.
A version of this article appeared on Medscape.com.
TOPLINE:
Most respondents, for instance, said patients should not be expected to understand how AI tools work, but many also felt patients could make treatment decisions based on AI-generated recommendations. Most oncologists also felt responsible for protecting patients from biased AI, but few were confident that they could do so.
METHODOLOGY:
- The US Food and Drug Administration (FDA) has for use in various medical specialties over the past few decades, and increasingly, AI tools are being integrated into cancer care.
- However, the uptake of these tools in oncology has raised ethical questions and concerns, including challenges with AI bias, error, or misuse, as well as issues explaining how an AI model reached a result.
- In the current study, researchers asked 204 oncologists from 37 states for their views on the ethical implications of using AI for cancer care.
- Among the survey respondents, 64% were men and 63% were non-Hispanic White; 29% were from academic practices, 47% had received some education on AI use in healthcare, and 45% were familiar with clinical decision models.
- The researchers assessed respondents’ answers to various questions, including whether to provide informed consent for AI use and how oncologists would approach a scenario where the AI model and the oncologist recommended a different treatment regimen.
TAKEAWAY:
- Overall, 81% of oncologists supported having patient consent to use an AI model during treatment decisions, and 85% felt that oncologists needed to be able to explain an AI-based clinical decision model to use it in the clinic; however, only 23% felt that patients also needed to be able to explain an AI model.
- When an AI decision model recommended a different treatment regimen than the treating oncologist, the most common response (36.8%) was to present both options to the patient and let the patient decide. Oncologists from academic settings were about 2.5 times more likely than those from other settings to let the patient decide. About 34% of respondents said they would present both options but recommend the oncologist’s regimen, whereas about 22% said they would present both but recommend the AI’s regimen. A small percentage would only present the oncologist’s regimen (5%) or the AI’s regimen (about 2.5%).
- About three of four respondents (76.5%) agreed that oncologists should protect patients from biased AI tools; however, only about one of four (27.9%) felt confident they could identify biased AI models.
- Most oncologists (91%) felt that AI developers were responsible for the medico-legal problems associated with AI use; less than half (47%) said oncologists or hospitals (43%) shared this responsibility.
IN PRACTICE:
“Together, these data characterize barriers that may impede the ethical adoption of AI into cancer care. The findings suggest that the implementation of AI in oncology must include rigorous assessments of its effect on care decisions, as well as decisional responsibility when problems related to AI use arise,” the authors concluded.
SOURCE:
The study, with first author Andrew Hantel, MD, from Dana-Farber Cancer Institute, Boston, was published last month in JAMA Network Open.
LIMITATIONS:
The study had a moderate sample size and response rate, although demographics of participating oncologists appear to be nationally representative. The cross-sectional study design limited the generalizability of the findings over time as AI is integrated into cancer care.
DISCLOSURES:
The study was funded by the National Cancer Institute, the Dana-Farber McGraw/Patterson Research Fund, and the Mark Foundation Emerging Leader Award. Dr. Hantel reported receiving personal fees from AbbVie, AstraZeneca, the American Journal of Managed Care, Genentech, and GSK.
A version of this article appeared on Medscape.com.
TOPLINE:
Most respondents, for instance, said patients should not be expected to understand how AI tools work, but many also felt patients could make treatment decisions based on AI-generated recommendations. Most oncologists also felt responsible for protecting patients from biased AI, but few were confident that they could do so.
METHODOLOGY:
- The US Food and Drug Administration (FDA) has for use in various medical specialties over the past few decades, and increasingly, AI tools are being integrated into cancer care.
- However, the uptake of these tools in oncology has raised ethical questions and concerns, including challenges with AI bias, error, or misuse, as well as issues explaining how an AI model reached a result.
- In the current study, researchers asked 204 oncologists from 37 states for their views on the ethical implications of using AI for cancer care.
- Among the survey respondents, 64% were men and 63% were non-Hispanic White; 29% were from academic practices, 47% had received some education on AI use in healthcare, and 45% were familiar with clinical decision models.
- The researchers assessed respondents’ answers to various questions, including whether to provide informed consent for AI use and how oncologists would approach a scenario where the AI model and the oncologist recommended a different treatment regimen.
TAKEAWAY:
- Overall, 81% of oncologists supported having patient consent to use an AI model during treatment decisions, and 85% felt that oncologists needed to be able to explain an AI-based clinical decision model to use it in the clinic; however, only 23% felt that patients also needed to be able to explain an AI model.
- When an AI decision model recommended a different treatment regimen than the treating oncologist, the most common response (36.8%) was to present both options to the patient and let the patient decide. Oncologists from academic settings were about 2.5 times more likely than those from other settings to let the patient decide. About 34% of respondents said they would present both options but recommend the oncologist’s regimen, whereas about 22% said they would present both but recommend the AI’s regimen. A small percentage would only present the oncologist’s regimen (5%) or the AI’s regimen (about 2.5%).
- About three of four respondents (76.5%) agreed that oncologists should protect patients from biased AI tools; however, only about one of four (27.9%) felt confident they could identify biased AI models.
- Most oncologists (91%) felt that AI developers were responsible for the medico-legal problems associated with AI use; less than half (47%) said oncologists or hospitals (43%) shared this responsibility.
IN PRACTICE:
“Together, these data characterize barriers that may impede the ethical adoption of AI into cancer care. The findings suggest that the implementation of AI in oncology must include rigorous assessments of its effect on care decisions, as well as decisional responsibility when problems related to AI use arise,” the authors concluded.
SOURCE:
The study, with first author Andrew Hantel, MD, from Dana-Farber Cancer Institute, Boston, was published last month in JAMA Network Open.
LIMITATIONS:
The study had a moderate sample size and response rate, although demographics of participating oncologists appear to be nationally representative. The cross-sectional study design limited the generalizability of the findings over time as AI is integrated into cancer care.
DISCLOSURES:
The study was funded by the National Cancer Institute, the Dana-Farber McGraw/Patterson Research Fund, and the Mark Foundation Emerging Leader Award. Dr. Hantel reported receiving personal fees from AbbVie, AstraZeneca, the American Journal of Managed Care, Genentech, and GSK.
A version of this article appeared on Medscape.com.
Delirium Linked to a Threefold Increased Risk for Dementia
, new research showed.
Incident dementia risk was more than three times higher in those who experienced just one episode of delirium, with each additional episode linked to a further 20% increase in dementia risk. The association was strongest in men.
Patients with delirium also had a 39% higher mortality risk than those with no history of delirium.
“We have known for a long time that delirium is dangerous, and this provides evidence that it’s even more dangerous than perhaps we had appreciated,” said study investigator Emily H. Gordon, PhD, MBBS, a geriatrician and senior lecturer at the University of Queensland, Brisbane, Australia.
“But we also know delirium is preventable. There are no excuses anymore; we really need to work together to improve the hospital system, to implement what are known to be effective interventions,” she added.
The findings were published online in The BMJ.
Close Matching
Prior studies that suggested an association between delirium and dementia were relatively small with short follow-up and varied in their adjustment for confounders. They also didn’t account for the competing risk for death, researchers noted.
Investigators used a linked New South Wales (NSW) statewide dataset that includes records of care episodes from all NSW hospitals as well as personal, administrative, clinical, and death information.
The study included an eligible sample of 626,467 older adults without dementia at baseline with at least one hospital admission between 2009 and 2014. For these patients, researchers calculated a hospital frailty risk score and collected other information including primary diagnosis and mean length of hospital stay and stay in the intensive care unit. From diagnostic codes, they categorized patients into no delirium and delirium groups and determined the number of delirium episodes.
Investigators matched patients in the delirium group to patients with no delirium according to characteristics with potential to confound the association between delirium and risk for dementia, including age, gender, frailty, reason for hospitalization, and length of stay in hospital and intensive care.
The matched study sample included 55,211 (mean age, 83 years) each in the delirium and the no delirium groups. Despite matching, the length of hospital stay for the index episode was longer for the delirium group than the no delirium group (mean, 9 days vs 6 days).
The primary outcomes were death and incident dementia, determined via diagnostic codes. During a follow-up of 5.25 years, 58% of patients died, and 17% had a new dementia diagnosis.
Among patients with at least one episode of delirium, the rate of incident dementia was 3.4 times higher than in those without delirium. After accounting for the competing risk for death, incident dementia risk remained three times higher among the delirium group (hazard ratio [HR], 3.00; 95% CI, 2.91-3.10).
This association was stronger for men than women (HR, 3.17 and 2.88, respectively; P = .004).
Sex Differences
The study is thought to be the first to identify a difference between sexes in dementia risk and delirium, Dr. Gordon said. It’s possible delirium in men is more severe in intensity or lasts longer than in women, or the male brain is, for whatever reason, more vulnerable to the effects of delirium, said Dr. Gordon. But she stressed these are only theories.
Investigators also found a mortality rate 1.4 times higher in the delirium group versus those without delirium, equating to a 39% increased risk for death (HR, 1.39; 95% CI, 1.37-1.41). The risk was similar for men and women (interaction P = .62).
When researchers categorized delirium by number of episodes, they found each additional episode was associated with a 10% increased risk for death (HR, 1.10; 95% CI, 1.09-1.12).
In addition to its large size, long follow-up, and close matching, what sets this new study apart from previous research is it accounted for the competing risk for death, said Dr. Gordon.
“This is really important because you’re not going to get dementia if you die, and in this population, the rate of death is incredibly high,” she said. “If we just assume people who died didn’t get dementia, then that screws the results.”
Causal Link?
For those who experienced at least one episode of delirium within the first 12 months, each additional episode of delirium was associated with a 20% increased risk for dementia (HR, 1.20; 95% CI, 1.18-1.23).
That dose-response association suggests a causal link between the two, Dr. Gordon said.
“The number one way to prove causality is to do a randomized controlled trial,” which isn’t feasible with delirium, she said. “By demonstrating a dose-response relationship suggests that it could be a causal pathway.”
Exact mechanisms linking delirium with dementia are unclear. Delirium might uncover preexisting or preclinical dementia, or it might cause dementia by accelerating underlying neuropathologic processes or de novo mechanisms, the authors noted.
Study limitations included the potential for residual confounding from unmeasured variables in the matching criteria. Delirium and dementia diagnoses depended on clinical coding of medical information recorded in the administrative dataset, and under-coding of dementia during hospitalization is well-recognized.
Although the study controlled for length of stay in hospital and in intensive care, this may not have fully captured differences in severity of medical conditions. Data about the duration and severity of delirium episodes were also unavailable, which limited the dose-response analysis.
Commenting on the findings, Christopher Weber, PhD, Alzheimer’s Association as director of Global Science Initiatives, said the results are consistent with other research on the association between delirium and incidents of dementia.
The increased risk for dementia following delirium in males is “an interesting finding,” said Dr. Weber. “This suggests a need for more research to understand the impact of sex differences in delirium, as well as research to see if preventing incidents of delirium could ultimately reduce rates of dementia.”
The study received support from the National Health and Medical Research Council: Partnership Centre for Health System Sustainability. Dr. Gordon and Dr. Weber reported no relevant conflicts of interest.
A version of this article appeared on Medscape.com.
, new research showed.
Incident dementia risk was more than three times higher in those who experienced just one episode of delirium, with each additional episode linked to a further 20% increase in dementia risk. The association was strongest in men.
Patients with delirium also had a 39% higher mortality risk than those with no history of delirium.
“We have known for a long time that delirium is dangerous, and this provides evidence that it’s even more dangerous than perhaps we had appreciated,” said study investigator Emily H. Gordon, PhD, MBBS, a geriatrician and senior lecturer at the University of Queensland, Brisbane, Australia.
“But we also know delirium is preventable. There are no excuses anymore; we really need to work together to improve the hospital system, to implement what are known to be effective interventions,” she added.
The findings were published online in The BMJ.
Close Matching
Prior studies that suggested an association between delirium and dementia were relatively small with short follow-up and varied in their adjustment for confounders. They also didn’t account for the competing risk for death, researchers noted.
Investigators used a linked New South Wales (NSW) statewide dataset that includes records of care episodes from all NSW hospitals as well as personal, administrative, clinical, and death information.
The study included an eligible sample of 626,467 older adults without dementia at baseline with at least one hospital admission between 2009 and 2014. For these patients, researchers calculated a hospital frailty risk score and collected other information including primary diagnosis and mean length of hospital stay and stay in the intensive care unit. From diagnostic codes, they categorized patients into no delirium and delirium groups and determined the number of delirium episodes.
Investigators matched patients in the delirium group to patients with no delirium according to characteristics with potential to confound the association between delirium and risk for dementia, including age, gender, frailty, reason for hospitalization, and length of stay in hospital and intensive care.
The matched study sample included 55,211 (mean age, 83 years) each in the delirium and the no delirium groups. Despite matching, the length of hospital stay for the index episode was longer for the delirium group than the no delirium group (mean, 9 days vs 6 days).
The primary outcomes were death and incident dementia, determined via diagnostic codes. During a follow-up of 5.25 years, 58% of patients died, and 17% had a new dementia diagnosis.
Among patients with at least one episode of delirium, the rate of incident dementia was 3.4 times higher than in those without delirium. After accounting for the competing risk for death, incident dementia risk remained three times higher among the delirium group (hazard ratio [HR], 3.00; 95% CI, 2.91-3.10).
This association was stronger for men than women (HR, 3.17 and 2.88, respectively; P = .004).
Sex Differences
The study is thought to be the first to identify a difference between sexes in dementia risk and delirium, Dr. Gordon said. It’s possible delirium in men is more severe in intensity or lasts longer than in women, or the male brain is, for whatever reason, more vulnerable to the effects of delirium, said Dr. Gordon. But she stressed these are only theories.
Investigators also found a mortality rate 1.4 times higher in the delirium group versus those without delirium, equating to a 39% increased risk for death (HR, 1.39; 95% CI, 1.37-1.41). The risk was similar for men and women (interaction P = .62).
When researchers categorized delirium by number of episodes, they found each additional episode was associated with a 10% increased risk for death (HR, 1.10; 95% CI, 1.09-1.12).
In addition to its large size, long follow-up, and close matching, what sets this new study apart from previous research is it accounted for the competing risk for death, said Dr. Gordon.
“This is really important because you’re not going to get dementia if you die, and in this population, the rate of death is incredibly high,” she said. “If we just assume people who died didn’t get dementia, then that screws the results.”
Causal Link?
For those who experienced at least one episode of delirium within the first 12 months, each additional episode of delirium was associated with a 20% increased risk for dementia (HR, 1.20; 95% CI, 1.18-1.23).
That dose-response association suggests a causal link between the two, Dr. Gordon said.
“The number one way to prove causality is to do a randomized controlled trial,” which isn’t feasible with delirium, she said. “By demonstrating a dose-response relationship suggests that it could be a causal pathway.”
Exact mechanisms linking delirium with dementia are unclear. Delirium might uncover preexisting or preclinical dementia, or it might cause dementia by accelerating underlying neuropathologic processes or de novo mechanisms, the authors noted.
Study limitations included the potential for residual confounding from unmeasured variables in the matching criteria. Delirium and dementia diagnoses depended on clinical coding of medical information recorded in the administrative dataset, and under-coding of dementia during hospitalization is well-recognized.
Although the study controlled for length of stay in hospital and in intensive care, this may not have fully captured differences in severity of medical conditions. Data about the duration and severity of delirium episodes were also unavailable, which limited the dose-response analysis.
Commenting on the findings, Christopher Weber, PhD, Alzheimer’s Association as director of Global Science Initiatives, said the results are consistent with other research on the association between delirium and incidents of dementia.
The increased risk for dementia following delirium in males is “an interesting finding,” said Dr. Weber. “This suggests a need for more research to understand the impact of sex differences in delirium, as well as research to see if preventing incidents of delirium could ultimately reduce rates of dementia.”
The study received support from the National Health and Medical Research Council: Partnership Centre for Health System Sustainability. Dr. Gordon and Dr. Weber reported no relevant conflicts of interest.
A version of this article appeared on Medscape.com.
, new research showed.
Incident dementia risk was more than three times higher in those who experienced just one episode of delirium, with each additional episode linked to a further 20% increase in dementia risk. The association was strongest in men.
Patients with delirium also had a 39% higher mortality risk than those with no history of delirium.
“We have known for a long time that delirium is dangerous, and this provides evidence that it’s even more dangerous than perhaps we had appreciated,” said study investigator Emily H. Gordon, PhD, MBBS, a geriatrician and senior lecturer at the University of Queensland, Brisbane, Australia.
“But we also know delirium is preventable. There are no excuses anymore; we really need to work together to improve the hospital system, to implement what are known to be effective interventions,” she added.
The findings were published online in The BMJ.
Close Matching
Prior studies that suggested an association between delirium and dementia were relatively small with short follow-up and varied in their adjustment for confounders. They also didn’t account for the competing risk for death, researchers noted.
Investigators used a linked New South Wales (NSW) statewide dataset that includes records of care episodes from all NSW hospitals as well as personal, administrative, clinical, and death information.
The study included an eligible sample of 626,467 older adults without dementia at baseline with at least one hospital admission between 2009 and 2014. For these patients, researchers calculated a hospital frailty risk score and collected other information including primary diagnosis and mean length of hospital stay and stay in the intensive care unit. From diagnostic codes, they categorized patients into no delirium and delirium groups and determined the number of delirium episodes.
Investigators matched patients in the delirium group to patients with no delirium according to characteristics with potential to confound the association between delirium and risk for dementia, including age, gender, frailty, reason for hospitalization, and length of stay in hospital and intensive care.
The matched study sample included 55,211 (mean age, 83 years) each in the delirium and the no delirium groups. Despite matching, the length of hospital stay for the index episode was longer for the delirium group than the no delirium group (mean, 9 days vs 6 days).
The primary outcomes were death and incident dementia, determined via diagnostic codes. During a follow-up of 5.25 years, 58% of patients died, and 17% had a new dementia diagnosis.
Among patients with at least one episode of delirium, the rate of incident dementia was 3.4 times higher than in those without delirium. After accounting for the competing risk for death, incident dementia risk remained three times higher among the delirium group (hazard ratio [HR], 3.00; 95% CI, 2.91-3.10).
This association was stronger for men than women (HR, 3.17 and 2.88, respectively; P = .004).
Sex Differences
The study is thought to be the first to identify a difference between sexes in dementia risk and delirium, Dr. Gordon said. It’s possible delirium in men is more severe in intensity or lasts longer than in women, or the male brain is, for whatever reason, more vulnerable to the effects of delirium, said Dr. Gordon. But she stressed these are only theories.
Investigators also found a mortality rate 1.4 times higher in the delirium group versus those without delirium, equating to a 39% increased risk for death (HR, 1.39; 95% CI, 1.37-1.41). The risk was similar for men and women (interaction P = .62).
When researchers categorized delirium by number of episodes, they found each additional episode was associated with a 10% increased risk for death (HR, 1.10; 95% CI, 1.09-1.12).
In addition to its large size, long follow-up, and close matching, what sets this new study apart from previous research is it accounted for the competing risk for death, said Dr. Gordon.
“This is really important because you’re not going to get dementia if you die, and in this population, the rate of death is incredibly high,” she said. “If we just assume people who died didn’t get dementia, then that screws the results.”
Causal Link?
For those who experienced at least one episode of delirium within the first 12 months, each additional episode of delirium was associated with a 20% increased risk for dementia (HR, 1.20; 95% CI, 1.18-1.23).
That dose-response association suggests a causal link between the two, Dr. Gordon said.
“The number one way to prove causality is to do a randomized controlled trial,” which isn’t feasible with delirium, she said. “By demonstrating a dose-response relationship suggests that it could be a causal pathway.”
Exact mechanisms linking delirium with dementia are unclear. Delirium might uncover preexisting or preclinical dementia, or it might cause dementia by accelerating underlying neuropathologic processes or de novo mechanisms, the authors noted.
Study limitations included the potential for residual confounding from unmeasured variables in the matching criteria. Delirium and dementia diagnoses depended on clinical coding of medical information recorded in the administrative dataset, and under-coding of dementia during hospitalization is well-recognized.
Although the study controlled for length of stay in hospital and in intensive care, this may not have fully captured differences in severity of medical conditions. Data about the duration and severity of delirium episodes were also unavailable, which limited the dose-response analysis.
Commenting on the findings, Christopher Weber, PhD, Alzheimer’s Association as director of Global Science Initiatives, said the results are consistent with other research on the association between delirium and incidents of dementia.
The increased risk for dementia following delirium in males is “an interesting finding,” said Dr. Weber. “This suggests a need for more research to understand the impact of sex differences in delirium, as well as research to see if preventing incidents of delirium could ultimately reduce rates of dementia.”
The study received support from the National Health and Medical Research Council: Partnership Centre for Health System Sustainability. Dr. Gordon and Dr. Weber reported no relevant conflicts of interest.
A version of this article appeared on Medscape.com.
FROM THE BMJ
Chronic Pain Linked to Accelerated Brain Aging
, new research showed.
Using structural MRI data from more than 9000 adults with knee osteoarthritis (KOA) from the UK Biobank, investigators developed a brain age model to compare an individual’s brain age with their chronological age. Those with KOA showed a much faster rate of brain aging than healthy individuals.
The acceleration in brain aging was largely driven by the hippocampus and predicted memory decline and incident dementia during follow-up. Researchers identified a gene highly expressed in glial cells as a possible genetic factor for accelerated brain aging.
“We demonstrate the accelerated brain aging and cognitive decline in chronic musculoskeletal pain, in particular knee osteoarthritis, and provide a neural marker for early detection and intervention,” said co-first author Jiao Liu, PhD candidate, Chinese Academy of Sciences, Beijing.
“We are interested to know how to slow down the aging brain in chronic musculoskeletal pain patients. Proper exercise and lifestyle may reduce the risk,” Dr. Liu said.
The study was published online in Nature Mental Health.
Common Condition
CMP affects more than 40% of the world’s population and has been shown to have a harmful impact on cognitive function, although the exact mechanisms remain unclear. Prior research suggests that inflammatory markers associated with brain aging are higher in patients with CMP, suggesting a link between brain aging and CMP.
To investigate further, researchers explored patterns of brain aging in healthy cohorts and cohorts with four common types of CMP — chronic knee pain, chronic back pain, chronic neck pain, and chronic hip pain.
Using their brain age model, investigators observed significantly increased brain aging, or “predicted age difference,” only in individuals with KOA (P < .001). The observation was validated in an independent dataset (P = .020), suggesting a pattern of brain aging acceleration specific to KOA.
This acceleration was primarily driven by key brain regions involved in cognitive processing, including hippocampus and orbitofrontal cortex, and was correlated with longitudinal memory decline and dementia risk.
These data also suggest that the SLC39A8 gene, which is highly expressed in glial cells, might be a key genetic factor underpinning this acceleration.
“We not only revealed the specificity of accelerated brain aging in knee osteoarthritis patients, but importantly, we also provided longitudinal evidence suggesting the ability of our brain aging marker to predict future memory decline and increased dementia risk,” corresponding author Yiheng Tu, PhD, also with Chinese Academy of Sciences, Beijing, said in a news release.
A Future Treatment Target?
Commenting on this research, Shaheen Lakhan, MD, PhD, a neurologist and researcher based in Miami, noted that in this study, people with KOA showed signs of “faster brain aging on scans. Think of it as your brain wearing a disguise, appearing older than its actual years,” Dr. Lakhan said.
“Inflammation, a key player in osteoarthritis, might be playing a double agent, wreaking havoc not just on your joints but potentially on your memory too. Researchers even identified a specific gene linked to both knee pain and faster brain aging, hinting at a potential target for future treatments,” he added.
“Importantly, the increased risk of cognitive decline and dementia associated with chronic pain is likely one of many factors, and probably not a very high one on its own,” Dr. Lakhan noted.
The “good news,” he said, is that there are many “well-established ways to keep your brain sharp. Regular exercise, a healthy diet, and staying mentally stimulated are all proven strategies to reduce dementia risk. Think of chronic pain management as another tool you can add to your brain health toolbox.”
Support for the study was provided by the STI-2030 Major Project, the National Natural Science Foundation of China, the Scientific Foundation of the Institute of Psychology, Chinese Academy of Sciences, and the Young Elite Scientist Sponsorship Program by the China Association for Science and Technology. Dr. Liu and Dr. Lakhan had no relevant disclosures.
A version of this article appeared on Medscape.com.
, new research showed.
Using structural MRI data from more than 9000 adults with knee osteoarthritis (KOA) from the UK Biobank, investigators developed a brain age model to compare an individual’s brain age with their chronological age. Those with KOA showed a much faster rate of brain aging than healthy individuals.
The acceleration in brain aging was largely driven by the hippocampus and predicted memory decline and incident dementia during follow-up. Researchers identified a gene highly expressed in glial cells as a possible genetic factor for accelerated brain aging.
“We demonstrate the accelerated brain aging and cognitive decline in chronic musculoskeletal pain, in particular knee osteoarthritis, and provide a neural marker for early detection and intervention,” said co-first author Jiao Liu, PhD candidate, Chinese Academy of Sciences, Beijing.
“We are interested to know how to slow down the aging brain in chronic musculoskeletal pain patients. Proper exercise and lifestyle may reduce the risk,” Dr. Liu said.
The study was published online in Nature Mental Health.
Common Condition
CMP affects more than 40% of the world’s population and has been shown to have a harmful impact on cognitive function, although the exact mechanisms remain unclear. Prior research suggests that inflammatory markers associated with brain aging are higher in patients with CMP, suggesting a link between brain aging and CMP.
To investigate further, researchers explored patterns of brain aging in healthy cohorts and cohorts with four common types of CMP — chronic knee pain, chronic back pain, chronic neck pain, and chronic hip pain.
Using their brain age model, investigators observed significantly increased brain aging, or “predicted age difference,” only in individuals with KOA (P < .001). The observation was validated in an independent dataset (P = .020), suggesting a pattern of brain aging acceleration specific to KOA.
This acceleration was primarily driven by key brain regions involved in cognitive processing, including hippocampus and orbitofrontal cortex, and was correlated with longitudinal memory decline and dementia risk.
These data also suggest that the SLC39A8 gene, which is highly expressed in glial cells, might be a key genetic factor underpinning this acceleration.
“We not only revealed the specificity of accelerated brain aging in knee osteoarthritis patients, but importantly, we also provided longitudinal evidence suggesting the ability of our brain aging marker to predict future memory decline and increased dementia risk,” corresponding author Yiheng Tu, PhD, also with Chinese Academy of Sciences, Beijing, said in a news release.
A Future Treatment Target?
Commenting on this research, Shaheen Lakhan, MD, PhD, a neurologist and researcher based in Miami, noted that in this study, people with KOA showed signs of “faster brain aging on scans. Think of it as your brain wearing a disguise, appearing older than its actual years,” Dr. Lakhan said.
“Inflammation, a key player in osteoarthritis, might be playing a double agent, wreaking havoc not just on your joints but potentially on your memory too. Researchers even identified a specific gene linked to both knee pain and faster brain aging, hinting at a potential target for future treatments,” he added.
“Importantly, the increased risk of cognitive decline and dementia associated with chronic pain is likely one of many factors, and probably not a very high one on its own,” Dr. Lakhan noted.
The “good news,” he said, is that there are many “well-established ways to keep your brain sharp. Regular exercise, a healthy diet, and staying mentally stimulated are all proven strategies to reduce dementia risk. Think of chronic pain management as another tool you can add to your brain health toolbox.”
Support for the study was provided by the STI-2030 Major Project, the National Natural Science Foundation of China, the Scientific Foundation of the Institute of Psychology, Chinese Academy of Sciences, and the Young Elite Scientist Sponsorship Program by the China Association for Science and Technology. Dr. Liu and Dr. Lakhan had no relevant disclosures.
A version of this article appeared on Medscape.com.
, new research showed.
Using structural MRI data from more than 9000 adults with knee osteoarthritis (KOA) from the UK Biobank, investigators developed a brain age model to compare an individual’s brain age with their chronological age. Those with KOA showed a much faster rate of brain aging than healthy individuals.
The acceleration in brain aging was largely driven by the hippocampus and predicted memory decline and incident dementia during follow-up. Researchers identified a gene highly expressed in glial cells as a possible genetic factor for accelerated brain aging.
“We demonstrate the accelerated brain aging and cognitive decline in chronic musculoskeletal pain, in particular knee osteoarthritis, and provide a neural marker for early detection and intervention,” said co-first author Jiao Liu, PhD candidate, Chinese Academy of Sciences, Beijing.
“We are interested to know how to slow down the aging brain in chronic musculoskeletal pain patients. Proper exercise and lifestyle may reduce the risk,” Dr. Liu said.
The study was published online in Nature Mental Health.
Common Condition
CMP affects more than 40% of the world’s population and has been shown to have a harmful impact on cognitive function, although the exact mechanisms remain unclear. Prior research suggests that inflammatory markers associated with brain aging are higher in patients with CMP, suggesting a link between brain aging and CMP.
To investigate further, researchers explored patterns of brain aging in healthy cohorts and cohorts with four common types of CMP — chronic knee pain, chronic back pain, chronic neck pain, and chronic hip pain.
Using their brain age model, investigators observed significantly increased brain aging, or “predicted age difference,” only in individuals with KOA (P < .001). The observation was validated in an independent dataset (P = .020), suggesting a pattern of brain aging acceleration specific to KOA.
This acceleration was primarily driven by key brain regions involved in cognitive processing, including hippocampus and orbitofrontal cortex, and was correlated with longitudinal memory decline and dementia risk.
These data also suggest that the SLC39A8 gene, which is highly expressed in glial cells, might be a key genetic factor underpinning this acceleration.
“We not only revealed the specificity of accelerated brain aging in knee osteoarthritis patients, but importantly, we also provided longitudinal evidence suggesting the ability of our brain aging marker to predict future memory decline and increased dementia risk,” corresponding author Yiheng Tu, PhD, also with Chinese Academy of Sciences, Beijing, said in a news release.
A Future Treatment Target?
Commenting on this research, Shaheen Lakhan, MD, PhD, a neurologist and researcher based in Miami, noted that in this study, people with KOA showed signs of “faster brain aging on scans. Think of it as your brain wearing a disguise, appearing older than its actual years,” Dr. Lakhan said.
“Inflammation, a key player in osteoarthritis, might be playing a double agent, wreaking havoc not just on your joints but potentially on your memory too. Researchers even identified a specific gene linked to both knee pain and faster brain aging, hinting at a potential target for future treatments,” he added.
“Importantly, the increased risk of cognitive decline and dementia associated with chronic pain is likely one of many factors, and probably not a very high one on its own,” Dr. Lakhan noted.
The “good news,” he said, is that there are many “well-established ways to keep your brain sharp. Regular exercise, a healthy diet, and staying mentally stimulated are all proven strategies to reduce dementia risk. Think of chronic pain management as another tool you can add to your brain health toolbox.”
Support for the study was provided by the STI-2030 Major Project, the National Natural Science Foundation of China, the Scientific Foundation of the Institute of Psychology, Chinese Academy of Sciences, and the Young Elite Scientist Sponsorship Program by the China Association for Science and Technology. Dr. Liu and Dr. Lakhan had no relevant disclosures.
A version of this article appeared on Medscape.com.
FROM NATURE MENTAL HEALTH
Prevalence of Dementia in Homeless Twice That in Housed
, according to the results of a new study.
The findings suggested that dementia occurs earlier in homeless individuals, and that these patients could benefit from proactive screening and housing interventions.
“Whether dementia caused the homelessness or homelessness caused the dementia, it’s a bidirectional relationship,” said lead author Richard G. Booth, PhD, RN, adjunct scientist at ICES (formerly Institute for Clinical Evaluative Sciences) and associate professor of nursing at Western University in London, Ontario, Canada.
The study was published in the April issue of The Lancet Public Health.
Dementia at Early Ages
The investigators used health administrative data from Ontario to compare the prevalence of dementia among homeless people with that among housed individuals in the general population and those living in low-income neighborhoods.
They included individuals aged 45 years or older on January 1, 2019, who visited hospital-based ambulatory care (such as emergency departments), were hospitalized, or visited a community health center in 2019. The researchers identified people as experiencing homelessness if they had one or more healthcare records with an indication of homelessness or unstable housing. The prevalence of dementia was ascertained as of December 31, 2019.
Included in the population-based, cross-sectional comparative analysis were 12,863 homeless people, 475,544 people in the low-income group, and 2,273,068 people in the general population group.
Dementia prevalence was 68.7 per 1000 individuals among the homeless population, 62.6 per 1000 in the low-income group, and 51.0 per 1000 in the general population group.
After adjustments for age, sex, geographical location of residence (urban vs rural), and health conditions associated with dementia, the prevalence ratio of dementia among homeless people was 1.71, compared with the low-income group, and 1.90, compared with the general population group.
Dementia also was detected in the 45- to 55-year age group among homeless people. This age is much earlier than the age at which doctors start screening their patients for cognitive decline (65 years).
“The study was not designed to define the causality but consider: If you have early-stage dementia and you are not intact enough to do basic functions of life, the likelihood of you becoming homeless is definitely increased, and vice versa. If you are homeless and suffer significant environmental and physical traumas just living on the street, you age much quicker, and you will experience geriatric symptoms such as dementia earlier in your life trajectory,” said Dr. Booth.
“The main takeaway here is that if you don’t have housing, bad things are going to happen in life.”
Public Health Problem
In an accompanying editorial, William J. Panenka, MD, associate professor of psychiatry at the University of British Columbia in Vancouver, British Columbia, Canada, and colleagues cited modifiable risk factors for dementia, including lower education, traumatic brain injury, substance use, smoking, mood disorders, and social isolation, many of which are disproportionately prevalent among homeless individuals.
“Ultimately, dementia could contribute to the cycle of homelessness, whereby housing instability increases the risk for brain impairment, and brain impairment makes breaking the cycle of homelessness progressively more challenging,” they wrote.
Dr. Panenka and colleagues also pointed out that the average age of homeless people is increasing. In the United States, it is now approximately 50 years. This fact underscores “the immediacy and gravity of the public health problem. A multifaceted approach that integrates healthcare, housing, and social services is needed to better understand and alleviate the health consequences of homelessness. A concerted effort at all levels is vital to inform future public health efforts and stem the tide of increasing morbidity, compromised function, and early mortality in homelessness,” they concluded.
Stephen Hwang, MD, director of the MAP Centre for Urban Health Solutions at St. Michael’s Hospital and Unity Health in Toronto, said that the study may underestimate the magnitude of the problem of dementia among homeless people.
“The methods used in this research study are very strong because they draw upon data for everyone living in the entire province of Ontario, and this is a very powerful way of looking at this challenging problem. The study probably underestimates the magnitude of the problem because to be diagnosed with dementia, patients have to have contact with healthcare providers that make that diagnosis. Often, people experiencing homelessness don’t have extensive contact with the healthcare system, and so their condition may go undiagnosed,” said Dr. Hwang.
A specialist in internal medicine, Dr. Hwang has provided healthcare for homeless people, and his research focuses on homelessness, housing, and health. He said that the findings from the Canadian study are applicable to the United States.
Forced clearances of homeless people and placing them in encampments, something that has been discussed in Florida, is unlikely to solve the problem, he said.
“The approach that has been shown to be beneficial is to engage with people and offer them housing and services that will allow them to exit homelessness without criminalizing the fact that they’re homeless. There really is no reason to think that this approach of forced clearances is going to help anyone.”
This study was supported by ICES (formerly the Institute for Clinical Evaluative Sciences), which is funded by the Ontario Ministry of Health and Ontario Ministry of Long-Term Care. Dr. Booth and Dr. Hwang reported no relevant financial relationships. Dr. Panenka reported receiving a research grant from the Canadian Institutes of Health Research.
A version of this article appeared on Medscape.com.
, according to the results of a new study.
The findings suggested that dementia occurs earlier in homeless individuals, and that these patients could benefit from proactive screening and housing interventions.
“Whether dementia caused the homelessness or homelessness caused the dementia, it’s a bidirectional relationship,” said lead author Richard G. Booth, PhD, RN, adjunct scientist at ICES (formerly Institute for Clinical Evaluative Sciences) and associate professor of nursing at Western University in London, Ontario, Canada.
The study was published in the April issue of The Lancet Public Health.
Dementia at Early Ages
The investigators used health administrative data from Ontario to compare the prevalence of dementia among homeless people with that among housed individuals in the general population and those living in low-income neighborhoods.
They included individuals aged 45 years or older on January 1, 2019, who visited hospital-based ambulatory care (such as emergency departments), were hospitalized, or visited a community health center in 2019. The researchers identified people as experiencing homelessness if they had one or more healthcare records with an indication of homelessness or unstable housing. The prevalence of dementia was ascertained as of December 31, 2019.
Included in the population-based, cross-sectional comparative analysis were 12,863 homeless people, 475,544 people in the low-income group, and 2,273,068 people in the general population group.
Dementia prevalence was 68.7 per 1000 individuals among the homeless population, 62.6 per 1000 in the low-income group, and 51.0 per 1000 in the general population group.
After adjustments for age, sex, geographical location of residence (urban vs rural), and health conditions associated with dementia, the prevalence ratio of dementia among homeless people was 1.71, compared with the low-income group, and 1.90, compared with the general population group.
Dementia also was detected in the 45- to 55-year age group among homeless people. This age is much earlier than the age at which doctors start screening their patients for cognitive decline (65 years).
“The study was not designed to define the causality but consider: If you have early-stage dementia and you are not intact enough to do basic functions of life, the likelihood of you becoming homeless is definitely increased, and vice versa. If you are homeless and suffer significant environmental and physical traumas just living on the street, you age much quicker, and you will experience geriatric symptoms such as dementia earlier in your life trajectory,” said Dr. Booth.
“The main takeaway here is that if you don’t have housing, bad things are going to happen in life.”
Public Health Problem
In an accompanying editorial, William J. Panenka, MD, associate professor of psychiatry at the University of British Columbia in Vancouver, British Columbia, Canada, and colleagues cited modifiable risk factors for dementia, including lower education, traumatic brain injury, substance use, smoking, mood disorders, and social isolation, many of which are disproportionately prevalent among homeless individuals.
“Ultimately, dementia could contribute to the cycle of homelessness, whereby housing instability increases the risk for brain impairment, and brain impairment makes breaking the cycle of homelessness progressively more challenging,” they wrote.
Dr. Panenka and colleagues also pointed out that the average age of homeless people is increasing. In the United States, it is now approximately 50 years. This fact underscores “the immediacy and gravity of the public health problem. A multifaceted approach that integrates healthcare, housing, and social services is needed to better understand and alleviate the health consequences of homelessness. A concerted effort at all levels is vital to inform future public health efforts and stem the tide of increasing morbidity, compromised function, and early mortality in homelessness,” they concluded.
Stephen Hwang, MD, director of the MAP Centre for Urban Health Solutions at St. Michael’s Hospital and Unity Health in Toronto, said that the study may underestimate the magnitude of the problem of dementia among homeless people.
“The methods used in this research study are very strong because they draw upon data for everyone living in the entire province of Ontario, and this is a very powerful way of looking at this challenging problem. The study probably underestimates the magnitude of the problem because to be diagnosed with dementia, patients have to have contact with healthcare providers that make that diagnosis. Often, people experiencing homelessness don’t have extensive contact with the healthcare system, and so their condition may go undiagnosed,” said Dr. Hwang.
A specialist in internal medicine, Dr. Hwang has provided healthcare for homeless people, and his research focuses on homelessness, housing, and health. He said that the findings from the Canadian study are applicable to the United States.
Forced clearances of homeless people and placing them in encampments, something that has been discussed in Florida, is unlikely to solve the problem, he said.
“The approach that has been shown to be beneficial is to engage with people and offer them housing and services that will allow them to exit homelessness without criminalizing the fact that they’re homeless. There really is no reason to think that this approach of forced clearances is going to help anyone.”
This study was supported by ICES (formerly the Institute for Clinical Evaluative Sciences), which is funded by the Ontario Ministry of Health and Ontario Ministry of Long-Term Care. Dr. Booth and Dr. Hwang reported no relevant financial relationships. Dr. Panenka reported receiving a research grant from the Canadian Institutes of Health Research.
A version of this article appeared on Medscape.com.
, according to the results of a new study.
The findings suggested that dementia occurs earlier in homeless individuals, and that these patients could benefit from proactive screening and housing interventions.
“Whether dementia caused the homelessness or homelessness caused the dementia, it’s a bidirectional relationship,” said lead author Richard G. Booth, PhD, RN, adjunct scientist at ICES (formerly Institute for Clinical Evaluative Sciences) and associate professor of nursing at Western University in London, Ontario, Canada.
The study was published in the April issue of The Lancet Public Health.
Dementia at Early Ages
The investigators used health administrative data from Ontario to compare the prevalence of dementia among homeless people with that among housed individuals in the general population and those living in low-income neighborhoods.
They included individuals aged 45 years or older on January 1, 2019, who visited hospital-based ambulatory care (such as emergency departments), were hospitalized, or visited a community health center in 2019. The researchers identified people as experiencing homelessness if they had one or more healthcare records with an indication of homelessness or unstable housing. The prevalence of dementia was ascertained as of December 31, 2019.
Included in the population-based, cross-sectional comparative analysis were 12,863 homeless people, 475,544 people in the low-income group, and 2,273,068 people in the general population group.
Dementia prevalence was 68.7 per 1000 individuals among the homeless population, 62.6 per 1000 in the low-income group, and 51.0 per 1000 in the general population group.
After adjustments for age, sex, geographical location of residence (urban vs rural), and health conditions associated with dementia, the prevalence ratio of dementia among homeless people was 1.71, compared with the low-income group, and 1.90, compared with the general population group.
Dementia also was detected in the 45- to 55-year age group among homeless people. This age is much earlier than the age at which doctors start screening their patients for cognitive decline (65 years).
“The study was not designed to define the causality but consider: If you have early-stage dementia and you are not intact enough to do basic functions of life, the likelihood of you becoming homeless is definitely increased, and vice versa. If you are homeless and suffer significant environmental and physical traumas just living on the street, you age much quicker, and you will experience geriatric symptoms such as dementia earlier in your life trajectory,” said Dr. Booth.
“The main takeaway here is that if you don’t have housing, bad things are going to happen in life.”
Public Health Problem
In an accompanying editorial, William J. Panenka, MD, associate professor of psychiatry at the University of British Columbia in Vancouver, British Columbia, Canada, and colleagues cited modifiable risk factors for dementia, including lower education, traumatic brain injury, substance use, smoking, mood disorders, and social isolation, many of which are disproportionately prevalent among homeless individuals.
“Ultimately, dementia could contribute to the cycle of homelessness, whereby housing instability increases the risk for brain impairment, and brain impairment makes breaking the cycle of homelessness progressively more challenging,” they wrote.
Dr. Panenka and colleagues also pointed out that the average age of homeless people is increasing. In the United States, it is now approximately 50 years. This fact underscores “the immediacy and gravity of the public health problem. A multifaceted approach that integrates healthcare, housing, and social services is needed to better understand and alleviate the health consequences of homelessness. A concerted effort at all levels is vital to inform future public health efforts and stem the tide of increasing morbidity, compromised function, and early mortality in homelessness,” they concluded.
Stephen Hwang, MD, director of the MAP Centre for Urban Health Solutions at St. Michael’s Hospital and Unity Health in Toronto, said that the study may underestimate the magnitude of the problem of dementia among homeless people.
“The methods used in this research study are very strong because they draw upon data for everyone living in the entire province of Ontario, and this is a very powerful way of looking at this challenging problem. The study probably underestimates the magnitude of the problem because to be diagnosed with dementia, patients have to have contact with healthcare providers that make that diagnosis. Often, people experiencing homelessness don’t have extensive contact with the healthcare system, and so their condition may go undiagnosed,” said Dr. Hwang.
A specialist in internal medicine, Dr. Hwang has provided healthcare for homeless people, and his research focuses on homelessness, housing, and health. He said that the findings from the Canadian study are applicable to the United States.
Forced clearances of homeless people and placing them in encampments, something that has been discussed in Florida, is unlikely to solve the problem, he said.
“The approach that has been shown to be beneficial is to engage with people and offer them housing and services that will allow them to exit homelessness without criminalizing the fact that they’re homeless. There really is no reason to think that this approach of forced clearances is going to help anyone.”
This study was supported by ICES (formerly the Institute for Clinical Evaluative Sciences), which is funded by the Ontario Ministry of Health and Ontario Ministry of Long-Term Care. Dr. Booth and Dr. Hwang reported no relevant financial relationships. Dr. Panenka reported receiving a research grant from the Canadian Institutes of Health Research.
A version of this article appeared on Medscape.com.
From Lancet Public Health
Three Distinct MS Subtypes Identified
, a new study suggests.
With further validation, determining a patient’s blood “immune signature,” or endophenotype, before starting immunomodulatory therapy may help predict clinical disease trajectories and lead to more personalized treatment decisions, investigators said.
“The characterization of an endophenotype at timepoints of diagnosis will help to determine likely trajectory of the disease course but also will help to refine the chosen immune therapy,” said Heinz Wiendl, MD, professor and chair, Department of Neurology, University of Münster, Germany. “This is a rationale way of precision medicine for the future.”
The study was published online in Science Translational Medicine.
Degenerative and Inflammatory Subtypes
MS is a highly heterogeneous disorder with different clinical manifestations and disease trajectories, making it a challenge to manage. Whether this heterogeneity is reflected by discrete immune signatures in the blood has been unclear.
To investigate, Dr. Wiendl and a multicenter team comprehensively analyzed the immunological properties of blood samples collected from 309 patients with early MS and an independent validation cohort of 232 patients with early MS.
In both cohorts, they found that cellular immune signatures split into three distinct immunological endophenotypes, dubbed E1, E2, and E3.
E1 is characterized by alterations in the CD4 T-cell compartment, with increases in inflammatory cytokines, namely interleukin-17A (IL-17A), IL-22, and granulocyte-macrophage colony-stimulating factor, as well as earlier structural brain damage, more severe disease, and higher disability.
Alterations in natural killer cells are a hallmark of the E2 subtype, while alterations in the CD8 T cells dominate the E3 subtype.
The different subtypes were associated with distinct clinical disease trajectories. E3 patients displayed a pattern reflecting higher inflammatory disease activity, as illustrated by a higher relapse rate (≥ 2) within the first year from baseline and more frequent use of highly active disease-modifying therapies as first immunomodulatory treatment.
E3 patients also had higher numbers of gadolinium-enhancing lesions at baseline, a higher conversion rate from clinically isolated syndrome to relapsing-remitting MS, and rapid disability accrual within 2 years after baseline.
This endophenotype was also associated with an increase in total cell numbers within the cerebrospinal fluid and intrathecal immunoglobulin (Ig) G synthesis at baseline.
E1 patients had a higher degree of early structural brain damage and disease severity, including disability and impaired evident at baseline, and increased serum neurofilament light and increased intrathecal IgM synthesis at baseline.
“According to these different patterns of disease trajectories, we therefore termed these subsets degenerative E1 and inflammatory E3. Overall, although some of the clinical and paraclinical parameters partially overlapped, our analysis reveals that distinct immunological endophenotypes might have predictive value with regard to clinically relevant disease trajectories,” the researchers wrote.
Toward Personalized Care
In addition, during up to 4-year follow-up of some patients, they observed that patients with the inflammatory E3 endophenotype treated with interferon-beta exhibited higher disease progression and MRI activity relative to E3 patients receiving other therapies. These differential effects of interferon-beta were not observed in the other endophenotypes.
With further study and refinement, the hope is to make this test a “clinical reality,” Dr. Wiendl said.
Commenting on the findings, Kimberly O’Neill, MD, clinical instructor, Department of Neurology, NYU Grossman School of Medicine, New York City, noted that people with MS can have “a broad variety of disease course and outcomes ranging from mild to a very severe and life-altering disease course. At this point, we are not great at predicting who is going to be on which path and also which medication is right for each patient.
“Research like this gives us hope for a more personalized precision medicine in MS,” said Dr. O’Neill, who was not part of the study. “The ideal world would be to have a blood test that could tell their disease course and which treatments will work for an individual patient, but we are certainly not there yet.”
Also providing an outside perspective, Mary Rensel, MD, director of wellness and pediatric MS at the Cleveland Clinic Mellen Center for MS, Cleveland, said, “Precision medicine is our goal and dream in MS care — to be able to do a blood test and know what medicine a patient may or may not respond to and save them years of ongoing symptoms or the risk of disability. This study is a great start.”
Support for this research was provided by grants from the Federal Ministry of Education and Research, the German Research Council, and the Hermann and Lilly Schilling Foundation. Disclosures for study authors are listed with the original article. Dr. O’Neill and Dr. Rensel had no relevant disclosures.
A version of this article appeared on Medscape.com.
, a new study suggests.
With further validation, determining a patient’s blood “immune signature,” or endophenotype, before starting immunomodulatory therapy may help predict clinical disease trajectories and lead to more personalized treatment decisions, investigators said.
“The characterization of an endophenotype at timepoints of diagnosis will help to determine likely trajectory of the disease course but also will help to refine the chosen immune therapy,” said Heinz Wiendl, MD, professor and chair, Department of Neurology, University of Münster, Germany. “This is a rationale way of precision medicine for the future.”
The study was published online in Science Translational Medicine.
Degenerative and Inflammatory Subtypes
MS is a highly heterogeneous disorder with different clinical manifestations and disease trajectories, making it a challenge to manage. Whether this heterogeneity is reflected by discrete immune signatures in the blood has been unclear.
To investigate, Dr. Wiendl and a multicenter team comprehensively analyzed the immunological properties of blood samples collected from 309 patients with early MS and an independent validation cohort of 232 patients with early MS.
In both cohorts, they found that cellular immune signatures split into three distinct immunological endophenotypes, dubbed E1, E2, and E3.
E1 is characterized by alterations in the CD4 T-cell compartment, with increases in inflammatory cytokines, namely interleukin-17A (IL-17A), IL-22, and granulocyte-macrophage colony-stimulating factor, as well as earlier structural brain damage, more severe disease, and higher disability.
Alterations in natural killer cells are a hallmark of the E2 subtype, while alterations in the CD8 T cells dominate the E3 subtype.
The different subtypes were associated with distinct clinical disease trajectories. E3 patients displayed a pattern reflecting higher inflammatory disease activity, as illustrated by a higher relapse rate (≥ 2) within the first year from baseline and more frequent use of highly active disease-modifying therapies as first immunomodulatory treatment.
E3 patients also had higher numbers of gadolinium-enhancing lesions at baseline, a higher conversion rate from clinically isolated syndrome to relapsing-remitting MS, and rapid disability accrual within 2 years after baseline.
This endophenotype was also associated with an increase in total cell numbers within the cerebrospinal fluid and intrathecal immunoglobulin (Ig) G synthesis at baseline.
E1 patients had a higher degree of early structural brain damage and disease severity, including disability and impaired evident at baseline, and increased serum neurofilament light and increased intrathecal IgM synthesis at baseline.
“According to these different patterns of disease trajectories, we therefore termed these subsets degenerative E1 and inflammatory E3. Overall, although some of the clinical and paraclinical parameters partially overlapped, our analysis reveals that distinct immunological endophenotypes might have predictive value with regard to clinically relevant disease trajectories,” the researchers wrote.
Toward Personalized Care
In addition, during up to 4-year follow-up of some patients, they observed that patients with the inflammatory E3 endophenotype treated with interferon-beta exhibited higher disease progression and MRI activity relative to E3 patients receiving other therapies. These differential effects of interferon-beta were not observed in the other endophenotypes.
With further study and refinement, the hope is to make this test a “clinical reality,” Dr. Wiendl said.
Commenting on the findings, Kimberly O’Neill, MD, clinical instructor, Department of Neurology, NYU Grossman School of Medicine, New York City, noted that people with MS can have “a broad variety of disease course and outcomes ranging from mild to a very severe and life-altering disease course. At this point, we are not great at predicting who is going to be on which path and also which medication is right for each patient.
“Research like this gives us hope for a more personalized precision medicine in MS,” said Dr. O’Neill, who was not part of the study. “The ideal world would be to have a blood test that could tell their disease course and which treatments will work for an individual patient, but we are certainly not there yet.”
Also providing an outside perspective, Mary Rensel, MD, director of wellness and pediatric MS at the Cleveland Clinic Mellen Center for MS, Cleveland, said, “Precision medicine is our goal and dream in MS care — to be able to do a blood test and know what medicine a patient may or may not respond to and save them years of ongoing symptoms or the risk of disability. This study is a great start.”
Support for this research was provided by grants from the Federal Ministry of Education and Research, the German Research Council, and the Hermann and Lilly Schilling Foundation. Disclosures for study authors are listed with the original article. Dr. O’Neill and Dr. Rensel had no relevant disclosures.
A version of this article appeared on Medscape.com.
, a new study suggests.
With further validation, determining a patient’s blood “immune signature,” or endophenotype, before starting immunomodulatory therapy may help predict clinical disease trajectories and lead to more personalized treatment decisions, investigators said.
“The characterization of an endophenotype at timepoints of diagnosis will help to determine likely trajectory of the disease course but also will help to refine the chosen immune therapy,” said Heinz Wiendl, MD, professor and chair, Department of Neurology, University of Münster, Germany. “This is a rationale way of precision medicine for the future.”
The study was published online in Science Translational Medicine.
Degenerative and Inflammatory Subtypes
MS is a highly heterogeneous disorder with different clinical manifestations and disease trajectories, making it a challenge to manage. Whether this heterogeneity is reflected by discrete immune signatures in the blood has been unclear.
To investigate, Dr. Wiendl and a multicenter team comprehensively analyzed the immunological properties of blood samples collected from 309 patients with early MS and an independent validation cohort of 232 patients with early MS.
In both cohorts, they found that cellular immune signatures split into three distinct immunological endophenotypes, dubbed E1, E2, and E3.
E1 is characterized by alterations in the CD4 T-cell compartment, with increases in inflammatory cytokines, namely interleukin-17A (IL-17A), IL-22, and granulocyte-macrophage colony-stimulating factor, as well as earlier structural brain damage, more severe disease, and higher disability.
Alterations in natural killer cells are a hallmark of the E2 subtype, while alterations in the CD8 T cells dominate the E3 subtype.
The different subtypes were associated with distinct clinical disease trajectories. E3 patients displayed a pattern reflecting higher inflammatory disease activity, as illustrated by a higher relapse rate (≥ 2) within the first year from baseline and more frequent use of highly active disease-modifying therapies as first immunomodulatory treatment.
E3 patients also had higher numbers of gadolinium-enhancing lesions at baseline, a higher conversion rate from clinically isolated syndrome to relapsing-remitting MS, and rapid disability accrual within 2 years after baseline.
This endophenotype was also associated with an increase in total cell numbers within the cerebrospinal fluid and intrathecal immunoglobulin (Ig) G synthesis at baseline.
E1 patients had a higher degree of early structural brain damage and disease severity, including disability and impaired evident at baseline, and increased serum neurofilament light and increased intrathecal IgM synthesis at baseline.
“According to these different patterns of disease trajectories, we therefore termed these subsets degenerative E1 and inflammatory E3. Overall, although some of the clinical and paraclinical parameters partially overlapped, our analysis reveals that distinct immunological endophenotypes might have predictive value with regard to clinically relevant disease trajectories,” the researchers wrote.
Toward Personalized Care
In addition, during up to 4-year follow-up of some patients, they observed that patients with the inflammatory E3 endophenotype treated with interferon-beta exhibited higher disease progression and MRI activity relative to E3 patients receiving other therapies. These differential effects of interferon-beta were not observed in the other endophenotypes.
With further study and refinement, the hope is to make this test a “clinical reality,” Dr. Wiendl said.
Commenting on the findings, Kimberly O’Neill, MD, clinical instructor, Department of Neurology, NYU Grossman School of Medicine, New York City, noted that people with MS can have “a broad variety of disease course and outcomes ranging from mild to a very severe and life-altering disease course. At this point, we are not great at predicting who is going to be on which path and also which medication is right for each patient.
“Research like this gives us hope for a more personalized precision medicine in MS,” said Dr. O’Neill, who was not part of the study. “The ideal world would be to have a blood test that could tell their disease course and which treatments will work for an individual patient, but we are certainly not there yet.”
Also providing an outside perspective, Mary Rensel, MD, director of wellness and pediatric MS at the Cleveland Clinic Mellen Center for MS, Cleveland, said, “Precision medicine is our goal and dream in MS care — to be able to do a blood test and know what medicine a patient may or may not respond to and save them years of ongoing symptoms or the risk of disability. This study is a great start.”
Support for this research was provided by grants from the Federal Ministry of Education and Research, the German Research Council, and the Hermann and Lilly Schilling Foundation. Disclosures for study authors are listed with the original article. Dr. O’Neill and Dr. Rensel had no relevant disclosures.
A version of this article appeared on Medscape.com.
FROM SCIENCE TRANSLATIONAL MEDICINE