User login
FDA approves first generics of pregabalin
The generics were approved to manage neuropathic pain associated with diabetic peripheral neuropathy, postherpetic neuralgia, and fibromyalgia, as well as neuropathic pain associated with spinal cord injury, and as an adjunctive therapy for the treatment of partial-onset seizures in patients aged 17 years and older. Approvals were granted to Alembic Pharmaceuticals, Alkem Laboratories, Amneal Pharmaceuticals, Dr. Reddy’s Laboratories, InvaGen Pharmaceuticals, MSN Laboratories, Rising Pharmaceuticals, Sciegen Pharmaceuticals, and Teva Pharmaceuticals.
The most common adverse events associated with pregabalin include dizziness, somnolence, dry mouth, swelling, blurred vision, weight gain, and abnormal thinking. Pregabalin must be dispensed with a patient Medication Guide containing a guide to the drug’s uses and risks. Angioedema, hypersensitivity reactions, increased seizure frequency, increased suicidal behavior, and peripheral edema are all possible.
“Today’s approval of the first generics for pregabalin, a widely used medication, is another example of the FDA’s long-standing commitment to advance patient access to lower-cost, high-quality generic medicines,” Janet Woodcock, MD, director of the FDA’s Center for Drug Evaluation and Research, said in a press release.
The generics were approved to manage neuropathic pain associated with diabetic peripheral neuropathy, postherpetic neuralgia, and fibromyalgia, as well as neuropathic pain associated with spinal cord injury, and as an adjunctive therapy for the treatment of partial-onset seizures in patients aged 17 years and older. Approvals were granted to Alembic Pharmaceuticals, Alkem Laboratories, Amneal Pharmaceuticals, Dr. Reddy’s Laboratories, InvaGen Pharmaceuticals, MSN Laboratories, Rising Pharmaceuticals, Sciegen Pharmaceuticals, and Teva Pharmaceuticals.
The most common adverse events associated with pregabalin include dizziness, somnolence, dry mouth, swelling, blurred vision, weight gain, and abnormal thinking. Pregabalin must be dispensed with a patient Medication Guide containing a guide to the drug’s uses and risks. Angioedema, hypersensitivity reactions, increased seizure frequency, increased suicidal behavior, and peripheral edema are all possible.
“Today’s approval of the first generics for pregabalin, a widely used medication, is another example of the FDA’s long-standing commitment to advance patient access to lower-cost, high-quality generic medicines,” Janet Woodcock, MD, director of the FDA’s Center for Drug Evaluation and Research, said in a press release.
The generics were approved to manage neuropathic pain associated with diabetic peripheral neuropathy, postherpetic neuralgia, and fibromyalgia, as well as neuropathic pain associated with spinal cord injury, and as an adjunctive therapy for the treatment of partial-onset seizures in patients aged 17 years and older. Approvals were granted to Alembic Pharmaceuticals, Alkem Laboratories, Amneal Pharmaceuticals, Dr. Reddy’s Laboratories, InvaGen Pharmaceuticals, MSN Laboratories, Rising Pharmaceuticals, Sciegen Pharmaceuticals, and Teva Pharmaceuticals.
The most common adverse events associated with pregabalin include dizziness, somnolence, dry mouth, swelling, blurred vision, weight gain, and abnormal thinking. Pregabalin must be dispensed with a patient Medication Guide containing a guide to the drug’s uses and risks. Angioedema, hypersensitivity reactions, increased seizure frequency, increased suicidal behavior, and peripheral edema are all possible.
“Today’s approval of the first generics for pregabalin, a widely used medication, is another example of the FDA’s long-standing commitment to advance patient access to lower-cost, high-quality generic medicines,” Janet Woodcock, MD, director of the FDA’s Center for Drug Evaluation and Research, said in a press release.
New measles outbreaks reported in Los Angeles and El Paso
according to the Centers for Disease Control and Prevention.
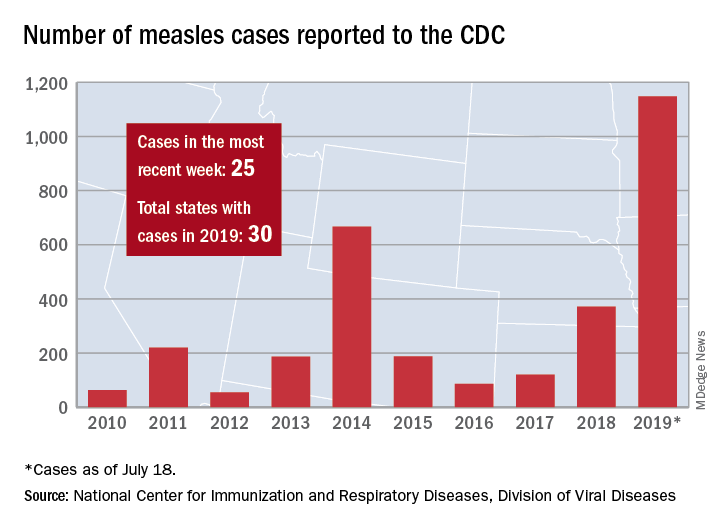
The total number of confirmed cases of measles in the United States is now up to 1,148 for the year, which is 25 more than the previous week, the CDC said on July 22. The highest 1-week total for the year was the 90 cases reported during the week of April 11.
The number of outbreaks is back up to five as California returned to the list after a 1-week absence and El Paso, Tex., made its first appearance of the year. The current outbreak in California – the state’s fifth – is occurring in Los Angeles, which is now up to 16 total cases in 2019. El Paso just reported its fourth case on July 17, and the city’s health department noted that “it had been more than 25 years since El Paso saw its last case of measles before these four recent cases.” Outbreaks also are ongoing in Rockland County, N.Y.; New York City; and three counties in Washington State.
States that joined the ranks of the measles-infected during this most recent reporting week were Alaska and Ohio, which brings the total number to 30 for the year, the CDC said.
The Alaska Department of Health and Social Services said that it “has confirmed a single case of measles in an unvaccinated teenager from the Kenai Peninsula who recently traveled out of state to Arizona via Seattle.” The Ohio case is a “young adult from Stark County [who] recently traveled to a state with confirmed measles cases,” according to the state’s health department.
according to the Centers for Disease Control and Prevention.

The total number of confirmed cases of measles in the United States is now up to 1,148 for the year, which is 25 more than the previous week, the CDC said on July 22. The highest 1-week total for the year was the 90 cases reported during the week of April 11.
The number of outbreaks is back up to five as California returned to the list after a 1-week absence and El Paso, Tex., made its first appearance of the year. The current outbreak in California – the state’s fifth – is occurring in Los Angeles, which is now up to 16 total cases in 2019. El Paso just reported its fourth case on July 17, and the city’s health department noted that “it had been more than 25 years since El Paso saw its last case of measles before these four recent cases.” Outbreaks also are ongoing in Rockland County, N.Y.; New York City; and three counties in Washington State.
States that joined the ranks of the measles-infected during this most recent reporting week were Alaska and Ohio, which brings the total number to 30 for the year, the CDC said.
The Alaska Department of Health and Social Services said that it “has confirmed a single case of measles in an unvaccinated teenager from the Kenai Peninsula who recently traveled out of state to Arizona via Seattle.” The Ohio case is a “young adult from Stark County [who] recently traveled to a state with confirmed measles cases,” according to the state’s health department.
according to the Centers for Disease Control and Prevention.

The total number of confirmed cases of measles in the United States is now up to 1,148 for the year, which is 25 more than the previous week, the CDC said on July 22. The highest 1-week total for the year was the 90 cases reported during the week of April 11.
The number of outbreaks is back up to five as California returned to the list after a 1-week absence and El Paso, Tex., made its first appearance of the year. The current outbreak in California – the state’s fifth – is occurring in Los Angeles, which is now up to 16 total cases in 2019. El Paso just reported its fourth case on July 17, and the city’s health department noted that “it had been more than 25 years since El Paso saw its last case of measles before these four recent cases.” Outbreaks also are ongoing in Rockland County, N.Y.; New York City; and three counties in Washington State.
States that joined the ranks of the measles-infected during this most recent reporting week were Alaska and Ohio, which brings the total number to 30 for the year, the CDC said.
The Alaska Department of Health and Social Services said that it “has confirmed a single case of measles in an unvaccinated teenager from the Kenai Peninsula who recently traveled out of state to Arizona via Seattle.” The Ohio case is a “young adult from Stark County [who] recently traveled to a state with confirmed measles cases,” according to the state’s health department.
Smoking-cessation attempts changed little over 7-year span
according to the Centers for Disease Control and Prevention.

The median percentage of adult smokers who tried to quit cigarettes over the past year went from 64.9% in 2011 to 65.4% in 2017, CDC investigators reported in the Morbidity and Mortality Weekly Report, but the rate has gone down since 2014, when it reached 66.9%.
“The limited progress in increasing quit attempts … together with the variation in quit attempt prevalence among states, underscores the importance of enhanced efforts to motivate and help smokers to quit,” wrote Kimp Walton, MS, of the CDC’s National Center for Chronic Disease Prevention and Health Promotion and associates.
State-specific trends in quit-attempt rates reflected the national situation. The prevalence of past-year cessation attempts went up significantly in four states (Kansas, Louisiana, Virginia, and West Virginia) from 2011 to 2017, went down significantly in two states (New York and Tennessee), and did not change significantly in the other 44 states and the District of Columbia, they wrote.
In 2017, cigarette smokers in Connecticut were the most likely to have tried to quit in the past year, with a rate of 71.6%. The only other places with rates greater than 70% were Delaware, D.C., New Jersey, and Texas. The lowest quit-attempt rate that year, 58.6%, belonged to Wisconsin, with Iowa and Missouri the only other states under 60%, the investigators reported based on data from annual Behavioral Risk Factor Surveillance System surveys.
“Because most smokers make multiple quit attempts before succeeding, as many as 30 on average, tobacco dependence is viewed as a chronic, relapsing condition that requires repeated intervention. Smokers should be encouraged to keep trying to quit until they succeed, and health care providers should be encouraged to keep supporting smokers until they quit,” investigators wrote.
SOURCE: Walton K et al. MMWR. 2019 Jul 19;68(28):621-6.
according to the Centers for Disease Control and Prevention.

The median percentage of adult smokers who tried to quit cigarettes over the past year went from 64.9% in 2011 to 65.4% in 2017, CDC investigators reported in the Morbidity and Mortality Weekly Report, but the rate has gone down since 2014, when it reached 66.9%.
“The limited progress in increasing quit attempts … together with the variation in quit attempt prevalence among states, underscores the importance of enhanced efforts to motivate and help smokers to quit,” wrote Kimp Walton, MS, of the CDC’s National Center for Chronic Disease Prevention and Health Promotion and associates.
State-specific trends in quit-attempt rates reflected the national situation. The prevalence of past-year cessation attempts went up significantly in four states (Kansas, Louisiana, Virginia, and West Virginia) from 2011 to 2017, went down significantly in two states (New York and Tennessee), and did not change significantly in the other 44 states and the District of Columbia, they wrote.
In 2017, cigarette smokers in Connecticut were the most likely to have tried to quit in the past year, with a rate of 71.6%. The only other places with rates greater than 70% were Delaware, D.C., New Jersey, and Texas. The lowest quit-attempt rate that year, 58.6%, belonged to Wisconsin, with Iowa and Missouri the only other states under 60%, the investigators reported based on data from annual Behavioral Risk Factor Surveillance System surveys.
“Because most smokers make multiple quit attempts before succeeding, as many as 30 on average, tobacco dependence is viewed as a chronic, relapsing condition that requires repeated intervention. Smokers should be encouraged to keep trying to quit until they succeed, and health care providers should be encouraged to keep supporting smokers until they quit,” investigators wrote.
SOURCE: Walton K et al. MMWR. 2019 Jul 19;68(28):621-6.
according to the Centers for Disease Control and Prevention.

The median percentage of adult smokers who tried to quit cigarettes over the past year went from 64.9% in 2011 to 65.4% in 2017, CDC investigators reported in the Morbidity and Mortality Weekly Report, but the rate has gone down since 2014, when it reached 66.9%.
“The limited progress in increasing quit attempts … together with the variation in quit attempt prevalence among states, underscores the importance of enhanced efforts to motivate and help smokers to quit,” wrote Kimp Walton, MS, of the CDC’s National Center for Chronic Disease Prevention and Health Promotion and associates.
State-specific trends in quit-attempt rates reflected the national situation. The prevalence of past-year cessation attempts went up significantly in four states (Kansas, Louisiana, Virginia, and West Virginia) from 2011 to 2017, went down significantly in two states (New York and Tennessee), and did not change significantly in the other 44 states and the District of Columbia, they wrote.
In 2017, cigarette smokers in Connecticut were the most likely to have tried to quit in the past year, with a rate of 71.6%. The only other places with rates greater than 70% were Delaware, D.C., New Jersey, and Texas. The lowest quit-attempt rate that year, 58.6%, belonged to Wisconsin, with Iowa and Missouri the only other states under 60%, the investigators reported based on data from annual Behavioral Risk Factor Surveillance System surveys.
“Because most smokers make multiple quit attempts before succeeding, as many as 30 on average, tobacco dependence is viewed as a chronic, relapsing condition that requires repeated intervention. Smokers should be encouraged to keep trying to quit until they succeed, and health care providers should be encouraged to keep supporting smokers until they quit,” investigators wrote.
SOURCE: Walton K et al. MMWR. 2019 Jul 19;68(28):621-6.
FROM MMWR
Drug overdose deaths declined in 2018
Reported drug overdose deaths in the United States declined by 4.2% from December 2017 to December 2018, the Centers for Disease Control and Prevention reported on July 17.
“The latest provisional data on overdose deaths show that America’s united efforts to curb opioid use disorder and addiction are working. Lives are being saved, and we’re beginning to win the fight against this crisis,” Health & Human Services Secretary Alex Azar said in a written statement. “Under President Trump’s leadership, and thanks to efforts on the ground by communities across America, the number of patients receiving medication-assisted treatment has risen, distribution of overdose-reversing drugs is up, and nationwide opioid prescriptions are down.”
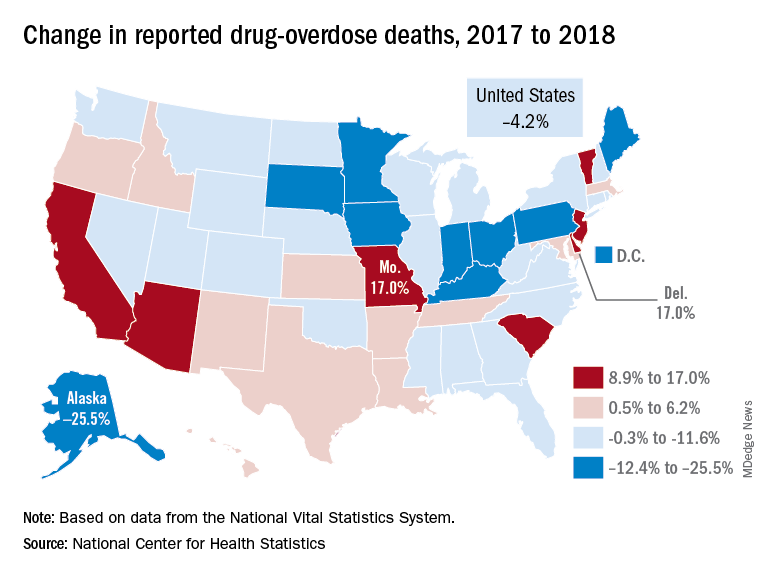
The new data show that total drug overdose deaths were down from 70,699 in 2017 to 67,744 in 2018, a drop of 4.2%, the CDC said.
States, of course, fell on both sides of that national figure. Delaware and Missouri wound up on the other end of the scale with increases of 17.0% from 2017 to 2018. Deaths in Vermont, Arizona, and South Carolina also rose by double digits, data from the National Vital Statistics System show.
“While the declining trend of overdose deaths is an encouraging sign, by no means have we declared victory against the epidemic or addiction in general,” Secretary Azar said. “This crisis developed over 2 decades and it will not be solved overnight. We also face other emerging threats, like concerning trends in cocaine and methamphetamine overdoses.”
Reported drug overdose deaths in the United States declined by 4.2% from December 2017 to December 2018, the Centers for Disease Control and Prevention reported on July 17.

“The latest provisional data on overdose deaths show that America’s united efforts to curb opioid use disorder and addiction are working. Lives are being saved, and we’re beginning to win the fight against this crisis,” Health & Human Services Secretary Alex Azar said in a written statement. “Under President Trump’s leadership, and thanks to efforts on the ground by communities across America, the number of patients receiving medication-assisted treatment has risen, distribution of overdose-reversing drugs is up, and nationwide opioid prescriptions are down.”
The new data show that total drug overdose deaths were down from 70,699 in 2017 to 67,744 in 2018, a drop of 4.2%, the CDC said.
States, of course, fell on both sides of that national figure. Delaware and Missouri wound up on the other end of the scale with increases of 17.0% from 2017 to 2018. Deaths in Vermont, Arizona, and South Carolina also rose by double digits, data from the National Vital Statistics System show.
“While the declining trend of overdose deaths is an encouraging sign, by no means have we declared victory against the epidemic or addiction in general,” Secretary Azar said. “This crisis developed over 2 decades and it will not be solved overnight. We also face other emerging threats, like concerning trends in cocaine and methamphetamine overdoses.”
Reported drug overdose deaths in the United States declined by 4.2% from December 2017 to December 2018, the Centers for Disease Control and Prevention reported on July 17.

“The latest provisional data on overdose deaths show that America’s united efforts to curb opioid use disorder and addiction are working. Lives are being saved, and we’re beginning to win the fight against this crisis,” Health & Human Services Secretary Alex Azar said in a written statement. “Under President Trump’s leadership, and thanks to efforts on the ground by communities across America, the number of patients receiving medication-assisted treatment has risen, distribution of overdose-reversing drugs is up, and nationwide opioid prescriptions are down.”
The new data show that total drug overdose deaths were down from 70,699 in 2017 to 67,744 in 2018, a drop of 4.2%, the CDC said.
States, of course, fell on both sides of that national figure. Delaware and Missouri wound up on the other end of the scale with increases of 17.0% from 2017 to 2018. Deaths in Vermont, Arizona, and South Carolina also rose by double digits, data from the National Vital Statistics System show.
“While the declining trend of overdose deaths is an encouraging sign, by no means have we declared victory against the epidemic or addiction in general,” Secretary Azar said. “This crisis developed over 2 decades and it will not be solved overnight. We also face other emerging threats, like concerning trends in cocaine and methamphetamine overdoses.”
Measles cases have slowed but not stopped
The United States continues to slowly add new cases of measles to 2019’s postelimination-record total, but California was officially removed from the outbreak list this week, according to the Centers for Disease Control and Prevention.
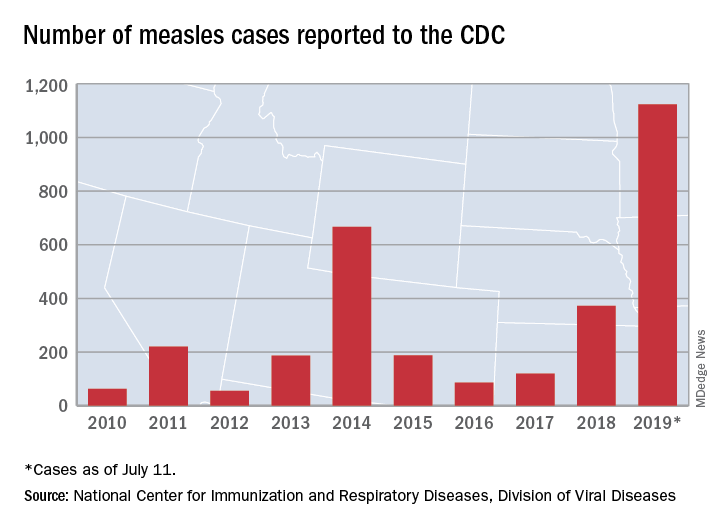
That is the highest number of cases reported since measles was declared eliminated in 2000 and the most in a single year since 1992.
The end of outbreak-related activity in California leaves three locations still dealing with ongoing cases: Rockland County, N.Y.; New York City; and King, Pierce, and Snohomish Counties in Washington, the CDC said.
Those three jurisdictions currently report the following:
- reported four new cases from July 3 to July 11 and is up to 175 cases for the year.
- had one new case from July 1 to July 8 and is now at 564 for the year.
- reported two cases from July 1 to July 10 and is now at 10 for the year (the other two counties have a total of three cases). Clark County in Washington reported 71 cases in an earlier, unrelated outbreak.
The United States continues to slowly add new cases of measles to 2019’s postelimination-record total, but California was officially removed from the outbreak list this week, according to the Centers for Disease Control and Prevention.

That is the highest number of cases reported since measles was declared eliminated in 2000 and the most in a single year since 1992.
The end of outbreak-related activity in California leaves three locations still dealing with ongoing cases: Rockland County, N.Y.; New York City; and King, Pierce, and Snohomish Counties in Washington, the CDC said.
Those three jurisdictions currently report the following:
- reported four new cases from July 3 to July 11 and is up to 175 cases for the year.
- had one new case from July 1 to July 8 and is now at 564 for the year.
- reported two cases from July 1 to July 10 and is now at 10 for the year (the other two counties have a total of three cases). Clark County in Washington reported 71 cases in an earlier, unrelated outbreak.
The United States continues to slowly add new cases of measles to 2019’s postelimination-record total, but California was officially removed from the outbreak list this week, according to the Centers for Disease Control and Prevention.

That is the highest number of cases reported since measles was declared eliminated in 2000 and the most in a single year since 1992.
The end of outbreak-related activity in California leaves three locations still dealing with ongoing cases: Rockland County, N.Y.; New York City; and King, Pierce, and Snohomish Counties in Washington, the CDC said.
Those three jurisdictions currently report the following:
- reported four new cases from July 3 to July 11 and is up to 175 cases for the year.
- had one new case from July 1 to July 8 and is now at 564 for the year.
- reported two cases from July 1 to July 10 and is now at 10 for the year (the other two counties have a total of three cases). Clark County in Washington reported 71 cases in an earlier, unrelated outbreak.
FDA pushes for more diversity in clinical trials in draft guidance
Officials at the Food and Drug Administration have issued draft guidance aimed at increasing the diversity of clinical trial populations, including adding children and adolescents earlier in drug development and making trial participation less burdensome for patients.
Despite efforts to enroll clinical trial participants who better reflect a real-world population, “challenges to participation in clinical trials remain, and certain groups continue to be unnecessarily underrepresented in many clinical trials,” the FDA noted in the document’s introduction.
The FDA noted that these challenges could have a significant impact on trial outcomes.
For example, the failure to include complex patients in a clinical trial “may lead to a failure to discover important safety information about the use of the investigational drug in patients who will take the drug after approval.”
In its draft recommendations, the FDA calls on trial sponsors to conduct a closer examination of exclusion criteria and to make it as narrow as possible; consider whether criteria from phase 2 studies, which are restrictive but often transferred to phase 3 protocols, can be eliminated or modified; and consider including children and adolescents when appropriate.
Recommendations related to trial design include characterizing early on the drug metabolism and clearance across populations that may metabolize or clear the drug differently, such as elderly or patients with liver or kidney dysfunction. The agency also called on trial sponsors to use “adaptive clinical trials,” which allow for prespecified trial design changes during the trial, and an early pediatric development program.
Another set of draft recommendations relates to ensuring trial participation is less burdensome for patients by reducing the frequency of study visits and making participants more aware of reimbursement for travel and lodging associated with the trial.
Additionally, the agency is recommending that trial sponsors adopt enrollment and retention practices that enhance inclusiveness, such as ensuring that trial sites include geographic locations with a higher concentration of racial and ethnic minority patients, and holding recruitment events on nights and weekends and in nonclinical locations.
Finally, the FDA issued a set of recommendations to trial sponsors aimed at broadening eligibility criteria when evaluating drugs intended for the treatment of rare diseases. In those trials, the FDA recommended reenrolling participants from early-phase trials in later-phase trials if it can be done safely.
“Because rare diseases often affect small, geographically dispersed patient populations with disease-related travel limitations, special efforts may be necessary to enroll and retain these participants to ensure that a broad spectrum of the patient population is represented,” the agency stated.
At first blush, the draft recommendations are being greeted with a positive response.
“We certainly need more diversity in clinical trial populations, so I think anything that FDA can say that will help encourage that is a good thing,” Richard Schilsky, MD, senior vice president and chief medical officer at the American Society of Clinical Oncology, said in an interview, noting that this new guidance builds off the work that ASCO and Friends of Cancer Research did with the FDA on previous efforts to expand clinical trial populations.
“I think that this new guidance, as best as I can tell, builds off of that, generalizes it across all therapeutic areas and goes a little bit beyond eligibility criteria to other features of clinical trials that may be impediments to patient participation and thereby limit the diversity of the populations in the studies,” he added.
That said, Dr. Schilsky said he welcomed the draft document as a step toward getting broader participation from patients who are more representative of the ultimate users of these treatments.
“All people who could potentially benefit from the trial should have the opportunity to participate as long as it’s safe for them to do so, and they should not be excluded based upon some of these arbitrary structural things,” he said.
“More importantly, we need data on how these new interventions perform in the patients who are seen and treated by doctors every day. We don’t want data just on patients who have no comorbid illnesses, patients who are otherwise perfectly well, patients who could run a marathon before they go to the doctor’s office. Those are generally not the patients that most doctors are seeing. If they are not represented in the trial population, then we are left with having to extrapolate from the trial data to a population for whom there is no information on how to use the treatment,” he added.
Public comments on the draft document are due on Aug. 6.
Officials at the Food and Drug Administration have issued draft guidance aimed at increasing the diversity of clinical trial populations, including adding children and adolescents earlier in drug development and making trial participation less burdensome for patients.
Despite efforts to enroll clinical trial participants who better reflect a real-world population, “challenges to participation in clinical trials remain, and certain groups continue to be unnecessarily underrepresented in many clinical trials,” the FDA noted in the document’s introduction.
The FDA noted that these challenges could have a significant impact on trial outcomes.
For example, the failure to include complex patients in a clinical trial “may lead to a failure to discover important safety information about the use of the investigational drug in patients who will take the drug after approval.”
In its draft recommendations, the FDA calls on trial sponsors to conduct a closer examination of exclusion criteria and to make it as narrow as possible; consider whether criteria from phase 2 studies, which are restrictive but often transferred to phase 3 protocols, can be eliminated or modified; and consider including children and adolescents when appropriate.
Recommendations related to trial design include characterizing early on the drug metabolism and clearance across populations that may metabolize or clear the drug differently, such as elderly or patients with liver or kidney dysfunction. The agency also called on trial sponsors to use “adaptive clinical trials,” which allow for prespecified trial design changes during the trial, and an early pediatric development program.
Another set of draft recommendations relates to ensuring trial participation is less burdensome for patients by reducing the frequency of study visits and making participants more aware of reimbursement for travel and lodging associated with the trial.
Additionally, the agency is recommending that trial sponsors adopt enrollment and retention practices that enhance inclusiveness, such as ensuring that trial sites include geographic locations with a higher concentration of racial and ethnic minority patients, and holding recruitment events on nights and weekends and in nonclinical locations.
Finally, the FDA issued a set of recommendations to trial sponsors aimed at broadening eligibility criteria when evaluating drugs intended for the treatment of rare diseases. In those trials, the FDA recommended reenrolling participants from early-phase trials in later-phase trials if it can be done safely.
“Because rare diseases often affect small, geographically dispersed patient populations with disease-related travel limitations, special efforts may be necessary to enroll and retain these participants to ensure that a broad spectrum of the patient population is represented,” the agency stated.
At first blush, the draft recommendations are being greeted with a positive response.
“We certainly need more diversity in clinical trial populations, so I think anything that FDA can say that will help encourage that is a good thing,” Richard Schilsky, MD, senior vice president and chief medical officer at the American Society of Clinical Oncology, said in an interview, noting that this new guidance builds off the work that ASCO and Friends of Cancer Research did with the FDA on previous efforts to expand clinical trial populations.
“I think that this new guidance, as best as I can tell, builds off of that, generalizes it across all therapeutic areas and goes a little bit beyond eligibility criteria to other features of clinical trials that may be impediments to patient participation and thereby limit the diversity of the populations in the studies,” he added.
That said, Dr. Schilsky said he welcomed the draft document as a step toward getting broader participation from patients who are more representative of the ultimate users of these treatments.
“All people who could potentially benefit from the trial should have the opportunity to participate as long as it’s safe for them to do so, and they should not be excluded based upon some of these arbitrary structural things,” he said.
“More importantly, we need data on how these new interventions perform in the patients who are seen and treated by doctors every day. We don’t want data just on patients who have no comorbid illnesses, patients who are otherwise perfectly well, patients who could run a marathon before they go to the doctor’s office. Those are generally not the patients that most doctors are seeing. If they are not represented in the trial population, then we are left with having to extrapolate from the trial data to a population for whom there is no information on how to use the treatment,” he added.
Public comments on the draft document are due on Aug. 6.
Officials at the Food and Drug Administration have issued draft guidance aimed at increasing the diversity of clinical trial populations, including adding children and adolescents earlier in drug development and making trial participation less burdensome for patients.
Despite efforts to enroll clinical trial participants who better reflect a real-world population, “challenges to participation in clinical trials remain, and certain groups continue to be unnecessarily underrepresented in many clinical trials,” the FDA noted in the document’s introduction.
The FDA noted that these challenges could have a significant impact on trial outcomes.
For example, the failure to include complex patients in a clinical trial “may lead to a failure to discover important safety information about the use of the investigational drug in patients who will take the drug after approval.”
In its draft recommendations, the FDA calls on trial sponsors to conduct a closer examination of exclusion criteria and to make it as narrow as possible; consider whether criteria from phase 2 studies, which are restrictive but often transferred to phase 3 protocols, can be eliminated or modified; and consider including children and adolescents when appropriate.
Recommendations related to trial design include characterizing early on the drug metabolism and clearance across populations that may metabolize or clear the drug differently, such as elderly or patients with liver or kidney dysfunction. The agency also called on trial sponsors to use “adaptive clinical trials,” which allow for prespecified trial design changes during the trial, and an early pediatric development program.
Another set of draft recommendations relates to ensuring trial participation is less burdensome for patients by reducing the frequency of study visits and making participants more aware of reimbursement for travel and lodging associated with the trial.
Additionally, the agency is recommending that trial sponsors adopt enrollment and retention practices that enhance inclusiveness, such as ensuring that trial sites include geographic locations with a higher concentration of racial and ethnic minority patients, and holding recruitment events on nights and weekends and in nonclinical locations.
Finally, the FDA issued a set of recommendations to trial sponsors aimed at broadening eligibility criteria when evaluating drugs intended for the treatment of rare diseases. In those trials, the FDA recommended reenrolling participants from early-phase trials in later-phase trials if it can be done safely.
“Because rare diseases often affect small, geographically dispersed patient populations with disease-related travel limitations, special efforts may be necessary to enroll and retain these participants to ensure that a broad spectrum of the patient population is represented,” the agency stated.
At first blush, the draft recommendations are being greeted with a positive response.
“We certainly need more diversity in clinical trial populations, so I think anything that FDA can say that will help encourage that is a good thing,” Richard Schilsky, MD, senior vice president and chief medical officer at the American Society of Clinical Oncology, said in an interview, noting that this new guidance builds off the work that ASCO and Friends of Cancer Research did with the FDA on previous efforts to expand clinical trial populations.
“I think that this new guidance, as best as I can tell, builds off of that, generalizes it across all therapeutic areas and goes a little bit beyond eligibility criteria to other features of clinical trials that may be impediments to patient participation and thereby limit the diversity of the populations in the studies,” he added.
That said, Dr. Schilsky said he welcomed the draft document as a step toward getting broader participation from patients who are more representative of the ultimate users of these treatments.
“All people who could potentially benefit from the trial should have the opportunity to participate as long as it’s safe for them to do so, and they should not be excluded based upon some of these arbitrary structural things,” he said.
“More importantly, we need data on how these new interventions perform in the patients who are seen and treated by doctors every day. We don’t want data just on patients who have no comorbid illnesses, patients who are otherwise perfectly well, patients who could run a marathon before they go to the doctor’s office. Those are generally not the patients that most doctors are seeing. If they are not represented in the trial population, then we are left with having to extrapolate from the trial data to a population for whom there is no information on how to use the treatment,” he added.
Public comments on the draft document are due on Aug. 6.
Long-term trend: Women receiving fewer pelvic exams
according to the National Center for Health Statistics.
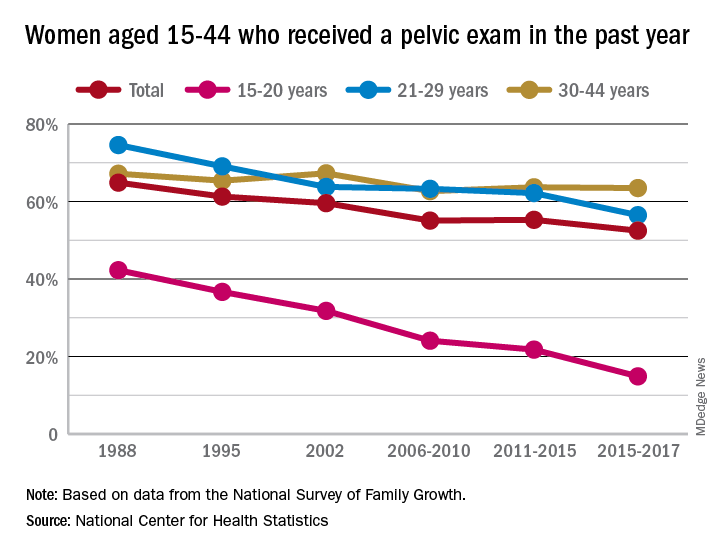
Sixty-five percent of women aged 15-44 years had received a pelvic examination in the past year when asked in 1988 as part of the National Survey of Family Growth, but the 3-year average for the 2015-2017 surveys was 53%, a significant decline, the NCHS said in a recent report.
The decrease was seen in all three of the age subgroups – 15-20 years, 21-29 years, and 30-44 years – over the length of the study period, with the trend in only the oldest women not reaching significance. The 30-44 group also was the only one of the three in which the rate ever increased at any point, the survey data show.
Data for other subgroups focused on the last 3-year period. From 2015 to 2017, non-Hispanic black women were more likely to have received a pelvic examination in the past year (60%) than were non-Hispanic white (54%) or Hispanic women (45%). An association with education level also was seen: Women with a bachelor’s degree or higher were most likely to get an exam (69%), and those with less than a high-school degree were least likely (52%), the researchers reported.
In 2018, the American College of Obstetricians and Gynecologists altered its recommendation that annual pelvic examinations be part of the well-woman visit for those aged 21 years and over, advising instead “that pelvic examinations be performed when indicated by medical history or symptoms,” the NCHS authors explained. They also suggested that their data “could provide a benchmark for estimates of the prevalence of pelvic examinations before the 2018 ACOG-updated guidelines.”
according to the National Center for Health Statistics.

Sixty-five percent of women aged 15-44 years had received a pelvic examination in the past year when asked in 1988 as part of the National Survey of Family Growth, but the 3-year average for the 2015-2017 surveys was 53%, a significant decline, the NCHS said in a recent report.
The decrease was seen in all three of the age subgroups – 15-20 years, 21-29 years, and 30-44 years – over the length of the study period, with the trend in only the oldest women not reaching significance. The 30-44 group also was the only one of the three in which the rate ever increased at any point, the survey data show.
Data for other subgroups focused on the last 3-year period. From 2015 to 2017, non-Hispanic black women were more likely to have received a pelvic examination in the past year (60%) than were non-Hispanic white (54%) or Hispanic women (45%). An association with education level also was seen: Women with a bachelor’s degree or higher were most likely to get an exam (69%), and those with less than a high-school degree were least likely (52%), the researchers reported.
In 2018, the American College of Obstetricians and Gynecologists altered its recommendation that annual pelvic examinations be part of the well-woman visit for those aged 21 years and over, advising instead “that pelvic examinations be performed when indicated by medical history or symptoms,” the NCHS authors explained. They also suggested that their data “could provide a benchmark for estimates of the prevalence of pelvic examinations before the 2018 ACOG-updated guidelines.”
according to the National Center for Health Statistics.

Sixty-five percent of women aged 15-44 years had received a pelvic examination in the past year when asked in 1988 as part of the National Survey of Family Growth, but the 3-year average for the 2015-2017 surveys was 53%, a significant decline, the NCHS said in a recent report.
The decrease was seen in all three of the age subgroups – 15-20 years, 21-29 years, and 30-44 years – over the length of the study period, with the trend in only the oldest women not reaching significance. The 30-44 group also was the only one of the three in which the rate ever increased at any point, the survey data show.
Data for other subgroups focused on the last 3-year period. From 2015 to 2017, non-Hispanic black women were more likely to have received a pelvic examination in the past year (60%) than were non-Hispanic white (54%) or Hispanic women (45%). An association with education level also was seen: Women with a bachelor’s degree or higher were most likely to get an exam (69%), and those with less than a high-school degree were least likely (52%), the researchers reported.
In 2018, the American College of Obstetricians and Gynecologists altered its recommendation that annual pelvic examinations be part of the well-woman visit for those aged 21 years and over, advising instead “that pelvic examinations be performed when indicated by medical history or symptoms,” the NCHS authors explained. They also suggested that their data “could provide a benchmark for estimates of the prevalence of pelvic examinations before the 2018 ACOG-updated guidelines.”
ACIP approves meningococcal booster for persons at increased risk
according to the Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices.
The committee voted unanimously in favor of a booster dose of MenB vaccine 1 year after completion of the primary series, with additional boosters every 2-3 years “for as long as risk remains” for high-risk persons, including microbiologists and persons with complement deficiency, complement inhibitor use, or asplenia.
The committee also voted unanimously in favor of a one-time MenB booster for individuals aged 10 years and older who are at least a year beyond completion of a MenB primary series and deemed at increased risk by public health officials in an outbreak situation.
In addition, “a booster dose interval of 6 months or more may be considered by public health officials depending on the specific outbreak, vaccine strategy, and projected duration of elevated risk” according to the language, which was included in the unanimously approved statement “Meningococcal Vaccination: Recommendations of The Advisory Committee on Immunization Practices.”
The updated statement on meningococcal vaccination was developed in 2019 “to consolidate all existing ACIP recommendations for MenACWY and MenB vaccines in a single document,” said Sarah Mbaeyi, MD, of the CDC’s National Center for Immunization and Respiratory Diseases, who presented immunogenicity data and the proposed recommendations.
The statement includes the recommendation of a MenB primary series for individuals aged 16-23 years based on shared clinical decision making. Kelly Moore, MD, of Vanderbilt University, Nashville, Tenn., noted the importance of ongoing data collection, and said clinicians must make clear to patients that, “if they want protection, they need the booster.”
Approximately 7% of serogroup B cases in the United States are related to disease outbreaks, mainly among college students, Dr. Mbaeyi said. All 13 universities that experienced outbreaks between 2013 and 2019 have implemented a MenB primary series, and one university has implemented an off-label booster program.
The work group concluded that a MenB booster dose is necessary to sustain protection against serogroup B disease in persons at increased risk during an outbreak, and that the potential benefits outweighed the harms given the seriousness of meningococcal disease.
Paul Hunter, MD, of the City of Milwaukee Health Department, noted that “the booster recommendation gives more flexibility” in an outbreak response.
The committee also voted unanimously to approve the Vaccines for Children resolution for the meningococcal vaccine that updates language to align with the new recommendations.
The ACIP members had no financial conflicts to disclose.
according to the Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices.
The committee voted unanimously in favor of a booster dose of MenB vaccine 1 year after completion of the primary series, with additional boosters every 2-3 years “for as long as risk remains” for high-risk persons, including microbiologists and persons with complement deficiency, complement inhibitor use, or asplenia.
The committee also voted unanimously in favor of a one-time MenB booster for individuals aged 10 years and older who are at least a year beyond completion of a MenB primary series and deemed at increased risk by public health officials in an outbreak situation.
In addition, “a booster dose interval of 6 months or more may be considered by public health officials depending on the specific outbreak, vaccine strategy, and projected duration of elevated risk” according to the language, which was included in the unanimously approved statement “Meningococcal Vaccination: Recommendations of The Advisory Committee on Immunization Practices.”
The updated statement on meningococcal vaccination was developed in 2019 “to consolidate all existing ACIP recommendations for MenACWY and MenB vaccines in a single document,” said Sarah Mbaeyi, MD, of the CDC’s National Center for Immunization and Respiratory Diseases, who presented immunogenicity data and the proposed recommendations.
The statement includes the recommendation of a MenB primary series for individuals aged 16-23 years based on shared clinical decision making. Kelly Moore, MD, of Vanderbilt University, Nashville, Tenn., noted the importance of ongoing data collection, and said clinicians must make clear to patients that, “if they want protection, they need the booster.”
Approximately 7% of serogroup B cases in the United States are related to disease outbreaks, mainly among college students, Dr. Mbaeyi said. All 13 universities that experienced outbreaks between 2013 and 2019 have implemented a MenB primary series, and one university has implemented an off-label booster program.
The work group concluded that a MenB booster dose is necessary to sustain protection against serogroup B disease in persons at increased risk during an outbreak, and that the potential benefits outweighed the harms given the seriousness of meningococcal disease.
Paul Hunter, MD, of the City of Milwaukee Health Department, noted that “the booster recommendation gives more flexibility” in an outbreak response.
The committee also voted unanimously to approve the Vaccines for Children resolution for the meningococcal vaccine that updates language to align with the new recommendations.
The ACIP members had no financial conflicts to disclose.
according to the Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices.
The committee voted unanimously in favor of a booster dose of MenB vaccine 1 year after completion of the primary series, with additional boosters every 2-3 years “for as long as risk remains” for high-risk persons, including microbiologists and persons with complement deficiency, complement inhibitor use, or asplenia.
The committee also voted unanimously in favor of a one-time MenB booster for individuals aged 10 years and older who are at least a year beyond completion of a MenB primary series and deemed at increased risk by public health officials in an outbreak situation.
In addition, “a booster dose interval of 6 months or more may be considered by public health officials depending on the specific outbreak, vaccine strategy, and projected duration of elevated risk” according to the language, which was included in the unanimously approved statement “Meningococcal Vaccination: Recommendations of The Advisory Committee on Immunization Practices.”
The updated statement on meningococcal vaccination was developed in 2019 “to consolidate all existing ACIP recommendations for MenACWY and MenB vaccines in a single document,” said Sarah Mbaeyi, MD, of the CDC’s National Center for Immunization and Respiratory Diseases, who presented immunogenicity data and the proposed recommendations.
The statement includes the recommendation of a MenB primary series for individuals aged 16-23 years based on shared clinical decision making. Kelly Moore, MD, of Vanderbilt University, Nashville, Tenn., noted the importance of ongoing data collection, and said clinicians must make clear to patients that, “if they want protection, they need the booster.”
Approximately 7% of serogroup B cases in the United States are related to disease outbreaks, mainly among college students, Dr. Mbaeyi said. All 13 universities that experienced outbreaks between 2013 and 2019 have implemented a MenB primary series, and one university has implemented an off-label booster program.
The work group concluded that a MenB booster dose is necessary to sustain protection against serogroup B disease in persons at increased risk during an outbreak, and that the potential benefits outweighed the harms given the seriousness of meningococcal disease.
Paul Hunter, MD, of the City of Milwaukee Health Department, noted that “the booster recommendation gives more flexibility” in an outbreak response.
The committee also voted unanimously to approve the Vaccines for Children resolution for the meningococcal vaccine that updates language to align with the new recommendations.
The ACIP members had no financial conflicts to disclose.
REPORTING FROM AN ACIP MEETING
FDA approves bevacizumab-bvzr for several cancers
The Food and Drug Administration has approved bevacizumab-bvzr (Zirabev) – a biosimilar to bevacizumab (Avastin) – for the treatment of five cancers: metastatic colorectal cancer (mCRC); unresectable, locally advanced, recurrent or metastatic non-squamous non–small cell lung cancer (NSCLC); recurrent glioblastoma; metastatic renal cell carcinoma (RCC); and persistent, recurrent or metastatic cervical cancer.
Approval was based on “review of a comprehensive data package which demonstrated biosimilarity of [bevacizumab-bvzr] to the reference product,” Pfizer said in a statement announcing the approval.
Bevacizumab-bvzr is the second bevacizumab biosimilar to be approved, following approval of Amgen’s bevacizumab-awwb (Mvasi) in 2017.
Warnings and precautions with the biosimilars, as with bevacizumab, include serious and sometimes fatal gastrointestinal perforation, surgery and wound healing complications, and sometimes serious and fatal hemorrhage.
The most common adverse events observed in bevacizumab patients are epistaxis, headache, hypertension, rhinitis, proteinuria, taste alteration, dry skin, rectal hemorrhage, lacrimation disorder, back pain, and exfoliative dermatitis.
Specific indications for the biosimilar are as follows:
Metastatic colorectal cancer
Bevacizumab-bvzr, in combination with intravenous fluorouracil-based chemotherapy, is indicated for the first- or second-line treatment of patients with mCRC.
Bevacizumab-bvzr, in combination with fluoropyrimidine-irinotecan or fluoropyrimidine-oxaliplatin–based chemotherapy, is indicated for the second-line treatment of patients with mCRC who have progressed on a first-line bevacizumab product–containing regimen.
Bevacizumab-bvzr is not indicated for adjuvant treatment of colon cancer.
First-line nonsquamous non–small cell lung cancer
Bevacizumab-bvzr, in combination with carboplatin and paclitaxel, is indicated for the first-line treatment of patients with unresectable, locally advanced, recurrent or metastatic NSCLC.
Recurrent glioblastoma
Bevacizumab-bvzr is indicated for the treatment of recurrent glioblastoma in adults.
Metastatic renal cell carcinoma
Bevacizumab-bvzr, in combination with interferon alfa, is indicated for the treatment of metastatic RCC.
Persistent, recurrent, or metastatic cervical cancer
Bevacizumab-bvzr, in combination with paclitaxel and cisplatin or paclitaxel and topotecan, is indicated for the treatment of patients with persistent, recurrent, or metastatic cervical cancer.
Complete prescribing information can be found on the FDA website.
The Food and Drug Administration has approved bevacizumab-bvzr (Zirabev) – a biosimilar to bevacizumab (Avastin) – for the treatment of five cancers: metastatic colorectal cancer (mCRC); unresectable, locally advanced, recurrent or metastatic non-squamous non–small cell lung cancer (NSCLC); recurrent glioblastoma; metastatic renal cell carcinoma (RCC); and persistent, recurrent or metastatic cervical cancer.
Approval was based on “review of a comprehensive data package which demonstrated biosimilarity of [bevacizumab-bvzr] to the reference product,” Pfizer said in a statement announcing the approval.
Bevacizumab-bvzr is the second bevacizumab biosimilar to be approved, following approval of Amgen’s bevacizumab-awwb (Mvasi) in 2017.
Warnings and precautions with the biosimilars, as with bevacizumab, include serious and sometimes fatal gastrointestinal perforation, surgery and wound healing complications, and sometimes serious and fatal hemorrhage.
The most common adverse events observed in bevacizumab patients are epistaxis, headache, hypertension, rhinitis, proteinuria, taste alteration, dry skin, rectal hemorrhage, lacrimation disorder, back pain, and exfoliative dermatitis.
Specific indications for the biosimilar are as follows:
Metastatic colorectal cancer
Bevacizumab-bvzr, in combination with intravenous fluorouracil-based chemotherapy, is indicated for the first- or second-line treatment of patients with mCRC.
Bevacizumab-bvzr, in combination with fluoropyrimidine-irinotecan or fluoropyrimidine-oxaliplatin–based chemotherapy, is indicated for the second-line treatment of patients with mCRC who have progressed on a first-line bevacizumab product–containing regimen.
Bevacizumab-bvzr is not indicated for adjuvant treatment of colon cancer.
First-line nonsquamous non–small cell lung cancer
Bevacizumab-bvzr, in combination with carboplatin and paclitaxel, is indicated for the first-line treatment of patients with unresectable, locally advanced, recurrent or metastatic NSCLC.
Recurrent glioblastoma
Bevacizumab-bvzr is indicated for the treatment of recurrent glioblastoma in adults.
Metastatic renal cell carcinoma
Bevacizumab-bvzr, in combination with interferon alfa, is indicated for the treatment of metastatic RCC.
Persistent, recurrent, or metastatic cervical cancer
Bevacizumab-bvzr, in combination with paclitaxel and cisplatin or paclitaxel and topotecan, is indicated for the treatment of patients with persistent, recurrent, or metastatic cervical cancer.
Complete prescribing information can be found on the FDA website.
The Food and Drug Administration has approved bevacizumab-bvzr (Zirabev) – a biosimilar to bevacizumab (Avastin) – for the treatment of five cancers: metastatic colorectal cancer (mCRC); unresectable, locally advanced, recurrent or metastatic non-squamous non–small cell lung cancer (NSCLC); recurrent glioblastoma; metastatic renal cell carcinoma (RCC); and persistent, recurrent or metastatic cervical cancer.
Approval was based on “review of a comprehensive data package which demonstrated biosimilarity of [bevacizumab-bvzr] to the reference product,” Pfizer said in a statement announcing the approval.
Bevacizumab-bvzr is the second bevacizumab biosimilar to be approved, following approval of Amgen’s bevacizumab-awwb (Mvasi) in 2017.
Warnings and precautions with the biosimilars, as with bevacizumab, include serious and sometimes fatal gastrointestinal perforation, surgery and wound healing complications, and sometimes serious and fatal hemorrhage.
The most common adverse events observed in bevacizumab patients are epistaxis, headache, hypertension, rhinitis, proteinuria, taste alteration, dry skin, rectal hemorrhage, lacrimation disorder, back pain, and exfoliative dermatitis.
Specific indications for the biosimilar are as follows:
Metastatic colorectal cancer
Bevacizumab-bvzr, in combination with intravenous fluorouracil-based chemotherapy, is indicated for the first- or second-line treatment of patients with mCRC.
Bevacizumab-bvzr, in combination with fluoropyrimidine-irinotecan or fluoropyrimidine-oxaliplatin–based chemotherapy, is indicated for the second-line treatment of patients with mCRC who have progressed on a first-line bevacizumab product–containing regimen.
Bevacizumab-bvzr is not indicated for adjuvant treatment of colon cancer.
First-line nonsquamous non–small cell lung cancer
Bevacizumab-bvzr, in combination with carboplatin and paclitaxel, is indicated for the first-line treatment of patients with unresectable, locally advanced, recurrent or metastatic NSCLC.
Recurrent glioblastoma
Bevacizumab-bvzr is indicated for the treatment of recurrent glioblastoma in adults.
Metastatic renal cell carcinoma
Bevacizumab-bvzr, in combination with interferon alfa, is indicated for the treatment of metastatic RCC.
Persistent, recurrent, or metastatic cervical cancer
Bevacizumab-bvzr, in combination with paclitaxel and cisplatin or paclitaxel and topotecan, is indicated for the treatment of patients with persistent, recurrent, or metastatic cervical cancer.
Complete prescribing information can be found on the FDA website.
Daratumumab wins new indication for newly diagnosed myeloma patients
Janssen’s daratumumab (Darzalex) has picked up a sixth adult multiple myeloma indication, this time in combination with lenalidomide (Revlimid) and dexamethasone in newly diagnosed patients ineligible for autologous stem cell transplants.
The phase 3 MAIA trial found that 97 of 368 patients (26.4%) treated with the combination – dubbed DRd – progressed or died at a median follow-up of 28 months, versus 143 of 269 (38.8%) treated with lenalidomide and dexamethasone alone (Rd). An estimated 55.6% of patients on lenalidomide and dexamethasone, versus 70.6% with the daratumumab add-on, were alive without progression at 30 months (N Engl J Med. 2019 May 30;380[22]:2104-15).
Previously approved indications for daratumumab include relapsed or refractory disease after at least one other therapy; and combination treatment with bortezomib, melphalan, and prednisone, also in newly diagnosed patients who are ineligible for transplant.
The most common grade 3 and 4 adverse events reported in the MAIA trial were neutropenia (50.0% for the DRd group versus 35.3% for the Rd group), anemia (11.8% vs. 19.7%), lymphopenia (15.1% vs. 10.7%), and pneumonia (13.7% vs. 7.9%).
Janssen’s daratumumab (Darzalex) has picked up a sixth adult multiple myeloma indication, this time in combination with lenalidomide (Revlimid) and dexamethasone in newly diagnosed patients ineligible for autologous stem cell transplants.
The phase 3 MAIA trial found that 97 of 368 patients (26.4%) treated with the combination – dubbed DRd – progressed or died at a median follow-up of 28 months, versus 143 of 269 (38.8%) treated with lenalidomide and dexamethasone alone (Rd). An estimated 55.6% of patients on lenalidomide and dexamethasone, versus 70.6% with the daratumumab add-on, were alive without progression at 30 months (N Engl J Med. 2019 May 30;380[22]:2104-15).
Previously approved indications for daratumumab include relapsed or refractory disease after at least one other therapy; and combination treatment with bortezomib, melphalan, and prednisone, also in newly diagnosed patients who are ineligible for transplant.
The most common grade 3 and 4 adverse events reported in the MAIA trial were neutropenia (50.0% for the DRd group versus 35.3% for the Rd group), anemia (11.8% vs. 19.7%), lymphopenia (15.1% vs. 10.7%), and pneumonia (13.7% vs. 7.9%).
Janssen’s daratumumab (Darzalex) has picked up a sixth adult multiple myeloma indication, this time in combination with lenalidomide (Revlimid) and dexamethasone in newly diagnosed patients ineligible for autologous stem cell transplants.
The phase 3 MAIA trial found that 97 of 368 patients (26.4%) treated with the combination – dubbed DRd – progressed or died at a median follow-up of 28 months, versus 143 of 269 (38.8%) treated with lenalidomide and dexamethasone alone (Rd). An estimated 55.6% of patients on lenalidomide and dexamethasone, versus 70.6% with the daratumumab add-on, were alive without progression at 30 months (N Engl J Med. 2019 May 30;380[22]:2104-15).
Previously approved indications for daratumumab include relapsed or refractory disease after at least one other therapy; and combination treatment with bortezomib, melphalan, and prednisone, also in newly diagnosed patients who are ineligible for transplant.
The most common grade 3 and 4 adverse events reported in the MAIA trial were neutropenia (50.0% for the DRd group versus 35.3% for the Rd group), anemia (11.8% vs. 19.7%), lymphopenia (15.1% vs. 10.7%), and pneumonia (13.7% vs. 7.9%).