User login
VIP Boot Camp: Expanding the Impact of VA Primary Care Mental Health With a Transdiagnostic Modular Group Program
VIP Boot Camp: Expanding the Impact of VA Primary Care Mental Health With a Transdiagnostic Modular Group Program
Since 2007, Primary Care Mental Health Integration (PCMHI) at the Veterans Health Administration (VHA) has improved access to mental health care services for veterans by directly embedding mental health care professionals (HCPs) within primary care teams.1 Veterans referred to PCMHI often have co-occurring physical and mental health disorders.2 Untreated chronic physical and mental comorbidities can diminish the effectiveness of medical and mental health interventions. Growing evidence suggests that treatment of mental health conditions can improve physical health outcomes and management of physical conditions can improve mental health outcomes.2,3
Chronic pain and sleep disorders are common reasons patients present to primary care, and often coexist together with mental health comorbidities.4 Sleep disorders affect 50% to 88% of patients with chronic pain, and 40% of patients with sleep disorders report chronic pain.4 Research has found that chronic pain and sleep disorders increase the risk of suicide attempts and deaths by suicide. Addressing suicide prevention simultaneously with treating chronic pain and insomnia is encouraged.5
Background
PCMHI treats physical and mental health comorbidities with a collaborative framework and a biopsychosocial integrative model.6 PCMHI staff provide mental health services as members of primary care teams. An interdisciplinary PCMHI team can include, but is not limited to, psychologists, mental health social workers, psychiatrists, nurse practitioners, clinical pharmacists, and mental health nurses. Quality of care within this model is elevated, as mental and physical health are recognized as interconnected. Collaboration between primary care and mental health benefits veterans and the VHA by increasing access to mental health care, decreasing stigma associated with mental health treatment, improving health outcomes, and enhancing the likelihood of recovery, resulting in high patient satisfaction.6-8
In the existing PCMHI model, HCPs are encouraged to use short-term, evidence-based psychotherapies (EBPs).9 Veterans referred to PCMHI from primary care are typically able to attend 1 to 6 brief sessions of mental health treatment, often 20 to 30 minutes long. Most EBPs in PCMHI are disorder- specific, providing interventions focused on a single presenting problem (eg, insomnia, chronic pain, or posttraumatic stress disorder [PTSD]). For veterans with a single issue, this model can be very effective. 1,10 However, the high rate of co-occurrence of mental and physical health issues can make it difficult to fully treat interrelated problems if the focus is on 1 specific diagnosis. Veterans with a need for additional (more comprehensive or intensive) mental health treatment are frequently referred to a higher, more resource-intensive level of mental health care, either in the VHA or the community. Examples of higher levels of mental health care include the longer term behavioral health interdisciplinary program (BHIP), sometimes called a mental health clinic (MHC), or a specialty mental health program such as a PTSD clinic.
As PCMHI continues to grow, new challenges have emerged related to staffing shortages and gaps in the clinical delivery of mental health treatment within the VHA. At the same time, demand for VHA mental health treatment has increased. However, a mental health professional shortage severely limits the ability of the VHA to meet this demand. In many systems, this shortage may result in more referrals being made to a higher level of mental health care because of fewer resources to provide comprehensive treatment in a less intensive PCMHI setting.8,10,11 This referral pattern can overburden higher level care, often with long wait times for treatment and lengthy lag times between appointments. Furthermore, these gaps in the clinical delivery of care cannot be effectively addressed by hiring additional mental health professionals. This strain on resources can impede access to care and negatively affect outcomes.10
Recent congressional reports highlight these issues, noting that demand for mental health services continues to outpace the capacity of both PCMHI and higher levels of mental health care, leading to delays in treatment that may negatively affect outcomes.8,10,11 These delays can be particularly detrimental for individuals with conditions requiring timely intervention.8,11 Some veterans are willing to engage with PCMHI in a primary care setting but may be reluctant to engage in general mental health treatment. These veterans might not receive the mental health care they need without PCMHI.
Group Psychotherapy
A group psychotherapy format can address gaps in care delivery and provide advantages for patients, mental health professionals, and the VHA. Group psychotherapy aligns with the US Department of Veterans Affairs (VA) 2018 Blueprint for Excellence and 2018 to 2024 strategic plan, underscoring the need for more timely and efficient mental health services.12,13
Benefits of group psychotherapy include reductions in symptoms, decreased feelings of isolation, increased social support, decreased emotional suppression, and enhanced satisfaction with overall quality of life.14-17 Studies of veterans with PTSD have found less attrition among those who chose group therapy compared with individual therapy.14,18 Group psychotherapy improves access to care by enabling delivery to more patients.14 When compared with individual therapy, the group format allows for a large number of patients to be treated simultaneously, maximizing resources and reducing costs.3,19-21
VISN 9 CRH Innovation
The VA provides care to veterans through regionally distinct administrative systems known as Veterans Integrated Service Networks (VISNs). Clinical resource hubs (CRH) are VISN-based programs created to cover VA staffing shortages by virtually deploying HCPs into local VA systems until vacancies are filled. The national CRH vision of effectively using resources and innovative technologies to meet veterans’ health care needs, along with the above-referenced clinical gaps in the delivery of care, inspired the development of VIP Boot Camp within the VISN 9 CRH.22
Program Description
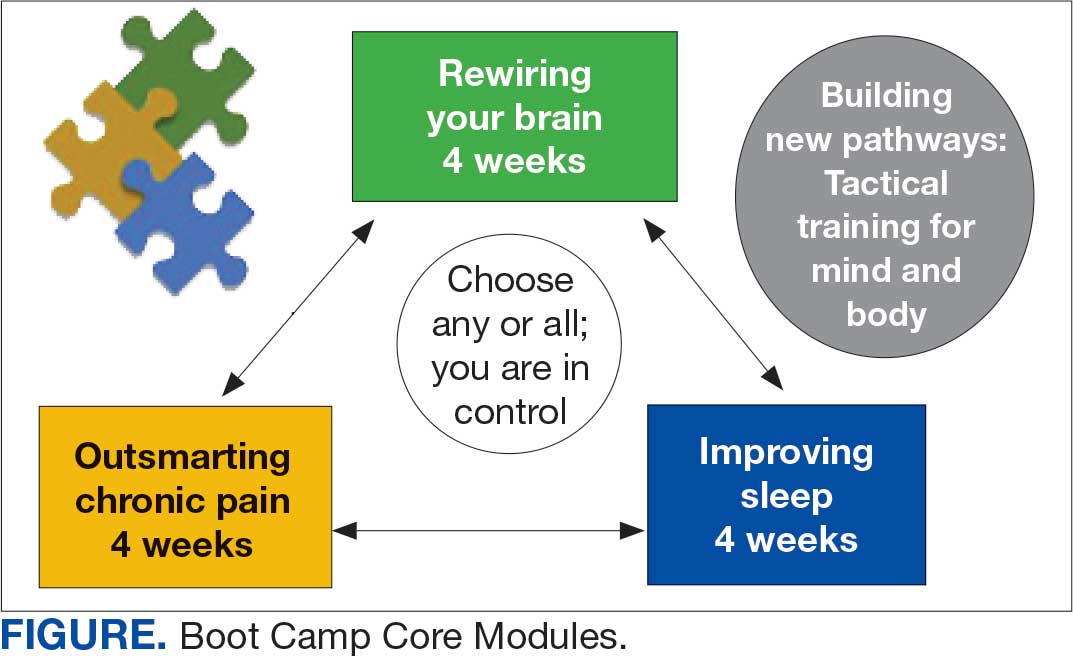
VIP Boot Camp is an evidence-informed group psychotherapy program designed to provide timely, brief, and comprehensive mental health treatment for veterans. VIP Boot Camp was developed to address the needs of veterans accessing PCMHI services who experience ≥ 1 of the often overlapping problems of anxiety/emotion regulation/stress, sleep difficulties, and chronic pain (Figure). VIP Boot Camp uses an integrative approach to highlight interconnections and similarities among these difficulties and their treatment. A primary vision of the program is to provide this comprehensive treatment within PCMHI (upstream) so additional referrals to higher levels of mental health care (downstream) may not be needed.
This design is intentional because it increases the number of individuals who can be treated upstream with comprehensive, preventive, and proactive care within PCMHI which, over time, frees up resources in the BHIP for individuals requiring higher levels of care. This approach also aligns with the importance of early treatment for chronic pain and sleep disturbances, which are linked to increased risk of suicide attempts and deaths by suicide for veterans.5 National interest for VIP Boot Camp grew during fiscal year 2024 after it received the Gold Medal Recognition for Most Adoptable and Greatest Potential for Impact during VHA National Access Sprint Wave 3—Mental Health Call of Champions.
History
VIP Boot Camp began in August 2021 at VISN 9 as a 6-week virtual group for veterans with chronic pain. It was established to assist a large VA medical center experiencing PCMHI staffing shortages and lacking available PCMHI groups. Many veterans in the chronic pain group discussed co-occurring issues such as sleep disturbances, anxiety, and stress. The CRH team considered launching 2 separate groups to address these additional PCMHI-level issues; however, in developing the group material which drew from multiple clinical approaches, the team recognized significant overlapping and interconnected themes.
The team discussed EBPs within the VHA and how certain interventions within these treatments could be helpful across many other co-occurring disorders. Integrated tactics (clinical interventions) were drawn from cognitive-behavioral therapy (for depression, insomnia, or chronic pain), acceptance and commitment therapy, prolonged exposure, cognitive processing therapy, dialectical behavior therapy, unified protocol, pain reprocessing therapy, emotional awareness and expression therapy, interpersonal neurobiology, and mindfulness. We collaborated with veterans during VIP Boot Camp groups to determine how to present and discuss complex interventions in ways that were clinically accurate, understandable, relatable, and relevant to their experiences.
To address accessibility issues, the chronic pain group was reduced to 4 weeks. A second 4-week module for anxiety, emotion regulation, and stress was developed, mirroring the tactics, language, and integrative approach of the revised chronic pain module. A similar integrative approach led to the development of the third and final 4-week module for sleep disturbances.
Current Program
The VIP Boot Camp consists of three 4-week integrated modules, each highlighting a critical area: sleep disturbances (Improving Sleep), chronic pain difficulties (Outsmarting Chronic Pain), and emotion regulation difficulties (Rewiring Your Brain). VIP Boot Camp is designed for veterans who are at the PCMHI level of care. Referrals are accepted for patients receiving treatment from primary care or PCMHI.
Guidelines for participation in VIP Boot Camp may differ across sites or VISNs. For example, a veteran who has been referred to the BHIP for medication management only or to a specialty MHC such as a pain clinic or PTSD clinic might also be appropriate and eligible for VIP Boot Camp.
Given the interconnectedness of foundational themes, elements, and practices across the VIP Boot Camp modules, the modules are offered in a rolling format with a veteran-centric “choose your own adventure” approach. Tactics are presented in the modules in a way that allows patients to begin with any 1 of the 3 modules and receive treatment that will help in the other areas. Participants choose their core module and initial treatment focus based on their values, needs, and goals. Individuals who complete a core module can end their VIP Boot Camp experience or continue to the next 4-week module for up to 3 modules.
The group is open to new individuals at the start of any 4-week module and closed for the remainder of its 4-week duration. This innovative rolling modular approach combines elements of open- and closed-group format, allowing for the flexibility and accessibility of an open group with the stability and peer support of a closed group.
Given the complicated and overlapping nature of chronic pain, emotion regulation/ stress, and sleep disturbances, VIP Boot Camp acknowledges that everything is interconnected and difficulties in 1 area may impact other areas. The 3 interconnected modules with repeating themes provide coherence and consistency. Veterans learn how interconnections across difficulties can be leveraged so that tactics learned and practiced in 1 area can assist in other areas, changing the cycle of suffering into a cycle of growth.
VIP Boot Camp sessions are 90 minutes long, once weekly for 4 weeks, with 2 mental health professionals trained to lead a dynamic group psychotherapy experience that aims to be fun for participants. VIP Boot Camp synthesizes evidence-based and evidence-informed interventions, as well as techniques from VHA complementary and integrative health programs, psychoeducation, and interpersonal interventions that model connection, playfulness, and healthy boundaries. These varied strategies combine to equip veterans with practical tactics for self-management outside of sessions, a process described as “finding puzzle pieces.” VIP Boot Camp is built on the idea that people are more likely to adopt and practice any tactic after being taught why that tactic is important, and how it fits into their larger interconnected puzzle. After each session, participants are provided with additional asynchronous educational material to help reinforce their learnings and practices.
Although individuals may hesitate to participate in a group setting, they often find the experience of community enhances and accelerates their treatment and gains. This involvement is highlighted in a core aspect of a VIP Boot Camp session called wins, during which participants learn how others on their Boot Camp team are implementing new skills and moving toward their personal values and objectives in a stepwise manner. Through these shared experiences, veterans discover how tactics working for others may serve as a model for their own personal objectives and plans for practice. The sense of relief described by many upon realizing they are not alone in their experiences, along with the satisfaction felt in discovering their ability to support others in Boot Camp, is described by many participants as deeply meaningful and in line with their personal values.
While developed as a fully virtual group program, VIP Boot Camp can also be conducted in person. The virtual program has been successful and continues to spread across VISN 9. There are 8 virtual VIP Boot Camps running in VISN 9, with plans for continued expansion. In the VISN 9 CRH, Boot Camps typically have 10 to 12 participants. Additionally, as VIP Boot Camp grows within a location there are frequently sufficient referrals to support a second rolling group, which enables staggering of the module offerings to allow for even more timely treatment.
Training Program
VISN 9 CRH also developed a VIP Boot Camp 3-day intensive training program for PCMHI HCPs that consists of learning and practicing VIP Boot Camp material for chronic pain, emotion regulation/ stress, sleep disturbances, mindfulness, and guided imagery, along with gaining experience as a VIP Boot Camp coleader. Feedback received from PCMHI HCPs who completed training has been positive. There is also a private Microsoft Teams channel for HCPs, which allows for resource sharing and community building among coleaders. More than 75 PCMHI HCPs have completed VIP Boot Camp training and > 25 VIP Boot Camps have been established at 4 additional VISNs.
The VISN 9 CRH VIP Boot Camp program initiated an implementation and effectiveness project with the Michael E. DeBakey VA Medical Center and the South Central Mental Illness Research, Education and Clinical Center. The focus of this collaboration is support for implementation and treatment effectiveness research with reports, articles, and a white paper on findings and best practices, alongside continued dissemination of the VIP Boot Camp program and training.
Conclusions
VIP Boot Camp is a PCMHI group program offering readily available, comprehensive, and integrative group psychotherapy services to veterans experiencing . 1 of the following: chronic pain, emotion regulation/ stress, and sleep disturbances. It was launched at the VISN 9 CRH with a goal of addressing clinical gaps in the delivery of mental health care, by increasing the number of patients treated within PCMHI. The VIP Boot Camp model provides veterans the opportunity to transform cycles of suffering into cycles of growth through a single approach that can address multiple presenting and interconnected issues.
A 3-day VIP Boot Camp training program provides a quick and effective path for a PCMHI program to train HCPs to launch a VIP Boot Camp. The VISN 9 CRH will continue to champion VIP Boot Camp as a model for the successful provision of comprehensive and integrative mental health treatment within PCMHI at the VA. Through readily available access to comprehensive mental health treatment in an environment that promotes participant empowerment and social engagement, VIP Boot Camp represents an integrative and innovative model of mental health treatment that offers benefits to veteran participants, HCPs, and the VHA.
- Leung LB, Yoon J, Escarce JJ, et al. Primary care-mental health integration in the VA: shifting mental health services for common mental illnesses to primary care. Psychiatr Serv. 2018;69:403-409. doi:10.1176/appi.ps.201700190
- Zhang A, Park S, Sullivan JE, et al. The effectiveness of problem-solving therapy for primary care patients’ depressive and/or anxiety disorders: a systematic review and meta-analysis. J Am Board Fam Med. 2018;31:139-150. doi:10.3122/jabfm.2018.01.170270
- Hundt NE, Barrera TL, Robinson A, et al. A systematic review of cognitive behavioral therapy for depression in veterans. Mil Med. 2014;179:942-949. doi:10.7205/milmed-d-14-00128
- Jank R, Gallee A, Boeckle M, et al. Chronic pain and sleep disorders in primary care. Pain Res Treat. 2017;2017:1-9. doi:10.1155/2017/9081802
- Ashrafioun L, Bishop TM, Pigeon WR. The relationship between pain severity, insomnia, and suicide attempts among a national veteran sample initiating pain care. Psychosom Med. 2021;83:733- 738. doi:10.1097/psy.0000000000000975
- Ramanuj P, Ferenchik E, Docherty M, et al. Evolving models of integrated behavioral health and primary care. Curr Psychiatry Rep. 2019;21:1. doi:10.1007/s11920-019-0985-4
- Post EP, Metzger M, Dumas P, et al. Integrating mental health into primary care within the Veterans Health Administration. Fam Syst Health. 2010;28:83-90. doi:10.1037/a0020130
- Smith TL, Kim B, Benzer JK, et al. FLOW: early results from a clinical demonstration project to improve the transition of patients with mental health disorders back to primary care. Psychol Serv. 2021;18:23-32. doi:10.1037/ser0000336
- Kearney LK, Post EP, Pomerantz AS, et al. Applying the interprofessional patient aligned care team in the department of veterans affairs transforming primary care. Am Psychol. 2014;69(4):399-408. doi:10.1037/a0035909
- US Government Accountability Office. Veterans health care: staffing challenges persist for fully integrating mental health and primary care services. December 15, 2022. Accessed July 9, 2025. https://www.gao.gov/products/gao-23-105372
- National Academies of Science and Engineering. Evaluation of the Department of Veterans Affairs Mental Health Services. National Academies Press; 2018. Accessed July 9, 2025. https://nap.nationalacademies.org/catalog/24915/evaluation-of-the-department-of-veterans-affairs-mental-health-services
- US Department of Veterans Affairs. Blueprint for excellence: achieving veterans’ excellence. October 6, 2014. Accessed July 9, 2025. https://www.volunteer.va.gov/docs/blueprintforexcellence_factsheet.PDF
- US Department of Veterans Affairs. Department of Veterans Affairs FY 2018-2024 strategic plan. Accessed July 9, 2025. https://www.calvet.ca.gov/Regulations/USDVA%20Strategic%20Plan%202018-2024.pdf
- Sripada RK, Bohnert KM, Ganoczy D, et al. Initial group versus individual therapy for posttraumatic stress disorder and subsequent follow-up treatment adequacy. Psychol Serv. 2016;13:349-355. doi:10.1037/ser0000077
- Burnett-Zeigler IE, Pfeiffer P, Zivin K, et al. Psychotherapy utilization for acute depression within the Veterans Affairs health care system. Psychol Serv. 2012;9:325-335. doi:10.1037/a0027957
- Kim JS, Prins A, Hirschhorn EW, et al. Preliminary investigation into the effectiveness of group webSTAIR for trauma-exposed veterans in primary care. Mil Med. 2024;189:e1403-e1408. doi:10.1093/milmed/usae052
- Jakupcak M, Blais RK, Grossbard J, et al. “Toughness” in association with mental health symptoms among Iraq and Afghanistan war veterans seeking Veterans Affairs health care. Psychol Men Masc. 2014;15:100-104. doi:10.1037/a0031508
- Stoycos SA, Berzenski SR, Beck JG, et al. Predictors of treatment completion in group psychotherapy for male veterans with posttraumatic stress disorder. J Trauma Stress. 2023;36:346-358. doi:10.1002/jts.22915
- Possemato K. The current state of intervention research for posttraumatic stress disorder within the primary care setting. J Clin Psychol Med Settings. 2011;18:268-280. doi:10.1007/s10880-011-9237-4
- Hunt MG, Rosenheck RA. Psychotherapy in mental health clinics of the Department of Veterans Affairs. J Clin Psychol. 2011;67:561-573. doi:10.1002/jclp.20788
- Khatri N, Marziali E, Tchernikov I, et al. Comparing telehealth-based and clinic-based group cognitive behavioral therapy for adults with depression and anxiety: a pilot study. Clin Interv Aging. 2014;9:765. doi:10.2147/cia.s57832
- Dangel J. Clinical resource hub increases veterans' access to care. VA News. January 12, 2025. Accessed September 3, 2025. https://news.va.gov/137439/clinical-resource-hub-increases-access-to-care/
Since 2007, Primary Care Mental Health Integration (PCMHI) at the Veterans Health Administration (VHA) has improved access to mental health care services for veterans by directly embedding mental health care professionals (HCPs) within primary care teams.1 Veterans referred to PCMHI often have co-occurring physical and mental health disorders.2 Untreated chronic physical and mental comorbidities can diminish the effectiveness of medical and mental health interventions. Growing evidence suggests that treatment of mental health conditions can improve physical health outcomes and management of physical conditions can improve mental health outcomes.2,3
Chronic pain and sleep disorders are common reasons patients present to primary care, and often coexist together with mental health comorbidities.4 Sleep disorders affect 50% to 88% of patients with chronic pain, and 40% of patients with sleep disorders report chronic pain.4 Research has found that chronic pain and sleep disorders increase the risk of suicide attempts and deaths by suicide. Addressing suicide prevention simultaneously with treating chronic pain and insomnia is encouraged.5
Background
PCMHI treats physical and mental health comorbidities with a collaborative framework and a biopsychosocial integrative model.6 PCMHI staff provide mental health services as members of primary care teams. An interdisciplinary PCMHI team can include, but is not limited to, psychologists, mental health social workers, psychiatrists, nurse practitioners, clinical pharmacists, and mental health nurses. Quality of care within this model is elevated, as mental and physical health are recognized as interconnected. Collaboration between primary care and mental health benefits veterans and the VHA by increasing access to mental health care, decreasing stigma associated with mental health treatment, improving health outcomes, and enhancing the likelihood of recovery, resulting in high patient satisfaction.6-8
In the existing PCMHI model, HCPs are encouraged to use short-term, evidence-based psychotherapies (EBPs).9 Veterans referred to PCMHI from primary care are typically able to attend 1 to 6 brief sessions of mental health treatment, often 20 to 30 minutes long. Most EBPs in PCMHI are disorder- specific, providing interventions focused on a single presenting problem (eg, insomnia, chronic pain, or posttraumatic stress disorder [PTSD]). For veterans with a single issue, this model can be very effective. 1,10 However, the high rate of co-occurrence of mental and physical health issues can make it difficult to fully treat interrelated problems if the focus is on 1 specific diagnosis. Veterans with a need for additional (more comprehensive or intensive) mental health treatment are frequently referred to a higher, more resource-intensive level of mental health care, either in the VHA or the community. Examples of higher levels of mental health care include the longer term behavioral health interdisciplinary program (BHIP), sometimes called a mental health clinic (MHC), or a specialty mental health program such as a PTSD clinic.
As PCMHI continues to grow, new challenges have emerged related to staffing shortages and gaps in the clinical delivery of mental health treatment within the VHA. At the same time, demand for VHA mental health treatment has increased. However, a mental health professional shortage severely limits the ability of the VHA to meet this demand. In many systems, this shortage may result in more referrals being made to a higher level of mental health care because of fewer resources to provide comprehensive treatment in a less intensive PCMHI setting.8,10,11 This referral pattern can overburden higher level care, often with long wait times for treatment and lengthy lag times between appointments. Furthermore, these gaps in the clinical delivery of care cannot be effectively addressed by hiring additional mental health professionals. This strain on resources can impede access to care and negatively affect outcomes.10
Recent congressional reports highlight these issues, noting that demand for mental health services continues to outpace the capacity of both PCMHI and higher levels of mental health care, leading to delays in treatment that may negatively affect outcomes.8,10,11 These delays can be particularly detrimental for individuals with conditions requiring timely intervention.8,11 Some veterans are willing to engage with PCMHI in a primary care setting but may be reluctant to engage in general mental health treatment. These veterans might not receive the mental health care they need without PCMHI.
Group Psychotherapy
A group psychotherapy format can address gaps in care delivery and provide advantages for patients, mental health professionals, and the VHA. Group psychotherapy aligns with the US Department of Veterans Affairs (VA) 2018 Blueprint for Excellence and 2018 to 2024 strategic plan, underscoring the need for more timely and efficient mental health services.12,13
Benefits of group psychotherapy include reductions in symptoms, decreased feelings of isolation, increased social support, decreased emotional suppression, and enhanced satisfaction with overall quality of life.14-17 Studies of veterans with PTSD have found less attrition among those who chose group therapy compared with individual therapy.14,18 Group psychotherapy improves access to care by enabling delivery to more patients.14 When compared with individual therapy, the group format allows for a large number of patients to be treated simultaneously, maximizing resources and reducing costs.3,19-21
VISN 9 CRH Innovation
The VA provides care to veterans through regionally distinct administrative systems known as Veterans Integrated Service Networks (VISNs). Clinical resource hubs (CRH) are VISN-based programs created to cover VA staffing shortages by virtually deploying HCPs into local VA systems until vacancies are filled. The national CRH vision of effectively using resources and innovative technologies to meet veterans’ health care needs, along with the above-referenced clinical gaps in the delivery of care, inspired the development of VIP Boot Camp within the VISN 9 CRH.22
Program Description
VIP Boot Camp is an evidence-informed group psychotherapy program designed to provide timely, brief, and comprehensive mental health treatment for veterans. VIP Boot Camp was developed to address the needs of veterans accessing PCMHI services who experience ≥ 1 of the often overlapping problems of anxiety/emotion regulation/stress, sleep difficulties, and chronic pain (Figure). VIP Boot Camp uses an integrative approach to highlight interconnections and similarities among these difficulties and their treatment. A primary vision of the program is to provide this comprehensive treatment within PCMHI (upstream) so additional referrals to higher levels of mental health care (downstream) may not be needed.

This design is intentional because it increases the number of individuals who can be treated upstream with comprehensive, preventive, and proactive care within PCMHI which, over time, frees up resources in the BHIP for individuals requiring higher levels of care. This approach also aligns with the importance of early treatment for chronic pain and sleep disturbances, which are linked to increased risk of suicide attempts and deaths by suicide for veterans.5 National interest for VIP Boot Camp grew during fiscal year 2024 after it received the Gold Medal Recognition for Most Adoptable and Greatest Potential for Impact during VHA National Access Sprint Wave 3—Mental Health Call of Champions.
History
VIP Boot Camp began in August 2021 at VISN 9 as a 6-week virtual group for veterans with chronic pain. It was established to assist a large VA medical center experiencing PCMHI staffing shortages and lacking available PCMHI groups. Many veterans in the chronic pain group discussed co-occurring issues such as sleep disturbances, anxiety, and stress. The CRH team considered launching 2 separate groups to address these additional PCMHI-level issues; however, in developing the group material which drew from multiple clinical approaches, the team recognized significant overlapping and interconnected themes.
The team discussed EBPs within the VHA and how certain interventions within these treatments could be helpful across many other co-occurring disorders. Integrated tactics (clinical interventions) were drawn from cognitive-behavioral therapy (for depression, insomnia, or chronic pain), acceptance and commitment therapy, prolonged exposure, cognitive processing therapy, dialectical behavior therapy, unified protocol, pain reprocessing therapy, emotional awareness and expression therapy, interpersonal neurobiology, and mindfulness. We collaborated with veterans during VIP Boot Camp groups to determine how to present and discuss complex interventions in ways that were clinically accurate, understandable, relatable, and relevant to their experiences.
To address accessibility issues, the chronic pain group was reduced to 4 weeks. A second 4-week module for anxiety, emotion regulation, and stress was developed, mirroring the tactics, language, and integrative approach of the revised chronic pain module. A similar integrative approach led to the development of the third and final 4-week module for sleep disturbances.
Current Program
The VIP Boot Camp consists of three 4-week integrated modules, each highlighting a critical area: sleep disturbances (Improving Sleep), chronic pain difficulties (Outsmarting Chronic Pain), and emotion regulation difficulties (Rewiring Your Brain). VIP Boot Camp is designed for veterans who are at the PCMHI level of care. Referrals are accepted for patients receiving treatment from primary care or PCMHI.
Guidelines for participation in VIP Boot Camp may differ across sites or VISNs. For example, a veteran who has been referred to the BHIP for medication management only or to a specialty MHC such as a pain clinic or PTSD clinic might also be appropriate and eligible for VIP Boot Camp.
Given the interconnectedness of foundational themes, elements, and practices across the VIP Boot Camp modules, the modules are offered in a rolling format with a veteran-centric “choose your own adventure” approach. Tactics are presented in the modules in a way that allows patients to begin with any 1 of the 3 modules and receive treatment that will help in the other areas. Participants choose their core module and initial treatment focus based on their values, needs, and goals. Individuals who complete a core module can end their VIP Boot Camp experience or continue to the next 4-week module for up to 3 modules.
The group is open to new individuals at the start of any 4-week module and closed for the remainder of its 4-week duration. This innovative rolling modular approach combines elements of open- and closed-group format, allowing for the flexibility and accessibility of an open group with the stability and peer support of a closed group.
Given the complicated and overlapping nature of chronic pain, emotion regulation/ stress, and sleep disturbances, VIP Boot Camp acknowledges that everything is interconnected and difficulties in 1 area may impact other areas. The 3 interconnected modules with repeating themes provide coherence and consistency. Veterans learn how interconnections across difficulties can be leveraged so that tactics learned and practiced in 1 area can assist in other areas, changing the cycle of suffering into a cycle of growth.
VIP Boot Camp sessions are 90 minutes long, once weekly for 4 weeks, with 2 mental health professionals trained to lead a dynamic group psychotherapy experience that aims to be fun for participants. VIP Boot Camp synthesizes evidence-based and evidence-informed interventions, as well as techniques from VHA complementary and integrative health programs, psychoeducation, and interpersonal interventions that model connection, playfulness, and healthy boundaries. These varied strategies combine to equip veterans with practical tactics for self-management outside of sessions, a process described as “finding puzzle pieces.” VIP Boot Camp is built on the idea that people are more likely to adopt and practice any tactic after being taught why that tactic is important, and how it fits into their larger interconnected puzzle. After each session, participants are provided with additional asynchronous educational material to help reinforce their learnings and practices.
Although individuals may hesitate to participate in a group setting, they often find the experience of community enhances and accelerates their treatment and gains. This involvement is highlighted in a core aspect of a VIP Boot Camp session called wins, during which participants learn how others on their Boot Camp team are implementing new skills and moving toward their personal values and objectives in a stepwise manner. Through these shared experiences, veterans discover how tactics working for others may serve as a model for their own personal objectives and plans for practice. The sense of relief described by many upon realizing they are not alone in their experiences, along with the satisfaction felt in discovering their ability to support others in Boot Camp, is described by many participants as deeply meaningful and in line with their personal values.
While developed as a fully virtual group program, VIP Boot Camp can also be conducted in person. The virtual program has been successful and continues to spread across VISN 9. There are 8 virtual VIP Boot Camps running in VISN 9, with plans for continued expansion. In the VISN 9 CRH, Boot Camps typically have 10 to 12 participants. Additionally, as VIP Boot Camp grows within a location there are frequently sufficient referrals to support a second rolling group, which enables staggering of the module offerings to allow for even more timely treatment.
Training Program
VISN 9 CRH also developed a VIP Boot Camp 3-day intensive training program for PCMHI HCPs that consists of learning and practicing VIP Boot Camp material for chronic pain, emotion regulation/ stress, sleep disturbances, mindfulness, and guided imagery, along with gaining experience as a VIP Boot Camp coleader. Feedback received from PCMHI HCPs who completed training has been positive. There is also a private Microsoft Teams channel for HCPs, which allows for resource sharing and community building among coleaders. More than 75 PCMHI HCPs have completed VIP Boot Camp training and > 25 VIP Boot Camps have been established at 4 additional VISNs.
The VISN 9 CRH VIP Boot Camp program initiated an implementation and effectiveness project with the Michael E. DeBakey VA Medical Center and the South Central Mental Illness Research, Education and Clinical Center. The focus of this collaboration is support for implementation and treatment effectiveness research with reports, articles, and a white paper on findings and best practices, alongside continued dissemination of the VIP Boot Camp program and training.
Conclusions
VIP Boot Camp is a PCMHI group program offering readily available, comprehensive, and integrative group psychotherapy services to veterans experiencing . 1 of the following: chronic pain, emotion regulation/ stress, and sleep disturbances. It was launched at the VISN 9 CRH with a goal of addressing clinical gaps in the delivery of mental health care, by increasing the number of patients treated within PCMHI. The VIP Boot Camp model provides veterans the opportunity to transform cycles of suffering into cycles of growth through a single approach that can address multiple presenting and interconnected issues.
A 3-day VIP Boot Camp training program provides a quick and effective path for a PCMHI program to train HCPs to launch a VIP Boot Camp. The VISN 9 CRH will continue to champion VIP Boot Camp as a model for the successful provision of comprehensive and integrative mental health treatment within PCMHI at the VA. Through readily available access to comprehensive mental health treatment in an environment that promotes participant empowerment and social engagement, VIP Boot Camp represents an integrative and innovative model of mental health treatment that offers benefits to veteran participants, HCPs, and the VHA.
Since 2007, Primary Care Mental Health Integration (PCMHI) at the Veterans Health Administration (VHA) has improved access to mental health care services for veterans by directly embedding mental health care professionals (HCPs) within primary care teams.1 Veterans referred to PCMHI often have co-occurring physical and mental health disorders.2 Untreated chronic physical and mental comorbidities can diminish the effectiveness of medical and mental health interventions. Growing evidence suggests that treatment of mental health conditions can improve physical health outcomes and management of physical conditions can improve mental health outcomes.2,3
Chronic pain and sleep disorders are common reasons patients present to primary care, and often coexist together with mental health comorbidities.4 Sleep disorders affect 50% to 88% of patients with chronic pain, and 40% of patients with sleep disorders report chronic pain.4 Research has found that chronic pain and sleep disorders increase the risk of suicide attempts and deaths by suicide. Addressing suicide prevention simultaneously with treating chronic pain and insomnia is encouraged.5
Background
PCMHI treats physical and mental health comorbidities with a collaborative framework and a biopsychosocial integrative model.6 PCMHI staff provide mental health services as members of primary care teams. An interdisciplinary PCMHI team can include, but is not limited to, psychologists, mental health social workers, psychiatrists, nurse practitioners, clinical pharmacists, and mental health nurses. Quality of care within this model is elevated, as mental and physical health are recognized as interconnected. Collaboration between primary care and mental health benefits veterans and the VHA by increasing access to mental health care, decreasing stigma associated with mental health treatment, improving health outcomes, and enhancing the likelihood of recovery, resulting in high patient satisfaction.6-8
In the existing PCMHI model, HCPs are encouraged to use short-term, evidence-based psychotherapies (EBPs).9 Veterans referred to PCMHI from primary care are typically able to attend 1 to 6 brief sessions of mental health treatment, often 20 to 30 minutes long. Most EBPs in PCMHI are disorder- specific, providing interventions focused on a single presenting problem (eg, insomnia, chronic pain, or posttraumatic stress disorder [PTSD]). For veterans with a single issue, this model can be very effective. 1,10 However, the high rate of co-occurrence of mental and physical health issues can make it difficult to fully treat interrelated problems if the focus is on 1 specific diagnosis. Veterans with a need for additional (more comprehensive or intensive) mental health treatment are frequently referred to a higher, more resource-intensive level of mental health care, either in the VHA or the community. Examples of higher levels of mental health care include the longer term behavioral health interdisciplinary program (BHIP), sometimes called a mental health clinic (MHC), or a specialty mental health program such as a PTSD clinic.
As PCMHI continues to grow, new challenges have emerged related to staffing shortages and gaps in the clinical delivery of mental health treatment within the VHA. At the same time, demand for VHA mental health treatment has increased. However, a mental health professional shortage severely limits the ability of the VHA to meet this demand. In many systems, this shortage may result in more referrals being made to a higher level of mental health care because of fewer resources to provide comprehensive treatment in a less intensive PCMHI setting.8,10,11 This referral pattern can overburden higher level care, often with long wait times for treatment and lengthy lag times between appointments. Furthermore, these gaps in the clinical delivery of care cannot be effectively addressed by hiring additional mental health professionals. This strain on resources can impede access to care and negatively affect outcomes.10
Recent congressional reports highlight these issues, noting that demand for mental health services continues to outpace the capacity of both PCMHI and higher levels of mental health care, leading to delays in treatment that may negatively affect outcomes.8,10,11 These delays can be particularly detrimental for individuals with conditions requiring timely intervention.8,11 Some veterans are willing to engage with PCMHI in a primary care setting but may be reluctant to engage in general mental health treatment. These veterans might not receive the mental health care they need without PCMHI.
Group Psychotherapy
A group psychotherapy format can address gaps in care delivery and provide advantages for patients, mental health professionals, and the VHA. Group psychotherapy aligns with the US Department of Veterans Affairs (VA) 2018 Blueprint for Excellence and 2018 to 2024 strategic plan, underscoring the need for more timely and efficient mental health services.12,13
Benefits of group psychotherapy include reductions in symptoms, decreased feelings of isolation, increased social support, decreased emotional suppression, and enhanced satisfaction with overall quality of life.14-17 Studies of veterans with PTSD have found less attrition among those who chose group therapy compared with individual therapy.14,18 Group psychotherapy improves access to care by enabling delivery to more patients.14 When compared with individual therapy, the group format allows for a large number of patients to be treated simultaneously, maximizing resources and reducing costs.3,19-21
VISN 9 CRH Innovation
The VA provides care to veterans through regionally distinct administrative systems known as Veterans Integrated Service Networks (VISNs). Clinical resource hubs (CRH) are VISN-based programs created to cover VA staffing shortages by virtually deploying HCPs into local VA systems until vacancies are filled. The national CRH vision of effectively using resources and innovative technologies to meet veterans’ health care needs, along with the above-referenced clinical gaps in the delivery of care, inspired the development of VIP Boot Camp within the VISN 9 CRH.22
Program Description
VIP Boot Camp is an evidence-informed group psychotherapy program designed to provide timely, brief, and comprehensive mental health treatment for veterans. VIP Boot Camp was developed to address the needs of veterans accessing PCMHI services who experience ≥ 1 of the often overlapping problems of anxiety/emotion regulation/stress, sleep difficulties, and chronic pain (Figure). VIP Boot Camp uses an integrative approach to highlight interconnections and similarities among these difficulties and their treatment. A primary vision of the program is to provide this comprehensive treatment within PCMHI (upstream) so additional referrals to higher levels of mental health care (downstream) may not be needed.

This design is intentional because it increases the number of individuals who can be treated upstream with comprehensive, preventive, and proactive care within PCMHI which, over time, frees up resources in the BHIP for individuals requiring higher levels of care. This approach also aligns with the importance of early treatment for chronic pain and sleep disturbances, which are linked to increased risk of suicide attempts and deaths by suicide for veterans.5 National interest for VIP Boot Camp grew during fiscal year 2024 after it received the Gold Medal Recognition for Most Adoptable and Greatest Potential for Impact during VHA National Access Sprint Wave 3—Mental Health Call of Champions.
History
VIP Boot Camp began in August 2021 at VISN 9 as a 6-week virtual group for veterans with chronic pain. It was established to assist a large VA medical center experiencing PCMHI staffing shortages and lacking available PCMHI groups. Many veterans in the chronic pain group discussed co-occurring issues such as sleep disturbances, anxiety, and stress. The CRH team considered launching 2 separate groups to address these additional PCMHI-level issues; however, in developing the group material which drew from multiple clinical approaches, the team recognized significant overlapping and interconnected themes.
The team discussed EBPs within the VHA and how certain interventions within these treatments could be helpful across many other co-occurring disorders. Integrated tactics (clinical interventions) were drawn from cognitive-behavioral therapy (for depression, insomnia, or chronic pain), acceptance and commitment therapy, prolonged exposure, cognitive processing therapy, dialectical behavior therapy, unified protocol, pain reprocessing therapy, emotional awareness and expression therapy, interpersonal neurobiology, and mindfulness. We collaborated with veterans during VIP Boot Camp groups to determine how to present and discuss complex interventions in ways that were clinically accurate, understandable, relatable, and relevant to their experiences.
To address accessibility issues, the chronic pain group was reduced to 4 weeks. A second 4-week module for anxiety, emotion regulation, and stress was developed, mirroring the tactics, language, and integrative approach of the revised chronic pain module. A similar integrative approach led to the development of the third and final 4-week module for sleep disturbances.
Current Program
The VIP Boot Camp consists of three 4-week integrated modules, each highlighting a critical area: sleep disturbances (Improving Sleep), chronic pain difficulties (Outsmarting Chronic Pain), and emotion regulation difficulties (Rewiring Your Brain). VIP Boot Camp is designed for veterans who are at the PCMHI level of care. Referrals are accepted for patients receiving treatment from primary care or PCMHI.
Guidelines for participation in VIP Boot Camp may differ across sites or VISNs. For example, a veteran who has been referred to the BHIP for medication management only or to a specialty MHC such as a pain clinic or PTSD clinic might also be appropriate and eligible for VIP Boot Camp.
Given the interconnectedness of foundational themes, elements, and practices across the VIP Boot Camp modules, the modules are offered in a rolling format with a veteran-centric “choose your own adventure” approach. Tactics are presented in the modules in a way that allows patients to begin with any 1 of the 3 modules and receive treatment that will help in the other areas. Participants choose their core module and initial treatment focus based on their values, needs, and goals. Individuals who complete a core module can end their VIP Boot Camp experience or continue to the next 4-week module for up to 3 modules.
The group is open to new individuals at the start of any 4-week module and closed for the remainder of its 4-week duration. This innovative rolling modular approach combines elements of open- and closed-group format, allowing for the flexibility and accessibility of an open group with the stability and peer support of a closed group.
Given the complicated and overlapping nature of chronic pain, emotion regulation/ stress, and sleep disturbances, VIP Boot Camp acknowledges that everything is interconnected and difficulties in 1 area may impact other areas. The 3 interconnected modules with repeating themes provide coherence and consistency. Veterans learn how interconnections across difficulties can be leveraged so that tactics learned and practiced in 1 area can assist in other areas, changing the cycle of suffering into a cycle of growth.
VIP Boot Camp sessions are 90 minutes long, once weekly for 4 weeks, with 2 mental health professionals trained to lead a dynamic group psychotherapy experience that aims to be fun for participants. VIP Boot Camp synthesizes evidence-based and evidence-informed interventions, as well as techniques from VHA complementary and integrative health programs, psychoeducation, and interpersonal interventions that model connection, playfulness, and healthy boundaries. These varied strategies combine to equip veterans with practical tactics for self-management outside of sessions, a process described as “finding puzzle pieces.” VIP Boot Camp is built on the idea that people are more likely to adopt and practice any tactic after being taught why that tactic is important, and how it fits into their larger interconnected puzzle. After each session, participants are provided with additional asynchronous educational material to help reinforce their learnings and practices.
Although individuals may hesitate to participate in a group setting, they often find the experience of community enhances and accelerates their treatment and gains. This involvement is highlighted in a core aspect of a VIP Boot Camp session called wins, during which participants learn how others on their Boot Camp team are implementing new skills and moving toward their personal values and objectives in a stepwise manner. Through these shared experiences, veterans discover how tactics working for others may serve as a model for their own personal objectives and plans for practice. The sense of relief described by many upon realizing they are not alone in their experiences, along with the satisfaction felt in discovering their ability to support others in Boot Camp, is described by many participants as deeply meaningful and in line with their personal values.
While developed as a fully virtual group program, VIP Boot Camp can also be conducted in person. The virtual program has been successful and continues to spread across VISN 9. There are 8 virtual VIP Boot Camps running in VISN 9, with plans for continued expansion. In the VISN 9 CRH, Boot Camps typically have 10 to 12 participants. Additionally, as VIP Boot Camp grows within a location there are frequently sufficient referrals to support a second rolling group, which enables staggering of the module offerings to allow for even more timely treatment.
Training Program
VISN 9 CRH also developed a VIP Boot Camp 3-day intensive training program for PCMHI HCPs that consists of learning and practicing VIP Boot Camp material for chronic pain, emotion regulation/ stress, sleep disturbances, mindfulness, and guided imagery, along with gaining experience as a VIP Boot Camp coleader. Feedback received from PCMHI HCPs who completed training has been positive. There is also a private Microsoft Teams channel for HCPs, which allows for resource sharing and community building among coleaders. More than 75 PCMHI HCPs have completed VIP Boot Camp training and > 25 VIP Boot Camps have been established at 4 additional VISNs.
The VISN 9 CRH VIP Boot Camp program initiated an implementation and effectiveness project with the Michael E. DeBakey VA Medical Center and the South Central Mental Illness Research, Education and Clinical Center. The focus of this collaboration is support for implementation and treatment effectiveness research with reports, articles, and a white paper on findings and best practices, alongside continued dissemination of the VIP Boot Camp program and training.
Conclusions
VIP Boot Camp is a PCMHI group program offering readily available, comprehensive, and integrative group psychotherapy services to veterans experiencing . 1 of the following: chronic pain, emotion regulation/ stress, and sleep disturbances. It was launched at the VISN 9 CRH with a goal of addressing clinical gaps in the delivery of mental health care, by increasing the number of patients treated within PCMHI. The VIP Boot Camp model provides veterans the opportunity to transform cycles of suffering into cycles of growth through a single approach that can address multiple presenting and interconnected issues.
A 3-day VIP Boot Camp training program provides a quick and effective path for a PCMHI program to train HCPs to launch a VIP Boot Camp. The VISN 9 CRH will continue to champion VIP Boot Camp as a model for the successful provision of comprehensive and integrative mental health treatment within PCMHI at the VA. Through readily available access to comprehensive mental health treatment in an environment that promotes participant empowerment and social engagement, VIP Boot Camp represents an integrative and innovative model of mental health treatment that offers benefits to veteran participants, HCPs, and the VHA.
- Leung LB, Yoon J, Escarce JJ, et al. Primary care-mental health integration in the VA: shifting mental health services for common mental illnesses to primary care. Psychiatr Serv. 2018;69:403-409. doi:10.1176/appi.ps.201700190
- Zhang A, Park S, Sullivan JE, et al. The effectiveness of problem-solving therapy for primary care patients’ depressive and/or anxiety disorders: a systematic review and meta-analysis. J Am Board Fam Med. 2018;31:139-150. doi:10.3122/jabfm.2018.01.170270
- Hundt NE, Barrera TL, Robinson A, et al. A systematic review of cognitive behavioral therapy for depression in veterans. Mil Med. 2014;179:942-949. doi:10.7205/milmed-d-14-00128
- Jank R, Gallee A, Boeckle M, et al. Chronic pain and sleep disorders in primary care. Pain Res Treat. 2017;2017:1-9. doi:10.1155/2017/9081802
- Ashrafioun L, Bishop TM, Pigeon WR. The relationship between pain severity, insomnia, and suicide attempts among a national veteran sample initiating pain care. Psychosom Med. 2021;83:733- 738. doi:10.1097/psy.0000000000000975
- Ramanuj P, Ferenchik E, Docherty M, et al. Evolving models of integrated behavioral health and primary care. Curr Psychiatry Rep. 2019;21:1. doi:10.1007/s11920-019-0985-4
- Post EP, Metzger M, Dumas P, et al. Integrating mental health into primary care within the Veterans Health Administration. Fam Syst Health. 2010;28:83-90. doi:10.1037/a0020130
- Smith TL, Kim B, Benzer JK, et al. FLOW: early results from a clinical demonstration project to improve the transition of patients with mental health disorders back to primary care. Psychol Serv. 2021;18:23-32. doi:10.1037/ser0000336
- Kearney LK, Post EP, Pomerantz AS, et al. Applying the interprofessional patient aligned care team in the department of veterans affairs transforming primary care. Am Psychol. 2014;69(4):399-408. doi:10.1037/a0035909
- US Government Accountability Office. Veterans health care: staffing challenges persist for fully integrating mental health and primary care services. December 15, 2022. Accessed July 9, 2025. https://www.gao.gov/products/gao-23-105372
- National Academies of Science and Engineering. Evaluation of the Department of Veterans Affairs Mental Health Services. National Academies Press; 2018. Accessed July 9, 2025. https://nap.nationalacademies.org/catalog/24915/evaluation-of-the-department-of-veterans-affairs-mental-health-services
- US Department of Veterans Affairs. Blueprint for excellence: achieving veterans’ excellence. October 6, 2014. Accessed July 9, 2025. https://www.volunteer.va.gov/docs/blueprintforexcellence_factsheet.PDF
- US Department of Veterans Affairs. Department of Veterans Affairs FY 2018-2024 strategic plan. Accessed July 9, 2025. https://www.calvet.ca.gov/Regulations/USDVA%20Strategic%20Plan%202018-2024.pdf
- Sripada RK, Bohnert KM, Ganoczy D, et al. Initial group versus individual therapy for posttraumatic stress disorder and subsequent follow-up treatment adequacy. Psychol Serv. 2016;13:349-355. doi:10.1037/ser0000077
- Burnett-Zeigler IE, Pfeiffer P, Zivin K, et al. Psychotherapy utilization for acute depression within the Veterans Affairs health care system. Psychol Serv. 2012;9:325-335. doi:10.1037/a0027957
- Kim JS, Prins A, Hirschhorn EW, et al. Preliminary investigation into the effectiveness of group webSTAIR for trauma-exposed veterans in primary care. Mil Med. 2024;189:e1403-e1408. doi:10.1093/milmed/usae052
- Jakupcak M, Blais RK, Grossbard J, et al. “Toughness” in association with mental health symptoms among Iraq and Afghanistan war veterans seeking Veterans Affairs health care. Psychol Men Masc. 2014;15:100-104. doi:10.1037/a0031508
- Stoycos SA, Berzenski SR, Beck JG, et al. Predictors of treatment completion in group psychotherapy for male veterans with posttraumatic stress disorder. J Trauma Stress. 2023;36:346-358. doi:10.1002/jts.22915
- Possemato K. The current state of intervention research for posttraumatic stress disorder within the primary care setting. J Clin Psychol Med Settings. 2011;18:268-280. doi:10.1007/s10880-011-9237-4
- Hunt MG, Rosenheck RA. Psychotherapy in mental health clinics of the Department of Veterans Affairs. J Clin Psychol. 2011;67:561-573. doi:10.1002/jclp.20788
- Khatri N, Marziali E, Tchernikov I, et al. Comparing telehealth-based and clinic-based group cognitive behavioral therapy for adults with depression and anxiety: a pilot study. Clin Interv Aging. 2014;9:765. doi:10.2147/cia.s57832
- Dangel J. Clinical resource hub increases veterans' access to care. VA News. January 12, 2025. Accessed September 3, 2025. https://news.va.gov/137439/clinical-resource-hub-increases-access-to-care/
- Leung LB, Yoon J, Escarce JJ, et al. Primary care-mental health integration in the VA: shifting mental health services for common mental illnesses to primary care. Psychiatr Serv. 2018;69:403-409. doi:10.1176/appi.ps.201700190
- Zhang A, Park S, Sullivan JE, et al. The effectiveness of problem-solving therapy for primary care patients’ depressive and/or anxiety disorders: a systematic review and meta-analysis. J Am Board Fam Med. 2018;31:139-150. doi:10.3122/jabfm.2018.01.170270
- Hundt NE, Barrera TL, Robinson A, et al. A systematic review of cognitive behavioral therapy for depression in veterans. Mil Med. 2014;179:942-949. doi:10.7205/milmed-d-14-00128
- Jank R, Gallee A, Boeckle M, et al. Chronic pain and sleep disorders in primary care. Pain Res Treat. 2017;2017:1-9. doi:10.1155/2017/9081802
- Ashrafioun L, Bishop TM, Pigeon WR. The relationship between pain severity, insomnia, and suicide attempts among a national veteran sample initiating pain care. Psychosom Med. 2021;83:733- 738. doi:10.1097/psy.0000000000000975
- Ramanuj P, Ferenchik E, Docherty M, et al. Evolving models of integrated behavioral health and primary care. Curr Psychiatry Rep. 2019;21:1. doi:10.1007/s11920-019-0985-4
- Post EP, Metzger M, Dumas P, et al. Integrating mental health into primary care within the Veterans Health Administration. Fam Syst Health. 2010;28:83-90. doi:10.1037/a0020130
- Smith TL, Kim B, Benzer JK, et al. FLOW: early results from a clinical demonstration project to improve the transition of patients with mental health disorders back to primary care. Psychol Serv. 2021;18:23-32. doi:10.1037/ser0000336
- Kearney LK, Post EP, Pomerantz AS, et al. Applying the interprofessional patient aligned care team in the department of veterans affairs transforming primary care. Am Psychol. 2014;69(4):399-408. doi:10.1037/a0035909
- US Government Accountability Office. Veterans health care: staffing challenges persist for fully integrating mental health and primary care services. December 15, 2022. Accessed July 9, 2025. https://www.gao.gov/products/gao-23-105372
- National Academies of Science and Engineering. Evaluation of the Department of Veterans Affairs Mental Health Services. National Academies Press; 2018. Accessed July 9, 2025. https://nap.nationalacademies.org/catalog/24915/evaluation-of-the-department-of-veterans-affairs-mental-health-services
- US Department of Veterans Affairs. Blueprint for excellence: achieving veterans’ excellence. October 6, 2014. Accessed July 9, 2025. https://www.volunteer.va.gov/docs/blueprintforexcellence_factsheet.PDF
- US Department of Veterans Affairs. Department of Veterans Affairs FY 2018-2024 strategic plan. Accessed July 9, 2025. https://www.calvet.ca.gov/Regulations/USDVA%20Strategic%20Plan%202018-2024.pdf
- Sripada RK, Bohnert KM, Ganoczy D, et al. Initial group versus individual therapy for posttraumatic stress disorder and subsequent follow-up treatment adequacy. Psychol Serv. 2016;13:349-355. doi:10.1037/ser0000077
- Burnett-Zeigler IE, Pfeiffer P, Zivin K, et al. Psychotherapy utilization for acute depression within the Veterans Affairs health care system. Psychol Serv. 2012;9:325-335. doi:10.1037/a0027957
- Kim JS, Prins A, Hirschhorn EW, et al. Preliminary investigation into the effectiveness of group webSTAIR for trauma-exposed veterans in primary care. Mil Med. 2024;189:e1403-e1408. doi:10.1093/milmed/usae052
- Jakupcak M, Blais RK, Grossbard J, et al. “Toughness” in association with mental health symptoms among Iraq and Afghanistan war veterans seeking Veterans Affairs health care. Psychol Men Masc. 2014;15:100-104. doi:10.1037/a0031508
- Stoycos SA, Berzenski SR, Beck JG, et al. Predictors of treatment completion in group psychotherapy for male veterans with posttraumatic stress disorder. J Trauma Stress. 2023;36:346-358. doi:10.1002/jts.22915
- Possemato K. The current state of intervention research for posttraumatic stress disorder within the primary care setting. J Clin Psychol Med Settings. 2011;18:268-280. doi:10.1007/s10880-011-9237-4
- Hunt MG, Rosenheck RA. Psychotherapy in mental health clinics of the Department of Veterans Affairs. J Clin Psychol. 2011;67:561-573. doi:10.1002/jclp.20788
- Khatri N, Marziali E, Tchernikov I, et al. Comparing telehealth-based and clinic-based group cognitive behavioral therapy for adults with depression and anxiety: a pilot study. Clin Interv Aging. 2014;9:765. doi:10.2147/cia.s57832
- Dangel J. Clinical resource hub increases veterans' access to care. VA News. January 12, 2025. Accessed September 3, 2025. https://news.va.gov/137439/clinical-resource-hub-increases-access-to-care/
VIP Boot Camp: Expanding the Impact of VA Primary Care Mental Health With a Transdiagnostic Modular Group Program
VIP Boot Camp: Expanding the Impact of VA Primary Care Mental Health With a Transdiagnostic Modular Group Program
E-Consults Bridge to Interdisciplinary Team Care for Rural Appalachian Veterans With Chronic Pain and Opioid Use Disorder
E-Consults Bridge to Interdisciplinary Team Care for Rural Appalachian Veterans With Chronic Pain and Opioid Use Disorder
Rural veterans are prescribed long-term opioid therapy for chronic pain at higher rates than urban veterans, increasing their risk of developing opioid use disorder (OUD).1,2 Veterans with co-occurring OUD and chronic pain have more severe health concerns, as well as higher rates of homelessness, psychoactive drug misuse, and mental health disorders, compared to veterans with either chronic pain or OUD alone.3 Interdisciplinary team (IDT) care is recommended for both chronic pain and OUD.4,5 Rural veterans with co-occurring chronic pain and OUD, however, are often unable to access IDTs due to long travel and wait times. As a result, these rural veterans often receive care from primary care practitioners (PCPs) who lack training in pain management and addiction and have low confidence in their ability to provide optimal treatment.6,7
In the Veterans Health Administration, electronic consultations (e-consults) provide support to PCPs by recommending evidence-based approaches such as buprenorphine for OUD and pain IDTs for chronic pain.5,8 However, research on the use of e-consults to connect to IDT care for co-occurring chronic pain and OUD are lacking, as well as studies on IDTs using innovative methods (eg, shared appointments) to overcome treatment barriers (eg, multiple appointments) for rural veterans at higher risk for co-occurring OUD and chronic pain.
This quality improvement study sought to determine the feasibility and impact of a pharmacy e-consult service that provided pain medication recommendations and subsequent referrals to RESTORE, a shared appointment program with an IDT, for assessment and treatment of chronic pain and OUD.
Methods
This retrospective chart review was approved as nonresearch by the Institutional Review Board Chair at the Salem Veterans Affairs Healthcare System (SVAHS), a low-complexity medical center in Virginia that primarily serves a rural and highly rural Central Appalachian veteran population.
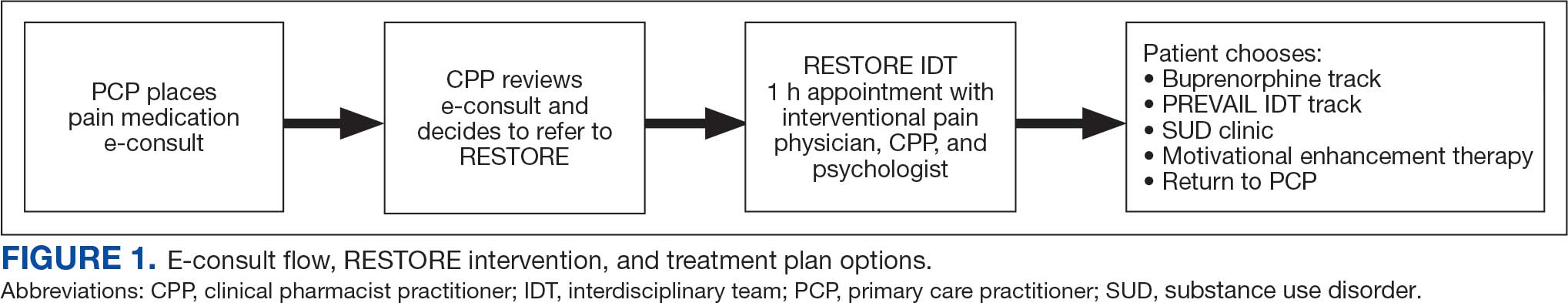
This study included veterans whose clinicians placed a pain medication e-consult requesting recommendations for medication adjustments and/or a referral to RESTORE from January 1, 2022, through January 6, 2023. Requests for services that could not be provided through an e-consult were excluded (Figure 1). Veterans who had a pain medication e-consult were identified in the SVAHS electronic medical record (EMR). Data extracted from the EMR included demographics, referral source, reason for referral, RESTORE appointment attendance, OUD diagnosis made during the RESTORE initial evaluation, implementation of medication recommendations by the referrer within 6 months, engagement in ≥ 3 pain education classes, and a shared appointment with a pain IDT within 6 months. Data were entered into a REDCap database, and descriptive statistics summarized the results. Feasibility was assessed by use of the e-consult by PCPs, attendance at the RESTORE appointment, and OUD diagnosis by the RESTORE team.
RESTORE Intervention
A pain medication e-consult was followed by referral to a shared appointment with the RESTORE IDT, with subsequent referrals to a pain IDT for chronic pain management if the veteran was amenable.
Pain medication e-consults in the EMR prompted a chart review by a clinical pharmacist practitioner (CPP). Recommendations for changes to medication regimens were documented in the EMR. At completion of the e-consult, the referring clinician received an automated view alert.
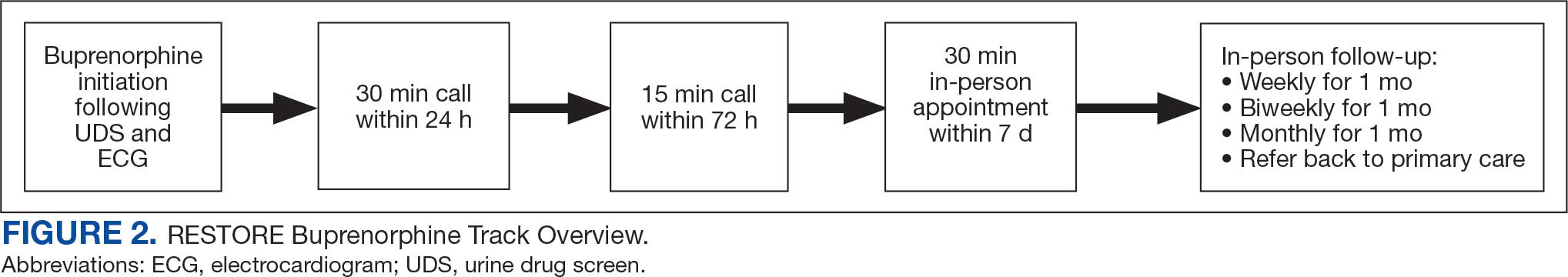
Veterans (and a support person, if preferred) were seen in a 60-minute, face-to-face shared appointment which included a psychologist, CPP, and pain physician. The psychologist conducted an OUD diagnostic interview, provided diagnostic feedback, and used motivational interviewing to provide psychoeducation on the biopsychosocial model of chronic pain, the IDT approach to chronic pain, and an overview of pain IDT care locally available. A CPP and physician then described medication options available to address OUD, if applicable. Together, the IDT and patient used shared decision making to determine a comprehensive treatment plan that may include a referral to the SVAHS PREVAIL Center for Chronic Pain IDT track (PREVAIL IDT track), a referral to substance use care in the case of polysubstance use, or medication initiation.9-11 If medication was prescribed, the patient was subsequently followed by the CPP through phone calls and face-to-face appointments at regularly scheduled intervals in coordination with the prescriber until they were stabilized. After stabilization, the prescription would be managed by their PCP (Figure 2). Veterans whose clinical condition changed significantly or worsened after returning to their PCP were invited to be reevaluated by the RESTORE team and restart care in that program. Individuals who were actively receiving RESTORE team care were discussed in a weekly care coordination meeting with all clinicians from both the PREVAIL and RESTORE teams.
Program Metrics
Pain medication e-consults were placed for 77 patients; 7 were excluded as inappropriate referral requests. Seventy (83%) e-consults were placed by PCPs (Table). Fifty-seven referring PCPs (81%) implemented ≥ 1 medication recommendation and 41 (59%) implemented all recommendations within 6 months. CPPs referred 19 individuals to RESTORE due to concerns related to high risk. All attended the initial evaluation appointment with the RESTORE team, 17 (89%) agreed to be referred to PREVAIL IDT track for nonpharmacologic pain care, and 9 (53%) engaged with that care within 6 months. Of those who attended RESTORE, 7 patients (37%) initiated buprenorphine for OUD with 6 (86%) being prescribed buprenorphine for ≥ 6 months.

Discussion
Most e-consults placed at SVAHS, which primarily serves a rural veteran population in Central Appalachia, resulted in veterans engaging in evidence-based treatment for co-occurring chronic pain and OUD. The use of e-consults and subsequent shared appointments with an IDT appears to be feasible, as the service was most often used by PCPs who often feel unequipped to manage chronic pain.7 The attendance rate for the RESTORE appointments was notable given the typically poor follow-up for patients with OUD. It supports the feasibility of a shared appointment approach which may overcome frequent barriers to care in this vulnerable population (ie, time, transportation). By attending 1 appointment with all clinicians present as opposed to multiple appointments, veterans experience fewer barriers than attending multiple appointments. RESTORE continues to be offered as an active clinical service whose implementation is now supported by changes to SVAHS policies. Since this study was conducted, the number of patients seen weekly has doubled and will soon be tripled based on high demand from PCPs.
While this study was limited to 1 site, had a small sample size, and was limited in scope, its results suggest that future research is warranted. Future studies using a larger sample size utilizing both a randomized control trial design and qualitative methods are needed to answer critical questions such as the role of patient characteristics on treatment effectiveness and the impact of the RESTORE model on long-term OUD medication adherence, patients’ perceptions and satisfaction, barriers to implementation, PCP confidence in providing pain care, and use of evidence-based nonpharmacologic pain management services.12-14
Conclusions
The results of this quality-improvement project suggest that e-consults may facilitate referrals to and patient follow-through with evidence-based treatment for co-occurring chronic pain and OUD among veterans living in rural communities in Central Appalachia who tend to experience significant barriers to traditional care and may require an innovative approach to facilitate effective treatment.
- Lund BC, Ohl ME, Hadlandsmyth K, et al. Regional and rural-urban variation in opioid prescribing in the Veterans Health Administration. Mil Med. 2019;184(11-12):894-900. doi:10.1093/milmed/usz104
- Edlund MJ, Martin BC, Russo JE, et al. The role of opioid prescription in incident opioid abuse and dependence among individuals with chronic noncancer pain: the role of opioid prescription. Clin J Pain. 2014;30(7):557-564. doi:10.1097/AJP.0000000000000021
- MacLean RR, Sofuoglu M, Stefanovics E, et al. Opioid use disorder with chronic pain increases disease burden and service use. Psychol Serv. 2023;20(1):157-165. doi:10.1037/ser0000607
- US Department of Veterans Affairs, US Department of Defense. VA/DoD clinical practice guidelines: use of opioids in the management of chronic pain. Version 4.0. Updated May 2022. Accessed August 4, 2025. https://www.healthquality.va.gov/guidelines/Pain/cot/VADoDOpioidsCPG.pdf
- US Department of Veterans Affairs, US Department of Defense. VA/DoD clinical practice guideline for the diagnosis and treatment of low back pain: the diagnosis and treatment of low back pain. Version 3.0. Updated February 2022. Accessed August 4, 2025. https://www.healthquality.va.gov/guidelines/Pain/lbp/VADoDLBPCPGFinal508.pdf
- Shipton EE, Bate F, Garrick R, et al. Systematic review of pain medicine content, teaching, and assessment in medical school curricula internationally. Pain Ther. 2018;7(2):139-161. doi:10.1007/s40122-018-0103-z
- Jamison RN, Scanlan E, Matthews ML, et al. Attitudes of primary care practitioners in managing chronic pain patients prescribed opioids for pain: a prospective longitudinal controlled trial. Pain Med. 2016;17(1):99-113. doi:10.1111/pme.12871
- Miller DM, Harvey TL. Pharmacist pain e-consults that result in a therapy change. Fed Pract. 2015;32(7):14-19.
- Courtney RE, Schadegg MJ. Chronic, noncancer pain care in the Veterans Administration: current trends and future directions. Anesthesiol Clin. 2023;41(2):519-529. doi:10.1016/j.anclin.2023.02.004
- Courtney RE, Schadegg MJ, Bolton R, et al. Using a whole health approach to build biopsychosocial-spiritual personal health plans for veterans with chronic pain. Pain Manag Nurs. 2024;25(1):69-74. doi:10.1016/j.pmn.2023.09.010
- Darnall BD, Edwards KA, Courtney RE, et al. Innovative treatment formats, technologies, and clinician trainings that improve access to behavioral pain treatment for youth and adults. Front Pain Res. 2023;4. doi:10.3389/fpain.2023.1223172
- Lister JJ, Weaver A, Ellis JD, et al. A systematic review of rural-specific barriers to medication treatment for opioid use disorder in the United States. Am J Drug Alcohol Abuse. 2020;46:273-288. doi:10.1080/00952990.2019.1694536
- Bhatraju EP, Radick AC, Leroux BG, et al. Buprenorphine adherence and illicit opioid use among patients in treatment for opioid use disorder. Am J Drug Alcohol Abuse. 2023;49. doi:10.1080/00952990.2023.2220876
- Courtney RE, Halsey E, Patil T, Mastronardi KV, Browne HS, Darnall BD. Prescription opioid tapering practices and outcomes at a rural VA health care system. Pain Med. 2024;25:480-482. doi:10.1093/pm/pnae013
Rural veterans are prescribed long-term opioid therapy for chronic pain at higher rates than urban veterans, increasing their risk of developing opioid use disorder (OUD).1,2 Veterans with co-occurring OUD and chronic pain have more severe health concerns, as well as higher rates of homelessness, psychoactive drug misuse, and mental health disorders, compared to veterans with either chronic pain or OUD alone.3 Interdisciplinary team (IDT) care is recommended for both chronic pain and OUD.4,5 Rural veterans with co-occurring chronic pain and OUD, however, are often unable to access IDTs due to long travel and wait times. As a result, these rural veterans often receive care from primary care practitioners (PCPs) who lack training in pain management and addiction and have low confidence in their ability to provide optimal treatment.6,7
In the Veterans Health Administration, electronic consultations (e-consults) provide support to PCPs by recommending evidence-based approaches such as buprenorphine for OUD and pain IDTs for chronic pain.5,8 However, research on the use of e-consults to connect to IDT care for co-occurring chronic pain and OUD are lacking, as well as studies on IDTs using innovative methods (eg, shared appointments) to overcome treatment barriers (eg, multiple appointments) for rural veterans at higher risk for co-occurring OUD and chronic pain.
This quality improvement study sought to determine the feasibility and impact of a pharmacy e-consult service that provided pain medication recommendations and subsequent referrals to RESTORE, a shared appointment program with an IDT, for assessment and treatment of chronic pain and OUD.
Methods
This retrospective chart review was approved as nonresearch by the Institutional Review Board Chair at the Salem Veterans Affairs Healthcare System (SVAHS), a low-complexity medical center in Virginia that primarily serves a rural and highly rural Central Appalachian veteran population.
This study included veterans whose clinicians placed a pain medication e-consult requesting recommendations for medication adjustments and/or a referral to RESTORE from January 1, 2022, through January 6, 2023. Requests for services that could not be provided through an e-consult were excluded (Figure 1). Veterans who had a pain medication e-consult were identified in the SVAHS electronic medical record (EMR). Data extracted from the EMR included demographics, referral source, reason for referral, RESTORE appointment attendance, OUD diagnosis made during the RESTORE initial evaluation, implementation of medication recommendations by the referrer within 6 months, engagement in ≥ 3 pain education classes, and a shared appointment with a pain IDT within 6 months. Data were entered into a REDCap database, and descriptive statistics summarized the results. Feasibility was assessed by use of the e-consult by PCPs, attendance at the RESTORE appointment, and OUD diagnosis by the RESTORE team.

RESTORE Intervention
A pain medication e-consult was followed by referral to a shared appointment with the RESTORE IDT, with subsequent referrals to a pain IDT for chronic pain management if the veteran was amenable.
Pain medication e-consults in the EMR prompted a chart review by a clinical pharmacist practitioner (CPP). Recommendations for changes to medication regimens were documented in the EMR. At completion of the e-consult, the referring clinician received an automated view alert.
Veterans (and a support person, if preferred) were seen in a 60-minute, face-to-face shared appointment which included a psychologist, CPP, and pain physician. The psychologist conducted an OUD diagnostic interview, provided diagnostic feedback, and used motivational interviewing to provide psychoeducation on the biopsychosocial model of chronic pain, the IDT approach to chronic pain, and an overview of pain IDT care locally available. A CPP and physician then described medication options available to address OUD, if applicable. Together, the IDT and patient used shared decision making to determine a comprehensive treatment plan that may include a referral to the SVAHS PREVAIL Center for Chronic Pain IDT track (PREVAIL IDT track), a referral to substance use care in the case of polysubstance use, or medication initiation.9-11 If medication was prescribed, the patient was subsequently followed by the CPP through phone calls and face-to-face appointments at regularly scheduled intervals in coordination with the prescriber until they were stabilized. After stabilization, the prescription would be managed by their PCP (Figure 2). Veterans whose clinical condition changed significantly or worsened after returning to their PCP were invited to be reevaluated by the RESTORE team and restart care in that program. Individuals who were actively receiving RESTORE team care were discussed in a weekly care coordination meeting with all clinicians from both the PREVAIL and RESTORE teams.

Program Metrics
Pain medication e-consults were placed for 77 patients; 7 were excluded as inappropriate referral requests. Seventy (83%) e-consults were placed by PCPs (Table). Fifty-seven referring PCPs (81%) implemented ≥ 1 medication recommendation and 41 (59%) implemented all recommendations within 6 months. CPPs referred 19 individuals to RESTORE due to concerns related to high risk. All attended the initial evaluation appointment with the RESTORE team, 17 (89%) agreed to be referred to PREVAIL IDT track for nonpharmacologic pain care, and 9 (53%) engaged with that care within 6 months. Of those who attended RESTORE, 7 patients (37%) initiated buprenorphine for OUD with 6 (86%) being prescribed buprenorphine for ≥ 6 months.

Discussion
Most e-consults placed at SVAHS, which primarily serves a rural veteran population in Central Appalachia, resulted in veterans engaging in evidence-based treatment for co-occurring chronic pain and OUD. The use of e-consults and subsequent shared appointments with an IDT appears to be feasible, as the service was most often used by PCPs who often feel unequipped to manage chronic pain.7 The attendance rate for the RESTORE appointments was notable given the typically poor follow-up for patients with OUD. It supports the feasibility of a shared appointment approach which may overcome frequent barriers to care in this vulnerable population (ie, time, transportation). By attending 1 appointment with all clinicians present as opposed to multiple appointments, veterans experience fewer barriers than attending multiple appointments. RESTORE continues to be offered as an active clinical service whose implementation is now supported by changes to SVAHS policies. Since this study was conducted, the number of patients seen weekly has doubled and will soon be tripled based on high demand from PCPs.
While this study was limited to 1 site, had a small sample size, and was limited in scope, its results suggest that future research is warranted. Future studies using a larger sample size utilizing both a randomized control trial design and qualitative methods are needed to answer critical questions such as the role of patient characteristics on treatment effectiveness and the impact of the RESTORE model on long-term OUD medication adherence, patients’ perceptions and satisfaction, barriers to implementation, PCP confidence in providing pain care, and use of evidence-based nonpharmacologic pain management services.12-14
Conclusions
The results of this quality-improvement project suggest that e-consults may facilitate referrals to and patient follow-through with evidence-based treatment for co-occurring chronic pain and OUD among veterans living in rural communities in Central Appalachia who tend to experience significant barriers to traditional care and may require an innovative approach to facilitate effective treatment.
Rural veterans are prescribed long-term opioid therapy for chronic pain at higher rates than urban veterans, increasing their risk of developing opioid use disorder (OUD).1,2 Veterans with co-occurring OUD and chronic pain have more severe health concerns, as well as higher rates of homelessness, psychoactive drug misuse, and mental health disorders, compared to veterans with either chronic pain or OUD alone.3 Interdisciplinary team (IDT) care is recommended for both chronic pain and OUD.4,5 Rural veterans with co-occurring chronic pain and OUD, however, are often unable to access IDTs due to long travel and wait times. As a result, these rural veterans often receive care from primary care practitioners (PCPs) who lack training in pain management and addiction and have low confidence in their ability to provide optimal treatment.6,7
In the Veterans Health Administration, electronic consultations (e-consults) provide support to PCPs by recommending evidence-based approaches such as buprenorphine for OUD and pain IDTs for chronic pain.5,8 However, research on the use of e-consults to connect to IDT care for co-occurring chronic pain and OUD are lacking, as well as studies on IDTs using innovative methods (eg, shared appointments) to overcome treatment barriers (eg, multiple appointments) for rural veterans at higher risk for co-occurring OUD and chronic pain.
This quality improvement study sought to determine the feasibility and impact of a pharmacy e-consult service that provided pain medication recommendations and subsequent referrals to RESTORE, a shared appointment program with an IDT, for assessment and treatment of chronic pain and OUD.
Methods
This retrospective chart review was approved as nonresearch by the Institutional Review Board Chair at the Salem Veterans Affairs Healthcare System (SVAHS), a low-complexity medical center in Virginia that primarily serves a rural and highly rural Central Appalachian veteran population.
This study included veterans whose clinicians placed a pain medication e-consult requesting recommendations for medication adjustments and/or a referral to RESTORE from January 1, 2022, through January 6, 2023. Requests for services that could not be provided through an e-consult were excluded (Figure 1). Veterans who had a pain medication e-consult were identified in the SVAHS electronic medical record (EMR). Data extracted from the EMR included demographics, referral source, reason for referral, RESTORE appointment attendance, OUD diagnosis made during the RESTORE initial evaluation, implementation of medication recommendations by the referrer within 6 months, engagement in ≥ 3 pain education classes, and a shared appointment with a pain IDT within 6 months. Data were entered into a REDCap database, and descriptive statistics summarized the results. Feasibility was assessed by use of the e-consult by PCPs, attendance at the RESTORE appointment, and OUD diagnosis by the RESTORE team.

RESTORE Intervention
A pain medication e-consult was followed by referral to a shared appointment with the RESTORE IDT, with subsequent referrals to a pain IDT for chronic pain management if the veteran was amenable.
Pain medication e-consults in the EMR prompted a chart review by a clinical pharmacist practitioner (CPP). Recommendations for changes to medication regimens were documented in the EMR. At completion of the e-consult, the referring clinician received an automated view alert.
Veterans (and a support person, if preferred) were seen in a 60-minute, face-to-face shared appointment which included a psychologist, CPP, and pain physician. The psychologist conducted an OUD diagnostic interview, provided diagnostic feedback, and used motivational interviewing to provide psychoeducation on the biopsychosocial model of chronic pain, the IDT approach to chronic pain, and an overview of pain IDT care locally available. A CPP and physician then described medication options available to address OUD, if applicable. Together, the IDT and patient used shared decision making to determine a comprehensive treatment plan that may include a referral to the SVAHS PREVAIL Center for Chronic Pain IDT track (PREVAIL IDT track), a referral to substance use care in the case of polysubstance use, or medication initiation.9-11 If medication was prescribed, the patient was subsequently followed by the CPP through phone calls and face-to-face appointments at regularly scheduled intervals in coordination with the prescriber until they were stabilized. After stabilization, the prescription would be managed by their PCP (Figure 2). Veterans whose clinical condition changed significantly or worsened after returning to their PCP were invited to be reevaluated by the RESTORE team and restart care in that program. Individuals who were actively receiving RESTORE team care were discussed in a weekly care coordination meeting with all clinicians from both the PREVAIL and RESTORE teams.

Program Metrics
Pain medication e-consults were placed for 77 patients; 7 were excluded as inappropriate referral requests. Seventy (83%) e-consults were placed by PCPs (Table). Fifty-seven referring PCPs (81%) implemented ≥ 1 medication recommendation and 41 (59%) implemented all recommendations within 6 months. CPPs referred 19 individuals to RESTORE due to concerns related to high risk. All attended the initial evaluation appointment with the RESTORE team, 17 (89%) agreed to be referred to PREVAIL IDT track for nonpharmacologic pain care, and 9 (53%) engaged with that care within 6 months. Of those who attended RESTORE, 7 patients (37%) initiated buprenorphine for OUD with 6 (86%) being prescribed buprenorphine for ≥ 6 months.

Discussion
Most e-consults placed at SVAHS, which primarily serves a rural veteran population in Central Appalachia, resulted in veterans engaging in evidence-based treatment for co-occurring chronic pain and OUD. The use of e-consults and subsequent shared appointments with an IDT appears to be feasible, as the service was most often used by PCPs who often feel unequipped to manage chronic pain.7 The attendance rate for the RESTORE appointments was notable given the typically poor follow-up for patients with OUD. It supports the feasibility of a shared appointment approach which may overcome frequent barriers to care in this vulnerable population (ie, time, transportation). By attending 1 appointment with all clinicians present as opposed to multiple appointments, veterans experience fewer barriers than attending multiple appointments. RESTORE continues to be offered as an active clinical service whose implementation is now supported by changes to SVAHS policies. Since this study was conducted, the number of patients seen weekly has doubled and will soon be tripled based on high demand from PCPs.
While this study was limited to 1 site, had a small sample size, and was limited in scope, its results suggest that future research is warranted. Future studies using a larger sample size utilizing both a randomized control trial design and qualitative methods are needed to answer critical questions such as the role of patient characteristics on treatment effectiveness and the impact of the RESTORE model on long-term OUD medication adherence, patients’ perceptions and satisfaction, barriers to implementation, PCP confidence in providing pain care, and use of evidence-based nonpharmacologic pain management services.12-14
Conclusions
The results of this quality-improvement project suggest that e-consults may facilitate referrals to and patient follow-through with evidence-based treatment for co-occurring chronic pain and OUD among veterans living in rural communities in Central Appalachia who tend to experience significant barriers to traditional care and may require an innovative approach to facilitate effective treatment.
- Lund BC, Ohl ME, Hadlandsmyth K, et al. Regional and rural-urban variation in opioid prescribing in the Veterans Health Administration. Mil Med. 2019;184(11-12):894-900. doi:10.1093/milmed/usz104
- Edlund MJ, Martin BC, Russo JE, et al. The role of opioid prescription in incident opioid abuse and dependence among individuals with chronic noncancer pain: the role of opioid prescription. Clin J Pain. 2014;30(7):557-564. doi:10.1097/AJP.0000000000000021
- MacLean RR, Sofuoglu M, Stefanovics E, et al. Opioid use disorder with chronic pain increases disease burden and service use. Psychol Serv. 2023;20(1):157-165. doi:10.1037/ser0000607
- US Department of Veterans Affairs, US Department of Defense. VA/DoD clinical practice guidelines: use of opioids in the management of chronic pain. Version 4.0. Updated May 2022. Accessed August 4, 2025. https://www.healthquality.va.gov/guidelines/Pain/cot/VADoDOpioidsCPG.pdf
- US Department of Veterans Affairs, US Department of Defense. VA/DoD clinical practice guideline for the diagnosis and treatment of low back pain: the diagnosis and treatment of low back pain. Version 3.0. Updated February 2022. Accessed August 4, 2025. https://www.healthquality.va.gov/guidelines/Pain/lbp/VADoDLBPCPGFinal508.pdf
- Shipton EE, Bate F, Garrick R, et al. Systematic review of pain medicine content, teaching, and assessment in medical school curricula internationally. Pain Ther. 2018;7(2):139-161. doi:10.1007/s40122-018-0103-z
- Jamison RN, Scanlan E, Matthews ML, et al. Attitudes of primary care practitioners in managing chronic pain patients prescribed opioids for pain: a prospective longitudinal controlled trial. Pain Med. 2016;17(1):99-113. doi:10.1111/pme.12871
- Miller DM, Harvey TL. Pharmacist pain e-consults that result in a therapy change. Fed Pract. 2015;32(7):14-19.
- Courtney RE, Schadegg MJ. Chronic, noncancer pain care in the Veterans Administration: current trends and future directions. Anesthesiol Clin. 2023;41(2):519-529. doi:10.1016/j.anclin.2023.02.004
- Courtney RE, Schadegg MJ, Bolton R, et al. Using a whole health approach to build biopsychosocial-spiritual personal health plans for veterans with chronic pain. Pain Manag Nurs. 2024;25(1):69-74. doi:10.1016/j.pmn.2023.09.010
- Darnall BD, Edwards KA, Courtney RE, et al. Innovative treatment formats, technologies, and clinician trainings that improve access to behavioral pain treatment for youth and adults. Front Pain Res. 2023;4. doi:10.3389/fpain.2023.1223172
- Lister JJ, Weaver A, Ellis JD, et al. A systematic review of rural-specific barriers to medication treatment for opioid use disorder in the United States. Am J Drug Alcohol Abuse. 2020;46:273-288. doi:10.1080/00952990.2019.1694536
- Bhatraju EP, Radick AC, Leroux BG, et al. Buprenorphine adherence and illicit opioid use among patients in treatment for opioid use disorder. Am J Drug Alcohol Abuse. 2023;49. doi:10.1080/00952990.2023.2220876
- Courtney RE, Halsey E, Patil T, Mastronardi KV, Browne HS, Darnall BD. Prescription opioid tapering practices and outcomes at a rural VA health care system. Pain Med. 2024;25:480-482. doi:10.1093/pm/pnae013
- Lund BC, Ohl ME, Hadlandsmyth K, et al. Regional and rural-urban variation in opioid prescribing in the Veterans Health Administration. Mil Med. 2019;184(11-12):894-900. doi:10.1093/milmed/usz104
- Edlund MJ, Martin BC, Russo JE, et al. The role of opioid prescription in incident opioid abuse and dependence among individuals with chronic noncancer pain: the role of opioid prescription. Clin J Pain. 2014;30(7):557-564. doi:10.1097/AJP.0000000000000021
- MacLean RR, Sofuoglu M, Stefanovics E, et al. Opioid use disorder with chronic pain increases disease burden and service use. Psychol Serv. 2023;20(1):157-165. doi:10.1037/ser0000607
- US Department of Veterans Affairs, US Department of Defense. VA/DoD clinical practice guidelines: use of opioids in the management of chronic pain. Version 4.0. Updated May 2022. Accessed August 4, 2025. https://www.healthquality.va.gov/guidelines/Pain/cot/VADoDOpioidsCPG.pdf
- US Department of Veterans Affairs, US Department of Defense. VA/DoD clinical practice guideline for the diagnosis and treatment of low back pain: the diagnosis and treatment of low back pain. Version 3.0. Updated February 2022. Accessed August 4, 2025. https://www.healthquality.va.gov/guidelines/Pain/lbp/VADoDLBPCPGFinal508.pdf
- Shipton EE, Bate F, Garrick R, et al. Systematic review of pain medicine content, teaching, and assessment in medical school curricula internationally. Pain Ther. 2018;7(2):139-161. doi:10.1007/s40122-018-0103-z
- Jamison RN, Scanlan E, Matthews ML, et al. Attitudes of primary care practitioners in managing chronic pain patients prescribed opioids for pain: a prospective longitudinal controlled trial. Pain Med. 2016;17(1):99-113. doi:10.1111/pme.12871
- Miller DM, Harvey TL. Pharmacist pain e-consults that result in a therapy change. Fed Pract. 2015;32(7):14-19.
- Courtney RE, Schadegg MJ. Chronic, noncancer pain care in the Veterans Administration: current trends and future directions. Anesthesiol Clin. 2023;41(2):519-529. doi:10.1016/j.anclin.2023.02.004
- Courtney RE, Schadegg MJ, Bolton R, et al. Using a whole health approach to build biopsychosocial-spiritual personal health plans for veterans with chronic pain. Pain Manag Nurs. 2024;25(1):69-74. doi:10.1016/j.pmn.2023.09.010
- Darnall BD, Edwards KA, Courtney RE, et al. Innovative treatment formats, technologies, and clinician trainings that improve access to behavioral pain treatment for youth and adults. Front Pain Res. 2023;4. doi:10.3389/fpain.2023.1223172
- Lister JJ, Weaver A, Ellis JD, et al. A systematic review of rural-specific barriers to medication treatment for opioid use disorder in the United States. Am J Drug Alcohol Abuse. 2020;46:273-288. doi:10.1080/00952990.2019.1694536
- Bhatraju EP, Radick AC, Leroux BG, et al. Buprenorphine adherence and illicit opioid use among patients in treatment for opioid use disorder. Am J Drug Alcohol Abuse. 2023;49. doi:10.1080/00952990.2023.2220876
- Courtney RE, Halsey E, Patil T, Mastronardi KV, Browne HS, Darnall BD. Prescription opioid tapering practices and outcomes at a rural VA health care system. Pain Med. 2024;25:480-482. doi:10.1093/pm/pnae013
E-Consults Bridge to Interdisciplinary Team Care for Rural Appalachian Veterans With Chronic Pain and Opioid Use Disorder
E-Consults Bridge to Interdisciplinary Team Care for Rural Appalachian Veterans With Chronic Pain and Opioid Use Disorder
Empowering Culture Change and Safety on the Journey to Zero Harm With Huddle Cards
Empowering Culture Change and Safety on the Journey to Zero Harm With Huddle Cards
Safety event reporting plays a vital role in fostering a culture of safety within a health care organization. The US Department of Veterans Affairs (VA) has shifted its focus from eradicating medical errors to minimizing or eliminating harm to patients.1 The National Center for Patient Safety’s objective is to prevent recurring errors by identifying and addressing systemic problems that may have been overlooked.2
Taking inspiration from industries known for high reliability, such as aviation and nuclear power, the Veterans Health Administration (VHA) patient safety program aims to identify and eliminate system vulnerabilities, such as medical errors. Learning from near misses, which occur more frequently than actual adverse events, is a crucial part of this process.3 By addressing these issues, the VHA can establish safer systems and encourage continuous identification of potential problems with proactive resolution.
All staff should participate actively in event reporting, which involves documenting and communicating details, outcomes, and relevant data about an event to understand what occurred, evaluate success, identify areas for improvement, and inform future decisions. This helps identify system weaknesses, create opportunities to standardize procedures and enhance patient care.
At the high complexity Central Texas Veterans Health Care System (CTVHCS), the fiscal year (FY) 2023 All Employee Survey (AES) found that staff members require additional education and awareness regarding the reporting of patient safety concerns.4 The survey highlighted areas such as lack of education on reporting, doubts about the effectiveness of reporting, confusion about the process after a report is made, and insufficient feedback.
BACKGROUND
To improve the culture of safety and address deficiencies noted in the AES, the CTVHCS patient safety (PS) and high reliability organization (HRO) teams partnered to develop a quality improvement initiative to increase staff understanding of safety event reporting and strengthen the safety culture. The PS and HRO teams developed an innovative education model that integrates Joint Patient Safety Reporting System (JPSR) education into huddles.
This initiative, called the JPSR Huddle Card Toolkit, sought to assess the impact of the toolkit on staff knowledge and behaviors related to patient safety event reporting. The toolkit consisted of educational materials encompassing 6 key areas: (1) reporting incidents; (2) close calls and near misses; (3) identification of root causes; (4) understanding the life cycle of a JPSR; (5) celebrating achievements; and (6) distinguishing between facts and fiction. Each JPSR huddle card included discussion points for the facilitator and was formatted on a 5 × 7-inch card (Figure 1). Topics were addressed during weekly safety huddles conducted in the pilot unit over a 6-week period. To evaluate its effectiveness, a pilot unit was selected and distributed an anonymous questionnaire paired with the JPSR huddle card toolkit to measure staff responses.

The pilot was conducted from November 2023 to January 2024. The participating pilot unit was a 10-bed critical care unit with 42 full-time employees. Nursing leadership, quality safety, and value personnel, and the Veterans Integrated Services Network (VISN) PS Team reviewed and approved the pilot.
Reporting of adverse events and near misses provides an opportunity to learn about latent systems errors.2 In 2018, the VHA began using the JPSR to standardize the capture and data management on medical errors and close calls across the Defense Health Administration (DHA) and VHA.1 The JPSR software is a joint application of the VHA and DHA. It improves the identification and documentation of patient safety-related events for VA medical centers, military hospitals and clinics, active-duty personnel, veterans and their families.
Event reporting is a key element in advancing high reliability and achieving zero preventable harm.1 Teams use these data to identify organizational patient safety trends and preempt common safety issues. All data are protected under 38 USC §5705 and 10 USC §1102.5 The JPSR single-source system standardizes the collection of core data points and increases collaboration between the DHA and VHA. This partnership increases insight into safety-related incidents, allowing for earlier detection and prevention of patient harm or injury incidents.
Numerous studies consistently commend huddles for their effectiveness in promoting teamwork and their positive impact on patient safety.6-8 Huddles facilitate connections between employees who may not typically interact, provide opportunities for discussions, and serve as a platform to encourage employees to voice their opinions. By fostering these interactions, huddles empower employees and create an environment for shared understanding, building trust, and promoting continuous learning.8
OBSERVATIONS
The JPSR huddle card initiative aimed to improve understanding of the JPSR process and promote knowledge and attitudes about patient safety and event reporting, while emphasizing shared responsibility. The goals focused on effective communication, respect for expertise, awareness of operational nuances, voicing concerns, and ensuring zero harm.
The facilitator initiated huddles by announcing their start to cultivate a constructive outcome.8 The JPSR huddle cards used a structured format designed to foster engagement and understanding of the topic. Each card begins with a factual statement or an open-ended question to gauge participants’ awareness or understanding. It then provides essential facts, principles, and relevant information to deepen knowledge. The card concludes with a discussion question, allowing facilitators to assess shared learning and encourage group reflection. This format promotes active participation and ensures that key concepts are both introduced and reinforced through dialogue.
The PS team standardized the format for all huddle cards, allowing 5 to 10 minutes for discussing training materials, receiving feedback, and concluding with a discussion question and call to action. Prior to each huddle, the facilitator would read a scripted remark that reviewed the objectives and ground rules for an effective huddle.
The PS and HRO teams promoted interactive discussions and welcomed ongoing feedback. Huddles provided a psychologically safe environment where individuals were encouraged to voice their thoughts and ideas.
Each weekly huddle card addressed a different patient safety topic. The Week 1 huddle card focuses on event reporting for safety improvement. The card outlines the purpose of JPSR as a tool to identify, manage, and analyze safety events to reduce preventable harm. The card emphasizes 3 core principles: (1) acknowledging mistakes, recognizing that errors happen; (2) no blame, no shame (encouraging a no-blame just culture to raise concerns); and (3) continuous improvement (committing to ongoing learning and prevention). It provides guidance on event details entry, advising staff to include facts in an SBAR (Situation, Background, Assessment, Response) format, avoid assumptions, and exclude personal identifiers. Tips include entering only relevant facts to help reviewers understand the incident. The card ends with discussion questions on reporting barriers and potential improvements in event reporting practices.
The Week 2 huddle card focuses on understanding and reporting near miss events, also known as close calls or good catches. A near miss is an incident where a potential hazard was identified and prevented before it reached the patient, avoiding harm due to timely intervention. The card emphasizes the importance of identifying these events to understand weaknesses and proactively reduce risks. Examples of near misses include discovering expired medication before use, catching a potential wrong-site surgery, and noticing incorrect medication dosages. Staff are encouraged to develop a mindset for anticipating and solving risks. The card ends with a discussion asking participants to share examples of near misses in their area.
The Week 3 huddle card covers root causes in preventing errors. The card highlights that errors in health care often stem from flawed processes rather than individual faults. By identifying root causes, systemic weaknesses can be addressed to reduce mistakes and build more error-tolerant and robust systems. All staff are advised to adopt a mindset of continuous improvement, error trapping behaviors and problem-solving. It concludes with discussion questions prompting reflection on assumptions and identifying weaknesses when something goes wrong.
The Week 4 huddle card covers the life of a JPSR, detailing that after entry JPSR events are viewed by the highest leadership levels at the morning report, and that lessons learned are distributed through frontline managers and chiefs in a monthly report to be shared with frontline staff. Additionally, JPSR trends are shared during monthly HRO safety forums. These practices promote a culture of safety through open communication and problem-solving. Staff and leaders are encouraged to prioritize safety daily. Discussion prompts ask team members if they had seen positive changes from JPSR reporting and what they would like leadership to communicate after investigations.
The Week 5 huddle card covers celebrating safety event reporting called Cue the Confetti. The VHA emphasizes recognizing staff who report safety events as part of their commitment to zero harm. By celebrating these contributions, the VHA fosters respect, joy, and satisfaction in the work. Staff are encouraged to nominate colleagues for recognition, reinforcing a supportive environment. Prompts invite teams to discuss how they celebrate JPSR reporting and how they’d like to enhance this culture of appreciation.
The Week 6 huddle card covers common misconceptions about JPSR. Key facts include that JPSRs are confidential, not for disciplinary action, and can be submitted by any staff member at any time. Only PS can view reporter identities for clarification purposes. The card concludes with prompts to ensure staff know how to access JPSR support and resources.
Measuring the impact on staff was essential to assess effectiveness and gather data for program improvement. To evaluate the impact of the huddle cards on the staff, the team provided a voluntary and anonymous 9 question survey (Figure 2). The survey was completed before the pilot began and again at the end of Week 6.
Questions 1 through 5 and 7 through 9 pertained to participants’ perceived knowledge and understanding of aspects of the JPSR. Perceived improvement among intensive care unit (ICU) participants ranged from 15% to 53%. There was a positive increase associated with every question with the top improvements: question 8, “How do you rate your understanding of how we share safety events for system and process improvement?” (53.4% increase); question 5, “How do you rate your understanding of what happens to a JPSR after it is entered?” (51.9% increase), and question 9, “How do you rate your understanding of the concepts of trust, psychological safety and a just culture?” (47.8% increase).
The survey analysis was not able to track individual changes. As a result, the findings reflect an overall change for the entire study group. Moreover, the questions assessed participants’ perceived knowledge rather than actual knowledge gained. It is important to note that there may be a significant gap between the actual knowledge gained and how participants perceive it. Additionally, improvement in knowledge and comprehension does not necessarily translate into behavior changes.
CONCLUSIONS
The use of JPSR huddle cards and direct engagement with staff during safety huddles yielded positive outcomes. On average, participants demonstrated higher scores in posttest questions compared to pretest questions. The posttest scores were consistently higher than the pretest scores, showing an average increase of around 2 standard deviations across all questions. This indicates an improvement in participants’ perceived knowledge and comprehension of the JPSR material.
During the pilot implementation of the huddle cards, there was a notable improvement in team member engagement. The structured format of the cards facilitated focused and meaningful discussions during safety huddles, encouraging open dialogue and fostering a culture of safety. Team members actively participated in identifying potential risks, sharing observations, and proposing actionable solutions, which reflected an enhanced sense of ownership regarding safety practices.
The support dialogue facilitated by the huddle cards highlighted the significance of mutual accountability and a collective commitment to achieving zero harm. This collaborative environment strengthened trust among team members and underscored the importance of shared vigilance in preventing adverse events. The pilot demonstrated the potential of huddle cards as an essential tool for enhancing team-based safety initiatives and promoting a culture of high reliability within the organization.
The total number of JPSR events in the ICU rose from 156 in FY 23 to 170 in FY 24. Adverse events increased from 19 to 31, while close calls saw a slight uptick from 137 to 139. Despite the overall rise in adverse events, a detailed analysis indicated that incidents of moderate harm decreased from 4 in FY 23 to 2 in FY 24. Furthermore, there was 1 reported case of death or severe harm in FY 23, which decreased to 0 in FY 24. This trend is consistent with the overarching objective of a high-reliability organization to achieve zero harm.
The next step is to expand this initiative across CTVHCS. This initiative aims to make this an annual education for all areas. The JPSR huddle card toolkit will be formatted by the media department for easy printing and retrieval. Leaders within units, clinics, and services will be empowered to facilitate the sessions in their safety huddles and reap the same outcomes as in the pilot. CTVHCS PS will monitor the effectiveness of this through ongoing CTVHCS patient safety rounding and future AES.
- Essen K, Villalobos C, Sculli GL, Steinbach L. Establishing a just culture: implications for the Veterans Health Administration journey to high reliability. Fed Pract. 2024;41:290-297. doi:10.12788/fp.0512
- Louis MY, Hussain LR, Dhanraj DN, et al. Improving patient safety event reporting among residents and teaching faculty. Ochsner J. 2016;16:73-80.
- Pimental CB, Snow AL, Carnes SL, et al. Huddles and their effectiveness at the frontlines of clinical care: a scoping review. J Gen Intern Med. 2021;36:2772-2783. doi:10.1007/s11606-021-06632-9
- National Academies of Sciences, Engineering, and Medicine. Appendix C: Nature of Veterans Health Administration Facilities Management (Engineering) Tasks and Staffing. Facilities Staffing Requirements for the Veterans Health Administration-Resource Planning and Methodology for the Future. National Academies Press. 2020:105-116. Accessed August 11, 2025. https://nap.nationalacademies.org/read/25454/chapter/11
- Woodier N, Burnett C, Moppett I. The value of learning from near misses to improve patient safety: a scoping review. J Patient Saf. 2023;19:42-47. doi:10.1097/pts.0000000000001078
- Ismail A, Khalid SNM. Patient safety culture and its determinants among healthcare professionals at a cluster hospital in Malaysia: a cross-sectional study. BMJ Open. 2022;12:e060546. doi:10.1136/bmjopen-2021-060546
- Ngo J, Lau D, Ploquin J, Receveur T, Stassen K, Del Castilho C. Improving incident reporting among physicians at south health campus hospital. BMJ Open Qual. 2022;11:e001945. doi:10.1136/bmjoq-2022-001945
- Oweidat I, Al-Mugheed K, Alsenany SA, et al. Awareness of reporting practices and barriers to incident reporting among nurses. BMC Nurs. 2023;22:231. doi:10.1186/s12912-023-01376-9
Safety event reporting plays a vital role in fostering a culture of safety within a health care organization. The US Department of Veterans Affairs (VA) has shifted its focus from eradicating medical errors to minimizing or eliminating harm to patients.1 The National Center for Patient Safety’s objective is to prevent recurring errors by identifying and addressing systemic problems that may have been overlooked.2
Taking inspiration from industries known for high reliability, such as aviation and nuclear power, the Veterans Health Administration (VHA) patient safety program aims to identify and eliminate system vulnerabilities, such as medical errors. Learning from near misses, which occur more frequently than actual adverse events, is a crucial part of this process.3 By addressing these issues, the VHA can establish safer systems and encourage continuous identification of potential problems with proactive resolution.
All staff should participate actively in event reporting, which involves documenting and communicating details, outcomes, and relevant data about an event to understand what occurred, evaluate success, identify areas for improvement, and inform future decisions. This helps identify system weaknesses, create opportunities to standardize procedures and enhance patient care.
At the high complexity Central Texas Veterans Health Care System (CTVHCS), the fiscal year (FY) 2023 All Employee Survey (AES) found that staff members require additional education and awareness regarding the reporting of patient safety concerns.4 The survey highlighted areas such as lack of education on reporting, doubts about the effectiveness of reporting, confusion about the process after a report is made, and insufficient feedback.
BACKGROUND
To improve the culture of safety and address deficiencies noted in the AES, the CTVHCS patient safety (PS) and high reliability organization (HRO) teams partnered to develop a quality improvement initiative to increase staff understanding of safety event reporting and strengthen the safety culture. The PS and HRO teams developed an innovative education model that integrates Joint Patient Safety Reporting System (JPSR) education into huddles.
This initiative, called the JPSR Huddle Card Toolkit, sought to assess the impact of the toolkit on staff knowledge and behaviors related to patient safety event reporting. The toolkit consisted of educational materials encompassing 6 key areas: (1) reporting incidents; (2) close calls and near misses; (3) identification of root causes; (4) understanding the life cycle of a JPSR; (5) celebrating achievements; and (6) distinguishing between facts and fiction. Each JPSR huddle card included discussion points for the facilitator and was formatted on a 5 × 7-inch card (Figure 1). Topics were addressed during weekly safety huddles conducted in the pilot unit over a 6-week period. To evaluate its effectiveness, a pilot unit was selected and distributed an anonymous questionnaire paired with the JPSR huddle card toolkit to measure staff responses.

The pilot was conducted from November 2023 to January 2024. The participating pilot unit was a 10-bed critical care unit with 42 full-time employees. Nursing leadership, quality safety, and value personnel, and the Veterans Integrated Services Network (VISN) PS Team reviewed and approved the pilot.
Reporting of adverse events and near misses provides an opportunity to learn about latent systems errors.2 In 2018, the VHA began using the JPSR to standardize the capture and data management on medical errors and close calls across the Defense Health Administration (DHA) and VHA.1 The JPSR software is a joint application of the VHA and DHA. It improves the identification and documentation of patient safety-related events for VA medical centers, military hospitals and clinics, active-duty personnel, veterans and their families.
Event reporting is a key element in advancing high reliability and achieving zero preventable harm.1 Teams use these data to identify organizational patient safety trends and preempt common safety issues. All data are protected under 38 USC §5705 and 10 USC §1102.5 The JPSR single-source system standardizes the collection of core data points and increases collaboration between the DHA and VHA. This partnership increases insight into safety-related incidents, allowing for earlier detection and prevention of patient harm or injury incidents.
Numerous studies consistently commend huddles for their effectiveness in promoting teamwork and their positive impact on patient safety.6-8 Huddles facilitate connections between employees who may not typically interact, provide opportunities for discussions, and serve as a platform to encourage employees to voice their opinions. By fostering these interactions, huddles empower employees and create an environment for shared understanding, building trust, and promoting continuous learning.8
OBSERVATIONS
The JPSR huddle card initiative aimed to improve understanding of the JPSR process and promote knowledge and attitudes about patient safety and event reporting, while emphasizing shared responsibility. The goals focused on effective communication, respect for expertise, awareness of operational nuances, voicing concerns, and ensuring zero harm.
The facilitator initiated huddles by announcing their start to cultivate a constructive outcome.8 The JPSR huddle cards used a structured format designed to foster engagement and understanding of the topic. Each card begins with a factual statement or an open-ended question to gauge participants’ awareness or understanding. It then provides essential facts, principles, and relevant information to deepen knowledge. The card concludes with a discussion question, allowing facilitators to assess shared learning and encourage group reflection. This format promotes active participation and ensures that key concepts are both introduced and reinforced through dialogue.
The PS team standardized the format for all huddle cards, allowing 5 to 10 minutes for discussing training materials, receiving feedback, and concluding with a discussion question and call to action. Prior to each huddle, the facilitator would read a scripted remark that reviewed the objectives and ground rules for an effective huddle.
The PS and HRO teams promoted interactive discussions and welcomed ongoing feedback. Huddles provided a psychologically safe environment where individuals were encouraged to voice their thoughts and ideas.
Each weekly huddle card addressed a different patient safety topic. The Week 1 huddle card focuses on event reporting for safety improvement. The card outlines the purpose of JPSR as a tool to identify, manage, and analyze safety events to reduce preventable harm. The card emphasizes 3 core principles: (1) acknowledging mistakes, recognizing that errors happen; (2) no blame, no shame (encouraging a no-blame just culture to raise concerns); and (3) continuous improvement (committing to ongoing learning and prevention). It provides guidance on event details entry, advising staff to include facts in an SBAR (Situation, Background, Assessment, Response) format, avoid assumptions, and exclude personal identifiers. Tips include entering only relevant facts to help reviewers understand the incident. The card ends with discussion questions on reporting barriers and potential improvements in event reporting practices.
The Week 2 huddle card focuses on understanding and reporting near miss events, also known as close calls or good catches. A near miss is an incident where a potential hazard was identified and prevented before it reached the patient, avoiding harm due to timely intervention. The card emphasizes the importance of identifying these events to understand weaknesses and proactively reduce risks. Examples of near misses include discovering expired medication before use, catching a potential wrong-site surgery, and noticing incorrect medication dosages. Staff are encouraged to develop a mindset for anticipating and solving risks. The card ends with a discussion asking participants to share examples of near misses in their area.
The Week 3 huddle card covers root causes in preventing errors. The card highlights that errors in health care often stem from flawed processes rather than individual faults. By identifying root causes, systemic weaknesses can be addressed to reduce mistakes and build more error-tolerant and robust systems. All staff are advised to adopt a mindset of continuous improvement, error trapping behaviors and problem-solving. It concludes with discussion questions prompting reflection on assumptions and identifying weaknesses when something goes wrong.
The Week 4 huddle card covers the life of a JPSR, detailing that after entry JPSR events are viewed by the highest leadership levels at the morning report, and that lessons learned are distributed through frontline managers and chiefs in a monthly report to be shared with frontline staff. Additionally, JPSR trends are shared during monthly HRO safety forums. These practices promote a culture of safety through open communication and problem-solving. Staff and leaders are encouraged to prioritize safety daily. Discussion prompts ask team members if they had seen positive changes from JPSR reporting and what they would like leadership to communicate after investigations.
The Week 5 huddle card covers celebrating safety event reporting called Cue the Confetti. The VHA emphasizes recognizing staff who report safety events as part of their commitment to zero harm. By celebrating these contributions, the VHA fosters respect, joy, and satisfaction in the work. Staff are encouraged to nominate colleagues for recognition, reinforcing a supportive environment. Prompts invite teams to discuss how they celebrate JPSR reporting and how they’d like to enhance this culture of appreciation.
The Week 6 huddle card covers common misconceptions about JPSR. Key facts include that JPSRs are confidential, not for disciplinary action, and can be submitted by any staff member at any time. Only PS can view reporter identities for clarification purposes. The card concludes with prompts to ensure staff know how to access JPSR support and resources.
Measuring the impact on staff was essential to assess effectiveness and gather data for program improvement. To evaluate the impact of the huddle cards on the staff, the team provided a voluntary and anonymous 9 question survey (Figure 2). The survey was completed before the pilot began and again at the end of Week 6.

Questions 1 through 5 and 7 through 9 pertained to participants’ perceived knowledge and understanding of aspects of the JPSR. Perceived improvement among intensive care unit (ICU) participants ranged from 15% to 53%. There was a positive increase associated with every question with the top improvements: question 8, “How do you rate your understanding of how we share safety events for system and process improvement?” (53.4% increase); question 5, “How do you rate your understanding of what happens to a JPSR after it is entered?” (51.9% increase), and question 9, “How do you rate your understanding of the concepts of trust, psychological safety and a just culture?” (47.8% increase).
The survey analysis was not able to track individual changes. As a result, the findings reflect an overall change for the entire study group. Moreover, the questions assessed participants’ perceived knowledge rather than actual knowledge gained. It is important to note that there may be a significant gap between the actual knowledge gained and how participants perceive it. Additionally, improvement in knowledge and comprehension does not necessarily translate into behavior changes.
CONCLUSIONS
The use of JPSR huddle cards and direct engagement with staff during safety huddles yielded positive outcomes. On average, participants demonstrated higher scores in posttest questions compared to pretest questions. The posttest scores were consistently higher than the pretest scores, showing an average increase of around 2 standard deviations across all questions. This indicates an improvement in participants’ perceived knowledge and comprehension of the JPSR material.
During the pilot implementation of the huddle cards, there was a notable improvement in team member engagement. The structured format of the cards facilitated focused and meaningful discussions during safety huddles, encouraging open dialogue and fostering a culture of safety. Team members actively participated in identifying potential risks, sharing observations, and proposing actionable solutions, which reflected an enhanced sense of ownership regarding safety practices.
The support dialogue facilitated by the huddle cards highlighted the significance of mutual accountability and a collective commitment to achieving zero harm. This collaborative environment strengthened trust among team members and underscored the importance of shared vigilance in preventing adverse events. The pilot demonstrated the potential of huddle cards as an essential tool for enhancing team-based safety initiatives and promoting a culture of high reliability within the organization.
The total number of JPSR events in the ICU rose from 156 in FY 23 to 170 in FY 24. Adverse events increased from 19 to 31, while close calls saw a slight uptick from 137 to 139. Despite the overall rise in adverse events, a detailed analysis indicated that incidents of moderate harm decreased from 4 in FY 23 to 2 in FY 24. Furthermore, there was 1 reported case of death or severe harm in FY 23, which decreased to 0 in FY 24. This trend is consistent with the overarching objective of a high-reliability organization to achieve zero harm.
The next step is to expand this initiative across CTVHCS. This initiative aims to make this an annual education for all areas. The JPSR huddle card toolkit will be formatted by the media department for easy printing and retrieval. Leaders within units, clinics, and services will be empowered to facilitate the sessions in their safety huddles and reap the same outcomes as in the pilot. CTVHCS PS will monitor the effectiveness of this through ongoing CTVHCS patient safety rounding and future AES.
Safety event reporting plays a vital role in fostering a culture of safety within a health care organization. The US Department of Veterans Affairs (VA) has shifted its focus from eradicating medical errors to minimizing or eliminating harm to patients.1 The National Center for Patient Safety’s objective is to prevent recurring errors by identifying and addressing systemic problems that may have been overlooked.2
Taking inspiration from industries known for high reliability, such as aviation and nuclear power, the Veterans Health Administration (VHA) patient safety program aims to identify and eliminate system vulnerabilities, such as medical errors. Learning from near misses, which occur more frequently than actual adverse events, is a crucial part of this process.3 By addressing these issues, the VHA can establish safer systems and encourage continuous identification of potential problems with proactive resolution.
All staff should participate actively in event reporting, which involves documenting and communicating details, outcomes, and relevant data about an event to understand what occurred, evaluate success, identify areas for improvement, and inform future decisions. This helps identify system weaknesses, create opportunities to standardize procedures and enhance patient care.
At the high complexity Central Texas Veterans Health Care System (CTVHCS), the fiscal year (FY) 2023 All Employee Survey (AES) found that staff members require additional education and awareness regarding the reporting of patient safety concerns.4 The survey highlighted areas such as lack of education on reporting, doubts about the effectiveness of reporting, confusion about the process after a report is made, and insufficient feedback.
BACKGROUND
To improve the culture of safety and address deficiencies noted in the AES, the CTVHCS patient safety (PS) and high reliability organization (HRO) teams partnered to develop a quality improvement initiative to increase staff understanding of safety event reporting and strengthen the safety culture. The PS and HRO teams developed an innovative education model that integrates Joint Patient Safety Reporting System (JPSR) education into huddles.
This initiative, called the JPSR Huddle Card Toolkit, sought to assess the impact of the toolkit on staff knowledge and behaviors related to patient safety event reporting. The toolkit consisted of educational materials encompassing 6 key areas: (1) reporting incidents; (2) close calls and near misses; (3) identification of root causes; (4) understanding the life cycle of a JPSR; (5) celebrating achievements; and (6) distinguishing between facts and fiction. Each JPSR huddle card included discussion points for the facilitator and was formatted on a 5 × 7-inch card (Figure 1). Topics were addressed during weekly safety huddles conducted in the pilot unit over a 6-week period. To evaluate its effectiveness, a pilot unit was selected and distributed an anonymous questionnaire paired with the JPSR huddle card toolkit to measure staff responses.

The pilot was conducted from November 2023 to January 2024. The participating pilot unit was a 10-bed critical care unit with 42 full-time employees. Nursing leadership, quality safety, and value personnel, and the Veterans Integrated Services Network (VISN) PS Team reviewed and approved the pilot.
Reporting of adverse events and near misses provides an opportunity to learn about latent systems errors.2 In 2018, the VHA began using the JPSR to standardize the capture and data management on medical errors and close calls across the Defense Health Administration (DHA) and VHA.1 The JPSR software is a joint application of the VHA and DHA. It improves the identification and documentation of patient safety-related events for VA medical centers, military hospitals and clinics, active-duty personnel, veterans and their families.
Event reporting is a key element in advancing high reliability and achieving zero preventable harm.1 Teams use these data to identify organizational patient safety trends and preempt common safety issues. All data are protected under 38 USC §5705 and 10 USC §1102.5 The JPSR single-source system standardizes the collection of core data points and increases collaboration between the DHA and VHA. This partnership increases insight into safety-related incidents, allowing for earlier detection and prevention of patient harm or injury incidents.
Numerous studies consistently commend huddles for their effectiveness in promoting teamwork and their positive impact on patient safety.6-8 Huddles facilitate connections between employees who may not typically interact, provide opportunities for discussions, and serve as a platform to encourage employees to voice their opinions. By fostering these interactions, huddles empower employees and create an environment for shared understanding, building trust, and promoting continuous learning.8
OBSERVATIONS
The JPSR huddle card initiative aimed to improve understanding of the JPSR process and promote knowledge and attitudes about patient safety and event reporting, while emphasizing shared responsibility. The goals focused on effective communication, respect for expertise, awareness of operational nuances, voicing concerns, and ensuring zero harm.
The facilitator initiated huddles by announcing their start to cultivate a constructive outcome.8 The JPSR huddle cards used a structured format designed to foster engagement and understanding of the topic. Each card begins with a factual statement or an open-ended question to gauge participants’ awareness or understanding. It then provides essential facts, principles, and relevant information to deepen knowledge. The card concludes with a discussion question, allowing facilitators to assess shared learning and encourage group reflection. This format promotes active participation and ensures that key concepts are both introduced and reinforced through dialogue.
The PS team standardized the format for all huddle cards, allowing 5 to 10 minutes for discussing training materials, receiving feedback, and concluding with a discussion question and call to action. Prior to each huddle, the facilitator would read a scripted remark that reviewed the objectives and ground rules for an effective huddle.
The PS and HRO teams promoted interactive discussions and welcomed ongoing feedback. Huddles provided a psychologically safe environment where individuals were encouraged to voice their thoughts and ideas.
Each weekly huddle card addressed a different patient safety topic. The Week 1 huddle card focuses on event reporting for safety improvement. The card outlines the purpose of JPSR as a tool to identify, manage, and analyze safety events to reduce preventable harm. The card emphasizes 3 core principles: (1) acknowledging mistakes, recognizing that errors happen; (2) no blame, no shame (encouraging a no-blame just culture to raise concerns); and (3) continuous improvement (committing to ongoing learning and prevention). It provides guidance on event details entry, advising staff to include facts in an SBAR (Situation, Background, Assessment, Response) format, avoid assumptions, and exclude personal identifiers. Tips include entering only relevant facts to help reviewers understand the incident. The card ends with discussion questions on reporting barriers and potential improvements in event reporting practices.
The Week 2 huddle card focuses on understanding and reporting near miss events, also known as close calls or good catches. A near miss is an incident where a potential hazard was identified and prevented before it reached the patient, avoiding harm due to timely intervention. The card emphasizes the importance of identifying these events to understand weaknesses and proactively reduce risks. Examples of near misses include discovering expired medication before use, catching a potential wrong-site surgery, and noticing incorrect medication dosages. Staff are encouraged to develop a mindset for anticipating and solving risks. The card ends with a discussion asking participants to share examples of near misses in their area.
The Week 3 huddle card covers root causes in preventing errors. The card highlights that errors in health care often stem from flawed processes rather than individual faults. By identifying root causes, systemic weaknesses can be addressed to reduce mistakes and build more error-tolerant and robust systems. All staff are advised to adopt a mindset of continuous improvement, error trapping behaviors and problem-solving. It concludes with discussion questions prompting reflection on assumptions and identifying weaknesses when something goes wrong.
The Week 4 huddle card covers the life of a JPSR, detailing that after entry JPSR events are viewed by the highest leadership levels at the morning report, and that lessons learned are distributed through frontline managers and chiefs in a monthly report to be shared with frontline staff. Additionally, JPSR trends are shared during monthly HRO safety forums. These practices promote a culture of safety through open communication and problem-solving. Staff and leaders are encouraged to prioritize safety daily. Discussion prompts ask team members if they had seen positive changes from JPSR reporting and what they would like leadership to communicate after investigations.
The Week 5 huddle card covers celebrating safety event reporting called Cue the Confetti. The VHA emphasizes recognizing staff who report safety events as part of their commitment to zero harm. By celebrating these contributions, the VHA fosters respect, joy, and satisfaction in the work. Staff are encouraged to nominate colleagues for recognition, reinforcing a supportive environment. Prompts invite teams to discuss how they celebrate JPSR reporting and how they’d like to enhance this culture of appreciation.
The Week 6 huddle card covers common misconceptions about JPSR. Key facts include that JPSRs are confidential, not for disciplinary action, and can be submitted by any staff member at any time. Only PS can view reporter identities for clarification purposes. The card concludes with prompts to ensure staff know how to access JPSR support and resources.
Measuring the impact on staff was essential to assess effectiveness and gather data for program improvement. To evaluate the impact of the huddle cards on the staff, the team provided a voluntary and anonymous 9 question survey (Figure 2). The survey was completed before the pilot began and again at the end of Week 6.

Questions 1 through 5 and 7 through 9 pertained to participants’ perceived knowledge and understanding of aspects of the JPSR. Perceived improvement among intensive care unit (ICU) participants ranged from 15% to 53%. There was a positive increase associated with every question with the top improvements: question 8, “How do you rate your understanding of how we share safety events for system and process improvement?” (53.4% increase); question 5, “How do you rate your understanding of what happens to a JPSR after it is entered?” (51.9% increase), and question 9, “How do you rate your understanding of the concepts of trust, psychological safety and a just culture?” (47.8% increase).
The survey analysis was not able to track individual changes. As a result, the findings reflect an overall change for the entire study group. Moreover, the questions assessed participants’ perceived knowledge rather than actual knowledge gained. It is important to note that there may be a significant gap between the actual knowledge gained and how participants perceive it. Additionally, improvement in knowledge and comprehension does not necessarily translate into behavior changes.
CONCLUSIONS
The use of JPSR huddle cards and direct engagement with staff during safety huddles yielded positive outcomes. On average, participants demonstrated higher scores in posttest questions compared to pretest questions. The posttest scores were consistently higher than the pretest scores, showing an average increase of around 2 standard deviations across all questions. This indicates an improvement in participants’ perceived knowledge and comprehension of the JPSR material.
During the pilot implementation of the huddle cards, there was a notable improvement in team member engagement. The structured format of the cards facilitated focused and meaningful discussions during safety huddles, encouraging open dialogue and fostering a culture of safety. Team members actively participated in identifying potential risks, sharing observations, and proposing actionable solutions, which reflected an enhanced sense of ownership regarding safety practices.
The support dialogue facilitated by the huddle cards highlighted the significance of mutual accountability and a collective commitment to achieving zero harm. This collaborative environment strengthened trust among team members and underscored the importance of shared vigilance in preventing adverse events. The pilot demonstrated the potential of huddle cards as an essential tool for enhancing team-based safety initiatives and promoting a culture of high reliability within the organization.
The total number of JPSR events in the ICU rose from 156 in FY 23 to 170 in FY 24. Adverse events increased from 19 to 31, while close calls saw a slight uptick from 137 to 139. Despite the overall rise in adverse events, a detailed analysis indicated that incidents of moderate harm decreased from 4 in FY 23 to 2 in FY 24. Furthermore, there was 1 reported case of death or severe harm in FY 23, which decreased to 0 in FY 24. This trend is consistent with the overarching objective of a high-reliability organization to achieve zero harm.
The next step is to expand this initiative across CTVHCS. This initiative aims to make this an annual education for all areas. The JPSR huddle card toolkit will be formatted by the media department for easy printing and retrieval. Leaders within units, clinics, and services will be empowered to facilitate the sessions in their safety huddles and reap the same outcomes as in the pilot. CTVHCS PS will monitor the effectiveness of this through ongoing CTVHCS patient safety rounding and future AES.
- Essen K, Villalobos C, Sculli GL, Steinbach L. Establishing a just culture: implications for the Veterans Health Administration journey to high reliability. Fed Pract. 2024;41:290-297. doi:10.12788/fp.0512
- Louis MY, Hussain LR, Dhanraj DN, et al. Improving patient safety event reporting among residents and teaching faculty. Ochsner J. 2016;16:73-80.
- Pimental CB, Snow AL, Carnes SL, et al. Huddles and their effectiveness at the frontlines of clinical care: a scoping review. J Gen Intern Med. 2021;36:2772-2783. doi:10.1007/s11606-021-06632-9
- National Academies of Sciences, Engineering, and Medicine. Appendix C: Nature of Veterans Health Administration Facilities Management (Engineering) Tasks and Staffing. Facilities Staffing Requirements for the Veterans Health Administration-Resource Planning and Methodology for the Future. National Academies Press. 2020:105-116. Accessed August 11, 2025. https://nap.nationalacademies.org/read/25454/chapter/11
- Woodier N, Burnett C, Moppett I. The value of learning from near misses to improve patient safety: a scoping review. J Patient Saf. 2023;19:42-47. doi:10.1097/pts.0000000000001078
- Ismail A, Khalid SNM. Patient safety culture and its determinants among healthcare professionals at a cluster hospital in Malaysia: a cross-sectional study. BMJ Open. 2022;12:e060546. doi:10.1136/bmjopen-2021-060546
- Ngo J, Lau D, Ploquin J, Receveur T, Stassen K, Del Castilho C. Improving incident reporting among physicians at south health campus hospital. BMJ Open Qual. 2022;11:e001945. doi:10.1136/bmjoq-2022-001945
- Oweidat I, Al-Mugheed K, Alsenany SA, et al. Awareness of reporting practices and barriers to incident reporting among nurses. BMC Nurs. 2023;22:231. doi:10.1186/s12912-023-01376-9
- Essen K, Villalobos C, Sculli GL, Steinbach L. Establishing a just culture: implications for the Veterans Health Administration journey to high reliability. Fed Pract. 2024;41:290-297. doi:10.12788/fp.0512
- Louis MY, Hussain LR, Dhanraj DN, et al. Improving patient safety event reporting among residents and teaching faculty. Ochsner J. 2016;16:73-80.
- Pimental CB, Snow AL, Carnes SL, et al. Huddles and their effectiveness at the frontlines of clinical care: a scoping review. J Gen Intern Med. 2021;36:2772-2783. doi:10.1007/s11606-021-06632-9
- National Academies of Sciences, Engineering, and Medicine. Appendix C: Nature of Veterans Health Administration Facilities Management (Engineering) Tasks and Staffing. Facilities Staffing Requirements for the Veterans Health Administration-Resource Planning and Methodology for the Future. National Academies Press. 2020:105-116. Accessed August 11, 2025. https://nap.nationalacademies.org/read/25454/chapter/11
- Woodier N, Burnett C, Moppett I. The value of learning from near misses to improve patient safety: a scoping review. J Patient Saf. 2023;19:42-47. doi:10.1097/pts.0000000000001078
- Ismail A, Khalid SNM. Patient safety culture and its determinants among healthcare professionals at a cluster hospital in Malaysia: a cross-sectional study. BMJ Open. 2022;12:e060546. doi:10.1136/bmjopen-2021-060546
- Ngo J, Lau D, Ploquin J, Receveur T, Stassen K, Del Castilho C. Improving incident reporting among physicians at south health campus hospital. BMJ Open Qual. 2022;11:e001945. doi:10.1136/bmjoq-2022-001945
- Oweidat I, Al-Mugheed K, Alsenany SA, et al. Awareness of reporting practices and barriers to incident reporting among nurses. BMC Nurs. 2023;22:231. doi:10.1186/s12912-023-01376-9
Empowering Culture Change and Safety on the Journey to Zero Harm With Huddle Cards
Empowering Culture Change and Safety on the Journey to Zero Harm With Huddle Cards
Enhancing Veteran Health Research: A Quality Improvement Initiative to Optimize Biorepository Efficiency
Purpose
Biorepositories are critical to scientific research within the VA. They offer high-quality, well-characterized biospecimens linked to clinical, demographic, and molecular data. Biorepositories support studies on disease mechanisms, personalized therapies, and emerging infectious diseases by systematically collecting, processing, storing, and distributing biological materials, including tissue, blood, and DNA samples. Within the Department of Veterans Affairs (VA), biorepositories provide essential support to clinical and translational research on service- related conditions such as PTSD, traumatic brain injury, cancers, and toxic exposures. While the need for harmonized quality processes and resource allocation has long been acknowledged within the biorepository community (Siwek, 2015), each biorepository operates independently, limiting scalability and standardization. This quality improvement project describes a collaboration between two VA biorepository sites supporting a national genomic study investigating disease risk and treatment outcomes. The project aimed to expand capacity, improve processing times, and enhance quality control. Each site mirrors the other’s functions, including receiving, accessioning, processing, storing, and shipping biospecimens, and serves as a contingency site to strengthen operational resilience.
Methods
To address space limitations and improve processing efficiency, one site implemented a custom rack design, expanding storage capacity per freezer. Robotic workflows were optimized, reducing biospecimen processing time. An in-process quality control step was introduced to identify data discrepancies earlier in the workflow, reducing investigation time and supporting overall data integrity. Efficiency was measured by the increase in storage capacity and decreased processing time. Descriptive statistics were used to evaluate changes in performance. Metrics were monitored over twelve months and compared against baseline data.
Results
Following implementation, storage capacity per freezer increased by 20%, and specimen processing time decreased by 30%. The new quality control checkpoint reduced investigation times by 98%, resulting in a more streamlined workflow. These improvements enhanced coordination between sites and improved support for ongoing studies.
Conclusions
This effort demonstrates that collaboration between biorepositories can significantly enhance efficiency, reduce turnaround times, and support high-quality research. Strengthening infrastructure through joint initiatives enables more effective support of large-scale clinical studies and contributes to improved outcomes for Veterans. These findings may also inform process improvements at other VA research facilities.
Purpose
Biorepositories are critical to scientific research within the VA. They offer high-quality, well-characterized biospecimens linked to clinical, demographic, and molecular data. Biorepositories support studies on disease mechanisms, personalized therapies, and emerging infectious diseases by systematically collecting, processing, storing, and distributing biological materials, including tissue, blood, and DNA samples. Within the Department of Veterans Affairs (VA), biorepositories provide essential support to clinical and translational research on service- related conditions such as PTSD, traumatic brain injury, cancers, and toxic exposures. While the need for harmonized quality processes and resource allocation has long been acknowledged within the biorepository community (Siwek, 2015), each biorepository operates independently, limiting scalability and standardization. This quality improvement project describes a collaboration between two VA biorepository sites supporting a national genomic study investigating disease risk and treatment outcomes. The project aimed to expand capacity, improve processing times, and enhance quality control. Each site mirrors the other’s functions, including receiving, accessioning, processing, storing, and shipping biospecimens, and serves as a contingency site to strengthen operational resilience.
Methods
To address space limitations and improve processing efficiency, one site implemented a custom rack design, expanding storage capacity per freezer. Robotic workflows were optimized, reducing biospecimen processing time. An in-process quality control step was introduced to identify data discrepancies earlier in the workflow, reducing investigation time and supporting overall data integrity. Efficiency was measured by the increase in storage capacity and decreased processing time. Descriptive statistics were used to evaluate changes in performance. Metrics were monitored over twelve months and compared against baseline data.
Results
Following implementation, storage capacity per freezer increased by 20%, and specimen processing time decreased by 30%. The new quality control checkpoint reduced investigation times by 98%, resulting in a more streamlined workflow. These improvements enhanced coordination between sites and improved support for ongoing studies.
Conclusions
This effort demonstrates that collaboration between biorepositories can significantly enhance efficiency, reduce turnaround times, and support high-quality research. Strengthening infrastructure through joint initiatives enables more effective support of large-scale clinical studies and contributes to improved outcomes for Veterans. These findings may also inform process improvements at other VA research facilities.
Purpose
Biorepositories are critical to scientific research within the VA. They offer high-quality, well-characterized biospecimens linked to clinical, demographic, and molecular data. Biorepositories support studies on disease mechanisms, personalized therapies, and emerging infectious diseases by systematically collecting, processing, storing, and distributing biological materials, including tissue, blood, and DNA samples. Within the Department of Veterans Affairs (VA), biorepositories provide essential support to clinical and translational research on service- related conditions such as PTSD, traumatic brain injury, cancers, and toxic exposures. While the need for harmonized quality processes and resource allocation has long been acknowledged within the biorepository community (Siwek, 2015), each biorepository operates independently, limiting scalability and standardization. This quality improvement project describes a collaboration between two VA biorepository sites supporting a national genomic study investigating disease risk and treatment outcomes. The project aimed to expand capacity, improve processing times, and enhance quality control. Each site mirrors the other’s functions, including receiving, accessioning, processing, storing, and shipping biospecimens, and serves as a contingency site to strengthen operational resilience.
Methods
To address space limitations and improve processing efficiency, one site implemented a custom rack design, expanding storage capacity per freezer. Robotic workflows were optimized, reducing biospecimen processing time. An in-process quality control step was introduced to identify data discrepancies earlier in the workflow, reducing investigation time and supporting overall data integrity. Efficiency was measured by the increase in storage capacity and decreased processing time. Descriptive statistics were used to evaluate changes in performance. Metrics were monitored over twelve months and compared against baseline data.
Results
Following implementation, storage capacity per freezer increased by 20%, and specimen processing time decreased by 30%. The new quality control checkpoint reduced investigation times by 98%, resulting in a more streamlined workflow. These improvements enhanced coordination between sites and improved support for ongoing studies.
Conclusions
This effort demonstrates that collaboration between biorepositories can significantly enhance efficiency, reduce turnaround times, and support high-quality research. Strengthening infrastructure through joint initiatives enables more effective support of large-scale clinical studies and contributes to improved outcomes for Veterans. These findings may also inform process improvements at other VA research facilities.
Enhancing Coding Accuracy at the Hematology/Oncology Clinic: Is It Time to Hire a Dedicated Coder?
Background
Accurate clinical coding that reflects all diagnoses and problems addressed during a patient encounter is essential for the cancer program’s data quality, research initiatives, and securing VERA (Veterans Equitable Resource Allocation) funding. However, providers often face barriers such as limited time during patient visits and difficulty navigating Electronic health record (EHR) systems. These challenges lead to inaccurate coding, which undermines downstream data integrity. This quality improvement (QI) study aimed to identify these barriers and implement an intervention to improve coding accuracy, while also assessing the financial implications of improved documentation.
Methods
This QI study was conducted at the Albany Stratton VA Medical Center, focusing on hematology/ oncology outpatient encounters. A baseline chart audit of diagnosis codes from June 2023 revealed an accuracy rate of 69.8%. To address this, an intervention was implemented in which dedicated coders were assigned to support attending physicians in coding for over a two-week period. These coders reviewed and corrected diagnosis codes in real-time. A follow-up audit conducted after the intervention showed an improved coding accuracy of 82%.
Discussion/Implications
Coding remains a timeconsuming task for providers, made more difficult by EHR systems that are not user-friendly. This study demonstrated that involving dedicated coders significantly improves documentation accuracy—from 69% to 82%. In addition to data quality, the financial benefits are notable. A projected annual return on investment of $216,094 was calculated, based on an internal analysis showing that in a sample of 124 patients, 10% could have qualified for higher VERA funding based on accurate coding, generating an estimated $17,427 in additional reimbursement per patient. This cost-benefit ratio supports the recommendation to staff dedicated coders. Other interventions were also utilised, such as updating the national encounter form and auto-populating documentation in Dragon software, but had limited impact and did not directly address diagnosis accuracy respectively.
Conclusions
Targeted interventions improved coding accuracy, but sustainability remains a challenge due to time and system limitations. Future efforts should focus on hiring full-time coders. These steps can further enhance coding quality and potentially increase hospital revenue.
Background
Accurate clinical coding that reflects all diagnoses and problems addressed during a patient encounter is essential for the cancer program’s data quality, research initiatives, and securing VERA (Veterans Equitable Resource Allocation) funding. However, providers often face barriers such as limited time during patient visits and difficulty navigating Electronic health record (EHR) systems. These challenges lead to inaccurate coding, which undermines downstream data integrity. This quality improvement (QI) study aimed to identify these barriers and implement an intervention to improve coding accuracy, while also assessing the financial implications of improved documentation.
Methods
This QI study was conducted at the Albany Stratton VA Medical Center, focusing on hematology/ oncology outpatient encounters. A baseline chart audit of diagnosis codes from June 2023 revealed an accuracy rate of 69.8%. To address this, an intervention was implemented in which dedicated coders were assigned to support attending physicians in coding for over a two-week period. These coders reviewed and corrected diagnosis codes in real-time. A follow-up audit conducted after the intervention showed an improved coding accuracy of 82%.
Discussion/Implications
Coding remains a timeconsuming task for providers, made more difficult by EHR systems that are not user-friendly. This study demonstrated that involving dedicated coders significantly improves documentation accuracy—from 69% to 82%. In addition to data quality, the financial benefits are notable. A projected annual return on investment of $216,094 was calculated, based on an internal analysis showing that in a sample of 124 patients, 10% could have qualified for higher VERA funding based on accurate coding, generating an estimated $17,427 in additional reimbursement per patient. This cost-benefit ratio supports the recommendation to staff dedicated coders. Other interventions were also utilised, such as updating the national encounter form and auto-populating documentation in Dragon software, but had limited impact and did not directly address diagnosis accuracy respectively.
Conclusions
Targeted interventions improved coding accuracy, but sustainability remains a challenge due to time and system limitations. Future efforts should focus on hiring full-time coders. These steps can further enhance coding quality and potentially increase hospital revenue.
Background
Accurate clinical coding that reflects all diagnoses and problems addressed during a patient encounter is essential for the cancer program’s data quality, research initiatives, and securing VERA (Veterans Equitable Resource Allocation) funding. However, providers often face barriers such as limited time during patient visits and difficulty navigating Electronic health record (EHR) systems. These challenges lead to inaccurate coding, which undermines downstream data integrity. This quality improvement (QI) study aimed to identify these barriers and implement an intervention to improve coding accuracy, while also assessing the financial implications of improved documentation.
Methods
This QI study was conducted at the Albany Stratton VA Medical Center, focusing on hematology/ oncology outpatient encounters. A baseline chart audit of diagnosis codes from June 2023 revealed an accuracy rate of 69.8%. To address this, an intervention was implemented in which dedicated coders were assigned to support attending physicians in coding for over a two-week period. These coders reviewed and corrected diagnosis codes in real-time. A follow-up audit conducted after the intervention showed an improved coding accuracy of 82%.
Discussion/Implications
Coding remains a timeconsuming task for providers, made more difficult by EHR systems that are not user-friendly. This study demonstrated that involving dedicated coders significantly improves documentation accuracy—from 69% to 82%. In addition to data quality, the financial benefits are notable. A projected annual return on investment of $216,094 was calculated, based on an internal analysis showing that in a sample of 124 patients, 10% could have qualified for higher VERA funding based on accurate coding, generating an estimated $17,427 in additional reimbursement per patient. This cost-benefit ratio supports the recommendation to staff dedicated coders. Other interventions were also utilised, such as updating the national encounter form and auto-populating documentation in Dragon software, but had limited impact and did not directly address diagnosis accuracy respectively.
Conclusions
Targeted interventions improved coding accuracy, but sustainability remains a challenge due to time and system limitations. Future efforts should focus on hiring full-time coders. These steps can further enhance coding quality and potentially increase hospital revenue.
Evaluating the Implementation of 60-minute Iron Dextran Infusions at a Rural Health Center
Background
Due to risk for infusion-related reactions (IRR), administration of iron dextran requires an initial test dose with an extended monitoring period and subsequent doses given as a slow infusion over 2-3 hours. Safe use of a 60-minute iron dextran infusion protocol has been demonstrated previously at fully staffed academic teaching institutions. This study sought to determine the impact on patient safety and infusion clinic efficiency after implementing a 60-minute iron dextran administration protocol at a small, rural facility utilizing a decentralized clinical model.
Methods
This single-site, prospective, interventional study was conducted at a rural level 1C Veterans Affairs secondary care facility. The Hematology/Oncology clinic staffing includes one onsite clinical pharmacy practitioner (CPP) and advanced practice nurse. Remote providers complete patient encounters through video and telehealth modalities. A 60-minute iron dextran infusion service line agreement was designed by the Hematology/Oncology CPP and approved by the facility prior to data collection. The protocol included administration of a test dose and 15-minute monitoring period for treatment naïve patients. Pre-medications were allowed at the discretion of the ordering providers. All patients who received iron dextran between May 31, 2024 and April 14, 2025 per protocol were included in data analysis and results were stratified by treatment naïve and pre-treated patients. Outcomes included the proportion of patients experiencing any grade of IRR based on the Common Criteria for Adverse Events Version 5.0, and the average duration of administration. Descriptive statistics were used for safety and efficiency outcomes.
Results
Eighty patients received 103 iron dextran infusions and were included for analysis. Pre-medications were administered for 16 of the 103 (15.5%) included infusions. Two patients experienced grade 1 IRR (nausea) on 4 occasions (3.8%) which quickly resolved with intravenous ondansetron, and full iron dextran doses were received. The mean infusion time was 94 minutes in the treatment naïve cohort vs 71 minutes in the pre-treated cohort.
Conclusions
This study suggests a Hematology/ Oncology CPP developed iron dextran 60-minute infusion protocol may be safely and efficiently administered for qualifying patients in a decentralized, rural healthcare setting.
Background
Due to risk for infusion-related reactions (IRR), administration of iron dextran requires an initial test dose with an extended monitoring period and subsequent doses given as a slow infusion over 2-3 hours. Safe use of a 60-minute iron dextran infusion protocol has been demonstrated previously at fully staffed academic teaching institutions. This study sought to determine the impact on patient safety and infusion clinic efficiency after implementing a 60-minute iron dextran administration protocol at a small, rural facility utilizing a decentralized clinical model.
Methods
This single-site, prospective, interventional study was conducted at a rural level 1C Veterans Affairs secondary care facility. The Hematology/Oncology clinic staffing includes one onsite clinical pharmacy practitioner (CPP) and advanced practice nurse. Remote providers complete patient encounters through video and telehealth modalities. A 60-minute iron dextran infusion service line agreement was designed by the Hematology/Oncology CPP and approved by the facility prior to data collection. The protocol included administration of a test dose and 15-minute monitoring period for treatment naïve patients. Pre-medications were allowed at the discretion of the ordering providers. All patients who received iron dextran between May 31, 2024 and April 14, 2025 per protocol were included in data analysis and results were stratified by treatment naïve and pre-treated patients. Outcomes included the proportion of patients experiencing any grade of IRR based on the Common Criteria for Adverse Events Version 5.0, and the average duration of administration. Descriptive statistics were used for safety and efficiency outcomes.
Results
Eighty patients received 103 iron dextran infusions and were included for analysis. Pre-medications were administered for 16 of the 103 (15.5%) included infusions. Two patients experienced grade 1 IRR (nausea) on 4 occasions (3.8%) which quickly resolved with intravenous ondansetron, and full iron dextran doses were received. The mean infusion time was 94 minutes in the treatment naïve cohort vs 71 minutes in the pre-treated cohort.
Conclusions
This study suggests a Hematology/ Oncology CPP developed iron dextran 60-minute infusion protocol may be safely and efficiently administered for qualifying patients in a decentralized, rural healthcare setting.
Background
Due to risk for infusion-related reactions (IRR), administration of iron dextran requires an initial test dose with an extended monitoring period and subsequent doses given as a slow infusion over 2-3 hours. Safe use of a 60-minute iron dextran infusion protocol has been demonstrated previously at fully staffed academic teaching institutions. This study sought to determine the impact on patient safety and infusion clinic efficiency after implementing a 60-minute iron dextran administration protocol at a small, rural facility utilizing a decentralized clinical model.
Methods
This single-site, prospective, interventional study was conducted at a rural level 1C Veterans Affairs secondary care facility. The Hematology/Oncology clinic staffing includes one onsite clinical pharmacy practitioner (CPP) and advanced practice nurse. Remote providers complete patient encounters through video and telehealth modalities. A 60-minute iron dextran infusion service line agreement was designed by the Hematology/Oncology CPP and approved by the facility prior to data collection. The protocol included administration of a test dose and 15-minute monitoring period for treatment naïve patients. Pre-medications were allowed at the discretion of the ordering providers. All patients who received iron dextran between May 31, 2024 and April 14, 2025 per protocol were included in data analysis and results were stratified by treatment naïve and pre-treated patients. Outcomes included the proportion of patients experiencing any grade of IRR based on the Common Criteria for Adverse Events Version 5.0, and the average duration of administration. Descriptive statistics were used for safety and efficiency outcomes.
Results
Eighty patients received 103 iron dextran infusions and were included for analysis. Pre-medications were administered for 16 of the 103 (15.5%) included infusions. Two patients experienced grade 1 IRR (nausea) on 4 occasions (3.8%) which quickly resolved with intravenous ondansetron, and full iron dextran doses were received. The mean infusion time was 94 minutes in the treatment naïve cohort vs 71 minutes in the pre-treated cohort.
Conclusions
This study suggests a Hematology/ Oncology CPP developed iron dextran 60-minute infusion protocol may be safely and efficiently administered for qualifying patients in a decentralized, rural healthcare setting.
Improving Palliative Care Referrals through Education of Hematology/Oncology Fellows: A QI Initiative
Purpose/Background
Palliative care referrals are recommended for patients with advanced or metastatic cancer to enhance patient and caregiver outcomes. However, challenges like delays or lack of referrals hinder implementation. This study identified rate of palliative care referrals at James A. Haley Veterans’ Hospital in Tampa, Florida; explored potential barriers to referral, and implemented targeted interventions to improve referral rates and patient outcomes.
Methods
A Plan-Do-Study-Act (PDSA) cycle was used for this quality improvement project. Data was collected from electronic medical record, focusing on consult dates, patient demographics, and reasons for seeking palliative care. Pre-intervention surveys were administered to Hematology-Oncology fellows at the institution to identify barriers to referral. Following a root cause analysis, a targeted intervention was developed, focusing on educational programs for fellows for streamlined referral processes.
Results
Before the intervention, monthly average for palliative care consults was low (3-8, typically 5). Pre-intervention surveys revealed that fellows lacked knowledge about palliative care resources, which contributed to low referral rates. To address this issue, a didactic session led by a palliative care specialist was conducted for the fellows in the fellowship program. This session provided education on the role of palliative care, how to initiate referrals, and the benefits of early involvement of palliative care teams in oncology patient management. Post-intervention surveys showed a marked improvement in fellows’ confidence regarding identification of patients suitable for palliative care. Following the session, 90% (9/10) of fellows reported being “very likely” to consult palliative care more often and 80% (8/10) indicated they were “very likely” to initiate palliative care discussions earlier in patient’s disease trajectory, with the remaining 20% (2/10) reporting a neutral stance. All fellows (100%) agreed that earlier palliative care involvement improves patient outcomes.
Implications/Significance
This PDSA cycle demonstrated that targeted education for fellows can increase awareness of palliative care resources and improve referral rates. Future work will focus on reassessing usage of palliative care consults post-intervention to evaluate effects of fellows’ education of appropriate palliative care consultation, make necessary interventions based on data and further evaluate the long-term impact on patient outcomes at James A. Haley Veterans’ Hospital.
Purpose/Background
Palliative care referrals are recommended for patients with advanced or metastatic cancer to enhance patient and caregiver outcomes. However, challenges like delays or lack of referrals hinder implementation. This study identified rate of palliative care referrals at James A. Haley Veterans’ Hospital in Tampa, Florida; explored potential barriers to referral, and implemented targeted interventions to improve referral rates and patient outcomes.
Methods
A Plan-Do-Study-Act (PDSA) cycle was used for this quality improvement project. Data was collected from electronic medical record, focusing on consult dates, patient demographics, and reasons for seeking palliative care. Pre-intervention surveys were administered to Hematology-Oncology fellows at the institution to identify barriers to referral. Following a root cause analysis, a targeted intervention was developed, focusing on educational programs for fellows for streamlined referral processes.
Results
Before the intervention, monthly average for palliative care consults was low (3-8, typically 5). Pre-intervention surveys revealed that fellows lacked knowledge about palliative care resources, which contributed to low referral rates. To address this issue, a didactic session led by a palliative care specialist was conducted for the fellows in the fellowship program. This session provided education on the role of palliative care, how to initiate referrals, and the benefits of early involvement of palliative care teams in oncology patient management. Post-intervention surveys showed a marked improvement in fellows’ confidence regarding identification of patients suitable for palliative care. Following the session, 90% (9/10) of fellows reported being “very likely” to consult palliative care more often and 80% (8/10) indicated they were “very likely” to initiate palliative care discussions earlier in patient’s disease trajectory, with the remaining 20% (2/10) reporting a neutral stance. All fellows (100%) agreed that earlier palliative care involvement improves patient outcomes.
Implications/Significance
This PDSA cycle demonstrated that targeted education for fellows can increase awareness of palliative care resources and improve referral rates. Future work will focus on reassessing usage of palliative care consults post-intervention to evaluate effects of fellows’ education of appropriate palliative care consultation, make necessary interventions based on data and further evaluate the long-term impact on patient outcomes at James A. Haley Veterans’ Hospital.
Purpose/Background
Palliative care referrals are recommended for patients with advanced or metastatic cancer to enhance patient and caregiver outcomes. However, challenges like delays or lack of referrals hinder implementation. This study identified rate of palliative care referrals at James A. Haley Veterans’ Hospital in Tampa, Florida; explored potential barriers to referral, and implemented targeted interventions to improve referral rates and patient outcomes.
Methods
A Plan-Do-Study-Act (PDSA) cycle was used for this quality improvement project. Data was collected from electronic medical record, focusing on consult dates, patient demographics, and reasons for seeking palliative care. Pre-intervention surveys were administered to Hematology-Oncology fellows at the institution to identify barriers to referral. Following a root cause analysis, a targeted intervention was developed, focusing on educational programs for fellows for streamlined referral processes.
Results
Before the intervention, monthly average for palliative care consults was low (3-8, typically 5). Pre-intervention surveys revealed that fellows lacked knowledge about palliative care resources, which contributed to low referral rates. To address this issue, a didactic session led by a palliative care specialist was conducted for the fellows in the fellowship program. This session provided education on the role of palliative care, how to initiate referrals, and the benefits of early involvement of palliative care teams in oncology patient management. Post-intervention surveys showed a marked improvement in fellows’ confidence regarding identification of patients suitable for palliative care. Following the session, 90% (9/10) of fellows reported being “very likely” to consult palliative care more often and 80% (8/10) indicated they were “very likely” to initiate palliative care discussions earlier in patient’s disease trajectory, with the remaining 20% (2/10) reporting a neutral stance. All fellows (100%) agreed that earlier palliative care involvement improves patient outcomes.
Implications/Significance
This PDSA cycle demonstrated that targeted education for fellows can increase awareness of palliative care resources and improve referral rates. Future work will focus on reassessing usage of palliative care consults post-intervention to evaluate effects of fellows’ education of appropriate palliative care consultation, make necessary interventions based on data and further evaluate the long-term impact on patient outcomes at James A. Haley Veterans’ Hospital.
Optimizing Symptom Management in VA Oncology: A Workflow-Based Quality Improvement Initiative
Background
Enhancing symptom assessment and management of patients undergoing cancer treatment presents several challenges, ranging from workflow integration to application of evidenced-based interventions (Minteer, et al., 2023). Previously, our team conducted a VA mixed-methods study and identified a lack of standardized approaches for symptom assessment, lack of technology support to optimize workflows, and the need for adaptable workflows that reflect both facility and patient preferences. In response, the National Oncology Program Office at Palo Alto VA (PAVA) launched the Proactive Patient-Centered Care Program (PPP) to address these care gaps and develop a feasible, replicable, sustainable workflow to guide broader VA-wide implementation based on prior work conducted by the PAVA team (Banks, et al., 2024).
Methods
Prior to launch, the PPP team engaged oncology leadership in VISN21 and VISN22. Long Beach VA (LBVA) was selected as the initial pilot implementation site. A multidisciplinary group from PAVA and LBVA comprised of oncology and palliative care clinicians, nurses, pharmacists, a lay health worker, and project manager guided the workflow adaptations. To support scalability and sustainability, the Empowering Learning, Innovation, and experiences through Implementation of health Informatics (ELIXIR) team designed an electronic health record agnostic technology-enabled tool to support workflow. The group met weekly to bi-monthly over 5 months, virtually and two in-person sessions, to map current practices, co-develop workflows, and identify key decisions regarding patient eligibility criteria, frequency of symptom assessments, triage responsibilities, escalation protocols, and closed-loop communication processes.
Results
A technology-enabled workflow was developed to deploy proactive symptom assessment and management across VA oncology sites with streamlined coordination between peer support staff and clinicians along with technology to support timely interventions.
Conclusions
Process improvement for symptom management requires on the ground adaptation even within an integrated health system like the VA. This initiative underscores the need for multidisciplinary collaboration, sustainability, and technology integration to support long-term intervention fidelity and scalability. The workflow developed will guide the PPP program’s expansion to LBVA, with patient enrollment beginning May 2025. The approach used to develop this workflow will serve as a model for standardizing supportive care processes across the VA to account for local needs.
Background
Enhancing symptom assessment and management of patients undergoing cancer treatment presents several challenges, ranging from workflow integration to application of evidenced-based interventions (Minteer, et al., 2023). Previously, our team conducted a VA mixed-methods study and identified a lack of standardized approaches for symptom assessment, lack of technology support to optimize workflows, and the need for adaptable workflows that reflect both facility and patient preferences. In response, the National Oncology Program Office at Palo Alto VA (PAVA) launched the Proactive Patient-Centered Care Program (PPP) to address these care gaps and develop a feasible, replicable, sustainable workflow to guide broader VA-wide implementation based on prior work conducted by the PAVA team (Banks, et al., 2024).
Methods
Prior to launch, the PPP team engaged oncology leadership in VISN21 and VISN22. Long Beach VA (LBVA) was selected as the initial pilot implementation site. A multidisciplinary group from PAVA and LBVA comprised of oncology and palliative care clinicians, nurses, pharmacists, a lay health worker, and project manager guided the workflow adaptations. To support scalability and sustainability, the Empowering Learning, Innovation, and experiences through Implementation of health Informatics (ELIXIR) team designed an electronic health record agnostic technology-enabled tool to support workflow. The group met weekly to bi-monthly over 5 months, virtually and two in-person sessions, to map current practices, co-develop workflows, and identify key decisions regarding patient eligibility criteria, frequency of symptom assessments, triage responsibilities, escalation protocols, and closed-loop communication processes.
Results
A technology-enabled workflow was developed to deploy proactive symptom assessment and management across VA oncology sites with streamlined coordination between peer support staff and clinicians along with technology to support timely interventions.
Conclusions
Process improvement for symptom management requires on the ground adaptation even within an integrated health system like the VA. This initiative underscores the need for multidisciplinary collaboration, sustainability, and technology integration to support long-term intervention fidelity and scalability. The workflow developed will guide the PPP program’s expansion to LBVA, with patient enrollment beginning May 2025. The approach used to develop this workflow will serve as a model for standardizing supportive care processes across the VA to account for local needs.
Background
Enhancing symptom assessment and management of patients undergoing cancer treatment presents several challenges, ranging from workflow integration to application of evidenced-based interventions (Minteer, et al., 2023). Previously, our team conducted a VA mixed-methods study and identified a lack of standardized approaches for symptom assessment, lack of technology support to optimize workflows, and the need for adaptable workflows that reflect both facility and patient preferences. In response, the National Oncology Program Office at Palo Alto VA (PAVA) launched the Proactive Patient-Centered Care Program (PPP) to address these care gaps and develop a feasible, replicable, sustainable workflow to guide broader VA-wide implementation based on prior work conducted by the PAVA team (Banks, et al., 2024).
Methods
Prior to launch, the PPP team engaged oncology leadership in VISN21 and VISN22. Long Beach VA (LBVA) was selected as the initial pilot implementation site. A multidisciplinary group from PAVA and LBVA comprised of oncology and palliative care clinicians, nurses, pharmacists, a lay health worker, and project manager guided the workflow adaptations. To support scalability and sustainability, the Empowering Learning, Innovation, and experiences through Implementation of health Informatics (ELIXIR) team designed an electronic health record agnostic technology-enabled tool to support workflow. The group met weekly to bi-monthly over 5 months, virtually and two in-person sessions, to map current practices, co-develop workflows, and identify key decisions regarding patient eligibility criteria, frequency of symptom assessments, triage responsibilities, escalation protocols, and closed-loop communication processes.
Results
A technology-enabled workflow was developed to deploy proactive symptom assessment and management across VA oncology sites with streamlined coordination between peer support staff and clinicians along with technology to support timely interventions.
Conclusions
Process improvement for symptom management requires on the ground adaptation even within an integrated health system like the VA. This initiative underscores the need for multidisciplinary collaboration, sustainability, and technology integration to support long-term intervention fidelity and scalability. The workflow developed will guide the PPP program’s expansion to LBVA, with patient enrollment beginning May 2025. The approach used to develop this workflow will serve as a model for standardizing supportive care processes across the VA to account for local needs.
Implementation of Consult Template Optimizes Hematology E-Consult Evaluation
Purpose/Background
The purpose of this project was to understand how implementing a consult template could optimize hematology E-consult evaluation. At the Tampa VA, providers can submit hematology E-consults for interpretation of lab abnormalities and management recommendations that do not require an in-person hematology evaluation. Previously, submission of an E-consult did not require prerequisite labs or imaging or for lab parameters to be met, leading to an increased number of hematology E-consults and subsequently, lower efficiency for hematologists.
Methods
A hematology E-consult template was created through collaboration between the hematology/ oncology and ambulatory care sections, which lists specific diagnoses and required parameters/workup needed for each diagnosis prior to submission of the E-consult. If those criteria were not met, the consult was cancelled. A representative sample of one month pre- and post-implementation data was analyzed.
Results
The E-consult template was implemented in September 2024. From April to August 2024, the average number of E-consults per month was 243, averaging at 11.0 per day, while from October 2024 to February 2025, the average number of E-consults per month was 146.4, averaging at 6.6 per day. In August 2024, the leading reasons for consult were anemia (77), leukocytosis (26), and thrombocytopenia (24). That month, there were 15 consult cancellations, with the primary reason being the patient was established in clinic (9). In October 2024, the leading reasons for consult were anemia (39), leukocytosis (14), and thrombocytopenia (13). That month, there were 34 consult cancellations, with the primary reason being that hematology advised a clinic consultation rather than an E-consult (10).
Implications/Significance
These data reveal that the hematology E-consult template was associated with a decreased number of E-consults per day and per month. Implementation of the hematology E-consult template allows the hematology consultants to focus on interpretation of lab results and providing management recommendations, as opposed to providing standard of care diagnostic recommendations. It also serves as an educational tool to referring providers, to understand appropriate indications for hematology E-consultation. Lastly, the template has created increased efficiency in providing hematology recommendations and ultimately, improved timely care for our veterans.
Purpose/Background
The purpose of this project was to understand how implementing a consult template could optimize hematology E-consult evaluation. At the Tampa VA, providers can submit hematology E-consults for interpretation of lab abnormalities and management recommendations that do not require an in-person hematology evaluation. Previously, submission of an E-consult did not require prerequisite labs or imaging or for lab parameters to be met, leading to an increased number of hematology E-consults and subsequently, lower efficiency for hematologists.
Methods
A hematology E-consult template was created through collaboration between the hematology/ oncology and ambulatory care sections, which lists specific diagnoses and required parameters/workup needed for each diagnosis prior to submission of the E-consult. If those criteria were not met, the consult was cancelled. A representative sample of one month pre- and post-implementation data was analyzed.
Results
The E-consult template was implemented in September 2024. From April to August 2024, the average number of E-consults per month was 243, averaging at 11.0 per day, while from October 2024 to February 2025, the average number of E-consults per month was 146.4, averaging at 6.6 per day. In August 2024, the leading reasons for consult were anemia (77), leukocytosis (26), and thrombocytopenia (24). That month, there were 15 consult cancellations, with the primary reason being the patient was established in clinic (9). In October 2024, the leading reasons for consult were anemia (39), leukocytosis (14), and thrombocytopenia (13). That month, there were 34 consult cancellations, with the primary reason being that hematology advised a clinic consultation rather than an E-consult (10).
Implications/Significance
These data reveal that the hematology E-consult template was associated with a decreased number of E-consults per day and per month. Implementation of the hematology E-consult template allows the hematology consultants to focus on interpretation of lab results and providing management recommendations, as opposed to providing standard of care diagnostic recommendations. It also serves as an educational tool to referring providers, to understand appropriate indications for hematology E-consultation. Lastly, the template has created increased efficiency in providing hematology recommendations and ultimately, improved timely care for our veterans.
Purpose/Background
The purpose of this project was to understand how implementing a consult template could optimize hematology E-consult evaluation. At the Tampa VA, providers can submit hematology E-consults for interpretation of lab abnormalities and management recommendations that do not require an in-person hematology evaluation. Previously, submission of an E-consult did not require prerequisite labs or imaging or for lab parameters to be met, leading to an increased number of hematology E-consults and subsequently, lower efficiency for hematologists.
Methods
A hematology E-consult template was created through collaboration between the hematology/ oncology and ambulatory care sections, which lists specific diagnoses and required parameters/workup needed for each diagnosis prior to submission of the E-consult. If those criteria were not met, the consult was cancelled. A representative sample of one month pre- and post-implementation data was analyzed.
Results
The E-consult template was implemented in September 2024. From April to August 2024, the average number of E-consults per month was 243, averaging at 11.0 per day, while from October 2024 to February 2025, the average number of E-consults per month was 146.4, averaging at 6.6 per day. In August 2024, the leading reasons for consult were anemia (77), leukocytosis (26), and thrombocytopenia (24). That month, there were 15 consult cancellations, with the primary reason being the patient was established in clinic (9). In October 2024, the leading reasons for consult were anemia (39), leukocytosis (14), and thrombocytopenia (13). That month, there were 34 consult cancellations, with the primary reason being that hematology advised a clinic consultation rather than an E-consult (10).
Implications/Significance
These data reveal that the hematology E-consult template was associated with a decreased number of E-consults per day and per month. Implementation of the hematology E-consult template allows the hematology consultants to focus on interpretation of lab results and providing management recommendations, as opposed to providing standard of care diagnostic recommendations. It also serves as an educational tool to referring providers, to understand appropriate indications for hematology E-consultation. Lastly, the template has created increased efficiency in providing hematology recommendations and ultimately, improved timely care for our veterans.
Enhancing Workforce Practices to Achieve Commission on Cancer Accreditation
Background
The American College of Surgeons’ Commission on Cancer (CoC) Accreditation requires establishment of a comprehensive cancer program, multi-disciplinary tumor boards, active cancer registry, quality improvement activities and cancer research.
Methods
In 2022, the Tibor Rubin VA Medical Center (TRVAMC) set out to obtain accreditation through enhancing workforce practices. Changes in workforce practices included (1) leadership engagement; (2) acquisition of staff; (3) enhancing staff efficiency and (4) inter-departmental collaboration, leading to CoC accreditation in August 2024. executive leadership team (ELT) buy-in was essential. ELT engagement included communicating the benefits of accreditation, alignment with organizational mission and values, protected time for Cancer Committee members, Chief of Staff presence in Cancer Committee, commitment to recruiting new staff, and membership in the Medical Executive Council to voice cancer program needs. New staff included a cancer program manager, cancer case conference RN care coordinator, certified oncology data specialist and survivorship nurse practitioner. Staff development included structured and focused training. Enhancing staff efficiency included developing standards of work with clear delineation of duties (delegation of specific CoC standards), decentralizing decision making, a shared governance council, and weekly Cancer Program meetings. These changes allowed staff members to be active, autonomous decision-making participants, and increased efficiency. Inter-departmental collaboration involved Hematology/Oncology, Surgery, Radiation Oncology, Pharmacy, Nutrition, Pathology, Palliative Care, Rehabilitation, Chaplaincy and Cancer Research, with key individuals serving as Cancer Committee members. Each department set performance goals and metrics. Each employee’s contribution was rated in annual performance reviews.
Results
TRVAMC thus elevated cancer care delivery standards through structured workforce practices within the framework of CoC standards required for accreditation. Additionally, the accreditation process achieved desirable and measurable outcomes, e.g. 100% growth in oncology dietitian referrals, 75% increase in early palliative care referrals (TRVAMC ranked in the top 5 in the US), and more than 200 patients enrolled in cancer clinical trials (TRVAMC was the highest enrolling VA in the US to NCI trials in 2024).
Conclusions
Our model demonstrates how strategic improvements in healthcare workforce practices at a VA can directly contribute to sustained improvements in quality and delivery of cancer care services.
Background
The American College of Surgeons’ Commission on Cancer (CoC) Accreditation requires establishment of a comprehensive cancer program, multi-disciplinary tumor boards, active cancer registry, quality improvement activities and cancer research.
Methods
In 2022, the Tibor Rubin VA Medical Center (TRVAMC) set out to obtain accreditation through enhancing workforce practices. Changes in workforce practices included (1) leadership engagement; (2) acquisition of staff; (3) enhancing staff efficiency and (4) inter-departmental collaboration, leading to CoC accreditation in August 2024. executive leadership team (ELT) buy-in was essential. ELT engagement included communicating the benefits of accreditation, alignment with organizational mission and values, protected time for Cancer Committee members, Chief of Staff presence in Cancer Committee, commitment to recruiting new staff, and membership in the Medical Executive Council to voice cancer program needs. New staff included a cancer program manager, cancer case conference RN care coordinator, certified oncology data specialist and survivorship nurse practitioner. Staff development included structured and focused training. Enhancing staff efficiency included developing standards of work with clear delineation of duties (delegation of specific CoC standards), decentralizing decision making, a shared governance council, and weekly Cancer Program meetings. These changes allowed staff members to be active, autonomous decision-making participants, and increased efficiency. Inter-departmental collaboration involved Hematology/Oncology, Surgery, Radiation Oncology, Pharmacy, Nutrition, Pathology, Palliative Care, Rehabilitation, Chaplaincy and Cancer Research, with key individuals serving as Cancer Committee members. Each department set performance goals and metrics. Each employee’s contribution was rated in annual performance reviews.
Results
TRVAMC thus elevated cancer care delivery standards through structured workforce practices within the framework of CoC standards required for accreditation. Additionally, the accreditation process achieved desirable and measurable outcomes, e.g. 100% growth in oncology dietitian referrals, 75% increase in early palliative care referrals (TRVAMC ranked in the top 5 in the US), and more than 200 patients enrolled in cancer clinical trials (TRVAMC was the highest enrolling VA in the US to NCI trials in 2024).
Conclusions
Our model demonstrates how strategic improvements in healthcare workforce practices at a VA can directly contribute to sustained improvements in quality and delivery of cancer care services.
Background
The American College of Surgeons’ Commission on Cancer (CoC) Accreditation requires establishment of a comprehensive cancer program, multi-disciplinary tumor boards, active cancer registry, quality improvement activities and cancer research.
Methods
In 2022, the Tibor Rubin VA Medical Center (TRVAMC) set out to obtain accreditation through enhancing workforce practices. Changes in workforce practices included (1) leadership engagement; (2) acquisition of staff; (3) enhancing staff efficiency and (4) inter-departmental collaboration, leading to CoC accreditation in August 2024. executive leadership team (ELT) buy-in was essential. ELT engagement included communicating the benefits of accreditation, alignment with organizational mission and values, protected time for Cancer Committee members, Chief of Staff presence in Cancer Committee, commitment to recruiting new staff, and membership in the Medical Executive Council to voice cancer program needs. New staff included a cancer program manager, cancer case conference RN care coordinator, certified oncology data specialist and survivorship nurse practitioner. Staff development included structured and focused training. Enhancing staff efficiency included developing standards of work with clear delineation of duties (delegation of specific CoC standards), decentralizing decision making, a shared governance council, and weekly Cancer Program meetings. These changes allowed staff members to be active, autonomous decision-making participants, and increased efficiency. Inter-departmental collaboration involved Hematology/Oncology, Surgery, Radiation Oncology, Pharmacy, Nutrition, Pathology, Palliative Care, Rehabilitation, Chaplaincy and Cancer Research, with key individuals serving as Cancer Committee members. Each department set performance goals and metrics. Each employee’s contribution was rated in annual performance reviews.
Results
TRVAMC thus elevated cancer care delivery standards through structured workforce practices within the framework of CoC standards required for accreditation. Additionally, the accreditation process achieved desirable and measurable outcomes, e.g. 100% growth in oncology dietitian referrals, 75% increase in early palliative care referrals (TRVAMC ranked in the top 5 in the US), and more than 200 patients enrolled in cancer clinical trials (TRVAMC was the highest enrolling VA in the US to NCI trials in 2024).
Conclusions
Our model demonstrates how strategic improvements in healthcare workforce practices at a VA can directly contribute to sustained improvements in quality and delivery of cancer care services.