User login
Acne before puberty: When to treat, when to worry
NEWPORT BEACH, CALIF. – according to Sheila Fallon Friedlander, MD.
“This is something you are going to see in your practice,” said Dr. Friedlander, a pediatric dermatologists at Rady Children’s Hospital–San Diego. It’s important to know when it’s time to be concerned and when another condition may be masquerading as acne, she said at the at Skin Disease Education Foundation’s Women’s & Pediatric Dermatology Seminar.
Dr. Friedlander, who is professor of dermatology and pediatrics at the University of California, San Diego, talked about treating acne in the following prepubertal age groups:
Neonatal acne (ages birth to 4 weeks)
Acne appears in this population up to 20% of the time, according to research, and it is much more common in males than in females, at a ratio of five to one.
The cause is “most likely the relationship between placental androgens and the baby’s adrenal glands,” Dr. Friedlander said. However, something more serious could be going on. “Look at the child and see if he’s sick. If he looks sick, then we need to worry.”
Hormonal abnormalities also could be a cause, she said. Refer a baby to a specialist if there are other signs of hyperandrogenism. However, “the likelihood is very low,” and she’s never needed to refer a neonate with acne for evaluation.
As for treatment, she said, “Mainly, I’m using tincture of time.” However, “many of my mothers have told me that topical yogurt application will work.” Why yogurt? It’s possible that its bacteria could play a role in combating acne, she said.
Masquerader alert! Beware of neonatal cephalic pustulosis, Dr. Friedlander cautioned, which may be an inflammatory response to yeast. Ketoconazole cream may be helpful.
Infantile acne (ages 0-12 months)
This form of acne is more common in males and may hint at the future development of severe adolescent acne. It does resolve but it may take months or years, Dr. Friedlander said.
In general, this acne isn’t a sign of something more serious. “You do not need to go crazy with the work-up,” she said. “With mild to moderate disease, with nothing else suspicious, I don’t do a big work-up.”
However, do consider whether the child is undergoing precocious puberty, Dr. Friedlander said. Signs include axillary hair, pubic hair, and body odor.
As for treatment of infantile acne, “start out topically” and consider options such as Bactrim (sulfamethoxazole/trimethoprim) and erythromycin.
Masquerader alert! Idiopathic facial aseptic granuloma can be mistaken for acne and abscess, and ultrasound is helpful to confirm it. “It’s not so easy to treat,” she said. “Ivermectin may be helpful. Sometimes you do cultures and make sure something else isn’t going on.”
Midchildhood (ages 1-7 years)
“It’s not as common to have acne develop in this age group, but when it develops you need to be concerned,” Dr. Friedlander said. “This is the age period when there is more often something really wrong.”
Be on the lookout for a family history of hormonal abnormalities, and check if the child is on medication. “You need to look carefully,” she said, adding that it’s important to check for signs of premature puberty such as giant spikes in growth, abnormally large hands and feet, genital changes, and body odor. Check blood pressure if you’re worried about an adrenal tumor.
It’s possible for children to develop precocious puberty – with acne – because of exposure to testosterone gel used by a father. Dehydroepiandrosterone (DHEA) creams also may cause the condition. “The more creams out there with androgenic effects, the more we may see it,” Dr. Friedlander said. “This is something to ask about because families may not be forthcoming.”
Masquerader alert! Perioral dermatitis may look like acne, and it may be linked to inhaled or topical steroids, she said.
Other masqueraders include demodex folliculitis, angiofibromas (think tuberous sclerosis), and keratosis pilaris (the most common type of bump on a children aged 1-7 years). The latter condition “is not the end of the world,” said Dr. Friedlander, who added that “I’ve never cured anyone of it.”
Prepubertal acne (ages 7 years to puberty)
Acne in this group is generally not worrisome, Dr. Friedlander said, but investigate further if there’s significant inflammation and signs of early sexual development or virilization.
Benzoyl peroxide wash may be enough to help the condition initially, and consider topical clindamycin or a combination product. “Start out slow,” she said. Twice a week to start might be appropriate. Moisturizers can be helpful, as can topical adapalene.
Also, keep in mind that even mild acne can be emotionally devastating to a child in this age group and worthy of treatment. “Your assessment may be very different than hers,” she said. It’s possible that “she has a few lesions, but she feels like an outcast.”
Dr. Friedlander reported no relevant financial disclosures. SDEF and this news organization are owned by the same parent company.
NEWPORT BEACH, CALIF. – according to Sheila Fallon Friedlander, MD.
“This is something you are going to see in your practice,” said Dr. Friedlander, a pediatric dermatologists at Rady Children’s Hospital–San Diego. It’s important to know when it’s time to be concerned and when another condition may be masquerading as acne, she said at the at Skin Disease Education Foundation’s Women’s & Pediatric Dermatology Seminar.
Dr. Friedlander, who is professor of dermatology and pediatrics at the University of California, San Diego, talked about treating acne in the following prepubertal age groups:
Neonatal acne (ages birth to 4 weeks)
Acne appears in this population up to 20% of the time, according to research, and it is much more common in males than in females, at a ratio of five to one.
The cause is “most likely the relationship between placental androgens and the baby’s adrenal glands,” Dr. Friedlander said. However, something more serious could be going on. “Look at the child and see if he’s sick. If he looks sick, then we need to worry.”
Hormonal abnormalities also could be a cause, she said. Refer a baby to a specialist if there are other signs of hyperandrogenism. However, “the likelihood is very low,” and she’s never needed to refer a neonate with acne for evaluation.
As for treatment, she said, “Mainly, I’m using tincture of time.” However, “many of my mothers have told me that topical yogurt application will work.” Why yogurt? It’s possible that its bacteria could play a role in combating acne, she said.
Masquerader alert! Beware of neonatal cephalic pustulosis, Dr. Friedlander cautioned, which may be an inflammatory response to yeast. Ketoconazole cream may be helpful.
Infantile acne (ages 0-12 months)
This form of acne is more common in males and may hint at the future development of severe adolescent acne. It does resolve but it may take months or years, Dr. Friedlander said.
In general, this acne isn’t a sign of something more serious. “You do not need to go crazy with the work-up,” she said. “With mild to moderate disease, with nothing else suspicious, I don’t do a big work-up.”
However, do consider whether the child is undergoing precocious puberty, Dr. Friedlander said. Signs include axillary hair, pubic hair, and body odor.
As for treatment of infantile acne, “start out topically” and consider options such as Bactrim (sulfamethoxazole/trimethoprim) and erythromycin.
Masquerader alert! Idiopathic facial aseptic granuloma can be mistaken for acne and abscess, and ultrasound is helpful to confirm it. “It’s not so easy to treat,” she said. “Ivermectin may be helpful. Sometimes you do cultures and make sure something else isn’t going on.”
Midchildhood (ages 1-7 years)
“It’s not as common to have acne develop in this age group, but when it develops you need to be concerned,” Dr. Friedlander said. “This is the age period when there is more often something really wrong.”
Be on the lookout for a family history of hormonal abnormalities, and check if the child is on medication. “You need to look carefully,” she said, adding that it’s important to check for signs of premature puberty such as giant spikes in growth, abnormally large hands and feet, genital changes, and body odor. Check blood pressure if you’re worried about an adrenal tumor.
It’s possible for children to develop precocious puberty – with acne – because of exposure to testosterone gel used by a father. Dehydroepiandrosterone (DHEA) creams also may cause the condition. “The more creams out there with androgenic effects, the more we may see it,” Dr. Friedlander said. “This is something to ask about because families may not be forthcoming.”
Masquerader alert! Perioral dermatitis may look like acne, and it may be linked to inhaled or topical steroids, she said.
Other masqueraders include demodex folliculitis, angiofibromas (think tuberous sclerosis), and keratosis pilaris (the most common type of bump on a children aged 1-7 years). The latter condition “is not the end of the world,” said Dr. Friedlander, who added that “I’ve never cured anyone of it.”
Prepubertal acne (ages 7 years to puberty)
Acne in this group is generally not worrisome, Dr. Friedlander said, but investigate further if there’s significant inflammation and signs of early sexual development or virilization.
Benzoyl peroxide wash may be enough to help the condition initially, and consider topical clindamycin or a combination product. “Start out slow,” she said. Twice a week to start might be appropriate. Moisturizers can be helpful, as can topical adapalene.
Also, keep in mind that even mild acne can be emotionally devastating to a child in this age group and worthy of treatment. “Your assessment may be very different than hers,” she said. It’s possible that “she has a few lesions, but she feels like an outcast.”
Dr. Friedlander reported no relevant financial disclosures. SDEF and this news organization are owned by the same parent company.
NEWPORT BEACH, CALIF. – according to Sheila Fallon Friedlander, MD.
“This is something you are going to see in your practice,” said Dr. Friedlander, a pediatric dermatologists at Rady Children’s Hospital–San Diego. It’s important to know when it’s time to be concerned and when another condition may be masquerading as acne, she said at the at Skin Disease Education Foundation’s Women’s & Pediatric Dermatology Seminar.
Dr. Friedlander, who is professor of dermatology and pediatrics at the University of California, San Diego, talked about treating acne in the following prepubertal age groups:
Neonatal acne (ages birth to 4 weeks)
Acne appears in this population up to 20% of the time, according to research, and it is much more common in males than in females, at a ratio of five to one.
The cause is “most likely the relationship between placental androgens and the baby’s adrenal glands,” Dr. Friedlander said. However, something more serious could be going on. “Look at the child and see if he’s sick. If he looks sick, then we need to worry.”
Hormonal abnormalities also could be a cause, she said. Refer a baby to a specialist if there are other signs of hyperandrogenism. However, “the likelihood is very low,” and she’s never needed to refer a neonate with acne for evaluation.
As for treatment, she said, “Mainly, I’m using tincture of time.” However, “many of my mothers have told me that topical yogurt application will work.” Why yogurt? It’s possible that its bacteria could play a role in combating acne, she said.
Masquerader alert! Beware of neonatal cephalic pustulosis, Dr. Friedlander cautioned, which may be an inflammatory response to yeast. Ketoconazole cream may be helpful.
Infantile acne (ages 0-12 months)
This form of acne is more common in males and may hint at the future development of severe adolescent acne. It does resolve but it may take months or years, Dr. Friedlander said.
In general, this acne isn’t a sign of something more serious. “You do not need to go crazy with the work-up,” she said. “With mild to moderate disease, with nothing else suspicious, I don’t do a big work-up.”
However, do consider whether the child is undergoing precocious puberty, Dr. Friedlander said. Signs include axillary hair, pubic hair, and body odor.
As for treatment of infantile acne, “start out topically” and consider options such as Bactrim (sulfamethoxazole/trimethoprim) and erythromycin.
Masquerader alert! Idiopathic facial aseptic granuloma can be mistaken for acne and abscess, and ultrasound is helpful to confirm it. “It’s not so easy to treat,” she said. “Ivermectin may be helpful. Sometimes you do cultures and make sure something else isn’t going on.”
Midchildhood (ages 1-7 years)
“It’s not as common to have acne develop in this age group, but when it develops you need to be concerned,” Dr. Friedlander said. “This is the age period when there is more often something really wrong.”
Be on the lookout for a family history of hormonal abnormalities, and check if the child is on medication. “You need to look carefully,” she said, adding that it’s important to check for signs of premature puberty such as giant spikes in growth, abnormally large hands and feet, genital changes, and body odor. Check blood pressure if you’re worried about an adrenal tumor.
It’s possible for children to develop precocious puberty – with acne – because of exposure to testosterone gel used by a father. Dehydroepiandrosterone (DHEA) creams also may cause the condition. “The more creams out there with androgenic effects, the more we may see it,” Dr. Friedlander said. “This is something to ask about because families may not be forthcoming.”
Masquerader alert! Perioral dermatitis may look like acne, and it may be linked to inhaled or topical steroids, she said.
Other masqueraders include demodex folliculitis, angiofibromas (think tuberous sclerosis), and keratosis pilaris (the most common type of bump on a children aged 1-7 years). The latter condition “is not the end of the world,” said Dr. Friedlander, who added that “I’ve never cured anyone of it.”
Prepubertal acne (ages 7 years to puberty)
Acne in this group is generally not worrisome, Dr. Friedlander said, but investigate further if there’s significant inflammation and signs of early sexual development or virilization.
Benzoyl peroxide wash may be enough to help the condition initially, and consider topical clindamycin or a combination product. “Start out slow,” she said. Twice a week to start might be appropriate. Moisturizers can be helpful, as can topical adapalene.
Also, keep in mind that even mild acne can be emotionally devastating to a child in this age group and worthy of treatment. “Your assessment may be very different than hers,” she said. It’s possible that “she has a few lesions, but she feels like an outcast.”
Dr. Friedlander reported no relevant financial disclosures. SDEF and this news organization are owned by the same parent company.
REPORTING FROM SDEF WOMEN’S & PEDIATRIC DERMATOLOGY SEMINAR
Update on Diet and Acne
Acne is a common condition that most often affects adolescents but is not uncommon in adults. It can result in considerable anxiety, depression, and medical and pharmaceutical costs. Additionally, oral antibiotics, the standard treatment for acne, are increasingly under suspicion for causing bacterial resistance as well as disruption of the cutaneous and gut microbiomes.1,2 These factors are among those that often drive patients and physicians to search for alternative and complementary treatments, including dietary modification.
Over the last few decades, the interaction between diet and acne has been one of the most fluid areas of research in dermatology. The role of diet in acne incidence and presentation has evolved from the general view in the 1970s that there was no connection to today’s more data-driven understanding that the acne disease course likely is modified by specific dietary components. Better designed and more rigorous studies have supported a link between acne severity and glycemic index (GI)/glycemic load (GL) and possibly dairy consumption. The ability to use data-driven evidence to counsel patients regarding dietary treatment of acne is increasingly important to counteract the pseudoadvice that patients can easily find on the Internet.
This article summarizes the history of beliefs about diet and acne, reviews more recent published data regarding dietary components that can modify acne severity, and outlines the current American Academy of Dermatology (AAD) guidelines and recommendations for diet and acne.
History of Diet and Acne
In most of the current literature, acne frequently is referred to as a disease of modern civilization or a consequence of the typical Western diet.3 For clarity, the Western diet is most commonly described as “a dietary regimen characterized by high amounts of sugary desserts, refined grains, high protein, high-fat dairy products, and high-sugar drinks.”4 The role of dairy in the etiology of acne typically is discussed separately from the Western diet. It has been reported that acne is not found in nonwesternized populations where a Paleolithic diet, which does not include consumption of high-GI carbohydrates, milk, or other dairy products, is common.5
Extending this line of argument, acne vulgaris has been called a metabolic syndrome of the sebaceous follicle and one of the mammalian target of rapamycin complex 1–driven diseases of civilization, along with cancer, obesity, and diabetes mellitus.3 This view seems somewhat extreme and discounts other drivers of acne incidence and severity. Twin studies have shown that acne is highly heritable, with 81% of the population variance attributed to genetic factors.6 Similar incidence numbers for acne vulgaris have been reported worldwide, and global incidence in late adolescence is rising; however, it is unknown whether this increase is a result of the adoption of the Western diet, which is thought to encourage early onset of puberty; genetic drift; changes in regional and cultural understanding and reporting of acne; or a byproduct of unknown environmental factors.4 More nuanced views acknowledge that acne is a multifactorial disease,7 and therefore genetic and possibly epigenetic factors as well as the cutaneous and gut microbiomes also must be taken into account. An interesting historical perspective on acne by Mahmood and Shipman8 outlined acne descriptions, diagnoses, topical treatments, and dietary advice going back to ancient Greek and Egyptian civilizations. They also cited recommendations from the 1930s that suggested avoiding “starchy foods, bread rolls, noodles, spaghetti, potatoes, oily nuts, chop suey, chow mein, and waffles” and listed the following foods as suitable to cure acne: “cooked and raw fruit, farina, rice, wheat, oatmeal, green vegetables, boiled or broiled meat and poultry, clear soup, vegetable soup, and an abundance of water.”8
More Recent Evidence of Dietary Influence on Acne
Importantly, the available research does not demonstrate that diet causes acne but rather that it may influence or aggravate existing acne. Data collection for acne studies also can be confounded by the interplay of many factors, such as increased access to health care, socioeconomic status, and shifting cultural perceptions of skin care and beauty.4 An important facet of any therapeutic recommendation is that it should be supported by confirmable mechanistic pathways.
GI and GL
Over the last few decades, a number of observational and intervention studies have focused on the possible influence of the GI/GL of foods on acne incidence and/or severity. A high GI diet is characterized by a relatively high intake of carbohydrate-containing foods that are quickly digested and absorbed, increasing blood glucose and insulin concentrations. Glycemic load takes the portion size of dietary carbohydrates into consideration and therefore is a measure of both the quality and quantity of carbohydrate-containing foods.9 TheGI/GL values of more than 2480 food items are available in the literature.10
Evidence from several studies supports the role of high GI/GL diets in exacerbating acne and suggests that transitioning to low GI/GL diets may lead to decreased lesion counts after 12 weeks.11-13 In one randomized controlled trial, male participants aged 15 to 25 years with mild to moderate facial acne were instructed either to eat a high protein/low GI diet or a conventional high GL control diet.13 After 12 weeks, total lesion counts had decreased more in the low GI diet group than the control. As partial confirmation of a mechanistic pathway for a high GI diet and acne, the low GI group demonstrated lower free androgen index and insulin levels than the control group.13 In a Korean study, a 10-week low GL regimen led to a reduction in acne lesion count, a decrease in sebaceous gland size, decreased inflammation, and reduced expression of sterol regulatory element-binding protein 1 and IL-8.14
More recent studies have further solidified the role of high GI/GL diets in acne severity.9,15,16 High GI/GL diets are believed to stimulate acne pathways by stimulating insulinlike growth factor 1 (IGF-1), which induces proliferation of both keratinocytes and sebocytes and simulates androgen production.17 An excellent diagram showing the connection between high GI diets (and dairy) and IGF-1, insulin and its receptors, androgen and its receptors, mammalian target of rapamycin, and the pilosebaceous unit was published in the literature in 2016.4 Interestingly, metformin has been shown to be an effective adjunctive therapy in the treatment of moderate to severe acne vulgaris.18,19
Milk and Dairy Consumption
Milk consumption also has been examined for its potential role in the pathogenesis of acne, including its ability to increase insulin and IGF-1 levels and bind to the human IGF-1 receptor as well as the fact that it contains bovine IGF-1 and dihydrotestosterone precursors.20 Although not studied quite as extensively or rigorously as GI/GL, consumption of milk and dairy products does appear to have the potential to exacerbate acne lesions. Beginning with a series of retrospective and prospective epidemiologic studies published from 2005 to 2008,21-23 a link between clinical acne and milk or dairy consumption in adolescent subjects was reported. A recent meta-analysis found a positive relationship between dairy, total milk, whole milk, low-fat milk, and skim milk consumption and acne occurrence but no significant association between yogurt/cheese consumption and acne development.24
AAD Guidelines
In their public forum, the AAD has advised that a low-glycemic diet may reduce the number of lesions in acne patients and highlighted data from around the world that support the concept that a high-glycemic diet and dairy are correlated with acne severity. They stated that consumption of milk—whole, low fat, and skim—may be linked to an increase in acne breakouts but that no studies have found that products made from milk, such as yogurt or cheese, lead to more breakouts.25
Other Considerations
Acne can be a serious quality-of-life issue with considerable psychological distress, physical morbidity, and social prejudice.9 Consequently, acne patients may be more willing to accept nonprofessional treatment advice, and there is no shortage of non–health care “experts” willing to provide an array of unfounded and fantastical advice. Dietary recommendations found online range from specific “miracle” foods to the more data-driven suggestions to “avoid dairy” or “eat low GI foods.” An important study recently published in Cutis concluded that most of the information found online regarding diet and acne is unfounded and/or misleading.26
Two additional reasons for recommending that acne patients consider dietary modification are not directly related to the disease: (1) the general health benefits of a lower GI/GL diet, and (2) the potential for decreasing the use of antibiotics. Antibiotic resistance is a growing problem across medicine, and dermatologists prescribe more antibiotics per provider than any other specialty.17 Dietary modification, where appropriate, could provide an approach to limiting the use of antibiotics in acne.
Final Thoughts
When advising acne patients, dermatologists can refer to the Table for general guidelines that incorporate the most current data-driven information on the relationship between diet and acne. Dietary modification, of course, will not work for all but can be safely recommended in cases of mild to moderate acne.

- Barbieri JS, Bhate K, Hartnett KP, et al. Trends in oral antibiotic prescription in dermatology, 2008 to 2016 [published online January 16, 2019]. JAMA Dermatol. doi:10.1001/jamadermatol.2018.4944.
- Barbieri JS, Spaccarelli N, Margolis DJ, et al. Approaches to limit systemic antibiotic use in acne: systemic alternatives, emerging topical therapies, dietary modification, and laser and light-based treatments. J Am Acad Dermatol. 2019;80:538-549.
- Melnik BC. Acne vulgaris: the metabolic syndrome of the pilosebaceous follicle [published online September 8, 2017]. Clin Dermatol. 2018;36:29-40.
- Lynn DD, Umari T, Dunnick CA, et al. The epidemiology of acne vulgaris in late adolescence. Adolesc Health Med Ther. 2016;7:13-25.
- Cordain L, Lindeberg S, Hurtado M, et al. Acne vulgaris: a disease of Western civilization. Arch Dermatol. 2002;138:1584-1590.
- Zaenglein AL, Pathy AL, Schlosser BJ, et al. Guidelines of care for the management of acne vulgaris [published online February 17, 2016]. J Am Acad Dermatol. 2016;74:945.e33-973.e33.
- Rezakovic´ S, Bukvic´ Mokos Z, Basta-Juzbašic´ A. Acne and diet: facts and controversies. Acta Dermatovenerol Croat. 2012;20:170-174.
- Mahmood NF, Shipman AR. The age-old problem of acne. Int J Womens Dermatol. 2017;3:71-76.
- Burris J, Shikany JM, Rietkerk W, et al. A low glycemic index and glycemic load diet decreases insulin-like growth factor-1 among adults with moderate and severe acne: a short-duration, 2-week randomized controlled trial. J Acad Nutr Diet. 2018;118:1874-1885.
- Atkinson FS, Foster-Powell K, Brand-Miller JC. International tables of glycemic index and glycemic load values: 2008 [published online October 3, 2008]. Diabetes Care. 2008;31:2281-2283.
- Smith RN, Braue A, Varigos GA, et al. The effect of a low glycemic load diet on acne vulgaris and the fatty acid composition of skin surface triglycerides. J Dermatol Sci. 2008;50:41-52
- Smith RN, Braue A, Varigos GA, et al. A low-glycemic-load diet improves symptoms in acne vulgaris patients: a randomized controlled trial. Am J Clin Nutr. 2007;86:107-115.
- Smith RN, Mann NJ, Braue A, et al. The effect of a high-protein, low glycemic-load diet versus a conventional, high glycemic-load diet on biochemical parameters associated with acne vulgaris: a randomized, investigator-masked, controlled trial. J Am Acad Dermatol. 2007;57:247-256.
- Kwon HH, Yoon JY, Hong JS, et al. Clinical and histological effect of a low glycaemic load diet in treatment of acne vulgaris in Korean patients: a randomized, controlled trial. Acta Derm Venereol. 2012;92:241-246.
- Burris J, Rietkerk W, Woolf K. Differences in dietary glycemic load and hormones in New York City adults with no and moderate/severe acne. J Acad Nutr Diet. 2017;117:1375-1383.
- Burris J, Rietkerk W, Woolf K. Relationships of self-reported dietary factors and perceived acne severity in a cohort of New York young adults [published online January 9, 2014]. J Acad Nutr Diet. 2014;114:384-392.
- Barbieri JS, Bhate K, Hartnett KP, et al. Trends in oral antibiotic prescription in dermatology, 2008 to 2016 [published online January 16, 2019]. JAMA Dermatol. 2019. doi:10.1001/jamadermatol.2018.4944.
- Lee JK, Smith AD. Metformin as an adjunct therapy for the treatment of moderate to severe acne vulgaris [published online November 15, 2017]. Dermatol Online J. 2017;23. pii:13030/qt53m2q13s.
- Robinson S, Kwan Z, Tang MM. Metformin as an adjunct therapy for the treatment of moderate to severe acne vulgaris: a randomized open-labeled study [published online May 1, 2019]. Dermatol Ther. 2019. doi:10.1111/dth.12953.
- Barbieri JS, Spaccarelli N, Margolis DJ, et al. Approaches to limitsystemic antibiotic use in acne: systemic alternatives, emerging topical therapies, dietary modification, and laser and light-based treatments [published online October 5, 2018]. J Am Acad Dermatol. 2019;80:538-549.
- Adebamowo CA, Spiegelman D, Berkey CS, et al. Milk consumption and acne in adolescent girls. Dermatol Online J. 2006;12:1.
- Adebamowo CA, Spiegelman D, Berkey CS, et al. Milk consumption and acne in teenaged boys. J Am Acad Dermatol. 2008;58:787-793.
- Adebamowo CA, Spiegelman D, Danby FW, et al. High school dietary dairy intake and teenage acne. J Am Acad Dermatol. 2005;52:207-214.
- Aghasi M, Golzarand M, Shab-Bidar S, et al. Dairy intake and acne development: a meta-analysis of observational studies. Clin Nutr. 2019;38:1067-1075.
- Can the right diet get rid of acne? American Academy of Dermatology website. https://www.aad.org/public/diseases/acne-and-rosacea/can-the-right-diet-get-rid-of-acne. Accessed June 13, 2019.
- Khanna R, Shifrin N, Nektalova T, et al. Diet and dermatology: Google search results for acne, psoriasis, and eczema. Cutis. 2018;102:44-46, 48.
Acne is a common condition that most often affects adolescents but is not uncommon in adults. It can result in considerable anxiety, depression, and medical and pharmaceutical costs. Additionally, oral antibiotics, the standard treatment for acne, are increasingly under suspicion for causing bacterial resistance as well as disruption of the cutaneous and gut microbiomes.1,2 These factors are among those that often drive patients and physicians to search for alternative and complementary treatments, including dietary modification.
Over the last few decades, the interaction between diet and acne has been one of the most fluid areas of research in dermatology. The role of diet in acne incidence and presentation has evolved from the general view in the 1970s that there was no connection to today’s more data-driven understanding that the acne disease course likely is modified by specific dietary components. Better designed and more rigorous studies have supported a link between acne severity and glycemic index (GI)/glycemic load (GL) and possibly dairy consumption. The ability to use data-driven evidence to counsel patients regarding dietary treatment of acne is increasingly important to counteract the pseudoadvice that patients can easily find on the Internet.
This article summarizes the history of beliefs about diet and acne, reviews more recent published data regarding dietary components that can modify acne severity, and outlines the current American Academy of Dermatology (AAD) guidelines and recommendations for diet and acne.
History of Diet and Acne
In most of the current literature, acne frequently is referred to as a disease of modern civilization or a consequence of the typical Western diet.3 For clarity, the Western diet is most commonly described as “a dietary regimen characterized by high amounts of sugary desserts, refined grains, high protein, high-fat dairy products, and high-sugar drinks.”4 The role of dairy in the etiology of acne typically is discussed separately from the Western diet. It has been reported that acne is not found in nonwesternized populations where a Paleolithic diet, which does not include consumption of high-GI carbohydrates, milk, or other dairy products, is common.5
Extending this line of argument, acne vulgaris has been called a metabolic syndrome of the sebaceous follicle and one of the mammalian target of rapamycin complex 1–driven diseases of civilization, along with cancer, obesity, and diabetes mellitus.3 This view seems somewhat extreme and discounts other drivers of acne incidence and severity. Twin studies have shown that acne is highly heritable, with 81% of the population variance attributed to genetic factors.6 Similar incidence numbers for acne vulgaris have been reported worldwide, and global incidence in late adolescence is rising; however, it is unknown whether this increase is a result of the adoption of the Western diet, which is thought to encourage early onset of puberty; genetic drift; changes in regional and cultural understanding and reporting of acne; or a byproduct of unknown environmental factors.4 More nuanced views acknowledge that acne is a multifactorial disease,7 and therefore genetic and possibly epigenetic factors as well as the cutaneous and gut microbiomes also must be taken into account. An interesting historical perspective on acne by Mahmood and Shipman8 outlined acne descriptions, diagnoses, topical treatments, and dietary advice going back to ancient Greek and Egyptian civilizations. They also cited recommendations from the 1930s that suggested avoiding “starchy foods, bread rolls, noodles, spaghetti, potatoes, oily nuts, chop suey, chow mein, and waffles” and listed the following foods as suitable to cure acne: “cooked and raw fruit, farina, rice, wheat, oatmeal, green vegetables, boiled or broiled meat and poultry, clear soup, vegetable soup, and an abundance of water.”8
More Recent Evidence of Dietary Influence on Acne
Importantly, the available research does not demonstrate that diet causes acne but rather that it may influence or aggravate existing acne. Data collection for acne studies also can be confounded by the interplay of many factors, such as increased access to health care, socioeconomic status, and shifting cultural perceptions of skin care and beauty.4 An important facet of any therapeutic recommendation is that it should be supported by confirmable mechanistic pathways.
GI and GL
Over the last few decades, a number of observational and intervention studies have focused on the possible influence of the GI/GL of foods on acne incidence and/or severity. A high GI diet is characterized by a relatively high intake of carbohydrate-containing foods that are quickly digested and absorbed, increasing blood glucose and insulin concentrations. Glycemic load takes the portion size of dietary carbohydrates into consideration and therefore is a measure of both the quality and quantity of carbohydrate-containing foods.9 TheGI/GL values of more than 2480 food items are available in the literature.10
Evidence from several studies supports the role of high GI/GL diets in exacerbating acne and suggests that transitioning to low GI/GL diets may lead to decreased lesion counts after 12 weeks.11-13 In one randomized controlled trial, male participants aged 15 to 25 years with mild to moderate facial acne were instructed either to eat a high protein/low GI diet or a conventional high GL control diet.13 After 12 weeks, total lesion counts had decreased more in the low GI diet group than the control. As partial confirmation of a mechanistic pathway for a high GI diet and acne, the low GI group demonstrated lower free androgen index and insulin levels than the control group.13 In a Korean study, a 10-week low GL regimen led to a reduction in acne lesion count, a decrease in sebaceous gland size, decreased inflammation, and reduced expression of sterol regulatory element-binding protein 1 and IL-8.14
More recent studies have further solidified the role of high GI/GL diets in acne severity.9,15,16 High GI/GL diets are believed to stimulate acne pathways by stimulating insulinlike growth factor 1 (IGF-1), which induces proliferation of both keratinocytes and sebocytes and simulates androgen production.17 An excellent diagram showing the connection between high GI diets (and dairy) and IGF-1, insulin and its receptors, androgen and its receptors, mammalian target of rapamycin, and the pilosebaceous unit was published in the literature in 2016.4 Interestingly, metformin has been shown to be an effective adjunctive therapy in the treatment of moderate to severe acne vulgaris.18,19
Milk and Dairy Consumption
Milk consumption also has been examined for its potential role in the pathogenesis of acne, including its ability to increase insulin and IGF-1 levels and bind to the human IGF-1 receptor as well as the fact that it contains bovine IGF-1 and dihydrotestosterone precursors.20 Although not studied quite as extensively or rigorously as GI/GL, consumption of milk and dairy products does appear to have the potential to exacerbate acne lesions. Beginning with a series of retrospective and prospective epidemiologic studies published from 2005 to 2008,21-23 a link between clinical acne and milk or dairy consumption in adolescent subjects was reported. A recent meta-analysis found a positive relationship between dairy, total milk, whole milk, low-fat milk, and skim milk consumption and acne occurrence but no significant association between yogurt/cheese consumption and acne development.24
AAD Guidelines
In their public forum, the AAD has advised that a low-glycemic diet may reduce the number of lesions in acne patients and highlighted data from around the world that support the concept that a high-glycemic diet and dairy are correlated with acne severity. They stated that consumption of milk—whole, low fat, and skim—may be linked to an increase in acne breakouts but that no studies have found that products made from milk, such as yogurt or cheese, lead to more breakouts.25
Other Considerations
Acne can be a serious quality-of-life issue with considerable psychological distress, physical morbidity, and social prejudice.9 Consequently, acne patients may be more willing to accept nonprofessional treatment advice, and there is no shortage of non–health care “experts” willing to provide an array of unfounded and fantastical advice. Dietary recommendations found online range from specific “miracle” foods to the more data-driven suggestions to “avoid dairy” or “eat low GI foods.” An important study recently published in Cutis concluded that most of the information found online regarding diet and acne is unfounded and/or misleading.26
Two additional reasons for recommending that acne patients consider dietary modification are not directly related to the disease: (1) the general health benefits of a lower GI/GL diet, and (2) the potential for decreasing the use of antibiotics. Antibiotic resistance is a growing problem across medicine, and dermatologists prescribe more antibiotics per provider than any other specialty.17 Dietary modification, where appropriate, could provide an approach to limiting the use of antibiotics in acne.
Final Thoughts
When advising acne patients, dermatologists can refer to the Table for general guidelines that incorporate the most current data-driven information on the relationship between diet and acne. Dietary modification, of course, will not work for all but can be safely recommended in cases of mild to moderate acne.

Acne is a common condition that most often affects adolescents but is not uncommon in adults. It can result in considerable anxiety, depression, and medical and pharmaceutical costs. Additionally, oral antibiotics, the standard treatment for acne, are increasingly under suspicion for causing bacterial resistance as well as disruption of the cutaneous and gut microbiomes.1,2 These factors are among those that often drive patients and physicians to search for alternative and complementary treatments, including dietary modification.
Over the last few decades, the interaction between diet and acne has been one of the most fluid areas of research in dermatology. The role of diet in acne incidence and presentation has evolved from the general view in the 1970s that there was no connection to today’s more data-driven understanding that the acne disease course likely is modified by specific dietary components. Better designed and more rigorous studies have supported a link between acne severity and glycemic index (GI)/glycemic load (GL) and possibly dairy consumption. The ability to use data-driven evidence to counsel patients regarding dietary treatment of acne is increasingly important to counteract the pseudoadvice that patients can easily find on the Internet.
This article summarizes the history of beliefs about diet and acne, reviews more recent published data regarding dietary components that can modify acne severity, and outlines the current American Academy of Dermatology (AAD) guidelines and recommendations for diet and acne.
History of Diet and Acne
In most of the current literature, acne frequently is referred to as a disease of modern civilization or a consequence of the typical Western diet.3 For clarity, the Western diet is most commonly described as “a dietary regimen characterized by high amounts of sugary desserts, refined grains, high protein, high-fat dairy products, and high-sugar drinks.”4 The role of dairy in the etiology of acne typically is discussed separately from the Western diet. It has been reported that acne is not found in nonwesternized populations where a Paleolithic diet, which does not include consumption of high-GI carbohydrates, milk, or other dairy products, is common.5
Extending this line of argument, acne vulgaris has been called a metabolic syndrome of the sebaceous follicle and one of the mammalian target of rapamycin complex 1–driven diseases of civilization, along with cancer, obesity, and diabetes mellitus.3 This view seems somewhat extreme and discounts other drivers of acne incidence and severity. Twin studies have shown that acne is highly heritable, with 81% of the population variance attributed to genetic factors.6 Similar incidence numbers for acne vulgaris have been reported worldwide, and global incidence in late adolescence is rising; however, it is unknown whether this increase is a result of the adoption of the Western diet, which is thought to encourage early onset of puberty; genetic drift; changes in regional and cultural understanding and reporting of acne; or a byproduct of unknown environmental factors.4 More nuanced views acknowledge that acne is a multifactorial disease,7 and therefore genetic and possibly epigenetic factors as well as the cutaneous and gut microbiomes also must be taken into account. An interesting historical perspective on acne by Mahmood and Shipman8 outlined acne descriptions, diagnoses, topical treatments, and dietary advice going back to ancient Greek and Egyptian civilizations. They also cited recommendations from the 1930s that suggested avoiding “starchy foods, bread rolls, noodles, spaghetti, potatoes, oily nuts, chop suey, chow mein, and waffles” and listed the following foods as suitable to cure acne: “cooked and raw fruit, farina, rice, wheat, oatmeal, green vegetables, boiled or broiled meat and poultry, clear soup, vegetable soup, and an abundance of water.”8
More Recent Evidence of Dietary Influence on Acne
Importantly, the available research does not demonstrate that diet causes acne but rather that it may influence or aggravate existing acne. Data collection for acne studies also can be confounded by the interplay of many factors, such as increased access to health care, socioeconomic status, and shifting cultural perceptions of skin care and beauty.4 An important facet of any therapeutic recommendation is that it should be supported by confirmable mechanistic pathways.
GI and GL
Over the last few decades, a number of observational and intervention studies have focused on the possible influence of the GI/GL of foods on acne incidence and/or severity. A high GI diet is characterized by a relatively high intake of carbohydrate-containing foods that are quickly digested and absorbed, increasing blood glucose and insulin concentrations. Glycemic load takes the portion size of dietary carbohydrates into consideration and therefore is a measure of both the quality and quantity of carbohydrate-containing foods.9 TheGI/GL values of more than 2480 food items are available in the literature.10
Evidence from several studies supports the role of high GI/GL diets in exacerbating acne and suggests that transitioning to low GI/GL diets may lead to decreased lesion counts after 12 weeks.11-13 In one randomized controlled trial, male participants aged 15 to 25 years with mild to moderate facial acne were instructed either to eat a high protein/low GI diet or a conventional high GL control diet.13 After 12 weeks, total lesion counts had decreased more in the low GI diet group than the control. As partial confirmation of a mechanistic pathway for a high GI diet and acne, the low GI group demonstrated lower free androgen index and insulin levels than the control group.13 In a Korean study, a 10-week low GL regimen led to a reduction in acne lesion count, a decrease in sebaceous gland size, decreased inflammation, and reduced expression of sterol regulatory element-binding protein 1 and IL-8.14
More recent studies have further solidified the role of high GI/GL diets in acne severity.9,15,16 High GI/GL diets are believed to stimulate acne pathways by stimulating insulinlike growth factor 1 (IGF-1), which induces proliferation of both keratinocytes and sebocytes and simulates androgen production.17 An excellent diagram showing the connection between high GI diets (and dairy) and IGF-1, insulin and its receptors, androgen and its receptors, mammalian target of rapamycin, and the pilosebaceous unit was published in the literature in 2016.4 Interestingly, metformin has been shown to be an effective adjunctive therapy in the treatment of moderate to severe acne vulgaris.18,19
Milk and Dairy Consumption
Milk consumption also has been examined for its potential role in the pathogenesis of acne, including its ability to increase insulin and IGF-1 levels and bind to the human IGF-1 receptor as well as the fact that it contains bovine IGF-1 and dihydrotestosterone precursors.20 Although not studied quite as extensively or rigorously as GI/GL, consumption of milk and dairy products does appear to have the potential to exacerbate acne lesions. Beginning with a series of retrospective and prospective epidemiologic studies published from 2005 to 2008,21-23 a link between clinical acne and milk or dairy consumption in adolescent subjects was reported. A recent meta-analysis found a positive relationship between dairy, total milk, whole milk, low-fat milk, and skim milk consumption and acne occurrence but no significant association between yogurt/cheese consumption and acne development.24
AAD Guidelines
In their public forum, the AAD has advised that a low-glycemic diet may reduce the number of lesions in acne patients and highlighted data from around the world that support the concept that a high-glycemic diet and dairy are correlated with acne severity. They stated that consumption of milk—whole, low fat, and skim—may be linked to an increase in acne breakouts but that no studies have found that products made from milk, such as yogurt or cheese, lead to more breakouts.25
Other Considerations
Acne can be a serious quality-of-life issue with considerable psychological distress, physical morbidity, and social prejudice.9 Consequently, acne patients may be more willing to accept nonprofessional treatment advice, and there is no shortage of non–health care “experts” willing to provide an array of unfounded and fantastical advice. Dietary recommendations found online range from specific “miracle” foods to the more data-driven suggestions to “avoid dairy” or “eat low GI foods.” An important study recently published in Cutis concluded that most of the information found online regarding diet and acne is unfounded and/or misleading.26
Two additional reasons for recommending that acne patients consider dietary modification are not directly related to the disease: (1) the general health benefits of a lower GI/GL diet, and (2) the potential for decreasing the use of antibiotics. Antibiotic resistance is a growing problem across medicine, and dermatologists prescribe more antibiotics per provider than any other specialty.17 Dietary modification, where appropriate, could provide an approach to limiting the use of antibiotics in acne.
Final Thoughts
When advising acne patients, dermatologists can refer to the Table for general guidelines that incorporate the most current data-driven information on the relationship between diet and acne. Dietary modification, of course, will not work for all but can be safely recommended in cases of mild to moderate acne.

- Barbieri JS, Bhate K, Hartnett KP, et al. Trends in oral antibiotic prescription in dermatology, 2008 to 2016 [published online January 16, 2019]. JAMA Dermatol. doi:10.1001/jamadermatol.2018.4944.
- Barbieri JS, Spaccarelli N, Margolis DJ, et al. Approaches to limit systemic antibiotic use in acne: systemic alternatives, emerging topical therapies, dietary modification, and laser and light-based treatments. J Am Acad Dermatol. 2019;80:538-549.
- Melnik BC. Acne vulgaris: the metabolic syndrome of the pilosebaceous follicle [published online September 8, 2017]. Clin Dermatol. 2018;36:29-40.
- Lynn DD, Umari T, Dunnick CA, et al. The epidemiology of acne vulgaris in late adolescence. Adolesc Health Med Ther. 2016;7:13-25.
- Cordain L, Lindeberg S, Hurtado M, et al. Acne vulgaris: a disease of Western civilization. Arch Dermatol. 2002;138:1584-1590.
- Zaenglein AL, Pathy AL, Schlosser BJ, et al. Guidelines of care for the management of acne vulgaris [published online February 17, 2016]. J Am Acad Dermatol. 2016;74:945.e33-973.e33.
- Rezakovic´ S, Bukvic´ Mokos Z, Basta-Juzbašic´ A. Acne and diet: facts and controversies. Acta Dermatovenerol Croat. 2012;20:170-174.
- Mahmood NF, Shipman AR. The age-old problem of acne. Int J Womens Dermatol. 2017;3:71-76.
- Burris J, Shikany JM, Rietkerk W, et al. A low glycemic index and glycemic load diet decreases insulin-like growth factor-1 among adults with moderate and severe acne: a short-duration, 2-week randomized controlled trial. J Acad Nutr Diet. 2018;118:1874-1885.
- Atkinson FS, Foster-Powell K, Brand-Miller JC. International tables of glycemic index and glycemic load values: 2008 [published online October 3, 2008]. Diabetes Care. 2008;31:2281-2283.
- Smith RN, Braue A, Varigos GA, et al. The effect of a low glycemic load diet on acne vulgaris and the fatty acid composition of skin surface triglycerides. J Dermatol Sci. 2008;50:41-52
- Smith RN, Braue A, Varigos GA, et al. A low-glycemic-load diet improves symptoms in acne vulgaris patients: a randomized controlled trial. Am J Clin Nutr. 2007;86:107-115.
- Smith RN, Mann NJ, Braue A, et al. The effect of a high-protein, low glycemic-load diet versus a conventional, high glycemic-load diet on biochemical parameters associated with acne vulgaris: a randomized, investigator-masked, controlled trial. J Am Acad Dermatol. 2007;57:247-256.
- Kwon HH, Yoon JY, Hong JS, et al. Clinical and histological effect of a low glycaemic load diet in treatment of acne vulgaris in Korean patients: a randomized, controlled trial. Acta Derm Venereol. 2012;92:241-246.
- Burris J, Rietkerk W, Woolf K. Differences in dietary glycemic load and hormones in New York City adults with no and moderate/severe acne. J Acad Nutr Diet. 2017;117:1375-1383.
- Burris J, Rietkerk W, Woolf K. Relationships of self-reported dietary factors and perceived acne severity in a cohort of New York young adults [published online January 9, 2014]. J Acad Nutr Diet. 2014;114:384-392.
- Barbieri JS, Bhate K, Hartnett KP, et al. Trends in oral antibiotic prescription in dermatology, 2008 to 2016 [published online January 16, 2019]. JAMA Dermatol. 2019. doi:10.1001/jamadermatol.2018.4944.
- Lee JK, Smith AD. Metformin as an adjunct therapy for the treatment of moderate to severe acne vulgaris [published online November 15, 2017]. Dermatol Online J. 2017;23. pii:13030/qt53m2q13s.
- Robinson S, Kwan Z, Tang MM. Metformin as an adjunct therapy for the treatment of moderate to severe acne vulgaris: a randomized open-labeled study [published online May 1, 2019]. Dermatol Ther. 2019. doi:10.1111/dth.12953.
- Barbieri JS, Spaccarelli N, Margolis DJ, et al. Approaches to limitsystemic antibiotic use in acne: systemic alternatives, emerging topical therapies, dietary modification, and laser and light-based treatments [published online October 5, 2018]. J Am Acad Dermatol. 2019;80:538-549.
- Adebamowo CA, Spiegelman D, Berkey CS, et al. Milk consumption and acne in adolescent girls. Dermatol Online J. 2006;12:1.
- Adebamowo CA, Spiegelman D, Berkey CS, et al. Milk consumption and acne in teenaged boys. J Am Acad Dermatol. 2008;58:787-793.
- Adebamowo CA, Spiegelman D, Danby FW, et al. High school dietary dairy intake and teenage acne. J Am Acad Dermatol. 2005;52:207-214.
- Aghasi M, Golzarand M, Shab-Bidar S, et al. Dairy intake and acne development: a meta-analysis of observational studies. Clin Nutr. 2019;38:1067-1075.
- Can the right diet get rid of acne? American Academy of Dermatology website. https://www.aad.org/public/diseases/acne-and-rosacea/can-the-right-diet-get-rid-of-acne. Accessed June 13, 2019.
- Khanna R, Shifrin N, Nektalova T, et al. Diet and dermatology: Google search results for acne, psoriasis, and eczema. Cutis. 2018;102:44-46, 48.
- Barbieri JS, Bhate K, Hartnett KP, et al. Trends in oral antibiotic prescription in dermatology, 2008 to 2016 [published online January 16, 2019]. JAMA Dermatol. doi:10.1001/jamadermatol.2018.4944.
- Barbieri JS, Spaccarelli N, Margolis DJ, et al. Approaches to limit systemic antibiotic use in acne: systemic alternatives, emerging topical therapies, dietary modification, and laser and light-based treatments. J Am Acad Dermatol. 2019;80:538-549.
- Melnik BC. Acne vulgaris: the metabolic syndrome of the pilosebaceous follicle [published online September 8, 2017]. Clin Dermatol. 2018;36:29-40.
- Lynn DD, Umari T, Dunnick CA, et al. The epidemiology of acne vulgaris in late adolescence. Adolesc Health Med Ther. 2016;7:13-25.
- Cordain L, Lindeberg S, Hurtado M, et al. Acne vulgaris: a disease of Western civilization. Arch Dermatol. 2002;138:1584-1590.
- Zaenglein AL, Pathy AL, Schlosser BJ, et al. Guidelines of care for the management of acne vulgaris [published online February 17, 2016]. J Am Acad Dermatol. 2016;74:945.e33-973.e33.
- Rezakovic´ S, Bukvic´ Mokos Z, Basta-Juzbašic´ A. Acne and diet: facts and controversies. Acta Dermatovenerol Croat. 2012;20:170-174.
- Mahmood NF, Shipman AR. The age-old problem of acne. Int J Womens Dermatol. 2017;3:71-76.
- Burris J, Shikany JM, Rietkerk W, et al. A low glycemic index and glycemic load diet decreases insulin-like growth factor-1 among adults with moderate and severe acne: a short-duration, 2-week randomized controlled trial. J Acad Nutr Diet. 2018;118:1874-1885.
- Atkinson FS, Foster-Powell K, Brand-Miller JC. International tables of glycemic index and glycemic load values: 2008 [published online October 3, 2008]. Diabetes Care. 2008;31:2281-2283.
- Smith RN, Braue A, Varigos GA, et al. The effect of a low glycemic load diet on acne vulgaris and the fatty acid composition of skin surface triglycerides. J Dermatol Sci. 2008;50:41-52
- Smith RN, Braue A, Varigos GA, et al. A low-glycemic-load diet improves symptoms in acne vulgaris patients: a randomized controlled trial. Am J Clin Nutr. 2007;86:107-115.
- Smith RN, Mann NJ, Braue A, et al. The effect of a high-protein, low glycemic-load diet versus a conventional, high glycemic-load diet on biochemical parameters associated with acne vulgaris: a randomized, investigator-masked, controlled trial. J Am Acad Dermatol. 2007;57:247-256.
- Kwon HH, Yoon JY, Hong JS, et al. Clinical and histological effect of a low glycaemic load diet in treatment of acne vulgaris in Korean patients: a randomized, controlled trial. Acta Derm Venereol. 2012;92:241-246.
- Burris J, Rietkerk W, Woolf K. Differences in dietary glycemic load and hormones in New York City adults with no and moderate/severe acne. J Acad Nutr Diet. 2017;117:1375-1383.
- Burris J, Rietkerk W, Woolf K. Relationships of self-reported dietary factors and perceived acne severity in a cohort of New York young adults [published online January 9, 2014]. J Acad Nutr Diet. 2014;114:384-392.
- Barbieri JS, Bhate K, Hartnett KP, et al. Trends in oral antibiotic prescription in dermatology, 2008 to 2016 [published online January 16, 2019]. JAMA Dermatol. 2019. doi:10.1001/jamadermatol.2018.4944.
- Lee JK, Smith AD. Metformin as an adjunct therapy for the treatment of moderate to severe acne vulgaris [published online November 15, 2017]. Dermatol Online J. 2017;23. pii:13030/qt53m2q13s.
- Robinson S, Kwan Z, Tang MM. Metformin as an adjunct therapy for the treatment of moderate to severe acne vulgaris: a randomized open-labeled study [published online May 1, 2019]. Dermatol Ther. 2019. doi:10.1111/dth.12953.
- Barbieri JS, Spaccarelli N, Margolis DJ, et al. Approaches to limitsystemic antibiotic use in acne: systemic alternatives, emerging topical therapies, dietary modification, and laser and light-based treatments [published online October 5, 2018]. J Am Acad Dermatol. 2019;80:538-549.
- Adebamowo CA, Spiegelman D, Berkey CS, et al. Milk consumption and acne in adolescent girls. Dermatol Online J. 2006;12:1.
- Adebamowo CA, Spiegelman D, Berkey CS, et al. Milk consumption and acne in teenaged boys. J Am Acad Dermatol. 2008;58:787-793.
- Adebamowo CA, Spiegelman D, Danby FW, et al. High school dietary dairy intake and teenage acne. J Am Acad Dermatol. 2005;52:207-214.
- Aghasi M, Golzarand M, Shab-Bidar S, et al. Dairy intake and acne development: a meta-analysis of observational studies. Clin Nutr. 2019;38:1067-1075.
- Can the right diet get rid of acne? American Academy of Dermatology website. https://www.aad.org/public/diseases/acne-and-rosacea/can-the-right-diet-get-rid-of-acne. Accessed June 13, 2019.
- Khanna R, Shifrin N, Nektalova T, et al. Diet and dermatology: Google search results for acne, psoriasis, and eczema. Cutis. 2018;102:44-46, 48.
The Role of Adolescent Acne Treatment in Formation of Scars Among Patients With Persistent Adult Acne: Evidence From an Observational Study
In the last 20 years, the incidence of acne lesions in adults has markedly increased. 1 Acne affects adults (individuals older than 25 years) and is no longer a condition limited to adolescents and young adults (individuals younger than 25 years). According to Dreno et al, 2 the accepted age threshold for the onset of adult acne is 25 years. 1-3 In 2013, the term adult acne was defined. 2 Among patients with adult acne, there are 2 subtypes: (1) persistent adult acne, which is a continuation or recurrence of adolescent acne, affecting approximately 80% of patients, and (2) late-onset acne, affecting approximately 20% of patients. 4
Clinical symptoms of adult acne and available treatment modalities have been explored in the literature. Daily clinical experience shows that additional difficulties involved in the management of adult acne patients are related mainly to a high therapeutic failure rate in acne patients older than 25 years. 5 Persistent adult acne seems to be noteworthy because it causes long-term symptoms, and patients experience uncontrollable recurrences.
It is believed that adult acne often is resistant to treatment. 2 Adult skin is more sensitive to topical agents, leading to more irritation by medications intended for external use and cosmetics. 6 Scars in these patients are a frequent and undesirable consequence. 3
Effective treatment of acne encompasses oral antibiotics, topical and systemic retinoids, and oral contraceptive pills (OCPs). For years, oral subantimicrobial doses of cyclines have been recommended for acne treatment. Topical and oral retinoids have been successfully used for more than 30 years as important therapeutic options. 7 More recent evidence-based guidelines for acne issued by the American Academy of Dermatology 8 and the European Dermatology Forum 9 also show that retinoids play an important role in acne therapy. Their anti-inflammatory activity acts against comedones and their precursors (microcomedones). Successful antiacne therapy not only achieves a smooth face without comedones but also minimizes scar formation, postinflammatory discoloration, and long-lasting postinflammatory erythema. 10 Oral contraceptives have a mainly antiseborrheic effect. 11
Our study sought to analyze the potential influence of therapy during adolescent acne on patients who later developed adult acne. Particular attention was given to the use of oral antibiotics, isotretinoin, and topical retinoids for adolescent acne and their potential role in diminishing scar formation in adult acne.
Materials and Methods
Patient Demographics and Selection
A population-based study of Polish patients with adult acne was conducted. Patients were included in the study group on a consecutive basis from among those who visited our outpatient dermatology center from May 2015 to January 2016. A total of 111 patients (101 women [90.99%] and 10 men [9.01%]) were examined. The study group comprised patients aged 25 years and older who were treated for adult acne (20 patients [18.02%] were aged 25–29 years, 61 [54.95%] were aged 30–39 years, and 30 [27.02%] were 40 years or older).
The following inclusion criteria were used: observation period of at least 6 months in our dermatologic center for patients diagnosed with adult acne, at least 2 dermatologic visits for adult acne prior to the study, written informed consent for study participation and data processing (the aim of the study was explained to each participant by a dermatologist), and age 25 years or older. Exclusion criteria included those who were younger than 25 years, those who had only 1 dermatologic visit at our dermatology center, and those who were unwilling to participate or did not provide written informed consent. Our study was conducted according to Good Clinical Practice.
Data Collection
To obtain data with the highest degree of reliability, 3 sources of information were used: (1) a detailed medical interview conducted by one experienced dermatologist (E.C.) at our dermatology center at the first visit in all study participants, (2) a clinical examination that yielded results necessary for the assessment of scars using a method outlined by Jacob et al, 12 and (3) information included in available medical records. These data were then statistically analyzed.
Statistical Analysis
The results were presented as frequency plots, and a Fisher exact test was conducted to obtain a statistical comparison of the distributions of analyzed data. Unless otherwise indicated, 5% was adopted as the significance level. The statistical analysis was performed using Stata 14 software (StataCorp LLC, College Station, Texas).
Results
Incidence of Different Forms of Adult Acne
To analyze the onset of acne, patients were categorized into 1 of 2 groups: those with persistent adult acne (81.98%) and those with late-onset adult acne (ie, developed after 25 years of age)(18.02%).
Age at Initiation of Dermatologic Treatment
Of the patients with persistent adult acne, 31.87% first visited a dermatologist the same year that the first acne lesions appeared, 36.26% postponed the first visit by at least 5 years (Figure 1), and 23.08% started treatment at least 10 years after acne first appeared. Among patients with persistent adult acne, 76.92% began dermatologic treatment before 25 years of age, and 23.08% began treatment after 25 years of age. Of the latter, 28.57% did not start therapy until they were older than 35 years.

Severity of Adolescent Acne
In the persistent adult acne group, the severity of adolescent acne was assessed during the medical interview as well as detailed histories in medical records. The activity of acne was evaluated at 2-year intervals with the use of a 10-point scale: 1 to 3 points indicated mild acne (7.69% of patients), 4 to 6 points indicated moderate acne (24.18%), and 7 to 10 points indicated severe acne (68.13%).
Treatment of Persistent Acne in Adolescence
Treatment was comprised of oral therapy with antibiotics, isotretinoin, and/or application of topical retinoids (sometimes supported with OCPs). Monotherapy was the standard of treatment more than 25 years ago when patients with persistent adult acne were treated as adolescents or young adults. As many as 43.96% of patients with persistent adult acne did not receive any of these therapies before 25 years of age; rather, they used antiacne cosmetics or beauty procedures. Furthermore, 50.55% of patients were treated with oral antibiotics (Figure 2). Topical retinoids were used in 19.78% of patients and isotretinoin was used in 16.48%. Incidentally, OCPs were given to 26.5%. In the course of adolescent acne, 31.87% of patients received 2 to 4 courses of treatment with either antibiotics or retinoids (oral or topical), and 5.49% were treated with 5 or more courses of treatment (Figure 3). The analysis of each treatment revealed that only 1 patient received 4 courses of isotretinoin. Five courses of oral antibiotics were given in 1 patient, and 3 courses of topical retinoids were given in the same patient.


Topical Retinoids
In an analysis of the number of treatments with topical retinoids completed by patients with persistent adult acne, it was established that 80.22% of patients never used topical retinoids for acne during adolescence. Additionally, 12.08% of these patients completed 1 course of treatment, and 7.69% completed 2 to 4 treatments. However, after 25 years of age, only 25.27% of the patients with persistent adult acne were not treated with topical retinoids, and 35.16% completed more than 2 courses of treatment.
Duration of Treatment
Because adult acne is a chronic disease, the mean number of years that patients received treatment over the disease course was analyzed. In the case of persistent adult acne, the mean duration of treatment, including therapy received during adolescence, was more than 13 years. At the time of the study, more than 30% of patients had been undergoing treatment of adult acne for more than 20 years. Scars— The proportion of patients with persistent adult acne who experienced scarring was evaluated. In the persistent adult acne group, scars were identified in 53.85% of patients. Scars appeared only during adolescence in 26.37% of patients with persistent adult acne, scars appeared only after 25 years of age in 21.97% of patients, and scars appeared in adolescence as well as adulthood in 30.77% of patients.
In an analysis of patients with persistent adult acne who experienced scarring after 25 years of age, the proportion of patients with untreated adolescent acne and those who were treated with antibiotics only was not significantly different (60% vs 64%; P = .478)(Table). The inclusion of topical retinoids into treatment decreased the proportion of scars (isotretinoin: 20%, P = .009; topical retinoids: 38.89%, P = .114).

Comment
Persistent Adult Acne
Patients with symptoms of persistent adult acne represented 81.98% of the study population, which was similar to a 1999 study by Goulden et al, 1 a 2001 study by Shaw and White, 13 and a 2009 report by Schmidt et al. 14 Of these patients with persistent adult acne, 23.08% initiated therapy after 25 years of age, and 23.08% started treatment at least 10 years after acne lesions first appeared. However, it is noteworthy that 68.13% of all patients with persistent adult acne assessed their disease as severe.
Treatment Modalities for Adult Acne
Over the last 5 years, some researchers have attempted to make recommendations for the treatment of adult acne based on standards adopted for the treatment of adolescent acne. 2,9,15 First-line treatment of patients with adult comedonal acne is topical retinoids. 9 The recommended treatment of mild to moderate adult inflammatory acne involves topical drugs, including retinoids, azelaic acid, or benzoyl peroxide, or oral medications, including antibiotics, OCPs, or antiandrogens. In severe inflammatory acne, the recommended treatment involves oral isotretinoin or combined therapies; the latter seems to be the most effective. 16 Furthermore, this therapy has been adjusted to the patient’s current clinical condition; general individual sensitivity of the skin to irritation and the risk for irritant activity of topical medications; and life situation, such as planned pregnancies and intended use of OCPs due to the risk for teratogenic effects of drugs. 17
To assess available treatment modalities, oral therapy with antibiotics or isotretinoin as well as topical retinoids were selected for our analysis. It is difficult to determine an exclusive impact of OCPs as acne treatment; according to our study, many female patients use hormone therapy for other medical conditions or contraception, and only a small proportion of these patients are prescribed hormone treatment for acne. We found that 43.96% of patients with persistent adult acne underwent no treatment with antibiotics, isotretinoin, or topical retinoids in adolescence. Patients who did not receive any of these treatments came only for single visits to a dermatologist, did not comply to a recommended therapy, or used only cosmetics or beauty procedures. We found that 80.22% of patients with persistent adult acne never used topical retinoids during adolescence and did not receive maintenance therapy, which may be attributed to the fact that there were no strict recommendations regarding retinoid treatment when these patients were adolescents or young adults. Published data indicate that retinoid use for acne treatment is not common. 18 Conversely, among patients older than 25 years with late-onset adult acne, there was only 1 patient (ie, < 1%) who had never received any oral antibiotic or isotretinoin treatment or therapy with topical retinoids. The reason for the lack of medical treatment is unknown. Only 25.27% of patients were not treated with topical retinoids, and 35.16% completed at least 2 courses of treatment.
Acne Scarring
The worst complication of acne is scarring. Scars develop for the duration of the disease, during both adolescent and adult acne. In the group with persistent adult acne, scarring was found in 53.85% of patients. Scar formation has been previously reported as a common complication of acne. 19 The effects of skin lesions that remain after acne are not only limited to impaired cosmetic appearance; they also negatively affect mental health and impair quality of life. 20 The aim of our study was to analyze types of treatment for adolescent acne in patients who later had persistent adult acne. Postacne scars observed later are objective evidence of the severity of disease. We found that using oral antibiotics did not diminish the number of scars among persistent adult acne patients in adulthood. In contrast, isotretinoin or topical retinoid treatment during adolescence decreased the risk for scars occurring during adulthood. In our opinion, these findings emphasize the role of this type of treatment among adolescents or young adults. The decrease of scar formation in adult acne due to retinoid treatment in adolescence indirectly justifies the role of maintenance therapy with topical retinoids. 21,22
- Goulden V, Stables GI, Cunliffe WJ. Prevalence of facial acne in adults. J Am Acad Dermatol. 1999;41:577-580.
- Dreno B, Layton A, Zouboulis CC, et al. Adult female acne: a new paradigm. J Eur Acad Dermatol Venereol. 2013;27:1063-1070.
- Preneau S, Dreno B. Female acne--a different subtype of teenager acne? J Eur Acad Dermatol Venereol. 2012;26:277-282.
- Goulden V, Clark SM, Cunliffe WJ. Post-adolescent acne: a review of clinical features. Br J Dermatol. 1997;136:66-70.
- Kamangar F, Shinkai K. Acne in the adult female patient: a practical approach. Int J Dermatol. 2012;51:1162-1174.
- Choi CW, Lee DH, Kim HS, et al. The clinical features of late onset acne compared with early onset acne in women. J Eur Acad Dermatol Venereol. 2011;25:454-461.
- Kligman AM, Fulton JE Jr, Plewig G. Topical vitamin A acid in acne vulgaris. Arch Dermatol. 1969;99:469-476.
- Zaenglein AL, Pathy AL, Schlosser BJ, et al. Guidelines of care for the management of acne vulgaris. J Am Acad Dermatol. 2016;74:945.e33-973.e33.
- Nast A, Dreno B, Bettoli V, et al. European evidence-based guidelines for the treatment of acne. J Eur Acad Dermatol Venereol. 2012;26(suppl 1):1-29.
- Levin J. The relationship of proper skin cleansing to pathophysiology, clinical benefits, and the concomitant use of prescription topical therapies in patients with acne vulgaris. Dermatol Clin. 2016;34:133-145.
- Savage LJ, Layton AM. Treating acne vulgaris: systemic, local and combination therapy. Expert Rev Clin Pharmacol. 2010;3:563-580.
- Jacob CL, Dover JS, Kaminer MS. Acne scarring: a classification system and review of treatment options. J Am Acad Dermatol. 2001;45:109-117.
- Shaw JC, White LE. Persistent acne in adult women. Arch Dermatol. 2001;137:1252-1253.
- Schmidt JV, Masuda PY, Miot HA. Acne in women: clinical patterns in different age groups. An Bras Dermatol. 2009;84:349-354.
- Thiboutot D, Gollnick H, Bettoli V, et al. New insights into the management of acne: an update from the Global Alliance to Improve Outcomes in Acne group. J Am Acad Dermatol. 2009;60(5 suppl):1-50.
- Williams C, Layton AM. Persistent acne in women: implications for the patient and for therapy. Am J Clin Dermatol. 2006;7:281-290.
- Holzmann R, Shakery K. Postadolescent acne in females. Skin Pharmacol Physiol. 2014;27(suppl 1):3-8.
- Pena S, Hill D, Feldman SR. Use of topical retinoids by dermatologist and non-dermatologist in the management of acne vulgaris. J Am Acad Dermatol. 2016;74:1252-1254.
- Layton AM, Henderson CA, Cunliffe WJ. A clinical evaluation of acne scarring and its incidence. Clin Exp Dermatol. 1994;19;303-308.
- Halvorsen JA, Stern RS, Dalgard F, et al. Suicidal ideation, mental health problems, and social impairment are increased in adolescents with acne: a population-based study. J Invest Dermatol. 2011;131:363-370.
- Thielitz A, Sidou F, Gollnick H. Control of microcomedone formation throughout a maintenance treatment with adapalene gel, 0.1%. J Eur Acad Dermatol Venereol. 2007;21:747-753.
- Leyden J, Thiboutot DM, Shalita R, et al. Comparison of tazarotene and minocycline maintenance therapies in acne vulgaris: a multicenter, double-blind, randomized, parallel-group study. Arch Dermatol. 2006;142:605-612.
In the last 20 years, the incidence of acne lesions in adults has markedly increased. 1 Acne affects adults (individuals older than 25 years) and is no longer a condition limited to adolescents and young adults (individuals younger than 25 years). According to Dreno et al, 2 the accepted age threshold for the onset of adult acne is 25 years. 1-3 In 2013, the term adult acne was defined. 2 Among patients with adult acne, there are 2 subtypes: (1) persistent adult acne, which is a continuation or recurrence of adolescent acne, affecting approximately 80% of patients, and (2) late-onset acne, affecting approximately 20% of patients. 4
Clinical symptoms of adult acne and available treatment modalities have been explored in the literature. Daily clinical experience shows that additional difficulties involved in the management of adult acne patients are related mainly to a high therapeutic failure rate in acne patients older than 25 years. 5 Persistent adult acne seems to be noteworthy because it causes long-term symptoms, and patients experience uncontrollable recurrences.
It is believed that adult acne often is resistant to treatment. 2 Adult skin is more sensitive to topical agents, leading to more irritation by medications intended for external use and cosmetics. 6 Scars in these patients are a frequent and undesirable consequence. 3
Effective treatment of acne encompasses oral antibiotics, topical and systemic retinoids, and oral contraceptive pills (OCPs). For years, oral subantimicrobial doses of cyclines have been recommended for acne treatment. Topical and oral retinoids have been successfully used for more than 30 years as important therapeutic options. 7 More recent evidence-based guidelines for acne issued by the American Academy of Dermatology 8 and the European Dermatology Forum 9 also show that retinoids play an important role in acne therapy. Their anti-inflammatory activity acts against comedones and their precursors (microcomedones). Successful antiacne therapy not only achieves a smooth face without comedones but also minimizes scar formation, postinflammatory discoloration, and long-lasting postinflammatory erythema. 10 Oral contraceptives have a mainly antiseborrheic effect. 11
Our study sought to analyze the potential influence of therapy during adolescent acne on patients who later developed adult acne. Particular attention was given to the use of oral antibiotics, isotretinoin, and topical retinoids for adolescent acne and their potential role in diminishing scar formation in adult acne.
Materials and Methods
Patient Demographics and Selection
A population-based study of Polish patients with adult acne was conducted. Patients were included in the study group on a consecutive basis from among those who visited our outpatient dermatology center from May 2015 to January 2016. A total of 111 patients (101 women [90.99%] and 10 men [9.01%]) were examined. The study group comprised patients aged 25 years and older who were treated for adult acne (20 patients [18.02%] were aged 25–29 years, 61 [54.95%] were aged 30–39 years, and 30 [27.02%] were 40 years or older).
The following inclusion criteria were used: observation period of at least 6 months in our dermatologic center for patients diagnosed with adult acne, at least 2 dermatologic visits for adult acne prior to the study, written informed consent for study participation and data processing (the aim of the study was explained to each participant by a dermatologist), and age 25 years or older. Exclusion criteria included those who were younger than 25 years, those who had only 1 dermatologic visit at our dermatology center, and those who were unwilling to participate or did not provide written informed consent. Our study was conducted according to Good Clinical Practice.
Data Collection
To obtain data with the highest degree of reliability, 3 sources of information were used: (1) a detailed medical interview conducted by one experienced dermatologist (E.C.) at our dermatology center at the first visit in all study participants, (2) a clinical examination that yielded results necessary for the assessment of scars using a method outlined by Jacob et al, 12 and (3) information included in available medical records. These data were then statistically analyzed.
Statistical Analysis
The results were presented as frequency plots, and a Fisher exact test was conducted to obtain a statistical comparison of the distributions of analyzed data. Unless otherwise indicated, 5% was adopted as the significance level. The statistical analysis was performed using Stata 14 software (StataCorp LLC, College Station, Texas).
Results
Incidence of Different Forms of Adult Acne
To analyze the onset of acne, patients were categorized into 1 of 2 groups: those with persistent adult acne (81.98%) and those with late-onset adult acne (ie, developed after 25 years of age)(18.02%).
Age at Initiation of Dermatologic Treatment
Of the patients with persistent adult acne, 31.87% first visited a dermatologist the same year that the first acne lesions appeared, 36.26% postponed the first visit by at least 5 years (Figure 1), and 23.08% started treatment at least 10 years after acne first appeared. Among patients with persistent adult acne, 76.92% began dermatologic treatment before 25 years of age, and 23.08% began treatment after 25 years of age. Of the latter, 28.57% did not start therapy until they were older than 35 years.

Severity of Adolescent Acne
In the persistent adult acne group, the severity of adolescent acne was assessed during the medical interview as well as detailed histories in medical records. The activity of acne was evaluated at 2-year intervals with the use of a 10-point scale: 1 to 3 points indicated mild acne (7.69% of patients), 4 to 6 points indicated moderate acne (24.18%), and 7 to 10 points indicated severe acne (68.13%).
Treatment of Persistent Acne in Adolescence
Treatment was comprised of oral therapy with antibiotics, isotretinoin, and/or application of topical retinoids (sometimes supported with OCPs). Monotherapy was the standard of treatment more than 25 years ago when patients with persistent adult acne were treated as adolescents or young adults. As many as 43.96% of patients with persistent adult acne did not receive any of these therapies before 25 years of age; rather, they used antiacne cosmetics or beauty procedures. Furthermore, 50.55% of patients were treated with oral antibiotics (Figure 2). Topical retinoids were used in 19.78% of patients and isotretinoin was used in 16.48%. Incidentally, OCPs were given to 26.5%. In the course of adolescent acne, 31.87% of patients received 2 to 4 courses of treatment with either antibiotics or retinoids (oral or topical), and 5.49% were treated with 5 or more courses of treatment (Figure 3). The analysis of each treatment revealed that only 1 patient received 4 courses of isotretinoin. Five courses of oral antibiotics were given in 1 patient, and 3 courses of topical retinoids were given in the same patient.


Topical Retinoids
In an analysis of the number of treatments with topical retinoids completed by patients with persistent adult acne, it was established that 80.22% of patients never used topical retinoids for acne during adolescence. Additionally, 12.08% of these patients completed 1 course of treatment, and 7.69% completed 2 to 4 treatments. However, after 25 years of age, only 25.27% of the patients with persistent adult acne were not treated with topical retinoids, and 35.16% completed more than 2 courses of treatment.
Duration of Treatment
Because adult acne is a chronic disease, the mean number of years that patients received treatment over the disease course was analyzed. In the case of persistent adult acne, the mean duration of treatment, including therapy received during adolescence, was more than 13 years. At the time of the study, more than 30% of patients had been undergoing treatment of adult acne for more than 20 years. Scars— The proportion of patients with persistent adult acne who experienced scarring was evaluated. In the persistent adult acne group, scars were identified in 53.85% of patients. Scars appeared only during adolescence in 26.37% of patients with persistent adult acne, scars appeared only after 25 years of age in 21.97% of patients, and scars appeared in adolescence as well as adulthood in 30.77% of patients.
In an analysis of patients with persistent adult acne who experienced scarring after 25 years of age, the proportion of patients with untreated adolescent acne and those who were treated with antibiotics only was not significantly different (60% vs 64%; P = .478)(Table). The inclusion of topical retinoids into treatment decreased the proportion of scars (isotretinoin: 20%, P = .009; topical retinoids: 38.89%, P = .114).

Comment
Persistent Adult Acne
Patients with symptoms of persistent adult acne represented 81.98% of the study population, which was similar to a 1999 study by Goulden et al, 1 a 2001 study by Shaw and White, 13 and a 2009 report by Schmidt et al. 14 Of these patients with persistent adult acne, 23.08% initiated therapy after 25 years of age, and 23.08% started treatment at least 10 years after acne lesions first appeared. However, it is noteworthy that 68.13% of all patients with persistent adult acne assessed their disease as severe.
Treatment Modalities for Adult Acne
Over the last 5 years, some researchers have attempted to make recommendations for the treatment of adult acne based on standards adopted for the treatment of adolescent acne. 2,9,15 First-line treatment of patients with adult comedonal acne is topical retinoids. 9 The recommended treatment of mild to moderate adult inflammatory acne involves topical drugs, including retinoids, azelaic acid, or benzoyl peroxide, or oral medications, including antibiotics, OCPs, or antiandrogens. In severe inflammatory acne, the recommended treatment involves oral isotretinoin or combined therapies; the latter seems to be the most effective. 16 Furthermore, this therapy has been adjusted to the patient’s current clinical condition; general individual sensitivity of the skin to irritation and the risk for irritant activity of topical medications; and life situation, such as planned pregnancies and intended use of OCPs due to the risk for teratogenic effects of drugs. 17
To assess available treatment modalities, oral therapy with antibiotics or isotretinoin as well as topical retinoids were selected for our analysis. It is difficult to determine an exclusive impact of OCPs as acne treatment; according to our study, many female patients use hormone therapy for other medical conditions or contraception, and only a small proportion of these patients are prescribed hormone treatment for acne. We found that 43.96% of patients with persistent adult acne underwent no treatment with antibiotics, isotretinoin, or topical retinoids in adolescence. Patients who did not receive any of these treatments came only for single visits to a dermatologist, did not comply to a recommended therapy, or used only cosmetics or beauty procedures. We found that 80.22% of patients with persistent adult acne never used topical retinoids during adolescence and did not receive maintenance therapy, which may be attributed to the fact that there were no strict recommendations regarding retinoid treatment when these patients were adolescents or young adults. Published data indicate that retinoid use for acne treatment is not common. 18 Conversely, among patients older than 25 years with late-onset adult acne, there was only 1 patient (ie, < 1%) who had never received any oral antibiotic or isotretinoin treatment or therapy with topical retinoids. The reason for the lack of medical treatment is unknown. Only 25.27% of patients were not treated with topical retinoids, and 35.16% completed at least 2 courses of treatment.
Acne Scarring
The worst complication of acne is scarring. Scars develop for the duration of the disease, during both adolescent and adult acne. In the group with persistent adult acne, scarring was found in 53.85% of patients. Scar formation has been previously reported as a common complication of acne. 19 The effects of skin lesions that remain after acne are not only limited to impaired cosmetic appearance; they also negatively affect mental health and impair quality of life. 20 The aim of our study was to analyze types of treatment for adolescent acne in patients who later had persistent adult acne. Postacne scars observed later are objective evidence of the severity of disease. We found that using oral antibiotics did not diminish the number of scars among persistent adult acne patients in adulthood. In contrast, isotretinoin or topical retinoid treatment during adolescence decreased the risk for scars occurring during adulthood. In our opinion, these findings emphasize the role of this type of treatment among adolescents or young adults. The decrease of scar formation in adult acne due to retinoid treatment in adolescence indirectly justifies the role of maintenance therapy with topical retinoids. 21,22
In the last 20 years, the incidence of acne lesions in adults has markedly increased. 1 Acne affects adults (individuals older than 25 years) and is no longer a condition limited to adolescents and young adults (individuals younger than 25 years). According to Dreno et al, 2 the accepted age threshold for the onset of adult acne is 25 years. 1-3 In 2013, the term adult acne was defined. 2 Among patients with adult acne, there are 2 subtypes: (1) persistent adult acne, which is a continuation or recurrence of adolescent acne, affecting approximately 80% of patients, and (2) late-onset acne, affecting approximately 20% of patients. 4
Clinical symptoms of adult acne and available treatment modalities have been explored in the literature. Daily clinical experience shows that additional difficulties involved in the management of adult acne patients are related mainly to a high therapeutic failure rate in acne patients older than 25 years. 5 Persistent adult acne seems to be noteworthy because it causes long-term symptoms, and patients experience uncontrollable recurrences.
It is believed that adult acne often is resistant to treatment. 2 Adult skin is more sensitive to topical agents, leading to more irritation by medications intended for external use and cosmetics. 6 Scars in these patients are a frequent and undesirable consequence. 3
Effective treatment of acne encompasses oral antibiotics, topical and systemic retinoids, and oral contraceptive pills (OCPs). For years, oral subantimicrobial doses of cyclines have been recommended for acne treatment. Topical and oral retinoids have been successfully used for more than 30 years as important therapeutic options. 7 More recent evidence-based guidelines for acne issued by the American Academy of Dermatology 8 and the European Dermatology Forum 9 also show that retinoids play an important role in acne therapy. Their anti-inflammatory activity acts against comedones and their precursors (microcomedones). Successful antiacne therapy not only achieves a smooth face without comedones but also minimizes scar formation, postinflammatory discoloration, and long-lasting postinflammatory erythema. 10 Oral contraceptives have a mainly antiseborrheic effect. 11
Our study sought to analyze the potential influence of therapy during adolescent acne on patients who later developed adult acne. Particular attention was given to the use of oral antibiotics, isotretinoin, and topical retinoids for adolescent acne and their potential role in diminishing scar formation in adult acne.
Materials and Methods
Patient Demographics and Selection
A population-based study of Polish patients with adult acne was conducted. Patients were included in the study group on a consecutive basis from among those who visited our outpatient dermatology center from May 2015 to January 2016. A total of 111 patients (101 women [90.99%] and 10 men [9.01%]) were examined. The study group comprised patients aged 25 years and older who were treated for adult acne (20 patients [18.02%] were aged 25–29 years, 61 [54.95%] were aged 30–39 years, and 30 [27.02%] were 40 years or older).
The following inclusion criteria were used: observation period of at least 6 months in our dermatologic center for patients diagnosed with adult acne, at least 2 dermatologic visits for adult acne prior to the study, written informed consent for study participation and data processing (the aim of the study was explained to each participant by a dermatologist), and age 25 years or older. Exclusion criteria included those who were younger than 25 years, those who had only 1 dermatologic visit at our dermatology center, and those who were unwilling to participate or did not provide written informed consent. Our study was conducted according to Good Clinical Practice.
Data Collection
To obtain data with the highest degree of reliability, 3 sources of information were used: (1) a detailed medical interview conducted by one experienced dermatologist (E.C.) at our dermatology center at the first visit in all study participants, (2) a clinical examination that yielded results necessary for the assessment of scars using a method outlined by Jacob et al, 12 and (3) information included in available medical records. These data were then statistically analyzed.
Statistical Analysis
The results were presented as frequency plots, and a Fisher exact test was conducted to obtain a statistical comparison of the distributions of analyzed data. Unless otherwise indicated, 5% was adopted as the significance level. The statistical analysis was performed using Stata 14 software (StataCorp LLC, College Station, Texas).
Results
Incidence of Different Forms of Adult Acne
To analyze the onset of acne, patients were categorized into 1 of 2 groups: those with persistent adult acne (81.98%) and those with late-onset adult acne (ie, developed after 25 years of age)(18.02%).
Age at Initiation of Dermatologic Treatment
Of the patients with persistent adult acne, 31.87% first visited a dermatologist the same year that the first acne lesions appeared, 36.26% postponed the first visit by at least 5 years (Figure 1), and 23.08% started treatment at least 10 years after acne first appeared. Among patients with persistent adult acne, 76.92% began dermatologic treatment before 25 years of age, and 23.08% began treatment after 25 years of age. Of the latter, 28.57% did not start therapy until they were older than 35 years.

Severity of Adolescent Acne
In the persistent adult acne group, the severity of adolescent acne was assessed during the medical interview as well as detailed histories in medical records. The activity of acne was evaluated at 2-year intervals with the use of a 10-point scale: 1 to 3 points indicated mild acne (7.69% of patients), 4 to 6 points indicated moderate acne (24.18%), and 7 to 10 points indicated severe acne (68.13%).
Treatment of Persistent Acne in Adolescence
Treatment was comprised of oral therapy with antibiotics, isotretinoin, and/or application of topical retinoids (sometimes supported with OCPs). Monotherapy was the standard of treatment more than 25 years ago when patients with persistent adult acne were treated as adolescents or young adults. As many as 43.96% of patients with persistent adult acne did not receive any of these therapies before 25 years of age; rather, they used antiacne cosmetics or beauty procedures. Furthermore, 50.55% of patients were treated with oral antibiotics (Figure 2). Topical retinoids were used in 19.78% of patients and isotretinoin was used in 16.48%. Incidentally, OCPs were given to 26.5%. In the course of adolescent acne, 31.87% of patients received 2 to 4 courses of treatment with either antibiotics or retinoids (oral or topical), and 5.49% were treated with 5 or more courses of treatment (Figure 3). The analysis of each treatment revealed that only 1 patient received 4 courses of isotretinoin. Five courses of oral antibiotics were given in 1 patient, and 3 courses of topical retinoids were given in the same patient.


Topical Retinoids
In an analysis of the number of treatments with topical retinoids completed by patients with persistent adult acne, it was established that 80.22% of patients never used topical retinoids for acne during adolescence. Additionally, 12.08% of these patients completed 1 course of treatment, and 7.69% completed 2 to 4 treatments. However, after 25 years of age, only 25.27% of the patients with persistent adult acne were not treated with topical retinoids, and 35.16% completed more than 2 courses of treatment.
Duration of Treatment
Because adult acne is a chronic disease, the mean number of years that patients received treatment over the disease course was analyzed. In the case of persistent adult acne, the mean duration of treatment, including therapy received during adolescence, was more than 13 years. At the time of the study, more than 30% of patients had been undergoing treatment of adult acne for more than 20 years. Scars— The proportion of patients with persistent adult acne who experienced scarring was evaluated. In the persistent adult acne group, scars were identified in 53.85% of patients. Scars appeared only during adolescence in 26.37% of patients with persistent adult acne, scars appeared only after 25 years of age in 21.97% of patients, and scars appeared in adolescence as well as adulthood in 30.77% of patients.
In an analysis of patients with persistent adult acne who experienced scarring after 25 years of age, the proportion of patients with untreated adolescent acne and those who were treated with antibiotics only was not significantly different (60% vs 64%; P = .478)(Table). The inclusion of topical retinoids into treatment decreased the proportion of scars (isotretinoin: 20%, P = .009; topical retinoids: 38.89%, P = .114).

Comment
Persistent Adult Acne
Patients with symptoms of persistent adult acne represented 81.98% of the study population, which was similar to a 1999 study by Goulden et al, 1 a 2001 study by Shaw and White, 13 and a 2009 report by Schmidt et al. 14 Of these patients with persistent adult acne, 23.08% initiated therapy after 25 years of age, and 23.08% started treatment at least 10 years after acne lesions first appeared. However, it is noteworthy that 68.13% of all patients with persistent adult acne assessed their disease as severe.
Treatment Modalities for Adult Acne
Over the last 5 years, some researchers have attempted to make recommendations for the treatment of adult acne based on standards adopted for the treatment of adolescent acne. 2,9,15 First-line treatment of patients with adult comedonal acne is topical retinoids. 9 The recommended treatment of mild to moderate adult inflammatory acne involves topical drugs, including retinoids, azelaic acid, or benzoyl peroxide, or oral medications, including antibiotics, OCPs, or antiandrogens. In severe inflammatory acne, the recommended treatment involves oral isotretinoin or combined therapies; the latter seems to be the most effective. 16 Furthermore, this therapy has been adjusted to the patient’s current clinical condition; general individual sensitivity of the skin to irritation and the risk for irritant activity of topical medications; and life situation, such as planned pregnancies and intended use of OCPs due to the risk for teratogenic effects of drugs. 17
To assess available treatment modalities, oral therapy with antibiotics or isotretinoin as well as topical retinoids were selected for our analysis. It is difficult to determine an exclusive impact of OCPs as acne treatment; according to our study, many female patients use hormone therapy for other medical conditions or contraception, and only a small proportion of these patients are prescribed hormone treatment for acne. We found that 43.96% of patients with persistent adult acne underwent no treatment with antibiotics, isotretinoin, or topical retinoids in adolescence. Patients who did not receive any of these treatments came only for single visits to a dermatologist, did not comply to a recommended therapy, or used only cosmetics or beauty procedures. We found that 80.22% of patients with persistent adult acne never used topical retinoids during adolescence and did not receive maintenance therapy, which may be attributed to the fact that there were no strict recommendations regarding retinoid treatment when these patients were adolescents or young adults. Published data indicate that retinoid use for acne treatment is not common. 18 Conversely, among patients older than 25 years with late-onset adult acne, there was only 1 patient (ie, < 1%) who had never received any oral antibiotic or isotretinoin treatment or therapy with topical retinoids. The reason for the lack of medical treatment is unknown. Only 25.27% of patients were not treated with topical retinoids, and 35.16% completed at least 2 courses of treatment.
Acne Scarring
The worst complication of acne is scarring. Scars develop for the duration of the disease, during both adolescent and adult acne. In the group with persistent adult acne, scarring was found in 53.85% of patients. Scar formation has been previously reported as a common complication of acne. 19 The effects of skin lesions that remain after acne are not only limited to impaired cosmetic appearance; they also negatively affect mental health and impair quality of life. 20 The aim of our study was to analyze types of treatment for adolescent acne in patients who later had persistent adult acne. Postacne scars observed later are objective evidence of the severity of disease. We found that using oral antibiotics did not diminish the number of scars among persistent adult acne patients in adulthood. In contrast, isotretinoin or topical retinoid treatment during adolescence decreased the risk for scars occurring during adulthood. In our opinion, these findings emphasize the role of this type of treatment among adolescents or young adults. The decrease of scar formation in adult acne due to retinoid treatment in adolescence indirectly justifies the role of maintenance therapy with topical retinoids. 21,22
- Goulden V, Stables GI, Cunliffe WJ. Prevalence of facial acne in adults. J Am Acad Dermatol. 1999;41:577-580.
- Dreno B, Layton A, Zouboulis CC, et al. Adult female acne: a new paradigm. J Eur Acad Dermatol Venereol. 2013;27:1063-1070.
- Preneau S, Dreno B. Female acne--a different subtype of teenager acne? J Eur Acad Dermatol Venereol. 2012;26:277-282.
- Goulden V, Clark SM, Cunliffe WJ. Post-adolescent acne: a review of clinical features. Br J Dermatol. 1997;136:66-70.
- Kamangar F, Shinkai K. Acne in the adult female patient: a practical approach. Int J Dermatol. 2012;51:1162-1174.
- Choi CW, Lee DH, Kim HS, et al. The clinical features of late onset acne compared with early onset acne in women. J Eur Acad Dermatol Venereol. 2011;25:454-461.
- Kligman AM, Fulton JE Jr, Plewig G. Topical vitamin A acid in acne vulgaris. Arch Dermatol. 1969;99:469-476.
- Zaenglein AL, Pathy AL, Schlosser BJ, et al. Guidelines of care for the management of acne vulgaris. J Am Acad Dermatol. 2016;74:945.e33-973.e33.
- Nast A, Dreno B, Bettoli V, et al. European evidence-based guidelines for the treatment of acne. J Eur Acad Dermatol Venereol. 2012;26(suppl 1):1-29.
- Levin J. The relationship of proper skin cleansing to pathophysiology, clinical benefits, and the concomitant use of prescription topical therapies in patients with acne vulgaris. Dermatol Clin. 2016;34:133-145.
- Savage LJ, Layton AM. Treating acne vulgaris: systemic, local and combination therapy. Expert Rev Clin Pharmacol. 2010;3:563-580.
- Jacob CL, Dover JS, Kaminer MS. Acne scarring: a classification system and review of treatment options. J Am Acad Dermatol. 2001;45:109-117.
- Shaw JC, White LE. Persistent acne in adult women. Arch Dermatol. 2001;137:1252-1253.
- Schmidt JV, Masuda PY, Miot HA. Acne in women: clinical patterns in different age groups. An Bras Dermatol. 2009;84:349-354.
- Thiboutot D, Gollnick H, Bettoli V, et al. New insights into the management of acne: an update from the Global Alliance to Improve Outcomes in Acne group. J Am Acad Dermatol. 2009;60(5 suppl):1-50.
- Williams C, Layton AM. Persistent acne in women: implications for the patient and for therapy. Am J Clin Dermatol. 2006;7:281-290.
- Holzmann R, Shakery K. Postadolescent acne in females. Skin Pharmacol Physiol. 2014;27(suppl 1):3-8.
- Pena S, Hill D, Feldman SR. Use of topical retinoids by dermatologist and non-dermatologist in the management of acne vulgaris. J Am Acad Dermatol. 2016;74:1252-1254.
- Layton AM, Henderson CA, Cunliffe WJ. A clinical evaluation of acne scarring and its incidence. Clin Exp Dermatol. 1994;19;303-308.
- Halvorsen JA, Stern RS, Dalgard F, et al. Suicidal ideation, mental health problems, and social impairment are increased in adolescents with acne: a population-based study. J Invest Dermatol. 2011;131:363-370.
- Thielitz A, Sidou F, Gollnick H. Control of microcomedone formation throughout a maintenance treatment with adapalene gel, 0.1%. J Eur Acad Dermatol Venereol. 2007;21:747-753.
- Leyden J, Thiboutot DM, Shalita R, et al. Comparison of tazarotene and minocycline maintenance therapies in acne vulgaris: a multicenter, double-blind, randomized, parallel-group study. Arch Dermatol. 2006;142:605-612.
- Goulden V, Stables GI, Cunliffe WJ. Prevalence of facial acne in adults. J Am Acad Dermatol. 1999;41:577-580.
- Dreno B, Layton A, Zouboulis CC, et al. Adult female acne: a new paradigm. J Eur Acad Dermatol Venereol. 2013;27:1063-1070.
- Preneau S, Dreno B. Female acne--a different subtype of teenager acne? J Eur Acad Dermatol Venereol. 2012;26:277-282.
- Goulden V, Clark SM, Cunliffe WJ. Post-adolescent acne: a review of clinical features. Br J Dermatol. 1997;136:66-70.
- Kamangar F, Shinkai K. Acne in the adult female patient: a practical approach. Int J Dermatol. 2012;51:1162-1174.
- Choi CW, Lee DH, Kim HS, et al. The clinical features of late onset acne compared with early onset acne in women. J Eur Acad Dermatol Venereol. 2011;25:454-461.
- Kligman AM, Fulton JE Jr, Plewig G. Topical vitamin A acid in acne vulgaris. Arch Dermatol. 1969;99:469-476.
- Zaenglein AL, Pathy AL, Schlosser BJ, et al. Guidelines of care for the management of acne vulgaris. J Am Acad Dermatol. 2016;74:945.e33-973.e33.
- Nast A, Dreno B, Bettoli V, et al. European evidence-based guidelines for the treatment of acne. J Eur Acad Dermatol Venereol. 2012;26(suppl 1):1-29.
- Levin J. The relationship of proper skin cleansing to pathophysiology, clinical benefits, and the concomitant use of prescription topical therapies in patients with acne vulgaris. Dermatol Clin. 2016;34:133-145.
- Savage LJ, Layton AM. Treating acne vulgaris: systemic, local and combination therapy. Expert Rev Clin Pharmacol. 2010;3:563-580.
- Jacob CL, Dover JS, Kaminer MS. Acne scarring: a classification system and review of treatment options. J Am Acad Dermatol. 2001;45:109-117.
- Shaw JC, White LE. Persistent acne in adult women. Arch Dermatol. 2001;137:1252-1253.
- Schmidt JV, Masuda PY, Miot HA. Acne in women: clinical patterns in different age groups. An Bras Dermatol. 2009;84:349-354.
- Thiboutot D, Gollnick H, Bettoli V, et al. New insights into the management of acne: an update from the Global Alliance to Improve Outcomes in Acne group. J Am Acad Dermatol. 2009;60(5 suppl):1-50.
- Williams C, Layton AM. Persistent acne in women: implications for the patient and for therapy. Am J Clin Dermatol. 2006;7:281-290.
- Holzmann R, Shakery K. Postadolescent acne in females. Skin Pharmacol Physiol. 2014;27(suppl 1):3-8.
- Pena S, Hill D, Feldman SR. Use of topical retinoids by dermatologist and non-dermatologist in the management of acne vulgaris. J Am Acad Dermatol. 2016;74:1252-1254.
- Layton AM, Henderson CA, Cunliffe WJ. A clinical evaluation of acne scarring and its incidence. Clin Exp Dermatol. 1994;19;303-308.
- Halvorsen JA, Stern RS, Dalgard F, et al. Suicidal ideation, mental health problems, and social impairment are increased in adolescents with acne: a population-based study. J Invest Dermatol. 2011;131:363-370.
- Thielitz A, Sidou F, Gollnick H. Control of microcomedone formation throughout a maintenance treatment with adapalene gel, 0.1%. J Eur Acad Dermatol Venereol. 2007;21:747-753.
- Leyden J, Thiboutot DM, Shalita R, et al. Comparison of tazarotene and minocycline maintenance therapies in acne vulgaris: a multicenter, double-blind, randomized, parallel-group study. Arch Dermatol. 2006;142:605-612.
Practice Points
- Postacne scarring is the most severe complication of acne.
- Isotretinoin or topical retinoid treatment in adolescence decreases the risk for scars during adult acne, justifying the role of maintenance therapy with topical retinoids.
What’s New in the Management of Acne Vulgaris
Inflammation is a backdrop to the commonly cited elements of the pathophysiology of acne: Propionibacterium acnes proliferation, increased sebum production with an increase in circulating androgens, and faulty keratinization.1,2 In fact, research shows that the initiating lesion of acne vulgaris—the microcomedone—is, in essence, an inflammatory lesion.3 This realization has clearly influenced the approach to acne treatment but has not yielded a bevy of new treatments.
A better understanding of acne pathophysiology and the role of inflammation has, however, yielded a better understanding of how existing therapies treat the disease and have led to more comprehensive treatment strategies that are multitargeted. Nonetheless, topical and oral antibiotics remain mainstays of acne therapy, along with topical retinoids and benzoyl peroxide. Current guidelines of care for acne emphasize strategies that reduce dependence on antibiotics and minimize the risk for resistance.4 The therapeutic landscape might at last be shifting, with new chemical entities for acne and several novel formulations in development.
Sarecycline: A Novel Tetracycline
Tetracycline antibiotics have been used to manage acne since the 1950s, but their method of action in the disease has not been fully elucidated.5 In addition to antibiotic effects, tetracyclines have been shown to confer anti-inflammatory properties and other biologic effects.6,7
First-generation tetracycline is broad spectrum. As such, it is associated with increased potential for antibiotic resistance and greater impact on gastrointestinal health. The novel compound sarecycline is a tetracycline with a narrower spectrum of activity compared to other tetracyclines and with reduced activity against enteric gram-negative bacteria8 (Figure 1). Sarecycline recently was approved by the US Food and Drug Administration (FDA) in a once-daily oral formulation for the treatment of inflammatory lesions of nonnodular moderate to severe acne vulgaris in patients 9 years and older. Sarecycline is dosed at 1.5 mg/kg daily. The FDA approval marks the first new antibiotic approved for acne in 4 decades.

In 2 phase 3 clinical trials, sarecycline demonstrated efficacy in reducing both inflammatory and noninflammatory lesions.9 At week 12, investigator global assessment (IGA) success (≥2 point reduction in IGA and score 0 [clear] or 1 [almost clear]) rates were 21.9% and 22.6% for active treatment (n=483 and n=519), respectively, in the 2 trials compared to 10.5% and 15.3% (n=485 and n=515), respectively, for controls. Sarecycline demonstrated rapid anti-inflammatory effect. Onset of action against inflammatory lesions was notable by week 3. At week 12, inflammatory lesions were reduced in the active treatment arms by 51.8% and 49.9%, respectively, compared to 35.1% and 35.4%, respectively, for controls.9
The most common reported treatment-emergent adverse events (TEAEs) were nausea, nasopharyngitis, headache, and vomiting.9 Vestibular (dizziness, tinnitus, vertigo) and phototoxic (sunburn, photosensitivity) TEAEs both occurred in 1% or fewer of sarecycline patients. Gastrointestinal TEAE rates for sarecycline were low.9
Sarecycline also was assessed in the 2 trials for efficacy in the treatment of back and chest acne; in the active treatment group, IGA success was achieved by 29.6% and 36.6%, respectively, compared to 19.6% and 21.6%, respectively, of controls.9
Tazarotene Foam in Focus
Topical tazarotene is commercially available in cream, gel, and foam formulations. Tazarotene foam 0.1% was FDA approved in 2012 for the treatment of acne vulgaris in patients 12 years and older. However, the product was recently relaunched to the market and therefore warrants discussion.
Similar to other retinoids, topical tazarotene has been associated with the potential for application-site irritation. This aqueous foam formulation of tazarotene was designed for ease of application and to attempt to impart moisturizing effects to offset potential irritation. It contains noncomedogenic light mineral oil, which is an emollient. The foam spreads easily, including on hair-bearing skin, with demonstrated penetration of the active drug into the epidermis and dermis. Nonetheless, compared to the gel formulation of tazarotene, the foam formulation was associated with reduced systemic exposure.10
The tazarotene foam formulation does not contain alcohol, fragrance, propylene glycol, or parabens. Clinical trial participants, blinded to whether they were on active treatment or vehicle foam, consistently rated the foam formulation favorably for ease of application and spreadability, lack of stickiness or residue, and moisturizing effect. The foam vehicle is suggested to increase compliance and satisfaction in some patients.11The efficacy and tolerability of tazarotene foam 0.1% was investigated in 2 randomized, double-blind, vehicle-controlled, parallel-group studies in the United States and Canada.12 The studies involved participants aged 2 to 45 years who were randomized to receive treatment with either tazarotene foam 0.1% or vehicle foam once daily for 12 weeks (N=1486). Lesion counts, investigator static global assessment, and subject global assessment were evaluated at baseline and at weeks 2, 4, 8, and 12. At week 12, mean reduction from baseline in noninflammatory lesions was 55.9% for active treatment, mean reduction in inflammatory lesions was 56.1%, and mean total lesion reduction was 56% compared to mean reductions of 37.7%, 45.3%, and 40.8%, respectively, for vehicle. In all, 28.2% of participants achieved treatment success with active treatment compared to 14.7% of controls. There was a greater proportion of active-treatment participants with investigator static global assessment scores of 0 or 1 compared to vehicle. The only adverse events reported by more than 5% of participants in the active-treatment groups in both studies were application-site skin irritation and dryness.12
Topical Minocycline
Systemic minocycline is the most commonly prescribed oral antibiotic for acne management.13 Despite its widespread use, it is not without potential safety concerns. Minocycline is distinct among tetracyclines for posing a small risk for systemic lupus erythematosus and autoimmune TEAE.Gastrointestinal side effects and bluish discoloration also are reported.14 Topical application of minocycline for acne would optimize the therapeutic effect while reducing systemic effects. FMX101 4%, an investigational minocycline foam, is being studied for the treatment of moderate to severe acne.
In a pharmacokinetic study, minocycline exposure was 730- to 765-times lower with foam application vs oral minocycline.15 No evidence of minocycline accumulation was identified over the 21 days of application of minocycline foam 4%. Minocycline foam 4% appeared to be safe and well tolerated, without serious TEAEs, treatment-related TEAEs, or TEAEs that led to treatment discontinuation.15
In 2 identical phase 3 studies in which 961 participants were randomized (2:1) to once-daily minocycline foam 4% or foam vehicle for 12 weeks, participants in the active-treatment group demonstrated a significantly greater reduction in both inflammatory and noninflammatory lesions in both studies (both P<.05) and a greater rate of treatment success (≥2 point reduction in IGA and score of 0 [clear] or 1 [almost clear]) in 1 study. Treatment was generally safe and well tolerated, with skin-related adverse events reported in fewer than 1% of participants receiving active treatment.16
In an open-label safety extension study that enrolled 657 patients, treatment with FMX101 continued for as long as 40 weeks.17 In total, 291 participants completed 52 weeks of therapy. Rates and types of reported TEAEs in the open-label extension phase were similar to those seen in the phase 3 trials. Application-site TEAEs occurred in fewer than 2% of participants. Participants reported a high level of treatment satisfaction at week 52.17
In a more recent phase 3 study, 1507 participants were randomized (1:1) to once-daily minocycline foam 4% or foam vehicle for 12 weeks to further evaluate the efficacy and safety of FMX101 4% for moderate to severe acne vulgaris.18 The study met both primary end points: absolute change from baseline in the inflammatory lesion count (−16.93 vs −13.40; P<.0001) and the noninflammatory lesion count (−18.80 vs −15.89; P<.05), as well as percentage of participants with IGA treatment success at week 12 (30.80% vs 19.63%; P<.0001). The percentage reduction in the inflammatory lesion count was statistically significantly greater for minocycline foam 4% compared to vehicle as early as week 3 (P<.0001). The safety profile was found to be consistent with the 2 earlier phase 3 studies.18
Topical Minocycline in Rosacea
A similar foam formulation of minocycline (1.5% concentration) has shown benefit in 2 identical phase 3 studies.19 A total of 1522 participants were enrolled in 2 phase 3, randomized, multicenter, double-blind, vehicle-controlled, 2-arm studies in participants 18 years and older with moderate to severe papulopustular rosacea. Participants were randomized (2:1) to either minocycline foam 1.5% or vehicle once daily to the face for 12 weeks.19
Treatment was associated with a statistically significant reduction in counts of inflammatory lesions of rosacea (Study FX2016-11: −17.57 vs −15.65 [P=.003]; Study FX2016-12: −18.54 vs −14.88 [P<.0001]) and a significantly higher rate of IGA treatment success compared to vehicle (Study FX2016-11: 52.1% vs 43.0% [P=.027]; Study FX2016-12: 49.1% vs 39.0% [P=.008]), highlighting the anti-inflammatory action of the topically applied agent.19
The most common TEAE for both studies was upper respiratory tract infection; there were no serious TEAEs. Overall, 9 participants across both studies discontinued because of a TEAE (foam, 7 participants; vehicle, 2 participants).19
Clascoterone: First-in-Class Topical
Clascoterone cream 1% is a new chemical entity under investigation for the treatment of moderate to severe acne in patients 9 years and older. Clascoterone targets androgen receptors in the skin to block the effects of circulating endogenous androgens; chemically, it shares a 4-ring backbone identical to dihydrotestosterone and spironolactone (Figure 2). Clascoterone competes with dihydrotestosterone for binding to the androgen receptor to limit or block transcription of androgen-responsive genes and modify specific gene expression.20

Androgens are known to promote both sebum production and inflammatory responses within the follicle, contributing to the cycle of acne.21 Antiandrogen therapy would, therefore, inhibit excess sebum production and directly reduce the presence of certain inflammatory mediators in skin. This effect is expected to lead to reduced follicular plugging and a reduction in growth of P acnes and its inflammatory by-products.
Direct and indirect hormonal modulation have been successfully employed to manage acne in women; however, such therapies have not been considered first-line interventions for the disease.22 Although systemic antiandrogens and hormonal modulation are effective for certain women with acne, there may be concerns about systemic exposure23; no hormone-modulating agent has been adopted for use in men with acne.
As an androgen inhibitor, clascoterone is thought to displace androgen hormones from androgen receptors located at the sebaceous gland and hair follicle, thus inhibiting the cycle of physiologic events that leads to acne formation. Clascoterone is applied topically and acts locally on androgen receptors in the skin, with no systemic exposure seen. In phase 2 trials, clascoterone was found to be safe and effective with no systemic exposure and was suggested to have better tolerability than topical tretinoin.
Preliminary individual study analysis of data from 2 phase 3 trials showed that topical clascoterone met its primary end points, achieving statistically significantly greater rates of IGA treatment success (≥2 point reduction in IGA and score of 0 [clear] or 1 [almost clear]) at week 12 (P<.0001).24 Rates of treatment success for actively treated participants were 16.1% and 18.7%, respectively, compared to 7% and 4.7%, respectively, for vehicle. The study population included both males and nonpregnant females 9 years and older who had a baseline IGA score of 3 (moderate) or 4 (severe). At baseline, participants had a mix of inflammatory lesions (≥30, to a maximum of 75) and noninflammatory lesions (≥30, to a maximum of 100).24
Intention-to-treat analysis at week 12 showed a mean total lesion reduction from baseline for active treatment of 37.1% and 37.7%, respectively, compared to 28.5% and 22.2%, respectively, for controls.24 Mean reductions from baseline in noninflammatory lesions for active treatment were 30.7% and 29.3%, respectively, compared to 21.9% and 15.8%, respectively, for controls. Mean reductions from baseline in inflammatory lesions for active treatment were 44.8% and 47%, respectively, compared to 36.6% and 29.8%, respectively, for controls. Similarly low rates of TEAEs were reported in active and placebo groups in both studies. No TEAE suggested systemic antiandrogen exposure.24
Advancements in Cannabinoids
Advancements in pharmaceutical development of cannabinoid compounds have largely coincided with the controversial national movement to legalize medical marijuana and decriminalize recreational marijuana use. Despite the temporal connection, the 2 topics are entirely distinct. Importantly, pharmaceutical development is largely focused on the effects of cannabidiol (CBD), which is 1 of approximately 113 cannabinoids identified from Cannabis sativa. Cannabidiol is not tetrahydrocannabinol, or THC, the compound responsible for marijuana’s psychoactive effects and addictive properties; CBD does not have any psychoactive effects and is not addictive (Figure 3).25

A CBD oral solution agent recently gained FDA approval for seizures associated with Lennox-Gastaut syndrome or Dravet syndrome in patients 2 years and older; it is estimated that more than 180 trials of CBD are ongoing in the United States for various indications.26 A notable question in the development of CBD-based therapies is: What is the role of natural plant-derived CBD compared to a pure synthetic form of CBD? The latter is akin to a pharmaceutical process in which a single molecule is developed as the active drug.27 Although the potency and composition of plant-derived CBD can vary with crop conditions, plant strains, and the extraction process, a synthetic molecule would allow for consistency in safety, potency, and pharmacokinetic properties, as well as efficacy, as a consequence.26
There are intriguing data to suggest a potential use for topical CBD in the management of skin diseases, including acne vulgaris. Researchers have, for at least a decade, been investigating the role of the endocannabinoid system, which has physiologic regulatory functions in proliferation, differentiation, apoptosis and cytokine, mediator, and hormone production of various cell types in skin, hair follicles, and sebaceous glands.28 Cannabidiol has been shown to suppress proliferation of sebocytes through activation of transient receptor potential vanilloid 4 ion channels and to have anti-inflammatory effects on sebocytes.29 It has been shown to inhibit human keratinocyte proliferation through a non-CB1/CB2 mechanism30 and to possess potent antimicrobial activity against gram-positive bacteria such as P acnes.31
Given these effects on sebocytes, modulation of keratinocyte proliferation, and anti-inflammatory and antibacterial effects, CBD could prove beneficial in the management of acne vulgaris. A new synthetic CBD topical formulation, BTX 1503, is under investigation for the treatment of acne vulgaris.
Early clinical data confirm both the anti-inflammatory effects of topical BTX 1503 as well as its effects on noninflammatory lesions, with 4-week reductions in inflammatory lesion counts similar to what are reported in clinical trials for leading FDA-approved topical therapies in the same time frame.
The phase 1b trial was a 4-week, open-label study in participants with moderate to severe acne vulgaris.32 The primary end point was safety, as demonstrated by the incidence of TEAE, laboratory monitoring, and assessment of cutaneous tolerability. Exploratory end points included changes in inflammatory and noninflammatory lesion counts and IGA score. A total of 21 participants aged 18 to 65 years with moderate to severe acne vulgaris were enrolled. BTX 1503 was applied topically twice daily. At baseline, eligible participants had 20 to 50 inflammatory lesions and 20 to 100 noninflammatory acne lesions on the face, an IGA of 3 (moderate) or 4 (severe), and 3 or fewer nodular or cystic lesions (>5 mm in diameter). No serious or severe TEAEs were reported; no participants withdrew due to a TEAE. Slight erythema, slight scaling, slight dryness, and slight burning and stinging were reported; there were no reports of irritant or allergic contact dermatitis. Only 1 TEAE was thought to be possibly related to treatment: mild pain at the application site.32
In addition to presenting a potential new chemical entity for the topical treatment of acne, the novel topical vehicle formulation of BTX 1503 represents an innovative approach to drug delivery. The formulation utilizes proprietary technology to deliver high doses of drug into the skin without controversial penetration enhancers, preservatives, or other potential irritating additives. Instead, volatile excipients are used that evaporate upon application to the skin, leaving a so-called superconcentrated secondary formulation on the skin. The concentration gradient effect then drives the concentrated drug into skin. Although the formulation efficiently delivers active drug into the skin and its appendages, systemic exposure has been reported to be very low. A phase 2 randomized, double-blind, vehicle-controlled trial ongoing in the United States and Australia in 360 patients with moderate to severe acne vulgaris will provide key data to confirm the efficacy and safety of BTX 1503 (ClinicalTrials.gov Identifier NCT03573518).
Conclusion
Drug development continues to focus on the challenge of treating acne effectively and safely. Vehicle innovations are optimizing existing active drugs and creating opportunities to deliver new compounds to the skin. The approval of sarecycline as the first new chemical entity approved for acne in several years may be followed in coming years by other new actives, including clascoterone and CBD.
- Webster GF. The pathophysiology of acne. Cutis. 2005;76(2 suppl):4-7.
- Burkhart CN, Gottwald L. Assessment of etiologic agents in acne pathogenesis. Skinmed. 2003;2:222-228.
- Kang S, Cho S, Chung JH, et al. Inflammation and extracellular matrix degradation mediated by activated transcription factors nuclear factor-kappaB and activator protein-1 in inflammatory acne lesions in vivo. Am J Pathol. 2005;166:1691-1699.
- Zaenglein AL, Pathy AL, Schlosser BJ, et al. Guidelines of care for the management of acne vulgaris. J Am Acad Dermatol. 2016;74:945-973.
- Garrido-Mesa N, Zarzuelo A, Gálvez J. Minocycline: far beyond an antibiotic. Br J Pharmacol. 2013;169:337-352.
- Griffin MO, Ceballos G, Villarreal FJ. Tetracycline compounds with non-antimicrobial organ protective properties: possible mechanisms of action. Pharmacol Res. 2011;63:102-107.
- Weinberg JM. The anti-inflammatory effects of tetracyclines. Cutis. 2005;75(4 suppl):6-11.
- Leyden JJ, Sniukiene V, Berk DR, et al. Efficacy and safety of sarecycline, a novel, once-daily, narrow spectrum antibiotic for the treatment of moderate to severe facial acne vulgaris: results of a phase 2, dose-ranging study. J Drugs Dermatol. 2018;17:333-338.
- Moore A, Green LJ, Bruce S, et al. Once-daily oral sarecycline 1.5 mg/kg/day is effective for moderate to severe acne vulgaris: results from two identically designed, phase 3, randomized, double-blind clinical trials. J Drugs Dermatol. 2018;17:987-996.
- Jarratt M, Werner CP, Alió Saenz AB. Tazarotene foam versus tazarotene gel: a randomized relative bioavailability study in acne vulgaris. Clin Drug Investig. 2013;33:283-289.
- Smith JA, Narahari S, Hill D, et al. Tazarotene foam, 0.1%, for the treatment of acne. Expert Opin Drug Saf. 2016;15:99-103.
- Feldman SR, Werner CP, Alió Saenz AB. The efficacy and tolerability of tazarotene foam, 0.1%, in the treatment of acne vulgaris in 2 multicenter, randomized, vehicle-controlled, double-blind studies. J Drugs Dermatol. 2013;12:438-446.
- Lee YH, Liu G, Thiboutot DM, et al. A retrospective analysis of the duration of oral antibiotic therapy for the treatment of acne among adolescents: investigating practice gaps and potential cost-savings. J Am Acad Dermatol. 2014;71:70-76.
- Garner SE, Eady A, Bennett C, et al. Minocycline for acne vulgaris: efficacy and safety. Cochrane Database Syst Rev. 2012(8):CD002086.
- Jones TM, Ellman H, deVries T. Pharmacokinetic comparison of once-daily topical minocycline foam 4% vs oral minocycline for moderate-to-severe acne. J Drugs Dermatol. 2017;16:1022-1028.
- Gold LS, Dhawan S, Weiss J, et al. A novel topical minocycline foam for the treatment of moderate-to-severe acne vulgaris: results of 2 randomized, double-blind, phase 3 studies. J Am Acad Dermatol. 2019;80:168-177.17.
- Gold LS, Dhawan S, Weiss J, et al. FMX101 4% minocycline foam for the treatment of acne vulgaris: safety and patient satisfaction from the open-label extension of 2 phase 3 studies. Poster presented at: 2018 Winter Clinical Dermatology Conference; January 12-17, 2018; Maui, HI.
- Raoof J, Hooper D, Moore A, et al. FMX101 4% topical minocycline foam for the treatment of moderate to severe acne vulgaris: efficacy and safety from a phase 3 randomized, double-blind, vehicle-controlled study. Poster presented at: 2018 Fall Clinical Dermatology Conference; October 18-21, 2018; Las Vegas, NV.
- Gold LS, Del Rosso JQ, Bhatia ND, et al. Efficacy and safety of FMX103 (1.5% minocycline foam) in the treatment of moderate-to-severe papulopustular rosacea: results from two phase 3 randomized, multicenter, double-blind, vehicle-controlled studies. Poster presented at: 2019 Winter Clinical Dermatology Conference; January 18-23; 2019; Koloa, HI.
- Data on file. CB-03-01 2017. Milan, Italy: Cassiopea SpA; 2017.
- Ju Q, Tao T, Hu T, et al. Sex hormones and acne. Clin Dermatol. 2017;35:130-137.
- Park JH, Bienenfeld A, Orlow SJ, et al. The use of hormonal antiandrogen therapy in female patients with acne: a 10-year retrospective study. Am J Clin Dermatol. 2018;19:449-455.
- Barros B, Thiboutot D. Hormonal therapies for acne. Clin Dermatol. 2017;35:168-172.
- Hebert A. Clascoterone topical cream, 1%: a novel, topical, local, selective androgen receptor antagonist: results from two phase 3 studies treating children and adult patients with facial acne vulgaris. Presented at: 2019 American Academy of Dermatology Annual Meeting; March 2, 2019; Washington, DC.
- Noreen N, Muhammad F, Akhtar B, et al. Is cannabidiol a promising substance for new drug development? a review of its potential therapeutic applications. Crit Rev Eukaryot Gene Expr. 2018;28:73-86.
- White CM. A review of human studies assessing cannabidiol’s (CBD) therapeutic actions and potential [published online February 7, 2019]. J Clin Pharmacol. 2019;59:923-934.
- Bonn-Miller MO, ElSohly MA, Loflin MJE, et al. Cannabis and cannabinoid drug development: evaluating botanical versus single molecule approaches. Int Rev Psychiatry. 2018;30:277-284.
- Bíró T, Tóth BI, Haskó G, et al. The endocannabinoid system of the skin in health and disease: novel perspectives and therapeutic opportunities. Trends Pharmacol Sci. 2009;30:411-420.
- Oláh A, Tóth BI, Borbíró I, et al. Cannabidiol exerts sebostatic and antiinflammatory effects on human sebocytes. J Clin Invest. 2014;124:3713-3724.
- Wilkinson JD, Williamson EM. Cannabinoids inhibit human keratinocyte proliferation through a non-CB1/CB2 mechanism and have a potential therapeutic value in the treatment of psoriasis. J Dermatol Sci. 2007;45:87-92.
- Appendino G, Gibbons S, Giana A, et al. Antibacterial cannabinoids from Cannabis sativa: a structure-activity study. J Nat Prod. 2008;71:1427-1430.
- Spleman L, Sinclair R, Freeman M, et al. The safety of topical cannabidiol (CBD) for the treatment of acne. J Invest Dermatol. 2018;138:S180.
Inflammation is a backdrop to the commonly cited elements of the pathophysiology of acne: Propionibacterium acnes proliferation, increased sebum production with an increase in circulating androgens, and faulty keratinization.1,2 In fact, research shows that the initiating lesion of acne vulgaris—the microcomedone—is, in essence, an inflammatory lesion.3 This realization has clearly influenced the approach to acne treatment but has not yielded a bevy of new treatments.
A better understanding of acne pathophysiology and the role of inflammation has, however, yielded a better understanding of how existing therapies treat the disease and have led to more comprehensive treatment strategies that are multitargeted. Nonetheless, topical and oral antibiotics remain mainstays of acne therapy, along with topical retinoids and benzoyl peroxide. Current guidelines of care for acne emphasize strategies that reduce dependence on antibiotics and minimize the risk for resistance.4 The therapeutic landscape might at last be shifting, with new chemical entities for acne and several novel formulations in development.
Sarecycline: A Novel Tetracycline
Tetracycline antibiotics have been used to manage acne since the 1950s, but their method of action in the disease has not been fully elucidated.5 In addition to antibiotic effects, tetracyclines have been shown to confer anti-inflammatory properties and other biologic effects.6,7
First-generation tetracycline is broad spectrum. As such, it is associated with increased potential for antibiotic resistance and greater impact on gastrointestinal health. The novel compound sarecycline is a tetracycline with a narrower spectrum of activity compared to other tetracyclines and with reduced activity against enteric gram-negative bacteria8 (Figure 1). Sarecycline recently was approved by the US Food and Drug Administration (FDA) in a once-daily oral formulation for the treatment of inflammatory lesions of nonnodular moderate to severe acne vulgaris in patients 9 years and older. Sarecycline is dosed at 1.5 mg/kg daily. The FDA approval marks the first new antibiotic approved for acne in 4 decades.

In 2 phase 3 clinical trials, sarecycline demonstrated efficacy in reducing both inflammatory and noninflammatory lesions.9 At week 12, investigator global assessment (IGA) success (≥2 point reduction in IGA and score 0 [clear] or 1 [almost clear]) rates were 21.9% and 22.6% for active treatment (n=483 and n=519), respectively, in the 2 trials compared to 10.5% and 15.3% (n=485 and n=515), respectively, for controls. Sarecycline demonstrated rapid anti-inflammatory effect. Onset of action against inflammatory lesions was notable by week 3. At week 12, inflammatory lesions were reduced in the active treatment arms by 51.8% and 49.9%, respectively, compared to 35.1% and 35.4%, respectively, for controls.9
The most common reported treatment-emergent adverse events (TEAEs) were nausea, nasopharyngitis, headache, and vomiting.9 Vestibular (dizziness, tinnitus, vertigo) and phototoxic (sunburn, photosensitivity) TEAEs both occurred in 1% or fewer of sarecycline patients. Gastrointestinal TEAE rates for sarecycline were low.9
Sarecycline also was assessed in the 2 trials for efficacy in the treatment of back and chest acne; in the active treatment group, IGA success was achieved by 29.6% and 36.6%, respectively, compared to 19.6% and 21.6%, respectively, of controls.9
Tazarotene Foam in Focus
Topical tazarotene is commercially available in cream, gel, and foam formulations. Tazarotene foam 0.1% was FDA approved in 2012 for the treatment of acne vulgaris in patients 12 years and older. However, the product was recently relaunched to the market and therefore warrants discussion.
Similar to other retinoids, topical tazarotene has been associated with the potential for application-site irritation. This aqueous foam formulation of tazarotene was designed for ease of application and to attempt to impart moisturizing effects to offset potential irritation. It contains noncomedogenic light mineral oil, which is an emollient. The foam spreads easily, including on hair-bearing skin, with demonstrated penetration of the active drug into the epidermis and dermis. Nonetheless, compared to the gel formulation of tazarotene, the foam formulation was associated with reduced systemic exposure.10
The tazarotene foam formulation does not contain alcohol, fragrance, propylene glycol, or parabens. Clinical trial participants, blinded to whether they were on active treatment or vehicle foam, consistently rated the foam formulation favorably for ease of application and spreadability, lack of stickiness or residue, and moisturizing effect. The foam vehicle is suggested to increase compliance and satisfaction in some patients.11The efficacy and tolerability of tazarotene foam 0.1% was investigated in 2 randomized, double-blind, vehicle-controlled, parallel-group studies in the United States and Canada.12 The studies involved participants aged 2 to 45 years who were randomized to receive treatment with either tazarotene foam 0.1% or vehicle foam once daily for 12 weeks (N=1486). Lesion counts, investigator static global assessment, and subject global assessment were evaluated at baseline and at weeks 2, 4, 8, and 12. At week 12, mean reduction from baseline in noninflammatory lesions was 55.9% for active treatment, mean reduction in inflammatory lesions was 56.1%, and mean total lesion reduction was 56% compared to mean reductions of 37.7%, 45.3%, and 40.8%, respectively, for vehicle. In all, 28.2% of participants achieved treatment success with active treatment compared to 14.7% of controls. There was a greater proportion of active-treatment participants with investigator static global assessment scores of 0 or 1 compared to vehicle. The only adverse events reported by more than 5% of participants in the active-treatment groups in both studies were application-site skin irritation and dryness.12
Topical Minocycline
Systemic minocycline is the most commonly prescribed oral antibiotic for acne management.13 Despite its widespread use, it is not without potential safety concerns. Minocycline is distinct among tetracyclines for posing a small risk for systemic lupus erythematosus and autoimmune TEAE.Gastrointestinal side effects and bluish discoloration also are reported.14 Topical application of minocycline for acne would optimize the therapeutic effect while reducing systemic effects. FMX101 4%, an investigational minocycline foam, is being studied for the treatment of moderate to severe acne.
In a pharmacokinetic study, minocycline exposure was 730- to 765-times lower with foam application vs oral minocycline.15 No evidence of minocycline accumulation was identified over the 21 days of application of minocycline foam 4%. Minocycline foam 4% appeared to be safe and well tolerated, without serious TEAEs, treatment-related TEAEs, or TEAEs that led to treatment discontinuation.15
In 2 identical phase 3 studies in which 961 participants were randomized (2:1) to once-daily minocycline foam 4% or foam vehicle for 12 weeks, participants in the active-treatment group demonstrated a significantly greater reduction in both inflammatory and noninflammatory lesions in both studies (both P<.05) and a greater rate of treatment success (≥2 point reduction in IGA and score of 0 [clear] or 1 [almost clear]) in 1 study. Treatment was generally safe and well tolerated, with skin-related adverse events reported in fewer than 1% of participants receiving active treatment.16
In an open-label safety extension study that enrolled 657 patients, treatment with FMX101 continued for as long as 40 weeks.17 In total, 291 participants completed 52 weeks of therapy. Rates and types of reported TEAEs in the open-label extension phase were similar to those seen in the phase 3 trials. Application-site TEAEs occurred in fewer than 2% of participants. Participants reported a high level of treatment satisfaction at week 52.17
In a more recent phase 3 study, 1507 participants were randomized (1:1) to once-daily minocycline foam 4% or foam vehicle for 12 weeks to further evaluate the efficacy and safety of FMX101 4% for moderate to severe acne vulgaris.18 The study met both primary end points: absolute change from baseline in the inflammatory lesion count (−16.93 vs −13.40; P<.0001) and the noninflammatory lesion count (−18.80 vs −15.89; P<.05), as well as percentage of participants with IGA treatment success at week 12 (30.80% vs 19.63%; P<.0001). The percentage reduction in the inflammatory lesion count was statistically significantly greater for minocycline foam 4% compared to vehicle as early as week 3 (P<.0001). The safety profile was found to be consistent with the 2 earlier phase 3 studies.18
Topical Minocycline in Rosacea
A similar foam formulation of minocycline (1.5% concentration) has shown benefit in 2 identical phase 3 studies.19 A total of 1522 participants were enrolled in 2 phase 3, randomized, multicenter, double-blind, vehicle-controlled, 2-arm studies in participants 18 years and older with moderate to severe papulopustular rosacea. Participants were randomized (2:1) to either minocycline foam 1.5% or vehicle once daily to the face for 12 weeks.19
Treatment was associated with a statistically significant reduction in counts of inflammatory lesions of rosacea (Study FX2016-11: −17.57 vs −15.65 [P=.003]; Study FX2016-12: −18.54 vs −14.88 [P<.0001]) and a significantly higher rate of IGA treatment success compared to vehicle (Study FX2016-11: 52.1% vs 43.0% [P=.027]; Study FX2016-12: 49.1% vs 39.0% [P=.008]), highlighting the anti-inflammatory action of the topically applied agent.19
The most common TEAE for both studies was upper respiratory tract infection; there were no serious TEAEs. Overall, 9 participants across both studies discontinued because of a TEAE (foam, 7 participants; vehicle, 2 participants).19
Clascoterone: First-in-Class Topical
Clascoterone cream 1% is a new chemical entity under investigation for the treatment of moderate to severe acne in patients 9 years and older. Clascoterone targets androgen receptors in the skin to block the effects of circulating endogenous androgens; chemically, it shares a 4-ring backbone identical to dihydrotestosterone and spironolactone (Figure 2). Clascoterone competes with dihydrotestosterone for binding to the androgen receptor to limit or block transcription of androgen-responsive genes and modify specific gene expression.20

Androgens are known to promote both sebum production and inflammatory responses within the follicle, contributing to the cycle of acne.21 Antiandrogen therapy would, therefore, inhibit excess sebum production and directly reduce the presence of certain inflammatory mediators in skin. This effect is expected to lead to reduced follicular plugging and a reduction in growth of P acnes and its inflammatory by-products.
Direct and indirect hormonal modulation have been successfully employed to manage acne in women; however, such therapies have not been considered first-line interventions for the disease.22 Although systemic antiandrogens and hormonal modulation are effective for certain women with acne, there may be concerns about systemic exposure23; no hormone-modulating agent has been adopted for use in men with acne.
As an androgen inhibitor, clascoterone is thought to displace androgen hormones from androgen receptors located at the sebaceous gland and hair follicle, thus inhibiting the cycle of physiologic events that leads to acne formation. Clascoterone is applied topically and acts locally on androgen receptors in the skin, with no systemic exposure seen. In phase 2 trials, clascoterone was found to be safe and effective with no systemic exposure and was suggested to have better tolerability than topical tretinoin.
Preliminary individual study analysis of data from 2 phase 3 trials showed that topical clascoterone met its primary end points, achieving statistically significantly greater rates of IGA treatment success (≥2 point reduction in IGA and score of 0 [clear] or 1 [almost clear]) at week 12 (P<.0001).24 Rates of treatment success for actively treated participants were 16.1% and 18.7%, respectively, compared to 7% and 4.7%, respectively, for vehicle. The study population included both males and nonpregnant females 9 years and older who had a baseline IGA score of 3 (moderate) or 4 (severe). At baseline, participants had a mix of inflammatory lesions (≥30, to a maximum of 75) and noninflammatory lesions (≥30, to a maximum of 100).24
Intention-to-treat analysis at week 12 showed a mean total lesion reduction from baseline for active treatment of 37.1% and 37.7%, respectively, compared to 28.5% and 22.2%, respectively, for controls.24 Mean reductions from baseline in noninflammatory lesions for active treatment were 30.7% and 29.3%, respectively, compared to 21.9% and 15.8%, respectively, for controls. Mean reductions from baseline in inflammatory lesions for active treatment were 44.8% and 47%, respectively, compared to 36.6% and 29.8%, respectively, for controls. Similarly low rates of TEAEs were reported in active and placebo groups in both studies. No TEAE suggested systemic antiandrogen exposure.24
Advancements in Cannabinoids
Advancements in pharmaceutical development of cannabinoid compounds have largely coincided with the controversial national movement to legalize medical marijuana and decriminalize recreational marijuana use. Despite the temporal connection, the 2 topics are entirely distinct. Importantly, pharmaceutical development is largely focused on the effects of cannabidiol (CBD), which is 1 of approximately 113 cannabinoids identified from Cannabis sativa. Cannabidiol is not tetrahydrocannabinol, or THC, the compound responsible for marijuana’s psychoactive effects and addictive properties; CBD does not have any psychoactive effects and is not addictive (Figure 3).25

A CBD oral solution agent recently gained FDA approval for seizures associated with Lennox-Gastaut syndrome or Dravet syndrome in patients 2 years and older; it is estimated that more than 180 trials of CBD are ongoing in the United States for various indications.26 A notable question in the development of CBD-based therapies is: What is the role of natural plant-derived CBD compared to a pure synthetic form of CBD? The latter is akin to a pharmaceutical process in which a single molecule is developed as the active drug.27 Although the potency and composition of plant-derived CBD can vary with crop conditions, plant strains, and the extraction process, a synthetic molecule would allow for consistency in safety, potency, and pharmacokinetic properties, as well as efficacy, as a consequence.26
There are intriguing data to suggest a potential use for topical CBD in the management of skin diseases, including acne vulgaris. Researchers have, for at least a decade, been investigating the role of the endocannabinoid system, which has physiologic regulatory functions in proliferation, differentiation, apoptosis and cytokine, mediator, and hormone production of various cell types in skin, hair follicles, and sebaceous glands.28 Cannabidiol has been shown to suppress proliferation of sebocytes through activation of transient receptor potential vanilloid 4 ion channels and to have anti-inflammatory effects on sebocytes.29 It has been shown to inhibit human keratinocyte proliferation through a non-CB1/CB2 mechanism30 and to possess potent antimicrobial activity against gram-positive bacteria such as P acnes.31
Given these effects on sebocytes, modulation of keratinocyte proliferation, and anti-inflammatory and antibacterial effects, CBD could prove beneficial in the management of acne vulgaris. A new synthetic CBD topical formulation, BTX 1503, is under investigation for the treatment of acne vulgaris.
Early clinical data confirm both the anti-inflammatory effects of topical BTX 1503 as well as its effects on noninflammatory lesions, with 4-week reductions in inflammatory lesion counts similar to what are reported in clinical trials for leading FDA-approved topical therapies in the same time frame.
The phase 1b trial was a 4-week, open-label study in participants with moderate to severe acne vulgaris.32 The primary end point was safety, as demonstrated by the incidence of TEAE, laboratory monitoring, and assessment of cutaneous tolerability. Exploratory end points included changes in inflammatory and noninflammatory lesion counts and IGA score. A total of 21 participants aged 18 to 65 years with moderate to severe acne vulgaris were enrolled. BTX 1503 was applied topically twice daily. At baseline, eligible participants had 20 to 50 inflammatory lesions and 20 to 100 noninflammatory acne lesions on the face, an IGA of 3 (moderate) or 4 (severe), and 3 or fewer nodular or cystic lesions (>5 mm in diameter). No serious or severe TEAEs were reported; no participants withdrew due to a TEAE. Slight erythema, slight scaling, slight dryness, and slight burning and stinging were reported; there were no reports of irritant or allergic contact dermatitis. Only 1 TEAE was thought to be possibly related to treatment: mild pain at the application site.32
In addition to presenting a potential new chemical entity for the topical treatment of acne, the novel topical vehicle formulation of BTX 1503 represents an innovative approach to drug delivery. The formulation utilizes proprietary technology to deliver high doses of drug into the skin without controversial penetration enhancers, preservatives, or other potential irritating additives. Instead, volatile excipients are used that evaporate upon application to the skin, leaving a so-called superconcentrated secondary formulation on the skin. The concentration gradient effect then drives the concentrated drug into skin. Although the formulation efficiently delivers active drug into the skin and its appendages, systemic exposure has been reported to be very low. A phase 2 randomized, double-blind, vehicle-controlled trial ongoing in the United States and Australia in 360 patients with moderate to severe acne vulgaris will provide key data to confirm the efficacy and safety of BTX 1503 (ClinicalTrials.gov Identifier NCT03573518).
Conclusion
Drug development continues to focus on the challenge of treating acne effectively and safely. Vehicle innovations are optimizing existing active drugs and creating opportunities to deliver new compounds to the skin. The approval of sarecycline as the first new chemical entity approved for acne in several years may be followed in coming years by other new actives, including clascoterone and CBD.
Inflammation is a backdrop to the commonly cited elements of the pathophysiology of acne: Propionibacterium acnes proliferation, increased sebum production with an increase in circulating androgens, and faulty keratinization.1,2 In fact, research shows that the initiating lesion of acne vulgaris—the microcomedone—is, in essence, an inflammatory lesion.3 This realization has clearly influenced the approach to acne treatment but has not yielded a bevy of new treatments.
A better understanding of acne pathophysiology and the role of inflammation has, however, yielded a better understanding of how existing therapies treat the disease and have led to more comprehensive treatment strategies that are multitargeted. Nonetheless, topical and oral antibiotics remain mainstays of acne therapy, along with topical retinoids and benzoyl peroxide. Current guidelines of care for acne emphasize strategies that reduce dependence on antibiotics and minimize the risk for resistance.4 The therapeutic landscape might at last be shifting, with new chemical entities for acne and several novel formulations in development.
Sarecycline: A Novel Tetracycline
Tetracycline antibiotics have been used to manage acne since the 1950s, but their method of action in the disease has not been fully elucidated.5 In addition to antibiotic effects, tetracyclines have been shown to confer anti-inflammatory properties and other biologic effects.6,7
First-generation tetracycline is broad spectrum. As such, it is associated with increased potential for antibiotic resistance and greater impact on gastrointestinal health. The novel compound sarecycline is a tetracycline with a narrower spectrum of activity compared to other tetracyclines and with reduced activity against enteric gram-negative bacteria8 (Figure 1). Sarecycline recently was approved by the US Food and Drug Administration (FDA) in a once-daily oral formulation for the treatment of inflammatory lesions of nonnodular moderate to severe acne vulgaris in patients 9 years and older. Sarecycline is dosed at 1.5 mg/kg daily. The FDA approval marks the first new antibiotic approved for acne in 4 decades.

In 2 phase 3 clinical trials, sarecycline demonstrated efficacy in reducing both inflammatory and noninflammatory lesions.9 At week 12, investigator global assessment (IGA) success (≥2 point reduction in IGA and score 0 [clear] or 1 [almost clear]) rates were 21.9% and 22.6% for active treatment (n=483 and n=519), respectively, in the 2 trials compared to 10.5% and 15.3% (n=485 and n=515), respectively, for controls. Sarecycline demonstrated rapid anti-inflammatory effect. Onset of action against inflammatory lesions was notable by week 3. At week 12, inflammatory lesions were reduced in the active treatment arms by 51.8% and 49.9%, respectively, compared to 35.1% and 35.4%, respectively, for controls.9
The most common reported treatment-emergent adverse events (TEAEs) were nausea, nasopharyngitis, headache, and vomiting.9 Vestibular (dizziness, tinnitus, vertigo) and phototoxic (sunburn, photosensitivity) TEAEs both occurred in 1% or fewer of sarecycline patients. Gastrointestinal TEAE rates for sarecycline were low.9
Sarecycline also was assessed in the 2 trials for efficacy in the treatment of back and chest acne; in the active treatment group, IGA success was achieved by 29.6% and 36.6%, respectively, compared to 19.6% and 21.6%, respectively, of controls.9
Tazarotene Foam in Focus
Topical tazarotene is commercially available in cream, gel, and foam formulations. Tazarotene foam 0.1% was FDA approved in 2012 for the treatment of acne vulgaris in patients 12 years and older. However, the product was recently relaunched to the market and therefore warrants discussion.
Similar to other retinoids, topical tazarotene has been associated with the potential for application-site irritation. This aqueous foam formulation of tazarotene was designed for ease of application and to attempt to impart moisturizing effects to offset potential irritation. It contains noncomedogenic light mineral oil, which is an emollient. The foam spreads easily, including on hair-bearing skin, with demonstrated penetration of the active drug into the epidermis and dermis. Nonetheless, compared to the gel formulation of tazarotene, the foam formulation was associated with reduced systemic exposure.10
The tazarotene foam formulation does not contain alcohol, fragrance, propylene glycol, or parabens. Clinical trial participants, blinded to whether they were on active treatment or vehicle foam, consistently rated the foam formulation favorably for ease of application and spreadability, lack of stickiness or residue, and moisturizing effect. The foam vehicle is suggested to increase compliance and satisfaction in some patients.11The efficacy and tolerability of tazarotene foam 0.1% was investigated in 2 randomized, double-blind, vehicle-controlled, parallel-group studies in the United States and Canada.12 The studies involved participants aged 2 to 45 years who were randomized to receive treatment with either tazarotene foam 0.1% or vehicle foam once daily for 12 weeks (N=1486). Lesion counts, investigator static global assessment, and subject global assessment were evaluated at baseline and at weeks 2, 4, 8, and 12. At week 12, mean reduction from baseline in noninflammatory lesions was 55.9% for active treatment, mean reduction in inflammatory lesions was 56.1%, and mean total lesion reduction was 56% compared to mean reductions of 37.7%, 45.3%, and 40.8%, respectively, for vehicle. In all, 28.2% of participants achieved treatment success with active treatment compared to 14.7% of controls. There was a greater proportion of active-treatment participants with investigator static global assessment scores of 0 or 1 compared to vehicle. The only adverse events reported by more than 5% of participants in the active-treatment groups in both studies were application-site skin irritation and dryness.12
Topical Minocycline
Systemic minocycline is the most commonly prescribed oral antibiotic for acne management.13 Despite its widespread use, it is not without potential safety concerns. Minocycline is distinct among tetracyclines for posing a small risk for systemic lupus erythematosus and autoimmune TEAE.Gastrointestinal side effects and bluish discoloration also are reported.14 Topical application of minocycline for acne would optimize the therapeutic effect while reducing systemic effects. FMX101 4%, an investigational minocycline foam, is being studied for the treatment of moderate to severe acne.
In a pharmacokinetic study, minocycline exposure was 730- to 765-times lower with foam application vs oral minocycline.15 No evidence of minocycline accumulation was identified over the 21 days of application of minocycline foam 4%. Minocycline foam 4% appeared to be safe and well tolerated, without serious TEAEs, treatment-related TEAEs, or TEAEs that led to treatment discontinuation.15
In 2 identical phase 3 studies in which 961 participants were randomized (2:1) to once-daily minocycline foam 4% or foam vehicle for 12 weeks, participants in the active-treatment group demonstrated a significantly greater reduction in both inflammatory and noninflammatory lesions in both studies (both P<.05) and a greater rate of treatment success (≥2 point reduction in IGA and score of 0 [clear] or 1 [almost clear]) in 1 study. Treatment was generally safe and well tolerated, with skin-related adverse events reported in fewer than 1% of participants receiving active treatment.16
In an open-label safety extension study that enrolled 657 patients, treatment with FMX101 continued for as long as 40 weeks.17 In total, 291 participants completed 52 weeks of therapy. Rates and types of reported TEAEs in the open-label extension phase were similar to those seen in the phase 3 trials. Application-site TEAEs occurred in fewer than 2% of participants. Participants reported a high level of treatment satisfaction at week 52.17
In a more recent phase 3 study, 1507 participants were randomized (1:1) to once-daily minocycline foam 4% or foam vehicle for 12 weeks to further evaluate the efficacy and safety of FMX101 4% for moderate to severe acne vulgaris.18 The study met both primary end points: absolute change from baseline in the inflammatory lesion count (−16.93 vs −13.40; P<.0001) and the noninflammatory lesion count (−18.80 vs −15.89; P<.05), as well as percentage of participants with IGA treatment success at week 12 (30.80% vs 19.63%; P<.0001). The percentage reduction in the inflammatory lesion count was statistically significantly greater for minocycline foam 4% compared to vehicle as early as week 3 (P<.0001). The safety profile was found to be consistent with the 2 earlier phase 3 studies.18
Topical Minocycline in Rosacea
A similar foam formulation of minocycline (1.5% concentration) has shown benefit in 2 identical phase 3 studies.19 A total of 1522 participants were enrolled in 2 phase 3, randomized, multicenter, double-blind, vehicle-controlled, 2-arm studies in participants 18 years and older with moderate to severe papulopustular rosacea. Participants were randomized (2:1) to either minocycline foam 1.5% or vehicle once daily to the face for 12 weeks.19
Treatment was associated with a statistically significant reduction in counts of inflammatory lesions of rosacea (Study FX2016-11: −17.57 vs −15.65 [P=.003]; Study FX2016-12: −18.54 vs −14.88 [P<.0001]) and a significantly higher rate of IGA treatment success compared to vehicle (Study FX2016-11: 52.1% vs 43.0% [P=.027]; Study FX2016-12: 49.1% vs 39.0% [P=.008]), highlighting the anti-inflammatory action of the topically applied agent.19
The most common TEAE for both studies was upper respiratory tract infection; there were no serious TEAEs. Overall, 9 participants across both studies discontinued because of a TEAE (foam, 7 participants; vehicle, 2 participants).19
Clascoterone: First-in-Class Topical
Clascoterone cream 1% is a new chemical entity under investigation for the treatment of moderate to severe acne in patients 9 years and older. Clascoterone targets androgen receptors in the skin to block the effects of circulating endogenous androgens; chemically, it shares a 4-ring backbone identical to dihydrotestosterone and spironolactone (Figure 2). Clascoterone competes with dihydrotestosterone for binding to the androgen receptor to limit or block transcription of androgen-responsive genes and modify specific gene expression.20

Androgens are known to promote both sebum production and inflammatory responses within the follicle, contributing to the cycle of acne.21 Antiandrogen therapy would, therefore, inhibit excess sebum production and directly reduce the presence of certain inflammatory mediators in skin. This effect is expected to lead to reduced follicular plugging and a reduction in growth of P acnes and its inflammatory by-products.
Direct and indirect hormonal modulation have been successfully employed to manage acne in women; however, such therapies have not been considered first-line interventions for the disease.22 Although systemic antiandrogens and hormonal modulation are effective for certain women with acne, there may be concerns about systemic exposure23; no hormone-modulating agent has been adopted for use in men with acne.
As an androgen inhibitor, clascoterone is thought to displace androgen hormones from androgen receptors located at the sebaceous gland and hair follicle, thus inhibiting the cycle of physiologic events that leads to acne formation. Clascoterone is applied topically and acts locally on androgen receptors in the skin, with no systemic exposure seen. In phase 2 trials, clascoterone was found to be safe and effective with no systemic exposure and was suggested to have better tolerability than topical tretinoin.
Preliminary individual study analysis of data from 2 phase 3 trials showed that topical clascoterone met its primary end points, achieving statistically significantly greater rates of IGA treatment success (≥2 point reduction in IGA and score of 0 [clear] or 1 [almost clear]) at week 12 (P<.0001).24 Rates of treatment success for actively treated participants were 16.1% and 18.7%, respectively, compared to 7% and 4.7%, respectively, for vehicle. The study population included both males and nonpregnant females 9 years and older who had a baseline IGA score of 3 (moderate) or 4 (severe). At baseline, participants had a mix of inflammatory lesions (≥30, to a maximum of 75) and noninflammatory lesions (≥30, to a maximum of 100).24
Intention-to-treat analysis at week 12 showed a mean total lesion reduction from baseline for active treatment of 37.1% and 37.7%, respectively, compared to 28.5% and 22.2%, respectively, for controls.24 Mean reductions from baseline in noninflammatory lesions for active treatment were 30.7% and 29.3%, respectively, compared to 21.9% and 15.8%, respectively, for controls. Mean reductions from baseline in inflammatory lesions for active treatment were 44.8% and 47%, respectively, compared to 36.6% and 29.8%, respectively, for controls. Similarly low rates of TEAEs were reported in active and placebo groups in both studies. No TEAE suggested systemic antiandrogen exposure.24
Advancements in Cannabinoids
Advancements in pharmaceutical development of cannabinoid compounds have largely coincided with the controversial national movement to legalize medical marijuana and decriminalize recreational marijuana use. Despite the temporal connection, the 2 topics are entirely distinct. Importantly, pharmaceutical development is largely focused on the effects of cannabidiol (CBD), which is 1 of approximately 113 cannabinoids identified from Cannabis sativa. Cannabidiol is not tetrahydrocannabinol, or THC, the compound responsible for marijuana’s psychoactive effects and addictive properties; CBD does not have any psychoactive effects and is not addictive (Figure 3).25

A CBD oral solution agent recently gained FDA approval for seizures associated with Lennox-Gastaut syndrome or Dravet syndrome in patients 2 years and older; it is estimated that more than 180 trials of CBD are ongoing in the United States for various indications.26 A notable question in the development of CBD-based therapies is: What is the role of natural plant-derived CBD compared to a pure synthetic form of CBD? The latter is akin to a pharmaceutical process in which a single molecule is developed as the active drug.27 Although the potency and composition of plant-derived CBD can vary with crop conditions, plant strains, and the extraction process, a synthetic molecule would allow for consistency in safety, potency, and pharmacokinetic properties, as well as efficacy, as a consequence.26
There are intriguing data to suggest a potential use for topical CBD in the management of skin diseases, including acne vulgaris. Researchers have, for at least a decade, been investigating the role of the endocannabinoid system, which has physiologic regulatory functions in proliferation, differentiation, apoptosis and cytokine, mediator, and hormone production of various cell types in skin, hair follicles, and sebaceous glands.28 Cannabidiol has been shown to suppress proliferation of sebocytes through activation of transient receptor potential vanilloid 4 ion channels and to have anti-inflammatory effects on sebocytes.29 It has been shown to inhibit human keratinocyte proliferation through a non-CB1/CB2 mechanism30 and to possess potent antimicrobial activity against gram-positive bacteria such as P acnes.31
Given these effects on sebocytes, modulation of keratinocyte proliferation, and anti-inflammatory and antibacterial effects, CBD could prove beneficial in the management of acne vulgaris. A new synthetic CBD topical formulation, BTX 1503, is under investigation for the treatment of acne vulgaris.
Early clinical data confirm both the anti-inflammatory effects of topical BTX 1503 as well as its effects on noninflammatory lesions, with 4-week reductions in inflammatory lesion counts similar to what are reported in clinical trials for leading FDA-approved topical therapies in the same time frame.
The phase 1b trial was a 4-week, open-label study in participants with moderate to severe acne vulgaris.32 The primary end point was safety, as demonstrated by the incidence of TEAE, laboratory monitoring, and assessment of cutaneous tolerability. Exploratory end points included changes in inflammatory and noninflammatory lesion counts and IGA score. A total of 21 participants aged 18 to 65 years with moderate to severe acne vulgaris were enrolled. BTX 1503 was applied topically twice daily. At baseline, eligible participants had 20 to 50 inflammatory lesions and 20 to 100 noninflammatory acne lesions on the face, an IGA of 3 (moderate) or 4 (severe), and 3 or fewer nodular or cystic lesions (>5 mm in diameter). No serious or severe TEAEs were reported; no participants withdrew due to a TEAE. Slight erythema, slight scaling, slight dryness, and slight burning and stinging were reported; there were no reports of irritant or allergic contact dermatitis. Only 1 TEAE was thought to be possibly related to treatment: mild pain at the application site.32
In addition to presenting a potential new chemical entity for the topical treatment of acne, the novel topical vehicle formulation of BTX 1503 represents an innovative approach to drug delivery. The formulation utilizes proprietary technology to deliver high doses of drug into the skin without controversial penetration enhancers, preservatives, or other potential irritating additives. Instead, volatile excipients are used that evaporate upon application to the skin, leaving a so-called superconcentrated secondary formulation on the skin. The concentration gradient effect then drives the concentrated drug into skin. Although the formulation efficiently delivers active drug into the skin and its appendages, systemic exposure has been reported to be very low. A phase 2 randomized, double-blind, vehicle-controlled trial ongoing in the United States and Australia in 360 patients with moderate to severe acne vulgaris will provide key data to confirm the efficacy and safety of BTX 1503 (ClinicalTrials.gov Identifier NCT03573518).
Conclusion
Drug development continues to focus on the challenge of treating acne effectively and safely. Vehicle innovations are optimizing existing active drugs and creating opportunities to deliver new compounds to the skin. The approval of sarecycline as the first new chemical entity approved for acne in several years may be followed in coming years by other new actives, including clascoterone and CBD.
- Webster GF. The pathophysiology of acne. Cutis. 2005;76(2 suppl):4-7.
- Burkhart CN, Gottwald L. Assessment of etiologic agents in acne pathogenesis. Skinmed. 2003;2:222-228.
- Kang S, Cho S, Chung JH, et al. Inflammation and extracellular matrix degradation mediated by activated transcription factors nuclear factor-kappaB and activator protein-1 in inflammatory acne lesions in vivo. Am J Pathol. 2005;166:1691-1699.
- Zaenglein AL, Pathy AL, Schlosser BJ, et al. Guidelines of care for the management of acne vulgaris. J Am Acad Dermatol. 2016;74:945-973.
- Garrido-Mesa N, Zarzuelo A, Gálvez J. Minocycline: far beyond an antibiotic. Br J Pharmacol. 2013;169:337-352.
- Griffin MO, Ceballos G, Villarreal FJ. Tetracycline compounds with non-antimicrobial organ protective properties: possible mechanisms of action. Pharmacol Res. 2011;63:102-107.
- Weinberg JM. The anti-inflammatory effects of tetracyclines. Cutis. 2005;75(4 suppl):6-11.
- Leyden JJ, Sniukiene V, Berk DR, et al. Efficacy and safety of sarecycline, a novel, once-daily, narrow spectrum antibiotic for the treatment of moderate to severe facial acne vulgaris: results of a phase 2, dose-ranging study. J Drugs Dermatol. 2018;17:333-338.
- Moore A, Green LJ, Bruce S, et al. Once-daily oral sarecycline 1.5 mg/kg/day is effective for moderate to severe acne vulgaris: results from two identically designed, phase 3, randomized, double-blind clinical trials. J Drugs Dermatol. 2018;17:987-996.
- Jarratt M, Werner CP, Alió Saenz AB. Tazarotene foam versus tazarotene gel: a randomized relative bioavailability study in acne vulgaris. Clin Drug Investig. 2013;33:283-289.
- Smith JA, Narahari S, Hill D, et al. Tazarotene foam, 0.1%, for the treatment of acne. Expert Opin Drug Saf. 2016;15:99-103.
- Feldman SR, Werner CP, Alió Saenz AB. The efficacy and tolerability of tazarotene foam, 0.1%, in the treatment of acne vulgaris in 2 multicenter, randomized, vehicle-controlled, double-blind studies. J Drugs Dermatol. 2013;12:438-446.
- Lee YH, Liu G, Thiboutot DM, et al. A retrospective analysis of the duration of oral antibiotic therapy for the treatment of acne among adolescents: investigating practice gaps and potential cost-savings. J Am Acad Dermatol. 2014;71:70-76.
- Garner SE, Eady A, Bennett C, et al. Minocycline for acne vulgaris: efficacy and safety. Cochrane Database Syst Rev. 2012(8):CD002086.
- Jones TM, Ellman H, deVries T. Pharmacokinetic comparison of once-daily topical minocycline foam 4% vs oral minocycline for moderate-to-severe acne. J Drugs Dermatol. 2017;16:1022-1028.
- Gold LS, Dhawan S, Weiss J, et al. A novel topical minocycline foam for the treatment of moderate-to-severe acne vulgaris: results of 2 randomized, double-blind, phase 3 studies. J Am Acad Dermatol. 2019;80:168-177.17.
- Gold LS, Dhawan S, Weiss J, et al. FMX101 4% minocycline foam for the treatment of acne vulgaris: safety and patient satisfaction from the open-label extension of 2 phase 3 studies. Poster presented at: 2018 Winter Clinical Dermatology Conference; January 12-17, 2018; Maui, HI.
- Raoof J, Hooper D, Moore A, et al. FMX101 4% topical minocycline foam for the treatment of moderate to severe acne vulgaris: efficacy and safety from a phase 3 randomized, double-blind, vehicle-controlled study. Poster presented at: 2018 Fall Clinical Dermatology Conference; October 18-21, 2018; Las Vegas, NV.
- Gold LS, Del Rosso JQ, Bhatia ND, et al. Efficacy and safety of FMX103 (1.5% minocycline foam) in the treatment of moderate-to-severe papulopustular rosacea: results from two phase 3 randomized, multicenter, double-blind, vehicle-controlled studies. Poster presented at: 2019 Winter Clinical Dermatology Conference; January 18-23; 2019; Koloa, HI.
- Data on file. CB-03-01 2017. Milan, Italy: Cassiopea SpA; 2017.
- Ju Q, Tao T, Hu T, et al. Sex hormones and acne. Clin Dermatol. 2017;35:130-137.
- Park JH, Bienenfeld A, Orlow SJ, et al. The use of hormonal antiandrogen therapy in female patients with acne: a 10-year retrospective study. Am J Clin Dermatol. 2018;19:449-455.
- Barros B, Thiboutot D. Hormonal therapies for acne. Clin Dermatol. 2017;35:168-172.
- Hebert A. Clascoterone topical cream, 1%: a novel, topical, local, selective androgen receptor antagonist: results from two phase 3 studies treating children and adult patients with facial acne vulgaris. Presented at: 2019 American Academy of Dermatology Annual Meeting; March 2, 2019; Washington, DC.
- Noreen N, Muhammad F, Akhtar B, et al. Is cannabidiol a promising substance for new drug development? a review of its potential therapeutic applications. Crit Rev Eukaryot Gene Expr. 2018;28:73-86.
- White CM. A review of human studies assessing cannabidiol’s (CBD) therapeutic actions and potential [published online February 7, 2019]. J Clin Pharmacol. 2019;59:923-934.
- Bonn-Miller MO, ElSohly MA, Loflin MJE, et al. Cannabis and cannabinoid drug development: evaluating botanical versus single molecule approaches. Int Rev Psychiatry. 2018;30:277-284.
- Bíró T, Tóth BI, Haskó G, et al. The endocannabinoid system of the skin in health and disease: novel perspectives and therapeutic opportunities. Trends Pharmacol Sci. 2009;30:411-420.
- Oláh A, Tóth BI, Borbíró I, et al. Cannabidiol exerts sebostatic and antiinflammatory effects on human sebocytes. J Clin Invest. 2014;124:3713-3724.
- Wilkinson JD, Williamson EM. Cannabinoids inhibit human keratinocyte proliferation through a non-CB1/CB2 mechanism and have a potential therapeutic value in the treatment of psoriasis. J Dermatol Sci. 2007;45:87-92.
- Appendino G, Gibbons S, Giana A, et al. Antibacterial cannabinoids from Cannabis sativa: a structure-activity study. J Nat Prod. 2008;71:1427-1430.
- Spleman L, Sinclair R, Freeman M, et al. The safety of topical cannabidiol (CBD) for the treatment of acne. J Invest Dermatol. 2018;138:S180.
- Webster GF. The pathophysiology of acne. Cutis. 2005;76(2 suppl):4-7.
- Burkhart CN, Gottwald L. Assessment of etiologic agents in acne pathogenesis. Skinmed. 2003;2:222-228.
- Kang S, Cho S, Chung JH, et al. Inflammation and extracellular matrix degradation mediated by activated transcription factors nuclear factor-kappaB and activator protein-1 in inflammatory acne lesions in vivo. Am J Pathol. 2005;166:1691-1699.
- Zaenglein AL, Pathy AL, Schlosser BJ, et al. Guidelines of care for the management of acne vulgaris. J Am Acad Dermatol. 2016;74:945-973.
- Garrido-Mesa N, Zarzuelo A, Gálvez J. Minocycline: far beyond an antibiotic. Br J Pharmacol. 2013;169:337-352.
- Griffin MO, Ceballos G, Villarreal FJ. Tetracycline compounds with non-antimicrobial organ protective properties: possible mechanisms of action. Pharmacol Res. 2011;63:102-107.
- Weinberg JM. The anti-inflammatory effects of tetracyclines. Cutis. 2005;75(4 suppl):6-11.
- Leyden JJ, Sniukiene V, Berk DR, et al. Efficacy and safety of sarecycline, a novel, once-daily, narrow spectrum antibiotic for the treatment of moderate to severe facial acne vulgaris: results of a phase 2, dose-ranging study. J Drugs Dermatol. 2018;17:333-338.
- Moore A, Green LJ, Bruce S, et al. Once-daily oral sarecycline 1.5 mg/kg/day is effective for moderate to severe acne vulgaris: results from two identically designed, phase 3, randomized, double-blind clinical trials. J Drugs Dermatol. 2018;17:987-996.
- Jarratt M, Werner CP, Alió Saenz AB. Tazarotene foam versus tazarotene gel: a randomized relative bioavailability study in acne vulgaris. Clin Drug Investig. 2013;33:283-289.
- Smith JA, Narahari S, Hill D, et al. Tazarotene foam, 0.1%, for the treatment of acne. Expert Opin Drug Saf. 2016;15:99-103.
- Feldman SR, Werner CP, Alió Saenz AB. The efficacy and tolerability of tazarotene foam, 0.1%, in the treatment of acne vulgaris in 2 multicenter, randomized, vehicle-controlled, double-blind studies. J Drugs Dermatol. 2013;12:438-446.
- Lee YH, Liu G, Thiboutot DM, et al. A retrospective analysis of the duration of oral antibiotic therapy for the treatment of acne among adolescents: investigating practice gaps and potential cost-savings. J Am Acad Dermatol. 2014;71:70-76.
- Garner SE, Eady A, Bennett C, et al. Minocycline for acne vulgaris: efficacy and safety. Cochrane Database Syst Rev. 2012(8):CD002086.
- Jones TM, Ellman H, deVries T. Pharmacokinetic comparison of once-daily topical minocycline foam 4% vs oral minocycline for moderate-to-severe acne. J Drugs Dermatol. 2017;16:1022-1028.
- Gold LS, Dhawan S, Weiss J, et al. A novel topical minocycline foam for the treatment of moderate-to-severe acne vulgaris: results of 2 randomized, double-blind, phase 3 studies. J Am Acad Dermatol. 2019;80:168-177.17.
- Gold LS, Dhawan S, Weiss J, et al. FMX101 4% minocycline foam for the treatment of acne vulgaris: safety and patient satisfaction from the open-label extension of 2 phase 3 studies. Poster presented at: 2018 Winter Clinical Dermatology Conference; January 12-17, 2018; Maui, HI.
- Raoof J, Hooper D, Moore A, et al. FMX101 4% topical minocycline foam for the treatment of moderate to severe acne vulgaris: efficacy and safety from a phase 3 randomized, double-blind, vehicle-controlled study. Poster presented at: 2018 Fall Clinical Dermatology Conference; October 18-21, 2018; Las Vegas, NV.
- Gold LS, Del Rosso JQ, Bhatia ND, et al. Efficacy and safety of FMX103 (1.5% minocycline foam) in the treatment of moderate-to-severe papulopustular rosacea: results from two phase 3 randomized, multicenter, double-blind, vehicle-controlled studies. Poster presented at: 2019 Winter Clinical Dermatology Conference; January 18-23; 2019; Koloa, HI.
- Data on file. CB-03-01 2017. Milan, Italy: Cassiopea SpA; 2017.
- Ju Q, Tao T, Hu T, et al. Sex hormones and acne. Clin Dermatol. 2017;35:130-137.
- Park JH, Bienenfeld A, Orlow SJ, et al. The use of hormonal antiandrogen therapy in female patients with acne: a 10-year retrospective study. Am J Clin Dermatol. 2018;19:449-455.
- Barros B, Thiboutot D. Hormonal therapies for acne. Clin Dermatol. 2017;35:168-172.
- Hebert A. Clascoterone topical cream, 1%: a novel, topical, local, selective androgen receptor antagonist: results from two phase 3 studies treating children and adult patients with facial acne vulgaris. Presented at: 2019 American Academy of Dermatology Annual Meeting; March 2, 2019; Washington, DC.
- Noreen N, Muhammad F, Akhtar B, et al. Is cannabidiol a promising substance for new drug development? a review of its potential therapeutic applications. Crit Rev Eukaryot Gene Expr. 2018;28:73-86.
- White CM. A review of human studies assessing cannabidiol’s (CBD) therapeutic actions and potential [published online February 7, 2019]. J Clin Pharmacol. 2019;59:923-934.
- Bonn-Miller MO, ElSohly MA, Loflin MJE, et al. Cannabis and cannabinoid drug development: evaluating botanical versus single molecule approaches. Int Rev Psychiatry. 2018;30:277-284.
- Bíró T, Tóth BI, Haskó G, et al. The endocannabinoid system of the skin in health and disease: novel perspectives and therapeutic opportunities. Trends Pharmacol Sci. 2009;30:411-420.
- Oláh A, Tóth BI, Borbíró I, et al. Cannabidiol exerts sebostatic and antiinflammatory effects on human sebocytes. J Clin Invest. 2014;124:3713-3724.
- Wilkinson JD, Williamson EM. Cannabinoids inhibit human keratinocyte proliferation through a non-CB1/CB2 mechanism and have a potential therapeutic value in the treatment of psoriasis. J Dermatol Sci. 2007;45:87-92.
- Appendino G, Gibbons S, Giana A, et al. Antibacterial cannabinoids from Cannabis sativa: a structure-activity study. J Nat Prod. 2008;71:1427-1430.
- Spleman L, Sinclair R, Freeman M, et al. The safety of topical cannabidiol (CBD) for the treatment of acne. J Invest Dermatol. 2018;138:S180.
Practice Points
- Sarecycline is the first new antibiotic approved for acne in several years.
- Tazarotene foam 0.1% was relaunched to the market. The foam formulation attempts to impart moisturizing effects to offset potential irritation.
- Topical minocycline for acne optimizes the therapeutic effects while reducing systemic effects.
- Clascoterone and cannabidiol currently are under investigation for acne treatment.
Acne in women: What new insights tell us
NEWPORT BEACH, CALIF. – When it comes to acne in adult women, look past the jawline, beyond traditional medications, and toward greater control. That’s the message of a dermatologist who spoke at Skin Disease Education Foundation’s Women’s & Pediatric Dermatology Seminar.
“We should be aiming to get our patients to clear or almost clear, and we have the tools necessary to help that happen,” said Linda Stein Gold, MD, director of dermatology research at Henry Ford Hospital in Detroit.
she noted. Acne appears to affect 51% of women aged 20-29 years, she said, and prevalence dips to 15% in women older than 50 years.
About 80% of cases continue from adolescence, compared with about 20% that are new-onset during adulthood, she said. According to studies, she added, “most adult women have acne on multiple different areas of their face, not just the jawline. It’s similar to what we see in the adolescent population.”
Dr. Stein Gold offered these tips about treatment in this group of patients:
Inflammation
Researchers now consider that “all acne is inflammatory acne.” Be aggressive with anti-inflammatory treatment, and “continue even after the lesion is resolved” if needed to prevent scarring.
Oral contraceptives (OCs)
OCs can be helpful, but “we have to proceed with caution,” she said. A 2012 Cochrane Library review of 31 trials found that six combination OCs (COCs) “evaluated in placebo-controlled trials are effective in reducing inflammatory and noninflammatory facial acne lesions. Few important and consistent differences were found between COC types in their effectiveness for treating acne,” the review concluded (Cochrane Database Syst Rev. 2012 Jul 11;[7]:CD004425).
Results take time, however, and it “can take 3 months to see an effect, and 6 months for full effect,” Dr. Stein Gold noted.
There are multiple contraindications to the use of OCs, and they’ve been linked – controversially – to an increased risk of blood clots and breast cancer. However, risk of thrombosis also spikes – to significantly higher levels than with OC use – during pregnancy and the postpartum period, she said.
Spironolactone
This antihypertensive drug can be helpful, Dr. Stein Gold noted, although the one study in a 2009 Cochrane review that had acne as an outcome failed to find evidence of efficacy versus placebo (Cochrane Database Syst Rev. 2009 Apr 15;[2]:CD000194). Be aware of the boxed warning about links to cancer in rat studies, and consider the risk of potassium elevation in certain populations, she added. Watch the dose: fewer side effects are seen at 50-100 mg daily, although they’re still common, and it can take 3 months or more for improvements to appear, she said.
Truncal acne
Patients may be hesitant to mention they have acne on their chest and back. “They may not tell you about it, and you may not ask about it but [some patients] expect you to know about it and treat it,” Dr. Stein Gold said. She referred to trifarotene, a topical retinoid cream that, although not yet approved, appears to be safe and effective in treating acne on the face and trunk in phase 3 studies.
“Some people will say the trunk will get too irritated if you put a retinoid on it. But it absolutely can be used on the chest and back. The first thing I say to my patients is to expect to have redness and scaling for first 2 weeks. People pay money for that. It’s a chemical peel! It’s okay to have some sloughing; use an oil-free moisturizer.”
Dr. Stein Gold disclosed relationships with Galderma, Foamix, and Sol Gel (investigator, consultant); Valeant (consultant, speaker); and Dermira (investigator, speaker).
SDEF and this news organization are owned by the same parent company.
NEWPORT BEACH, CALIF. – When it comes to acne in adult women, look past the jawline, beyond traditional medications, and toward greater control. That’s the message of a dermatologist who spoke at Skin Disease Education Foundation’s Women’s & Pediatric Dermatology Seminar.
“We should be aiming to get our patients to clear or almost clear, and we have the tools necessary to help that happen,” said Linda Stein Gold, MD, director of dermatology research at Henry Ford Hospital in Detroit.
she noted. Acne appears to affect 51% of women aged 20-29 years, she said, and prevalence dips to 15% in women older than 50 years.
About 80% of cases continue from adolescence, compared with about 20% that are new-onset during adulthood, she said. According to studies, she added, “most adult women have acne on multiple different areas of their face, not just the jawline. It’s similar to what we see in the adolescent population.”
Dr. Stein Gold offered these tips about treatment in this group of patients:
Inflammation
Researchers now consider that “all acne is inflammatory acne.” Be aggressive with anti-inflammatory treatment, and “continue even after the lesion is resolved” if needed to prevent scarring.
Oral contraceptives (OCs)
OCs can be helpful, but “we have to proceed with caution,” she said. A 2012 Cochrane Library review of 31 trials found that six combination OCs (COCs) “evaluated in placebo-controlled trials are effective in reducing inflammatory and noninflammatory facial acne lesions. Few important and consistent differences were found between COC types in their effectiveness for treating acne,” the review concluded (Cochrane Database Syst Rev. 2012 Jul 11;[7]:CD004425).
Results take time, however, and it “can take 3 months to see an effect, and 6 months for full effect,” Dr. Stein Gold noted.
There are multiple contraindications to the use of OCs, and they’ve been linked – controversially – to an increased risk of blood clots and breast cancer. However, risk of thrombosis also spikes – to significantly higher levels than with OC use – during pregnancy and the postpartum period, she said.
Spironolactone
This antihypertensive drug can be helpful, Dr. Stein Gold noted, although the one study in a 2009 Cochrane review that had acne as an outcome failed to find evidence of efficacy versus placebo (Cochrane Database Syst Rev. 2009 Apr 15;[2]:CD000194). Be aware of the boxed warning about links to cancer in rat studies, and consider the risk of potassium elevation in certain populations, she added. Watch the dose: fewer side effects are seen at 50-100 mg daily, although they’re still common, and it can take 3 months or more for improvements to appear, she said.
Truncal acne
Patients may be hesitant to mention they have acne on their chest and back. “They may not tell you about it, and you may not ask about it but [some patients] expect you to know about it and treat it,” Dr. Stein Gold said. She referred to trifarotene, a topical retinoid cream that, although not yet approved, appears to be safe and effective in treating acne on the face and trunk in phase 3 studies.
“Some people will say the trunk will get too irritated if you put a retinoid on it. But it absolutely can be used on the chest and back. The first thing I say to my patients is to expect to have redness and scaling for first 2 weeks. People pay money for that. It’s a chemical peel! It’s okay to have some sloughing; use an oil-free moisturizer.”
Dr. Stein Gold disclosed relationships with Galderma, Foamix, and Sol Gel (investigator, consultant); Valeant (consultant, speaker); and Dermira (investigator, speaker).
SDEF and this news organization are owned by the same parent company.
NEWPORT BEACH, CALIF. – When it comes to acne in adult women, look past the jawline, beyond traditional medications, and toward greater control. That’s the message of a dermatologist who spoke at Skin Disease Education Foundation’s Women’s & Pediatric Dermatology Seminar.
“We should be aiming to get our patients to clear or almost clear, and we have the tools necessary to help that happen,” said Linda Stein Gold, MD, director of dermatology research at Henry Ford Hospital in Detroit.
she noted. Acne appears to affect 51% of women aged 20-29 years, she said, and prevalence dips to 15% in women older than 50 years.
About 80% of cases continue from adolescence, compared with about 20% that are new-onset during adulthood, she said. According to studies, she added, “most adult women have acne on multiple different areas of their face, not just the jawline. It’s similar to what we see in the adolescent population.”
Dr. Stein Gold offered these tips about treatment in this group of patients:
Inflammation
Researchers now consider that “all acne is inflammatory acne.” Be aggressive with anti-inflammatory treatment, and “continue even after the lesion is resolved” if needed to prevent scarring.
Oral contraceptives (OCs)
OCs can be helpful, but “we have to proceed with caution,” she said. A 2012 Cochrane Library review of 31 trials found that six combination OCs (COCs) “evaluated in placebo-controlled trials are effective in reducing inflammatory and noninflammatory facial acne lesions. Few important and consistent differences were found between COC types in their effectiveness for treating acne,” the review concluded (Cochrane Database Syst Rev. 2012 Jul 11;[7]:CD004425).
Results take time, however, and it “can take 3 months to see an effect, and 6 months for full effect,” Dr. Stein Gold noted.
There are multiple contraindications to the use of OCs, and they’ve been linked – controversially – to an increased risk of blood clots and breast cancer. However, risk of thrombosis also spikes – to significantly higher levels than with OC use – during pregnancy and the postpartum period, she said.
Spironolactone
This antihypertensive drug can be helpful, Dr. Stein Gold noted, although the one study in a 2009 Cochrane review that had acne as an outcome failed to find evidence of efficacy versus placebo (Cochrane Database Syst Rev. 2009 Apr 15;[2]:CD000194). Be aware of the boxed warning about links to cancer in rat studies, and consider the risk of potassium elevation in certain populations, she added. Watch the dose: fewer side effects are seen at 50-100 mg daily, although they’re still common, and it can take 3 months or more for improvements to appear, she said.
Truncal acne
Patients may be hesitant to mention they have acne on their chest and back. “They may not tell you about it, and you may not ask about it but [some patients] expect you to know about it and treat it,” Dr. Stein Gold said. She referred to trifarotene, a topical retinoid cream that, although not yet approved, appears to be safe and effective in treating acne on the face and trunk in phase 3 studies.
“Some people will say the trunk will get too irritated if you put a retinoid on it. But it absolutely can be used on the chest and back. The first thing I say to my patients is to expect to have redness and scaling for first 2 weeks. People pay money for that. It’s a chemical peel! It’s okay to have some sloughing; use an oil-free moisturizer.”
Dr. Stein Gold disclosed relationships with Galderma, Foamix, and Sol Gel (investigator, consultant); Valeant (consultant, speaker); and Dermira (investigator, speaker).
SDEF and this news organization are owned by the same parent company.
EXPERT ANALYSIS FROM SDEF WOMEN’S & PEDIATRIC DERMATOLOGY SEMINAR
No increased risk of psychiatric problems tied to isotretinoin
Isotretinoin use may increase vulnerability to psychiatric conditions, but available evidence does not support a causal relationship, on the basis of data from a retrospective study of 17,829 psychiatric adverse events reported to the Food and Drug Administration over 2 decades.
“Although one study highlighted consistent reporting of depression and suicide in patients taking isotretinoin in the United States from 1982 to 2000, few studies have examined reports of psychiatric adverse events at the national level since 2000,” wrote Sean Singer of Harvard University, Boston, and his colleagues.
In a study published in JAMA Dermatology, the researchers reviewed data from the FDA’s Adverse Event Reporting System between 1997 and 2017.
A total of 17,829 psychiatric adverse events in which isotretinoin was the primary suspect drug were reported during the study period. The researchers classified the events into 12 categories; the most common were depressive disorders (42%), emotional lability (17%), and anxiety (14%). The number of reported psychiatric adverse events was similar between men and women (8,936 and 8,362 events, respectively).
The researchers also identified 2,278 reports of suicidal ideation, 602 reports of attempted suicide, and 368 reports of completed suicide.
In addition, the researchers examined data from the iPLEDGE program and found completed suicide rates of 8.4 per 100,000 patients in 2009 and 5.6 per 100,000 patients in 2010. However, these rates were lower than national suicide rates in the general population of 11.8 per 100,000 people in 2009 and 12.1 per 100,000 people in 2010.
Patient age was available for 13,553 adverse event reports, and patients aged 10-19 years accounted for 53% of the reports overall and 58% of completed suicides for which age was reported.
The high number of psychiatric adverse events in the youngest age group “could reflect more isotretinoin prescriptions in this age group or may suggest that teenagers are particularly vulnerable to psychiatric adverse events while taking isotretinoin,” the researchers said.
The findings were limited by several factors, including the reliance on proper clinician reports to the Adverse Event Reporting System database and the separation of some psychiatric terms into categories that may reflect symptoms of other psychiatric diagnoses, the researchers said.
However, “Our data showed high numbers of reports of emotional lability, anxiety disorders, insomnia, self-injurious behavior, and psychotic disorders with isotretinoin as the primary suspect drug,” they noted.
“Although no causal link has been established between isotretinoin and psychiatric adverse events, it is important to recognize that there are data that suggest patients using this drug may be vulnerable to a number of psychiatric conditions” and that monthly iPLEDGE visits are an opportunity to screen patients for these conditions, they said.
They also stressed that “the risk of psychiatric adverse events in patients taking isotretinoin must be considered in the context of a known increased risk of suicidal ideation in patients with acne independent of isotretinoin therapy.”
Mr. Singer had no financial conflicts to disclose. Study coauthor John S. Barbieri, MD, disclosed partial salary support from Pfizer and grand support from the National Institute of Arthritis and Musculoskeletal and Skin Diseases, and Arash Mostaghimi, MD, disclosed personal fees from Pfizer.
SOURCE: Singer S et al. JAMA Dermatol. 2019. Jul 3. doi: 10.1001/jamadermatol.2019.1416.
Isotretinoin use may increase vulnerability to psychiatric conditions, but available evidence does not support a causal relationship, on the basis of data from a retrospective study of 17,829 psychiatric adverse events reported to the Food and Drug Administration over 2 decades.
“Although one study highlighted consistent reporting of depression and suicide in patients taking isotretinoin in the United States from 1982 to 2000, few studies have examined reports of psychiatric adverse events at the national level since 2000,” wrote Sean Singer of Harvard University, Boston, and his colleagues.
In a study published in JAMA Dermatology, the researchers reviewed data from the FDA’s Adverse Event Reporting System between 1997 and 2017.
A total of 17,829 psychiatric adverse events in which isotretinoin was the primary suspect drug were reported during the study period. The researchers classified the events into 12 categories; the most common were depressive disorders (42%), emotional lability (17%), and anxiety (14%). The number of reported psychiatric adverse events was similar between men and women (8,936 and 8,362 events, respectively).
The researchers also identified 2,278 reports of suicidal ideation, 602 reports of attempted suicide, and 368 reports of completed suicide.
In addition, the researchers examined data from the iPLEDGE program and found completed suicide rates of 8.4 per 100,000 patients in 2009 and 5.6 per 100,000 patients in 2010. However, these rates were lower than national suicide rates in the general population of 11.8 per 100,000 people in 2009 and 12.1 per 100,000 people in 2010.
Patient age was available for 13,553 adverse event reports, and patients aged 10-19 years accounted for 53% of the reports overall and 58% of completed suicides for which age was reported.
The high number of psychiatric adverse events in the youngest age group “could reflect more isotretinoin prescriptions in this age group or may suggest that teenagers are particularly vulnerable to psychiatric adverse events while taking isotretinoin,” the researchers said.
The findings were limited by several factors, including the reliance on proper clinician reports to the Adverse Event Reporting System database and the separation of some psychiatric terms into categories that may reflect symptoms of other psychiatric diagnoses, the researchers said.
However, “Our data showed high numbers of reports of emotional lability, anxiety disorders, insomnia, self-injurious behavior, and psychotic disorders with isotretinoin as the primary suspect drug,” they noted.
“Although no causal link has been established between isotretinoin and psychiatric adverse events, it is important to recognize that there are data that suggest patients using this drug may be vulnerable to a number of psychiatric conditions” and that monthly iPLEDGE visits are an opportunity to screen patients for these conditions, they said.
They also stressed that “the risk of psychiatric adverse events in patients taking isotretinoin must be considered in the context of a known increased risk of suicidal ideation in patients with acne independent of isotretinoin therapy.”
Mr. Singer had no financial conflicts to disclose. Study coauthor John S. Barbieri, MD, disclosed partial salary support from Pfizer and grand support from the National Institute of Arthritis and Musculoskeletal and Skin Diseases, and Arash Mostaghimi, MD, disclosed personal fees from Pfizer.
SOURCE: Singer S et al. JAMA Dermatol. 2019. Jul 3. doi: 10.1001/jamadermatol.2019.1416.
Isotretinoin use may increase vulnerability to psychiatric conditions, but available evidence does not support a causal relationship, on the basis of data from a retrospective study of 17,829 psychiatric adverse events reported to the Food and Drug Administration over 2 decades.
“Although one study highlighted consistent reporting of depression and suicide in patients taking isotretinoin in the United States from 1982 to 2000, few studies have examined reports of psychiatric adverse events at the national level since 2000,” wrote Sean Singer of Harvard University, Boston, and his colleagues.
In a study published in JAMA Dermatology, the researchers reviewed data from the FDA’s Adverse Event Reporting System between 1997 and 2017.
A total of 17,829 psychiatric adverse events in which isotretinoin was the primary suspect drug were reported during the study period. The researchers classified the events into 12 categories; the most common were depressive disorders (42%), emotional lability (17%), and anxiety (14%). The number of reported psychiatric adverse events was similar between men and women (8,936 and 8,362 events, respectively).
The researchers also identified 2,278 reports of suicidal ideation, 602 reports of attempted suicide, and 368 reports of completed suicide.
In addition, the researchers examined data from the iPLEDGE program and found completed suicide rates of 8.4 per 100,000 patients in 2009 and 5.6 per 100,000 patients in 2010. However, these rates were lower than national suicide rates in the general population of 11.8 per 100,000 people in 2009 and 12.1 per 100,000 people in 2010.
Patient age was available for 13,553 adverse event reports, and patients aged 10-19 years accounted for 53% of the reports overall and 58% of completed suicides for which age was reported.
The high number of psychiatric adverse events in the youngest age group “could reflect more isotretinoin prescriptions in this age group or may suggest that teenagers are particularly vulnerable to psychiatric adverse events while taking isotretinoin,” the researchers said.
The findings were limited by several factors, including the reliance on proper clinician reports to the Adverse Event Reporting System database and the separation of some psychiatric terms into categories that may reflect symptoms of other psychiatric diagnoses, the researchers said.
However, “Our data showed high numbers of reports of emotional lability, anxiety disorders, insomnia, self-injurious behavior, and psychotic disorders with isotretinoin as the primary suspect drug,” they noted.
“Although no causal link has been established between isotretinoin and psychiatric adverse events, it is important to recognize that there are data that suggest patients using this drug may be vulnerable to a number of psychiatric conditions” and that monthly iPLEDGE visits are an opportunity to screen patients for these conditions, they said.
They also stressed that “the risk of psychiatric adverse events in patients taking isotretinoin must be considered in the context of a known increased risk of suicidal ideation in patients with acne independent of isotretinoin therapy.”
Mr. Singer had no financial conflicts to disclose. Study coauthor John S. Barbieri, MD, disclosed partial salary support from Pfizer and grand support from the National Institute of Arthritis and Musculoskeletal and Skin Diseases, and Arash Mostaghimi, MD, disclosed personal fees from Pfizer.
SOURCE: Singer S et al. JAMA Dermatol. 2019. Jul 3. doi: 10.1001/jamadermatol.2019.1416.
FROM JAMA DERMATOLOGY
Severe Acne Fulminans Following Low-Dose Isotretinoin and Testosterone Use
To the Editor:
Acne fulminans (AF), the most severe form of acne, is a rare condition with an incidence of less than 1% of total acne cases.1 Adolescent boys are the most susceptible group of patients.2 Painful inflammatory pustules that transform into deep ulcerations covered by abundant hemorrhagic crust are typical of AF. Commonly affected areas include the face, back, neck, and chest. Additionally, fever and polyarthralgia may be present, and there often is myopathy due to rapid weight loss.3,4 Less often, erythema nodosum and splenomegaly may be observed.5 Laboratory testing also may reveal markers of systemic inflammation such as leukocytosis with neutrophilia, elevated C-reactive protein levels, increased erythrocyte sedimentation rate, and thrombocytosis. Anemia and elevated hepatic enzyme levels also may be present in AF.2 It is suspected that AF may be induced by low doses of isotretinoin therapy with concomitant inherited susceptibility.6
We report the case of a 21-year-old man who was referred to the Department of Dermatology by his primary care physician for evaluation of severe hemorrhagic lesions on the trunk following use of oral isotretinoin (Figure 1). Prior to development of the lesions, the patient had started weekly intramuscular injections of testosterone 500 mg, which he purchased online without consulting a physician, to address muscle mass reduction associated with sudden weight loss from intense physical training. After 8 months of testosterone supplementation along with continued physical training, the patient presented to his primary care physician for treatment of acne vulgaris on the back and trunk of 2 months’ duration. Oral isotretinoin 20 mg once daily was initiated; however, the patient reported that the acne lesions showed progression after 1 month of treatment. Isotretinoin was increased to a more weight-appropriate dosage of 60 mg once daily 2 weeks before admission to our dermatology clinic.
At the current presentation, dermatologic examination revealed numerous inflamed ulcerations covered by a hemorrhagic crust on the back and trunk. The patient also reported knee, elbow, and inguinal pain, especially at night. No fever or loss of appetite was reported. The patient was otherwise healthy and had no remarkable family history of acne or other dermatologic diseases.
Laboratory testing showed leukocytosis (11,000/µL [reference range, 4500–11,000/µL]), an elevated C-reactive protein level (66 mg/L [reference range, 0.08–3.1 mg/L]), and an elevated erythrocyte sedimentation rate (46 mm/h [reference range, 0–20 mm/h]). There were laboratory and clinical signs of a secondary bacterial infection in the affected areas, and a culture of secretions collected from lesions on the back grew Staphylococcus aureus with sensitivity to erythromycin, clindamycin, doxycycline, and trimethoprim-sulfamethoxazole and resistance to penicillin. A diagnosis of AF was made based on the clinical presentation and systemic symptoms, and anabolic-androgenic steroids and low-dose isotretinoin were identified as etiologic factors.
Treatment initially included cessation of isotretinoin and administration of prednisone, omeprazole, clindamycin, and doxycycline. Prednisone was given at a dosage of 40 mg once daily for 1 week, then decreased by 5 mg every 7 days. Omeprazole was given concurrently as prophylaxis for the gastrointestinal tract side effects of long-term prednisone use. Clindamycin was given at a dosage of 300 mg 3 times daily. Doxycycline was given for 6 weeks at a dosage of 100 mg twice daily. Topical octenidine dihydrochloride also was given.
Marked improvement was noted after 24 hours (Figure 2) as well as on the third day of treatment (Figure 3A). After 6 weeks, only disfiguring scars were visible (Figure 3B). Oral isotretinoin was reincorporated after 8 weeks and was subsequently discontinued after 5 months of therapy with a cumulative dose of 150 mg/kg.

It is important to differentiate AF from exacerbation of acne vulgaris because patients typically have mild or moderate acne vulgaris before the onset of acute symptoms.1 Acne fulminans is characterized by systemic symptoms such as myalgia, polyarthralgia, fatigue, and osteolytic bone lesions.1,7 Additionally, hematologic symptoms such as fever, leukocytosis, anemia, splenomegaly, and hepatomegaly may be present.1,5,7 Our patient demonstrated the polysymptomatic form of AF. The patient had severe acne with a tendency to scar. There also were some systemic manifestations such as polyarthralgia, weight loss, leukocytosis, an elevated erythrocyte sedimentation rate, and an elevated C-reactive protein level.
The clinical diagnosis in our patient also was supported by the hypothesis that heredity, overactive immune reactions, bacterial infections, and use of some drugs (eg, isotretinoin, tetracycline, testosterone) can trigger AF.8 The most well-known theory is that low doses of isotretinoin induce AF.6 The majority of cases are caused by doses of less than 20 mg/kg once daily, but there have been reports of patients using full doses and developing this condition.9 The fact that the use of low- and high-dose isotretinoin can provoke AF suggests an idiosyncratic reaction that is not clearly dose related. The most dangerous triggering factor of AF is concomitant usage of testosterone and isotretinoin.10 Our patient used testosterone injections to increase muscle mass and underwent treatment with isotretinoin for acne.
Treatment of AF is controversial, as there is no standard therapy. Currently, steroids and isotretinoin are the treatments of choice. Antibiotic use is controversial because of a lack of randomized trials.11
In the first stage of treatment, prednisone 0.5 to 1 mg/kg once daily is recommended as an initial anti-inflammatory therapy, with gradual dose reduction. According to evidence-based recommendations, a low dose of isotretinoin can be introduced after crusted lesions have healed. The overlapping therapy with steroids and isotretinoin should be provided for at least 4 weeks. High-potency topical corticosteroids can be used on granulation tissue, which can shorten the systemic treatment with prednisone or can be an alternative treatment for patients with contraindications to systemic corticosteroids.11
Additionally, local care of the lesions including compresses and topical emollients is crucial. There are some case reports in which there is introduction of high doses of isotretinoin, subsequently with systemic steroids.7,8,12 Seukeran and Cunliffe5 proved that it is beneficial to give acne prophylaxis to prevent further episodes. Our patient was similarly treated with systemic steroids and isotretinoin. Treatment guidelines for AF do not recommend oral antibiotics,11 but data are limited in the case of isotretinoin-induced AF. Our patient was given doxycycline concomitant with systemic steroids, which was necessary due to signs of secondary infection from a lesion culture. Doxycycline was stopped when isotretinoin treatment was initiated to prevent pseudotumor cerebri. The patient achieved good clinical improvement with no relapse.
Using isotretinoin to treat acne vulgaris has many benefits, despite the possibility of developing AF as an extremely rare complication. Clinicians should be aware of the risk of this complication to make the diagnosis and provide appropriate care, especially in young men. It is particularly important to consider the possibility of concomitant testosterone and isotretinoin when documenting the patient’s medical history.
- Romiti R, Jansen T, Plewig G. Acne fulminans. An Bras Dermatol. 2000;75:611-617.
- Karvonen SL. Acne fulminans: report of clinical findings and treatment of twenty-four patients. J Am Acad Dermatol. 1993;28:572-579.
- Kelly AP, Burns RE. Acute febrile ulcerative conglobate acne with polyarthralgia. Arch Dermatol. 1971;104:182-187.
- Plewig G, Kligman AM. Vitamin A acid in acneiform dermatoses. Acta Derm Venereol Suppl. 1975;74:119-127.
- Seukeran DC, Cunliffe WJ. The treatment of acne fulminans: a review of 25 cases. Br J Dermatol. 1999;141:307-309.
- Kraus SL, Emmert S, Schön MP, et al. The dark side of beauty: acne fulminans induced by anabolic steroids in a male bodybuilder. Arch Dermatol. 2012;148:1210-1212.
- Jansen T, Plewig G. Acne fulminans. Int J Dermatol. 1998;37:254-257.
- Zanelato TP, Gontijo GM, Alves CA, et al. Disabling acne fulminans. An Bras Dermatol. 2011;86:9-12.
- Azulay DR, Abulafia LA, Costa JAN, et al. Tecido de granulação exuberante. efeito colateral da terapêutica com isotretinoína. An Bras Dermatol. 1985;60:179-182.
- Traupe H, von Mühlendahl KE, Brämswig J, et al. Acne of the fulminans type following testosterone therapy in three excessively tall boys. Arch Dermatol. 1988;124:414-417.
- Greywal T, Zaenglein AL, Baldwin HE, et al. Evidence-based recommendations for the management of acne fulminans and its variants. J Am Acad Dermatol. 2017;77:109-117.
- Honma M, Murakami M, Iinuma S, et al. Acne fulminans following measles infection. J Dermatol. 2009;36:471-473.
To the Editor:
Acne fulminans (AF), the most severe form of acne, is a rare condition with an incidence of less than 1% of total acne cases.1 Adolescent boys are the most susceptible group of patients.2 Painful inflammatory pustules that transform into deep ulcerations covered by abundant hemorrhagic crust are typical of AF. Commonly affected areas include the face, back, neck, and chest. Additionally, fever and polyarthralgia may be present, and there often is myopathy due to rapid weight loss.3,4 Less often, erythema nodosum and splenomegaly may be observed.5 Laboratory testing also may reveal markers of systemic inflammation such as leukocytosis with neutrophilia, elevated C-reactive protein levels, increased erythrocyte sedimentation rate, and thrombocytosis. Anemia and elevated hepatic enzyme levels also may be present in AF.2 It is suspected that AF may be induced by low doses of isotretinoin therapy with concomitant inherited susceptibility.6
We report the case of a 21-year-old man who was referred to the Department of Dermatology by his primary care physician for evaluation of severe hemorrhagic lesions on the trunk following use of oral isotretinoin (Figure 1). Prior to development of the lesions, the patient had started weekly intramuscular injections of testosterone 500 mg, which he purchased online without consulting a physician, to address muscle mass reduction associated with sudden weight loss from intense physical training. After 8 months of testosterone supplementation along with continued physical training, the patient presented to his primary care physician for treatment of acne vulgaris on the back and trunk of 2 months’ duration. Oral isotretinoin 20 mg once daily was initiated; however, the patient reported that the acne lesions showed progression after 1 month of treatment. Isotretinoin was increased to a more weight-appropriate dosage of 60 mg once daily 2 weeks before admission to our dermatology clinic.

At the current presentation, dermatologic examination revealed numerous inflamed ulcerations covered by a hemorrhagic crust on the back and trunk. The patient also reported knee, elbow, and inguinal pain, especially at night. No fever or loss of appetite was reported. The patient was otherwise healthy and had no remarkable family history of acne or other dermatologic diseases.
Laboratory testing showed leukocytosis (11,000/µL [reference range, 4500–11,000/µL]), an elevated C-reactive protein level (66 mg/L [reference range, 0.08–3.1 mg/L]), and an elevated erythrocyte sedimentation rate (46 mm/h [reference range, 0–20 mm/h]). There were laboratory and clinical signs of a secondary bacterial infection in the affected areas, and a culture of secretions collected from lesions on the back grew Staphylococcus aureus with sensitivity to erythromycin, clindamycin, doxycycline, and trimethoprim-sulfamethoxazole and resistance to penicillin. A diagnosis of AF was made based on the clinical presentation and systemic symptoms, and anabolic-androgenic steroids and low-dose isotretinoin were identified as etiologic factors.
Treatment initially included cessation of isotretinoin and administration of prednisone, omeprazole, clindamycin, and doxycycline. Prednisone was given at a dosage of 40 mg once daily for 1 week, then decreased by 5 mg every 7 days. Omeprazole was given concurrently as prophylaxis for the gastrointestinal tract side effects of long-term prednisone use. Clindamycin was given at a dosage of 300 mg 3 times daily. Doxycycline was given for 6 weeks at a dosage of 100 mg twice daily. Topical octenidine dihydrochloride also was given.
Marked improvement was noted after 24 hours (Figure 2) as well as on the third day of treatment (Figure 3A). After 6 weeks, only disfiguring scars were visible (Figure 3B). Oral isotretinoin was reincorporated after 8 weeks and was subsequently discontinued after 5 months of therapy with a cumulative dose of 150 mg/kg.


It is important to differentiate AF from exacerbation of acne vulgaris because patients typically have mild or moderate acne vulgaris before the onset of acute symptoms.1 Acne fulminans is characterized by systemic symptoms such as myalgia, polyarthralgia, fatigue, and osteolytic bone lesions.1,7 Additionally, hematologic symptoms such as fever, leukocytosis, anemia, splenomegaly, and hepatomegaly may be present.1,5,7 Our patient demonstrated the polysymptomatic form of AF. The patient had severe acne with a tendency to scar. There also were some systemic manifestations such as polyarthralgia, weight loss, leukocytosis, an elevated erythrocyte sedimentation rate, and an elevated C-reactive protein level.
The clinical diagnosis in our patient also was supported by the hypothesis that heredity, overactive immune reactions, bacterial infections, and use of some drugs (eg, isotretinoin, tetracycline, testosterone) can trigger AF.8 The most well-known theory is that low doses of isotretinoin induce AF.6 The majority of cases are caused by doses of less than 20 mg/kg once daily, but there have been reports of patients using full doses and developing this condition.9 The fact that the use of low- and high-dose isotretinoin can provoke AF suggests an idiosyncratic reaction that is not clearly dose related. The most dangerous triggering factor of AF is concomitant usage of testosterone and isotretinoin.10 Our patient used testosterone injections to increase muscle mass and underwent treatment with isotretinoin for acne.
Treatment of AF is controversial, as there is no standard therapy. Currently, steroids and isotretinoin are the treatments of choice. Antibiotic use is controversial because of a lack of randomized trials.11
In the first stage of treatment, prednisone 0.5 to 1 mg/kg once daily is recommended as an initial anti-inflammatory therapy, with gradual dose reduction. According to evidence-based recommendations, a low dose of isotretinoin can be introduced after crusted lesions have healed. The overlapping therapy with steroids and isotretinoin should be provided for at least 4 weeks. High-potency topical corticosteroids can be used on granulation tissue, which can shorten the systemic treatment with prednisone or can be an alternative treatment for patients with contraindications to systemic corticosteroids.11
Additionally, local care of the lesions including compresses and topical emollients is crucial. There are some case reports in which there is introduction of high doses of isotretinoin, subsequently with systemic steroids.7,8,12 Seukeran and Cunliffe5 proved that it is beneficial to give acne prophylaxis to prevent further episodes. Our patient was similarly treated with systemic steroids and isotretinoin. Treatment guidelines for AF do not recommend oral antibiotics,11 but data are limited in the case of isotretinoin-induced AF. Our patient was given doxycycline concomitant with systemic steroids, which was necessary due to signs of secondary infection from a lesion culture. Doxycycline was stopped when isotretinoin treatment was initiated to prevent pseudotumor cerebri. The patient achieved good clinical improvement with no relapse.
Using isotretinoin to treat acne vulgaris has many benefits, despite the possibility of developing AF as an extremely rare complication. Clinicians should be aware of the risk of this complication to make the diagnosis and provide appropriate care, especially in young men. It is particularly important to consider the possibility of concomitant testosterone and isotretinoin when documenting the patient’s medical history.
To the Editor:
Acne fulminans (AF), the most severe form of acne, is a rare condition with an incidence of less than 1% of total acne cases.1 Adolescent boys are the most susceptible group of patients.2 Painful inflammatory pustules that transform into deep ulcerations covered by abundant hemorrhagic crust are typical of AF. Commonly affected areas include the face, back, neck, and chest. Additionally, fever and polyarthralgia may be present, and there often is myopathy due to rapid weight loss.3,4 Less often, erythema nodosum and splenomegaly may be observed.5 Laboratory testing also may reveal markers of systemic inflammation such as leukocytosis with neutrophilia, elevated C-reactive protein levels, increased erythrocyte sedimentation rate, and thrombocytosis. Anemia and elevated hepatic enzyme levels also may be present in AF.2 It is suspected that AF may be induced by low doses of isotretinoin therapy with concomitant inherited susceptibility.6
We report the case of a 21-year-old man who was referred to the Department of Dermatology by his primary care physician for evaluation of severe hemorrhagic lesions on the trunk following use of oral isotretinoin (Figure 1). Prior to development of the lesions, the patient had started weekly intramuscular injections of testosterone 500 mg, which he purchased online without consulting a physician, to address muscle mass reduction associated with sudden weight loss from intense physical training. After 8 months of testosterone supplementation along with continued physical training, the patient presented to his primary care physician for treatment of acne vulgaris on the back and trunk of 2 months’ duration. Oral isotretinoin 20 mg once daily was initiated; however, the patient reported that the acne lesions showed progression after 1 month of treatment. Isotretinoin was increased to a more weight-appropriate dosage of 60 mg once daily 2 weeks before admission to our dermatology clinic.

At the current presentation, dermatologic examination revealed numerous inflamed ulcerations covered by a hemorrhagic crust on the back and trunk. The patient also reported knee, elbow, and inguinal pain, especially at night. No fever or loss of appetite was reported. The patient was otherwise healthy and had no remarkable family history of acne or other dermatologic diseases.
Laboratory testing showed leukocytosis (11,000/µL [reference range, 4500–11,000/µL]), an elevated C-reactive protein level (66 mg/L [reference range, 0.08–3.1 mg/L]), and an elevated erythrocyte sedimentation rate (46 mm/h [reference range, 0–20 mm/h]). There were laboratory and clinical signs of a secondary bacterial infection in the affected areas, and a culture of secretions collected from lesions on the back grew Staphylococcus aureus with sensitivity to erythromycin, clindamycin, doxycycline, and trimethoprim-sulfamethoxazole and resistance to penicillin. A diagnosis of AF was made based on the clinical presentation and systemic symptoms, and anabolic-androgenic steroids and low-dose isotretinoin were identified as etiologic factors.
Treatment initially included cessation of isotretinoin and administration of prednisone, omeprazole, clindamycin, and doxycycline. Prednisone was given at a dosage of 40 mg once daily for 1 week, then decreased by 5 mg every 7 days. Omeprazole was given concurrently as prophylaxis for the gastrointestinal tract side effects of long-term prednisone use. Clindamycin was given at a dosage of 300 mg 3 times daily. Doxycycline was given for 6 weeks at a dosage of 100 mg twice daily. Topical octenidine dihydrochloride also was given.
Marked improvement was noted after 24 hours (Figure 2) as well as on the third day of treatment (Figure 3A). After 6 weeks, only disfiguring scars were visible (Figure 3B). Oral isotretinoin was reincorporated after 8 weeks and was subsequently discontinued after 5 months of therapy with a cumulative dose of 150 mg/kg.


It is important to differentiate AF from exacerbation of acne vulgaris because patients typically have mild or moderate acne vulgaris before the onset of acute symptoms.1 Acne fulminans is characterized by systemic symptoms such as myalgia, polyarthralgia, fatigue, and osteolytic bone lesions.1,7 Additionally, hematologic symptoms such as fever, leukocytosis, anemia, splenomegaly, and hepatomegaly may be present.1,5,7 Our patient demonstrated the polysymptomatic form of AF. The patient had severe acne with a tendency to scar. There also were some systemic manifestations such as polyarthralgia, weight loss, leukocytosis, an elevated erythrocyte sedimentation rate, and an elevated C-reactive protein level.
The clinical diagnosis in our patient also was supported by the hypothesis that heredity, overactive immune reactions, bacterial infections, and use of some drugs (eg, isotretinoin, tetracycline, testosterone) can trigger AF.8 The most well-known theory is that low doses of isotretinoin induce AF.6 The majority of cases are caused by doses of less than 20 mg/kg once daily, but there have been reports of patients using full doses and developing this condition.9 The fact that the use of low- and high-dose isotretinoin can provoke AF suggests an idiosyncratic reaction that is not clearly dose related. The most dangerous triggering factor of AF is concomitant usage of testosterone and isotretinoin.10 Our patient used testosterone injections to increase muscle mass and underwent treatment with isotretinoin for acne.
Treatment of AF is controversial, as there is no standard therapy. Currently, steroids and isotretinoin are the treatments of choice. Antibiotic use is controversial because of a lack of randomized trials.11
In the first stage of treatment, prednisone 0.5 to 1 mg/kg once daily is recommended as an initial anti-inflammatory therapy, with gradual dose reduction. According to evidence-based recommendations, a low dose of isotretinoin can be introduced after crusted lesions have healed. The overlapping therapy with steroids and isotretinoin should be provided for at least 4 weeks. High-potency topical corticosteroids can be used on granulation tissue, which can shorten the systemic treatment with prednisone or can be an alternative treatment for patients with contraindications to systemic corticosteroids.11
Additionally, local care of the lesions including compresses and topical emollients is crucial. There are some case reports in which there is introduction of high doses of isotretinoin, subsequently with systemic steroids.7,8,12 Seukeran and Cunliffe5 proved that it is beneficial to give acne prophylaxis to prevent further episodes. Our patient was similarly treated with systemic steroids and isotretinoin. Treatment guidelines for AF do not recommend oral antibiotics,11 but data are limited in the case of isotretinoin-induced AF. Our patient was given doxycycline concomitant with systemic steroids, which was necessary due to signs of secondary infection from a lesion culture. Doxycycline was stopped when isotretinoin treatment was initiated to prevent pseudotumor cerebri. The patient achieved good clinical improvement with no relapse.
Using isotretinoin to treat acne vulgaris has many benefits, despite the possibility of developing AF as an extremely rare complication. Clinicians should be aware of the risk of this complication to make the diagnosis and provide appropriate care, especially in young men. It is particularly important to consider the possibility of concomitant testosterone and isotretinoin when documenting the patient’s medical history.
- Romiti R, Jansen T, Plewig G. Acne fulminans. An Bras Dermatol. 2000;75:611-617.
- Karvonen SL. Acne fulminans: report of clinical findings and treatment of twenty-four patients. J Am Acad Dermatol. 1993;28:572-579.
- Kelly AP, Burns RE. Acute febrile ulcerative conglobate acne with polyarthralgia. Arch Dermatol. 1971;104:182-187.
- Plewig G, Kligman AM. Vitamin A acid in acneiform dermatoses. Acta Derm Venereol Suppl. 1975;74:119-127.
- Seukeran DC, Cunliffe WJ. The treatment of acne fulminans: a review of 25 cases. Br J Dermatol. 1999;141:307-309.
- Kraus SL, Emmert S, Schön MP, et al. The dark side of beauty: acne fulminans induced by anabolic steroids in a male bodybuilder. Arch Dermatol. 2012;148:1210-1212.
- Jansen T, Plewig G. Acne fulminans. Int J Dermatol. 1998;37:254-257.
- Zanelato TP, Gontijo GM, Alves CA, et al. Disabling acne fulminans. An Bras Dermatol. 2011;86:9-12.
- Azulay DR, Abulafia LA, Costa JAN, et al. Tecido de granulação exuberante. efeito colateral da terapêutica com isotretinoína. An Bras Dermatol. 1985;60:179-182.
- Traupe H, von Mühlendahl KE, Brämswig J, et al. Acne of the fulminans type following testosterone therapy in three excessively tall boys. Arch Dermatol. 1988;124:414-417.
- Greywal T, Zaenglein AL, Baldwin HE, et al. Evidence-based recommendations for the management of acne fulminans and its variants. J Am Acad Dermatol. 2017;77:109-117.
- Honma M, Murakami M, Iinuma S, et al. Acne fulminans following measles infection. J Dermatol. 2009;36:471-473.
- Romiti R, Jansen T, Plewig G. Acne fulminans. An Bras Dermatol. 2000;75:611-617.
- Karvonen SL. Acne fulminans: report of clinical findings and treatment of twenty-four patients. J Am Acad Dermatol. 1993;28:572-579.
- Kelly AP, Burns RE. Acute febrile ulcerative conglobate acne with polyarthralgia. Arch Dermatol. 1971;104:182-187.
- Plewig G, Kligman AM. Vitamin A acid in acneiform dermatoses. Acta Derm Venereol Suppl. 1975;74:119-127.
- Seukeran DC, Cunliffe WJ. The treatment of acne fulminans: a review of 25 cases. Br J Dermatol. 1999;141:307-309.
- Kraus SL, Emmert S, Schön MP, et al. The dark side of beauty: acne fulminans induced by anabolic steroids in a male bodybuilder. Arch Dermatol. 2012;148:1210-1212.
- Jansen T, Plewig G. Acne fulminans. Int J Dermatol. 1998;37:254-257.
- Zanelato TP, Gontijo GM, Alves CA, et al. Disabling acne fulminans. An Bras Dermatol. 2011;86:9-12.
- Azulay DR, Abulafia LA, Costa JAN, et al. Tecido de granulação exuberante. efeito colateral da terapêutica com isotretinoína. An Bras Dermatol. 1985;60:179-182.
- Traupe H, von Mühlendahl KE, Brämswig J, et al. Acne of the fulminans type following testosterone therapy in three excessively tall boys. Arch Dermatol. 1988;124:414-417.
- Greywal T, Zaenglein AL, Baldwin HE, et al. Evidence-based recommendations for the management of acne fulminans and its variants. J Am Acad Dermatol. 2017;77:109-117.
- Honma M, Murakami M, Iinuma S, et al. Acne fulminans following measles infection. J Dermatol. 2009;36:471-473.
Practice Points
- Acne fulminans, the most severe form of acne, is characterized by deep ulcerations covered by a hemorrhagic crust. It is commonly associated with fever, polyarthralgia, and myopathy caused by rapid weight loss.
- This rare condition is recognized as a potential complication of oral isotretinoin therapy.
Ovarian reserve markers fall on isotretinoin, but rebound after stopping treatment
MILAN – according to data presented at the World Congress of Dermatology.
Although markers for ovarian reserve, including anti-Müllerian hormone (AMH) serum levels, ovarian volume, and antral follicle count, were significantly lower during a period of isotretinoin use than at baseline, these values were were not significantly different from pretreatment levels by 1 month after stopping isotretinoin.
For patients taking isotretinoin at a dose of 0.5 mg/kg/day, AMH levels fell from a baseline level of 5.29 ng/mL to 4.16 ng/mL during treatment, but rebounded to 4.77 ng/mL 1 month after stopping treatment (P less than .001 for difference between baseline and on-drug values), Tuğba Özkök Akbulut, MD, said during a late-breaking abstracts session.
For women taking isotretinoin 1 mg/kg/day, AMH levels went from 5.14 ng/mL at baseline to 4.24 ng/mL on treatment, to 4.65 ng/mL 1 month after treatment (P less than .001 for difference between baseline and on-drug values), reported Dr. Akbulut a dermatologist at the Haseki Training Research Hospital, Istanbul.
Women on the higher dose of isotretinoin had a similar pattern of decline while on treatment and rebound after ceasing isotretinoin for ovarian volume and antral follicle count (P less than .001 for all values). These differences were not statistically significant for women taking 0.5 mg/kg/day of isotretinoin, except for right ovarian volume (P = 0.013).
Although values were numerically lower for many markers of ovarian reserve after ceasing treatment, compared with baseline figures, these differences were not statistically significantly different. Markers of ovarian reserve did not change significantly for a control group of women without acne.
Dr. Akbulut and her colleagues conducted this prospective case-control study of 42 women of reproductive age who sought dermatologist care for severe acne unresponsive to conservative therapy; 26 women who did not have acne constituted the control group. Smokers, patients with thyroid disease, and those with known polycystic ovary syndrome were excluded from participation.
The women with acne received oral isotretinoin dosed either at 0.5 or 1.0 mg/kg/day, with treatment lasting 5-9 months. For each patient, treatment was stopped when the cumulative dose reached 120 mg/kg.
After an initial visit at which blood was collected from all participants to measure serum AMH levels, those receiving isotretinoin were seen every 4 weeks to check serum lipid and liver enzyme levels.
At the 3-month mark during the study period and 1 month after the end of completing isotretinoin treatment, or at the end of the study period for the control group, blood samples also were drawn for AMH levels.
To measure hormone levels, also blood was drawn between days 2 and 5 of the follicular phase of the menstrual cycle. Participants received ultrasounds to measure antral follicle count and ovarian volume between days 2 and 5 of the menstrual cycle at the initial visit, at the 3-month visit, and at the final visit. Results were interpreted by a trained gynecologist.
Patients, who were mostly in their early 20s, had a mean body mass index of about 22 kg/m2. Hormone levels, ovarian volume, and antral follicle count did not differ among study arms at baseline.
“There are contradictory reports in the literature regarding the effect of retinoic acid on ovarian reserve,” noted Dr. Akbulut. Some preclinical studies found that retinoic acid increased fertility and ovarian reserve in rodents; however, some human studies had shown lower serum AMH concentrations in patients using isotretinoin.
This new demonstration of the reversibility of isotretinoin’s negative effect on ovarian reserve helps clarify a confused picture in the medical literature, said Dr. Akbulut. “The results of our study demonstrated that systemic isotretinoin had a reversible effect on ovarian reserve.”
Dr. Akbulut reported no outside sources of funding and that she had no relevant financial disclosures.
MILAN – according to data presented at the World Congress of Dermatology.
Although markers for ovarian reserve, including anti-Müllerian hormone (AMH) serum levels, ovarian volume, and antral follicle count, were significantly lower during a period of isotretinoin use than at baseline, these values were were not significantly different from pretreatment levels by 1 month after stopping isotretinoin.
For patients taking isotretinoin at a dose of 0.5 mg/kg/day, AMH levels fell from a baseline level of 5.29 ng/mL to 4.16 ng/mL during treatment, but rebounded to 4.77 ng/mL 1 month after stopping treatment (P less than .001 for difference between baseline and on-drug values), Tuğba Özkök Akbulut, MD, said during a late-breaking abstracts session.
For women taking isotretinoin 1 mg/kg/day, AMH levels went from 5.14 ng/mL at baseline to 4.24 ng/mL on treatment, to 4.65 ng/mL 1 month after treatment (P less than .001 for difference between baseline and on-drug values), reported Dr. Akbulut a dermatologist at the Haseki Training Research Hospital, Istanbul.
Women on the higher dose of isotretinoin had a similar pattern of decline while on treatment and rebound after ceasing isotretinoin for ovarian volume and antral follicle count (P less than .001 for all values). These differences were not statistically significant for women taking 0.5 mg/kg/day of isotretinoin, except for right ovarian volume (P = 0.013).
Although values were numerically lower for many markers of ovarian reserve after ceasing treatment, compared with baseline figures, these differences were not statistically significantly different. Markers of ovarian reserve did not change significantly for a control group of women without acne.
Dr. Akbulut and her colleagues conducted this prospective case-control study of 42 women of reproductive age who sought dermatologist care for severe acne unresponsive to conservative therapy; 26 women who did not have acne constituted the control group. Smokers, patients with thyroid disease, and those with known polycystic ovary syndrome were excluded from participation.
The women with acne received oral isotretinoin dosed either at 0.5 or 1.0 mg/kg/day, with treatment lasting 5-9 months. For each patient, treatment was stopped when the cumulative dose reached 120 mg/kg.
After an initial visit at which blood was collected from all participants to measure serum AMH levels, those receiving isotretinoin were seen every 4 weeks to check serum lipid and liver enzyme levels.
At the 3-month mark during the study period and 1 month after the end of completing isotretinoin treatment, or at the end of the study period for the control group, blood samples also were drawn for AMH levels.
To measure hormone levels, also blood was drawn between days 2 and 5 of the follicular phase of the menstrual cycle. Participants received ultrasounds to measure antral follicle count and ovarian volume between days 2 and 5 of the menstrual cycle at the initial visit, at the 3-month visit, and at the final visit. Results were interpreted by a trained gynecologist.
Patients, who were mostly in their early 20s, had a mean body mass index of about 22 kg/m2. Hormone levels, ovarian volume, and antral follicle count did not differ among study arms at baseline.
“There are contradictory reports in the literature regarding the effect of retinoic acid on ovarian reserve,” noted Dr. Akbulut. Some preclinical studies found that retinoic acid increased fertility and ovarian reserve in rodents; however, some human studies had shown lower serum AMH concentrations in patients using isotretinoin.
This new demonstration of the reversibility of isotretinoin’s negative effect on ovarian reserve helps clarify a confused picture in the medical literature, said Dr. Akbulut. “The results of our study demonstrated that systemic isotretinoin had a reversible effect on ovarian reserve.”
Dr. Akbulut reported no outside sources of funding and that she had no relevant financial disclosures.
MILAN – according to data presented at the World Congress of Dermatology.
Although markers for ovarian reserve, including anti-Müllerian hormone (AMH) serum levels, ovarian volume, and antral follicle count, were significantly lower during a period of isotretinoin use than at baseline, these values were were not significantly different from pretreatment levels by 1 month after stopping isotretinoin.
For patients taking isotretinoin at a dose of 0.5 mg/kg/day, AMH levels fell from a baseline level of 5.29 ng/mL to 4.16 ng/mL during treatment, but rebounded to 4.77 ng/mL 1 month after stopping treatment (P less than .001 for difference between baseline and on-drug values), Tuğba Özkök Akbulut, MD, said during a late-breaking abstracts session.
For women taking isotretinoin 1 mg/kg/day, AMH levels went from 5.14 ng/mL at baseline to 4.24 ng/mL on treatment, to 4.65 ng/mL 1 month after treatment (P less than .001 for difference between baseline and on-drug values), reported Dr. Akbulut a dermatologist at the Haseki Training Research Hospital, Istanbul.
Women on the higher dose of isotretinoin had a similar pattern of decline while on treatment and rebound after ceasing isotretinoin for ovarian volume and antral follicle count (P less than .001 for all values). These differences were not statistically significant for women taking 0.5 mg/kg/day of isotretinoin, except for right ovarian volume (P = 0.013).
Although values were numerically lower for many markers of ovarian reserve after ceasing treatment, compared with baseline figures, these differences were not statistically significantly different. Markers of ovarian reserve did not change significantly for a control group of women without acne.
Dr. Akbulut and her colleagues conducted this prospective case-control study of 42 women of reproductive age who sought dermatologist care for severe acne unresponsive to conservative therapy; 26 women who did not have acne constituted the control group. Smokers, patients with thyroid disease, and those with known polycystic ovary syndrome were excluded from participation.
The women with acne received oral isotretinoin dosed either at 0.5 or 1.0 mg/kg/day, with treatment lasting 5-9 months. For each patient, treatment was stopped when the cumulative dose reached 120 mg/kg.
After an initial visit at which blood was collected from all participants to measure serum AMH levels, those receiving isotretinoin were seen every 4 weeks to check serum lipid and liver enzyme levels.
At the 3-month mark during the study period and 1 month after the end of completing isotretinoin treatment, or at the end of the study period for the control group, blood samples also were drawn for AMH levels.
To measure hormone levels, also blood was drawn between days 2 and 5 of the follicular phase of the menstrual cycle. Participants received ultrasounds to measure antral follicle count and ovarian volume between days 2 and 5 of the menstrual cycle at the initial visit, at the 3-month visit, and at the final visit. Results were interpreted by a trained gynecologist.
Patients, who were mostly in their early 20s, had a mean body mass index of about 22 kg/m2. Hormone levels, ovarian volume, and antral follicle count did not differ among study arms at baseline.
“There are contradictory reports in the literature regarding the effect of retinoic acid on ovarian reserve,” noted Dr. Akbulut. Some preclinical studies found that retinoic acid increased fertility and ovarian reserve in rodents; however, some human studies had shown lower serum AMH concentrations in patients using isotretinoin.
This new demonstration of the reversibility of isotretinoin’s negative effect on ovarian reserve helps clarify a confused picture in the medical literature, said Dr. Akbulut. “The results of our study demonstrated that systemic isotretinoin had a reversible effect on ovarian reserve.”
Dr. Akbulut reported no outside sources of funding and that she had no relevant financial disclosures.
REPORTING FROM WCD2019
Treatment Consideration for US Military Members With Skin Disease
The National Defense Authorization Act for Fiscal Year 20171 has changed military medicine, including substantial reduction in military medical personnel as positions are converted to combat functions. As a result, there will be fewer military dermatologists, which means many US soldiers, sailors, airmen, and marines will seek medical care outside of military treatment facilities. This article highlights some unique treatment considerations in this patient population for our civilian dermatology colleagues.
Medical Readiness
In 2015, General Joseph F. Dunford Jr, 19th Chairman of the Joint Chiefs of Staff, made readiness his top priority for the US Armed Forces.2 Readiness refers to service members’ ability to deploy to locations across the globe and perform their military duties with little advanced notice, which requires personnel to be medically prepared at all times to leave home and perform their duties in locations with limited medical support.
Medical readiness is maintaining a unit that is medically able to perform its military function both at home and in a deployed environment. Military members’ medical readiness status is carefully tracked and determined via annual physical, dental, hearing, and vision examinations, as well as human immunodeficiency virus status and immunizations. The readiness status of the unit (ie, the number of troops ready to deploy at any given time) is available to commanders at all levels at any time. Each military branch has tracking systems that allow commanders to know when a member is past due for an examination or if a member’s medical status has changed, making them nondeployable. When a member is nondeployable, it affects the unit’s ability to perform its mission and degrades its readiness. If readiness is suboptimal, the military cannot deploy and complete its missions, which is why readiness is a top priority. The primary function of military medicine is to support the medical readiness of the force.
Deployment Eligibility
A unique aspect of military medicine that can be foreign to civilian physicians is the unit commanders’ authority to request and receive information on military members’ medical conditions as they relate to readiness. Under most circumstances, an individual’s medical information is his/her private information; however, that is not always the case in the military. If a member’s medical status changes and he/she becomes nondeployable, by regulation the commander can be privy to pertinent aspects of that member’s medical condition as it affects unit readiness, including the diagnosis, treatment plan, and prognosis. Commanders need this information to aid in the member’s recovery, ensure training does not impact his/her care, and identify possible need of replacement.
Published accession guidelines are used to determine medical eligibility for service.3 These instructions are organized by major organ systems and broad disease categories. They provide guidance on medically disqualifying conditions. The Table outlines those conditions that apply to the skin.3 Individual military branches may have additional regulations with guidance on medically disqualifying conditions that are job specific. Additional regulations also are available based on an area of military operation that can be more restrictive and specific to those locations.4
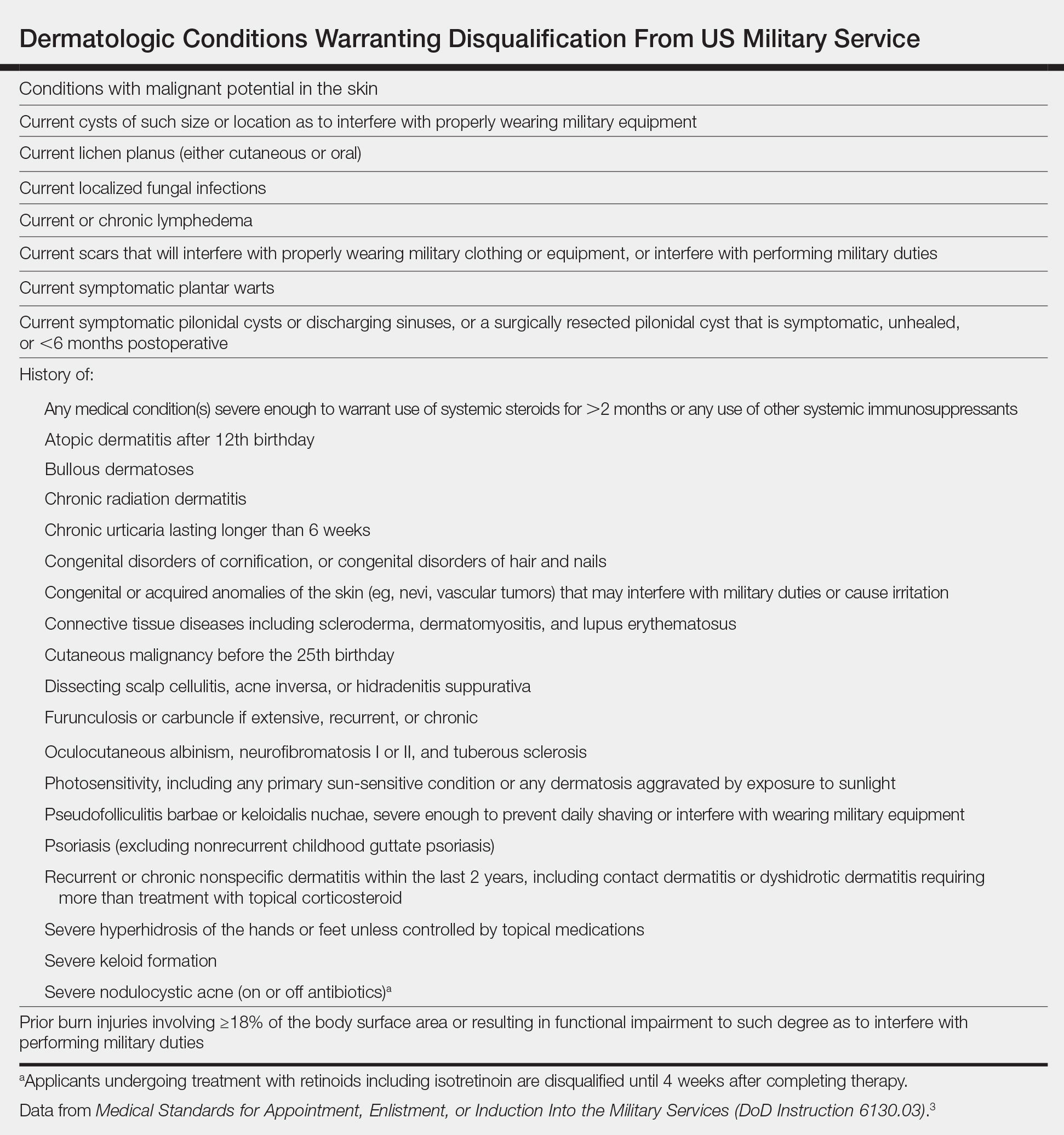
Similarly, each military branch has its own retention standards.5,6 Previously healthy individuals can develop new medical conditions, and commanders are notified if a service member becomes medically nondeployable. If a medical condition limits a service member’s ability to deploy, he/she will be evaluated for retention by a medical evaluation board (MEB). Three outcomes are possible: return in current function, retain the service member but retrain in another military occupation, or separate from military service.7 Rarely, waivers are provided so that the service member can return to duty.
Readiness and Patient Care
Importantly, readiness should not be seen as a roadblock to appropriate patient care. Patients should receive treatment that is appropriate for their medical condition. Much of the difficulty within military medicine is understanding and communicating how the natural disease history, prognosis, and treatment of their respective medical conditions will impact members’ service.
In some cases, the condition and/or treatment is incompatible with military service. Consider the following scenario: A 23-year-old active-duty soldier with a history of psoriasis developed widespread disease of 1 year’s duration and was referred to a civilian dermatologist due to nonavailability of a military dermatologist. After topical and light-based therapies failed, he was started on ustekinumab, which cleared the psoriasis. He wanted to continue on ustekinumab due to its good efficacy, but his unit was set to deploy in the coming year, and the drug made him medically nondeployable due to its immunosuppressive nature.
This real-life example was a difficult case to disposition. The service member was unsure if he could perform his military duties and deploy without continuing treatment with ustekinumab. His prior dermatology notes were requested to better assess the severity of his baseline disease, followed by a candid discussion between the military dermatologist and the patient about treatment options and their respective ramifications to his military career. One option included continuing ustekinumab, which would initiate an MEB evaluation and likely result in separation. Another option was UV therapy, which would not prompt an MEB evaluation but would not be available in deployed environments. Apremilast was offered as a third treatment option and could be used in place of UV therapy during deployment along with topical medications. This patient opted to continue treatment with ustekinumab, resulting in MEB review and separation from military service.
Dermatology Treatment Considerations
Civilian dermatologists should be aware of specific considerations when treating active US service members with common cutaneous diagnoses such as acne, atopic dermatitis (AD), psoriasis, dissecting cellulitis of the scalp (DCS), and lupus erythematosus (LE). This discussion is not meant to be all-inclusive but provides information and examples related to common treatment challenges in this patient population.
Acne
Acne is common in the active-duty military population. Typically, acne should be treated per recommended guidelines based on type and severity.8 Medical evaluation board review is warranted in cases of severe acne that is unresponsive to treatment and interferes with a service member’s performance.5,6 Unique situations in the active-duty military population include the following:
• Use of minocycline. Aircrew members have unique restrictions on many medications,6 including minocycline, which is restricted in this population due to vestibular side effects. Doxycycline is an acceptable alternative for aircrew members; however, even this medication may require a ground trial to ensure there are no idiosyncratic effects.
• Use of isotretinoin, which is not permitted in aircrew members, submariners, or divers. If they take this medication, they will be temporarily removed from duty for the duration of treatment and for a period of time after completion (1–3 months, depending on service). Isotretinoin also is not used during deployment due to potential side effects, the need for laboratory monitoring, and iPLEDGE system requirements.
Atopic Dermatitis
A history of AD after the 12th birthday is considered a disqualifying condition with regard to military service,3 though mild and well-controlled disease can easily be overlooked during entrance physical examinations. Members frequently present with eczema flares following field training exercises where they are outdoors for many hours and have been exposed to grass or other environmental triggers while wearing military gear that is heavy and occlusive, which is further exacerbated by being unable to bathe or care for their skin as they would at home.
Separation from the military is considered when AD is moderate to severe, is unresponsive to treatment, and/or interferes with performance of duty. Severity often can be evaluated based on the impact of AD on performance of duties in addition to clinical appearance. A pilot who is distracted by itching presents a potentially dangerous situation. A soldier whose AD flares every time he/she goes to the field, requiring him/her to return home early to control symptoms, can be considered moderate to severe due to lack of ability to do his/her job away from home base.
Response to treatment is more often where trouble lies for military members with AD, as patients are only permitted to take emollients, preferred cleansers, and topical medications to field training exercises and deployments. UV therapy is used to control disease in the military population but is not an option in deployed environments. Classic immunosuppressants (eg, methotrexate, mycophenolate mofetil, azathioprine, cyclosporine) may result in a good response to treatment; however, due to their side-effect profiles, need for laboratory monitoring, and immunosuppressive nature, long-term use of those medications will result in a nondeployable status. Dupilumab does not appear to have the immunosuppressive effects of other biologics; however, the medication requires refrigeration,9 which currently precludes its use in the deployed environment, as it would be difficult to ensure supply and storage in remote areas.
Service members with a history of AD are exempt from the smallpox vaccine due to concerns about eczema vaccinatum.10
Psoriasis
Psoriasis is another dermatologic condition that does not meet military admission standards,3 and mild undiagnosed cases may be overlooked during the entrance physical examination. Because psoriasis commonly affects young adults, it may manifest in service members after entering service. If psoriasis is extensive or refractory to treatment, an MEB evaluation may be required.5,6 Widespread psoriasis can result in considerable discomfort when wearing body armor and other military gear. Severe localized disease can have duty implications; service members with treatment-resistant scalp psoriasis or pustular psoriasis of the feet may have difficulty wearing helmets or military boots, respectively.
Most service members with limited psoriasis vulgaris can be managed with topical steroids and steroid-sparing agents such as calcipotriene. Some service members opt not to aggressively treat their psoriasis if it is limited in nature and not symptomatic.
When discussing systemic treatments beyond light therapy in those with refractory disease, apremilast can be a good first-line treatment option.11 It is an oral medication, has minimal monitoring requirements, and lacks immunosuppressive side effects; therefore, it does not adversely impact deployability. If patients do not improve in 4 months with apremilast, biologics should then be considered; however, biologics have service implications, the most important being inability to deploy while taking the medication. In rare circumstances, military dermatologists may discuss utilizing biologic therapy only in the nondeployed setting. In these cases, service members are counseled that biologic therapy will be discontinued if they deploy in the future and treatment will be sustained with topicals and/or apremilast through the deployment. The treatment plan also should be communicated to the patient’s primary care provider to ensure that he/she is in agreement.
Dissecting Cellulitis of the Scalp
Dissecting cellulitis of the scalp may result in separation if the condition is unresponsive to treatment and/or interferes with satisfactory performance of duty.5 In addition to causing considerable pain, this condition can prevent service members from wearing combat helmets, which limits their ability to train and deploy. One of the authors (S.C.) has had more service members undergo an MEB evaluation for DCS than any of the other conditions mentioned.
Topical tretinoin and topical antibiotics can be used in conjunction with either doxycycline or minocycline to treat DCS, with the addition of intralesional corticosteroids for painful nodules. Fluctuant lesions are treated with incision and drainage. If there is inadequate response to treatment after 2 to 3 months, oral clindamycin and rifampin can be tried for 3 months. As an alternative measure or if the condition is refractory to oral clindamycin and rifampin, isotretinoin can then be used. One of the authors (S.C.) typically recommends a temporary no-helmet profile to the patient’s primary care provider until his/her next dermatology appointment. If the patient still has substantial disease despite these treatment options, it is recommended that the patient be issued a permanent profile for no helmet wear, which will prompt an MEB evaluation. Although tumor necrosis factor α inhibitors can work well in patients with DCS, the use of biologics is not conducive to continued service.
Lupus Erythematosus
A history of LE is disqualifying from military service. Patients who develop LE while on active duty will be referred for MEB evaluation if their disease is unresponsive to treatment and/or interferes with the satisfactory performance of duty.5,6 In general, connective tissue diseases have an array of physical implications that can affect military service, including photosensitivity, joint inflammation, and internal organ involvement. Similar to the other dermatologic conditions described, treatment of connective tissue diseases also can present challenges to continued military service. Considerations in the case of LE that are unique to military service members include the following:
• Sun exposure. Most military service members are required to work outside in all manners of conditions, which include hot, sunny, humid, and/or dry climates. Often physicians might counsel sun-sensitive patients with LE to avoid being outside during daylight hours, limit window exposure at work, and avoid daytime driving when possible; however, these recommendations are not possible for many, if not most, service members.
• Immunosuppressive therapies are incompatible with military deployment; therefore, prescribing methotrexate, cyclosporine, mycophenolate mofetil, rituximab, or belimumab for treatment of LE would prompt an MEB evaluation if the treatment is necessary to control the disease.
Final Thoughts
The recent changes to military medicine are needed to meet our country’s defense requirements and will ultimately result in civilian specialists playing a larger role in the care of our military population. This article highlights unique factors civilian dermatologists must consider when treating active-duty military patients to ensure they remain deployable during treatment.
- National Defense Authorization Act for Fiscal Year 2017, S 2943, 114th Congress, 2nd Sess (2016).
- Garamone J. Dunford sends message to joint force, stresses readiness, warfighting, education [news release]. Washington, DC: US Department of Defense; October 2, 2015. https://dod.defense.gov/News/Article/Article/621725/dunford-sends-message-to-joint-force-stresses-readiness-warfighting-education/. Accessed May 17, 2019.
- Medical Standards for Appointment, Enlistment, or Induction Into the Military Services (DoD Instruction 6130.03). Washington, DC: Department of Defense; March 30, 2018. https://www.esd.whs.mil/Portals/54/Documents/DD/issuances/dodi/613003p.pdf?ver=2018-05-04-113917-883. Accessed May 17, 2019.
- Force health protection guidance for deployment in USSOUTHCOM as of 7 December 2017. US Southern Command website. https://www.southcom.mil/Portals/7/Documents/Operational%20Contract%20Support/USSOUTHCOM_Force_Health_Protection_Guidance_AS_OF_7_DEC_2017.pdf?ver=2018-01-29-100603-957. Published December 7, 2017. Accessed May 28, 2019.
- US Department of the Army. Standards of medical fitness. http://www.au.af.mil/au/awc/awcgate/army/r40_501.pdf. Published August 26, 2003. Accessed May 17, 2019.
- US Department of the Air Force. Medical examinations and standards. https://static.e-publishing.af.mil/production/1/af_sg/publication/afi48-123/afi48-123.pdf. Published November 5, 2013. Accessed May 17, 2019.
- Medical and physical evaluation boards (MEB/PEB). US Army Warrior Care and Transition website. https://wct.army.mil/modules/soldier/s6-medicalBoards.html. Accessed May 28, 2019.
- Zaenglein AL, Pathy AL, Schlosser BJ, et al. Guidelines of care for the management of acne vulgaris. J Am Acad Dermatol. 2016;74:945-973.
- Dupixent [package insert]. Tarrytown, NY: Regeneron, Inc; 2017.
- Departments of the Army, the Navy, the Air Force, and the Coast Guard. Immunizations and chemoprophylaxis for the prevention of infectious diseases. https://health.mil/Reference-Center/Policies/2013/10/07/Immunizations-and-Chemoprophylaxis-for-the-Prevention-of-Infectious-Diseases. Published October 7, 2013. Accessed May 28, 2019.
- Rosenberg A, Meyerle J. The use of apremilast to treat psoriasis during deployment. Mil Med. 2017;182:1628-1631.
The National Defense Authorization Act for Fiscal Year 20171 has changed military medicine, including substantial reduction in military medical personnel as positions are converted to combat functions. As a result, there will be fewer military dermatologists, which means many US soldiers, sailors, airmen, and marines will seek medical care outside of military treatment facilities. This article highlights some unique treatment considerations in this patient population for our civilian dermatology colleagues.
Medical Readiness
In 2015, General Joseph F. Dunford Jr, 19th Chairman of the Joint Chiefs of Staff, made readiness his top priority for the US Armed Forces.2 Readiness refers to service members’ ability to deploy to locations across the globe and perform their military duties with little advanced notice, which requires personnel to be medically prepared at all times to leave home and perform their duties in locations with limited medical support.
Medical readiness is maintaining a unit that is medically able to perform its military function both at home and in a deployed environment. Military members’ medical readiness status is carefully tracked and determined via annual physical, dental, hearing, and vision examinations, as well as human immunodeficiency virus status and immunizations. The readiness status of the unit (ie, the number of troops ready to deploy at any given time) is available to commanders at all levels at any time. Each military branch has tracking systems that allow commanders to know when a member is past due for an examination or if a member’s medical status has changed, making them nondeployable. When a member is nondeployable, it affects the unit’s ability to perform its mission and degrades its readiness. If readiness is suboptimal, the military cannot deploy and complete its missions, which is why readiness is a top priority. The primary function of military medicine is to support the medical readiness of the force.
Deployment Eligibility
A unique aspect of military medicine that can be foreign to civilian physicians is the unit commanders’ authority to request and receive information on military members’ medical conditions as they relate to readiness. Under most circumstances, an individual’s medical information is his/her private information; however, that is not always the case in the military. If a member’s medical status changes and he/she becomes nondeployable, by regulation the commander can be privy to pertinent aspects of that member’s medical condition as it affects unit readiness, including the diagnosis, treatment plan, and prognosis. Commanders need this information to aid in the member’s recovery, ensure training does not impact his/her care, and identify possible need of replacement.
Published accession guidelines are used to determine medical eligibility for service.3 These instructions are organized by major organ systems and broad disease categories. They provide guidance on medically disqualifying conditions. The Table outlines those conditions that apply to the skin.3 Individual military branches may have additional regulations with guidance on medically disqualifying conditions that are job specific. Additional regulations also are available based on an area of military operation that can be more restrictive and specific to those locations.4

Similarly, each military branch has its own retention standards.5,6 Previously healthy individuals can develop new medical conditions, and commanders are notified if a service member becomes medically nondeployable. If a medical condition limits a service member’s ability to deploy, he/she will be evaluated for retention by a medical evaluation board (MEB). Three outcomes are possible: return in current function, retain the service member but retrain in another military occupation, or separate from military service.7 Rarely, waivers are provided so that the service member can return to duty.
Readiness and Patient Care
Importantly, readiness should not be seen as a roadblock to appropriate patient care. Patients should receive treatment that is appropriate for their medical condition. Much of the difficulty within military medicine is understanding and communicating how the natural disease history, prognosis, and treatment of their respective medical conditions will impact members’ service.
In some cases, the condition and/or treatment is incompatible with military service. Consider the following scenario: A 23-year-old active-duty soldier with a history of psoriasis developed widespread disease of 1 year’s duration and was referred to a civilian dermatologist due to nonavailability of a military dermatologist. After topical and light-based therapies failed, he was started on ustekinumab, which cleared the psoriasis. He wanted to continue on ustekinumab due to its good efficacy, but his unit was set to deploy in the coming year, and the drug made him medically nondeployable due to its immunosuppressive nature.
This real-life example was a difficult case to disposition. The service member was unsure if he could perform his military duties and deploy without continuing treatment with ustekinumab. His prior dermatology notes were requested to better assess the severity of his baseline disease, followed by a candid discussion between the military dermatologist and the patient about treatment options and their respective ramifications to his military career. One option included continuing ustekinumab, which would initiate an MEB evaluation and likely result in separation. Another option was UV therapy, which would not prompt an MEB evaluation but would not be available in deployed environments. Apremilast was offered as a third treatment option and could be used in place of UV therapy during deployment along with topical medications. This patient opted to continue treatment with ustekinumab, resulting in MEB review and separation from military service.
Dermatology Treatment Considerations
Civilian dermatologists should be aware of specific considerations when treating active US service members with common cutaneous diagnoses such as acne, atopic dermatitis (AD), psoriasis, dissecting cellulitis of the scalp (DCS), and lupus erythematosus (LE). This discussion is not meant to be all-inclusive but provides information and examples related to common treatment challenges in this patient population.
Acne
Acne is common in the active-duty military population. Typically, acne should be treated per recommended guidelines based on type and severity.8 Medical evaluation board review is warranted in cases of severe acne that is unresponsive to treatment and interferes with a service member’s performance.5,6 Unique situations in the active-duty military population include the following:
• Use of minocycline. Aircrew members have unique restrictions on many medications,6 including minocycline, which is restricted in this population due to vestibular side effects. Doxycycline is an acceptable alternative for aircrew members; however, even this medication may require a ground trial to ensure there are no idiosyncratic effects.
• Use of isotretinoin, which is not permitted in aircrew members, submariners, or divers. If they take this medication, they will be temporarily removed from duty for the duration of treatment and for a period of time after completion (1–3 months, depending on service). Isotretinoin also is not used during deployment due to potential side effects, the need for laboratory monitoring, and iPLEDGE system requirements.
Atopic Dermatitis
A history of AD after the 12th birthday is considered a disqualifying condition with regard to military service,3 though mild and well-controlled disease can easily be overlooked during entrance physical examinations. Members frequently present with eczema flares following field training exercises where they are outdoors for many hours and have been exposed to grass or other environmental triggers while wearing military gear that is heavy and occlusive, which is further exacerbated by being unable to bathe or care for their skin as they would at home.
Separation from the military is considered when AD is moderate to severe, is unresponsive to treatment, and/or interferes with performance of duty. Severity often can be evaluated based on the impact of AD on performance of duties in addition to clinical appearance. A pilot who is distracted by itching presents a potentially dangerous situation. A soldier whose AD flares every time he/she goes to the field, requiring him/her to return home early to control symptoms, can be considered moderate to severe due to lack of ability to do his/her job away from home base.
Response to treatment is more often where trouble lies for military members with AD, as patients are only permitted to take emollients, preferred cleansers, and topical medications to field training exercises and deployments. UV therapy is used to control disease in the military population but is not an option in deployed environments. Classic immunosuppressants (eg, methotrexate, mycophenolate mofetil, azathioprine, cyclosporine) may result in a good response to treatment; however, due to their side-effect profiles, need for laboratory monitoring, and immunosuppressive nature, long-term use of those medications will result in a nondeployable status. Dupilumab does not appear to have the immunosuppressive effects of other biologics; however, the medication requires refrigeration,9 which currently precludes its use in the deployed environment, as it would be difficult to ensure supply and storage in remote areas.
Service members with a history of AD are exempt from the smallpox vaccine due to concerns about eczema vaccinatum.10
Psoriasis
Psoriasis is another dermatologic condition that does not meet military admission standards,3 and mild undiagnosed cases may be overlooked during the entrance physical examination. Because psoriasis commonly affects young adults, it may manifest in service members after entering service. If psoriasis is extensive or refractory to treatment, an MEB evaluation may be required.5,6 Widespread psoriasis can result in considerable discomfort when wearing body armor and other military gear. Severe localized disease can have duty implications; service members with treatment-resistant scalp psoriasis or pustular psoriasis of the feet may have difficulty wearing helmets or military boots, respectively.
Most service members with limited psoriasis vulgaris can be managed with topical steroids and steroid-sparing agents such as calcipotriene. Some service members opt not to aggressively treat their psoriasis if it is limited in nature and not symptomatic.
When discussing systemic treatments beyond light therapy in those with refractory disease, apremilast can be a good first-line treatment option.11 It is an oral medication, has minimal monitoring requirements, and lacks immunosuppressive side effects; therefore, it does not adversely impact deployability. If patients do not improve in 4 months with apremilast, biologics should then be considered; however, biologics have service implications, the most important being inability to deploy while taking the medication. In rare circumstances, military dermatologists may discuss utilizing biologic therapy only in the nondeployed setting. In these cases, service members are counseled that biologic therapy will be discontinued if they deploy in the future and treatment will be sustained with topicals and/or apremilast through the deployment. The treatment plan also should be communicated to the patient’s primary care provider to ensure that he/she is in agreement.
Dissecting Cellulitis of the Scalp
Dissecting cellulitis of the scalp may result in separation if the condition is unresponsive to treatment and/or interferes with satisfactory performance of duty.5 In addition to causing considerable pain, this condition can prevent service members from wearing combat helmets, which limits their ability to train and deploy. One of the authors (S.C.) has had more service members undergo an MEB evaluation for DCS than any of the other conditions mentioned.
Topical tretinoin and topical antibiotics can be used in conjunction with either doxycycline or minocycline to treat DCS, with the addition of intralesional corticosteroids for painful nodules. Fluctuant lesions are treated with incision and drainage. If there is inadequate response to treatment after 2 to 3 months, oral clindamycin and rifampin can be tried for 3 months. As an alternative measure or if the condition is refractory to oral clindamycin and rifampin, isotretinoin can then be used. One of the authors (S.C.) typically recommends a temporary no-helmet profile to the patient’s primary care provider until his/her next dermatology appointment. If the patient still has substantial disease despite these treatment options, it is recommended that the patient be issued a permanent profile for no helmet wear, which will prompt an MEB evaluation. Although tumor necrosis factor α inhibitors can work well in patients with DCS, the use of biologics is not conducive to continued service.
Lupus Erythematosus
A history of LE is disqualifying from military service. Patients who develop LE while on active duty will be referred for MEB evaluation if their disease is unresponsive to treatment and/or interferes with the satisfactory performance of duty.5,6 In general, connective tissue diseases have an array of physical implications that can affect military service, including photosensitivity, joint inflammation, and internal organ involvement. Similar to the other dermatologic conditions described, treatment of connective tissue diseases also can present challenges to continued military service. Considerations in the case of LE that are unique to military service members include the following:
• Sun exposure. Most military service members are required to work outside in all manners of conditions, which include hot, sunny, humid, and/or dry climates. Often physicians might counsel sun-sensitive patients with LE to avoid being outside during daylight hours, limit window exposure at work, and avoid daytime driving when possible; however, these recommendations are not possible for many, if not most, service members.
• Immunosuppressive therapies are incompatible with military deployment; therefore, prescribing methotrexate, cyclosporine, mycophenolate mofetil, rituximab, or belimumab for treatment of LE would prompt an MEB evaluation if the treatment is necessary to control the disease.
Final Thoughts
The recent changes to military medicine are needed to meet our country’s defense requirements and will ultimately result in civilian specialists playing a larger role in the care of our military population. This article highlights unique factors civilian dermatologists must consider when treating active-duty military patients to ensure they remain deployable during treatment.
The National Defense Authorization Act for Fiscal Year 20171 has changed military medicine, including substantial reduction in military medical personnel as positions are converted to combat functions. As a result, there will be fewer military dermatologists, which means many US soldiers, sailors, airmen, and marines will seek medical care outside of military treatment facilities. This article highlights some unique treatment considerations in this patient population for our civilian dermatology colleagues.
Medical Readiness
In 2015, General Joseph F. Dunford Jr, 19th Chairman of the Joint Chiefs of Staff, made readiness his top priority for the US Armed Forces.2 Readiness refers to service members’ ability to deploy to locations across the globe and perform their military duties with little advanced notice, which requires personnel to be medically prepared at all times to leave home and perform their duties in locations with limited medical support.
Medical readiness is maintaining a unit that is medically able to perform its military function both at home and in a deployed environment. Military members’ medical readiness status is carefully tracked and determined via annual physical, dental, hearing, and vision examinations, as well as human immunodeficiency virus status and immunizations. The readiness status of the unit (ie, the number of troops ready to deploy at any given time) is available to commanders at all levels at any time. Each military branch has tracking systems that allow commanders to know when a member is past due for an examination or if a member’s medical status has changed, making them nondeployable. When a member is nondeployable, it affects the unit’s ability to perform its mission and degrades its readiness. If readiness is suboptimal, the military cannot deploy and complete its missions, which is why readiness is a top priority. The primary function of military medicine is to support the medical readiness of the force.
Deployment Eligibility
A unique aspect of military medicine that can be foreign to civilian physicians is the unit commanders’ authority to request and receive information on military members’ medical conditions as they relate to readiness. Under most circumstances, an individual’s medical information is his/her private information; however, that is not always the case in the military. If a member’s medical status changes and he/she becomes nondeployable, by regulation the commander can be privy to pertinent aspects of that member’s medical condition as it affects unit readiness, including the diagnosis, treatment plan, and prognosis. Commanders need this information to aid in the member’s recovery, ensure training does not impact his/her care, and identify possible need of replacement.
Published accession guidelines are used to determine medical eligibility for service.3 These instructions are organized by major organ systems and broad disease categories. They provide guidance on medically disqualifying conditions. The Table outlines those conditions that apply to the skin.3 Individual military branches may have additional regulations with guidance on medically disqualifying conditions that are job specific. Additional regulations also are available based on an area of military operation that can be more restrictive and specific to those locations.4

Similarly, each military branch has its own retention standards.5,6 Previously healthy individuals can develop new medical conditions, and commanders are notified if a service member becomes medically nondeployable. If a medical condition limits a service member’s ability to deploy, he/she will be evaluated for retention by a medical evaluation board (MEB). Three outcomes are possible: return in current function, retain the service member but retrain in another military occupation, or separate from military service.7 Rarely, waivers are provided so that the service member can return to duty.
Readiness and Patient Care
Importantly, readiness should not be seen as a roadblock to appropriate patient care. Patients should receive treatment that is appropriate for their medical condition. Much of the difficulty within military medicine is understanding and communicating how the natural disease history, prognosis, and treatment of their respective medical conditions will impact members’ service.
In some cases, the condition and/or treatment is incompatible with military service. Consider the following scenario: A 23-year-old active-duty soldier with a history of psoriasis developed widespread disease of 1 year’s duration and was referred to a civilian dermatologist due to nonavailability of a military dermatologist. After topical and light-based therapies failed, he was started on ustekinumab, which cleared the psoriasis. He wanted to continue on ustekinumab due to its good efficacy, but his unit was set to deploy in the coming year, and the drug made him medically nondeployable due to its immunosuppressive nature.
This real-life example was a difficult case to disposition. The service member was unsure if he could perform his military duties and deploy without continuing treatment with ustekinumab. His prior dermatology notes were requested to better assess the severity of his baseline disease, followed by a candid discussion between the military dermatologist and the patient about treatment options and their respective ramifications to his military career. One option included continuing ustekinumab, which would initiate an MEB evaluation and likely result in separation. Another option was UV therapy, which would not prompt an MEB evaluation but would not be available in deployed environments. Apremilast was offered as a third treatment option and could be used in place of UV therapy during deployment along with topical medications. This patient opted to continue treatment with ustekinumab, resulting in MEB review and separation from military service.
Dermatology Treatment Considerations
Civilian dermatologists should be aware of specific considerations when treating active US service members with common cutaneous diagnoses such as acne, atopic dermatitis (AD), psoriasis, dissecting cellulitis of the scalp (DCS), and lupus erythematosus (LE). This discussion is not meant to be all-inclusive but provides information and examples related to common treatment challenges in this patient population.
Acne
Acne is common in the active-duty military population. Typically, acne should be treated per recommended guidelines based on type and severity.8 Medical evaluation board review is warranted in cases of severe acne that is unresponsive to treatment and interferes with a service member’s performance.5,6 Unique situations in the active-duty military population include the following:
• Use of minocycline. Aircrew members have unique restrictions on many medications,6 including minocycline, which is restricted in this population due to vestibular side effects. Doxycycline is an acceptable alternative for aircrew members; however, even this medication may require a ground trial to ensure there are no idiosyncratic effects.
• Use of isotretinoin, which is not permitted in aircrew members, submariners, or divers. If they take this medication, they will be temporarily removed from duty for the duration of treatment and for a period of time after completion (1–3 months, depending on service). Isotretinoin also is not used during deployment due to potential side effects, the need for laboratory monitoring, and iPLEDGE system requirements.
Atopic Dermatitis
A history of AD after the 12th birthday is considered a disqualifying condition with regard to military service,3 though mild and well-controlled disease can easily be overlooked during entrance physical examinations. Members frequently present with eczema flares following field training exercises where they are outdoors for many hours and have been exposed to grass or other environmental triggers while wearing military gear that is heavy and occlusive, which is further exacerbated by being unable to bathe or care for their skin as they would at home.
Separation from the military is considered when AD is moderate to severe, is unresponsive to treatment, and/or interferes with performance of duty. Severity often can be evaluated based on the impact of AD on performance of duties in addition to clinical appearance. A pilot who is distracted by itching presents a potentially dangerous situation. A soldier whose AD flares every time he/she goes to the field, requiring him/her to return home early to control symptoms, can be considered moderate to severe due to lack of ability to do his/her job away from home base.
Response to treatment is more often where trouble lies for military members with AD, as patients are only permitted to take emollients, preferred cleansers, and topical medications to field training exercises and deployments. UV therapy is used to control disease in the military population but is not an option in deployed environments. Classic immunosuppressants (eg, methotrexate, mycophenolate mofetil, azathioprine, cyclosporine) may result in a good response to treatment; however, due to their side-effect profiles, need for laboratory monitoring, and immunosuppressive nature, long-term use of those medications will result in a nondeployable status. Dupilumab does not appear to have the immunosuppressive effects of other biologics; however, the medication requires refrigeration,9 which currently precludes its use in the deployed environment, as it would be difficult to ensure supply and storage in remote areas.
Service members with a history of AD are exempt from the smallpox vaccine due to concerns about eczema vaccinatum.10
Psoriasis
Psoriasis is another dermatologic condition that does not meet military admission standards,3 and mild undiagnosed cases may be overlooked during the entrance physical examination. Because psoriasis commonly affects young adults, it may manifest in service members after entering service. If psoriasis is extensive or refractory to treatment, an MEB evaluation may be required.5,6 Widespread psoriasis can result in considerable discomfort when wearing body armor and other military gear. Severe localized disease can have duty implications; service members with treatment-resistant scalp psoriasis or pustular psoriasis of the feet may have difficulty wearing helmets or military boots, respectively.
Most service members with limited psoriasis vulgaris can be managed with topical steroids and steroid-sparing agents such as calcipotriene. Some service members opt not to aggressively treat their psoriasis if it is limited in nature and not symptomatic.
When discussing systemic treatments beyond light therapy in those with refractory disease, apremilast can be a good first-line treatment option.11 It is an oral medication, has minimal monitoring requirements, and lacks immunosuppressive side effects; therefore, it does not adversely impact deployability. If patients do not improve in 4 months with apremilast, biologics should then be considered; however, biologics have service implications, the most important being inability to deploy while taking the medication. In rare circumstances, military dermatologists may discuss utilizing biologic therapy only in the nondeployed setting. In these cases, service members are counseled that biologic therapy will be discontinued if they deploy in the future and treatment will be sustained with topicals and/or apremilast through the deployment. The treatment plan also should be communicated to the patient’s primary care provider to ensure that he/she is in agreement.
Dissecting Cellulitis of the Scalp
Dissecting cellulitis of the scalp may result in separation if the condition is unresponsive to treatment and/or interferes with satisfactory performance of duty.5 In addition to causing considerable pain, this condition can prevent service members from wearing combat helmets, which limits their ability to train and deploy. One of the authors (S.C.) has had more service members undergo an MEB evaluation for DCS than any of the other conditions mentioned.
Topical tretinoin and topical antibiotics can be used in conjunction with either doxycycline or minocycline to treat DCS, with the addition of intralesional corticosteroids for painful nodules. Fluctuant lesions are treated with incision and drainage. If there is inadequate response to treatment after 2 to 3 months, oral clindamycin and rifampin can be tried for 3 months. As an alternative measure or if the condition is refractory to oral clindamycin and rifampin, isotretinoin can then be used. One of the authors (S.C.) typically recommends a temporary no-helmet profile to the patient’s primary care provider until his/her next dermatology appointment. If the patient still has substantial disease despite these treatment options, it is recommended that the patient be issued a permanent profile for no helmet wear, which will prompt an MEB evaluation. Although tumor necrosis factor α inhibitors can work well in patients with DCS, the use of biologics is not conducive to continued service.
Lupus Erythematosus
A history of LE is disqualifying from military service. Patients who develop LE while on active duty will be referred for MEB evaluation if their disease is unresponsive to treatment and/or interferes with the satisfactory performance of duty.5,6 In general, connective tissue diseases have an array of physical implications that can affect military service, including photosensitivity, joint inflammation, and internal organ involvement. Similar to the other dermatologic conditions described, treatment of connective tissue diseases also can present challenges to continued military service. Considerations in the case of LE that are unique to military service members include the following:
• Sun exposure. Most military service members are required to work outside in all manners of conditions, which include hot, sunny, humid, and/or dry climates. Often physicians might counsel sun-sensitive patients with LE to avoid being outside during daylight hours, limit window exposure at work, and avoid daytime driving when possible; however, these recommendations are not possible for many, if not most, service members.
• Immunosuppressive therapies are incompatible with military deployment; therefore, prescribing methotrexate, cyclosporine, mycophenolate mofetil, rituximab, or belimumab for treatment of LE would prompt an MEB evaluation if the treatment is necessary to control the disease.
Final Thoughts
The recent changes to military medicine are needed to meet our country’s defense requirements and will ultimately result in civilian specialists playing a larger role in the care of our military population. This article highlights unique factors civilian dermatologists must consider when treating active-duty military patients to ensure they remain deployable during treatment.
- National Defense Authorization Act for Fiscal Year 2017, S 2943, 114th Congress, 2nd Sess (2016).
- Garamone J. Dunford sends message to joint force, stresses readiness, warfighting, education [news release]. Washington, DC: US Department of Defense; October 2, 2015. https://dod.defense.gov/News/Article/Article/621725/dunford-sends-message-to-joint-force-stresses-readiness-warfighting-education/. Accessed May 17, 2019.
- Medical Standards for Appointment, Enlistment, or Induction Into the Military Services (DoD Instruction 6130.03). Washington, DC: Department of Defense; March 30, 2018. https://www.esd.whs.mil/Portals/54/Documents/DD/issuances/dodi/613003p.pdf?ver=2018-05-04-113917-883. Accessed May 17, 2019.
- Force health protection guidance for deployment in USSOUTHCOM as of 7 December 2017. US Southern Command website. https://www.southcom.mil/Portals/7/Documents/Operational%20Contract%20Support/USSOUTHCOM_Force_Health_Protection_Guidance_AS_OF_7_DEC_2017.pdf?ver=2018-01-29-100603-957. Published December 7, 2017. Accessed May 28, 2019.
- US Department of the Army. Standards of medical fitness. http://www.au.af.mil/au/awc/awcgate/army/r40_501.pdf. Published August 26, 2003. Accessed May 17, 2019.
- US Department of the Air Force. Medical examinations and standards. https://static.e-publishing.af.mil/production/1/af_sg/publication/afi48-123/afi48-123.pdf. Published November 5, 2013. Accessed May 17, 2019.
- Medical and physical evaluation boards (MEB/PEB). US Army Warrior Care and Transition website. https://wct.army.mil/modules/soldier/s6-medicalBoards.html. Accessed May 28, 2019.
- Zaenglein AL, Pathy AL, Schlosser BJ, et al. Guidelines of care for the management of acne vulgaris. J Am Acad Dermatol. 2016;74:945-973.
- Dupixent [package insert]. Tarrytown, NY: Regeneron, Inc; 2017.
- Departments of the Army, the Navy, the Air Force, and the Coast Guard. Immunizations and chemoprophylaxis for the prevention of infectious diseases. https://health.mil/Reference-Center/Policies/2013/10/07/Immunizations-and-Chemoprophylaxis-for-the-Prevention-of-Infectious-Diseases. Published October 7, 2013. Accessed May 28, 2019.
- Rosenberg A, Meyerle J. The use of apremilast to treat psoriasis during deployment. Mil Med. 2017;182:1628-1631.
- National Defense Authorization Act for Fiscal Year 2017, S 2943, 114th Congress, 2nd Sess (2016).
- Garamone J. Dunford sends message to joint force, stresses readiness, warfighting, education [news release]. Washington, DC: US Department of Defense; October 2, 2015. https://dod.defense.gov/News/Article/Article/621725/dunford-sends-message-to-joint-force-stresses-readiness-warfighting-education/. Accessed May 17, 2019.
- Medical Standards for Appointment, Enlistment, or Induction Into the Military Services (DoD Instruction 6130.03). Washington, DC: Department of Defense; March 30, 2018. https://www.esd.whs.mil/Portals/54/Documents/DD/issuances/dodi/613003p.pdf?ver=2018-05-04-113917-883. Accessed May 17, 2019.
- Force health protection guidance for deployment in USSOUTHCOM as of 7 December 2017. US Southern Command website. https://www.southcom.mil/Portals/7/Documents/Operational%20Contract%20Support/USSOUTHCOM_Force_Health_Protection_Guidance_AS_OF_7_DEC_2017.pdf?ver=2018-01-29-100603-957. Published December 7, 2017. Accessed May 28, 2019.
- US Department of the Army. Standards of medical fitness. http://www.au.af.mil/au/awc/awcgate/army/r40_501.pdf. Published August 26, 2003. Accessed May 17, 2019.
- US Department of the Air Force. Medical examinations and standards. https://static.e-publishing.af.mil/production/1/af_sg/publication/afi48-123/afi48-123.pdf. Published November 5, 2013. Accessed May 17, 2019.
- Medical and physical evaluation boards (MEB/PEB). US Army Warrior Care and Transition website. https://wct.army.mil/modules/soldier/s6-medicalBoards.html. Accessed May 28, 2019.
- Zaenglein AL, Pathy AL, Schlosser BJ, et al. Guidelines of care for the management of acne vulgaris. J Am Acad Dermatol. 2016;74:945-973.
- Dupixent [package insert]. Tarrytown, NY: Regeneron, Inc; 2017.
- Departments of the Army, the Navy, the Air Force, and the Coast Guard. Immunizations and chemoprophylaxis for the prevention of infectious diseases. https://health.mil/Reference-Center/Policies/2013/10/07/Immunizations-and-Chemoprophylaxis-for-the-Prevention-of-Infectious-Diseases. Published October 7, 2013. Accessed May 28, 2019.
- Rosenberg A, Meyerle J. The use of apremilast to treat psoriasis during deployment. Mil Med. 2017;182:1628-1631.
Practice Points
- Certain conditions and treatments are incompatible with military service and may result in separation.
- Dermatologists must consider a patient’s profession when choosing a treatment modality.
Acetyl-coenzyme-A carboxylase inhibition shows early promise for acne vulgaris
A potent oral inhibitor of acetyl-coenzyme-A carboxylase approximately halved the production of facial sebum, most of which arises from de novo lipogenesis, researchers reported.
The production of sebum triglycerides, wax esters, and free fatty acids all depend on local flux through this de novo lipogenesis (DNL) pathway in sebocytes, explained William P. Esler, PhD, and associates. Oral treatment with the investigational agent PF-05175157, a potent inhibitor of acetyl-coenzyme-A carboxylase (ACC) 1 and 2, reduced levels of these sebum components by about 66%, but did not affect levels of compounds that do not depend on the DNL pathway. The results of their mechanistic studies “identify sebocyte DNL as a pathway of importance in the biology of human skin and in the pathogenesis of acne vulgaris,” the researchers wrote in Science Translational Medicine. “Moreover, the observed dependence of human sebum production on local DNL flux and the effectiveness of DNL inhibition by an
Sebum helps moisturize and protect human skin, but increased production is linked to acne vulgaris severity. While sebaceous glands contain ACC and undergo DNL, the role of this pathway in sebum production relative to the recycling of circulating lipids was unknown. For the study, Dr. Esler of Pfizer Global Research and Development in Cambridge, Mass., and associates administered heavy water to 22 healthy volunteers to measure how much stable isotope was incorporated into newly synthesized fatty acids in sebocytes. They found that most skin sebum originated from local flux through the DNL pathway, including 80% of sebum palmitate and more than 80% of sebum sapienate. Furthermore, compared with 10 individuals with acne-free skin, 9 patients with acne vulgaris had about 20% greater sebum production and DNL pathway flux.
Oral therapy for 2 weeks with the ACC inhibitor (200 mg twice daily) was well tolerated and reduced baseline sebum production by 49% when administered to 10 healthy volunteers, the investigators wrote. This effect was not observed in a small placebo comparator group. Importantly, studies of hamsters and guinea pigs failed to implicate the DNL pathway in sebum production, even though these animals have been widely used to model sebum production in humans.
The researchers recommended studying the effects of ACC inhibition in patients with acne vulgaris, the effects of topical ACC inhibition on sebum production, and whether DNL pathway inhibition reduces the number and severity of acne lesions.
Pfizer provided funding; Dr. Esler and 12 coinvestigators are Pfizer employees and stockholders. Two additional coinvestigators are former Pfizer employees while three are current or prior scientific consultants for Pfizer.
SOURCE: Esler WP et al. Sci Transl Med. 2019 May 15. doi: 10.1126/scitranslmed.aau8465.
A potent oral inhibitor of acetyl-coenzyme-A carboxylase approximately halved the production of facial sebum, most of which arises from de novo lipogenesis, researchers reported.
The production of sebum triglycerides, wax esters, and free fatty acids all depend on local flux through this de novo lipogenesis (DNL) pathway in sebocytes, explained William P. Esler, PhD, and associates. Oral treatment with the investigational agent PF-05175157, a potent inhibitor of acetyl-coenzyme-A carboxylase (ACC) 1 and 2, reduced levels of these sebum components by about 66%, but did not affect levels of compounds that do not depend on the DNL pathway. The results of their mechanistic studies “identify sebocyte DNL as a pathway of importance in the biology of human skin and in the pathogenesis of acne vulgaris,” the researchers wrote in Science Translational Medicine. “Moreover, the observed dependence of human sebum production on local DNL flux and the effectiveness of DNL inhibition by an
Sebum helps moisturize and protect human skin, but increased production is linked to acne vulgaris severity. While sebaceous glands contain ACC and undergo DNL, the role of this pathway in sebum production relative to the recycling of circulating lipids was unknown. For the study, Dr. Esler of Pfizer Global Research and Development in Cambridge, Mass., and associates administered heavy water to 22 healthy volunteers to measure how much stable isotope was incorporated into newly synthesized fatty acids in sebocytes. They found that most skin sebum originated from local flux through the DNL pathway, including 80% of sebum palmitate and more than 80% of sebum sapienate. Furthermore, compared with 10 individuals with acne-free skin, 9 patients with acne vulgaris had about 20% greater sebum production and DNL pathway flux.
Oral therapy for 2 weeks with the ACC inhibitor (200 mg twice daily) was well tolerated and reduced baseline sebum production by 49% when administered to 10 healthy volunteers, the investigators wrote. This effect was not observed in a small placebo comparator group. Importantly, studies of hamsters and guinea pigs failed to implicate the DNL pathway in sebum production, even though these animals have been widely used to model sebum production in humans.
The researchers recommended studying the effects of ACC inhibition in patients with acne vulgaris, the effects of topical ACC inhibition on sebum production, and whether DNL pathway inhibition reduces the number and severity of acne lesions.
Pfizer provided funding; Dr. Esler and 12 coinvestigators are Pfizer employees and stockholders. Two additional coinvestigators are former Pfizer employees while three are current or prior scientific consultants for Pfizer.
SOURCE: Esler WP et al. Sci Transl Med. 2019 May 15. doi: 10.1126/scitranslmed.aau8465.
A potent oral inhibitor of acetyl-coenzyme-A carboxylase approximately halved the production of facial sebum, most of which arises from de novo lipogenesis, researchers reported.
The production of sebum triglycerides, wax esters, and free fatty acids all depend on local flux through this de novo lipogenesis (DNL) pathway in sebocytes, explained William P. Esler, PhD, and associates. Oral treatment with the investigational agent PF-05175157, a potent inhibitor of acetyl-coenzyme-A carboxylase (ACC) 1 and 2, reduced levels of these sebum components by about 66%, but did not affect levels of compounds that do not depend on the DNL pathway. The results of their mechanistic studies “identify sebocyte DNL as a pathway of importance in the biology of human skin and in the pathogenesis of acne vulgaris,” the researchers wrote in Science Translational Medicine. “Moreover, the observed dependence of human sebum production on local DNL flux and the effectiveness of DNL inhibition by an
Sebum helps moisturize and protect human skin, but increased production is linked to acne vulgaris severity. While sebaceous glands contain ACC and undergo DNL, the role of this pathway in sebum production relative to the recycling of circulating lipids was unknown. For the study, Dr. Esler of Pfizer Global Research and Development in Cambridge, Mass., and associates administered heavy water to 22 healthy volunteers to measure how much stable isotope was incorporated into newly synthesized fatty acids in sebocytes. They found that most skin sebum originated from local flux through the DNL pathway, including 80% of sebum palmitate and more than 80% of sebum sapienate. Furthermore, compared with 10 individuals with acne-free skin, 9 patients with acne vulgaris had about 20% greater sebum production and DNL pathway flux.
Oral therapy for 2 weeks with the ACC inhibitor (200 mg twice daily) was well tolerated and reduced baseline sebum production by 49% when administered to 10 healthy volunteers, the investigators wrote. This effect was not observed in a small placebo comparator group. Importantly, studies of hamsters and guinea pigs failed to implicate the DNL pathway in sebum production, even though these animals have been widely used to model sebum production in humans.
The researchers recommended studying the effects of ACC inhibition in patients with acne vulgaris, the effects of topical ACC inhibition on sebum production, and whether DNL pathway inhibition reduces the number and severity of acne lesions.
Pfizer provided funding; Dr. Esler and 12 coinvestigators are Pfizer employees and stockholders. Two additional coinvestigators are former Pfizer employees while three are current or prior scientific consultants for Pfizer.
SOURCE: Esler WP et al. Sci Transl Med. 2019 May 15. doi: 10.1126/scitranslmed.aau8465.
FROM SCIENCE TRANSLATIONAL MEDICINE